User login
Elderly Americans carry heavier opioid burden
according to the Agency for Healthcare Quality and Research.
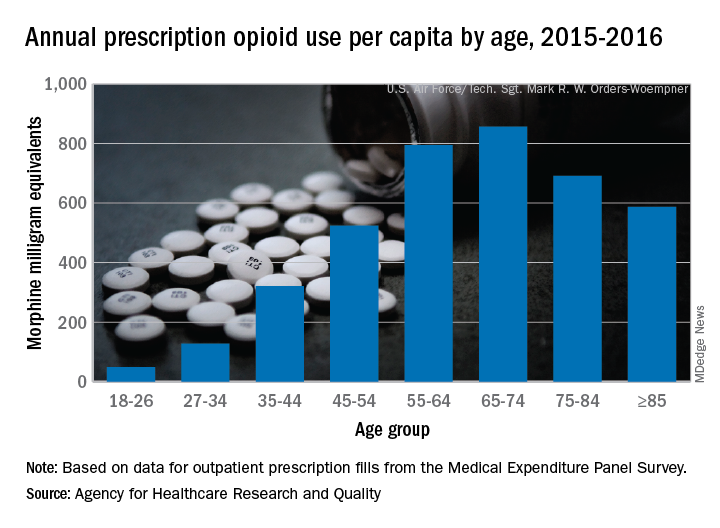
Elderly adults with chronic and acute pain obtained an average of 774 morphine milligram equivalents (MMEs) of prescription opioids annually during 2015-2016 from outpatient clinicians, compared with 376 MMEs a year for nonelderly adults, said Asako S. Moriya, PhD, and G. Edward Miller, PhD, of the AHRQ.
Narrowing the age groups shows that opioid MMEs increased with age, starting at 49 MMEs for 18- to 26-year-olds and rising to a high of 856 MMEs in the 65- to 74-year-old group, before dropping off in the oldest adults, the investigators said in a Medical Expenditure Panel Survey (MEPS) research findings report.
The analysis included “all opioid medications that are commonly used to treat pain” and excluded respiratory agents, antitussives, and drugs used for medication-assisted treatment, they noted. The MEPS data cover prescriptions purchased or obtained in outpatient settings but not those administered in inpatient settings or in clinics or physician offices.
according to the Agency for Healthcare Quality and Research.

Elderly adults with chronic and acute pain obtained an average of 774 morphine milligram equivalents (MMEs) of prescription opioids annually during 2015-2016 from outpatient clinicians, compared with 376 MMEs a year for nonelderly adults, said Asako S. Moriya, PhD, and G. Edward Miller, PhD, of the AHRQ.
Narrowing the age groups shows that opioid MMEs increased with age, starting at 49 MMEs for 18- to 26-year-olds and rising to a high of 856 MMEs in the 65- to 74-year-old group, before dropping off in the oldest adults, the investigators said in a Medical Expenditure Panel Survey (MEPS) research findings report.
The analysis included “all opioid medications that are commonly used to treat pain” and excluded respiratory agents, antitussives, and drugs used for medication-assisted treatment, they noted. The MEPS data cover prescriptions purchased or obtained in outpatient settings but not those administered in inpatient settings or in clinics or physician offices.
according to the Agency for Healthcare Quality and Research.

Elderly adults with chronic and acute pain obtained an average of 774 morphine milligram equivalents (MMEs) of prescription opioids annually during 2015-2016 from outpatient clinicians, compared with 376 MMEs a year for nonelderly adults, said Asako S. Moriya, PhD, and G. Edward Miller, PhD, of the AHRQ.
Narrowing the age groups shows that opioid MMEs increased with age, starting at 49 MMEs for 18- to 26-year-olds and rising to a high of 856 MMEs in the 65- to 74-year-old group, before dropping off in the oldest adults, the investigators said in a Medical Expenditure Panel Survey (MEPS) research findings report.
The analysis included “all opioid medications that are commonly used to treat pain” and excluded respiratory agents, antitussives, and drugs used for medication-assisted treatment, they noted. The MEPS data cover prescriptions purchased or obtained in outpatient settings but not those administered in inpatient settings or in clinics or physician offices.
FDA approves ozanimod for relapsing and secondary progressive forms of MS
The Food and Drug Administration has approved the oral medication ozanimod (Zeposia) for relapsing forms of multiple sclerosis (MS), including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, according to a release from Bristol-Myers Squibb.
Ozanimod is a sphingosine 1-phosphate (S1P) receptor modulator that binds with high affinity to S1P receptors 1 and 5. It blocks the capacity of lymphocytes to egress from lymph nodes, reducing the number of lymphocytes in peripheral blood. Although its therapeutic mechanism of action in MS is unknown, it may involve the reduction of lymphocyte migration into the central nervous system. A genetic test is not required to start the drug, and no patient observation is required for the first dose, although up-titration of initial doses are required to reach the maintenance dose because a transient decrease in heart rate and atrioventricular conduction delays may occur, according to the company.
The approval is based on a pair of head-to-head studies that compared it with interferon beta-1a (Avonex) and together included more than 2,600 patients. It delivered better efficacy in terms of relative reduction in annualized relapse rate (48% at 1 year and 38% at 2 years). It also demonstrated better relative reduction of the number of T1-weighted gadolinium-enhanced brain lesions (63% fewer at 1 year and 53% fewer at 2 years) and number of new or enlarging T2 lesions (48% fewer at 1 year and 42% at 2 years).
Ozanimod is contraindicated in patients who, in the past 6 months, experienced a myocardial infarction, unstable angina, stroke, or other conditions. It is associated with other health risks, including infections, liver injury, additive immunosuppressive effects from prior immune-modulating therapies, and increased blood pressure. Certain assessments, such as recent complete blood count, ECG, liver function test, and current and prior medications and vaccinations, are required before initiation of treatment.
In its announcement, Bristol-Myers Squibb said that it has decided to delay the commercial launch of ozanimod during the COVID-19 pandemic until a later date.
The drug is also in development for additional immune-inflammatory indications, including ulcerative colitis and Crohn’s disease.
The full prescribing information can be found on the company’s website.
The Food and Drug Administration has approved the oral medication ozanimod (Zeposia) for relapsing forms of multiple sclerosis (MS), including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, according to a release from Bristol-Myers Squibb.
Ozanimod is a sphingosine 1-phosphate (S1P) receptor modulator that binds with high affinity to S1P receptors 1 and 5. It blocks the capacity of lymphocytes to egress from lymph nodes, reducing the number of lymphocytes in peripheral blood. Although its therapeutic mechanism of action in MS is unknown, it may involve the reduction of lymphocyte migration into the central nervous system. A genetic test is not required to start the drug, and no patient observation is required for the first dose, although up-titration of initial doses are required to reach the maintenance dose because a transient decrease in heart rate and atrioventricular conduction delays may occur, according to the company.
The approval is based on a pair of head-to-head studies that compared it with interferon beta-1a (Avonex) and together included more than 2,600 patients. It delivered better efficacy in terms of relative reduction in annualized relapse rate (48% at 1 year and 38% at 2 years). It also demonstrated better relative reduction of the number of T1-weighted gadolinium-enhanced brain lesions (63% fewer at 1 year and 53% fewer at 2 years) and number of new or enlarging T2 lesions (48% fewer at 1 year and 42% at 2 years).
Ozanimod is contraindicated in patients who, in the past 6 months, experienced a myocardial infarction, unstable angina, stroke, or other conditions. It is associated with other health risks, including infections, liver injury, additive immunosuppressive effects from prior immune-modulating therapies, and increased blood pressure. Certain assessments, such as recent complete blood count, ECG, liver function test, and current and prior medications and vaccinations, are required before initiation of treatment.
In its announcement, Bristol-Myers Squibb said that it has decided to delay the commercial launch of ozanimod during the COVID-19 pandemic until a later date.
The drug is also in development for additional immune-inflammatory indications, including ulcerative colitis and Crohn’s disease.
The full prescribing information can be found on the company’s website.
The Food and Drug Administration has approved the oral medication ozanimod (Zeposia) for relapsing forms of multiple sclerosis (MS), including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, according to a release from Bristol-Myers Squibb.
Ozanimod is a sphingosine 1-phosphate (S1P) receptor modulator that binds with high affinity to S1P receptors 1 and 5. It blocks the capacity of lymphocytes to egress from lymph nodes, reducing the number of lymphocytes in peripheral blood. Although its therapeutic mechanism of action in MS is unknown, it may involve the reduction of lymphocyte migration into the central nervous system. A genetic test is not required to start the drug, and no patient observation is required for the first dose, although up-titration of initial doses are required to reach the maintenance dose because a transient decrease in heart rate and atrioventricular conduction delays may occur, according to the company.
The approval is based on a pair of head-to-head studies that compared it with interferon beta-1a (Avonex) and together included more than 2,600 patients. It delivered better efficacy in terms of relative reduction in annualized relapse rate (48% at 1 year and 38% at 2 years). It also demonstrated better relative reduction of the number of T1-weighted gadolinium-enhanced brain lesions (63% fewer at 1 year and 53% fewer at 2 years) and number of new or enlarging T2 lesions (48% fewer at 1 year and 42% at 2 years).
Ozanimod is contraindicated in patients who, in the past 6 months, experienced a myocardial infarction, unstable angina, stroke, or other conditions. It is associated with other health risks, including infections, liver injury, additive immunosuppressive effects from prior immune-modulating therapies, and increased blood pressure. Certain assessments, such as recent complete blood count, ECG, liver function test, and current and prior medications and vaccinations, are required before initiation of treatment.
In its announcement, Bristol-Myers Squibb said that it has decided to delay the commercial launch of ozanimod during the COVID-19 pandemic until a later date.
The drug is also in development for additional immune-inflammatory indications, including ulcerative colitis and Crohn’s disease.
The full prescribing information can be found on the company’s website.
Due to the COVID-19 pandemic, the AAN urges feds to further expand telehealth benefits
On March 17, the Trump administration announced an expansion of telehealth benefits to help stop the spread of COVID-19 and allow more Medicare patients to receive virtual care without having to visit a healthcare center or physician office.
Under the expansion, Medicare will pay for office, hospital, and other visits furnished via telehealth across the country and including in the patient’s home, delivered by a range of providers, such as physicians, nurse practitioners, clinical psychologists, and licensed clinical social workers.
Prior to this waiver, Medicare would only pay for telehealth on a limited basis, such as when the patient receiving the service was in a designated rural area.
However, in a letter to Alex Azar, secretary of the U.S. Department of Health & Human Services (HHS), the AAN says the easing of restrictions on telehealth should be extended beyond Medicare fee-for-service to both Medicare Advantage and Medicaid patients.
Practice changing?
“It is very heartening that the government is stepping up to the plate” and lifting many telemedicine restrictions, Neil Busis, MD, member of the AAN Health Policy Subcommittee, said in an interview.
Dr. Busis, who leads the telemedicine program for the department of neurology at NYU Langone Health in New York, said the global pandemic has “heightened, focused, and sharpened” attention to the need for telehealth services, particularly for neurology.
“By definition, a lot of neurology patients have mobility problems, traveling is a burden, making it difficult to see a neurologist,” he said.
Dr. Busis hopes these waivers in telehealth, made on a temporary and emergency basis, will become permanent once the COVID-19 pandemic has passed.
“What we hope is that the usefulness of various virtual technologies tested in the crucible of this pandemic will stimulate people to think about it once the pandemic is over and not rescind these loosening of restrictions, and that this will be the beginning of a new era for telemedicine,” he said.
The COVID-19 pandemic may be a “catalyst to accelerate the incorporation of non-face-to-face care into our armamentarium,” he added.
“What we have discovered in recent years is non-face-to-face care with enabling communication technologies is as effective in many clinical situations as face-to-face care. Now is the time to really focus on making the virtual experience as good as possible and to make it as available to as many people as possible,” said Dr. Busis.
Reduce regulatory burdens
The AAN also calls on the federal government to urge states to take action to ensure access to telehealth services and allow telehealth companies to provide telehealth technology and education free of charge to providers who don’t currently use telehealth in their practices.
“The AAN notes that doing so may implicate provisions of the Anti-Kickback Statute. We believe during the current emergency that HHS should issue guidance making it clear to providers that accepting free access to telehealth platforms and education does not put them at risk of violating fraud and abuse laws,” the letter, signed by AAN President James Stevens, MD, stated.
The AAN also wants the government to reduce regulatory burdens during this public health emergency to allow physicians more time to focus on patient care. “This is especially true for providers that are self-quarantining or are in a practice that is experiencing staffing shortages due to self-quarantines,” he wrote.
Specifically, the AAN asked the Centers for Medicare & Medicaid Services to extend the March 31 deadline for physicians to submit their data for the Merit-based Incentive Payment System program for calendar year 2019 (and other compliance deadlines) by at least 30 days.
The AAN also calls on the CMS to delay implementation of the Appropriate Use Criteria program by 1 year, saying that many providers will not have the capacity to “meaningfully” participate in the current testing year for the program.
A version of this article originally appeared on Medscape.com.
On March 17, the Trump administration announced an expansion of telehealth benefits to help stop the spread of COVID-19 and allow more Medicare patients to receive virtual care without having to visit a healthcare center or physician office.
Under the expansion, Medicare will pay for office, hospital, and other visits furnished via telehealth across the country and including in the patient’s home, delivered by a range of providers, such as physicians, nurse practitioners, clinical psychologists, and licensed clinical social workers.
Prior to this waiver, Medicare would only pay for telehealth on a limited basis, such as when the patient receiving the service was in a designated rural area.
However, in a letter to Alex Azar, secretary of the U.S. Department of Health & Human Services (HHS), the AAN says the easing of restrictions on telehealth should be extended beyond Medicare fee-for-service to both Medicare Advantage and Medicaid patients.
Practice changing?
“It is very heartening that the government is stepping up to the plate” and lifting many telemedicine restrictions, Neil Busis, MD, member of the AAN Health Policy Subcommittee, said in an interview.
Dr. Busis, who leads the telemedicine program for the department of neurology at NYU Langone Health in New York, said the global pandemic has “heightened, focused, and sharpened” attention to the need for telehealth services, particularly for neurology.
“By definition, a lot of neurology patients have mobility problems, traveling is a burden, making it difficult to see a neurologist,” he said.
Dr. Busis hopes these waivers in telehealth, made on a temporary and emergency basis, will become permanent once the COVID-19 pandemic has passed.
“What we hope is that the usefulness of various virtual technologies tested in the crucible of this pandemic will stimulate people to think about it once the pandemic is over and not rescind these loosening of restrictions, and that this will be the beginning of a new era for telemedicine,” he said.
The COVID-19 pandemic may be a “catalyst to accelerate the incorporation of non-face-to-face care into our armamentarium,” he added.
“What we have discovered in recent years is non-face-to-face care with enabling communication technologies is as effective in many clinical situations as face-to-face care. Now is the time to really focus on making the virtual experience as good as possible and to make it as available to as many people as possible,” said Dr. Busis.
Reduce regulatory burdens
The AAN also calls on the federal government to urge states to take action to ensure access to telehealth services and allow telehealth companies to provide telehealth technology and education free of charge to providers who don’t currently use telehealth in their practices.
“The AAN notes that doing so may implicate provisions of the Anti-Kickback Statute. We believe during the current emergency that HHS should issue guidance making it clear to providers that accepting free access to telehealth platforms and education does not put them at risk of violating fraud and abuse laws,” the letter, signed by AAN President James Stevens, MD, stated.
The AAN also wants the government to reduce regulatory burdens during this public health emergency to allow physicians more time to focus on patient care. “This is especially true for providers that are self-quarantining or are in a practice that is experiencing staffing shortages due to self-quarantines,” he wrote.
Specifically, the AAN asked the Centers for Medicare & Medicaid Services to extend the March 31 deadline for physicians to submit their data for the Merit-based Incentive Payment System program for calendar year 2019 (and other compliance deadlines) by at least 30 days.
The AAN also calls on the CMS to delay implementation of the Appropriate Use Criteria program by 1 year, saying that many providers will not have the capacity to “meaningfully” participate in the current testing year for the program.
A version of this article originally appeared on Medscape.com.
On March 17, the Trump administration announced an expansion of telehealth benefits to help stop the spread of COVID-19 and allow more Medicare patients to receive virtual care without having to visit a healthcare center or physician office.
Under the expansion, Medicare will pay for office, hospital, and other visits furnished via telehealth across the country and including in the patient’s home, delivered by a range of providers, such as physicians, nurse practitioners, clinical psychologists, and licensed clinical social workers.
Prior to this waiver, Medicare would only pay for telehealth on a limited basis, such as when the patient receiving the service was in a designated rural area.
However, in a letter to Alex Azar, secretary of the U.S. Department of Health & Human Services (HHS), the AAN says the easing of restrictions on telehealth should be extended beyond Medicare fee-for-service to both Medicare Advantage and Medicaid patients.
Practice changing?
“It is very heartening that the government is stepping up to the plate” and lifting many telemedicine restrictions, Neil Busis, MD, member of the AAN Health Policy Subcommittee, said in an interview.
Dr. Busis, who leads the telemedicine program for the department of neurology at NYU Langone Health in New York, said the global pandemic has “heightened, focused, and sharpened” attention to the need for telehealth services, particularly for neurology.
“By definition, a lot of neurology patients have mobility problems, traveling is a burden, making it difficult to see a neurologist,” he said.
Dr. Busis hopes these waivers in telehealth, made on a temporary and emergency basis, will become permanent once the COVID-19 pandemic has passed.
“What we hope is that the usefulness of various virtual technologies tested in the crucible of this pandemic will stimulate people to think about it once the pandemic is over and not rescind these loosening of restrictions, and that this will be the beginning of a new era for telemedicine,” he said.
The COVID-19 pandemic may be a “catalyst to accelerate the incorporation of non-face-to-face care into our armamentarium,” he added.
“What we have discovered in recent years is non-face-to-face care with enabling communication technologies is as effective in many clinical situations as face-to-face care. Now is the time to really focus on making the virtual experience as good as possible and to make it as available to as many people as possible,” said Dr. Busis.
Reduce regulatory burdens
The AAN also calls on the federal government to urge states to take action to ensure access to telehealth services and allow telehealth companies to provide telehealth technology and education free of charge to providers who don’t currently use telehealth in their practices.
“The AAN notes that doing so may implicate provisions of the Anti-Kickback Statute. We believe during the current emergency that HHS should issue guidance making it clear to providers that accepting free access to telehealth platforms and education does not put them at risk of violating fraud and abuse laws,” the letter, signed by AAN President James Stevens, MD, stated.
The AAN also wants the government to reduce regulatory burdens during this public health emergency to allow physicians more time to focus on patient care. “This is especially true for providers that are self-quarantining or are in a practice that is experiencing staffing shortages due to self-quarantines,” he wrote.
Specifically, the AAN asked the Centers for Medicare & Medicaid Services to extend the March 31 deadline for physicians to submit their data for the Merit-based Incentive Payment System program for calendar year 2019 (and other compliance deadlines) by at least 30 days.
The AAN also calls on the CMS to delay implementation of the Appropriate Use Criteria program by 1 year, saying that many providers will not have the capacity to “meaningfully” participate in the current testing year for the program.
A version of this article originally appeared on Medscape.com.
Sleep-disordered breathing linked with Alzheimer’s disease biomarkers in cognitively normal older adults
investigators have found.
Among 127 adults enrolled in a randomized clinical trial of interventions to promote mental well-being in older adults, those with sleep-disordered breathing had significantly greater amyloid burden and gray-matter volume, as well as increased perfusion and metabolism in parietal-occipital regions, reported Claire André, PhD, from the French Institute of Health and Medical Research (INSERM) unit in Caen, and colleagues.
“Our findings highlight the need to treat sleep disorders in the older population, even in the absence of cognitive or behavioral manifestations,” they wrote in a study published in JAMA Neurology.
Previous studies of the possible association between sleep-disordered breathing and dementia risk have shown conflicting or inconsistent results, the authors noted.
“These discrepancies may be explained by the characteristics of patients with sleep-disordered breathing (e.g., recruited from sleep clinics versus from the community, differences in age and disease duration), the scoring criteria of respiratory events, sample sizes, or the lack of controls for possibly biasing covariates,” they wrote.
To see whether they could clear up the confusion, the investigators conducted a retrospective analysis of 127 patients who were enrolled in the Age-Well randomized, controlled trial of the Medit-Ageing European project. The participants were community-dwelling adults (mean age, 69.1 years; 63% women), who were enrolled in the trial and underwent evaluation from 2016 to 2018 at the Cyceron Cancer Center in Caen.
The participants, all of whom were cognitively unimpaired at baseline, underwent neuropsychological assessment, polysomnography, MRI, plus florbetapir- and fluorodeoxyglucose-labeled PET.
The investigators defined sleep-disordered breathing as 15 apnea-hypopnea index events per hour or higher, and compared results between those with sleep-disordered breathing and those without for each imaging modality.
Participants with sleep-disordered breathing has significantly greater amyloid burden (P = .04), gray-matter volume (P = .04), perfusion (P = .04), and metabolism (P = .001), primarily overlapping the posterior cingulate cortex and precuneus, areas known to be significantly involved in Alzheimer’s disease.
When the investigators looked for behavioral and cognitive correlates of sleep-disordered breathing severity with associated brain changes, however, they found no associations with either cognitive performance, self-reported cognitive or sleep difficulties, or symptoms of daytime sleepiness.
“Importantly, to the best of our knowledge, our results show in vivo for the first time that greater amyloid burden colocalizes with greater gray-matter volume, perfusion, and metabolism in older participants with sleep-disordered breathing who are cognitively unimpaired. We believe that these overlapping patterns reinforce the likelihood of common underlying mechanisms,” they wrote.
The Age-Well randomized clinical trial is part of the Medit-Ageing project and is funded through the European Union’s Horizon 2020 Research and Innovation Program, INSERM, and Fondation d’ Entreprise MMA des Entrepreneurs du Futur. Dr. André reported no conflicts of interest to disclose.
SOURCE: André C et al. JAMA Neurol. 2020 Mar 23. doi: 10.1001/jamaneurol.2020.0311.
investigators have found.
Among 127 adults enrolled in a randomized clinical trial of interventions to promote mental well-being in older adults, those with sleep-disordered breathing had significantly greater amyloid burden and gray-matter volume, as well as increased perfusion and metabolism in parietal-occipital regions, reported Claire André, PhD, from the French Institute of Health and Medical Research (INSERM) unit in Caen, and colleagues.
“Our findings highlight the need to treat sleep disorders in the older population, even in the absence of cognitive or behavioral manifestations,” they wrote in a study published in JAMA Neurology.
Previous studies of the possible association between sleep-disordered breathing and dementia risk have shown conflicting or inconsistent results, the authors noted.
“These discrepancies may be explained by the characteristics of patients with sleep-disordered breathing (e.g., recruited from sleep clinics versus from the community, differences in age and disease duration), the scoring criteria of respiratory events, sample sizes, or the lack of controls for possibly biasing covariates,” they wrote.
To see whether they could clear up the confusion, the investigators conducted a retrospective analysis of 127 patients who were enrolled in the Age-Well randomized, controlled trial of the Medit-Ageing European project. The participants were community-dwelling adults (mean age, 69.1 years; 63% women), who were enrolled in the trial and underwent evaluation from 2016 to 2018 at the Cyceron Cancer Center in Caen.
The participants, all of whom were cognitively unimpaired at baseline, underwent neuropsychological assessment, polysomnography, MRI, plus florbetapir- and fluorodeoxyglucose-labeled PET.
The investigators defined sleep-disordered breathing as 15 apnea-hypopnea index events per hour or higher, and compared results between those with sleep-disordered breathing and those without for each imaging modality.
Participants with sleep-disordered breathing has significantly greater amyloid burden (P = .04), gray-matter volume (P = .04), perfusion (P = .04), and metabolism (P = .001), primarily overlapping the posterior cingulate cortex and precuneus, areas known to be significantly involved in Alzheimer’s disease.
When the investigators looked for behavioral and cognitive correlates of sleep-disordered breathing severity with associated brain changes, however, they found no associations with either cognitive performance, self-reported cognitive or sleep difficulties, or symptoms of daytime sleepiness.
“Importantly, to the best of our knowledge, our results show in vivo for the first time that greater amyloid burden colocalizes with greater gray-matter volume, perfusion, and metabolism in older participants with sleep-disordered breathing who are cognitively unimpaired. We believe that these overlapping patterns reinforce the likelihood of common underlying mechanisms,” they wrote.
The Age-Well randomized clinical trial is part of the Medit-Ageing project and is funded through the European Union’s Horizon 2020 Research and Innovation Program, INSERM, and Fondation d’ Entreprise MMA des Entrepreneurs du Futur. Dr. André reported no conflicts of interest to disclose.
SOURCE: André C et al. JAMA Neurol. 2020 Mar 23. doi: 10.1001/jamaneurol.2020.0311.
investigators have found.
Among 127 adults enrolled in a randomized clinical trial of interventions to promote mental well-being in older adults, those with sleep-disordered breathing had significantly greater amyloid burden and gray-matter volume, as well as increased perfusion and metabolism in parietal-occipital regions, reported Claire André, PhD, from the French Institute of Health and Medical Research (INSERM) unit in Caen, and colleagues.
“Our findings highlight the need to treat sleep disorders in the older population, even in the absence of cognitive or behavioral manifestations,” they wrote in a study published in JAMA Neurology.
Previous studies of the possible association between sleep-disordered breathing and dementia risk have shown conflicting or inconsistent results, the authors noted.
“These discrepancies may be explained by the characteristics of patients with sleep-disordered breathing (e.g., recruited from sleep clinics versus from the community, differences in age and disease duration), the scoring criteria of respiratory events, sample sizes, or the lack of controls for possibly biasing covariates,” they wrote.
To see whether they could clear up the confusion, the investigators conducted a retrospective analysis of 127 patients who were enrolled in the Age-Well randomized, controlled trial of the Medit-Ageing European project. The participants were community-dwelling adults (mean age, 69.1 years; 63% women), who were enrolled in the trial and underwent evaluation from 2016 to 2018 at the Cyceron Cancer Center in Caen.
The participants, all of whom were cognitively unimpaired at baseline, underwent neuropsychological assessment, polysomnography, MRI, plus florbetapir- and fluorodeoxyglucose-labeled PET.
The investigators defined sleep-disordered breathing as 15 apnea-hypopnea index events per hour or higher, and compared results between those with sleep-disordered breathing and those without for each imaging modality.
Participants with sleep-disordered breathing has significantly greater amyloid burden (P = .04), gray-matter volume (P = .04), perfusion (P = .04), and metabolism (P = .001), primarily overlapping the posterior cingulate cortex and precuneus, areas known to be significantly involved in Alzheimer’s disease.
When the investigators looked for behavioral and cognitive correlates of sleep-disordered breathing severity with associated brain changes, however, they found no associations with either cognitive performance, self-reported cognitive or sleep difficulties, or symptoms of daytime sleepiness.
“Importantly, to the best of our knowledge, our results show in vivo for the first time that greater amyloid burden colocalizes with greater gray-matter volume, perfusion, and metabolism in older participants with sleep-disordered breathing who are cognitively unimpaired. We believe that these overlapping patterns reinforce the likelihood of common underlying mechanisms,” they wrote.
The Age-Well randomized clinical trial is part of the Medit-Ageing project and is funded through the European Union’s Horizon 2020 Research and Innovation Program, INSERM, and Fondation d’ Entreprise MMA des Entrepreneurs du Futur. Dr. André reported no conflicts of interest to disclose.
SOURCE: André C et al. JAMA Neurol. 2020 Mar 23. doi: 10.1001/jamaneurol.2020.0311.
FROM JAMA NEUROLOGY
New ASAM guideline released amid COVID-19 concerns
Home-based buprenorphine induction deemed safe for OUD
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
Home-based buprenorphine induction deemed safe for OUD
Home-based buprenorphine induction deemed safe for OUD
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
Screen asymptomatic older adults for cognitive impairment? Not so fast
Reference
1. US Preventive Services Task Force. Final recommendation statement: cognitive impairment in older adults: screening. www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/cognitive-impairment-in-older-adults-screening. Published February 2020. Accessed March 19, 2020.
Reference
1. US Preventive Services Task Force. Final recommendation statement: cognitive impairment in older adults: screening. www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/cognitive-impairment-in-older-adults-screening. Published February 2020. Accessed March 19, 2020.
Reference
1. US Preventive Services Task Force. Final recommendation statement: cognitive impairment in older adults: screening. www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/cognitive-impairment-in-older-adults-screening. Published February 2020. Accessed March 19, 2020.
ICH survival lags in the community setting
LOS ANGELES – Although recent findings from circumscribed patient populations enrolled in intervention studies have shown improved survival rates in patients with a recent intracerebral hemorrhagic stroke, data from a large, observational study in the Netherlands suggested a much darker real-world picture, with a 6-month mortality of 64% identified in a total cohort of nearly 15,000 people followed prospectively starting in 1990.
In striking contrast to the survival pattern over time of patients in the same Dutch study who had a first acute ischemic stroke, which showed a statistically significant and meaningful cut in mortality for ischemic stroke patients during the 25-year period examined, survival rates for patients during the first months following a first intracerebral hemorrhage (ICH) stayed flat during 1991-2015, Reem Waziry, MD, said at the International Stroke Conference sponsored by the American Heart Association.
“The promising treatment advances [applied to patients] in the recent ICH trials may not be reflected in community-based treatment,” suggested Dr. Waziry, a research and teaching fellow in clinical epidemiology at the Harvard School of Public Health in Boston.
The data she reported came from the Rotterdam Study, which followed unselected, older people in the Rotterdam community with no stroke history, and during 25 years of monitoring identified 162 incident ICH strokes and 988 acute ischemic strokes. Concurrently with Dr. Waziry’s talk at the conference, the data she reported were published in Stroke. The data she reported also showed that, during the 25 years studied, mortality at 3 years following a first ICH stroke rose to 73% on average.
During her talk, Dr. Waziry also presented an unpublished comparison of the 64% 6-month mortality in the Rotterdam Study with the 3- to 6-month mortality reported in the control arms of four recent, randomized intervention trials, including the MISTIE III trial. Among the four randomized trials Dr. Waziry selected to make this post-hoc comparison, the study with the highest mortality among control patients was MISTIE III, which showed about 25% mortality after 6 months. In contrast, the 19% 6-month mortality among ischemic stroke patients in the Rotterdam Study was roughly similar to the mortality seem in the control arms of some recent studies of interventions for patients with acute ischemic stroke.
The Rotterdam Study receives no commercial funding. Dr. Waziry had no disclosures.
SOURCE: Waziry R et al. ISC 2020, Abstract LB14.
LOS ANGELES – Although recent findings from circumscribed patient populations enrolled in intervention studies have shown improved survival rates in patients with a recent intracerebral hemorrhagic stroke, data from a large, observational study in the Netherlands suggested a much darker real-world picture, with a 6-month mortality of 64% identified in a total cohort of nearly 15,000 people followed prospectively starting in 1990.
In striking contrast to the survival pattern over time of patients in the same Dutch study who had a first acute ischemic stroke, which showed a statistically significant and meaningful cut in mortality for ischemic stroke patients during the 25-year period examined, survival rates for patients during the first months following a first intracerebral hemorrhage (ICH) stayed flat during 1991-2015, Reem Waziry, MD, said at the International Stroke Conference sponsored by the American Heart Association.
“The promising treatment advances [applied to patients] in the recent ICH trials may not be reflected in community-based treatment,” suggested Dr. Waziry, a research and teaching fellow in clinical epidemiology at the Harvard School of Public Health in Boston.
The data she reported came from the Rotterdam Study, which followed unselected, older people in the Rotterdam community with no stroke history, and during 25 years of monitoring identified 162 incident ICH strokes and 988 acute ischemic strokes. Concurrently with Dr. Waziry’s talk at the conference, the data she reported were published in Stroke. The data she reported also showed that, during the 25 years studied, mortality at 3 years following a first ICH stroke rose to 73% on average.
During her talk, Dr. Waziry also presented an unpublished comparison of the 64% 6-month mortality in the Rotterdam Study with the 3- to 6-month mortality reported in the control arms of four recent, randomized intervention trials, including the MISTIE III trial. Among the four randomized trials Dr. Waziry selected to make this post-hoc comparison, the study with the highest mortality among control patients was MISTIE III, which showed about 25% mortality after 6 months. In contrast, the 19% 6-month mortality among ischemic stroke patients in the Rotterdam Study was roughly similar to the mortality seem in the control arms of some recent studies of interventions for patients with acute ischemic stroke.
The Rotterdam Study receives no commercial funding. Dr. Waziry had no disclosures.
SOURCE: Waziry R et al. ISC 2020, Abstract LB14.
LOS ANGELES – Although recent findings from circumscribed patient populations enrolled in intervention studies have shown improved survival rates in patients with a recent intracerebral hemorrhagic stroke, data from a large, observational study in the Netherlands suggested a much darker real-world picture, with a 6-month mortality of 64% identified in a total cohort of nearly 15,000 people followed prospectively starting in 1990.
In striking contrast to the survival pattern over time of patients in the same Dutch study who had a first acute ischemic stroke, which showed a statistically significant and meaningful cut in mortality for ischemic stroke patients during the 25-year period examined, survival rates for patients during the first months following a first intracerebral hemorrhage (ICH) stayed flat during 1991-2015, Reem Waziry, MD, said at the International Stroke Conference sponsored by the American Heart Association.
“The promising treatment advances [applied to patients] in the recent ICH trials may not be reflected in community-based treatment,” suggested Dr. Waziry, a research and teaching fellow in clinical epidemiology at the Harvard School of Public Health in Boston.
The data she reported came from the Rotterdam Study, which followed unselected, older people in the Rotterdam community with no stroke history, and during 25 years of monitoring identified 162 incident ICH strokes and 988 acute ischemic strokes. Concurrently with Dr. Waziry’s talk at the conference, the data she reported were published in Stroke. The data she reported also showed that, during the 25 years studied, mortality at 3 years following a first ICH stroke rose to 73% on average.
During her talk, Dr. Waziry also presented an unpublished comparison of the 64% 6-month mortality in the Rotterdam Study with the 3- to 6-month mortality reported in the control arms of four recent, randomized intervention trials, including the MISTIE III trial. Among the four randomized trials Dr. Waziry selected to make this post-hoc comparison, the study with the highest mortality among control patients was MISTIE III, which showed about 25% mortality after 6 months. In contrast, the 19% 6-month mortality among ischemic stroke patients in the Rotterdam Study was roughly similar to the mortality seem in the control arms of some recent studies of interventions for patients with acute ischemic stroke.
The Rotterdam Study receives no commercial funding. Dr. Waziry had no disclosures.
SOURCE: Waziry R et al. ISC 2020, Abstract LB14.
REPORTING FROM ISC 2020
Get With the Guidelines – Stroke targets ICH
The Get With the Guidelines – Stroke program is finally turning its attention to hemorrhagic strokes after having spurred improved patient management performance from participating U.S. stroke centers since its start in 2003 with a focus on acute ischemic stroke.
The advisers who craft policy for Get With the Guidelines – Stroke (GWTG–S) are planning to launch a pilot program later in 2020 that will initiate data monitoring and quality improvement aimed at optimizing care for patients following an intracerebral hemorrhage (ICH) starting at 15 U.S. stroke centers, with announcement of these 15 participating centers expected later in 2020. The program will start by targeting nine specific, evidence-based, key aspects of the acute management of ICH patients, said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn, and a volunteer expert who is part of the team developing the ICH initiative.
According to Dr. Sheth, the nine imperatives of acute ICH care that the program plans to monitor at participating centers are:
- Obtain a baseline severity score.
- Identify etiology as spontaneous or treatment related.
- Perform coagulopathy reversal or anticoagulant reversal.
- Administer venous thromboembolism prophylaxis.
- Apply dysphagia screening within 24 hours, and delay oral intake until patient passes dysphagia screen.
- Provide patient management in a multidisciplinary stroke or ICU unit.
- Prescribe appropriate blood pressure treatment at discharge.
- Perform assessment for rehabilitation.
- Avoid prescribing corticosteroids and other contraindicated drugs.
GWTG–S is adopting these metrics for assessing the acute care of ICH patients based largely on the recommendations of an expert 2018 panel organized by the American Heart Association and American Stroke. Association that proposed a set of performance measures for the care of ICH patients. This set of performance measures served as the primary basis for designing the new GWTG–S program, along with considerations of feasibility for collecting data on these measures, Dr. Sheth said in an interview. “We hope to make it easy” for centers to collect the data needed to participate.
The existing GWTG–S program is now 17-years old, and has spread to nearly 2,400 U.S. stroke centers as of early 2020, but the time has come to broaden its reach to patients with ICH and the programs that treat these patients, Dr. Sheth said. After years of nihilism about the prospects for patients following an ICH stroke, survival rates have increased, presenting “an opportunity to optimize care, for quality improvement,” he explained. “It’s a huge shift.” ICH patients “do better than we used to think.”
The Get With the Guidelines – Stroke program is finally turning its attention to hemorrhagic strokes after having spurred improved patient management performance from participating U.S. stroke centers since its start in 2003 with a focus on acute ischemic stroke.
The advisers who craft policy for Get With the Guidelines – Stroke (GWTG–S) are planning to launch a pilot program later in 2020 that will initiate data monitoring and quality improvement aimed at optimizing care for patients following an intracerebral hemorrhage (ICH) starting at 15 U.S. stroke centers, with announcement of these 15 participating centers expected later in 2020. The program will start by targeting nine specific, evidence-based, key aspects of the acute management of ICH patients, said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn, and a volunteer expert who is part of the team developing the ICH initiative.
According to Dr. Sheth, the nine imperatives of acute ICH care that the program plans to monitor at participating centers are:
- Obtain a baseline severity score.
- Identify etiology as spontaneous or treatment related.
- Perform coagulopathy reversal or anticoagulant reversal.
- Administer venous thromboembolism prophylaxis.
- Apply dysphagia screening within 24 hours, and delay oral intake until patient passes dysphagia screen.
- Provide patient management in a multidisciplinary stroke or ICU unit.
- Prescribe appropriate blood pressure treatment at discharge.
- Perform assessment for rehabilitation.
- Avoid prescribing corticosteroids and other contraindicated drugs.
GWTG–S is adopting these metrics for assessing the acute care of ICH patients based largely on the recommendations of an expert 2018 panel organized by the American Heart Association and American Stroke. Association that proposed a set of performance measures for the care of ICH patients. This set of performance measures served as the primary basis for designing the new GWTG–S program, along with considerations of feasibility for collecting data on these measures, Dr. Sheth said in an interview. “We hope to make it easy” for centers to collect the data needed to participate.
The existing GWTG–S program is now 17-years old, and has spread to nearly 2,400 U.S. stroke centers as of early 2020, but the time has come to broaden its reach to patients with ICH and the programs that treat these patients, Dr. Sheth said. After years of nihilism about the prospects for patients following an ICH stroke, survival rates have increased, presenting “an opportunity to optimize care, for quality improvement,” he explained. “It’s a huge shift.” ICH patients “do better than we used to think.”
The Get With the Guidelines – Stroke program is finally turning its attention to hemorrhagic strokes after having spurred improved patient management performance from participating U.S. stroke centers since its start in 2003 with a focus on acute ischemic stroke.
The advisers who craft policy for Get With the Guidelines – Stroke (GWTG–S) are planning to launch a pilot program later in 2020 that will initiate data monitoring and quality improvement aimed at optimizing care for patients following an intracerebral hemorrhage (ICH) starting at 15 U.S. stroke centers, with announcement of these 15 participating centers expected later in 2020. The program will start by targeting nine specific, evidence-based, key aspects of the acute management of ICH patients, said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn, and a volunteer expert who is part of the team developing the ICH initiative.
According to Dr. Sheth, the nine imperatives of acute ICH care that the program plans to monitor at participating centers are:
- Obtain a baseline severity score.
- Identify etiology as spontaneous or treatment related.
- Perform coagulopathy reversal or anticoagulant reversal.
- Administer venous thromboembolism prophylaxis.
- Apply dysphagia screening within 24 hours, and delay oral intake until patient passes dysphagia screen.
- Provide patient management in a multidisciplinary stroke or ICU unit.
- Prescribe appropriate blood pressure treatment at discharge.
- Perform assessment for rehabilitation.
- Avoid prescribing corticosteroids and other contraindicated drugs.
GWTG–S is adopting these metrics for assessing the acute care of ICH patients based largely on the recommendations of an expert 2018 panel organized by the American Heart Association and American Stroke. Association that proposed a set of performance measures for the care of ICH patients. This set of performance measures served as the primary basis for designing the new GWTG–S program, along with considerations of feasibility for collecting data on these measures, Dr. Sheth said in an interview. “We hope to make it easy” for centers to collect the data needed to participate.
The existing GWTG–S program is now 17-years old, and has spread to nearly 2,400 U.S. stroke centers as of early 2020, but the time has come to broaden its reach to patients with ICH and the programs that treat these patients, Dr. Sheth said. After years of nihilism about the prospects for patients following an ICH stroke, survival rates have increased, presenting “an opportunity to optimize care, for quality improvement,” he explained. “It’s a huge shift.” ICH patients “do better than we used to think.”
Recent treatment advances brighten prospects for intracerebral hemorrhage patients
LOS ANGELES – Intracerebral hemorrhage (ICH) appears to be not nearly as uniformly devastating to patients as its reputation suggests. Recent study results documented unexpectedly decent recovery prospects for hemorrhagic stroke patients assessed after 1 year who were earlier considered moderately severe or severely disabled based on their 30-day status. And these data provide further support for the growing impression among clinicians that a way forward for improving outcomes even more is with a “gentle” surgical intervention designed to substantially reduce ICH clot volume.
“Historically, there’s been a lot of nihilism around these patients. Intracerebral hemorrhage has always been the deadliest stroke type, but one of the great advances of the past 10-20 years is that ICH survival has improved. Patients do better than we used to think,” said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn. “Even though ICH remains a difficult disease, this change has two big implications,” Dr. Sheth said in an interview during the International Stroke Conference sponsored by the American Heart Association. First, increased ICH survival offers an opportunity to expand the reach of recent management advances through quality improvement programs that emphasize new strategies that work better and incentivize delivery of these successful strategies to more patients.
The second implication is simply a growing number of ICH survivors, expanding the population of patients who stand to gain from these new management strategies. Dr. Sheth is working with the Get With the Guidelines – Stroke program, a quality-improvement program begun in 2003 and until now aimed at patients with acute ischemic stroke, to develop a 15-site pilot program planned to start in 2020 that will begin implementing and studying a Get With the Guidelines – Stroke quality-improvement program focused on patients with an ICH. The current conception of a quality measurement and improvement program like Get with the Guidelines – Stroke for patients with ICH stems from an important, earlier milestone in the emergence of effective ICH treatments, the 2018 publication of performance measures for ICH care that identified nine key management steps for assessing quality of care and documented the evidence behind them.
“Evidence for optimal treatment of ICH has lagged behind that for ischemic stroke, and consequently, metrics specific to ICH care have not been widely promulgated,” said the authors of the 2018 ICH performance measures, a panel that included Dr. Sheth. “However, numerous more recent studies and clinical trials of various medical and surgical interventions for ICH have been published and form the basis of evidence-based guidelines for the management of ICH,” they explained.
MISTIE III showcases better ICH outcomes
Perhaps the most dramatic recent evidence of brighter prospects for ICH patients came in data collected during the MISTIE III (Minimally Invasive Surgery with Thrombolysis in Intracerebral Hemorrhage Evacuation III) trial, which randomized 506 ICH patients with a hematoma of at least 30 mL to standard care or to a “gentle” clot-reduction protocol using a small-bore catheter placed with stereotactic guidance to both evacuate clot and introduce a serial infusion of alteplase into the clot to try to shrink its volume to less than 15 mL. The study’s results showed a neutral effect for the primary outcome, the incidence of recovery to a modified Rankin Scale (mRS) score of 0-3 at 1 year after entry, which occurred in 45% of the surgically treated patients and 41% of the controls in a modified intention-to-treat analysis that included 499 of the randomized patients, a difference that did not reach statistical significance.
However, when the analysis focused on the 146 of 247 patients (59%) randomized to surgical plus lytic intervention who underwent the procedure and actually had their clot volume reduced to 15 mL or less per protocol, the adjusted incidence of the primary endpoint was double that of patients who underwent the procedure but failed to have their residual clot reduced to this size. A similar doubling of good outcomes occurred when MISTIE patients had their residual clot cut to 20 mL or less, compared with those who didn’t reach this, with the differences in both analyses statistically significant. The actual rates showed patients with clot cut to 15 mL or less having a 53% rate of a mRS score of 0-3 after 1 year, compared with 33% of patients who received the intervention but had their residual clot remain above 15 mL.
The MISTIE III investigators looked at their data to try to get better insight into the outcome of all “poor prognosis” patients in the study regardless of their treatment arm assignment, and how patients and their family members made decisions for withdrawal of life-sustaining therapy. In MISTIE III, 61 patients had withdrawal of life-sustaining treatment (WoLST), with more than 40% of the WoLST occurring with patients randomized to the intervention arm including 10 patients treated to a residual clot volume of 15 mL or less. To quantify the disease severity in these 61 patients, the researchers applied a six-item formula at 30 days after the stroke, a metric their 2019 report described in detail. They then used these severity scores to identify 104 matched patients who were alive at 30 days and remained on life-sustaining treatment to see their 1-year outcomes. At 30 days, the 104 matched patients included 82 (79%) with a mRS score of 5 (severe disability) and 22 patients (21%) with a mRS score of 4 (moderately severe disability). Overall, an mRS score of 4 or 5 was quite prevalent 30 days after the stroke, with 87% of the patients treated with the MISTIE intervention and 90% of the control patients having this degree of disability at 30 days.
When the MISTIE III investigators followed these patients for a year, they made an unexpected finding: A substantial incidence of patients whose condition had improved since day 30. One year out, 40 (39%) of these 104 patients had improved to a mRS score of 1-3, including 10 (10%) with a mRS score of 1 or 2. Another indicator of the reasonable outcome many of these patients achieved was that after 1 year 69% were living at home.
“Our data show that many ICH subjects with clinical factors that suggest ‘poor prognosis,’ when given time, can achieve a favorable outcome and return home,” concluded Noeleen Ostapkovich, who presented these results at the Stroke Conference.
She cited these findings as potentially helpful for refining the information given to patients and families on the prognosis for ICH patients at about 30 days after their event, the usual time for assessment. “These patients looked like they weren’t going to do well after 30 days, but by 365 days they had improved physically and in their ability to care for themselves at home,” noted Ms. Ostapkovich, a researcher in the Brain Injury Outcomes Clinical Trial Coordinating Center of Johns Hopkins University in Baltimore.
A message for acute-care clinicians
She and her colleagues highlighted the implications these new findings have for clinical decision making in the first weeks after an ICH.
“Acute-care physicians see these patients at day 30, not at day 365, so it’s important that they have a clear picture of what these patients could look like a year later. It’s an important message,” Ms. Ostapkovich said in an interview.
In fact, a colleague of hers at Johns Hopkins ran an analysis that looked at factors that contributed to families opting for WoLST for 61 of the MISTIE III patients, and found that 38 family groups (62%) cited the anticipated outcome of the patient in a dependent state as their primary reason for opting for WoLST, Lourdes J. Carhuapoma reported in a separate talk at the conference.
“The main message is that many patients with significant ICH did well and recovered despite having very poor prognostic factors at 30 days, but it took more time. A concern is that the [prognostic] information families receive may be wrong. There is a disconnect,” between what families get told to expect and what actually happens, said Ms. Carhuapoma, an acute care nurse practitioner at Johns Hopkins.
“When physicians, nurses, and family members get together” to discuss ICH patients like these after 30 days, “they see the glass as empty. But the real message is that the glass is half full,” summed up Daniel F. Hanley, MD, lead investigator of MISTIE III and professor of neurology at Johns Hopkins. “These data show a large amount of improvement between 30 and 180 days.” The 104 patients with exclusively mRS scores of 4 or 5 at day 30 had a 30% incidence of improvement to an mRS score of 2 or 3 after 180 days, on their way to a 39% rate of mRS scores of 1-3 at 1 year.
An additional analysis that has not yet been presented showed that the “strongest predictor” of whether or not patients who presented with a mRS score of 4 or 5 after 30 days improved their status at 1 year was if their residual hematoma volume shrank to 15 mL or less, Dr. Hanley said in an interview. “It’s not rocket science. If you had to choose between a 45-mL hematoma and less than 15 mL, which would you choose? What’s new here is how this recovery can play out,” taking 180 days or longer in some patients to become apparent.
More evidence needed to prove MISTIE’s hypothesis
According to Dr. Hanley, the MISTIE III findings have begun to influence practice despite its neutral primary finding, with more attention being paid to reducing residual clot volume following an ICH. And evidence continues to mount that more aggressive minimization of hematoma size can have an important effect on outcomes. For example, another study presented at the conference assessed the incremental change in prognostic accuracy when the ICH score, a five-item formula for estimating the prognosis of an ICH patient, substituted a precise quantification of residual hematoma volume rather than the original, dichotomous entry for either a hematoma volume of 30 mL or greater, or less than 30 mL, and when the severity score also quantified intraventricular hemorrhage (IVH) volume rather than simply designating IVH as present or absent.
Using data from 933 patients who had been enrolled in either MISTIE III or in another study of hematoma volume reduction, CLEAR III, the analysis showed that including specific quantification of both residual ICH volume as well as residual IVH volume improved the area under the receiver operating characteristic curve of the ICH score as a prognostic assessment from 0.70 to 0.75 in the intervention arms of the two trials, and from 0.60 to 0.68 in the two combined control arms, Adam de Havenon, MD, reported in a talk at the conference. “These data show that quantifying ICH and IVH volume improves mortality prognostication,” concluded Dr. de Havenon, a vascular and stroke neurologist at the University of Utah in Salt Lake City.
Furthermore, it’s “certainly evidence for the importance of volume reduction,” he said during discussion of his talk. “The MISTIE procedure can reset patients” so that their outcomes become more like patients with much smaller clot volumes even if they start with large hematomas. “In our experience, if the volume is reduced to 5 mL, there is real benefit regardless of how big the clot was initially,” Dr. de Havenon said.
But the neutral result for the MISTIE III primary endpoint will, for the time being, hobble application of this concept and keep the MISTIE intervention from rising to a level I recommendation until greater evidence for its efficacy comes out.
“It’s been known for many years that clot size matters when it comes to ICH. The MISTIE team has made a very compelling case that [reducing clot volume] is a very reasonable hypothesis, but we must continue to acquire data that can confirm it,” Dr. Sheth commented.
Dr. Sheth’s institution receives research funding from Novartis and Bard for studies that Dr. Sheth helps run. The MISTIE III study received the alteplase used in the study at no cost from Genentech. Ms. Ostapkovich and Ms. Carhuapoma had no disclosures. Dr. Hanley has received personal fees from BrainScope, Medtronic, Neurotrope, Op2Lysis, and Portola. Dr. de Havenon has received research funding from Regeneron.
LOS ANGELES – Intracerebral hemorrhage (ICH) appears to be not nearly as uniformly devastating to patients as its reputation suggests. Recent study results documented unexpectedly decent recovery prospects for hemorrhagic stroke patients assessed after 1 year who were earlier considered moderately severe or severely disabled based on their 30-day status. And these data provide further support for the growing impression among clinicians that a way forward for improving outcomes even more is with a “gentle” surgical intervention designed to substantially reduce ICH clot volume.
“Historically, there’s been a lot of nihilism around these patients. Intracerebral hemorrhage has always been the deadliest stroke type, but one of the great advances of the past 10-20 years is that ICH survival has improved. Patients do better than we used to think,” said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn. “Even though ICH remains a difficult disease, this change has two big implications,” Dr. Sheth said in an interview during the International Stroke Conference sponsored by the American Heart Association. First, increased ICH survival offers an opportunity to expand the reach of recent management advances through quality improvement programs that emphasize new strategies that work better and incentivize delivery of these successful strategies to more patients.
The second implication is simply a growing number of ICH survivors, expanding the population of patients who stand to gain from these new management strategies. Dr. Sheth is working with the Get With the Guidelines – Stroke program, a quality-improvement program begun in 2003 and until now aimed at patients with acute ischemic stroke, to develop a 15-site pilot program planned to start in 2020 that will begin implementing and studying a Get With the Guidelines – Stroke quality-improvement program focused on patients with an ICH. The current conception of a quality measurement and improvement program like Get with the Guidelines – Stroke for patients with ICH stems from an important, earlier milestone in the emergence of effective ICH treatments, the 2018 publication of performance measures for ICH care that identified nine key management steps for assessing quality of care and documented the evidence behind them.
“Evidence for optimal treatment of ICH has lagged behind that for ischemic stroke, and consequently, metrics specific to ICH care have not been widely promulgated,” said the authors of the 2018 ICH performance measures, a panel that included Dr. Sheth. “However, numerous more recent studies and clinical trials of various medical and surgical interventions for ICH have been published and form the basis of evidence-based guidelines for the management of ICH,” they explained.
MISTIE III showcases better ICH outcomes
Perhaps the most dramatic recent evidence of brighter prospects for ICH patients came in data collected during the MISTIE III (Minimally Invasive Surgery with Thrombolysis in Intracerebral Hemorrhage Evacuation III) trial, which randomized 506 ICH patients with a hematoma of at least 30 mL to standard care or to a “gentle” clot-reduction protocol using a small-bore catheter placed with stereotactic guidance to both evacuate clot and introduce a serial infusion of alteplase into the clot to try to shrink its volume to less than 15 mL. The study’s results showed a neutral effect for the primary outcome, the incidence of recovery to a modified Rankin Scale (mRS) score of 0-3 at 1 year after entry, which occurred in 45% of the surgically treated patients and 41% of the controls in a modified intention-to-treat analysis that included 499 of the randomized patients, a difference that did not reach statistical significance.
However, when the analysis focused on the 146 of 247 patients (59%) randomized to surgical plus lytic intervention who underwent the procedure and actually had their clot volume reduced to 15 mL or less per protocol, the adjusted incidence of the primary endpoint was double that of patients who underwent the procedure but failed to have their residual clot reduced to this size. A similar doubling of good outcomes occurred when MISTIE patients had their residual clot cut to 20 mL or less, compared with those who didn’t reach this, with the differences in both analyses statistically significant. The actual rates showed patients with clot cut to 15 mL or less having a 53% rate of a mRS score of 0-3 after 1 year, compared with 33% of patients who received the intervention but had their residual clot remain above 15 mL.
The MISTIE III investigators looked at their data to try to get better insight into the outcome of all “poor prognosis” patients in the study regardless of their treatment arm assignment, and how patients and their family members made decisions for withdrawal of life-sustaining therapy. In MISTIE III, 61 patients had withdrawal of life-sustaining treatment (WoLST), with more than 40% of the WoLST occurring with patients randomized to the intervention arm including 10 patients treated to a residual clot volume of 15 mL or less. To quantify the disease severity in these 61 patients, the researchers applied a six-item formula at 30 days after the stroke, a metric their 2019 report described in detail. They then used these severity scores to identify 104 matched patients who were alive at 30 days and remained on life-sustaining treatment to see their 1-year outcomes. At 30 days, the 104 matched patients included 82 (79%) with a mRS score of 5 (severe disability) and 22 patients (21%) with a mRS score of 4 (moderately severe disability). Overall, an mRS score of 4 or 5 was quite prevalent 30 days after the stroke, with 87% of the patients treated with the MISTIE intervention and 90% of the control patients having this degree of disability at 30 days.
When the MISTIE III investigators followed these patients for a year, they made an unexpected finding: A substantial incidence of patients whose condition had improved since day 30. One year out, 40 (39%) of these 104 patients had improved to a mRS score of 1-3, including 10 (10%) with a mRS score of 1 or 2. Another indicator of the reasonable outcome many of these patients achieved was that after 1 year 69% were living at home.
“Our data show that many ICH subjects with clinical factors that suggest ‘poor prognosis,’ when given time, can achieve a favorable outcome and return home,” concluded Noeleen Ostapkovich, who presented these results at the Stroke Conference.
She cited these findings as potentially helpful for refining the information given to patients and families on the prognosis for ICH patients at about 30 days after their event, the usual time for assessment. “These patients looked like they weren’t going to do well after 30 days, but by 365 days they had improved physically and in their ability to care for themselves at home,” noted Ms. Ostapkovich, a researcher in the Brain Injury Outcomes Clinical Trial Coordinating Center of Johns Hopkins University in Baltimore.
A message for acute-care clinicians
She and her colleagues highlighted the implications these new findings have for clinical decision making in the first weeks after an ICH.
“Acute-care physicians see these patients at day 30, not at day 365, so it’s important that they have a clear picture of what these patients could look like a year later. It’s an important message,” Ms. Ostapkovich said in an interview.
In fact, a colleague of hers at Johns Hopkins ran an analysis that looked at factors that contributed to families opting for WoLST for 61 of the MISTIE III patients, and found that 38 family groups (62%) cited the anticipated outcome of the patient in a dependent state as their primary reason for opting for WoLST, Lourdes J. Carhuapoma reported in a separate talk at the conference.
“The main message is that many patients with significant ICH did well and recovered despite having very poor prognostic factors at 30 days, but it took more time. A concern is that the [prognostic] information families receive may be wrong. There is a disconnect,” between what families get told to expect and what actually happens, said Ms. Carhuapoma, an acute care nurse practitioner at Johns Hopkins.
“When physicians, nurses, and family members get together” to discuss ICH patients like these after 30 days, “they see the glass as empty. But the real message is that the glass is half full,” summed up Daniel F. Hanley, MD, lead investigator of MISTIE III and professor of neurology at Johns Hopkins. “These data show a large amount of improvement between 30 and 180 days.” The 104 patients with exclusively mRS scores of 4 or 5 at day 30 had a 30% incidence of improvement to an mRS score of 2 or 3 after 180 days, on their way to a 39% rate of mRS scores of 1-3 at 1 year.
An additional analysis that has not yet been presented showed that the “strongest predictor” of whether or not patients who presented with a mRS score of 4 or 5 after 30 days improved their status at 1 year was if their residual hematoma volume shrank to 15 mL or less, Dr. Hanley said in an interview. “It’s not rocket science. If you had to choose between a 45-mL hematoma and less than 15 mL, which would you choose? What’s new here is how this recovery can play out,” taking 180 days or longer in some patients to become apparent.
More evidence needed to prove MISTIE’s hypothesis
According to Dr. Hanley, the MISTIE III findings have begun to influence practice despite its neutral primary finding, with more attention being paid to reducing residual clot volume following an ICH. And evidence continues to mount that more aggressive minimization of hematoma size can have an important effect on outcomes. For example, another study presented at the conference assessed the incremental change in prognostic accuracy when the ICH score, a five-item formula for estimating the prognosis of an ICH patient, substituted a precise quantification of residual hematoma volume rather than the original, dichotomous entry for either a hematoma volume of 30 mL or greater, or less than 30 mL, and when the severity score also quantified intraventricular hemorrhage (IVH) volume rather than simply designating IVH as present or absent.
Using data from 933 patients who had been enrolled in either MISTIE III or in another study of hematoma volume reduction, CLEAR III, the analysis showed that including specific quantification of both residual ICH volume as well as residual IVH volume improved the area under the receiver operating characteristic curve of the ICH score as a prognostic assessment from 0.70 to 0.75 in the intervention arms of the two trials, and from 0.60 to 0.68 in the two combined control arms, Adam de Havenon, MD, reported in a talk at the conference. “These data show that quantifying ICH and IVH volume improves mortality prognostication,” concluded Dr. de Havenon, a vascular and stroke neurologist at the University of Utah in Salt Lake City.
Furthermore, it’s “certainly evidence for the importance of volume reduction,” he said during discussion of his talk. “The MISTIE procedure can reset patients” so that their outcomes become more like patients with much smaller clot volumes even if they start with large hematomas. “In our experience, if the volume is reduced to 5 mL, there is real benefit regardless of how big the clot was initially,” Dr. de Havenon said.
But the neutral result for the MISTIE III primary endpoint will, for the time being, hobble application of this concept and keep the MISTIE intervention from rising to a level I recommendation until greater evidence for its efficacy comes out.
“It’s been known for many years that clot size matters when it comes to ICH. The MISTIE team has made a very compelling case that [reducing clot volume] is a very reasonable hypothesis, but we must continue to acquire data that can confirm it,” Dr. Sheth commented.
Dr. Sheth’s institution receives research funding from Novartis and Bard for studies that Dr. Sheth helps run. The MISTIE III study received the alteplase used in the study at no cost from Genentech. Ms. Ostapkovich and Ms. Carhuapoma had no disclosures. Dr. Hanley has received personal fees from BrainScope, Medtronic, Neurotrope, Op2Lysis, and Portola. Dr. de Havenon has received research funding from Regeneron.
LOS ANGELES – Intracerebral hemorrhage (ICH) appears to be not nearly as uniformly devastating to patients as its reputation suggests. Recent study results documented unexpectedly decent recovery prospects for hemorrhagic stroke patients assessed after 1 year who were earlier considered moderately severe or severely disabled based on their 30-day status. And these data provide further support for the growing impression among clinicians that a way forward for improving outcomes even more is with a “gentle” surgical intervention designed to substantially reduce ICH clot volume.
“Historically, there’s been a lot of nihilism around these patients. Intracerebral hemorrhage has always been the deadliest stroke type, but one of the great advances of the past 10-20 years is that ICH survival has improved. Patients do better than we used to think,” said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn. “Even though ICH remains a difficult disease, this change has two big implications,” Dr. Sheth said in an interview during the International Stroke Conference sponsored by the American Heart Association. First, increased ICH survival offers an opportunity to expand the reach of recent management advances through quality improvement programs that emphasize new strategies that work better and incentivize delivery of these successful strategies to more patients.
The second implication is simply a growing number of ICH survivors, expanding the population of patients who stand to gain from these new management strategies. Dr. Sheth is working with the Get With the Guidelines – Stroke program, a quality-improvement program begun in 2003 and until now aimed at patients with acute ischemic stroke, to develop a 15-site pilot program planned to start in 2020 that will begin implementing and studying a Get With the Guidelines – Stroke quality-improvement program focused on patients with an ICH. The current conception of a quality measurement and improvement program like Get with the Guidelines – Stroke for patients with ICH stems from an important, earlier milestone in the emergence of effective ICH treatments, the 2018 publication of performance measures for ICH care that identified nine key management steps for assessing quality of care and documented the evidence behind them.
“Evidence for optimal treatment of ICH has lagged behind that for ischemic stroke, and consequently, metrics specific to ICH care have not been widely promulgated,” said the authors of the 2018 ICH performance measures, a panel that included Dr. Sheth. “However, numerous more recent studies and clinical trials of various medical and surgical interventions for ICH have been published and form the basis of evidence-based guidelines for the management of ICH,” they explained.
MISTIE III showcases better ICH outcomes
Perhaps the most dramatic recent evidence of brighter prospects for ICH patients came in data collected during the MISTIE III (Minimally Invasive Surgery with Thrombolysis in Intracerebral Hemorrhage Evacuation III) trial, which randomized 506 ICH patients with a hematoma of at least 30 mL to standard care or to a “gentle” clot-reduction protocol using a small-bore catheter placed with stereotactic guidance to both evacuate clot and introduce a serial infusion of alteplase into the clot to try to shrink its volume to less than 15 mL. The study’s results showed a neutral effect for the primary outcome, the incidence of recovery to a modified Rankin Scale (mRS) score of 0-3 at 1 year after entry, which occurred in 45% of the surgically treated patients and 41% of the controls in a modified intention-to-treat analysis that included 499 of the randomized patients, a difference that did not reach statistical significance.
However, when the analysis focused on the 146 of 247 patients (59%) randomized to surgical plus lytic intervention who underwent the procedure and actually had their clot volume reduced to 15 mL or less per protocol, the adjusted incidence of the primary endpoint was double that of patients who underwent the procedure but failed to have their residual clot reduced to this size. A similar doubling of good outcomes occurred when MISTIE patients had their residual clot cut to 20 mL or less, compared with those who didn’t reach this, with the differences in both analyses statistically significant. The actual rates showed patients with clot cut to 15 mL or less having a 53% rate of a mRS score of 0-3 after 1 year, compared with 33% of patients who received the intervention but had their residual clot remain above 15 mL.
The MISTIE III investigators looked at their data to try to get better insight into the outcome of all “poor prognosis” patients in the study regardless of their treatment arm assignment, and how patients and their family members made decisions for withdrawal of life-sustaining therapy. In MISTIE III, 61 patients had withdrawal of life-sustaining treatment (WoLST), with more than 40% of the WoLST occurring with patients randomized to the intervention arm including 10 patients treated to a residual clot volume of 15 mL or less. To quantify the disease severity in these 61 patients, the researchers applied a six-item formula at 30 days after the stroke, a metric their 2019 report described in detail. They then used these severity scores to identify 104 matched patients who were alive at 30 days and remained on life-sustaining treatment to see their 1-year outcomes. At 30 days, the 104 matched patients included 82 (79%) with a mRS score of 5 (severe disability) and 22 patients (21%) with a mRS score of 4 (moderately severe disability). Overall, an mRS score of 4 or 5 was quite prevalent 30 days after the stroke, with 87% of the patients treated with the MISTIE intervention and 90% of the control patients having this degree of disability at 30 days.
When the MISTIE III investigators followed these patients for a year, they made an unexpected finding: A substantial incidence of patients whose condition had improved since day 30. One year out, 40 (39%) of these 104 patients had improved to a mRS score of 1-3, including 10 (10%) with a mRS score of 1 or 2. Another indicator of the reasonable outcome many of these patients achieved was that after 1 year 69% were living at home.
“Our data show that many ICH subjects with clinical factors that suggest ‘poor prognosis,’ when given time, can achieve a favorable outcome and return home,” concluded Noeleen Ostapkovich, who presented these results at the Stroke Conference.
She cited these findings as potentially helpful for refining the information given to patients and families on the prognosis for ICH patients at about 30 days after their event, the usual time for assessment. “These patients looked like they weren’t going to do well after 30 days, but by 365 days they had improved physically and in their ability to care for themselves at home,” noted Ms. Ostapkovich, a researcher in the Brain Injury Outcomes Clinical Trial Coordinating Center of Johns Hopkins University in Baltimore.
A message for acute-care clinicians
She and her colleagues highlighted the implications these new findings have for clinical decision making in the first weeks after an ICH.
“Acute-care physicians see these patients at day 30, not at day 365, so it’s important that they have a clear picture of what these patients could look like a year later. It’s an important message,” Ms. Ostapkovich said in an interview.
In fact, a colleague of hers at Johns Hopkins ran an analysis that looked at factors that contributed to families opting for WoLST for 61 of the MISTIE III patients, and found that 38 family groups (62%) cited the anticipated outcome of the patient in a dependent state as their primary reason for opting for WoLST, Lourdes J. Carhuapoma reported in a separate talk at the conference.
“The main message is that many patients with significant ICH did well and recovered despite having very poor prognostic factors at 30 days, but it took more time. A concern is that the [prognostic] information families receive may be wrong. There is a disconnect,” between what families get told to expect and what actually happens, said Ms. Carhuapoma, an acute care nurse practitioner at Johns Hopkins.
“When physicians, nurses, and family members get together” to discuss ICH patients like these after 30 days, “they see the glass as empty. But the real message is that the glass is half full,” summed up Daniel F. Hanley, MD, lead investigator of MISTIE III and professor of neurology at Johns Hopkins. “These data show a large amount of improvement between 30 and 180 days.” The 104 patients with exclusively mRS scores of 4 or 5 at day 30 had a 30% incidence of improvement to an mRS score of 2 or 3 after 180 days, on their way to a 39% rate of mRS scores of 1-3 at 1 year.
An additional analysis that has not yet been presented showed that the “strongest predictor” of whether or not patients who presented with a mRS score of 4 or 5 after 30 days improved their status at 1 year was if their residual hematoma volume shrank to 15 mL or less, Dr. Hanley said in an interview. “It’s not rocket science. If you had to choose between a 45-mL hematoma and less than 15 mL, which would you choose? What’s new here is how this recovery can play out,” taking 180 days or longer in some patients to become apparent.
More evidence needed to prove MISTIE’s hypothesis
According to Dr. Hanley, the MISTIE III findings have begun to influence practice despite its neutral primary finding, with more attention being paid to reducing residual clot volume following an ICH. And evidence continues to mount that more aggressive minimization of hematoma size can have an important effect on outcomes. For example, another study presented at the conference assessed the incremental change in prognostic accuracy when the ICH score, a five-item formula for estimating the prognosis of an ICH patient, substituted a precise quantification of residual hematoma volume rather than the original, dichotomous entry for either a hematoma volume of 30 mL or greater, or less than 30 mL, and when the severity score also quantified intraventricular hemorrhage (IVH) volume rather than simply designating IVH as present or absent.
Using data from 933 patients who had been enrolled in either MISTIE III or in another study of hematoma volume reduction, CLEAR III, the analysis showed that including specific quantification of both residual ICH volume as well as residual IVH volume improved the area under the receiver operating characteristic curve of the ICH score as a prognostic assessment from 0.70 to 0.75 in the intervention arms of the two trials, and from 0.60 to 0.68 in the two combined control arms, Adam de Havenon, MD, reported in a talk at the conference. “These data show that quantifying ICH and IVH volume improves mortality prognostication,” concluded Dr. de Havenon, a vascular and stroke neurologist at the University of Utah in Salt Lake City.
Furthermore, it’s “certainly evidence for the importance of volume reduction,” he said during discussion of his talk. “The MISTIE procedure can reset patients” so that their outcomes become more like patients with much smaller clot volumes even if they start with large hematomas. “In our experience, if the volume is reduced to 5 mL, there is real benefit regardless of how big the clot was initially,” Dr. de Havenon said.
But the neutral result for the MISTIE III primary endpoint will, for the time being, hobble application of this concept and keep the MISTIE intervention from rising to a level I recommendation until greater evidence for its efficacy comes out.
“It’s been known for many years that clot size matters when it comes to ICH. The MISTIE team has made a very compelling case that [reducing clot volume] is a very reasonable hypothesis, but we must continue to acquire data that can confirm it,” Dr. Sheth commented.
Dr. Sheth’s institution receives research funding from Novartis and Bard for studies that Dr. Sheth helps run. The MISTIE III study received the alteplase used in the study at no cost from Genentech. Ms. Ostapkovich and Ms. Carhuapoma had no disclosures. Dr. Hanley has received personal fees from BrainScope, Medtronic, Neurotrope, Op2Lysis, and Portola. Dr. de Havenon has received research funding from Regeneron.
REPORTING FROM ISC 2020
Do urgent care centers use optimal medications for acute migraine?
according to a study published in the March issue of Headache. Pain and nausea or vomiting associated with migraine may go undertreated, and treatment may not be consistent with American Headache Society (AHS) guidelines for EDs, said Mia T. Minen, MD, of the department of neurology and population health at NYU Langone Health in New York and colleagues.
“Our study findings raise the question as to whether the patients with migraine in the urgent care setting should be managed similarly to the ED, and whether the AHS guidelines for the ED should be revisited and applied to urgent care,” the researchers noted.
Relative to the ED, urgent care centers may provide cost savings and emerge “as a preferred place for treatment for people with migraine, perhaps as they are potentially more quiet medical settings where people with migraine might expeditiously receive care,” the authors said.
Dr. Minen and colleagues conducted a retrospective chart review to assess migraine management at two urgent care centers in New York. They examined the number of urgent care visits for migraine, treatments used, and how closely clinicians followed the AHS recommendations for administration of antiemetic medication and triptans, among other outcomes.
The study population included adults diagnosed with migraine at the NYU Langone Medhattan Urgent Care center between Dec. 1, 2015, and Dec. 1, 2018, or at the NYU Langone Ambulatory Care Urgent Care West Side center between May 1, 2017, and Dec. 1, 2018. Of more than 32,000 urgent care visits during the study period, 78 patients received a migraine diagnosis. Patients with migraine had an average age of 32.5 years, and 79.5% were female. More than half had a documented history of migraine. Two of the patients (2.6%) had been to an emergency department for headache or migraine.
Less than half of the patients who presented with pain (46.6%) were given medication, most commonly ketorolac injection. Most patients (78.2%) received prescriptions, and 25.6% received a triptan prescription. About 60% of patients were told to follow up with a neurologist. In addition, 11.5% revisited urgent care with a migraine or headache or to request a prescription refill.
“Patients in this study appeared to be using the urgent care centers specifically for acute care,” the researchers said. “The patients generally had infrequent headaches and the majority would not have qualified for migraine preventive treatment.”
Although AHS guidelines include three “should offer” medications for acute management of migraine in the ED – intravenous metoclopramide, intravenous prochlorperazine, and subcutaneous sumatriptan – two of the medications, subcutaneous sumatriptan and intravenous prochlorperazine, were not available in the urgent care pharmacy. “Of the level B migraine medications, only metoclopramide IV was in the pharmacy, and only 12.3% was given this at their urgent care visit,” the researchers said. “There was also likely undertreatment of nausea/vomiting; despite 39 patients with recorded nausea or vomiting with their migraine, less than half (46.2%) received an antiemetic at the visit,” including metoclopramide or ondansetron through oral or intravenous administration.
Future studies should look at headache and migraine visits at urgent care centers across the United States, the investigators suggested.
One of the authors of the study (Leslie Miller, MD) is the head of the NYU Langone Health Urgent Care Centers. Dr. Minen has received grant support, honoraria, or travel funds from the National Institutes of Health, the American Academy of Neurology, the American Brain Foundation, the National Multiple Sclerosis Society, the National Headache Foundation, the American Headache Society, Barnard College, and NYU. Dr. Minen is associate editor of Headache.
SOURCE: Minen MT et al. Headache. 2020;60(3):542-52.
according to a study published in the March issue of Headache. Pain and nausea or vomiting associated with migraine may go undertreated, and treatment may not be consistent with American Headache Society (AHS) guidelines for EDs, said Mia T. Minen, MD, of the department of neurology and population health at NYU Langone Health in New York and colleagues.
“Our study findings raise the question as to whether the patients with migraine in the urgent care setting should be managed similarly to the ED, and whether the AHS guidelines for the ED should be revisited and applied to urgent care,” the researchers noted.
Relative to the ED, urgent care centers may provide cost savings and emerge “as a preferred place for treatment for people with migraine, perhaps as they are potentially more quiet medical settings where people with migraine might expeditiously receive care,” the authors said.
Dr. Minen and colleagues conducted a retrospective chart review to assess migraine management at two urgent care centers in New York. They examined the number of urgent care visits for migraine, treatments used, and how closely clinicians followed the AHS recommendations for administration of antiemetic medication and triptans, among other outcomes.
The study population included adults diagnosed with migraine at the NYU Langone Medhattan Urgent Care center between Dec. 1, 2015, and Dec. 1, 2018, or at the NYU Langone Ambulatory Care Urgent Care West Side center between May 1, 2017, and Dec. 1, 2018. Of more than 32,000 urgent care visits during the study period, 78 patients received a migraine diagnosis. Patients with migraine had an average age of 32.5 years, and 79.5% were female. More than half had a documented history of migraine. Two of the patients (2.6%) had been to an emergency department for headache or migraine.
Less than half of the patients who presented with pain (46.6%) were given medication, most commonly ketorolac injection. Most patients (78.2%) received prescriptions, and 25.6% received a triptan prescription. About 60% of patients were told to follow up with a neurologist. In addition, 11.5% revisited urgent care with a migraine or headache or to request a prescription refill.
“Patients in this study appeared to be using the urgent care centers specifically for acute care,” the researchers said. “The patients generally had infrequent headaches and the majority would not have qualified for migraine preventive treatment.”
Although AHS guidelines include three “should offer” medications for acute management of migraine in the ED – intravenous metoclopramide, intravenous prochlorperazine, and subcutaneous sumatriptan – two of the medications, subcutaneous sumatriptan and intravenous prochlorperazine, were not available in the urgent care pharmacy. “Of the level B migraine medications, only metoclopramide IV was in the pharmacy, and only 12.3% was given this at their urgent care visit,” the researchers said. “There was also likely undertreatment of nausea/vomiting; despite 39 patients with recorded nausea or vomiting with their migraine, less than half (46.2%) received an antiemetic at the visit,” including metoclopramide or ondansetron through oral or intravenous administration.
Future studies should look at headache and migraine visits at urgent care centers across the United States, the investigators suggested.
One of the authors of the study (Leslie Miller, MD) is the head of the NYU Langone Health Urgent Care Centers. Dr. Minen has received grant support, honoraria, or travel funds from the National Institutes of Health, the American Academy of Neurology, the American Brain Foundation, the National Multiple Sclerosis Society, the National Headache Foundation, the American Headache Society, Barnard College, and NYU. Dr. Minen is associate editor of Headache.
SOURCE: Minen MT et al. Headache. 2020;60(3):542-52.
according to a study published in the March issue of Headache. Pain and nausea or vomiting associated with migraine may go undertreated, and treatment may not be consistent with American Headache Society (AHS) guidelines for EDs, said Mia T. Minen, MD, of the department of neurology and population health at NYU Langone Health in New York and colleagues.
“Our study findings raise the question as to whether the patients with migraine in the urgent care setting should be managed similarly to the ED, and whether the AHS guidelines for the ED should be revisited and applied to urgent care,” the researchers noted.
Relative to the ED, urgent care centers may provide cost savings and emerge “as a preferred place for treatment for people with migraine, perhaps as they are potentially more quiet medical settings where people with migraine might expeditiously receive care,” the authors said.
Dr. Minen and colleagues conducted a retrospective chart review to assess migraine management at two urgent care centers in New York. They examined the number of urgent care visits for migraine, treatments used, and how closely clinicians followed the AHS recommendations for administration of antiemetic medication and triptans, among other outcomes.
The study population included adults diagnosed with migraine at the NYU Langone Medhattan Urgent Care center between Dec. 1, 2015, and Dec. 1, 2018, or at the NYU Langone Ambulatory Care Urgent Care West Side center between May 1, 2017, and Dec. 1, 2018. Of more than 32,000 urgent care visits during the study period, 78 patients received a migraine diagnosis. Patients with migraine had an average age of 32.5 years, and 79.5% were female. More than half had a documented history of migraine. Two of the patients (2.6%) had been to an emergency department for headache or migraine.
Less than half of the patients who presented with pain (46.6%) were given medication, most commonly ketorolac injection. Most patients (78.2%) received prescriptions, and 25.6% received a triptan prescription. About 60% of patients were told to follow up with a neurologist. In addition, 11.5% revisited urgent care with a migraine or headache or to request a prescription refill.
“Patients in this study appeared to be using the urgent care centers specifically for acute care,” the researchers said. “The patients generally had infrequent headaches and the majority would not have qualified for migraine preventive treatment.”
Although AHS guidelines include three “should offer” medications for acute management of migraine in the ED – intravenous metoclopramide, intravenous prochlorperazine, and subcutaneous sumatriptan – two of the medications, subcutaneous sumatriptan and intravenous prochlorperazine, were not available in the urgent care pharmacy. “Of the level B migraine medications, only metoclopramide IV was in the pharmacy, and only 12.3% was given this at their urgent care visit,” the researchers said. “There was also likely undertreatment of nausea/vomiting; despite 39 patients with recorded nausea or vomiting with their migraine, less than half (46.2%) received an antiemetic at the visit,” including metoclopramide or ondansetron through oral or intravenous administration.
Future studies should look at headache and migraine visits at urgent care centers across the United States, the investigators suggested.
One of the authors of the study (Leslie Miller, MD) is the head of the NYU Langone Health Urgent Care Centers. Dr. Minen has received grant support, honoraria, or travel funds from the National Institutes of Health, the American Academy of Neurology, the American Brain Foundation, the National Multiple Sclerosis Society, the National Headache Foundation, the American Headache Society, Barnard College, and NYU. Dr. Minen is associate editor of Headache.
SOURCE: Minen MT et al. Headache. 2020;60(3):542-52.
FROM HEADACHE








