User login
Reprogramming the immune system

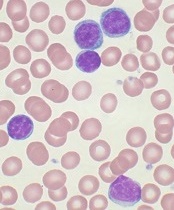
showing Hodgkin lymphoma
NEW YORK—Using a 3-pronged approach to reprogram the immune system—inhibition of critical pathways, activation of others, and depletion of malignant cells—may be the best strategy to optimize immune function in B-cell lymphomas, according to Stephen M. Ansell, MD, PhD, of the Mayo Clinic in Rochester, Minnesota.
“[A]ll told, there are multiple immunological barriers to an effective immune response,” Dr Ansell said at Lymphoma & Myeloma 2015.
“So the questions are how can you use an immune checkpoint approach to try and modulate this and improve the outcome.”
Dr Ansell discussed checkpoint inhibitors, immune signal activators, and the potential of combining the approaches in Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL) in a way that enhances rather than antagonizes their effects.
Blocking CTLA-4
Cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) functions as an immune checkpoint that downregulates the immune system. A receptor found on the surface of inhibitor T cells, it acts as an off switch when it binds to CD80 or CD86 on the surface of antigen-presenting cells.
Ipilimumab, an antibody that targets CTLA-4, has been approved by the US Food and Drug Administration for the treatment of melanoma and is in clinical trials for lung, bladder, and prostate cancer.
Investigators wanted to see whether it also works in lymphoma, so they conducted a phase 1 study in relapsed/refractory B-cell NHL.
Eighteen patients received 3 mg/kg of ipilimumab. Two patients responded, 1 with a complete response (CR) that lasted more than 31 months, and 1 with a partial response (PR) that lasted 19 months. In 5 of 16 patients (31%), T-cell proliferation to recall antigens increased more than 2-fold.
As Dr Ansell explained, “Immune response doesn’t always correlate directly with the clinical responses. So I think we really have a lot to learn about what is really a biomarker of efficacy.”
Ipilimumab was also evaluated to treat relapse after allogeneic hematopoietic stem cell transplantation (HSCT) in 29 patients with relapsed hematologic disease. Two patients with HL achieved a CR and 1 patient with mantle cell lymphoma achieved a PR.
The investigators observed that ipilimumab did not induce or exacerbate clinical graft-versus-host disease.
Blocking PD-1
Programmed cell death protein 1 (PD-1) is a surface receptor expressed on T cells and pro-B cells. PD-1 binds 2 ligands, PD-L1 and PD-L2.
PD-1 ligands are overexpressed in inflammatory environments and attenuate the immune response through PD-1 on immune effector cells. In addition, PD-L1 expressed on malignant cells or in the tumor microenvironment suppresses tumor-infiltrating lymphocyte activity.
Pidilizumab, a humanized monoclonal antibody that binds to PD-1, weakens the apoptotic processes in lymphocytes and augments the antitumor activities of NK cells.
Investigators conducted a phase 2 trial of pidilizumab in patients with diffuse large B-cell lymphoma (DLBCL) after autologous HSCT to modulate the immune system after a transplant.
The team treated 66 patients with the antibody. At 16 months, progression-free survival (PFS) was 72%. For the 24 high-risk patients who were PET-positive after salvage chemotherapy, the 16-month PFS was 70%.
“And I think that what was most interesting,” Dr Ansell said, when focusing on the 35 patients with measurable disease after transplant, pidilizumab produced a 51% response rate “even in patients that actually had active disease.”
When pidilizumab was combined with rituximab in another trial in patients with relapsed follicular lymphoma (FL), 19 of 29 evaluable patients (66%) achieved an objective response: 15 (52%) CRs and 4 (14%) PRs.
“You might say, ‘Who cares? That’s not that great,’” Dr Ansell said. “But I think what was pretty impressive is that 52% CR rate. And most of you who treat patients with rituximab would know that that’s quite surprising, suggesting that there may be additional benefit for the use of PD-1 blockade in this subset of patients.”
Nivolumab, another monoclonal antibody that blocks the PD-1 pathway, is being investigated in a number of lymphoid malignancies, including HL, DLBCL, and T-cell lymphomas.
In a phase 1 study of nivolumab in 81 patients with relapsed or refractory lymphoid malignancies, the best preliminary overall response has been in FL and DLBCL patients, with an objective response rate of 40% and 36%, respectively, including 1 PR and 3 PRs in each subtype.
“I think what is important,” Dr Ansell said, “is that the side effects, as expected, were mainly immune-mediated, not as dramatic as have been seen with other agents, and very similar to what has been seen in solid tumor studies.”
Dr Ansell pointed out that the response rates with nivolumab varied widely by histology, suggesting that “we have a lot to learn about why patients benefit and who exactly benefits.”
There were no responses in patients with multiple myeloma or primary mediastinal B-cell lymphoma, although many patients achieved stable disease.
“Hodgkin lymphoma was completely different,” Dr Ansell said, “and there were responses in virtually every patient.”
Of 23 patients treated with nivolumab, 20 responded—4 achieved a CR and 16 a PR—including patients who had failed autologous HSCT and brentuximab vedotin treatment. Eleven patients, including 2 with CRs, have an ongoing response, some approaching 2 years. So the responses have been durable, Dr Ansell noted.
Yet another PD-1 antibody, pembrolizumab, has prompted reduction in tumor burden in HL in all but 2 of 29 evaluable patients, including 6 CRs and 13 PRs. The median duration of response has not yet been reached, and the side effect profile was similar to what has been seen with nivolumab and in solid tumors.
Activating immune stimulatory signals
Another approach to boosting the immune system is to activate immune stimulatory signals, eg, CD27 and CD40, and get a benefit that way. Varlilumab (CDX-1127) is an unconjugated monoclonal antibody that binds CD27 and activates CD27-expressing T cells.
In a phase 1 trial of varlilumab in 24 lymphoma patients, investigators found no significant depletion in absolute lymphocyte counts, T cells, or B cells. “Not quite the same success story,” Dr Ansell said, with a response—a CR—in only 1 patient.
Investigators did observe, however, evidence of increased soluble CD27, a reduction of circulating Tregs, and the induction of pro-inflammatory cytokines.
And in a phase 1 study of the anti-CD40 monoclonal antibody dacetuzumab in recurrent NHL, “the response rate was disappointingly low,” Dr Ansell said.
Investigators observed 6 objective responses, including 1 CR and 5 PRs, and a decrease in tumor size in approximately one-third of the 50 patients treated.
The investigators of the subsequent phase 2 trial did not want to take the agent forward for further study, Dr Ansell noted, “but there are antibodies now being developed in this space that will hopefully be more effective and create a greater benefit.”
Optimizing immune function
Dr Ansell suggested there are 3 main approaches to treating patients. One is going directly after the malignant cells and depleting them. A second is to inhibit critical pathways that the malignant cell is dependent upon, “starving them, if you like.” The third way is to activate the immune system and thereby create a greater benefit for patients.
“[P]robably our best strategy is to use all 3 in a reprogram approach,” he said. “Because unless you target each one of these areas, the likelihood is that the other sides of the 3-legged stool will take over.”
“This is an encouraging and exciting time for immune checkpoints therapy and an encouraging and exciting time for immune therapies in general. I think this is really the new frontier in lymphomas.” ![]()

showing Hodgkin lymphoma
NEW YORK—Using a 3-pronged approach to reprogram the immune system—inhibition of critical pathways, activation of others, and depletion of malignant cells—may be the best strategy to optimize immune function in B-cell lymphomas, according to Stephen M. Ansell, MD, PhD, of the Mayo Clinic in Rochester, Minnesota.
“[A]ll told, there are multiple immunological barriers to an effective immune response,” Dr Ansell said at Lymphoma & Myeloma 2015.
“So the questions are how can you use an immune checkpoint approach to try and modulate this and improve the outcome.”
Dr Ansell discussed checkpoint inhibitors, immune signal activators, and the potential of combining the approaches in Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL) in a way that enhances rather than antagonizes their effects.
Blocking CTLA-4
Cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) functions as an immune checkpoint that downregulates the immune system. A receptor found on the surface of inhibitor T cells, it acts as an off switch when it binds to CD80 or CD86 on the surface of antigen-presenting cells.
Ipilimumab, an antibody that targets CTLA-4, has been approved by the US Food and Drug Administration for the treatment of melanoma and is in clinical trials for lung, bladder, and prostate cancer.
Investigators wanted to see whether it also works in lymphoma, so they conducted a phase 1 study in relapsed/refractory B-cell NHL.
Eighteen patients received 3 mg/kg of ipilimumab. Two patients responded, 1 with a complete response (CR) that lasted more than 31 months, and 1 with a partial response (PR) that lasted 19 months. In 5 of 16 patients (31%), T-cell proliferation to recall antigens increased more than 2-fold.
As Dr Ansell explained, “Immune response doesn’t always correlate directly with the clinical responses. So I think we really have a lot to learn about what is really a biomarker of efficacy.”
Ipilimumab was also evaluated to treat relapse after allogeneic hematopoietic stem cell transplantation (HSCT) in 29 patients with relapsed hematologic disease. Two patients with HL achieved a CR and 1 patient with mantle cell lymphoma achieved a PR.
The investigators observed that ipilimumab did not induce or exacerbate clinical graft-versus-host disease.
Blocking PD-1
Programmed cell death protein 1 (PD-1) is a surface receptor expressed on T cells and pro-B cells. PD-1 binds 2 ligands, PD-L1 and PD-L2.
PD-1 ligands are overexpressed in inflammatory environments and attenuate the immune response through PD-1 on immune effector cells. In addition, PD-L1 expressed on malignant cells or in the tumor microenvironment suppresses tumor-infiltrating lymphocyte activity.
Pidilizumab, a humanized monoclonal antibody that binds to PD-1, weakens the apoptotic processes in lymphocytes and augments the antitumor activities of NK cells.
Investigators conducted a phase 2 trial of pidilizumab in patients with diffuse large B-cell lymphoma (DLBCL) after autologous HSCT to modulate the immune system after a transplant.
The team treated 66 patients with the antibody. At 16 months, progression-free survival (PFS) was 72%. For the 24 high-risk patients who were PET-positive after salvage chemotherapy, the 16-month PFS was 70%.
“And I think that what was most interesting,” Dr Ansell said, when focusing on the 35 patients with measurable disease after transplant, pidilizumab produced a 51% response rate “even in patients that actually had active disease.”
When pidilizumab was combined with rituximab in another trial in patients with relapsed follicular lymphoma (FL), 19 of 29 evaluable patients (66%) achieved an objective response: 15 (52%) CRs and 4 (14%) PRs.
“You might say, ‘Who cares? That’s not that great,’” Dr Ansell said. “But I think what was pretty impressive is that 52% CR rate. And most of you who treat patients with rituximab would know that that’s quite surprising, suggesting that there may be additional benefit for the use of PD-1 blockade in this subset of patients.”
Nivolumab, another monoclonal antibody that blocks the PD-1 pathway, is being investigated in a number of lymphoid malignancies, including HL, DLBCL, and T-cell lymphomas.
In a phase 1 study of nivolumab in 81 patients with relapsed or refractory lymphoid malignancies, the best preliminary overall response has been in FL and DLBCL patients, with an objective response rate of 40% and 36%, respectively, including 1 PR and 3 PRs in each subtype.
“I think what is important,” Dr Ansell said, “is that the side effects, as expected, were mainly immune-mediated, not as dramatic as have been seen with other agents, and very similar to what has been seen in solid tumor studies.”
Dr Ansell pointed out that the response rates with nivolumab varied widely by histology, suggesting that “we have a lot to learn about why patients benefit and who exactly benefits.”
There were no responses in patients with multiple myeloma or primary mediastinal B-cell lymphoma, although many patients achieved stable disease.
“Hodgkin lymphoma was completely different,” Dr Ansell said, “and there were responses in virtually every patient.”
Of 23 patients treated with nivolumab, 20 responded—4 achieved a CR and 16 a PR—including patients who had failed autologous HSCT and brentuximab vedotin treatment. Eleven patients, including 2 with CRs, have an ongoing response, some approaching 2 years. So the responses have been durable, Dr Ansell noted.
Yet another PD-1 antibody, pembrolizumab, has prompted reduction in tumor burden in HL in all but 2 of 29 evaluable patients, including 6 CRs and 13 PRs. The median duration of response has not yet been reached, and the side effect profile was similar to what has been seen with nivolumab and in solid tumors.
Activating immune stimulatory signals
Another approach to boosting the immune system is to activate immune stimulatory signals, eg, CD27 and CD40, and get a benefit that way. Varlilumab (CDX-1127) is an unconjugated monoclonal antibody that binds CD27 and activates CD27-expressing T cells.
In a phase 1 trial of varlilumab in 24 lymphoma patients, investigators found no significant depletion in absolute lymphocyte counts, T cells, or B cells. “Not quite the same success story,” Dr Ansell said, with a response—a CR—in only 1 patient.
Investigators did observe, however, evidence of increased soluble CD27, a reduction of circulating Tregs, and the induction of pro-inflammatory cytokines.
And in a phase 1 study of the anti-CD40 monoclonal antibody dacetuzumab in recurrent NHL, “the response rate was disappointingly low,” Dr Ansell said.
Investigators observed 6 objective responses, including 1 CR and 5 PRs, and a decrease in tumor size in approximately one-third of the 50 patients treated.
The investigators of the subsequent phase 2 trial did not want to take the agent forward for further study, Dr Ansell noted, “but there are antibodies now being developed in this space that will hopefully be more effective and create a greater benefit.”
Optimizing immune function
Dr Ansell suggested there are 3 main approaches to treating patients. One is going directly after the malignant cells and depleting them. A second is to inhibit critical pathways that the malignant cell is dependent upon, “starving them, if you like.” The third way is to activate the immune system and thereby create a greater benefit for patients.
“[P]robably our best strategy is to use all 3 in a reprogram approach,” he said. “Because unless you target each one of these areas, the likelihood is that the other sides of the 3-legged stool will take over.”
“This is an encouraging and exciting time for immune checkpoints therapy and an encouraging and exciting time for immune therapies in general. I think this is really the new frontier in lymphomas.” ![]()

showing Hodgkin lymphoma
NEW YORK—Using a 3-pronged approach to reprogram the immune system—inhibition of critical pathways, activation of others, and depletion of malignant cells—may be the best strategy to optimize immune function in B-cell lymphomas, according to Stephen M. Ansell, MD, PhD, of the Mayo Clinic in Rochester, Minnesota.
“[A]ll told, there are multiple immunological barriers to an effective immune response,” Dr Ansell said at Lymphoma & Myeloma 2015.
“So the questions are how can you use an immune checkpoint approach to try and modulate this and improve the outcome.”
Dr Ansell discussed checkpoint inhibitors, immune signal activators, and the potential of combining the approaches in Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL) in a way that enhances rather than antagonizes their effects.
Blocking CTLA-4
Cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) functions as an immune checkpoint that downregulates the immune system. A receptor found on the surface of inhibitor T cells, it acts as an off switch when it binds to CD80 or CD86 on the surface of antigen-presenting cells.
Ipilimumab, an antibody that targets CTLA-4, has been approved by the US Food and Drug Administration for the treatment of melanoma and is in clinical trials for lung, bladder, and prostate cancer.
Investigators wanted to see whether it also works in lymphoma, so they conducted a phase 1 study in relapsed/refractory B-cell NHL.
Eighteen patients received 3 mg/kg of ipilimumab. Two patients responded, 1 with a complete response (CR) that lasted more than 31 months, and 1 with a partial response (PR) that lasted 19 months. In 5 of 16 patients (31%), T-cell proliferation to recall antigens increased more than 2-fold.
As Dr Ansell explained, “Immune response doesn’t always correlate directly with the clinical responses. So I think we really have a lot to learn about what is really a biomarker of efficacy.”
Ipilimumab was also evaluated to treat relapse after allogeneic hematopoietic stem cell transplantation (HSCT) in 29 patients with relapsed hematologic disease. Two patients with HL achieved a CR and 1 patient with mantle cell lymphoma achieved a PR.
The investigators observed that ipilimumab did not induce or exacerbate clinical graft-versus-host disease.
Blocking PD-1
Programmed cell death protein 1 (PD-1) is a surface receptor expressed on T cells and pro-B cells. PD-1 binds 2 ligands, PD-L1 and PD-L2.
PD-1 ligands are overexpressed in inflammatory environments and attenuate the immune response through PD-1 on immune effector cells. In addition, PD-L1 expressed on malignant cells or in the tumor microenvironment suppresses tumor-infiltrating lymphocyte activity.
Pidilizumab, a humanized monoclonal antibody that binds to PD-1, weakens the apoptotic processes in lymphocytes and augments the antitumor activities of NK cells.
Investigators conducted a phase 2 trial of pidilizumab in patients with diffuse large B-cell lymphoma (DLBCL) after autologous HSCT to modulate the immune system after a transplant.
The team treated 66 patients with the antibody. At 16 months, progression-free survival (PFS) was 72%. For the 24 high-risk patients who were PET-positive after salvage chemotherapy, the 16-month PFS was 70%.
“And I think that what was most interesting,” Dr Ansell said, when focusing on the 35 patients with measurable disease after transplant, pidilizumab produced a 51% response rate “even in patients that actually had active disease.”
When pidilizumab was combined with rituximab in another trial in patients with relapsed follicular lymphoma (FL), 19 of 29 evaluable patients (66%) achieved an objective response: 15 (52%) CRs and 4 (14%) PRs.
“You might say, ‘Who cares? That’s not that great,’” Dr Ansell said. “But I think what was pretty impressive is that 52% CR rate. And most of you who treat patients with rituximab would know that that’s quite surprising, suggesting that there may be additional benefit for the use of PD-1 blockade in this subset of patients.”
Nivolumab, another monoclonal antibody that blocks the PD-1 pathway, is being investigated in a number of lymphoid malignancies, including HL, DLBCL, and T-cell lymphomas.
In a phase 1 study of nivolumab in 81 patients with relapsed or refractory lymphoid malignancies, the best preliminary overall response has been in FL and DLBCL patients, with an objective response rate of 40% and 36%, respectively, including 1 PR and 3 PRs in each subtype.
“I think what is important,” Dr Ansell said, “is that the side effects, as expected, were mainly immune-mediated, not as dramatic as have been seen with other agents, and very similar to what has been seen in solid tumor studies.”
Dr Ansell pointed out that the response rates with nivolumab varied widely by histology, suggesting that “we have a lot to learn about why patients benefit and who exactly benefits.”
There were no responses in patients with multiple myeloma or primary mediastinal B-cell lymphoma, although many patients achieved stable disease.
“Hodgkin lymphoma was completely different,” Dr Ansell said, “and there were responses in virtually every patient.”
Of 23 patients treated with nivolumab, 20 responded—4 achieved a CR and 16 a PR—including patients who had failed autologous HSCT and brentuximab vedotin treatment. Eleven patients, including 2 with CRs, have an ongoing response, some approaching 2 years. So the responses have been durable, Dr Ansell noted.
Yet another PD-1 antibody, pembrolizumab, has prompted reduction in tumor burden in HL in all but 2 of 29 evaluable patients, including 6 CRs and 13 PRs. The median duration of response has not yet been reached, and the side effect profile was similar to what has been seen with nivolumab and in solid tumors.
Activating immune stimulatory signals
Another approach to boosting the immune system is to activate immune stimulatory signals, eg, CD27 and CD40, and get a benefit that way. Varlilumab (CDX-1127) is an unconjugated monoclonal antibody that binds CD27 and activates CD27-expressing T cells.
In a phase 1 trial of varlilumab in 24 lymphoma patients, investigators found no significant depletion in absolute lymphocyte counts, T cells, or B cells. “Not quite the same success story,” Dr Ansell said, with a response—a CR—in only 1 patient.
Investigators did observe, however, evidence of increased soluble CD27, a reduction of circulating Tregs, and the induction of pro-inflammatory cytokines.
And in a phase 1 study of the anti-CD40 monoclonal antibody dacetuzumab in recurrent NHL, “the response rate was disappointingly low,” Dr Ansell said.
Investigators observed 6 objective responses, including 1 CR and 5 PRs, and a decrease in tumor size in approximately one-third of the 50 patients treated.
The investigators of the subsequent phase 2 trial did not want to take the agent forward for further study, Dr Ansell noted, “but there are antibodies now being developed in this space that will hopefully be more effective and create a greater benefit.”
Optimizing immune function
Dr Ansell suggested there are 3 main approaches to treating patients. One is going directly after the malignant cells and depleting them. A second is to inhibit critical pathways that the malignant cell is dependent upon, “starving them, if you like.” The third way is to activate the immune system and thereby create a greater benefit for patients.
“[P]robably our best strategy is to use all 3 in a reprogram approach,” he said. “Because unless you target each one of these areas, the likelihood is that the other sides of the 3-legged stool will take over.”
“This is an encouraging and exciting time for immune checkpoints therapy and an encouraging and exciting time for immune therapies in general. I think this is really the new frontier in lymphomas.” ![]()
Median DOR, PFS not yet reached for ibrutinib in CLL

Photo courtesy of
Janssen Biotech, Inc.
NEW YORK—Long-term follow-up of single-agent ibrutinib at the approved dose of 420 mg daily confirms that the Bruton’s tyrosine kinase inhibitor produces rapid and durable responses in patients with chronic lymphocytic leukemia (CLL), according to an update presented at Lymphoma & Myeloma 2015.
At up to 44 months of follow-up, the median duration of response (DOR) and progression-free survival (PFS) have not yet been reached.
At 30 months, the PFS rate was 96% for treatment-naïve patients and 76% for relapsed or refractory patients. Patients with del 17p had a median PFS of 32.4 months.
“Virtually all the patients do respond to treatment,” said Steven Coutre, MD, of Stanford University School of Medicine in California.
“Only a handful of patients achieve less than CR [complete response] or PR [partial response],” he said during his presentation at the meeting.
Phase 1/2b and extension studies
Ninety-four patients enrolled in the phase 1/2b (PCYC-1102) and extension (PCYC-1103) studies received 420 mg of ibrutinib once daily.
“We initially enrolled patients with relapsed/refractory CLL,” Dr Coutre clarified. “Then, because of the significant efficacy and safety that was observed, we added a second cohort of treatment-naïve patients age 65 and older.”
The treatment-naïve (TN) cohort consisted of 27 CLL patients. The relapsed or refractory (R/R) cohort consisted of 67 patients with CLL or small lymphocytic lymphoma, including patients with high-risk disease, which was defined as disease progression less than 24 months after the start of a chemoimmunotherapy regimen or refractory to the most recent regimen.
The median time on study was 32 months (range, 0–44).
In the TN cohort, the median age was 71, 78% were ECOG performance status 0, and most had advanced disease as indicated by Rai stage.
In the R/R cohort, the median age was 66, 40% were ECOG performance status 0, 57% were ECOG performance status 1, and 52% had bulky nodes greater than 5 cm.
“We had a significant representation of high-risk cytogenetic abnormalities,” Dr Coutre noted.
In the R/R group, 34% of patients had del 17p, and 33% had del 11q. In the TN cohort, 7% of patients had del 17p, and none had del 11q.
“There were also a significant number of cytopenias,” Dr Coutre said, “as one might expect in a heavily pretreated patient population.”
The number of prior therapies was also “quite significant,” he said, with 55% having a median of 4 or more therapies (range, 1–12).
“It really stretches the imagination to figure out what those 12 different regimens were,” he commented.
All R/R patients had prior chemotherapy, 94% a nucleoside analog, 90% an alkylator (including bendamustine), 99% anti-CD20-based therapy, 97% anti-CD20-based chemoimmunotherapy, 24% alemtuzumab, and 6% idelalisib.
The median time on treatment was 30.4 months (range, 1.3–44.2) for TN patients and 21.9 months (range, 0.3–44.6) for R/R patients. The majority of patients in both groups remain on ibrutinib—81% of the TN patients and 60% of R/R patients.
Safety
“Only 1 patient in the treatment-naïve cohort has progressed,” Dr Coutre noted. “That was a patient with deletion 17p [who progressed in about 8 months].”
The primary reasons for discontinuing therapy were progressive disease (1 TN, 11 R/R), adverse events (AEs; 3 TN, 9 R/R), consent withdrawal (1 TN, 2 R/R), investigators’ decision (0 TN, 4 R/R), and other reasons (0 TN, 1 R/R).
“Discontinuations due to AEs occurred predominantly early,” Dr Coutre observed. “So of the 12 patients [who discontinued due to AEs], 7 discontinued in the first year, 3 in the second year, and only 2 beyond year 3.”
Grade 3 or higher AEs occurred in 55 R/R patients (82%) and 17 TN patients (63%). Infection occurred in 48% of R/R patients and 11% of TN patients. Dr Coutre pointed out that most of these AEs were not related to ibrutinib.
Grade 3 or higher ibrutinib-related AEs occurred in 6 TN patients (22%) and 25 R/R patients (37%). One TN patient and 8 R/R patients experienced grade 3 or higher serious ibrutinib-related AEs.
One TN patient and 7 R/R patients required a dose reduction due to an AE. However, the dose reductions occurred predominantly during the first year, Dr Coutre noted.
Regarding time to onset of grade 3 or higher AEs, Dr Coutre said most of the events occurred early and decreased with time. Pneumonia and atrial fibrillation followed this pattern, as did neutropenia and thrombocytopenia. Hypertension was the exception, occurring during all years.
Nonhematologic AEs of grade 3 or higher that occurred in at least 5% of patients were pneumonia, hypertension, diarrhea, hyponatremia, and atrial fibrillation in TN patients, and sepsis, cellulitis, dehydration, and fatigue in R/R patients.
Hematologic AEs of grade 3 or higher in each cohort included neutropenia, thrombocytopenia, and anemia.
“The drug doesn’t seem to be myelosuppressive,” Dr Coutre noted. “We don’t have prolonged cytopenias as patients stay on treatment.”
One TN patient and 7 R/R patients died during the study.
Response and survival
The response rate (as assessed by the investigators) was 85% for TN patients. Twenty-six percent of patients achieved a complete response, 52% a partial response (PR), and 7% a PR with lymphocytosis.
The response rate for R/R patients was 94%. Nine percent achieved a complete response, 82% a PR, and 3% a PR with lymphocytosis.
The median time to the best response was 7.4 months for both cohorts.
The median DOR has not been reached in either cohort, but the 30-month DOR was 95.2% for TN patients and 79.1% for R/R patients.
The 30-month PFS was 95.8% for TN patients and 75.9% for R/R patients.
At 30 months, the PFS rate was 59.6% for patients with del 17p and 82.4% for patients with del 11q. The median PFS for patients with del 17p was 32.4 months, and it was not reached for patients with del 11q. For patients with neither of these abnormalities, the median PFS has not been reached.
“Overall survival was equally impressive,” Dr Coutre said.
The median overall survival has not been reached for any group, and 30-month overall survival is 81.3% for del 17p patients, 88.2% for patients with del 11q, and 90.3% for patients with neither abnormality.
“[I]brutinib induces rapid and durable responses that continue to improve over time . . . ,” Dr Coutre said.
He added that the drug is well-tolerated, “allowing us to continue patients on treatment, which, I think, is particularly important for these types of drugs because we clearly see that patients have significant clinical benefit, despite the fact that they still often have easily detectable disease, particularly in the bone marrow.”
“So one of the challenges is going to be [to determine] how to use these drugs on a long-term basis and [see if we can] use them in a more time-limited fashion.”
Ibrutinib is approved by the US Food and Drug Administration for 4 indications: patients with CLL who have received at least 1 prior therapy, CLL patients with del 17p, patients with mantle cell lymphoma, and patients with Waldenström’s macroglobulinemia.
Ibrutinib is distributed and marketed as Imbruvica by Pharmacyclics and also marketed by Janssen Biotech, Inc. ![]()

Photo courtesy of
Janssen Biotech, Inc.
NEW YORK—Long-term follow-up of single-agent ibrutinib at the approved dose of 420 mg daily confirms that the Bruton’s tyrosine kinase inhibitor produces rapid and durable responses in patients with chronic lymphocytic leukemia (CLL), according to an update presented at Lymphoma & Myeloma 2015.
At up to 44 months of follow-up, the median duration of response (DOR) and progression-free survival (PFS) have not yet been reached.
At 30 months, the PFS rate was 96% for treatment-naïve patients and 76% for relapsed or refractory patients. Patients with del 17p had a median PFS of 32.4 months.
“Virtually all the patients do respond to treatment,” said Steven Coutre, MD, of Stanford University School of Medicine in California.
“Only a handful of patients achieve less than CR [complete response] or PR [partial response],” he said during his presentation at the meeting.
Phase 1/2b and extension studies
Ninety-four patients enrolled in the phase 1/2b (PCYC-1102) and extension (PCYC-1103) studies received 420 mg of ibrutinib once daily.
“We initially enrolled patients with relapsed/refractory CLL,” Dr Coutre clarified. “Then, because of the significant efficacy and safety that was observed, we added a second cohort of treatment-naïve patients age 65 and older.”
The treatment-naïve (TN) cohort consisted of 27 CLL patients. The relapsed or refractory (R/R) cohort consisted of 67 patients with CLL or small lymphocytic lymphoma, including patients with high-risk disease, which was defined as disease progression less than 24 months after the start of a chemoimmunotherapy regimen or refractory to the most recent regimen.
The median time on study was 32 months (range, 0–44).
In the TN cohort, the median age was 71, 78% were ECOG performance status 0, and most had advanced disease as indicated by Rai stage.
In the R/R cohort, the median age was 66, 40% were ECOG performance status 0, 57% were ECOG performance status 1, and 52% had bulky nodes greater than 5 cm.
“We had a significant representation of high-risk cytogenetic abnormalities,” Dr Coutre noted.
In the R/R group, 34% of patients had del 17p, and 33% had del 11q. In the TN cohort, 7% of patients had del 17p, and none had del 11q.
“There were also a significant number of cytopenias,” Dr Coutre said, “as one might expect in a heavily pretreated patient population.”
The number of prior therapies was also “quite significant,” he said, with 55% having a median of 4 or more therapies (range, 1–12).
“It really stretches the imagination to figure out what those 12 different regimens were,” he commented.
All R/R patients had prior chemotherapy, 94% a nucleoside analog, 90% an alkylator (including bendamustine), 99% anti-CD20-based therapy, 97% anti-CD20-based chemoimmunotherapy, 24% alemtuzumab, and 6% idelalisib.
The median time on treatment was 30.4 months (range, 1.3–44.2) for TN patients and 21.9 months (range, 0.3–44.6) for R/R patients. The majority of patients in both groups remain on ibrutinib—81% of the TN patients and 60% of R/R patients.
Safety
“Only 1 patient in the treatment-naïve cohort has progressed,” Dr Coutre noted. “That was a patient with deletion 17p [who progressed in about 8 months].”
The primary reasons for discontinuing therapy were progressive disease (1 TN, 11 R/R), adverse events (AEs; 3 TN, 9 R/R), consent withdrawal (1 TN, 2 R/R), investigators’ decision (0 TN, 4 R/R), and other reasons (0 TN, 1 R/R).
“Discontinuations due to AEs occurred predominantly early,” Dr Coutre observed. “So of the 12 patients [who discontinued due to AEs], 7 discontinued in the first year, 3 in the second year, and only 2 beyond year 3.”
Grade 3 or higher AEs occurred in 55 R/R patients (82%) and 17 TN patients (63%). Infection occurred in 48% of R/R patients and 11% of TN patients. Dr Coutre pointed out that most of these AEs were not related to ibrutinib.
Grade 3 or higher ibrutinib-related AEs occurred in 6 TN patients (22%) and 25 R/R patients (37%). One TN patient and 8 R/R patients experienced grade 3 or higher serious ibrutinib-related AEs.
One TN patient and 7 R/R patients required a dose reduction due to an AE. However, the dose reductions occurred predominantly during the first year, Dr Coutre noted.
Regarding time to onset of grade 3 or higher AEs, Dr Coutre said most of the events occurred early and decreased with time. Pneumonia and atrial fibrillation followed this pattern, as did neutropenia and thrombocytopenia. Hypertension was the exception, occurring during all years.
Nonhematologic AEs of grade 3 or higher that occurred in at least 5% of patients were pneumonia, hypertension, diarrhea, hyponatremia, and atrial fibrillation in TN patients, and sepsis, cellulitis, dehydration, and fatigue in R/R patients.
Hematologic AEs of grade 3 or higher in each cohort included neutropenia, thrombocytopenia, and anemia.
“The drug doesn’t seem to be myelosuppressive,” Dr Coutre noted. “We don’t have prolonged cytopenias as patients stay on treatment.”
One TN patient and 7 R/R patients died during the study.
Response and survival
The response rate (as assessed by the investigators) was 85% for TN patients. Twenty-six percent of patients achieved a complete response, 52% a partial response (PR), and 7% a PR with lymphocytosis.
The response rate for R/R patients was 94%. Nine percent achieved a complete response, 82% a PR, and 3% a PR with lymphocytosis.
The median time to the best response was 7.4 months for both cohorts.
The median DOR has not been reached in either cohort, but the 30-month DOR was 95.2% for TN patients and 79.1% for R/R patients.
The 30-month PFS was 95.8% for TN patients and 75.9% for R/R patients.
At 30 months, the PFS rate was 59.6% for patients with del 17p and 82.4% for patients with del 11q. The median PFS for patients with del 17p was 32.4 months, and it was not reached for patients with del 11q. For patients with neither of these abnormalities, the median PFS has not been reached.
“Overall survival was equally impressive,” Dr Coutre said.
The median overall survival has not been reached for any group, and 30-month overall survival is 81.3% for del 17p patients, 88.2% for patients with del 11q, and 90.3% for patients with neither abnormality.
“[I]brutinib induces rapid and durable responses that continue to improve over time . . . ,” Dr Coutre said.
He added that the drug is well-tolerated, “allowing us to continue patients on treatment, which, I think, is particularly important for these types of drugs because we clearly see that patients have significant clinical benefit, despite the fact that they still often have easily detectable disease, particularly in the bone marrow.”
“So one of the challenges is going to be [to determine] how to use these drugs on a long-term basis and [see if we can] use them in a more time-limited fashion.”
Ibrutinib is approved by the US Food and Drug Administration for 4 indications: patients with CLL who have received at least 1 prior therapy, CLL patients with del 17p, patients with mantle cell lymphoma, and patients with Waldenström’s macroglobulinemia.
Ibrutinib is distributed and marketed as Imbruvica by Pharmacyclics and also marketed by Janssen Biotech, Inc. ![]()

Photo courtesy of
Janssen Biotech, Inc.
NEW YORK—Long-term follow-up of single-agent ibrutinib at the approved dose of 420 mg daily confirms that the Bruton’s tyrosine kinase inhibitor produces rapid and durable responses in patients with chronic lymphocytic leukemia (CLL), according to an update presented at Lymphoma & Myeloma 2015.
At up to 44 months of follow-up, the median duration of response (DOR) and progression-free survival (PFS) have not yet been reached.
At 30 months, the PFS rate was 96% for treatment-naïve patients and 76% for relapsed or refractory patients. Patients with del 17p had a median PFS of 32.4 months.
“Virtually all the patients do respond to treatment,” said Steven Coutre, MD, of Stanford University School of Medicine in California.
“Only a handful of patients achieve less than CR [complete response] or PR [partial response],” he said during his presentation at the meeting.
Phase 1/2b and extension studies
Ninety-four patients enrolled in the phase 1/2b (PCYC-1102) and extension (PCYC-1103) studies received 420 mg of ibrutinib once daily.
“We initially enrolled patients with relapsed/refractory CLL,” Dr Coutre clarified. “Then, because of the significant efficacy and safety that was observed, we added a second cohort of treatment-naïve patients age 65 and older.”
The treatment-naïve (TN) cohort consisted of 27 CLL patients. The relapsed or refractory (R/R) cohort consisted of 67 patients with CLL or small lymphocytic lymphoma, including patients with high-risk disease, which was defined as disease progression less than 24 months after the start of a chemoimmunotherapy regimen or refractory to the most recent regimen.
The median time on study was 32 months (range, 0–44).
In the TN cohort, the median age was 71, 78% were ECOG performance status 0, and most had advanced disease as indicated by Rai stage.
In the R/R cohort, the median age was 66, 40% were ECOG performance status 0, 57% were ECOG performance status 1, and 52% had bulky nodes greater than 5 cm.
“We had a significant representation of high-risk cytogenetic abnormalities,” Dr Coutre noted.
In the R/R group, 34% of patients had del 17p, and 33% had del 11q. In the TN cohort, 7% of patients had del 17p, and none had del 11q.
“There were also a significant number of cytopenias,” Dr Coutre said, “as one might expect in a heavily pretreated patient population.”
The number of prior therapies was also “quite significant,” he said, with 55% having a median of 4 or more therapies (range, 1–12).
“It really stretches the imagination to figure out what those 12 different regimens were,” he commented.
All R/R patients had prior chemotherapy, 94% a nucleoside analog, 90% an alkylator (including bendamustine), 99% anti-CD20-based therapy, 97% anti-CD20-based chemoimmunotherapy, 24% alemtuzumab, and 6% idelalisib.
The median time on treatment was 30.4 months (range, 1.3–44.2) for TN patients and 21.9 months (range, 0.3–44.6) for R/R patients. The majority of patients in both groups remain on ibrutinib—81% of the TN patients and 60% of R/R patients.
Safety
“Only 1 patient in the treatment-naïve cohort has progressed,” Dr Coutre noted. “That was a patient with deletion 17p [who progressed in about 8 months].”
The primary reasons for discontinuing therapy were progressive disease (1 TN, 11 R/R), adverse events (AEs; 3 TN, 9 R/R), consent withdrawal (1 TN, 2 R/R), investigators’ decision (0 TN, 4 R/R), and other reasons (0 TN, 1 R/R).
“Discontinuations due to AEs occurred predominantly early,” Dr Coutre observed. “So of the 12 patients [who discontinued due to AEs], 7 discontinued in the first year, 3 in the second year, and only 2 beyond year 3.”
Grade 3 or higher AEs occurred in 55 R/R patients (82%) and 17 TN patients (63%). Infection occurred in 48% of R/R patients and 11% of TN patients. Dr Coutre pointed out that most of these AEs were not related to ibrutinib.
Grade 3 or higher ibrutinib-related AEs occurred in 6 TN patients (22%) and 25 R/R patients (37%). One TN patient and 8 R/R patients experienced grade 3 or higher serious ibrutinib-related AEs.
One TN patient and 7 R/R patients required a dose reduction due to an AE. However, the dose reductions occurred predominantly during the first year, Dr Coutre noted.
Regarding time to onset of grade 3 or higher AEs, Dr Coutre said most of the events occurred early and decreased with time. Pneumonia and atrial fibrillation followed this pattern, as did neutropenia and thrombocytopenia. Hypertension was the exception, occurring during all years.
Nonhematologic AEs of grade 3 or higher that occurred in at least 5% of patients were pneumonia, hypertension, diarrhea, hyponatremia, and atrial fibrillation in TN patients, and sepsis, cellulitis, dehydration, and fatigue in R/R patients.
Hematologic AEs of grade 3 or higher in each cohort included neutropenia, thrombocytopenia, and anemia.
“The drug doesn’t seem to be myelosuppressive,” Dr Coutre noted. “We don’t have prolonged cytopenias as patients stay on treatment.”
One TN patient and 7 R/R patients died during the study.
Response and survival
The response rate (as assessed by the investigators) was 85% for TN patients. Twenty-six percent of patients achieved a complete response, 52% a partial response (PR), and 7% a PR with lymphocytosis.
The response rate for R/R patients was 94%. Nine percent achieved a complete response, 82% a PR, and 3% a PR with lymphocytosis.
The median time to the best response was 7.4 months for both cohorts.
The median DOR has not been reached in either cohort, but the 30-month DOR was 95.2% for TN patients and 79.1% for R/R patients.
The 30-month PFS was 95.8% for TN patients and 75.9% for R/R patients.
At 30 months, the PFS rate was 59.6% for patients with del 17p and 82.4% for patients with del 11q. The median PFS for patients with del 17p was 32.4 months, and it was not reached for patients with del 11q. For patients with neither of these abnormalities, the median PFS has not been reached.
“Overall survival was equally impressive,” Dr Coutre said.
The median overall survival has not been reached for any group, and 30-month overall survival is 81.3% for del 17p patients, 88.2% for patients with del 11q, and 90.3% for patients with neither abnormality.
“[I]brutinib induces rapid and durable responses that continue to improve over time . . . ,” Dr Coutre said.
He added that the drug is well-tolerated, “allowing us to continue patients on treatment, which, I think, is particularly important for these types of drugs because we clearly see that patients have significant clinical benefit, despite the fact that they still often have easily detectable disease, particularly in the bone marrow.”
“So one of the challenges is going to be [to determine] how to use these drugs on a long-term basis and [see if we can] use them in a more time-limited fashion.”
Ibrutinib is approved by the US Food and Drug Administration for 4 indications: patients with CLL who have received at least 1 prior therapy, CLL patients with del 17p, patients with mantle cell lymphoma, and patients with Waldenström’s macroglobulinemia.
Ibrutinib is distributed and marketed as Imbruvica by Pharmacyclics and also marketed by Janssen Biotech, Inc. ![]()
Cancer care in Latin America

patient and her father
Photo by Rhoda Baer
Despite progress made in cancer care in Latin America in the last 2 years, substantial barriers remain to ensure optimal clinical management, according to a report commissioned by The Lancet Oncology.
The report, an update from a report published in 2013, details a number of improvements in cancer care in Latin America, either specifically related to cancer or to general healthcare initiatives that will also benefit cancer patients.
However, the updated report also suggests that major changes are needed in many areas to increase the standard of cancer care in Latin America.
Progress made
According to the report, progress has been made in the following areas.
The proportion of people in Latin America affiliated with any kind of health insurance program grew from 46% to 60% between 2008 and 2013.
For 2014, the World Health Organization (WHO) reported an 8% increase in the number of countries (60% of the whole region) with a National Cancer Plan. The following countries have newly adopted plans: Suriname, Ecuador, Dominican Republic, Trinidad and Tobago, Puerto Rico, Peru, El Salvador, and Colombia.
In addition, Latin America—most notably, Brazil and Argentina—has begun to address the shortage of cancer specialists.
Brazil has shown an increase of 77% in oncologists since 2011 (from 1457 to 2577). Concurrently, the number of hematologists has also increased by 40% (from 1420 in 2011 to 1985 in 2015), and that of radiotherapists by 12% (from 444 in 2011 to 497 in 2015). These rises are in the context of an 11% increase in cancer cases in Brazil (from 518,000 new cases in 2012 to 576,000 in 2014).
Many countries across Latin America have signed on to the Global Action Plan for the Prevention and Control of Non-Communicable Diseases 2013-2020, endorsed by the WHO, which aims to achieve a 25% reduction in premature mortality from non-communicable diseases (including cancer) by 2025.
The Colombian Ministry of Health and Social Protection has expanded its social insurance program to cover all types of cancer.
Since January 2014, the administration of chemotherapy and radiation treatments is free of charge in Uruguay.
The Atlas of Palliative Care was published in Latin America, which revealed a growth of more than 400% in the number of palliative services since 2006.
Room for improvement
The report indicates that the following issues are still problems in Latin America.
Compared with high-income countries, Latin America in 2015 remains behind in terms of public expenditure on health and cancer care.
Argentina and Mexico spend around 6% of their gross national product on healthcare, compared to 9% for the UK, 11% for Germany, and 17% for the US, which reflects a gap between Latin American and other countries not only proportionately but also in terms of absolute dollars. Only Brazil, at 9%, is close to the proportion spent in high-income countries.
In Latin America, only Brazil, Cuba, Costa Rica, and Uruguay are considered to have integration of social security and public insurance, and only Brazil, Cuba, and Costa Rica can be judged to have universal healthcare.
Many countries still have no specific training in palliative care (including Bolivia, El Salvador, Honduras, and Nicaragua).
Additionally, data from 2002 showed that Latin America accounted for less than 1% of the world’s opioid drug consumption for pain relief. Consumption of strong opioids still lags behind developed countries today, with no Latin American country exceeding 15 mg/capita per year.
Under-implementation of new technologies has not improved substantially since the previous Lancet Oncology Commission in 2013. There are a few exceptions, however, such as PET scanning technology improvements in Uruguay.
Pharmaceutical trials for expensive new anticancer therapies are largely unhelpful to most patients in Latin America. Patients participating in trials of expensive new anticancer therapies sometimes cannot complete treatment once their trial ends, and the trials often do not lead to approval in these regions.
There are often geographical disparities where most cancer specialists are located in major hospitals in big cities, requiring patients from rural and remote areas to travel far distances to these hospitals for cancer care.
In addition, waiting times in these centers can be unacceptably long, with reports from Mexico and Brazil describing median waiting times of 7 months or more for patients with breast cancer from symptomatic presentation to initial treatment.
Better cancer registries are desperately needed in all Latin American countries to more accurately quantify the cancer burden in the region and the resources required to combat it, according to the report. ![]()

patient and her father
Photo by Rhoda Baer
Despite progress made in cancer care in Latin America in the last 2 years, substantial barriers remain to ensure optimal clinical management, according to a report commissioned by The Lancet Oncology.
The report, an update from a report published in 2013, details a number of improvements in cancer care in Latin America, either specifically related to cancer or to general healthcare initiatives that will also benefit cancer patients.
However, the updated report also suggests that major changes are needed in many areas to increase the standard of cancer care in Latin America.
Progress made
According to the report, progress has been made in the following areas.
The proportion of people in Latin America affiliated with any kind of health insurance program grew from 46% to 60% between 2008 and 2013.
For 2014, the World Health Organization (WHO) reported an 8% increase in the number of countries (60% of the whole region) with a National Cancer Plan. The following countries have newly adopted plans: Suriname, Ecuador, Dominican Republic, Trinidad and Tobago, Puerto Rico, Peru, El Salvador, and Colombia.
In addition, Latin America—most notably, Brazil and Argentina—has begun to address the shortage of cancer specialists.
Brazil has shown an increase of 77% in oncologists since 2011 (from 1457 to 2577). Concurrently, the number of hematologists has also increased by 40% (from 1420 in 2011 to 1985 in 2015), and that of radiotherapists by 12% (from 444 in 2011 to 497 in 2015). These rises are in the context of an 11% increase in cancer cases in Brazil (from 518,000 new cases in 2012 to 576,000 in 2014).
Many countries across Latin America have signed on to the Global Action Plan for the Prevention and Control of Non-Communicable Diseases 2013-2020, endorsed by the WHO, which aims to achieve a 25% reduction in premature mortality from non-communicable diseases (including cancer) by 2025.
The Colombian Ministry of Health and Social Protection has expanded its social insurance program to cover all types of cancer.
Since January 2014, the administration of chemotherapy and radiation treatments is free of charge in Uruguay.
The Atlas of Palliative Care was published in Latin America, which revealed a growth of more than 400% in the number of palliative services since 2006.
Room for improvement
The report indicates that the following issues are still problems in Latin America.
Compared with high-income countries, Latin America in 2015 remains behind in terms of public expenditure on health and cancer care.
Argentina and Mexico spend around 6% of their gross national product on healthcare, compared to 9% for the UK, 11% for Germany, and 17% for the US, which reflects a gap between Latin American and other countries not only proportionately but also in terms of absolute dollars. Only Brazil, at 9%, is close to the proportion spent in high-income countries.
In Latin America, only Brazil, Cuba, Costa Rica, and Uruguay are considered to have integration of social security and public insurance, and only Brazil, Cuba, and Costa Rica can be judged to have universal healthcare.
Many countries still have no specific training in palliative care (including Bolivia, El Salvador, Honduras, and Nicaragua).
Additionally, data from 2002 showed that Latin America accounted for less than 1% of the world’s opioid drug consumption for pain relief. Consumption of strong opioids still lags behind developed countries today, with no Latin American country exceeding 15 mg/capita per year.
Under-implementation of new technologies has not improved substantially since the previous Lancet Oncology Commission in 2013. There are a few exceptions, however, such as PET scanning technology improvements in Uruguay.
Pharmaceutical trials for expensive new anticancer therapies are largely unhelpful to most patients in Latin America. Patients participating in trials of expensive new anticancer therapies sometimes cannot complete treatment once their trial ends, and the trials often do not lead to approval in these regions.
There are often geographical disparities where most cancer specialists are located in major hospitals in big cities, requiring patients from rural and remote areas to travel far distances to these hospitals for cancer care.
In addition, waiting times in these centers can be unacceptably long, with reports from Mexico and Brazil describing median waiting times of 7 months or more for patients with breast cancer from symptomatic presentation to initial treatment.
Better cancer registries are desperately needed in all Latin American countries to more accurately quantify the cancer burden in the region and the resources required to combat it, according to the report. ![]()

patient and her father
Photo by Rhoda Baer
Despite progress made in cancer care in Latin America in the last 2 years, substantial barriers remain to ensure optimal clinical management, according to a report commissioned by The Lancet Oncology.
The report, an update from a report published in 2013, details a number of improvements in cancer care in Latin America, either specifically related to cancer or to general healthcare initiatives that will also benefit cancer patients.
However, the updated report also suggests that major changes are needed in many areas to increase the standard of cancer care in Latin America.
Progress made
According to the report, progress has been made in the following areas.
The proportion of people in Latin America affiliated with any kind of health insurance program grew from 46% to 60% between 2008 and 2013.
For 2014, the World Health Organization (WHO) reported an 8% increase in the number of countries (60% of the whole region) with a National Cancer Plan. The following countries have newly adopted plans: Suriname, Ecuador, Dominican Republic, Trinidad and Tobago, Puerto Rico, Peru, El Salvador, and Colombia.
In addition, Latin America—most notably, Brazil and Argentina—has begun to address the shortage of cancer specialists.
Brazil has shown an increase of 77% in oncologists since 2011 (from 1457 to 2577). Concurrently, the number of hematologists has also increased by 40% (from 1420 in 2011 to 1985 in 2015), and that of radiotherapists by 12% (from 444 in 2011 to 497 in 2015). These rises are in the context of an 11% increase in cancer cases in Brazil (from 518,000 new cases in 2012 to 576,000 in 2014).
Many countries across Latin America have signed on to the Global Action Plan for the Prevention and Control of Non-Communicable Diseases 2013-2020, endorsed by the WHO, which aims to achieve a 25% reduction in premature mortality from non-communicable diseases (including cancer) by 2025.
The Colombian Ministry of Health and Social Protection has expanded its social insurance program to cover all types of cancer.
Since January 2014, the administration of chemotherapy and radiation treatments is free of charge in Uruguay.
The Atlas of Palliative Care was published in Latin America, which revealed a growth of more than 400% in the number of palliative services since 2006.
Room for improvement
The report indicates that the following issues are still problems in Latin America.
Compared with high-income countries, Latin America in 2015 remains behind in terms of public expenditure on health and cancer care.
Argentina and Mexico spend around 6% of their gross national product on healthcare, compared to 9% for the UK, 11% for Germany, and 17% for the US, which reflects a gap between Latin American and other countries not only proportionately but also in terms of absolute dollars. Only Brazil, at 9%, is close to the proportion spent in high-income countries.
In Latin America, only Brazil, Cuba, Costa Rica, and Uruguay are considered to have integration of social security and public insurance, and only Brazil, Cuba, and Costa Rica can be judged to have universal healthcare.
Many countries still have no specific training in palliative care (including Bolivia, El Salvador, Honduras, and Nicaragua).
Additionally, data from 2002 showed that Latin America accounted for less than 1% of the world’s opioid drug consumption for pain relief. Consumption of strong opioids still lags behind developed countries today, with no Latin American country exceeding 15 mg/capita per year.
Under-implementation of new technologies has not improved substantially since the previous Lancet Oncology Commission in 2013. There are a few exceptions, however, such as PET scanning technology improvements in Uruguay.
Pharmaceutical trials for expensive new anticancer therapies are largely unhelpful to most patients in Latin America. Patients participating in trials of expensive new anticancer therapies sometimes cannot complete treatment once their trial ends, and the trials often do not lead to approval in these regions.
There are often geographical disparities where most cancer specialists are located in major hospitals in big cities, requiring patients from rural and remote areas to travel far distances to these hospitals for cancer care.
In addition, waiting times in these centers can be unacceptably long, with reports from Mexico and Brazil describing median waiting times of 7 months or more for patients with breast cancer from symptomatic presentation to initial treatment.
Better cancer registries are desperately needed in all Latin American countries to more accurately quantify the cancer burden in the region and the resources required to combat it, according to the report. ![]()
The search continues for additional targets in CLL
showing CLL
Image by Mary Ann Thompson
NEW YORK—Despite enormous advances in therapies for chronic lymphocytic leukemia (CLL) that target the B-cell receptor (BCR) signaling pathway, there is still room for improvement, according to investigators at the Mayo Clinic.
Bruton’s tyrosine kinase (BTK) and phosphoinositide-3 kinase delta (PI3Kδ) inhibitors are major players in mediating BCR signaling, yet both have off-target effects.
“These agents are not curative, and resistance develops,” explained Neil Kay, MD, of the Mayo Clinic in Rochester, Minnesota.
He described the Axl receptor tyrosine kinase and its inhibitor, TP-0903, at Lymphoma & Myeloma 2015, and discussed how it may be another promising target for CLL therapy.
Pros and cons of current therapies
Ibrutinib, a potent, irreversible, covalent inhibitor of BTK, inhibits interleukin 2 kinase, an essential enzyme in Th2 T cells.
“The potential benefit of this,” Dr Kay said, “is it shifts the balance between Th1 and Th2 T cells and potentially enhances antitumor immune responses.”
However, ibrutinib may cause off-target effects, including defects of platelet function, atrial fibrillation probably related to the inhibition of cardiac PI3K-AKT signaling, and decreased efficacy of anti-CD20 antibodies as mediated by natural killer cells.
In addition, long-term exposure to ibrutinib can induce signal mutations associated with resistance.
Idelalisib, a reversible inhibitor of the delta isoform of PI3K, modulates malignant B-cell signaling and inhibits the chemokines CCL3 and 4. It does not inhibit antibody-dependent cell-mediated cytotoxicity mediated by anti-CD20 antibodies, and it may stimulate antitumor responses.
However, idelalisib may also increase the incidence of diarrhea and colitis and can cause transaminitis and pneumonitis.
Axl receptor tyrosine kinase
So Dr Kay and fellow researchers explored whether CLL B cells express other active receptor tyrosine kinases (RTKs), and if so, what their functional implication is in CLL B-cell survival.
And what they detected is active Axl RTK and basic fibroblast growth factor receptor 3 (FGFR3) RTK in CLL B cells. The human Axl gene, a member of the TAM family of RTKs, is located on chromosome 19q13.2 and encodes a protein weighing 100–140 kD.
When Axl is activated, it initiates various signaling pathways, including cell survival, proliferation, apoptosis inhibition, migration, cell adhesion, and cytokine production. It does this through interactions with a number of signaling molecules, including PI3K and phospholipase C γ2 (PLCγ2), among others.
Most important, Axl is overexpressed in a number of human malignancies, including acute myeloid leukemia, and is associated with poor survivorship.
The research team immunoprecipitated Axl RTK from CLL B-cell lysates and examined them for co-localization by Western blot for the proteins PI3K, c-Src, Syk, PLCγ2, ZAP-70, and Lyn.
“What we think currently is that Axl provides a docking site for these key signaling molecules,” Dr Kay said.
The team found that, by inhibiting Axl with TP-0903, they were able to induce robust apoptosis in CLL B cells, including those from high-risk 17p– and 11q– CLL. They also observed that Axl inhibition resulted in significant reduction of the anti-apoptotic proteins Bcl-2, XIAP, and Mcl-1 and upregulation of the pro-apoptotic protein BIM in CLL B cells.
They then tested the BTK inhibitors ibrutinib and TP-4216 with and without the Axl inhibitor TP-0903 and found that concurrent administration of TP-0903 with the reversible BTK inhibitor TP-4216 had additive effects on apoptosis. This was not the case when it was added to ibrutinib.
“There was much more dramatic upregulation with the reversible BTK inhibitor,” Dr Kay emphasized.
He said Axl inhibition is a therapeutic opportunity in that these inhibitors are orally bioavailable and would be candidates for clinical testing. These inhibitors may minimize drug resistance and prevent the emergence of Richter’s transformation, he added.
The researchers received TP-0903 from Tolero Pharmaceuticals. ![]()

showing CLL
Image by Mary Ann Thompson
NEW YORK—Despite enormous advances in therapies for chronic lymphocytic leukemia (CLL) that target the B-cell receptor (BCR) signaling pathway, there is still room for improvement, according to investigators at the Mayo Clinic.
Bruton’s tyrosine kinase (BTK) and phosphoinositide-3 kinase delta (PI3Kδ) inhibitors are major players in mediating BCR signaling, yet both have off-target effects.
“These agents are not curative, and resistance develops,” explained Neil Kay, MD, of the Mayo Clinic in Rochester, Minnesota.
He described the Axl receptor tyrosine kinase and its inhibitor, TP-0903, at Lymphoma & Myeloma 2015, and discussed how it may be another promising target for CLL therapy.
Pros and cons of current therapies
Ibrutinib, a potent, irreversible, covalent inhibitor of BTK, inhibits interleukin 2 kinase, an essential enzyme in Th2 T cells.
“The potential benefit of this,” Dr Kay said, “is it shifts the balance between Th1 and Th2 T cells and potentially enhances antitumor immune responses.”
However, ibrutinib may cause off-target effects, including defects of platelet function, atrial fibrillation probably related to the inhibition of cardiac PI3K-AKT signaling, and decreased efficacy of anti-CD20 antibodies as mediated by natural killer cells.
In addition, long-term exposure to ibrutinib can induce signal mutations associated with resistance.
Idelalisib, a reversible inhibitor of the delta isoform of PI3K, modulates malignant B-cell signaling and inhibits the chemokines CCL3 and 4. It does not inhibit antibody-dependent cell-mediated cytotoxicity mediated by anti-CD20 antibodies, and it may stimulate antitumor responses.
However, idelalisib may also increase the incidence of diarrhea and colitis and can cause transaminitis and pneumonitis.
Axl receptor tyrosine kinase
So Dr Kay and fellow researchers explored whether CLL B cells express other active receptor tyrosine kinases (RTKs), and if so, what their functional implication is in CLL B-cell survival.
And what they detected is active Axl RTK and basic fibroblast growth factor receptor 3 (FGFR3) RTK in CLL B cells. The human Axl gene, a member of the TAM family of RTKs, is located on chromosome 19q13.2 and encodes a protein weighing 100–140 kD.
When Axl is activated, it initiates various signaling pathways, including cell survival, proliferation, apoptosis inhibition, migration, cell adhesion, and cytokine production. It does this through interactions with a number of signaling molecules, including PI3K and phospholipase C γ2 (PLCγ2), among others.
Most important, Axl is overexpressed in a number of human malignancies, including acute myeloid leukemia, and is associated with poor survivorship.
The research team immunoprecipitated Axl RTK from CLL B-cell lysates and examined them for co-localization by Western blot for the proteins PI3K, c-Src, Syk, PLCγ2, ZAP-70, and Lyn.
“What we think currently is that Axl provides a docking site for these key signaling molecules,” Dr Kay said.
The team found that, by inhibiting Axl with TP-0903, they were able to induce robust apoptosis in CLL B cells, including those from high-risk 17p– and 11q– CLL. They also observed that Axl inhibition resulted in significant reduction of the anti-apoptotic proteins Bcl-2, XIAP, and Mcl-1 and upregulation of the pro-apoptotic protein BIM in CLL B cells.
They then tested the BTK inhibitors ibrutinib and TP-4216 with and without the Axl inhibitor TP-0903 and found that concurrent administration of TP-0903 with the reversible BTK inhibitor TP-4216 had additive effects on apoptosis. This was not the case when it was added to ibrutinib.
“There was much more dramatic upregulation with the reversible BTK inhibitor,” Dr Kay emphasized.
He said Axl inhibition is a therapeutic opportunity in that these inhibitors are orally bioavailable and would be candidates for clinical testing. These inhibitors may minimize drug resistance and prevent the emergence of Richter’s transformation, he added.
The researchers received TP-0903 from Tolero Pharmaceuticals. ![]()

showing CLL
Image by Mary Ann Thompson
NEW YORK—Despite enormous advances in therapies for chronic lymphocytic leukemia (CLL) that target the B-cell receptor (BCR) signaling pathway, there is still room for improvement, according to investigators at the Mayo Clinic.
Bruton’s tyrosine kinase (BTK) and phosphoinositide-3 kinase delta (PI3Kδ) inhibitors are major players in mediating BCR signaling, yet both have off-target effects.
“These agents are not curative, and resistance develops,” explained Neil Kay, MD, of the Mayo Clinic in Rochester, Minnesota.
He described the Axl receptor tyrosine kinase and its inhibitor, TP-0903, at Lymphoma & Myeloma 2015, and discussed how it may be another promising target for CLL therapy.
Pros and cons of current therapies
Ibrutinib, a potent, irreversible, covalent inhibitor of BTK, inhibits interleukin 2 kinase, an essential enzyme in Th2 T cells.
“The potential benefit of this,” Dr Kay said, “is it shifts the balance between Th1 and Th2 T cells and potentially enhances antitumor immune responses.”
However, ibrutinib may cause off-target effects, including defects of platelet function, atrial fibrillation probably related to the inhibition of cardiac PI3K-AKT signaling, and decreased efficacy of anti-CD20 antibodies as mediated by natural killer cells.
In addition, long-term exposure to ibrutinib can induce signal mutations associated with resistance.
Idelalisib, a reversible inhibitor of the delta isoform of PI3K, modulates malignant B-cell signaling and inhibits the chemokines CCL3 and 4. It does not inhibit antibody-dependent cell-mediated cytotoxicity mediated by anti-CD20 antibodies, and it may stimulate antitumor responses.
However, idelalisib may also increase the incidence of diarrhea and colitis and can cause transaminitis and pneumonitis.
Axl receptor tyrosine kinase
So Dr Kay and fellow researchers explored whether CLL B cells express other active receptor tyrosine kinases (RTKs), and if so, what their functional implication is in CLL B-cell survival.
And what they detected is active Axl RTK and basic fibroblast growth factor receptor 3 (FGFR3) RTK in CLL B cells. The human Axl gene, a member of the TAM family of RTKs, is located on chromosome 19q13.2 and encodes a protein weighing 100–140 kD.
When Axl is activated, it initiates various signaling pathways, including cell survival, proliferation, apoptosis inhibition, migration, cell adhesion, and cytokine production. It does this through interactions with a number of signaling molecules, including PI3K and phospholipase C γ2 (PLCγ2), among others.
Most important, Axl is overexpressed in a number of human malignancies, including acute myeloid leukemia, and is associated with poor survivorship.
The research team immunoprecipitated Axl RTK from CLL B-cell lysates and examined them for co-localization by Western blot for the proteins PI3K, c-Src, Syk, PLCγ2, ZAP-70, and Lyn.
“What we think currently is that Axl provides a docking site for these key signaling molecules,” Dr Kay said.
The team found that, by inhibiting Axl with TP-0903, they were able to induce robust apoptosis in CLL B cells, including those from high-risk 17p– and 11q– CLL. They also observed that Axl inhibition resulted in significant reduction of the anti-apoptotic proteins Bcl-2, XIAP, and Mcl-1 and upregulation of the pro-apoptotic protein BIM in CLL B cells.
They then tested the BTK inhibitors ibrutinib and TP-4216 with and without the Axl inhibitor TP-0903 and found that concurrent administration of TP-0903 with the reversible BTK inhibitor TP-4216 had additive effects on apoptosis. This was not the case when it was added to ibrutinib.
“There was much more dramatic upregulation with the reversible BTK inhibitor,” Dr Kay emphasized.
He said Axl inhibition is a therapeutic opportunity in that these inhibitors are orally bioavailable and would be candidates for clinical testing. These inhibitors may minimize drug resistance and prevent the emergence of Richter’s transformation, he added.
The researchers received TP-0903 from Tolero Pharmaceuticals. ![]()
MicroRNA may be therapeutic target for MF

A Notch-related microRNA may be a therapeutic target for mycosis fungoides (MF), according to research published in the Journal of Investigative Dermatology.
The Notch pathway has been implicated in the progression of cutaneous T-cell lymphomas, but the mechanisms driving Notch activation has been unclear.
So investigators studied a series of skin samples from patients with MF in tumor phase, focusing on the Notch pathway.
“The purpose of this project has been to research the state of the Notch pathway in a series of samples from patients with mycosis fungoides and compare the results to a control group to discover if Notch activation in tumors is influenced by epigenetic modifications,” said Fernando Gallardo, MD, of Hospital del Mar Investigacions Mèdiques in Barcelona, Spain.
So he and his colleagues looked at methylation patterns in several components of the Notch pathway and confirmed that Notch1 was activated in samples from patients with MF.
They then identified a microRNA, miR-200C, that was epigenetically repressed in the samples. Further investigation revealed that this repression leads to the activation of the Notch pathway.
“The restoration of miR-200C expression, silenced in the tumor cells, could represent a potential therapeutic target for this subtype of lymphomas,” Dr Gallardo concluded. ![]()

A Notch-related microRNA may be a therapeutic target for mycosis fungoides (MF), according to research published in the Journal of Investigative Dermatology.
The Notch pathway has been implicated in the progression of cutaneous T-cell lymphomas, but the mechanisms driving Notch activation has been unclear.
So investigators studied a series of skin samples from patients with MF in tumor phase, focusing on the Notch pathway.
“The purpose of this project has been to research the state of the Notch pathway in a series of samples from patients with mycosis fungoides and compare the results to a control group to discover if Notch activation in tumors is influenced by epigenetic modifications,” said Fernando Gallardo, MD, of Hospital del Mar Investigacions Mèdiques in Barcelona, Spain.
So he and his colleagues looked at methylation patterns in several components of the Notch pathway and confirmed that Notch1 was activated in samples from patients with MF.
They then identified a microRNA, miR-200C, that was epigenetically repressed in the samples. Further investigation revealed that this repression leads to the activation of the Notch pathway.
“The restoration of miR-200C expression, silenced in the tumor cells, could represent a potential therapeutic target for this subtype of lymphomas,” Dr Gallardo concluded. ![]()

A Notch-related microRNA may be a therapeutic target for mycosis fungoides (MF), according to research published in the Journal of Investigative Dermatology.
The Notch pathway has been implicated in the progression of cutaneous T-cell lymphomas, but the mechanisms driving Notch activation has been unclear.
So investigators studied a series of skin samples from patients with MF in tumor phase, focusing on the Notch pathway.
“The purpose of this project has been to research the state of the Notch pathway in a series of samples from patients with mycosis fungoides and compare the results to a control group to discover if Notch activation in tumors is influenced by epigenetic modifications,” said Fernando Gallardo, MD, of Hospital del Mar Investigacions Mèdiques in Barcelona, Spain.
So he and his colleagues looked at methylation patterns in several components of the Notch pathway and confirmed that Notch1 was activated in samples from patients with MF.
They then identified a microRNA, miR-200C, that was epigenetically repressed in the samples. Further investigation revealed that this repression leads to the activation of the Notch pathway.
“The restoration of miR-200C expression, silenced in the tumor cells, could represent a potential therapeutic target for this subtype of lymphomas,” Dr Gallardo concluded. ![]()
Rapid, cheap test prognostic in multiple myeloma
A rapid, low-cost assay measuring DNA and immunoglobulin in multiple myeloma cells is both a powerful predictor of prognosis and a tool for identifying new, previously unidentified therapeutic targets, its developers say.
The test, labeled DNA/CIG, uses two-color flow cytometry to evaluate DNA in the nucleus and immunoglobulin in the cytoplasm of bone marrow taken from patients with multiple myeloma (MM) at baseline, before the start of the aggressive “Total Therapy 3b” (TT3b) protocol developed at the Myeloma Institute for Research and Therapy at the University of Arkansas for Medical Sciences in Little Rock.
The light chain cytoplasmic immunoglobulin (CIg) index generated by the test, combined with an associated 12-gene panel, serves as an independent prognostic factor in patients with myeloma. The CIg index is a marker for immunoglobulin production in myeloma cells, with lower index scores suggesting ineffective immunologic responses.
“We show that the presence of low CIg as detected by DNA/CIG is a major adverse prognostic factor in TT3b, even when other GEP [gene expression profiling]–derived prognostic factors were accounted for,” Dr. Xenon Papanikolaou of the University of Arkansas and colleagues wrote in the journal Leukemia (2015;29:1713-20).
The investigators performed DNA/CIG as part of the routine work-up of patients with newly diagnosed MM who were scheduled to undergo the current iteration of the Total Therapy protocol, consisting of two induction chemotherapy cycles of VTD-PACE (bortezomib, thalidomide, dexamethasone and 4-day continuous infusions of cisplatin, doxorubicin, cyclophosphamide and etoposide), two planned hematopoietic stem cell transplants, and 3 years of planned maintenance with bortezomib, lenalidomide and dexamethasone.
The authors looked at the association of the CIg index, gene-expression profiling, and standard prognostic measures with both overall and progression-free survival (OS and PFS).
They found that both the percentage of light chain–restricted cells and the CIg as determined by DNA/CIG were associated with poor clinical outcomes. Additionally, a CIg lower than 2.8, albumin lower than 3.5 g/dL, and age 65 and older were significantly associated with worse PFS and OS, independent of gene-expression profiling data.
Any CIg lower than 2.8 was associated with a twofold risk for worse OS (hazard ratio, 2.08; P = .012) and a nearly doubling of risk for worse PFS (HR, 1.84; P = .001).
In multivariate models, CIg remained an independent predictor for prognosis when GEP-defined high-risk and low albumin were added in. CIg was also linked to other prognostic factors, including beta2-microglobulin greater than 5.5 mg/L, percentage of LCR cells exceeding 50%, C-reactive protein equal to or greater than 8 mg/L, and GEP-derived high centrosome index.
Low CIg was also associated with an experimental group of 12 gene probes believed to be involved in cell cycle regulation, cell differentiation, and cellular drug transport. The investigators used the gene signatures to develop a risk score, called GEP12, that offered important prognostic information for patients in TT3b and in earlier clinical studies.
The finding that a low CIg index was linked to a high centrosome index as defined by GEP points to a possible role for CIg index as a treatment selection tool.
“Interestingly, a centrosome inhibitor has shown promising activity in preclinical models of MM, thus potentially providing a selective drug for patients with low CIg myeloma,” the authors write.
The study was supported in part by a grant from the National Cancer Institute. Senior author Dr. Bart Barlogies reports research funding from Celgene Corp and Millennium Pharmaceuticals, and consulting with those companies as well as Onyx Pharmaceuticals and Amgen. He is a coinventor on patents and patent applications related to use of gene expression profiling in cancer medicine that have been licensed to Myeloma Health, but has no financial interests in this company. The remaining authors reported having no conflicts of interest.
A rapid, low-cost assay measuring DNA and immunoglobulin in multiple myeloma cells is both a powerful predictor of prognosis and a tool for identifying new, previously unidentified therapeutic targets, its developers say.
The test, labeled DNA/CIG, uses two-color flow cytometry to evaluate DNA in the nucleus and immunoglobulin in the cytoplasm of bone marrow taken from patients with multiple myeloma (MM) at baseline, before the start of the aggressive “Total Therapy 3b” (TT3b) protocol developed at the Myeloma Institute for Research and Therapy at the University of Arkansas for Medical Sciences in Little Rock.
The light chain cytoplasmic immunoglobulin (CIg) index generated by the test, combined with an associated 12-gene panel, serves as an independent prognostic factor in patients with myeloma. The CIg index is a marker for immunoglobulin production in myeloma cells, with lower index scores suggesting ineffective immunologic responses.
“We show that the presence of low CIg as detected by DNA/CIG is a major adverse prognostic factor in TT3b, even when other GEP [gene expression profiling]–derived prognostic factors were accounted for,” Dr. Xenon Papanikolaou of the University of Arkansas and colleagues wrote in the journal Leukemia (2015;29:1713-20).
The investigators performed DNA/CIG as part of the routine work-up of patients with newly diagnosed MM who were scheduled to undergo the current iteration of the Total Therapy protocol, consisting of two induction chemotherapy cycles of VTD-PACE (bortezomib, thalidomide, dexamethasone and 4-day continuous infusions of cisplatin, doxorubicin, cyclophosphamide and etoposide), two planned hematopoietic stem cell transplants, and 3 years of planned maintenance with bortezomib, lenalidomide and dexamethasone.
The authors looked at the association of the CIg index, gene-expression profiling, and standard prognostic measures with both overall and progression-free survival (OS and PFS).
They found that both the percentage of light chain–restricted cells and the CIg as determined by DNA/CIG were associated with poor clinical outcomes. Additionally, a CIg lower than 2.8, albumin lower than 3.5 g/dL, and age 65 and older were significantly associated with worse PFS and OS, independent of gene-expression profiling data.
Any CIg lower than 2.8 was associated with a twofold risk for worse OS (hazard ratio, 2.08; P = .012) and a nearly doubling of risk for worse PFS (HR, 1.84; P = .001).
In multivariate models, CIg remained an independent predictor for prognosis when GEP-defined high-risk and low albumin were added in. CIg was also linked to other prognostic factors, including beta2-microglobulin greater than 5.5 mg/L, percentage of LCR cells exceeding 50%, C-reactive protein equal to or greater than 8 mg/L, and GEP-derived high centrosome index.
Low CIg was also associated with an experimental group of 12 gene probes believed to be involved in cell cycle regulation, cell differentiation, and cellular drug transport. The investigators used the gene signatures to develop a risk score, called GEP12, that offered important prognostic information for patients in TT3b and in earlier clinical studies.
The finding that a low CIg index was linked to a high centrosome index as defined by GEP points to a possible role for CIg index as a treatment selection tool.
“Interestingly, a centrosome inhibitor has shown promising activity in preclinical models of MM, thus potentially providing a selective drug for patients with low CIg myeloma,” the authors write.
The study was supported in part by a grant from the National Cancer Institute. Senior author Dr. Bart Barlogies reports research funding from Celgene Corp and Millennium Pharmaceuticals, and consulting with those companies as well as Onyx Pharmaceuticals and Amgen. He is a coinventor on patents and patent applications related to use of gene expression profiling in cancer medicine that have been licensed to Myeloma Health, but has no financial interests in this company. The remaining authors reported having no conflicts of interest.
A rapid, low-cost assay measuring DNA and immunoglobulin in multiple myeloma cells is both a powerful predictor of prognosis and a tool for identifying new, previously unidentified therapeutic targets, its developers say.
The test, labeled DNA/CIG, uses two-color flow cytometry to evaluate DNA in the nucleus and immunoglobulin in the cytoplasm of bone marrow taken from patients with multiple myeloma (MM) at baseline, before the start of the aggressive “Total Therapy 3b” (TT3b) protocol developed at the Myeloma Institute for Research and Therapy at the University of Arkansas for Medical Sciences in Little Rock.
The light chain cytoplasmic immunoglobulin (CIg) index generated by the test, combined with an associated 12-gene panel, serves as an independent prognostic factor in patients with myeloma. The CIg index is a marker for immunoglobulin production in myeloma cells, with lower index scores suggesting ineffective immunologic responses.
“We show that the presence of low CIg as detected by DNA/CIG is a major adverse prognostic factor in TT3b, even when other GEP [gene expression profiling]–derived prognostic factors were accounted for,” Dr. Xenon Papanikolaou of the University of Arkansas and colleagues wrote in the journal Leukemia (2015;29:1713-20).
The investigators performed DNA/CIG as part of the routine work-up of patients with newly diagnosed MM who were scheduled to undergo the current iteration of the Total Therapy protocol, consisting of two induction chemotherapy cycles of VTD-PACE (bortezomib, thalidomide, dexamethasone and 4-day continuous infusions of cisplatin, doxorubicin, cyclophosphamide and etoposide), two planned hematopoietic stem cell transplants, and 3 years of planned maintenance with bortezomib, lenalidomide and dexamethasone.
The authors looked at the association of the CIg index, gene-expression profiling, and standard prognostic measures with both overall and progression-free survival (OS and PFS).
They found that both the percentage of light chain–restricted cells and the CIg as determined by DNA/CIG were associated with poor clinical outcomes. Additionally, a CIg lower than 2.8, albumin lower than 3.5 g/dL, and age 65 and older were significantly associated with worse PFS and OS, independent of gene-expression profiling data.
Any CIg lower than 2.8 was associated with a twofold risk for worse OS (hazard ratio, 2.08; P = .012) and a nearly doubling of risk for worse PFS (HR, 1.84; P = .001).
In multivariate models, CIg remained an independent predictor for prognosis when GEP-defined high-risk and low albumin were added in. CIg was also linked to other prognostic factors, including beta2-microglobulin greater than 5.5 mg/L, percentage of LCR cells exceeding 50%, C-reactive protein equal to or greater than 8 mg/L, and GEP-derived high centrosome index.
Low CIg was also associated with an experimental group of 12 gene probes believed to be involved in cell cycle regulation, cell differentiation, and cellular drug transport. The investigators used the gene signatures to develop a risk score, called GEP12, that offered important prognostic information for patients in TT3b and in earlier clinical studies.
The finding that a low CIg index was linked to a high centrosome index as defined by GEP points to a possible role for CIg index as a treatment selection tool.
“Interestingly, a centrosome inhibitor has shown promising activity in preclinical models of MM, thus potentially providing a selective drug for patients with low CIg myeloma,” the authors write.
The study was supported in part by a grant from the National Cancer Institute. Senior author Dr. Bart Barlogies reports research funding from Celgene Corp and Millennium Pharmaceuticals, and consulting with those companies as well as Onyx Pharmaceuticals and Amgen. He is a coinventor on patents and patent applications related to use of gene expression profiling in cancer medicine that have been licensed to Myeloma Health, but has no financial interests in this company. The remaining authors reported having no conflicts of interest.
FROM LEUKEMIA
Key clinical point: Cytoplasmic immunoglobulin index (CIg) is prognostic of clinical outcomes in patients with multiple myeloma.
Major finding: A CIg lower than 2.8 was prognostic for poor overall and progression-free survival, with hazard ratios of 2.08 and 1.84, respectively.
Data source: Prospective study in 177 patients with newly diagnosed multiple myeloma.
Disclosures: The study was supported in part by a grant from the National Cancer Institute. Senior author Dr. Bart Barlogies reports research funding from Celgene Corp and Millennium Pharmaceuticals, and consulting with those companies as well as Onyx Pharmaceuticals and Amgen. He is a coinventor on patents and patent applications related to use of gene expression profiling in cancer medicine that have been licensed to Myeloma Health, but has no financial interests in this company. The remaining authors reported having no conflicts of interest.
How a cancer diagnosis affects income

receiving treatment
Photo by Rhoda Baer
A new study indicates that when American adults are diagnosed with
cancer, they experience significant decreases in the probability of
working, in the number of hours they work, and correspondingly, in their
incomes.
Additionally, these effects appear to be more pronounced among men than women.
Anna Zajacova, PhD, of the University of Wyoming in Laramie, and her colleagues reported these findings in Cancer.
The researchers analyzed data from the Panel Study of Income Dynamics, a nationally representative, prospective, population-based, observational study with individual and family level economic information. The data spanned the period from 1999 to 2009.
The team used models to estimate the impact of cancer on employment, hours worked, individual income, and total family income.
The results showed that, after a cancer diagnosis, the probability of a patient being employed dropped by almost 10 percentage points, and hours worked declined by up to 200 hours, or about 5 weeks of full-time work, in the first year.
Annual labor market earnings dropped almost 40% within 2 years of diagnosis, and they remained lower than before diagnosis. Total family income declined by 20%, although it recovered within 4 years of cancer diagnosis.
These effects were primarily driven by losses among male cancer survivors. For women diagnosed with cancer, the losses were largely not statistically significant.
“Fifteen million American adults are cancer survivors, and American families need economic support while they are dealing with the rigors of cancer treatment,” Dr Zajacova said.
“Our paper suggests that families where an adult—especially a working-age male—is diagnosed with cancer suffer short-term and long-term declines in their economic well-being. We need to improve workplace and insurance safety nets so families can focus on dealing with the cancer treatment rather than deal with the financial and employment fallout.” ![]()

receiving treatment
Photo by Rhoda Baer
A new study indicates that when American adults are diagnosed with
cancer, they experience significant decreases in the probability of
working, in the number of hours they work, and correspondingly, in their
incomes.
Additionally, these effects appear to be more pronounced among men than women.
Anna Zajacova, PhD, of the University of Wyoming in Laramie, and her colleagues reported these findings in Cancer.
The researchers analyzed data from the Panel Study of Income Dynamics, a nationally representative, prospective, population-based, observational study with individual and family level economic information. The data spanned the period from 1999 to 2009.
The team used models to estimate the impact of cancer on employment, hours worked, individual income, and total family income.
The results showed that, after a cancer diagnosis, the probability of a patient being employed dropped by almost 10 percentage points, and hours worked declined by up to 200 hours, or about 5 weeks of full-time work, in the first year.
Annual labor market earnings dropped almost 40% within 2 years of diagnosis, and they remained lower than before diagnosis. Total family income declined by 20%, although it recovered within 4 years of cancer diagnosis.
These effects were primarily driven by losses among male cancer survivors. For women diagnosed with cancer, the losses were largely not statistically significant.
“Fifteen million American adults are cancer survivors, and American families need economic support while they are dealing with the rigors of cancer treatment,” Dr Zajacova said.
“Our paper suggests that families where an adult—especially a working-age male—is diagnosed with cancer suffer short-term and long-term declines in their economic well-being. We need to improve workplace and insurance safety nets so families can focus on dealing with the cancer treatment rather than deal with the financial and employment fallout.” ![]()

receiving treatment
Photo by Rhoda Baer
A new study indicates that when American adults are diagnosed with
cancer, they experience significant decreases in the probability of
working, in the number of hours they work, and correspondingly, in their
incomes.
Additionally, these effects appear to be more pronounced among men than women.
Anna Zajacova, PhD, of the University of Wyoming in Laramie, and her colleagues reported these findings in Cancer.
The researchers analyzed data from the Panel Study of Income Dynamics, a nationally representative, prospective, population-based, observational study with individual and family level economic information. The data spanned the period from 1999 to 2009.
The team used models to estimate the impact of cancer on employment, hours worked, individual income, and total family income.
The results showed that, after a cancer diagnosis, the probability of a patient being employed dropped by almost 10 percentage points, and hours worked declined by up to 200 hours, or about 5 weeks of full-time work, in the first year.
Annual labor market earnings dropped almost 40% within 2 years of diagnosis, and they remained lower than before diagnosis. Total family income declined by 20%, although it recovered within 4 years of cancer diagnosis.
These effects were primarily driven by losses among male cancer survivors. For women diagnosed with cancer, the losses were largely not statistically significant.
“Fifteen million American adults are cancer survivors, and American families need economic support while they are dealing with the rigors of cancer treatment,” Dr Zajacova said.
“Our paper suggests that families where an adult—especially a working-age male—is diagnosed with cancer suffer short-term and long-term declines in their economic well-being. We need to improve workplace and insurance safety nets so families can focus on dealing with the cancer treatment rather than deal with the financial and employment fallout.” ![]()
Radiation often underused in follicular lymphoma

woman for radiotherapy
Photo by Rhoda Baer
SAN ANTONIO—A new study indicates that patients with early stage follicular lymphoma (FL) are increasingly receiving no treatment or single-agent chemotherapy, despite evidence suggesting that radiation therapy can produce better outcomes.
Guidelines from the National Comprehensive Cancer Network and the European Society for Medical Oncology both list radiation therapy as the preferred treatment for low-grade FL.
However, investigators found that, in recent years, radiation has been replaced by alternative strategies.
“Our study highlights the increasing omission of radiation therapy in [FL] and its associated negative effect on overall survival at a national level,” said John Austin Vargo, MD, of the University of Pittsburg Cancer Institute in Pennsylvania.
“This increasing bias towards the omission of radiation therapy is despite proven efficacy and increasing adoption of lower radiation therapy doses and more modern radiation therapy techniques which decrease risk of side effects.”
Dr Vargo presented these findings at the 57th Annual Meeting of the American Society for Radiation Oncology (presentation #183).
He and his colleagues analyzed patterns of care and survival outcomes for 35,961 patients diagnosed with early stage FL as listed in the National Cancer Data Base. A majority of patients were older than 60 (61%), and most had stage I disease (63%).
The use of radiation therapy in this group of patients decreased from 37% in 1999 to 24% in 2012 (P<0.0001).
The use of observation increased from 34% in 1998 to 44% in 2012 (P<0.0001). And the use of single-agent chemotherapy increased from 5.4% in 1999 to 11.7% in 2006 (P=0.01).
The 5-year overall survival rate was 86% in patients who received radiation and 74% in those who did not (P<0.0001). Ten-year overall survival rates were 68% and 54%, respectively (P<0.0001).
In multivariate analysis, radiation therapy remained significantly associated with improved overall survival (P<0.0001). ![]()

woman for radiotherapy
Photo by Rhoda Baer
SAN ANTONIO—A new study indicates that patients with early stage follicular lymphoma (FL) are increasingly receiving no treatment or single-agent chemotherapy, despite evidence suggesting that radiation therapy can produce better outcomes.
Guidelines from the National Comprehensive Cancer Network and the European Society for Medical Oncology both list radiation therapy as the preferred treatment for low-grade FL.
However, investigators found that, in recent years, radiation has been replaced by alternative strategies.
“Our study highlights the increasing omission of radiation therapy in [FL] and its associated negative effect on overall survival at a national level,” said John Austin Vargo, MD, of the University of Pittsburg Cancer Institute in Pennsylvania.
“This increasing bias towards the omission of radiation therapy is despite proven efficacy and increasing adoption of lower radiation therapy doses and more modern radiation therapy techniques which decrease risk of side effects.”
Dr Vargo presented these findings at the 57th Annual Meeting of the American Society for Radiation Oncology (presentation #183).
He and his colleagues analyzed patterns of care and survival outcomes for 35,961 patients diagnosed with early stage FL as listed in the National Cancer Data Base. A majority of patients were older than 60 (61%), and most had stage I disease (63%).
The use of radiation therapy in this group of patients decreased from 37% in 1999 to 24% in 2012 (P<0.0001).
The use of observation increased from 34% in 1998 to 44% in 2012 (P<0.0001). And the use of single-agent chemotherapy increased from 5.4% in 1999 to 11.7% in 2006 (P=0.01).
The 5-year overall survival rate was 86% in patients who received radiation and 74% in those who did not (P<0.0001). Ten-year overall survival rates were 68% and 54%, respectively (P<0.0001).
In multivariate analysis, radiation therapy remained significantly associated with improved overall survival (P<0.0001). ![]()

woman for radiotherapy
Photo by Rhoda Baer
SAN ANTONIO—A new study indicates that patients with early stage follicular lymphoma (FL) are increasingly receiving no treatment or single-agent chemotherapy, despite evidence suggesting that radiation therapy can produce better outcomes.
Guidelines from the National Comprehensive Cancer Network and the European Society for Medical Oncology both list radiation therapy as the preferred treatment for low-grade FL.
However, investigators found that, in recent years, radiation has been replaced by alternative strategies.
“Our study highlights the increasing omission of radiation therapy in [FL] and its associated negative effect on overall survival at a national level,” said John Austin Vargo, MD, of the University of Pittsburg Cancer Institute in Pennsylvania.
“This increasing bias towards the omission of radiation therapy is despite proven efficacy and increasing adoption of lower radiation therapy doses and more modern radiation therapy techniques which decrease risk of side effects.”
Dr Vargo presented these findings at the 57th Annual Meeting of the American Society for Radiation Oncology (presentation #183).
He and his colleagues analyzed patterns of care and survival outcomes for 35,961 patients diagnosed with early stage FL as listed in the National Cancer Data Base. A majority of patients were older than 60 (61%), and most had stage I disease (63%).
The use of radiation therapy in this group of patients decreased from 37% in 1999 to 24% in 2012 (P<0.0001).
The use of observation increased from 34% in 1998 to 44% in 2012 (P<0.0001). And the use of single-agent chemotherapy increased from 5.4% in 1999 to 11.7% in 2006 (P=0.01).
The 5-year overall survival rate was 86% in patients who received radiation and 74% in those who did not (P<0.0001). Ten-year overall survival rates were 68% and 54%, respectively (P<0.0001).
In multivariate analysis, radiation therapy remained significantly associated with improved overall survival (P<0.0001).
Radioimmunotherapy shows promise for HL

Copyright 2010 Nephron
Results of a small trial suggest the radiolabeled anti-CD25 antibody 90Y-daclizumab can treat certain patients with relapsed/refractory Hodgkin lymphoma (HL).
90Y-daclizumab produced responses in 50% of patients studied, and most of these were complete responses.
Several patients developed myelodysplastic syndrome (MDS) after receiving 90Y-daclizumab, but researchers found they could prevent this side effect with pretreatment screening.
John Janik, MD, of the National Cancer Institute in Bethesda, Maryland, and his colleagues described this research in PNAS.
The researchers conducted this study to determine if CD25 is a favorable target for systemic radioimmunotherapy in HL. The team noted that although most normal cells don’t express CD25, it is expressed by a minority of Reed–Sternberg cells and by most polyclonal T cells rosetting around Reed–Sternberg cells.
So the researchers tested the anti-CD25 antibody 90Y-daclizumab in 46 patients with relapsed and refractory HL. Patients received 90Y-daclizumab at 10 mCi to 15 mCi every 6 to 10 weeks for up to 7 doses.
The overall response rate was 50%. There were 14 complete responses and 9 partial responses. Fourteen patients had stable disease, and 9 progressed.
The researchers noted that responses occurred in patients whose Reed–Sternberg cells expressed CD25 and in those whose neoplastic cells were CD25-negative, as long as associated rosetting T cells expressed CD25.
Grade 3 or higher toxicities observed in this study included lymphopenia (n=10), neutropenia (n=7), MDS (n=6), thrombocytopenia (n=5), anemia (n=5), and pneumonia (n=2).
At a median follow-up of 9 years, MDS had occurred in 6 patients. They had received a mean of 3.2 doses of 90Y-daclizumab. Prior to 90Y-daclizumab, they had received a mean of 6.2 courses of chemotherapy.
The median time from the initiation of 90Y-daclizumab to MDS diagnosis was 37 months. A retrospective review of 1 patient with MDS revealed that the patient had cytogenetic aberrations on chromosomes 5 and 7 after receiving chemotherapy but before starting 90Y-daclizumab.
So the researchers changed the trial’s entry criteria to require a bone marrow karyotype analysis, and patients who had aberrations were excluded. None of the 16 patients who entered the trial after this change developed MDS.
The researchers said this study is too small to draw meaningful conclusions about the relative roles of chemotherapy and 90Y-daclizumab in the pathogenesis of MDS.
However, this complication is a serious concern, and any subsequent trial should require cytogenetic studies of bone marrow specimens before patients receive systemic radioimmunotherapy.

Copyright 2010 Nephron
Results of a small trial suggest the radiolabeled anti-CD25 antibody 90Y-daclizumab can treat certain patients with relapsed/refractory Hodgkin lymphoma (HL).
90Y-daclizumab produced responses in 50% of patients studied, and most of these were complete responses.
Several patients developed myelodysplastic syndrome (MDS) after receiving 90Y-daclizumab, but researchers found they could prevent this side effect with pretreatment screening.
John Janik, MD, of the National Cancer Institute in Bethesda, Maryland, and his colleagues described this research in PNAS.
The researchers conducted this study to determine if CD25 is a favorable target for systemic radioimmunotherapy in HL. The team noted that although most normal cells don’t express CD25, it is expressed by a minority of Reed–Sternberg cells and by most polyclonal T cells rosetting around Reed–Sternberg cells.
So the researchers tested the anti-CD25 antibody 90Y-daclizumab in 46 patients with relapsed and refractory HL. Patients received 90Y-daclizumab at 10 mCi to 15 mCi every 6 to 10 weeks for up to 7 doses.
The overall response rate was 50%. There were 14 complete responses and 9 partial responses. Fourteen patients had stable disease, and 9 progressed.
The researchers noted that responses occurred in patients whose Reed–Sternberg cells expressed CD25 and in those whose neoplastic cells were CD25-negative, as long as associated rosetting T cells expressed CD25.
Grade 3 or higher toxicities observed in this study included lymphopenia (n=10), neutropenia (n=7), MDS (n=6), thrombocytopenia (n=5), anemia (n=5), and pneumonia (n=2).
At a median follow-up of 9 years, MDS had occurred in 6 patients. They had received a mean of 3.2 doses of 90Y-daclizumab. Prior to 90Y-daclizumab, they had received a mean of 6.2 courses of chemotherapy.
The median time from the initiation of 90Y-daclizumab to MDS diagnosis was 37 months. A retrospective review of 1 patient with MDS revealed that the patient had cytogenetic aberrations on chromosomes 5 and 7 after receiving chemotherapy but before starting 90Y-daclizumab.
So the researchers changed the trial’s entry criteria to require a bone marrow karyotype analysis, and patients who had aberrations were excluded. None of the 16 patients who entered the trial after this change developed MDS.
The researchers said this study is too small to draw meaningful conclusions about the relative roles of chemotherapy and 90Y-daclizumab in the pathogenesis of MDS.
However, this complication is a serious concern, and any subsequent trial should require cytogenetic studies of bone marrow specimens before patients receive systemic radioimmunotherapy.

Copyright 2010 Nephron
Results of a small trial suggest the radiolabeled anti-CD25 antibody 90Y-daclizumab can treat certain patients with relapsed/refractory Hodgkin lymphoma (HL).
90Y-daclizumab produced responses in 50% of patients studied, and most of these were complete responses.
Several patients developed myelodysplastic syndrome (MDS) after receiving 90Y-daclizumab, but researchers found they could prevent this side effect with pretreatment screening.
John Janik, MD, of the National Cancer Institute in Bethesda, Maryland, and his colleagues described this research in PNAS.
The researchers conducted this study to determine if CD25 is a favorable target for systemic radioimmunotherapy in HL. The team noted that although most normal cells don’t express CD25, it is expressed by a minority of Reed–Sternberg cells and by most polyclonal T cells rosetting around Reed–Sternberg cells.
So the researchers tested the anti-CD25 antibody 90Y-daclizumab in 46 patients with relapsed and refractory HL. Patients received 90Y-daclizumab at 10 mCi to 15 mCi every 6 to 10 weeks for up to 7 doses.
The overall response rate was 50%. There were 14 complete responses and 9 partial responses. Fourteen patients had stable disease, and 9 progressed.
The researchers noted that responses occurred in patients whose Reed–Sternberg cells expressed CD25 and in those whose neoplastic cells were CD25-negative, as long as associated rosetting T cells expressed CD25.
Grade 3 or higher toxicities observed in this study included lymphopenia (n=10), neutropenia (n=7), MDS (n=6), thrombocytopenia (n=5), anemia (n=5), and pneumonia (n=2).
At a median follow-up of 9 years, MDS had occurred in 6 patients. They had received a mean of 3.2 doses of 90Y-daclizumab. Prior to 90Y-daclizumab, they had received a mean of 6.2 courses of chemotherapy.
The median time from the initiation of 90Y-daclizumab to MDS diagnosis was 37 months. A retrospective review of 1 patient with MDS revealed that the patient had cytogenetic aberrations on chromosomes 5 and 7 after receiving chemotherapy but before starting 90Y-daclizumab.
So the researchers changed the trial’s entry criteria to require a bone marrow karyotype analysis, and patients who had aberrations were excluded. None of the 16 patients who entered the trial after this change developed MDS.
The researchers said this study is too small to draw meaningful conclusions about the relative roles of chemotherapy and 90Y-daclizumab in the pathogenesis of MDS.
However, this complication is a serious concern, and any subsequent trial should require cytogenetic studies of bone marrow specimens before patients receive systemic radioimmunotherapy.
HSC finding may have range of implications
Image by Matthias Zepper
Murine research has provided new insight into the functionality of hematopoietic stem cells (HSCs).
Investigators found that a full complement of mini-chromosome maintenance (MCM) proteins is required to preserve HSC functionality and the proper differentiation and maturation of erythrocytes.
Downregulation of the gene MCM3 during embryonic development caused replication stress in hematopoietic progenitors and led to fetal anemia.
In adult mice, downregulation of MCM3 reduced life expectancy and promoted lymphomagenesis.
The investigators therefore believe that therapies designed to modulate replication stress could fight aging, anemia, and hematopoietic malignancies.
This research was published in Nature Communications.
A previous study revealed that replication stress drives the functional decline of HSCs that occurs with age. With the new study, investigators have managed to replicate this phenomenon in mouse embryos.
The team did this by reducing levels of MCM3, one of the components of the MCM complex that is responsible for separating the strands of the DNA double helix during replication. Cells need to maintain high levels of MCM during DNA replication or they experience replication stress.
“When we reduce the levels of the MCM3 gene in the entire organism, we observe that replication stress especially affects the stem cells that give rise to the other blood cells and, in particular, the red blood cell precursors,” explained study author Juan Méndez, PhD, of Centro Nacional de Investigaciones Oncologicas (CNIO) in Madrid, Spain.
“In adult organisms, the production and maturation of red blood cells takes place in the bone marrow, but, during embryonic development, it occurs mainly in the fetal liver. In animals with MCM3 deficiency, the stem cells of the fetal liver are deteriorated, and the embryos develop a severe form of anemia that prevents them from being born.”
“We could say that replication stress turns fetal stem cells, which should be in perfect working order, into very old cells. We have verified this finding in transplantation experiments, where fetal cells with replication stress were unable to adequately reconstitute the blood system in the recipient animals.”
However, the investigators managed to prevent embryonic lethality and reduce anemia by increasing the levels of another gene, CHK1.
“CHK1 is one of the genes responsible for protecting cells from replication stress,” Dr Méndez noted. “It supervises DNA replication. When something goes wrong, CHK1 slows down or halts cell division until the problem has been resolved.”
Embryos that were subjected to replication stress (due to loss of MCM3) but had higher levels of CHK1 showed less pronounced anemia. Four of every 10 embryos developed normally and completed their gestation.
The investigators said an interesting implication of this work is that the type of anemia caused by replication stress is very similar to the aplastic anemia that arises in patients receiving chemotherapy or radiation therapy.
Therefore, the team believes these results could aid the development of novel therapies for aplastic anemia.

Image by Matthias Zepper
Murine research has provided new insight into the functionality of hematopoietic stem cells (HSCs).
Investigators found that a full complement of mini-chromosome maintenance (MCM) proteins is required to preserve HSC functionality and the proper differentiation and maturation of erythrocytes.
Downregulation of the gene MCM3 during embryonic development caused replication stress in hematopoietic progenitors and led to fetal anemia.
In adult mice, downregulation of MCM3 reduced life expectancy and promoted lymphomagenesis.
The investigators therefore believe that therapies designed to modulate replication stress could fight aging, anemia, and hematopoietic malignancies.
This research was published in Nature Communications.
A previous study revealed that replication stress drives the functional decline of HSCs that occurs with age. With the new study, investigators have managed to replicate this phenomenon in mouse embryos.
The team did this by reducing levels of MCM3, one of the components of the MCM complex that is responsible for separating the strands of the DNA double helix during replication. Cells need to maintain high levels of MCM during DNA replication or they experience replication stress.
“When we reduce the levels of the MCM3 gene in the entire organism, we observe that replication stress especially affects the stem cells that give rise to the other blood cells and, in particular, the red blood cell precursors,” explained study author Juan Méndez, PhD, of Centro Nacional de Investigaciones Oncologicas (CNIO) in Madrid, Spain.
“In adult organisms, the production and maturation of red blood cells takes place in the bone marrow, but, during embryonic development, it occurs mainly in the fetal liver. In animals with MCM3 deficiency, the stem cells of the fetal liver are deteriorated, and the embryos develop a severe form of anemia that prevents them from being born.”
“We could say that replication stress turns fetal stem cells, which should be in perfect working order, into very old cells. We have verified this finding in transplantation experiments, where fetal cells with replication stress were unable to adequately reconstitute the blood system in the recipient animals.”
However, the investigators managed to prevent embryonic lethality and reduce anemia by increasing the levels of another gene, CHK1.
“CHK1 is one of the genes responsible for protecting cells from replication stress,” Dr Méndez noted. “It supervises DNA replication. When something goes wrong, CHK1 slows down or halts cell division until the problem has been resolved.”
Embryos that were subjected to replication stress (due to loss of MCM3) but had higher levels of CHK1 showed less pronounced anemia. Four of every 10 embryos developed normally and completed their gestation.
The investigators said an interesting implication of this work is that the type of anemia caused by replication stress is very similar to the aplastic anemia that arises in patients receiving chemotherapy or radiation therapy.
Therefore, the team believes these results could aid the development of novel therapies for aplastic anemia.

Image by Matthias Zepper
Murine research has provided new insight into the functionality of hematopoietic stem cells (HSCs).
Investigators found that a full complement of mini-chromosome maintenance (MCM) proteins is required to preserve HSC functionality and the proper differentiation and maturation of erythrocytes.
Downregulation of the gene MCM3 during embryonic development caused replication stress in hematopoietic progenitors and led to fetal anemia.
In adult mice, downregulation of MCM3 reduced life expectancy and promoted lymphomagenesis.
The investigators therefore believe that therapies designed to modulate replication stress could fight aging, anemia, and hematopoietic malignancies.
This research was published in Nature Communications.
A previous study revealed that replication stress drives the functional decline of HSCs that occurs with age. With the new study, investigators have managed to replicate this phenomenon in mouse embryos.
The team did this by reducing levels of MCM3, one of the components of the MCM complex that is responsible for separating the strands of the DNA double helix during replication. Cells need to maintain high levels of MCM during DNA replication or they experience replication stress.
“When we reduce the levels of the MCM3 gene in the entire organism, we observe that replication stress especially affects the stem cells that give rise to the other blood cells and, in particular, the red blood cell precursors,” explained study author Juan Méndez, PhD, of Centro Nacional de Investigaciones Oncologicas (CNIO) in Madrid, Spain.
“In adult organisms, the production and maturation of red blood cells takes place in the bone marrow, but, during embryonic development, it occurs mainly in the fetal liver. In animals with MCM3 deficiency, the stem cells of the fetal liver are deteriorated, and the embryos develop a severe form of anemia that prevents them from being born.”
“We could say that replication stress turns fetal stem cells, which should be in perfect working order, into very old cells. We have verified this finding in transplantation experiments, where fetal cells with replication stress were unable to adequately reconstitute the blood system in the recipient animals.”
However, the investigators managed to prevent embryonic lethality and reduce anemia by increasing the levels of another gene, CHK1.
“CHK1 is one of the genes responsible for protecting cells from replication stress,” Dr Méndez noted. “It supervises DNA replication. When something goes wrong, CHK1 slows down or halts cell division until the problem has been resolved.”
Embryos that were subjected to replication stress (due to loss of MCM3) but had higher levels of CHK1 showed less pronounced anemia. Four of every 10 embryos developed normally and completed their gestation.
The investigators said an interesting implication of this work is that the type of anemia caused by replication stress is very similar to the aplastic anemia that arises in patients receiving chemotherapy or radiation therapy.
Therefore, the team believes these results could aid the development of novel therapies for aplastic anemia.