User login
For MD-IQ use only
Early FMT shows promise for preventing recurrent C. difficile
Fecal microbiota transplantation (FMT) is safe and highly effective as first-line therapy for patients with first or second Clostridioides difficile infection, according to the first randomized, double-blind, placebo-controlled trial of its kind.
Study enrollment was halted after an interim analysis revealed significantly better outcomes among patients who received vancomycin plus FMT versus vancomycin alone, reported lead author Simon Mark Dahl Baunwall, MD, of Aarhus (Denmark) University Hospital and colleagues in The Lancet Gastroenterology & Hepatology.
The investigators noted that the participants represented a real-world patient population, so the data support FMT “as a necessary, effective first-line option” in routine management of C. difficile infection.
“Previous studies have demonstrated clinical cure rates [with FMT] of up to 92%,” Dr. Baunwall and colleagues wrote. “Early use of FMT for first or second C. difficile infection has therapeutic potential, but no formal randomized trials to support use of the approach as a first-line therapy have been done.”
The present trial, conducted at a university hospital in Denmark, involved 42 adult patients with first or second C. difficile infection. Patients were randomized in a 1:1 ratio to receive either vancomycin alone or vancomycin plus FMT. All patients received 125 mg oral vancomycin four times daily for a minimum of 10 days after diagnosis. On day 1 after completion of vancomycin therapy and again between day 3 and 7, patients received either oral FMT or matching placebo, depending on their group. After completing the protocol, patients were followed for 8 weeks or C. difficile recurrence to evaluate resolution of C. difficile–associated diarrhea.
“In this trial, patients were treated with two sequential FMT procedures on separate days,” the investigators noted. “This practice might have overtreated some patients and differs from previous trials. It remains unknown whether optimal effect is achieved by one or two treatments.”
The trial design called for 84 patients, but enrollment was halted after an interim analysis of the above cohort of 42 patients because of significantly lower rate resolution in the placebo group. At the 2-month mark, 90% (95% confidence interval, 70%-99%) of patients in the FMT group had resolution, compared with only 33% (95% CI, 15%-57%) of patients in the placebo group (P = .0003), constituting a 57% (95% CI, 33%-81%) absolute risk reduction.
Most patients experienced adverse events, including 20 in the FMT group and all 21 in the placebo group, although most were transient and nonserious. The most common adverse events were diarrhea, which occurred more frequently in the FMT group (23 vs. 14 events), followed by abdominal pain(14 vs. 11 events) and nausea (12 vs. 5 events).
One limitation of the study was its single-center design with regional uptake; the authors noted that, despite having high statistical power for the clinical effect, the study’s premature termination and low patient number prevent inferences regarding mortality, time to effect, and cost.
“The results of this trial highlight how the use of fecal microbiota transplantation as a first-line treatment can effectively prevent C. difficile recurrence and suggests that microbiota restoration might be necessary to obtain sustained resolution,” the investigators wrote. “At present, only 10% of patients with multiple, recurrent C. difficile infection and indication for FMT receive it. International initiatives address the unmet need, but logistic and regulatory obstacles remain unsolved.”
Encouraging findings, lingering concerns
Nicholas Turner, MD, assistant professor in the division of infectious diseases at Duke University, Durham, N.C., praised the study for “pushing the boundaries for FMT,” and noted that the methodology appeared sound. Results in the placebo group, however, cast doubt on the generalizability of the findings, he said.
“If you look at the group that received vancomycin plus placebo, their failure rate was really astoundingly high,” Dr. Turner said in an interview, referring to the 67% failure rate in the control group; he noted previous studies had reported failure rates closer to 10%. “I think that just calls into question just a little bit what happened with that control group.”
Dr. Turner said his confidence would go “way, way up” if the findings were reproduced in a larger study. Ideally, these future trials would use fidaxomicin, he added, which is becoming the preferred option over vancomycin for treating C. difficile.
John Y. Kao, MD, professor of medicine and codirector of the FMT program at University of Michigan Medicine, Ann Arbor, offered a different perspective, suggesting that the control group findings shouldn’t overshadow the efficacy of FMT.
“I agree that historical data would tell us that the placebo population should see a much higher response,” Dr. Kao said in an interview. “In my mind though, the success rate of FMT over placebo is what I would expect. The message of the study should be upheld: that FMT is an effective therapy whether it’s given early or, as the way we give it now, as a sort of rescue therapy.”
Despite this confidence in FMT as an efficacious first-line option, Dr. Kao said it is unlikely to be routinely used in this way anytime soon, even if a larger trial echoes the present results.
“We don’t know the long-term risks of FMT therapy, although we’ve been doing this now probably close to 20 years,” Dr. Kao said.
Specifically, Dr. Kao was most concerned about the long-term risk of colon cancer, as mouse models suggest that microbiome characteristics may affect risk level, and risk may vary based on host-microbiome relationships. In other words, an organism may pose no risk in the gut of the donor, but the same may not be true for the recipient.
While increased rates of colon cancer or other serious illnesses have not been detected in humans who have undergone FMT over the past 2 decades, Dr. Kao said that these findings cannot be extrapolated over a patient’s entire lifetime, especially for younger individuals.
“In a patient that’s 80, you would say, yeah, let’s go ahead and treat you [with FMT] as first-line therapy, whereas someone who’s 20, and has maybe another 50 or 60 years longevity, you may not want to give FMT as first-line therapy,” Dr. Kao said.
This study was supported by Innovation Fund Denmark. The investigators disclosed no competing interests. Dr. Turner previously performed statistical analyses for a Merck study comparing vancomycin, fidaxomicin, and metronidazole for C. difficile infection. Dr. Kao disclosed no relevant conflicts of interest.
Fecal microbiota transplantation (FMT) is safe and highly effective as first-line therapy for patients with first or second Clostridioides difficile infection, according to the first randomized, double-blind, placebo-controlled trial of its kind.
Study enrollment was halted after an interim analysis revealed significantly better outcomes among patients who received vancomycin plus FMT versus vancomycin alone, reported lead author Simon Mark Dahl Baunwall, MD, of Aarhus (Denmark) University Hospital and colleagues in The Lancet Gastroenterology & Hepatology.
The investigators noted that the participants represented a real-world patient population, so the data support FMT “as a necessary, effective first-line option” in routine management of C. difficile infection.
“Previous studies have demonstrated clinical cure rates [with FMT] of up to 92%,” Dr. Baunwall and colleagues wrote. “Early use of FMT for first or second C. difficile infection has therapeutic potential, but no formal randomized trials to support use of the approach as a first-line therapy have been done.”
The present trial, conducted at a university hospital in Denmark, involved 42 adult patients with first or second C. difficile infection. Patients were randomized in a 1:1 ratio to receive either vancomycin alone or vancomycin plus FMT. All patients received 125 mg oral vancomycin four times daily for a minimum of 10 days after diagnosis. On day 1 after completion of vancomycin therapy and again between day 3 and 7, patients received either oral FMT or matching placebo, depending on their group. After completing the protocol, patients were followed for 8 weeks or C. difficile recurrence to evaluate resolution of C. difficile–associated diarrhea.
“In this trial, patients were treated with two sequential FMT procedures on separate days,” the investigators noted. “This practice might have overtreated some patients and differs from previous trials. It remains unknown whether optimal effect is achieved by one or two treatments.”
The trial design called for 84 patients, but enrollment was halted after an interim analysis of the above cohort of 42 patients because of significantly lower rate resolution in the placebo group. At the 2-month mark, 90% (95% confidence interval, 70%-99%) of patients in the FMT group had resolution, compared with only 33% (95% CI, 15%-57%) of patients in the placebo group (P = .0003), constituting a 57% (95% CI, 33%-81%) absolute risk reduction.
Most patients experienced adverse events, including 20 in the FMT group and all 21 in the placebo group, although most were transient and nonserious. The most common adverse events were diarrhea, which occurred more frequently in the FMT group (23 vs. 14 events), followed by abdominal pain(14 vs. 11 events) and nausea (12 vs. 5 events).
One limitation of the study was its single-center design with regional uptake; the authors noted that, despite having high statistical power for the clinical effect, the study’s premature termination and low patient number prevent inferences regarding mortality, time to effect, and cost.
“The results of this trial highlight how the use of fecal microbiota transplantation as a first-line treatment can effectively prevent C. difficile recurrence and suggests that microbiota restoration might be necessary to obtain sustained resolution,” the investigators wrote. “At present, only 10% of patients with multiple, recurrent C. difficile infection and indication for FMT receive it. International initiatives address the unmet need, but logistic and regulatory obstacles remain unsolved.”
Encouraging findings, lingering concerns
Nicholas Turner, MD, assistant professor in the division of infectious diseases at Duke University, Durham, N.C., praised the study for “pushing the boundaries for FMT,” and noted that the methodology appeared sound. Results in the placebo group, however, cast doubt on the generalizability of the findings, he said.
“If you look at the group that received vancomycin plus placebo, their failure rate was really astoundingly high,” Dr. Turner said in an interview, referring to the 67% failure rate in the control group; he noted previous studies had reported failure rates closer to 10%. “I think that just calls into question just a little bit what happened with that control group.”
Dr. Turner said his confidence would go “way, way up” if the findings were reproduced in a larger study. Ideally, these future trials would use fidaxomicin, he added, which is becoming the preferred option over vancomycin for treating C. difficile.
John Y. Kao, MD, professor of medicine and codirector of the FMT program at University of Michigan Medicine, Ann Arbor, offered a different perspective, suggesting that the control group findings shouldn’t overshadow the efficacy of FMT.
“I agree that historical data would tell us that the placebo population should see a much higher response,” Dr. Kao said in an interview. “In my mind though, the success rate of FMT over placebo is what I would expect. The message of the study should be upheld: that FMT is an effective therapy whether it’s given early or, as the way we give it now, as a sort of rescue therapy.”
Despite this confidence in FMT as an efficacious first-line option, Dr. Kao said it is unlikely to be routinely used in this way anytime soon, even if a larger trial echoes the present results.
“We don’t know the long-term risks of FMT therapy, although we’ve been doing this now probably close to 20 years,” Dr. Kao said.
Specifically, Dr. Kao was most concerned about the long-term risk of colon cancer, as mouse models suggest that microbiome characteristics may affect risk level, and risk may vary based on host-microbiome relationships. In other words, an organism may pose no risk in the gut of the donor, but the same may not be true for the recipient.
While increased rates of colon cancer or other serious illnesses have not been detected in humans who have undergone FMT over the past 2 decades, Dr. Kao said that these findings cannot be extrapolated over a patient’s entire lifetime, especially for younger individuals.
“In a patient that’s 80, you would say, yeah, let’s go ahead and treat you [with FMT] as first-line therapy, whereas someone who’s 20, and has maybe another 50 or 60 years longevity, you may not want to give FMT as first-line therapy,” Dr. Kao said.
This study was supported by Innovation Fund Denmark. The investigators disclosed no competing interests. Dr. Turner previously performed statistical analyses for a Merck study comparing vancomycin, fidaxomicin, and metronidazole for C. difficile infection. Dr. Kao disclosed no relevant conflicts of interest.
Fecal microbiota transplantation (FMT) is safe and highly effective as first-line therapy for patients with first or second Clostridioides difficile infection, according to the first randomized, double-blind, placebo-controlled trial of its kind.
Study enrollment was halted after an interim analysis revealed significantly better outcomes among patients who received vancomycin plus FMT versus vancomycin alone, reported lead author Simon Mark Dahl Baunwall, MD, of Aarhus (Denmark) University Hospital and colleagues in The Lancet Gastroenterology & Hepatology.
The investigators noted that the participants represented a real-world patient population, so the data support FMT “as a necessary, effective first-line option” in routine management of C. difficile infection.
“Previous studies have demonstrated clinical cure rates [with FMT] of up to 92%,” Dr. Baunwall and colleagues wrote. “Early use of FMT for first or second C. difficile infection has therapeutic potential, but no formal randomized trials to support use of the approach as a first-line therapy have been done.”
The present trial, conducted at a university hospital in Denmark, involved 42 adult patients with first or second C. difficile infection. Patients were randomized in a 1:1 ratio to receive either vancomycin alone or vancomycin plus FMT. All patients received 125 mg oral vancomycin four times daily for a minimum of 10 days after diagnosis. On day 1 after completion of vancomycin therapy and again between day 3 and 7, patients received either oral FMT or matching placebo, depending on their group. After completing the protocol, patients were followed for 8 weeks or C. difficile recurrence to evaluate resolution of C. difficile–associated diarrhea.
“In this trial, patients were treated with two sequential FMT procedures on separate days,” the investigators noted. “This practice might have overtreated some patients and differs from previous trials. It remains unknown whether optimal effect is achieved by one or two treatments.”
The trial design called for 84 patients, but enrollment was halted after an interim analysis of the above cohort of 42 patients because of significantly lower rate resolution in the placebo group. At the 2-month mark, 90% (95% confidence interval, 70%-99%) of patients in the FMT group had resolution, compared with only 33% (95% CI, 15%-57%) of patients in the placebo group (P = .0003), constituting a 57% (95% CI, 33%-81%) absolute risk reduction.
Most patients experienced adverse events, including 20 in the FMT group and all 21 in the placebo group, although most were transient and nonserious. The most common adverse events were diarrhea, which occurred more frequently in the FMT group (23 vs. 14 events), followed by abdominal pain(14 vs. 11 events) and nausea (12 vs. 5 events).
One limitation of the study was its single-center design with regional uptake; the authors noted that, despite having high statistical power for the clinical effect, the study’s premature termination and low patient number prevent inferences regarding mortality, time to effect, and cost.
“The results of this trial highlight how the use of fecal microbiota transplantation as a first-line treatment can effectively prevent C. difficile recurrence and suggests that microbiota restoration might be necessary to obtain sustained resolution,” the investigators wrote. “At present, only 10% of patients with multiple, recurrent C. difficile infection and indication for FMT receive it. International initiatives address the unmet need, but logistic and regulatory obstacles remain unsolved.”
Encouraging findings, lingering concerns
Nicholas Turner, MD, assistant professor in the division of infectious diseases at Duke University, Durham, N.C., praised the study for “pushing the boundaries for FMT,” and noted that the methodology appeared sound. Results in the placebo group, however, cast doubt on the generalizability of the findings, he said.
“If you look at the group that received vancomycin plus placebo, their failure rate was really astoundingly high,” Dr. Turner said in an interview, referring to the 67% failure rate in the control group; he noted previous studies had reported failure rates closer to 10%. “I think that just calls into question just a little bit what happened with that control group.”
Dr. Turner said his confidence would go “way, way up” if the findings were reproduced in a larger study. Ideally, these future trials would use fidaxomicin, he added, which is becoming the preferred option over vancomycin for treating C. difficile.
John Y. Kao, MD, professor of medicine and codirector of the FMT program at University of Michigan Medicine, Ann Arbor, offered a different perspective, suggesting that the control group findings shouldn’t overshadow the efficacy of FMT.
“I agree that historical data would tell us that the placebo population should see a much higher response,” Dr. Kao said in an interview. “In my mind though, the success rate of FMT over placebo is what I would expect. The message of the study should be upheld: that FMT is an effective therapy whether it’s given early or, as the way we give it now, as a sort of rescue therapy.”
Despite this confidence in FMT as an efficacious first-line option, Dr. Kao said it is unlikely to be routinely used in this way anytime soon, even if a larger trial echoes the present results.
“We don’t know the long-term risks of FMT therapy, although we’ve been doing this now probably close to 20 years,” Dr. Kao said.
Specifically, Dr. Kao was most concerned about the long-term risk of colon cancer, as mouse models suggest that microbiome characteristics may affect risk level, and risk may vary based on host-microbiome relationships. In other words, an organism may pose no risk in the gut of the donor, but the same may not be true for the recipient.
While increased rates of colon cancer or other serious illnesses have not been detected in humans who have undergone FMT over the past 2 decades, Dr. Kao said that these findings cannot be extrapolated over a patient’s entire lifetime, especially for younger individuals.
“In a patient that’s 80, you would say, yeah, let’s go ahead and treat you [with FMT] as first-line therapy, whereas someone who’s 20, and has maybe another 50 or 60 years longevity, you may not want to give FMT as first-line therapy,” Dr. Kao said.
This study was supported by Innovation Fund Denmark. The investigators disclosed no competing interests. Dr. Turner previously performed statistical analyses for a Merck study comparing vancomycin, fidaxomicin, and metronidazole for C. difficile infection. Dr. Kao disclosed no relevant conflicts of interest.
FROM THE LANCET GASTROENTEROLOGY & HEPATOLOGY
Malaria vaccine gets special delivery by tiny health personnel
Don’t like needles? Have we got a vaccine for you
Here’s a quick question: How do you turn the most annoying thing ever into something positive?
No, we’re not talking about politicians this time. No, not Elon Musk, either. Infomercials? Guess again. Humidity? Nope, even more annoying than that.
Give up? The most annoying thing ever is mosquitoes. This time, however, NPR reports that mosquitoes have been used to deliver a vaccine for the very disease they’ve been transmitting to their human food sources all these years.
In a recent proof-of-concept trial, investigators used CRISPR technology to genetically modify malaria-causing Plasmodium falciparum sporozoites, which just happen to live in the salivary glands of Anopheles mosquitoes. And since the Plasmodium parasites are already in the mosquitoes, it made sense to use the buzzy little critters as the delivery device for the vaccine.
More sense than a syringe, you ask? Have you ever tried to poke a syringe into the salivary gland of a mosquito? No, we thought not. Well, we can tell you from experience that it’s really, really hard. Never mind how we know. We just do.
The 14 study volunteers – who were paid $4,100 for their participation – were first exposed to hundreds of mosquitoes carrying the altered Plasmodium parasites. Then, to test the vaccine, they were exposed to mosquitoes that had actual, malaria-carrying Plasmodium. Half of the subjects got malaria, so the vaccine was only 50% effective, meaning there’s still work to do.
Meanwhile, the scientists here at LOTMEco are all over this mosquito-delivery business, working on a vaccine to prevent Elon Musk. Plan B involves some sort of really big swatter.
Climate change: Sleeping your life away
It’s no secret that climate change is raising the temperature on everything. You may think you’re getting relief when the sun goes down, but in some places it’s still hot. A new survey conducted in central Japan shows how bad it can be and how higher nighttime temperatures can have a serious impact on people’s health.
That online survey, the Sleep Quality Index for Daily Sleep, enabled the investigators to correlate sleep quality with daily temperature for 1,284 adults in 2011 and 2012 who completed the survey over 10 days.
Not only was there a significant difference in sleep disturbance among younger men (higher) versus older men, but the prevalence of sleep disturbance went up when the daytime temperature was above 24.8° C. They also found that disability-adjusted life-years (DALYs), which measure time lost through premature death and time lived in certain conditions that put one’s health at risk, were 81.8 years for the city of Nagoya (population, 2.2 million) in 2012.
The damage to health from sleep disorders caused by daily temperatures higher than 25° C “is comparable to that of heatstroke and must be addressed,” lead author Tomohiko Ihara of the University of Tokyo said in a written statement.
The researchers hope that this information will help sway legislators to consider the impact of higher nighttime temperatures and that it can be used to provide guidance for better sleep. The solution for now? Sleep with the air conditioner on. Your energy bill might increase, but just think about those DALYs. If using the AC lowers DALYs and increases time lived, then we say it’s worth it.
Maybe it would have been a dragon WITH cancer
If you ask a random person on the street to tell you all they know about the country of Wales, they’ll probably mention two things: One, the contorted collection of jumbled-up letters that is the Welsh language (looking at you, Llanfairpwllgwyngyllgogerychwyrndrobwllllantysiliogogogoch) and, two, the association with dragons. The Welsh flag even has a dragon on it.
With that in mind, take a guess as to what sort of statue art dealer Simon Wingett wanted to build in the Welsh town of Wrexham. No, not a monument to the second-longest place name in the world. Try again. His dragon would not be some piddly little thing either; he wanted a virtual kaiju overlooking the town, with the whole statue to stand about 60 meters high. That’s taller than the original 1954 Godzilla.
Artistic masterpieces may sell for frankly insane prices, but art dealers themselves are not the wealthiest of individuals, so Mr. Wingett needed money to fund his dragon-based dream. Lucky for him, he also happened to be the manager of a cancer charity – initially set up by Mr. Wingett’s father, who had throat cancer – which nominally aimed to provide equipment and resources to cancer patients in the Wrexham area.
Yes, this is going precisely where you think it’s going. From 2011 to 2018, when the charity closed, Mr. Wingett used the charity’s donations to fund his dragon statue – which never actually got built, by the way – to the tune of over 400,000 pounds. Of course, Mr. Wingett came under scrutiny when people started to notice that his cancer charity hadn’t actually done anything charitable since 2011, and he was recently banned by the Welsh High Court from serving as trustee of any charity for 10 years. Oh no, tragedy and horror! Truly a punishment worse than death itself.
Okay fine, he also has to pay back 117,000 pounds to actual legitimate cancer charities. The astute mathematicians out there may notice that 117,000 is a lot less than 400,000. But it’s just as the old saying goes: One-quarter of crime doesn’t pay. You can keep three-quarters of it, though, that’s completely fine.
Don’t like needles? Have we got a vaccine for you
Here’s a quick question: How do you turn the most annoying thing ever into something positive?
No, we’re not talking about politicians this time. No, not Elon Musk, either. Infomercials? Guess again. Humidity? Nope, even more annoying than that.
Give up? The most annoying thing ever is mosquitoes. This time, however, NPR reports that mosquitoes have been used to deliver a vaccine for the very disease they’ve been transmitting to their human food sources all these years.
In a recent proof-of-concept trial, investigators used CRISPR technology to genetically modify malaria-causing Plasmodium falciparum sporozoites, which just happen to live in the salivary glands of Anopheles mosquitoes. And since the Plasmodium parasites are already in the mosquitoes, it made sense to use the buzzy little critters as the delivery device for the vaccine.
More sense than a syringe, you ask? Have you ever tried to poke a syringe into the salivary gland of a mosquito? No, we thought not. Well, we can tell you from experience that it’s really, really hard. Never mind how we know. We just do.
The 14 study volunteers – who were paid $4,100 for their participation – were first exposed to hundreds of mosquitoes carrying the altered Plasmodium parasites. Then, to test the vaccine, they were exposed to mosquitoes that had actual, malaria-carrying Plasmodium. Half of the subjects got malaria, so the vaccine was only 50% effective, meaning there’s still work to do.
Meanwhile, the scientists here at LOTMEco are all over this mosquito-delivery business, working on a vaccine to prevent Elon Musk. Plan B involves some sort of really big swatter.
Climate change: Sleeping your life away
It’s no secret that climate change is raising the temperature on everything. You may think you’re getting relief when the sun goes down, but in some places it’s still hot. A new survey conducted in central Japan shows how bad it can be and how higher nighttime temperatures can have a serious impact on people’s health.
That online survey, the Sleep Quality Index for Daily Sleep, enabled the investigators to correlate sleep quality with daily temperature for 1,284 adults in 2011 and 2012 who completed the survey over 10 days.
Not only was there a significant difference in sleep disturbance among younger men (higher) versus older men, but the prevalence of sleep disturbance went up when the daytime temperature was above 24.8° C. They also found that disability-adjusted life-years (DALYs), which measure time lost through premature death and time lived in certain conditions that put one’s health at risk, were 81.8 years for the city of Nagoya (population, 2.2 million) in 2012.
The damage to health from sleep disorders caused by daily temperatures higher than 25° C “is comparable to that of heatstroke and must be addressed,” lead author Tomohiko Ihara of the University of Tokyo said in a written statement.
The researchers hope that this information will help sway legislators to consider the impact of higher nighttime temperatures and that it can be used to provide guidance for better sleep. The solution for now? Sleep with the air conditioner on. Your energy bill might increase, but just think about those DALYs. If using the AC lowers DALYs and increases time lived, then we say it’s worth it.
Maybe it would have been a dragon WITH cancer
If you ask a random person on the street to tell you all they know about the country of Wales, they’ll probably mention two things: One, the contorted collection of jumbled-up letters that is the Welsh language (looking at you, Llanfairpwllgwyngyllgogerychwyrndrobwllllantysiliogogogoch) and, two, the association with dragons. The Welsh flag even has a dragon on it.
With that in mind, take a guess as to what sort of statue art dealer Simon Wingett wanted to build in the Welsh town of Wrexham. No, not a monument to the second-longest place name in the world. Try again. His dragon would not be some piddly little thing either; he wanted a virtual kaiju overlooking the town, with the whole statue to stand about 60 meters high. That’s taller than the original 1954 Godzilla.
Artistic masterpieces may sell for frankly insane prices, but art dealers themselves are not the wealthiest of individuals, so Mr. Wingett needed money to fund his dragon-based dream. Lucky for him, he also happened to be the manager of a cancer charity – initially set up by Mr. Wingett’s father, who had throat cancer – which nominally aimed to provide equipment and resources to cancer patients in the Wrexham area.
Yes, this is going precisely where you think it’s going. From 2011 to 2018, when the charity closed, Mr. Wingett used the charity’s donations to fund his dragon statue – which never actually got built, by the way – to the tune of over 400,000 pounds. Of course, Mr. Wingett came under scrutiny when people started to notice that his cancer charity hadn’t actually done anything charitable since 2011, and he was recently banned by the Welsh High Court from serving as trustee of any charity for 10 years. Oh no, tragedy and horror! Truly a punishment worse than death itself.
Okay fine, he also has to pay back 117,000 pounds to actual legitimate cancer charities. The astute mathematicians out there may notice that 117,000 is a lot less than 400,000. But it’s just as the old saying goes: One-quarter of crime doesn’t pay. You can keep three-quarters of it, though, that’s completely fine.
Don’t like needles? Have we got a vaccine for you
Here’s a quick question: How do you turn the most annoying thing ever into something positive?
No, we’re not talking about politicians this time. No, not Elon Musk, either. Infomercials? Guess again. Humidity? Nope, even more annoying than that.
Give up? The most annoying thing ever is mosquitoes. This time, however, NPR reports that mosquitoes have been used to deliver a vaccine for the very disease they’ve been transmitting to their human food sources all these years.
In a recent proof-of-concept trial, investigators used CRISPR technology to genetically modify malaria-causing Plasmodium falciparum sporozoites, which just happen to live in the salivary glands of Anopheles mosquitoes. And since the Plasmodium parasites are already in the mosquitoes, it made sense to use the buzzy little critters as the delivery device for the vaccine.
More sense than a syringe, you ask? Have you ever tried to poke a syringe into the salivary gland of a mosquito? No, we thought not. Well, we can tell you from experience that it’s really, really hard. Never mind how we know. We just do.
The 14 study volunteers – who were paid $4,100 for their participation – were first exposed to hundreds of mosquitoes carrying the altered Plasmodium parasites. Then, to test the vaccine, they were exposed to mosquitoes that had actual, malaria-carrying Plasmodium. Half of the subjects got malaria, so the vaccine was only 50% effective, meaning there’s still work to do.
Meanwhile, the scientists here at LOTMEco are all over this mosquito-delivery business, working on a vaccine to prevent Elon Musk. Plan B involves some sort of really big swatter.
Climate change: Sleeping your life away
It’s no secret that climate change is raising the temperature on everything. You may think you’re getting relief when the sun goes down, but in some places it’s still hot. A new survey conducted in central Japan shows how bad it can be and how higher nighttime temperatures can have a serious impact on people’s health.
That online survey, the Sleep Quality Index for Daily Sleep, enabled the investigators to correlate sleep quality with daily temperature for 1,284 adults in 2011 and 2012 who completed the survey over 10 days.
Not only was there a significant difference in sleep disturbance among younger men (higher) versus older men, but the prevalence of sleep disturbance went up when the daytime temperature was above 24.8° C. They also found that disability-adjusted life-years (DALYs), which measure time lost through premature death and time lived in certain conditions that put one’s health at risk, were 81.8 years for the city of Nagoya (population, 2.2 million) in 2012.
The damage to health from sleep disorders caused by daily temperatures higher than 25° C “is comparable to that of heatstroke and must be addressed,” lead author Tomohiko Ihara of the University of Tokyo said in a written statement.
The researchers hope that this information will help sway legislators to consider the impact of higher nighttime temperatures and that it can be used to provide guidance for better sleep. The solution for now? Sleep with the air conditioner on. Your energy bill might increase, but just think about those DALYs. If using the AC lowers DALYs and increases time lived, then we say it’s worth it.
Maybe it would have been a dragon WITH cancer
If you ask a random person on the street to tell you all they know about the country of Wales, they’ll probably mention two things: One, the contorted collection of jumbled-up letters that is the Welsh language (looking at you, Llanfairpwllgwyngyllgogerychwyrndrobwllllantysiliogogogoch) and, two, the association with dragons. The Welsh flag even has a dragon on it.
With that in mind, take a guess as to what sort of statue art dealer Simon Wingett wanted to build in the Welsh town of Wrexham. No, not a monument to the second-longest place name in the world. Try again. His dragon would not be some piddly little thing either; he wanted a virtual kaiju overlooking the town, with the whole statue to stand about 60 meters high. That’s taller than the original 1954 Godzilla.
Artistic masterpieces may sell for frankly insane prices, but art dealers themselves are not the wealthiest of individuals, so Mr. Wingett needed money to fund his dragon-based dream. Lucky for him, he also happened to be the manager of a cancer charity – initially set up by Mr. Wingett’s father, who had throat cancer – which nominally aimed to provide equipment and resources to cancer patients in the Wrexham area.
Yes, this is going precisely where you think it’s going. From 2011 to 2018, when the charity closed, Mr. Wingett used the charity’s donations to fund his dragon statue – which never actually got built, by the way – to the tune of over 400,000 pounds. Of course, Mr. Wingett came under scrutiny when people started to notice that his cancer charity hadn’t actually done anything charitable since 2011, and he was recently banned by the Welsh High Court from serving as trustee of any charity for 10 years. Oh no, tragedy and horror! Truly a punishment worse than death itself.
Okay fine, he also has to pay back 117,000 pounds to actual legitimate cancer charities. The astute mathematicians out there may notice that 117,000 is a lot less than 400,000. But it’s just as the old saying goes: One-quarter of crime doesn’t pay. You can keep three-quarters of it, though, that’s completely fine.
Glucocorticoid-Induced Bone Loss: Dietary Supplementation Recommendations to Reduce the Risk for Osteoporosis and Osteoporotic Fractures
Glucocorticoids (GCs) are among the most widely prescribed medications in dermatologic practice. Although GCs are highly effective anti-inflammatory agents, long-term systemic therapy can result in dangerous adverse effects, including GC-induced osteoporosis (GIO), a bone disease associated with a heightened risk for fragility fractures.1,2 In the United States, an estimated 10.2 million adults have osteoporosis—defined as a T-score lower than −2.5 measured via a bone densitometry scan—and 43.4 million adults have low bone mineral density (BMD).3,4 The prevalence of osteoporosis is increasing, and the diagnosis is more common in females and adults 55 years and older.2 More than 2 million individuals have osteoporosis-related fractures annually, and the mortality risk is increased at 5 and 10 years following low-energy osteoporosis-related fractures.3-5
Glucocorticoid therapy is the leading iatrogenic cause of secondary osteoporosis. As many as 30% of all patients treated with systemic GCs for more than 6 months develop GIO.1,6,7 Glucocorticoid-induced BMD loss occurs at a rate of 6% to 12% of total BMD during the first year, slowing to approximately 3% per year during subsequent therapy.1 The risk for insufficiency fractures increases by as much as 75% from baseline in adults with rheumatic, pulmonary, and skin disorders within the first 3 months of therapy and peaks at approximately 12 months.1,2
Despite the risks, many long-term GC users never receive therapy to prevent bone loss; others are only started on therapy once they have sustained an insufficiency fracture. A 5-year international observational study including more than 40,000 postmenopausal women found that only 51% of patients who were on continuous GC therapy were undergoing BMD testing and appropriate medical management.8 This review highlights the existing evidence on the risks of osteoporosis and osteoporotic (OP) fractures in the setting of topical, intralesional, intramuscular, and systemic GC treatment, as well as recommendations for nutritional supplementation to reduce these risks.
Pathophysiology
The pathophysiology of GIO is multifactorial and occurs in both early and late phases.9,10 The early phase is characterized by rapid BMD reduction due to excessive bone resorption. The late phase is characterized by slower and more progressive BMD reduction due to impaired bone formation.9 At the osteocyte level, GCs decrease cell viability and induce apoptosis.11 At the osteoblast level, GCs impair cell replication and differentiation and have proapoptotic effects, resulting in decreased cell numbers and subsequent bone formation.10 At the osteoclast level, GCs increase expression of pro-osteoclastic cytokines and decrease mature osteoclast apoptosis, resulting in an expanded osteoclastic life span and prolonged bone resorption.12,13 Indirectly, GCs alter calcium metabolism by decreasing gastrointestinal calcium absorption and impairing renal absorption.14,15
GCs and Osteoporosis
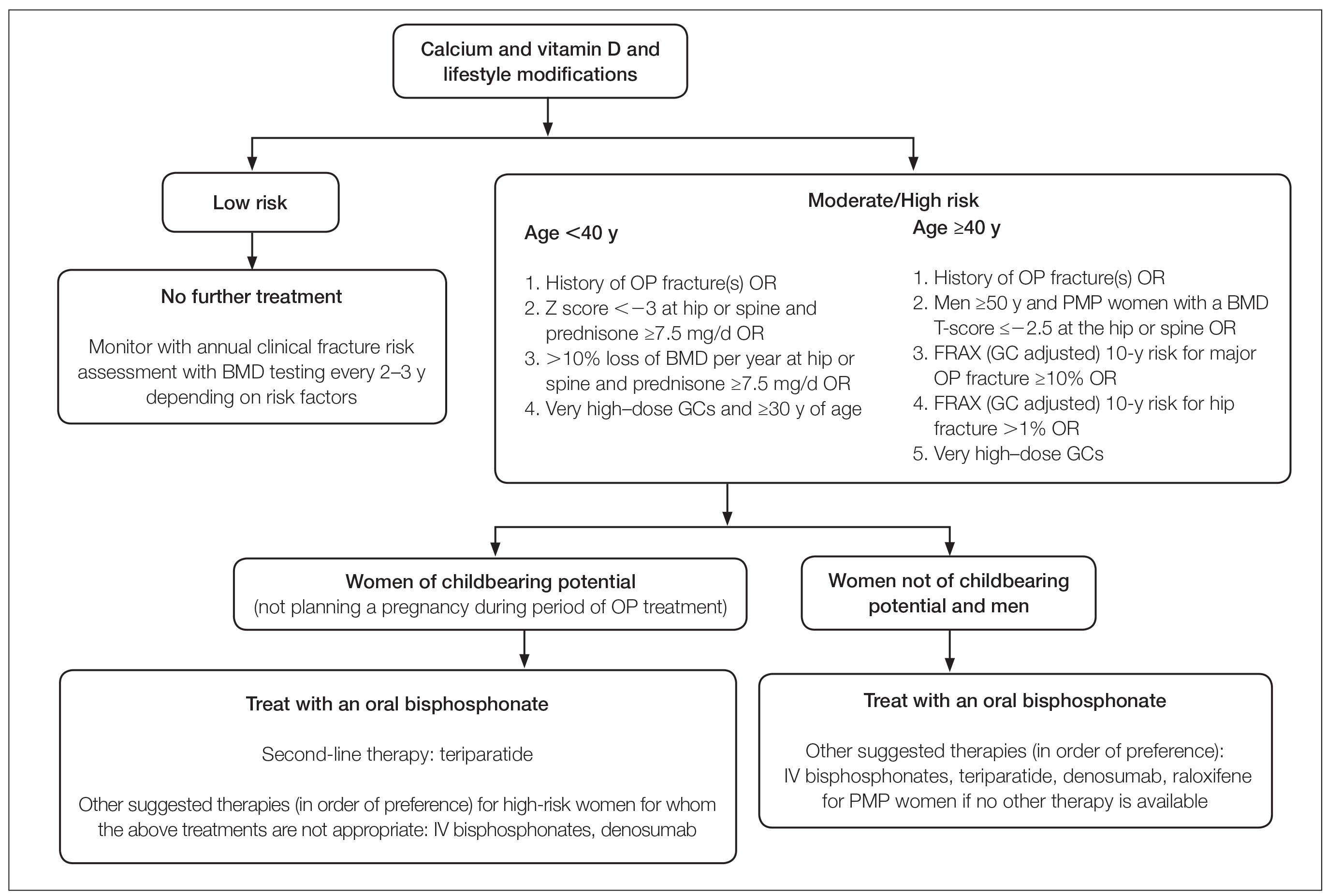
Oral GCs—Glucocorticoid-induced osteoporosis and fracture risk are dose and duration dependent.6 A study of 244,235 patients taking GCs and 244,235 controls found the relative risk of vertebral fracture was 1.55 (range, 1.20–2.01) for daily prednisone use at less than 2.5 mg, 2.59 (range, 2.16–3.10) for daily prednisone use from 2.5 to 7.4 mg, and 5.18 (range, 4.25–6.31) for daily doses of 7.5 mg or higher; the relative risk for hip fractures was 0.99 (range, 0.82–1.20), 1.77 (range, 1.55–2.02), and 2.27 (range, 1.94–2.66), respectively.16 Another large retrospective cohort study found that continuous treatment with prednisone 10 mg/d for more than 90 days compared to no GC exposure increased the risk for hip fractures 7-fold and 17-fold for vertebral fractures.17 Although the minimum cumulative dose of GCs known to cause osteoporosis is not clearly established, the American College of Rheumatology has proposed an algorithm as a basic approach to anticipate, prevent, and treat GIO (Figure).18,19 Fracture risk should be assessed in all patients who are prescribed prednisone 2.5 mg/d for 3 months or longer or an anticipated cumulative dose of more than 1 g per year. Patients 40 years and older with anticipated GC use of 3 months or longer should have both a bone densitometry scan and a Fracture Risk Assessment (FRAX) score. The FRAX tool estimates the 10-year probability of fracture in patients aged 40 to 80 years, and those patients can be further risk stratified as low (FRAX <10%), moderate (FRAX 10%–19%), or high (FRAX ≥20%) risk. In patients with moderate to high risk of fracture (FRAX >10%), initiation of pharmacologic treatment or referral to a metabolic bone specialist should be considered.18,19 First-line therapy is an oral bisphosphonate, and second-line therapies include intravenous bisphosphonates, teriparatide, denosumab, or raloxifene for patients at high risk for GIO.19 Adults younger than 40 years with a history of OP fracture or considerable risk factors for OP fractures should have a bone densitometry scan, and, if results are abnormal, the patient should be referred to a metabolic bone specialist. Those with low fracture risk based on bone densitometry and FRAX and those with no risk factors should be assessed annually for bone health (additional risk factors, GC dose and duration, bone densitometry/FRAX if indicated).18 In addition to GC dose and duration, additional risk factors for GIO, which are factored into the FRAX tool, include advanced age, low body mass index, history of bone fracture, smoking, excessive alcohol use (≥3 drinks/d), history of falls, low BMD, family history of bone fracture, and hypovitaminosis D.6
Topical GCs—Although there is strong evidence and clear guidelines regarding oral GIO, there is a dearth of data surrounding OP risk due to treatment with topical GCs. A recent retrospective nationwide Danish study evaluating the risk of osteoporosis and major OP fracture in 723,251 adults treated with potent or very potent topical steroids sought to evaluate these risks.20 Patients were included if they had filled prescriptions of at least 500 g of topical mometasone or an equivalent alternative. The investigators reported a 3% increase in relative risk of osteoporosis and major OP fracture with doubling of the cumulative topical GC dose (hazard ratio [HR], 1.03 [95% CI, 1.02-1.04] for both). The overall population-attributable risk was 4.3% (95% CI, 2.7%-5.8%) for osteoporosis and 2.7% (95% CI, 1.7%-3.8%) for major OP fracture. Notably, at least 10,000 g of mometasone was required for 1 additional patient to have a major OP fracture.20 In a commentary based on this study, Jackson21 noted that the number of patient-years of topical GC use needed for 1 fracture was 4-fold higher than that for high-dose oral GCs (40 mg/d prednisolone for ≥30 days). Another study assessed the effects of topical GCs on BMD in adults with moderate to severe atopic dermatitis over a 2-year period.22 No significant difference in BMD assessed via bone densitometry of either the lumbar spine or total hip at baseline or at 2-year follow-up was reported for either group treated with corticosteroids (<75 g per month or ≥75 g per month). Of note, the authors did not account for steroid potency, which ranged from class 1 through class 4.22 Although limited data exist, these studies suggest topical GCs used at conventional doses with appropriate breaks in therapy will not substantially increase risk for GIO or OP fracture; however, in the small subset of patients requiring chronic use of superpotent topical corticosteroids with other OP risk factors, transitioning to non–GC-based therapy or initiating bone health therapy may be advised to improve patient outcomes. Risk assessment, as in cases of chronic topical GC use, may be beneficial.
Intralesional GCs—Intralesional GCs are indicated for numerous inflammatory conditions including alopecia areata, discoid lupus erythematosus, keloids, and granuloma annulare. It generally is accepted that doses of triamcinolone acetonide should not exceed 20 mg per session spaced at least 3 weeks apart or up to 40 mg per month.18 One study demonstrated that doses of triamcinolone diacetate of 25 mg or less were unlikely to produce systemic effects and were determined to be a safe dose for intralesional injections.23 A retrospective cross-sectional case series including 18 patients with alopecia areata reported decreased BMD in 9 patients receiving intralesional triamcinolone acetonide 10 mg/mL at 4- to 8-week intervals for at least 20 months, with cumulative doses greater than 500 mg. This was particularly notable in postmenopausal women and men older than 50 years; participants with a body mass index less than 18.5 kg/m2, history of a stress fracture, family history of osteopenia or osteoporosis, and history of smoking; and those who did not regularly engage in weight-bearing exercises.24 Patients receiving long-term (ie, >1 year) intralesional steroids should be evaluated for osteoporosis risk and preventative strategies should be considered (ie, regular weight-bearing exercises, calcium and vitamin D supplementation, bisphosphate therapy). As with topical GCs, there are no clear guidelines for risk assessment or treatment recommendations for GIO.
Intramuscular GCs—The data regarding intramuscular (IM) GCs and dermatologic disease is severely limited, and to the best of our knowledge, no studies specifically assess the risk for GIO or fracture secondary to intramuscular GCs; however, a retrospective study of 27 patients (4 female, 23 male; mean age, 33 years [range, 12–61 years]) with refractory alopecia areata receiving IM triamcinolone acetonide (40 mg every 4 weeks for 3–6 months) reported 1 patient (a 56-year-old woman) with notably decreased bone densitometry from baseline requiring treatment discontinuation.25 No other patients at risk for osteoporosis had decreased BMD from treatment with IM triamcinolone; however, it was noted that 1 month following treatment, 10 of 11 assessed patients demonstrated decreased levels of morning serum cortisol and plasma adrenocorticotropic hormone—despite baseline levels within reference range—that resolved 3 months after treatment completion,25 which suggests a prolonged release of IM triamcinolone and sustained systemic effect. One systematic review of 342 patients with dermatologic diseases treated with IM corticosteroids found the primary side effects included dysmenorrhea, injection-site lipoatrophy, and adrenocortical suppression, with only a single reported case of low BMD.26 Given the paucity of evidence, additional studies are required to assess the effect of IM triamcinolone on BMD and risk for major OP fractures with regard to dosing and frequency. As there are no clear guidelines for osteoporosis evaluation in the setting of intramuscular GCs, it may be prudent to follow the algorithmic model recommended for oral steroids when anticipating at least 3 months of intramuscular GCs.
Diet and Prevention of Bone Loss
Given the profound impact that systemic GCs have on osteoporosis and fracture risk and the sparse data regarding risk from topical, intralesional, or intramuscular GCs, diet and nutrition represent a simple, safe, and potentially preventative method of slowing BMD loss and minimizing fracture risk. In higher-risk patients, nutritional assessment in combination with medical therapy also is likely warranted.
Calcium and Vitamin D3—Patients treated with any GC dose longer than 3 months should undergo calcium and vitamin D optimization.19 Exceptions for supplementation include certain patients with sarcoidosis, which can be associated with high vitamin D levels; patients with a history of hypercalcemia or hypercalciuria; and patients with chronic kidney disease.6 In a meta-analysis including 30,970 patients in 8 randomized controlled trials, calcium (500–1200 mg/d) and vitamin D (400–800 IU/d) supplementation reduced the risk of total fractures by 15% (summary relative risk estimate, 0.85 [95% CI, 0.73-0.98]) and hip fractures by 30% (summary relative risk estimate, 0.70 [95% CI, 0.56-0.87]).4 One double-blind, placebo-controlled clinical trial conducted by the Women’s Health Initiative that included 36,282 postmenopausal women who were taking 1000 mg of calcium and 400 IU of vitamin D3 daily for more than 5 years reported an HR of 0.62 (95% CI, 0.38-1.00) for hip fracture for supplementation vs placebo.27 Lastly, a 2016 Cochrane Review including 12 randomized trials and 1343 participants reported a 43% lower risk of new vertebral fractures following supplementation with calcium, vitamin D, or both compared with controls.28
Specific recommendations for calcium and vitamin D3 supplementation vary based on age and sex. The US Preventive Services Task Force concluded that insufficient evidence exists to support calcium and vitamin D3 supplementation in asymptomatic men and premenopausal women.29 The National Osteoporosis Foundation (NOF) supports the use of calcium supplementation for fracture risk reduction in middle-aged and older adults.4 Furthermore, the NOF supports the Institute of Medicine recommendations31 that men aged 50 to 70 years consume 1000 mg/d of calcium and that women 51 years and older as well as men 71 years and older consume 1200 mg/d of calcium.30 The NOF recommends 800 to 1000 IU/d of vitamin D in adults 50 years and older, while the Institute of Medicine recommends 600 IU/d in adults 70 years and younger and 800 IU/d in adults 71 years and older.31 These recommendations are similar to both the Endocrine Society and the American Geriatric Society.32,33 Total calcium should not exceed 2000 mg/d due to risk of adverse effects.
Dietary sources of vitamin D include fatty fish, mushrooms, and fortified dairy products, though recommended doses rarely can be achieved through diet alone.34 Dairy products are the primary source of dietary calcium. Other high-calcium foods include green leafy vegetables, nuts and seeds, soft-boned fish, and fortified beverages and cereals.35
Probiotics—A growing body of evidence suggests that probiotics may be beneficial in promoting bone health by improving calcium homeostasis, reducing risk for hyperparathyroidism secondary to GC therapy, and decreasing age-related bone resorption.36 An animal study demonstrated that probiotics can regulate bone resorption and formation as well as reduce bone loss secondary to GC therapy.37 A randomized, double-blind, placebo-controlled, multicenter trial randomly assigned 249 healthy, early postmenopausal women to receive probiotic treatment containing 3 lactobacillus strains (Lactobacillus paracasei DSM 13434, Lactobacillus plantarum DSM 15312, and L plantarum DSM 15313) or placebo once daily for 12 months.38 Bone mineral density was measured at baseline and at 12 months. Of the 234 participants who completed the study, lactobacillus treatment reduced lumbosacral BMD loss compared to the placebo group (mean difference, 0.71% [95% CI, 0.06-1.35]). They also reported significant lumbosacral BMD loss in the placebo group (−0.72% [95% CI, −1.22 to −0.22]) compared to no BMD loss in the group treated with lactobacillus (−0.01% [95% CI, −0.50 to 0.48]).38 Although the data may be encouraging, more studies are needed to determine if probiotics should be regarded as an adjuvant treatment to calcium, vitamin D, and pharmacologic therapy for long-term prevention of bone loss in the setting of GIO.39 Because existing studies on probiotics include varying compositions and doses, larger studies with consistent supplementation are required. Encouraging probiotic intake through fermented dairy products may represent a simple low-risk intervention to support bone health.
Anti-inflammatory Diet—The traditional Mediterranean diet is rich in fruits, vegetables, fish, nuts, whole grains, legumes, and monounsaturated fats and low in meat and dairy products. The Mediterranean diet has been shown to be modestly protective against osteoporosis and fracture risk. A large US observational study including 93,676 women showed that those with the highest quintile of the alternate Mediterranean diet score had a lower risk for hip fracture (HR, 0.80 [95% CI, 0.66-0.97]), with an absolute risk reduction of 0.29% and number needed to treat at 342.40 A multicenter study involving adults from 8 European countries found that increased adherence to the Mediterranean diet was associated with a 7% reduction in hip fracture incidence (HR per 1 unit increase in Mediterranean diet, 0.93 [95% CI, 0.89-0.98]). High vegetable and fruit intake was associated with decreased hip fracture incidence (HR, 0.86 and 0.89 [95% CI, 0.79-0.94 and 0.82-0.97, respectively]), and high meat and excessive ethanol consumption were associated with increased fracture incidence (HR, 1.18 and 1.74 [95% CI, 1.06-1.31 and 1.32-2.31, respectively]).41 Similarly, a large observational study in Sweden that included 37,903 men and 33,403 women reported similar findings, noting a 6% lower hip fracture rate per one unit increase in alternate Mediterranean diet score (adjusted HR, 0.94 [95% CI, 0.92-0.96]).42 This is thought to be due in part to higher levels of dietary vitamin D present in many foods traditionally included in the Mediterranean diet.43 Additionally, olive oil, a staple in the Mediterranean diet, appears to reduce bone loss by promoting osteoblast proliferation and maturation, inhibiting bone resorption, suppressing oxidative stress and inflammation, and increasing calcium deposition in the extracellular matrix.44,45 Fruits, vegetables, legumes, and nuts also are rich in minerals including potassium and magnesium, which are important in bone health to promote osteoblast proliferation and vitamin D activation.36,46-48
Final Thoughts
Osteoporosis-related fractures are common and are associated with high morbidity and health care costs. Dermatologists using and prescribing corticosteroids must be aware of the risk for GIO, particularly in patients with a pre-existing diagnosis of osteopenia or osteoporosis. There likely is no oral corticosteroid dose that does not increase a patient’s risk for osteoporosis; therefore, oral GCs should be used at the lowest effective daily dose for the shortest duration possible. Patients with an anticipated duration of at least 3 months—regardless of dose—should be assessed for their risk for GIO. Patients using topical and intralesional corticosteroids are unlikely to develop GIO; however, those with risk factors and a considerable cumulative dose may warrant further evaluation. In all cases, we advocate for supplementing with calcium and vitamin D as well as promoting probiotic intake and the Mediterranean diet. Those at moderate to high risk for fracture may require additional medical therapy. Dermatologists are uniquely positioned to identify this at-risk population, and because osteoporosis is a chronic illness, primary care providers should be notified of prolonged GC therapy to help with risk assessment, initiation of vitamin and mineral supplementation, and follow-up with metabolic bone health specialists. Through a multidisciplinary approach and patient education, GIO and the potential risk for fracture can be successfully mitigated in most patients.
- Weinstein RS. Clinical practice. glucocorticoid-induced bone disease. N Engl J Med. 2011;365:62-70.
- Buckley L, Humphrey MB. Glucocorticoid-induced osteoporosis. N Engl J Med. 2018;379:2547-2556.
- Wright NC, Looker AC, Saag KG, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29:2520-2526.
- Weaver CM, Alexander DD, Boushey CJ, et al. Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int. 2016;27:367-376.
- Bliuc D, Nguyen ND, Milch VE, et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301:513-521.
- Caplan A, Fett N, Rosenbach M, et al. Prevention and management of glucocorticoid-induced side effects: a comprehensive review: a review of glucocorticoid pharmacology and bone health. J Am Acad Dermatol. 2017;76:1-9.
- Gudbjornsson B, Juliusson UI, Gudjonsson FV. Prevalence of long term steroid treatment and the frequency of decision making to prevent steroid induced osteoporosis in daily clinical practice. Ann Rheum Dis. 2002;61:32-36.
- Silverman S, Curtis J, Saag K, et al. International management of bone health in glucocorticoid-exposed individuals in the observational GLOW study. Osteoporos Int. 2015;26:419-420.
- Canalis E, Bilezikian JP, Angeli A, et al. Perspectives on glucocorticoid-induced osteoporosis. Bone. 2004;34:593-598.
- Canalis E, Mazziotti G, Giustina A, et al. Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int. 2007;18:1319-1328.
- Lane NE, Yao W, Balooch M, et al. Glucocorticoid-treated mice have localized changes in trabecular bone material properties and osteocyte lacunar size that are not observed in placebo-treated or estrogen-deficient mice. J Bone Miner Res. 2006;21:466-476.
- Hofbauer LC, Gori F, Riggs BL, et al. Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblastic lineage cells: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology. 1999;140:4382-4389.
- Jia D, O’Brien CA, Stewart SA, et al. Glucocorticoids act directly on osteoclasts to increase their life span and reduce bone density. Endocrinology. 2006;147:5592-5599.
- Mazziotti G, Angeli A, Bilezikian JP, et al. Glucocorticoid-induced osteoporosis: an update. Trends Endocrinol Metab. 2006;17:144-149.
- Huybers S, Naber TH, Bindels RJ, et al. Prednisolone-induced Ca2+ malabsorption is caused by diminished expression of the epithelial Ca2+ channel TRPV6. Am J Physiol Gastrointest Liver Physiol. 2007;292:G92-G97.
- Van Staa TP, Leufkens HG, Abenhaim L, et al. Use of oral corticosteroids and risk of fractures. J Bone Miner Res. 2000;15:993-1000.
- Steinbuch M, Youket TE, Cohen S. Oral glucocorticoid use is associated with an increased risk of fracture. Osteoporos Int. 2004;15:323-328.
- Lupsa BC, Insogna KL, Micheletti RG, et al. Corticosteroid use in chronic dermatologic disorders and osteoporosis. Int J Womens Dermatol. 2021;7:545-551.
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken). 2017;69:1095-1110.
- Egeberg A, Schwarz P, Harsløf T, et al. Association of potent and very potent topical corticosteroids and the risk of osteoporosis and major osteoporotic fractures. JAMA Dermatol. 2021;157:275-282.
- Jackson RD. Topical corticosteroids and glucocorticoid-induced osteoporosis-cumulative dose and duration matter. JAMA Dermatol. 2021;157:269-270.
- van Velsen SG, Haeck IM, Knol MJ, et al. Two-year assessment of effect of topical corticosteroids on bone mineral density in adults with moderate to severe atopic dermatitis. J Am Acad Dermatol. 2012;66:691-693.
- McGugan AD, Shuster S, Bottoms E. Adrenal suppression from intradermal triamcinolone. J Invest Dermatol. 1963;40:271-272.
- Samrao A, Fu JM, Harris ST, et al. Bone mineral density in patients with alopecia areata treated with long-term intralesional corticosteroids. J Drugs Dermatol. 2013;12:E36-E40.
- Seo J, Lee YI, Hwang S, et al. Intramuscular triamcinolone acetonide: an undervalued option for refractory alopecia areata. J Dermatol. 2017;44:173-179.
- Thomas LW, Elsensohn A, Bergheim T, et al. Intramuscular steroids in the treatment of dermatologic disease: a systematic review. J Drugs Dermatol. 2018;17:323-329.
- Prentice RL, Pettinger MB, Jackson RD, et al. Health risks and benefits from calcium and vitamin D supplementation: Women’s Health Initiative clinical trial and cohort study. Osteoporos Int. 2013;24:567-580.
- Allen CS, Yeung JH, Vandermeer B, et al. Bisphosphonates for steroid-induced osteoporosis. Cochrane Database Syst Rev. 2016;10:CD001347. doi:10.1002/14651858.CD001347.pub2
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, et al. Vitamin D, calcium, or combined supplementation for the primary prevention of fractures in community-dwelling adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319:1592-1599.
- Cosman F, de Beur SJ, LeBoff MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359-2381.
- Institute of Medicine. Dietary reference intakes for calcium and vitamin D. Washington, DC: National Academies Press; 2011.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911-1930.
- American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults. Recommendations abstracted from the American Geriatrics Society Consensus Statement on vitamin D for prevention of falls and their consequences. J Am Geriatr Soc. 2014;62:147-152.
- Vitamin D fact sheet for health professionals. National Institutes of Health Office of Dietary Supplements website. Updated August 12, 2022. Accessed September 16, 2022. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
- Calcium fact sheet for health professionals. National Institutes of Health Office of Dietary Supplements website. Updated June 2, 2022. Accessed September 16, 2022. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/
- Muñoz-Garach A, García-Fontana B, Muñoz-Torres M. Nutrients and dietary patterns related to osteoporosis. Nutrients. 2020;12:1986.
- Schepper JD, Collins F, Rios-Arce ND, et al. Involvement of the gut microbiota and barrier function in glucocorticoid-induced osteoporosis. J Bone Miner Res. 2020;35:801-820.
- Jansson PA, Curiac D, Ahrén IL, et al. Probiotic treatment using a mix of three Lactobacillus strains for lumbar spine bone loss in postmenopausal women: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet Rheumatol. 2019;1:E154-E162.
- Rizzoli R, Biver E. Are probiotics the new calcium and vitamin D for bone health? Curr Osteoporos Rep. 2020;18:273-284.
- Haring B, Crandall CJ, Wu C, et al. Dietary patterns and fractures in postmenopausal women: results from the Women’s Health Initiative. JAMA Intern Med. 2016;176:645-652.
- Benetou V, Orfanos P, Pettersson-Kymmer U, et al. Mediterranean diet and incidence of hip fractures in a European cohort. Osteoporos Int. 2013;24:1587-1598.
- Byberg L, Bellavia A, Larsson SC, et al. Mediterranean diet and hip fracture in Swedish men and women. J Bone Miner Res. 2016;31:2098-2105.
- Zupo R, Lampignano L, Lattanzio A, et al. Association between adherence to the Mediterranean diet and circulating vitamin D levels. Int J Food Sci Nutr. 2020;71:884-890.
- Chin KY, Ima-Nirwana S. Olives and bone: a green osteoporosis prevention option. Int J Environ Res Public Health. 2016;13:755.
- García-Martínez O, Rivas A, Ramos-Torrecillas J, et al. The effect of olive oil on osteoporosis prevention. Int J Food Sci Nutr. 2014;65:834-840.
- Uwitonze AM, Razzaque MS. Role of magnesium in vitamin D activation and function. J Am Osteopath Assoc. 2018;118:181-189.
- Veronese N, Stubbs B, Solmi M, et al. Dietary magnesium intake and fracture risk: data from a large prospective study. Br J Nutr. 2017;117:1570-1576.
- Kong SH, Kim JH, Hong AR, et al. Dietary potassium intake is beneficial to bone health in a low calcium intake population: the Korean National Health and Nutrition Examination Survey (KNHANES)(2008-2011). Osteoporos Int. 2017;28:1577-1585.
Glucocorticoids (GCs) are among the most widely prescribed medications in dermatologic practice. Although GCs are highly effective anti-inflammatory agents, long-term systemic therapy can result in dangerous adverse effects, including GC-induced osteoporosis (GIO), a bone disease associated with a heightened risk for fragility fractures.1,2 In the United States, an estimated 10.2 million adults have osteoporosis—defined as a T-score lower than −2.5 measured via a bone densitometry scan—and 43.4 million adults have low bone mineral density (BMD).3,4 The prevalence of osteoporosis is increasing, and the diagnosis is more common in females and adults 55 years and older.2 More than 2 million individuals have osteoporosis-related fractures annually, and the mortality risk is increased at 5 and 10 years following low-energy osteoporosis-related fractures.3-5
Glucocorticoid therapy is the leading iatrogenic cause of secondary osteoporosis. As many as 30% of all patients treated with systemic GCs for more than 6 months develop GIO.1,6,7 Glucocorticoid-induced BMD loss occurs at a rate of 6% to 12% of total BMD during the first year, slowing to approximately 3% per year during subsequent therapy.1 The risk for insufficiency fractures increases by as much as 75% from baseline in adults with rheumatic, pulmonary, and skin disorders within the first 3 months of therapy and peaks at approximately 12 months.1,2
Despite the risks, many long-term GC users never receive therapy to prevent bone loss; others are only started on therapy once they have sustained an insufficiency fracture. A 5-year international observational study including more than 40,000 postmenopausal women found that only 51% of patients who were on continuous GC therapy were undergoing BMD testing and appropriate medical management.8 This review highlights the existing evidence on the risks of osteoporosis and osteoporotic (OP) fractures in the setting of topical, intralesional, intramuscular, and systemic GC treatment, as well as recommendations for nutritional supplementation to reduce these risks.
Pathophysiology
The pathophysiology of GIO is multifactorial and occurs in both early and late phases.9,10 The early phase is characterized by rapid BMD reduction due to excessive bone resorption. The late phase is characterized by slower and more progressive BMD reduction due to impaired bone formation.9 At the osteocyte level, GCs decrease cell viability and induce apoptosis.11 At the osteoblast level, GCs impair cell replication and differentiation and have proapoptotic effects, resulting in decreased cell numbers and subsequent bone formation.10 At the osteoclast level, GCs increase expression of pro-osteoclastic cytokines and decrease mature osteoclast apoptosis, resulting in an expanded osteoclastic life span and prolonged bone resorption.12,13 Indirectly, GCs alter calcium metabolism by decreasing gastrointestinal calcium absorption and impairing renal absorption.14,15
GCs and Osteoporosis
Oral GCs—Glucocorticoid-induced osteoporosis and fracture risk are dose and duration dependent.6 A study of 244,235 patients taking GCs and 244,235 controls found the relative risk of vertebral fracture was 1.55 (range, 1.20–2.01) for daily prednisone use at less than 2.5 mg, 2.59 (range, 2.16–3.10) for daily prednisone use from 2.5 to 7.4 mg, and 5.18 (range, 4.25–6.31) for daily doses of 7.5 mg or higher; the relative risk for hip fractures was 0.99 (range, 0.82–1.20), 1.77 (range, 1.55–2.02), and 2.27 (range, 1.94–2.66), respectively.16 Another large retrospective cohort study found that continuous treatment with prednisone 10 mg/d for more than 90 days compared to no GC exposure increased the risk for hip fractures 7-fold and 17-fold for vertebral fractures.17 Although the minimum cumulative dose of GCs known to cause osteoporosis is not clearly established, the American College of Rheumatology has proposed an algorithm as a basic approach to anticipate, prevent, and treat GIO (Figure).18,19 Fracture risk should be assessed in all patients who are prescribed prednisone 2.5 mg/d for 3 months or longer or an anticipated cumulative dose of more than 1 g per year. Patients 40 years and older with anticipated GC use of 3 months or longer should have both a bone densitometry scan and a Fracture Risk Assessment (FRAX) score. The FRAX tool estimates the 10-year probability of fracture in patients aged 40 to 80 years, and those patients can be further risk stratified as low (FRAX <10%), moderate (FRAX 10%–19%), or high (FRAX ≥20%) risk. In patients with moderate to high risk of fracture (FRAX >10%), initiation of pharmacologic treatment or referral to a metabolic bone specialist should be considered.18,19 First-line therapy is an oral bisphosphonate, and second-line therapies include intravenous bisphosphonates, teriparatide, denosumab, or raloxifene for patients at high risk for GIO.19 Adults younger than 40 years with a history of OP fracture or considerable risk factors for OP fractures should have a bone densitometry scan, and, if results are abnormal, the patient should be referred to a metabolic bone specialist. Those with low fracture risk based on bone densitometry and FRAX and those with no risk factors should be assessed annually for bone health (additional risk factors, GC dose and duration, bone densitometry/FRAX if indicated).18 In addition to GC dose and duration, additional risk factors for GIO, which are factored into the FRAX tool, include advanced age, low body mass index, history of bone fracture, smoking, excessive alcohol use (≥3 drinks/d), history of falls, low BMD, family history of bone fracture, and hypovitaminosis D.6

Topical GCs—Although there is strong evidence and clear guidelines regarding oral GIO, there is a dearth of data surrounding OP risk due to treatment with topical GCs. A recent retrospective nationwide Danish study evaluating the risk of osteoporosis and major OP fracture in 723,251 adults treated with potent or very potent topical steroids sought to evaluate these risks.20 Patients were included if they had filled prescriptions of at least 500 g of topical mometasone or an equivalent alternative. The investigators reported a 3% increase in relative risk of osteoporosis and major OP fracture with doubling of the cumulative topical GC dose (hazard ratio [HR], 1.03 [95% CI, 1.02-1.04] for both). The overall population-attributable risk was 4.3% (95% CI, 2.7%-5.8%) for osteoporosis and 2.7% (95% CI, 1.7%-3.8%) for major OP fracture. Notably, at least 10,000 g of mometasone was required for 1 additional patient to have a major OP fracture.20 In a commentary based on this study, Jackson21 noted that the number of patient-years of topical GC use needed for 1 fracture was 4-fold higher than that for high-dose oral GCs (40 mg/d prednisolone for ≥30 days). Another study assessed the effects of topical GCs on BMD in adults with moderate to severe atopic dermatitis over a 2-year period.22 No significant difference in BMD assessed via bone densitometry of either the lumbar spine or total hip at baseline or at 2-year follow-up was reported for either group treated with corticosteroids (<75 g per month or ≥75 g per month). Of note, the authors did not account for steroid potency, which ranged from class 1 through class 4.22 Although limited data exist, these studies suggest topical GCs used at conventional doses with appropriate breaks in therapy will not substantially increase risk for GIO or OP fracture; however, in the small subset of patients requiring chronic use of superpotent topical corticosteroids with other OP risk factors, transitioning to non–GC-based therapy or initiating bone health therapy may be advised to improve patient outcomes. Risk assessment, as in cases of chronic topical GC use, may be beneficial.
Intralesional GCs—Intralesional GCs are indicated for numerous inflammatory conditions including alopecia areata, discoid lupus erythematosus, keloids, and granuloma annulare. It generally is accepted that doses of triamcinolone acetonide should not exceed 20 mg per session spaced at least 3 weeks apart or up to 40 mg per month.18 One study demonstrated that doses of triamcinolone diacetate of 25 mg or less were unlikely to produce systemic effects and were determined to be a safe dose for intralesional injections.23 A retrospective cross-sectional case series including 18 patients with alopecia areata reported decreased BMD in 9 patients receiving intralesional triamcinolone acetonide 10 mg/mL at 4- to 8-week intervals for at least 20 months, with cumulative doses greater than 500 mg. This was particularly notable in postmenopausal women and men older than 50 years; participants with a body mass index less than 18.5 kg/m2, history of a stress fracture, family history of osteopenia or osteoporosis, and history of smoking; and those who did not regularly engage in weight-bearing exercises.24 Patients receiving long-term (ie, >1 year) intralesional steroids should be evaluated for osteoporosis risk and preventative strategies should be considered (ie, regular weight-bearing exercises, calcium and vitamin D supplementation, bisphosphate therapy). As with topical GCs, there are no clear guidelines for risk assessment or treatment recommendations for GIO.
Intramuscular GCs—The data regarding intramuscular (IM) GCs and dermatologic disease is severely limited, and to the best of our knowledge, no studies specifically assess the risk for GIO or fracture secondary to intramuscular GCs; however, a retrospective study of 27 patients (4 female, 23 male; mean age, 33 years [range, 12–61 years]) with refractory alopecia areata receiving IM triamcinolone acetonide (40 mg every 4 weeks for 3–6 months) reported 1 patient (a 56-year-old woman) with notably decreased bone densitometry from baseline requiring treatment discontinuation.25 No other patients at risk for osteoporosis had decreased BMD from treatment with IM triamcinolone; however, it was noted that 1 month following treatment, 10 of 11 assessed patients demonstrated decreased levels of morning serum cortisol and plasma adrenocorticotropic hormone—despite baseline levels within reference range—that resolved 3 months after treatment completion,25 which suggests a prolonged release of IM triamcinolone and sustained systemic effect. One systematic review of 342 patients with dermatologic diseases treated with IM corticosteroids found the primary side effects included dysmenorrhea, injection-site lipoatrophy, and adrenocortical suppression, with only a single reported case of low BMD.26 Given the paucity of evidence, additional studies are required to assess the effect of IM triamcinolone on BMD and risk for major OP fractures with regard to dosing and frequency. As there are no clear guidelines for osteoporosis evaluation in the setting of intramuscular GCs, it may be prudent to follow the algorithmic model recommended for oral steroids when anticipating at least 3 months of intramuscular GCs.
Diet and Prevention of Bone Loss
Given the profound impact that systemic GCs have on osteoporosis and fracture risk and the sparse data regarding risk from topical, intralesional, or intramuscular GCs, diet and nutrition represent a simple, safe, and potentially preventative method of slowing BMD loss and minimizing fracture risk. In higher-risk patients, nutritional assessment in combination with medical therapy also is likely warranted.
Calcium and Vitamin D3—Patients treated with any GC dose longer than 3 months should undergo calcium and vitamin D optimization.19 Exceptions for supplementation include certain patients with sarcoidosis, which can be associated with high vitamin D levels; patients with a history of hypercalcemia or hypercalciuria; and patients with chronic kidney disease.6 In a meta-analysis including 30,970 patients in 8 randomized controlled trials, calcium (500–1200 mg/d) and vitamin D (400–800 IU/d) supplementation reduced the risk of total fractures by 15% (summary relative risk estimate, 0.85 [95% CI, 0.73-0.98]) and hip fractures by 30% (summary relative risk estimate, 0.70 [95% CI, 0.56-0.87]).4 One double-blind, placebo-controlled clinical trial conducted by the Women’s Health Initiative that included 36,282 postmenopausal women who were taking 1000 mg of calcium and 400 IU of vitamin D3 daily for more than 5 years reported an HR of 0.62 (95% CI, 0.38-1.00) for hip fracture for supplementation vs placebo.27 Lastly, a 2016 Cochrane Review including 12 randomized trials and 1343 participants reported a 43% lower risk of new vertebral fractures following supplementation with calcium, vitamin D, or both compared with controls.28
Specific recommendations for calcium and vitamin D3 supplementation vary based on age and sex. The US Preventive Services Task Force concluded that insufficient evidence exists to support calcium and vitamin D3 supplementation in asymptomatic men and premenopausal women.29 The National Osteoporosis Foundation (NOF) supports the use of calcium supplementation for fracture risk reduction in middle-aged and older adults.4 Furthermore, the NOF supports the Institute of Medicine recommendations31 that men aged 50 to 70 years consume 1000 mg/d of calcium and that women 51 years and older as well as men 71 years and older consume 1200 mg/d of calcium.30 The NOF recommends 800 to 1000 IU/d of vitamin D in adults 50 years and older, while the Institute of Medicine recommends 600 IU/d in adults 70 years and younger and 800 IU/d in adults 71 years and older.31 These recommendations are similar to both the Endocrine Society and the American Geriatric Society.32,33 Total calcium should not exceed 2000 mg/d due to risk of adverse effects.
Dietary sources of vitamin D include fatty fish, mushrooms, and fortified dairy products, though recommended doses rarely can be achieved through diet alone.34 Dairy products are the primary source of dietary calcium. Other high-calcium foods include green leafy vegetables, nuts and seeds, soft-boned fish, and fortified beverages and cereals.35
Probiotics—A growing body of evidence suggests that probiotics may be beneficial in promoting bone health by improving calcium homeostasis, reducing risk for hyperparathyroidism secondary to GC therapy, and decreasing age-related bone resorption.36 An animal study demonstrated that probiotics can regulate bone resorption and formation as well as reduce bone loss secondary to GC therapy.37 A randomized, double-blind, placebo-controlled, multicenter trial randomly assigned 249 healthy, early postmenopausal women to receive probiotic treatment containing 3 lactobacillus strains (Lactobacillus paracasei DSM 13434, Lactobacillus plantarum DSM 15312, and L plantarum DSM 15313) or placebo once daily for 12 months.38 Bone mineral density was measured at baseline and at 12 months. Of the 234 participants who completed the study, lactobacillus treatment reduced lumbosacral BMD loss compared to the placebo group (mean difference, 0.71% [95% CI, 0.06-1.35]). They also reported significant lumbosacral BMD loss in the placebo group (−0.72% [95% CI, −1.22 to −0.22]) compared to no BMD loss in the group treated with lactobacillus (−0.01% [95% CI, −0.50 to 0.48]).38 Although the data may be encouraging, more studies are needed to determine if probiotics should be regarded as an adjuvant treatment to calcium, vitamin D, and pharmacologic therapy for long-term prevention of bone loss in the setting of GIO.39 Because existing studies on probiotics include varying compositions and doses, larger studies with consistent supplementation are required. Encouraging probiotic intake through fermented dairy products may represent a simple low-risk intervention to support bone health.
Anti-inflammatory Diet—The traditional Mediterranean diet is rich in fruits, vegetables, fish, nuts, whole grains, legumes, and monounsaturated fats and low in meat and dairy products. The Mediterranean diet has been shown to be modestly protective against osteoporosis and fracture risk. A large US observational study including 93,676 women showed that those with the highest quintile of the alternate Mediterranean diet score had a lower risk for hip fracture (HR, 0.80 [95% CI, 0.66-0.97]), with an absolute risk reduction of 0.29% and number needed to treat at 342.40 A multicenter study involving adults from 8 European countries found that increased adherence to the Mediterranean diet was associated with a 7% reduction in hip fracture incidence (HR per 1 unit increase in Mediterranean diet, 0.93 [95% CI, 0.89-0.98]). High vegetable and fruit intake was associated with decreased hip fracture incidence (HR, 0.86 and 0.89 [95% CI, 0.79-0.94 and 0.82-0.97, respectively]), and high meat and excessive ethanol consumption were associated with increased fracture incidence (HR, 1.18 and 1.74 [95% CI, 1.06-1.31 and 1.32-2.31, respectively]).41 Similarly, a large observational study in Sweden that included 37,903 men and 33,403 women reported similar findings, noting a 6% lower hip fracture rate per one unit increase in alternate Mediterranean diet score (adjusted HR, 0.94 [95% CI, 0.92-0.96]).42 This is thought to be due in part to higher levels of dietary vitamin D present in many foods traditionally included in the Mediterranean diet.43 Additionally, olive oil, a staple in the Mediterranean diet, appears to reduce bone loss by promoting osteoblast proliferation and maturation, inhibiting bone resorption, suppressing oxidative stress and inflammation, and increasing calcium deposition in the extracellular matrix.44,45 Fruits, vegetables, legumes, and nuts also are rich in minerals including potassium and magnesium, which are important in bone health to promote osteoblast proliferation and vitamin D activation.36,46-48
Final Thoughts
Osteoporosis-related fractures are common and are associated with high morbidity and health care costs. Dermatologists using and prescribing corticosteroids must be aware of the risk for GIO, particularly in patients with a pre-existing diagnosis of osteopenia or osteoporosis. There likely is no oral corticosteroid dose that does not increase a patient’s risk for osteoporosis; therefore, oral GCs should be used at the lowest effective daily dose for the shortest duration possible. Patients with an anticipated duration of at least 3 months—regardless of dose—should be assessed for their risk for GIO. Patients using topical and intralesional corticosteroids are unlikely to develop GIO; however, those with risk factors and a considerable cumulative dose may warrant further evaluation. In all cases, we advocate for supplementing with calcium and vitamin D as well as promoting probiotic intake and the Mediterranean diet. Those at moderate to high risk for fracture may require additional medical therapy. Dermatologists are uniquely positioned to identify this at-risk population, and because osteoporosis is a chronic illness, primary care providers should be notified of prolonged GC therapy to help with risk assessment, initiation of vitamin and mineral supplementation, and follow-up with metabolic bone health specialists. Through a multidisciplinary approach and patient education, GIO and the potential risk for fracture can be successfully mitigated in most patients.
Glucocorticoids (GCs) are among the most widely prescribed medications in dermatologic practice. Although GCs are highly effective anti-inflammatory agents, long-term systemic therapy can result in dangerous adverse effects, including GC-induced osteoporosis (GIO), a bone disease associated with a heightened risk for fragility fractures.1,2 In the United States, an estimated 10.2 million adults have osteoporosis—defined as a T-score lower than −2.5 measured via a bone densitometry scan—and 43.4 million adults have low bone mineral density (BMD).3,4 The prevalence of osteoporosis is increasing, and the diagnosis is more common in females and adults 55 years and older.2 More than 2 million individuals have osteoporosis-related fractures annually, and the mortality risk is increased at 5 and 10 years following low-energy osteoporosis-related fractures.3-5
Glucocorticoid therapy is the leading iatrogenic cause of secondary osteoporosis. As many as 30% of all patients treated with systemic GCs for more than 6 months develop GIO.1,6,7 Glucocorticoid-induced BMD loss occurs at a rate of 6% to 12% of total BMD during the first year, slowing to approximately 3% per year during subsequent therapy.1 The risk for insufficiency fractures increases by as much as 75% from baseline in adults with rheumatic, pulmonary, and skin disorders within the first 3 months of therapy and peaks at approximately 12 months.1,2
Despite the risks, many long-term GC users never receive therapy to prevent bone loss; others are only started on therapy once they have sustained an insufficiency fracture. A 5-year international observational study including more than 40,000 postmenopausal women found that only 51% of patients who were on continuous GC therapy were undergoing BMD testing and appropriate medical management.8 This review highlights the existing evidence on the risks of osteoporosis and osteoporotic (OP) fractures in the setting of topical, intralesional, intramuscular, and systemic GC treatment, as well as recommendations for nutritional supplementation to reduce these risks.
Pathophysiology
The pathophysiology of GIO is multifactorial and occurs in both early and late phases.9,10 The early phase is characterized by rapid BMD reduction due to excessive bone resorption. The late phase is characterized by slower and more progressive BMD reduction due to impaired bone formation.9 At the osteocyte level, GCs decrease cell viability and induce apoptosis.11 At the osteoblast level, GCs impair cell replication and differentiation and have proapoptotic effects, resulting in decreased cell numbers and subsequent bone formation.10 At the osteoclast level, GCs increase expression of pro-osteoclastic cytokines and decrease mature osteoclast apoptosis, resulting in an expanded osteoclastic life span and prolonged bone resorption.12,13 Indirectly, GCs alter calcium metabolism by decreasing gastrointestinal calcium absorption and impairing renal absorption.14,15
GCs and Osteoporosis
Oral GCs—Glucocorticoid-induced osteoporosis and fracture risk are dose and duration dependent.6 A study of 244,235 patients taking GCs and 244,235 controls found the relative risk of vertebral fracture was 1.55 (range, 1.20–2.01) for daily prednisone use at less than 2.5 mg, 2.59 (range, 2.16–3.10) for daily prednisone use from 2.5 to 7.4 mg, and 5.18 (range, 4.25–6.31) for daily doses of 7.5 mg or higher; the relative risk for hip fractures was 0.99 (range, 0.82–1.20), 1.77 (range, 1.55–2.02), and 2.27 (range, 1.94–2.66), respectively.16 Another large retrospective cohort study found that continuous treatment with prednisone 10 mg/d for more than 90 days compared to no GC exposure increased the risk for hip fractures 7-fold and 17-fold for vertebral fractures.17 Although the minimum cumulative dose of GCs known to cause osteoporosis is not clearly established, the American College of Rheumatology has proposed an algorithm as a basic approach to anticipate, prevent, and treat GIO (Figure).18,19 Fracture risk should be assessed in all patients who are prescribed prednisone 2.5 mg/d for 3 months or longer or an anticipated cumulative dose of more than 1 g per year. Patients 40 years and older with anticipated GC use of 3 months or longer should have both a bone densitometry scan and a Fracture Risk Assessment (FRAX) score. The FRAX tool estimates the 10-year probability of fracture in patients aged 40 to 80 years, and those patients can be further risk stratified as low (FRAX <10%), moderate (FRAX 10%–19%), or high (FRAX ≥20%) risk. In patients with moderate to high risk of fracture (FRAX >10%), initiation of pharmacologic treatment or referral to a metabolic bone specialist should be considered.18,19 First-line therapy is an oral bisphosphonate, and second-line therapies include intravenous bisphosphonates, teriparatide, denosumab, or raloxifene for patients at high risk for GIO.19 Adults younger than 40 years with a history of OP fracture or considerable risk factors for OP fractures should have a bone densitometry scan, and, if results are abnormal, the patient should be referred to a metabolic bone specialist. Those with low fracture risk based on bone densitometry and FRAX and those with no risk factors should be assessed annually for bone health (additional risk factors, GC dose and duration, bone densitometry/FRAX if indicated).18 In addition to GC dose and duration, additional risk factors for GIO, which are factored into the FRAX tool, include advanced age, low body mass index, history of bone fracture, smoking, excessive alcohol use (≥3 drinks/d), history of falls, low BMD, family history of bone fracture, and hypovitaminosis D.6

Topical GCs—Although there is strong evidence and clear guidelines regarding oral GIO, there is a dearth of data surrounding OP risk due to treatment with topical GCs. A recent retrospective nationwide Danish study evaluating the risk of osteoporosis and major OP fracture in 723,251 adults treated with potent or very potent topical steroids sought to evaluate these risks.20 Patients were included if they had filled prescriptions of at least 500 g of topical mometasone or an equivalent alternative. The investigators reported a 3% increase in relative risk of osteoporosis and major OP fracture with doubling of the cumulative topical GC dose (hazard ratio [HR], 1.03 [95% CI, 1.02-1.04] for both). The overall population-attributable risk was 4.3% (95% CI, 2.7%-5.8%) for osteoporosis and 2.7% (95% CI, 1.7%-3.8%) for major OP fracture. Notably, at least 10,000 g of mometasone was required for 1 additional patient to have a major OP fracture.20 In a commentary based on this study, Jackson21 noted that the number of patient-years of topical GC use needed for 1 fracture was 4-fold higher than that for high-dose oral GCs (40 mg/d prednisolone for ≥30 days). Another study assessed the effects of topical GCs on BMD in adults with moderate to severe atopic dermatitis over a 2-year period.22 No significant difference in BMD assessed via bone densitometry of either the lumbar spine or total hip at baseline or at 2-year follow-up was reported for either group treated with corticosteroids (<75 g per month or ≥75 g per month). Of note, the authors did not account for steroid potency, which ranged from class 1 through class 4.22 Although limited data exist, these studies suggest topical GCs used at conventional doses with appropriate breaks in therapy will not substantially increase risk for GIO or OP fracture; however, in the small subset of patients requiring chronic use of superpotent topical corticosteroids with other OP risk factors, transitioning to non–GC-based therapy or initiating bone health therapy may be advised to improve patient outcomes. Risk assessment, as in cases of chronic topical GC use, may be beneficial.
Intralesional GCs—Intralesional GCs are indicated for numerous inflammatory conditions including alopecia areata, discoid lupus erythematosus, keloids, and granuloma annulare. It generally is accepted that doses of triamcinolone acetonide should not exceed 20 mg per session spaced at least 3 weeks apart or up to 40 mg per month.18 One study demonstrated that doses of triamcinolone diacetate of 25 mg or less were unlikely to produce systemic effects and were determined to be a safe dose for intralesional injections.23 A retrospective cross-sectional case series including 18 patients with alopecia areata reported decreased BMD in 9 patients receiving intralesional triamcinolone acetonide 10 mg/mL at 4- to 8-week intervals for at least 20 months, with cumulative doses greater than 500 mg. This was particularly notable in postmenopausal women and men older than 50 years; participants with a body mass index less than 18.5 kg/m2, history of a stress fracture, family history of osteopenia or osteoporosis, and history of smoking; and those who did not regularly engage in weight-bearing exercises.24 Patients receiving long-term (ie, >1 year) intralesional steroids should be evaluated for osteoporosis risk and preventative strategies should be considered (ie, regular weight-bearing exercises, calcium and vitamin D supplementation, bisphosphate therapy). As with topical GCs, there are no clear guidelines for risk assessment or treatment recommendations for GIO.
Intramuscular GCs—The data regarding intramuscular (IM) GCs and dermatologic disease is severely limited, and to the best of our knowledge, no studies specifically assess the risk for GIO or fracture secondary to intramuscular GCs; however, a retrospective study of 27 patients (4 female, 23 male; mean age, 33 years [range, 12–61 years]) with refractory alopecia areata receiving IM triamcinolone acetonide (40 mg every 4 weeks for 3–6 months) reported 1 patient (a 56-year-old woman) with notably decreased bone densitometry from baseline requiring treatment discontinuation.25 No other patients at risk for osteoporosis had decreased BMD from treatment with IM triamcinolone; however, it was noted that 1 month following treatment, 10 of 11 assessed patients demonstrated decreased levels of morning serum cortisol and plasma adrenocorticotropic hormone—despite baseline levels within reference range—that resolved 3 months after treatment completion,25 which suggests a prolonged release of IM triamcinolone and sustained systemic effect. One systematic review of 342 patients with dermatologic diseases treated with IM corticosteroids found the primary side effects included dysmenorrhea, injection-site lipoatrophy, and adrenocortical suppression, with only a single reported case of low BMD.26 Given the paucity of evidence, additional studies are required to assess the effect of IM triamcinolone on BMD and risk for major OP fractures with regard to dosing and frequency. As there are no clear guidelines for osteoporosis evaluation in the setting of intramuscular GCs, it may be prudent to follow the algorithmic model recommended for oral steroids when anticipating at least 3 months of intramuscular GCs.
Diet and Prevention of Bone Loss
Given the profound impact that systemic GCs have on osteoporosis and fracture risk and the sparse data regarding risk from topical, intralesional, or intramuscular GCs, diet and nutrition represent a simple, safe, and potentially preventative method of slowing BMD loss and minimizing fracture risk. In higher-risk patients, nutritional assessment in combination with medical therapy also is likely warranted.
Calcium and Vitamin D3—Patients treated with any GC dose longer than 3 months should undergo calcium and vitamin D optimization.19 Exceptions for supplementation include certain patients with sarcoidosis, which can be associated with high vitamin D levels; patients with a history of hypercalcemia or hypercalciuria; and patients with chronic kidney disease.6 In a meta-analysis including 30,970 patients in 8 randomized controlled trials, calcium (500–1200 mg/d) and vitamin D (400–800 IU/d) supplementation reduced the risk of total fractures by 15% (summary relative risk estimate, 0.85 [95% CI, 0.73-0.98]) and hip fractures by 30% (summary relative risk estimate, 0.70 [95% CI, 0.56-0.87]).4 One double-blind, placebo-controlled clinical trial conducted by the Women’s Health Initiative that included 36,282 postmenopausal women who were taking 1000 mg of calcium and 400 IU of vitamin D3 daily for more than 5 years reported an HR of 0.62 (95% CI, 0.38-1.00) for hip fracture for supplementation vs placebo.27 Lastly, a 2016 Cochrane Review including 12 randomized trials and 1343 participants reported a 43% lower risk of new vertebral fractures following supplementation with calcium, vitamin D, or both compared with controls.28
Specific recommendations for calcium and vitamin D3 supplementation vary based on age and sex. The US Preventive Services Task Force concluded that insufficient evidence exists to support calcium and vitamin D3 supplementation in asymptomatic men and premenopausal women.29 The National Osteoporosis Foundation (NOF) supports the use of calcium supplementation for fracture risk reduction in middle-aged and older adults.4 Furthermore, the NOF supports the Institute of Medicine recommendations31 that men aged 50 to 70 years consume 1000 mg/d of calcium and that women 51 years and older as well as men 71 years and older consume 1200 mg/d of calcium.30 The NOF recommends 800 to 1000 IU/d of vitamin D in adults 50 years and older, while the Institute of Medicine recommends 600 IU/d in adults 70 years and younger and 800 IU/d in adults 71 years and older.31 These recommendations are similar to both the Endocrine Society and the American Geriatric Society.32,33 Total calcium should not exceed 2000 mg/d due to risk of adverse effects.
Dietary sources of vitamin D include fatty fish, mushrooms, and fortified dairy products, though recommended doses rarely can be achieved through diet alone.34 Dairy products are the primary source of dietary calcium. Other high-calcium foods include green leafy vegetables, nuts and seeds, soft-boned fish, and fortified beverages and cereals.35
Probiotics—A growing body of evidence suggests that probiotics may be beneficial in promoting bone health by improving calcium homeostasis, reducing risk for hyperparathyroidism secondary to GC therapy, and decreasing age-related bone resorption.36 An animal study demonstrated that probiotics can regulate bone resorption and formation as well as reduce bone loss secondary to GC therapy.37 A randomized, double-blind, placebo-controlled, multicenter trial randomly assigned 249 healthy, early postmenopausal women to receive probiotic treatment containing 3 lactobacillus strains (Lactobacillus paracasei DSM 13434, Lactobacillus plantarum DSM 15312, and L plantarum DSM 15313) or placebo once daily for 12 months.38 Bone mineral density was measured at baseline and at 12 months. Of the 234 participants who completed the study, lactobacillus treatment reduced lumbosacral BMD loss compared to the placebo group (mean difference, 0.71% [95% CI, 0.06-1.35]). They also reported significant lumbosacral BMD loss in the placebo group (−0.72% [95% CI, −1.22 to −0.22]) compared to no BMD loss in the group treated with lactobacillus (−0.01% [95% CI, −0.50 to 0.48]).38 Although the data may be encouraging, more studies are needed to determine if probiotics should be regarded as an adjuvant treatment to calcium, vitamin D, and pharmacologic therapy for long-term prevention of bone loss in the setting of GIO.39 Because existing studies on probiotics include varying compositions and doses, larger studies with consistent supplementation are required. Encouraging probiotic intake through fermented dairy products may represent a simple low-risk intervention to support bone health.
Anti-inflammatory Diet—The traditional Mediterranean diet is rich in fruits, vegetables, fish, nuts, whole grains, legumes, and monounsaturated fats and low in meat and dairy products. The Mediterranean diet has been shown to be modestly protective against osteoporosis and fracture risk. A large US observational study including 93,676 women showed that those with the highest quintile of the alternate Mediterranean diet score had a lower risk for hip fracture (HR, 0.80 [95% CI, 0.66-0.97]), with an absolute risk reduction of 0.29% and number needed to treat at 342.40 A multicenter study involving adults from 8 European countries found that increased adherence to the Mediterranean diet was associated with a 7% reduction in hip fracture incidence (HR per 1 unit increase in Mediterranean diet, 0.93 [95% CI, 0.89-0.98]). High vegetable and fruit intake was associated with decreased hip fracture incidence (HR, 0.86 and 0.89 [95% CI, 0.79-0.94 and 0.82-0.97, respectively]), and high meat and excessive ethanol consumption were associated with increased fracture incidence (HR, 1.18 and 1.74 [95% CI, 1.06-1.31 and 1.32-2.31, respectively]).41 Similarly, a large observational study in Sweden that included 37,903 men and 33,403 women reported similar findings, noting a 6% lower hip fracture rate per one unit increase in alternate Mediterranean diet score (adjusted HR, 0.94 [95% CI, 0.92-0.96]).42 This is thought to be due in part to higher levels of dietary vitamin D present in many foods traditionally included in the Mediterranean diet.43 Additionally, olive oil, a staple in the Mediterranean diet, appears to reduce bone loss by promoting osteoblast proliferation and maturation, inhibiting bone resorption, suppressing oxidative stress and inflammation, and increasing calcium deposition in the extracellular matrix.44,45 Fruits, vegetables, legumes, and nuts also are rich in minerals including potassium and magnesium, which are important in bone health to promote osteoblast proliferation and vitamin D activation.36,46-48
Final Thoughts
Osteoporosis-related fractures are common and are associated with high morbidity and health care costs. Dermatologists using and prescribing corticosteroids must be aware of the risk for GIO, particularly in patients with a pre-existing diagnosis of osteopenia or osteoporosis. There likely is no oral corticosteroid dose that does not increase a patient’s risk for osteoporosis; therefore, oral GCs should be used at the lowest effective daily dose for the shortest duration possible. Patients with an anticipated duration of at least 3 months—regardless of dose—should be assessed for their risk for GIO. Patients using topical and intralesional corticosteroids are unlikely to develop GIO; however, those with risk factors and a considerable cumulative dose may warrant further evaluation. In all cases, we advocate for supplementing with calcium and vitamin D as well as promoting probiotic intake and the Mediterranean diet. Those at moderate to high risk for fracture may require additional medical therapy. Dermatologists are uniquely positioned to identify this at-risk population, and because osteoporosis is a chronic illness, primary care providers should be notified of prolonged GC therapy to help with risk assessment, initiation of vitamin and mineral supplementation, and follow-up with metabolic bone health specialists. Through a multidisciplinary approach and patient education, GIO and the potential risk for fracture can be successfully mitigated in most patients.
- Weinstein RS. Clinical practice. glucocorticoid-induced bone disease. N Engl J Med. 2011;365:62-70.
- Buckley L, Humphrey MB. Glucocorticoid-induced osteoporosis. N Engl J Med. 2018;379:2547-2556.
- Wright NC, Looker AC, Saag KG, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29:2520-2526.
- Weaver CM, Alexander DD, Boushey CJ, et al. Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int. 2016;27:367-376.
- Bliuc D, Nguyen ND, Milch VE, et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301:513-521.
- Caplan A, Fett N, Rosenbach M, et al. Prevention and management of glucocorticoid-induced side effects: a comprehensive review: a review of glucocorticoid pharmacology and bone health. J Am Acad Dermatol. 2017;76:1-9.
- Gudbjornsson B, Juliusson UI, Gudjonsson FV. Prevalence of long term steroid treatment and the frequency of decision making to prevent steroid induced osteoporosis in daily clinical practice. Ann Rheum Dis. 2002;61:32-36.
- Silverman S, Curtis J, Saag K, et al. International management of bone health in glucocorticoid-exposed individuals in the observational GLOW study. Osteoporos Int. 2015;26:419-420.
- Canalis E, Bilezikian JP, Angeli A, et al. Perspectives on glucocorticoid-induced osteoporosis. Bone. 2004;34:593-598.
- Canalis E, Mazziotti G, Giustina A, et al. Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int. 2007;18:1319-1328.
- Lane NE, Yao W, Balooch M, et al. Glucocorticoid-treated mice have localized changes in trabecular bone material properties and osteocyte lacunar size that are not observed in placebo-treated or estrogen-deficient mice. J Bone Miner Res. 2006;21:466-476.
- Hofbauer LC, Gori F, Riggs BL, et al. Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblastic lineage cells: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology. 1999;140:4382-4389.
- Jia D, O’Brien CA, Stewart SA, et al. Glucocorticoids act directly on osteoclasts to increase their life span and reduce bone density. Endocrinology. 2006;147:5592-5599.
- Mazziotti G, Angeli A, Bilezikian JP, et al. Glucocorticoid-induced osteoporosis: an update. Trends Endocrinol Metab. 2006;17:144-149.
- Huybers S, Naber TH, Bindels RJ, et al. Prednisolone-induced Ca2+ malabsorption is caused by diminished expression of the epithelial Ca2+ channel TRPV6. Am J Physiol Gastrointest Liver Physiol. 2007;292:G92-G97.
- Van Staa TP, Leufkens HG, Abenhaim L, et al. Use of oral corticosteroids and risk of fractures. J Bone Miner Res. 2000;15:993-1000.
- Steinbuch M, Youket TE, Cohen S. Oral glucocorticoid use is associated with an increased risk of fracture. Osteoporos Int. 2004;15:323-328.
- Lupsa BC, Insogna KL, Micheletti RG, et al. Corticosteroid use in chronic dermatologic disorders and osteoporosis. Int J Womens Dermatol. 2021;7:545-551.
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken). 2017;69:1095-1110.
- Egeberg A, Schwarz P, Harsløf T, et al. Association of potent and very potent topical corticosteroids and the risk of osteoporosis and major osteoporotic fractures. JAMA Dermatol. 2021;157:275-282.
- Jackson RD. Topical corticosteroids and glucocorticoid-induced osteoporosis-cumulative dose and duration matter. JAMA Dermatol. 2021;157:269-270.
- van Velsen SG, Haeck IM, Knol MJ, et al. Two-year assessment of effect of topical corticosteroids on bone mineral density in adults with moderate to severe atopic dermatitis. J Am Acad Dermatol. 2012;66:691-693.
- McGugan AD, Shuster S, Bottoms E. Adrenal suppression from intradermal triamcinolone. J Invest Dermatol. 1963;40:271-272.
- Samrao A, Fu JM, Harris ST, et al. Bone mineral density in patients with alopecia areata treated with long-term intralesional corticosteroids. J Drugs Dermatol. 2013;12:E36-E40.
- Seo J, Lee YI, Hwang S, et al. Intramuscular triamcinolone acetonide: an undervalued option for refractory alopecia areata. J Dermatol. 2017;44:173-179.
- Thomas LW, Elsensohn A, Bergheim T, et al. Intramuscular steroids in the treatment of dermatologic disease: a systematic review. J Drugs Dermatol. 2018;17:323-329.
- Prentice RL, Pettinger MB, Jackson RD, et al. Health risks and benefits from calcium and vitamin D supplementation: Women’s Health Initiative clinical trial and cohort study. Osteoporos Int. 2013;24:567-580.
- Allen CS, Yeung JH, Vandermeer B, et al. Bisphosphonates for steroid-induced osteoporosis. Cochrane Database Syst Rev. 2016;10:CD001347. doi:10.1002/14651858.CD001347.pub2
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, et al. Vitamin D, calcium, or combined supplementation for the primary prevention of fractures in community-dwelling adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319:1592-1599.
- Cosman F, de Beur SJ, LeBoff MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359-2381.
- Institute of Medicine. Dietary reference intakes for calcium and vitamin D. Washington, DC: National Academies Press; 2011.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911-1930.
- American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults. Recommendations abstracted from the American Geriatrics Society Consensus Statement on vitamin D for prevention of falls and their consequences. J Am Geriatr Soc. 2014;62:147-152.
- Vitamin D fact sheet for health professionals. National Institutes of Health Office of Dietary Supplements website. Updated August 12, 2022. Accessed September 16, 2022. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
- Calcium fact sheet for health professionals. National Institutes of Health Office of Dietary Supplements website. Updated June 2, 2022. Accessed September 16, 2022. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/
- Muñoz-Garach A, García-Fontana B, Muñoz-Torres M. Nutrients and dietary patterns related to osteoporosis. Nutrients. 2020;12:1986.
- Schepper JD, Collins F, Rios-Arce ND, et al. Involvement of the gut microbiota and barrier function in glucocorticoid-induced osteoporosis. J Bone Miner Res. 2020;35:801-820.
- Jansson PA, Curiac D, Ahrén IL, et al. Probiotic treatment using a mix of three Lactobacillus strains for lumbar spine bone loss in postmenopausal women: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet Rheumatol. 2019;1:E154-E162.
- Rizzoli R, Biver E. Are probiotics the new calcium and vitamin D for bone health? Curr Osteoporos Rep. 2020;18:273-284.
- Haring B, Crandall CJ, Wu C, et al. Dietary patterns and fractures in postmenopausal women: results from the Women’s Health Initiative. JAMA Intern Med. 2016;176:645-652.
- Benetou V, Orfanos P, Pettersson-Kymmer U, et al. Mediterranean diet and incidence of hip fractures in a European cohort. Osteoporos Int. 2013;24:1587-1598.
- Byberg L, Bellavia A, Larsson SC, et al. Mediterranean diet and hip fracture in Swedish men and women. J Bone Miner Res. 2016;31:2098-2105.
- Zupo R, Lampignano L, Lattanzio A, et al. Association between adherence to the Mediterranean diet and circulating vitamin D levels. Int J Food Sci Nutr. 2020;71:884-890.
- Chin KY, Ima-Nirwana S. Olives and bone: a green osteoporosis prevention option. Int J Environ Res Public Health. 2016;13:755.
- García-Martínez O, Rivas A, Ramos-Torrecillas J, et al. The effect of olive oil on osteoporosis prevention. Int J Food Sci Nutr. 2014;65:834-840.
- Uwitonze AM, Razzaque MS. Role of magnesium in vitamin D activation and function. J Am Osteopath Assoc. 2018;118:181-189.
- Veronese N, Stubbs B, Solmi M, et al. Dietary magnesium intake and fracture risk: data from a large prospective study. Br J Nutr. 2017;117:1570-1576.
- Kong SH, Kim JH, Hong AR, et al. Dietary potassium intake is beneficial to bone health in a low calcium intake population: the Korean National Health and Nutrition Examination Survey (KNHANES)(2008-2011). Osteoporos Int. 2017;28:1577-1585.
- Weinstein RS. Clinical practice. glucocorticoid-induced bone disease. N Engl J Med. 2011;365:62-70.
- Buckley L, Humphrey MB. Glucocorticoid-induced osteoporosis. N Engl J Med. 2018;379:2547-2556.
- Wright NC, Looker AC, Saag KG, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29:2520-2526.
- Weaver CM, Alexander DD, Boushey CJ, et al. Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int. 2016;27:367-376.
- Bliuc D, Nguyen ND, Milch VE, et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301:513-521.
- Caplan A, Fett N, Rosenbach M, et al. Prevention and management of glucocorticoid-induced side effects: a comprehensive review: a review of glucocorticoid pharmacology and bone health. J Am Acad Dermatol. 2017;76:1-9.
- Gudbjornsson B, Juliusson UI, Gudjonsson FV. Prevalence of long term steroid treatment and the frequency of decision making to prevent steroid induced osteoporosis in daily clinical practice. Ann Rheum Dis. 2002;61:32-36.
- Silverman S, Curtis J, Saag K, et al. International management of bone health in glucocorticoid-exposed individuals in the observational GLOW study. Osteoporos Int. 2015;26:419-420.
- Canalis E, Bilezikian JP, Angeli A, et al. Perspectives on glucocorticoid-induced osteoporosis. Bone. 2004;34:593-598.
- Canalis E, Mazziotti G, Giustina A, et al. Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int. 2007;18:1319-1328.
- Lane NE, Yao W, Balooch M, et al. Glucocorticoid-treated mice have localized changes in trabecular bone material properties and osteocyte lacunar size that are not observed in placebo-treated or estrogen-deficient mice. J Bone Miner Res. 2006;21:466-476.
- Hofbauer LC, Gori F, Riggs BL, et al. Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblastic lineage cells: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology. 1999;140:4382-4389.
- Jia D, O’Brien CA, Stewart SA, et al. Glucocorticoids act directly on osteoclasts to increase their life span and reduce bone density. Endocrinology. 2006;147:5592-5599.
- Mazziotti G, Angeli A, Bilezikian JP, et al. Glucocorticoid-induced osteoporosis: an update. Trends Endocrinol Metab. 2006;17:144-149.
- Huybers S, Naber TH, Bindels RJ, et al. Prednisolone-induced Ca2+ malabsorption is caused by diminished expression of the epithelial Ca2+ channel TRPV6. Am J Physiol Gastrointest Liver Physiol. 2007;292:G92-G97.
- Van Staa TP, Leufkens HG, Abenhaim L, et al. Use of oral corticosteroids and risk of fractures. J Bone Miner Res. 2000;15:993-1000.
- Steinbuch M, Youket TE, Cohen S. Oral glucocorticoid use is associated with an increased risk of fracture. Osteoporos Int. 2004;15:323-328.
- Lupsa BC, Insogna KL, Micheletti RG, et al. Corticosteroid use in chronic dermatologic disorders and osteoporosis. Int J Womens Dermatol. 2021;7:545-551.
- Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken). 2017;69:1095-1110.
- Egeberg A, Schwarz P, Harsløf T, et al. Association of potent and very potent topical corticosteroids and the risk of osteoporosis and major osteoporotic fractures. JAMA Dermatol. 2021;157:275-282.
- Jackson RD. Topical corticosteroids and glucocorticoid-induced osteoporosis-cumulative dose and duration matter. JAMA Dermatol. 2021;157:269-270.
- van Velsen SG, Haeck IM, Knol MJ, et al. Two-year assessment of effect of topical corticosteroids on bone mineral density in adults with moderate to severe atopic dermatitis. J Am Acad Dermatol. 2012;66:691-693.
- McGugan AD, Shuster S, Bottoms E. Adrenal suppression from intradermal triamcinolone. J Invest Dermatol. 1963;40:271-272.
- Samrao A, Fu JM, Harris ST, et al. Bone mineral density in patients with alopecia areata treated with long-term intralesional corticosteroids. J Drugs Dermatol. 2013;12:E36-E40.
- Seo J, Lee YI, Hwang S, et al. Intramuscular triamcinolone acetonide: an undervalued option for refractory alopecia areata. J Dermatol. 2017;44:173-179.
- Thomas LW, Elsensohn A, Bergheim T, et al. Intramuscular steroids in the treatment of dermatologic disease: a systematic review. J Drugs Dermatol. 2018;17:323-329.
- Prentice RL, Pettinger MB, Jackson RD, et al. Health risks and benefits from calcium and vitamin D supplementation: Women’s Health Initiative clinical trial and cohort study. Osteoporos Int. 2013;24:567-580.
- Allen CS, Yeung JH, Vandermeer B, et al. Bisphosphonates for steroid-induced osteoporosis. Cochrane Database Syst Rev. 2016;10:CD001347. doi:10.1002/14651858.CD001347.pub2
- US Preventive Services Task Force; Grossman DC, Curry SJ, Owens DK, et al. Vitamin D, calcium, or combined supplementation for the primary prevention of fractures in community-dwelling adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319:1592-1599.
- Cosman F, de Beur SJ, LeBoff MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359-2381.
- Institute of Medicine. Dietary reference intakes for calcium and vitamin D. Washington, DC: National Academies Press; 2011.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911-1930.
- American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults. Recommendations abstracted from the American Geriatrics Society Consensus Statement on vitamin D for prevention of falls and their consequences. J Am Geriatr Soc. 2014;62:147-152.
- Vitamin D fact sheet for health professionals. National Institutes of Health Office of Dietary Supplements website. Updated August 12, 2022. Accessed September 16, 2022. https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/
- Calcium fact sheet for health professionals. National Institutes of Health Office of Dietary Supplements website. Updated June 2, 2022. Accessed September 16, 2022. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/
- Muñoz-Garach A, García-Fontana B, Muñoz-Torres M. Nutrients and dietary patterns related to osteoporosis. Nutrients. 2020;12:1986.
- Schepper JD, Collins F, Rios-Arce ND, et al. Involvement of the gut microbiota and barrier function in glucocorticoid-induced osteoporosis. J Bone Miner Res. 2020;35:801-820.
- Jansson PA, Curiac D, Ahrén IL, et al. Probiotic treatment using a mix of three Lactobacillus strains for lumbar spine bone loss in postmenopausal women: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet Rheumatol. 2019;1:E154-E162.
- Rizzoli R, Biver E. Are probiotics the new calcium and vitamin D for bone health? Curr Osteoporos Rep. 2020;18:273-284.
- Haring B, Crandall CJ, Wu C, et al. Dietary patterns and fractures in postmenopausal women: results from the Women’s Health Initiative. JAMA Intern Med. 2016;176:645-652.
- Benetou V, Orfanos P, Pettersson-Kymmer U, et al. Mediterranean diet and incidence of hip fractures in a European cohort. Osteoporos Int. 2013;24:1587-1598.
- Byberg L, Bellavia A, Larsson SC, et al. Mediterranean diet and hip fracture in Swedish men and women. J Bone Miner Res. 2016;31:2098-2105.
- Zupo R, Lampignano L, Lattanzio A, et al. Association between adherence to the Mediterranean diet and circulating vitamin D levels. Int J Food Sci Nutr. 2020;71:884-890.
- Chin KY, Ima-Nirwana S. Olives and bone: a green osteoporosis prevention option. Int J Environ Res Public Health. 2016;13:755.
- García-Martínez O, Rivas A, Ramos-Torrecillas J, et al. The effect of olive oil on osteoporosis prevention. Int J Food Sci Nutr. 2014;65:834-840.
- Uwitonze AM, Razzaque MS. Role of magnesium in vitamin D activation and function. J Am Osteopath Assoc. 2018;118:181-189.
- Veronese N, Stubbs B, Solmi M, et al. Dietary magnesium intake and fracture risk: data from a large prospective study. Br J Nutr. 2017;117:1570-1576.
- Kong SH, Kim JH, Hong AR, et al. Dietary potassium intake is beneficial to bone health in a low calcium intake population: the Korean National Health and Nutrition Examination Survey (KNHANES)(2008-2011). Osteoporos Int. 2017;28:1577-1585.
Practice Points
- Many long-term glucocorticoid (GC) users never receive therapy to prevent bone loss, and others are only started on therapy once they have sustained an insufficiency fracture.
- Oral GCs should be used at the lowest effective daily dose for the shortest duration possible.
- Patients using topical and intralesional corticosteroids are unlikely to develop GC-induced osteoporosis.
ESMO’s new focus on cancer prevention
This transcript has been edited for clarity.
John Whyte, MD: Welcome, everyone. I’m Dr John Whyte. I’m the chief medical officer at WebMD, and I’m joined today by Fabrice André. He is the chair of the scientific committee at the European Society for Medical Oncology, where we are today in Paris, France. Bonjour, Fabrice.
So, remind our viewers: What is ESMO? What does it do? And why is it so important?
Fabrice André, MD, PhD: First ESMO, is a scientific society – a member-based organization with around 25,000 members.
Dr. Whyte: Equivalent to ASCO (American Society of Clinical Oncology) in the United States, correct?
Dr. André: It has members worldwide, from all over the world. And it aims at disseminating science, educating. The name is European, so it has some roots in Europe; but it is really a global organization for education, dissemination, and also more and more to generate frameworks for the standards of treatment and the common terminology for healthcare professionals to better care for patients.
Dr. Whyte: What are you most excited by at this conference in terms of the innovations that are being discussed?
Dr. André: Today we are at ESMO 2022 in Paris, with 28,000 people registered and the vast majority on site, and what has been the editorial line – the tagline – for the scientific committee is “understand the disease to better treat the patient.” This is extremely important; What are the determinants of outcomes if we want to integrate all the wealth of innovation that is coming?
So, then, what are the new things? In the Presidential Symposium, where we usually have the very new things, we will have very important presentations on the role of pollution on cancer and the biological mechanism that induces cancer. Why is it important? First, it has impact on public health. But also, it’s important because, for us, it’s raising the signal that the oncology community must start to invest in this field of prevention.
Dr. Whyte: I was at your booth, by the way, the ESMO booth here, and you have two bicycles, which impressed me. Nobody was on them, I might point out, but the focus was on prevention. But let’s also address how historically, the academic community, the scientific community hasn’t really been focused on prevention. It’s about treatment. So it’s fascinating that you’re talking about prevention, because usually we talk about precision medicine, right? We talk about checkpoint inhibitors; we talk about immunomodulators. And here you’re saying, “Hey, John, we need to understand how we prevent cancer,” which is really a misnomer in a way, because there are many different diseases. Would you agree with that?
Dr. André: I fully agree with you. But what is the premise we are trying to address here? The premise is that prevention has always been very low in the agenda of international conferences. And we think we want to give the signal that it’s really time now that clinical infrastructure, hospitals, invest in this field, create teams dedicated to prevention, new structures for prevention. Why? Because we are discovering step by step that it could be that some drugs we use for patients with cancer could also be developed in the field of prevention. And for this, we need the oncologists. So, more and more, our conviction is that it is the oncology community that will transform the field of prevention, and we need to invest now. Having said that, we have two very important abstracts on this question. The other one is about early cancer detection. But of course, we have our traditional session on immunotherapeutics, precision medicine, and all the wealth of randomized trials. And so in this field, for patients with cancer, what is the new information?
Dr. Whyte: We have this whole continuum. So you talk about prevention – how much cancer is preventable? Eighty percent? Seventy percent? What do you estimate?
Dr. André: You know, I’m also a scientist. So as a scientist, I will say that there is no limit for this question. No, the only limit is the knowledge.
Dr. Whyte: Well, there is some inherited mutation, so we do know that.
Dr. André: We can just go to the current status – what we know now – but I don’t see why we would put some limit on how much we can prevent cancer. But indeed, so far, what are the risk factors? Genetics, hereditary cancer, all habits, and we know them. It’s about tobacco, alcohol, sun, some sexual behavior, etc., that indeed account. In France, we say that around 40% of cancer could be preventable.
Dr. Whyte: More and more, we learn about the issues of gout, other inflammatory diseases; it can have an association, but then we have early screening as well. So, if we’re on this continuum, how excited are you by what’s happening with liquid biopsies, with other testing? Because if we can get a cancer instead of at 500,000 cells at the time of imaging, at 10 or 50 cells, while there are fragments, that’s revolutionary, isn’t it?
Dr. André: I fully agree with you. We will have an important trial presented during ESMO that is the first prospective trial testing the device called Galleri, a tool for early cancer detection based on ctDNA (circulating tumor DNA) analysis by methylation pattern.
Dr. Whyte: General screening of the population or a more tailored population with certain indications? Because right now, most of those have focused on a limited population or are used for patients who already have a cancer, and testing that way – you think it’s going to be broader?
Dr. André: What this trial is investigating is in participants who do not have cancer, 6,000 participants ...
Dr. Whyte: Pas de tout? No cancer at all?
Dr. André: No cancer.
Dr. Whyte: No family history?
Dr. André: They can have family history, but no detectable cancer – can ctDNA analysis detect cancer? And the answer is, indeed, there is around 1% positivity, and around 40% of them, indeed, had cancer. So why is it important? Because it’s really a landmark prospective trial that is telling us that a device based on ctDNA can detect cancer at early stage. Then, how many cancers? What percentage?
Dr. Whyte: Which type of cancer?
Dr. André: And is it going to have an impact on outcome? And for all the questions, we don’t have the answer here. But the answer we have here today is that with this device, done prospectively, you can detect some cancer that would not be detectable without symptoms.
Dr. Whyte: It’s only going to get better, too.
Dr. André: Yeah. So then the next step is improving technology, integrating this technology with other ones we already have, in order to increase the percentage of patients in which we detect cancer at an earlier stage.
Dr. Whyte: What about pancreatic cancer, cancers we can’t detect through screening? People forget that most cancers cannot be detected through screening, so we need better tools. We do know that there are inherited mutations. Those really aren’t preventable in many ways; the goal is to get them early. So then we move to treatments, and you talked about precision medicine. What excites you about what’s going on these days at ESMO right now.
Dr. André: We have many trials on precision medicine. We will have two randomized trials that investigate two new targets; one is gamma secretase inhibitor. So, it’s a first-in-class, first time we even hear about this target at a clinical conference. And the second highly expected trial is a clinical trial in patients with metastatic lung cancer, KRAS mutated, testing sotorasib, which is a KRAS inhibitor, and showing the magnitude of improvement associated with sotorasib. The trial is positive, and it improved PFS [progression-free survival] in these patients. So these are two new targets that are validated at this conference.
Then, if we go on another topic of genomics, there is a question that is extremely important: Can we define patients who present an outlier sensitivity to immunotherapeutics? There will be one trial presented in the Presidential Symposium of immunotherapeutics in patients with colon cancer and microsatellite instability (MSI), showing that a few weeks of immunotherapeutics followed by surgery can cure patients. Why is it important? It’s important because we are all facing a shortage in the healthcare workforce. We have fewer nurses, fewer doctors, and we all have issues of sustainability. So, really now is the time to think about precision medicine, how precision medicine, by identifying outlier responders, can decrease the amount of resources we need to cure a patient. And this trial on immunotherapeutics, guided by genomics, is exactly this point: 8 weeks of treatment to cure a patient.
Dr. Whyte: Do you think there’s going to be a cure for cancer 10 years from now?
Dr. André: What I’m convinced of is that, in the 10 years that are coming, we are going step by step; we’re going to continue to increase the life expectancy of patients with cancer.
Dr. Whyte: And quality of life too, right?
Dr. André: Quality of life is a major issue. We had today a keynote on digital medicine and how ePRO (electronic patient-reported outcomes) can help the patient to really decrease the burden of symptoms. Quality of life is, of course, extremely important because of the very high number of patients who are cured of cancer; we need to decrease the burden of symptom in patients.
Dr. Whyte: And even though cancer rates are going down in most areas of the world, we still globally have millions of deaths from cancer every year. And sometimes people forget that, because they hear about some of the innovations. But I want to end with this: Are we investing enough in cancer care? Because let’s be honest – there are other diseases that we also need to spend time on. Cardiovascular disease is a global burden; infectious disease is a global burden. Are governments, are industries spending enough on cancer research and development?
Dr. André: Well, we can always claim for more, no? This is how everyone is trying to be, I think. But the reality is that we are living in a world where we have limited resources. I think what is more important for me is to be sure that any euro or dollar invested in cancer research is well used and generates an impact for patients. That is the most important, I think.
Dr. Whyte: And that’s why outcomes are so important in this research.
Dr. André: My conviction is that we have the tools, meaning the knowledge, the biotechnology, to really go the next step in terms of improving outcomes for patients. And for this, we now need clinical trials and translational research, but the tools, meaning basic science, basic knowledge, biotechnology – the basement for progress is here. We need now to transform this into direct impact for the patient. But I would not like to finish by saying we need more money in the field; what we need are people who can transform one euro, one dollar into concrete and measurable advances.
Dr. Whyte: We’re going to need more time on another day because I want to ask you about diversity in clinical trials, how important that is. I want to ask you about pediatric cancers; there are a whole bunch of things that I want to talk to you about. So hopefully we’ll find more time when we’re not at a big international conference such as ESMO. So, Dr Fabrice André, I want to thank you for taking time today.
Dr. André: Thank you and have a nice day.
Dr. Whyte: Stay tuned for a future discussion with Dr André on more about where we’re going in terms of cancer research and development. Thanks for watching, everyone.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
John Whyte, MD: Welcome, everyone. I’m Dr John Whyte. I’m the chief medical officer at WebMD, and I’m joined today by Fabrice André. He is the chair of the scientific committee at the European Society for Medical Oncology, where we are today in Paris, France. Bonjour, Fabrice.
So, remind our viewers: What is ESMO? What does it do? And why is it so important?
Fabrice André, MD, PhD: First ESMO, is a scientific society – a member-based organization with around 25,000 members.
Dr. Whyte: Equivalent to ASCO (American Society of Clinical Oncology) in the United States, correct?
Dr. André: It has members worldwide, from all over the world. And it aims at disseminating science, educating. The name is European, so it has some roots in Europe; but it is really a global organization for education, dissemination, and also more and more to generate frameworks for the standards of treatment and the common terminology for healthcare professionals to better care for patients.
Dr. Whyte: What are you most excited by at this conference in terms of the innovations that are being discussed?
Dr. André: Today we are at ESMO 2022 in Paris, with 28,000 people registered and the vast majority on site, and what has been the editorial line – the tagline – for the scientific committee is “understand the disease to better treat the patient.” This is extremely important; What are the determinants of outcomes if we want to integrate all the wealth of innovation that is coming?
So, then, what are the new things? In the Presidential Symposium, where we usually have the very new things, we will have very important presentations on the role of pollution on cancer and the biological mechanism that induces cancer. Why is it important? First, it has impact on public health. But also, it’s important because, for us, it’s raising the signal that the oncology community must start to invest in this field of prevention.
Dr. Whyte: I was at your booth, by the way, the ESMO booth here, and you have two bicycles, which impressed me. Nobody was on them, I might point out, but the focus was on prevention. But let’s also address how historically, the academic community, the scientific community hasn’t really been focused on prevention. It’s about treatment. So it’s fascinating that you’re talking about prevention, because usually we talk about precision medicine, right? We talk about checkpoint inhibitors; we talk about immunomodulators. And here you’re saying, “Hey, John, we need to understand how we prevent cancer,” which is really a misnomer in a way, because there are many different diseases. Would you agree with that?
Dr. André: I fully agree with you. But what is the premise we are trying to address here? The premise is that prevention has always been very low in the agenda of international conferences. And we think we want to give the signal that it’s really time now that clinical infrastructure, hospitals, invest in this field, create teams dedicated to prevention, new structures for prevention. Why? Because we are discovering step by step that it could be that some drugs we use for patients with cancer could also be developed in the field of prevention. And for this, we need the oncologists. So, more and more, our conviction is that it is the oncology community that will transform the field of prevention, and we need to invest now. Having said that, we have two very important abstracts on this question. The other one is about early cancer detection. But of course, we have our traditional session on immunotherapeutics, precision medicine, and all the wealth of randomized trials. And so in this field, for patients with cancer, what is the new information?
Dr. Whyte: We have this whole continuum. So you talk about prevention – how much cancer is preventable? Eighty percent? Seventy percent? What do you estimate?
Dr. André: You know, I’m also a scientist. So as a scientist, I will say that there is no limit for this question. No, the only limit is the knowledge.
Dr. Whyte: Well, there is some inherited mutation, so we do know that.
Dr. André: We can just go to the current status – what we know now – but I don’t see why we would put some limit on how much we can prevent cancer. But indeed, so far, what are the risk factors? Genetics, hereditary cancer, all habits, and we know them. It’s about tobacco, alcohol, sun, some sexual behavior, etc., that indeed account. In France, we say that around 40% of cancer could be preventable.
Dr. Whyte: More and more, we learn about the issues of gout, other inflammatory diseases; it can have an association, but then we have early screening as well. So, if we’re on this continuum, how excited are you by what’s happening with liquid biopsies, with other testing? Because if we can get a cancer instead of at 500,000 cells at the time of imaging, at 10 or 50 cells, while there are fragments, that’s revolutionary, isn’t it?
Dr. André: I fully agree with you. We will have an important trial presented during ESMO that is the first prospective trial testing the device called Galleri, a tool for early cancer detection based on ctDNA (circulating tumor DNA) analysis by methylation pattern.
Dr. Whyte: General screening of the population or a more tailored population with certain indications? Because right now, most of those have focused on a limited population or are used for patients who already have a cancer, and testing that way – you think it’s going to be broader?
Dr. André: What this trial is investigating is in participants who do not have cancer, 6,000 participants ...
Dr. Whyte: Pas de tout? No cancer at all?
Dr. André: No cancer.
Dr. Whyte: No family history?
Dr. André: They can have family history, but no detectable cancer – can ctDNA analysis detect cancer? And the answer is, indeed, there is around 1% positivity, and around 40% of them, indeed, had cancer. So why is it important? Because it’s really a landmark prospective trial that is telling us that a device based on ctDNA can detect cancer at early stage. Then, how many cancers? What percentage?
Dr. Whyte: Which type of cancer?
Dr. André: And is it going to have an impact on outcome? And for all the questions, we don’t have the answer here. But the answer we have here today is that with this device, done prospectively, you can detect some cancer that would not be detectable without symptoms.
Dr. Whyte: It’s only going to get better, too.
Dr. André: Yeah. So then the next step is improving technology, integrating this technology with other ones we already have, in order to increase the percentage of patients in which we detect cancer at an earlier stage.
Dr. Whyte: What about pancreatic cancer, cancers we can’t detect through screening? People forget that most cancers cannot be detected through screening, so we need better tools. We do know that there are inherited mutations. Those really aren’t preventable in many ways; the goal is to get them early. So then we move to treatments, and you talked about precision medicine. What excites you about what’s going on these days at ESMO right now.
Dr. André: We have many trials on precision medicine. We will have two randomized trials that investigate two new targets; one is gamma secretase inhibitor. So, it’s a first-in-class, first time we even hear about this target at a clinical conference. And the second highly expected trial is a clinical trial in patients with metastatic lung cancer, KRAS mutated, testing sotorasib, which is a KRAS inhibitor, and showing the magnitude of improvement associated with sotorasib. The trial is positive, and it improved PFS [progression-free survival] in these patients. So these are two new targets that are validated at this conference.
Then, if we go on another topic of genomics, there is a question that is extremely important: Can we define patients who present an outlier sensitivity to immunotherapeutics? There will be one trial presented in the Presidential Symposium of immunotherapeutics in patients with colon cancer and microsatellite instability (MSI), showing that a few weeks of immunotherapeutics followed by surgery can cure patients. Why is it important? It’s important because we are all facing a shortage in the healthcare workforce. We have fewer nurses, fewer doctors, and we all have issues of sustainability. So, really now is the time to think about precision medicine, how precision medicine, by identifying outlier responders, can decrease the amount of resources we need to cure a patient. And this trial on immunotherapeutics, guided by genomics, is exactly this point: 8 weeks of treatment to cure a patient.
Dr. Whyte: Do you think there’s going to be a cure for cancer 10 years from now?
Dr. André: What I’m convinced of is that, in the 10 years that are coming, we are going step by step; we’re going to continue to increase the life expectancy of patients with cancer.
Dr. Whyte: And quality of life too, right?
Dr. André: Quality of life is a major issue. We had today a keynote on digital medicine and how ePRO (electronic patient-reported outcomes) can help the patient to really decrease the burden of symptoms. Quality of life is, of course, extremely important because of the very high number of patients who are cured of cancer; we need to decrease the burden of symptom in patients.
Dr. Whyte: And even though cancer rates are going down in most areas of the world, we still globally have millions of deaths from cancer every year. And sometimes people forget that, because they hear about some of the innovations. But I want to end with this: Are we investing enough in cancer care? Because let’s be honest – there are other diseases that we also need to spend time on. Cardiovascular disease is a global burden; infectious disease is a global burden. Are governments, are industries spending enough on cancer research and development?
Dr. André: Well, we can always claim for more, no? This is how everyone is trying to be, I think. But the reality is that we are living in a world where we have limited resources. I think what is more important for me is to be sure that any euro or dollar invested in cancer research is well used and generates an impact for patients. That is the most important, I think.
Dr. Whyte: And that’s why outcomes are so important in this research.
Dr. André: My conviction is that we have the tools, meaning the knowledge, the biotechnology, to really go the next step in terms of improving outcomes for patients. And for this, we now need clinical trials and translational research, but the tools, meaning basic science, basic knowledge, biotechnology – the basement for progress is here. We need now to transform this into direct impact for the patient. But I would not like to finish by saying we need more money in the field; what we need are people who can transform one euro, one dollar into concrete and measurable advances.
Dr. Whyte: We’re going to need more time on another day because I want to ask you about diversity in clinical trials, how important that is. I want to ask you about pediatric cancers; there are a whole bunch of things that I want to talk to you about. So hopefully we’ll find more time when we’re not at a big international conference such as ESMO. So, Dr Fabrice André, I want to thank you for taking time today.
Dr. André: Thank you and have a nice day.
Dr. Whyte: Stay tuned for a future discussion with Dr André on more about where we’re going in terms of cancer research and development. Thanks for watching, everyone.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
John Whyte, MD: Welcome, everyone. I’m Dr John Whyte. I’m the chief medical officer at WebMD, and I’m joined today by Fabrice André. He is the chair of the scientific committee at the European Society for Medical Oncology, where we are today in Paris, France. Bonjour, Fabrice.
So, remind our viewers: What is ESMO? What does it do? And why is it so important?
Fabrice André, MD, PhD: First ESMO, is a scientific society – a member-based organization with around 25,000 members.
Dr. Whyte: Equivalent to ASCO (American Society of Clinical Oncology) in the United States, correct?
Dr. André: It has members worldwide, from all over the world. And it aims at disseminating science, educating. The name is European, so it has some roots in Europe; but it is really a global organization for education, dissemination, and also more and more to generate frameworks for the standards of treatment and the common terminology for healthcare professionals to better care for patients.
Dr. Whyte: What are you most excited by at this conference in terms of the innovations that are being discussed?
Dr. André: Today we are at ESMO 2022 in Paris, with 28,000 people registered and the vast majority on site, and what has been the editorial line – the tagline – for the scientific committee is “understand the disease to better treat the patient.” This is extremely important; What are the determinants of outcomes if we want to integrate all the wealth of innovation that is coming?
So, then, what are the new things? In the Presidential Symposium, where we usually have the very new things, we will have very important presentations on the role of pollution on cancer and the biological mechanism that induces cancer. Why is it important? First, it has impact on public health. But also, it’s important because, for us, it’s raising the signal that the oncology community must start to invest in this field of prevention.
Dr. Whyte: I was at your booth, by the way, the ESMO booth here, and you have two bicycles, which impressed me. Nobody was on them, I might point out, but the focus was on prevention. But let’s also address how historically, the academic community, the scientific community hasn’t really been focused on prevention. It’s about treatment. So it’s fascinating that you’re talking about prevention, because usually we talk about precision medicine, right? We talk about checkpoint inhibitors; we talk about immunomodulators. And here you’re saying, “Hey, John, we need to understand how we prevent cancer,” which is really a misnomer in a way, because there are many different diseases. Would you agree with that?
Dr. André: I fully agree with you. But what is the premise we are trying to address here? The premise is that prevention has always been very low in the agenda of international conferences. And we think we want to give the signal that it’s really time now that clinical infrastructure, hospitals, invest in this field, create teams dedicated to prevention, new structures for prevention. Why? Because we are discovering step by step that it could be that some drugs we use for patients with cancer could also be developed in the field of prevention. And for this, we need the oncologists. So, more and more, our conviction is that it is the oncology community that will transform the field of prevention, and we need to invest now. Having said that, we have two very important abstracts on this question. The other one is about early cancer detection. But of course, we have our traditional session on immunotherapeutics, precision medicine, and all the wealth of randomized trials. And so in this field, for patients with cancer, what is the new information?
Dr. Whyte: We have this whole continuum. So you talk about prevention – how much cancer is preventable? Eighty percent? Seventy percent? What do you estimate?
Dr. André: You know, I’m also a scientist. So as a scientist, I will say that there is no limit for this question. No, the only limit is the knowledge.
Dr. Whyte: Well, there is some inherited mutation, so we do know that.
Dr. André: We can just go to the current status – what we know now – but I don’t see why we would put some limit on how much we can prevent cancer. But indeed, so far, what are the risk factors? Genetics, hereditary cancer, all habits, and we know them. It’s about tobacco, alcohol, sun, some sexual behavior, etc., that indeed account. In France, we say that around 40% of cancer could be preventable.
Dr. Whyte: More and more, we learn about the issues of gout, other inflammatory diseases; it can have an association, but then we have early screening as well. So, if we’re on this continuum, how excited are you by what’s happening with liquid biopsies, with other testing? Because if we can get a cancer instead of at 500,000 cells at the time of imaging, at 10 or 50 cells, while there are fragments, that’s revolutionary, isn’t it?
Dr. André: I fully agree with you. We will have an important trial presented during ESMO that is the first prospective trial testing the device called Galleri, a tool for early cancer detection based on ctDNA (circulating tumor DNA) analysis by methylation pattern.
Dr. Whyte: General screening of the population or a more tailored population with certain indications? Because right now, most of those have focused on a limited population or are used for patients who already have a cancer, and testing that way – you think it’s going to be broader?
Dr. André: What this trial is investigating is in participants who do not have cancer, 6,000 participants ...
Dr. Whyte: Pas de tout? No cancer at all?
Dr. André: No cancer.
Dr. Whyte: No family history?
Dr. André: They can have family history, but no detectable cancer – can ctDNA analysis detect cancer? And the answer is, indeed, there is around 1% positivity, and around 40% of them, indeed, had cancer. So why is it important? Because it’s really a landmark prospective trial that is telling us that a device based on ctDNA can detect cancer at early stage. Then, how many cancers? What percentage?
Dr. Whyte: Which type of cancer?
Dr. André: And is it going to have an impact on outcome? And for all the questions, we don’t have the answer here. But the answer we have here today is that with this device, done prospectively, you can detect some cancer that would not be detectable without symptoms.
Dr. Whyte: It’s only going to get better, too.
Dr. André: Yeah. So then the next step is improving technology, integrating this technology with other ones we already have, in order to increase the percentage of patients in which we detect cancer at an earlier stage.
Dr. Whyte: What about pancreatic cancer, cancers we can’t detect through screening? People forget that most cancers cannot be detected through screening, so we need better tools. We do know that there are inherited mutations. Those really aren’t preventable in many ways; the goal is to get them early. So then we move to treatments, and you talked about precision medicine. What excites you about what’s going on these days at ESMO right now.
Dr. André: We have many trials on precision medicine. We will have two randomized trials that investigate two new targets; one is gamma secretase inhibitor. So, it’s a first-in-class, first time we even hear about this target at a clinical conference. And the second highly expected trial is a clinical trial in patients with metastatic lung cancer, KRAS mutated, testing sotorasib, which is a KRAS inhibitor, and showing the magnitude of improvement associated with sotorasib. The trial is positive, and it improved PFS [progression-free survival] in these patients. So these are two new targets that are validated at this conference.
Then, if we go on another topic of genomics, there is a question that is extremely important: Can we define patients who present an outlier sensitivity to immunotherapeutics? There will be one trial presented in the Presidential Symposium of immunotherapeutics in patients with colon cancer and microsatellite instability (MSI), showing that a few weeks of immunotherapeutics followed by surgery can cure patients. Why is it important? It’s important because we are all facing a shortage in the healthcare workforce. We have fewer nurses, fewer doctors, and we all have issues of sustainability. So, really now is the time to think about precision medicine, how precision medicine, by identifying outlier responders, can decrease the amount of resources we need to cure a patient. And this trial on immunotherapeutics, guided by genomics, is exactly this point: 8 weeks of treatment to cure a patient.
Dr. Whyte: Do you think there’s going to be a cure for cancer 10 years from now?
Dr. André: What I’m convinced of is that, in the 10 years that are coming, we are going step by step; we’re going to continue to increase the life expectancy of patients with cancer.
Dr. Whyte: And quality of life too, right?
Dr. André: Quality of life is a major issue. We had today a keynote on digital medicine and how ePRO (electronic patient-reported outcomes) can help the patient to really decrease the burden of symptoms. Quality of life is, of course, extremely important because of the very high number of patients who are cured of cancer; we need to decrease the burden of symptom in patients.
Dr. Whyte: And even though cancer rates are going down in most areas of the world, we still globally have millions of deaths from cancer every year. And sometimes people forget that, because they hear about some of the innovations. But I want to end with this: Are we investing enough in cancer care? Because let’s be honest – there are other diseases that we also need to spend time on. Cardiovascular disease is a global burden; infectious disease is a global burden. Are governments, are industries spending enough on cancer research and development?
Dr. André: Well, we can always claim for more, no? This is how everyone is trying to be, I think. But the reality is that we are living in a world where we have limited resources. I think what is more important for me is to be sure that any euro or dollar invested in cancer research is well used and generates an impact for patients. That is the most important, I think.
Dr. Whyte: And that’s why outcomes are so important in this research.
Dr. André: My conviction is that we have the tools, meaning the knowledge, the biotechnology, to really go the next step in terms of improving outcomes for patients. And for this, we now need clinical trials and translational research, but the tools, meaning basic science, basic knowledge, biotechnology – the basement for progress is here. We need now to transform this into direct impact for the patient. But I would not like to finish by saying we need more money in the field; what we need are people who can transform one euro, one dollar into concrete and measurable advances.
Dr. Whyte: We’re going to need more time on another day because I want to ask you about diversity in clinical trials, how important that is. I want to ask you about pediatric cancers; there are a whole bunch of things that I want to talk to you about. So hopefully we’ll find more time when we’re not at a big international conference such as ESMO. So, Dr Fabrice André, I want to thank you for taking time today.
Dr. André: Thank you and have a nice day.
Dr. Whyte: Stay tuned for a future discussion with Dr André on more about where we’re going in terms of cancer research and development. Thanks for watching, everyone.
A version of this article first appeared on Medscape.com.
I am not fine: The heavy toll cancer takes
PARIS – “I thought I was as exhausted, and isolated, and neglected as I could get, and then he came home.”
Those were the words of Kate Washington, PhD, from Sacramento as she gave a moving account of the immense burden she felt as caregiver to her husband with cancer.
She was taking part in the session, “I am FINE: Frustrated * Isolated * Neglected * Emotional,” at the annual meeting of the European Society for Medical Oncology. In that session,
Dr. Washington, author of “Already Toast: Caregiving and Burnout in America” (Boston: Beacon Press, 2021), explained that she cared for her husband and young family while he was “suffering through two different kinds of lymphoma and really devastating stem cell transplants.”
When her husband was first diagnosed with a rare form of lymphoma in 2015, he was placed on a watch-and-wait protocol. At that point, he seemed fine, Dr. Washington said.
A few months later, he started coughing up blood. After being rushed to the emergency department, doctors found that a slow-growing lung tumor had ruptured.
Three weeks later, he came out of the hospital with a collapsed lung – an effect of his chemotherapy, Dr. Washington said.
But that was hardly the last word. He soon experienced relapse with a “very aggressive” form of his disease, and in 2016, he underwent a stem cell transplant.
“He spent 1½ months in the hospital ... in isolation, not seeing our daughters,” Dr. Washington said. He lost his vision and developed grade 4 graft-versus-host disease, among other problems.
He was alive, just barely, Dr. Washington said.
“As you might imagine, I was pulled between the hospital and the home, taking care of our daughters, who were not seeing him during that time,” she recalled.
But every time someone asked her whether she was okay, she replied: “I am fine.”
“A total lie,” she admitted.
Dr. Washington felt frustrated, not only from the financial strain of out-of-pocket health care costs and lost earnings but also from fast evolving relationships and a feeling of being “unseen and underappreciated.”
Another jarring change: When her husband was discharged from the hospital, Dr. Washington was suddenly thrust into the role of full-time caretaker.
Her husband could not be left alone, his doctor had said. And with two young children, Dr. Washington did not know how she would manage.
The demands of being a full-time caregiver are intense. Caregivers, Dr. Washington explained, can spend 32 hours a week looking after a loved one with cancer.
Like Dr. Washington, most caregivers feel they have no choice but to take on this intense role – one for which they have little or no training or preparation. The nonstop demands leave little time for self-care and can lead to high rates of caregiver injury and illness.
Isolation often creeps in because it can be “hard to ask for help,” she said. About 30% of caregivers report having depression or anxiety, and 21% feel lonely.
“When he was very ill, I found it really difficult to connect with other people and my friends,” Dr. Washington recalled. “I didn’t feel like I could really adequately explain the kind of strain that I was under.”
Are patients fine?
Like caregivers, patients often say they are fine when they are not.
The toll cancer takes on patients is immense. Natacha Bolanos Fernandez, from the Lymphoma Coalition Europe, highlighted the physical, mental, and social strain that can affect patients with cancer.
The physical aspects can encompass a host of problems – fatigue, night sweats, weight loss, and the vomiting that accompanies many cancer treatments. Patients may face changes in their mobility and independence as well. The mental side of cancer can include anxiety, depression, and psychological distress, while the social aspects span changing, perhaps strained, relationships with family and friends.
Fatigue, in particular, is an underreported, underdiagnosed, and undertreated problem, Ms. Fernandez noted. According to recent survey data from the Lymphoma Coalition’s Global Patient Survey, 72% of patients reported fatigue. This problem worsened over time, with 59% reporting fatigue after their diagnosis and up to 82% among patients who experienced relapse two or more times.
Fatigue “may be getting worse rather than better over time,” Ms. Fernandez said, and many patients felt that their life had changed completely because of cancer-related fatigue.
To help patients manage, the Lymphoma Coalition has published a report on the impact of cancer-related fatigue and how to improve outcomes. Methods include greater awareness, regular screening, and interventions such as yoga or mindfulness-based cognitive therapy.
Are clinicians fine?
Nurses and physicians face challenges caring for patients with cancer.
Although “nurses love their jobs and are extremely committed,” the impact cancer has on a nursing career is often undervalued or “neglected,” said Lena Sharp, RN, PhD, of the Regional Cancer Centre, Stockholm-Gotland.
Burnout, in particular, remains a problem among oncologists and nurses, and it was made worse during the COVID-19 pandemic.
Fatima Cardoso, MD, explained that burnout has an impact on doctors as well as patients because it affects communication with patients and performance. Physicians can, for instance, appear detached, emotional, or tired.
Patients may then feel less inclined to tell their oncologist how they’re feeling, said Dr. Cardoso, director of the breast unit at Champalimaud Clinical Center, Lisbon.
It is important to remember to not just focus on the patient’s disease or treatment but to also ask how they are doing and what is going on in their lives.
Above all, “show that you care,” said Dr. Cardoso.
The Lymphoma Coalition Europe has relationships with Bristol-Myers Squibb, Establishment Labs, Kyowa Kirin, Novartis, Roche, Takeda. Dr. Cardoso has relationships with Amgen, Astellas/Medivation, AstraZeneca, Celgene, Daiichi Sankyo, Eisai, GE Oncology, Genentech, GlaxoSmithKline, and other companies. No other relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
PARIS – “I thought I was as exhausted, and isolated, and neglected as I could get, and then he came home.”
Those were the words of Kate Washington, PhD, from Sacramento as she gave a moving account of the immense burden she felt as caregiver to her husband with cancer.
She was taking part in the session, “I am FINE: Frustrated * Isolated * Neglected * Emotional,” at the annual meeting of the European Society for Medical Oncology. In that session,
Dr. Washington, author of “Already Toast: Caregiving and Burnout in America” (Boston: Beacon Press, 2021), explained that she cared for her husband and young family while he was “suffering through two different kinds of lymphoma and really devastating stem cell transplants.”
When her husband was first diagnosed with a rare form of lymphoma in 2015, he was placed on a watch-and-wait protocol. At that point, he seemed fine, Dr. Washington said.
A few months later, he started coughing up blood. After being rushed to the emergency department, doctors found that a slow-growing lung tumor had ruptured.
Three weeks later, he came out of the hospital with a collapsed lung – an effect of his chemotherapy, Dr. Washington said.
But that was hardly the last word. He soon experienced relapse with a “very aggressive” form of his disease, and in 2016, he underwent a stem cell transplant.
“He spent 1½ months in the hospital ... in isolation, not seeing our daughters,” Dr. Washington said. He lost his vision and developed grade 4 graft-versus-host disease, among other problems.
He was alive, just barely, Dr. Washington said.
“As you might imagine, I was pulled between the hospital and the home, taking care of our daughters, who were not seeing him during that time,” she recalled.
But every time someone asked her whether she was okay, she replied: “I am fine.”
“A total lie,” she admitted.
Dr. Washington felt frustrated, not only from the financial strain of out-of-pocket health care costs and lost earnings but also from fast evolving relationships and a feeling of being “unseen and underappreciated.”
Another jarring change: When her husband was discharged from the hospital, Dr. Washington was suddenly thrust into the role of full-time caretaker.
Her husband could not be left alone, his doctor had said. And with two young children, Dr. Washington did not know how she would manage.
The demands of being a full-time caregiver are intense. Caregivers, Dr. Washington explained, can spend 32 hours a week looking after a loved one with cancer.
Like Dr. Washington, most caregivers feel they have no choice but to take on this intense role – one for which they have little or no training or preparation. The nonstop demands leave little time for self-care and can lead to high rates of caregiver injury and illness.
Isolation often creeps in because it can be “hard to ask for help,” she said. About 30% of caregivers report having depression or anxiety, and 21% feel lonely.
“When he was very ill, I found it really difficult to connect with other people and my friends,” Dr. Washington recalled. “I didn’t feel like I could really adequately explain the kind of strain that I was under.”
Are patients fine?
Like caregivers, patients often say they are fine when they are not.
The toll cancer takes on patients is immense. Natacha Bolanos Fernandez, from the Lymphoma Coalition Europe, highlighted the physical, mental, and social strain that can affect patients with cancer.
The physical aspects can encompass a host of problems – fatigue, night sweats, weight loss, and the vomiting that accompanies many cancer treatments. Patients may face changes in their mobility and independence as well. The mental side of cancer can include anxiety, depression, and psychological distress, while the social aspects span changing, perhaps strained, relationships with family and friends.
Fatigue, in particular, is an underreported, underdiagnosed, and undertreated problem, Ms. Fernandez noted. According to recent survey data from the Lymphoma Coalition’s Global Patient Survey, 72% of patients reported fatigue. This problem worsened over time, with 59% reporting fatigue after their diagnosis and up to 82% among patients who experienced relapse two or more times.
Fatigue “may be getting worse rather than better over time,” Ms. Fernandez said, and many patients felt that their life had changed completely because of cancer-related fatigue.
To help patients manage, the Lymphoma Coalition has published a report on the impact of cancer-related fatigue and how to improve outcomes. Methods include greater awareness, regular screening, and interventions such as yoga or mindfulness-based cognitive therapy.
Are clinicians fine?
Nurses and physicians face challenges caring for patients with cancer.
Although “nurses love their jobs and are extremely committed,” the impact cancer has on a nursing career is often undervalued or “neglected,” said Lena Sharp, RN, PhD, of the Regional Cancer Centre, Stockholm-Gotland.
Burnout, in particular, remains a problem among oncologists and nurses, and it was made worse during the COVID-19 pandemic.
Fatima Cardoso, MD, explained that burnout has an impact on doctors as well as patients because it affects communication with patients and performance. Physicians can, for instance, appear detached, emotional, or tired.
Patients may then feel less inclined to tell their oncologist how they’re feeling, said Dr. Cardoso, director of the breast unit at Champalimaud Clinical Center, Lisbon.
It is important to remember to not just focus on the patient’s disease or treatment but to also ask how they are doing and what is going on in their lives.
Above all, “show that you care,” said Dr. Cardoso.
The Lymphoma Coalition Europe has relationships with Bristol-Myers Squibb, Establishment Labs, Kyowa Kirin, Novartis, Roche, Takeda. Dr. Cardoso has relationships with Amgen, Astellas/Medivation, AstraZeneca, Celgene, Daiichi Sankyo, Eisai, GE Oncology, Genentech, GlaxoSmithKline, and other companies. No other relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
PARIS – “I thought I was as exhausted, and isolated, and neglected as I could get, and then he came home.”
Those were the words of Kate Washington, PhD, from Sacramento as she gave a moving account of the immense burden she felt as caregiver to her husband with cancer.
She was taking part in the session, “I am FINE: Frustrated * Isolated * Neglected * Emotional,” at the annual meeting of the European Society for Medical Oncology. In that session,
Dr. Washington, author of “Already Toast: Caregiving and Burnout in America” (Boston: Beacon Press, 2021), explained that she cared for her husband and young family while he was “suffering through two different kinds of lymphoma and really devastating stem cell transplants.”
When her husband was first diagnosed with a rare form of lymphoma in 2015, he was placed on a watch-and-wait protocol. At that point, he seemed fine, Dr. Washington said.
A few months later, he started coughing up blood. After being rushed to the emergency department, doctors found that a slow-growing lung tumor had ruptured.
Three weeks later, he came out of the hospital with a collapsed lung – an effect of his chemotherapy, Dr. Washington said.
But that was hardly the last word. He soon experienced relapse with a “very aggressive” form of his disease, and in 2016, he underwent a stem cell transplant.
“He spent 1½ months in the hospital ... in isolation, not seeing our daughters,” Dr. Washington said. He lost his vision and developed grade 4 graft-versus-host disease, among other problems.
He was alive, just barely, Dr. Washington said.
“As you might imagine, I was pulled between the hospital and the home, taking care of our daughters, who were not seeing him during that time,” she recalled.
But every time someone asked her whether she was okay, she replied: “I am fine.”
“A total lie,” she admitted.
Dr. Washington felt frustrated, not only from the financial strain of out-of-pocket health care costs and lost earnings but also from fast evolving relationships and a feeling of being “unseen and underappreciated.”
Another jarring change: When her husband was discharged from the hospital, Dr. Washington was suddenly thrust into the role of full-time caretaker.
Her husband could not be left alone, his doctor had said. And with two young children, Dr. Washington did not know how she would manage.
The demands of being a full-time caregiver are intense. Caregivers, Dr. Washington explained, can spend 32 hours a week looking after a loved one with cancer.
Like Dr. Washington, most caregivers feel they have no choice but to take on this intense role – one for which they have little or no training or preparation. The nonstop demands leave little time for self-care and can lead to high rates of caregiver injury and illness.
Isolation often creeps in because it can be “hard to ask for help,” she said. About 30% of caregivers report having depression or anxiety, and 21% feel lonely.
“When he was very ill, I found it really difficult to connect with other people and my friends,” Dr. Washington recalled. “I didn’t feel like I could really adequately explain the kind of strain that I was under.”
Are patients fine?
Like caregivers, patients often say they are fine when they are not.
The toll cancer takes on patients is immense. Natacha Bolanos Fernandez, from the Lymphoma Coalition Europe, highlighted the physical, mental, and social strain that can affect patients with cancer.
The physical aspects can encompass a host of problems – fatigue, night sweats, weight loss, and the vomiting that accompanies many cancer treatments. Patients may face changes in their mobility and independence as well. The mental side of cancer can include anxiety, depression, and psychological distress, while the social aspects span changing, perhaps strained, relationships with family and friends.
Fatigue, in particular, is an underreported, underdiagnosed, and undertreated problem, Ms. Fernandez noted. According to recent survey data from the Lymphoma Coalition’s Global Patient Survey, 72% of patients reported fatigue. This problem worsened over time, with 59% reporting fatigue after their diagnosis and up to 82% among patients who experienced relapse two or more times.
Fatigue “may be getting worse rather than better over time,” Ms. Fernandez said, and many patients felt that their life had changed completely because of cancer-related fatigue.
To help patients manage, the Lymphoma Coalition has published a report on the impact of cancer-related fatigue and how to improve outcomes. Methods include greater awareness, regular screening, and interventions such as yoga or mindfulness-based cognitive therapy.
Are clinicians fine?
Nurses and physicians face challenges caring for patients with cancer.
Although “nurses love their jobs and are extremely committed,” the impact cancer has on a nursing career is often undervalued or “neglected,” said Lena Sharp, RN, PhD, of the Regional Cancer Centre, Stockholm-Gotland.
Burnout, in particular, remains a problem among oncologists and nurses, and it was made worse during the COVID-19 pandemic.
Fatima Cardoso, MD, explained that burnout has an impact on doctors as well as patients because it affects communication with patients and performance. Physicians can, for instance, appear detached, emotional, or tired.
Patients may then feel less inclined to tell their oncologist how they’re feeling, said Dr. Cardoso, director of the breast unit at Champalimaud Clinical Center, Lisbon.
It is important to remember to not just focus on the patient’s disease or treatment but to also ask how they are doing and what is going on in their lives.
Above all, “show that you care,” said Dr. Cardoso.
The Lymphoma Coalition Europe has relationships with Bristol-Myers Squibb, Establishment Labs, Kyowa Kirin, Novartis, Roche, Takeda. Dr. Cardoso has relationships with Amgen, Astellas/Medivation, AstraZeneca, Celgene, Daiichi Sankyo, Eisai, GE Oncology, Genentech, GlaxoSmithKline, and other companies. No other relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
AT ESMO CONGRESS 2022
Folic acid tied to a reduction in suicide attempts
new research suggests.
After adjusting for multiple factors, results from a large pharmaco-epidemiological study showed taking folic acid was associated with a 44% reduction in suicide events.
“These results are really putting folic acid squarely on the map as a potential for large-scale, population-level prevention,” lead author Robert D. Gibbons, PhD, professor of biostatistics, Center for Health Statistics, University of Chicago, said in an interview.
“Folic acid is safe, inexpensive, and generally available, and if future randomized controlled trials show this association is beyond a shadow of a doubt causal, we have a new tool in the arsenal,” Dr. Gibbons said.
Having such a tool would be extremely important given that suicide is such a significant public health crisis worldwide, he added.
The findings were published online in JAMA Psychiatry.
Previous research ‘fairly thin’
Folate, the naturally occurring form of B9, is essential for neurogenesis, nucleotide synthesis, and methylation of homocysteine. Past research has suggested that taking folate can prevent neural tube and heart defects in the fetus during pregnancy – and may prevent strokes and reduce age-related hearing loss in adults.
In psychiatry, the role of folate has been recognized for more than a decade. It may enhance the effects of antidepressants; and folate deficiency can predict poorer response to SSRIs.
This has led to recommendations for folate augmentation in patients with low or normal levels at the start of depression treatment.
Although previous research has shown a link between folic acid and suicidality, the findings have been “fairly thin,” with studies being “generally small, and many are case series,” Dr. Gibbons said.
The current study follows an earlier analysis that used a novel statistical methodology for generating drug safety signals that was developed by Dr. Gibbons and colleagues. That study compared rates of suicide attempts before and after initiation of 922 drugs with at least 3,000 prescriptions.
Its results showed 10 drugs were associated with increased risk after exposure, with the strongest associations for alprazolam, butalbital, hydrocodone, and combination codeine/promethazine. In addition, 44 drugs were associated with decreased risk, many of which were antidepressants and antipsychotics.
“One of the most interesting findings in terms of the decreased risk was for folic acid,” said Dr. Gibbons.
He and his colleagues initially thought this was because of women taking folic acid during pregnancy. But when restricting the analysis to men, they found the same effect.
Their next step was to carry out the current large-scale pharmaco-epidemiological study.
Prescriptions for pain
The researchers used a health claims database that included 164 million enrollees. The study cohort was comprised of 866,586 adults with private health insurance (81.3% women; 10.4% aged 60 years and older) who filled a folic acid prescription between 2012 and 2017.
More than half of the folic acid prescriptions were associated with pain disorders. About 48% were for a single agent at a dosage of 1 mg/d, which is the upper tolerable limit for adults – including in pregnancy and lactation.
Other single-agent daily dosages ranging from 0.4 mg to 5 mg accounted for 0.11% of prescriptions. The remainder were multivitamins.
The participants were followed for 24 months. The within-person analysis compared suicide attempts or self-harm events resulting in an outpatient visit or inpatient admission during periods of folic acid treatment versus during periods without treatment.
During the study period, the overall suicidal event rate was 133 per 100,000 population, which is one-fourth the national rate reported by the National Institutes of Health of 600 per 100,000.
After adjusting for age, sex, diagnoses related to suicidal behavior and folic acid deficiency, history of folate-reducing medications, and history of suicidal events, the estimated hazard ratio for suicide events when taking folic acid was 0.56 (95% confidence interal, 0.48-0.65) – which indicates a 44% reduction in suicide events.
“This is a very large decrease and is extremely significant and exciting,” Dr. Gibbons said.
He noted the decrease in suicidal events may have been even greater considering the study captured only prescription folic acid, and participants may also have also taken over-the-counter products.
“The 44% reduction in suicide attempts may actually be an underestimate,” said Dr. Gibbons.
Age and sex did not moderate the association between folic acid and suicide attempts, and a similar association was found in women of childbearing age.
Provocative results?
The investigators also assessed a negative control group of 236,610 individuals using cyanocobalamin during the study period. Cyanocobalamin is a form of vitamin B12 that is essential for metabolism, blood cell synthesis, and the nervous system. It does not contain folic acid and is commonly used to treat anemia.
Results showed no association between cyanocobalamin and suicidal events in the adjusted analysis (HR, 1.01; 95% CI, 0.80-1.27) or unadjusted analysis (HR, 1.02; 95% CI, 0.80-1.28).
Dr. Gibbons noted this result boosts the argument that the association between folic acid and reduced suicidal attempts “isn’t just about health-seeking behavior like taking vitamin supplements.”
Another sensitivity analysis showed every additional month of treatment was associated with a 5% reduction in the suicidal event rate.
“This means the longer you take folic acid, the greater the benefit, which is what you would expect to see if there was a real association between a treatment and an outcome,” said Dr. Gibbons.
The new results “are so provocative that they really mandate the need for a well-controlled randomized controlled trial of folic acid and suicide events,” possibly in a high-risk population such as veterans, he noted.
Such a study could use longitudinal assessments of suicidal events, such as the validated Computerized Adaptive Test Suicide Scale, he added. This continuous scale of suicidality ranges from subclinical, signifying helplessness, hopelessness, and loss of pleasure, to suicide attempts and completion.
As for study limitations, the investigators noted that this study was observational, so there could be selection effects. And using claims data likely underrepresented the number of suicidal events because of incomplete reporting. As the researchers pointed out, the rate of suicidal events in this study was much lower than the national rate.
Other limitations cited were that the association between folic acid and suicidal events may be explained by healthy user bias; and although the investigators conducted a sensitivity analysis in women of childbearing age, they did not have data on women actively planning for a pregnancy.
‘Impressive, encouraging’
In a comment, Shirley Yen, PhD, associate professor of psychology, Beth Israel Deaconess Medical Center and Harvard Medical School, both in Boston, described the new findings as “quite impressive” and “extremely encouraging.”
However, she noted “it’s too premature” to suggest widespread use of folic acid in patients with depressive symptoms.
Dr. Yen, who has researched suicide risks previously, was not involved with the current study.
She did agree with the investigators that the results call for “more robustly controlled studies. These could include double-blind, randomized, controlled trials that could “more formally assess” all folic acid usage as opposed to prescriptions only, Dr. Yen said.
The study was funded by the NIH, the Agency for Healthcare Research and Quality, and the Center of Excellence for Suicide Prevention. Dr. Gibbons reported serving as an expert witness in cases for the Department of Justice; receiving expert witness fees from Merck, GlaxoSmithKline, Pfizer, and Wyeth; and having founded Adaptive Testing Technologies, which distributes the Computerized Adaptive Test Suicide Scale. Dr. Yen reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
After adjusting for multiple factors, results from a large pharmaco-epidemiological study showed taking folic acid was associated with a 44% reduction in suicide events.
“These results are really putting folic acid squarely on the map as a potential for large-scale, population-level prevention,” lead author Robert D. Gibbons, PhD, professor of biostatistics, Center for Health Statistics, University of Chicago, said in an interview.
“Folic acid is safe, inexpensive, and generally available, and if future randomized controlled trials show this association is beyond a shadow of a doubt causal, we have a new tool in the arsenal,” Dr. Gibbons said.
Having such a tool would be extremely important given that suicide is such a significant public health crisis worldwide, he added.
The findings were published online in JAMA Psychiatry.
Previous research ‘fairly thin’
Folate, the naturally occurring form of B9, is essential for neurogenesis, nucleotide synthesis, and methylation of homocysteine. Past research has suggested that taking folate can prevent neural tube and heart defects in the fetus during pregnancy – and may prevent strokes and reduce age-related hearing loss in adults.
In psychiatry, the role of folate has been recognized for more than a decade. It may enhance the effects of antidepressants; and folate deficiency can predict poorer response to SSRIs.
This has led to recommendations for folate augmentation in patients with low or normal levels at the start of depression treatment.
Although previous research has shown a link between folic acid and suicidality, the findings have been “fairly thin,” with studies being “generally small, and many are case series,” Dr. Gibbons said.
The current study follows an earlier analysis that used a novel statistical methodology for generating drug safety signals that was developed by Dr. Gibbons and colleagues. That study compared rates of suicide attempts before and after initiation of 922 drugs with at least 3,000 prescriptions.
Its results showed 10 drugs were associated with increased risk after exposure, with the strongest associations for alprazolam, butalbital, hydrocodone, and combination codeine/promethazine. In addition, 44 drugs were associated with decreased risk, many of which were antidepressants and antipsychotics.
“One of the most interesting findings in terms of the decreased risk was for folic acid,” said Dr. Gibbons.
He and his colleagues initially thought this was because of women taking folic acid during pregnancy. But when restricting the analysis to men, they found the same effect.
Their next step was to carry out the current large-scale pharmaco-epidemiological study.
Prescriptions for pain
The researchers used a health claims database that included 164 million enrollees. The study cohort was comprised of 866,586 adults with private health insurance (81.3% women; 10.4% aged 60 years and older) who filled a folic acid prescription between 2012 and 2017.
More than half of the folic acid prescriptions were associated with pain disorders. About 48% were for a single agent at a dosage of 1 mg/d, which is the upper tolerable limit for adults – including in pregnancy and lactation.
Other single-agent daily dosages ranging from 0.4 mg to 5 mg accounted for 0.11% of prescriptions. The remainder were multivitamins.
The participants were followed for 24 months. The within-person analysis compared suicide attempts or self-harm events resulting in an outpatient visit or inpatient admission during periods of folic acid treatment versus during periods without treatment.
During the study period, the overall suicidal event rate was 133 per 100,000 population, which is one-fourth the national rate reported by the National Institutes of Health of 600 per 100,000.
After adjusting for age, sex, diagnoses related to suicidal behavior and folic acid deficiency, history of folate-reducing medications, and history of suicidal events, the estimated hazard ratio for suicide events when taking folic acid was 0.56 (95% confidence interal, 0.48-0.65) – which indicates a 44% reduction in suicide events.
“This is a very large decrease and is extremely significant and exciting,” Dr. Gibbons said.
He noted the decrease in suicidal events may have been even greater considering the study captured only prescription folic acid, and participants may also have also taken over-the-counter products.
“The 44% reduction in suicide attempts may actually be an underestimate,” said Dr. Gibbons.
Age and sex did not moderate the association between folic acid and suicide attempts, and a similar association was found in women of childbearing age.
Provocative results?
The investigators also assessed a negative control group of 236,610 individuals using cyanocobalamin during the study period. Cyanocobalamin is a form of vitamin B12 that is essential for metabolism, blood cell synthesis, and the nervous system. It does not contain folic acid and is commonly used to treat anemia.
Results showed no association between cyanocobalamin and suicidal events in the adjusted analysis (HR, 1.01; 95% CI, 0.80-1.27) or unadjusted analysis (HR, 1.02; 95% CI, 0.80-1.28).
Dr. Gibbons noted this result boosts the argument that the association between folic acid and reduced suicidal attempts “isn’t just about health-seeking behavior like taking vitamin supplements.”
Another sensitivity analysis showed every additional month of treatment was associated with a 5% reduction in the suicidal event rate.
“This means the longer you take folic acid, the greater the benefit, which is what you would expect to see if there was a real association between a treatment and an outcome,” said Dr. Gibbons.
The new results “are so provocative that they really mandate the need for a well-controlled randomized controlled trial of folic acid and suicide events,” possibly in a high-risk population such as veterans, he noted.
Such a study could use longitudinal assessments of suicidal events, such as the validated Computerized Adaptive Test Suicide Scale, he added. This continuous scale of suicidality ranges from subclinical, signifying helplessness, hopelessness, and loss of pleasure, to suicide attempts and completion.
As for study limitations, the investigators noted that this study was observational, so there could be selection effects. And using claims data likely underrepresented the number of suicidal events because of incomplete reporting. As the researchers pointed out, the rate of suicidal events in this study was much lower than the national rate.
Other limitations cited were that the association between folic acid and suicidal events may be explained by healthy user bias; and although the investigators conducted a sensitivity analysis in women of childbearing age, they did not have data on women actively planning for a pregnancy.
‘Impressive, encouraging’
In a comment, Shirley Yen, PhD, associate professor of psychology, Beth Israel Deaconess Medical Center and Harvard Medical School, both in Boston, described the new findings as “quite impressive” and “extremely encouraging.”
However, she noted “it’s too premature” to suggest widespread use of folic acid in patients with depressive symptoms.
Dr. Yen, who has researched suicide risks previously, was not involved with the current study.
She did agree with the investigators that the results call for “more robustly controlled studies. These could include double-blind, randomized, controlled trials that could “more formally assess” all folic acid usage as opposed to prescriptions only, Dr. Yen said.
The study was funded by the NIH, the Agency for Healthcare Research and Quality, and the Center of Excellence for Suicide Prevention. Dr. Gibbons reported serving as an expert witness in cases for the Department of Justice; receiving expert witness fees from Merck, GlaxoSmithKline, Pfizer, and Wyeth; and having founded Adaptive Testing Technologies, which distributes the Computerized Adaptive Test Suicide Scale. Dr. Yen reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
After adjusting for multiple factors, results from a large pharmaco-epidemiological study showed taking folic acid was associated with a 44% reduction in suicide events.
“These results are really putting folic acid squarely on the map as a potential for large-scale, population-level prevention,” lead author Robert D. Gibbons, PhD, professor of biostatistics, Center for Health Statistics, University of Chicago, said in an interview.
“Folic acid is safe, inexpensive, and generally available, and if future randomized controlled trials show this association is beyond a shadow of a doubt causal, we have a new tool in the arsenal,” Dr. Gibbons said.
Having such a tool would be extremely important given that suicide is such a significant public health crisis worldwide, he added.
The findings were published online in JAMA Psychiatry.
Previous research ‘fairly thin’
Folate, the naturally occurring form of B9, is essential for neurogenesis, nucleotide synthesis, and methylation of homocysteine. Past research has suggested that taking folate can prevent neural tube and heart defects in the fetus during pregnancy – and may prevent strokes and reduce age-related hearing loss in adults.
In psychiatry, the role of folate has been recognized for more than a decade. It may enhance the effects of antidepressants; and folate deficiency can predict poorer response to SSRIs.
This has led to recommendations for folate augmentation in patients with low or normal levels at the start of depression treatment.
Although previous research has shown a link between folic acid and suicidality, the findings have been “fairly thin,” with studies being “generally small, and many are case series,” Dr. Gibbons said.
The current study follows an earlier analysis that used a novel statistical methodology for generating drug safety signals that was developed by Dr. Gibbons and colleagues. That study compared rates of suicide attempts before and after initiation of 922 drugs with at least 3,000 prescriptions.
Its results showed 10 drugs were associated with increased risk after exposure, with the strongest associations for alprazolam, butalbital, hydrocodone, and combination codeine/promethazine. In addition, 44 drugs were associated with decreased risk, many of which were antidepressants and antipsychotics.
“One of the most interesting findings in terms of the decreased risk was for folic acid,” said Dr. Gibbons.
He and his colleagues initially thought this was because of women taking folic acid during pregnancy. But when restricting the analysis to men, they found the same effect.
Their next step was to carry out the current large-scale pharmaco-epidemiological study.
Prescriptions for pain
The researchers used a health claims database that included 164 million enrollees. The study cohort was comprised of 866,586 adults with private health insurance (81.3% women; 10.4% aged 60 years and older) who filled a folic acid prescription between 2012 and 2017.
More than half of the folic acid prescriptions were associated with pain disorders. About 48% were for a single agent at a dosage of 1 mg/d, which is the upper tolerable limit for adults – including in pregnancy and lactation.
Other single-agent daily dosages ranging from 0.4 mg to 5 mg accounted for 0.11% of prescriptions. The remainder were multivitamins.
The participants were followed for 24 months. The within-person analysis compared suicide attempts or self-harm events resulting in an outpatient visit or inpatient admission during periods of folic acid treatment versus during periods without treatment.
During the study period, the overall suicidal event rate was 133 per 100,000 population, which is one-fourth the national rate reported by the National Institutes of Health of 600 per 100,000.
After adjusting for age, sex, diagnoses related to suicidal behavior and folic acid deficiency, history of folate-reducing medications, and history of suicidal events, the estimated hazard ratio for suicide events when taking folic acid was 0.56 (95% confidence interal, 0.48-0.65) – which indicates a 44% reduction in suicide events.
“This is a very large decrease and is extremely significant and exciting,” Dr. Gibbons said.
He noted the decrease in suicidal events may have been even greater considering the study captured only prescription folic acid, and participants may also have also taken over-the-counter products.
“The 44% reduction in suicide attempts may actually be an underestimate,” said Dr. Gibbons.
Age and sex did not moderate the association between folic acid and suicide attempts, and a similar association was found in women of childbearing age.
Provocative results?
The investigators also assessed a negative control group of 236,610 individuals using cyanocobalamin during the study period. Cyanocobalamin is a form of vitamin B12 that is essential for metabolism, blood cell synthesis, and the nervous system. It does not contain folic acid and is commonly used to treat anemia.
Results showed no association between cyanocobalamin and suicidal events in the adjusted analysis (HR, 1.01; 95% CI, 0.80-1.27) or unadjusted analysis (HR, 1.02; 95% CI, 0.80-1.28).
Dr. Gibbons noted this result boosts the argument that the association between folic acid and reduced suicidal attempts “isn’t just about health-seeking behavior like taking vitamin supplements.”
Another sensitivity analysis showed every additional month of treatment was associated with a 5% reduction in the suicidal event rate.
“This means the longer you take folic acid, the greater the benefit, which is what you would expect to see if there was a real association between a treatment and an outcome,” said Dr. Gibbons.
The new results “are so provocative that they really mandate the need for a well-controlled randomized controlled trial of folic acid and suicide events,” possibly in a high-risk population such as veterans, he noted.
Such a study could use longitudinal assessments of suicidal events, such as the validated Computerized Adaptive Test Suicide Scale, he added. This continuous scale of suicidality ranges from subclinical, signifying helplessness, hopelessness, and loss of pleasure, to suicide attempts and completion.
As for study limitations, the investigators noted that this study was observational, so there could be selection effects. And using claims data likely underrepresented the number of suicidal events because of incomplete reporting. As the researchers pointed out, the rate of suicidal events in this study was much lower than the national rate.
Other limitations cited were that the association between folic acid and suicidal events may be explained by healthy user bias; and although the investigators conducted a sensitivity analysis in women of childbearing age, they did not have data on women actively planning for a pregnancy.
‘Impressive, encouraging’
In a comment, Shirley Yen, PhD, associate professor of psychology, Beth Israel Deaconess Medical Center and Harvard Medical School, both in Boston, described the new findings as “quite impressive” and “extremely encouraging.”
However, she noted “it’s too premature” to suggest widespread use of folic acid in patients with depressive symptoms.
Dr. Yen, who has researched suicide risks previously, was not involved with the current study.
She did agree with the investigators that the results call for “more robustly controlled studies. These could include double-blind, randomized, controlled trials that could “more formally assess” all folic acid usage as opposed to prescriptions only, Dr. Yen said.
The study was funded by the NIH, the Agency for Healthcare Research and Quality, and the Center of Excellence for Suicide Prevention. Dr. Gibbons reported serving as an expert witness in cases for the Department of Justice; receiving expert witness fees from Merck, GlaxoSmithKline, Pfizer, and Wyeth; and having founded Adaptive Testing Technologies, which distributes the Computerized Adaptive Test Suicide Scale. Dr. Yen reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Ancient DNA discoveries lead to Nobel Prize in medicine and help explain how humans evolved
It all started with a 40,000-year-old bone. That Neanderthal bone contained enough DNA that Dr. Pääbo could start decades of research showing us modern humans, Homo sapiens, are genetically distinct from other now-extinct hominins such as Neanderthals and Denisovans.
The connection to physiology and medicine comes from the genes some modern humans carry from these ancient relatives. For example, Neanderthal genes transferred to Homo sapiens can explain how our immune systems react to different infections. Dr. Pääbo also discovered that a copy of a gene from another extinct relative, the Denisovans, gives modern-day Tibetans a survival advantage for living at high altitudes.
“Svante Pääbo accomplished something seemingly impossible: sequencing the genome of the Neanderthal, an extinct relative of present-day humans. He also made the sensational discovery of a previously unknown hominin, Denisova,” the Nobel Committee stated in a news release announcing the award. “Importantly, [Dr.] Pääbo also found that gene transfer had occurred from these now extinct hominins to Homo sapiens following the migration out of Africa around 70,000 years ago.”
Dr. Pääbo’s scientific discoveries only happened by pairing ancient DNA evidence with modern technology. An initial discovery occurred when Dr. Pääbo worked at the University of Munich (Germany), where he sequenced mitochondrial DNA from the 40,000-year-old Neanderthal bone. The sequencing of the human genome in the 1990s coupled with genomic DNA recovered from ancient bones allowed Dr. Pääbo to compare our DNA with that of Neanderthals and Denisovans. A lot of this work happened at the Max Planck Institute for Evolutionary Anthropology in Leipzig, Germany, which Dr. Pääbo founded in 1999 and where he still works today.
In 2008, a different 40,000-year-old fragment from a finger bone was discovered in a Denisova cave in the southern part of Siberia. Fortunately for Dr. Pääbo and his team, the DNA was well preserved and could be sequenced.
“The results caused a sensation: the DNA sequence was unique when compared to all known sequences from Neanderthals and present-day humans,” the Nobel Committee said.
In a big picture sense, Dr. Pääbo’s research helps answer where modern-day humans come from and how we interacted with our hominin relatives tens of thousands of years ago. His research is so pioneering that it created a new field of science called paleogenomics, the Nobel Committee noted.
“On behalf of The Physiological Society, I am delighted to congratulate Svante Pääbo for being awarded the Nobel Prize. His pioneering research shines a light on the physiology that makes humans unique from their ancestors,” David Paterson, DPhil, president of The Physiological Society, said in a statement.
“This is an important science discovery in evolutionary biology,” Dr. Paterson added. “Ascribing physiological function to highly conserved mitochondrial genes has been important in our understanding in high-altitude acclimatization as populations move and adapt to new environments, and how genetic variants affect us on a day-to-day basis in health and disease.”
A version of this article first appeared on Medscape.com.
It all started with a 40,000-year-old bone. That Neanderthal bone contained enough DNA that Dr. Pääbo could start decades of research showing us modern humans, Homo sapiens, are genetically distinct from other now-extinct hominins such as Neanderthals and Denisovans.
The connection to physiology and medicine comes from the genes some modern humans carry from these ancient relatives. For example, Neanderthal genes transferred to Homo sapiens can explain how our immune systems react to different infections. Dr. Pääbo also discovered that a copy of a gene from another extinct relative, the Denisovans, gives modern-day Tibetans a survival advantage for living at high altitudes.
“Svante Pääbo accomplished something seemingly impossible: sequencing the genome of the Neanderthal, an extinct relative of present-day humans. He also made the sensational discovery of a previously unknown hominin, Denisova,” the Nobel Committee stated in a news release announcing the award. “Importantly, [Dr.] Pääbo also found that gene transfer had occurred from these now extinct hominins to Homo sapiens following the migration out of Africa around 70,000 years ago.”
Dr. Pääbo’s scientific discoveries only happened by pairing ancient DNA evidence with modern technology. An initial discovery occurred when Dr. Pääbo worked at the University of Munich (Germany), where he sequenced mitochondrial DNA from the 40,000-year-old Neanderthal bone. The sequencing of the human genome in the 1990s coupled with genomic DNA recovered from ancient bones allowed Dr. Pääbo to compare our DNA with that of Neanderthals and Denisovans. A lot of this work happened at the Max Planck Institute for Evolutionary Anthropology in Leipzig, Germany, which Dr. Pääbo founded in 1999 and where he still works today.
In 2008, a different 40,000-year-old fragment from a finger bone was discovered in a Denisova cave in the southern part of Siberia. Fortunately for Dr. Pääbo and his team, the DNA was well preserved and could be sequenced.
“The results caused a sensation: the DNA sequence was unique when compared to all known sequences from Neanderthals and present-day humans,” the Nobel Committee said.
In a big picture sense, Dr. Pääbo’s research helps answer where modern-day humans come from and how we interacted with our hominin relatives tens of thousands of years ago. His research is so pioneering that it created a new field of science called paleogenomics, the Nobel Committee noted.
“On behalf of The Physiological Society, I am delighted to congratulate Svante Pääbo for being awarded the Nobel Prize. His pioneering research shines a light on the physiology that makes humans unique from their ancestors,” David Paterson, DPhil, president of The Physiological Society, said in a statement.
“This is an important science discovery in evolutionary biology,” Dr. Paterson added. “Ascribing physiological function to highly conserved mitochondrial genes has been important in our understanding in high-altitude acclimatization as populations move and adapt to new environments, and how genetic variants affect us on a day-to-day basis in health and disease.”
A version of this article first appeared on Medscape.com.
It all started with a 40,000-year-old bone. That Neanderthal bone contained enough DNA that Dr. Pääbo could start decades of research showing us modern humans, Homo sapiens, are genetically distinct from other now-extinct hominins such as Neanderthals and Denisovans.
The connection to physiology and medicine comes from the genes some modern humans carry from these ancient relatives. For example, Neanderthal genes transferred to Homo sapiens can explain how our immune systems react to different infections. Dr. Pääbo also discovered that a copy of a gene from another extinct relative, the Denisovans, gives modern-day Tibetans a survival advantage for living at high altitudes.
“Svante Pääbo accomplished something seemingly impossible: sequencing the genome of the Neanderthal, an extinct relative of present-day humans. He also made the sensational discovery of a previously unknown hominin, Denisova,” the Nobel Committee stated in a news release announcing the award. “Importantly, [Dr.] Pääbo also found that gene transfer had occurred from these now extinct hominins to Homo sapiens following the migration out of Africa around 70,000 years ago.”
Dr. Pääbo’s scientific discoveries only happened by pairing ancient DNA evidence with modern technology. An initial discovery occurred when Dr. Pääbo worked at the University of Munich (Germany), where he sequenced mitochondrial DNA from the 40,000-year-old Neanderthal bone. The sequencing of the human genome in the 1990s coupled with genomic DNA recovered from ancient bones allowed Dr. Pääbo to compare our DNA with that of Neanderthals and Denisovans. A lot of this work happened at the Max Planck Institute for Evolutionary Anthropology in Leipzig, Germany, which Dr. Pääbo founded in 1999 and where he still works today.
In 2008, a different 40,000-year-old fragment from a finger bone was discovered in a Denisova cave in the southern part of Siberia. Fortunately for Dr. Pääbo and his team, the DNA was well preserved and could be sequenced.
“The results caused a sensation: the DNA sequence was unique when compared to all known sequences from Neanderthals and present-day humans,” the Nobel Committee said.
In a big picture sense, Dr. Pääbo’s research helps answer where modern-day humans come from and how we interacted with our hominin relatives tens of thousands of years ago. His research is so pioneering that it created a new field of science called paleogenomics, the Nobel Committee noted.
“On behalf of The Physiological Society, I am delighted to congratulate Svante Pääbo for being awarded the Nobel Prize. His pioneering research shines a light on the physiology that makes humans unique from their ancestors,” David Paterson, DPhil, president of The Physiological Society, said in a statement.
“This is an important science discovery in evolutionary biology,” Dr. Paterson added. “Ascribing physiological function to highly conserved mitochondrial genes has been important in our understanding in high-altitude acclimatization as populations move and adapt to new environments, and how genetic variants affect us on a day-to-day basis in health and disease.”
A version of this article first appeared on Medscape.com.
Strong link found between enterovirus and type 1 diabetes
STOCKHOLM – Enterovirus infection appears to be strongly linked to both type 1 diabetes and islet cell autoantibodies, new research suggests.
The strength of the relationship, particularly within the first month of type 1 diabetes diagnosis, “further supports the rationale for development of enterovirus-targeted vaccines and antiviral therapy to prevent and reduce the impact of type 1 diabetes,” according to lead investigator Sonia Isaacs, MD, of the department of pediatrics and child health at the University of New South Wales, Sydney, Australia.
Enteroviruses are a large family of viruses responsible for many infections in children. These live in the intestinal tract but can cause a wide variety of illnesses. There are more than 70 different strains, which include the group A and group B coxsackieviruses, the polioviruses, hepatitis A virus, and several strains that just go by the name enterovirus.
Dr. Isaacs presented the data, from a meta-analysis of studies using modern molecular techniques, at the annual meeting of the European Association for the Study of Diabetes.
The findings raise the question of whether people should be routinely tested for enterovirus at the time of type 1 diabetes diagnosis, she said during her presentation.
Asked by this news organization about the implications for first-degree relatives of people with type 1 diabetes, Dr. Isaacs said that they are “definitely a population to watch out for,” with regard to enteroviral infections. “Type 1 diabetes is very diverse and has different endotypes. Different environmental factors may be implicated in these different endotypes, and it may be that the enteroviruses are quite important in the first-degree relative group.”
Asked to comment, session moderator Kamlesh Khunti, MD, PhD, told this news organization that the data were “compelling,” particularly in the short term after type 1 diabetes diagnosis. “It seems that there may be plausibility for enterovirus associated with the development of type 1 diabetes ... Are there methods by which we can reduce this risk with either antivirals or vaccinations? I think that needs to be tested.”
And in regard to first-degree relatives, “I think that’s the group to go for because the association is so highly correlated. I think that’s the group worth testing with any interventions,” said Dr. Khunti, professor of primary care diabetes and vascular medicine at the University of Leicester, England.
Link stronger a month after diagnosis, in close relatives, in Europe
The new meta-analysis is an update to a prior review published in 2011 by Dr. Isaacs’ group, which found that people with islet cell autoimmunity were more than four times as likely as were controls to have an enterovirus infection, and people with type 1 diabetes were almost 10 times as likely.
This new analysis focuses on studies using more modern molecular techniques for detecting viruses, including high throughput sequencing and single-cell technologies.
The analysis identified 60 studies with a total of 12,077 participants, of whom 900 had islet autoimmunity, 5,081 had type 1 diabetes, and 6,096 were controls. Thirty-five of the studies were from Europe, while others were from the United States, Asia, and the Middle East.
Of 16 studies examining enterovirus infection in islet autoimmunity, cases with islet autoimmunity were twice as likely to have an enterovirus infection at any time point compared to controls, a significant difference (odds ratio [OR], 2.07, P = .002.)
Among 48 studies reporting enterovirus infection in type 1 diabetes, those with type 1 diabetes were eight times as likely to have an enterovirus infection compared with controls (OR, 8.0, P < .00001).
In 25 studies including 2,977 participants with onset of type 1 diabetes within the prior month, those individuals were more than 16 times more likely to present with an enterovirus infection (OR, 16.2, P < .00001).
“The strength of this is association is greater than previously reported by both us and others,” Dr. Isaacs noted.
The association between enterovirus infection and islet autoimmunity was greater in individuals who later progressed to type 1 diabetes, with odds ratio 5.1 vs. 2.0 for those who didn’t. The association was most evident at or shortly after seroconversion (5.1), was stronger in Europe (3.2) than in other regions (1.9), and was stronger among those with a first-degree relative with type 1 diabetes (9.8) than those recruited via a high-risk human leukocyte antigen (HLA), in whom the relationship wasn’t significant.
Having multiple or consecutive enteroviral infections was also associated with islet autoimmunity (2.0).
With type 1 diabetes, the relationship with enterovirus was greater in children (9.0) than in adults (4.1), and was greater for type 1 diabetes onset within 1 year (13.8) and within 1 month (16.2) than for those with established type 1 diabetes (7.0). Here, too, the relationship was stronger in Europe (10.2) than outside Europe (7.5).
The link with type 1 diabetes and enterovirus was particularly strong for those with both a first-degree relative and a high-risk HLA (141.4).
The relationship with type 1 diabetes was significant for enterovirus species A (3.7), B (12.7) and C (13.8), including coxsackie virus genotypes, but not D.
“Future studies should focus on characterizing enterovirus genomes in at-risk cohorts rather than just the presence or absence of the virus,” Dr. Isaacs said.
However, she added, “type 1 diabetes is such a heterogenous condition, viruses may be implicated more in one type than another. It’s important that we start to look into this.”
Dr. Isaacs reports no relevant financial relationships. Dr. Khunti disclosed ties with AstraZeneca, Novartis, Novo Nordisk, Sanofi-Aventis, Lilly, Merck Sharp & Dohme, Boehringer Ingelheim, Bayer, Berlin-Chemie AG / Menarini Group, Janssen, and Napp.
A version of this article first appeared on Medscape.com.
STOCKHOLM – Enterovirus infection appears to be strongly linked to both type 1 diabetes and islet cell autoantibodies, new research suggests.
The strength of the relationship, particularly within the first month of type 1 diabetes diagnosis, “further supports the rationale for development of enterovirus-targeted vaccines and antiviral therapy to prevent and reduce the impact of type 1 diabetes,” according to lead investigator Sonia Isaacs, MD, of the department of pediatrics and child health at the University of New South Wales, Sydney, Australia.
Enteroviruses are a large family of viruses responsible for many infections in children. These live in the intestinal tract but can cause a wide variety of illnesses. There are more than 70 different strains, which include the group A and group B coxsackieviruses, the polioviruses, hepatitis A virus, and several strains that just go by the name enterovirus.
Dr. Isaacs presented the data, from a meta-analysis of studies using modern molecular techniques, at the annual meeting of the European Association for the Study of Diabetes.
The findings raise the question of whether people should be routinely tested for enterovirus at the time of type 1 diabetes diagnosis, she said during her presentation.
Asked by this news organization about the implications for first-degree relatives of people with type 1 diabetes, Dr. Isaacs said that they are “definitely a population to watch out for,” with regard to enteroviral infections. “Type 1 diabetes is very diverse and has different endotypes. Different environmental factors may be implicated in these different endotypes, and it may be that the enteroviruses are quite important in the first-degree relative group.”
Asked to comment, session moderator Kamlesh Khunti, MD, PhD, told this news organization that the data were “compelling,” particularly in the short term after type 1 diabetes diagnosis. “It seems that there may be plausibility for enterovirus associated with the development of type 1 diabetes ... Are there methods by which we can reduce this risk with either antivirals or vaccinations? I think that needs to be tested.”
And in regard to first-degree relatives, “I think that’s the group to go for because the association is so highly correlated. I think that’s the group worth testing with any interventions,” said Dr. Khunti, professor of primary care diabetes and vascular medicine at the University of Leicester, England.
Link stronger a month after diagnosis, in close relatives, in Europe
The new meta-analysis is an update to a prior review published in 2011 by Dr. Isaacs’ group, which found that people with islet cell autoimmunity were more than four times as likely as were controls to have an enterovirus infection, and people with type 1 diabetes were almost 10 times as likely.
This new analysis focuses on studies using more modern molecular techniques for detecting viruses, including high throughput sequencing and single-cell technologies.
The analysis identified 60 studies with a total of 12,077 participants, of whom 900 had islet autoimmunity, 5,081 had type 1 diabetes, and 6,096 were controls. Thirty-five of the studies were from Europe, while others were from the United States, Asia, and the Middle East.
Of 16 studies examining enterovirus infection in islet autoimmunity, cases with islet autoimmunity were twice as likely to have an enterovirus infection at any time point compared to controls, a significant difference (odds ratio [OR], 2.07, P = .002.)
Among 48 studies reporting enterovirus infection in type 1 diabetes, those with type 1 diabetes were eight times as likely to have an enterovirus infection compared with controls (OR, 8.0, P < .00001).
In 25 studies including 2,977 participants with onset of type 1 diabetes within the prior month, those individuals were more than 16 times more likely to present with an enterovirus infection (OR, 16.2, P < .00001).
“The strength of this is association is greater than previously reported by both us and others,” Dr. Isaacs noted.
The association between enterovirus infection and islet autoimmunity was greater in individuals who later progressed to type 1 diabetes, with odds ratio 5.1 vs. 2.0 for those who didn’t. The association was most evident at or shortly after seroconversion (5.1), was stronger in Europe (3.2) than in other regions (1.9), and was stronger among those with a first-degree relative with type 1 diabetes (9.8) than those recruited via a high-risk human leukocyte antigen (HLA), in whom the relationship wasn’t significant.
Having multiple or consecutive enteroviral infections was also associated with islet autoimmunity (2.0).
With type 1 diabetes, the relationship with enterovirus was greater in children (9.0) than in adults (4.1), and was greater for type 1 diabetes onset within 1 year (13.8) and within 1 month (16.2) than for those with established type 1 diabetes (7.0). Here, too, the relationship was stronger in Europe (10.2) than outside Europe (7.5).
The link with type 1 diabetes and enterovirus was particularly strong for those with both a first-degree relative and a high-risk HLA (141.4).
The relationship with type 1 diabetes was significant for enterovirus species A (3.7), B (12.7) and C (13.8), including coxsackie virus genotypes, but not D.
“Future studies should focus on characterizing enterovirus genomes in at-risk cohorts rather than just the presence or absence of the virus,” Dr. Isaacs said.
However, she added, “type 1 diabetes is such a heterogenous condition, viruses may be implicated more in one type than another. It’s important that we start to look into this.”
Dr. Isaacs reports no relevant financial relationships. Dr. Khunti disclosed ties with AstraZeneca, Novartis, Novo Nordisk, Sanofi-Aventis, Lilly, Merck Sharp & Dohme, Boehringer Ingelheim, Bayer, Berlin-Chemie AG / Menarini Group, Janssen, and Napp.
A version of this article first appeared on Medscape.com.
STOCKHOLM – Enterovirus infection appears to be strongly linked to both type 1 diabetes and islet cell autoantibodies, new research suggests.
The strength of the relationship, particularly within the first month of type 1 diabetes diagnosis, “further supports the rationale for development of enterovirus-targeted vaccines and antiviral therapy to prevent and reduce the impact of type 1 diabetes,” according to lead investigator Sonia Isaacs, MD, of the department of pediatrics and child health at the University of New South Wales, Sydney, Australia.
Enteroviruses are a large family of viruses responsible for many infections in children. These live in the intestinal tract but can cause a wide variety of illnesses. There are more than 70 different strains, which include the group A and group B coxsackieviruses, the polioviruses, hepatitis A virus, and several strains that just go by the name enterovirus.
Dr. Isaacs presented the data, from a meta-analysis of studies using modern molecular techniques, at the annual meeting of the European Association for the Study of Diabetes.
The findings raise the question of whether people should be routinely tested for enterovirus at the time of type 1 diabetes diagnosis, she said during her presentation.
Asked by this news organization about the implications for first-degree relatives of people with type 1 diabetes, Dr. Isaacs said that they are “definitely a population to watch out for,” with regard to enteroviral infections. “Type 1 diabetes is very diverse and has different endotypes. Different environmental factors may be implicated in these different endotypes, and it may be that the enteroviruses are quite important in the first-degree relative group.”
Asked to comment, session moderator Kamlesh Khunti, MD, PhD, told this news organization that the data were “compelling,” particularly in the short term after type 1 diabetes diagnosis. “It seems that there may be plausibility for enterovirus associated with the development of type 1 diabetes ... Are there methods by which we can reduce this risk with either antivirals or vaccinations? I think that needs to be tested.”
And in regard to first-degree relatives, “I think that’s the group to go for because the association is so highly correlated. I think that’s the group worth testing with any interventions,” said Dr. Khunti, professor of primary care diabetes and vascular medicine at the University of Leicester, England.
Link stronger a month after diagnosis, in close relatives, in Europe
The new meta-analysis is an update to a prior review published in 2011 by Dr. Isaacs’ group, which found that people with islet cell autoimmunity were more than four times as likely as were controls to have an enterovirus infection, and people with type 1 diabetes were almost 10 times as likely.
This new analysis focuses on studies using more modern molecular techniques for detecting viruses, including high throughput sequencing and single-cell technologies.
The analysis identified 60 studies with a total of 12,077 participants, of whom 900 had islet autoimmunity, 5,081 had type 1 diabetes, and 6,096 were controls. Thirty-five of the studies were from Europe, while others were from the United States, Asia, and the Middle East.
Of 16 studies examining enterovirus infection in islet autoimmunity, cases with islet autoimmunity were twice as likely to have an enterovirus infection at any time point compared to controls, a significant difference (odds ratio [OR], 2.07, P = .002.)
Among 48 studies reporting enterovirus infection in type 1 diabetes, those with type 1 diabetes were eight times as likely to have an enterovirus infection compared with controls (OR, 8.0, P < .00001).
In 25 studies including 2,977 participants with onset of type 1 diabetes within the prior month, those individuals were more than 16 times more likely to present with an enterovirus infection (OR, 16.2, P < .00001).
“The strength of this is association is greater than previously reported by both us and others,” Dr. Isaacs noted.
The association between enterovirus infection and islet autoimmunity was greater in individuals who later progressed to type 1 diabetes, with odds ratio 5.1 vs. 2.0 for those who didn’t. The association was most evident at or shortly after seroconversion (5.1), was stronger in Europe (3.2) than in other regions (1.9), and was stronger among those with a first-degree relative with type 1 diabetes (9.8) than those recruited via a high-risk human leukocyte antigen (HLA), in whom the relationship wasn’t significant.
Having multiple or consecutive enteroviral infections was also associated with islet autoimmunity (2.0).
With type 1 diabetes, the relationship with enterovirus was greater in children (9.0) than in adults (4.1), and was greater for type 1 diabetes onset within 1 year (13.8) and within 1 month (16.2) than for those with established type 1 diabetes (7.0). Here, too, the relationship was stronger in Europe (10.2) than outside Europe (7.5).
The link with type 1 diabetes and enterovirus was particularly strong for those with both a first-degree relative and a high-risk HLA (141.4).
The relationship with type 1 diabetes was significant for enterovirus species A (3.7), B (12.7) and C (13.8), including coxsackie virus genotypes, but not D.
“Future studies should focus on characterizing enterovirus genomes in at-risk cohorts rather than just the presence or absence of the virus,” Dr. Isaacs said.
However, she added, “type 1 diabetes is such a heterogenous condition, viruses may be implicated more in one type than another. It’s important that we start to look into this.”
Dr. Isaacs reports no relevant financial relationships. Dr. Khunti disclosed ties with AstraZeneca, Novartis, Novo Nordisk, Sanofi-Aventis, Lilly, Merck Sharp & Dohme, Boehringer Ingelheim, Bayer, Berlin-Chemie AG / Menarini Group, Janssen, and Napp.
A version of this article first appeared on Medscape.com.
AT EASD 2022
Cancer as a full contact sport
John worked as a handyman and lived on a small sailboat in a marina. When he was diagnosed with metastatic kidney cancer at age 48, he quickly fell through the cracks. He failed to show to appointments and took oral anticancer treatments, but just sporadically. He had Medicaid, so insurance wasn’t the issue. It was everything else.
John was behind on his slip fees; he hadn’t been able to work for some time because of his progressive weakness and pain. He was chronically in danger of getting kicked out of his makeshift home aboard the boat. He had no reliable transportation to the clinic and so he didn’t come to appointments regularly. The specialty pharmacy refused to deliver his expensive oral chemotherapy to his address at the marina. He went days without eating full meals because he was too weak to cook for himself. Plus, he was estranged from his family who were unaware of his illness. His oncologist was overwhelmed trying to take care of him. He had a reasonable chance of achieving disease control on first-line oral therapy, but his problems seemed to hinder these chances at every turn. She was distraught – what could she do?
Enter the team approach. John’s oncologist reached out to our palliative care program for help. We recognized that this was a job too big for us alone so we connected John with the Extensivist Medicine program at UCLA Health, a high-intensity primary care program led by a physician specializing in primary care for high-risk individuals. The program provides wraparound outpatient services for chronically and seriously ill patients, like John, who are at risk for falling through the cracks. John went from receiving very little support to now having an entire team of caring professionals focused on helping him achieve his best possible outcome despite the seriousness of his disease.
He now had the support of a high-functioning team with clearly defined roles. Social work connected him with housing, food, and transportation resources. A nurse called him every day to check in and make sure he was taking medications and reminded him about his upcoming appointments. Case management helped him get needed equipment, such as grab bars and a walker. As his palliative care nurse practitioner, I counseled him on understanding his prognosis and planning ahead for medical emergencies. Our psycho-oncology clinicians helped John reconcile with his family, who were more than willing to take him in once they realized how ill he was. Once these social factors were addressed, John could more easily stay current with his oral chemotherapy, giving him the best chance possible to achieve a robust treatment response that could buy him more time.
And, John did get that time – he got 6 months of improved quality of life, during which he reconnected with his family, including his children, and rebuilt these important relationships. Eventually treatment failed him. His disease, already widely metastatic, became more active and painful. He accepted hospice care at his sister’s house and we transitioned him from our team to the hospice team. He died peacefully surrounded by family.
Interprofessional teamwork is fundamental to treat ‘total pain’
None of this would have been possible without the work of high-functioning teams. It is a commonly held belief that interprofessional teamwork is fundamental to the care of patients and families living with serious illness. But why? How did this idea come about? And what evidence is there to support teamwork?
Dame Cicely Saunders, who founded the modern hospice movement in mid-20th century England, embodied the interdisciplinary team by working first as a nurse, then a social worker, and finally as a physician. She wrote about patients’ “total pain,” the crisis of physical, spiritual, social, and emotional distress that many people have at the end of life. She understood that no single health care discipline was adequate to the task of addressing each of these domains equally well. Thus, hospice became synonymous with care provided by a quartet of specialists – physicians, nurses, social workers, and chaplains. Nowadays, there are other specialists that are added to the mix – home health aides, pharmacists, physical and occupational therapists, music and pet therapists, and so on.
But in medicine, like all areas of science, convention and tradition only go so far. What evidence is there to support the work of an interdisciplinary team in managing the distress of patients and families living with advanced illnesses? It turns out that there is good evidence to support the use of high-functioning interdisciplinary teams in the care of the seriously ill. Palliative care is associated with improved patient outcomes, including improvements in symptom control, quality of life, and end of life care, when it is delivered by an interdisciplinary team rather than by a solo practitioner.
You may think that teamwork is most useful for patients like John who have seemingly intractable social barriers. But it is also true that for even patients with many more social advantages teamwork improves quality of life. I got to see this up close recently in my own life.
Teamwork improves quality of life
My father recently passed away after a 9-month battle with advanced cancer. He had every advantage possible – financial stability, high health literacy, an incredibly devoted spouse who happens to be an RN, good insurance, and access to top-notch medical care. Yet, even he benefited from a team approach. It started small, with the oncologist and oncology NP providing excellent, patient-centered care. Then it grew to include myself as the daughter/palliative care nurse practitioner who made recommendations for treating his nausea and ensured that his advance directive was completed and uploaded to his chart. When my dad needed physical therapy, the home health agency sent a wonderful physical therapist, who brought all sorts of equipment that kept him more functional than he would have been otherwise. Other family members helped out – my sisters helped connect my dad with a priest who came to the home to provide spiritual care, which was crucial to ensuring that he was at peace. And, in his final days, my dad had the hospice team to help manage his symptoms and his family members to provide hands-on care.
The complexity of cancer care has long necessitated a team approach to planning cancer treatment – known as a tumor board – with medical oncology, radiation oncology, surgery, and pathology all weighing in. It makes sense that patients and their families would also need a team of clinicians representing different specialty areas to assist with the wide array of physical, psychosocial, practical, and spiritual concerns that arise throughout the cancer disease trajectory.
Ms. D’Ambruoso is a hospice and palliative care nurse practitioner for UCLA Health Cancer Care, Santa Monica, Calif.
John worked as a handyman and lived on a small sailboat in a marina. When he was diagnosed with metastatic kidney cancer at age 48, he quickly fell through the cracks. He failed to show to appointments and took oral anticancer treatments, but just sporadically. He had Medicaid, so insurance wasn’t the issue. It was everything else.
John was behind on his slip fees; he hadn’t been able to work for some time because of his progressive weakness and pain. He was chronically in danger of getting kicked out of his makeshift home aboard the boat. He had no reliable transportation to the clinic and so he didn’t come to appointments regularly. The specialty pharmacy refused to deliver his expensive oral chemotherapy to his address at the marina. He went days without eating full meals because he was too weak to cook for himself. Plus, he was estranged from his family who were unaware of his illness. His oncologist was overwhelmed trying to take care of him. He had a reasonable chance of achieving disease control on first-line oral therapy, but his problems seemed to hinder these chances at every turn. She was distraught – what could she do?
Enter the team approach. John’s oncologist reached out to our palliative care program for help. We recognized that this was a job too big for us alone so we connected John with the Extensivist Medicine program at UCLA Health, a high-intensity primary care program led by a physician specializing in primary care for high-risk individuals. The program provides wraparound outpatient services for chronically and seriously ill patients, like John, who are at risk for falling through the cracks. John went from receiving very little support to now having an entire team of caring professionals focused on helping him achieve his best possible outcome despite the seriousness of his disease.
He now had the support of a high-functioning team with clearly defined roles. Social work connected him with housing, food, and transportation resources. A nurse called him every day to check in and make sure he was taking medications and reminded him about his upcoming appointments. Case management helped him get needed equipment, such as grab bars and a walker. As his palliative care nurse practitioner, I counseled him on understanding his prognosis and planning ahead for medical emergencies. Our psycho-oncology clinicians helped John reconcile with his family, who were more than willing to take him in once they realized how ill he was. Once these social factors were addressed, John could more easily stay current with his oral chemotherapy, giving him the best chance possible to achieve a robust treatment response that could buy him more time.
And, John did get that time – he got 6 months of improved quality of life, during which he reconnected with his family, including his children, and rebuilt these important relationships. Eventually treatment failed him. His disease, already widely metastatic, became more active and painful. He accepted hospice care at his sister’s house and we transitioned him from our team to the hospice team. He died peacefully surrounded by family.
Interprofessional teamwork is fundamental to treat ‘total pain’
None of this would have been possible without the work of high-functioning teams. It is a commonly held belief that interprofessional teamwork is fundamental to the care of patients and families living with serious illness. But why? How did this idea come about? And what evidence is there to support teamwork?
Dame Cicely Saunders, who founded the modern hospice movement in mid-20th century England, embodied the interdisciplinary team by working first as a nurse, then a social worker, and finally as a physician. She wrote about patients’ “total pain,” the crisis of physical, spiritual, social, and emotional distress that many people have at the end of life. She understood that no single health care discipline was adequate to the task of addressing each of these domains equally well. Thus, hospice became synonymous with care provided by a quartet of specialists – physicians, nurses, social workers, and chaplains. Nowadays, there are other specialists that are added to the mix – home health aides, pharmacists, physical and occupational therapists, music and pet therapists, and so on.
But in medicine, like all areas of science, convention and tradition only go so far. What evidence is there to support the work of an interdisciplinary team in managing the distress of patients and families living with advanced illnesses? It turns out that there is good evidence to support the use of high-functioning interdisciplinary teams in the care of the seriously ill. Palliative care is associated with improved patient outcomes, including improvements in symptom control, quality of life, and end of life care, when it is delivered by an interdisciplinary team rather than by a solo practitioner.
You may think that teamwork is most useful for patients like John who have seemingly intractable social barriers. But it is also true that for even patients with many more social advantages teamwork improves quality of life. I got to see this up close recently in my own life.
Teamwork improves quality of life
My father recently passed away after a 9-month battle with advanced cancer. He had every advantage possible – financial stability, high health literacy, an incredibly devoted spouse who happens to be an RN, good insurance, and access to top-notch medical care. Yet, even he benefited from a team approach. It started small, with the oncologist and oncology NP providing excellent, patient-centered care. Then it grew to include myself as the daughter/palliative care nurse practitioner who made recommendations for treating his nausea and ensured that his advance directive was completed and uploaded to his chart. When my dad needed physical therapy, the home health agency sent a wonderful physical therapist, who brought all sorts of equipment that kept him more functional than he would have been otherwise. Other family members helped out – my sisters helped connect my dad with a priest who came to the home to provide spiritual care, which was crucial to ensuring that he was at peace. And, in his final days, my dad had the hospice team to help manage his symptoms and his family members to provide hands-on care.
The complexity of cancer care has long necessitated a team approach to planning cancer treatment – known as a tumor board – with medical oncology, radiation oncology, surgery, and pathology all weighing in. It makes sense that patients and their families would also need a team of clinicians representing different specialty areas to assist with the wide array of physical, psychosocial, practical, and spiritual concerns that arise throughout the cancer disease trajectory.
Ms. D’Ambruoso is a hospice and palliative care nurse practitioner for UCLA Health Cancer Care, Santa Monica, Calif.
John worked as a handyman and lived on a small sailboat in a marina. When he was diagnosed with metastatic kidney cancer at age 48, he quickly fell through the cracks. He failed to show to appointments and took oral anticancer treatments, but just sporadically. He had Medicaid, so insurance wasn’t the issue. It was everything else.
John was behind on his slip fees; he hadn’t been able to work for some time because of his progressive weakness and pain. He was chronically in danger of getting kicked out of his makeshift home aboard the boat. He had no reliable transportation to the clinic and so he didn’t come to appointments regularly. The specialty pharmacy refused to deliver his expensive oral chemotherapy to his address at the marina. He went days without eating full meals because he was too weak to cook for himself. Plus, he was estranged from his family who were unaware of his illness. His oncologist was overwhelmed trying to take care of him. He had a reasonable chance of achieving disease control on first-line oral therapy, but his problems seemed to hinder these chances at every turn. She was distraught – what could she do?
Enter the team approach. John’s oncologist reached out to our palliative care program for help. We recognized that this was a job too big for us alone so we connected John with the Extensivist Medicine program at UCLA Health, a high-intensity primary care program led by a physician specializing in primary care for high-risk individuals. The program provides wraparound outpatient services for chronically and seriously ill patients, like John, who are at risk for falling through the cracks. John went from receiving very little support to now having an entire team of caring professionals focused on helping him achieve his best possible outcome despite the seriousness of his disease.
He now had the support of a high-functioning team with clearly defined roles. Social work connected him with housing, food, and transportation resources. A nurse called him every day to check in and make sure he was taking medications and reminded him about his upcoming appointments. Case management helped him get needed equipment, such as grab bars and a walker. As his palliative care nurse practitioner, I counseled him on understanding his prognosis and planning ahead for medical emergencies. Our psycho-oncology clinicians helped John reconcile with his family, who were more than willing to take him in once they realized how ill he was. Once these social factors were addressed, John could more easily stay current with his oral chemotherapy, giving him the best chance possible to achieve a robust treatment response that could buy him more time.
And, John did get that time – he got 6 months of improved quality of life, during which he reconnected with his family, including his children, and rebuilt these important relationships. Eventually treatment failed him. His disease, already widely metastatic, became more active and painful. He accepted hospice care at his sister’s house and we transitioned him from our team to the hospice team. He died peacefully surrounded by family.
Interprofessional teamwork is fundamental to treat ‘total pain’
None of this would have been possible without the work of high-functioning teams. It is a commonly held belief that interprofessional teamwork is fundamental to the care of patients and families living with serious illness. But why? How did this idea come about? And what evidence is there to support teamwork?
Dame Cicely Saunders, who founded the modern hospice movement in mid-20th century England, embodied the interdisciplinary team by working first as a nurse, then a social worker, and finally as a physician. She wrote about patients’ “total pain,” the crisis of physical, spiritual, social, and emotional distress that many people have at the end of life. She understood that no single health care discipline was adequate to the task of addressing each of these domains equally well. Thus, hospice became synonymous with care provided by a quartet of specialists – physicians, nurses, social workers, and chaplains. Nowadays, there are other specialists that are added to the mix – home health aides, pharmacists, physical and occupational therapists, music and pet therapists, and so on.
But in medicine, like all areas of science, convention and tradition only go so far. What evidence is there to support the work of an interdisciplinary team in managing the distress of patients and families living with advanced illnesses? It turns out that there is good evidence to support the use of high-functioning interdisciplinary teams in the care of the seriously ill. Palliative care is associated with improved patient outcomes, including improvements in symptom control, quality of life, and end of life care, when it is delivered by an interdisciplinary team rather than by a solo practitioner.
You may think that teamwork is most useful for patients like John who have seemingly intractable social barriers. But it is also true that for even patients with many more social advantages teamwork improves quality of life. I got to see this up close recently in my own life.
Teamwork improves quality of life
My father recently passed away after a 9-month battle with advanced cancer. He had every advantage possible – financial stability, high health literacy, an incredibly devoted spouse who happens to be an RN, good insurance, and access to top-notch medical care. Yet, even he benefited from a team approach. It started small, with the oncologist and oncology NP providing excellent, patient-centered care. Then it grew to include myself as the daughter/palliative care nurse practitioner who made recommendations for treating his nausea and ensured that his advance directive was completed and uploaded to his chart. When my dad needed physical therapy, the home health agency sent a wonderful physical therapist, who brought all sorts of equipment that kept him more functional than he would have been otherwise. Other family members helped out – my sisters helped connect my dad with a priest who came to the home to provide spiritual care, which was crucial to ensuring that he was at peace. And, in his final days, my dad had the hospice team to help manage his symptoms and his family members to provide hands-on care.
The complexity of cancer care has long necessitated a team approach to planning cancer treatment – known as a tumor board – with medical oncology, radiation oncology, surgery, and pathology all weighing in. It makes sense that patients and their families would also need a team of clinicians representing different specialty areas to assist with the wide array of physical, psychosocial, practical, and spiritual concerns that arise throughout the cancer disease trajectory.
Ms. D’Ambruoso is a hospice and palliative care nurse practitioner for UCLA Health Cancer Care, Santa Monica, Calif.
CAR T-cell therapy neurotoxicity linked to NfL elevations
“This is the first study to show NfL levels are elevated even before CAR T treatment is given,” first author Omar H. Butt, MD, PhD, of the Siteman Cancer Center at Barnes-Jewish Hospital and Washington University in St. Louis, said in an interview.
“While unlikely to be the sole driver of [the neurotoxicity], neural injury reflected by NfL may aid in identifying a high-risk subset of patients undergoing cellular therapy,” the authors concluded in the study, published in JAMA Oncology.
CAR T-cell therapy has gained favor for virtually revolutionizing the treatment of some leukemias and lymphomas, however, as many as 40%-60% of patients develop the neurotoxicity side effect, called immune effector cell–associated neurotoxicity syndrome (ICANS), which, though usually low grade, in more severe cases can cause substantial morbidity and even mortality.
Hence, “the early identification of patients at risk for ICANS is critical for preemptive management,” the authors noted.
NfL, an established marker of neuroaxonal injury in neurodegenerative diseases including multiple sclerosis and Alzheimer’s disease, has been shown in previous studies to be elevated following the development of ICANS and up to 5 days prior to its peak symptoms.
To further evaluate NfL elevations in relation to ICANS, Dr. Butt and colleagues identified 30 patients undergoing CD19 CART-cell therapy, including 77% for diffuse large B-cell lymphoma, at two U.S. centers: Washington University in St. Louis and Case Western Reserve University, Cleveland.
The patients had a median age of 64 and were 40% female.
Among them, four developed low-grade ICANS grade 1-2, and 7 developed ICANS grade 3 or higher.
Of those developing any-grade ICANS, baseline elevations of NfL prior to the CAR T-cell treatment, were significantly higher, compared with those who did not develop ICANs (mean 87.6 pg/mL vs. 29.4 pg/mL, P < .001), with no significant differences between the low-grade (1 and 2) and higher-grade (3 or higher) ICANS groups.
A receiver operating characteristic analysis showed baseline NfL levels significantly predicted the development of ICANS with high accuracy (area under the ROC curve, 0.96), as well as sensitivity (AUROC, 0.91) and specificity (AUROC, 0.95).
Notably, baseline NfL levels were associated with ICANS severity, but did not correlate with other factors including demographic, oncologic history, nononcologic neurologic history, or history of exposure to neurotoxic therapies.
However, Dr. Butt added, “it is important to note that our study was insufficiently powered to examine those relationships in earnest. Therefore, [a correlation between NfL and those factors] remains possible,” he said.
The elevated NfL levels observed prior to the development of ICANS remained high across the study’s seven time points, up to day 30 post infusion.
Interest in NfL levels on the rise
NfL assessment is currently only clinically validated in amyotrophic lateral sclerosis, where it is used to assess neuroaxonal health and integrity. However, testing is available as interest and evidence of NfL’s potential role in other settings grows.
Meanwhile, Dr. Butt and associates are themselves developing an assay to predict the development of ICANS, which will likely include NfL, if the role is validated in further studies.
“Future studies will explore validating NfL for ICANS and additional indications,” he said.
ICANS symptoms can range from headaches and confusion to seizures or strokes in more severe cases.
The current gold standard for treatment includes early intervention with high-dose steroids and careful monitoring, but there is reluctance to use such therapies because of concerns about their blunting the anticancer effects of the CAR T cells.
Importantly, if validated, elevations in NfL could signal the need for more precautionary measures with CAR T-cell therapy, Dr. Butt noted.
“Our data suggests patients with high NfL levels at baseline would benefit most from perhaps closer monitoring with frequent checks and possible early intervention at the first sign of symptoms, a period of time when it may be hard to distinguish ICANS from other causes of confusion, such as delirium,” he explained.
Limitations: Validation, preventive measures needed
Commenting on the study, Sattva S. Neelapu, MD, a professor and deputy chair of the department of lymphoma and myeloma at the University of Texas MD Anderson Cancer Center, Houston, agreed that the findings have potentially important implications.
“I think this is a very intriguing and novel finding that needs to be investigated further prospectively in a larger cohort and across different CAR T products in patients with lymphoma, leukemia, and myeloma,” Dr. Neelapu said in an interview.
The NfL elevations observed even before CAR T-cell therapy among those who went on to develop ICANS are notable, he added.
“This is the surprising finding in the study,” Dr. Neelapu said. “It raises the question whether neurologic injury is caused by prior therapies that these patients received or whether it is an age-related phenomenon, as we do see higher incidence and severity of ICANS in older patients or some other mechanisms.”
A key caveat, however, is that even if a risk is identified, options to prevent ICANS are currently limited, Dr. Neelapu noted.
“I think it is too early to implement this into clinical practice,” he said. In addition to needing further validation, “assessing NfL levels would be useful when there is an effective prophylactic or therapeutic strategy – both of which also need to be investigated.”
Dr. Butt and colleagues are developing a clinical assay for ICANS and reported a provisional patent pending on the use of plasma NfL as a predictive biomarker for ICANS. The study received support from the Washington University in St. Louis, the Paula and Rodger O. Riney Fund, the Daniel J. Brennan MD Fund, the Fred Simmons and Olga Mohan Fund; the National Cancer Institute, the National Multiple Sclerosis Society, and the National Institute of Neurological Disorders and Stroke. Dr. Neelapu reported conflicts of interest with numerous pharmaceutical companies.
“This is the first study to show NfL levels are elevated even before CAR T treatment is given,” first author Omar H. Butt, MD, PhD, of the Siteman Cancer Center at Barnes-Jewish Hospital and Washington University in St. Louis, said in an interview.
“While unlikely to be the sole driver of [the neurotoxicity], neural injury reflected by NfL may aid in identifying a high-risk subset of patients undergoing cellular therapy,” the authors concluded in the study, published in JAMA Oncology.
CAR T-cell therapy has gained favor for virtually revolutionizing the treatment of some leukemias and lymphomas, however, as many as 40%-60% of patients develop the neurotoxicity side effect, called immune effector cell–associated neurotoxicity syndrome (ICANS), which, though usually low grade, in more severe cases can cause substantial morbidity and even mortality.
Hence, “the early identification of patients at risk for ICANS is critical for preemptive management,” the authors noted.
NfL, an established marker of neuroaxonal injury in neurodegenerative diseases including multiple sclerosis and Alzheimer’s disease, has been shown in previous studies to be elevated following the development of ICANS and up to 5 days prior to its peak symptoms.
To further evaluate NfL elevations in relation to ICANS, Dr. Butt and colleagues identified 30 patients undergoing CD19 CART-cell therapy, including 77% for diffuse large B-cell lymphoma, at two U.S. centers: Washington University in St. Louis and Case Western Reserve University, Cleveland.
The patients had a median age of 64 and were 40% female.
Among them, four developed low-grade ICANS grade 1-2, and 7 developed ICANS grade 3 or higher.
Of those developing any-grade ICANS, baseline elevations of NfL prior to the CAR T-cell treatment, were significantly higher, compared with those who did not develop ICANs (mean 87.6 pg/mL vs. 29.4 pg/mL, P < .001), with no significant differences between the low-grade (1 and 2) and higher-grade (3 or higher) ICANS groups.
A receiver operating characteristic analysis showed baseline NfL levels significantly predicted the development of ICANS with high accuracy (area under the ROC curve, 0.96), as well as sensitivity (AUROC, 0.91) and specificity (AUROC, 0.95).
Notably, baseline NfL levels were associated with ICANS severity, but did not correlate with other factors including demographic, oncologic history, nononcologic neurologic history, or history of exposure to neurotoxic therapies.
However, Dr. Butt added, “it is important to note that our study was insufficiently powered to examine those relationships in earnest. Therefore, [a correlation between NfL and those factors] remains possible,” he said.
The elevated NfL levels observed prior to the development of ICANS remained high across the study’s seven time points, up to day 30 post infusion.
Interest in NfL levels on the rise
NfL assessment is currently only clinically validated in amyotrophic lateral sclerosis, where it is used to assess neuroaxonal health and integrity. However, testing is available as interest and evidence of NfL’s potential role in other settings grows.
Meanwhile, Dr. Butt and associates are themselves developing an assay to predict the development of ICANS, which will likely include NfL, if the role is validated in further studies.
“Future studies will explore validating NfL for ICANS and additional indications,” he said.
ICANS symptoms can range from headaches and confusion to seizures or strokes in more severe cases.
The current gold standard for treatment includes early intervention with high-dose steroids and careful monitoring, but there is reluctance to use such therapies because of concerns about their blunting the anticancer effects of the CAR T cells.
Importantly, if validated, elevations in NfL could signal the need for more precautionary measures with CAR T-cell therapy, Dr. Butt noted.
“Our data suggests patients with high NfL levels at baseline would benefit most from perhaps closer monitoring with frequent checks and possible early intervention at the first sign of symptoms, a period of time when it may be hard to distinguish ICANS from other causes of confusion, such as delirium,” he explained.
Limitations: Validation, preventive measures needed
Commenting on the study, Sattva S. Neelapu, MD, a professor and deputy chair of the department of lymphoma and myeloma at the University of Texas MD Anderson Cancer Center, Houston, agreed that the findings have potentially important implications.
“I think this is a very intriguing and novel finding that needs to be investigated further prospectively in a larger cohort and across different CAR T products in patients with lymphoma, leukemia, and myeloma,” Dr. Neelapu said in an interview.
The NfL elevations observed even before CAR T-cell therapy among those who went on to develop ICANS are notable, he added.
“This is the surprising finding in the study,” Dr. Neelapu said. “It raises the question whether neurologic injury is caused by prior therapies that these patients received or whether it is an age-related phenomenon, as we do see higher incidence and severity of ICANS in older patients or some other mechanisms.”
A key caveat, however, is that even if a risk is identified, options to prevent ICANS are currently limited, Dr. Neelapu noted.
“I think it is too early to implement this into clinical practice,” he said. In addition to needing further validation, “assessing NfL levels would be useful when there is an effective prophylactic or therapeutic strategy – both of which also need to be investigated.”
Dr. Butt and colleagues are developing a clinical assay for ICANS and reported a provisional patent pending on the use of plasma NfL as a predictive biomarker for ICANS. The study received support from the Washington University in St. Louis, the Paula and Rodger O. Riney Fund, the Daniel J. Brennan MD Fund, the Fred Simmons and Olga Mohan Fund; the National Cancer Institute, the National Multiple Sclerosis Society, and the National Institute of Neurological Disorders and Stroke. Dr. Neelapu reported conflicts of interest with numerous pharmaceutical companies.
“This is the first study to show NfL levels are elevated even before CAR T treatment is given,” first author Omar H. Butt, MD, PhD, of the Siteman Cancer Center at Barnes-Jewish Hospital and Washington University in St. Louis, said in an interview.
“While unlikely to be the sole driver of [the neurotoxicity], neural injury reflected by NfL may aid in identifying a high-risk subset of patients undergoing cellular therapy,” the authors concluded in the study, published in JAMA Oncology.
CAR T-cell therapy has gained favor for virtually revolutionizing the treatment of some leukemias and lymphomas, however, as many as 40%-60% of patients develop the neurotoxicity side effect, called immune effector cell–associated neurotoxicity syndrome (ICANS), which, though usually low grade, in more severe cases can cause substantial morbidity and even mortality.
Hence, “the early identification of patients at risk for ICANS is critical for preemptive management,” the authors noted.
NfL, an established marker of neuroaxonal injury in neurodegenerative diseases including multiple sclerosis and Alzheimer’s disease, has been shown in previous studies to be elevated following the development of ICANS and up to 5 days prior to its peak symptoms.
To further evaluate NfL elevations in relation to ICANS, Dr. Butt and colleagues identified 30 patients undergoing CD19 CART-cell therapy, including 77% for diffuse large B-cell lymphoma, at two U.S. centers: Washington University in St. Louis and Case Western Reserve University, Cleveland.
The patients had a median age of 64 and were 40% female.
Among them, four developed low-grade ICANS grade 1-2, and 7 developed ICANS grade 3 or higher.
Of those developing any-grade ICANS, baseline elevations of NfL prior to the CAR T-cell treatment, were significantly higher, compared with those who did not develop ICANs (mean 87.6 pg/mL vs. 29.4 pg/mL, P < .001), with no significant differences between the low-grade (1 and 2) and higher-grade (3 or higher) ICANS groups.
A receiver operating characteristic analysis showed baseline NfL levels significantly predicted the development of ICANS with high accuracy (area under the ROC curve, 0.96), as well as sensitivity (AUROC, 0.91) and specificity (AUROC, 0.95).
Notably, baseline NfL levels were associated with ICANS severity, but did not correlate with other factors including demographic, oncologic history, nononcologic neurologic history, or history of exposure to neurotoxic therapies.
However, Dr. Butt added, “it is important to note that our study was insufficiently powered to examine those relationships in earnest. Therefore, [a correlation between NfL and those factors] remains possible,” he said.
The elevated NfL levels observed prior to the development of ICANS remained high across the study’s seven time points, up to day 30 post infusion.
Interest in NfL levels on the rise
NfL assessment is currently only clinically validated in amyotrophic lateral sclerosis, where it is used to assess neuroaxonal health and integrity. However, testing is available as interest and evidence of NfL’s potential role in other settings grows.
Meanwhile, Dr. Butt and associates are themselves developing an assay to predict the development of ICANS, which will likely include NfL, if the role is validated in further studies.
“Future studies will explore validating NfL for ICANS and additional indications,” he said.
ICANS symptoms can range from headaches and confusion to seizures or strokes in more severe cases.
The current gold standard for treatment includes early intervention with high-dose steroids and careful monitoring, but there is reluctance to use such therapies because of concerns about their blunting the anticancer effects of the CAR T cells.
Importantly, if validated, elevations in NfL could signal the need for more precautionary measures with CAR T-cell therapy, Dr. Butt noted.
“Our data suggests patients with high NfL levels at baseline would benefit most from perhaps closer monitoring with frequent checks and possible early intervention at the first sign of symptoms, a period of time when it may be hard to distinguish ICANS from other causes of confusion, such as delirium,” he explained.
Limitations: Validation, preventive measures needed
Commenting on the study, Sattva S. Neelapu, MD, a professor and deputy chair of the department of lymphoma and myeloma at the University of Texas MD Anderson Cancer Center, Houston, agreed that the findings have potentially important implications.
“I think this is a very intriguing and novel finding that needs to be investigated further prospectively in a larger cohort and across different CAR T products in patients with lymphoma, leukemia, and myeloma,” Dr. Neelapu said in an interview.
The NfL elevations observed even before CAR T-cell therapy among those who went on to develop ICANS are notable, he added.
“This is the surprising finding in the study,” Dr. Neelapu said. “It raises the question whether neurologic injury is caused by prior therapies that these patients received or whether it is an age-related phenomenon, as we do see higher incidence and severity of ICANS in older patients or some other mechanisms.”
A key caveat, however, is that even if a risk is identified, options to prevent ICANS are currently limited, Dr. Neelapu noted.
“I think it is too early to implement this into clinical practice,” he said. In addition to needing further validation, “assessing NfL levels would be useful when there is an effective prophylactic or therapeutic strategy – both of which also need to be investigated.”
Dr. Butt and colleagues are developing a clinical assay for ICANS and reported a provisional patent pending on the use of plasma NfL as a predictive biomarker for ICANS. The study received support from the Washington University in St. Louis, the Paula and Rodger O. Riney Fund, the Daniel J. Brennan MD Fund, the Fred Simmons and Olga Mohan Fund; the National Cancer Institute, the National Multiple Sclerosis Society, and the National Institute of Neurological Disorders and Stroke. Dr. Neelapu reported conflicts of interest with numerous pharmaceutical companies.
FROM JAMA ONCOLOGY