User login
For MD-IQ use only
Analysis of a Pilot Curriculum for Business Education in Dermatology Residency
To the Editor:
With health care constituting one of the larger segments of the US economy, medical practice is increasingly subject to business considerations.1 Patients, providers, and organizations are all required to make decisions that reflect choices beyond clinical needs alone. Given the impact of market forces, clinicians often are asked to navigate operational and business decisions. Accordingly, education about the policy and systems that shape care delivery can improve quality and help patients.2
The ability to understand the ecosystem of health care is of utmost importance for medical providers and can be achieved through resident education. Teaching fundamental business concepts enables residents to deliver care that is responsive to the constraints and opportunities encountered by patients and organizations, which ultimately will better prepare them to serve as advocates in alignment with their principal duties as physicians.
Despite the recognizable relationship between business and medicine, training has not yet been standardized to include topics in business education, and clinicians in dermatology are remarkably positioned to benefit because of the variety of practice settings and services they can provide. In dermatology, the diversity of services provided gives rise to complex coding and use of modifiers. Proper utilization of coding and billing is critical to create accurate documentation and receive appropriate reimbursement.3 Furthermore, clinicians in dermatology have to contend with the influence of insurance at many points of care, such as with coverage of pharmaceuticals. Formularies often have wide variability in coverage and are changing as new drugs come to market in the dermatologic space.4
The landscape of practice structure also has undergone change with increasing consolidation and mergers. The acquisition of practices by private equity firms has induced changes in practice infrastructure. The impact of changing organizational and managerial influences continues to be a topic of debate, with disparate opinions on how these developments shape standards of physician satisfaction and patient care.5
The convergence of these factors points to an important question that is gaining popularity: How will young dermatologists work within the context of all these parameters to best advocate and care for their patients? These questions are garnering more attention and were recently investigated through a survey of participants in a pilot program to evaluate the importance of business education in dermatology residency.
A survey of residency program directors was created by Patrinley and Dewan,6 which found that business education during residency was important and additional training should be implemented. Despite the perceived importance of business education, only half of the programs represented by survey respondents offered any structured educational opportunities, revealing a discrepancy between believed importance and practical implementation of business training, which suggests the need to develop a standardized, dermatology-specific curriculum that could be accessed by all residents in training.6
We performed a search of the medical literature to identify models of business education in residency programs. Only a few programs were identified, in which courses were predominantly instructed to trainees in primary care–based fields. According to course graduates, the programs were beneficial.7,8 Programs that had descriptive information about curriculum structure and content were chosen for further investigation and included internal medicine programs at the University of California San Francisco (UCSF) and Columbia University Vagelos College of Physicians and Surgeons (New York, New York). UCSF implemented a Program in Residency Investigation Methods and Epidemiology (PRIME program) to deliver seven 90-minute sessions dedicated to introducing residents to medical economics. Sessions were constructed with the intent of being interactive seminars that took on a variety of forms, including reading-based discussions, case-based analysis, and simulation-based learning.7 Columbia University developed a pilot program of week-long didactic sessions that were delivered to third-year internal medicine residents. These seminars featured discussions on health policy and economics, health insurance, technology and cost assessment, legal medicine, public health, community-oriented primary care, and local health department initiatives.8 We drew on both courses to build a lecture series focused on the business of dermatology that was delivered to dermatology residents at UMass Chan Medical School (Worcester, Massachusetts). Topic selection also was informed by qualitative input collected via email from recent graduates of the UMass dermatology residency program, focusing on the following areas: the US medical economy and health care costs; billing, coding, and claims processing; quality, relative value units (RVUs), reimbursement, and the merit-based incentive payment system; coverage of pharmaceuticals and teledermatology; and management. Residents were not required to prepare for any of the sessions; they were provided with handouts and slideshow presentations for reference to review at their convenience if desired. Five seminars were virtually conducted by an MD/MBA candidate at the institution (E.H.). They were recorded over the course of an academic year at 1- to 2-month intervals. Each 45-minute session was conducted in a lecture-discussion format and included case examples to help illustrate key principles and stimulate conversation. For example, the lecture on reimbursement incorporated a fee schedule calculation for a shave biopsy, using RVU and geographic pricing cost index (GCPI) multipliers. This demonstrated the variation in Centers for Medicare & Medicaid Services reimbursement in relation to (1) constituents of the RVU calculation (ie, work, practice expense, and malpractice) and (2) practice in a particular location (ie, the GCPI). Following this example, a conversation ensued among participants regarding the factors that drive valuation, with particular interest in variation based on urban vs suburban locations across the United States. Participants also found it of interest to examine the percentage of the valuation dedicated to each constituent and how features such as lesion size informed the final assessment of the charge. Another stylistic choice in developing the model was to include prompts for further consideration prior to transitioning topics in the lectures. For example: when examining the burden of skin disease, the audience was prompted to consider: “What is driving cost escalations, and how will services of the clinical domain meet these evolving needs?” At another point in the introductory lecture, residents were asked: “How do different types of insurance plans impact the management of patients with dermatologic concerns?” These questions were intended to transition residents to the next topic of discussion and highlight take-home points of consideration for medical practice. The project was reviewed by the UMass institutional review board and met criteria for exemption.
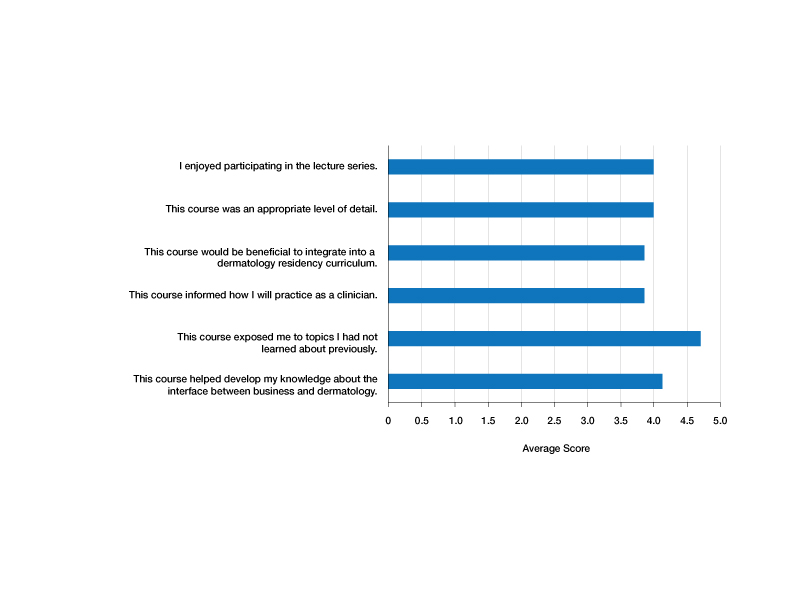
Residents who participated in at least 1 lecture (N=10) were surveyed after attendance; there were 7 responses (70% response rate). Residents were asked to rate a series of statements on a scale of 1 (strongly disagree) to 5 (strongly agree) and to provide commentary via an online form. Respondents indicated that the course was enjoyable (average score, 4.00), provided an appropriate level of detail (average score, 4.00), would be beneficial to integrate into a dermatology residency curriculum (average score, 3.86), and informed how they would practice as a clinician (average score, 3.86)(Figure). The respondents agreed that the course met the main goals of this initiative: it helped them develop knowledge about the interface between business and dermatology (4.14) and exposed residents to topics they had not learned about previously (4.71).
Although the course generally was well received, areas for improvement were identified from respondents’ comments, relating to audience engagement and refining the level of detail in the lectures. Recommendations included “less technical jargon and more focus on ‘big picture’ concepts, given audience’s low baseline knowledge”; “more case examples in each module”; and “more diagrams or interactive activities (polls, quizzes, break-out rooms) because the lectures were a bit dense.” This input was taken into consideration when revising the lectures for future use; they were reconstructed to have more case-based examples and prompts to encourage participation.
Resident commentary also demonstrated appreciation for education in this subject material. Statements such as “this is an important topic for future dermatologists” and “thank you so much for taking the time to implement this course” reflected the perceived value of this material during critical academic time. Another resident remarked: “This was great, thanks for putting it together.”
Given the positive experience of the residents and successful implementation of the series, this course was made available to all dermatology trainees on a network server with accompanying written documents. It is planned to be offered on a 3-year cycle in the future and will be updated to reflect inevitable changes in health care.
Although the relationship between business and medicine is increasingly important, teaching business principles has not become standardized or required in medical training. Despite the perception that this content is of value, implementation of programming has lagged behind that recognition, likely due to challenges in designing the curriculum and diffusing content into an already-saturated schedule. A model course that can be replicated in other residency programs would be valuable. We introduced a dermatology-specific lecture series to help prepare trainees for dermatology practice in a variety of clinical settings and train them with the language of business and operations that will equip them to respond to the needs of their patients, their practice, and the medical environment. Findings of this pilot study may not be generalizable to all dermatology residency programs because the sample size was small; the study was conducted at a single institution; and the content was delivered entirely online.
1. Tan S, Seiger K, Renehan P, et al. Trends in private equity acquisition of dermatology practices in the United States. JAMA Dermatol. 2019;155:1013-1021. doi:10.1001/jamadermatol.2019.1634
2. The business of health care in the United States. Harvard Online [Internet]. June 27, 2022. Accessed July 24, 2023. https://www.harvardonline.harvard.edu/blog/business-health-care-united-states
3. Ranpariya V, Cull D, Feldman SR, et al. Evaluation and management 2021 coding guidelines: key changes and implications. The Dermatologist. December 2020. Accessed July 24, 2023. https://www.hmpgloballearningnetwork.com/site/thederm/article/evaluation-and-management-2021-coding-guidelines-key-changes-and-implications?key=Ranpariya&elastic%5B0%5D=brand%3A73468
4. Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76:958-972.e2. doi:10.1016/j.jaad.2016.12.043
5. Resneck JS Jr. Dermatology practice consolidation fueled by private equity investment: potential consequences for the specialty and patients. JAMA Dermatol. 2018;154:13-14. doi:10.1001/jamadermatol.2017.5558
6. Patrinely JR Jr, Dewan AK. Business education in dermatology residency: a survey of program directors. Cutis. 2021;108:E7-E19. doi:10.12788/cutis.0331
7. Kohlwes RJ, Chou CL. A curriculum in medical economics for residents. Acad Med. 2002;77:465-466. doi:10.1097/00001888-200205000-00040
8. Fiebach NH, Rao D, Hamm ME. A curriculum in health systems and public health for internal medicine residents. Am J Prev Med. 2011;41(4 suppl 3):S264-S269. doi:10.1016/j.amepre.2011.05.025
To the Editor:
With health care constituting one of the larger segments of the US economy, medical practice is increasingly subject to business considerations.1 Patients, providers, and organizations are all required to make decisions that reflect choices beyond clinical needs alone. Given the impact of market forces, clinicians often are asked to navigate operational and business decisions. Accordingly, education about the policy and systems that shape care delivery can improve quality and help patients.2
The ability to understand the ecosystem of health care is of utmost importance for medical providers and can be achieved through resident education. Teaching fundamental business concepts enables residents to deliver care that is responsive to the constraints and opportunities encountered by patients and organizations, which ultimately will better prepare them to serve as advocates in alignment with their principal duties as physicians.
Despite the recognizable relationship between business and medicine, training has not yet been standardized to include topics in business education, and clinicians in dermatology are remarkably positioned to benefit because of the variety of practice settings and services they can provide. In dermatology, the diversity of services provided gives rise to complex coding and use of modifiers. Proper utilization of coding and billing is critical to create accurate documentation and receive appropriate reimbursement.3 Furthermore, clinicians in dermatology have to contend with the influence of insurance at many points of care, such as with coverage of pharmaceuticals. Formularies often have wide variability in coverage and are changing as new drugs come to market in the dermatologic space.4
The landscape of practice structure also has undergone change with increasing consolidation and mergers. The acquisition of practices by private equity firms has induced changes in practice infrastructure. The impact of changing organizational and managerial influences continues to be a topic of debate, with disparate opinions on how these developments shape standards of physician satisfaction and patient care.5
The convergence of these factors points to an important question that is gaining popularity: How will young dermatologists work within the context of all these parameters to best advocate and care for their patients? These questions are garnering more attention and were recently investigated through a survey of participants in a pilot program to evaluate the importance of business education in dermatology residency.
A survey of residency program directors was created by Patrinley and Dewan,6 which found that business education during residency was important and additional training should be implemented. Despite the perceived importance of business education, only half of the programs represented by survey respondents offered any structured educational opportunities, revealing a discrepancy between believed importance and practical implementation of business training, which suggests the need to develop a standardized, dermatology-specific curriculum that could be accessed by all residents in training.6
We performed a search of the medical literature to identify models of business education in residency programs. Only a few programs were identified, in which courses were predominantly instructed to trainees in primary care–based fields. According to course graduates, the programs were beneficial.7,8 Programs that had descriptive information about curriculum structure and content were chosen for further investigation and included internal medicine programs at the University of California San Francisco (UCSF) and Columbia University Vagelos College of Physicians and Surgeons (New York, New York). UCSF implemented a Program in Residency Investigation Methods and Epidemiology (PRIME program) to deliver seven 90-minute sessions dedicated to introducing residents to medical economics. Sessions were constructed with the intent of being interactive seminars that took on a variety of forms, including reading-based discussions, case-based analysis, and simulation-based learning.7 Columbia University developed a pilot program of week-long didactic sessions that were delivered to third-year internal medicine residents. These seminars featured discussions on health policy and economics, health insurance, technology and cost assessment, legal medicine, public health, community-oriented primary care, and local health department initiatives.8 We drew on both courses to build a lecture series focused on the business of dermatology that was delivered to dermatology residents at UMass Chan Medical School (Worcester, Massachusetts). Topic selection also was informed by qualitative input collected via email from recent graduates of the UMass dermatology residency program, focusing on the following areas: the US medical economy and health care costs; billing, coding, and claims processing; quality, relative value units (RVUs), reimbursement, and the merit-based incentive payment system; coverage of pharmaceuticals and teledermatology; and management. Residents were not required to prepare for any of the sessions; they were provided with handouts and slideshow presentations for reference to review at their convenience if desired. Five seminars were virtually conducted by an MD/MBA candidate at the institution (E.H.). They were recorded over the course of an academic year at 1- to 2-month intervals. Each 45-minute session was conducted in a lecture-discussion format and included case examples to help illustrate key principles and stimulate conversation. For example, the lecture on reimbursement incorporated a fee schedule calculation for a shave biopsy, using RVU and geographic pricing cost index (GCPI) multipliers. This demonstrated the variation in Centers for Medicare & Medicaid Services reimbursement in relation to (1) constituents of the RVU calculation (ie, work, practice expense, and malpractice) and (2) practice in a particular location (ie, the GCPI). Following this example, a conversation ensued among participants regarding the factors that drive valuation, with particular interest in variation based on urban vs suburban locations across the United States. Participants also found it of interest to examine the percentage of the valuation dedicated to each constituent and how features such as lesion size informed the final assessment of the charge. Another stylistic choice in developing the model was to include prompts for further consideration prior to transitioning topics in the lectures. For example: when examining the burden of skin disease, the audience was prompted to consider: “What is driving cost escalations, and how will services of the clinical domain meet these evolving needs?” At another point in the introductory lecture, residents were asked: “How do different types of insurance plans impact the management of patients with dermatologic concerns?” These questions were intended to transition residents to the next topic of discussion and highlight take-home points of consideration for medical practice. The project was reviewed by the UMass institutional review board and met criteria for exemption.
Residents who participated in at least 1 lecture (N=10) were surveyed after attendance; there were 7 responses (70% response rate). Residents were asked to rate a series of statements on a scale of 1 (strongly disagree) to 5 (strongly agree) and to provide commentary via an online form. Respondents indicated that the course was enjoyable (average score, 4.00), provided an appropriate level of detail (average score, 4.00), would be beneficial to integrate into a dermatology residency curriculum (average score, 3.86), and informed how they would practice as a clinician (average score, 3.86)(Figure). The respondents agreed that the course met the main goals of this initiative: it helped them develop knowledge about the interface between business and dermatology (4.14) and exposed residents to topics they had not learned about previously (4.71).

Although the course generally was well received, areas for improvement were identified from respondents’ comments, relating to audience engagement and refining the level of detail in the lectures. Recommendations included “less technical jargon and more focus on ‘big picture’ concepts, given audience’s low baseline knowledge”; “more case examples in each module”; and “more diagrams or interactive activities (polls, quizzes, break-out rooms) because the lectures were a bit dense.” This input was taken into consideration when revising the lectures for future use; they were reconstructed to have more case-based examples and prompts to encourage participation.
Resident commentary also demonstrated appreciation for education in this subject material. Statements such as “this is an important topic for future dermatologists” and “thank you so much for taking the time to implement this course” reflected the perceived value of this material during critical academic time. Another resident remarked: “This was great, thanks for putting it together.”
Given the positive experience of the residents and successful implementation of the series, this course was made available to all dermatology trainees on a network server with accompanying written documents. It is planned to be offered on a 3-year cycle in the future and will be updated to reflect inevitable changes in health care.
Although the relationship between business and medicine is increasingly important, teaching business principles has not become standardized or required in medical training. Despite the perception that this content is of value, implementation of programming has lagged behind that recognition, likely due to challenges in designing the curriculum and diffusing content into an already-saturated schedule. A model course that can be replicated in other residency programs would be valuable. We introduced a dermatology-specific lecture series to help prepare trainees for dermatology practice in a variety of clinical settings and train them with the language of business and operations that will equip them to respond to the needs of their patients, their practice, and the medical environment. Findings of this pilot study may not be generalizable to all dermatology residency programs because the sample size was small; the study was conducted at a single institution; and the content was delivered entirely online.
To the Editor:
With health care constituting one of the larger segments of the US economy, medical practice is increasingly subject to business considerations.1 Patients, providers, and organizations are all required to make decisions that reflect choices beyond clinical needs alone. Given the impact of market forces, clinicians often are asked to navigate operational and business decisions. Accordingly, education about the policy and systems that shape care delivery can improve quality and help patients.2
The ability to understand the ecosystem of health care is of utmost importance for medical providers and can be achieved through resident education. Teaching fundamental business concepts enables residents to deliver care that is responsive to the constraints and opportunities encountered by patients and organizations, which ultimately will better prepare them to serve as advocates in alignment with their principal duties as physicians.
Despite the recognizable relationship between business and medicine, training has not yet been standardized to include topics in business education, and clinicians in dermatology are remarkably positioned to benefit because of the variety of practice settings and services they can provide. In dermatology, the diversity of services provided gives rise to complex coding and use of modifiers. Proper utilization of coding and billing is critical to create accurate documentation and receive appropriate reimbursement.3 Furthermore, clinicians in dermatology have to contend with the influence of insurance at many points of care, such as with coverage of pharmaceuticals. Formularies often have wide variability in coverage and are changing as new drugs come to market in the dermatologic space.4
The landscape of practice structure also has undergone change with increasing consolidation and mergers. The acquisition of practices by private equity firms has induced changes in practice infrastructure. The impact of changing organizational and managerial influences continues to be a topic of debate, with disparate opinions on how these developments shape standards of physician satisfaction and patient care.5
The convergence of these factors points to an important question that is gaining popularity: How will young dermatologists work within the context of all these parameters to best advocate and care for their patients? These questions are garnering more attention and were recently investigated through a survey of participants in a pilot program to evaluate the importance of business education in dermatology residency.
A survey of residency program directors was created by Patrinley and Dewan,6 which found that business education during residency was important and additional training should be implemented. Despite the perceived importance of business education, only half of the programs represented by survey respondents offered any structured educational opportunities, revealing a discrepancy between believed importance and practical implementation of business training, which suggests the need to develop a standardized, dermatology-specific curriculum that could be accessed by all residents in training.6
We performed a search of the medical literature to identify models of business education in residency programs. Only a few programs were identified, in which courses were predominantly instructed to trainees in primary care–based fields. According to course graduates, the programs were beneficial.7,8 Programs that had descriptive information about curriculum structure and content were chosen for further investigation and included internal medicine programs at the University of California San Francisco (UCSF) and Columbia University Vagelos College of Physicians and Surgeons (New York, New York). UCSF implemented a Program in Residency Investigation Methods and Epidemiology (PRIME program) to deliver seven 90-minute sessions dedicated to introducing residents to medical economics. Sessions were constructed with the intent of being interactive seminars that took on a variety of forms, including reading-based discussions, case-based analysis, and simulation-based learning.7 Columbia University developed a pilot program of week-long didactic sessions that were delivered to third-year internal medicine residents. These seminars featured discussions on health policy and economics, health insurance, technology and cost assessment, legal medicine, public health, community-oriented primary care, and local health department initiatives.8 We drew on both courses to build a lecture series focused on the business of dermatology that was delivered to dermatology residents at UMass Chan Medical School (Worcester, Massachusetts). Topic selection also was informed by qualitative input collected via email from recent graduates of the UMass dermatology residency program, focusing on the following areas: the US medical economy and health care costs; billing, coding, and claims processing; quality, relative value units (RVUs), reimbursement, and the merit-based incentive payment system; coverage of pharmaceuticals and teledermatology; and management. Residents were not required to prepare for any of the sessions; they were provided with handouts and slideshow presentations for reference to review at their convenience if desired. Five seminars were virtually conducted by an MD/MBA candidate at the institution (E.H.). They were recorded over the course of an academic year at 1- to 2-month intervals. Each 45-minute session was conducted in a lecture-discussion format and included case examples to help illustrate key principles and stimulate conversation. For example, the lecture on reimbursement incorporated a fee schedule calculation for a shave biopsy, using RVU and geographic pricing cost index (GCPI) multipliers. This demonstrated the variation in Centers for Medicare & Medicaid Services reimbursement in relation to (1) constituents of the RVU calculation (ie, work, practice expense, and malpractice) and (2) practice in a particular location (ie, the GCPI). Following this example, a conversation ensued among participants regarding the factors that drive valuation, with particular interest in variation based on urban vs suburban locations across the United States. Participants also found it of interest to examine the percentage of the valuation dedicated to each constituent and how features such as lesion size informed the final assessment of the charge. Another stylistic choice in developing the model was to include prompts for further consideration prior to transitioning topics in the lectures. For example: when examining the burden of skin disease, the audience was prompted to consider: “What is driving cost escalations, and how will services of the clinical domain meet these evolving needs?” At another point in the introductory lecture, residents were asked: “How do different types of insurance plans impact the management of patients with dermatologic concerns?” These questions were intended to transition residents to the next topic of discussion and highlight take-home points of consideration for medical practice. The project was reviewed by the UMass institutional review board and met criteria for exemption.
Residents who participated in at least 1 lecture (N=10) were surveyed after attendance; there were 7 responses (70% response rate). Residents were asked to rate a series of statements on a scale of 1 (strongly disagree) to 5 (strongly agree) and to provide commentary via an online form. Respondents indicated that the course was enjoyable (average score, 4.00), provided an appropriate level of detail (average score, 4.00), would be beneficial to integrate into a dermatology residency curriculum (average score, 3.86), and informed how they would practice as a clinician (average score, 3.86)(Figure). The respondents agreed that the course met the main goals of this initiative: it helped them develop knowledge about the interface between business and dermatology (4.14) and exposed residents to topics they had not learned about previously (4.71).

Although the course generally was well received, areas for improvement were identified from respondents’ comments, relating to audience engagement and refining the level of detail in the lectures. Recommendations included “less technical jargon and more focus on ‘big picture’ concepts, given audience’s low baseline knowledge”; “more case examples in each module”; and “more diagrams or interactive activities (polls, quizzes, break-out rooms) because the lectures were a bit dense.” This input was taken into consideration when revising the lectures for future use; they were reconstructed to have more case-based examples and prompts to encourage participation.
Resident commentary also demonstrated appreciation for education in this subject material. Statements such as “this is an important topic for future dermatologists” and “thank you so much for taking the time to implement this course” reflected the perceived value of this material during critical academic time. Another resident remarked: “This was great, thanks for putting it together.”
Given the positive experience of the residents and successful implementation of the series, this course was made available to all dermatology trainees on a network server with accompanying written documents. It is planned to be offered on a 3-year cycle in the future and will be updated to reflect inevitable changes in health care.
Although the relationship between business and medicine is increasingly important, teaching business principles has not become standardized or required in medical training. Despite the perception that this content is of value, implementation of programming has lagged behind that recognition, likely due to challenges in designing the curriculum and diffusing content into an already-saturated schedule. A model course that can be replicated in other residency programs would be valuable. We introduced a dermatology-specific lecture series to help prepare trainees for dermatology practice in a variety of clinical settings and train them with the language of business and operations that will equip them to respond to the needs of their patients, their practice, and the medical environment. Findings of this pilot study may not be generalizable to all dermatology residency programs because the sample size was small; the study was conducted at a single institution; and the content was delivered entirely online.
1. Tan S, Seiger K, Renehan P, et al. Trends in private equity acquisition of dermatology practices in the United States. JAMA Dermatol. 2019;155:1013-1021. doi:10.1001/jamadermatol.2019.1634
2. The business of health care in the United States. Harvard Online [Internet]. June 27, 2022. Accessed July 24, 2023. https://www.harvardonline.harvard.edu/blog/business-health-care-united-states
3. Ranpariya V, Cull D, Feldman SR, et al. Evaluation and management 2021 coding guidelines: key changes and implications. The Dermatologist. December 2020. Accessed July 24, 2023. https://www.hmpgloballearningnetwork.com/site/thederm/article/evaluation-and-management-2021-coding-guidelines-key-changes-and-implications?key=Ranpariya&elastic%5B0%5D=brand%3A73468
4. Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76:958-972.e2. doi:10.1016/j.jaad.2016.12.043
5. Resneck JS Jr. Dermatology practice consolidation fueled by private equity investment: potential consequences for the specialty and patients. JAMA Dermatol. 2018;154:13-14. doi:10.1001/jamadermatol.2017.5558
6. Patrinely JR Jr, Dewan AK. Business education in dermatology residency: a survey of program directors. Cutis. 2021;108:E7-E19. doi:10.12788/cutis.0331
7. Kohlwes RJ, Chou CL. A curriculum in medical economics for residents. Acad Med. 2002;77:465-466. doi:10.1097/00001888-200205000-00040
8. Fiebach NH, Rao D, Hamm ME. A curriculum in health systems and public health for internal medicine residents. Am J Prev Med. 2011;41(4 suppl 3):S264-S269. doi:10.1016/j.amepre.2011.05.025
1. Tan S, Seiger K, Renehan P, et al. Trends in private equity acquisition of dermatology practices in the United States. JAMA Dermatol. 2019;155:1013-1021. doi:10.1001/jamadermatol.2019.1634
2. The business of health care in the United States. Harvard Online [Internet]. June 27, 2022. Accessed July 24, 2023. https://www.harvardonline.harvard.edu/blog/business-health-care-united-states
3. Ranpariya V, Cull D, Feldman SR, et al. Evaluation and management 2021 coding guidelines: key changes and implications. The Dermatologist. December 2020. Accessed July 24, 2023. https://www.hmpgloballearningnetwork.com/site/thederm/article/evaluation-and-management-2021-coding-guidelines-key-changes-and-implications?key=Ranpariya&elastic%5B0%5D=brand%3A73468
4. Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76:958-972.e2. doi:10.1016/j.jaad.2016.12.043
5. Resneck JS Jr. Dermatology practice consolidation fueled by private equity investment: potential consequences for the specialty and patients. JAMA Dermatol. 2018;154:13-14. doi:10.1001/jamadermatol.2017.5558
6. Patrinely JR Jr, Dewan AK. Business education in dermatology residency: a survey of program directors. Cutis. 2021;108:E7-E19. doi:10.12788/cutis.0331
7. Kohlwes RJ, Chou CL. A curriculum in medical economics for residents. Acad Med. 2002;77:465-466. doi:10.1097/00001888-200205000-00040
8. Fiebach NH, Rao D, Hamm ME. A curriculum in health systems and public health for internal medicine residents. Am J Prev Med. 2011;41(4 suppl 3):S264-S269. doi:10.1016/j.amepre.2011.05.025
Practice Points
- Business education in dermatology residency promotes understanding of the health care ecosystem and can enable residents to more effectively deliver care that is responsive to the needs of their patients.
- Teaching fundamental business principles to residents can inform decision-making on patient, provider, and systems levels.
- A pilot curriculum supports implementation of business education teaching and will be particularly helpful in dermatology.
Stiff arteries may cause metabolic syndrome
New research published in the American Journal of Physiology found that arterial stiffness occurred before the presence of metabolic syndrome. A progressive rise in stiffness was associated with a cumulative increase in risk for the condition among the 3,862 people studied over a 7-year period starting in late adolescence.
Results revealed a notable sex difference: Arterial stiffness increased the risk for metabolic syndrome by 9% for males but only by 1% for females. Males were also five times more likely than females to have metabolic syndrome.
“It seems metabolic syndrome has a new risk factor we haven’t thought about,” said author Andrew O. Agbaje, MD, clinical epidemiologist and researcher, University of Eastern Finland, Kuopio.
Arterial stiffness previously was associated with metabolic syndrome in numerous studies. But the new work is the first to find evidence for causality, Dr. Agbaje said in an interview.
“Interventions have focused on addressing the components of metabolic syndrome such as obesity, dyslipidemia, hyperglycemia, and hypertension,” Dr. Agbaje said. “But arterial stiffness may independently cause metabolic syndrome in 1 out of 10 male teens. I encourage clinicians to think about its role in preventing and managing metabolic syndrome, not just as a consequence but as a cause.”
The results have important implications for physicians, according to Sissi Cossio, MD, pediatric endocrinologist, Pediatrix Medical Group, Fort Lauderdale, Fla.
“The fact that arterial stiffness progression preceded metabolic syndrome is important because it could be used as an earlier detection marker of disease,” Dr. Cossio said.
To conduct the study, Dr. Agbaje and his research team used data collected by the Avon Longitudinal Study of Parents and Children at the University of Bristol in England. Arterial stiffness was measured using carotid-femoral pulse wave velocity, the speed of blood flow from the upper to the lower aorta. They assessed for metabolic syndrome by the presence of three or more risk factors, including high cholesterol, high triglycerides, and high trunk fat mass.
Participants were studied starting in gestation in the early 1990s, and were measured for arterial stiffness and metabolic syndrome starting at age 17 through age 24.
The overall risk for metabolic syndrome doubled within the 7-year study period of follow-up between 2009 and 2017, indicating that early intervention during adolescence is essential.
Dr. Agbaje recommended that physicians start treating arterial stiffness and other markers of metabolic syndrome as early as possible, noting that, “potentially irreversible cardiovascular health damage might occur after age 17.”
Arterial stiffness can be negated through physical activity and dietary changes that lower inflammation. Physicians should refer at-risk teens to a preventative clinic where they can be monitored and receive repeated measurements of arterial stiffness, lipid levels, blood pressure, glucose levels, and obesity every 3 months, Dr. Agbaje said.
“The health progress made after a year would be an indicator for physicians whether a more aggressive therapeutic approach is needed since it takes about 7 years for the risk of metabolic syndrome attributed to arterial stiffness to worsen remarkably in the young population,” he said.
Dr. Agbaje pointed to a few potential pathways through which arterial stiffness might create a disease cascade. Stiffer arteries disrupt blood flow to the liver and pancreas, which could adversely affect their functioning, he said. Damage to these organs may increase insulin and LDL cholesterol blood levels, increasing the risk for metabolic syndrome.
Arterial stiffness also can lead to higher blood pressure and insulin resistance, potentially inducing musculogenesis and vasculogenesis. The resulting excessive muscle mass may also increase the risk for the condition, he said.
Dr. Cossio acknowledged that treatments for metabolic syndrome become less effective with age, but emphasized that reversal is possible in adults with lifestyle changes and medications.
“Early detection will give patients the best chance at reversing the disease, and [primary care physicians] are a key factor in this process,” she said.
Dr. Cossio said that at-risk teens should receive treatment in a weight loss or endocrinology clinic. Treatment may include behavioral, surgical, and pharmacotherapeutic interventions.
“Teens with signs of insulin resistance and impaired fasting glucose, acanthosis, or prediabetes, should start metformin as the first line of therapy,” Dr. Cossio said.
For weight management, she recommends antiobesity medications such as liraglutide, semaglutide, and the combination of phentermine/topiramate in children aged 12 years or older. In teenagers 16 years or older, phentermine alone is another option.
The research group that conducted the study reported received funding from the Jenny and Antti Wihuri Foundation, the North Savo Regional Fund and Central Finnish Cultural Foundation, the Aarne Koskelo Foundation, the Foundation for Pediatric Research, and the Finnish Foundation for Cardiovascular Research, among others. The authors declared no conflicts of interest, financial or otherwise.
A version of this article appeared on Medscape.com.
New research published in the American Journal of Physiology found that arterial stiffness occurred before the presence of metabolic syndrome. A progressive rise in stiffness was associated with a cumulative increase in risk for the condition among the 3,862 people studied over a 7-year period starting in late adolescence.
Results revealed a notable sex difference: Arterial stiffness increased the risk for metabolic syndrome by 9% for males but only by 1% for females. Males were also five times more likely than females to have metabolic syndrome.
“It seems metabolic syndrome has a new risk factor we haven’t thought about,” said author Andrew O. Agbaje, MD, clinical epidemiologist and researcher, University of Eastern Finland, Kuopio.
Arterial stiffness previously was associated with metabolic syndrome in numerous studies. But the new work is the first to find evidence for causality, Dr. Agbaje said in an interview.
“Interventions have focused on addressing the components of metabolic syndrome such as obesity, dyslipidemia, hyperglycemia, and hypertension,” Dr. Agbaje said. “But arterial stiffness may independently cause metabolic syndrome in 1 out of 10 male teens. I encourage clinicians to think about its role in preventing and managing metabolic syndrome, not just as a consequence but as a cause.”
The results have important implications for physicians, according to Sissi Cossio, MD, pediatric endocrinologist, Pediatrix Medical Group, Fort Lauderdale, Fla.
“The fact that arterial stiffness progression preceded metabolic syndrome is important because it could be used as an earlier detection marker of disease,” Dr. Cossio said.
To conduct the study, Dr. Agbaje and his research team used data collected by the Avon Longitudinal Study of Parents and Children at the University of Bristol in England. Arterial stiffness was measured using carotid-femoral pulse wave velocity, the speed of blood flow from the upper to the lower aorta. They assessed for metabolic syndrome by the presence of three or more risk factors, including high cholesterol, high triglycerides, and high trunk fat mass.
Participants were studied starting in gestation in the early 1990s, and were measured for arterial stiffness and metabolic syndrome starting at age 17 through age 24.
The overall risk for metabolic syndrome doubled within the 7-year study period of follow-up between 2009 and 2017, indicating that early intervention during adolescence is essential.
Dr. Agbaje recommended that physicians start treating arterial stiffness and other markers of metabolic syndrome as early as possible, noting that, “potentially irreversible cardiovascular health damage might occur after age 17.”
Arterial stiffness can be negated through physical activity and dietary changes that lower inflammation. Physicians should refer at-risk teens to a preventative clinic where they can be monitored and receive repeated measurements of arterial stiffness, lipid levels, blood pressure, glucose levels, and obesity every 3 months, Dr. Agbaje said.
“The health progress made after a year would be an indicator for physicians whether a more aggressive therapeutic approach is needed since it takes about 7 years for the risk of metabolic syndrome attributed to arterial stiffness to worsen remarkably in the young population,” he said.
Dr. Agbaje pointed to a few potential pathways through which arterial stiffness might create a disease cascade. Stiffer arteries disrupt blood flow to the liver and pancreas, which could adversely affect their functioning, he said. Damage to these organs may increase insulin and LDL cholesterol blood levels, increasing the risk for metabolic syndrome.
Arterial stiffness also can lead to higher blood pressure and insulin resistance, potentially inducing musculogenesis and vasculogenesis. The resulting excessive muscle mass may also increase the risk for the condition, he said.
Dr. Cossio acknowledged that treatments for metabolic syndrome become less effective with age, but emphasized that reversal is possible in adults with lifestyle changes and medications.
“Early detection will give patients the best chance at reversing the disease, and [primary care physicians] are a key factor in this process,” she said.
Dr. Cossio said that at-risk teens should receive treatment in a weight loss or endocrinology clinic. Treatment may include behavioral, surgical, and pharmacotherapeutic interventions.
“Teens with signs of insulin resistance and impaired fasting glucose, acanthosis, or prediabetes, should start metformin as the first line of therapy,” Dr. Cossio said.
For weight management, she recommends antiobesity medications such as liraglutide, semaglutide, and the combination of phentermine/topiramate in children aged 12 years or older. In teenagers 16 years or older, phentermine alone is another option.
The research group that conducted the study reported received funding from the Jenny and Antti Wihuri Foundation, the North Savo Regional Fund and Central Finnish Cultural Foundation, the Aarne Koskelo Foundation, the Foundation for Pediatric Research, and the Finnish Foundation for Cardiovascular Research, among others. The authors declared no conflicts of interest, financial or otherwise.
A version of this article appeared on Medscape.com.
New research published in the American Journal of Physiology found that arterial stiffness occurred before the presence of metabolic syndrome. A progressive rise in stiffness was associated with a cumulative increase in risk for the condition among the 3,862 people studied over a 7-year period starting in late adolescence.
Results revealed a notable sex difference: Arterial stiffness increased the risk for metabolic syndrome by 9% for males but only by 1% for females. Males were also five times more likely than females to have metabolic syndrome.
“It seems metabolic syndrome has a new risk factor we haven’t thought about,” said author Andrew O. Agbaje, MD, clinical epidemiologist and researcher, University of Eastern Finland, Kuopio.
Arterial stiffness previously was associated with metabolic syndrome in numerous studies. But the new work is the first to find evidence for causality, Dr. Agbaje said in an interview.
“Interventions have focused on addressing the components of metabolic syndrome such as obesity, dyslipidemia, hyperglycemia, and hypertension,” Dr. Agbaje said. “But arterial stiffness may independently cause metabolic syndrome in 1 out of 10 male teens. I encourage clinicians to think about its role in preventing and managing metabolic syndrome, not just as a consequence but as a cause.”
The results have important implications for physicians, according to Sissi Cossio, MD, pediatric endocrinologist, Pediatrix Medical Group, Fort Lauderdale, Fla.
“The fact that arterial stiffness progression preceded metabolic syndrome is important because it could be used as an earlier detection marker of disease,” Dr. Cossio said.
To conduct the study, Dr. Agbaje and his research team used data collected by the Avon Longitudinal Study of Parents and Children at the University of Bristol in England. Arterial stiffness was measured using carotid-femoral pulse wave velocity, the speed of blood flow from the upper to the lower aorta. They assessed for metabolic syndrome by the presence of three or more risk factors, including high cholesterol, high triglycerides, and high trunk fat mass.
Participants were studied starting in gestation in the early 1990s, and were measured for arterial stiffness and metabolic syndrome starting at age 17 through age 24.
The overall risk for metabolic syndrome doubled within the 7-year study period of follow-up between 2009 and 2017, indicating that early intervention during adolescence is essential.
Dr. Agbaje recommended that physicians start treating arterial stiffness and other markers of metabolic syndrome as early as possible, noting that, “potentially irreversible cardiovascular health damage might occur after age 17.”
Arterial stiffness can be negated through physical activity and dietary changes that lower inflammation. Physicians should refer at-risk teens to a preventative clinic where they can be monitored and receive repeated measurements of arterial stiffness, lipid levels, blood pressure, glucose levels, and obesity every 3 months, Dr. Agbaje said.
“The health progress made after a year would be an indicator for physicians whether a more aggressive therapeutic approach is needed since it takes about 7 years for the risk of metabolic syndrome attributed to arterial stiffness to worsen remarkably in the young population,” he said.
Dr. Agbaje pointed to a few potential pathways through which arterial stiffness might create a disease cascade. Stiffer arteries disrupt blood flow to the liver and pancreas, which could adversely affect their functioning, he said. Damage to these organs may increase insulin and LDL cholesterol blood levels, increasing the risk for metabolic syndrome.
Arterial stiffness also can lead to higher blood pressure and insulin resistance, potentially inducing musculogenesis and vasculogenesis. The resulting excessive muscle mass may also increase the risk for the condition, he said.
Dr. Cossio acknowledged that treatments for metabolic syndrome become less effective with age, but emphasized that reversal is possible in adults with lifestyle changes and medications.
“Early detection will give patients the best chance at reversing the disease, and [primary care physicians] are a key factor in this process,” she said.
Dr. Cossio said that at-risk teens should receive treatment in a weight loss or endocrinology clinic. Treatment may include behavioral, surgical, and pharmacotherapeutic interventions.
“Teens with signs of insulin resistance and impaired fasting glucose, acanthosis, or prediabetes, should start metformin as the first line of therapy,” Dr. Cossio said.
For weight management, she recommends antiobesity medications such as liraglutide, semaglutide, and the combination of phentermine/topiramate in children aged 12 years or older. In teenagers 16 years or older, phentermine alone is another option.
The research group that conducted the study reported received funding from the Jenny and Antti Wihuri Foundation, the North Savo Regional Fund and Central Finnish Cultural Foundation, the Aarne Koskelo Foundation, the Foundation for Pediatric Research, and the Finnish Foundation for Cardiovascular Research, among others. The authors declared no conflicts of interest, financial or otherwise.
A version of this article appeared on Medscape.com.
FROM AMERICAN JOURNAL OF PHYSIOLOGY
Just a simple country doctor
Whenever someone asks me what I do, I happily reply, “I’m just a simple country doctor.” That is, in part, why I am honored to be granted the opportunity to serve as editor-in-chief of The Journal of Family Practice (JFP). As our late colleague Dr. John Hickner noted in his first JFP editorial, he and the 2 editors-in-chief before him (Drs. Jeff Susman and Mark Ebell) were also of the small-town family doc tradition.1
My small-town roots trace back to rural South Carolina. I am a first-generation college student and attended medical school on a Navy Health Professions Scholarship. After completing my residency training, I had the privilege of serving for 5 years in the Navy (2 of those years were overseas), where I practiced and taught full-scope family medicine. I saw patients of all ages, attended deliveries, and provided inpatient hospital care, as well as performed a full range of procedures and tests, including colposcopies, skin procedures, vasectomies, flexible sigmoidoscopies, and exercise treadmill testing.
Following military service and completion of a 2-year fellowship and Master of Public Health degree (while working nights at a rural emergency department), I began work at the University of North Carolina at Chapel Hill. I had the good fortune of spending the next 11 years as a faculty member there, where I advanced my research and teaching career. In 2017, I was named the Chair of Family Medicine and Community Health at Duke University School of Medicine, where I continue to have an active outpatient practice.
My experiences have shaped my belief that it is critical that family medicine maintain its presence (and advance its prominence) both in our communities and at our large academic medicine centers, championing service to rural areas, promoting health equity, and advocating for the importance of high-quality primary care delivery and training. No matter where we are, our work is valuable, and we make a difference. Like my predecessors, I have a love of evidence-based medicine. I also have a love of writing, which I can trace back to my days as an intern. I am excited to be able to apply what I have learned over the years to help maintain the rigor, practicality, and relevance of JFP while simultaneously helping to nurture new authors and peer reviewers.
My goal as this journal’s editor-in-chief will be to continue its high academic standing while maintaining its utility for busy clinicians. The provision of evidence-based clinical review articles that are succinct and practical, along with departments (eg, Photo Rounds, Behavioral Health Consult, Practice Alert, PURLs), will remain the journal’s major focus. Within this framework, I also want to share the best evidence and ideas on other aspects of practicing medicine, such as quality improvement, population health, and health equity. I’ll be looking to increase recruitment and mentorship of authors from diverse backgrounds, including those historically underrepresented in medicine.
I look forward to working with the editorial board, associate and assistant editors, and staff of JFP to serve the diverse interests and needs of our readers. To that end, we’ll be looking for your guidance. How else can JFP help you in your day-to-day practice? Please let us know your ideas. Drop us a line at jfp.eic@mdedge.com.
Finally, please join me in thanking Drs. Henry Barry and Kate Rowland for all of their work this past year in keeping JFP going strong!
1. Hickner J. Meet JFP’s new editor-in-chief. J Fam Pract. 2012;61: 581.
Whenever someone asks me what I do, I happily reply, “I’m just a simple country doctor.” That is, in part, why I am honored to be granted the opportunity to serve as editor-in-chief of The Journal of Family Practice (JFP). As our late colleague Dr. John Hickner noted in his first JFP editorial, he and the 2 editors-in-chief before him (Drs. Jeff Susman and Mark Ebell) were also of the small-town family doc tradition.1
My small-town roots trace back to rural South Carolina. I am a first-generation college student and attended medical school on a Navy Health Professions Scholarship. After completing my residency training, I had the privilege of serving for 5 years in the Navy (2 of those years were overseas), where I practiced and taught full-scope family medicine. I saw patients of all ages, attended deliveries, and provided inpatient hospital care, as well as performed a full range of procedures and tests, including colposcopies, skin procedures, vasectomies, flexible sigmoidoscopies, and exercise treadmill testing.
Following military service and completion of a 2-year fellowship and Master of Public Health degree (while working nights at a rural emergency department), I began work at the University of North Carolina at Chapel Hill. I had the good fortune of spending the next 11 years as a faculty member there, where I advanced my research and teaching career. In 2017, I was named the Chair of Family Medicine and Community Health at Duke University School of Medicine, where I continue to have an active outpatient practice.
My experiences have shaped my belief that it is critical that family medicine maintain its presence (and advance its prominence) both in our communities and at our large academic medicine centers, championing service to rural areas, promoting health equity, and advocating for the importance of high-quality primary care delivery and training. No matter where we are, our work is valuable, and we make a difference. Like my predecessors, I have a love of evidence-based medicine. I also have a love of writing, which I can trace back to my days as an intern. I am excited to be able to apply what I have learned over the years to help maintain the rigor, practicality, and relevance of JFP while simultaneously helping to nurture new authors and peer reviewers.
My goal as this journal’s editor-in-chief will be to continue its high academic standing while maintaining its utility for busy clinicians. The provision of evidence-based clinical review articles that are succinct and practical, along with departments (eg, Photo Rounds, Behavioral Health Consult, Practice Alert, PURLs), will remain the journal’s major focus. Within this framework, I also want to share the best evidence and ideas on other aspects of practicing medicine, such as quality improvement, population health, and health equity. I’ll be looking to increase recruitment and mentorship of authors from diverse backgrounds, including those historically underrepresented in medicine.
I look forward to working with the editorial board, associate and assistant editors, and staff of JFP to serve the diverse interests and needs of our readers. To that end, we’ll be looking for your guidance. How else can JFP help you in your day-to-day practice? Please let us know your ideas. Drop us a line at jfp.eic@mdedge.com.
Finally, please join me in thanking Drs. Henry Barry and Kate Rowland for all of their work this past year in keeping JFP going strong!
Whenever someone asks me what I do, I happily reply, “I’m just a simple country doctor.” That is, in part, why I am honored to be granted the opportunity to serve as editor-in-chief of The Journal of Family Practice (JFP). As our late colleague Dr. John Hickner noted in his first JFP editorial, he and the 2 editors-in-chief before him (Drs. Jeff Susman and Mark Ebell) were also of the small-town family doc tradition.1
My small-town roots trace back to rural South Carolina. I am a first-generation college student and attended medical school on a Navy Health Professions Scholarship. After completing my residency training, I had the privilege of serving for 5 years in the Navy (2 of those years were overseas), where I practiced and taught full-scope family medicine. I saw patients of all ages, attended deliveries, and provided inpatient hospital care, as well as performed a full range of procedures and tests, including colposcopies, skin procedures, vasectomies, flexible sigmoidoscopies, and exercise treadmill testing.
Following military service and completion of a 2-year fellowship and Master of Public Health degree (while working nights at a rural emergency department), I began work at the University of North Carolina at Chapel Hill. I had the good fortune of spending the next 11 years as a faculty member there, where I advanced my research and teaching career. In 2017, I was named the Chair of Family Medicine and Community Health at Duke University School of Medicine, where I continue to have an active outpatient practice.
My experiences have shaped my belief that it is critical that family medicine maintain its presence (and advance its prominence) both in our communities and at our large academic medicine centers, championing service to rural areas, promoting health equity, and advocating for the importance of high-quality primary care delivery and training. No matter where we are, our work is valuable, and we make a difference. Like my predecessors, I have a love of evidence-based medicine. I also have a love of writing, which I can trace back to my days as an intern. I am excited to be able to apply what I have learned over the years to help maintain the rigor, practicality, and relevance of JFP while simultaneously helping to nurture new authors and peer reviewers.
My goal as this journal’s editor-in-chief will be to continue its high academic standing while maintaining its utility for busy clinicians. The provision of evidence-based clinical review articles that are succinct and practical, along with departments (eg, Photo Rounds, Behavioral Health Consult, Practice Alert, PURLs), will remain the journal’s major focus. Within this framework, I also want to share the best evidence and ideas on other aspects of practicing medicine, such as quality improvement, population health, and health equity. I’ll be looking to increase recruitment and mentorship of authors from diverse backgrounds, including those historically underrepresented in medicine.
I look forward to working with the editorial board, associate and assistant editors, and staff of JFP to serve the diverse interests and needs of our readers. To that end, we’ll be looking for your guidance. How else can JFP help you in your day-to-day practice? Please let us know your ideas. Drop us a line at jfp.eic@mdedge.com.
Finally, please join me in thanking Drs. Henry Barry and Kate Rowland for all of their work this past year in keeping JFP going strong!
1. Hickner J. Meet JFP’s new editor-in-chief. J Fam Pract. 2012;61: 581.
1. Hickner J. Meet JFP’s new editor-in-chief. J Fam Pract. 2012;61: 581.
Generalized maculopapular rash and fever
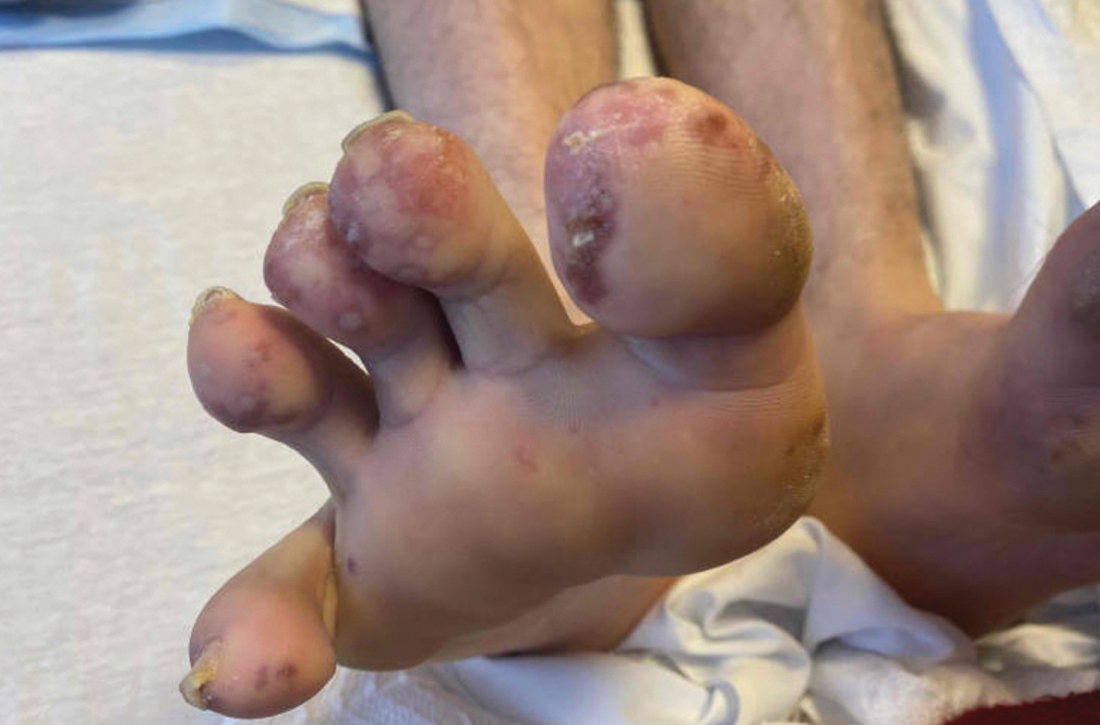
A 29-YEAR-OLD MAN was referred to the emergency department for fever and rash. Two months prior, he had noticed painful spots on his toes (FIGURE 1). Soon after, a rash of different morphology appeared on his chest and upper extremities. Associated symptoms included hair loss, generalized arthralgias, chills, trouble with balance, and photophobia. The patient denied genital lesions but reported 3 recent cold sores.
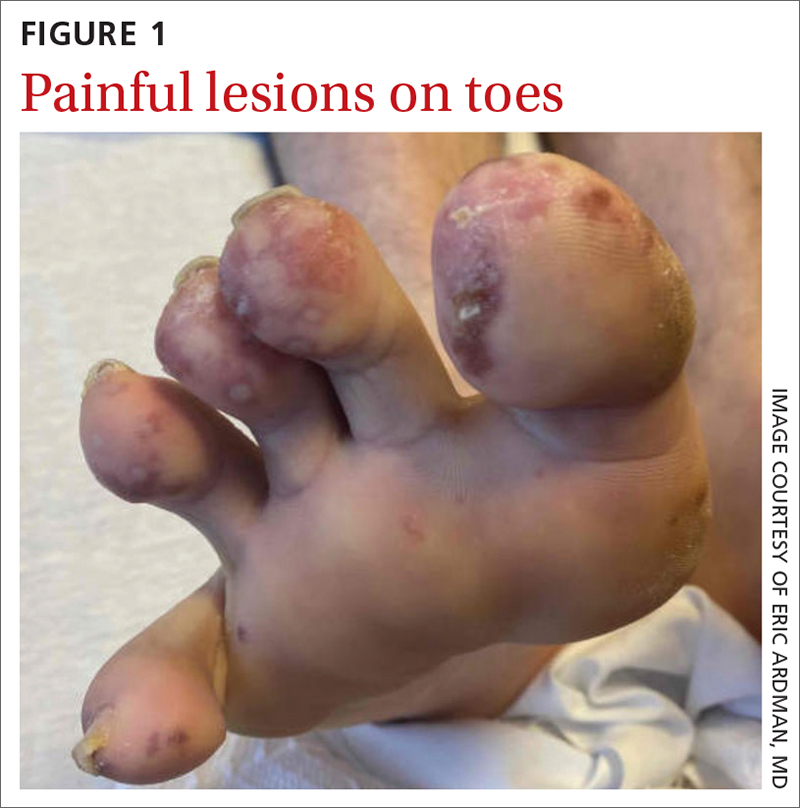
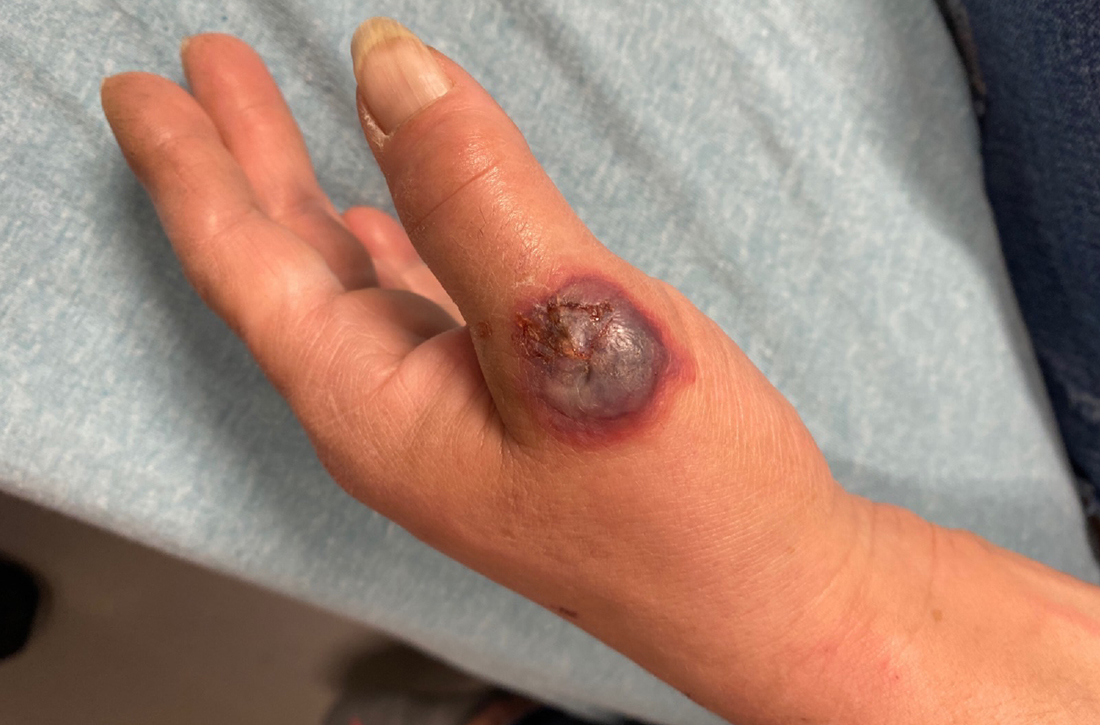
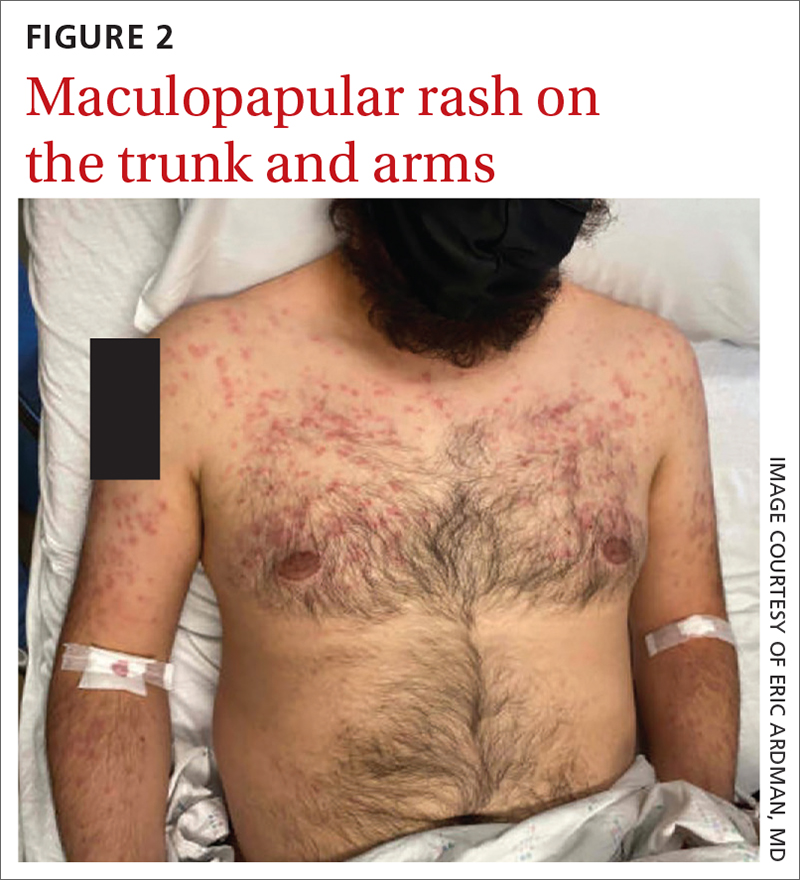
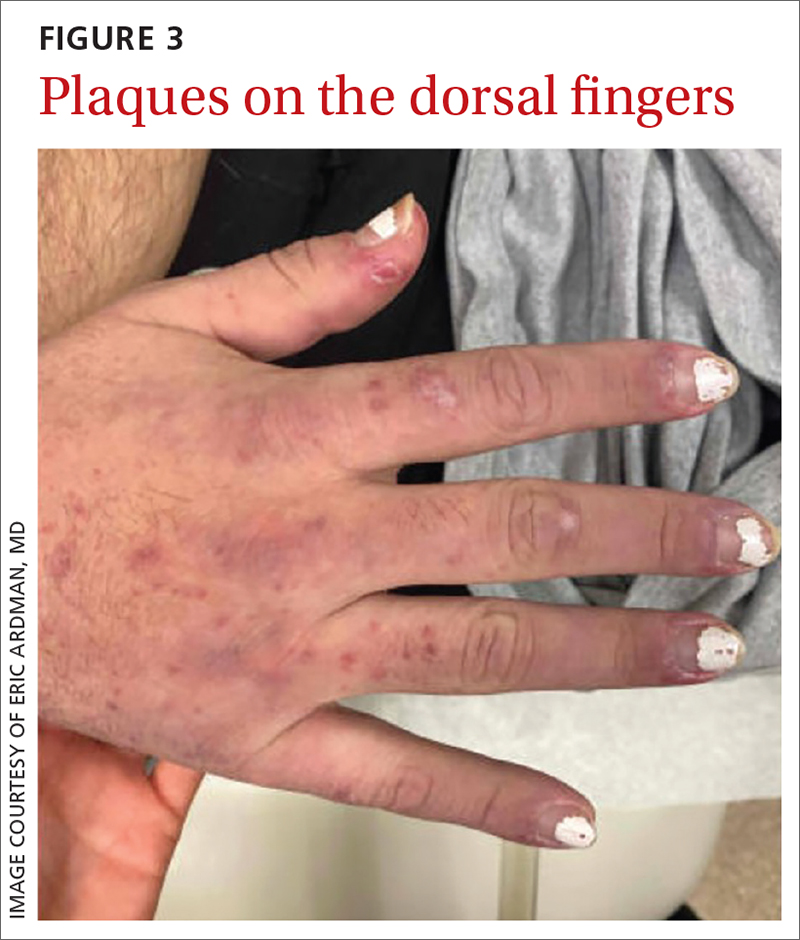
On exam, the patient was febrile (102 °F). Skin exam revealed a generalized maculopapular rash on the trunk, arms, and legs with scattered lesions on the palms (FIGURE 2). There were tender purpuric macules on the tips of his toes with areas of blanching. His hands had pink plaques on the dorsal fingers (FIGURE 3). Additionally, there was an erythematous papular rash with scale on his cheeks and nasal bridge, patchy areas of hair loss, and a single oral ulcer.
A neurologic exam was notable for mildly unstable gait, an abnormal Babinski reflex on the left side, and a positive Romberg sign. Musculoskeletal exam revealed joint tenderness in his shoulders, elbows, and wrists. Lymphadenopathy was present bilaterally in the axilla.
Lab work was ordered, including a VDRL test and a treponemal antibodies test. Skin biopsies also were taken from lesions on his arm and chest.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Systemic lupus erythematosus
Our patient’s fever and rash were highly suggestive of either systemic lupus erythematosus (SLE) or secondary syphilis (the “great masquerader”).
In addition to the joint tenderness revealed during the musculoskeletal exam, our patient had several nonspecific lupus findings on skin exam: malar rash, discoid rash on hands, subacute vasculitis (generalized rash), alopecia, and an oral ulcer; he also had the specific finding of chilblains vasculitis of the toes. Lab work and pathology results made the diagnosis clear. Lab work revealed leukopenia, an antinuclear antibody (ANA) result of 1:2560 with speckled appearance, and positive anti-SM antibodies. A dipstick was negative for protein; VDRL and treponemal antibodies tests were also negative. Histopathology showed perivascular lymph histiocytic vacuolar dermatitis with a differential of connective tissue disease, including lupus.
Our patient met the criteria
SLE is a systemic autoimmune disease resulting in chronic inflammation in multiple organ systems; it commonly manifests with vague symptoms of fatigue, fever, and weight loss. The prevalence of SLE in the United States has been reported as high as 241 per 100,000 people. 1 Women are more likely to be affected, and the incidence is highest among Black people and lowest among Caucasians.1,2 Risk factors include cigarette smoking and exposure to silica particulate air pollution.
The 2019 European League Against Rheumatism/American College of Rheumatology criteria for a diagnosis of SLE require that a patient have a positive ANA and some, but not all, additive lab, clinical, and organ-specific findings.3 Findings that clinicians should look for include3,4
- elevated ANA (≥ 1:80)
- constitutional symptoms (fever)
- hematologic findings (leukopenia, thrombocytopenia, autoimmune hemolysis)
- neuropsychiatric findings (delirium, psychosis, seizure)
- mucocutaneous findings (alopecia, oral ulcers, others)
- serosal findings (effusion, acute pericarditis)
- musculoskeletal findings (joint involvement)
- renal findings (proteinuria)
- antiphospholipid antibodies
- decreased complement proteins
- SLE-specific antibodies.
Dermatologic findings occur in more than 70% of patients with SLE.5 They can be nonspecific—eg, classic discoid rash, malar rash, alopecia, maculopapular rash (most commonly on sun-exposed areas, mimicking polymorphous light eruption)—or specific (eg, chilblains
Continue to: The differential for rash and fever is broad
The differential for rash and fever is broad
Syphilis also can manifest with rash and fever. The rash of syphilis is nonpainful and affects the torso and face, with concentration on the palms and soles.6,7
Dermatomyositis is a rare disorder of inflammation in both the skin and muscles. Symptoms include rash, muscle aches, and weakness. Lab abnormalities include elevated creatine kinase levels
Erythema multiforme is an immunologic-mediated rash consisting of firm targetoid erythematous papules distributed symmetrically on the extremities, including palms/soles. It typically appears after a viral infection, immunization, or new medications (eg, antibiotics, nonsteroidal anti-inflammatory drugs, or phenothiazines) initiated 1 to 3 weeks prior to the appearance of the rash. History and appearance inform the diagnosis.
Polymorphic light eruption is a rash of variable appearance on sun-exposed areas that results from a sensitivity to sunlight after lack of exposure for a period of time. Symptoms include burning and itching.
Treat patients with SLE with hydroxychloroquine (200-400 mg/d) to suppress inflammation and with low-dose oral steroids such as prednisone (7.5 mg/d) for intermittent exacerbations. Higher steroid doses are sometimes needed for signs of organ inflammation. Patients with increased disease activity will require immunosuppressive therapy with disease-modifying antirheumatic drugs,
Our patient was admitted for further evaluation. A lumbar puncture was performed because of his balance issues; it showed an elevated protein level, but further work-up did not find an infectious or malignant source. Balance improved with hydration. The patient remained hospitalized for 9 days, during which his fever subsided. His pain improved after initiation of hydroxychloroquine 400 mg/d. Follow-up with Rheumatology was arranged for further care.
1. Rees F, Doherty M, Grainge MJ, et al. The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford). 2017;56:1945-1961. doi: 10.1093/rheumatology/kex260
2. CDC. Systemic lupus erythematosus (SLE). Updated July 5, 2022. Accessed April 11, 2023. www.cdc.gov/lupus/facts/detailed.html
3. Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019;71:1400-1412. doi: 10.1002/art.40930
4. Lam NV, Brown JA, Sharma R. Systemic lupus erythematosus: diagnosis and treatment. Am Fam Physician. 2023;107:383-395.
5. Albrecht J, Berlin JA, Braverman IM, et al. Dermatology position paper on revision of the 1982 ACR criteria for systemic lupus erythematosus. Lupus. 2004;13:839-849. doi: 10.1191/0961203304lu2020oa
6. Dylewski J, Duong M. The rash of secondary syphilis. CMAJ. 2007;176:33-35. doi: 10.1503/cmaj.060665
7. Lautenschlager S. Cutaneous manifestations of syphilis: recognition and management. Am J Clin Dermatol. 2006;7:291-304. doi: 10.2165/00128071-200607050-00003:
8. Brown DL, Frank JE. Diagnosis and management of syphilis. Am Fam Physician. 2003;68:283-290.
9. Ricco J, Westby A. Syphilis: far from ancient history. Am Fam Physician. 2020;102:91-98.
A 29-YEAR-OLD MAN was referred to the emergency department for fever and rash. Two months prior, he had noticed painful spots on his toes (FIGURE 1). Soon after, a rash of different morphology appeared on his chest and upper extremities. Associated symptoms included hair loss, generalized arthralgias, chills, trouble with balance, and photophobia. The patient denied genital lesions but reported 3 recent cold sores.

On exam, the patient was febrile (102 °F). Skin exam revealed a generalized maculopapular rash on the trunk, arms, and legs with scattered lesions on the palms (FIGURE 2). There were tender purpuric macules on the tips of his toes with areas of blanching. His hands had pink plaques on the dorsal fingers (FIGURE 3). Additionally, there was an erythematous papular rash with scale on his cheeks and nasal bridge, patchy areas of hair loss, and a single oral ulcer.

A neurologic exam was notable for mildly unstable gait, an abnormal Babinski reflex on the left side, and a positive Romberg sign. Musculoskeletal exam revealed joint tenderness in his shoulders, elbows, and wrists. Lymphadenopathy was present bilaterally in the axilla.

Lab work was ordered, including a VDRL test and a treponemal antibodies test. Skin biopsies also were taken from lesions on his arm and chest.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Systemic lupus erythematosus
Our patient’s fever and rash were highly suggestive of either systemic lupus erythematosus (SLE) or secondary syphilis (the “great masquerader”).
In addition to the joint tenderness revealed during the musculoskeletal exam, our patient had several nonspecific lupus findings on skin exam: malar rash, discoid rash on hands, subacute vasculitis (generalized rash), alopecia, and an oral ulcer; he also had the specific finding of chilblains vasculitis of the toes. Lab work and pathology results made the diagnosis clear. Lab work revealed leukopenia, an antinuclear antibody (ANA) result of 1:2560 with speckled appearance, and positive anti-SM antibodies. A dipstick was negative for protein; VDRL and treponemal antibodies tests were also negative. Histopathology showed perivascular lymph histiocytic vacuolar dermatitis with a differential of connective tissue disease, including lupus.
Our patient met the criteria
SLE is a systemic autoimmune disease resulting in chronic inflammation in multiple organ systems; it commonly manifests with vague symptoms of fatigue, fever, and weight loss. The prevalence of SLE in the United States has been reported as high as 241 per 100,000 people. 1 Women are more likely to be affected, and the incidence is highest among Black people and lowest among Caucasians.1,2 Risk factors include cigarette smoking and exposure to silica particulate air pollution.
The 2019 European League Against Rheumatism/American College of Rheumatology criteria for a diagnosis of SLE require that a patient have a positive ANA and some, but not all, additive lab, clinical, and organ-specific findings.3 Findings that clinicians should look for include3,4
- elevated ANA (≥ 1:80)
- constitutional symptoms (fever)
- hematologic findings (leukopenia, thrombocytopenia, autoimmune hemolysis)
- neuropsychiatric findings (delirium, psychosis, seizure)
- mucocutaneous findings (alopecia, oral ulcers, others)
- serosal findings (effusion, acute pericarditis)
- musculoskeletal findings (joint involvement)
- renal findings (proteinuria)
- antiphospholipid antibodies
- decreased complement proteins
- SLE-specific antibodies.
Dermatologic findings occur in more than 70% of patients with SLE.5 They can be nonspecific—eg, classic discoid rash, malar rash, alopecia, maculopapular rash (most commonly on sun-exposed areas, mimicking polymorphous light eruption)—or specific (eg, chilblains
Continue to: The differential for rash and fever is broad
The differential for rash and fever is broad
Syphilis also can manifest with rash and fever. The rash of syphilis is nonpainful and affects the torso and face, with concentration on the palms and soles.6,7
Dermatomyositis is a rare disorder of inflammation in both the skin and muscles. Symptoms include rash, muscle aches, and weakness. Lab abnormalities include elevated creatine kinase levels
Erythema multiforme is an immunologic-mediated rash consisting of firm targetoid erythematous papules distributed symmetrically on the extremities, including palms/soles. It typically appears after a viral infection, immunization, or new medications (eg, antibiotics, nonsteroidal anti-inflammatory drugs, or phenothiazines) initiated 1 to 3 weeks prior to the appearance of the rash. History and appearance inform the diagnosis.
Polymorphic light eruption is a rash of variable appearance on sun-exposed areas that results from a sensitivity to sunlight after lack of exposure for a period of time. Symptoms include burning and itching.
Treat patients with SLE with hydroxychloroquine (200-400 mg/d) to suppress inflammation and with low-dose oral steroids such as prednisone (7.5 mg/d) for intermittent exacerbations. Higher steroid doses are sometimes needed for signs of organ inflammation. Patients with increased disease activity will require immunosuppressive therapy with disease-modifying antirheumatic drugs,
Our patient was admitted for further evaluation. A lumbar puncture was performed because of his balance issues; it showed an elevated protein level, but further work-up did not find an infectious or malignant source. Balance improved with hydration. The patient remained hospitalized for 9 days, during which his fever subsided. His pain improved after initiation of hydroxychloroquine 400 mg/d. Follow-up with Rheumatology was arranged for further care.
A 29-YEAR-OLD MAN was referred to the emergency department for fever and rash. Two months prior, he had noticed painful spots on his toes (FIGURE 1). Soon after, a rash of different morphology appeared on his chest and upper extremities. Associated symptoms included hair loss, generalized arthralgias, chills, trouble with balance, and photophobia. The patient denied genital lesions but reported 3 recent cold sores.

On exam, the patient was febrile (102 °F). Skin exam revealed a generalized maculopapular rash on the trunk, arms, and legs with scattered lesions on the palms (FIGURE 2). There were tender purpuric macules on the tips of his toes with areas of blanching. His hands had pink plaques on the dorsal fingers (FIGURE 3). Additionally, there was an erythematous papular rash with scale on his cheeks and nasal bridge, patchy areas of hair loss, and a single oral ulcer.

A neurologic exam was notable for mildly unstable gait, an abnormal Babinski reflex on the left side, and a positive Romberg sign. Musculoskeletal exam revealed joint tenderness in his shoulders, elbows, and wrists. Lymphadenopathy was present bilaterally in the axilla.

Lab work was ordered, including a VDRL test and a treponemal antibodies test. Skin biopsies also were taken from lesions on his arm and chest.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Systemic lupus erythematosus
Our patient’s fever and rash were highly suggestive of either systemic lupus erythematosus (SLE) or secondary syphilis (the “great masquerader”).
In addition to the joint tenderness revealed during the musculoskeletal exam, our patient had several nonspecific lupus findings on skin exam: malar rash, discoid rash on hands, subacute vasculitis (generalized rash), alopecia, and an oral ulcer; he also had the specific finding of chilblains vasculitis of the toes. Lab work and pathology results made the diagnosis clear. Lab work revealed leukopenia, an antinuclear antibody (ANA) result of 1:2560 with speckled appearance, and positive anti-SM antibodies. A dipstick was negative for protein; VDRL and treponemal antibodies tests were also negative. Histopathology showed perivascular lymph histiocytic vacuolar dermatitis with a differential of connective tissue disease, including lupus.
Our patient met the criteria
SLE is a systemic autoimmune disease resulting in chronic inflammation in multiple organ systems; it commonly manifests with vague symptoms of fatigue, fever, and weight loss. The prevalence of SLE in the United States has been reported as high as 241 per 100,000 people. 1 Women are more likely to be affected, and the incidence is highest among Black people and lowest among Caucasians.1,2 Risk factors include cigarette smoking and exposure to silica particulate air pollution.
The 2019 European League Against Rheumatism/American College of Rheumatology criteria for a diagnosis of SLE require that a patient have a positive ANA and some, but not all, additive lab, clinical, and organ-specific findings.3 Findings that clinicians should look for include3,4
- elevated ANA (≥ 1:80)
- constitutional symptoms (fever)
- hematologic findings (leukopenia, thrombocytopenia, autoimmune hemolysis)
- neuropsychiatric findings (delirium, psychosis, seizure)
- mucocutaneous findings (alopecia, oral ulcers, others)
- serosal findings (effusion, acute pericarditis)
- musculoskeletal findings (joint involvement)
- renal findings (proteinuria)
- antiphospholipid antibodies
- decreased complement proteins
- SLE-specific antibodies.
Dermatologic findings occur in more than 70% of patients with SLE.5 They can be nonspecific—eg, classic discoid rash, malar rash, alopecia, maculopapular rash (most commonly on sun-exposed areas, mimicking polymorphous light eruption)—or specific (eg, chilblains
Continue to: The differential for rash and fever is broad
The differential for rash and fever is broad
Syphilis also can manifest with rash and fever. The rash of syphilis is nonpainful and affects the torso and face, with concentration on the palms and soles.6,7
Dermatomyositis is a rare disorder of inflammation in both the skin and muscles. Symptoms include rash, muscle aches, and weakness. Lab abnormalities include elevated creatine kinase levels
Erythema multiforme is an immunologic-mediated rash consisting of firm targetoid erythematous papules distributed symmetrically on the extremities, including palms/soles. It typically appears after a viral infection, immunization, or new medications (eg, antibiotics, nonsteroidal anti-inflammatory drugs, or phenothiazines) initiated 1 to 3 weeks prior to the appearance of the rash. History and appearance inform the diagnosis.
Polymorphic light eruption is a rash of variable appearance on sun-exposed areas that results from a sensitivity to sunlight after lack of exposure for a period of time. Symptoms include burning and itching.
Treat patients with SLE with hydroxychloroquine (200-400 mg/d) to suppress inflammation and with low-dose oral steroids such as prednisone (7.5 mg/d) for intermittent exacerbations. Higher steroid doses are sometimes needed for signs of organ inflammation. Patients with increased disease activity will require immunosuppressive therapy with disease-modifying antirheumatic drugs,
Our patient was admitted for further evaluation. A lumbar puncture was performed because of his balance issues; it showed an elevated protein level, but further work-up did not find an infectious or malignant source. Balance improved with hydration. The patient remained hospitalized for 9 days, during which his fever subsided. His pain improved after initiation of hydroxychloroquine 400 mg/d. Follow-up with Rheumatology was arranged for further care.
1. Rees F, Doherty M, Grainge MJ, et al. The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford). 2017;56:1945-1961. doi: 10.1093/rheumatology/kex260
2. CDC. Systemic lupus erythematosus (SLE). Updated July 5, 2022. Accessed April 11, 2023. www.cdc.gov/lupus/facts/detailed.html
3. Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019;71:1400-1412. doi: 10.1002/art.40930
4. Lam NV, Brown JA, Sharma R. Systemic lupus erythematosus: diagnosis and treatment. Am Fam Physician. 2023;107:383-395.
5. Albrecht J, Berlin JA, Braverman IM, et al. Dermatology position paper on revision of the 1982 ACR criteria for systemic lupus erythematosus. Lupus. 2004;13:839-849. doi: 10.1191/0961203304lu2020oa
6. Dylewski J, Duong M. The rash of secondary syphilis. CMAJ. 2007;176:33-35. doi: 10.1503/cmaj.060665
7. Lautenschlager S. Cutaneous manifestations of syphilis: recognition and management. Am J Clin Dermatol. 2006;7:291-304. doi: 10.2165/00128071-200607050-00003:
8. Brown DL, Frank JE. Diagnosis and management of syphilis. Am Fam Physician. 2003;68:283-290.
9. Ricco J, Westby A. Syphilis: far from ancient history. Am Fam Physician. 2020;102:91-98.
1. Rees F, Doherty M, Grainge MJ, et al. The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford). 2017;56:1945-1961. doi: 10.1093/rheumatology/kex260
2. CDC. Systemic lupus erythematosus (SLE). Updated July 5, 2022. Accessed April 11, 2023. www.cdc.gov/lupus/facts/detailed.html
3. Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019;71:1400-1412. doi: 10.1002/art.40930
4. Lam NV, Brown JA, Sharma R. Systemic lupus erythematosus: diagnosis and treatment. Am Fam Physician. 2023;107:383-395.
5. Albrecht J, Berlin JA, Braverman IM, et al. Dermatology position paper on revision of the 1982 ACR criteria for systemic lupus erythematosus. Lupus. 2004;13:839-849. doi: 10.1191/0961203304lu2020oa
6. Dylewski J, Duong M. The rash of secondary syphilis. CMAJ. 2007;176:33-35. doi: 10.1503/cmaj.060665
7. Lautenschlager S. Cutaneous manifestations of syphilis: recognition and management. Am J Clin Dermatol. 2006;7:291-304. doi: 10.2165/00128071-200607050-00003:
8. Brown DL, Frank JE. Diagnosis and management of syphilis. Am Fam Physician. 2003;68:283-290.
9. Ricco J, Westby A. Syphilis: far from ancient history. Am Fam Physician. 2020;102:91-98.
Rapid-onset ulcerative hand nodule
A 55-YEAR-OLD WOMAN developed a small red papule on her left hand that, over the course of a week, progressed rapidly into an ulcerated nodule with accompanying swelling and pain. She reported concomitant fatigue, unintentional weight loss, and swollen axillary lymph nodes. Past medical history included rheumatoid arthritis.
A physical examination of her left hand revealed a tender, erythematous to violaceous nodule with ulceration and crust and surrounding diffuse erythema and edema (FIGURE). She also had several enlarged, nontender right axillary lymph nodes. Initial lab evaluation was significant for leukocytosis (13.8 K/uL) with increased neutrophils, lymphocytes, and eosinophils. Two punch biopsies were performed and the samples submitted for hematoxylin and eosin (H&E) staining and tissue culture.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Neutrophilic dermatosis of the dorsal hands
The results of H&E were consistent with neutrophilic dermatosis of the dorsal hands (NDDH). Tissue culture was negative for fungus, bacteria, and atypical mycobacteria, confirming the diagnosis.
NDDH is a neutrophilic dermatosis and considered a localized variant of Sweet syndrome, manifesting on the dorsal hands as suppurative, erythematous to violaceous papules, plaques, or nodules that often undergo necrosis, blistering, and ulceration. The diagnosis can be made clinically, although a biopsy is usually performed for confirmation. It is characterized histologically by a dense dermal neutrophilic infiltrate along with dermal edema.1
The pathogenesis of NDDH is not fully known.2 It is often preceded by trauma and may be associated with recent infection (respiratory, gastrointestinal), inflammatory bowel disease, autoimmune disease (eg, rheumatoid arthritis), or malignancy.1 The most common associated malignancies are hematologic, such as myelodysplastic syndrome, leukemia, or lymphoma, although solid tumors also can be seen.1,3 Therefore, patients who receive a diagnosis of NDDH typically require further work-up to rule out these associated conditions. NDDH is a rare enough entity that incidence/prevalence data aren’t available or likely to be accurate.
The differential includes infection and neoplastic processes
NDDH often is mistaken for an infectious abscess and unsuccessfully treated with antimicrobial agents, such as those commonly used for staphylococcus and streptococcus skin and soft-tissue infections. Thus, wound or tissue culture may be considered to exclude infection from the differential diagnosis. In addition to infectious processes such as sporotrichosis or an atypical mycobacterial infection, the differential includes other neutrophilic dermatoses and neoplastic processes such as lymphoma or leukemia cutis.
Sporotrichosis is caused by Sporothrix schenckii and usually spreads proximally after entering through a wound or cut. Special stains on histology and culture are needed to make the diagnosis.
Continue to: Atypical mycobacterial infections
Atypical mycobacterial infections usually enter through an area of trauma and spread proximally after inoculation. Atypical mycobacterial infections can be diagnosed via biopsy with special stains, culture, and polymerase chain reaction of the tissue.
Neutrophilic dermatoses are a broad category of dermatoses that include NDDH, pyoderma gangrenosum, and Sweet syndrome. This category of dermatoses is differentiated by morphology and distribution of lesions.
Lymphoma can be primary cutaneous or secondary to a systemic lymphoma. A biopsy will show a collection of atypical lymphocytes.
Treatment begins with steroids
Treatment with topical (eg, 0.05% clobetasol ointment bid), intralesional (10 to 40 mg/mL triamcinolone acetonide), or systemic (eg, prednisone 0.5 to 1 mg/kg tapered over the course of 1-2 months) steroids is considered first-line therapy and often results in rapid clinical improvement. Agents such as dapsone (25 to 150 mg/d) and/or colchicine (0.6 mg bid to tid) may be used in recalcitrant cases or in patients for whom steroids are contraindicated.2
Our patient’s NDDH was treated with prednisone (~1.0 mg/kg daily tapered over the course of 6 weeks). She was referred to Hematology/Oncology for further work-up of her constitutional symptoms, lymphadenopathy, and leukocytosis. Ultimately, she received a diagnosis of concomitant chronic lymphocytic leukemia/small lymphocytic lymphoma. The patient required no immediate treatment for her indolent lymphoma and was advised that she would need to get blood work done on a regular basis and have annual check-ups.
1. Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and Sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63.
2. Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 Cases. J Am Acad Dermatol. 2019;S0190-9622(19)32678-7. doi: 10.1016/j.jaad.2019.08.070
3. Mobini N, Sadrolashrafi K, Michaels S. Neutrophilic dermatosis of the dorsal hands: report of a case and review of the literature. Case Rep Dermatol Med. 2019;2019:8301585. doi: 10.1155/2019/8301585
A 55-YEAR-OLD WOMAN developed a small red papule on her left hand that, over the course of a week, progressed rapidly into an ulcerated nodule with accompanying swelling and pain. She reported concomitant fatigue, unintentional weight loss, and swollen axillary lymph nodes. Past medical history included rheumatoid arthritis.
A physical examination of her left hand revealed a tender, erythematous to violaceous nodule with ulceration and crust and surrounding diffuse erythema and edema (FIGURE). She also had several enlarged, nontender right axillary lymph nodes. Initial lab evaluation was significant for leukocytosis (13.8 K/uL) with increased neutrophils, lymphocytes, and eosinophils. Two punch biopsies were performed and the samples submitted for hematoxylin and eosin (H&E) staining and tissue culture.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Neutrophilic dermatosis of the dorsal hands
The results of H&E were consistent with neutrophilic dermatosis of the dorsal hands (NDDH). Tissue culture was negative for fungus, bacteria, and atypical mycobacteria, confirming the diagnosis.
NDDH is a neutrophilic dermatosis and considered a localized variant of Sweet syndrome, manifesting on the dorsal hands as suppurative, erythematous to violaceous papules, plaques, or nodules that often undergo necrosis, blistering, and ulceration. The diagnosis can be made clinically, although a biopsy is usually performed for confirmation. It is characterized histologically by a dense dermal neutrophilic infiltrate along with dermal edema.1
The pathogenesis of NDDH is not fully known.2 It is often preceded by trauma and may be associated with recent infection (respiratory, gastrointestinal), inflammatory bowel disease, autoimmune disease (eg, rheumatoid arthritis), or malignancy.1 The most common associated malignancies are hematologic, such as myelodysplastic syndrome, leukemia, or lymphoma, although solid tumors also can be seen.1,3 Therefore, patients who receive a diagnosis of NDDH typically require further work-up to rule out these associated conditions. NDDH is a rare enough entity that incidence/prevalence data aren’t available or likely to be accurate.
The differential includes infection and neoplastic processes
NDDH often is mistaken for an infectious abscess and unsuccessfully treated with antimicrobial agents, such as those commonly used for staphylococcus and streptococcus skin and soft-tissue infections. Thus, wound or tissue culture may be considered to exclude infection from the differential diagnosis. In addition to infectious processes such as sporotrichosis or an atypical mycobacterial infection, the differential includes other neutrophilic dermatoses and neoplastic processes such as lymphoma or leukemia cutis.
Sporotrichosis is caused by Sporothrix schenckii and usually spreads proximally after entering through a wound or cut. Special stains on histology and culture are needed to make the diagnosis.
Continue to: Atypical mycobacterial infections
Atypical mycobacterial infections usually enter through an area of trauma and spread proximally after inoculation. Atypical mycobacterial infections can be diagnosed via biopsy with special stains, culture, and polymerase chain reaction of the tissue.
Neutrophilic dermatoses are a broad category of dermatoses that include NDDH, pyoderma gangrenosum, and Sweet syndrome. This category of dermatoses is differentiated by morphology and distribution of lesions.
Lymphoma can be primary cutaneous or secondary to a systemic lymphoma. A biopsy will show a collection of atypical lymphocytes.
Treatment begins with steroids
Treatment with topical (eg, 0.05% clobetasol ointment bid), intralesional (10 to 40 mg/mL triamcinolone acetonide), or systemic (eg, prednisone 0.5 to 1 mg/kg tapered over the course of 1-2 months) steroids is considered first-line therapy and often results in rapid clinical improvement. Agents such as dapsone (25 to 150 mg/d) and/or colchicine (0.6 mg bid to tid) may be used in recalcitrant cases or in patients for whom steroids are contraindicated.2
Our patient’s NDDH was treated with prednisone (~1.0 mg/kg daily tapered over the course of 6 weeks). She was referred to Hematology/Oncology for further work-up of her constitutional symptoms, lymphadenopathy, and leukocytosis. Ultimately, she received a diagnosis of concomitant chronic lymphocytic leukemia/small lymphocytic lymphoma. The patient required no immediate treatment for her indolent lymphoma and was advised that she would need to get blood work done on a regular basis and have annual check-ups.
A 55-YEAR-OLD WOMAN developed a small red papule on her left hand that, over the course of a week, progressed rapidly into an ulcerated nodule with accompanying swelling and pain. She reported concomitant fatigue, unintentional weight loss, and swollen axillary lymph nodes. Past medical history included rheumatoid arthritis.
A physical examination of her left hand revealed a tender, erythematous to violaceous nodule with ulceration and crust and surrounding diffuse erythema and edema (FIGURE). She also had several enlarged, nontender right axillary lymph nodes. Initial lab evaluation was significant for leukocytosis (13.8 K/uL) with increased neutrophils, lymphocytes, and eosinophils. Two punch biopsies were performed and the samples submitted for hematoxylin and eosin (H&E) staining and tissue culture.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Neutrophilic dermatosis of the dorsal hands
The results of H&E were consistent with neutrophilic dermatosis of the dorsal hands (NDDH). Tissue culture was negative for fungus, bacteria, and atypical mycobacteria, confirming the diagnosis.
NDDH is a neutrophilic dermatosis and considered a localized variant of Sweet syndrome, manifesting on the dorsal hands as suppurative, erythematous to violaceous papules, plaques, or nodules that often undergo necrosis, blistering, and ulceration. The diagnosis can be made clinically, although a biopsy is usually performed for confirmation. It is characterized histologically by a dense dermal neutrophilic infiltrate along with dermal edema.1
The pathogenesis of NDDH is not fully known.2 It is often preceded by trauma and may be associated with recent infection (respiratory, gastrointestinal), inflammatory bowel disease, autoimmune disease (eg, rheumatoid arthritis), or malignancy.1 The most common associated malignancies are hematologic, such as myelodysplastic syndrome, leukemia, or lymphoma, although solid tumors also can be seen.1,3 Therefore, patients who receive a diagnosis of NDDH typically require further work-up to rule out these associated conditions. NDDH is a rare enough entity that incidence/prevalence data aren’t available or likely to be accurate.
The differential includes infection and neoplastic processes
NDDH often is mistaken for an infectious abscess and unsuccessfully treated with antimicrobial agents, such as those commonly used for staphylococcus and streptococcus skin and soft-tissue infections. Thus, wound or tissue culture may be considered to exclude infection from the differential diagnosis. In addition to infectious processes such as sporotrichosis or an atypical mycobacterial infection, the differential includes other neutrophilic dermatoses and neoplastic processes such as lymphoma or leukemia cutis.
Sporotrichosis is caused by Sporothrix schenckii and usually spreads proximally after entering through a wound or cut. Special stains on histology and culture are needed to make the diagnosis.
Continue to: Atypical mycobacterial infections
Atypical mycobacterial infections usually enter through an area of trauma and spread proximally after inoculation. Atypical mycobacterial infections can be diagnosed via biopsy with special stains, culture, and polymerase chain reaction of the tissue.
Neutrophilic dermatoses are a broad category of dermatoses that include NDDH, pyoderma gangrenosum, and Sweet syndrome. This category of dermatoses is differentiated by morphology and distribution of lesions.
Lymphoma can be primary cutaneous or secondary to a systemic lymphoma. A biopsy will show a collection of atypical lymphocytes.
Treatment begins with steroids
Treatment with topical (eg, 0.05% clobetasol ointment bid), intralesional (10 to 40 mg/mL triamcinolone acetonide), or systemic (eg, prednisone 0.5 to 1 mg/kg tapered over the course of 1-2 months) steroids is considered first-line therapy and often results in rapid clinical improvement. Agents such as dapsone (25 to 150 mg/d) and/or colchicine (0.6 mg bid to tid) may be used in recalcitrant cases or in patients for whom steroids are contraindicated.2
Our patient’s NDDH was treated with prednisone (~1.0 mg/kg daily tapered over the course of 6 weeks). She was referred to Hematology/Oncology for further work-up of her constitutional symptoms, lymphadenopathy, and leukocytosis. Ultimately, she received a diagnosis of concomitant chronic lymphocytic leukemia/small lymphocytic lymphoma. The patient required no immediate treatment for her indolent lymphoma and was advised that she would need to get blood work done on a regular basis and have annual check-ups.
1. Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and Sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63.
2. Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 Cases. J Am Acad Dermatol. 2019;S0190-9622(19)32678-7. doi: 10.1016/j.jaad.2019.08.070
3. Mobini N, Sadrolashrafi K, Michaels S. Neutrophilic dermatosis of the dorsal hands: report of a case and review of the literature. Case Rep Dermatol Med. 2019;2019:8301585. doi: 10.1155/2019/8301585
1. Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and Sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63.
2. Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 Cases. J Am Acad Dermatol. 2019;S0190-9622(19)32678-7. doi: 10.1016/j.jaad.2019.08.070
3. Mobini N, Sadrolashrafi K, Michaels S. Neutrophilic dermatosis of the dorsal hands: report of a case and review of the literature. Case Rep Dermatol Med. 2019;2019:8301585. doi: 10.1155/2019/8301585
Is injectable PrEP superior to oral therapy for HIV protection?
ILLUSTRATIVE CASE
A 24-year-old cisgender man with no significant past medical history comes to your office requesting PrEP after starting a new sexual relationship. His partner is a 26-year-old cisgender man with known HIV. The patient reports that balancing graduate school and work has made him very forgetful, and he worries that he won’t remember to take a daily pill. Are there any other PrEP methods you can offer?
The efficacy of PrEP to reduce HIV acquisition has been established across varying populations at high risk for transmission.1 PrEP has been found to reduce the risk for sexual acquisition of HIV by nearly 99%.2
Although the use of PrEP in the United States has increased steadily since 2012, adherence to an oral formulation remains a significant issue. One study of > 13,000 people found that daily oral PrEP was discontinued by 52% of participants, only 60% of whom reinitiated the therapy after discontinuation.2 Although the federal government has required Medicaid and other insurance providers to cover PrEP in an effort to increase access to the medication, this does not necessarily increase adherence to a daily medication in an often otherwise healthy population.
Long-acting injectable forms of PrEP, which have a reduced dosing frequency that may support adherence, have been studied to potentially replace daily oral pills. This latest study compared cabotegravir (CAB-LA), a long-acting IM injection given every 8 weeks, to daily oral PrEP with tenofovir disoproxil fumarate–emtricitabine (TDF-FTC).1
STUDY SUMMARY
Decreased seroconversion without daily pills
This randomized, double-blind, double-dummy, noninferiority trial compared long-acting injectable vs daily oral PrEP formulations for the prevention of HIV across an international population. Patients were randomized to receive either CAB-LA 600 mg IM every 8 weeks or TDF-FTC 300/200 mg orally daily. The double-dummy methodology meant that those patients receiving active CAB-LA also received a daily oral placebo, while those patients receiving active TDF-FTC also received a placebo injection every 8 weeks.
Study participants were cisgender MSM or transgender women who have sex with men; ages 18 years and older; and in good health but considered to be at high risk for HIV infection. To be included, participants had to have a negative HIV serologic test at enrollment, undetectable blood HIV RNA viral load within 14 days of enrollment, and creatinine clearance ≥ 60 mL/min. Exclusion criteria included intravenous (IV) drug use within 90 days of enrollment, coagulopathy, buttock implants or fillers, a seizure disorder, or a QTc interval > 500 ms.1
The intention-to-treat population included 4566 patients: 2282 in the CAB-LA group and 2284 in the TDF-FTC group. Demographic characteristics—including age, race, geographic region, and cohort (MSM vs transgender women)—were not significantly different between groups at baseline. The study lasted 153 weeks, and > 86% of patients were retained at 1 year (median follow-up, 1.4 years; interquartile range, 0.8-1.9).
Continue to: The primary efficacy and safety...
The primary efficacy and safety outcomes of interest were HIV infection and occurrence of a grade ≥ 2 adverse drug reaction, respectively. HIV seroconversion occurred in 13 of 2282 (0.57%) patients in the CAB-LA group and 39 of 2284 (1.7%) patients in the TDF-FTC group (hazard ratio = 0.34; 95% CI, 0.18-0.62). The rate of severe adverse drug reactions was similar between groups. The study was stopped early due to the superiority of CAB-LA.
WHAT’S NEW
Demonstrated superiority of injectable vs oral PrEP
The results of this study could have a monumental impact on the spread of HIV. Since adherence is a known limitation of daily oral PrEP, a long-acting injectable is an intriguing option. The 8-week period between injections offers convenience, allowing primary care physicians (PCPs) to schedule their patients in advance. And because every injection is administered in the office, this option would help PCPs track adherence. Witnessed adherence to the medication, and its demonstrated superiority, could have a significant effect on HIV prevention.
The limited serious adverse effects reported by both groups may ease some PCPs’ hesitation to prescribe CAB-LA.
CAVEATS
More injection-site reactions (but little impact on adherence)
Notably, 81.4% of patients in the CAB-LA group had injection-site reactions vs 31.3% in the TDF-FTC group. However, only 2.4% of patients in the CAB-LA group opted to stop receiving the injections because of these reactions.
Standard PrEP reduces the risk for HIV acquisition from IV drug use by 74%.2 However, because IV drug use was an exclusion criterion in this study, future research will need to assess CAB-LA’s effectiveness in that population.
CHALLENGES TO IMPLEMENTATION
Price and storage requirementsof CAB-LA may create issues
CAB-LA is expensive, costing more than $25,000 per year—significantly outpricing TDF-FTC, which costs approximately $8300 per year.3 Insurance coverage for PrEP, including CAB-LA, varies widely. Given the superiority reflected in this study, more efforts should be made to lower the cost of the medication.
Another hurdle for CAB-LA is that it requires refrigeration for storage. Although likely not an issue in most of the United States, it will make adoption of this method difficult in other parts of the world.
1. Landovitz RJ, Donnell D, Clement ME, et al; HPTN 083 Study Team. Cabotegravir for HIV prevention in cisgender men and transgender women. N Engl J Med. 2021;385:595-608. doi: 10.1056/NEJMoa2101016
2. Hojilla JC, Hurley LB, Marcus JL, et al. Characterization of HIV preexposure prophylaxis use behaviors and HIV incidence among US adults in an integrated health care system. JAMA Netw Open. 2021;4:e2122692. doi: 10.1001/jamanetworkopen.2021.22692
3. Neilan AM, Landovitz RJ, Le MH, et al. Cost-effectiveness of long-acting injectable HIV preexposure prophylaxis in the United States: a cost-effectiveness analysis. Ann Intern Med. 2022;175:479-489. doi: 10.7326/M21-1548
ILLUSTRATIVE CASE
A 24-year-old cisgender man with no significant past medical history comes to your office requesting PrEP after starting a new sexual relationship. His partner is a 26-year-old cisgender man with known HIV. The patient reports that balancing graduate school and work has made him very forgetful, and he worries that he won’t remember to take a daily pill. Are there any other PrEP methods you can offer?
The efficacy of PrEP to reduce HIV acquisition has been established across varying populations at high risk for transmission.1 PrEP has been found to reduce the risk for sexual acquisition of HIV by nearly 99%.2
Although the use of PrEP in the United States has increased steadily since 2012, adherence to an oral formulation remains a significant issue. One study of > 13,000 people found that daily oral PrEP was discontinued by 52% of participants, only 60% of whom reinitiated the therapy after discontinuation.2 Although the federal government has required Medicaid and other insurance providers to cover PrEP in an effort to increase access to the medication, this does not necessarily increase adherence to a daily medication in an often otherwise healthy population.
Long-acting injectable forms of PrEP, which have a reduced dosing frequency that may support adherence, have been studied to potentially replace daily oral pills. This latest study compared cabotegravir (CAB-LA), a long-acting IM injection given every 8 weeks, to daily oral PrEP with tenofovir disoproxil fumarate–emtricitabine (TDF-FTC).1
STUDY SUMMARY
Decreased seroconversion without daily pills
This randomized, double-blind, double-dummy, noninferiority trial compared long-acting injectable vs daily oral PrEP formulations for the prevention of HIV across an international population. Patients were randomized to receive either CAB-LA 600 mg IM every 8 weeks or TDF-FTC 300/200 mg orally daily. The double-dummy methodology meant that those patients receiving active CAB-LA also received a daily oral placebo, while those patients receiving active TDF-FTC also received a placebo injection every 8 weeks.
Study participants were cisgender MSM or transgender women who have sex with men; ages 18 years and older; and in good health but considered to be at high risk for HIV infection. To be included, participants had to have a negative HIV serologic test at enrollment, undetectable blood HIV RNA viral load within 14 days of enrollment, and creatinine clearance ≥ 60 mL/min. Exclusion criteria included intravenous (IV) drug use within 90 days of enrollment, coagulopathy, buttock implants or fillers, a seizure disorder, or a QTc interval > 500 ms.1
The intention-to-treat population included 4566 patients: 2282 in the CAB-LA group and 2284 in the TDF-FTC group. Demographic characteristics—including age, race, geographic region, and cohort (MSM vs transgender women)—were not significantly different between groups at baseline. The study lasted 153 weeks, and > 86% of patients were retained at 1 year (median follow-up, 1.4 years; interquartile range, 0.8-1.9).
Continue to: The primary efficacy and safety...
The primary efficacy and safety outcomes of interest were HIV infection and occurrence of a grade ≥ 2 adverse drug reaction, respectively. HIV seroconversion occurred in 13 of 2282 (0.57%) patients in the CAB-LA group and 39 of 2284 (1.7%) patients in the TDF-FTC group (hazard ratio = 0.34; 95% CI, 0.18-0.62). The rate of severe adverse drug reactions was similar between groups. The study was stopped early due to the superiority of CAB-LA.
WHAT’S NEW
Demonstrated superiority of injectable vs oral PrEP
The results of this study could have a monumental impact on the spread of HIV. Since adherence is a known limitation of daily oral PrEP, a long-acting injectable is an intriguing option. The 8-week period between injections offers convenience, allowing primary care physicians (PCPs) to schedule their patients in advance. And because every injection is administered in the office, this option would help PCPs track adherence. Witnessed adherence to the medication, and its demonstrated superiority, could have a significant effect on HIV prevention.
The limited serious adverse effects reported by both groups may ease some PCPs’ hesitation to prescribe CAB-LA.
CAVEATS
More injection-site reactions (but little impact on adherence)
Notably, 81.4% of patients in the CAB-LA group had injection-site reactions vs 31.3% in the TDF-FTC group. However, only 2.4% of patients in the CAB-LA group opted to stop receiving the injections because of these reactions.
Standard PrEP reduces the risk for HIV acquisition from IV drug use by 74%.2 However, because IV drug use was an exclusion criterion in this study, future research will need to assess CAB-LA’s effectiveness in that population.
CHALLENGES TO IMPLEMENTATION
Price and storage requirementsof CAB-LA may create issues
CAB-LA is expensive, costing more than $25,000 per year—significantly outpricing TDF-FTC, which costs approximately $8300 per year.3 Insurance coverage for PrEP, including CAB-LA, varies widely. Given the superiority reflected in this study, more efforts should be made to lower the cost of the medication.
Another hurdle for CAB-LA is that it requires refrigeration for storage. Although likely not an issue in most of the United States, it will make adoption of this method difficult in other parts of the world.
ILLUSTRATIVE CASE
A 24-year-old cisgender man with no significant past medical history comes to your office requesting PrEP after starting a new sexual relationship. His partner is a 26-year-old cisgender man with known HIV. The patient reports that balancing graduate school and work has made him very forgetful, and he worries that he won’t remember to take a daily pill. Are there any other PrEP methods you can offer?
The efficacy of PrEP to reduce HIV acquisition has been established across varying populations at high risk for transmission.1 PrEP has been found to reduce the risk for sexual acquisition of HIV by nearly 99%.2
Although the use of PrEP in the United States has increased steadily since 2012, adherence to an oral formulation remains a significant issue. One study of > 13,000 people found that daily oral PrEP was discontinued by 52% of participants, only 60% of whom reinitiated the therapy after discontinuation.2 Although the federal government has required Medicaid and other insurance providers to cover PrEP in an effort to increase access to the medication, this does not necessarily increase adherence to a daily medication in an often otherwise healthy population.
Long-acting injectable forms of PrEP, which have a reduced dosing frequency that may support adherence, have been studied to potentially replace daily oral pills. This latest study compared cabotegravir (CAB-LA), a long-acting IM injection given every 8 weeks, to daily oral PrEP with tenofovir disoproxil fumarate–emtricitabine (TDF-FTC).1
STUDY SUMMARY
Decreased seroconversion without daily pills
This randomized, double-blind, double-dummy, noninferiority trial compared long-acting injectable vs daily oral PrEP formulations for the prevention of HIV across an international population. Patients were randomized to receive either CAB-LA 600 mg IM every 8 weeks or TDF-FTC 300/200 mg orally daily. The double-dummy methodology meant that those patients receiving active CAB-LA also received a daily oral placebo, while those patients receiving active TDF-FTC also received a placebo injection every 8 weeks.
Study participants were cisgender MSM or transgender women who have sex with men; ages 18 years and older; and in good health but considered to be at high risk for HIV infection. To be included, participants had to have a negative HIV serologic test at enrollment, undetectable blood HIV RNA viral load within 14 days of enrollment, and creatinine clearance ≥ 60 mL/min. Exclusion criteria included intravenous (IV) drug use within 90 days of enrollment, coagulopathy, buttock implants or fillers, a seizure disorder, or a QTc interval > 500 ms.1
The intention-to-treat population included 4566 patients: 2282 in the CAB-LA group and 2284 in the TDF-FTC group. Demographic characteristics—including age, race, geographic region, and cohort (MSM vs transgender women)—were not significantly different between groups at baseline. The study lasted 153 weeks, and > 86% of patients were retained at 1 year (median follow-up, 1.4 years; interquartile range, 0.8-1.9).
Continue to: The primary efficacy and safety...
The primary efficacy and safety outcomes of interest were HIV infection and occurrence of a grade ≥ 2 adverse drug reaction, respectively. HIV seroconversion occurred in 13 of 2282 (0.57%) patients in the CAB-LA group and 39 of 2284 (1.7%) patients in the TDF-FTC group (hazard ratio = 0.34; 95% CI, 0.18-0.62). The rate of severe adverse drug reactions was similar between groups. The study was stopped early due to the superiority of CAB-LA.
WHAT’S NEW
Demonstrated superiority of injectable vs oral PrEP
The results of this study could have a monumental impact on the spread of HIV. Since adherence is a known limitation of daily oral PrEP, a long-acting injectable is an intriguing option. The 8-week period between injections offers convenience, allowing primary care physicians (PCPs) to schedule their patients in advance. And because every injection is administered in the office, this option would help PCPs track adherence. Witnessed adherence to the medication, and its demonstrated superiority, could have a significant effect on HIV prevention.
The limited serious adverse effects reported by both groups may ease some PCPs’ hesitation to prescribe CAB-LA.
CAVEATS
More injection-site reactions (but little impact on adherence)
Notably, 81.4% of patients in the CAB-LA group had injection-site reactions vs 31.3% in the TDF-FTC group. However, only 2.4% of patients in the CAB-LA group opted to stop receiving the injections because of these reactions.
Standard PrEP reduces the risk for HIV acquisition from IV drug use by 74%.2 However, because IV drug use was an exclusion criterion in this study, future research will need to assess CAB-LA’s effectiveness in that population.
CHALLENGES TO IMPLEMENTATION
Price and storage requirementsof CAB-LA may create issues
CAB-LA is expensive, costing more than $25,000 per year—significantly outpricing TDF-FTC, which costs approximately $8300 per year.3 Insurance coverage for PrEP, including CAB-LA, varies widely. Given the superiority reflected in this study, more efforts should be made to lower the cost of the medication.
Another hurdle for CAB-LA is that it requires refrigeration for storage. Although likely not an issue in most of the United States, it will make adoption of this method difficult in other parts of the world.
1. Landovitz RJ, Donnell D, Clement ME, et al; HPTN 083 Study Team. Cabotegravir for HIV prevention in cisgender men and transgender women. N Engl J Med. 2021;385:595-608. doi: 10.1056/NEJMoa2101016
2. Hojilla JC, Hurley LB, Marcus JL, et al. Characterization of HIV preexposure prophylaxis use behaviors and HIV incidence among US adults in an integrated health care system. JAMA Netw Open. 2021;4:e2122692. doi: 10.1001/jamanetworkopen.2021.22692
3. Neilan AM, Landovitz RJ, Le MH, et al. Cost-effectiveness of long-acting injectable HIV preexposure prophylaxis in the United States: a cost-effectiveness analysis. Ann Intern Med. 2022;175:479-489. doi: 10.7326/M21-1548
1. Landovitz RJ, Donnell D, Clement ME, et al; HPTN 083 Study Team. Cabotegravir for HIV prevention in cisgender men and transgender women. N Engl J Med. 2021;385:595-608. doi: 10.1056/NEJMoa2101016
2. Hojilla JC, Hurley LB, Marcus JL, et al. Characterization of HIV preexposure prophylaxis use behaviors and HIV incidence among US adults in an integrated health care system. JAMA Netw Open. 2021;4:e2122692. doi: 10.1001/jamanetworkopen.2021.22692
3. Neilan AM, Landovitz RJ, Le MH, et al. Cost-effectiveness of long-acting injectable HIV preexposure prophylaxis in the United States: a cost-effectiveness analysis. Ann Intern Med. 2022;175:479-489. doi: 10.7326/M21-1548
PRACTICE CHANGER
Consider intramuscular (IM) injectable cabotegravir every 8 weeks for HIV preexposure prophylaxis (PrEP) in cisgender men who have sex with men (MSM) and in transgender women.
STRENGTH OF RECOMMENDATION
B: Based on a single randomized controlled trial.1
Landovitz RJ, Donnell D, Clement ME, et al; HPTN 083 Study Team. Cabotegravir for HIV prevention in cisgender men and transgender women. N Engl J Med. 2021;385:595-608. doi: 10.1056/NEJMoa2101016
Prescribing DOACs with specific patient populations in mind
Four medications comprise the drug category known as direct oral anticoagulants (DOACs). Dabigatran (Pradaxa)1 was the first to gain approval. It was approved by the US Food and Drug Administration (FDA) in 2010 for the reduction of stroke and systemic embolism in patients with nonvalvular atrial fibrillation (AF). This was followed by approvals for rivaroxaban (Xarelto)2 in 2011, apixaban (Eliquis)3 in 2012, and edoxaban (Savaysa)4 in 2015. Betrixaban (Bevyxxa)5 was approved in 2017 for venous thromboembolism (VTE) prophylaxis in acutely ill hospitalized patients with restricted mobility, but it was removed from the market in 2020.

In addition to stroke prevention in nonvalvular AF, each DOAC has been approved for other indications and has been addressed further in guideline-based recommendations outside FDA-approved indications.
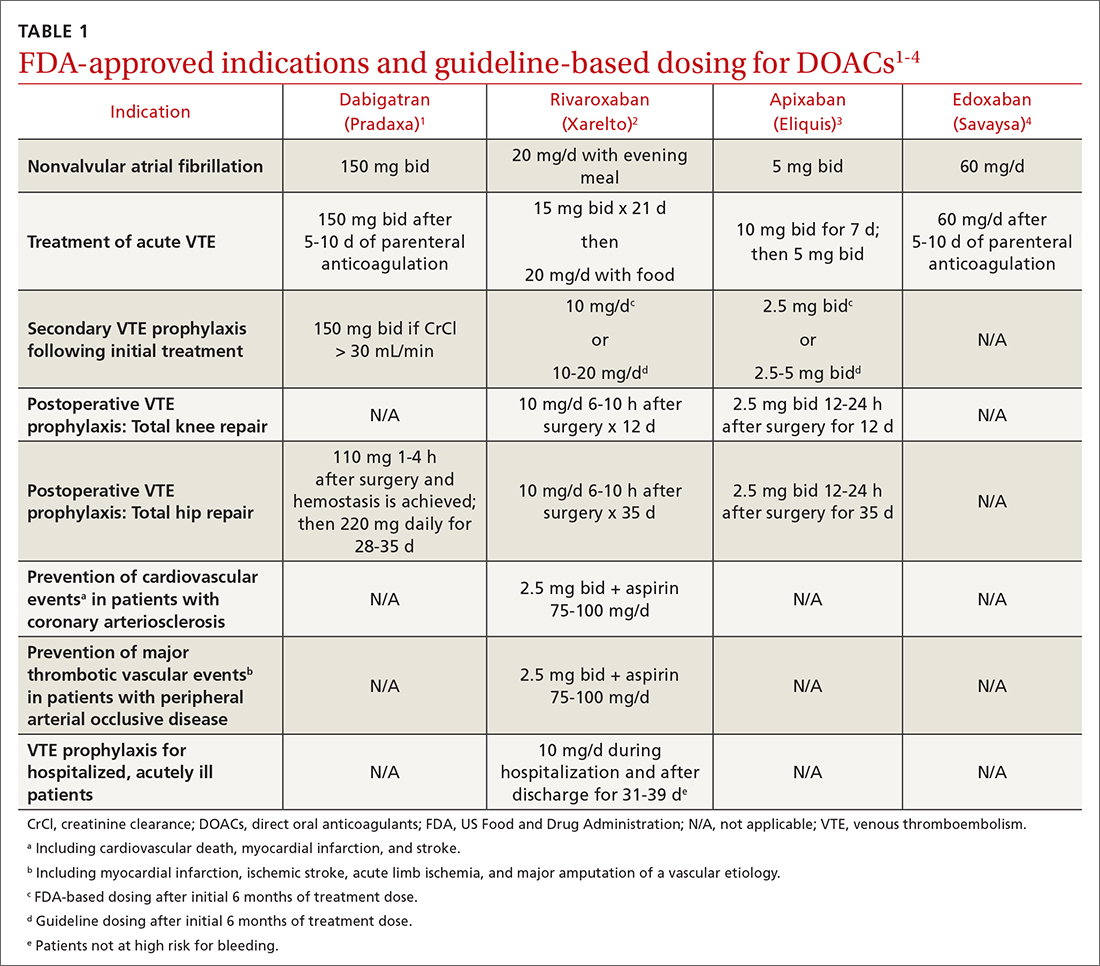
Overview of DOACs
Dabigatran is the only direct thrombin inhibitor; the other agents inhibit factor Xa. TABLE 11-4 summarizes FDA-approved indications and dosing and guideline-based dosing. Dabigatran and edoxaban require parenteral anticoagulation for 5 to 10 days prior to initiation for acute VTE, limiting their use.1,4 TABLE 21-4 highlights pharmacokinetic differences among the agents. For example, dabigatran is 80% renally cleared, is somewhat dialyzable, and can accumulate in patients with renal dysfunction.1 Edoxaban is contraindicated for nonvalvular AF in patients with a creatinine clearance (CrCl) > 95 mL/min because an increased stroke risk was demonstrated.4 Therefore, rivaroxaban and apixaban are prescribed most often in the United States.6,7
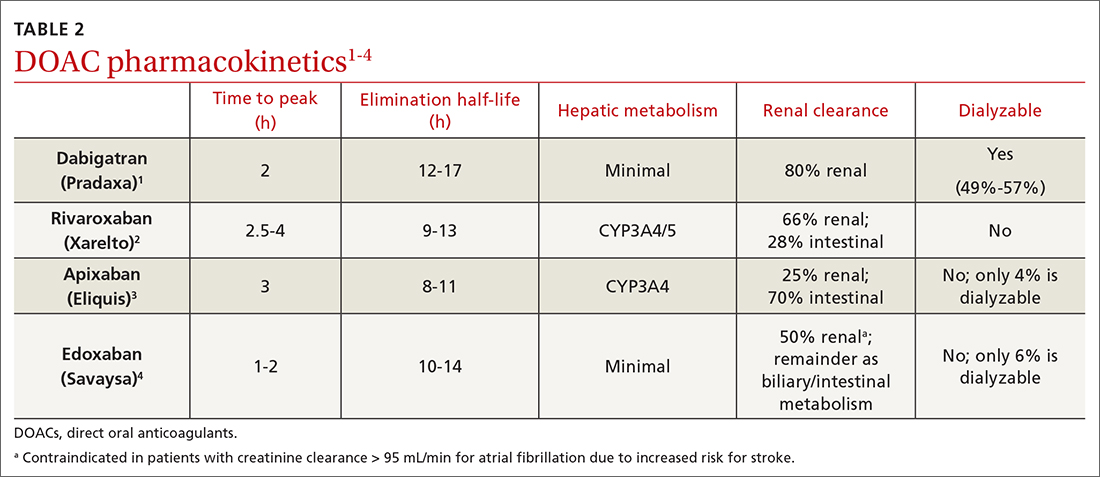
Applications in special patient populations
Obesity
As of 2020, more than 40% of adults in the United States were obese (body mass index [BMI] ≥ 30), with 9% classified as class 3 or severely obese (BMI ≥ 40).8 Altered drug pharmacokinetics in patients with severe obesity raises concern for undertreatment with fixed-dose DOACs. Phase III DOAC approval trials included patients with obesity, but weight cutoffs differed, making extrapolating efficacy and safety data difficult across different obesity stages.9 Although no FDA-labeled dosing adjustments exist for patients with obesity, the International Society on Thrombosis and Haemostasis (ISTH) does provide such recommendations.
ISTH changes position on measuring drug levels. ISTH previously recommended avoiding DOACs in those with a BMI > 40 or body weight > 120 kg. If a DOAC was used, ISTH advised obtaining peak and trough drug levels.10 However, DOAC drug levels have not been associated with clinical outcomes or sufficient degrees of anticoagulation.11
Men and women are affected equally by fibrolipomas. Prevalence does not differ by race or ethnicity.
In April 2021, ISTH updated guidance on DOACs in obesity, indicating standard doses of rivaroxaban or apixaban can be used for the treatment and prevention of VTE in all patients regardless of weight or BMI. Because data in obesity are lacking for dabigatran and edoxaban, avoid using these agents in patients with a BMI > 40 or weight > 120 kg. Additionally, assessing drug levels is no longer recommended, as there is insufficient evidence that these impact clinical outcomes.12
The 2021 American College of Chest Physicians (CHEST) guideline update
Continue to: Effectiveness of DOACs for AF in patients with obesity isn't clear
Effectiveness of DOACs for AF in patients with obesity isn’t clear, as most data are from retrospective cohort analyses. In patients weighing > 120 kg, dabigatran has shown efficacy in thrombosis prevention similar to that achieved in those weighing ≤ 120 kg, but it has increased the risk for gastrointestinal (GI) bleeding.15 Another study indicated a 15-mg dose of rivaroxaban may be associated with increased thromboembolic complications in patients with a BMI ≥ 35.16 Alternatively, another retrospective study of rivaroxaban demonstrated a small absolute risk reduction in ischemic stroke among patients in all stages of obesity and no difference in significant bleeding events.17 One further retrospective cohort showed that, in patients with a BMI ≥ 50 kg, the effectiveness of rivaroxaban and apixaban in thrombosis prevention and bleeding safety outcomes was comparable to that seen in those with a BMI < 30.18
As a result of conflicting data, and a lack of prospective randomized controlled trials (RCTs), ISTH continued recommending international normalized ratio (INR)–based dosing of warfarin for class 3 or severely obese patients with AF. The 2018 CHEST guidelines19 and the 2020 ESC guidelines20 make no mention of DOAC avoidance in patients with obesity and AF.
Advanced and end-stage renal disease
DOACs are renally dosed based on indication, drug-drug interactions, and degree of renal function (TABLE 31-4). For example, patients with AF who are anticoagulated with apixaban are prescribed 2.5 mg twice daily when 2 of the 3 following criteria are met: age ≥ 80 years, body weight ≤ 60 kg, serum creatinine ≥ 1.5 mg/dL. However, no dosage adjustment is necessary for VTE treatment or prophylaxis with apixaban regardless of renal function.3
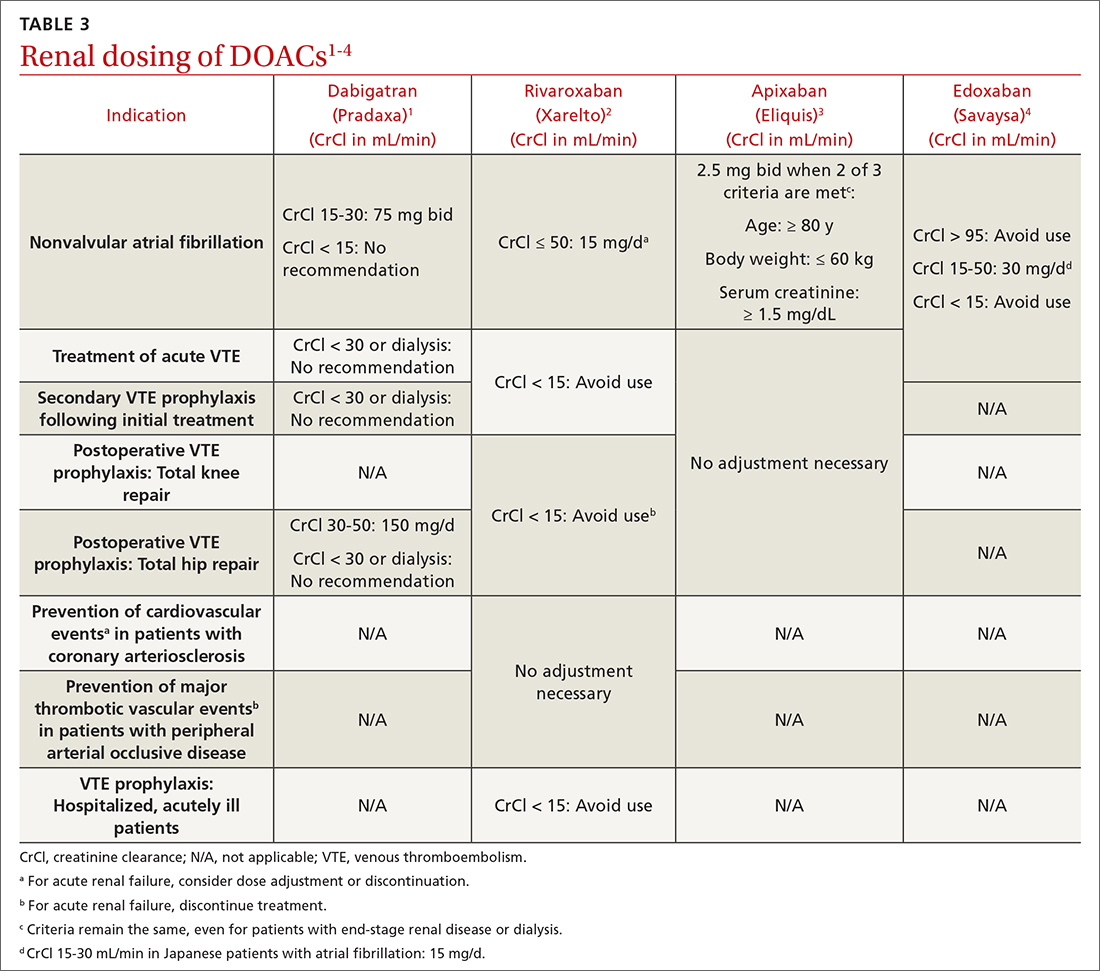
Data supporting the safety and efficacy of DOACs in end-stage renal disease (ESRD) are sparse. All DOACs are renally cleared to varying degrees (TABLE 21-4), theoretically increasing bleeding risk as kidney disease progresses. Apixaban is the least renally cleared of the DOACs and has been evaluated in the greatest number of trials for patients with ESRD for both VTE treatment and prevention and nonvalvular AF.21 As a result, the FDA approved standard-dose apixaban (5 mg twice daily) for VTE treatment and prevention and nonvalvular AF in patients with ESRD, even those requiring dialysis. Use the reduced apixaban dose (2.5 mg twice daily) in patients with ESRD and AF only if they are ≥ 80 years of age or their body weight is ≤ 60 kg.3
Patients with cancer
Cancer-associated acute VTE treatment. Cancer is an established risk factor for acute VTE but it also increases the risk for treatment-associated bleeding compared with patients without cancer.22 Historically, low-molecular-weight heparin (LMWH) was recommended over warfarin and DOACs for cancer-associated thromboses (CAT).23 Compared with warfarin, LMWH reduced the rate of recurrent VTE and had similar or reduced bleeding rates at 6 to 12 months.24-26 However, clinicians and patients often chose warfarin to avoid subcutaneous injections.27
CHEST guidelines recommend oral Xa inhibitors over LMWH for the treatment of CAT.13 The 2020 guidelines of the National Institute for Health and Care Excellence (NICE) recommend DOACs as an option for CAT along with LMWH or LMWH transitioned to warfarin.28 The American Society of Clinical Oncology (ASCO) recommends rivaroxaban for acute VTE treatment in CAT. No head-to-head trials have evaluated comparative efficacy of DOACs for CAT. However, edoxaban and rivaroxaban are associated with a greater risk for GI bleeding; therefore, apixaban is preferred in patients with GI malignancies.29 Standard DOAC VTE treatment dosing is recommended for all 3 agents.2-4
When using DOACs for patients with CAT, consider potential drug-drug interactions with chemotherapy regimens. All DOACs are transported by p-glycoprotein, while rivaroxaban and apixaban are substrates of cytochrome P450, leading to potentially significant drug-drug interactions.30 These interactions could affect the patient’s chemotherapeutic regimen, decrease the efficacy of the DOAC, or increase the risk for bleeding. Therefore, anticoagulation choice should be made in collaboration with the hematology/oncology team.
Continue to: Cancer-associated VTE prophylaxis...
Cancer-associated VTE prophylaxis. VTE prophylaxis for patients with cancer is complex and necessitates a global assessment of cancer location and treatment regimen and setting. Hospitalized patients receiving chemotherapy are at high risk for VTE if mobility is reduced or if other VTE risk factors are present. The International Initiative on Thrombosis and Cancer (ITAC)31 and ISTH32 recommend VTE prophylaxis with unfractionated heparin or LMWH (ISTH recommends LMWH more strongly). The 2020 ASCO Guidelines recommend pharmacologic anticoagulation but make no drug-specific recommendation.29 Parenteral treatment in hospitalized patients is not as burdensome as it is in ambulatory patients; therefore, these recommendations are less likely to elicit inpatient opposition.
In the ambulatory setting, patient avoidance of subcutaneous injections necessitates consideration of DOACs for CAT prophylaxis. The Khorana Risk Score (KRS) is a validated tool (scale, 0-7) to predict VTE risk in ambulatory patients receiving chemotherapy.33 KRS scores ≥ 2 indicate high thrombotic risk and the need for prophylactic anticoagulation. ASCO recommends apixaban, rivaroxaban, or LMWH.29 ISTH and ITAC both recommend apixaban or rivaroxaban over LMWH.31,34 An RCT published in June 2023 confirmed that, for adults with cancer and VTE, DOACs were noninferior to LMWH for preventing recurrent VTE for 6 months.35 The recommended doses for apixaban (2.5 mg twice daily) and rivaroxaban (10 mg daily) for CAT VTE prophylaxis are lower than FDA-approved treatment doses.31
Patients with thrombophilia: VTE prevention
Thrombophilias are broadly categorized as inherited or acquired, with inherited thrombophilia being more prevalent. The Factor V Leiden (FVL) variant affects 2% to 7% of the population, and prothrombin gene mutation (PGM) affects 1% to 2% of the population.36 Other forms of inherited thrombophilia, such as protein C deficiency, protein S deficiency, and antithrombin deficiency, occur less commonly (< 0.7% of the population).36 Antiphospholipid syndrome (APS), the most common acquired thrombophilia, affects approximately 2% of the population.36 APS involves multiple antibodies: anticardiolipin antibodies, lupus anticoagulant, and anti-beta-2 glycoprotein 1 antibodies. Establishing risk for thrombosis across the varying types of thrombophilia has proven difficult, but APS is considered the most thrombogenic thrombophilia apart from extremely rare homozygous inherited thrombophilias.36 Therefore, DOAC recommendations are thrombophilia specific.
A prospective cohort study evaluated DOACs compared with heparin/warfarin for VTE treatment in patients with inherited thrombophilias.37 Although all 4 available DOACs were included, most patients (61.1%) received rivaroxaban. Patients with an array of inherited thrombophilias, including rare homozygous mutations, were enrolled in this trial. While most patients (66.9%) had a “mild thrombophilia” defined as either FVL or PGM, the remainder had more severe thrombophilias.37 VTE recurrence was similar and uncommon in the DOAC and heparin/warfarin groups, consistent with a previous meta-analysis.38 Surprisingly, an increase in the cumulative risk for bleeding was seen in the DOAC group compared with the warfarin group, a finding inconsistent with prior trials.38 There were no major bleeding events in the DOAC group, but 3 such events occurred in the heparin/warfarin group, including 2 intracranial hemorrhages.
Currently NICE, CHEST, and ISTH do not make a recommendation for a preferred agent in patients with an acute VTE and inherited thrombophilia; however, DOACs would not be inappropriate.23,28,32 The American Society of Hematology (ASH) had planned to release recommendations related to the treatment of thrombophilia in 2020, but they were delayed by the COVID-19 pandemic.39
APS presents challenges for acute VTE anticoagulation. First, it causes a strongly thrombogenic state necessitating therapeutic anticoagulation. Second, for patients with positive lupus anticoagulant, INR monitoring and standardized INR goals may be inadequate.40 Therefore, using fixed-dose DOACs without the need for therapeutic monitoring is appealing, but significant concerns exist for using DOACs in patients with APS.41-45 ISTH and CHEST recommend warfarin for the treatment and prevention of acute VTE in patients with APS, especially those with triple-positive (anticardiolipin, lupus anticoagulant, and anti-beta-2 glycoprotein 1) APS.13,46 Package labeling for all DOACs recommends avoidance in triple-positive APS.1-4
ASTRO-APS is the most recent RCT to compare apixaban and warfarin for patients with APS,47 and it was terminated early after 6 of 23 patients in the apixaban group had thrombotic events, while no one in the warfarin group had such an event.48 Subsequently, a meta-analysis49 demonstrated that patients with thrombotic APS appear to have a greater risk for arterial thrombosis when treated with DOACs compared with warfarin. These 2 studies may lead to changes in recommendations to avoid DOACs in all patients with APS or may prompt more focused trials for DOAC use in patients with APS plus an antiplatelet to mitigate arterial thrombotic risk.
Continue to: Expanded clinical indications
Expanded clinical indications
Superficial vein thrombosis
Superficial thrombophlebitis or superficial vein thrombosis (SVT) is estimated to occur 6 times more frequently than VTE.50 Management of patients with isolated, uncomplicated thrombophlebitis who are at low risk for extension of the SVT involves symptomatic treatment with nonsteroidal anti-inflammatory drugs, topical agents, or compression therapy. However, depending on risk for progression, anticoagulation may be recommended.51
Patients at intermediate risk for extension or propagation of SVT are candidates for anticoagulation. The CHEST guidelines recommend
Certain situations should prompt one to consider using a treatment dose of a DOAC for 3 months. These include cases in which the SVT is located within 3 cm of the deep venous system, expands despite an appropriate prophylactic regimen, or recurs after discontinuation of prophylactic anticoagulation.13,50
Acute coronary syndrome
The American College of Cardiology/American Heart Association (ACC/AHA) recommend combination antiplatelet therapy and anticoagulation for management of acute coronary syndrome in hospitalized patients.52 Data are mixed regarding longer-term anticoagulation in addition to dual antiplatelet therapy in outpatient settings to prevent thrombosis recurrence in the absence of AF.
The APPRAISE-2 trial enrolled high-risk patients with ACS within 7 days of the event.53 Apixaban 5 mg twice daily was compared with placebo in patients taking aspirin or aspirin plus clopidogrel. The trial was terminated early because major bleeding events increased with apixaban without reduction in recurrent ischemic events. The ATLAS ACS-TIMI 46 trial evaluated different rivaroxaban doses (5-20 mg daily) in ACS patients.54 The study revealed possible thrombosis benefit but also increased risk for bleeding, particularly at higher doses. As a result, another study—ATLAS ACS 2-TIMI 51—was conducted and compared the use of low-dose rivaroxaban (2.5 mg twice daily or 5 mg twice daily) vs placebo for patients with recent ACS.55 All patients were receiving low-dose aspirin, and approximately 93% of patients in each group also were receiving clopidogrel or ticlopidine. As in the APPRAISE-2 trial, rivaroxaban increased the rate of major bleeding and intracranial hemorrhage; however, it did not increase the incidence of fatal bleeding. Unlike APPRAISE-2, rivaroxaban significantly reduced the primary efficacy end point, a composite of death from cardiovascular causes, myocardial infarction, or stroke (absolute risk reduction = 1.8%; number needed to treat = 56 for combined rivaroxaban doses).55
A secondary subgroup analysis combined data from the ATLAS ACMS-TIMI 46 and ATLAS ACS 2-TIMI 51 trials to evaluate outcomes in patients receiving aspirin monotherapy when combined with rivaroxaban 2.5 mg twice daily or 5 mg twice daily or with placebo.56 The primary efficacy end point was a composite of cardiovascular death, myocardial infarction, or stroke. When the 2 trials were evaluated separately, neither rivaroxaban dose was associated with reduction of the primary efficacy outcomes compared with aspirin alone. However, when the data were pooled, both the combined rivaroxaban doses (particularly the 5-mg dose) were associated with reduced cardiovascular outcomes. From a safety perspective, the 2.5-mg twice-daily dose of rivaroxaban was the only dose not associated with increased major bleeding risk. Thus, the 2.5-mg twice-daily dose of rivaroxaban may not provide sufficient cardiovascular benefit in patients with ACS, while the larger dose may increase the risk for nonfatal major bleeding events.56
The European Medicines Agency57 approved rivaroxaban 2.5 mg twice daily for ACS, and the 2020 ESC guidelines58 consider it an appropriate therapeutic option in addition to aspirin for patients at high ischemic risk and low bleeding risk. ACS is not an FDA-approved indication for DOACs, and the ACC/AHA Guideline for the Management of ACS, last updated in 2014, does not include DOACs for ACS unless patients have AF.52 Ongoing trials are further investigating rivaroxaban for ACS, so the use of DOACs in the post-acute phase of ACS may become clearer in the future.59
Continue to: Heparin-induced thrombocytopenia
Heparin-induced thrombocytopenia
Historically, nonheparin parenteral anticoagulants argatroban, bivalirudin, and fondaparinux were recommended for patients at risk for or who had heparin-induced thrombocytopenia (HIT). Argatroban is the only drug FDA approved for the treatment and prophylaxis of HIT; recommendations for the others are based on guideline recommendations.23,60,61 The nonheparin parenteral anticoagulants cost between $700 and $1500 per day; therefore most patients with HIT are transitioned to warfarin.62 However, protein C and S inhibition and a subsequent prothrombotic state conveyed by warfarin initiation necessitates a minimum 5-day bridge to therapeutic warfarin with a nonheparin parenteral anticoagulant.
In vitro tests show that DOACs do not promote development of HIT antibodies63 or affect platelet activation or aggregation.64 A literature summary of DOACs for HIT determined that in 104 patients, all but 1 achieved platelet recovery (defined as > 150,000/mcL) within a median time of 7 days. Therapeutically, DOACs prevented new or recurrent VTE in 102/104 cases (98%), and only 3% of patients experienced significant bleeding events.62
The 2018 ASH guidelines for VTE management in HIT include (with very low certainty of evidence) dabigatran, rivaroxaban, or apixaban for consideration in addition to previously recommended nonheparin parenteral anticoagulants.61 The dosing of each agent is contingent upon treatment of patients with HIT and an acute thrombosis (HITT) or HIT in the absence of VTE. For patients with HITT, treatment doses for acute VTE should be used for the appropriate duration of therapy (ie, 3 months). Importantly, dabigatran requires a 5-day pretreatment period with a parenteral anticoagulant, so it is not an ideal option. When treating isolated HIT (in the absence of VTE), ASH recommends all agents be dosed twice daily—dabigatran 150 mg twice daily (no 5-day parenteral pretreatment necessary), rivaroxaban 15 mg twice daily, or apixaban 5 mg twice daily—until platelet recovery (≥ 150,000/mcL) is achieved.61
CORRESPONDENCE
Kevin Schleich, PharmD, BCACP, Departments of Pharmaceutical Care and Family Medicine, University of Iowa, 200 Hawkins Drive, 01102-D PFP, Iowa City, IA, 52242; kevin-schleich@uiowa.edu
1. Dabigatran. Package Insert. Boehringer Ingelheim Pharmaceuticals, Inc.; 2021.
2. Rivaroxaban. Package insert. Janssen Pharmaceuticals, Inc; 2022.
3. Apixaban. Package insert. Bristol-Myers Squibb; 2021.
4. Edoxaban. Package insert. Daiichi Sankyo, Inc; 2015.
5. Betrixaban. Package insert. Portola Pharmaceuticals, Inc; 2017.
6. Wheelock KM, Ross JS, Murugiah K, et al. Clinician trends in prescribing direct oral anticoagulants for US Medicare beneficiaries. JAMA Netw Open. 2021;4:e2137288. doi: 10.1001/jamanetworkopen.2021.37288
7. Colacci M, Tseng EK, Sacks CA, et al. Oral anticoagulant utilization in the United States and United Kingdom. J Gen Intern Med. 2020;35:2505-2507. doi: 10.1007/s11606-020-05904-0
8. CDC. Adult obesity facts. Accessed May 9, 2023. www.cdc.gov/obesity/data/adult.html
9. Mocini D, Di Fusco SA, Mocini E, et al. Direct oral anticoagulants in patients with obesity and atrial fibrillation: position paper of Italian National Association of Hospital Cardiologists (ANMCO). J Clin Med. 2021;10:4185. doi: 10.3390/jcm10184185
10. Martin K, Beyer-Westendorf J, Davidson BL, et al. Use of the direct oral anticoagulants in obese patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14:1308-1313. doi: 10.1111/jth.13323
11. Gu TM, Garcia DA, Sabath DE. Assessment of direct oral anticoagulant assay use in clinical practice. J Thromb Thrombolysis. 2019;47:403-408. doi: 10.1007/s11239-018-1793-0
12. Martin KA, Beyer-Westendorf J, Davidson BL, et al. Use of direct oral anticoagulants in patients with obesity for treatment and prevention of venous thromboembolism: updated communication from the ISTH SSC Subcommittee on Control of Anticoagulation. J Thromb Haemost. 2021;19:1874-1882. doi: 10.1111/jth.15358
13. Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST Guideline and Expert Panel Report. Chest. 2021;160:e545-e608. doi: 10.1016/j.chest.2021.07.055
14. Konstantinides SV, Meyer G, Becattini C, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur Heart J. 2020;41:543-603. doi: 10.1093/eurheartj/ehz405
15. Coates J, Bitton E, Hendje A, et al. Clinical outcomes of dabigatran use in patients with non-valvular atrial fibrillation and weight >120 kg. Thromb Res. 2021;208:176-180. doi: 10.1016/j.thromres.2021.11.007
16. Li X, Zuo C, Ji Q, et al. Body mass index influence on the clinical outcomes for nonvalvular atrial fibrillation patients admitted to a hospital treated with direct oral anticoagulants: a retrospective cohort study. Drug Des Devel Ther. 2021;15:1931-1943. doi: 10.2147/dddt.S303219
17. Barakat AF, Jain S, Masri A, et al. Outcomes of direct oral anticoagulants in atrial fibrillation patients across different body mass index categories. JACC Clin Electrophysiol. 2021;7:649-658. doi: 10.1016/j.jacep.2021.02.002
18. O’Kane CP, Avalon JCO, Lacoste JL, et al. Apixaban and rivaroxaban use for atrial fibrillation in patients with obesity and BMI ≥50 kg/m2. Pharmacotherapy. 2022;42:112-118. doi: https://doi.org/10.1002/phar.2651
19. Lip GYH, Banerjee A, Boriani G, et al. Antithrombotic therapy for atrial fibrillation: CHEST Guideline and Expert Panel Report. Chest. 2018;154:1121-1201. doi: 10.1016/j.chest.2018.07.040
20. Sepehri Shamloo A, Dagres N, Hindricks G. [2020 ESC guidelines on atrial fibrillation: summary of the most relevant recommendations and innovations]. Herz. 2021;46:28-37. doi: 10.1007/s00059-020-05005-y
21. Chokesuwattanaskul R, Thongprayoon C, Tanawuttiwat T, et al. Safety and efficacy of apixaban versus warfarin in patients with end-stage renal disease: meta-analysis. Pacing Clin Electrophysiol. 2018;41:627-634. doi: 10.1111/pace.13331
22. Wang T-F, Li A, Garcia D. Managing thrombosis in cancer patients. Res Pract Thromb Haemost. 2018;2:429-438. doi: https://doi.org/10.1002/rth2.12102
23. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. CHEST. 2016;149:315-352. doi: 10.1016/j.chest.2015.11.026
24. Lee AY, Levine MN, Baker RI, et al. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349:146-153. doi: 10.1056/NEJMoa025313
25. Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med. 2002;162:1729-1735. doi: 10.1001/archinte.162.15.1729
26. Hull RD, Pineo GF, Brant RF, et al. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006;119:1062-1072. doi: 10.1016/j.amjmed.2006.02.022
27. Lee AYY, Kamphuisen PW, Meyer G, et al. Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 2015;314:677-686. doi: 10.1001/jama.2015.9243
28. NICE Guideline. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing. Accessed May 9, 2023. www.ncbi.nlm.nih.gov/books/NBK556698/
29. Key NS, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: ASCO Clinical Practice Guideline Update. J Clin Oncol. 2020;38:496-520. doi: 10.1200/jco.19.01461
30. Galgani A, Palleria C, Iannone LF, et al. Pharmacokinetic interactions of clinical interest between direct oral anticoagulants and antiepileptic drugs. Front Neurol. 2018;9:1067. doi: 10.3389/fneur.2018.01067
31. Farge D, Frere C, Connors JM, et al. 2019 International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. Lancet Oncol. 2019;20:e566-e581. doi: 10.1016/s1470-2045(19)30336-5
32. Di Nisio M, Carrier M, Lyman GH, et al. Prevention of venous thromboembolism in hospitalized medical cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2014;12:1746-1749. doi: 10.1111/jth.12683
33. Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902-4907. doi: 10.1182/blood-2007-10-116327
34. Wang TF, Zwicker JI, Ay C, et al. The use of direct oral anticoagulants for primary thromboprophylaxis in ambulatory cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2019;17:1772-1778. doi: 10.1111/jth.14564
35. Schrag D, Uno H, Rosovsky R, et al. Direct oral anticoagulants vs low-molecular-weight heparin and recurrent VTE in patients with cancer: a randomized clinical trial. JAMA. 2023;329:1924-1933. doi: 10.1001/jama.2023.7843
36. Stevens SM, Woller SC, Bauer KA, et al. Guidance for the evaluation and treatment of hereditary and acquired thrombophilia. J Thromb Thrombolysis. 2016;41:154-164. doi: 10.1007/s11239-015-1316-1
37. Campello E, Spiezia L, Simion C, et al. Direct oral anticoagulants in patients with inherited thrombophilia and venous thromboembolism: a prospective cohort study. J Am Heart Assoc. 2020;9:e018917. doi: 10.1161/jaha.120.018917
38. Elsebaie MAT, van Es N, Langston A, et al. Direct oral anticoagulants in patients with venous thromboembolism and thrombophilia: a systematic review and meta-analysis. J Thromb Haemost. 2019;17:645-656. doi: 10.1111/jth.14398
39. ASH. ASH Clinical Practice Guidelines on Venous Thromboembolism. Accessed May 10, 2023. www.hematology.org/education/clinicians/guidelines-and-quality-care/clinical-practice-guidelines/venous-thromboembolism-guidelines
40. Baquero-Salamanca M, Téllez-Arévalo AM, Calderon-Ospina C. Variability in the international normalised ratio (INR) in patients with antiphospholipid syndrome and positive lupus anticoagulant: should the INR targets be higher? BMJ Case Rep. 2015;2015:bcr2014209013. doi: 10.1136/bcr-2014-209013
41. Pengo V, Denas G, Zoppellaro G, et al. Rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome. Blood. 2018;132:1365-1371. doi: 10.1182/blood-2018-04-848333
42. Ordi-Ros J, Sáez-Comet L, Pérez-Conesa M, et al. Rivaroxaban versus vitamin K antagonist in antiphospholipid syndrome: a randomized noninferiority trial. Ann Intern Med. 2019;171:685-694. doi: 10.7326/m19-0291
43. Sato T, Nakamura H, Fujieda Y, et al. Factor Xa inhibitors for preventing recurrent thrombosis in patients with antiphospholipid syndrome: a longitudinal cohort study. Lupus. 2019;28:1577-1582. doi: 10.1177/0961203319881200
44. Malec K, Broniatowska E, Undas A. Direct oral anticoagulants in patients with antiphospholipid syndrome: a cohort study. Lupus. 2020;29:37-44. doi: 10.1177/0961203319889156
45. Rivaroxaban versus warfarin to treat patients with thrombotic antiphospholipid syndrome. Dr. Hannah Cohen about the results of the RAPS trial (Lancet Haematol 2016; 3: e426-36). Rheumatology (Oxford). 2017;56:e23. doi: 10.1093/rheumatology/kex290
46. Zuily S, Cohen H, Isenberg D, et al. Use of direct oral anticoagulants in patients with thrombotic antiphospholipid syndrome: guidance from the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. J Thromb Haemost. 2020;18:2126-2137. doi: https://doi.org/10.1111/jth.14935
47. NIH. ClinicalTrials.gov. Apixaban for the secondary prevention of thromboembolism among patients with antiphospholipid syndrome (ASTRO-APS). Accessed May 10, 2023. https://clinicaltrials.gov/ct2/show/NCT02295475?term=apixaban&cond=Anti+Phospholipid+Syndrome&draw=2&rank=1
48. Woller SC, Stevens SM, Kaplan D, et al. Apixaban compared with warfarin to prevent thrombosis in thrombotic antiphospholipid syndrome: a randomized trial. Blood Adv. 2022;6:1661-1670. doi: 10.1182/bloodadvances.2021005808
49. Khairani CD, Bejjani A, Piazza G, et al. Direct oral anticoagulants vs vitamin K antagonists in patients with antiphospholipid syndromes: meta-analysis of randomized trials. J Am Coll Cardiol. 2023;81:16-30. doi: 10.1016/j.jacc.2022.10.008
50. Superficial thrombophlebitis, superficial vein thrombosis. 2021. Accessed May 10, 2023. thrombosiscanada.ca/wp-content/uploads/2021/07/47.-Superficial-Vein-Thrombosis_16July2021.pdf
51. Di Nisio M, Wichers IM, Middeldorp S. Treatment for superficial thrombophlebitis of the leg. Cochrane Database Syst Rev. 2018;2:CD004982. doi: 10.1002/14651858.CD004982.pub6
52. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the Management of Patients With Non–ST-Elevation Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;64:e139-e228. doi: 10.1016/j.jacc.2014.09.017
53. Alexander JH, Lopes RD, James S, et al. Apixaban with antiplatelet therapy after acute coronary syndrome. N Engl J Med. 2011;365:699-708. doi: 10.1056/NEJMoa1105819
54. Mega JL, Braunwald E, Mohanavelu S, et al. Rivaroxaban versus placebo in patients with acute coronary syndromes (ATLAS ACS-TIMI 46): a randomised, double-blind, phase II trial. Lancet. 2009;374:29-38. doi: 10.1016/s0140-6736(09)60738-8
55. Mega JL, Braunwald E, Wiviott SD, et al. Rivaroxaban in patients with a recent acute coronary syndrome. N Engl J Med. 2012;366:9-19. doi: 10.1056/NEJMoa1112277
56. Gibson WJ, Gibson CM, Yee MK, et al. Safety and efficacy of rivaroxaban when added to aspirin monotherapy among stabilized post‐acute coronary syndrome patients: a pooled analysis study of ATLAS ACS‐TIMI 46 and ATLAS ACS 2‐TIMI 51. J Am Heart Assoc. 2019. Accessed May 10, 2023. Doi: 10.1161/JAHA.118.009451
57. European Medicines Agency. Xarelto (rivaroxaban). 2008. Accessed June 23, 2023.
58. Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42:1289-1367. doi: 10.1093/eurheartj/ehaa575
59. NIH. ClinicalTrials.gov. Accessed May 10, 2023. www.clinicaltrials.gov/ct2/results?cond=Acute+Coronary+Syndrome&term=rivaroxaban+&cntry=&state=&city=&dist=#
60. Watson H, Davidson S, Keeling D. Guidelines on the diagnosis and management of heparin-induced thrombocytopenia: second edition. Br J Haematol. 2012;159:528-40. doi: 10.1111/bjh.12059
61. Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv. 2018;2:3360-3392. doi: 10.1182/bloodadvances.2018024489
62. Momin J, Lee C-S. The role of direct oral anticoagulants in the management of heparin-induced thrombocytopenia US Pharmacist. 2020;45:3-10. Accessed May 10, 2023. www.uspharmacist.com/article/the-role-of-direct-oral-anticoagulants-in-the-management-of-heparininduced-thrombocytopenia
63. Warkentin TE, Pai M, Linkins LA. Direct oral anticoagulants for treatment of HIT: update of Hamilton experience and literature review. Blood. 2017;130:1104-1113. doi: 10.1182/blood-2017-04-778993
64. Krauel K, Hackbarth C, Fürll B, et al. Heparin-induced thrombocytopenia: in vitro studies on the interaction of dabigatran, rivaroxaban, and low-sulfated heparin, with platelet factor 4 and anti-PF4/heparin antibodies. Blood. 2012;119:1248-1255. doi: 10.1182/blood-2011-05-353391
Four medications comprise the drug category known as direct oral anticoagulants (DOACs). Dabigatran (Pradaxa)1 was the first to gain approval. It was approved by the US Food and Drug Administration (FDA) in 2010 for the reduction of stroke and systemic embolism in patients with nonvalvular atrial fibrillation (AF). This was followed by approvals for rivaroxaban (Xarelto)2 in 2011, apixaban (Eliquis)3 in 2012, and edoxaban (Savaysa)4 in 2015. Betrixaban (Bevyxxa)5 was approved in 2017 for venous thromboembolism (VTE) prophylaxis in acutely ill hospitalized patients with restricted mobility, but it was removed from the market in 2020.

In addition to stroke prevention in nonvalvular AF, each DOAC has been approved for other indications and has been addressed further in guideline-based recommendations outside FDA-approved indications.

Overview of DOACs
Dabigatran is the only direct thrombin inhibitor; the other agents inhibit factor Xa. TABLE 11-4 summarizes FDA-approved indications and dosing and guideline-based dosing. Dabigatran and edoxaban require parenteral anticoagulation for 5 to 10 days prior to initiation for acute VTE, limiting their use.1,4 TABLE 21-4 highlights pharmacokinetic differences among the agents. For example, dabigatran is 80% renally cleared, is somewhat dialyzable, and can accumulate in patients with renal dysfunction.1 Edoxaban is contraindicated for nonvalvular AF in patients with a creatinine clearance (CrCl) > 95 mL/min because an increased stroke risk was demonstrated.4 Therefore, rivaroxaban and apixaban are prescribed most often in the United States.6,7

Applications in special patient populations
Obesity
As of 2020, more than 40% of adults in the United States were obese (body mass index [BMI] ≥ 30), with 9% classified as class 3 or severely obese (BMI ≥ 40).8 Altered drug pharmacokinetics in patients with severe obesity raises concern for undertreatment with fixed-dose DOACs. Phase III DOAC approval trials included patients with obesity, but weight cutoffs differed, making extrapolating efficacy and safety data difficult across different obesity stages.9 Although no FDA-labeled dosing adjustments exist for patients with obesity, the International Society on Thrombosis and Haemostasis (ISTH) does provide such recommendations.
ISTH changes position on measuring drug levels. ISTH previously recommended avoiding DOACs in those with a BMI > 40 or body weight > 120 kg. If a DOAC was used, ISTH advised obtaining peak and trough drug levels.10 However, DOAC drug levels have not been associated with clinical outcomes or sufficient degrees of anticoagulation.11
Men and women are affected equally by fibrolipomas. Prevalence does not differ by race or ethnicity.
In April 2021, ISTH updated guidance on DOACs in obesity, indicating standard doses of rivaroxaban or apixaban can be used for the treatment and prevention of VTE in all patients regardless of weight or BMI. Because data in obesity are lacking for dabigatran and edoxaban, avoid using these agents in patients with a BMI > 40 or weight > 120 kg. Additionally, assessing drug levels is no longer recommended, as there is insufficient evidence that these impact clinical outcomes.12
The 2021 American College of Chest Physicians (CHEST) guideline update
Continue to: Effectiveness of DOACs for AF in patients with obesity isn't clear
Effectiveness of DOACs for AF in patients with obesity isn’t clear, as most data are from retrospective cohort analyses. In patients weighing > 120 kg, dabigatran has shown efficacy in thrombosis prevention similar to that achieved in those weighing ≤ 120 kg, but it has increased the risk for gastrointestinal (GI) bleeding.15 Another study indicated a 15-mg dose of rivaroxaban may be associated with increased thromboembolic complications in patients with a BMI ≥ 35.16 Alternatively, another retrospective study of rivaroxaban demonstrated a small absolute risk reduction in ischemic stroke among patients in all stages of obesity and no difference in significant bleeding events.17 One further retrospective cohort showed that, in patients with a BMI ≥ 50 kg, the effectiveness of rivaroxaban and apixaban in thrombosis prevention and bleeding safety outcomes was comparable to that seen in those with a BMI < 30.18
As a result of conflicting data, and a lack of prospective randomized controlled trials (RCTs), ISTH continued recommending international normalized ratio (INR)–based dosing of warfarin for class 3 or severely obese patients with AF. The 2018 CHEST guidelines19 and the 2020 ESC guidelines20 make no mention of DOAC avoidance in patients with obesity and AF.
Advanced and end-stage renal disease
DOACs are renally dosed based on indication, drug-drug interactions, and degree of renal function (TABLE 31-4). For example, patients with AF who are anticoagulated with apixaban are prescribed 2.5 mg twice daily when 2 of the 3 following criteria are met: age ≥ 80 years, body weight ≤ 60 kg, serum creatinine ≥ 1.5 mg/dL. However, no dosage adjustment is necessary for VTE treatment or prophylaxis with apixaban regardless of renal function.3

Data supporting the safety and efficacy of DOACs in end-stage renal disease (ESRD) are sparse. All DOACs are renally cleared to varying degrees (TABLE 21-4), theoretically increasing bleeding risk as kidney disease progresses. Apixaban is the least renally cleared of the DOACs and has been evaluated in the greatest number of trials for patients with ESRD for both VTE treatment and prevention and nonvalvular AF.21 As a result, the FDA approved standard-dose apixaban (5 mg twice daily) for VTE treatment and prevention and nonvalvular AF in patients with ESRD, even those requiring dialysis. Use the reduced apixaban dose (2.5 mg twice daily) in patients with ESRD and AF only if they are ≥ 80 years of age or their body weight is ≤ 60 kg.3
Patients with cancer
Cancer-associated acute VTE treatment. Cancer is an established risk factor for acute VTE but it also increases the risk for treatment-associated bleeding compared with patients without cancer.22 Historically, low-molecular-weight heparin (LMWH) was recommended over warfarin and DOACs for cancer-associated thromboses (CAT).23 Compared with warfarin, LMWH reduced the rate of recurrent VTE and had similar or reduced bleeding rates at 6 to 12 months.24-26 However, clinicians and patients often chose warfarin to avoid subcutaneous injections.27
CHEST guidelines recommend oral Xa inhibitors over LMWH for the treatment of CAT.13 The 2020 guidelines of the National Institute for Health and Care Excellence (NICE) recommend DOACs as an option for CAT along with LMWH or LMWH transitioned to warfarin.28 The American Society of Clinical Oncology (ASCO) recommends rivaroxaban for acute VTE treatment in CAT. No head-to-head trials have evaluated comparative efficacy of DOACs for CAT. However, edoxaban and rivaroxaban are associated with a greater risk for GI bleeding; therefore, apixaban is preferred in patients with GI malignancies.29 Standard DOAC VTE treatment dosing is recommended for all 3 agents.2-4
When using DOACs for patients with CAT, consider potential drug-drug interactions with chemotherapy regimens. All DOACs are transported by p-glycoprotein, while rivaroxaban and apixaban are substrates of cytochrome P450, leading to potentially significant drug-drug interactions.30 These interactions could affect the patient’s chemotherapeutic regimen, decrease the efficacy of the DOAC, or increase the risk for bleeding. Therefore, anticoagulation choice should be made in collaboration with the hematology/oncology team.
Continue to: Cancer-associated VTE prophylaxis...
Cancer-associated VTE prophylaxis. VTE prophylaxis for patients with cancer is complex and necessitates a global assessment of cancer location and treatment regimen and setting. Hospitalized patients receiving chemotherapy are at high risk for VTE if mobility is reduced or if other VTE risk factors are present. The International Initiative on Thrombosis and Cancer (ITAC)31 and ISTH32 recommend VTE prophylaxis with unfractionated heparin or LMWH (ISTH recommends LMWH more strongly). The 2020 ASCO Guidelines recommend pharmacologic anticoagulation but make no drug-specific recommendation.29 Parenteral treatment in hospitalized patients is not as burdensome as it is in ambulatory patients; therefore, these recommendations are less likely to elicit inpatient opposition.
In the ambulatory setting, patient avoidance of subcutaneous injections necessitates consideration of DOACs for CAT prophylaxis. The Khorana Risk Score (KRS) is a validated tool (scale, 0-7) to predict VTE risk in ambulatory patients receiving chemotherapy.33 KRS scores ≥ 2 indicate high thrombotic risk and the need for prophylactic anticoagulation. ASCO recommends apixaban, rivaroxaban, or LMWH.29 ISTH and ITAC both recommend apixaban or rivaroxaban over LMWH.31,34 An RCT published in June 2023 confirmed that, for adults with cancer and VTE, DOACs were noninferior to LMWH for preventing recurrent VTE for 6 months.35 The recommended doses for apixaban (2.5 mg twice daily) and rivaroxaban (10 mg daily) for CAT VTE prophylaxis are lower than FDA-approved treatment doses.31
Patients with thrombophilia: VTE prevention
Thrombophilias are broadly categorized as inherited or acquired, with inherited thrombophilia being more prevalent. The Factor V Leiden (FVL) variant affects 2% to 7% of the population, and prothrombin gene mutation (PGM) affects 1% to 2% of the population.36 Other forms of inherited thrombophilia, such as protein C deficiency, protein S deficiency, and antithrombin deficiency, occur less commonly (< 0.7% of the population).36 Antiphospholipid syndrome (APS), the most common acquired thrombophilia, affects approximately 2% of the population.36 APS involves multiple antibodies: anticardiolipin antibodies, lupus anticoagulant, and anti-beta-2 glycoprotein 1 antibodies. Establishing risk for thrombosis across the varying types of thrombophilia has proven difficult, but APS is considered the most thrombogenic thrombophilia apart from extremely rare homozygous inherited thrombophilias.36 Therefore, DOAC recommendations are thrombophilia specific.
A prospective cohort study evaluated DOACs compared with heparin/warfarin for VTE treatment in patients with inherited thrombophilias.37 Although all 4 available DOACs were included, most patients (61.1%) received rivaroxaban. Patients with an array of inherited thrombophilias, including rare homozygous mutations, were enrolled in this trial. While most patients (66.9%) had a “mild thrombophilia” defined as either FVL or PGM, the remainder had more severe thrombophilias.37 VTE recurrence was similar and uncommon in the DOAC and heparin/warfarin groups, consistent with a previous meta-analysis.38 Surprisingly, an increase in the cumulative risk for bleeding was seen in the DOAC group compared with the warfarin group, a finding inconsistent with prior trials.38 There were no major bleeding events in the DOAC group, but 3 such events occurred in the heparin/warfarin group, including 2 intracranial hemorrhages.
Currently NICE, CHEST, and ISTH do not make a recommendation for a preferred agent in patients with an acute VTE and inherited thrombophilia; however, DOACs would not be inappropriate.23,28,32 The American Society of Hematology (ASH) had planned to release recommendations related to the treatment of thrombophilia in 2020, but they were delayed by the COVID-19 pandemic.39
APS presents challenges for acute VTE anticoagulation. First, it causes a strongly thrombogenic state necessitating therapeutic anticoagulation. Second, for patients with positive lupus anticoagulant, INR monitoring and standardized INR goals may be inadequate.40 Therefore, using fixed-dose DOACs without the need for therapeutic monitoring is appealing, but significant concerns exist for using DOACs in patients with APS.41-45 ISTH and CHEST recommend warfarin for the treatment and prevention of acute VTE in patients with APS, especially those with triple-positive (anticardiolipin, lupus anticoagulant, and anti-beta-2 glycoprotein 1) APS.13,46 Package labeling for all DOACs recommends avoidance in triple-positive APS.1-4
ASTRO-APS is the most recent RCT to compare apixaban and warfarin for patients with APS,47 and it was terminated early after 6 of 23 patients in the apixaban group had thrombotic events, while no one in the warfarin group had such an event.48 Subsequently, a meta-analysis49 demonstrated that patients with thrombotic APS appear to have a greater risk for arterial thrombosis when treated with DOACs compared with warfarin. These 2 studies may lead to changes in recommendations to avoid DOACs in all patients with APS or may prompt more focused trials for DOAC use in patients with APS plus an antiplatelet to mitigate arterial thrombotic risk.
Continue to: Expanded clinical indications
Expanded clinical indications
Superficial vein thrombosis
Superficial thrombophlebitis or superficial vein thrombosis (SVT) is estimated to occur 6 times more frequently than VTE.50 Management of patients with isolated, uncomplicated thrombophlebitis who are at low risk for extension of the SVT involves symptomatic treatment with nonsteroidal anti-inflammatory drugs, topical agents, or compression therapy. However, depending on risk for progression, anticoagulation may be recommended.51
Patients at intermediate risk for extension or propagation of SVT are candidates for anticoagulation. The CHEST guidelines recommend
Certain situations should prompt one to consider using a treatment dose of a DOAC for 3 months. These include cases in which the SVT is located within 3 cm of the deep venous system, expands despite an appropriate prophylactic regimen, or recurs after discontinuation of prophylactic anticoagulation.13,50
Acute coronary syndrome
The American College of Cardiology/American Heart Association (ACC/AHA) recommend combination antiplatelet therapy and anticoagulation for management of acute coronary syndrome in hospitalized patients.52 Data are mixed regarding longer-term anticoagulation in addition to dual antiplatelet therapy in outpatient settings to prevent thrombosis recurrence in the absence of AF.
The APPRAISE-2 trial enrolled high-risk patients with ACS within 7 days of the event.53 Apixaban 5 mg twice daily was compared with placebo in patients taking aspirin or aspirin plus clopidogrel. The trial was terminated early because major bleeding events increased with apixaban without reduction in recurrent ischemic events. The ATLAS ACS-TIMI 46 trial evaluated different rivaroxaban doses (5-20 mg daily) in ACS patients.54 The study revealed possible thrombosis benefit but also increased risk for bleeding, particularly at higher doses. As a result, another study—ATLAS ACS 2-TIMI 51—was conducted and compared the use of low-dose rivaroxaban (2.5 mg twice daily or 5 mg twice daily) vs placebo for patients with recent ACS.55 All patients were receiving low-dose aspirin, and approximately 93% of patients in each group also were receiving clopidogrel or ticlopidine. As in the APPRAISE-2 trial, rivaroxaban increased the rate of major bleeding and intracranial hemorrhage; however, it did not increase the incidence of fatal bleeding. Unlike APPRAISE-2, rivaroxaban significantly reduced the primary efficacy end point, a composite of death from cardiovascular causes, myocardial infarction, or stroke (absolute risk reduction = 1.8%; number needed to treat = 56 for combined rivaroxaban doses).55
A secondary subgroup analysis combined data from the ATLAS ACMS-TIMI 46 and ATLAS ACS 2-TIMI 51 trials to evaluate outcomes in patients receiving aspirin monotherapy when combined with rivaroxaban 2.5 mg twice daily or 5 mg twice daily or with placebo.56 The primary efficacy end point was a composite of cardiovascular death, myocardial infarction, or stroke. When the 2 trials were evaluated separately, neither rivaroxaban dose was associated with reduction of the primary efficacy outcomes compared with aspirin alone. However, when the data were pooled, both the combined rivaroxaban doses (particularly the 5-mg dose) were associated with reduced cardiovascular outcomes. From a safety perspective, the 2.5-mg twice-daily dose of rivaroxaban was the only dose not associated with increased major bleeding risk. Thus, the 2.5-mg twice-daily dose of rivaroxaban may not provide sufficient cardiovascular benefit in patients with ACS, while the larger dose may increase the risk for nonfatal major bleeding events.56
The European Medicines Agency57 approved rivaroxaban 2.5 mg twice daily for ACS, and the 2020 ESC guidelines58 consider it an appropriate therapeutic option in addition to aspirin for patients at high ischemic risk and low bleeding risk. ACS is not an FDA-approved indication for DOACs, and the ACC/AHA Guideline for the Management of ACS, last updated in 2014, does not include DOACs for ACS unless patients have AF.52 Ongoing trials are further investigating rivaroxaban for ACS, so the use of DOACs in the post-acute phase of ACS may become clearer in the future.59
Continue to: Heparin-induced thrombocytopenia
Heparin-induced thrombocytopenia
Historically, nonheparin parenteral anticoagulants argatroban, bivalirudin, and fondaparinux were recommended for patients at risk for or who had heparin-induced thrombocytopenia (HIT). Argatroban is the only drug FDA approved for the treatment and prophylaxis of HIT; recommendations for the others are based on guideline recommendations.23,60,61 The nonheparin parenteral anticoagulants cost between $700 and $1500 per day; therefore most patients with HIT are transitioned to warfarin.62 However, protein C and S inhibition and a subsequent prothrombotic state conveyed by warfarin initiation necessitates a minimum 5-day bridge to therapeutic warfarin with a nonheparin parenteral anticoagulant.
In vitro tests show that DOACs do not promote development of HIT antibodies63 or affect platelet activation or aggregation.64 A literature summary of DOACs for HIT determined that in 104 patients, all but 1 achieved platelet recovery (defined as > 150,000/mcL) within a median time of 7 days. Therapeutically, DOACs prevented new or recurrent VTE in 102/104 cases (98%), and only 3% of patients experienced significant bleeding events.62
The 2018 ASH guidelines for VTE management in HIT include (with very low certainty of evidence) dabigatran, rivaroxaban, or apixaban for consideration in addition to previously recommended nonheparin parenteral anticoagulants.61 The dosing of each agent is contingent upon treatment of patients with HIT and an acute thrombosis (HITT) or HIT in the absence of VTE. For patients with HITT, treatment doses for acute VTE should be used for the appropriate duration of therapy (ie, 3 months). Importantly, dabigatran requires a 5-day pretreatment period with a parenteral anticoagulant, so it is not an ideal option. When treating isolated HIT (in the absence of VTE), ASH recommends all agents be dosed twice daily—dabigatran 150 mg twice daily (no 5-day parenteral pretreatment necessary), rivaroxaban 15 mg twice daily, or apixaban 5 mg twice daily—until platelet recovery (≥ 150,000/mcL) is achieved.61
CORRESPONDENCE
Kevin Schleich, PharmD, BCACP, Departments of Pharmaceutical Care and Family Medicine, University of Iowa, 200 Hawkins Drive, 01102-D PFP, Iowa City, IA, 52242; kevin-schleich@uiowa.edu
Four medications comprise the drug category known as direct oral anticoagulants (DOACs). Dabigatran (Pradaxa)1 was the first to gain approval. It was approved by the US Food and Drug Administration (FDA) in 2010 for the reduction of stroke and systemic embolism in patients with nonvalvular atrial fibrillation (AF). This was followed by approvals for rivaroxaban (Xarelto)2 in 2011, apixaban (Eliquis)3 in 2012, and edoxaban (Savaysa)4 in 2015. Betrixaban (Bevyxxa)5 was approved in 2017 for venous thromboembolism (VTE) prophylaxis in acutely ill hospitalized patients with restricted mobility, but it was removed from the market in 2020.

In addition to stroke prevention in nonvalvular AF, each DOAC has been approved for other indications and has been addressed further in guideline-based recommendations outside FDA-approved indications.

Overview of DOACs
Dabigatran is the only direct thrombin inhibitor; the other agents inhibit factor Xa. TABLE 11-4 summarizes FDA-approved indications and dosing and guideline-based dosing. Dabigatran and edoxaban require parenteral anticoagulation for 5 to 10 days prior to initiation for acute VTE, limiting their use.1,4 TABLE 21-4 highlights pharmacokinetic differences among the agents. For example, dabigatran is 80% renally cleared, is somewhat dialyzable, and can accumulate in patients with renal dysfunction.1 Edoxaban is contraindicated for nonvalvular AF in patients with a creatinine clearance (CrCl) > 95 mL/min because an increased stroke risk was demonstrated.4 Therefore, rivaroxaban and apixaban are prescribed most often in the United States.6,7

Applications in special patient populations
Obesity
As of 2020, more than 40% of adults in the United States were obese (body mass index [BMI] ≥ 30), with 9% classified as class 3 or severely obese (BMI ≥ 40).8 Altered drug pharmacokinetics in patients with severe obesity raises concern for undertreatment with fixed-dose DOACs. Phase III DOAC approval trials included patients with obesity, but weight cutoffs differed, making extrapolating efficacy and safety data difficult across different obesity stages.9 Although no FDA-labeled dosing adjustments exist for patients with obesity, the International Society on Thrombosis and Haemostasis (ISTH) does provide such recommendations.
ISTH changes position on measuring drug levels. ISTH previously recommended avoiding DOACs in those with a BMI > 40 or body weight > 120 kg. If a DOAC was used, ISTH advised obtaining peak and trough drug levels.10 However, DOAC drug levels have not been associated with clinical outcomes or sufficient degrees of anticoagulation.11
Men and women are affected equally by fibrolipomas. Prevalence does not differ by race or ethnicity.
In April 2021, ISTH updated guidance on DOACs in obesity, indicating standard doses of rivaroxaban or apixaban can be used for the treatment and prevention of VTE in all patients regardless of weight or BMI. Because data in obesity are lacking for dabigatran and edoxaban, avoid using these agents in patients with a BMI > 40 or weight > 120 kg. Additionally, assessing drug levels is no longer recommended, as there is insufficient evidence that these impact clinical outcomes.12
The 2021 American College of Chest Physicians (CHEST) guideline update
Continue to: Effectiveness of DOACs for AF in patients with obesity isn't clear
Effectiveness of DOACs for AF in patients with obesity isn’t clear, as most data are from retrospective cohort analyses. In patients weighing > 120 kg, dabigatran has shown efficacy in thrombosis prevention similar to that achieved in those weighing ≤ 120 kg, but it has increased the risk for gastrointestinal (GI) bleeding.15 Another study indicated a 15-mg dose of rivaroxaban may be associated with increased thromboembolic complications in patients with a BMI ≥ 35.16 Alternatively, another retrospective study of rivaroxaban demonstrated a small absolute risk reduction in ischemic stroke among patients in all stages of obesity and no difference in significant bleeding events.17 One further retrospective cohort showed that, in patients with a BMI ≥ 50 kg, the effectiveness of rivaroxaban and apixaban in thrombosis prevention and bleeding safety outcomes was comparable to that seen in those with a BMI < 30.18
As a result of conflicting data, and a lack of prospective randomized controlled trials (RCTs), ISTH continued recommending international normalized ratio (INR)–based dosing of warfarin for class 3 or severely obese patients with AF. The 2018 CHEST guidelines19 and the 2020 ESC guidelines20 make no mention of DOAC avoidance in patients with obesity and AF.
Advanced and end-stage renal disease
DOACs are renally dosed based on indication, drug-drug interactions, and degree of renal function (TABLE 31-4). For example, patients with AF who are anticoagulated with apixaban are prescribed 2.5 mg twice daily when 2 of the 3 following criteria are met: age ≥ 80 years, body weight ≤ 60 kg, serum creatinine ≥ 1.5 mg/dL. However, no dosage adjustment is necessary for VTE treatment or prophylaxis with apixaban regardless of renal function.3

Data supporting the safety and efficacy of DOACs in end-stage renal disease (ESRD) are sparse. All DOACs are renally cleared to varying degrees (TABLE 21-4), theoretically increasing bleeding risk as kidney disease progresses. Apixaban is the least renally cleared of the DOACs and has been evaluated in the greatest number of trials for patients with ESRD for both VTE treatment and prevention and nonvalvular AF.21 As a result, the FDA approved standard-dose apixaban (5 mg twice daily) for VTE treatment and prevention and nonvalvular AF in patients with ESRD, even those requiring dialysis. Use the reduced apixaban dose (2.5 mg twice daily) in patients with ESRD and AF only if they are ≥ 80 years of age or their body weight is ≤ 60 kg.3
Patients with cancer
Cancer-associated acute VTE treatment. Cancer is an established risk factor for acute VTE but it also increases the risk for treatment-associated bleeding compared with patients without cancer.22 Historically, low-molecular-weight heparin (LMWH) was recommended over warfarin and DOACs for cancer-associated thromboses (CAT).23 Compared with warfarin, LMWH reduced the rate of recurrent VTE and had similar or reduced bleeding rates at 6 to 12 months.24-26 However, clinicians and patients often chose warfarin to avoid subcutaneous injections.27
CHEST guidelines recommend oral Xa inhibitors over LMWH for the treatment of CAT.13 The 2020 guidelines of the National Institute for Health and Care Excellence (NICE) recommend DOACs as an option for CAT along with LMWH or LMWH transitioned to warfarin.28 The American Society of Clinical Oncology (ASCO) recommends rivaroxaban for acute VTE treatment in CAT. No head-to-head trials have evaluated comparative efficacy of DOACs for CAT. However, edoxaban and rivaroxaban are associated with a greater risk for GI bleeding; therefore, apixaban is preferred in patients with GI malignancies.29 Standard DOAC VTE treatment dosing is recommended for all 3 agents.2-4
When using DOACs for patients with CAT, consider potential drug-drug interactions with chemotherapy regimens. All DOACs are transported by p-glycoprotein, while rivaroxaban and apixaban are substrates of cytochrome P450, leading to potentially significant drug-drug interactions.30 These interactions could affect the patient’s chemotherapeutic regimen, decrease the efficacy of the DOAC, or increase the risk for bleeding. Therefore, anticoagulation choice should be made in collaboration with the hematology/oncology team.
Continue to: Cancer-associated VTE prophylaxis...
Cancer-associated VTE prophylaxis. VTE prophylaxis for patients with cancer is complex and necessitates a global assessment of cancer location and treatment regimen and setting. Hospitalized patients receiving chemotherapy are at high risk for VTE if mobility is reduced or if other VTE risk factors are present. The International Initiative on Thrombosis and Cancer (ITAC)31 and ISTH32 recommend VTE prophylaxis with unfractionated heparin or LMWH (ISTH recommends LMWH more strongly). The 2020 ASCO Guidelines recommend pharmacologic anticoagulation but make no drug-specific recommendation.29 Parenteral treatment in hospitalized patients is not as burdensome as it is in ambulatory patients; therefore, these recommendations are less likely to elicit inpatient opposition.
In the ambulatory setting, patient avoidance of subcutaneous injections necessitates consideration of DOACs for CAT prophylaxis. The Khorana Risk Score (KRS) is a validated tool (scale, 0-7) to predict VTE risk in ambulatory patients receiving chemotherapy.33 KRS scores ≥ 2 indicate high thrombotic risk and the need for prophylactic anticoagulation. ASCO recommends apixaban, rivaroxaban, or LMWH.29 ISTH and ITAC both recommend apixaban or rivaroxaban over LMWH.31,34 An RCT published in June 2023 confirmed that, for adults with cancer and VTE, DOACs were noninferior to LMWH for preventing recurrent VTE for 6 months.35 The recommended doses for apixaban (2.5 mg twice daily) and rivaroxaban (10 mg daily) for CAT VTE prophylaxis are lower than FDA-approved treatment doses.31
Patients with thrombophilia: VTE prevention
Thrombophilias are broadly categorized as inherited or acquired, with inherited thrombophilia being more prevalent. The Factor V Leiden (FVL) variant affects 2% to 7% of the population, and prothrombin gene mutation (PGM) affects 1% to 2% of the population.36 Other forms of inherited thrombophilia, such as protein C deficiency, protein S deficiency, and antithrombin deficiency, occur less commonly (< 0.7% of the population).36 Antiphospholipid syndrome (APS), the most common acquired thrombophilia, affects approximately 2% of the population.36 APS involves multiple antibodies: anticardiolipin antibodies, lupus anticoagulant, and anti-beta-2 glycoprotein 1 antibodies. Establishing risk for thrombosis across the varying types of thrombophilia has proven difficult, but APS is considered the most thrombogenic thrombophilia apart from extremely rare homozygous inherited thrombophilias.36 Therefore, DOAC recommendations are thrombophilia specific.
A prospective cohort study evaluated DOACs compared with heparin/warfarin for VTE treatment in patients with inherited thrombophilias.37 Although all 4 available DOACs were included, most patients (61.1%) received rivaroxaban. Patients with an array of inherited thrombophilias, including rare homozygous mutations, were enrolled in this trial. While most patients (66.9%) had a “mild thrombophilia” defined as either FVL or PGM, the remainder had more severe thrombophilias.37 VTE recurrence was similar and uncommon in the DOAC and heparin/warfarin groups, consistent with a previous meta-analysis.38 Surprisingly, an increase in the cumulative risk for bleeding was seen in the DOAC group compared with the warfarin group, a finding inconsistent with prior trials.38 There were no major bleeding events in the DOAC group, but 3 such events occurred in the heparin/warfarin group, including 2 intracranial hemorrhages.
Currently NICE, CHEST, and ISTH do not make a recommendation for a preferred agent in patients with an acute VTE and inherited thrombophilia; however, DOACs would not be inappropriate.23,28,32 The American Society of Hematology (ASH) had planned to release recommendations related to the treatment of thrombophilia in 2020, but they were delayed by the COVID-19 pandemic.39
APS presents challenges for acute VTE anticoagulation. First, it causes a strongly thrombogenic state necessitating therapeutic anticoagulation. Second, for patients with positive lupus anticoagulant, INR monitoring and standardized INR goals may be inadequate.40 Therefore, using fixed-dose DOACs without the need for therapeutic monitoring is appealing, but significant concerns exist for using DOACs in patients with APS.41-45 ISTH and CHEST recommend warfarin for the treatment and prevention of acute VTE in patients with APS, especially those with triple-positive (anticardiolipin, lupus anticoagulant, and anti-beta-2 glycoprotein 1) APS.13,46 Package labeling for all DOACs recommends avoidance in triple-positive APS.1-4
ASTRO-APS is the most recent RCT to compare apixaban and warfarin for patients with APS,47 and it was terminated early after 6 of 23 patients in the apixaban group had thrombotic events, while no one in the warfarin group had such an event.48 Subsequently, a meta-analysis49 demonstrated that patients with thrombotic APS appear to have a greater risk for arterial thrombosis when treated with DOACs compared with warfarin. These 2 studies may lead to changes in recommendations to avoid DOACs in all patients with APS or may prompt more focused trials for DOAC use in patients with APS plus an antiplatelet to mitigate arterial thrombotic risk.
Continue to: Expanded clinical indications
Expanded clinical indications
Superficial vein thrombosis
Superficial thrombophlebitis or superficial vein thrombosis (SVT) is estimated to occur 6 times more frequently than VTE.50 Management of patients with isolated, uncomplicated thrombophlebitis who are at low risk for extension of the SVT involves symptomatic treatment with nonsteroidal anti-inflammatory drugs, topical agents, or compression therapy. However, depending on risk for progression, anticoagulation may be recommended.51
Patients at intermediate risk for extension or propagation of SVT are candidates for anticoagulation. The CHEST guidelines recommend
Certain situations should prompt one to consider using a treatment dose of a DOAC for 3 months. These include cases in which the SVT is located within 3 cm of the deep venous system, expands despite an appropriate prophylactic regimen, or recurs after discontinuation of prophylactic anticoagulation.13,50
Acute coronary syndrome
The American College of Cardiology/American Heart Association (ACC/AHA) recommend combination antiplatelet therapy and anticoagulation for management of acute coronary syndrome in hospitalized patients.52 Data are mixed regarding longer-term anticoagulation in addition to dual antiplatelet therapy in outpatient settings to prevent thrombosis recurrence in the absence of AF.
The APPRAISE-2 trial enrolled high-risk patients with ACS within 7 days of the event.53 Apixaban 5 mg twice daily was compared with placebo in patients taking aspirin or aspirin plus clopidogrel. The trial was terminated early because major bleeding events increased with apixaban without reduction in recurrent ischemic events. The ATLAS ACS-TIMI 46 trial evaluated different rivaroxaban doses (5-20 mg daily) in ACS patients.54 The study revealed possible thrombosis benefit but also increased risk for bleeding, particularly at higher doses. As a result, another study—ATLAS ACS 2-TIMI 51—was conducted and compared the use of low-dose rivaroxaban (2.5 mg twice daily or 5 mg twice daily) vs placebo for patients with recent ACS.55 All patients were receiving low-dose aspirin, and approximately 93% of patients in each group also were receiving clopidogrel or ticlopidine. As in the APPRAISE-2 trial, rivaroxaban increased the rate of major bleeding and intracranial hemorrhage; however, it did not increase the incidence of fatal bleeding. Unlike APPRAISE-2, rivaroxaban significantly reduced the primary efficacy end point, a composite of death from cardiovascular causes, myocardial infarction, or stroke (absolute risk reduction = 1.8%; number needed to treat = 56 for combined rivaroxaban doses).55
A secondary subgroup analysis combined data from the ATLAS ACMS-TIMI 46 and ATLAS ACS 2-TIMI 51 trials to evaluate outcomes in patients receiving aspirin monotherapy when combined with rivaroxaban 2.5 mg twice daily or 5 mg twice daily or with placebo.56 The primary efficacy end point was a composite of cardiovascular death, myocardial infarction, or stroke. When the 2 trials were evaluated separately, neither rivaroxaban dose was associated with reduction of the primary efficacy outcomes compared with aspirin alone. However, when the data were pooled, both the combined rivaroxaban doses (particularly the 5-mg dose) were associated with reduced cardiovascular outcomes. From a safety perspective, the 2.5-mg twice-daily dose of rivaroxaban was the only dose not associated with increased major bleeding risk. Thus, the 2.5-mg twice-daily dose of rivaroxaban may not provide sufficient cardiovascular benefit in patients with ACS, while the larger dose may increase the risk for nonfatal major bleeding events.56
The European Medicines Agency57 approved rivaroxaban 2.5 mg twice daily for ACS, and the 2020 ESC guidelines58 consider it an appropriate therapeutic option in addition to aspirin for patients at high ischemic risk and low bleeding risk. ACS is not an FDA-approved indication for DOACs, and the ACC/AHA Guideline for the Management of ACS, last updated in 2014, does not include DOACs for ACS unless patients have AF.52 Ongoing trials are further investigating rivaroxaban for ACS, so the use of DOACs in the post-acute phase of ACS may become clearer in the future.59
Continue to: Heparin-induced thrombocytopenia
Heparin-induced thrombocytopenia
Historically, nonheparin parenteral anticoagulants argatroban, bivalirudin, and fondaparinux were recommended for patients at risk for or who had heparin-induced thrombocytopenia (HIT). Argatroban is the only drug FDA approved for the treatment and prophylaxis of HIT; recommendations for the others are based on guideline recommendations.23,60,61 The nonheparin parenteral anticoagulants cost between $700 and $1500 per day; therefore most patients with HIT are transitioned to warfarin.62 However, protein C and S inhibition and a subsequent prothrombotic state conveyed by warfarin initiation necessitates a minimum 5-day bridge to therapeutic warfarin with a nonheparin parenteral anticoagulant.
In vitro tests show that DOACs do not promote development of HIT antibodies63 or affect platelet activation or aggregation.64 A literature summary of DOACs for HIT determined that in 104 patients, all but 1 achieved platelet recovery (defined as > 150,000/mcL) within a median time of 7 days. Therapeutically, DOACs prevented new or recurrent VTE in 102/104 cases (98%), and only 3% of patients experienced significant bleeding events.62
The 2018 ASH guidelines for VTE management in HIT include (with very low certainty of evidence) dabigatran, rivaroxaban, or apixaban for consideration in addition to previously recommended nonheparin parenteral anticoagulants.61 The dosing of each agent is contingent upon treatment of patients with HIT and an acute thrombosis (HITT) or HIT in the absence of VTE. For patients with HITT, treatment doses for acute VTE should be used for the appropriate duration of therapy (ie, 3 months). Importantly, dabigatran requires a 5-day pretreatment period with a parenteral anticoagulant, so it is not an ideal option. When treating isolated HIT (in the absence of VTE), ASH recommends all agents be dosed twice daily—dabigatran 150 mg twice daily (no 5-day parenteral pretreatment necessary), rivaroxaban 15 mg twice daily, or apixaban 5 mg twice daily—until platelet recovery (≥ 150,000/mcL) is achieved.61
CORRESPONDENCE
Kevin Schleich, PharmD, BCACP, Departments of Pharmaceutical Care and Family Medicine, University of Iowa, 200 Hawkins Drive, 01102-D PFP, Iowa City, IA, 52242; kevin-schleich@uiowa.edu
1. Dabigatran. Package Insert. Boehringer Ingelheim Pharmaceuticals, Inc.; 2021.
2. Rivaroxaban. Package insert. Janssen Pharmaceuticals, Inc; 2022.
3. Apixaban. Package insert. Bristol-Myers Squibb; 2021.
4. Edoxaban. Package insert. Daiichi Sankyo, Inc; 2015.
5. Betrixaban. Package insert. Portola Pharmaceuticals, Inc; 2017.
6. Wheelock KM, Ross JS, Murugiah K, et al. Clinician trends in prescribing direct oral anticoagulants for US Medicare beneficiaries. JAMA Netw Open. 2021;4:e2137288. doi: 10.1001/jamanetworkopen.2021.37288
7. Colacci M, Tseng EK, Sacks CA, et al. Oral anticoagulant utilization in the United States and United Kingdom. J Gen Intern Med. 2020;35:2505-2507. doi: 10.1007/s11606-020-05904-0
8. CDC. Adult obesity facts. Accessed May 9, 2023. www.cdc.gov/obesity/data/adult.html
9. Mocini D, Di Fusco SA, Mocini E, et al. Direct oral anticoagulants in patients with obesity and atrial fibrillation: position paper of Italian National Association of Hospital Cardiologists (ANMCO). J Clin Med. 2021;10:4185. doi: 10.3390/jcm10184185
10. Martin K, Beyer-Westendorf J, Davidson BL, et al. Use of the direct oral anticoagulants in obese patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14:1308-1313. doi: 10.1111/jth.13323
11. Gu TM, Garcia DA, Sabath DE. Assessment of direct oral anticoagulant assay use in clinical practice. J Thromb Thrombolysis. 2019;47:403-408. doi: 10.1007/s11239-018-1793-0
12. Martin KA, Beyer-Westendorf J, Davidson BL, et al. Use of direct oral anticoagulants in patients with obesity for treatment and prevention of venous thromboembolism: updated communication from the ISTH SSC Subcommittee on Control of Anticoagulation. J Thromb Haemost. 2021;19:1874-1882. doi: 10.1111/jth.15358
13. Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST Guideline and Expert Panel Report. Chest. 2021;160:e545-e608. doi: 10.1016/j.chest.2021.07.055
14. Konstantinides SV, Meyer G, Becattini C, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur Heart J. 2020;41:543-603. doi: 10.1093/eurheartj/ehz405
15. Coates J, Bitton E, Hendje A, et al. Clinical outcomes of dabigatran use in patients with non-valvular atrial fibrillation and weight >120 kg. Thromb Res. 2021;208:176-180. doi: 10.1016/j.thromres.2021.11.007
16. Li X, Zuo C, Ji Q, et al. Body mass index influence on the clinical outcomes for nonvalvular atrial fibrillation patients admitted to a hospital treated with direct oral anticoagulants: a retrospective cohort study. Drug Des Devel Ther. 2021;15:1931-1943. doi: 10.2147/dddt.S303219
17. Barakat AF, Jain S, Masri A, et al. Outcomes of direct oral anticoagulants in atrial fibrillation patients across different body mass index categories. JACC Clin Electrophysiol. 2021;7:649-658. doi: 10.1016/j.jacep.2021.02.002
18. O’Kane CP, Avalon JCO, Lacoste JL, et al. Apixaban and rivaroxaban use for atrial fibrillation in patients with obesity and BMI ≥50 kg/m2. Pharmacotherapy. 2022;42:112-118. doi: https://doi.org/10.1002/phar.2651
19. Lip GYH, Banerjee A, Boriani G, et al. Antithrombotic therapy for atrial fibrillation: CHEST Guideline and Expert Panel Report. Chest. 2018;154:1121-1201. doi: 10.1016/j.chest.2018.07.040
20. Sepehri Shamloo A, Dagres N, Hindricks G. [2020 ESC guidelines on atrial fibrillation: summary of the most relevant recommendations and innovations]. Herz. 2021;46:28-37. doi: 10.1007/s00059-020-05005-y
21. Chokesuwattanaskul R, Thongprayoon C, Tanawuttiwat T, et al. Safety and efficacy of apixaban versus warfarin in patients with end-stage renal disease: meta-analysis. Pacing Clin Electrophysiol. 2018;41:627-634. doi: 10.1111/pace.13331
22. Wang T-F, Li A, Garcia D. Managing thrombosis in cancer patients. Res Pract Thromb Haemost. 2018;2:429-438. doi: https://doi.org/10.1002/rth2.12102
23. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. CHEST. 2016;149:315-352. doi: 10.1016/j.chest.2015.11.026
24. Lee AY, Levine MN, Baker RI, et al. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349:146-153. doi: 10.1056/NEJMoa025313
25. Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med. 2002;162:1729-1735. doi: 10.1001/archinte.162.15.1729
26. Hull RD, Pineo GF, Brant RF, et al. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006;119:1062-1072. doi: 10.1016/j.amjmed.2006.02.022
27. Lee AYY, Kamphuisen PW, Meyer G, et al. Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 2015;314:677-686. doi: 10.1001/jama.2015.9243
28. NICE Guideline. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing. Accessed May 9, 2023. www.ncbi.nlm.nih.gov/books/NBK556698/
29. Key NS, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: ASCO Clinical Practice Guideline Update. J Clin Oncol. 2020;38:496-520. doi: 10.1200/jco.19.01461
30. Galgani A, Palleria C, Iannone LF, et al. Pharmacokinetic interactions of clinical interest between direct oral anticoagulants and antiepileptic drugs. Front Neurol. 2018;9:1067. doi: 10.3389/fneur.2018.01067
31. Farge D, Frere C, Connors JM, et al. 2019 International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. Lancet Oncol. 2019;20:e566-e581. doi: 10.1016/s1470-2045(19)30336-5
32. Di Nisio M, Carrier M, Lyman GH, et al. Prevention of venous thromboembolism in hospitalized medical cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2014;12:1746-1749. doi: 10.1111/jth.12683
33. Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902-4907. doi: 10.1182/blood-2007-10-116327
34. Wang TF, Zwicker JI, Ay C, et al. The use of direct oral anticoagulants for primary thromboprophylaxis in ambulatory cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2019;17:1772-1778. doi: 10.1111/jth.14564
35. Schrag D, Uno H, Rosovsky R, et al. Direct oral anticoagulants vs low-molecular-weight heparin and recurrent VTE in patients with cancer: a randomized clinical trial. JAMA. 2023;329:1924-1933. doi: 10.1001/jama.2023.7843
36. Stevens SM, Woller SC, Bauer KA, et al. Guidance for the evaluation and treatment of hereditary and acquired thrombophilia. J Thromb Thrombolysis. 2016;41:154-164. doi: 10.1007/s11239-015-1316-1
37. Campello E, Spiezia L, Simion C, et al. Direct oral anticoagulants in patients with inherited thrombophilia and venous thromboembolism: a prospective cohort study. J Am Heart Assoc. 2020;9:e018917. doi: 10.1161/jaha.120.018917
38. Elsebaie MAT, van Es N, Langston A, et al. Direct oral anticoagulants in patients with venous thromboembolism and thrombophilia: a systematic review and meta-analysis. J Thromb Haemost. 2019;17:645-656. doi: 10.1111/jth.14398
39. ASH. ASH Clinical Practice Guidelines on Venous Thromboembolism. Accessed May 10, 2023. www.hematology.org/education/clinicians/guidelines-and-quality-care/clinical-practice-guidelines/venous-thromboembolism-guidelines
40. Baquero-Salamanca M, Téllez-Arévalo AM, Calderon-Ospina C. Variability in the international normalised ratio (INR) in patients with antiphospholipid syndrome and positive lupus anticoagulant: should the INR targets be higher? BMJ Case Rep. 2015;2015:bcr2014209013. doi: 10.1136/bcr-2014-209013
41. Pengo V, Denas G, Zoppellaro G, et al. Rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome. Blood. 2018;132:1365-1371. doi: 10.1182/blood-2018-04-848333
42. Ordi-Ros J, Sáez-Comet L, Pérez-Conesa M, et al. Rivaroxaban versus vitamin K antagonist in antiphospholipid syndrome: a randomized noninferiority trial. Ann Intern Med. 2019;171:685-694. doi: 10.7326/m19-0291
43. Sato T, Nakamura H, Fujieda Y, et al. Factor Xa inhibitors for preventing recurrent thrombosis in patients with antiphospholipid syndrome: a longitudinal cohort study. Lupus. 2019;28:1577-1582. doi: 10.1177/0961203319881200
44. Malec K, Broniatowska E, Undas A. Direct oral anticoagulants in patients with antiphospholipid syndrome: a cohort study. Lupus. 2020;29:37-44. doi: 10.1177/0961203319889156
45. Rivaroxaban versus warfarin to treat patients with thrombotic antiphospholipid syndrome. Dr. Hannah Cohen about the results of the RAPS trial (Lancet Haematol 2016; 3: e426-36). Rheumatology (Oxford). 2017;56:e23. doi: 10.1093/rheumatology/kex290
46. Zuily S, Cohen H, Isenberg D, et al. Use of direct oral anticoagulants in patients with thrombotic antiphospholipid syndrome: guidance from the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. J Thromb Haemost. 2020;18:2126-2137. doi: https://doi.org/10.1111/jth.14935
47. NIH. ClinicalTrials.gov. Apixaban for the secondary prevention of thromboembolism among patients with antiphospholipid syndrome (ASTRO-APS). Accessed May 10, 2023. https://clinicaltrials.gov/ct2/show/NCT02295475?term=apixaban&cond=Anti+Phospholipid+Syndrome&draw=2&rank=1
48. Woller SC, Stevens SM, Kaplan D, et al. Apixaban compared with warfarin to prevent thrombosis in thrombotic antiphospholipid syndrome: a randomized trial. Blood Adv. 2022;6:1661-1670. doi: 10.1182/bloodadvances.2021005808
49. Khairani CD, Bejjani A, Piazza G, et al. Direct oral anticoagulants vs vitamin K antagonists in patients with antiphospholipid syndromes: meta-analysis of randomized trials. J Am Coll Cardiol. 2023;81:16-30. doi: 10.1016/j.jacc.2022.10.008
50. Superficial thrombophlebitis, superficial vein thrombosis. 2021. Accessed May 10, 2023. thrombosiscanada.ca/wp-content/uploads/2021/07/47.-Superficial-Vein-Thrombosis_16July2021.pdf
51. Di Nisio M, Wichers IM, Middeldorp S. Treatment for superficial thrombophlebitis of the leg. Cochrane Database Syst Rev. 2018;2:CD004982. doi: 10.1002/14651858.CD004982.pub6
52. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the Management of Patients With Non–ST-Elevation Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;64:e139-e228. doi: 10.1016/j.jacc.2014.09.017
53. Alexander JH, Lopes RD, James S, et al. Apixaban with antiplatelet therapy after acute coronary syndrome. N Engl J Med. 2011;365:699-708. doi: 10.1056/NEJMoa1105819
54. Mega JL, Braunwald E, Mohanavelu S, et al. Rivaroxaban versus placebo in patients with acute coronary syndromes (ATLAS ACS-TIMI 46): a randomised, double-blind, phase II trial. Lancet. 2009;374:29-38. doi: 10.1016/s0140-6736(09)60738-8
55. Mega JL, Braunwald E, Wiviott SD, et al. Rivaroxaban in patients with a recent acute coronary syndrome. N Engl J Med. 2012;366:9-19. doi: 10.1056/NEJMoa1112277
56. Gibson WJ, Gibson CM, Yee MK, et al. Safety and efficacy of rivaroxaban when added to aspirin monotherapy among stabilized post‐acute coronary syndrome patients: a pooled analysis study of ATLAS ACS‐TIMI 46 and ATLAS ACS 2‐TIMI 51. J Am Heart Assoc. 2019. Accessed May 10, 2023. Doi: 10.1161/JAHA.118.009451
57. European Medicines Agency. Xarelto (rivaroxaban). 2008. Accessed June 23, 2023.
58. Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42:1289-1367. doi: 10.1093/eurheartj/ehaa575
59. NIH. ClinicalTrials.gov. Accessed May 10, 2023. www.clinicaltrials.gov/ct2/results?cond=Acute+Coronary+Syndrome&term=rivaroxaban+&cntry=&state=&city=&dist=#
60. Watson H, Davidson S, Keeling D. Guidelines on the diagnosis and management of heparin-induced thrombocytopenia: second edition. Br J Haematol. 2012;159:528-40. doi: 10.1111/bjh.12059
61. Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv. 2018;2:3360-3392. doi: 10.1182/bloodadvances.2018024489
62. Momin J, Lee C-S. The role of direct oral anticoagulants in the management of heparin-induced thrombocytopenia US Pharmacist. 2020;45:3-10. Accessed May 10, 2023. www.uspharmacist.com/article/the-role-of-direct-oral-anticoagulants-in-the-management-of-heparininduced-thrombocytopenia
63. Warkentin TE, Pai M, Linkins LA. Direct oral anticoagulants for treatment of HIT: update of Hamilton experience and literature review. Blood. 2017;130:1104-1113. doi: 10.1182/blood-2017-04-778993
64. Krauel K, Hackbarth C, Fürll B, et al. Heparin-induced thrombocytopenia: in vitro studies on the interaction of dabigatran, rivaroxaban, and low-sulfated heparin, with platelet factor 4 and anti-PF4/heparin antibodies. Blood. 2012;119:1248-1255. doi: 10.1182/blood-2011-05-353391
1. Dabigatran. Package Insert. Boehringer Ingelheim Pharmaceuticals, Inc.; 2021.
2. Rivaroxaban. Package insert. Janssen Pharmaceuticals, Inc; 2022.
3. Apixaban. Package insert. Bristol-Myers Squibb; 2021.
4. Edoxaban. Package insert. Daiichi Sankyo, Inc; 2015.
5. Betrixaban. Package insert. Portola Pharmaceuticals, Inc; 2017.
6. Wheelock KM, Ross JS, Murugiah K, et al. Clinician trends in prescribing direct oral anticoagulants for US Medicare beneficiaries. JAMA Netw Open. 2021;4:e2137288. doi: 10.1001/jamanetworkopen.2021.37288
7. Colacci M, Tseng EK, Sacks CA, et al. Oral anticoagulant utilization in the United States and United Kingdom. J Gen Intern Med. 2020;35:2505-2507. doi: 10.1007/s11606-020-05904-0
8. CDC. Adult obesity facts. Accessed May 9, 2023. www.cdc.gov/obesity/data/adult.html
9. Mocini D, Di Fusco SA, Mocini E, et al. Direct oral anticoagulants in patients with obesity and atrial fibrillation: position paper of Italian National Association of Hospital Cardiologists (ANMCO). J Clin Med. 2021;10:4185. doi: 10.3390/jcm10184185
10. Martin K, Beyer-Westendorf J, Davidson BL, et al. Use of the direct oral anticoagulants in obese patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14:1308-1313. doi: 10.1111/jth.13323
11. Gu TM, Garcia DA, Sabath DE. Assessment of direct oral anticoagulant assay use in clinical practice. J Thromb Thrombolysis. 2019;47:403-408. doi: 10.1007/s11239-018-1793-0
12. Martin KA, Beyer-Westendorf J, Davidson BL, et al. Use of direct oral anticoagulants in patients with obesity for treatment and prevention of venous thromboembolism: updated communication from the ISTH SSC Subcommittee on Control of Anticoagulation. J Thromb Haemost. 2021;19:1874-1882. doi: 10.1111/jth.15358
13. Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST Guideline and Expert Panel Report. Chest. 2021;160:e545-e608. doi: 10.1016/j.chest.2021.07.055
14. Konstantinides SV, Meyer G, Becattini C, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur Heart J. 2020;41:543-603. doi: 10.1093/eurheartj/ehz405
15. Coates J, Bitton E, Hendje A, et al. Clinical outcomes of dabigatran use in patients with non-valvular atrial fibrillation and weight >120 kg. Thromb Res. 2021;208:176-180. doi: 10.1016/j.thromres.2021.11.007
16. Li X, Zuo C, Ji Q, et al. Body mass index influence on the clinical outcomes for nonvalvular atrial fibrillation patients admitted to a hospital treated with direct oral anticoagulants: a retrospective cohort study. Drug Des Devel Ther. 2021;15:1931-1943. doi: 10.2147/dddt.S303219
17. Barakat AF, Jain S, Masri A, et al. Outcomes of direct oral anticoagulants in atrial fibrillation patients across different body mass index categories. JACC Clin Electrophysiol. 2021;7:649-658. doi: 10.1016/j.jacep.2021.02.002
18. O’Kane CP, Avalon JCO, Lacoste JL, et al. Apixaban and rivaroxaban use for atrial fibrillation in patients with obesity and BMI ≥50 kg/m2. Pharmacotherapy. 2022;42:112-118. doi: https://doi.org/10.1002/phar.2651
19. Lip GYH, Banerjee A, Boriani G, et al. Antithrombotic therapy for atrial fibrillation: CHEST Guideline and Expert Panel Report. Chest. 2018;154:1121-1201. doi: 10.1016/j.chest.2018.07.040
20. Sepehri Shamloo A, Dagres N, Hindricks G. [2020 ESC guidelines on atrial fibrillation: summary of the most relevant recommendations and innovations]. Herz. 2021;46:28-37. doi: 10.1007/s00059-020-05005-y
21. Chokesuwattanaskul R, Thongprayoon C, Tanawuttiwat T, et al. Safety and efficacy of apixaban versus warfarin in patients with end-stage renal disease: meta-analysis. Pacing Clin Electrophysiol. 2018;41:627-634. doi: 10.1111/pace.13331
22. Wang T-F, Li A, Garcia D. Managing thrombosis in cancer patients. Res Pract Thromb Haemost. 2018;2:429-438. doi: https://doi.org/10.1002/rth2.12102
23. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. CHEST. 2016;149:315-352. doi: 10.1016/j.chest.2015.11.026
24. Lee AY, Levine MN, Baker RI, et al. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349:146-153. doi: 10.1056/NEJMoa025313
25. Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med. 2002;162:1729-1735. doi: 10.1001/archinte.162.15.1729
26. Hull RD, Pineo GF, Brant RF, et al. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006;119:1062-1072. doi: 10.1016/j.amjmed.2006.02.022
27. Lee AYY, Kamphuisen PW, Meyer G, et al. Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 2015;314:677-686. doi: 10.1001/jama.2015.9243
28. NICE Guideline. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing. Accessed May 9, 2023. www.ncbi.nlm.nih.gov/books/NBK556698/
29. Key NS, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: ASCO Clinical Practice Guideline Update. J Clin Oncol. 2020;38:496-520. doi: 10.1200/jco.19.01461
30. Galgani A, Palleria C, Iannone LF, et al. Pharmacokinetic interactions of clinical interest between direct oral anticoagulants and antiepileptic drugs. Front Neurol. 2018;9:1067. doi: 10.3389/fneur.2018.01067
31. Farge D, Frere C, Connors JM, et al. 2019 International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. Lancet Oncol. 2019;20:e566-e581. doi: 10.1016/s1470-2045(19)30336-5
32. Di Nisio M, Carrier M, Lyman GH, et al. Prevention of venous thromboembolism in hospitalized medical cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2014;12:1746-1749. doi: 10.1111/jth.12683
33. Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902-4907. doi: 10.1182/blood-2007-10-116327
34. Wang TF, Zwicker JI, Ay C, et al. The use of direct oral anticoagulants for primary thromboprophylaxis in ambulatory cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost. 2019;17:1772-1778. doi: 10.1111/jth.14564
35. Schrag D, Uno H, Rosovsky R, et al. Direct oral anticoagulants vs low-molecular-weight heparin and recurrent VTE in patients with cancer: a randomized clinical trial. JAMA. 2023;329:1924-1933. doi: 10.1001/jama.2023.7843
36. Stevens SM, Woller SC, Bauer KA, et al. Guidance for the evaluation and treatment of hereditary and acquired thrombophilia. J Thromb Thrombolysis. 2016;41:154-164. doi: 10.1007/s11239-015-1316-1
37. Campello E, Spiezia L, Simion C, et al. Direct oral anticoagulants in patients with inherited thrombophilia and venous thromboembolism: a prospective cohort study. J Am Heart Assoc. 2020;9:e018917. doi: 10.1161/jaha.120.018917
38. Elsebaie MAT, van Es N, Langston A, et al. Direct oral anticoagulants in patients with venous thromboembolism and thrombophilia: a systematic review and meta-analysis. J Thromb Haemost. 2019;17:645-656. doi: 10.1111/jth.14398
39. ASH. ASH Clinical Practice Guidelines on Venous Thromboembolism. Accessed May 10, 2023. www.hematology.org/education/clinicians/guidelines-and-quality-care/clinical-practice-guidelines/venous-thromboembolism-guidelines
40. Baquero-Salamanca M, Téllez-Arévalo AM, Calderon-Ospina C. Variability in the international normalised ratio (INR) in patients with antiphospholipid syndrome and positive lupus anticoagulant: should the INR targets be higher? BMJ Case Rep. 2015;2015:bcr2014209013. doi: 10.1136/bcr-2014-209013
41. Pengo V, Denas G, Zoppellaro G, et al. Rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome. Blood. 2018;132:1365-1371. doi: 10.1182/blood-2018-04-848333
42. Ordi-Ros J, Sáez-Comet L, Pérez-Conesa M, et al. Rivaroxaban versus vitamin K antagonist in antiphospholipid syndrome: a randomized noninferiority trial. Ann Intern Med. 2019;171:685-694. doi: 10.7326/m19-0291
43. Sato T, Nakamura H, Fujieda Y, et al. Factor Xa inhibitors for preventing recurrent thrombosis in patients with antiphospholipid syndrome: a longitudinal cohort study. Lupus. 2019;28:1577-1582. doi: 10.1177/0961203319881200
44. Malec K, Broniatowska E, Undas A. Direct oral anticoagulants in patients with antiphospholipid syndrome: a cohort study. Lupus. 2020;29:37-44. doi: 10.1177/0961203319889156
45. Rivaroxaban versus warfarin to treat patients with thrombotic antiphospholipid syndrome. Dr. Hannah Cohen about the results of the RAPS trial (Lancet Haematol 2016; 3: e426-36). Rheumatology (Oxford). 2017;56:e23. doi: 10.1093/rheumatology/kex290
46. Zuily S, Cohen H, Isenberg D, et al. Use of direct oral anticoagulants in patients with thrombotic antiphospholipid syndrome: guidance from the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. J Thromb Haemost. 2020;18:2126-2137. doi: https://doi.org/10.1111/jth.14935
47. NIH. ClinicalTrials.gov. Apixaban for the secondary prevention of thromboembolism among patients with antiphospholipid syndrome (ASTRO-APS). Accessed May 10, 2023. https://clinicaltrials.gov/ct2/show/NCT02295475?term=apixaban&cond=Anti+Phospholipid+Syndrome&draw=2&rank=1
48. Woller SC, Stevens SM, Kaplan D, et al. Apixaban compared with warfarin to prevent thrombosis in thrombotic antiphospholipid syndrome: a randomized trial. Blood Adv. 2022;6:1661-1670. doi: 10.1182/bloodadvances.2021005808
49. Khairani CD, Bejjani A, Piazza G, et al. Direct oral anticoagulants vs vitamin K antagonists in patients with antiphospholipid syndromes: meta-analysis of randomized trials. J Am Coll Cardiol. 2023;81:16-30. doi: 10.1016/j.jacc.2022.10.008
50. Superficial thrombophlebitis, superficial vein thrombosis. 2021. Accessed May 10, 2023. thrombosiscanada.ca/wp-content/uploads/2021/07/47.-Superficial-Vein-Thrombosis_16July2021.pdf
51. Di Nisio M, Wichers IM, Middeldorp S. Treatment for superficial thrombophlebitis of the leg. Cochrane Database Syst Rev. 2018;2:CD004982. doi: 10.1002/14651858.CD004982.pub6
52. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the Management of Patients With Non–ST-Elevation Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;64:e139-e228. doi: 10.1016/j.jacc.2014.09.017
53. Alexander JH, Lopes RD, James S, et al. Apixaban with antiplatelet therapy after acute coronary syndrome. N Engl J Med. 2011;365:699-708. doi: 10.1056/NEJMoa1105819
54. Mega JL, Braunwald E, Mohanavelu S, et al. Rivaroxaban versus placebo in patients with acute coronary syndromes (ATLAS ACS-TIMI 46): a randomised, double-blind, phase II trial. Lancet. 2009;374:29-38. doi: 10.1016/s0140-6736(09)60738-8
55. Mega JL, Braunwald E, Wiviott SD, et al. Rivaroxaban in patients with a recent acute coronary syndrome. N Engl J Med. 2012;366:9-19. doi: 10.1056/NEJMoa1112277
56. Gibson WJ, Gibson CM, Yee MK, et al. Safety and efficacy of rivaroxaban when added to aspirin monotherapy among stabilized post‐acute coronary syndrome patients: a pooled analysis study of ATLAS ACS‐TIMI 46 and ATLAS ACS 2‐TIMI 51. J Am Heart Assoc. 2019. Accessed May 10, 2023. Doi: 10.1161/JAHA.118.009451
57. European Medicines Agency. Xarelto (rivaroxaban). 2008. Accessed June 23, 2023.
58. Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42:1289-1367. doi: 10.1093/eurheartj/ehaa575
59. NIH. ClinicalTrials.gov. Accessed May 10, 2023. www.clinicaltrials.gov/ct2/results?cond=Acute+Coronary+Syndrome&term=rivaroxaban+&cntry=&state=&city=&dist=#
60. Watson H, Davidson S, Keeling D. Guidelines on the diagnosis and management of heparin-induced thrombocytopenia: second edition. Br J Haematol. 2012;159:528-40. doi: 10.1111/bjh.12059
61. Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv. 2018;2:3360-3392. doi: 10.1182/bloodadvances.2018024489
62. Momin J, Lee C-S. The role of direct oral anticoagulants in the management of heparin-induced thrombocytopenia US Pharmacist. 2020;45:3-10. Accessed May 10, 2023. www.uspharmacist.com/article/the-role-of-direct-oral-anticoagulants-in-the-management-of-heparininduced-thrombocytopenia
63. Warkentin TE, Pai M, Linkins LA. Direct oral anticoagulants for treatment of HIT: update of Hamilton experience and literature review. Blood. 2017;130:1104-1113. doi: 10.1182/blood-2017-04-778993
64. Krauel K, Hackbarth C, Fürll B, et al. Heparin-induced thrombocytopenia: in vitro studies on the interaction of dabigatran, rivaroxaban, and low-sulfated heparin, with platelet factor 4 and anti-PF4/heparin antibodies. Blood. 2012;119:1248-1255. doi: 10.1182/blood-2011-05-353391
PRACTICE RECOMMENDATIONS
› Consider a direct oral anticoagulant (DOAC) when treating venous thromboembolism (VTE) in patients with advanced chronic kidney disease or obesity. C
› Select apixaban for treatment of VTE or nonvalvular atrial fibrillation in patients with end-stage renal disease, due to its minimal renal clearance compared with other DOACs. B
› Consider DOACs such as dabigatran, rivaroxaban, or apixaban for treatment of VTE in the context of heparin-induced thrombocytopenia. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
What makes teens choose to use sunscreen?
a cornerstone of skin cancer prevention, according to results from a systematic review.
“We know that skin cancer is one of the most common malignancies in the world, and sun protection methods such as sunscreen make it highly preventable,” first author Carly R. Stevens, a student at Tulane University, New Orleans, said in an interview. “This study demonstrates the adolescent populations that are most vulnerable to sun damage and how we can help mitigate their risk of developing skin cancer through education methods, such as Sun Protection Outreach Teaching by Students.”
Ms. Stevens and coauthors presented the findings during a poster session at the annual meeting of the Society for Pediatric Dermatology.
To investigate predictors of sunscreen use among high school students, they searched PubMed, Embase, and Web of Science using the terms (“sunscreen” or “SPF” or “sun protection”) and (“high school” or “teen” or “teenager” or “adolescent”) and limited the analysis to English studies reporting data on sunscreen use in U.S. high school students up to November 2021.
A total of 20 studies were included in the final review. The study populations ranged in number from 208 to 24,645. Of 11 studies that examined gender, all showed increased sunscreen use in females compared with males. Of five studies that examined age, all showed increased sunscreen use in younger adolescents, compared with their older counterparts.
Of four studies that examined the role of ethnicity on sunscreen use, White students were more likely to use sunscreen, compared with their peers of other ethnicities. “This may be due to perceived sun sensitivity, as [these four studies] also showed increased sunscreen use in populations that believed were more susceptible to sun damage,” the researchers wrote in their abstract.
In other findings, two studies that examined perceived self-efficacy concluded that higher levels of sunscreen use correlated with higher self-efficacy, while four studies concluded that high school students were more likely to use sunscreen if their parents encouraged them the wear it or if the parent used it themselves.
“With 40%-50% of ultraviolet damage being done before the age of 20, it’s crucial that we find ways to educate adolescents on the importance of sunscreen use and target those populations who were found to rarely use sunscreen in our study,” Ms. Stevens said.
In one outreach program, Sun Protection Outreach Teaching by Students (SPOTS), medical students visit middle and high schools to educate them about the importance of practicing sun protection. The program began as a collaboration between Saint Louis University and Washington University in St. Louis, but has expanded nationwide. Ms. Stevens described SPOTS as “a great way for medical students to present the information to middle and high school students in a way that is engaging and interactive.”
The researchers reported having no disclosures.
a cornerstone of skin cancer prevention, according to results from a systematic review.
“We know that skin cancer is one of the most common malignancies in the world, and sun protection methods such as sunscreen make it highly preventable,” first author Carly R. Stevens, a student at Tulane University, New Orleans, said in an interview. “This study demonstrates the adolescent populations that are most vulnerable to sun damage and how we can help mitigate their risk of developing skin cancer through education methods, such as Sun Protection Outreach Teaching by Students.”
Ms. Stevens and coauthors presented the findings during a poster session at the annual meeting of the Society for Pediatric Dermatology.
To investigate predictors of sunscreen use among high school students, they searched PubMed, Embase, and Web of Science using the terms (“sunscreen” or “SPF” or “sun protection”) and (“high school” or “teen” or “teenager” or “adolescent”) and limited the analysis to English studies reporting data on sunscreen use in U.S. high school students up to November 2021.
A total of 20 studies were included in the final review. The study populations ranged in number from 208 to 24,645. Of 11 studies that examined gender, all showed increased sunscreen use in females compared with males. Of five studies that examined age, all showed increased sunscreen use in younger adolescents, compared with their older counterparts.
Of four studies that examined the role of ethnicity on sunscreen use, White students were more likely to use sunscreen, compared with their peers of other ethnicities. “This may be due to perceived sun sensitivity, as [these four studies] also showed increased sunscreen use in populations that believed were more susceptible to sun damage,” the researchers wrote in their abstract.
In other findings, two studies that examined perceived self-efficacy concluded that higher levels of sunscreen use correlated with higher self-efficacy, while four studies concluded that high school students were more likely to use sunscreen if their parents encouraged them the wear it or if the parent used it themselves.
“With 40%-50% of ultraviolet damage being done before the age of 20, it’s crucial that we find ways to educate adolescents on the importance of sunscreen use and target those populations who were found to rarely use sunscreen in our study,” Ms. Stevens said.
In one outreach program, Sun Protection Outreach Teaching by Students (SPOTS), medical students visit middle and high schools to educate them about the importance of practicing sun protection. The program began as a collaboration between Saint Louis University and Washington University in St. Louis, but has expanded nationwide. Ms. Stevens described SPOTS as “a great way for medical students to present the information to middle and high school students in a way that is engaging and interactive.”
The researchers reported having no disclosures.
a cornerstone of skin cancer prevention, according to results from a systematic review.
“We know that skin cancer is one of the most common malignancies in the world, and sun protection methods such as sunscreen make it highly preventable,” first author Carly R. Stevens, a student at Tulane University, New Orleans, said in an interview. “This study demonstrates the adolescent populations that are most vulnerable to sun damage and how we can help mitigate their risk of developing skin cancer through education methods, such as Sun Protection Outreach Teaching by Students.”
Ms. Stevens and coauthors presented the findings during a poster session at the annual meeting of the Society for Pediatric Dermatology.
To investigate predictors of sunscreen use among high school students, they searched PubMed, Embase, and Web of Science using the terms (“sunscreen” or “SPF” or “sun protection”) and (“high school” or “teen” or “teenager” or “adolescent”) and limited the analysis to English studies reporting data on sunscreen use in U.S. high school students up to November 2021.
A total of 20 studies were included in the final review. The study populations ranged in number from 208 to 24,645. Of 11 studies that examined gender, all showed increased sunscreen use in females compared with males. Of five studies that examined age, all showed increased sunscreen use in younger adolescents, compared with their older counterparts.
Of four studies that examined the role of ethnicity on sunscreen use, White students were more likely to use sunscreen, compared with their peers of other ethnicities. “This may be due to perceived sun sensitivity, as [these four studies] also showed increased sunscreen use in populations that believed were more susceptible to sun damage,” the researchers wrote in their abstract.
In other findings, two studies that examined perceived self-efficacy concluded that higher levels of sunscreen use correlated with higher self-efficacy, while four studies concluded that high school students were more likely to use sunscreen if their parents encouraged them the wear it or if the parent used it themselves.
“With 40%-50% of ultraviolet damage being done before the age of 20, it’s crucial that we find ways to educate adolescents on the importance of sunscreen use and target those populations who were found to rarely use sunscreen in our study,” Ms. Stevens said.
In one outreach program, Sun Protection Outreach Teaching by Students (SPOTS), medical students visit middle and high schools to educate them about the importance of practicing sun protection. The program began as a collaboration between Saint Louis University and Washington University in St. Louis, but has expanded nationwide. Ms. Stevens described SPOTS as “a great way for medical students to present the information to middle and high school students in a way that is engaging and interactive.”
The researchers reported having no disclosures.
FROM SPD 2023
Most Americans in favor of regulated therapeutic psychedelics
It is a surprisingly large percentage, said officials at the University of California, Berkeley, Center for the Science of Psychedelics, which conducted the online survey of 1,500 registered voters in early June.
“That is a stunning number,” said Michael Pollan, cofounder of the center, and author of “How to Change Your Mind,” a book that explored potential uses of psychedelics.
In a briefing with reporters, Mr. Pollan said that he believes the large support base, in part, reflects campaigns that have “been successful by highlighting the effectiveness of psychedelics as therapy for mental illness.”
However, the poll also showed that 61% of voters said that they do not perceive psychedelics as “good for society,” and 69% do not perceive them as “something for people like me.”
These negative sentiments “suggest a fragile kind of support – the kind of support where you’re only hearing one side of the story,” said Mr. Pollan.
Still, poll respondents supported other potential policy changes, including 56% in support of the U.S. Food and Drug Administration vetting and approving psychedelics so they could be available by prescription.
50% have tried psychedelics
Almost 80% said that it should be easier for researchers to study psychedelics, and just under one-half said that they backed removing criminal penalties for personal use and possession.
The poll results also show that almost half of respondents had heard about psychedelics recently, with 48% saying they had heard about the drugs’ use in treating mental illness.
Respondents who were most familiar with and positive about psychedelics tended to be White, male, aged 30-50 years, liberal, highly educated, living in a Western state, and have little to no religious or spiritual practice.
Overall, 52% of survey respondents said that they or someone close to them had used a psychedelic, with almost half of that use coming in the past 5 years. Some 40% said that the use had been more than a decade ago.
Almost three-quarters of psychedelic use was reported as recreational, but the second-biggest category was therapeutic use at 39%. About one-third of respondents said that they or someone close to them had microdosed.
Conservative voters had lower levels of awareness and first-degree connection use as well as the least amount of support for regulated therapeutic use, with only 45% saying they would back such a policy, compared with 80% of liberal voters and 66% of moderate voters.
Black individuals were the least likely to be familiar with psychedelics: Just 29% said that they had heard a little or a lot about the drugs, compared with 39% of Latinx individuals and 51% of White individuals. And just one-quarter reported first-degree use, compared with half of Latinx individuals and 56% of White individuals.
Who should be eligible?
When asked who should be eligible for treatment with psychedelics, 80% said that they were comfortable with its use for those with terminal illnesses. More than two-thirds expressed comfort with the drugs being used to help veterans and people with treatment-resistant depression and anxiety.
Less than one-half of respondents said that psychedelics should be available to everyone older than 21 years. And voters seemed to be less inclined to say psychedelics should be used to treat people with addiction, with just 45% indicating that they were very or somewhat comfortable with that use.
Mr. Pollan said that reflects perhaps some lack of knowledge or education.
“The story about addiction and psychedelics hasn’t gotten out,” he said. “I kind of get that intuitively the idea of using a drug to treat a drug doesn’t sound right to a lot of people. But in fact, there’s good evidence it works,” Mr. Pollan said.
Respondents said that doctors, nurses, and scientists were the most trusted source of information about psychedelics, whereas the FDA received lower confidence. Law enforcement was least trusted by liberals and most trusted by conservatives.
Mr. Pollan noted the reversal in attitudes, with Americans mostly now looking to the scientific and medical establishment for guidance on psychedelics.
“We went from a counterculture drug to something that is being taken seriously by scientists as a potential therapy,” he said.
The poll’s margin of error was ± 2.5%.
A version of this article first appeared on Medscape.com.
It is a surprisingly large percentage, said officials at the University of California, Berkeley, Center for the Science of Psychedelics, which conducted the online survey of 1,500 registered voters in early June.
“That is a stunning number,” said Michael Pollan, cofounder of the center, and author of “How to Change Your Mind,” a book that explored potential uses of psychedelics.
In a briefing with reporters, Mr. Pollan said that he believes the large support base, in part, reflects campaigns that have “been successful by highlighting the effectiveness of psychedelics as therapy for mental illness.”
However, the poll also showed that 61% of voters said that they do not perceive psychedelics as “good for society,” and 69% do not perceive them as “something for people like me.”
These negative sentiments “suggest a fragile kind of support – the kind of support where you’re only hearing one side of the story,” said Mr. Pollan.
Still, poll respondents supported other potential policy changes, including 56% in support of the U.S. Food and Drug Administration vetting and approving psychedelics so they could be available by prescription.
50% have tried psychedelics
Almost 80% said that it should be easier for researchers to study psychedelics, and just under one-half said that they backed removing criminal penalties for personal use and possession.
The poll results also show that almost half of respondents had heard about psychedelics recently, with 48% saying they had heard about the drugs’ use in treating mental illness.
Respondents who were most familiar with and positive about psychedelics tended to be White, male, aged 30-50 years, liberal, highly educated, living in a Western state, and have little to no religious or spiritual practice.
Overall, 52% of survey respondents said that they or someone close to them had used a psychedelic, with almost half of that use coming in the past 5 years. Some 40% said that the use had been more than a decade ago.
Almost three-quarters of psychedelic use was reported as recreational, but the second-biggest category was therapeutic use at 39%. About one-third of respondents said that they or someone close to them had microdosed.
Conservative voters had lower levels of awareness and first-degree connection use as well as the least amount of support for regulated therapeutic use, with only 45% saying they would back such a policy, compared with 80% of liberal voters and 66% of moderate voters.
Black individuals were the least likely to be familiar with psychedelics: Just 29% said that they had heard a little or a lot about the drugs, compared with 39% of Latinx individuals and 51% of White individuals. And just one-quarter reported first-degree use, compared with half of Latinx individuals and 56% of White individuals.
Who should be eligible?
When asked who should be eligible for treatment with psychedelics, 80% said that they were comfortable with its use for those with terminal illnesses. More than two-thirds expressed comfort with the drugs being used to help veterans and people with treatment-resistant depression and anxiety.
Less than one-half of respondents said that psychedelics should be available to everyone older than 21 years. And voters seemed to be less inclined to say psychedelics should be used to treat people with addiction, with just 45% indicating that they were very or somewhat comfortable with that use.
Mr. Pollan said that reflects perhaps some lack of knowledge or education.
“The story about addiction and psychedelics hasn’t gotten out,” he said. “I kind of get that intuitively the idea of using a drug to treat a drug doesn’t sound right to a lot of people. But in fact, there’s good evidence it works,” Mr. Pollan said.
Respondents said that doctors, nurses, and scientists were the most trusted source of information about psychedelics, whereas the FDA received lower confidence. Law enforcement was least trusted by liberals and most trusted by conservatives.
Mr. Pollan noted the reversal in attitudes, with Americans mostly now looking to the scientific and medical establishment for guidance on psychedelics.
“We went from a counterculture drug to something that is being taken seriously by scientists as a potential therapy,” he said.
The poll’s margin of error was ± 2.5%.
A version of this article first appeared on Medscape.com.
It is a surprisingly large percentage, said officials at the University of California, Berkeley, Center for the Science of Psychedelics, which conducted the online survey of 1,500 registered voters in early June.
“That is a stunning number,” said Michael Pollan, cofounder of the center, and author of “How to Change Your Mind,” a book that explored potential uses of psychedelics.
In a briefing with reporters, Mr. Pollan said that he believes the large support base, in part, reflects campaigns that have “been successful by highlighting the effectiveness of psychedelics as therapy for mental illness.”
However, the poll also showed that 61% of voters said that they do not perceive psychedelics as “good for society,” and 69% do not perceive them as “something for people like me.”
These negative sentiments “suggest a fragile kind of support – the kind of support where you’re only hearing one side of the story,” said Mr. Pollan.
Still, poll respondents supported other potential policy changes, including 56% in support of the U.S. Food and Drug Administration vetting and approving psychedelics so they could be available by prescription.
50% have tried psychedelics
Almost 80% said that it should be easier for researchers to study psychedelics, and just under one-half said that they backed removing criminal penalties for personal use and possession.
The poll results also show that almost half of respondents had heard about psychedelics recently, with 48% saying they had heard about the drugs’ use in treating mental illness.
Respondents who were most familiar with and positive about psychedelics tended to be White, male, aged 30-50 years, liberal, highly educated, living in a Western state, and have little to no religious or spiritual practice.
Overall, 52% of survey respondents said that they or someone close to them had used a psychedelic, with almost half of that use coming in the past 5 years. Some 40% said that the use had been more than a decade ago.
Almost three-quarters of psychedelic use was reported as recreational, but the second-biggest category was therapeutic use at 39%. About one-third of respondents said that they or someone close to them had microdosed.
Conservative voters had lower levels of awareness and first-degree connection use as well as the least amount of support for regulated therapeutic use, with only 45% saying they would back such a policy, compared with 80% of liberal voters and 66% of moderate voters.
Black individuals were the least likely to be familiar with psychedelics: Just 29% said that they had heard a little or a lot about the drugs, compared with 39% of Latinx individuals and 51% of White individuals. And just one-quarter reported first-degree use, compared with half of Latinx individuals and 56% of White individuals.
Who should be eligible?
When asked who should be eligible for treatment with psychedelics, 80% said that they were comfortable with its use for those with terminal illnesses. More than two-thirds expressed comfort with the drugs being used to help veterans and people with treatment-resistant depression and anxiety.
Less than one-half of respondents said that psychedelics should be available to everyone older than 21 years. And voters seemed to be less inclined to say psychedelics should be used to treat people with addiction, with just 45% indicating that they were very or somewhat comfortable with that use.
Mr. Pollan said that reflects perhaps some lack of knowledge or education.
“The story about addiction and psychedelics hasn’t gotten out,” he said. “I kind of get that intuitively the idea of using a drug to treat a drug doesn’t sound right to a lot of people. But in fact, there’s good evidence it works,” Mr. Pollan said.
Respondents said that doctors, nurses, and scientists were the most trusted source of information about psychedelics, whereas the FDA received lower confidence. Law enforcement was least trusted by liberals and most trusted by conservatives.
Mr. Pollan noted the reversal in attitudes, with Americans mostly now looking to the scientific and medical establishment for guidance on psychedelics.
“We went from a counterculture drug to something that is being taken seriously by scientists as a potential therapy,” he said.
The poll’s margin of error was ± 2.5%.
A version of this article first appeared on Medscape.com.
High-intensity interval training before major surgery may boost postoperative outcomes
TOPLINE:
It cuts the risk of postoperative complications and may shorten hospital length of stay and improve postoperative quality of life.
METHODOLOGY:
Evidence suggests CRF – which improves physical and cognitive function and is associated with a reduction in cardiovascular risk – can be enhanced before major surgeries, but reported postoperative outcomes in previous reviews have been inconsistent.
In the study, HIIT involved repeated aerobic high-intensity exercise intervals at about 80% of maximum heart rate, followed by active recovery.
The meta-analysis included 12 studies with 832 patients (mean age, 67) that compared preoperative HIIT – supervised at hospitals, gyms, or community or physical therapy centers, or unsupervised at home – with standard care for patients slated for major surgery, including liver, lung, colorectal, urologic, and mixed major abdominal operations.
The primary outcome was change in CRF by peak VO2 or 6-minute walk test; other endpoints included change in endurance time and postoperative outcomes.
TAKEAWAY:
Preoperative HIIT (median total, 160 minutes; range, 80-240 minutes; intense exercise during 6-40 sessions) was associated with an increase in peak oxygen consumption (VO2 peak) by 2.59 mL/kg/min (95% confidence interval, 1.52-3.65 mL/kg/min; P < .001), compared with standard care, which represents about a 10% increase in CRF.
In eight studies that involved 770 patients, there was moderate evidence that preoperative HIIT cut the odds ratio for postoperative complications by more than half (OR, 0.44; 95% CI, 0.32-0.60; P < .001); there was a similar apparent benefit in an analysis that was limited to patients who were slated for abdominal surgery (OR, 0.45; 95% CI, 0.29-0.68; P < .001).
An analysis that was limited to studies that reported hospital length of stay showed a clinically relevant but nonsignificant 3-day reduction among patients in the HIIT groups.
Most quality of life assessments did not show post-HIIT improvements; some showed a significant benefit 6 weeks after surgery.
IN PRACTICE:
The results suggest preoperative HIIT may improve postoperative outcomes. By extension, it could be cost-effective and “should be included in prehabilitation programs,” the report states.
SOURCE:
The study was carried out by Kari Clifford, PhD, Otago Medical School, University of Otago, Dunedin, New Zealand, and colleagues. It was published online June 30, 2023, in JAMA Network Open.
LIMITATIONS:
Included studies were heterogeneous in methodology; for example, HIIT definitions and protocols varied across almost every study. Data reporting was incomplete, the samples sizes in the studies were limited, and patients could not be blinded to their intervention. The patients could not be stratified on the basis of frailty. There were limited HIIT data from patients who underwent orthopedic surgeries.
DISCLOSURES:
The study received funding from the University of Otago. The authors reported no conflicts.
A version of this article first appeared on Medscape.com.
TOPLINE:
It cuts the risk of postoperative complications and may shorten hospital length of stay and improve postoperative quality of life.
METHODOLOGY:
Evidence suggests CRF – which improves physical and cognitive function and is associated with a reduction in cardiovascular risk – can be enhanced before major surgeries, but reported postoperative outcomes in previous reviews have been inconsistent.
In the study, HIIT involved repeated aerobic high-intensity exercise intervals at about 80% of maximum heart rate, followed by active recovery.
The meta-analysis included 12 studies with 832 patients (mean age, 67) that compared preoperative HIIT – supervised at hospitals, gyms, or community or physical therapy centers, or unsupervised at home – with standard care for patients slated for major surgery, including liver, lung, colorectal, urologic, and mixed major abdominal operations.
The primary outcome was change in CRF by peak VO2 or 6-minute walk test; other endpoints included change in endurance time and postoperative outcomes.
TAKEAWAY:
Preoperative HIIT (median total, 160 minutes; range, 80-240 minutes; intense exercise during 6-40 sessions) was associated with an increase in peak oxygen consumption (VO2 peak) by 2.59 mL/kg/min (95% confidence interval, 1.52-3.65 mL/kg/min; P < .001), compared with standard care, which represents about a 10% increase in CRF.
In eight studies that involved 770 patients, there was moderate evidence that preoperative HIIT cut the odds ratio for postoperative complications by more than half (OR, 0.44; 95% CI, 0.32-0.60; P < .001); there was a similar apparent benefit in an analysis that was limited to patients who were slated for abdominal surgery (OR, 0.45; 95% CI, 0.29-0.68; P < .001).
An analysis that was limited to studies that reported hospital length of stay showed a clinically relevant but nonsignificant 3-day reduction among patients in the HIIT groups.
Most quality of life assessments did not show post-HIIT improvements; some showed a significant benefit 6 weeks after surgery.
IN PRACTICE:
The results suggest preoperative HIIT may improve postoperative outcomes. By extension, it could be cost-effective and “should be included in prehabilitation programs,” the report states.
SOURCE:
The study was carried out by Kari Clifford, PhD, Otago Medical School, University of Otago, Dunedin, New Zealand, and colleagues. It was published online June 30, 2023, in JAMA Network Open.
LIMITATIONS:
Included studies were heterogeneous in methodology; for example, HIIT definitions and protocols varied across almost every study. Data reporting was incomplete, the samples sizes in the studies were limited, and patients could not be blinded to their intervention. The patients could not be stratified on the basis of frailty. There were limited HIIT data from patients who underwent orthopedic surgeries.
DISCLOSURES:
The study received funding from the University of Otago. The authors reported no conflicts.
A version of this article first appeared on Medscape.com.
TOPLINE:
It cuts the risk of postoperative complications and may shorten hospital length of stay and improve postoperative quality of life.
METHODOLOGY:
Evidence suggests CRF – which improves physical and cognitive function and is associated with a reduction in cardiovascular risk – can be enhanced before major surgeries, but reported postoperative outcomes in previous reviews have been inconsistent.
In the study, HIIT involved repeated aerobic high-intensity exercise intervals at about 80% of maximum heart rate, followed by active recovery.
The meta-analysis included 12 studies with 832 patients (mean age, 67) that compared preoperative HIIT – supervised at hospitals, gyms, or community or physical therapy centers, or unsupervised at home – with standard care for patients slated for major surgery, including liver, lung, colorectal, urologic, and mixed major abdominal operations.
The primary outcome was change in CRF by peak VO2 or 6-minute walk test; other endpoints included change in endurance time and postoperative outcomes.
TAKEAWAY:
Preoperative HIIT (median total, 160 minutes; range, 80-240 minutes; intense exercise during 6-40 sessions) was associated with an increase in peak oxygen consumption (VO2 peak) by 2.59 mL/kg/min (95% confidence interval, 1.52-3.65 mL/kg/min; P < .001), compared with standard care, which represents about a 10% increase in CRF.
In eight studies that involved 770 patients, there was moderate evidence that preoperative HIIT cut the odds ratio for postoperative complications by more than half (OR, 0.44; 95% CI, 0.32-0.60; P < .001); there was a similar apparent benefit in an analysis that was limited to patients who were slated for abdominal surgery (OR, 0.45; 95% CI, 0.29-0.68; P < .001).
An analysis that was limited to studies that reported hospital length of stay showed a clinically relevant but nonsignificant 3-day reduction among patients in the HIIT groups.
Most quality of life assessments did not show post-HIIT improvements; some showed a significant benefit 6 weeks after surgery.
IN PRACTICE:
The results suggest preoperative HIIT may improve postoperative outcomes. By extension, it could be cost-effective and “should be included in prehabilitation programs,” the report states.
SOURCE:
The study was carried out by Kari Clifford, PhD, Otago Medical School, University of Otago, Dunedin, New Zealand, and colleagues. It was published online June 30, 2023, in JAMA Network Open.
LIMITATIONS:
Included studies were heterogeneous in methodology; for example, HIIT definitions and protocols varied across almost every study. Data reporting was incomplete, the samples sizes in the studies were limited, and patients could not be blinded to their intervention. The patients could not be stratified on the basis of frailty. There were limited HIIT data from patients who underwent orthopedic surgeries.
DISCLOSURES:
The study received funding from the University of Otago. The authors reported no conflicts.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN