User login
For MD-IQ use only
PV: Novel rusfertide shows ‘impressive’ efficacy
“The results are surprisingly positive,” said senior author Ronald Hoffman, MD, of the Icahn School of Medicine at Mount Sinai, New York, in discussing the late-breaking research at a press briefing during the European Hematology Association Hybrid Congress 2023.
“Importantly, the study met all of its efficacy endpoints, including the proportion of responders, absence of phlebotomy eligibility, and hematocrit control,” Dr. Hoffman said.
PV, a relatively common clonal myeloproliferative neoplasm, is characterized by uncontrolled erythrocytosis, or excessive production of red blood cells, increasing the risk for serious complications such as thromboembolic and cardiovascular events – the most common causes of morbidity and mortality in this blood cancer.
To treat PV, the maintenance of hematocrit levels at below 45% is critical. However, the current standard of care, therapeutic phlebotomy, with or without cytoreductive agents, falls short in maintaining those lower levels in the majority of patients, Dr. Hoffman explained.
To improve responses, rusfertide was developed as a novel, synthetic form of hepcidin, a peptide hormone that is produced by the liver and functions to maintain iron homeostasis and control the formation of red blood cells.
“This is somewhat of a paradigm shift,” said Dr. Hoffman in the press briefing. “We’re trying to use a hormone made by the liver to control excessive red blood cell production from polycythemia vera.”
For the phase 2 REVIVE study evaluating rusfertide in PV, the authors enrolled 53 patients with PV who had a high phlebotomy burden while receiving the current standard of care. The study’s criteria called for patients to have received at least three therapeutic phlebotomies in the 28 weeks prior to enrollment, with or without concurrent cytoreductive agents.
During a first part of the study, patients received subcutaneous rusfertide once weekly over 28 weeks, during which period the dose was adjusted individually to achieve control of HCT levels below 45%.
The second part was a withdrawal phase extending from weeks 29 to 41, in which patients were randomized in a blinded fashion to either continue on rusfertide (n = 26) or receive a placebo (n = 27).
The patients had a median age of 58; they were 71.7% male, and 54.7% had previously been treated with therapeutic phlebotomy alone while 45.3% received therapeutic phlebotomy plus cytoreductive agents.
Patients were considered to be responders if they met three criteria, including having HCT control without phlebotomy eligibility, no therapeutic phlebotomy, and having completed 12 weeks of treatment.
At the end of the second phase, 69.2% of patients receiving rusfertide were responders versus just 18.5% in the placebo group (P = .0003).
Notably, the improvement with rusfertide was observed among those receiving therapeutic phlebotomy alone, as well as with cytoreductive agents (both P = .02).
Compared with placebo, rusfertide provided significant improvement in measures including the maintenance of response, the absence of the need for therapeutic phlebotomy, and persistent HCT control (P < .0001 for all).
Whereas the phlebotomy-free rate with rusfertide during the dose-finding weeks of 1-17 was 76.9% and in weeks 17-29, 87.3%, the rate increased in part 2 of the study to 92.3%.
Additional symptom benefits reported with rusfertide at week 29 versus baseline in part 1 of the study included significant improvements in concentration (P = .0018), itching (P = .0054), fatigue (P = .0074), and inactivity (P = .0005).
In terms of safety, rusfertide was generally well tolerated, with 83% of treatment-emergent adverse events (TEAEs) being grade 1-2, while 17% were grade 3, and none were grade 4 or 5.
The most common TEAEs consisted of injection-site reactions, which were localized, and grade 1-2 in severity. The incidence of reactions decreased with ongoing treatment. There were only two discontinuations resulting from TEAEs.
Among a total of 70 patients who were enrolled, 52 (74.3%) have continued to receive rusfertide for at least 1 year, 32 (45.7%) for at least 1.5 years, and 10 (14.3%) for at least 2 years, indicating the long-term tolerability of rusfertide.
Further commenting, first author Marina Kremyanskaya, MD, PhD, an assistant professor of medicine, hematology, and medical oncology at the Icahn School of Medicine at Mount Sinai, added that a key benefit is rusfertide’s tolerability with combination therapies, which is important in enabling the avoidance of phlebotomies.
“Many patients on cytoreductive therapies still require phlebotomies, and they can’t tolerate a dose increase, either due to cytopenias or other adverse reactions,” she said in an interview. “So adding rusfertide allows for better control of their hematocrits on a lower dose of their respective cytoreductive drug.”
“The combination treatment thus allows for elimination of phlebotomy requirements and potentially improves their symptoms,” Dr. Kremyanskaya said, adding that “using a lower dose of cytoreductive drug such as interferon or hydroxyurea could offer a symptomatic relief to patients as well.”
Overall, she agreed that the responses are remarkably positive.
“I think this is what is so impressive about this agent – basically everybody responds,” Dr. Kremyanskaya said. “When we first started treating patients, we were so impressed, as none of the other drugs we use to treat PV, or any other hematologic malignancy, come anywhere close to this response rate.”
In commenting on the study, Claire Harrison, MD, a professor of myeloproliferative neoplasms and deputy medical director of research at Guy’s and St Thomas’ NHS Foundation Trust in London, agreed that “these data show a strong signal for effectiveness of this therapy in controlling red cell proliferation in PV without inducing iron deficiency and adding to the symptom burden of patients.”
The alternative of phlebotomy “is painful and consumes patient time and hospital resources,” she said in an interview.
Dr. Harrison noted that an earlier signal suggested squamous cell cancer might be of potential concern, but the signal “has not re-emerged [suggesting] this does indeed seem to be a safe and extremely effective therapy.”
Further commenting on the study during the press briefing, Konstanze Döhner, MD, of the University of Ulm (Germany) added that “this is exciting data.”
“For a long time, we had no therapeutic options for PV, and now the field is rapidly developing,” she said.
In ongoing research, rusfertide is currently being studied in the phase 3, placebo-controlled VERIFY randomized trial.
The study was sponsored by Protagonist Therapeutics. Dr. Hoffman reports being on the advisory board for Protagonist Therapeutics, and Dr. Kremyanskaya is a consultant for Protagonist Therapeutics. Dr. Harrison had no disclosures to report.
“The results are surprisingly positive,” said senior author Ronald Hoffman, MD, of the Icahn School of Medicine at Mount Sinai, New York, in discussing the late-breaking research at a press briefing during the European Hematology Association Hybrid Congress 2023.
“Importantly, the study met all of its efficacy endpoints, including the proportion of responders, absence of phlebotomy eligibility, and hematocrit control,” Dr. Hoffman said.
PV, a relatively common clonal myeloproliferative neoplasm, is characterized by uncontrolled erythrocytosis, or excessive production of red blood cells, increasing the risk for serious complications such as thromboembolic and cardiovascular events – the most common causes of morbidity and mortality in this blood cancer.
To treat PV, the maintenance of hematocrit levels at below 45% is critical. However, the current standard of care, therapeutic phlebotomy, with or without cytoreductive agents, falls short in maintaining those lower levels in the majority of patients, Dr. Hoffman explained.
To improve responses, rusfertide was developed as a novel, synthetic form of hepcidin, a peptide hormone that is produced by the liver and functions to maintain iron homeostasis and control the formation of red blood cells.
“This is somewhat of a paradigm shift,” said Dr. Hoffman in the press briefing. “We’re trying to use a hormone made by the liver to control excessive red blood cell production from polycythemia vera.”
For the phase 2 REVIVE study evaluating rusfertide in PV, the authors enrolled 53 patients with PV who had a high phlebotomy burden while receiving the current standard of care. The study’s criteria called for patients to have received at least three therapeutic phlebotomies in the 28 weeks prior to enrollment, with or without concurrent cytoreductive agents.
During a first part of the study, patients received subcutaneous rusfertide once weekly over 28 weeks, during which period the dose was adjusted individually to achieve control of HCT levels below 45%.
The second part was a withdrawal phase extending from weeks 29 to 41, in which patients were randomized in a blinded fashion to either continue on rusfertide (n = 26) or receive a placebo (n = 27).
The patients had a median age of 58; they were 71.7% male, and 54.7% had previously been treated with therapeutic phlebotomy alone while 45.3% received therapeutic phlebotomy plus cytoreductive agents.
Patients were considered to be responders if they met three criteria, including having HCT control without phlebotomy eligibility, no therapeutic phlebotomy, and having completed 12 weeks of treatment.
At the end of the second phase, 69.2% of patients receiving rusfertide were responders versus just 18.5% in the placebo group (P = .0003).
Notably, the improvement with rusfertide was observed among those receiving therapeutic phlebotomy alone, as well as with cytoreductive agents (both P = .02).
Compared with placebo, rusfertide provided significant improvement in measures including the maintenance of response, the absence of the need for therapeutic phlebotomy, and persistent HCT control (P < .0001 for all).
Whereas the phlebotomy-free rate with rusfertide during the dose-finding weeks of 1-17 was 76.9% and in weeks 17-29, 87.3%, the rate increased in part 2 of the study to 92.3%.
Additional symptom benefits reported with rusfertide at week 29 versus baseline in part 1 of the study included significant improvements in concentration (P = .0018), itching (P = .0054), fatigue (P = .0074), and inactivity (P = .0005).
In terms of safety, rusfertide was generally well tolerated, with 83% of treatment-emergent adverse events (TEAEs) being grade 1-2, while 17% were grade 3, and none were grade 4 or 5.
The most common TEAEs consisted of injection-site reactions, which were localized, and grade 1-2 in severity. The incidence of reactions decreased with ongoing treatment. There were only two discontinuations resulting from TEAEs.
Among a total of 70 patients who were enrolled, 52 (74.3%) have continued to receive rusfertide for at least 1 year, 32 (45.7%) for at least 1.5 years, and 10 (14.3%) for at least 2 years, indicating the long-term tolerability of rusfertide.
Further commenting, first author Marina Kremyanskaya, MD, PhD, an assistant professor of medicine, hematology, and medical oncology at the Icahn School of Medicine at Mount Sinai, added that a key benefit is rusfertide’s tolerability with combination therapies, which is important in enabling the avoidance of phlebotomies.
“Many patients on cytoreductive therapies still require phlebotomies, and they can’t tolerate a dose increase, either due to cytopenias or other adverse reactions,” she said in an interview. “So adding rusfertide allows for better control of their hematocrits on a lower dose of their respective cytoreductive drug.”
“The combination treatment thus allows for elimination of phlebotomy requirements and potentially improves their symptoms,” Dr. Kremyanskaya said, adding that “using a lower dose of cytoreductive drug such as interferon or hydroxyurea could offer a symptomatic relief to patients as well.”
Overall, she agreed that the responses are remarkably positive.
“I think this is what is so impressive about this agent – basically everybody responds,” Dr. Kremyanskaya said. “When we first started treating patients, we were so impressed, as none of the other drugs we use to treat PV, or any other hematologic malignancy, come anywhere close to this response rate.”
In commenting on the study, Claire Harrison, MD, a professor of myeloproliferative neoplasms and deputy medical director of research at Guy’s and St Thomas’ NHS Foundation Trust in London, agreed that “these data show a strong signal for effectiveness of this therapy in controlling red cell proliferation in PV without inducing iron deficiency and adding to the symptom burden of patients.”
The alternative of phlebotomy “is painful and consumes patient time and hospital resources,” she said in an interview.
Dr. Harrison noted that an earlier signal suggested squamous cell cancer might be of potential concern, but the signal “has not re-emerged [suggesting] this does indeed seem to be a safe and extremely effective therapy.”
Further commenting on the study during the press briefing, Konstanze Döhner, MD, of the University of Ulm (Germany) added that “this is exciting data.”
“For a long time, we had no therapeutic options for PV, and now the field is rapidly developing,” she said.
In ongoing research, rusfertide is currently being studied in the phase 3, placebo-controlled VERIFY randomized trial.
The study was sponsored by Protagonist Therapeutics. Dr. Hoffman reports being on the advisory board for Protagonist Therapeutics, and Dr. Kremyanskaya is a consultant for Protagonist Therapeutics. Dr. Harrison had no disclosures to report.
“The results are surprisingly positive,” said senior author Ronald Hoffman, MD, of the Icahn School of Medicine at Mount Sinai, New York, in discussing the late-breaking research at a press briefing during the European Hematology Association Hybrid Congress 2023.
“Importantly, the study met all of its efficacy endpoints, including the proportion of responders, absence of phlebotomy eligibility, and hematocrit control,” Dr. Hoffman said.
PV, a relatively common clonal myeloproliferative neoplasm, is characterized by uncontrolled erythrocytosis, or excessive production of red blood cells, increasing the risk for serious complications such as thromboembolic and cardiovascular events – the most common causes of morbidity and mortality in this blood cancer.
To treat PV, the maintenance of hematocrit levels at below 45% is critical. However, the current standard of care, therapeutic phlebotomy, with or without cytoreductive agents, falls short in maintaining those lower levels in the majority of patients, Dr. Hoffman explained.
To improve responses, rusfertide was developed as a novel, synthetic form of hepcidin, a peptide hormone that is produced by the liver and functions to maintain iron homeostasis and control the formation of red blood cells.
“This is somewhat of a paradigm shift,” said Dr. Hoffman in the press briefing. “We’re trying to use a hormone made by the liver to control excessive red blood cell production from polycythemia vera.”
For the phase 2 REVIVE study evaluating rusfertide in PV, the authors enrolled 53 patients with PV who had a high phlebotomy burden while receiving the current standard of care. The study’s criteria called for patients to have received at least three therapeutic phlebotomies in the 28 weeks prior to enrollment, with or without concurrent cytoreductive agents.
During a first part of the study, patients received subcutaneous rusfertide once weekly over 28 weeks, during which period the dose was adjusted individually to achieve control of HCT levels below 45%.
The second part was a withdrawal phase extending from weeks 29 to 41, in which patients were randomized in a blinded fashion to either continue on rusfertide (n = 26) or receive a placebo (n = 27).
The patients had a median age of 58; they were 71.7% male, and 54.7% had previously been treated with therapeutic phlebotomy alone while 45.3% received therapeutic phlebotomy plus cytoreductive agents.
Patients were considered to be responders if they met three criteria, including having HCT control without phlebotomy eligibility, no therapeutic phlebotomy, and having completed 12 weeks of treatment.
At the end of the second phase, 69.2% of patients receiving rusfertide were responders versus just 18.5% in the placebo group (P = .0003).
Notably, the improvement with rusfertide was observed among those receiving therapeutic phlebotomy alone, as well as with cytoreductive agents (both P = .02).
Compared with placebo, rusfertide provided significant improvement in measures including the maintenance of response, the absence of the need for therapeutic phlebotomy, and persistent HCT control (P < .0001 for all).
Whereas the phlebotomy-free rate with rusfertide during the dose-finding weeks of 1-17 was 76.9% and in weeks 17-29, 87.3%, the rate increased in part 2 of the study to 92.3%.
Additional symptom benefits reported with rusfertide at week 29 versus baseline in part 1 of the study included significant improvements in concentration (P = .0018), itching (P = .0054), fatigue (P = .0074), and inactivity (P = .0005).
In terms of safety, rusfertide was generally well tolerated, with 83% of treatment-emergent adverse events (TEAEs) being grade 1-2, while 17% were grade 3, and none were grade 4 or 5.
The most common TEAEs consisted of injection-site reactions, which were localized, and grade 1-2 in severity. The incidence of reactions decreased with ongoing treatment. There were only two discontinuations resulting from TEAEs.
Among a total of 70 patients who were enrolled, 52 (74.3%) have continued to receive rusfertide for at least 1 year, 32 (45.7%) for at least 1.5 years, and 10 (14.3%) for at least 2 years, indicating the long-term tolerability of rusfertide.
Further commenting, first author Marina Kremyanskaya, MD, PhD, an assistant professor of medicine, hematology, and medical oncology at the Icahn School of Medicine at Mount Sinai, added that a key benefit is rusfertide’s tolerability with combination therapies, which is important in enabling the avoidance of phlebotomies.
“Many patients on cytoreductive therapies still require phlebotomies, and they can’t tolerate a dose increase, either due to cytopenias or other adverse reactions,” she said in an interview. “So adding rusfertide allows for better control of their hematocrits on a lower dose of their respective cytoreductive drug.”
“The combination treatment thus allows for elimination of phlebotomy requirements and potentially improves their symptoms,” Dr. Kremyanskaya said, adding that “using a lower dose of cytoreductive drug such as interferon or hydroxyurea could offer a symptomatic relief to patients as well.”
Overall, she agreed that the responses are remarkably positive.
“I think this is what is so impressive about this agent – basically everybody responds,” Dr. Kremyanskaya said. “When we first started treating patients, we were so impressed, as none of the other drugs we use to treat PV, or any other hematologic malignancy, come anywhere close to this response rate.”
In commenting on the study, Claire Harrison, MD, a professor of myeloproliferative neoplasms and deputy medical director of research at Guy’s and St Thomas’ NHS Foundation Trust in London, agreed that “these data show a strong signal for effectiveness of this therapy in controlling red cell proliferation in PV without inducing iron deficiency and adding to the symptom burden of patients.”
The alternative of phlebotomy “is painful and consumes patient time and hospital resources,” she said in an interview.
Dr. Harrison noted that an earlier signal suggested squamous cell cancer might be of potential concern, but the signal “has not re-emerged [suggesting] this does indeed seem to be a safe and extremely effective therapy.”
Further commenting on the study during the press briefing, Konstanze Döhner, MD, of the University of Ulm (Germany) added that “this is exciting data.”
“For a long time, we had no therapeutic options for PV, and now the field is rapidly developing,” she said.
In ongoing research, rusfertide is currently being studied in the phase 3, placebo-controlled VERIFY randomized trial.
The study was sponsored by Protagonist Therapeutics. Dr. Hoffman reports being on the advisory board for Protagonist Therapeutics, and Dr. Kremyanskaya is a consultant for Protagonist Therapeutics. Dr. Harrison had no disclosures to report.
FROM EHA 2023
FDA warns of tattoo ink tied to dangerous infections
The Food and Drug Administration draft guidance released recently on possible contamination of tattoo ink was not concerning Whitney Donohue, 34, owner of Forget Me Not Tattoo in Billings, Mont.
“I get our ink directly through the manufacturer – not at a store or through Amazon or eBay,” she said. “You never know if it’s going to be repackaged.”
Tattoo artists themselves, she said, regulate the quality of ink they use.
Still, the threat is real, said Bruce Brod, MD, a clinical professor of dermatology at the University of Pennsylvania Health System. “I’ve seen several different infections from tattooing, and they are from organisms that tend to contaminate things in damp, liquid-type environments.”
, dermatologists said.
“Tattooing involves puncturing the epidermis about 100 times per second with needles and depositing ink 1.5 to 2 millimeters below the surface of the skin, deep into the dermis,” the guidance states. “Contaminated tattoo ink can cause infections and serious injuries. Because these inks are injected, pathogens or other harmful substances in these inks can travel from the injection site through the blood and lymphatic systems to other parts of the body.”
The guidance comes as body art continues to get more popular. According to a 2019 poll, 30% of Americans had at least one tattoo – up from 21% in 2012. Forty percent of people 18-34 and 36% of those ages 35-54 had at least one tattoo. And though they are commonplace, tattoos come with medical risks that should be known beforehand, doctors said.
Commonly reported symptoms of tattoo ink–associated infections include rashes, blisters, painful nodules, and severe abscesses. One of the most common bacteria found in contaminated tattoo ink is nontuberculous mycobacteria, which is related to the bacteria that causes tuberculosis and can be found in soil and water.
The guidance lists several unsanitary manufacturing conditions that may lead to ink contamination, including:
- Preparing or packing of tattoo inks in facilities that are hard to sanitize, such as carpeted areas
- Ink or ink components left uncovered, especially near open air ducts
- Unsanitary mixing of tattoo inks, including with unclean utensils or containers
- Lack of appropriate attire by staff, failure to use hairnets, lab coats, aprons, gowns, masks, or gloves
“Infections will often spread along the drainage channels in the skin and create squiggly, uneven lines of big red, lumpy nodules,” Dr. Brod said.
Between 2003 and 2023, there were 18 recalls of tattoo inks that were contaminated with various microorganisms, according to the FDA. In May 2019, the FDA issued a safety alert advising consumers, tattoo artists, and retailers to avoid using or selling certain tattoo inks contaminated with microorganisms.
Reputable ink manufacturers use a process called gamma radiation, which refers to electromagnetic radiation of high frequencies to kill microorganisms in the ink and its packaging.
Most of the trustworthy, high-quality ink manufacturers are well-known among tattoo artists, Ms. Donohue said.
While she has seen customers with sensitive skin have allergic reactions, she has not seen someone come back with an infection in her 9 years working in the tattoo industry.
Because tattoo ink is considered a cosmetic product, there is not much regulatory oversight involved, which means the sterility and quality of ingredients vary, said Teo Soleymani, MD, an assistant clinical professor of dermatology and dermatological surgery at the UCLA David Geffen School of Medicine.
“Cosmeceuticals aren’t regulated by the FDA like prescription medication,” he said. “What we’ve seen many times is inadvertent contamination during the application process or contamination while the inks are being made.”
In years past, unclean needles spreading hepatitis and HIV were more of a concern, but those rates have dropped significantly, Dr. Soleymani said.
The infections that have increased are from rare bacteria that exist in stagnant water. And they are injected into a part of the body that allows them to evade the immune system, he said: shallow enough that there aren’t many associated blood vessels, but not still below the layer of skin that gets sloughed off every 28 days.
Sometimes, antibiotics alone won’t cut it, and the tattoo will require surgical removal.
“The aesthetic you were going for has to be not only removed, but you’re left with a surgical scar,” Dr. Soleymani said. “Tattoos can be beautiful, but they can come with unwanted visitors that can cause months of misery.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration draft guidance released recently on possible contamination of tattoo ink was not concerning Whitney Donohue, 34, owner of Forget Me Not Tattoo in Billings, Mont.
“I get our ink directly through the manufacturer – not at a store or through Amazon or eBay,” she said. “You never know if it’s going to be repackaged.”
Tattoo artists themselves, she said, regulate the quality of ink they use.
Still, the threat is real, said Bruce Brod, MD, a clinical professor of dermatology at the University of Pennsylvania Health System. “I’ve seen several different infections from tattooing, and they are from organisms that tend to contaminate things in damp, liquid-type environments.”
, dermatologists said.
“Tattooing involves puncturing the epidermis about 100 times per second with needles and depositing ink 1.5 to 2 millimeters below the surface of the skin, deep into the dermis,” the guidance states. “Contaminated tattoo ink can cause infections and serious injuries. Because these inks are injected, pathogens or other harmful substances in these inks can travel from the injection site through the blood and lymphatic systems to other parts of the body.”
The guidance comes as body art continues to get more popular. According to a 2019 poll, 30% of Americans had at least one tattoo – up from 21% in 2012. Forty percent of people 18-34 and 36% of those ages 35-54 had at least one tattoo. And though they are commonplace, tattoos come with medical risks that should be known beforehand, doctors said.
Commonly reported symptoms of tattoo ink–associated infections include rashes, blisters, painful nodules, and severe abscesses. One of the most common bacteria found in contaminated tattoo ink is nontuberculous mycobacteria, which is related to the bacteria that causes tuberculosis and can be found in soil and water.
The guidance lists several unsanitary manufacturing conditions that may lead to ink contamination, including:
- Preparing or packing of tattoo inks in facilities that are hard to sanitize, such as carpeted areas
- Ink or ink components left uncovered, especially near open air ducts
- Unsanitary mixing of tattoo inks, including with unclean utensils or containers
- Lack of appropriate attire by staff, failure to use hairnets, lab coats, aprons, gowns, masks, or gloves
“Infections will often spread along the drainage channels in the skin and create squiggly, uneven lines of big red, lumpy nodules,” Dr. Brod said.
Between 2003 and 2023, there were 18 recalls of tattoo inks that were contaminated with various microorganisms, according to the FDA. In May 2019, the FDA issued a safety alert advising consumers, tattoo artists, and retailers to avoid using or selling certain tattoo inks contaminated with microorganisms.
Reputable ink manufacturers use a process called gamma radiation, which refers to electromagnetic radiation of high frequencies to kill microorganisms in the ink and its packaging.
Most of the trustworthy, high-quality ink manufacturers are well-known among tattoo artists, Ms. Donohue said.
While she has seen customers with sensitive skin have allergic reactions, she has not seen someone come back with an infection in her 9 years working in the tattoo industry.
Because tattoo ink is considered a cosmetic product, there is not much regulatory oversight involved, which means the sterility and quality of ingredients vary, said Teo Soleymani, MD, an assistant clinical professor of dermatology and dermatological surgery at the UCLA David Geffen School of Medicine.
“Cosmeceuticals aren’t regulated by the FDA like prescription medication,” he said. “What we’ve seen many times is inadvertent contamination during the application process or contamination while the inks are being made.”
In years past, unclean needles spreading hepatitis and HIV were more of a concern, but those rates have dropped significantly, Dr. Soleymani said.
The infections that have increased are from rare bacteria that exist in stagnant water. And they are injected into a part of the body that allows them to evade the immune system, he said: shallow enough that there aren’t many associated blood vessels, but not still below the layer of skin that gets sloughed off every 28 days.
Sometimes, antibiotics alone won’t cut it, and the tattoo will require surgical removal.
“The aesthetic you were going for has to be not only removed, but you’re left with a surgical scar,” Dr. Soleymani said. “Tattoos can be beautiful, but they can come with unwanted visitors that can cause months of misery.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration draft guidance released recently on possible contamination of tattoo ink was not concerning Whitney Donohue, 34, owner of Forget Me Not Tattoo in Billings, Mont.
“I get our ink directly through the manufacturer – not at a store or through Amazon or eBay,” she said. “You never know if it’s going to be repackaged.”
Tattoo artists themselves, she said, regulate the quality of ink they use.
Still, the threat is real, said Bruce Brod, MD, a clinical professor of dermatology at the University of Pennsylvania Health System. “I’ve seen several different infections from tattooing, and they are from organisms that tend to contaminate things in damp, liquid-type environments.”
, dermatologists said.
“Tattooing involves puncturing the epidermis about 100 times per second with needles and depositing ink 1.5 to 2 millimeters below the surface of the skin, deep into the dermis,” the guidance states. “Contaminated tattoo ink can cause infections and serious injuries. Because these inks are injected, pathogens or other harmful substances in these inks can travel from the injection site through the blood and lymphatic systems to other parts of the body.”
The guidance comes as body art continues to get more popular. According to a 2019 poll, 30% of Americans had at least one tattoo – up from 21% in 2012. Forty percent of people 18-34 and 36% of those ages 35-54 had at least one tattoo. And though they are commonplace, tattoos come with medical risks that should be known beforehand, doctors said.
Commonly reported symptoms of tattoo ink–associated infections include rashes, blisters, painful nodules, and severe abscesses. One of the most common bacteria found in contaminated tattoo ink is nontuberculous mycobacteria, which is related to the bacteria that causes tuberculosis and can be found in soil and water.
The guidance lists several unsanitary manufacturing conditions that may lead to ink contamination, including:
- Preparing or packing of tattoo inks in facilities that are hard to sanitize, such as carpeted areas
- Ink or ink components left uncovered, especially near open air ducts
- Unsanitary mixing of tattoo inks, including with unclean utensils or containers
- Lack of appropriate attire by staff, failure to use hairnets, lab coats, aprons, gowns, masks, or gloves
“Infections will often spread along the drainage channels in the skin and create squiggly, uneven lines of big red, lumpy nodules,” Dr. Brod said.
Between 2003 and 2023, there were 18 recalls of tattoo inks that were contaminated with various microorganisms, according to the FDA. In May 2019, the FDA issued a safety alert advising consumers, tattoo artists, and retailers to avoid using or selling certain tattoo inks contaminated with microorganisms.
Reputable ink manufacturers use a process called gamma radiation, which refers to electromagnetic radiation of high frequencies to kill microorganisms in the ink and its packaging.
Most of the trustworthy, high-quality ink manufacturers are well-known among tattoo artists, Ms. Donohue said.
While she has seen customers with sensitive skin have allergic reactions, she has not seen someone come back with an infection in her 9 years working in the tattoo industry.
Because tattoo ink is considered a cosmetic product, there is not much regulatory oversight involved, which means the sterility and quality of ingredients vary, said Teo Soleymani, MD, an assistant clinical professor of dermatology and dermatological surgery at the UCLA David Geffen School of Medicine.
“Cosmeceuticals aren’t regulated by the FDA like prescription medication,” he said. “What we’ve seen many times is inadvertent contamination during the application process or contamination while the inks are being made.”
In years past, unclean needles spreading hepatitis and HIV were more of a concern, but those rates have dropped significantly, Dr. Soleymani said.
The infections that have increased are from rare bacteria that exist in stagnant water. And they are injected into a part of the body that allows them to evade the immune system, he said: shallow enough that there aren’t many associated blood vessels, but not still below the layer of skin that gets sloughed off every 28 days.
Sometimes, antibiotics alone won’t cut it, and the tattoo will require surgical removal.
“The aesthetic you were going for has to be not only removed, but you’re left with a surgical scar,” Dr. Soleymani said. “Tattoos can be beautiful, but they can come with unwanted visitors that can cause months of misery.”
A version of this article first appeared on WebMD.com.
PCOS associated with shorter lifespan
CHICAGO –
In the study, involving nearly 10,000 women with PCOS and matched controls from Finland, women with PCOS died on average a year earlier than their age-matched counterparts, primarily from diseases of the circulatory system, cancer, and diabetes.
PCOS is the most common endocrine disorder of reproductive-age women, of whom about 50%-70% also have obesity.
“I think we need to acknowledge that this is a health burden and not just a reproductive problem. In many cases we deal with the reproductive problem, and then these women are left alone. … So I think the message is we need to look beyond the reproductive outcomes, which are … really good. We can manage that,” said Terhi T. Piltonen, MD, PhD, during a press briefing held June 15 at the annual meeting of the Endocrine Society.
“I think the difficult part is [managing] the lifelong health for these women and supporting them to achieve the best health they can get. We need a multidisciplinary effort and to put more resources into the research,” added Dr. Piltonen, professor in the departments of ob.gyn. and reproductive endocrinology at the University of Oulu, Finland.
Indeed, Punith Kempegowda, MD, PhD, of the University of Birmingham (England) observed: “In our medical schools in the U.K., over 5 years, students get 45 minutes [of education] on PCOS, and they’re expected to learn about it.”
And over the last 20 years, funding for research into the condition has totaled less than a half percent of overall medical funding. “And we’re talking about 10% of all women. …We need to acknowledge it and educate people more. We need more published studies to understand more about it,” he noted.
Asked to comment, Greg Dodell, MD, owner and president of Central Park Endocrinology, New York, said: “PCOS is about a lot more than fertility, and that may not be the goal or on the mind of a woman at the time they start having symptoms of PCOS or get the diagnosis.”
“PCOS is largely a metabolic condition rooted in insulin resistance, and therefore, the potential clinical outcomes, including mortality, are important to recognize.”
Dr. Dodell, who has a special interest in PCOS, advised that, for women with the condition, “focus on reducing insulin resistance with health-promoting behaviors and medications as needed. Data demonstrate that improving fitness, irrespective of a change in weight, can improve metabolic markers.” And, he advised that these women be routinely screened for mental health issues.
He also noted, “PCOS occurs across the size spectrum, but those patients in larger bodies may face weight stigma which has negative health consequences. These patients may avoid going to doctors for routine health screenings, so it is an important issue to continue to address.”
Women with PCOS lose a year of life
The new data come from 9,839 women with PCOS and 70,705 age- and region-matched controls from the Finnish Care Register for Health Care. The group with PCOS had been diagnosed at a mean age of 27 years.
The mean follow-up time was 13.1 years in both groups, during which 1,003 controls and 177 women with PCOS died. The mean age at death was 51.4 years for the PCOS group versus 52.6 years for the control women, a significant difference (P < .001).
Causes of death that were significantly higher among the women with PCOS versus controls after adjustments were cancer (hazard ratio, 1.39), and diseases of the circulatory system (1.68).
In more specific subcategories, after adjustment for education, the women with PCOS had increased mortality from nonischemic diseases, such as hypertensive heart disease, pulmonary embolism, etc. (HR, 2.06), and diabetes (HR, 2.85).
One study limitation was the inability to adjust for body mass index, Dr. Piltonen noted.
Dr. Piltonen, Dr. Kempegowda, and Dr. Dodell have no disclosures.
CHICAGO –
In the study, involving nearly 10,000 women with PCOS and matched controls from Finland, women with PCOS died on average a year earlier than their age-matched counterparts, primarily from diseases of the circulatory system, cancer, and diabetes.
PCOS is the most common endocrine disorder of reproductive-age women, of whom about 50%-70% also have obesity.
“I think we need to acknowledge that this is a health burden and not just a reproductive problem. In many cases we deal with the reproductive problem, and then these women are left alone. … So I think the message is we need to look beyond the reproductive outcomes, which are … really good. We can manage that,” said Terhi T. Piltonen, MD, PhD, during a press briefing held June 15 at the annual meeting of the Endocrine Society.
“I think the difficult part is [managing] the lifelong health for these women and supporting them to achieve the best health they can get. We need a multidisciplinary effort and to put more resources into the research,” added Dr. Piltonen, professor in the departments of ob.gyn. and reproductive endocrinology at the University of Oulu, Finland.
Indeed, Punith Kempegowda, MD, PhD, of the University of Birmingham (England) observed: “In our medical schools in the U.K., over 5 years, students get 45 minutes [of education] on PCOS, and they’re expected to learn about it.”
And over the last 20 years, funding for research into the condition has totaled less than a half percent of overall medical funding. “And we’re talking about 10% of all women. …We need to acknowledge it and educate people more. We need more published studies to understand more about it,” he noted.
Asked to comment, Greg Dodell, MD, owner and president of Central Park Endocrinology, New York, said: “PCOS is about a lot more than fertility, and that may not be the goal or on the mind of a woman at the time they start having symptoms of PCOS or get the diagnosis.”
“PCOS is largely a metabolic condition rooted in insulin resistance, and therefore, the potential clinical outcomes, including mortality, are important to recognize.”
Dr. Dodell, who has a special interest in PCOS, advised that, for women with the condition, “focus on reducing insulin resistance with health-promoting behaviors and medications as needed. Data demonstrate that improving fitness, irrespective of a change in weight, can improve metabolic markers.” And, he advised that these women be routinely screened for mental health issues.
He also noted, “PCOS occurs across the size spectrum, but those patients in larger bodies may face weight stigma which has negative health consequences. These patients may avoid going to doctors for routine health screenings, so it is an important issue to continue to address.”
Women with PCOS lose a year of life
The new data come from 9,839 women with PCOS and 70,705 age- and region-matched controls from the Finnish Care Register for Health Care. The group with PCOS had been diagnosed at a mean age of 27 years.
The mean follow-up time was 13.1 years in both groups, during which 1,003 controls and 177 women with PCOS died. The mean age at death was 51.4 years for the PCOS group versus 52.6 years for the control women, a significant difference (P < .001).
Causes of death that were significantly higher among the women with PCOS versus controls after adjustments were cancer (hazard ratio, 1.39), and diseases of the circulatory system (1.68).
In more specific subcategories, after adjustment for education, the women with PCOS had increased mortality from nonischemic diseases, such as hypertensive heart disease, pulmonary embolism, etc. (HR, 2.06), and diabetes (HR, 2.85).
One study limitation was the inability to adjust for body mass index, Dr. Piltonen noted.
Dr. Piltonen, Dr. Kempegowda, and Dr. Dodell have no disclosures.
CHICAGO –
In the study, involving nearly 10,000 women with PCOS and matched controls from Finland, women with PCOS died on average a year earlier than their age-matched counterparts, primarily from diseases of the circulatory system, cancer, and diabetes.
PCOS is the most common endocrine disorder of reproductive-age women, of whom about 50%-70% also have obesity.
“I think we need to acknowledge that this is a health burden and not just a reproductive problem. In many cases we deal with the reproductive problem, and then these women are left alone. … So I think the message is we need to look beyond the reproductive outcomes, which are … really good. We can manage that,” said Terhi T. Piltonen, MD, PhD, during a press briefing held June 15 at the annual meeting of the Endocrine Society.
“I think the difficult part is [managing] the lifelong health for these women and supporting them to achieve the best health they can get. We need a multidisciplinary effort and to put more resources into the research,” added Dr. Piltonen, professor in the departments of ob.gyn. and reproductive endocrinology at the University of Oulu, Finland.
Indeed, Punith Kempegowda, MD, PhD, of the University of Birmingham (England) observed: “In our medical schools in the U.K., over 5 years, students get 45 minutes [of education] on PCOS, and they’re expected to learn about it.”
And over the last 20 years, funding for research into the condition has totaled less than a half percent of overall medical funding. “And we’re talking about 10% of all women. …We need to acknowledge it and educate people more. We need more published studies to understand more about it,” he noted.
Asked to comment, Greg Dodell, MD, owner and president of Central Park Endocrinology, New York, said: “PCOS is about a lot more than fertility, and that may not be the goal or on the mind of a woman at the time they start having symptoms of PCOS or get the diagnosis.”
“PCOS is largely a metabolic condition rooted in insulin resistance, and therefore, the potential clinical outcomes, including mortality, are important to recognize.”
Dr. Dodell, who has a special interest in PCOS, advised that, for women with the condition, “focus on reducing insulin resistance with health-promoting behaviors and medications as needed. Data demonstrate that improving fitness, irrespective of a change in weight, can improve metabolic markers.” And, he advised that these women be routinely screened for mental health issues.
He also noted, “PCOS occurs across the size spectrum, but those patients in larger bodies may face weight stigma which has negative health consequences. These patients may avoid going to doctors for routine health screenings, so it is an important issue to continue to address.”
Women with PCOS lose a year of life
The new data come from 9,839 women with PCOS and 70,705 age- and region-matched controls from the Finnish Care Register for Health Care. The group with PCOS had been diagnosed at a mean age of 27 years.
The mean follow-up time was 13.1 years in both groups, during which 1,003 controls and 177 women with PCOS died. The mean age at death was 51.4 years for the PCOS group versus 52.6 years for the control women, a significant difference (P < .001).
Causes of death that were significantly higher among the women with PCOS versus controls after adjustments were cancer (hazard ratio, 1.39), and diseases of the circulatory system (1.68).
In more specific subcategories, after adjustment for education, the women with PCOS had increased mortality from nonischemic diseases, such as hypertensive heart disease, pulmonary embolism, etc. (HR, 2.06), and diabetes (HR, 2.85).
One study limitation was the inability to adjust for body mass index, Dr. Piltonen noted.
Dr. Piltonen, Dr. Kempegowda, and Dr. Dodell have no disclosures.
AT ENDO 2023
Cancer drug shortages spur worry, rationing, and tough choices
CHICAGO – Oncologist Denise Yardley, MD, isn’t used to expressing uncertainty when she tells patients about what’s in store for them in terms of drug treatment. But things are dramatically different now amid a severe national shortage of carboplatin and cisplatin, two common and crucial cancer drugs.
“There’s a regimen I’m thinking about,” Dr. Yardley told a new patient recently, “but we’ll have to wait until you finish your staging evaluation to see whether I can deliver this. Another regimen that’s a little more toxic is my second choice.” And, she added, the alternative chemotherapy treatment – anthracycline instead of carboplatin – requires a longer treatment period.
This ambiguity is hardly ideal, said Dr. Yardley, of Tennessee Oncology and Sarah Cannon Research Institute in Nashville. “It’s another factor in being overwhelmed in a first-time visit and wanting to know the details about what your treatment is going to look like. You’re not walking out knowing exactly what you’re going to take or the exact timing so you can start mapping out your calendar and work schedule.”
This kind of scenario is becoming all too familiar this spring, according to oncologists who gathered at the annual meeting of the American Society of Clinical Oncology (ASCO). In interviews, these physicians said the limited supply of multiple cancer drugs – including the chemotherapies carboplatin and cisplatin – is having an unprecedented negative effect since their use is so widespread in cancer care.
“Every patient could get impacted. That’s why we need to address this sooner rather than later,” said oncologist Aditya Baria, MBBS, MPH, director of the Breast Cancer Research Program at Massachusetts General Hospital, Boston.
Shortages of cancer drugs are not unusual. Three-quarters of oncology pharmacists at 68 organizations surveyed from 2019 to 2020 said shortages prompted treatment delays, reduced doses, or alternative regimens. But the current shortages are having a much wider impact.
The National Comprehensive Cancer Network recently reported that 93% of 27 member institutions surveyed in late May are short on carboplatin, and 70% have reported a shortage of cisplatin. Plus, 20% of 19 centers said they weren’t able to continue carboplatin regimens for all patients.
The drugs are mainstays of multiple types of treatment for a long list of cancer types including lung, breast, gynecologic, and many others.
Several scenarios are possible when the drugs are in short supply, said Dr. Yardley, who noted that the shortage is more severe than any she’s seen in her medical career of more than 3 decades. Patients may need to be switched to regimens with more side effects, even when they’re in the middle of a treatment, she said. Or patients might have to go longer between treatments.
In some cases, Dr. Yardley said, the shortage is forcing patients to go without an important component of a larger combination therapy regimen. “The Keynote 522 neoadjuvant regimen for triple-negative breast cancer has carboplatin given with Taxol [paclitaxel] and Keytruda [pembrolizumab]. We are just deleting the carboplatin.”
She added that carboplatin is part of the following so-called TCHP regimen for HER2+ early-stage breast cancer: Taxotere (docetaxel), carboplatin, Herceptin (trastuzumab), and Perjeta (pertuzumab).
“You can delete [carboplatin] or consider substituting cyclophosphamide for carboplatin,” she said. But she cautioned the Keynote 522 and TCHP regimens haven’t been tested without carboplatin in curative-intent trials.
At Duke University in Durham, N.C., doses of carboplatin for many patients are being lowered by a third to the level that’s commonly used for older and frail patients, said oncologist Arif Kamal, MD, MBA, MHS, who works at the academic center and is the chief patient officer at the American Cancer Society.
“We don’t know if [the lower doses will negatively affect cancer patients’ outcomes]. What’s amazing is how many patients [are understanding about having to take smaller amounts of the chemotherapy],” he said.
Medical organizations are offering guidance. The Society of Gynecologic Oncology, for example, in late April recommended that oncologists increase intervals between chemotherapy treatments when appropriate, round down vial sizes to ensure “efficient use,” and eliminate or minimize use of cisplatin and carboplatin in certain platinum-resistant cancers.
In early June, ASCO published guidance regarding alternatives to cisplatin, carboplatin, and 5-fluorouracil, which is also in short supply, in gastrointestinal cancer. As the guidance notes, some alternatives are more untested or more toxic than ideal treatments.
In addition, ASCO has a webpage devoted to news and resources about shortages of cancer drugs. It offers drug availability updates, general guidance, and breast cancer guidance. ASCO also offers ethical guidance about handling drug shortages.
Patients in clinical trials and those who hope to join them are especially vulnerable to the drug shortage, oncologists interviewed for this story said. Cisplatin and carboplatin are the backbones of many clinical trials, Dr. Yardley said. “When you can’t supply a drug in one of the [trial] arms, that puts the whole trial on pause.”
Even clinics that have managed to find adequate supplies of the drugs are planning for when they run out.
“Our institution and other institutions are trying to come up with a rationing protocol, deciding which patients are going to get access, and which ones have reasonable alternatives,” radiation oncologist Corey Speers MD, PhD, of University Hospitals Seidman Cancer Center and Case Western Reserve University, Cleveland, said in an interview. “In some settings, there really isn’t an effective alternative. Or the alternatives are tens of thousands of dollars more expensive.”
Oncologists also noted that cisplatin and carboplatin aren’t the only cancer drugs in short supply.
“Methotrexate is critically low, and 5FU [fluorouracil] is critically low,” Dr. Yardley said, referring to drugs that each treat several types of cancer. According to the May NCNN survey, 67% of respondents reported low supplies of methotrexate, and 26% said they were low on 5FU.
“Viscous lidocaine is a component of many supportive care mouth rinses for the stomatitis caused by our drugs but is not available at all,” Dr. Yardley said.
She added that there are also low supplies of fludarabine, which is used to treat chronic lymphocytic lymphom; clofarabine, which is used to treat acute lymphoblastic leukemia; and rasburicase, which is used to treat high levels of uric acid in patients on chemotherapy.
Dr. Speers said his institution is facing a shortage of capecitabine, which is used to treat several types of cancer.
“Numerous trials have demonstrated the improved, safety, efficacy, and convenience of oral capecitabine. With the shortage we’re having to use infusional 5FU, which not only is less convenient but also ends up being more costly and requires infusion room space or continuous infusion pumps. This impacts our ability to treat cancer patients,” he said. “Our capacity is becoming more limited to accommodate these added patients, and we have to use infusional formulations of a drug that previously was readily available via an oral formulation. Patients and caregivers now have to come to the cancer center for appointments and infusions that previously weren’t needed as they could take an oral pill.”
Dr. Speers added that his institution is rationing methotrexate. “We are now prioritizing patients being treated with curative intent and adjusting protocols to use the lowest allowable doses to conserve supply,” he said.
The roots of the platinum chemotherapy drug shortage link back to the India-based Intas Pharmaceuticals company, a major manufacturer of cisplatin and carboplatin. According to Kellyann Zuzulo, spokeperson for Accord Healthcare, an Instas U.S. subsidiary, a facility inspection in December 2022 prompted a decision to temporarily stop making the drugs. The inspection identified multiple problems.
“Intas and Accord are working with the FDA on a plan to return to manufacturing,” Ms. Zuzulo said in an interview. “This will allow for continued production of products that will be prioritized based on medical necessity. A date has not yet been confirmed in which the facility will return to manufacturing for cisplatin, carboplatin or any other products.”
Ms. Zuzulo said the company is not a health care provider and cannot offer advice to patients about alternatives.
Other companies that make cisplatin and carboplatin have also reported shortages. In interviews, representatives for Fresenius Kabi and Pfizer said the companies have limited supplies because of increased demand – not because of manufacturing problems.
On June 12, the American Society of Health-System Pharmacists (ASHP) reported that carboplatin remains in short supply, with all five companies that sell the drug listed as having limited or back-ordered supplies. Cisplatin is also in short supply, the organization reported in a June 9 update, although some is available.
In a June 12 update on methotrexate, ASHP said manufacturing delays at Accord have caused a shortage, and other companies are running low due to increased demand.
As for the future, Congress and the Biden administration, according to a report by Bloomberg, are trying to figure out what to do regarding shortages of cheap generic drugs such as cisplatin and carboplatin. The FDA is exploring a partnership with a Chinese drugmaker to make cisplatin, NBC News reported.
However, fixes will be challenging, according to former FDA commissioner and Pfizer board member, Scott Gottlieb, MD.
“This generic business, particularly for these complex drugs, these complex formulations, is not a healthy business right now. Yet it’s a vital business from a public standpoint,” he told CBS News.
In an interview, Dr. Kamal said that there is even talk about boosting the prices of cheap generic drugs “to ensure that there’s enough incentive for multiple manufacturers to be involved.”
Dr. Kamal said he is crossing his fingers that cutting chemotherapy doses at his clinic doesn’t result in worse outcomes for his patients.
“Right now, I think dropping someone by 25% or 30% is okay. And for some patients, particularly in a curative setting, we try to keep them at as much as 100% as possible. But there’s just a lot of unknowns,” he said.
CHICAGO – Oncologist Denise Yardley, MD, isn’t used to expressing uncertainty when she tells patients about what’s in store for them in terms of drug treatment. But things are dramatically different now amid a severe national shortage of carboplatin and cisplatin, two common and crucial cancer drugs.
“There’s a regimen I’m thinking about,” Dr. Yardley told a new patient recently, “but we’ll have to wait until you finish your staging evaluation to see whether I can deliver this. Another regimen that’s a little more toxic is my second choice.” And, she added, the alternative chemotherapy treatment – anthracycline instead of carboplatin – requires a longer treatment period.
This ambiguity is hardly ideal, said Dr. Yardley, of Tennessee Oncology and Sarah Cannon Research Institute in Nashville. “It’s another factor in being overwhelmed in a first-time visit and wanting to know the details about what your treatment is going to look like. You’re not walking out knowing exactly what you’re going to take or the exact timing so you can start mapping out your calendar and work schedule.”
This kind of scenario is becoming all too familiar this spring, according to oncologists who gathered at the annual meeting of the American Society of Clinical Oncology (ASCO). In interviews, these physicians said the limited supply of multiple cancer drugs – including the chemotherapies carboplatin and cisplatin – is having an unprecedented negative effect since their use is so widespread in cancer care.
“Every patient could get impacted. That’s why we need to address this sooner rather than later,” said oncologist Aditya Baria, MBBS, MPH, director of the Breast Cancer Research Program at Massachusetts General Hospital, Boston.
Shortages of cancer drugs are not unusual. Three-quarters of oncology pharmacists at 68 organizations surveyed from 2019 to 2020 said shortages prompted treatment delays, reduced doses, or alternative regimens. But the current shortages are having a much wider impact.
The National Comprehensive Cancer Network recently reported that 93% of 27 member institutions surveyed in late May are short on carboplatin, and 70% have reported a shortage of cisplatin. Plus, 20% of 19 centers said they weren’t able to continue carboplatin regimens for all patients.
The drugs are mainstays of multiple types of treatment for a long list of cancer types including lung, breast, gynecologic, and many others.
Several scenarios are possible when the drugs are in short supply, said Dr. Yardley, who noted that the shortage is more severe than any she’s seen in her medical career of more than 3 decades. Patients may need to be switched to regimens with more side effects, even when they’re in the middle of a treatment, she said. Or patients might have to go longer between treatments.
In some cases, Dr. Yardley said, the shortage is forcing patients to go without an important component of a larger combination therapy regimen. “The Keynote 522 neoadjuvant regimen for triple-negative breast cancer has carboplatin given with Taxol [paclitaxel] and Keytruda [pembrolizumab]. We are just deleting the carboplatin.”
She added that carboplatin is part of the following so-called TCHP regimen for HER2+ early-stage breast cancer: Taxotere (docetaxel), carboplatin, Herceptin (trastuzumab), and Perjeta (pertuzumab).
“You can delete [carboplatin] or consider substituting cyclophosphamide for carboplatin,” she said. But she cautioned the Keynote 522 and TCHP regimens haven’t been tested without carboplatin in curative-intent trials.
At Duke University in Durham, N.C., doses of carboplatin for many patients are being lowered by a third to the level that’s commonly used for older and frail patients, said oncologist Arif Kamal, MD, MBA, MHS, who works at the academic center and is the chief patient officer at the American Cancer Society.
“We don’t know if [the lower doses will negatively affect cancer patients’ outcomes]. What’s amazing is how many patients [are understanding about having to take smaller amounts of the chemotherapy],” he said.
Medical organizations are offering guidance. The Society of Gynecologic Oncology, for example, in late April recommended that oncologists increase intervals between chemotherapy treatments when appropriate, round down vial sizes to ensure “efficient use,” and eliminate or minimize use of cisplatin and carboplatin in certain platinum-resistant cancers.
In early June, ASCO published guidance regarding alternatives to cisplatin, carboplatin, and 5-fluorouracil, which is also in short supply, in gastrointestinal cancer. As the guidance notes, some alternatives are more untested or more toxic than ideal treatments.
In addition, ASCO has a webpage devoted to news and resources about shortages of cancer drugs. It offers drug availability updates, general guidance, and breast cancer guidance. ASCO also offers ethical guidance about handling drug shortages.
Patients in clinical trials and those who hope to join them are especially vulnerable to the drug shortage, oncologists interviewed for this story said. Cisplatin and carboplatin are the backbones of many clinical trials, Dr. Yardley said. “When you can’t supply a drug in one of the [trial] arms, that puts the whole trial on pause.”
Even clinics that have managed to find adequate supplies of the drugs are planning for when they run out.
“Our institution and other institutions are trying to come up with a rationing protocol, deciding which patients are going to get access, and which ones have reasonable alternatives,” radiation oncologist Corey Speers MD, PhD, of University Hospitals Seidman Cancer Center and Case Western Reserve University, Cleveland, said in an interview. “In some settings, there really isn’t an effective alternative. Or the alternatives are tens of thousands of dollars more expensive.”
Oncologists also noted that cisplatin and carboplatin aren’t the only cancer drugs in short supply.
“Methotrexate is critically low, and 5FU [fluorouracil] is critically low,” Dr. Yardley said, referring to drugs that each treat several types of cancer. According to the May NCNN survey, 67% of respondents reported low supplies of methotrexate, and 26% said they were low on 5FU.
“Viscous lidocaine is a component of many supportive care mouth rinses for the stomatitis caused by our drugs but is not available at all,” Dr. Yardley said.
She added that there are also low supplies of fludarabine, which is used to treat chronic lymphocytic lymphom; clofarabine, which is used to treat acute lymphoblastic leukemia; and rasburicase, which is used to treat high levels of uric acid in patients on chemotherapy.
Dr. Speers said his institution is facing a shortage of capecitabine, which is used to treat several types of cancer.
“Numerous trials have demonstrated the improved, safety, efficacy, and convenience of oral capecitabine. With the shortage we’re having to use infusional 5FU, which not only is less convenient but also ends up being more costly and requires infusion room space or continuous infusion pumps. This impacts our ability to treat cancer patients,” he said. “Our capacity is becoming more limited to accommodate these added patients, and we have to use infusional formulations of a drug that previously was readily available via an oral formulation. Patients and caregivers now have to come to the cancer center for appointments and infusions that previously weren’t needed as they could take an oral pill.”
Dr. Speers added that his institution is rationing methotrexate. “We are now prioritizing patients being treated with curative intent and adjusting protocols to use the lowest allowable doses to conserve supply,” he said.
The roots of the platinum chemotherapy drug shortage link back to the India-based Intas Pharmaceuticals company, a major manufacturer of cisplatin and carboplatin. According to Kellyann Zuzulo, spokeperson for Accord Healthcare, an Instas U.S. subsidiary, a facility inspection in December 2022 prompted a decision to temporarily stop making the drugs. The inspection identified multiple problems.
“Intas and Accord are working with the FDA on a plan to return to manufacturing,” Ms. Zuzulo said in an interview. “This will allow for continued production of products that will be prioritized based on medical necessity. A date has not yet been confirmed in which the facility will return to manufacturing for cisplatin, carboplatin or any other products.”
Ms. Zuzulo said the company is not a health care provider and cannot offer advice to patients about alternatives.
Other companies that make cisplatin and carboplatin have also reported shortages. In interviews, representatives for Fresenius Kabi and Pfizer said the companies have limited supplies because of increased demand – not because of manufacturing problems.
On June 12, the American Society of Health-System Pharmacists (ASHP) reported that carboplatin remains in short supply, with all five companies that sell the drug listed as having limited or back-ordered supplies. Cisplatin is also in short supply, the organization reported in a June 9 update, although some is available.
In a June 12 update on methotrexate, ASHP said manufacturing delays at Accord have caused a shortage, and other companies are running low due to increased demand.
As for the future, Congress and the Biden administration, according to a report by Bloomberg, are trying to figure out what to do regarding shortages of cheap generic drugs such as cisplatin and carboplatin. The FDA is exploring a partnership with a Chinese drugmaker to make cisplatin, NBC News reported.
However, fixes will be challenging, according to former FDA commissioner and Pfizer board member, Scott Gottlieb, MD.
“This generic business, particularly for these complex drugs, these complex formulations, is not a healthy business right now. Yet it’s a vital business from a public standpoint,” he told CBS News.
In an interview, Dr. Kamal said that there is even talk about boosting the prices of cheap generic drugs “to ensure that there’s enough incentive for multiple manufacturers to be involved.”
Dr. Kamal said he is crossing his fingers that cutting chemotherapy doses at his clinic doesn’t result in worse outcomes for his patients.
“Right now, I think dropping someone by 25% or 30% is okay. And for some patients, particularly in a curative setting, we try to keep them at as much as 100% as possible. But there’s just a lot of unknowns,” he said.
CHICAGO – Oncologist Denise Yardley, MD, isn’t used to expressing uncertainty when she tells patients about what’s in store for them in terms of drug treatment. But things are dramatically different now amid a severe national shortage of carboplatin and cisplatin, two common and crucial cancer drugs.
“There’s a regimen I’m thinking about,” Dr. Yardley told a new patient recently, “but we’ll have to wait until you finish your staging evaluation to see whether I can deliver this. Another regimen that’s a little more toxic is my second choice.” And, she added, the alternative chemotherapy treatment – anthracycline instead of carboplatin – requires a longer treatment period.
This ambiguity is hardly ideal, said Dr. Yardley, of Tennessee Oncology and Sarah Cannon Research Institute in Nashville. “It’s another factor in being overwhelmed in a first-time visit and wanting to know the details about what your treatment is going to look like. You’re not walking out knowing exactly what you’re going to take or the exact timing so you can start mapping out your calendar and work schedule.”
This kind of scenario is becoming all too familiar this spring, according to oncologists who gathered at the annual meeting of the American Society of Clinical Oncology (ASCO). In interviews, these physicians said the limited supply of multiple cancer drugs – including the chemotherapies carboplatin and cisplatin – is having an unprecedented negative effect since their use is so widespread in cancer care.
“Every patient could get impacted. That’s why we need to address this sooner rather than later,” said oncologist Aditya Baria, MBBS, MPH, director of the Breast Cancer Research Program at Massachusetts General Hospital, Boston.
Shortages of cancer drugs are not unusual. Three-quarters of oncology pharmacists at 68 organizations surveyed from 2019 to 2020 said shortages prompted treatment delays, reduced doses, or alternative regimens. But the current shortages are having a much wider impact.
The National Comprehensive Cancer Network recently reported that 93% of 27 member institutions surveyed in late May are short on carboplatin, and 70% have reported a shortage of cisplatin. Plus, 20% of 19 centers said they weren’t able to continue carboplatin regimens for all patients.
The drugs are mainstays of multiple types of treatment for a long list of cancer types including lung, breast, gynecologic, and many others.
Several scenarios are possible when the drugs are in short supply, said Dr. Yardley, who noted that the shortage is more severe than any she’s seen in her medical career of more than 3 decades. Patients may need to be switched to regimens with more side effects, even when they’re in the middle of a treatment, she said. Or patients might have to go longer between treatments.
In some cases, Dr. Yardley said, the shortage is forcing patients to go without an important component of a larger combination therapy regimen. “The Keynote 522 neoadjuvant regimen for triple-negative breast cancer has carboplatin given with Taxol [paclitaxel] and Keytruda [pembrolizumab]. We are just deleting the carboplatin.”
She added that carboplatin is part of the following so-called TCHP regimen for HER2+ early-stage breast cancer: Taxotere (docetaxel), carboplatin, Herceptin (trastuzumab), and Perjeta (pertuzumab).
“You can delete [carboplatin] or consider substituting cyclophosphamide for carboplatin,” she said. But she cautioned the Keynote 522 and TCHP regimens haven’t been tested without carboplatin in curative-intent trials.
At Duke University in Durham, N.C., doses of carboplatin for many patients are being lowered by a third to the level that’s commonly used for older and frail patients, said oncologist Arif Kamal, MD, MBA, MHS, who works at the academic center and is the chief patient officer at the American Cancer Society.
“We don’t know if [the lower doses will negatively affect cancer patients’ outcomes]. What’s amazing is how many patients [are understanding about having to take smaller amounts of the chemotherapy],” he said.
Medical organizations are offering guidance. The Society of Gynecologic Oncology, for example, in late April recommended that oncologists increase intervals between chemotherapy treatments when appropriate, round down vial sizes to ensure “efficient use,” and eliminate or minimize use of cisplatin and carboplatin in certain platinum-resistant cancers.
In early June, ASCO published guidance regarding alternatives to cisplatin, carboplatin, and 5-fluorouracil, which is also in short supply, in gastrointestinal cancer. As the guidance notes, some alternatives are more untested or more toxic than ideal treatments.
In addition, ASCO has a webpage devoted to news and resources about shortages of cancer drugs. It offers drug availability updates, general guidance, and breast cancer guidance. ASCO also offers ethical guidance about handling drug shortages.
Patients in clinical trials and those who hope to join them are especially vulnerable to the drug shortage, oncologists interviewed for this story said. Cisplatin and carboplatin are the backbones of many clinical trials, Dr. Yardley said. “When you can’t supply a drug in one of the [trial] arms, that puts the whole trial on pause.”
Even clinics that have managed to find adequate supplies of the drugs are planning for when they run out.
“Our institution and other institutions are trying to come up with a rationing protocol, deciding which patients are going to get access, and which ones have reasonable alternatives,” radiation oncologist Corey Speers MD, PhD, of University Hospitals Seidman Cancer Center and Case Western Reserve University, Cleveland, said in an interview. “In some settings, there really isn’t an effective alternative. Or the alternatives are tens of thousands of dollars more expensive.”
Oncologists also noted that cisplatin and carboplatin aren’t the only cancer drugs in short supply.
“Methotrexate is critically low, and 5FU [fluorouracil] is critically low,” Dr. Yardley said, referring to drugs that each treat several types of cancer. According to the May NCNN survey, 67% of respondents reported low supplies of methotrexate, and 26% said they were low on 5FU.
“Viscous lidocaine is a component of many supportive care mouth rinses for the stomatitis caused by our drugs but is not available at all,” Dr. Yardley said.
She added that there are also low supplies of fludarabine, which is used to treat chronic lymphocytic lymphom; clofarabine, which is used to treat acute lymphoblastic leukemia; and rasburicase, which is used to treat high levels of uric acid in patients on chemotherapy.
Dr. Speers said his institution is facing a shortage of capecitabine, which is used to treat several types of cancer.
“Numerous trials have demonstrated the improved, safety, efficacy, and convenience of oral capecitabine. With the shortage we’re having to use infusional 5FU, which not only is less convenient but also ends up being more costly and requires infusion room space or continuous infusion pumps. This impacts our ability to treat cancer patients,” he said. “Our capacity is becoming more limited to accommodate these added patients, and we have to use infusional formulations of a drug that previously was readily available via an oral formulation. Patients and caregivers now have to come to the cancer center for appointments and infusions that previously weren’t needed as they could take an oral pill.”
Dr. Speers added that his institution is rationing methotrexate. “We are now prioritizing patients being treated with curative intent and adjusting protocols to use the lowest allowable doses to conserve supply,” he said.
The roots of the platinum chemotherapy drug shortage link back to the India-based Intas Pharmaceuticals company, a major manufacturer of cisplatin and carboplatin. According to Kellyann Zuzulo, spokeperson for Accord Healthcare, an Instas U.S. subsidiary, a facility inspection in December 2022 prompted a decision to temporarily stop making the drugs. The inspection identified multiple problems.
“Intas and Accord are working with the FDA on a plan to return to manufacturing,” Ms. Zuzulo said in an interview. “This will allow for continued production of products that will be prioritized based on medical necessity. A date has not yet been confirmed in which the facility will return to manufacturing for cisplatin, carboplatin or any other products.”
Ms. Zuzulo said the company is not a health care provider and cannot offer advice to patients about alternatives.
Other companies that make cisplatin and carboplatin have also reported shortages. In interviews, representatives for Fresenius Kabi and Pfizer said the companies have limited supplies because of increased demand – not because of manufacturing problems.
On June 12, the American Society of Health-System Pharmacists (ASHP) reported that carboplatin remains in short supply, with all five companies that sell the drug listed as having limited or back-ordered supplies. Cisplatin is also in short supply, the organization reported in a June 9 update, although some is available.
In a June 12 update on methotrexate, ASHP said manufacturing delays at Accord have caused a shortage, and other companies are running low due to increased demand.
As for the future, Congress and the Biden administration, according to a report by Bloomberg, are trying to figure out what to do regarding shortages of cheap generic drugs such as cisplatin and carboplatin. The FDA is exploring a partnership with a Chinese drugmaker to make cisplatin, NBC News reported.
However, fixes will be challenging, according to former FDA commissioner and Pfizer board member, Scott Gottlieb, MD.
“This generic business, particularly for these complex drugs, these complex formulations, is not a healthy business right now. Yet it’s a vital business from a public standpoint,” he told CBS News.
In an interview, Dr. Kamal said that there is even talk about boosting the prices of cheap generic drugs “to ensure that there’s enough incentive for multiple manufacturers to be involved.”
Dr. Kamal said he is crossing his fingers that cutting chemotherapy doses at his clinic doesn’t result in worse outcomes for his patients.
“Right now, I think dropping someone by 25% or 30% is okay. And for some patients, particularly in a curative setting, we try to keep them at as much as 100% as possible. But there’s just a lot of unknowns,” he said.
AT ASCO 2023
10 ways in which ObGyn care can be more environmentally sustainable
Climate change has been called the biggest health threat of the 21st century.1 The health care sector is a huge contributor to global carbon emissions, accounting for almost double the emissions of global aviation. While other industries and countries are implementing mitigation measures to decrease their emissions, health care is currently on track to double its carbon emissions by 2050, even though it should be carbon neutral by that time to comply with the Paris Climate Agreement.2 There have been some national efforts to curb health care emissions, including the creation of the Office of Climate Change and Health Equity in 2021 and the passage of the Inflation Reduction Act in 2022.3 These are top-down, administrative approaches, and to be successful we will also need clinicians to understand and address this problem.
The negative impacts of heat, air pollution, and exposure to toxic substances on human health have been well documented in multiple regions across multiple specialties.4-7 The United States makes up 27% of the global health care carbon footprint—more emissions than the entire United Kingdom as a country—despite having only 4% of the world’s population.2 Culture and incentives for an overabundance of single-use supplies, not evidence for patient safety, have led to this uniquely American problem. It is evident that our health care industry is an excellent place to implement mitigation measures for carbon emissions that contribute to climate change and can improve health outcomes.
In this article, we recommend 10 practices that can decrease our carbon footprint in ObGyn. We focus on the classic motto of “Reduce, Reuse, Recycle,” while adding “Remove” and “Reimagine” to classify the ways in which we can reduce emissions while not compromising our care to patients.
Reduce
1. Minimize opened materials and single-use devices in the OR and labor and delivery
Health care is a unique setting where a culture of infection prevention and efficiency has led low-cost, single-use supplies to dominate over reusable items. While single-use items can have inexpensive purchasing costs compared to reusable items, the environmental costs required for the production and disposal of the former are often much greater. In operating rooms (ORs) and labor and delivery (LD) units, single-use items are omnipresent. Over the past decade, researchers and clinicians have started to take a closer look at these items and their carbon footprint. One group evaluated hysterectomy through a waste audit and found that the vast majority of waste from all of the cases was Spunbond Meltblown Spunbond, or SMS; plastic materialthat comprises gowns; blue wraps; and drapes; followed by hard plastic material that comprises trays and packaging.8 Moreover, production and manufacturing processes contributed to 95% of the environmental impacts of these items.8
In an effort to be time efficient, OR staff will open sterile surgical packs and individual peel-pack items prior to surgery to minimize having to find items during surgery. However, this creates an inordinate amount of waste. One group of neurosurgeons who evaluated their opened but unused supplies found that 85% of their unused items were individually opened items, leading to a waste of $2.9 million per year.9 Minor procedures like dilation and curettage, cystoscopy, and hysteroscopy do not need such a large sterile field, as these procedures are also safe to perform in the office. Hand surgeons have been quick to lead in this space, particularly with minor procedures such as carpal tunnel release. One division was able to eliminate 2.8 tons of waste and save $13,000 in a 2-year period by reducing the sterile field.10 ObGyns can work with OR and LD staff to create custom packs that minimize unused or underutilized items, helping to reduce both the carbon footprint and health care spending.
Bottom line: ObGyns can help foster a culture of having supplies available but not opened until needed during a case.
Continue to: 2. Decrease regulated medical waste...
2. Decrease regulated medical waste
Health care is unique from other fields in that there are multiple waste streams to consider. Infectious waste and items saturated in blood or capable of causing infection must be placed into regulated medical waste (RMW), or more commonly, red biohazard bags. RMW is autoclaved or incinerated prior to disposal in a landfill. This process is more financially and environmentally costly than general municipal waste (GMW). This process also requires more transport—1 study revealed that GMW traveled 20 km to a landfill for disposal, compared with the 50 km that RMW traveled for sterilized-prior-to-landfill disposal.11
Unfortunately, the vast majority of items placed in RMW are incorrectly triaged and should instead be disposed in GMW.12,13 One study performed in an emergency department revealed that 85% of waste was incorrectly placed in the RMW.12
Bottom line: ObGyns can avoid placing items in RMW that may not qualify and advocate for institution policy changes to remove RMW from places such as waiting rooms, at the patient bedside, or next to scrub sinks.
3. Reduce energy use
ORs and LD units use a lot of energy, and numerous studies have demonstrated that the heating, ventilation, and air conditioning (HVAC) system plays a large role in emissions.8,11 This can easily be fixed by “HVAC setbacks” and powering down rooms when not in use. One institution powered down ORs when not in use and reduced 234 metric tons of CO2 emissions and saved $33,000 per year.14 Transitioning to light-emitting diode (LED) lights reduced energy usage at 1 institution by almost 50%.15 Finally, computers in clinical offices, examination rooms, and administrative offices can be powered down at the end of the day. One study found that in 1 radiology department, 29 computers left on overnight and on weekends emitted 17.7 tons of CO2 emissions in 1 year.16
Bottom line: We as ObGyns can advocate for how energy can be saved outside of surgical cases, including powering down ORs and LD units, transitioning to LED lighting, and powering down workstations.
Reuse
4. Choose reusable equipment
In ObGyn practice, the most commonly used tool is the speculum. Given its omnipresence, the speculum is a great place to start to decrease our carbon footprint. Two studies have evaluated the environmental impact of reusable versus single-use disposable specula, and both demonstrated that the stainless-steel versions have less global warming potential than the acrylic varieties.17,18 Donahue and colleagues17 demonstrated that it only took 2 to 3 pelvic examinations for the cost of stainless-steel specula to break even, even when sterilized in a half-filled autoclave tray. Rodriquez, et al18 revealed that, compared with an acrylic model, the stainless-steel specula had fewer negative impacts in terms of global warming, acidification, respiratory effects, smog, and fossil fuel depletion.18
Bottom line: Strongly consider using stainless-steel specula to reduce costs and carbon emissions.
In addition to specula, ObGyns can choose reusable equipment in the OR. For example, surgeons can use stainless-steel trocars instead of disposable trocars.19 In vaginal cases, Breisky-Navratil retractors can be used instead of disposable self-retaining retractors. Plastic basins that often are included in sterile supply packs can be replaced with stainless-steel basins, which could have profound positive effects on the carbon footprint of gynecologic surgery.8 One study of ObGyns demonstrated that 95% of physicians supported waste-reduction efforts, and 66% supported utilizing reusable surgical tools instead of disposable tools.20
Bottom line: As surgeons, ObGyns have influence over what they want to use in the OR, and they can petition for reusable options over disposable options.
5. Launder the sterile blue towels
Sterile blue towels, which are made of cotton, have the largest environmental footprint compared with other disposable materials, such as plastics, and contribute greatly to toxicity in human health.8,11 Although these towels cannot be laundered and sterilized again for use in a sterile surgical field, they can be laundered and repurposed, including by environmental services to clean hospital rooms. Blue towels should be able to be laundered no matter how saturated in body fluids they are.
Bottom line: ObGyns should strive to always launder the blue towels and educate trainees and other staff in the OR to do the same.
Recycle
6. Recycle and reprocess materials and devices
While recycling is immensely important, it requires a large amount of energy to break down a material to its raw components for manufacturing. It likely reduces our carbon footprint from OR procedures by only 5%.8 However, recycling is still a good way to divert appropriate materials from landfill, saving costs and emissions at the end of a material’s life. One example is sterile blue wrap, which is a petroleum product with a recycling number of 6 and a filtration rating of N99. Blue wrap can be recycled into plastic pellets, or it can be recreated into other hospital supplies, such as gowns.
Bottom line: ObGyns can petition their hospitals to work with suppliers and waste-processing companies who have recycling programs built into their supply chains.
By contrast, reprocessing can have a much larger impact on carbon emissions. Complex items, such as advanced energy devices that can be reprocessed, result in a greater reduction in carbon emissions due to the reuse of their complex materials and manufacturing when compared with such devices that cannot be reprocessed. Recycling and reprocessing programs are already in place for several devices (TABLE). Authors of a systematic review showed that there is no evidence to support the use of single-use supplies and instruments over reprocessed items when considering instrument function, ease of use, patient safety, transmission of infection, or long-term patient outcomes.21
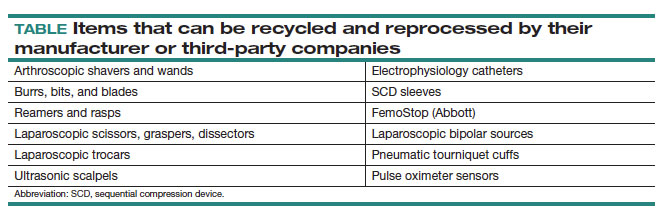
Bottom line: ObGyns can choose to use reprocessed items in ORs instead of single-use devices and educate staff on the safety of these items.
Continue to: Remove...
Remove
7. Remove desflurane and other volatile gases from formularies
Volatile anesthetic gases, such as desflurane, isoflurane, and nitrous oxide, are themselves potent greenhouse gases, comprising a large portion of the carbon emissions that come from the OR.22 Desflurane was developed to have a rapid onset for induction and quick recovery; however, studies have shown no clinical benefit over other gases.23 Furthermore, the costs and greenhouse gas potential are substantial. Desflurane costs 2 to 3 times more and has more than 20 times the global warming potential of the other volatile gases (FIGURE).8 Using 1 hour of desflurane is equivalent to driving 378 miles in a gas-powered vehicle, while the use of isoflurane and sevoflurane create equivalents of only 15 and 8 miles, respectively.23
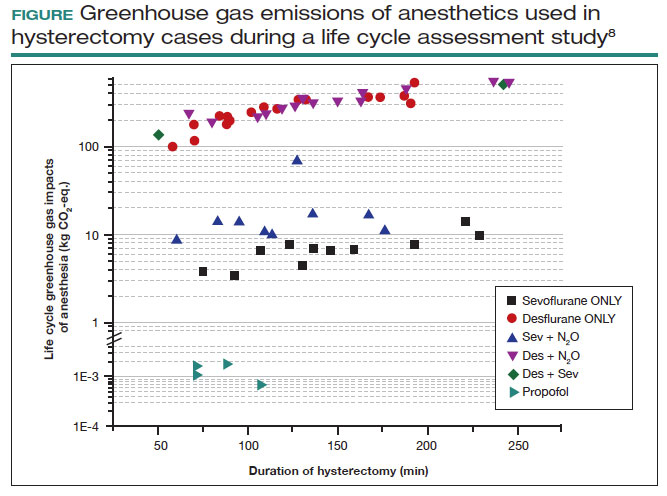
Nitrous oxide is another powerful greenhouse gas that is a direct ozone depletor and can stay in the atmosphere for 114 years.22 Nitrous oxide has limited clinical use in hospitals, but it is often stored in central hospital piping. Most of the impact of nitrous oxide comes through leaks in a poor system design rather than patient delivery. One estimate reveals that more than 13 million liters of nitrous oxide are lost annually from leaks in European hospitals.22 The American Society of Anesthesiologists recommends decommissioning central piping of nitrous oxide in favor of cylinders at the point of care.24
Literature on enhanced recovery after surgery in gynecology promotes the use of propofol over volatile gases for our patients because of the high rate of postoperative nausea and vomiting seen with gases.25 Volatile gases should be a last-choice anesthetic for our patients.
Bottom line: It is critical that ObGyns work with colleagues in anesthesia to develop climate- and patient-friendly protocols for procedures.
8. Remove endocrine-disrupting chemicals from clinical supplies
Endocrine-disrupting chemicals (EDCs) are a type of chemical that alter the hormonal systems of humans, which can result in adverse health effects. Multiple studies and reviews have tied EDCs to reproductive abnormalities, such as the effects of bisphenol A (BPA) on estradiol levels, antral follicle counts, oocyte quality, and implantation rates; phthalates on fibroid burden; triclosan on embryo quality; parabens on live birth rates; and perfluoroalkylsubstances (PFAS or “forever substances”) on hypertensive disorders of pregnancy.5,26,27
What might be most shocking is that these EDCs are incorporated into medical supplies and pharmaceuticals. For example, BPA is known to line dialysis and ointment tubes, parabens are used for their antimicrobial properties in ultrasound gel and hep-locks, and phthalates are found in up to 40% of medical-use plastics and controlled-release medications. Authors of an observational study found that 74% of patients admitted to an LD unit were exposed to EDCs. In a neonatal intensive care unit (NICU), most of the supplies contained an EDC, and urinary BPA levels were elevated in neonates admitted to a NICU, raising concerns about long-term health risks.5
Bottom line: Physicians and health care institutions have an obligation to petition industry partners and suppliers to remove EDCs from their supply chains.
Reimagine
9. Educate
The field of health care sustainability remains in its infancy, but from 2007 to 2019, publications on climate change and health in academia increased by a factor of 8.29 Additionally, through waste audits, quality-improvement projects, and life cycle analyses (analytical tools to evaluate product or process emissions from materials extraction to disposal), we have gained insight into the scope of the problem, with evidence showing that our practices are largely derived from culture. It is time to provide formal education on health care sustainability to medical trainees, staff, and clinicians alike, who desire to see this topic reflected in their formal curricula.30 Start talking about it!
Bottom line: Commentaries, webinars, formal didactics sessions, in-services, and hospital workgroups to introduce this topic are a good way to teach others about the carbon footprint of our care and solutions to minimize it.
10. Engage in advocacy
Physicians have an ethical duty to advocate for change at the local, regional, and national levels if we want to see a better future for our patients, their children, and even ourselves. We should reimagine this work as an important public health initiative.31 Surveys of physicians, including ObGyns, reveal a concern about the sustainability of health care and a commitment to addressing this issue.20 ObGyns are on the frontlines of delivering care every day, so we are poised to implement changes that can impact our patients, especially when we can lead and petition hospital or local committees.20,28,32 There is much to be done, but every voice counts and can make impactful changes at every level. ●
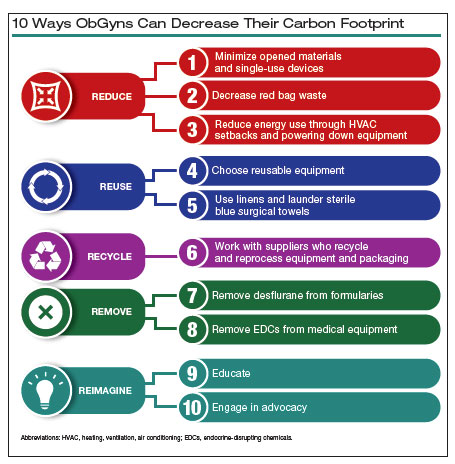
- Costello A, Abbas M, Allen et al. Managing the health effects of climate change: Lancet and University College London Institute for Global Health Commission. Lancet. 2009;373:1693-1733.
- Health care climate footprint report. Health Care Without Harm website. https://www.noharm.org/ClimateFootprintReport. Accessed May 12, 2023.
- Balbus JM, McCannon CJ, Mataka A, et al. After COP26—putting health and equity at the center of the climate movement. N Engl J Med. 2022;386:1295-1297.
- Bekkar B, Pacheco S, Basu R, et al. Association of air pollution and heat exposure with preterm birth, low birth weight, and stillbirth in the US: a systematic review. JAMA Netw Open. 2020;3:e208243.
- Genco M, Anderson-Shaw L, Sargis RM. Unwitting accomplices: endocrine disruptors confounding clinical care. J Clin Endocrinol Metab. 2020;105:e3822-e3827.
- Al-Kindi SG, Sarode A, Zullo M, et al. Ambient air pollution and mortality after cardiac transplantation. J Am Coll Cardiol. 2019;74:30263035.
- Ghosh R, Gauderman WJ, Minor H, et al. Air pollution, weight loss and metabolic benefits of bariatric surgery: a potential model for study of metabolic effects of environmental exposures. Pediatr Obes. 2018;13:312-320.
- Thiel CL, Eckelman M, Guido R, et al. Environmental impacts of surgical procedures: life cycle assessment of hysterectomy in the United States. Environ Sci Technol. 2015;49:1779-1786.
- Zygourakis CC, Yoon S, Valencia V, et al. Operating room waste: disposable supply utilization in neurosurgical procedures. J Neurosurg. 2017;126:620-625.
- van Demark RE, Smith VJS, Fiegen A. Lean and green hand surgery. J Hand Surg. 2018;43:179-181.
- Campion N, Thiel CL, DeBlois J, et al. Life cycle assessment perspectives on delivering an infant in the US. Sci Total Environ. 2012;425:191198.
- Hsu S, Thiel CL, Mello MJ, Slutzman JE. Dumpster diving in the emergency department. West J Emerg Med. 2020;21:1211-1217.
- Mcgain F, Story D, Hendel S. An audit of intensive care unit recyclable waste. Anaesthesia. 2009;64:1299-1302.
- Wormer BA, Augenstein VA, Carpenter CL, et al. The green operating room: simple changes to reduce cost and our carbon footprint. Am Surg. 2013;79:666-671.
- Kagoma Y, Stall N, Rubinstein E, et al. People, planet and profits: the case for greening operating rooms. Can Med Assoc J. 2012;184:19051911.
- McCarthy CJ, Gerstenmaier JF, O’ Neill AC, et al. “EcoRadiology”— pulling the plug on wasted energy in the radiology department. Acad Radiol. 2014;21:1563-1566.
- Donahue LM, Hilton S, Bell SG, et al. A comparative carbon footprint analysis of disposable and reusable vaginal specula. Am J Obstet Gynecol. 2020;223:225.e1-225.e7.
- Rodriguez Morris MI, Hicks A. Life cycle assessment of stainless-steel reusable speculums versus disposable acrylic speculums in a university clinic setting: a case study. Environ Res Commun. 2022;4:025002.
- MacNeill AJ, Lillywhite R, Brown CJ. The impact of surgery on global climate: a carbon footprinting study of operating theatres in three health systems. Lancet Planet Health. 2017;1:e381-e388.
- Thiel C, Duncan P, Woods N. Attitude of US obstetricians and gynaecologists to global warming and medical waste. J Health Serv Res Policy. 2017;22:162-167.
- Siu J, Hill AG, MacCormick AD. Systematic review of reusable versus disposable laparoscopic instruments: costs and safety. ANZ J Surg. 2017;87:28-33.
- Ryan SM, Nielsen CJ. Global warming potential of inhaled anesthetics: application to clinical use. Anesth Analg. 2010;111:92-98.
- Meyer MJ. Desflurane should des-appear: global and financial rationale. Anesth Analg. 2020;131:1317-1322.
- Rollins MD, Arendt KW, Carvalho B, et al. ASA Committee on Obstetric Anesthesia Working Group. Nitrous oxide. American Society of Anesthesiologists website. Accessed May 12, 2023. https://www .asahq.org/about-asa/governance-and-committees/asa-committees /committee-on-obstetric-anesthesia/nitrous-oxide.
- Kalogera E, Dowdy SC. Enhanced recovery pathway in gynecologic surgery: improving outcomes through evidence-based medicine. Obstet Gynecol Clin North Am. 2016;43:551-573.
- Zota AR, Geller RJ, Calafat AM, et al. Phthalates exposure and uterine fibroid burden among women undergoing surgical treatment for fibroids: a preliminary study. Fertil Steril. 2019;111:112-121.
- Bommartio PA, Ferguson KK, Meeker JD, et al. Maternal levels of perfluoroalkyl substances (PFAS) during early pregnancy in relation to preeclampsia subtypes and biomarkers of preeclampsia risk. Environ Health Perspect. 2021;129:107004.
- Azouz S, Boyll P, Swanson M, et al. Managing barriers to recycling in the operating room. Am J Surg. 2019;217:634-638.
- Watts N, Amann M, Arnell N, et al. The 2020 report of The Lancet Countdown on health and climate change: responding to converging crises. Lancet. 2021;397:129-170.
- Ryan EC, Dubrow R, Sherman JD. Medical, nursing, and physician assistant student knowledge and attitudes toward climate change, pollution, and resource conservation in health care. BMC Med Educ. 2020;20:200.
- Giudice LC, Llamas-Clark EF, DeNicola Net al; FIGO Committee on Climate Change and Toxic Environmental Exposures. Climate change, women’s health, and the role of obstetricians and gynecologists in leadership. Int J Gynaecol Obstet. 2021;155:345-356.
- Yates EF, Bowder AN, Roa L, et al. Empowering surgeons, anesthesiologists, and obstetricians to incorporate environmental sustainability in the operating room. Ann Surg. 2021;273:1108-1114.
Climate change has been called the biggest health threat of the 21st century.1 The health care sector is a huge contributor to global carbon emissions, accounting for almost double the emissions of global aviation. While other industries and countries are implementing mitigation measures to decrease their emissions, health care is currently on track to double its carbon emissions by 2050, even though it should be carbon neutral by that time to comply with the Paris Climate Agreement.2 There have been some national efforts to curb health care emissions, including the creation of the Office of Climate Change and Health Equity in 2021 and the passage of the Inflation Reduction Act in 2022.3 These are top-down, administrative approaches, and to be successful we will also need clinicians to understand and address this problem.
The negative impacts of heat, air pollution, and exposure to toxic substances on human health have been well documented in multiple regions across multiple specialties.4-7 The United States makes up 27% of the global health care carbon footprint—more emissions than the entire United Kingdom as a country—despite having only 4% of the world’s population.2 Culture and incentives for an overabundance of single-use supplies, not evidence for patient safety, have led to this uniquely American problem. It is evident that our health care industry is an excellent place to implement mitigation measures for carbon emissions that contribute to climate change and can improve health outcomes.
In this article, we recommend 10 practices that can decrease our carbon footprint in ObGyn. We focus on the classic motto of “Reduce, Reuse, Recycle,” while adding “Remove” and “Reimagine” to classify the ways in which we can reduce emissions while not compromising our care to patients.
Reduce
1. Minimize opened materials and single-use devices in the OR and labor and delivery
Health care is a unique setting where a culture of infection prevention and efficiency has led low-cost, single-use supplies to dominate over reusable items. While single-use items can have inexpensive purchasing costs compared to reusable items, the environmental costs required for the production and disposal of the former are often much greater. In operating rooms (ORs) and labor and delivery (LD) units, single-use items are omnipresent. Over the past decade, researchers and clinicians have started to take a closer look at these items and their carbon footprint. One group evaluated hysterectomy through a waste audit and found that the vast majority of waste from all of the cases was Spunbond Meltblown Spunbond, or SMS; plastic materialthat comprises gowns; blue wraps; and drapes; followed by hard plastic material that comprises trays and packaging.8 Moreover, production and manufacturing processes contributed to 95% of the environmental impacts of these items.8
In an effort to be time efficient, OR staff will open sterile surgical packs and individual peel-pack items prior to surgery to minimize having to find items during surgery. However, this creates an inordinate amount of waste. One group of neurosurgeons who evaluated their opened but unused supplies found that 85% of their unused items were individually opened items, leading to a waste of $2.9 million per year.9 Minor procedures like dilation and curettage, cystoscopy, and hysteroscopy do not need such a large sterile field, as these procedures are also safe to perform in the office. Hand surgeons have been quick to lead in this space, particularly with minor procedures such as carpal tunnel release. One division was able to eliminate 2.8 tons of waste and save $13,000 in a 2-year period by reducing the sterile field.10 ObGyns can work with OR and LD staff to create custom packs that minimize unused or underutilized items, helping to reduce both the carbon footprint and health care spending.
Bottom line: ObGyns can help foster a culture of having supplies available but not opened until needed during a case.
Continue to: 2. Decrease regulated medical waste...
2. Decrease regulated medical waste
Health care is unique from other fields in that there are multiple waste streams to consider. Infectious waste and items saturated in blood or capable of causing infection must be placed into regulated medical waste (RMW), or more commonly, red biohazard bags. RMW is autoclaved or incinerated prior to disposal in a landfill. This process is more financially and environmentally costly than general municipal waste (GMW). This process also requires more transport—1 study revealed that GMW traveled 20 km to a landfill for disposal, compared with the 50 km that RMW traveled for sterilized-prior-to-landfill disposal.11
Unfortunately, the vast majority of items placed in RMW are incorrectly triaged and should instead be disposed in GMW.12,13 One study performed in an emergency department revealed that 85% of waste was incorrectly placed in the RMW.12
Bottom line: ObGyns can avoid placing items in RMW that may not qualify and advocate for institution policy changes to remove RMW from places such as waiting rooms, at the patient bedside, or next to scrub sinks.
3. Reduce energy use
ORs and LD units use a lot of energy, and numerous studies have demonstrated that the heating, ventilation, and air conditioning (HVAC) system plays a large role in emissions.8,11 This can easily be fixed by “HVAC setbacks” and powering down rooms when not in use. One institution powered down ORs when not in use and reduced 234 metric tons of CO2 emissions and saved $33,000 per year.14 Transitioning to light-emitting diode (LED) lights reduced energy usage at 1 institution by almost 50%.15 Finally, computers in clinical offices, examination rooms, and administrative offices can be powered down at the end of the day. One study found that in 1 radiology department, 29 computers left on overnight and on weekends emitted 17.7 tons of CO2 emissions in 1 year.16
Bottom line: We as ObGyns can advocate for how energy can be saved outside of surgical cases, including powering down ORs and LD units, transitioning to LED lighting, and powering down workstations.
Reuse
4. Choose reusable equipment
In ObGyn practice, the most commonly used tool is the speculum. Given its omnipresence, the speculum is a great place to start to decrease our carbon footprint. Two studies have evaluated the environmental impact of reusable versus single-use disposable specula, and both demonstrated that the stainless-steel versions have less global warming potential than the acrylic varieties.17,18 Donahue and colleagues17 demonstrated that it only took 2 to 3 pelvic examinations for the cost of stainless-steel specula to break even, even when sterilized in a half-filled autoclave tray. Rodriquez, et al18 revealed that, compared with an acrylic model, the stainless-steel specula had fewer negative impacts in terms of global warming, acidification, respiratory effects, smog, and fossil fuel depletion.18
Bottom line: Strongly consider using stainless-steel specula to reduce costs and carbon emissions.
In addition to specula, ObGyns can choose reusable equipment in the OR. For example, surgeons can use stainless-steel trocars instead of disposable trocars.19 In vaginal cases, Breisky-Navratil retractors can be used instead of disposable self-retaining retractors. Plastic basins that often are included in sterile supply packs can be replaced with stainless-steel basins, which could have profound positive effects on the carbon footprint of gynecologic surgery.8 One study of ObGyns demonstrated that 95% of physicians supported waste-reduction efforts, and 66% supported utilizing reusable surgical tools instead of disposable tools.20
Bottom line: As surgeons, ObGyns have influence over what they want to use in the OR, and they can petition for reusable options over disposable options.
5. Launder the sterile blue towels
Sterile blue towels, which are made of cotton, have the largest environmental footprint compared with other disposable materials, such as plastics, and contribute greatly to toxicity in human health.8,11 Although these towels cannot be laundered and sterilized again for use in a sterile surgical field, they can be laundered and repurposed, including by environmental services to clean hospital rooms. Blue towels should be able to be laundered no matter how saturated in body fluids they are.
Bottom line: ObGyns should strive to always launder the blue towels and educate trainees and other staff in the OR to do the same.
Recycle
6. Recycle and reprocess materials and devices
While recycling is immensely important, it requires a large amount of energy to break down a material to its raw components for manufacturing. It likely reduces our carbon footprint from OR procedures by only 5%.8 However, recycling is still a good way to divert appropriate materials from landfill, saving costs and emissions at the end of a material’s life. One example is sterile blue wrap, which is a petroleum product with a recycling number of 6 and a filtration rating of N99. Blue wrap can be recycled into plastic pellets, or it can be recreated into other hospital supplies, such as gowns.
Bottom line: ObGyns can petition their hospitals to work with suppliers and waste-processing companies who have recycling programs built into their supply chains.
By contrast, reprocessing can have a much larger impact on carbon emissions. Complex items, such as advanced energy devices that can be reprocessed, result in a greater reduction in carbon emissions due to the reuse of their complex materials and manufacturing when compared with such devices that cannot be reprocessed. Recycling and reprocessing programs are already in place for several devices (TABLE). Authors of a systematic review showed that there is no evidence to support the use of single-use supplies and instruments over reprocessed items when considering instrument function, ease of use, patient safety, transmission of infection, or long-term patient outcomes.21

Bottom line: ObGyns can choose to use reprocessed items in ORs instead of single-use devices and educate staff on the safety of these items.
Continue to: Remove...
Remove
7. Remove desflurane and other volatile gases from formularies
Volatile anesthetic gases, such as desflurane, isoflurane, and nitrous oxide, are themselves potent greenhouse gases, comprising a large portion of the carbon emissions that come from the OR.22 Desflurane was developed to have a rapid onset for induction and quick recovery; however, studies have shown no clinical benefit over other gases.23 Furthermore, the costs and greenhouse gas potential are substantial. Desflurane costs 2 to 3 times more and has more than 20 times the global warming potential of the other volatile gases (FIGURE).8 Using 1 hour of desflurane is equivalent to driving 378 miles in a gas-powered vehicle, while the use of isoflurane and sevoflurane create equivalents of only 15 and 8 miles, respectively.23

Nitrous oxide is another powerful greenhouse gas that is a direct ozone depletor and can stay in the atmosphere for 114 years.22 Nitrous oxide has limited clinical use in hospitals, but it is often stored in central hospital piping. Most of the impact of nitrous oxide comes through leaks in a poor system design rather than patient delivery. One estimate reveals that more than 13 million liters of nitrous oxide are lost annually from leaks in European hospitals.22 The American Society of Anesthesiologists recommends decommissioning central piping of nitrous oxide in favor of cylinders at the point of care.24
Literature on enhanced recovery after surgery in gynecology promotes the use of propofol over volatile gases for our patients because of the high rate of postoperative nausea and vomiting seen with gases.25 Volatile gases should be a last-choice anesthetic for our patients.
Bottom line: It is critical that ObGyns work with colleagues in anesthesia to develop climate- and patient-friendly protocols for procedures.
8. Remove endocrine-disrupting chemicals from clinical supplies
Endocrine-disrupting chemicals (EDCs) are a type of chemical that alter the hormonal systems of humans, which can result in adverse health effects. Multiple studies and reviews have tied EDCs to reproductive abnormalities, such as the effects of bisphenol A (BPA) on estradiol levels, antral follicle counts, oocyte quality, and implantation rates; phthalates on fibroid burden; triclosan on embryo quality; parabens on live birth rates; and perfluoroalkylsubstances (PFAS or “forever substances”) on hypertensive disorders of pregnancy.5,26,27
What might be most shocking is that these EDCs are incorporated into medical supplies and pharmaceuticals. For example, BPA is known to line dialysis and ointment tubes, parabens are used for their antimicrobial properties in ultrasound gel and hep-locks, and phthalates are found in up to 40% of medical-use plastics and controlled-release medications. Authors of an observational study found that 74% of patients admitted to an LD unit were exposed to EDCs. In a neonatal intensive care unit (NICU), most of the supplies contained an EDC, and urinary BPA levels were elevated in neonates admitted to a NICU, raising concerns about long-term health risks.5
Bottom line: Physicians and health care institutions have an obligation to petition industry partners and suppliers to remove EDCs from their supply chains.
Reimagine
9. Educate
The field of health care sustainability remains in its infancy, but from 2007 to 2019, publications on climate change and health in academia increased by a factor of 8.29 Additionally, through waste audits, quality-improvement projects, and life cycle analyses (analytical tools to evaluate product or process emissions from materials extraction to disposal), we have gained insight into the scope of the problem, with evidence showing that our practices are largely derived from culture. It is time to provide formal education on health care sustainability to medical trainees, staff, and clinicians alike, who desire to see this topic reflected in their formal curricula.30 Start talking about it!
Bottom line: Commentaries, webinars, formal didactics sessions, in-services, and hospital workgroups to introduce this topic are a good way to teach others about the carbon footprint of our care and solutions to minimize it.
10. Engage in advocacy
Physicians have an ethical duty to advocate for change at the local, regional, and national levels if we want to see a better future for our patients, their children, and even ourselves. We should reimagine this work as an important public health initiative.31 Surveys of physicians, including ObGyns, reveal a concern about the sustainability of health care and a commitment to addressing this issue.20 ObGyns are on the frontlines of delivering care every day, so we are poised to implement changes that can impact our patients, especially when we can lead and petition hospital or local committees.20,28,32 There is much to be done, but every voice counts and can make impactful changes at every level. ●

Climate change has been called the biggest health threat of the 21st century.1 The health care sector is a huge contributor to global carbon emissions, accounting for almost double the emissions of global aviation. While other industries and countries are implementing mitigation measures to decrease their emissions, health care is currently on track to double its carbon emissions by 2050, even though it should be carbon neutral by that time to comply with the Paris Climate Agreement.2 There have been some national efforts to curb health care emissions, including the creation of the Office of Climate Change and Health Equity in 2021 and the passage of the Inflation Reduction Act in 2022.3 These are top-down, administrative approaches, and to be successful we will also need clinicians to understand and address this problem.
The negative impacts of heat, air pollution, and exposure to toxic substances on human health have been well documented in multiple regions across multiple specialties.4-7 The United States makes up 27% of the global health care carbon footprint—more emissions than the entire United Kingdom as a country—despite having only 4% of the world’s population.2 Culture and incentives for an overabundance of single-use supplies, not evidence for patient safety, have led to this uniquely American problem. It is evident that our health care industry is an excellent place to implement mitigation measures for carbon emissions that contribute to climate change and can improve health outcomes.
In this article, we recommend 10 practices that can decrease our carbon footprint in ObGyn. We focus on the classic motto of “Reduce, Reuse, Recycle,” while adding “Remove” and “Reimagine” to classify the ways in which we can reduce emissions while not compromising our care to patients.
Reduce
1. Minimize opened materials and single-use devices in the OR and labor and delivery
Health care is a unique setting where a culture of infection prevention and efficiency has led low-cost, single-use supplies to dominate over reusable items. While single-use items can have inexpensive purchasing costs compared to reusable items, the environmental costs required for the production and disposal of the former are often much greater. In operating rooms (ORs) and labor and delivery (LD) units, single-use items are omnipresent. Over the past decade, researchers and clinicians have started to take a closer look at these items and their carbon footprint. One group evaluated hysterectomy through a waste audit and found that the vast majority of waste from all of the cases was Spunbond Meltblown Spunbond, or SMS; plastic materialthat comprises gowns; blue wraps; and drapes; followed by hard plastic material that comprises trays and packaging.8 Moreover, production and manufacturing processes contributed to 95% of the environmental impacts of these items.8
In an effort to be time efficient, OR staff will open sterile surgical packs and individual peel-pack items prior to surgery to minimize having to find items during surgery. However, this creates an inordinate amount of waste. One group of neurosurgeons who evaluated their opened but unused supplies found that 85% of their unused items were individually opened items, leading to a waste of $2.9 million per year.9 Minor procedures like dilation and curettage, cystoscopy, and hysteroscopy do not need such a large sterile field, as these procedures are also safe to perform in the office. Hand surgeons have been quick to lead in this space, particularly with minor procedures such as carpal tunnel release. One division was able to eliminate 2.8 tons of waste and save $13,000 in a 2-year period by reducing the sterile field.10 ObGyns can work with OR and LD staff to create custom packs that minimize unused or underutilized items, helping to reduce both the carbon footprint and health care spending.
Bottom line: ObGyns can help foster a culture of having supplies available but not opened until needed during a case.
Continue to: 2. Decrease regulated medical waste...
2. Decrease regulated medical waste
Health care is unique from other fields in that there are multiple waste streams to consider. Infectious waste and items saturated in blood or capable of causing infection must be placed into regulated medical waste (RMW), or more commonly, red biohazard bags. RMW is autoclaved or incinerated prior to disposal in a landfill. This process is more financially and environmentally costly than general municipal waste (GMW). This process also requires more transport—1 study revealed that GMW traveled 20 km to a landfill for disposal, compared with the 50 km that RMW traveled for sterilized-prior-to-landfill disposal.11
Unfortunately, the vast majority of items placed in RMW are incorrectly triaged and should instead be disposed in GMW.12,13 One study performed in an emergency department revealed that 85% of waste was incorrectly placed in the RMW.12
Bottom line: ObGyns can avoid placing items in RMW that may not qualify and advocate for institution policy changes to remove RMW from places such as waiting rooms, at the patient bedside, or next to scrub sinks.
3. Reduce energy use
ORs and LD units use a lot of energy, and numerous studies have demonstrated that the heating, ventilation, and air conditioning (HVAC) system plays a large role in emissions.8,11 This can easily be fixed by “HVAC setbacks” and powering down rooms when not in use. One institution powered down ORs when not in use and reduced 234 metric tons of CO2 emissions and saved $33,000 per year.14 Transitioning to light-emitting diode (LED) lights reduced energy usage at 1 institution by almost 50%.15 Finally, computers in clinical offices, examination rooms, and administrative offices can be powered down at the end of the day. One study found that in 1 radiology department, 29 computers left on overnight and on weekends emitted 17.7 tons of CO2 emissions in 1 year.16
Bottom line: We as ObGyns can advocate for how energy can be saved outside of surgical cases, including powering down ORs and LD units, transitioning to LED lighting, and powering down workstations.
Reuse
4. Choose reusable equipment
In ObGyn practice, the most commonly used tool is the speculum. Given its omnipresence, the speculum is a great place to start to decrease our carbon footprint. Two studies have evaluated the environmental impact of reusable versus single-use disposable specula, and both demonstrated that the stainless-steel versions have less global warming potential than the acrylic varieties.17,18 Donahue and colleagues17 demonstrated that it only took 2 to 3 pelvic examinations for the cost of stainless-steel specula to break even, even when sterilized in a half-filled autoclave tray. Rodriquez, et al18 revealed that, compared with an acrylic model, the stainless-steel specula had fewer negative impacts in terms of global warming, acidification, respiratory effects, smog, and fossil fuel depletion.18
Bottom line: Strongly consider using stainless-steel specula to reduce costs and carbon emissions.
In addition to specula, ObGyns can choose reusable equipment in the OR. For example, surgeons can use stainless-steel trocars instead of disposable trocars.19 In vaginal cases, Breisky-Navratil retractors can be used instead of disposable self-retaining retractors. Plastic basins that often are included in sterile supply packs can be replaced with stainless-steel basins, which could have profound positive effects on the carbon footprint of gynecologic surgery.8 One study of ObGyns demonstrated that 95% of physicians supported waste-reduction efforts, and 66% supported utilizing reusable surgical tools instead of disposable tools.20
Bottom line: As surgeons, ObGyns have influence over what they want to use in the OR, and they can petition for reusable options over disposable options.
5. Launder the sterile blue towels
Sterile blue towels, which are made of cotton, have the largest environmental footprint compared with other disposable materials, such as plastics, and contribute greatly to toxicity in human health.8,11 Although these towels cannot be laundered and sterilized again for use in a sterile surgical field, they can be laundered and repurposed, including by environmental services to clean hospital rooms. Blue towels should be able to be laundered no matter how saturated in body fluids they are.
Bottom line: ObGyns should strive to always launder the blue towels and educate trainees and other staff in the OR to do the same.
Recycle
6. Recycle and reprocess materials and devices
While recycling is immensely important, it requires a large amount of energy to break down a material to its raw components for manufacturing. It likely reduces our carbon footprint from OR procedures by only 5%.8 However, recycling is still a good way to divert appropriate materials from landfill, saving costs and emissions at the end of a material’s life. One example is sterile blue wrap, which is a petroleum product with a recycling number of 6 and a filtration rating of N99. Blue wrap can be recycled into plastic pellets, or it can be recreated into other hospital supplies, such as gowns.
Bottom line: ObGyns can petition their hospitals to work with suppliers and waste-processing companies who have recycling programs built into their supply chains.
By contrast, reprocessing can have a much larger impact on carbon emissions. Complex items, such as advanced energy devices that can be reprocessed, result in a greater reduction in carbon emissions due to the reuse of their complex materials and manufacturing when compared with such devices that cannot be reprocessed. Recycling and reprocessing programs are already in place for several devices (TABLE). Authors of a systematic review showed that there is no evidence to support the use of single-use supplies and instruments over reprocessed items when considering instrument function, ease of use, patient safety, transmission of infection, or long-term patient outcomes.21

Bottom line: ObGyns can choose to use reprocessed items in ORs instead of single-use devices and educate staff on the safety of these items.
Continue to: Remove...
Remove
7. Remove desflurane and other volatile gases from formularies
Volatile anesthetic gases, such as desflurane, isoflurane, and nitrous oxide, are themselves potent greenhouse gases, comprising a large portion of the carbon emissions that come from the OR.22 Desflurane was developed to have a rapid onset for induction and quick recovery; however, studies have shown no clinical benefit over other gases.23 Furthermore, the costs and greenhouse gas potential are substantial. Desflurane costs 2 to 3 times more and has more than 20 times the global warming potential of the other volatile gases (FIGURE).8 Using 1 hour of desflurane is equivalent to driving 378 miles in a gas-powered vehicle, while the use of isoflurane and sevoflurane create equivalents of only 15 and 8 miles, respectively.23

Nitrous oxide is another powerful greenhouse gas that is a direct ozone depletor and can stay in the atmosphere for 114 years.22 Nitrous oxide has limited clinical use in hospitals, but it is often stored in central hospital piping. Most of the impact of nitrous oxide comes through leaks in a poor system design rather than patient delivery. One estimate reveals that more than 13 million liters of nitrous oxide are lost annually from leaks in European hospitals.22 The American Society of Anesthesiologists recommends decommissioning central piping of nitrous oxide in favor of cylinders at the point of care.24
Literature on enhanced recovery after surgery in gynecology promotes the use of propofol over volatile gases for our patients because of the high rate of postoperative nausea and vomiting seen with gases.25 Volatile gases should be a last-choice anesthetic for our patients.
Bottom line: It is critical that ObGyns work with colleagues in anesthesia to develop climate- and patient-friendly protocols for procedures.
8. Remove endocrine-disrupting chemicals from clinical supplies
Endocrine-disrupting chemicals (EDCs) are a type of chemical that alter the hormonal systems of humans, which can result in adverse health effects. Multiple studies and reviews have tied EDCs to reproductive abnormalities, such as the effects of bisphenol A (BPA) on estradiol levels, antral follicle counts, oocyte quality, and implantation rates; phthalates on fibroid burden; triclosan on embryo quality; parabens on live birth rates; and perfluoroalkylsubstances (PFAS or “forever substances”) on hypertensive disorders of pregnancy.5,26,27
What might be most shocking is that these EDCs are incorporated into medical supplies and pharmaceuticals. For example, BPA is known to line dialysis and ointment tubes, parabens are used for their antimicrobial properties in ultrasound gel and hep-locks, and phthalates are found in up to 40% of medical-use plastics and controlled-release medications. Authors of an observational study found that 74% of patients admitted to an LD unit were exposed to EDCs. In a neonatal intensive care unit (NICU), most of the supplies contained an EDC, and urinary BPA levels were elevated in neonates admitted to a NICU, raising concerns about long-term health risks.5
Bottom line: Physicians and health care institutions have an obligation to petition industry partners and suppliers to remove EDCs from their supply chains.
Reimagine
9. Educate
The field of health care sustainability remains in its infancy, but from 2007 to 2019, publications on climate change and health in academia increased by a factor of 8.29 Additionally, through waste audits, quality-improvement projects, and life cycle analyses (analytical tools to evaluate product or process emissions from materials extraction to disposal), we have gained insight into the scope of the problem, with evidence showing that our practices are largely derived from culture. It is time to provide formal education on health care sustainability to medical trainees, staff, and clinicians alike, who desire to see this topic reflected in their formal curricula.30 Start talking about it!
Bottom line: Commentaries, webinars, formal didactics sessions, in-services, and hospital workgroups to introduce this topic are a good way to teach others about the carbon footprint of our care and solutions to minimize it.
10. Engage in advocacy
Physicians have an ethical duty to advocate for change at the local, regional, and national levels if we want to see a better future for our patients, their children, and even ourselves. We should reimagine this work as an important public health initiative.31 Surveys of physicians, including ObGyns, reveal a concern about the sustainability of health care and a commitment to addressing this issue.20 ObGyns are on the frontlines of delivering care every day, so we are poised to implement changes that can impact our patients, especially when we can lead and petition hospital or local committees.20,28,32 There is much to be done, but every voice counts and can make impactful changes at every level. ●

- Costello A, Abbas M, Allen et al. Managing the health effects of climate change: Lancet and University College London Institute for Global Health Commission. Lancet. 2009;373:1693-1733.
- Health care climate footprint report. Health Care Without Harm website. https://www.noharm.org/ClimateFootprintReport. Accessed May 12, 2023.
- Balbus JM, McCannon CJ, Mataka A, et al. After COP26—putting health and equity at the center of the climate movement. N Engl J Med. 2022;386:1295-1297.
- Bekkar B, Pacheco S, Basu R, et al. Association of air pollution and heat exposure with preterm birth, low birth weight, and stillbirth in the US: a systematic review. JAMA Netw Open. 2020;3:e208243.
- Genco M, Anderson-Shaw L, Sargis RM. Unwitting accomplices: endocrine disruptors confounding clinical care. J Clin Endocrinol Metab. 2020;105:e3822-e3827.
- Al-Kindi SG, Sarode A, Zullo M, et al. Ambient air pollution and mortality after cardiac transplantation. J Am Coll Cardiol. 2019;74:30263035.
- Ghosh R, Gauderman WJ, Minor H, et al. Air pollution, weight loss and metabolic benefits of bariatric surgery: a potential model for study of metabolic effects of environmental exposures. Pediatr Obes. 2018;13:312-320.
- Thiel CL, Eckelman M, Guido R, et al. Environmental impacts of surgical procedures: life cycle assessment of hysterectomy in the United States. Environ Sci Technol. 2015;49:1779-1786.
- Zygourakis CC, Yoon S, Valencia V, et al. Operating room waste: disposable supply utilization in neurosurgical procedures. J Neurosurg. 2017;126:620-625.
- van Demark RE, Smith VJS, Fiegen A. Lean and green hand surgery. J Hand Surg. 2018;43:179-181.
- Campion N, Thiel CL, DeBlois J, et al. Life cycle assessment perspectives on delivering an infant in the US. Sci Total Environ. 2012;425:191198.
- Hsu S, Thiel CL, Mello MJ, Slutzman JE. Dumpster diving in the emergency department. West J Emerg Med. 2020;21:1211-1217.
- Mcgain F, Story D, Hendel S. An audit of intensive care unit recyclable waste. Anaesthesia. 2009;64:1299-1302.
- Wormer BA, Augenstein VA, Carpenter CL, et al. The green operating room: simple changes to reduce cost and our carbon footprint. Am Surg. 2013;79:666-671.
- Kagoma Y, Stall N, Rubinstein E, et al. People, planet and profits: the case for greening operating rooms. Can Med Assoc J. 2012;184:19051911.
- McCarthy CJ, Gerstenmaier JF, O’ Neill AC, et al. “EcoRadiology”— pulling the plug on wasted energy in the radiology department. Acad Radiol. 2014;21:1563-1566.
- Donahue LM, Hilton S, Bell SG, et al. A comparative carbon footprint analysis of disposable and reusable vaginal specula. Am J Obstet Gynecol. 2020;223:225.e1-225.e7.
- Rodriguez Morris MI, Hicks A. Life cycle assessment of stainless-steel reusable speculums versus disposable acrylic speculums in a university clinic setting: a case study. Environ Res Commun. 2022;4:025002.
- MacNeill AJ, Lillywhite R, Brown CJ. The impact of surgery on global climate: a carbon footprinting study of operating theatres in three health systems. Lancet Planet Health. 2017;1:e381-e388.
- Thiel C, Duncan P, Woods N. Attitude of US obstetricians and gynaecologists to global warming and medical waste. J Health Serv Res Policy. 2017;22:162-167.
- Siu J, Hill AG, MacCormick AD. Systematic review of reusable versus disposable laparoscopic instruments: costs and safety. ANZ J Surg. 2017;87:28-33.
- Ryan SM, Nielsen CJ. Global warming potential of inhaled anesthetics: application to clinical use. Anesth Analg. 2010;111:92-98.
- Meyer MJ. Desflurane should des-appear: global and financial rationale. Anesth Analg. 2020;131:1317-1322.
- Rollins MD, Arendt KW, Carvalho B, et al. ASA Committee on Obstetric Anesthesia Working Group. Nitrous oxide. American Society of Anesthesiologists website. Accessed May 12, 2023. https://www .asahq.org/about-asa/governance-and-committees/asa-committees /committee-on-obstetric-anesthesia/nitrous-oxide.
- Kalogera E, Dowdy SC. Enhanced recovery pathway in gynecologic surgery: improving outcomes through evidence-based medicine. Obstet Gynecol Clin North Am. 2016;43:551-573.
- Zota AR, Geller RJ, Calafat AM, et al. Phthalates exposure and uterine fibroid burden among women undergoing surgical treatment for fibroids: a preliminary study. Fertil Steril. 2019;111:112-121.
- Bommartio PA, Ferguson KK, Meeker JD, et al. Maternal levels of perfluoroalkyl substances (PFAS) during early pregnancy in relation to preeclampsia subtypes and biomarkers of preeclampsia risk. Environ Health Perspect. 2021;129:107004.
- Azouz S, Boyll P, Swanson M, et al. Managing barriers to recycling in the operating room. Am J Surg. 2019;217:634-638.
- Watts N, Amann M, Arnell N, et al. The 2020 report of The Lancet Countdown on health and climate change: responding to converging crises. Lancet. 2021;397:129-170.
- Ryan EC, Dubrow R, Sherman JD. Medical, nursing, and physician assistant student knowledge and attitudes toward climate change, pollution, and resource conservation in health care. BMC Med Educ. 2020;20:200.
- Giudice LC, Llamas-Clark EF, DeNicola Net al; FIGO Committee on Climate Change and Toxic Environmental Exposures. Climate change, women’s health, and the role of obstetricians and gynecologists in leadership. Int J Gynaecol Obstet. 2021;155:345-356.
- Yates EF, Bowder AN, Roa L, et al. Empowering surgeons, anesthesiologists, and obstetricians to incorporate environmental sustainability in the operating room. Ann Surg. 2021;273:1108-1114.
- Costello A, Abbas M, Allen et al. Managing the health effects of climate change: Lancet and University College London Institute for Global Health Commission. Lancet. 2009;373:1693-1733.
- Health care climate footprint report. Health Care Without Harm website. https://www.noharm.org/ClimateFootprintReport. Accessed May 12, 2023.
- Balbus JM, McCannon CJ, Mataka A, et al. After COP26—putting health and equity at the center of the climate movement. N Engl J Med. 2022;386:1295-1297.
- Bekkar B, Pacheco S, Basu R, et al. Association of air pollution and heat exposure with preterm birth, low birth weight, and stillbirth in the US: a systematic review. JAMA Netw Open. 2020;3:e208243.
- Genco M, Anderson-Shaw L, Sargis RM. Unwitting accomplices: endocrine disruptors confounding clinical care. J Clin Endocrinol Metab. 2020;105:e3822-e3827.
- Al-Kindi SG, Sarode A, Zullo M, et al. Ambient air pollution and mortality after cardiac transplantation. J Am Coll Cardiol. 2019;74:30263035.
- Ghosh R, Gauderman WJ, Minor H, et al. Air pollution, weight loss and metabolic benefits of bariatric surgery: a potential model for study of metabolic effects of environmental exposures. Pediatr Obes. 2018;13:312-320.
- Thiel CL, Eckelman M, Guido R, et al. Environmental impacts of surgical procedures: life cycle assessment of hysterectomy in the United States. Environ Sci Technol. 2015;49:1779-1786.
- Zygourakis CC, Yoon S, Valencia V, et al. Operating room waste: disposable supply utilization in neurosurgical procedures. J Neurosurg. 2017;126:620-625.
- van Demark RE, Smith VJS, Fiegen A. Lean and green hand surgery. J Hand Surg. 2018;43:179-181.
- Campion N, Thiel CL, DeBlois J, et al. Life cycle assessment perspectives on delivering an infant in the US. Sci Total Environ. 2012;425:191198.
- Hsu S, Thiel CL, Mello MJ, Slutzman JE. Dumpster diving in the emergency department. West J Emerg Med. 2020;21:1211-1217.
- Mcgain F, Story D, Hendel S. An audit of intensive care unit recyclable waste. Anaesthesia. 2009;64:1299-1302.
- Wormer BA, Augenstein VA, Carpenter CL, et al. The green operating room: simple changes to reduce cost and our carbon footprint. Am Surg. 2013;79:666-671.
- Kagoma Y, Stall N, Rubinstein E, et al. People, planet and profits: the case for greening operating rooms. Can Med Assoc J. 2012;184:19051911.
- McCarthy CJ, Gerstenmaier JF, O’ Neill AC, et al. “EcoRadiology”— pulling the plug on wasted energy in the radiology department. Acad Radiol. 2014;21:1563-1566.
- Donahue LM, Hilton S, Bell SG, et al. A comparative carbon footprint analysis of disposable and reusable vaginal specula. Am J Obstet Gynecol. 2020;223:225.e1-225.e7.
- Rodriguez Morris MI, Hicks A. Life cycle assessment of stainless-steel reusable speculums versus disposable acrylic speculums in a university clinic setting: a case study. Environ Res Commun. 2022;4:025002.
- MacNeill AJ, Lillywhite R, Brown CJ. The impact of surgery on global climate: a carbon footprinting study of operating theatres in three health systems. Lancet Planet Health. 2017;1:e381-e388.
- Thiel C, Duncan P, Woods N. Attitude of US obstetricians and gynaecologists to global warming and medical waste. J Health Serv Res Policy. 2017;22:162-167.
- Siu J, Hill AG, MacCormick AD. Systematic review of reusable versus disposable laparoscopic instruments: costs and safety. ANZ J Surg. 2017;87:28-33.
- Ryan SM, Nielsen CJ. Global warming potential of inhaled anesthetics: application to clinical use. Anesth Analg. 2010;111:92-98.
- Meyer MJ. Desflurane should des-appear: global and financial rationale. Anesth Analg. 2020;131:1317-1322.
- Rollins MD, Arendt KW, Carvalho B, et al. ASA Committee on Obstetric Anesthesia Working Group. Nitrous oxide. American Society of Anesthesiologists website. Accessed May 12, 2023. https://www .asahq.org/about-asa/governance-and-committees/asa-committees /committee-on-obstetric-anesthesia/nitrous-oxide.
- Kalogera E, Dowdy SC. Enhanced recovery pathway in gynecologic surgery: improving outcomes through evidence-based medicine. Obstet Gynecol Clin North Am. 2016;43:551-573.
- Zota AR, Geller RJ, Calafat AM, et al. Phthalates exposure and uterine fibroid burden among women undergoing surgical treatment for fibroids: a preliminary study. Fertil Steril. 2019;111:112-121.
- Bommartio PA, Ferguson KK, Meeker JD, et al. Maternal levels of perfluoroalkyl substances (PFAS) during early pregnancy in relation to preeclampsia subtypes and biomarkers of preeclampsia risk. Environ Health Perspect. 2021;129:107004.
- Azouz S, Boyll P, Swanson M, et al. Managing barriers to recycling in the operating room. Am J Surg. 2019;217:634-638.
- Watts N, Amann M, Arnell N, et al. The 2020 report of The Lancet Countdown on health and climate change: responding to converging crises. Lancet. 2021;397:129-170.
- Ryan EC, Dubrow R, Sherman JD. Medical, nursing, and physician assistant student knowledge and attitudes toward climate change, pollution, and resource conservation in health care. BMC Med Educ. 2020;20:200.
- Giudice LC, Llamas-Clark EF, DeNicola Net al; FIGO Committee on Climate Change and Toxic Environmental Exposures. Climate change, women’s health, and the role of obstetricians and gynecologists in leadership. Int J Gynaecol Obstet. 2021;155:345-356.
- Yates EF, Bowder AN, Roa L, et al. Empowering surgeons, anesthesiologists, and obstetricians to incorporate environmental sustainability in the operating room. Ann Surg. 2021;273:1108-1114.
ACS officer provides ASCO highlights: Targeting hidden cancer, AI in oncology
And it didn’t just sparkle because of the sequined Taylor Swift fans clogging the nearby streets during the meeting.
Arif Kamal, MD, MBA, MHS, who is also an oncologist at Duke University, Durham, N.C., said he was impressed by a pair of landmark studies released at the meeting that show hidden cancer can be targeted with “really remarkable outcomes.” He also highlighted sessions that examined the role of artificial intelligence (AI) in oncology, during an interview.
Below are lightly edited excerpts from a conversation with Dr. Kamal:
Question: What are some of most groundbreaking studies released at ASCO?
Answer: One is an interim analysis of the NATALEE trial, which involved patients with early-stage hormone receptor-positive, HER2-negative (HR+/HER2–) breast tumors. This phase 3 randomized trial compared maintenance therapy with the cyclin-dependent kinase 4/6 (CDK4/6) inhibitor ribociclib (Kisqali) plus endocrine therapy with an aromatase inhibitor to endocrine therapy alone in patients with node-positive or node-negative and stage II or III HR+/HER– breast cancer.
For a long time, the standard care in these patients has been to use endocrine therapy alone. This is the first big trial to show that upstream usage of additional therapy in early stages is also beneficial for disease-free survival. The 3-year invasive disease-free survival rate was 90.4% in the rebociclib-endocrine therapy group vs. 87.1% for patients who received only endocrine therapy (P = .0014).
Q: How do these findings add to current knowledge?
A: Typically, we let people get metastatic disease before we use CDK4/6 inhibitors. These findings show that systemic treatment beyond endocrine therapy will be helpful in cases where you’ve got smaller disease that has not spread yet.
Even in patients with node-negative breast cancer, micrometastatic disease is clearly there, because the medication killed the negative lymph nodes.
Q: What else struck you as especially important research?
A: The NATALEE findings match what we saw in another study – the ADAURA trial, which looked at adjuvant osimertinib in non–small-cell lung cancer patients with EGFR-mutated, stage IB to IIIA disease – cancer that has not spread to the lymph nodes.
This is another example where you have a treatment being used in earlier-stage disease that’s showing really remarkable outcomes. The study found that 5-year overall survival was 88% in an osimertinib group vs. 78% in a placebo group (P < .001). This is a disease where, in stage IB, we wouldn’t even necessarily give these patients treatment at all, other than surgical resection of the tumor and maybe give them a little bit of chemotherapy.
Even in these smaller, early tumors, osimertinib makes a difference.
Q: As a whole, what are these studies telling us about cancer cells that can’t be easily detected?
A: To find a disease-free survival benefit with adding ribociclib in a stage II, stage III setting, particularly in node-negative disease, is remarkable because it says that the cells in hiding are bad actors, and they are going to cause trouble. The study shows that medications can find these cells and reverse that risk of bad outcomes.
If you think about the paradigm of cancer, that’s pretty remarkable because the ADAURA trial does the same thing: You do surgery for [early-stage] lung cancers that have not spread to the lymph nodes and you figure, “Well, I’ve got it all, right? The margins are real big, healthy, clean.” And yet, people still have recurrences, and you ask the same question: “Can any medicine find those few cells, the hundreds of cells that are still left somewhere in hiding?” And the answer is again, yes. It’s changing the paradigm of our understanding of minimal residual disease.
That’s why there’s so much interest in liquid biopsies. Let’s say that after treatment we don’t see any cancer radiologically, but there’s a signal from a liquid biopsy [detecting residual cancer]. These two trials demonstrate that there’s something we can do about it.
Q: There were quite a few studies about artificial intelligence released at ASCO. Where do we stand on that front?
A: We’re just at the beginning of people thinking about the use of generative AI for clinical decision support, clinical trial matching, and pathology review. But AI, at least for now, still has the issue of making up things that aren’t true. That’s not something patients are going to be okay with.
Q: How can AI be helpful to medical providers considering its limitations?
A: AI is going to be very good at the data-to-information transition. You’ll start seeing people use AI to start clinical notes for them and to match patients to the best clinical trials for them. But fundamentally, the clinician’s role will continue to be to check facts and offer wisdom.
Q: Will AI threaten the careers of oncologists?
A: The body of knowledge about oncology is growing exponentially, and no one can actually keep up. There’s so much data that’s out there that needs to be turned into usable information amid a shortage of oncologists. At the same time, the prevalence of cancer is going up, even though mortality is going down.
Synthesis of data is what oncologists are waiting for from AI. They’ll welcome it as opposed to being worried. That’s the sentiment I heard from my colleagues.
Dr. Kamal has no disclosures.
And it didn’t just sparkle because of the sequined Taylor Swift fans clogging the nearby streets during the meeting.
Arif Kamal, MD, MBA, MHS, who is also an oncologist at Duke University, Durham, N.C., said he was impressed by a pair of landmark studies released at the meeting that show hidden cancer can be targeted with “really remarkable outcomes.” He also highlighted sessions that examined the role of artificial intelligence (AI) in oncology, during an interview.
Below are lightly edited excerpts from a conversation with Dr. Kamal:
Question: What are some of most groundbreaking studies released at ASCO?
Answer: One is an interim analysis of the NATALEE trial, which involved patients with early-stage hormone receptor-positive, HER2-negative (HR+/HER2–) breast tumors. This phase 3 randomized trial compared maintenance therapy with the cyclin-dependent kinase 4/6 (CDK4/6) inhibitor ribociclib (Kisqali) plus endocrine therapy with an aromatase inhibitor to endocrine therapy alone in patients with node-positive or node-negative and stage II or III HR+/HER– breast cancer.
For a long time, the standard care in these patients has been to use endocrine therapy alone. This is the first big trial to show that upstream usage of additional therapy in early stages is also beneficial for disease-free survival. The 3-year invasive disease-free survival rate was 90.4% in the rebociclib-endocrine therapy group vs. 87.1% for patients who received only endocrine therapy (P = .0014).
Q: How do these findings add to current knowledge?
A: Typically, we let people get metastatic disease before we use CDK4/6 inhibitors. These findings show that systemic treatment beyond endocrine therapy will be helpful in cases where you’ve got smaller disease that has not spread yet.
Even in patients with node-negative breast cancer, micrometastatic disease is clearly there, because the medication killed the negative lymph nodes.
Q: What else struck you as especially important research?
A: The NATALEE findings match what we saw in another study – the ADAURA trial, which looked at adjuvant osimertinib in non–small-cell lung cancer patients with EGFR-mutated, stage IB to IIIA disease – cancer that has not spread to the lymph nodes.
This is another example where you have a treatment being used in earlier-stage disease that’s showing really remarkable outcomes. The study found that 5-year overall survival was 88% in an osimertinib group vs. 78% in a placebo group (P < .001). This is a disease where, in stage IB, we wouldn’t even necessarily give these patients treatment at all, other than surgical resection of the tumor and maybe give them a little bit of chemotherapy.
Even in these smaller, early tumors, osimertinib makes a difference.
Q: As a whole, what are these studies telling us about cancer cells that can’t be easily detected?
A: To find a disease-free survival benefit with adding ribociclib in a stage II, stage III setting, particularly in node-negative disease, is remarkable because it says that the cells in hiding are bad actors, and they are going to cause trouble. The study shows that medications can find these cells and reverse that risk of bad outcomes.
If you think about the paradigm of cancer, that’s pretty remarkable because the ADAURA trial does the same thing: You do surgery for [early-stage] lung cancers that have not spread to the lymph nodes and you figure, “Well, I’ve got it all, right? The margins are real big, healthy, clean.” And yet, people still have recurrences, and you ask the same question: “Can any medicine find those few cells, the hundreds of cells that are still left somewhere in hiding?” And the answer is again, yes. It’s changing the paradigm of our understanding of minimal residual disease.
That’s why there’s so much interest in liquid biopsies. Let’s say that after treatment we don’t see any cancer radiologically, but there’s a signal from a liquid biopsy [detecting residual cancer]. These two trials demonstrate that there’s something we can do about it.
Q: There were quite a few studies about artificial intelligence released at ASCO. Where do we stand on that front?
A: We’re just at the beginning of people thinking about the use of generative AI for clinical decision support, clinical trial matching, and pathology review. But AI, at least for now, still has the issue of making up things that aren’t true. That’s not something patients are going to be okay with.
Q: How can AI be helpful to medical providers considering its limitations?
A: AI is going to be very good at the data-to-information transition. You’ll start seeing people use AI to start clinical notes for them and to match patients to the best clinical trials for them. But fundamentally, the clinician’s role will continue to be to check facts and offer wisdom.
Q: Will AI threaten the careers of oncologists?
A: The body of knowledge about oncology is growing exponentially, and no one can actually keep up. There’s so much data that’s out there that needs to be turned into usable information amid a shortage of oncologists. At the same time, the prevalence of cancer is going up, even though mortality is going down.
Synthesis of data is what oncologists are waiting for from AI. They’ll welcome it as opposed to being worried. That’s the sentiment I heard from my colleagues.
Dr. Kamal has no disclosures.
And it didn’t just sparkle because of the sequined Taylor Swift fans clogging the nearby streets during the meeting.
Arif Kamal, MD, MBA, MHS, who is also an oncologist at Duke University, Durham, N.C., said he was impressed by a pair of landmark studies released at the meeting that show hidden cancer can be targeted with “really remarkable outcomes.” He also highlighted sessions that examined the role of artificial intelligence (AI) in oncology, during an interview.
Below are lightly edited excerpts from a conversation with Dr. Kamal:
Question: What are some of most groundbreaking studies released at ASCO?
Answer: One is an interim analysis of the NATALEE trial, which involved patients with early-stage hormone receptor-positive, HER2-negative (HR+/HER2–) breast tumors. This phase 3 randomized trial compared maintenance therapy with the cyclin-dependent kinase 4/6 (CDK4/6) inhibitor ribociclib (Kisqali) plus endocrine therapy with an aromatase inhibitor to endocrine therapy alone in patients with node-positive or node-negative and stage II or III HR+/HER– breast cancer.
For a long time, the standard care in these patients has been to use endocrine therapy alone. This is the first big trial to show that upstream usage of additional therapy in early stages is also beneficial for disease-free survival. The 3-year invasive disease-free survival rate was 90.4% in the rebociclib-endocrine therapy group vs. 87.1% for patients who received only endocrine therapy (P = .0014).
Q: How do these findings add to current knowledge?
A: Typically, we let people get metastatic disease before we use CDK4/6 inhibitors. These findings show that systemic treatment beyond endocrine therapy will be helpful in cases where you’ve got smaller disease that has not spread yet.
Even in patients with node-negative breast cancer, micrometastatic disease is clearly there, because the medication killed the negative lymph nodes.
Q: What else struck you as especially important research?
A: The NATALEE findings match what we saw in another study – the ADAURA trial, which looked at adjuvant osimertinib in non–small-cell lung cancer patients with EGFR-mutated, stage IB to IIIA disease – cancer that has not spread to the lymph nodes.
This is another example where you have a treatment being used in earlier-stage disease that’s showing really remarkable outcomes. The study found that 5-year overall survival was 88% in an osimertinib group vs. 78% in a placebo group (P < .001). This is a disease where, in stage IB, we wouldn’t even necessarily give these patients treatment at all, other than surgical resection of the tumor and maybe give them a little bit of chemotherapy.
Even in these smaller, early tumors, osimertinib makes a difference.
Q: As a whole, what are these studies telling us about cancer cells that can’t be easily detected?
A: To find a disease-free survival benefit with adding ribociclib in a stage II, stage III setting, particularly in node-negative disease, is remarkable because it says that the cells in hiding are bad actors, and they are going to cause trouble. The study shows that medications can find these cells and reverse that risk of bad outcomes.
If you think about the paradigm of cancer, that’s pretty remarkable because the ADAURA trial does the same thing: You do surgery for [early-stage] lung cancers that have not spread to the lymph nodes and you figure, “Well, I’ve got it all, right? The margins are real big, healthy, clean.” And yet, people still have recurrences, and you ask the same question: “Can any medicine find those few cells, the hundreds of cells that are still left somewhere in hiding?” And the answer is again, yes. It’s changing the paradigm of our understanding of minimal residual disease.
That’s why there’s so much interest in liquid biopsies. Let’s say that after treatment we don’t see any cancer radiologically, but there’s a signal from a liquid biopsy [detecting residual cancer]. These two trials demonstrate that there’s something we can do about it.
Q: There were quite a few studies about artificial intelligence released at ASCO. Where do we stand on that front?
A: We’re just at the beginning of people thinking about the use of generative AI for clinical decision support, clinical trial matching, and pathology review. But AI, at least for now, still has the issue of making up things that aren’t true. That’s not something patients are going to be okay with.
Q: How can AI be helpful to medical providers considering its limitations?
A: AI is going to be very good at the data-to-information transition. You’ll start seeing people use AI to start clinical notes for them and to match patients to the best clinical trials for them. But fundamentally, the clinician’s role will continue to be to check facts and offer wisdom.
Q: Will AI threaten the careers of oncologists?
A: The body of knowledge about oncology is growing exponentially, and no one can actually keep up. There’s so much data that’s out there that needs to be turned into usable information amid a shortage of oncologists. At the same time, the prevalence of cancer is going up, even though mortality is going down.
Synthesis of data is what oncologists are waiting for from AI. They’ll welcome it as opposed to being worried. That’s the sentiment I heard from my colleagues.
Dr. Kamal has no disclosures.
AT ASCO 2023
‘New standard of care’ for capecitabine hand-foot syndrome
researchers reported in a study that has been hailed by experts as “practice changing.”
Hand-foot syndrome causes painful, bleeding blisters and ulcers on the palms and soles. It often leads to dose reductions and sometimes even discontinuations, both of which limit the effectiveness of capecitabine, a standard oral chemotherapy drug widely used for colorectal and breast cancers.
In a new study presented at the annual meeting of the American Society of Clinical Oncology, Indian researchers reported that a cheap, safe, and widely available over-the-counter nonsteroidal anti-inflammatory gel containing 1% diclofenac reduced the incidence of hand-foot syndrome by 75% among patients with cancer being treated with capecitabine.
Up until now, the oral anti-inflammatory celecoxib (Celebrex) was the only agent proven to prevent the problem, but it’s rarely used because of the risk for strokes, gastric bleeding, and other issues, none of which are a concern with topical diclofenac, which osteoarthritis patients have used safely for years.
The Indian trial, dubbed D-Torch, establishes “1% topical diclofenac gel as the new standard of care to prevent capecitabine-associated hand-foot syndrome,” said investigator and study presenter Atul Batra, MD, a medical oncologist at the All India Institute of Medical Sciences, New Delhi.
Dr. Batra told ASCO Daily News that there is no need for a second trial. “We don’t feel there’s a need to replicate these results” in a larger study “because this was adequately powered, and the results speak for themselves. There’s no confusion about these results. Diclofenac is clearly effective.”
Dr. Batra also commented that his clinic now uses topical diclofenac routinely during capecitabine treatment and that he hopes oncology practices elsewhere will do the same.
Diclofenac gel is sold under the brand name Voltaren and is also available as a generic; in the United States, a 150-gram tube costs about $18 at Walmart.
‘The most practice-changing study’ at ASCO 2023
Audience members at ASCO’s annual meeting immediately saw the importance of the study.
Tarah Ballinger, MD, a breast cancer specialist at Indiana University, Indianapolis, said on Twitter that “this might be the most practice changing study I heard at ASCO23.” Topical diclofenac is “widely available, affordable, [and] addresses [a] major” quality of life issue.
The study discussant at the meeting, gastrointestinal cancer specialist Pallavi Kumar, MD, of the University of Pennsylvania, Philadelphia, concurred: “For me as a GI oncologist, topical diclofenac for prevention of HFS for patients on capecitabine is practice changing,” she said.
The takeaway is “that topical diclofenac significantly reduces the incidence of grade 2 or higher HFS in patients receiving capecitabine.” The results are “very impressive,” Dr. Kumar said.
Study details
The idea for the new study came after Batra and colleagues realized that celecoxib, a COX-2 enzyme inhibitor, helps prevent capecitabine hand-foot syndrome (HFS) by blocking a key process that leads to it, the up-regulation of COX-2 and subsequent release of proinflammatory prostaglandins.
They turned to diclofenac gel hoping to get the same effect but more safely; diclofenac is also a COX-2 blocker, and its topical formulation has a strong safety record.
To test the approach, the team randomly assigned 130 patients to topical diclofenac and 133 to placebo – the gel vehicle without the medication – while they were being treated with capecitabine for 12 weeks; 56% were being treated for breast cancer and the rest for gastrointestinal cancers.
Subjects rubbed one fingertip’s worth of gel – about half a gram – on each palm and the back of each hand twice a day. The dose was about 4 grams/day, which is well below maximal dosages for osteoarthritis (up to 32 g/day over all affected joints). Adherence to treatment was about 95% in both arms.
By the end of 12 weeks, the incidence of grade 2 or higher HFS was 3.8% in the diclofenac arm (5 patients) versus 15% (n = 20) with placebo (P = .003), a 75% risk reduction.
The incidence of any grade HFS was 6.1% in the treatment group versus 18.1% with placebo (P = .003).
Hand-foot syndrome led to dose reductions of capecitabine in 13.5% of placebo but only 3.8% of those in the diclofenac group (P = .002).
The findings held regardless of whether patients were being treated for breast or GI cancer or if they were men or women.
Other capecitabine-induced adverse events, including diarrhea, mucositis, and myelosuppression, were not significantly different between the groups.
The treatment arms were well balanced, with a median age of 47 years in both groups and women making up about 70% of each. About 40% of subjects in each group were on capecitabine monotherapy with the rest on combination treatments. The mean dose of capecitabine was just over 1,880 mg/m2 in both groups.
At the meeting, Dr. Batra was asked if topical diclofenac would also work for another common problem in oncology: hand-food syndrome occurring as a side-effect with VEGF–tyrosine kinase inhibitors. He didn’t think so because it probably has a different cause than capecitabine HFS, one not strongly related to COX-2 up-regulation.
The study was partly funded by the Indian Supportive Care of Cancer Association. The investigators reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
researchers reported in a study that has been hailed by experts as “practice changing.”
Hand-foot syndrome causes painful, bleeding blisters and ulcers on the palms and soles. It often leads to dose reductions and sometimes even discontinuations, both of which limit the effectiveness of capecitabine, a standard oral chemotherapy drug widely used for colorectal and breast cancers.
In a new study presented at the annual meeting of the American Society of Clinical Oncology, Indian researchers reported that a cheap, safe, and widely available over-the-counter nonsteroidal anti-inflammatory gel containing 1% diclofenac reduced the incidence of hand-foot syndrome by 75% among patients with cancer being treated with capecitabine.
Up until now, the oral anti-inflammatory celecoxib (Celebrex) was the only agent proven to prevent the problem, but it’s rarely used because of the risk for strokes, gastric bleeding, and other issues, none of which are a concern with topical diclofenac, which osteoarthritis patients have used safely for years.
The Indian trial, dubbed D-Torch, establishes “1% topical diclofenac gel as the new standard of care to prevent capecitabine-associated hand-foot syndrome,” said investigator and study presenter Atul Batra, MD, a medical oncologist at the All India Institute of Medical Sciences, New Delhi.
Dr. Batra told ASCO Daily News that there is no need for a second trial. “We don’t feel there’s a need to replicate these results” in a larger study “because this was adequately powered, and the results speak for themselves. There’s no confusion about these results. Diclofenac is clearly effective.”
Dr. Batra also commented that his clinic now uses topical diclofenac routinely during capecitabine treatment and that he hopes oncology practices elsewhere will do the same.
Diclofenac gel is sold under the brand name Voltaren and is also available as a generic; in the United States, a 150-gram tube costs about $18 at Walmart.
‘The most practice-changing study’ at ASCO 2023
Audience members at ASCO’s annual meeting immediately saw the importance of the study.
Tarah Ballinger, MD, a breast cancer specialist at Indiana University, Indianapolis, said on Twitter that “this might be the most practice changing study I heard at ASCO23.” Topical diclofenac is “widely available, affordable, [and] addresses [a] major” quality of life issue.
The study discussant at the meeting, gastrointestinal cancer specialist Pallavi Kumar, MD, of the University of Pennsylvania, Philadelphia, concurred: “For me as a GI oncologist, topical diclofenac for prevention of HFS for patients on capecitabine is practice changing,” she said.
The takeaway is “that topical diclofenac significantly reduces the incidence of grade 2 or higher HFS in patients receiving capecitabine.” The results are “very impressive,” Dr. Kumar said.
Study details
The idea for the new study came after Batra and colleagues realized that celecoxib, a COX-2 enzyme inhibitor, helps prevent capecitabine hand-foot syndrome (HFS) by blocking a key process that leads to it, the up-regulation of COX-2 and subsequent release of proinflammatory prostaglandins.
They turned to diclofenac gel hoping to get the same effect but more safely; diclofenac is also a COX-2 blocker, and its topical formulation has a strong safety record.
To test the approach, the team randomly assigned 130 patients to topical diclofenac and 133 to placebo – the gel vehicle without the medication – while they were being treated with capecitabine for 12 weeks; 56% were being treated for breast cancer and the rest for gastrointestinal cancers.
Subjects rubbed one fingertip’s worth of gel – about half a gram – on each palm and the back of each hand twice a day. The dose was about 4 grams/day, which is well below maximal dosages for osteoarthritis (up to 32 g/day over all affected joints). Adherence to treatment was about 95% in both arms.
By the end of 12 weeks, the incidence of grade 2 or higher HFS was 3.8% in the diclofenac arm (5 patients) versus 15% (n = 20) with placebo (P = .003), a 75% risk reduction.
The incidence of any grade HFS was 6.1% in the treatment group versus 18.1% with placebo (P = .003).
Hand-foot syndrome led to dose reductions of capecitabine in 13.5% of placebo but only 3.8% of those in the diclofenac group (P = .002).
The findings held regardless of whether patients were being treated for breast or GI cancer or if they were men or women.
Other capecitabine-induced adverse events, including diarrhea, mucositis, and myelosuppression, were not significantly different between the groups.
The treatment arms were well balanced, with a median age of 47 years in both groups and women making up about 70% of each. About 40% of subjects in each group were on capecitabine monotherapy with the rest on combination treatments. The mean dose of capecitabine was just over 1,880 mg/m2 in both groups.
At the meeting, Dr. Batra was asked if topical diclofenac would also work for another common problem in oncology: hand-food syndrome occurring as a side-effect with VEGF–tyrosine kinase inhibitors. He didn’t think so because it probably has a different cause than capecitabine HFS, one not strongly related to COX-2 up-regulation.
The study was partly funded by the Indian Supportive Care of Cancer Association. The investigators reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
researchers reported in a study that has been hailed by experts as “practice changing.”
Hand-foot syndrome causes painful, bleeding blisters and ulcers on the palms and soles. It often leads to dose reductions and sometimes even discontinuations, both of which limit the effectiveness of capecitabine, a standard oral chemotherapy drug widely used for colorectal and breast cancers.
In a new study presented at the annual meeting of the American Society of Clinical Oncology, Indian researchers reported that a cheap, safe, and widely available over-the-counter nonsteroidal anti-inflammatory gel containing 1% diclofenac reduced the incidence of hand-foot syndrome by 75% among patients with cancer being treated with capecitabine.
Up until now, the oral anti-inflammatory celecoxib (Celebrex) was the only agent proven to prevent the problem, but it’s rarely used because of the risk for strokes, gastric bleeding, and other issues, none of which are a concern with topical diclofenac, which osteoarthritis patients have used safely for years.
The Indian trial, dubbed D-Torch, establishes “1% topical diclofenac gel as the new standard of care to prevent capecitabine-associated hand-foot syndrome,” said investigator and study presenter Atul Batra, MD, a medical oncologist at the All India Institute of Medical Sciences, New Delhi.
Dr. Batra told ASCO Daily News that there is no need for a second trial. “We don’t feel there’s a need to replicate these results” in a larger study “because this was adequately powered, and the results speak for themselves. There’s no confusion about these results. Diclofenac is clearly effective.”
Dr. Batra also commented that his clinic now uses topical diclofenac routinely during capecitabine treatment and that he hopes oncology practices elsewhere will do the same.
Diclofenac gel is sold under the brand name Voltaren and is also available as a generic; in the United States, a 150-gram tube costs about $18 at Walmart.
‘The most practice-changing study’ at ASCO 2023
Audience members at ASCO’s annual meeting immediately saw the importance of the study.
Tarah Ballinger, MD, a breast cancer specialist at Indiana University, Indianapolis, said on Twitter that “this might be the most practice changing study I heard at ASCO23.” Topical diclofenac is “widely available, affordable, [and] addresses [a] major” quality of life issue.
The study discussant at the meeting, gastrointestinal cancer specialist Pallavi Kumar, MD, of the University of Pennsylvania, Philadelphia, concurred: “For me as a GI oncologist, topical diclofenac for prevention of HFS for patients on capecitabine is practice changing,” she said.
The takeaway is “that topical diclofenac significantly reduces the incidence of grade 2 or higher HFS in patients receiving capecitabine.” The results are “very impressive,” Dr. Kumar said.
Study details
The idea for the new study came after Batra and colleagues realized that celecoxib, a COX-2 enzyme inhibitor, helps prevent capecitabine hand-foot syndrome (HFS) by blocking a key process that leads to it, the up-regulation of COX-2 and subsequent release of proinflammatory prostaglandins.
They turned to diclofenac gel hoping to get the same effect but more safely; diclofenac is also a COX-2 blocker, and its topical formulation has a strong safety record.
To test the approach, the team randomly assigned 130 patients to topical diclofenac and 133 to placebo – the gel vehicle without the medication – while they were being treated with capecitabine for 12 weeks; 56% were being treated for breast cancer and the rest for gastrointestinal cancers.
Subjects rubbed one fingertip’s worth of gel – about half a gram – on each palm and the back of each hand twice a day. The dose was about 4 grams/day, which is well below maximal dosages for osteoarthritis (up to 32 g/day over all affected joints). Adherence to treatment was about 95% in both arms.
By the end of 12 weeks, the incidence of grade 2 or higher HFS was 3.8% in the diclofenac arm (5 patients) versus 15% (n = 20) with placebo (P = .003), a 75% risk reduction.
The incidence of any grade HFS was 6.1% in the treatment group versus 18.1% with placebo (P = .003).
Hand-foot syndrome led to dose reductions of capecitabine in 13.5% of placebo but only 3.8% of those in the diclofenac group (P = .002).
The findings held regardless of whether patients were being treated for breast or GI cancer or if they were men or women.
Other capecitabine-induced adverse events, including diarrhea, mucositis, and myelosuppression, were not significantly different between the groups.
The treatment arms were well balanced, with a median age of 47 years in both groups and women making up about 70% of each. About 40% of subjects in each group were on capecitabine monotherapy with the rest on combination treatments. The mean dose of capecitabine was just over 1,880 mg/m2 in both groups.
At the meeting, Dr. Batra was asked if topical diclofenac would also work for another common problem in oncology: hand-food syndrome occurring as a side-effect with VEGF–tyrosine kinase inhibitors. He didn’t think so because it probably has a different cause than capecitabine HFS, one not strongly related to COX-2 up-regulation.
The study was partly funded by the Indian Supportive Care of Cancer Association. The investigators reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ASCO 2023
Hospital patient catches on fire, highlighting need for prevention
On Thanksgiving Day 2022, Kathy Stark watched as her husband of 35 years, Bobby Ray Stark, caught fire at a Nashville hospital. According to Clint Kelly, Kathy Stark’s attorney, the hospital staff was performing cardioversion to restore Bobby Ray’s heart rhythm when a spark ignited the oxygen and set the patient aflame.
Mr. Stark, 64, died of “a combination of cardiovascular disease and thermal burns,” according to a local news report. In May, Kathy Stark filed a malpractice lawsuit in U.S. District Court. Mr. Kelly hopes that the lawsuit will help improve patient safety. Meanwhile, Kathy Stark “goes to bed at night and sees her husband on fire,” Mr. Kelly says. A similar incident occurred last December in the operating room at Oregon Health & Science University, resulting in minor injuries to a patient.
Underreported, but likely dropping
Reliable data on the incidence of surgical fires is lacking because incidents may go unreported over litigation fears, says Jeffrey Feldman, MD, MSE, anesthesiologist at Children’s Hospital of Philadelphia and chair of the Anesthesia Patient Safety Foundation’s Committee on Technology.
The Pennsylvania Patient Safety Authority has been tracking surgical fires for decades, however, and experts have used the agency’s data to extrapolate how often they occur in the United States.
In 2005, nationwide incidence was estimated to be somewhere in the neighborhood of 550-600 fires annually, says Barbara G. Malanga, acting director of health care incident investigation and technology consulting at ECRI (formerly the Emergency Care Research Institute). By 2011, that number appeared to have dropped to 200-240 incidents per year.
A similar analysis in 2018 found the incidence may now be as low as 88-105 a year. The drop is likely a result of increased awareness because of educational efforts on the part of the ECRI and the APSF, including a widely disseminated video on fire safety.
The decline of surgical fires “sounds great,” says Dr. Feldman, “except that it’s a 100% preventable complication, and they’re still happening.”
Accidents waiting to happen
How do these fires happen? It comes down to the ‘fire triangle’ often taught in grade school. Fire requires three things: an ignition source, fuel, and oxygen or an oxidizing agent. Ignition sources are plentiful in a surgical suite, including any of a variety of electrical devices commonly used in surgical procedures, including defibrillators. Gowns, gauze, drapes, sponges, oxygen masks, nasal cannulae, a patient’s hair or their clothing – all provide the necessary fuel.
But the key factor for surgical fire risk is the presence of high concentrations of oxygen.
Safety protocols
The best and most obvious way to mitigate risk is to reduce the amount of supplemental oxygen, explains Dr. Feldman.
“Many patients do not require a high concentration of oxygen during sedation,” he says.
When a patient does require a higher concentration for their safety, the APSF and ECRI recommend placing an endotracheal tube or supraglottic airway rather than using an oxygen mask or a nasal cannula. “You want to deliver the oxygen in such a way that high concentration doesn’t exist in the surgical field,” Dr. Feldman says. In cases where supplemental oxygen is necessary, ECRI and APSF recommend reducing the oxygen concentration to less than 30%.
In addition, safety protocols include giving flammable prep solutions time to dry before applying towels or drapes and beginning the procedure. These precautions to ensure the safety of patients take just a moment, says Chester H. Lake Jr, MD, MS, of the department of anesthesiology at the University of Mississippi Medical Center, Jackson.
Making fire safety part of the preop routine
These safety protocols are straightforward but not always observed, experts say. Part of the reason is a matter of culture. Both anesthesiologists and surgeons have absorbed the attitude that placing an airway escalates the procedure beyond what the patient needs, says Dr. Feldman. And indeed, according to a 2013 analysis of the American Society of Anesthesiologists closed claims database, 85% of surgical fires occur in outpatient settings where airways are less likely to be placed, and 81% of those claims were for procedures that used monitored anesthesia care.
In an article on prevention of surgical fires, Dr. Lake and colleagues recommend in-house education on preventing and responding to fires at least once a year. But it shouldn’t stop there. Because these fires – horrific as they are – are fairly rare, it’s important to maintain awareness. Making fire safety a regular part of the surgical “time-out” can help further reduce incidents, he says. ECRI and the APSF have teamed up to create a poster that can help surgical teams make fire safety a regular part of their routines.
Although the national decline in surgical fires is encouraging, the problem remains serious. “You can classify these incidents as low, but it’s not low if it happens to you or a family member,” says Dr. Lake. “One is too many.”
ECRI’s Ms. Malanga agrees. “I do like to emphasize that it’s rare,” she says. “But I’d like to see us reduce this until it’s zero.”
A version of this article originally appeared on Medscape.com.
On Thanksgiving Day 2022, Kathy Stark watched as her husband of 35 years, Bobby Ray Stark, caught fire at a Nashville hospital. According to Clint Kelly, Kathy Stark’s attorney, the hospital staff was performing cardioversion to restore Bobby Ray’s heart rhythm when a spark ignited the oxygen and set the patient aflame.
Mr. Stark, 64, died of “a combination of cardiovascular disease and thermal burns,” according to a local news report. In May, Kathy Stark filed a malpractice lawsuit in U.S. District Court. Mr. Kelly hopes that the lawsuit will help improve patient safety. Meanwhile, Kathy Stark “goes to bed at night and sees her husband on fire,” Mr. Kelly says. A similar incident occurred last December in the operating room at Oregon Health & Science University, resulting in minor injuries to a patient.
Underreported, but likely dropping
Reliable data on the incidence of surgical fires is lacking because incidents may go unreported over litigation fears, says Jeffrey Feldman, MD, MSE, anesthesiologist at Children’s Hospital of Philadelphia and chair of the Anesthesia Patient Safety Foundation’s Committee on Technology.
The Pennsylvania Patient Safety Authority has been tracking surgical fires for decades, however, and experts have used the agency’s data to extrapolate how often they occur in the United States.
In 2005, nationwide incidence was estimated to be somewhere in the neighborhood of 550-600 fires annually, says Barbara G. Malanga, acting director of health care incident investigation and technology consulting at ECRI (formerly the Emergency Care Research Institute). By 2011, that number appeared to have dropped to 200-240 incidents per year.
A similar analysis in 2018 found the incidence may now be as low as 88-105 a year. The drop is likely a result of increased awareness because of educational efforts on the part of the ECRI and the APSF, including a widely disseminated video on fire safety.
The decline of surgical fires “sounds great,” says Dr. Feldman, “except that it’s a 100% preventable complication, and they’re still happening.”
Accidents waiting to happen
How do these fires happen? It comes down to the ‘fire triangle’ often taught in grade school. Fire requires three things: an ignition source, fuel, and oxygen or an oxidizing agent. Ignition sources are plentiful in a surgical suite, including any of a variety of electrical devices commonly used in surgical procedures, including defibrillators. Gowns, gauze, drapes, sponges, oxygen masks, nasal cannulae, a patient’s hair or their clothing – all provide the necessary fuel.
But the key factor for surgical fire risk is the presence of high concentrations of oxygen.
Safety protocols
The best and most obvious way to mitigate risk is to reduce the amount of supplemental oxygen, explains Dr. Feldman.
“Many patients do not require a high concentration of oxygen during sedation,” he says.
When a patient does require a higher concentration for their safety, the APSF and ECRI recommend placing an endotracheal tube or supraglottic airway rather than using an oxygen mask or a nasal cannula. “You want to deliver the oxygen in such a way that high concentration doesn’t exist in the surgical field,” Dr. Feldman says. In cases where supplemental oxygen is necessary, ECRI and APSF recommend reducing the oxygen concentration to less than 30%.
In addition, safety protocols include giving flammable prep solutions time to dry before applying towels or drapes and beginning the procedure. These precautions to ensure the safety of patients take just a moment, says Chester H. Lake Jr, MD, MS, of the department of anesthesiology at the University of Mississippi Medical Center, Jackson.
Making fire safety part of the preop routine
These safety protocols are straightforward but not always observed, experts say. Part of the reason is a matter of culture. Both anesthesiologists and surgeons have absorbed the attitude that placing an airway escalates the procedure beyond what the patient needs, says Dr. Feldman. And indeed, according to a 2013 analysis of the American Society of Anesthesiologists closed claims database, 85% of surgical fires occur in outpatient settings where airways are less likely to be placed, and 81% of those claims were for procedures that used monitored anesthesia care.
In an article on prevention of surgical fires, Dr. Lake and colleagues recommend in-house education on preventing and responding to fires at least once a year. But it shouldn’t stop there. Because these fires – horrific as they are – are fairly rare, it’s important to maintain awareness. Making fire safety a regular part of the surgical “time-out” can help further reduce incidents, he says. ECRI and the APSF have teamed up to create a poster that can help surgical teams make fire safety a regular part of their routines.
Although the national decline in surgical fires is encouraging, the problem remains serious. “You can classify these incidents as low, but it’s not low if it happens to you or a family member,” says Dr. Lake. “One is too many.”
ECRI’s Ms. Malanga agrees. “I do like to emphasize that it’s rare,” she says. “But I’d like to see us reduce this until it’s zero.”
A version of this article originally appeared on Medscape.com.
On Thanksgiving Day 2022, Kathy Stark watched as her husband of 35 years, Bobby Ray Stark, caught fire at a Nashville hospital. According to Clint Kelly, Kathy Stark’s attorney, the hospital staff was performing cardioversion to restore Bobby Ray’s heart rhythm when a spark ignited the oxygen and set the patient aflame.
Mr. Stark, 64, died of “a combination of cardiovascular disease and thermal burns,” according to a local news report. In May, Kathy Stark filed a malpractice lawsuit in U.S. District Court. Mr. Kelly hopes that the lawsuit will help improve patient safety. Meanwhile, Kathy Stark “goes to bed at night and sees her husband on fire,” Mr. Kelly says. A similar incident occurred last December in the operating room at Oregon Health & Science University, resulting in minor injuries to a patient.
Underreported, but likely dropping
Reliable data on the incidence of surgical fires is lacking because incidents may go unreported over litigation fears, says Jeffrey Feldman, MD, MSE, anesthesiologist at Children’s Hospital of Philadelphia and chair of the Anesthesia Patient Safety Foundation’s Committee on Technology.
The Pennsylvania Patient Safety Authority has been tracking surgical fires for decades, however, and experts have used the agency’s data to extrapolate how often they occur in the United States.
In 2005, nationwide incidence was estimated to be somewhere in the neighborhood of 550-600 fires annually, says Barbara G. Malanga, acting director of health care incident investigation and technology consulting at ECRI (formerly the Emergency Care Research Institute). By 2011, that number appeared to have dropped to 200-240 incidents per year.
A similar analysis in 2018 found the incidence may now be as low as 88-105 a year. The drop is likely a result of increased awareness because of educational efforts on the part of the ECRI and the APSF, including a widely disseminated video on fire safety.
The decline of surgical fires “sounds great,” says Dr. Feldman, “except that it’s a 100% preventable complication, and they’re still happening.”
Accidents waiting to happen
How do these fires happen? It comes down to the ‘fire triangle’ often taught in grade school. Fire requires three things: an ignition source, fuel, and oxygen or an oxidizing agent. Ignition sources are plentiful in a surgical suite, including any of a variety of electrical devices commonly used in surgical procedures, including defibrillators. Gowns, gauze, drapes, sponges, oxygen masks, nasal cannulae, a patient’s hair or their clothing – all provide the necessary fuel.
But the key factor for surgical fire risk is the presence of high concentrations of oxygen.
Safety protocols
The best and most obvious way to mitigate risk is to reduce the amount of supplemental oxygen, explains Dr. Feldman.
“Many patients do not require a high concentration of oxygen during sedation,” he says.
When a patient does require a higher concentration for their safety, the APSF and ECRI recommend placing an endotracheal tube or supraglottic airway rather than using an oxygen mask or a nasal cannula. “You want to deliver the oxygen in such a way that high concentration doesn’t exist in the surgical field,” Dr. Feldman says. In cases where supplemental oxygen is necessary, ECRI and APSF recommend reducing the oxygen concentration to less than 30%.
In addition, safety protocols include giving flammable prep solutions time to dry before applying towels or drapes and beginning the procedure. These precautions to ensure the safety of patients take just a moment, says Chester H. Lake Jr, MD, MS, of the department of anesthesiology at the University of Mississippi Medical Center, Jackson.
Making fire safety part of the preop routine
These safety protocols are straightforward but not always observed, experts say. Part of the reason is a matter of culture. Both anesthesiologists and surgeons have absorbed the attitude that placing an airway escalates the procedure beyond what the patient needs, says Dr. Feldman. And indeed, according to a 2013 analysis of the American Society of Anesthesiologists closed claims database, 85% of surgical fires occur in outpatient settings where airways are less likely to be placed, and 81% of those claims were for procedures that used monitored anesthesia care.
In an article on prevention of surgical fires, Dr. Lake and colleagues recommend in-house education on preventing and responding to fires at least once a year. But it shouldn’t stop there. Because these fires – horrific as they are – are fairly rare, it’s important to maintain awareness. Making fire safety a regular part of the surgical “time-out” can help further reduce incidents, he says. ECRI and the APSF have teamed up to create a poster that can help surgical teams make fire safety a regular part of their routines.
Although the national decline in surgical fires is encouraging, the problem remains serious. “You can classify these incidents as low, but it’s not low if it happens to you or a family member,” says Dr. Lake. “One is too many.”
ECRI’s Ms. Malanga agrees. “I do like to emphasize that it’s rare,” she says. “But I’d like to see us reduce this until it’s zero.”
A version of this article originally appeared on Medscape.com.
Dramatic rise in hallucinogen use among young adults
With the exception of lysergic acid diethylamide,
In 2018, the prevalence of young adults’ past-year use of non-LSD hallucinogens was 3.4%. By 2021, it had jumped to 6.6%.
The increase in non-LSD hallucinogen use occurred while LSD use remained stable at around 4% in 2018 and 2021.
“While non-LSD hallucinogen use remains substantially less prevalent than use of substances such as alcohol and cannabis, a doubling of prevalence in just three years is a dramatic increase and raises possible public health concerns,” co-author Megan Patrick, PhD, with the University of Michigan Institute for Social Research, Ann Arbor, said in a news release.
The results were published online in the journal Addiction.
Health concerns
The estimates are derived from the Monitoring the Future study, which includes annual assessments of adolescent and adult health in the United States.
The analysis focused on 11,304 persons (52% female) aged 9-30 years from the U.S. general population who were interviewed between 2018 and 2021.
Participants were asked about past 12-month use of LSD, as well as use of non-LSD hallucinogens, such as psilocybin.
From 2018 to 2021, past 12-month use of LSD remained relatively stable; it was 3.7% in 2018 and 4.2% in 2021.
However, non-LSD hallucinogen use increased in prevalence from 3.4% to 6.6% from 2018 to 2021.
Across years, the odds of non-LSD use were higher among males, White people, and individuals from households with higher parental education – a proxy for higher socioeconomic status.
The most commonly used non-LSD hallucinogen was psilocybin.
The survey did not ask whether young adults used non-LSD hallucinogens for therapeutic or medical reasons.
“The use of psychedelic and hallucinogenic drugs for a range of therapeutic uses is increasing, given accumulating yet still preliminary data from randomized trials on clinical effectiveness,” lead author Katherine Keyes, PhD, with Columbia University Mailman School of Public Health, New York, said in the release.
“With increased visibility for medical and therapeutic use, however, potentially comes diversion and unregulated product availability, as well as a lack of understanding among the public of potential risks,” Dr. Keyes added.
“However, approved therapeutic use of psychedelics under a trained health professional’s care remains uncommon in the United States, thus the trends we observe here are undoubtedly in nonmedical and nontherapeutic use,” Dr. Keyes noted.
Dr. Patrick said the increased use of hallucinogens raises “concern for young adult health” and is not without risk. While hallucinogen dependence has historically been rare in the U.S. population, it could become more common as use increases, she noted.
The researchers will continue to track these trends to see whether the increases continue.
“We need additional research, including about the motives for hallucinogen use and how young adults are using these substances, in order to be able to mitigate the associated negative consequences,” Dr. Patrick said.
The study was funded by the National Institute on Drug Abuse, part of the National Institutes of Health. Dr. Keyes and Dr. Patrick have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
With the exception of lysergic acid diethylamide,
In 2018, the prevalence of young adults’ past-year use of non-LSD hallucinogens was 3.4%. By 2021, it had jumped to 6.6%.
The increase in non-LSD hallucinogen use occurred while LSD use remained stable at around 4% in 2018 and 2021.
“While non-LSD hallucinogen use remains substantially less prevalent than use of substances such as alcohol and cannabis, a doubling of prevalence in just three years is a dramatic increase and raises possible public health concerns,” co-author Megan Patrick, PhD, with the University of Michigan Institute for Social Research, Ann Arbor, said in a news release.
The results were published online in the journal Addiction.
Health concerns
The estimates are derived from the Monitoring the Future study, which includes annual assessments of adolescent and adult health in the United States.
The analysis focused on 11,304 persons (52% female) aged 9-30 years from the U.S. general population who were interviewed between 2018 and 2021.
Participants were asked about past 12-month use of LSD, as well as use of non-LSD hallucinogens, such as psilocybin.
From 2018 to 2021, past 12-month use of LSD remained relatively stable; it was 3.7% in 2018 and 4.2% in 2021.
However, non-LSD hallucinogen use increased in prevalence from 3.4% to 6.6% from 2018 to 2021.
Across years, the odds of non-LSD use were higher among males, White people, and individuals from households with higher parental education – a proxy for higher socioeconomic status.
The most commonly used non-LSD hallucinogen was psilocybin.
The survey did not ask whether young adults used non-LSD hallucinogens for therapeutic or medical reasons.
“The use of psychedelic and hallucinogenic drugs for a range of therapeutic uses is increasing, given accumulating yet still preliminary data from randomized trials on clinical effectiveness,” lead author Katherine Keyes, PhD, with Columbia University Mailman School of Public Health, New York, said in the release.
“With increased visibility for medical and therapeutic use, however, potentially comes diversion and unregulated product availability, as well as a lack of understanding among the public of potential risks,” Dr. Keyes added.
“However, approved therapeutic use of psychedelics under a trained health professional’s care remains uncommon in the United States, thus the trends we observe here are undoubtedly in nonmedical and nontherapeutic use,” Dr. Keyes noted.
Dr. Patrick said the increased use of hallucinogens raises “concern for young adult health” and is not without risk. While hallucinogen dependence has historically been rare in the U.S. population, it could become more common as use increases, she noted.
The researchers will continue to track these trends to see whether the increases continue.
“We need additional research, including about the motives for hallucinogen use and how young adults are using these substances, in order to be able to mitigate the associated negative consequences,” Dr. Patrick said.
The study was funded by the National Institute on Drug Abuse, part of the National Institutes of Health. Dr. Keyes and Dr. Patrick have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
With the exception of lysergic acid diethylamide,
In 2018, the prevalence of young adults’ past-year use of non-LSD hallucinogens was 3.4%. By 2021, it had jumped to 6.6%.
The increase in non-LSD hallucinogen use occurred while LSD use remained stable at around 4% in 2018 and 2021.
“While non-LSD hallucinogen use remains substantially less prevalent than use of substances such as alcohol and cannabis, a doubling of prevalence in just three years is a dramatic increase and raises possible public health concerns,” co-author Megan Patrick, PhD, with the University of Michigan Institute for Social Research, Ann Arbor, said in a news release.
The results were published online in the journal Addiction.
Health concerns
The estimates are derived from the Monitoring the Future study, which includes annual assessments of adolescent and adult health in the United States.
The analysis focused on 11,304 persons (52% female) aged 9-30 years from the U.S. general population who were interviewed between 2018 and 2021.
Participants were asked about past 12-month use of LSD, as well as use of non-LSD hallucinogens, such as psilocybin.
From 2018 to 2021, past 12-month use of LSD remained relatively stable; it was 3.7% in 2018 and 4.2% in 2021.
However, non-LSD hallucinogen use increased in prevalence from 3.4% to 6.6% from 2018 to 2021.
Across years, the odds of non-LSD use were higher among males, White people, and individuals from households with higher parental education – a proxy for higher socioeconomic status.
The most commonly used non-LSD hallucinogen was psilocybin.
The survey did not ask whether young adults used non-LSD hallucinogens for therapeutic or medical reasons.
“The use of psychedelic and hallucinogenic drugs for a range of therapeutic uses is increasing, given accumulating yet still preliminary data from randomized trials on clinical effectiveness,” lead author Katherine Keyes, PhD, with Columbia University Mailman School of Public Health, New York, said in the release.
“With increased visibility for medical and therapeutic use, however, potentially comes diversion and unregulated product availability, as well as a lack of understanding among the public of potential risks,” Dr. Keyes added.
“However, approved therapeutic use of psychedelics under a trained health professional’s care remains uncommon in the United States, thus the trends we observe here are undoubtedly in nonmedical and nontherapeutic use,” Dr. Keyes noted.
Dr. Patrick said the increased use of hallucinogens raises “concern for young adult health” and is not without risk. While hallucinogen dependence has historically been rare in the U.S. population, it could become more common as use increases, she noted.
The researchers will continue to track these trends to see whether the increases continue.
“We need additional research, including about the motives for hallucinogen use and how young adults are using these substances, in order to be able to mitigate the associated negative consequences,” Dr. Patrick said.
The study was funded by the National Institute on Drug Abuse, part of the National Institutes of Health. Dr. Keyes and Dr. Patrick have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ADDICTION
The road to weight loss is paved with collusion and sabotage
Three big bumps on the weight-loss journey
The search for the Holy Grail. The destruction of the One Ring. The never-ending struggle to Lose Weight.
Like most legendary quests, weight loss is a journey, and we need support to help us achieve our goal. Maybe it’s gaining a new workout partner or finding a similarly-goaled Facebook Group. For a lot of people, it’s as simple as your friends and family. A recent study, however, suggests that the people closest to you may be your worst weight-loss enemies, and they might not even know it.
Researchers at the University of Surrey reviewed the literature on the positives and negatives of social support when it comes to weight loss and identified three types of negative effects: acts of sabotage, feeding behavior, and collusion.
Let’s start with the softest of intentions and work our way up. Collusion is the least negative. Friends and family may just go with the flow, even if it doesn’t agree with the goals of the person who’s trying to lose weight. It can even happen when health care professionals try to help their patients navigate or avoid obesity, ultimately killing with kindness, so to speak.
Next up, feeding behavior. Maybe you know someone whose love language is cooking. There are also people who share food because they don’t want to waste it or because they’re trying to be polite. They act out of the goodness of their hearts, but they’re putting up roadblocks to someone’s goals. These types of acts are usually one-sided, the researchers found. Remember, it’s okay to say, “No thanks.”
The last method, sabotage, is the most sinister. The saboteur may discourage others from eating healthy, undermine their efforts to be physically active, or take jabs at their confidence or self-esteem. Something as simple as criticizing someone for eating a salad or refusing to go on a walk with them can cause a setback.
“We need to explore this area further to develop interventions which could target family and friends and help them be more supportive in helping those they are close to lose weight,” said lead author Jane Odgen, PhD, of the University of Surrey, Guildford, England.
Like we said before, weight loss is a journey. The right support can only improve the odds of success.
Robots vs. mosquitoes
If there’s one thing robots are bad at, it’s giving solid mental health advice to people in crisis. If there’s one thing robots are very, very good at, it’s causing apocalypses. And joyous day for humanity, this time we’re not the ones being apocalypsed.
Yet.
Taiwan has a big mosquito problem. Not only do the mosquitoes in Taiwan carry dengue – among other dangerous diseases – but they’ve urbanized. Not urbanized in the sense that they’ve acquired a taste for organic coffee and avocado toast (that would be the millennial mosquito, a separate but even more terrifying creature), but more that they’ve adapted to reproduce literally anywhere and everywhere. Taiwanese mosquitoes like to breed in roadside sewer ditches, and this is where our genocidal robot comes in.
To combat the new, dangerous form of street-savvy mosquito, researchers built a robot armed with both insecticide and high-temperature, high-pressure water jets and sent it into the sewers of Kaohsiung City. The robot’s goal was simple: Whenever it came across signs of heavy mosquito breeding – eggs, larvae, pupae, and so on – the robot went to work. Utilizing both its primary weapons, the robot scrubbed numerous breeding sites across the city clean.
The researchers could just sit back and wait to see how effective their robot was. In the immediate aftermath, at various monitoring sites placed alongside the ditches, adult mosquito density fell by two-thirds in areas targeted by the robot. That’s nothing to sniff at, and it does make sense. After all, mosquitoes are quite difficult to kill in their adult stage, why not target them when they’re young and basically immobile?
The researchers saw promise with their mosquito-killing robot, but we’ve noticed a rather large issue. Killing two-thirds of mosquitoes is fine, but the third that’s left will be very angry. Very angry indeed. After all, we’re targeting the mosquito equivalent of children. Let’s hope our mosquito Terminator managed to kill mosquito Sarah Connor, or we’re going to have a big problem on our hands a bit later down the line.
This is knot what you were expecting
Physicians who aren’t surgeons probably don’t realize it, but the big thing that’s been getting between the knot-tying specialists and perfect suturing technique all these years is a lack of physics. Don’t believe us? Well, maybe you’ll believe plastic surgeon Samia Guerid, MD, of Lausanne, Switzerland: “The lack of physics-based analysis has been a limitation.” Nuff said.
That’s not enough for you, is it? Fine, we were warned.
Any surgical knot, Dr. Guerid and associates explained in a written statement, involves the “complex interplay” between six key factors: topology, geometry, elasticity, contact, friction, and polymer plasticity of the suturing filament. The strength of a suture “depends on the tension applied during the tying of the knot, [which] permanently deforms, or stretches the filament, creating a holding force.” Not enough tension and the knot comes undone, while too much snaps the filament.
For the experiment, Dr. Guerid tied a few dozen surgical knots, which were then scanned using x-ray micro–computed tomography to facilitate finite element modeling with a “3D continuum-level constitutive model for elastic-viscoplastic mechanical behavior” – no, we have no idea what that means, either – developed by the research team.
That model, and a great deal of math – so much math – allowed the researchers to define a threshold between loose and tight knots and uncover “relationships between knot strength and pretension, friction, and number of throws,” they said.
But what about the big question? The one about the ideal amount of tension? You may want to sit down. The answer to the ultimate question of the relationship between knot pretension and strength is … Did we mention that the team had its own mathematician? Their predictive model for safe knot-tying is … You’re not going to like this. The best way to teach safe knot-tying to both trainees and robots is … not ready yet.
The secret to targeting the knot tension sweet spot, for now, anyway, is still intuition gained from years of experience. Nobody ever said science was perfect … or easy … or quick.
Three big bumps on the weight-loss journey
The search for the Holy Grail. The destruction of the One Ring. The never-ending struggle to Lose Weight.
Like most legendary quests, weight loss is a journey, and we need support to help us achieve our goal. Maybe it’s gaining a new workout partner or finding a similarly-goaled Facebook Group. For a lot of people, it’s as simple as your friends and family. A recent study, however, suggests that the people closest to you may be your worst weight-loss enemies, and they might not even know it.
Researchers at the University of Surrey reviewed the literature on the positives and negatives of social support when it comes to weight loss and identified three types of negative effects: acts of sabotage, feeding behavior, and collusion.
Let’s start with the softest of intentions and work our way up. Collusion is the least negative. Friends and family may just go with the flow, even if it doesn’t agree with the goals of the person who’s trying to lose weight. It can even happen when health care professionals try to help their patients navigate or avoid obesity, ultimately killing with kindness, so to speak.
Next up, feeding behavior. Maybe you know someone whose love language is cooking. There are also people who share food because they don’t want to waste it or because they’re trying to be polite. They act out of the goodness of their hearts, but they’re putting up roadblocks to someone’s goals. These types of acts are usually one-sided, the researchers found. Remember, it’s okay to say, “No thanks.”
The last method, sabotage, is the most sinister. The saboteur may discourage others from eating healthy, undermine their efforts to be physically active, or take jabs at their confidence or self-esteem. Something as simple as criticizing someone for eating a salad or refusing to go on a walk with them can cause a setback.
“We need to explore this area further to develop interventions which could target family and friends and help them be more supportive in helping those they are close to lose weight,” said lead author Jane Odgen, PhD, of the University of Surrey, Guildford, England.
Like we said before, weight loss is a journey. The right support can only improve the odds of success.
Robots vs. mosquitoes
If there’s one thing robots are bad at, it’s giving solid mental health advice to people in crisis. If there’s one thing robots are very, very good at, it’s causing apocalypses. And joyous day for humanity, this time we’re not the ones being apocalypsed.
Yet.
Taiwan has a big mosquito problem. Not only do the mosquitoes in Taiwan carry dengue – among other dangerous diseases – but they’ve urbanized. Not urbanized in the sense that they’ve acquired a taste for organic coffee and avocado toast (that would be the millennial mosquito, a separate but even more terrifying creature), but more that they’ve adapted to reproduce literally anywhere and everywhere. Taiwanese mosquitoes like to breed in roadside sewer ditches, and this is where our genocidal robot comes in.
To combat the new, dangerous form of street-savvy mosquito, researchers built a robot armed with both insecticide and high-temperature, high-pressure water jets and sent it into the sewers of Kaohsiung City. The robot’s goal was simple: Whenever it came across signs of heavy mosquito breeding – eggs, larvae, pupae, and so on – the robot went to work. Utilizing both its primary weapons, the robot scrubbed numerous breeding sites across the city clean.
The researchers could just sit back and wait to see how effective their robot was. In the immediate aftermath, at various monitoring sites placed alongside the ditches, adult mosquito density fell by two-thirds in areas targeted by the robot. That’s nothing to sniff at, and it does make sense. After all, mosquitoes are quite difficult to kill in their adult stage, why not target them when they’re young and basically immobile?
The researchers saw promise with their mosquito-killing robot, but we’ve noticed a rather large issue. Killing two-thirds of mosquitoes is fine, but the third that’s left will be very angry. Very angry indeed. After all, we’re targeting the mosquito equivalent of children. Let’s hope our mosquito Terminator managed to kill mosquito Sarah Connor, or we’re going to have a big problem on our hands a bit later down the line.
This is knot what you were expecting
Physicians who aren’t surgeons probably don’t realize it, but the big thing that’s been getting between the knot-tying specialists and perfect suturing technique all these years is a lack of physics. Don’t believe us? Well, maybe you’ll believe plastic surgeon Samia Guerid, MD, of Lausanne, Switzerland: “The lack of physics-based analysis has been a limitation.” Nuff said.
That’s not enough for you, is it? Fine, we were warned.
Any surgical knot, Dr. Guerid and associates explained in a written statement, involves the “complex interplay” between six key factors: topology, geometry, elasticity, contact, friction, and polymer plasticity of the suturing filament. The strength of a suture “depends on the tension applied during the tying of the knot, [which] permanently deforms, or stretches the filament, creating a holding force.” Not enough tension and the knot comes undone, while too much snaps the filament.
For the experiment, Dr. Guerid tied a few dozen surgical knots, which were then scanned using x-ray micro–computed tomography to facilitate finite element modeling with a “3D continuum-level constitutive model for elastic-viscoplastic mechanical behavior” – no, we have no idea what that means, either – developed by the research team.
That model, and a great deal of math – so much math – allowed the researchers to define a threshold between loose and tight knots and uncover “relationships between knot strength and pretension, friction, and number of throws,” they said.
But what about the big question? The one about the ideal amount of tension? You may want to sit down. The answer to the ultimate question of the relationship between knot pretension and strength is … Did we mention that the team had its own mathematician? Their predictive model for safe knot-tying is … You’re not going to like this. The best way to teach safe knot-tying to both trainees and robots is … not ready yet.
The secret to targeting the knot tension sweet spot, for now, anyway, is still intuition gained from years of experience. Nobody ever said science was perfect … or easy … or quick.
Three big bumps on the weight-loss journey
The search for the Holy Grail. The destruction of the One Ring. The never-ending struggle to Lose Weight.
Like most legendary quests, weight loss is a journey, and we need support to help us achieve our goal. Maybe it’s gaining a new workout partner or finding a similarly-goaled Facebook Group. For a lot of people, it’s as simple as your friends and family. A recent study, however, suggests that the people closest to you may be your worst weight-loss enemies, and they might not even know it.
Researchers at the University of Surrey reviewed the literature on the positives and negatives of social support when it comes to weight loss and identified three types of negative effects: acts of sabotage, feeding behavior, and collusion.
Let’s start with the softest of intentions and work our way up. Collusion is the least negative. Friends and family may just go with the flow, even if it doesn’t agree with the goals of the person who’s trying to lose weight. It can even happen when health care professionals try to help their patients navigate or avoid obesity, ultimately killing with kindness, so to speak.
Next up, feeding behavior. Maybe you know someone whose love language is cooking. There are also people who share food because they don’t want to waste it or because they’re trying to be polite. They act out of the goodness of their hearts, but they’re putting up roadblocks to someone’s goals. These types of acts are usually one-sided, the researchers found. Remember, it’s okay to say, “No thanks.”
The last method, sabotage, is the most sinister. The saboteur may discourage others from eating healthy, undermine their efforts to be physically active, or take jabs at their confidence or self-esteem. Something as simple as criticizing someone for eating a salad or refusing to go on a walk with them can cause a setback.
“We need to explore this area further to develop interventions which could target family and friends and help them be more supportive in helping those they are close to lose weight,” said lead author Jane Odgen, PhD, of the University of Surrey, Guildford, England.
Like we said before, weight loss is a journey. The right support can only improve the odds of success.
Robots vs. mosquitoes
If there’s one thing robots are bad at, it’s giving solid mental health advice to people in crisis. If there’s one thing robots are very, very good at, it’s causing apocalypses. And joyous day for humanity, this time we’re not the ones being apocalypsed.
Yet.
Taiwan has a big mosquito problem. Not only do the mosquitoes in Taiwan carry dengue – among other dangerous diseases – but they’ve urbanized. Not urbanized in the sense that they’ve acquired a taste for organic coffee and avocado toast (that would be the millennial mosquito, a separate but even more terrifying creature), but more that they’ve adapted to reproduce literally anywhere and everywhere. Taiwanese mosquitoes like to breed in roadside sewer ditches, and this is where our genocidal robot comes in.
To combat the new, dangerous form of street-savvy mosquito, researchers built a robot armed with both insecticide and high-temperature, high-pressure water jets and sent it into the sewers of Kaohsiung City. The robot’s goal was simple: Whenever it came across signs of heavy mosquito breeding – eggs, larvae, pupae, and so on – the robot went to work. Utilizing both its primary weapons, the robot scrubbed numerous breeding sites across the city clean.
The researchers could just sit back and wait to see how effective their robot was. In the immediate aftermath, at various monitoring sites placed alongside the ditches, adult mosquito density fell by two-thirds in areas targeted by the robot. That’s nothing to sniff at, and it does make sense. After all, mosquitoes are quite difficult to kill in their adult stage, why not target them when they’re young and basically immobile?
The researchers saw promise with their mosquito-killing robot, but we’ve noticed a rather large issue. Killing two-thirds of mosquitoes is fine, but the third that’s left will be very angry. Very angry indeed. After all, we’re targeting the mosquito equivalent of children. Let’s hope our mosquito Terminator managed to kill mosquito Sarah Connor, or we’re going to have a big problem on our hands a bit later down the line.
This is knot what you were expecting
Physicians who aren’t surgeons probably don’t realize it, but the big thing that’s been getting between the knot-tying specialists and perfect suturing technique all these years is a lack of physics. Don’t believe us? Well, maybe you’ll believe plastic surgeon Samia Guerid, MD, of Lausanne, Switzerland: “The lack of physics-based analysis has been a limitation.” Nuff said.
That’s not enough for you, is it? Fine, we were warned.
Any surgical knot, Dr. Guerid and associates explained in a written statement, involves the “complex interplay” between six key factors: topology, geometry, elasticity, contact, friction, and polymer plasticity of the suturing filament. The strength of a suture “depends on the tension applied during the tying of the knot, [which] permanently deforms, or stretches the filament, creating a holding force.” Not enough tension and the knot comes undone, while too much snaps the filament.
For the experiment, Dr. Guerid tied a few dozen surgical knots, which were then scanned using x-ray micro–computed tomography to facilitate finite element modeling with a “3D continuum-level constitutive model for elastic-viscoplastic mechanical behavior” – no, we have no idea what that means, either – developed by the research team.
That model, and a great deal of math – so much math – allowed the researchers to define a threshold between loose and tight knots and uncover “relationships between knot strength and pretension, friction, and number of throws,” they said.
But what about the big question? The one about the ideal amount of tension? You may want to sit down. The answer to the ultimate question of the relationship between knot pretension and strength is … Did we mention that the team had its own mathematician? Their predictive model for safe knot-tying is … You’re not going to like this. The best way to teach safe knot-tying to both trainees and robots is … not ready yet.
The secret to targeting the knot tension sweet spot, for now, anyway, is still intuition gained from years of experience. Nobody ever said science was perfect … or easy … or quick.