User login
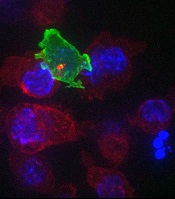
CTLs captured on video destroying cancer cells
cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()
Adding mAb to treatment extends PFS in MM

Photo by Linda Bartlett
The monoclonal antibody (mAb) elotuzumab may be a useful addition to the multiple myeloma (MM) arsenal, according to investigators involved in the phase 3 ELOQUENT-2 trial.
The trial showed that adding elotuzumab to treatment with lenalidomide and dexamethasone extended progression-free survival (PFS) in relapsed MM patients by about 5 months, on average, when compared to treatment with just lenalidomide and dexamethasone.
“It appears that, for patients with relapsed multiple myeloma who would otherwise be offered lenalidomide and dexamethasone, addition of this new targeted drug makes the outcomes even better,” said study investigator Sagar Lonial, MD, of Emory University in Atlanta, Georgia.
Dr Lonial presented data from ELOQUENT-2 at a presscast in advance of the 2015 ASCO Annual Meeting. Full data from the study will be presented at the meeting on June 2 as abstract 8508.
The study was funded by Bristol-Myers Squibb and AbbVie, the companies developing elotuzumab.
Dr Lonial explained that elotuzumab attaches to the cell surface protein SLAMF7, which is found on MM cells and natural killer (NK) cells. Scientists believe that elotuzumab mounts a 2-pronged attack on MM by targeting myeloma cells directly and by enhancing NK cells’ ability to kill myeloma cells.
In ELOQUENT-2, 646 patients with recurrent MM were randomized to receive elotuzumab plus lenalidomide and dexamethasone or only lenalidomide and dexamethasone (control).
The patients’ median age was 66. They had failed 1 to 3 prior treatments, and 35% of them were refractory to their last therapy. Thirty-two percent of patients had del(17p), and 9% had t[4;14].
At a median follow-up of 24 months, elotuzumab had reduced the risk of MM progression and death by 30%. Patients in the elotuzumab arm had significantly longer PFS than patients in the control arm—a median of 19.4 months and 14.9 months, respectively (P=0.0004).
The 1-year PFS was 68% in the elotuzumab arm and 57% in the control arm. The 2-year PFS was 41% and 27%, respectively. Dr Lonial pointed out that, unlike some other therapies, elotuzumab continued to improve PFS over time.
“[T]he idea of the maintenance of benefit over time really speaks to the power of an immune-based approach when we treat cancer,” he said.
“Patients who received elotuzumab had a longer duration of remission [and] a higher overall response rate, and this improvement in clinical parameters occurred without a significant increase in adverse events or toxicity. In fact, there was no reduction in quality of life for [patients in the] 3-drug arm.”
Mild infusion reactions occurred after the first few doses in 10% of patients who received elotuzumab. Most of these reactions were grade 1 or 2.
Common grade 3-4 adverse events (occurring in ≥ 15% of patients) in both the elotuzumab and control arms were neutropenia (25% and 33%, respectively) and anemia (15% and 16%, respectively).
In all, 210 patients died, 94 in the elotuzumab arm and 116 in the control arm.
“Based on this randomized, phase 3 trial, we hope that we will soon have a new treatment option for patients with relapsed or refractory myeloma . . . ,” Dr Lonial said.
The US Food and Drug Administration has already granted elotuzumab breakthrough therapy designation to treat MM patients who have received at least 1 prior therapy. ![]()

Photo by Linda Bartlett
The monoclonal antibody (mAb) elotuzumab may be a useful addition to the multiple myeloma (MM) arsenal, according to investigators involved in the phase 3 ELOQUENT-2 trial.
The trial showed that adding elotuzumab to treatment with lenalidomide and dexamethasone extended progression-free survival (PFS) in relapsed MM patients by about 5 months, on average, when compared to treatment with just lenalidomide and dexamethasone.
“It appears that, for patients with relapsed multiple myeloma who would otherwise be offered lenalidomide and dexamethasone, addition of this new targeted drug makes the outcomes even better,” said study investigator Sagar Lonial, MD, of Emory University in Atlanta, Georgia.
Dr Lonial presented data from ELOQUENT-2 at a presscast in advance of the 2015 ASCO Annual Meeting. Full data from the study will be presented at the meeting on June 2 as abstract 8508.
The study was funded by Bristol-Myers Squibb and AbbVie, the companies developing elotuzumab.
Dr Lonial explained that elotuzumab attaches to the cell surface protein SLAMF7, which is found on MM cells and natural killer (NK) cells. Scientists believe that elotuzumab mounts a 2-pronged attack on MM by targeting myeloma cells directly and by enhancing NK cells’ ability to kill myeloma cells.
In ELOQUENT-2, 646 patients with recurrent MM were randomized to receive elotuzumab plus lenalidomide and dexamethasone or only lenalidomide and dexamethasone (control).
The patients’ median age was 66. They had failed 1 to 3 prior treatments, and 35% of them were refractory to their last therapy. Thirty-two percent of patients had del(17p), and 9% had t[4;14].
At a median follow-up of 24 months, elotuzumab had reduced the risk of MM progression and death by 30%. Patients in the elotuzumab arm had significantly longer PFS than patients in the control arm—a median of 19.4 months and 14.9 months, respectively (P=0.0004).
The 1-year PFS was 68% in the elotuzumab arm and 57% in the control arm. The 2-year PFS was 41% and 27%, respectively. Dr Lonial pointed out that, unlike some other therapies, elotuzumab continued to improve PFS over time.
“[T]he idea of the maintenance of benefit over time really speaks to the power of an immune-based approach when we treat cancer,” he said.
“Patients who received elotuzumab had a longer duration of remission [and] a higher overall response rate, and this improvement in clinical parameters occurred without a significant increase in adverse events or toxicity. In fact, there was no reduction in quality of life for [patients in the] 3-drug arm.”
Mild infusion reactions occurred after the first few doses in 10% of patients who received elotuzumab. Most of these reactions were grade 1 or 2.
Common grade 3-4 adverse events (occurring in ≥ 15% of patients) in both the elotuzumab and control arms were neutropenia (25% and 33%, respectively) and anemia (15% and 16%, respectively).
In all, 210 patients died, 94 in the elotuzumab arm and 116 in the control arm.
“Based on this randomized, phase 3 trial, we hope that we will soon have a new treatment option for patients with relapsed or refractory myeloma . . . ,” Dr Lonial said.
The US Food and Drug Administration has already granted elotuzumab breakthrough therapy designation to treat MM patients who have received at least 1 prior therapy. ![]()

Photo by Linda Bartlett
The monoclonal antibody (mAb) elotuzumab may be a useful addition to the multiple myeloma (MM) arsenal, according to investigators involved in the phase 3 ELOQUENT-2 trial.
The trial showed that adding elotuzumab to treatment with lenalidomide and dexamethasone extended progression-free survival (PFS) in relapsed MM patients by about 5 months, on average, when compared to treatment with just lenalidomide and dexamethasone.
“It appears that, for patients with relapsed multiple myeloma who would otherwise be offered lenalidomide and dexamethasone, addition of this new targeted drug makes the outcomes even better,” said study investigator Sagar Lonial, MD, of Emory University in Atlanta, Georgia.
Dr Lonial presented data from ELOQUENT-2 at a presscast in advance of the 2015 ASCO Annual Meeting. Full data from the study will be presented at the meeting on June 2 as abstract 8508.
The study was funded by Bristol-Myers Squibb and AbbVie, the companies developing elotuzumab.
Dr Lonial explained that elotuzumab attaches to the cell surface protein SLAMF7, which is found on MM cells and natural killer (NK) cells. Scientists believe that elotuzumab mounts a 2-pronged attack on MM by targeting myeloma cells directly and by enhancing NK cells’ ability to kill myeloma cells.
In ELOQUENT-2, 646 patients with recurrent MM were randomized to receive elotuzumab plus lenalidomide and dexamethasone or only lenalidomide and dexamethasone (control).
The patients’ median age was 66. They had failed 1 to 3 prior treatments, and 35% of them were refractory to their last therapy. Thirty-two percent of patients had del(17p), and 9% had t[4;14].
At a median follow-up of 24 months, elotuzumab had reduced the risk of MM progression and death by 30%. Patients in the elotuzumab arm had significantly longer PFS than patients in the control arm—a median of 19.4 months and 14.9 months, respectively (P=0.0004).
The 1-year PFS was 68% in the elotuzumab arm and 57% in the control arm. The 2-year PFS was 41% and 27%, respectively. Dr Lonial pointed out that, unlike some other therapies, elotuzumab continued to improve PFS over time.
“[T]he idea of the maintenance of benefit over time really speaks to the power of an immune-based approach when we treat cancer,” he said.
“Patients who received elotuzumab had a longer duration of remission [and] a higher overall response rate, and this improvement in clinical parameters occurred without a significant increase in adverse events or toxicity. In fact, there was no reduction in quality of life for [patients in the] 3-drug arm.”
Mild infusion reactions occurred after the first few doses in 10% of patients who received elotuzumab. Most of these reactions were grade 1 or 2.
Common grade 3-4 adverse events (occurring in ≥ 15% of patients) in both the elotuzumab and control arms were neutropenia (25% and 33%, respectively) and anemia (15% and 16%, respectively).
In all, 210 patients died, 94 in the elotuzumab arm and 116 in the control arm.
“Based on this randomized, phase 3 trial, we hope that we will soon have a new treatment option for patients with relapsed or refractory myeloma . . . ,” Dr Lonial said.
The US Food and Drug Administration has already granted elotuzumab breakthrough therapy designation to treat MM patients who have received at least 1 prior therapy. ![]()
Preventing Skeletal-Related Events
Supplementation with calcium and vitamin D can be effective ways to prevent skeletal or teeth-related issues during treatment for bone metastasis or multiple myeloma, according to Sheetal Malhotra, MD, of the James J. Peters VAMC in the Bronx, New York.
“Data suggest that if you optimize vitamin D levels when you are giving patients bisphosphonates it can further improve response to bisphosphonate therapy,” Malhotra said. “Supplementation should be given to these patients who are on treatment with bisphosphonates unless there is some contraindication.”
Supplementation with calcium and vitamin D can be effective ways to prevent skeletal or teeth-related issues during treatment for bone metastasis or multiple myeloma, according to Sheetal Malhotra, MD, of the James J. Peters VAMC in the Bronx, New York.
“Data suggest that if you optimize vitamin D levels when you are giving patients bisphosphonates it can further improve response to bisphosphonate therapy,” Malhotra said. “Supplementation should be given to these patients who are on treatment with bisphosphonates unless there is some contraindication.”
Supplementation with calcium and vitamin D can be effective ways to prevent skeletal or teeth-related issues during treatment for bone metastasis or multiple myeloma, according to Sheetal Malhotra, MD, of the James J. Peters VAMC in the Bronx, New York.
“Data suggest that if you optimize vitamin D levels when you are giving patients bisphosphonates it can further improve response to bisphosphonate therapy,” Malhotra said. “Supplementation should be given to these patients who are on treatment with bisphosphonates unless there is some contraindication.”
Elotuzumab plus len-dex extends remissions of multiple myeloma
Adding the investigational monoclonal antibody elotuzumab to a standard regimen for early relapsed/refractory multiple myeloma extended the duration of remissions by a mean of 5 months.
The combination of elotuzumab (elo), lenalidomide (len), and dexamethasone (dex) was associated with a 30% reduction in the risk of disease progression or death compared with len/dex alone, and the benefit of the drug was also seen, although to a lesser degree, in patients with high-risk disease mutations, reported Dr. Sagar Lonial of the Winship Cancer Institute of Emory University, Atlanta.
“We were certainly excited about the fact that there is such a big difference in progression-free survival between the two arms,” he said at a briefing on studies to be presented at the annual meeting of the American Society of Clinical Oncology.
“Patients who received elotuzumab had a longer duration of remission and had a higher overall response rate, and this improvement in clinical parameters occurred without a significant increase in adverse events or toxicity,” he added.
Dr. Julie M. Vose, ASCO president-elect, who comoderated the briefing, said that “although we have had monoclonal antibodies in a number of other malignancies for years, this truly is the first one for myeloma.”
Dr. Lonial reported interim results from ELOQUENT-2, a phase III study comparing the efficacy and safety of the elo/len/dex combination vs. len/dex alone in 646 patients with relapsed/refractory multiple myeloma who had received one, two, or three prior lines of therapy and whose disease was not resistant to len/dex.
The patients were randomized to receive either oral len/dex on a standard schedule, or oral lenalidomide plus oral dexamethasone on weeks when there was no elotuzumab infusion, with both oral and intravenous dexamethasone delivered on weeks when the antibody was delivered by solutional infusion. The regimens were delivered in 28-day cycles until disease progression or unacceptable toxicities occurred.
At 24 months median follow-up, the mean progression-free survival (PFS) for patients on elo/len/dex was 19.4 months, compared with 14.9 months for len/dex alone (P = .0004). One-year PFS rates were 68% for elo/len/dex, compared with 57% for len/dex. Two-year PFS rates were 41% vs. 27%, respectively.
The overall response rate with the antibody-containing combination was 79% compared with 66% for len/dex alone (P = .0002). Dr. Lonial noted that the antibody-containing combination was effective both in patients with average risk, and in those with the high risk cytogenetic abnormalities, including the 17p deletion and the t(4;14) chromosomal translocation, although the response was not as robust in these patients.
A total of 210 patients died during follow-up, 94 who had been treated with elo/len/dex, and 116 who had been treated with len/dex.
Grade 3 or 4 adverse events occurring in 15% or more of patients included neutropenia in 25% of patients on elo/len/dex, and 33% of patients on len/dex, and anemia, which occurred in 15% and 16% of patients, respectively. In all, 10% of patients in the elotuzumab arm had mild infusion reactions after the first few doses.
Based on data from earlier trials, the Food and Drug Administration has granted elotuzumab breakthrough designation for treatment of multiple myeloma that has relapsed or is refractory to at least one prior line of therapy. Breakthrough status entitles the manufacturer to an expedited review of the drug.
Adding the investigational monoclonal antibody elotuzumab to a standard regimen for early relapsed/refractory multiple myeloma extended the duration of remissions by a mean of 5 months.
The combination of elotuzumab (elo), lenalidomide (len), and dexamethasone (dex) was associated with a 30% reduction in the risk of disease progression or death compared with len/dex alone, and the benefit of the drug was also seen, although to a lesser degree, in patients with high-risk disease mutations, reported Dr. Sagar Lonial of the Winship Cancer Institute of Emory University, Atlanta.
“We were certainly excited about the fact that there is such a big difference in progression-free survival between the two arms,” he said at a briefing on studies to be presented at the annual meeting of the American Society of Clinical Oncology.
“Patients who received elotuzumab had a longer duration of remission and had a higher overall response rate, and this improvement in clinical parameters occurred without a significant increase in adverse events or toxicity,” he added.
Dr. Julie M. Vose, ASCO president-elect, who comoderated the briefing, said that “although we have had monoclonal antibodies in a number of other malignancies for years, this truly is the first one for myeloma.”
Dr. Lonial reported interim results from ELOQUENT-2, a phase III study comparing the efficacy and safety of the elo/len/dex combination vs. len/dex alone in 646 patients with relapsed/refractory multiple myeloma who had received one, two, or three prior lines of therapy and whose disease was not resistant to len/dex.
The patients were randomized to receive either oral len/dex on a standard schedule, or oral lenalidomide plus oral dexamethasone on weeks when there was no elotuzumab infusion, with both oral and intravenous dexamethasone delivered on weeks when the antibody was delivered by solutional infusion. The regimens were delivered in 28-day cycles until disease progression or unacceptable toxicities occurred.
At 24 months median follow-up, the mean progression-free survival (PFS) for patients on elo/len/dex was 19.4 months, compared with 14.9 months for len/dex alone (P = .0004). One-year PFS rates were 68% for elo/len/dex, compared with 57% for len/dex. Two-year PFS rates were 41% vs. 27%, respectively.
The overall response rate with the antibody-containing combination was 79% compared with 66% for len/dex alone (P = .0002). Dr. Lonial noted that the antibody-containing combination was effective both in patients with average risk, and in those with the high risk cytogenetic abnormalities, including the 17p deletion and the t(4;14) chromosomal translocation, although the response was not as robust in these patients.
A total of 210 patients died during follow-up, 94 who had been treated with elo/len/dex, and 116 who had been treated with len/dex.
Grade 3 or 4 adverse events occurring in 15% or more of patients included neutropenia in 25% of patients on elo/len/dex, and 33% of patients on len/dex, and anemia, which occurred in 15% and 16% of patients, respectively. In all, 10% of patients in the elotuzumab arm had mild infusion reactions after the first few doses.
Based on data from earlier trials, the Food and Drug Administration has granted elotuzumab breakthrough designation for treatment of multiple myeloma that has relapsed or is refractory to at least one prior line of therapy. Breakthrough status entitles the manufacturer to an expedited review of the drug.
Adding the investigational monoclonal antibody elotuzumab to a standard regimen for early relapsed/refractory multiple myeloma extended the duration of remissions by a mean of 5 months.
The combination of elotuzumab (elo), lenalidomide (len), and dexamethasone (dex) was associated with a 30% reduction in the risk of disease progression or death compared with len/dex alone, and the benefit of the drug was also seen, although to a lesser degree, in patients with high-risk disease mutations, reported Dr. Sagar Lonial of the Winship Cancer Institute of Emory University, Atlanta.
“We were certainly excited about the fact that there is such a big difference in progression-free survival between the two arms,” he said at a briefing on studies to be presented at the annual meeting of the American Society of Clinical Oncology.
“Patients who received elotuzumab had a longer duration of remission and had a higher overall response rate, and this improvement in clinical parameters occurred without a significant increase in adverse events or toxicity,” he added.
Dr. Julie M. Vose, ASCO president-elect, who comoderated the briefing, said that “although we have had monoclonal antibodies in a number of other malignancies for years, this truly is the first one for myeloma.”
Dr. Lonial reported interim results from ELOQUENT-2, a phase III study comparing the efficacy and safety of the elo/len/dex combination vs. len/dex alone in 646 patients with relapsed/refractory multiple myeloma who had received one, two, or three prior lines of therapy and whose disease was not resistant to len/dex.
The patients were randomized to receive either oral len/dex on a standard schedule, or oral lenalidomide plus oral dexamethasone on weeks when there was no elotuzumab infusion, with both oral and intravenous dexamethasone delivered on weeks when the antibody was delivered by solutional infusion. The regimens were delivered in 28-day cycles until disease progression or unacceptable toxicities occurred.
At 24 months median follow-up, the mean progression-free survival (PFS) for patients on elo/len/dex was 19.4 months, compared with 14.9 months for len/dex alone (P = .0004). One-year PFS rates were 68% for elo/len/dex, compared with 57% for len/dex. Two-year PFS rates were 41% vs. 27%, respectively.
The overall response rate with the antibody-containing combination was 79% compared with 66% for len/dex alone (P = .0002). Dr. Lonial noted that the antibody-containing combination was effective both in patients with average risk, and in those with the high risk cytogenetic abnormalities, including the 17p deletion and the t(4;14) chromosomal translocation, although the response was not as robust in these patients.
A total of 210 patients died during follow-up, 94 who had been treated with elo/len/dex, and 116 who had been treated with len/dex.
Grade 3 or 4 adverse events occurring in 15% or more of patients included neutropenia in 25% of patients on elo/len/dex, and 33% of patients on len/dex, and anemia, which occurred in 15% and 16% of patients, respectively. In all, 10% of patients in the elotuzumab arm had mild infusion reactions after the first few doses.
Based on data from earlier trials, the Food and Drug Administration has granted elotuzumab breakthrough designation for treatment of multiple myeloma that has relapsed or is refractory to at least one prior line of therapy. Breakthrough status entitles the manufacturer to an expedited review of the drug.
Key clinical point: The novel antibody elotuzumab extended multiple myeloma remissions when added to standard therapy.
Major finding: Mean progression-free survival at 24-months median follow-up was 19.4 months for elotuzumab plus lenalidomide/dexamethasone, vs. 14.9 months for lenalidomide/dexamethasone alone.
Data source: Interim analysis of a phase III randomized trial of 646 patients with relapsed/refractory multiple myeloma.
Disclosures: Bristol-Myers Squibb and AbbVie sponsored the trial. Dr. Lonial disclosed consulting/advisory roles with BMS and other companies. His coauthors disclosed multiple industry relationships.
Familial factors linked to child’s risk of blood cancer

A new study has linked a father’s age at his child’s birth to the risk that the child will develop a hematologic malignancy as an adult, but this risk only proved significant among children without siblings.
Only-children whose fathers were 35 or older at the child’s birth were significantly more likely to develop hematologic malignancies than only-children with fathers who were younger than 25 at the child’s birth.
There was no association between these cancers and a mother’s age, either among only-children or those with siblings.
A previous study of more than 100,000 women also showed an association between paternal—but not maternal—age at a child’s birth and the risk of hematologic malignancy.
To further investigate the association, Lauren Teras, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues analyzed data from women and men enrolled in the American Cancer Society Cancer Prevention Study-II Nutrition Cohort.
The team reported their findings in the American Journal of Epidemiology.
Among the 138,003 participants, there were 2532 cases of hematologic malignancies diagnosed between 1992 and 2009.
Subjects’ mothers tended to be younger at their birth than fathers, with median ages of 27 and 31, respectively. Almost a third of the fathers were 35 or older when a subject was born, compared with 17% of the mothers.
In the categorical analysis, the researchers found a positive association between older paternal age at a subject’s birth and the risk of hematologic malignancies in male, but not female, subjects. The hazard ratio (HR) was 1.35 for male subjects with fathers aged 35 and older compared to those whose fathers were younger than 25.
On the other hand, when paternal age was modeled as a continuous variable, there was no association with the risk of hematologic malignancy for males or females. Likewise, there was no association between maternal age at a subject’s birth and the risk of hematologic malignancy in male or female subjects.
However, among subjects without siblings, there was a significant, positive association with paternal age and the risk of hematologic malignancy (P=0.002).
When the researchers separated only-children by sex, they found a suggestive positive association between paternal age and hematologic malignancy for females (HR=1.40) and a significant association for males (HR=1.84). However, the linear spline was significant for males (P=0.01) and females (P=0.04).
There was no association between paternal age at a subject’s birth and the risk of hematologic malignancy among subjects with at least 1 sibling (HR=1.06).
The researchers said the fact that the association between paternal age and malignancy was significant in subjects with no siblings suggests it may be related to the “hygiene hypothesis”—the idea that exposure to mild infections in childhood, which might be more numerous with more siblings, are important to immune system development and may reduce the risk of immune-related diseases.
It is possible that the combination of having an older father and no siblings may promote cell proliferation in those individuals with an underdeveloped immune system and, as such, favors the development of cancers related to the immune system, the team said.
They added that this study suggests a need for further research to better understand the association between paternal age at a child’s birth and hematologic malignancies.
“The lifetime risk of these cancers is fairly low—about 1 in 20 men and women will be diagnosed with lymphoma, leukemia, or myeloma at some point during their lifetime—so people born to older fathers should not be alarmed,” Dr Teras said.
“Still, the study does highlight the need for more research to confirm these findings and to clarify the biologic underpinning for this association, given the growing number of children born to older fathers in the United States and worldwide.” ![]()

A new study has linked a father’s age at his child’s birth to the risk that the child will develop a hematologic malignancy as an adult, but this risk only proved significant among children without siblings.
Only-children whose fathers were 35 or older at the child’s birth were significantly more likely to develop hematologic malignancies than only-children with fathers who were younger than 25 at the child’s birth.
There was no association between these cancers and a mother’s age, either among only-children or those with siblings.
A previous study of more than 100,000 women also showed an association between paternal—but not maternal—age at a child’s birth and the risk of hematologic malignancy.
To further investigate the association, Lauren Teras, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues analyzed data from women and men enrolled in the American Cancer Society Cancer Prevention Study-II Nutrition Cohort.
The team reported their findings in the American Journal of Epidemiology.
Among the 138,003 participants, there were 2532 cases of hematologic malignancies diagnosed between 1992 and 2009.
Subjects’ mothers tended to be younger at their birth than fathers, with median ages of 27 and 31, respectively. Almost a third of the fathers were 35 or older when a subject was born, compared with 17% of the mothers.
In the categorical analysis, the researchers found a positive association between older paternal age at a subject’s birth and the risk of hematologic malignancies in male, but not female, subjects. The hazard ratio (HR) was 1.35 for male subjects with fathers aged 35 and older compared to those whose fathers were younger than 25.
On the other hand, when paternal age was modeled as a continuous variable, there was no association with the risk of hematologic malignancy for males or females. Likewise, there was no association between maternal age at a subject’s birth and the risk of hematologic malignancy in male or female subjects.
However, among subjects without siblings, there was a significant, positive association with paternal age and the risk of hematologic malignancy (P=0.002).
When the researchers separated only-children by sex, they found a suggestive positive association between paternal age and hematologic malignancy for females (HR=1.40) and a significant association for males (HR=1.84). However, the linear spline was significant for males (P=0.01) and females (P=0.04).
There was no association between paternal age at a subject’s birth and the risk of hematologic malignancy among subjects with at least 1 sibling (HR=1.06).
The researchers said the fact that the association between paternal age and malignancy was significant in subjects with no siblings suggests it may be related to the “hygiene hypothesis”—the idea that exposure to mild infections in childhood, which might be more numerous with more siblings, are important to immune system development and may reduce the risk of immune-related diseases.
It is possible that the combination of having an older father and no siblings may promote cell proliferation in those individuals with an underdeveloped immune system and, as such, favors the development of cancers related to the immune system, the team said.
They added that this study suggests a need for further research to better understand the association between paternal age at a child’s birth and hematologic malignancies.
“The lifetime risk of these cancers is fairly low—about 1 in 20 men and women will be diagnosed with lymphoma, leukemia, or myeloma at some point during their lifetime—so people born to older fathers should not be alarmed,” Dr Teras said.
“Still, the study does highlight the need for more research to confirm these findings and to clarify the biologic underpinning for this association, given the growing number of children born to older fathers in the United States and worldwide.” ![]()

A new study has linked a father’s age at his child’s birth to the risk that the child will develop a hematologic malignancy as an adult, but this risk only proved significant among children without siblings.
Only-children whose fathers were 35 or older at the child’s birth were significantly more likely to develop hematologic malignancies than only-children with fathers who were younger than 25 at the child’s birth.
There was no association between these cancers and a mother’s age, either among only-children or those with siblings.
A previous study of more than 100,000 women also showed an association between paternal—but not maternal—age at a child’s birth and the risk of hematologic malignancy.
To further investigate the association, Lauren Teras, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues analyzed data from women and men enrolled in the American Cancer Society Cancer Prevention Study-II Nutrition Cohort.
The team reported their findings in the American Journal of Epidemiology.
Among the 138,003 participants, there were 2532 cases of hematologic malignancies diagnosed between 1992 and 2009.
Subjects’ mothers tended to be younger at their birth than fathers, with median ages of 27 and 31, respectively. Almost a third of the fathers were 35 or older when a subject was born, compared with 17% of the mothers.
In the categorical analysis, the researchers found a positive association between older paternal age at a subject’s birth and the risk of hematologic malignancies in male, but not female, subjects. The hazard ratio (HR) was 1.35 for male subjects with fathers aged 35 and older compared to those whose fathers were younger than 25.
On the other hand, when paternal age was modeled as a continuous variable, there was no association with the risk of hematologic malignancy for males or females. Likewise, there was no association between maternal age at a subject’s birth and the risk of hematologic malignancy in male or female subjects.
However, among subjects without siblings, there was a significant, positive association with paternal age and the risk of hematologic malignancy (P=0.002).
When the researchers separated only-children by sex, they found a suggestive positive association between paternal age and hematologic malignancy for females (HR=1.40) and a significant association for males (HR=1.84). However, the linear spline was significant for males (P=0.01) and females (P=0.04).
There was no association between paternal age at a subject’s birth and the risk of hematologic malignancy among subjects with at least 1 sibling (HR=1.06).
The researchers said the fact that the association between paternal age and malignancy was significant in subjects with no siblings suggests it may be related to the “hygiene hypothesis”—the idea that exposure to mild infections in childhood, which might be more numerous with more siblings, are important to immune system development and may reduce the risk of immune-related diseases.
It is possible that the combination of having an older father and no siblings may promote cell proliferation in those individuals with an underdeveloped immune system and, as such, favors the development of cancers related to the immune system, the team said.
They added that this study suggests a need for further research to better understand the association between paternal age at a child’s birth and hematologic malignancies.
“The lifetime risk of these cancers is fairly low—about 1 in 20 men and women will be diagnosed with lymphoma, leukemia, or myeloma at some point during their lifetime—so people born to older fathers should not be alarmed,” Dr Teras said.
“Still, the study does highlight the need for more research to confirm these findings and to clarify the biologic underpinning for this association, given the growing number of children born to older fathers in the United States and worldwide.” ![]()
Explaining obesity in cancer survivors

Researchers have identified several factors that may influence the risk of obesity in childhood cancer survivors.
Previous research showed that obesity rates are elevated in childhood cancer survivors who were exposed to cranial radiation.
But the new study has shown that other types of treatment, a patient’s age, and certain genetic variants are associated with obesity in this population.
Carmen Wilson, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues reported these findings in Cancer.
The researchers evaluated 1,996 childhood cancer survivors treated at St. Jude. The patients’ median age at diagnosis was 7.2 years (range, 0.1-24.8), and their median age at follow-up was 32.4 years (range, 18.9-63.8).
At the time of evaluation, 645 patients (32.3%) were of normal weight, 71 (3.6%) were underweight, 556 (27.9%) were overweight, and 723 (36.2%) were obese.
The prevalence of obesity was highest in male leukemia survivors (42.5%) and females who survived neuroblastoma (43.6%), followed closely by those who survived leukemia (43.1%).
Multivariable analyses showed that 3 factors were independently associated with an increased risk of obesity: older age at the time of evaluation (≥30 years vs <30 years; P<0.001), undergoing cranial radiation (P<0.001), and receiving glucocorticoids (P=0.004).
On the other hand, receiving chest, abdominal, or pelvic radiation was associated with a decreased risk of obesity (P<0.001).
The researchers also identified 166 single nucleotide polymorphisms that were associated with obesity among cancer survivors who had received cranial radiation. The strongest association was in variants of genes involved in neuron growth, repair, and connectivity.
Among survivors who did not receive cranial radiation, only 1 single nucleotide polymorphism—rs12073359, located on chromosome 1—was associated with an increased risk of obesity.
Dr Wilson said these findings might help us identify the childhood cancer survivors who are most likely to become obese. The results may also provide a foundation for future research efforts aimed at characterizing molecular pathways involved in the link between childhood cancer treatment and obesity. ![]()

Researchers have identified several factors that may influence the risk of obesity in childhood cancer survivors.
Previous research showed that obesity rates are elevated in childhood cancer survivors who were exposed to cranial radiation.
But the new study has shown that other types of treatment, a patient’s age, and certain genetic variants are associated with obesity in this population.
Carmen Wilson, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues reported these findings in Cancer.
The researchers evaluated 1,996 childhood cancer survivors treated at St. Jude. The patients’ median age at diagnosis was 7.2 years (range, 0.1-24.8), and their median age at follow-up was 32.4 years (range, 18.9-63.8).
At the time of evaluation, 645 patients (32.3%) were of normal weight, 71 (3.6%) were underweight, 556 (27.9%) were overweight, and 723 (36.2%) were obese.
The prevalence of obesity was highest in male leukemia survivors (42.5%) and females who survived neuroblastoma (43.6%), followed closely by those who survived leukemia (43.1%).
Multivariable analyses showed that 3 factors were independently associated with an increased risk of obesity: older age at the time of evaluation (≥30 years vs <30 years; P<0.001), undergoing cranial radiation (P<0.001), and receiving glucocorticoids (P=0.004).
On the other hand, receiving chest, abdominal, or pelvic radiation was associated with a decreased risk of obesity (P<0.001).
The researchers also identified 166 single nucleotide polymorphisms that were associated with obesity among cancer survivors who had received cranial radiation. The strongest association was in variants of genes involved in neuron growth, repair, and connectivity.
Among survivors who did not receive cranial radiation, only 1 single nucleotide polymorphism—rs12073359, located on chromosome 1—was associated with an increased risk of obesity.
Dr Wilson said these findings might help us identify the childhood cancer survivors who are most likely to become obese. The results may also provide a foundation for future research efforts aimed at characterizing molecular pathways involved in the link between childhood cancer treatment and obesity. ![]()

Researchers have identified several factors that may influence the risk of obesity in childhood cancer survivors.
Previous research showed that obesity rates are elevated in childhood cancer survivors who were exposed to cranial radiation.
But the new study has shown that other types of treatment, a patient’s age, and certain genetic variants are associated with obesity in this population.
Carmen Wilson, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues reported these findings in Cancer.
The researchers evaluated 1,996 childhood cancer survivors treated at St. Jude. The patients’ median age at diagnosis was 7.2 years (range, 0.1-24.8), and their median age at follow-up was 32.4 years (range, 18.9-63.8).
At the time of evaluation, 645 patients (32.3%) were of normal weight, 71 (3.6%) were underweight, 556 (27.9%) were overweight, and 723 (36.2%) were obese.
The prevalence of obesity was highest in male leukemia survivors (42.5%) and females who survived neuroblastoma (43.6%), followed closely by those who survived leukemia (43.1%).
Multivariable analyses showed that 3 factors were independently associated with an increased risk of obesity: older age at the time of evaluation (≥30 years vs <30 years; P<0.001), undergoing cranial radiation (P<0.001), and receiving glucocorticoids (P=0.004).
On the other hand, receiving chest, abdominal, or pelvic radiation was associated with a decreased risk of obesity (P<0.001).
The researchers also identified 166 single nucleotide polymorphisms that were associated with obesity among cancer survivors who had received cranial radiation. The strongest association was in variants of genes involved in neuron growth, repair, and connectivity.
Among survivors who did not receive cranial radiation, only 1 single nucleotide polymorphism—rs12073359, located on chromosome 1—was associated with an increased risk of obesity.
Dr Wilson said these findings might help us identify the childhood cancer survivors who are most likely to become obese. The results may also provide a foundation for future research efforts aimed at characterizing molecular pathways involved in the link between childhood cancer treatment and obesity. ![]()
New model simulates human MM

Photo by Ben Skála
By growing human tumors on chicken embryos, scientists have created a new model system for screening drugs to treat multiple myeloma (MM).
Several MM drug candidates have shown promising activity in this system, according to the researchers.
Gerold Untergasser, PhD, of Innsbruck Medical University in Austria, and his colleagues described how they developed the system in the Journal of Visualized Experiments.
First, the researchers prepared cultures of the MM cell lines OPM2 and RPMI 8226, as well as human mesenchymal stem cells. They then transfected MM cells with enhanced green fluorescent protein, making the cells easy to observe through a fluorescence microscope.
Next, the team cultured MM cells with mesenchymal cells and collagen to create 3-dimensional cell spheres, thereby simulating the natural microenvironment of the tumor.
The researchers then transferred their cell spheres to the outer membrane of chicken embryos. This choriallantoid membrane provides a suitable base for growing miniature human tumors in culture.
The team has introduced anti-myeloma drugs, such as bortezomib, into this system and assessed the drugs’ ability to target MM cells and prevent tumor growth and angiogenesis. They were also able to evaluate toxicity.
“The chicken egg is much easier to handle and cheaper than mice, and it reduces the number of animal experiments,” Dr Untergasser said. “In our new video publication, we give detailed information on how our system works.”
“We provide an easy-to-repeat protocol for broad use within the research community. Our vision for the future is that a separate test is performed for each patient to determine which drug is best suitable.” ![]()

Photo by Ben Skála
By growing human tumors on chicken embryos, scientists have created a new model system for screening drugs to treat multiple myeloma (MM).
Several MM drug candidates have shown promising activity in this system, according to the researchers.
Gerold Untergasser, PhD, of Innsbruck Medical University in Austria, and his colleagues described how they developed the system in the Journal of Visualized Experiments.
First, the researchers prepared cultures of the MM cell lines OPM2 and RPMI 8226, as well as human mesenchymal stem cells. They then transfected MM cells with enhanced green fluorescent protein, making the cells easy to observe through a fluorescence microscope.
Next, the team cultured MM cells with mesenchymal cells and collagen to create 3-dimensional cell spheres, thereby simulating the natural microenvironment of the tumor.
The researchers then transferred their cell spheres to the outer membrane of chicken embryos. This choriallantoid membrane provides a suitable base for growing miniature human tumors in culture.
The team has introduced anti-myeloma drugs, such as bortezomib, into this system and assessed the drugs’ ability to target MM cells and prevent tumor growth and angiogenesis. They were also able to evaluate toxicity.
“The chicken egg is much easier to handle and cheaper than mice, and it reduces the number of animal experiments,” Dr Untergasser said. “In our new video publication, we give detailed information on how our system works.”
“We provide an easy-to-repeat protocol for broad use within the research community. Our vision for the future is that a separate test is performed for each patient to determine which drug is best suitable.” ![]()

Photo by Ben Skála
By growing human tumors on chicken embryos, scientists have created a new model system for screening drugs to treat multiple myeloma (MM).
Several MM drug candidates have shown promising activity in this system, according to the researchers.
Gerold Untergasser, PhD, of Innsbruck Medical University in Austria, and his colleagues described how they developed the system in the Journal of Visualized Experiments.
First, the researchers prepared cultures of the MM cell lines OPM2 and RPMI 8226, as well as human mesenchymal stem cells. They then transfected MM cells with enhanced green fluorescent protein, making the cells easy to observe through a fluorescence microscope.
Next, the team cultured MM cells with mesenchymal cells and collagen to create 3-dimensional cell spheres, thereby simulating the natural microenvironment of the tumor.
The researchers then transferred their cell spheres to the outer membrane of chicken embryos. This choriallantoid membrane provides a suitable base for growing miniature human tumors in culture.
The team has introduced anti-myeloma drugs, such as bortezomib, into this system and assessed the drugs’ ability to target MM cells and prevent tumor growth and angiogenesis. They were also able to evaluate toxicity.
“The chicken egg is much easier to handle and cheaper than mice, and it reduces the number of animal experiments,” Dr Untergasser said. “In our new video publication, we give detailed information on how our system works.”
“We provide an easy-to-repeat protocol for broad use within the research community. Our vision for the future is that a separate test is performed for each patient to determine which drug is best suitable.” ![]()
Susceptibility to 2nd cancers in WM/LPL survivors

PHILADELPHIA—A retrospective study has revealed factors that appear to influence a person’s susceptibility to Waldenström’s macroglobulinemia (WM)/lymphoplasmacytic lymphoma (LPL) and other malignancies.
Study investigators looked at patients diagnosed with WM or LPL over a 20-year period and found about a 50% excess of second primary cancers in this population.
The patients had a significantly increased risk of multiple hematologic and solid tumor malignancies, and a few of these malignancies had shared susceptibility factors with WM/LPL.
The investigators believe that identifying these factors may prove useful for determining genetic susceptibility to WM/LPL.
Mary L. McMaster, MD, of the National Cancer Institute in Bethesda, Maryland, and her colleagues presented these findings at the AACR Annual Meeting 2015 (abstract 3709).
The team used data from the National Cancer Institute’s Surveillance, Epidemiology and End Results (SSER) database to evaluate the risk of subsequent primary cancer in 3825 patients diagnosed with WM (n=2163) or LPL (n=1662) from 1992 to 2011. The patients’ median age was 70, most of them were male (n=2221), and most were white (n=3153).
Dr McMaster said she and her colleagues looked at both WM and LPL in this study because SEER does not include information about immunoglobulin subtype, which makes it difficult to identify all WM cases with absolute certainty.
“[D]epending on what information a pathologist has when they review a bone marrow biopsy, for example, they may or may not know whether there’s IgM present,” Dr McMaster said. “So you may have a diagnosis of LPL and not have the information required to make the diagnosis of WM. For that reason, we combined both entities for this study.”
Dr McMaster and her colleagues calculated the observed-to-expected standardized incidence ratios (SIRs) for invasive cancers. After adjusting for multiple comparisons, the team found that survivors of WM/LPL had a significantly increased risk of developing a second primary malignancy (SIR=1.49).
This increased risk was seen for males and females and persisted throughout follow-up. The risk was higher for patients younger than 65 years of age (SIR=1.95).
Hematologic malignancies
WM/LPL survivors had a significantly increased risk of several hematologic malignancies. The SIR was 4.09 for all hematologic malignancies, 4.29 for lymphomas, and 3.16 for leukemias.
Dr McMaster pointed out that several lymphoma subtypes can have lymphoplasmacytic differentiation, the most common being marginal zone lymphoma. And this could potentially result in misclassification.
“So we actually ran the study with and without marginal zone lymphoma and saw no difference in the results,” she said. “So we don’t think misclassification accounts for the majority of what we’re seeing.”
The investigators found that WM/LPL survivors had the highest risk of developing Burkitt lymphoma (SIR=13.45), followed by Hodgkin lymphoma (SIR=9.80), T-cell non-Hodgkin lymphoma (SIR=6.62), mantle cell lymphoma (SIR=5.37), diffuse large B-cell lymphoma (DLBCL, SIR=4.76), multiple myeloma (SIR=4.40), any non-Hodgkin lymphoma (SIR=4.08), and acute myeloid leukemia (AML, SIR=3.27).
“Waldenström’s is known to transform, on occasion, to DLBCL,” Dr McMaster said. “So that may well account for the excess of DLBCL that we see in this population.”
She also noted that, prior to the early 2000s, WM was typically treated with alkylating agents. And alkylating agents have been linked to an increased risk of AML.
In this population, the risk of AML peaked 5 to 10 years after WM/LPL diagnosis and was only present in patients treated prior to 2002. This suggests the AML observed in this study was likely treatment-related.
Dr McMaster and her colleagues also found that WM/LPL survivors did not have a significantly increased risk of developing acute lymphocytic leukemia (SIR=0), hairy cell leukemia (SIR=0), chronic lymphocytic leukemia/small lymphocytic lymphoma (SIR=0.97), or follicular lymphoma (SIR=2.25).
Solid tumors
WM/LPL survivors did have a significantly increased risk of certain solid tumor malignancies. The overall SIR for solid tumors was 1.21.
The risk was significant for non-epithelial skin cancers (SIR=5.15), thyroid cancers (SIR=3.13), melanoma (SIR=1.72), and cancers of the lung and bronchus (SIR=1.44) or respiratory system (SIR=1.42).
“Melanoma has an immunological basis, as does Waldenström’s, so we think there may be some shared etiology there,” Dr McMaster said.
She also noted that a strong risk factor for thyroid cancer, particularly papillary thyroid cancer, is a history of autoimmune thyroid disease.
“Autoimmune disease of any sort is a risk factor for Waldenström’s macroglobulinemia,” she said. “So again, we think there might be a basis for shared susceptibility there.”
Dr McMaster said this research suggests that multiple primary cancers may occur in a single individual because of shared genetic susceptibility, shared environmental exposures, treatment effects, or chance. She believes future research will show that both genetic and environmental factors contribute to WM.
Investigators are currently conducting whole-exome sequencing studies and genome-wide association studies in patients with familial and spontaneous WM, with the hopes of identifying genes that contribute to WM susceptibility. ![]()

PHILADELPHIA—A retrospective study has revealed factors that appear to influence a person’s susceptibility to Waldenström’s macroglobulinemia (WM)/lymphoplasmacytic lymphoma (LPL) and other malignancies.
Study investigators looked at patients diagnosed with WM or LPL over a 20-year period and found about a 50% excess of second primary cancers in this population.
The patients had a significantly increased risk of multiple hematologic and solid tumor malignancies, and a few of these malignancies had shared susceptibility factors with WM/LPL.
The investigators believe that identifying these factors may prove useful for determining genetic susceptibility to WM/LPL.
Mary L. McMaster, MD, of the National Cancer Institute in Bethesda, Maryland, and her colleagues presented these findings at the AACR Annual Meeting 2015 (abstract 3709).
The team used data from the National Cancer Institute’s Surveillance, Epidemiology and End Results (SSER) database to evaluate the risk of subsequent primary cancer in 3825 patients diagnosed with WM (n=2163) or LPL (n=1662) from 1992 to 2011. The patients’ median age was 70, most of them were male (n=2221), and most were white (n=3153).
Dr McMaster said she and her colleagues looked at both WM and LPL in this study because SEER does not include information about immunoglobulin subtype, which makes it difficult to identify all WM cases with absolute certainty.
“[D]epending on what information a pathologist has when they review a bone marrow biopsy, for example, they may or may not know whether there’s IgM present,” Dr McMaster said. “So you may have a diagnosis of LPL and not have the information required to make the diagnosis of WM. For that reason, we combined both entities for this study.”
Dr McMaster and her colleagues calculated the observed-to-expected standardized incidence ratios (SIRs) for invasive cancers. After adjusting for multiple comparisons, the team found that survivors of WM/LPL had a significantly increased risk of developing a second primary malignancy (SIR=1.49).
This increased risk was seen for males and females and persisted throughout follow-up. The risk was higher for patients younger than 65 years of age (SIR=1.95).
Hematologic malignancies
WM/LPL survivors had a significantly increased risk of several hematologic malignancies. The SIR was 4.09 for all hematologic malignancies, 4.29 for lymphomas, and 3.16 for leukemias.
Dr McMaster pointed out that several lymphoma subtypes can have lymphoplasmacytic differentiation, the most common being marginal zone lymphoma. And this could potentially result in misclassification.
“So we actually ran the study with and without marginal zone lymphoma and saw no difference in the results,” she said. “So we don’t think misclassification accounts for the majority of what we’re seeing.”
The investigators found that WM/LPL survivors had the highest risk of developing Burkitt lymphoma (SIR=13.45), followed by Hodgkin lymphoma (SIR=9.80), T-cell non-Hodgkin lymphoma (SIR=6.62), mantle cell lymphoma (SIR=5.37), diffuse large B-cell lymphoma (DLBCL, SIR=4.76), multiple myeloma (SIR=4.40), any non-Hodgkin lymphoma (SIR=4.08), and acute myeloid leukemia (AML, SIR=3.27).
“Waldenström’s is known to transform, on occasion, to DLBCL,” Dr McMaster said. “So that may well account for the excess of DLBCL that we see in this population.”
She also noted that, prior to the early 2000s, WM was typically treated with alkylating agents. And alkylating agents have been linked to an increased risk of AML.
In this population, the risk of AML peaked 5 to 10 years after WM/LPL diagnosis and was only present in patients treated prior to 2002. This suggests the AML observed in this study was likely treatment-related.
Dr McMaster and her colleagues also found that WM/LPL survivors did not have a significantly increased risk of developing acute lymphocytic leukemia (SIR=0), hairy cell leukemia (SIR=0), chronic lymphocytic leukemia/small lymphocytic lymphoma (SIR=0.97), or follicular lymphoma (SIR=2.25).
Solid tumors
WM/LPL survivors did have a significantly increased risk of certain solid tumor malignancies. The overall SIR for solid tumors was 1.21.
The risk was significant for non-epithelial skin cancers (SIR=5.15), thyroid cancers (SIR=3.13), melanoma (SIR=1.72), and cancers of the lung and bronchus (SIR=1.44) or respiratory system (SIR=1.42).
“Melanoma has an immunological basis, as does Waldenström’s, so we think there may be some shared etiology there,” Dr McMaster said.
She also noted that a strong risk factor for thyroid cancer, particularly papillary thyroid cancer, is a history of autoimmune thyroid disease.
“Autoimmune disease of any sort is a risk factor for Waldenström’s macroglobulinemia,” she said. “So again, we think there might be a basis for shared susceptibility there.”
Dr McMaster said this research suggests that multiple primary cancers may occur in a single individual because of shared genetic susceptibility, shared environmental exposures, treatment effects, or chance. She believes future research will show that both genetic and environmental factors contribute to WM.
Investigators are currently conducting whole-exome sequencing studies and genome-wide association studies in patients with familial and spontaneous WM, with the hopes of identifying genes that contribute to WM susceptibility. ![]()

PHILADELPHIA—A retrospective study has revealed factors that appear to influence a person’s susceptibility to Waldenström’s macroglobulinemia (WM)/lymphoplasmacytic lymphoma (LPL) and other malignancies.
Study investigators looked at patients diagnosed with WM or LPL over a 20-year period and found about a 50% excess of second primary cancers in this population.
The patients had a significantly increased risk of multiple hematologic and solid tumor malignancies, and a few of these malignancies had shared susceptibility factors with WM/LPL.
The investigators believe that identifying these factors may prove useful for determining genetic susceptibility to WM/LPL.
Mary L. McMaster, MD, of the National Cancer Institute in Bethesda, Maryland, and her colleagues presented these findings at the AACR Annual Meeting 2015 (abstract 3709).
The team used data from the National Cancer Institute’s Surveillance, Epidemiology and End Results (SSER) database to evaluate the risk of subsequent primary cancer in 3825 patients diagnosed with WM (n=2163) or LPL (n=1662) from 1992 to 2011. The patients’ median age was 70, most of them were male (n=2221), and most were white (n=3153).
Dr McMaster said she and her colleagues looked at both WM and LPL in this study because SEER does not include information about immunoglobulin subtype, which makes it difficult to identify all WM cases with absolute certainty.
“[D]epending on what information a pathologist has when they review a bone marrow biopsy, for example, they may or may not know whether there’s IgM present,” Dr McMaster said. “So you may have a diagnosis of LPL and not have the information required to make the diagnosis of WM. For that reason, we combined both entities for this study.”
Dr McMaster and her colleagues calculated the observed-to-expected standardized incidence ratios (SIRs) for invasive cancers. After adjusting for multiple comparisons, the team found that survivors of WM/LPL had a significantly increased risk of developing a second primary malignancy (SIR=1.49).
This increased risk was seen for males and females and persisted throughout follow-up. The risk was higher for patients younger than 65 years of age (SIR=1.95).
Hematologic malignancies
WM/LPL survivors had a significantly increased risk of several hematologic malignancies. The SIR was 4.09 for all hematologic malignancies, 4.29 for lymphomas, and 3.16 for leukemias.
Dr McMaster pointed out that several lymphoma subtypes can have lymphoplasmacytic differentiation, the most common being marginal zone lymphoma. And this could potentially result in misclassification.
“So we actually ran the study with and without marginal zone lymphoma and saw no difference in the results,” she said. “So we don’t think misclassification accounts for the majority of what we’re seeing.”
The investigators found that WM/LPL survivors had the highest risk of developing Burkitt lymphoma (SIR=13.45), followed by Hodgkin lymphoma (SIR=9.80), T-cell non-Hodgkin lymphoma (SIR=6.62), mantle cell lymphoma (SIR=5.37), diffuse large B-cell lymphoma (DLBCL, SIR=4.76), multiple myeloma (SIR=4.40), any non-Hodgkin lymphoma (SIR=4.08), and acute myeloid leukemia (AML, SIR=3.27).
“Waldenström’s is known to transform, on occasion, to DLBCL,” Dr McMaster said. “So that may well account for the excess of DLBCL that we see in this population.”
She also noted that, prior to the early 2000s, WM was typically treated with alkylating agents. And alkylating agents have been linked to an increased risk of AML.
In this population, the risk of AML peaked 5 to 10 years after WM/LPL diagnosis and was only present in patients treated prior to 2002. This suggests the AML observed in this study was likely treatment-related.
Dr McMaster and her colleagues also found that WM/LPL survivors did not have a significantly increased risk of developing acute lymphocytic leukemia (SIR=0), hairy cell leukemia (SIR=0), chronic lymphocytic leukemia/small lymphocytic lymphoma (SIR=0.97), or follicular lymphoma (SIR=2.25).
Solid tumors
WM/LPL survivors did have a significantly increased risk of certain solid tumor malignancies. The overall SIR for solid tumors was 1.21.
The risk was significant for non-epithelial skin cancers (SIR=5.15), thyroid cancers (SIR=3.13), melanoma (SIR=1.72), and cancers of the lung and bronchus (SIR=1.44) or respiratory system (SIR=1.42).
“Melanoma has an immunological basis, as does Waldenström’s, so we think there may be some shared etiology there,” Dr McMaster said.
She also noted that a strong risk factor for thyroid cancer, particularly papillary thyroid cancer, is a history of autoimmune thyroid disease.
“Autoimmune disease of any sort is a risk factor for Waldenström’s macroglobulinemia,” she said. “So again, we think there might be a basis for shared susceptibility there.”
Dr McMaster said this research suggests that multiple primary cancers may occur in a single individual because of shared genetic susceptibility, shared environmental exposures, treatment effects, or chance. She believes future research will show that both genetic and environmental factors contribute to WM.
Investigators are currently conducting whole-exome sequencing studies and genome-wide association studies in patients with familial and spontaneous WM, with the hopes of identifying genes that contribute to WM susceptibility. ![]()
MM drug met accelerated approval requirements

Photo courtesy of the CDC
Celgene Corporation has fulfilled the requirements for accelerated approval of pomalidomide (Pomalyst) in the US, based on results from the phase 3 MM-003 trial.
The trial showed that pomalidomide in combination with dexamethasone can improve survival in patients with relapsed or refractory multiple
myeloma (MM).
A drug can be granted accelerated approval in the US based on a surrogate endpoint thought to predict clinical benefit.
To retain approval from the US Food and Drug Administration (FDA), the drug must demonstrate an actual clinical benefit.
In 2013, the FDA granted pomalidomide accelerated approval for use in combination with dexamethasone to treat MM patients who have received at least 2 prior therapies, including lenalidomide and a proteasome inhibitor, and have demonstrated disease progression on or within 60 days of completing their last therapy.
The FDA’s approval was based on results from a phase 2 trial known as MM-002. The trial showed that pomalidomide plus dexamethasone can improve the overall response rate in relapsed/refractory MM patients when compared to pomalidomide alone.
About 29% of patients in the pomalidomide-dexamethasone arm achieved a partial response or better, compared to about 7% of patients in the pomalidomide-alone arm.
Now, results of the MM-003 trial have shown that pomalidomide plus low-dose dexamethasone can improve progression-free survival and overall survival in relapsed/refractory MM patients, when compared to high-dose dexamethasone alone.
The median progression-free survival was 3.6 months in the pomalidomide-dexamethasone arm and 1.8 months in the dexamethasone arm (P<0.001). And the median overall survival was 12.4 months and 8 months, respectively (P=0.009).
These outcomes suggest pomalidomide, in combination with dexamethasone, provides a clinical benefit for previously treated MM patients, which fulfills the requirements for accelerated approval. So the drug’s label has been updated to reflect his change.
For more details on pomalidomide, see the full prescribing information. ![]()

Photo courtesy of the CDC
Celgene Corporation has fulfilled the requirements for accelerated approval of pomalidomide (Pomalyst) in the US, based on results from the phase 3 MM-003 trial.
The trial showed that pomalidomide in combination with dexamethasone can improve survival in patients with relapsed or refractory multiple
myeloma (MM).
A drug can be granted accelerated approval in the US based on a surrogate endpoint thought to predict clinical benefit.
To retain approval from the US Food and Drug Administration (FDA), the drug must demonstrate an actual clinical benefit.
In 2013, the FDA granted pomalidomide accelerated approval for use in combination with dexamethasone to treat MM patients who have received at least 2 prior therapies, including lenalidomide and a proteasome inhibitor, and have demonstrated disease progression on or within 60 days of completing their last therapy.
The FDA’s approval was based on results from a phase 2 trial known as MM-002. The trial showed that pomalidomide plus dexamethasone can improve the overall response rate in relapsed/refractory MM patients when compared to pomalidomide alone.
About 29% of patients in the pomalidomide-dexamethasone arm achieved a partial response or better, compared to about 7% of patients in the pomalidomide-alone arm.
Now, results of the MM-003 trial have shown that pomalidomide plus low-dose dexamethasone can improve progression-free survival and overall survival in relapsed/refractory MM patients, when compared to high-dose dexamethasone alone.
The median progression-free survival was 3.6 months in the pomalidomide-dexamethasone arm and 1.8 months in the dexamethasone arm (P<0.001). And the median overall survival was 12.4 months and 8 months, respectively (P=0.009).
These outcomes suggest pomalidomide, in combination with dexamethasone, provides a clinical benefit for previously treated MM patients, which fulfills the requirements for accelerated approval. So the drug’s label has been updated to reflect his change.
For more details on pomalidomide, see the full prescribing information. ![]()

Photo courtesy of the CDC
Celgene Corporation has fulfilled the requirements for accelerated approval of pomalidomide (Pomalyst) in the US, based on results from the phase 3 MM-003 trial.
The trial showed that pomalidomide in combination with dexamethasone can improve survival in patients with relapsed or refractory multiple
myeloma (MM).
A drug can be granted accelerated approval in the US based on a surrogate endpoint thought to predict clinical benefit.
To retain approval from the US Food and Drug Administration (FDA), the drug must demonstrate an actual clinical benefit.
In 2013, the FDA granted pomalidomide accelerated approval for use in combination with dexamethasone to treat MM patients who have received at least 2 prior therapies, including lenalidomide and a proteasome inhibitor, and have demonstrated disease progression on or within 60 days of completing their last therapy.
The FDA’s approval was based on results from a phase 2 trial known as MM-002. The trial showed that pomalidomide plus dexamethasone can improve the overall response rate in relapsed/refractory MM patients when compared to pomalidomide alone.
About 29% of patients in the pomalidomide-dexamethasone arm achieved a partial response or better, compared to about 7% of patients in the pomalidomide-alone arm.
Now, results of the MM-003 trial have shown that pomalidomide plus low-dose dexamethasone can improve progression-free survival and overall survival in relapsed/refractory MM patients, when compared to high-dose dexamethasone alone.
The median progression-free survival was 3.6 months in the pomalidomide-dexamethasone arm and 1.8 months in the dexamethasone arm (P<0.001). And the median overall survival was 12.4 months and 8 months, respectively (P=0.009).
These outcomes suggest pomalidomide, in combination with dexamethasone, provides a clinical benefit for previously treated MM patients, which fulfills the requirements for accelerated approval. So the drug’s label has been updated to reflect his change.
For more details on pomalidomide, see the full prescribing information.
Drug that fell short in prostate cancer could treat MM
PHILADELPHIA—A drug that has fallen short of expectations in clinical trials of prostate cancer may be effective for treating multiple myeloma (MM), according to research presented at the AACR Annual Meeting 2015.
The drug, tasquinimod, inhibits the function of S100A9, a pro-inflammatory protein that is elevated in MM, prostate cancer, and other malignancies.
Researchers found that tasquinimod can reduce tumor growth and improve survival in mouse models of MM. And these effects are associated with reduced angiogenesis in the bone marrow.
Cindy Lin, PhD, of the Wistar Institute in Philadelphia, Pennsylvania, and her colleagues presented these findings as abstract 1364.* The research was supported by Active Biotech and Ipsen, the companies developing tasquinimod.
Dr Lin noted that tasquinimod has already been tested in clinical trials of prostate cancer and initially appeared to be very effective. However, recent results from a phase 3 trial suggested the drug does not confer a favorable risk-benefit ratio for this population.
So Active Biotech and Ipsen decided to discontinue all trials of tasquinimod in prostate cancer. But the preclinical results observed in MM suggest tasquinimod may hold promise for treating these patients.
Activity in MM
Dr Lin said previous preclinical experiments revealed that myeloid-derived suppressor cells are involved in regulating MM progression, and these cells produce S100A9.
Tasquinimod is a quinoline-3-carboxamide derivative that binds to S100A9 and inhibits interaction with its receptors. So Dr Lin and her colleagues decided to investigate the antitumor effect of the drug in mouse models of MM.
The researchers initially tested tasquinimod in a syngeneic MM model, randomizing mice to treatment or control. Mice in the treatment group received tasquinimod at 30 mg/kg/day in their drinking water for 28 days.
Tasquinimod significantly improved survival in this model (P<0.005). All control mice died within 30 days of tumor inoculation, but about 40% of tasquinimod-treated mice were still alive more than 80 days out.
The researchers then tested tasquinimod in xenograft models of human MM. The drug significantly reduced tumor size in both H929 (P=0.0042) and RPMI-8226 models (P=0.0003). Treatment significantly improved survival in the H929 (P=0.0008) and RPMI-8226 models as well (P=0.0243).
Dr Lin said she and her colleagues did not see any side effects of treatment in any of the mice.
Investigating the mechanism
To determine if the antitumor effect of tasquinimod is, in fact, mediated through inhibition of S100A9, the researchers administered the drug to S100A9-knockout mice with MM. Tasquinimod did not improve survival in these mice, which suggests its anti-MM effects are mediated through S100A9 inhibition.
“To try and investigate some of the mechanisms of how survival is improved in tasquinimod-treated, tumor-bearing mice, we looked at a variety of different things, including angiogenesis,” Dr Lin said.
“We used CD31 immunohistochemistry to look at angiogenesis, and, in the untreated mice, we didn’t see a lot of staining. But in the tumor-bearing mice, there was a lot more staining [P=0.0231]. And when we gave the mice tasquinimod, angiogenesis was significantly decreased [P<0.0001].”
The researchers also looked at different angiogenic factors. And they found that, compared to control-treated mice with MM, tumor-bearing mice that received tasquinimod had a significant decrease in serum levels of VEGF, FGF2, tissue factor, and endoglin.
The team is now assessing the effects of S100A9 and tasquinimod on megakaryocytes and platelets, 2 of the major cell populations that promote angiogenesis. ![]()
*Information in the abstract differs from that presented at the meeting.
PHILADELPHIA—A drug that has fallen short of expectations in clinical trials of prostate cancer may be effective for treating multiple myeloma (MM), according to research presented at the AACR Annual Meeting 2015.
The drug, tasquinimod, inhibits the function of S100A9, a pro-inflammatory protein that is elevated in MM, prostate cancer, and other malignancies.
Researchers found that tasquinimod can reduce tumor growth and improve survival in mouse models of MM. And these effects are associated with reduced angiogenesis in the bone marrow.
Cindy Lin, PhD, of the Wistar Institute in Philadelphia, Pennsylvania, and her colleagues presented these findings as abstract 1364.* The research was supported by Active Biotech and Ipsen, the companies developing tasquinimod.
Dr Lin noted that tasquinimod has already been tested in clinical trials of prostate cancer and initially appeared to be very effective. However, recent results from a phase 3 trial suggested the drug does not confer a favorable risk-benefit ratio for this population.
So Active Biotech and Ipsen decided to discontinue all trials of tasquinimod in prostate cancer. But the preclinical results observed in MM suggest tasquinimod may hold promise for treating these patients.
Activity in MM
Dr Lin said previous preclinical experiments revealed that myeloid-derived suppressor cells are involved in regulating MM progression, and these cells produce S100A9.
Tasquinimod is a quinoline-3-carboxamide derivative that binds to S100A9 and inhibits interaction with its receptors. So Dr Lin and her colleagues decided to investigate the antitumor effect of the drug in mouse models of MM.
The researchers initially tested tasquinimod in a syngeneic MM model, randomizing mice to treatment or control. Mice in the treatment group received tasquinimod at 30 mg/kg/day in their drinking water for 28 days.
Tasquinimod significantly improved survival in this model (P<0.005). All control mice died within 30 days of tumor inoculation, but about 40% of tasquinimod-treated mice were still alive more than 80 days out.
The researchers then tested tasquinimod in xenograft models of human MM. The drug significantly reduced tumor size in both H929 (P=0.0042) and RPMI-8226 models (P=0.0003). Treatment significantly improved survival in the H929 (P=0.0008) and RPMI-8226 models as well (P=0.0243).
Dr Lin said she and her colleagues did not see any side effects of treatment in any of the mice.
Investigating the mechanism
To determine if the antitumor effect of tasquinimod is, in fact, mediated through inhibition of S100A9, the researchers administered the drug to S100A9-knockout mice with MM. Tasquinimod did not improve survival in these mice, which suggests its anti-MM effects are mediated through S100A9 inhibition.
“To try and investigate some of the mechanisms of how survival is improved in tasquinimod-treated, tumor-bearing mice, we looked at a variety of different things, including angiogenesis,” Dr Lin said.
“We used CD31 immunohistochemistry to look at angiogenesis, and, in the untreated mice, we didn’t see a lot of staining. But in the tumor-bearing mice, there was a lot more staining [P=0.0231]. And when we gave the mice tasquinimod, angiogenesis was significantly decreased [P<0.0001].”
The researchers also looked at different angiogenic factors. And they found that, compared to control-treated mice with MM, tumor-bearing mice that received tasquinimod had a significant decrease in serum levels of VEGF, FGF2, tissue factor, and endoglin.
The team is now assessing the effects of S100A9 and tasquinimod on megakaryocytes and platelets, 2 of the major cell populations that promote angiogenesis. ![]()
*Information in the abstract differs from that presented at the meeting.
PHILADELPHIA—A drug that has fallen short of expectations in clinical trials of prostate cancer may be effective for treating multiple myeloma (MM), according to research presented at the AACR Annual Meeting 2015.
The drug, tasquinimod, inhibits the function of S100A9, a pro-inflammatory protein that is elevated in MM, prostate cancer, and other malignancies.
Researchers found that tasquinimod can reduce tumor growth and improve survival in mouse models of MM. And these effects are associated with reduced angiogenesis in the bone marrow.
Cindy Lin, PhD, of the Wistar Institute in Philadelphia, Pennsylvania, and her colleagues presented these findings as abstract 1364.* The research was supported by Active Biotech and Ipsen, the companies developing tasquinimod.
Dr Lin noted that tasquinimod has already been tested in clinical trials of prostate cancer and initially appeared to be very effective. However, recent results from a phase 3 trial suggested the drug does not confer a favorable risk-benefit ratio for this population.
So Active Biotech and Ipsen decided to discontinue all trials of tasquinimod in prostate cancer. But the preclinical results observed in MM suggest tasquinimod may hold promise for treating these patients.
Activity in MM
Dr Lin said previous preclinical experiments revealed that myeloid-derived suppressor cells are involved in regulating MM progression, and these cells produce S100A9.
Tasquinimod is a quinoline-3-carboxamide derivative that binds to S100A9 and inhibits interaction with its receptors. So Dr Lin and her colleagues decided to investigate the antitumor effect of the drug in mouse models of MM.
The researchers initially tested tasquinimod in a syngeneic MM model, randomizing mice to treatment or control. Mice in the treatment group received tasquinimod at 30 mg/kg/day in their drinking water for 28 days.
Tasquinimod significantly improved survival in this model (P<0.005). All control mice died within 30 days of tumor inoculation, but about 40% of tasquinimod-treated mice were still alive more than 80 days out.
The researchers then tested tasquinimod in xenograft models of human MM. The drug significantly reduced tumor size in both H929 (P=0.0042) and RPMI-8226 models (P=0.0003). Treatment significantly improved survival in the H929 (P=0.0008) and RPMI-8226 models as well (P=0.0243).
Dr Lin said she and her colleagues did not see any side effects of treatment in any of the mice.
Investigating the mechanism
To determine if the antitumor effect of tasquinimod is, in fact, mediated through inhibition of S100A9, the researchers administered the drug to S100A9-knockout mice with MM. Tasquinimod did not improve survival in these mice, which suggests its anti-MM effects are mediated through S100A9 inhibition.
“To try and investigate some of the mechanisms of how survival is improved in tasquinimod-treated, tumor-bearing mice, we looked at a variety of different things, including angiogenesis,” Dr Lin said.
“We used CD31 immunohistochemistry to look at angiogenesis, and, in the untreated mice, we didn’t see a lot of staining. But in the tumor-bearing mice, there was a lot more staining [P=0.0231]. And when we gave the mice tasquinimod, angiogenesis was significantly decreased [P<0.0001].”
The researchers also looked at different angiogenic factors. And they found that, compared to control-treated mice with MM, tumor-bearing mice that received tasquinimod had a significant decrease in serum levels of VEGF, FGF2, tissue factor, and endoglin.
The team is now assessing the effects of S100A9 and tasquinimod on megakaryocytes and platelets, 2 of the major cell populations that promote angiogenesis. ![]()
*Information in the abstract differs from that presented at the meeting.


