User login
BCL-2 inhibitor shows potential for treating MM

to an attendee at ASCO 2015
© ASCO/Zach Boyden-Holmes
CHICAGO—A BCL-2 inhibitor that has shown activity in patients with chronic and acute leukemias could be a feasible treatment option for multiple myeloma (MM) too, according to research presented at the 2015 ASCO Annual Meeting.
In a pair of phase 1 studies, the drug, venetoclax, prompted responses in certain patients with relapsed or refractory MM, both when given alone and in combination with bortezomib and dexamethasone.
As monotherapy, venetoclax induced complete responses (CRs) in 2 patients with t(11;14), but the overall response rate (ORR) was low, and most patients discontinued treatment, largely due to progression.
Venetoclax in combination with bortezomib and dexamethasone prompted a response in nearly half of the patients studied, but more than half discontinued treatment. The combination produced a high ORR in patients who were bortezomib-naïve and bortezomib-sensitive, but there was no response among bortezomib-refractory patients.
“Multiple myeloma remains a high area of unmet medical need, and additional research to identify new therapies is important,” said investigator Cyrille Touzeau, MD, of the University of Nantes in France.
“The response rates shown in these studies suggest potential of venetoclax in this patient population and warrant further evaluation.”
Dr Touzeau and his colleagues presented these studies at ASCO as abstracts 8580 and 8576. Both studies were funded by AbbVie, the company developing venetoclax.
Venetoclax in combination
In a phase 1b study (abstract 8580*), the researchers evaluated venetoclax in combination with bortezomib and dexamethasone in 38 patients with relapsed or refractory MM.
The patients’ median age was 65 (range, 38-79). Five patients were t(11;14)-positive, 3 were t(4;14)-positive, 9 had del17p, and 19 had del13q. They had received a median of 5 prior lines of therapy (range, 1-15). Ten patients were bortezomib-refractory, 21 were lenalidomide-refractory, and 8 patients were refractory to both drugs.
The patients received venetoclax at doses ranging from 50 mg to 600 mg daily (with a 1-week lead-in period for the 50 mg cohort). On cycles 1-8, patients received dexamethasone (20 mg) and bortezomib (1.3 mg/m2) with venetoclax on days 1, 4, 8, and 11. They received dexamethasone (20 mg) with venetoclax on days 2, 5, 9, and 12.
On cycles 9-11, patients received dexamethasone (20 mg) and bortezomib (1.3 mg/m2) with venetoclax on days 1, 8, 15, and 22. Patients received venetoclax monotherapy for cycles 12 and beyond. They also received prophylaxis for tumor lysis syndrome.
Twenty-five patients (66%) discontinued treatment, 19 due to disease progression (including 4 deaths), 2 due to adverse events (AEs), 3 due to patient decision, and 1 due to withdrawn consent.
The ORR was 47% among the 36 evaluable patients, 83% for bortezomib-naïve patients (5/6), 60% for bortezomib-sensitive patients (12/20), and 0% for bortezomib-refractory patients (0/10).
Two patients had a stringent CR, 1 had a CR, 4 had a very good partial response, 10 had a partial response, and 2 had a minimal response. Six patients had stable disease, and 9 progressed.
The most common AEs—occurring in at least 20% of patients—were constipation (37%), diarrhea (32%), insomnia (32%), thrombocytopenia (29%), peripheral neuropathy (26%), asthenia (26%), dyspnea (26%), peripheral edema (24%), and anemia (21%).
Grade 3/4 AEs—occurring in at least 10% of patients—were thrombocytopenia (21%), anemia (13%), and dyspnea (11%). Serious AEs—each occurring in 5% of patients—were cardiac failure, pyrexia, sepsis, respiratory failure, pneumonia, and embolism.
Venetoclax as monotherapy
In a phase 1 study (abstract 8576*), researchers evaluated venetoclax as monotherapy in 29 patients with relapsed or refractory MM. Eleven patients were t(11;14)-positive, 2 were t(4;14)-positive, 4 had del17p, and 14 had del13q.
The patients’ median age was 66 (range, 42-79), and they had received a median of 6 prior therapies (range, 1-13). Fifteen patients were bortezomib-refractory, 12 were refractory to lenalidomide, and 10 patients were refractory to both drugs.
After a 2-week lead-in period, patients received venetoclax daily on a 21-day cycle, ranging from 300 mg to 1200 mg on a 3+3 design. They also received prophylaxis for tumor lysis syndrome. Patients who progressed during treatment were allowed to receive dexamethasone as well and continue the study.
Twenty-three patients (79%) discontinued treatment, 18 due to disease progression, 4 due to AEs, and 1 due to patient decision.
The ORR was 7% (n=2). Both patients achieved a CR, and both were t(11;14)-positive. One of the patients is in the 600 mg cohort and is still responding to treatment. The duration of response, thus far, is 2.1 months.
The other patient with a CR is in the 900 mg dose cohort. This patient is still responding as well, and the response duration, thus far, is 2.8 months.
Common AEs—occurring in at least 20% of patients—were diarrhea (41%), nausea (41%), fatigue (24%), vomiting (21%), asthenia (21%), and neutropenia (21%).
Grade 3/4 AEs—occurring in at least 10% of patients—were thrombocytopenia (24%), neutropenia (14%), and anemia (10%). Serious AEs—each occurring in 7% of patients—were pyrexia, malignant neoplasm progression, and cough.
Two patients had dose-limiting toxicities at the 600 mg dose of venetoclax. However, the researchers said the maximum tolerated dose was not reached, and the recommended phase 2 dose is 1200 mg. ![]()
*Information in the abstract differs from that presented at the meeting.

to an attendee at ASCO 2015
© ASCO/Zach Boyden-Holmes
CHICAGO—A BCL-2 inhibitor that has shown activity in patients with chronic and acute leukemias could be a feasible treatment option for multiple myeloma (MM) too, according to research presented at the 2015 ASCO Annual Meeting.
In a pair of phase 1 studies, the drug, venetoclax, prompted responses in certain patients with relapsed or refractory MM, both when given alone and in combination with bortezomib and dexamethasone.
As monotherapy, venetoclax induced complete responses (CRs) in 2 patients with t(11;14), but the overall response rate (ORR) was low, and most patients discontinued treatment, largely due to progression.
Venetoclax in combination with bortezomib and dexamethasone prompted a response in nearly half of the patients studied, but more than half discontinued treatment. The combination produced a high ORR in patients who were bortezomib-naïve and bortezomib-sensitive, but there was no response among bortezomib-refractory patients.
“Multiple myeloma remains a high area of unmet medical need, and additional research to identify new therapies is important,” said investigator Cyrille Touzeau, MD, of the University of Nantes in France.
“The response rates shown in these studies suggest potential of venetoclax in this patient population and warrant further evaluation.”
Dr Touzeau and his colleagues presented these studies at ASCO as abstracts 8580 and 8576. Both studies were funded by AbbVie, the company developing venetoclax.
Venetoclax in combination
In a phase 1b study (abstract 8580*), the researchers evaluated venetoclax in combination with bortezomib and dexamethasone in 38 patients with relapsed or refractory MM.
The patients’ median age was 65 (range, 38-79). Five patients were t(11;14)-positive, 3 were t(4;14)-positive, 9 had del17p, and 19 had del13q. They had received a median of 5 prior lines of therapy (range, 1-15). Ten patients were bortezomib-refractory, 21 were lenalidomide-refractory, and 8 patients were refractory to both drugs.
The patients received venetoclax at doses ranging from 50 mg to 600 mg daily (with a 1-week lead-in period for the 50 mg cohort). On cycles 1-8, patients received dexamethasone (20 mg) and bortezomib (1.3 mg/m2) with venetoclax on days 1, 4, 8, and 11. They received dexamethasone (20 mg) with venetoclax on days 2, 5, 9, and 12.
On cycles 9-11, patients received dexamethasone (20 mg) and bortezomib (1.3 mg/m2) with venetoclax on days 1, 8, 15, and 22. Patients received venetoclax monotherapy for cycles 12 and beyond. They also received prophylaxis for tumor lysis syndrome.
Twenty-five patients (66%) discontinued treatment, 19 due to disease progression (including 4 deaths), 2 due to adverse events (AEs), 3 due to patient decision, and 1 due to withdrawn consent.
The ORR was 47% among the 36 evaluable patients, 83% for bortezomib-naïve patients (5/6), 60% for bortezomib-sensitive patients (12/20), and 0% for bortezomib-refractory patients (0/10).
Two patients had a stringent CR, 1 had a CR, 4 had a very good partial response, 10 had a partial response, and 2 had a minimal response. Six patients had stable disease, and 9 progressed.
The most common AEs—occurring in at least 20% of patients—were constipation (37%), diarrhea (32%), insomnia (32%), thrombocytopenia (29%), peripheral neuropathy (26%), asthenia (26%), dyspnea (26%), peripheral edema (24%), and anemia (21%).
Grade 3/4 AEs—occurring in at least 10% of patients—were thrombocytopenia (21%), anemia (13%), and dyspnea (11%). Serious AEs—each occurring in 5% of patients—were cardiac failure, pyrexia, sepsis, respiratory failure, pneumonia, and embolism.
Venetoclax as monotherapy
In a phase 1 study (abstract 8576*), researchers evaluated venetoclax as monotherapy in 29 patients with relapsed or refractory MM. Eleven patients were t(11;14)-positive, 2 were t(4;14)-positive, 4 had del17p, and 14 had del13q.
The patients’ median age was 66 (range, 42-79), and they had received a median of 6 prior therapies (range, 1-13). Fifteen patients were bortezomib-refractory, 12 were refractory to lenalidomide, and 10 patients were refractory to both drugs.
After a 2-week lead-in period, patients received venetoclax daily on a 21-day cycle, ranging from 300 mg to 1200 mg on a 3+3 design. They also received prophylaxis for tumor lysis syndrome. Patients who progressed during treatment were allowed to receive dexamethasone as well and continue the study.
Twenty-three patients (79%) discontinued treatment, 18 due to disease progression, 4 due to AEs, and 1 due to patient decision.
The ORR was 7% (n=2). Both patients achieved a CR, and both were t(11;14)-positive. One of the patients is in the 600 mg cohort and is still responding to treatment. The duration of response, thus far, is 2.1 months.
The other patient with a CR is in the 900 mg dose cohort. This patient is still responding as well, and the response duration, thus far, is 2.8 months.
Common AEs—occurring in at least 20% of patients—were diarrhea (41%), nausea (41%), fatigue (24%), vomiting (21%), asthenia (21%), and neutropenia (21%).
Grade 3/4 AEs—occurring in at least 10% of patients—were thrombocytopenia (24%), neutropenia (14%), and anemia (10%). Serious AEs—each occurring in 7% of patients—were pyrexia, malignant neoplasm progression, and cough.
Two patients had dose-limiting toxicities at the 600 mg dose of venetoclax. However, the researchers said the maximum tolerated dose was not reached, and the recommended phase 2 dose is 1200 mg. ![]()
*Information in the abstract differs from that presented at the meeting.

to an attendee at ASCO 2015
© ASCO/Zach Boyden-Holmes
CHICAGO—A BCL-2 inhibitor that has shown activity in patients with chronic and acute leukemias could be a feasible treatment option for multiple myeloma (MM) too, according to research presented at the 2015 ASCO Annual Meeting.
In a pair of phase 1 studies, the drug, venetoclax, prompted responses in certain patients with relapsed or refractory MM, both when given alone and in combination with bortezomib and dexamethasone.
As monotherapy, venetoclax induced complete responses (CRs) in 2 patients with t(11;14), but the overall response rate (ORR) was low, and most patients discontinued treatment, largely due to progression.
Venetoclax in combination with bortezomib and dexamethasone prompted a response in nearly half of the patients studied, but more than half discontinued treatment. The combination produced a high ORR in patients who were bortezomib-naïve and bortezomib-sensitive, but there was no response among bortezomib-refractory patients.
“Multiple myeloma remains a high area of unmet medical need, and additional research to identify new therapies is important,” said investigator Cyrille Touzeau, MD, of the University of Nantes in France.
“The response rates shown in these studies suggest potential of venetoclax in this patient population and warrant further evaluation.”
Dr Touzeau and his colleagues presented these studies at ASCO as abstracts 8580 and 8576. Both studies were funded by AbbVie, the company developing venetoclax.
Venetoclax in combination
In a phase 1b study (abstract 8580*), the researchers evaluated venetoclax in combination with bortezomib and dexamethasone in 38 patients with relapsed or refractory MM.
The patients’ median age was 65 (range, 38-79). Five patients were t(11;14)-positive, 3 were t(4;14)-positive, 9 had del17p, and 19 had del13q. They had received a median of 5 prior lines of therapy (range, 1-15). Ten patients were bortezomib-refractory, 21 were lenalidomide-refractory, and 8 patients were refractory to both drugs.
The patients received venetoclax at doses ranging from 50 mg to 600 mg daily (with a 1-week lead-in period for the 50 mg cohort). On cycles 1-8, patients received dexamethasone (20 mg) and bortezomib (1.3 mg/m2) with venetoclax on days 1, 4, 8, and 11. They received dexamethasone (20 mg) with venetoclax on days 2, 5, 9, and 12.
On cycles 9-11, patients received dexamethasone (20 mg) and bortezomib (1.3 mg/m2) with venetoclax on days 1, 8, 15, and 22. Patients received venetoclax monotherapy for cycles 12 and beyond. They also received prophylaxis for tumor lysis syndrome.
Twenty-five patients (66%) discontinued treatment, 19 due to disease progression (including 4 deaths), 2 due to adverse events (AEs), 3 due to patient decision, and 1 due to withdrawn consent.
The ORR was 47% among the 36 evaluable patients, 83% for bortezomib-naïve patients (5/6), 60% for bortezomib-sensitive patients (12/20), and 0% for bortezomib-refractory patients (0/10).
Two patients had a stringent CR, 1 had a CR, 4 had a very good partial response, 10 had a partial response, and 2 had a minimal response. Six patients had stable disease, and 9 progressed.
The most common AEs—occurring in at least 20% of patients—were constipation (37%), diarrhea (32%), insomnia (32%), thrombocytopenia (29%), peripheral neuropathy (26%), asthenia (26%), dyspnea (26%), peripheral edema (24%), and anemia (21%).
Grade 3/4 AEs—occurring in at least 10% of patients—were thrombocytopenia (21%), anemia (13%), and dyspnea (11%). Serious AEs—each occurring in 5% of patients—were cardiac failure, pyrexia, sepsis, respiratory failure, pneumonia, and embolism.
Venetoclax as monotherapy
In a phase 1 study (abstract 8576*), researchers evaluated venetoclax as monotherapy in 29 patients with relapsed or refractory MM. Eleven patients were t(11;14)-positive, 2 were t(4;14)-positive, 4 had del17p, and 14 had del13q.
The patients’ median age was 66 (range, 42-79), and they had received a median of 6 prior therapies (range, 1-13). Fifteen patients were bortezomib-refractory, 12 were refractory to lenalidomide, and 10 patients were refractory to both drugs.
After a 2-week lead-in period, patients received venetoclax daily on a 21-day cycle, ranging from 300 mg to 1200 mg on a 3+3 design. They also received prophylaxis for tumor lysis syndrome. Patients who progressed during treatment were allowed to receive dexamethasone as well and continue the study.
Twenty-three patients (79%) discontinued treatment, 18 due to disease progression, 4 due to AEs, and 1 due to patient decision.
The ORR was 7% (n=2). Both patients achieved a CR, and both were t(11;14)-positive. One of the patients is in the 600 mg cohort and is still responding to treatment. The duration of response, thus far, is 2.1 months.
The other patient with a CR is in the 900 mg dose cohort. This patient is still responding as well, and the response duration, thus far, is 2.8 months.
Common AEs—occurring in at least 20% of patients—were diarrhea (41%), nausea (41%), fatigue (24%), vomiting (21%), asthenia (21%), and neutropenia (21%).
Grade 3/4 AEs—occurring in at least 10% of patients—were thrombocytopenia (24%), neutropenia (14%), and anemia (10%). Serious AEs—each occurring in 7% of patients—were pyrexia, malignant neoplasm progression, and cough.
Two patients had dose-limiting toxicities at the 600 mg dose of venetoclax. However, the researchers said the maximum tolerated dose was not reached, and the recommended phase 2 dose is 1200 mg. ![]()
*Information in the abstract differs from that presented at the meeting.
Biochemist Irwin Rose dies at 88

Photo courtesy of UCI
Biochemist and Nobel laureate Irwin “Ernie” Rose, PhD, has passed away at the age of 88.
Dr Rose and colleagues from Israel won the Nobel Prize in Chemistry in 2004 for their discovery of ubiquitin-mediated protein degradation.
This research has wide-ranging implications for medicine and led to the development of anticancer drugs such as bortezomib, which is approved in the US to treat multiple myeloma and mantle cell lymphoma.
According to his friends and colleagues, Dr Rose was humble, generous, and endlessly curious.
“Ernie was not interested in personal fame and was oblivious to the politics of science,” said Ann Skalka, PhD, of Fox Chase Cancer Center in Philadelphia, Pennsylvania.
“His total satisfaction came from solving intricate biochemical puzzles. Although Ernie was an intellectual leader on the project that ultimately won him the Nobel, he took no personal credit. He was rather surprised at being recognized, but all of us at Fox Chase knew that the Nobel Committee had gotten it right.”
Dr Rose was born in Brooklyn, New York, on July 16, 1926. His scientific ambitions began to take shape after he moved to Spokane, Washington, at 13. While in high school, he spent summers working at a local hospital. And this inspired him to pursue a career that involved “solving medical problems.”
Dr Rose attended Washington State College for his undergraduate work and went on to earn a doctoral degree at the University of Chicago, after a brief stint in the Navy. He spent the better part of his career as a research scientist at the Fox Chase Cancer Center.
There, during the late 1970s and early 1980s, Dr Rose helped reveal how ubiquitin molecules facilitate the breakdown of old and damaged proteins. The discovery of this process fostered a new understanding of the molecular activity present in cancers and other diseases.
For the work, Dr Rose shared the 2004 Nobel Prize in Chemistry with Avram Hershko, MD, PhD, and Aaron Ciechanover, MD, PhD, of the Israel Institute of Technology.
“Ernie had a genius for asking the right questions,” said Jonathan Chernoff, MD, PhD, of Fox Chase Cancer Center.
“In the mid-1950s, when many scientists were interested in how proteins are synthesized, Ernie became fascinated with the opposite issue—how are proteins degraded? With the collaboration of his Israeli colleagues, he cracked that problem with the discovery of the ubiquitin conjugating system.”
After retiring to Laguna Woods, California, in 1997, Dr Rose accepted a special research position with the University of California Irvine (UCI).
There, he studied the mechanisms of fumarase, an enzyme involved in the citric acid cycle, the cellular pathway by which higher organisms convert food into energy. And he quickly became a beloved colleague and mentor to students and faculty.
“[B]oth prior to and after winning the Nobel Prize, he would help any student or young postdoctoral researcher who was having a hard time with an experiment,” said Ralph Bradshaw, PhD, a former professor at UCI.
“It was a lot of fun working with him,” said James Nowick, PhD, of UCI. “He worked with his own hands, not relying on others, with old instrumentation, and was able to do literally superb science.”
“He was the quintessential scientist—perseverant, soft-spoken, and interested in science for science’s sake,” Dr Chernoff said. “We will miss him very much.”
Dr Rose died in his sleep on June 2 in Deerfield, Massachusetts. He is survived by his wife, Zelda; their sons, Howard, Frederic, and Robert; and 5 grandchildren. Dr Rose’s daughter, Sarah, died in 2005. ![]()

Photo courtesy of UCI
Biochemist and Nobel laureate Irwin “Ernie” Rose, PhD, has passed away at the age of 88.
Dr Rose and colleagues from Israel won the Nobel Prize in Chemistry in 2004 for their discovery of ubiquitin-mediated protein degradation.
This research has wide-ranging implications for medicine and led to the development of anticancer drugs such as bortezomib, which is approved in the US to treat multiple myeloma and mantle cell lymphoma.
According to his friends and colleagues, Dr Rose was humble, generous, and endlessly curious.
“Ernie was not interested in personal fame and was oblivious to the politics of science,” said Ann Skalka, PhD, of Fox Chase Cancer Center in Philadelphia, Pennsylvania.
“His total satisfaction came from solving intricate biochemical puzzles. Although Ernie was an intellectual leader on the project that ultimately won him the Nobel, he took no personal credit. He was rather surprised at being recognized, but all of us at Fox Chase knew that the Nobel Committee had gotten it right.”
Dr Rose was born in Brooklyn, New York, on July 16, 1926. His scientific ambitions began to take shape after he moved to Spokane, Washington, at 13. While in high school, he spent summers working at a local hospital. And this inspired him to pursue a career that involved “solving medical problems.”
Dr Rose attended Washington State College for his undergraduate work and went on to earn a doctoral degree at the University of Chicago, after a brief stint in the Navy. He spent the better part of his career as a research scientist at the Fox Chase Cancer Center.
There, during the late 1970s and early 1980s, Dr Rose helped reveal how ubiquitin molecules facilitate the breakdown of old and damaged proteins. The discovery of this process fostered a new understanding of the molecular activity present in cancers and other diseases.
For the work, Dr Rose shared the 2004 Nobel Prize in Chemistry with Avram Hershko, MD, PhD, and Aaron Ciechanover, MD, PhD, of the Israel Institute of Technology.
“Ernie had a genius for asking the right questions,” said Jonathan Chernoff, MD, PhD, of Fox Chase Cancer Center.
“In the mid-1950s, when many scientists were interested in how proteins are synthesized, Ernie became fascinated with the opposite issue—how are proteins degraded? With the collaboration of his Israeli colleagues, he cracked that problem with the discovery of the ubiquitin conjugating system.”
After retiring to Laguna Woods, California, in 1997, Dr Rose accepted a special research position with the University of California Irvine (UCI).
There, he studied the mechanisms of fumarase, an enzyme involved in the citric acid cycle, the cellular pathway by which higher organisms convert food into energy. And he quickly became a beloved colleague and mentor to students and faculty.
“[B]oth prior to and after winning the Nobel Prize, he would help any student or young postdoctoral researcher who was having a hard time with an experiment,” said Ralph Bradshaw, PhD, a former professor at UCI.
“It was a lot of fun working with him,” said James Nowick, PhD, of UCI. “He worked with his own hands, not relying on others, with old instrumentation, and was able to do literally superb science.”
“He was the quintessential scientist—perseverant, soft-spoken, and interested in science for science’s sake,” Dr Chernoff said. “We will miss him very much.”
Dr Rose died in his sleep on June 2 in Deerfield, Massachusetts. He is survived by his wife, Zelda; their sons, Howard, Frederic, and Robert; and 5 grandchildren. Dr Rose’s daughter, Sarah, died in 2005. ![]()

Photo courtesy of UCI
Biochemist and Nobel laureate Irwin “Ernie” Rose, PhD, has passed away at the age of 88.
Dr Rose and colleagues from Israel won the Nobel Prize in Chemistry in 2004 for their discovery of ubiquitin-mediated protein degradation.
This research has wide-ranging implications for medicine and led to the development of anticancer drugs such as bortezomib, which is approved in the US to treat multiple myeloma and mantle cell lymphoma.
According to his friends and colleagues, Dr Rose was humble, generous, and endlessly curious.
“Ernie was not interested in personal fame and was oblivious to the politics of science,” said Ann Skalka, PhD, of Fox Chase Cancer Center in Philadelphia, Pennsylvania.
“His total satisfaction came from solving intricate biochemical puzzles. Although Ernie was an intellectual leader on the project that ultimately won him the Nobel, he took no personal credit. He was rather surprised at being recognized, but all of us at Fox Chase knew that the Nobel Committee had gotten it right.”
Dr Rose was born in Brooklyn, New York, on July 16, 1926. His scientific ambitions began to take shape after he moved to Spokane, Washington, at 13. While in high school, he spent summers working at a local hospital. And this inspired him to pursue a career that involved “solving medical problems.”
Dr Rose attended Washington State College for his undergraduate work and went on to earn a doctoral degree at the University of Chicago, after a brief stint in the Navy. He spent the better part of his career as a research scientist at the Fox Chase Cancer Center.
There, during the late 1970s and early 1980s, Dr Rose helped reveal how ubiquitin molecules facilitate the breakdown of old and damaged proteins. The discovery of this process fostered a new understanding of the molecular activity present in cancers and other diseases.
For the work, Dr Rose shared the 2004 Nobel Prize in Chemistry with Avram Hershko, MD, PhD, and Aaron Ciechanover, MD, PhD, of the Israel Institute of Technology.
“Ernie had a genius for asking the right questions,” said Jonathan Chernoff, MD, PhD, of Fox Chase Cancer Center.
“In the mid-1950s, when many scientists were interested in how proteins are synthesized, Ernie became fascinated with the opposite issue—how are proteins degraded? With the collaboration of his Israeli colleagues, he cracked that problem with the discovery of the ubiquitin conjugating system.”
After retiring to Laguna Woods, California, in 1997, Dr Rose accepted a special research position with the University of California Irvine (UCI).
There, he studied the mechanisms of fumarase, an enzyme involved in the citric acid cycle, the cellular pathway by which higher organisms convert food into energy. And he quickly became a beloved colleague and mentor to students and faculty.
“[B]oth prior to and after winning the Nobel Prize, he would help any student or young postdoctoral researcher who was having a hard time with an experiment,” said Ralph Bradshaw, PhD, a former professor at UCI.
“It was a lot of fun working with him,” said James Nowick, PhD, of UCI. “He worked with his own hands, not relying on others, with old instrumentation, and was able to do literally superb science.”
“He was the quintessential scientist—perseverant, soft-spoken, and interested in science for science’s sake,” Dr Chernoff said. “We will miss him very much.”
Dr Rose died in his sleep on June 2 in Deerfield, Massachusetts. He is survived by his wife, Zelda; their sons, Howard, Frederic, and Robert; and 5 grandchildren. Dr Rose’s daughter, Sarah, died in 2005. ![]()
Single agent can treat resistant MM

© ASCO/Todd Buchanan
CHICAGO—The anti-CD38 monoclonal antibody daratumumab can be effective as a stand-alone therapy for some heavily pretreated patients with multiple myeloma (MM), results of an ongoing phase 2 trial suggest.
The study, known as SIRIUS or MMY2002, included more than 100 patients who had received 3 or more prior lines of therapy.
Roughly 30% of these subjects responded to daratumumab, with a median response duration of about 7 months.
The median progression-free survival was close to 4 months, and the estimated 1-year overall survival rate was 65%.
Serious adverse events (AEs) occurred in 30% of patients.
“These findings speak to the potential of daratumumab as an effective and tolerable option for people with multiple myeloma who have exhausted other available treatment options,” said study investigator Sagar Lonial, MD, of Emory University School of Medicine in Atlanta, Georgia.
Dr Lonial presented these findings at the 2015 ASCO Annual Meeting (abstract LBA8512). The research was funded by Janssen Research & Development, the company developing daratumumab.
In part 1 of this study, 34 patients were randomized to receive either 8 mg/kg of daratumumab once every 4 weeks or 16 mg/kg once a week for 8 weeks, then once every 2 weeks for 16 weeks and once every 4 weeks after that, until disease progression or unacceptable toxicity.
In part 2, an additional 90 patients were enrolled to receive 16 mg/kg of daratumumab on the same dosing schedule as in part 1.
Dr Lonial reported results for all patients in parts 1 and 2 who received 16 mg/kg of daratumumab. These 106 patients had received a median of 5 prior lines of therapy, including a proteasome inhibitor and an immunomodulatory drug.
According to an independent review committee, 29.2% of patients responded to daratumumab. Eighteen patients had a partial response, 10 had a very good partial response, and 3 had a stringent complete response. The median duration of response was 7.4 months.
“It is particularly noteworthy to see this level of response with a single-agent in this heavily pretreated population,” Dr Lonial said. “Ninety-seven percent of patients in this study were refractory to their last line of therapy, and 95% were double-refractory to both a [proteasome inhibitor] and an [immunomodulatory drug].”
The median overall survival has not been reached, and the estimated 1-year overall survival rate is 65%. The median progression-free survival was 3.7 months.
After a median follow up of 9.4 months, 45.2% of responders remain on therapy.
The most common AEs were fatigue (39.6%), anemia (33%), nausea (29.2%), thrombocytopenia (25.5%), neutropenia (22.6%), back pain (22.6%), and cough (20.8%).
Thirty percent of patients experienced serious AEs. And 4.7% of patients discontinued treatment due to AEs, none of which were considered drug-related.
Infusion-related reactions (IRR) were reported in 42.5% of patients and were predominantly grade 1 or 2 (4.7% grade 3; no grade 4). These occurred mainly during the first infusion.
The most common IRRs included nasal congestion (12%), throat irritation (7%), cough (6%), dyspnea (6%), chills (6%), and vomiting (6%)—all of which were treated with standard of care and slower infusion rates. ![]()

© ASCO/Todd Buchanan
CHICAGO—The anti-CD38 monoclonal antibody daratumumab can be effective as a stand-alone therapy for some heavily pretreated patients with multiple myeloma (MM), results of an ongoing phase 2 trial suggest.
The study, known as SIRIUS or MMY2002, included more than 100 patients who had received 3 or more prior lines of therapy.
Roughly 30% of these subjects responded to daratumumab, with a median response duration of about 7 months.
The median progression-free survival was close to 4 months, and the estimated 1-year overall survival rate was 65%.
Serious adverse events (AEs) occurred in 30% of patients.
“These findings speak to the potential of daratumumab as an effective and tolerable option for people with multiple myeloma who have exhausted other available treatment options,” said study investigator Sagar Lonial, MD, of Emory University School of Medicine in Atlanta, Georgia.
Dr Lonial presented these findings at the 2015 ASCO Annual Meeting (abstract LBA8512). The research was funded by Janssen Research & Development, the company developing daratumumab.
In part 1 of this study, 34 patients were randomized to receive either 8 mg/kg of daratumumab once every 4 weeks or 16 mg/kg once a week for 8 weeks, then once every 2 weeks for 16 weeks and once every 4 weeks after that, until disease progression or unacceptable toxicity.
In part 2, an additional 90 patients were enrolled to receive 16 mg/kg of daratumumab on the same dosing schedule as in part 1.
Dr Lonial reported results for all patients in parts 1 and 2 who received 16 mg/kg of daratumumab. These 106 patients had received a median of 5 prior lines of therapy, including a proteasome inhibitor and an immunomodulatory drug.
According to an independent review committee, 29.2% of patients responded to daratumumab. Eighteen patients had a partial response, 10 had a very good partial response, and 3 had a stringent complete response. The median duration of response was 7.4 months.
“It is particularly noteworthy to see this level of response with a single-agent in this heavily pretreated population,” Dr Lonial said. “Ninety-seven percent of patients in this study were refractory to their last line of therapy, and 95% were double-refractory to both a [proteasome inhibitor] and an [immunomodulatory drug].”
The median overall survival has not been reached, and the estimated 1-year overall survival rate is 65%. The median progression-free survival was 3.7 months.
After a median follow up of 9.4 months, 45.2% of responders remain on therapy.
The most common AEs were fatigue (39.6%), anemia (33%), nausea (29.2%), thrombocytopenia (25.5%), neutropenia (22.6%), back pain (22.6%), and cough (20.8%).
Thirty percent of patients experienced serious AEs. And 4.7% of patients discontinued treatment due to AEs, none of which were considered drug-related.
Infusion-related reactions (IRR) were reported in 42.5% of patients and were predominantly grade 1 or 2 (4.7% grade 3; no grade 4). These occurred mainly during the first infusion.
The most common IRRs included nasal congestion (12%), throat irritation (7%), cough (6%), dyspnea (6%), chills (6%), and vomiting (6%)—all of which were treated with standard of care and slower infusion rates. ![]()

© ASCO/Todd Buchanan
CHICAGO—The anti-CD38 monoclonal antibody daratumumab can be effective as a stand-alone therapy for some heavily pretreated patients with multiple myeloma (MM), results of an ongoing phase 2 trial suggest.
The study, known as SIRIUS or MMY2002, included more than 100 patients who had received 3 or more prior lines of therapy.
Roughly 30% of these subjects responded to daratumumab, with a median response duration of about 7 months.
The median progression-free survival was close to 4 months, and the estimated 1-year overall survival rate was 65%.
Serious adverse events (AEs) occurred in 30% of patients.
“These findings speak to the potential of daratumumab as an effective and tolerable option for people with multiple myeloma who have exhausted other available treatment options,” said study investigator Sagar Lonial, MD, of Emory University School of Medicine in Atlanta, Georgia.
Dr Lonial presented these findings at the 2015 ASCO Annual Meeting (abstract LBA8512). The research was funded by Janssen Research & Development, the company developing daratumumab.
In part 1 of this study, 34 patients were randomized to receive either 8 mg/kg of daratumumab once every 4 weeks or 16 mg/kg once a week for 8 weeks, then once every 2 weeks for 16 weeks and once every 4 weeks after that, until disease progression or unacceptable toxicity.
In part 2, an additional 90 patients were enrolled to receive 16 mg/kg of daratumumab on the same dosing schedule as in part 1.
Dr Lonial reported results for all patients in parts 1 and 2 who received 16 mg/kg of daratumumab. These 106 patients had received a median of 5 prior lines of therapy, including a proteasome inhibitor and an immunomodulatory drug.
According to an independent review committee, 29.2% of patients responded to daratumumab. Eighteen patients had a partial response, 10 had a very good partial response, and 3 had a stringent complete response. The median duration of response was 7.4 months.
“It is particularly noteworthy to see this level of response with a single-agent in this heavily pretreated population,” Dr Lonial said. “Ninety-seven percent of patients in this study were refractory to their last line of therapy, and 95% were double-refractory to both a [proteasome inhibitor] and an [immunomodulatory drug].”
The median overall survival has not been reached, and the estimated 1-year overall survival rate is 65%. The median progression-free survival was 3.7 months.
After a median follow up of 9.4 months, 45.2% of responders remain on therapy.
The most common AEs were fatigue (39.6%), anemia (33%), nausea (29.2%), thrombocytopenia (25.5%), neutropenia (22.6%), back pain (22.6%), and cough (20.8%).
Thirty percent of patients experienced serious AEs. And 4.7% of patients discontinued treatment due to AEs, none of which were considered drug-related.
Infusion-related reactions (IRR) were reported in 42.5% of patients and were predominantly grade 1 or 2 (4.7% grade 3; no grade 4). These occurred mainly during the first infusion.
The most common IRRs included nasal congestion (12%), throat irritation (7%), cough (6%), dyspnea (6%), chills (6%), and vomiting (6%)—all of which were treated with standard of care and slower infusion rates. ![]()
Cancer survivors mirror spouses’ moods

chemotherapy
Photo by Rhoda Baer
Cancer survivors’ moods are impacted—both positively and negatively—by their spouses’ moods, according to research published in Cancer Epidemiology, Biomarkers & Prevention.
In the study, cancer survivors whose spouses reported depressed moods were more likely to be depressed after about a year of follow-up, and survivors whose spouses reported better mental and physical health-related quality of life (HRQOL) were less likely to be depressed.
However, survivors’ moods did not have the same impact on their spouses.
“We were surprised that the effects of the spouses on the survivors were so much larger in this study than the effect of the survivors on their spouses,” said study author Kristin Litzelman, PhD, of the National Cancer Institute in Bethesda, Maryland. “We expected to see a more reciprocal relationship.”
Dr Litzelman and her colleagues conducted this research in an attempt to understand how cancer survivors and their families influence one another. The team hoped to identify ways to improve the healthcare both parties receive and thereby improve their health and well-being.
The researchers analyzed data from 910 cancer patients and their spouses, comparing them to 910 couples without any kind of cancer-related health problem.
The team used statistical models to assess how each spouse’s quality of life or depression at one time point was associated with his or her partner’s risk of depression around 11 months later. The researchers took into account a person’s previously reported mood, demographic characteristics, and other factors.
The results showed that, when spouses reported feeling depressed, cancer survivors were about 4 times more likely to report being depressed 11 months later (odds ratio [OR]=4.27). This association was stronger among female cancer survivors (OR=9.49) than male survivors (OR=3.98).
Cancer survivors whose spouses reported better HRQOL had a 30% decrease in depressed mood per 10-point improvement in HRQOL score. The ORs were 0.72 for mental health and 0.68 for physical health. The associations between spousal HRQOL and survivor depressed mood were similar for male and female survivors.
The researchers noted that cancer survivors’ moods did not have a significant impact on their spouses’ risk of depressed mood 11 months later.
And the team did not see mood associations in couples without any cancer-related health problems.
“This finding certainly needs to be backed up by other studies, but it highlights the importance of family well-being in cancer survivor outcomes,” Dr Litzelman said. “Our research highlights that spouses need to take care of themselves, not just for their own sake, but also for the sake of the cancer survivor.”
“Our findings also suggest that, when caring for cancer survivors, clinicians may want to assess the well-being of spousal caregivers. Future research could test whether including caregivers in the survivorship care plan might help to improve outcomes for both caregivers and for cancer survivors.” ![]()

chemotherapy
Photo by Rhoda Baer
Cancer survivors’ moods are impacted—both positively and negatively—by their spouses’ moods, according to research published in Cancer Epidemiology, Biomarkers & Prevention.
In the study, cancer survivors whose spouses reported depressed moods were more likely to be depressed after about a year of follow-up, and survivors whose spouses reported better mental and physical health-related quality of life (HRQOL) were less likely to be depressed.
However, survivors’ moods did not have the same impact on their spouses.
“We were surprised that the effects of the spouses on the survivors were so much larger in this study than the effect of the survivors on their spouses,” said study author Kristin Litzelman, PhD, of the National Cancer Institute in Bethesda, Maryland. “We expected to see a more reciprocal relationship.”
Dr Litzelman and her colleagues conducted this research in an attempt to understand how cancer survivors and their families influence one another. The team hoped to identify ways to improve the healthcare both parties receive and thereby improve their health and well-being.
The researchers analyzed data from 910 cancer patients and their spouses, comparing them to 910 couples without any kind of cancer-related health problem.
The team used statistical models to assess how each spouse’s quality of life or depression at one time point was associated with his or her partner’s risk of depression around 11 months later. The researchers took into account a person’s previously reported mood, demographic characteristics, and other factors.
The results showed that, when spouses reported feeling depressed, cancer survivors were about 4 times more likely to report being depressed 11 months later (odds ratio [OR]=4.27). This association was stronger among female cancer survivors (OR=9.49) than male survivors (OR=3.98).
Cancer survivors whose spouses reported better HRQOL had a 30% decrease in depressed mood per 10-point improvement in HRQOL score. The ORs were 0.72 for mental health and 0.68 for physical health. The associations between spousal HRQOL and survivor depressed mood were similar for male and female survivors.
The researchers noted that cancer survivors’ moods did not have a significant impact on their spouses’ risk of depressed mood 11 months later.
And the team did not see mood associations in couples without any cancer-related health problems.
“This finding certainly needs to be backed up by other studies, but it highlights the importance of family well-being in cancer survivor outcomes,” Dr Litzelman said. “Our research highlights that spouses need to take care of themselves, not just for their own sake, but also for the sake of the cancer survivor.”
“Our findings also suggest that, when caring for cancer survivors, clinicians may want to assess the well-being of spousal caregivers. Future research could test whether including caregivers in the survivorship care plan might help to improve outcomes for both caregivers and for cancer survivors.” ![]()

chemotherapy
Photo by Rhoda Baer
Cancer survivors’ moods are impacted—both positively and negatively—by their spouses’ moods, according to research published in Cancer Epidemiology, Biomarkers & Prevention.
In the study, cancer survivors whose spouses reported depressed moods were more likely to be depressed after about a year of follow-up, and survivors whose spouses reported better mental and physical health-related quality of life (HRQOL) were less likely to be depressed.
However, survivors’ moods did not have the same impact on their spouses.
“We were surprised that the effects of the spouses on the survivors were so much larger in this study than the effect of the survivors on their spouses,” said study author Kristin Litzelman, PhD, of the National Cancer Institute in Bethesda, Maryland. “We expected to see a more reciprocal relationship.”
Dr Litzelman and her colleagues conducted this research in an attempt to understand how cancer survivors and their families influence one another. The team hoped to identify ways to improve the healthcare both parties receive and thereby improve their health and well-being.
The researchers analyzed data from 910 cancer patients and their spouses, comparing them to 910 couples without any kind of cancer-related health problem.
The team used statistical models to assess how each spouse’s quality of life or depression at one time point was associated with his or her partner’s risk of depression around 11 months later. The researchers took into account a person’s previously reported mood, demographic characteristics, and other factors.
The results showed that, when spouses reported feeling depressed, cancer survivors were about 4 times more likely to report being depressed 11 months later (odds ratio [OR]=4.27). This association was stronger among female cancer survivors (OR=9.49) than male survivors (OR=3.98).
Cancer survivors whose spouses reported better HRQOL had a 30% decrease in depressed mood per 10-point improvement in HRQOL score. The ORs were 0.72 for mental health and 0.68 for physical health. The associations between spousal HRQOL and survivor depressed mood were similar for male and female survivors.
The researchers noted that cancer survivors’ moods did not have a significant impact on their spouses’ risk of depressed mood 11 months later.
And the team did not see mood associations in couples without any cancer-related health problems.
“This finding certainly needs to be backed up by other studies, but it highlights the importance of family well-being in cancer survivor outcomes,” Dr Litzelman said. “Our research highlights that spouses need to take care of themselves, not just for their own sake, but also for the sake of the cancer survivor.”
“Our findings also suggest that, when caring for cancer survivors, clinicians may want to assess the well-being of spousal caregivers. Future research could test whether including caregivers in the survivorship care plan might help to improve outcomes for both caregivers and for cancer survivors.” ![]()
VIDEO: Heavily pretreated multiple myeloma yields to novel antibody
CHICAGO – An investigational targeted therapy for multiple myeloma delayed disease progression in patients for whom as many as five prior lines of therapy had failed.
Daratumumab, an experimental antibody targeted to the CD38 receptor expressed at high levels on the surface of myeloma cells, was associated with a 29.2% overall response rate (ORR) at a median follow-up of 9.4 months, including three patients with a complete remission, Dr. Saad Zafar Usmani, a hematologist at Levine Cancer Institute-Carolinas Healthcare System in Charlotte, N.C., said in a video interview.
Dr. Usmani says that daratumumab monotherapy produced unprecedented overall responses that deepened over time in this heavily pretreated multiple myeloma patient population and that the results highlight the potential of daratumumab as a novel, well-tolerated, single-agent therapy that might be combined with existing therapies for treatment of refractory disease.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – An investigational targeted therapy for multiple myeloma delayed disease progression in patients for whom as many as five prior lines of therapy had failed.
Daratumumab, an experimental antibody targeted to the CD38 receptor expressed at high levels on the surface of myeloma cells, was associated with a 29.2% overall response rate (ORR) at a median follow-up of 9.4 months, including three patients with a complete remission, Dr. Saad Zafar Usmani, a hematologist at Levine Cancer Institute-Carolinas Healthcare System in Charlotte, N.C., said in a video interview.
Dr. Usmani says that daratumumab monotherapy produced unprecedented overall responses that deepened over time in this heavily pretreated multiple myeloma patient population and that the results highlight the potential of daratumumab as a novel, well-tolerated, single-agent therapy that might be combined with existing therapies for treatment of refractory disease.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – An investigational targeted therapy for multiple myeloma delayed disease progression in patients for whom as many as five prior lines of therapy had failed.
Daratumumab, an experimental antibody targeted to the CD38 receptor expressed at high levels on the surface of myeloma cells, was associated with a 29.2% overall response rate (ORR) at a median follow-up of 9.4 months, including three patients with a complete remission, Dr. Saad Zafar Usmani, a hematologist at Levine Cancer Institute-Carolinas Healthcare System in Charlotte, N.C., said in a video interview.
Dr. Usmani says that daratumumab monotherapy produced unprecedented overall responses that deepened over time in this heavily pretreated multiple myeloma patient population and that the results highlight the potential of daratumumab as a novel, well-tolerated, single-agent therapy that might be combined with existing therapies for treatment of refractory disease.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE 2015 ASCO ANNUAL MEETING
Novel antibody evokes responses in relapsed/refractory myeloma
CHICAGO – An investigational targeted therapy for multiple myeloma delayed disease progression in patients for whom as many as five prior lines of therapy had failed.
Daratumumab, an experimental antibody targeted to the CD38 receptor expressed at high levels on the surface of myeloma cells, was associated with a 29.2% overall response rate (ORR) at a median follow-up of 9.4 months, including three patients with a complete remission, said Dr. Saad Zafar Usmani of Levine Cancer Institute–Carolinas Healthcare System, Charlotte, N.C.
“Daratumumab monotherapy produced unprecedented overall responses that deepened over time in this heavily pretreated multiple myeloma patient population. These results highlight the potential of daratumumab as a novel, well-tolerated, single-agent therapy in various stages of myeloma treatment,” Dr. Usmani said at a briefing prior to the presentation of the data in a session at the annual meeting of the American Society of Clinical Oncology.
The results support further exploration of daratumumab in combination with currently available therapies for multiple myeloma, he added.
In the dose-selection portion of a phase II open-label, international, multicenter study, patients with multiple myeloma who had received at least three prior lines of therapy including a proteasome inhibitor and an immunomodulatory agent, or who had no treatment response to the most recent proteasome inhibitor and immunomodulator, were randomly assigned to receive either 8 mg/kg or 16 mg/kg daratumumab. After a response evaluation, the 16 mg/kg dose was chosen, and an additional 90 patients were enrolled at this dose.
As noted, the overall response rate, the primary endpoint, was 29.2%, consisting of three stringent complete responses, 10 very good partial responses, and 18 partial responses. The median duration of response was 7.4 months. The responses were consistent across all clinically relevant subgroups, Dr. Usmani said.
The median time to progression was 3.7 months. Median overall survival had not been reached at the time of the last data analysis, and the estimated 1-year overall survival rate was 65%.
At a median follow-up of 9.4 months, 14 of 31 patients with responses remained on therapy.
Patients tolerated the 16 mg/kg dose well, and none discontinued because of drug-related events.
Minor infusion site reactions (grade 1 or 2) were common during the first infusion and were managed with standard therapies, Dr. Usmani said.
“This is the first monoclonal antibody in myeloma that has shown single-agent activity,” Dr. Usmani said.
ASCO expert Dr. Merry-Jennifer Markham from the University of Florida in Gainesville, who was not involved in the study, commented that “there have been substantial treatment advances in multiple myeloma over the last decade, and daratumumab will be an important addition to the list of options. This therapy may offer a glimmer of hope for those myeloma patients who have run out of treatment options.
The study was funded by Janssen Research and Development. Dr. Usmani is on the speaker’s bureau for Janssen Oncology.
CHICAGO – An investigational targeted therapy for multiple myeloma delayed disease progression in patients for whom as many as five prior lines of therapy had failed.
Daratumumab, an experimental antibody targeted to the CD38 receptor expressed at high levels on the surface of myeloma cells, was associated with a 29.2% overall response rate (ORR) at a median follow-up of 9.4 months, including three patients with a complete remission, said Dr. Saad Zafar Usmani of Levine Cancer Institute–Carolinas Healthcare System, Charlotte, N.C.
“Daratumumab monotherapy produced unprecedented overall responses that deepened over time in this heavily pretreated multiple myeloma patient population. These results highlight the potential of daratumumab as a novel, well-tolerated, single-agent therapy in various stages of myeloma treatment,” Dr. Usmani said at a briefing prior to the presentation of the data in a session at the annual meeting of the American Society of Clinical Oncology.
The results support further exploration of daratumumab in combination with currently available therapies for multiple myeloma, he added.
In the dose-selection portion of a phase II open-label, international, multicenter study, patients with multiple myeloma who had received at least three prior lines of therapy including a proteasome inhibitor and an immunomodulatory agent, or who had no treatment response to the most recent proteasome inhibitor and immunomodulator, were randomly assigned to receive either 8 mg/kg or 16 mg/kg daratumumab. After a response evaluation, the 16 mg/kg dose was chosen, and an additional 90 patients were enrolled at this dose.
As noted, the overall response rate, the primary endpoint, was 29.2%, consisting of three stringent complete responses, 10 very good partial responses, and 18 partial responses. The median duration of response was 7.4 months. The responses were consistent across all clinically relevant subgroups, Dr. Usmani said.
The median time to progression was 3.7 months. Median overall survival had not been reached at the time of the last data analysis, and the estimated 1-year overall survival rate was 65%.
At a median follow-up of 9.4 months, 14 of 31 patients with responses remained on therapy.
Patients tolerated the 16 mg/kg dose well, and none discontinued because of drug-related events.
Minor infusion site reactions (grade 1 or 2) were common during the first infusion and were managed with standard therapies, Dr. Usmani said.
“This is the first monoclonal antibody in myeloma that has shown single-agent activity,” Dr. Usmani said.
ASCO expert Dr. Merry-Jennifer Markham from the University of Florida in Gainesville, who was not involved in the study, commented that “there have been substantial treatment advances in multiple myeloma over the last decade, and daratumumab will be an important addition to the list of options. This therapy may offer a glimmer of hope for those myeloma patients who have run out of treatment options.
The study was funded by Janssen Research and Development. Dr. Usmani is on the speaker’s bureau for Janssen Oncology.
CHICAGO – An investigational targeted therapy for multiple myeloma delayed disease progression in patients for whom as many as five prior lines of therapy had failed.
Daratumumab, an experimental antibody targeted to the CD38 receptor expressed at high levels on the surface of myeloma cells, was associated with a 29.2% overall response rate (ORR) at a median follow-up of 9.4 months, including three patients with a complete remission, said Dr. Saad Zafar Usmani of Levine Cancer Institute–Carolinas Healthcare System, Charlotte, N.C.
“Daratumumab monotherapy produced unprecedented overall responses that deepened over time in this heavily pretreated multiple myeloma patient population. These results highlight the potential of daratumumab as a novel, well-tolerated, single-agent therapy in various stages of myeloma treatment,” Dr. Usmani said at a briefing prior to the presentation of the data in a session at the annual meeting of the American Society of Clinical Oncology.
The results support further exploration of daratumumab in combination with currently available therapies for multiple myeloma, he added.
In the dose-selection portion of a phase II open-label, international, multicenter study, patients with multiple myeloma who had received at least three prior lines of therapy including a proteasome inhibitor and an immunomodulatory agent, or who had no treatment response to the most recent proteasome inhibitor and immunomodulator, were randomly assigned to receive either 8 mg/kg or 16 mg/kg daratumumab. After a response evaluation, the 16 mg/kg dose was chosen, and an additional 90 patients were enrolled at this dose.
As noted, the overall response rate, the primary endpoint, was 29.2%, consisting of three stringent complete responses, 10 very good partial responses, and 18 partial responses. The median duration of response was 7.4 months. The responses were consistent across all clinically relevant subgroups, Dr. Usmani said.
The median time to progression was 3.7 months. Median overall survival had not been reached at the time of the last data analysis, and the estimated 1-year overall survival rate was 65%.
At a median follow-up of 9.4 months, 14 of 31 patients with responses remained on therapy.
Patients tolerated the 16 mg/kg dose well, and none discontinued because of drug-related events.
Minor infusion site reactions (grade 1 or 2) were common during the first infusion and were managed with standard therapies, Dr. Usmani said.
“This is the first monoclonal antibody in myeloma that has shown single-agent activity,” Dr. Usmani said.
ASCO expert Dr. Merry-Jennifer Markham from the University of Florida in Gainesville, who was not involved in the study, commented that “there have been substantial treatment advances in multiple myeloma over the last decade, and daratumumab will be an important addition to the list of options. This therapy may offer a glimmer of hope for those myeloma patients who have run out of treatment options.
The study was funded by Janssen Research and Development. Dr. Usmani is on the speaker’s bureau for Janssen Oncology.
AT ASCO 2015
Key clinical point: The investigational monoclonal antibody daratumumab was effective in patients with heavily pretreated multiple myeloma.
Major finding: The overall response rate to daratumumab monotherapy was 29.2%.
Data source: A phase II open-label trial in 108 patients with previously treated or refractory multiple myeloma.
Disclosures: The study was funded by Janssen Research and Development. Dr. Usmani is on the speaker’s bureau for Janssen Oncology.
Hodgkin lymphoma incidence on the decline worldwide

Photo courtesy of NIH
In trying to estimate the global cancer burden, researchers found that cases of Hodgkin lymphoma (HL) have decreased worldwide over the last 2 decades.
The team studied 28 cancer types in 188 countries, and HL was the only malignancy whose incidence decreased from 1990 to 2013.
And the number of HL deaths in 2013 was comparatively low. When the researchers ranked cancers according to the number of global deaths, HL was 26th on the list of 28.
The researchers disclosed these results in JAMA Oncology.
The team collected data from cancer registries, vital records, verbal autopsy reports, and other sources to estimate the global cancer burden.
The data suggested that, in 2013, there were 14.9 million new cancer cases and 8.2 million cancer deaths worldwide. The proportion of cancer deaths as part of all deaths increased from 12% in 1990 to 15% in 2013.
The most common malignancy in men was prostate cancer, with 1.4 million cases in 2013. For women, it was breast cancer, with 1.8 million cases in 2013.
Tracheal, bronchus, and lung cancers were the leading cause of cancer death in men and women, with 1.6 million deaths in 2013.
Hematologic malignancies
Globally, the age-standardized incidence of HL per 100,000 people decreased by 34% during the time period studied. Cases of HL fell from about 103,000 in 1990 to 93,000 in 2013.
When the researchers ranked cancer types according to the number of global deaths in 2013, HL came in 26th. There were about 24,000 HL deaths in 2013—14,000 among men and 10,000 among women.
Non-Hodgkin lymphoma (NHL) came in 11th for global cancer deaths in 2013. There were about 226,000 NHL deaths—133,000 among men and 92,000 among women.
In addition, the incidence of NHL more than doubled from 1990 to 2013, rising from about 227,000 to 465,000. According to 2013 data, 1 in 103 men and 1 in 151 women developed NHL between birth and 79 years of age.
The researchers observed an increase in cases of multiple myeloma (MM) as well, from about 63,000 in 1990 to 117,000 in 2013.
In 2013, there were about 79,000 MM deaths—42,000 among men and 37,000 among women. MM ranked 19th on the list of global cancer deaths in 2013.
Leukemia ranked 9th on the list. There were about 265,000 leukemia deaths in 2013—149,000 among men and 116,000 among women.
Cases of leukemia increased from 297,000 in 1990 to 414,000 in 2013. According to 2013 data, 1 in 127 men and 1 in 203 women developed leukemia between birth and 79 years of age.
This research shows that cancer remains a major threat to people’s health around the world, said study author Christina Fitzmaurice, MD, of the University of Washington in Seattle.
“Cancer prevention, screening, and treatment programs are costly,” she noted, “and it is very important for countries to know which cancers cause the highest disease burden in order to allocate scarce resources appropriately.” ![]()

Photo courtesy of NIH
In trying to estimate the global cancer burden, researchers found that cases of Hodgkin lymphoma (HL) have decreased worldwide over the last 2 decades.
The team studied 28 cancer types in 188 countries, and HL was the only malignancy whose incidence decreased from 1990 to 2013.
And the number of HL deaths in 2013 was comparatively low. When the researchers ranked cancers according to the number of global deaths, HL was 26th on the list of 28.
The researchers disclosed these results in JAMA Oncology.
The team collected data from cancer registries, vital records, verbal autopsy reports, and other sources to estimate the global cancer burden.
The data suggested that, in 2013, there were 14.9 million new cancer cases and 8.2 million cancer deaths worldwide. The proportion of cancer deaths as part of all deaths increased from 12% in 1990 to 15% in 2013.
The most common malignancy in men was prostate cancer, with 1.4 million cases in 2013. For women, it was breast cancer, with 1.8 million cases in 2013.
Tracheal, bronchus, and lung cancers were the leading cause of cancer death in men and women, with 1.6 million deaths in 2013.
Hematologic malignancies
Globally, the age-standardized incidence of HL per 100,000 people decreased by 34% during the time period studied. Cases of HL fell from about 103,000 in 1990 to 93,000 in 2013.
When the researchers ranked cancer types according to the number of global deaths in 2013, HL came in 26th. There were about 24,000 HL deaths in 2013—14,000 among men and 10,000 among women.
Non-Hodgkin lymphoma (NHL) came in 11th for global cancer deaths in 2013. There were about 226,000 NHL deaths—133,000 among men and 92,000 among women.
In addition, the incidence of NHL more than doubled from 1990 to 2013, rising from about 227,000 to 465,000. According to 2013 data, 1 in 103 men and 1 in 151 women developed NHL between birth and 79 years of age.
The researchers observed an increase in cases of multiple myeloma (MM) as well, from about 63,000 in 1990 to 117,000 in 2013.
In 2013, there were about 79,000 MM deaths—42,000 among men and 37,000 among women. MM ranked 19th on the list of global cancer deaths in 2013.
Leukemia ranked 9th on the list. There were about 265,000 leukemia deaths in 2013—149,000 among men and 116,000 among women.
Cases of leukemia increased from 297,000 in 1990 to 414,000 in 2013. According to 2013 data, 1 in 127 men and 1 in 203 women developed leukemia between birth and 79 years of age.
This research shows that cancer remains a major threat to people’s health around the world, said study author Christina Fitzmaurice, MD, of the University of Washington in Seattle.
“Cancer prevention, screening, and treatment programs are costly,” she noted, “and it is very important for countries to know which cancers cause the highest disease burden in order to allocate scarce resources appropriately.” ![]()

Photo courtesy of NIH
In trying to estimate the global cancer burden, researchers found that cases of Hodgkin lymphoma (HL) have decreased worldwide over the last 2 decades.
The team studied 28 cancer types in 188 countries, and HL was the only malignancy whose incidence decreased from 1990 to 2013.
And the number of HL deaths in 2013 was comparatively low. When the researchers ranked cancers according to the number of global deaths, HL was 26th on the list of 28.
The researchers disclosed these results in JAMA Oncology.
The team collected data from cancer registries, vital records, verbal autopsy reports, and other sources to estimate the global cancer burden.
The data suggested that, in 2013, there were 14.9 million new cancer cases and 8.2 million cancer deaths worldwide. The proportion of cancer deaths as part of all deaths increased from 12% in 1990 to 15% in 2013.
The most common malignancy in men was prostate cancer, with 1.4 million cases in 2013. For women, it was breast cancer, with 1.8 million cases in 2013.
Tracheal, bronchus, and lung cancers were the leading cause of cancer death in men and women, with 1.6 million deaths in 2013.
Hematologic malignancies
Globally, the age-standardized incidence of HL per 100,000 people decreased by 34% during the time period studied. Cases of HL fell from about 103,000 in 1990 to 93,000 in 2013.
When the researchers ranked cancer types according to the number of global deaths in 2013, HL came in 26th. There were about 24,000 HL deaths in 2013—14,000 among men and 10,000 among women.
Non-Hodgkin lymphoma (NHL) came in 11th for global cancer deaths in 2013. There were about 226,000 NHL deaths—133,000 among men and 92,000 among women.
In addition, the incidence of NHL more than doubled from 1990 to 2013, rising from about 227,000 to 465,000. According to 2013 data, 1 in 103 men and 1 in 151 women developed NHL between birth and 79 years of age.
The researchers observed an increase in cases of multiple myeloma (MM) as well, from about 63,000 in 1990 to 117,000 in 2013.
In 2013, there were about 79,000 MM deaths—42,000 among men and 37,000 among women. MM ranked 19th on the list of global cancer deaths in 2013.
Leukemia ranked 9th on the list. There were about 265,000 leukemia deaths in 2013—149,000 among men and 116,000 among women.
Cases of leukemia increased from 297,000 in 1990 to 414,000 in 2013. According to 2013 data, 1 in 127 men and 1 in 203 women developed leukemia between birth and 79 years of age.
This research shows that cancer remains a major threat to people’s health around the world, said study author Christina Fitzmaurice, MD, of the University of Washington in Seattle.
“Cancer prevention, screening, and treatment programs are costly,” she noted, “and it is very important for countries to know which cancers cause the highest disease burden in order to allocate scarce resources appropriately.” ![]()
Herbs reduce fatigue in cancer patients

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()
Modified T cells may enhance MM treatment
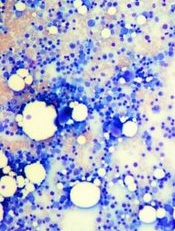
Image by Daniel E. Sabath
Modified T cells derived from patients’ bone marrow can help treat multiple myeloma (MM), according to researchers.
They modified marrow-infiltrating lymphocytes (MILs) to target MM cells and administered them to patients following chemotherapy and hematopoietic stem cell transplant (HSCT).
Seven of the 22 patients treated achieved at least a 90% reduction in disease burden, which translated to an improvement in progression-free survival.
The researchers reported these results in Science Translational Medicine.
“What we learned in this small trial is that large numbers of activated MILs can selectively target and kill myeloma cells,” said Ivan Borrello, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
Dr Borrello and his colleagues set out to test the MILs in 25 patients with newly diagnosed (n=14) or relapsed (n=11) MM. The patients’ median age was 56 years (range, 30-71), 64% were male, and 82% were white.
The median number of prior treatments was 2.13 (range, 1-6). For this trial, the patients received high-dose melphalan, autologous HSCT, and MIL therapy.
Patient outcomes
Prior to HSCT, 14 patients had a partial response (PR, 64%), 3 had a very good PR (14%), 2 had a minor response (8%), and 3 had stable disease (SD, 14%). After HSCT, 7 patients were in complete response (CR, 32%), 6 had a PR (27%), 6 had SD (27%), and 3 had progressive disease (PD, 14%).
Patients received MILs when they obtained the maximal response to the last therapy. The researchers retrieved MILs from each patient’s bone marrow and expanded them with anti-CD3/CD28 beads plus interleukin-2.
Twenty-two patients received a median of 9.5×108 MILs. Three patients relapsed before they could receive the MILs.
After MIL treatment, the overall response rate was 54%. Twenty-seven percent of patients achieved a CR, 27% had a PR, 23% had SD, and 14% had PD.
Patients who achieved at least a 90% reduction in disease burden (n=7) had superior progression-free survival compared to patients who did not respond as well to treatment (n=15).
The median progression-free survival was 25.1 months and 11.8 months, respectively (P=0.01). There was no significant difference between the groups with regard to overall survival.
The researchers said none of the patients experienced serious adverse events related to the MIL therapy.
Future directions
Dr Borrello said several US cancer centers have tested similar experimental treatments, but his team is believed to be the only one testing MILs. Other types of tumor-infiltrating cells can be used, but they are usually less plentiful in patients’ tumors and may not grow as well outside the body, he said.
“Typically, immune cells from solid tumors, called tumor-infiltrating lymphocytes, can be harvested and grown in only about 25% of patients who could potentially be eligible for the therapy,” said Kimberly Noonan, PhD, also of the Johns Hopkins University School of Medicine. “But in our clinical trial, we were able to harvest and grow MILs from all 22 patients.”
Dr Noonan said this small trial helped the researchers learn more about which patients may benefit from MIL therapy. For example, the team was able to determine how many of the MILs grown in the lab were specifically targeted to a patient’s tumor and whether they continued to target the tumor after being infused.
Additionally, the researchers found that patients with a high number of central memory cells before treatment had a better response to MIL therapy. Patients who began treatment with signs of an overactive immune response did not respond as well.
Dr Noonan said the team has used these data to guide 2 other ongoing trials of MILs. Those studies, she said, are trying to extend antitumor response and tumor specificity by combining the MILs with a cancer vaccine called GVAX and the drug lenalidomide, which stimulates T-cell responses.
All of the trials have also shed light on new ways to grow the MILs.
“In most of these trials, you see that the more cells you get, the better response you get in patients,” Dr Noonan said. “Learning how to improve cell growth may therefore improve the therapy.”
Researchers are also developing MILs to treat solid tumors such as lung, esophageal, and gastric cancers, as well as the pediatric cancers neuroblastoma and Ewing’s sarcoma. ![]()

Image by Daniel E. Sabath
Modified T cells derived from patients’ bone marrow can help treat multiple myeloma (MM), according to researchers.
They modified marrow-infiltrating lymphocytes (MILs) to target MM cells and administered them to patients following chemotherapy and hematopoietic stem cell transplant (HSCT).
Seven of the 22 patients treated achieved at least a 90% reduction in disease burden, which translated to an improvement in progression-free survival.
The researchers reported these results in Science Translational Medicine.
“What we learned in this small trial is that large numbers of activated MILs can selectively target and kill myeloma cells,” said Ivan Borrello, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
Dr Borrello and his colleagues set out to test the MILs in 25 patients with newly diagnosed (n=14) or relapsed (n=11) MM. The patients’ median age was 56 years (range, 30-71), 64% were male, and 82% were white.
The median number of prior treatments was 2.13 (range, 1-6). For this trial, the patients received high-dose melphalan, autologous HSCT, and MIL therapy.
Patient outcomes
Prior to HSCT, 14 patients had a partial response (PR, 64%), 3 had a very good PR (14%), 2 had a minor response (8%), and 3 had stable disease (SD, 14%). After HSCT, 7 patients were in complete response (CR, 32%), 6 had a PR (27%), 6 had SD (27%), and 3 had progressive disease (PD, 14%).
Patients received MILs when they obtained the maximal response to the last therapy. The researchers retrieved MILs from each patient’s bone marrow and expanded them with anti-CD3/CD28 beads plus interleukin-2.
Twenty-two patients received a median of 9.5×108 MILs. Three patients relapsed before they could receive the MILs.
After MIL treatment, the overall response rate was 54%. Twenty-seven percent of patients achieved a CR, 27% had a PR, 23% had SD, and 14% had PD.
Patients who achieved at least a 90% reduction in disease burden (n=7) had superior progression-free survival compared to patients who did not respond as well to treatment (n=15).
The median progression-free survival was 25.1 months and 11.8 months, respectively (P=0.01). There was no significant difference between the groups with regard to overall survival.
The researchers said none of the patients experienced serious adverse events related to the MIL therapy.
Future directions
Dr Borrello said several US cancer centers have tested similar experimental treatments, but his team is believed to be the only one testing MILs. Other types of tumor-infiltrating cells can be used, but they are usually less plentiful in patients’ tumors and may not grow as well outside the body, he said.
“Typically, immune cells from solid tumors, called tumor-infiltrating lymphocytes, can be harvested and grown in only about 25% of patients who could potentially be eligible for the therapy,” said Kimberly Noonan, PhD, also of the Johns Hopkins University School of Medicine. “But in our clinical trial, we were able to harvest and grow MILs from all 22 patients.”
Dr Noonan said this small trial helped the researchers learn more about which patients may benefit from MIL therapy. For example, the team was able to determine how many of the MILs grown in the lab were specifically targeted to a patient’s tumor and whether they continued to target the tumor after being infused.
Additionally, the researchers found that patients with a high number of central memory cells before treatment had a better response to MIL therapy. Patients who began treatment with signs of an overactive immune response did not respond as well.
Dr Noonan said the team has used these data to guide 2 other ongoing trials of MILs. Those studies, she said, are trying to extend antitumor response and tumor specificity by combining the MILs with a cancer vaccine called GVAX and the drug lenalidomide, which stimulates T-cell responses.
All of the trials have also shed light on new ways to grow the MILs.
“In most of these trials, you see that the more cells you get, the better response you get in patients,” Dr Noonan said. “Learning how to improve cell growth may therefore improve the therapy.”
Researchers are also developing MILs to treat solid tumors such as lung, esophageal, and gastric cancers, as well as the pediatric cancers neuroblastoma and Ewing’s sarcoma. ![]()

Image by Daniel E. Sabath
Modified T cells derived from patients’ bone marrow can help treat multiple myeloma (MM), according to researchers.
They modified marrow-infiltrating lymphocytes (MILs) to target MM cells and administered them to patients following chemotherapy and hematopoietic stem cell transplant (HSCT).
Seven of the 22 patients treated achieved at least a 90% reduction in disease burden, which translated to an improvement in progression-free survival.
The researchers reported these results in Science Translational Medicine.
“What we learned in this small trial is that large numbers of activated MILs can selectively target and kill myeloma cells,” said Ivan Borrello, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
Dr Borrello and his colleagues set out to test the MILs in 25 patients with newly diagnosed (n=14) or relapsed (n=11) MM. The patients’ median age was 56 years (range, 30-71), 64% were male, and 82% were white.
The median number of prior treatments was 2.13 (range, 1-6). For this trial, the patients received high-dose melphalan, autologous HSCT, and MIL therapy.
Patient outcomes
Prior to HSCT, 14 patients had a partial response (PR, 64%), 3 had a very good PR (14%), 2 had a minor response (8%), and 3 had stable disease (SD, 14%). After HSCT, 7 patients were in complete response (CR, 32%), 6 had a PR (27%), 6 had SD (27%), and 3 had progressive disease (PD, 14%).
Patients received MILs when they obtained the maximal response to the last therapy. The researchers retrieved MILs from each patient’s bone marrow and expanded them with anti-CD3/CD28 beads plus interleukin-2.
Twenty-two patients received a median of 9.5×108 MILs. Three patients relapsed before they could receive the MILs.
After MIL treatment, the overall response rate was 54%. Twenty-seven percent of patients achieved a CR, 27% had a PR, 23% had SD, and 14% had PD.
Patients who achieved at least a 90% reduction in disease burden (n=7) had superior progression-free survival compared to patients who did not respond as well to treatment (n=15).
The median progression-free survival was 25.1 months and 11.8 months, respectively (P=0.01). There was no significant difference between the groups with regard to overall survival.
The researchers said none of the patients experienced serious adverse events related to the MIL therapy.
Future directions
Dr Borrello said several US cancer centers have tested similar experimental treatments, but his team is believed to be the only one testing MILs. Other types of tumor-infiltrating cells can be used, but they are usually less plentiful in patients’ tumors and may not grow as well outside the body, he said.
“Typically, immune cells from solid tumors, called tumor-infiltrating lymphocytes, can be harvested and grown in only about 25% of patients who could potentially be eligible for the therapy,” said Kimberly Noonan, PhD, also of the Johns Hopkins University School of Medicine. “But in our clinical trial, we were able to harvest and grow MILs from all 22 patients.”
Dr Noonan said this small trial helped the researchers learn more about which patients may benefit from MIL therapy. For example, the team was able to determine how many of the MILs grown in the lab were specifically targeted to a patient’s tumor and whether they continued to target the tumor after being infused.
Additionally, the researchers found that patients with a high number of central memory cells before treatment had a better response to MIL therapy. Patients who began treatment with signs of an overactive immune response did not respond as well.
Dr Noonan said the team has used these data to guide 2 other ongoing trials of MILs. Those studies, she said, are trying to extend antitumor response and tumor specificity by combining the MILs with a cancer vaccine called GVAX and the drug lenalidomide, which stimulates T-cell responses.
All of the trials have also shed light on new ways to grow the MILs.
“In most of these trials, you see that the more cells you get, the better response you get in patients,” Dr Noonan said. “Learning how to improve cell growth may therefore improve the therapy.”
Researchers are also developing MILs to treat solid tumors such as lung, esophageal, and gastric cancers, as well as the pediatric cancers neuroblastoma and Ewing’s sarcoma.
Nanotherapy appears effective against MM

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work.

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work.

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work.



