User login
Imetelstat elicits response in myelofibrosis, thrombocythemia
The telomerase inhibitor imetelstat showed promise against advanced myelofibrosis and essential thrombocythemia in two industry-funded preliminary studies, according to separate reports published online Sept. 3 in the New England Journal of Medicine.
In previous in vitro and animal studies, imetelstat inhibited the proliferation of various types of malignant cells but was not active in normal somatic tissue. Researchers assessed the agent for advanced myelofibrosis in part because, at present, only one available treatment – allogeneic stem-cell transplantation (ASCT) – sometimes induces long-term remission. ASCT carries a relatively high rate of treatment-related death and complications, and is contraindicated in many older patients.
In the first report, researchers conducted a small, single-center cohort study to collect preliminary data on the agent’s efficacy and safety in 33 patients with primary myelofibrosis (18 participants), myelofibrosis that was related to polycythemia (10 participants), or myelofibrosis associated with essential thrombocytopenia (10 participants). Imetelstat was administered in 2-hour intravenous infusions given in 3-week cycles, said Dr. Ayalew Tefferi of the division of hematology, Mayo Clinic, Rochester Minn.
The median duration of treatment was 8.6 months (range, 1.4-21.7 months). Seven patients (21%) had either a complete or partial response; the 4 patients with a complete response had documented complete reversal of bone marrow fibrosis. The time to onset of response was 3.5 months (range, 1.4-7.2 months), and the median duration of response was 18 months (range, 13-20 months).
These remissions “confirm selective anticlonal activity, which has not previously been documented in drug treatment of myelofibrosis,” noted Dr. Tefferi and his associates (N Engl J Med 2015 Sep 3. doi:10.1056/NEJMoa1310523). Three of the seven patients who responded to imetelstat “had been heavily dependent on red-cell transfusions at study entry and became transfusion-independent and sustained a hemoglobin level of more than 10 g/dL for a minimum of 3 months during therapy,” they noted.
In addition, 8 of 10 patients who had marked leukocytosis at baseline had either a complete resolution (3 patients) or a reduction of at least 50% in white-cell counts (5 patients). All 11 participants who had thrombocytosis at baseline had either complete resolution (10 patients) or a reduction in platelet count of at least 50% (1 patient). Of the 27 participants who had leukoerythroblastosis at baseline, 22 showed either complete resolution (13 patients) or a reduction of at least 50% in the percentage of immature myeloid cells and nucleated red cells (9 patients). Also, 17 of the 21 participants who had at least 1% circulating blasts at baseline had either a complete disappearance of circulating blasts (14 patients) or a reduction of at least 50% (3 patients).
The most clinically significant adverse effect of imetelstat, myelosuppression, occurred in 22 patients (67%) and often necessitated dose reductions. Low-grade elevations in liver enzymes also were a concern. One patient died from an intracranial hemorrhage that the treating physician attributed to drug-induced grade 4 thrombocytopenia. Other adverse events that may or may not have been treatment related included fever, epistaxis, bruising, hematoma, lung infection, skin infection, and upper-GI hemorrhage.
These findings not only identify imetelstat as a possible treatment for myelofibrosis, they also suggest that other telomerase-targeting strategies may be beneficial in this disease, Dr. Tefferi and his associates added.
In the second report, researchers performing a phase-II study at seven medical centers in the United States, Germany, and Switzerland found that imetelstat produced rapid and durable hematologic and molecular responses in all 18 patients in their study of essential thrombocythemia refractory to other treatments. This result is particularly encouraging because current standard therapies “induce nonspecific reductions in platelet counts but do not typically eliminate or alter the biologic characteristics of the disease,” said Dr. Gabriela M. Baerlocher of the department of hematology and the Stem Cell Molecular Diagnostics Laboratory, University of Bern, Switzerland.
These study participants had either failed to respond to hydroxyurea, anagrelide, and interferon therapy or were forced to discontinue these agents because of adverse effects. After weekly treatment with imetelstat at one of two doses, 100% of the patients achieved a hematologic response, attaining platelet counts of 250,000-300,000 per cc. Sixteen participants (89%) achieved a complete hematologic response. The median time to complete response was 6.1 weeks, Dr. Baerlocher and her associates said (N Engl J Med. 2015 Sep 3. doi:10.1056/NEJMoa1503479).
After a median follow-up of 17 months on a maintenance dose of imetelstat, 10 patients were still receiving treatment. The median duration of response had not been reached as of press time (range, 5-30 months).
The most important adverse events were neutropenia (15 patients) and abnormal results on liver-function tests (14 patients). The treating physicians attributed 18 adverse events of grade 3 or higher to the study drug, including headache, anemia, and one syncopal episode. Other adverse events included fatigue, nausea, diarrhea, infections, and rash.
The results of both of these studies are compelling and certainly warrant further research, given the limited treatment options for myeloproliferative disorders.
Although imetelstat’s mechanism of action remains to be elucidated, both studies hint at the possibility that the agent may actually change the natural history of these debilitating disorders.
More important, assessing imetelstat’s long-term safety profile is a vital next step for researchers.
Dr. Mary Armanios and Carol W. Greider, Ph.D., are at Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, Baltimore. Dr. Armanios reported having no relevant disclosures; Dr. Greider reported patents related to an RNA component of telomerase and telomerase-associated proteins. Dr. Armanios and Dr. Greider made these remarks in an editorial accompanying the two reports on imetelstat (N Engl J Med. 2015 Sep 3. doi:10.1056/NEJMe1508740).
The results of both of these studies are compelling and certainly warrant further research, given the limited treatment options for myeloproliferative disorders.
Although imetelstat’s mechanism of action remains to be elucidated, both studies hint at the possibility that the agent may actually change the natural history of these debilitating disorders.
More important, assessing imetelstat’s long-term safety profile is a vital next step for researchers.
Dr. Mary Armanios and Carol W. Greider, Ph.D., are at Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, Baltimore. Dr. Armanios reported having no relevant disclosures; Dr. Greider reported patents related to an RNA component of telomerase and telomerase-associated proteins. Dr. Armanios and Dr. Greider made these remarks in an editorial accompanying the two reports on imetelstat (N Engl J Med. 2015 Sep 3. doi:10.1056/NEJMe1508740).
The results of both of these studies are compelling and certainly warrant further research, given the limited treatment options for myeloproliferative disorders.
Although imetelstat’s mechanism of action remains to be elucidated, both studies hint at the possibility that the agent may actually change the natural history of these debilitating disorders.
More important, assessing imetelstat’s long-term safety profile is a vital next step for researchers.
Dr. Mary Armanios and Carol W. Greider, Ph.D., are at Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, Baltimore. Dr. Armanios reported having no relevant disclosures; Dr. Greider reported patents related to an RNA component of telomerase and telomerase-associated proteins. Dr. Armanios and Dr. Greider made these remarks in an editorial accompanying the two reports on imetelstat (N Engl J Med. 2015 Sep 3. doi:10.1056/NEJMe1508740).
The telomerase inhibitor imetelstat showed promise against advanced myelofibrosis and essential thrombocythemia in two industry-funded preliminary studies, according to separate reports published online Sept. 3 in the New England Journal of Medicine.
In previous in vitro and animal studies, imetelstat inhibited the proliferation of various types of malignant cells but was not active in normal somatic tissue. Researchers assessed the agent for advanced myelofibrosis in part because, at present, only one available treatment – allogeneic stem-cell transplantation (ASCT) – sometimes induces long-term remission. ASCT carries a relatively high rate of treatment-related death and complications, and is contraindicated in many older patients.
In the first report, researchers conducted a small, single-center cohort study to collect preliminary data on the agent’s efficacy and safety in 33 patients with primary myelofibrosis (18 participants), myelofibrosis that was related to polycythemia (10 participants), or myelofibrosis associated with essential thrombocytopenia (10 participants). Imetelstat was administered in 2-hour intravenous infusions given in 3-week cycles, said Dr. Ayalew Tefferi of the division of hematology, Mayo Clinic, Rochester Minn.
The median duration of treatment was 8.6 months (range, 1.4-21.7 months). Seven patients (21%) had either a complete or partial response; the 4 patients with a complete response had documented complete reversal of bone marrow fibrosis. The time to onset of response was 3.5 months (range, 1.4-7.2 months), and the median duration of response was 18 months (range, 13-20 months).
These remissions “confirm selective anticlonal activity, which has not previously been documented in drug treatment of myelofibrosis,” noted Dr. Tefferi and his associates (N Engl J Med 2015 Sep 3. doi:10.1056/NEJMoa1310523). Three of the seven patients who responded to imetelstat “had been heavily dependent on red-cell transfusions at study entry and became transfusion-independent and sustained a hemoglobin level of more than 10 g/dL for a minimum of 3 months during therapy,” they noted.
In addition, 8 of 10 patients who had marked leukocytosis at baseline had either a complete resolution (3 patients) or a reduction of at least 50% in white-cell counts (5 patients). All 11 participants who had thrombocytosis at baseline had either complete resolution (10 patients) or a reduction in platelet count of at least 50% (1 patient). Of the 27 participants who had leukoerythroblastosis at baseline, 22 showed either complete resolution (13 patients) or a reduction of at least 50% in the percentage of immature myeloid cells and nucleated red cells (9 patients). Also, 17 of the 21 participants who had at least 1% circulating blasts at baseline had either a complete disappearance of circulating blasts (14 patients) or a reduction of at least 50% (3 patients).
The most clinically significant adverse effect of imetelstat, myelosuppression, occurred in 22 patients (67%) and often necessitated dose reductions. Low-grade elevations in liver enzymes also were a concern. One patient died from an intracranial hemorrhage that the treating physician attributed to drug-induced grade 4 thrombocytopenia. Other adverse events that may or may not have been treatment related included fever, epistaxis, bruising, hematoma, lung infection, skin infection, and upper-GI hemorrhage.
These findings not only identify imetelstat as a possible treatment for myelofibrosis, they also suggest that other telomerase-targeting strategies may be beneficial in this disease, Dr. Tefferi and his associates added.
In the second report, researchers performing a phase-II study at seven medical centers in the United States, Germany, and Switzerland found that imetelstat produced rapid and durable hematologic and molecular responses in all 18 patients in their study of essential thrombocythemia refractory to other treatments. This result is particularly encouraging because current standard therapies “induce nonspecific reductions in platelet counts but do not typically eliminate or alter the biologic characteristics of the disease,” said Dr. Gabriela M. Baerlocher of the department of hematology and the Stem Cell Molecular Diagnostics Laboratory, University of Bern, Switzerland.
These study participants had either failed to respond to hydroxyurea, anagrelide, and interferon therapy or were forced to discontinue these agents because of adverse effects. After weekly treatment with imetelstat at one of two doses, 100% of the patients achieved a hematologic response, attaining platelet counts of 250,000-300,000 per cc. Sixteen participants (89%) achieved a complete hematologic response. The median time to complete response was 6.1 weeks, Dr. Baerlocher and her associates said (N Engl J Med. 2015 Sep 3. doi:10.1056/NEJMoa1503479).
After a median follow-up of 17 months on a maintenance dose of imetelstat, 10 patients were still receiving treatment. The median duration of response had not been reached as of press time (range, 5-30 months).
The most important adverse events were neutropenia (15 patients) and abnormal results on liver-function tests (14 patients). The treating physicians attributed 18 adverse events of grade 3 or higher to the study drug, including headache, anemia, and one syncopal episode. Other adverse events included fatigue, nausea, diarrhea, infections, and rash.
The telomerase inhibitor imetelstat showed promise against advanced myelofibrosis and essential thrombocythemia in two industry-funded preliminary studies, according to separate reports published online Sept. 3 in the New England Journal of Medicine.
In previous in vitro and animal studies, imetelstat inhibited the proliferation of various types of malignant cells but was not active in normal somatic tissue. Researchers assessed the agent for advanced myelofibrosis in part because, at present, only one available treatment – allogeneic stem-cell transplantation (ASCT) – sometimes induces long-term remission. ASCT carries a relatively high rate of treatment-related death and complications, and is contraindicated in many older patients.
In the first report, researchers conducted a small, single-center cohort study to collect preliminary data on the agent’s efficacy and safety in 33 patients with primary myelofibrosis (18 participants), myelofibrosis that was related to polycythemia (10 participants), or myelofibrosis associated with essential thrombocytopenia (10 participants). Imetelstat was administered in 2-hour intravenous infusions given in 3-week cycles, said Dr. Ayalew Tefferi of the division of hematology, Mayo Clinic, Rochester Minn.
The median duration of treatment was 8.6 months (range, 1.4-21.7 months). Seven patients (21%) had either a complete or partial response; the 4 patients with a complete response had documented complete reversal of bone marrow fibrosis. The time to onset of response was 3.5 months (range, 1.4-7.2 months), and the median duration of response was 18 months (range, 13-20 months).
These remissions “confirm selective anticlonal activity, which has not previously been documented in drug treatment of myelofibrosis,” noted Dr. Tefferi and his associates (N Engl J Med 2015 Sep 3. doi:10.1056/NEJMoa1310523). Three of the seven patients who responded to imetelstat “had been heavily dependent on red-cell transfusions at study entry and became transfusion-independent and sustained a hemoglobin level of more than 10 g/dL for a minimum of 3 months during therapy,” they noted.
In addition, 8 of 10 patients who had marked leukocytosis at baseline had either a complete resolution (3 patients) or a reduction of at least 50% in white-cell counts (5 patients). All 11 participants who had thrombocytosis at baseline had either complete resolution (10 patients) or a reduction in platelet count of at least 50% (1 patient). Of the 27 participants who had leukoerythroblastosis at baseline, 22 showed either complete resolution (13 patients) or a reduction of at least 50% in the percentage of immature myeloid cells and nucleated red cells (9 patients). Also, 17 of the 21 participants who had at least 1% circulating blasts at baseline had either a complete disappearance of circulating blasts (14 patients) or a reduction of at least 50% (3 patients).
The most clinically significant adverse effect of imetelstat, myelosuppression, occurred in 22 patients (67%) and often necessitated dose reductions. Low-grade elevations in liver enzymes also were a concern. One patient died from an intracranial hemorrhage that the treating physician attributed to drug-induced grade 4 thrombocytopenia. Other adverse events that may or may not have been treatment related included fever, epistaxis, bruising, hematoma, lung infection, skin infection, and upper-GI hemorrhage.
These findings not only identify imetelstat as a possible treatment for myelofibrosis, they also suggest that other telomerase-targeting strategies may be beneficial in this disease, Dr. Tefferi and his associates added.
In the second report, researchers performing a phase-II study at seven medical centers in the United States, Germany, and Switzerland found that imetelstat produced rapid and durable hematologic and molecular responses in all 18 patients in their study of essential thrombocythemia refractory to other treatments. This result is particularly encouraging because current standard therapies “induce nonspecific reductions in platelet counts but do not typically eliminate or alter the biologic characteristics of the disease,” said Dr. Gabriela M. Baerlocher of the department of hematology and the Stem Cell Molecular Diagnostics Laboratory, University of Bern, Switzerland.
These study participants had either failed to respond to hydroxyurea, anagrelide, and interferon therapy or were forced to discontinue these agents because of adverse effects. After weekly treatment with imetelstat at one of two doses, 100% of the patients achieved a hematologic response, attaining platelet counts of 250,000-300,000 per cc. Sixteen participants (89%) achieved a complete hematologic response. The median time to complete response was 6.1 weeks, Dr. Baerlocher and her associates said (N Engl J Med. 2015 Sep 3. doi:10.1056/NEJMoa1503479).
After a median follow-up of 17 months on a maintenance dose of imetelstat, 10 patients were still receiving treatment. The median duration of response had not been reached as of press time (range, 5-30 months).
The most important adverse events were neutropenia (15 patients) and abnormal results on liver-function tests (14 patients). The treating physicians attributed 18 adverse events of grade 3 or higher to the study drug, including headache, anemia, and one syncopal episode. Other adverse events included fatigue, nausea, diarrhea, infections, and rash.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: The telomerase inhibitor imetelstat showed promise in separate preliminary studies for treatment of myelofibrosis and thrombocythemia.
Major finding: A complete or partial response to imetelstat was seen in 7 of 33 patients with advanced myelofibrosis and 18 of 18 with thrombocythemia.
Data source: An international phase-II open-label study involving 18 patients with essential thrombocythemia and a single-center observational cohort study involving 33 patients with myelofibrosis.
Disclosures: Both studies were funded by Geron.
Castleman Disease
Castleman disease (CD) is a rare nonclonal lymphoproliferative disorder, also known as angiofollicular lymph-node hyperplasia or giant node hyperplasia. It was first reported in 1954 and in 1956 described by Benjamin Castleman, MD, in a case series of localized mediastinal lymph-node hyperplasia.1 Unicentric Castleman (UCD) disease presents as a localized disease affecting a single lymph node/lymph node chain. Multicentric Castleman disease (MCD) is a more widespread or generalized disease (Table 1). About 4,000 to 6,000 new cases of CD are diagnosed per year of which about 20% to 25% cases are MCD. The estimated incidence rate for CD has recently been calculated as 21 to 25 per million person-years, or about 6,000 new cases annually.2
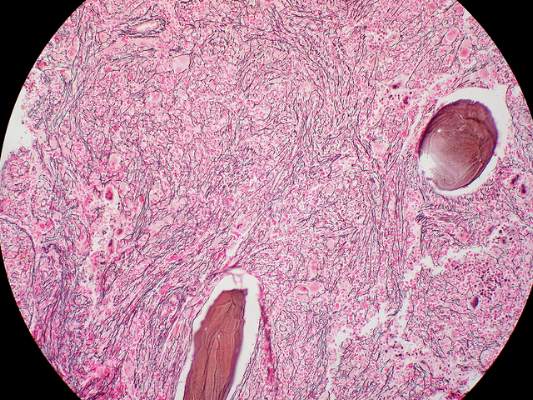
The clinical presentation of CD often overlaps with autoimmune, infectious, or other malignant diseases. The diagnosis is confirmed by a biopsy of the affected lymph-node tissue. Interleukin-6 (IL-6) and a viral analog of IL-6 play major role in the pathogenesis by stimulating a widespread inflammatory response that results in systemic manifestations. It is often associated with HIV and human herpesvirus-8 (HHV-8) infections. Castleman disease is histologically characterized into the hyaline vascular variant, the plasma-cell variant, and the mixed form. The plasmablastic variety is associated with HIV and HHV-8 infections. The prognosis ranges from good in UCD (91% overall survival [OS] at 5 y) to poor in MCD (65% OS at 5 y).3
Treatment options range from local surgical excision to systemic treatments. Newer therapies include monoclonal antibodies against both IL-6 and CD20 and a few other targets in the inflammatory cascade. This article discusses the updated approach to diagnosis and management of CD.
Unicentric Castleman Disease
Unicentric CD is more common than MCD, presents as a localized lymph node or chain involvement, and is generally diagnosed in the third or fourth decade of life but has been reported in children. The presenting symptoms of UCD vary by site. It presents as nontender lymphadenopathy when confined to peripheral lymph nodes, whereas respiratory symptoms or bowel obstruction may be seen with lymphadenopathy in the chest/mediastinum, neck, or abdomen. The systemic
symptoms, such as fever, night sweats, and weight loss, are uncommon.
Dysplastic follicular dendritic cells characterize UCD. Histologically, it is usually classified as hyaline vascular disease with the follicles comprising small lymphocytes
and dendritic cells forming concentric rings with prominent vascularity.4,5 No association with HIV or HHV-8 has been seen.
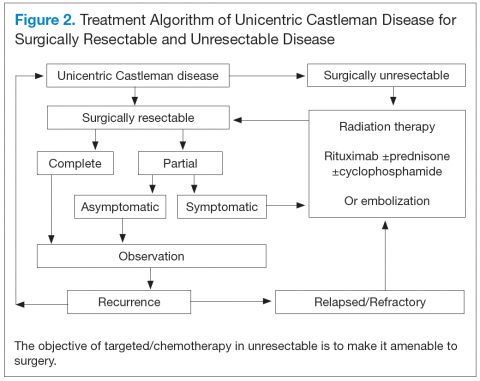
Unicentric CD is often amenable to resection, and a complete cure can be achieved.6 Partial resection may be attempted when complete resection is not possible. Radiation therapy is offered for unresectable disease.7 In patients who are not candidates for any intervention, close long-term follow-up is recommended unless patients are symptomatic, in which case systemic treatment should be considered.
Multicentric Castleman Disease
The more widespread MCD is generally diagnosed in the fifth or sixth decade of life. It is more aggressive than UCD and presents a wide spectrum of symptoms and abnormal laboratory findings (Table 2).8
It is histologically classified into (a) plasmablastic or HHV-8 associated: It is often seen in patients with MCD infected with HIV, which can give rise to large B-cell lymphoma, known as HHV-8 plasmablastic lymphoma9; (b) plasmacytic variant has marked paracortical plasmacytosis with retained nodal architecture10; and (c) mixed MCD has abundant plasma cells with features similar to those of the hyaline-vascular variant.
Most patients with HIV-associated MCD are co-infected with HHV-8. The HHV-8 infection is also present in about 50% of HIV-negative cases.11 The incidence of HIVassociated MCD is increasing in the highly active antiretroviral therapy (HAART) era secondary to improved survival of patients infected with HIV.12 To diagnose active HIV MCD, the French Agence Nationale de Recherche sur le SIDA 117 CastlemaB trial group has described criteria based on the clinical symptoms, including fever, a raised serum C-reactive protein > 20 mg/L without any other cause, and 3 of 12 additional clinical findings described as peripheral lymphadenopathy, splenomegaly, ascites, edema, pleural effusion, cough, nasal obstruction, xerostomia, rash, central neurologic symptoms, jaundice, and autoimmune hemolytic anemia.13 The reported 2-year survival of patients who are HIV-negative is 97% compared with HIV-positive cases at 67%.14
Idiopathic MCD is diagnosed when there is no evidence of any underlying infectious, autoimmune, and neoplastic process.15
Patients with MCD are at an increased risk of developing non-Hodgkin and Hodgkin lymphoma, Kaposi sarcoma, primary effusion lymphoma, and follicular dendritic
cell sarcoma. POEMS (peripheral neuropathy, organomegaly, endocrinopathy, monoclonal protein, skin changes) syndrome and paraneoplastic disease, such as paraneoplastic pemphigus myasthenia gravis, may be commonly diagnosed concurrently or sequentially with MCD.16-20
The disease course of MCD ranges from indolent to rapidly progressive, and its 5-year OS is about 65%. When associated with POEMS syndrome, the 5-year survival was estimated to be 90% with the osteosclerotic variant and 27% without osteosclerotic lesions.3 Treatment options for MCD include systemic chemotherapy, including antiviral therapy for HHV-8 positive and HAART for HIV positive and newer monoclonal antibody therapies targeting CD20 or IL-6.
Pathophysiology
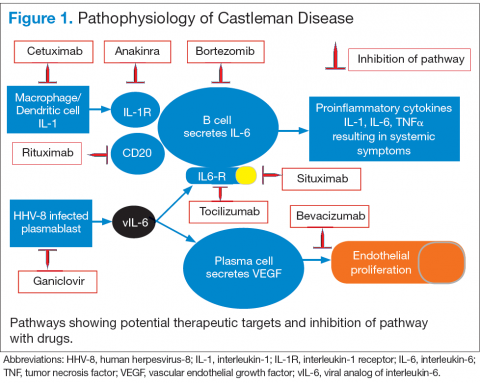
Interleukin-6 plays an important role for inflammation in both UCD and MCD (Figure 1). There is dysregulation and overproduction of IL-6, which further stimulates the production of acute-phase reactants, resulting in various systemic manifestations.15,21,22 There is increased expression of IL-1 and IL-6, upregulation of IL-6 secondary to interaction of IL-1 with nuclear factor-kappa B (NF-kappa B), thus stimulating B-cell proliferation. IL-6 binding to IL-6 receptor (IL6-R) results in downstream activation of transcription Janus kinases/signal transducers and activators of the transcription pathway. This promotes the transcription of genes encoding the acute-phase reactant proteins. Hence, interfering with IL-6 transduction by blocking downstream signals are potential therapeutic targets. The mitogen-activated protein kinase cascade, the rapidly accelerated fibrosarcoma kinases, and the overexpression of the endothelial growth factor receptor (EGFR), all contribute to disease pathogenesis by promoting increased B-cell proliferation and vascular EGFR mediated angiogenesis. 23,24
In HHV-8–associated MCD, the virus replicates within lymph node plasmablasts, causing increased production of viral IL-6 analog, human IL-6, and other proinflammatory proteins resulting in B-cell and plasma-cell proliferation, increased vascular endothelial growth factor secretion and angiogenesis.25,26 The HHV-8–infected plasmablasts are marked by variable expression of CD20, and therefore, anti-CD20 is also shown to be an effective treatment. The calmodulin/calcineurin nuclear factor assists in the proliferation of HHV-8, thereby making calcineurin another potential target for the antiviral proliferation.27
Staging
The treatment decisions and prognosis for patients with CD is based on the clinical and histologic staging. The initial workup includes but is not limited to routine laboratory evaluation, imaging, and HIV and HHV-8 testing (Table 3). Routine tests of the levels of cytokines are not recommended. Other relevant tests for known disease associations should be obtained when relevant.
Treatment
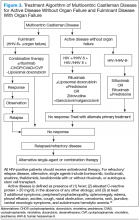
Better understanding of the disease process in CD has helped to identify potential therapeutic targets (Figures 2 and 3).
For UCD, surgery is the mainstay of treatment.4,28,29 In surgically unresectable cases, radiation therapy is helpful for local disease control. Alternatively, neoadjuvant
chemotherapy and rituximab are used. Corticosteroids are generally used to treat acute exacerbations and as adjuncts to chemotherapy.
For MCD, the treatment approach depends on the HIV and HHV-8 status of the patient. For patients with HHV-8 infection, both with and without HIV co-infection, antiviral agents, such as ganciclovir, foscarnet, or cidofovir, have shown in vitro activity against HHV-8 but with limited clinical success.30 In patients infected with HIV, the aim of treating with HAART is to control the disease, prevent opportunistic infections, and improve tolerance to chemotherapy.31-33 Rituximab with or without chemotherapy is the standard treatment approach. The additional chemotherapeutic agents are used depending on the presence or absence of organ failure. This approach has improved the OS in HIV-associated MCD.34,35 Treatment with HAART does not decrease the risk of relapse in HIV MCD; therefore, the role of rituximab and antiherpesvirus agents as maintenance therapy has been explored.36 In patients who fail to respond to or relapse rapidly following rituximab monotherapy, the use of either single-agent chemotherapy with or without rituximab or antiherpesvirus therapy with high-dose zidovudine and valganciclovir is recommended.37
The cytotoxic chemotherapy with single agents, such as etoposide, vinblastine, cyclophosphamide, cladribine, chlorambucil, and liposomal doxorubicin, has been used with limited success.22 The combination chemotherapy with cyclophosphamide/doxorubicin/vincristine/prednisone (CHOP) or cyclophosphamide/vincristine/
prednisone (CVP) without rituximab has been shown to achieve durable remissions. Corticosteroids are usually administered as an initial adjunct to chemotherapy or for acute exacerbations. In patients with MCD, regardless of HIV status, the interferon therapy was found to achieve long-term remission.38,39 The interferon therapy
exerts antiviral effects via downregulation of the IL-6R and inhibition of HHV-8 replication. For patients in remission, maintenance therapy with oral valganciclovir is promising.40
Immunomodulators & Targeted Therapies
For unresectable UCD or MCD with organ failure or relapse, the use of alternativesingle-agent or combination chemotherapies with or without rituximab is recommended. Thalidomide has shown some success, probably secondary to disruption of IL-6 production.41 In cases of progression following second-line therapy, bortezomib, antiherpesvirus therapies, or IL-6–directed therapy with siltuximab or tocilizumab should be considered.
Rituximab is a monoclonal chimeric antibody that targets CD20 on B cells, thus leading to B-cell lymphodepletion via activating complement-dependent cytotoxicity and antibody-dependent cell-mediated cytotoxicity. As monotherapy, it has been shown to achieve 2-year progression-free survival in 80% of patients.42 In patients with MCD who are HIV positive, rituximab with and without chemotherapy has shown improved overall and disease-free survival of 70% to 80% at 2 years.43
Siltuximab is a chimeric human-mouse monoclonal antibody to IL-6 that has been approved for treatment of patients with MCD who are both HIV negative and HHV-8 negative.44-46 Tocilizumab targets the IL-6R. The antibody has shown improvement in a study in HIVseronegative adults with MCD.47,48
Bortezomib is a proteasome inhibitor that inhibits the NF-kappa B pathway, which induces the expression of numerous proinflammatory proteins, including IL-6. It is recommended for relapsed or refractory disease.49,50
Anakinra is a recombinant IL-1R antagonist that blocks IL-1 effects and controls disease by decreasing IL-6 production.51
Conculsion
There has been significant progress in disease diagnosis and management as more information is available about the incidence, clinical presentation, and pathophysiology of CD. The understanding of the disease pathogenesis and biology has helped to discover multiple potential therapeutic targets. Successful treatment has been achieved through targeting HHV-8 replication, CD20, and IL-6 and anti– IL-6R antibodies. Although surgical resection continues to be the
standard of therapy for UCD, the management of MCD and relapsed or refractory disease continues to evolve. Exploration of various treatment strategies in different clinical presentations is warranted.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S.
Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information
for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to
patients.
Click here to read the digital edition.
1. Castleman B, Iverson L, Menendez VP. Localized mediastinal lymph-node hyperplasia
resembling thymoma. Cancer. 1956;9(4):822-830.
2. Munshi N, Mehra M, van de Velde H, Desai A, Potluri R, Vermeulen J. Use of a claims database to characterize and estimate the incidence rate for Castleman disease. Leuk Lymphoma. 2015;56(5):1252-1260.
3. Dispenzieri A, Armitage JO, Loe MJ, et al. The clinical spectrum of Castleman’s disease. Am J Hematol. 2012;87(11):997-1002.
4. Keller AR, Hochholzer L, Castleman B. Hyaline-vascular and plasma-cell types of giant lymph node hyperplasia of the mediastinum and other locations. Cancer. 1972;29(3):670-683.
5. Cronin DM, Warnke RA. Castleman disease: an update on classification and the
spectrum of associated lesions. Adv Anat Pathol. 2009;16(4):236-246.
6. Talat N, Belgaumkar AP, Schulte KM. Surgery in Castleman’s disease: a systematic review of 404 published cases. Ann Surg. 2012;255(4):677-684.
7. Chronowski GM, Ha CS, Wilder RB, Cabanillas F, Manning J, Cox JD. Treatment of unicentric and multicentric Castleman disease and the role of radiotherapy. Cancer. 2001;92(3):670-676.
8. Herrada J, Cabanillas F, Rice L, Manning J, Pugh W. The clinical behavior of localized and multicentric Castleman disease. Ann Intern Med. 1998;128(8):657-662.
9. Dupin N, Diss TL, Kellam P, et al. HHV-8 is associated with a plasmablastic variant of Castleman disease that is linked to HHV-8-positive plasmablastic lymphoma. Blood. 2000;95(4):1406-1412.
10. Ferry JA, Harris NL. Atlas of Lymphoid Hyperplasia and Lymphoma. Philadelphia, PA: W.B. Saunders; 1997.
11. Soulier J, Grollet L, Oksenhendler E, et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood. 1995;86(4):1276-1280.
12. Powles T, Stebbing J, Bazeos A, et al. The role of immune suppression and HHV-8 in the increasing incidence of HIV-associated multicentric Castleman’s disease. Ann Oncol. 2009;20(4):775-779.
13. Gérard L, Bérezné A, Galicier L, et al. Prospective study of rituximab in chemotherapy-dependent human immunodeficiency virus associated multicentric Castleman’s disease: ANRS 117 CastlemaB Trial. J Clin Oncol. 2007;25(22):3350-3356.
14. Casper C, Teltsch DY, Robinson D Jr, et al. Clinical characteristics and healthcare utilization of patients with multicentric Castleman disease. Br J Haematol. 2015;168(1):82-93.
15. Fajgenbaum DC, van Rhee F, Nabel CS. HHV-8-negative, idiopathic multicentric Castleman disease: novel insights into biology, pathogenesis, and therapy. Blood. 2014;123(19):2924-2933.
16. Larroche C, Cacoub P, Soulier J, et al. Castleman’s disease and lymphoma: report of eight cases in HIV-negative patients and literature review. Am J Hematol. 2002;69(2):119-126.
17. Dispenzieri A. POEMS syndrome: 2014 update on diagnosis, risk-stratification, and management. Am J Hematol. 2014;89(2):214-223.
18. Andhavarapu S, Jiang L. POEMS syndrome and Castleman disease. Blood. 2013;122(2):159.
19. Bélec L, Mohamed AS, Authier FJ, et al. Human herpesvirus 8 infection in patients with POEMS syndrome-associated multicentric Castleman’s disease. Blood. 1999;93(11):3643-3653.
20. Oksenhendler E, Boulanger E, Galicier L, et al. High incidence of Kaposi sarcomaassociated herpesvirus-related non-Hodgkin lymphoma in patients with HIV infection and multicentric Castleman disease. Blood. 2002;99(7):2331-2336.
21. Yoshizaki K, Matsuda T, Nishimoto N, et al. Pathogenic significance of interleukin-6 (IL-6/BSF-2) in Castleman’s disease. Blood. 1989;74(4):1360-1367.
22. El-Osta HE, Kurzrock R. Castleman’s disease: from basic mechanisms to molecular therapeutics. Oncologist. 2011;16(4):497-511.
23. Akula SM, Ford PW, Whitman AG, et al. B-Raf-dependent expression of vascular endothelial growth factor-A in Kaposi sarcoma-associated herpesvirus-infected human B cells. Blood. 2005;105(11):4516-4522.
24. Sun X, Chang KC, Abruzzo LV, Lai R, Younes A, Jones D. Epidermal growth factor receptor expression in follicular dendritic cells: a shared feature of follicular dendritic cell sarcoma and Castleman’s disease. Hum Pathol. 2003;34(9):835-840.
25. Adam N, Rabe B, Suthaus J, Grötzinger J, Rose-John S, Scheller J. Unraveling viral interleukin-6 binding to gp130 and activation of STAT-signaling pathways independently of the interleukin-6 receptor. J Virol. 2009;83(10):5117-5126.
26. Suda T, Katano H, Delsol G, et al. HHV-8 infection status of AIDS-unrelated and
AIDS-associated multicentric Castleman’s disease. Pathol Int. 2001;51(9):671-679.
27. Zoeteweij JP, Moses AV, Rinderknecht AS, et al. Targeted inhibition of calcineurin signaling blocks calcium-dependent reactivation of Kaposi sarcoma-associated herpesvirus. Blood. 2001;97(8):2374-2380.
28. McCarty MJ, Vukelja SJ, Banks PM, Weiss RB. Angiofollicular lymph node hyperplasia
(Castleman’s disease). Cancer Treat Rev. 1995;21(4):291-310.
29. Bowne WB, Lewis JJ, Filippa DA, et al. The management of unicentric and multicentric Castleman’s disease: a report of 16 cases and a review of the literature. Cancer. 1999;85(3):706-717.
30. Reddy D, Mitsuyasu R. HIV-associated multicentric Castleman disease. Curr Opin Oncol. 2011;23(5):475-481.
31. Aaron L, Lidove O, Yousry C, Roudiere L, Dupont B, Viard JP. Human herpesvirus 8-positive Castleman disease in human immunodeficiency virus-infected patients: the impact of highly active antiretroviral therapy. Clin Infect Dis. 2002;35(7):880-882.
32. Sprinz E, Jeffman M, Liedke P, Putten A, Schwartsmann G. Successful treatment of AIDS-related Castleman’s disease following the administration of highly active antiretroviral therapy (HAART). Ann Oncol. 2004;15(2):356-358.
33. Lee SM, Edwards SG, Chilton DN, Ramsay A, Miller RF. Highly active antiretroviral therapy alone may be an effective treatment for HIV-associated multi-centric Castleman’s disease. Haematologica. 2010;95(11):1979-1981.
34. Bower M. How I treat HIV-associated multicentric Castleman disease. Blood. 2010;116(22):4415-4421.
35. Bower M, Newsom-Davis T, Naresh K, et al. Clinical features and outcome in HIVassociated
multicentric Castleman’s disease. J Clin Oncol. 2011;29(18):2481-2486.
36. Casper C, Nichols WG, Huang ML, Corey L, Wald A. Remission of HHV-8 and HIV-associated multicentric Castleman disease with ganciclovir treatment. Blood. 2004;103(5):1632-1634.
37. Uldrick TS, Polizzotto MN, Aleman K, et al. High-dose zidovudine plus valganciclovir for Kaposi sarcoma herpesvirus-associated multicentric Castleman disease: a pilot study of virus-activated cytotoxic therapy. Blood. 2011;117(26):6977-6986.
38. Kumari P, Schechter GP, Saini N, Benator DA. Successful treatment of human immunodeficiency virus-related Castleman’s disease with interferon-alpha. Clin Infect Dis. 2000;31(2):602-604.
39. Nord JA, Karter D. Low dose interferon-alpha therapy for HIV-associated multicentric Castleman’s disease. Int J STD AIDS. 2003;14(1):61-62.
40. Oksenhendler E. HIV-associated multicentric Castleman disease. Curr Opin HIV AIDS. 2009;4(1):16-21.
41. Jung CP, Emmerich B, Goebel FD, Bogner JR. Successful treatment of a patient with HIV-associated multicentric Castleman disease (MCD) with thalidomide. Am J Hematol. 2004;75(3):176-177.
42. Ide M, Kawachi Y, Izumi Y, Kasagi K, Ogino T. Long-term remission in HIV negative patients with multicentric Castleman’s disease using rituximab. Eur J Haematol. 2006;76(2):119-123.
43. Marcelin AG, Aaron L, Mateus C, et al. Rituximab therapy for HIV-associated Castleman disease. Blood. 2003;102(8):2786-2788.
44. Van Rhee F, Fayad L, Voorhees P, et al. Siltuximab, a novel anti-interleukin-6 monoclonal antibody, for Castleman’s disease. J Clin Oncol. 2010;28(23):3701-3708.
45. Wong RS, Casper C, Munshi N, et al. A multicenter, randomized, doubleblind, placebo-controlled study of the efficacy and safety of siltuximab, an antiinterleukin-6 monoclonal antibody, in patients with multicentric Castleman’s disease. Blood. 2013;122(21):505.
46. Van Rhee F, Casper C, Voorhees PM, et al. An open-label, phase 2, multicenter study of the safety of long-term treatment with siltuximab (an anti-interleukin-6 monoclonal antibody) in patients with multicentric Castleman’s disease. Blood. 2013;122(21):1806.
47. Nishimoto N, Kanakura Y, Aozasa K, et al. Humanized anti-interleukin-6 receptor antibody treatment of multicentric Castleman disease. Blood. 2005;106(8):2627-2632.
48. Müzes G, Sipos F, Csomor J, Sréter L. Successful tocilizumab treatment in a patient with human herpesvirus 8-positive and human immunodeficiency virusnegative multicentric Castleman’s disease of plasma cell type nonresponsive to rituximab-CVP therapy. APMIS. 2013;121(7):668-674.
49. Hess G, Wagner V, Kreft A, Heussel CP, Huber C. Effects of bortezomib on proinflammatory cytokine levels and transfusion dependency in a patient with multicentric Castleman disease. Br J Haematol. 2006;134(5):544-545.
50. Sobas MA, Alonso Vence N, Diaz Arias J, Bendaña Lopez A, Fraga Rodriguez M, Bello Lopez JL. Efficacy of bortezomib in refractory form of multicentric Castleman disease associated to poems syndrome (MCD-POEMS variant). Ann Hematol. 2010;89(2):217-219.
51. El-Osta H, Janku F, Kurzrock R. Successful treatment of Castleman’s disease with interleukin-1 receptor antagonist (Anakinra). Mol Cancer Ther. 2010;9(6):1485-1488.
Castleman disease (CD) is a rare nonclonal lymphoproliferative disorder, also known as angiofollicular lymph-node hyperplasia or giant node hyperplasia. It was first reported in 1954 and in 1956 described by Benjamin Castleman, MD, in a case series of localized mediastinal lymph-node hyperplasia.1 Unicentric Castleman (UCD) disease presents as a localized disease affecting a single lymph node/lymph node chain. Multicentric Castleman disease (MCD) is a more widespread or generalized disease (Table 1). About 4,000 to 6,000 new cases of CD are diagnosed per year of which about 20% to 25% cases are MCD. The estimated incidence rate for CD has recently been calculated as 21 to 25 per million person-years, or about 6,000 new cases annually.2
The clinical presentation of CD often overlaps with autoimmune, infectious, or other malignant diseases. The diagnosis is confirmed by a biopsy of the affected lymph-node tissue. Interleukin-6 (IL-6) and a viral analog of IL-6 play major role in the pathogenesis by stimulating a widespread inflammatory response that results in systemic manifestations. It is often associated with HIV and human herpesvirus-8 (HHV-8) infections. Castleman disease is histologically characterized into the hyaline vascular variant, the plasma-cell variant, and the mixed form. The plasmablastic variety is associated with HIV and HHV-8 infections. The prognosis ranges from good in UCD (91% overall survival [OS] at 5 y) to poor in MCD (65% OS at 5 y).3
Treatment options range from local surgical excision to systemic treatments. Newer therapies include monoclonal antibodies against both IL-6 and CD20 and a few other targets in the inflammatory cascade. This article discusses the updated approach to diagnosis and management of CD.
Unicentric Castleman Disease
Unicentric CD is more common than MCD, presents as a localized lymph node or chain involvement, and is generally diagnosed in the third or fourth decade of life but has been reported in children. The presenting symptoms of UCD vary by site. It presents as nontender lymphadenopathy when confined to peripheral lymph nodes, whereas respiratory symptoms or bowel obstruction may be seen with lymphadenopathy in the chest/mediastinum, neck, or abdomen. The systemic
symptoms, such as fever, night sweats, and weight loss, are uncommon.
Dysplastic follicular dendritic cells characterize UCD. Histologically, it is usually classified as hyaline vascular disease with the follicles comprising small lymphocytes
and dendritic cells forming concentric rings with prominent vascularity.4,5 No association with HIV or HHV-8 has been seen.
Unicentric CD is often amenable to resection, and a complete cure can be achieved.6 Partial resection may be attempted when complete resection is not possible. Radiation therapy is offered for unresectable disease.7 In patients who are not candidates for any intervention, close long-term follow-up is recommended unless patients are symptomatic, in which case systemic treatment should be considered.
Multicentric Castleman Disease
The more widespread MCD is generally diagnosed in the fifth or sixth decade of life. It is more aggressive than UCD and presents a wide spectrum of symptoms and abnormal laboratory findings (Table 2).8
It is histologically classified into (a) plasmablastic or HHV-8 associated: It is often seen in patients with MCD infected with HIV, which can give rise to large B-cell lymphoma, known as HHV-8 plasmablastic lymphoma9; (b) plasmacytic variant has marked paracortical plasmacytosis with retained nodal architecture10; and (c) mixed MCD has abundant plasma cells with features similar to those of the hyaline-vascular variant.
Most patients with HIV-associated MCD are co-infected with HHV-8. The HHV-8 infection is also present in about 50% of HIV-negative cases.11 The incidence of HIVassociated MCD is increasing in the highly active antiretroviral therapy (HAART) era secondary to improved survival of patients infected with HIV.12 To diagnose active HIV MCD, the French Agence Nationale de Recherche sur le SIDA 117 CastlemaB trial group has described criteria based on the clinical symptoms, including fever, a raised serum C-reactive protein > 20 mg/L without any other cause, and 3 of 12 additional clinical findings described as peripheral lymphadenopathy, splenomegaly, ascites, edema, pleural effusion, cough, nasal obstruction, xerostomia, rash, central neurologic symptoms, jaundice, and autoimmune hemolytic anemia.13 The reported 2-year survival of patients who are HIV-negative is 97% compared with HIV-positive cases at 67%.14
Idiopathic MCD is diagnosed when there is no evidence of any underlying infectious, autoimmune, and neoplastic process.15
Patients with MCD are at an increased risk of developing non-Hodgkin and Hodgkin lymphoma, Kaposi sarcoma, primary effusion lymphoma, and follicular dendritic
cell sarcoma. POEMS (peripheral neuropathy, organomegaly, endocrinopathy, monoclonal protein, skin changes) syndrome and paraneoplastic disease, such as paraneoplastic pemphigus myasthenia gravis, may be commonly diagnosed concurrently or sequentially with MCD.16-20
The disease course of MCD ranges from indolent to rapidly progressive, and its 5-year OS is about 65%. When associated with POEMS syndrome, the 5-year survival was estimated to be 90% with the osteosclerotic variant and 27% without osteosclerotic lesions.3 Treatment options for MCD include systemic chemotherapy, including antiviral therapy for HHV-8 positive and HAART for HIV positive and newer monoclonal antibody therapies targeting CD20 or IL-6.
Pathophysiology
Interleukin-6 plays an important role for inflammation in both UCD and MCD (Figure 1). There is dysregulation and overproduction of IL-6, which further stimulates the production of acute-phase reactants, resulting in various systemic manifestations.15,21,22 There is increased expression of IL-1 and IL-6, upregulation of IL-6 secondary to interaction of IL-1 with nuclear factor-kappa B (NF-kappa B), thus stimulating B-cell proliferation. IL-6 binding to IL-6 receptor (IL6-R) results in downstream activation of transcription Janus kinases/signal transducers and activators of the transcription pathway. This promotes the transcription of genes encoding the acute-phase reactant proteins. Hence, interfering with IL-6 transduction by blocking downstream signals are potential therapeutic targets. The mitogen-activated protein kinase cascade, the rapidly accelerated fibrosarcoma kinases, and the overexpression of the endothelial growth factor receptor (EGFR), all contribute to disease pathogenesis by promoting increased B-cell proliferation and vascular EGFR mediated angiogenesis. 23,24
In HHV-8–associated MCD, the virus replicates within lymph node plasmablasts, causing increased production of viral IL-6 analog, human IL-6, and other proinflammatory proteins resulting in B-cell and plasma-cell proliferation, increased vascular endothelial growth factor secretion and angiogenesis.25,26 The HHV-8–infected plasmablasts are marked by variable expression of CD20, and therefore, anti-CD20 is also shown to be an effective treatment. The calmodulin/calcineurin nuclear factor assists in the proliferation of HHV-8, thereby making calcineurin another potential target for the antiviral proliferation.27
Staging
The treatment decisions and prognosis for patients with CD is based on the clinical and histologic staging. The initial workup includes but is not limited to routine laboratory evaluation, imaging, and HIV and HHV-8 testing (Table 3). Routine tests of the levels of cytokines are not recommended. Other relevant tests for known disease associations should be obtained when relevant.
Treatment
Better understanding of the disease process in CD has helped to identify potential therapeutic targets (Figures 2 and 3).
For UCD, surgery is the mainstay of treatment.4,28,29 In surgically unresectable cases, radiation therapy is helpful for local disease control. Alternatively, neoadjuvant
chemotherapy and rituximab are used. Corticosteroids are generally used to treat acute exacerbations and as adjuncts to chemotherapy.
For MCD, the treatment approach depends on the HIV and HHV-8 status of the patient. For patients with HHV-8 infection, both with and without HIV co-infection, antiviral agents, such as ganciclovir, foscarnet, or cidofovir, have shown in vitro activity against HHV-8 but with limited clinical success.30 In patients infected with HIV, the aim of treating with HAART is to control the disease, prevent opportunistic infections, and improve tolerance to chemotherapy.31-33 Rituximab with or without chemotherapy is the standard treatment approach. The additional chemotherapeutic agents are used depending on the presence or absence of organ failure. This approach has improved the OS in HIV-associated MCD.34,35 Treatment with HAART does not decrease the risk of relapse in HIV MCD; therefore, the role of rituximab and antiherpesvirus agents as maintenance therapy has been explored.36 In patients who fail to respond to or relapse rapidly following rituximab monotherapy, the use of either single-agent chemotherapy with or without rituximab or antiherpesvirus therapy with high-dose zidovudine and valganciclovir is recommended.37
The cytotoxic chemotherapy with single agents, such as etoposide, vinblastine, cyclophosphamide, cladribine, chlorambucil, and liposomal doxorubicin, has been used with limited success.22 The combination chemotherapy with cyclophosphamide/doxorubicin/vincristine/prednisone (CHOP) or cyclophosphamide/vincristine/
prednisone (CVP) without rituximab has been shown to achieve durable remissions. Corticosteroids are usually administered as an initial adjunct to chemotherapy or for acute exacerbations. In patients with MCD, regardless of HIV status, the interferon therapy was found to achieve long-term remission.38,39 The interferon therapy
exerts antiviral effects via downregulation of the IL-6R and inhibition of HHV-8 replication. For patients in remission, maintenance therapy with oral valganciclovir is promising.40
Immunomodulators & Targeted Therapies
For unresectable UCD or MCD with organ failure or relapse, the use of alternativesingle-agent or combination chemotherapies with or without rituximab is recommended. Thalidomide has shown some success, probably secondary to disruption of IL-6 production.41 In cases of progression following second-line therapy, bortezomib, antiherpesvirus therapies, or IL-6–directed therapy with siltuximab or tocilizumab should be considered.
Rituximab is a monoclonal chimeric antibody that targets CD20 on B cells, thus leading to B-cell lymphodepletion via activating complement-dependent cytotoxicity and antibody-dependent cell-mediated cytotoxicity. As monotherapy, it has been shown to achieve 2-year progression-free survival in 80% of patients.42 In patients with MCD who are HIV positive, rituximab with and without chemotherapy has shown improved overall and disease-free survival of 70% to 80% at 2 years.43
Siltuximab is a chimeric human-mouse monoclonal antibody to IL-6 that has been approved for treatment of patients with MCD who are both HIV negative and HHV-8 negative.44-46 Tocilizumab targets the IL-6R. The antibody has shown improvement in a study in HIVseronegative adults with MCD.47,48
Bortezomib is a proteasome inhibitor that inhibits the NF-kappa B pathway, which induces the expression of numerous proinflammatory proteins, including IL-6. It is recommended for relapsed or refractory disease.49,50
Anakinra is a recombinant IL-1R antagonist that blocks IL-1 effects and controls disease by decreasing IL-6 production.51
Conculsion
There has been significant progress in disease diagnosis and management as more information is available about the incidence, clinical presentation, and pathophysiology of CD. The understanding of the disease pathogenesis and biology has helped to discover multiple potential therapeutic targets. Successful treatment has been achieved through targeting HHV-8 replication, CD20, and IL-6 and anti– IL-6R antibodies. Although surgical resection continues to be the
standard of therapy for UCD, the management of MCD and relapsed or refractory disease continues to evolve. Exploration of various treatment strategies in different clinical presentations is warranted.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S.
Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information
for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to
patients.
Click here to read the digital edition.
Castleman disease (CD) is a rare nonclonal lymphoproliferative disorder, also known as angiofollicular lymph-node hyperplasia or giant node hyperplasia. It was first reported in 1954 and in 1956 described by Benjamin Castleman, MD, in a case series of localized mediastinal lymph-node hyperplasia.1 Unicentric Castleman (UCD) disease presents as a localized disease affecting a single lymph node/lymph node chain. Multicentric Castleman disease (MCD) is a more widespread or generalized disease (Table 1). About 4,000 to 6,000 new cases of CD are diagnosed per year of which about 20% to 25% cases are MCD. The estimated incidence rate for CD has recently been calculated as 21 to 25 per million person-years, or about 6,000 new cases annually.2
The clinical presentation of CD often overlaps with autoimmune, infectious, or other malignant diseases. The diagnosis is confirmed by a biopsy of the affected lymph-node tissue. Interleukin-6 (IL-6) and a viral analog of IL-6 play major role in the pathogenesis by stimulating a widespread inflammatory response that results in systemic manifestations. It is often associated with HIV and human herpesvirus-8 (HHV-8) infections. Castleman disease is histologically characterized into the hyaline vascular variant, the plasma-cell variant, and the mixed form. The plasmablastic variety is associated with HIV and HHV-8 infections. The prognosis ranges from good in UCD (91% overall survival [OS] at 5 y) to poor in MCD (65% OS at 5 y).3
Treatment options range from local surgical excision to systemic treatments. Newer therapies include monoclonal antibodies against both IL-6 and CD20 and a few other targets in the inflammatory cascade. This article discusses the updated approach to diagnosis and management of CD.
Unicentric Castleman Disease
Unicentric CD is more common than MCD, presents as a localized lymph node or chain involvement, and is generally diagnosed in the third or fourth decade of life but has been reported in children. The presenting symptoms of UCD vary by site. It presents as nontender lymphadenopathy when confined to peripheral lymph nodes, whereas respiratory symptoms or bowel obstruction may be seen with lymphadenopathy in the chest/mediastinum, neck, or abdomen. The systemic
symptoms, such as fever, night sweats, and weight loss, are uncommon.
Dysplastic follicular dendritic cells characterize UCD. Histologically, it is usually classified as hyaline vascular disease with the follicles comprising small lymphocytes
and dendritic cells forming concentric rings with prominent vascularity.4,5 No association with HIV or HHV-8 has been seen.
Unicentric CD is often amenable to resection, and a complete cure can be achieved.6 Partial resection may be attempted when complete resection is not possible. Radiation therapy is offered for unresectable disease.7 In patients who are not candidates for any intervention, close long-term follow-up is recommended unless patients are symptomatic, in which case systemic treatment should be considered.
Multicentric Castleman Disease
The more widespread MCD is generally diagnosed in the fifth or sixth decade of life. It is more aggressive than UCD and presents a wide spectrum of symptoms and abnormal laboratory findings (Table 2).8
It is histologically classified into (a) plasmablastic or HHV-8 associated: It is often seen in patients with MCD infected with HIV, which can give rise to large B-cell lymphoma, known as HHV-8 plasmablastic lymphoma9; (b) plasmacytic variant has marked paracortical plasmacytosis with retained nodal architecture10; and (c) mixed MCD has abundant plasma cells with features similar to those of the hyaline-vascular variant.
Most patients with HIV-associated MCD are co-infected with HHV-8. The HHV-8 infection is also present in about 50% of HIV-negative cases.11 The incidence of HIVassociated MCD is increasing in the highly active antiretroviral therapy (HAART) era secondary to improved survival of patients infected with HIV.12 To diagnose active HIV MCD, the French Agence Nationale de Recherche sur le SIDA 117 CastlemaB trial group has described criteria based on the clinical symptoms, including fever, a raised serum C-reactive protein > 20 mg/L without any other cause, and 3 of 12 additional clinical findings described as peripheral lymphadenopathy, splenomegaly, ascites, edema, pleural effusion, cough, nasal obstruction, xerostomia, rash, central neurologic symptoms, jaundice, and autoimmune hemolytic anemia.13 The reported 2-year survival of patients who are HIV-negative is 97% compared with HIV-positive cases at 67%.14
Idiopathic MCD is diagnosed when there is no evidence of any underlying infectious, autoimmune, and neoplastic process.15
Patients with MCD are at an increased risk of developing non-Hodgkin and Hodgkin lymphoma, Kaposi sarcoma, primary effusion lymphoma, and follicular dendritic
cell sarcoma. POEMS (peripheral neuropathy, organomegaly, endocrinopathy, monoclonal protein, skin changes) syndrome and paraneoplastic disease, such as paraneoplastic pemphigus myasthenia gravis, may be commonly diagnosed concurrently or sequentially with MCD.16-20
The disease course of MCD ranges from indolent to rapidly progressive, and its 5-year OS is about 65%. When associated with POEMS syndrome, the 5-year survival was estimated to be 90% with the osteosclerotic variant and 27% without osteosclerotic lesions.3 Treatment options for MCD include systemic chemotherapy, including antiviral therapy for HHV-8 positive and HAART for HIV positive and newer monoclonal antibody therapies targeting CD20 or IL-6.
Pathophysiology
Interleukin-6 plays an important role for inflammation in both UCD and MCD (Figure 1). There is dysregulation and overproduction of IL-6, which further stimulates the production of acute-phase reactants, resulting in various systemic manifestations.15,21,22 There is increased expression of IL-1 and IL-6, upregulation of IL-6 secondary to interaction of IL-1 with nuclear factor-kappa B (NF-kappa B), thus stimulating B-cell proliferation. IL-6 binding to IL-6 receptor (IL6-R) results in downstream activation of transcription Janus kinases/signal transducers and activators of the transcription pathway. This promotes the transcription of genes encoding the acute-phase reactant proteins. Hence, interfering with IL-6 transduction by blocking downstream signals are potential therapeutic targets. The mitogen-activated protein kinase cascade, the rapidly accelerated fibrosarcoma kinases, and the overexpression of the endothelial growth factor receptor (EGFR), all contribute to disease pathogenesis by promoting increased B-cell proliferation and vascular EGFR mediated angiogenesis. 23,24
In HHV-8–associated MCD, the virus replicates within lymph node plasmablasts, causing increased production of viral IL-6 analog, human IL-6, and other proinflammatory proteins resulting in B-cell and plasma-cell proliferation, increased vascular endothelial growth factor secretion and angiogenesis.25,26 The HHV-8–infected plasmablasts are marked by variable expression of CD20, and therefore, anti-CD20 is also shown to be an effective treatment. The calmodulin/calcineurin nuclear factor assists in the proliferation of HHV-8, thereby making calcineurin another potential target for the antiviral proliferation.27
Staging
The treatment decisions and prognosis for patients with CD is based on the clinical and histologic staging. The initial workup includes but is not limited to routine laboratory evaluation, imaging, and HIV and HHV-8 testing (Table 3). Routine tests of the levels of cytokines are not recommended. Other relevant tests for known disease associations should be obtained when relevant.
Treatment
Better understanding of the disease process in CD has helped to identify potential therapeutic targets (Figures 2 and 3).
For UCD, surgery is the mainstay of treatment.4,28,29 In surgically unresectable cases, radiation therapy is helpful for local disease control. Alternatively, neoadjuvant
chemotherapy and rituximab are used. Corticosteroids are generally used to treat acute exacerbations and as adjuncts to chemotherapy.
For MCD, the treatment approach depends on the HIV and HHV-8 status of the patient. For patients with HHV-8 infection, both with and without HIV co-infection, antiviral agents, such as ganciclovir, foscarnet, or cidofovir, have shown in vitro activity against HHV-8 but with limited clinical success.30 In patients infected with HIV, the aim of treating with HAART is to control the disease, prevent opportunistic infections, and improve tolerance to chemotherapy.31-33 Rituximab with or without chemotherapy is the standard treatment approach. The additional chemotherapeutic agents are used depending on the presence or absence of organ failure. This approach has improved the OS in HIV-associated MCD.34,35 Treatment with HAART does not decrease the risk of relapse in HIV MCD; therefore, the role of rituximab and antiherpesvirus agents as maintenance therapy has been explored.36 In patients who fail to respond to or relapse rapidly following rituximab monotherapy, the use of either single-agent chemotherapy with or without rituximab or antiherpesvirus therapy with high-dose zidovudine and valganciclovir is recommended.37
The cytotoxic chemotherapy with single agents, such as etoposide, vinblastine, cyclophosphamide, cladribine, chlorambucil, and liposomal doxorubicin, has been used with limited success.22 The combination chemotherapy with cyclophosphamide/doxorubicin/vincristine/prednisone (CHOP) or cyclophosphamide/vincristine/
prednisone (CVP) without rituximab has been shown to achieve durable remissions. Corticosteroids are usually administered as an initial adjunct to chemotherapy or for acute exacerbations. In patients with MCD, regardless of HIV status, the interferon therapy was found to achieve long-term remission.38,39 The interferon therapy
exerts antiviral effects via downregulation of the IL-6R and inhibition of HHV-8 replication. For patients in remission, maintenance therapy with oral valganciclovir is promising.40
Immunomodulators & Targeted Therapies
For unresectable UCD or MCD with organ failure or relapse, the use of alternativesingle-agent or combination chemotherapies with or without rituximab is recommended. Thalidomide has shown some success, probably secondary to disruption of IL-6 production.41 In cases of progression following second-line therapy, bortezomib, antiherpesvirus therapies, or IL-6–directed therapy with siltuximab or tocilizumab should be considered.
Rituximab is a monoclonal chimeric antibody that targets CD20 on B cells, thus leading to B-cell lymphodepletion via activating complement-dependent cytotoxicity and antibody-dependent cell-mediated cytotoxicity. As monotherapy, it has been shown to achieve 2-year progression-free survival in 80% of patients.42 In patients with MCD who are HIV positive, rituximab with and without chemotherapy has shown improved overall and disease-free survival of 70% to 80% at 2 years.43
Siltuximab is a chimeric human-mouse monoclonal antibody to IL-6 that has been approved for treatment of patients with MCD who are both HIV negative and HHV-8 negative.44-46 Tocilizumab targets the IL-6R. The antibody has shown improvement in a study in HIVseronegative adults with MCD.47,48
Bortezomib is a proteasome inhibitor that inhibits the NF-kappa B pathway, which induces the expression of numerous proinflammatory proteins, including IL-6. It is recommended for relapsed or refractory disease.49,50
Anakinra is a recombinant IL-1R antagonist that blocks IL-1 effects and controls disease by decreasing IL-6 production.51
Conculsion
There has been significant progress in disease diagnosis and management as more information is available about the incidence, clinical presentation, and pathophysiology of CD. The understanding of the disease pathogenesis and biology has helped to discover multiple potential therapeutic targets. Successful treatment has been achieved through targeting HHV-8 replication, CD20, and IL-6 and anti– IL-6R antibodies. Although surgical resection continues to be the
standard of therapy for UCD, the management of MCD and relapsed or refractory disease continues to evolve. Exploration of various treatment strategies in different clinical presentations is warranted.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S.
Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information
for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to
patients.
Click here to read the digital edition.
1. Castleman B, Iverson L, Menendez VP. Localized mediastinal lymph-node hyperplasia
resembling thymoma. Cancer. 1956;9(4):822-830.
2. Munshi N, Mehra M, van de Velde H, Desai A, Potluri R, Vermeulen J. Use of a claims database to characterize and estimate the incidence rate for Castleman disease. Leuk Lymphoma. 2015;56(5):1252-1260.
3. Dispenzieri A, Armitage JO, Loe MJ, et al. The clinical spectrum of Castleman’s disease. Am J Hematol. 2012;87(11):997-1002.
4. Keller AR, Hochholzer L, Castleman B. Hyaline-vascular and plasma-cell types of giant lymph node hyperplasia of the mediastinum and other locations. Cancer. 1972;29(3):670-683.
5. Cronin DM, Warnke RA. Castleman disease: an update on classification and the
spectrum of associated lesions. Adv Anat Pathol. 2009;16(4):236-246.
6. Talat N, Belgaumkar AP, Schulte KM. Surgery in Castleman’s disease: a systematic review of 404 published cases. Ann Surg. 2012;255(4):677-684.
7. Chronowski GM, Ha CS, Wilder RB, Cabanillas F, Manning J, Cox JD. Treatment of unicentric and multicentric Castleman disease and the role of radiotherapy. Cancer. 2001;92(3):670-676.
8. Herrada J, Cabanillas F, Rice L, Manning J, Pugh W. The clinical behavior of localized and multicentric Castleman disease. Ann Intern Med. 1998;128(8):657-662.
9. Dupin N, Diss TL, Kellam P, et al. HHV-8 is associated with a plasmablastic variant of Castleman disease that is linked to HHV-8-positive plasmablastic lymphoma. Blood. 2000;95(4):1406-1412.
10. Ferry JA, Harris NL. Atlas of Lymphoid Hyperplasia and Lymphoma. Philadelphia, PA: W.B. Saunders; 1997.
11. Soulier J, Grollet L, Oksenhendler E, et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood. 1995;86(4):1276-1280.
12. Powles T, Stebbing J, Bazeos A, et al. The role of immune suppression and HHV-8 in the increasing incidence of HIV-associated multicentric Castleman’s disease. Ann Oncol. 2009;20(4):775-779.
13. Gérard L, Bérezné A, Galicier L, et al. Prospective study of rituximab in chemotherapy-dependent human immunodeficiency virus associated multicentric Castleman’s disease: ANRS 117 CastlemaB Trial. J Clin Oncol. 2007;25(22):3350-3356.
14. Casper C, Teltsch DY, Robinson D Jr, et al. Clinical characteristics and healthcare utilization of patients with multicentric Castleman disease. Br J Haematol. 2015;168(1):82-93.
15. Fajgenbaum DC, van Rhee F, Nabel CS. HHV-8-negative, idiopathic multicentric Castleman disease: novel insights into biology, pathogenesis, and therapy. Blood. 2014;123(19):2924-2933.
16. Larroche C, Cacoub P, Soulier J, et al. Castleman’s disease and lymphoma: report of eight cases in HIV-negative patients and literature review. Am J Hematol. 2002;69(2):119-126.
17. Dispenzieri A. POEMS syndrome: 2014 update on diagnosis, risk-stratification, and management. Am J Hematol. 2014;89(2):214-223.
18. Andhavarapu S, Jiang L. POEMS syndrome and Castleman disease. Blood. 2013;122(2):159.
19. Bélec L, Mohamed AS, Authier FJ, et al. Human herpesvirus 8 infection in patients with POEMS syndrome-associated multicentric Castleman’s disease. Blood. 1999;93(11):3643-3653.
20. Oksenhendler E, Boulanger E, Galicier L, et al. High incidence of Kaposi sarcomaassociated herpesvirus-related non-Hodgkin lymphoma in patients with HIV infection and multicentric Castleman disease. Blood. 2002;99(7):2331-2336.
21. Yoshizaki K, Matsuda T, Nishimoto N, et al. Pathogenic significance of interleukin-6 (IL-6/BSF-2) in Castleman’s disease. Blood. 1989;74(4):1360-1367.
22. El-Osta HE, Kurzrock R. Castleman’s disease: from basic mechanisms to molecular therapeutics. Oncologist. 2011;16(4):497-511.
23. Akula SM, Ford PW, Whitman AG, et al. B-Raf-dependent expression of vascular endothelial growth factor-A in Kaposi sarcoma-associated herpesvirus-infected human B cells. Blood. 2005;105(11):4516-4522.
24. Sun X, Chang KC, Abruzzo LV, Lai R, Younes A, Jones D. Epidermal growth factor receptor expression in follicular dendritic cells: a shared feature of follicular dendritic cell sarcoma and Castleman’s disease. Hum Pathol. 2003;34(9):835-840.
25. Adam N, Rabe B, Suthaus J, Grötzinger J, Rose-John S, Scheller J. Unraveling viral interleukin-6 binding to gp130 and activation of STAT-signaling pathways independently of the interleukin-6 receptor. J Virol. 2009;83(10):5117-5126.
26. Suda T, Katano H, Delsol G, et al. HHV-8 infection status of AIDS-unrelated and
AIDS-associated multicentric Castleman’s disease. Pathol Int. 2001;51(9):671-679.
27. Zoeteweij JP, Moses AV, Rinderknecht AS, et al. Targeted inhibition of calcineurin signaling blocks calcium-dependent reactivation of Kaposi sarcoma-associated herpesvirus. Blood. 2001;97(8):2374-2380.
28. McCarty MJ, Vukelja SJ, Banks PM, Weiss RB. Angiofollicular lymph node hyperplasia
(Castleman’s disease). Cancer Treat Rev. 1995;21(4):291-310.
29. Bowne WB, Lewis JJ, Filippa DA, et al. The management of unicentric and multicentric Castleman’s disease: a report of 16 cases and a review of the literature. Cancer. 1999;85(3):706-717.
30. Reddy D, Mitsuyasu R. HIV-associated multicentric Castleman disease. Curr Opin Oncol. 2011;23(5):475-481.
31. Aaron L, Lidove O, Yousry C, Roudiere L, Dupont B, Viard JP. Human herpesvirus 8-positive Castleman disease in human immunodeficiency virus-infected patients: the impact of highly active antiretroviral therapy. Clin Infect Dis. 2002;35(7):880-882.
32. Sprinz E, Jeffman M, Liedke P, Putten A, Schwartsmann G. Successful treatment of AIDS-related Castleman’s disease following the administration of highly active antiretroviral therapy (HAART). Ann Oncol. 2004;15(2):356-358.
33. Lee SM, Edwards SG, Chilton DN, Ramsay A, Miller RF. Highly active antiretroviral therapy alone may be an effective treatment for HIV-associated multi-centric Castleman’s disease. Haematologica. 2010;95(11):1979-1981.
34. Bower M. How I treat HIV-associated multicentric Castleman disease. Blood. 2010;116(22):4415-4421.
35. Bower M, Newsom-Davis T, Naresh K, et al. Clinical features and outcome in HIVassociated
multicentric Castleman’s disease. J Clin Oncol. 2011;29(18):2481-2486.
36. Casper C, Nichols WG, Huang ML, Corey L, Wald A. Remission of HHV-8 and HIV-associated multicentric Castleman disease with ganciclovir treatment. Blood. 2004;103(5):1632-1634.
37. Uldrick TS, Polizzotto MN, Aleman K, et al. High-dose zidovudine plus valganciclovir for Kaposi sarcoma herpesvirus-associated multicentric Castleman disease: a pilot study of virus-activated cytotoxic therapy. Blood. 2011;117(26):6977-6986.
38. Kumari P, Schechter GP, Saini N, Benator DA. Successful treatment of human immunodeficiency virus-related Castleman’s disease with interferon-alpha. Clin Infect Dis. 2000;31(2):602-604.
39. Nord JA, Karter D. Low dose interferon-alpha therapy for HIV-associated multicentric Castleman’s disease. Int J STD AIDS. 2003;14(1):61-62.
40. Oksenhendler E. HIV-associated multicentric Castleman disease. Curr Opin HIV AIDS. 2009;4(1):16-21.
41. Jung CP, Emmerich B, Goebel FD, Bogner JR. Successful treatment of a patient with HIV-associated multicentric Castleman disease (MCD) with thalidomide. Am J Hematol. 2004;75(3):176-177.
42. Ide M, Kawachi Y, Izumi Y, Kasagi K, Ogino T. Long-term remission in HIV negative patients with multicentric Castleman’s disease using rituximab. Eur J Haematol. 2006;76(2):119-123.
43. Marcelin AG, Aaron L, Mateus C, et al. Rituximab therapy for HIV-associated Castleman disease. Blood. 2003;102(8):2786-2788.
44. Van Rhee F, Fayad L, Voorhees P, et al. Siltuximab, a novel anti-interleukin-6 monoclonal antibody, for Castleman’s disease. J Clin Oncol. 2010;28(23):3701-3708.
45. Wong RS, Casper C, Munshi N, et al. A multicenter, randomized, doubleblind, placebo-controlled study of the efficacy and safety of siltuximab, an antiinterleukin-6 monoclonal antibody, in patients with multicentric Castleman’s disease. Blood. 2013;122(21):505.
46. Van Rhee F, Casper C, Voorhees PM, et al. An open-label, phase 2, multicenter study of the safety of long-term treatment with siltuximab (an anti-interleukin-6 monoclonal antibody) in patients with multicentric Castleman’s disease. Blood. 2013;122(21):1806.
47. Nishimoto N, Kanakura Y, Aozasa K, et al. Humanized anti-interleukin-6 receptor antibody treatment of multicentric Castleman disease. Blood. 2005;106(8):2627-2632.
48. Müzes G, Sipos F, Csomor J, Sréter L. Successful tocilizumab treatment in a patient with human herpesvirus 8-positive and human immunodeficiency virusnegative multicentric Castleman’s disease of plasma cell type nonresponsive to rituximab-CVP therapy. APMIS. 2013;121(7):668-674.
49. Hess G, Wagner V, Kreft A, Heussel CP, Huber C. Effects of bortezomib on proinflammatory cytokine levels and transfusion dependency in a patient with multicentric Castleman disease. Br J Haematol. 2006;134(5):544-545.
50. Sobas MA, Alonso Vence N, Diaz Arias J, Bendaña Lopez A, Fraga Rodriguez M, Bello Lopez JL. Efficacy of bortezomib in refractory form of multicentric Castleman disease associated to poems syndrome (MCD-POEMS variant). Ann Hematol. 2010;89(2):217-219.
51. El-Osta H, Janku F, Kurzrock R. Successful treatment of Castleman’s disease with interleukin-1 receptor antagonist (Anakinra). Mol Cancer Ther. 2010;9(6):1485-1488.
1. Castleman B, Iverson L, Menendez VP. Localized mediastinal lymph-node hyperplasia
resembling thymoma. Cancer. 1956;9(4):822-830.
2. Munshi N, Mehra M, van de Velde H, Desai A, Potluri R, Vermeulen J. Use of a claims database to characterize and estimate the incidence rate for Castleman disease. Leuk Lymphoma. 2015;56(5):1252-1260.
3. Dispenzieri A, Armitage JO, Loe MJ, et al. The clinical spectrum of Castleman’s disease. Am J Hematol. 2012;87(11):997-1002.
4. Keller AR, Hochholzer L, Castleman B. Hyaline-vascular and plasma-cell types of giant lymph node hyperplasia of the mediastinum and other locations. Cancer. 1972;29(3):670-683.
5. Cronin DM, Warnke RA. Castleman disease: an update on classification and the
spectrum of associated lesions. Adv Anat Pathol. 2009;16(4):236-246.
6. Talat N, Belgaumkar AP, Schulte KM. Surgery in Castleman’s disease: a systematic review of 404 published cases. Ann Surg. 2012;255(4):677-684.
7. Chronowski GM, Ha CS, Wilder RB, Cabanillas F, Manning J, Cox JD. Treatment of unicentric and multicentric Castleman disease and the role of radiotherapy. Cancer. 2001;92(3):670-676.
8. Herrada J, Cabanillas F, Rice L, Manning J, Pugh W. The clinical behavior of localized and multicentric Castleman disease. Ann Intern Med. 1998;128(8):657-662.
9. Dupin N, Diss TL, Kellam P, et al. HHV-8 is associated with a plasmablastic variant of Castleman disease that is linked to HHV-8-positive plasmablastic lymphoma. Blood. 2000;95(4):1406-1412.
10. Ferry JA, Harris NL. Atlas of Lymphoid Hyperplasia and Lymphoma. Philadelphia, PA: W.B. Saunders; 1997.
11. Soulier J, Grollet L, Oksenhendler E, et al. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood. 1995;86(4):1276-1280.
12. Powles T, Stebbing J, Bazeos A, et al. The role of immune suppression and HHV-8 in the increasing incidence of HIV-associated multicentric Castleman’s disease. Ann Oncol. 2009;20(4):775-779.
13. Gérard L, Bérezné A, Galicier L, et al. Prospective study of rituximab in chemotherapy-dependent human immunodeficiency virus associated multicentric Castleman’s disease: ANRS 117 CastlemaB Trial. J Clin Oncol. 2007;25(22):3350-3356.
14. Casper C, Teltsch DY, Robinson D Jr, et al. Clinical characteristics and healthcare utilization of patients with multicentric Castleman disease. Br J Haematol. 2015;168(1):82-93.
15. Fajgenbaum DC, van Rhee F, Nabel CS. HHV-8-negative, idiopathic multicentric Castleman disease: novel insights into biology, pathogenesis, and therapy. Blood. 2014;123(19):2924-2933.
16. Larroche C, Cacoub P, Soulier J, et al. Castleman’s disease and lymphoma: report of eight cases in HIV-negative patients and literature review. Am J Hematol. 2002;69(2):119-126.
17. Dispenzieri A. POEMS syndrome: 2014 update on diagnosis, risk-stratification, and management. Am J Hematol. 2014;89(2):214-223.
18. Andhavarapu S, Jiang L. POEMS syndrome and Castleman disease. Blood. 2013;122(2):159.
19. Bélec L, Mohamed AS, Authier FJ, et al. Human herpesvirus 8 infection in patients with POEMS syndrome-associated multicentric Castleman’s disease. Blood. 1999;93(11):3643-3653.
20. Oksenhendler E, Boulanger E, Galicier L, et al. High incidence of Kaposi sarcomaassociated herpesvirus-related non-Hodgkin lymphoma in patients with HIV infection and multicentric Castleman disease. Blood. 2002;99(7):2331-2336.
21. Yoshizaki K, Matsuda T, Nishimoto N, et al. Pathogenic significance of interleukin-6 (IL-6/BSF-2) in Castleman’s disease. Blood. 1989;74(4):1360-1367.
22. El-Osta HE, Kurzrock R. Castleman’s disease: from basic mechanisms to molecular therapeutics. Oncologist. 2011;16(4):497-511.
23. Akula SM, Ford PW, Whitman AG, et al. B-Raf-dependent expression of vascular endothelial growth factor-A in Kaposi sarcoma-associated herpesvirus-infected human B cells. Blood. 2005;105(11):4516-4522.
24. Sun X, Chang KC, Abruzzo LV, Lai R, Younes A, Jones D. Epidermal growth factor receptor expression in follicular dendritic cells: a shared feature of follicular dendritic cell sarcoma and Castleman’s disease. Hum Pathol. 2003;34(9):835-840.
25. Adam N, Rabe B, Suthaus J, Grötzinger J, Rose-John S, Scheller J. Unraveling viral interleukin-6 binding to gp130 and activation of STAT-signaling pathways independently of the interleukin-6 receptor. J Virol. 2009;83(10):5117-5126.
26. Suda T, Katano H, Delsol G, et al. HHV-8 infection status of AIDS-unrelated and
AIDS-associated multicentric Castleman’s disease. Pathol Int. 2001;51(9):671-679.
27. Zoeteweij JP, Moses AV, Rinderknecht AS, et al. Targeted inhibition of calcineurin signaling blocks calcium-dependent reactivation of Kaposi sarcoma-associated herpesvirus. Blood. 2001;97(8):2374-2380.
28. McCarty MJ, Vukelja SJ, Banks PM, Weiss RB. Angiofollicular lymph node hyperplasia
(Castleman’s disease). Cancer Treat Rev. 1995;21(4):291-310.
29. Bowne WB, Lewis JJ, Filippa DA, et al. The management of unicentric and multicentric Castleman’s disease: a report of 16 cases and a review of the literature. Cancer. 1999;85(3):706-717.
30. Reddy D, Mitsuyasu R. HIV-associated multicentric Castleman disease. Curr Opin Oncol. 2011;23(5):475-481.
31. Aaron L, Lidove O, Yousry C, Roudiere L, Dupont B, Viard JP. Human herpesvirus 8-positive Castleman disease in human immunodeficiency virus-infected patients: the impact of highly active antiretroviral therapy. Clin Infect Dis. 2002;35(7):880-882.
32. Sprinz E, Jeffman M, Liedke P, Putten A, Schwartsmann G. Successful treatment of AIDS-related Castleman’s disease following the administration of highly active antiretroviral therapy (HAART). Ann Oncol. 2004;15(2):356-358.
33. Lee SM, Edwards SG, Chilton DN, Ramsay A, Miller RF. Highly active antiretroviral therapy alone may be an effective treatment for HIV-associated multi-centric Castleman’s disease. Haematologica. 2010;95(11):1979-1981.
34. Bower M. How I treat HIV-associated multicentric Castleman disease. Blood. 2010;116(22):4415-4421.
35. Bower M, Newsom-Davis T, Naresh K, et al. Clinical features and outcome in HIVassociated
multicentric Castleman’s disease. J Clin Oncol. 2011;29(18):2481-2486.
36. Casper C, Nichols WG, Huang ML, Corey L, Wald A. Remission of HHV-8 and HIV-associated multicentric Castleman disease with ganciclovir treatment. Blood. 2004;103(5):1632-1634.
37. Uldrick TS, Polizzotto MN, Aleman K, et al. High-dose zidovudine plus valganciclovir for Kaposi sarcoma herpesvirus-associated multicentric Castleman disease: a pilot study of virus-activated cytotoxic therapy. Blood. 2011;117(26):6977-6986.
38. Kumari P, Schechter GP, Saini N, Benator DA. Successful treatment of human immunodeficiency virus-related Castleman’s disease with interferon-alpha. Clin Infect Dis. 2000;31(2):602-604.
39. Nord JA, Karter D. Low dose interferon-alpha therapy for HIV-associated multicentric Castleman’s disease. Int J STD AIDS. 2003;14(1):61-62.
40. Oksenhendler E. HIV-associated multicentric Castleman disease. Curr Opin HIV AIDS. 2009;4(1):16-21.
41. Jung CP, Emmerich B, Goebel FD, Bogner JR. Successful treatment of a patient with HIV-associated multicentric Castleman disease (MCD) with thalidomide. Am J Hematol. 2004;75(3):176-177.
42. Ide M, Kawachi Y, Izumi Y, Kasagi K, Ogino T. Long-term remission in HIV negative patients with multicentric Castleman’s disease using rituximab. Eur J Haematol. 2006;76(2):119-123.
43. Marcelin AG, Aaron L, Mateus C, et al. Rituximab therapy for HIV-associated Castleman disease. Blood. 2003;102(8):2786-2788.
44. Van Rhee F, Fayad L, Voorhees P, et al. Siltuximab, a novel anti-interleukin-6 monoclonal antibody, for Castleman’s disease. J Clin Oncol. 2010;28(23):3701-3708.
45. Wong RS, Casper C, Munshi N, et al. A multicenter, randomized, doubleblind, placebo-controlled study of the efficacy and safety of siltuximab, an antiinterleukin-6 monoclonal antibody, in patients with multicentric Castleman’s disease. Blood. 2013;122(21):505.
46. Van Rhee F, Casper C, Voorhees PM, et al. An open-label, phase 2, multicenter study of the safety of long-term treatment with siltuximab (an anti-interleukin-6 monoclonal antibody) in patients with multicentric Castleman’s disease. Blood. 2013;122(21):1806.
47. Nishimoto N, Kanakura Y, Aozasa K, et al. Humanized anti-interleukin-6 receptor antibody treatment of multicentric Castleman disease. Blood. 2005;106(8):2627-2632.
48. Müzes G, Sipos F, Csomor J, Sréter L. Successful tocilizumab treatment in a patient with human herpesvirus 8-positive and human immunodeficiency virusnegative multicentric Castleman’s disease of plasma cell type nonresponsive to rituximab-CVP therapy. APMIS. 2013;121(7):668-674.
49. Hess G, Wagner V, Kreft A, Heussel CP, Huber C. Effects of bortezomib on proinflammatory cytokine levels and transfusion dependency in a patient with multicentric Castleman disease. Br J Haematol. 2006;134(5):544-545.
50. Sobas MA, Alonso Vence N, Diaz Arias J, Bendaña Lopez A, Fraga Rodriguez M, Bello Lopez JL. Efficacy of bortezomib in refractory form of multicentric Castleman disease associated to poems syndrome (MCD-POEMS variant). Ann Hematol. 2010;89(2):217-219.
51. El-Osta H, Janku F, Kurzrock R. Successful treatment of Castleman’s disease with interleukin-1 receptor antagonist (Anakinra). Mol Cancer Ther. 2010;9(6):1485-1488.
Sequencing aids management of young cancer patients

patient and her father
Photo by Rhoda Baer
Exome and transcriptome sequencing results can inform the management of young patients with relapsed, refractory, and rare malignancies, a new study suggests.
In a consecutive case series, sequencing data revealed potentially actionable findings for 46% of patients.
As a result, 15% of patients changed treatment, and 10% underwent genetic counseling.
Investigators described this research in JAMA.
“We found that, for some children with rare, difficult-to-treat, and aggressive cancers, this technology can dramatically change the course of their treatment,” said study author Rajen Mody, MBBS, of the University of Michigan in Ann Arbor.
Dr Mody and his colleagues evaluated 102 patients with relapsed, refractory, or rare cancers. Their median age was 11.5 (range, 0-22).
The patients underwent integrative clinical exome (tumor and germline DNA) and transcriptome (tumor RNA) sequencing. Ninety-one patients (89%) had adequate tumor tissue to complete sequencing, including 28 patients (31%) with hematologic malignancies and 63 (69%) with solid tumors.
All sequencing results were discussed at a precision medicine tumor board, which included pediatric and adult oncologists, pathologists, genetics specialists, and other professionals. This group discussed the results and assessed the feasibility of pursuing treatment options based on the findings.
Actionable findings
Forty-two patients (46%) had potentially actionable findings, 15 (54%) with hematologic malignancies and 27 (43%) with solid tumors.
Actionable findings included a change in diagnosis (n=2), the presence of a genetic anomaly that could be targeted by an approved or experimental drug (n=31), and the need for genetic counseling for inherited cancer risk that could affect the patient or the whole family (n=9).
“We were excited to see an actionable finding in such a substantial percentage of patients, and we think it could potentially be higher over time,” said study author Arul Chinnaiyan, MD, PhD, also of the University of Michigan.
“These are patients who had exhausted all proven therapeutic options or who had an extremely rare diagnosis. If we can find a clinically actionable event and have a chance to act upon it, we show in this study that it can have a big impact on that patient.”
Actions were taken in 23 of the 42 patients. Fourteen patients (15%) had their treatment changed, and 9 of these patients (10%) had durable partial or complete remissions (CRs) as a result.
Nine patients (10%) underwent genetic counseling because of sequencing results. The researchers noted that 4 of these patients had no notable family history to suggest an inherited risk, and they would not otherwise have been referred for genetic counseling.
Hematologic malignancies
Fifteen patients with hematologic malignancies had potentially actionable findings, and 4 underwent treatment changes as a result. (None of the patients required genetic counseling.)
For a patient with pre-B acute lymphoblastic leukemia (ALL), sequencing revealed a homozygous CDKN2A deletion and an ETV6-ABL1 fusion. So the patient was placed on imatinib and had a sustained CR for 21 months.
A patient with early T-cell precursor ALL had a FLT3-ITD mutation, Chr16p gain, Chr16q loss, and FLT3 overexpression. The patient achieved a CR after transplant, was placed on the FLT3 inhibitor sorafenib, and remained in CR for 15 months.
Another patient with pre-B ALL had a FLT3 nonframeshift deletion and BLK and FLT3 overexpression. The patient was in CR for 9 months after a transplant and received sorafenib for 6 months.
A patient with biphenotypic leukemia had mutations in NRAS and PHF6; SPI1, ASXL1, and CBLC frameshift insertions; a JAK3-activating mutation; and JAK3 overexpression. The patient received the JAK3 inhibitor tofacitinib but could not tolerate the full dose and died of progressive disease.
Cost and turn-around time
The cost for sequencing was approximately $6000 per patient and was covered under the research protocol.
It took the researchers about 7 to 8 weeks to report the sequencing results back to treating physicians and families.
“These are early days, and the full promise of precision medicine is yet to be fully realized,” Dr Mody said. “We need better targeted therapies designed for children, and turnaround time for sequencing needs to be less than 2 weeks for it to be a regular part of a patient’s treatment plan.” ![]()

patient and her father
Photo by Rhoda Baer
Exome and transcriptome sequencing results can inform the management of young patients with relapsed, refractory, and rare malignancies, a new study suggests.
In a consecutive case series, sequencing data revealed potentially actionable findings for 46% of patients.
As a result, 15% of patients changed treatment, and 10% underwent genetic counseling.
Investigators described this research in JAMA.
“We found that, for some children with rare, difficult-to-treat, and aggressive cancers, this technology can dramatically change the course of their treatment,” said study author Rajen Mody, MBBS, of the University of Michigan in Ann Arbor.
Dr Mody and his colleagues evaluated 102 patients with relapsed, refractory, or rare cancers. Their median age was 11.5 (range, 0-22).
The patients underwent integrative clinical exome (tumor and germline DNA) and transcriptome (tumor RNA) sequencing. Ninety-one patients (89%) had adequate tumor tissue to complete sequencing, including 28 patients (31%) with hematologic malignancies and 63 (69%) with solid tumors.
All sequencing results were discussed at a precision medicine tumor board, which included pediatric and adult oncologists, pathologists, genetics specialists, and other professionals. This group discussed the results and assessed the feasibility of pursuing treatment options based on the findings.
Actionable findings
Forty-two patients (46%) had potentially actionable findings, 15 (54%) with hematologic malignancies and 27 (43%) with solid tumors.
Actionable findings included a change in diagnosis (n=2), the presence of a genetic anomaly that could be targeted by an approved or experimental drug (n=31), and the need for genetic counseling for inherited cancer risk that could affect the patient or the whole family (n=9).
“We were excited to see an actionable finding in such a substantial percentage of patients, and we think it could potentially be higher over time,” said study author Arul Chinnaiyan, MD, PhD, also of the University of Michigan.
“These are patients who had exhausted all proven therapeutic options or who had an extremely rare diagnosis. If we can find a clinically actionable event and have a chance to act upon it, we show in this study that it can have a big impact on that patient.”
Actions were taken in 23 of the 42 patients. Fourteen patients (15%) had their treatment changed, and 9 of these patients (10%) had durable partial or complete remissions (CRs) as a result.
Nine patients (10%) underwent genetic counseling because of sequencing results. The researchers noted that 4 of these patients had no notable family history to suggest an inherited risk, and they would not otherwise have been referred for genetic counseling.
Hematologic malignancies
Fifteen patients with hematologic malignancies had potentially actionable findings, and 4 underwent treatment changes as a result. (None of the patients required genetic counseling.)
For a patient with pre-B acute lymphoblastic leukemia (ALL), sequencing revealed a homozygous CDKN2A deletion and an ETV6-ABL1 fusion. So the patient was placed on imatinib and had a sustained CR for 21 months.
A patient with early T-cell precursor ALL had a FLT3-ITD mutation, Chr16p gain, Chr16q loss, and FLT3 overexpression. The patient achieved a CR after transplant, was placed on the FLT3 inhibitor sorafenib, and remained in CR for 15 months.
Another patient with pre-B ALL had a FLT3 nonframeshift deletion and BLK and FLT3 overexpression. The patient was in CR for 9 months after a transplant and received sorafenib for 6 months.
A patient with biphenotypic leukemia had mutations in NRAS and PHF6; SPI1, ASXL1, and CBLC frameshift insertions; a JAK3-activating mutation; and JAK3 overexpression. The patient received the JAK3 inhibitor tofacitinib but could not tolerate the full dose and died of progressive disease.
Cost and turn-around time
The cost for sequencing was approximately $6000 per patient and was covered under the research protocol.
It took the researchers about 7 to 8 weeks to report the sequencing results back to treating physicians and families.
“These are early days, and the full promise of precision medicine is yet to be fully realized,” Dr Mody said. “We need better targeted therapies designed for children, and turnaround time for sequencing needs to be less than 2 weeks for it to be a regular part of a patient’s treatment plan.” ![]()

patient and her father
Photo by Rhoda Baer
Exome and transcriptome sequencing results can inform the management of young patients with relapsed, refractory, and rare malignancies, a new study suggests.
In a consecutive case series, sequencing data revealed potentially actionable findings for 46% of patients.
As a result, 15% of patients changed treatment, and 10% underwent genetic counseling.
Investigators described this research in JAMA.
“We found that, for some children with rare, difficult-to-treat, and aggressive cancers, this technology can dramatically change the course of their treatment,” said study author Rajen Mody, MBBS, of the University of Michigan in Ann Arbor.
Dr Mody and his colleagues evaluated 102 patients with relapsed, refractory, or rare cancers. Their median age was 11.5 (range, 0-22).
The patients underwent integrative clinical exome (tumor and germline DNA) and transcriptome (tumor RNA) sequencing. Ninety-one patients (89%) had adequate tumor tissue to complete sequencing, including 28 patients (31%) with hematologic malignancies and 63 (69%) with solid tumors.
All sequencing results were discussed at a precision medicine tumor board, which included pediatric and adult oncologists, pathologists, genetics specialists, and other professionals. This group discussed the results and assessed the feasibility of pursuing treatment options based on the findings.
Actionable findings
Forty-two patients (46%) had potentially actionable findings, 15 (54%) with hematologic malignancies and 27 (43%) with solid tumors.
Actionable findings included a change in diagnosis (n=2), the presence of a genetic anomaly that could be targeted by an approved or experimental drug (n=31), and the need for genetic counseling for inherited cancer risk that could affect the patient or the whole family (n=9).
“We were excited to see an actionable finding in such a substantial percentage of patients, and we think it could potentially be higher over time,” said study author Arul Chinnaiyan, MD, PhD, also of the University of Michigan.
“These are patients who had exhausted all proven therapeutic options or who had an extremely rare diagnosis. If we can find a clinically actionable event and have a chance to act upon it, we show in this study that it can have a big impact on that patient.”
Actions were taken in 23 of the 42 patients. Fourteen patients (15%) had their treatment changed, and 9 of these patients (10%) had durable partial or complete remissions (CRs) as a result.
Nine patients (10%) underwent genetic counseling because of sequencing results. The researchers noted that 4 of these patients had no notable family history to suggest an inherited risk, and they would not otherwise have been referred for genetic counseling.
Hematologic malignancies
Fifteen patients with hematologic malignancies had potentially actionable findings, and 4 underwent treatment changes as a result. (None of the patients required genetic counseling.)
For a patient with pre-B acute lymphoblastic leukemia (ALL), sequencing revealed a homozygous CDKN2A deletion and an ETV6-ABL1 fusion. So the patient was placed on imatinib and had a sustained CR for 21 months.
A patient with early T-cell precursor ALL had a FLT3-ITD mutation, Chr16p gain, Chr16q loss, and FLT3 overexpression. The patient achieved a CR after transplant, was placed on the FLT3 inhibitor sorafenib, and remained in CR for 15 months.
Another patient with pre-B ALL had a FLT3 nonframeshift deletion and BLK and FLT3 overexpression. The patient was in CR for 9 months after a transplant and received sorafenib for 6 months.
A patient with biphenotypic leukemia had mutations in NRAS and PHF6; SPI1, ASXL1, and CBLC frameshift insertions; a JAK3-activating mutation; and JAK3 overexpression. The patient received the JAK3 inhibitor tofacitinib but could not tolerate the full dose and died of progressive disease.
Cost and turn-around time
The cost for sequencing was approximately $6000 per patient and was covered under the research protocol.
It took the researchers about 7 to 8 weeks to report the sequencing results back to treating physicians and families.
“These are early days, and the full promise of precision medicine is yet to be fully realized,” Dr Mody said. “We need better targeted therapies designed for children, and turnaround time for sequencing needs to be less than 2 weeks for it to be a regular part of a patient’s treatment plan.” ![]()
A new, improved treatment approach for lymphoma?

Researchers believe they have discovered how therapy targeting CD47 harnesses the immune system to fight lymphoma and other cancers.
Conducting experiments in immune-competent mice, the team found that anti-CD47 therapy drives T-cell mediated elimination of lymphoma, colon cancer, and breast cancer.
The group’s research also revealed how the timing of chemotherapy administration affects anti-CD47 therapy.
Yang-Xin Fu, MD, PhD, of the University of Chicago in Illinois, and his colleagues described this research in Nature Medicine.
Previous research had shown that many cancer cells have CD47 on their surface. The protein instructs circulating macrophages not to devour the cells, but anti-CD47 therapy can negate this effect. This research relied on human tumors transplanted in immunocompromised mice.
With the current study, Dr Fu and his colleagues transplanted tumors from mice into genetically identical hosts with intact immune systems.
The team’s experiments revealed that anti-CD47-mediated tumor rejection requires both innate and adaptive immune responses. And the bulk of the therapeutic effect from CD47 blockade relies not on macrophages but on dendritic cells.
Dendritic cells proved more potent than macrophages at priming CD8+ T cells. Dendritic cells also caused type-1 interferon to boost adaptive immunity and activated the STING pathway, which was “absolutely essential for the antitumor effect of anti-CD47 therapy.”
The researchers also found evidence to suggest that chemotherapy should be administered before, rather than after, anti-CD47 therapy.
The team tested the anti-CD47 monoclonal antibody (mAb) MIAP301 in combination with clinically equivalent doses of cyclophosphamide or paclitaxel in mouse models of lymphoma (established A20 tumors).
When the chemotherapy was administered after the mAb, tumor regression was no faster than when the mAb was given alone.
In fact, the chemotherapy appeared to hinder antitumor memory responses generated by the mAb. When the researchers removed all tumors and rechallenged the mice with A20 cells, all of the mice that had received the mAb alone rejected the tumor rechallenge.
But mice that had received the mAb followed by chemotherapy were susceptible to tumor outgrowth—50% of cyclophosphamide-treated mice and 80% of paclitaxel-treated mice.
When chemotherapy was given before the mAb, however, it conferred benefits. A single dose of either chemotherapy drug synergized with the mAb to fight lymphoma.
And the treatment preserved the host memory response against relapsing tumors. All of the cyclophosphamide-treated mice and 80% of the paclitaxel-treated mice were resistant to tumor rechallenge.
The researchers said this suggests the order of treatment administration could have a major impact on primary and memory immune responses to tumors and alter outcomes of anti-CD47 therapy.
“Our results point to a new and more personalized strategy to modulate the tumor microenvironment,” Dr Fu said. “We think our approach, along with further investigation of scheduling and dosing, could improve survival and quality of life for patients battling advanced cancer.” ![]()

Researchers believe they have discovered how therapy targeting CD47 harnesses the immune system to fight lymphoma and other cancers.
Conducting experiments in immune-competent mice, the team found that anti-CD47 therapy drives T-cell mediated elimination of lymphoma, colon cancer, and breast cancer.
The group’s research also revealed how the timing of chemotherapy administration affects anti-CD47 therapy.
Yang-Xin Fu, MD, PhD, of the University of Chicago in Illinois, and his colleagues described this research in Nature Medicine.
Previous research had shown that many cancer cells have CD47 on their surface. The protein instructs circulating macrophages not to devour the cells, but anti-CD47 therapy can negate this effect. This research relied on human tumors transplanted in immunocompromised mice.
With the current study, Dr Fu and his colleagues transplanted tumors from mice into genetically identical hosts with intact immune systems.
The team’s experiments revealed that anti-CD47-mediated tumor rejection requires both innate and adaptive immune responses. And the bulk of the therapeutic effect from CD47 blockade relies not on macrophages but on dendritic cells.
Dendritic cells proved more potent than macrophages at priming CD8+ T cells. Dendritic cells also caused type-1 interferon to boost adaptive immunity and activated the STING pathway, which was “absolutely essential for the antitumor effect of anti-CD47 therapy.”
The researchers also found evidence to suggest that chemotherapy should be administered before, rather than after, anti-CD47 therapy.
The team tested the anti-CD47 monoclonal antibody (mAb) MIAP301 in combination with clinically equivalent doses of cyclophosphamide or paclitaxel in mouse models of lymphoma (established A20 tumors).
When the chemotherapy was administered after the mAb, tumor regression was no faster than when the mAb was given alone.
In fact, the chemotherapy appeared to hinder antitumor memory responses generated by the mAb. When the researchers removed all tumors and rechallenged the mice with A20 cells, all of the mice that had received the mAb alone rejected the tumor rechallenge.
But mice that had received the mAb followed by chemotherapy were susceptible to tumor outgrowth—50% of cyclophosphamide-treated mice and 80% of paclitaxel-treated mice.
When chemotherapy was given before the mAb, however, it conferred benefits. A single dose of either chemotherapy drug synergized with the mAb to fight lymphoma.
And the treatment preserved the host memory response against relapsing tumors. All of the cyclophosphamide-treated mice and 80% of the paclitaxel-treated mice were resistant to tumor rechallenge.
The researchers said this suggests the order of treatment administration could have a major impact on primary and memory immune responses to tumors and alter outcomes of anti-CD47 therapy.
“Our results point to a new and more personalized strategy to modulate the tumor microenvironment,” Dr Fu said. “We think our approach, along with further investigation of scheduling and dosing, could improve survival and quality of life for patients battling advanced cancer.” ![]()

Researchers believe they have discovered how therapy targeting CD47 harnesses the immune system to fight lymphoma and other cancers.
Conducting experiments in immune-competent mice, the team found that anti-CD47 therapy drives T-cell mediated elimination of lymphoma, colon cancer, and breast cancer.
The group’s research also revealed how the timing of chemotherapy administration affects anti-CD47 therapy.
Yang-Xin Fu, MD, PhD, of the University of Chicago in Illinois, and his colleagues described this research in Nature Medicine.
Previous research had shown that many cancer cells have CD47 on their surface. The protein instructs circulating macrophages not to devour the cells, but anti-CD47 therapy can negate this effect. This research relied on human tumors transplanted in immunocompromised mice.
With the current study, Dr Fu and his colleagues transplanted tumors from mice into genetically identical hosts with intact immune systems.
The team’s experiments revealed that anti-CD47-mediated tumor rejection requires both innate and adaptive immune responses. And the bulk of the therapeutic effect from CD47 blockade relies not on macrophages but on dendritic cells.
Dendritic cells proved more potent than macrophages at priming CD8+ T cells. Dendritic cells also caused type-1 interferon to boost adaptive immunity and activated the STING pathway, which was “absolutely essential for the antitumor effect of anti-CD47 therapy.”
The researchers also found evidence to suggest that chemotherapy should be administered before, rather than after, anti-CD47 therapy.
The team tested the anti-CD47 monoclonal antibody (mAb) MIAP301 in combination with clinically equivalent doses of cyclophosphamide or paclitaxel in mouse models of lymphoma (established A20 tumors).
When the chemotherapy was administered after the mAb, tumor regression was no faster than when the mAb was given alone.
In fact, the chemotherapy appeared to hinder antitumor memory responses generated by the mAb. When the researchers removed all tumors and rechallenged the mice with A20 cells, all of the mice that had received the mAb alone rejected the tumor rechallenge.
But mice that had received the mAb followed by chemotherapy were susceptible to tumor outgrowth—50% of cyclophosphamide-treated mice and 80% of paclitaxel-treated mice.
When chemotherapy was given before the mAb, however, it conferred benefits. A single dose of either chemotherapy drug synergized with the mAb to fight lymphoma.
And the treatment preserved the host memory response against relapsing tumors. All of the cyclophosphamide-treated mice and 80% of the paclitaxel-treated mice were resistant to tumor rechallenge.
The researchers said this suggests the order of treatment administration could have a major impact on primary and memory immune responses to tumors and alter outcomes of anti-CD47 therapy.
“Our results point to a new and more personalized strategy to modulate the tumor microenvironment,” Dr Fu said. “We think our approach, along with further investigation of scheduling and dosing, could improve survival and quality of life for patients battling advanced cancer.” ![]()
Daratumumab monotherapy impresses in hard-to-treat multiple myeloma
Daratumumab monotherapy yielded impressive results in patients with heavily pretreated and refractory multiple myeloma, according to the full results of a phase I/II study published online by the New England Journal of Medicine.
Among 42 patients receiving 16 mg/kg of daratumumab in part 2 of the study, the overall response rate was 36%, including 2 complete responses, 2 very good partial responses, and 11 partial responses.
Median progression-free survival was 5.6 months. Responses deepened over time, with 65% of responders remaining progression-free at 12 months, study author Dr. Henk M. Lokhorst, of University Medical Center Utrecht, and the VU University Medical Center, Amsterdam, both in the Netherlands, and his associates reported (N Engl J Med. 2015 Aug. 26 [doi:10.1056/NEJMoa1506348]).
In 30 patients given daratumumab 8 mg/kg, the response rate was 10%, including three partial responses.
Patients in part 2 of the study were heavily pretreated with a median of four prior lines of therapy (range 2-12) and 64% were refractory to the current standard treatment of protease inhibitors (PIs) and immunomodulatory agents (IMiDs). Such patients have a poor prognosis, with an estimated median overall survival of 9 months and estimated event-free survival of 5 months at best, Dr. Lokhorst and his associates noted.
“Daratumumab showed single-agent antitumor activity in a population of patients with highly difficult-to-treat myeloma who had very few effective treatment options,” the investigators concluded. “Its target and mechanisms of action differentiate it from existing therapies.”
Daratumumab is a human monoclonal antibody that targets CD38, which is uniformly overexpressed in multiple myeloma cells.
In part 1, the dose-escalation phase of the study, no maximum-tolerated dose was identified at doses up to 24 mg/kg in 32 patients. Two dose-limiting toxicities – grade 3 anemia and grade 3 elevated aspartate aminotransferase levels – occurred at 0.1 mg/kg and 1 mg/kg, but no further events were seen with dose escalation, Dr. Lokhorst and associates reported.
The most-common adverse events in part 2 of the study – occurring in at least 25% of patients – were fatigue, allergic rhinitis, and pyrexia. Infusion-related reactions were mild and were seen in 71% of patients, typically during the first infusion.
The most-common grade 3/4 events were pneumonia (five patients) and thrombocytopenia (four patients). Neutropenia, leukopenia, anemia, and hyperglycemia occurred in two patients each. There was one grade 5 pneumonia, but investigators did not think it was related to the study drug, they wrote.
Exploratory subgroup analyses revealed that response rates were similar in patients with disease refractory to both bortezomib (Velcade) and lenalidomide (Revlimid) and the total population, but were higher in patients with two or three prior lines of therapy vs. more heavily pretreated patients (56% vs. 23%).
Data from the study were included in a Biologics License Application submitted in July 2015 to the U.S. Food and Drug Administration for daratumumab as a treatment for patients with multiple myeloma who have received at least three prior lines of therapy including both a PI and an IMiD or who are double refractory to a PI and an IMiD, according to Genmab.
Daratumumab, which has already been granted breakthrough therapy status by the FDA, is being develop by Janssen Biotech, under an exclusive license from Genmab.
On Twitter @pwendl
The single-agent activity of daratumumab, including complete responses, in this patient population is surprising and very encouraging. These results are probably due to its pleiotropic mechanisms of action against myeloma.
The introduction of monoclonal antibodies into the arsenal against myeloma is game-changing in multiple myeloma treatment. These agents have the advantage of an immune-based approach without the need for patient-specific cell manipulation. Their limited toxicity allows for easy combining with existing therapies.
Even with this enthusiasm, unanswered questions remain. How do tumors escape the effects of daratumumab? Can daratumumab, like rituximab in the treatment of lymphoma, be active in many phases of treatment, such as in induction, consolidation, and maintenance therapies? Can daratumumab resistance be predicted? As we begin to tackle the complexity of these questions, it is reassuring to know that we have yet one more treatment option that will contribute in an important way to improvement in outcomes in patients with myeloma.
These comments were excerpted from an editorial accompanying the report by Lokhorst et al. (N Engl J Med. 2015 Aug 26 [doi: 10.1056/NEJMe1509419]).
Dr. Noopur Raje is director of the multiple myeloma program at Massachusetts General Hospital, Boston. Dr. Dan L. Longo is a professor of medicine at Harvard Medical School, Boston.
The single-agent activity of daratumumab, including complete responses, in this patient population is surprising and very encouraging. These results are probably due to its pleiotropic mechanisms of action against myeloma.
The introduction of monoclonal antibodies into the arsenal against myeloma is game-changing in multiple myeloma treatment. These agents have the advantage of an immune-based approach without the need for patient-specific cell manipulation. Their limited toxicity allows for easy combining with existing therapies.
Even with this enthusiasm, unanswered questions remain. How do tumors escape the effects of daratumumab? Can daratumumab, like rituximab in the treatment of lymphoma, be active in many phases of treatment, such as in induction, consolidation, and maintenance therapies? Can daratumumab resistance be predicted? As we begin to tackle the complexity of these questions, it is reassuring to know that we have yet one more treatment option that will contribute in an important way to improvement in outcomes in patients with myeloma.
These comments were excerpted from an editorial accompanying the report by Lokhorst et al. (N Engl J Med. 2015 Aug 26 [doi: 10.1056/NEJMe1509419]).
Dr. Noopur Raje is director of the multiple myeloma program at Massachusetts General Hospital, Boston. Dr. Dan L. Longo is a professor of medicine at Harvard Medical School, Boston.
The single-agent activity of daratumumab, including complete responses, in this patient population is surprising and very encouraging. These results are probably due to its pleiotropic mechanisms of action against myeloma.
The introduction of monoclonal antibodies into the arsenal against myeloma is game-changing in multiple myeloma treatment. These agents have the advantage of an immune-based approach without the need for patient-specific cell manipulation. Their limited toxicity allows for easy combining with existing therapies.
Even with this enthusiasm, unanswered questions remain. How do tumors escape the effects of daratumumab? Can daratumumab, like rituximab in the treatment of lymphoma, be active in many phases of treatment, such as in induction, consolidation, and maintenance therapies? Can daratumumab resistance be predicted? As we begin to tackle the complexity of these questions, it is reassuring to know that we have yet one more treatment option that will contribute in an important way to improvement in outcomes in patients with myeloma.
These comments were excerpted from an editorial accompanying the report by Lokhorst et al. (N Engl J Med. 2015 Aug 26 [doi: 10.1056/NEJMe1509419]).
Dr. Noopur Raje is director of the multiple myeloma program at Massachusetts General Hospital, Boston. Dr. Dan L. Longo is a professor of medicine at Harvard Medical School, Boston.
Daratumumab monotherapy yielded impressive results in patients with heavily pretreated and refractory multiple myeloma, according to the full results of a phase I/II study published online by the New England Journal of Medicine.
Among 42 patients receiving 16 mg/kg of daratumumab in part 2 of the study, the overall response rate was 36%, including 2 complete responses, 2 very good partial responses, and 11 partial responses.
Median progression-free survival was 5.6 months. Responses deepened over time, with 65% of responders remaining progression-free at 12 months, study author Dr. Henk M. Lokhorst, of University Medical Center Utrecht, and the VU University Medical Center, Amsterdam, both in the Netherlands, and his associates reported (N Engl J Med. 2015 Aug. 26 [doi:10.1056/NEJMoa1506348]).
In 30 patients given daratumumab 8 mg/kg, the response rate was 10%, including three partial responses.
Patients in part 2 of the study were heavily pretreated with a median of four prior lines of therapy (range 2-12) and 64% were refractory to the current standard treatment of protease inhibitors (PIs) and immunomodulatory agents (IMiDs). Such patients have a poor prognosis, with an estimated median overall survival of 9 months and estimated event-free survival of 5 months at best, Dr. Lokhorst and his associates noted.
“Daratumumab showed single-agent antitumor activity in a population of patients with highly difficult-to-treat myeloma who had very few effective treatment options,” the investigators concluded. “Its target and mechanisms of action differentiate it from existing therapies.”
Daratumumab is a human monoclonal antibody that targets CD38, which is uniformly overexpressed in multiple myeloma cells.
In part 1, the dose-escalation phase of the study, no maximum-tolerated dose was identified at doses up to 24 mg/kg in 32 patients. Two dose-limiting toxicities – grade 3 anemia and grade 3 elevated aspartate aminotransferase levels – occurred at 0.1 mg/kg and 1 mg/kg, but no further events were seen with dose escalation, Dr. Lokhorst and associates reported.
The most-common adverse events in part 2 of the study – occurring in at least 25% of patients – were fatigue, allergic rhinitis, and pyrexia. Infusion-related reactions were mild and were seen in 71% of patients, typically during the first infusion.
The most-common grade 3/4 events were pneumonia (five patients) and thrombocytopenia (four patients). Neutropenia, leukopenia, anemia, and hyperglycemia occurred in two patients each. There was one grade 5 pneumonia, but investigators did not think it was related to the study drug, they wrote.
Exploratory subgroup analyses revealed that response rates were similar in patients with disease refractory to both bortezomib (Velcade) and lenalidomide (Revlimid) and the total population, but were higher in patients with two or three prior lines of therapy vs. more heavily pretreated patients (56% vs. 23%).
Data from the study were included in a Biologics License Application submitted in July 2015 to the U.S. Food and Drug Administration for daratumumab as a treatment for patients with multiple myeloma who have received at least three prior lines of therapy including both a PI and an IMiD or who are double refractory to a PI and an IMiD, according to Genmab.
Daratumumab, which has already been granted breakthrough therapy status by the FDA, is being develop by Janssen Biotech, under an exclusive license from Genmab.
On Twitter @pwendl
Daratumumab monotherapy yielded impressive results in patients with heavily pretreated and refractory multiple myeloma, according to the full results of a phase I/II study published online by the New England Journal of Medicine.
Among 42 patients receiving 16 mg/kg of daratumumab in part 2 of the study, the overall response rate was 36%, including 2 complete responses, 2 very good partial responses, and 11 partial responses.
Median progression-free survival was 5.6 months. Responses deepened over time, with 65% of responders remaining progression-free at 12 months, study author Dr. Henk M. Lokhorst, of University Medical Center Utrecht, and the VU University Medical Center, Amsterdam, both in the Netherlands, and his associates reported (N Engl J Med. 2015 Aug. 26 [doi:10.1056/NEJMoa1506348]).
In 30 patients given daratumumab 8 mg/kg, the response rate was 10%, including three partial responses.
Patients in part 2 of the study were heavily pretreated with a median of four prior lines of therapy (range 2-12) and 64% were refractory to the current standard treatment of protease inhibitors (PIs) and immunomodulatory agents (IMiDs). Such patients have a poor prognosis, with an estimated median overall survival of 9 months and estimated event-free survival of 5 months at best, Dr. Lokhorst and his associates noted.
“Daratumumab showed single-agent antitumor activity in a population of patients with highly difficult-to-treat myeloma who had very few effective treatment options,” the investigators concluded. “Its target and mechanisms of action differentiate it from existing therapies.”
Daratumumab is a human monoclonal antibody that targets CD38, which is uniformly overexpressed in multiple myeloma cells.
In part 1, the dose-escalation phase of the study, no maximum-tolerated dose was identified at doses up to 24 mg/kg in 32 patients. Two dose-limiting toxicities – grade 3 anemia and grade 3 elevated aspartate aminotransferase levels – occurred at 0.1 mg/kg and 1 mg/kg, but no further events were seen with dose escalation, Dr. Lokhorst and associates reported.
The most-common adverse events in part 2 of the study – occurring in at least 25% of patients – were fatigue, allergic rhinitis, and pyrexia. Infusion-related reactions were mild and were seen in 71% of patients, typically during the first infusion.
The most-common grade 3/4 events were pneumonia (five patients) and thrombocytopenia (four patients). Neutropenia, leukopenia, anemia, and hyperglycemia occurred in two patients each. There was one grade 5 pneumonia, but investigators did not think it was related to the study drug, they wrote.
Exploratory subgroup analyses revealed that response rates were similar in patients with disease refractory to both bortezomib (Velcade) and lenalidomide (Revlimid) and the total population, but were higher in patients with two or three prior lines of therapy vs. more heavily pretreated patients (56% vs. 23%).
Data from the study were included in a Biologics License Application submitted in July 2015 to the U.S. Food and Drug Administration for daratumumab as a treatment for patients with multiple myeloma who have received at least three prior lines of therapy including both a PI and an IMiD or who are double refractory to a PI and an IMiD, according to Genmab.
Daratumumab, which has already been granted breakthrough therapy status by the FDA, is being develop by Janssen Biotech, under an exclusive license from Genmab.
On Twitter @pwendl
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Daratumumab monotherapy has encouraging efficacy and a favorable safety profile in heavily pretreated and refractory multiple myeloma.
Major finding: The overall response rate was 36% with daratumumab 16 mg/kg.
Data source: Open-label phase I/II study in 104 patients with relapsed or refractory multiple myeloma.
Disclosures: The study was funded by Janssen Biotech and Genmab. Dr. Lokhorst reported grant support and personal fees from Genmab and Janssen and personal fees from Amgen. Several coauthors also reported fees from Janssen and/or Genmab. Dr. Raje reported personal fees from Celgene and Millennium/Takeda and grant support from Eli Lilly and AstraZeneca. Dr. Longo disclosed being employed as deputy editor at the New England Journal of Medicine.
Public uninformed about cancer therapies, survey suggests

receiving chemotherapy
Photo by Rhoda Baer
Results of a new survey suggest many adults in the UK may be uninformed about cancer treatment options, despite broad media coverage of these therapies.
Personalized drug treatment, immunotherapy, and proton beam therapy have all been covered by the lay media and featured in news stories across the globe.
But a survey of more than 2000 UK adults showed that most respondents were not aware of these treatment types.
Only 19% of respondents said they had heard about immunotherapy, 29% had heard of personalized drug treatment, and 30% had heard of proton beam therapy.
The survey, which included 2081 adults, was conducted online by YouGov in June. It was commissioned by Cancer Research UK and other members of the Radiotherapy Awareness Programme.
The primary goal of the survey was to examine public awareness of radiotherapy. And the results showed that many respondents were unaware of newer, more targeted radiotherapy options.
Respondents were largely uninformed about other types of cancer treatment as well. However, of the respondents who elected to give their opinion (n=1877), most said the National Health Service (NHS) should fund chemotherapy and other drug treatments over radiotherapy.
Survey questions and responses were as follows.
Radiotherapy
Before taking this survey, which, if any, of the following types of radiotherapy had you heard of?
| Intensity-modulated radiotherapy | 4% |
| Stereotactic radiotherapy/
stereotactic ablative radiotherapy |
3% |
| Image-guided radiotherapy | 9% |
| Proton beam therapy | 30% |
| Brachytherapy | 5% |
| Radiofrequency ablation | 7% |
| Cyberknife | 4% |
| Gammaknife | 6% |
| Higgs-boson radiotherapy
(red herring option) |
6% |
| Carbon ion radiotherapy
(red herring option) |
3% |
| None of these | 50% |
| Prefer not to say | 11% |
Other cancer treatments
Which, if any, of the following specific types of cancer treatments/tests had you heard of before taking this survey?
| Immunotherapy | 19% |
| Personalized drugs | 29% |
| Monoclonal antibodies | 5% |
| High-dose chemotherapy
with stem cell transplant |
26% |
| Tablet chemotherapy | 28% |
| Molecular diagnostic tests | 6% |
| Robotically assisted surgery/Da Vinci robot | 12% |
| Laparoscopic (keyhole) surgery | 39% |
| None of these | 32% |
| Prefer not to say | 11% |
NHS funding
What level of priority do you think the NHS should give to funding each of the following 4 types of cancer treatments?
| Treatment | 1st priority | 2nd priority | 3rd priority | Lowest priority |
| Chemotherapy &
other drug treatments |
57% | 29% | 10% | 4% |
| Surgery | 29% | 35% | 31% | 5% |
| Radiotherapy | 9% | 32% | 53% | 5% |
| Alternative treatments | 5% | 4% | 6% | 86% |
![]()

receiving chemotherapy
Photo by Rhoda Baer
Results of a new survey suggest many adults in the UK may be uninformed about cancer treatment options, despite broad media coverage of these therapies.
Personalized drug treatment, immunotherapy, and proton beam therapy have all been covered by the lay media and featured in news stories across the globe.
But a survey of more than 2000 UK adults showed that most respondents were not aware of these treatment types.
Only 19% of respondents said they had heard about immunotherapy, 29% had heard of personalized drug treatment, and 30% had heard of proton beam therapy.
The survey, which included 2081 adults, was conducted online by YouGov in June. It was commissioned by Cancer Research UK and other members of the Radiotherapy Awareness Programme.
The primary goal of the survey was to examine public awareness of radiotherapy. And the results showed that many respondents were unaware of newer, more targeted radiotherapy options.
Respondents were largely uninformed about other types of cancer treatment as well. However, of the respondents who elected to give their opinion (n=1877), most said the National Health Service (NHS) should fund chemotherapy and other drug treatments over radiotherapy.
Survey questions and responses were as follows.
Radiotherapy
Before taking this survey, which, if any, of the following types of radiotherapy had you heard of?
| Intensity-modulated radiotherapy | 4% |
| Stereotactic radiotherapy/
stereotactic ablative radiotherapy |
3% |
| Image-guided radiotherapy | 9% |
| Proton beam therapy | 30% |
| Brachytherapy | 5% |
| Radiofrequency ablation | 7% |
| Cyberknife | 4% |
| Gammaknife | 6% |
| Higgs-boson radiotherapy
(red herring option) |
6% |
| Carbon ion radiotherapy
(red herring option) |
3% |
| None of these | 50% |
| Prefer not to say | 11% |
Other cancer treatments
Which, if any, of the following specific types of cancer treatments/tests had you heard of before taking this survey?
| Immunotherapy | 19% |
| Personalized drugs | 29% |
| Monoclonal antibodies | 5% |
| High-dose chemotherapy
with stem cell transplant |
26% |
| Tablet chemotherapy | 28% |
| Molecular diagnostic tests | 6% |
| Robotically assisted surgery/Da Vinci robot | 12% |
| Laparoscopic (keyhole) surgery | 39% |
| None of these | 32% |
| Prefer not to say | 11% |
NHS funding
What level of priority do you think the NHS should give to funding each of the following 4 types of cancer treatments?
| Treatment | 1st priority | 2nd priority | 3rd priority | Lowest priority |
| Chemotherapy &
other drug treatments |
57% | 29% | 10% | 4% |
| Surgery | 29% | 35% | 31% | 5% |
| Radiotherapy | 9% | 32% | 53% | 5% |
| Alternative treatments | 5% | 4% | 6% | 86% |
![]()

receiving chemotherapy
Photo by Rhoda Baer
Results of a new survey suggest many adults in the UK may be uninformed about cancer treatment options, despite broad media coverage of these therapies.
Personalized drug treatment, immunotherapy, and proton beam therapy have all been covered by the lay media and featured in news stories across the globe.
But a survey of more than 2000 UK adults showed that most respondents were not aware of these treatment types.
Only 19% of respondents said they had heard about immunotherapy, 29% had heard of personalized drug treatment, and 30% had heard of proton beam therapy.
The survey, which included 2081 adults, was conducted online by YouGov in June. It was commissioned by Cancer Research UK and other members of the Radiotherapy Awareness Programme.
The primary goal of the survey was to examine public awareness of radiotherapy. And the results showed that many respondents were unaware of newer, more targeted radiotherapy options.
Respondents were largely uninformed about other types of cancer treatment as well. However, of the respondents who elected to give their opinion (n=1877), most said the National Health Service (NHS) should fund chemotherapy and other drug treatments over radiotherapy.
Survey questions and responses were as follows.
Radiotherapy
Before taking this survey, which, if any, of the following types of radiotherapy had you heard of?
| Intensity-modulated radiotherapy | 4% |
| Stereotactic radiotherapy/
stereotactic ablative radiotherapy |
3% |
| Image-guided radiotherapy | 9% |
| Proton beam therapy | 30% |
| Brachytherapy | 5% |
| Radiofrequency ablation | 7% |
| Cyberknife | 4% |
| Gammaknife | 6% |
| Higgs-boson radiotherapy
(red herring option) |
6% |
| Carbon ion radiotherapy
(red herring option) |
3% |
| None of these | 50% |
| Prefer not to say | 11% |
Other cancer treatments
Which, if any, of the following specific types of cancer treatments/tests had you heard of before taking this survey?
| Immunotherapy | 19% |
| Personalized drugs | 29% |
| Monoclonal antibodies | 5% |
| High-dose chemotherapy
with stem cell transplant |
26% |
| Tablet chemotherapy | 28% |
| Molecular diagnostic tests | 6% |
| Robotically assisted surgery/Da Vinci robot | 12% |
| Laparoscopic (keyhole) surgery | 39% |
| None of these | 32% |
| Prefer not to say | 11% |
NHS funding
What level of priority do you think the NHS should give to funding each of the following 4 types of cancer treatments?
| Treatment | 1st priority | 2nd priority | 3rd priority | Lowest priority |
| Chemotherapy &
other drug treatments |
57% | 29% | 10% | 4% |
| Surgery | 29% | 35% | 31% | 5% |
| Radiotherapy | 9% | 32% | 53% | 5% |
| Alternative treatments | 5% | 4% | 6% | 86% |
![]()
Protein may be key in virus, cancer research

Image courtesy of the
University of North Carolina
Researchers say they have uncovered a viral protein that inhibits cGAS, the principal cytosolic DNA sensor that detects invading viral DNA and triggers antiviral responses.
The protein, Kaposi’s sarcoma-associated herpesvirus (KSHV) ORF52, subverts cytosolic DNA sensing by directly inhibiting cGAS enzymatic activity.
The team believes this finding could have a range of therapeutic implications.
“We can manipulate the protein and/or the sensor to boost or tune down the immune response in order to fight infectious and autoimmune diseases, as well as cancers,” said Fanxiu Zhu, PhD, of Florida State University in Tallahassee.
Dr Zhu and his colleagues described this research in Cell Host and Microbe.
The authors noted that, although cGAS senses several DNA viruses, viral strategies targeting cGAS are “virtually unknown.”
To uncover a cGAS inhibitor, the researchers screened every protein in a KSHV cell—90 in total. This revealed KSHV ORF52, which the team renamed “KicGas,” an abbreviation for “KSHV inhibitor of cGAS.”
Further investigation revealed how KicGas inhibits cGAS activity: it must bind to both DNA and cGAS.
The researchers then found that ORF52 homologs in other gammaherpesviruses also inhibit cGAS activity and similarly bind cGAS and DNA.
Finally, the team infected human cell lines with KSHV to mimic natural infection. They found that KSHV triggers a cGAS-dependent immune response that can be partially mitigated by KicGas.
When the researchers eliminated KicGas from infected cells, the cells produced a much stronger immune response.
For the next phase of research, the team is building a 3-dimensional model to help them better understand how KicGas functions. They hope this will help them utilize KicGas to fight disease. ![]()

Image courtesy of the
University of North Carolina
Researchers say they have uncovered a viral protein that inhibits cGAS, the principal cytosolic DNA sensor that detects invading viral DNA and triggers antiviral responses.
The protein, Kaposi’s sarcoma-associated herpesvirus (KSHV) ORF52, subverts cytosolic DNA sensing by directly inhibiting cGAS enzymatic activity.
The team believes this finding could have a range of therapeutic implications.
“We can manipulate the protein and/or the sensor to boost or tune down the immune response in order to fight infectious and autoimmune diseases, as well as cancers,” said Fanxiu Zhu, PhD, of Florida State University in Tallahassee.
Dr Zhu and his colleagues described this research in Cell Host and Microbe.
The authors noted that, although cGAS senses several DNA viruses, viral strategies targeting cGAS are “virtually unknown.”
To uncover a cGAS inhibitor, the researchers screened every protein in a KSHV cell—90 in total. This revealed KSHV ORF52, which the team renamed “KicGas,” an abbreviation for “KSHV inhibitor of cGAS.”
Further investigation revealed how KicGas inhibits cGAS activity: it must bind to both DNA and cGAS.
The researchers then found that ORF52 homologs in other gammaherpesviruses also inhibit cGAS activity and similarly bind cGAS and DNA.
Finally, the team infected human cell lines with KSHV to mimic natural infection. They found that KSHV triggers a cGAS-dependent immune response that can be partially mitigated by KicGas.
When the researchers eliminated KicGas from infected cells, the cells produced a much stronger immune response.
For the next phase of research, the team is building a 3-dimensional model to help them better understand how KicGas functions. They hope this will help them utilize KicGas to fight disease. ![]()

Image courtesy of the
University of North Carolina
Researchers say they have uncovered a viral protein that inhibits cGAS, the principal cytosolic DNA sensor that detects invading viral DNA and triggers antiviral responses.
The protein, Kaposi’s sarcoma-associated herpesvirus (KSHV) ORF52, subverts cytosolic DNA sensing by directly inhibiting cGAS enzymatic activity.
The team believes this finding could have a range of therapeutic implications.
“We can manipulate the protein and/or the sensor to boost or tune down the immune response in order to fight infectious and autoimmune diseases, as well as cancers,” said Fanxiu Zhu, PhD, of Florida State University in Tallahassee.
Dr Zhu and his colleagues described this research in Cell Host and Microbe.
The authors noted that, although cGAS senses several DNA viruses, viral strategies targeting cGAS are “virtually unknown.”
To uncover a cGAS inhibitor, the researchers screened every protein in a KSHV cell—90 in total. This revealed KSHV ORF52, which the team renamed “KicGas,” an abbreviation for “KSHV inhibitor of cGAS.”
Further investigation revealed how KicGas inhibits cGAS activity: it must bind to both DNA and cGAS.
The researchers then found that ORF52 homologs in other gammaherpesviruses also inhibit cGAS activity and similarly bind cGAS and DNA.
Finally, the team infected human cell lines with KSHV to mimic natural infection. They found that KSHV triggers a cGAS-dependent immune response that can be partially mitigated by KicGas.
When the researchers eliminated KicGas from infected cells, the cells produced a much stronger immune response.
For the next phase of research, the team is building a 3-dimensional model to help them better understand how KicGas functions. They hope this will help them utilize KicGas to fight disease. ![]()
Discovery reveals potential for viral cancer treatment

Image by Eric Smith
Researchers say they have discovered critical details that explain how a cellular response system tells the difference between damage to the body’s own DNA and the foreign DNA of an invading virus.
The team believes this discovery could aid the development of new cancer-selective viral therapies, and it may help explain why aging, cancers, and other diseases
seem to open the door to viral infections.
“Our study reveals fundamental mechanisms that distinguish DNA breaks at cellular and viral genomes to trigger different responses that protect the host,” said Clodagh O’Shea, of the Salk Institute for Biological Studies in La Jolla, California.
“The findings may also explain why certain conditions like aging, cancer chemotherapy, and inflammation make us more susceptible to viral infection.”
Dr O’Shea and Govind Shah, PhD, also of the Salk Institute, reported these findings in Cell.
The pair described how a cluster of proteins known as the MRN complex detects DNA breaks and amplifies its response through histones.
MRN starts a domino effect, activating histones on surrounding chromosomes, which summons a cascade of additional proteins and results in a cell-wide, all-hands-on-deck alarm to help mend the DNA.
If the cell can’t fix the DNA break, it will induce apoptosis—a self-destruct mechanism that helps to prevent mutated cells from replicating and therefore prevents tumor growth.
“What’s interesting is that even a single break transmits a global signal through the cell, halting cell division and growth,” Dr O’Shea said. “This response prevents replication so the cell doesn’t pass on a break.”
Drs O’Shea and Shah also found that, when it comes to defending against DNA viruses, the cell’s response system begins the same way—with MRN detecting breaks. But it never progresses to the global alarm signal in the case of the virus.
Typically, a common DNA virus enters the cell’s nucleus and turns on genes to replicate its own DNA. The cell detects the unauthorized replication, and the MRN complex grabs and selectively neutralizes viral DNA without triggering a global response that would arrest or kill the cell.
So the MRN response to the virus stays localized and only selectively prevents viral, but not cellular, replication.
When both threats to the genome are present, MRN will activate the massive response at the DNA break, and no MRN is left to respond to the virus. This means the virus is effectively ignored while the cell responds to the more massive alarm.
“The requirement of MRN for sensing both cellular and viral genome breaks has profound consequences,” Dr O’Shea said.
“When MRN is recruited to cellular DNA breaks, it can no longer sense and respond to incoming viral genomes. Thus, the act of responding to cellular genome breaks inactivates the host’s defenses to viral replication.”
Dr O’Shea said this may explain why people who have high levels of cellular DNA damage—such as cancer patients—are more susceptible to viral infections.
“Having damaged DNA compromises our cells’ ability to fight viral infection, while having healthy DNA boosts our cells’ ability to catch viral DNA,” Dr Shah said. “Our work implies that we may be able to engineer viruses that selectively kill cancer cells.”
The researchers aim to use this new knowledge to create viruses that are destroyed in normal cells but replicate specifically in cancer cells.
Unlike normal cells, cancer cells almost always have very high levels of DNA damage. In cancer cells, MRN is already so preoccupied with responding to DNA breaks that an engineered virus could sneak in undetected.
“Cancer cells, by definition, have high mutation rates and genomic instability even at the very earliest stages,” Dr O’Shea said. “So you could imagine building a virus that could destroy even the earliest lesions and be used as a prophylactic.” ![]()

Image by Eric Smith
Researchers say they have discovered critical details that explain how a cellular response system tells the difference between damage to the body’s own DNA and the foreign DNA of an invading virus.
The team believes this discovery could aid the development of new cancer-selective viral therapies, and it may help explain why aging, cancers, and other diseases
seem to open the door to viral infections.
“Our study reveals fundamental mechanisms that distinguish DNA breaks at cellular and viral genomes to trigger different responses that protect the host,” said Clodagh O’Shea, of the Salk Institute for Biological Studies in La Jolla, California.
“The findings may also explain why certain conditions like aging, cancer chemotherapy, and inflammation make us more susceptible to viral infection.”
Dr O’Shea and Govind Shah, PhD, also of the Salk Institute, reported these findings in Cell.
The pair described how a cluster of proteins known as the MRN complex detects DNA breaks and amplifies its response through histones.
MRN starts a domino effect, activating histones on surrounding chromosomes, which summons a cascade of additional proteins and results in a cell-wide, all-hands-on-deck alarm to help mend the DNA.
If the cell can’t fix the DNA break, it will induce apoptosis—a self-destruct mechanism that helps to prevent mutated cells from replicating and therefore prevents tumor growth.
“What’s interesting is that even a single break transmits a global signal through the cell, halting cell division and growth,” Dr O’Shea said. “This response prevents replication so the cell doesn’t pass on a break.”
Drs O’Shea and Shah also found that, when it comes to defending against DNA viruses, the cell’s response system begins the same way—with MRN detecting breaks. But it never progresses to the global alarm signal in the case of the virus.
Typically, a common DNA virus enters the cell’s nucleus and turns on genes to replicate its own DNA. The cell detects the unauthorized replication, and the MRN complex grabs and selectively neutralizes viral DNA without triggering a global response that would arrest or kill the cell.
So the MRN response to the virus stays localized and only selectively prevents viral, but not cellular, replication.
When both threats to the genome are present, MRN will activate the massive response at the DNA break, and no MRN is left to respond to the virus. This means the virus is effectively ignored while the cell responds to the more massive alarm.
“The requirement of MRN for sensing both cellular and viral genome breaks has profound consequences,” Dr O’Shea said.
“When MRN is recruited to cellular DNA breaks, it can no longer sense and respond to incoming viral genomes. Thus, the act of responding to cellular genome breaks inactivates the host’s defenses to viral replication.”
Dr O’Shea said this may explain why people who have high levels of cellular DNA damage—such as cancer patients—are more susceptible to viral infections.
“Having damaged DNA compromises our cells’ ability to fight viral infection, while having healthy DNA boosts our cells’ ability to catch viral DNA,” Dr Shah said. “Our work implies that we may be able to engineer viruses that selectively kill cancer cells.”
The researchers aim to use this new knowledge to create viruses that are destroyed in normal cells but replicate specifically in cancer cells.
Unlike normal cells, cancer cells almost always have very high levels of DNA damage. In cancer cells, MRN is already so preoccupied with responding to DNA breaks that an engineered virus could sneak in undetected.
“Cancer cells, by definition, have high mutation rates and genomic instability even at the very earliest stages,” Dr O’Shea said. “So you could imagine building a virus that could destroy even the earliest lesions and be used as a prophylactic.” ![]()

Image by Eric Smith
Researchers say they have discovered critical details that explain how a cellular response system tells the difference between damage to the body’s own DNA and the foreign DNA of an invading virus.
The team believes this discovery could aid the development of new cancer-selective viral therapies, and it may help explain why aging, cancers, and other diseases
seem to open the door to viral infections.
“Our study reveals fundamental mechanisms that distinguish DNA breaks at cellular and viral genomes to trigger different responses that protect the host,” said Clodagh O’Shea, of the Salk Institute for Biological Studies in La Jolla, California.
“The findings may also explain why certain conditions like aging, cancer chemotherapy, and inflammation make us more susceptible to viral infection.”
Dr O’Shea and Govind Shah, PhD, also of the Salk Institute, reported these findings in Cell.
The pair described how a cluster of proteins known as the MRN complex detects DNA breaks and amplifies its response through histones.
MRN starts a domino effect, activating histones on surrounding chromosomes, which summons a cascade of additional proteins and results in a cell-wide, all-hands-on-deck alarm to help mend the DNA.
If the cell can’t fix the DNA break, it will induce apoptosis—a self-destruct mechanism that helps to prevent mutated cells from replicating and therefore prevents tumor growth.
“What’s interesting is that even a single break transmits a global signal through the cell, halting cell division and growth,” Dr O’Shea said. “This response prevents replication so the cell doesn’t pass on a break.”
Drs O’Shea and Shah also found that, when it comes to defending against DNA viruses, the cell’s response system begins the same way—with MRN detecting breaks. But it never progresses to the global alarm signal in the case of the virus.
Typically, a common DNA virus enters the cell’s nucleus and turns on genes to replicate its own DNA. The cell detects the unauthorized replication, and the MRN complex grabs and selectively neutralizes viral DNA without triggering a global response that would arrest or kill the cell.
So the MRN response to the virus stays localized and only selectively prevents viral, but not cellular, replication.
When both threats to the genome are present, MRN will activate the massive response at the DNA break, and no MRN is left to respond to the virus. This means the virus is effectively ignored while the cell responds to the more massive alarm.
“The requirement of MRN for sensing both cellular and viral genome breaks has profound consequences,” Dr O’Shea said.
“When MRN is recruited to cellular DNA breaks, it can no longer sense and respond to incoming viral genomes. Thus, the act of responding to cellular genome breaks inactivates the host’s defenses to viral replication.”
Dr O’Shea said this may explain why people who have high levels of cellular DNA damage—such as cancer patients—are more susceptible to viral infections.
“Having damaged DNA compromises our cells’ ability to fight viral infection, while having healthy DNA boosts our cells’ ability to catch viral DNA,” Dr Shah said. “Our work implies that we may be able to engineer viruses that selectively kill cancer cells.”
The researchers aim to use this new knowledge to create viruses that are destroyed in normal cells but replicate specifically in cancer cells.
Unlike normal cells, cancer cells almost always have very high levels of DNA damage. In cancer cells, MRN is already so preoccupied with responding to DNA breaks that an engineered virus could sneak in undetected.
“Cancer cells, by definition, have high mutation rates and genomic instability even at the very earliest stages,” Dr O’Shea said. “So you could imagine building a virus that could destroy even the earliest lesions and be used as a prophylactic.” ![]()
Team quantifies CAM use among seniors with cancer

Photo by Rhoda Baer
A new study suggests that seniors with cancer may be taking complementary or alternative medicines (CAMs) without their oncologists’ knowledge.
In this single-center study, 27% of senior cancer patients took CAMs at some point during their cancer care.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Polypharmacy and certain comorbidities were linked to CAM use as well.
Researchers reported these findings in the Journal of Geriatric Oncology.
“Currently, few oncologists are aware of the alternative medicines their patients take,” said study author Ginah Nightingale, PharmD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“Patients often fail to disclose the CAMs they take because they think they are safe, natural, nontoxic, and not relevant to their cancer care; because they think their doctor will disapprove; or because the doctor doesn’t specifically ask.”
To quantify CAM use in older cancer patients treated at their institution, Dr Nightingale and her colleagues surveyed patients who came to the Senior Adult Oncology Center at Thomas Jefferson University.
In a single visit, patients were seen by a medical oncologist, geriatrician, clinical pharmacist, social worker, and dietician. As part of this assessment, the patients brought in the contents of their medicine cabinets, and the medications they actively used were reviewed and recorded.
A total of 234 patients were included in the final analysis. Their mean age was 79.9 (range, 61–98). Most (87%) had solid tumor malignancies, were Caucasian (74%), and were female (64%).
In all, 26.5% of patients (n=62) had taken at least 1 CAM during their cancer care, with 19.2% taking 1 CAM, 6.4% taking 2, 0.4% taking 3, and 0.4% taking 4 or more CAMs. The highest number of CAMs taken was 10.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Comorbidities significantly associated with CAM use were vision impairment (P=0.048) and urologic comorbidities (P=0.021). Polypharmacy (concurrent use of 5 or more medications) was significantly associated with CAM use as well (P=0.045).
Some of the commonly used CAMs were mega-dose vitamins or minerals, as well as treatments for macular degeneration, stomach probiotics, and joint health.
The researchers did not examine the potential adverse effects of these medications, but Dr Nightingale said some are known to have a biochemical effect on the body and other drugs.
“It is very important to do a comprehensive screen of all of the medications that older cancer patients take, including CAMs,” she added. “Clear and transparent documentation of CAM use should be recorded in the patient’s medical record. This documentation should indicate that patient-specific communication and/or education was provided so that shared and informed decisions by the patient can be made regarding the continued use of these medications.” ![]()

Photo by Rhoda Baer
A new study suggests that seniors with cancer may be taking complementary or alternative medicines (CAMs) without their oncologists’ knowledge.
In this single-center study, 27% of senior cancer patients took CAMs at some point during their cancer care.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Polypharmacy and certain comorbidities were linked to CAM use as well.
Researchers reported these findings in the Journal of Geriatric Oncology.
“Currently, few oncologists are aware of the alternative medicines their patients take,” said study author Ginah Nightingale, PharmD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“Patients often fail to disclose the CAMs they take because they think they are safe, natural, nontoxic, and not relevant to their cancer care; because they think their doctor will disapprove; or because the doctor doesn’t specifically ask.”
To quantify CAM use in older cancer patients treated at their institution, Dr Nightingale and her colleagues surveyed patients who came to the Senior Adult Oncology Center at Thomas Jefferson University.
In a single visit, patients were seen by a medical oncologist, geriatrician, clinical pharmacist, social worker, and dietician. As part of this assessment, the patients brought in the contents of their medicine cabinets, and the medications they actively used were reviewed and recorded.
A total of 234 patients were included in the final analysis. Their mean age was 79.9 (range, 61–98). Most (87%) had solid tumor malignancies, were Caucasian (74%), and were female (64%).
In all, 26.5% of patients (n=62) had taken at least 1 CAM during their cancer care, with 19.2% taking 1 CAM, 6.4% taking 2, 0.4% taking 3, and 0.4% taking 4 or more CAMs. The highest number of CAMs taken was 10.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Comorbidities significantly associated with CAM use were vision impairment (P=0.048) and urologic comorbidities (P=0.021). Polypharmacy (concurrent use of 5 or more medications) was significantly associated with CAM use as well (P=0.045).
Some of the commonly used CAMs were mega-dose vitamins or minerals, as well as treatments for macular degeneration, stomach probiotics, and joint health.
The researchers did not examine the potential adverse effects of these medications, but Dr Nightingale said some are known to have a biochemical effect on the body and other drugs.
“It is very important to do a comprehensive screen of all of the medications that older cancer patients take, including CAMs,” she added. “Clear and transparent documentation of CAM use should be recorded in the patient’s medical record. This documentation should indicate that patient-specific communication and/or education was provided so that shared and informed decisions by the patient can be made regarding the continued use of these medications.” ![]()

Photo by Rhoda Baer
A new study suggests that seniors with cancer may be taking complementary or alternative medicines (CAMs) without their oncologists’ knowledge.
In this single-center study, 27% of senior cancer patients took CAMs at some point during their cancer care.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Polypharmacy and certain comorbidities were linked to CAM use as well.
Researchers reported these findings in the Journal of Geriatric Oncology.
“Currently, few oncologists are aware of the alternative medicines their patients take,” said study author Ginah Nightingale, PharmD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“Patients often fail to disclose the CAMs they take because they think they are safe, natural, nontoxic, and not relevant to their cancer care; because they think their doctor will disapprove; or because the doctor doesn’t specifically ask.”
To quantify CAM use in older cancer patients treated at their institution, Dr Nightingale and her colleagues surveyed patients who came to the Senior Adult Oncology Center at Thomas Jefferson University.
In a single visit, patients were seen by a medical oncologist, geriatrician, clinical pharmacist, social worker, and dietician. As part of this assessment, the patients brought in the contents of their medicine cabinets, and the medications they actively used were reviewed and recorded.
A total of 234 patients were included in the final analysis. Their mean age was 79.9 (range, 61–98). Most (87%) had solid tumor malignancies, were Caucasian (74%), and were female (64%).
In all, 26.5% of patients (n=62) had taken at least 1 CAM during their cancer care, with 19.2% taking 1 CAM, 6.4% taking 2, 0.4% taking 3, and 0.4% taking 4 or more CAMs. The highest number of CAMs taken was 10.
CAM usage was highest among patients ages 80 to 89, women, Caucasians, and patients with solid tumor malignancies.
Comorbidities significantly associated with CAM use were vision impairment (P=0.048) and urologic comorbidities (P=0.021). Polypharmacy (concurrent use of 5 or more medications) was significantly associated with CAM use as well (P=0.045).
Some of the commonly used CAMs were mega-dose vitamins or minerals, as well as treatments for macular degeneration, stomach probiotics, and joint health.
The researchers did not examine the potential adverse effects of these medications, but Dr Nightingale said some are known to have a biochemical effect on the body and other drugs.
“It is very important to do a comprehensive screen of all of the medications that older cancer patients take, including CAMs,” she added. “Clear and transparent documentation of CAM use should be recorded in the patient’s medical record. This documentation should indicate that patient-specific communication and/or education was provided so that shared and informed decisions by the patient can be made regarding the continued use of these medications.” ![]()
Genomic oncology: moving beyond the tip of the iceberg
In the 15 years since the first map of the human genome emerged, genetics has become an integral part of medical practice worldwide.1 Oncology is no exception; the genetic origins of cancer were suspected more than a century ago and it is now well understood that most cancers are driven by genetic alterations that disrupt key cellular pathways involved in tumor survival and progression.2
In the 15 years since the first map of the human genome emerged, genetics has become an integral part of medical practice worldwide.1 Oncology is no exception; the genetic origins of cancer were suspected more than a century ago and it is now well understood that most cancers are driven by genetic alterations that disrupt key cellular pathways involved in tumor survival and progression.2
In the 15 years since the first map of the human genome emerged, genetics has become an integral part of medical practice worldwide.1 Oncology is no exception; the genetic origins of cancer were suspected more than a century ago and it is now well understood that most cancers are driven by genetic alterations that disrupt key cellular pathways involved in tumor survival and progression.2