User login
For MD-IQ use only
SPF is only the start when recommending sunscreens
CHICAGO – at the inaugural Pigmentary Disorders Exchange Symposium.
Among the first factors physicians should consider before recommending sunscreen are a patient’s Fitzpatrick skin type, risks for burning or tanning, underlying skin disorders, and medications the patient is taking, Dr. Taylor, professor of dermatology at the University of Pennsylvania, Philadelphia, said at the meeting, provided by MedscapeLIVE! If patients are on hypertensives, for example, medications can make them more photosensitive.
Consider skin type
Dr. Taylor said she was dismayed by the results of a recent study, which found that 43% of dermatologists who responded to a survey reported that they never, rarely, or only sometimes took a patient’s skin type into account when making sunscreen recommendations. The article is referenced in a 2022 expert panel consensus paper she coauthored on photoprotection “for skin of all color.” But she pointed out that considering skin type alone is inadequate.
Questions for patients in joint decision-making should include lifestyle and work choices such as whether they work inside or outside, and how much sun exposure they get in a typical day. Heat and humidity levels should also be considered as should a patient’s susceptibility to dyspigmentation. “That could be overall darkening of the skin, mottled hyperpigmentation, actinic dyspigmentation, and, of course, propensity for skin cancer,” she said.
Use differs by race
Dr. Taylor, who is also vice chair for diversity, equity and inclusion in the department of dermatology at the University of Pennsylvania, pointed out that sunscreen use differs considerably by race.
In study of 8,952 adults in the United States who reported that they were sun sensitive found that a subset of adults with skin of color were significantly less likely to use sunscreen when compared with non-Hispanic White adults: Non-Hispanic Black (adjusted odds ratio, 0.43); non-Hispanic Asian (aOR. 0.54); and Hispanic (aOR, 0.70) adults.
In the study, non-Hispanic Black and Hispanic adults were significantly less likely to use sunscreens with an SPF greater than 15. In addition, non-Hispanic Black, non-Hispanic Asian, and Hispanic adults were significantly more likely than non-Hispanic Whites to wear long sleeves when outside. Such differences are important to keep in mind when advising patients about sunscreens, she said.
Protection for lighter-colored skin
Dr. Taylor said that, for patients with lighter skin tones, “we really want to protect against ultraviolet B as well as ultraviolet A, particularly ultraviolet A2. Ultraviolet radiation is going to cause DNA damage.” Patients with Fitzpatrick skin types I, II, or III are most susceptible to the effects of UVB with sunburn inflammation, which will cause erythema and tanning, and immunosuppression.
“For those who are I, II, and III, we do want to recommend a broad-spectrum, photostable sunscreen with a critical wavelength of 370 nanometers, which is going to protect from both UVB and UVA2,” she said.
Sunscreen recommendations are meant to be paired with advice to avoid midday sun from 10 a.m. to 2 p.m., wearing protective clothing and accessories, and seeking shade, she noted.
Dr. Taylor said, for those patients with lighter skin who are more susceptible to photodamage and premature aging, physicians should recommend sunscreens that contain DNA repair enzymes such as photolyases and sunscreens that contain antioxidants that can prevent or reverse DNA damage. “The exogenous form of these lyases have been manufactured and added to sunscreens,” Dr. Taylor said. “They’re readily available in the United States. That is something to consider for patients with significant photodamage.”
Retinoids can also help alleviate or reverse photodamage, she added.
Protection for darker-colored skin
“Many people of color do not believe they need sunscreen,” Dr. Taylor said. But studies show that, although there may be more intrinsic protection, sunscreen is still needed.
Over 30 years ago, Halder and colleagues reported that melanin in skin of color can filter two to five times more UV radiation, and in a paper on the photoprotective role of melanin, Kaidbey and colleagues found that skin types V and VI had an intrinsic SPF of 13 when compared with those who have lighter complexions, which had an SPF of 3.
Sunburns seem to occur less frequently in people with skin of color, but that may be because erythema is less apparent in people with darker skin tones or because of differences in personal definitions of sunburn, Dr. Taylor said.
“Skin of color can and does sustain sunburns and sunscreen will help prevent that,” she said, adding that a recommendation of an SPF 30 is likely sufficient for these patients. Dr. Taylor noted that sunscreens for patients with darker skin often cost substantially more than those for lighter skin, and that should be considered in recommendations.
Tinted sunscreens
Dr. Taylor said that, while broad-spectrum photostable sunscreens protect against UVB and UVA 2, they don’t protect from visible light and UVA1. Two methods to add that protection are using inorganic tinted sunscreens that contain iron oxide or pigmentary titanium dioxide. Dr. Taylor was a coauthor of a practical guide to tinted sunscreens published in 2022.
“For iron oxide, we want a concentration of 3% or greater,” she said, adding that the percentage often is not known because if it is contained in a sunscreen, it is listed as an inactive ingredient.
Another method to address visible light and UVA1 is the use of antioxidant-containing sunscreens with vitamin E, vitamin C, or licochalcone A, Dr. Taylor said.
During the question-and-answer period following her presentation, Amit Pandya, MD, adjunct professor of dermatology at University of Texas Southwestern Medical Center, Dallas, asked why “every makeup, every sunscreen, just says iron oxide,” since it is known that visible light will cause pigmentation, especially in those with darker skin tones.
He urged pushing for a law that would require listing the percentage of iron oxide on products to assure it is sufficient, according to what the literature recommends.
Conference Chair Pearl Grimes, MD, director of the Vitiligo and Pigmentation Institute of Southern California, Los Angeles, said that she recommends tinted sunscreens almost exclusively for her patients, but those with darker skin colors struggle to match color.
Dr. Taylor referred to an analysis published in 2022 of 58 over-the counter sunscreens, which found that only 38% of tinted sunscreens was available in more than one shade, “which is a problem for many of our patients.” She said that providing samples with different hues and tactile sensations may help patients find the right product.
Dr. Taylor disclosed being on the advisory boards for AbbVie, Avita Medical, Beiersdorf, Biorez, Eli Lily, EPI Health, Evolus, Galderma, Hugel America, Johnson and Johnson, L’Oreal USA, MedScape, Pfizer, Scientis US, UCB, Vichy Laboratories. She is a consultant for Arcutis Biothermapeutics, Beiersdorf, Bristol-Myers Squibb, Cara Therapeutics, Dior, and Sanofi. She has done contracted research for Allergan Aesthetics, Concert Pharmaceuticals, Croma-Pharma, Eli Lilly, and Pfizer, and has an ownership interest in Armis Scientific, GloGetter, and Piction Health.
Medscape and this news organization are owned by the same parent company.
CHICAGO – at the inaugural Pigmentary Disorders Exchange Symposium.
Among the first factors physicians should consider before recommending sunscreen are a patient’s Fitzpatrick skin type, risks for burning or tanning, underlying skin disorders, and medications the patient is taking, Dr. Taylor, professor of dermatology at the University of Pennsylvania, Philadelphia, said at the meeting, provided by MedscapeLIVE! If patients are on hypertensives, for example, medications can make them more photosensitive.
Consider skin type
Dr. Taylor said she was dismayed by the results of a recent study, which found that 43% of dermatologists who responded to a survey reported that they never, rarely, or only sometimes took a patient’s skin type into account when making sunscreen recommendations. The article is referenced in a 2022 expert panel consensus paper she coauthored on photoprotection “for skin of all color.” But she pointed out that considering skin type alone is inadequate.
Questions for patients in joint decision-making should include lifestyle and work choices such as whether they work inside or outside, and how much sun exposure they get in a typical day. Heat and humidity levels should also be considered as should a patient’s susceptibility to dyspigmentation. “That could be overall darkening of the skin, mottled hyperpigmentation, actinic dyspigmentation, and, of course, propensity for skin cancer,” she said.
Use differs by race
Dr. Taylor, who is also vice chair for diversity, equity and inclusion in the department of dermatology at the University of Pennsylvania, pointed out that sunscreen use differs considerably by race.
In study of 8,952 adults in the United States who reported that they were sun sensitive found that a subset of adults with skin of color were significantly less likely to use sunscreen when compared with non-Hispanic White adults: Non-Hispanic Black (adjusted odds ratio, 0.43); non-Hispanic Asian (aOR. 0.54); and Hispanic (aOR, 0.70) adults.
In the study, non-Hispanic Black and Hispanic adults were significantly less likely to use sunscreens with an SPF greater than 15. In addition, non-Hispanic Black, non-Hispanic Asian, and Hispanic adults were significantly more likely than non-Hispanic Whites to wear long sleeves when outside. Such differences are important to keep in mind when advising patients about sunscreens, she said.
Protection for lighter-colored skin
Dr. Taylor said that, for patients with lighter skin tones, “we really want to protect against ultraviolet B as well as ultraviolet A, particularly ultraviolet A2. Ultraviolet radiation is going to cause DNA damage.” Patients with Fitzpatrick skin types I, II, or III are most susceptible to the effects of UVB with sunburn inflammation, which will cause erythema and tanning, and immunosuppression.
“For those who are I, II, and III, we do want to recommend a broad-spectrum, photostable sunscreen with a critical wavelength of 370 nanometers, which is going to protect from both UVB and UVA2,” she said.
Sunscreen recommendations are meant to be paired with advice to avoid midday sun from 10 a.m. to 2 p.m., wearing protective clothing and accessories, and seeking shade, she noted.
Dr. Taylor said, for those patients with lighter skin who are more susceptible to photodamage and premature aging, physicians should recommend sunscreens that contain DNA repair enzymes such as photolyases and sunscreens that contain antioxidants that can prevent or reverse DNA damage. “The exogenous form of these lyases have been manufactured and added to sunscreens,” Dr. Taylor said. “They’re readily available in the United States. That is something to consider for patients with significant photodamage.”
Retinoids can also help alleviate or reverse photodamage, she added.
Protection for darker-colored skin
“Many people of color do not believe they need sunscreen,” Dr. Taylor said. But studies show that, although there may be more intrinsic protection, sunscreen is still needed.
Over 30 years ago, Halder and colleagues reported that melanin in skin of color can filter two to five times more UV radiation, and in a paper on the photoprotective role of melanin, Kaidbey and colleagues found that skin types V and VI had an intrinsic SPF of 13 when compared with those who have lighter complexions, which had an SPF of 3.
Sunburns seem to occur less frequently in people with skin of color, but that may be because erythema is less apparent in people with darker skin tones or because of differences in personal definitions of sunburn, Dr. Taylor said.
“Skin of color can and does sustain sunburns and sunscreen will help prevent that,” she said, adding that a recommendation of an SPF 30 is likely sufficient for these patients. Dr. Taylor noted that sunscreens for patients with darker skin often cost substantially more than those for lighter skin, and that should be considered in recommendations.
Tinted sunscreens
Dr. Taylor said that, while broad-spectrum photostable sunscreens protect against UVB and UVA 2, they don’t protect from visible light and UVA1. Two methods to add that protection are using inorganic tinted sunscreens that contain iron oxide or pigmentary titanium dioxide. Dr. Taylor was a coauthor of a practical guide to tinted sunscreens published in 2022.
“For iron oxide, we want a concentration of 3% or greater,” she said, adding that the percentage often is not known because if it is contained in a sunscreen, it is listed as an inactive ingredient.
Another method to address visible light and UVA1 is the use of antioxidant-containing sunscreens with vitamin E, vitamin C, or licochalcone A, Dr. Taylor said.
During the question-and-answer period following her presentation, Amit Pandya, MD, adjunct professor of dermatology at University of Texas Southwestern Medical Center, Dallas, asked why “every makeup, every sunscreen, just says iron oxide,” since it is known that visible light will cause pigmentation, especially in those with darker skin tones.
He urged pushing for a law that would require listing the percentage of iron oxide on products to assure it is sufficient, according to what the literature recommends.
Conference Chair Pearl Grimes, MD, director of the Vitiligo and Pigmentation Institute of Southern California, Los Angeles, said that she recommends tinted sunscreens almost exclusively for her patients, but those with darker skin colors struggle to match color.
Dr. Taylor referred to an analysis published in 2022 of 58 over-the counter sunscreens, which found that only 38% of tinted sunscreens was available in more than one shade, “which is a problem for many of our patients.” She said that providing samples with different hues and tactile sensations may help patients find the right product.
Dr. Taylor disclosed being on the advisory boards for AbbVie, Avita Medical, Beiersdorf, Biorez, Eli Lily, EPI Health, Evolus, Galderma, Hugel America, Johnson and Johnson, L’Oreal USA, MedScape, Pfizer, Scientis US, UCB, Vichy Laboratories. She is a consultant for Arcutis Biothermapeutics, Beiersdorf, Bristol-Myers Squibb, Cara Therapeutics, Dior, and Sanofi. She has done contracted research for Allergan Aesthetics, Concert Pharmaceuticals, Croma-Pharma, Eli Lilly, and Pfizer, and has an ownership interest in Armis Scientific, GloGetter, and Piction Health.
Medscape and this news organization are owned by the same parent company.
CHICAGO – at the inaugural Pigmentary Disorders Exchange Symposium.
Among the first factors physicians should consider before recommending sunscreen are a patient’s Fitzpatrick skin type, risks for burning or tanning, underlying skin disorders, and medications the patient is taking, Dr. Taylor, professor of dermatology at the University of Pennsylvania, Philadelphia, said at the meeting, provided by MedscapeLIVE! If patients are on hypertensives, for example, medications can make them more photosensitive.
Consider skin type
Dr. Taylor said she was dismayed by the results of a recent study, which found that 43% of dermatologists who responded to a survey reported that they never, rarely, or only sometimes took a patient’s skin type into account when making sunscreen recommendations. The article is referenced in a 2022 expert panel consensus paper she coauthored on photoprotection “for skin of all color.” But she pointed out that considering skin type alone is inadequate.
Questions for patients in joint decision-making should include lifestyle and work choices such as whether they work inside or outside, and how much sun exposure they get in a typical day. Heat and humidity levels should also be considered as should a patient’s susceptibility to dyspigmentation. “That could be overall darkening of the skin, mottled hyperpigmentation, actinic dyspigmentation, and, of course, propensity for skin cancer,” she said.
Use differs by race
Dr. Taylor, who is also vice chair for diversity, equity and inclusion in the department of dermatology at the University of Pennsylvania, pointed out that sunscreen use differs considerably by race.
In study of 8,952 adults in the United States who reported that they were sun sensitive found that a subset of adults with skin of color were significantly less likely to use sunscreen when compared with non-Hispanic White adults: Non-Hispanic Black (adjusted odds ratio, 0.43); non-Hispanic Asian (aOR. 0.54); and Hispanic (aOR, 0.70) adults.
In the study, non-Hispanic Black and Hispanic adults were significantly less likely to use sunscreens with an SPF greater than 15. In addition, non-Hispanic Black, non-Hispanic Asian, and Hispanic adults were significantly more likely than non-Hispanic Whites to wear long sleeves when outside. Such differences are important to keep in mind when advising patients about sunscreens, she said.
Protection for lighter-colored skin
Dr. Taylor said that, for patients with lighter skin tones, “we really want to protect against ultraviolet B as well as ultraviolet A, particularly ultraviolet A2. Ultraviolet radiation is going to cause DNA damage.” Patients with Fitzpatrick skin types I, II, or III are most susceptible to the effects of UVB with sunburn inflammation, which will cause erythema and tanning, and immunosuppression.
“For those who are I, II, and III, we do want to recommend a broad-spectrum, photostable sunscreen with a critical wavelength of 370 nanometers, which is going to protect from both UVB and UVA2,” she said.
Sunscreen recommendations are meant to be paired with advice to avoid midday sun from 10 a.m. to 2 p.m., wearing protective clothing and accessories, and seeking shade, she noted.
Dr. Taylor said, for those patients with lighter skin who are more susceptible to photodamage and premature aging, physicians should recommend sunscreens that contain DNA repair enzymes such as photolyases and sunscreens that contain antioxidants that can prevent or reverse DNA damage. “The exogenous form of these lyases have been manufactured and added to sunscreens,” Dr. Taylor said. “They’re readily available in the United States. That is something to consider for patients with significant photodamage.”
Retinoids can also help alleviate or reverse photodamage, she added.
Protection for darker-colored skin
“Many people of color do not believe they need sunscreen,” Dr. Taylor said. But studies show that, although there may be more intrinsic protection, sunscreen is still needed.
Over 30 years ago, Halder and colleagues reported that melanin in skin of color can filter two to five times more UV radiation, and in a paper on the photoprotective role of melanin, Kaidbey and colleagues found that skin types V and VI had an intrinsic SPF of 13 when compared with those who have lighter complexions, which had an SPF of 3.
Sunburns seem to occur less frequently in people with skin of color, but that may be because erythema is less apparent in people with darker skin tones or because of differences in personal definitions of sunburn, Dr. Taylor said.
“Skin of color can and does sustain sunburns and sunscreen will help prevent that,” she said, adding that a recommendation of an SPF 30 is likely sufficient for these patients. Dr. Taylor noted that sunscreens for patients with darker skin often cost substantially more than those for lighter skin, and that should be considered in recommendations.
Tinted sunscreens
Dr. Taylor said that, while broad-spectrum photostable sunscreens protect against UVB and UVA 2, they don’t protect from visible light and UVA1. Two methods to add that protection are using inorganic tinted sunscreens that contain iron oxide or pigmentary titanium dioxide. Dr. Taylor was a coauthor of a practical guide to tinted sunscreens published in 2022.
“For iron oxide, we want a concentration of 3% or greater,” she said, adding that the percentage often is not known because if it is contained in a sunscreen, it is listed as an inactive ingredient.
Another method to address visible light and UVA1 is the use of antioxidant-containing sunscreens with vitamin E, vitamin C, or licochalcone A, Dr. Taylor said.
During the question-and-answer period following her presentation, Amit Pandya, MD, adjunct professor of dermatology at University of Texas Southwestern Medical Center, Dallas, asked why “every makeup, every sunscreen, just says iron oxide,” since it is known that visible light will cause pigmentation, especially in those with darker skin tones.
He urged pushing for a law that would require listing the percentage of iron oxide on products to assure it is sufficient, according to what the literature recommends.
Conference Chair Pearl Grimes, MD, director of the Vitiligo and Pigmentation Institute of Southern California, Los Angeles, said that she recommends tinted sunscreens almost exclusively for her patients, but those with darker skin colors struggle to match color.
Dr. Taylor referred to an analysis published in 2022 of 58 over-the counter sunscreens, which found that only 38% of tinted sunscreens was available in more than one shade, “which is a problem for many of our patients.” She said that providing samples with different hues and tactile sensations may help patients find the right product.
Dr. Taylor disclosed being on the advisory boards for AbbVie, Avita Medical, Beiersdorf, Biorez, Eli Lily, EPI Health, Evolus, Galderma, Hugel America, Johnson and Johnson, L’Oreal USA, MedScape, Pfizer, Scientis US, UCB, Vichy Laboratories. She is a consultant for Arcutis Biothermapeutics, Beiersdorf, Bristol-Myers Squibb, Cara Therapeutics, Dior, and Sanofi. She has done contracted research for Allergan Aesthetics, Concert Pharmaceuticals, Croma-Pharma, Eli Lilly, and Pfizer, and has an ownership interest in Armis Scientific, GloGetter, and Piction Health.
Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPELIVE! PIGMENTARY DISORDERS SYMPOSIUM
Low-dose atropine improves myopia in children
Myopia affects roughly one-third of the population worldwide - a figure that is projected to reach 50% by 2050. Low-dose atropine, which helps curb the condition, currently is available in the United States only through compounding pharmacies. The products contain preservatives – raising questions about potential toxicities to the eye – and may not be of pharmaceutical grade. In a new study published in JAMA Ophthalmology,
Methodology
- The CHAMP study was a double-masked, placebo-controlled, randomized, phase 3 trial conducted between Nov. 20, 2017, and Aug. 22, 2022, that involved children at 26 sites in North America and 5 centers in Europe.
- Children received either 0.01% or 0.02% atropine drops once per day.
- Patients were aged 3-16 years. They demonstrated a spherical equivalent refractive error (SER) of −0.50 diopter (D) to −6.00 D astigmatism no worse than −1.50 D.
- Of these patients, 573 were included in a safety analysis, and 489 were included in a modified intention-to-treat analysis.
Takeaways
- After 36 months, the 0.01% dose of atropine was associated with a significantly lower responder proportion (odds ratio, 4.54) and slower progression of SER and axial elongation.
- The effect of the 0.02% dose on responder proportion and SER progression was not statistically significant, but the treatment was associated with slower axial elongation.
- The researchers observed no serious ocular adverse events and few serious nonocular events, none of which was determined to be associated with the treatment.
In practice: According to the researchers, “from a risk/benefit perspective, the efficacy and safety observed suggests that low-dose atropine may provide a treatment option for children aged 3-17 years with myopia progression, which may lead to less frequent or delayed change in glasses, progression to less severe correction, and potentially reduce long-term sequelae, which could lead to vision loss later in life, such as myopic maculopathy.”
Study details: The CHAMP study was led by Karla Zadnik, OD, PhD, of Ohio State University, Columbus, and was funded by Vylulma.
Limitations: The researchers said the trial was potentially limited by the fact that patients switched from the study drug to confounding treatments. In addition, patients at the low and high age ranges were not well represented.
Disclosures: Dr. Zadnik received consultant fees from Vyluma during the study.
A version of this article first appeared on Medscape.com.
Myopia affects roughly one-third of the population worldwide - a figure that is projected to reach 50% by 2050. Low-dose atropine, which helps curb the condition, currently is available in the United States only through compounding pharmacies. The products contain preservatives – raising questions about potential toxicities to the eye – and may not be of pharmaceutical grade. In a new study published in JAMA Ophthalmology,
Methodology
- The CHAMP study was a double-masked, placebo-controlled, randomized, phase 3 trial conducted between Nov. 20, 2017, and Aug. 22, 2022, that involved children at 26 sites in North America and 5 centers in Europe.
- Children received either 0.01% or 0.02% atropine drops once per day.
- Patients were aged 3-16 years. They demonstrated a spherical equivalent refractive error (SER) of −0.50 diopter (D) to −6.00 D astigmatism no worse than −1.50 D.
- Of these patients, 573 were included in a safety analysis, and 489 were included in a modified intention-to-treat analysis.
Takeaways
- After 36 months, the 0.01% dose of atropine was associated with a significantly lower responder proportion (odds ratio, 4.54) and slower progression of SER and axial elongation.
- The effect of the 0.02% dose on responder proportion and SER progression was not statistically significant, but the treatment was associated with slower axial elongation.
- The researchers observed no serious ocular adverse events and few serious nonocular events, none of which was determined to be associated with the treatment.
In practice: According to the researchers, “from a risk/benefit perspective, the efficacy and safety observed suggests that low-dose atropine may provide a treatment option for children aged 3-17 years with myopia progression, which may lead to less frequent or delayed change in glasses, progression to less severe correction, and potentially reduce long-term sequelae, which could lead to vision loss later in life, such as myopic maculopathy.”
Study details: The CHAMP study was led by Karla Zadnik, OD, PhD, of Ohio State University, Columbus, and was funded by Vylulma.
Limitations: The researchers said the trial was potentially limited by the fact that patients switched from the study drug to confounding treatments. In addition, patients at the low and high age ranges were not well represented.
Disclosures: Dr. Zadnik received consultant fees from Vyluma during the study.
A version of this article first appeared on Medscape.com.
Myopia affects roughly one-third of the population worldwide - a figure that is projected to reach 50% by 2050. Low-dose atropine, which helps curb the condition, currently is available in the United States only through compounding pharmacies. The products contain preservatives – raising questions about potential toxicities to the eye – and may not be of pharmaceutical grade. In a new study published in JAMA Ophthalmology,
Methodology
- The CHAMP study was a double-masked, placebo-controlled, randomized, phase 3 trial conducted between Nov. 20, 2017, and Aug. 22, 2022, that involved children at 26 sites in North America and 5 centers in Europe.
- Children received either 0.01% or 0.02% atropine drops once per day.
- Patients were aged 3-16 years. They demonstrated a spherical equivalent refractive error (SER) of −0.50 diopter (D) to −6.00 D astigmatism no worse than −1.50 D.
- Of these patients, 573 were included in a safety analysis, and 489 were included in a modified intention-to-treat analysis.
Takeaways
- After 36 months, the 0.01% dose of atropine was associated with a significantly lower responder proportion (odds ratio, 4.54) and slower progression of SER and axial elongation.
- The effect of the 0.02% dose on responder proportion and SER progression was not statistically significant, but the treatment was associated with slower axial elongation.
- The researchers observed no serious ocular adverse events and few serious nonocular events, none of which was determined to be associated with the treatment.
In practice: According to the researchers, “from a risk/benefit perspective, the efficacy and safety observed suggests that low-dose atropine may provide a treatment option for children aged 3-17 years with myopia progression, which may lead to less frequent or delayed change in glasses, progression to less severe correction, and potentially reduce long-term sequelae, which could lead to vision loss later in life, such as myopic maculopathy.”
Study details: The CHAMP study was led by Karla Zadnik, OD, PhD, of Ohio State University, Columbus, and was funded by Vylulma.
Limitations: The researchers said the trial was potentially limited by the fact that patients switched from the study drug to confounding treatments. In addition, patients at the low and high age ranges were not well represented.
Disclosures: Dr. Zadnik received consultant fees from Vyluma during the study.
A version of this article first appeared on Medscape.com.
FROM JAMA OPHTHALMOLOGY
United Healthcare ditches prior authorization in favor of new policy
Instead, the giant health insurer will adopt an “advance notification” program for nonscreening and nonemergent gastrointestinal procedures.
The company has not made any changes to their policy regarding screening colonoscopies for preventive care, and the advance notification policy does not impact screening colonoscopies.
UHC alerted physicians to changes to the program yesterday, including updated notices on UHCProvider.com with a new Frequently Asked Questions document.
The advance notification program “will not result in the denial of care for clinical reasons or for failure to notify and will help educate physicians who are not following clinical best practices. Provider groups who do not submit advance notification during this period will not be eligible for the United Healthcare Gold Card program,” a spokesperson for the company said.
The previously announced Gold Card program, which is scheduled to start in early 2024, would eliminate prior authorization requirements for providers that meet certain eligibility requirements.
The American Gastroenterological Association remains “extremely concerned” that UHC’s advance notification program is a “temporary patch” likely to have significant repercussions for patient access. The organization says the program only temporarily postpones prior authorization requirements set to impact the insurer’s 27.4 million commercial beneficiaries while increasing the administrative burden on clinicians.
The AGA called the program “nebulous” and “poorly defined.” It would ostensibly require physicians to input “copious” amounts of highly complex and granular patient data prior to performing colonoscopies and endoscopies, the AGA says.
AGA President Barbara H. Jung, MD, AGAF, said UHC’s “slap-dash approach to rolling out a policy that will ultimately control patient access to critical, often life-saving, medical procedures flies in the face of common sense and responsible medical practice.”
“It also indicates that UHC does not currently have data that show any significant overutilization of critical endoscopy and colonoscopy procedures that would ostensibly justify this program or prior authorization. UHC is not acting in good faith, and its actions will compromise patient access to potentially lifesaving procedures,” Dr. Jung added.
Recent data show 62% of high-risk patients in the United States who had polyps removed had evidence of delayed or no use of surveillance colonoscopies after 10 years.
“If other prior authorization requirements imposed on patients for specialty care are any indication, we expect to see negative patient outcomes with an enormous cost to patient well-being and physician resources,” AGA Vice President Lawrence Kim, MD, wrote in a news release.
“Given the high percentage of eventual approvals by insurers mandating prior authorization, we anticipate there will be little to no benefit from this prior authorization requirement. When utilized this way, it becomes a nonsensical and harmful policy,” Dr. Kim added.
AGA says it will continue to work closely with its members to assess the full impact of the new requirements and urges UHC to make endoscopy procedures more accessible to patients.
A recent American Medical Association survey on prior authorization found that one-third (33%) of doctors said the insurance barrier has led to a serious adverse event such as hospitalization, permanent disability, or death for a patient in their care. Nearly half (46%) of physicians reported that prior authorization has led to immediate care and/or emergency department visits.
In a 2023 survey of AGA membership, conducted before UHC announced its proposed prior authorization policy, 95% of respondents said prior authorization restrictions have impacted patient access to clinically appropriate treatments and patient clinical outcomes. And 84% said the burdens associated with prior authorization policies have increased “significantly” (60%) or “somewhat” (24%) over the last 5 years.
A version of this article first appeared on Medscape.com.
Instead, the giant health insurer will adopt an “advance notification” program for nonscreening and nonemergent gastrointestinal procedures.
The company has not made any changes to their policy regarding screening colonoscopies for preventive care, and the advance notification policy does not impact screening colonoscopies.
UHC alerted physicians to changes to the program yesterday, including updated notices on UHCProvider.com with a new Frequently Asked Questions document.
The advance notification program “will not result in the denial of care for clinical reasons or for failure to notify and will help educate physicians who are not following clinical best practices. Provider groups who do not submit advance notification during this period will not be eligible for the United Healthcare Gold Card program,” a spokesperson for the company said.
The previously announced Gold Card program, which is scheduled to start in early 2024, would eliminate prior authorization requirements for providers that meet certain eligibility requirements.
The American Gastroenterological Association remains “extremely concerned” that UHC’s advance notification program is a “temporary patch” likely to have significant repercussions for patient access. The organization says the program only temporarily postpones prior authorization requirements set to impact the insurer’s 27.4 million commercial beneficiaries while increasing the administrative burden on clinicians.
The AGA called the program “nebulous” and “poorly defined.” It would ostensibly require physicians to input “copious” amounts of highly complex and granular patient data prior to performing colonoscopies and endoscopies, the AGA says.
AGA President Barbara H. Jung, MD, AGAF, said UHC’s “slap-dash approach to rolling out a policy that will ultimately control patient access to critical, often life-saving, medical procedures flies in the face of common sense and responsible medical practice.”
“It also indicates that UHC does not currently have data that show any significant overutilization of critical endoscopy and colonoscopy procedures that would ostensibly justify this program or prior authorization. UHC is not acting in good faith, and its actions will compromise patient access to potentially lifesaving procedures,” Dr. Jung added.
Recent data show 62% of high-risk patients in the United States who had polyps removed had evidence of delayed or no use of surveillance colonoscopies after 10 years.
“If other prior authorization requirements imposed on patients for specialty care are any indication, we expect to see negative patient outcomes with an enormous cost to patient well-being and physician resources,” AGA Vice President Lawrence Kim, MD, wrote in a news release.
“Given the high percentage of eventual approvals by insurers mandating prior authorization, we anticipate there will be little to no benefit from this prior authorization requirement. When utilized this way, it becomes a nonsensical and harmful policy,” Dr. Kim added.
AGA says it will continue to work closely with its members to assess the full impact of the new requirements and urges UHC to make endoscopy procedures more accessible to patients.
A recent American Medical Association survey on prior authorization found that one-third (33%) of doctors said the insurance barrier has led to a serious adverse event such as hospitalization, permanent disability, or death for a patient in their care. Nearly half (46%) of physicians reported that prior authorization has led to immediate care and/or emergency department visits.
In a 2023 survey of AGA membership, conducted before UHC announced its proposed prior authorization policy, 95% of respondents said prior authorization restrictions have impacted patient access to clinically appropriate treatments and patient clinical outcomes. And 84% said the burdens associated with prior authorization policies have increased “significantly” (60%) or “somewhat” (24%) over the last 5 years.
A version of this article first appeared on Medscape.com.
Instead, the giant health insurer will adopt an “advance notification” program for nonscreening and nonemergent gastrointestinal procedures.
The company has not made any changes to their policy regarding screening colonoscopies for preventive care, and the advance notification policy does not impact screening colonoscopies.
UHC alerted physicians to changes to the program yesterday, including updated notices on UHCProvider.com with a new Frequently Asked Questions document.
The advance notification program “will not result in the denial of care for clinical reasons or for failure to notify and will help educate physicians who are not following clinical best practices. Provider groups who do not submit advance notification during this period will not be eligible for the United Healthcare Gold Card program,” a spokesperson for the company said.
The previously announced Gold Card program, which is scheduled to start in early 2024, would eliminate prior authorization requirements for providers that meet certain eligibility requirements.
The American Gastroenterological Association remains “extremely concerned” that UHC’s advance notification program is a “temporary patch” likely to have significant repercussions for patient access. The organization says the program only temporarily postpones prior authorization requirements set to impact the insurer’s 27.4 million commercial beneficiaries while increasing the administrative burden on clinicians.
The AGA called the program “nebulous” and “poorly defined.” It would ostensibly require physicians to input “copious” amounts of highly complex and granular patient data prior to performing colonoscopies and endoscopies, the AGA says.
AGA President Barbara H. Jung, MD, AGAF, said UHC’s “slap-dash approach to rolling out a policy that will ultimately control patient access to critical, often life-saving, medical procedures flies in the face of common sense and responsible medical practice.”
“It also indicates that UHC does not currently have data that show any significant overutilization of critical endoscopy and colonoscopy procedures that would ostensibly justify this program or prior authorization. UHC is not acting in good faith, and its actions will compromise patient access to potentially lifesaving procedures,” Dr. Jung added.
Recent data show 62% of high-risk patients in the United States who had polyps removed had evidence of delayed or no use of surveillance colonoscopies after 10 years.
“If other prior authorization requirements imposed on patients for specialty care are any indication, we expect to see negative patient outcomes with an enormous cost to patient well-being and physician resources,” AGA Vice President Lawrence Kim, MD, wrote in a news release.
“Given the high percentage of eventual approvals by insurers mandating prior authorization, we anticipate there will be little to no benefit from this prior authorization requirement. When utilized this way, it becomes a nonsensical and harmful policy,” Dr. Kim added.
AGA says it will continue to work closely with its members to assess the full impact of the new requirements and urges UHC to make endoscopy procedures more accessible to patients.
A recent American Medical Association survey on prior authorization found that one-third (33%) of doctors said the insurance barrier has led to a serious adverse event such as hospitalization, permanent disability, or death for a patient in their care. Nearly half (46%) of physicians reported that prior authorization has led to immediate care and/or emergency department visits.
In a 2023 survey of AGA membership, conducted before UHC announced its proposed prior authorization policy, 95% of respondents said prior authorization restrictions have impacted patient access to clinically appropriate treatments and patient clinical outcomes. And 84% said the burdens associated with prior authorization policies have increased “significantly” (60%) or “somewhat” (24%) over the last 5 years.
A version of this article first appeared on Medscape.com.
Scientists discover variants, therapy for disabling pansclerotic morphea
A team of especially in patients who have not responded to other interventions.
DPM was first reported in 1923, and while a genetic cause has been suspected, it had not been identified until now. The disease is the most severe form of deep morphea, which affects individuals with juvenile localized scleroderma. Patients, generally children under age 14, experience rapid sclerosis of all layers of the skin, fascia, muscle, and bone. DPM is also deadly: Most patients do not live more than 10 years after diagnosis, as they contract squamous cell carcinoma, restrictive pulmonary disease, sepsis, and gangrene.
In the study, published in the New England Journal of Medicine, the researchers discovered that people with DPM have an overactive version of the protein STAT4, which regulates inflammation and wound healing. The scientists studied four patients from three unrelated families with an autosomal dominant pattern of inheritance of DPM.
“Researchers previously thought that this disorder was caused by the immune system attacking the skin,” Sarah Blackstone, a predoctoral fellow in the inflammatory disease section at the National Human Genome Research Institute and co–first author of the study, said in a statement from the National Institutes of Health describing the results. “However, we found that this is an oversimplification, and that both skin and the immune system play an active role in disabling pansclerotic morphea,” added Ms. Blackstone, also a medical student at the University of South Dakota, Sioux Falls.
The overactive STAT4 protein creates a positive feedback loop of inflammation and impaired wound-healing. By targeting JAK, the researchers were able to stop the feedback and patients’ wounds dramatically improved. After 18 months of treatment with oral ruxolitinib, one patient had discontinued all other medications, and had complete resolution of a chest rash, substantial clearing on the arms and legs, and global clinical improvement.
The authors said that oral systemic JAK inhibitor therapy is preferred over topical therapy. Their research also suggested that anti–interleukin-6 monoclonal antibodies – such as tocilizumab, approved for indications that include rheumatoid arthritis and systemic sclerosis–associated interstitial lung disease, “may be an alternative therapy or may be useful in combination with JAK inhibitors in patients with DPM,” the authors wrote.
Most current DPM therapies – including methotrexate, mycophenolate mofetil, and ultraviolet A light therapy – have been ineffective, and some have severe side effects.
“The findings of this study open doors for JAK inhibitors to be a potential treatment for other inflammatory skin disorders or disorders related to tissue scarring, whether it is scarring of the lungs, liver or bone marrow,” Dan Kastner, MD, PhD, an NIH distinguished investigator, head of the NHGRI’s inflammatory disease section, and a senior author of the paper, said in the NIH statement.
“We hope to continue studying other molecules in this pathway and how they are altered in patients with disabling pansclerotic morphea and related conditions to find clues to understanding a broader array of more common diseases,” Lori Broderick, MD, PhD, a senior author of the paper and an associate professor at University of California, San Diego, said in the statement.
The study was led by researchers at NHGRI in collaboration with researchers from UCSD and the University of Pittsburgh. Researchers from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the National Institute of Allergy and Infectious Diseases also participated.
The study was supported by grants from the American Academy of Allergy, Asthma, and Immunology Foundation; the Ludwig Institute for Cancer Research; the University of California, San Diego, department of pediatrics; and the Novo Nordisk Foundation. Additional support and grants were given by the Deutsche Forschungsgemeinschaft, various institutes at the NIH, the California Institute for Regenerative Medicine, the Hydrocephalus Association, the Scleroderma Research Foundation, the Biowulf High-Performance Computing Cluster of the Center for Information Technology, the Undiagnosed Diseases Program of the Common Fund of the Office of the Director of the NIH, and the NIH Clinical Center.
A team of especially in patients who have not responded to other interventions.
DPM was first reported in 1923, and while a genetic cause has been suspected, it had not been identified until now. The disease is the most severe form of deep morphea, which affects individuals with juvenile localized scleroderma. Patients, generally children under age 14, experience rapid sclerosis of all layers of the skin, fascia, muscle, and bone. DPM is also deadly: Most patients do not live more than 10 years after diagnosis, as they contract squamous cell carcinoma, restrictive pulmonary disease, sepsis, and gangrene.
In the study, published in the New England Journal of Medicine, the researchers discovered that people with DPM have an overactive version of the protein STAT4, which regulates inflammation and wound healing. The scientists studied four patients from three unrelated families with an autosomal dominant pattern of inheritance of DPM.
“Researchers previously thought that this disorder was caused by the immune system attacking the skin,” Sarah Blackstone, a predoctoral fellow in the inflammatory disease section at the National Human Genome Research Institute and co–first author of the study, said in a statement from the National Institutes of Health describing the results. “However, we found that this is an oversimplification, and that both skin and the immune system play an active role in disabling pansclerotic morphea,” added Ms. Blackstone, also a medical student at the University of South Dakota, Sioux Falls.
The overactive STAT4 protein creates a positive feedback loop of inflammation and impaired wound-healing. By targeting JAK, the researchers were able to stop the feedback and patients’ wounds dramatically improved. After 18 months of treatment with oral ruxolitinib, one patient had discontinued all other medications, and had complete resolution of a chest rash, substantial clearing on the arms and legs, and global clinical improvement.
The authors said that oral systemic JAK inhibitor therapy is preferred over topical therapy. Their research also suggested that anti–interleukin-6 monoclonal antibodies – such as tocilizumab, approved for indications that include rheumatoid arthritis and systemic sclerosis–associated interstitial lung disease, “may be an alternative therapy or may be useful in combination with JAK inhibitors in patients with DPM,” the authors wrote.
Most current DPM therapies – including methotrexate, mycophenolate mofetil, and ultraviolet A light therapy – have been ineffective, and some have severe side effects.
“The findings of this study open doors for JAK inhibitors to be a potential treatment for other inflammatory skin disorders or disorders related to tissue scarring, whether it is scarring of the lungs, liver or bone marrow,” Dan Kastner, MD, PhD, an NIH distinguished investigator, head of the NHGRI’s inflammatory disease section, and a senior author of the paper, said in the NIH statement.
“We hope to continue studying other molecules in this pathway and how they are altered in patients with disabling pansclerotic morphea and related conditions to find clues to understanding a broader array of more common diseases,” Lori Broderick, MD, PhD, a senior author of the paper and an associate professor at University of California, San Diego, said in the statement.
The study was led by researchers at NHGRI in collaboration with researchers from UCSD and the University of Pittsburgh. Researchers from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the National Institute of Allergy and Infectious Diseases also participated.
The study was supported by grants from the American Academy of Allergy, Asthma, and Immunology Foundation; the Ludwig Institute for Cancer Research; the University of California, San Diego, department of pediatrics; and the Novo Nordisk Foundation. Additional support and grants were given by the Deutsche Forschungsgemeinschaft, various institutes at the NIH, the California Institute for Regenerative Medicine, the Hydrocephalus Association, the Scleroderma Research Foundation, the Biowulf High-Performance Computing Cluster of the Center for Information Technology, the Undiagnosed Diseases Program of the Common Fund of the Office of the Director of the NIH, and the NIH Clinical Center.
A team of especially in patients who have not responded to other interventions.
DPM was first reported in 1923, and while a genetic cause has been suspected, it had not been identified until now. The disease is the most severe form of deep morphea, which affects individuals with juvenile localized scleroderma. Patients, generally children under age 14, experience rapid sclerosis of all layers of the skin, fascia, muscle, and bone. DPM is also deadly: Most patients do not live more than 10 years after diagnosis, as they contract squamous cell carcinoma, restrictive pulmonary disease, sepsis, and gangrene.
In the study, published in the New England Journal of Medicine, the researchers discovered that people with DPM have an overactive version of the protein STAT4, which regulates inflammation and wound healing. The scientists studied four patients from three unrelated families with an autosomal dominant pattern of inheritance of DPM.
“Researchers previously thought that this disorder was caused by the immune system attacking the skin,” Sarah Blackstone, a predoctoral fellow in the inflammatory disease section at the National Human Genome Research Institute and co–first author of the study, said in a statement from the National Institutes of Health describing the results. “However, we found that this is an oversimplification, and that both skin and the immune system play an active role in disabling pansclerotic morphea,” added Ms. Blackstone, also a medical student at the University of South Dakota, Sioux Falls.
The overactive STAT4 protein creates a positive feedback loop of inflammation and impaired wound-healing. By targeting JAK, the researchers were able to stop the feedback and patients’ wounds dramatically improved. After 18 months of treatment with oral ruxolitinib, one patient had discontinued all other medications, and had complete resolution of a chest rash, substantial clearing on the arms and legs, and global clinical improvement.
The authors said that oral systemic JAK inhibitor therapy is preferred over topical therapy. Their research also suggested that anti–interleukin-6 monoclonal antibodies – such as tocilizumab, approved for indications that include rheumatoid arthritis and systemic sclerosis–associated interstitial lung disease, “may be an alternative therapy or may be useful in combination with JAK inhibitors in patients with DPM,” the authors wrote.
Most current DPM therapies – including methotrexate, mycophenolate mofetil, and ultraviolet A light therapy – have been ineffective, and some have severe side effects.
“The findings of this study open doors for JAK inhibitors to be a potential treatment for other inflammatory skin disorders or disorders related to tissue scarring, whether it is scarring of the lungs, liver or bone marrow,” Dan Kastner, MD, PhD, an NIH distinguished investigator, head of the NHGRI’s inflammatory disease section, and a senior author of the paper, said in the NIH statement.
“We hope to continue studying other molecules in this pathway and how they are altered in patients with disabling pansclerotic morphea and related conditions to find clues to understanding a broader array of more common diseases,” Lori Broderick, MD, PhD, a senior author of the paper and an associate professor at University of California, San Diego, said in the statement.
The study was led by researchers at NHGRI in collaboration with researchers from UCSD and the University of Pittsburgh. Researchers from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the National Institute of Allergy and Infectious Diseases also participated.
The study was supported by grants from the American Academy of Allergy, Asthma, and Immunology Foundation; the Ludwig Institute for Cancer Research; the University of California, San Diego, department of pediatrics; and the Novo Nordisk Foundation. Additional support and grants were given by the Deutsche Forschungsgemeinschaft, various institutes at the NIH, the California Institute for Regenerative Medicine, the Hydrocephalus Association, the Scleroderma Research Foundation, the Biowulf High-Performance Computing Cluster of the Center for Information Technology, the Undiagnosed Diseases Program of the Common Fund of the Office of the Director of the NIH, and the NIH Clinical Center.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Traditional Mexican food has health benefits
, according to researchers from the Institute of Sciences at Benemérita Autonomous University of Puebla, Puebla, Mexico.
Their study, published in the journal Foods, is the first to produce tables showing the phenolic content of Mexican dishes. Physicians and nutritionists can use this information as a reference tool when drawing up diet recommendations for patients who could benefit from a higher intake of phenolic compounds (PC).
“Up until now, there hasn’t been a table that we – nutritionists and physicians – could look at and see exactly which foods were richest in these compounds. In the United States, European countries, Asian countries – they’ve all had food tables; in Mexico, we didn’t,” said lead author Julia Alatorre-Cruz, PhD, a biological scientist and postdoctoral researcher at BUAP. “So, it’s a fairly innovative contribution. As a bonus, the information can be used to analyze the relationship between diet and noncommunicable diseases in the Mexican population.”
In recent years, nutrition science has focused on counteracting nutrient deficiency and some diseases by identifying active-food components. Diet offers the possibility to improve the patient’s health conditions by using these components or functional food.
PCs are a diverse group of plant micronutrients, some of which modulate physiologic and molecular pathways involved in energy metabolism. They can act by different mechanisms; the most important of them are conducted by anti-inflammatory, antioxidant activities, and are antiallergic.
Moreover, recent studies explain how PCs positively affect certain illnesses, such as obesity, diabetes, cardiovascular diseases, thrombocytopenia, and metabolic syndrome. Several common features characterize these pathologies – among them are the redox balance and a notable inflammatory response that strongly alters the biochemical and functional characteristics of the affected tissues.
Traditional Mexican food is characterized by grains, tubers, legumes, vegetables, and spices, most of which are rich in PCs. However, the Mexican diet has changed over the past decades because traditional food has been replaced with ultraprocessed food with high-caloric values. Moreover, some vegetables and fruits are preferably consumed after processing, which affects the quantity, quality, and bioavailability of the PCs. In addition, diseases associated with eating habits have increased by more than 27% in the Mexican population.
The objective of the study was to determine whether participants with a higher PC intake from beverages or Mexican dishes have better health conditions than those with a lower intake.
A total of 973 adults (798 females, 175 males) aged 18-79 years were enrolled in this cross-sectional study. The data were obtained from a validated, self-administered food consumption survey that was posted on social media (Facebook) or sent via WhatsApp or email. In one section, there was a list of fruits, vegetables, cereals, legumes, seeds, spices, beverages, and Mexican dishes. The participants were asked to indicate how often in the past month they had consumed these items. There were also sections for providing identification data (for example, age, sex, marital status), height and weight information, and medical history.
“The study was carried out during the pandemic, so that limited contact with the participants,” said Dr. Alatorre-Cruz. “Not being able to directly interact with them was a challenge. For example, we would have liked to have taken those anthropometric measurements ourselves.”
The researchers performed K-means clustering to determine the participant’s health-condition level, resulting in two groups: those with less diseases (LD, n = 649) and those with more diseases (MD, n = 324).
Using the food biochemistry composition reported in multiple papers, the researchers computed the average total phenolic compounds (TPC) for each item listed in the survey. For Mexican dishes, they added the TPC of each recipe’s ingredient, then recalculated to come up with TPC for an individual portion of each recipe (TPCr). To analyze the results of the LD group and of the MD group, phenolic compounds intake of recipe was calculated for each participant.
To Dr. Alatorre-Cruz, the biggest challenge was determining the content of compounds in traditional dishes. “Extensive, in-depth research was done to gather as much information as possible about all of the foods – especially about local and regional ones, because we have such a wide variety – looking to see where that information would match up with the exact method with which the compounds of interest were extracted.”
As expected, the team found that food with high PC was associated with a better health condition. However, the consumption of beverages and Mexican dishes was lower than their expectations. As noted in the article, the Mexican diet has changed over the last decades because traditional food has been replaced with ultraprocessed food with high-caloric values.
The authors suggest that their data confirm the alarming changes previously reported in the Mexican diet – changes possibly attributable to the increased influence of other countries via social media and economic globalization. However, they also found that beans, corn (mainly tortilla), and nopal intake remained preserved in the Mexican eating habits.
Their statistical analyses revealed that sex, age, and education seem to play a role in the presence or absence of diseases in the Mexican cohort. Men, participants over age 29 years, and those with lower levels of education had more diseases.
Dr. Alatorre-Cruz told this news organization that the Foods article opens new lines of research for the group to pursue. “One future proposal looks to enroll patients with conditions where there’s an increase in oxidative stress – which we know has quite a detrimental effect – and look into possible links with their diet. But now, maybe the focus can be on patients with a single condition and using these traditional-food tables to find links. We also want to know more about the molecular or biochemical mechanisms that are modulating the association that we saw first in animal models.”
Laura Álvarez, MD, a specialist in clinical nutrition, is the founder of the NUTRIENT project, which focuses on nutrition and strength training. In her opinion, the study extols the benefits of Mexican food. “I’ve always thought that people have this idea that Mexican food isn’t good. For example, thinking that it’s very high in fat. But in reality, Mexican food is very rich in nutrients.”
She added that research should be broadened, by enrolling a larger group of participants and evaluating not only a greater number and a greater variety of Mexican dishes, and new variables as well. “I think that body composition could be taken into account. In this study, they took the BMI into account to assess overweight and obesity, but ... the BMI doesn’t tell us much. Overweight can be due to fat or due to muscle. If we could get even more specific and find out what type of fat, that would give us more information about other [possible] diseases that ... need to be taken into account.”
This research was funded by the Institute of Sciences, Benemérita Autonomous University of Puebla. The funders had no role in the design of the study; in the collection, analyses, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results. Dr. Alatorre-Cruz and Dr. Álvarez have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to researchers from the Institute of Sciences at Benemérita Autonomous University of Puebla, Puebla, Mexico.
Their study, published in the journal Foods, is the first to produce tables showing the phenolic content of Mexican dishes. Physicians and nutritionists can use this information as a reference tool when drawing up diet recommendations for patients who could benefit from a higher intake of phenolic compounds (PC).
“Up until now, there hasn’t been a table that we – nutritionists and physicians – could look at and see exactly which foods were richest in these compounds. In the United States, European countries, Asian countries – they’ve all had food tables; in Mexico, we didn’t,” said lead author Julia Alatorre-Cruz, PhD, a biological scientist and postdoctoral researcher at BUAP. “So, it’s a fairly innovative contribution. As a bonus, the information can be used to analyze the relationship between diet and noncommunicable diseases in the Mexican population.”
In recent years, nutrition science has focused on counteracting nutrient deficiency and some diseases by identifying active-food components. Diet offers the possibility to improve the patient’s health conditions by using these components or functional food.
PCs are a diverse group of plant micronutrients, some of which modulate physiologic and molecular pathways involved in energy metabolism. They can act by different mechanisms; the most important of them are conducted by anti-inflammatory, antioxidant activities, and are antiallergic.
Moreover, recent studies explain how PCs positively affect certain illnesses, such as obesity, diabetes, cardiovascular diseases, thrombocytopenia, and metabolic syndrome. Several common features characterize these pathologies – among them are the redox balance and a notable inflammatory response that strongly alters the biochemical and functional characteristics of the affected tissues.
Traditional Mexican food is characterized by grains, tubers, legumes, vegetables, and spices, most of which are rich in PCs. However, the Mexican diet has changed over the past decades because traditional food has been replaced with ultraprocessed food with high-caloric values. Moreover, some vegetables and fruits are preferably consumed after processing, which affects the quantity, quality, and bioavailability of the PCs. In addition, diseases associated with eating habits have increased by more than 27% in the Mexican population.
The objective of the study was to determine whether participants with a higher PC intake from beverages or Mexican dishes have better health conditions than those with a lower intake.
A total of 973 adults (798 females, 175 males) aged 18-79 years were enrolled in this cross-sectional study. The data were obtained from a validated, self-administered food consumption survey that was posted on social media (Facebook) or sent via WhatsApp or email. In one section, there was a list of fruits, vegetables, cereals, legumes, seeds, spices, beverages, and Mexican dishes. The participants were asked to indicate how often in the past month they had consumed these items. There were also sections for providing identification data (for example, age, sex, marital status), height and weight information, and medical history.
“The study was carried out during the pandemic, so that limited contact with the participants,” said Dr. Alatorre-Cruz. “Not being able to directly interact with them was a challenge. For example, we would have liked to have taken those anthropometric measurements ourselves.”
The researchers performed K-means clustering to determine the participant’s health-condition level, resulting in two groups: those with less diseases (LD, n = 649) and those with more diseases (MD, n = 324).
Using the food biochemistry composition reported in multiple papers, the researchers computed the average total phenolic compounds (TPC) for each item listed in the survey. For Mexican dishes, they added the TPC of each recipe’s ingredient, then recalculated to come up with TPC for an individual portion of each recipe (TPCr). To analyze the results of the LD group and of the MD group, phenolic compounds intake of recipe was calculated for each participant.
To Dr. Alatorre-Cruz, the biggest challenge was determining the content of compounds in traditional dishes. “Extensive, in-depth research was done to gather as much information as possible about all of the foods – especially about local and regional ones, because we have such a wide variety – looking to see where that information would match up with the exact method with which the compounds of interest were extracted.”
As expected, the team found that food with high PC was associated with a better health condition. However, the consumption of beverages and Mexican dishes was lower than their expectations. As noted in the article, the Mexican diet has changed over the last decades because traditional food has been replaced with ultraprocessed food with high-caloric values.
The authors suggest that their data confirm the alarming changes previously reported in the Mexican diet – changes possibly attributable to the increased influence of other countries via social media and economic globalization. However, they also found that beans, corn (mainly tortilla), and nopal intake remained preserved in the Mexican eating habits.
Their statistical analyses revealed that sex, age, and education seem to play a role in the presence or absence of diseases in the Mexican cohort. Men, participants over age 29 years, and those with lower levels of education had more diseases.
Dr. Alatorre-Cruz told this news organization that the Foods article opens new lines of research for the group to pursue. “One future proposal looks to enroll patients with conditions where there’s an increase in oxidative stress – which we know has quite a detrimental effect – and look into possible links with their diet. But now, maybe the focus can be on patients with a single condition and using these traditional-food tables to find links. We also want to know more about the molecular or biochemical mechanisms that are modulating the association that we saw first in animal models.”
Laura Álvarez, MD, a specialist in clinical nutrition, is the founder of the NUTRIENT project, which focuses on nutrition and strength training. In her opinion, the study extols the benefits of Mexican food. “I’ve always thought that people have this idea that Mexican food isn’t good. For example, thinking that it’s very high in fat. But in reality, Mexican food is very rich in nutrients.”
She added that research should be broadened, by enrolling a larger group of participants and evaluating not only a greater number and a greater variety of Mexican dishes, and new variables as well. “I think that body composition could be taken into account. In this study, they took the BMI into account to assess overweight and obesity, but ... the BMI doesn’t tell us much. Overweight can be due to fat or due to muscle. If we could get even more specific and find out what type of fat, that would give us more information about other [possible] diseases that ... need to be taken into account.”
This research was funded by the Institute of Sciences, Benemérita Autonomous University of Puebla. The funders had no role in the design of the study; in the collection, analyses, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results. Dr. Alatorre-Cruz and Dr. Álvarez have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to researchers from the Institute of Sciences at Benemérita Autonomous University of Puebla, Puebla, Mexico.
Their study, published in the journal Foods, is the first to produce tables showing the phenolic content of Mexican dishes. Physicians and nutritionists can use this information as a reference tool when drawing up diet recommendations for patients who could benefit from a higher intake of phenolic compounds (PC).
“Up until now, there hasn’t been a table that we – nutritionists and physicians – could look at and see exactly which foods were richest in these compounds. In the United States, European countries, Asian countries – they’ve all had food tables; in Mexico, we didn’t,” said lead author Julia Alatorre-Cruz, PhD, a biological scientist and postdoctoral researcher at BUAP. “So, it’s a fairly innovative contribution. As a bonus, the information can be used to analyze the relationship between diet and noncommunicable diseases in the Mexican population.”
In recent years, nutrition science has focused on counteracting nutrient deficiency and some diseases by identifying active-food components. Diet offers the possibility to improve the patient’s health conditions by using these components or functional food.
PCs are a diverse group of plant micronutrients, some of which modulate physiologic and molecular pathways involved in energy metabolism. They can act by different mechanisms; the most important of them are conducted by anti-inflammatory, antioxidant activities, and are antiallergic.
Moreover, recent studies explain how PCs positively affect certain illnesses, such as obesity, diabetes, cardiovascular diseases, thrombocytopenia, and metabolic syndrome. Several common features characterize these pathologies – among them are the redox balance and a notable inflammatory response that strongly alters the biochemical and functional characteristics of the affected tissues.
Traditional Mexican food is characterized by grains, tubers, legumes, vegetables, and spices, most of which are rich in PCs. However, the Mexican diet has changed over the past decades because traditional food has been replaced with ultraprocessed food with high-caloric values. Moreover, some vegetables and fruits are preferably consumed after processing, which affects the quantity, quality, and bioavailability of the PCs. In addition, diseases associated with eating habits have increased by more than 27% in the Mexican population.
The objective of the study was to determine whether participants with a higher PC intake from beverages or Mexican dishes have better health conditions than those with a lower intake.
A total of 973 adults (798 females, 175 males) aged 18-79 years were enrolled in this cross-sectional study. The data were obtained from a validated, self-administered food consumption survey that was posted on social media (Facebook) or sent via WhatsApp or email. In one section, there was a list of fruits, vegetables, cereals, legumes, seeds, spices, beverages, and Mexican dishes. The participants were asked to indicate how often in the past month they had consumed these items. There were also sections for providing identification data (for example, age, sex, marital status), height and weight information, and medical history.
“The study was carried out during the pandemic, so that limited contact with the participants,” said Dr. Alatorre-Cruz. “Not being able to directly interact with them was a challenge. For example, we would have liked to have taken those anthropometric measurements ourselves.”
The researchers performed K-means clustering to determine the participant’s health-condition level, resulting in two groups: those with less diseases (LD, n = 649) and those with more diseases (MD, n = 324).
Using the food biochemistry composition reported in multiple papers, the researchers computed the average total phenolic compounds (TPC) for each item listed in the survey. For Mexican dishes, they added the TPC of each recipe’s ingredient, then recalculated to come up with TPC for an individual portion of each recipe (TPCr). To analyze the results of the LD group and of the MD group, phenolic compounds intake of recipe was calculated for each participant.
To Dr. Alatorre-Cruz, the biggest challenge was determining the content of compounds in traditional dishes. “Extensive, in-depth research was done to gather as much information as possible about all of the foods – especially about local and regional ones, because we have such a wide variety – looking to see where that information would match up with the exact method with which the compounds of interest were extracted.”
As expected, the team found that food with high PC was associated with a better health condition. However, the consumption of beverages and Mexican dishes was lower than their expectations. As noted in the article, the Mexican diet has changed over the last decades because traditional food has been replaced with ultraprocessed food with high-caloric values.
The authors suggest that their data confirm the alarming changes previously reported in the Mexican diet – changes possibly attributable to the increased influence of other countries via social media and economic globalization. However, they also found that beans, corn (mainly tortilla), and nopal intake remained preserved in the Mexican eating habits.
Their statistical analyses revealed that sex, age, and education seem to play a role in the presence or absence of diseases in the Mexican cohort. Men, participants over age 29 years, and those with lower levels of education had more diseases.
Dr. Alatorre-Cruz told this news organization that the Foods article opens new lines of research for the group to pursue. “One future proposal looks to enroll patients with conditions where there’s an increase in oxidative stress – which we know has quite a detrimental effect – and look into possible links with their diet. But now, maybe the focus can be on patients with a single condition and using these traditional-food tables to find links. We also want to know more about the molecular or biochemical mechanisms that are modulating the association that we saw first in animal models.”
Laura Álvarez, MD, a specialist in clinical nutrition, is the founder of the NUTRIENT project, which focuses on nutrition and strength training. In her opinion, the study extols the benefits of Mexican food. “I’ve always thought that people have this idea that Mexican food isn’t good. For example, thinking that it’s very high in fat. But in reality, Mexican food is very rich in nutrients.”
She added that research should be broadened, by enrolling a larger group of participants and evaluating not only a greater number and a greater variety of Mexican dishes, and new variables as well. “I think that body composition could be taken into account. In this study, they took the BMI into account to assess overweight and obesity, but ... the BMI doesn’t tell us much. Overweight can be due to fat or due to muscle. If we could get even more specific and find out what type of fat, that would give us more information about other [possible] diseases that ... need to be taken into account.”
This research was funded by the Institute of Sciences, Benemérita Autonomous University of Puebla. The funders had no role in the design of the study; in the collection, analyses, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results. Dr. Alatorre-Cruz and Dr. Álvarez have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM FOODS
What's your diagnosis?
Answer to ‘What’s your diagnosis?’: Gastric adenocarcinoma and proximal polyposis of the stomach syndrome.
Fundic gland polyps (FGPs) are the most common gastric polyps and when occurring in the sporadic setting are typically benign; however, FGPs that occur in gastrointestinal polyposis syndromes such as familial adenomatosis polyposis can progress to adenocarcinoma and require surveillance. Therefore, it is important to distinguish sporadic versus syndromic fundic gland polyposis. Gastric adenocarcinoma and proximal polyposis of the stomach is a recently described condition that significantly increases the risk of developing invasive gastric adenocarcinoma from FGPs. Diagnostic criteria include (1) gastric polyposis restricted to the body and fundus with no small bowel or colonic involvement, (2) >100 gastric polyps or >30 polyps in a first-degree relative, (3) histology consistent with FGP with areas of dysplasia, (4) a family history consistent with an autosomal-dominant pattern of inheritance, and (5) exclusion of other syndromes and proton pump inhibitor use.1 Unlike familial adenomatosis polyposis, the polyposis is restricted to the oxyntic mucosa of the gastric body and fundus with sparing of the gastric antrum, small bowel, and colon. The genetic basis of the disease has been attributed to a point mutation in the APC gene promotor IB region leading to a loss of tumor suppressor function.2 Typical histology shows large FGPs with areas of low-grade and high-grade dysplasia, as seen in our patient.
There are few data on the natural history of gastric adenocarcinoma and proximal polyposis of the stomach, but effective surveillance is limited by the degree of polyposis. There are multiple reports of hidden adenocarcinoma on surgically resected specimens, as well as rapid progression to metastatic adenocarcinoma despite adequate diagnosis and surveillance.1,3 Therefore, total gastrectomy should be offered to patients who are surgical candidates. Our patient underwent genetic testing that revealed a point mutation in the APC promotor IB. He declined surgical intervention and opted for surveillance endoscopy every 6 months.
References
1. Worthley D.L. et al. Gut. 2012;61:774-9
2. Li J et al. Am J Hum Genet. 2016;98:830-42
3. Rudloff U. Clin Exp Gastroenterol. 2018;11:447-59
Answer to ‘What’s your diagnosis?’: Gastric adenocarcinoma and proximal polyposis of the stomach syndrome.
Fundic gland polyps (FGPs) are the most common gastric polyps and when occurring in the sporadic setting are typically benign; however, FGPs that occur in gastrointestinal polyposis syndromes such as familial adenomatosis polyposis can progress to adenocarcinoma and require surveillance. Therefore, it is important to distinguish sporadic versus syndromic fundic gland polyposis. Gastric adenocarcinoma and proximal polyposis of the stomach is a recently described condition that significantly increases the risk of developing invasive gastric adenocarcinoma from FGPs. Diagnostic criteria include (1) gastric polyposis restricted to the body and fundus with no small bowel or colonic involvement, (2) >100 gastric polyps or >30 polyps in a first-degree relative, (3) histology consistent with FGP with areas of dysplasia, (4) a family history consistent with an autosomal-dominant pattern of inheritance, and (5) exclusion of other syndromes and proton pump inhibitor use.1 Unlike familial adenomatosis polyposis, the polyposis is restricted to the oxyntic mucosa of the gastric body and fundus with sparing of the gastric antrum, small bowel, and colon. The genetic basis of the disease has been attributed to a point mutation in the APC gene promotor IB region leading to a loss of tumor suppressor function.2 Typical histology shows large FGPs with areas of low-grade and high-grade dysplasia, as seen in our patient.
There are few data on the natural history of gastric adenocarcinoma and proximal polyposis of the stomach, but effective surveillance is limited by the degree of polyposis. There are multiple reports of hidden adenocarcinoma on surgically resected specimens, as well as rapid progression to metastatic adenocarcinoma despite adequate diagnosis and surveillance.1,3 Therefore, total gastrectomy should be offered to patients who are surgical candidates. Our patient underwent genetic testing that revealed a point mutation in the APC promotor IB. He declined surgical intervention and opted for surveillance endoscopy every 6 months.
References
1. Worthley D.L. et al. Gut. 2012;61:774-9
2. Li J et al. Am J Hum Genet. 2016;98:830-42
3. Rudloff U. Clin Exp Gastroenterol. 2018;11:447-59
Answer to ‘What’s your diagnosis?’: Gastric adenocarcinoma and proximal polyposis of the stomach syndrome.
Fundic gland polyps (FGPs) are the most common gastric polyps and when occurring in the sporadic setting are typically benign; however, FGPs that occur in gastrointestinal polyposis syndromes such as familial adenomatosis polyposis can progress to adenocarcinoma and require surveillance. Therefore, it is important to distinguish sporadic versus syndromic fundic gland polyposis. Gastric adenocarcinoma and proximal polyposis of the stomach is a recently described condition that significantly increases the risk of developing invasive gastric adenocarcinoma from FGPs. Diagnostic criteria include (1) gastric polyposis restricted to the body and fundus with no small bowel or colonic involvement, (2) >100 gastric polyps or >30 polyps in a first-degree relative, (3) histology consistent with FGP with areas of dysplasia, (4) a family history consistent with an autosomal-dominant pattern of inheritance, and (5) exclusion of other syndromes and proton pump inhibitor use.1 Unlike familial adenomatosis polyposis, the polyposis is restricted to the oxyntic mucosa of the gastric body and fundus with sparing of the gastric antrum, small bowel, and colon. The genetic basis of the disease has been attributed to a point mutation in the APC gene promotor IB region leading to a loss of tumor suppressor function.2 Typical histology shows large FGPs with areas of low-grade and high-grade dysplasia, as seen in our patient.
There are few data on the natural history of gastric adenocarcinoma and proximal polyposis of the stomach, but effective surveillance is limited by the degree of polyposis. There are multiple reports of hidden adenocarcinoma on surgically resected specimens, as well as rapid progression to metastatic adenocarcinoma despite adequate diagnosis and surveillance.1,3 Therefore, total gastrectomy should be offered to patients who are surgical candidates. Our patient underwent genetic testing that revealed a point mutation in the APC promotor IB. He declined surgical intervention and opted for surveillance endoscopy every 6 months.
References
1. Worthley D.L. et al. Gut. 2012;61:774-9
2. Li J et al. Am J Hum Genet. 2016;98:830-42
3. Rudloff U. Clin Exp Gastroenterol. 2018;11:447-59
A 72-year-old man with compensated cirrhosis owing to autoimmune hepatitis presented for evaluation of an indeterminate gastric lesion found during an otherwise normal endoscopic retrograde cholangiopancreatography performed for incidental ductal dilation seen on cross-sectional imaging. He did not endorse any abdominal pain, dyspepsia, or weight loss and was not on a proton pump inhibitor. Family history was notable for a daughter diagnosed with metastatic gastric adenocarcinoma at the age of 44 years.
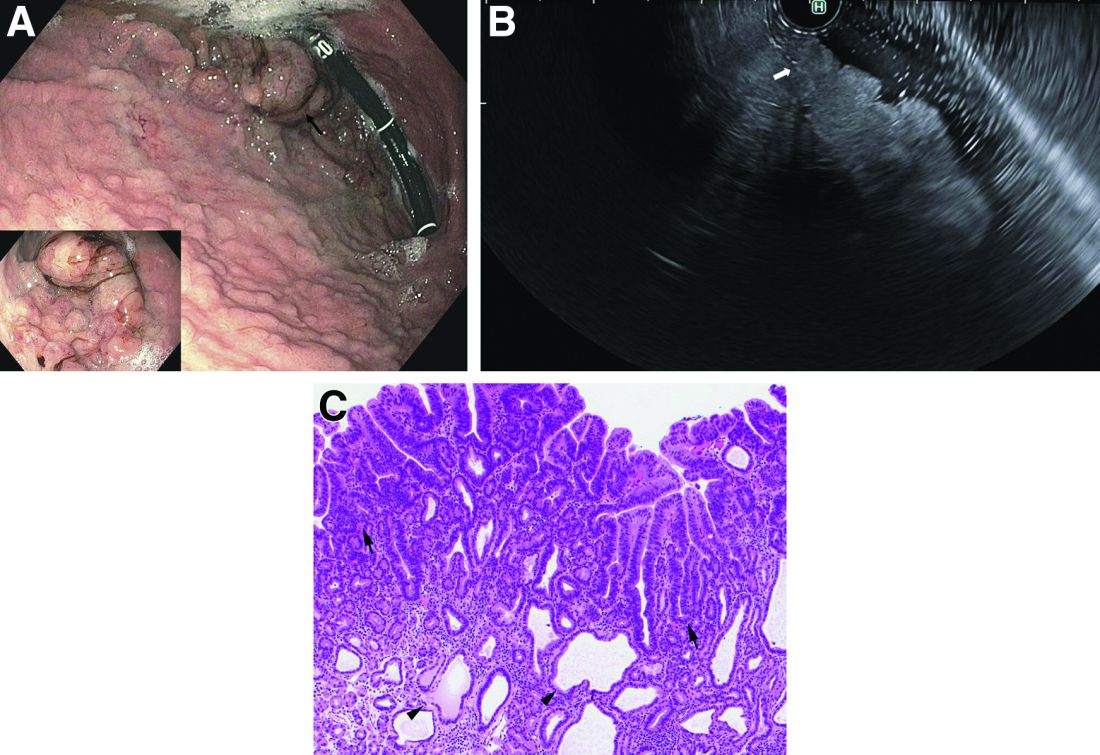
Upper endoscopy showed innumerable sessile polyps of variable size carpeting the gastric body and fundus (Figure A) with a large, mound-like mass lesion in the fundus (Figure A, arrow and inset). Echoendoscopy revealed a hypoechoic, noncircumferential mass restricted to the mucosal surface with well-defined borders (Figure B, arrow). A technically challenging, piecemeal endoscopic mucosal resection was performed. The patient also underwent a colonoscopy that was unremarkable. Pathology of the gastric lesion was consistent with a fundic gland polyp (Figure C, arrowheads) containing low-grade and high-grade dysplasia (Figure C, arrows).
What is the most likely diagnosis?
Ancient plague, cyclical pandemics … history lesson?
Even the plague wanted to visit Stonehenge
We’re about to blow your mind: The history you learned in school was often inaccurate. Shocking, we know, so we’ll give you a minute to process this incredible news.
Better? Good. Now, let’s look back at high school European history. The Black Death, specifically. The common narrative is that the Mongols, while besieging a Crimean city belonging to the Genoese, catapulted dead bodies infected with some mystery disease that turned out to be the plague. The Genoese then brought the plague back to Italy, and from there, we all know the rest of the story.
The Black Death was certainly extremely important to the development of modern Europe as we know it, but the history books gloss over the much longer history of the plague. Yersinia pestis did not suddenly appear unbidden in a Mongol war camp in 1347. The Black Death wasn’t even the first horrific, continent-wide pandemic caused by the plague; the Plague of Justinian 800 years earlier crippled the Byzantine Empire during an expansionist phase and killed anywhere between 15 million and 100 million.
Today, though, LOTME looks even deeper into history, nearly beyond even history itself, back into the depths of early Bronze Age northern Europe. Specifically, to two ancient burial sites in England, where researchers have identified three 4,000-year-old cases of Y. pestis, the first recorded incidence of the disease in Britain.
Two of the individuals, identified through analysis of dental pulp, were young children buried at a mass grave in Somerset, while the third, a middle-aged woman, was found in a ring cairn in Cumbria. These sites are hundreds of miles apart, yet carbon dating suggests all three people lived and died at roughly the same time. The strain found is very similar to other samples of plague found across central and western Europe starting around 3,000 BCE, suggesting a single, easily spread disease affecting a large area in a relatively small period of time. In other words, a pandemic. Even in these ancient times, the world was connected. Not even the island of Britain could escape.
Beyond that though, the research helps confirm the cyclical nature of the plague; over time, it loses its effectiveness and goes into hiding, only to mutate and come roaring back. This is a story with absolutely no relevance at all to the modern world. Nope, no plagues or pandemics going around right now, no viruses fading into the background in any way. What a ridiculous inference to make.
Uncovering the invisible with artificial intelligence
This week in “What Else Can AI Do?” new research shows that a computer program can reveal brain injury that couldn’t be seen before with typical MRI.
The hot new AI, birthed by researchers at New York University, could potentially be a game changer by linking repeated head impacts with tiny, structural changes in the brains of athletes who have not been diagnosed with a concussion. By using machine learning to train the AI, the researchers were, for the first time, able to distinguish the brain of athletes who played contact sports (football, soccer, lacrosse) from those participating in noncontact sports such as baseball, basketball, and cross-country.
How did they do it? The investigators “designed statistical techniques that gave their computer program the ability to ‘learn’ how to predict exposure to repeated head impacts using mathematical models,” they explained in a written statement. Adding in data from the MRI scans of 81 male athletes with no known concussion diagnosis and the ability to identify unusual brain features between athletes with and without head trauma allowed the AI to predict results with accuracy even Miss Cleo would envy.
“This method may provide an important diagnostic tool not only for concussion, but also for detecting the damage that stems from subtler and more frequent head impacts,” said lead author Junbo Chen, an engineering doctoral candidate at NYU. That could make this new AI a valuable asset to science and medicine.
There are many things the human brain can do that AI can’t, and delegation could be one of them. Examining the data that represent the human brain in minute detail? Maybe we leave that to the machine.
Talk about your field promotions
If you’re a surgeon doing an amputation, the list of possible assistants pretty much starts and ends in only one place: Not the closest available janitor.
That may seem like an oddly obvious thing for us to say, but there’s at least one former Mainz (Germany) University Hospital physician who really needed to get this bit of advice before he attempted an unassisted toe amputation back in October of 2020. Yes, that does seem like kind of a long time ago for us to be reporting it now, but the details of the incident only just came to light a few days ago, thanks to German public broadcaster SWR.
Since it was just a toe, the surgeon thought he could perform the operation without any help. The toe, unfortunately, had other plans. The partially anesthetized patient got restless in the operating room, but with no actual trained nurse in the vicinity, the surgeon asked the closest available person – that would be the janitor – to lend a hand.
The surgical manager heard about these goings-on and got to the operating room too late to stop the procedure but soon enough to see the cleaning staffer “at the operating table with a bloody suction cup and a bloody compress in their hands,” SWR recently reported.
The incident was reported to the hospital’s medical director and the surgeon was fired, but since the patient experienced no complications not much fuss was made about it at the time.
Well, guess what? It’s toe-tally our job to make a fuss about these kinds of things. Or could it be that our job, much like the surgeon’s employment and the patient’s digit, is here toe-day and gone toe-morrow?
Even the plague wanted to visit Stonehenge
We’re about to blow your mind: The history you learned in school was often inaccurate. Shocking, we know, so we’ll give you a minute to process this incredible news.
Better? Good. Now, let’s look back at high school European history. The Black Death, specifically. The common narrative is that the Mongols, while besieging a Crimean city belonging to the Genoese, catapulted dead bodies infected with some mystery disease that turned out to be the plague. The Genoese then brought the plague back to Italy, and from there, we all know the rest of the story.
The Black Death was certainly extremely important to the development of modern Europe as we know it, but the history books gloss over the much longer history of the plague. Yersinia pestis did not suddenly appear unbidden in a Mongol war camp in 1347. The Black Death wasn’t even the first horrific, continent-wide pandemic caused by the plague; the Plague of Justinian 800 years earlier crippled the Byzantine Empire during an expansionist phase and killed anywhere between 15 million and 100 million.
Today, though, LOTME looks even deeper into history, nearly beyond even history itself, back into the depths of early Bronze Age northern Europe. Specifically, to two ancient burial sites in England, where researchers have identified three 4,000-year-old cases of Y. pestis, the first recorded incidence of the disease in Britain.
Two of the individuals, identified through analysis of dental pulp, were young children buried at a mass grave in Somerset, while the third, a middle-aged woman, was found in a ring cairn in Cumbria. These sites are hundreds of miles apart, yet carbon dating suggests all three people lived and died at roughly the same time. The strain found is very similar to other samples of plague found across central and western Europe starting around 3,000 BCE, suggesting a single, easily spread disease affecting a large area in a relatively small period of time. In other words, a pandemic. Even in these ancient times, the world was connected. Not even the island of Britain could escape.
Beyond that though, the research helps confirm the cyclical nature of the plague; over time, it loses its effectiveness and goes into hiding, only to mutate and come roaring back. This is a story with absolutely no relevance at all to the modern world. Nope, no plagues or pandemics going around right now, no viruses fading into the background in any way. What a ridiculous inference to make.
Uncovering the invisible with artificial intelligence
This week in “What Else Can AI Do?” new research shows that a computer program can reveal brain injury that couldn’t be seen before with typical MRI.
The hot new AI, birthed by researchers at New York University, could potentially be a game changer by linking repeated head impacts with tiny, structural changes in the brains of athletes who have not been diagnosed with a concussion. By using machine learning to train the AI, the researchers were, for the first time, able to distinguish the brain of athletes who played contact sports (football, soccer, lacrosse) from those participating in noncontact sports such as baseball, basketball, and cross-country.
How did they do it? The investigators “designed statistical techniques that gave their computer program the ability to ‘learn’ how to predict exposure to repeated head impacts using mathematical models,” they explained in a written statement. Adding in data from the MRI scans of 81 male athletes with no known concussion diagnosis and the ability to identify unusual brain features between athletes with and without head trauma allowed the AI to predict results with accuracy even Miss Cleo would envy.
“This method may provide an important diagnostic tool not only for concussion, but also for detecting the damage that stems from subtler and more frequent head impacts,” said lead author Junbo Chen, an engineering doctoral candidate at NYU. That could make this new AI a valuable asset to science and medicine.
There are many things the human brain can do that AI can’t, and delegation could be one of them. Examining the data that represent the human brain in minute detail? Maybe we leave that to the machine.
Talk about your field promotions
If you’re a surgeon doing an amputation, the list of possible assistants pretty much starts and ends in only one place: Not the closest available janitor.
That may seem like an oddly obvious thing for us to say, but there’s at least one former Mainz (Germany) University Hospital physician who really needed to get this bit of advice before he attempted an unassisted toe amputation back in October of 2020. Yes, that does seem like kind of a long time ago for us to be reporting it now, but the details of the incident only just came to light a few days ago, thanks to German public broadcaster SWR.
Since it was just a toe, the surgeon thought he could perform the operation without any help. The toe, unfortunately, had other plans. The partially anesthetized patient got restless in the operating room, but with no actual trained nurse in the vicinity, the surgeon asked the closest available person – that would be the janitor – to lend a hand.
The surgical manager heard about these goings-on and got to the operating room too late to stop the procedure but soon enough to see the cleaning staffer “at the operating table with a bloody suction cup and a bloody compress in their hands,” SWR recently reported.
The incident was reported to the hospital’s medical director and the surgeon was fired, but since the patient experienced no complications not much fuss was made about it at the time.
Well, guess what? It’s toe-tally our job to make a fuss about these kinds of things. Or could it be that our job, much like the surgeon’s employment and the patient’s digit, is here toe-day and gone toe-morrow?
Even the plague wanted to visit Stonehenge
We’re about to blow your mind: The history you learned in school was often inaccurate. Shocking, we know, so we’ll give you a minute to process this incredible news.
Better? Good. Now, let’s look back at high school European history. The Black Death, specifically. The common narrative is that the Mongols, while besieging a Crimean city belonging to the Genoese, catapulted dead bodies infected with some mystery disease that turned out to be the plague. The Genoese then brought the plague back to Italy, and from there, we all know the rest of the story.
The Black Death was certainly extremely important to the development of modern Europe as we know it, but the history books gloss over the much longer history of the plague. Yersinia pestis did not suddenly appear unbidden in a Mongol war camp in 1347. The Black Death wasn’t even the first horrific, continent-wide pandemic caused by the plague; the Plague of Justinian 800 years earlier crippled the Byzantine Empire during an expansionist phase and killed anywhere between 15 million and 100 million.
Today, though, LOTME looks even deeper into history, nearly beyond even history itself, back into the depths of early Bronze Age northern Europe. Specifically, to two ancient burial sites in England, where researchers have identified three 4,000-year-old cases of Y. pestis, the first recorded incidence of the disease in Britain.
Two of the individuals, identified through analysis of dental pulp, were young children buried at a mass grave in Somerset, while the third, a middle-aged woman, was found in a ring cairn in Cumbria. These sites are hundreds of miles apart, yet carbon dating suggests all three people lived and died at roughly the same time. The strain found is very similar to other samples of plague found across central and western Europe starting around 3,000 BCE, suggesting a single, easily spread disease affecting a large area in a relatively small period of time. In other words, a pandemic. Even in these ancient times, the world was connected. Not even the island of Britain could escape.
Beyond that though, the research helps confirm the cyclical nature of the plague; over time, it loses its effectiveness and goes into hiding, only to mutate and come roaring back. This is a story with absolutely no relevance at all to the modern world. Nope, no plagues or pandemics going around right now, no viruses fading into the background in any way. What a ridiculous inference to make.
Uncovering the invisible with artificial intelligence
This week in “What Else Can AI Do?” new research shows that a computer program can reveal brain injury that couldn’t be seen before with typical MRI.
The hot new AI, birthed by researchers at New York University, could potentially be a game changer by linking repeated head impacts with tiny, structural changes in the brains of athletes who have not been diagnosed with a concussion. By using machine learning to train the AI, the researchers were, for the first time, able to distinguish the brain of athletes who played contact sports (football, soccer, lacrosse) from those participating in noncontact sports such as baseball, basketball, and cross-country.
How did they do it? The investigators “designed statistical techniques that gave their computer program the ability to ‘learn’ how to predict exposure to repeated head impacts using mathematical models,” they explained in a written statement. Adding in data from the MRI scans of 81 male athletes with no known concussion diagnosis and the ability to identify unusual brain features between athletes with and without head trauma allowed the AI to predict results with accuracy even Miss Cleo would envy.
“This method may provide an important diagnostic tool not only for concussion, but also for detecting the damage that stems from subtler and more frequent head impacts,” said lead author Junbo Chen, an engineering doctoral candidate at NYU. That could make this new AI a valuable asset to science and medicine.
There are many things the human brain can do that AI can’t, and delegation could be one of them. Examining the data that represent the human brain in minute detail? Maybe we leave that to the machine.
Talk about your field promotions
If you’re a surgeon doing an amputation, the list of possible assistants pretty much starts and ends in only one place: Not the closest available janitor.
That may seem like an oddly obvious thing for us to say, but there’s at least one former Mainz (Germany) University Hospital physician who really needed to get this bit of advice before he attempted an unassisted toe amputation back in October of 2020. Yes, that does seem like kind of a long time ago for us to be reporting it now, but the details of the incident only just came to light a few days ago, thanks to German public broadcaster SWR.
Since it was just a toe, the surgeon thought he could perform the operation without any help. The toe, unfortunately, had other plans. The partially anesthetized patient got restless in the operating room, but with no actual trained nurse in the vicinity, the surgeon asked the closest available person – that would be the janitor – to lend a hand.
The surgical manager heard about these goings-on and got to the operating room too late to stop the procedure but soon enough to see the cleaning staffer “at the operating table with a bloody suction cup and a bloody compress in their hands,” SWR recently reported.
The incident was reported to the hospital’s medical director and the surgeon was fired, but since the patient experienced no complications not much fuss was made about it at the time.
Well, guess what? It’s toe-tally our job to make a fuss about these kinds of things. Or could it be that our job, much like the surgeon’s employment and the patient’s digit, is here toe-day and gone toe-morrow?
Brooklyn gastroenterologist: Good listening skills make a doctor a better teacher, person
After the 2004 Indian Ocean earthquake and tsunami, he traveled to his home country of Sri Lanka to help people who were in need and establish an orphanage. He has applied his skills as a gastroenterologist in the U.S. military and the New York City Police Department.
He was in New York during the 9-11 terrorist attacks. To this day, he treats patients with residual GI problems and precancerous changes associated with 9-11. “I’m involved in screening those people who were the first responders referred by the NYPD,” he says.
This year Dr. Iswara earned the Distinguished Clinician Award in Private Practice from the American Gastroenterological Association. “He puts his patients first in every endeavor – and every question that he asks with regards to research and education is linked to the ultimate measuring stick of improving patient care,” according to an AGA announcement of the award.
When dealing with patients and colleagues, he offers this simple pearl of advice: Listen and then listen some more.
“Once you listen more, you can find out their issues much more in depth, and you can give a satisfying answer to them and their problems. Listening is a kindness and a compassionate thing. It not only makes you become a better teacher, but a better person,” said Dr. Iswara, attending gastroenterologist at Maimonides Medical Center in New York.
In an interview, he talked more in depth about his GI beginnings, his role as a mentor, and why he always starts the day with a prayer. He also confided about the useful time management habit kept from his military days that gives him energy.
Question: What gives you joy in day-to-day practice?
Dr. Iswara: One of the main joys is my colleagues, coworkers, fellows, and my patients. The patients come No. 1. As I walk into my practice area or in the hospital, there is a sense of inner happiness in my mind to see the smiles of the patients and the greetings I get from the patients and all the coworkers. I also see smiling patients with anxiety in their face, trying to get my attention to take care of them.
After I see the patient, I change to a different mode, a kind of a professional mode to give the best to the people whom I’m caring for, who are trusting me with their lives.
One thing I do in my mind before I even start the day, I do a silent prayer to guide me, to give compassionate care and safe care. I will not harm anyone who is depending on my care.
Q: Who was your mentor?
Dr. Iswara: I was lucky enough to have been trained by Baroukh El Kodsi, MD, at Maimonides Medical Center. He recently passed away and was a legend in Brooklyn. I was his first-generation trainee, and I was able to pass on my skills to my trainees. Now so many people who are in Brooklyn; they were trained by me, so it’s kind of growth by generations.
When I finished the training with Dr. Kodsi, he hired me as an associate director of the GI department at Maimonides. I became the program director, then division chief, then I became a director of advanced endoscopy. All these gastroenterology procedures started after 1975 while I was doing the training, so I was one of the pioneers to bring all this new technology to our hospital. I’m still involved in fellowship education.
Q: Can we talk more about your accomplishments? Perhaps you can discuss your AGA award and what you received it for.
Dr. Iswara: I’m humbled and honored by this role, and I’ll be forever grateful to AGA for this prestigious honor at the late stage of my career.
I have been a continuous AGA member for the last 45 years. I probably have one of the longest durations of being an actively practicing gastroenterologist in Brooklyn. I’ve also done academic work, teaching so many young gastroenterologists, motivating several of them to become leading gastroenterologists.
Q: If you could describe a scene of your vision for the future, what it would it be in terms of how gastroenterology is practiced?
Dr. Iswara: I’d like to see the newer generation practice more of a clinical medicine than technical medicine. Sometimes when I see the young people, they sit in front of the computer more than talking and touching the patient. There has to be some sort of a balance where the newer people should be taught more bedside personal care, touching the patient, looking at the patient’s face. They are kind of under pressure to write longer notes than to examine the patient, so I think this has to change.
Q: Describe how you would spend a free Saturday afternoon.
Dr. Iswara: When I was in the military, I was told that to prevent battle fatigue you had to take a rest. I really try to take a rest almost 2 hours every day in the daytime. This rejuvenates me.
We live in New York, and I love to go to shows, especially magic shows. I love magic and illusion.
On free Saturday evenings, I also spend time with my grandchildren in the city, watching them in their baseball, soccer, swimming, and other activities. I love to spend time with them.
Lightning round
Texting or talking?
Texting
Favorite city in the U.S. besides the one you live?
Naples, Fla.
Favorite breakfast?
Pancakes
Dark Chocolate or milk chocolate?
Cadbury from England
Last movie you watched?
“To Sir, With Love”
After the 2004 Indian Ocean earthquake and tsunami, he traveled to his home country of Sri Lanka to help people who were in need and establish an orphanage. He has applied his skills as a gastroenterologist in the U.S. military and the New York City Police Department.
He was in New York during the 9-11 terrorist attacks. To this day, he treats patients with residual GI problems and precancerous changes associated with 9-11. “I’m involved in screening those people who were the first responders referred by the NYPD,” he says.
This year Dr. Iswara earned the Distinguished Clinician Award in Private Practice from the American Gastroenterological Association. “He puts his patients first in every endeavor – and every question that he asks with regards to research and education is linked to the ultimate measuring stick of improving patient care,” according to an AGA announcement of the award.
When dealing with patients and colleagues, he offers this simple pearl of advice: Listen and then listen some more.
“Once you listen more, you can find out their issues much more in depth, and you can give a satisfying answer to them and their problems. Listening is a kindness and a compassionate thing. It not only makes you become a better teacher, but a better person,” said Dr. Iswara, attending gastroenterologist at Maimonides Medical Center in New York.
In an interview, he talked more in depth about his GI beginnings, his role as a mentor, and why he always starts the day with a prayer. He also confided about the useful time management habit kept from his military days that gives him energy.
Question: What gives you joy in day-to-day practice?
Dr. Iswara: One of the main joys is my colleagues, coworkers, fellows, and my patients. The patients come No. 1. As I walk into my practice area or in the hospital, there is a sense of inner happiness in my mind to see the smiles of the patients and the greetings I get from the patients and all the coworkers. I also see smiling patients with anxiety in their face, trying to get my attention to take care of them.
After I see the patient, I change to a different mode, a kind of a professional mode to give the best to the people whom I’m caring for, who are trusting me with their lives.
One thing I do in my mind before I even start the day, I do a silent prayer to guide me, to give compassionate care and safe care. I will not harm anyone who is depending on my care.
Q: Who was your mentor?
Dr. Iswara: I was lucky enough to have been trained by Baroukh El Kodsi, MD, at Maimonides Medical Center. He recently passed away and was a legend in Brooklyn. I was his first-generation trainee, and I was able to pass on my skills to my trainees. Now so many people who are in Brooklyn; they were trained by me, so it’s kind of growth by generations.
When I finished the training with Dr. Kodsi, he hired me as an associate director of the GI department at Maimonides. I became the program director, then division chief, then I became a director of advanced endoscopy. All these gastroenterology procedures started after 1975 while I was doing the training, so I was one of the pioneers to bring all this new technology to our hospital. I’m still involved in fellowship education.
Q: Can we talk more about your accomplishments? Perhaps you can discuss your AGA award and what you received it for.
Dr. Iswara: I’m humbled and honored by this role, and I’ll be forever grateful to AGA for this prestigious honor at the late stage of my career.
I have been a continuous AGA member for the last 45 years. I probably have one of the longest durations of being an actively practicing gastroenterologist in Brooklyn. I’ve also done academic work, teaching so many young gastroenterologists, motivating several of them to become leading gastroenterologists.
Q: If you could describe a scene of your vision for the future, what it would it be in terms of how gastroenterology is practiced?
Dr. Iswara: I’d like to see the newer generation practice more of a clinical medicine than technical medicine. Sometimes when I see the young people, they sit in front of the computer more than talking and touching the patient. There has to be some sort of a balance where the newer people should be taught more bedside personal care, touching the patient, looking at the patient’s face. They are kind of under pressure to write longer notes than to examine the patient, so I think this has to change.
Q: Describe how you would spend a free Saturday afternoon.
Dr. Iswara: When I was in the military, I was told that to prevent battle fatigue you had to take a rest. I really try to take a rest almost 2 hours every day in the daytime. This rejuvenates me.
We live in New York, and I love to go to shows, especially magic shows. I love magic and illusion.
On free Saturday evenings, I also spend time with my grandchildren in the city, watching them in their baseball, soccer, swimming, and other activities. I love to spend time with them.
Lightning round
Texting or talking?
Texting
Favorite city in the U.S. besides the one you live?
Naples, Fla.
Favorite breakfast?
Pancakes
Dark Chocolate or milk chocolate?
Cadbury from England
Last movie you watched?
“To Sir, With Love”
After the 2004 Indian Ocean earthquake and tsunami, he traveled to his home country of Sri Lanka to help people who were in need and establish an orphanage. He has applied his skills as a gastroenterologist in the U.S. military and the New York City Police Department.
He was in New York during the 9-11 terrorist attacks. To this day, he treats patients with residual GI problems and precancerous changes associated with 9-11. “I’m involved in screening those people who were the first responders referred by the NYPD,” he says.
This year Dr. Iswara earned the Distinguished Clinician Award in Private Practice from the American Gastroenterological Association. “He puts his patients first in every endeavor – and every question that he asks with regards to research and education is linked to the ultimate measuring stick of improving patient care,” according to an AGA announcement of the award.
When dealing with patients and colleagues, he offers this simple pearl of advice: Listen and then listen some more.
“Once you listen more, you can find out their issues much more in depth, and you can give a satisfying answer to them and their problems. Listening is a kindness and a compassionate thing. It not only makes you become a better teacher, but a better person,” said Dr. Iswara, attending gastroenterologist at Maimonides Medical Center in New York.
In an interview, he talked more in depth about his GI beginnings, his role as a mentor, and why he always starts the day with a prayer. He also confided about the useful time management habit kept from his military days that gives him energy.
Question: What gives you joy in day-to-day practice?
Dr. Iswara: One of the main joys is my colleagues, coworkers, fellows, and my patients. The patients come No. 1. As I walk into my practice area or in the hospital, there is a sense of inner happiness in my mind to see the smiles of the patients and the greetings I get from the patients and all the coworkers. I also see smiling patients with anxiety in their face, trying to get my attention to take care of them.
After I see the patient, I change to a different mode, a kind of a professional mode to give the best to the people whom I’m caring for, who are trusting me with their lives.
One thing I do in my mind before I even start the day, I do a silent prayer to guide me, to give compassionate care and safe care. I will not harm anyone who is depending on my care.
Q: Who was your mentor?
Dr. Iswara: I was lucky enough to have been trained by Baroukh El Kodsi, MD, at Maimonides Medical Center. He recently passed away and was a legend in Brooklyn. I was his first-generation trainee, and I was able to pass on my skills to my trainees. Now so many people who are in Brooklyn; they were trained by me, so it’s kind of growth by generations.
When I finished the training with Dr. Kodsi, he hired me as an associate director of the GI department at Maimonides. I became the program director, then division chief, then I became a director of advanced endoscopy. All these gastroenterology procedures started after 1975 while I was doing the training, so I was one of the pioneers to bring all this new technology to our hospital. I’m still involved in fellowship education.
Q: Can we talk more about your accomplishments? Perhaps you can discuss your AGA award and what you received it for.
Dr. Iswara: I’m humbled and honored by this role, and I’ll be forever grateful to AGA for this prestigious honor at the late stage of my career.
I have been a continuous AGA member for the last 45 years. I probably have one of the longest durations of being an actively practicing gastroenterologist in Brooklyn. I’ve also done academic work, teaching so many young gastroenterologists, motivating several of them to become leading gastroenterologists.
Q: If you could describe a scene of your vision for the future, what it would it be in terms of how gastroenterology is practiced?
Dr. Iswara: I’d like to see the newer generation practice more of a clinical medicine than technical medicine. Sometimes when I see the young people, they sit in front of the computer more than talking and touching the patient. There has to be some sort of a balance where the newer people should be taught more bedside personal care, touching the patient, looking at the patient’s face. They are kind of under pressure to write longer notes than to examine the patient, so I think this has to change.
Q: Describe how you would spend a free Saturday afternoon.
Dr. Iswara: When I was in the military, I was told that to prevent battle fatigue you had to take a rest. I really try to take a rest almost 2 hours every day in the daytime. This rejuvenates me.
We live in New York, and I love to go to shows, especially magic shows. I love magic and illusion.
On free Saturday evenings, I also spend time with my grandchildren in the city, watching them in their baseball, soccer, swimming, and other activities. I love to spend time with them.
Lightning round
Texting or talking?
Texting
Favorite city in the U.S. besides the one you live?
Naples, Fla.
Favorite breakfast?
Pancakes
Dark Chocolate or milk chocolate?
Cadbury from England
Last movie you watched?
“To Sir, With Love”
The power of mentorship
In a 2018 JAMA Viewpoint, Dr. Vineet Chopra, a former colleague of mine at the University of Michigan (now chair of medicine at the University of Colorado) and colleagues wrote about four archetypes of mentorship: mentor, coach, sponsor, and connector. along the way.
For me, DDW serves as an annual reminder of the power of mentorship in building and sustaining careers. Each May, trainees and early career faculty present their projects in oral or poster sessions, cheered on by their research mentors. Senior thought leaders offer career advice and guidance to more junior colleagues through structured sessions or informal conversations and facilitate introductions to new collaborators. Department chairs, division chiefs, and senior practice leaders take time to reconnect with their early mentors who believed in their potential and provided them with opportunities to take their careers to new heights. And, we see the incredible payoff of programs like AGA’s FORWARD and Future Leaders Programs in serving as springboards for career advancement and creating powerful role models and mentors for the future.
This year’s AGA presidential leadership transition served as a particularly poignant example of the power of mentorship as incoming AGA President Dr. Barbara Jung succeeded one of her early mentors, outgoing AGA President Dr. John Carethers, in this prestigious role. I hope you’ll join me in reflecting on the tremendous impact that mentors, coaches, sponsors, and connectors have had on your career, and continue to pay it forward to the next generation.
In this month’s issue, we feature several stories from DDW 2023, including summaries of the AGA presidential address and a study evaluating the impact of state Medicaid expansion on uptake of CRC screening in safety-net practices. From AGA’s flagship journals, we highlight a propensity-matched cohort study assessing the impact of pancreatic cancer surveillance of high-risk patients on important clinical outcomes and a new AGA CPU on management of extraesophageal GERD. In this month’s AGA Policy and Advocacy column, Dr. Amit Patel and Dr. Rotonya Carr review the results of a recent membership survey on policy priorities and outline the many ways you can get involved in advocacy efforts. Finally, our Member Spotlight column celebrates gastroenterologist and humanitarian Kadirawel Iswara, MD, recipient of this year’s AGA Distinguished Clinician Award in Private Practice, who is a cherished mentor to many prominent members of our field.
Megan A. Adams, M.D., J.D., MSc
Editor-in-Chief
In a 2018 JAMA Viewpoint, Dr. Vineet Chopra, a former colleague of mine at the University of Michigan (now chair of medicine at the University of Colorado) and colleagues wrote about four archetypes of mentorship: mentor, coach, sponsor, and connector. along the way.
For me, DDW serves as an annual reminder of the power of mentorship in building and sustaining careers. Each May, trainees and early career faculty present their projects in oral or poster sessions, cheered on by their research mentors. Senior thought leaders offer career advice and guidance to more junior colleagues through structured sessions or informal conversations and facilitate introductions to new collaborators. Department chairs, division chiefs, and senior practice leaders take time to reconnect with their early mentors who believed in their potential and provided them with opportunities to take their careers to new heights. And, we see the incredible payoff of programs like AGA’s FORWARD and Future Leaders Programs in serving as springboards for career advancement and creating powerful role models and mentors for the future.
This year’s AGA presidential leadership transition served as a particularly poignant example of the power of mentorship as incoming AGA President Dr. Barbara Jung succeeded one of her early mentors, outgoing AGA President Dr. John Carethers, in this prestigious role. I hope you’ll join me in reflecting on the tremendous impact that mentors, coaches, sponsors, and connectors have had on your career, and continue to pay it forward to the next generation.
In this month’s issue, we feature several stories from DDW 2023, including summaries of the AGA presidential address and a study evaluating the impact of state Medicaid expansion on uptake of CRC screening in safety-net practices. From AGA’s flagship journals, we highlight a propensity-matched cohort study assessing the impact of pancreatic cancer surveillance of high-risk patients on important clinical outcomes and a new AGA CPU on management of extraesophageal GERD. In this month’s AGA Policy and Advocacy column, Dr. Amit Patel and Dr. Rotonya Carr review the results of a recent membership survey on policy priorities and outline the many ways you can get involved in advocacy efforts. Finally, our Member Spotlight column celebrates gastroenterologist and humanitarian Kadirawel Iswara, MD, recipient of this year’s AGA Distinguished Clinician Award in Private Practice, who is a cherished mentor to many prominent members of our field.
Megan A. Adams, M.D., J.D., MSc
Editor-in-Chief
In a 2018 JAMA Viewpoint, Dr. Vineet Chopra, a former colleague of mine at the University of Michigan (now chair of medicine at the University of Colorado) and colleagues wrote about four archetypes of mentorship: mentor, coach, sponsor, and connector. along the way.
For me, DDW serves as an annual reminder of the power of mentorship in building and sustaining careers. Each May, trainees and early career faculty present their projects in oral or poster sessions, cheered on by their research mentors. Senior thought leaders offer career advice and guidance to more junior colleagues through structured sessions or informal conversations and facilitate introductions to new collaborators. Department chairs, division chiefs, and senior practice leaders take time to reconnect with their early mentors who believed in their potential and provided them with opportunities to take their careers to new heights. And, we see the incredible payoff of programs like AGA’s FORWARD and Future Leaders Programs in serving as springboards for career advancement and creating powerful role models and mentors for the future.
This year’s AGA presidential leadership transition served as a particularly poignant example of the power of mentorship as incoming AGA President Dr. Barbara Jung succeeded one of her early mentors, outgoing AGA President Dr. John Carethers, in this prestigious role. I hope you’ll join me in reflecting on the tremendous impact that mentors, coaches, sponsors, and connectors have had on your career, and continue to pay it forward to the next generation.
In this month’s issue, we feature several stories from DDW 2023, including summaries of the AGA presidential address and a study evaluating the impact of state Medicaid expansion on uptake of CRC screening in safety-net practices. From AGA’s flagship journals, we highlight a propensity-matched cohort study assessing the impact of pancreatic cancer surveillance of high-risk patients on important clinical outcomes and a new AGA CPU on management of extraesophageal GERD. In this month’s AGA Policy and Advocacy column, Dr. Amit Patel and Dr. Rotonya Carr review the results of a recent membership survey on policy priorities and outline the many ways you can get involved in advocacy efforts. Finally, our Member Spotlight column celebrates gastroenterologist and humanitarian Kadirawel Iswara, MD, recipient of this year’s AGA Distinguished Clinician Award in Private Practice, who is a cherished mentor to many prominent members of our field.
Megan A. Adams, M.D., J.D., MSc
Editor-in-Chief
Blood cancer patient takes on bias and ‘gaslighting’
Diagnosed with Hodgkin lymphoma in 2021, Ms. Ngon underwent port surgery to allow chemotherapy to be administered. Her right arm lost circulation and went numb, so she sought guidance from her blood cancer specialist. He dismissed her worries, saying that her tumors were pinching a nerve. She’d get better, he predicted, after more chemo.
“I knew in my body that something was wrong,” Ms. Ngon recalled. When the oncologist continued to downplay her concerns, she and a fellow communications specialist sat down together in the hospital lobby to draft an email to her physician. “We were trying to articulate the urgency in an email that expresses that I’m not being dramatic. We had to do it in a way that didn’t insult his intelligence: ‘Respectfully, you’re the doctor, but I know something is wrong.’ ”
In essence, Ms. Ngon was trying to be diplomatic and not trigger her oncologist’s defenses, while still convincing him to take action. Her approach to getting her doctor’s attention worked. He referred Ms. Ngon to a radiologist, who discovered that she had blood clots in her arm. Ms. Ngon then landed in the ICU for a week, as clinicians tried to break up the clots.
“I was the perfect person for this to happen to, because of my job and education. But it makes me sad because I understand I was in a fortunate position, with a background in communication. Most people don’t have that,” Ms. Ngon said.
This and other negative experiences during her medical saga inspired Ms. Ngon to partner with the Lymphoma Research Foundation in order to spread the word about unique challenges facing patients like her: people of color.
Ms. Ngon, who is Black, said her goal as a patient advocate is to “empower communities of color to speak up for themselves and hold oncologists responsible for listening and understanding differences across cultures.” And she wants to take a stand against the “gaslighting” of patients.
African Americans with hematologic disease like Ms. Ngon face a higher risk of poor outcomes than Whites, even as they are less likely than Whites to develop certain blood cancers. The reasons for this disparity aren’t clear, but researchers suspect they’re related to factors such as poverty, lack of insurance, genetics, and limited access to high-quality care.
Some researchers have blamed another factor: racism. A 2022 study sought to explain why Black and Hispanic patients with acute myeloid leukemia in urban areas have higher mortality rates than Whites, “despite more favorable genetics and younger age” (hazard ratio, 1.59, 95% confidence interval, 1.15-2.22 and HR, 1.25; 95% CI, 0.88-1.79). The study authors determined that “structural racism” – which they measured by examining segregation and “disadvantage” in neighborhoods where patients lived – accounted for nearly all of the disparities.
Ms. Ngon said her experiences and her awareness about poorer outcomes in medicine for African Americans – such as higher death rates for Black women during pregnancy – affect how she interacts with clinicians. “I automatically assume a barrier between me and my doctors, and it’s their responsibility to dismantle it.”
Making an connection with a physician can make a huge difference, she said. “I walked into my primary care doctor’s office and saw that she was a Latino woman. My guard went down, and I could feel her care for me as a human being. Whether that was because she was also a woman of color or not, I don’t know. But I did feel more cared for.”
However, Ms. Ngon could not find a Black oncologist to care for her in New York City, and that’s no surprise.
Ethnic and gender diversity remains an immense challenge in the hematology/oncology field. According to the American Society of Clinical Oncology, only about a third of oncologists are women, and the percentages identifying themselves as Black/African American and Hispanic are just 2.3% and 5.8%, respectively.
These numbers don’t seem likely to budge much any time soon. An analysis of medical students in U.S. oncology training programs from 2015-2020 found that just 3.8% identified themselves as Black/African American and 5.1% as Hispanic/Latino versus 52.15% as White and 31% as Asian/Pacific Islander/Native Hawaiian.
Ms. Ngon encountered challenges on other fronts during her cancer care. When she needed a wig during chemotherapy, a list of insurer-approved shops didn’t include any that catered to African Americans. Essentially, she said, she was being told that she couldn’t “purchase a wig from a place that makes you feel comfortable and from a woman who understand your needs as a Black woman. It needs to be from these specific shops that really don’t cater to my community.”
She also found it difficult to find fellow patients who shared her unique challenges. “I remember when I was diagnosed, I was looking through the support groups on Facebook, trying to find someone Black to ask about whether braiding my hair might stop it from falling out.”
Now, Ms. Ngon is in remission. And she’s happy with her oncologist, who’s White. “He listened to me, and he promised me that I would have the most boring recovery process ever, after everything I’d experienced. That explains a lot of why I felt so comfortable with him.”
She hopes to use her partnership with the Lymphoma Research Foundation to be a resource for people of color and alert them to the support that’s available for them. “I would love to let them know how to advocate for themselves as patients, how to trust their bodies, how to push back if they feel like they’re not getting the care that they deserve.”
Ms. Ngon would also like to see more support for medical students of color. “I hope to exist in a world one day where it wouldn’t be so hard to find an oncologist who looks like me in a city as large as this one,” she said.
As for oncologists, she urged them to “go the extra mile and really, really listen to what patients are saying. It’s easier said than done because there are natural biases in this world, and it’s hard to overcome those obstacles. But to not be heard and have to push every time. It was just exhausting to do that on top of trying to beat cancer.”
Diagnosed with Hodgkin lymphoma in 2021, Ms. Ngon underwent port surgery to allow chemotherapy to be administered. Her right arm lost circulation and went numb, so she sought guidance from her blood cancer specialist. He dismissed her worries, saying that her tumors were pinching a nerve. She’d get better, he predicted, after more chemo.

“I knew in my body that something was wrong,” Ms. Ngon recalled. When the oncologist continued to downplay her concerns, she and a fellow communications specialist sat down together in the hospital lobby to draft an email to her physician. “We were trying to articulate the urgency in an email that expresses that I’m not being dramatic. We had to do it in a way that didn’t insult his intelligence: ‘Respectfully, you’re the doctor, but I know something is wrong.’ ”
In essence, Ms. Ngon was trying to be diplomatic and not trigger her oncologist’s defenses, while still convincing him to take action. Her approach to getting her doctor’s attention worked. He referred Ms. Ngon to a radiologist, who discovered that she had blood clots in her arm. Ms. Ngon then landed in the ICU for a week, as clinicians tried to break up the clots.
“I was the perfect person for this to happen to, because of my job and education. But it makes me sad because I understand I was in a fortunate position, with a background in communication. Most people don’t have that,” Ms. Ngon said.
This and other negative experiences during her medical saga inspired Ms. Ngon to partner with the Lymphoma Research Foundation in order to spread the word about unique challenges facing patients like her: people of color.
Ms. Ngon, who is Black, said her goal as a patient advocate is to “empower communities of color to speak up for themselves and hold oncologists responsible for listening and understanding differences across cultures.” And she wants to take a stand against the “gaslighting” of patients.
African Americans with hematologic disease like Ms. Ngon face a higher risk of poor outcomes than Whites, even as they are less likely than Whites to develop certain blood cancers. The reasons for this disparity aren’t clear, but researchers suspect they’re related to factors such as poverty, lack of insurance, genetics, and limited access to high-quality care.
Some researchers have blamed another factor: racism. A 2022 study sought to explain why Black and Hispanic patients with acute myeloid leukemia in urban areas have higher mortality rates than Whites, “despite more favorable genetics and younger age” (hazard ratio, 1.59, 95% confidence interval, 1.15-2.22 and HR, 1.25; 95% CI, 0.88-1.79). The study authors determined that “structural racism” – which they measured by examining segregation and “disadvantage” in neighborhoods where patients lived – accounted for nearly all of the disparities.
Ms. Ngon said her experiences and her awareness about poorer outcomes in medicine for African Americans – such as higher death rates for Black women during pregnancy – affect how she interacts with clinicians. “I automatically assume a barrier between me and my doctors, and it’s their responsibility to dismantle it.”
Making an connection with a physician can make a huge difference, she said. “I walked into my primary care doctor’s office and saw that she was a Latino woman. My guard went down, and I could feel her care for me as a human being. Whether that was because she was also a woman of color or not, I don’t know. But I did feel more cared for.”
However, Ms. Ngon could not find a Black oncologist to care for her in New York City, and that’s no surprise.
Ethnic and gender diversity remains an immense challenge in the hematology/oncology field. According to the American Society of Clinical Oncology, only about a third of oncologists are women, and the percentages identifying themselves as Black/African American and Hispanic are just 2.3% and 5.8%, respectively.
These numbers don’t seem likely to budge much any time soon. An analysis of medical students in U.S. oncology training programs from 2015-2020 found that just 3.8% identified themselves as Black/African American and 5.1% as Hispanic/Latino versus 52.15% as White and 31% as Asian/Pacific Islander/Native Hawaiian.
Ms. Ngon encountered challenges on other fronts during her cancer care. When she needed a wig during chemotherapy, a list of insurer-approved shops didn’t include any that catered to African Americans. Essentially, she said, she was being told that she couldn’t “purchase a wig from a place that makes you feel comfortable and from a woman who understand your needs as a Black woman. It needs to be from these specific shops that really don’t cater to my community.”
She also found it difficult to find fellow patients who shared her unique challenges. “I remember when I was diagnosed, I was looking through the support groups on Facebook, trying to find someone Black to ask about whether braiding my hair might stop it from falling out.”
Now, Ms. Ngon is in remission. And she’s happy with her oncologist, who’s White. “He listened to me, and he promised me that I would have the most boring recovery process ever, after everything I’d experienced. That explains a lot of why I felt so comfortable with him.”
She hopes to use her partnership with the Lymphoma Research Foundation to be a resource for people of color and alert them to the support that’s available for them. “I would love to let them know how to advocate for themselves as patients, how to trust their bodies, how to push back if they feel like they’re not getting the care that they deserve.”
Ms. Ngon would also like to see more support for medical students of color. “I hope to exist in a world one day where it wouldn’t be so hard to find an oncologist who looks like me in a city as large as this one,” she said.
As for oncologists, she urged them to “go the extra mile and really, really listen to what patients are saying. It’s easier said than done because there are natural biases in this world, and it’s hard to overcome those obstacles. But to not be heard and have to push every time. It was just exhausting to do that on top of trying to beat cancer.”
Diagnosed with Hodgkin lymphoma in 2021, Ms. Ngon underwent port surgery to allow chemotherapy to be administered. Her right arm lost circulation and went numb, so she sought guidance from her blood cancer specialist. He dismissed her worries, saying that her tumors were pinching a nerve. She’d get better, he predicted, after more chemo.

“I knew in my body that something was wrong,” Ms. Ngon recalled. When the oncologist continued to downplay her concerns, she and a fellow communications specialist sat down together in the hospital lobby to draft an email to her physician. “We were trying to articulate the urgency in an email that expresses that I’m not being dramatic. We had to do it in a way that didn’t insult his intelligence: ‘Respectfully, you’re the doctor, but I know something is wrong.’ ”
In essence, Ms. Ngon was trying to be diplomatic and not trigger her oncologist’s defenses, while still convincing him to take action. Her approach to getting her doctor’s attention worked. He referred Ms. Ngon to a radiologist, who discovered that she had blood clots in her arm. Ms. Ngon then landed in the ICU for a week, as clinicians tried to break up the clots.
“I was the perfect person for this to happen to, because of my job and education. But it makes me sad because I understand I was in a fortunate position, with a background in communication. Most people don’t have that,” Ms. Ngon said.
This and other negative experiences during her medical saga inspired Ms. Ngon to partner with the Lymphoma Research Foundation in order to spread the word about unique challenges facing patients like her: people of color.
Ms. Ngon, who is Black, said her goal as a patient advocate is to “empower communities of color to speak up for themselves and hold oncologists responsible for listening and understanding differences across cultures.” And she wants to take a stand against the “gaslighting” of patients.
African Americans with hematologic disease like Ms. Ngon face a higher risk of poor outcomes than Whites, even as they are less likely than Whites to develop certain blood cancers. The reasons for this disparity aren’t clear, but researchers suspect they’re related to factors such as poverty, lack of insurance, genetics, and limited access to high-quality care.
Some researchers have blamed another factor: racism. A 2022 study sought to explain why Black and Hispanic patients with acute myeloid leukemia in urban areas have higher mortality rates than Whites, “despite more favorable genetics and younger age” (hazard ratio, 1.59, 95% confidence interval, 1.15-2.22 and HR, 1.25; 95% CI, 0.88-1.79). The study authors determined that “structural racism” – which they measured by examining segregation and “disadvantage” in neighborhoods where patients lived – accounted for nearly all of the disparities.
Ms. Ngon said her experiences and her awareness about poorer outcomes in medicine for African Americans – such as higher death rates for Black women during pregnancy – affect how she interacts with clinicians. “I automatically assume a barrier between me and my doctors, and it’s their responsibility to dismantle it.”
Making an connection with a physician can make a huge difference, she said. “I walked into my primary care doctor’s office and saw that she was a Latino woman. My guard went down, and I could feel her care for me as a human being. Whether that was because she was also a woman of color or not, I don’t know. But I did feel more cared for.”
However, Ms. Ngon could not find a Black oncologist to care for her in New York City, and that’s no surprise.
Ethnic and gender diversity remains an immense challenge in the hematology/oncology field. According to the American Society of Clinical Oncology, only about a third of oncologists are women, and the percentages identifying themselves as Black/African American and Hispanic are just 2.3% and 5.8%, respectively.
These numbers don’t seem likely to budge much any time soon. An analysis of medical students in U.S. oncology training programs from 2015-2020 found that just 3.8% identified themselves as Black/African American and 5.1% as Hispanic/Latino versus 52.15% as White and 31% as Asian/Pacific Islander/Native Hawaiian.
Ms. Ngon encountered challenges on other fronts during her cancer care. When she needed a wig during chemotherapy, a list of insurer-approved shops didn’t include any that catered to African Americans. Essentially, she said, she was being told that she couldn’t “purchase a wig from a place that makes you feel comfortable and from a woman who understand your needs as a Black woman. It needs to be from these specific shops that really don’t cater to my community.”
She also found it difficult to find fellow patients who shared her unique challenges. “I remember when I was diagnosed, I was looking through the support groups on Facebook, trying to find someone Black to ask about whether braiding my hair might stop it from falling out.”
Now, Ms. Ngon is in remission. And she’s happy with her oncologist, who’s White. “He listened to me, and he promised me that I would have the most boring recovery process ever, after everything I’d experienced. That explains a lot of why I felt so comfortable with him.”
She hopes to use her partnership with the Lymphoma Research Foundation to be a resource for people of color and alert them to the support that’s available for them. “I would love to let them know how to advocate for themselves as patients, how to trust their bodies, how to push back if they feel like they’re not getting the care that they deserve.”
Ms. Ngon would also like to see more support for medical students of color. “I hope to exist in a world one day where it wouldn’t be so hard to find an oncologist who looks like me in a city as large as this one,” she said.
As for oncologists, she urged them to “go the extra mile and really, really listen to what patients are saying. It’s easier said than done because there are natural biases in this world, and it’s hard to overcome those obstacles. But to not be heard and have to push every time. It was just exhausting to do that on top of trying to beat cancer.”









