User login
For MD-IQ use only
Doctors are disappearing from emergency departments as hospitals look to cut costs
She didn’t know much about miscarriage, but this seemed like one.
In the emergency department, she was examined then sent home, she said. She went back when her cramping became excruciating. Then home again. It ultimately took three trips to the ED on 3 consecutive days, generating three separate bills, before she saw a doctor who looked at her blood work and confirmed her fears.
“At the time I wasn’t thinking, ‘Oh, I need to see a doctor,’ ” Ms. Valle recalled. “But when you think about it, it’s like, ‘Well, dang – why didn’t I see a doctor?’ ” It’s unclear whether the repeat visits were due to delays in seeing a physician, but the experience worried her. And she’s still paying the bills.
The hospital declined to discuss Ms. Valle’s care, citing patient privacy. But 17 months before her 3-day ordeal, Tennova had outsourced its emergency departments to American Physician Partners, a medical staffing company owned by private equity investors. APP employs fewer doctors in its EDs as one of its cost-saving initiatives to increase earnings, according to a confidential company document obtained by KHN and NPR.
This staffing strategy has permeated hospitals, and particularly emergency departments, that seek to reduce their top expense: physician labor. While diagnosing and treating patients was once their domain, doctors are increasingly being replaced by nurse practitioners and physician assistants, collectively known as “midlevel practitioners,” who can perform many of the same duties and generate much of the same revenue for less than half of the pay.
“APP has numerous cost saving initiatives underway as part of the Company’s continual focus on cost optimization,” the document says, including a “shift of staffing” between doctors and midlevel practitioners.
In a statement to KHN, American Physician Partners said this strategy is a way to ensure all EDs remain fully staffed, calling it a “blended model” that allows doctors, nurse practitioners, and physician assistants “to provide care to their fullest potential.”
Critics of this strategy say the quest to save money results in treatment meted out by someone with far less training than a physician, leaving patients vulnerable to misdiagnoses, higher medical bills, and inadequate care. And these fears are bolstered by evidence that suggests dropping doctors from EDs may not be good for patients.
A working paper, published in October by the National Bureau of Economic Research, analyzed roughly 1.1 million visits to 44 EDs throughout the Veterans Health Administration, where nurse practitioners can treat patients without oversight from doctors.
Researchers found that treatment by a nurse practitioner resulted on average in a 7% increase in cost of care and an 11% increase in length of stay, extending patients’ time in the ED by minutes for minor visits and hours for longer ones. These gaps widened among patients with more severe diagnoses, the study said, but could be somewhat mitigated by nurse practitioners with more experience.
The study also found that ED patients treated by a nurse practitioner were 20% more likely to be readmitted to the hospital for a preventable reason within 30 days, although the overall risk of readmission remained very small.
Yiqun Chen, PhD, who is an assistant professor of economics at the University of Illinois at Chicago and coauthored the study, said these findings are not an indictment of nurse practitioners in the ED. Instead, she said, she hopes the study will guide how to best deploy nurse practitioners: in treatment of simpler patients or circumstances when no doctor is available.
“It’s not just a simple question of if we can substitute physicians with nurse practitioners or not,” Dr. Chen said. “It depends on how we use them. If we just use them as independent providers, especially ... for relatively complicated patients, it doesn’t seem to be a very good use.”
Dr. Chen’s research echoes smaller studies, like one from The Harvey L. Neiman Health Policy Institute that found nonphysician practitioners in EDs were associated with a 5.3% increase in imaging, which could unnecessarily increase bills for patients. Separately, a study at the Hattiesburg Clinic in Mississippi found that midlevel practitioners in primary care – not in the emergency department – increased the out-of-pocket costs to patients while also leading to worse performance on 9 of 10 quality-of-care metrics, including cancer screenings and vaccination rates.
But definitive evidence remains elusive that replacing ER doctors with nonphysicians has a negative impact on patients, said Cameron Gettel, MD, an assistant professor of emergency medicine at Yale University, New Haven, Conn. Private equity investment and the use of midlevel practitioners rose in lockstep in the ED, Dr. Gettel said, and in the absence of game-changing research, the pattern will likely continue.
“Worse patient outcomes haven’t really been shown across the board,” he said. “And I think until that is shown, then they will continue to play an increasing role.”
For private equity, dropping ED docs is a “simple equation”
Private equity companies pool money from wealthy investors to buy their way into various industries, often slashing spending and seeking to flip businesses in 3 to 7 years. While this business model is a proven moneymaker on Wall Street, it raises concerns in health care, where critics worry the pressure to turn big profits will influence life-or-death decisions that were once left solely to medical professionals.
Nearly $1 trillion in private equity funds have gone into almost 8,000 health care transactions over the past decade, according to industry tracker PitchBook, including buying into medical staffing companies that many hospitals hire to manage their emergency departments.
Two firms dominate the ED staffing industry: TeamHealth, bought by private equity firm Blackstone in 2016, and Envision Healthcare, bought by KKR in 2018. Trying to undercut these staffing giants is American Physician Partners, a rapidly expanding company that runs EDs in at least 17 states and is 50% owned by private equity firm BBH Capital Partners.
These staffing companies have been among the most aggressive in replacing doctors to cut costs, said Robert McNamara, MD, a founder of the American Academy of Emergency Medicine and chair of emergency medicine at Temple University, Philadelphia.
“It’s a relatively simple equation,” Dr. McNamara said. “Their No. 1 expense is the board-certified emergency physician. So they are going to want to keep that expense as low as possible.”
Not everyone sees the trend of private equity in ED staffing in a negative light. Jennifer Orozco, president of the American Academy of Physician Associates, which represents physician assistants, said even if the change – to use more nonphysician providers – is driven by the staffing firms’ desire to make more money, patients are still well served by a team approach that includes nurse practitioners and physician assistants.
“Though I see that shift, it’s not about profits at the end of the day,” Ms. Orozco said. “It’s about the patient.”
The “shift” is nearly invisible to patients because hospitals rarely promote branding from their ED staffing firms and there is little public documentation of private equity investments.
Arthur Smolensky, MD, a Tennessee emergency medicine specialist attempting to measure private equity’s intrusion into EDs, said his review of hospital job postings and employment contracts in 14 major metropolitan areas found that 43% of ED patients were seen in EDs staffed by companies with nonphysician owners, nearly all of whom are private equity investors.
Dr. Smolensky hopes to publish his full study, expanding to 55 metro areas, later this year. But this research will merely quantify what many doctors already know: The ED has changed. Demoralized by an increased focus on profit, and wary of a looming surplus of emergency medicine residents because there are fewer jobs to fill, many experienced doctors are leaving the ED on their own, he said.
“Most of us didn’t go into medicine to supervise an army of people that are not as well trained as we are,” Dr. Smolensky said. “We want to take care of patients.”
“I guess we’re the first guinea pigs for our ER”
Joshua Allen, a nurse practitioner at a small Kentucky hospital, snaked a rubber hose through a rack of pork ribs to practice inserting a chest tube to fix a collapsed lung.
It was 2020, and American Physician Partners was restructuring the ED where Mr. Allen worked, reducing shifts from two doctors to one. Once Mr. Allen had placed 10 tubes under a doctor’s supervision, he would be allowed to do it on his own.
“I guess we’re the first guinea pigs for our ER,” he said. “If we do have a major trauma and multiple victims come in, there’s only one doctor there. ... We need to be prepared.”
Mr. Allen is one of many midlevel practitioners finding work in emergency departments. Nurse practitioners and physician assistants are among the fastest-growing occupations in the nation, according to the U.S. Bureau of Labor Statistics.
Generally, they have master’s degrees and receive several years of specialized schooling but have significantly less training than doctors. Many are permitted to diagnose patients and prescribe medication with little or no supervision from a doctor, although limitations vary by state.
The Neiman Institute found that the share of ED visits in which a midlevel practitioner was the main clinician increased by more than 172% between 2005 and 2020. Another study, in the Journal of Emergency Medicine, reported that if trends continue there may be equal numbers of midlevel practitioners and doctors in EDs by 2030.
There is little mystery as to why. Federal data shows emergency medicine doctors are paid about $310,000 a year on average, while nurse practitioners and physician assistants earn less than $120,000. Generally, hospitals can bill for care by a midlevel practitioner at 85% the rate of a doctor while paying them less than half as much.
Private equity can make millions in the gap.
For example, Envision once encouraged EDs to employ “the least expensive resource” and treat up to 35% of patients with midlevel practitioners, according to a 2017 PowerPoint presentation. The presentation drew scorn on social media and disappeared from Envision’s website.
Envision declined a request for a phone interview. In a written statement to KHN, spokesperson Aliese Polk said the company does not direct its physician leaders on how to care for patients and called the presentation a “concept guide” that does not represent current views.
American Physician Partners touted roughly the same staffing strategy in 2021 in response to the No Surprises Act, which threatened the company’s profits by outlawing surprise medical bills. In its confidential pitch to lenders, the company estimated it could cut almost $6 million by shifting more staffing from physicians to midlevel practitioners.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
She didn’t know much about miscarriage, but this seemed like one.
In the emergency department, she was examined then sent home, she said. She went back when her cramping became excruciating. Then home again. It ultimately took three trips to the ED on 3 consecutive days, generating three separate bills, before she saw a doctor who looked at her blood work and confirmed her fears.
“At the time I wasn’t thinking, ‘Oh, I need to see a doctor,’ ” Ms. Valle recalled. “But when you think about it, it’s like, ‘Well, dang – why didn’t I see a doctor?’ ” It’s unclear whether the repeat visits were due to delays in seeing a physician, but the experience worried her. And she’s still paying the bills.
The hospital declined to discuss Ms. Valle’s care, citing patient privacy. But 17 months before her 3-day ordeal, Tennova had outsourced its emergency departments to American Physician Partners, a medical staffing company owned by private equity investors. APP employs fewer doctors in its EDs as one of its cost-saving initiatives to increase earnings, according to a confidential company document obtained by KHN and NPR.
This staffing strategy has permeated hospitals, and particularly emergency departments, that seek to reduce their top expense: physician labor. While diagnosing and treating patients was once their domain, doctors are increasingly being replaced by nurse practitioners and physician assistants, collectively known as “midlevel practitioners,” who can perform many of the same duties and generate much of the same revenue for less than half of the pay.
“APP has numerous cost saving initiatives underway as part of the Company’s continual focus on cost optimization,” the document says, including a “shift of staffing” between doctors and midlevel practitioners.
In a statement to KHN, American Physician Partners said this strategy is a way to ensure all EDs remain fully staffed, calling it a “blended model” that allows doctors, nurse practitioners, and physician assistants “to provide care to their fullest potential.”
Critics of this strategy say the quest to save money results in treatment meted out by someone with far less training than a physician, leaving patients vulnerable to misdiagnoses, higher medical bills, and inadequate care. And these fears are bolstered by evidence that suggests dropping doctors from EDs may not be good for patients.
A working paper, published in October by the National Bureau of Economic Research, analyzed roughly 1.1 million visits to 44 EDs throughout the Veterans Health Administration, where nurse practitioners can treat patients without oversight from doctors.
Researchers found that treatment by a nurse practitioner resulted on average in a 7% increase in cost of care and an 11% increase in length of stay, extending patients’ time in the ED by minutes for minor visits and hours for longer ones. These gaps widened among patients with more severe diagnoses, the study said, but could be somewhat mitigated by nurse practitioners with more experience.
The study also found that ED patients treated by a nurse practitioner were 20% more likely to be readmitted to the hospital for a preventable reason within 30 days, although the overall risk of readmission remained very small.
Yiqun Chen, PhD, who is an assistant professor of economics at the University of Illinois at Chicago and coauthored the study, said these findings are not an indictment of nurse practitioners in the ED. Instead, she said, she hopes the study will guide how to best deploy nurse practitioners: in treatment of simpler patients or circumstances when no doctor is available.
“It’s not just a simple question of if we can substitute physicians with nurse practitioners or not,” Dr. Chen said. “It depends on how we use them. If we just use them as independent providers, especially ... for relatively complicated patients, it doesn’t seem to be a very good use.”
Dr. Chen’s research echoes smaller studies, like one from The Harvey L. Neiman Health Policy Institute that found nonphysician practitioners in EDs were associated with a 5.3% increase in imaging, which could unnecessarily increase bills for patients. Separately, a study at the Hattiesburg Clinic in Mississippi found that midlevel practitioners in primary care – not in the emergency department – increased the out-of-pocket costs to patients while also leading to worse performance on 9 of 10 quality-of-care metrics, including cancer screenings and vaccination rates.
But definitive evidence remains elusive that replacing ER doctors with nonphysicians has a negative impact on patients, said Cameron Gettel, MD, an assistant professor of emergency medicine at Yale University, New Haven, Conn. Private equity investment and the use of midlevel practitioners rose in lockstep in the ED, Dr. Gettel said, and in the absence of game-changing research, the pattern will likely continue.
“Worse patient outcomes haven’t really been shown across the board,” he said. “And I think until that is shown, then they will continue to play an increasing role.”
For private equity, dropping ED docs is a “simple equation”
Private equity companies pool money from wealthy investors to buy their way into various industries, often slashing spending and seeking to flip businesses in 3 to 7 years. While this business model is a proven moneymaker on Wall Street, it raises concerns in health care, where critics worry the pressure to turn big profits will influence life-or-death decisions that were once left solely to medical professionals.
Nearly $1 trillion in private equity funds have gone into almost 8,000 health care transactions over the past decade, according to industry tracker PitchBook, including buying into medical staffing companies that many hospitals hire to manage their emergency departments.
Two firms dominate the ED staffing industry: TeamHealth, bought by private equity firm Blackstone in 2016, and Envision Healthcare, bought by KKR in 2018. Trying to undercut these staffing giants is American Physician Partners, a rapidly expanding company that runs EDs in at least 17 states and is 50% owned by private equity firm BBH Capital Partners.
These staffing companies have been among the most aggressive in replacing doctors to cut costs, said Robert McNamara, MD, a founder of the American Academy of Emergency Medicine and chair of emergency medicine at Temple University, Philadelphia.
“It’s a relatively simple equation,” Dr. McNamara said. “Their No. 1 expense is the board-certified emergency physician. So they are going to want to keep that expense as low as possible.”
Not everyone sees the trend of private equity in ED staffing in a negative light. Jennifer Orozco, president of the American Academy of Physician Associates, which represents physician assistants, said even if the change – to use more nonphysician providers – is driven by the staffing firms’ desire to make more money, patients are still well served by a team approach that includes nurse practitioners and physician assistants.
“Though I see that shift, it’s not about profits at the end of the day,” Ms. Orozco said. “It’s about the patient.”
The “shift” is nearly invisible to patients because hospitals rarely promote branding from their ED staffing firms and there is little public documentation of private equity investments.
Arthur Smolensky, MD, a Tennessee emergency medicine specialist attempting to measure private equity’s intrusion into EDs, said his review of hospital job postings and employment contracts in 14 major metropolitan areas found that 43% of ED patients were seen in EDs staffed by companies with nonphysician owners, nearly all of whom are private equity investors.
Dr. Smolensky hopes to publish his full study, expanding to 55 metro areas, later this year. But this research will merely quantify what many doctors already know: The ED has changed. Demoralized by an increased focus on profit, and wary of a looming surplus of emergency medicine residents because there are fewer jobs to fill, many experienced doctors are leaving the ED on their own, he said.
“Most of us didn’t go into medicine to supervise an army of people that are not as well trained as we are,” Dr. Smolensky said. “We want to take care of patients.”
“I guess we’re the first guinea pigs for our ER”
Joshua Allen, a nurse practitioner at a small Kentucky hospital, snaked a rubber hose through a rack of pork ribs to practice inserting a chest tube to fix a collapsed lung.
It was 2020, and American Physician Partners was restructuring the ED where Mr. Allen worked, reducing shifts from two doctors to one. Once Mr. Allen had placed 10 tubes under a doctor’s supervision, he would be allowed to do it on his own.
“I guess we’re the first guinea pigs for our ER,” he said. “If we do have a major trauma and multiple victims come in, there’s only one doctor there. ... We need to be prepared.”
Mr. Allen is one of many midlevel practitioners finding work in emergency departments. Nurse practitioners and physician assistants are among the fastest-growing occupations in the nation, according to the U.S. Bureau of Labor Statistics.
Generally, they have master’s degrees and receive several years of specialized schooling but have significantly less training than doctors. Many are permitted to diagnose patients and prescribe medication with little or no supervision from a doctor, although limitations vary by state.
The Neiman Institute found that the share of ED visits in which a midlevel practitioner was the main clinician increased by more than 172% between 2005 and 2020. Another study, in the Journal of Emergency Medicine, reported that if trends continue there may be equal numbers of midlevel practitioners and doctors in EDs by 2030.
There is little mystery as to why. Federal data shows emergency medicine doctors are paid about $310,000 a year on average, while nurse practitioners and physician assistants earn less than $120,000. Generally, hospitals can bill for care by a midlevel practitioner at 85% the rate of a doctor while paying them less than half as much.
Private equity can make millions in the gap.
For example, Envision once encouraged EDs to employ “the least expensive resource” and treat up to 35% of patients with midlevel practitioners, according to a 2017 PowerPoint presentation. The presentation drew scorn on social media and disappeared from Envision’s website.
Envision declined a request for a phone interview. In a written statement to KHN, spokesperson Aliese Polk said the company does not direct its physician leaders on how to care for patients and called the presentation a “concept guide” that does not represent current views.
American Physician Partners touted roughly the same staffing strategy in 2021 in response to the No Surprises Act, which threatened the company’s profits by outlawing surprise medical bills. In its confidential pitch to lenders, the company estimated it could cut almost $6 million by shifting more staffing from physicians to midlevel practitioners.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
She didn’t know much about miscarriage, but this seemed like one.
In the emergency department, she was examined then sent home, she said. She went back when her cramping became excruciating. Then home again. It ultimately took three trips to the ED on 3 consecutive days, generating three separate bills, before she saw a doctor who looked at her blood work and confirmed her fears.
“At the time I wasn’t thinking, ‘Oh, I need to see a doctor,’ ” Ms. Valle recalled. “But when you think about it, it’s like, ‘Well, dang – why didn’t I see a doctor?’ ” It’s unclear whether the repeat visits were due to delays in seeing a physician, but the experience worried her. And she’s still paying the bills.
The hospital declined to discuss Ms. Valle’s care, citing patient privacy. But 17 months before her 3-day ordeal, Tennova had outsourced its emergency departments to American Physician Partners, a medical staffing company owned by private equity investors. APP employs fewer doctors in its EDs as one of its cost-saving initiatives to increase earnings, according to a confidential company document obtained by KHN and NPR.
This staffing strategy has permeated hospitals, and particularly emergency departments, that seek to reduce their top expense: physician labor. While diagnosing and treating patients was once their domain, doctors are increasingly being replaced by nurse practitioners and physician assistants, collectively known as “midlevel practitioners,” who can perform many of the same duties and generate much of the same revenue for less than half of the pay.
“APP has numerous cost saving initiatives underway as part of the Company’s continual focus on cost optimization,” the document says, including a “shift of staffing” between doctors and midlevel practitioners.
In a statement to KHN, American Physician Partners said this strategy is a way to ensure all EDs remain fully staffed, calling it a “blended model” that allows doctors, nurse practitioners, and physician assistants “to provide care to their fullest potential.”
Critics of this strategy say the quest to save money results in treatment meted out by someone with far less training than a physician, leaving patients vulnerable to misdiagnoses, higher medical bills, and inadequate care. And these fears are bolstered by evidence that suggests dropping doctors from EDs may not be good for patients.
A working paper, published in October by the National Bureau of Economic Research, analyzed roughly 1.1 million visits to 44 EDs throughout the Veterans Health Administration, where nurse practitioners can treat patients without oversight from doctors.
Researchers found that treatment by a nurse practitioner resulted on average in a 7% increase in cost of care and an 11% increase in length of stay, extending patients’ time in the ED by minutes for minor visits and hours for longer ones. These gaps widened among patients with more severe diagnoses, the study said, but could be somewhat mitigated by nurse practitioners with more experience.
The study also found that ED patients treated by a nurse practitioner were 20% more likely to be readmitted to the hospital for a preventable reason within 30 days, although the overall risk of readmission remained very small.
Yiqun Chen, PhD, who is an assistant professor of economics at the University of Illinois at Chicago and coauthored the study, said these findings are not an indictment of nurse practitioners in the ED. Instead, she said, she hopes the study will guide how to best deploy nurse practitioners: in treatment of simpler patients or circumstances when no doctor is available.
“It’s not just a simple question of if we can substitute physicians with nurse practitioners or not,” Dr. Chen said. “It depends on how we use them. If we just use them as independent providers, especially ... for relatively complicated patients, it doesn’t seem to be a very good use.”
Dr. Chen’s research echoes smaller studies, like one from The Harvey L. Neiman Health Policy Institute that found nonphysician practitioners in EDs were associated with a 5.3% increase in imaging, which could unnecessarily increase bills for patients. Separately, a study at the Hattiesburg Clinic in Mississippi found that midlevel practitioners in primary care – not in the emergency department – increased the out-of-pocket costs to patients while also leading to worse performance on 9 of 10 quality-of-care metrics, including cancer screenings and vaccination rates.
But definitive evidence remains elusive that replacing ER doctors with nonphysicians has a negative impact on patients, said Cameron Gettel, MD, an assistant professor of emergency medicine at Yale University, New Haven, Conn. Private equity investment and the use of midlevel practitioners rose in lockstep in the ED, Dr. Gettel said, and in the absence of game-changing research, the pattern will likely continue.
“Worse patient outcomes haven’t really been shown across the board,” he said. “And I think until that is shown, then they will continue to play an increasing role.”
For private equity, dropping ED docs is a “simple equation”
Private equity companies pool money from wealthy investors to buy their way into various industries, often slashing spending and seeking to flip businesses in 3 to 7 years. While this business model is a proven moneymaker on Wall Street, it raises concerns in health care, where critics worry the pressure to turn big profits will influence life-or-death decisions that were once left solely to medical professionals.
Nearly $1 trillion in private equity funds have gone into almost 8,000 health care transactions over the past decade, according to industry tracker PitchBook, including buying into medical staffing companies that many hospitals hire to manage their emergency departments.
Two firms dominate the ED staffing industry: TeamHealth, bought by private equity firm Blackstone in 2016, and Envision Healthcare, bought by KKR in 2018. Trying to undercut these staffing giants is American Physician Partners, a rapidly expanding company that runs EDs in at least 17 states and is 50% owned by private equity firm BBH Capital Partners.
These staffing companies have been among the most aggressive in replacing doctors to cut costs, said Robert McNamara, MD, a founder of the American Academy of Emergency Medicine and chair of emergency medicine at Temple University, Philadelphia.
“It’s a relatively simple equation,” Dr. McNamara said. “Their No. 1 expense is the board-certified emergency physician. So they are going to want to keep that expense as low as possible.”
Not everyone sees the trend of private equity in ED staffing in a negative light. Jennifer Orozco, president of the American Academy of Physician Associates, which represents physician assistants, said even if the change – to use more nonphysician providers – is driven by the staffing firms’ desire to make more money, patients are still well served by a team approach that includes nurse practitioners and physician assistants.
“Though I see that shift, it’s not about profits at the end of the day,” Ms. Orozco said. “It’s about the patient.”
The “shift” is nearly invisible to patients because hospitals rarely promote branding from their ED staffing firms and there is little public documentation of private equity investments.
Arthur Smolensky, MD, a Tennessee emergency medicine specialist attempting to measure private equity’s intrusion into EDs, said his review of hospital job postings and employment contracts in 14 major metropolitan areas found that 43% of ED patients were seen in EDs staffed by companies with nonphysician owners, nearly all of whom are private equity investors.
Dr. Smolensky hopes to publish his full study, expanding to 55 metro areas, later this year. But this research will merely quantify what many doctors already know: The ED has changed. Demoralized by an increased focus on profit, and wary of a looming surplus of emergency medicine residents because there are fewer jobs to fill, many experienced doctors are leaving the ED on their own, he said.
“Most of us didn’t go into medicine to supervise an army of people that are not as well trained as we are,” Dr. Smolensky said. “We want to take care of patients.”
“I guess we’re the first guinea pigs for our ER”
Joshua Allen, a nurse practitioner at a small Kentucky hospital, snaked a rubber hose through a rack of pork ribs to practice inserting a chest tube to fix a collapsed lung.
It was 2020, and American Physician Partners was restructuring the ED where Mr. Allen worked, reducing shifts from two doctors to one. Once Mr. Allen had placed 10 tubes under a doctor’s supervision, he would be allowed to do it on his own.
“I guess we’re the first guinea pigs for our ER,” he said. “If we do have a major trauma and multiple victims come in, there’s only one doctor there. ... We need to be prepared.”
Mr. Allen is one of many midlevel practitioners finding work in emergency departments. Nurse practitioners and physician assistants are among the fastest-growing occupations in the nation, according to the U.S. Bureau of Labor Statistics.
Generally, they have master’s degrees and receive several years of specialized schooling but have significantly less training than doctors. Many are permitted to diagnose patients and prescribe medication with little or no supervision from a doctor, although limitations vary by state.
The Neiman Institute found that the share of ED visits in which a midlevel practitioner was the main clinician increased by more than 172% between 2005 and 2020. Another study, in the Journal of Emergency Medicine, reported that if trends continue there may be equal numbers of midlevel practitioners and doctors in EDs by 2030.
There is little mystery as to why. Federal data shows emergency medicine doctors are paid about $310,000 a year on average, while nurse practitioners and physician assistants earn less than $120,000. Generally, hospitals can bill for care by a midlevel practitioner at 85% the rate of a doctor while paying them less than half as much.
Private equity can make millions in the gap.
For example, Envision once encouraged EDs to employ “the least expensive resource” and treat up to 35% of patients with midlevel practitioners, according to a 2017 PowerPoint presentation. The presentation drew scorn on social media and disappeared from Envision’s website.
Envision declined a request for a phone interview. In a written statement to KHN, spokesperson Aliese Polk said the company does not direct its physician leaders on how to care for patients and called the presentation a “concept guide” that does not represent current views.
American Physician Partners touted roughly the same staffing strategy in 2021 in response to the No Surprises Act, which threatened the company’s profits by outlawing surprise medical bills. In its confidential pitch to lenders, the company estimated it could cut almost $6 million by shifting more staffing from physicians to midlevel practitioners.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
New report says suicide rates rising among young Black people
Significant increases in suicide occurred among Native American, Black and Hispanic people, with a startling rise among young Black people. Meanwhile, the rate of suicide among older people declined between 2018 and 2021, the Centers for Disease Control and Prevention has reported.
In 2021, 48,183 people died by suicide in the United States, which equates to a suicide rate of 14.1 per 100,000 people. That level equals the 2018 suicide rate, which had seen a peak that was followed by declines associated with the pandemic.
Experts said rebounding suicide rates are common following times of crisis, such as the COVID-19 pandemic. Suicide declines have also occurred during times of war and natural disaster, when psychological resilience tends to increase and people work together to overcome shared adversity.
“That will wane, and then you will see rebounding in suicide rates. That is, in fact, what we feared would happen. And it has happened, at least in 2021,” Christine Moutier, MD, chief medical officer of the American Foundation for Suicide Prevention, told the New York Times.
The new CDC report found that the largest increase was among Black people aged 10-24 years, who experienced a 36.6% increase in suicide rate between 2018 and 2021. While Black people experience mental illness at the same rates as that of the general population, historically they have disproportionately limited access to mental health care, according to the American Psychiatric Association.
CDC report authors noted that some of the biggest increases in suicide rates occurred among groups most affected by the pandemic.
From 2018 to 2021, the suicide rate for people aged 25-44 increased among Native Americans by 33.7% and among Black people by 22.9%. Suicide increased among multiracial people by 20.6% and among Hispanic or Latinx people by 19.4%. Among White people of all ages, the suicide rate declined or remained steady.
“As the nation continues to respond to the short- and long-term impacts of the COVID-19 pandemic, remaining vigilant in prevention efforts is critical, especially among disproportionately affected populations where longer-term impacts might compound preexisting inequities in suicide risk,” the CDC researchers wrote.
A version of this article first appeared on WebMD.com.
Significant increases in suicide occurred among Native American, Black and Hispanic people, with a startling rise among young Black people. Meanwhile, the rate of suicide among older people declined between 2018 and 2021, the Centers for Disease Control and Prevention has reported.
In 2021, 48,183 people died by suicide in the United States, which equates to a suicide rate of 14.1 per 100,000 people. That level equals the 2018 suicide rate, which had seen a peak that was followed by declines associated with the pandemic.
Experts said rebounding suicide rates are common following times of crisis, such as the COVID-19 pandemic. Suicide declines have also occurred during times of war and natural disaster, when psychological resilience tends to increase and people work together to overcome shared adversity.
“That will wane, and then you will see rebounding in suicide rates. That is, in fact, what we feared would happen. And it has happened, at least in 2021,” Christine Moutier, MD, chief medical officer of the American Foundation for Suicide Prevention, told the New York Times.
The new CDC report found that the largest increase was among Black people aged 10-24 years, who experienced a 36.6% increase in suicide rate between 2018 and 2021. While Black people experience mental illness at the same rates as that of the general population, historically they have disproportionately limited access to mental health care, according to the American Psychiatric Association.
CDC report authors noted that some of the biggest increases in suicide rates occurred among groups most affected by the pandemic.
From 2018 to 2021, the suicide rate for people aged 25-44 increased among Native Americans by 33.7% and among Black people by 22.9%. Suicide increased among multiracial people by 20.6% and among Hispanic or Latinx people by 19.4%. Among White people of all ages, the suicide rate declined or remained steady.
“As the nation continues to respond to the short- and long-term impacts of the COVID-19 pandemic, remaining vigilant in prevention efforts is critical, especially among disproportionately affected populations where longer-term impacts might compound preexisting inequities in suicide risk,” the CDC researchers wrote.
A version of this article first appeared on WebMD.com.
Significant increases in suicide occurred among Native American, Black and Hispanic people, with a startling rise among young Black people. Meanwhile, the rate of suicide among older people declined between 2018 and 2021, the Centers for Disease Control and Prevention has reported.
In 2021, 48,183 people died by suicide in the United States, which equates to a suicide rate of 14.1 per 100,000 people. That level equals the 2018 suicide rate, which had seen a peak that was followed by declines associated with the pandemic.
Experts said rebounding suicide rates are common following times of crisis, such as the COVID-19 pandemic. Suicide declines have also occurred during times of war and natural disaster, when psychological resilience tends to increase and people work together to overcome shared adversity.
“That will wane, and then you will see rebounding in suicide rates. That is, in fact, what we feared would happen. And it has happened, at least in 2021,” Christine Moutier, MD, chief medical officer of the American Foundation for Suicide Prevention, told the New York Times.
The new CDC report found that the largest increase was among Black people aged 10-24 years, who experienced a 36.6% increase in suicide rate between 2018 and 2021. While Black people experience mental illness at the same rates as that of the general population, historically they have disproportionately limited access to mental health care, according to the American Psychiatric Association.
CDC report authors noted that some of the biggest increases in suicide rates occurred among groups most affected by the pandemic.
From 2018 to 2021, the suicide rate for people aged 25-44 increased among Native Americans by 33.7% and among Black people by 22.9%. Suicide increased among multiracial people by 20.6% and among Hispanic or Latinx people by 19.4%. Among White people of all ages, the suicide rate declined or remained steady.
“As the nation continues to respond to the short- and long-term impacts of the COVID-19 pandemic, remaining vigilant in prevention efforts is critical, especially among disproportionately affected populations where longer-term impacts might compound preexisting inequities in suicide risk,” the CDC researchers wrote.
A version of this article first appeared on WebMD.com.
Forced hospitalization for mental illness not a permanent solution
I met Eleanor when I was writing a book on involuntary psychiatric treatment. She was very ill when she presented to an emergency department in Northern California. She was looking for help and would have signed herself in, but after waiting 8 hours with no food or medical attention, she walked out and went to another hospital.
At this point, she was agitated and distressed and began screaming uncontrollably. The physician in the second ED did not offer her the option of signing in, and she was placed on a 72-hour hold and subsequently held in the hospital for 3 weeks after a judge committed her.
Like so many issues, involuntary psychiatric care is highly polarized. Some groups favor legislation to make involuntary treatment easier, while patient advocacy and civil rights groups vehemently oppose such legislation.
We don’t hear from these combatants as much as we hear from those who trumpet their views on abortion or gun control, yet this battlefield exists. It is not surprising that when New York City Mayor Eric Adams announced a plan to hospitalize homeless people with mental illnesses – involuntarily if necessary, and at the discretion of the police – people were outraged.
New York City is not the only place using this strategy to address the problem of mental illness and homelessness; California has enacted similar legislation, and every major city has homeless citizens.
Eleanor was not homeless, and fortunately, she recovered and returned to her family. However, she remained distressed and traumatized by her hospitalization for years. “It sticks with you,” she told me. “I would rather die than go in again.”
I wish I could tell you that Eleanor is unique in saying that she would rather die than go to a hospital unit for treatment, but it is not an uncommon sentiment for patients. Some people who are charged with crimes and end up in the judicial system will opt to go to jail rather than to a psychiatric hospital. It is also not easy to access outpatient psychiatric treatment.
Barriers to care
Many psychiatrists don’t participate with insurance networks, and publicly funded clinics may have long waiting lists, so illnesses escalate until there is a crisis and hospitalization is necessary. For many, stigma and fear of potential professional repercussions are significant barriers to care.
What are the issues that legislation attempts to address? The first is the standard for hospitalizing individuals against their will. In some states, the patient must be dangerous, while in others there is a lower standard of “gravely disabled,” and finally there are those that promote a standard of a “need for treatment.”
The second is related to medicating people against their will, a process that can be rightly perceived as an assault if the patient refuses to take oral medications and must be held down for injections. Next, the use of outpatient civil commitment – legally requiring people to get treatment if they are not in the hospital – has been increasingly invoked as a way to prevent mass murders and random violence against strangers.
All but four states have some legislation for outpatient commitment, euphemistically called Assisted Outpatient Treatment (AOT), yet these laws are difficult to enforce and expensive to enact. They are also not fully effective.
In New York City, Kendra’s Law has not eliminated subway violence by people with psychiatric disturbances, and the shooter who killed 32 people and wounded 17 others at Virginia Tech in 2007 had previously been ordered by a judge to go to outpatient treatment, but he simply never showed up for his appointment.
Finally, the battle includes the right of patients to refuse to have their psychiatric information released to their caretakers under the Health Insurance Portability and Accountability Act of 1996 – a measure that many families believe would help them to get loved ones to take medications and go to appointments.
The concern about how to negotiate the needs of society and the civil rights of people with psychiatric disorders has been with us for centuries. There is a strong antipsychiatry movement that asserts that psychotropic medications are ineffective or harmful and refers to patients as “psychiatric survivors.” We value the right to medical autonomy, and when there is controversy over the validity of a treatment, there is even more controversy over forcing it upon people.
Psychiatric medications are very effective and benefit many people, but they don’t help everyone, and some people experience side effects. Also, we can’t deny that involuntary care can go wrong; the conservatorship of Britney Spears for 13 years is a very public example.
Multiple stakeholders
Many have a stake in how this plays out. There are the patients, who may be suffering and unable to recognize that they are ill, who may have valid reasons for not wanting the treatments, and who ideally should have the right to refuse care.
There are the families who watch their loved ones suffer, deteriorate, and miss the opportunities that life has to offer; who do not want their children to be homeless or incarcerated; and who may be at risk from violent behavior.
There are the mental health professionals who want to do what’s in the best interest of their patients while following legal and ethical mandates, who worry about being sued for tragic outcomes, and who can’t meet the current demand for services.
There is the taxpayer who foots the bill for disability payments, lost productivity, and institutionalization. There is our society that worries that people with psychiatric disorders will commit random acts of violence.
Finally, there are the insurers, who want to pay for as little care as possible and throw up constant hurdles in the treatment process. We must acknowledge that resources used for involuntary treatment are diverted away from those who want care.
Eleanor had many advantages that unhoused people don’t have: a supportive family, health insurance, and the financial means to pay a psychiatrist who respected her wishes to wean off her medications. She returned to a comfortable home and to personal and occupational success.
It is tragic that we have people living on the streets because of a psychiatric disorder, addiction, poverty, or some combination of these. No one should be unhoused. If the rationale of hospitalization is to decrease violence, I am not hopeful. The Epidemiologic Catchment Area study shows that people with psychiatric disorders are responsible for only 4% of all violence.
The logistics of determining which people living on the streets have psychiatric disorders, transporting them safely to medical facilities, and then finding the resources to provide for compassionate and thoughtful care in meaningful and sustained ways are very challenging.
If we don’t want people living on the streets, we need to create supports, including infrastructure to facilitate housing, access to mental health care, and addiction treatment before we resort to involuntary hospitalization.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
I met Eleanor when I was writing a book on involuntary psychiatric treatment. She was very ill when she presented to an emergency department in Northern California. She was looking for help and would have signed herself in, but after waiting 8 hours with no food or medical attention, she walked out and went to another hospital.
At this point, she was agitated and distressed and began screaming uncontrollably. The physician in the second ED did not offer her the option of signing in, and she was placed on a 72-hour hold and subsequently held in the hospital for 3 weeks after a judge committed her.
Like so many issues, involuntary psychiatric care is highly polarized. Some groups favor legislation to make involuntary treatment easier, while patient advocacy and civil rights groups vehemently oppose such legislation.
We don’t hear from these combatants as much as we hear from those who trumpet their views on abortion or gun control, yet this battlefield exists. It is not surprising that when New York City Mayor Eric Adams announced a plan to hospitalize homeless people with mental illnesses – involuntarily if necessary, and at the discretion of the police – people were outraged.
New York City is not the only place using this strategy to address the problem of mental illness and homelessness; California has enacted similar legislation, and every major city has homeless citizens.
Eleanor was not homeless, and fortunately, she recovered and returned to her family. However, she remained distressed and traumatized by her hospitalization for years. “It sticks with you,” she told me. “I would rather die than go in again.”
I wish I could tell you that Eleanor is unique in saying that she would rather die than go to a hospital unit for treatment, but it is not an uncommon sentiment for patients. Some people who are charged with crimes and end up in the judicial system will opt to go to jail rather than to a psychiatric hospital. It is also not easy to access outpatient psychiatric treatment.
Barriers to care
Many psychiatrists don’t participate with insurance networks, and publicly funded clinics may have long waiting lists, so illnesses escalate until there is a crisis and hospitalization is necessary. For many, stigma and fear of potential professional repercussions are significant barriers to care.
What are the issues that legislation attempts to address? The first is the standard for hospitalizing individuals against their will. In some states, the patient must be dangerous, while in others there is a lower standard of “gravely disabled,” and finally there are those that promote a standard of a “need for treatment.”
The second is related to medicating people against their will, a process that can be rightly perceived as an assault if the patient refuses to take oral medications and must be held down for injections. Next, the use of outpatient civil commitment – legally requiring people to get treatment if they are not in the hospital – has been increasingly invoked as a way to prevent mass murders and random violence against strangers.
All but four states have some legislation for outpatient commitment, euphemistically called Assisted Outpatient Treatment (AOT), yet these laws are difficult to enforce and expensive to enact. They are also not fully effective.
In New York City, Kendra’s Law has not eliminated subway violence by people with psychiatric disturbances, and the shooter who killed 32 people and wounded 17 others at Virginia Tech in 2007 had previously been ordered by a judge to go to outpatient treatment, but he simply never showed up for his appointment.
Finally, the battle includes the right of patients to refuse to have their psychiatric information released to their caretakers under the Health Insurance Portability and Accountability Act of 1996 – a measure that many families believe would help them to get loved ones to take medications and go to appointments.
The concern about how to negotiate the needs of society and the civil rights of people with psychiatric disorders has been with us for centuries. There is a strong antipsychiatry movement that asserts that psychotropic medications are ineffective or harmful and refers to patients as “psychiatric survivors.” We value the right to medical autonomy, and when there is controversy over the validity of a treatment, there is even more controversy over forcing it upon people.
Psychiatric medications are very effective and benefit many people, but they don’t help everyone, and some people experience side effects. Also, we can’t deny that involuntary care can go wrong; the conservatorship of Britney Spears for 13 years is a very public example.
Multiple stakeholders
Many have a stake in how this plays out. There are the patients, who may be suffering and unable to recognize that they are ill, who may have valid reasons for not wanting the treatments, and who ideally should have the right to refuse care.
There are the families who watch their loved ones suffer, deteriorate, and miss the opportunities that life has to offer; who do not want their children to be homeless or incarcerated; and who may be at risk from violent behavior.
There are the mental health professionals who want to do what’s in the best interest of their patients while following legal and ethical mandates, who worry about being sued for tragic outcomes, and who can’t meet the current demand for services.
There is the taxpayer who foots the bill for disability payments, lost productivity, and institutionalization. There is our society that worries that people with psychiatric disorders will commit random acts of violence.
Finally, there are the insurers, who want to pay for as little care as possible and throw up constant hurdles in the treatment process. We must acknowledge that resources used for involuntary treatment are diverted away from those who want care.
Eleanor had many advantages that unhoused people don’t have: a supportive family, health insurance, and the financial means to pay a psychiatrist who respected her wishes to wean off her medications. She returned to a comfortable home and to personal and occupational success.
It is tragic that we have people living on the streets because of a psychiatric disorder, addiction, poverty, or some combination of these. No one should be unhoused. If the rationale of hospitalization is to decrease violence, I am not hopeful. The Epidemiologic Catchment Area study shows that people with psychiatric disorders are responsible for only 4% of all violence.
The logistics of determining which people living on the streets have psychiatric disorders, transporting them safely to medical facilities, and then finding the resources to provide for compassionate and thoughtful care in meaningful and sustained ways are very challenging.
If we don’t want people living on the streets, we need to create supports, including infrastructure to facilitate housing, access to mental health care, and addiction treatment before we resort to involuntary hospitalization.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
I met Eleanor when I was writing a book on involuntary psychiatric treatment. She was very ill when she presented to an emergency department in Northern California. She was looking for help and would have signed herself in, but after waiting 8 hours with no food or medical attention, she walked out and went to another hospital.
At this point, she was agitated and distressed and began screaming uncontrollably. The physician in the second ED did not offer her the option of signing in, and she was placed on a 72-hour hold and subsequently held in the hospital for 3 weeks after a judge committed her.
Like so many issues, involuntary psychiatric care is highly polarized. Some groups favor legislation to make involuntary treatment easier, while patient advocacy and civil rights groups vehemently oppose such legislation.
We don’t hear from these combatants as much as we hear from those who trumpet their views on abortion or gun control, yet this battlefield exists. It is not surprising that when New York City Mayor Eric Adams announced a plan to hospitalize homeless people with mental illnesses – involuntarily if necessary, and at the discretion of the police – people were outraged.
New York City is not the only place using this strategy to address the problem of mental illness and homelessness; California has enacted similar legislation, and every major city has homeless citizens.
Eleanor was not homeless, and fortunately, she recovered and returned to her family. However, she remained distressed and traumatized by her hospitalization for years. “It sticks with you,” she told me. “I would rather die than go in again.”
I wish I could tell you that Eleanor is unique in saying that she would rather die than go to a hospital unit for treatment, but it is not an uncommon sentiment for patients. Some people who are charged with crimes and end up in the judicial system will opt to go to jail rather than to a psychiatric hospital. It is also not easy to access outpatient psychiatric treatment.
Barriers to care
Many psychiatrists don’t participate with insurance networks, and publicly funded clinics may have long waiting lists, so illnesses escalate until there is a crisis and hospitalization is necessary. For many, stigma and fear of potential professional repercussions are significant barriers to care.
What are the issues that legislation attempts to address? The first is the standard for hospitalizing individuals against their will. In some states, the patient must be dangerous, while in others there is a lower standard of “gravely disabled,” and finally there are those that promote a standard of a “need for treatment.”
The second is related to medicating people against their will, a process that can be rightly perceived as an assault if the patient refuses to take oral medications and must be held down for injections. Next, the use of outpatient civil commitment – legally requiring people to get treatment if they are not in the hospital – has been increasingly invoked as a way to prevent mass murders and random violence against strangers.
All but four states have some legislation for outpatient commitment, euphemistically called Assisted Outpatient Treatment (AOT), yet these laws are difficult to enforce and expensive to enact. They are also not fully effective.
In New York City, Kendra’s Law has not eliminated subway violence by people with psychiatric disturbances, and the shooter who killed 32 people and wounded 17 others at Virginia Tech in 2007 had previously been ordered by a judge to go to outpatient treatment, but he simply never showed up for his appointment.
Finally, the battle includes the right of patients to refuse to have their psychiatric information released to their caretakers under the Health Insurance Portability and Accountability Act of 1996 – a measure that many families believe would help them to get loved ones to take medications and go to appointments.
The concern about how to negotiate the needs of society and the civil rights of people with psychiatric disorders has been with us for centuries. There is a strong antipsychiatry movement that asserts that psychotropic medications are ineffective or harmful and refers to patients as “psychiatric survivors.” We value the right to medical autonomy, and when there is controversy over the validity of a treatment, there is even more controversy over forcing it upon people.
Psychiatric medications are very effective and benefit many people, but they don’t help everyone, and some people experience side effects. Also, we can’t deny that involuntary care can go wrong; the conservatorship of Britney Spears for 13 years is a very public example.
Multiple stakeholders
Many have a stake in how this plays out. There are the patients, who may be suffering and unable to recognize that they are ill, who may have valid reasons for not wanting the treatments, and who ideally should have the right to refuse care.
There are the families who watch their loved ones suffer, deteriorate, and miss the opportunities that life has to offer; who do not want their children to be homeless or incarcerated; and who may be at risk from violent behavior.
There are the mental health professionals who want to do what’s in the best interest of their patients while following legal and ethical mandates, who worry about being sued for tragic outcomes, and who can’t meet the current demand for services.
There is the taxpayer who foots the bill for disability payments, lost productivity, and institutionalization. There is our society that worries that people with psychiatric disorders will commit random acts of violence.
Finally, there are the insurers, who want to pay for as little care as possible and throw up constant hurdles in the treatment process. We must acknowledge that resources used for involuntary treatment are diverted away from those who want care.
Eleanor had many advantages that unhoused people don’t have: a supportive family, health insurance, and the financial means to pay a psychiatrist who respected her wishes to wean off her medications. She returned to a comfortable home and to personal and occupational success.
It is tragic that we have people living on the streets because of a psychiatric disorder, addiction, poverty, or some combination of these. No one should be unhoused. If the rationale of hospitalization is to decrease violence, I am not hopeful. The Epidemiologic Catchment Area study shows that people with psychiatric disorders are responsible for only 4% of all violence.
The logistics of determining which people living on the streets have psychiatric disorders, transporting them safely to medical facilities, and then finding the resources to provide for compassionate and thoughtful care in meaningful and sustained ways are very challenging.
If we don’t want people living on the streets, we need to create supports, including infrastructure to facilitate housing, access to mental health care, and addiction treatment before we resort to involuntary hospitalization.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
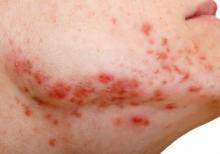
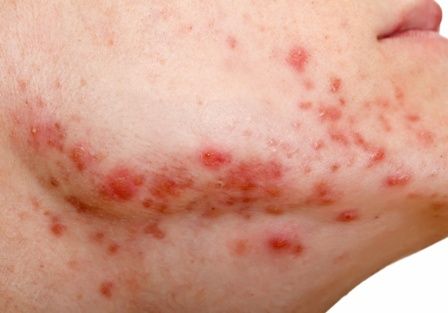
Notalgia paresthetica: Difelikefalin helps upper-back itch, but with side effects
from a randomized, double-blinded placebo-controlled trial suggest.
However, side effects were significant and caused 19% in the intervention group to discontinue the trial versus 6% in the placebo group.
Results of the study were published online in the New England Journal of Medicine.
There is currently no treatment approved by the U.S. Food and Drug Administration for the common condition, which typically causes itch in the hard-to-reach area between the shoulder blades or mid-back.
Drug reduced moderate to severe itch
Difelikefalin – a selective kappa-opioid receptor agonist – is FDA approved only as an injection for treating moderate to severe itch from chronic kidney disease in adults undergoing hemodialysis, and is marketed as Korsuva for that indication.
However, in a new trial, led by Brian S. Kim, MD, professor of dermatology and vice chair of research at the Icahn School of Medicine at Mount Sinai, New York, the drug gave moderate relief to patients with notalgia paresthetica who had moderate to severe itch.
Patients were randomly assigned 1:1 to receive oral difelikefalin 2 mg or a placebo twice daily for 8 weeks. The primary outcome was change in the weekly average of the daily 0-10 Worst Itch Numeric Rating Scale, for which 0 is “no itch” and 10 is “worst itch imaginable.”
Secondary clinical outcomes were itch-related quality-of-life and itch-related sleep measures.
The study included 126 patients; 62 received difelikefalin and 63 received placebo. One patient assigned to the difelikefalin group withdrew consent before the first dose.
The average baseline score on the Worst Itch scale was 7.6 (severe itch) in each group. Mean scores in the difelikefalin dropped by 4 points versus 2.4 points in the placebo group (95% confidence interval, −2.6 to −0.6; P = .001).
Difelikefalin did not help with sleep disturbance, compared with placebo, “except possibly in patients with impaired sleep at baseline,” the authors write. “Larger and longer trials are required to determine the effect and risks of difelikefalin treatment in this disorder.”
In a Mount Sinai press release, Dr. Kim, who is also director of the Lebwohl Center for Neuroinflammation and Sensation at Mount Sinai, called the team’s findings “encouraging.”
“The encouraging results achieved in this trial could reenergize the field and mark an important step toward improving symptoms of itch for patients with notalgia paresthetica,” he said.
Side effects ‘worrisome’
The main side effects reported included headaches, dizziness, constipation and increased urine output.
Shawn Kwatra, MD, director of the Johns Hopkins Itch Center, Baltimore, told this news organization that dizziness was “especially worrisome,” noting the average age of participants in the trial was 59-60 years. “We are very concerned about folks having falls or hip fractures,” he said.
“Things we use more commonly are topical steroids, topical capsaicin, the capsaicin patch, muscle strengthening, and gabapentin,” Dr. Kwatra said. “Off-label we use botulinum toxin (Botox) as well. I’m able to control” almost all of my notalgia paresthetica patients, he added.
In his view, for this type of drug, he said, “the right home for it is more for a generalized neuropathic pruritus or nociplastic itch vs. something very localized which is more amenable to topical therapies.”
He said that the associated central nervous system effects, such as dizziness and headache, “would limit therapeutic use to only the most severe cases in my mind.”
The trial was funded by Cara Therapeutics, manufacturer of difelikefalin.
Dr. Kim and coauthor Mark Lebwohl, MD, are paid consultants/advisers to Cara Therapeutics. Other coauthors also reported ties to Cara. Dr. Kwatra previously had done consulting work for Cara Therapeutics and is an advisory board member/consultant for AbbVie, Amgen, Arcutis Biotherapeutics, Aslan Pharmaceuticals, Castle Biosciences, Celldex Therapeutics, Galderma, Genzada Pharmaceuticals, Incyte Corporation, Johnson & Johnson, Leo Pharma, Novartis Pharmaceuticals Corporation, Pfizer, Regeneron Pharmaceuticals, and Sanofi and has served as an investigator for Galderma, Incyte, Pfizer, and Sanofi.
A version of this article first appeared on Medscape.com.
from a randomized, double-blinded placebo-controlled trial suggest.
However, side effects were significant and caused 19% in the intervention group to discontinue the trial versus 6% in the placebo group.
Results of the study were published online in the New England Journal of Medicine.
There is currently no treatment approved by the U.S. Food and Drug Administration for the common condition, which typically causes itch in the hard-to-reach area between the shoulder blades or mid-back.
Drug reduced moderate to severe itch
Difelikefalin – a selective kappa-opioid receptor agonist – is FDA approved only as an injection for treating moderate to severe itch from chronic kidney disease in adults undergoing hemodialysis, and is marketed as Korsuva for that indication.
However, in a new trial, led by Brian S. Kim, MD, professor of dermatology and vice chair of research at the Icahn School of Medicine at Mount Sinai, New York, the drug gave moderate relief to patients with notalgia paresthetica who had moderate to severe itch.
Patients were randomly assigned 1:1 to receive oral difelikefalin 2 mg or a placebo twice daily for 8 weeks. The primary outcome was change in the weekly average of the daily 0-10 Worst Itch Numeric Rating Scale, for which 0 is “no itch” and 10 is “worst itch imaginable.”
Secondary clinical outcomes were itch-related quality-of-life and itch-related sleep measures.
The study included 126 patients; 62 received difelikefalin and 63 received placebo. One patient assigned to the difelikefalin group withdrew consent before the first dose.
The average baseline score on the Worst Itch scale was 7.6 (severe itch) in each group. Mean scores in the difelikefalin dropped by 4 points versus 2.4 points in the placebo group (95% confidence interval, −2.6 to −0.6; P = .001).
Difelikefalin did not help with sleep disturbance, compared with placebo, “except possibly in patients with impaired sleep at baseline,” the authors write. “Larger and longer trials are required to determine the effect and risks of difelikefalin treatment in this disorder.”
In a Mount Sinai press release, Dr. Kim, who is also director of the Lebwohl Center for Neuroinflammation and Sensation at Mount Sinai, called the team’s findings “encouraging.”
“The encouraging results achieved in this trial could reenergize the field and mark an important step toward improving symptoms of itch for patients with notalgia paresthetica,” he said.
Side effects ‘worrisome’
The main side effects reported included headaches, dizziness, constipation and increased urine output.
Shawn Kwatra, MD, director of the Johns Hopkins Itch Center, Baltimore, told this news organization that dizziness was “especially worrisome,” noting the average age of participants in the trial was 59-60 years. “We are very concerned about folks having falls or hip fractures,” he said.
“Things we use more commonly are topical steroids, topical capsaicin, the capsaicin patch, muscle strengthening, and gabapentin,” Dr. Kwatra said. “Off-label we use botulinum toxin (Botox) as well. I’m able to control” almost all of my notalgia paresthetica patients, he added.
In his view, for this type of drug, he said, “the right home for it is more for a generalized neuropathic pruritus or nociplastic itch vs. something very localized which is more amenable to topical therapies.”
He said that the associated central nervous system effects, such as dizziness and headache, “would limit therapeutic use to only the most severe cases in my mind.”
The trial was funded by Cara Therapeutics, manufacturer of difelikefalin.
Dr. Kim and coauthor Mark Lebwohl, MD, are paid consultants/advisers to Cara Therapeutics. Other coauthors also reported ties to Cara. Dr. Kwatra previously had done consulting work for Cara Therapeutics and is an advisory board member/consultant for AbbVie, Amgen, Arcutis Biotherapeutics, Aslan Pharmaceuticals, Castle Biosciences, Celldex Therapeutics, Galderma, Genzada Pharmaceuticals, Incyte Corporation, Johnson & Johnson, Leo Pharma, Novartis Pharmaceuticals Corporation, Pfizer, Regeneron Pharmaceuticals, and Sanofi and has served as an investigator for Galderma, Incyte, Pfizer, and Sanofi.
A version of this article first appeared on Medscape.com.
from a randomized, double-blinded placebo-controlled trial suggest.
However, side effects were significant and caused 19% in the intervention group to discontinue the trial versus 6% in the placebo group.
Results of the study were published online in the New England Journal of Medicine.
There is currently no treatment approved by the U.S. Food and Drug Administration for the common condition, which typically causes itch in the hard-to-reach area between the shoulder blades or mid-back.
Drug reduced moderate to severe itch
Difelikefalin – a selective kappa-opioid receptor agonist – is FDA approved only as an injection for treating moderate to severe itch from chronic kidney disease in adults undergoing hemodialysis, and is marketed as Korsuva for that indication.
However, in a new trial, led by Brian S. Kim, MD, professor of dermatology and vice chair of research at the Icahn School of Medicine at Mount Sinai, New York, the drug gave moderate relief to patients with notalgia paresthetica who had moderate to severe itch.
Patients were randomly assigned 1:1 to receive oral difelikefalin 2 mg or a placebo twice daily for 8 weeks. The primary outcome was change in the weekly average of the daily 0-10 Worst Itch Numeric Rating Scale, for which 0 is “no itch” and 10 is “worst itch imaginable.”
Secondary clinical outcomes were itch-related quality-of-life and itch-related sleep measures.
The study included 126 patients; 62 received difelikefalin and 63 received placebo. One patient assigned to the difelikefalin group withdrew consent before the first dose.
The average baseline score on the Worst Itch scale was 7.6 (severe itch) in each group. Mean scores in the difelikefalin dropped by 4 points versus 2.4 points in the placebo group (95% confidence interval, −2.6 to −0.6; P = .001).
Difelikefalin did not help with sleep disturbance, compared with placebo, “except possibly in patients with impaired sleep at baseline,” the authors write. “Larger and longer trials are required to determine the effect and risks of difelikefalin treatment in this disorder.”
In a Mount Sinai press release, Dr. Kim, who is also director of the Lebwohl Center for Neuroinflammation and Sensation at Mount Sinai, called the team’s findings “encouraging.”
“The encouraging results achieved in this trial could reenergize the field and mark an important step toward improving symptoms of itch for patients with notalgia paresthetica,” he said.
Side effects ‘worrisome’
The main side effects reported included headaches, dizziness, constipation and increased urine output.
Shawn Kwatra, MD, director of the Johns Hopkins Itch Center, Baltimore, told this news organization that dizziness was “especially worrisome,” noting the average age of participants in the trial was 59-60 years. “We are very concerned about folks having falls or hip fractures,” he said.
“Things we use more commonly are topical steroids, topical capsaicin, the capsaicin patch, muscle strengthening, and gabapentin,” Dr. Kwatra said. “Off-label we use botulinum toxin (Botox) as well. I’m able to control” almost all of my notalgia paresthetica patients, he added.
In his view, for this type of drug, he said, “the right home for it is more for a generalized neuropathic pruritus or nociplastic itch vs. something very localized which is more amenable to topical therapies.”
He said that the associated central nervous system effects, such as dizziness and headache, “would limit therapeutic use to only the most severe cases in my mind.”
The trial was funded by Cara Therapeutics, manufacturer of difelikefalin.
Dr. Kim and coauthor Mark Lebwohl, MD, are paid consultants/advisers to Cara Therapeutics. Other coauthors also reported ties to Cara. Dr. Kwatra previously had done consulting work for Cara Therapeutics and is an advisory board member/consultant for AbbVie, Amgen, Arcutis Biotherapeutics, Aslan Pharmaceuticals, Castle Biosciences, Celldex Therapeutics, Galderma, Genzada Pharmaceuticals, Incyte Corporation, Johnson & Johnson, Leo Pharma, Novartis Pharmaceuticals Corporation, Pfizer, Regeneron Pharmaceuticals, and Sanofi and has served as an investigator for Galderma, Incyte, Pfizer, and Sanofi.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Vibrating pill can help treat constipation
The drug-free solution is designed for daily use. In a trial, the pill produced at least one additional weekly bowel movement for 41% of participants, compared with at least one additional bowel movement for 23% of participants who took a placebo pill.
Vibrant was approved by the Food and Drug Administration in August but is just now becoming available for doctors to prescribe, the company announced Wednesday.
Because it is not a drug, Vibrant is considered a class 2 medical device by the FDA, which is the same class as contact lenses.
Here’s how it works: Around bedtime, the pill is inserted in a pod to activate it, then swallowed. It travels the digestive tract and reaches the large intestine about 14 hours later.
“Then it goes to work,” the company explained in a news release. “After it’s swallowed, it is active for about 2 hours, goes quiet for around 6, hours and then activates again for another 2 hours.”
“There are little vibrations for 3 seconds on, 3 seconds off,” said Cathy Collis, chief commercial officer for Israel-based Vibrant Gastro, in a statement.
The vibrations help trigger peristalsis, the wave-like muscle contractions that move food through the gastrointestinal tract, the company said. Decreased peristalsis is a cause of constipation, which is defined as having less than three bowel movements per week, according to the Cleveland Clinic.
About 2.5 million people see their doctor each year for constipation. The pills are made of what the company called “medical-grade material” that is the same as what’s used to make gastroenterology cameras.
In the trial, most people did not report feeling the pill inside of them.
“A minority could feel it,” said Eamonn Quigley, MD, chief of gastroenterology at Houston Methodist Hospital, in a statement. “None of them felt it was being uncomfortable. And none of them stopped taking it because of that.”
Dr. Quigley helped test the capsules and does not have a financial stake in the company, according to Vibrant.
The pills do not dissolve inside a person’s body. Rather, “after they’ve done their job, the person’s body poops them out, and they’re flushed away,” the company said.
A version of this article first appeared on WebMD.com.
The drug-free solution is designed for daily use. In a trial, the pill produced at least one additional weekly bowel movement for 41% of participants, compared with at least one additional bowel movement for 23% of participants who took a placebo pill.
Vibrant was approved by the Food and Drug Administration in August but is just now becoming available for doctors to prescribe, the company announced Wednesday.
Because it is not a drug, Vibrant is considered a class 2 medical device by the FDA, which is the same class as contact lenses.
Here’s how it works: Around bedtime, the pill is inserted in a pod to activate it, then swallowed. It travels the digestive tract and reaches the large intestine about 14 hours later.
“Then it goes to work,” the company explained in a news release. “After it’s swallowed, it is active for about 2 hours, goes quiet for around 6, hours and then activates again for another 2 hours.”
“There are little vibrations for 3 seconds on, 3 seconds off,” said Cathy Collis, chief commercial officer for Israel-based Vibrant Gastro, in a statement.
The vibrations help trigger peristalsis, the wave-like muscle contractions that move food through the gastrointestinal tract, the company said. Decreased peristalsis is a cause of constipation, which is defined as having less than three bowel movements per week, according to the Cleveland Clinic.
About 2.5 million people see their doctor each year for constipation. The pills are made of what the company called “medical-grade material” that is the same as what’s used to make gastroenterology cameras.
In the trial, most people did not report feeling the pill inside of them.
“A minority could feel it,” said Eamonn Quigley, MD, chief of gastroenterology at Houston Methodist Hospital, in a statement. “None of them felt it was being uncomfortable. And none of them stopped taking it because of that.”
Dr. Quigley helped test the capsules and does not have a financial stake in the company, according to Vibrant.
The pills do not dissolve inside a person’s body. Rather, “after they’ve done their job, the person’s body poops them out, and they’re flushed away,” the company said.
A version of this article first appeared on WebMD.com.
The drug-free solution is designed for daily use. In a trial, the pill produced at least one additional weekly bowel movement for 41% of participants, compared with at least one additional bowel movement for 23% of participants who took a placebo pill.
Vibrant was approved by the Food and Drug Administration in August but is just now becoming available for doctors to prescribe, the company announced Wednesday.
Because it is not a drug, Vibrant is considered a class 2 medical device by the FDA, which is the same class as contact lenses.
Here’s how it works: Around bedtime, the pill is inserted in a pod to activate it, then swallowed. It travels the digestive tract and reaches the large intestine about 14 hours later.
“Then it goes to work,” the company explained in a news release. “After it’s swallowed, it is active for about 2 hours, goes quiet for around 6, hours and then activates again for another 2 hours.”
“There are little vibrations for 3 seconds on, 3 seconds off,” said Cathy Collis, chief commercial officer for Israel-based Vibrant Gastro, in a statement.
The vibrations help trigger peristalsis, the wave-like muscle contractions that move food through the gastrointestinal tract, the company said. Decreased peristalsis is a cause of constipation, which is defined as having less than three bowel movements per week, according to the Cleveland Clinic.
About 2.5 million people see their doctor each year for constipation. The pills are made of what the company called “medical-grade material” that is the same as what’s used to make gastroenterology cameras.
In the trial, most people did not report feeling the pill inside of them.
“A minority could feel it,” said Eamonn Quigley, MD, chief of gastroenterology at Houston Methodist Hospital, in a statement. “None of them felt it was being uncomfortable. And none of them stopped taking it because of that.”
Dr. Quigley helped test the capsules and does not have a financial stake in the company, according to Vibrant.
The pills do not dissolve inside a person’s body. Rather, “after they’ve done their job, the person’s body poops them out, and they’re flushed away,” the company said.
A version of this article first appeared on WebMD.com.
Embattled iPLEDGE program: Changes ahead?
In December 2021, major changes took effect in the iPLEDGE program, the Food and Drug Administration–required safety program for managing the risks of isotretinoin’s teratogenicity and preventing exposure during pregnancy. Now, more modifications may be coming to the acne drug’s safety program.
The risk evaluation and mitigation strategy (REMS) requirements. The aim, according to the FDA meeting announcement, is “to minimize burden on patients, pharmacies, and prescribers while maintaining safe use of isotretinoin oral capsules for patients.”
Isotretinoin is marketed as Absorica, Absorica LD, Claravis, Amnesteem, Myorisan, and Zenatane. Its former brand name was Accutane.
Problems began to surface days after a new, gender-neutral approach to the risk mitigation program was launched on Dec. 13, 2021. That program had been approved earlier by the FDA.
However, the problems that were encountered were a result of glitches in changes in the platform that had been planned, and were not related to the gender-neutral changes. The iPLEDGE program had transitioned to the new platform, and the rollout was far from smooth. Dermatologists, pharmacists, patients, parents of patients, and others were frustrated and angry that they could not access the new platform and obtain the medication promptly. Reaching the help line to sort out problems was another exercise in frustration. Wait times while on hold were unbearably long, or problems were not resolved over the phone.
(The new gender-neutral approach, which advocates said was needed to preserve inclusiveness of their patients, including transgender patients, places potential patients into two categories: those who can become pregnant, and those who cannot. Previously, there were three categories into which patients were classified: females who have reproductive potential, females who do not have reproductive potential, and males.)
Before pharmacists can fill a prescription for isotretinoin, a medical provider must confirm a patient’s negative pregnancy test and inform a patient with reproductive potential of the risks of the medication.
In January 2022, to deal with the chaotic launch and subsequent problems, the FDA said it would continue to meet with the Isotretinoin Products Manufacturers Group (IPMG) to resolve the problems reported by clinicians, pharmacists, and patients.
The American Academy of Dermatology Association formed an iPLEDGE work group to address the issues and suggest solutions. It has made several requests of and suggestions for the IPMG, which manages the program, according to Andrea L. Zaenglein, MD, professor of dermatology and pediatrics at Penn State Hershey (Pa.) Medical Center, and a member of the work group.
“We are asking them to eliminate the monthly attestation for patients who can’t get pregnant and to review and modify restrictive and punitive waiting and lockout periods for all patients,” she told this news organization.
As of February 2023, most of the platform glitches had been smoothed out, Dr. Zaenglein said. Still, “improvements to the design of the website could improve the user interface,” she added.
The FDA has established a docket for the public to submit comments before the meeting. The docket number is FDA-2022-N-3071. The electronic filing system will accept comments until 11:59 p.m. Eastern time on March 27. Background material and a link to the live webcast of the panel meeting will be available to the public no later than 2 days before the meeting and will be posted on the FDA web page or at the time of the meeting.
A version of this article first appeared on Medscape.com.
In December 2021, major changes took effect in the iPLEDGE program, the Food and Drug Administration–required safety program for managing the risks of isotretinoin’s teratogenicity and preventing exposure during pregnancy. Now, more modifications may be coming to the acne drug’s safety program.
The risk evaluation and mitigation strategy (REMS) requirements. The aim, according to the FDA meeting announcement, is “to minimize burden on patients, pharmacies, and prescribers while maintaining safe use of isotretinoin oral capsules for patients.”
Isotretinoin is marketed as Absorica, Absorica LD, Claravis, Amnesteem, Myorisan, and Zenatane. Its former brand name was Accutane.
Problems began to surface days after a new, gender-neutral approach to the risk mitigation program was launched on Dec. 13, 2021. That program had been approved earlier by the FDA.
However, the problems that were encountered were a result of glitches in changes in the platform that had been planned, and were not related to the gender-neutral changes. The iPLEDGE program had transitioned to the new platform, and the rollout was far from smooth. Dermatologists, pharmacists, patients, parents of patients, and others were frustrated and angry that they could not access the new platform and obtain the medication promptly. Reaching the help line to sort out problems was another exercise in frustration. Wait times while on hold were unbearably long, or problems were not resolved over the phone.
(The new gender-neutral approach, which advocates said was needed to preserve inclusiveness of their patients, including transgender patients, places potential patients into two categories: those who can become pregnant, and those who cannot. Previously, there were three categories into which patients were classified: females who have reproductive potential, females who do not have reproductive potential, and males.)
Before pharmacists can fill a prescription for isotretinoin, a medical provider must confirm a patient’s negative pregnancy test and inform a patient with reproductive potential of the risks of the medication.
In January 2022, to deal with the chaotic launch and subsequent problems, the FDA said it would continue to meet with the Isotretinoin Products Manufacturers Group (IPMG) to resolve the problems reported by clinicians, pharmacists, and patients.
The American Academy of Dermatology Association formed an iPLEDGE work group to address the issues and suggest solutions. It has made several requests of and suggestions for the IPMG, which manages the program, according to Andrea L. Zaenglein, MD, professor of dermatology and pediatrics at Penn State Hershey (Pa.) Medical Center, and a member of the work group.
“We are asking them to eliminate the monthly attestation for patients who can’t get pregnant and to review and modify restrictive and punitive waiting and lockout periods for all patients,” she told this news organization.
As of February 2023, most of the platform glitches had been smoothed out, Dr. Zaenglein said. Still, “improvements to the design of the website could improve the user interface,” she added.
The FDA has established a docket for the public to submit comments before the meeting. The docket number is FDA-2022-N-3071. The electronic filing system will accept comments until 11:59 p.m. Eastern time on March 27. Background material and a link to the live webcast of the panel meeting will be available to the public no later than 2 days before the meeting and will be posted on the FDA web page or at the time of the meeting.
A version of this article first appeared on Medscape.com.
In December 2021, major changes took effect in the iPLEDGE program, the Food and Drug Administration–required safety program for managing the risks of isotretinoin’s teratogenicity and preventing exposure during pregnancy. Now, more modifications may be coming to the acne drug’s safety program.
The risk evaluation and mitigation strategy (REMS) requirements. The aim, according to the FDA meeting announcement, is “to minimize burden on patients, pharmacies, and prescribers while maintaining safe use of isotretinoin oral capsules for patients.”
Isotretinoin is marketed as Absorica, Absorica LD, Claravis, Amnesteem, Myorisan, and Zenatane. Its former brand name was Accutane.
Problems began to surface days after a new, gender-neutral approach to the risk mitigation program was launched on Dec. 13, 2021. That program had been approved earlier by the FDA.
However, the problems that were encountered were a result of glitches in changes in the platform that had been planned, and were not related to the gender-neutral changes. The iPLEDGE program had transitioned to the new platform, and the rollout was far from smooth. Dermatologists, pharmacists, patients, parents of patients, and others were frustrated and angry that they could not access the new platform and obtain the medication promptly. Reaching the help line to sort out problems was another exercise in frustration. Wait times while on hold were unbearably long, or problems were not resolved over the phone.
(The new gender-neutral approach, which advocates said was needed to preserve inclusiveness of their patients, including transgender patients, places potential patients into two categories: those who can become pregnant, and those who cannot. Previously, there were three categories into which patients were classified: females who have reproductive potential, females who do not have reproductive potential, and males.)
Before pharmacists can fill a prescription for isotretinoin, a medical provider must confirm a patient’s negative pregnancy test and inform a patient with reproductive potential of the risks of the medication.
In January 2022, to deal with the chaotic launch and subsequent problems, the FDA said it would continue to meet with the Isotretinoin Products Manufacturers Group (IPMG) to resolve the problems reported by clinicians, pharmacists, and patients.
The American Academy of Dermatology Association formed an iPLEDGE work group to address the issues and suggest solutions. It has made several requests of and suggestions for the IPMG, which manages the program, according to Andrea L. Zaenglein, MD, professor of dermatology and pediatrics at Penn State Hershey (Pa.) Medical Center, and a member of the work group.
“We are asking them to eliminate the monthly attestation for patients who can’t get pregnant and to review and modify restrictive and punitive waiting and lockout periods for all patients,” she told this news organization.
As of February 2023, most of the platform glitches had been smoothed out, Dr. Zaenglein said. Still, “improvements to the design of the website could improve the user interface,” she added.
The FDA has established a docket for the public to submit comments before the meeting. The docket number is FDA-2022-N-3071. The electronic filing system will accept comments until 11:59 p.m. Eastern time on March 27. Background material and a link to the live webcast of the panel meeting will be available to the public no later than 2 days before the meeting and will be posted on the FDA web page or at the time of the meeting.
A version of this article first appeared on Medscape.com.
Doc never met patient who died from insect bite, but negligence suit moves forward; more
On-call specialist incurred a clear ‘duty of care,’ court rules
a state appeals court ruled late in January.
The appeals decision is the result of a case involving the late Dennis Blagden.
On July 26, 2017, Mr. Blagden arrived at the Graham Hospital ED, in Canton, Ill., complaining of neck pain and an insect bite that had resulted in a swollen elbow. His ED doctor, Matthew McMillin, MD, who worked for Coleman Medical Associates, ordered tests and prescribed an anti-inflammatory pain medication and a muscle relaxant.
Dr. McMillin consulted via telephone with Kenneth Krock, MD, an internal medicine specialist and pediatrician, who was on call that day and who enjoyed admitting privileges at Graham. (Krock was also an employee of Coleman Medical Associates, which provided clinical staffing for the hospital.)
Dr. Krock had final admitting authority in this instance. Court records show that Dr. McMillin and he agreed that the patient could be discharged from the ED, despite Krock’s differential diagnosis indicating a possible infection.
Three days later, now with “hypercapnic respiratory failure, sepsis, and an altered mental state,” Mr. Blagden was again seen at the Graham Hospital ED. Mr. Blagden underwent intubation by Dr. McMillin, his original ED doctor, and was airlifted to Methodist Medical Center, in Peoria, 30 miles away. There, an MRI showed that he’d developed a spinal epidural abscess. On Aug. 7, 2017, a little over a week after his admission to Methodist, Mr. Blagden died from complications of his infection.
In January 2019, Mr. Blagden’s wife, Judy, filed a suit against Dr. McMillin, his practice, and Graham Hospital, which is a part of Graham Health System. Her suit alleged medical negligence in the death of her husband.
About 6 months later, Mr.s Blagden amended her original complaint, adding a second count of medical negligence against Dr. Krock; his practice and employer, Coleman Medical Associates; and Graham Hospital. In her amended complaint, Mrs. Blagden alleged that although Krock hadn’t actually seen her husband Dennis, his consultation with Dr. McMillin was sufficient to establish a doctor-patient relationship and thus a legal duty of care. That duty, Mrs. Blagden further alleged, was breached when Dr. Krock failed both to rule out her husband’s “infectious process” and to admit him for proper follow-up monitoring.
In July 2021, after the case had been transferred from Peoria County to Fulton County, Dr. Krock cried foul. In a motion to the court for summary judgment – that is, a ruling prior to an actual trial – he and his practice put forth the following argument: As a mere on-call consultant that day in 2017, he had neither seen the patient nor established a relationship with him, thereby precluding his legal duty of care.
The trial court judge agreed and granted both Dr. Krock and Dr. Coleman the summary judgment they had sought.
Mrs. Blagden then appealed to the Appellate Court of Illinois, Fourth District, which is located in Springfield.
In its unanimous decision, the three-judge panel reversed the lower court’s ruling. Taking direct aim at Dr. Krock’s earlier motion, Justice Eugene Doherty, who wrote the panel’s opinion, said that state law had long established that “the special relationship giving rise to a duty of care may exist even in the absence of any meeting between the physician and the patient where the physician performs specific services for the benefit of the patient.”
As Justice Doherty explained, Dr. Krock’s status that day as both the on-call doctor and the one with final admitting authority undermined his argument for summary judgment. Also undermining it, Justice Doherty added, was the fact that the conversation between the two doctors that day in 2017 was a formal exchange “contemplated by hospital bylaws.”
“While public policy should encourage informal consultations between physicians,” the justice continued, “it must not ignore actual physician involvement in decisions that directly affect a patient’s care.”
Following the Fourth District decision, the suit against Dr. McMillin, Dr. Krock, and the other defendants has now been tossed back to the trial court for further proceedings. At press time, no trial date had been set.
Will this proposed damages cap help retain more physicians?
Fear of a doctor shortage, triggered in part by a recent history of large payouts, has prompted Iowa lawmakers to push for new state caps on medical malpractice awards, as a story in the Des Moines Register reports.
Currently, Iowa caps most noneconomic damages – including those for pain and suffering – at $250,000, which is among the lowest such caps in the nation.
Under existing Iowa law, however, the limit doesn’t apply in extraordinary cases – that is, those involving “substantial or permanent loss of body function, substantial disfigurement, or death.” It also isn’t applicable in cases in which a jury decides that a defendant acted with intentional malice.
Lawmakers and Iowa Gov. Kim Reynolds would like to change this.
Under a Senate bill that has now passed out of committee and is awaiting debate on the Senate floor, even plaintiffs involved in extreme cases would receive no more than $1 million to compensate for their pain, suffering, or emotional distress. (The bill also includes a 2.1% annual hike to compensate for inflation. A similar bill, which adds “loss of pregnancy” to the list of extreme cases, has advanced to the House floor.)
Supporters say the proposed cap would help to limit mega awards. In Johnson County in March 2022, for instance, a jury awarded $97.4 million to the parents of a young boy who sustained severe brain injuries during his delivery, causing the clinic that had been involved in the case to file for bankruptcy. This award was nearly three times the total payouts ($35 million) in the entire state of Iowa in all of 2021, a year in which there were 192 closed claims, including at least a dozen that resulted in payouts of $1 million or more.
Supporters also think the proposed cap will mitigate what they see as a looming doctor shortage, especially among ob.gyns. in eastern Iowa. “I just cannot overstate how much this is affecting our workforce, and that turns into effects for the women and the children, the babies, in our state,” Shannon Leveridge, MD, an obstetrician in Davenport said. “In order to keep these women and their babies safe, we need doctors.”
But critics of the bill, including some lawmakers and the trial bar, say it overreaches, even in the case of the $97.4 million award.
“They don’t want to talk about the actual damages that are caused by medical negligence,” explained a spokesman for the trial lawyers. “So, you don’t hear about the fact that, of the $50 million of economic damages ... most of that is going to go to the 24/7 care for this child for the rest of his life.”
A version of this article first appeared on Medscape.com.
On-call specialist incurred a clear ‘duty of care,’ court rules
a state appeals court ruled late in January.
The appeals decision is the result of a case involving the late Dennis Blagden.
On July 26, 2017, Mr. Blagden arrived at the Graham Hospital ED, in Canton, Ill., complaining of neck pain and an insect bite that had resulted in a swollen elbow. His ED doctor, Matthew McMillin, MD, who worked for Coleman Medical Associates, ordered tests and prescribed an anti-inflammatory pain medication and a muscle relaxant.
Dr. McMillin consulted via telephone with Kenneth Krock, MD, an internal medicine specialist and pediatrician, who was on call that day and who enjoyed admitting privileges at Graham. (Krock was also an employee of Coleman Medical Associates, which provided clinical staffing for the hospital.)
Dr. Krock had final admitting authority in this instance. Court records show that Dr. McMillin and he agreed that the patient could be discharged from the ED, despite Krock’s differential diagnosis indicating a possible infection.
Three days later, now with “hypercapnic respiratory failure, sepsis, and an altered mental state,” Mr. Blagden was again seen at the Graham Hospital ED. Mr. Blagden underwent intubation by Dr. McMillin, his original ED doctor, and was airlifted to Methodist Medical Center, in Peoria, 30 miles away. There, an MRI showed that he’d developed a spinal epidural abscess. On Aug. 7, 2017, a little over a week after his admission to Methodist, Mr. Blagden died from complications of his infection.
In January 2019, Mr. Blagden’s wife, Judy, filed a suit against Dr. McMillin, his practice, and Graham Hospital, which is a part of Graham Health System. Her suit alleged medical negligence in the death of her husband.
About 6 months later, Mr.s Blagden amended her original complaint, adding a second count of medical negligence against Dr. Krock; his practice and employer, Coleman Medical Associates; and Graham Hospital. In her amended complaint, Mrs. Blagden alleged that although Krock hadn’t actually seen her husband Dennis, his consultation with Dr. McMillin was sufficient to establish a doctor-patient relationship and thus a legal duty of care. That duty, Mrs. Blagden further alleged, was breached when Dr. Krock failed both to rule out her husband’s “infectious process” and to admit him for proper follow-up monitoring.
In July 2021, after the case had been transferred from Peoria County to Fulton County, Dr. Krock cried foul. In a motion to the court for summary judgment – that is, a ruling prior to an actual trial – he and his practice put forth the following argument: As a mere on-call consultant that day in 2017, he had neither seen the patient nor established a relationship with him, thereby precluding his legal duty of care.
The trial court judge agreed and granted both Dr. Krock and Dr. Coleman the summary judgment they had sought.
Mrs. Blagden then appealed to the Appellate Court of Illinois, Fourth District, which is located in Springfield.
In its unanimous decision, the three-judge panel reversed the lower court’s ruling. Taking direct aim at Dr. Krock’s earlier motion, Justice Eugene Doherty, who wrote the panel’s opinion, said that state law had long established that “the special relationship giving rise to a duty of care may exist even in the absence of any meeting between the physician and the patient where the physician performs specific services for the benefit of the patient.”
As Justice Doherty explained, Dr. Krock’s status that day as both the on-call doctor and the one with final admitting authority undermined his argument for summary judgment. Also undermining it, Justice Doherty added, was the fact that the conversation between the two doctors that day in 2017 was a formal exchange “contemplated by hospital bylaws.”
“While public policy should encourage informal consultations between physicians,” the justice continued, “it must not ignore actual physician involvement in decisions that directly affect a patient’s care.”
Following the Fourth District decision, the suit against Dr. McMillin, Dr. Krock, and the other defendants has now been tossed back to the trial court for further proceedings. At press time, no trial date had been set.
Will this proposed damages cap help retain more physicians?
Fear of a doctor shortage, triggered in part by a recent history of large payouts, has prompted Iowa lawmakers to push for new state caps on medical malpractice awards, as a story in the Des Moines Register reports.
Currently, Iowa caps most noneconomic damages – including those for pain and suffering – at $250,000, which is among the lowest such caps in the nation.
Under existing Iowa law, however, the limit doesn’t apply in extraordinary cases – that is, those involving “substantial or permanent loss of body function, substantial disfigurement, or death.” It also isn’t applicable in cases in which a jury decides that a defendant acted with intentional malice.
Lawmakers and Iowa Gov. Kim Reynolds would like to change this.
Under a Senate bill that has now passed out of committee and is awaiting debate on the Senate floor, even plaintiffs involved in extreme cases would receive no more than $1 million to compensate for their pain, suffering, or emotional distress. (The bill also includes a 2.1% annual hike to compensate for inflation. A similar bill, which adds “loss of pregnancy” to the list of extreme cases, has advanced to the House floor.)
Supporters say the proposed cap would help to limit mega awards. In Johnson County in March 2022, for instance, a jury awarded $97.4 million to the parents of a young boy who sustained severe brain injuries during his delivery, causing the clinic that had been involved in the case to file for bankruptcy. This award was nearly three times the total payouts ($35 million) in the entire state of Iowa in all of 2021, a year in which there were 192 closed claims, including at least a dozen that resulted in payouts of $1 million or more.
Supporters also think the proposed cap will mitigate what they see as a looming doctor shortage, especially among ob.gyns. in eastern Iowa. “I just cannot overstate how much this is affecting our workforce, and that turns into effects for the women and the children, the babies, in our state,” Shannon Leveridge, MD, an obstetrician in Davenport said. “In order to keep these women and their babies safe, we need doctors.”
But critics of the bill, including some lawmakers and the trial bar, say it overreaches, even in the case of the $97.4 million award.
“They don’t want to talk about the actual damages that are caused by medical negligence,” explained a spokesman for the trial lawyers. “So, you don’t hear about the fact that, of the $50 million of economic damages ... most of that is going to go to the 24/7 care for this child for the rest of his life.”
A version of this article first appeared on Medscape.com.
On-call specialist incurred a clear ‘duty of care,’ court rules
a state appeals court ruled late in January.
The appeals decision is the result of a case involving the late Dennis Blagden.
On July 26, 2017, Mr. Blagden arrived at the Graham Hospital ED, in Canton, Ill., complaining of neck pain and an insect bite that had resulted in a swollen elbow. His ED doctor, Matthew McMillin, MD, who worked for Coleman Medical Associates, ordered tests and prescribed an anti-inflammatory pain medication and a muscle relaxant.
Dr. McMillin consulted via telephone with Kenneth Krock, MD, an internal medicine specialist and pediatrician, who was on call that day and who enjoyed admitting privileges at Graham. (Krock was also an employee of Coleman Medical Associates, which provided clinical staffing for the hospital.)
Dr. Krock had final admitting authority in this instance. Court records show that Dr. McMillin and he agreed that the patient could be discharged from the ED, despite Krock’s differential diagnosis indicating a possible infection.
Three days later, now with “hypercapnic respiratory failure, sepsis, and an altered mental state,” Mr. Blagden was again seen at the Graham Hospital ED. Mr. Blagden underwent intubation by Dr. McMillin, his original ED doctor, and was airlifted to Methodist Medical Center, in Peoria, 30 miles away. There, an MRI showed that he’d developed a spinal epidural abscess. On Aug. 7, 2017, a little over a week after his admission to Methodist, Mr. Blagden died from complications of his infection.
In January 2019, Mr. Blagden’s wife, Judy, filed a suit against Dr. McMillin, his practice, and Graham Hospital, which is a part of Graham Health System. Her suit alleged medical negligence in the death of her husband.
About 6 months later, Mr.s Blagden amended her original complaint, adding a second count of medical negligence against Dr. Krock; his practice and employer, Coleman Medical Associates; and Graham Hospital. In her amended complaint, Mrs. Blagden alleged that although Krock hadn’t actually seen her husband Dennis, his consultation with Dr. McMillin was sufficient to establish a doctor-patient relationship and thus a legal duty of care. That duty, Mrs. Blagden further alleged, was breached when Dr. Krock failed both to rule out her husband’s “infectious process” and to admit him for proper follow-up monitoring.
In July 2021, after the case had been transferred from Peoria County to Fulton County, Dr. Krock cried foul. In a motion to the court for summary judgment – that is, a ruling prior to an actual trial – he and his practice put forth the following argument: As a mere on-call consultant that day in 2017, he had neither seen the patient nor established a relationship with him, thereby precluding his legal duty of care.
The trial court judge agreed and granted both Dr. Krock and Dr. Coleman the summary judgment they had sought.
Mrs. Blagden then appealed to the Appellate Court of Illinois, Fourth District, which is located in Springfield.
In its unanimous decision, the three-judge panel reversed the lower court’s ruling. Taking direct aim at Dr. Krock’s earlier motion, Justice Eugene Doherty, who wrote the panel’s opinion, said that state law had long established that “the special relationship giving rise to a duty of care may exist even in the absence of any meeting between the physician and the patient where the physician performs specific services for the benefit of the patient.”
As Justice Doherty explained, Dr. Krock’s status that day as both the on-call doctor and the one with final admitting authority undermined his argument for summary judgment. Also undermining it, Justice Doherty added, was the fact that the conversation between the two doctors that day in 2017 was a formal exchange “contemplated by hospital bylaws.”
“While public policy should encourage informal consultations between physicians,” the justice continued, “it must not ignore actual physician involvement in decisions that directly affect a patient’s care.”
Following the Fourth District decision, the suit against Dr. McMillin, Dr. Krock, and the other defendants has now been tossed back to the trial court for further proceedings. At press time, no trial date had been set.
Will this proposed damages cap help retain more physicians?
Fear of a doctor shortage, triggered in part by a recent history of large payouts, has prompted Iowa lawmakers to push for new state caps on medical malpractice awards, as a story in the Des Moines Register reports.
Currently, Iowa caps most noneconomic damages – including those for pain and suffering – at $250,000, which is among the lowest such caps in the nation.
Under existing Iowa law, however, the limit doesn’t apply in extraordinary cases – that is, those involving “substantial or permanent loss of body function, substantial disfigurement, or death.” It also isn’t applicable in cases in which a jury decides that a defendant acted with intentional malice.
Lawmakers and Iowa Gov. Kim Reynolds would like to change this.
Under a Senate bill that has now passed out of committee and is awaiting debate on the Senate floor, even plaintiffs involved in extreme cases would receive no more than $1 million to compensate for their pain, suffering, or emotional distress. (The bill also includes a 2.1% annual hike to compensate for inflation. A similar bill, which adds “loss of pregnancy” to the list of extreme cases, has advanced to the House floor.)
Supporters say the proposed cap would help to limit mega awards. In Johnson County in March 2022, for instance, a jury awarded $97.4 million to the parents of a young boy who sustained severe brain injuries during his delivery, causing the clinic that had been involved in the case to file for bankruptcy. This award was nearly three times the total payouts ($35 million) in the entire state of Iowa in all of 2021, a year in which there were 192 closed claims, including at least a dozen that resulted in payouts of $1 million or more.
Supporters also think the proposed cap will mitigate what they see as a looming doctor shortage, especially among ob.gyns. in eastern Iowa. “I just cannot overstate how much this is affecting our workforce, and that turns into effects for the women and the children, the babies, in our state,” Shannon Leveridge, MD, an obstetrician in Davenport said. “In order to keep these women and their babies safe, we need doctors.”
But critics of the bill, including some lawmakers and the trial bar, say it overreaches, even in the case of the $97.4 million award.
“They don’t want to talk about the actual damages that are caused by medical negligence,” explained a spokesman for the trial lawyers. “So, you don’t hear about the fact that, of the $50 million of economic damages ... most of that is going to go to the 24/7 care for this child for the rest of his life.”
A version of this article first appeared on Medscape.com.
The five biggest changes in the 2023 adult vaccine schedules
This transcript has been edited for clarity.
Hello. I’m Dr Sandra Fryhofer. Welcome to Medicine Matters.
It’s a new year, which means a new ACIP adult immunization schedule – a valuable resource collating ACIP’s most up-to-date vaccination recommendations.
Here are this year’s five most important changes:
- COVID vaccines now front and center
- New emphasis on polio vaccination
- Inclusion of some nonvaccine products (such as monoclonal antibody products)
- Pharmacists group has approved the schedule for the first time
- New shared clinical decision-making option for pneumococcal vaccines
The schedule’s organization remains the same. It still has four sections:
- Table 1: vaccinations by age
- Table 2: vaccinations by medical condition and other indications
- The Notes section (alphabetically ordered by vaccine type)
- Appendix listing of vaccine-specific contraindications and precautions
But what’s unique this year is that some of the abbreviations have historical implications. The first change is no big surprise in light of what we’ve gone through in the past few years. COVID vaccines are listed first on the cover page by brand name for those authorized and by company name for those still under US emergency use authorization. They’re also listed first on the graphics and in the notes.
COVID and mRNA and protein-based vaccines have now been assigned official abbreviations based on vaccine platform and valency.
- 1vCOV-mRNA: Comirnaty/Pfizer-BioNTech and Spikevax Moderna COVID-19 vaccines
- 2vCOV-mRNA: Pfizer-BioNTech and Moderna bivalent COVID-19 vaccines
- 1vCOV-aPS: Novavax COVID-19 vaccine
Also remarkable is the absence of COVID viral vector vaccines on the list. However, the viral vector COVID vaccine (which has been available but is not preferred) does have a CDC website link in the Notes section.
A sad but necessary inclusion was triggered by recent polio cases in New York. Polio was believed to be eradicated, and we thought adults no longer needed to be vaccinated against polio. In the new schedule, the polio vaccine is listed on the cover page but is not included in the tables. Current polio vaccination recommendations are now in the Notes section.
Also of historical significance and something that may set a precedent is the inclusion of nonvaccine products. The value of COVID preexposure prophylaxis with products including monoclonal antibodies (such as Evusheld) for people who are moderately or severely immunocompromised is mentioned in the Notes section.
For the first time ever, the schedule has been approved by the American Pharmacists Association, which validates pharmacists as established partners in vaccine administration.
Color-code key
One aspect of the schedule that has not changed is the color-code key:
- Yellow: Recommended if the patient meets the age requirement
- Purple: Indicated for those with additional risk factors or another indication
- Blue: Recommended based on shared clinical decision-making
- Orange: Precaution
- Red: Contraindicated or not recommended; the vaccine should not be administered. Overlays on the red more precisely clarify whether a vaccine is really contraindicated or just not recommended. An asterisk on red means vaccinate after pregnancy if indicated.
- Gray: No recommendation or not applicable
Vaccinations by age
Table 1 lists recommended vaccinations by age. There is one major change. COVID vaccines are on the first row of the graphic, with the need for both a primary series and boosters emphasized on the overlay. The notes have hyperlinks to the most up-to-date COVID vaccination recommendations.
Pneumococcal vaccination. Pneumococcal vaccination is routinely recommended starting at age 65. Current recommendations for those not previously vaccinated have not changed since last year. But on Table 1, the bottom half of the row for those 65 or older is now blue (and that’s new). This new color blue means shared clinical decision-making and applies to people who were previously considered fully vaccinated with the now extinct combination of PCV13 and PPSV23. These patients now have the option of getting a dose of PCV20 five years after completing their PCV13-PPSV23 combo series. This option is blue because the decision is up to you and your patient.
Check the notes for more pneumococcal vaccination details. For example, for those partially vaccinated using lower valency vaccines, there’s an option of substituting PCV20 for PPSV23 to broaden and increase durability of protection.
The pneumococcal vaccination recommendation options are complicated. A new pneumococcal vaccination app can help.
Hepatitis B. For adults under age 60, the color code for the hepatitis B vaccine is yellow, meaning it’s indicated for all. For older patients, the color code is purple. If a patient who is age 60 or older wants the hepatitis B vaccine, they can have it even in the absence of additional risk indications.
Vaccinations by medical condition or other indications
Other than a few minor word changes on the overlay, the only thing that’s new is the COVID vaccine row.
This table is helpful for matching vaccine recommendations with specific medical conditions, including pregnancy, immunocompromise, HIV (with specifics according to CD4 count), asplenia, complement deficiencies, heart disease, lung disease, alcoholism, chronic liver disease, diabetes, health care personnel, and men who have sex with men.
Use this table to dot the i’s and cross the t’s when it comes to vaccination recommendations. For example, take a look at the pregnancy column. Live virus vaccines, including LAIV, MMR, and varicella, are contraindicated and color-coded red. MMR and varicella also have an asterisk, meaning vaccinate after pregnancy if indicated. HPV vaccines are not live virus vaccines, but the overlay says they are not recommended during pregnancy. The asterisk indicates that you can vaccinate after pregnancy.
Vaccine notes
The notes are in alphabetical order, and their organization (routine, special situations, and shared clinical decision-making when indicated) has not changed. They are concise and succinct, but sometimes they’re not enough. That’s why vaccine-specific links to more complete recommendations are so convenient.
Notes for hepatitis B contain nuances on specific dosing for vaccinating patients on dialysis, as well as a reminder that newer hepatitis C vaccines such as Heplisav and PreHevbrio are not recommended during pregnancy due to lack of safety data.
For influenza, everyone 6 months or older still needs yearly flu vaccination with an age- and health-appropriate flu vaccine. But for those aged 65 or older, the notes specify the three vaccine versions now preferred: high-dose, recombinant, or adjuvanted versions. However, if these aren’t available, it’s better to get any flu vaccine than to go without.
Under meningococcal vaccines, the notes for MenACWY and MenB are combined. For MenB, trade names Bexsero and Trumenba are specified because the products are not interchangeable. Booster intervals for those still at risk are different for each vaccine type: every 5 years for MenACWY boosters, and every 2-3 years for boosts of MenB.
The recent polio cases in New York have put polio vaccination in the spotlight. ACIP has now reinstated its Polio Vaccine Work Group. The new schedule lists polio vaccines on the cover page. Current recommendations have been added to the notes section. Routine vaccination for adults is not necessary, at least for now. However, those at increased risk for exposure to polio fall in the special-situation category. For those at increased risk who have completed a polio vaccine series, a single lifetime IPV booster can be given. For those at increased risk who have not completed their polio vaccine series, now would be the time to finish the series.
Appendix
The final step in using the new schedule is checking the appendix and its list of vaccine-specific contraindications and precautions.
I hope this review of the new ACIP adult immunization schedule has been helpful. For Medicine Matters, I’m Dr. Sandra Fryhofer.
Dr. Fryhofer is clinical associate professor of medicine, Emory University, Atlanta. She reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hello. I’m Dr Sandra Fryhofer. Welcome to Medicine Matters.
It’s a new year, which means a new ACIP adult immunization schedule – a valuable resource collating ACIP’s most up-to-date vaccination recommendations.
Here are this year’s five most important changes:
- COVID vaccines now front and center
- New emphasis on polio vaccination
- Inclusion of some nonvaccine products (such as monoclonal antibody products)
- Pharmacists group has approved the schedule for the first time
- New shared clinical decision-making option for pneumococcal vaccines
The schedule’s organization remains the same. It still has four sections:
- Table 1: vaccinations by age
- Table 2: vaccinations by medical condition and other indications
- The Notes section (alphabetically ordered by vaccine type)
- Appendix listing of vaccine-specific contraindications and precautions
But what’s unique this year is that some of the abbreviations have historical implications. The first change is no big surprise in light of what we’ve gone through in the past few years. COVID vaccines are listed first on the cover page by brand name for those authorized and by company name for those still under US emergency use authorization. They’re also listed first on the graphics and in the notes.
COVID and mRNA and protein-based vaccines have now been assigned official abbreviations based on vaccine platform and valency.
- 1vCOV-mRNA: Comirnaty/Pfizer-BioNTech and Spikevax Moderna COVID-19 vaccines
- 2vCOV-mRNA: Pfizer-BioNTech and Moderna bivalent COVID-19 vaccines
- 1vCOV-aPS: Novavax COVID-19 vaccine
Also remarkable is the absence of COVID viral vector vaccines on the list. However, the viral vector COVID vaccine (which has been available but is not preferred) does have a CDC website link in the Notes section.
A sad but necessary inclusion was triggered by recent polio cases in New York. Polio was believed to be eradicated, and we thought adults no longer needed to be vaccinated against polio. In the new schedule, the polio vaccine is listed on the cover page but is not included in the tables. Current polio vaccination recommendations are now in the Notes section.
Also of historical significance and something that may set a precedent is the inclusion of nonvaccine products. The value of COVID preexposure prophylaxis with products including monoclonal antibodies (such as Evusheld) for people who are moderately or severely immunocompromised is mentioned in the Notes section.
For the first time ever, the schedule has been approved by the American Pharmacists Association, which validates pharmacists as established partners in vaccine administration.
Color-code key
One aspect of the schedule that has not changed is the color-code key:
- Yellow: Recommended if the patient meets the age requirement
- Purple: Indicated for those with additional risk factors or another indication
- Blue: Recommended based on shared clinical decision-making
- Orange: Precaution
- Red: Contraindicated or not recommended; the vaccine should not be administered. Overlays on the red more precisely clarify whether a vaccine is really contraindicated or just not recommended. An asterisk on red means vaccinate after pregnancy if indicated.
- Gray: No recommendation or not applicable
Vaccinations by age
Table 1 lists recommended vaccinations by age. There is one major change. COVID vaccines are on the first row of the graphic, with the need for both a primary series and boosters emphasized on the overlay. The notes have hyperlinks to the most up-to-date COVID vaccination recommendations.
Pneumococcal vaccination. Pneumococcal vaccination is routinely recommended starting at age 65. Current recommendations for those not previously vaccinated have not changed since last year. But on Table 1, the bottom half of the row for those 65 or older is now blue (and that’s new). This new color blue means shared clinical decision-making and applies to people who were previously considered fully vaccinated with the now extinct combination of PCV13 and PPSV23. These patients now have the option of getting a dose of PCV20 five years after completing their PCV13-PPSV23 combo series. This option is blue because the decision is up to you and your patient.
Check the notes for more pneumococcal vaccination details. For example, for those partially vaccinated using lower valency vaccines, there’s an option of substituting PCV20 for PPSV23 to broaden and increase durability of protection.
The pneumococcal vaccination recommendation options are complicated. A new pneumococcal vaccination app can help.
Hepatitis B. For adults under age 60, the color code for the hepatitis B vaccine is yellow, meaning it’s indicated for all. For older patients, the color code is purple. If a patient who is age 60 or older wants the hepatitis B vaccine, they can have it even in the absence of additional risk indications.
Vaccinations by medical condition or other indications
Other than a few minor word changes on the overlay, the only thing that’s new is the COVID vaccine row.
This table is helpful for matching vaccine recommendations with specific medical conditions, including pregnancy, immunocompromise, HIV (with specifics according to CD4 count), asplenia, complement deficiencies, heart disease, lung disease, alcoholism, chronic liver disease, diabetes, health care personnel, and men who have sex with men.
Use this table to dot the i’s and cross the t’s when it comes to vaccination recommendations. For example, take a look at the pregnancy column. Live virus vaccines, including LAIV, MMR, and varicella, are contraindicated and color-coded red. MMR and varicella also have an asterisk, meaning vaccinate after pregnancy if indicated. HPV vaccines are not live virus vaccines, but the overlay says they are not recommended during pregnancy. The asterisk indicates that you can vaccinate after pregnancy.
Vaccine notes
The notes are in alphabetical order, and their organization (routine, special situations, and shared clinical decision-making when indicated) has not changed. They are concise and succinct, but sometimes they’re not enough. That’s why vaccine-specific links to more complete recommendations are so convenient.
Notes for hepatitis B contain nuances on specific dosing for vaccinating patients on dialysis, as well as a reminder that newer hepatitis C vaccines such as Heplisav and PreHevbrio are not recommended during pregnancy due to lack of safety data.
For influenza, everyone 6 months or older still needs yearly flu vaccination with an age- and health-appropriate flu vaccine. But for those aged 65 or older, the notes specify the three vaccine versions now preferred: high-dose, recombinant, or adjuvanted versions. However, if these aren’t available, it’s better to get any flu vaccine than to go without.
Under meningococcal vaccines, the notes for MenACWY and MenB are combined. For MenB, trade names Bexsero and Trumenba are specified because the products are not interchangeable. Booster intervals for those still at risk are different for each vaccine type: every 5 years for MenACWY boosters, and every 2-3 years for boosts of MenB.
The recent polio cases in New York have put polio vaccination in the spotlight. ACIP has now reinstated its Polio Vaccine Work Group. The new schedule lists polio vaccines on the cover page. Current recommendations have been added to the notes section. Routine vaccination for adults is not necessary, at least for now. However, those at increased risk for exposure to polio fall in the special-situation category. For those at increased risk who have completed a polio vaccine series, a single lifetime IPV booster can be given. For those at increased risk who have not completed their polio vaccine series, now would be the time to finish the series.
Appendix
The final step in using the new schedule is checking the appendix and its list of vaccine-specific contraindications and precautions.
I hope this review of the new ACIP adult immunization schedule has been helpful. For Medicine Matters, I’m Dr. Sandra Fryhofer.
Dr. Fryhofer is clinical associate professor of medicine, Emory University, Atlanta. She reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hello. I’m Dr Sandra Fryhofer. Welcome to Medicine Matters.
It’s a new year, which means a new ACIP adult immunization schedule – a valuable resource collating ACIP’s most up-to-date vaccination recommendations.
Here are this year’s five most important changes:
- COVID vaccines now front and center
- New emphasis on polio vaccination
- Inclusion of some nonvaccine products (such as monoclonal antibody products)
- Pharmacists group has approved the schedule for the first time
- New shared clinical decision-making option for pneumococcal vaccines
The schedule’s organization remains the same. It still has four sections:
- Table 1: vaccinations by age
- Table 2: vaccinations by medical condition and other indications
- The Notes section (alphabetically ordered by vaccine type)
- Appendix listing of vaccine-specific contraindications and precautions
But what’s unique this year is that some of the abbreviations have historical implications. The first change is no big surprise in light of what we’ve gone through in the past few years. COVID vaccines are listed first on the cover page by brand name for those authorized and by company name for those still under US emergency use authorization. They’re also listed first on the graphics and in the notes.
COVID and mRNA and protein-based vaccines have now been assigned official abbreviations based on vaccine platform and valency.
- 1vCOV-mRNA: Comirnaty/Pfizer-BioNTech and Spikevax Moderna COVID-19 vaccines
- 2vCOV-mRNA: Pfizer-BioNTech and Moderna bivalent COVID-19 vaccines
- 1vCOV-aPS: Novavax COVID-19 vaccine
Also remarkable is the absence of COVID viral vector vaccines on the list. However, the viral vector COVID vaccine (which has been available but is not preferred) does have a CDC website link in the Notes section.
A sad but necessary inclusion was triggered by recent polio cases in New York. Polio was believed to be eradicated, and we thought adults no longer needed to be vaccinated against polio. In the new schedule, the polio vaccine is listed on the cover page but is not included in the tables. Current polio vaccination recommendations are now in the Notes section.
Also of historical significance and something that may set a precedent is the inclusion of nonvaccine products. The value of COVID preexposure prophylaxis with products including monoclonal antibodies (such as Evusheld) for people who are moderately or severely immunocompromised is mentioned in the Notes section.
For the first time ever, the schedule has been approved by the American Pharmacists Association, which validates pharmacists as established partners in vaccine administration.
Color-code key
One aspect of the schedule that has not changed is the color-code key:
- Yellow: Recommended if the patient meets the age requirement
- Purple: Indicated for those with additional risk factors or another indication
- Blue: Recommended based on shared clinical decision-making
- Orange: Precaution
- Red: Contraindicated or not recommended; the vaccine should not be administered. Overlays on the red more precisely clarify whether a vaccine is really contraindicated or just not recommended. An asterisk on red means vaccinate after pregnancy if indicated.
- Gray: No recommendation or not applicable
Vaccinations by age
Table 1 lists recommended vaccinations by age. There is one major change. COVID vaccines are on the first row of the graphic, with the need for both a primary series and boosters emphasized on the overlay. The notes have hyperlinks to the most up-to-date COVID vaccination recommendations.
Pneumococcal vaccination. Pneumococcal vaccination is routinely recommended starting at age 65. Current recommendations for those not previously vaccinated have not changed since last year. But on Table 1, the bottom half of the row for those 65 or older is now blue (and that’s new). This new color blue means shared clinical decision-making and applies to people who were previously considered fully vaccinated with the now extinct combination of PCV13 and PPSV23. These patients now have the option of getting a dose of PCV20 five years after completing their PCV13-PPSV23 combo series. This option is blue because the decision is up to you and your patient.
Check the notes for more pneumococcal vaccination details. For example, for those partially vaccinated using lower valency vaccines, there’s an option of substituting PCV20 for PPSV23 to broaden and increase durability of protection.
The pneumococcal vaccination recommendation options are complicated. A new pneumococcal vaccination app can help.
Hepatitis B. For adults under age 60, the color code for the hepatitis B vaccine is yellow, meaning it’s indicated for all. For older patients, the color code is purple. If a patient who is age 60 or older wants the hepatitis B vaccine, they can have it even in the absence of additional risk indications.
Vaccinations by medical condition or other indications
Other than a few minor word changes on the overlay, the only thing that’s new is the COVID vaccine row.
This table is helpful for matching vaccine recommendations with specific medical conditions, including pregnancy, immunocompromise, HIV (with specifics according to CD4 count), asplenia, complement deficiencies, heart disease, lung disease, alcoholism, chronic liver disease, diabetes, health care personnel, and men who have sex with men.
Use this table to dot the i’s and cross the t’s when it comes to vaccination recommendations. For example, take a look at the pregnancy column. Live virus vaccines, including LAIV, MMR, and varicella, are contraindicated and color-coded red. MMR and varicella also have an asterisk, meaning vaccinate after pregnancy if indicated. HPV vaccines are not live virus vaccines, but the overlay says they are not recommended during pregnancy. The asterisk indicates that you can vaccinate after pregnancy.
Vaccine notes
The notes are in alphabetical order, and their organization (routine, special situations, and shared clinical decision-making when indicated) has not changed. They are concise and succinct, but sometimes they’re not enough. That’s why vaccine-specific links to more complete recommendations are so convenient.
Notes for hepatitis B contain nuances on specific dosing for vaccinating patients on dialysis, as well as a reminder that newer hepatitis C vaccines such as Heplisav and PreHevbrio are not recommended during pregnancy due to lack of safety data.
For influenza, everyone 6 months or older still needs yearly flu vaccination with an age- and health-appropriate flu vaccine. But for those aged 65 or older, the notes specify the three vaccine versions now preferred: high-dose, recombinant, or adjuvanted versions. However, if these aren’t available, it’s better to get any flu vaccine than to go without.
Under meningococcal vaccines, the notes for MenACWY and MenB are combined. For MenB, trade names Bexsero and Trumenba are specified because the products are not interchangeable. Booster intervals for those still at risk are different for each vaccine type: every 5 years for MenACWY boosters, and every 2-3 years for boosts of MenB.
The recent polio cases in New York have put polio vaccination in the spotlight. ACIP has now reinstated its Polio Vaccine Work Group. The new schedule lists polio vaccines on the cover page. Current recommendations have been added to the notes section. Routine vaccination for adults is not necessary, at least for now. However, those at increased risk for exposure to polio fall in the special-situation category. For those at increased risk who have completed a polio vaccine series, a single lifetime IPV booster can be given. For those at increased risk who have not completed their polio vaccine series, now would be the time to finish the series.
Appendix
The final step in using the new schedule is checking the appendix and its list of vaccine-specific contraindications and precautions.
I hope this review of the new ACIP adult immunization schedule has been helpful. For Medicine Matters, I’m Dr. Sandra Fryhofer.
Dr. Fryhofer is clinical associate professor of medicine, Emory University, Atlanta. She reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
Weaponizing Education: The Rise, Fall, and Return of the GI Bill
Growing up I can remember my father telling stories of service members in the medical battalion he commanded in World War II (WWII) who after the war with his encouragement and their GI Bill educational benefits went to school to become doctors, nurses, and dentists. They were among the 2,300,000 veterans who attended US colleges and universities through the Servicemen’s Readjustment Act passed in 1944. The American Legion navigated the bill through the twists and turns of congressional support, and it was one of their leaders who invented the catchy GI Bill shorthand.2
As with most political legislation, there were mixed motives driving passage of the act, and like many policies in America, the primary impetus was economic. While the war was raging overseas, at home the US Department of Labor predicted that by the war’s end, 16 million service members would be jobless. Apprehensive about the prospect of yet another financial depression, in 1943 a White House agency recommended that the federal government fund education and training for the individuals who had served during the war.2
While troops stormed the beaches of Normandy, wartime President Franklin D. Roosevelt (FDR) signed the bill that delivered not only educational and training opportunities for service members and veterans, but also funded home loans and US Department of Veterans Affairs (VA) hospitals. The bill was practical in that it provided not only tuition, but also books, supplies, a living stipend, and counseling for the students. The bill technically expired in 1956, but a series of extensions and expansions has been true to the original intention to offer those who served their nation in the military a better life as citizens.
Articles describing the impact of the GI Bill use terms like life changing and transformative.3,4 Our contemporary culture makes it difficult to imagine how out of reach a college education was for the generation that fought WWII. Universities were primarily for the rich and connected, the powerful and privileged. Were it not for the upward social mobility the GI Bill propelled, the American dream would not have become a reality for many farmers, small town merchants, and factory workers. The GI Bill though could not by itself ensure equity. The systemic racism endemic in the United States and among the elected representatives who debated the bill resulted in many Black service members especially in the South being denied entrance to institutions of higher learning.5 Despite this invidious discrimination, the bill was a profound effort to help many other service members to successfully reintegrate into the society they had preserved and defended.4
“With the signing of this bill, a well-rounded program of special veterans’ benefits is nearly completed,” FDR said, capturing its noble intent: “It gives emphatic notice to the men and women in our armed forces that the American people do not intend to let them down.”6
Regrettably, we have not kept FDR’s pledge. Now unscrupulous businesses are preying on the aspirations of military personnel and veterans for an education and thwarting their ability to seek gainful employment. For more than a decade, respected news media have reported that for-profit universities were exploiting service members trying to improve their lives through obtaining a college education via the GI Bill.7 The sad irony is that what enabled the exploitation to occur was a major expansion of the benefits under the Post-9/11 GI Bill. This version granted educational funding to any individual who had served on active duty for 90 days or more after September 10, 2001.8 Federal law prohibits for-profit educational institutions from receiving more than 90% of their total revenue from federal student aid. A loophole in the law enabled these institutions to categorize GI Bill funding as private not government dollars. Bad old American greed drove these for-profit colleges and universities to aggressively recruit veterans who trusted in the good faith of the academic institutions. Once the GI Bill monies were exhausted, veterans had already invested so much time and energy in a degree or certificate, the schools could persuade them to take out student loans with the promise of job placement assistance that never materialized. They took advantage of the veterans’ hopes to fatten their own bottom line in the face of declining enrolments.9 Journalists, government, think tank reports, and even a documentary described the tragic stories of service members left unemployed with immense debt and degrees that to many of them were now worthless.10
After years of reporters exposing the scam and politically thwarted efforts to stop it, Congress and President Biden closed what was known as the 90/10 loophole. This ended the weaponization of education it had promoted. In October 2022, the US Department of Education announced its final rule to prohibit the widespread educational fraud that had betrayed so many veterans and service members, which Secretary Dennis McDonough described as “abuse.”11Some readers may wonder why I have devoted an editorial to a topic that seems somewhat distant from the health care that is the primary domain of Federal Practitioner. It happens that education is in closer proximity to health for our patients than many of us might have realized. A 2018 Military Medicine study found that veterans who took advantage of the educational opportunities of the GI Bill had better health and reduced smoking, among other benefits.12 This connection between health and education should serve as a source of pride for all of us in federal practice as we are part of organizations that affirm the holistic concept of health that embraces not just medicine but education, housing, and other services essential for comprehensive well-being.
1. Mandela NR. Lighting your way to a better future: speech delivered by Mr. N R Mandela at the launch of Mindset Network. July 16, 2003. Accessed January 23, 2023. http://db.nelsonmandela.org/speeches/pub_view.asp?pg=item&ItemID=NMS909&txtstr=Lighting%20your%20way%20to%20a%20better%20future
2. US National Archives and Records Administration. Milestones Documents: Servicemen’s Readjustment Act (1944). Updated May 3, 2022. Accessed January 23, 2023. https://www.archives.gov/milestone-documents/servicemens-readjustment-act
3. O’Brien C. A brief history of the GI Bill. Army Times. March 10, 2021. Accessed January 23, 2023. https://www.armytimes.com/education-transition/2021/03/10/a-brief-history-of-the-gi-bill
4. US Department of Defense. 75 years of the GI Bill: how transformative it’s been. June 9, 2019. Accessed January 23, 2023. https://www.defense.gov/News/Feature-Stories/story/Article/1727086/75-years-of-the-gi-bill-how-transformative-its-been
5. Thompson J. The GI Bill should’ve been race neutral, politicos made sure it wasn’t. Army Times. November 9, 2019. Accessed January 23, 2023. https://www.armytimes.com/military-honor/salute-veterans/2019/11/10/the-gi-bill-shouldve-been-race-neutral-politicos-made-sure-it-wasnt
6. US Department of Veterans Affairs. Born of controversy: the GI Bill of Rights. Accessed January 23, 2023. https://www.va.gov/opa/publications/celebrate/gi-bill.pdf
7. Lipton E. Profit and scrutiny for colleges courting veterans. New York Times. December 8, 2010. Accessed January 23, 2023. https://www.nytimes.com/2010/12/09/education/09colleges.html
8. Post-9/11 GI Bill. Accessed January 23, 2023. https://www.military.com/education/gi-bill/post-9-11
9. Veterans Education Success. Large for-profit schools remain dependent on recruiting GI Bill students despite overall enrollment declines. Veterans Perspective Brief 2018;4. Accessed January 23, 2023. https://static1.squarespace.com/static/556718b2e4b02e470eb1b186/t/5ae241e588251be6319e24a5/1524777445871/VES+Issue+Brief+%234+Enrollment.FINAL.v2.pdf
10. Hernandez K. Why these veterans regret their for-profit degrees—and debt. PBS Newshour. October 23, 2018. Accessed January 23, 2023. https://www.pbs.org/newshour/education/why-these-veterans-regret-their-for-profit-college-degrees-and-debt
11. US Department of Education. Education Department unveils final rules to protect veterans and service members, improve college access for incarcerated individuals and improve oversight when colleges change owners. Press release. Published October 27, 2022. Accessed January 23, 2023. https://www.ed.gov/news/press-releases/education-department-unveils-final-rules-protect-veterans-and-service-members-improve-college-access-incarcerated-individuals-and-improve-oversight-when-colleges-change-owners
12. Rumery ZR, Patel N, Richard P. The association between the use of the education benefits from the G.I. Bill and veterans’ health. Mil Med. 2018;183(5-6):e241-e248. doi:10.1093/milmed/usx102
Growing up I can remember my father telling stories of service members in the medical battalion he commanded in World War II (WWII) who after the war with his encouragement and their GI Bill educational benefits went to school to become doctors, nurses, and dentists. They were among the 2,300,000 veterans who attended US colleges and universities through the Servicemen’s Readjustment Act passed in 1944. The American Legion navigated the bill through the twists and turns of congressional support, and it was one of their leaders who invented the catchy GI Bill shorthand.2
As with most political legislation, there were mixed motives driving passage of the act, and like many policies in America, the primary impetus was economic. While the war was raging overseas, at home the US Department of Labor predicted that by the war’s end, 16 million service members would be jobless. Apprehensive about the prospect of yet another financial depression, in 1943 a White House agency recommended that the federal government fund education and training for the individuals who had served during the war.2
While troops stormed the beaches of Normandy, wartime President Franklin D. Roosevelt (FDR) signed the bill that delivered not only educational and training opportunities for service members and veterans, but also funded home loans and US Department of Veterans Affairs (VA) hospitals. The bill was practical in that it provided not only tuition, but also books, supplies, a living stipend, and counseling for the students. The bill technically expired in 1956, but a series of extensions and expansions has been true to the original intention to offer those who served their nation in the military a better life as citizens.
Articles describing the impact of the GI Bill use terms like life changing and transformative.3,4 Our contemporary culture makes it difficult to imagine how out of reach a college education was for the generation that fought WWII. Universities were primarily for the rich and connected, the powerful and privileged. Were it not for the upward social mobility the GI Bill propelled, the American dream would not have become a reality for many farmers, small town merchants, and factory workers. The GI Bill though could not by itself ensure equity. The systemic racism endemic in the United States and among the elected representatives who debated the bill resulted in many Black service members especially in the South being denied entrance to institutions of higher learning.5 Despite this invidious discrimination, the bill was a profound effort to help many other service members to successfully reintegrate into the society they had preserved and defended.4
“With the signing of this bill, a well-rounded program of special veterans’ benefits is nearly completed,” FDR said, capturing its noble intent: “It gives emphatic notice to the men and women in our armed forces that the American people do not intend to let them down.”6
Regrettably, we have not kept FDR’s pledge. Now unscrupulous businesses are preying on the aspirations of military personnel and veterans for an education and thwarting their ability to seek gainful employment. For more than a decade, respected news media have reported that for-profit universities were exploiting service members trying to improve their lives through obtaining a college education via the GI Bill.7 The sad irony is that what enabled the exploitation to occur was a major expansion of the benefits under the Post-9/11 GI Bill. This version granted educational funding to any individual who had served on active duty for 90 days or more after September 10, 2001.8 Federal law prohibits for-profit educational institutions from receiving more than 90% of their total revenue from federal student aid. A loophole in the law enabled these institutions to categorize GI Bill funding as private not government dollars. Bad old American greed drove these for-profit colleges and universities to aggressively recruit veterans who trusted in the good faith of the academic institutions. Once the GI Bill monies were exhausted, veterans had already invested so much time and energy in a degree or certificate, the schools could persuade them to take out student loans with the promise of job placement assistance that never materialized. They took advantage of the veterans’ hopes to fatten their own bottom line in the face of declining enrolments.9 Journalists, government, think tank reports, and even a documentary described the tragic stories of service members left unemployed with immense debt and degrees that to many of them were now worthless.10
After years of reporters exposing the scam and politically thwarted efforts to stop it, Congress and President Biden closed what was known as the 90/10 loophole. This ended the weaponization of education it had promoted. In October 2022, the US Department of Education announced its final rule to prohibit the widespread educational fraud that had betrayed so many veterans and service members, which Secretary Dennis McDonough described as “abuse.”11Some readers may wonder why I have devoted an editorial to a topic that seems somewhat distant from the health care that is the primary domain of Federal Practitioner. It happens that education is in closer proximity to health for our patients than many of us might have realized. A 2018 Military Medicine study found that veterans who took advantage of the educational opportunities of the GI Bill had better health and reduced smoking, among other benefits.12 This connection between health and education should serve as a source of pride for all of us in federal practice as we are part of organizations that affirm the holistic concept of health that embraces not just medicine but education, housing, and other services essential for comprehensive well-being.
Growing up I can remember my father telling stories of service members in the medical battalion he commanded in World War II (WWII) who after the war with his encouragement and their GI Bill educational benefits went to school to become doctors, nurses, and dentists. They were among the 2,300,000 veterans who attended US colleges and universities through the Servicemen’s Readjustment Act passed in 1944. The American Legion navigated the bill through the twists and turns of congressional support, and it was one of their leaders who invented the catchy GI Bill shorthand.2
As with most political legislation, there were mixed motives driving passage of the act, and like many policies in America, the primary impetus was economic. While the war was raging overseas, at home the US Department of Labor predicted that by the war’s end, 16 million service members would be jobless. Apprehensive about the prospect of yet another financial depression, in 1943 a White House agency recommended that the federal government fund education and training for the individuals who had served during the war.2
While troops stormed the beaches of Normandy, wartime President Franklin D. Roosevelt (FDR) signed the bill that delivered not only educational and training opportunities for service members and veterans, but also funded home loans and US Department of Veterans Affairs (VA) hospitals. The bill was practical in that it provided not only tuition, but also books, supplies, a living stipend, and counseling for the students. The bill technically expired in 1956, but a series of extensions and expansions has been true to the original intention to offer those who served their nation in the military a better life as citizens.
Articles describing the impact of the GI Bill use terms like life changing and transformative.3,4 Our contemporary culture makes it difficult to imagine how out of reach a college education was for the generation that fought WWII. Universities were primarily for the rich and connected, the powerful and privileged. Were it not for the upward social mobility the GI Bill propelled, the American dream would not have become a reality for many farmers, small town merchants, and factory workers. The GI Bill though could not by itself ensure equity. The systemic racism endemic in the United States and among the elected representatives who debated the bill resulted in many Black service members especially in the South being denied entrance to institutions of higher learning.5 Despite this invidious discrimination, the bill was a profound effort to help many other service members to successfully reintegrate into the society they had preserved and defended.4
“With the signing of this bill, a well-rounded program of special veterans’ benefits is nearly completed,” FDR said, capturing its noble intent: “It gives emphatic notice to the men and women in our armed forces that the American people do not intend to let them down.”6
Regrettably, we have not kept FDR’s pledge. Now unscrupulous businesses are preying on the aspirations of military personnel and veterans for an education and thwarting their ability to seek gainful employment. For more than a decade, respected news media have reported that for-profit universities were exploiting service members trying to improve their lives through obtaining a college education via the GI Bill.7 The sad irony is that what enabled the exploitation to occur was a major expansion of the benefits under the Post-9/11 GI Bill. This version granted educational funding to any individual who had served on active duty for 90 days or more after September 10, 2001.8 Federal law prohibits for-profit educational institutions from receiving more than 90% of their total revenue from federal student aid. A loophole in the law enabled these institutions to categorize GI Bill funding as private not government dollars. Bad old American greed drove these for-profit colleges and universities to aggressively recruit veterans who trusted in the good faith of the academic institutions. Once the GI Bill monies were exhausted, veterans had already invested so much time and energy in a degree or certificate, the schools could persuade them to take out student loans with the promise of job placement assistance that never materialized. They took advantage of the veterans’ hopes to fatten their own bottom line in the face of declining enrolments.9 Journalists, government, think tank reports, and even a documentary described the tragic stories of service members left unemployed with immense debt and degrees that to many of them were now worthless.10
After years of reporters exposing the scam and politically thwarted efforts to stop it, Congress and President Biden closed what was known as the 90/10 loophole. This ended the weaponization of education it had promoted. In October 2022, the US Department of Education announced its final rule to prohibit the widespread educational fraud that had betrayed so many veterans and service members, which Secretary Dennis McDonough described as “abuse.”11Some readers may wonder why I have devoted an editorial to a topic that seems somewhat distant from the health care that is the primary domain of Federal Practitioner. It happens that education is in closer proximity to health for our patients than many of us might have realized. A 2018 Military Medicine study found that veterans who took advantage of the educational opportunities of the GI Bill had better health and reduced smoking, among other benefits.12 This connection between health and education should serve as a source of pride for all of us in federal practice as we are part of organizations that affirm the holistic concept of health that embraces not just medicine but education, housing, and other services essential for comprehensive well-being.
1. Mandela NR. Lighting your way to a better future: speech delivered by Mr. N R Mandela at the launch of Mindset Network. July 16, 2003. Accessed January 23, 2023. http://db.nelsonmandela.org/speeches/pub_view.asp?pg=item&ItemID=NMS909&txtstr=Lighting%20your%20way%20to%20a%20better%20future
2. US National Archives and Records Administration. Milestones Documents: Servicemen’s Readjustment Act (1944). Updated May 3, 2022. Accessed January 23, 2023. https://www.archives.gov/milestone-documents/servicemens-readjustment-act
3. O’Brien C. A brief history of the GI Bill. Army Times. March 10, 2021. Accessed January 23, 2023. https://www.armytimes.com/education-transition/2021/03/10/a-brief-history-of-the-gi-bill
4. US Department of Defense. 75 years of the GI Bill: how transformative it’s been. June 9, 2019. Accessed January 23, 2023. https://www.defense.gov/News/Feature-Stories/story/Article/1727086/75-years-of-the-gi-bill-how-transformative-its-been
5. Thompson J. The GI Bill should’ve been race neutral, politicos made sure it wasn’t. Army Times. November 9, 2019. Accessed January 23, 2023. https://www.armytimes.com/military-honor/salute-veterans/2019/11/10/the-gi-bill-shouldve-been-race-neutral-politicos-made-sure-it-wasnt
6. US Department of Veterans Affairs. Born of controversy: the GI Bill of Rights. Accessed January 23, 2023. https://www.va.gov/opa/publications/celebrate/gi-bill.pdf
7. Lipton E. Profit and scrutiny for colleges courting veterans. New York Times. December 8, 2010. Accessed January 23, 2023. https://www.nytimes.com/2010/12/09/education/09colleges.html
8. Post-9/11 GI Bill. Accessed January 23, 2023. https://www.military.com/education/gi-bill/post-9-11
9. Veterans Education Success. Large for-profit schools remain dependent on recruiting GI Bill students despite overall enrollment declines. Veterans Perspective Brief 2018;4. Accessed January 23, 2023. https://static1.squarespace.com/static/556718b2e4b02e470eb1b186/t/5ae241e588251be6319e24a5/1524777445871/VES+Issue+Brief+%234+Enrollment.FINAL.v2.pdf
10. Hernandez K. Why these veterans regret their for-profit degrees—and debt. PBS Newshour. October 23, 2018. Accessed January 23, 2023. https://www.pbs.org/newshour/education/why-these-veterans-regret-their-for-profit-college-degrees-and-debt
11. US Department of Education. Education Department unveils final rules to protect veterans and service members, improve college access for incarcerated individuals and improve oversight when colleges change owners. Press release. Published October 27, 2022. Accessed January 23, 2023. https://www.ed.gov/news/press-releases/education-department-unveils-final-rules-protect-veterans-and-service-members-improve-college-access-incarcerated-individuals-and-improve-oversight-when-colleges-change-owners
12. Rumery ZR, Patel N, Richard P. The association between the use of the education benefits from the G.I. Bill and veterans’ health. Mil Med. 2018;183(5-6):e241-e248. doi:10.1093/milmed/usx102
1. Mandela NR. Lighting your way to a better future: speech delivered by Mr. N R Mandela at the launch of Mindset Network. July 16, 2003. Accessed January 23, 2023. http://db.nelsonmandela.org/speeches/pub_view.asp?pg=item&ItemID=NMS909&txtstr=Lighting%20your%20way%20to%20a%20better%20future
2. US National Archives and Records Administration. Milestones Documents: Servicemen’s Readjustment Act (1944). Updated May 3, 2022. Accessed January 23, 2023. https://www.archives.gov/milestone-documents/servicemens-readjustment-act
3. O’Brien C. A brief history of the GI Bill. Army Times. March 10, 2021. Accessed January 23, 2023. https://www.armytimes.com/education-transition/2021/03/10/a-brief-history-of-the-gi-bill
4. US Department of Defense. 75 years of the GI Bill: how transformative it’s been. June 9, 2019. Accessed January 23, 2023. https://www.defense.gov/News/Feature-Stories/story/Article/1727086/75-years-of-the-gi-bill-how-transformative-its-been
5. Thompson J. The GI Bill should’ve been race neutral, politicos made sure it wasn’t. Army Times. November 9, 2019. Accessed January 23, 2023. https://www.armytimes.com/military-honor/salute-veterans/2019/11/10/the-gi-bill-shouldve-been-race-neutral-politicos-made-sure-it-wasnt
6. US Department of Veterans Affairs. Born of controversy: the GI Bill of Rights. Accessed January 23, 2023. https://www.va.gov/opa/publications/celebrate/gi-bill.pdf
7. Lipton E. Profit and scrutiny for colleges courting veterans. New York Times. December 8, 2010. Accessed January 23, 2023. https://www.nytimes.com/2010/12/09/education/09colleges.html
8. Post-9/11 GI Bill. Accessed January 23, 2023. https://www.military.com/education/gi-bill/post-9-11
9. Veterans Education Success. Large for-profit schools remain dependent on recruiting GI Bill students despite overall enrollment declines. Veterans Perspective Brief 2018;4. Accessed January 23, 2023. https://static1.squarespace.com/static/556718b2e4b02e470eb1b186/t/5ae241e588251be6319e24a5/1524777445871/VES+Issue+Brief+%234+Enrollment.FINAL.v2.pdf
10. Hernandez K. Why these veterans regret their for-profit degrees—and debt. PBS Newshour. October 23, 2018. Accessed January 23, 2023. https://www.pbs.org/newshour/education/why-these-veterans-regret-their-for-profit-college-degrees-and-debt
11. US Department of Education. Education Department unveils final rules to protect veterans and service members, improve college access for incarcerated individuals and improve oversight when colleges change owners. Press release. Published October 27, 2022. Accessed January 23, 2023. https://www.ed.gov/news/press-releases/education-department-unveils-final-rules-protect-veterans-and-service-members-improve-college-access-incarcerated-individuals-and-improve-oversight-when-colleges-change-owners
12. Rumery ZR, Patel N, Richard P. The association between the use of the education benefits from the G.I. Bill and veterans’ health. Mil Med. 2018;183(5-6):e241-e248. doi:10.1093/milmed/usx102
Three wild technologies about to change health care
When I was a child, I watched syndicated episodes of the original “Star Trek.” I was dazzled by the space travel, sure, but also the medical technology.
A handheld “tricorder” detected diseases, while an intramuscular injector (“hypospray”) could treat them. Sickbay “biobeds” came with real-time health monitors that looked futuristic at the time but seem primitive today.
Such visions inspired a lot of us kids to pursue science. Little did we know the real-life advances many of us would see in our lifetimes.
Artificial intelligence helping to spot disease, robots performing surgery, even video calls between doctor and patient – all these once sounded fantastical but now happen in clinical care.
Now, in the 23rd year of the 21st century, you might not believe wht we’ll be capable of next. Three especially wild examples are moving closer to clinical reality.
Human hibernation
Captain America, Han Solo, and “Star Trek” villain Khan – all were preserved at low temperatures and then revived, waking up alive and well months, decades, or centuries later. These are fictional examples, to be sure, but the science they’re rooted in is real.
(In one extreme case, a climber survived after almost 9 hours of efforts to revive him.)
Useful for a space traveler? Maybe not. But it’s potentially huge for someone with life-threatening injuries from a car accident or a gunshot wound.
That’s the thinking behind a breakthrough procedure that came after decades of research on pigs and dogs, now in a clinical trial. The idea: A person with massive blood loss whose heart has stopped is injected with an ice-cold fluid, cooling them from the inside, down to about 50° F.
Doctors already induce more modest hypothermia to protect the brain and other organs after cardiac arrest and during surgery on the aortic arch (the main artery carrying blood from the heart).
But this experimental procedure – called emergency preservation and resuscitation (EPR) – goes far beyond that, dramatically “decreasing the body’s need for oxygen and blood flow,” says Samuel Tisherman, MD, a trauma surgeon at the University of Maryland Medical Center and the trial’s lead researcher. This puts the patient in a state of suspended animation that “could buy time for surgeons to stop the bleeding and save more of these patients.”
The technique has been done on at least six patients, though none were reported to survive. The trial is expected to include 20 people by the time it wraps up in December, according to the listing on the U.S. clinical trials database. Though given the strict requirements for candidates (emergency trauma victims who are not likely to survive), one can’t exactly rely on a set schedule.
Still, the technology is promising. Someday we may even use it to keep patients in suspended animation for months or years, experts predict, helping astronauts through decades-long spaceflights, or stalling death in sick patients awaiting a cure.
Artificial womb
Another sci-fi classic: growing human babies outside the womb. Think the fetus fields from “The Matrix,” or the frozen embryos in “Alien: Covenant.”
In 1923, British biologist J.B.S. Haldane coined a term for that – ectogenesis. He predicted that 70% of pregnancies would take place, from fertilization to birth, in artificial wombs by 2074. That many seems unlikely, but the timeline is on track.
Developing an embryo outside the womb is already routine in in vitro fertilization. And technology enables preterm babies to survive through much of the second half of gestation. Normal human pregnancy is 40 weeks, and the youngest preterm baby ever to survive was 21 weeks and 1 day old, just a few days younger than a smattering of others who lived.
The biggest obstacle for babies younger than that is lung viability. Mechanical ventilation can damage the lungs and lead to a chronic (sometimes fatal) lung disease known as bronchopulmonary dysplasia. Avoiding this would mean figuring out a way to maintain fetal circulation – the intricate system that delivers oxygenated blood from the placenta to the fetus via the umbilical cord. Researchers at Children’s Hospital of Philadelphia have done this using a fetal lamb.
The key to their invention is a substitute placenta: an oxygenator connected to the lamb’s umbilical cord. Tubes inserted through the umbilical vein and arteries carry oxygenated blood from the “placenta” to the fetus, and deoxygenated blood back out. The lamb resides in an artificial, fluid-filled amniotic sac until its lungs and other organs are developed.
Fertility treatment could benefit, too. “An artificial womb may substitute in situations in which a gestational carrier – surrogate – is indicated,” says Paula Amato, MD, a professor of obstetrics and gynecology at Oregon Health and Science University, Portland. (Dr. Amato is not involved in the CHOP research.) For example: when the mother is missing a uterus or can’t carry a pregnancy safely.
No date is set for clinical trials yet. But according to the research, the main difference between human and lamb may come down to size. A lamb’s umbilical vessels are larger, so feeding in a tube is easier. With today’s advances in miniaturizing surgical methods, that seems like a challenge scientists can overcome.
Messenger RNA therapeutics
Back to “Star Trek.” The hypospray injector’s contents could cure just about any disease, even one newly discovered on a strange planet. That’s not unlike messenger RNA (mRNA) technology, a breakthrough that enabled scientists to quickly develop some of the first COVID-19 vaccines.
But vaccines are just the beginning of what this technology can do.
A whole field of immunotherapy is emerging that uses mRNA to deliver instructions to produce chimeric antigen receptor–modified immune cells (CAR-modified immune cells). These cells are engineered to target diseased cells and tissues, like cancer cells and harmful fibroblasts (scar tissue) that promote fibrosis in, for example, the heart and lungs.
The field is bursting with rodent research, and clinical trials have started for treating some advanced-stage malignancies.
Actual clinical use may be years away, but if all goes well, these medicines could help treat or even cure the core medical problems facing humanity. We’re talking cancer, heart disease, neurodegenerative disease – transforming one therapy into another by simply changing the mRNA’s “nucleotide sequence,” the blueprint containing instructions telling it what to do, and what disease to attack.
As this technology matures, we may start to feel as if we’re really on “Star Trek,” where Dr. Leonard “Bones” McCoy pulls out the same device to treat just about every disease or injury.
A version of this article first appeared on WebMD.com.
When I was a child, I watched syndicated episodes of the original “Star Trek.” I was dazzled by the space travel, sure, but also the medical technology.
A handheld “tricorder” detected diseases, while an intramuscular injector (“hypospray”) could treat them. Sickbay “biobeds” came with real-time health monitors that looked futuristic at the time but seem primitive today.
Such visions inspired a lot of us kids to pursue science. Little did we know the real-life advances many of us would see in our lifetimes.
Artificial intelligence helping to spot disease, robots performing surgery, even video calls between doctor and patient – all these once sounded fantastical but now happen in clinical care.
Now, in the 23rd year of the 21st century, you might not believe wht we’ll be capable of next. Three especially wild examples are moving closer to clinical reality.
Human hibernation
Captain America, Han Solo, and “Star Trek” villain Khan – all were preserved at low temperatures and then revived, waking up alive and well months, decades, or centuries later. These are fictional examples, to be sure, but the science they’re rooted in is real.
(In one extreme case, a climber survived after almost 9 hours of efforts to revive him.)
Useful for a space traveler? Maybe not. But it’s potentially huge for someone with life-threatening injuries from a car accident or a gunshot wound.
That’s the thinking behind a breakthrough procedure that came after decades of research on pigs and dogs, now in a clinical trial. The idea: A person with massive blood loss whose heart has stopped is injected with an ice-cold fluid, cooling them from the inside, down to about 50° F.
Doctors already induce more modest hypothermia to protect the brain and other organs after cardiac arrest and during surgery on the aortic arch (the main artery carrying blood from the heart).
But this experimental procedure – called emergency preservation and resuscitation (EPR) – goes far beyond that, dramatically “decreasing the body’s need for oxygen and blood flow,” says Samuel Tisherman, MD, a trauma surgeon at the University of Maryland Medical Center and the trial’s lead researcher. This puts the patient in a state of suspended animation that “could buy time for surgeons to stop the bleeding and save more of these patients.”
The technique has been done on at least six patients, though none were reported to survive. The trial is expected to include 20 people by the time it wraps up in December, according to the listing on the U.S. clinical trials database. Though given the strict requirements for candidates (emergency trauma victims who are not likely to survive), one can’t exactly rely on a set schedule.
Still, the technology is promising. Someday we may even use it to keep patients in suspended animation for months or years, experts predict, helping astronauts through decades-long spaceflights, or stalling death in sick patients awaiting a cure.
Artificial womb
Another sci-fi classic: growing human babies outside the womb. Think the fetus fields from “The Matrix,” or the frozen embryos in “Alien: Covenant.”
In 1923, British biologist J.B.S. Haldane coined a term for that – ectogenesis. He predicted that 70% of pregnancies would take place, from fertilization to birth, in artificial wombs by 2074. That many seems unlikely, but the timeline is on track.
Developing an embryo outside the womb is already routine in in vitro fertilization. And technology enables preterm babies to survive through much of the second half of gestation. Normal human pregnancy is 40 weeks, and the youngest preterm baby ever to survive was 21 weeks and 1 day old, just a few days younger than a smattering of others who lived.
The biggest obstacle for babies younger than that is lung viability. Mechanical ventilation can damage the lungs and lead to a chronic (sometimes fatal) lung disease known as bronchopulmonary dysplasia. Avoiding this would mean figuring out a way to maintain fetal circulation – the intricate system that delivers oxygenated blood from the placenta to the fetus via the umbilical cord. Researchers at Children’s Hospital of Philadelphia have done this using a fetal lamb.
The key to their invention is a substitute placenta: an oxygenator connected to the lamb’s umbilical cord. Tubes inserted through the umbilical vein and arteries carry oxygenated blood from the “placenta” to the fetus, and deoxygenated blood back out. The lamb resides in an artificial, fluid-filled amniotic sac until its lungs and other organs are developed.
Fertility treatment could benefit, too. “An artificial womb may substitute in situations in which a gestational carrier – surrogate – is indicated,” says Paula Amato, MD, a professor of obstetrics and gynecology at Oregon Health and Science University, Portland. (Dr. Amato is not involved in the CHOP research.) For example: when the mother is missing a uterus or can’t carry a pregnancy safely.
No date is set for clinical trials yet. But according to the research, the main difference between human and lamb may come down to size. A lamb’s umbilical vessels are larger, so feeding in a tube is easier. With today’s advances in miniaturizing surgical methods, that seems like a challenge scientists can overcome.
Messenger RNA therapeutics
Back to “Star Trek.” The hypospray injector’s contents could cure just about any disease, even one newly discovered on a strange planet. That’s not unlike messenger RNA (mRNA) technology, a breakthrough that enabled scientists to quickly develop some of the first COVID-19 vaccines.
But vaccines are just the beginning of what this technology can do.
A whole field of immunotherapy is emerging that uses mRNA to deliver instructions to produce chimeric antigen receptor–modified immune cells (CAR-modified immune cells). These cells are engineered to target diseased cells and tissues, like cancer cells and harmful fibroblasts (scar tissue) that promote fibrosis in, for example, the heart and lungs.
The field is bursting with rodent research, and clinical trials have started for treating some advanced-stage malignancies.
Actual clinical use may be years away, but if all goes well, these medicines could help treat or even cure the core medical problems facing humanity. We’re talking cancer, heart disease, neurodegenerative disease – transforming one therapy into another by simply changing the mRNA’s “nucleotide sequence,” the blueprint containing instructions telling it what to do, and what disease to attack.
As this technology matures, we may start to feel as if we’re really on “Star Trek,” where Dr. Leonard “Bones” McCoy pulls out the same device to treat just about every disease or injury.
A version of this article first appeared on WebMD.com.
When I was a child, I watched syndicated episodes of the original “Star Trek.” I was dazzled by the space travel, sure, but also the medical technology.
A handheld “tricorder” detected diseases, while an intramuscular injector (“hypospray”) could treat them. Sickbay “biobeds” came with real-time health monitors that looked futuristic at the time but seem primitive today.
Such visions inspired a lot of us kids to pursue science. Little did we know the real-life advances many of us would see in our lifetimes.
Artificial intelligence helping to spot disease, robots performing surgery, even video calls between doctor and patient – all these once sounded fantastical but now happen in clinical care.
Now, in the 23rd year of the 21st century, you might not believe wht we’ll be capable of next. Three especially wild examples are moving closer to clinical reality.
Human hibernation
Captain America, Han Solo, and “Star Trek” villain Khan – all were preserved at low temperatures and then revived, waking up alive and well months, decades, or centuries later. These are fictional examples, to be sure, but the science they’re rooted in is real.
(In one extreme case, a climber survived after almost 9 hours of efforts to revive him.)
Useful for a space traveler? Maybe not. But it’s potentially huge for someone with life-threatening injuries from a car accident or a gunshot wound.
That’s the thinking behind a breakthrough procedure that came after decades of research on pigs and dogs, now in a clinical trial. The idea: A person with massive blood loss whose heart has stopped is injected with an ice-cold fluid, cooling them from the inside, down to about 50° F.
Doctors already induce more modest hypothermia to protect the brain and other organs after cardiac arrest and during surgery on the aortic arch (the main artery carrying blood from the heart).
But this experimental procedure – called emergency preservation and resuscitation (EPR) – goes far beyond that, dramatically “decreasing the body’s need for oxygen and blood flow,” says Samuel Tisherman, MD, a trauma surgeon at the University of Maryland Medical Center and the trial’s lead researcher. This puts the patient in a state of suspended animation that “could buy time for surgeons to stop the bleeding and save more of these patients.”
The technique has been done on at least six patients, though none were reported to survive. The trial is expected to include 20 people by the time it wraps up in December, according to the listing on the U.S. clinical trials database. Though given the strict requirements for candidates (emergency trauma victims who are not likely to survive), one can’t exactly rely on a set schedule.
Still, the technology is promising. Someday we may even use it to keep patients in suspended animation for months or years, experts predict, helping astronauts through decades-long spaceflights, or stalling death in sick patients awaiting a cure.
Artificial womb
Another sci-fi classic: growing human babies outside the womb. Think the fetus fields from “The Matrix,” or the frozen embryos in “Alien: Covenant.”
In 1923, British biologist J.B.S. Haldane coined a term for that – ectogenesis. He predicted that 70% of pregnancies would take place, from fertilization to birth, in artificial wombs by 2074. That many seems unlikely, but the timeline is on track.
Developing an embryo outside the womb is already routine in in vitro fertilization. And technology enables preterm babies to survive through much of the second half of gestation. Normal human pregnancy is 40 weeks, and the youngest preterm baby ever to survive was 21 weeks and 1 day old, just a few days younger than a smattering of others who lived.
The biggest obstacle for babies younger than that is lung viability. Mechanical ventilation can damage the lungs and lead to a chronic (sometimes fatal) lung disease known as bronchopulmonary dysplasia. Avoiding this would mean figuring out a way to maintain fetal circulation – the intricate system that delivers oxygenated blood from the placenta to the fetus via the umbilical cord. Researchers at Children’s Hospital of Philadelphia have done this using a fetal lamb.
The key to their invention is a substitute placenta: an oxygenator connected to the lamb’s umbilical cord. Tubes inserted through the umbilical vein and arteries carry oxygenated blood from the “placenta” to the fetus, and deoxygenated blood back out. The lamb resides in an artificial, fluid-filled amniotic sac until its lungs and other organs are developed.
Fertility treatment could benefit, too. “An artificial womb may substitute in situations in which a gestational carrier – surrogate – is indicated,” says Paula Amato, MD, a professor of obstetrics and gynecology at Oregon Health and Science University, Portland. (Dr. Amato is not involved in the CHOP research.) For example: when the mother is missing a uterus or can’t carry a pregnancy safely.
No date is set for clinical trials yet. But according to the research, the main difference between human and lamb may come down to size. A lamb’s umbilical vessels are larger, so feeding in a tube is easier. With today’s advances in miniaturizing surgical methods, that seems like a challenge scientists can overcome.
Messenger RNA therapeutics
Back to “Star Trek.” The hypospray injector’s contents could cure just about any disease, even one newly discovered on a strange planet. That’s not unlike messenger RNA (mRNA) technology, a breakthrough that enabled scientists to quickly develop some of the first COVID-19 vaccines.
But vaccines are just the beginning of what this technology can do.
A whole field of immunotherapy is emerging that uses mRNA to deliver instructions to produce chimeric antigen receptor–modified immune cells (CAR-modified immune cells). These cells are engineered to target diseased cells and tissues, like cancer cells and harmful fibroblasts (scar tissue) that promote fibrosis in, for example, the heart and lungs.
The field is bursting with rodent research, and clinical trials have started for treating some advanced-stage malignancies.
Actual clinical use may be years away, but if all goes well, these medicines could help treat or even cure the core medical problems facing humanity. We’re talking cancer, heart disease, neurodegenerative disease – transforming one therapy into another by simply changing the mRNA’s “nucleotide sequence,” the blueprint containing instructions telling it what to do, and what disease to attack.
As this technology matures, we may start to feel as if we’re really on “Star Trek,” where Dr. Leonard “Bones” McCoy pulls out the same device to treat just about every disease or injury.
A version of this article first appeared on WebMD.com.