User login
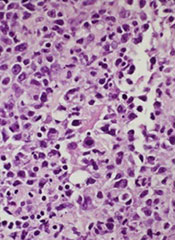
Study reveals potential drivers of CLL

Photo courtesy of Dana-
Farber Cancer Institute
Researchers say they have identified dozens of genetic abnormalities that may drive chronic lymphocytic leukemia (CLL), including some that were never before linked to human cancer.
The team began to trace how some of these abnormalities affect the course of the disease and its susceptibility to treatment.
And they started tracking the evolutionary path of CLL as its genome spawns new groups and subgroups of tumor cells in a single patient.
“Sequencing the DNA of CLL has taught us a great deal about the genetic basis of the disease,” said Catherine Wu, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts.
“Previous studies, however, were limited by the relatively small number of tumor tissue samples analyzed and by the fact that those samples were taken at different stages of the treatment process from patients treated with different drug agents.”
“In our new study, we wanted to determine if analyzing tissue samples from a large, similarly treated group of patients provides the statistical power necessary to study the disease in all its genetic diversity, to draw connections between certain mutations and the aggressiveness of the disease, and to chart the emergence of new mutations and their role in helping the disease advance. Our results demonstrate the range of insights to be gained by this approach.”
Dr Wu and her colleagues reported these results in Nature.
The researchers collected tumor and normal tissue samples from 538 patients with CLL and performed whole-exome sequencing on each sample.
In this way, the team identified 44 putative CLL driver genes, including 18 CLL mutated drivers that were previously identified and 26 additional putative CLL genes. About 34% of the CLL samples harbored a mutation in at least 1 of these 26 genes.
Nearly 9% of the patients had mutations in CLL genes in the MAPK-ERK pathway. The researchers therefore believe that further exploration of MAPK-ERK pathway inhibitors may be warranted.
The team found that mutations differed between IGHV-mutated and unmutated CLL. IGHV-unmutated CLL tended to have a higher proportion of most driving mutations, while only 3 driver genes were enriched in IGHV-mutated CLL—del(13q), MYD88, and CHD2.
The researchers also discovered that certain mutations were common in patients who had already undergone treatment.
Previous treatment was associated with enrichment in TP53 and BIRC3 mutations del(17p) and del(11q), as well as in mutated DDX3X and MAP2K1. The team therefore believes these mutations may help CLL rebound after initial therapy.
Another key finding was that therapy tended to produce shorter remissions in patients with mutations in TP53 or SF3B1.
“We found that genomic evolution after therapy is the rule rather than the exception,” Dr Wu noted. “Certain mutations were present in a greater number of leukemia cells within a sample after relapse, showing that these mutations, presumably, allow the tumor to persevere.” ![]()

Photo courtesy of Dana-
Farber Cancer Institute
Researchers say they have identified dozens of genetic abnormalities that may drive chronic lymphocytic leukemia (CLL), including some that were never before linked to human cancer.
The team began to trace how some of these abnormalities affect the course of the disease and its susceptibility to treatment.
And they started tracking the evolutionary path of CLL as its genome spawns new groups and subgroups of tumor cells in a single patient.
“Sequencing the DNA of CLL has taught us a great deal about the genetic basis of the disease,” said Catherine Wu, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts.
“Previous studies, however, were limited by the relatively small number of tumor tissue samples analyzed and by the fact that those samples were taken at different stages of the treatment process from patients treated with different drug agents.”
“In our new study, we wanted to determine if analyzing tissue samples from a large, similarly treated group of patients provides the statistical power necessary to study the disease in all its genetic diversity, to draw connections between certain mutations and the aggressiveness of the disease, and to chart the emergence of new mutations and their role in helping the disease advance. Our results demonstrate the range of insights to be gained by this approach.”
Dr Wu and her colleagues reported these results in Nature.
The researchers collected tumor and normal tissue samples from 538 patients with CLL and performed whole-exome sequencing on each sample.
In this way, the team identified 44 putative CLL driver genes, including 18 CLL mutated drivers that were previously identified and 26 additional putative CLL genes. About 34% of the CLL samples harbored a mutation in at least 1 of these 26 genes.
Nearly 9% of the patients had mutations in CLL genes in the MAPK-ERK pathway. The researchers therefore believe that further exploration of MAPK-ERK pathway inhibitors may be warranted.
The team found that mutations differed between IGHV-mutated and unmutated CLL. IGHV-unmutated CLL tended to have a higher proportion of most driving mutations, while only 3 driver genes were enriched in IGHV-mutated CLL—del(13q), MYD88, and CHD2.
The researchers also discovered that certain mutations were common in patients who had already undergone treatment.
Previous treatment was associated with enrichment in TP53 and BIRC3 mutations del(17p) and del(11q), as well as in mutated DDX3X and MAP2K1. The team therefore believes these mutations may help CLL rebound after initial therapy.
Another key finding was that therapy tended to produce shorter remissions in patients with mutations in TP53 or SF3B1.
“We found that genomic evolution after therapy is the rule rather than the exception,” Dr Wu noted. “Certain mutations were present in a greater number of leukemia cells within a sample after relapse, showing that these mutations, presumably, allow the tumor to persevere.” ![]()

Photo courtesy of Dana-
Farber Cancer Institute
Researchers say they have identified dozens of genetic abnormalities that may drive chronic lymphocytic leukemia (CLL), including some that were never before linked to human cancer.
The team began to trace how some of these abnormalities affect the course of the disease and its susceptibility to treatment.
And they started tracking the evolutionary path of CLL as its genome spawns new groups and subgroups of tumor cells in a single patient.
“Sequencing the DNA of CLL has taught us a great deal about the genetic basis of the disease,” said Catherine Wu, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts.
“Previous studies, however, were limited by the relatively small number of tumor tissue samples analyzed and by the fact that those samples were taken at different stages of the treatment process from patients treated with different drug agents.”
“In our new study, we wanted to determine if analyzing tissue samples from a large, similarly treated group of patients provides the statistical power necessary to study the disease in all its genetic diversity, to draw connections between certain mutations and the aggressiveness of the disease, and to chart the emergence of new mutations and their role in helping the disease advance. Our results demonstrate the range of insights to be gained by this approach.”
Dr Wu and her colleagues reported these results in Nature.
The researchers collected tumor and normal tissue samples from 538 patients with CLL and performed whole-exome sequencing on each sample.
In this way, the team identified 44 putative CLL driver genes, including 18 CLL mutated drivers that were previously identified and 26 additional putative CLL genes. About 34% of the CLL samples harbored a mutation in at least 1 of these 26 genes.
Nearly 9% of the patients had mutations in CLL genes in the MAPK-ERK pathway. The researchers therefore believe that further exploration of MAPK-ERK pathway inhibitors may be warranted.
The team found that mutations differed between IGHV-mutated and unmutated CLL. IGHV-unmutated CLL tended to have a higher proportion of most driving mutations, while only 3 driver genes were enriched in IGHV-mutated CLL—del(13q), MYD88, and CHD2.
The researchers also discovered that certain mutations were common in patients who had already undergone treatment.
Previous treatment was associated with enrichment in TP53 and BIRC3 mutations del(17p) and del(11q), as well as in mutated DDX3X and MAP2K1. The team therefore believes these mutations may help CLL rebound after initial therapy.
Another key finding was that therapy tended to produce shorter remissions in patients with mutations in TP53 or SF3B1.
“We found that genomic evolution after therapy is the rule rather than the exception,” Dr Wu noted. “Certain mutations were present in a greater number of leukemia cells within a sample after relapse, showing that these mutations, presumably, allow the tumor to persevere.” ![]()
COMP recommends orphan designations for KTE-C19

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) has adopted positive opinions recommending orphan designation for KTE-C19 to treat acute lymphoblastic leukemia, chronic lymphocytic leukemia/small lymphocytic lymphoma, and follicular lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the surface of B cells.
The CAR T-cell therapy already has orphan designation for the treatment of diffuse large B-cell lymphoma in the US and the European Union (EU).
KTE-C19 also has COMP positive opinions for orphan designation in the EU for primary mediastinal B-cell lymphoma and mantle cell lymphoma.
About orphan designation
The COMP adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required.
KTE-C19 research
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of the CAR T-cell therapy. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory diffuse large B-cell lymphoma, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
KTE-C19 is currently under investigation in a phase 1/2 trial (ZUMA-1) of patients with refractory, aggressive non-Hodgkin lymphomas. Kite Pharma, Inc., the company developing KTE-C19, plans to present top-line phase 1 data at the 2015 ASH Annual Meeting. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) has adopted positive opinions recommending orphan designation for KTE-C19 to treat acute lymphoblastic leukemia, chronic lymphocytic leukemia/small lymphocytic lymphoma, and follicular lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the surface of B cells.
The CAR T-cell therapy already has orphan designation for the treatment of diffuse large B-cell lymphoma in the US and the European Union (EU).
KTE-C19 also has COMP positive opinions for orphan designation in the EU for primary mediastinal B-cell lymphoma and mantle cell lymphoma.
About orphan designation
The COMP adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required.
KTE-C19 research
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of the CAR T-cell therapy. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory diffuse large B-cell lymphoma, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
KTE-C19 is currently under investigation in a phase 1/2 trial (ZUMA-1) of patients with refractory, aggressive non-Hodgkin lymphomas. Kite Pharma, Inc., the company developing KTE-C19, plans to present top-line phase 1 data at the 2015 ASH Annual Meeting. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) has adopted positive opinions recommending orphan designation for KTE-C19 to treat acute lymphoblastic leukemia, chronic lymphocytic leukemia/small lymphocytic lymphoma, and follicular lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the surface of B cells.
The CAR T-cell therapy already has orphan designation for the treatment of diffuse large B-cell lymphoma in the US and the European Union (EU).
KTE-C19 also has COMP positive opinions for orphan designation in the EU for primary mediastinal B-cell lymphoma and mantle cell lymphoma.
About orphan designation
The COMP adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required.
KTE-C19 research
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of the CAR T-cell therapy. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory diffuse large B-cell lymphoma, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
KTE-C19 is currently under investigation in a phase 1/2 trial (ZUMA-1) of patients with refractory, aggressive non-Hodgkin lymphomas. Kite Pharma, Inc., the company developing KTE-C19, plans to present top-line phase 1 data at the 2015 ASH Annual Meeting. ![]()
MMF may increase risk of CNS lymphoma
A new study has linked the immunosuppressive drug mycophenolate mofetil (MMF) to an increased risk of central nervous system (CNS) lymphoma in solid organ transplant recipients.
However, the research also suggests that calcineurin inhibitors (CNIs), when given alone or in combination with MMF, may protect transplant recipients from CNS lymphoma.
Researchers reported these findings in Oncotarget.
“MMF remains one of the best current medications for immunosuppression that we have,” said study author Amy Duffield, MD, PhD, of The Johns Hopkins Medical Institutions in Baltimore, Maryland.
“But a better understanding of its association with CNS lymphoproliferative disease will be crucial to further improving patients’ transplant regimens based on all of the risks these patients face.”
Dr Duffield and her colleagues noted that lymphomas and leukemias are known to be complications of solid organ transplants, but these malignancies rarely start in the CNS.
Still, in recent years, clinicians have begun to notice a rise in primary CNS lymphoproliferative disorders among transplant recipients. The current study is thought to be the first large enough to identify a link between MMF and these tumors.
For this work, Dr Duffield and her colleagues analyzed information on 177 patients with post-transplant lymphoproliferative disorder (PTLD) who were seen at Johns Hopkins Hospital between 1986 and 2014.
In that group, 29 patients—mostly kidney transplant recipients—were diagnosed with primary CNS lymphoproliferative disorders. The researchers said these were predominantly classified as monomorphic PTLD (72%), and most of the classifiable lymphomas were large B-cell lymphomas.
There were no cases of primary CNS PTLD diagnosed between 1986 and 1997, but the diagnosis increased markedly in the next decades.
The proportion of primary CNS PTLD cases compared to other PTLDs was 4.4-fold higher in the period from 2005 to 2014 than in the period from 1995 to 2004 (P<0.0001), even though the total number of PTLD cases remained relatively stable over time.
The researchers had prescription records on 16 of the patients who developed primary CNS lymphoproliferative disease.
Fifteen of the 16 patients had been taking MMF in the year prior to, or at the time of, their PTLD diagnosis. On the other hand, 37 of the 102 patients with PTLD outside the CNS had taken MMF (P<0.001).
The researchers also found that patients who took CNIs, either alone or in combination with MMF, seemed to be protected from developing primary CNS disease.
Primary CNS lymphoproliferative disease accounted for 66.7% of PTLDs among patients who took MMF but not a CNI (n=6), 23.9% of PTLDs among patients who took both an MMF and a CNI (n=46), and 1.7% of PTLDs among patients who took just a CNI (n=60).
The researchers found similar trends in a set of 6966 patients with PTLD. Those patients’ records were gleaned from an organ transplant database managed by the Organ Procurement and Transplantation Network and the United Network for Organ Sharing.
“More research needs to be done to confirm our results,” said Genevieve Crane, MD, PhD, of The Johns Hopkins Medical Institutions.
“But our work suggests that, at least in some patients, the combination of MMF and CNIs may be protective against CNS lymphoproliferative disease in a way that had not previously been appreciated.” ![]()

A new study has linked the immunosuppressive drug mycophenolate mofetil (MMF) to an increased risk of central nervous system (CNS) lymphoma in solid organ transplant recipients.
However, the research also suggests that calcineurin inhibitors (CNIs), when given alone or in combination with MMF, may protect transplant recipients from CNS lymphoma.
Researchers reported these findings in Oncotarget.
“MMF remains one of the best current medications for immunosuppression that we have,” said study author Amy Duffield, MD, PhD, of The Johns Hopkins Medical Institutions in Baltimore, Maryland.
“But a better understanding of its association with CNS lymphoproliferative disease will be crucial to further improving patients’ transplant regimens based on all of the risks these patients face.”
Dr Duffield and her colleagues noted that lymphomas and leukemias are known to be complications of solid organ transplants, but these malignancies rarely start in the CNS.
Still, in recent years, clinicians have begun to notice a rise in primary CNS lymphoproliferative disorders among transplant recipients. The current study is thought to be the first large enough to identify a link between MMF and these tumors.
For this work, Dr Duffield and her colleagues analyzed information on 177 patients with post-transplant lymphoproliferative disorder (PTLD) who were seen at Johns Hopkins Hospital between 1986 and 2014.
In that group, 29 patients—mostly kidney transplant recipients—were diagnosed with primary CNS lymphoproliferative disorders. The researchers said these were predominantly classified as monomorphic PTLD (72%), and most of the classifiable lymphomas were large B-cell lymphomas.
There were no cases of primary CNS PTLD diagnosed between 1986 and 1997, but the diagnosis increased markedly in the next decades.
The proportion of primary CNS PTLD cases compared to other PTLDs was 4.4-fold higher in the period from 2005 to 2014 than in the period from 1995 to 2004 (P<0.0001), even though the total number of PTLD cases remained relatively stable over time.
The researchers had prescription records on 16 of the patients who developed primary CNS lymphoproliferative disease.
Fifteen of the 16 patients had been taking MMF in the year prior to, or at the time of, their PTLD diagnosis. On the other hand, 37 of the 102 patients with PTLD outside the CNS had taken MMF (P<0.001).
The researchers also found that patients who took CNIs, either alone or in combination with MMF, seemed to be protected from developing primary CNS disease.
Primary CNS lymphoproliferative disease accounted for 66.7% of PTLDs among patients who took MMF but not a CNI (n=6), 23.9% of PTLDs among patients who took both an MMF and a CNI (n=46), and 1.7% of PTLDs among patients who took just a CNI (n=60).
The researchers found similar trends in a set of 6966 patients with PTLD. Those patients’ records were gleaned from an organ transplant database managed by the Organ Procurement and Transplantation Network and the United Network for Organ Sharing.
“More research needs to be done to confirm our results,” said Genevieve Crane, MD, PhD, of The Johns Hopkins Medical Institutions.
“But our work suggests that, at least in some patients, the combination of MMF and CNIs may be protective against CNS lymphoproliferative disease in a way that had not previously been appreciated.” ![]()

A new study has linked the immunosuppressive drug mycophenolate mofetil (MMF) to an increased risk of central nervous system (CNS) lymphoma in solid organ transplant recipients.
However, the research also suggests that calcineurin inhibitors (CNIs), when given alone or in combination with MMF, may protect transplant recipients from CNS lymphoma.
Researchers reported these findings in Oncotarget.
“MMF remains one of the best current medications for immunosuppression that we have,” said study author Amy Duffield, MD, PhD, of The Johns Hopkins Medical Institutions in Baltimore, Maryland.
“But a better understanding of its association with CNS lymphoproliferative disease will be crucial to further improving patients’ transplant regimens based on all of the risks these patients face.”
Dr Duffield and her colleagues noted that lymphomas and leukemias are known to be complications of solid organ transplants, but these malignancies rarely start in the CNS.
Still, in recent years, clinicians have begun to notice a rise in primary CNS lymphoproliferative disorders among transplant recipients. The current study is thought to be the first large enough to identify a link between MMF and these tumors.
For this work, Dr Duffield and her colleagues analyzed information on 177 patients with post-transplant lymphoproliferative disorder (PTLD) who were seen at Johns Hopkins Hospital between 1986 and 2014.
In that group, 29 patients—mostly kidney transplant recipients—were diagnosed with primary CNS lymphoproliferative disorders. The researchers said these were predominantly classified as monomorphic PTLD (72%), and most of the classifiable lymphomas were large B-cell lymphomas.
There were no cases of primary CNS PTLD diagnosed between 1986 and 1997, but the diagnosis increased markedly in the next decades.
The proportion of primary CNS PTLD cases compared to other PTLDs was 4.4-fold higher in the period from 2005 to 2014 than in the period from 1995 to 2004 (P<0.0001), even though the total number of PTLD cases remained relatively stable over time.
The researchers had prescription records on 16 of the patients who developed primary CNS lymphoproliferative disease.
Fifteen of the 16 patients had been taking MMF in the year prior to, or at the time of, their PTLD diagnosis. On the other hand, 37 of the 102 patients with PTLD outside the CNS had taken MMF (P<0.001).
The researchers also found that patients who took CNIs, either alone or in combination with MMF, seemed to be protected from developing primary CNS disease.
Primary CNS lymphoproliferative disease accounted for 66.7% of PTLDs among patients who took MMF but not a CNI (n=6), 23.9% of PTLDs among patients who took both an MMF and a CNI (n=46), and 1.7% of PTLDs among patients who took just a CNI (n=60).
The researchers found similar trends in a set of 6966 patients with PTLD. Those patients’ records were gleaned from an organ transplant database managed by the Organ Procurement and Transplantation Network and the United Network for Organ Sharing.
“More research needs to be done to confirm our results,” said Genevieve Crane, MD, PhD, of The Johns Hopkins Medical Institutions.
“But our work suggests that, at least in some patients, the combination of MMF and CNIs may be protective against CNS lymphoproliferative disease in a way that had not previously been appreciated.” ![]()
Malarial protein is basis for potential cancer therapy

Photo by Vivian Sum
A protein expressed by the malaria parasite Plasmodium falciparum may prove useful for treating a range of cancers, according to research published in Cancer Cell.
This protein, VAR2CSA, binds a chondroitin sulfate (CS) that is found in placenta but is also present in many different cancer cells.
So investigators combined recombinant VAR2CSA (rVAR2) with 2 different toxins to create cancer-targeting treatments.
The treatments effectively targeted cancers in vitro and in vivo, impeding tumor growth and even eradicating cancer in some mice.
An idea is born
This research was born while the investigators were exploring why pregnant women are particularly susceptible to malaria. The team found that P falciparum produces VAR2CSA, which binds to a particular CS in the placenta, and that placenta-like CS (pl-CS) is found in most cancers.
This suggested the pl-CS could be a target for anticancer drugs, and VAR2CSA could provide the tool for carrying such drugs to tumors.
“Scientists have spent decades trying to find biochemical similarities between placenta tissue and cancer, but we just didn’t have the technology to find it,” said project leader Mads Daugaard, PhD, of the University of British Columbia in Vancouver, Canada.
“When my colleagues discovered how malaria uses VAR2CSA to embed itself in the placenta, we immediately saw its potential to deliver cancer drugs in a precise, controlled way to tumors.”
Testing rVAR2
After demonstrating that rVAR2 binds only to pl-CS, the investigators tested rVAR2 in patient-derived cancer cell lines of hematopoietic, epithelial, and mesenchymal origin. rVAR2 reacted with 95% (106/111) of these cell lines.
To determine whether rVAR2 could be used as a pl-CS-specific tumor-targeting system, the investigators fused the cytotoxic domain of diphtheria toxin (DT388) to rVAR2, creating a recombinant rDT388-VAR2 (rVAR2-DT) fusion protein.
The rVAR2-DT protein killed tumor cell lines of epithelial and mesenchymal origin, but it had no effect on normal primary human endothelial cells.
The investigators also tested rVAR2-DT in mouse models of prostate cancer and found that as few as 3 doses were enough to significantly inhibit tumor growth.
However, the team noted that clinical trials with DT fusions have shown that high drug concentrations are not well-tolerated.
So they chemically conjugated a hemiasterlin analog (KT886) to rVAR2 via a protease cleavable linker. The rVAR2-KT886 drug conjugate (VDC886) carried an average of 3 toxins per rVAR2 molecule.
The investigators tested VDC886 in 33 cancer cell lines and observed cytotoxicity in all cell lines.
So the team went on to test VDC886 in mouse models of non-Hodgkin lymphoma, prostate cancer, and metastatic breast cancer. VDC886 significantly inhibited tumor growth in all 3 models.
In mice with non-Hodgkin lymphoma, treated tumors were about a quarter the size of tumors in control mice. For the mice with prostate cancer, tumors completely disappeared in 2 of the 6 treated mice a month after they received the first dose of VDC886.
In mice with metastatic breast cancer, 5 of the 6 treated mice were cured and alive after almost 8 weeks. None of the control mice with metastatic breast cancer survived that long.
The investigators said they did not observe any adverse effects in the mice, and their organs were unharmed by the therapy.
“It appears that the malaria protein attaches itself to the tumor without any significant attachment to other tissue,” said Thomas Mandel Clausen, a PhD student at the University of Copenhagen in Denmark.
“And the mice that were given doses of protein and toxin showed far higher survival rates than the untreated mice. We have seen that 3 doses can arrest growth in a tumor and even make it shrink.”
Based on these results, 2 companies—Vancouver-based Kairos Therapeutics and Copenhagen-based VAR2 Pharmaceuticals—are developing the compound for clinical trials. The investigators believe this will take a few years. ![]()

Photo by Vivian Sum
A protein expressed by the malaria parasite Plasmodium falciparum may prove useful for treating a range of cancers, according to research published in Cancer Cell.
This protein, VAR2CSA, binds a chondroitin sulfate (CS) that is found in placenta but is also present in many different cancer cells.
So investigators combined recombinant VAR2CSA (rVAR2) with 2 different toxins to create cancer-targeting treatments.
The treatments effectively targeted cancers in vitro and in vivo, impeding tumor growth and even eradicating cancer in some mice.
An idea is born
This research was born while the investigators were exploring why pregnant women are particularly susceptible to malaria. The team found that P falciparum produces VAR2CSA, which binds to a particular CS in the placenta, and that placenta-like CS (pl-CS) is found in most cancers.
This suggested the pl-CS could be a target for anticancer drugs, and VAR2CSA could provide the tool for carrying such drugs to tumors.
“Scientists have spent decades trying to find biochemical similarities between placenta tissue and cancer, but we just didn’t have the technology to find it,” said project leader Mads Daugaard, PhD, of the University of British Columbia in Vancouver, Canada.
“When my colleagues discovered how malaria uses VAR2CSA to embed itself in the placenta, we immediately saw its potential to deliver cancer drugs in a precise, controlled way to tumors.”
Testing rVAR2
After demonstrating that rVAR2 binds only to pl-CS, the investigators tested rVAR2 in patient-derived cancer cell lines of hematopoietic, epithelial, and mesenchymal origin. rVAR2 reacted with 95% (106/111) of these cell lines.
To determine whether rVAR2 could be used as a pl-CS-specific tumor-targeting system, the investigators fused the cytotoxic domain of diphtheria toxin (DT388) to rVAR2, creating a recombinant rDT388-VAR2 (rVAR2-DT) fusion protein.
The rVAR2-DT protein killed tumor cell lines of epithelial and mesenchymal origin, but it had no effect on normal primary human endothelial cells.
The investigators also tested rVAR2-DT in mouse models of prostate cancer and found that as few as 3 doses were enough to significantly inhibit tumor growth.
However, the team noted that clinical trials with DT fusions have shown that high drug concentrations are not well-tolerated.
So they chemically conjugated a hemiasterlin analog (KT886) to rVAR2 via a protease cleavable linker. The rVAR2-KT886 drug conjugate (VDC886) carried an average of 3 toxins per rVAR2 molecule.
The investigators tested VDC886 in 33 cancer cell lines and observed cytotoxicity in all cell lines.
So the team went on to test VDC886 in mouse models of non-Hodgkin lymphoma, prostate cancer, and metastatic breast cancer. VDC886 significantly inhibited tumor growth in all 3 models.
In mice with non-Hodgkin lymphoma, treated tumors were about a quarter the size of tumors in control mice. For the mice with prostate cancer, tumors completely disappeared in 2 of the 6 treated mice a month after they received the first dose of VDC886.
In mice with metastatic breast cancer, 5 of the 6 treated mice were cured and alive after almost 8 weeks. None of the control mice with metastatic breast cancer survived that long.
The investigators said they did not observe any adverse effects in the mice, and their organs were unharmed by the therapy.
“It appears that the malaria protein attaches itself to the tumor without any significant attachment to other tissue,” said Thomas Mandel Clausen, a PhD student at the University of Copenhagen in Denmark.
“And the mice that were given doses of protein and toxin showed far higher survival rates than the untreated mice. We have seen that 3 doses can arrest growth in a tumor and even make it shrink.”
Based on these results, 2 companies—Vancouver-based Kairos Therapeutics and Copenhagen-based VAR2 Pharmaceuticals—are developing the compound for clinical trials. The investigators believe this will take a few years. ![]()

Photo by Vivian Sum
A protein expressed by the malaria parasite Plasmodium falciparum may prove useful for treating a range of cancers, according to research published in Cancer Cell.
This protein, VAR2CSA, binds a chondroitin sulfate (CS) that is found in placenta but is also present in many different cancer cells.
So investigators combined recombinant VAR2CSA (rVAR2) with 2 different toxins to create cancer-targeting treatments.
The treatments effectively targeted cancers in vitro and in vivo, impeding tumor growth and even eradicating cancer in some mice.
An idea is born
This research was born while the investigators were exploring why pregnant women are particularly susceptible to malaria. The team found that P falciparum produces VAR2CSA, which binds to a particular CS in the placenta, and that placenta-like CS (pl-CS) is found in most cancers.
This suggested the pl-CS could be a target for anticancer drugs, and VAR2CSA could provide the tool for carrying such drugs to tumors.
“Scientists have spent decades trying to find biochemical similarities between placenta tissue and cancer, but we just didn’t have the technology to find it,” said project leader Mads Daugaard, PhD, of the University of British Columbia in Vancouver, Canada.
“When my colleagues discovered how malaria uses VAR2CSA to embed itself in the placenta, we immediately saw its potential to deliver cancer drugs in a precise, controlled way to tumors.”
Testing rVAR2
After demonstrating that rVAR2 binds only to pl-CS, the investigators tested rVAR2 in patient-derived cancer cell lines of hematopoietic, epithelial, and mesenchymal origin. rVAR2 reacted with 95% (106/111) of these cell lines.
To determine whether rVAR2 could be used as a pl-CS-specific tumor-targeting system, the investigators fused the cytotoxic domain of diphtheria toxin (DT388) to rVAR2, creating a recombinant rDT388-VAR2 (rVAR2-DT) fusion protein.
The rVAR2-DT protein killed tumor cell lines of epithelial and mesenchymal origin, but it had no effect on normal primary human endothelial cells.
The investigators also tested rVAR2-DT in mouse models of prostate cancer and found that as few as 3 doses were enough to significantly inhibit tumor growth.
However, the team noted that clinical trials with DT fusions have shown that high drug concentrations are not well-tolerated.
So they chemically conjugated a hemiasterlin analog (KT886) to rVAR2 via a protease cleavable linker. The rVAR2-KT886 drug conjugate (VDC886) carried an average of 3 toxins per rVAR2 molecule.
The investigators tested VDC886 in 33 cancer cell lines and observed cytotoxicity in all cell lines.
So the team went on to test VDC886 in mouse models of non-Hodgkin lymphoma, prostate cancer, and metastatic breast cancer. VDC886 significantly inhibited tumor growth in all 3 models.
In mice with non-Hodgkin lymphoma, treated tumors were about a quarter the size of tumors in control mice. For the mice with prostate cancer, tumors completely disappeared in 2 of the 6 treated mice a month after they received the first dose of VDC886.
In mice with metastatic breast cancer, 5 of the 6 treated mice were cured and alive after almost 8 weeks. None of the control mice with metastatic breast cancer survived that long.
The investigators said they did not observe any adverse effects in the mice, and their organs were unharmed by the therapy.
“It appears that the malaria protein attaches itself to the tumor without any significant attachment to other tissue,” said Thomas Mandel Clausen, a PhD student at the University of Copenhagen in Denmark.
“And the mice that were given doses of protein and toxin showed far higher survival rates than the untreated mice. We have seen that 3 doses can arrest growth in a tumor and even make it shrink.”
Based on these results, 2 companies—Vancouver-based Kairos Therapeutics and Copenhagen-based VAR2 Pharmaceuticals—are developing the compound for clinical trials. The investigators believe this will take a few years. ![]()
Bacterium may increase risk of DLBCL, FL
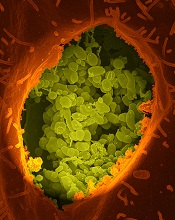
Coxiella burnetii
Image courtesy of NIAID
The bacterium that causes Q fever may confer an increased risk of diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL), according to a study published in Blood.
Q fever is an infectious disease caused by Coxiella burnetii, a bacterium primarily transmitted through the excrements of cattle, sheep, and goats.
Because some patients with Q fever develop lymphoma, researchers believed the malignancy could be a risk factor for Q fever.
However, the experience of 1 patient prompted a group of researchers in France to consider the opposite—that the infection might cause the lymphoma.
“During a follow-up scan in a patient we had successfully treated for Q fever, we observed a tumor close to the location of the previous infection,” said study author Didier Raoult, MD, PhD, of Aix-Marseille University in Marseille, France.
“The discovery that it was a lymphoma tumor containing C burnetii encouraged us to consider that the infection might have contributed to the development of the cancer.”
To better understand the association between C burnetii and lymphoma, Dr Raoult and his colleagues screened 1468 patients treated at the French National Referral Center for Q Fever from 2004 to 2014.
The researchers imaged patient tissue samples and identified 7 people, including the initial patient, who developed lymphoma after C burnetii infection. Six patients were diagnosed with DLBCL and 1 with FL.
To determine if patients with Q fever have a higher risk of lymphoma than the general population, the researchers compared the incidence of lymphoma in the Q fever registry to the incidence reported in France’s general population.
This revealed an excess risk of DLBCL and FL in the Q fever population, with standardized incidence ratios of 25.4 for DLBCL and 6.7 for FL.
In addition, the odds of developing lymphoma were higher in patients with persistent, concentrated infections than in those with other forms of Q fever. The hazard ratio for patients with persistent, concentrated infection was 9.35.
Finally, the researchers observed interleukin-10 overproduction in Q fever patients with lymphoma. The team said this suggests that suppression of the immune system may have allowed the lymphoma cells to evade immune detection and multiply.
“As we continue to learn more about the association between C burnetii and lymphoma, these results should encourage clinicians to survey high-risk patients as early as possible for potential cancer,” Dr Raoult said.
“Ultimately, this early diagnosis and treatment would improve outcomes for Q fever patients who subsequently develop lymphoma, particularly those with B-cell non-Hodgkin lymphoma.” ![]()

Coxiella burnetii
Image courtesy of NIAID
The bacterium that causes Q fever may confer an increased risk of diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL), according to a study published in Blood.
Q fever is an infectious disease caused by Coxiella burnetii, a bacterium primarily transmitted through the excrements of cattle, sheep, and goats.
Because some patients with Q fever develop lymphoma, researchers believed the malignancy could be a risk factor for Q fever.
However, the experience of 1 patient prompted a group of researchers in France to consider the opposite—that the infection might cause the lymphoma.
“During a follow-up scan in a patient we had successfully treated for Q fever, we observed a tumor close to the location of the previous infection,” said study author Didier Raoult, MD, PhD, of Aix-Marseille University in Marseille, France.
“The discovery that it was a lymphoma tumor containing C burnetii encouraged us to consider that the infection might have contributed to the development of the cancer.”
To better understand the association between C burnetii and lymphoma, Dr Raoult and his colleagues screened 1468 patients treated at the French National Referral Center for Q Fever from 2004 to 2014.
The researchers imaged patient tissue samples and identified 7 people, including the initial patient, who developed lymphoma after C burnetii infection. Six patients were diagnosed with DLBCL and 1 with FL.
To determine if patients with Q fever have a higher risk of lymphoma than the general population, the researchers compared the incidence of lymphoma in the Q fever registry to the incidence reported in France’s general population.
This revealed an excess risk of DLBCL and FL in the Q fever population, with standardized incidence ratios of 25.4 for DLBCL and 6.7 for FL.
In addition, the odds of developing lymphoma were higher in patients with persistent, concentrated infections than in those with other forms of Q fever. The hazard ratio for patients with persistent, concentrated infection was 9.35.
Finally, the researchers observed interleukin-10 overproduction in Q fever patients with lymphoma. The team said this suggests that suppression of the immune system may have allowed the lymphoma cells to evade immune detection and multiply.
“As we continue to learn more about the association between C burnetii and lymphoma, these results should encourage clinicians to survey high-risk patients as early as possible for potential cancer,” Dr Raoult said.
“Ultimately, this early diagnosis and treatment would improve outcomes for Q fever patients who subsequently develop lymphoma, particularly those with B-cell non-Hodgkin lymphoma.” ![]()

Coxiella burnetii
Image courtesy of NIAID
The bacterium that causes Q fever may confer an increased risk of diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL), according to a study published in Blood.
Q fever is an infectious disease caused by Coxiella burnetii, a bacterium primarily transmitted through the excrements of cattle, sheep, and goats.
Because some patients with Q fever develop lymphoma, researchers believed the malignancy could be a risk factor for Q fever.
However, the experience of 1 patient prompted a group of researchers in France to consider the opposite—that the infection might cause the lymphoma.
“During a follow-up scan in a patient we had successfully treated for Q fever, we observed a tumor close to the location of the previous infection,” said study author Didier Raoult, MD, PhD, of Aix-Marseille University in Marseille, France.
“The discovery that it was a lymphoma tumor containing C burnetii encouraged us to consider that the infection might have contributed to the development of the cancer.”
To better understand the association between C burnetii and lymphoma, Dr Raoult and his colleagues screened 1468 patients treated at the French National Referral Center for Q Fever from 2004 to 2014.
The researchers imaged patient tissue samples and identified 7 people, including the initial patient, who developed lymphoma after C burnetii infection. Six patients were diagnosed with DLBCL and 1 with FL.
To determine if patients with Q fever have a higher risk of lymphoma than the general population, the researchers compared the incidence of lymphoma in the Q fever registry to the incidence reported in France’s general population.
This revealed an excess risk of DLBCL and FL in the Q fever population, with standardized incidence ratios of 25.4 for DLBCL and 6.7 for FL.
In addition, the odds of developing lymphoma were higher in patients with persistent, concentrated infections than in those with other forms of Q fever. The hazard ratio for patients with persistent, concentrated infection was 9.35.
Finally, the researchers observed interleukin-10 overproduction in Q fever patients with lymphoma. The team said this suggests that suppression of the immune system may have allowed the lymphoma cells to evade immune detection and multiply.
“As we continue to learn more about the association between C burnetii and lymphoma, these results should encourage clinicians to survey high-risk patients as early as possible for potential cancer,” Dr Raoult said.
“Ultimately, this early diagnosis and treatment would improve outcomes for Q fever patients who subsequently develop lymphoma, particularly those with B-cell non-Hodgkin lymphoma.” ![]()
Cancer survivors have poor diets, study suggests

New research suggests that cancer survivors in the US may need dietary interventions to improve their health.
The study showed that, overall, cancer survivors did not adhere to federal dietary guidelines as well as a matched control population.
Cancer survivors tended to consume more empty calories and less fiber than controls.
Fang Fang Zhang, MD, PhD, of Tufts University in Boston, Massachusetts, and his colleagues reported these findings in Cancer.
The team evaluated the diets of 1533 adult cancer survivors who participated in the National Health and Nutrition Examination Survey from 1999 to 2010. The investigators also assessed the diets of 3075 individuals who had no history of cancer and were matched to the cancer survivors by age, sex, and race/ethnicity.
The goal was to determine how subjects’ diets aligned with advice from the 2010 Dietary Guidelines for Americans, which was jointly issued by the Department of Agriculture and the Department of Health and Human Services.
Cancer survivors had poor adherence to the guidelines, with a total Healthy Eating Index score of 47.2 out of 100, compared with a score of 48.3 in the adults without a history of cancer (P=0.03).
Cancer survivors had a significantly lower mean score for empty calories compared to the noncancer group—13.6 and 14.4, respectively (P=0.001). This suggested the cancer group had a higher consumption of calories from solid fats, alcohol, and added sugars.
Cancer survivors also had significantly lower dietary intake of fiber than the noncancer group—15.0 and 15.9 g per day, respectively (P=0.02).
Compared to recommended values, cancer survivors had low dietary intakes of vitamin D (31% of the recommended intake), vitamin E (47%), potassium (55%), and calcium (73%) but high intakes of saturated fat (112%) and sodium (133%).
The investigators noted that diet quality in cancer survivors increased linearly with age. The older the age, the better the diet quality.
Survivors with lower education (high school or less) had significantly worse diet quality than those with higher education. And survivors who were current smokers had significantly worse diet quality than non-smokers or former smokers.
For the 4 major cancer types in the US (breast, prostate, lung, and colorectal), breast cancer survivors had the best diet quality, and lung cancer survivors had the worst diet quality.
The investigators said that knowing how well cancer survivors adhere to federal dietary guidelines can help inform evidence-based priorities for improving their nutritional intake.
“Dietary changes that include more fiber, fruit, and vegetables in the diet and less fat, sodium, and added sugar would be important for cancer survivors,” Dr Zhang said.
“Oncology care providers can play critical roles in reinforcing the importance of a healthful diet and can refer patients to registered dietitians who are experts in oncology care or to other reputable sources in order to improve survivors’ overall health.” ![]()

New research suggests that cancer survivors in the US may need dietary interventions to improve their health.
The study showed that, overall, cancer survivors did not adhere to federal dietary guidelines as well as a matched control population.
Cancer survivors tended to consume more empty calories and less fiber than controls.
Fang Fang Zhang, MD, PhD, of Tufts University in Boston, Massachusetts, and his colleagues reported these findings in Cancer.
The team evaluated the diets of 1533 adult cancer survivors who participated in the National Health and Nutrition Examination Survey from 1999 to 2010. The investigators also assessed the diets of 3075 individuals who had no history of cancer and were matched to the cancer survivors by age, sex, and race/ethnicity.
The goal was to determine how subjects’ diets aligned with advice from the 2010 Dietary Guidelines for Americans, which was jointly issued by the Department of Agriculture and the Department of Health and Human Services.
Cancer survivors had poor adherence to the guidelines, with a total Healthy Eating Index score of 47.2 out of 100, compared with a score of 48.3 in the adults without a history of cancer (P=0.03).
Cancer survivors had a significantly lower mean score for empty calories compared to the noncancer group—13.6 and 14.4, respectively (P=0.001). This suggested the cancer group had a higher consumption of calories from solid fats, alcohol, and added sugars.
Cancer survivors also had significantly lower dietary intake of fiber than the noncancer group—15.0 and 15.9 g per day, respectively (P=0.02).
Compared to recommended values, cancer survivors had low dietary intakes of vitamin D (31% of the recommended intake), vitamin E (47%), potassium (55%), and calcium (73%) but high intakes of saturated fat (112%) and sodium (133%).
The investigators noted that diet quality in cancer survivors increased linearly with age. The older the age, the better the diet quality.
Survivors with lower education (high school or less) had significantly worse diet quality than those with higher education. And survivors who were current smokers had significantly worse diet quality than non-smokers or former smokers.
For the 4 major cancer types in the US (breast, prostate, lung, and colorectal), breast cancer survivors had the best diet quality, and lung cancer survivors had the worst diet quality.
The investigators said that knowing how well cancer survivors adhere to federal dietary guidelines can help inform evidence-based priorities for improving their nutritional intake.
“Dietary changes that include more fiber, fruit, and vegetables in the diet and less fat, sodium, and added sugar would be important for cancer survivors,” Dr Zhang said.
“Oncology care providers can play critical roles in reinforcing the importance of a healthful diet and can refer patients to registered dietitians who are experts in oncology care or to other reputable sources in order to improve survivors’ overall health.” ![]()

New research suggests that cancer survivors in the US may need dietary interventions to improve their health.
The study showed that, overall, cancer survivors did not adhere to federal dietary guidelines as well as a matched control population.
Cancer survivors tended to consume more empty calories and less fiber than controls.
Fang Fang Zhang, MD, PhD, of Tufts University in Boston, Massachusetts, and his colleagues reported these findings in Cancer.
The team evaluated the diets of 1533 adult cancer survivors who participated in the National Health and Nutrition Examination Survey from 1999 to 2010. The investigators also assessed the diets of 3075 individuals who had no history of cancer and were matched to the cancer survivors by age, sex, and race/ethnicity.
The goal was to determine how subjects’ diets aligned with advice from the 2010 Dietary Guidelines for Americans, which was jointly issued by the Department of Agriculture and the Department of Health and Human Services.
Cancer survivors had poor adherence to the guidelines, with a total Healthy Eating Index score of 47.2 out of 100, compared with a score of 48.3 in the adults without a history of cancer (P=0.03).
Cancer survivors had a significantly lower mean score for empty calories compared to the noncancer group—13.6 and 14.4, respectively (P=0.001). This suggested the cancer group had a higher consumption of calories from solid fats, alcohol, and added sugars.
Cancer survivors also had significantly lower dietary intake of fiber than the noncancer group—15.0 and 15.9 g per day, respectively (P=0.02).
Compared to recommended values, cancer survivors had low dietary intakes of vitamin D (31% of the recommended intake), vitamin E (47%), potassium (55%), and calcium (73%) but high intakes of saturated fat (112%) and sodium (133%).
The investigators noted that diet quality in cancer survivors increased linearly with age. The older the age, the better the diet quality.
Survivors with lower education (high school or less) had significantly worse diet quality than those with higher education. And survivors who were current smokers had significantly worse diet quality than non-smokers or former smokers.
For the 4 major cancer types in the US (breast, prostate, lung, and colorectal), breast cancer survivors had the best diet quality, and lung cancer survivors had the worst diet quality.
The investigators said that knowing how well cancer survivors adhere to federal dietary guidelines can help inform evidence-based priorities for improving their nutritional intake.
“Dietary changes that include more fiber, fruit, and vegetables in the diet and less fat, sodium, and added sugar would be important for cancer survivors,” Dr Zhang said.
“Oncology care providers can play critical roles in reinforcing the importance of a healthful diet and can refer patients to registered dietitians who are experts in oncology care or to other reputable sources in order to improve survivors’ overall health.” ![]()
Baseline factors predict early response to multiple myeloma therapy
CHICAGO – Age and the results of select baseline lab measures reflected early response to initial therapy in a review of 1,304 newly diagnosed patients with symptomatic multiple myeloma.
Characteristics associated with increased odds of achieving very good partial response or better (VGPR+) within four cycles of treatment in the study participants, who were seen between 2001 and 2013, were absolute free light chain difference of more than 175 mg/dL (odds ratio, 2.38), age less than 75 years at diagnosis (OR, 2.18), hemoglobin concentration less than 10/12.5 (OR, 1.68), and IgA vs. IgG serum heavy chain secretion (OR, 1.66), Dr. Moritz Binder of the Mayo Clinic, Rochester, Minn., reported in a poster at the American Society of Hematology Meeting on Hematologic Malignancies.
In patients receiving proteasome inhibitors, better response was associated with creatinine concentrations greater than 1.5 mg/dL (OR, 3.83), calcium concentration less than 9.0 mg/dL (OR, 3.37), and greater absolute free light chain difference greater than 175 mg/dL (OR, 2.50), Dr. Binder noted.
High-risk cytogenetic features, conversely, were not associated with treatment response, he said.
The findings are important because the initial response to therapy in newly diagnosed disease can have an effect on long-term outcomes; achieving at least a VGPR to initial treatment can improve progression-free and overall survival. Further, novel agents and risk-adapted treatment strategies now in use have improved response rates and overall survival, Dr. Binder said, adding that it has been unclear, however, whether baseline laboratory parameters could predict the likelihood of early, deep response to initial therapy.
“The ability to predict the likelihood of response to the current therapies can have implications for the treatment approaches in NDMM,” he wrote.
In this study, 288 patients achieved VGPR+ after 4 months, and those patients had a decreased risk of subsequent mortality (hazard ratio, 0.69). This remained true after adjusting for sex, age, International Staging System stage, bone marrow plasma cell involvement, lactate dehydrogenase concentration, initial treatment regimen group, and hematopoietic stem cell transplantation during the disease course (HR, 0.68), he said.
The three most common regimens with immunomodulators that were used in the cohort were lenalidomide-dexamethasone, thalidomide-dexamethasone, and cyclophosphamide-lenalidomide-dexamethasone, and the three most common regimens with proteasome inhibitors were bortezomib-cyclophosphamide-dexamethasone, bortezomib-lenalidomide-dexamethasone, and bortezomib-dexamethasone.
Dr. Binder reported receiving research funding from ASH. Coauthors reported receiving research funding from Celgene, Janssen, Millennium, and Pfizer, and/or serving in an advisory role for Pfizer.
CHICAGO – Age and the results of select baseline lab measures reflected early response to initial therapy in a review of 1,304 newly diagnosed patients with symptomatic multiple myeloma.
Characteristics associated with increased odds of achieving very good partial response or better (VGPR+) within four cycles of treatment in the study participants, who were seen between 2001 and 2013, were absolute free light chain difference of more than 175 mg/dL (odds ratio, 2.38), age less than 75 years at diagnosis (OR, 2.18), hemoglobin concentration less than 10/12.5 (OR, 1.68), and IgA vs. IgG serum heavy chain secretion (OR, 1.66), Dr. Moritz Binder of the Mayo Clinic, Rochester, Minn., reported in a poster at the American Society of Hematology Meeting on Hematologic Malignancies.
In patients receiving proteasome inhibitors, better response was associated with creatinine concentrations greater than 1.5 mg/dL (OR, 3.83), calcium concentration less than 9.0 mg/dL (OR, 3.37), and greater absolute free light chain difference greater than 175 mg/dL (OR, 2.50), Dr. Binder noted.
High-risk cytogenetic features, conversely, were not associated with treatment response, he said.
The findings are important because the initial response to therapy in newly diagnosed disease can have an effect on long-term outcomes; achieving at least a VGPR to initial treatment can improve progression-free and overall survival. Further, novel agents and risk-adapted treatment strategies now in use have improved response rates and overall survival, Dr. Binder said, adding that it has been unclear, however, whether baseline laboratory parameters could predict the likelihood of early, deep response to initial therapy.
“The ability to predict the likelihood of response to the current therapies can have implications for the treatment approaches in NDMM,” he wrote.
In this study, 288 patients achieved VGPR+ after 4 months, and those patients had a decreased risk of subsequent mortality (hazard ratio, 0.69). This remained true after adjusting for sex, age, International Staging System stage, bone marrow plasma cell involvement, lactate dehydrogenase concentration, initial treatment regimen group, and hematopoietic stem cell transplantation during the disease course (HR, 0.68), he said.
The three most common regimens with immunomodulators that were used in the cohort were lenalidomide-dexamethasone, thalidomide-dexamethasone, and cyclophosphamide-lenalidomide-dexamethasone, and the three most common regimens with proteasome inhibitors were bortezomib-cyclophosphamide-dexamethasone, bortezomib-lenalidomide-dexamethasone, and bortezomib-dexamethasone.
Dr. Binder reported receiving research funding from ASH. Coauthors reported receiving research funding from Celgene, Janssen, Millennium, and Pfizer, and/or serving in an advisory role for Pfizer.
CHICAGO – Age and the results of select baseline lab measures reflected early response to initial therapy in a review of 1,304 newly diagnosed patients with symptomatic multiple myeloma.
Characteristics associated with increased odds of achieving very good partial response or better (VGPR+) within four cycles of treatment in the study participants, who were seen between 2001 and 2013, were absolute free light chain difference of more than 175 mg/dL (odds ratio, 2.38), age less than 75 years at diagnosis (OR, 2.18), hemoglobin concentration less than 10/12.5 (OR, 1.68), and IgA vs. IgG serum heavy chain secretion (OR, 1.66), Dr. Moritz Binder of the Mayo Clinic, Rochester, Minn., reported in a poster at the American Society of Hematology Meeting on Hematologic Malignancies.
In patients receiving proteasome inhibitors, better response was associated with creatinine concentrations greater than 1.5 mg/dL (OR, 3.83), calcium concentration less than 9.0 mg/dL (OR, 3.37), and greater absolute free light chain difference greater than 175 mg/dL (OR, 2.50), Dr. Binder noted.
High-risk cytogenetic features, conversely, were not associated with treatment response, he said.
The findings are important because the initial response to therapy in newly diagnosed disease can have an effect on long-term outcomes; achieving at least a VGPR to initial treatment can improve progression-free and overall survival. Further, novel agents and risk-adapted treatment strategies now in use have improved response rates and overall survival, Dr. Binder said, adding that it has been unclear, however, whether baseline laboratory parameters could predict the likelihood of early, deep response to initial therapy.
“The ability to predict the likelihood of response to the current therapies can have implications for the treatment approaches in NDMM,” he wrote.
In this study, 288 patients achieved VGPR+ after 4 months, and those patients had a decreased risk of subsequent mortality (hazard ratio, 0.69). This remained true after adjusting for sex, age, International Staging System stage, bone marrow plasma cell involvement, lactate dehydrogenase concentration, initial treatment regimen group, and hematopoietic stem cell transplantation during the disease course (HR, 0.68), he said.
The three most common regimens with immunomodulators that were used in the cohort were lenalidomide-dexamethasone, thalidomide-dexamethasone, and cyclophosphamide-lenalidomide-dexamethasone, and the three most common regimens with proteasome inhibitors were bortezomib-cyclophosphamide-dexamethasone, bortezomib-lenalidomide-dexamethasone, and bortezomib-dexamethasone.
Dr. Binder reported receiving research funding from ASH. Coauthors reported receiving research funding from Celgene, Janssen, Millennium, and Pfizer, and/or serving in an advisory role for Pfizer.
AT MHM 2015
Key clinical point: A number of factors predict early response to initial therapy in patients with symptomatic multiple myeloma, according to findings from a review of 1,304 cases.
Major finding: Predictors of early response were greater absolute free light chain difference (OR, 2.38), younger age (OR, 2.18), hemoglobin concentration (OR, 1.68), and IgA vs. IgG heavy chain secretion (OR, 1.66).
Data source: A retrospective cohort study involving 1,304 patients.
Disclosures: Dr. Binder reported receiving research funding from ASH. Coauthors reported receiving research funding from Celgene, Janssen, Millennium, and Pfizer, and/or serving in an advisory role for Pfizer.
Haplo-HSCT appears comparable to fully matched HSCT

Photo by Chad McNeeley
A retrospective study suggests that, for patients with hematologic disorders, a haploidentical hematopoietic stem cell transplant (HSCT)
can be roughly as safe and effective as a fully matched HSCT.
The study showed that, when patients received an identical conditioning regimen, graft T-cell dose, and graft-vs-host disease (GVHD) prophylaxis, haploidentical and fully matched HSCTs produced comparable results.
Patients had similar rates of overall and progression-free survival, relapse, non-relapse mortality, and chronic GVHD.
However, patients who received haploidentical transplants had higher rates of grade 2-4 acute GVHD and cytomegalovirus reactivation.
Researchers reported these results in Biology of Blood and Marrow Transplantation.
“This is the first study to compare the gold standard to a half-match using an identical protocol,” said Neal Flomenberg, MD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“The field has debated whether the differences in outcomes between full and partial matches were caused by the quality of the match or by all the procedures the patient goes through before and after the donor cells are administered. We haven’t had a clear answer.”
With that in mind, Dr Flomenberg and his colleagues compared 3-year outcome data from patients who received haploidentical HSCTs (n=50) or fully matched HSCTs (n=27), when both groups of patients were treated with a 2-step protocol.
The patients had acute myeloid leukemia (n=38), acute lymphoblastic leukemia (n=20), myelodysplastic syndromes/myeloproliferative neoplasms (n=7), non-Hodgkin lymphoma (n=11), and aplastic anemia (n=1).
The 2-step protocol
All patients received a myeloablative conditioning regimen consisting of 12 Gy of total body irradiation administered in 8 fractions over 4 days. After the last fraction, they received a fixed T-cell dose (2 x 108 cells/kg), which was followed, 2 days later, by cyclophosphamide at 60 mg/kg/day for 2 days.
Twenty-four hours after they completed cyclophosphamide, patients received CD34-selected peripheral blood stem cells from a half-matched or fully matched donor.
On day -1, patients began taking tacrolimus and mycophenolate mofetil as GVHD prophylaxis. They also received growth factor support (granulocyte-macrophage colony-stimulating factor at 250 μg/m2) starting on day +1.
In the absence of GVHD, mycophenolate mofetil was discontinued on day 28 and tacrolimus was tapered, starting on day +60 after HSCT.
Results
The researchers said that early immune recovery was comparable between the patient groups in nearly all assessed T-cell subsets. The exception was the median CD3/CD8 cell count, which was significantly higher at day 28 in the fully matched group than the haploidentical group (P=0.029).
Survival rates were comparable between the groups. The estimated 3-year overall survival was 70% in the haploidentical group and 71% in the fully matched group (P=0.81). The 3-year progression-free survival was 68% and 70%, respectively (P=0.97).
The 3-year cumulative incidence of non-relapse mortality was 10% in the haploidentical group and 4% in the fully matched group (P=0.34). The 3-year cumulative incidence of relapse was 21% and 27%, respectively (P=0.93).
The 100-day cumulative incidence of grade 2-4 acute GVHD was significantly higher in the haploidentical group than the fully matched group—40% and 8%, respectively (P<0.001). But there was no significant difference in the incidence of grade 3-4 acute GVHD—6% and 4%, respectively (P=0.49).
The cumulative incidence of chronic GVHD at 2 years was not significantly different between the haploidentical and fully matched groups—19% and 12%, respectively (P=0.47). The same was true for severe chronic GVHD—4% and 8%, respectively (P=0.49).
The cumulative incidence of cytomegalovirus reactivation was significantly higher in the haploidentical group than the fully matched group—68% and 19%, respectively (P<0.001).
There were no deaths from infections or GVHD in either group.
“The results of the current study are certainly encouraging and suggest that outcomes from a half-matched, related donor are similar to fully matched donors,” said study author Sameh Gaballa, MD, also of Thomas Jefferson University.
“It might be time to reassess whether half-matched, related transplants can be considered the best alternative donor source for patients lacking a fully matched family member donor. For that, we’ll need more evidence from a randomly controlled, prospective trial, rather than studies that look at patient data retrospectively, to help solidify our findings here.” ![]()

Photo by Chad McNeeley
A retrospective study suggests that, for patients with hematologic disorders, a haploidentical hematopoietic stem cell transplant (HSCT)
can be roughly as safe and effective as a fully matched HSCT.
The study showed that, when patients received an identical conditioning regimen, graft T-cell dose, and graft-vs-host disease (GVHD) prophylaxis, haploidentical and fully matched HSCTs produced comparable results.
Patients had similar rates of overall and progression-free survival, relapse, non-relapse mortality, and chronic GVHD.
However, patients who received haploidentical transplants had higher rates of grade 2-4 acute GVHD and cytomegalovirus reactivation.
Researchers reported these results in Biology of Blood and Marrow Transplantation.
“This is the first study to compare the gold standard to a half-match using an identical protocol,” said Neal Flomenberg, MD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“The field has debated whether the differences in outcomes between full and partial matches were caused by the quality of the match or by all the procedures the patient goes through before and after the donor cells are administered. We haven’t had a clear answer.”
With that in mind, Dr Flomenberg and his colleagues compared 3-year outcome data from patients who received haploidentical HSCTs (n=50) or fully matched HSCTs (n=27), when both groups of patients were treated with a 2-step protocol.
The patients had acute myeloid leukemia (n=38), acute lymphoblastic leukemia (n=20), myelodysplastic syndromes/myeloproliferative neoplasms (n=7), non-Hodgkin lymphoma (n=11), and aplastic anemia (n=1).
The 2-step protocol
All patients received a myeloablative conditioning regimen consisting of 12 Gy of total body irradiation administered in 8 fractions over 4 days. After the last fraction, they received a fixed T-cell dose (2 x 108 cells/kg), which was followed, 2 days later, by cyclophosphamide at 60 mg/kg/day for 2 days.
Twenty-four hours after they completed cyclophosphamide, patients received CD34-selected peripheral blood stem cells from a half-matched or fully matched donor.
On day -1, patients began taking tacrolimus and mycophenolate mofetil as GVHD prophylaxis. They also received growth factor support (granulocyte-macrophage colony-stimulating factor at 250 μg/m2) starting on day +1.
In the absence of GVHD, mycophenolate mofetil was discontinued on day 28 and tacrolimus was tapered, starting on day +60 after HSCT.
Results
The researchers said that early immune recovery was comparable between the patient groups in nearly all assessed T-cell subsets. The exception was the median CD3/CD8 cell count, which was significantly higher at day 28 in the fully matched group than the haploidentical group (P=0.029).
Survival rates were comparable between the groups. The estimated 3-year overall survival was 70% in the haploidentical group and 71% in the fully matched group (P=0.81). The 3-year progression-free survival was 68% and 70%, respectively (P=0.97).
The 3-year cumulative incidence of non-relapse mortality was 10% in the haploidentical group and 4% in the fully matched group (P=0.34). The 3-year cumulative incidence of relapse was 21% and 27%, respectively (P=0.93).
The 100-day cumulative incidence of grade 2-4 acute GVHD was significantly higher in the haploidentical group than the fully matched group—40% and 8%, respectively (P<0.001). But there was no significant difference in the incidence of grade 3-4 acute GVHD—6% and 4%, respectively (P=0.49).
The cumulative incidence of chronic GVHD at 2 years was not significantly different between the haploidentical and fully matched groups—19% and 12%, respectively (P=0.47). The same was true for severe chronic GVHD—4% and 8%, respectively (P=0.49).
The cumulative incidence of cytomegalovirus reactivation was significantly higher in the haploidentical group than the fully matched group—68% and 19%, respectively (P<0.001).
There were no deaths from infections or GVHD in either group.
“The results of the current study are certainly encouraging and suggest that outcomes from a half-matched, related donor are similar to fully matched donors,” said study author Sameh Gaballa, MD, also of Thomas Jefferson University.
“It might be time to reassess whether half-matched, related transplants can be considered the best alternative donor source for patients lacking a fully matched family member donor. For that, we’ll need more evidence from a randomly controlled, prospective trial, rather than studies that look at patient data retrospectively, to help solidify our findings here.” ![]()

Photo by Chad McNeeley
A retrospective study suggests that, for patients with hematologic disorders, a haploidentical hematopoietic stem cell transplant (HSCT)
can be roughly as safe and effective as a fully matched HSCT.
The study showed that, when patients received an identical conditioning regimen, graft T-cell dose, and graft-vs-host disease (GVHD) prophylaxis, haploidentical and fully matched HSCTs produced comparable results.
Patients had similar rates of overall and progression-free survival, relapse, non-relapse mortality, and chronic GVHD.
However, patients who received haploidentical transplants had higher rates of grade 2-4 acute GVHD and cytomegalovirus reactivation.
Researchers reported these results in Biology of Blood and Marrow Transplantation.
“This is the first study to compare the gold standard to a half-match using an identical protocol,” said Neal Flomenberg, MD, of Thomas Jefferson University in Philadelphia, Pennsylvania.
“The field has debated whether the differences in outcomes between full and partial matches were caused by the quality of the match or by all the procedures the patient goes through before and after the donor cells are administered. We haven’t had a clear answer.”
With that in mind, Dr Flomenberg and his colleagues compared 3-year outcome data from patients who received haploidentical HSCTs (n=50) or fully matched HSCTs (n=27), when both groups of patients were treated with a 2-step protocol.
The patients had acute myeloid leukemia (n=38), acute lymphoblastic leukemia (n=20), myelodysplastic syndromes/myeloproliferative neoplasms (n=7), non-Hodgkin lymphoma (n=11), and aplastic anemia (n=1).
The 2-step protocol
All patients received a myeloablative conditioning regimen consisting of 12 Gy of total body irradiation administered in 8 fractions over 4 days. After the last fraction, they received a fixed T-cell dose (2 x 108 cells/kg), which was followed, 2 days later, by cyclophosphamide at 60 mg/kg/day for 2 days.
Twenty-four hours after they completed cyclophosphamide, patients received CD34-selected peripheral blood stem cells from a half-matched or fully matched donor.
On day -1, patients began taking tacrolimus and mycophenolate mofetil as GVHD prophylaxis. They also received growth factor support (granulocyte-macrophage colony-stimulating factor at 250 μg/m2) starting on day +1.
In the absence of GVHD, mycophenolate mofetil was discontinued on day 28 and tacrolimus was tapered, starting on day +60 after HSCT.
Results
The researchers said that early immune recovery was comparable between the patient groups in nearly all assessed T-cell subsets. The exception was the median CD3/CD8 cell count, which was significantly higher at day 28 in the fully matched group than the haploidentical group (P=0.029).
Survival rates were comparable between the groups. The estimated 3-year overall survival was 70% in the haploidentical group and 71% in the fully matched group (P=0.81). The 3-year progression-free survival was 68% and 70%, respectively (P=0.97).
The 3-year cumulative incidence of non-relapse mortality was 10% in the haploidentical group and 4% in the fully matched group (P=0.34). The 3-year cumulative incidence of relapse was 21% and 27%, respectively (P=0.93).
The 100-day cumulative incidence of grade 2-4 acute GVHD was significantly higher in the haploidentical group than the fully matched group—40% and 8%, respectively (P<0.001). But there was no significant difference in the incidence of grade 3-4 acute GVHD—6% and 4%, respectively (P=0.49).
The cumulative incidence of chronic GVHD at 2 years was not significantly different between the haploidentical and fully matched groups—19% and 12%, respectively (P=0.47). The same was true for severe chronic GVHD—4% and 8%, respectively (P=0.49).
The cumulative incidence of cytomegalovirus reactivation was significantly higher in the haploidentical group than the fully matched group—68% and 19%, respectively (P<0.001).
There were no deaths from infections or GVHD in either group.
“The results of the current study are certainly encouraging and suggest that outcomes from a half-matched, related donor are similar to fully matched donors,” said study author Sameh Gaballa, MD, also of Thomas Jefferson University.
“It might be time to reassess whether half-matched, related transplants can be considered the best alternative donor source for patients lacking a fully matched family member donor. For that, we’ll need more evidence from a randomly controlled, prospective trial, rather than studies that look at patient data retrospectively, to help solidify our findings here.”
Method can identify CTCL quickly, study suggests
High-throughput sequencing (HTS) may enable earlier diagnosis of cutaneous T-cell lymphoma (CTCL), according to a study published in Science Translational Medicine.
Researchers said HTS, which provided new insights into the biology of CTCL, accurately diagnosed 100% of patients with the disease, including those with early stage and recurrent CTCL.
HTS exhibited greater sensitivity and specificity than polymerase chain reaction.
The researchers noted that, in CTCL, cancerous T cells accumulate in the skin, causing skin tumors but also rashes that are often mistaken for benign inflammatory skin conditions. And this delays diagnosis.
“Using current methods, it takes, on average, 6 years for patients to receive a definitive CTCL diagnosis,” said Rachael Clark, MD, PhD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“As a result, appropriate therapy is delayed, often until the disease worsens to the point where diagnosis is clear, but effective treatment is more difficult.”
In search of a new diagnostic approach, Dr Clark and her colleagues used HTS to analyze T-cell receptor genes from blood and skin tissue samples. The team compared samples from 46 CTCL patients to samples from healthy individuals and patients with psoriasis, eczema, and other non-cancerous, inflammatory skin diseases.
By identifying and quantifying malignant T cells, HTS accurately distinguished CTCL from other skin diseases in all 46 patients. In contrast, polymerase chain reaction correctly identified 70% of the CTCL samples, often missing earlier-stage tumors.
For a subset of patients, the researchers used HTS to track malignant T cells over time, allowing them to detect early recurrence and monitor patient response to treatment.
HTS also revealed new insights into the cell of origin for CTCL, which has remained unresolved. The researchers found that CTCL arises from mature T cells, a discovery that may aid the development of more effective therapy.
The team said that, altogether, their findings suggest HTS is a promising tool for guiding earlier diagnosis and, thus, treatment of CTCL patients.
HTS was performed using ImmunoSEQ, an assay developed by Adaptive Biotechnologies. The company did not sponsor the study, but company employees were involved in the research.

High-throughput sequencing (HTS) may enable earlier diagnosis of cutaneous T-cell lymphoma (CTCL), according to a study published in Science Translational Medicine.
Researchers said HTS, which provided new insights into the biology of CTCL, accurately diagnosed 100% of patients with the disease, including those with early stage and recurrent CTCL.
HTS exhibited greater sensitivity and specificity than polymerase chain reaction.
The researchers noted that, in CTCL, cancerous T cells accumulate in the skin, causing skin tumors but also rashes that are often mistaken for benign inflammatory skin conditions. And this delays diagnosis.
“Using current methods, it takes, on average, 6 years for patients to receive a definitive CTCL diagnosis,” said Rachael Clark, MD, PhD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“As a result, appropriate therapy is delayed, often until the disease worsens to the point where diagnosis is clear, but effective treatment is more difficult.”
In search of a new diagnostic approach, Dr Clark and her colleagues used HTS to analyze T-cell receptor genes from blood and skin tissue samples. The team compared samples from 46 CTCL patients to samples from healthy individuals and patients with psoriasis, eczema, and other non-cancerous, inflammatory skin diseases.
By identifying and quantifying malignant T cells, HTS accurately distinguished CTCL from other skin diseases in all 46 patients. In contrast, polymerase chain reaction correctly identified 70% of the CTCL samples, often missing earlier-stage tumors.
For a subset of patients, the researchers used HTS to track malignant T cells over time, allowing them to detect early recurrence and monitor patient response to treatment.
HTS also revealed new insights into the cell of origin for CTCL, which has remained unresolved. The researchers found that CTCL arises from mature T cells, a discovery that may aid the development of more effective therapy.
The team said that, altogether, their findings suggest HTS is a promising tool for guiding earlier diagnosis and, thus, treatment of CTCL patients.
HTS was performed using ImmunoSEQ, an assay developed by Adaptive Biotechnologies. The company did not sponsor the study, but company employees were involved in the research.

High-throughput sequencing (HTS) may enable earlier diagnosis of cutaneous T-cell lymphoma (CTCL), according to a study published in Science Translational Medicine.
Researchers said HTS, which provided new insights into the biology of CTCL, accurately diagnosed 100% of patients with the disease, including those with early stage and recurrent CTCL.
HTS exhibited greater sensitivity and specificity than polymerase chain reaction.
The researchers noted that, in CTCL, cancerous T cells accumulate in the skin, causing skin tumors but also rashes that are often mistaken for benign inflammatory skin conditions. And this delays diagnosis.
“Using current methods, it takes, on average, 6 years for patients to receive a definitive CTCL diagnosis,” said Rachael Clark, MD, PhD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“As a result, appropriate therapy is delayed, often until the disease worsens to the point where diagnosis is clear, but effective treatment is more difficult.”
In search of a new diagnostic approach, Dr Clark and her colleagues used HTS to analyze T-cell receptor genes from blood and skin tissue samples. The team compared samples from 46 CTCL patients to samples from healthy individuals and patients with psoriasis, eczema, and other non-cancerous, inflammatory skin diseases.
By identifying and quantifying malignant T cells, HTS accurately distinguished CTCL from other skin diseases in all 46 patients. In contrast, polymerase chain reaction correctly identified 70% of the CTCL samples, often missing earlier-stage tumors.
For a subset of patients, the researchers used HTS to track malignant T cells over time, allowing them to detect early recurrence and monitor patient response to treatment.
HTS also revealed new insights into the cell of origin for CTCL, which has remained unresolved. The researchers found that CTCL arises from mature T cells, a discovery that may aid the development of more effective therapy.
The team said that, altogether, their findings suggest HTS is a promising tool for guiding earlier diagnosis and, thus, treatment of CTCL patients.
HTS was performed using ImmunoSEQ, an assay developed by Adaptive Biotechnologies. The company did not sponsor the study, but company employees were involved in the research.
New guidelines address multiple myeloma–related complications
New guidelines on managing the complications of multiple myeloma and its treatment recommend whole-body, low-dose computed tomography over conventional radiography because of its superior sensitivity for detecting osteolytic lesions. The guidelines, drafted by the European Myeloma Network, also provide an imaging algorithm to address various clinical scenarios.
This is just one of the recommendations from the interdisciplinary panel that reviewed published randomized trials, guidelines, meta-analyses, systematic reviews, observational studies, and case reports on the topic. They graded their recommendations according to the strength of the evidence and, when evidence was insufficient, expert consensus.
“Multiple myeloma … is characterized by bone destruction, anemia, renal and immunological impairment. These complications may lead to severe impairment of the quality of life of myeloma patients and may deteriorate their life expectancy,” note the authors, who were led by Dr. Evangelos Terpos of the department of clinical therapeutics, National and Kapodistrian University of Athens (Haematologica. 2015;100:1254-66).
The panel endorsed addition of zoledronic acid (Zometa) or pamidronate (Aredia) to specific antimyeloma therapy for patients with adequate renal function who have bone disease at diagnosis. Although evidence is weaker, they note that symptomatic patients who do not have lytic lesions on conventional radiography can be treated with zoledronic acid; however, its use in patients with no bone involvement on computed tomography or magnetic resonance imaging has uncertain benefit. Additionally, they do not recommend use of bisphosphonates in asymptomatic patients.
The panel recommends that zoledronic acid be given continuously, although they add that benefit of continuous use is not clear in patients who achieve at least a very good partial response.
Treatment can be initiated with erythropoiesis-stimulating agents such as epoetin alfa (various brand names) and darbepoetin (Aranesp) in patients who have persistent symptomatic anemia (defined as a hemoglobin level of less than 10 g/dL) without any other apparent cause. However, these agents should be stopped after 6 to 8 weeks if hemoglobin response has not been adequate.
The panel notes that bortezomib(Velcade)-based regimens are the standard of care for patients with multiple myeloma who have renal impairment. Lenalidomide is an option, albeit with weaker evidence, in cases of mild to moderate renal impairment.
In patients who experience treatment-induced peripheral neuropathy, therapy should be modified by either altering the schedule or route of administration (as appropriate) or reducing the dose, according to the guidelines.
Patients with multiple myeloma (and their contacts) should be vaccinated against influenza, the panel recommends; vaccination against Streptococcus pneumonia and Haemophilus influenzae is “appropriate,” although efficacy is not guaranteed as patients are immunologically compromised.
The panel endorsed acyclovir (Zovirax) or valacyclovir (Valtrex) for herpes zoster virus prophylaxis in patients receiving proteasome inhibitors or undergoing autologous or allogeneic transplantation, mainly directed to patients who are seropositive.
Finally, clinicians should assess risk of venous thromboembolism in patients who are due to start immunomodulatory drug therapy and should use appropriate risk-based antiplatelet or anticoagulation therapy throughout treatment, according to the guidelines.
Dr. Terpos reported having no relevant disclosures.
New guidelines on managing the complications of multiple myeloma and its treatment recommend whole-body, low-dose computed tomography over conventional radiography because of its superior sensitivity for detecting osteolytic lesions. The guidelines, drafted by the European Myeloma Network, also provide an imaging algorithm to address various clinical scenarios.
This is just one of the recommendations from the interdisciplinary panel that reviewed published randomized trials, guidelines, meta-analyses, systematic reviews, observational studies, and case reports on the topic. They graded their recommendations according to the strength of the evidence and, when evidence was insufficient, expert consensus.
“Multiple myeloma … is characterized by bone destruction, anemia, renal and immunological impairment. These complications may lead to severe impairment of the quality of life of myeloma patients and may deteriorate their life expectancy,” note the authors, who were led by Dr. Evangelos Terpos of the department of clinical therapeutics, National and Kapodistrian University of Athens (Haematologica. 2015;100:1254-66).
The panel endorsed addition of zoledronic acid (Zometa) or pamidronate (Aredia) to specific antimyeloma therapy for patients with adequate renal function who have bone disease at diagnosis. Although evidence is weaker, they note that symptomatic patients who do not have lytic lesions on conventional radiography can be treated with zoledronic acid; however, its use in patients with no bone involvement on computed tomography or magnetic resonance imaging has uncertain benefit. Additionally, they do not recommend use of bisphosphonates in asymptomatic patients.
The panel recommends that zoledronic acid be given continuously, although they add that benefit of continuous use is not clear in patients who achieve at least a very good partial response.
Treatment can be initiated with erythropoiesis-stimulating agents such as epoetin alfa (various brand names) and darbepoetin (Aranesp) in patients who have persistent symptomatic anemia (defined as a hemoglobin level of less than 10 g/dL) without any other apparent cause. However, these agents should be stopped after 6 to 8 weeks if hemoglobin response has not been adequate.
The panel notes that bortezomib(Velcade)-based regimens are the standard of care for patients with multiple myeloma who have renal impairment. Lenalidomide is an option, albeit with weaker evidence, in cases of mild to moderate renal impairment.
In patients who experience treatment-induced peripheral neuropathy, therapy should be modified by either altering the schedule or route of administration (as appropriate) or reducing the dose, according to the guidelines.
Patients with multiple myeloma (and their contacts) should be vaccinated against influenza, the panel recommends; vaccination against Streptococcus pneumonia and Haemophilus influenzae is “appropriate,” although efficacy is not guaranteed as patients are immunologically compromised.
The panel endorsed acyclovir (Zovirax) or valacyclovir (Valtrex) for herpes zoster virus prophylaxis in patients receiving proteasome inhibitors or undergoing autologous or allogeneic transplantation, mainly directed to patients who are seropositive.
Finally, clinicians should assess risk of venous thromboembolism in patients who are due to start immunomodulatory drug therapy and should use appropriate risk-based antiplatelet or anticoagulation therapy throughout treatment, according to the guidelines.
Dr. Terpos reported having no relevant disclosures.
New guidelines on managing the complications of multiple myeloma and its treatment recommend whole-body, low-dose computed tomography over conventional radiography because of its superior sensitivity for detecting osteolytic lesions. The guidelines, drafted by the European Myeloma Network, also provide an imaging algorithm to address various clinical scenarios.
This is just one of the recommendations from the interdisciplinary panel that reviewed published randomized trials, guidelines, meta-analyses, systematic reviews, observational studies, and case reports on the topic. They graded their recommendations according to the strength of the evidence and, when evidence was insufficient, expert consensus.
“Multiple myeloma … is characterized by bone destruction, anemia, renal and immunological impairment. These complications may lead to severe impairment of the quality of life of myeloma patients and may deteriorate their life expectancy,” note the authors, who were led by Dr. Evangelos Terpos of the department of clinical therapeutics, National and Kapodistrian University of Athens (Haematologica. 2015;100:1254-66).
The panel endorsed addition of zoledronic acid (Zometa) or pamidronate (Aredia) to specific antimyeloma therapy for patients with adequate renal function who have bone disease at diagnosis. Although evidence is weaker, they note that symptomatic patients who do not have lytic lesions on conventional radiography can be treated with zoledronic acid; however, its use in patients with no bone involvement on computed tomography or magnetic resonance imaging has uncertain benefit. Additionally, they do not recommend use of bisphosphonates in asymptomatic patients.
The panel recommends that zoledronic acid be given continuously, although they add that benefit of continuous use is not clear in patients who achieve at least a very good partial response.
Treatment can be initiated with erythropoiesis-stimulating agents such as epoetin alfa (various brand names) and darbepoetin (Aranesp) in patients who have persistent symptomatic anemia (defined as a hemoglobin level of less than 10 g/dL) without any other apparent cause. However, these agents should be stopped after 6 to 8 weeks if hemoglobin response has not been adequate.
The panel notes that bortezomib(Velcade)-based regimens are the standard of care for patients with multiple myeloma who have renal impairment. Lenalidomide is an option, albeit with weaker evidence, in cases of mild to moderate renal impairment.
In patients who experience treatment-induced peripheral neuropathy, therapy should be modified by either altering the schedule or route of administration (as appropriate) or reducing the dose, according to the guidelines.
Patients with multiple myeloma (and their contacts) should be vaccinated against influenza, the panel recommends; vaccination against Streptococcus pneumonia and Haemophilus influenzae is “appropriate,” although efficacy is not guaranteed as patients are immunologically compromised.
The panel endorsed acyclovir (Zovirax) or valacyclovir (Valtrex) for herpes zoster virus prophylaxis in patients receiving proteasome inhibitors or undergoing autologous or allogeneic transplantation, mainly directed to patients who are seropositive.
Finally, clinicians should assess risk of venous thromboembolism in patients who are due to start immunomodulatory drug therapy and should use appropriate risk-based antiplatelet or anticoagulation therapy throughout treatment, according to the guidelines.
Dr. Terpos reported having no relevant disclosures.
FROM HAEMATOLOGICA

