User login
CCSs have increased risk of hypertension
A study of childhood cancer survivors (CCSs) suggests these individuals have an increased risk of developing hypertension as adults.
The CCSs studied had more than double the rate of hypertension observed in the matched general population.
Sex, age, race, and weight were all significantly associated with hypertension among CCSs, but most treatment types were not.
The exception was nephrectomy, which was associated with an increased risk of hypertension.
Todd M. Gibson, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted this research and reported the results in Cancer Epidemiology, Biomarkers & Prevention.
“High blood pressure is an important modifiable risk factor that increases the risk of heart problems in everyone,” Dr Gibson said. “Research has shown that high blood pressure can have an even greater negative impact on survivors of childhood cancer who were treated with cardiotoxic therapies such as anthracyclines or chest radiation.”
To assess the prevalence of hypertension among CCSs, Dr Gibson and his colleagues examined 3016 adults who were 10-year survivors of childhood cancers. The subjects were enrolled in the St. Jude Lifetime Cohort Study, which provides ongoing medical assessments of CCSs to advance knowledge of their long-term health outcomes.
The subjects’ mean age at the initial study assessment was 32, and 52% were male. Most (83%) were non-Hispanic white, 14% were non-Hispanic black, 2% were Hispanic, and 1% were “other.”
Thirty-seven percent of subjects had leukemia, 12% had Hodgkin lymphoma, and 7% had non-Hodgkin lymphoma.
Eighty-six percent of subjects had received chemotherapy, and 59% received radiation.
Results
Subjects were considered to have hypertension if their systolic blood pressure was 140 or greater, their diastolic blood pressure was 90 or greater, or if they had been previously diagnosed with hypertension and were taking antihypertensive medication.
The prevalence of hypertension was 2.6 times higher among CCSs than expected, based on age-, sex-, race- and body mass index-specific rates in the general population.
In addition, the incidence of hypertension increased for CCSs over time. Thirteen percent of CCSs had hypertension at age 30, 37% had it at age 40, and more than 70% had it at age 50.
Dr Gibson said rates of hypertension in CCSs matched rates in the general population of people about a decade older.
The researchers identified several factors that were significantly associated with hypertension among CCSs, including:
- Male sex (odd ratio [OR], 1.38; 95% CI, 1.14–1.67)
- Non-Hispanic black race (OR, 1.66; 95% CI, 1.28–2.16)
- Older age at assessment (OR per 1 year of age, 1.10; 95% CI, 1.08–1.11)
- Being overweight (OR, 1.58; 95% CI, 1.21–2.07)
- Obesity (OR, 3.02; 95% CI, 2.34–3.88).
Exposure to any type of radiation or chemotherapy was not significantly associated with hypertension, but nephrectomy was (OR, 1.68; 95% CI, 1.11–2.53).
Dr Gibson said the lack of an association between hypertension and radiation/chemotherapy was surprising. It suggests the connection between childhood cancer survival and adult hypertension is multifactorial and worthy of future research.
In the meantime, he said, clinicians should be mindful that CCSs are more likely than the general public to develop hypertension.
“The good news is that, unlike prior cancer therapy, high blood pressure is a modifiable risk factor,” Dr Gibson noted. “Research is needed to identify effective interventions to prevent hypertension in survivors, but our results emphasize the importance of blood pressure surveillance and management.”
Dr Gibson said a limitation of this study is that it was based on blood pressure measurements taken at a single study visit. A clinical diagnosis of hypertension typically requires measurements taken at multiple intervals.
In addition, the St. Jude Lifetime Cohort is a group of CCSs who undergo frequent clinical follow-up, so its participants may have benefited from being monitored and may therefore be in better health than CCSs who have less comprehensive follow-up. ![]()
A study of childhood cancer survivors (CCSs) suggests these individuals have an increased risk of developing hypertension as adults.
The CCSs studied had more than double the rate of hypertension observed in the matched general population.
Sex, age, race, and weight were all significantly associated with hypertension among CCSs, but most treatment types were not.
The exception was nephrectomy, which was associated with an increased risk of hypertension.
Todd M. Gibson, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted this research and reported the results in Cancer Epidemiology, Biomarkers & Prevention.
“High blood pressure is an important modifiable risk factor that increases the risk of heart problems in everyone,” Dr Gibson said. “Research has shown that high blood pressure can have an even greater negative impact on survivors of childhood cancer who were treated with cardiotoxic therapies such as anthracyclines or chest radiation.”
To assess the prevalence of hypertension among CCSs, Dr Gibson and his colleagues examined 3016 adults who were 10-year survivors of childhood cancers. The subjects were enrolled in the St. Jude Lifetime Cohort Study, which provides ongoing medical assessments of CCSs to advance knowledge of their long-term health outcomes.
The subjects’ mean age at the initial study assessment was 32, and 52% were male. Most (83%) were non-Hispanic white, 14% were non-Hispanic black, 2% were Hispanic, and 1% were “other.”
Thirty-seven percent of subjects had leukemia, 12% had Hodgkin lymphoma, and 7% had non-Hodgkin lymphoma.
Eighty-six percent of subjects had received chemotherapy, and 59% received radiation.
Results
Subjects were considered to have hypertension if their systolic blood pressure was 140 or greater, their diastolic blood pressure was 90 or greater, or if they had been previously diagnosed with hypertension and were taking antihypertensive medication.
The prevalence of hypertension was 2.6 times higher among CCSs than expected, based on age-, sex-, race- and body mass index-specific rates in the general population.
In addition, the incidence of hypertension increased for CCSs over time. Thirteen percent of CCSs had hypertension at age 30, 37% had it at age 40, and more than 70% had it at age 50.
Dr Gibson said rates of hypertension in CCSs matched rates in the general population of people about a decade older.
The researchers identified several factors that were significantly associated with hypertension among CCSs, including:
- Male sex (odd ratio [OR], 1.38; 95% CI, 1.14–1.67)
- Non-Hispanic black race (OR, 1.66; 95% CI, 1.28–2.16)
- Older age at assessment (OR per 1 year of age, 1.10; 95% CI, 1.08–1.11)
- Being overweight (OR, 1.58; 95% CI, 1.21–2.07)
- Obesity (OR, 3.02; 95% CI, 2.34–3.88).
Exposure to any type of radiation or chemotherapy was not significantly associated with hypertension, but nephrectomy was (OR, 1.68; 95% CI, 1.11–2.53).
Dr Gibson said the lack of an association between hypertension and radiation/chemotherapy was surprising. It suggests the connection between childhood cancer survival and adult hypertension is multifactorial and worthy of future research.
In the meantime, he said, clinicians should be mindful that CCSs are more likely than the general public to develop hypertension.
“The good news is that, unlike prior cancer therapy, high blood pressure is a modifiable risk factor,” Dr Gibson noted. “Research is needed to identify effective interventions to prevent hypertension in survivors, but our results emphasize the importance of blood pressure surveillance and management.”
Dr Gibson said a limitation of this study is that it was based on blood pressure measurements taken at a single study visit. A clinical diagnosis of hypertension typically requires measurements taken at multiple intervals.
In addition, the St. Jude Lifetime Cohort is a group of CCSs who undergo frequent clinical follow-up, so its participants may have benefited from being monitored and may therefore be in better health than CCSs who have less comprehensive follow-up. ![]()
A study of childhood cancer survivors (CCSs) suggests these individuals have an increased risk of developing hypertension as adults.
The CCSs studied had more than double the rate of hypertension observed in the matched general population.
Sex, age, race, and weight were all significantly associated with hypertension among CCSs, but most treatment types were not.
The exception was nephrectomy, which was associated with an increased risk of hypertension.
Todd M. Gibson, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted this research and reported the results in Cancer Epidemiology, Biomarkers & Prevention.
“High blood pressure is an important modifiable risk factor that increases the risk of heart problems in everyone,” Dr Gibson said. “Research has shown that high blood pressure can have an even greater negative impact on survivors of childhood cancer who were treated with cardiotoxic therapies such as anthracyclines or chest radiation.”
To assess the prevalence of hypertension among CCSs, Dr Gibson and his colleagues examined 3016 adults who were 10-year survivors of childhood cancers. The subjects were enrolled in the St. Jude Lifetime Cohort Study, which provides ongoing medical assessments of CCSs to advance knowledge of their long-term health outcomes.
The subjects’ mean age at the initial study assessment was 32, and 52% were male. Most (83%) were non-Hispanic white, 14% were non-Hispanic black, 2% were Hispanic, and 1% were “other.”
Thirty-seven percent of subjects had leukemia, 12% had Hodgkin lymphoma, and 7% had non-Hodgkin lymphoma.
Eighty-six percent of subjects had received chemotherapy, and 59% received radiation.
Results
Subjects were considered to have hypertension if their systolic blood pressure was 140 or greater, their diastolic blood pressure was 90 or greater, or if they had been previously diagnosed with hypertension and were taking antihypertensive medication.
The prevalence of hypertension was 2.6 times higher among CCSs than expected, based on age-, sex-, race- and body mass index-specific rates in the general population.
In addition, the incidence of hypertension increased for CCSs over time. Thirteen percent of CCSs had hypertension at age 30, 37% had it at age 40, and more than 70% had it at age 50.
Dr Gibson said rates of hypertension in CCSs matched rates in the general population of people about a decade older.
The researchers identified several factors that were significantly associated with hypertension among CCSs, including:
- Male sex (odd ratio [OR], 1.38; 95% CI, 1.14–1.67)
- Non-Hispanic black race (OR, 1.66; 95% CI, 1.28–2.16)
- Older age at assessment (OR per 1 year of age, 1.10; 95% CI, 1.08–1.11)
- Being overweight (OR, 1.58; 95% CI, 1.21–2.07)
- Obesity (OR, 3.02; 95% CI, 2.34–3.88).
Exposure to any type of radiation or chemotherapy was not significantly associated with hypertension, but nephrectomy was (OR, 1.68; 95% CI, 1.11–2.53).
Dr Gibson said the lack of an association between hypertension and radiation/chemotherapy was surprising. It suggests the connection between childhood cancer survival and adult hypertension is multifactorial and worthy of future research.
In the meantime, he said, clinicians should be mindful that CCSs are more likely than the general public to develop hypertension.
“The good news is that, unlike prior cancer therapy, high blood pressure is a modifiable risk factor,” Dr Gibson noted. “Research is needed to identify effective interventions to prevent hypertension in survivors, but our results emphasize the importance of blood pressure surveillance and management.”
Dr Gibson said a limitation of this study is that it was based on blood pressure measurements taken at a single study visit. A clinical diagnosis of hypertension typically requires measurements taken at multiple intervals.
In addition, the St. Jude Lifetime Cohort is a group of CCSs who undergo frequent clinical follow-up, so its participants may have benefited from being monitored and may therefore be in better health than CCSs who have less comprehensive follow-up. ![]()
CAR T-cells gain ground against hematologic cancers
Chimeric antigen receptor (CAR) T-cell therapies are driving ever faster forward, with impressive response rates – including a high level of complete responses – in treatment of patients with lymphomas and multiple myeloma as shown by clinical trial results to be presented at the annual meeting of the American Society of Hematology.
Investigators will be presenting data on the first two CAR T constructs to receive Food and Drug Administration approval, each directed against CD19.
ZUMA-1 for non-Hodgkin lymphoma
Long-term follow-up results with the use of axicabtagene ciloleucel (Yescarta; axi-cel) in patients with refractory aggressive non-Hodgkin lymphoma will be presented by Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center in Houston, on behalf of colleagues in the ZUMA-1 trial (Abstract 578).
At the 2017 European Hematology Association Congress in Madrid, ZUMA-1 investigators reported that axi-cel, an autologous anti-CD19 CAR-T product, was associated with an 82% objective response rate (ORR), including 54% complete responses, in patients with refractory diffuse large B cell lymphoma (DLBCL), primary mediastinal B-cell lymphoma, or transformed follicular lymphoma. The 54% complete response rate was nearly sevenfold higher than that reported for historical controls, according to coinvestigator Yi Lin, MD, PhD, of the Mayo Clinic, Rochester, Minn.
The treatment was generally safe, with 13% of patients experiencing grade 3 or greater cytokine release syndrome (CRS), and 28% having grade 3 or greater neurologic events. The events were generally reversible, and the rates of each declined over time. The use of tocilizumab or steroids to control adverse events did not have a negative effect on responses.
This CAR T-cell construct received FDA approval in October 2017.
At ASH 2017, Dr. Neelapu will present 1-year follow-up data from the trial which will include both data on responses and toxicity, “but also some very interesting information on mechanisms of resistance, whether patients’ tumor cells become CD19 negative, or checkpoint, like PD-L1, positive,” said Kenneth Anderson, MD, of the Dana-Farber Cancer Institute in Boston, who discussed noteworthy abstracts in a media briefing prior to the meeting.
JULIET for DLBCL
Data on the use of another anti-CD19 CAR construct, tisagenlecleucel (Kymriah) in adults with relapsed or refractory DLBCL will be presented by Stephen J. Schuster, MD, of the University of Pennsylvania, Philadelphia, on behalf of investigators in the JULIET trial.
At the 2017 European Hematology Association Congress, coinvestigator Gilles Salles, MD, PhD, of the University of Lyon, France, reported an interim analysis from the trial, showing that the CAR T construct was associated with a 59% ORR, consisting of 43% complete responses and 16% partial responses in patients with relapsed or refractory DLBCL. The analysis “confirms the high response rates and durable responses observed in the previous single-center trial,” Dr. Salles said.
Dr. Schuster will present the primary analysis of the global phase 2 pivotal trial at ASH 2017 (Abstract 577), looking at patients who received the product from the U.S. manufacturing site.
As of the data cutoff in March 2017, the best ORR among 81 patients with at least 3 months of follow-up, or earlier discontinuation, was 53.1% with 39.5% complete responses and 13.6% partial responses. For 46 patients evaluable at 6 months, the complete response rate was 30% and partial response rate was 7%.
The response rates were consistent across subgroups, including patients who had previously undergone autologous stem cell transplant and those with double-hit lymphoma (i.e, with mutation in MYC and BCL2 or BCL6).
The responses also appeared to be durable, with the median duration not reached. The 6-month probability of being relapse free was 73.5%. Similarly, median overall survival was not reached; the 6-month probability of overall survival was 64.5%, according to the published abstract.
Adverse events included CRS in 58% of infused patients, with 15% grade 3 and 8% grade 4 in severity. CRS was managed according to protocol with tocilizumab and/or corticosteroids. There were no deaths associated with either the CRS or neurologic toxicities.
“This is a multicenter trial: 27 centers, 10 countries, 4 continents, so it sort of demonstrates to all of us that this technology can be done in an international scope,” Dr. Anderson said.
Anti-BCMA for multiple myeloma
CAR T-cells are relative newcomers in the treatment of multiple myeloma, but James N. Kochenderfer, MD, of the National Cancer Institute, Bethesda, Md., will be presenting updated results from a multicenter study of bb2121, a CAR T-cell construct directed against B-cell maturation antigen (BCMA).
BCMA was first described in myeloma in 2004 as a mechanism for the growth and survival of malignant plasma cells. As previously reported, a different anti-BCMA T-cell product induced clinical remissions in 33 of 35 patients with relapsed/refractory multiple myeloma who were treated in an early clinical trial.
With the construct being investigated by Dr. Kochenderfer and colleagues, 21 patients (median 58 years old) with a median of 5 years since a diagnosis of multiple myeloma were infused with bb2121. One-month data on clinical response was available for 18 patients at the time of data cutoff in May 2017 (Abstract 740).
In this heavily pretreated population, the ORR was 89% overall, and was 100% for patients treated with doses of 150 x 106 CAR+ T cells or higher. At this dose range or higher no patients had disease progression at up to 54 weeks after infusion. Of the four patients evaluable for minimal residual disease (MRD), all were MRD negative.
As of the data cut-off there were no dose-limiting toxicities and no treatment-related grade 3 or higher neurotoxicities. CRS was primarily grade 1 or 2 and was reported in 15 of 21 patients. Two patients had grade 3 CRS that resolved in 24 hours, and four patients received tocilizumab (one with steroids) for CRS management.
There was one on-study death, from cardiopulmonary arrest more than 4 months after bb2121 infusion in a patient with an extensive cardiac history. The death was judged to be unrelated to CAR T-cell therapy.
Dr. Kochenderfer will present data and analysis on an additional 5 months of follow-up.
“This abstract demonstrates that again, in patients, this time with myeloma with literally no other option, you can achieve very impressive response,” Dr. Anderson said.
The ZUMA-1 study is funded by Kite Pharma. Dr. Neelapu reported research funding from BMS, Poseida, Merck, Kite Pharma, and Cellectis. He reported consultancy and membership on the board of directors or advisory committees for Merck, Kite Pharma, and Celgene.
The JULIET study is funded by Novartis. Dr. Schuster reported consultancy and research funding from Novartis Pharmaceuticals, Celgene, Genentech, Bristol-Myers Squibb, Janssen R&D, and Gilead. He reported consultancy with Seattle Genetics and Nordic Nanovector, and research funding from Merck.
The multicenter study of bb2121 is sponsored by bluebird bio and Celgene. Dr. Kochenderfer reported research funding from bluebird bio and Kite Pharma. He reported having multiple patents in the CAR field.
Abstract 578 Long-Term Follow-up ZUMA-1: A Pivotal Trial of Axicabtagene Ciloleucel (Axi-Cel; KTE-C19) in Patients with Refractory Aggressive Non-Hodgkin Lymphoma (NHL) will be presented in session 626, Monday, Dec. 11, 2017, at 7:15 a.m.
Abstract 577 Primary Analysis of Juliet: A Global, Pivotal, Phase 2 Trial of CTL019 in Adult Patients with Relapsed or Refractory Diffuse Large B-Cell Lymphoma will be presented in session 626, Monday, Dec. 11, 2017 at 7:00 a.m.
Abstract 740 Durable Clinical Responses in Heavily Pretreated Patients with Relapsed/Refractory Multiple Myeloma: Updated Results from a Multicenter Study of bb2121 Anti-BCMA CAR T Cell Therapy will be presented in session 653, Monday, Dec. 11, 2017 at 3:00 p.m.
Chimeric antigen receptor (CAR) T-cell therapies are driving ever faster forward, with impressive response rates – including a high level of complete responses – in treatment of patients with lymphomas and multiple myeloma as shown by clinical trial results to be presented at the annual meeting of the American Society of Hematology.
Investigators will be presenting data on the first two CAR T constructs to receive Food and Drug Administration approval, each directed against CD19.
ZUMA-1 for non-Hodgkin lymphoma
Long-term follow-up results with the use of axicabtagene ciloleucel (Yescarta; axi-cel) in patients with refractory aggressive non-Hodgkin lymphoma will be presented by Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center in Houston, on behalf of colleagues in the ZUMA-1 trial (Abstract 578).
At the 2017 European Hematology Association Congress in Madrid, ZUMA-1 investigators reported that axi-cel, an autologous anti-CD19 CAR-T product, was associated with an 82% objective response rate (ORR), including 54% complete responses, in patients with refractory diffuse large B cell lymphoma (DLBCL), primary mediastinal B-cell lymphoma, or transformed follicular lymphoma. The 54% complete response rate was nearly sevenfold higher than that reported for historical controls, according to coinvestigator Yi Lin, MD, PhD, of the Mayo Clinic, Rochester, Minn.
The treatment was generally safe, with 13% of patients experiencing grade 3 or greater cytokine release syndrome (CRS), and 28% having grade 3 or greater neurologic events. The events were generally reversible, and the rates of each declined over time. The use of tocilizumab or steroids to control adverse events did not have a negative effect on responses.
This CAR T-cell construct received FDA approval in October 2017.
At ASH 2017, Dr. Neelapu will present 1-year follow-up data from the trial which will include both data on responses and toxicity, “but also some very interesting information on mechanisms of resistance, whether patients’ tumor cells become CD19 negative, or checkpoint, like PD-L1, positive,” said Kenneth Anderson, MD, of the Dana-Farber Cancer Institute in Boston, who discussed noteworthy abstracts in a media briefing prior to the meeting.
JULIET for DLBCL
Data on the use of another anti-CD19 CAR construct, tisagenlecleucel (Kymriah) in adults with relapsed or refractory DLBCL will be presented by Stephen J. Schuster, MD, of the University of Pennsylvania, Philadelphia, on behalf of investigators in the JULIET trial.
At the 2017 European Hematology Association Congress, coinvestigator Gilles Salles, MD, PhD, of the University of Lyon, France, reported an interim analysis from the trial, showing that the CAR T construct was associated with a 59% ORR, consisting of 43% complete responses and 16% partial responses in patients with relapsed or refractory DLBCL. The analysis “confirms the high response rates and durable responses observed in the previous single-center trial,” Dr. Salles said.
Dr. Schuster will present the primary analysis of the global phase 2 pivotal trial at ASH 2017 (Abstract 577), looking at patients who received the product from the U.S. manufacturing site.
As of the data cutoff in March 2017, the best ORR among 81 patients with at least 3 months of follow-up, or earlier discontinuation, was 53.1% with 39.5% complete responses and 13.6% partial responses. For 46 patients evaluable at 6 months, the complete response rate was 30% and partial response rate was 7%.
The response rates were consistent across subgroups, including patients who had previously undergone autologous stem cell transplant and those with double-hit lymphoma (i.e, with mutation in MYC and BCL2 or BCL6).
The responses also appeared to be durable, with the median duration not reached. The 6-month probability of being relapse free was 73.5%. Similarly, median overall survival was not reached; the 6-month probability of overall survival was 64.5%, according to the published abstract.
Adverse events included CRS in 58% of infused patients, with 15% grade 3 and 8% grade 4 in severity. CRS was managed according to protocol with tocilizumab and/or corticosteroids. There were no deaths associated with either the CRS or neurologic toxicities.
“This is a multicenter trial: 27 centers, 10 countries, 4 continents, so it sort of demonstrates to all of us that this technology can be done in an international scope,” Dr. Anderson said.
Anti-BCMA for multiple myeloma
CAR T-cells are relative newcomers in the treatment of multiple myeloma, but James N. Kochenderfer, MD, of the National Cancer Institute, Bethesda, Md., will be presenting updated results from a multicenter study of bb2121, a CAR T-cell construct directed against B-cell maturation antigen (BCMA).
BCMA was first described in myeloma in 2004 as a mechanism for the growth and survival of malignant plasma cells. As previously reported, a different anti-BCMA T-cell product induced clinical remissions in 33 of 35 patients with relapsed/refractory multiple myeloma who were treated in an early clinical trial.
With the construct being investigated by Dr. Kochenderfer and colleagues, 21 patients (median 58 years old) with a median of 5 years since a diagnosis of multiple myeloma were infused with bb2121. One-month data on clinical response was available for 18 patients at the time of data cutoff in May 2017 (Abstract 740).
In this heavily pretreated population, the ORR was 89% overall, and was 100% for patients treated with doses of 150 x 106 CAR+ T cells or higher. At this dose range or higher no patients had disease progression at up to 54 weeks after infusion. Of the four patients evaluable for minimal residual disease (MRD), all were MRD negative.
As of the data cut-off there were no dose-limiting toxicities and no treatment-related grade 3 or higher neurotoxicities. CRS was primarily grade 1 or 2 and was reported in 15 of 21 patients. Two patients had grade 3 CRS that resolved in 24 hours, and four patients received tocilizumab (one with steroids) for CRS management.
There was one on-study death, from cardiopulmonary arrest more than 4 months after bb2121 infusion in a patient with an extensive cardiac history. The death was judged to be unrelated to CAR T-cell therapy.
Dr. Kochenderfer will present data and analysis on an additional 5 months of follow-up.
“This abstract demonstrates that again, in patients, this time with myeloma with literally no other option, you can achieve very impressive response,” Dr. Anderson said.
The ZUMA-1 study is funded by Kite Pharma. Dr. Neelapu reported research funding from BMS, Poseida, Merck, Kite Pharma, and Cellectis. He reported consultancy and membership on the board of directors or advisory committees for Merck, Kite Pharma, and Celgene.
The JULIET study is funded by Novartis. Dr. Schuster reported consultancy and research funding from Novartis Pharmaceuticals, Celgene, Genentech, Bristol-Myers Squibb, Janssen R&D, and Gilead. He reported consultancy with Seattle Genetics and Nordic Nanovector, and research funding from Merck.
The multicenter study of bb2121 is sponsored by bluebird bio and Celgene. Dr. Kochenderfer reported research funding from bluebird bio and Kite Pharma. He reported having multiple patents in the CAR field.
Abstract 578 Long-Term Follow-up ZUMA-1: A Pivotal Trial of Axicabtagene Ciloleucel (Axi-Cel; KTE-C19) in Patients with Refractory Aggressive Non-Hodgkin Lymphoma (NHL) will be presented in session 626, Monday, Dec. 11, 2017, at 7:15 a.m.
Abstract 577 Primary Analysis of Juliet: A Global, Pivotal, Phase 2 Trial of CTL019 in Adult Patients with Relapsed or Refractory Diffuse Large B-Cell Lymphoma will be presented in session 626, Monday, Dec. 11, 2017 at 7:00 a.m.
Abstract 740 Durable Clinical Responses in Heavily Pretreated Patients with Relapsed/Refractory Multiple Myeloma: Updated Results from a Multicenter Study of bb2121 Anti-BCMA CAR T Cell Therapy will be presented in session 653, Monday, Dec. 11, 2017 at 3:00 p.m.
Chimeric antigen receptor (CAR) T-cell therapies are driving ever faster forward, with impressive response rates – including a high level of complete responses – in treatment of patients with lymphomas and multiple myeloma as shown by clinical trial results to be presented at the annual meeting of the American Society of Hematology.
Investigators will be presenting data on the first two CAR T constructs to receive Food and Drug Administration approval, each directed against CD19.
ZUMA-1 for non-Hodgkin lymphoma
Long-term follow-up results with the use of axicabtagene ciloleucel (Yescarta; axi-cel) in patients with refractory aggressive non-Hodgkin lymphoma will be presented by Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center in Houston, on behalf of colleagues in the ZUMA-1 trial (Abstract 578).
At the 2017 European Hematology Association Congress in Madrid, ZUMA-1 investigators reported that axi-cel, an autologous anti-CD19 CAR-T product, was associated with an 82% objective response rate (ORR), including 54% complete responses, in patients with refractory diffuse large B cell lymphoma (DLBCL), primary mediastinal B-cell lymphoma, or transformed follicular lymphoma. The 54% complete response rate was nearly sevenfold higher than that reported for historical controls, according to coinvestigator Yi Lin, MD, PhD, of the Mayo Clinic, Rochester, Minn.
The treatment was generally safe, with 13% of patients experiencing grade 3 or greater cytokine release syndrome (CRS), and 28% having grade 3 or greater neurologic events. The events were generally reversible, and the rates of each declined over time. The use of tocilizumab or steroids to control adverse events did not have a negative effect on responses.
This CAR T-cell construct received FDA approval in October 2017.
At ASH 2017, Dr. Neelapu will present 1-year follow-up data from the trial which will include both data on responses and toxicity, “but also some very interesting information on mechanisms of resistance, whether patients’ tumor cells become CD19 negative, or checkpoint, like PD-L1, positive,” said Kenneth Anderson, MD, of the Dana-Farber Cancer Institute in Boston, who discussed noteworthy abstracts in a media briefing prior to the meeting.
JULIET for DLBCL
Data on the use of another anti-CD19 CAR construct, tisagenlecleucel (Kymriah) in adults with relapsed or refractory DLBCL will be presented by Stephen J. Schuster, MD, of the University of Pennsylvania, Philadelphia, on behalf of investigators in the JULIET trial.
At the 2017 European Hematology Association Congress, coinvestigator Gilles Salles, MD, PhD, of the University of Lyon, France, reported an interim analysis from the trial, showing that the CAR T construct was associated with a 59% ORR, consisting of 43% complete responses and 16% partial responses in patients with relapsed or refractory DLBCL. The analysis “confirms the high response rates and durable responses observed in the previous single-center trial,” Dr. Salles said.
Dr. Schuster will present the primary analysis of the global phase 2 pivotal trial at ASH 2017 (Abstract 577), looking at patients who received the product from the U.S. manufacturing site.
As of the data cutoff in March 2017, the best ORR among 81 patients with at least 3 months of follow-up, or earlier discontinuation, was 53.1% with 39.5% complete responses and 13.6% partial responses. For 46 patients evaluable at 6 months, the complete response rate was 30% and partial response rate was 7%.
The response rates were consistent across subgroups, including patients who had previously undergone autologous stem cell transplant and those with double-hit lymphoma (i.e, with mutation in MYC and BCL2 or BCL6).
The responses also appeared to be durable, with the median duration not reached. The 6-month probability of being relapse free was 73.5%. Similarly, median overall survival was not reached; the 6-month probability of overall survival was 64.5%, according to the published abstract.
Adverse events included CRS in 58% of infused patients, with 15% grade 3 and 8% grade 4 in severity. CRS was managed according to protocol with tocilizumab and/or corticosteroids. There were no deaths associated with either the CRS or neurologic toxicities.
“This is a multicenter trial: 27 centers, 10 countries, 4 continents, so it sort of demonstrates to all of us that this technology can be done in an international scope,” Dr. Anderson said.
Anti-BCMA for multiple myeloma
CAR T-cells are relative newcomers in the treatment of multiple myeloma, but James N. Kochenderfer, MD, of the National Cancer Institute, Bethesda, Md., will be presenting updated results from a multicenter study of bb2121, a CAR T-cell construct directed against B-cell maturation antigen (BCMA).
BCMA was first described in myeloma in 2004 as a mechanism for the growth and survival of malignant plasma cells. As previously reported, a different anti-BCMA T-cell product induced clinical remissions in 33 of 35 patients with relapsed/refractory multiple myeloma who were treated in an early clinical trial.
With the construct being investigated by Dr. Kochenderfer and colleagues, 21 patients (median 58 years old) with a median of 5 years since a diagnosis of multiple myeloma were infused with bb2121. One-month data on clinical response was available for 18 patients at the time of data cutoff in May 2017 (Abstract 740).
In this heavily pretreated population, the ORR was 89% overall, and was 100% for patients treated with doses of 150 x 106 CAR+ T cells or higher. At this dose range or higher no patients had disease progression at up to 54 weeks after infusion. Of the four patients evaluable for minimal residual disease (MRD), all were MRD negative.
As of the data cut-off there were no dose-limiting toxicities and no treatment-related grade 3 or higher neurotoxicities. CRS was primarily grade 1 or 2 and was reported in 15 of 21 patients. Two patients had grade 3 CRS that resolved in 24 hours, and four patients received tocilizumab (one with steroids) for CRS management.
There was one on-study death, from cardiopulmonary arrest more than 4 months after bb2121 infusion in a patient with an extensive cardiac history. The death was judged to be unrelated to CAR T-cell therapy.
Dr. Kochenderfer will present data and analysis on an additional 5 months of follow-up.
“This abstract demonstrates that again, in patients, this time with myeloma with literally no other option, you can achieve very impressive response,” Dr. Anderson said.
The ZUMA-1 study is funded by Kite Pharma. Dr. Neelapu reported research funding from BMS, Poseida, Merck, Kite Pharma, and Cellectis. He reported consultancy and membership on the board of directors or advisory committees for Merck, Kite Pharma, and Celgene.
The JULIET study is funded by Novartis. Dr. Schuster reported consultancy and research funding from Novartis Pharmaceuticals, Celgene, Genentech, Bristol-Myers Squibb, Janssen R&D, and Gilead. He reported consultancy with Seattle Genetics and Nordic Nanovector, and research funding from Merck.
The multicenter study of bb2121 is sponsored by bluebird bio and Celgene. Dr. Kochenderfer reported research funding from bluebird bio and Kite Pharma. He reported having multiple patents in the CAR field.
Abstract 578 Long-Term Follow-up ZUMA-1: A Pivotal Trial of Axicabtagene Ciloleucel (Axi-Cel; KTE-C19) in Patients with Refractory Aggressive Non-Hodgkin Lymphoma (NHL) will be presented in session 626, Monday, Dec. 11, 2017, at 7:15 a.m.
Abstract 577 Primary Analysis of Juliet: A Global, Pivotal, Phase 2 Trial of CTL019 in Adult Patients with Relapsed or Refractory Diffuse Large B-Cell Lymphoma will be presented in session 626, Monday, Dec. 11, 2017 at 7:00 a.m.
Abstract 740 Durable Clinical Responses in Heavily Pretreated Patients with Relapsed/Refractory Multiple Myeloma: Updated Results from a Multicenter Study of bb2121 Anti-BCMA CAR T Cell Therapy will be presented in session 653, Monday, Dec. 11, 2017 at 3:00 p.m.
FROM ASH 2017
Late-breaking abstracts highlight treatment advances in CLL, myeloma, and more
.
In a preplanned interim analysis of data from 389 patients in the randomized phase III Murano trial, venetoclax and rituximab therapy proved “superior to the standard of care and well tolerated, and a major advance in the management of [relapsed/refractory] CLL,” ASH President Kenneth C. Anderson, MD said during a premeeting preview session for the media.
In Murano, venetoclax plus rituximab bettered bendamustine plus rituximab in progression-free survival, overall survival, overall and complete response rates, and number of patients achieving minimal residual disease (MRD) negativity, said Dr. Anderson, who is also director of the Lebow Institute for Myeloma Therapeutics and Jerome Lipper Myeloma Center at Dana-Farber Cancer Institute, Boston.
The results were consistent in all risk subsets, including patients who had high-risk disease by virtue of chromosome 17p deletion, according Dr. Anderson.
In another late-breaking randomized phase III study, known as ALCYONE, adding the CD38-targeting monoclonal antibody daratumumab to standard therapy with bortezomib, melphalan, and prednisone (VMP) resulted in a “doubling” of progression-free survival in patients who had newly diagnosed multiple myeloma and were ineligible for transplantation, he reported.
In the trial of more than 700 patients, daratumumab plus VMP as initial treatment for nontransplant patients was well tolerated and improved outcomes, including overall response rate and the percent of patients who achieved MRD negative status.
“As we saw in CLL, so it’s true in this abstract in myeloma: this is a very major advance,” Dr. Anderson said.
Also during the preview session, ASH Secretary Robert A. Brodsky, MD, discussed the randomized, phase III HERCULES study results, which showed that patients with acquired thrombotic thrombocytopenic purpura (TTP) may benefit when caplacizumab is added to standard therapy. Caplacizumab targets the A1 domain of von Willebrand factor, which inhibits interaction between ultra-large von Willebrand factor and platelets.
In the trial, 145 patients were randomized to receive either plasma exchange alone or plasma exchange and caplacizumab.
Preliminary results suggest “this was a very positive trial” with a primary endpoint of time to platelet response that “greatly favored the caplacizumab arm,” said Dr. Brodsky, professor of medicine and oncology and director of the division of hematology at Johns Hopkins University, Baltimore. “Even the secondary composite endpoint of death, recurrence, and/or major thromboembolic events was much improved with caplacizumab, so this is a very positive trial and potentially a game-changing drug for the management of TTP, which can be very challenging.”
Dr. Brodsky also discussed the Hokusai VTE-Cancer Study, a randomized, open-label, blinded outcome assessment trial that showed the oral factor Xa inhibitor edoxaban was noninferior to subcutaneous dalteparin for the prevention of cancer-associated venous thromboembolism.
With more than 1,000 patients enrolled in 114 centers, the Hokusai VTE-Cancer Study had a primary outcome of the composite of the first recurrent VTE or major bleeding event during follow-up. The primary outcome occurred in 12.8% of patients in the edoxaban group, compared with 13.5% of patients in the dalteparin group (P = .0056 for noninferiority), according to the preliminary published results.
The key question addressed by the trial is whether a newer oral anticoagulant, edoxaban, can substitute for the older, subcutaneously administered low-molecular-weight heparin, dalteparin. The results “confirmed that a newer oral anticoagulant is at least as good and as safe as the low molecular weight heparin,” allowing patients the convenience of an oral therapy, Dr. Brodsky noted.
This year’s late-breaking abstracts at ASH are:
LBA-1 Results of the Randomized, Double-Blind, Placebo-Controlled, Phase III Hercules Study of Caplacizumab in Patients with Acquired Thrombotic Thrombocytopenic Purpura.
LBA-2 Venetoclax Plus Rituximab Is Superior to Bendamustine Plus Rituximab in Patients with Relapsed/ Refractory Chronic Lymphocytic Leukemia - Results from Pre-Planned Interim Analysis of the Randomized Phase III Murano Study.
LBA-3 Mutations in SRP54 Gene Cause Severe Primary Neutropenia As Well As Shwachman-Diamond-like Syndrome.
LBA-4 Phase III Randomized Study of Daratumumab Plus Bortezomib, Melphalan, and Prednisone (D-VMP) Versus Bortezomib, Melphalan, and Prednisone (VMP) in Newly Diagnosed Multiple Myeloma (NDMM) Patients (Pts) Ineligible for Transplant (ALCYONE).
LBA-5 Prospective Molecular MRD Detection By NGS: A Powerful Independent Predictor for Relapse and Survival in Adults with Newly Diagnosed AML.
LBA-6 A Randomized, Open-Label, Blinded Outcome Assessment Trial Evaluating the Efficacy and Safety of LMWH/Edoxaban Versus Dalteparin for Venous Thromboembolism Associated with Cancer: Hokusai VTE-Cancer Study
.
In a preplanned interim analysis of data from 389 patients in the randomized phase III Murano trial, venetoclax and rituximab therapy proved “superior to the standard of care and well tolerated, and a major advance in the management of [relapsed/refractory] CLL,” ASH President Kenneth C. Anderson, MD said during a premeeting preview session for the media.
In Murano, venetoclax plus rituximab bettered bendamustine plus rituximab in progression-free survival, overall survival, overall and complete response rates, and number of patients achieving minimal residual disease (MRD) negativity, said Dr. Anderson, who is also director of the Lebow Institute for Myeloma Therapeutics and Jerome Lipper Myeloma Center at Dana-Farber Cancer Institute, Boston.
The results were consistent in all risk subsets, including patients who had high-risk disease by virtue of chromosome 17p deletion, according Dr. Anderson.
In another late-breaking randomized phase III study, known as ALCYONE, adding the CD38-targeting monoclonal antibody daratumumab to standard therapy with bortezomib, melphalan, and prednisone (VMP) resulted in a “doubling” of progression-free survival in patients who had newly diagnosed multiple myeloma and were ineligible for transplantation, he reported.
In the trial of more than 700 patients, daratumumab plus VMP as initial treatment for nontransplant patients was well tolerated and improved outcomes, including overall response rate and the percent of patients who achieved MRD negative status.
“As we saw in CLL, so it’s true in this abstract in myeloma: this is a very major advance,” Dr. Anderson said.
Also during the preview session, ASH Secretary Robert A. Brodsky, MD, discussed the randomized, phase III HERCULES study results, which showed that patients with acquired thrombotic thrombocytopenic purpura (TTP) may benefit when caplacizumab is added to standard therapy. Caplacizumab targets the A1 domain of von Willebrand factor, which inhibits interaction between ultra-large von Willebrand factor and platelets.
In the trial, 145 patients were randomized to receive either plasma exchange alone or plasma exchange and caplacizumab.
Preliminary results suggest “this was a very positive trial” with a primary endpoint of time to platelet response that “greatly favored the caplacizumab arm,” said Dr. Brodsky, professor of medicine and oncology and director of the division of hematology at Johns Hopkins University, Baltimore. “Even the secondary composite endpoint of death, recurrence, and/or major thromboembolic events was much improved with caplacizumab, so this is a very positive trial and potentially a game-changing drug for the management of TTP, which can be very challenging.”
Dr. Brodsky also discussed the Hokusai VTE-Cancer Study, a randomized, open-label, blinded outcome assessment trial that showed the oral factor Xa inhibitor edoxaban was noninferior to subcutaneous dalteparin for the prevention of cancer-associated venous thromboembolism.
With more than 1,000 patients enrolled in 114 centers, the Hokusai VTE-Cancer Study had a primary outcome of the composite of the first recurrent VTE or major bleeding event during follow-up. The primary outcome occurred in 12.8% of patients in the edoxaban group, compared with 13.5% of patients in the dalteparin group (P = .0056 for noninferiority), according to the preliminary published results.
The key question addressed by the trial is whether a newer oral anticoagulant, edoxaban, can substitute for the older, subcutaneously administered low-molecular-weight heparin, dalteparin. The results “confirmed that a newer oral anticoagulant is at least as good and as safe as the low molecular weight heparin,” allowing patients the convenience of an oral therapy, Dr. Brodsky noted.
This year’s late-breaking abstracts at ASH are:
LBA-1 Results of the Randomized, Double-Blind, Placebo-Controlled, Phase III Hercules Study of Caplacizumab in Patients with Acquired Thrombotic Thrombocytopenic Purpura.
LBA-2 Venetoclax Plus Rituximab Is Superior to Bendamustine Plus Rituximab in Patients with Relapsed/ Refractory Chronic Lymphocytic Leukemia - Results from Pre-Planned Interim Analysis of the Randomized Phase III Murano Study.
LBA-3 Mutations in SRP54 Gene Cause Severe Primary Neutropenia As Well As Shwachman-Diamond-like Syndrome.
LBA-4 Phase III Randomized Study of Daratumumab Plus Bortezomib, Melphalan, and Prednisone (D-VMP) Versus Bortezomib, Melphalan, and Prednisone (VMP) in Newly Diagnosed Multiple Myeloma (NDMM) Patients (Pts) Ineligible for Transplant (ALCYONE).
LBA-5 Prospective Molecular MRD Detection By NGS: A Powerful Independent Predictor for Relapse and Survival in Adults with Newly Diagnosed AML.
LBA-6 A Randomized, Open-Label, Blinded Outcome Assessment Trial Evaluating the Efficacy and Safety of LMWH/Edoxaban Versus Dalteparin for Venous Thromboembolism Associated with Cancer: Hokusai VTE-Cancer Study
.
In a preplanned interim analysis of data from 389 patients in the randomized phase III Murano trial, venetoclax and rituximab therapy proved “superior to the standard of care and well tolerated, and a major advance in the management of [relapsed/refractory] CLL,” ASH President Kenneth C. Anderson, MD said during a premeeting preview session for the media.
In Murano, venetoclax plus rituximab bettered bendamustine plus rituximab in progression-free survival, overall survival, overall and complete response rates, and number of patients achieving minimal residual disease (MRD) negativity, said Dr. Anderson, who is also director of the Lebow Institute for Myeloma Therapeutics and Jerome Lipper Myeloma Center at Dana-Farber Cancer Institute, Boston.
The results were consistent in all risk subsets, including patients who had high-risk disease by virtue of chromosome 17p deletion, according Dr. Anderson.
In another late-breaking randomized phase III study, known as ALCYONE, adding the CD38-targeting monoclonal antibody daratumumab to standard therapy with bortezomib, melphalan, and prednisone (VMP) resulted in a “doubling” of progression-free survival in patients who had newly diagnosed multiple myeloma and were ineligible for transplantation, he reported.
In the trial of more than 700 patients, daratumumab plus VMP as initial treatment for nontransplant patients was well tolerated and improved outcomes, including overall response rate and the percent of patients who achieved MRD negative status.
“As we saw in CLL, so it’s true in this abstract in myeloma: this is a very major advance,” Dr. Anderson said.
Also during the preview session, ASH Secretary Robert A. Brodsky, MD, discussed the randomized, phase III HERCULES study results, which showed that patients with acquired thrombotic thrombocytopenic purpura (TTP) may benefit when caplacizumab is added to standard therapy. Caplacizumab targets the A1 domain of von Willebrand factor, which inhibits interaction between ultra-large von Willebrand factor and platelets.
In the trial, 145 patients were randomized to receive either plasma exchange alone or plasma exchange and caplacizumab.
Preliminary results suggest “this was a very positive trial” with a primary endpoint of time to platelet response that “greatly favored the caplacizumab arm,” said Dr. Brodsky, professor of medicine and oncology and director of the division of hematology at Johns Hopkins University, Baltimore. “Even the secondary composite endpoint of death, recurrence, and/or major thromboembolic events was much improved with caplacizumab, so this is a very positive trial and potentially a game-changing drug for the management of TTP, which can be very challenging.”
Dr. Brodsky also discussed the Hokusai VTE-Cancer Study, a randomized, open-label, blinded outcome assessment trial that showed the oral factor Xa inhibitor edoxaban was noninferior to subcutaneous dalteparin for the prevention of cancer-associated venous thromboembolism.
With more than 1,000 patients enrolled in 114 centers, the Hokusai VTE-Cancer Study had a primary outcome of the composite of the first recurrent VTE or major bleeding event during follow-up. The primary outcome occurred in 12.8% of patients in the edoxaban group, compared with 13.5% of patients in the dalteparin group (P = .0056 for noninferiority), according to the preliminary published results.
The key question addressed by the trial is whether a newer oral anticoagulant, edoxaban, can substitute for the older, subcutaneously administered low-molecular-weight heparin, dalteparin. The results “confirmed that a newer oral anticoagulant is at least as good and as safe as the low molecular weight heparin,” allowing patients the convenience of an oral therapy, Dr. Brodsky noted.
This year’s late-breaking abstracts at ASH are:
LBA-1 Results of the Randomized, Double-Blind, Placebo-Controlled, Phase III Hercules Study of Caplacizumab in Patients with Acquired Thrombotic Thrombocytopenic Purpura.
LBA-2 Venetoclax Plus Rituximab Is Superior to Bendamustine Plus Rituximab in Patients with Relapsed/ Refractory Chronic Lymphocytic Leukemia - Results from Pre-Planned Interim Analysis of the Randomized Phase III Murano Study.
LBA-3 Mutations in SRP54 Gene Cause Severe Primary Neutropenia As Well As Shwachman-Diamond-like Syndrome.
LBA-4 Phase III Randomized Study of Daratumumab Plus Bortezomib, Melphalan, and Prednisone (D-VMP) Versus Bortezomib, Melphalan, and Prednisone (VMP) in Newly Diagnosed Multiple Myeloma (NDMM) Patients (Pts) Ineligible for Transplant (ALCYONE).
LBA-5 Prospective Molecular MRD Detection By NGS: A Powerful Independent Predictor for Relapse and Survival in Adults with Newly Diagnosed AML.
LBA-6 A Randomized, Open-Label, Blinded Outcome Assessment Trial Evaluating the Efficacy and Safety of LMWH/Edoxaban Versus Dalteparin for Venous Thromboembolism Associated with Cancer: Hokusai VTE-Cancer Study
FROM ASH 2017
Method identifies effective treatments for leukemias, lymphomas
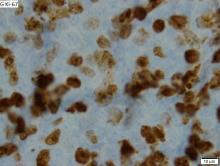
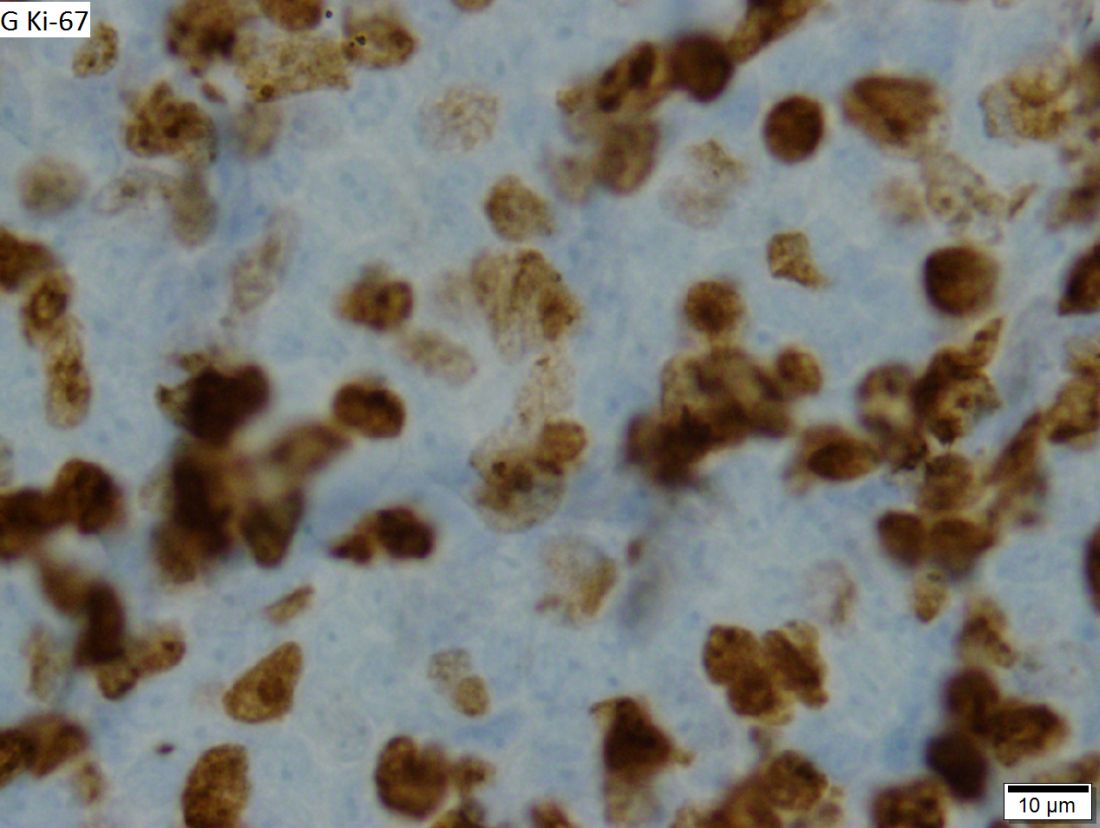
An ex vivo drug screening method can reveal optimal therapies for patients with hematologic malignancies, according to research published in The Lancet Haematology.
Researchers used a method called pharmacoscopy to measure single-cell responses to possible treatments in samples from patients with leukemias and lymphomas.
The team then used these results to guide treatment decisions and found that pharmacoscopy-guided treatment greatly improved response rates and progression-free survival (PFS).
“Having a robust, fast, and reliable predictive test at our disposal during the patient treatment process, especially at the time of relapse where a new intervention must be selected quickly, will change how medical doctors prioritize drugs to use for late-stage patients,” said study author Philipp Staber, MD, of Medical University of Vienna in Austria.
With pharmacoscopy, hundreds of drug options can be pre-tested ex vivo in small liquid biopsy samples collected from individual patients. The effects of each drug on the individual cells are quantified using high-throughput and high-content automated confocal microscopy.
In combination with specially developed analysis methods, machine learning, and other algorithms, pharmacoscopy allows quantification of never-before visualized phenotypes. The method was first described last April in Nature Chemical Biology.
Now, Dr Staber and his colleagues have reported, in The Lancet Haematology, an interim analysis of the first clinical trial testing pharmacoscopy-guided treatment.
There were 17 evaluable patients, all of whom had aggressive hematologic malignancies. This included diffuse large B-cell lymphoma (n=6), acute myeloid leukemia (n=3), B-cell acute lymphoblastic leukemia (n=2), precursor B-cell lymphoblastic lymphoma (n=1), peripheral T-cell lymphoma (n=1), primary mediastinal B-cell lymphoma (n=1), T-cell lymphoblastic lymphoma (n=1), follicular lymphoma (n=1), and T-cell prolymphocytic leukemia (n=1).
The researchers compared outcomes with pharmacoscopy-guided treatment to outcomes with the most recent regimen on which the patient had progressed.
The overall response rate was 88% with pharmacoscopy-guided treatment and 24% with the patients’ most recent previous treatment regimen (odds ratio=24.38; 95%, CI 3.99–125.4; P=0.0013).
None of the patients had progressive disease as their best overall response when they received pharmacoscopy-guided treatment. However, 7 patients had progressive disease in response to their most recent prior regimen.
At the time of analysis, 8 patients (47%) still had ongoing responses after pharmacoscopy-guided treatment.
In addition, pharmacoscopy-guided treatment significantly improved PFS. The median PFS was 22.6 weeks with pharmacoscopy-guided treatment and 5.7 weeks with the most recent prior regimen (hazard ratio=3.14; 95%, CI 1.37–7.22; P=0.0075).
“Evidence that the pharmacoscopy approach is helpful for clinical evaluation of therapy is wonderful,” said study author Giulio Superti-Furga, PhD, of CeMM Research Center for Molecular Medicine in Vienna, Austria.
“Single-cell functional analysis of primary material gives unprecedented resolution and precision that we are sure to further develop in the future to address yet more diseases.” ![]()
An ex vivo drug screening method can reveal optimal therapies for patients with hematologic malignancies, according to research published in The Lancet Haematology.
Researchers used a method called pharmacoscopy to measure single-cell responses to possible treatments in samples from patients with leukemias and lymphomas.
The team then used these results to guide treatment decisions and found that pharmacoscopy-guided treatment greatly improved response rates and progression-free survival (PFS).
“Having a robust, fast, and reliable predictive test at our disposal during the patient treatment process, especially at the time of relapse where a new intervention must be selected quickly, will change how medical doctors prioritize drugs to use for late-stage patients,” said study author Philipp Staber, MD, of Medical University of Vienna in Austria.
With pharmacoscopy, hundreds of drug options can be pre-tested ex vivo in small liquid biopsy samples collected from individual patients. The effects of each drug on the individual cells are quantified using high-throughput and high-content automated confocal microscopy.
In combination with specially developed analysis methods, machine learning, and other algorithms, pharmacoscopy allows quantification of never-before visualized phenotypes. The method was first described last April in Nature Chemical Biology.
Now, Dr Staber and his colleagues have reported, in The Lancet Haematology, an interim analysis of the first clinical trial testing pharmacoscopy-guided treatment.
There were 17 evaluable patients, all of whom had aggressive hematologic malignancies. This included diffuse large B-cell lymphoma (n=6), acute myeloid leukemia (n=3), B-cell acute lymphoblastic leukemia (n=2), precursor B-cell lymphoblastic lymphoma (n=1), peripheral T-cell lymphoma (n=1), primary mediastinal B-cell lymphoma (n=1), T-cell lymphoblastic lymphoma (n=1), follicular lymphoma (n=1), and T-cell prolymphocytic leukemia (n=1).
The researchers compared outcomes with pharmacoscopy-guided treatment to outcomes with the most recent regimen on which the patient had progressed.
The overall response rate was 88% with pharmacoscopy-guided treatment and 24% with the patients’ most recent previous treatment regimen (odds ratio=24.38; 95%, CI 3.99–125.4; P=0.0013).
None of the patients had progressive disease as their best overall response when they received pharmacoscopy-guided treatment. However, 7 patients had progressive disease in response to their most recent prior regimen.
At the time of analysis, 8 patients (47%) still had ongoing responses after pharmacoscopy-guided treatment.
In addition, pharmacoscopy-guided treatment significantly improved PFS. The median PFS was 22.6 weeks with pharmacoscopy-guided treatment and 5.7 weeks with the most recent prior regimen (hazard ratio=3.14; 95%, CI 1.37–7.22; P=0.0075).
“Evidence that the pharmacoscopy approach is helpful for clinical evaluation of therapy is wonderful,” said study author Giulio Superti-Furga, PhD, of CeMM Research Center for Molecular Medicine in Vienna, Austria.
“Single-cell functional analysis of primary material gives unprecedented resolution and precision that we are sure to further develop in the future to address yet more diseases.” ![]()
An ex vivo drug screening method can reveal optimal therapies for patients with hematologic malignancies, according to research published in The Lancet Haematology.
Researchers used a method called pharmacoscopy to measure single-cell responses to possible treatments in samples from patients with leukemias and lymphomas.
The team then used these results to guide treatment decisions and found that pharmacoscopy-guided treatment greatly improved response rates and progression-free survival (PFS).
“Having a robust, fast, and reliable predictive test at our disposal during the patient treatment process, especially at the time of relapse where a new intervention must be selected quickly, will change how medical doctors prioritize drugs to use for late-stage patients,” said study author Philipp Staber, MD, of Medical University of Vienna in Austria.
With pharmacoscopy, hundreds of drug options can be pre-tested ex vivo in small liquid biopsy samples collected from individual patients. The effects of each drug on the individual cells are quantified using high-throughput and high-content automated confocal microscopy.
In combination with specially developed analysis methods, machine learning, and other algorithms, pharmacoscopy allows quantification of never-before visualized phenotypes. The method was first described last April in Nature Chemical Biology.
Now, Dr Staber and his colleagues have reported, in The Lancet Haematology, an interim analysis of the first clinical trial testing pharmacoscopy-guided treatment.
There were 17 evaluable patients, all of whom had aggressive hematologic malignancies. This included diffuse large B-cell lymphoma (n=6), acute myeloid leukemia (n=3), B-cell acute lymphoblastic leukemia (n=2), precursor B-cell lymphoblastic lymphoma (n=1), peripheral T-cell lymphoma (n=1), primary mediastinal B-cell lymphoma (n=1), T-cell lymphoblastic lymphoma (n=1), follicular lymphoma (n=1), and T-cell prolymphocytic leukemia (n=1).
The researchers compared outcomes with pharmacoscopy-guided treatment to outcomes with the most recent regimen on which the patient had progressed.
The overall response rate was 88% with pharmacoscopy-guided treatment and 24% with the patients’ most recent previous treatment regimen (odds ratio=24.38; 95%, CI 3.99–125.4; P=0.0013).
None of the patients had progressive disease as their best overall response when they received pharmacoscopy-guided treatment. However, 7 patients had progressive disease in response to their most recent prior regimen.
At the time of analysis, 8 patients (47%) still had ongoing responses after pharmacoscopy-guided treatment.
In addition, pharmacoscopy-guided treatment significantly improved PFS. The median PFS was 22.6 weeks with pharmacoscopy-guided treatment and 5.7 weeks with the most recent prior regimen (hazard ratio=3.14; 95%, CI 1.37–7.22; P=0.0075).
“Evidence that the pharmacoscopy approach is helpful for clinical evaluation of therapy is wonderful,” said study author Giulio Superti-Furga, PhD, of CeMM Research Center for Molecular Medicine in Vienna, Austria.
“Single-cell functional analysis of primary material gives unprecedented resolution and precision that we are sure to further develop in the future to address yet more diseases.” ![]()
PTSD can persist in cancer survivors
Cancer patients may experience lasting post-traumatic stress disorder (PTSD), according to a study published in the journal Cancer.
Approximately one-fifth of patients involved in the study experienced PTSD several months after their cancer diagnosis, and roughly a third of these patients continued to live with PTSD 4 years later.
Researchers say these findings highlight the need for early identification, careful monitoring, and treatment of PTSD in cancer survivors.
Caryn Mei Hsien Chan, PhD, of the National University of Malaysia in Kuala Lumpur, and her colleagues conducted this research.
The study included 469 adults with various cancers who were within 1 month of cancer diagnosis at enrollment.
Patients who had significant psychological distress (defined as a Hospital Anxiety and Depression Scale total cutoff score of 16 or higher) underwent
testing for PTSD at 6 months of follow-up. All patients were tested for PTSD at 4 years of follow-up (regardless of their Hospital Anxiety and Depression Scale score).
The incidence of PTSD was 21.7% at 6 months and 6.1% at 4 years. Although overall rates of PTSD decreased with time, roughly one-third of patients initially diagnosed with PTSD were found to have persistent or worsening symptoms 4 years later.
“Many cancer patients believe they need to adopt a ‘warrior mentality’ and remain positive and optimistic from diagnosis through treatment to stand a better chance of beating their cancer,” Dr Chan said.
“To these patients, seeking help for the emotional issues they face is akin to admitting weakness. There needs to be greater awareness that there is nothing wrong with getting help to manage the emotional upheaval—particularly depression, anxiety, and PTSD—post-cancer.”
Dr Chan also stressed that many patients live in fear that their cancer may come back, and they may think the cancer has returned with every lump or bump, pain or ache, fatigue or fever.
In addition, cancer survivors might skip visits to their oncologists or other physicians to avoid triggering memories of their past cancer experience. This can lead to delays in seeking help for new symptoms or even refusal of treatment for unrelated conditions.
“We need psychological evaluation and support services for patients with cancer at an initial stage and at continued follows-up because psychological well-being and mental health—and by extension, quality of life—are just as important as physical health,” Dr Chan noted. ![]()
Cancer patients may experience lasting post-traumatic stress disorder (PTSD), according to a study published in the journal Cancer.
Approximately one-fifth of patients involved in the study experienced PTSD several months after their cancer diagnosis, and roughly a third of these patients continued to live with PTSD 4 years later.
Researchers say these findings highlight the need for early identification, careful monitoring, and treatment of PTSD in cancer survivors.
Caryn Mei Hsien Chan, PhD, of the National University of Malaysia in Kuala Lumpur, and her colleagues conducted this research.
The study included 469 adults with various cancers who were within 1 month of cancer diagnosis at enrollment.
Patients who had significant psychological distress (defined as a Hospital Anxiety and Depression Scale total cutoff score of 16 or higher) underwent
testing for PTSD at 6 months of follow-up. All patients were tested for PTSD at 4 years of follow-up (regardless of their Hospital Anxiety and Depression Scale score).
The incidence of PTSD was 21.7% at 6 months and 6.1% at 4 years. Although overall rates of PTSD decreased with time, roughly one-third of patients initially diagnosed with PTSD were found to have persistent or worsening symptoms 4 years later.
“Many cancer patients believe they need to adopt a ‘warrior mentality’ and remain positive and optimistic from diagnosis through treatment to stand a better chance of beating their cancer,” Dr Chan said.
“To these patients, seeking help for the emotional issues they face is akin to admitting weakness. There needs to be greater awareness that there is nothing wrong with getting help to manage the emotional upheaval—particularly depression, anxiety, and PTSD—post-cancer.”
Dr Chan also stressed that many patients live in fear that their cancer may come back, and they may think the cancer has returned with every lump or bump, pain or ache, fatigue or fever.
In addition, cancer survivors might skip visits to their oncologists or other physicians to avoid triggering memories of their past cancer experience. This can lead to delays in seeking help for new symptoms or even refusal of treatment for unrelated conditions.
“We need psychological evaluation and support services for patients with cancer at an initial stage and at continued follows-up because psychological well-being and mental health—and by extension, quality of life—are just as important as physical health,” Dr Chan noted. ![]()
Cancer patients may experience lasting post-traumatic stress disorder (PTSD), according to a study published in the journal Cancer.
Approximately one-fifth of patients involved in the study experienced PTSD several months after their cancer diagnosis, and roughly a third of these patients continued to live with PTSD 4 years later.
Researchers say these findings highlight the need for early identification, careful monitoring, and treatment of PTSD in cancer survivors.
Caryn Mei Hsien Chan, PhD, of the National University of Malaysia in Kuala Lumpur, and her colleagues conducted this research.
The study included 469 adults with various cancers who were within 1 month of cancer diagnosis at enrollment.
Patients who had significant psychological distress (defined as a Hospital Anxiety and Depression Scale total cutoff score of 16 or higher) underwent
testing for PTSD at 6 months of follow-up. All patients were tested for PTSD at 4 years of follow-up (regardless of their Hospital Anxiety and Depression Scale score).
The incidence of PTSD was 21.7% at 6 months and 6.1% at 4 years. Although overall rates of PTSD decreased with time, roughly one-third of patients initially diagnosed with PTSD were found to have persistent or worsening symptoms 4 years later.
“Many cancer patients believe they need to adopt a ‘warrior mentality’ and remain positive and optimistic from diagnosis through treatment to stand a better chance of beating their cancer,” Dr Chan said.
“To these patients, seeking help for the emotional issues they face is akin to admitting weakness. There needs to be greater awareness that there is nothing wrong with getting help to manage the emotional upheaval—particularly depression, anxiety, and PTSD—post-cancer.”
Dr Chan also stressed that many patients live in fear that their cancer may come back, and they may think the cancer has returned with every lump or bump, pain or ache, fatigue or fever.
In addition, cancer survivors might skip visits to their oncologists or other physicians to avoid triggering memories of their past cancer experience. This can lead to delays in seeking help for new symptoms or even refusal of treatment for unrelated conditions.
“We need psychological evaluation and support services for patients with cancer at an initial stage and at continued follows-up because psychological well-being and mental health—and by extension, quality of life—are just as important as physical health,” Dr Chan noted. ![]()
CD22 CAR activity in B-ALL highlights promise of multispecific CARs
NATIONAL HARBOR, MD. – A new CD22-targeted chimeric antigen receptor (CAR) demonstrated clinical activity in a phase 1 study of adults and children with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL), including several who were previously treated with CD19-directed immunotherapy.
In 21 children and adults with B-ALL who were treated with the CD22 CAR, dose-dependent antileukemic activity was observed; complete remission occurred in 11 of 15 (73%) who received bioactive doses (at least 1 x 106 CD22 CAR T-cells per kg of body weight), including 5 of 5 patients with CD19dim or CD19neg B-ALL, Terry J. Fry, MD, of the National Institutes of Health, Bethesda, Md., and colleagues reported in Nature Medicine (2017 Nov 20. doi: 10.1038.nm.4441).
This study is the first to establish the clinical activity of a CD22 CAR in B-ALL. The investigators developed the CAR in an effort to counter the resistance sometimes seen in patients who receive CD19 CAR T-cell therapy. CD22 is also expressed in most B-ALL cases – and usually is retained following CD19 loss, they explained.
The findings, when considered in light of efficacy demonstrated in leukemia that is resistant to anti-CD19 immunotherapy, highlights the potential for – and importance of – developing multispecific CARs, Crystal L. Mackall, MD, the senior author of the study, said during an update on CAR T-cell research at the annual meeting of the Society for Immunotherapy of Cancer.
“All in all, once we got to the dose that was appropriate ... this CAR had really impressive activity,” said Dr. Mackall, director of the Parker Institute for Cancer Immunotherapy at Stanford (Calif.) University. “In some patients, this was all the patient needed for a prolonged disease-free interval.”
Three patients had ongoing responses, at 21 months, 9 months, and 6 months, she said. There was a high rate of relapse among the study participants, but all patients had previously received at least one bone marrow transplant, and 17 had received CD19-based immunotherapy.
“But nonetheless, the interrogation of these relapses was really essential to understand more about the Achilles heels of these CAR T-cells,” she said. “What we saw is that it was all about the antigen.”
Unlike CD19, which tends to disappear after relapse, CD22-expressing tumors that relapse tend to come back with “simply lower expression of CD22,” she said. The CD22 CAR was unable to control the CD22lo leukemias. This is not unique to the CD22 CAR, she said.
“Every CAR we’ve looked at so far has this exquisite dependence on antigen density for functionality,” she explained, noting that heterogeneity in antigen expression will pose major challenges for the development of therapies, and “maybe has been one of the main reasons we haven’t yet seen the effectiveness of CAR T-cells in solid tumors that we have for hematological malignancies where we’ve typically had targets that are expressed homogenously and at high levels.”
“So we believe very strongly that multispecific CARS are going to be essential for progress, especially as we move into solid tumors,” she added.
Early attempts at developing multispecific CARS suggest that coadministration is not ideal, but two other approaches – coexpression using two vectors or a bicistronic vector, or by creation of a bivalent-bispecific CAR (also known as a tandem CAR) – are both still on the table, she said.
Two clinical first-in-human trials evaluating a CD19/22-bispecific CAR (one in children and one in adults) for relapsed/refractory B-cell malignancies are underway at Stanford.
“We predict that this is going to be the beginning of a wave of bispecific, trispecific, and maybe even quad CARs,” she said. “There’s a lot of work to do, but this is an area that’s going to be very active in the coming years.”
Dr. Mackall has received consulting fees from Adaptimmune, GSK, Roche, Unum Therapeutics, and Vore Pharmaceuticals; has conducted research for Bluebird Bio; and has ownership interest from Juno Therapeutics.
NATIONAL HARBOR, MD. – A new CD22-targeted chimeric antigen receptor (CAR) demonstrated clinical activity in a phase 1 study of adults and children with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL), including several who were previously treated with CD19-directed immunotherapy.
In 21 children and adults with B-ALL who were treated with the CD22 CAR, dose-dependent antileukemic activity was observed; complete remission occurred in 11 of 15 (73%) who received bioactive doses (at least 1 x 106 CD22 CAR T-cells per kg of body weight), including 5 of 5 patients with CD19dim or CD19neg B-ALL, Terry J. Fry, MD, of the National Institutes of Health, Bethesda, Md., and colleagues reported in Nature Medicine (2017 Nov 20. doi: 10.1038.nm.4441).
This study is the first to establish the clinical activity of a CD22 CAR in B-ALL. The investigators developed the CAR in an effort to counter the resistance sometimes seen in patients who receive CD19 CAR T-cell therapy. CD22 is also expressed in most B-ALL cases – and usually is retained following CD19 loss, they explained.
The findings, when considered in light of efficacy demonstrated in leukemia that is resistant to anti-CD19 immunotherapy, highlights the potential for – and importance of – developing multispecific CARs, Crystal L. Mackall, MD, the senior author of the study, said during an update on CAR T-cell research at the annual meeting of the Society for Immunotherapy of Cancer.
“All in all, once we got to the dose that was appropriate ... this CAR had really impressive activity,” said Dr. Mackall, director of the Parker Institute for Cancer Immunotherapy at Stanford (Calif.) University. “In some patients, this was all the patient needed for a prolonged disease-free interval.”
Three patients had ongoing responses, at 21 months, 9 months, and 6 months, she said. There was a high rate of relapse among the study participants, but all patients had previously received at least one bone marrow transplant, and 17 had received CD19-based immunotherapy.
“But nonetheless, the interrogation of these relapses was really essential to understand more about the Achilles heels of these CAR T-cells,” she said. “What we saw is that it was all about the antigen.”
Unlike CD19, which tends to disappear after relapse, CD22-expressing tumors that relapse tend to come back with “simply lower expression of CD22,” she said. The CD22 CAR was unable to control the CD22lo leukemias. This is not unique to the CD22 CAR, she said.
“Every CAR we’ve looked at so far has this exquisite dependence on antigen density for functionality,” she explained, noting that heterogeneity in antigen expression will pose major challenges for the development of therapies, and “maybe has been one of the main reasons we haven’t yet seen the effectiveness of CAR T-cells in solid tumors that we have for hematological malignancies where we’ve typically had targets that are expressed homogenously and at high levels.”
“So we believe very strongly that multispecific CARS are going to be essential for progress, especially as we move into solid tumors,” she added.
Early attempts at developing multispecific CARS suggest that coadministration is not ideal, but two other approaches – coexpression using two vectors or a bicistronic vector, or by creation of a bivalent-bispecific CAR (also known as a tandem CAR) – are both still on the table, she said.
Two clinical first-in-human trials evaluating a CD19/22-bispecific CAR (one in children and one in adults) for relapsed/refractory B-cell malignancies are underway at Stanford.
“We predict that this is going to be the beginning of a wave of bispecific, trispecific, and maybe even quad CARs,” she said. “There’s a lot of work to do, but this is an area that’s going to be very active in the coming years.”
Dr. Mackall has received consulting fees from Adaptimmune, GSK, Roche, Unum Therapeutics, and Vore Pharmaceuticals; has conducted research for Bluebird Bio; and has ownership interest from Juno Therapeutics.
NATIONAL HARBOR, MD. – A new CD22-targeted chimeric antigen receptor (CAR) demonstrated clinical activity in a phase 1 study of adults and children with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL), including several who were previously treated with CD19-directed immunotherapy.
In 21 children and adults with B-ALL who were treated with the CD22 CAR, dose-dependent antileukemic activity was observed; complete remission occurred in 11 of 15 (73%) who received bioactive doses (at least 1 x 106 CD22 CAR T-cells per kg of body weight), including 5 of 5 patients with CD19dim or CD19neg B-ALL, Terry J. Fry, MD, of the National Institutes of Health, Bethesda, Md., and colleagues reported in Nature Medicine (2017 Nov 20. doi: 10.1038.nm.4441).
This study is the first to establish the clinical activity of a CD22 CAR in B-ALL. The investigators developed the CAR in an effort to counter the resistance sometimes seen in patients who receive CD19 CAR T-cell therapy. CD22 is also expressed in most B-ALL cases – and usually is retained following CD19 loss, they explained.
The findings, when considered in light of efficacy demonstrated in leukemia that is resistant to anti-CD19 immunotherapy, highlights the potential for – and importance of – developing multispecific CARs, Crystal L. Mackall, MD, the senior author of the study, said during an update on CAR T-cell research at the annual meeting of the Society for Immunotherapy of Cancer.
“All in all, once we got to the dose that was appropriate ... this CAR had really impressive activity,” said Dr. Mackall, director of the Parker Institute for Cancer Immunotherapy at Stanford (Calif.) University. “In some patients, this was all the patient needed for a prolonged disease-free interval.”
Three patients had ongoing responses, at 21 months, 9 months, and 6 months, she said. There was a high rate of relapse among the study participants, but all patients had previously received at least one bone marrow transplant, and 17 had received CD19-based immunotherapy.
“But nonetheless, the interrogation of these relapses was really essential to understand more about the Achilles heels of these CAR T-cells,” she said. “What we saw is that it was all about the antigen.”
Unlike CD19, which tends to disappear after relapse, CD22-expressing tumors that relapse tend to come back with “simply lower expression of CD22,” she said. The CD22 CAR was unable to control the CD22lo leukemias. This is not unique to the CD22 CAR, she said.
“Every CAR we’ve looked at so far has this exquisite dependence on antigen density for functionality,” she explained, noting that heterogeneity in antigen expression will pose major challenges for the development of therapies, and “maybe has been one of the main reasons we haven’t yet seen the effectiveness of CAR T-cells in solid tumors that we have for hematological malignancies where we’ve typically had targets that are expressed homogenously and at high levels.”
“So we believe very strongly that multispecific CARS are going to be essential for progress, especially as we move into solid tumors,” she added.
Early attempts at developing multispecific CARS suggest that coadministration is not ideal, but two other approaches – coexpression using two vectors or a bicistronic vector, or by creation of a bivalent-bispecific CAR (also known as a tandem CAR) – are both still on the table, she said.
Two clinical first-in-human trials evaluating a CD19/22-bispecific CAR (one in children and one in adults) for relapsed/refractory B-cell malignancies are underway at Stanford.
“We predict that this is going to be the beginning of a wave of bispecific, trispecific, and maybe even quad CARs,” she said. “There’s a lot of work to do, but this is an area that’s going to be very active in the coming years.”
Dr. Mackall has received consulting fees from Adaptimmune, GSK, Roche, Unum Therapeutics, and Vore Pharmaceuticals; has conducted research for Bluebird Bio; and has ownership interest from Juno Therapeutics.
AT SITC 2017
Key clinical point:
Major finding: Complete remission occurred in 73% of patients who received bioactive doses of the CD22 CAR.
Data source: A phase 1 study of 21 patients.
Disclosures: Dr. Mackall has received consulting fees from Adaptimmune, GSK, Roche, Unum Therapeutics, and Vore Pharmaceuticals; has conducted research for Bluebird Bio; and has ownership interest from Juno Therapeutics.
Cyclophosphamide after transplant reduced GVHD in myeloma patients
Post-transplantation cyclophosphamide may help decrease the rate of nonrelapse mortality and graft-versus-host disease (GVHD) in patients who have undergone allogeneic blood or marrow transplantation, according to new findings published in Biology of Blood and Marrow Transplantation.
Even though it is a potentially curative option for patients with poor-risk hematologic malignancies, allogeneic blood or marrow transplantation (alloBMT) has not been commonly used in multiple myeloma because of its association with high nonrelapse mortality and high relapse rates. However, the use of post-transplantation cyclophosphamide (PTCy) in this study led to low rates of both acute and chronic GVHD, which translated to low rates of nonrelapse mortality and maintenance of long-term remissions in a subset of patients with multiple myeloma treated at Johns Hopkins Hospital, Baltimore.
“The favorable toxicity profile of alloBMT using PTCy in patients with MM offers the potential to further explore the use of post-transplantation strategies to improve disease control,” wrote Nilanjan Ghosh, MD, of the Levine Cancer Institute, Charlotte, N.C., and his colleagues.
The researchers examined outcomes of 39 patients with multiple myeloma who underwent bone marrow or peripheral blood alloBMT from HLA-matched related/unrelated or haploidentical related donors after either myeloablative or nonmyeloablative conditioning.
Patients who underwent myeloablative conditioning received PTCy 50 mg/kg per day (given intravenously on days 3 and 4 after alloBMT) as GVHD prophylaxis, while those who received nonmyeloablative conditioning received PTCy (same dose and schedule) and additional immunosuppression with mycophenolate mofetil 15 mg/kg (orally two to three times daily, up to 1,000 mg/dose on days 4-35 after transplantation).
At a median follow-up of 10.3 years following alloBMT, 23% of the cohort remained alive and without any evidence of disease. The median overall survival was 4.4 years. The 5-year survival probability was 49% (95% confidence interval, 32%-67%) and 10-year survival probability was 43% (95% CI, 29%-62%).
Following transplantation, 10 (26%) patients achieved a complete response, 8 (21%) had a very good partial response, 6 (15%) had a partial response, and 15 (38%) had progressive disease, the investigators reported (Biol Blood Marrow Transplant. 2017 Nov;23[11]:1903-9).
Among the 36 patients with evidence of donor engraftment, the cumulative incidences of grade 2-4 and grade 3 and 4 acute GVHD were 0.41 (95% CI, 0.25-0.57) and 0.08 (95% CI, 0.01-0.16), respectively. For chronic GVHD, the cumulative incidence was 0.13 (95% CI, 0.02-0.23), and the median time to development was 109 days after alloBMT.
The median progression-free survival was 12 months (95% CI, 7.6-40), and the estimated cumulative incidence of relapse was 0.46 (95% CI, 0.3-0.62) at 1 year and 0.56 (95% CI, 0.41-0.72) at 2 years.
In univariate analysis, achievement of a complete response following alloBMT correlated with overall survival. Factors such as the intensity of the alloBMT conditioning regimen, stem cell source, age, and disease response before alloBMT did not appear to impact overall survival.
“Given the low incidence of GVHD and [nonrelapse mortality] in our study, it is possible that post-transplantation maintenance therapies can be used to improve diseases control,” Dr. Ghosh and his associates wrote. “Emerging data suggest that many anticancer agents may be more active after alloBMT than before transplantation.”
The study was funded in part by grants from the National Institutes of Health. The researchers reported having no relevant financial disclosures.
Post-transplantation cyclophosphamide may help decrease the rate of nonrelapse mortality and graft-versus-host disease (GVHD) in patients who have undergone allogeneic blood or marrow transplantation, according to new findings published in Biology of Blood and Marrow Transplantation.
Even though it is a potentially curative option for patients with poor-risk hematologic malignancies, allogeneic blood or marrow transplantation (alloBMT) has not been commonly used in multiple myeloma because of its association with high nonrelapse mortality and high relapse rates. However, the use of post-transplantation cyclophosphamide (PTCy) in this study led to low rates of both acute and chronic GVHD, which translated to low rates of nonrelapse mortality and maintenance of long-term remissions in a subset of patients with multiple myeloma treated at Johns Hopkins Hospital, Baltimore.
“The favorable toxicity profile of alloBMT using PTCy in patients with MM offers the potential to further explore the use of post-transplantation strategies to improve disease control,” wrote Nilanjan Ghosh, MD, of the Levine Cancer Institute, Charlotte, N.C., and his colleagues.
The researchers examined outcomes of 39 patients with multiple myeloma who underwent bone marrow or peripheral blood alloBMT from HLA-matched related/unrelated or haploidentical related donors after either myeloablative or nonmyeloablative conditioning.
Patients who underwent myeloablative conditioning received PTCy 50 mg/kg per day (given intravenously on days 3 and 4 after alloBMT) as GVHD prophylaxis, while those who received nonmyeloablative conditioning received PTCy (same dose and schedule) and additional immunosuppression with mycophenolate mofetil 15 mg/kg (orally two to three times daily, up to 1,000 mg/dose on days 4-35 after transplantation).
At a median follow-up of 10.3 years following alloBMT, 23% of the cohort remained alive and without any evidence of disease. The median overall survival was 4.4 years. The 5-year survival probability was 49% (95% confidence interval, 32%-67%) and 10-year survival probability was 43% (95% CI, 29%-62%).
Following transplantation, 10 (26%) patients achieved a complete response, 8 (21%) had a very good partial response, 6 (15%) had a partial response, and 15 (38%) had progressive disease, the investigators reported (Biol Blood Marrow Transplant. 2017 Nov;23[11]:1903-9).
Among the 36 patients with evidence of donor engraftment, the cumulative incidences of grade 2-4 and grade 3 and 4 acute GVHD were 0.41 (95% CI, 0.25-0.57) and 0.08 (95% CI, 0.01-0.16), respectively. For chronic GVHD, the cumulative incidence was 0.13 (95% CI, 0.02-0.23), and the median time to development was 109 days after alloBMT.
The median progression-free survival was 12 months (95% CI, 7.6-40), and the estimated cumulative incidence of relapse was 0.46 (95% CI, 0.3-0.62) at 1 year and 0.56 (95% CI, 0.41-0.72) at 2 years.
In univariate analysis, achievement of a complete response following alloBMT correlated with overall survival. Factors such as the intensity of the alloBMT conditioning regimen, stem cell source, age, and disease response before alloBMT did not appear to impact overall survival.
“Given the low incidence of GVHD and [nonrelapse mortality] in our study, it is possible that post-transplantation maintenance therapies can be used to improve diseases control,” Dr. Ghosh and his associates wrote. “Emerging data suggest that many anticancer agents may be more active after alloBMT than before transplantation.”
The study was funded in part by grants from the National Institutes of Health. The researchers reported having no relevant financial disclosures.
Post-transplantation cyclophosphamide may help decrease the rate of nonrelapse mortality and graft-versus-host disease (GVHD) in patients who have undergone allogeneic blood or marrow transplantation, according to new findings published in Biology of Blood and Marrow Transplantation.
Even though it is a potentially curative option for patients with poor-risk hematologic malignancies, allogeneic blood or marrow transplantation (alloBMT) has not been commonly used in multiple myeloma because of its association with high nonrelapse mortality and high relapse rates. However, the use of post-transplantation cyclophosphamide (PTCy) in this study led to low rates of both acute and chronic GVHD, which translated to low rates of nonrelapse mortality and maintenance of long-term remissions in a subset of patients with multiple myeloma treated at Johns Hopkins Hospital, Baltimore.
“The favorable toxicity profile of alloBMT using PTCy in patients with MM offers the potential to further explore the use of post-transplantation strategies to improve disease control,” wrote Nilanjan Ghosh, MD, of the Levine Cancer Institute, Charlotte, N.C., and his colleagues.
The researchers examined outcomes of 39 patients with multiple myeloma who underwent bone marrow or peripheral blood alloBMT from HLA-matched related/unrelated or haploidentical related donors after either myeloablative or nonmyeloablative conditioning.
Patients who underwent myeloablative conditioning received PTCy 50 mg/kg per day (given intravenously on days 3 and 4 after alloBMT) as GVHD prophylaxis, while those who received nonmyeloablative conditioning received PTCy (same dose and schedule) and additional immunosuppression with mycophenolate mofetil 15 mg/kg (orally two to three times daily, up to 1,000 mg/dose on days 4-35 after transplantation).
At a median follow-up of 10.3 years following alloBMT, 23% of the cohort remained alive and without any evidence of disease. The median overall survival was 4.4 years. The 5-year survival probability was 49% (95% confidence interval, 32%-67%) and 10-year survival probability was 43% (95% CI, 29%-62%).
Following transplantation, 10 (26%) patients achieved a complete response, 8 (21%) had a very good partial response, 6 (15%) had a partial response, and 15 (38%) had progressive disease, the investigators reported (Biol Blood Marrow Transplant. 2017 Nov;23[11]:1903-9).
Among the 36 patients with evidence of donor engraftment, the cumulative incidences of grade 2-4 and grade 3 and 4 acute GVHD were 0.41 (95% CI, 0.25-0.57) and 0.08 (95% CI, 0.01-0.16), respectively. For chronic GVHD, the cumulative incidence was 0.13 (95% CI, 0.02-0.23), and the median time to development was 109 days after alloBMT.
The median progression-free survival was 12 months (95% CI, 7.6-40), and the estimated cumulative incidence of relapse was 0.46 (95% CI, 0.3-0.62) at 1 year and 0.56 (95% CI, 0.41-0.72) at 2 years.
In univariate analysis, achievement of a complete response following alloBMT correlated with overall survival. Factors such as the intensity of the alloBMT conditioning regimen, stem cell source, age, and disease response before alloBMT did not appear to impact overall survival.
“Given the low incidence of GVHD and [nonrelapse mortality] in our study, it is possible that post-transplantation maintenance therapies can be used to improve diseases control,” Dr. Ghosh and his associates wrote. “Emerging data suggest that many anticancer agents may be more active after alloBMT than before transplantation.”
The study was funded in part by grants from the National Institutes of Health. The researchers reported having no relevant financial disclosures.
FROM BIOLOGY OF BLOOD AND MARROW TRANSPLANTATION
Key clinical point:
Major finding: At 10.3 years following alloBMT, 16 of 39 (23%) of patients were alive and free of disease. Median overall survival was 4.4 years.
Data source: A single-institution series involving 39 patients with multiple myeloma who underwent alloBMT and received post-transplantation cyclophosphamide.
Disclosures: The study was funded in part by grants from the National Institutes of Health. The researchers reported having no relevant financial disclosures.
FDA approves obinutuzumab for follicular lymphoma
The Food and Drug Administration has approved obinutuzumab in combination with chemotherapy, followed by obinutuzumab alone in those who responded, for people with previously untreated advanced follicular lymphoma (stage II bulky, III or IV).
The most common adverse events associated with obinutuzumab were infusion reactions, low white blood cell count, upper respiratory tract infection, cough, constipation, and diarrhea. The most common significant adverse events are low white blood cell count, low white blood cell count with fever, and low platelet count.
Obinutuzumab is marketed as Gazyva by Genentech.
“Today’s Gazyva approval is an important advance for the thousands of people diagnosed each year with follicular lymphoma who hope to delay disease progression for as long as possible,” said Sarah Horning, MD, chief medical officer and head of global product development at Genentech, in the company press release.
The Food and Drug Administration has approved obinutuzumab in combination with chemotherapy, followed by obinutuzumab alone in those who responded, for people with previously untreated advanced follicular lymphoma (stage II bulky, III or IV).
The most common adverse events associated with obinutuzumab were infusion reactions, low white blood cell count, upper respiratory tract infection, cough, constipation, and diarrhea. The most common significant adverse events are low white blood cell count, low white blood cell count with fever, and low platelet count.
Obinutuzumab is marketed as Gazyva by Genentech.
“Today’s Gazyva approval is an important advance for the thousands of people diagnosed each year with follicular lymphoma who hope to delay disease progression for as long as possible,” said Sarah Horning, MD, chief medical officer and head of global product development at Genentech, in the company press release.
The Food and Drug Administration has approved obinutuzumab in combination with chemotherapy, followed by obinutuzumab alone in those who responded, for people with previously untreated advanced follicular lymphoma (stage II bulky, III or IV).
The most common adverse events associated with obinutuzumab were infusion reactions, low white blood cell count, upper respiratory tract infection, cough, constipation, and diarrhea. The most common significant adverse events are low white blood cell count, low white blood cell count with fever, and low platelet count.
Obinutuzumab is marketed as Gazyva by Genentech.
“Today’s Gazyva approval is an important advance for the thousands of people diagnosed each year with follicular lymphoma who hope to delay disease progression for as long as possible,” said Sarah Horning, MD, chief medical officer and head of global product development at Genentech, in the company press release.
FDA expands approval of obinutuzumab
The US Food and Drug Administration (FDA) has expanded the approved use of obinutuzumab (Gazyva®).
The drug is now approved for use in combination with chemotherapy to treat patients with previously untreated follicular lymphoma (FL) that is advanced (stage II bulky, stage III, or stage IV) .
In patients who respond to this treatment, obinutuzumab monotherapy can be given as maintenance.
The FDA granted this new approval of obinutuzumab to Genentech, Inc. The application for obinutuzumab in this indication received priority review.
The latest FDA approval means obinutuzumab is available in the US for the following indications:
- In combination with chlorambucil to treat chronic lymphocytic leukemia (CLL) in adults who have not had previous CLL treatment
- In combination with bendamustine, followed by obinutuzumab alone, to treat FL in adults who did not respond to a rituximab-containing regimen or whose FL returned after such treatment
- In combination with chemotherapy, followed by obinutuzumab alone in responders, to treat stage II bulky, stage III, or stage IV FL in adults who have not had previous FL treatment.
Phase 3 results
The latest approval of obinutuzumab is based on results from the phase 3 GALLIUM study, which were presented at the 2016 ASH Annual Meeting and published in NEJM in October.
The following are updated data from the obinutuzumab prescribing information.
GALLIUM included 1385 patients with previously untreated non-Hodgkin lymphoma, and 1202 of these patients had advanced FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
At a median observation time of 38 months, the overall response rate was 91% in the obinutuzumab arm and 88% in the rituximab arm. The complete response rates were 28% and 27%, respectively.
The median progression-free survival was not reached in either arm. The hazard ratio, for obinutuzumab compared to rituximab, was 0.72 (95% CI, 0.56-0.93, P=0.0118).
Safety was evaluated based on all 1385 patients in the study, 86% of whom had previously untreated FL and 14% of whom had marginal zone lymphoma.
Serious adverse events (AEs) occurred in 50% of patients in the obinutuzumab arm and 43% in the rituximab arm. Fatal AEs occurred in 5% and 4%, respectively. Infections and second malignancies were the leading causes of these deaths.
The most common AEs (incidence ≥ 20%) observed at least 2% more patients in the obinutuzumab arm were infusion-related reactions, neutropenia, upper respiratory tract infection, cough, constipation, and diarrhea.
The most common grade 3 to 5 AEs (incidence ≥ 5%) observed more frequently in the obinutuzumab arm were neutropenia, infusion-related reactions, febrile neutropenia, and thrombocytopenia. ![]()
The US Food and Drug Administration (FDA) has expanded the approved use of obinutuzumab (Gazyva®).
The drug is now approved for use in combination with chemotherapy to treat patients with previously untreated follicular lymphoma (FL) that is advanced (stage II bulky, stage III, or stage IV) .
In patients who respond to this treatment, obinutuzumab monotherapy can be given as maintenance.
The FDA granted this new approval of obinutuzumab to Genentech, Inc. The application for obinutuzumab in this indication received priority review.
The latest FDA approval means obinutuzumab is available in the US for the following indications:
- In combination with chlorambucil to treat chronic lymphocytic leukemia (CLL) in adults who have not had previous CLL treatment
- In combination with bendamustine, followed by obinutuzumab alone, to treat FL in adults who did not respond to a rituximab-containing regimen or whose FL returned after such treatment
- In combination with chemotherapy, followed by obinutuzumab alone in responders, to treat stage II bulky, stage III, or stage IV FL in adults who have not had previous FL treatment.
Phase 3 results
The latest approval of obinutuzumab is based on results from the phase 3 GALLIUM study, which were presented at the 2016 ASH Annual Meeting and published in NEJM in October.
The following are updated data from the obinutuzumab prescribing information.
GALLIUM included 1385 patients with previously untreated non-Hodgkin lymphoma, and 1202 of these patients had advanced FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
At a median observation time of 38 months, the overall response rate was 91% in the obinutuzumab arm and 88% in the rituximab arm. The complete response rates were 28% and 27%, respectively.
The median progression-free survival was not reached in either arm. The hazard ratio, for obinutuzumab compared to rituximab, was 0.72 (95% CI, 0.56-0.93, P=0.0118).
Safety was evaluated based on all 1385 patients in the study, 86% of whom had previously untreated FL and 14% of whom had marginal zone lymphoma.
Serious adverse events (AEs) occurred in 50% of patients in the obinutuzumab arm and 43% in the rituximab arm. Fatal AEs occurred in 5% and 4%, respectively. Infections and second malignancies were the leading causes of these deaths.
The most common AEs (incidence ≥ 20%) observed at least 2% more patients in the obinutuzumab arm were infusion-related reactions, neutropenia, upper respiratory tract infection, cough, constipation, and diarrhea.
The most common grade 3 to 5 AEs (incidence ≥ 5%) observed more frequently in the obinutuzumab arm were neutropenia, infusion-related reactions, febrile neutropenia, and thrombocytopenia. ![]()
The US Food and Drug Administration (FDA) has expanded the approved use of obinutuzumab (Gazyva®).
The drug is now approved for use in combination with chemotherapy to treat patients with previously untreated follicular lymphoma (FL) that is advanced (stage II bulky, stage III, or stage IV) .
In patients who respond to this treatment, obinutuzumab monotherapy can be given as maintenance.
The FDA granted this new approval of obinutuzumab to Genentech, Inc. The application for obinutuzumab in this indication received priority review.
The latest FDA approval means obinutuzumab is available in the US for the following indications:
- In combination with chlorambucil to treat chronic lymphocytic leukemia (CLL) in adults who have not had previous CLL treatment
- In combination with bendamustine, followed by obinutuzumab alone, to treat FL in adults who did not respond to a rituximab-containing regimen or whose FL returned after such treatment
- In combination with chemotherapy, followed by obinutuzumab alone in responders, to treat stage II bulky, stage III, or stage IV FL in adults who have not had previous FL treatment.
Phase 3 results
The latest approval of obinutuzumab is based on results from the phase 3 GALLIUM study, which were presented at the 2016 ASH Annual Meeting and published in NEJM in October.
The following are updated data from the obinutuzumab prescribing information.
GALLIUM included 1385 patients with previously untreated non-Hodgkin lymphoma, and 1202 of these patients had advanced FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
At a median observation time of 38 months, the overall response rate was 91% in the obinutuzumab arm and 88% in the rituximab arm. The complete response rates were 28% and 27%, respectively.
The median progression-free survival was not reached in either arm. The hazard ratio, for obinutuzumab compared to rituximab, was 0.72 (95% CI, 0.56-0.93, P=0.0118).
Safety was evaluated based on all 1385 patients in the study, 86% of whom had previously untreated FL and 14% of whom had marginal zone lymphoma.
Serious adverse events (AEs) occurred in 50% of patients in the obinutuzumab arm and 43% in the rituximab arm. Fatal AEs occurred in 5% and 4%, respectively. Infections and second malignancies were the leading causes of these deaths.
The most common AEs (incidence ≥ 20%) observed at least 2% more patients in the obinutuzumab arm were infusion-related reactions, neutropenia, upper respiratory tract infection, cough, constipation, and diarrhea.
The most common grade 3 to 5 AEs (incidence ≥ 5%) observed more frequently in the obinutuzumab arm were neutropenia, infusion-related reactions, febrile neutropenia, and thrombocytopenia. ![]()
Rituximab key to survival after transplant for mantle cell lymphoma
The study, which outlines the experience across a variety of different treatment patterns at City of Hope National Medical Center, Duarte, Calif., between January 1997 and November 2013, suggests a “large benefit” of adding rituximab, wrote Matthew G. Mei, MD, of the center’s department of hematology and hematopoietic cell transplantation, and his colleagues. Further, maintenance rituximab was associated with improved survival outcomes in patients with positron emission tomography (PET)-negative status at first complete remission.
The benefit of rituximab “stands out, and adds to the increasing body of evidence supporting this practice for all MCL patients after ASCT, regardless of age and frontline induction regimens,” wrote Dr. Mei and his colleagues (Biol Blood Marrow Transplant 2017 November. doi: 10.1016/j.bbmt.2017.07.006). This was the case even with improvements in early diagnosis and supportive care, and the incorporation of novel agents such as bortezomib, lenalidomide, and ibrutinib, they wrote, noting significantly better outcomes for patients who underwent ASCT after 2007.
In multivariate analysis, maintenance rituximab therapy after ASCT was the single most important factor associated with improvement in progression-free survival (relative risk [RR], .25; 95% confidence interval, .14-.44) and overall survival (RR, .17; 95% CI, .07-.38).
Positron emission tomography scans were done prior to ASCT for 133 patients; after ASCT, 105 (79%) were found to be in a PET-negative complete remission. All but one of the patients with PET-negative disease received rituximab before ASCT. For that PET-negative subset, maintenance rituximab was significantly associated with improvements in progression-free survival (RR, .20; 95% CI, .09-.43) and overall survival (RR, .17; 95% CI, .05-.59).
This study represents one of the largest single-center reports to date on MCL patients who have undergone ASCT, according to the authors. “This study also sets the stage for prospective investigation aiming at optimization of maintenance therapy following ASCT.”
Dr. Mei reported no disclosures, and senior author Lihua E. Budde, MD, PhD, reported being a member of the Lymphoma Research Foundation MCL consortium. The study was supported by research funding from the National Cancer Institute.
This study confirms the value of maintenance rituximab for a large cohort of patients with mantle cell lymphoma who have undergone high-dose chemotherapy and autologous stem cell transplantation outside of clinical trials.
The findings also affirm results of a recent phase 3 randomized trial (LyMa) suggesting that in previously untreated MCL patients who have undergone ASCT, rituximab maintenance is superior to observation in improving overall survival and progression-free survival.
However, the most interesting aspect of this study is the positron emission tomography data. Namely, the benefit of rituximab maintenance was apparent in patients regardless of whether they were in a PET-positive or PET-negative first complete remission at ASCT. “This important finding implies that the benefit of rituximab maintenance after ASCT is present for low- and high-risk MCL patients.”
Despite these confirmatory findings, the treatment landscape for MCL has changed significantly in recent years, particularly with the introduction of treatments such as ibrutinib.
In a clinical trial currently underway, the European Mantle Cell Lymphoma Network is evaluating ibrutinib as an upfront treatment for young and fit patients. Specifically, the study compares first-line ASCT and rituximab maintenance, ASCT with ibrutinib maintenance, or a transplant-free approach with ibrutinib and chemotherapy.
Unless and until the data from this study “redefine the value of ASCT in the ibrutinib era, ASCT and rituximab maintenance should be recommended as the standard treatment after ASCT for transplant-eligible patients with MCL.”
Tobias Roider, MD, and Sascha Dietrich, MD, are with the Department of Medicine V, University of Heidelberg, Germany. Their comments are in an editorial (Biol Blood Marrow Transplant 2017 November. doi: 10.1016/j.bbmt.2017.09.008). The authors reported no financial disclosures or conflicts of interest.
This study confirms the value of maintenance rituximab for a large cohort of patients with mantle cell lymphoma who have undergone high-dose chemotherapy and autologous stem cell transplantation outside of clinical trials.
The findings also affirm results of a recent phase 3 randomized trial (LyMa) suggesting that in previously untreated MCL patients who have undergone ASCT, rituximab maintenance is superior to observation in improving overall survival and progression-free survival.
However, the most interesting aspect of this study is the positron emission tomography data. Namely, the benefit of rituximab maintenance was apparent in patients regardless of whether they were in a PET-positive or PET-negative first complete remission at ASCT. “This important finding implies that the benefit of rituximab maintenance after ASCT is present for low- and high-risk MCL patients.”
Despite these confirmatory findings, the treatment landscape for MCL has changed significantly in recent years, particularly with the introduction of treatments such as ibrutinib.
In a clinical trial currently underway, the European Mantle Cell Lymphoma Network is evaluating ibrutinib as an upfront treatment for young and fit patients. Specifically, the study compares first-line ASCT and rituximab maintenance, ASCT with ibrutinib maintenance, or a transplant-free approach with ibrutinib and chemotherapy.
Unless and until the data from this study “redefine the value of ASCT in the ibrutinib era, ASCT and rituximab maintenance should be recommended as the standard treatment after ASCT for transplant-eligible patients with MCL.”
Tobias Roider, MD, and Sascha Dietrich, MD, are with the Department of Medicine V, University of Heidelberg, Germany. Their comments are in an editorial (Biol Blood Marrow Transplant 2017 November. doi: 10.1016/j.bbmt.2017.09.008). The authors reported no financial disclosures or conflicts of interest.
This study confirms the value of maintenance rituximab for a large cohort of patients with mantle cell lymphoma who have undergone high-dose chemotherapy and autologous stem cell transplantation outside of clinical trials.
The findings also affirm results of a recent phase 3 randomized trial (LyMa) suggesting that in previously untreated MCL patients who have undergone ASCT, rituximab maintenance is superior to observation in improving overall survival and progression-free survival.
However, the most interesting aspect of this study is the positron emission tomography data. Namely, the benefit of rituximab maintenance was apparent in patients regardless of whether they were in a PET-positive or PET-negative first complete remission at ASCT. “This important finding implies that the benefit of rituximab maintenance after ASCT is present for low- and high-risk MCL patients.”
Despite these confirmatory findings, the treatment landscape for MCL has changed significantly in recent years, particularly with the introduction of treatments such as ibrutinib.
In a clinical trial currently underway, the European Mantle Cell Lymphoma Network is evaluating ibrutinib as an upfront treatment for young and fit patients. Specifically, the study compares first-line ASCT and rituximab maintenance, ASCT with ibrutinib maintenance, or a transplant-free approach with ibrutinib and chemotherapy.
Unless and until the data from this study “redefine the value of ASCT in the ibrutinib era, ASCT and rituximab maintenance should be recommended as the standard treatment after ASCT for transplant-eligible patients with MCL.”
Tobias Roider, MD, and Sascha Dietrich, MD, are with the Department of Medicine V, University of Heidelberg, Germany. Their comments are in an editorial (Biol Blood Marrow Transplant 2017 November. doi: 10.1016/j.bbmt.2017.09.008). The authors reported no financial disclosures or conflicts of interest.
The study, which outlines the experience across a variety of different treatment patterns at City of Hope National Medical Center, Duarte, Calif., between January 1997 and November 2013, suggests a “large benefit” of adding rituximab, wrote Matthew G. Mei, MD, of the center’s department of hematology and hematopoietic cell transplantation, and his colleagues. Further, maintenance rituximab was associated with improved survival outcomes in patients with positron emission tomography (PET)-negative status at first complete remission.
The benefit of rituximab “stands out, and adds to the increasing body of evidence supporting this practice for all MCL patients after ASCT, regardless of age and frontline induction regimens,” wrote Dr. Mei and his colleagues (Biol Blood Marrow Transplant 2017 November. doi: 10.1016/j.bbmt.2017.07.006). This was the case even with improvements in early diagnosis and supportive care, and the incorporation of novel agents such as bortezomib, lenalidomide, and ibrutinib, they wrote, noting significantly better outcomes for patients who underwent ASCT after 2007.
In multivariate analysis, maintenance rituximab therapy after ASCT was the single most important factor associated with improvement in progression-free survival (relative risk [RR], .25; 95% confidence interval, .14-.44) and overall survival (RR, .17; 95% CI, .07-.38).
Positron emission tomography scans were done prior to ASCT for 133 patients; after ASCT, 105 (79%) were found to be in a PET-negative complete remission. All but one of the patients with PET-negative disease received rituximab before ASCT. For that PET-negative subset, maintenance rituximab was significantly associated with improvements in progression-free survival (RR, .20; 95% CI, .09-.43) and overall survival (RR, .17; 95% CI, .05-.59).
This study represents one of the largest single-center reports to date on MCL patients who have undergone ASCT, according to the authors. “This study also sets the stage for prospective investigation aiming at optimization of maintenance therapy following ASCT.”
Dr. Mei reported no disclosures, and senior author Lihua E. Budde, MD, PhD, reported being a member of the Lymphoma Research Foundation MCL consortium. The study was supported by research funding from the National Cancer Institute.
The study, which outlines the experience across a variety of different treatment patterns at City of Hope National Medical Center, Duarte, Calif., between January 1997 and November 2013, suggests a “large benefit” of adding rituximab, wrote Matthew G. Mei, MD, of the center’s department of hematology and hematopoietic cell transplantation, and his colleagues. Further, maintenance rituximab was associated with improved survival outcomes in patients with positron emission tomography (PET)-negative status at first complete remission.
The benefit of rituximab “stands out, and adds to the increasing body of evidence supporting this practice for all MCL patients after ASCT, regardless of age and frontline induction regimens,” wrote Dr. Mei and his colleagues (Biol Blood Marrow Transplant 2017 November. doi: 10.1016/j.bbmt.2017.07.006). This was the case even with improvements in early diagnosis and supportive care, and the incorporation of novel agents such as bortezomib, lenalidomide, and ibrutinib, they wrote, noting significantly better outcomes for patients who underwent ASCT after 2007.
In multivariate analysis, maintenance rituximab therapy after ASCT was the single most important factor associated with improvement in progression-free survival (relative risk [RR], .25; 95% confidence interval, .14-.44) and overall survival (RR, .17; 95% CI, .07-.38).
Positron emission tomography scans were done prior to ASCT for 133 patients; after ASCT, 105 (79%) were found to be in a PET-negative complete remission. All but one of the patients with PET-negative disease received rituximab before ASCT. For that PET-negative subset, maintenance rituximab was significantly associated with improvements in progression-free survival (RR, .20; 95% CI, .09-.43) and overall survival (RR, .17; 95% CI, .05-.59).
This study represents one of the largest single-center reports to date on MCL patients who have undergone ASCT, according to the authors. “This study also sets the stage for prospective investigation aiming at optimization of maintenance therapy following ASCT.”
Dr. Mei reported no disclosures, and senior author Lihua E. Budde, MD, PhD, reported being a member of the Lymphoma Research Foundation MCL consortium. The study was supported by research funding from the National Cancer Institute.
FROM BIOLOGY OF BLOOD AND MARROW TRANSPLANTATION
Key clinical point: Over time and in many different patterns, rituximab maintenance therapy stood out as the prominent factor influencing survival in patients with mantle cell lymphoma who undergo autologous stem cell transplant.
Major finding: Maintenance rituximab was significantly associated with superior progression-free survival (relative risk, .25; 95% confidence interval, .14-.44) and overall survival (RR, .17; 95% CI, .07-.38).
Data source: Retrospective analysis of data for 191 patients with MCL who underwent ASCT at a medical center in California between January 1997 and November 2013.
Disclosures: The study was supported by research funding from the National Cancer Institute. Senior author Lihua E. Budde, MD, PhD, reported being a member of the Lymphoma Research Foundation MCL consortium.