User login
VIDEO: Targeted agent, len/dex combo perform ‘impressively’ in advanced multiple myeloma
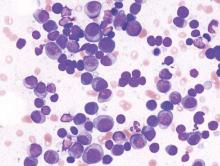
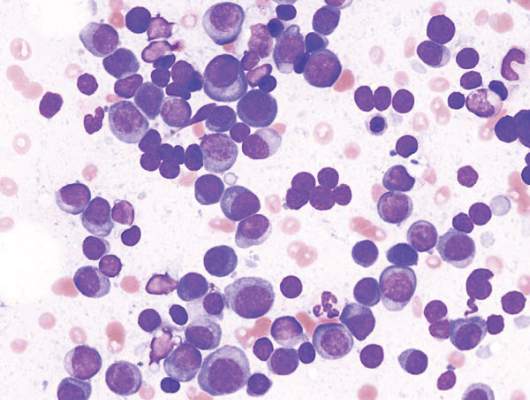
ORLANDO – A combination of daratumumab (Darzalex) with lenalidomide and dexamethasone (len/dex) was associated with a high overall response rate, including stringent complete responses, in patients with heavily pretreated relapsed/refractory multiple myeloma.
Daratumumab was recently approved by the Food and Drug Administration as monotherapy in patients with relapsed/refractory multiple myeloma.
Dr. Torben Plesner of Vejle Hospital and the University of Southern Denmark in Vejle, who dosed the first patient with daratumumab in 2007, discusses updated findings from a phase I/II trial of the daratumumab and len/dex combination.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ORLANDO – A combination of daratumumab (Darzalex) with lenalidomide and dexamethasone (len/dex) was associated with a high overall response rate, including stringent complete responses, in patients with heavily pretreated relapsed/refractory multiple myeloma.
Daratumumab was recently approved by the Food and Drug Administration as monotherapy in patients with relapsed/refractory multiple myeloma.
Dr. Torben Plesner of Vejle Hospital and the University of Southern Denmark in Vejle, who dosed the first patient with daratumumab in 2007, discusses updated findings from a phase I/II trial of the daratumumab and len/dex combination.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ORLANDO – A combination of daratumumab (Darzalex) with lenalidomide and dexamethasone (len/dex) was associated with a high overall response rate, including stringent complete responses, in patients with heavily pretreated relapsed/refractory multiple myeloma.
Daratumumab was recently approved by the Food and Drug Administration as monotherapy in patients with relapsed/refractory multiple myeloma.
Dr. Torben Plesner of Vejle Hospital and the University of Southern Denmark in Vejle, who dosed the first patient with daratumumab in 2007, discusses updated findings from a phase I/II trial of the daratumumab and len/dex combination.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT ASH 2015
Cancer drug prices vary widely from country to country

Photo by Bill Branson
The price of cancer drugs varies widely between European countries, Australia, and New Zealand, according to a study published in The Lancet Oncology.
The study indicates that, overall, the UK and Mediterranean countries such as Greece, Spain, and Portugal pay the lowest average unit manufacturer prices for a group of 31 originator cancer drugs (new drugs under patent).
And Sweden, Switzerland, and Germany pay the highest prices.
The greatest differences in price were noted for gemcitabine, which costs €209 per vial in New Zealand and €43 in Australia, and zoledronic acid, which costs €330 per vial in New Zealand but €128 in Greece.*
“Public payers in Germany are paying 223% more in terms of official prices for interferon alfa 2b for melanoma and leukemia treatment than those in Greece,” noted study author Sabine Vogler, PhD, of the WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies in Vienna, Austria.
“For gefitinib to treat non-small-lung cancer, the price in Germany is 172% higher than in New Zealand.”
To uncover these price differences, Dr Vogler and her colleagues reviewed official drug price data from the Pharma Price Information (PPI) service of the Austrian Public Health Institute for 16 European countries**, and from the pharmaceutical schedules in Australia and New Zealand.
The researchers compared what manufacturers charged for a unit (ie, price per tablet or vial) of 31 originator cancer drugs in June 2013.
None of these drugs had a unit price lower than €10. Four drugs (13%) had an average unit manufacturer price between €250 and €500, and 2 drugs (6%) had an average unit price between €500 and €1000.
Seven drugs (23%) had an average unit price higher than €1000. For example, plerixafor cost over €5000 per injection.
The price differences between the highest- and lowest-priced countries ranged from 28% to 50% for a third of the drugs sampled, between 50% and 100% for half of the drugs, and between 100% and 200% for 3 drugs (10%).
The researchers noted that information on real drug prices is scarce. The cancer drug prices they surveyed did not include confidential discounts such as those agreed upon in managed-entry arrangements that are increasingly used in countries such as Australia, Italy, the UK, and the Netherlands.
“Some high-income countries have managed to barter the manufacturers down to lower prices, but these agreements, including the agreed prices, are confidential,” Dr Vogler explained.
“Although these agreements ensure patient access to new drugs, other countries risk overpaying when setting drug prices through the common practice of external price referencing, or international price comparison, because they can only use the official undiscounted prices as a benchmark. There needs to be far more transparency.”
“We hope that our findings will provide concrete evidence for policymakers to take action to address high prices and ensure more transparency in cancer drug pricing so that costs and access to new drugs does not depend on where a patient lives.” ![]()
*Gemcitabine and zoledronic acid have generic versions in several countries, and originator prices were decreased in some countries following patent expiry but not in others.
**Austria, Belgium, Denmark, Germany, Greece, Finland, France, Italy, Ireland, the Netherlands, Norway, Portugal, Spain, Sweden, Switzerland, and the UK.

Photo by Bill Branson
The price of cancer drugs varies widely between European countries, Australia, and New Zealand, according to a study published in The Lancet Oncology.
The study indicates that, overall, the UK and Mediterranean countries such as Greece, Spain, and Portugal pay the lowest average unit manufacturer prices for a group of 31 originator cancer drugs (new drugs under patent).
And Sweden, Switzerland, and Germany pay the highest prices.
The greatest differences in price were noted for gemcitabine, which costs €209 per vial in New Zealand and €43 in Australia, and zoledronic acid, which costs €330 per vial in New Zealand but €128 in Greece.*
“Public payers in Germany are paying 223% more in terms of official prices for interferon alfa 2b for melanoma and leukemia treatment than those in Greece,” noted study author Sabine Vogler, PhD, of the WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies in Vienna, Austria.
“For gefitinib to treat non-small-lung cancer, the price in Germany is 172% higher than in New Zealand.”
To uncover these price differences, Dr Vogler and her colleagues reviewed official drug price data from the Pharma Price Information (PPI) service of the Austrian Public Health Institute for 16 European countries**, and from the pharmaceutical schedules in Australia and New Zealand.
The researchers compared what manufacturers charged for a unit (ie, price per tablet or vial) of 31 originator cancer drugs in June 2013.
None of these drugs had a unit price lower than €10. Four drugs (13%) had an average unit manufacturer price between €250 and €500, and 2 drugs (6%) had an average unit price between €500 and €1000.
Seven drugs (23%) had an average unit price higher than €1000. For example, plerixafor cost over €5000 per injection.
The price differences between the highest- and lowest-priced countries ranged from 28% to 50% for a third of the drugs sampled, between 50% and 100% for half of the drugs, and between 100% and 200% for 3 drugs (10%).
The researchers noted that information on real drug prices is scarce. The cancer drug prices they surveyed did not include confidential discounts such as those agreed upon in managed-entry arrangements that are increasingly used in countries such as Australia, Italy, the UK, and the Netherlands.
“Some high-income countries have managed to barter the manufacturers down to lower prices, but these agreements, including the agreed prices, are confidential,” Dr Vogler explained.
“Although these agreements ensure patient access to new drugs, other countries risk overpaying when setting drug prices through the common practice of external price referencing, or international price comparison, because they can only use the official undiscounted prices as a benchmark. There needs to be far more transparency.”
“We hope that our findings will provide concrete evidence for policymakers to take action to address high prices and ensure more transparency in cancer drug pricing so that costs and access to new drugs does not depend on where a patient lives.” ![]()
*Gemcitabine and zoledronic acid have generic versions in several countries, and originator prices were decreased in some countries following patent expiry but not in others.
**Austria, Belgium, Denmark, Germany, Greece, Finland, France, Italy, Ireland, the Netherlands, Norway, Portugal, Spain, Sweden, Switzerland, and the UK.

Photo by Bill Branson
The price of cancer drugs varies widely between European countries, Australia, and New Zealand, according to a study published in The Lancet Oncology.
The study indicates that, overall, the UK and Mediterranean countries such as Greece, Spain, and Portugal pay the lowest average unit manufacturer prices for a group of 31 originator cancer drugs (new drugs under patent).
And Sweden, Switzerland, and Germany pay the highest prices.
The greatest differences in price were noted for gemcitabine, which costs €209 per vial in New Zealand and €43 in Australia, and zoledronic acid, which costs €330 per vial in New Zealand but €128 in Greece.*
“Public payers in Germany are paying 223% more in terms of official prices for interferon alfa 2b for melanoma and leukemia treatment than those in Greece,” noted study author Sabine Vogler, PhD, of the WHO Collaborating Centre for Pharmaceutical Pricing and Reimbursement Policies in Vienna, Austria.
“For gefitinib to treat non-small-lung cancer, the price in Germany is 172% higher than in New Zealand.”
To uncover these price differences, Dr Vogler and her colleagues reviewed official drug price data from the Pharma Price Information (PPI) service of the Austrian Public Health Institute for 16 European countries**, and from the pharmaceutical schedules in Australia and New Zealand.
The researchers compared what manufacturers charged for a unit (ie, price per tablet or vial) of 31 originator cancer drugs in June 2013.
None of these drugs had a unit price lower than €10. Four drugs (13%) had an average unit manufacturer price between €250 and €500, and 2 drugs (6%) had an average unit price between €500 and €1000.
Seven drugs (23%) had an average unit price higher than €1000. For example, plerixafor cost over €5000 per injection.
The price differences between the highest- and lowest-priced countries ranged from 28% to 50% for a third of the drugs sampled, between 50% and 100% for half of the drugs, and between 100% and 200% for 3 drugs (10%).
The researchers noted that information on real drug prices is scarce. The cancer drug prices they surveyed did not include confidential discounts such as those agreed upon in managed-entry arrangements that are increasingly used in countries such as Australia, Italy, the UK, and the Netherlands.
“Some high-income countries have managed to barter the manufacturers down to lower prices, but these agreements, including the agreed prices, are confidential,” Dr Vogler explained.
“Although these agreements ensure patient access to new drugs, other countries risk overpaying when setting drug prices through the common practice of external price referencing, or international price comparison, because they can only use the official undiscounted prices as a benchmark. There needs to be far more transparency.”
“We hope that our findings will provide concrete evidence for policymakers to take action to address high prices and ensure more transparency in cancer drug pricing so that costs and access to new drugs does not depend on where a patient lives.” ![]()
*Gemcitabine and zoledronic acid have generic versions in several countries, and originator prices were decreased in some countries following patent expiry but not in others.
**Austria, Belgium, Denmark, Germany, Greece, Finland, France, Italy, Ireland, the Netherlands, Norway, Portugal, Spain, Sweden, Switzerland, and the UK.
ASH: Gene therapy restores immune function to older children with SCID-X1
ORLANDO – – Gene therapy can help restore immune function to older children and young adults with X-linked severe combined immunodeficiency, a team of US investigators reports.
“This is the first demonstration of the use of gene therapy to salvage failed allogeneic hematopoietic stem cell transplants in older SCID-X1 patients,” said Dr. Suk See De Ravin from the Laboratory of Host Defenses at the National Institutes of Health in Bethesda, Maryland.
Although allogeneic hematopoietic stem cell transplantation (HSCT) from a matched sibling donor can be curative, transplants using parental bone marrow or genetically modified autologous transplants without myeloconditiong restore T-cell-mediated immunity but not humoral immunity, Dr. De Ravin said at the American Society of Hematology annual meeting.
Additionally, transplants in older children with SCID-X1 who have persistent immune system defects – despite having received a transplant from a haploidentical donor in infancy – leave the patients with serious medical problems, including the life-long need for immunoglobulin G (IgG) supplementation, recurrent and chronic infections, warts, malnutrition, growth failure, and progressive diseases of the gut and lung.
Previous attempts at using gene therapy to correct mutations in IL2RG, the cause of SCID-X1, used mouse retroviral vectors to insert normal IL2RG into autologous hematopoietic stem cells without chemotherapy conditioning. This treatment restored T-cell immunity, but not B-cell- or natural killer (NK)-cell immunity
Of even greater concern is the fact that among infants with SCID-X1 who received autologous stem cells transduced with murine gamma retrovirus carrying the common gamma chain, 25% developed vector-associated leukemia.
To overcome these problems, investigators in the current study used a lentiviral vector containing an insulator fragment from a chicken beta-globin gene. The insulator fragment allows expression of the gamma chain complementary DNA while protecting against up-regulation of neighboring oncogenes.
Dr. De Ravin reported data on five patients from the ages of 7 to 24 years who had worsening immune dysfunction and complex medical problems, including dependence on immunoglobulin G (IgG) supplementations. All of the patients had previously undergone one or more HSCT from haploidentical donors.
The patients were treated with granulocyte-colony stimulating factor and plerixafor to mobilize peripheral blood cells, and then underwent apheresis to isolate CD34 cells. The cells were transduced in vitro with a lentiviral vector, and reinfused into patients after conditioning with low-dose busulfan (6 mg/kg). The vector was developed by researchers at St Jude Children’s Hospital in Memphis, Tennessee.
At the most recent follow-up, the first two patients treated with the protocol had stable engraftment of gamma-chain expressing cells gene with enhanced expression of B, T, and NK cells, with the cells continuing to show improvements, Early data on the remaining four patients indicates a similar positive trend.
Chimerism studies of the patients’ T-cells showed that the host cells were continuing to increase their contribution, suggesting gradual replacement over time of the T-cell graft, Dr. De Ravin said.
In the second patient treated, an increase in NK cells corresponded with an improvement in chronic warts. In addition, the first two patients began to produce both IgG and antigen-specific responses to vaccination, clearance of chronic norovirus infections, and resolution of their protein-losing enteropathy.
“Gene therapy does not appear to reverse prior organ damage, supporting early intervention to improve immunity in these patients before such damage occurs,” Dr. De Ravin said.
The study was supported by the National Institutes of Health. The viral vector was developed at St. Jude Children’s Research Hospital in Memphis, Tennessee. The authors reported no relevant conflicts of interest.
ORLANDO – – Gene therapy can help restore immune function to older children and young adults with X-linked severe combined immunodeficiency, a team of US investigators reports.
“This is the first demonstration of the use of gene therapy to salvage failed allogeneic hematopoietic stem cell transplants in older SCID-X1 patients,” said Dr. Suk See De Ravin from the Laboratory of Host Defenses at the National Institutes of Health in Bethesda, Maryland.
Although allogeneic hematopoietic stem cell transplantation (HSCT) from a matched sibling donor can be curative, transplants using parental bone marrow or genetically modified autologous transplants without myeloconditiong restore T-cell-mediated immunity but not humoral immunity, Dr. De Ravin said at the American Society of Hematology annual meeting.
Additionally, transplants in older children with SCID-X1 who have persistent immune system defects – despite having received a transplant from a haploidentical donor in infancy – leave the patients with serious medical problems, including the life-long need for immunoglobulin G (IgG) supplementation, recurrent and chronic infections, warts, malnutrition, growth failure, and progressive diseases of the gut and lung.
Previous attempts at using gene therapy to correct mutations in IL2RG, the cause of SCID-X1, used mouse retroviral vectors to insert normal IL2RG into autologous hematopoietic stem cells without chemotherapy conditioning. This treatment restored T-cell immunity, but not B-cell- or natural killer (NK)-cell immunity
Of even greater concern is the fact that among infants with SCID-X1 who received autologous stem cells transduced with murine gamma retrovirus carrying the common gamma chain, 25% developed vector-associated leukemia.
To overcome these problems, investigators in the current study used a lentiviral vector containing an insulator fragment from a chicken beta-globin gene. The insulator fragment allows expression of the gamma chain complementary DNA while protecting against up-regulation of neighboring oncogenes.
Dr. De Ravin reported data on five patients from the ages of 7 to 24 years who had worsening immune dysfunction and complex medical problems, including dependence on immunoglobulin G (IgG) supplementations. All of the patients had previously undergone one or more HSCT from haploidentical donors.
The patients were treated with granulocyte-colony stimulating factor and plerixafor to mobilize peripheral blood cells, and then underwent apheresis to isolate CD34 cells. The cells were transduced in vitro with a lentiviral vector, and reinfused into patients after conditioning with low-dose busulfan (6 mg/kg). The vector was developed by researchers at St Jude Children’s Hospital in Memphis, Tennessee.
At the most recent follow-up, the first two patients treated with the protocol had stable engraftment of gamma-chain expressing cells gene with enhanced expression of B, T, and NK cells, with the cells continuing to show improvements, Early data on the remaining four patients indicates a similar positive trend.
Chimerism studies of the patients’ T-cells showed that the host cells were continuing to increase their contribution, suggesting gradual replacement over time of the T-cell graft, Dr. De Ravin said.
In the second patient treated, an increase in NK cells corresponded with an improvement in chronic warts. In addition, the first two patients began to produce both IgG and antigen-specific responses to vaccination, clearance of chronic norovirus infections, and resolution of their protein-losing enteropathy.
“Gene therapy does not appear to reverse prior organ damage, supporting early intervention to improve immunity in these patients before such damage occurs,” Dr. De Ravin said.
The study was supported by the National Institutes of Health. The viral vector was developed at St. Jude Children’s Research Hospital in Memphis, Tennessee. The authors reported no relevant conflicts of interest.
ORLANDO – – Gene therapy can help restore immune function to older children and young adults with X-linked severe combined immunodeficiency, a team of US investigators reports.
“This is the first demonstration of the use of gene therapy to salvage failed allogeneic hematopoietic stem cell transplants in older SCID-X1 patients,” said Dr. Suk See De Ravin from the Laboratory of Host Defenses at the National Institutes of Health in Bethesda, Maryland.
Although allogeneic hematopoietic stem cell transplantation (HSCT) from a matched sibling donor can be curative, transplants using parental bone marrow or genetically modified autologous transplants without myeloconditiong restore T-cell-mediated immunity but not humoral immunity, Dr. De Ravin said at the American Society of Hematology annual meeting.
Additionally, transplants in older children with SCID-X1 who have persistent immune system defects – despite having received a transplant from a haploidentical donor in infancy – leave the patients with serious medical problems, including the life-long need for immunoglobulin G (IgG) supplementation, recurrent and chronic infections, warts, malnutrition, growth failure, and progressive diseases of the gut and lung.
Previous attempts at using gene therapy to correct mutations in IL2RG, the cause of SCID-X1, used mouse retroviral vectors to insert normal IL2RG into autologous hematopoietic stem cells without chemotherapy conditioning. This treatment restored T-cell immunity, but not B-cell- or natural killer (NK)-cell immunity
Of even greater concern is the fact that among infants with SCID-X1 who received autologous stem cells transduced with murine gamma retrovirus carrying the common gamma chain, 25% developed vector-associated leukemia.
To overcome these problems, investigators in the current study used a lentiviral vector containing an insulator fragment from a chicken beta-globin gene. The insulator fragment allows expression of the gamma chain complementary DNA while protecting against up-regulation of neighboring oncogenes.
Dr. De Ravin reported data on five patients from the ages of 7 to 24 years who had worsening immune dysfunction and complex medical problems, including dependence on immunoglobulin G (IgG) supplementations. All of the patients had previously undergone one or more HSCT from haploidentical donors.
The patients were treated with granulocyte-colony stimulating factor and plerixafor to mobilize peripheral blood cells, and then underwent apheresis to isolate CD34 cells. The cells were transduced in vitro with a lentiviral vector, and reinfused into patients after conditioning with low-dose busulfan (6 mg/kg). The vector was developed by researchers at St Jude Children’s Hospital in Memphis, Tennessee.
At the most recent follow-up, the first two patients treated with the protocol had stable engraftment of gamma-chain expressing cells gene with enhanced expression of B, T, and NK cells, with the cells continuing to show improvements, Early data on the remaining four patients indicates a similar positive trend.
Chimerism studies of the patients’ T-cells showed that the host cells were continuing to increase their contribution, suggesting gradual replacement over time of the T-cell graft, Dr. De Ravin said.
In the second patient treated, an increase in NK cells corresponded with an improvement in chronic warts. In addition, the first two patients began to produce both IgG and antigen-specific responses to vaccination, clearance of chronic norovirus infections, and resolution of their protein-losing enteropathy.
“Gene therapy does not appear to reverse prior organ damage, supporting early intervention to improve immunity in these patients before such damage occurs,” Dr. De Ravin said.
The study was supported by the National Institutes of Health. The viral vector was developed at St. Jude Children’s Research Hospital in Memphis, Tennessee. The authors reported no relevant conflicts of interest.
AT ASH 2015
Key clinical point: Gene therapy can correct B, T, and NK cell immunity in older patients with SCID-X1.
Major finding: The first two patients treated with the protocol had stable engraftment of gamma-chain expressing cells gene with enhanced expression of B, T, and NK cells, with the cells continuing to show improvements.
Data source: Clinical study of 5 patients with X-linked severe combined immunodeficiency syndrome (SCID-X1).
Disclosures: The study was supported by the National Institutes of Health. The viral vector was developed at St. Jude Children’s Research Hospital in Memphis, Tennessee. The authors reported no relevant conflicts of interest.
ASH: Carfilzomib doubles progression free survival in head-to-head trial with bortezomib
In a head-to-head clinical trial, carfilzomib (Kyprolis) plus dexamethasone doubled the length of progression-free survival (PFS) when compared with bortezomib (Velcade) plus dexamethasone for individuals with relapsed or refractory multiple myeloma. Response rates were also significantly higher for those receiving carfilzomib, though neither medication could overcome high-risk cytogenetic factors.
The international multi-center phase 3 open-label ENDEAVOR study (clinicaltrials.gov ID: NCT01568866) compared outcomes for a total of 929 patients who were randomized 1:1 to receive one of the treatments. Randomization was stratified to account for prior treatments, including prior proteasome treatment, as well as disease severity by International Staging System stage.
Reporting for the ENDEAVOR investigators, Dr. Meletios Dimopoulos and his co-authors published their results in The Lancet simultaneously with a presentation at the annual meeting of the American Society of Hematology [Lancet Oncol, http://dx.doi.org/10.1016/S1470-2045(15)00464-7] . Dr. Dimopoulos is a professor in the department of clinical therapeutics at the school of medicine of the National and Kapodistrian University of Athens.
The primary endpoint, PFS in the intention-to-treat population, differed significantly between the study arms when the data were analyzed at a pre-planned interim analysis. For those receiving carfilzomib, PFS was 18.7 months, compared with 9.4 months in the bortezomib group, for a hazard ratio of 0.53 (P less than 0.0001). Data were drawn from a pre-specified interim analysis, and study participation is ongoing, with 28-day dosing cycles repeated until disease progression occurrs or the patients experience unacceptable toxicity or withdraw consent.
Investigators had pre-specified subgroup analyses to examine what populations fared better and worse in each arm. Prior bortezomib exposure did not significantly affect PFS for those receiving carfilzomib. Not enough patients had received carfilzomib prior to the study to permit analysis of this effect. Though carfilzomib arm patients with high-risk factors on cytogenetic analysis fared slightly better than those on bortezomib, “neither proteasome inhibitor appears to significantly overcome the adverse prognostic effect of high-risk cytogenetics,” wrote Dr. Dimopoulos and his co-authors.
Secondary outcome measures included the numbers of patients in each arm achieving complete response or better (with this group broken into “stringent complete response” as well as complete response). Significantly more carfilzomib than bortezomib patients reached this mark (58 patients [13%] vs 29 patients [6%], P equal to 0.001). A larger group of patients in each arm met the secondary outcome measure of experiencing very good partial response or better, with those on the carfelzomib arm faring significantly better than those receiving bortezomib (252 [54%] vs 133 [29%], P less than 0.0001).
Dr. Dimopoulos and his co-authors remarked on the importance of these findings, noting that “The finding that the proportion of patients with a complete response or better and very good partial response or better was higher in the carfilzomib group than in the bortezomib group is encouraging because studies have shown an association between depth of response and improved survival in patients with multiple myeloma.”
ENDEAVOR’s safety analyses included all patients who received at least one dose of study drug. Overall, the carfilzomib patients had a 48% rate of serious adverse events (224 of 463 patients for this analysis), compared to 36% of the bortezomib group (162 of 456 patients).
On-study death rates were similar between the two groups, with 18 of 464 patients (4%) in the carfilzomib group and 16 of 465 patients (3%) in the bortezomib group dying during the study period. Four deaths in each arm were judged related to disease progression.
A pre-planned substudy examined cardiac function for patients in both study arms, and did not find increased risk of left or right ventricular dysfunction or cumulative cardiac injury for carfilzomib when compared with bortezomib.
The open label nature of the study, an acknowledged limitation, was necessitated by the different dosing regimens of the two drugs, but the treatment allocation was hidden from the independent review committee that assessed participant disease status. The funder was also masked to per-group treatment results.
Carfilzomib’s irreversable proteosome inhibition may lead to more sustained proteasomal inhibition, Dr. Dimopoulous and his co-authors posited. Also, carfilzomib can overcome bortezomib resistance and is more potent against cell lines naive to this class of drugs in preclinical studies, they said.
“Taken together, the results from the ENDEAVOR study suggest an important role for carfilzomib-based regimens for patients with relapsed or refractory jultiple myeloma,” wrote Dr. Dimopoulos and his collaborators.
The study was supported by Onyx Pharmaceuticals, Inc, a subsidiary of Amgen. Dr. Dimopoulos reported non-financial support from Onyx Pharmaceuticals, Celgene Corporation, and Ortho-Biotech. Several co-authors reported multiple financial associations with pharmaceutical companies.
On Twitter @karioakes
In a head-to-head clinical trial, carfilzomib (Kyprolis) plus dexamethasone doubled the length of progression-free survival (PFS) when compared with bortezomib (Velcade) plus dexamethasone for individuals with relapsed or refractory multiple myeloma. Response rates were also significantly higher for those receiving carfilzomib, though neither medication could overcome high-risk cytogenetic factors.
The international multi-center phase 3 open-label ENDEAVOR study (clinicaltrials.gov ID: NCT01568866) compared outcomes for a total of 929 patients who were randomized 1:1 to receive one of the treatments. Randomization was stratified to account for prior treatments, including prior proteasome treatment, as well as disease severity by International Staging System stage.
Reporting for the ENDEAVOR investigators, Dr. Meletios Dimopoulos and his co-authors published their results in The Lancet simultaneously with a presentation at the annual meeting of the American Society of Hematology [Lancet Oncol, http://dx.doi.org/10.1016/S1470-2045(15)00464-7] . Dr. Dimopoulos is a professor in the department of clinical therapeutics at the school of medicine of the National and Kapodistrian University of Athens.
The primary endpoint, PFS in the intention-to-treat population, differed significantly between the study arms when the data were analyzed at a pre-planned interim analysis. For those receiving carfilzomib, PFS was 18.7 months, compared with 9.4 months in the bortezomib group, for a hazard ratio of 0.53 (P less than 0.0001). Data were drawn from a pre-specified interim analysis, and study participation is ongoing, with 28-day dosing cycles repeated until disease progression occurrs or the patients experience unacceptable toxicity or withdraw consent.
Investigators had pre-specified subgroup analyses to examine what populations fared better and worse in each arm. Prior bortezomib exposure did not significantly affect PFS for those receiving carfilzomib. Not enough patients had received carfilzomib prior to the study to permit analysis of this effect. Though carfilzomib arm patients with high-risk factors on cytogenetic analysis fared slightly better than those on bortezomib, “neither proteasome inhibitor appears to significantly overcome the adverse prognostic effect of high-risk cytogenetics,” wrote Dr. Dimopoulos and his co-authors.
Secondary outcome measures included the numbers of patients in each arm achieving complete response or better (with this group broken into “stringent complete response” as well as complete response). Significantly more carfilzomib than bortezomib patients reached this mark (58 patients [13%] vs 29 patients [6%], P equal to 0.001). A larger group of patients in each arm met the secondary outcome measure of experiencing very good partial response or better, with those on the carfelzomib arm faring significantly better than those receiving bortezomib (252 [54%] vs 133 [29%], P less than 0.0001).
Dr. Dimopoulos and his co-authors remarked on the importance of these findings, noting that “The finding that the proportion of patients with a complete response or better and very good partial response or better was higher in the carfilzomib group than in the bortezomib group is encouraging because studies have shown an association between depth of response and improved survival in patients with multiple myeloma.”
ENDEAVOR’s safety analyses included all patients who received at least one dose of study drug. Overall, the carfilzomib patients had a 48% rate of serious adverse events (224 of 463 patients for this analysis), compared to 36% of the bortezomib group (162 of 456 patients).
On-study death rates were similar between the two groups, with 18 of 464 patients (4%) in the carfilzomib group and 16 of 465 patients (3%) in the bortezomib group dying during the study period. Four deaths in each arm were judged related to disease progression.
A pre-planned substudy examined cardiac function for patients in both study arms, and did not find increased risk of left or right ventricular dysfunction or cumulative cardiac injury for carfilzomib when compared with bortezomib.
The open label nature of the study, an acknowledged limitation, was necessitated by the different dosing regimens of the two drugs, but the treatment allocation was hidden from the independent review committee that assessed participant disease status. The funder was also masked to per-group treatment results.
Carfilzomib’s irreversable proteosome inhibition may lead to more sustained proteasomal inhibition, Dr. Dimopoulous and his co-authors posited. Also, carfilzomib can overcome bortezomib resistance and is more potent against cell lines naive to this class of drugs in preclinical studies, they said.
“Taken together, the results from the ENDEAVOR study suggest an important role for carfilzomib-based regimens for patients with relapsed or refractory jultiple myeloma,” wrote Dr. Dimopoulos and his collaborators.
The study was supported by Onyx Pharmaceuticals, Inc, a subsidiary of Amgen. Dr. Dimopoulos reported non-financial support from Onyx Pharmaceuticals, Celgene Corporation, and Ortho-Biotech. Several co-authors reported multiple financial associations with pharmaceutical companies.
On Twitter @karioakes
In a head-to-head clinical trial, carfilzomib (Kyprolis) plus dexamethasone doubled the length of progression-free survival (PFS) when compared with bortezomib (Velcade) plus dexamethasone for individuals with relapsed or refractory multiple myeloma. Response rates were also significantly higher for those receiving carfilzomib, though neither medication could overcome high-risk cytogenetic factors.
The international multi-center phase 3 open-label ENDEAVOR study (clinicaltrials.gov ID: NCT01568866) compared outcomes for a total of 929 patients who were randomized 1:1 to receive one of the treatments. Randomization was stratified to account for prior treatments, including prior proteasome treatment, as well as disease severity by International Staging System stage.
Reporting for the ENDEAVOR investigators, Dr. Meletios Dimopoulos and his co-authors published their results in The Lancet simultaneously with a presentation at the annual meeting of the American Society of Hematology [Lancet Oncol, http://dx.doi.org/10.1016/S1470-2045(15)00464-7] . Dr. Dimopoulos is a professor in the department of clinical therapeutics at the school of medicine of the National and Kapodistrian University of Athens.
The primary endpoint, PFS in the intention-to-treat population, differed significantly between the study arms when the data were analyzed at a pre-planned interim analysis. For those receiving carfilzomib, PFS was 18.7 months, compared with 9.4 months in the bortezomib group, for a hazard ratio of 0.53 (P less than 0.0001). Data were drawn from a pre-specified interim analysis, and study participation is ongoing, with 28-day dosing cycles repeated until disease progression occurrs or the patients experience unacceptable toxicity or withdraw consent.
Investigators had pre-specified subgroup analyses to examine what populations fared better and worse in each arm. Prior bortezomib exposure did not significantly affect PFS for those receiving carfilzomib. Not enough patients had received carfilzomib prior to the study to permit analysis of this effect. Though carfilzomib arm patients with high-risk factors on cytogenetic analysis fared slightly better than those on bortezomib, “neither proteasome inhibitor appears to significantly overcome the adverse prognostic effect of high-risk cytogenetics,” wrote Dr. Dimopoulos and his co-authors.
Secondary outcome measures included the numbers of patients in each arm achieving complete response or better (with this group broken into “stringent complete response” as well as complete response). Significantly more carfilzomib than bortezomib patients reached this mark (58 patients [13%] vs 29 patients [6%], P equal to 0.001). A larger group of patients in each arm met the secondary outcome measure of experiencing very good partial response or better, with those on the carfelzomib arm faring significantly better than those receiving bortezomib (252 [54%] vs 133 [29%], P less than 0.0001).
Dr. Dimopoulos and his co-authors remarked on the importance of these findings, noting that “The finding that the proportion of patients with a complete response or better and very good partial response or better was higher in the carfilzomib group than in the bortezomib group is encouraging because studies have shown an association between depth of response and improved survival in patients with multiple myeloma.”
ENDEAVOR’s safety analyses included all patients who received at least one dose of study drug. Overall, the carfilzomib patients had a 48% rate of serious adverse events (224 of 463 patients for this analysis), compared to 36% of the bortezomib group (162 of 456 patients).
On-study death rates were similar between the two groups, with 18 of 464 patients (4%) in the carfilzomib group and 16 of 465 patients (3%) in the bortezomib group dying during the study period. Four deaths in each arm were judged related to disease progression.
A pre-planned substudy examined cardiac function for patients in both study arms, and did not find increased risk of left or right ventricular dysfunction or cumulative cardiac injury for carfilzomib when compared with bortezomib.
The open label nature of the study, an acknowledged limitation, was necessitated by the different dosing regimens of the two drugs, but the treatment allocation was hidden from the independent review committee that assessed participant disease status. The funder was also masked to per-group treatment results.
Carfilzomib’s irreversable proteosome inhibition may lead to more sustained proteasomal inhibition, Dr. Dimopoulous and his co-authors posited. Also, carfilzomib can overcome bortezomib resistance and is more potent against cell lines naive to this class of drugs in preclinical studies, they said.
“Taken together, the results from the ENDEAVOR study suggest an important role for carfilzomib-based regimens for patients with relapsed or refractory jultiple myeloma,” wrote Dr. Dimopoulos and his collaborators.
The study was supported by Onyx Pharmaceuticals, Inc, a subsidiary of Amgen. Dr. Dimopoulos reported non-financial support from Onyx Pharmaceuticals, Celgene Corporation, and Ortho-Biotech. Several co-authors reported multiple financial associations with pharmaceutical companies.
On Twitter @karioakes
FROM ASH 2015
Key clinical point: Carfilzomib nearly doubled progression-free survival (PFS) compared with bortezomib for relapsed and refractory multiple myeloma
Major finding: For those receiving carfilzomib, PFS was 18.7 months, compared with 9.4 months in the bortezomib group, for a hazard ratio of 0.53 (P less than .0001).
Data source: International randomized open-label clinical trial of 979 patients with refractory or relapsed multiple myeloma.
Disclosures: The study was supported by Onyx Pharmaceuticals, Inc, a subsidiary of Amgen. Dr. Dimopoulos reported non-financial support from Onyx Pharmaceuticals, Celgene Corporation, and Ortho-Biotech. Several co-authors reported multiple financial associations with pharmaceutical companies.
Complete excision most effective for BI-ALCL
![]()
Photo courtesy of the FDA
The optimal treatment approach for most women with breast implant-associated anaplastic large-cell lymphoma (BI-ALCL) is complete surgical excision of the implant and surrounding capsule, a new study suggests.
The study, published in the Journal of Clinical Oncology, represents the most comprehensive study of BI-ALCL to date, including 87 patients and 30 investigators from 14 institutions across 5 continents.
BI-ALCL is a rare T-cell lymphoma that forms in the scar tissue or in the fluid surrounding a breast implant. The disease manifests as a large fluid collection around the implant over a year after implantation, usually taking an average of 8 years to develop.
An estimated 10 million women worldwide have breast implants, and the annual incidence of BI-ALCL is estimated to be 0.1 to 0.3 per 100,000 women with breast implants.
“Although this disease is rare, it appears to be amenable to treatment, and, in the vast majority of patients, the outcome is very good,” said Mark Clemens, MD, of The University of Texas MD Anderson Cancer Center in Houston.
“The disease can be reliably diagnosed, and, when treated appropriately, it has a good prognosis.”
Still, the optimal management for BI-ALCL hasn’t been clear. So with this study, Dr Clemens and his colleagues sought to evaluate treatment efficacy on disease outcomes and determine the best treatment approach. The study expands on previous research published in the Journal of Clinical Oncology in 2014.
The researchers gathered detailed treatment and outcome information from a total of 87 BI-ALCL patients, including 37 whose information had not previously been published. A review of treatment approaches in relation to event-free survival and overall survival revealed that surgery was the optimal frontline therapy for BI-ALCL.
“We determined that complete surgical excision was essential for the management of this disease,” Dr Clemens said. “Patients did not do as well unless they were treated with full removal of the breast implant and complete excision of the capsule around the implant.”
Patients with complete surgical excision had a recurrence rate of 4% at 5 years, compared to 28% for patients who received radiation therapy and 32% for chemotherapy.
In addition, patients who underwent a complete surgical excision had better overall survival (P=0.022) and event-free survival (P=0.014) than patients who received a partial capsulectomy, systemic chemotherapy, or radiation.
“This lymphoma represents a different paradigm from systemic anaplastic large-cell lymphoma, in particular because of its strong association with breast implants,” said Roberto N. Miranda, MD, of The University of Texas MD Anderson Cancer Center.
“We have demonstrated that this is a predominantly localized disease where surgical excision has a primary role.”
The researchers emphasized that, despite the overall good prognosis, some rare cases of BI-ALCL exhibit more aggressive behavior with systemic dissemination. As a part of this study, the team is gathering tissue from these patients to assess underlying mechanisms for progression of disease.
Additional research is ongoing to optimize therapy for these cases through genetic profiling and defining the role of chemotherapy and radiation. The researchers are also studying animal models to further assess the role of breast implants in the pathogenesis of this lymphoma. ![]()
![]()
Photo courtesy of the FDA
The optimal treatment approach for most women with breast implant-associated anaplastic large-cell lymphoma (BI-ALCL) is complete surgical excision of the implant and surrounding capsule, a new study suggests.
The study, published in the Journal of Clinical Oncology, represents the most comprehensive study of BI-ALCL to date, including 87 patients and 30 investigators from 14 institutions across 5 continents.
BI-ALCL is a rare T-cell lymphoma that forms in the scar tissue or in the fluid surrounding a breast implant. The disease manifests as a large fluid collection around the implant over a year after implantation, usually taking an average of 8 years to develop.
An estimated 10 million women worldwide have breast implants, and the annual incidence of BI-ALCL is estimated to be 0.1 to 0.3 per 100,000 women with breast implants.
“Although this disease is rare, it appears to be amenable to treatment, and, in the vast majority of patients, the outcome is very good,” said Mark Clemens, MD, of The University of Texas MD Anderson Cancer Center in Houston.
“The disease can be reliably diagnosed, and, when treated appropriately, it has a good prognosis.”
Still, the optimal management for BI-ALCL hasn’t been clear. So with this study, Dr Clemens and his colleagues sought to evaluate treatment efficacy on disease outcomes and determine the best treatment approach. The study expands on previous research published in the Journal of Clinical Oncology in 2014.
The researchers gathered detailed treatment and outcome information from a total of 87 BI-ALCL patients, including 37 whose information had not previously been published. A review of treatment approaches in relation to event-free survival and overall survival revealed that surgery was the optimal frontline therapy for BI-ALCL.
“We determined that complete surgical excision was essential for the management of this disease,” Dr Clemens said. “Patients did not do as well unless they were treated with full removal of the breast implant and complete excision of the capsule around the implant.”
Patients with complete surgical excision had a recurrence rate of 4% at 5 years, compared to 28% for patients who received radiation therapy and 32% for chemotherapy.
In addition, patients who underwent a complete surgical excision had better overall survival (P=0.022) and event-free survival (P=0.014) than patients who received a partial capsulectomy, systemic chemotherapy, or radiation.
“This lymphoma represents a different paradigm from systemic anaplastic large-cell lymphoma, in particular because of its strong association with breast implants,” said Roberto N. Miranda, MD, of The University of Texas MD Anderson Cancer Center.
“We have demonstrated that this is a predominantly localized disease where surgical excision has a primary role.”
The researchers emphasized that, despite the overall good prognosis, some rare cases of BI-ALCL exhibit more aggressive behavior with systemic dissemination. As a part of this study, the team is gathering tissue from these patients to assess underlying mechanisms for progression of disease.
Additional research is ongoing to optimize therapy for these cases through genetic profiling and defining the role of chemotherapy and radiation. The researchers are also studying animal models to further assess the role of breast implants in the pathogenesis of this lymphoma. ![]()
![]()
Photo courtesy of the FDA
The optimal treatment approach for most women with breast implant-associated anaplastic large-cell lymphoma (BI-ALCL) is complete surgical excision of the implant and surrounding capsule, a new study suggests.
The study, published in the Journal of Clinical Oncology, represents the most comprehensive study of BI-ALCL to date, including 87 patients and 30 investigators from 14 institutions across 5 continents.
BI-ALCL is a rare T-cell lymphoma that forms in the scar tissue or in the fluid surrounding a breast implant. The disease manifests as a large fluid collection around the implant over a year after implantation, usually taking an average of 8 years to develop.
An estimated 10 million women worldwide have breast implants, and the annual incidence of BI-ALCL is estimated to be 0.1 to 0.3 per 100,000 women with breast implants.
“Although this disease is rare, it appears to be amenable to treatment, and, in the vast majority of patients, the outcome is very good,” said Mark Clemens, MD, of The University of Texas MD Anderson Cancer Center in Houston.
“The disease can be reliably diagnosed, and, when treated appropriately, it has a good prognosis.”
Still, the optimal management for BI-ALCL hasn’t been clear. So with this study, Dr Clemens and his colleagues sought to evaluate treatment efficacy on disease outcomes and determine the best treatment approach. The study expands on previous research published in the Journal of Clinical Oncology in 2014.
The researchers gathered detailed treatment and outcome information from a total of 87 BI-ALCL patients, including 37 whose information had not previously been published. A review of treatment approaches in relation to event-free survival and overall survival revealed that surgery was the optimal frontline therapy for BI-ALCL.
“We determined that complete surgical excision was essential for the management of this disease,” Dr Clemens said. “Patients did not do as well unless they were treated with full removal of the breast implant and complete excision of the capsule around the implant.”
Patients with complete surgical excision had a recurrence rate of 4% at 5 years, compared to 28% for patients who received radiation therapy and 32% for chemotherapy.
In addition, patients who underwent a complete surgical excision had better overall survival (P=0.022) and event-free survival (P=0.014) than patients who received a partial capsulectomy, systemic chemotherapy, or radiation.
“This lymphoma represents a different paradigm from systemic anaplastic large-cell lymphoma, in particular because of its strong association with breast implants,” said Roberto N. Miranda, MD, of The University of Texas MD Anderson Cancer Center.
“We have demonstrated that this is a predominantly localized disease where surgical excision has a primary role.”
The researchers emphasized that, despite the overall good prognosis, some rare cases of BI-ALCL exhibit more aggressive behavior with systemic dissemination. As a part of this study, the team is gathering tissue from these patients to assess underlying mechanisms for progression of disease.
Additional research is ongoing to optimize therapy for these cases through genetic profiling and defining the role of chemotherapy and radiation. The researchers are also studying animal models to further assess the role of breast implants in the pathogenesis of this lymphoma. ![]()
Multiple myeloma advances will be a hot topic at ASH
With the approvals of three new therapies for multiple myeloma in just the last month – elotuzumab, ixazomib, and daratumumab – sessions at ASH 2015 will be buzzing with discussions of how the availability of these new therapies is likely to influence disease management. Other leading abstracts, highlighted at a press conference prior to the annual meeting of the American Society of Hematology, address gene therapy, precision medicine, and stem cell transplant for sickle cell disease. We’re providing the links to these top abstracts here, and invite you to look for our upcoming daily newsletters and next-best-thing-to-being-there coverage of ASH.
Experimental, targeted treatments for blood cancer
• 507: Daratumumab in Combination with Lenalidomide and Dexamethasone in Patients with Relapsed or Relapsed and Refractory Multiple Myeloma: Updated Results of a Phase I/II Study (GEN503).
• 727: Ixazomib, an Investigational Oral Proteasome Inhibitor (PI), in Combination with Lenalidomide and Dexamethasone (IRd), Significantly Extends Progression-Free Survival (PFS) for Patients (Pts) with Relapsed and/or Refractory Multiple Myeloma (RRMM): The Phase III Tourmaline-MM1 Study (NCT01564537).
• 391: Autologous Transplantation for Multiple Myeloma in the Era of New Drugs: A Phase III Study of the Intergroupe Francophone Du Myelome (IFM/DFCI 2009 Trial).
• 495: Results from the International, Randomized Phase III Study of Ibrutinib Versus Chlorambucil in Patients 65 Years and Older with Treatment-Naïve CLL/SLL (RESONATE-2TM).
Experimental gene therapy
• 201: Update of Results from the Northstar Study (HGB-204): A Phase I/II Study of Gene Therapy for Beta-Thalassemia Major Via Transplantation of Autologous Hematopoietic Stem Cells Transduced Ex-Vivo with a Lentiviral Beta AT87Q-Globin Vector (LentiGlobin BB305 Drug Product).
• 261: Lentiviral Hematopoietic Stem Cell Gene Therapy for Older Patients with X-Linked Severe Combined Immunodeficiency.
Precision medicine
• 250: Genetic Risk Factors for the Development of Osteonecrosis in Children Under Age 10 Treated for Acute Lymphoblastic Leukemia.
• Special Scientific Symposium: Precision Medicine in Cancer Therapy.
Coagulation
• 5: Source of Factor VIII Replacement (PLASMATIC OR RECOMBINANT) and Incidence of Inhibitory Alloantibodies in Previously Untreated Patients with Severe Hemophilia a: The Multicenter Randomized Sippet Study.
New sickle cell disease research
•541: Hematopoietic Stem Cell Transplantation from HLA Identical Sibling For Sickle Cell Disease an International Survey on Behalf of Eurocord-Monacord, EBMT Paediatric Disease Working Party and CIBMTR.
With the approvals of three new therapies for multiple myeloma in just the last month – elotuzumab, ixazomib, and daratumumab – sessions at ASH 2015 will be buzzing with discussions of how the availability of these new therapies is likely to influence disease management. Other leading abstracts, highlighted at a press conference prior to the annual meeting of the American Society of Hematology, address gene therapy, precision medicine, and stem cell transplant for sickle cell disease. We’re providing the links to these top abstracts here, and invite you to look for our upcoming daily newsletters and next-best-thing-to-being-there coverage of ASH.
Experimental, targeted treatments for blood cancer
• 507: Daratumumab in Combination with Lenalidomide and Dexamethasone in Patients with Relapsed or Relapsed and Refractory Multiple Myeloma: Updated Results of a Phase I/II Study (GEN503).
• 727: Ixazomib, an Investigational Oral Proteasome Inhibitor (PI), in Combination with Lenalidomide and Dexamethasone (IRd), Significantly Extends Progression-Free Survival (PFS) for Patients (Pts) with Relapsed and/or Refractory Multiple Myeloma (RRMM): The Phase III Tourmaline-MM1 Study (NCT01564537).
• 391: Autologous Transplantation for Multiple Myeloma in the Era of New Drugs: A Phase III Study of the Intergroupe Francophone Du Myelome (IFM/DFCI 2009 Trial).
• 495: Results from the International, Randomized Phase III Study of Ibrutinib Versus Chlorambucil in Patients 65 Years and Older with Treatment-Naïve CLL/SLL (RESONATE-2TM).
Experimental gene therapy
• 201: Update of Results from the Northstar Study (HGB-204): A Phase I/II Study of Gene Therapy for Beta-Thalassemia Major Via Transplantation of Autologous Hematopoietic Stem Cells Transduced Ex-Vivo with a Lentiviral Beta AT87Q-Globin Vector (LentiGlobin BB305 Drug Product).
• 261: Lentiviral Hematopoietic Stem Cell Gene Therapy for Older Patients with X-Linked Severe Combined Immunodeficiency.
Precision medicine
• 250: Genetic Risk Factors for the Development of Osteonecrosis in Children Under Age 10 Treated for Acute Lymphoblastic Leukemia.
• Special Scientific Symposium: Precision Medicine in Cancer Therapy.
Coagulation
• 5: Source of Factor VIII Replacement (PLASMATIC OR RECOMBINANT) and Incidence of Inhibitory Alloantibodies in Previously Untreated Patients with Severe Hemophilia a: The Multicenter Randomized Sippet Study.
New sickle cell disease research
•541: Hematopoietic Stem Cell Transplantation from HLA Identical Sibling For Sickle Cell Disease an International Survey on Behalf of Eurocord-Monacord, EBMT Paediatric Disease Working Party and CIBMTR.
With the approvals of three new therapies for multiple myeloma in just the last month – elotuzumab, ixazomib, and daratumumab – sessions at ASH 2015 will be buzzing with discussions of how the availability of these new therapies is likely to influence disease management. Other leading abstracts, highlighted at a press conference prior to the annual meeting of the American Society of Hematology, address gene therapy, precision medicine, and stem cell transplant for sickle cell disease. We’re providing the links to these top abstracts here, and invite you to look for our upcoming daily newsletters and next-best-thing-to-being-there coverage of ASH.
Experimental, targeted treatments for blood cancer
• 507: Daratumumab in Combination with Lenalidomide and Dexamethasone in Patients with Relapsed or Relapsed and Refractory Multiple Myeloma: Updated Results of a Phase I/II Study (GEN503).
• 727: Ixazomib, an Investigational Oral Proteasome Inhibitor (PI), in Combination with Lenalidomide and Dexamethasone (IRd), Significantly Extends Progression-Free Survival (PFS) for Patients (Pts) with Relapsed and/or Refractory Multiple Myeloma (RRMM): The Phase III Tourmaline-MM1 Study (NCT01564537).
• 391: Autologous Transplantation for Multiple Myeloma in the Era of New Drugs: A Phase III Study of the Intergroupe Francophone Du Myelome (IFM/DFCI 2009 Trial).
• 495: Results from the International, Randomized Phase III Study of Ibrutinib Versus Chlorambucil in Patients 65 Years and Older with Treatment-Naïve CLL/SLL (RESONATE-2TM).
Experimental gene therapy
• 201: Update of Results from the Northstar Study (HGB-204): A Phase I/II Study of Gene Therapy for Beta-Thalassemia Major Via Transplantation of Autologous Hematopoietic Stem Cells Transduced Ex-Vivo with a Lentiviral Beta AT87Q-Globin Vector (LentiGlobin BB305 Drug Product).
• 261: Lentiviral Hematopoietic Stem Cell Gene Therapy for Older Patients with X-Linked Severe Combined Immunodeficiency.
Precision medicine
• 250: Genetic Risk Factors for the Development of Osteonecrosis in Children Under Age 10 Treated for Acute Lymphoblastic Leukemia.
• Special Scientific Symposium: Precision Medicine in Cancer Therapy.
Coagulation
• 5: Source of Factor VIII Replacement (PLASMATIC OR RECOMBINANT) and Incidence of Inhibitory Alloantibodies in Previously Untreated Patients with Severe Hemophilia a: The Multicenter Randomized Sippet Study.
New sickle cell disease research
•541: Hematopoietic Stem Cell Transplantation from HLA Identical Sibling For Sickle Cell Disease an International Survey on Behalf of Eurocord-Monacord, EBMT Paediatric Disease Working Party and CIBMTR.
FROM ASH 2015
Expert panel issues guidelines for treatment of hematologic cancers in pregnancy
Consensus guidelines for the perinatal management of hematologic malignancies detected during pregnancy have been issued by a panel of international experts.
The guidelines, published online in the Journal of Clinical Oncology, aim to ensure that timely treatment of the cancers is not delayed in pregnant women (doi: 10.1200/JCO.2015.62.4445).
While rare, hematologic malignancies in pregnancy introduce clinical, social, ethical, and moral dilemmas. Evidence-based data are scarce, according to the researchers, who note the International Network on Cancer, Infertility and Pregnancy registers all cancers occurring during gestation.
“Patient accrual is ongoing and essential, because registration of new cases and long-term follow-up will improve clinical knowledge and increase the level of evidence,” Dr. Michael Lishner of Tel Aviv University and Meir Medical Center, Kfar Saba, Israel, and his fellow panelists wrote.
Hodgkin lymphoma
The researchers note that Hodgkin lymphoma is the most common hematologic cancer in pregnancy, and the prognosis for these patients is excellent. When diagnosed during the first trimester, a regimen based on vinblastine monotherapy has been used. ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) therapy can be used postpartum and has been used in cases of progression during pregnancy, the panelists wrote.
“The limited data available suggest that ABVD may be administered safely and effectively during the latter phases of pregnancy,” the panel wrote. “Although it may be associated with prematurity and lower birth weights, studies have not reported significant disadvantages.”
Non-Hodgkin lymphoma
The second most common cancer in pregnancy is non-Hodgkin lymphoma. In the case of indolent disease, watchful waiting is possible, with the intent to treat with monoclonal antibodies – with or without chemotherapy – if symptoms or evidence of disease progression are noted. Steroids can be administered during the first trimester as a bridge to the second trimester, when chemotherapy can be used with relatively greater safety, the panelists noted.
Aggressive lymphomas diagnosed before 20 weeks’ gestation warrant pregnancy termination and treatment, they recommend. When diagnosed after 20 weeks, therapy should be comparable to that given a nonpregnant woman, including monoclonal antibodies (R-CHOP).
Chronic myeloid leukemia
Chronic myeloid leukemia occurs in approximately 1 in 100,000 pregnancies and is typically diagnosed during routine blood testing in an asymptomatic patient. As a result, treatment may not be needed until the patient’s white count or platelet count have risen to levels associated with the onset of symptoms. An approximate guideline is a white cell count greater than 100 X 109/L and a platelet count greater than 500 X 109/L.
Therapeutic approaches in pregnancy include interferon-a (INF-a), which does not inhibit DNA synthesis or readily cross the placenta, and leukapheresis, which is frequently required two to three times per week during the first and second trimesters. Counts tend to drop during the third trimester, allowing less frequent intervention.
Consideration should be given to adding aspirin or low-molecular-weight heparin (LMWH) when the platelet count exceeds 1,000 X 109/L.
Myeloproliferative neoplasms
The most common myeloproliferative neoplasm seen in women of childbearing age is essential thrombocytosis.
“A large meta-analysis of pregnant women with essential thrombocytosis reported a live birth rate of 50%-70%, first trimester loss in 25%-40%, late pregnancy loss in 10%, placental abruption in 3.6%, and intrauterine growth restriction in 4.5%. Maternal morbidity is rare, but stroke has been reported,” according to the panelists.
Limited literature suggests similar outcomes for pregnant women with polycythemia vera and primary myelofibrosis.
In low-risk pregnancies, aspirin (75 mg/day) should be offered unless clearly contraindicated. For women with polycythemia vera, venesection may be continued when tolerated to maintain the hematocrit within the gestation-appropriate range.
Fetal ultrasound scans should be performed at 20, 26, and 34 weeks of gestation and uterine artery Doppler should be performed at 20 weeks’ gestation. If the mean pulsatility index exceeds 1.4, the pregnancy may be considered high risk, and treatment and monitoring should be increased.
In high-risk pregnancies, additional treatment includes cytoreductive therapy with or without LMWH. If cytoreductive therapy is required, INF-a should be titrated to maintain a platelet count of less than 400 X 109/L and hematocrit within appropriate range.
Local protocols regarding interruption of LMWH should be adhered to during labor, and dehydration should be avoided. Platelet count and hematocrit may increase postpartum, requiring cytoreductive therapy. Thromboprophylaxis should be considered at 6 weeks’ postpartum because of the increased risk of thrombosis, the guidelines note.
Acute leukemia
“The remarkable anemia, thrombocytopenia, and neutropenia that characterize acute myeloid and lymphoblastic leukemia” require prompt treatment. Leukapheresis in the presence of clinically significant evidence of leukostasis can be considered, regardless of gestational stage. When patients are diagnosed with acute myeloid leukemia during the first trimester, pregnancy termination followed by conventional induction therapy (cytarabine/anthracycline) is recommended.
Those diagnosed later in pregnancy can receive conventional induction therapy, although this seems to be associated with increased risk of fetal growth restriction and even fetal loss. “Notably, neonates rarely experience neutropenia and cardiac impairment unless exposed to lipophilic idarubicin, which should not be used,” the panelists wrote.
When acute promyelocytic leukemia is diagnosed in the first trimester, pregnancy termination is recommended before initiating conventional ATRA-anthracycline therapy. Later in pregnancy, the regimen demonstrates low teratogenicity and can be used in women diagnosed after that stage. Arsenic treatment is highly teratogenic and is prohibited throughout gestation.
Acute lymphocytic leukemia (ALL) requires prophylactic CNS therapy, including methotrexate and L-asparaginase, which are fetotoxic. Methotrexate interferes with organogenesis and is prohibited before week 20 of gestation. L-asparaginase may increase the high risk for thromboembolic events attributed to the combination of pregnancy and malignancy.
Notably, tyrosine kinase inhibitors, essential for patients with Philadelphia chromosome–positive ALL, are teratogenic. Given these limitations, women diagnosed with ALL before 20 weeks’ gestation should undergo termination of the pregnancy and start conventional treatment. After week 20, conventional chemotherapy can be administered during pregnancy. Tyrosine kinase inhibitors can be initiated postpartum.
The guidelines also contain recommendations on diagnostic testing and radiotherapy, maternal supportive care, and perinatal and pediatric aspects of hematologic malignancies in pregnancy. An online appendix offers recommendations on the treatment of rare hematologic malignancies, including hairy cell leukemia, multiple myeloma, and myelodysplastic syndromes.
Dr. Lishner and nine of his coauthors had no financial relationships to disclose. Three coauthors received honoraria and research funding or are consultants to a wide variety of drug makers.
On Twitter @maryjodales
Consensus guidelines for the perinatal management of hematologic malignancies detected during pregnancy have been issued by a panel of international experts.
The guidelines, published online in the Journal of Clinical Oncology, aim to ensure that timely treatment of the cancers is not delayed in pregnant women (doi: 10.1200/JCO.2015.62.4445).
While rare, hematologic malignancies in pregnancy introduce clinical, social, ethical, and moral dilemmas. Evidence-based data are scarce, according to the researchers, who note the International Network on Cancer, Infertility and Pregnancy registers all cancers occurring during gestation.
“Patient accrual is ongoing and essential, because registration of new cases and long-term follow-up will improve clinical knowledge and increase the level of evidence,” Dr. Michael Lishner of Tel Aviv University and Meir Medical Center, Kfar Saba, Israel, and his fellow panelists wrote.
Hodgkin lymphoma
The researchers note that Hodgkin lymphoma is the most common hematologic cancer in pregnancy, and the prognosis for these patients is excellent. When diagnosed during the first trimester, a regimen based on vinblastine monotherapy has been used. ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) therapy can be used postpartum and has been used in cases of progression during pregnancy, the panelists wrote.
“The limited data available suggest that ABVD may be administered safely and effectively during the latter phases of pregnancy,” the panel wrote. “Although it may be associated with prematurity and lower birth weights, studies have not reported significant disadvantages.”
Non-Hodgkin lymphoma
The second most common cancer in pregnancy is non-Hodgkin lymphoma. In the case of indolent disease, watchful waiting is possible, with the intent to treat with monoclonal antibodies – with or without chemotherapy – if symptoms or evidence of disease progression are noted. Steroids can be administered during the first trimester as a bridge to the second trimester, when chemotherapy can be used with relatively greater safety, the panelists noted.
Aggressive lymphomas diagnosed before 20 weeks’ gestation warrant pregnancy termination and treatment, they recommend. When diagnosed after 20 weeks, therapy should be comparable to that given a nonpregnant woman, including monoclonal antibodies (R-CHOP).
Chronic myeloid leukemia
Chronic myeloid leukemia occurs in approximately 1 in 100,000 pregnancies and is typically diagnosed during routine blood testing in an asymptomatic patient. As a result, treatment may not be needed until the patient’s white count or platelet count have risen to levels associated with the onset of symptoms. An approximate guideline is a white cell count greater than 100 X 109/L and a platelet count greater than 500 X 109/L.
Therapeutic approaches in pregnancy include interferon-a (INF-a), which does not inhibit DNA synthesis or readily cross the placenta, and leukapheresis, which is frequently required two to three times per week during the first and second trimesters. Counts tend to drop during the third trimester, allowing less frequent intervention.
Consideration should be given to adding aspirin or low-molecular-weight heparin (LMWH) when the platelet count exceeds 1,000 X 109/L.
Myeloproliferative neoplasms
The most common myeloproliferative neoplasm seen in women of childbearing age is essential thrombocytosis.
“A large meta-analysis of pregnant women with essential thrombocytosis reported a live birth rate of 50%-70%, first trimester loss in 25%-40%, late pregnancy loss in 10%, placental abruption in 3.6%, and intrauterine growth restriction in 4.5%. Maternal morbidity is rare, but stroke has been reported,” according to the panelists.
Limited literature suggests similar outcomes for pregnant women with polycythemia vera and primary myelofibrosis.
In low-risk pregnancies, aspirin (75 mg/day) should be offered unless clearly contraindicated. For women with polycythemia vera, venesection may be continued when tolerated to maintain the hematocrit within the gestation-appropriate range.
Fetal ultrasound scans should be performed at 20, 26, and 34 weeks of gestation and uterine artery Doppler should be performed at 20 weeks’ gestation. If the mean pulsatility index exceeds 1.4, the pregnancy may be considered high risk, and treatment and monitoring should be increased.
In high-risk pregnancies, additional treatment includes cytoreductive therapy with or without LMWH. If cytoreductive therapy is required, INF-a should be titrated to maintain a platelet count of less than 400 X 109/L and hematocrit within appropriate range.
Local protocols regarding interruption of LMWH should be adhered to during labor, and dehydration should be avoided. Platelet count and hematocrit may increase postpartum, requiring cytoreductive therapy. Thromboprophylaxis should be considered at 6 weeks’ postpartum because of the increased risk of thrombosis, the guidelines note.
Acute leukemia
“The remarkable anemia, thrombocytopenia, and neutropenia that characterize acute myeloid and lymphoblastic leukemia” require prompt treatment. Leukapheresis in the presence of clinically significant evidence of leukostasis can be considered, regardless of gestational stage. When patients are diagnosed with acute myeloid leukemia during the first trimester, pregnancy termination followed by conventional induction therapy (cytarabine/anthracycline) is recommended.
Those diagnosed later in pregnancy can receive conventional induction therapy, although this seems to be associated with increased risk of fetal growth restriction and even fetal loss. “Notably, neonates rarely experience neutropenia and cardiac impairment unless exposed to lipophilic idarubicin, which should not be used,” the panelists wrote.
When acute promyelocytic leukemia is diagnosed in the first trimester, pregnancy termination is recommended before initiating conventional ATRA-anthracycline therapy. Later in pregnancy, the regimen demonstrates low teratogenicity and can be used in women diagnosed after that stage. Arsenic treatment is highly teratogenic and is prohibited throughout gestation.
Acute lymphocytic leukemia (ALL) requires prophylactic CNS therapy, including methotrexate and L-asparaginase, which are fetotoxic. Methotrexate interferes with organogenesis and is prohibited before week 20 of gestation. L-asparaginase may increase the high risk for thromboembolic events attributed to the combination of pregnancy and malignancy.
Notably, tyrosine kinase inhibitors, essential for patients with Philadelphia chromosome–positive ALL, are teratogenic. Given these limitations, women diagnosed with ALL before 20 weeks’ gestation should undergo termination of the pregnancy and start conventional treatment. After week 20, conventional chemotherapy can be administered during pregnancy. Tyrosine kinase inhibitors can be initiated postpartum.
The guidelines also contain recommendations on diagnostic testing and radiotherapy, maternal supportive care, and perinatal and pediatric aspects of hematologic malignancies in pregnancy. An online appendix offers recommendations on the treatment of rare hematologic malignancies, including hairy cell leukemia, multiple myeloma, and myelodysplastic syndromes.
Dr. Lishner and nine of his coauthors had no financial relationships to disclose. Three coauthors received honoraria and research funding or are consultants to a wide variety of drug makers.
On Twitter @maryjodales
Consensus guidelines for the perinatal management of hematologic malignancies detected during pregnancy have been issued by a panel of international experts.
The guidelines, published online in the Journal of Clinical Oncology, aim to ensure that timely treatment of the cancers is not delayed in pregnant women (doi: 10.1200/JCO.2015.62.4445).
While rare, hematologic malignancies in pregnancy introduce clinical, social, ethical, and moral dilemmas. Evidence-based data are scarce, according to the researchers, who note the International Network on Cancer, Infertility and Pregnancy registers all cancers occurring during gestation.
“Patient accrual is ongoing and essential, because registration of new cases and long-term follow-up will improve clinical knowledge and increase the level of evidence,” Dr. Michael Lishner of Tel Aviv University and Meir Medical Center, Kfar Saba, Israel, and his fellow panelists wrote.
Hodgkin lymphoma
The researchers note that Hodgkin lymphoma is the most common hematologic cancer in pregnancy, and the prognosis for these patients is excellent. When diagnosed during the first trimester, a regimen based on vinblastine monotherapy has been used. ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) therapy can be used postpartum and has been used in cases of progression during pregnancy, the panelists wrote.
“The limited data available suggest that ABVD may be administered safely and effectively during the latter phases of pregnancy,” the panel wrote. “Although it may be associated with prematurity and lower birth weights, studies have not reported significant disadvantages.”
Non-Hodgkin lymphoma
The second most common cancer in pregnancy is non-Hodgkin lymphoma. In the case of indolent disease, watchful waiting is possible, with the intent to treat with monoclonal antibodies – with or without chemotherapy – if symptoms or evidence of disease progression are noted. Steroids can be administered during the first trimester as a bridge to the second trimester, when chemotherapy can be used with relatively greater safety, the panelists noted.
Aggressive lymphomas diagnosed before 20 weeks’ gestation warrant pregnancy termination and treatment, they recommend. When diagnosed after 20 weeks, therapy should be comparable to that given a nonpregnant woman, including monoclonal antibodies (R-CHOP).
Chronic myeloid leukemia
Chronic myeloid leukemia occurs in approximately 1 in 100,000 pregnancies and is typically diagnosed during routine blood testing in an asymptomatic patient. As a result, treatment may not be needed until the patient’s white count or platelet count have risen to levels associated with the onset of symptoms. An approximate guideline is a white cell count greater than 100 X 109/L and a platelet count greater than 500 X 109/L.
Therapeutic approaches in pregnancy include interferon-a (INF-a), which does not inhibit DNA synthesis or readily cross the placenta, and leukapheresis, which is frequently required two to three times per week during the first and second trimesters. Counts tend to drop during the third trimester, allowing less frequent intervention.
Consideration should be given to adding aspirin or low-molecular-weight heparin (LMWH) when the platelet count exceeds 1,000 X 109/L.
Myeloproliferative neoplasms
The most common myeloproliferative neoplasm seen in women of childbearing age is essential thrombocytosis.
“A large meta-analysis of pregnant women with essential thrombocytosis reported a live birth rate of 50%-70%, first trimester loss in 25%-40%, late pregnancy loss in 10%, placental abruption in 3.6%, and intrauterine growth restriction in 4.5%. Maternal morbidity is rare, but stroke has been reported,” according to the panelists.
Limited literature suggests similar outcomes for pregnant women with polycythemia vera and primary myelofibrosis.
In low-risk pregnancies, aspirin (75 mg/day) should be offered unless clearly contraindicated. For women with polycythemia vera, venesection may be continued when tolerated to maintain the hematocrit within the gestation-appropriate range.
Fetal ultrasound scans should be performed at 20, 26, and 34 weeks of gestation and uterine artery Doppler should be performed at 20 weeks’ gestation. If the mean pulsatility index exceeds 1.4, the pregnancy may be considered high risk, and treatment and monitoring should be increased.
In high-risk pregnancies, additional treatment includes cytoreductive therapy with or without LMWH. If cytoreductive therapy is required, INF-a should be titrated to maintain a platelet count of less than 400 X 109/L and hematocrit within appropriate range.
Local protocols regarding interruption of LMWH should be adhered to during labor, and dehydration should be avoided. Platelet count and hematocrit may increase postpartum, requiring cytoreductive therapy. Thromboprophylaxis should be considered at 6 weeks’ postpartum because of the increased risk of thrombosis, the guidelines note.
Acute leukemia
“The remarkable anemia, thrombocytopenia, and neutropenia that characterize acute myeloid and lymphoblastic leukemia” require prompt treatment. Leukapheresis in the presence of clinically significant evidence of leukostasis can be considered, regardless of gestational stage. When patients are diagnosed with acute myeloid leukemia during the first trimester, pregnancy termination followed by conventional induction therapy (cytarabine/anthracycline) is recommended.
Those diagnosed later in pregnancy can receive conventional induction therapy, although this seems to be associated with increased risk of fetal growth restriction and even fetal loss. “Notably, neonates rarely experience neutropenia and cardiac impairment unless exposed to lipophilic idarubicin, which should not be used,” the panelists wrote.
When acute promyelocytic leukemia is diagnosed in the first trimester, pregnancy termination is recommended before initiating conventional ATRA-anthracycline therapy. Later in pregnancy, the regimen demonstrates low teratogenicity and can be used in women diagnosed after that stage. Arsenic treatment is highly teratogenic and is prohibited throughout gestation.
Acute lymphocytic leukemia (ALL) requires prophylactic CNS therapy, including methotrexate and L-asparaginase, which are fetotoxic. Methotrexate interferes with organogenesis and is prohibited before week 20 of gestation. L-asparaginase may increase the high risk for thromboembolic events attributed to the combination of pregnancy and malignancy.
Notably, tyrosine kinase inhibitors, essential for patients with Philadelphia chromosome–positive ALL, are teratogenic. Given these limitations, women diagnosed with ALL before 20 weeks’ gestation should undergo termination of the pregnancy and start conventional treatment. After week 20, conventional chemotherapy can be administered during pregnancy. Tyrosine kinase inhibitors can be initiated postpartum.
The guidelines also contain recommendations on diagnostic testing and radiotherapy, maternal supportive care, and perinatal and pediatric aspects of hematologic malignancies in pregnancy. An online appendix offers recommendations on the treatment of rare hematologic malignancies, including hairy cell leukemia, multiple myeloma, and myelodysplastic syndromes.
Dr. Lishner and nine of his coauthors had no financial relationships to disclose. Three coauthors received honoraria and research funding or are consultants to a wide variety of drug makers.
On Twitter @maryjodales
FROM JOURNAL OF CLINICAL ONCOLOGY
Elotuzumab approved for multiple myeloma
Elotuzumab (Empliciti) has been approved as combination therapy for individuals with relapsed or refractory multiple myeloma, the Food and Drug Administration announced Nov. 30.
Elotuzumab’s approval was based on a randomized, open-label clinical trial of 646 individuals with relapsed or refractory multiple myeloma. The multicenter trial compared progression-free survival (PFS) and overall response rate (ORR) for 321 individuals receiving lenalidomide (Revlimid) and dexamethasone to the PFS and ORR for 325 individuals receiving elotuzumab, lenalidomide, and dexamethasone.
For individuals receiving elotuzumab together with lenalidomide and dexamethasone, the median PFS of 19.4 months was significantly longer than the median 14.9 months of PFS seen in those receiving just lenalidomide and dexamethasone. For the elotuzumab-containing arm, the ORR was also significantly greater at 78.5%, compared with an ORR of 65.5% for the arm not receiving elotuzumab.
Slightly more patients in the elotuzumab-containing arm experienced serious adverse events, at 65.4% vs. 56.5% for those not receiving elotuzumab. Of the adverse reactions seen in more than 20% of patients, those that occurred more frequently in patients receiving elotuzumab included fatigue, diarrhea, fever, constipation, cough, peripheral neuropathy, upper respiratory tract infection, decreased appetite, and pneumonia.
Elotuzumab is a monoclonal antibody given intravenously; it can interfere with determination of complete response, and infusion reactions, infections, second primary malignancies, and hepatotoxicity may also occur.
Elotuzumab’s target is the surface antigen signaling lymphocyte activation molecule family 7 (SLAMF7). This protein is present on malignant cells in multiple myeloma and is also present on human immune cells such as natural killer cells and monocytes. The recommended dosing is 10 mg/kg intravenously weekly for 2 weeks, then every 2 weeks until disease progression or unacceptable toxicity occurs. Acetaminophen and histamine1 (H1) and H2 blockade should be given preadministration.
“The approval of elotuzumab marks another important milestone in the treatment of multiple myeloma in a short period of time,” Walter M. Capone, president and CEO of the Multiple Myeloma Research Foundation, said in a statement.
Today’s approval is the third in November for multiple myeloma therapies, following the Nov. 16 approval of the monoclonal antibody daratumumab (Darzalex) and the Nov. 20 approval of the oral proteasome inhibitor ixazomib (Ninlaro).
Elotuzumab received priority review by the FDA as a breakthrough drug and also has orphan drug designation. Elotuzumab will be marketed as Empliciti by Bristol-Myers Squibb.
On Twitter @karioakes
Elotuzumab (Empliciti) has been approved as combination therapy for individuals with relapsed or refractory multiple myeloma, the Food and Drug Administration announced Nov. 30.
Elotuzumab’s approval was based on a randomized, open-label clinical trial of 646 individuals with relapsed or refractory multiple myeloma. The multicenter trial compared progression-free survival (PFS) and overall response rate (ORR) for 321 individuals receiving lenalidomide (Revlimid) and dexamethasone to the PFS and ORR for 325 individuals receiving elotuzumab, lenalidomide, and dexamethasone.
For individuals receiving elotuzumab together with lenalidomide and dexamethasone, the median PFS of 19.4 months was significantly longer than the median 14.9 months of PFS seen in those receiving just lenalidomide and dexamethasone. For the elotuzumab-containing arm, the ORR was also significantly greater at 78.5%, compared with an ORR of 65.5% for the arm not receiving elotuzumab.
Slightly more patients in the elotuzumab-containing arm experienced serious adverse events, at 65.4% vs. 56.5% for those not receiving elotuzumab. Of the adverse reactions seen in more than 20% of patients, those that occurred more frequently in patients receiving elotuzumab included fatigue, diarrhea, fever, constipation, cough, peripheral neuropathy, upper respiratory tract infection, decreased appetite, and pneumonia.
Elotuzumab is a monoclonal antibody given intravenously; it can interfere with determination of complete response, and infusion reactions, infections, second primary malignancies, and hepatotoxicity may also occur.
Elotuzumab’s target is the surface antigen signaling lymphocyte activation molecule family 7 (SLAMF7). This protein is present on malignant cells in multiple myeloma and is also present on human immune cells such as natural killer cells and monocytes. The recommended dosing is 10 mg/kg intravenously weekly for 2 weeks, then every 2 weeks until disease progression or unacceptable toxicity occurs. Acetaminophen and histamine1 (H1) and H2 blockade should be given preadministration.
“The approval of elotuzumab marks another important milestone in the treatment of multiple myeloma in a short period of time,” Walter M. Capone, president and CEO of the Multiple Myeloma Research Foundation, said in a statement.
Today’s approval is the third in November for multiple myeloma therapies, following the Nov. 16 approval of the monoclonal antibody daratumumab (Darzalex) and the Nov. 20 approval of the oral proteasome inhibitor ixazomib (Ninlaro).
Elotuzumab received priority review by the FDA as a breakthrough drug and also has orphan drug designation. Elotuzumab will be marketed as Empliciti by Bristol-Myers Squibb.
On Twitter @karioakes
Elotuzumab (Empliciti) has been approved as combination therapy for individuals with relapsed or refractory multiple myeloma, the Food and Drug Administration announced Nov. 30.
Elotuzumab’s approval was based on a randomized, open-label clinical trial of 646 individuals with relapsed or refractory multiple myeloma. The multicenter trial compared progression-free survival (PFS) and overall response rate (ORR) for 321 individuals receiving lenalidomide (Revlimid) and dexamethasone to the PFS and ORR for 325 individuals receiving elotuzumab, lenalidomide, and dexamethasone.
For individuals receiving elotuzumab together with lenalidomide and dexamethasone, the median PFS of 19.4 months was significantly longer than the median 14.9 months of PFS seen in those receiving just lenalidomide and dexamethasone. For the elotuzumab-containing arm, the ORR was also significantly greater at 78.5%, compared with an ORR of 65.5% for the arm not receiving elotuzumab.
Slightly more patients in the elotuzumab-containing arm experienced serious adverse events, at 65.4% vs. 56.5% for those not receiving elotuzumab. Of the adverse reactions seen in more than 20% of patients, those that occurred more frequently in patients receiving elotuzumab included fatigue, diarrhea, fever, constipation, cough, peripheral neuropathy, upper respiratory tract infection, decreased appetite, and pneumonia.
Elotuzumab is a monoclonal antibody given intravenously; it can interfere with determination of complete response, and infusion reactions, infections, second primary malignancies, and hepatotoxicity may also occur.
Elotuzumab’s target is the surface antigen signaling lymphocyte activation molecule family 7 (SLAMF7). This protein is present on malignant cells in multiple myeloma and is also present on human immune cells such as natural killer cells and monocytes. The recommended dosing is 10 mg/kg intravenously weekly for 2 weeks, then every 2 weeks until disease progression or unacceptable toxicity occurs. Acetaminophen and histamine1 (H1) and H2 blockade should be given preadministration.
“The approval of elotuzumab marks another important milestone in the treatment of multiple myeloma in a short period of time,” Walter M. Capone, president and CEO of the Multiple Myeloma Research Foundation, said in a statement.
Today’s approval is the third in November for multiple myeloma therapies, following the Nov. 16 approval of the monoclonal antibody daratumumab (Darzalex) and the Nov. 20 approval of the oral proteasome inhibitor ixazomib (Ninlaro).
Elotuzumab received priority review by the FDA as a breakthrough drug and also has orphan drug designation. Elotuzumab will be marketed as Empliciti by Bristol-Myers Squibb.
On Twitter @karioakes
Trial of ofatumumab in FL stopped early

Photo courtesy of GSK
Genmab A/S said it is stopping a phase 3 trial of ofatumumab in follicular lymphoma (FL) after a planned interim analysis suggested the drug was unlikely to demonstrate superiority over rituximab.
The trial was designed to compare these 2 drugs as single-agent treatment in FL patients who had relapsed at least 6 months after completing treatment with a rituximab-containing regimen.
An interim analysis performed by an independent data monitoring committee indicated that, if the trial was to be completed as planned, it was unlikely that ofatumumab would improve progression-free survival (PFS) when compared to rituximab.
Genmab noted that this analysis did not reveal any new safety signals associated with ofatumumab, and no other ongoing trials of ofatumumab will be affected by the results of this analysis.
“The outcome of the interim analysis in this study is disappointing, as we had hoped to see superiority of ofatumumab,” said Jan van de Winkel, PhD, chief executive officer of Genmab. “The data from the study will now be prepared so that it can be presented at a future scientific conference.”
The goal of the study was to randomize up to 516 patients to receive ofatumumab (at 1000 mg) or rituximab (at 375 mg/m2) by intravenous infusion for 4 weekly doses.
Patients who had stable or responsive disease would then receive single infusions of ofatumumab or rituximab every 2 months for 4 additional doses—a total of 8 doses over 9 months. The primary endpoint of the study was PFS.
Past disappointments
This is not the first time ofatumumab has fallen short of expectations. Last year, the drug failed to meet the primary endpoint in two phase 3 trials.
In the OMB114242 trial, ofatumumab failed to improve PFS when compared to physicians’ choice in patients with bulky, fludarabine-refractory chronic lymphocytic leukemia (CLL).
In the ORCHARRD trial, ofatumumab plus chemotherapy failed to improve PFS when compared to rituximab plus chemotherapy in patients with relapsed or refractory diffuse large B-cell lymphoma.
About ofatumumab
Ofatumumab is a human monoclonal antibody designed to target the CD20 molecule found on the surface of CLL cells and normal B lymphocytes.
In the US, ofatumumab is approved for use in combination with chlorambucil to treat previously untreated patients with CLL for whom fludarabine-based therapy is considered inappropriate.
In the European Union (EU), ofatumumab is approved for use in combination with chlorambucil or bendamustine to treat patients with CLL who have not received prior therapy and who are not eligible for fludarabine-based therapy.
In more than 50 countries worldwide, including the US and EU member countries, ofatumumab is approved as monotherapy for the treatment of patients with CLL who are refractory after prior treatment with fludarabine and alemtuzumab.
Ofatumumab is marketed as Arzerra under a collaboration agreement between Genmab and Novartis (formerly GSK). ![]()

Photo courtesy of GSK
Genmab A/S said it is stopping a phase 3 trial of ofatumumab in follicular lymphoma (FL) after a planned interim analysis suggested the drug was unlikely to demonstrate superiority over rituximab.
The trial was designed to compare these 2 drugs as single-agent treatment in FL patients who had relapsed at least 6 months after completing treatment with a rituximab-containing regimen.
An interim analysis performed by an independent data monitoring committee indicated that, if the trial was to be completed as planned, it was unlikely that ofatumumab would improve progression-free survival (PFS) when compared to rituximab.
Genmab noted that this analysis did not reveal any new safety signals associated with ofatumumab, and no other ongoing trials of ofatumumab will be affected by the results of this analysis.
“The outcome of the interim analysis in this study is disappointing, as we had hoped to see superiority of ofatumumab,” said Jan van de Winkel, PhD, chief executive officer of Genmab. “The data from the study will now be prepared so that it can be presented at a future scientific conference.”
The goal of the study was to randomize up to 516 patients to receive ofatumumab (at 1000 mg) or rituximab (at 375 mg/m2) by intravenous infusion for 4 weekly doses.
Patients who had stable or responsive disease would then receive single infusions of ofatumumab or rituximab every 2 months for 4 additional doses—a total of 8 doses over 9 months. The primary endpoint of the study was PFS.
Past disappointments
This is not the first time ofatumumab has fallen short of expectations. Last year, the drug failed to meet the primary endpoint in two phase 3 trials.
In the OMB114242 trial, ofatumumab failed to improve PFS when compared to physicians’ choice in patients with bulky, fludarabine-refractory chronic lymphocytic leukemia (CLL).
In the ORCHARRD trial, ofatumumab plus chemotherapy failed to improve PFS when compared to rituximab plus chemotherapy in patients with relapsed or refractory diffuse large B-cell lymphoma.
About ofatumumab
Ofatumumab is a human monoclonal antibody designed to target the CD20 molecule found on the surface of CLL cells and normal B lymphocytes.
In the US, ofatumumab is approved for use in combination with chlorambucil to treat previously untreated patients with CLL for whom fludarabine-based therapy is considered inappropriate.
In the European Union (EU), ofatumumab is approved for use in combination with chlorambucil or bendamustine to treat patients with CLL who have not received prior therapy and who are not eligible for fludarabine-based therapy.
In more than 50 countries worldwide, including the US and EU member countries, ofatumumab is approved as monotherapy for the treatment of patients with CLL who are refractory after prior treatment with fludarabine and alemtuzumab.
Ofatumumab is marketed as Arzerra under a collaboration agreement between Genmab and Novartis (formerly GSK). ![]()

Photo courtesy of GSK
Genmab A/S said it is stopping a phase 3 trial of ofatumumab in follicular lymphoma (FL) after a planned interim analysis suggested the drug was unlikely to demonstrate superiority over rituximab.
The trial was designed to compare these 2 drugs as single-agent treatment in FL patients who had relapsed at least 6 months after completing treatment with a rituximab-containing regimen.
An interim analysis performed by an independent data monitoring committee indicated that, if the trial was to be completed as planned, it was unlikely that ofatumumab would improve progression-free survival (PFS) when compared to rituximab.
Genmab noted that this analysis did not reveal any new safety signals associated with ofatumumab, and no other ongoing trials of ofatumumab will be affected by the results of this analysis.
“The outcome of the interim analysis in this study is disappointing, as we had hoped to see superiority of ofatumumab,” said Jan van de Winkel, PhD, chief executive officer of Genmab. “The data from the study will now be prepared so that it can be presented at a future scientific conference.”
The goal of the study was to randomize up to 516 patients to receive ofatumumab (at 1000 mg) or rituximab (at 375 mg/m2) by intravenous infusion for 4 weekly doses.
Patients who had stable or responsive disease would then receive single infusions of ofatumumab or rituximab every 2 months for 4 additional doses—a total of 8 doses over 9 months. The primary endpoint of the study was PFS.
Past disappointments
This is not the first time ofatumumab has fallen short of expectations. Last year, the drug failed to meet the primary endpoint in two phase 3 trials.
In the OMB114242 trial, ofatumumab failed to improve PFS when compared to physicians’ choice in patients with bulky, fludarabine-refractory chronic lymphocytic leukemia (CLL).
In the ORCHARRD trial, ofatumumab plus chemotherapy failed to improve PFS when compared to rituximab plus chemotherapy in patients with relapsed or refractory diffuse large B-cell lymphoma.
About ofatumumab
Ofatumumab is a human monoclonal antibody designed to target the CD20 molecule found on the surface of CLL cells and normal B lymphocytes.
In the US, ofatumumab is approved for use in combination with chlorambucil to treat previously untreated patients with CLL for whom fludarabine-based therapy is considered inappropriate.
In the European Union (EU), ofatumumab is approved for use in combination with chlorambucil or bendamustine to treat patients with CLL who have not received prior therapy and who are not eligible for fludarabine-based therapy.
In more than 50 countries worldwide, including the US and EU member countries, ofatumumab is approved as monotherapy for the treatment of patients with CLL who are refractory after prior treatment with fludarabine and alemtuzumab.
Ofatumumab is marketed as Arzerra under a collaboration agreement between Genmab and Novartis (formerly GSK). ![]()
Template can help guide care for cancer survivors

for radiation therapy
Photo by Rhoda Baer
A new template can help standardize plans for long-term care of cancer survivors who have undergone radiation therapy (RT), according to the American Society for Radiation Oncology (ASTRO).
An ASTRO advisory committee created the template to coordinate post-treatment care for cancer survivors among primary care providers (PCPs) and oncology
specialists (radiation, medical, and surgical), as well as patients themselves.
Details on the template appear in Practical Radiation Oncology.
“Factors such as earlier detection of cancer, increasingly effective treatment options, and an aging population lead to a growing number of cancer survivors and, ultimately, a need to educate and empower these individuals for their ongoing care,” said ASTRO chair Bruce D. Minsky, MD.
“The ASTRO template is designed to foster better coordination of post-treatment care for cancer survivors, including greater clarity in the dialogue between radiation oncologists and PCPs for issues such as less common side effects that may appear well after treatment is complete.”
Many radiation oncologists may already provide their patients with post-treatment materials such as diagnosis and treatment summaries, contacts for ancillary services such as financial or nutritional counselling, and information on potential late treatment effects.
But the ASTRO template coordinates these components in a central, plain-language document.
It also enables practices to meet new accreditation requirements set by the American College of Surgeons Commission on Cancer (CoC).
In response to a 2006 recommendation from the Institutes of Medicine that cancer patients be provided with a survivorship care plan (SCP) following treatment, CoC issued a mandate that cancer programs provide SCPs for all curative cancer patients by 2019 to maintain accreditation.
ASTRO’s template includes both elements required by the CoC in SCPs—namely, a summary of past treatment and directions for future care.
The treatment summary outlines the survivor’s diagnosis and stage information; treatment details such as the site, dosage, and schedule of RT; and contact information for providers who delivered the treatment.
The plan for follow-up care covers anticipated toxicities from RT, expected course of recovery from treatment-related toxicities, possible functional and/or social limitations, recommendations for preventative measures and behaviors, cancer information resources, and referrals to supportive care providers.
“This 2-page template facilitates consistency in SCPs across the discipline and also reduces the time and effort required by providers to complete each individual plan,” said Ronald Chen, MD, of the University of North Carolina at Chapel Hill.
“The field of radiation oncology has a long tradition of creating treatment summaries for each patient, even before the Institute of Medicine recommended survivorship care plans in 2006. This radiation-oncology-specific template will serve a dual purpose as both a traditional radiation oncology treatment summary and a plan for survivorship care that meets CoC requirements, thus reducing the burden on radiation oncologists from having to create 2 documents for each patient.” ![]()

for radiation therapy
Photo by Rhoda Baer
A new template can help standardize plans for long-term care of cancer survivors who have undergone radiation therapy (RT), according to the American Society for Radiation Oncology (ASTRO).
An ASTRO advisory committee created the template to coordinate post-treatment care for cancer survivors among primary care providers (PCPs) and oncology
specialists (radiation, medical, and surgical), as well as patients themselves.
Details on the template appear in Practical Radiation Oncology.
“Factors such as earlier detection of cancer, increasingly effective treatment options, and an aging population lead to a growing number of cancer survivors and, ultimately, a need to educate and empower these individuals for their ongoing care,” said ASTRO chair Bruce D. Minsky, MD.
“The ASTRO template is designed to foster better coordination of post-treatment care for cancer survivors, including greater clarity in the dialogue between radiation oncologists and PCPs for issues such as less common side effects that may appear well after treatment is complete.”
Many radiation oncologists may already provide their patients with post-treatment materials such as diagnosis and treatment summaries, contacts for ancillary services such as financial or nutritional counselling, and information on potential late treatment effects.
But the ASTRO template coordinates these components in a central, plain-language document.
It also enables practices to meet new accreditation requirements set by the American College of Surgeons Commission on Cancer (CoC).
In response to a 2006 recommendation from the Institutes of Medicine that cancer patients be provided with a survivorship care plan (SCP) following treatment, CoC issued a mandate that cancer programs provide SCPs for all curative cancer patients by 2019 to maintain accreditation.
ASTRO’s template includes both elements required by the CoC in SCPs—namely, a summary of past treatment and directions for future care.
The treatment summary outlines the survivor’s diagnosis and stage information; treatment details such as the site, dosage, and schedule of RT; and contact information for providers who delivered the treatment.
The plan for follow-up care covers anticipated toxicities from RT, expected course of recovery from treatment-related toxicities, possible functional and/or social limitations, recommendations for preventative measures and behaviors, cancer information resources, and referrals to supportive care providers.
“This 2-page template facilitates consistency in SCPs across the discipline and also reduces the time and effort required by providers to complete each individual plan,” said Ronald Chen, MD, of the University of North Carolina at Chapel Hill.
“The field of radiation oncology has a long tradition of creating treatment summaries for each patient, even before the Institute of Medicine recommended survivorship care plans in 2006. This radiation-oncology-specific template will serve a dual purpose as both a traditional radiation oncology treatment summary and a plan for survivorship care that meets CoC requirements, thus reducing the burden on radiation oncologists from having to create 2 documents for each patient.” ![]()

for radiation therapy
Photo by Rhoda Baer
A new template can help standardize plans for long-term care of cancer survivors who have undergone radiation therapy (RT), according to the American Society for Radiation Oncology (ASTRO).
An ASTRO advisory committee created the template to coordinate post-treatment care for cancer survivors among primary care providers (PCPs) and oncology
specialists (radiation, medical, and surgical), as well as patients themselves.
Details on the template appear in Practical Radiation Oncology.
“Factors such as earlier detection of cancer, increasingly effective treatment options, and an aging population lead to a growing number of cancer survivors and, ultimately, a need to educate and empower these individuals for their ongoing care,” said ASTRO chair Bruce D. Minsky, MD.
“The ASTRO template is designed to foster better coordination of post-treatment care for cancer survivors, including greater clarity in the dialogue between radiation oncologists and PCPs for issues such as less common side effects that may appear well after treatment is complete.”
Many radiation oncologists may already provide their patients with post-treatment materials such as diagnosis and treatment summaries, contacts for ancillary services such as financial or nutritional counselling, and information on potential late treatment effects.
But the ASTRO template coordinates these components in a central, plain-language document.
It also enables practices to meet new accreditation requirements set by the American College of Surgeons Commission on Cancer (CoC).
In response to a 2006 recommendation from the Institutes of Medicine that cancer patients be provided with a survivorship care plan (SCP) following treatment, CoC issued a mandate that cancer programs provide SCPs for all curative cancer patients by 2019 to maintain accreditation.
ASTRO’s template includes both elements required by the CoC in SCPs—namely, a summary of past treatment and directions for future care.
The treatment summary outlines the survivor’s diagnosis and stage information; treatment details such as the site, dosage, and schedule of RT; and contact information for providers who delivered the treatment.
The plan for follow-up care covers anticipated toxicities from RT, expected course of recovery from treatment-related toxicities, possible functional and/or social limitations, recommendations for preventative measures and behaviors, cancer information resources, and referrals to supportive care providers.
“This 2-page template facilitates consistency in SCPs across the discipline and also reduces the time and effort required by providers to complete each individual plan,” said Ronald Chen, MD, of the University of North Carolina at Chapel Hill.
“The field of radiation oncology has a long tradition of creating treatment summaries for each patient, even before the Institute of Medicine recommended survivorship care plans in 2006. This radiation-oncology-specific template will serve a dual purpose as both a traditional radiation oncology treatment summary and a plan for survivorship care that meets CoC requirements, thus reducing the burden on radiation oncologists from having to create 2 documents for each patient.” ![]()