User login
For MD-IQ use only
Red blood cells made to deliver antibiotics to bacteria
Over several years, “we developed experimental and computational techniques to study how proteins and drugs interact with membranes,” Hannah Krivić, a graduate student, and Maikel C. Rheinstädter, PhD, a professor of physics, both at McMaster University in Hamilton, Ont., told this news organization.
In earlier work, these researchers investigated how antibiotics target bacterial membranes and how those membranes enable the development of antibiotic resistance. Then, they said, “we started to ... manipulate membranes by tuning their properties [with] synthetic lipid molecules to create ‘hybrid’ membranes – that is, functionalized biological membranes with optimized properties.
“We are now using this approach to functionalize red blood cells by using them as drug carriers. We optimize these cells to carry certain loads, such as drug molecules, and anchor proteins in their membranes that target receptors in bacteria to selectively and efficiently deliver that load.”
The strategy, they said, “has become a universal red blood cell–based delivery platform that we call ‘smart blood’ ... that can safely and selectively deliver antibiotics to certain bacterial targets.”
The platform currently is being tested in vitro, and in vivo testing is slated to begin in early 2023. Their study was published online in ACS Infectious Diseases.
Optimizing dosing
Polymyxin B (PmB) is one of a few potent antibiotics with promising efficacy against drug-resistant bacteria such as E. coli. PmB, however, is widely considered a treatment of last resort because of its toxic side effects (which include nephrotoxicity, neurotoxicity, and neuromuscular blockade) particularly at higher doses.
The researchers hypothesized that targeted delivery of PmB might lead to optimized dosing and potentially reduce the need for higher or repeated doses. In the current study, they tested the ability of the smart blood platform to deliver PmB to E. coli.
Creating “erythro-PmBs” involves removing the inner components of red blood cells, loading the cells with PmB, and coating the cell membranes (liposomes) with antibacterial (in this case, anti–E. coli) antibodies.
The in vitro experiments showed that the cells could be loaded with PmB and retain and selectively deliver the drug to E. coli, with no apparent hemolytic activity or nephrotoxicity. Specifically, the erythro-PmBs had a loading efficiency of approximately 90% and delivered PmB to E. coli with values for the minimum inhibitory concentration that were comparable with those of free PmB.
“In contrast to drug-delivery systems based on synthetic carriers, our erythrocytes have a high biocompatibility and can stay in circulation in the body for several weeks to provide a sustained and targeted release of the drug,” said Ms. Krivić and Dr. Rheinstädter. “This [profile] can make existing drugs safer by, for instance, increasing their efficacy while at the same time lowering the required dosage, thereby reducing side effects.”
The researchers are now exploring the ability of the smart blood platform to deliver neurotrophic factors across the blood-brain barrier to potentially treat neurodegenerative diseases. Their approach is identical to that used in the current study, they said, except in this case, the red blood cell membranes are designed to deliver neurotrophic factors specifically to the blood-brain barrier.
‘Certainly promising’
David W. Deamer, PhD, research professor of biomolecular engineering at the University of California, Santa Cruz, said in a comment: “This is certainly promising. The erythro-PmBs have a greater loading capacity and longer circulation time than ordinary liposomes used for drug delivery. They can also be prepared with specific antibodies so that the antibiotic is delivered more directly when they bind to bacterial pathogens.”
The effect on bacterial growth, however, was tested in a model system, not in an actual infection, he said, adding that an important next step will be to perform animal testing. “One of the simplest tests is induced sepsis in mice, which mimics a burst appendix. If the erythro-PmBs can treat sepsis effectively, it will be an encouraging sign that they have potential therapeutic value. It will also be interesting to see whether the antigens responsible for ABO blood groups are retained on the surfaces of the erythro-PmBs. If they are, it might be necessary to match donor blood to that of the recipient.
“Getting a product ready for clinical use will require partnership with a major pharmaceutical firm, several years of animal testing, and then several more years carrying out phase 1, 2, and 3 clinical trials in human patients,” Dr. Deamer concluded.
No commercial funding was disclosed. Ms. Krivić, Dr. Rheinstädter, and Dr. Deamer reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Over several years, “we developed experimental and computational techniques to study how proteins and drugs interact with membranes,” Hannah Krivić, a graduate student, and Maikel C. Rheinstädter, PhD, a professor of physics, both at McMaster University in Hamilton, Ont., told this news organization.
In earlier work, these researchers investigated how antibiotics target bacterial membranes and how those membranes enable the development of antibiotic resistance. Then, they said, “we started to ... manipulate membranes by tuning their properties [with] synthetic lipid molecules to create ‘hybrid’ membranes – that is, functionalized biological membranes with optimized properties.
“We are now using this approach to functionalize red blood cells by using them as drug carriers. We optimize these cells to carry certain loads, such as drug molecules, and anchor proteins in their membranes that target receptors in bacteria to selectively and efficiently deliver that load.”
The strategy, they said, “has become a universal red blood cell–based delivery platform that we call ‘smart blood’ ... that can safely and selectively deliver antibiotics to certain bacterial targets.”
The platform currently is being tested in vitro, and in vivo testing is slated to begin in early 2023. Their study was published online in ACS Infectious Diseases.
Optimizing dosing
Polymyxin B (PmB) is one of a few potent antibiotics with promising efficacy against drug-resistant bacteria such as E. coli. PmB, however, is widely considered a treatment of last resort because of its toxic side effects (which include nephrotoxicity, neurotoxicity, and neuromuscular blockade) particularly at higher doses.
The researchers hypothesized that targeted delivery of PmB might lead to optimized dosing and potentially reduce the need for higher or repeated doses. In the current study, they tested the ability of the smart blood platform to deliver PmB to E. coli.
Creating “erythro-PmBs” involves removing the inner components of red blood cells, loading the cells with PmB, and coating the cell membranes (liposomes) with antibacterial (in this case, anti–E. coli) antibodies.
The in vitro experiments showed that the cells could be loaded with PmB and retain and selectively deliver the drug to E. coli, with no apparent hemolytic activity or nephrotoxicity. Specifically, the erythro-PmBs had a loading efficiency of approximately 90% and delivered PmB to E. coli with values for the minimum inhibitory concentration that were comparable with those of free PmB.
“In contrast to drug-delivery systems based on synthetic carriers, our erythrocytes have a high biocompatibility and can stay in circulation in the body for several weeks to provide a sustained and targeted release of the drug,” said Ms. Krivić and Dr. Rheinstädter. “This [profile] can make existing drugs safer by, for instance, increasing their efficacy while at the same time lowering the required dosage, thereby reducing side effects.”
The researchers are now exploring the ability of the smart blood platform to deliver neurotrophic factors across the blood-brain barrier to potentially treat neurodegenerative diseases. Their approach is identical to that used in the current study, they said, except in this case, the red blood cell membranes are designed to deliver neurotrophic factors specifically to the blood-brain barrier.
‘Certainly promising’
David W. Deamer, PhD, research professor of biomolecular engineering at the University of California, Santa Cruz, said in a comment: “This is certainly promising. The erythro-PmBs have a greater loading capacity and longer circulation time than ordinary liposomes used for drug delivery. They can also be prepared with specific antibodies so that the antibiotic is delivered more directly when they bind to bacterial pathogens.”
The effect on bacterial growth, however, was tested in a model system, not in an actual infection, he said, adding that an important next step will be to perform animal testing. “One of the simplest tests is induced sepsis in mice, which mimics a burst appendix. If the erythro-PmBs can treat sepsis effectively, it will be an encouraging sign that they have potential therapeutic value. It will also be interesting to see whether the antigens responsible for ABO blood groups are retained on the surfaces of the erythro-PmBs. If they are, it might be necessary to match donor blood to that of the recipient.
“Getting a product ready for clinical use will require partnership with a major pharmaceutical firm, several years of animal testing, and then several more years carrying out phase 1, 2, and 3 clinical trials in human patients,” Dr. Deamer concluded.
No commercial funding was disclosed. Ms. Krivić, Dr. Rheinstädter, and Dr. Deamer reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Over several years, “we developed experimental and computational techniques to study how proteins and drugs interact with membranes,” Hannah Krivić, a graduate student, and Maikel C. Rheinstädter, PhD, a professor of physics, both at McMaster University in Hamilton, Ont., told this news organization.
In earlier work, these researchers investigated how antibiotics target bacterial membranes and how those membranes enable the development of antibiotic resistance. Then, they said, “we started to ... manipulate membranes by tuning their properties [with] synthetic lipid molecules to create ‘hybrid’ membranes – that is, functionalized biological membranes with optimized properties.
“We are now using this approach to functionalize red blood cells by using them as drug carriers. We optimize these cells to carry certain loads, such as drug molecules, and anchor proteins in their membranes that target receptors in bacteria to selectively and efficiently deliver that load.”
The strategy, they said, “has become a universal red blood cell–based delivery platform that we call ‘smart blood’ ... that can safely and selectively deliver antibiotics to certain bacterial targets.”
The platform currently is being tested in vitro, and in vivo testing is slated to begin in early 2023. Their study was published online in ACS Infectious Diseases.
Optimizing dosing
Polymyxin B (PmB) is one of a few potent antibiotics with promising efficacy against drug-resistant bacteria such as E. coli. PmB, however, is widely considered a treatment of last resort because of its toxic side effects (which include nephrotoxicity, neurotoxicity, and neuromuscular blockade) particularly at higher doses.
The researchers hypothesized that targeted delivery of PmB might lead to optimized dosing and potentially reduce the need for higher or repeated doses. In the current study, they tested the ability of the smart blood platform to deliver PmB to E. coli.
Creating “erythro-PmBs” involves removing the inner components of red blood cells, loading the cells with PmB, and coating the cell membranes (liposomes) with antibacterial (in this case, anti–E. coli) antibodies.
The in vitro experiments showed that the cells could be loaded with PmB and retain and selectively deliver the drug to E. coli, with no apparent hemolytic activity or nephrotoxicity. Specifically, the erythro-PmBs had a loading efficiency of approximately 90% and delivered PmB to E. coli with values for the minimum inhibitory concentration that were comparable with those of free PmB.
“In contrast to drug-delivery systems based on synthetic carriers, our erythrocytes have a high biocompatibility and can stay in circulation in the body for several weeks to provide a sustained and targeted release of the drug,” said Ms. Krivić and Dr. Rheinstädter. “This [profile] can make existing drugs safer by, for instance, increasing their efficacy while at the same time lowering the required dosage, thereby reducing side effects.”
The researchers are now exploring the ability of the smart blood platform to deliver neurotrophic factors across the blood-brain barrier to potentially treat neurodegenerative diseases. Their approach is identical to that used in the current study, they said, except in this case, the red blood cell membranes are designed to deliver neurotrophic factors specifically to the blood-brain barrier.
‘Certainly promising’
David W. Deamer, PhD, research professor of biomolecular engineering at the University of California, Santa Cruz, said in a comment: “This is certainly promising. The erythro-PmBs have a greater loading capacity and longer circulation time than ordinary liposomes used for drug delivery. They can also be prepared with specific antibodies so that the antibiotic is delivered more directly when they bind to bacterial pathogens.”
The effect on bacterial growth, however, was tested in a model system, not in an actual infection, he said, adding that an important next step will be to perform animal testing. “One of the simplest tests is induced sepsis in mice, which mimics a burst appendix. If the erythro-PmBs can treat sepsis effectively, it will be an encouraging sign that they have potential therapeutic value. It will also be interesting to see whether the antigens responsible for ABO blood groups are retained on the surfaces of the erythro-PmBs. If they are, it might be necessary to match donor blood to that of the recipient.
“Getting a product ready for clinical use will require partnership with a major pharmaceutical firm, several years of animal testing, and then several more years carrying out phase 1, 2, and 3 clinical trials in human patients,” Dr. Deamer concluded.
No commercial funding was disclosed. Ms. Krivić, Dr. Rheinstädter, and Dr. Deamer reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM ACS INFECTIOUS DISEASES
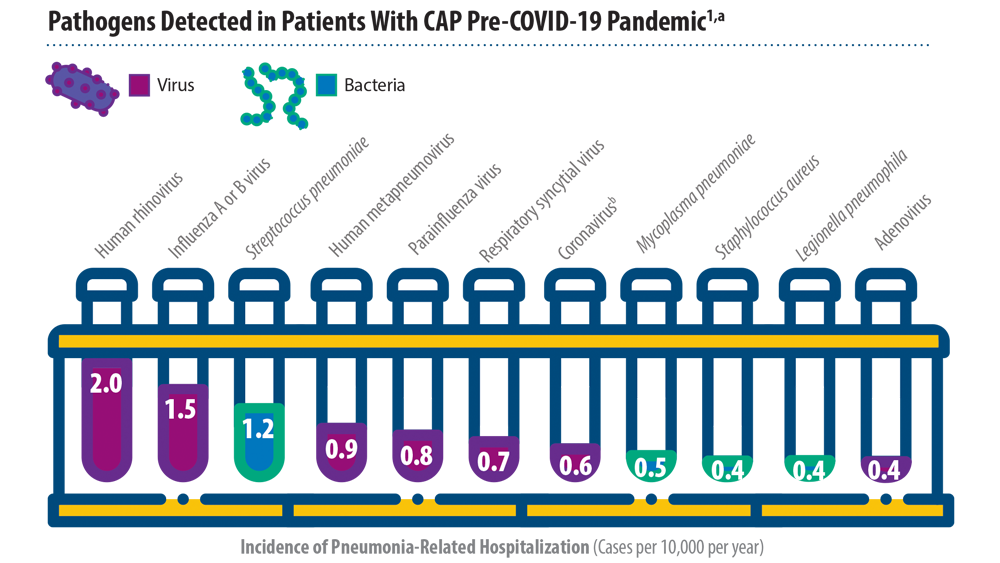
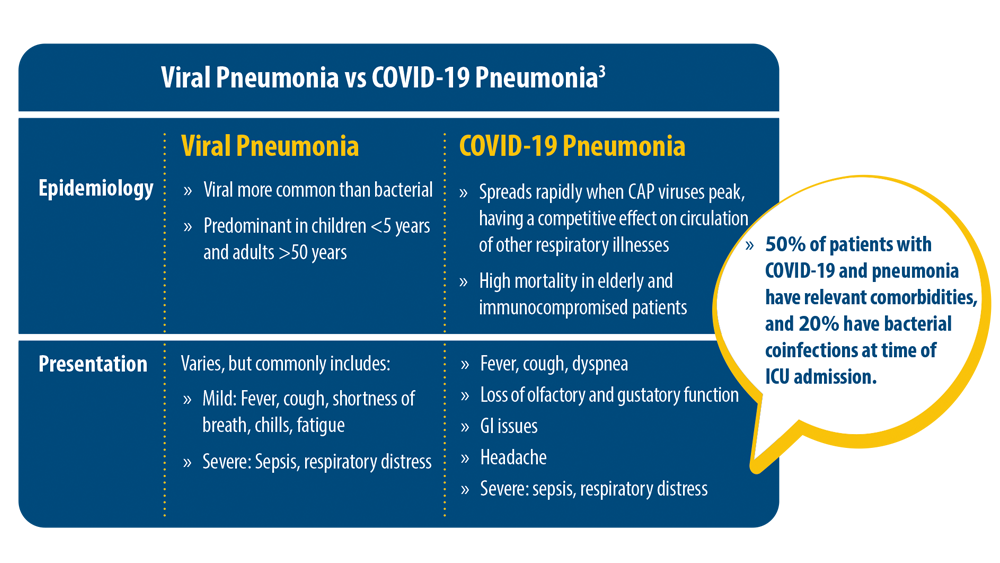
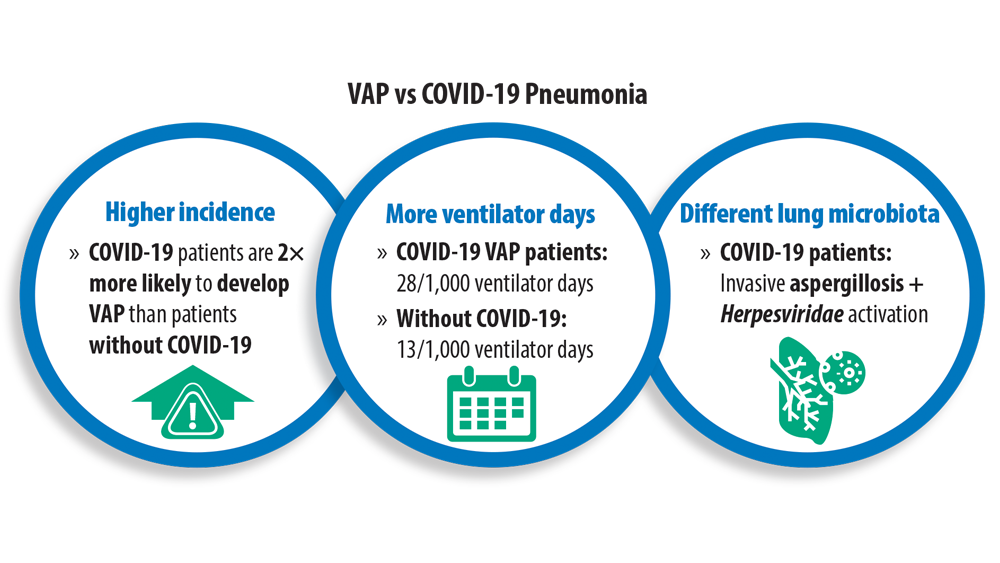
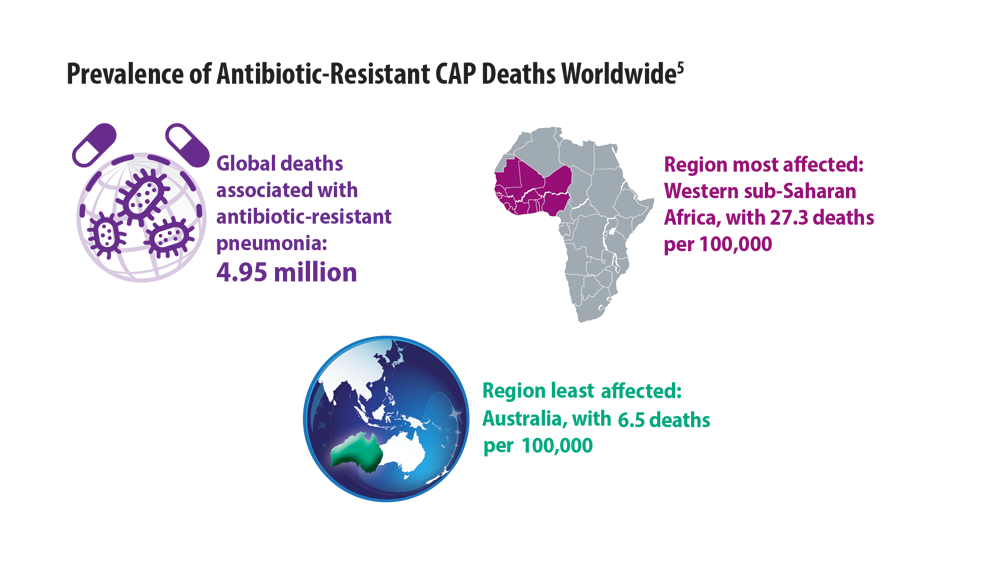
New Pathogens, COVID-19, and Antibiotic Resistance in the Field of Pneumonia
- Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among US adults. N Engl J Med. 2015;373(5):415-427. doi:10.1056/NEJMoa1500245
- Aliberti S, Dela Cruz CS, Amati F, Sotgiu G, Restrepo MI. Community-acquired pneumonia. Lancet. 2021;398(10303):906-919. doi:10.1016/S0140-6736(21)00630-9
- Pagliano P, Sellitto C, Conti V, Ascione T, Esposito S. Characteristics of viral pneumonia in the COVID-19 era: an update. Infection. 2021;49(4):607-616. doi:10.1007/s15010-021-01603-y
- Maes M, Higginson E, Pereira-Dias J, et al. Ventilator-associated pneumonia in critically ill patients with COVID-19 [published correction appears in Crit Care. 2021 Apr 6;25(1):130]. Crit Care. 2021;25(1):25. doi:10.1186/s13054-021-03460-5
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629-655. doi:10.1016/S0140- 6736(21)02724-0
- Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among US adults. N Engl J Med. 2015;373(5):415-427. doi:10.1056/NEJMoa1500245
- Aliberti S, Dela Cruz CS, Amati F, Sotgiu G, Restrepo MI. Community-acquired pneumonia. Lancet. 2021;398(10303):906-919. doi:10.1016/S0140-6736(21)00630-9
- Pagliano P, Sellitto C, Conti V, Ascione T, Esposito S. Characteristics of viral pneumonia in the COVID-19 era: an update. Infection. 2021;49(4):607-616. doi:10.1007/s15010-021-01603-y
- Maes M, Higginson E, Pereira-Dias J, et al. Ventilator-associated pneumonia in critically ill patients with COVID-19 [published correction appears in Crit Care. 2021 Apr 6;25(1):130]. Crit Care. 2021;25(1):25. doi:10.1186/s13054-021-03460-5
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629-655. doi:10.1016/S0140- 6736(21)02724-0
- Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among US adults. N Engl J Med. 2015;373(5):415-427. doi:10.1056/NEJMoa1500245
- Aliberti S, Dela Cruz CS, Amati F, Sotgiu G, Restrepo MI. Community-acquired pneumonia. Lancet. 2021;398(10303):906-919. doi:10.1016/S0140-6736(21)00630-9
- Pagliano P, Sellitto C, Conti V, Ascione T, Esposito S. Characteristics of viral pneumonia in the COVID-19 era: an update. Infection. 2021;49(4):607-616. doi:10.1007/s15010-021-01603-y
- Maes M, Higginson E, Pereira-Dias J, et al. Ventilator-associated pneumonia in critically ill patients with COVID-19 [published correction appears in Crit Care. 2021 Apr 6;25(1):130]. Crit Care. 2021;25(1):25. doi:10.1186/s13054-021-03460-5
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629-655. doi:10.1016/S0140- 6736(21)02724-0
Avoid routine early ECMO in severe cardiogenic shock: ECMO-CS
CHICAGO – Routine early, expeditious use of extracorporeal membrane oxygenation (ECMO) is a common strategy in patients with severe cardiogenic shock, but a less aggressive initial approach may be just as effective, a randomized trial suggests.
In the study that assigned patients with “rapidly deteriorating or severe” cardiogenic shock to one or the other approach, clinical outcomes were no better for those who received immediate ECMO than for those initially managed with inotropes and vasopressors, researchers said.
The conservative strategy, importantly, allowed for downstream ECMO in the event of hemodynamic deterioration, which occurred in a substantial 39% of cases, observed Petr Ostadal, MD, PhD, when presenting the results at the American Heart Association scientific sessions.
Dr. Ostadal of Na Homolce Hospital, Prague, is also first author on the published report of the study, called Extracorporeal Membrane Oxygenation in the Therapy of Cardiogenic Shock (ECMO-CS), which was published the same day in Circulation.
The trial makes a firm case for preferring the conservative initial approach over routine early ECMO in the kind of patients it entered, Larry A. Allen, MD, MHS, University of Coloradoat Denver, Aurora, told this news organization.
More than 60% of the trial’s 117 patients had shock secondary to an acute coronary syndrome; another 23% were in heart failure decompensation.
A preference for the conservative initial approach would be welcome, he said. The early aggressive ECMO approach is resource intensive and carries some important risks, such as stroke or coagulopathy, said Dr. Allen, who is not connected with ECMO-CS. Yet it is increasingly the go-to approach in such patients, based primarily on observational data.
Although early ECMO apparently didn’t benefit patients in this study in their specific stage of cardiogenic shock, Dr. Allen observed, it would presumably help some, but identifying them in practice presents challenges. “Defining where people are in the spectrum of early versus middle versus late cardiogenic shock is actually very tricky.”
It will therefore be important, he said, to identify ways to predict which conservatively managed patients do well with the strategy, and which are most at risk for hemodynamic deterioration and for whom ECMO should be readily available.
“I think part of what ECMO-CS tells us is that, if a patient is stable on intravenous inotropic and vasopressor support, you can defer ECMO while you’re thinking about the patient – about their larger context and the right medical decision-making for them.”
The trial randomly assigned 122 patients with rapidly deteriorating or severe cardiogenic shock to the immediate-ECMO or the conservative strategy at four centers in the Czech Republic. The 117 patients for whom informed consent could be obtained were included in the analysis, 58 and 59 patients, respectively. Their mean age was about 65 years and three-fourths were male.
The primary endpoint, the only endpoint for which the study was powered, consisted of death from any cause, resuscitated circulatory arrest, or use of a different form of mechanical circulatory support (MCS) by 30 days.
It occurred in 63.8% of patients assigned to immediate ECMO and 71.2% of those in the conservative strategy group, for a hazard ratio of 0.72 (95% confidence interval, 0.46-1.12; P = .21).
As individual endpoints, rates of death from any cause and resuscitated arrest did not significantly differ between the groups, but conservatively managed patients more often used another form of MCS. The HRs were 1.11 (95% CI, 0.66-1.87) for death from any cause, 0.79 (95% CI, 0.27-2.28) for resuscitated cardiac arrest, and 0.38 (95% CI, 0.18-0.79) for use of another MCS device.
The rates for serious adverse events – including bleeding, ischemia, stroke, pneumonia, or sepsis – were similar at 60.3% in the early-ECMO group and 61% in group with conservative initial management, Dr. Ostadal reported.
Other than the 23 patients in the conservative initial strategy group who went on to receive ECMO (1.9 days after randomization, on average), 1 went on to undergo implantation with a HeartMate (Abbott) ventricular assist device and 3 received an Impella pump (Abiomed).
Six patients in the early-ECMO group were already receiving intra-aortic balloon pump (IABP) support at randomization, two underwent temporary implantation with a Centrimag device (Abbott), and three went on to receive a HeartMate device, the published report notes.
ECMO is the optimal first choice for MCS in such patients with cardiogenic shock who need a circulatory support device, especially because it also oxygenates the blood, Dr. Ostadal told this news organization.
But ECMO doesn’t help with ventricular unloading. Indeed, it can sometimes reduce ventricular preload, especially if right-heart pressures are low. So MCS devices that unload the ventricle, typically an IABP, can complement ECMO.
Dr. Ostadal speculates, however, that there may be a better pairing option. “Impella plus ECMO, I think, is the combination which has a future,” he said, for patients in cardiogenic shock who need a short-term percutaneous hemodynamic support device. Impella “supports the whole circulation” and unloads the left ventricle.
“A balloon pump in combination with ECMO is still not a bad choice. It’s very cheap in comparison with Impella.” But in his opinion, Dr. Ostadal said, “The combination of Impella plus ECMO is more efficient from a hemodynamic point of view.”
As the published report notes, ongoing randomized trials looking at ECMO plus other MCS devices in cardiogenic shock include ECLS-SHOCK, with a projected enrollment of 420 patients, and EURO-SHOCK, aiming for a similar number of patients; both compare routine ECMO to conservative management.
In addition, ANCHOR, in which ECMO is combined with IABP, and DanShock, which looks at early use of Impella rather than ECMO, are enrolling patients with shock secondary to acute coronary syndromes.
Dr. Ostadal disclosed consulting for Getinge, Edwards, Medtronic, Biomedica, and Xenios/Fresenius, and receiving research support from Xenios/Fresenius. Dr. Allen disclosed modest or significant relationships with ACI Clinical, Novartis, UpToDate, Boston Scientific, and Cytokinetics.
A version of this article first appeared on Medscape.com.
CHICAGO – Routine early, expeditious use of extracorporeal membrane oxygenation (ECMO) is a common strategy in patients with severe cardiogenic shock, but a less aggressive initial approach may be just as effective, a randomized trial suggests.
In the study that assigned patients with “rapidly deteriorating or severe” cardiogenic shock to one or the other approach, clinical outcomes were no better for those who received immediate ECMO than for those initially managed with inotropes and vasopressors, researchers said.
The conservative strategy, importantly, allowed for downstream ECMO in the event of hemodynamic deterioration, which occurred in a substantial 39% of cases, observed Petr Ostadal, MD, PhD, when presenting the results at the American Heart Association scientific sessions.
Dr. Ostadal of Na Homolce Hospital, Prague, is also first author on the published report of the study, called Extracorporeal Membrane Oxygenation in the Therapy of Cardiogenic Shock (ECMO-CS), which was published the same day in Circulation.
The trial makes a firm case for preferring the conservative initial approach over routine early ECMO in the kind of patients it entered, Larry A. Allen, MD, MHS, University of Coloradoat Denver, Aurora, told this news organization.
More than 60% of the trial’s 117 patients had shock secondary to an acute coronary syndrome; another 23% were in heart failure decompensation.
A preference for the conservative initial approach would be welcome, he said. The early aggressive ECMO approach is resource intensive and carries some important risks, such as stroke or coagulopathy, said Dr. Allen, who is not connected with ECMO-CS. Yet it is increasingly the go-to approach in such patients, based primarily on observational data.
Although early ECMO apparently didn’t benefit patients in this study in their specific stage of cardiogenic shock, Dr. Allen observed, it would presumably help some, but identifying them in practice presents challenges. “Defining where people are in the spectrum of early versus middle versus late cardiogenic shock is actually very tricky.”
It will therefore be important, he said, to identify ways to predict which conservatively managed patients do well with the strategy, and which are most at risk for hemodynamic deterioration and for whom ECMO should be readily available.
“I think part of what ECMO-CS tells us is that, if a patient is stable on intravenous inotropic and vasopressor support, you can defer ECMO while you’re thinking about the patient – about their larger context and the right medical decision-making for them.”
The trial randomly assigned 122 patients with rapidly deteriorating or severe cardiogenic shock to the immediate-ECMO or the conservative strategy at four centers in the Czech Republic. The 117 patients for whom informed consent could be obtained were included in the analysis, 58 and 59 patients, respectively. Their mean age was about 65 years and three-fourths were male.
The primary endpoint, the only endpoint for which the study was powered, consisted of death from any cause, resuscitated circulatory arrest, or use of a different form of mechanical circulatory support (MCS) by 30 days.
It occurred in 63.8% of patients assigned to immediate ECMO and 71.2% of those in the conservative strategy group, for a hazard ratio of 0.72 (95% confidence interval, 0.46-1.12; P = .21).
As individual endpoints, rates of death from any cause and resuscitated arrest did not significantly differ between the groups, but conservatively managed patients more often used another form of MCS. The HRs were 1.11 (95% CI, 0.66-1.87) for death from any cause, 0.79 (95% CI, 0.27-2.28) for resuscitated cardiac arrest, and 0.38 (95% CI, 0.18-0.79) for use of another MCS device.
The rates for serious adverse events – including bleeding, ischemia, stroke, pneumonia, or sepsis – were similar at 60.3% in the early-ECMO group and 61% in group with conservative initial management, Dr. Ostadal reported.
Other than the 23 patients in the conservative initial strategy group who went on to receive ECMO (1.9 days after randomization, on average), 1 went on to undergo implantation with a HeartMate (Abbott) ventricular assist device and 3 received an Impella pump (Abiomed).
Six patients in the early-ECMO group were already receiving intra-aortic balloon pump (IABP) support at randomization, two underwent temporary implantation with a Centrimag device (Abbott), and three went on to receive a HeartMate device, the published report notes.
ECMO is the optimal first choice for MCS in such patients with cardiogenic shock who need a circulatory support device, especially because it also oxygenates the blood, Dr. Ostadal told this news organization.
But ECMO doesn’t help with ventricular unloading. Indeed, it can sometimes reduce ventricular preload, especially if right-heart pressures are low. So MCS devices that unload the ventricle, typically an IABP, can complement ECMO.
Dr. Ostadal speculates, however, that there may be a better pairing option. “Impella plus ECMO, I think, is the combination which has a future,” he said, for patients in cardiogenic shock who need a short-term percutaneous hemodynamic support device. Impella “supports the whole circulation” and unloads the left ventricle.
“A balloon pump in combination with ECMO is still not a bad choice. It’s very cheap in comparison with Impella.” But in his opinion, Dr. Ostadal said, “The combination of Impella plus ECMO is more efficient from a hemodynamic point of view.”
As the published report notes, ongoing randomized trials looking at ECMO plus other MCS devices in cardiogenic shock include ECLS-SHOCK, with a projected enrollment of 420 patients, and EURO-SHOCK, aiming for a similar number of patients; both compare routine ECMO to conservative management.
In addition, ANCHOR, in which ECMO is combined with IABP, and DanShock, which looks at early use of Impella rather than ECMO, are enrolling patients with shock secondary to acute coronary syndromes.
Dr. Ostadal disclosed consulting for Getinge, Edwards, Medtronic, Biomedica, and Xenios/Fresenius, and receiving research support from Xenios/Fresenius. Dr. Allen disclosed modest or significant relationships with ACI Clinical, Novartis, UpToDate, Boston Scientific, and Cytokinetics.
A version of this article first appeared on Medscape.com.
CHICAGO – Routine early, expeditious use of extracorporeal membrane oxygenation (ECMO) is a common strategy in patients with severe cardiogenic shock, but a less aggressive initial approach may be just as effective, a randomized trial suggests.
In the study that assigned patients with “rapidly deteriorating or severe” cardiogenic shock to one or the other approach, clinical outcomes were no better for those who received immediate ECMO than for those initially managed with inotropes and vasopressors, researchers said.
The conservative strategy, importantly, allowed for downstream ECMO in the event of hemodynamic deterioration, which occurred in a substantial 39% of cases, observed Petr Ostadal, MD, PhD, when presenting the results at the American Heart Association scientific sessions.
Dr. Ostadal of Na Homolce Hospital, Prague, is also first author on the published report of the study, called Extracorporeal Membrane Oxygenation in the Therapy of Cardiogenic Shock (ECMO-CS), which was published the same day in Circulation.
The trial makes a firm case for preferring the conservative initial approach over routine early ECMO in the kind of patients it entered, Larry A. Allen, MD, MHS, University of Coloradoat Denver, Aurora, told this news organization.
More than 60% of the trial’s 117 patients had shock secondary to an acute coronary syndrome; another 23% were in heart failure decompensation.
A preference for the conservative initial approach would be welcome, he said. The early aggressive ECMO approach is resource intensive and carries some important risks, such as stroke or coagulopathy, said Dr. Allen, who is not connected with ECMO-CS. Yet it is increasingly the go-to approach in such patients, based primarily on observational data.
Although early ECMO apparently didn’t benefit patients in this study in their specific stage of cardiogenic shock, Dr. Allen observed, it would presumably help some, but identifying them in practice presents challenges. “Defining where people are in the spectrum of early versus middle versus late cardiogenic shock is actually very tricky.”
It will therefore be important, he said, to identify ways to predict which conservatively managed patients do well with the strategy, and which are most at risk for hemodynamic deterioration and for whom ECMO should be readily available.
“I think part of what ECMO-CS tells us is that, if a patient is stable on intravenous inotropic and vasopressor support, you can defer ECMO while you’re thinking about the patient – about their larger context and the right medical decision-making for them.”
The trial randomly assigned 122 patients with rapidly deteriorating or severe cardiogenic shock to the immediate-ECMO or the conservative strategy at four centers in the Czech Republic. The 117 patients for whom informed consent could be obtained were included in the analysis, 58 and 59 patients, respectively. Their mean age was about 65 years and three-fourths were male.
The primary endpoint, the only endpoint for which the study was powered, consisted of death from any cause, resuscitated circulatory arrest, or use of a different form of mechanical circulatory support (MCS) by 30 days.
It occurred in 63.8% of patients assigned to immediate ECMO and 71.2% of those in the conservative strategy group, for a hazard ratio of 0.72 (95% confidence interval, 0.46-1.12; P = .21).
As individual endpoints, rates of death from any cause and resuscitated arrest did not significantly differ between the groups, but conservatively managed patients more often used another form of MCS. The HRs were 1.11 (95% CI, 0.66-1.87) for death from any cause, 0.79 (95% CI, 0.27-2.28) for resuscitated cardiac arrest, and 0.38 (95% CI, 0.18-0.79) for use of another MCS device.
The rates for serious adverse events – including bleeding, ischemia, stroke, pneumonia, or sepsis – were similar at 60.3% in the early-ECMO group and 61% in group with conservative initial management, Dr. Ostadal reported.
Other than the 23 patients in the conservative initial strategy group who went on to receive ECMO (1.9 days after randomization, on average), 1 went on to undergo implantation with a HeartMate (Abbott) ventricular assist device and 3 received an Impella pump (Abiomed).
Six patients in the early-ECMO group were already receiving intra-aortic balloon pump (IABP) support at randomization, two underwent temporary implantation with a Centrimag device (Abbott), and three went on to receive a HeartMate device, the published report notes.
ECMO is the optimal first choice for MCS in such patients with cardiogenic shock who need a circulatory support device, especially because it also oxygenates the blood, Dr. Ostadal told this news organization.
But ECMO doesn’t help with ventricular unloading. Indeed, it can sometimes reduce ventricular preload, especially if right-heart pressures are low. So MCS devices that unload the ventricle, typically an IABP, can complement ECMO.
Dr. Ostadal speculates, however, that there may be a better pairing option. “Impella plus ECMO, I think, is the combination which has a future,” he said, for patients in cardiogenic shock who need a short-term percutaneous hemodynamic support device. Impella “supports the whole circulation” and unloads the left ventricle.
“A balloon pump in combination with ECMO is still not a bad choice. It’s very cheap in comparison with Impella.” But in his opinion, Dr. Ostadal said, “The combination of Impella plus ECMO is more efficient from a hemodynamic point of view.”
As the published report notes, ongoing randomized trials looking at ECMO plus other MCS devices in cardiogenic shock include ECLS-SHOCK, with a projected enrollment of 420 patients, and EURO-SHOCK, aiming for a similar number of patients; both compare routine ECMO to conservative management.
In addition, ANCHOR, in which ECMO is combined with IABP, and DanShock, which looks at early use of Impella rather than ECMO, are enrolling patients with shock secondary to acute coronary syndromes.
Dr. Ostadal disclosed consulting for Getinge, Edwards, Medtronic, Biomedica, and Xenios/Fresenius, and receiving research support from Xenios/Fresenius. Dr. Allen disclosed modest or significant relationships with ACI Clinical, Novartis, UpToDate, Boston Scientific, and Cytokinetics.
A version of this article first appeared on Medscape.com.
AT AHA 2022
Study sheds new light on RAS inhibitors’ role for advanced CKD
ORLANDO – Treatment with a renin-angiotensin system (RAS) inhibitor is widely accepted as standard practice for slowing progression of chronic kidney disease (CKD), but data have been inconsistent as to whether there is benefit to continuing RAS inhibition when patients develop advanced CKD, defined as an estimated glomerular filtration rate (eGFR) of less than 30 mL/min per 1.73 m2.
Now, in STOP ACEi, a new multicenter, randomized trial of 411 patients, , for 3 years.
People who continued RAS inhibitor treatment did not develop a significant or clinically relevant decrease in eGFR, the study’s primary outcome, both overall as well as in several prespecified subgroups compared with those who discontinued treatment, said Sunil Bhandari, MBChB, PhD, and associates, who presented the research in a poster at the annual meeting of the American Society of Nephrology.
“I hope these results will reassure clinicians to continue ACE inhibitors or ARBs” in patients with advanced CKD, “with their known beneficial cardiovascular effects,” Dr. Bhandari said in an interview.
The results were simultaneously published in the New England Journal of Medicine.
Similar eGFR levels after 3 years
While it’s clear that in patients with mild or moderate CKD, treatment with a RAS inhibitor, which includes angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (ARBs), reduces blood pressure, slows decline in eGFR, reduces proteinuria, and delays progression to advanced CKD, there has been little evidence that the use of RAS inhibitors benefits patients with advanced CKD.
Data from previous trials have been inconsistent regarding whether the use of RAS inhibitors is nephroprotective in patients with advanced CKD, say Dr. Bhandari, a nephrologist and professor at Hull York Medical School, Hull, England, and colleagues.
“Current guidelines do not provide specific advice on whether to continue or stop ACE inhibitors or ARBs for advanced chronic kidney disease,” they also note.
And so they decided to assess whether discontinuation of ACE inhibitors/ARBs could slow progression of CKD in patients with advanced CKD.
Three years after 206 study participants stopped RAS inhibitor treatment, the least-squares mean eGFR was 12.6 mL/min per 1.73m2 in the discontinuation group and 13.3 mL/min per 1.73 m2 in the 205 patients in the continuation group, a difference that was not significant.
In addition to the primary outcome, 62% of patients who stopped RAS inhibitor treatment and 56% of those who continued developed end-stage kidney disease or required renal-replacement therapy, which translated into an adjusted hazard ratio of 1.28 for this outcome among those who discontinued compared with those who continued, which was just short of significance (95% CI, 0.99-1.65).
The two study groups also showed no significant differences in the 3-year incidence of hospitalization for any reason, cardiovascular events, or deaths. The two groups also showed no meaningful differences in various domains of quality of life and no differences in serious adverse effects.
Participants had an eGFR less than 30 mL/min per 1.73 m2
The study ran at 39 United Kingdom centers in 2014-2019. Investigators enrolled adults with an eGFR of less than 30 mL/min per 1.73 m2 who were not on dialysis and had not received a kidney transplant. In addition, all enrolled patients had to have an annual drop in eGFR of more than 2 mL/min per 1.73 m2 during the prior 2 years and had to have been on treatment with at least one RAS inhibitor for more than 6 months.
The randomization protocol insured balanced distribution of subjects between the two study arms by age, eGFR, presence of diabetes, and level of proteinuria, among other factors. The study design also mandated that participants maintain a blood pressure of no more than 140/85 mm Hg.
Those who discontinued RAS-inhibitor treatment could receive any guideline-recommended antihypertensive agent that was not a RAS inhibitor, although adding a RAS inhibitor was permitted as a last treatment resort.
People in the maintenance group could receive whichever additional antihypertensive agents their treating clinicians deemed necessary for maintaining the target blood pressure.
The enrolled population was a median age of 63 years old and 68% were men. Their average eGFR at baseline was 18 mL/min per 1.73 m2, and 118 (29%) had an eGFR of less than 15 mL/min per 1.73 m2. Their median level of proteinuria was 115 mg/mmol (about 1,018 mg/g). Diabetes was prevalent in 37%, and 58% of participants were taking at least three antihypertensive medications at entry.
Among the study’s limitations, the researchers cited the open-label design, which may have affected clinical care and the tally of subjective endpoints, including quality of life and exercise capacity. Also, because the study enrolled people who were on a RAS inhibitor at the time of randomization, it did not include anyone who had already discontinued these agents.
Continue RAS inhibitors in advanced CKD for best outcomes
Dr. Bhandari and colleagues note that in a large observational trial published in January 2021, Swedish researchers found an increase in the incidence of major cardiovascular events and death among patients with advanced CKD who had discontinued RAS inhibitors.
But they observe, “Our trial did not have sufficient power to investigate the effect of the discontinuation of RAS inhibitors on cardiovascular events or mortality. However, because our findings are consistent with a lack of advantage for such discontinuation with respect to kidney function, there is little rationale to conduct a larger randomized trial to investigate cardiovascular safety.”
“Our findings do not support the hypothesis that the discontinuation of RAS inhibitors in patients with advanced and progressive chronic kidney disease would improve kidney function, quality of life, or exercise capacity.”
“The results of this trial will inform future clinical practice worldwide and guideline recommendations,” they conclude.
STOP ACEi received no commercial funding. Dr. Bhandari has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ORLANDO – Treatment with a renin-angiotensin system (RAS) inhibitor is widely accepted as standard practice for slowing progression of chronic kidney disease (CKD), but data have been inconsistent as to whether there is benefit to continuing RAS inhibition when patients develop advanced CKD, defined as an estimated glomerular filtration rate (eGFR) of less than 30 mL/min per 1.73 m2.
Now, in STOP ACEi, a new multicenter, randomized trial of 411 patients, , for 3 years.
People who continued RAS inhibitor treatment did not develop a significant or clinically relevant decrease in eGFR, the study’s primary outcome, both overall as well as in several prespecified subgroups compared with those who discontinued treatment, said Sunil Bhandari, MBChB, PhD, and associates, who presented the research in a poster at the annual meeting of the American Society of Nephrology.
“I hope these results will reassure clinicians to continue ACE inhibitors or ARBs” in patients with advanced CKD, “with their known beneficial cardiovascular effects,” Dr. Bhandari said in an interview.
The results were simultaneously published in the New England Journal of Medicine.
Similar eGFR levels after 3 years
While it’s clear that in patients with mild or moderate CKD, treatment with a RAS inhibitor, which includes angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (ARBs), reduces blood pressure, slows decline in eGFR, reduces proteinuria, and delays progression to advanced CKD, there has been little evidence that the use of RAS inhibitors benefits patients with advanced CKD.
Data from previous trials have been inconsistent regarding whether the use of RAS inhibitors is nephroprotective in patients with advanced CKD, say Dr. Bhandari, a nephrologist and professor at Hull York Medical School, Hull, England, and colleagues.
“Current guidelines do not provide specific advice on whether to continue or stop ACE inhibitors or ARBs for advanced chronic kidney disease,” they also note.
And so they decided to assess whether discontinuation of ACE inhibitors/ARBs could slow progression of CKD in patients with advanced CKD.
Three years after 206 study participants stopped RAS inhibitor treatment, the least-squares mean eGFR was 12.6 mL/min per 1.73m2 in the discontinuation group and 13.3 mL/min per 1.73 m2 in the 205 patients in the continuation group, a difference that was not significant.
In addition to the primary outcome, 62% of patients who stopped RAS inhibitor treatment and 56% of those who continued developed end-stage kidney disease or required renal-replacement therapy, which translated into an adjusted hazard ratio of 1.28 for this outcome among those who discontinued compared with those who continued, which was just short of significance (95% CI, 0.99-1.65).
The two study groups also showed no significant differences in the 3-year incidence of hospitalization for any reason, cardiovascular events, or deaths. The two groups also showed no meaningful differences in various domains of quality of life and no differences in serious adverse effects.
Participants had an eGFR less than 30 mL/min per 1.73 m2
The study ran at 39 United Kingdom centers in 2014-2019. Investigators enrolled adults with an eGFR of less than 30 mL/min per 1.73 m2 who were not on dialysis and had not received a kidney transplant. In addition, all enrolled patients had to have an annual drop in eGFR of more than 2 mL/min per 1.73 m2 during the prior 2 years and had to have been on treatment with at least one RAS inhibitor for more than 6 months.
The randomization protocol insured balanced distribution of subjects between the two study arms by age, eGFR, presence of diabetes, and level of proteinuria, among other factors. The study design also mandated that participants maintain a blood pressure of no more than 140/85 mm Hg.
Those who discontinued RAS-inhibitor treatment could receive any guideline-recommended antihypertensive agent that was not a RAS inhibitor, although adding a RAS inhibitor was permitted as a last treatment resort.
People in the maintenance group could receive whichever additional antihypertensive agents their treating clinicians deemed necessary for maintaining the target blood pressure.
The enrolled population was a median age of 63 years old and 68% were men. Their average eGFR at baseline was 18 mL/min per 1.73 m2, and 118 (29%) had an eGFR of less than 15 mL/min per 1.73 m2. Their median level of proteinuria was 115 mg/mmol (about 1,018 mg/g). Diabetes was prevalent in 37%, and 58% of participants were taking at least three antihypertensive medications at entry.
Among the study’s limitations, the researchers cited the open-label design, which may have affected clinical care and the tally of subjective endpoints, including quality of life and exercise capacity. Also, because the study enrolled people who were on a RAS inhibitor at the time of randomization, it did not include anyone who had already discontinued these agents.
Continue RAS inhibitors in advanced CKD for best outcomes
Dr. Bhandari and colleagues note that in a large observational trial published in January 2021, Swedish researchers found an increase in the incidence of major cardiovascular events and death among patients with advanced CKD who had discontinued RAS inhibitors.
But they observe, “Our trial did not have sufficient power to investigate the effect of the discontinuation of RAS inhibitors on cardiovascular events or mortality. However, because our findings are consistent with a lack of advantage for such discontinuation with respect to kidney function, there is little rationale to conduct a larger randomized trial to investigate cardiovascular safety.”
“Our findings do not support the hypothesis that the discontinuation of RAS inhibitors in patients with advanced and progressive chronic kidney disease would improve kidney function, quality of life, or exercise capacity.”
“The results of this trial will inform future clinical practice worldwide and guideline recommendations,” they conclude.
STOP ACEi received no commercial funding. Dr. Bhandari has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ORLANDO – Treatment with a renin-angiotensin system (RAS) inhibitor is widely accepted as standard practice for slowing progression of chronic kidney disease (CKD), but data have been inconsistent as to whether there is benefit to continuing RAS inhibition when patients develop advanced CKD, defined as an estimated glomerular filtration rate (eGFR) of less than 30 mL/min per 1.73 m2.
Now, in STOP ACEi, a new multicenter, randomized trial of 411 patients, , for 3 years.
People who continued RAS inhibitor treatment did not develop a significant or clinically relevant decrease in eGFR, the study’s primary outcome, both overall as well as in several prespecified subgroups compared with those who discontinued treatment, said Sunil Bhandari, MBChB, PhD, and associates, who presented the research in a poster at the annual meeting of the American Society of Nephrology.
“I hope these results will reassure clinicians to continue ACE inhibitors or ARBs” in patients with advanced CKD, “with their known beneficial cardiovascular effects,” Dr. Bhandari said in an interview.
The results were simultaneously published in the New England Journal of Medicine.
Similar eGFR levels after 3 years
While it’s clear that in patients with mild or moderate CKD, treatment with a RAS inhibitor, which includes angiotensin-converting enzyme (ACE) inhibitors and angiotensin-receptor blockers (ARBs), reduces blood pressure, slows decline in eGFR, reduces proteinuria, and delays progression to advanced CKD, there has been little evidence that the use of RAS inhibitors benefits patients with advanced CKD.
Data from previous trials have been inconsistent regarding whether the use of RAS inhibitors is nephroprotective in patients with advanced CKD, say Dr. Bhandari, a nephrologist and professor at Hull York Medical School, Hull, England, and colleagues.
“Current guidelines do not provide specific advice on whether to continue or stop ACE inhibitors or ARBs for advanced chronic kidney disease,” they also note.
And so they decided to assess whether discontinuation of ACE inhibitors/ARBs could slow progression of CKD in patients with advanced CKD.
Three years after 206 study participants stopped RAS inhibitor treatment, the least-squares mean eGFR was 12.6 mL/min per 1.73m2 in the discontinuation group and 13.3 mL/min per 1.73 m2 in the 205 patients in the continuation group, a difference that was not significant.
In addition to the primary outcome, 62% of patients who stopped RAS inhibitor treatment and 56% of those who continued developed end-stage kidney disease or required renal-replacement therapy, which translated into an adjusted hazard ratio of 1.28 for this outcome among those who discontinued compared with those who continued, which was just short of significance (95% CI, 0.99-1.65).
The two study groups also showed no significant differences in the 3-year incidence of hospitalization for any reason, cardiovascular events, or deaths. The two groups also showed no meaningful differences in various domains of quality of life and no differences in serious adverse effects.
Participants had an eGFR less than 30 mL/min per 1.73 m2
The study ran at 39 United Kingdom centers in 2014-2019. Investigators enrolled adults with an eGFR of less than 30 mL/min per 1.73 m2 who were not on dialysis and had not received a kidney transplant. In addition, all enrolled patients had to have an annual drop in eGFR of more than 2 mL/min per 1.73 m2 during the prior 2 years and had to have been on treatment with at least one RAS inhibitor for more than 6 months.
The randomization protocol insured balanced distribution of subjects between the two study arms by age, eGFR, presence of diabetes, and level of proteinuria, among other factors. The study design also mandated that participants maintain a blood pressure of no more than 140/85 mm Hg.
Those who discontinued RAS-inhibitor treatment could receive any guideline-recommended antihypertensive agent that was not a RAS inhibitor, although adding a RAS inhibitor was permitted as a last treatment resort.
People in the maintenance group could receive whichever additional antihypertensive agents their treating clinicians deemed necessary for maintaining the target blood pressure.
The enrolled population was a median age of 63 years old and 68% were men. Their average eGFR at baseline was 18 mL/min per 1.73 m2, and 118 (29%) had an eGFR of less than 15 mL/min per 1.73 m2. Their median level of proteinuria was 115 mg/mmol (about 1,018 mg/g). Diabetes was prevalent in 37%, and 58% of participants were taking at least three antihypertensive medications at entry.
Among the study’s limitations, the researchers cited the open-label design, which may have affected clinical care and the tally of subjective endpoints, including quality of life and exercise capacity. Also, because the study enrolled people who were on a RAS inhibitor at the time of randomization, it did not include anyone who had already discontinued these agents.
Continue RAS inhibitors in advanced CKD for best outcomes
Dr. Bhandari and colleagues note that in a large observational trial published in January 2021, Swedish researchers found an increase in the incidence of major cardiovascular events and death among patients with advanced CKD who had discontinued RAS inhibitors.
But they observe, “Our trial did not have sufficient power to investigate the effect of the discontinuation of RAS inhibitors on cardiovascular events or mortality. However, because our findings are consistent with a lack of advantage for such discontinuation with respect to kidney function, there is little rationale to conduct a larger randomized trial to investigate cardiovascular safety.”
“Our findings do not support the hypothesis that the discontinuation of RAS inhibitors in patients with advanced and progressive chronic kidney disease would improve kidney function, quality of life, or exercise capacity.”
“The results of this trial will inform future clinical practice worldwide and guideline recommendations,” they conclude.
STOP ACEi received no commercial funding. Dr. Bhandari has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT KIDNEY WEEK 2022
Moving the needle: SGLT2 inhibitor role for isolated kidney disease
ORLANDO – in a pivotal trial with more than 6,600 patients.
This confirms the efficacy for this population that was previously seen with dapagliflozin, another agent from the same class, in the DAPA-CKD trial.
In the new trial, EMPA-Kidney, treatment with empagliflozin 10 mg daily for a median of 2.0 years led to a significant 28% relative risk reduction in the primary combined endpoint in comparison with placebo, William G. Herrington, MD, reported at the annual meeting of the American Society of Nephrology.
The results were simultaneously published in the New England Journal of Medicine.
In 2020, a different team of researchers running DAPA-CKD reported that during a median of 2.4 years, treatment of 4,304 patients with dapagliflozin 10 mg daily resulted in a significant 39% relative risk reduction, compared with placebo for an identical combined primary endpoint. Enrollment criteria for the DAPA-CKD trial were mostly similar to that of the current trial.
‘Remarkably similar’ findings
Results from EMPA-Kidney and DAPA-CKD are “remarkably similar,” said Dr. Herrington during a press briefing at the meeting.
He also noted that when the EMPA-Kidney study began – before results from DAPA-CKD were known – “we never imagined such a large effect” on important endpoints in people with CKD.
In addition to cardiovascular death, the combined primary endpoint included the incidence of renal death, incident end-stage kidney disease, a sustained decrease in estimated glomerular filtration rate to less than 10 mL/min per 1.73m2, or a sustained decrease in eGFR of at least 40% from baseline.
Having similar evidence from both trials “will hopefully provide people with the confidence to start to use SGLT2 inhibitors as standard care in people with CKD” who match enrollment criteria of the two trials, added Dr. Herrington, a nephrologist at the University of Oxford (England).
The analyses he reported also showed that empagliflozin had similar efficacy for the primary endpoint regardless of whether patients had type 2 diabetes at the time of enrollment and regardless of their eGFR at entry.
To enter EMPA-Kidney, people needed to have either an eGFR of 20-44 mL/min per 1.73m2 with no minimum level of albuminuria or an eGFR of 45-89 mL/min per 1.73m2 with a urine albumin-to-creatinine ratio (UACR) of at least 200 mg/g.
In contrast, to enroll in DAPA-CKD, patients had to have a UACR of at least 200 mg/g. This means that for the first time, EMPA-Kidney produced data on the relationship between albuminuria severity and the impact of treatment with an SGLT2 inhibitor in the enrolled population.
A signal of greater efficacy with higher UACR
A total of 6,609 patients underwent randomization in EMPA-Kidney. During a median of 2.0 years of follow-up, the primary endpoint – progression of kidney disease or death from cardiovascular causes – occurred in 432 of 3,304 patients (13.1%) in the empagliflozin group and in 558 of 3,305 patients (16.9%) in the placebo group (hazard ratio, 0.72; P < .001).
The results “suggested that the effects [of empagliflozin] are greater in patients with higher levels of albuminuria, with statistically significant heterogeneity between this subgroup and those with a UACR of less than 200 mg/g (P = .02),” Dr. Herrington said.
Of the study population, 54% had no evidence of diabetes at enrollment.
Having data from a second large trial of an SGLT2 inhibitor that included people with isolated CKD who did not have diabetes or heart failure “will start to move the needle” on using this class of drugs in these types of patients, commented F. Perry Wilson, MD, a nephrologist at Yale University, New Haven, Conn.
On the basis of the DAPA-CKD results, in April 2021 the Food and Drug Administration expanded dapagliflozin’s indications to include CKD, yet, “a lot of nephrologists consider SGLT2 inhibitors to be agents for people with diabetes or heart failure, and they defer prescribing them to endocrinologists and cardiologists,” Dr. Wilson said in an interview.
‘Flozinators’ rising
But Pascale H. Lane, MD, a pediatric nephrologist at the University of Oklahoma Health Sciences Center, Oklahoma City, commented that many nephrologists she knows have been prescribing dapagliflozin “widely” to their patients with CKD.
“I know many adult nephrologists who use it almost universally now,” Dr. Lane said. “They call themselves ‘flozinators.’ ”
EMPA-Kidney was sponsored by Boehringer Ingelheim, the company that along with Lilly markets empagliflozin (Jardiance). Dr. Herrington, Dr. Wilson, and Dr. Lane disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ORLANDO – in a pivotal trial with more than 6,600 patients.
This confirms the efficacy for this population that was previously seen with dapagliflozin, another agent from the same class, in the DAPA-CKD trial.
In the new trial, EMPA-Kidney, treatment with empagliflozin 10 mg daily for a median of 2.0 years led to a significant 28% relative risk reduction in the primary combined endpoint in comparison with placebo, William G. Herrington, MD, reported at the annual meeting of the American Society of Nephrology.
The results were simultaneously published in the New England Journal of Medicine.
In 2020, a different team of researchers running DAPA-CKD reported that during a median of 2.4 years, treatment of 4,304 patients with dapagliflozin 10 mg daily resulted in a significant 39% relative risk reduction, compared with placebo for an identical combined primary endpoint. Enrollment criteria for the DAPA-CKD trial were mostly similar to that of the current trial.
‘Remarkably similar’ findings
Results from EMPA-Kidney and DAPA-CKD are “remarkably similar,” said Dr. Herrington during a press briefing at the meeting.
He also noted that when the EMPA-Kidney study began – before results from DAPA-CKD were known – “we never imagined such a large effect” on important endpoints in people with CKD.
In addition to cardiovascular death, the combined primary endpoint included the incidence of renal death, incident end-stage kidney disease, a sustained decrease in estimated glomerular filtration rate to less than 10 mL/min per 1.73m2, or a sustained decrease in eGFR of at least 40% from baseline.
Having similar evidence from both trials “will hopefully provide people with the confidence to start to use SGLT2 inhibitors as standard care in people with CKD” who match enrollment criteria of the two trials, added Dr. Herrington, a nephrologist at the University of Oxford (England).
The analyses he reported also showed that empagliflozin had similar efficacy for the primary endpoint regardless of whether patients had type 2 diabetes at the time of enrollment and regardless of their eGFR at entry.
To enter EMPA-Kidney, people needed to have either an eGFR of 20-44 mL/min per 1.73m2 with no minimum level of albuminuria or an eGFR of 45-89 mL/min per 1.73m2 with a urine albumin-to-creatinine ratio (UACR) of at least 200 mg/g.
In contrast, to enroll in DAPA-CKD, patients had to have a UACR of at least 200 mg/g. This means that for the first time, EMPA-Kidney produced data on the relationship between albuminuria severity and the impact of treatment with an SGLT2 inhibitor in the enrolled population.
A signal of greater efficacy with higher UACR
A total of 6,609 patients underwent randomization in EMPA-Kidney. During a median of 2.0 years of follow-up, the primary endpoint – progression of kidney disease or death from cardiovascular causes – occurred in 432 of 3,304 patients (13.1%) in the empagliflozin group and in 558 of 3,305 patients (16.9%) in the placebo group (hazard ratio, 0.72; P < .001).
The results “suggested that the effects [of empagliflozin] are greater in patients with higher levels of albuminuria, with statistically significant heterogeneity between this subgroup and those with a UACR of less than 200 mg/g (P = .02),” Dr. Herrington said.
Of the study population, 54% had no evidence of diabetes at enrollment.
Having data from a second large trial of an SGLT2 inhibitor that included people with isolated CKD who did not have diabetes or heart failure “will start to move the needle” on using this class of drugs in these types of patients, commented F. Perry Wilson, MD, a nephrologist at Yale University, New Haven, Conn.
On the basis of the DAPA-CKD results, in April 2021 the Food and Drug Administration expanded dapagliflozin’s indications to include CKD, yet, “a lot of nephrologists consider SGLT2 inhibitors to be agents for people with diabetes or heart failure, and they defer prescribing them to endocrinologists and cardiologists,” Dr. Wilson said in an interview.
‘Flozinators’ rising
But Pascale H. Lane, MD, a pediatric nephrologist at the University of Oklahoma Health Sciences Center, Oklahoma City, commented that many nephrologists she knows have been prescribing dapagliflozin “widely” to their patients with CKD.
“I know many adult nephrologists who use it almost universally now,” Dr. Lane said. “They call themselves ‘flozinators.’ ”
EMPA-Kidney was sponsored by Boehringer Ingelheim, the company that along with Lilly markets empagliflozin (Jardiance). Dr. Herrington, Dr. Wilson, and Dr. Lane disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ORLANDO – in a pivotal trial with more than 6,600 patients.
This confirms the efficacy for this population that was previously seen with dapagliflozin, another agent from the same class, in the DAPA-CKD trial.
In the new trial, EMPA-Kidney, treatment with empagliflozin 10 mg daily for a median of 2.0 years led to a significant 28% relative risk reduction in the primary combined endpoint in comparison with placebo, William G. Herrington, MD, reported at the annual meeting of the American Society of Nephrology.
The results were simultaneously published in the New England Journal of Medicine.
In 2020, a different team of researchers running DAPA-CKD reported that during a median of 2.4 years, treatment of 4,304 patients with dapagliflozin 10 mg daily resulted in a significant 39% relative risk reduction, compared with placebo for an identical combined primary endpoint. Enrollment criteria for the DAPA-CKD trial were mostly similar to that of the current trial.
‘Remarkably similar’ findings
Results from EMPA-Kidney and DAPA-CKD are “remarkably similar,” said Dr. Herrington during a press briefing at the meeting.
He also noted that when the EMPA-Kidney study began – before results from DAPA-CKD were known – “we never imagined such a large effect” on important endpoints in people with CKD.
In addition to cardiovascular death, the combined primary endpoint included the incidence of renal death, incident end-stage kidney disease, a sustained decrease in estimated glomerular filtration rate to less than 10 mL/min per 1.73m2, or a sustained decrease in eGFR of at least 40% from baseline.
Having similar evidence from both trials “will hopefully provide people with the confidence to start to use SGLT2 inhibitors as standard care in people with CKD” who match enrollment criteria of the two trials, added Dr. Herrington, a nephrologist at the University of Oxford (England).
The analyses he reported also showed that empagliflozin had similar efficacy for the primary endpoint regardless of whether patients had type 2 diabetes at the time of enrollment and regardless of their eGFR at entry.
To enter EMPA-Kidney, people needed to have either an eGFR of 20-44 mL/min per 1.73m2 with no minimum level of albuminuria or an eGFR of 45-89 mL/min per 1.73m2 with a urine albumin-to-creatinine ratio (UACR) of at least 200 mg/g.
In contrast, to enroll in DAPA-CKD, patients had to have a UACR of at least 200 mg/g. This means that for the first time, EMPA-Kidney produced data on the relationship between albuminuria severity and the impact of treatment with an SGLT2 inhibitor in the enrolled population.
A signal of greater efficacy with higher UACR
A total of 6,609 patients underwent randomization in EMPA-Kidney. During a median of 2.0 years of follow-up, the primary endpoint – progression of kidney disease or death from cardiovascular causes – occurred in 432 of 3,304 patients (13.1%) in the empagliflozin group and in 558 of 3,305 patients (16.9%) in the placebo group (hazard ratio, 0.72; P < .001).
The results “suggested that the effects [of empagliflozin] are greater in patients with higher levels of albuminuria, with statistically significant heterogeneity between this subgroup and those with a UACR of less than 200 mg/g (P = .02),” Dr. Herrington said.
Of the study population, 54% had no evidence of diabetes at enrollment.
Having data from a second large trial of an SGLT2 inhibitor that included people with isolated CKD who did not have diabetes or heart failure “will start to move the needle” on using this class of drugs in these types of patients, commented F. Perry Wilson, MD, a nephrologist at Yale University, New Haven, Conn.
On the basis of the DAPA-CKD results, in April 2021 the Food and Drug Administration expanded dapagliflozin’s indications to include CKD, yet, “a lot of nephrologists consider SGLT2 inhibitors to be agents for people with diabetes or heart failure, and they defer prescribing them to endocrinologists and cardiologists,” Dr. Wilson said in an interview.
‘Flozinators’ rising
But Pascale H. Lane, MD, a pediatric nephrologist at the University of Oklahoma Health Sciences Center, Oklahoma City, commented that many nephrologists she knows have been prescribing dapagliflozin “widely” to their patients with CKD.
“I know many adult nephrologists who use it almost universally now,” Dr. Lane said. “They call themselves ‘flozinators.’ ”
EMPA-Kidney was sponsored by Boehringer Ingelheim, the company that along with Lilly markets empagliflozin (Jardiance). Dr. Herrington, Dr. Wilson, and Dr. Lane disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT KIDNEY WEEK 2022
Four methods to chip away at imposter syndrome
Regardless of the setting, one of the most frequently discussed topics in health care is imposter syndrome.
Imposter syndrome was first defined by Clance and Imes as an inability to internalize success, and the tendency to attribute success to external causes such as luck, error, or knowing the appropriate individual.1 This definition is essential because most health care professionals have had a sense of doubt or questioned the full extent of their competencies in various situations. I would argue that this is normal and – within reason – helpful to the practice of medicine. The problem with true imposter syndrome is that the individual does not incorporate success in a way that builds healthy self-esteem and self-efficacy.2
Imposter syndrome has a very nasty way of interacting with burnout. Studies have shown that imposter syndrome can be associated with high levels of emotional exhaustion at work.3 In my experience, this makes clinical sense. Professionals suffering from imposter syndrome can spend a great deal of time and energy trying to maintain a particular image.4 They are acting a part 24/7. Have you ever seriously tried to act? It’s arduous work. A friend once asked me to read a role for a play because “you’d be great; you’re a natural.” By the time I was done with rehearsal, I felt like I had run a 4-by-400-meter relay, by myself, in Victoria, Tex.
And any talk of imposter syndrome must include its running mate, perfectionism. These two conditions exist together so commonly it can be a bit of a chicken or egg question as to which came first.
Imposter syndrome, perfectionism, and burnout can form a deadly triad if not recognized and addressed quickly. In medicine, perfectionism can be a coping strategy that sets up unrelenting standards. Failure to meet unrelenting standards then serves as fuel and validation for imposter syndrome and emotional exhaustion. The consequences of this cycle going unchecked over a health care professional’s career are seismic and can include downstream effects ranging from depression to suicide.
Some readers will relate to this, while others will shrug their shoulders and say that this has never happened in their professional life. I get it. However, I would now ask if you have ever felt like an imposter in your personal life. I’ll make a cup of tea and wait for you to figure out precisely what is the boundary between your personal and professional life. Okay, all done? Great. Now I’ll give you some more time to sincerely reflect if any of the traits of imposter syndrome have described you at times in your personal life. Hmmm, interesting to think about, isn’t it?
I believe that health care professionals frequently use one credit card to pay off another, but the debt remains the same. So even if things are going well at work, we may have just shifted the debt to our personal lives. (At some point in the future, I’ll share my 10 greatest father fails to date to elucidate my point.)
In my work at the GW Resiliency and Well-Being Center, I’ve gravitated toward a few methods supported by evidence that help alleviate imposter syndrome symptoms and potentially serve as protective factors against the future development of imposter syndrome.4 These include but are not limited to:
- Keep a record of small personal success that is yours alone.
- Have a mentor to share failures with.
- Use personal reflection to examine what it means to successfully reach your goals and fulfill your purpose, not a relative value unit target.
- Share experiences with each other, so you know you’re not alone.
The last method is one of my favorites because it involves connecting to others and shining a light on our shared experiences and, coincidentally, our collective strengths. Once this collective strength is realized, the circumstances of that 4-by-400-meter relay change drastically. Be safe and well, everyone.
Lorenzo Norris, MD, is a psychiatrist and chief wellness officer for the George Washington University Medical Enterprise and serves as associate dean of student affairs and administration for the George Washington University School of Medicine and Health Sciences. A version of this article first appeared on Medscape.com.
References
1. Clance PR, Imes SA. The imposter phenomenon in high achieving women: Dynamics and therapeutic intervention. Psychotherapy: Theory, Research & Practice. 1978;15(3): 241-7. doi: 10.1037/h0086006.
2. Thomas M, Bigatti S. Perfectionism, impostor phenomenon, and mental health in medicine: A literature review. Int J Med Educ. 2020 Sep 28;11:201-3. doi: 10.5116/ijme.5f54.c8f8.
3. Liu RQ et al. Impostorism and anxiety contribute to burnout among resident physicians. Med Teach. 2022 Jul;44(7):758-64. doi: 10.1080/0142159X.2022.2028751.
4. Gottlieb M et al. Impostor syndrome among physicians and physicians in training: A scoping review. Med Educ. 2020 Feb;54(2):116-24. doi: 10.1111/medu.13956.
Regardless of the setting, one of the most frequently discussed topics in health care is imposter syndrome.
Imposter syndrome was first defined by Clance and Imes as an inability to internalize success, and the tendency to attribute success to external causes such as luck, error, or knowing the appropriate individual.1 This definition is essential because most health care professionals have had a sense of doubt or questioned the full extent of their competencies in various situations. I would argue that this is normal and – within reason – helpful to the practice of medicine. The problem with true imposter syndrome is that the individual does not incorporate success in a way that builds healthy self-esteem and self-efficacy.2
Imposter syndrome has a very nasty way of interacting with burnout. Studies have shown that imposter syndrome can be associated with high levels of emotional exhaustion at work.3 In my experience, this makes clinical sense. Professionals suffering from imposter syndrome can spend a great deal of time and energy trying to maintain a particular image.4 They are acting a part 24/7. Have you ever seriously tried to act? It’s arduous work. A friend once asked me to read a role for a play because “you’d be great; you’re a natural.” By the time I was done with rehearsal, I felt like I had run a 4-by-400-meter relay, by myself, in Victoria, Tex.
And any talk of imposter syndrome must include its running mate, perfectionism. These two conditions exist together so commonly it can be a bit of a chicken or egg question as to which came first.
Imposter syndrome, perfectionism, and burnout can form a deadly triad if not recognized and addressed quickly. In medicine, perfectionism can be a coping strategy that sets up unrelenting standards. Failure to meet unrelenting standards then serves as fuel and validation for imposter syndrome and emotional exhaustion. The consequences of this cycle going unchecked over a health care professional’s career are seismic and can include downstream effects ranging from depression to suicide.
Some readers will relate to this, while others will shrug their shoulders and say that this has never happened in their professional life. I get it. However, I would now ask if you have ever felt like an imposter in your personal life. I’ll make a cup of tea and wait for you to figure out precisely what is the boundary between your personal and professional life. Okay, all done? Great. Now I’ll give you some more time to sincerely reflect if any of the traits of imposter syndrome have described you at times in your personal life. Hmmm, interesting to think about, isn’t it?
I believe that health care professionals frequently use one credit card to pay off another, but the debt remains the same. So even if things are going well at work, we may have just shifted the debt to our personal lives. (At some point in the future, I’ll share my 10 greatest father fails to date to elucidate my point.)
In my work at the GW Resiliency and Well-Being Center, I’ve gravitated toward a few methods supported by evidence that help alleviate imposter syndrome symptoms and potentially serve as protective factors against the future development of imposter syndrome.4 These include but are not limited to:
- Keep a record of small personal success that is yours alone.
- Have a mentor to share failures with.
- Use personal reflection to examine what it means to successfully reach your goals and fulfill your purpose, not a relative value unit target.
- Share experiences with each other, so you know you’re not alone.
The last method is one of my favorites because it involves connecting to others and shining a light on our shared experiences and, coincidentally, our collective strengths. Once this collective strength is realized, the circumstances of that 4-by-400-meter relay change drastically. Be safe and well, everyone.
Lorenzo Norris, MD, is a psychiatrist and chief wellness officer for the George Washington University Medical Enterprise and serves as associate dean of student affairs and administration for the George Washington University School of Medicine and Health Sciences. A version of this article first appeared on Medscape.com.
References
1. Clance PR, Imes SA. The imposter phenomenon in high achieving women: Dynamics and therapeutic intervention. Psychotherapy: Theory, Research & Practice. 1978;15(3): 241-7. doi: 10.1037/h0086006.
2. Thomas M, Bigatti S. Perfectionism, impostor phenomenon, and mental health in medicine: A literature review. Int J Med Educ. 2020 Sep 28;11:201-3. doi: 10.5116/ijme.5f54.c8f8.
3. Liu RQ et al. Impostorism and anxiety contribute to burnout among resident physicians. Med Teach. 2022 Jul;44(7):758-64. doi: 10.1080/0142159X.2022.2028751.
4. Gottlieb M et al. Impostor syndrome among physicians and physicians in training: A scoping review. Med Educ. 2020 Feb;54(2):116-24. doi: 10.1111/medu.13956.
Regardless of the setting, one of the most frequently discussed topics in health care is imposter syndrome.
Imposter syndrome was first defined by Clance and Imes as an inability to internalize success, and the tendency to attribute success to external causes such as luck, error, or knowing the appropriate individual.1 This definition is essential because most health care professionals have had a sense of doubt or questioned the full extent of their competencies in various situations. I would argue that this is normal and – within reason – helpful to the practice of medicine. The problem with true imposter syndrome is that the individual does not incorporate success in a way that builds healthy self-esteem and self-efficacy.2
Imposter syndrome has a very nasty way of interacting with burnout. Studies have shown that imposter syndrome can be associated with high levels of emotional exhaustion at work.3 In my experience, this makes clinical sense. Professionals suffering from imposter syndrome can spend a great deal of time and energy trying to maintain a particular image.4 They are acting a part 24/7. Have you ever seriously tried to act? It’s arduous work. A friend once asked me to read a role for a play because “you’d be great; you’re a natural.” By the time I was done with rehearsal, I felt like I had run a 4-by-400-meter relay, by myself, in Victoria, Tex.
And any talk of imposter syndrome must include its running mate, perfectionism. These two conditions exist together so commonly it can be a bit of a chicken or egg question as to which came first.
Imposter syndrome, perfectionism, and burnout can form a deadly triad if not recognized and addressed quickly. In medicine, perfectionism can be a coping strategy that sets up unrelenting standards. Failure to meet unrelenting standards then serves as fuel and validation for imposter syndrome and emotional exhaustion. The consequences of this cycle going unchecked over a health care professional’s career are seismic and can include downstream effects ranging from depression to suicide.
Some readers will relate to this, while others will shrug their shoulders and say that this has never happened in their professional life. I get it. However, I would now ask if you have ever felt like an imposter in your personal life. I’ll make a cup of tea and wait for you to figure out precisely what is the boundary between your personal and professional life. Okay, all done? Great. Now I’ll give you some more time to sincerely reflect if any of the traits of imposter syndrome have described you at times in your personal life. Hmmm, interesting to think about, isn’t it?
I believe that health care professionals frequently use one credit card to pay off another, but the debt remains the same. So even if things are going well at work, we may have just shifted the debt to our personal lives. (At some point in the future, I’ll share my 10 greatest father fails to date to elucidate my point.)
In my work at the GW Resiliency and Well-Being Center, I’ve gravitated toward a few methods supported by evidence that help alleviate imposter syndrome symptoms and potentially serve as protective factors against the future development of imposter syndrome.4 These include but are not limited to:
- Keep a record of small personal success that is yours alone.
- Have a mentor to share failures with.
- Use personal reflection to examine what it means to successfully reach your goals and fulfill your purpose, not a relative value unit target.
- Share experiences with each other, so you know you’re not alone.
The last method is one of my favorites because it involves connecting to others and shining a light on our shared experiences and, coincidentally, our collective strengths. Once this collective strength is realized, the circumstances of that 4-by-400-meter relay change drastically. Be safe and well, everyone.
Lorenzo Norris, MD, is a psychiatrist and chief wellness officer for the George Washington University Medical Enterprise and serves as associate dean of student affairs and administration for the George Washington University School of Medicine and Health Sciences. A version of this article first appeared on Medscape.com.
References
1. Clance PR, Imes SA. The imposter phenomenon in high achieving women: Dynamics and therapeutic intervention. Psychotherapy: Theory, Research & Practice. 1978;15(3): 241-7. doi: 10.1037/h0086006.
2. Thomas M, Bigatti S. Perfectionism, impostor phenomenon, and mental health in medicine: A literature review. Int J Med Educ. 2020 Sep 28;11:201-3. doi: 10.5116/ijme.5f54.c8f8.
3. Liu RQ et al. Impostorism and anxiety contribute to burnout among resident physicians. Med Teach. 2022 Jul;44(7):758-64. doi: 10.1080/0142159X.2022.2028751.
4. Gottlieb M et al. Impostor syndrome among physicians and physicians in training: A scoping review. Med Educ. 2020 Feb;54(2):116-24. doi: 10.1111/medu.13956.
Microplastics and health risks: What do we really know?
You eat a credit card’s worth of plastic in a week. That may bother you. But does it harm you?
The answer depends on who you ask. Awareness of microplastics in general is certainly increasing; the most recent news is the detection of microplastics in human breast milk. Other research has suggested that we may be consuming up to 5 grams of plastic each week from our food, water, and certain consumer products.
The World Health Organization has been releasing reports on microplastics and human health since 2019. Their most recent report was released in late August 2022.
“Although the limited data provide little evidence that nano- and microplastic particles have adverse effects in humans, there is increasing public awareness and an overwhelming consensus among all stakeholders that plastics do not belong in the environment, and measures should be taken to mitigate exposure,” the WHO said at the time.
The WHO can’t go beyond what the data shows, of course. If microplastics are wreaking long-term havoc in our bodies as we speak, science hasn’t connected the dots enough to definitively say “this is the problem.”
But some researchers are willing to speculate – and, at the very least, the risks are becoming impossible to ignore. Dick Vethaak, PhD, a microplastics researcher and emeritus professor of ecotoxicology at Vrije Universiteit, Amsterdam, is blunt, calling them “a plastic time bomb.”
The plastic problem
Every piece of plastic that has ever been created is still on our planet today, apart from what has been burned. Past estimates show we only recycle about 9% of all plastic, leaving 9 billion tons in our landfills, oceans, and ecosystems. For context, that amount is 1,500 times heavier than the Great Pyramid of Khufu.
New data is even more dire. A 2022 report from Greenpeace showed a 5% U.S. recycling rate in 2021, with a large portion of what consumers think of as “recycled” still winding up in garbage piles or bodies of water.
And this plastic doesn’t disappear. Instead, it breaks down into smaller and smaller pieces known as microplastics and nanoplastics.
Microplastics have been confirmed in human blood, lung tissue, colons, placentas, stool, and breast milk. But how they impact our health is still unknown.
To assess risk, we must ask: “How hazardous is the material?” said Flemming Cassee, PhD, professor of inhalation toxicology at Utrecht (the Netherlands) University and coauthor of the WHO’s recent microplastics report.
There are three potential hazards of microplastics: their physical presence in our bodies, what they’re made of, and what they carry. To determine the extent of these risks, we need to know how much we’re exposed to, said Dr. Cassee.
The first initiative to research the impact of microplastics on human health came from the European Union in 2018. Although microplastics were around before then, we were unable to detect them, said Dr. Cassee.
That’s the real problem: warned Dr. Vethaak.
What, exactly, are microplastics?
Microplastics are plastic particles between 5 mm and 100 nm in diameter, or the width of a pencil eraser and something 10 times thinner than a human hair. Anything smaller than that is known as a nanoplastic.
“Microplastics include a wide range of different materials, different sizes, different shapes, different densities, and different colors,” said Evangelos Danopoulos, PhD, a microplastics researcher at Hull York (England) Medical School.
“Primary” microplastics are manufactured to be small and used in things like cosmetics and paints. “Secondary” microplastics result from the breaking down of larger plastic materials, like water bottles and plastic bags.
Secondary microplastics are more diverse than primary microplastics and can take forms ranging from fibers shed from synthetic clothing (like polyester) to pieces of a plastic spoon left in our rivers, lakes, and oceans. Any plastic in the environment will eventually become a secondary microplastic as natural forces such as wind, water currents, and UV radiation break it down into smaller and smaller pieces.
Plastic is a diverse material. Heather Leslie, PhD, senior researcher in Vrije Universiteit’s department of environment and health, likens it to spaghetti with sauce. The noodles are the long polymer backbone that all plastic shares. The sauces are “the pigments, the antioxidants, the flame retardants, etc., that make it functional,” she said.
What makes microplastics dangerous?
There are more than 10,000 different chemicals, or “sauces,” used to alter a plastic’s physical characteristics – making it softer, more rigid, or more flexible, said Hanna Dusza, PhD, of the Institute for Risk Assessment Sciences at Utrecht University.
As plastics degrade and become microplastics, these chemicals likely remain. Recent research has shown that microplastics leach these chemicals locally in human tissues, or other areas of accumulation, said Dr. Dusza. Some 2,400 of the 10,000 chemical additives were classified as substances of potential concern, meeting the European Union’s criteria for persistence, bioaccumulation, or toxicity.
Many of these chemicals also act as endocrine-disrupting compounds, or toxicants that imitate hormones when they enter the body. Hormones are active at very low concentrations in your bloodstream, explained Dr. Leslie. To your body, some chemical additives in plastic resemble hormones, so the body responds.
“Sometimes even a low dose of some of these additives can cause unwanted effects,” said Dr. Leslie.
Bisphenol A (BPA), for example, is one of the more infamous endocrine disruptors. It is used as an additive to make plastics more rigid and can be found in any number of plastic products, though areas of concern have been plastic water bottles, baby bottles, and the protective coatings in canned foods.
BPA may mimic estrogen, the female sex hormone essential for reproduction, neurodevelopment, and bone density. In men, estrogen regulates sperm count, sex drive, and erectile function. BPA exposure has been linked with – but not proven to cause – multiple cancer types, ADHD, obesity, and low sperm count. Most everyone has some amount of BPA circulating within their blood, but microplastics may retain BPA as they degrade, potentially increasing our exposure, leading to its unwanted consequences, said Dr. Dusza.
And BPA is just one of those 2,400 substances of “potential concern.”
The inflammation problem
A potentially larger health issue emerges from our bodies yet again doing what they are supposed to do when encountering microplastics. Particles can trigger an immune response when they enter your bloodstream, explains Nienke Vrisekoop, PhD, assistant professor at UMC Utrecht.
White blood cells have no issue breaking down things like bacteria, but microplastics cannot be degraded. When a white blood cell engulfs a certain mass of microplastics – either many small particles or a singular large one – it dies, releasing its enzymes and causing local inflammation.
Meanwhile, the plastic particle remains. So more white blood cells attack.
“This triggers continual activation that can result in various adverse effects, including oxidative stress and the release of cytokines that trigger inflammatory reactions, said Dr. Vethaak.
And “chronic inflammation is the prelude to chronic diseases,” said Dr. Leslie. “Every chronic disease, like cancer, heart disease, and even neuropsychiatric diseases like Parkinson’s or major depression, begins with inflammation.”
Meanwhile, inhaling microplastic particles can lead to respiratory diseases and cancer.
“The smallest particles – less than one-tenth of a micrometer – penetrate deep into the lungs and even into the bloodstream, causing damage to the heart, blood vessels and brain,” said Dr. Vetaak. “The only direct evidence comes from workers in the textile and plastic industries that had been exposed to very high amounts of plastic fibrous dust.”
Microplastics as carriers
Microplastics can also pick up harmful substances and deliver them into your body.
“When they’re in an environment, they basically can suck up [chemicals] like a sponge,” said Dr. Dusza. “These chemicals are known environmental pollutants, like pesticides, fluorinated compounds, flame retardants, and so on.”
Once in the body, these chemicals can be released, potentially leading to cancer, chronic inflammation, or other unknown effects.
Particles can also act as a vector for microbes, bacteria, and viruses. A September 2022 study found that infectious viruses can survive for 3 days in fresh water by “hitchhiking” on microplastics. Their porous nature provides microbes with a perfect environment in which to live and reproduce, said Dr. Dusza. If you ingest the plastics, you ingest the microbes.
How to minimize exposure
There is no way to avoid microplastics. They’re in the air we breathe, the products we use, the water we drink, and the food we eat.
Dr. Danopoulos reviewed 72 studies to quantify our consumption of microplastics in drinking water, salt, and seafood.
“We are exposed to millions of microplastics every year, and I was only looking at three food sources, so there are really a lot more,” he said. “Once plastic waste is mismanaged and it enters the environment, there is very little we can do to extract it.”
That said, we can take steps to lower our exposure and keep the problem from getting worse.
Water filtration is one option, though it is not perfect. Research has shown that municipal water treatment can be effective. An October 2021 study found that two methods – electrocoagulation-electroflotation and membrane filtration – can be 100% effective in removing microplastics from treated water. The problem? Not all municipal water treatment uses these methods – and you would have to investigate to find out if your locality does.
As for at-home filtration methods, they can be effective but can also be dicey. Some consumer brands claim they remove microplastics, but how well depends on not just the type of filter but the size of the particles in the water. Meanwhile, how do you know if a filter is working on your water without testing it, something few people will do? Best not to take a brand’s claims on face-value, but look for independent testing on at-home brands.
A longer-term project: Reduce our risk by reusing and recycling plastic waste. Limiting our consumption of plastic, especially single use plastic, decreases the amount available to become micro- and nanoplastics.
We must all learn to not treat plastic as waste, but rather as a renewable material, said Dr. Cassee. But if that seems like a tall order, it’s because it is.
“You’re a human being and you have a voice and there are a lot of other humans out there with voices,” said Dr. Leslie.
“You sign a petition in your community. You talk about it with your friends at the pub. If you’re a teacher, you discuss it in your class. You call your elected representatives and tell them what you think and how you want them to vote on bills.”
When people start working together, you can really amplify that voice, said Dr. Leslie.
What’s the bottom line right now, today?
Numerous sources have declared microplastics do not impact human health. But that’s largely because no direct evidence of this exists yet.
Even the WHO in its report suggests that progress must happen if we’re to fully understand the scope of the problem.
“Strengthening of the evidence necessary for reliable characterization and quantification of the risks to human health posed by [nano- and microplastics] will require active participation by all stakeholders,” it said.
All researchers interviewed for this article agree we don’t have enough evidence to draw any definite conclusions. But “if you look at the wrong endpoints, things will look safe, until you look at the endpoint where it’s really causing the problem,” said Dr. Leslie.
We must research our blind spots and continually ask: Where could we be wrong?
“It is a problem; it’s not going to go away,” said Dr. Danopoulos. “It’s going to get worse, and will continue to get worse, not by something that we are doing now but by something we did 5 years ago.”
Perhaps the question to be asked, then, is the hardest to answer: Are we willing to wait for the science?
A version of this article first appeared on WebMD.com.
You eat a credit card’s worth of plastic in a week. That may bother you. But does it harm you?
The answer depends on who you ask. Awareness of microplastics in general is certainly increasing; the most recent news is the detection of microplastics in human breast milk. Other research has suggested that we may be consuming up to 5 grams of plastic each week from our food, water, and certain consumer products.
The World Health Organization has been releasing reports on microplastics and human health since 2019. Their most recent report was released in late August 2022.
“Although the limited data provide little evidence that nano- and microplastic particles have adverse effects in humans, there is increasing public awareness and an overwhelming consensus among all stakeholders that plastics do not belong in the environment, and measures should be taken to mitigate exposure,” the WHO said at the time.
The WHO can’t go beyond what the data shows, of course. If microplastics are wreaking long-term havoc in our bodies as we speak, science hasn’t connected the dots enough to definitively say “this is the problem.”
But some researchers are willing to speculate – and, at the very least, the risks are becoming impossible to ignore. Dick Vethaak, PhD, a microplastics researcher and emeritus professor of ecotoxicology at Vrije Universiteit, Amsterdam, is blunt, calling them “a plastic time bomb.”
The plastic problem
Every piece of plastic that has ever been created is still on our planet today, apart from what has been burned. Past estimates show we only recycle about 9% of all plastic, leaving 9 billion tons in our landfills, oceans, and ecosystems. For context, that amount is 1,500 times heavier than the Great Pyramid of Khufu.
New data is even more dire. A 2022 report from Greenpeace showed a 5% U.S. recycling rate in 2021, with a large portion of what consumers think of as “recycled” still winding up in garbage piles or bodies of water.
And this plastic doesn’t disappear. Instead, it breaks down into smaller and smaller pieces known as microplastics and nanoplastics.
Microplastics have been confirmed in human blood, lung tissue, colons, placentas, stool, and breast milk. But how they impact our health is still unknown.
To assess risk, we must ask: “How hazardous is the material?” said Flemming Cassee, PhD, professor of inhalation toxicology at Utrecht (the Netherlands) University and coauthor of the WHO’s recent microplastics report.
There are three potential hazards of microplastics: their physical presence in our bodies, what they’re made of, and what they carry. To determine the extent of these risks, we need to know how much we’re exposed to, said Dr. Cassee.
The first initiative to research the impact of microplastics on human health came from the European Union in 2018. Although microplastics were around before then, we were unable to detect them, said Dr. Cassee.
That’s the real problem: warned Dr. Vethaak.
What, exactly, are microplastics?
Microplastics are plastic particles between 5 mm and 100 nm in diameter, or the width of a pencil eraser and something 10 times thinner than a human hair. Anything smaller than that is known as a nanoplastic.
“Microplastics include a wide range of different materials, different sizes, different shapes, different densities, and different colors,” said Evangelos Danopoulos, PhD, a microplastics researcher at Hull York (England) Medical School.
“Primary” microplastics are manufactured to be small and used in things like cosmetics and paints. “Secondary” microplastics result from the breaking down of larger plastic materials, like water bottles and plastic bags.
Secondary microplastics are more diverse than primary microplastics and can take forms ranging from fibers shed from synthetic clothing (like polyester) to pieces of a plastic spoon left in our rivers, lakes, and oceans. Any plastic in the environment will eventually become a secondary microplastic as natural forces such as wind, water currents, and UV radiation break it down into smaller and smaller pieces.
Plastic is a diverse material. Heather Leslie, PhD, senior researcher in Vrije Universiteit’s department of environment and health, likens it to spaghetti with sauce. The noodles are the long polymer backbone that all plastic shares. The sauces are “the pigments, the antioxidants, the flame retardants, etc., that make it functional,” she said.
What makes microplastics dangerous?
There are more than 10,000 different chemicals, or “sauces,” used to alter a plastic’s physical characteristics – making it softer, more rigid, or more flexible, said Hanna Dusza, PhD, of the Institute for Risk Assessment Sciences at Utrecht University.
As plastics degrade and become microplastics, these chemicals likely remain. Recent research has shown that microplastics leach these chemicals locally in human tissues, or other areas of accumulation, said Dr. Dusza. Some 2,400 of the 10,000 chemical additives were classified as substances of potential concern, meeting the European Union’s criteria for persistence, bioaccumulation, or toxicity.
Many of these chemicals also act as endocrine-disrupting compounds, or toxicants that imitate hormones when they enter the body. Hormones are active at very low concentrations in your bloodstream, explained Dr. Leslie. To your body, some chemical additives in plastic resemble hormones, so the body responds.
“Sometimes even a low dose of some of these additives can cause unwanted effects,” said Dr. Leslie.
Bisphenol A (BPA), for example, is one of the more infamous endocrine disruptors. It is used as an additive to make plastics more rigid and can be found in any number of plastic products, though areas of concern have been plastic water bottles, baby bottles, and the protective coatings in canned foods.
BPA may mimic estrogen, the female sex hormone essential for reproduction, neurodevelopment, and bone density. In men, estrogen regulates sperm count, sex drive, and erectile function. BPA exposure has been linked with – but not proven to cause – multiple cancer types, ADHD, obesity, and low sperm count. Most everyone has some amount of BPA circulating within their blood, but microplastics may retain BPA as they degrade, potentially increasing our exposure, leading to its unwanted consequences, said Dr. Dusza.
And BPA is just one of those 2,400 substances of “potential concern.”
The inflammation problem
A potentially larger health issue emerges from our bodies yet again doing what they are supposed to do when encountering microplastics. Particles can trigger an immune response when they enter your bloodstream, explains Nienke Vrisekoop, PhD, assistant professor at UMC Utrecht.
White blood cells have no issue breaking down things like bacteria, but microplastics cannot be degraded. When a white blood cell engulfs a certain mass of microplastics – either many small particles or a singular large one – it dies, releasing its enzymes and causing local inflammation.
Meanwhile, the plastic particle remains. So more white blood cells attack.
“This triggers continual activation that can result in various adverse effects, including oxidative stress and the release of cytokines that trigger inflammatory reactions, said Dr. Vethaak.
And “chronic inflammation is the prelude to chronic diseases,” said Dr. Leslie. “Every chronic disease, like cancer, heart disease, and even neuropsychiatric diseases like Parkinson’s or major depression, begins with inflammation.”
Meanwhile, inhaling microplastic particles can lead to respiratory diseases and cancer.
“The smallest particles – less than one-tenth of a micrometer – penetrate deep into the lungs and even into the bloodstream, causing damage to the heart, blood vessels and brain,” said Dr. Vetaak. “The only direct evidence comes from workers in the textile and plastic industries that had been exposed to very high amounts of plastic fibrous dust.”
Microplastics as carriers
Microplastics can also pick up harmful substances and deliver them into your body.
“When they’re in an environment, they basically can suck up [chemicals] like a sponge,” said Dr. Dusza. “These chemicals are known environmental pollutants, like pesticides, fluorinated compounds, flame retardants, and so on.”
Once in the body, these chemicals can be released, potentially leading to cancer, chronic inflammation, or other unknown effects.
Particles can also act as a vector for microbes, bacteria, and viruses. A September 2022 study found that infectious viruses can survive for 3 days in fresh water by “hitchhiking” on microplastics. Their porous nature provides microbes with a perfect environment in which to live and reproduce, said Dr. Dusza. If you ingest the plastics, you ingest the microbes.
How to minimize exposure
There is no way to avoid microplastics. They’re in the air we breathe, the products we use, the water we drink, and the food we eat.
Dr. Danopoulos reviewed 72 studies to quantify our consumption of microplastics in drinking water, salt, and seafood.
“We are exposed to millions of microplastics every year, and I was only looking at three food sources, so there are really a lot more,” he said. “Once plastic waste is mismanaged and it enters the environment, there is very little we can do to extract it.”
That said, we can take steps to lower our exposure and keep the problem from getting worse.
Water filtration is one option, though it is not perfect. Research has shown that municipal water treatment can be effective. An October 2021 study found that two methods – electrocoagulation-electroflotation and membrane filtration – can be 100% effective in removing microplastics from treated water. The problem? Not all municipal water treatment uses these methods – and you would have to investigate to find out if your locality does.
As for at-home filtration methods, they can be effective but can also be dicey. Some consumer brands claim they remove microplastics, but how well depends on not just the type of filter but the size of the particles in the water. Meanwhile, how do you know if a filter is working on your water without testing it, something few people will do? Best not to take a brand’s claims on face-value, but look for independent testing on at-home brands.
A longer-term project: Reduce our risk by reusing and recycling plastic waste. Limiting our consumption of plastic, especially single use plastic, decreases the amount available to become micro- and nanoplastics.
We must all learn to not treat plastic as waste, but rather as a renewable material, said Dr. Cassee. But if that seems like a tall order, it’s because it is.
“You’re a human being and you have a voice and there are a lot of other humans out there with voices,” said Dr. Leslie.
“You sign a petition in your community. You talk about it with your friends at the pub. If you’re a teacher, you discuss it in your class. You call your elected representatives and tell them what you think and how you want them to vote on bills.”
When people start working together, you can really amplify that voice, said Dr. Leslie.
What’s the bottom line right now, today?
Numerous sources have declared microplastics do not impact human health. But that’s largely because no direct evidence of this exists yet.
Even the WHO in its report suggests that progress must happen if we’re to fully understand the scope of the problem.
“Strengthening of the evidence necessary for reliable characterization and quantification of the risks to human health posed by [nano- and microplastics] will require active participation by all stakeholders,” it said.
All researchers interviewed for this article agree we don’t have enough evidence to draw any definite conclusions. But “if you look at the wrong endpoints, things will look safe, until you look at the endpoint where it’s really causing the problem,” said Dr. Leslie.
We must research our blind spots and continually ask: Where could we be wrong?
“It is a problem; it’s not going to go away,” said Dr. Danopoulos. “It’s going to get worse, and will continue to get worse, not by something that we are doing now but by something we did 5 years ago.”
Perhaps the question to be asked, then, is the hardest to answer: Are we willing to wait for the science?
A version of this article first appeared on WebMD.com.
You eat a credit card’s worth of plastic in a week. That may bother you. But does it harm you?
The answer depends on who you ask. Awareness of microplastics in general is certainly increasing; the most recent news is the detection of microplastics in human breast milk. Other research has suggested that we may be consuming up to 5 grams of plastic each week from our food, water, and certain consumer products.
The World Health Organization has been releasing reports on microplastics and human health since 2019. Their most recent report was released in late August 2022.
“Although the limited data provide little evidence that nano- and microplastic particles have adverse effects in humans, there is increasing public awareness and an overwhelming consensus among all stakeholders that plastics do not belong in the environment, and measures should be taken to mitigate exposure,” the WHO said at the time.
The WHO can’t go beyond what the data shows, of course. If microplastics are wreaking long-term havoc in our bodies as we speak, science hasn’t connected the dots enough to definitively say “this is the problem.”
But some researchers are willing to speculate – and, at the very least, the risks are becoming impossible to ignore. Dick Vethaak, PhD, a microplastics researcher and emeritus professor of ecotoxicology at Vrije Universiteit, Amsterdam, is blunt, calling them “a plastic time bomb.”
The plastic problem
Every piece of plastic that has ever been created is still on our planet today, apart from what has been burned. Past estimates show we only recycle about 9% of all plastic, leaving 9 billion tons in our landfills, oceans, and ecosystems. For context, that amount is 1,500 times heavier than the Great Pyramid of Khufu.
New data is even more dire. A 2022 report from Greenpeace showed a 5% U.S. recycling rate in 2021, with a large portion of what consumers think of as “recycled” still winding up in garbage piles or bodies of water.
And this plastic doesn’t disappear. Instead, it breaks down into smaller and smaller pieces known as microplastics and nanoplastics.
Microplastics have been confirmed in human blood, lung tissue, colons, placentas, stool, and breast milk. But how they impact our health is still unknown.
To assess risk, we must ask: “How hazardous is the material?” said Flemming Cassee, PhD, professor of inhalation toxicology at Utrecht (the Netherlands) University and coauthor of the WHO’s recent microplastics report.
There are three potential hazards of microplastics: their physical presence in our bodies, what they’re made of, and what they carry. To determine the extent of these risks, we need to know how much we’re exposed to, said Dr. Cassee.
The first initiative to research the impact of microplastics on human health came from the European Union in 2018. Although microplastics were around before then, we were unable to detect them, said Dr. Cassee.
That’s the real problem: warned Dr. Vethaak.
What, exactly, are microplastics?
Microplastics are plastic particles between 5 mm and 100 nm in diameter, or the width of a pencil eraser and something 10 times thinner than a human hair. Anything smaller than that is known as a nanoplastic.
“Microplastics include a wide range of different materials, different sizes, different shapes, different densities, and different colors,” said Evangelos Danopoulos, PhD, a microplastics researcher at Hull York (England) Medical School.
“Primary” microplastics are manufactured to be small and used in things like cosmetics and paints. “Secondary” microplastics result from the breaking down of larger plastic materials, like water bottles and plastic bags.
Secondary microplastics are more diverse than primary microplastics and can take forms ranging from fibers shed from synthetic clothing (like polyester) to pieces of a plastic spoon left in our rivers, lakes, and oceans. Any plastic in the environment will eventually become a secondary microplastic as natural forces such as wind, water currents, and UV radiation break it down into smaller and smaller pieces.
Plastic is a diverse material. Heather Leslie, PhD, senior researcher in Vrije Universiteit’s department of environment and health, likens it to spaghetti with sauce. The noodles are the long polymer backbone that all plastic shares. The sauces are “the pigments, the antioxidants, the flame retardants, etc., that make it functional,” she said.
What makes microplastics dangerous?
There are more than 10,000 different chemicals, or “sauces,” used to alter a plastic’s physical characteristics – making it softer, more rigid, or more flexible, said Hanna Dusza, PhD, of the Institute for Risk Assessment Sciences at Utrecht University.
As plastics degrade and become microplastics, these chemicals likely remain. Recent research has shown that microplastics leach these chemicals locally in human tissues, or other areas of accumulation, said Dr. Dusza. Some 2,400 of the 10,000 chemical additives were classified as substances of potential concern, meeting the European Union’s criteria for persistence, bioaccumulation, or toxicity.
Many of these chemicals also act as endocrine-disrupting compounds, or toxicants that imitate hormones when they enter the body. Hormones are active at very low concentrations in your bloodstream, explained Dr. Leslie. To your body, some chemical additives in plastic resemble hormones, so the body responds.
“Sometimes even a low dose of some of these additives can cause unwanted effects,” said Dr. Leslie.
Bisphenol A (BPA), for example, is one of the more infamous endocrine disruptors. It is used as an additive to make plastics more rigid and can be found in any number of plastic products, though areas of concern have been plastic water bottles, baby bottles, and the protective coatings in canned foods.
BPA may mimic estrogen, the female sex hormone essential for reproduction, neurodevelopment, and bone density. In men, estrogen regulates sperm count, sex drive, and erectile function. BPA exposure has been linked with – but not proven to cause – multiple cancer types, ADHD, obesity, and low sperm count. Most everyone has some amount of BPA circulating within their blood, but microplastics may retain BPA as they degrade, potentially increasing our exposure, leading to its unwanted consequences, said Dr. Dusza.
And BPA is just one of those 2,400 substances of “potential concern.”
The inflammation problem
A potentially larger health issue emerges from our bodies yet again doing what they are supposed to do when encountering microplastics. Particles can trigger an immune response when they enter your bloodstream, explains Nienke Vrisekoop, PhD, assistant professor at UMC Utrecht.
White blood cells have no issue breaking down things like bacteria, but microplastics cannot be degraded. When a white blood cell engulfs a certain mass of microplastics – either many small particles or a singular large one – it dies, releasing its enzymes and causing local inflammation.
Meanwhile, the plastic particle remains. So more white blood cells attack.
“This triggers continual activation that can result in various adverse effects, including oxidative stress and the release of cytokines that trigger inflammatory reactions, said Dr. Vethaak.
And “chronic inflammation is the prelude to chronic diseases,” said Dr. Leslie. “Every chronic disease, like cancer, heart disease, and even neuropsychiatric diseases like Parkinson’s or major depression, begins with inflammation.”
Meanwhile, inhaling microplastic particles can lead to respiratory diseases and cancer.
“The smallest particles – less than one-tenth of a micrometer – penetrate deep into the lungs and even into the bloodstream, causing damage to the heart, blood vessels and brain,” said Dr. Vetaak. “The only direct evidence comes from workers in the textile and plastic industries that had been exposed to very high amounts of plastic fibrous dust.”
Microplastics as carriers
Microplastics can also pick up harmful substances and deliver them into your body.
“When they’re in an environment, they basically can suck up [chemicals] like a sponge,” said Dr. Dusza. “These chemicals are known environmental pollutants, like pesticides, fluorinated compounds, flame retardants, and so on.”
Once in the body, these chemicals can be released, potentially leading to cancer, chronic inflammation, or other unknown effects.
Particles can also act as a vector for microbes, bacteria, and viruses. A September 2022 study found that infectious viruses can survive for 3 days in fresh water by “hitchhiking” on microplastics. Their porous nature provides microbes with a perfect environment in which to live and reproduce, said Dr. Dusza. If you ingest the plastics, you ingest the microbes.
How to minimize exposure
There is no way to avoid microplastics. They’re in the air we breathe, the products we use, the water we drink, and the food we eat.
Dr. Danopoulos reviewed 72 studies to quantify our consumption of microplastics in drinking water, salt, and seafood.
“We are exposed to millions of microplastics every year, and I was only looking at three food sources, so there are really a lot more,” he said. “Once plastic waste is mismanaged and it enters the environment, there is very little we can do to extract it.”
That said, we can take steps to lower our exposure and keep the problem from getting worse.
Water filtration is one option, though it is not perfect. Research has shown that municipal water treatment can be effective. An October 2021 study found that two methods – electrocoagulation-electroflotation and membrane filtration – can be 100% effective in removing microplastics from treated water. The problem? Not all municipal water treatment uses these methods – and you would have to investigate to find out if your locality does.
As for at-home filtration methods, they can be effective but can also be dicey. Some consumer brands claim they remove microplastics, but how well depends on not just the type of filter but the size of the particles in the water. Meanwhile, how do you know if a filter is working on your water without testing it, something few people will do? Best not to take a brand’s claims on face-value, but look for independent testing on at-home brands.
A longer-term project: Reduce our risk by reusing and recycling plastic waste. Limiting our consumption of plastic, especially single use plastic, decreases the amount available to become micro- and nanoplastics.
We must all learn to not treat plastic as waste, but rather as a renewable material, said Dr. Cassee. But if that seems like a tall order, it’s because it is.
“You’re a human being and you have a voice and there are a lot of other humans out there with voices,” said Dr. Leslie.
“You sign a petition in your community. You talk about it with your friends at the pub. If you’re a teacher, you discuss it in your class. You call your elected representatives and tell them what you think and how you want them to vote on bills.”
When people start working together, you can really amplify that voice, said Dr. Leslie.
What’s the bottom line right now, today?
Numerous sources have declared microplastics do not impact human health. But that’s largely because no direct evidence of this exists yet.
Even the WHO in its report suggests that progress must happen if we’re to fully understand the scope of the problem.
“Strengthening of the evidence necessary for reliable characterization and quantification of the risks to human health posed by [nano- and microplastics] will require active participation by all stakeholders,” it said.
All researchers interviewed for this article agree we don’t have enough evidence to draw any definite conclusions. But “if you look at the wrong endpoints, things will look safe, until you look at the endpoint where it’s really causing the problem,” said Dr. Leslie.
We must research our blind spots and continually ask: Where could we be wrong?
“It is a problem; it’s not going to go away,” said Dr. Danopoulos. “It’s going to get worse, and will continue to get worse, not by something that we are doing now but by something we did 5 years ago.”
Perhaps the question to be asked, then, is the hardest to answer: Are we willing to wait for the science?
A version of this article first appeared on WebMD.com.
VA Delays EHR Rollout—Again
The US Department of Veterans Affairs (VA) is pushing further deployments of the system to June 2023 “to address challenges” and make sure it’s functioning optimally.
Among the challenges: Safety concerns “voluminous enough and prevalent enough” to prompt the VA to disclose to 41,500 veterans enrolled in Washington, Idaho, Oregon, Montana, and Ohio that their care “may have been impacted as a result of the system’s deployment as it is currently configured,” VA Undersecretary for Health Shereef Elnahal said in a news conference.
The plan was to launch in the first quarter of 2023 in Western Washington, Michigan, and Ohio. But in a recent release, the VA said an investigation had found several technical and system issues, such as latency and slowness, and problems with patient scheduling, referrals, medication management, and other types of medical orders. During this “assess and address” period, the VA says, it will correct outstanding issues—especially those that may have patient safety implications—before restarting deployments at other VA medical centers.
“Right now, the Oracle Cerner [EHR] system is not delivering for veterans or VA health care providers—and we are holding Oracle Cerner and ourselves accountable to get this right,” said VA Deputy Secretary Donald Remy, who has oversight over the EHR program. “We are delaying all future deployments of the new EHR while we fully assess performance and address every concern. Veterans and clinicians deserve a seamless, modernized health record system, and we will not rest until they get it.”
The modernized EHR, intended to replace the Veterans Health Information Systems and Technology Architecture (VistA), has been plagued by problems from the very first launch in October 2020 at Mann-Grandstaff VA Medical Center and associated clinics in the Northwest. Deputy Inspector David Case, of the Office of Inspector General (OIG), reported to the House Committee on Veterans’ Affairs on oversight between 2020 and July 2021. Among other things, the OIG identified problems with the infrastructure and with users’ experiences. Clinical and administrative staff at Mann-Grandstaff and a Columbus clinic shared their frustration with OIG personnel about the “significant system and process limitations that raised concerns about the continuity of and prompt access to quality patient care.”
For example, according to an OIG report from July 2022, the new EHR sent thousands of orders for medical care to an “undetectable location, or unknown queue” instead of the intended location. The mis-delivery caused 149 patient harm events.
On October 11, the VA confirmed to The Spokesman-Review, a Spokane-based newspaper, that a patient had died at the VA clinic in Columbus. The death was attributed to the patient not receiving medication due to incorrect information. The incident is being treated as a potential “sentinel event.”
Elnahal, who met with employees in September at the Columbus clinic where the Oracle Cerner system was launched in April, said he found that the highly complex system made it hard for clinicians to perform routine tasks, such as ordering tests or follow-up appointments. Delays in follow-ups—including a yearlong delay in treatment for a veteran ultimately diagnosed with terminal cancer—were the main cause of the cases of harm cited in the July OIG report.
The veterans who received the letter about the potential impact on their health care “got caught up in this phenomenon of commands not getting where they need to go,” Elnahal said in a news conference in September.
Senator Patty Murray (D-WA), a senior member of the Veterans Affairs Committee, has been consistently pressing the VA to do something about the EHR system’s flaws. “It’s painfully clear,” she said in a statement, “we need to stop this program until the VA can fix these serious issues before they hurt anyone else.”
After finding more than 200 orders in the unknown queue in May 2022, the OIG said, it “has concerns with the effectiveness of Cerner’s plan to mitigate the safety risk.” While executing its “assess and address” plan, the VA will continue to focus on the 5 facilities where the new system has been deployed. “Sometimes, you’re not presented with options to immediately resolve the safety concerns that are in front of you,” Elnahal told reporters. “It is simply the case that the best option in front of us to resolve these patient safety concerns is to work with Oracle Cerner over the next several months to resolve the Cerner system issues at the sites where it exists. We know that this is possible, because other health systems have gone through this journey before, and I think we can do it.”
Veterans who believe their care may have been affected can call a dedicated call center at 800.319.9446. A VA health care team will follow up within 5 days.
The US Department of Veterans Affairs (VA) is pushing further deployments of the system to June 2023 “to address challenges” and make sure it’s functioning optimally.
Among the challenges: Safety concerns “voluminous enough and prevalent enough” to prompt the VA to disclose to 41,500 veterans enrolled in Washington, Idaho, Oregon, Montana, and Ohio that their care “may have been impacted as a result of the system’s deployment as it is currently configured,” VA Undersecretary for Health Shereef Elnahal said in a news conference.
The plan was to launch in the first quarter of 2023 in Western Washington, Michigan, and Ohio. But in a recent release, the VA said an investigation had found several technical and system issues, such as latency and slowness, and problems with patient scheduling, referrals, medication management, and other types of medical orders. During this “assess and address” period, the VA says, it will correct outstanding issues—especially those that may have patient safety implications—before restarting deployments at other VA medical centers.
“Right now, the Oracle Cerner [EHR] system is not delivering for veterans or VA health care providers—and we are holding Oracle Cerner and ourselves accountable to get this right,” said VA Deputy Secretary Donald Remy, who has oversight over the EHR program. “We are delaying all future deployments of the new EHR while we fully assess performance and address every concern. Veterans and clinicians deserve a seamless, modernized health record system, and we will not rest until they get it.”
The modernized EHR, intended to replace the Veterans Health Information Systems and Technology Architecture (VistA), has been plagued by problems from the very first launch in October 2020 at Mann-Grandstaff VA Medical Center and associated clinics in the Northwest. Deputy Inspector David Case, of the Office of Inspector General (OIG), reported to the House Committee on Veterans’ Affairs on oversight between 2020 and July 2021. Among other things, the OIG identified problems with the infrastructure and with users’ experiences. Clinical and administrative staff at Mann-Grandstaff and a Columbus clinic shared their frustration with OIG personnel about the “significant system and process limitations that raised concerns about the continuity of and prompt access to quality patient care.”
For example, according to an OIG report from July 2022, the new EHR sent thousands of orders for medical care to an “undetectable location, or unknown queue” instead of the intended location. The mis-delivery caused 149 patient harm events.
On October 11, the VA confirmed to The Spokesman-Review, a Spokane-based newspaper, that a patient had died at the VA clinic in Columbus. The death was attributed to the patient not receiving medication due to incorrect information. The incident is being treated as a potential “sentinel event.”
Elnahal, who met with employees in September at the Columbus clinic where the Oracle Cerner system was launched in April, said he found that the highly complex system made it hard for clinicians to perform routine tasks, such as ordering tests or follow-up appointments. Delays in follow-ups—including a yearlong delay in treatment for a veteran ultimately diagnosed with terminal cancer—were the main cause of the cases of harm cited in the July OIG report.
The veterans who received the letter about the potential impact on their health care “got caught up in this phenomenon of commands not getting where they need to go,” Elnahal said in a news conference in September.
Senator Patty Murray (D-WA), a senior member of the Veterans Affairs Committee, has been consistently pressing the VA to do something about the EHR system’s flaws. “It’s painfully clear,” she said in a statement, “we need to stop this program until the VA can fix these serious issues before they hurt anyone else.”
After finding more than 200 orders in the unknown queue in May 2022, the OIG said, it “has concerns with the effectiveness of Cerner’s plan to mitigate the safety risk.” While executing its “assess and address” plan, the VA will continue to focus on the 5 facilities where the new system has been deployed. “Sometimes, you’re not presented with options to immediately resolve the safety concerns that are in front of you,” Elnahal told reporters. “It is simply the case that the best option in front of us to resolve these patient safety concerns is to work with Oracle Cerner over the next several months to resolve the Cerner system issues at the sites where it exists. We know that this is possible, because other health systems have gone through this journey before, and I think we can do it.”
Veterans who believe their care may have been affected can call a dedicated call center at 800.319.9446. A VA health care team will follow up within 5 days.
The US Department of Veterans Affairs (VA) is pushing further deployments of the system to June 2023 “to address challenges” and make sure it’s functioning optimally.
Among the challenges: Safety concerns “voluminous enough and prevalent enough” to prompt the VA to disclose to 41,500 veterans enrolled in Washington, Idaho, Oregon, Montana, and Ohio that their care “may have been impacted as a result of the system’s deployment as it is currently configured,” VA Undersecretary for Health Shereef Elnahal said in a news conference.
The plan was to launch in the first quarter of 2023 in Western Washington, Michigan, and Ohio. But in a recent release, the VA said an investigation had found several technical and system issues, such as latency and slowness, and problems with patient scheduling, referrals, medication management, and other types of medical orders. During this “assess and address” period, the VA says, it will correct outstanding issues—especially those that may have patient safety implications—before restarting deployments at other VA medical centers.
“Right now, the Oracle Cerner [EHR] system is not delivering for veterans or VA health care providers—and we are holding Oracle Cerner and ourselves accountable to get this right,” said VA Deputy Secretary Donald Remy, who has oversight over the EHR program. “We are delaying all future deployments of the new EHR while we fully assess performance and address every concern. Veterans and clinicians deserve a seamless, modernized health record system, and we will not rest until they get it.”
The modernized EHR, intended to replace the Veterans Health Information Systems and Technology Architecture (VistA), has been plagued by problems from the very first launch in October 2020 at Mann-Grandstaff VA Medical Center and associated clinics in the Northwest. Deputy Inspector David Case, of the Office of Inspector General (OIG), reported to the House Committee on Veterans’ Affairs on oversight between 2020 and July 2021. Among other things, the OIG identified problems with the infrastructure and with users’ experiences. Clinical and administrative staff at Mann-Grandstaff and a Columbus clinic shared their frustration with OIG personnel about the “significant system and process limitations that raised concerns about the continuity of and prompt access to quality patient care.”
For example, according to an OIG report from July 2022, the new EHR sent thousands of orders for medical care to an “undetectable location, or unknown queue” instead of the intended location. The mis-delivery caused 149 patient harm events.
On October 11, the VA confirmed to The Spokesman-Review, a Spokane-based newspaper, that a patient had died at the VA clinic in Columbus. The death was attributed to the patient not receiving medication due to incorrect information. The incident is being treated as a potential “sentinel event.”
Elnahal, who met with employees in September at the Columbus clinic where the Oracle Cerner system was launched in April, said he found that the highly complex system made it hard for clinicians to perform routine tasks, such as ordering tests or follow-up appointments. Delays in follow-ups—including a yearlong delay in treatment for a veteran ultimately diagnosed with terminal cancer—were the main cause of the cases of harm cited in the July OIG report.
The veterans who received the letter about the potential impact on their health care “got caught up in this phenomenon of commands not getting where they need to go,” Elnahal said in a news conference in September.
Senator Patty Murray (D-WA), a senior member of the Veterans Affairs Committee, has been consistently pressing the VA to do something about the EHR system’s flaws. “It’s painfully clear,” she said in a statement, “we need to stop this program until the VA can fix these serious issues before they hurt anyone else.”
After finding more than 200 orders in the unknown queue in May 2022, the OIG said, it “has concerns with the effectiveness of Cerner’s plan to mitigate the safety risk.” While executing its “assess and address” plan, the VA will continue to focus on the 5 facilities where the new system has been deployed. “Sometimes, you’re not presented with options to immediately resolve the safety concerns that are in front of you,” Elnahal told reporters. “It is simply the case that the best option in front of us to resolve these patient safety concerns is to work with Oracle Cerner over the next several months to resolve the Cerner system issues at the sites where it exists. We know that this is possible, because other health systems have gone through this journey before, and I think we can do it.”
Veterans who believe their care may have been affected can call a dedicated call center at 800.319.9446. A VA health care team will follow up within 5 days.
Marital stress tied to worse outcome in young MI patients
Severe marital stress was associated with worse recovery after myocardial infarction in a large U.S. cohort of married/partnered patients aged 55 years or younger.
Compared with patients who reported no or mild marital stress a month after their MI, patients who reported severe marital stress had worse physical and mental health, worse generic and cardiovascular quality of life, more frequent angina symptoms, and a greater likelihood of having a hospital readmission a year later.
These findings held true after adjusting for gender, age, race/ethnicity, and baseline health status (model 1) and after further adjusting for education and income levels and employment and insurance status (model 2).
A greater percentage of women than men reported having severe marital stress (39% vs. 30%; P = .001).
Cenjing Zhu, MPhil, a PhD candidate at Yale University, New Haven, Conn., and colleagues will present this study at the American Heart Association scientific sessions.
The results show that “both patients and care providers should be aware that stress experienced in one’s everyday life, such as marital stress, can affect AMI [acute MI] recovery,” Ms. Zhu said in an email.
Health care providers should consider incorporating screening for everyday stress during follow-up patient visits to better spot people at high risk of a poor recovery and further hospitalizations, she added. When possible, they could guide patients to resources to help them manage and reduce their stress levels.
According to Ms. Zhu, the findings suggest that “managing personal stress may be as important as managing other clinical risk factors during the recovery process.”
This study in younger patients with MI “shows that high levels of marital stress impair heart attack recovery, and women have greater impairment in their heart attack recovery compared to men,” AHA spokesperson Nieca Goldberg, MD, who was not involved with this research, told this news organization.
The study shows that “clinicians have to incorporate mental health as part of their assessment of all patients,” said Dr. Goldberg, a clinical associate professor of medicine at New York University and medical director of Atria New York City.
“Our mental health impacts our physical health,” she noted. “Questions about marital stress should be included as part of an overall assessment of mental health. This means assessing all patients for stress, anxiety, and depression.”
Patients who are experiencing marital stress should share the information with their doctor and discuss ways to be referred to therapists and cardiac rehabilitation providers, she said. “My final thought is, women have often been told that their cardiac symptoms are due to stress by doctors. Now we know stress impacts physical health and [is] no longer an excuse but a contributing factor to our physical health.”
Does marital stress affect young MI recovery?
Previous literature has linked psychological stress with worse cardiovascular outcomes, Ms. Zhu noted.
However, little is known about the prognostic impact of marital stress on 1-year health outcomes for younger people who survive an MI.
To investigate this, the researchers analyzed data from participants in the Variation in Recovery: Role of Gender on Outcomes of Young AMI Patients (VIRGO) study.
The current study comprised 1,593 adults, including 1,020 female participants (64%), who were treated for MI at 103 hospitals in 30 U.S. states.
VIRGO enrolled participants in a 2:1 female-to-male ratio so as to enrich the inclusion of women, Ms. Zhu explained.
In the study, “partnered” participants were individuals who self-reported as “living as married/living with a partner.” There were 126 such patients (8%) in the current study.
The mean age of the patients was 47, and about 90% were 40-55 years old. Three quarters were White, 13% were Black, and 7% were Hispanic.
Marital stress was assessed on the basis of patients’ replies to 17 questions in the Stockholm Marital Stress Scale regarding the quality of their emotional and sexual relationships with their spouses/partners.
The researchers divided patients into three groups on the basis of their marital stress: mild or absent (lowest quartile), moderate (second quartile), and severe (upper two quartiles).
At 1 year after their MI, patients replied to questionnaires that assessed their health, quality of life, and depressive and angina symptoms. Hospital readmissions were determined on the basis of self-reports and medical records.
Compared to participants who reported no or mild marital stress, those who reported severe mental stress had significantly worse scores for physical and mental health and generic and cardiovascular quality of life, after adjusting for baseline health and demographics. They had worse scores for mental health and quality of life, after further adjusting for socioeconomic status.
In the fully adjusted model, patients who reported severe marital stress were significantly more likely to report more frequent chest pain/angina (odds ratio, 1.49; 95% confidence interval, 1.06-2.10; P = .023) and to have been readmitted to hospital for any cause (OR, 1.45; 95% CI, 1.04-2.00; P = .006), compared with the patients who reported no or mild marital stress.
Study limitations include the fact that the findings are based on self-reported questionnaire replies; they may not be generalizable to patients in other countries; and they do not extend beyond a period of 1 year.
The researchers call for further research “to understand this complex relationship and potential causal pathway associated with these findings.”
“Additional stressors beyond marital stress, such as financial strain or work stress, may also play a role in young adults’ recovery, and the interaction between these factors require further research,” Ms. Zhu noted in a press release from the AHA.
The study was funded by Canadian Institutes of Health Research. The VIRGO study was funded by the National Heart, Lung, and Blood Institute. Ms. Zhu and Dr. Goldberg have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Severe marital stress was associated with worse recovery after myocardial infarction in a large U.S. cohort of married/partnered patients aged 55 years or younger.
Compared with patients who reported no or mild marital stress a month after their MI, patients who reported severe marital stress had worse physical and mental health, worse generic and cardiovascular quality of life, more frequent angina symptoms, and a greater likelihood of having a hospital readmission a year later.
These findings held true after adjusting for gender, age, race/ethnicity, and baseline health status (model 1) and after further adjusting for education and income levels and employment and insurance status (model 2).
A greater percentage of women than men reported having severe marital stress (39% vs. 30%; P = .001).
Cenjing Zhu, MPhil, a PhD candidate at Yale University, New Haven, Conn., and colleagues will present this study at the American Heart Association scientific sessions.
The results show that “both patients and care providers should be aware that stress experienced in one’s everyday life, such as marital stress, can affect AMI [acute MI] recovery,” Ms. Zhu said in an email.
Health care providers should consider incorporating screening for everyday stress during follow-up patient visits to better spot people at high risk of a poor recovery and further hospitalizations, she added. When possible, they could guide patients to resources to help them manage and reduce their stress levels.
According to Ms. Zhu, the findings suggest that “managing personal stress may be as important as managing other clinical risk factors during the recovery process.”
This study in younger patients with MI “shows that high levels of marital stress impair heart attack recovery, and women have greater impairment in their heart attack recovery compared to men,” AHA spokesperson Nieca Goldberg, MD, who was not involved with this research, told this news organization.
The study shows that “clinicians have to incorporate mental health as part of their assessment of all patients,” said Dr. Goldberg, a clinical associate professor of medicine at New York University and medical director of Atria New York City.
“Our mental health impacts our physical health,” she noted. “Questions about marital stress should be included as part of an overall assessment of mental health. This means assessing all patients for stress, anxiety, and depression.”
Patients who are experiencing marital stress should share the information with their doctor and discuss ways to be referred to therapists and cardiac rehabilitation providers, she said. “My final thought is, women have often been told that their cardiac symptoms are due to stress by doctors. Now we know stress impacts physical health and [is] no longer an excuse but a contributing factor to our physical health.”
Does marital stress affect young MI recovery?
Previous literature has linked psychological stress with worse cardiovascular outcomes, Ms. Zhu noted.
However, little is known about the prognostic impact of marital stress on 1-year health outcomes for younger people who survive an MI.
To investigate this, the researchers analyzed data from participants in the Variation in Recovery: Role of Gender on Outcomes of Young AMI Patients (VIRGO) study.
The current study comprised 1,593 adults, including 1,020 female participants (64%), who were treated for MI at 103 hospitals in 30 U.S. states.
VIRGO enrolled participants in a 2:1 female-to-male ratio so as to enrich the inclusion of women, Ms. Zhu explained.
In the study, “partnered” participants were individuals who self-reported as “living as married/living with a partner.” There were 126 such patients (8%) in the current study.
The mean age of the patients was 47, and about 90% were 40-55 years old. Three quarters were White, 13% were Black, and 7% were Hispanic.
Marital stress was assessed on the basis of patients’ replies to 17 questions in the Stockholm Marital Stress Scale regarding the quality of their emotional and sexual relationships with their spouses/partners.
The researchers divided patients into three groups on the basis of their marital stress: mild or absent (lowest quartile), moderate (second quartile), and severe (upper two quartiles).
At 1 year after their MI, patients replied to questionnaires that assessed their health, quality of life, and depressive and angina symptoms. Hospital readmissions were determined on the basis of self-reports and medical records.
Compared to participants who reported no or mild marital stress, those who reported severe mental stress had significantly worse scores for physical and mental health and generic and cardiovascular quality of life, after adjusting for baseline health and demographics. They had worse scores for mental health and quality of life, after further adjusting for socioeconomic status.
In the fully adjusted model, patients who reported severe marital stress were significantly more likely to report more frequent chest pain/angina (odds ratio, 1.49; 95% confidence interval, 1.06-2.10; P = .023) and to have been readmitted to hospital for any cause (OR, 1.45; 95% CI, 1.04-2.00; P = .006), compared with the patients who reported no or mild marital stress.
Study limitations include the fact that the findings are based on self-reported questionnaire replies; they may not be generalizable to patients in other countries; and they do not extend beyond a period of 1 year.
The researchers call for further research “to understand this complex relationship and potential causal pathway associated with these findings.”
“Additional stressors beyond marital stress, such as financial strain or work stress, may also play a role in young adults’ recovery, and the interaction between these factors require further research,” Ms. Zhu noted in a press release from the AHA.
The study was funded by Canadian Institutes of Health Research. The VIRGO study was funded by the National Heart, Lung, and Blood Institute. Ms. Zhu and Dr. Goldberg have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Severe marital stress was associated with worse recovery after myocardial infarction in a large U.S. cohort of married/partnered patients aged 55 years or younger.
Compared with patients who reported no or mild marital stress a month after their MI, patients who reported severe marital stress had worse physical and mental health, worse generic and cardiovascular quality of life, more frequent angina symptoms, and a greater likelihood of having a hospital readmission a year later.
These findings held true after adjusting for gender, age, race/ethnicity, and baseline health status (model 1) and after further adjusting for education and income levels and employment and insurance status (model 2).
A greater percentage of women than men reported having severe marital stress (39% vs. 30%; P = .001).
Cenjing Zhu, MPhil, a PhD candidate at Yale University, New Haven, Conn., and colleagues will present this study at the American Heart Association scientific sessions.
The results show that “both patients and care providers should be aware that stress experienced in one’s everyday life, such as marital stress, can affect AMI [acute MI] recovery,” Ms. Zhu said in an email.
Health care providers should consider incorporating screening for everyday stress during follow-up patient visits to better spot people at high risk of a poor recovery and further hospitalizations, she added. When possible, they could guide patients to resources to help them manage and reduce their stress levels.
According to Ms. Zhu, the findings suggest that “managing personal stress may be as important as managing other clinical risk factors during the recovery process.”
This study in younger patients with MI “shows that high levels of marital stress impair heart attack recovery, and women have greater impairment in their heart attack recovery compared to men,” AHA spokesperson Nieca Goldberg, MD, who was not involved with this research, told this news organization.
The study shows that “clinicians have to incorporate mental health as part of their assessment of all patients,” said Dr. Goldberg, a clinical associate professor of medicine at New York University and medical director of Atria New York City.
“Our mental health impacts our physical health,” she noted. “Questions about marital stress should be included as part of an overall assessment of mental health. This means assessing all patients for stress, anxiety, and depression.”
Patients who are experiencing marital stress should share the information with their doctor and discuss ways to be referred to therapists and cardiac rehabilitation providers, she said. “My final thought is, women have often been told that their cardiac symptoms are due to stress by doctors. Now we know stress impacts physical health and [is] no longer an excuse but a contributing factor to our physical health.”
Does marital stress affect young MI recovery?
Previous literature has linked psychological stress with worse cardiovascular outcomes, Ms. Zhu noted.
However, little is known about the prognostic impact of marital stress on 1-year health outcomes for younger people who survive an MI.
To investigate this, the researchers analyzed data from participants in the Variation in Recovery: Role of Gender on Outcomes of Young AMI Patients (VIRGO) study.
The current study comprised 1,593 adults, including 1,020 female participants (64%), who were treated for MI at 103 hospitals in 30 U.S. states.
VIRGO enrolled participants in a 2:1 female-to-male ratio so as to enrich the inclusion of women, Ms. Zhu explained.
In the study, “partnered” participants were individuals who self-reported as “living as married/living with a partner.” There were 126 such patients (8%) in the current study.
The mean age of the patients was 47, and about 90% were 40-55 years old. Three quarters were White, 13% were Black, and 7% were Hispanic.
Marital stress was assessed on the basis of patients’ replies to 17 questions in the Stockholm Marital Stress Scale regarding the quality of their emotional and sexual relationships with their spouses/partners.
The researchers divided patients into three groups on the basis of their marital stress: mild or absent (lowest quartile), moderate (second quartile), and severe (upper two quartiles).
At 1 year after their MI, patients replied to questionnaires that assessed their health, quality of life, and depressive and angina symptoms. Hospital readmissions were determined on the basis of self-reports and medical records.
Compared to participants who reported no or mild marital stress, those who reported severe mental stress had significantly worse scores for physical and mental health and generic and cardiovascular quality of life, after adjusting for baseline health and demographics. They had worse scores for mental health and quality of life, after further adjusting for socioeconomic status.
In the fully adjusted model, patients who reported severe marital stress were significantly more likely to report more frequent chest pain/angina (odds ratio, 1.49; 95% confidence interval, 1.06-2.10; P = .023) and to have been readmitted to hospital for any cause (OR, 1.45; 95% CI, 1.04-2.00; P = .006), compared with the patients who reported no or mild marital stress.
Study limitations include the fact that the findings are based on self-reported questionnaire replies; they may not be generalizable to patients in other countries; and they do not extend beyond a period of 1 year.
The researchers call for further research “to understand this complex relationship and potential causal pathway associated with these findings.”
“Additional stressors beyond marital stress, such as financial strain or work stress, may also play a role in young adults’ recovery, and the interaction between these factors require further research,” Ms. Zhu noted in a press release from the AHA.
The study was funded by Canadian Institutes of Health Research. The VIRGO study was funded by the National Heart, Lung, and Blood Institute. Ms. Zhu and Dr. Goldberg have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AHA 2022
The truth of alcohol consequences
Bad drinking consequence No. 87: Joining the LOTME team
Alcohol and college students go together like peanut butter and jelly. Or peanut butter and chocolate. Or peanut butter and toothpaste. Peanut butter goes with a lot of things.
Naturally, when you combine alcohol and college students, bad decisions are sure to follow. But have you ever wondered just how many bad decisions alcohol causes? A team of researchers from Penn State University, the undisputed champion of poor drinking decisions (trust us, we know), sure has. They’ve even conducted a 4-year study of 1,700 students as they carved a drunken swath through the many fine local drinking establishments, such as East Halls or that one frat house that hosts medieval battle–style ping pong tournaments.
The students were surveyed twice a year throughout the study, and the researchers compiled a list of all the various consequences their subjects experienced. Ultimately, college students will experience an average of 102 consequences from drinking during their 4-year college careers, which is an impressive number. Try thinking up a hundred consequences for anything.
Some consequences are less common than others – we imagine “missing the Renaissance Faire because you felt drunker the morning after than while you were drinking” is pretty low on the list – but more than 96% of students reported that they’d experienced a hangover and that drinking had caused them to say or do embarrassing things. Also, more than 70% said they needed additional alcohol to feel any effect, a potential sign of alcohol use disorder.
Once they had their list, the researchers focused on 12 of the more common and severe consequences, such as blacking out, hangovers, and missing work/class, and asked the study participants how their parents would react to their drinking and those specific consequences. Students who believed their parents would disapprove of alcohol-related consequences actually experienced fewer consequences overall.
College students, it seems, really do care what their parents think, even if they don’t express it, the researchers said. That gives space for parents to offer advice about the consequences of hard drinking, making decisions while drunk, or bringing godawful Fireball whiskey to parties. Seriously, don’t do that. Stuff’s bad, and you should feel bad for bringing it. Your parents raised you better than that.
COVID ‘expert’ discusses data sharing
We interrupt our regularly scheduled programming to bring you this special news event. Elon Musk, the world’s second-most annoying human, is holding a press conference to discuss, of all things, COVID-19.
Reporter: Hey, Mr. Musketeer, what qualifies you to talk about a global pandemic?
EM: As the official king of the Twitterverse, I’m pretty much an expert on any topic.
Reporter: Okay then, Mr. Muskmelon, what can you tell us about the new study in Agricultural Economics, which looked at consumers’ knowledge of local COVID infection rates and their willingness to eat at restaurants?
EM: Well, I know that one of the investigators, Rigoberto Lopez, PhD, of the University of Connecticut, said “no news is bad news.” Restaurants located in cities where local regulations required COVID tracking recovered faster than those in areas that did not, according to data from 87 restaurants in 10 Chinese cities that were gathered between Dec. 1, 2019, and March 27, 2020. Having access to local infection rate data made customers more comfortable going out to eat, the investigators explained.
Second reporter: Interesting, Mr. Muskox, but how about this headline from CNN: “Workers flee China’s biggest iPhone factory over Covid outbreak”? Do you agree with analysts, who said that “the chaos at Zhengzhou could jeopardize Apple and Foxconn’s output in the coming weeks,” as CNN put it?
EM: I did see that a manager at Foxconn, which owns the factory and is known to its friends as Hon Hai Precision Industry, told a Chinese media outlet that “workers are panicking over the spread of the virus at the factory and lack of access to official information.” As we’ve already discussed, no news is bad news.
That’s all the time I have to chat with you today. I’m off to fire some more Twitter employees.
In case you hadn’t already guessed, Vlad Putin is officially more annoying than Elon Musk. We now return to this week’s typical LOTME shenanigans, already in progress.
The deadliest month
With climate change making the world hotter, leading to more heat stroke and organ failure, you would think the summer months would be the most deadly. In reality, though, it’s quite the opposite.
There are multiple factors that make January the most deadly month out of the year, as LiveScience discovered in a recent analysis.
Let’s go through them, shall we?
Respiratory viruses: Robert Glatter, MD, of Lenox Hill Hospital in New York, told LiveScence that winter is the time for illnesses like the flu, bacterial pneumonia, and RSV. Millions of people worldwide die from the flu, according to the CDC. And the World Health Organization reported lower respiratory infections as the fourth-leading cause of death worldwide before COVID came along.
Heart disease: Heart conditions are actually more fatal in the winter months, according to a study published in Circulation. The cold puts more stress on the heart to keep the body warm, which can be a challenge for people who already have preexisting heart conditions.
Space heaters: Dr. Glatter also told Live Science that the use of space heaters could be a factor in the cold winter months since they can lead to carbon monoxide poisoning and even fires. Silent killers.
Holiday season: A time for joy and merriment, certainly, but Christmas et al. have their downsides. By January we’re coming off a 3-month food and alcohol binge, which leads to cardiac stress. There’s also the psychological stress that comes with the season. Sometimes the most wonderful time of the year just isn’t.
So even though summer is hot, fall has hurricanes, and spring tends to have the highest suicide rate, winter still ends up being the deadliest season.
Bad drinking consequence No. 87: Joining the LOTME team
Alcohol and college students go together like peanut butter and jelly. Or peanut butter and chocolate. Or peanut butter and toothpaste. Peanut butter goes with a lot of things.
Naturally, when you combine alcohol and college students, bad decisions are sure to follow. But have you ever wondered just how many bad decisions alcohol causes? A team of researchers from Penn State University, the undisputed champion of poor drinking decisions (trust us, we know), sure has. They’ve even conducted a 4-year study of 1,700 students as they carved a drunken swath through the many fine local drinking establishments, such as East Halls or that one frat house that hosts medieval battle–style ping pong tournaments.
The students were surveyed twice a year throughout the study, and the researchers compiled a list of all the various consequences their subjects experienced. Ultimately, college students will experience an average of 102 consequences from drinking during their 4-year college careers, which is an impressive number. Try thinking up a hundred consequences for anything.
Some consequences are less common than others – we imagine “missing the Renaissance Faire because you felt drunker the morning after than while you were drinking” is pretty low on the list – but more than 96% of students reported that they’d experienced a hangover and that drinking had caused them to say or do embarrassing things. Also, more than 70% said they needed additional alcohol to feel any effect, a potential sign of alcohol use disorder.
Once they had their list, the researchers focused on 12 of the more common and severe consequences, such as blacking out, hangovers, and missing work/class, and asked the study participants how their parents would react to their drinking and those specific consequences. Students who believed their parents would disapprove of alcohol-related consequences actually experienced fewer consequences overall.
College students, it seems, really do care what their parents think, even if they don’t express it, the researchers said. That gives space for parents to offer advice about the consequences of hard drinking, making decisions while drunk, or bringing godawful Fireball whiskey to parties. Seriously, don’t do that. Stuff’s bad, and you should feel bad for bringing it. Your parents raised you better than that.
COVID ‘expert’ discusses data sharing
We interrupt our regularly scheduled programming to bring you this special news event. Elon Musk, the world’s second-most annoying human, is holding a press conference to discuss, of all things, COVID-19.
Reporter: Hey, Mr. Musketeer, what qualifies you to talk about a global pandemic?
EM: As the official king of the Twitterverse, I’m pretty much an expert on any topic.
Reporter: Okay then, Mr. Muskmelon, what can you tell us about the new study in Agricultural Economics, which looked at consumers’ knowledge of local COVID infection rates and their willingness to eat at restaurants?
EM: Well, I know that one of the investigators, Rigoberto Lopez, PhD, of the University of Connecticut, said “no news is bad news.” Restaurants located in cities where local regulations required COVID tracking recovered faster than those in areas that did not, according to data from 87 restaurants in 10 Chinese cities that were gathered between Dec. 1, 2019, and March 27, 2020. Having access to local infection rate data made customers more comfortable going out to eat, the investigators explained.
Second reporter: Interesting, Mr. Muskox, but how about this headline from CNN: “Workers flee China’s biggest iPhone factory over Covid outbreak”? Do you agree with analysts, who said that “the chaos at Zhengzhou could jeopardize Apple and Foxconn’s output in the coming weeks,” as CNN put it?
EM: I did see that a manager at Foxconn, which owns the factory and is known to its friends as Hon Hai Precision Industry, told a Chinese media outlet that “workers are panicking over the spread of the virus at the factory and lack of access to official information.” As we’ve already discussed, no news is bad news.
That’s all the time I have to chat with you today. I’m off to fire some more Twitter employees.
In case you hadn’t already guessed, Vlad Putin is officially more annoying than Elon Musk. We now return to this week’s typical LOTME shenanigans, already in progress.
The deadliest month
With climate change making the world hotter, leading to more heat stroke and organ failure, you would think the summer months would be the most deadly. In reality, though, it’s quite the opposite.
There are multiple factors that make January the most deadly month out of the year, as LiveScience discovered in a recent analysis.
Let’s go through them, shall we?
Respiratory viruses: Robert Glatter, MD, of Lenox Hill Hospital in New York, told LiveScence that winter is the time for illnesses like the flu, bacterial pneumonia, and RSV. Millions of people worldwide die from the flu, according to the CDC. And the World Health Organization reported lower respiratory infections as the fourth-leading cause of death worldwide before COVID came along.
Heart disease: Heart conditions are actually more fatal in the winter months, according to a study published in Circulation. The cold puts more stress on the heart to keep the body warm, which can be a challenge for people who already have preexisting heart conditions.
Space heaters: Dr. Glatter also told Live Science that the use of space heaters could be a factor in the cold winter months since they can lead to carbon monoxide poisoning and even fires. Silent killers.
Holiday season: A time for joy and merriment, certainly, but Christmas et al. have their downsides. By January we’re coming off a 3-month food and alcohol binge, which leads to cardiac stress. There’s also the psychological stress that comes with the season. Sometimes the most wonderful time of the year just isn’t.
So even though summer is hot, fall has hurricanes, and spring tends to have the highest suicide rate, winter still ends up being the deadliest season.
Bad drinking consequence No. 87: Joining the LOTME team
Alcohol and college students go together like peanut butter and jelly. Or peanut butter and chocolate. Or peanut butter and toothpaste. Peanut butter goes with a lot of things.
Naturally, when you combine alcohol and college students, bad decisions are sure to follow. But have you ever wondered just how many bad decisions alcohol causes? A team of researchers from Penn State University, the undisputed champion of poor drinking decisions (trust us, we know), sure has. They’ve even conducted a 4-year study of 1,700 students as they carved a drunken swath through the many fine local drinking establishments, such as East Halls or that one frat house that hosts medieval battle–style ping pong tournaments.
The students were surveyed twice a year throughout the study, and the researchers compiled a list of all the various consequences their subjects experienced. Ultimately, college students will experience an average of 102 consequences from drinking during their 4-year college careers, which is an impressive number. Try thinking up a hundred consequences for anything.
Some consequences are less common than others – we imagine “missing the Renaissance Faire because you felt drunker the morning after than while you were drinking” is pretty low on the list – but more than 96% of students reported that they’d experienced a hangover and that drinking had caused them to say or do embarrassing things. Also, more than 70% said they needed additional alcohol to feel any effect, a potential sign of alcohol use disorder.
Once they had their list, the researchers focused on 12 of the more common and severe consequences, such as blacking out, hangovers, and missing work/class, and asked the study participants how their parents would react to their drinking and those specific consequences. Students who believed their parents would disapprove of alcohol-related consequences actually experienced fewer consequences overall.
College students, it seems, really do care what their parents think, even if they don’t express it, the researchers said. That gives space for parents to offer advice about the consequences of hard drinking, making decisions while drunk, or bringing godawful Fireball whiskey to parties. Seriously, don’t do that. Stuff’s bad, and you should feel bad for bringing it. Your parents raised you better than that.
COVID ‘expert’ discusses data sharing
We interrupt our regularly scheduled programming to bring you this special news event. Elon Musk, the world’s second-most annoying human, is holding a press conference to discuss, of all things, COVID-19.
Reporter: Hey, Mr. Musketeer, what qualifies you to talk about a global pandemic?
EM: As the official king of the Twitterverse, I’m pretty much an expert on any topic.
Reporter: Okay then, Mr. Muskmelon, what can you tell us about the new study in Agricultural Economics, which looked at consumers’ knowledge of local COVID infection rates and their willingness to eat at restaurants?
EM: Well, I know that one of the investigators, Rigoberto Lopez, PhD, of the University of Connecticut, said “no news is bad news.” Restaurants located in cities where local regulations required COVID tracking recovered faster than those in areas that did not, according to data from 87 restaurants in 10 Chinese cities that were gathered between Dec. 1, 2019, and March 27, 2020. Having access to local infection rate data made customers more comfortable going out to eat, the investigators explained.
Second reporter: Interesting, Mr. Muskox, but how about this headline from CNN: “Workers flee China’s biggest iPhone factory over Covid outbreak”? Do you agree with analysts, who said that “the chaos at Zhengzhou could jeopardize Apple and Foxconn’s output in the coming weeks,” as CNN put it?
EM: I did see that a manager at Foxconn, which owns the factory and is known to its friends as Hon Hai Precision Industry, told a Chinese media outlet that “workers are panicking over the spread of the virus at the factory and lack of access to official information.” As we’ve already discussed, no news is bad news.
That’s all the time I have to chat with you today. I’m off to fire some more Twitter employees.
In case you hadn’t already guessed, Vlad Putin is officially more annoying than Elon Musk. We now return to this week’s typical LOTME shenanigans, already in progress.
The deadliest month
With climate change making the world hotter, leading to more heat stroke and organ failure, you would think the summer months would be the most deadly. In reality, though, it’s quite the opposite.
There are multiple factors that make January the most deadly month out of the year, as LiveScience discovered in a recent analysis.
Let’s go through them, shall we?
Respiratory viruses: Robert Glatter, MD, of Lenox Hill Hospital in New York, told LiveScence that winter is the time for illnesses like the flu, bacterial pneumonia, and RSV. Millions of people worldwide die from the flu, according to the CDC. And the World Health Organization reported lower respiratory infections as the fourth-leading cause of death worldwide before COVID came along.
Heart disease: Heart conditions are actually more fatal in the winter months, according to a study published in Circulation. The cold puts more stress on the heart to keep the body warm, which can be a challenge for people who already have preexisting heart conditions.
Space heaters: Dr. Glatter also told Live Science that the use of space heaters could be a factor in the cold winter months since they can lead to carbon monoxide poisoning and even fires. Silent killers.
Holiday season: A time for joy and merriment, certainly, but Christmas et al. have their downsides. By January we’re coming off a 3-month food and alcohol binge, which leads to cardiac stress. There’s also the psychological stress that comes with the season. Sometimes the most wonderful time of the year just isn’t.
So even though summer is hot, fall has hurricanes, and spring tends to have the highest suicide rate, winter still ends up being the deadliest season.