User login
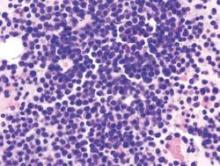
Whole-exome sequencing reveals important mutations in myeloma
The first comprehensive molecular analysis of patients in a clinical trial with myeloma has identified key copy number and structural abnormalities (CNSAs) and mutations that may help target high risk patients for more appropriate treatments, according to a new study published online in the Journal of Clinical Oncology.
“The detection of mutations can improve our ability to detect high-risk patients who experience relapse and die early, but who may benefit from specific therapeutic interventions,” said Dr. Brian Walker, from the Institute of Cancer Research, London, UK, and his colleagues (J Clin Oncol. 2015 Aug 17. doi:10.1200/JCO.2014.59.1503).
Samples were taken from 463 patients who were newly diagnosed with symptomatic myeloma and enrolled onto the National Cancer Research Institute Myeloma XI trial. This was a phase III, open-label trial in which patients were randomized to triplet immunomodulatory drug induction of either cyclophosphamide, thalidomide, and dexamethasone, or cyclophosphamide, lenalidomide, and dexamethasone.
Whole-exome sequencing of the patients identified 15 significantly mutated genes: IRF4, KRAS, NRAS, MAX, HIST1H1E, RB1, EGR1, TP53, TRAF3, FAM46C, DIS3, BRAF, LTB, CYLD, and FGFR3.
With the exception of NRAS and KRAS, all of the genes are mutated at a low percentage. The finding could be of importance, as it indicates that key pathways are deregulated, instead of mutations arising in single genes.
A number of the mutations appear to have an effect on survival. CCND1 mutations were associated with a negative impact on overall survival (2-year OS, 38%; [95% confidence interval, 14%-100%] vs. 80% [95% CI, 76%-84%]; P = .005). TP53 mutations and deletions were also associated with a significant negative impact on progression-free survival (PFS) and overall survival, as were ATM and ATR mutations.
In contrast, mutations in IRF4 had a positive impact on survival, with a trend toward an improvement in progression free survival (2-year PFS, 71%; [95% CI, 50%-100%] vs. 54% [95% CI, 49%-60%]; P = .09) and a significant impact on overall survival (2-year OS, 100% vs. 79% [95% CI, 75%-83%]; P = .05).
Aside from being prognostic for outcomes, some of these mutations are potential therapeutic targets. The deregulation of the RAS/MAPK pathway with the most common mutations being in NRAS and KRAS is a major therapeutic target, while the other targetable pathway is NF-kappa-B.
“Overall, we identified a set of potential actionable mutations comprising 309 targets applicable to 53% of patients,” the researchers wrote. “In the years to come, we foresee this to increase to 440 targets applicable to 62% of patients.”
The study was supported by several U.K. government grants, as well as funds from the National Institute of Health Biomedical Research Centre at the Royal Marsden Hospital. Coauthor Dr. Boyle was supported by the Fédération Française de Recherche sur le Myélome et les Gammapathies grant. Dr. Walker reports receiving honoraria from Celgene and Onyx Pharmaceuticals.
The first comprehensive molecular analysis of patients in a clinical trial with myeloma has identified key copy number and structural abnormalities (CNSAs) and mutations that may help target high risk patients for more appropriate treatments, according to a new study published online in the Journal of Clinical Oncology.
“The detection of mutations can improve our ability to detect high-risk patients who experience relapse and die early, but who may benefit from specific therapeutic interventions,” said Dr. Brian Walker, from the Institute of Cancer Research, London, UK, and his colleagues (J Clin Oncol. 2015 Aug 17. doi:10.1200/JCO.2014.59.1503).
Samples were taken from 463 patients who were newly diagnosed with symptomatic myeloma and enrolled onto the National Cancer Research Institute Myeloma XI trial. This was a phase III, open-label trial in which patients were randomized to triplet immunomodulatory drug induction of either cyclophosphamide, thalidomide, and dexamethasone, or cyclophosphamide, lenalidomide, and dexamethasone.
Whole-exome sequencing of the patients identified 15 significantly mutated genes: IRF4, KRAS, NRAS, MAX, HIST1H1E, RB1, EGR1, TP53, TRAF3, FAM46C, DIS3, BRAF, LTB, CYLD, and FGFR3.
With the exception of NRAS and KRAS, all of the genes are mutated at a low percentage. The finding could be of importance, as it indicates that key pathways are deregulated, instead of mutations arising in single genes.
A number of the mutations appear to have an effect on survival. CCND1 mutations were associated with a negative impact on overall survival (2-year OS, 38%; [95% confidence interval, 14%-100%] vs. 80% [95% CI, 76%-84%]; P = .005). TP53 mutations and deletions were also associated with a significant negative impact on progression-free survival (PFS) and overall survival, as were ATM and ATR mutations.
In contrast, mutations in IRF4 had a positive impact on survival, with a trend toward an improvement in progression free survival (2-year PFS, 71%; [95% CI, 50%-100%] vs. 54% [95% CI, 49%-60%]; P = .09) and a significant impact on overall survival (2-year OS, 100% vs. 79% [95% CI, 75%-83%]; P = .05).
Aside from being prognostic for outcomes, some of these mutations are potential therapeutic targets. The deregulation of the RAS/MAPK pathway with the most common mutations being in NRAS and KRAS is a major therapeutic target, while the other targetable pathway is NF-kappa-B.
“Overall, we identified a set of potential actionable mutations comprising 309 targets applicable to 53% of patients,” the researchers wrote. “In the years to come, we foresee this to increase to 440 targets applicable to 62% of patients.”
The study was supported by several U.K. government grants, as well as funds from the National Institute of Health Biomedical Research Centre at the Royal Marsden Hospital. Coauthor Dr. Boyle was supported by the Fédération Française de Recherche sur le Myélome et les Gammapathies grant. Dr. Walker reports receiving honoraria from Celgene and Onyx Pharmaceuticals.
The first comprehensive molecular analysis of patients in a clinical trial with myeloma has identified key copy number and structural abnormalities (CNSAs) and mutations that may help target high risk patients for more appropriate treatments, according to a new study published online in the Journal of Clinical Oncology.
“The detection of mutations can improve our ability to detect high-risk patients who experience relapse and die early, but who may benefit from specific therapeutic interventions,” said Dr. Brian Walker, from the Institute of Cancer Research, London, UK, and his colleagues (J Clin Oncol. 2015 Aug 17. doi:10.1200/JCO.2014.59.1503).
Samples were taken from 463 patients who were newly diagnosed with symptomatic myeloma and enrolled onto the National Cancer Research Institute Myeloma XI trial. This was a phase III, open-label trial in which patients were randomized to triplet immunomodulatory drug induction of either cyclophosphamide, thalidomide, and dexamethasone, or cyclophosphamide, lenalidomide, and dexamethasone.
Whole-exome sequencing of the patients identified 15 significantly mutated genes: IRF4, KRAS, NRAS, MAX, HIST1H1E, RB1, EGR1, TP53, TRAF3, FAM46C, DIS3, BRAF, LTB, CYLD, and FGFR3.
With the exception of NRAS and KRAS, all of the genes are mutated at a low percentage. The finding could be of importance, as it indicates that key pathways are deregulated, instead of mutations arising in single genes.
A number of the mutations appear to have an effect on survival. CCND1 mutations were associated with a negative impact on overall survival (2-year OS, 38%; [95% confidence interval, 14%-100%] vs. 80% [95% CI, 76%-84%]; P = .005). TP53 mutations and deletions were also associated with a significant negative impact on progression-free survival (PFS) and overall survival, as were ATM and ATR mutations.
In contrast, mutations in IRF4 had a positive impact on survival, with a trend toward an improvement in progression free survival (2-year PFS, 71%; [95% CI, 50%-100%] vs. 54% [95% CI, 49%-60%]; P = .09) and a significant impact on overall survival (2-year OS, 100% vs. 79% [95% CI, 75%-83%]; P = .05).
Aside from being prognostic for outcomes, some of these mutations are potential therapeutic targets. The deregulation of the RAS/MAPK pathway with the most common mutations being in NRAS and KRAS is a major therapeutic target, while the other targetable pathway is NF-kappa-B.
“Overall, we identified a set of potential actionable mutations comprising 309 targets applicable to 53% of patients,” the researchers wrote. “In the years to come, we foresee this to increase to 440 targets applicable to 62% of patients.”
The study was supported by several U.K. government grants, as well as funds from the National Institute of Health Biomedical Research Centre at the Royal Marsden Hospital. Coauthor Dr. Boyle was supported by the Fédération Française de Recherche sur le Myélome et les Gammapathies grant. Dr. Walker reports receiving honoraria from Celgene and Onyx Pharmaceuticals.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: A comprehensive analysis identified key copy number and structural abnormalities (CNSAs) and mutations in multiple myeloma.
Major finding: Fifteen significantly mutated genes were identified, primarily mutations in the RAS (43%) and nuclear factor kB pathway (17%).
Data source: Whole-exome sequencing for 463 patients who presented with myeloma and were enrolled onto the National Cancer Research Institute Myeloma XI trial.
Disclosures: The study was supported by several U.K. government grants, as well as funds from the National Institute of Health Biomedical Research Centre at the Royal Marsden Hospital, London. Coauthor Dr. Boyle was supported by the Fédération Française de Recherche sur le Myélome et les Gammapathies grant. Dr. Walker reports receiving honoraria from Celgene and Onyx Pharmaceuticals.
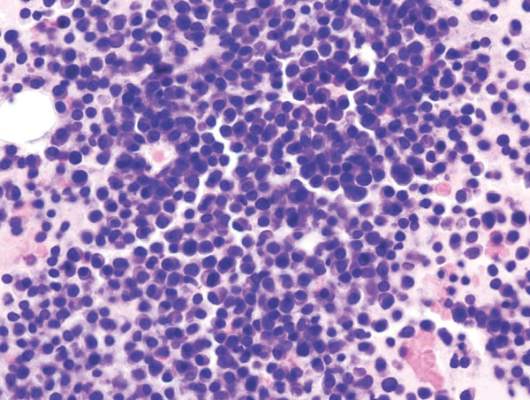
CTL019 induced a durable complete response in refractory MM
The first recipient of CTL019 for advanced refractory multiple myeloma achieved a durable complete response without developing cytokine release syndrome, according to a study published Sept. 9 in the New England Journal of Medicine.
“Twelve months after transplantation, the patient had no evidence of monoclonal immunoglobulin on serum and urine immunofixation and no clinical signs or symptoms of multiple myeloma,” said Dr. Alfred Garfall and his associates at the University of Pennsylvania in Philadelphia. “This response was achieved despite the absence of CD19 expression in 99.95% of the patient’s neoplastic plasma cells.”
CTL019 consists of autologous T cells modified to express an anti-CD19 chimeric antigen receptor (CAR) from a lentiviral vector. The cell therapy has yielded promising results in relapsed/refractory CLL and ALL,but was overlooked in MM because it was thought to infrequently express CD19, the researchers said.
“Several reports, however, have suggested that a minor component of the MM clone with drug-resistant, disease-propagating properties has a B-cell (i.e., CD19-positive) phenotype,” they noted. “In addition, our unpublished observations suggest that neoplastic plasma cells express low levels of CD19” (N Engl J Med. 2015 Sep 9;373:1040-7).
In response, they designed a pilot trial of adults whose MM relapsed or progressed within a year after initial autologous stem cell transplant. The first participant, a 43-year-old woman with IgA kappa MM, partially responded to lenalidomide, bortezomib, and dexamethasone but progressed when therapy was paused to collect stem cells for transplant. She then partially responded to cisplatin, doxorubicin, cyclophosphamide, and etoposide followed by high-dose melphalan and ASCT, but progressed again and continued to worsen despite a total of nine lines of therapy. A bone marrow sample revealed more than 95% plasma cells when the patient began the CTL019 trial, the researchers said.
For the study, the patient received a lower melphalan dose (140 mg/m2 of body surface area), followed by ASCT, CTL019 starting 2 weeks later, and maintenance lenalidomide. On day 100, her tumor burden had dropped by 5-log10, the researchers said. She also did not develop cytokine release syndrome, they added.
So far, 10 patients have been treated on study, of whom six remain progression free, according to the investigators. “The only additional CTL019-attributable toxic effects observed have been one instance of grade 1 cytokine release syndrome and one instance of grade 3 enterocolitis due to autologous graft-versus-host disease,” they reported.
Novartis supported the study and approved the manuscript. The work was also funded by the National Institutes of Health, the International Society for Advancement of Cytometry, the University of Pennsylvania Institute or Translational Medicine and Therapeutics, and a Conquer Cancer Foundation Young Investigator Award. The University of Pennsylvania has licensed technologies involved in this trial to Novartis. Several scientists involved in this trial hold patents for these technologies. As a result of the licensing relationship with Novartis, the University of Pennsylvania receives significant financial benefit, and these inventors have benefited financially or may benefit in the future.
The first recipient of CTL019 for advanced refractory multiple myeloma achieved a durable complete response without developing cytokine release syndrome, according to a study published Sept. 9 in the New England Journal of Medicine.
“Twelve months after transplantation, the patient had no evidence of monoclonal immunoglobulin on serum and urine immunofixation and no clinical signs or symptoms of multiple myeloma,” said Dr. Alfred Garfall and his associates at the University of Pennsylvania in Philadelphia. “This response was achieved despite the absence of CD19 expression in 99.95% of the patient’s neoplastic plasma cells.”
CTL019 consists of autologous T cells modified to express an anti-CD19 chimeric antigen receptor (CAR) from a lentiviral vector. The cell therapy has yielded promising results in relapsed/refractory CLL and ALL,but was overlooked in MM because it was thought to infrequently express CD19, the researchers said.
“Several reports, however, have suggested that a minor component of the MM clone with drug-resistant, disease-propagating properties has a B-cell (i.e., CD19-positive) phenotype,” they noted. “In addition, our unpublished observations suggest that neoplastic plasma cells express low levels of CD19” (N Engl J Med. 2015 Sep 9;373:1040-7).
In response, they designed a pilot trial of adults whose MM relapsed or progressed within a year after initial autologous stem cell transplant. The first participant, a 43-year-old woman with IgA kappa MM, partially responded to lenalidomide, bortezomib, and dexamethasone but progressed when therapy was paused to collect stem cells for transplant. She then partially responded to cisplatin, doxorubicin, cyclophosphamide, and etoposide followed by high-dose melphalan and ASCT, but progressed again and continued to worsen despite a total of nine lines of therapy. A bone marrow sample revealed more than 95% plasma cells when the patient began the CTL019 trial, the researchers said.
For the study, the patient received a lower melphalan dose (140 mg/m2 of body surface area), followed by ASCT, CTL019 starting 2 weeks later, and maintenance lenalidomide. On day 100, her tumor burden had dropped by 5-log10, the researchers said. She also did not develop cytokine release syndrome, they added.
So far, 10 patients have been treated on study, of whom six remain progression free, according to the investigators. “The only additional CTL019-attributable toxic effects observed have been one instance of grade 1 cytokine release syndrome and one instance of grade 3 enterocolitis due to autologous graft-versus-host disease,” they reported.
Novartis supported the study and approved the manuscript. The work was also funded by the National Institutes of Health, the International Society for Advancement of Cytometry, the University of Pennsylvania Institute or Translational Medicine and Therapeutics, and a Conquer Cancer Foundation Young Investigator Award. The University of Pennsylvania has licensed technologies involved in this trial to Novartis. Several scientists involved in this trial hold patents for these technologies. As a result of the licensing relationship with Novartis, the University of Pennsylvania receives significant financial benefit, and these inventors have benefited financially or may benefit in the future.
The first recipient of CTL019 for advanced refractory multiple myeloma achieved a durable complete response without developing cytokine release syndrome, according to a study published Sept. 9 in the New England Journal of Medicine.
“Twelve months after transplantation, the patient had no evidence of monoclonal immunoglobulin on serum and urine immunofixation and no clinical signs or symptoms of multiple myeloma,” said Dr. Alfred Garfall and his associates at the University of Pennsylvania in Philadelphia. “This response was achieved despite the absence of CD19 expression in 99.95% of the patient’s neoplastic plasma cells.”
CTL019 consists of autologous T cells modified to express an anti-CD19 chimeric antigen receptor (CAR) from a lentiviral vector. The cell therapy has yielded promising results in relapsed/refractory CLL and ALL,but was overlooked in MM because it was thought to infrequently express CD19, the researchers said.
“Several reports, however, have suggested that a minor component of the MM clone with drug-resistant, disease-propagating properties has a B-cell (i.e., CD19-positive) phenotype,” they noted. “In addition, our unpublished observations suggest that neoplastic plasma cells express low levels of CD19” (N Engl J Med. 2015 Sep 9;373:1040-7).
In response, they designed a pilot trial of adults whose MM relapsed or progressed within a year after initial autologous stem cell transplant. The first participant, a 43-year-old woman with IgA kappa MM, partially responded to lenalidomide, bortezomib, and dexamethasone but progressed when therapy was paused to collect stem cells for transplant. She then partially responded to cisplatin, doxorubicin, cyclophosphamide, and etoposide followed by high-dose melphalan and ASCT, but progressed again and continued to worsen despite a total of nine lines of therapy. A bone marrow sample revealed more than 95% plasma cells when the patient began the CTL019 trial, the researchers said.
For the study, the patient received a lower melphalan dose (140 mg/m2 of body surface area), followed by ASCT, CTL019 starting 2 weeks later, and maintenance lenalidomide. On day 100, her tumor burden had dropped by 5-log10, the researchers said. She also did not develop cytokine release syndrome, they added.
So far, 10 patients have been treated on study, of whom six remain progression free, according to the investigators. “The only additional CTL019-attributable toxic effects observed have been one instance of grade 1 cytokine release syndrome and one instance of grade 3 enterocolitis due to autologous graft-versus-host disease,” they reported.
Novartis supported the study and approved the manuscript. The work was also funded by the National Institutes of Health, the International Society for Advancement of Cytometry, the University of Pennsylvania Institute or Translational Medicine and Therapeutics, and a Conquer Cancer Foundation Young Investigator Award. The University of Pennsylvania has licensed technologies involved in this trial to Novartis. Several scientists involved in this trial hold patents for these technologies. As a result of the licensing relationship with Novartis, the University of Pennsylvania receives significant financial benefit, and these inventors have benefited financially or may benefit in the future.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: CTL019 cell therapy after stem cell transplant led to a durable complete response that persisted at 12 months in a patient with advanced, refractory multiple myeloma.
Major finding: The patient received a lower melphalan dose (140 mg/m2 of body surface area), followed by ASCT, CTL019 starting 2 weeks later, and maintenance lenalidomide. On day 100, her tumor burden had dropped by 5-log10.
Data source: Ongoing pilot trial of CTL019 for patients with MM who relapsed within 12 months of their first transplant.
Disclosures: Novartis funded the study and approved the manuscript. The work was also funded by the National Institutes of Health, the International Society for Advancement of Cytometry, the University of Pennsylvania Institute or Translational Medicine and Therapeutics, and a Conquer Cancer Foundation Young Investigator Award. The University of Pennsylvania has licensed technologies involved in this trial to Novartis. Several scientists involved in this trial hold patents for these technologies. As a result of the licensing relationship with Novartis, the University of Pennsylvania receives significant financial benefit, and these inventors have benefited financially or may benefit in the future.
Model may aid cancer drug discovery

Photo by Darren Baker
Researchers say they have created a model that can show how nearly any drug behaves in P-glycoprotein (P-gp), a protein associated with chemotherapy failure.
The team developed this computer-generated model to overcome the problem of relying on static images for the structure of P-gp.
When the researchers introduced drugs into the model, the drugs responded the way they do in real life and behaved according to predictions.
John G. Wise, PhD, of Southern Methodist University in Dallas, Texas, and his colleagues described the model in Biochemistry.
“The value of this fundamental research is that it generates dynamic mechanisms that let us understand something in biochemistry, in biology,” Dr Wise said. “And by understanding P-gp in such detail, we can now think of ways to better and more specifically inhibit it.”
Dr Wise and his colleagues noted that P-gp protects cells by pumping out toxins, but that can include chemotherapy drugs. So inhibiting P-gp’s pumping action might circumvent chemotherapy failure.
With than in mind, the team tested tariquidar, a P-gp inhibitor in clinical trials, in their model.
It hasn’t been clear exactly where tariquidar binds in P-gp. But the model showed the drug prefers to bind high in the protein. Tariquidar also behaved as expected. It wasn’t effectively pumped from the cell.
“Now we have more details on how tariquidar inhibits P-gp, where it inhibits, and what it’s actually binding to,” Dr Wise said.
He and his colleagues also used their model to uncover additional details about the behavior of other drugs in P-gp.
“For a long time, it’s been thought that there are at least a couple of distinct binding sites for drugs,” Dr Wise said.
“Sure enough, with our models, we found that [the chemotherapeutic agent] daunorubicin, at least, prefers to bind on one side of the P-gp model, while verapamil—a commonly prescribed blood pressure medicine—prefers the other side.”
Not only did the researchers show computationally that there are 2 different starting points for drugs, they also showed that there are 2 different pathways to get the drugs through.
“The 2 different drugs start at different sites, and they’re funneled to the outside by being pushed by the protein,” Dr Wise said. “But the actual parts of the protein that are pushing the drugs out are different.”
Drug discovery
Being able to watch molecular machinery up close, while it is doing its job the way it does in real life, may spark new drug discoveries to fight cancer, Dr Wise said.
“Having an accurate model that actually moves—that shows the dynamics of the thing—is incredibly helpful in developing therapies against a molecular target to inhibit it,” Dr Wise said. “The only other ways to do it are blind, and the chances of success using blind methods are very low.”
“Scientists have tried for 30 years to find inhibitors of this pump and have done it without knowing the structure and with only little knowledge about the mechanism, screening more or less blindly for compounds that inhibit the thing.”
“They found drugs that worked in the test tube and that worked in cultured cells but that didn’t work in the patient. With our model, because we can see the pump moving, we can probably predict better what’s going to make an inhibitor actually work well.”
Dr Wise and his colleagues used the P-gp model to virtually screen millions of publicly available compounds. They discovered 3 new drug leads that could ultimately inhibit P-gp and offer better odds of survival to prostate cancer patients.
The researchers reported these findings in Pharmacology Research & Perspectives.
Creating the model
To build the P-gp model, Dr Wise and his colleagues used static structures from the US Protein Data Bank repository. They used structures showing various stages of transport to simulate 4 points of reference.
From there, the team fed a supercomputer parameters and characteristics of the protein, as well as how it should behave physically, including when kinetic energy was added to bring the protein and its surrounding membrane and water up to body temperature.
The animated model resulted from calculating differences between 2 structures and using targeted molecular dynamics programs to slightly nudge the model to the next step.
“You do that several million times and make several trillion calculations, and you arrive at the next structure,” Dr Wise said. “In this way, we can nudge P-gp through a full catalytic transport cycle.”
Finally, using a docking program, the researchers individually introduced daunorubicin and other drugs into the protein and watched the drugs move through P-gp’s catalytic cycle.
“What happened was: the drugs moved,” Dr Wise said. “And they moved the way they should move, clinically, biochemically, physiologically, to pump the compounds out of the cell.”
Challenging the model
The researchers ran a critical control to further test if the model worked.
“We thought maybe anything you put in the protein, relevant or not, would get pumped through,” Dr Wise said. “So we put in something that is not a transport substrate of P-gp, something that, biochemically, would never be transported by P-gp.”
“We put it in, starting where daunorubicin is effectively pumped out, and, very quickly, the compound left the protein. But it left the opposite way, back into the cell. This experiment gave us more confidence that what we are seeing in these models is reflecting what happens in the cell.”
Dr Wise admitted that, until he saw it for himself, he had doubts the virtual P-gp model would behave like real-life P-gp.
“It’s a crude approximation of a complex, sophisticated human protein, but it’s so much better than the static images available now,” Dr Wise said.
“I’ve got to emphasize for all the disbelievers, for the ‘culture of doubters’ out there, that this model works. It moves the drugs through the membrane. That speaks for itself. What P-gp does in the cell, cancerous or normal, it does in our simulations.” ![]()

Photo by Darren Baker
Researchers say they have created a model that can show how nearly any drug behaves in P-glycoprotein (P-gp), a protein associated with chemotherapy failure.
The team developed this computer-generated model to overcome the problem of relying on static images for the structure of P-gp.
When the researchers introduced drugs into the model, the drugs responded the way they do in real life and behaved according to predictions.
John G. Wise, PhD, of Southern Methodist University in Dallas, Texas, and his colleagues described the model in Biochemistry.
“The value of this fundamental research is that it generates dynamic mechanisms that let us understand something in biochemistry, in biology,” Dr Wise said. “And by understanding P-gp in such detail, we can now think of ways to better and more specifically inhibit it.”
Dr Wise and his colleagues noted that P-gp protects cells by pumping out toxins, but that can include chemotherapy drugs. So inhibiting P-gp’s pumping action might circumvent chemotherapy failure.
With than in mind, the team tested tariquidar, a P-gp inhibitor in clinical trials, in their model.
It hasn’t been clear exactly where tariquidar binds in P-gp. But the model showed the drug prefers to bind high in the protein. Tariquidar also behaved as expected. It wasn’t effectively pumped from the cell.
“Now we have more details on how tariquidar inhibits P-gp, where it inhibits, and what it’s actually binding to,” Dr Wise said.
He and his colleagues also used their model to uncover additional details about the behavior of other drugs in P-gp.
“For a long time, it’s been thought that there are at least a couple of distinct binding sites for drugs,” Dr Wise said.
“Sure enough, with our models, we found that [the chemotherapeutic agent] daunorubicin, at least, prefers to bind on one side of the P-gp model, while verapamil—a commonly prescribed blood pressure medicine—prefers the other side.”
Not only did the researchers show computationally that there are 2 different starting points for drugs, they also showed that there are 2 different pathways to get the drugs through.
“The 2 different drugs start at different sites, and they’re funneled to the outside by being pushed by the protein,” Dr Wise said. “But the actual parts of the protein that are pushing the drugs out are different.”
Drug discovery
Being able to watch molecular machinery up close, while it is doing its job the way it does in real life, may spark new drug discoveries to fight cancer, Dr Wise said.
“Having an accurate model that actually moves—that shows the dynamics of the thing—is incredibly helpful in developing therapies against a molecular target to inhibit it,” Dr Wise said. “The only other ways to do it are blind, and the chances of success using blind methods are very low.”
“Scientists have tried for 30 years to find inhibitors of this pump and have done it without knowing the structure and with only little knowledge about the mechanism, screening more or less blindly for compounds that inhibit the thing.”
“They found drugs that worked in the test tube and that worked in cultured cells but that didn’t work in the patient. With our model, because we can see the pump moving, we can probably predict better what’s going to make an inhibitor actually work well.”
Dr Wise and his colleagues used the P-gp model to virtually screen millions of publicly available compounds. They discovered 3 new drug leads that could ultimately inhibit P-gp and offer better odds of survival to prostate cancer patients.
The researchers reported these findings in Pharmacology Research & Perspectives.
Creating the model
To build the P-gp model, Dr Wise and his colleagues used static structures from the US Protein Data Bank repository. They used structures showing various stages of transport to simulate 4 points of reference.
From there, the team fed a supercomputer parameters and characteristics of the protein, as well as how it should behave physically, including when kinetic energy was added to bring the protein and its surrounding membrane and water up to body temperature.
The animated model resulted from calculating differences between 2 structures and using targeted molecular dynamics programs to slightly nudge the model to the next step.
“You do that several million times and make several trillion calculations, and you arrive at the next structure,” Dr Wise said. “In this way, we can nudge P-gp through a full catalytic transport cycle.”
Finally, using a docking program, the researchers individually introduced daunorubicin and other drugs into the protein and watched the drugs move through P-gp’s catalytic cycle.
“What happened was: the drugs moved,” Dr Wise said. “And they moved the way they should move, clinically, biochemically, physiologically, to pump the compounds out of the cell.”
Challenging the model
The researchers ran a critical control to further test if the model worked.
“We thought maybe anything you put in the protein, relevant or not, would get pumped through,” Dr Wise said. “So we put in something that is not a transport substrate of P-gp, something that, biochemically, would never be transported by P-gp.”
“We put it in, starting where daunorubicin is effectively pumped out, and, very quickly, the compound left the protein. But it left the opposite way, back into the cell. This experiment gave us more confidence that what we are seeing in these models is reflecting what happens in the cell.”
Dr Wise admitted that, until he saw it for himself, he had doubts the virtual P-gp model would behave like real-life P-gp.
“It’s a crude approximation of a complex, sophisticated human protein, but it’s so much better than the static images available now,” Dr Wise said.
“I’ve got to emphasize for all the disbelievers, for the ‘culture of doubters’ out there, that this model works. It moves the drugs through the membrane. That speaks for itself. What P-gp does in the cell, cancerous or normal, it does in our simulations.” ![]()

Photo by Darren Baker
Researchers say they have created a model that can show how nearly any drug behaves in P-glycoprotein (P-gp), a protein associated with chemotherapy failure.
The team developed this computer-generated model to overcome the problem of relying on static images for the structure of P-gp.
When the researchers introduced drugs into the model, the drugs responded the way they do in real life and behaved according to predictions.
John G. Wise, PhD, of Southern Methodist University in Dallas, Texas, and his colleagues described the model in Biochemistry.
“The value of this fundamental research is that it generates dynamic mechanisms that let us understand something in biochemistry, in biology,” Dr Wise said. “And by understanding P-gp in such detail, we can now think of ways to better and more specifically inhibit it.”
Dr Wise and his colleagues noted that P-gp protects cells by pumping out toxins, but that can include chemotherapy drugs. So inhibiting P-gp’s pumping action might circumvent chemotherapy failure.
With than in mind, the team tested tariquidar, a P-gp inhibitor in clinical trials, in their model.
It hasn’t been clear exactly where tariquidar binds in P-gp. But the model showed the drug prefers to bind high in the protein. Tariquidar also behaved as expected. It wasn’t effectively pumped from the cell.
“Now we have more details on how tariquidar inhibits P-gp, where it inhibits, and what it’s actually binding to,” Dr Wise said.
He and his colleagues also used their model to uncover additional details about the behavior of other drugs in P-gp.
“For a long time, it’s been thought that there are at least a couple of distinct binding sites for drugs,” Dr Wise said.
“Sure enough, with our models, we found that [the chemotherapeutic agent] daunorubicin, at least, prefers to bind on one side of the P-gp model, while verapamil—a commonly prescribed blood pressure medicine—prefers the other side.”
Not only did the researchers show computationally that there are 2 different starting points for drugs, they also showed that there are 2 different pathways to get the drugs through.
“The 2 different drugs start at different sites, and they’re funneled to the outside by being pushed by the protein,” Dr Wise said. “But the actual parts of the protein that are pushing the drugs out are different.”
Drug discovery
Being able to watch molecular machinery up close, while it is doing its job the way it does in real life, may spark new drug discoveries to fight cancer, Dr Wise said.
“Having an accurate model that actually moves—that shows the dynamics of the thing—is incredibly helpful in developing therapies against a molecular target to inhibit it,” Dr Wise said. “The only other ways to do it are blind, and the chances of success using blind methods are very low.”
“Scientists have tried for 30 years to find inhibitors of this pump and have done it without knowing the structure and with only little knowledge about the mechanism, screening more or less blindly for compounds that inhibit the thing.”
“They found drugs that worked in the test tube and that worked in cultured cells but that didn’t work in the patient. With our model, because we can see the pump moving, we can probably predict better what’s going to make an inhibitor actually work well.”
Dr Wise and his colleagues used the P-gp model to virtually screen millions of publicly available compounds. They discovered 3 new drug leads that could ultimately inhibit P-gp and offer better odds of survival to prostate cancer patients.
The researchers reported these findings in Pharmacology Research & Perspectives.
Creating the model
To build the P-gp model, Dr Wise and his colleagues used static structures from the US Protein Data Bank repository. They used structures showing various stages of transport to simulate 4 points of reference.
From there, the team fed a supercomputer parameters and characteristics of the protein, as well as how it should behave physically, including when kinetic energy was added to bring the protein and its surrounding membrane and water up to body temperature.
The animated model resulted from calculating differences between 2 structures and using targeted molecular dynamics programs to slightly nudge the model to the next step.
“You do that several million times and make several trillion calculations, and you arrive at the next structure,” Dr Wise said. “In this way, we can nudge P-gp through a full catalytic transport cycle.”
Finally, using a docking program, the researchers individually introduced daunorubicin and other drugs into the protein and watched the drugs move through P-gp’s catalytic cycle.
“What happened was: the drugs moved,” Dr Wise said. “And they moved the way they should move, clinically, biochemically, physiologically, to pump the compounds out of the cell.”
Challenging the model
The researchers ran a critical control to further test if the model worked.
“We thought maybe anything you put in the protein, relevant or not, would get pumped through,” Dr Wise said. “So we put in something that is not a transport substrate of P-gp, something that, biochemically, would never be transported by P-gp.”
“We put it in, starting where daunorubicin is effectively pumped out, and, very quickly, the compound left the protein. But it left the opposite way, back into the cell. This experiment gave us more confidence that what we are seeing in these models is reflecting what happens in the cell.”
Dr Wise admitted that, until he saw it for himself, he had doubts the virtual P-gp model would behave like real-life P-gp.
“It’s a crude approximation of a complex, sophisticated human protein, but it’s so much better than the static images available now,” Dr Wise said.
“I’ve got to emphasize for all the disbelievers, for the ‘culture of doubters’ out there, that this model works. It moves the drugs through the membrane. That speaks for itself. What P-gp does in the cell, cancerous or normal, it does in our simulations.” ![]()
Blood cancer drugs set to be removed from CDF

Photo courtesy of CDC
England’s National Health Service (NHS) plans to remove several drugs used to treat hematologic malignancies from the Cancer Drugs Fund (CDF).
The plan is that, as of November 4, 2015, pomalidomide, lenalidomide, ibrutinib, dasatinib, brentuximab, bosutinib, and bendamustine will no longer be funded via the CDF for certain indications.
Ofatumumab was removed from the CDF list yesterday but is now available through the NHS.
Drugs used to treat solid tumor malignancies are set to be de-funded through CDF in November as well.
However, the NHS said the proposal to remove a drug from the CDF is not necessarily a final decision.
In cases where a drug offers enough clinical benefit, the pharmaceutical company developing that drug has the opportunity to reduce the price they are asking the NHS to pay to ensure that it achieves a satisfactory level of value for money. The NHS said a number of such negotiations are underway.
In addition, patients who are currently receiving the drugs set to be removed from the CDF will continue to have access to those drugs.
About the CDF and the NHS
The CDF—set up in 2010 and currently due to run until March 2016—is money the government has set aside to pay for cancer drugs that haven’t been approved by the National Institute for Health and Care Excellence (NICE) and aren’t available within the NHS in England. Most cancer drugs are routinely funded outside of the CDF.
NHS England and NICE are planning to consult on a proposed new system for commissioning cancer drugs. The NHS said the new system will be designed to provide the agency with a more systematic approach to getting the best price for cancer drugs.
Reason for drug removals
The NHS previously increased the budget for the CDF from £200 million in 2013/14, to £280 million in 2014/15, and £340 million from April 2015. This represents a total increase of 70% since August 2014.
However, current projections suggest that spending would rise to around £410 million for this year, an over-spend of £70 million, in the absence of further prioritization. The NHS said this money could be used for other aspects of cancer treatment or NHS services for other patient groups.
Therefore, some drugs are set to be removed from the CDF. The NHS said all decisions on drugs to be maintained in the CDF were based on the advice of clinicians, the best available evidence, and the cost of the treatment.
“There is no escaping the fact that we face a difficult set of choices, but it is our duty to ensure we get maximum value from every penny available on behalf of patients,” said Peter Clark, chair of the CDF.
“We must ensure we invest in those treatments that offer the most benefit, based on rigorous evidence-based clinical analysis and an assessment of the cost of those treatments.”
While de-funding certain drugs will reduce costs, the CDF is not expected to be back on budget this financial year. The NHS does expect the CDF will be operating within its budget during 2016/17.
Blood cancer drugs to be removed
The following drugs are currently on the CDF list for the following indications, but they are set to be de-listed on November 4, 2015.
Bendamustine
For the treatment of chronic lymphocytic leukemia (CLL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- CLL (not licensed in this indication)
- Second-line indication, third-line indication, or fourth-line indication
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
For the treatment of relapsed mantle cell lymphoma (MCL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MCL
- Option for second- or subsequent-line chemotherapy
- No previous treatment with bendamustine
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
*Bendamustine will remain on the CDF for other indications.
Bosutinib
For the treatment of refractory, chronic phase chronic myeloid leukemia (CML) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Chronic phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
For the treatment of refractory, accelerated phase CML where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
For the treatment of accelerated phase CML where there is intolerance of treatments and where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Significant intolerance to dasatinib (grade 3 or 4 adverse events; if dasatinib accessed via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
*Bosutinib will still be available through the CDF for patients with chronic phase CML that is intolerant of other treatments.
Brentuximab
For the treatment of refractory, systemic anaplastic lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory systemic anaplastic large-cell lymphoma
For the treatment of relapsed or refractory CD30+ Hodgkin lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory CD30+ Hodgkin lymphoma
- Following autologous stem cell transplant or following at least 2 prior therapies when autologous stem cell transplant or multi-agent chemotherapy is not an option
Dasatinib
For the treatment of Philadelphia-chromosome-positive (Ph+) acute lymphoblastic leukemia where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Refractory or significant intolerance or resistance to prior therapy including imatinib (grade 3 or 4 adverse events)
- Second-line indication or third-line indication
*Dasatinib will still be available for chronic phase and accelerated phase CML.
Ibrutinib
For the treatment of relapsed/refractory CLL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed CLL
- Must have received at least 1 prior therapy for CLL
- Considered not appropriate for treatment or retreatment with purine-analogue-based therapy due to:
- Failure to respond to chemo-immunotherapy or
- A progression-free interval of less than 3 years or
- Age of 70 years or more or
- Age of 65 years or more plus the presence of comorbidities or
- A 17p or TP53 deletion
- ECOG performance status of 0-2
- A neutrophil count of ≥0.75 x 10⁹/L
- A platelet count of ≥30 x 10⁹/L
- Patient not on warfarin or CYP3A4/5 inhibitors
- No prior treatment with idelalisib
For the treatment of relapsed/refractory MCL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed MCL with cyclin D1 overexpression or translocation breakpoints at t(11;14)
- Failure to achieve at least partial response with, or documented disease progression disease after, the most recent treatment regimen
- ECOG performance status of 0-2
- At least 1 but no more than 5 previous lines of treatment
Lenalidomide
For the second-line treatment of multiple myeloma (MM) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MM
- Second-line indication
- Contraindication to bortezomib or previously received bortezomib in the first-line setting
*Lenalidomide will still be available for patients with myelodysplastic syndromes with 5q deletion.
Pomalidomide
For the treatment of relapsed and refractory MM where the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically
- MM
- Performance status of 0-2
- Previously received treatment with adequate trials of at least all of the following options of therapy: bortezomib, lenalidomide, and alkylating agents
- Failed treatment with bortezomib or lenalidomide, as defined by: progression on or before 60 days of treatment, progressive disease 6 months or less after achieving a partial response, or intolerance to bortezomib
- Refractory disease to previous treatment
- No resistance to high-dose dexamethasone used in the last line of therapy
- No peripheral neuropathy of grade 2 or more
A complete list of proposed changes to the CDF, as well as the drugs that were de-listed on March 12, 2015, is available on the NHS website. ![]()

Photo courtesy of CDC
England’s National Health Service (NHS) plans to remove several drugs used to treat hematologic malignancies from the Cancer Drugs Fund (CDF).
The plan is that, as of November 4, 2015, pomalidomide, lenalidomide, ibrutinib, dasatinib, brentuximab, bosutinib, and bendamustine will no longer be funded via the CDF for certain indications.
Ofatumumab was removed from the CDF list yesterday but is now available through the NHS.
Drugs used to treat solid tumor malignancies are set to be de-funded through CDF in November as well.
However, the NHS said the proposal to remove a drug from the CDF is not necessarily a final decision.
In cases where a drug offers enough clinical benefit, the pharmaceutical company developing that drug has the opportunity to reduce the price they are asking the NHS to pay to ensure that it achieves a satisfactory level of value for money. The NHS said a number of such negotiations are underway.
In addition, patients who are currently receiving the drugs set to be removed from the CDF will continue to have access to those drugs.
About the CDF and the NHS
The CDF—set up in 2010 and currently due to run until March 2016—is money the government has set aside to pay for cancer drugs that haven’t been approved by the National Institute for Health and Care Excellence (NICE) and aren’t available within the NHS in England. Most cancer drugs are routinely funded outside of the CDF.
NHS England and NICE are planning to consult on a proposed new system for commissioning cancer drugs. The NHS said the new system will be designed to provide the agency with a more systematic approach to getting the best price for cancer drugs.
Reason for drug removals
The NHS previously increased the budget for the CDF from £200 million in 2013/14, to £280 million in 2014/15, and £340 million from April 2015. This represents a total increase of 70% since August 2014.
However, current projections suggest that spending would rise to around £410 million for this year, an over-spend of £70 million, in the absence of further prioritization. The NHS said this money could be used for other aspects of cancer treatment or NHS services for other patient groups.
Therefore, some drugs are set to be removed from the CDF. The NHS said all decisions on drugs to be maintained in the CDF were based on the advice of clinicians, the best available evidence, and the cost of the treatment.
“There is no escaping the fact that we face a difficult set of choices, but it is our duty to ensure we get maximum value from every penny available on behalf of patients,” said Peter Clark, chair of the CDF.
“We must ensure we invest in those treatments that offer the most benefit, based on rigorous evidence-based clinical analysis and an assessment of the cost of those treatments.”
While de-funding certain drugs will reduce costs, the CDF is not expected to be back on budget this financial year. The NHS does expect the CDF will be operating within its budget during 2016/17.
Blood cancer drugs to be removed
The following drugs are currently on the CDF list for the following indications, but they are set to be de-listed on November 4, 2015.
Bendamustine
For the treatment of chronic lymphocytic leukemia (CLL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- CLL (not licensed in this indication)
- Second-line indication, third-line indication, or fourth-line indication
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
For the treatment of relapsed mantle cell lymphoma (MCL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MCL
- Option for second- or subsequent-line chemotherapy
- No previous treatment with bendamustine
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
*Bendamustine will remain on the CDF for other indications.
Bosutinib
For the treatment of refractory, chronic phase chronic myeloid leukemia (CML) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Chronic phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
For the treatment of refractory, accelerated phase CML where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
For the treatment of accelerated phase CML where there is intolerance of treatments and where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Significant intolerance to dasatinib (grade 3 or 4 adverse events; if dasatinib accessed via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
*Bosutinib will still be available through the CDF for patients with chronic phase CML that is intolerant of other treatments.
Brentuximab
For the treatment of refractory, systemic anaplastic lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory systemic anaplastic large-cell lymphoma
For the treatment of relapsed or refractory CD30+ Hodgkin lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory CD30+ Hodgkin lymphoma
- Following autologous stem cell transplant or following at least 2 prior therapies when autologous stem cell transplant or multi-agent chemotherapy is not an option
Dasatinib
For the treatment of Philadelphia-chromosome-positive (Ph+) acute lymphoblastic leukemia where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Refractory or significant intolerance or resistance to prior therapy including imatinib (grade 3 or 4 adverse events)
- Second-line indication or third-line indication
*Dasatinib will still be available for chronic phase and accelerated phase CML.
Ibrutinib
For the treatment of relapsed/refractory CLL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed CLL
- Must have received at least 1 prior therapy for CLL
- Considered not appropriate for treatment or retreatment with purine-analogue-based therapy due to:
- Failure to respond to chemo-immunotherapy or
- A progression-free interval of less than 3 years or
- Age of 70 years or more or
- Age of 65 years or more plus the presence of comorbidities or
- A 17p or TP53 deletion
- ECOG performance status of 0-2
- A neutrophil count of ≥0.75 x 10⁹/L
- A platelet count of ≥30 x 10⁹/L
- Patient not on warfarin or CYP3A4/5 inhibitors
- No prior treatment with idelalisib
For the treatment of relapsed/refractory MCL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed MCL with cyclin D1 overexpression or translocation breakpoints at t(11;14)
- Failure to achieve at least partial response with, or documented disease progression disease after, the most recent treatment regimen
- ECOG performance status of 0-2
- At least 1 but no more than 5 previous lines of treatment
Lenalidomide
For the second-line treatment of multiple myeloma (MM) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MM
- Second-line indication
- Contraindication to bortezomib or previously received bortezomib in the first-line setting
*Lenalidomide will still be available for patients with myelodysplastic syndromes with 5q deletion.
Pomalidomide
For the treatment of relapsed and refractory MM where the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically
- MM
- Performance status of 0-2
- Previously received treatment with adequate trials of at least all of the following options of therapy: bortezomib, lenalidomide, and alkylating agents
- Failed treatment with bortezomib or lenalidomide, as defined by: progression on or before 60 days of treatment, progressive disease 6 months or less after achieving a partial response, or intolerance to bortezomib
- Refractory disease to previous treatment
- No resistance to high-dose dexamethasone used in the last line of therapy
- No peripheral neuropathy of grade 2 or more
A complete list of proposed changes to the CDF, as well as the drugs that were de-listed on March 12, 2015, is available on the NHS website. ![]()

Photo courtesy of CDC
England’s National Health Service (NHS) plans to remove several drugs used to treat hematologic malignancies from the Cancer Drugs Fund (CDF).
The plan is that, as of November 4, 2015, pomalidomide, lenalidomide, ibrutinib, dasatinib, brentuximab, bosutinib, and bendamustine will no longer be funded via the CDF for certain indications.
Ofatumumab was removed from the CDF list yesterday but is now available through the NHS.
Drugs used to treat solid tumor malignancies are set to be de-funded through CDF in November as well.
However, the NHS said the proposal to remove a drug from the CDF is not necessarily a final decision.
In cases where a drug offers enough clinical benefit, the pharmaceutical company developing that drug has the opportunity to reduce the price they are asking the NHS to pay to ensure that it achieves a satisfactory level of value for money. The NHS said a number of such negotiations are underway.
In addition, patients who are currently receiving the drugs set to be removed from the CDF will continue to have access to those drugs.
About the CDF and the NHS
The CDF—set up in 2010 and currently due to run until March 2016—is money the government has set aside to pay for cancer drugs that haven’t been approved by the National Institute for Health and Care Excellence (NICE) and aren’t available within the NHS in England. Most cancer drugs are routinely funded outside of the CDF.
NHS England and NICE are planning to consult on a proposed new system for commissioning cancer drugs. The NHS said the new system will be designed to provide the agency with a more systematic approach to getting the best price for cancer drugs.
Reason for drug removals
The NHS previously increased the budget for the CDF from £200 million in 2013/14, to £280 million in 2014/15, and £340 million from April 2015. This represents a total increase of 70% since August 2014.
However, current projections suggest that spending would rise to around £410 million for this year, an over-spend of £70 million, in the absence of further prioritization. The NHS said this money could be used for other aspects of cancer treatment or NHS services for other patient groups.
Therefore, some drugs are set to be removed from the CDF. The NHS said all decisions on drugs to be maintained in the CDF were based on the advice of clinicians, the best available evidence, and the cost of the treatment.
“There is no escaping the fact that we face a difficult set of choices, but it is our duty to ensure we get maximum value from every penny available on behalf of patients,” said Peter Clark, chair of the CDF.
“We must ensure we invest in those treatments that offer the most benefit, based on rigorous evidence-based clinical analysis and an assessment of the cost of those treatments.”
While de-funding certain drugs will reduce costs, the CDF is not expected to be back on budget this financial year. The NHS does expect the CDF will be operating within its budget during 2016/17.
Blood cancer drugs to be removed
The following drugs are currently on the CDF list for the following indications, but they are set to be de-listed on November 4, 2015.
Bendamustine
For the treatment of chronic lymphocytic leukemia (CLL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- CLL (not licensed in this indication)
- Second-line indication, third-line indication, or fourth-line indication
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
For the treatment of relapsed mantle cell lymphoma (MCL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MCL
- Option for second- or subsequent-line chemotherapy
- No previous treatment with bendamustine
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
*Bendamustine will remain on the CDF for other indications.
Bosutinib
For the treatment of refractory, chronic phase chronic myeloid leukemia (CML) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Chronic phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
For the treatment of refractory, accelerated phase CML where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
For the treatment of accelerated phase CML where there is intolerance of treatments and where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Significant intolerance to dasatinib (grade 3 or 4 adverse events; if dasatinib accessed via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
*Bosutinib will still be available through the CDF for patients with chronic phase CML that is intolerant of other treatments.
Brentuximab
For the treatment of refractory, systemic anaplastic lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory systemic anaplastic large-cell lymphoma
For the treatment of relapsed or refractory CD30+ Hodgkin lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory CD30+ Hodgkin lymphoma
- Following autologous stem cell transplant or following at least 2 prior therapies when autologous stem cell transplant or multi-agent chemotherapy is not an option
Dasatinib
For the treatment of Philadelphia-chromosome-positive (Ph+) acute lymphoblastic leukemia where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Refractory or significant intolerance or resistance to prior therapy including imatinib (grade 3 or 4 adverse events)
- Second-line indication or third-line indication
*Dasatinib will still be available for chronic phase and accelerated phase CML.
Ibrutinib
For the treatment of relapsed/refractory CLL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed CLL
- Must have received at least 1 prior therapy for CLL
- Considered not appropriate for treatment or retreatment with purine-analogue-based therapy due to:
- Failure to respond to chemo-immunotherapy or
- A progression-free interval of less than 3 years or
- Age of 70 years or more or
- Age of 65 years or more plus the presence of comorbidities or
- A 17p or TP53 deletion
- ECOG performance status of 0-2
- A neutrophil count of ≥0.75 x 10⁹/L
- A platelet count of ≥30 x 10⁹/L
- Patient not on warfarin or CYP3A4/5 inhibitors
- No prior treatment with idelalisib
For the treatment of relapsed/refractory MCL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed MCL with cyclin D1 overexpression or translocation breakpoints at t(11;14)
- Failure to achieve at least partial response with, or documented disease progression disease after, the most recent treatment regimen
- ECOG performance status of 0-2
- At least 1 but no more than 5 previous lines of treatment
Lenalidomide
For the second-line treatment of multiple myeloma (MM) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MM
- Second-line indication
- Contraindication to bortezomib or previously received bortezomib in the first-line setting
*Lenalidomide will still be available for patients with myelodysplastic syndromes with 5q deletion.
Pomalidomide
For the treatment of relapsed and refractory MM where the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically
- MM
- Performance status of 0-2
- Previously received treatment with adequate trials of at least all of the following options of therapy: bortezomib, lenalidomide, and alkylating agents
- Failed treatment with bortezomib or lenalidomide, as defined by: progression on or before 60 days of treatment, progressive disease 6 months or less after achieving a partial response, or intolerance to bortezomib
- Refractory disease to previous treatment
- No resistance to high-dose dexamethasone used in the last line of therapy
- No peripheral neuropathy of grade 2 or more
A complete list of proposed changes to the CDF, as well as the drugs that were de-listed on March 12, 2015, is available on the NHS website. ![]()
FDA updates warning about Treanda

Photo by Rhoda Baer
Last March, the US Food and Drug Administration (FDA) issued a statement warning healthcare professionals not to use the chemotherapy drug Treanda Injection (bendamustine hydrochloride) with closed system transfer devices (CSTDs), adapters, and syringes containing polycarbonate or acrylonitrile-butadiene-styrene (ABS).
Now, the FDA is providing a list of devices that were tested and deemed compatible with the drug (see the tables below).
The devices were tested by Treanda’s manufacturer, Teva Pharmaceuticals.
Treanda is used to treat patients with chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma that has progressed during or within 6 months of treatment with rituximab or a rituximab-containing regimen.
Treanda is available in 2 formulations: a solution, Treanda Injection (45 mg/0.5 mL or 180 mg/2 mL solution), and a lyophilized powder, Treanda for Injection (25 mg/vial or 100 mg/vial lyophilized powder). The information discussed here is referring to compatibility with the solution, Treanda Injection.
Treanda Injection contains N, N-dimethylacetamide (DMA), which is incompatible with devices that contain polycarbonate or ABS. Devices including CSTDs, adapters, and syringes that contain polycarbonate or ABS have been shown to dissolve when they come in contact with DMA in the drug.
This incompatibility leads to device failure, such as leaking, breaking, or operational failure of CSTD components; possible product contamination; and potential serious adverse health consequences to practitioners, such as skin reactions, or to patients, including the risk of small blood vessel blockage if the product is contaminated with dissolved ABS or polycarbonate.
Users should contact device manufacturers prior to using the specific devices listed below to ensure there have been no changes made to the material composition of the devices and that the devices are compatible with Treanda use.
Table 1. The compatibility of Treanda Injection with specific CSTDs, syringes, vial adapters, and gloves (based on testing conducted by Teva from February 2015 through June 2015).
| Component tested | Component brand name (part number) |
| Closed system transfer devices (CSTDs) | BD Phaseal System consisting of:
BD Phaseal Protector P14 (REF 515100), BD Phaseal Injector Luer Lock N35 (REF 515003), BD Phaseal Infusion Adapter C100 (REF 515306), BD syringe 5 mL (REF 309646 and 309657) |
| Vial adapters | Baxter CHEMO-AIDE Dispensing Pin (REF 2N9106)
Medimop Swabable Vial Adapter (REF 8070101) Alaris Smartsite (REF 2202E and 2203E) |
| Polypropylene syringes | BD (Becton Dickinson), 5 mL (REF 309646) and 3 mL (REF 309657)
Covidien Monoject, 5 mL (REF 1180600777) and 3 mL (REF 1180300777) B. Braun, 5 mL (REF 4617053V-02) and 3 mL (REF 4610303-02) Air-Tite Norm Jet, 5 mL (REF 4050.X00V0) and 3 mL (REF 4020.X00V0) Medline, 5 mL (REF SYR105010) and 3 mL (REF SYR103010) Terumo, 5 mL (REF SS-05L) |
| Disposable gloves* | ChemoPlus (REF CT0194-1)
EP-Blue (REF 181350) Jackson Safety G29 (REF 49824) NeoPro (REF NPG-888) NitriDerm (REF 182350) Purple (REF 50604) Purple KC 500 (REF 55084) UltraSense EC (REF USE-880) |
*Part numbers reflect a specific size glove used in the compatibility tests.
Table 2. The IV administration set found to be compatible with Treanda Injection after dilution in a 500 mL 0.9% sodium chloride IV infusion bags (based on testing conducted by Teva from February 2015 through June 2015*).
| Component tested | Brand name (part number) |
| IV administration sets | B. Braun Safeline (REF NF3482) and AdditIV (REF V1921)
Baxter DuoVent Spike (REF 2C7575) and Clearlink System (2H8480) BD Phaseal Secondary set (REF 515301) ICU Medical Clave (REF CH3011) |
*Compatibility studies did not include testing with 2.5% dextrose/0.45% sodium chloride injection. However, the results of these studies are not expected to change. So either diluent, 0.9% sodium chloride or 2.5% dextrose/0.45% sodium chloride injection, can be used with Treanda injection.
The FDA required label changes for both the solution and the powder formulations of Treanda to include information for safe preparation and handling for IV administration. See the full prescribing information for details.
For more details on the compatibility of Treanda Injection with specific CSTDs, syringes, vial adapters, gloves, and IV administration sets, see Teva’s Dear Health Care Provider letter.
Adverse events or quality problems associated with the use of Treanda products can be reported to the FDA’s MedWatch Adverse Event Reporting Program. ![]()

Photo by Rhoda Baer
Last March, the US Food and Drug Administration (FDA) issued a statement warning healthcare professionals not to use the chemotherapy drug Treanda Injection (bendamustine hydrochloride) with closed system transfer devices (CSTDs), adapters, and syringes containing polycarbonate or acrylonitrile-butadiene-styrene (ABS).
Now, the FDA is providing a list of devices that were tested and deemed compatible with the drug (see the tables below).
The devices were tested by Treanda’s manufacturer, Teva Pharmaceuticals.
Treanda is used to treat patients with chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma that has progressed during or within 6 months of treatment with rituximab or a rituximab-containing regimen.
Treanda is available in 2 formulations: a solution, Treanda Injection (45 mg/0.5 mL or 180 mg/2 mL solution), and a lyophilized powder, Treanda for Injection (25 mg/vial or 100 mg/vial lyophilized powder). The information discussed here is referring to compatibility with the solution, Treanda Injection.
Treanda Injection contains N, N-dimethylacetamide (DMA), which is incompatible with devices that contain polycarbonate or ABS. Devices including CSTDs, adapters, and syringes that contain polycarbonate or ABS have been shown to dissolve when they come in contact with DMA in the drug.
This incompatibility leads to device failure, such as leaking, breaking, or operational failure of CSTD components; possible product contamination; and potential serious adverse health consequences to practitioners, such as skin reactions, or to patients, including the risk of small blood vessel blockage if the product is contaminated with dissolved ABS or polycarbonate.
Users should contact device manufacturers prior to using the specific devices listed below to ensure there have been no changes made to the material composition of the devices and that the devices are compatible with Treanda use.
Table 1. The compatibility of Treanda Injection with specific CSTDs, syringes, vial adapters, and gloves (based on testing conducted by Teva from February 2015 through June 2015).
| Component tested | Component brand name (part number) |
| Closed system transfer devices (CSTDs) | BD Phaseal System consisting of:
BD Phaseal Protector P14 (REF 515100), BD Phaseal Injector Luer Lock N35 (REF 515003), BD Phaseal Infusion Adapter C100 (REF 515306), BD syringe 5 mL (REF 309646 and 309657) |
| Vial adapters | Baxter CHEMO-AIDE Dispensing Pin (REF 2N9106)
Medimop Swabable Vial Adapter (REF 8070101) Alaris Smartsite (REF 2202E and 2203E) |
| Polypropylene syringes | BD (Becton Dickinson), 5 mL (REF 309646) and 3 mL (REF 309657)
Covidien Monoject, 5 mL (REF 1180600777) and 3 mL (REF 1180300777) B. Braun, 5 mL (REF 4617053V-02) and 3 mL (REF 4610303-02) Air-Tite Norm Jet, 5 mL (REF 4050.X00V0) and 3 mL (REF 4020.X00V0) Medline, 5 mL (REF SYR105010) and 3 mL (REF SYR103010) Terumo, 5 mL (REF SS-05L) |
| Disposable gloves* | ChemoPlus (REF CT0194-1)
EP-Blue (REF 181350) Jackson Safety G29 (REF 49824) NeoPro (REF NPG-888) NitriDerm (REF 182350) Purple (REF 50604) Purple KC 500 (REF 55084) UltraSense EC (REF USE-880) |
*Part numbers reflect a specific size glove used in the compatibility tests.
Table 2. The IV administration set found to be compatible with Treanda Injection after dilution in a 500 mL 0.9% sodium chloride IV infusion bags (based on testing conducted by Teva from February 2015 through June 2015*).
| Component tested | Brand name (part number) |
| IV administration sets | B. Braun Safeline (REF NF3482) and AdditIV (REF V1921)
Baxter DuoVent Spike (REF 2C7575) and Clearlink System (2H8480) BD Phaseal Secondary set (REF 515301) ICU Medical Clave (REF CH3011) |
*Compatibility studies did not include testing with 2.5% dextrose/0.45% sodium chloride injection. However, the results of these studies are not expected to change. So either diluent, 0.9% sodium chloride or 2.5% dextrose/0.45% sodium chloride injection, can be used with Treanda injection.
The FDA required label changes for both the solution and the powder formulations of Treanda to include information for safe preparation and handling for IV administration. See the full prescribing information for details.
For more details on the compatibility of Treanda Injection with specific CSTDs, syringes, vial adapters, gloves, and IV administration sets, see Teva’s Dear Health Care Provider letter.
Adverse events or quality problems associated with the use of Treanda products can be reported to the FDA’s MedWatch Adverse Event Reporting Program. ![]()

Photo by Rhoda Baer
Last March, the US Food and Drug Administration (FDA) issued a statement warning healthcare professionals not to use the chemotherapy drug Treanda Injection (bendamustine hydrochloride) with closed system transfer devices (CSTDs), adapters, and syringes containing polycarbonate or acrylonitrile-butadiene-styrene (ABS).
Now, the FDA is providing a list of devices that were tested and deemed compatible with the drug (see the tables below).
The devices were tested by Treanda’s manufacturer, Teva Pharmaceuticals.
Treanda is used to treat patients with chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma that has progressed during or within 6 months of treatment with rituximab or a rituximab-containing regimen.
Treanda is available in 2 formulations: a solution, Treanda Injection (45 mg/0.5 mL or 180 mg/2 mL solution), and a lyophilized powder, Treanda for Injection (25 mg/vial or 100 mg/vial lyophilized powder). The information discussed here is referring to compatibility with the solution, Treanda Injection.
Treanda Injection contains N, N-dimethylacetamide (DMA), which is incompatible with devices that contain polycarbonate or ABS. Devices including CSTDs, adapters, and syringes that contain polycarbonate or ABS have been shown to dissolve when they come in contact with DMA in the drug.
This incompatibility leads to device failure, such as leaking, breaking, or operational failure of CSTD components; possible product contamination; and potential serious adverse health consequences to practitioners, such as skin reactions, or to patients, including the risk of small blood vessel blockage if the product is contaminated with dissolved ABS or polycarbonate.
Users should contact device manufacturers prior to using the specific devices listed below to ensure there have been no changes made to the material composition of the devices and that the devices are compatible with Treanda use.
Table 1. The compatibility of Treanda Injection with specific CSTDs, syringes, vial adapters, and gloves (based on testing conducted by Teva from February 2015 through June 2015).
| Component tested | Component brand name (part number) |
| Closed system transfer devices (CSTDs) | BD Phaseal System consisting of:
BD Phaseal Protector P14 (REF 515100), BD Phaseal Injector Luer Lock N35 (REF 515003), BD Phaseal Infusion Adapter C100 (REF 515306), BD syringe 5 mL (REF 309646 and 309657) |
| Vial adapters | Baxter CHEMO-AIDE Dispensing Pin (REF 2N9106)
Medimop Swabable Vial Adapter (REF 8070101) Alaris Smartsite (REF 2202E and 2203E) |
| Polypropylene syringes | BD (Becton Dickinson), 5 mL (REF 309646) and 3 mL (REF 309657)
Covidien Monoject, 5 mL (REF 1180600777) and 3 mL (REF 1180300777) B. Braun, 5 mL (REF 4617053V-02) and 3 mL (REF 4610303-02) Air-Tite Norm Jet, 5 mL (REF 4050.X00V0) and 3 mL (REF 4020.X00V0) Medline, 5 mL (REF SYR105010) and 3 mL (REF SYR103010) Terumo, 5 mL (REF SS-05L) |
| Disposable gloves* | ChemoPlus (REF CT0194-1)
EP-Blue (REF 181350) Jackson Safety G29 (REF 49824) NeoPro (REF NPG-888) NitriDerm (REF 182350) Purple (REF 50604) Purple KC 500 (REF 55084) UltraSense EC (REF USE-880) |
*Part numbers reflect a specific size glove used in the compatibility tests.
Table 2. The IV administration set found to be compatible with Treanda Injection after dilution in a 500 mL 0.9% sodium chloride IV infusion bags (based on testing conducted by Teva from February 2015 through June 2015*).
| Component tested | Brand name (part number) |
| IV administration sets | B. Braun Safeline (REF NF3482) and AdditIV (REF V1921)
Baxter DuoVent Spike (REF 2C7575) and Clearlink System (2H8480) BD Phaseal Secondary set (REF 515301) ICU Medical Clave (REF CH3011) |
*Compatibility studies did not include testing with 2.5% dextrose/0.45% sodium chloride injection. However, the results of these studies are not expected to change. So either diluent, 0.9% sodium chloride or 2.5% dextrose/0.45% sodium chloride injection, can be used with Treanda injection.
The FDA required label changes for both the solution and the powder formulations of Treanda to include information for safe preparation and handling for IV administration. See the full prescribing information for details.
For more details on the compatibility of Treanda Injection with specific CSTDs, syringes, vial adapters, gloves, and IV administration sets, see Teva’s Dear Health Care Provider letter.
Adverse events or quality problems associated with the use of Treanda products can be reported to the FDA’s MedWatch Adverse Event Reporting Program. ![]()
First biosimilar launched in US
© Sandoz Inc. 2015
The leukocyte growth factor Zarxio (filgrastim-sndz), the first biosimilar product to gain approval from the US Food and Drug Administration (FDA), is now available in the US.
Zarxio was approved by the FDA on March 6. The product, made by Sandoz, Inc., is biosimilar to Amgen Inc.’s Neupogen, which was originally licensed in 1991.
Zarxio is marketed as Zarzio outside the US. The biosimilar is available in more than 60 countries worldwide.
In the US, Zarxio is approved for the same indications as Neupogen. So Zarxio can be prescribed for the following 5 indications.
Patients with cancer receiving myelosuppressive chemotherapy: to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with nonmyeloid malignancies receiving myelosuppressive anticancer drugs associated with a significant incidence of severe neutropenia with fever.
Patients with acute myeloid leukemia receiving induction or consolidation chemotherapy: to reduce the time to neutrophil recovery and the duration of fever, following induction or consolidation chemotherapy.
Patients with cancer undergoing bone marrow transplant: to reduce the duration of neutropenia and neutropenia-related clinical sequelae—eg, febrile neutropenia—in patients with nonmyeloid malignancies undergoing myeloablative chemotherapy followed by bone marrow transplant.
Patients undergoing autologous peripheral blood progenitor cell collection and therapy: for the mobilization of autologous hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis.
Patients with severe chronic neutropenia: for chronic administration to reduce the incidence and duration of sequelae of neutropenia—eg, fever, infections, oropharyngeal ulcers—in symptomatic patients with congenital neutropenia, cyclic neutropenia, or idiopathic neutropenia.
PIONEER trial
The FDA’s approval of Zarxio was based on data showing that Zarxio is highly similar to Neupogen, with no clinically meaningful differences between the products.
The head-to-head PIONEER study was the final piece of evidence the FDA used to approve Zarxio as biosimilar to Neupogen. Results of the trial were presented at ASH 2014.
Zarxio and Neupogen both produced the expected reduction in the duration of severe neutropenia in breast cancer patients undergoing myelosuppressive chemotherapy—1.17 ± 1.11 and 1.20 ±1.02 days, respectively.
The mean time to absolute neutrophil count recovery in cycle 1 was also similar—1.8 ± 0.97 days in the Zarxio arm and 1.7 ± 0.81 days in the Neupogen arm. No immunogenicity or antibodies against rhG-CSF were detected throughout the study.
The researchers said there were no obvious differences between Zarxio and Neupogen with regard to treatment-emergent adverse events.
The most common side effects observed with Zarxio are aching bones/muscles and redness, swelling, or itching at the injection site. Serious side effects may include spleen rupture; serious allergic reactions that may cause rash, shortness of breath, wheezing and/or swelling around the mouth and eyes; fast pulse and sweating; and acute respiratory distress syndrome.
For more details on Zarxio, see the full prescribing information or visit www.zarxio.com. ![]()

© Sandoz Inc. 2015
The leukocyte growth factor Zarxio (filgrastim-sndz), the first biosimilar product to gain approval from the US Food and Drug Administration (FDA), is now available in the US.
Zarxio was approved by the FDA on March 6. The product, made by Sandoz, Inc., is biosimilar to Amgen Inc.’s Neupogen, which was originally licensed in 1991.
Zarxio is marketed as Zarzio outside the US. The biosimilar is available in more than 60 countries worldwide.
In the US, Zarxio is approved for the same indications as Neupogen. So Zarxio can be prescribed for the following 5 indications.
Patients with cancer receiving myelosuppressive chemotherapy: to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with nonmyeloid malignancies receiving myelosuppressive anticancer drugs associated with a significant incidence of severe neutropenia with fever.
Patients with acute myeloid leukemia receiving induction or consolidation chemotherapy: to reduce the time to neutrophil recovery and the duration of fever, following induction or consolidation chemotherapy.
Patients with cancer undergoing bone marrow transplant: to reduce the duration of neutropenia and neutropenia-related clinical sequelae—eg, febrile neutropenia—in patients with nonmyeloid malignancies undergoing myeloablative chemotherapy followed by bone marrow transplant.
Patients undergoing autologous peripheral blood progenitor cell collection and therapy: for the mobilization of autologous hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis.
Patients with severe chronic neutropenia: for chronic administration to reduce the incidence and duration of sequelae of neutropenia—eg, fever, infections, oropharyngeal ulcers—in symptomatic patients with congenital neutropenia, cyclic neutropenia, or idiopathic neutropenia.
PIONEER trial
The FDA’s approval of Zarxio was based on data showing that Zarxio is highly similar to Neupogen, with no clinically meaningful differences between the products.
The head-to-head PIONEER study was the final piece of evidence the FDA used to approve Zarxio as biosimilar to Neupogen. Results of the trial were presented at ASH 2014.
Zarxio and Neupogen both produced the expected reduction in the duration of severe neutropenia in breast cancer patients undergoing myelosuppressive chemotherapy—1.17 ± 1.11 and 1.20 ±1.02 days, respectively.
The mean time to absolute neutrophil count recovery in cycle 1 was also similar—1.8 ± 0.97 days in the Zarxio arm and 1.7 ± 0.81 days in the Neupogen arm. No immunogenicity or antibodies against rhG-CSF were detected throughout the study.
The researchers said there were no obvious differences between Zarxio and Neupogen with regard to treatment-emergent adverse events.
The most common side effects observed with Zarxio are aching bones/muscles and redness, swelling, or itching at the injection site. Serious side effects may include spleen rupture; serious allergic reactions that may cause rash, shortness of breath, wheezing and/or swelling around the mouth and eyes; fast pulse and sweating; and acute respiratory distress syndrome.
For more details on Zarxio, see the full prescribing information or visit www.zarxio.com. ![]()

© Sandoz Inc. 2015
The leukocyte growth factor Zarxio (filgrastim-sndz), the first biosimilar product to gain approval from the US Food and Drug Administration (FDA), is now available in the US.
Zarxio was approved by the FDA on March 6. The product, made by Sandoz, Inc., is biosimilar to Amgen Inc.’s Neupogen, which was originally licensed in 1991.
Zarxio is marketed as Zarzio outside the US. The biosimilar is available in more than 60 countries worldwide.
In the US, Zarxio is approved for the same indications as Neupogen. So Zarxio can be prescribed for the following 5 indications.
Patients with cancer receiving myelosuppressive chemotherapy: to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with nonmyeloid malignancies receiving myelosuppressive anticancer drugs associated with a significant incidence of severe neutropenia with fever.
Patients with acute myeloid leukemia receiving induction or consolidation chemotherapy: to reduce the time to neutrophil recovery and the duration of fever, following induction or consolidation chemotherapy.
Patients with cancer undergoing bone marrow transplant: to reduce the duration of neutropenia and neutropenia-related clinical sequelae—eg, febrile neutropenia—in patients with nonmyeloid malignancies undergoing myeloablative chemotherapy followed by bone marrow transplant.
Patients undergoing autologous peripheral blood progenitor cell collection and therapy: for the mobilization of autologous hematopoietic progenitor cells into the peripheral blood for collection by leukapheresis.
Patients with severe chronic neutropenia: for chronic administration to reduce the incidence and duration of sequelae of neutropenia—eg, fever, infections, oropharyngeal ulcers—in symptomatic patients with congenital neutropenia, cyclic neutropenia, or idiopathic neutropenia.
PIONEER trial
The FDA’s approval of Zarxio was based on data showing that Zarxio is highly similar to Neupogen, with no clinically meaningful differences between the products.
The head-to-head PIONEER study was the final piece of evidence the FDA used to approve Zarxio as biosimilar to Neupogen. Results of the trial were presented at ASH 2014.
Zarxio and Neupogen both produced the expected reduction in the duration of severe neutropenia in breast cancer patients undergoing myelosuppressive chemotherapy—1.17 ± 1.11 and 1.20 ±1.02 days, respectively.
The mean time to absolute neutrophil count recovery in cycle 1 was also similar—1.8 ± 0.97 days in the Zarxio arm and 1.7 ± 0.81 days in the Neupogen arm. No immunogenicity or antibodies against rhG-CSF were detected throughout the study.
The researchers said there were no obvious differences between Zarxio and Neupogen with regard to treatment-emergent adverse events.
The most common side effects observed with Zarxio are aching bones/muscles and redness, swelling, or itching at the injection site. Serious side effects may include spleen rupture; serious allergic reactions that may cause rash, shortness of breath, wheezing and/or swelling around the mouth and eyes; fast pulse and sweating; and acute respiratory distress syndrome.
For more details on Zarxio, see the full prescribing information or visit www.zarxio.com. ![]()
Obinutuzumab trends better than rituxumab in relapsed indolent lymphoma
Patients with relapsed follicular lymphoma who were treated with obinutuzumab experienced higher response rates than did patients given rituximab with an acceptable safety profile, according to new findings.
However, the difference did not translate into an improvement in progression-free survival, so the clinical value of obinutuzumab in this patient population is still unclear.
The quality of remissions was better with obinutuzumab, with an almost twofold higher complete response/unconfirmed complete response rate (41.9% vs. 22.7%; P = .006),” wrote Dr. Laurie Sehn from the Centre for Lymphoid Cancer, British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues (J Clin Oncol. 2015 Aug 17. doi:10.1200/JCO.2014.59.2139).
On the basis of an independent review, the best overall response was better in the obinutuzumab arm (P = .04), but the complete response/unconfirmed response rate was not different for the two groups.
The study was published online Aug. 17 in the Journal of Clinical Oncology.
A total of 175 patients with relapsed CD20+ indolent lymphoma were randomized 1:1 to four once-per-week infusions of either obinutuzumab (1,000 mg) or rituximab (375 mg/m2). Those without any evidence of disease progression after completing induction therapy received obinutuzumab or rituximab maintenance therapy every 2 months for up to 2 years.
At the end of induction, the investigator assessed overall response rate was 44.6% in the obinutuzumab arm and 33.3% in the rituximab arm (P = .08); nine patients receiving obinutuzumab (12.2%) and four given rituximab (5.3%) achieved complete response or unconfirmed complete response, but the difference was not significant (P = .07).
Independent review also found the overall response rate to be higher with obinutuzumab vs. rituximab (44.6% vs. 26.7%; P = .01), but with no difference in complete response/unconfirmed complete response rate (5.4 vs. 4.0; P = .34).
Adverse events were similar in each group, and most episodes were grade 1 to 2. Higher rates of infusion-related reactions (74% vs. 51%) and cough (24% vs. 9%) were observed in the obinutuzumab vs. the rituximab arm.
Dr. Sehn receives research funding and honoraria from, and serves in a consulting or advisory role to, Roche/Genentech, the maker of obinutuzumab (Gyzyva) and rituximab (Rituxan). She also receives honoraria from and serves in a consulting or advisory role to Amgen, Janssen, Seattle Genetics, Lundbeck, and Celgene.
Patients with relapsed follicular lymphoma who were treated with obinutuzumab experienced higher response rates than did patients given rituximab with an acceptable safety profile, according to new findings.
However, the difference did not translate into an improvement in progression-free survival, so the clinical value of obinutuzumab in this patient population is still unclear.
The quality of remissions was better with obinutuzumab, with an almost twofold higher complete response/unconfirmed complete response rate (41.9% vs. 22.7%; P = .006),” wrote Dr. Laurie Sehn from the Centre for Lymphoid Cancer, British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues (J Clin Oncol. 2015 Aug 17. doi:10.1200/JCO.2014.59.2139).
On the basis of an independent review, the best overall response was better in the obinutuzumab arm (P = .04), but the complete response/unconfirmed response rate was not different for the two groups.
The study was published online Aug. 17 in the Journal of Clinical Oncology.
A total of 175 patients with relapsed CD20+ indolent lymphoma were randomized 1:1 to four once-per-week infusions of either obinutuzumab (1,000 mg) or rituximab (375 mg/m2). Those without any evidence of disease progression after completing induction therapy received obinutuzumab or rituximab maintenance therapy every 2 months for up to 2 years.
At the end of induction, the investigator assessed overall response rate was 44.6% in the obinutuzumab arm and 33.3% in the rituximab arm (P = .08); nine patients receiving obinutuzumab (12.2%) and four given rituximab (5.3%) achieved complete response or unconfirmed complete response, but the difference was not significant (P = .07).
Independent review also found the overall response rate to be higher with obinutuzumab vs. rituximab (44.6% vs. 26.7%; P = .01), but with no difference in complete response/unconfirmed complete response rate (5.4 vs. 4.0; P = .34).
Adverse events were similar in each group, and most episodes were grade 1 to 2. Higher rates of infusion-related reactions (74% vs. 51%) and cough (24% vs. 9%) were observed in the obinutuzumab vs. the rituximab arm.
Dr. Sehn receives research funding and honoraria from, and serves in a consulting or advisory role to, Roche/Genentech, the maker of obinutuzumab (Gyzyva) and rituximab (Rituxan). She also receives honoraria from and serves in a consulting or advisory role to Amgen, Janssen, Seattle Genetics, Lundbeck, and Celgene.
Patients with relapsed follicular lymphoma who were treated with obinutuzumab experienced higher response rates than did patients given rituximab with an acceptable safety profile, according to new findings.
However, the difference did not translate into an improvement in progression-free survival, so the clinical value of obinutuzumab in this patient population is still unclear.
The quality of remissions was better with obinutuzumab, with an almost twofold higher complete response/unconfirmed complete response rate (41.9% vs. 22.7%; P = .006),” wrote Dr. Laurie Sehn from the Centre for Lymphoid Cancer, British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues (J Clin Oncol. 2015 Aug 17. doi:10.1200/JCO.2014.59.2139).
On the basis of an independent review, the best overall response was better in the obinutuzumab arm (P = .04), but the complete response/unconfirmed response rate was not different for the two groups.
The study was published online Aug. 17 in the Journal of Clinical Oncology.
A total of 175 patients with relapsed CD20+ indolent lymphoma were randomized 1:1 to four once-per-week infusions of either obinutuzumab (1,000 mg) or rituximab (375 mg/m2). Those without any evidence of disease progression after completing induction therapy received obinutuzumab or rituximab maintenance therapy every 2 months for up to 2 years.
At the end of induction, the investigator assessed overall response rate was 44.6% in the obinutuzumab arm and 33.3% in the rituximab arm (P = .08); nine patients receiving obinutuzumab (12.2%) and four given rituximab (5.3%) achieved complete response or unconfirmed complete response, but the difference was not significant (P = .07).
Independent review also found the overall response rate to be higher with obinutuzumab vs. rituximab (44.6% vs. 26.7%; P = .01), but with no difference in complete response/unconfirmed complete response rate (5.4 vs. 4.0; P = .34).
Adverse events were similar in each group, and most episodes were grade 1 to 2. Higher rates of infusion-related reactions (74% vs. 51%) and cough (24% vs. 9%) were observed in the obinutuzumab vs. the rituximab arm.
Dr. Sehn receives research funding and honoraria from, and serves in a consulting or advisory role to, Roche/Genentech, the maker of obinutuzumab (Gyzyva) and rituximab (Rituxan). She also receives honoraria from and serves in a consulting or advisory role to Amgen, Janssen, Seattle Genetics, Lundbeck, and Celgene.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Obinutuzumab was associated with a higher overall response rate as compared with rituximab, but obinutuzumab’s clinical benefit in non–Hodgkin lymphoma is still unclear.
Major finding: Among patients with follicular lymphoma (n = 149), overall response rate trended higher for obinutuzumab, compared with rituximab (44.6% vs. 33.3%; P = .08).
Data source: An open-label, multicenter, randomized, phase II study of 175 patients with relapsed CD20+ indolent lymphoma that compared induction with obinutuzumab vs. rituximab.
Disclosures: Dr. Sehn receives research funding and honoraria from, and serves in a consulting or advisory role to Roche/Genentech, the maker of obinutuzumab (Gyzyva) and rituximab (Rituxan). She also receives honoraria from and serves in a consulting or advisory role to Amgen, Janssen, Seattle Genetics, Lundbeck, and Celgene.
Myeloma precursor linked to Agent Orange exposure
Vietnam War veterans exposed to Agent Orange have a twofold higher prevalence of monoclonal gammopathy of undetermined significance (MGUS), compared with control veterans, providing the first scientific evidence for a link between the multiple myeloma precursor and Agent Orange exposure, researchers reported online in JAMA Oncology.
Serum samples from U.S. Air Force personnel who conducted aerial herbicide spray missions of Agent Orange in the Vietnam War from 1962 to 1971 (Operation Ranch Hand) were compared with samples from veterans who served in Vietnam during the same time period but were not involved in herbicide spray missions. The human carcinogen TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin) was a contaminant found in variable amounts in Agent Orange, and levels of TCDD measured in the veteran’s serum samples were associated with cohort status. For example, TCDD levels greater than 10.92 parts per trillion were observed in 47.5% of the Ranch Hand cohort, compared with just 2.5% of the control veteran cohort. The risk of MGUS increased with increasing body burden of TCDD, although the trend was not significant.
MGUS prevalence in the Ranch Hand group was 7.1% (34 of 479) compared with 3.1% (15 of 479) in the control group (adjusted odds ratio, 2.37; 95% confidence interval, 1.27-4.44; P = .007).
“Our findings of increased MGUS risk among Ranch Hand veterans supports an association between Agent Orange exposure and multiple myeloma,” wrote Dr. Ola Landgren, Chief of Myeloma Service at Memorial Sloan Kettering, New York, and his colleagues (JAMA Oncol. 2015 Sep 3; [doi:10.1001/jamaoncol.2015.2938].
Serum samples were collected in 2002 from U.S. Air Force personnel who conducted aerial herbicide spray missions from 1962 to 1971 (n = 479) and control veterans who were not involved in the aerial missions (n = 479). The study was a follow-up of the Air Force Health Study. The first TCDD measurements were made in 1987, up to 25 years after Agent Orange exposure.
Ranch Hand veterans younger than 70 years had a significantly increased MGUS risk (OR, 3.4; 95% CI, 1.46-8.13; P = .004), but those older than 70 years had no increased MGUS risk.
Previous studies have pointed to an elevated risk of multiple myeloma among agricultural workers, and pesticides are thought to be responsible for the association.
The study was supported by the Agency for Toxic Substances and Disease Registry, the National Cancer Institute, and the Air Force Health Study Assets Research Program. Dr. Landgren reported having consulting or advisory roles with Onyx Pharmaceuticals/AMGEN, Celgene, Bristol-Myers Squibb, Jansen, and Millennium Pharmaceuticals/Takeda.
Given that all multiple myeloma cases originate from MGUS, the study by Landgren et al. provides the first scientific evidence for a direct link between multiple myeloma and exposure to Agent Orange. The study was based on well-characterized samples, a long follow-up period (25 years), and measurements of toxin exposure. The results are in line with an Agricultural Health Study that showed an almost twofold higher prevalence of MGUS among male pesticide applicators.
A weakness of this study was the variable time between exposure to Agent Orange and measurement of serum TCDD, which ranged from 16 to 25 years. It is unclear whether the peak level of TCDD exposure or the longer-term persistence of the substance in the body plays a role in MGUS transformation.
The study also highlights the importance of tissue banking to allow modern methods to investigate unanswered questions. Newer technologies can be applied to stored samples to better characterize the occurrence and progression of diseases. An important question that remains is whether TCDD exposure induces MGUS, which in turn requires additional mutations to undergo malignant transformation, or whether TCDD exposure creates the genomic instability that leads myeloma.
Dr. Nikhil Munshi is professor of medicine at the Dana-Farber Cancer Institute, Boston. These remarks were part of an editorial accompanying the report (JAMA Oncol. 2015 Sep 3; [doi:10.1001/jamaoncol.2015.2938]. Dr. Munshi had no disclosures to report.
Given that all multiple myeloma cases originate from MGUS, the study by Landgren et al. provides the first scientific evidence for a direct link between multiple myeloma and exposure to Agent Orange. The study was based on well-characterized samples, a long follow-up period (25 years), and measurements of toxin exposure. The results are in line with an Agricultural Health Study that showed an almost twofold higher prevalence of MGUS among male pesticide applicators.
A weakness of this study was the variable time between exposure to Agent Orange and measurement of serum TCDD, which ranged from 16 to 25 years. It is unclear whether the peak level of TCDD exposure or the longer-term persistence of the substance in the body plays a role in MGUS transformation.
The study also highlights the importance of tissue banking to allow modern methods to investigate unanswered questions. Newer technologies can be applied to stored samples to better characterize the occurrence and progression of diseases. An important question that remains is whether TCDD exposure induces MGUS, which in turn requires additional mutations to undergo malignant transformation, or whether TCDD exposure creates the genomic instability that leads myeloma.
Dr. Nikhil Munshi is professor of medicine at the Dana-Farber Cancer Institute, Boston. These remarks were part of an editorial accompanying the report (JAMA Oncol. 2015 Sep 3; [doi:10.1001/jamaoncol.2015.2938]. Dr. Munshi had no disclosures to report.
Given that all multiple myeloma cases originate from MGUS, the study by Landgren et al. provides the first scientific evidence for a direct link between multiple myeloma and exposure to Agent Orange. The study was based on well-characterized samples, a long follow-up period (25 years), and measurements of toxin exposure. The results are in line with an Agricultural Health Study that showed an almost twofold higher prevalence of MGUS among male pesticide applicators.
A weakness of this study was the variable time between exposure to Agent Orange and measurement of serum TCDD, which ranged from 16 to 25 years. It is unclear whether the peak level of TCDD exposure or the longer-term persistence of the substance in the body plays a role in MGUS transformation.
The study also highlights the importance of tissue banking to allow modern methods to investigate unanswered questions. Newer technologies can be applied to stored samples to better characterize the occurrence and progression of diseases. An important question that remains is whether TCDD exposure induces MGUS, which in turn requires additional mutations to undergo malignant transformation, or whether TCDD exposure creates the genomic instability that leads myeloma.
Dr. Nikhil Munshi is professor of medicine at the Dana-Farber Cancer Institute, Boston. These remarks were part of an editorial accompanying the report (JAMA Oncol. 2015 Sep 3; [doi:10.1001/jamaoncol.2015.2938]. Dr. Munshi had no disclosures to report.
Vietnam War veterans exposed to Agent Orange have a twofold higher prevalence of monoclonal gammopathy of undetermined significance (MGUS), compared with control veterans, providing the first scientific evidence for a link between the multiple myeloma precursor and Agent Orange exposure, researchers reported online in JAMA Oncology.
Serum samples from U.S. Air Force personnel who conducted aerial herbicide spray missions of Agent Orange in the Vietnam War from 1962 to 1971 (Operation Ranch Hand) were compared with samples from veterans who served in Vietnam during the same time period but were not involved in herbicide spray missions. The human carcinogen TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin) was a contaminant found in variable amounts in Agent Orange, and levels of TCDD measured in the veteran’s serum samples were associated with cohort status. For example, TCDD levels greater than 10.92 parts per trillion were observed in 47.5% of the Ranch Hand cohort, compared with just 2.5% of the control veteran cohort. The risk of MGUS increased with increasing body burden of TCDD, although the trend was not significant.
MGUS prevalence in the Ranch Hand group was 7.1% (34 of 479) compared with 3.1% (15 of 479) in the control group (adjusted odds ratio, 2.37; 95% confidence interval, 1.27-4.44; P = .007).
“Our findings of increased MGUS risk among Ranch Hand veterans supports an association between Agent Orange exposure and multiple myeloma,” wrote Dr. Ola Landgren, Chief of Myeloma Service at Memorial Sloan Kettering, New York, and his colleagues (JAMA Oncol. 2015 Sep 3; [doi:10.1001/jamaoncol.2015.2938].
Serum samples were collected in 2002 from U.S. Air Force personnel who conducted aerial herbicide spray missions from 1962 to 1971 (n = 479) and control veterans who were not involved in the aerial missions (n = 479). The study was a follow-up of the Air Force Health Study. The first TCDD measurements were made in 1987, up to 25 years after Agent Orange exposure.
Ranch Hand veterans younger than 70 years had a significantly increased MGUS risk (OR, 3.4; 95% CI, 1.46-8.13; P = .004), but those older than 70 years had no increased MGUS risk.
Previous studies have pointed to an elevated risk of multiple myeloma among agricultural workers, and pesticides are thought to be responsible for the association.
The study was supported by the Agency for Toxic Substances and Disease Registry, the National Cancer Institute, and the Air Force Health Study Assets Research Program. Dr. Landgren reported having consulting or advisory roles with Onyx Pharmaceuticals/AMGEN, Celgene, Bristol-Myers Squibb, Jansen, and Millennium Pharmaceuticals/Takeda.
Vietnam War veterans exposed to Agent Orange have a twofold higher prevalence of monoclonal gammopathy of undetermined significance (MGUS), compared with control veterans, providing the first scientific evidence for a link between the multiple myeloma precursor and Agent Orange exposure, researchers reported online in JAMA Oncology.
Serum samples from U.S. Air Force personnel who conducted aerial herbicide spray missions of Agent Orange in the Vietnam War from 1962 to 1971 (Operation Ranch Hand) were compared with samples from veterans who served in Vietnam during the same time period but were not involved in herbicide spray missions. The human carcinogen TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin) was a contaminant found in variable amounts in Agent Orange, and levels of TCDD measured in the veteran’s serum samples were associated with cohort status. For example, TCDD levels greater than 10.92 parts per trillion were observed in 47.5% of the Ranch Hand cohort, compared with just 2.5% of the control veteran cohort. The risk of MGUS increased with increasing body burden of TCDD, although the trend was not significant.
MGUS prevalence in the Ranch Hand group was 7.1% (34 of 479) compared with 3.1% (15 of 479) in the control group (adjusted odds ratio, 2.37; 95% confidence interval, 1.27-4.44; P = .007).
“Our findings of increased MGUS risk among Ranch Hand veterans supports an association between Agent Orange exposure and multiple myeloma,” wrote Dr. Ola Landgren, Chief of Myeloma Service at Memorial Sloan Kettering, New York, and his colleagues (JAMA Oncol. 2015 Sep 3; [doi:10.1001/jamaoncol.2015.2938].
Serum samples were collected in 2002 from U.S. Air Force personnel who conducted aerial herbicide spray missions from 1962 to 1971 (n = 479) and control veterans who were not involved in the aerial missions (n = 479). The study was a follow-up of the Air Force Health Study. The first TCDD measurements were made in 1987, up to 25 years after Agent Orange exposure.
Ranch Hand veterans younger than 70 years had a significantly increased MGUS risk (OR, 3.4; 95% CI, 1.46-8.13; P = .004), but those older than 70 years had no increased MGUS risk.
Previous studies have pointed to an elevated risk of multiple myeloma among agricultural workers, and pesticides are thought to be responsible for the association.
The study was supported by the Agency for Toxic Substances and Disease Registry, the National Cancer Institute, and the Air Force Health Study Assets Research Program. Dr. Landgren reported having consulting or advisory roles with Onyx Pharmaceuticals/AMGEN, Celgene, Bristol-Myers Squibb, Jansen, and Millennium Pharmaceuticals/Takeda.
FROM JAMA ONCOLOGY
Key clinical point: Prevalence of monoclonal gammopathy of undetermined significance (MGUS) was twofold higher in Vietnam War veterans exposed to Agent Orange, compared with veterans not exposed.
Major finding: Among veterans who conducted aerial spray missions using Agent Orange, MGUS prevalence was 7.1% (34 of 479), compared with 3.1% (15 of 479) among comparison veterans (adjusted odds ratio, 2.37; 95% CI, 1.27-4.44; P = .007).
Data source: Serum samples collected in 2002 from U.S. Air Force personnel exposed to Agent Orange from 1962 to 1971 (n = 479) and control veterans who were not involved in aerial spray missions (n = 479).
Disclosures: The study was supported by the Agency for Toxic Substances and Disease Registry, the National Cancer Institute, and the Air Force Health Study Assets Research Program. Dr. Landgren reported having consulting or advisory roles with Onyx Pharmaceuticals/AMGEN, Celgene, Bristol-Myers Squibb, Jansen, and Millennium Pharmaceuticals/Takeda.
Team reports latest results of CTL019 in CLL

Photo from Penn Medicine
The chimeric antigen receptor (CAR) T-cell therapy CTL019 can produce durable responses in patients with relapsed/refractory chronic lymphocytic leukemia (CLL), according to research published in Science Translational Medicine.
Eight of 14 patients responded to CTL019—4 complete responses (CRs) and 4 partial responses (PRs).
Three of the patients with CRs were still alive and in remission at last follow-up. The longest remission has lasted 53 months.
Only 1 patient with a PR was still alive at last follow-up, and that patient progressed.
Nine patients developed cytokine release syndrome (CRS), some requiring intensive care. And there were 2 cases of tumor lysis syndrome.
These results are the most mature data from this trial. Results from this study were previously presented at ASH 2013 and ASH 2012, and they were published in NEJM and Science Translational Medicine in August 2011.
This study was supported by grants from Novartis, the Leukemia and Lymphoma Society, and the National Institutes of Health. CTL019 was originally developed at the University of Pennsylvania, but the university licensed the technology to Novartis.
Treatment and outcomes
The trial enrolled 23 CLL patients, but only 14 received CTL019. The 14 patients had a median age of 66 (range, 51 to 78), and most (n=14) were male.
They had received a median of 5 prior therapies (range, 1 to 11), and 8 patients had 17p deletion. All patients had active disease at the time of CTL019 infusion.
Patients received CTL019 at doses of 0.14 × 108 to 11 × 108 cells (median, 1.6 × 108 cells). Eight patients responded to the treatment, for an overall response rate of 57%.
Four patients (29%) achieved a CR. One of these patients died while in remission at 21 months due to infectious complications that occurred after removal of a basal cell carcinoma on his leg.
The other 3 CR patients remained alive at the time of analysis, with no evidence of leukemia at 28 months, 52 months, and 53 months after receiving their infusions. They did not receive additional therapy after CTL019.
“The durability of the remissions we have observed in this study are remarkable and have given us great hope that personalized cell therapies are going to be important options for patients whose cancers are no longer treatable with standard approaches,” said study author David L. Porter, MD, of the University of Pennsylvania Perelman School of Medicine in Philadelphia.
Four patients (29%) achieved a PR after receiving CTL019, with responses lasting a median of 7 months. Two of these patients died of disease progression 10 months and 27 months after receiving CTL019.
One PR patient died after suffering a pulmonary embolism 6 months after CTL019 infusion. The last PR patient experienced disease progression at 13 months, but the patient remained alive on other therapies 36 months after receiving CTL019.
Six patients (43%) did not respond to CTL019 and progressed within 1 month to 9 months. Tests revealed that the modified T cells did not expand as robustly in these patients as in those who experienced remissions.
Two of these patients later died from their disease or complications of other therapies, and 4 are receiving other types of treatment.
CRS and other toxicity
The investigators said infusional toxicities were infrequent and mild (less than grade 2). They were primarily low-grade fevers and chills.
The most frequent related adverse events were associated with complications of neutropenia (including fevers) and delayed CRS, which was correlated with in vivo CTL019 expansion. Nine patients (including all 8 responders) developed CRS.
Five patients with CRS required anti-cytokine-directed therapy—tocilizumab (n=4) and/or steroids (n=3). Four patients required intensive care for complications related to CRS, such as hypotension and hypoxia. They remained in the intensive care unit for a median of 6 days (range, 1-9).
Concurrent with CRS were 6 neurologic events in 5 patients—grade 1/2 hallucinations, confusion, or delirium typically associated with high fevers, intensive care, or medication use. There was 1 case of grade 4 confusion that lasted 2 days and was attributed, at least partly, to CTL019.
There were 2 cases of tumor lysis syndrome. And 1 patient died in remission 21 months after CTL019 infusion, having developed overwhelming ecthyma gangrenosum from a pseudomonas wound infection from a skin biopsy site.
CTL019 durability
“Importantly, our tests of patients who experienced complete remissions showed that the modified cells remain in patients’ bodies for years after their infusions, with no sign of cancerous or normal B cells,” said study author Carl H. June, MD, of the University of Pennsylvania Perelman School of Medicine.
“This suggests that at least some of the CTL019 cells retain their ability to hunt for cancerous cells for long periods of time.”
A lab experiment using CAR T cells isolated from one of the first patients to receive CTL019 confirmed the potential for long-term function of these cells. At nearly 3 years after infusion, the patient’s CTL019 cells demonstrated immediate and specific reactivity against cells expressing CD19.
CTL019 development
The investigators did not identify demographic or disease-related factors, such as age or types of prior therapies, that could be used to predict response to CTL019. And there was no association between T-cell dose and patient response.
An ongoing dose-optimization study is exploring this relationship in greater detail. Further future areas of study may include strategies to combine CTL019 with immune checkpoint inhibitors or other therapies to stimulate T-cell recognition of tumor cells.
In addition to CLL, CTL019 is under investigation in patients with acute lymphoblastic leukemia, non-Hodgkin lymphoma, and myeloma. The product has breakthrough designation from the US Food and Drug Administration for acute lymphoblastic leukemia. ![]()

Photo from Penn Medicine
The chimeric antigen receptor (CAR) T-cell therapy CTL019 can produce durable responses in patients with relapsed/refractory chronic lymphocytic leukemia (CLL), according to research published in Science Translational Medicine.
Eight of 14 patients responded to CTL019—4 complete responses (CRs) and 4 partial responses (PRs).
Three of the patients with CRs were still alive and in remission at last follow-up. The longest remission has lasted 53 months.
Only 1 patient with a PR was still alive at last follow-up, and that patient progressed.
Nine patients developed cytokine release syndrome (CRS), some requiring intensive care. And there were 2 cases of tumor lysis syndrome.
These results are the most mature data from this trial. Results from this study were previously presented at ASH 2013 and ASH 2012, and they were published in NEJM and Science Translational Medicine in August 2011.
This study was supported by grants from Novartis, the Leukemia and Lymphoma Society, and the National Institutes of Health. CTL019 was originally developed at the University of Pennsylvania, but the university licensed the technology to Novartis.
Treatment and outcomes
The trial enrolled 23 CLL patients, but only 14 received CTL019. The 14 patients had a median age of 66 (range, 51 to 78), and most (n=14) were male.
They had received a median of 5 prior therapies (range, 1 to 11), and 8 patients had 17p deletion. All patients had active disease at the time of CTL019 infusion.
Patients received CTL019 at doses of 0.14 × 108 to 11 × 108 cells (median, 1.6 × 108 cells). Eight patients responded to the treatment, for an overall response rate of 57%.
Four patients (29%) achieved a CR. One of these patients died while in remission at 21 months due to infectious complications that occurred after removal of a basal cell carcinoma on his leg.
The other 3 CR patients remained alive at the time of analysis, with no evidence of leukemia at 28 months, 52 months, and 53 months after receiving their infusions. They did not receive additional therapy after CTL019.
“The durability of the remissions we have observed in this study are remarkable and have given us great hope that personalized cell therapies are going to be important options for patients whose cancers are no longer treatable with standard approaches,” said study author David L. Porter, MD, of the University of Pennsylvania Perelman School of Medicine in Philadelphia.
Four patients (29%) achieved a PR after receiving CTL019, with responses lasting a median of 7 months. Two of these patients died of disease progression 10 months and 27 months after receiving CTL019.
One PR patient died after suffering a pulmonary embolism 6 months after CTL019 infusion. The last PR patient experienced disease progression at 13 months, but the patient remained alive on other therapies 36 months after receiving CTL019.
Six patients (43%) did not respond to CTL019 and progressed within 1 month to 9 months. Tests revealed that the modified T cells did not expand as robustly in these patients as in those who experienced remissions.
Two of these patients later died from their disease or complications of other therapies, and 4 are receiving other types of treatment.
CRS and other toxicity
The investigators said infusional toxicities were infrequent and mild (less than grade 2). They were primarily low-grade fevers and chills.
The most frequent related adverse events were associated with complications of neutropenia (including fevers) and delayed CRS, which was correlated with in vivo CTL019 expansion. Nine patients (including all 8 responders) developed CRS.
Five patients with CRS required anti-cytokine-directed therapy—tocilizumab (n=4) and/or steroids (n=3). Four patients required intensive care for complications related to CRS, such as hypotension and hypoxia. They remained in the intensive care unit for a median of 6 days (range, 1-9).
Concurrent with CRS were 6 neurologic events in 5 patients—grade 1/2 hallucinations, confusion, or delirium typically associated with high fevers, intensive care, or medication use. There was 1 case of grade 4 confusion that lasted 2 days and was attributed, at least partly, to CTL019.
There were 2 cases of tumor lysis syndrome. And 1 patient died in remission 21 months after CTL019 infusion, having developed overwhelming ecthyma gangrenosum from a pseudomonas wound infection from a skin biopsy site.
CTL019 durability
“Importantly, our tests of patients who experienced complete remissions showed that the modified cells remain in patients’ bodies for years after their infusions, with no sign of cancerous or normal B cells,” said study author Carl H. June, MD, of the University of Pennsylvania Perelman School of Medicine.
“This suggests that at least some of the CTL019 cells retain their ability to hunt for cancerous cells for long periods of time.”
A lab experiment using CAR T cells isolated from one of the first patients to receive CTL019 confirmed the potential for long-term function of these cells. At nearly 3 years after infusion, the patient’s CTL019 cells demonstrated immediate and specific reactivity against cells expressing CD19.
CTL019 development
The investigators did not identify demographic or disease-related factors, such as age or types of prior therapies, that could be used to predict response to CTL019. And there was no association between T-cell dose and patient response.
An ongoing dose-optimization study is exploring this relationship in greater detail. Further future areas of study may include strategies to combine CTL019 with immune checkpoint inhibitors or other therapies to stimulate T-cell recognition of tumor cells.
In addition to CLL, CTL019 is under investigation in patients with acute lymphoblastic leukemia, non-Hodgkin lymphoma, and myeloma. The product has breakthrough designation from the US Food and Drug Administration for acute lymphoblastic leukemia. ![]()

Photo from Penn Medicine
The chimeric antigen receptor (CAR) T-cell therapy CTL019 can produce durable responses in patients with relapsed/refractory chronic lymphocytic leukemia (CLL), according to research published in Science Translational Medicine.
Eight of 14 patients responded to CTL019—4 complete responses (CRs) and 4 partial responses (PRs).
Three of the patients with CRs were still alive and in remission at last follow-up. The longest remission has lasted 53 months.
Only 1 patient with a PR was still alive at last follow-up, and that patient progressed.
Nine patients developed cytokine release syndrome (CRS), some requiring intensive care. And there were 2 cases of tumor lysis syndrome.
These results are the most mature data from this trial. Results from this study were previously presented at ASH 2013 and ASH 2012, and they were published in NEJM and Science Translational Medicine in August 2011.
This study was supported by grants from Novartis, the Leukemia and Lymphoma Society, and the National Institutes of Health. CTL019 was originally developed at the University of Pennsylvania, but the university licensed the technology to Novartis.
Treatment and outcomes
The trial enrolled 23 CLL patients, but only 14 received CTL019. The 14 patients had a median age of 66 (range, 51 to 78), and most (n=14) were male.
They had received a median of 5 prior therapies (range, 1 to 11), and 8 patients had 17p deletion. All patients had active disease at the time of CTL019 infusion.
Patients received CTL019 at doses of 0.14 × 108 to 11 × 108 cells (median, 1.6 × 108 cells). Eight patients responded to the treatment, for an overall response rate of 57%.
Four patients (29%) achieved a CR. One of these patients died while in remission at 21 months due to infectious complications that occurred after removal of a basal cell carcinoma on his leg.
The other 3 CR patients remained alive at the time of analysis, with no evidence of leukemia at 28 months, 52 months, and 53 months after receiving their infusions. They did not receive additional therapy after CTL019.
“The durability of the remissions we have observed in this study are remarkable and have given us great hope that personalized cell therapies are going to be important options for patients whose cancers are no longer treatable with standard approaches,” said study author David L. Porter, MD, of the University of Pennsylvania Perelman School of Medicine in Philadelphia.
Four patients (29%) achieved a PR after receiving CTL019, with responses lasting a median of 7 months. Two of these patients died of disease progression 10 months and 27 months after receiving CTL019.
One PR patient died after suffering a pulmonary embolism 6 months after CTL019 infusion. The last PR patient experienced disease progression at 13 months, but the patient remained alive on other therapies 36 months after receiving CTL019.
Six patients (43%) did not respond to CTL019 and progressed within 1 month to 9 months. Tests revealed that the modified T cells did not expand as robustly in these patients as in those who experienced remissions.
Two of these patients later died from their disease or complications of other therapies, and 4 are receiving other types of treatment.
CRS and other toxicity
The investigators said infusional toxicities were infrequent and mild (less than grade 2). They were primarily low-grade fevers and chills.
The most frequent related adverse events were associated with complications of neutropenia (including fevers) and delayed CRS, which was correlated with in vivo CTL019 expansion. Nine patients (including all 8 responders) developed CRS.
Five patients with CRS required anti-cytokine-directed therapy—tocilizumab (n=4) and/or steroids (n=3). Four patients required intensive care for complications related to CRS, such as hypotension and hypoxia. They remained in the intensive care unit for a median of 6 days (range, 1-9).
Concurrent with CRS were 6 neurologic events in 5 patients—grade 1/2 hallucinations, confusion, or delirium typically associated with high fevers, intensive care, or medication use. There was 1 case of grade 4 confusion that lasted 2 days and was attributed, at least partly, to CTL019.
There were 2 cases of tumor lysis syndrome. And 1 patient died in remission 21 months after CTL019 infusion, having developed overwhelming ecthyma gangrenosum from a pseudomonas wound infection from a skin biopsy site.
CTL019 durability
“Importantly, our tests of patients who experienced complete remissions showed that the modified cells remain in patients’ bodies for years after their infusions, with no sign of cancerous or normal B cells,” said study author Carl H. June, MD, of the University of Pennsylvania Perelman School of Medicine.
“This suggests that at least some of the CTL019 cells retain their ability to hunt for cancerous cells for long periods of time.”
A lab experiment using CAR T cells isolated from one of the first patients to receive CTL019 confirmed the potential for long-term function of these cells. At nearly 3 years after infusion, the patient’s CTL019 cells demonstrated immediate and specific reactivity against cells expressing CD19.
CTL019 development
The investigators did not identify demographic or disease-related factors, such as age or types of prior therapies, that could be used to predict response to CTL019. And there was no association between T-cell dose and patient response.
An ongoing dose-optimization study is exploring this relationship in greater detail. Further future areas of study may include strategies to combine CTL019 with immune checkpoint inhibitors or other therapies to stimulate T-cell recognition of tumor cells.
In addition to CLL, CTL019 is under investigation in patients with acute lymphoblastic leukemia, non-Hodgkin lymphoma, and myeloma. The product has breakthrough designation from the US Food and Drug Administration for acute lymphoblastic leukemia. ![]()
FDA approves drug to prevent delayed CINV in adults

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved rolapitant (Varubi) for use in adult cancer patients receiving initial and repeat courses of emetogenic chemotherapy.
Rolapitant is to be used in combination with other antiemetic agents to prevent delayed chemotherapy-induced nausea and vomiting (CINV).
Tesaro, Inc., the company developing rolapitant, plans to launch the drug in the fourth quarter of this year.
Rolapitant is a selective and competitive antagonist of human substance P/neurokinin 1 (NK-1) receptors, with a plasma half-life of approximately 7 days. Activation of NK-1 receptors plays a central role in CINV, particularly in the delayed phase (the 25- to 120-hour period after chemotherapy administration).
Rolapitant comes in tablet form. The recommended dose is 180 mg, given approximately 1 to 2 hours prior to chemotherapy administration in combination with a 5-HT3 receptor antagonist and dexamethasone. No dosage adjustment is required for dexamethasone when administering rolapitant.
Rolapitant inhibits the CYP2D6 enzyme, so it is contraindicated with the use of thioridazine, a drug metabolized by the CYP2D6 enzyme. Use of these drugs together may increase the amount of thioridazine in the blood and cause an abnormal heart rhythm that can be serious.
Rolapitant clinical trials
Results from three phase 3 trials suggested that rolapitant (at 180 mg) in combination with a 5-HT3 receptor antagonist and dexamethasone was more effective than the 5-HT3 receptor antagonist and dexamethasone on their own (active control).
The 3-drug combination demonstrated a significant reduction in episodes of vomiting or use of rescue medication during the 25- to 120-hour period following administration of highly emetogenic and moderately emetogenic chemotherapy regimens.
In addition, patients who received rolapitant reported experiencing less nausea that interfered with normal daily life and fewer episodes of vomiting or retching over multiple cycles of chemotherapy.
Highly emetogenic chemotherapy
The clinical profile of rolapitant in cisplatin-based, highly emetogenic chemotherapy (HEC) was confirmed in two phase 3 studies: HEC1 and HEC2. Results from these trials were recently published in The Lancet Oncology.
Both trials met their primary endpoint of complete response (CR) and demonstrated statistical superiority of the rolapitant combination compared to active control.
In HEC1, 264 patients received the rolapitant combination, and 262 received active control. The proportion of patients achieving a CR was 72.7% and 58.4%, respectively (P<0.001).
In HEC2, 271 patients received the rolapitant combination, and 273 received active control. The proportion of patients achieving a CR was 70.1% and 61.9%, respectively (P=0.043).
The most common adverse events (≥3%) were neutropenia (9% rolapitant and 8% control), hiccups (5% and 4%), and abdominal pain (3% and 2%).
Moderately emetogenic chemotherapy
Researchers conducted another phase 3 trial to compare the rolapitant combination with active control in 1332 patients receiving moderately emetogenic chemotherapy regimens. Results from this trial were recently published in The Lancet Oncology.
This trial met its primary endpoint of CR and demonstrated statistical superiority of the rolapitant combination compared to active control. The proportion of patients achieving a CR was 71.3% and 61.6%, respectively (P<0.001).
The most common adverse events (≥3%) were decreased appetite (9% rolapitant and 7% control), neutropenia (7% and 6%), dizziness (6% and 4%), dyspepsia (4% and 2%), urinary tract infection (4% and 3%), stomatitis (4% and 2%), and anemia (3% and 2%).
The full prescribing information for rolapitant is available at www.varubirx.com. ![]()

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved rolapitant (Varubi) for use in adult cancer patients receiving initial and repeat courses of emetogenic chemotherapy.
Rolapitant is to be used in combination with other antiemetic agents to prevent delayed chemotherapy-induced nausea and vomiting (CINV).
Tesaro, Inc., the company developing rolapitant, plans to launch the drug in the fourth quarter of this year.
Rolapitant is a selective and competitive antagonist of human substance P/neurokinin 1 (NK-1) receptors, with a plasma half-life of approximately 7 days. Activation of NK-1 receptors plays a central role in CINV, particularly in the delayed phase (the 25- to 120-hour period after chemotherapy administration).
Rolapitant comes in tablet form. The recommended dose is 180 mg, given approximately 1 to 2 hours prior to chemotherapy administration in combination with a 5-HT3 receptor antagonist and dexamethasone. No dosage adjustment is required for dexamethasone when administering rolapitant.
Rolapitant inhibits the CYP2D6 enzyme, so it is contraindicated with the use of thioridazine, a drug metabolized by the CYP2D6 enzyme. Use of these drugs together may increase the amount of thioridazine in the blood and cause an abnormal heart rhythm that can be serious.
Rolapitant clinical trials
Results from three phase 3 trials suggested that rolapitant (at 180 mg) in combination with a 5-HT3 receptor antagonist and dexamethasone was more effective than the 5-HT3 receptor antagonist and dexamethasone on their own (active control).
The 3-drug combination demonstrated a significant reduction in episodes of vomiting or use of rescue medication during the 25- to 120-hour period following administration of highly emetogenic and moderately emetogenic chemotherapy regimens.
In addition, patients who received rolapitant reported experiencing less nausea that interfered with normal daily life and fewer episodes of vomiting or retching over multiple cycles of chemotherapy.
Highly emetogenic chemotherapy
The clinical profile of rolapitant in cisplatin-based, highly emetogenic chemotherapy (HEC) was confirmed in two phase 3 studies: HEC1 and HEC2. Results from these trials were recently published in The Lancet Oncology.
Both trials met their primary endpoint of complete response (CR) and demonstrated statistical superiority of the rolapitant combination compared to active control.
In HEC1, 264 patients received the rolapitant combination, and 262 received active control. The proportion of patients achieving a CR was 72.7% and 58.4%, respectively (P<0.001).
In HEC2, 271 patients received the rolapitant combination, and 273 received active control. The proportion of patients achieving a CR was 70.1% and 61.9%, respectively (P=0.043).
The most common adverse events (≥3%) were neutropenia (9% rolapitant and 8% control), hiccups (5% and 4%), and abdominal pain (3% and 2%).
Moderately emetogenic chemotherapy
Researchers conducted another phase 3 trial to compare the rolapitant combination with active control in 1332 patients receiving moderately emetogenic chemotherapy regimens. Results from this trial were recently published in The Lancet Oncology.
This trial met its primary endpoint of CR and demonstrated statistical superiority of the rolapitant combination compared to active control. The proportion of patients achieving a CR was 71.3% and 61.6%, respectively (P<0.001).
The most common adverse events (≥3%) were decreased appetite (9% rolapitant and 7% control), neutropenia (7% and 6%), dizziness (6% and 4%), dyspepsia (4% and 2%), urinary tract infection (4% and 3%), stomatitis (4% and 2%), and anemia (3% and 2%).
The full prescribing information for rolapitant is available at www.varubirx.com. ![]()

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved rolapitant (Varubi) for use in adult cancer patients receiving initial and repeat courses of emetogenic chemotherapy.
Rolapitant is to be used in combination with other antiemetic agents to prevent delayed chemotherapy-induced nausea and vomiting (CINV).
Tesaro, Inc., the company developing rolapitant, plans to launch the drug in the fourth quarter of this year.
Rolapitant is a selective and competitive antagonist of human substance P/neurokinin 1 (NK-1) receptors, with a plasma half-life of approximately 7 days. Activation of NK-1 receptors plays a central role in CINV, particularly in the delayed phase (the 25- to 120-hour period after chemotherapy administration).
Rolapitant comes in tablet form. The recommended dose is 180 mg, given approximately 1 to 2 hours prior to chemotherapy administration in combination with a 5-HT3 receptor antagonist and dexamethasone. No dosage adjustment is required for dexamethasone when administering rolapitant.
Rolapitant inhibits the CYP2D6 enzyme, so it is contraindicated with the use of thioridazine, a drug metabolized by the CYP2D6 enzyme. Use of these drugs together may increase the amount of thioridazine in the blood and cause an abnormal heart rhythm that can be serious.
Rolapitant clinical trials
Results from three phase 3 trials suggested that rolapitant (at 180 mg) in combination with a 5-HT3 receptor antagonist and dexamethasone was more effective than the 5-HT3 receptor antagonist and dexamethasone on their own (active control).
The 3-drug combination demonstrated a significant reduction in episodes of vomiting or use of rescue medication during the 25- to 120-hour period following administration of highly emetogenic and moderately emetogenic chemotherapy regimens.
In addition, patients who received rolapitant reported experiencing less nausea that interfered with normal daily life and fewer episodes of vomiting or retching over multiple cycles of chemotherapy.
Highly emetogenic chemotherapy
The clinical profile of rolapitant in cisplatin-based, highly emetogenic chemotherapy (HEC) was confirmed in two phase 3 studies: HEC1 and HEC2. Results from these trials were recently published in The Lancet Oncology.
Both trials met their primary endpoint of complete response (CR) and demonstrated statistical superiority of the rolapitant combination compared to active control.
In HEC1, 264 patients received the rolapitant combination, and 262 received active control. The proportion of patients achieving a CR was 72.7% and 58.4%, respectively (P<0.001).
In HEC2, 271 patients received the rolapitant combination, and 273 received active control. The proportion of patients achieving a CR was 70.1% and 61.9%, respectively (P=0.043).
The most common adverse events (≥3%) were neutropenia (9% rolapitant and 8% control), hiccups (5% and 4%), and abdominal pain (3% and 2%).
Moderately emetogenic chemotherapy
Researchers conducted another phase 3 trial to compare the rolapitant combination with active control in 1332 patients receiving moderately emetogenic chemotherapy regimens. Results from this trial were recently published in The Lancet Oncology.
This trial met its primary endpoint of CR and demonstrated statistical superiority of the rolapitant combination compared to active control. The proportion of patients achieving a CR was 71.3% and 61.6%, respectively (P<0.001).
The most common adverse events (≥3%) were decreased appetite (9% rolapitant and 7% control), neutropenia (7% and 6%), dizziness (6% and 4%), dyspepsia (4% and 2%), urinary tract infection (4% and 3%), stomatitis (4% and 2%), and anemia (3% and 2%).
The full prescribing information for rolapitant is available at www.varubirx.com. ![]()