User login
Researchers develop score to predict risk of stroke among migraineurs with aura
LOS ANGELES – The study on which the risk score is based was presented at the International Stroke Conference sponsored by the American Heart Association. Migraine with aura, for which younger women are at higher risk, increases the risk of ischemic stroke. “With our new risk-prediction tool, we could start identifying those at higher risk, treat their risk factors, and lower their risk of stroke,” said Souvik Sen, MD, MPH, professor and chair of neurology at the University of South Carolina in Columbia, in a press release.
Risk groups significantly discriminated stroke risk
To create the score, Dr. Sen and colleagues examined data from the ARIC (Atherosclerosis Risk in Communities) cohort, which includes community-dwelling people in Forsyth County, N.C.; Jackson, Miss.; Washington County, Md.; and the suburbs of Minneapolis. Researchers have been following the participants since 1987. From this population, Dr. Sen and colleagues identified 429 participants with a history of migraine with aura. Most of these participants were women aged 50-59 years at their first visit. The researchers analyzed the association between potential risk factors and ischemic stroke using Cox proportional hazards analysis.
Of the 429 participants, 31 had an ischemic stroke during a follow-up period of 20 years. Dr. Sen’s group created a risk score by identifying five risk factors for stroke and assigning them points in proportion to their influence (i.e., their regression coefficients). They assigned diabetes mellitus – 7 points; age older than 65 years – 5 points; heart rate variability (i.e., the standard deviation of all normal-to-normal RR intervals) – 3 points; hypertension – 3 points – and sex – 1 point. Then the researchers calculated risk scores for each patient and defined a low-risk group (from 0-4 points), a moderate-risk group (5-10 points), and a high-risk group (11-21 points).
After 18 years of follow-up, the incidence of stroke was 3% in the low-risk group, 8% in the moderate-risk group, and 34% in the high-risk group. The hazard ratio for ischemic stroke in the high-risk group, compared with the low-risk group, was 7.35. Kaplan Meier curves indicated that the risk-stratification groups significantly discriminated stroke risk among the sample. The risk score should be validated in an independent population cohort, said the investigators.
Dr. Sen and colleagues did not report any funding for this study. Investigators reported receiving grants from the National Institutes of Health, the American Heart Association, and the American Academy of Neurology.
Score may leave important variables unexamined
One mechanism through which migraine increases the risk of stroke is the constriction of blood vessels, said Louis R. Caplan, MD, professor of neurology at Harvard Medical School in Boston and member of the editorial advisory board of Neurology Reviews. Triptans, which many patients use to treat migraine, also cause vasoconstriction. In addition, migraine increases blood coagulation.
Although the risk score developed by Dr. Sen and colleagues accounts for various comorbidities, it may not apply equally to all patients. “As I understand it, they’re just using migraine with aura as a single factor,” said Dr. Caplan. Variables such as prolonged aura, frequent episodes, and aura-related deficit are associated with increased risk of stroke, but the risk score does not examine these factors.
Patients with severe, long-lasting attacks or attacks that involve weakness or aphasia should receive prophylactic treatment to prevent vasoconstriction, such as verapamil (Verelan), said Dr. Caplan. Antithrombotic agents such as aspirin also may be appropriate prophylaxis. Whether effective treatment of migraine with aura decreases the risk of stroke remains unknown.
SOURCE: Trivedi T et al. ISC 2020. Abstract WMP117.
LOS ANGELES – The study on which the risk score is based was presented at the International Stroke Conference sponsored by the American Heart Association. Migraine with aura, for which younger women are at higher risk, increases the risk of ischemic stroke. “With our new risk-prediction tool, we could start identifying those at higher risk, treat their risk factors, and lower their risk of stroke,” said Souvik Sen, MD, MPH, professor and chair of neurology at the University of South Carolina in Columbia, in a press release.
Risk groups significantly discriminated stroke risk
To create the score, Dr. Sen and colleagues examined data from the ARIC (Atherosclerosis Risk in Communities) cohort, which includes community-dwelling people in Forsyth County, N.C.; Jackson, Miss.; Washington County, Md.; and the suburbs of Minneapolis. Researchers have been following the participants since 1987. From this population, Dr. Sen and colleagues identified 429 participants with a history of migraine with aura. Most of these participants were women aged 50-59 years at their first visit. The researchers analyzed the association between potential risk factors and ischemic stroke using Cox proportional hazards analysis.
Of the 429 participants, 31 had an ischemic stroke during a follow-up period of 20 years. Dr. Sen’s group created a risk score by identifying five risk factors for stroke and assigning them points in proportion to their influence (i.e., their regression coefficients). They assigned diabetes mellitus – 7 points; age older than 65 years – 5 points; heart rate variability (i.e., the standard deviation of all normal-to-normal RR intervals) – 3 points; hypertension – 3 points – and sex – 1 point. Then the researchers calculated risk scores for each patient and defined a low-risk group (from 0-4 points), a moderate-risk group (5-10 points), and a high-risk group (11-21 points).
After 18 years of follow-up, the incidence of stroke was 3% in the low-risk group, 8% in the moderate-risk group, and 34% in the high-risk group. The hazard ratio for ischemic stroke in the high-risk group, compared with the low-risk group, was 7.35. Kaplan Meier curves indicated that the risk-stratification groups significantly discriminated stroke risk among the sample. The risk score should be validated in an independent population cohort, said the investigators.
Dr. Sen and colleagues did not report any funding for this study. Investigators reported receiving grants from the National Institutes of Health, the American Heart Association, and the American Academy of Neurology.
Score may leave important variables unexamined
One mechanism through which migraine increases the risk of stroke is the constriction of blood vessels, said Louis R. Caplan, MD, professor of neurology at Harvard Medical School in Boston and member of the editorial advisory board of Neurology Reviews. Triptans, which many patients use to treat migraine, also cause vasoconstriction. In addition, migraine increases blood coagulation.
Although the risk score developed by Dr. Sen and colleagues accounts for various comorbidities, it may not apply equally to all patients. “As I understand it, they’re just using migraine with aura as a single factor,” said Dr. Caplan. Variables such as prolonged aura, frequent episodes, and aura-related deficit are associated with increased risk of stroke, but the risk score does not examine these factors.
Patients with severe, long-lasting attacks or attacks that involve weakness or aphasia should receive prophylactic treatment to prevent vasoconstriction, such as verapamil (Verelan), said Dr. Caplan. Antithrombotic agents such as aspirin also may be appropriate prophylaxis. Whether effective treatment of migraine with aura decreases the risk of stroke remains unknown.
SOURCE: Trivedi T et al. ISC 2020. Abstract WMP117.
LOS ANGELES – The study on which the risk score is based was presented at the International Stroke Conference sponsored by the American Heart Association. Migraine with aura, for which younger women are at higher risk, increases the risk of ischemic stroke. “With our new risk-prediction tool, we could start identifying those at higher risk, treat their risk factors, and lower their risk of stroke,” said Souvik Sen, MD, MPH, professor and chair of neurology at the University of South Carolina in Columbia, in a press release.
Risk groups significantly discriminated stroke risk
To create the score, Dr. Sen and colleagues examined data from the ARIC (Atherosclerosis Risk in Communities) cohort, which includes community-dwelling people in Forsyth County, N.C.; Jackson, Miss.; Washington County, Md.; and the suburbs of Minneapolis. Researchers have been following the participants since 1987. From this population, Dr. Sen and colleagues identified 429 participants with a history of migraine with aura. Most of these participants were women aged 50-59 years at their first visit. The researchers analyzed the association between potential risk factors and ischemic stroke using Cox proportional hazards analysis.
Of the 429 participants, 31 had an ischemic stroke during a follow-up period of 20 years. Dr. Sen’s group created a risk score by identifying five risk factors for stroke and assigning them points in proportion to their influence (i.e., their regression coefficients). They assigned diabetes mellitus – 7 points; age older than 65 years – 5 points; heart rate variability (i.e., the standard deviation of all normal-to-normal RR intervals) – 3 points; hypertension – 3 points – and sex – 1 point. Then the researchers calculated risk scores for each patient and defined a low-risk group (from 0-4 points), a moderate-risk group (5-10 points), and a high-risk group (11-21 points).
After 18 years of follow-up, the incidence of stroke was 3% in the low-risk group, 8% in the moderate-risk group, and 34% in the high-risk group. The hazard ratio for ischemic stroke in the high-risk group, compared with the low-risk group, was 7.35. Kaplan Meier curves indicated that the risk-stratification groups significantly discriminated stroke risk among the sample. The risk score should be validated in an independent population cohort, said the investigators.
Dr. Sen and colleagues did not report any funding for this study. Investigators reported receiving grants from the National Institutes of Health, the American Heart Association, and the American Academy of Neurology.
Score may leave important variables unexamined
One mechanism through which migraine increases the risk of stroke is the constriction of blood vessels, said Louis R. Caplan, MD, professor of neurology at Harvard Medical School in Boston and member of the editorial advisory board of Neurology Reviews. Triptans, which many patients use to treat migraine, also cause vasoconstriction. In addition, migraine increases blood coagulation.
Although the risk score developed by Dr. Sen and colleagues accounts for various comorbidities, it may not apply equally to all patients. “As I understand it, they’re just using migraine with aura as a single factor,” said Dr. Caplan. Variables such as prolonged aura, frequent episodes, and aura-related deficit are associated with increased risk of stroke, but the risk score does not examine these factors.
Patients with severe, long-lasting attacks or attacks that involve weakness or aphasia should receive prophylactic treatment to prevent vasoconstriction, such as verapamil (Verelan), said Dr. Caplan. Antithrombotic agents such as aspirin also may be appropriate prophylaxis. Whether effective treatment of migraine with aura decreases the risk of stroke remains unknown.
SOURCE: Trivedi T et al. ISC 2020. Abstract WMP117.
REPORTING FROM ISC 2020
Scientific community ‘shocked’ by loss of MIND diet pioneer
Martha Clare Morris, ScD, a pioneer in research linking nutrition to brain health and a creator of the breakthrough MIND diet, has died of cancer at the age of 64.
Morris was a professor in the Department of Internal Medicine, assistant provost of community research, and director of the Rush Institute for Healthy Aging at Rush University, in Chicago, Illinois. She was also a director of the internal medicine department’s Section of Community Epidemiology.
Long-time friend and colleague Julie A. Schneider, MD, the Deborah R. and Edgar D. Jannotta Presidential Professor of Pathology and Neurological Sciences, Rush University Medical Center, described Morris as creative, passionate, and adventurous.
Her death was “a shock” to the scientific community, Schneider told Medscape Medical News.
“It’s a tragic loss in so many ways,” said Schneider, who is also associate director of the Rush Alzheimer’s Disease Center. She was a very well-respected nutritional epidemiologist and was passionate about her work; she had just so much unwavering commitment to it.
Diet, said Schneider, is “notoriously a hard thing to study” because “it’s so intertwined with lifestyle” and other factors that create “barriers” to conducting such research.
But Morris had a unique and creative talent for filtering out what might be the individual contribution of a particular modifiable risk factor, said Heather Snyder, PhD, vice president of medical and scientific relations, Alzheimer’s Association, who also knew Morris both personally and professionally.
“Humble” trailblazer
Morris’s pioneering research examined the connection between nutrition and the prevention of cognitive decline. Taking results from this research, she developed the MIND diet – a hybrid of the Mediterranean diet and the Dietary Approaches to Stop Hypertension – along with colleagues at both Rush and Harvard Universities.
The MIND diet – an acronym for Mediterranean-DASH Intervention for Neurodegenerative Delay – emphasizes brain-healthy foods, including leafy green vegetables, nuts, berries, chicken, fish, whole grains, beans, olive oil, and moderate amounts of red wine. The diet limits consumption of red meat, butter, margarine, and processed foods.
In 2015, Morris published her initial findings on the MIND diet in Alzheimer’s and Dementia. Reported by Medscape Medical News at that time, the study showed that the diet protected cardiovascular health and slowed cognitive decline in older individuals.
The excitement around the findings inspired Morris to write “Diet for the Mind,” which was published in 2017. The book summarizes the benefits of the MIND diet and includes brain-healthy recipes created by her daughter Laura, who is a chef. Despite many accolades, Morris was “humble” about this project, said Schneider.
“This was not about publicity and trying to get a book out; she wanted to see if this diet really was going to change people’s lives. She wanted to bring it into the community,” she said.
Proud legacy
Since 2017, Morris had led a large clinical trial of the effectiveness of the MIND diet in preventing cognitive decline. The first study of its kind, the trial received a $14.5 million grant from the National Institutes of Health (NIH). Results of this study are expected in 2021.
The MIND diet was ranked among the top 10 diets for five consecutive years in US News and World Report.
Morris’s nutrition-related research went beyond diets and examined the impact of individual nutrients. One of her studies, published in 2018 and reported by Medscape Medical News, suggested that the presence of folate, phylloquinone, and lutein – nutrients found in relatively large amounts in green leafy vegetables – may account for why consuming a daily serving of these vegetables slows cognitive decline.
One of the most recent studies from Morris’ group, published in January 2020 and reported by Medscape Medical News at that time, provided the first evidence that dietary flavonols, which are found in many fruits and vegetables, are associated with a significantly reduced risk for dementia.
What Morris did so well was to “look at the big picture” and “think about commonalities that cross nutritional components” of diets such as MIND, DASH, the Mediterranean diet, and the Nordic diet, which is similar to the Mediterranean diet but highlights local foods such as fish from Nordic regions, Snyder told Medscape Medical News.
Morris was instrumental in getting the Alzheimer’s Association’s US POINTER (US Study to Protect Brain Health Through Lifestyle Intervention to Reduce Risk) study off the ground. The 2-year clinical trial is testing whether combining a healthy diet with exercise, cognitive and social stimulation, and the management of cardiovascular conditions protects cognitive function in older adults who are at increased risk for cognitive decline.
This study will be part of her legacy, said Snyder.
“She will be remembered for her perseverance to get us to a place where we can be looking at nutrition as a modifiable risk factor and now testing it in trials that she helped to set up,” she said.
Even before her involvement with US POINTER, Morris had long been an active volunteer for the Alzheimer’s Association, said Snyder.
“She contributed significant time and expertise as we looked at the state of the evidence around nutrition and other lifestyle and behavioral interventions.”
We’ll ‘always have Paris’
While Morris was “truly passionate” about diet and health “both professionally and personally,” she also had a fun side, said Schneider. She remembers she and Morris had a chance meeting in Paris, where they spent an entire day going to museums and restaurants and just talking about life and their travels. To the end, they joked they would “always have Paris,” said Schneider.
She was also a loyal friend. Morris threw a baby shower when Schneider was pregnant, organizing every detail, despite her extremely busy schedule.
Family was another of Morris’s passions. Snyder recalls Morris’s face lighting up when she talked about her children and grandchildren. She also remembers her friend’s zest for life. “She had an energy that was contagious.”
Morris also loved the outdoors and was a keen adventurer. She once trained for weeks before a long bike trip with her daughter and would take a helicopter to access remote backcountry on hiking excursions.
“She wanted to try everything,” said Schneider.
An author or contributor to more than 80 articles in peer-reviewed journals, Morris also served two terms (from 2011 to 2013) as chair of the NIH’s Neurological, Aging and Musculoskeletal Epidemiology Study Section.
She left behind multiple grants for various studies. One unique study, said Schneider, investigated the relationship between iron and other metals in the brain and the neuropathology of Alzheimer disease.
“She was really in the prime of her career,” noted Schneider. “She had so much left to give and to offer, so this is tremendously sad.”
According to news reports, Morris (nee Chinn) grew up in Homewood, Illinois, and earned bachelor’s and master’s degrees in sociology from the University of Iowa in Iowa City, where she met her husband, James Morris. The two married in 1978 and had three children.
Morris completed a doctorate in epidemiology at the Harvard School of Public Health. James died in 2012, also from cancer. Morris passed away peacefully at her home on February 15.
Martha Clare Morris, ScD, a pioneer in research linking nutrition to brain health and a creator of the breakthrough MIND diet, has died of cancer at the age of 64.
Morris was a professor in the Department of Internal Medicine, assistant provost of community research, and director of the Rush Institute for Healthy Aging at Rush University, in Chicago, Illinois. She was also a director of the internal medicine department’s Section of Community Epidemiology.
Long-time friend and colleague Julie A. Schneider, MD, the Deborah R. and Edgar D. Jannotta Presidential Professor of Pathology and Neurological Sciences, Rush University Medical Center, described Morris as creative, passionate, and adventurous.
Her death was “a shock” to the scientific community, Schneider told Medscape Medical News.
“It’s a tragic loss in so many ways,” said Schneider, who is also associate director of the Rush Alzheimer’s Disease Center. She was a very well-respected nutritional epidemiologist and was passionate about her work; she had just so much unwavering commitment to it.
Diet, said Schneider, is “notoriously a hard thing to study” because “it’s so intertwined with lifestyle” and other factors that create “barriers” to conducting such research.
But Morris had a unique and creative talent for filtering out what might be the individual contribution of a particular modifiable risk factor, said Heather Snyder, PhD, vice president of medical and scientific relations, Alzheimer’s Association, who also knew Morris both personally and professionally.
“Humble” trailblazer
Morris’s pioneering research examined the connection between nutrition and the prevention of cognitive decline. Taking results from this research, she developed the MIND diet – a hybrid of the Mediterranean diet and the Dietary Approaches to Stop Hypertension – along with colleagues at both Rush and Harvard Universities.
The MIND diet – an acronym for Mediterranean-DASH Intervention for Neurodegenerative Delay – emphasizes brain-healthy foods, including leafy green vegetables, nuts, berries, chicken, fish, whole grains, beans, olive oil, and moderate amounts of red wine. The diet limits consumption of red meat, butter, margarine, and processed foods.
In 2015, Morris published her initial findings on the MIND diet in Alzheimer’s and Dementia. Reported by Medscape Medical News at that time, the study showed that the diet protected cardiovascular health and slowed cognitive decline in older individuals.
The excitement around the findings inspired Morris to write “Diet for the Mind,” which was published in 2017. The book summarizes the benefits of the MIND diet and includes brain-healthy recipes created by her daughter Laura, who is a chef. Despite many accolades, Morris was “humble” about this project, said Schneider.
“This was not about publicity and trying to get a book out; she wanted to see if this diet really was going to change people’s lives. She wanted to bring it into the community,” she said.
Proud legacy
Since 2017, Morris had led a large clinical trial of the effectiveness of the MIND diet in preventing cognitive decline. The first study of its kind, the trial received a $14.5 million grant from the National Institutes of Health (NIH). Results of this study are expected in 2021.
The MIND diet was ranked among the top 10 diets for five consecutive years in US News and World Report.
Morris’s nutrition-related research went beyond diets and examined the impact of individual nutrients. One of her studies, published in 2018 and reported by Medscape Medical News, suggested that the presence of folate, phylloquinone, and lutein – nutrients found in relatively large amounts in green leafy vegetables – may account for why consuming a daily serving of these vegetables slows cognitive decline.
One of the most recent studies from Morris’ group, published in January 2020 and reported by Medscape Medical News at that time, provided the first evidence that dietary flavonols, which are found in many fruits and vegetables, are associated with a significantly reduced risk for dementia.
What Morris did so well was to “look at the big picture” and “think about commonalities that cross nutritional components” of diets such as MIND, DASH, the Mediterranean diet, and the Nordic diet, which is similar to the Mediterranean diet but highlights local foods such as fish from Nordic regions, Snyder told Medscape Medical News.
Morris was instrumental in getting the Alzheimer’s Association’s US POINTER (US Study to Protect Brain Health Through Lifestyle Intervention to Reduce Risk) study off the ground. The 2-year clinical trial is testing whether combining a healthy diet with exercise, cognitive and social stimulation, and the management of cardiovascular conditions protects cognitive function in older adults who are at increased risk for cognitive decline.
This study will be part of her legacy, said Snyder.
“She will be remembered for her perseverance to get us to a place where we can be looking at nutrition as a modifiable risk factor and now testing it in trials that she helped to set up,” she said.
Even before her involvement with US POINTER, Morris had long been an active volunteer for the Alzheimer’s Association, said Snyder.
“She contributed significant time and expertise as we looked at the state of the evidence around nutrition and other lifestyle and behavioral interventions.”
We’ll ‘always have Paris’
While Morris was “truly passionate” about diet and health “both professionally and personally,” she also had a fun side, said Schneider. She remembers she and Morris had a chance meeting in Paris, where they spent an entire day going to museums and restaurants and just talking about life and their travels. To the end, they joked they would “always have Paris,” said Schneider.
She was also a loyal friend. Morris threw a baby shower when Schneider was pregnant, organizing every detail, despite her extremely busy schedule.
Family was another of Morris’s passions. Snyder recalls Morris’s face lighting up when she talked about her children and grandchildren. She also remembers her friend’s zest for life. “She had an energy that was contagious.”
Morris also loved the outdoors and was a keen adventurer. She once trained for weeks before a long bike trip with her daughter and would take a helicopter to access remote backcountry on hiking excursions.
“She wanted to try everything,” said Schneider.
An author or contributor to more than 80 articles in peer-reviewed journals, Morris also served two terms (from 2011 to 2013) as chair of the NIH’s Neurological, Aging and Musculoskeletal Epidemiology Study Section.
She left behind multiple grants for various studies. One unique study, said Schneider, investigated the relationship between iron and other metals in the brain and the neuropathology of Alzheimer disease.
“She was really in the prime of her career,” noted Schneider. “She had so much left to give and to offer, so this is tremendously sad.”
According to news reports, Morris (nee Chinn) grew up in Homewood, Illinois, and earned bachelor’s and master’s degrees in sociology from the University of Iowa in Iowa City, where she met her husband, James Morris. The two married in 1978 and had three children.
Morris completed a doctorate in epidemiology at the Harvard School of Public Health. James died in 2012, also from cancer. Morris passed away peacefully at her home on February 15.
Martha Clare Morris, ScD, a pioneer in research linking nutrition to brain health and a creator of the breakthrough MIND diet, has died of cancer at the age of 64.
Morris was a professor in the Department of Internal Medicine, assistant provost of community research, and director of the Rush Institute for Healthy Aging at Rush University, in Chicago, Illinois. She was also a director of the internal medicine department’s Section of Community Epidemiology.
Long-time friend and colleague Julie A. Schneider, MD, the Deborah R. and Edgar D. Jannotta Presidential Professor of Pathology and Neurological Sciences, Rush University Medical Center, described Morris as creative, passionate, and adventurous.
Her death was “a shock” to the scientific community, Schneider told Medscape Medical News.
“It’s a tragic loss in so many ways,” said Schneider, who is also associate director of the Rush Alzheimer’s Disease Center. She was a very well-respected nutritional epidemiologist and was passionate about her work; she had just so much unwavering commitment to it.
Diet, said Schneider, is “notoriously a hard thing to study” because “it’s so intertwined with lifestyle” and other factors that create “barriers” to conducting such research.
But Morris had a unique and creative talent for filtering out what might be the individual contribution of a particular modifiable risk factor, said Heather Snyder, PhD, vice president of medical and scientific relations, Alzheimer’s Association, who also knew Morris both personally and professionally.
“Humble” trailblazer
Morris’s pioneering research examined the connection between nutrition and the prevention of cognitive decline. Taking results from this research, she developed the MIND diet – a hybrid of the Mediterranean diet and the Dietary Approaches to Stop Hypertension – along with colleagues at both Rush and Harvard Universities.
The MIND diet – an acronym for Mediterranean-DASH Intervention for Neurodegenerative Delay – emphasizes brain-healthy foods, including leafy green vegetables, nuts, berries, chicken, fish, whole grains, beans, olive oil, and moderate amounts of red wine. The diet limits consumption of red meat, butter, margarine, and processed foods.
In 2015, Morris published her initial findings on the MIND diet in Alzheimer’s and Dementia. Reported by Medscape Medical News at that time, the study showed that the diet protected cardiovascular health and slowed cognitive decline in older individuals.
The excitement around the findings inspired Morris to write “Diet for the Mind,” which was published in 2017. The book summarizes the benefits of the MIND diet and includes brain-healthy recipes created by her daughter Laura, who is a chef. Despite many accolades, Morris was “humble” about this project, said Schneider.
“This was not about publicity and trying to get a book out; she wanted to see if this diet really was going to change people’s lives. She wanted to bring it into the community,” she said.
Proud legacy
Since 2017, Morris had led a large clinical trial of the effectiveness of the MIND diet in preventing cognitive decline. The first study of its kind, the trial received a $14.5 million grant from the National Institutes of Health (NIH). Results of this study are expected in 2021.
The MIND diet was ranked among the top 10 diets for five consecutive years in US News and World Report.
Morris’s nutrition-related research went beyond diets and examined the impact of individual nutrients. One of her studies, published in 2018 and reported by Medscape Medical News, suggested that the presence of folate, phylloquinone, and lutein – nutrients found in relatively large amounts in green leafy vegetables – may account for why consuming a daily serving of these vegetables slows cognitive decline.
One of the most recent studies from Morris’ group, published in January 2020 and reported by Medscape Medical News at that time, provided the first evidence that dietary flavonols, which are found in many fruits and vegetables, are associated with a significantly reduced risk for dementia.
What Morris did so well was to “look at the big picture” and “think about commonalities that cross nutritional components” of diets such as MIND, DASH, the Mediterranean diet, and the Nordic diet, which is similar to the Mediterranean diet but highlights local foods such as fish from Nordic regions, Snyder told Medscape Medical News.
Morris was instrumental in getting the Alzheimer’s Association’s US POINTER (US Study to Protect Brain Health Through Lifestyle Intervention to Reduce Risk) study off the ground. The 2-year clinical trial is testing whether combining a healthy diet with exercise, cognitive and social stimulation, and the management of cardiovascular conditions protects cognitive function in older adults who are at increased risk for cognitive decline.
This study will be part of her legacy, said Snyder.
“She will be remembered for her perseverance to get us to a place where we can be looking at nutrition as a modifiable risk factor and now testing it in trials that she helped to set up,” she said.
Even before her involvement with US POINTER, Morris had long been an active volunteer for the Alzheimer’s Association, said Snyder.
“She contributed significant time and expertise as we looked at the state of the evidence around nutrition and other lifestyle and behavioral interventions.”
We’ll ‘always have Paris’
While Morris was “truly passionate” about diet and health “both professionally and personally,” she also had a fun side, said Schneider. She remembers she and Morris had a chance meeting in Paris, where they spent an entire day going to museums and restaurants and just talking about life and their travels. To the end, they joked they would “always have Paris,” said Schneider.
She was also a loyal friend. Morris threw a baby shower when Schneider was pregnant, organizing every detail, despite her extremely busy schedule.
Family was another of Morris’s passions. Snyder recalls Morris’s face lighting up when she talked about her children and grandchildren. She also remembers her friend’s zest for life. “She had an energy that was contagious.”
Morris also loved the outdoors and was a keen adventurer. She once trained for weeks before a long bike trip with her daughter and would take a helicopter to access remote backcountry on hiking excursions.
“She wanted to try everything,” said Schneider.
An author or contributor to more than 80 articles in peer-reviewed journals, Morris also served two terms (from 2011 to 2013) as chair of the NIH’s Neurological, Aging and Musculoskeletal Epidemiology Study Section.
She left behind multiple grants for various studies. One unique study, said Schneider, investigated the relationship between iron and other metals in the brain and the neuropathology of Alzheimer disease.
“She was really in the prime of her career,” noted Schneider. “She had so much left to give and to offer, so this is tremendously sad.”
According to news reports, Morris (nee Chinn) grew up in Homewood, Illinois, and earned bachelor’s and master’s degrees in sociology from the University of Iowa in Iowa City, where she met her husband, James Morris. The two married in 1978 and had three children.
Morris completed a doctorate in epidemiology at the Harvard School of Public Health. James died in 2012, also from cancer. Morris passed away peacefully at her home on February 15.
Manual dexterity may decline more rapidly in pediatric-onset MS
WEST PALM BEACH, FLA. – (AOMS), according to an analysis presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
When MS onset occurs before the patient is age 18 years, the patient is considered to have POMS. Compared with AOMS, POMS is less prevalent and has distinct features. To determine whether changes in physical performance differ between POMS and AOMS, Sarah M. Planchon, PhD, a project scientist at the Mellen Center for MS at the Cleveland Clinic, and colleagues analyzed data cut 9 from the MS PATHS (MS Partners Advancing Technology and Health Solutions) initiative. As part of this initiative, which is sponsored by Biogen, investigators collect MS performance measures longitudinally at each patient visit. Among these measures are the manual dexterity test (MDT), an iPad version of the Nine-Hole Peg Test, and the walking speed test (WST), which is the iPad version of the Timed 25-Foot Walk.
Dr. Planchon and colleagues matched each patient with POMS to five patients with AOMS according to disease duration. They calculated descriptive statistics for the sample and performed Tukey’s honestly significant difference test to compare patient groups on several categorical variables.
Overall, function was better in POMS than in AOMS
The investigators included 3 years’ worth of data from 6,457 patients in their analysis. The average age was approximately 50 years for patients with AOMS and 31 years for patients with POMS. The time elapsed since diagnosis was approximately 14 years in the AOMS group and 17 years in the POMS group. The proportion of female patients was about 74% in the AOMS group and 73% in the POMS group. Compared with the AOMS group, the POMS group had higher proportions of patients who were Asian (0.5% vs 2.6%), black (9.3% vs 11.5%), and other race (2.8% vs 9.3%).
Overall, patients with POMS performed better than patients with AOMS by 1.39 seconds on the MDT and by 0.79 seconds on the WST. Regression analyses indicated that with increasing age, patients with AOMS declined more quickly on the MDT and the WST than patients with POMS did. When the investigators stratified the results by disease duration, however, patients with POMS declined more rapidly on the MDT than did patients with AOMS. There was no significant difference between groups in WST in this analysis. When Dr. Planchon and colleagues performed linear regression and adjusted for variables such as age, sex, race, education, insurance, employment, MS phenotype, disease duration, number of relapses, and Patient-Determined Disease Steps (PDDS), the MS onset type did not significantly affect outcomes. Age, sex, PDDS, and MS type were significant covariates for both tests.
The role of occupational and physical therapy
“POMS patients tend to have a greater dysfunction of the cerebellar and brainstem regions of the brain, both of which may impact motor skills to a greater degree than other regions of the brain,” said Dr. Planchon. The increased rate of manual impairment in POMS, compared with AOMS, does not necessarily indicate more severe disease, she added. Getting a true picture of disease severity would require consideration of factors such as ambulation, cognitive functioning, vision, fatigue, and depression.
“We would recommend introducing POMS patients to occupational and physical therapy early in their disease course, before significant deficits accrue,” said Dr. Planchon. “Early familiarity with rehabilitation services should help the patient and family optimize what exercises are being done to improve and maintain function.”
The optimal pharmacologic treatment for POMS is unknown. One therapy (i.e., fingolimod) has Food and Drug Administration approval, and clinical trials of other treatments are ongoing. Some MS treatments not indicated for a pediatric population are used off label in children.
“We plan to delve deeper into the data set, including using regression modeling to try to better define differences between individuals with POMS and AOMS that may lead to the functional outcome changes we have already observed,” said Dr. Planchon. “We also plan to investigate further the impact of POMS on cognition and quality of life measures and to better understand disease-modifying therapy prescribing patterns and benefits in individuals with POMS. We will look for associations in the MRI imaging findings and various biomarkers to help us understand the disease process in this special population of MS.”
Dr. Planchon has received research support from the Guthy-Jackson Charitable Foundation. Her coinvestigators received funding from Biogen, Genentech, Genzyme, MedImmune, Novartis, Serono, and Teva.
SOURCE: Planchon SM et al. ACTRIMS 2020. Abstract P043.
WEST PALM BEACH, FLA. – (AOMS), according to an analysis presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
When MS onset occurs before the patient is age 18 years, the patient is considered to have POMS. Compared with AOMS, POMS is less prevalent and has distinct features. To determine whether changes in physical performance differ between POMS and AOMS, Sarah M. Planchon, PhD, a project scientist at the Mellen Center for MS at the Cleveland Clinic, and colleagues analyzed data cut 9 from the MS PATHS (MS Partners Advancing Technology and Health Solutions) initiative. As part of this initiative, which is sponsored by Biogen, investigators collect MS performance measures longitudinally at each patient visit. Among these measures are the manual dexterity test (MDT), an iPad version of the Nine-Hole Peg Test, and the walking speed test (WST), which is the iPad version of the Timed 25-Foot Walk.
Dr. Planchon and colleagues matched each patient with POMS to five patients with AOMS according to disease duration. They calculated descriptive statistics for the sample and performed Tukey’s honestly significant difference test to compare patient groups on several categorical variables.
Overall, function was better in POMS than in AOMS
The investigators included 3 years’ worth of data from 6,457 patients in their analysis. The average age was approximately 50 years for patients with AOMS and 31 years for patients with POMS. The time elapsed since diagnosis was approximately 14 years in the AOMS group and 17 years in the POMS group. The proportion of female patients was about 74% in the AOMS group and 73% in the POMS group. Compared with the AOMS group, the POMS group had higher proportions of patients who were Asian (0.5% vs 2.6%), black (9.3% vs 11.5%), and other race (2.8% vs 9.3%).
Overall, patients with POMS performed better than patients with AOMS by 1.39 seconds on the MDT and by 0.79 seconds on the WST. Regression analyses indicated that with increasing age, patients with AOMS declined more quickly on the MDT and the WST than patients with POMS did. When the investigators stratified the results by disease duration, however, patients with POMS declined more rapidly on the MDT than did patients with AOMS. There was no significant difference between groups in WST in this analysis. When Dr. Planchon and colleagues performed linear regression and adjusted for variables such as age, sex, race, education, insurance, employment, MS phenotype, disease duration, number of relapses, and Patient-Determined Disease Steps (PDDS), the MS onset type did not significantly affect outcomes. Age, sex, PDDS, and MS type were significant covariates for both tests.
The role of occupational and physical therapy
“POMS patients tend to have a greater dysfunction of the cerebellar and brainstem regions of the brain, both of which may impact motor skills to a greater degree than other regions of the brain,” said Dr. Planchon. The increased rate of manual impairment in POMS, compared with AOMS, does not necessarily indicate more severe disease, she added. Getting a true picture of disease severity would require consideration of factors such as ambulation, cognitive functioning, vision, fatigue, and depression.
“We would recommend introducing POMS patients to occupational and physical therapy early in their disease course, before significant deficits accrue,” said Dr. Planchon. “Early familiarity with rehabilitation services should help the patient and family optimize what exercises are being done to improve and maintain function.”
The optimal pharmacologic treatment for POMS is unknown. One therapy (i.e., fingolimod) has Food and Drug Administration approval, and clinical trials of other treatments are ongoing. Some MS treatments not indicated for a pediatric population are used off label in children.
“We plan to delve deeper into the data set, including using regression modeling to try to better define differences between individuals with POMS and AOMS that may lead to the functional outcome changes we have already observed,” said Dr. Planchon. “We also plan to investigate further the impact of POMS on cognition and quality of life measures and to better understand disease-modifying therapy prescribing patterns and benefits in individuals with POMS. We will look for associations in the MRI imaging findings and various biomarkers to help us understand the disease process in this special population of MS.”
Dr. Planchon has received research support from the Guthy-Jackson Charitable Foundation. Her coinvestigators received funding from Biogen, Genentech, Genzyme, MedImmune, Novartis, Serono, and Teva.
SOURCE: Planchon SM et al. ACTRIMS 2020. Abstract P043.
WEST PALM BEACH, FLA. – (AOMS), according to an analysis presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
When MS onset occurs before the patient is age 18 years, the patient is considered to have POMS. Compared with AOMS, POMS is less prevalent and has distinct features. To determine whether changes in physical performance differ between POMS and AOMS, Sarah M. Planchon, PhD, a project scientist at the Mellen Center for MS at the Cleveland Clinic, and colleagues analyzed data cut 9 from the MS PATHS (MS Partners Advancing Technology and Health Solutions) initiative. As part of this initiative, which is sponsored by Biogen, investigators collect MS performance measures longitudinally at each patient visit. Among these measures are the manual dexterity test (MDT), an iPad version of the Nine-Hole Peg Test, and the walking speed test (WST), which is the iPad version of the Timed 25-Foot Walk.
Dr. Planchon and colleagues matched each patient with POMS to five patients with AOMS according to disease duration. They calculated descriptive statistics for the sample and performed Tukey’s honestly significant difference test to compare patient groups on several categorical variables.
Overall, function was better in POMS than in AOMS
The investigators included 3 years’ worth of data from 6,457 patients in their analysis. The average age was approximately 50 years for patients with AOMS and 31 years for patients with POMS. The time elapsed since diagnosis was approximately 14 years in the AOMS group and 17 years in the POMS group. The proportion of female patients was about 74% in the AOMS group and 73% in the POMS group. Compared with the AOMS group, the POMS group had higher proportions of patients who were Asian (0.5% vs 2.6%), black (9.3% vs 11.5%), and other race (2.8% vs 9.3%).
Overall, patients with POMS performed better than patients with AOMS by 1.39 seconds on the MDT and by 0.79 seconds on the WST. Regression analyses indicated that with increasing age, patients with AOMS declined more quickly on the MDT and the WST than patients with POMS did. When the investigators stratified the results by disease duration, however, patients with POMS declined more rapidly on the MDT than did patients with AOMS. There was no significant difference between groups in WST in this analysis. When Dr. Planchon and colleagues performed linear regression and adjusted for variables such as age, sex, race, education, insurance, employment, MS phenotype, disease duration, number of relapses, and Patient-Determined Disease Steps (PDDS), the MS onset type did not significantly affect outcomes. Age, sex, PDDS, and MS type were significant covariates for both tests.
The role of occupational and physical therapy
“POMS patients tend to have a greater dysfunction of the cerebellar and brainstem regions of the brain, both of which may impact motor skills to a greater degree than other regions of the brain,” said Dr. Planchon. The increased rate of manual impairment in POMS, compared with AOMS, does not necessarily indicate more severe disease, she added. Getting a true picture of disease severity would require consideration of factors such as ambulation, cognitive functioning, vision, fatigue, and depression.
“We would recommend introducing POMS patients to occupational and physical therapy early in their disease course, before significant deficits accrue,” said Dr. Planchon. “Early familiarity with rehabilitation services should help the patient and family optimize what exercises are being done to improve and maintain function.”
The optimal pharmacologic treatment for POMS is unknown. One therapy (i.e., fingolimod) has Food and Drug Administration approval, and clinical trials of other treatments are ongoing. Some MS treatments not indicated for a pediatric population are used off label in children.
“We plan to delve deeper into the data set, including using regression modeling to try to better define differences between individuals with POMS and AOMS that may lead to the functional outcome changes we have already observed,” said Dr. Planchon. “We also plan to investigate further the impact of POMS on cognition and quality of life measures and to better understand disease-modifying therapy prescribing patterns and benefits in individuals with POMS. We will look for associations in the MRI imaging findings and various biomarkers to help us understand the disease process in this special population of MS.”
Dr. Planchon has received research support from the Guthy-Jackson Charitable Foundation. Her coinvestigators received funding from Biogen, Genentech, Genzyme, MedImmune, Novartis, Serono, and Teva.
SOURCE: Planchon SM et al. ACTRIMS 2020. Abstract P043.
REPORTING FROM ACTRIMS FORUM 2020
DMT use is common in older patients with MS
WEST PALM BEACH, FLA. –
MS disease activity typically declines with age. At the same time, evidence to support the efficacy of MS drugs in older patients is limited, said Yinan Zhang, MD, a researcher at Icahn School of Medicine at Mount Sinai, New York. Clinical trials have tended to enroll younger patients and to include only patients with active disease, which is not representative of most older patients in the real world, Dr. Zhang said.
“DMTs for MS may be less efficacious in the elderly, especially in the absence of active disease, yet real-world prescribing patterns still show widespread use of DMTs in older patients,” Dr. Zhang and colleagues said. Physicians may be able to use the presence of disease activity to identify older patients who should receive therapy. “Continuing DMTs in elderly patients who have no evidence of disease activity should be questioned rather than accepted,” they said at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To investigate whether age affects the efficacy of DMTs in patients with relapsing-remitting MS and how often DMTs are used in different age groups, Dr. Zhang and coinvestigators conducted a meta-analysis of group-level data from clinical trial, analyzed individual-level data from one of the trials, and reviewed survey data from two registries.
The meta-analysis included 26 clinical trials of 13 DMTs with more than 12,400 patients. Participants had an average age of about 37 years. “An age-dependent relationship of DMTs on relapse rate in RRMS [relapsing-remitting MS] cannot be established with currently published aggregate summary data,” the researchers said. “The meta-analysis was limited by the use of group-level data resulting in a narrow range of mean age.”
In an effort to overcome the limitations of group-level data, they analyzed individual-level data from approximately 1,000 patients in the CombiRx trial, which compared interferon beta-1a plus glatiramer acetate versus the agents alone. Thirty-seven of the patients were aged 55 years or older. The results suggest that each “1-year increase in baseline age was associated with a 3.2% reduction in the odds of having a relapse” during the trial, the investigators said. Change in annualized relapse rate was not significantly associated with age group, which may have resulted from “enrollment criteria selecting for patients with active disease, where DMTs are expected to show the greatest efficacy,” the researchers said.
Finally, Dr. Zhang and colleagues reviewed data on DMT use by age group from the North American Research Committee on Multiple Sclerosis (NARCOMS) and the Multiple Sclerosis Surveillance Registry (MSSR) from Veterans Affairs. In a 2018 survey of nearly 7,000 patients in the NARCOMS registry, 39.2% of patients older than 60 years were taking a DMT, including 44.5% of patients aged 61-70, 28.6% of patients aged 71-80, and 11% of patients aged 81 years and older. In comparison, about 62% of patients aged 41-50 years were taking DMT.
A 2019 survey of about 1,700 veterans in the MSSR found that 36.3% of patients older than 60 years were taking a DMT, including 41.1% of patients aged 61-70, 27.2% of patients aged 71-80, and 7.1% of patients aged 81 years and older. Among patients aged 41-50 years, more than 72% were taking a DMT. “The continued use of DMTs in the elderly may be the result of the perceived notion that disease inactivity is due to the effect of DMTs rather than the natural disease course with aging,” they said.
Dr. Zhang had no relevant disclosures. Coauthors disclosed consulting for and grant support from various pharmaceutical companies.
SOURCE: Zhang Y et al. ACTRIMS Forum 2020. Abstract P263.
WEST PALM BEACH, FLA. –
MS disease activity typically declines with age. At the same time, evidence to support the efficacy of MS drugs in older patients is limited, said Yinan Zhang, MD, a researcher at Icahn School of Medicine at Mount Sinai, New York. Clinical trials have tended to enroll younger patients and to include only patients with active disease, which is not representative of most older patients in the real world, Dr. Zhang said.
“DMTs for MS may be less efficacious in the elderly, especially in the absence of active disease, yet real-world prescribing patterns still show widespread use of DMTs in older patients,” Dr. Zhang and colleagues said. Physicians may be able to use the presence of disease activity to identify older patients who should receive therapy. “Continuing DMTs in elderly patients who have no evidence of disease activity should be questioned rather than accepted,” they said at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To investigate whether age affects the efficacy of DMTs in patients with relapsing-remitting MS and how often DMTs are used in different age groups, Dr. Zhang and coinvestigators conducted a meta-analysis of group-level data from clinical trial, analyzed individual-level data from one of the trials, and reviewed survey data from two registries.
The meta-analysis included 26 clinical trials of 13 DMTs with more than 12,400 patients. Participants had an average age of about 37 years. “An age-dependent relationship of DMTs on relapse rate in RRMS [relapsing-remitting MS] cannot be established with currently published aggregate summary data,” the researchers said. “The meta-analysis was limited by the use of group-level data resulting in a narrow range of mean age.”
In an effort to overcome the limitations of group-level data, they analyzed individual-level data from approximately 1,000 patients in the CombiRx trial, which compared interferon beta-1a plus glatiramer acetate versus the agents alone. Thirty-seven of the patients were aged 55 years or older. The results suggest that each “1-year increase in baseline age was associated with a 3.2% reduction in the odds of having a relapse” during the trial, the investigators said. Change in annualized relapse rate was not significantly associated with age group, which may have resulted from “enrollment criteria selecting for patients with active disease, where DMTs are expected to show the greatest efficacy,” the researchers said.
Finally, Dr. Zhang and colleagues reviewed data on DMT use by age group from the North American Research Committee on Multiple Sclerosis (NARCOMS) and the Multiple Sclerosis Surveillance Registry (MSSR) from Veterans Affairs. In a 2018 survey of nearly 7,000 patients in the NARCOMS registry, 39.2% of patients older than 60 years were taking a DMT, including 44.5% of patients aged 61-70, 28.6% of patients aged 71-80, and 11% of patients aged 81 years and older. In comparison, about 62% of patients aged 41-50 years were taking DMT.
A 2019 survey of about 1,700 veterans in the MSSR found that 36.3% of patients older than 60 years were taking a DMT, including 41.1% of patients aged 61-70, 27.2% of patients aged 71-80, and 7.1% of patients aged 81 years and older. Among patients aged 41-50 years, more than 72% were taking a DMT. “The continued use of DMTs in the elderly may be the result of the perceived notion that disease inactivity is due to the effect of DMTs rather than the natural disease course with aging,” they said.
Dr. Zhang had no relevant disclosures. Coauthors disclosed consulting for and grant support from various pharmaceutical companies.
SOURCE: Zhang Y et al. ACTRIMS Forum 2020. Abstract P263.
WEST PALM BEACH, FLA. –
MS disease activity typically declines with age. At the same time, evidence to support the efficacy of MS drugs in older patients is limited, said Yinan Zhang, MD, a researcher at Icahn School of Medicine at Mount Sinai, New York. Clinical trials have tended to enroll younger patients and to include only patients with active disease, which is not representative of most older patients in the real world, Dr. Zhang said.
“DMTs for MS may be less efficacious in the elderly, especially in the absence of active disease, yet real-world prescribing patterns still show widespread use of DMTs in older patients,” Dr. Zhang and colleagues said. Physicians may be able to use the presence of disease activity to identify older patients who should receive therapy. “Continuing DMTs in elderly patients who have no evidence of disease activity should be questioned rather than accepted,” they said at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To investigate whether age affects the efficacy of DMTs in patients with relapsing-remitting MS and how often DMTs are used in different age groups, Dr. Zhang and coinvestigators conducted a meta-analysis of group-level data from clinical trial, analyzed individual-level data from one of the trials, and reviewed survey data from two registries.
The meta-analysis included 26 clinical trials of 13 DMTs with more than 12,400 patients. Participants had an average age of about 37 years. “An age-dependent relationship of DMTs on relapse rate in RRMS [relapsing-remitting MS] cannot be established with currently published aggregate summary data,” the researchers said. “The meta-analysis was limited by the use of group-level data resulting in a narrow range of mean age.”
In an effort to overcome the limitations of group-level data, they analyzed individual-level data from approximately 1,000 patients in the CombiRx trial, which compared interferon beta-1a plus glatiramer acetate versus the agents alone. Thirty-seven of the patients were aged 55 years or older. The results suggest that each “1-year increase in baseline age was associated with a 3.2% reduction in the odds of having a relapse” during the trial, the investigators said. Change in annualized relapse rate was not significantly associated with age group, which may have resulted from “enrollment criteria selecting for patients with active disease, where DMTs are expected to show the greatest efficacy,” the researchers said.
Finally, Dr. Zhang and colleagues reviewed data on DMT use by age group from the North American Research Committee on Multiple Sclerosis (NARCOMS) and the Multiple Sclerosis Surveillance Registry (MSSR) from Veterans Affairs. In a 2018 survey of nearly 7,000 patients in the NARCOMS registry, 39.2% of patients older than 60 years were taking a DMT, including 44.5% of patients aged 61-70, 28.6% of patients aged 71-80, and 11% of patients aged 81 years and older. In comparison, about 62% of patients aged 41-50 years were taking DMT.
A 2019 survey of about 1,700 veterans in the MSSR found that 36.3% of patients older than 60 years were taking a DMT, including 41.1% of patients aged 61-70, 27.2% of patients aged 71-80, and 7.1% of patients aged 81 years and older. Among patients aged 41-50 years, more than 72% were taking a DMT. “The continued use of DMTs in the elderly may be the result of the perceived notion that disease inactivity is due to the effect of DMTs rather than the natural disease course with aging,” they said.
Dr. Zhang had no relevant disclosures. Coauthors disclosed consulting for and grant support from various pharmaceutical companies.
SOURCE: Zhang Y et al. ACTRIMS Forum 2020. Abstract P263.
REPORTING FROM ACTRIMS FORUM 2020
American Headache Society updates guideline on neuroimaging for migraine
Migraine with atypical features may require neuroimaging, according to the guideline. These include an unusual aura; change in clinical features; a first or worst migraine; a migraine that presents with brainstem aura, confusion, or motor manifestation; migraine accompaniments in later life; headaches that are side-locked or posttraumatic; and aura that presents without headache.
Assessing the evidence
The recommendation to avoid MRI or CT in otherwise neurologically normal patients with migraine carried a grade A recommendation from the American Headache Society, while the specific considerations for neuroimaging was based on consensus and carried a grade C recommendation, according to lead author Randolph W. Evans, MD, of the department of neurology at Baylor College of Medicine in Houston, and colleagues.
The recommendations, published in the journal Headache (2020 Feb;60(2):318-36), came from a systematic review of 23 studies of adults at least 18 years old who underwent MRI or CT during outpatient treatment for migraine between 1973 and 2018. Ten studies looked at CT neuroimaging in patients with migraine, nine studies examined MRI neuroimaging alone in patients with migraine, and four studies contained adults with headache or migraine who underwent either MRI or CT. The majority of studies analyzed were retrospective or cross-sectional in nature, while four studies were prospective observational studies.
Dr. Evans and colleagues noted that neuroimaging for patients with suspected migraine is ordered for a variety of reasons, such as excluding conditions that aren’t migraine, diagnostic certainty, cognitive bias, practice workflow, medicolegal concerns, addressing patient and family anxiety, and addressing clinician anxiety. Neuroimaging also can be costly, they said, adding up to an estimated $1 billion annually according to one study, and can lead to additional testing from findings that may not be clinically significant.
Good advice, with caveats
In an interview, Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews, said that while he generally does not like broad guideline recommendations, the recommendation made by the American Headache Society to avoid neuroimaging in patients with a normal neurological examination without any atypical features and red flags “takes most of the important factors into consideration and will work almost all the time.” The recommendation made by consensus for specific considerations of neuroimaging was issued by top headache specialists in the United States who reviewed the data, and it is unlikely a patient with a migraine as diagnosed by the International Classification of Headache Disorders with a normal neurological examination would have a significant abnormality that would appear with imaging, Dr. Rapoport said.
“If everyone caring for migraine patients knew these recommendations, and used them unless the patients fit the exclusions mentioned, we would have more efficient clinical practice and save lots of money on unnecessary scanning,” he said.
However, Dr. Rapoport, clinical professor of neurology at the University of California, Los Angeles, founder of the New England Center for Headache, and past president of The International Headache Society, said that not all clinicians will be convinced by the American Headache Society’s recommendations.
“Various third parties often jump on society recommendations or guidelines and prevent smart clinicians from doing what they need to do when they want to disregard the recommendation or guideline,” he explained. “More importantly, if a physician feels the need to think out of the box and image a patient without a clear reason, and the patient cannot pay for the scan when a medical insurance company refuses to authorize it, there can be a bad result if the patient does not get the study.”
Dr. Rapoport noted that the guideline does not address situations where neuroimaging may not pick up conditions that lead to migraine, such as a subarachnoid or subdural hemorrhage, reversible cerebral vasoconstriction syndrome, or early aspects of low cerebrospinal fluid pressure syndrome. Anxiety on the part of the patient or the clinician is another area that can be addressed by future research, he said.
“If the clinician does a good job of explaining the odds of anything significant being found with a typical migraine history and normal examination, and the patient says [they] need an MRI with contrast to be sure, it will be difficult to dissuade them,” said Dr. Rapoport. “If you don’t order one, they will find a way to get one. If it is abnormal, you could be in trouble. Also, if the clinician has no good reason to do a scan but has anxiety about what is being missed, it will probably get done.”
There was no funding source for the guidelines. The authors reported personal and institutional relationships in the form of advisory board memberships, investigator appointments, speakers bureau positions, research support, and consultancies for a variety of pharmaceutical companies, agencies, institutions, publishers, and other organizations.
Migraine with atypical features may require neuroimaging, according to the guideline. These include an unusual aura; change in clinical features; a first or worst migraine; a migraine that presents with brainstem aura, confusion, or motor manifestation; migraine accompaniments in later life; headaches that are side-locked or posttraumatic; and aura that presents without headache.
Assessing the evidence
The recommendation to avoid MRI or CT in otherwise neurologically normal patients with migraine carried a grade A recommendation from the American Headache Society, while the specific considerations for neuroimaging was based on consensus and carried a grade C recommendation, according to lead author Randolph W. Evans, MD, of the department of neurology at Baylor College of Medicine in Houston, and colleagues.
The recommendations, published in the journal Headache (2020 Feb;60(2):318-36), came from a systematic review of 23 studies of adults at least 18 years old who underwent MRI or CT during outpatient treatment for migraine between 1973 and 2018. Ten studies looked at CT neuroimaging in patients with migraine, nine studies examined MRI neuroimaging alone in patients with migraine, and four studies contained adults with headache or migraine who underwent either MRI or CT. The majority of studies analyzed were retrospective or cross-sectional in nature, while four studies were prospective observational studies.
Dr. Evans and colleagues noted that neuroimaging for patients with suspected migraine is ordered for a variety of reasons, such as excluding conditions that aren’t migraine, diagnostic certainty, cognitive bias, practice workflow, medicolegal concerns, addressing patient and family anxiety, and addressing clinician anxiety. Neuroimaging also can be costly, they said, adding up to an estimated $1 billion annually according to one study, and can lead to additional testing from findings that may not be clinically significant.
Good advice, with caveats
In an interview, Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews, said that while he generally does not like broad guideline recommendations, the recommendation made by the American Headache Society to avoid neuroimaging in patients with a normal neurological examination without any atypical features and red flags “takes most of the important factors into consideration and will work almost all the time.” The recommendation made by consensus for specific considerations of neuroimaging was issued by top headache specialists in the United States who reviewed the data, and it is unlikely a patient with a migraine as diagnosed by the International Classification of Headache Disorders with a normal neurological examination would have a significant abnormality that would appear with imaging, Dr. Rapoport said.
“If everyone caring for migraine patients knew these recommendations, and used them unless the patients fit the exclusions mentioned, we would have more efficient clinical practice and save lots of money on unnecessary scanning,” he said.
However, Dr. Rapoport, clinical professor of neurology at the University of California, Los Angeles, founder of the New England Center for Headache, and past president of The International Headache Society, said that not all clinicians will be convinced by the American Headache Society’s recommendations.
“Various third parties often jump on society recommendations or guidelines and prevent smart clinicians from doing what they need to do when they want to disregard the recommendation or guideline,” he explained. “More importantly, if a physician feels the need to think out of the box and image a patient without a clear reason, and the patient cannot pay for the scan when a medical insurance company refuses to authorize it, there can be a bad result if the patient does not get the study.”
Dr. Rapoport noted that the guideline does not address situations where neuroimaging may not pick up conditions that lead to migraine, such as a subarachnoid or subdural hemorrhage, reversible cerebral vasoconstriction syndrome, or early aspects of low cerebrospinal fluid pressure syndrome. Anxiety on the part of the patient or the clinician is another area that can be addressed by future research, he said.
“If the clinician does a good job of explaining the odds of anything significant being found with a typical migraine history and normal examination, and the patient says [they] need an MRI with contrast to be sure, it will be difficult to dissuade them,” said Dr. Rapoport. “If you don’t order one, they will find a way to get one. If it is abnormal, you could be in trouble. Also, if the clinician has no good reason to do a scan but has anxiety about what is being missed, it will probably get done.”
There was no funding source for the guidelines. The authors reported personal and institutional relationships in the form of advisory board memberships, investigator appointments, speakers bureau positions, research support, and consultancies for a variety of pharmaceutical companies, agencies, institutions, publishers, and other organizations.
Migraine with atypical features may require neuroimaging, according to the guideline. These include an unusual aura; change in clinical features; a first or worst migraine; a migraine that presents with brainstem aura, confusion, or motor manifestation; migraine accompaniments in later life; headaches that are side-locked or posttraumatic; and aura that presents without headache.
Assessing the evidence
The recommendation to avoid MRI or CT in otherwise neurologically normal patients with migraine carried a grade A recommendation from the American Headache Society, while the specific considerations for neuroimaging was based on consensus and carried a grade C recommendation, according to lead author Randolph W. Evans, MD, of the department of neurology at Baylor College of Medicine in Houston, and colleagues.
The recommendations, published in the journal Headache (2020 Feb;60(2):318-36), came from a systematic review of 23 studies of adults at least 18 years old who underwent MRI or CT during outpatient treatment for migraine between 1973 and 2018. Ten studies looked at CT neuroimaging in patients with migraine, nine studies examined MRI neuroimaging alone in patients with migraine, and four studies contained adults with headache or migraine who underwent either MRI or CT. The majority of studies analyzed were retrospective or cross-sectional in nature, while four studies were prospective observational studies.
Dr. Evans and colleagues noted that neuroimaging for patients with suspected migraine is ordered for a variety of reasons, such as excluding conditions that aren’t migraine, diagnostic certainty, cognitive bias, practice workflow, medicolegal concerns, addressing patient and family anxiety, and addressing clinician anxiety. Neuroimaging also can be costly, they said, adding up to an estimated $1 billion annually according to one study, and can lead to additional testing from findings that may not be clinically significant.
Good advice, with caveats
In an interview, Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews, said that while he generally does not like broad guideline recommendations, the recommendation made by the American Headache Society to avoid neuroimaging in patients with a normal neurological examination without any atypical features and red flags “takes most of the important factors into consideration and will work almost all the time.” The recommendation made by consensus for specific considerations of neuroimaging was issued by top headache specialists in the United States who reviewed the data, and it is unlikely a patient with a migraine as diagnosed by the International Classification of Headache Disorders with a normal neurological examination would have a significant abnormality that would appear with imaging, Dr. Rapoport said.
“If everyone caring for migraine patients knew these recommendations, and used them unless the patients fit the exclusions mentioned, we would have more efficient clinical practice and save lots of money on unnecessary scanning,” he said.
However, Dr. Rapoport, clinical professor of neurology at the University of California, Los Angeles, founder of the New England Center for Headache, and past president of The International Headache Society, said that not all clinicians will be convinced by the American Headache Society’s recommendations.
“Various third parties often jump on society recommendations or guidelines and prevent smart clinicians from doing what they need to do when they want to disregard the recommendation or guideline,” he explained. “More importantly, if a physician feels the need to think out of the box and image a patient without a clear reason, and the patient cannot pay for the scan when a medical insurance company refuses to authorize it, there can be a bad result if the patient does not get the study.”
Dr. Rapoport noted that the guideline does not address situations where neuroimaging may not pick up conditions that lead to migraine, such as a subarachnoid or subdural hemorrhage, reversible cerebral vasoconstriction syndrome, or early aspects of low cerebrospinal fluid pressure syndrome. Anxiety on the part of the patient or the clinician is another area that can be addressed by future research, he said.
“If the clinician does a good job of explaining the odds of anything significant being found with a typical migraine history and normal examination, and the patient says [they] need an MRI with contrast to be sure, it will be difficult to dissuade them,” said Dr. Rapoport. “If you don’t order one, they will find a way to get one. If it is abnormal, you could be in trouble. Also, if the clinician has no good reason to do a scan but has anxiety about what is being missed, it will probably get done.”
There was no funding source for the guidelines. The authors reported personal and institutional relationships in the form of advisory board memberships, investigator appointments, speakers bureau positions, research support, and consultancies for a variety of pharmaceutical companies, agencies, institutions, publishers, and other organizations.
FROM HEADACHE
TBI deaths from falls on the rise
A 17% surge in mortality from fall-related traumatic brain injuries from 2008 to 2017 was driven largely by increases among those aged 75 years and older, according to investigators from the Centers for Disease Control and Prevention.
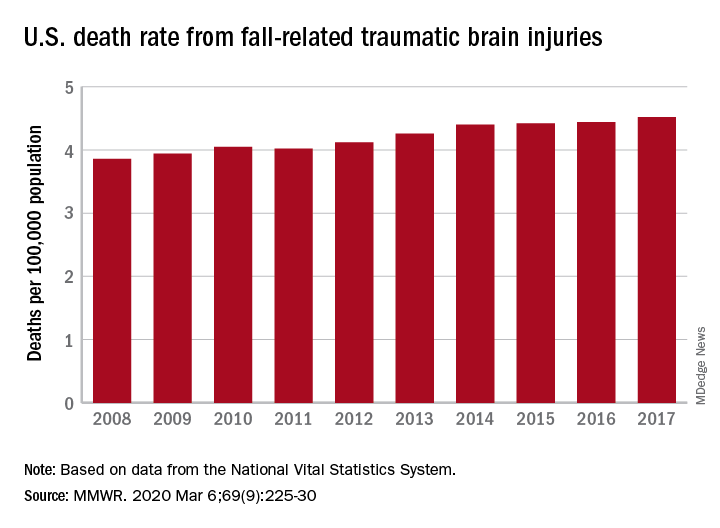
Nationally, the rate of deaths from traumatic brain injuries (TBIs) caused by unintentional falls rose from 3.86 per 100,000 population in 2008 to 4.52 per 100,000 in 2017, as the number of deaths went from 12,311 to 17,408, said Alexis B. Peterson, PhD, and Scott R. Kegler, PhD, of the CDC’s National Center for Injury Prevention and Control in Atlanta.
“This increase might be explained by longer survival following the onset of common diseases such as stroke, cancer, and heart disease or be attributable to the increasing population of older adults in the United States,” they suggested in the Mortality and Morbidity Weekly Report.
The rate of fall-related TBI among Americans aged 75 years and older increased by an average of 2.6% per year from 2008 to 2017, compared with 1.8% in those aged 55-74. Over that same time, death rates dropped for those aged 35-44 (–0.3%), 18-34 (–1.1%), and 0-17 (–4.3%), they said, based on data from the National Vital Statistics System’s multiple cause-of-death database.
The death rate increased fastest in residents of rural areas (2.9% per year), but deaths from fall-related TBI were up at all levels of urbanization. The largest central cities and fringe metro areas were up by 1.4% a year, with larger annual increases seen in medium-size cities (2.1%), small cities (2.2%), and small towns (2.1%), Dr. Peterson and Dr. Kegler said.
Rates of TBI-related mortality in general are higher in rural areas, they noted, and “heterogeneity in the availability and accessibility of resources (e.g., access to high-level trauma centers and rehabilitative services) can result in disparities in postinjury outcomes.”
State-specific rates increased in 45 states, although Alaska was excluded from the analysis because of its small number of cases (less than 20). Increases were significant in 29 states, but none of the changes were significant in the 4 states with lower rates at the end of the study period, the investigators reported.
“In older adults, evidence-based fall prevention strategies can prevent falls and avert costly medical expenditures,” Dr. Peterson and Dr. Kegler said, suggesting that health care providers “consider prescribing exercises that incorporate balance, strength and gait activities, such as tai chi, and reviewing and managing medications linked to falls.”
SOURCE: Peterson AB, Kegler SR. MMWR. 2019 Mar 6;69(9):225-30.
A 17% surge in mortality from fall-related traumatic brain injuries from 2008 to 2017 was driven largely by increases among those aged 75 years and older, according to investigators from the Centers for Disease Control and Prevention.

Nationally, the rate of deaths from traumatic brain injuries (TBIs) caused by unintentional falls rose from 3.86 per 100,000 population in 2008 to 4.52 per 100,000 in 2017, as the number of deaths went from 12,311 to 17,408, said Alexis B. Peterson, PhD, and Scott R. Kegler, PhD, of the CDC’s National Center for Injury Prevention and Control in Atlanta.
“This increase might be explained by longer survival following the onset of common diseases such as stroke, cancer, and heart disease or be attributable to the increasing population of older adults in the United States,” they suggested in the Mortality and Morbidity Weekly Report.
The rate of fall-related TBI among Americans aged 75 years and older increased by an average of 2.6% per year from 2008 to 2017, compared with 1.8% in those aged 55-74. Over that same time, death rates dropped for those aged 35-44 (–0.3%), 18-34 (–1.1%), and 0-17 (–4.3%), they said, based on data from the National Vital Statistics System’s multiple cause-of-death database.
The death rate increased fastest in residents of rural areas (2.9% per year), but deaths from fall-related TBI were up at all levels of urbanization. The largest central cities and fringe metro areas were up by 1.4% a year, with larger annual increases seen in medium-size cities (2.1%), small cities (2.2%), and small towns (2.1%), Dr. Peterson and Dr. Kegler said.
Rates of TBI-related mortality in general are higher in rural areas, they noted, and “heterogeneity in the availability and accessibility of resources (e.g., access to high-level trauma centers and rehabilitative services) can result in disparities in postinjury outcomes.”
State-specific rates increased in 45 states, although Alaska was excluded from the analysis because of its small number of cases (less than 20). Increases were significant in 29 states, but none of the changes were significant in the 4 states with lower rates at the end of the study period, the investigators reported.
“In older adults, evidence-based fall prevention strategies can prevent falls and avert costly medical expenditures,” Dr. Peterson and Dr. Kegler said, suggesting that health care providers “consider prescribing exercises that incorporate balance, strength and gait activities, such as tai chi, and reviewing and managing medications linked to falls.”
SOURCE: Peterson AB, Kegler SR. MMWR. 2019 Mar 6;69(9):225-30.
A 17% surge in mortality from fall-related traumatic brain injuries from 2008 to 2017 was driven largely by increases among those aged 75 years and older, according to investigators from the Centers for Disease Control and Prevention.

Nationally, the rate of deaths from traumatic brain injuries (TBIs) caused by unintentional falls rose from 3.86 per 100,000 population in 2008 to 4.52 per 100,000 in 2017, as the number of deaths went from 12,311 to 17,408, said Alexis B. Peterson, PhD, and Scott R. Kegler, PhD, of the CDC’s National Center for Injury Prevention and Control in Atlanta.
“This increase might be explained by longer survival following the onset of common diseases such as stroke, cancer, and heart disease or be attributable to the increasing population of older adults in the United States,” they suggested in the Mortality and Morbidity Weekly Report.
The rate of fall-related TBI among Americans aged 75 years and older increased by an average of 2.6% per year from 2008 to 2017, compared with 1.8% in those aged 55-74. Over that same time, death rates dropped for those aged 35-44 (–0.3%), 18-34 (–1.1%), and 0-17 (–4.3%), they said, based on data from the National Vital Statistics System’s multiple cause-of-death database.
The death rate increased fastest in residents of rural areas (2.9% per year), but deaths from fall-related TBI were up at all levels of urbanization. The largest central cities and fringe metro areas were up by 1.4% a year, with larger annual increases seen in medium-size cities (2.1%), small cities (2.2%), and small towns (2.1%), Dr. Peterson and Dr. Kegler said.
Rates of TBI-related mortality in general are higher in rural areas, they noted, and “heterogeneity in the availability and accessibility of resources (e.g., access to high-level trauma centers and rehabilitative services) can result in disparities in postinjury outcomes.”
State-specific rates increased in 45 states, although Alaska was excluded from the analysis because of its small number of cases (less than 20). Increases were significant in 29 states, but none of the changes were significant in the 4 states with lower rates at the end of the study period, the investigators reported.
“In older adults, evidence-based fall prevention strategies can prevent falls and avert costly medical expenditures,” Dr. Peterson and Dr. Kegler said, suggesting that health care providers “consider prescribing exercises that incorporate balance, strength and gait activities, such as tai chi, and reviewing and managing medications linked to falls.”
SOURCE: Peterson AB, Kegler SR. MMWR. 2019 Mar 6;69(9):225-30.
FROM MMWR
Stress-related disorders linked to later neurodegenerative diseases
Individuals with posttraumatic stress disorder (PTSD), acute stress reaction, adjustment disorder, or other stress reactions had an 80% increased risk of vascular neurodegenerative diseases, according to results of the study, which was based on Swedish population registry data.
Risk of primary neurodegenerative diseases was increased as well in people with those conditions, but only by 31%, according to lead author Huan Song, MD, PhD, of Sichuan University in Chengdu, China.
“The stronger association observed for neurodegenerative diseases with a vascular component, compared with primary neurodegenerative diseases, suggested a considerable role of a possible cerebrovascular pathway,” Dr. Song and coauthors said in a report on the study appearing in JAMA Neurology.
While some previous studies have linked stress-related disorders to neurodegenerative diseases – particularly PTSD and dementia – this is believed to be the first, according to the investigators, to comprehensively evaluate all stress-related disorders in relation to the most common neurodegenerative conditions.
When considering neurodegenerative conditions separately, they found a statistically significant association between stress-related disorders and Alzheimer’s disease, while linkages with Parkinson’s disease and amyotrophic lateral sclerosis (ALS) were “comparable” but associations did not reach statistical significance, according to investigators.
Based on these findings, stress reduction should be recommended in addition to daily physical activity, mental activity, and a heart-healthy diet to potentially reduce risk of onset or worsening of cognitive decline, according to Chun Lim, MD, PhD, medical director of the cognitive neurology unit at Beth Israel Deaconess Medical Center in Boston.
“We don’t really have great evidence that anything slows down the progression of Alzheimer’s disease, but there are some suggestions that for people who lead heart-healthy lifestyles or adhere to a Mediterranean diet, fewer develop cognitive issues over 5-10 years,” Dr. Lim said in an interview. “Because of this paper, stress reduction may be one additional way to hopefully help these patients these patients that have or are concerned about cognitive issues.”
The population-matched cohort of the study included 61,748 individuals with stress-related disorders and 595,335 matched individuals without those disorders, while the sibling-matched cohort included 44,839 individuals with those disorders and 78,482 without. The median age at the start of follow-up was 47 years and 39.4% of those with stress-related disorders were male.
During follow-up, the incidence of neurodegenerative diseases per 1,000 person-years was 1.50 for individuals with stress-related disorders, versus 0.82 for those without stress-related disorders, according to the report. Risk of primary neurodegenerative diseases was increased among those with stress-related disorders, compared with those without, with a hazard ratio of 1.31 (95% confidence interval, 1.15-1.48). However, the risk of vascular neurodegenerative diseases was significantly higher, with an HR of 1.80 (95% CI, 1.40-2.31; P = .03 for the difference between hazard ratios).
Results of the matched sibling cohort supported results of the population-matched cohort, though the elevated risk of vascular neurodegenerative diseases among those with stress-related disorders was “slightly lower” than in the population-based cohort, Dr. Song and coauthors wrote in their report.
Beyond causing a host of hormonal and medical issues, stress can lead to sleep issues that may have long-term consequences, Dr. Lim noted in the interview.
“There’s some thought that quality sleep is important for memory formation, and if people are under a fair amount of stress and they have really poor sleep, that can also lead to cognitive issues including memory impairment,” he said.
“There are these multiple avenues that may be contributing to the accelerated development of these kinds of issues,” he added, “so I think this paper suggests more ways to counsel the patients about using lifestyle modifications to slow down the development of these cognitive impairments.”
Funding for the study came from the Swedish Research Council, Icelandic Research Fund; ,European Research Council the Karolinska Institutet, Swedish Research Council, and West China Hospital. Authors of the study provided disclosures related to those organizations as well as Shire/Takeda and Evolan.
SOURCE: Song H et al. JAMA Neurol. 2020 Mar 9. doi: 10.1001/jamaneurol.2020.0117.
Individuals with posttraumatic stress disorder (PTSD), acute stress reaction, adjustment disorder, or other stress reactions had an 80% increased risk of vascular neurodegenerative diseases, according to results of the study, which was based on Swedish population registry data.
Risk of primary neurodegenerative diseases was increased as well in people with those conditions, but only by 31%, according to lead author Huan Song, MD, PhD, of Sichuan University in Chengdu, China.
“The stronger association observed for neurodegenerative diseases with a vascular component, compared with primary neurodegenerative diseases, suggested a considerable role of a possible cerebrovascular pathway,” Dr. Song and coauthors said in a report on the study appearing in JAMA Neurology.
While some previous studies have linked stress-related disorders to neurodegenerative diseases – particularly PTSD and dementia – this is believed to be the first, according to the investigators, to comprehensively evaluate all stress-related disorders in relation to the most common neurodegenerative conditions.
When considering neurodegenerative conditions separately, they found a statistically significant association between stress-related disorders and Alzheimer’s disease, while linkages with Parkinson’s disease and amyotrophic lateral sclerosis (ALS) were “comparable” but associations did not reach statistical significance, according to investigators.
Based on these findings, stress reduction should be recommended in addition to daily physical activity, mental activity, and a heart-healthy diet to potentially reduce risk of onset or worsening of cognitive decline, according to Chun Lim, MD, PhD, medical director of the cognitive neurology unit at Beth Israel Deaconess Medical Center in Boston.
“We don’t really have great evidence that anything slows down the progression of Alzheimer’s disease, but there are some suggestions that for people who lead heart-healthy lifestyles or adhere to a Mediterranean diet, fewer develop cognitive issues over 5-10 years,” Dr. Lim said in an interview. “Because of this paper, stress reduction may be one additional way to hopefully help these patients these patients that have or are concerned about cognitive issues.”
The population-matched cohort of the study included 61,748 individuals with stress-related disorders and 595,335 matched individuals without those disorders, while the sibling-matched cohort included 44,839 individuals with those disorders and 78,482 without. The median age at the start of follow-up was 47 years and 39.4% of those with stress-related disorders were male.
During follow-up, the incidence of neurodegenerative diseases per 1,000 person-years was 1.50 for individuals with stress-related disorders, versus 0.82 for those without stress-related disorders, according to the report. Risk of primary neurodegenerative diseases was increased among those with stress-related disorders, compared with those without, with a hazard ratio of 1.31 (95% confidence interval, 1.15-1.48). However, the risk of vascular neurodegenerative diseases was significantly higher, with an HR of 1.80 (95% CI, 1.40-2.31; P = .03 for the difference between hazard ratios).
Results of the matched sibling cohort supported results of the population-matched cohort, though the elevated risk of vascular neurodegenerative diseases among those with stress-related disorders was “slightly lower” than in the population-based cohort, Dr. Song and coauthors wrote in their report.
Beyond causing a host of hormonal and medical issues, stress can lead to sleep issues that may have long-term consequences, Dr. Lim noted in the interview.
“There’s some thought that quality sleep is important for memory formation, and if people are under a fair amount of stress and they have really poor sleep, that can also lead to cognitive issues including memory impairment,” he said.
“There are these multiple avenues that may be contributing to the accelerated development of these kinds of issues,” he added, “so I think this paper suggests more ways to counsel the patients about using lifestyle modifications to slow down the development of these cognitive impairments.”
Funding for the study came from the Swedish Research Council, Icelandic Research Fund; ,European Research Council the Karolinska Institutet, Swedish Research Council, and West China Hospital. Authors of the study provided disclosures related to those organizations as well as Shire/Takeda and Evolan.
SOURCE: Song H et al. JAMA Neurol. 2020 Mar 9. doi: 10.1001/jamaneurol.2020.0117.
Individuals with posttraumatic stress disorder (PTSD), acute stress reaction, adjustment disorder, or other stress reactions had an 80% increased risk of vascular neurodegenerative diseases, according to results of the study, which was based on Swedish population registry data.
Risk of primary neurodegenerative diseases was increased as well in people with those conditions, but only by 31%, according to lead author Huan Song, MD, PhD, of Sichuan University in Chengdu, China.
“The stronger association observed for neurodegenerative diseases with a vascular component, compared with primary neurodegenerative diseases, suggested a considerable role of a possible cerebrovascular pathway,” Dr. Song and coauthors said in a report on the study appearing in JAMA Neurology.
While some previous studies have linked stress-related disorders to neurodegenerative diseases – particularly PTSD and dementia – this is believed to be the first, according to the investigators, to comprehensively evaluate all stress-related disorders in relation to the most common neurodegenerative conditions.
When considering neurodegenerative conditions separately, they found a statistically significant association between stress-related disorders and Alzheimer’s disease, while linkages with Parkinson’s disease and amyotrophic lateral sclerosis (ALS) were “comparable” but associations did not reach statistical significance, according to investigators.
Based on these findings, stress reduction should be recommended in addition to daily physical activity, mental activity, and a heart-healthy diet to potentially reduce risk of onset or worsening of cognitive decline, according to Chun Lim, MD, PhD, medical director of the cognitive neurology unit at Beth Israel Deaconess Medical Center in Boston.
“We don’t really have great evidence that anything slows down the progression of Alzheimer’s disease, but there are some suggestions that for people who lead heart-healthy lifestyles or adhere to a Mediterranean diet, fewer develop cognitive issues over 5-10 years,” Dr. Lim said in an interview. “Because of this paper, stress reduction may be one additional way to hopefully help these patients these patients that have or are concerned about cognitive issues.”
The population-matched cohort of the study included 61,748 individuals with stress-related disorders and 595,335 matched individuals without those disorders, while the sibling-matched cohort included 44,839 individuals with those disorders and 78,482 without. The median age at the start of follow-up was 47 years and 39.4% of those with stress-related disorders were male.
During follow-up, the incidence of neurodegenerative diseases per 1,000 person-years was 1.50 for individuals with stress-related disorders, versus 0.82 for those without stress-related disorders, according to the report. Risk of primary neurodegenerative diseases was increased among those with stress-related disorders, compared with those without, with a hazard ratio of 1.31 (95% confidence interval, 1.15-1.48). However, the risk of vascular neurodegenerative diseases was significantly higher, with an HR of 1.80 (95% CI, 1.40-2.31; P = .03 for the difference between hazard ratios).
Results of the matched sibling cohort supported results of the population-matched cohort, though the elevated risk of vascular neurodegenerative diseases among those with stress-related disorders was “slightly lower” than in the population-based cohort, Dr. Song and coauthors wrote in their report.
Beyond causing a host of hormonal and medical issues, stress can lead to sleep issues that may have long-term consequences, Dr. Lim noted in the interview.
“There’s some thought that quality sleep is important for memory formation, and if people are under a fair amount of stress and they have really poor sleep, that can also lead to cognitive issues including memory impairment,” he said.
“There are these multiple avenues that may be contributing to the accelerated development of these kinds of issues,” he added, “so I think this paper suggests more ways to counsel the patients about using lifestyle modifications to slow down the development of these cognitive impairments.”
Funding for the study came from the Swedish Research Council, Icelandic Research Fund; ,European Research Council the Karolinska Institutet, Swedish Research Council, and West China Hospital. Authors of the study provided disclosures related to those organizations as well as Shire/Takeda and Evolan.
SOURCE: Song H et al. JAMA Neurol. 2020 Mar 9. doi: 10.1001/jamaneurol.2020.0117.
FROM JAMA NEUROLOGY
Shared medical appointments educate and encourage MS patients
WEST PALM BEACH, FLA. – according to a presentation at the Americas Committee for Treatment and Research in Multiple Sclerosis.
“At first, this may sound to patients like an awkward concept – they may say, ‘Why would I want to have a medical appointment with other people?’ ” Mary R. Rensel, MD, who is the director of the program at the Mellen Center for Multiple Sclerosis, Cleveland Clinic Foundation, said. “But once they get there, it’s wonderful to see what happens – patients start to encourage each other and share resources, and it’s enjoyable for the patients and providers alike,” she said.
The main objective of the shared appointments concept was to increase education regarding comorbidity prevention and management of MS, however, importantly, if patients wish to discuss any issues privately, they are accommodated. In addition, family members, children, and caregivers are all welcome to attend. “Caregivers need support as well, so their participation is welcome,” Dr. Rensel said.
A significant benefit of the program is the extended time with providers – an hour and a half – which is a substantially longer period than patients and providers typically spend together, Dr. Rensel noted. “Medical visits are often so rushed, but this gives us much more time together, to learn more and talk about things like brain health,” she said.
With guidance from a multidisciplinary team including nurses, wellness providers, psychologists, and other experts, there are currently seven meeting themes that are rotated through the year, focusing on a variety of subjects. One, for instance, includes education from a nutritionist, and the center includes a kitchen for the group to learn about and try recipes. Other sessions include chair yoga, art therapy, guided imagery, and exercise physiology.
The Cleveland Clinic is a leader in the concept of SMA and offers it to as many as 360 disease states. With the pilot program now underway for more than 3 years, Dr. Rensel and her team conducted a study to investigate its effects.
For the study, the authors collected clinical data on 50 patients who had attended at least one session between January 2016 and June 2019. Among the patients, 94% were female, 80% had relapsing-remitting MS, and mean age was 50. Patients had a mean Determined Disease Steps (PDSS) score of 3.1 plus or minus 2.4 and the average 25-foot walk and nine-hole peg test (dominant hand) times were 9.4 plus or minus 7.8 seconds and 25.8 plus or minus 9.1 seconds, respectively.
The most common comorbidity was depression/anxiety, occurring in 44% of patients, however after participation in the shared medical appointment program, their mean Patient Health Questionnaire-9 scores, with higher scores indicative of worse depression, decreased from pretreatment scores of 7.3 plus or minus 5.5 to posttreatment scores of 5.1 plus or minus 5.6 (P = .001).
Notably, the program appears to have had a positive effect on patients’ use of health care services – while there was a significant decrease in the mean number of emergency room visits (n = 13 to n = 2; P = .0005), the results showed a favorable increase in mean number of follow-up visits with attendees’ primary care providers (n = 19 to n = 41; P = 3.47), physical therapists (n = 15 to n = 27; P = .004), or psychologists (n = 6 to n = 19; P = .003).
“The study was to evaluate the effect of the program after even just one appointment, and we found it really seemed to increase the use of more appropriate care, with less ER utilization and more visits to primary care,” Dr. Rensel said. The study even showed a small but significant reduction in pre- and postoutcome body mass index (BMI, 30.2 plus or minus 7.3 vs. 28.8 plus or minus 7.1; P = .03).
A critical metric that was not measured in the study – the effect of social interaction and camaraderie in a condition that can, for many, feel socially isolating – is clearly profound, Dr. Rensel said.
Amar Dhand, MD, associate professor of neurology at Brigham and Women’s Hospital, Harvard University, Boston, agreed that the peer support in such medical group settings can be highly valuable.
“Shared medical appointments offer an opportunity for peer-to-peer engagement, support, and education,” he said in an interview. “For many patients, this is a chance to bond with persons who are coexperiencing similar problems, allowing new social connections to emerge.”
Dr. Dhand, who spoke on the issue of the importance of social networks at the meeting, noted that, although there are numerous benefits with shared medical appointments, not all patients may respond well.
“Health care settings are one place to stimulate community among peers. This is one important ingredient of addressing social isolation,” he said. “However, there remain challenges such as sustainability of such relationships, paradoxical depression when persons see others with more severe disease, and infrastructure to support such programs.”
The findings from the study, however, do suggest favorable responses, he noted.
“I think, mechanistically, improved psychosocial outcomes are the most pertinent to the intervention,” Dr. Dhand said. “The health care utilization may be attributed to other factors and will need to be assessed in a case control design.”
Key benefits of the shared medical appointment concept
A recent article from Cleveland Clinic researchers reviewing the concept of shared medical appointments summarizes that the programs offer benefits based on nine key principles:
- Group exposure in shared medical appointments combats isolation, which in turn helps to remove doubts about one’s ability to manage illness.
- Patients learn about disease self-management vicariously by witnessing others’ illness experiences.
- Patients feel inspired by seeing others who are coping well.
- Group dynamics lead patients and providers to developing more equitable relationships.
- Providers feel increased appreciation and rapport toward colleagues leading to increased efficiency.
- Providers learn from the patients how better to meet their patients’ needs.
- Adequate time allotment of the SMA leads patients to feel supported.
- Patients receive professional expertise from the provider in combination with firsthand information from peers, resulting in more robust health knowledge.
- Patients have the opportunity to see how the physicians interact with fellow patients, which allows them to get to know the physician and better determine their level of trust.
The take-home message from the shared medical appointments concept is that “it may hit a quadruple aim,” Dr. Rensel said. “Access, cost, outcomes, and provider satisfaction.”
The Shared Medical Appointments program received a grant from Genzyme. Dr. Rensel reported consulting or advisory board relationships with Serono, Biogen, Teva, Genzyme, Novartis, and the National Multiple Sclerosis Society. Dr. Dhand had no disclosures to report.
WEST PALM BEACH, FLA. – according to a presentation at the Americas Committee for Treatment and Research in Multiple Sclerosis.
“At first, this may sound to patients like an awkward concept – they may say, ‘Why would I want to have a medical appointment with other people?’ ” Mary R. Rensel, MD, who is the director of the program at the Mellen Center for Multiple Sclerosis, Cleveland Clinic Foundation, said. “But once they get there, it’s wonderful to see what happens – patients start to encourage each other and share resources, and it’s enjoyable for the patients and providers alike,” she said.
The main objective of the shared appointments concept was to increase education regarding comorbidity prevention and management of MS, however, importantly, if patients wish to discuss any issues privately, they are accommodated. In addition, family members, children, and caregivers are all welcome to attend. “Caregivers need support as well, so their participation is welcome,” Dr. Rensel said.
A significant benefit of the program is the extended time with providers – an hour and a half – which is a substantially longer period than patients and providers typically spend together, Dr. Rensel noted. “Medical visits are often so rushed, but this gives us much more time together, to learn more and talk about things like brain health,” she said.
With guidance from a multidisciplinary team including nurses, wellness providers, psychologists, and other experts, there are currently seven meeting themes that are rotated through the year, focusing on a variety of subjects. One, for instance, includes education from a nutritionist, and the center includes a kitchen for the group to learn about and try recipes. Other sessions include chair yoga, art therapy, guided imagery, and exercise physiology.
The Cleveland Clinic is a leader in the concept of SMA and offers it to as many as 360 disease states. With the pilot program now underway for more than 3 years, Dr. Rensel and her team conducted a study to investigate its effects.
For the study, the authors collected clinical data on 50 patients who had attended at least one session between January 2016 and June 2019. Among the patients, 94% were female, 80% had relapsing-remitting MS, and mean age was 50. Patients had a mean Determined Disease Steps (PDSS) score of 3.1 plus or minus 2.4 and the average 25-foot walk and nine-hole peg test (dominant hand) times were 9.4 plus or minus 7.8 seconds and 25.8 plus or minus 9.1 seconds, respectively.
The most common comorbidity was depression/anxiety, occurring in 44% of patients, however after participation in the shared medical appointment program, their mean Patient Health Questionnaire-9 scores, with higher scores indicative of worse depression, decreased from pretreatment scores of 7.3 plus or minus 5.5 to posttreatment scores of 5.1 plus or minus 5.6 (P = .001).
Notably, the program appears to have had a positive effect on patients’ use of health care services – while there was a significant decrease in the mean number of emergency room visits (n = 13 to n = 2; P = .0005), the results showed a favorable increase in mean number of follow-up visits with attendees’ primary care providers (n = 19 to n = 41; P = 3.47), physical therapists (n = 15 to n = 27; P = .004), or psychologists (n = 6 to n = 19; P = .003).
“The study was to evaluate the effect of the program after even just one appointment, and we found it really seemed to increase the use of more appropriate care, with less ER utilization and more visits to primary care,” Dr. Rensel said. The study even showed a small but significant reduction in pre- and postoutcome body mass index (BMI, 30.2 plus or minus 7.3 vs. 28.8 plus or minus 7.1; P = .03).
A critical metric that was not measured in the study – the effect of social interaction and camaraderie in a condition that can, for many, feel socially isolating – is clearly profound, Dr. Rensel said.
Amar Dhand, MD, associate professor of neurology at Brigham and Women’s Hospital, Harvard University, Boston, agreed that the peer support in such medical group settings can be highly valuable.
“Shared medical appointments offer an opportunity for peer-to-peer engagement, support, and education,” he said in an interview. “For many patients, this is a chance to bond with persons who are coexperiencing similar problems, allowing new social connections to emerge.”
Dr. Dhand, who spoke on the issue of the importance of social networks at the meeting, noted that, although there are numerous benefits with shared medical appointments, not all patients may respond well.
“Health care settings are one place to stimulate community among peers. This is one important ingredient of addressing social isolation,” he said. “However, there remain challenges such as sustainability of such relationships, paradoxical depression when persons see others with more severe disease, and infrastructure to support such programs.”
The findings from the study, however, do suggest favorable responses, he noted.
“I think, mechanistically, improved psychosocial outcomes are the most pertinent to the intervention,” Dr. Dhand said. “The health care utilization may be attributed to other factors and will need to be assessed in a case control design.”
Key benefits of the shared medical appointment concept
A recent article from Cleveland Clinic researchers reviewing the concept of shared medical appointments summarizes that the programs offer benefits based on nine key principles:
- Group exposure in shared medical appointments combats isolation, which in turn helps to remove doubts about one’s ability to manage illness.
- Patients learn about disease self-management vicariously by witnessing others’ illness experiences.
- Patients feel inspired by seeing others who are coping well.
- Group dynamics lead patients and providers to developing more equitable relationships.
- Providers feel increased appreciation and rapport toward colleagues leading to increased efficiency.
- Providers learn from the patients how better to meet their patients’ needs.
- Adequate time allotment of the SMA leads patients to feel supported.
- Patients receive professional expertise from the provider in combination with firsthand information from peers, resulting in more robust health knowledge.
- Patients have the opportunity to see how the physicians interact with fellow patients, which allows them to get to know the physician and better determine their level of trust.
The take-home message from the shared medical appointments concept is that “it may hit a quadruple aim,” Dr. Rensel said. “Access, cost, outcomes, and provider satisfaction.”
The Shared Medical Appointments program received a grant from Genzyme. Dr. Rensel reported consulting or advisory board relationships with Serono, Biogen, Teva, Genzyme, Novartis, and the National Multiple Sclerosis Society. Dr. Dhand had no disclosures to report.
WEST PALM BEACH, FLA. – according to a presentation at the Americas Committee for Treatment and Research in Multiple Sclerosis.
“At first, this may sound to patients like an awkward concept – they may say, ‘Why would I want to have a medical appointment with other people?’ ” Mary R. Rensel, MD, who is the director of the program at the Mellen Center for Multiple Sclerosis, Cleveland Clinic Foundation, said. “But once they get there, it’s wonderful to see what happens – patients start to encourage each other and share resources, and it’s enjoyable for the patients and providers alike,” she said.
The main objective of the shared appointments concept was to increase education regarding comorbidity prevention and management of MS, however, importantly, if patients wish to discuss any issues privately, they are accommodated. In addition, family members, children, and caregivers are all welcome to attend. “Caregivers need support as well, so their participation is welcome,” Dr. Rensel said.
A significant benefit of the program is the extended time with providers – an hour and a half – which is a substantially longer period than patients and providers typically spend together, Dr. Rensel noted. “Medical visits are often so rushed, but this gives us much more time together, to learn more and talk about things like brain health,” she said.
With guidance from a multidisciplinary team including nurses, wellness providers, psychologists, and other experts, there are currently seven meeting themes that are rotated through the year, focusing on a variety of subjects. One, for instance, includes education from a nutritionist, and the center includes a kitchen for the group to learn about and try recipes. Other sessions include chair yoga, art therapy, guided imagery, and exercise physiology.
The Cleveland Clinic is a leader in the concept of SMA and offers it to as many as 360 disease states. With the pilot program now underway for more than 3 years, Dr. Rensel and her team conducted a study to investigate its effects.
For the study, the authors collected clinical data on 50 patients who had attended at least one session between January 2016 and June 2019. Among the patients, 94% were female, 80% had relapsing-remitting MS, and mean age was 50. Patients had a mean Determined Disease Steps (PDSS) score of 3.1 plus or minus 2.4 and the average 25-foot walk and nine-hole peg test (dominant hand) times were 9.4 plus or minus 7.8 seconds and 25.8 plus or minus 9.1 seconds, respectively.
The most common comorbidity was depression/anxiety, occurring in 44% of patients, however after participation in the shared medical appointment program, their mean Patient Health Questionnaire-9 scores, with higher scores indicative of worse depression, decreased from pretreatment scores of 7.3 plus or minus 5.5 to posttreatment scores of 5.1 plus or minus 5.6 (P = .001).
Notably, the program appears to have had a positive effect on patients’ use of health care services – while there was a significant decrease in the mean number of emergency room visits (n = 13 to n = 2; P = .0005), the results showed a favorable increase in mean number of follow-up visits with attendees’ primary care providers (n = 19 to n = 41; P = 3.47), physical therapists (n = 15 to n = 27; P = .004), or psychologists (n = 6 to n = 19; P = .003).
“The study was to evaluate the effect of the program after even just one appointment, and we found it really seemed to increase the use of more appropriate care, with less ER utilization and more visits to primary care,” Dr. Rensel said. The study even showed a small but significant reduction in pre- and postoutcome body mass index (BMI, 30.2 plus or minus 7.3 vs. 28.8 plus or minus 7.1; P = .03).
A critical metric that was not measured in the study – the effect of social interaction and camaraderie in a condition that can, for many, feel socially isolating – is clearly profound, Dr. Rensel said.
Amar Dhand, MD, associate professor of neurology at Brigham and Women’s Hospital, Harvard University, Boston, agreed that the peer support in such medical group settings can be highly valuable.
“Shared medical appointments offer an opportunity for peer-to-peer engagement, support, and education,” he said in an interview. “For many patients, this is a chance to bond with persons who are coexperiencing similar problems, allowing new social connections to emerge.”
Dr. Dhand, who spoke on the issue of the importance of social networks at the meeting, noted that, although there are numerous benefits with shared medical appointments, not all patients may respond well.
“Health care settings are one place to stimulate community among peers. This is one important ingredient of addressing social isolation,” he said. “However, there remain challenges such as sustainability of such relationships, paradoxical depression when persons see others with more severe disease, and infrastructure to support such programs.”
The findings from the study, however, do suggest favorable responses, he noted.
“I think, mechanistically, improved psychosocial outcomes are the most pertinent to the intervention,” Dr. Dhand said. “The health care utilization may be attributed to other factors and will need to be assessed in a case control design.”
Key benefits of the shared medical appointment concept
A recent article from Cleveland Clinic researchers reviewing the concept of shared medical appointments summarizes that the programs offer benefits based on nine key principles:
- Group exposure in shared medical appointments combats isolation, which in turn helps to remove doubts about one’s ability to manage illness.
- Patients learn about disease self-management vicariously by witnessing others’ illness experiences.
- Patients feel inspired by seeing others who are coping well.
- Group dynamics lead patients and providers to developing more equitable relationships.
- Providers feel increased appreciation and rapport toward colleagues leading to increased efficiency.
- Providers learn from the patients how better to meet their patients’ needs.
- Adequate time allotment of the SMA leads patients to feel supported.
- Patients receive professional expertise from the provider in combination with firsthand information from peers, resulting in more robust health knowledge.
- Patients have the opportunity to see how the physicians interact with fellow patients, which allows them to get to know the physician and better determine their level of trust.
The take-home message from the shared medical appointments concept is that “it may hit a quadruple aim,” Dr. Rensel said. “Access, cost, outcomes, and provider satisfaction.”
The Shared Medical Appointments program received a grant from Genzyme. Dr. Rensel reported consulting or advisory board relationships with Serono, Biogen, Teva, Genzyme, Novartis, and the National Multiple Sclerosis Society. Dr. Dhand had no disclosures to report.
REPORTING FROM ACTRIMS FORUM 2020
Depression, or something else?
CASE Suicidal behavior, severe headaches
Ms. A, age 60, presents to the emergency department (ED) with depression, suicidal behavior, and 3 days of severe headaches. Neurology is consulted and an MRI is ordered, which shows a 3.0-cm mass lesion in the left temporal lobe with associated vasogenic edema that is suspicious for metastatic disease (Figure).
Ms. A is admitted to the hospital for further workup of her brain lesion. She is started on IV dexamethasone, 10 mg every 6 hours, a glucocorticosteroid, for brain edema, and levetiracetam, 500 mg twice a day, for seizure prophylaxis.
Upon admission, in addition to oncology and neurosurgery, psychiatry is also consulted to evaluate Ms. A for depression and suicidality.
EVALUATION Mood changes and poor judgment
Ms. A has a psychiatric history of depression and alcohol use disorder but says she has not consumed any alcohol in years. Her medical history includes hypertension, diabetes, and stage 4 non-small–cell lung cancer, for which she received surgery and adjuvant chemoradiotherapy 1 year ago.
On initial intake, Ms. A reports that in addition to the headaches, she has also been experiencing worsening depression and suicidal behavior. For the past 2 months, she has had a severely depressed mood, with notable anhedonia, poor appetite, insomnia, low energy, and decreased concentration. The changes in her mental health were triggered by her mother’s death. Three days prior to admission, the patient planned to overdose on antihypertensive pills, but her suicide attempt was interrupted when her family called. She denies any current suicidal ideation, intent, or plan.
According to her family, Ms. A has been increasingly irritable and her personality has changed in the past month. She also has been repeatedly sorting through her neighbors’ garbage.
Ms. A’s current psychiatric medications are duloxetine, 30 mg/d; quetiapine, 50 mg every night at bedtime; and buspirone, 10 mg/d. However, it is unclear if she is consistently taking these medications.
Continue to: On mental status examination...
On mental status examination, Ms. A is calm and she has no abnormal movements. She says she is depressed. Her affect is reactive and labile. She is alert and oriented to person, place, and time. Her attention, registration, and recall are intact. Her executive function is not tested. However, Ms. A’s insight and judgment seem poor.
To address Ms. A’s worsening depression, the psychiatry team increases her duloxetine from 30 to 60 mg/d, and she continues quetiapine, 50 mg every night at bedtime, for mood lability. Buspirone is not continued because she was not taking a therapeutic dosage in the community.
Within 4 days, Ms. A shows improvement in sleep, appetite, and mood. She has no further suicidal ideation.
[polldaddy:10511743]
The authors’ observations
Ms. A had a recurrence of what was presumed to be major depressive disorder (MDD) in the context of her mother’s death. However, she also exhibited irritability, mood lability, and impulsivity, all of which could be part of her depression, or a separate problem related to her brain tumor. Because Ms. A had never displayed bizarre behavior before the past few weeks, it is likely that her CNS lesion was directly affecting her personality and possibly underlying her planned suicide attempt.
Fifty to 80% of patients with CNS tumors, either primary or metastatic, present with psychiatric symptoms.1 Table 11-3 lists common psychiatric symptoms of brain tumors. Unfortunately, there is little reliable evidence that directly correlates tumor location with specific psychiatric symptoms. A 2010 meta-analysis found a statistically significant link between anorexia nervosa and hypothalamic tumors.1 However, for other brain regions, there is only an increased likelihood that any given tumor location will produce psychiatric symptoms.1,4 For instance, compared to patients with tumors in other locations, those with temporal lobe tumors are more likely to present with mood disorders, personality changes, and memory problems.1 In contrast, patients with frontal lobe tumors have an increased likelihood of psychosis, mood disorders, and personality changes.1 Patients with tumors in the pituitary region often present with anxiety.1
Continue to: When considering treatment options...
When considering treatment options for Ms. A, alcohol withdrawal was unlikely given the remote history of alcohol use, low alcohol blood level, and lack of evidence of unstable vital signs or tremor. Although she might have benefited from inpatient psychiatric treatment, this needed to wait until there was a definitive treatment plan for her brain tumor. Finally, although a paraneoplastic syndrome, such as limbic encephalitis, could be causing her psychiatric symptoms, this scenario is less likely with non-small–cell lung cancer.
Although uncommon, CNS tumors can present with psychiatric symptoms as the only manifestation. This is more likely when a patient exhibits new-onset or atypical symptoms, or fails to respond to standard psychiatric treatment.4 Case reports have described patients with brain tumors being misdiagnosed as having a primary psychiatric condition, which delays treatment of their CNS cancer.2 Additionally, frontal and limbic tumors are more likely to present with psychiatric manifestations; up to 90% of patients exhibit altered mental status or personality changes, as did Ms. A.1,4 Clearly, it is easier to identify patients with psychiatric symptoms resulting from a brain tumor when they also present with focal neurologic deficits or systemic symptoms, such as headache or nausea and vomiting. Ms. A presented with severe headaches, which is what led to her early imaging and prompt diagnosis.
Numerous proposed mechanisms might account for the psychiatric symptoms that occur during the course of a brain tumor, including direct injury to neuronal cells, secretion of hormones or other tumor-derived substances, and peri-ictal phenomena.3
TREATMENT Tumor is removed, but memory is impaired
Ms. A is scheduled for craniotomy and surgical resection of the frontal mass. Prior to surgery, Ms. A shows interest in improving her health, cooperates with staff, and seeks her daughter’s input on treatment. One week after admission, Ms. A has her mass resected, which is confirmed on biopsy to be a lung metastasis. Post-surgery, Ms. A receives codeine, 30 mg every 6 hours as needed, for pain; she continues dexamethasone, 4 mg IV every 6 hours, for brain edema and levetiracetam, 500 mg twice a day, for seizure prophylaxis.
On Day 2 after surgery, Ms. A attempts to elope. When she is approached by a psychiatrist on the treatment team, she does not recognize him. Although her long-term memory seems intact, she is unable to remember the details of recent events, including her medical and surgical treatments.
[polldaddy:10511745]
Continue to: The authors' observations
The authors’ observations
Ms. A’s memory impairment may be secondary to a surgically acquired neurocognitive deficit. In the United States, brain metastases represent a significant public health issue, affecting >100,000 patients per year.5 Metastatic lesions are the most common brain tumors. Lung cancer, breast cancer, and melanoma are the leading solid tumors to spread to the CNS.5 In cases of single brain metastasis, similar to Ms. A’s solitary left temporal lobe lesion, surgical resection plays a critical role in treatment. It provides histological confirmation of metastatic disease and can relieve mass effect if present. Studies have shown that combined surgical resection with radiation improves survival relative to patients who undergo radiation therapy alone.6,7
However, the benefits of surgical resection need to be balanced with preservation of neurologic function. Emerging evidence suggests that a majority of patients have surgically-acquired cognitive deficits due to damage of normal surrounding tissues, and these deficits are associated with reduced quality of life.8,9 Further, a study examining glioma surgical resections found that patients with left temporal lobe tumors exhibit more frequent and severe neurocognitive decline than patients with right temporal lobe tumors, especially in domains such as verbal memory.8 Ms. A’s memory impairment was persistent during her postoperative course, which suggests that it was not just an immediate post-surgical phenomenon, but a longer-lasting cognitive change directly related to the resection.
It is also possible that Ms. A had a prior neurocognitive disorder that manifested to a greater degree as a result of the CNS tumor. Ms. A might have had early-onset Alzheimer’s disease, although her intact memory before surgery makes this less likely. Alternatively, she could have had vascular dementia, especially given her long-standing hypertension and diabetes. This might have been missed in the initial evaluation because executive function was not tested. However, the relatively abrupt onset of memory problems after surgery suggests that she had no underlying neurocognitive disorder.
Ms. A’s presumed episode of MDD might also explain her memory changes. Major depressive disorder is increasingly common among geriatric patients, affecting approximately 5% of community-dwelling older adults.10 Its incidence increases with medical comorbidities, as suggested by depression rates of 5% to 10% in the primary care setting vs 37% in patients after critical-care hospitalizations.10 Late-life depression (LLD) occurs in adults age ≥60. Unlike depression in younger patients, LLD is more likely to be associated with cognitive impairment, specifically impairment of executive function and memory.11 The incidence of cognitive impairment in LLD is higher in patients with a history of depression, such as Ms. A.11,12 However, in general, patients who are depressed have memory complaints out of proportion to the clinical findings, and they show poor effort on cognitive testing. Ms. A exhibited neither of these, which makes it less likely that LLD was the exclusive cause of her memory loss.13 Table 214 outlines the management of cognitive deficits in a patient with a brain tumor.
EVALUATION Increasingly agitated and paranoid
After the tumor resection, Ms. A becomes increasingly irritable, uncooperative, and agitated. She repeatedly demands to be discharged. She insists she is fine and refuses medications and further laboratory workup. She becomes paranoid about the nursing staff and believes they are trying to kill her.
Continue to: On psychiatric re-evaluation...
On psychiatric re-evaluation, Ms. A demonstrates pressured speech, perseveration about going home, paranoid delusions, and anger at her family and physicians.
[polldaddy:10511747]
The authors’ observations
Ms. A’s refusal of medications and agitation may be explained by postoperative delirium, a surgical complication that is increasingly common among geriatric patients and is associated with poor clinical outcomes. Delirium is characterized by an acute onset and fluctuating course of symptoms that include inattention, motoric hypo- or hyperactivity, inappropriate behavior, emotional lability, cognitive dysfunction, and psychotic symptoms.15 Risk factors that contribute to postoperative delirium include older age, alcohol use, and poor baseline functional and cognitive status.16 The pathophysiology of delirium is not fully understood, but accumulating evidence suggests that different sets of interacting biologic factors (ie, neurotransmitters and inflammation) contribute to a disruption of large-scale neuronal networks in the brain, resulting in cognitive dysfunction.15 Patients who develop postoperative delirium are more likely to develop long-term cognitive dysfunction and have an increased risk of dementia.16
Another potential source of Ms. A’s agitation is steroid use. Ms. A received IV dexamethasone, 8 to 16 mg/d, around the time of her surgery. Steroids are commonly used to treat brain tumors, particularly when there is vasogenic edema. Steroid psychosis is a term loosely used to describe a wide range of psychiatric symptoms induced by corticosteroids that includes, but is not limited to, depression, mania, psychosis, delirium, and cognitive impairment.17 Steroid-induced psychiatric adverse effects occur in 5% to 18% of patients receiving corticosteroids and often happen early in treatment, although they can occur at any point.18 Corticosteroids influence brain activity via glucocorticoid and mineralocorticoid receptors. These receptors are widely distributed throughout the brain and affect neurotransmitter systems, such as the serotonergic system, that are associated with changes in mood, behavior, and cognition.17 While the adverse psychiatric manifestations of steroid use vary, higher dosages are associated with an increased risk of psychiatric complications; mania is more prevalent early in the course of treatment, and depression is more common with long-term use.17,19 Table 317,18 outlines the evidence-based treatment of corticosteroid-induced adverse psychiatric effects.
Although there are no clinical guidelines or FDA-approved medications for treating steroid-induced psychiatric adverse events, these are best managed by tapering and discontinuing steroids when possible and simultaneously using psychotropic medications to treat psychiatric symptoms. Case reports and limited evidence-based literature have demonstrated that steroid-induced mania responds to mood stabilizers or antipsychotics, while depression can be managed with antidepressants or lithium.17
Additionally, patients with CNS tumors are at risk for seizures and often are prescribed antiepileptics. Because it is easy to administer and does not need to be titrated, levetiracetam is a commonly used agent. However, levetiracetam can cause psychiatric adverse effects, including behavior changes and frank psychosis.20
Continue to: Finally, Ms. A's altered mental status...
Finally, Ms. A’s altered mental status could have been related to opioid intoxication. Opioids are used to manage postsurgical pain, and studies have shown these medications can be a precipitating factor for delirium in geriatric patients.21
TREATMENT Medication adjustments
At the request of the psychiatry team, levetiracetam is discontinued due to its potential for psychiatric adverse effects. The neurosurgery team replaces it with valproic acid, 500 mg every 12 hours. Ms. A is also tapered off steroids fairly rapidly because of the potential for steroid-induced psychiatric adverse effects. Her quetiapine is titrated from 50 to 150 mg every night at bedtime, and duloxetine is discontinued.
OUTCOME Agitation improves dramatically
Ms. A’s new medication regimen dramatically improves her agitation, which allows Ms. A, her family, and the medical team to work together to establish treatment goals. Ms. A ultimately returns home with the assistance of her family. She continues to have memory issues, but with improved emotion regulation. Several months later, Ms. A is readmitted to the hospital because her cancer has progressed despite treatment.
Bottom Line
Brain tumors may present with various psychiatric manifestations that can change during the course of the patient’s treatment. A comprehensive psychiatric evaluation should parse out the interplay between direct effects of the tumor and any adverse effects that are the result of medical and/or surgical interventions to determine the cause of psychiatric symptoms and their appropriate management.
Related Resource
Madhusoodanan S, Ting MB, Farah T, et al. Psychiatric aspects of brain tumors: a review. World J Psychiatry. 2015;5(3):273-285.
Drug Brand Names
Aripiprazole • Abilify
Buspirone • Buspar
Chlorpromazine • Thorazine
Codeine • Codeine systemic
Dexamethasone • Decadron
Duloxetine • Cymbalta
Haloperidol • Haldol
Levetiracetam • Keppra
Lorazepam • Ativan
Lithium • Eskalith, Lithobid
Olanzapine • Zyprexa
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproic acid • Depakene
1. Madhusoodanan S, Opler MG, Moise D, et al. Brain tumor location and psychiatric symptoms: is there any association? A meta-analysis of published case studies. Expert Rev Neurother. 2010;10(10):1529-1536.
2. Bunevicius A, Deltuva VP, Deltuviene D, et al. Brain lesions manifesting as psychiatric disorders: eight cases. CNS Spectr. 2008;13(11):950-958.
3. Pearl ML, Talgat G, Valea FA, et al. Psychiatric symptoms due to brain metastases. Med Update Psychiatr. 1998;3(4):91-94.
4. Madhusoodanan S, Danan D, Moise D. Psychiatric manifestations of brain tumors: diagnostic implications. Expert Rev Neurother. 2007;7(4):343-349.
5. Ferguson SD, Wagner KM, Prabhu SS, et al. Neurosurgical management of brain metastases. Clin Exp Metastasis. 2017;34(6-7):377-389.
6. Husain ZA, Regine WF, Kwok Y, et al. Brain metastases: contemporary management and future directions. Eur J Clin Med Oncol. 2011;3(3):38-45.
7. Vecht CJ, Haaxmareiche H, Noordijk EM, et al. Treatment of single brain metastasis - radiotherapy alone or combined with neurosurgery. Ann Neurol. 1993;33(6):583-590.
8. Barry RL, Byun NE, Tantawy MN, et al. In vivo neuroimaging and behavioral correlates in a rat model of chemotherapy-induced cognitive dysfunction. Brain Imaging Behav. 2018;12(1):87-95.
9. Wu AS, Witgert ME, Lang FF, et al. Neurocognitive function before and after surgery for insular gliomas. J Neurosurg. 2011;115(6):1115-1125.
10. Taylor WD. Depression in the elderly. N Engl J Med. 2014;371(13):1228-1236.
11. Liguori C, Pierantozzi M, Chiaravalloti A, et al. When cognitive decline and depression coexist in the elderly: CSF biomarkers analysis can differentiate Alzheimer’s disease from late-life depression. Front Aging Neurosci. 2018;10:38.
12. Luijendijk HJ, van den Berg JF, Dekker MJHJ, et al. Incidence and recurrence of late-life depression. Arch Gen Psychiatry. 2008;65(12):1394-1401.
13. Potter GG, Steffens DC. Contribution of depression to cognitive impairment and dementia in older adults. Neurologist. 2007;13(3):105-117.
14. Taphoorn MJB, Klein M. Cognitive deficits in adult patients with brain tumours. Lancet Neurol. 2004;3(3):159-168.
15. Inouye SK, Westendorp RGJ, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911-922.
16. Sprung J, Roberts RO, Weingarten TN, et al. Postoperative delirium in elderly patients is associated with subsequent cognitive impairment. Br J Anaesth. 2017;119(2):316-323.
17. Kusljic S, Manias E, Gogos A. Corticosteroid-induced psychiatric disturbances: it is time for pharmacists to take notice. Res Soc Adm Pharm. 2016;12(2):355-360.
18. Cerullo MA. Corticosteroid-induced mania: prepare for the unpredictable. Current Psychiatry. 2006;5(6):43-50.
19. Dubovsky AN, Arvikar S, Stern TA, et al. Steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
20. Habets JGV, Leentjens AFG, Schijns OEMG. Serious and reversible levetiracetam-induced psychiatric symptoms after resection of frontal low-grade glioma: two case histories. Br J Neurosurg. 2017;31(4):471-473.
21
CASE Suicidal behavior, severe headaches
Ms. A, age 60, presents to the emergency department (ED) with depression, suicidal behavior, and 3 days of severe headaches. Neurology is consulted and an MRI is ordered, which shows a 3.0-cm mass lesion in the left temporal lobe with associated vasogenic edema that is suspicious for metastatic disease (Figure).
Ms. A is admitted to the hospital for further workup of her brain lesion. She is started on IV dexamethasone, 10 mg every 6 hours, a glucocorticosteroid, for brain edema, and levetiracetam, 500 mg twice a day, for seizure prophylaxis.
Upon admission, in addition to oncology and neurosurgery, psychiatry is also consulted to evaluate Ms. A for depression and suicidality.
EVALUATION Mood changes and poor judgment
Ms. A has a psychiatric history of depression and alcohol use disorder but says she has not consumed any alcohol in years. Her medical history includes hypertension, diabetes, and stage 4 non-small–cell lung cancer, for which she received surgery and adjuvant chemoradiotherapy 1 year ago.
On initial intake, Ms. A reports that in addition to the headaches, she has also been experiencing worsening depression and suicidal behavior. For the past 2 months, she has had a severely depressed mood, with notable anhedonia, poor appetite, insomnia, low energy, and decreased concentration. The changes in her mental health were triggered by her mother’s death. Three days prior to admission, the patient planned to overdose on antihypertensive pills, but her suicide attempt was interrupted when her family called. She denies any current suicidal ideation, intent, or plan.
According to her family, Ms. A has been increasingly irritable and her personality has changed in the past month. She also has been repeatedly sorting through her neighbors’ garbage.
Ms. A’s current psychiatric medications are duloxetine, 30 mg/d; quetiapine, 50 mg every night at bedtime; and buspirone, 10 mg/d. However, it is unclear if she is consistently taking these medications.
Continue to: On mental status examination...
On mental status examination, Ms. A is calm and she has no abnormal movements. She says she is depressed. Her affect is reactive and labile. She is alert and oriented to person, place, and time. Her attention, registration, and recall are intact. Her executive function is not tested. However, Ms. A’s insight and judgment seem poor.
To address Ms. A’s worsening depression, the psychiatry team increases her duloxetine from 30 to 60 mg/d, and she continues quetiapine, 50 mg every night at bedtime, for mood lability. Buspirone is not continued because she was not taking a therapeutic dosage in the community.
Within 4 days, Ms. A shows improvement in sleep, appetite, and mood. She has no further suicidal ideation.
[polldaddy:10511743]
The authors’ observations
Ms. A had a recurrence of what was presumed to be major depressive disorder (MDD) in the context of her mother’s death. However, she also exhibited irritability, mood lability, and impulsivity, all of which could be part of her depression, or a separate problem related to her brain tumor. Because Ms. A had never displayed bizarre behavior before the past few weeks, it is likely that her CNS lesion was directly affecting her personality and possibly underlying her planned suicide attempt.
Fifty to 80% of patients with CNS tumors, either primary or metastatic, present with psychiatric symptoms.1 Table 11-3 lists common psychiatric symptoms of brain tumors. Unfortunately, there is little reliable evidence that directly correlates tumor location with specific psychiatric symptoms. A 2010 meta-analysis found a statistically significant link between anorexia nervosa and hypothalamic tumors.1 However, for other brain regions, there is only an increased likelihood that any given tumor location will produce psychiatric symptoms.1,4 For instance, compared to patients with tumors in other locations, those with temporal lobe tumors are more likely to present with mood disorders, personality changes, and memory problems.1 In contrast, patients with frontal lobe tumors have an increased likelihood of psychosis, mood disorders, and personality changes.1 Patients with tumors in the pituitary region often present with anxiety.1
Continue to: When considering treatment options...
When considering treatment options for Ms. A, alcohol withdrawal was unlikely given the remote history of alcohol use, low alcohol blood level, and lack of evidence of unstable vital signs or tremor. Although she might have benefited from inpatient psychiatric treatment, this needed to wait until there was a definitive treatment plan for her brain tumor. Finally, although a paraneoplastic syndrome, such as limbic encephalitis, could be causing her psychiatric symptoms, this scenario is less likely with non-small–cell lung cancer.
Although uncommon, CNS tumors can present with psychiatric symptoms as the only manifestation. This is more likely when a patient exhibits new-onset or atypical symptoms, or fails to respond to standard psychiatric treatment.4 Case reports have described patients with brain tumors being misdiagnosed as having a primary psychiatric condition, which delays treatment of their CNS cancer.2 Additionally, frontal and limbic tumors are more likely to present with psychiatric manifestations; up to 90% of patients exhibit altered mental status or personality changes, as did Ms. A.1,4 Clearly, it is easier to identify patients with psychiatric symptoms resulting from a brain tumor when they also present with focal neurologic deficits or systemic symptoms, such as headache or nausea and vomiting. Ms. A presented with severe headaches, which is what led to her early imaging and prompt diagnosis.
Numerous proposed mechanisms might account for the psychiatric symptoms that occur during the course of a brain tumor, including direct injury to neuronal cells, secretion of hormones or other tumor-derived substances, and peri-ictal phenomena.3
TREATMENT Tumor is removed, but memory is impaired
Ms. A is scheduled for craniotomy and surgical resection of the frontal mass. Prior to surgery, Ms. A shows interest in improving her health, cooperates with staff, and seeks her daughter’s input on treatment. One week after admission, Ms. A has her mass resected, which is confirmed on biopsy to be a lung metastasis. Post-surgery, Ms. A receives codeine, 30 mg every 6 hours as needed, for pain; she continues dexamethasone, 4 mg IV every 6 hours, for brain edema and levetiracetam, 500 mg twice a day, for seizure prophylaxis.
On Day 2 after surgery, Ms. A attempts to elope. When she is approached by a psychiatrist on the treatment team, she does not recognize him. Although her long-term memory seems intact, she is unable to remember the details of recent events, including her medical and surgical treatments.
[polldaddy:10511745]
Continue to: The authors' observations
The authors’ observations
Ms. A’s memory impairment may be secondary to a surgically acquired neurocognitive deficit. In the United States, brain metastases represent a significant public health issue, affecting >100,000 patients per year.5 Metastatic lesions are the most common brain tumors. Lung cancer, breast cancer, and melanoma are the leading solid tumors to spread to the CNS.5 In cases of single brain metastasis, similar to Ms. A’s solitary left temporal lobe lesion, surgical resection plays a critical role in treatment. It provides histological confirmation of metastatic disease and can relieve mass effect if present. Studies have shown that combined surgical resection with radiation improves survival relative to patients who undergo radiation therapy alone.6,7
However, the benefits of surgical resection need to be balanced with preservation of neurologic function. Emerging evidence suggests that a majority of patients have surgically-acquired cognitive deficits due to damage of normal surrounding tissues, and these deficits are associated with reduced quality of life.8,9 Further, a study examining glioma surgical resections found that patients with left temporal lobe tumors exhibit more frequent and severe neurocognitive decline than patients with right temporal lobe tumors, especially in domains such as verbal memory.8 Ms. A’s memory impairment was persistent during her postoperative course, which suggests that it was not just an immediate post-surgical phenomenon, but a longer-lasting cognitive change directly related to the resection.
It is also possible that Ms. A had a prior neurocognitive disorder that manifested to a greater degree as a result of the CNS tumor. Ms. A might have had early-onset Alzheimer’s disease, although her intact memory before surgery makes this less likely. Alternatively, she could have had vascular dementia, especially given her long-standing hypertension and diabetes. This might have been missed in the initial evaluation because executive function was not tested. However, the relatively abrupt onset of memory problems after surgery suggests that she had no underlying neurocognitive disorder.
Ms. A’s presumed episode of MDD might also explain her memory changes. Major depressive disorder is increasingly common among geriatric patients, affecting approximately 5% of community-dwelling older adults.10 Its incidence increases with medical comorbidities, as suggested by depression rates of 5% to 10% in the primary care setting vs 37% in patients after critical-care hospitalizations.10 Late-life depression (LLD) occurs in adults age ≥60. Unlike depression in younger patients, LLD is more likely to be associated with cognitive impairment, specifically impairment of executive function and memory.11 The incidence of cognitive impairment in LLD is higher in patients with a history of depression, such as Ms. A.11,12 However, in general, patients who are depressed have memory complaints out of proportion to the clinical findings, and they show poor effort on cognitive testing. Ms. A exhibited neither of these, which makes it less likely that LLD was the exclusive cause of her memory loss.13 Table 214 outlines the management of cognitive deficits in a patient with a brain tumor.
EVALUATION Increasingly agitated and paranoid
After the tumor resection, Ms. A becomes increasingly irritable, uncooperative, and agitated. She repeatedly demands to be discharged. She insists she is fine and refuses medications and further laboratory workup. She becomes paranoid about the nursing staff and believes they are trying to kill her.
Continue to: On psychiatric re-evaluation...
On psychiatric re-evaluation, Ms. A demonstrates pressured speech, perseveration about going home, paranoid delusions, and anger at her family and physicians.
[polldaddy:10511747]
The authors’ observations
Ms. A’s refusal of medications and agitation may be explained by postoperative delirium, a surgical complication that is increasingly common among geriatric patients and is associated with poor clinical outcomes. Delirium is characterized by an acute onset and fluctuating course of symptoms that include inattention, motoric hypo- or hyperactivity, inappropriate behavior, emotional lability, cognitive dysfunction, and psychotic symptoms.15 Risk factors that contribute to postoperative delirium include older age, alcohol use, and poor baseline functional and cognitive status.16 The pathophysiology of delirium is not fully understood, but accumulating evidence suggests that different sets of interacting biologic factors (ie, neurotransmitters and inflammation) contribute to a disruption of large-scale neuronal networks in the brain, resulting in cognitive dysfunction.15 Patients who develop postoperative delirium are more likely to develop long-term cognitive dysfunction and have an increased risk of dementia.16
Another potential source of Ms. A’s agitation is steroid use. Ms. A received IV dexamethasone, 8 to 16 mg/d, around the time of her surgery. Steroids are commonly used to treat brain tumors, particularly when there is vasogenic edema. Steroid psychosis is a term loosely used to describe a wide range of psychiatric symptoms induced by corticosteroids that includes, but is not limited to, depression, mania, psychosis, delirium, and cognitive impairment.17 Steroid-induced psychiatric adverse effects occur in 5% to 18% of patients receiving corticosteroids and often happen early in treatment, although they can occur at any point.18 Corticosteroids influence brain activity via glucocorticoid and mineralocorticoid receptors. These receptors are widely distributed throughout the brain and affect neurotransmitter systems, such as the serotonergic system, that are associated with changes in mood, behavior, and cognition.17 While the adverse psychiatric manifestations of steroid use vary, higher dosages are associated with an increased risk of psychiatric complications; mania is more prevalent early in the course of treatment, and depression is more common with long-term use.17,19 Table 317,18 outlines the evidence-based treatment of corticosteroid-induced adverse psychiatric effects.
Although there are no clinical guidelines or FDA-approved medications for treating steroid-induced psychiatric adverse events, these are best managed by tapering and discontinuing steroids when possible and simultaneously using psychotropic medications to treat psychiatric symptoms. Case reports and limited evidence-based literature have demonstrated that steroid-induced mania responds to mood stabilizers or antipsychotics, while depression can be managed with antidepressants or lithium.17
Additionally, patients with CNS tumors are at risk for seizures and often are prescribed antiepileptics. Because it is easy to administer and does not need to be titrated, levetiracetam is a commonly used agent. However, levetiracetam can cause psychiatric adverse effects, including behavior changes and frank psychosis.20
Continue to: Finally, Ms. A's altered mental status...
Finally, Ms. A’s altered mental status could have been related to opioid intoxication. Opioids are used to manage postsurgical pain, and studies have shown these medications can be a precipitating factor for delirium in geriatric patients.21
TREATMENT Medication adjustments
At the request of the psychiatry team, levetiracetam is discontinued due to its potential for psychiatric adverse effects. The neurosurgery team replaces it with valproic acid, 500 mg every 12 hours. Ms. A is also tapered off steroids fairly rapidly because of the potential for steroid-induced psychiatric adverse effects. Her quetiapine is titrated from 50 to 150 mg every night at bedtime, and duloxetine is discontinued.
OUTCOME Agitation improves dramatically
Ms. A’s new medication regimen dramatically improves her agitation, which allows Ms. A, her family, and the medical team to work together to establish treatment goals. Ms. A ultimately returns home with the assistance of her family. She continues to have memory issues, but with improved emotion regulation. Several months later, Ms. A is readmitted to the hospital because her cancer has progressed despite treatment.
Bottom Line
Brain tumors may present with various psychiatric manifestations that can change during the course of the patient’s treatment. A comprehensive psychiatric evaluation should parse out the interplay between direct effects of the tumor and any adverse effects that are the result of medical and/or surgical interventions to determine the cause of psychiatric symptoms and their appropriate management.
Related Resource
Madhusoodanan S, Ting MB, Farah T, et al. Psychiatric aspects of brain tumors: a review. World J Psychiatry. 2015;5(3):273-285.
Drug Brand Names
Aripiprazole • Abilify
Buspirone • Buspar
Chlorpromazine • Thorazine
Codeine • Codeine systemic
Dexamethasone • Decadron
Duloxetine • Cymbalta
Haloperidol • Haldol
Levetiracetam • Keppra
Lorazepam • Ativan
Lithium • Eskalith, Lithobid
Olanzapine • Zyprexa
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproic acid • Depakene
CASE Suicidal behavior, severe headaches
Ms. A, age 60, presents to the emergency department (ED) with depression, suicidal behavior, and 3 days of severe headaches. Neurology is consulted and an MRI is ordered, which shows a 3.0-cm mass lesion in the left temporal lobe with associated vasogenic edema that is suspicious for metastatic disease (Figure).
Ms. A is admitted to the hospital for further workup of her brain lesion. She is started on IV dexamethasone, 10 mg every 6 hours, a glucocorticosteroid, for brain edema, and levetiracetam, 500 mg twice a day, for seizure prophylaxis.
Upon admission, in addition to oncology and neurosurgery, psychiatry is also consulted to evaluate Ms. A for depression and suicidality.
EVALUATION Mood changes and poor judgment
Ms. A has a psychiatric history of depression and alcohol use disorder but says she has not consumed any alcohol in years. Her medical history includes hypertension, diabetes, and stage 4 non-small–cell lung cancer, for which she received surgery and adjuvant chemoradiotherapy 1 year ago.
On initial intake, Ms. A reports that in addition to the headaches, she has also been experiencing worsening depression and suicidal behavior. For the past 2 months, she has had a severely depressed mood, with notable anhedonia, poor appetite, insomnia, low energy, and decreased concentration. The changes in her mental health were triggered by her mother’s death. Three days prior to admission, the patient planned to overdose on antihypertensive pills, but her suicide attempt was interrupted when her family called. She denies any current suicidal ideation, intent, or plan.
According to her family, Ms. A has been increasingly irritable and her personality has changed in the past month. She also has been repeatedly sorting through her neighbors’ garbage.
Ms. A’s current psychiatric medications are duloxetine, 30 mg/d; quetiapine, 50 mg every night at bedtime; and buspirone, 10 mg/d. However, it is unclear if she is consistently taking these medications.
Continue to: On mental status examination...
On mental status examination, Ms. A is calm and she has no abnormal movements. She says she is depressed. Her affect is reactive and labile. She is alert and oriented to person, place, and time. Her attention, registration, and recall are intact. Her executive function is not tested. However, Ms. A’s insight and judgment seem poor.
To address Ms. A’s worsening depression, the psychiatry team increases her duloxetine from 30 to 60 mg/d, and she continues quetiapine, 50 mg every night at bedtime, for mood lability. Buspirone is not continued because she was not taking a therapeutic dosage in the community.
Within 4 days, Ms. A shows improvement in sleep, appetite, and mood. She has no further suicidal ideation.
[polldaddy:10511743]
The authors’ observations
Ms. A had a recurrence of what was presumed to be major depressive disorder (MDD) in the context of her mother’s death. However, she also exhibited irritability, mood lability, and impulsivity, all of which could be part of her depression, or a separate problem related to her brain tumor. Because Ms. A had never displayed bizarre behavior before the past few weeks, it is likely that her CNS lesion was directly affecting her personality and possibly underlying her planned suicide attempt.
Fifty to 80% of patients with CNS tumors, either primary or metastatic, present with psychiatric symptoms.1 Table 11-3 lists common psychiatric symptoms of brain tumors. Unfortunately, there is little reliable evidence that directly correlates tumor location with specific psychiatric symptoms. A 2010 meta-analysis found a statistically significant link between anorexia nervosa and hypothalamic tumors.1 However, for other brain regions, there is only an increased likelihood that any given tumor location will produce psychiatric symptoms.1,4 For instance, compared to patients with tumors in other locations, those with temporal lobe tumors are more likely to present with mood disorders, personality changes, and memory problems.1 In contrast, patients with frontal lobe tumors have an increased likelihood of psychosis, mood disorders, and personality changes.1 Patients with tumors in the pituitary region often present with anxiety.1
Continue to: When considering treatment options...
When considering treatment options for Ms. A, alcohol withdrawal was unlikely given the remote history of alcohol use, low alcohol blood level, and lack of evidence of unstable vital signs or tremor. Although she might have benefited from inpatient psychiatric treatment, this needed to wait until there was a definitive treatment plan for her brain tumor. Finally, although a paraneoplastic syndrome, such as limbic encephalitis, could be causing her psychiatric symptoms, this scenario is less likely with non-small–cell lung cancer.
Although uncommon, CNS tumors can present with psychiatric symptoms as the only manifestation. This is more likely when a patient exhibits new-onset or atypical symptoms, or fails to respond to standard psychiatric treatment.4 Case reports have described patients with brain tumors being misdiagnosed as having a primary psychiatric condition, which delays treatment of their CNS cancer.2 Additionally, frontal and limbic tumors are more likely to present with psychiatric manifestations; up to 90% of patients exhibit altered mental status or personality changes, as did Ms. A.1,4 Clearly, it is easier to identify patients with psychiatric symptoms resulting from a brain tumor when they also present with focal neurologic deficits or systemic symptoms, such as headache or nausea and vomiting. Ms. A presented with severe headaches, which is what led to her early imaging and prompt diagnosis.
Numerous proposed mechanisms might account for the psychiatric symptoms that occur during the course of a brain tumor, including direct injury to neuronal cells, secretion of hormones or other tumor-derived substances, and peri-ictal phenomena.3
TREATMENT Tumor is removed, but memory is impaired
Ms. A is scheduled for craniotomy and surgical resection of the frontal mass. Prior to surgery, Ms. A shows interest in improving her health, cooperates with staff, and seeks her daughter’s input on treatment. One week after admission, Ms. A has her mass resected, which is confirmed on biopsy to be a lung metastasis. Post-surgery, Ms. A receives codeine, 30 mg every 6 hours as needed, for pain; she continues dexamethasone, 4 mg IV every 6 hours, for brain edema and levetiracetam, 500 mg twice a day, for seizure prophylaxis.
On Day 2 after surgery, Ms. A attempts to elope. When she is approached by a psychiatrist on the treatment team, she does not recognize him. Although her long-term memory seems intact, she is unable to remember the details of recent events, including her medical and surgical treatments.
[polldaddy:10511745]
Continue to: The authors' observations
The authors’ observations
Ms. A’s memory impairment may be secondary to a surgically acquired neurocognitive deficit. In the United States, brain metastases represent a significant public health issue, affecting >100,000 patients per year.5 Metastatic lesions are the most common brain tumors. Lung cancer, breast cancer, and melanoma are the leading solid tumors to spread to the CNS.5 In cases of single brain metastasis, similar to Ms. A’s solitary left temporal lobe lesion, surgical resection plays a critical role in treatment. It provides histological confirmation of metastatic disease and can relieve mass effect if present. Studies have shown that combined surgical resection with radiation improves survival relative to patients who undergo radiation therapy alone.6,7
However, the benefits of surgical resection need to be balanced with preservation of neurologic function. Emerging evidence suggests that a majority of patients have surgically-acquired cognitive deficits due to damage of normal surrounding tissues, and these deficits are associated with reduced quality of life.8,9 Further, a study examining glioma surgical resections found that patients with left temporal lobe tumors exhibit more frequent and severe neurocognitive decline than patients with right temporal lobe tumors, especially in domains such as verbal memory.8 Ms. A’s memory impairment was persistent during her postoperative course, which suggests that it was not just an immediate post-surgical phenomenon, but a longer-lasting cognitive change directly related to the resection.
It is also possible that Ms. A had a prior neurocognitive disorder that manifested to a greater degree as a result of the CNS tumor. Ms. A might have had early-onset Alzheimer’s disease, although her intact memory before surgery makes this less likely. Alternatively, she could have had vascular dementia, especially given her long-standing hypertension and diabetes. This might have been missed in the initial evaluation because executive function was not tested. However, the relatively abrupt onset of memory problems after surgery suggests that she had no underlying neurocognitive disorder.
Ms. A’s presumed episode of MDD might also explain her memory changes. Major depressive disorder is increasingly common among geriatric patients, affecting approximately 5% of community-dwelling older adults.10 Its incidence increases with medical comorbidities, as suggested by depression rates of 5% to 10% in the primary care setting vs 37% in patients after critical-care hospitalizations.10 Late-life depression (LLD) occurs in adults age ≥60. Unlike depression in younger patients, LLD is more likely to be associated with cognitive impairment, specifically impairment of executive function and memory.11 The incidence of cognitive impairment in LLD is higher in patients with a history of depression, such as Ms. A.11,12 However, in general, patients who are depressed have memory complaints out of proportion to the clinical findings, and they show poor effort on cognitive testing. Ms. A exhibited neither of these, which makes it less likely that LLD was the exclusive cause of her memory loss.13 Table 214 outlines the management of cognitive deficits in a patient with a brain tumor.
EVALUATION Increasingly agitated and paranoid
After the tumor resection, Ms. A becomes increasingly irritable, uncooperative, and agitated. She repeatedly demands to be discharged. She insists she is fine and refuses medications and further laboratory workup. She becomes paranoid about the nursing staff and believes they are trying to kill her.
Continue to: On psychiatric re-evaluation...
On psychiatric re-evaluation, Ms. A demonstrates pressured speech, perseveration about going home, paranoid delusions, and anger at her family and physicians.
[polldaddy:10511747]
The authors’ observations
Ms. A’s refusal of medications and agitation may be explained by postoperative delirium, a surgical complication that is increasingly common among geriatric patients and is associated with poor clinical outcomes. Delirium is characterized by an acute onset and fluctuating course of symptoms that include inattention, motoric hypo- or hyperactivity, inappropriate behavior, emotional lability, cognitive dysfunction, and psychotic symptoms.15 Risk factors that contribute to postoperative delirium include older age, alcohol use, and poor baseline functional and cognitive status.16 The pathophysiology of delirium is not fully understood, but accumulating evidence suggests that different sets of interacting biologic factors (ie, neurotransmitters and inflammation) contribute to a disruption of large-scale neuronal networks in the brain, resulting in cognitive dysfunction.15 Patients who develop postoperative delirium are more likely to develop long-term cognitive dysfunction and have an increased risk of dementia.16
Another potential source of Ms. A’s agitation is steroid use. Ms. A received IV dexamethasone, 8 to 16 mg/d, around the time of her surgery. Steroids are commonly used to treat brain tumors, particularly when there is vasogenic edema. Steroid psychosis is a term loosely used to describe a wide range of psychiatric symptoms induced by corticosteroids that includes, but is not limited to, depression, mania, psychosis, delirium, and cognitive impairment.17 Steroid-induced psychiatric adverse effects occur in 5% to 18% of patients receiving corticosteroids and often happen early in treatment, although they can occur at any point.18 Corticosteroids influence brain activity via glucocorticoid and mineralocorticoid receptors. These receptors are widely distributed throughout the brain and affect neurotransmitter systems, such as the serotonergic system, that are associated with changes in mood, behavior, and cognition.17 While the adverse psychiatric manifestations of steroid use vary, higher dosages are associated with an increased risk of psychiatric complications; mania is more prevalent early in the course of treatment, and depression is more common with long-term use.17,19 Table 317,18 outlines the evidence-based treatment of corticosteroid-induced adverse psychiatric effects.
Although there are no clinical guidelines or FDA-approved medications for treating steroid-induced psychiatric adverse events, these are best managed by tapering and discontinuing steroids when possible and simultaneously using psychotropic medications to treat psychiatric symptoms. Case reports and limited evidence-based literature have demonstrated that steroid-induced mania responds to mood stabilizers or antipsychotics, while depression can be managed with antidepressants or lithium.17
Additionally, patients with CNS tumors are at risk for seizures and often are prescribed antiepileptics. Because it is easy to administer and does not need to be titrated, levetiracetam is a commonly used agent. However, levetiracetam can cause psychiatric adverse effects, including behavior changes and frank psychosis.20
Continue to: Finally, Ms. A's altered mental status...
Finally, Ms. A’s altered mental status could have been related to opioid intoxication. Opioids are used to manage postsurgical pain, and studies have shown these medications can be a precipitating factor for delirium in geriatric patients.21
TREATMENT Medication adjustments
At the request of the psychiatry team, levetiracetam is discontinued due to its potential for psychiatric adverse effects. The neurosurgery team replaces it with valproic acid, 500 mg every 12 hours. Ms. A is also tapered off steroids fairly rapidly because of the potential for steroid-induced psychiatric adverse effects. Her quetiapine is titrated from 50 to 150 mg every night at bedtime, and duloxetine is discontinued.
OUTCOME Agitation improves dramatically
Ms. A’s new medication regimen dramatically improves her agitation, which allows Ms. A, her family, and the medical team to work together to establish treatment goals. Ms. A ultimately returns home with the assistance of her family. She continues to have memory issues, but with improved emotion regulation. Several months later, Ms. A is readmitted to the hospital because her cancer has progressed despite treatment.
Bottom Line
Brain tumors may present with various psychiatric manifestations that can change during the course of the patient’s treatment. A comprehensive psychiatric evaluation should parse out the interplay between direct effects of the tumor and any adverse effects that are the result of medical and/or surgical interventions to determine the cause of psychiatric symptoms and their appropriate management.
Related Resource
Madhusoodanan S, Ting MB, Farah T, et al. Psychiatric aspects of brain tumors: a review. World J Psychiatry. 2015;5(3):273-285.
Drug Brand Names
Aripiprazole • Abilify
Buspirone • Buspar
Chlorpromazine • Thorazine
Codeine • Codeine systemic
Dexamethasone • Decadron
Duloxetine • Cymbalta
Haloperidol • Haldol
Levetiracetam • Keppra
Lorazepam • Ativan
Lithium • Eskalith, Lithobid
Olanzapine • Zyprexa
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproic acid • Depakene
1. Madhusoodanan S, Opler MG, Moise D, et al. Brain tumor location and psychiatric symptoms: is there any association? A meta-analysis of published case studies. Expert Rev Neurother. 2010;10(10):1529-1536.
2. Bunevicius A, Deltuva VP, Deltuviene D, et al. Brain lesions manifesting as psychiatric disorders: eight cases. CNS Spectr. 2008;13(11):950-958.
3. Pearl ML, Talgat G, Valea FA, et al. Psychiatric symptoms due to brain metastases. Med Update Psychiatr. 1998;3(4):91-94.
4. Madhusoodanan S, Danan D, Moise D. Psychiatric manifestations of brain tumors: diagnostic implications. Expert Rev Neurother. 2007;7(4):343-349.
5. Ferguson SD, Wagner KM, Prabhu SS, et al. Neurosurgical management of brain metastases. Clin Exp Metastasis. 2017;34(6-7):377-389.
6. Husain ZA, Regine WF, Kwok Y, et al. Brain metastases: contemporary management and future directions. Eur J Clin Med Oncol. 2011;3(3):38-45.
7. Vecht CJ, Haaxmareiche H, Noordijk EM, et al. Treatment of single brain metastasis - radiotherapy alone or combined with neurosurgery. Ann Neurol. 1993;33(6):583-590.
8. Barry RL, Byun NE, Tantawy MN, et al. In vivo neuroimaging and behavioral correlates in a rat model of chemotherapy-induced cognitive dysfunction. Brain Imaging Behav. 2018;12(1):87-95.
9. Wu AS, Witgert ME, Lang FF, et al. Neurocognitive function before and after surgery for insular gliomas. J Neurosurg. 2011;115(6):1115-1125.
10. Taylor WD. Depression in the elderly. N Engl J Med. 2014;371(13):1228-1236.
11. Liguori C, Pierantozzi M, Chiaravalloti A, et al. When cognitive decline and depression coexist in the elderly: CSF biomarkers analysis can differentiate Alzheimer’s disease from late-life depression. Front Aging Neurosci. 2018;10:38.
12. Luijendijk HJ, van den Berg JF, Dekker MJHJ, et al. Incidence and recurrence of late-life depression. Arch Gen Psychiatry. 2008;65(12):1394-1401.
13. Potter GG, Steffens DC. Contribution of depression to cognitive impairment and dementia in older adults. Neurologist. 2007;13(3):105-117.
14. Taphoorn MJB, Klein M. Cognitive deficits in adult patients with brain tumours. Lancet Neurol. 2004;3(3):159-168.
15. Inouye SK, Westendorp RGJ, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911-922.
16. Sprung J, Roberts RO, Weingarten TN, et al. Postoperative delirium in elderly patients is associated with subsequent cognitive impairment. Br J Anaesth. 2017;119(2):316-323.
17. Kusljic S, Manias E, Gogos A. Corticosteroid-induced psychiatric disturbances: it is time for pharmacists to take notice. Res Soc Adm Pharm. 2016;12(2):355-360.
18. Cerullo MA. Corticosteroid-induced mania: prepare for the unpredictable. Current Psychiatry. 2006;5(6):43-50.
19. Dubovsky AN, Arvikar S, Stern TA, et al. Steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
20. Habets JGV, Leentjens AFG, Schijns OEMG. Serious and reversible levetiracetam-induced psychiatric symptoms after resection of frontal low-grade glioma: two case histories. Br J Neurosurg. 2017;31(4):471-473.
21
1. Madhusoodanan S, Opler MG, Moise D, et al. Brain tumor location and psychiatric symptoms: is there any association? A meta-analysis of published case studies. Expert Rev Neurother. 2010;10(10):1529-1536.
2. Bunevicius A, Deltuva VP, Deltuviene D, et al. Brain lesions manifesting as psychiatric disorders: eight cases. CNS Spectr. 2008;13(11):950-958.
3. Pearl ML, Talgat G, Valea FA, et al. Psychiatric symptoms due to brain metastases. Med Update Psychiatr. 1998;3(4):91-94.
4. Madhusoodanan S, Danan D, Moise D. Psychiatric manifestations of brain tumors: diagnostic implications. Expert Rev Neurother. 2007;7(4):343-349.
5. Ferguson SD, Wagner KM, Prabhu SS, et al. Neurosurgical management of brain metastases. Clin Exp Metastasis. 2017;34(6-7):377-389.
6. Husain ZA, Regine WF, Kwok Y, et al. Brain metastases: contemporary management and future directions. Eur J Clin Med Oncol. 2011;3(3):38-45.
7. Vecht CJ, Haaxmareiche H, Noordijk EM, et al. Treatment of single brain metastasis - radiotherapy alone or combined with neurosurgery. Ann Neurol. 1993;33(6):583-590.
8. Barry RL, Byun NE, Tantawy MN, et al. In vivo neuroimaging and behavioral correlates in a rat model of chemotherapy-induced cognitive dysfunction. Brain Imaging Behav. 2018;12(1):87-95.
9. Wu AS, Witgert ME, Lang FF, et al. Neurocognitive function before and after surgery for insular gliomas. J Neurosurg. 2011;115(6):1115-1125.
10. Taylor WD. Depression in the elderly. N Engl J Med. 2014;371(13):1228-1236.
11. Liguori C, Pierantozzi M, Chiaravalloti A, et al. When cognitive decline and depression coexist in the elderly: CSF biomarkers analysis can differentiate Alzheimer’s disease from late-life depression. Front Aging Neurosci. 2018;10:38.
12. Luijendijk HJ, van den Berg JF, Dekker MJHJ, et al. Incidence and recurrence of late-life depression. Arch Gen Psychiatry. 2008;65(12):1394-1401.
13. Potter GG, Steffens DC. Contribution of depression to cognitive impairment and dementia in older adults. Neurologist. 2007;13(3):105-117.
14. Taphoorn MJB, Klein M. Cognitive deficits in adult patients with brain tumours. Lancet Neurol. 2004;3(3):159-168.
15. Inouye SK, Westendorp RGJ, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911-922.
16. Sprung J, Roberts RO, Weingarten TN, et al. Postoperative delirium in elderly patients is associated with subsequent cognitive impairment. Br J Anaesth. 2017;119(2):316-323.
17. Kusljic S, Manias E, Gogos A. Corticosteroid-induced psychiatric disturbances: it is time for pharmacists to take notice. Res Soc Adm Pharm. 2016;12(2):355-360.
18. Cerullo MA. Corticosteroid-induced mania: prepare for the unpredictable. Current Psychiatry. 2006;5(6):43-50.
19. Dubovsky AN, Arvikar S, Stern TA, et al. Steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
20. Habets JGV, Leentjens AFG, Schijns OEMG. Serious and reversible levetiracetam-induced psychiatric symptoms after resection of frontal low-grade glioma: two case histories. Br J Neurosurg. 2017;31(4):471-473.
21
Incomplete MS relapse recovery predicted greater long-term disability
WEST PALM BEACH, FLA. – Failure to recover completely from early relapses in multiple sclerosis (MS) is significantly associated with higher long-term disability, according to research presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Incomplete recovery thus should be given more consideration when evaluating research and clinical practice outcomes, the study investigators cautioned.
“We found that the recovery from early relapses is an important predictor of future disability,” first author Marinos G. Sotiropoulos, MD, of the department of neurology, Brigham and Women’s Hospital, Boston, said in an interview. “It should be incorporated in future predictive models of disease severity and clinical trials, [and] it could be useful in clinical decision making as well.”
Incomplete recovery from relapses is known to be linked to disability progression and to the likelihood of transitioning to secondary progressive MS. Research on its role in longer-term outcomes is lacking, however.
To investigate the effect of incomplete relapse recovery in the first 3 years of MS on rates of disability at 10 years, Dr. Sotiropoulos and colleagues evaluated data on 360 patients enrolled in the CLIMB (Comprehensive Longitudinal Investigation in Multiple Sclerosis at Brigham and Women’s Hospital) study. CLIMB is a natural history study spanning 20 years, with more than 2,000 patients.
Patients were included if at least 8.5 years had passed since their first documented symptom, if they were at least 18 years at their first visit to the Partners MS Center, if that visit occurred within 1 year of their first symptom, and if they had a diagnosis of relapsing-remitting MS or secondary progressive MS.
Among the 308 patients included in the study, 74% were female and 89% were white, with a mean age at the first symptom of 35.9 years.
A total of 403 early attacks from those 308 patients were included in the study. Half of the attacks (50.4%) were followed by incomplete recovery after 6 months, defined specifically as an increase in the Expanded Disability Status Scale (EDSS) scores from baseline to at least 6 months after the onset of the attack.
As of their 10-year visit, 27.3% of patients had a normal examination, defined as EDSS 0, and 64.1% had no significant disability (EDSS less than 2). The mean EDSS at 10 years was 1.52.
Patients’ recovery index, defined as the percentage of early attacks that recovered completely, was significantly associated with 10-year EDSS scores (P less than .001).
Patient age at first symptom was also a significant predictor of 10-year disability (P less than .004). Factors that were significantly associated with incomplete relapse recovery were the duration of time from first symptom (P less than .001) and moderate severity of the relapse (P = .029).
With the type of drug treatment likely representing an important factor in whether a patient has incomplete relapse recovery, the issue should be the subject of further research, Dr. Sotiropoulos said.
“This is something that is important to look at because none of the clinical trials for the drugs we currently have looked at relapse recovery as an outcome,” he explained.
“There have been some post hoc analyses [that] have shown that some of the new medications can improve recovery from relapses, but there is a lot to look into now that we know relapse recovery is an important clinical parameter,” he said. “We have to factor in the treatment effect in preventing residual disability after relapses.”
The findings suggest that “patients with incomplete early recovery might be considered for highly effective disease-modifying therapy,” added senior author Tanuja Chitnis, MD, also of the department of neurology at Brigham and Women’s Hospital. “We are now analyzing the biological mechanisms associated with relapse recovery.”
The authors of a recent study that echoes the importance of relapse recovery call it “the forgotten variable in multiple sclerosis clinical trials.” In that study, the researchers found an increased likelihood of a benign disease course among patients who received immediate disease-modifying therapy (DMT) initiation after failing to have a good recovery from an initial relapse (Neurol Neuroimmunol Neuroinflamm. 2019 Dec 17;7[2]).
“Some clinicians may choose to hold off DMTs because the patient may not have high disease activity levels,” Burcu Zeydan, MD, a coauthor of that study and an assistant professor of radiology in the Center of MS and Autoimmune Neurology at the Mayo Clinic, Rochester, Minn., said in an interview.
“What these studies add is that, if a patient is a poor recoverer despite not having highly active disease, that patient should be considered for immediate treatment initiation,” she said. “Otherwise, there is the possibility of a next relapse, which may not happen often. But when it happens, it may lead to more residual deficit with additional disability burden.”
The CLIMB study received funding from Mallinckrodt and the National MS Society Nancy Davis Center Without Walls. Dr. Sotiropoulos has received research support from Mallinckrodt. Dr. Chitnis has served on advisory boards for Biogen, Novartis, and Sanofi-Genzyme, and she has received research support from the Department of Defense, National MS Society, Guthy-Jackson Charitable Foundation, Novartis, Octave, Serono, and Verily. Dr. Zeydan had no disclosures to report.
SOURCE: Sotiropoulos MG et al. ACTRIMS Forum 2020. Abstract LB 317.
WEST PALM BEACH, FLA. – Failure to recover completely from early relapses in multiple sclerosis (MS) is significantly associated with higher long-term disability, according to research presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Incomplete recovery thus should be given more consideration when evaluating research and clinical practice outcomes, the study investigators cautioned.
“We found that the recovery from early relapses is an important predictor of future disability,” first author Marinos G. Sotiropoulos, MD, of the department of neurology, Brigham and Women’s Hospital, Boston, said in an interview. “It should be incorporated in future predictive models of disease severity and clinical trials, [and] it could be useful in clinical decision making as well.”
Incomplete recovery from relapses is known to be linked to disability progression and to the likelihood of transitioning to secondary progressive MS. Research on its role in longer-term outcomes is lacking, however.
To investigate the effect of incomplete relapse recovery in the first 3 years of MS on rates of disability at 10 years, Dr. Sotiropoulos and colleagues evaluated data on 360 patients enrolled in the CLIMB (Comprehensive Longitudinal Investigation in Multiple Sclerosis at Brigham and Women’s Hospital) study. CLIMB is a natural history study spanning 20 years, with more than 2,000 patients.
Patients were included if at least 8.5 years had passed since their first documented symptom, if they were at least 18 years at their first visit to the Partners MS Center, if that visit occurred within 1 year of their first symptom, and if they had a diagnosis of relapsing-remitting MS or secondary progressive MS.
Among the 308 patients included in the study, 74% were female and 89% were white, with a mean age at the first symptom of 35.9 years.
A total of 403 early attacks from those 308 patients were included in the study. Half of the attacks (50.4%) were followed by incomplete recovery after 6 months, defined specifically as an increase in the Expanded Disability Status Scale (EDSS) scores from baseline to at least 6 months after the onset of the attack.
As of their 10-year visit, 27.3% of patients had a normal examination, defined as EDSS 0, and 64.1% had no significant disability (EDSS less than 2). The mean EDSS at 10 years was 1.52.
Patients’ recovery index, defined as the percentage of early attacks that recovered completely, was significantly associated with 10-year EDSS scores (P less than .001).
Patient age at first symptom was also a significant predictor of 10-year disability (P less than .004). Factors that were significantly associated with incomplete relapse recovery were the duration of time from first symptom (P less than .001) and moderate severity of the relapse (P = .029).
With the type of drug treatment likely representing an important factor in whether a patient has incomplete relapse recovery, the issue should be the subject of further research, Dr. Sotiropoulos said.
“This is something that is important to look at because none of the clinical trials for the drugs we currently have looked at relapse recovery as an outcome,” he explained.
“There have been some post hoc analyses [that] have shown that some of the new medications can improve recovery from relapses, but there is a lot to look into now that we know relapse recovery is an important clinical parameter,” he said. “We have to factor in the treatment effect in preventing residual disability after relapses.”
The findings suggest that “patients with incomplete early recovery might be considered for highly effective disease-modifying therapy,” added senior author Tanuja Chitnis, MD, also of the department of neurology at Brigham and Women’s Hospital. “We are now analyzing the biological mechanisms associated with relapse recovery.”
The authors of a recent study that echoes the importance of relapse recovery call it “the forgotten variable in multiple sclerosis clinical trials.” In that study, the researchers found an increased likelihood of a benign disease course among patients who received immediate disease-modifying therapy (DMT) initiation after failing to have a good recovery from an initial relapse (Neurol Neuroimmunol Neuroinflamm. 2019 Dec 17;7[2]).
“Some clinicians may choose to hold off DMTs because the patient may not have high disease activity levels,” Burcu Zeydan, MD, a coauthor of that study and an assistant professor of radiology in the Center of MS and Autoimmune Neurology at the Mayo Clinic, Rochester, Minn., said in an interview.
“What these studies add is that, if a patient is a poor recoverer despite not having highly active disease, that patient should be considered for immediate treatment initiation,” she said. “Otherwise, there is the possibility of a next relapse, which may not happen often. But when it happens, it may lead to more residual deficit with additional disability burden.”
The CLIMB study received funding from Mallinckrodt and the National MS Society Nancy Davis Center Without Walls. Dr. Sotiropoulos has received research support from Mallinckrodt. Dr. Chitnis has served on advisory boards for Biogen, Novartis, and Sanofi-Genzyme, and she has received research support from the Department of Defense, National MS Society, Guthy-Jackson Charitable Foundation, Novartis, Octave, Serono, and Verily. Dr. Zeydan had no disclosures to report.
SOURCE: Sotiropoulos MG et al. ACTRIMS Forum 2020. Abstract LB 317.
WEST PALM BEACH, FLA. – Failure to recover completely from early relapses in multiple sclerosis (MS) is significantly associated with higher long-term disability, according to research presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Incomplete recovery thus should be given more consideration when evaluating research and clinical practice outcomes, the study investigators cautioned.
“We found that the recovery from early relapses is an important predictor of future disability,” first author Marinos G. Sotiropoulos, MD, of the department of neurology, Brigham and Women’s Hospital, Boston, said in an interview. “It should be incorporated in future predictive models of disease severity and clinical trials, [and] it could be useful in clinical decision making as well.”
Incomplete recovery from relapses is known to be linked to disability progression and to the likelihood of transitioning to secondary progressive MS. Research on its role in longer-term outcomes is lacking, however.
To investigate the effect of incomplete relapse recovery in the first 3 years of MS on rates of disability at 10 years, Dr. Sotiropoulos and colleagues evaluated data on 360 patients enrolled in the CLIMB (Comprehensive Longitudinal Investigation in Multiple Sclerosis at Brigham and Women’s Hospital) study. CLIMB is a natural history study spanning 20 years, with more than 2,000 patients.
Patients were included if at least 8.5 years had passed since their first documented symptom, if they were at least 18 years at their first visit to the Partners MS Center, if that visit occurred within 1 year of their first symptom, and if they had a diagnosis of relapsing-remitting MS or secondary progressive MS.
Among the 308 patients included in the study, 74% were female and 89% were white, with a mean age at the first symptom of 35.9 years.
A total of 403 early attacks from those 308 patients were included in the study. Half of the attacks (50.4%) were followed by incomplete recovery after 6 months, defined specifically as an increase in the Expanded Disability Status Scale (EDSS) scores from baseline to at least 6 months after the onset of the attack.
As of their 10-year visit, 27.3% of patients had a normal examination, defined as EDSS 0, and 64.1% had no significant disability (EDSS less than 2). The mean EDSS at 10 years was 1.52.
Patients’ recovery index, defined as the percentage of early attacks that recovered completely, was significantly associated with 10-year EDSS scores (P less than .001).
Patient age at first symptom was also a significant predictor of 10-year disability (P less than .004). Factors that were significantly associated with incomplete relapse recovery were the duration of time from first symptom (P less than .001) and moderate severity of the relapse (P = .029).
With the type of drug treatment likely representing an important factor in whether a patient has incomplete relapse recovery, the issue should be the subject of further research, Dr. Sotiropoulos said.
“This is something that is important to look at because none of the clinical trials for the drugs we currently have looked at relapse recovery as an outcome,” he explained.
“There have been some post hoc analyses [that] have shown that some of the new medications can improve recovery from relapses, but there is a lot to look into now that we know relapse recovery is an important clinical parameter,” he said. “We have to factor in the treatment effect in preventing residual disability after relapses.”
The findings suggest that “patients with incomplete early recovery might be considered for highly effective disease-modifying therapy,” added senior author Tanuja Chitnis, MD, also of the department of neurology at Brigham and Women’s Hospital. “We are now analyzing the biological mechanisms associated with relapse recovery.”
The authors of a recent study that echoes the importance of relapse recovery call it “the forgotten variable in multiple sclerosis clinical trials.” In that study, the researchers found an increased likelihood of a benign disease course among patients who received immediate disease-modifying therapy (DMT) initiation after failing to have a good recovery from an initial relapse (Neurol Neuroimmunol Neuroinflamm. 2019 Dec 17;7[2]).
“Some clinicians may choose to hold off DMTs because the patient may not have high disease activity levels,” Burcu Zeydan, MD, a coauthor of that study and an assistant professor of radiology in the Center of MS and Autoimmune Neurology at the Mayo Clinic, Rochester, Minn., said in an interview.
“What these studies add is that, if a patient is a poor recoverer despite not having highly active disease, that patient should be considered for immediate treatment initiation,” she said. “Otherwise, there is the possibility of a next relapse, which may not happen often. But when it happens, it may lead to more residual deficit with additional disability burden.”
The CLIMB study received funding from Mallinckrodt and the National MS Society Nancy Davis Center Without Walls. Dr. Sotiropoulos has received research support from Mallinckrodt. Dr. Chitnis has served on advisory boards for Biogen, Novartis, and Sanofi-Genzyme, and she has received research support from the Department of Defense, National MS Society, Guthy-Jackson Charitable Foundation, Novartis, Octave, Serono, and Verily. Dr. Zeydan had no disclosures to report.
SOURCE: Sotiropoulos MG et al. ACTRIMS Forum 2020. Abstract LB 317.
REPORTING FROM ACTRIMS FORUM 2020