User login
For MD-IQ use only
What's your diagnosis?
Whipple's disease
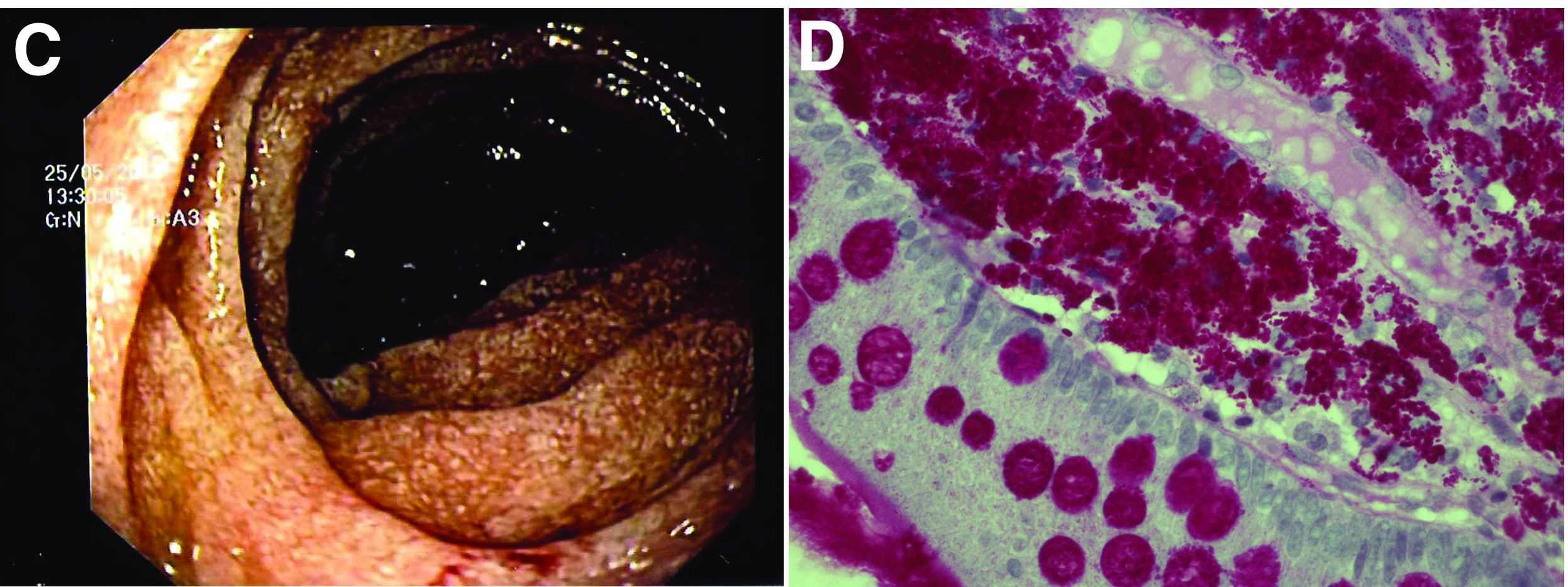
The ultrasound features were highly suggestive of malabsorption, a hypothesis that was supported by the laboratory findings. Celiac disease, one of the most common causes of malabsorption, was excluded by serology tests. Esophagogastroduodenoscopy was therefore repeated: The mucosa of the distal first part and second part of the duodenum appeared completely covered with tiny white spots (Figure C). Histologic examination revealed that the mucosal architecture of the villi was altered by the presence of infiltrates of macrophages with wide cytoplasm filled with round periodic acid-Schiff (PAS)-positive inclusions, associated to aggregates of neutrophils attacking the epithelium (Figure D). These histologic findings are consistent with Whipple's disease.
Whipple's disease is a chronic infectious disease caused by a gram-positive ubiquitous bacterium named Tropheryma whipplei. In predisposed subjects with an insufficient T-helper response, for example, those undergoing treatment with tumor necrosis factor-alpha inhibitors as in our patient, T. whipplei is able to survive and replicate inside the macrophages of the intestinal mucosa and to spread to other organs.1 Whipple's disease can thus manifest as a multisystemic disease or as a single-organ disease with extraintestinal involvement (e.g., central nervous system, eyes, heart, or lung). The classic form is characterized by weight loss, diarrhea, abdominal pain, and signs of malabsorption, typically preceded by a history of arthralgia. The arthralgia is often misdiagnosed as a form of rheumatoid arthritis and therefore treated with immunosuppressant therapy, which favors the onset of the classic intestinal symptoms.
In the literature, few case reports describe the ultrasound findings in patients with Whipple's disease. The most frequent sonographic features include small-bowel dilatation with wall thickening, the presence of peri-intestinal fluid effusion and mesenteric and retroperitoneal lymphadenopathy.2,3
The final diagnosis relies on intestinal biopsy and the histologic finding of foamy macrophages containing large amounts of diastase-resistant PAS-positive particles in the lamina propria of the duodenum, jejunum, ileum, or gastric antral region.
The diagnosis, particularly in cases of extraintestinal involvement, can be confirmed by polymerase chain reaction positivity for T. whipplei in the examined tissue.
Therapy consists of the administration of ceftriaxone (2 g IV once daily) for 2 weeks followed by oral therapy with trimethoprim-sulfamethoxazole for 1 year.
References
1. Schneider T et al. Whipple's disease: New aspects of pathogenesis and treatment. Lancet Infect Dis. 2008;8:179-90.
2. Brindicci D et al. Ultrasonic findings in Whipple's disease. J Clin Ultrasound. 1984;12:286-8.
3. Neye H et al. Der Morbus Whipple's Disease - A rare intestinal disease and its sonographic characteristics. Ultraschall Med. 2012;33(04):314-5.
Whipple's disease
The ultrasound features were highly suggestive of malabsorption, a hypothesis that was supported by the laboratory findings. Celiac disease, one of the most common causes of malabsorption, was excluded by serology tests. Esophagogastroduodenoscopy was therefore repeated: The mucosa of the distal first part and second part of the duodenum appeared completely covered with tiny white spots (Figure C). Histologic examination revealed that the mucosal architecture of the villi was altered by the presence of infiltrates of macrophages with wide cytoplasm filled with round periodic acid-Schiff (PAS)-positive inclusions, associated to aggregates of neutrophils attacking the epithelium (Figure D). These histologic findings are consistent with Whipple's disease.

Whipple's disease is a chronic infectious disease caused by a gram-positive ubiquitous bacterium named Tropheryma whipplei. In predisposed subjects with an insufficient T-helper response, for example, those undergoing treatment with tumor necrosis factor-alpha inhibitors as in our patient, T. whipplei is able to survive and replicate inside the macrophages of the intestinal mucosa and to spread to other organs.1 Whipple's disease can thus manifest as a multisystemic disease or as a single-organ disease with extraintestinal involvement (e.g., central nervous system, eyes, heart, or lung). The classic form is characterized by weight loss, diarrhea, abdominal pain, and signs of malabsorption, typically preceded by a history of arthralgia. The arthralgia is often misdiagnosed as a form of rheumatoid arthritis and therefore treated with immunosuppressant therapy, which favors the onset of the classic intestinal symptoms.
In the literature, few case reports describe the ultrasound findings in patients with Whipple's disease. The most frequent sonographic features include small-bowel dilatation with wall thickening, the presence of peri-intestinal fluid effusion and mesenteric and retroperitoneal lymphadenopathy.2,3
The final diagnosis relies on intestinal biopsy and the histologic finding of foamy macrophages containing large amounts of diastase-resistant PAS-positive particles in the lamina propria of the duodenum, jejunum, ileum, or gastric antral region.
The diagnosis, particularly in cases of extraintestinal involvement, can be confirmed by polymerase chain reaction positivity for T. whipplei in the examined tissue.
Therapy consists of the administration of ceftriaxone (2 g IV once daily) for 2 weeks followed by oral therapy with trimethoprim-sulfamethoxazole for 1 year.
References
1. Schneider T et al. Whipple's disease: New aspects of pathogenesis and treatment. Lancet Infect Dis. 2008;8:179-90.
2. Brindicci D et al. Ultrasonic findings in Whipple's disease. J Clin Ultrasound. 1984;12:286-8.
3. Neye H et al. Der Morbus Whipple's Disease - A rare intestinal disease and its sonographic characteristics. Ultraschall Med. 2012;33(04):314-5.
Whipple's disease
The ultrasound features were highly suggestive of malabsorption, a hypothesis that was supported by the laboratory findings. Celiac disease, one of the most common causes of malabsorption, was excluded by serology tests. Esophagogastroduodenoscopy was therefore repeated: The mucosa of the distal first part and second part of the duodenum appeared completely covered with tiny white spots (Figure C). Histologic examination revealed that the mucosal architecture of the villi was altered by the presence of infiltrates of macrophages with wide cytoplasm filled with round periodic acid-Schiff (PAS)-positive inclusions, associated to aggregates of neutrophils attacking the epithelium (Figure D). These histologic findings are consistent with Whipple's disease.

Whipple's disease is a chronic infectious disease caused by a gram-positive ubiquitous bacterium named Tropheryma whipplei. In predisposed subjects with an insufficient T-helper response, for example, those undergoing treatment with tumor necrosis factor-alpha inhibitors as in our patient, T. whipplei is able to survive and replicate inside the macrophages of the intestinal mucosa and to spread to other organs.1 Whipple's disease can thus manifest as a multisystemic disease or as a single-organ disease with extraintestinal involvement (e.g., central nervous system, eyes, heart, or lung). The classic form is characterized by weight loss, diarrhea, abdominal pain, and signs of malabsorption, typically preceded by a history of arthralgia. The arthralgia is often misdiagnosed as a form of rheumatoid arthritis and therefore treated with immunosuppressant therapy, which favors the onset of the classic intestinal symptoms.
In the literature, few case reports describe the ultrasound findings in patients with Whipple's disease. The most frequent sonographic features include small-bowel dilatation with wall thickening, the presence of peri-intestinal fluid effusion and mesenteric and retroperitoneal lymphadenopathy.2,3
The final diagnosis relies on intestinal biopsy and the histologic finding of foamy macrophages containing large amounts of diastase-resistant PAS-positive particles in the lamina propria of the duodenum, jejunum, ileum, or gastric antral region.
The diagnosis, particularly in cases of extraintestinal involvement, can be confirmed by polymerase chain reaction positivity for T. whipplei in the examined tissue.
Therapy consists of the administration of ceftriaxone (2 g IV once daily) for 2 weeks followed by oral therapy with trimethoprim-sulfamethoxazole for 1 year.
References
1. Schneider T et al. Whipple's disease: New aspects of pathogenesis and treatment. Lancet Infect Dis. 2008;8:179-90.
2. Brindicci D et al. Ultrasonic findings in Whipple's disease. J Clin Ultrasound. 1984;12:286-8.
3. Neye H et al. Der Morbus Whipple's Disease - A rare intestinal disease and its sonographic characteristics. Ultraschall Med. 2012;33(04):314-5.
67-year-old woman presented with a year-long history of general malaise, low-grade fever, diarrhea, and a 20-kg weight loss. She had a history of hypertension and depressive disorder. In the previous 4 years, she had undergone several rheumatologic examinations for polyarthritis and, having been diagnosed with seronegative rheumatoid arthritis, she had been treated with steroids, methotrexate, and etanercept, with little benefit.
Recent laboratory tests showed: hemoglobin, 8.3 g/dL; mean corpuscular volume, 70 fL; erythrocyte sedimentation rate, 78; and C-reactive protein, 6.4 mg/dL. To evaluate the microcytic anemia and the diarrhea, endoscopic investigations had been performed a few months earlier. Esophagogastroduodenoscopy showed villous atrophy at the level of DII; histology was compatible with intramucosal xanthoma. There were no pathologic findings at colonoscopy. The situation had not been further investigated.
At presentation, the physical examination revealed lower-limb edema, skin and mucosal pallor, and a body mass index of 17.4 kg/m2. Laboratory tests showed microcytic anemia (hemoglobin, 10.0 g/dL; mean corpuscular volume, 74 fL), increased acute-phase proteins (erythrocyte sedimentation rate, 59; C-reactive protein, 8.53 mg/dL), and malabsorption (albumin, 2.5 g/dL; multiple electrolytes deficiencies including iron, vitamin A, and vitamin D deficiency).
Abdominal ultrasound examination revealed three small lymph nodes in the periaortic region (maximum diameter, 10 mm), marked mesenteric and ileal wall thickening, mild jejunal wall thickening, an increased number of connivent valves, and a mild amount of peri-intestinal fluid effusion (Figure A, B).

What is the likely diagnosis and the appropriate treatment?
AHA targets physician burnout in academic CV medicine
In a new scientific statement, the American Heart Association highlights the unique drivers of burnout in academic cardiovascular medicine physicians and proposes system-level and personal interventions to support individual wellness in this setting.
“The future cardiovascular health of Americans relies on a well-trained and experienced physician workforce created by rigorous academic medical training,” the writing group says in Circulation.
“Cardiovascular physicians pursuing careers in academic medicine are critical to continuing this mission, which includes providing clinical care for common and increasingly complex disease, educating and training the next generation of physicians/health care workers, and pursuing scientific discovery and innovation to treat and cure disease,” write Elisa Bradley, Penn State Health Heart and Vascular Institute, Hershey, Pa., and coauthors.
Given the multitasking nature of academic medicine, exhaustion and burnout uniquely threaten future and early career academic physicians, they say.
Drivers of burnout in this setting include productivity-driven compensation models that force competition for time between clinical care and academics; the requirement for promotion in systems that have not evolved to consider combined clinical and academic expectations; and distinct expectations based on faculty pathway, such as grant funding and publications.
In addition, at the early career and fellow-in-training level, drivers of burnout also include significant changes in personal and family life, coupled with long hours and high clinical and research demands, as well as financial strain and educational debt.
Many of the drivers of burnout in academic medicine are external and beyond the control of a single individual. Therefore, proposed solutions must be largely at the level of organizations, institutions, and government, the writing group says.
These solutions include appropriate mentorship, goal planning, efficiency in the workplace, time management and time “protection,” and manageable schedules.
Professional satisfaction “should be a shared responsibility between the clinician and the institution. Each must adapt their values to find a middle ground that meets the needs of both, recognizing that health care is both personal and a business,” the writing group says.
“Interventions to support efficiency of practice and a culture of wellness span normalizing and supporting flexible work environments to enhancing clinical support,” they add.
To enhance flexible clinical environments, organizations should consider “float teams” to provide care to bridge gaps when a physician is not available, job sharing and flexible hours, and telemedicine, the writing group says.
At the individual level, academic cardiovascular professionals should build individualized strategies to combat fatigue and to promote wellness, focusing on self-care and healthy habits (adequate sleep, healthy nutrition, exercise, outside interests, meaningful social relationships), they advise.
With help, “young academicians can look forward to a fulfilling and long career in academic cardiovascular medicine,” they conclude.
This research had no commercial funding. Members of the writing group reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a new scientific statement, the American Heart Association highlights the unique drivers of burnout in academic cardiovascular medicine physicians and proposes system-level and personal interventions to support individual wellness in this setting.
“The future cardiovascular health of Americans relies on a well-trained and experienced physician workforce created by rigorous academic medical training,” the writing group says in Circulation.
“Cardiovascular physicians pursuing careers in academic medicine are critical to continuing this mission, which includes providing clinical care for common and increasingly complex disease, educating and training the next generation of physicians/health care workers, and pursuing scientific discovery and innovation to treat and cure disease,” write Elisa Bradley, Penn State Health Heart and Vascular Institute, Hershey, Pa., and coauthors.
Given the multitasking nature of academic medicine, exhaustion and burnout uniquely threaten future and early career academic physicians, they say.
Drivers of burnout in this setting include productivity-driven compensation models that force competition for time between clinical care and academics; the requirement for promotion in systems that have not evolved to consider combined clinical and academic expectations; and distinct expectations based on faculty pathway, such as grant funding and publications.
In addition, at the early career and fellow-in-training level, drivers of burnout also include significant changes in personal and family life, coupled with long hours and high clinical and research demands, as well as financial strain and educational debt.
Many of the drivers of burnout in academic medicine are external and beyond the control of a single individual. Therefore, proposed solutions must be largely at the level of organizations, institutions, and government, the writing group says.
These solutions include appropriate mentorship, goal planning, efficiency in the workplace, time management and time “protection,” and manageable schedules.
Professional satisfaction “should be a shared responsibility between the clinician and the institution. Each must adapt their values to find a middle ground that meets the needs of both, recognizing that health care is both personal and a business,” the writing group says.
“Interventions to support efficiency of practice and a culture of wellness span normalizing and supporting flexible work environments to enhancing clinical support,” they add.
To enhance flexible clinical environments, organizations should consider “float teams” to provide care to bridge gaps when a physician is not available, job sharing and flexible hours, and telemedicine, the writing group says.
At the individual level, academic cardiovascular professionals should build individualized strategies to combat fatigue and to promote wellness, focusing on self-care and healthy habits (adequate sleep, healthy nutrition, exercise, outside interests, meaningful social relationships), they advise.
With help, “young academicians can look forward to a fulfilling and long career in academic cardiovascular medicine,” they conclude.
This research had no commercial funding. Members of the writing group reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a new scientific statement, the American Heart Association highlights the unique drivers of burnout in academic cardiovascular medicine physicians and proposes system-level and personal interventions to support individual wellness in this setting.
“The future cardiovascular health of Americans relies on a well-trained and experienced physician workforce created by rigorous academic medical training,” the writing group says in Circulation.
“Cardiovascular physicians pursuing careers in academic medicine are critical to continuing this mission, which includes providing clinical care for common and increasingly complex disease, educating and training the next generation of physicians/health care workers, and pursuing scientific discovery and innovation to treat and cure disease,” write Elisa Bradley, Penn State Health Heart and Vascular Institute, Hershey, Pa., and coauthors.
Given the multitasking nature of academic medicine, exhaustion and burnout uniquely threaten future and early career academic physicians, they say.
Drivers of burnout in this setting include productivity-driven compensation models that force competition for time between clinical care and academics; the requirement for promotion in systems that have not evolved to consider combined clinical and academic expectations; and distinct expectations based on faculty pathway, such as grant funding and publications.
In addition, at the early career and fellow-in-training level, drivers of burnout also include significant changes in personal and family life, coupled with long hours and high clinical and research demands, as well as financial strain and educational debt.
Many of the drivers of burnout in academic medicine are external and beyond the control of a single individual. Therefore, proposed solutions must be largely at the level of organizations, institutions, and government, the writing group says.
These solutions include appropriate mentorship, goal planning, efficiency in the workplace, time management and time “protection,” and manageable schedules.
Professional satisfaction “should be a shared responsibility between the clinician and the institution. Each must adapt their values to find a middle ground that meets the needs of both, recognizing that health care is both personal and a business,” the writing group says.
“Interventions to support efficiency of practice and a culture of wellness span normalizing and supporting flexible work environments to enhancing clinical support,” they add.
To enhance flexible clinical environments, organizations should consider “float teams” to provide care to bridge gaps when a physician is not available, job sharing and flexible hours, and telemedicine, the writing group says.
At the individual level, academic cardiovascular professionals should build individualized strategies to combat fatigue and to promote wellness, focusing on self-care and healthy habits (adequate sleep, healthy nutrition, exercise, outside interests, meaningful social relationships), they advise.
With help, “young academicians can look forward to a fulfilling and long career in academic cardiovascular medicine,” they conclude.
This research had no commercial funding. Members of the writing group reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Gastroenterology Data Trends 2022
Inside this issue:
- The Impact of COVID-19 on Colorectal Cancer Screening Programs
Rachel B. Issaka, MD, MAS - Early Onset Colorectal Cancer: Trends in Incidence and Screening
Aasma Shaukat, MD, MPH, AGAF - Diversity in the Gastroenterology Workforce and its Implications for Patients
Sandra M. Quezada, MD, MS, AGAF - Trends in Surveillance and Management of Dysplasia in IBD
Joseph D. Feuerstein, MD, AGAF - Environmental Factors in IBD: Diet and Stress
Ashwin Ananthakrishnan, MBBS, MPH - Evolving Therapeutic Goals in Crohn’s Disease Management
Ryan Ungaro, MD, MS - Switching to Disposable Duodenoscopes: Risks and Rewards
Rajesh N. Keswani, MD, MS - Increasing Surveillance Programs and Expanding Treatment Options in HCC
Amit Singal, MD, MS - Achalasia Remains a Challenging Disorder for the Community Gastroenterologist
Benson T. Massey, MD
Inside this issue:
- The Impact of COVID-19 on Colorectal Cancer Screening Programs
Rachel B. Issaka, MD, MAS - Early Onset Colorectal Cancer: Trends in Incidence and Screening
Aasma Shaukat, MD, MPH, AGAF - Diversity in the Gastroenterology Workforce and its Implications for Patients
Sandra M. Quezada, MD, MS, AGAF - Trends in Surveillance and Management of Dysplasia in IBD
Joseph D. Feuerstein, MD, AGAF - Environmental Factors in IBD: Diet and Stress
Ashwin Ananthakrishnan, MBBS, MPH - Evolving Therapeutic Goals in Crohn’s Disease Management
Ryan Ungaro, MD, MS - Switching to Disposable Duodenoscopes: Risks and Rewards
Rajesh N. Keswani, MD, MS - Increasing Surveillance Programs and Expanding Treatment Options in HCC
Amit Singal, MD, MS - Achalasia Remains a Challenging Disorder for the Community Gastroenterologist
Benson T. Massey, MD
Inside this issue:
- The Impact of COVID-19 on Colorectal Cancer Screening Programs
Rachel B. Issaka, MD, MAS - Early Onset Colorectal Cancer: Trends in Incidence and Screening
Aasma Shaukat, MD, MPH, AGAF - Diversity in the Gastroenterology Workforce and its Implications for Patients
Sandra M. Quezada, MD, MS, AGAF - Trends in Surveillance and Management of Dysplasia in IBD
Joseph D. Feuerstein, MD, AGAF - Environmental Factors in IBD: Diet and Stress
Ashwin Ananthakrishnan, MBBS, MPH - Evolving Therapeutic Goals in Crohn’s Disease Management
Ryan Ungaro, MD, MS - Switching to Disposable Duodenoscopes: Risks and Rewards
Rajesh N. Keswani, MD, MS - Increasing Surveillance Programs and Expanding Treatment Options in HCC
Amit Singal, MD, MS - Achalasia Remains a Challenging Disorder for the Community Gastroenterologist
Benson T. Massey, MD
Novel approach brings hospice-bound MM patient into remission
In a case that researchers hope might pave the way for similar responses, a hospice-bound relapsed/refractory multiple myeloma (RRMM) patient who relapsed after chimeric antigen receptor (CAR) T-cell therapy was brought back into remission with the help of next-generation genomic sequencing, targeted molecular analysis and a novel combination of MAP kinase (MAPK)–inhibiting drugs.
“We have shown that comprehensive molecular profiling of advanced myeloma patients may provide critical information to guide treatment beyond standard of care,” senior author Alessandro Lagana, PhD, of the Tisch Cancer Institute, Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“This represents proof of concept that, while not curative, targeted molecules may serve as potential bridging therapies to clinical trial enrollment,” the authors further report in the case study, published recently in the Journal of Hematology & Oncology.
The use of B-cell maturation antigen (BCMA) CAR T-cell therapy approaches has transformed the treatment of multiple myeloma and leukemias, resulting in high response rates. However, most patients ultimately relapse, and no clear treatment options beyond CAR T therapy are established.
Such was the case for a 61-year old patient described in the study, who had relapsed 6 months after undergoing anti-BCMA CAR T-cell therapy and progressed after being salvaged for a short period with autologous stem cell transplantation. The patient had developed skin extramedullary disease, manifested as subcutaneous nodules.
“The subcutaneous skin lesions in lower extremities made him [ineligible] for another clinical trial and left him with no options,” Dr. Lagana said.
Using next-generation whole-exome sequencing, Dr. Lagana and colleagues had observed that a previously identified BRAF V600E–dominant subclone had persisted, despite the CAR T-cell treatment, in the patient’s bone marrow and cutaneous plasmacytoma.
The finding was not uncommon. More than half of RRMM patients (about 53%) show emerging clones with mutations within the MAPK signaling pathway, and in about 7% of patients, those include BRAF V600E, which can be targeted, the authors noted.
Further assessment of the patient’s CD138-positive MM cells using western blot signaling pathway analysis looking at DNA and RNA markers did indeed show an increase in MAPK signaling as a consequence of the mutation. This suggested a potential benefit of triple MAPK inhibition, compared with standard strategies.
Based on that information and on insights the researchers had gained from previous research, they implemented the novel, orally administered triple-combination treatment strategy, consisting of monomeric inhibition of BRAF dabrafenib (100 mg, twice daily), as well as dimeric inhibition with the multi–kinase inhibitor regorafenib (40 mg, once daily) and a MEK inhibitor (trametinib, 1.5 mg, for 21/28 days daily).
Of note, previous efforts using only monomeric inhibition of BRAF have not shown much success, but early data has shown some potential, with the inclusion of dimeric inhibition.
“Monomeric inhibition of BRAF has been attempted in patients with V600E, but the efficacy has been limited, likely due to feedback activation of the MAPK pathway via induction of BRAF dimer formation,” Dr. Lagana explained.
Meanwhile, “previous in vitro data from our colleagues at Mount Sinai has shown that inhibition of both monomeric and dimeric forms of BRAF in combination with MEK inhibition can overcome the negative feedback and lead to more efficacious and tolerable treatment,” he said.
With the treatment, the patient achieved a very good partial response for 110 days, with prompt reduction of the subcutaneous skin lesions and an 80% reduction in lambda free light chain (27.5 mg/L).
The triple-drug combination was well tolerated with minimal side effects, primarily involving grade 1 fatigue, and the patient was able to carry out activities of daily living and return to work.
“The triple inhibition allowed us to use less of each drug, which resulted in a well-tolerated regimen without any significant side effects,” Dr. Lagana said.
While the patient relapsed about 3 months later, there was, importantly, no recurrence of the subcutaneous nodules.
“We believe that the triple MAPK inhibition completely eradicated the disease clones driving the extramedullary disease,” Dr. Lagana said.
The therapy meanwhile enabled the patient to bridge to a new clinical trial, where he went into complete remission, and still was as of Sept. 29.
“To our knowledge, this was the first reported successful case of this treatment in an RRMM patient,” Dr. Lagana explained.
Case suggests ‘hope’ for relapsing patients
Importantly, currently many patients in the same position may wind up going to hospice, until such targeted medicine gains momentum, coauthor Samir Parekh, MD, a professor of hematology-oncology at the Hess Center for Science and Medicine, Icahn School of Medicine at Mount Sinai, said in an interview.
“As precision medicine is in its infancy in myeloma, these patients are not routinely sequenced for drug options that may be identified by next-generation sequencing,” said Dr. Parekh.
But for clinicians, the message of this case should be that “there is hope for patients relapsing after CAR T,” he added.
“Precision medicine approaches may be applicable even for this relapsed patient population,” he added. “MAP kinase mutations are common and drugs targeting them may be useful in myeloma.”
Noting that “the infrastructure to test and guide application of these therapies needs to be developed for myeloma, Dr. Parekh predicted that, “in the future, more effective MAPK inhibitors and other mutation or RNA-seq guided therapies will be applicable and hopefully provide more durable remissions.”
Approach may help address unmet need
Until then, however, treatment for patients who relapse after CAR-T and BCMA-targeted therapies has emerged as a significant unmet need. Therefore, this case highlights an important potential strategy, said Hans Lee, MD, an associate professor in the department of lymphoma/myeloma, division of cancer medicine, University of Texas MD Anderson Cancer Center, Houston, commenting on the study.
“This case report provides impetus for oncologists to strongly consider performing next-generation sequencing on myeloma tumor samples to look for potential actionable mutations, such as those in the MAPK pathway – which are common in myeloma,” he said. “With limited treatment options in the post–CAR T and post-BCMA setting, identifying such actional mutations may at least provide a bridge to other effective therapies available through clinical trials such as this patient’s case.”
Dr. Lee noted that key caveats include the fact that most physicians currently don’t have access to the type of next-generation sequencing and drug sensitivity testing used in the study.
Nevertheless, considering the limited options in the post–CAR T and post-BCMA setting, “the successful use of triple MAPK pathway inhibition through monomeric and dimeric inhibition of BRAF and MEK inhibition warrants further study in multiple myeloma in a clinical trial,” he said.
Dr. Lagana and associates are doing just that.
“We are about to launch the clinical trial, where we will match advanced RRMM patients with potential targeted treatments using different DNA and RNA markers,” Dr. Lagana said.
Dr. Lagana and Dr. Parekh had no disclosures to report. Three study coauthors reported receiving research grants or consulting fees from numerous pharmaceutical companies.
In a case that researchers hope might pave the way for similar responses, a hospice-bound relapsed/refractory multiple myeloma (RRMM) patient who relapsed after chimeric antigen receptor (CAR) T-cell therapy was brought back into remission with the help of next-generation genomic sequencing, targeted molecular analysis and a novel combination of MAP kinase (MAPK)–inhibiting drugs.
“We have shown that comprehensive molecular profiling of advanced myeloma patients may provide critical information to guide treatment beyond standard of care,” senior author Alessandro Lagana, PhD, of the Tisch Cancer Institute, Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“This represents proof of concept that, while not curative, targeted molecules may serve as potential bridging therapies to clinical trial enrollment,” the authors further report in the case study, published recently in the Journal of Hematology & Oncology.
The use of B-cell maturation antigen (BCMA) CAR T-cell therapy approaches has transformed the treatment of multiple myeloma and leukemias, resulting in high response rates. However, most patients ultimately relapse, and no clear treatment options beyond CAR T therapy are established.
Such was the case for a 61-year old patient described in the study, who had relapsed 6 months after undergoing anti-BCMA CAR T-cell therapy and progressed after being salvaged for a short period with autologous stem cell transplantation. The patient had developed skin extramedullary disease, manifested as subcutaneous nodules.
“The subcutaneous skin lesions in lower extremities made him [ineligible] for another clinical trial and left him with no options,” Dr. Lagana said.
Using next-generation whole-exome sequencing, Dr. Lagana and colleagues had observed that a previously identified BRAF V600E–dominant subclone had persisted, despite the CAR T-cell treatment, in the patient’s bone marrow and cutaneous plasmacytoma.
The finding was not uncommon. More than half of RRMM patients (about 53%) show emerging clones with mutations within the MAPK signaling pathway, and in about 7% of patients, those include BRAF V600E, which can be targeted, the authors noted.
Further assessment of the patient’s CD138-positive MM cells using western blot signaling pathway analysis looking at DNA and RNA markers did indeed show an increase in MAPK signaling as a consequence of the mutation. This suggested a potential benefit of triple MAPK inhibition, compared with standard strategies.
Based on that information and on insights the researchers had gained from previous research, they implemented the novel, orally administered triple-combination treatment strategy, consisting of monomeric inhibition of BRAF dabrafenib (100 mg, twice daily), as well as dimeric inhibition with the multi–kinase inhibitor regorafenib (40 mg, once daily) and a MEK inhibitor (trametinib, 1.5 mg, for 21/28 days daily).
Of note, previous efforts using only monomeric inhibition of BRAF have not shown much success, but early data has shown some potential, with the inclusion of dimeric inhibition.
“Monomeric inhibition of BRAF has been attempted in patients with V600E, but the efficacy has been limited, likely due to feedback activation of the MAPK pathway via induction of BRAF dimer formation,” Dr. Lagana explained.
Meanwhile, “previous in vitro data from our colleagues at Mount Sinai has shown that inhibition of both monomeric and dimeric forms of BRAF in combination with MEK inhibition can overcome the negative feedback and lead to more efficacious and tolerable treatment,” he said.
With the treatment, the patient achieved a very good partial response for 110 days, with prompt reduction of the subcutaneous skin lesions and an 80% reduction in lambda free light chain (27.5 mg/L).
The triple-drug combination was well tolerated with minimal side effects, primarily involving grade 1 fatigue, and the patient was able to carry out activities of daily living and return to work.
“The triple inhibition allowed us to use less of each drug, which resulted in a well-tolerated regimen without any significant side effects,” Dr. Lagana said.
While the patient relapsed about 3 months later, there was, importantly, no recurrence of the subcutaneous nodules.
“We believe that the triple MAPK inhibition completely eradicated the disease clones driving the extramedullary disease,” Dr. Lagana said.
The therapy meanwhile enabled the patient to bridge to a new clinical trial, where he went into complete remission, and still was as of Sept. 29.
“To our knowledge, this was the first reported successful case of this treatment in an RRMM patient,” Dr. Lagana explained.
Case suggests ‘hope’ for relapsing patients
Importantly, currently many patients in the same position may wind up going to hospice, until such targeted medicine gains momentum, coauthor Samir Parekh, MD, a professor of hematology-oncology at the Hess Center for Science and Medicine, Icahn School of Medicine at Mount Sinai, said in an interview.
“As precision medicine is in its infancy in myeloma, these patients are not routinely sequenced for drug options that may be identified by next-generation sequencing,” said Dr. Parekh.
But for clinicians, the message of this case should be that “there is hope for patients relapsing after CAR T,” he added.
“Precision medicine approaches may be applicable even for this relapsed patient population,” he added. “MAP kinase mutations are common and drugs targeting them may be useful in myeloma.”
Noting that “the infrastructure to test and guide application of these therapies needs to be developed for myeloma, Dr. Parekh predicted that, “in the future, more effective MAPK inhibitors and other mutation or RNA-seq guided therapies will be applicable and hopefully provide more durable remissions.”
Approach may help address unmet need
Until then, however, treatment for patients who relapse after CAR-T and BCMA-targeted therapies has emerged as a significant unmet need. Therefore, this case highlights an important potential strategy, said Hans Lee, MD, an associate professor in the department of lymphoma/myeloma, division of cancer medicine, University of Texas MD Anderson Cancer Center, Houston, commenting on the study.
“This case report provides impetus for oncologists to strongly consider performing next-generation sequencing on myeloma tumor samples to look for potential actionable mutations, such as those in the MAPK pathway – which are common in myeloma,” he said. “With limited treatment options in the post–CAR T and post-BCMA setting, identifying such actional mutations may at least provide a bridge to other effective therapies available through clinical trials such as this patient’s case.”
Dr. Lee noted that key caveats include the fact that most physicians currently don’t have access to the type of next-generation sequencing and drug sensitivity testing used in the study.
Nevertheless, considering the limited options in the post–CAR T and post-BCMA setting, “the successful use of triple MAPK pathway inhibition through monomeric and dimeric inhibition of BRAF and MEK inhibition warrants further study in multiple myeloma in a clinical trial,” he said.
Dr. Lagana and associates are doing just that.
“We are about to launch the clinical trial, where we will match advanced RRMM patients with potential targeted treatments using different DNA and RNA markers,” Dr. Lagana said.
Dr. Lagana and Dr. Parekh had no disclosures to report. Three study coauthors reported receiving research grants or consulting fees from numerous pharmaceutical companies.
In a case that researchers hope might pave the way for similar responses, a hospice-bound relapsed/refractory multiple myeloma (RRMM) patient who relapsed after chimeric antigen receptor (CAR) T-cell therapy was brought back into remission with the help of next-generation genomic sequencing, targeted molecular analysis and a novel combination of MAP kinase (MAPK)–inhibiting drugs.
“We have shown that comprehensive molecular profiling of advanced myeloma patients may provide critical information to guide treatment beyond standard of care,” senior author Alessandro Lagana, PhD, of the Tisch Cancer Institute, Icahn School of Medicine at Mount Sinai, New York, said in an interview.
“This represents proof of concept that, while not curative, targeted molecules may serve as potential bridging therapies to clinical trial enrollment,” the authors further report in the case study, published recently in the Journal of Hematology & Oncology.
The use of B-cell maturation antigen (BCMA) CAR T-cell therapy approaches has transformed the treatment of multiple myeloma and leukemias, resulting in high response rates. However, most patients ultimately relapse, and no clear treatment options beyond CAR T therapy are established.
Such was the case for a 61-year old patient described in the study, who had relapsed 6 months after undergoing anti-BCMA CAR T-cell therapy and progressed after being salvaged for a short period with autologous stem cell transplantation. The patient had developed skin extramedullary disease, manifested as subcutaneous nodules.
“The subcutaneous skin lesions in lower extremities made him [ineligible] for another clinical trial and left him with no options,” Dr. Lagana said.
Using next-generation whole-exome sequencing, Dr. Lagana and colleagues had observed that a previously identified BRAF V600E–dominant subclone had persisted, despite the CAR T-cell treatment, in the patient’s bone marrow and cutaneous plasmacytoma.
The finding was not uncommon. More than half of RRMM patients (about 53%) show emerging clones with mutations within the MAPK signaling pathway, and in about 7% of patients, those include BRAF V600E, which can be targeted, the authors noted.
Further assessment of the patient’s CD138-positive MM cells using western blot signaling pathway analysis looking at DNA and RNA markers did indeed show an increase in MAPK signaling as a consequence of the mutation. This suggested a potential benefit of triple MAPK inhibition, compared with standard strategies.
Based on that information and on insights the researchers had gained from previous research, they implemented the novel, orally administered triple-combination treatment strategy, consisting of monomeric inhibition of BRAF dabrafenib (100 mg, twice daily), as well as dimeric inhibition with the multi–kinase inhibitor regorafenib (40 mg, once daily) and a MEK inhibitor (trametinib, 1.5 mg, for 21/28 days daily).
Of note, previous efforts using only monomeric inhibition of BRAF have not shown much success, but early data has shown some potential, with the inclusion of dimeric inhibition.
“Monomeric inhibition of BRAF has been attempted in patients with V600E, but the efficacy has been limited, likely due to feedback activation of the MAPK pathway via induction of BRAF dimer formation,” Dr. Lagana explained.
Meanwhile, “previous in vitro data from our colleagues at Mount Sinai has shown that inhibition of both monomeric and dimeric forms of BRAF in combination with MEK inhibition can overcome the negative feedback and lead to more efficacious and tolerable treatment,” he said.
With the treatment, the patient achieved a very good partial response for 110 days, with prompt reduction of the subcutaneous skin lesions and an 80% reduction in lambda free light chain (27.5 mg/L).
The triple-drug combination was well tolerated with minimal side effects, primarily involving grade 1 fatigue, and the patient was able to carry out activities of daily living and return to work.
“The triple inhibition allowed us to use less of each drug, which resulted in a well-tolerated regimen without any significant side effects,” Dr. Lagana said.
While the patient relapsed about 3 months later, there was, importantly, no recurrence of the subcutaneous nodules.
“We believe that the triple MAPK inhibition completely eradicated the disease clones driving the extramedullary disease,” Dr. Lagana said.
The therapy meanwhile enabled the patient to bridge to a new clinical trial, where he went into complete remission, and still was as of Sept. 29.
“To our knowledge, this was the first reported successful case of this treatment in an RRMM patient,” Dr. Lagana explained.
Case suggests ‘hope’ for relapsing patients
Importantly, currently many patients in the same position may wind up going to hospice, until such targeted medicine gains momentum, coauthor Samir Parekh, MD, a professor of hematology-oncology at the Hess Center for Science and Medicine, Icahn School of Medicine at Mount Sinai, said in an interview.
“As precision medicine is in its infancy in myeloma, these patients are not routinely sequenced for drug options that may be identified by next-generation sequencing,” said Dr. Parekh.
But for clinicians, the message of this case should be that “there is hope for patients relapsing after CAR T,” he added.
“Precision medicine approaches may be applicable even for this relapsed patient population,” he added. “MAP kinase mutations are common and drugs targeting them may be useful in myeloma.”
Noting that “the infrastructure to test and guide application of these therapies needs to be developed for myeloma, Dr. Parekh predicted that, “in the future, more effective MAPK inhibitors and other mutation or RNA-seq guided therapies will be applicable and hopefully provide more durable remissions.”
Approach may help address unmet need
Until then, however, treatment for patients who relapse after CAR-T and BCMA-targeted therapies has emerged as a significant unmet need. Therefore, this case highlights an important potential strategy, said Hans Lee, MD, an associate professor in the department of lymphoma/myeloma, division of cancer medicine, University of Texas MD Anderson Cancer Center, Houston, commenting on the study.
“This case report provides impetus for oncologists to strongly consider performing next-generation sequencing on myeloma tumor samples to look for potential actionable mutations, such as those in the MAPK pathway – which are common in myeloma,” he said. “With limited treatment options in the post–CAR T and post-BCMA setting, identifying such actional mutations may at least provide a bridge to other effective therapies available through clinical trials such as this patient’s case.”
Dr. Lee noted that key caveats include the fact that most physicians currently don’t have access to the type of next-generation sequencing and drug sensitivity testing used in the study.
Nevertheless, considering the limited options in the post–CAR T and post-BCMA setting, “the successful use of triple MAPK pathway inhibition through monomeric and dimeric inhibition of BRAF and MEK inhibition warrants further study in multiple myeloma in a clinical trial,” he said.
Dr. Lagana and associates are doing just that.
“We are about to launch the clinical trial, where we will match advanced RRMM patients with potential targeted treatments using different DNA and RNA markers,” Dr. Lagana said.
Dr. Lagana and Dr. Parekh had no disclosures to report. Three study coauthors reported receiving research grants or consulting fees from numerous pharmaceutical companies.
FROM THE JOURNAL OF HEMATOLOGY & ONCOLOGY
Meet our newest genetically engineered frenemy, herpes
Herpes to the rescue
Let’s face it: When people hear the word “herpes,” their first thoughts are not positive. But what if herpes could be a hero?
Scientists have found a way to make a strain of herpes that kills cancer because, hey, it’s 2022, and anything is possible. Trials have been going well and this seems like a safe and effective way to fight cancer.
Viruses may be one of our oldest enemies, but it’s also been said that the enemy of my enemy is my friend. So why not make herpes the enemy of cancer, thereby turning it into our friend? The genetically modified herpes virus is injected directly into tumors, where it destroys cancer cells from within. But wait, there’s more! The patient’s immune system also senses the virus and springs into action against it and the cancer in which it is residing.
During the phase 1 trial, three of the nine patients saw tumor reduction and the therapy proved safe as well. Future trials will be able to more specifically target various cancer types and make the treatment better. For once, we are rooting for you, herpes.
A breath of not-so-fresh air
There’s nothing quite like that first real warm day of spring. You can finally open the windows and clear out the old stuffy air that’s been hanging around all winter long. It’s a ritual that’s now backed up with some science in the form of a new study. Turns out that there’s actually a fair amount of smog in the average home. That’s right, smog’s not just for the big city anymore.
As part of the HOMEChem project, a whole host of scientists gathered together under one roof in a typical suburban house and immediately started doing chores. Cooking, cleaning, the works. No, it wasn’t because they had trashed the place the night before. They had set up instrumentation all around the house to measure the chemical makeup of the air inside. A scientist’s idea of a wild party.
The results are perhaps not all that surprising, but interesting nonetheless. Your homemade smog certainly won’t kill you, but there’s both an increased amount and higher concentration of airborne toxins in indoor air, compared with outdoors. Benzene and formaldehyde were common, as were acrolein (a pulmonary toxicant emitted by lumber and burning fats) and isocyanic acid (which can react with proteins in the human body). The researchers noted that most of these chemicals can be removed with proper ventilation.
Although cleaning is certainly responsible for a fair share of the chemicals, cooking generally produced more toxic compounds, similar to what’s found in wildfire smoke. One of the researchers said this makes sense, since a wildfire can be considered an “extreme form of cooking.” Scientists may not know how to party, but their idea of a barbecue sounds … interesting. We’re looking forward to an upcoming study out of California: Can a 1-million acre wildfire adequately cook a ribeye steak?
We’re dying to try composting ... with humans, that is
We here at LOTME are not really fans of politicians, except as objects of ridicule. That is kind of fun. Whether we’re watching Fox News, listening to NPR, or reading Vladimir Putin’s fashion blog, one thing remains clear: If you want actual information, don’t ask a politician.
There are, of course, always exceptions, and we just found one: California state representative Cristina Garcia. Rep. Garcia sponsored a bill just signed into law by Gov. Gavin Newsom that legalizes the practice of human composting, the reduction of remains by “placing bodies in individual vessels and fostering gentle transformation into a nutrient-dense soil.”
Since we’ve written about this sort of thing before – Washington was the first state to legalize the process back in 2019 – we’re more interested now in what Rep. Garcia told NBC News while describing her motivation: “I’ve always wanted to be a tree. The idea of having my family sitting under my shade one day – that brings a lot of joy.” How great is that? Tree-hugging is just not enough. Be the tree.
California is the fifth state to provide its residents with the human composting option, the other three being Colorado, Oregon, and Vermont. The process “typically involves putting a body into a steel vessel, then covering it with organic materials like straw, wood chips and alfalfa. Microbes break down the corpse and the plant matter, transforming the various components into nutrient-rich soil in roughly 30 days,” Smithsonian Magazine explained.
We just happen to have some good news for Rep. Garcia about that wanting-to-be-a-tree business. She’s already pretty close. For more on that, we go to our correspondent from beyond the grave, Carl Sagan, who shares a thought about trees. And no, we couldn’t just write out his quote here. You have to hear it in Dr. Sagan’s own voice.
That’ll be one pandemic with extra distress. Hold the goals
When the COVID-19 pandemic first hit it put a lot of stuff on hold for everyone. Couldn’t eat inside at your favorite restaurant, attend that long-awaited concert, or travel out of the country. Those were all pretty bad, but it was the disruption of pursuing long-term goals that seemed to have the most effect on people’s mental health.
Investigators from the University of Waterloo (Ont.) looked at how putting such goals on hold affected people’s mental well-being. The study’s 226 participants were asked about their “COVID-frozen” goals and the degree to which they were able to actively pursue each goal and how committed they were to achieving it.
What they found was that the participants’ COVID-frozen goals were associated with feelings of psychological distress, such as anxiety, depressive symptoms, stress, and lowered life satisfaction. It was only when participants were able to disengage from goal rumination that well-being was impacted positively.
“Goal rumination is compulsive and can aggravate worries and frustrations while also taking away mental resources from other goals,” Candice Hubley, lead author and a PhD candidate in psychology, said in a written statement. So in short, you’re only stressing yourself out more about something that is far off in the distance when you could be focusing more on short-term, tangible goals instead.
Now, no one is saying to give up on your goals. Just take them one at a time. You’ll have better life satisfaction and your COVID-frozen goals will thaw out before you know it.
Herpes to the rescue
Let’s face it: When people hear the word “herpes,” their first thoughts are not positive. But what if herpes could be a hero?
Scientists have found a way to make a strain of herpes that kills cancer because, hey, it’s 2022, and anything is possible. Trials have been going well and this seems like a safe and effective way to fight cancer.
Viruses may be one of our oldest enemies, but it’s also been said that the enemy of my enemy is my friend. So why not make herpes the enemy of cancer, thereby turning it into our friend? The genetically modified herpes virus is injected directly into tumors, where it destroys cancer cells from within. But wait, there’s more! The patient’s immune system also senses the virus and springs into action against it and the cancer in which it is residing.
During the phase 1 trial, three of the nine patients saw tumor reduction and the therapy proved safe as well. Future trials will be able to more specifically target various cancer types and make the treatment better. For once, we are rooting for you, herpes.
A breath of not-so-fresh air
There’s nothing quite like that first real warm day of spring. You can finally open the windows and clear out the old stuffy air that’s been hanging around all winter long. It’s a ritual that’s now backed up with some science in the form of a new study. Turns out that there’s actually a fair amount of smog in the average home. That’s right, smog’s not just for the big city anymore.
As part of the HOMEChem project, a whole host of scientists gathered together under one roof in a typical suburban house and immediately started doing chores. Cooking, cleaning, the works. No, it wasn’t because they had trashed the place the night before. They had set up instrumentation all around the house to measure the chemical makeup of the air inside. A scientist’s idea of a wild party.
The results are perhaps not all that surprising, but interesting nonetheless. Your homemade smog certainly won’t kill you, but there’s both an increased amount and higher concentration of airborne toxins in indoor air, compared with outdoors. Benzene and formaldehyde were common, as were acrolein (a pulmonary toxicant emitted by lumber and burning fats) and isocyanic acid (which can react with proteins in the human body). The researchers noted that most of these chemicals can be removed with proper ventilation.
Although cleaning is certainly responsible for a fair share of the chemicals, cooking generally produced more toxic compounds, similar to what’s found in wildfire smoke. One of the researchers said this makes sense, since a wildfire can be considered an “extreme form of cooking.” Scientists may not know how to party, but their idea of a barbecue sounds … interesting. We’re looking forward to an upcoming study out of California: Can a 1-million acre wildfire adequately cook a ribeye steak?
We’re dying to try composting ... with humans, that is
We here at LOTME are not really fans of politicians, except as objects of ridicule. That is kind of fun. Whether we’re watching Fox News, listening to NPR, or reading Vladimir Putin’s fashion blog, one thing remains clear: If you want actual information, don’t ask a politician.
There are, of course, always exceptions, and we just found one: California state representative Cristina Garcia. Rep. Garcia sponsored a bill just signed into law by Gov. Gavin Newsom that legalizes the practice of human composting, the reduction of remains by “placing bodies in individual vessels and fostering gentle transformation into a nutrient-dense soil.”
Since we’ve written about this sort of thing before – Washington was the first state to legalize the process back in 2019 – we’re more interested now in what Rep. Garcia told NBC News while describing her motivation: “I’ve always wanted to be a tree. The idea of having my family sitting under my shade one day – that brings a lot of joy.” How great is that? Tree-hugging is just not enough. Be the tree.
California is the fifth state to provide its residents with the human composting option, the other three being Colorado, Oregon, and Vermont. The process “typically involves putting a body into a steel vessel, then covering it with organic materials like straw, wood chips and alfalfa. Microbes break down the corpse and the plant matter, transforming the various components into nutrient-rich soil in roughly 30 days,” Smithsonian Magazine explained.
We just happen to have some good news for Rep. Garcia about that wanting-to-be-a-tree business. She’s already pretty close. For more on that, we go to our correspondent from beyond the grave, Carl Sagan, who shares a thought about trees. And no, we couldn’t just write out his quote here. You have to hear it in Dr. Sagan’s own voice.
That’ll be one pandemic with extra distress. Hold the goals
When the COVID-19 pandemic first hit it put a lot of stuff on hold for everyone. Couldn’t eat inside at your favorite restaurant, attend that long-awaited concert, or travel out of the country. Those were all pretty bad, but it was the disruption of pursuing long-term goals that seemed to have the most effect on people’s mental health.
Investigators from the University of Waterloo (Ont.) looked at how putting such goals on hold affected people’s mental well-being. The study’s 226 participants were asked about their “COVID-frozen” goals and the degree to which they were able to actively pursue each goal and how committed they were to achieving it.
What they found was that the participants’ COVID-frozen goals were associated with feelings of psychological distress, such as anxiety, depressive symptoms, stress, and lowered life satisfaction. It was only when participants were able to disengage from goal rumination that well-being was impacted positively.
“Goal rumination is compulsive and can aggravate worries and frustrations while also taking away mental resources from other goals,” Candice Hubley, lead author and a PhD candidate in psychology, said in a written statement. So in short, you’re only stressing yourself out more about something that is far off in the distance when you could be focusing more on short-term, tangible goals instead.
Now, no one is saying to give up on your goals. Just take them one at a time. You’ll have better life satisfaction and your COVID-frozen goals will thaw out before you know it.
Herpes to the rescue
Let’s face it: When people hear the word “herpes,” their first thoughts are not positive. But what if herpes could be a hero?
Scientists have found a way to make a strain of herpes that kills cancer because, hey, it’s 2022, and anything is possible. Trials have been going well and this seems like a safe and effective way to fight cancer.
Viruses may be one of our oldest enemies, but it’s also been said that the enemy of my enemy is my friend. So why not make herpes the enemy of cancer, thereby turning it into our friend? The genetically modified herpes virus is injected directly into tumors, where it destroys cancer cells from within. But wait, there’s more! The patient’s immune system also senses the virus and springs into action against it and the cancer in which it is residing.
During the phase 1 trial, three of the nine patients saw tumor reduction and the therapy proved safe as well. Future trials will be able to more specifically target various cancer types and make the treatment better. For once, we are rooting for you, herpes.
A breath of not-so-fresh air
There’s nothing quite like that first real warm day of spring. You can finally open the windows and clear out the old stuffy air that’s been hanging around all winter long. It’s a ritual that’s now backed up with some science in the form of a new study. Turns out that there’s actually a fair amount of smog in the average home. That’s right, smog’s not just for the big city anymore.
As part of the HOMEChem project, a whole host of scientists gathered together under one roof in a typical suburban house and immediately started doing chores. Cooking, cleaning, the works. No, it wasn’t because they had trashed the place the night before. They had set up instrumentation all around the house to measure the chemical makeup of the air inside. A scientist’s idea of a wild party.
The results are perhaps not all that surprising, but interesting nonetheless. Your homemade smog certainly won’t kill you, but there’s both an increased amount and higher concentration of airborne toxins in indoor air, compared with outdoors. Benzene and formaldehyde were common, as were acrolein (a pulmonary toxicant emitted by lumber and burning fats) and isocyanic acid (which can react with proteins in the human body). The researchers noted that most of these chemicals can be removed with proper ventilation.
Although cleaning is certainly responsible for a fair share of the chemicals, cooking generally produced more toxic compounds, similar to what’s found in wildfire smoke. One of the researchers said this makes sense, since a wildfire can be considered an “extreme form of cooking.” Scientists may not know how to party, but their idea of a barbecue sounds … interesting. We’re looking forward to an upcoming study out of California: Can a 1-million acre wildfire adequately cook a ribeye steak?
We’re dying to try composting ... with humans, that is
We here at LOTME are not really fans of politicians, except as objects of ridicule. That is kind of fun. Whether we’re watching Fox News, listening to NPR, or reading Vladimir Putin’s fashion blog, one thing remains clear: If you want actual information, don’t ask a politician.
There are, of course, always exceptions, and we just found one: California state representative Cristina Garcia. Rep. Garcia sponsored a bill just signed into law by Gov. Gavin Newsom that legalizes the practice of human composting, the reduction of remains by “placing bodies in individual vessels and fostering gentle transformation into a nutrient-dense soil.”
Since we’ve written about this sort of thing before – Washington was the first state to legalize the process back in 2019 – we’re more interested now in what Rep. Garcia told NBC News while describing her motivation: “I’ve always wanted to be a tree. The idea of having my family sitting under my shade one day – that brings a lot of joy.” How great is that? Tree-hugging is just not enough. Be the tree.
California is the fifth state to provide its residents with the human composting option, the other three being Colorado, Oregon, and Vermont. The process “typically involves putting a body into a steel vessel, then covering it with organic materials like straw, wood chips and alfalfa. Microbes break down the corpse and the plant matter, transforming the various components into nutrient-rich soil in roughly 30 days,” Smithsonian Magazine explained.
We just happen to have some good news for Rep. Garcia about that wanting-to-be-a-tree business. She’s already pretty close. For more on that, we go to our correspondent from beyond the grave, Carl Sagan, who shares a thought about trees. And no, we couldn’t just write out his quote here. You have to hear it in Dr. Sagan’s own voice.
That’ll be one pandemic with extra distress. Hold the goals
When the COVID-19 pandemic first hit it put a lot of stuff on hold for everyone. Couldn’t eat inside at your favorite restaurant, attend that long-awaited concert, or travel out of the country. Those were all pretty bad, but it was the disruption of pursuing long-term goals that seemed to have the most effect on people’s mental health.
Investigators from the University of Waterloo (Ont.) looked at how putting such goals on hold affected people’s mental well-being. The study’s 226 participants were asked about their “COVID-frozen” goals and the degree to which they were able to actively pursue each goal and how committed they were to achieving it.
What they found was that the participants’ COVID-frozen goals were associated with feelings of psychological distress, such as anxiety, depressive symptoms, stress, and lowered life satisfaction. It was only when participants were able to disengage from goal rumination that well-being was impacted positively.
“Goal rumination is compulsive and can aggravate worries and frustrations while also taking away mental resources from other goals,” Candice Hubley, lead author and a PhD candidate in psychology, said in a written statement. So in short, you’re only stressing yourself out more about something that is far off in the distance when you could be focusing more on short-term, tangible goals instead.
Now, no one is saying to give up on your goals. Just take them one at a time. You’ll have better life satisfaction and your COVID-frozen goals will thaw out before you know it.
Apremilast may have some cardiometabolic benefits in patients with psoriasis
The trial, led by Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania, Philadelphia, found that apremilast (Otezla) has a neutral effect on aortic vascular inflammation in patients with moderate to severe psoriasis.
It also had variable, but generally favorable, associations with 68 cardiometabolic biomarkers tested and associations with reductions in both visceral and subcutaneous fat. Findings of the study were published online in JAMA Dermatology.
Fat reductions maintained at 1-year mark
The researchers found a 5%-6% reduction in subcutaneous and visceral fat at week 16 of the study that was maintained at the 1-year mark. “The fact that it was rock stable a year later is pretty encouraging,” Dr. Gelfand told this news organization.
As for effects on vascular inflammation, Dr. Gelfand said, “The good news is we didn’t find any adverse effects on aortic vascular inflammation, but we didn’t find any beneficial effects either. That was a little disappointing.
“The most surprising thing was really the effects on visceral adiposity,” he added. “I’m not aware of any other drug having demonstrated that effect.”
Michael S. Garshick, MD, a cardiologist with NYU Langone Health in New York, who was not involved with the trial, told this news organization that despite seemingly good epidemiologic evidence in observational studies that by treating psoriasis surrogates of cardiovascular risk can be reduced, this trial, like others before it, failed to reduce aortic vascular inflammation.
The trial does help answer the question of whether apremilast can induce weight loss, he said, something that earlier trials suggested. “This trial confirms that, which is exciting,” he said. The reduction in both visceral and subcutaneous fat “deserves a lot further study.”
Several questions remain, Dr. Garshick said. Both he and Dr. Gelfand pointed to the need for large, placebo-controlled trials. “We still don’t know which medications may be preferrable in psoriasis to reduce [cardiovascular] risk if any at all,” Dr. Garshick said.
Seventy patients enrolled
In total, 70 patients with moderate to severe psoriasis were enrolled, 60 completed week 16, and 39 completed week 52 of the single-arm, open-label trial conducted between April 2017 and August 2021 at seven dermatology sites in the United States.
Participants took 30 mg of apremilast, an oral phosphodiesterase-4 (PDE-4) inhibitor approved for treating psoriasis and psoriatic arthritis, twice daily. Participants’ average age was 47.5 years; most were male (77.1%) and White (82.9%); almost 6% were Black. Average body mass index was 30 kg/m2. Patients could not have received biologics within 90 days of study baseline (or 180 days for ustekinumab [Stelara]).
There was no change in aortic vascular inflammation at week 16 (target to background ratio, −0.02; 95% confidence interval [CI], −0.08 to 0.05; P = .61) or week 52 (target to background ratio, −0.07; 95% CI, −0.15 to 0.01; P = .09) compared with baseline.
“At week 16, there were reductions in levels of interleukin-1b, fetuin A, valine, leucine, and isoleucine,” the authors wrote, adding that at week 52, compared with baseline, “there were reductions in levels of ferritin, cholesterol efflux capacity, beta-hydroxybutyrate, acetone, and ketone bodies, and an increase in levels of apolipoprotein A-1.”
This study highlights the importance of screening, Dr. Garshick said.
He and Dr. Gelfand said people with psoriatic disease tend to be vastly underscreened for cardiovascular risk factors.
Dr. Gelfand said, “If we did what we knew worked – meaning we screened them for diabetes, we screen their cholesterol, we check their blood pressure, and we adequately treated those traditional cardiovascular risk factors, we probably could narrow the gap quite a bit” in terms of the lower life expectancy people face when they have more significant psoriasis.
Celgene was the initial funding sponsor; sponsorship was then transferred to Amgen. The authors designed, executed, analyzed, and reported the study. Celgene provided nonbinding input into study design, and Amgen provided nonbinding input into the reporting of results. Dr. Gelfand reported numerous disclosures with various pharmaceutical companies and organizations. Dr. Garshick reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The trial, led by Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania, Philadelphia, found that apremilast (Otezla) has a neutral effect on aortic vascular inflammation in patients with moderate to severe psoriasis.
It also had variable, but generally favorable, associations with 68 cardiometabolic biomarkers tested and associations with reductions in both visceral and subcutaneous fat. Findings of the study were published online in JAMA Dermatology.
Fat reductions maintained at 1-year mark
The researchers found a 5%-6% reduction in subcutaneous and visceral fat at week 16 of the study that was maintained at the 1-year mark. “The fact that it was rock stable a year later is pretty encouraging,” Dr. Gelfand told this news organization.
As for effects on vascular inflammation, Dr. Gelfand said, “The good news is we didn’t find any adverse effects on aortic vascular inflammation, but we didn’t find any beneficial effects either. That was a little disappointing.
“The most surprising thing was really the effects on visceral adiposity,” he added. “I’m not aware of any other drug having demonstrated that effect.”
Michael S. Garshick, MD, a cardiologist with NYU Langone Health in New York, who was not involved with the trial, told this news organization that despite seemingly good epidemiologic evidence in observational studies that by treating psoriasis surrogates of cardiovascular risk can be reduced, this trial, like others before it, failed to reduce aortic vascular inflammation.
The trial does help answer the question of whether apremilast can induce weight loss, he said, something that earlier trials suggested. “This trial confirms that, which is exciting,” he said. The reduction in both visceral and subcutaneous fat “deserves a lot further study.”
Several questions remain, Dr. Garshick said. Both he and Dr. Gelfand pointed to the need for large, placebo-controlled trials. “We still don’t know which medications may be preferrable in psoriasis to reduce [cardiovascular] risk if any at all,” Dr. Garshick said.
Seventy patients enrolled
In total, 70 patients with moderate to severe psoriasis were enrolled, 60 completed week 16, and 39 completed week 52 of the single-arm, open-label trial conducted between April 2017 and August 2021 at seven dermatology sites in the United States.
Participants took 30 mg of apremilast, an oral phosphodiesterase-4 (PDE-4) inhibitor approved for treating psoriasis and psoriatic arthritis, twice daily. Participants’ average age was 47.5 years; most were male (77.1%) and White (82.9%); almost 6% were Black. Average body mass index was 30 kg/m2. Patients could not have received biologics within 90 days of study baseline (or 180 days for ustekinumab [Stelara]).
There was no change in aortic vascular inflammation at week 16 (target to background ratio, −0.02; 95% confidence interval [CI], −0.08 to 0.05; P = .61) or week 52 (target to background ratio, −0.07; 95% CI, −0.15 to 0.01; P = .09) compared with baseline.
“At week 16, there were reductions in levels of interleukin-1b, fetuin A, valine, leucine, and isoleucine,” the authors wrote, adding that at week 52, compared with baseline, “there were reductions in levels of ferritin, cholesterol efflux capacity, beta-hydroxybutyrate, acetone, and ketone bodies, and an increase in levels of apolipoprotein A-1.”
This study highlights the importance of screening, Dr. Garshick said.
He and Dr. Gelfand said people with psoriatic disease tend to be vastly underscreened for cardiovascular risk factors.
Dr. Gelfand said, “If we did what we knew worked – meaning we screened them for diabetes, we screen their cholesterol, we check their blood pressure, and we adequately treated those traditional cardiovascular risk factors, we probably could narrow the gap quite a bit” in terms of the lower life expectancy people face when they have more significant psoriasis.
Celgene was the initial funding sponsor; sponsorship was then transferred to Amgen. The authors designed, executed, analyzed, and reported the study. Celgene provided nonbinding input into study design, and Amgen provided nonbinding input into the reporting of results. Dr. Gelfand reported numerous disclosures with various pharmaceutical companies and organizations. Dr. Garshick reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The trial, led by Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania, Philadelphia, found that apremilast (Otezla) has a neutral effect on aortic vascular inflammation in patients with moderate to severe psoriasis.
It also had variable, but generally favorable, associations with 68 cardiometabolic biomarkers tested and associations with reductions in both visceral and subcutaneous fat. Findings of the study were published online in JAMA Dermatology.
Fat reductions maintained at 1-year mark
The researchers found a 5%-6% reduction in subcutaneous and visceral fat at week 16 of the study that was maintained at the 1-year mark. “The fact that it was rock stable a year later is pretty encouraging,” Dr. Gelfand told this news organization.
As for effects on vascular inflammation, Dr. Gelfand said, “The good news is we didn’t find any adverse effects on aortic vascular inflammation, but we didn’t find any beneficial effects either. That was a little disappointing.
“The most surprising thing was really the effects on visceral adiposity,” he added. “I’m not aware of any other drug having demonstrated that effect.”
Michael S. Garshick, MD, a cardiologist with NYU Langone Health in New York, who was not involved with the trial, told this news organization that despite seemingly good epidemiologic evidence in observational studies that by treating psoriasis surrogates of cardiovascular risk can be reduced, this trial, like others before it, failed to reduce aortic vascular inflammation.
The trial does help answer the question of whether apremilast can induce weight loss, he said, something that earlier trials suggested. “This trial confirms that, which is exciting,” he said. The reduction in both visceral and subcutaneous fat “deserves a lot further study.”
Several questions remain, Dr. Garshick said. Both he and Dr. Gelfand pointed to the need for large, placebo-controlled trials. “We still don’t know which medications may be preferrable in psoriasis to reduce [cardiovascular] risk if any at all,” Dr. Garshick said.
Seventy patients enrolled
In total, 70 patients with moderate to severe psoriasis were enrolled, 60 completed week 16, and 39 completed week 52 of the single-arm, open-label trial conducted between April 2017 and August 2021 at seven dermatology sites in the United States.
Participants took 30 mg of apremilast, an oral phosphodiesterase-4 (PDE-4) inhibitor approved for treating psoriasis and psoriatic arthritis, twice daily. Participants’ average age was 47.5 years; most were male (77.1%) and White (82.9%); almost 6% were Black. Average body mass index was 30 kg/m2. Patients could not have received biologics within 90 days of study baseline (or 180 days for ustekinumab [Stelara]).
There was no change in aortic vascular inflammation at week 16 (target to background ratio, −0.02; 95% confidence interval [CI], −0.08 to 0.05; P = .61) or week 52 (target to background ratio, −0.07; 95% CI, −0.15 to 0.01; P = .09) compared with baseline.
“At week 16, there were reductions in levels of interleukin-1b, fetuin A, valine, leucine, and isoleucine,” the authors wrote, adding that at week 52, compared with baseline, “there were reductions in levels of ferritin, cholesterol efflux capacity, beta-hydroxybutyrate, acetone, and ketone bodies, and an increase in levels of apolipoprotein A-1.”
This study highlights the importance of screening, Dr. Garshick said.
He and Dr. Gelfand said people with psoriatic disease tend to be vastly underscreened for cardiovascular risk factors.
Dr. Gelfand said, “If we did what we knew worked – meaning we screened them for diabetes, we screen their cholesterol, we check their blood pressure, and we adequately treated those traditional cardiovascular risk factors, we probably could narrow the gap quite a bit” in terms of the lower life expectancy people face when they have more significant psoriasis.
Celgene was the initial funding sponsor; sponsorship was then transferred to Amgen. The authors designed, executed, analyzed, and reported the study. Celgene provided nonbinding input into study design, and Amgen provided nonbinding input into the reporting of results. Dr. Gelfand reported numerous disclosures with various pharmaceutical companies and organizations. Dr. Garshick reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Yes, we should talk politics and religion with patients
From our first days as medical students, we are told that politics and religion are topics to be avoided with patients, but I disagree. Knowing more about our patients allows us to deliver better care.
Politics and religion: New risk factors
The importance of politics and religion in the health of patients was clearly demonstrated during the COVID-19 pandemic. Lives were needlessly lost because of stands taken based on religious beliefs or a political ideology. Families, friends, and the community at large were impacted.
Over my years of practice, I have found that while these are difficult topics to address, they should not be avoided. Studies have shown that open acknowledgement of religious beliefs can affect both clinical outcomes and well-being. Religion and spirituality are as much a part of our patient’s lives as the physical parameters that we measure. To neglect these significant aspects is to miss the very essence of the individual.
I made it a practice to ask patients about their religious beliefs, the extent to which religion shaped their life, and whether they were part of a church community. Knowing this allowed me to separate deep personal belief from stances based on personal freedom, misinformation, conspiracies, and politics.
I found that information about political leanings flowed naturally in discussions with patients as we trusted and respected each other over time. If I approached politics objectively and nonjudgmentally, it generally led to meaningful conversation. This helped me to understand the patient as an individual and informed my diagnosis and treatment plan.
Politics as stress
For example, on more than one occasion, a patient with atrial fibrillation presented with persistent elevated blood pressure and pulse rate despite adherence to the medical regimen that I had prescribed. After a few minutes of discussion, it was clear that excessive attention to political commentary on TV and social media raised their anxiety and anger level, putting them at greater risk for adverse outcomes. I advised that they refocus their leisure activities rather than change or increase medication.
It is disappointing to see how one of the great scientific advances of our lifetime, vaccination science, has been tarnished because of political or religious ideology and to see the extent to which these beliefs influenced COVID-19 vaccination compliance. As health care providers, we must promote information based on the scientific method. If patients challenge us and point out that recommendations based on science seem to change over time, we must explain that science evolves on the basis of new information objectively gathered. We need to find out what information the patient has gotten from the Internet, TV, or conspiracy theories and counter this with scientific facts. If we do not discuss religion and politics with our patients along with other risk factors, we may compromise our ability to give them the best advice and treatment.
Our patients have a right to their own spiritual and political ideology. If it differs dramatically from our own, this should not influence our commitment to care for them. But we have an obligation to challenge unfounded beliefs about medicine and counter with scientific facts. There are times when individual freedoms must be secondary to public health. Ultimately, it is up to the patient to choose, but they should not be given a “free pass” on the basis of religion or politics. If I know something is true and I would do it myself or recommend it for my family, I have an obligation to provide this recommendation to my patients.
Religious preference is included in medical records. It is not appropriate to add political preference, but the patient benefits if a long-term caregiver knows this information.
During the pandemic, for the first time in my 40+ years of practice, some patients questioned my recommendations and placed equal or greater weight on religion, politics, or conspiracy theories. This continues to be a very real struggle.
Knowing and understanding our patients as individuals is critical to providing optimum care and that means tackling these formally taboo topics. If having a potentially uncomfortable conversation with patients allows us to save one life, it is worth it.
Dr. Francis is a cardiologist at Inova Heart and Vascular Institute, McLean, Va. He disclosed no relevant conflict of interest. A version of this article first appeared on Medscape.com.
From our first days as medical students, we are told that politics and religion are topics to be avoided with patients, but I disagree. Knowing more about our patients allows us to deliver better care.
Politics and religion: New risk factors
The importance of politics and religion in the health of patients was clearly demonstrated during the COVID-19 pandemic. Lives were needlessly lost because of stands taken based on religious beliefs or a political ideology. Families, friends, and the community at large were impacted.
Over my years of practice, I have found that while these are difficult topics to address, they should not be avoided. Studies have shown that open acknowledgement of religious beliefs can affect both clinical outcomes and well-being. Religion and spirituality are as much a part of our patient’s lives as the physical parameters that we measure. To neglect these significant aspects is to miss the very essence of the individual.
I made it a practice to ask patients about their religious beliefs, the extent to which religion shaped their life, and whether they were part of a church community. Knowing this allowed me to separate deep personal belief from stances based on personal freedom, misinformation, conspiracies, and politics.
I found that information about political leanings flowed naturally in discussions with patients as we trusted and respected each other over time. If I approached politics objectively and nonjudgmentally, it generally led to meaningful conversation. This helped me to understand the patient as an individual and informed my diagnosis and treatment plan.
Politics as stress
For example, on more than one occasion, a patient with atrial fibrillation presented with persistent elevated blood pressure and pulse rate despite adherence to the medical regimen that I had prescribed. After a few minutes of discussion, it was clear that excessive attention to political commentary on TV and social media raised their anxiety and anger level, putting them at greater risk for adverse outcomes. I advised that they refocus their leisure activities rather than change or increase medication.
It is disappointing to see how one of the great scientific advances of our lifetime, vaccination science, has been tarnished because of political or religious ideology and to see the extent to which these beliefs influenced COVID-19 vaccination compliance. As health care providers, we must promote information based on the scientific method. If patients challenge us and point out that recommendations based on science seem to change over time, we must explain that science evolves on the basis of new information objectively gathered. We need to find out what information the patient has gotten from the Internet, TV, or conspiracy theories and counter this with scientific facts. If we do not discuss religion and politics with our patients along with other risk factors, we may compromise our ability to give them the best advice and treatment.
Our patients have a right to their own spiritual and political ideology. If it differs dramatically from our own, this should not influence our commitment to care for them. But we have an obligation to challenge unfounded beliefs about medicine and counter with scientific facts. There are times when individual freedoms must be secondary to public health. Ultimately, it is up to the patient to choose, but they should not be given a “free pass” on the basis of religion or politics. If I know something is true and I would do it myself or recommend it for my family, I have an obligation to provide this recommendation to my patients.
Religious preference is included in medical records. It is not appropriate to add political preference, but the patient benefits if a long-term caregiver knows this information.
During the pandemic, for the first time in my 40+ years of practice, some patients questioned my recommendations and placed equal or greater weight on religion, politics, or conspiracy theories. This continues to be a very real struggle.
Knowing and understanding our patients as individuals is critical to providing optimum care and that means tackling these formally taboo topics. If having a potentially uncomfortable conversation with patients allows us to save one life, it is worth it.
Dr. Francis is a cardiologist at Inova Heart and Vascular Institute, McLean, Va. He disclosed no relevant conflict of interest. A version of this article first appeared on Medscape.com.
From our first days as medical students, we are told that politics and religion are topics to be avoided with patients, but I disagree. Knowing more about our patients allows us to deliver better care.
Politics and religion: New risk factors
The importance of politics and religion in the health of patients was clearly demonstrated during the COVID-19 pandemic. Lives were needlessly lost because of stands taken based on religious beliefs or a political ideology. Families, friends, and the community at large were impacted.
Over my years of practice, I have found that while these are difficult topics to address, they should not be avoided. Studies have shown that open acknowledgement of religious beliefs can affect both clinical outcomes and well-being. Religion and spirituality are as much a part of our patient’s lives as the physical parameters that we measure. To neglect these significant aspects is to miss the very essence of the individual.
I made it a practice to ask patients about their religious beliefs, the extent to which religion shaped their life, and whether they were part of a church community. Knowing this allowed me to separate deep personal belief from stances based on personal freedom, misinformation, conspiracies, and politics.
I found that information about political leanings flowed naturally in discussions with patients as we trusted and respected each other over time. If I approached politics objectively and nonjudgmentally, it generally led to meaningful conversation. This helped me to understand the patient as an individual and informed my diagnosis and treatment plan.
Politics as stress
For example, on more than one occasion, a patient with atrial fibrillation presented with persistent elevated blood pressure and pulse rate despite adherence to the medical regimen that I had prescribed. After a few minutes of discussion, it was clear that excessive attention to political commentary on TV and social media raised their anxiety and anger level, putting them at greater risk for adverse outcomes. I advised that they refocus their leisure activities rather than change or increase medication.
It is disappointing to see how one of the great scientific advances of our lifetime, vaccination science, has been tarnished because of political or religious ideology and to see the extent to which these beliefs influenced COVID-19 vaccination compliance. As health care providers, we must promote information based on the scientific method. If patients challenge us and point out that recommendations based on science seem to change over time, we must explain that science evolves on the basis of new information objectively gathered. We need to find out what information the patient has gotten from the Internet, TV, or conspiracy theories and counter this with scientific facts. If we do not discuss religion and politics with our patients along with other risk factors, we may compromise our ability to give them the best advice and treatment.
Our patients have a right to their own spiritual and political ideology. If it differs dramatically from our own, this should not influence our commitment to care for them. But we have an obligation to challenge unfounded beliefs about medicine and counter with scientific facts. There are times when individual freedoms must be secondary to public health. Ultimately, it is up to the patient to choose, but they should not be given a “free pass” on the basis of religion or politics. If I know something is true and I would do it myself or recommend it for my family, I have an obligation to provide this recommendation to my patients.
Religious preference is included in medical records. It is not appropriate to add political preference, but the patient benefits if a long-term caregiver knows this information.
During the pandemic, for the first time in my 40+ years of practice, some patients questioned my recommendations and placed equal or greater weight on religion, politics, or conspiracy theories. This continues to be a very real struggle.
Knowing and understanding our patients as individuals is critical to providing optimum care and that means tackling these formally taboo topics. If having a potentially uncomfortable conversation with patients allows us to save one life, it is worth it.
Dr. Francis is a cardiologist at Inova Heart and Vascular Institute, McLean, Va. He disclosed no relevant conflict of interest. A version of this article first appeared on Medscape.com.
Visual impairment more common in minority youth
Race, ethnicity, and socioeconomic status are closely associated with visual impairment in adolescents in the United States, researchers have found.
The study showed that adolescents who identified as Black, Mexican-American, of low income, or as non–U.S. citizens were two to three times more likely than White adolescents to report vision problems and to perform worse on objective tests of visual acuity.
Although disparities in visual impairment with respect to ethnic, racial, and socioeconomic status have been described among adults, little research has explored the adolescent population, according to the researchers, whose findings appear in JAMA Ophthalmology.
“The primary motivation behind trying to figure out exactly when these disparities emerge is that it gives us an opportunity to come up with ideas for interventions that can potentially address them before they manifest in a way that they are no longer treatable,” said Isdin Oke, MD, an instructor in ophthalmology at Harvard Medical School, Boston, who led the latest study.
Dr. Oke and his colleagues analyzed the records of 2,833 children and adolescents aged 12 through 18 years (mean, 15.5 years; 49% female) in the National Health and Nutrition Examination Survey. All the participants had completed a visual function questionnaire and had undergone an eye examination. The primary outcomes of the study were subjective (self-reported) poor vision and objective measures of visual function (visual acuity worse than 20/40 in the better-seeing eye).
Of the study participants, 14% were non-Hispanic Black, 11% were Mexican-American, 63% were non-Hispanic White, and 11% were of other race and ethnicity. Five percent of participants were not U.S. citizens, and 19% had a family income below the poverty threshold.
After accounting for potential confounders, self-reported poor vision was more common among Black (odds ratio, 2.85; 95% CI, 2.00-4.05; P < .001), Mexican-American (OR, 2.83; 95% CI, 1.70-4.73; P < .001), and low-income youth (OR, 2.44; 95% CI, 1.63-3.65; P < .001), the researchers report.
The study also found increased odds of visual acuity below 20/40 in the better-seeing eye among Black (OR, 2.13; 95% CI, 1.41-3.24; P = .001) and Mexican-American adolescents (OR, 2.13; 95% CI, 1.39-3.26; P = .001) and non–U.S. citizens (OR, 1.96; 95% CI, 1.10-3.49; P = .02).
Black and Mexican-American adolescents were almost three times more likely to suffer poor subjective visual function and twice as likely to have low objective visual acuity than non-Hispanic White youth, according to the researchers.
“I think it’s something that health care providers, and even the population as a whole, should be more aware of,” Dr. Oke said.
Opportunities for intervention
Dr. Oke said the findings likely reflect the underlying inequities in access to vision care experienced by these persons.
“There are a lot of opportunities for early intervention, whether it’s through vision screening or improving access to vision services for children and adolescents that can really make a big difference over the long term,” he told this news organization.
Michael F. Chiang, MD, director of the National Eye Institute at the National Institutes of Health, Bethesda, Md., agreed that more steps need to be taken to improve access to vision care for all Americans.
“There are good data showing that we don’t have a sufficient number of eye care providers and health-services clinical researchers who come from backgrounds that are currently underrepresented in medicine and science,” Dr. Chiang said. “Therefore, we need to find ways to inspire, recruit, and train a larger number of people to strengthen our vision workforce.”
Other ways to address these gaps in care, he added, include improving understanding of the social determinants of vision health as well as developing ways to improve access to eye care.
“That may include new models of eye care, like telehealth to improve outreach, and will likely need to include what we call implementation science, so clinicians can better adopt these measures,” he said.
Dr. Oke and Dr. Chiang reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Race, ethnicity, and socioeconomic status are closely associated with visual impairment in adolescents in the United States, researchers have found.
The study showed that adolescents who identified as Black, Mexican-American, of low income, or as non–U.S. citizens were two to three times more likely than White adolescents to report vision problems and to perform worse on objective tests of visual acuity.
Although disparities in visual impairment with respect to ethnic, racial, and socioeconomic status have been described among adults, little research has explored the adolescent population, according to the researchers, whose findings appear in JAMA Ophthalmology.
“The primary motivation behind trying to figure out exactly when these disparities emerge is that it gives us an opportunity to come up with ideas for interventions that can potentially address them before they manifest in a way that they are no longer treatable,” said Isdin Oke, MD, an instructor in ophthalmology at Harvard Medical School, Boston, who led the latest study.
Dr. Oke and his colleagues analyzed the records of 2,833 children and adolescents aged 12 through 18 years (mean, 15.5 years; 49% female) in the National Health and Nutrition Examination Survey. All the participants had completed a visual function questionnaire and had undergone an eye examination. The primary outcomes of the study were subjective (self-reported) poor vision and objective measures of visual function (visual acuity worse than 20/40 in the better-seeing eye).
Of the study participants, 14% were non-Hispanic Black, 11% were Mexican-American, 63% were non-Hispanic White, and 11% were of other race and ethnicity. Five percent of participants were not U.S. citizens, and 19% had a family income below the poverty threshold.
After accounting for potential confounders, self-reported poor vision was more common among Black (odds ratio, 2.85; 95% CI, 2.00-4.05; P < .001), Mexican-American (OR, 2.83; 95% CI, 1.70-4.73; P < .001), and low-income youth (OR, 2.44; 95% CI, 1.63-3.65; P < .001), the researchers report.
The study also found increased odds of visual acuity below 20/40 in the better-seeing eye among Black (OR, 2.13; 95% CI, 1.41-3.24; P = .001) and Mexican-American adolescents (OR, 2.13; 95% CI, 1.39-3.26; P = .001) and non–U.S. citizens (OR, 1.96; 95% CI, 1.10-3.49; P = .02).
Black and Mexican-American adolescents were almost three times more likely to suffer poor subjective visual function and twice as likely to have low objective visual acuity than non-Hispanic White youth, according to the researchers.
“I think it’s something that health care providers, and even the population as a whole, should be more aware of,” Dr. Oke said.
Opportunities for intervention
Dr. Oke said the findings likely reflect the underlying inequities in access to vision care experienced by these persons.
“There are a lot of opportunities for early intervention, whether it’s through vision screening or improving access to vision services for children and adolescents that can really make a big difference over the long term,” he told this news organization.
Michael F. Chiang, MD, director of the National Eye Institute at the National Institutes of Health, Bethesda, Md., agreed that more steps need to be taken to improve access to vision care for all Americans.
“There are good data showing that we don’t have a sufficient number of eye care providers and health-services clinical researchers who come from backgrounds that are currently underrepresented in medicine and science,” Dr. Chiang said. “Therefore, we need to find ways to inspire, recruit, and train a larger number of people to strengthen our vision workforce.”
Other ways to address these gaps in care, he added, include improving understanding of the social determinants of vision health as well as developing ways to improve access to eye care.
“That may include new models of eye care, like telehealth to improve outreach, and will likely need to include what we call implementation science, so clinicians can better adopt these measures,” he said.
Dr. Oke and Dr. Chiang reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Race, ethnicity, and socioeconomic status are closely associated with visual impairment in adolescents in the United States, researchers have found.
The study showed that adolescents who identified as Black, Mexican-American, of low income, or as non–U.S. citizens were two to three times more likely than White adolescents to report vision problems and to perform worse on objective tests of visual acuity.
Although disparities in visual impairment with respect to ethnic, racial, and socioeconomic status have been described among adults, little research has explored the adolescent population, according to the researchers, whose findings appear in JAMA Ophthalmology.
“The primary motivation behind trying to figure out exactly when these disparities emerge is that it gives us an opportunity to come up with ideas for interventions that can potentially address them before they manifest in a way that they are no longer treatable,” said Isdin Oke, MD, an instructor in ophthalmology at Harvard Medical School, Boston, who led the latest study.
Dr. Oke and his colleagues analyzed the records of 2,833 children and adolescents aged 12 through 18 years (mean, 15.5 years; 49% female) in the National Health and Nutrition Examination Survey. All the participants had completed a visual function questionnaire and had undergone an eye examination. The primary outcomes of the study were subjective (self-reported) poor vision and objective measures of visual function (visual acuity worse than 20/40 in the better-seeing eye).
Of the study participants, 14% were non-Hispanic Black, 11% were Mexican-American, 63% were non-Hispanic White, and 11% were of other race and ethnicity. Five percent of participants were not U.S. citizens, and 19% had a family income below the poverty threshold.
After accounting for potential confounders, self-reported poor vision was more common among Black (odds ratio, 2.85; 95% CI, 2.00-4.05; P < .001), Mexican-American (OR, 2.83; 95% CI, 1.70-4.73; P < .001), and low-income youth (OR, 2.44; 95% CI, 1.63-3.65; P < .001), the researchers report.
The study also found increased odds of visual acuity below 20/40 in the better-seeing eye among Black (OR, 2.13; 95% CI, 1.41-3.24; P = .001) and Mexican-American adolescents (OR, 2.13; 95% CI, 1.39-3.26; P = .001) and non–U.S. citizens (OR, 1.96; 95% CI, 1.10-3.49; P = .02).
Black and Mexican-American adolescents were almost three times more likely to suffer poor subjective visual function and twice as likely to have low objective visual acuity than non-Hispanic White youth, according to the researchers.
“I think it’s something that health care providers, and even the population as a whole, should be more aware of,” Dr. Oke said.
Opportunities for intervention
Dr. Oke said the findings likely reflect the underlying inequities in access to vision care experienced by these persons.
“There are a lot of opportunities for early intervention, whether it’s through vision screening or improving access to vision services for children and adolescents that can really make a big difference over the long term,” he told this news organization.
Michael F. Chiang, MD, director of the National Eye Institute at the National Institutes of Health, Bethesda, Md., agreed that more steps need to be taken to improve access to vision care for all Americans.
“There are good data showing that we don’t have a sufficient number of eye care providers and health-services clinical researchers who come from backgrounds that are currently underrepresented in medicine and science,” Dr. Chiang said. “Therefore, we need to find ways to inspire, recruit, and train a larger number of people to strengthen our vision workforce.”
Other ways to address these gaps in care, he added, include improving understanding of the social determinants of vision health as well as developing ways to improve access to eye care.
“That may include new models of eye care, like telehealth to improve outreach, and will likely need to include what we call implementation science, so clinicians can better adopt these measures,” he said.
Dr. Oke and Dr. Chiang reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Can we eliminate measles and rubella worldwide?
A study in The Lancet Global Health takes a pessimistic view of our ability to eradicate measles by 2100, although rubella forecasts look a bit more promising.
So far, measles has been eliminated in 81 countries and rubella in 93. But factors such as antivaccination sentiment and misinformation linking vaccination to autism have led to occasional outbreaks. In addition, because the COVID-19 pandemic fueled lower routine vaccination coverage and postponed public health campaigns, some countries have also lost previously gained ground.
The study, which is slated for publication in the Oct. 1 issue of the Lancet Global Health, explored the likelihood of eliminating measles and rubella, based on vaccination strategies in 93 countries with the highest measles and rubella burden, under two vaccination scenarios: 1) a “business as usual” approach, that is, continuing current vaccination coverage via routine childhood immunization schedules and intermittent vaccination campaigns that target age groups to vaccinate quickly (known as SIAs); and 2) an “intensified investment approach” that scales up SIA vaccination coverage into the future.
Both vaccination scenarios were evaluated within the context of two national models (Johns Hopkins University and Public Health England), and one subnational model (Nigeria) for rubella transmission.
Lead author Amy Winter, PhD, assistant professor of epidemiology and biostatistics, University of Georgia College of Public Health, Athens, told this news organization that “under the intensified investment scenario, rubella elimination is likely to be achieved in all 93 countries that were modeled [but] measles elimination is likely in some but not all countries.”
This is especially the case if the goal is cessation of vaccination campaigns, study authors noted when placing the research in context.
But Dr. Winter also emphasized that Nigeria offered specific lessons not seen in the national models.
For one,
In addition, she stressed a need to improve vaccine equity by focusing on areas with really low coverage and then moving into areas with higher coverage.
“The Nigerian subnational analysis definitely illustrates the importance of achieving equitable vaccination and the need for potentially targeted strategies to improve vaccination,” she said. “The initial focus should be on getting areas with low coverage up to par.”
Still, “even with the intensified investment approach, we won’t be able to eradicate measles,” William Moss, MD, professor of epidemiology and executive director, International Vaccine Access Center, Johns Hopkins University, Baltimore, who was not directly involved in the study, told this news organization.
Pandemic interruptions, future strategies
In a related editorial (The Lancet Global Health. 2022 Oct 1. doi: 10.1016/S2214-109X[22]00388-6), the authors noted that COVID-19 has markedly disrupted vaccination campaigns globally.
In 2017, 118 (61%) countries achieved the Global Vaccine Action Plan 2020 target of 90% or more national MCV1 (first dose of measles vaccine) coverage. Since that time, measles coverage has declined from 84%-85% in 2017 to 81% in 2021, leaving 24.7 million completely unprotected (also known as zero-dose children) and 14.7 million children underimmunized (that is, recipients of only 1 dose).
Notably, this is the lowest immunization level since 2008, with more than 5 million more children missing their first measles dose.
Dr. Moss has previously written on the biological feasibility of measles eradication and said that it’s not tenable to rely on increased vaccination coverage alone.
We need “new tools and the new strategies. One of the ones that we’re most excited about [is] microarray patches,” he said, noting that they are thermostable and can be administered by anyone.
Dr. Moss also said that, while he is hoping for point-of-care rapid diagnostics, the focus of the efforts needs to change.
“Where’s [the] measles virus coming from? Where’s it being exported from and where is it being imported to?” he posited, adding that the focus should be on these areas “to try to shut down transmission … a radical kind of second phase of a measles eradication puts aside equity and focuses on sources and sinks.”
In the interim, rubella elimination looks promising.
“It’s not as contagious [as measles] and has a lower sort of herd immunity threshold because of it,” Dr. Winter said.
Dr. Winter and Dr. Moss report no relevant financial relationships. The study was funded by the World Health Organization, Gavi, the Vaccine Alliance, the Centers for Disease Control and Prevention, and the Bill & Melinda Gates Foundation.
A version of this article first appeared on Medscape.com.
A study in The Lancet Global Health takes a pessimistic view of our ability to eradicate measles by 2100, although rubella forecasts look a bit more promising.
So far, measles has been eliminated in 81 countries and rubella in 93. But factors such as antivaccination sentiment and misinformation linking vaccination to autism have led to occasional outbreaks. In addition, because the COVID-19 pandemic fueled lower routine vaccination coverage and postponed public health campaigns, some countries have also lost previously gained ground.
The study, which is slated for publication in the Oct. 1 issue of the Lancet Global Health, explored the likelihood of eliminating measles and rubella, based on vaccination strategies in 93 countries with the highest measles and rubella burden, under two vaccination scenarios: 1) a “business as usual” approach, that is, continuing current vaccination coverage via routine childhood immunization schedules and intermittent vaccination campaigns that target age groups to vaccinate quickly (known as SIAs); and 2) an “intensified investment approach” that scales up SIA vaccination coverage into the future.
Both vaccination scenarios were evaluated within the context of two national models (Johns Hopkins University and Public Health England), and one subnational model (Nigeria) for rubella transmission.
Lead author Amy Winter, PhD, assistant professor of epidemiology and biostatistics, University of Georgia College of Public Health, Athens, told this news organization that “under the intensified investment scenario, rubella elimination is likely to be achieved in all 93 countries that were modeled [but] measles elimination is likely in some but not all countries.”
This is especially the case if the goal is cessation of vaccination campaigns, study authors noted when placing the research in context.
But Dr. Winter also emphasized that Nigeria offered specific lessons not seen in the national models.
For one,
In addition, she stressed a need to improve vaccine equity by focusing on areas with really low coverage and then moving into areas with higher coverage.
“The Nigerian subnational analysis definitely illustrates the importance of achieving equitable vaccination and the need for potentially targeted strategies to improve vaccination,” she said. “The initial focus should be on getting areas with low coverage up to par.”
Still, “even with the intensified investment approach, we won’t be able to eradicate measles,” William Moss, MD, professor of epidemiology and executive director, International Vaccine Access Center, Johns Hopkins University, Baltimore, who was not directly involved in the study, told this news organization.
Pandemic interruptions, future strategies
In a related editorial (The Lancet Global Health. 2022 Oct 1. doi: 10.1016/S2214-109X[22]00388-6), the authors noted that COVID-19 has markedly disrupted vaccination campaigns globally.
In 2017, 118 (61%) countries achieved the Global Vaccine Action Plan 2020 target of 90% or more national MCV1 (first dose of measles vaccine) coverage. Since that time, measles coverage has declined from 84%-85% in 2017 to 81% in 2021, leaving 24.7 million completely unprotected (also known as zero-dose children) and 14.7 million children underimmunized (that is, recipients of only 1 dose).
Notably, this is the lowest immunization level since 2008, with more than 5 million more children missing their first measles dose.
Dr. Moss has previously written on the biological feasibility of measles eradication and said that it’s not tenable to rely on increased vaccination coverage alone.
We need “new tools and the new strategies. One of the ones that we’re most excited about [is] microarray patches,” he said, noting that they are thermostable and can be administered by anyone.
Dr. Moss also said that, while he is hoping for point-of-care rapid diagnostics, the focus of the efforts needs to change.
“Where’s [the] measles virus coming from? Where’s it being exported from and where is it being imported to?” he posited, adding that the focus should be on these areas “to try to shut down transmission … a radical kind of second phase of a measles eradication puts aside equity and focuses on sources and sinks.”
In the interim, rubella elimination looks promising.
“It’s not as contagious [as measles] and has a lower sort of herd immunity threshold because of it,” Dr. Winter said.
Dr. Winter and Dr. Moss report no relevant financial relationships. The study was funded by the World Health Organization, Gavi, the Vaccine Alliance, the Centers for Disease Control and Prevention, and the Bill & Melinda Gates Foundation.
A version of this article first appeared on Medscape.com.
A study in The Lancet Global Health takes a pessimistic view of our ability to eradicate measles by 2100, although rubella forecasts look a bit more promising.
So far, measles has been eliminated in 81 countries and rubella in 93. But factors such as antivaccination sentiment and misinformation linking vaccination to autism have led to occasional outbreaks. In addition, because the COVID-19 pandemic fueled lower routine vaccination coverage and postponed public health campaigns, some countries have also lost previously gained ground.
The study, which is slated for publication in the Oct. 1 issue of the Lancet Global Health, explored the likelihood of eliminating measles and rubella, based on vaccination strategies in 93 countries with the highest measles and rubella burden, under two vaccination scenarios: 1) a “business as usual” approach, that is, continuing current vaccination coverage via routine childhood immunization schedules and intermittent vaccination campaigns that target age groups to vaccinate quickly (known as SIAs); and 2) an “intensified investment approach” that scales up SIA vaccination coverage into the future.
Both vaccination scenarios were evaluated within the context of two national models (Johns Hopkins University and Public Health England), and one subnational model (Nigeria) for rubella transmission.
Lead author Amy Winter, PhD, assistant professor of epidemiology and biostatistics, University of Georgia College of Public Health, Athens, told this news organization that “under the intensified investment scenario, rubella elimination is likely to be achieved in all 93 countries that were modeled [but] measles elimination is likely in some but not all countries.”
This is especially the case if the goal is cessation of vaccination campaigns, study authors noted when placing the research in context.
But Dr. Winter also emphasized that Nigeria offered specific lessons not seen in the national models.
For one,
In addition, she stressed a need to improve vaccine equity by focusing on areas with really low coverage and then moving into areas with higher coverage.
“The Nigerian subnational analysis definitely illustrates the importance of achieving equitable vaccination and the need for potentially targeted strategies to improve vaccination,” she said. “The initial focus should be on getting areas with low coverage up to par.”
Still, “even with the intensified investment approach, we won’t be able to eradicate measles,” William Moss, MD, professor of epidemiology and executive director, International Vaccine Access Center, Johns Hopkins University, Baltimore, who was not directly involved in the study, told this news organization.
Pandemic interruptions, future strategies
In a related editorial (The Lancet Global Health. 2022 Oct 1. doi: 10.1016/S2214-109X[22]00388-6), the authors noted that COVID-19 has markedly disrupted vaccination campaigns globally.
In 2017, 118 (61%) countries achieved the Global Vaccine Action Plan 2020 target of 90% or more national MCV1 (first dose of measles vaccine) coverage. Since that time, measles coverage has declined from 84%-85% in 2017 to 81% in 2021, leaving 24.7 million completely unprotected (also known as zero-dose children) and 14.7 million children underimmunized (that is, recipients of only 1 dose).
Notably, this is the lowest immunization level since 2008, with more than 5 million more children missing their first measles dose.
Dr. Moss has previously written on the biological feasibility of measles eradication and said that it’s not tenable to rely on increased vaccination coverage alone.
We need “new tools and the new strategies. One of the ones that we’re most excited about [is] microarray patches,” he said, noting that they are thermostable and can be administered by anyone.
Dr. Moss also said that, while he is hoping for point-of-care rapid diagnostics, the focus of the efforts needs to change.
“Where’s [the] measles virus coming from? Where’s it being exported from and where is it being imported to?” he posited, adding that the focus should be on these areas “to try to shut down transmission … a radical kind of second phase of a measles eradication puts aside equity and focuses on sources and sinks.”
In the interim, rubella elimination looks promising.
“It’s not as contagious [as measles] and has a lower sort of herd immunity threshold because of it,” Dr. Winter said.
Dr. Winter and Dr. Moss report no relevant financial relationships. The study was funded by the World Health Organization, Gavi, the Vaccine Alliance, the Centers for Disease Control and Prevention, and the Bill & Melinda Gates Foundation.
A version of this article first appeared on Medscape.com.
FROM THE LANCET GLOBAL HEALTH
Noted oncologist ponders death, life, care inequities
In 2020, he published a book aimed at cancer specialists and their patients on how to die “with hope and dignity,” titled “Between Life and Death” (Penguin Random House India).
When Dr. Patel, the CEO of Carolina Blood and Cancer Care Associates in Rock Hill, S.C., became president of the Washington-based Community Oncology Alliance 2 years ago, he stepped into a leadership role in community oncology. As an advocate for health care payment reform on Capitol Hill, the South Carolina legislature, and within his own practice, Dr. Patel has long worked to eliminate disparities in U.S. cancer care.
This news organization spoke with Dr. Patel about his unusual career path.
Question: Your father had a great influence on you. Can you tell us more about him?
Answer: My dad was a hermit and a saint. He lost his dad when he was 4 years old and moved to the big city with his cousins. When he was 9 or so, he got a message saying that his mum was very ill. So, he and his cousin raised some money, got a doctor and one of those old, rugged jeeps, and they started driving to the village, but rains had destroyed the road. So, without penicillin, his mum died of pneumonia.
He felt that roads and doctor access were the two big factors that could have saved her life. He eventually became the Superintending Engineer for four districts in Gujarat State, building roads connecting every village, but he never gave up his simplistic, minimalist life.
When I was in elementary school, every other weekend my dad would literally dump me at the Mahatma Gandhi Ashram and come back in 2 hours. So, I’m looking at Gandhi’s cabinets, his pictures, reading about his life. So, my formative years were born in that.
Q: I read that you were intending to become an engineer and join the space race. How did your father nudge you toward medicine?
A: When I was 9 years old, my favorite movie hero died of cancer. To comfort me, my father inserted the idea into my brain: When you grow up, you can become a doctor to cure cancer. So, when I finished high school, I was 24th in the state and had an option to go to the space school in India. On the day when I was going for the interview, I could see tears in my father’s eyes, and he said, You know what, boy? I thought you’re going to become a doctor and cure cancer. So, to honor him, I went to med school instead.

Q: I understand that your father also triggered your interest in photography?
A: I started photographing Kutchi tribal people in 1977, after I bought a camera from a famous architect [Hasmukh Patel], while traveling with my dad. And then my dad bought me a motorcycle, so I started riding myself. From the time I entered med school in 1978 until I finished my residency in 1987, I made several trips following Kutchi migrant families and livestock. They leave their homeland in Kutch [district] during summer in search of grass and water to keep their livestock alive and walk across the state from the desert of Kutch all the way to central Gujarat until monsoon begins. Then they return, only to resume the journey next year. I would catch them along their journey, would talk to them, drink tea and eat millet crepes with them.

In 1984, between Dr. Patel’s medical school and residency, the Lions Club in his hometown, Ahmedabad, India, sponsored him and three buddies to document people and wildlife in Gujarat state. Traveling by motorcycle, the four friends stayed for free with local families by knocking on doors and explaining that they were medical students. Dr. Patel’s photographs were exhibited by the Lions Club of Ahmedabad and at India’s top art institution, the Lalit Kala gallery.
In the 3rd year of his internal-medicine residency in Bombay (now Mumbai), Dr. Patel approached a national newspaper, The Indian Express, for work. He was immediately sent on assignment to cover a cholera epidemic and filed his story and photographs the following day. He worked as a photojournalist and subeditor for a year.
Q: Among all your thousands of pictures, do you have a favorite?
A: There were two photos of Kutchi people that touched me. There was one photo of a lady. All of her worldly belongings were in the picture and a smile on her face showed that we don’t need so many things to be happy. The second photo is of an elderly lady shifting her water pan on her head to a younger family member. And a little girl looks up with a look of curiosity: Will I be doing this when I grow up? We seek so much materialistic happiness. But when you look at the curiosity, smiles, and happiness [in these photos], you realize we could have a lot of happiness in minimalism, as well.
Q: After you finished your residency in Ahmedabad, how did you get started in oncology?
A: In 1986, Ahmedabad City and Gujarat State did not have structured training programs in oncology, so I went to Bombay [Mumbai], where Dr. B.C. Mehta, a true legend and pioneer in India, had started hematology-oncology training. I was a post-doc research fellow with him for a little over a year but when I started seeing patients, I had to answer to myself, Am I doing everything I can to help these people? I saw that the U.K. had one of the best training programs in hem malignancy, so I started applying. Then something happened that was almost like a miracle.
In April 1992, Dr. Patel was working at the Institute of Kidney Diseases in Ahmedabad. One afternoon, just as the clinic was closing for siesta, a family brought in a young girl. She had drug-induced thrombocytopenia and needed an immediate transfusion. The father offered to sell his wedding ring to pay Dr. Patel if he would supervise the treatment and stay by the girl’s side. Dr. Patel told the man to keep his ring, then he remained in the office with the child. At 4 p.m., the office phone rang. It was Dr. H.K. Parikh, an eminent British physician who was wintering in India and needed to make a medical appointment for his wife. On a normal day, Dr. Patel would have missed the call.
“This is how I got to meet Dr. Parikh, out of the blue,” said Dr. Patel. “His wife came to the office for 6 weeks and after 6 weeks, he said, You’re a smart guy; you should come to England. That was in April. I sent a resume and all the usual paperwork. On July 16, 1992, at 2 in the morning, I got a call from the U.K. saying, Your job is confirmed. I’m going to fax your appointment through the Royal College of Physicians, and you’re coming to Manchester to work with us. I’d been sponsored by the Overseas Doctors Training Program.
“So, it turns out that if I’d declined to see that patient and declined to stay in my clinic that afternoon, if I’d declined to see this doctor’s wife, I would never have been in the U.K. And that opened up the doors for me. I like that story because I’ve found that standing up for people who do not have a voice, who do not have hope, always leads to what is destined for me.”
Q: After working as a registrar in the United Kingdom 4 years, you found yourself in the United States and, once again, had to train as an internist. What was new about U.S. oncology?
A: I took 3 years to get recertified in Jamaica Hospital in Queens, then became a fellow in hematology-oncology at the Thomas Jefferson in Philadelphia. My U.K. training was all based on hematological malignancy. In the United States, I shifted into solid tumors.
Q: You have a long history of advocating for affordable oncology at the community, state, and federal level, and you recently launched a disparities initiative in your center called NOLA (No One Left Alone). What was the trigger for NOLA?
A: In the spring of 2020, when we started seeing the COVID surge and the difference in mortality rate between the multiple races, at the same time I saw the AACR [American Association for Cancer Research] 2020 disparity report showing that 34% of cancer deaths are preventable – one in three – if we took care of disparities. The same year, the Community Oncology Alliance asked me to become the president. So, I felt that there is something herding me, leading me, to this position. Eighty percent of cancer patients are treated in community clinics like ours. It put the onus on me to do something.
I learned from Gandhi that I cannot depend on government, I cannot depend on the policy, I have to act myself.
I said, I would not worry about making money, I would rather lose funding on this. So, we started. I read 400+ papers; I spent over 1,000 hours reading about disparities. And I realized that it’s not complicated. There are five pillars to eliminate disparity: access to care for financial reasons, access to biomarker testing or precision medicine, access to social determinants of health, access to cancer screening, and trials. If we focus on these five, we can at least bring that number from 34% to 20%, if not lower.
So, we put that plan in place. I dedicated three employees whose only role is to ensure that not a single patient has to take financial burden from my practice. And we showed it’s doable.
This has now become my mission for the last quarter of my life.
In 2020, Dr. Patel published a book on dying well titled “Between Life and Death.” It’s framed as a series of his conversations with a former patient, Harry Falls. Harry wanted to understand death better, so Dr. Patel narrated five patient stories, drawing the threads together to help Harry face the inevitable. Dr. Patel now uses a similar approach to train clinicians on having meaningful end-of-life conversations with patients.
Q: Why did you feel the need to write a book about dying?
A: The more I’ve witnessed, the more I’m convinced that there are things that we don’t know about this process, which needs to be explored much more. However, I do feel that there’s a power within all of us to steer the process of leaving this world.
Before I sat down with Harry, I loved to counsel patients, but I didn’t have any structural ideas. It was Harry himself who told me that I now had a simple way to explain dying to a much larger audience.
Q: What is your secret for fitting everything into your life?
A: I’ll tell you, it’s very simple. If I put my soul, heart, mind, actions, and language on the one plane and don’t let my brain and conditioning influence my choices, then I live in the moment. Whenever I let my conditioned mind take all the decisions, those are crooked, because you know, we’re selfish creatures – we can use what we call the convenient lie to hide inconvenient truth. And I try not to do that. I mean, it’s been a journey. It didn’t come overnight. I learned. And I feel that over all these years, the only thing that rewarded me, that opened the door of where I am today, was pure, selfless process, whether it’s the act of talking, speaking, or doing.
In 2020, he published a book aimed at cancer specialists and their patients on how to die “with hope and dignity,” titled “Between Life and Death” (Penguin Random House India).
When Dr. Patel, the CEO of Carolina Blood and Cancer Care Associates in Rock Hill, S.C., became president of the Washington-based Community Oncology Alliance 2 years ago, he stepped into a leadership role in community oncology. As an advocate for health care payment reform on Capitol Hill, the South Carolina legislature, and within his own practice, Dr. Patel has long worked to eliminate disparities in U.S. cancer care.
This news organization spoke with Dr. Patel about his unusual career path.
Question: Your father had a great influence on you. Can you tell us more about him?
Answer: My dad was a hermit and a saint. He lost his dad when he was 4 years old and moved to the big city with his cousins. When he was 9 or so, he got a message saying that his mum was very ill. So, he and his cousin raised some money, got a doctor and one of those old, rugged jeeps, and they started driving to the village, but rains had destroyed the road. So, without penicillin, his mum died of pneumonia.
He felt that roads and doctor access were the two big factors that could have saved her life. He eventually became the Superintending Engineer for four districts in Gujarat State, building roads connecting every village, but he never gave up his simplistic, minimalist life.
When I was in elementary school, every other weekend my dad would literally dump me at the Mahatma Gandhi Ashram and come back in 2 hours. So, I’m looking at Gandhi’s cabinets, his pictures, reading about his life. So, my formative years were born in that.
Q: I read that you were intending to become an engineer and join the space race. How did your father nudge you toward medicine?
A: When I was 9 years old, my favorite movie hero died of cancer. To comfort me, my father inserted the idea into my brain: When you grow up, you can become a doctor to cure cancer. So, when I finished high school, I was 24th in the state and had an option to go to the space school in India. On the day when I was going for the interview, I could see tears in my father’s eyes, and he said, You know what, boy? I thought you’re going to become a doctor and cure cancer. So, to honor him, I went to med school instead.

Q: I understand that your father also triggered your interest in photography?
A: I started photographing Kutchi tribal people in 1977, after I bought a camera from a famous architect [Hasmukh Patel], while traveling with my dad. And then my dad bought me a motorcycle, so I started riding myself. From the time I entered med school in 1978 until I finished my residency in 1987, I made several trips following Kutchi migrant families and livestock. They leave their homeland in Kutch [district] during summer in search of grass and water to keep their livestock alive and walk across the state from the desert of Kutch all the way to central Gujarat until monsoon begins. Then they return, only to resume the journey next year. I would catch them along their journey, would talk to them, drink tea and eat millet crepes with them.

In 1984, between Dr. Patel’s medical school and residency, the Lions Club in his hometown, Ahmedabad, India, sponsored him and three buddies to document people and wildlife in Gujarat state. Traveling by motorcycle, the four friends stayed for free with local families by knocking on doors and explaining that they were medical students. Dr. Patel’s photographs were exhibited by the Lions Club of Ahmedabad and at India’s top art institution, the Lalit Kala gallery.
In the 3rd year of his internal-medicine residency in Bombay (now Mumbai), Dr. Patel approached a national newspaper, The Indian Express, for work. He was immediately sent on assignment to cover a cholera epidemic and filed his story and photographs the following day. He worked as a photojournalist and subeditor for a year.
Q: Among all your thousands of pictures, do you have a favorite?
A: There were two photos of Kutchi people that touched me. There was one photo of a lady. All of her worldly belongings were in the picture and a smile on her face showed that we don’t need so many things to be happy. The second photo is of an elderly lady shifting her water pan on her head to a younger family member. And a little girl looks up with a look of curiosity: Will I be doing this when I grow up? We seek so much materialistic happiness. But when you look at the curiosity, smiles, and happiness [in these photos], you realize we could have a lot of happiness in minimalism, as well.
Q: After you finished your residency in Ahmedabad, how did you get started in oncology?
A: In 1986, Ahmedabad City and Gujarat State did not have structured training programs in oncology, so I went to Bombay [Mumbai], where Dr. B.C. Mehta, a true legend and pioneer in India, had started hematology-oncology training. I was a post-doc research fellow with him for a little over a year but when I started seeing patients, I had to answer to myself, Am I doing everything I can to help these people? I saw that the U.K. had one of the best training programs in hem malignancy, so I started applying. Then something happened that was almost like a miracle.
In April 1992, Dr. Patel was working at the Institute of Kidney Diseases in Ahmedabad. One afternoon, just as the clinic was closing for siesta, a family brought in a young girl. She had drug-induced thrombocytopenia and needed an immediate transfusion. The father offered to sell his wedding ring to pay Dr. Patel if he would supervise the treatment and stay by the girl’s side. Dr. Patel told the man to keep his ring, then he remained in the office with the child. At 4 p.m., the office phone rang. It was Dr. H.K. Parikh, an eminent British physician who was wintering in India and needed to make a medical appointment for his wife. On a normal day, Dr. Patel would have missed the call.
“This is how I got to meet Dr. Parikh, out of the blue,” said Dr. Patel. “His wife came to the office for 6 weeks and after 6 weeks, he said, You’re a smart guy; you should come to England. That was in April. I sent a resume and all the usual paperwork. On July 16, 1992, at 2 in the morning, I got a call from the U.K. saying, Your job is confirmed. I’m going to fax your appointment through the Royal College of Physicians, and you’re coming to Manchester to work with us. I’d been sponsored by the Overseas Doctors Training Program.
“So, it turns out that if I’d declined to see that patient and declined to stay in my clinic that afternoon, if I’d declined to see this doctor’s wife, I would never have been in the U.K. And that opened up the doors for me. I like that story because I’ve found that standing up for people who do not have a voice, who do not have hope, always leads to what is destined for me.”
Q: After working as a registrar in the United Kingdom 4 years, you found yourself in the United States and, once again, had to train as an internist. What was new about U.S. oncology?
A: I took 3 years to get recertified in Jamaica Hospital in Queens, then became a fellow in hematology-oncology at the Thomas Jefferson in Philadelphia. My U.K. training was all based on hematological malignancy. In the United States, I shifted into solid tumors.
Q: You have a long history of advocating for affordable oncology at the community, state, and federal level, and you recently launched a disparities initiative in your center called NOLA (No One Left Alone). What was the trigger for NOLA?
A: In the spring of 2020, when we started seeing the COVID surge and the difference in mortality rate between the multiple races, at the same time I saw the AACR [American Association for Cancer Research] 2020 disparity report showing that 34% of cancer deaths are preventable – one in three – if we took care of disparities. The same year, the Community Oncology Alliance asked me to become the president. So, I felt that there is something herding me, leading me, to this position. Eighty percent of cancer patients are treated in community clinics like ours. It put the onus on me to do something.
I learned from Gandhi that I cannot depend on government, I cannot depend on the policy, I have to act myself.
I said, I would not worry about making money, I would rather lose funding on this. So, we started. I read 400+ papers; I spent over 1,000 hours reading about disparities. And I realized that it’s not complicated. There are five pillars to eliminate disparity: access to care for financial reasons, access to biomarker testing or precision medicine, access to social determinants of health, access to cancer screening, and trials. If we focus on these five, we can at least bring that number from 34% to 20%, if not lower.
So, we put that plan in place. I dedicated three employees whose only role is to ensure that not a single patient has to take financial burden from my practice. And we showed it’s doable.
This has now become my mission for the last quarter of my life.
In 2020, Dr. Patel published a book on dying well titled “Between Life and Death.” It’s framed as a series of his conversations with a former patient, Harry Falls. Harry wanted to understand death better, so Dr. Patel narrated five patient stories, drawing the threads together to help Harry face the inevitable. Dr. Patel now uses a similar approach to train clinicians on having meaningful end-of-life conversations with patients.
Q: Why did you feel the need to write a book about dying?
A: The more I’ve witnessed, the more I’m convinced that there are things that we don’t know about this process, which needs to be explored much more. However, I do feel that there’s a power within all of us to steer the process of leaving this world.
Before I sat down with Harry, I loved to counsel patients, but I didn’t have any structural ideas. It was Harry himself who told me that I now had a simple way to explain dying to a much larger audience.
Q: What is your secret for fitting everything into your life?
A: I’ll tell you, it’s very simple. If I put my soul, heart, mind, actions, and language on the one plane and don’t let my brain and conditioning influence my choices, then I live in the moment. Whenever I let my conditioned mind take all the decisions, those are crooked, because you know, we’re selfish creatures – we can use what we call the convenient lie to hide inconvenient truth. And I try not to do that. I mean, it’s been a journey. It didn’t come overnight. I learned. And I feel that over all these years, the only thing that rewarded me, that opened the door of where I am today, was pure, selfless process, whether it’s the act of talking, speaking, or doing.
In 2020, he published a book aimed at cancer specialists and their patients on how to die “with hope and dignity,” titled “Between Life and Death” (Penguin Random House India).
When Dr. Patel, the CEO of Carolina Blood and Cancer Care Associates in Rock Hill, S.C., became president of the Washington-based Community Oncology Alliance 2 years ago, he stepped into a leadership role in community oncology. As an advocate for health care payment reform on Capitol Hill, the South Carolina legislature, and within his own practice, Dr. Patel has long worked to eliminate disparities in U.S. cancer care.
This news organization spoke with Dr. Patel about his unusual career path.
Question: Your father had a great influence on you. Can you tell us more about him?
Answer: My dad was a hermit and a saint. He lost his dad when he was 4 years old and moved to the big city with his cousins. When he was 9 or so, he got a message saying that his mum was very ill. So, he and his cousin raised some money, got a doctor and one of those old, rugged jeeps, and they started driving to the village, but rains had destroyed the road. So, without penicillin, his mum died of pneumonia.
He felt that roads and doctor access were the two big factors that could have saved her life. He eventually became the Superintending Engineer for four districts in Gujarat State, building roads connecting every village, but he never gave up his simplistic, minimalist life.
When I was in elementary school, every other weekend my dad would literally dump me at the Mahatma Gandhi Ashram and come back in 2 hours. So, I’m looking at Gandhi’s cabinets, his pictures, reading about his life. So, my formative years were born in that.
Q: I read that you were intending to become an engineer and join the space race. How did your father nudge you toward medicine?
A: When I was 9 years old, my favorite movie hero died of cancer. To comfort me, my father inserted the idea into my brain: When you grow up, you can become a doctor to cure cancer. So, when I finished high school, I was 24th in the state and had an option to go to the space school in India. On the day when I was going for the interview, I could see tears in my father’s eyes, and he said, You know what, boy? I thought you’re going to become a doctor and cure cancer. So, to honor him, I went to med school instead.

Q: I understand that your father also triggered your interest in photography?
A: I started photographing Kutchi tribal people in 1977, after I bought a camera from a famous architect [Hasmukh Patel], while traveling with my dad. And then my dad bought me a motorcycle, so I started riding myself. From the time I entered med school in 1978 until I finished my residency in 1987, I made several trips following Kutchi migrant families and livestock. They leave their homeland in Kutch [district] during summer in search of grass and water to keep their livestock alive and walk across the state from the desert of Kutch all the way to central Gujarat until monsoon begins. Then they return, only to resume the journey next year. I would catch them along their journey, would talk to them, drink tea and eat millet crepes with them.

In 1984, between Dr. Patel’s medical school and residency, the Lions Club in his hometown, Ahmedabad, India, sponsored him and three buddies to document people and wildlife in Gujarat state. Traveling by motorcycle, the four friends stayed for free with local families by knocking on doors and explaining that they were medical students. Dr. Patel’s photographs were exhibited by the Lions Club of Ahmedabad and at India’s top art institution, the Lalit Kala gallery.
In the 3rd year of his internal-medicine residency in Bombay (now Mumbai), Dr. Patel approached a national newspaper, The Indian Express, for work. He was immediately sent on assignment to cover a cholera epidemic and filed his story and photographs the following day. He worked as a photojournalist and subeditor for a year.
Q: Among all your thousands of pictures, do you have a favorite?
A: There were two photos of Kutchi people that touched me. There was one photo of a lady. All of her worldly belongings were in the picture and a smile on her face showed that we don’t need so many things to be happy. The second photo is of an elderly lady shifting her water pan on her head to a younger family member. And a little girl looks up with a look of curiosity: Will I be doing this when I grow up? We seek so much materialistic happiness. But when you look at the curiosity, smiles, and happiness [in these photos], you realize we could have a lot of happiness in minimalism, as well.
Q: After you finished your residency in Ahmedabad, how did you get started in oncology?
A: In 1986, Ahmedabad City and Gujarat State did not have structured training programs in oncology, so I went to Bombay [Mumbai], where Dr. B.C. Mehta, a true legend and pioneer in India, had started hematology-oncology training. I was a post-doc research fellow with him for a little over a year but when I started seeing patients, I had to answer to myself, Am I doing everything I can to help these people? I saw that the U.K. had one of the best training programs in hem malignancy, so I started applying. Then something happened that was almost like a miracle.
In April 1992, Dr. Patel was working at the Institute of Kidney Diseases in Ahmedabad. One afternoon, just as the clinic was closing for siesta, a family brought in a young girl. She had drug-induced thrombocytopenia and needed an immediate transfusion. The father offered to sell his wedding ring to pay Dr. Patel if he would supervise the treatment and stay by the girl’s side. Dr. Patel told the man to keep his ring, then he remained in the office with the child. At 4 p.m., the office phone rang. It was Dr. H.K. Parikh, an eminent British physician who was wintering in India and needed to make a medical appointment for his wife. On a normal day, Dr. Patel would have missed the call.
“This is how I got to meet Dr. Parikh, out of the blue,” said Dr. Patel. “His wife came to the office for 6 weeks and after 6 weeks, he said, You’re a smart guy; you should come to England. That was in April. I sent a resume and all the usual paperwork. On July 16, 1992, at 2 in the morning, I got a call from the U.K. saying, Your job is confirmed. I’m going to fax your appointment through the Royal College of Physicians, and you’re coming to Manchester to work with us. I’d been sponsored by the Overseas Doctors Training Program.
“So, it turns out that if I’d declined to see that patient and declined to stay in my clinic that afternoon, if I’d declined to see this doctor’s wife, I would never have been in the U.K. And that opened up the doors for me. I like that story because I’ve found that standing up for people who do not have a voice, who do not have hope, always leads to what is destined for me.”
Q: After working as a registrar in the United Kingdom 4 years, you found yourself in the United States and, once again, had to train as an internist. What was new about U.S. oncology?
A: I took 3 years to get recertified in Jamaica Hospital in Queens, then became a fellow in hematology-oncology at the Thomas Jefferson in Philadelphia. My U.K. training was all based on hematological malignancy. In the United States, I shifted into solid tumors.
Q: You have a long history of advocating for affordable oncology at the community, state, and federal level, and you recently launched a disparities initiative in your center called NOLA (No One Left Alone). What was the trigger for NOLA?
A: In the spring of 2020, when we started seeing the COVID surge and the difference in mortality rate between the multiple races, at the same time I saw the AACR [American Association for Cancer Research] 2020 disparity report showing that 34% of cancer deaths are preventable – one in three – if we took care of disparities. The same year, the Community Oncology Alliance asked me to become the president. So, I felt that there is something herding me, leading me, to this position. Eighty percent of cancer patients are treated in community clinics like ours. It put the onus on me to do something.
I learned from Gandhi that I cannot depend on government, I cannot depend on the policy, I have to act myself.
I said, I would not worry about making money, I would rather lose funding on this. So, we started. I read 400+ papers; I spent over 1,000 hours reading about disparities. And I realized that it’s not complicated. There are five pillars to eliminate disparity: access to care for financial reasons, access to biomarker testing or precision medicine, access to social determinants of health, access to cancer screening, and trials. If we focus on these five, we can at least bring that number from 34% to 20%, if not lower.
So, we put that plan in place. I dedicated three employees whose only role is to ensure that not a single patient has to take financial burden from my practice. And we showed it’s doable.
This has now become my mission for the last quarter of my life.
In 2020, Dr. Patel published a book on dying well titled “Between Life and Death.” It’s framed as a series of his conversations with a former patient, Harry Falls. Harry wanted to understand death better, so Dr. Patel narrated five patient stories, drawing the threads together to help Harry face the inevitable. Dr. Patel now uses a similar approach to train clinicians on having meaningful end-of-life conversations with patients.
Q: Why did you feel the need to write a book about dying?
A: The more I’ve witnessed, the more I’m convinced that there are things that we don’t know about this process, which needs to be explored much more. However, I do feel that there’s a power within all of us to steer the process of leaving this world.
Before I sat down with Harry, I loved to counsel patients, but I didn’t have any structural ideas. It was Harry himself who told me that I now had a simple way to explain dying to a much larger audience.
Q: What is your secret for fitting everything into your life?
A: I’ll tell you, it’s very simple. If I put my soul, heart, mind, actions, and language on the one plane and don’t let my brain and conditioning influence my choices, then I live in the moment. Whenever I let my conditioned mind take all the decisions, those are crooked, because you know, we’re selfish creatures – we can use what we call the convenient lie to hide inconvenient truth. And I try not to do that. I mean, it’s been a journey. It didn’t come overnight. I learned. And I feel that over all these years, the only thing that rewarded me, that opened the door of where I am today, was pure, selfless process, whether it’s the act of talking, speaking, or doing.