User login
NHL patients may have higher risk of second cancer

Photo courtesy of NIH
Compared to patients with other common cancers, patients with non-Hodgkin lymphoma (NHL) have a higher risk of developing a second, unrelated malignancy, according to a new study.
Researchers looked at data on more than 2.1 million patients with 10 of the most common cancers and found that patients with NHL or bladder cancer had the highest risk of developing a second malignancy.
The researchers reported these findings in Cancer.
For this study, Karim Chamie, MD, of the University of California, Los Angeles, and his colleagues looked at data from Surveillance, Epidemiology, and End Results database.
The team identified patients age 18 and older who were diagnosed with one of the 10 most common cancers—NHL, melanoma, and prostate, breast, lung, colon, rectal, bladder, uterine, and kidney cancers—between 1992 and 2008.
Of the 2,116,163 patients identified, 170,865 (8.1%) developed a second primary malignancy.
In multivariable analysis, patients with NHL or bladder cancer had the highest risk of developing a second malignancy.
The hazard ratios for patients with NHL were 2.70 for men and 2.88 for women. The hazard ratios for bladder cancer were 1.88 for men and 1.66 for women.
Lung cancer was a common second malignancy for both NHL and bladder cancer patients. NHL patients also tended to develop prostate and breast cancer.
Among patients with 2 incident cancers, 13% died of their initial cancer, and 55% died of their second primary malignancy. Lung cancer was the cause of death in 12% of the patients.
“As clinicians, we can become so focused on surveilling our patients to see if a primary cancer recurs that we sometimes may not be aware that patients can be at risk of developing a second, unrelated cancer,” Dr Chamie said.
He and his colleagues believe this study makes a case for monitoring cancer patients for second malignancies. ![]()

Photo courtesy of NIH
Compared to patients with other common cancers, patients with non-Hodgkin lymphoma (NHL) have a higher risk of developing a second, unrelated malignancy, according to a new study.
Researchers looked at data on more than 2.1 million patients with 10 of the most common cancers and found that patients with NHL or bladder cancer had the highest risk of developing a second malignancy.
The researchers reported these findings in Cancer.
For this study, Karim Chamie, MD, of the University of California, Los Angeles, and his colleagues looked at data from Surveillance, Epidemiology, and End Results database.
The team identified patients age 18 and older who were diagnosed with one of the 10 most common cancers—NHL, melanoma, and prostate, breast, lung, colon, rectal, bladder, uterine, and kidney cancers—between 1992 and 2008.
Of the 2,116,163 patients identified, 170,865 (8.1%) developed a second primary malignancy.
In multivariable analysis, patients with NHL or bladder cancer had the highest risk of developing a second malignancy.
The hazard ratios for patients with NHL were 2.70 for men and 2.88 for women. The hazard ratios for bladder cancer were 1.88 for men and 1.66 for women.
Lung cancer was a common second malignancy for both NHL and bladder cancer patients. NHL patients also tended to develop prostate and breast cancer.
Among patients with 2 incident cancers, 13% died of their initial cancer, and 55% died of their second primary malignancy. Lung cancer was the cause of death in 12% of the patients.
“As clinicians, we can become so focused on surveilling our patients to see if a primary cancer recurs that we sometimes may not be aware that patients can be at risk of developing a second, unrelated cancer,” Dr Chamie said.
He and his colleagues believe this study makes a case for monitoring cancer patients for second malignancies. ![]()

Photo courtesy of NIH
Compared to patients with other common cancers, patients with non-Hodgkin lymphoma (NHL) have a higher risk of developing a second, unrelated malignancy, according to a new study.
Researchers looked at data on more than 2.1 million patients with 10 of the most common cancers and found that patients with NHL or bladder cancer had the highest risk of developing a second malignancy.
The researchers reported these findings in Cancer.
For this study, Karim Chamie, MD, of the University of California, Los Angeles, and his colleagues looked at data from Surveillance, Epidemiology, and End Results database.
The team identified patients age 18 and older who were diagnosed with one of the 10 most common cancers—NHL, melanoma, and prostate, breast, lung, colon, rectal, bladder, uterine, and kidney cancers—between 1992 and 2008.
Of the 2,116,163 patients identified, 170,865 (8.1%) developed a second primary malignancy.
In multivariable analysis, patients with NHL or bladder cancer had the highest risk of developing a second malignancy.
The hazard ratios for patients with NHL were 2.70 for men and 2.88 for women. The hazard ratios for bladder cancer were 1.88 for men and 1.66 for women.
Lung cancer was a common second malignancy for both NHL and bladder cancer patients. NHL patients also tended to develop prostate and breast cancer.
Among patients with 2 incident cancers, 13% died of their initial cancer, and 55% died of their second primary malignancy. Lung cancer was the cause of death in 12% of the patients.
“As clinicians, we can become so focused on surveilling our patients to see if a primary cancer recurs that we sometimes may not be aware that patients can be at risk of developing a second, unrelated cancer,” Dr Chamie said.
He and his colleagues believe this study makes a case for monitoring cancer patients for second malignancies. ![]()
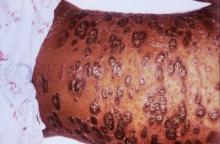
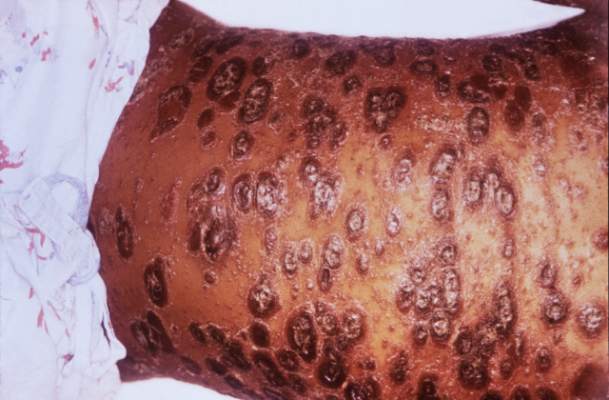
Severe psoriasis upped lymphoma risk in large cohort study
SCOTTSDALE, ARIZ. – Psoriasis of all severities was linked to a 3.5-fold increase in risk of cutaneous T-cell lymphoma, and severe psoriasis upped the associated risk of Hodgkin lymphoma by about 2.5 times, in a large, longitudinal, population-based cohort study.
Psoriasis also was tied to a smaller but statistically significant increase in the risk of non-Hodgkin lymphoma, said Zelma Chiesa Fuxench, MD, of the department of dermatology, the University of Pennsylvania, Philadelphia. Overall, lymphoma risk was highest in people with severe psoriasis, independent of traditional risk factors and exposure to immunosuppressive medications, Dr. Fuxench said at the annual meeting of the Society for Investigative Dermatology.
Psoriasis affects more than 125 million people worldwide, and severe cases are a major cause of cancer-related mortality. “Prior studies have suggested an increased risk of lymphoma in psoriasis patients, but it is unclear if this due to chronic inflammation, exposure to immunosuppressive therapies, or a combination of both factors,” Dr. Fuxench said.
To further explore these links, she and her associates analyzed electronic medical records from THIN (The Health Information Network), which includes about 12 million patients across the United Kingdom. Adults with psoriasis were matched to up to five nonpsoriatic controls based on date and clinic location. Patients who needed systemic medications or phototherapy were categorized as having severe psoriasis. The final dataset included more than 12,000 such patients, as well as 184,000 patients with mild psoriasis and more than 965,000 patients without psoriasis.
Psoriasis patients were younger and more likely to be overweight, male, and smoke and drink alcohol than patients without psoriasis, Dr. Fuxench said. Almost 80% of patients with severe disease had received systemic therapies, most often methotrexate (70% of systemic treatments) or cyclosporine (10%), while only 1% had received biologics.
Patients with severe psoriasis were more likely to be diagnosed with Hodgkin disease, non-Hodgkin lymphoma, and cutaneous T-cell lymphoma than were patients with mild psoriasis or controls. Over a median follow-up of 5.3 years, 34 patients with severe psoriasis were diagnosed with any type of lymphoma, for an incidence of 5.2 cases per 10,000 person-years (95% confidence interval, 3.7-7.3). In contrast, incidence rates for patients with mild psoriasis and controls were 3.3 and 3.2 cases per 10,000 person-years, respectively, Dr. Fuxench said.
In the multivariable analysis, patients with psoriasis were about 18% more likely to develop any type of lymphoma than were controls, an association that reached statistical significance (adjusted hazard ratio, 1.18; 95% CI, 1.06-1.31). Mild psoriasis increased lymphoma risk by 14%, and severe psoriasis upped it by about 83%, and both associations were statistically significant.
The increase in risk of non-Hodgkin lymphoma was 13% greater with mild psoriasis and 56% greater with severe disease, compared with controls, and these associations also reached statistical significance. Mild psoriasis was not linked to Hodgkin lymphoma, but patients with severe psoriasis were about 250% more likely to develop it than controls, with a trend toward statistical significance (aHR, 2.54; 95% CI, 0.94-6.87).
Finally, severe psoriasis was linked to a more than ninefold increase in risk of cutaneous T-cell lymphoma (aHR, 9.3; 95% CI, 4.1-21.4), while mild psoriasis was linked to about a threefold increase in risk.
“These results were robust in multiple sensitivity analyses, including analyses that excluded patients with rheumatoid arthritis, psoriatic arthritis, or a history of exposure to methotrexate, cyclosporine, or biologics,” Dr. Fuxench said. Future studies should explore the effect of treatment timing and selection on cancer risk, she added. “For those of us who care for these patients, we are increasingly using systemic agents that selectively target the immune system, and these questions will arise in clinics.”
The study’s design made it possible to pinpoint dates of diagnosis more effectively than investigators could estimate disease duration or confirm whether patients initially diagnosed with psoriasis actually had cutaneous T-cell lymphoma, Dr. Fuxench noted. “Ideally, we could have another cohort study of incident psoriasis with prospective follow-up, but lymphoma is so rare that there is currently not enough power [in the THIN database] to determine associations.”
The study was funded by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Fuxench disclosed unrestricted research funding from Pfizer outside the submitted work.
SCOTTSDALE, ARIZ. – Psoriasis of all severities was linked to a 3.5-fold increase in risk of cutaneous T-cell lymphoma, and severe psoriasis upped the associated risk of Hodgkin lymphoma by about 2.5 times, in a large, longitudinal, population-based cohort study.
Psoriasis also was tied to a smaller but statistically significant increase in the risk of non-Hodgkin lymphoma, said Zelma Chiesa Fuxench, MD, of the department of dermatology, the University of Pennsylvania, Philadelphia. Overall, lymphoma risk was highest in people with severe psoriasis, independent of traditional risk factors and exposure to immunosuppressive medications, Dr. Fuxench said at the annual meeting of the Society for Investigative Dermatology.
Psoriasis affects more than 125 million people worldwide, and severe cases are a major cause of cancer-related mortality. “Prior studies have suggested an increased risk of lymphoma in psoriasis patients, but it is unclear if this due to chronic inflammation, exposure to immunosuppressive therapies, or a combination of both factors,” Dr. Fuxench said.
To further explore these links, she and her associates analyzed electronic medical records from THIN (The Health Information Network), which includes about 12 million patients across the United Kingdom. Adults with psoriasis were matched to up to five nonpsoriatic controls based on date and clinic location. Patients who needed systemic medications or phototherapy were categorized as having severe psoriasis. The final dataset included more than 12,000 such patients, as well as 184,000 patients with mild psoriasis and more than 965,000 patients without psoriasis.
Psoriasis patients were younger and more likely to be overweight, male, and smoke and drink alcohol than patients without psoriasis, Dr. Fuxench said. Almost 80% of patients with severe disease had received systemic therapies, most often methotrexate (70% of systemic treatments) or cyclosporine (10%), while only 1% had received biologics.
Patients with severe psoriasis were more likely to be diagnosed with Hodgkin disease, non-Hodgkin lymphoma, and cutaneous T-cell lymphoma than were patients with mild psoriasis or controls. Over a median follow-up of 5.3 years, 34 patients with severe psoriasis were diagnosed with any type of lymphoma, for an incidence of 5.2 cases per 10,000 person-years (95% confidence interval, 3.7-7.3). In contrast, incidence rates for patients with mild psoriasis and controls were 3.3 and 3.2 cases per 10,000 person-years, respectively, Dr. Fuxench said.
In the multivariable analysis, patients with psoriasis were about 18% more likely to develop any type of lymphoma than were controls, an association that reached statistical significance (adjusted hazard ratio, 1.18; 95% CI, 1.06-1.31). Mild psoriasis increased lymphoma risk by 14%, and severe psoriasis upped it by about 83%, and both associations were statistically significant.
The increase in risk of non-Hodgkin lymphoma was 13% greater with mild psoriasis and 56% greater with severe disease, compared with controls, and these associations also reached statistical significance. Mild psoriasis was not linked to Hodgkin lymphoma, but patients with severe psoriasis were about 250% more likely to develop it than controls, with a trend toward statistical significance (aHR, 2.54; 95% CI, 0.94-6.87).
Finally, severe psoriasis was linked to a more than ninefold increase in risk of cutaneous T-cell lymphoma (aHR, 9.3; 95% CI, 4.1-21.4), while mild psoriasis was linked to about a threefold increase in risk.
“These results were robust in multiple sensitivity analyses, including analyses that excluded patients with rheumatoid arthritis, psoriatic arthritis, or a history of exposure to methotrexate, cyclosporine, or biologics,” Dr. Fuxench said. Future studies should explore the effect of treatment timing and selection on cancer risk, she added. “For those of us who care for these patients, we are increasingly using systemic agents that selectively target the immune system, and these questions will arise in clinics.”
The study’s design made it possible to pinpoint dates of diagnosis more effectively than investigators could estimate disease duration or confirm whether patients initially diagnosed with psoriasis actually had cutaneous T-cell lymphoma, Dr. Fuxench noted. “Ideally, we could have another cohort study of incident psoriasis with prospective follow-up, but lymphoma is so rare that there is currently not enough power [in the THIN database] to determine associations.”
The study was funded by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Fuxench disclosed unrestricted research funding from Pfizer outside the submitted work.
SCOTTSDALE, ARIZ. – Psoriasis of all severities was linked to a 3.5-fold increase in risk of cutaneous T-cell lymphoma, and severe psoriasis upped the associated risk of Hodgkin lymphoma by about 2.5 times, in a large, longitudinal, population-based cohort study.
Psoriasis also was tied to a smaller but statistically significant increase in the risk of non-Hodgkin lymphoma, said Zelma Chiesa Fuxench, MD, of the department of dermatology, the University of Pennsylvania, Philadelphia. Overall, lymphoma risk was highest in people with severe psoriasis, independent of traditional risk factors and exposure to immunosuppressive medications, Dr. Fuxench said at the annual meeting of the Society for Investigative Dermatology.
Psoriasis affects more than 125 million people worldwide, and severe cases are a major cause of cancer-related mortality. “Prior studies have suggested an increased risk of lymphoma in psoriasis patients, but it is unclear if this due to chronic inflammation, exposure to immunosuppressive therapies, or a combination of both factors,” Dr. Fuxench said.
To further explore these links, she and her associates analyzed electronic medical records from THIN (The Health Information Network), which includes about 12 million patients across the United Kingdom. Adults with psoriasis were matched to up to five nonpsoriatic controls based on date and clinic location. Patients who needed systemic medications or phototherapy were categorized as having severe psoriasis. The final dataset included more than 12,000 such patients, as well as 184,000 patients with mild psoriasis and more than 965,000 patients without psoriasis.
Psoriasis patients were younger and more likely to be overweight, male, and smoke and drink alcohol than patients without psoriasis, Dr. Fuxench said. Almost 80% of patients with severe disease had received systemic therapies, most often methotrexate (70% of systemic treatments) or cyclosporine (10%), while only 1% had received biologics.
Patients with severe psoriasis were more likely to be diagnosed with Hodgkin disease, non-Hodgkin lymphoma, and cutaneous T-cell lymphoma than were patients with mild psoriasis or controls. Over a median follow-up of 5.3 years, 34 patients with severe psoriasis were diagnosed with any type of lymphoma, for an incidence of 5.2 cases per 10,000 person-years (95% confidence interval, 3.7-7.3). In contrast, incidence rates for patients with mild psoriasis and controls were 3.3 and 3.2 cases per 10,000 person-years, respectively, Dr. Fuxench said.
In the multivariable analysis, patients with psoriasis were about 18% more likely to develop any type of lymphoma than were controls, an association that reached statistical significance (adjusted hazard ratio, 1.18; 95% CI, 1.06-1.31). Mild psoriasis increased lymphoma risk by 14%, and severe psoriasis upped it by about 83%, and both associations were statistically significant.
The increase in risk of non-Hodgkin lymphoma was 13% greater with mild psoriasis and 56% greater with severe disease, compared with controls, and these associations also reached statistical significance. Mild psoriasis was not linked to Hodgkin lymphoma, but patients with severe psoriasis were about 250% more likely to develop it than controls, with a trend toward statistical significance (aHR, 2.54; 95% CI, 0.94-6.87).
Finally, severe psoriasis was linked to a more than ninefold increase in risk of cutaneous T-cell lymphoma (aHR, 9.3; 95% CI, 4.1-21.4), while mild psoriasis was linked to about a threefold increase in risk.
“These results were robust in multiple sensitivity analyses, including analyses that excluded patients with rheumatoid arthritis, psoriatic arthritis, or a history of exposure to methotrexate, cyclosporine, or biologics,” Dr. Fuxench said. Future studies should explore the effect of treatment timing and selection on cancer risk, she added. “For those of us who care for these patients, we are increasingly using systemic agents that selectively target the immune system, and these questions will arise in clinics.”
The study’s design made it possible to pinpoint dates of diagnosis more effectively than investigators could estimate disease duration or confirm whether patients initially diagnosed with psoriasis actually had cutaneous T-cell lymphoma, Dr. Fuxench noted. “Ideally, we could have another cohort study of incident psoriasis with prospective follow-up, but lymphoma is so rare that there is currently not enough power [in the THIN database] to determine associations.”
The study was funded by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Fuxench disclosed unrestricted research funding from Pfizer outside the submitted work.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Psoriasis was identified as an independent risk factor for lymphoma, with the risk of lymphoma increasing with disease severity.
Major finding: The strongest association was between severe psoriasis and cutaneous T-cell lymphoma (aHR, 9.3; 95% CI, 4.1-21.4).
Data source: A population-based longitudinal cohort study of 12,198 patients with severe psoriasis, 184,870 patients with mild psoriasis, and 965,730 nonpsoriatic controls.
Disclosures: The study was funded by grants from the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Fuxench disclosed unrestricted research support from Pfizer outside the submitted work.
Pembrolizumab shows signs of efficacy in relapsed/refractory classical Hodgkin lymphoma
PD-1 checkpoint blockade via pembrolizumab is a potential option for classical Hodgkin’s lymphoma that progressed despite brentuximab vedotin therapy, based on a phase Ib, single-arm, open-label, industry-sponsored study of 31 patients.
After a median follow-up of 17 months, five (16%) patients achieved complete remission (90% confidence interval, 7%-31%), and 15 (48%) patients achieved partial remission (90% CI, 33%-64%), for an overall response rate of 65% (48%-79%). Furthermore, 70% of responses lasted at least 24 weeks, reported Philippe Armand, MD, of Dana-Farber Cancer Institute, Boston, and his associates.
“Since the time of study design, it has become apparent that complete responses are not commonly achieved with checkpoint blockade in solid tumors or hematologic malignancies,” they added. “Yet partial responses can be durable, suggesting that the achievement of complete response with checkpoint blockade is not necessary to derive significant clinical benefit.”
Pembrolizumab (Keytruda) is a humanized, highly selective IgG4 anti-PD-1 (programmed death-1) monoclonal antibody approved in the United States for patients with unresectable or metastatic melanoma or metastatic PDL-1 expressing non–small cell lung cancer. Tumor cells in classical Hodgkin lymphoma (HL) often overexpress PD-1 ligands, which “strongly suggests” that HL is PD-1 dependent, the researchers noted (J Clin Oncol. 2016 Jun 27. doi: 10.1200/JCO.2016.67.3467).
The researchers analyzed data for one group of patients within a phase Ib study of pembrolizumab in hematologic malignancies (KEYNOTE-013; NCT01953692). These 31 adults (median age, 32 years; 58% male) all had heavily pretreated classical HL – more than half had received at least five prior lines of therapy, and all had progressed on or after brentuximab vedotin therapy. Most patients (71%) also had received autologous stem cell transplantation. All 31 patients in the study received intravenous pembrolizumab (10 mg/kg) every other week.
Rates of progression-free survival were 69% at 24 weeks and 46% at 52 weeks, the researchers said. “Biomarker analyses demonstrated a high prevalence of PD-L1 and PD-L2 expression, treatment-induced expansion of T cells and natural killer cells, and activation of interferon-gamma, T-cell receptor, and expanded immune-related signaling pathways,” they reported. Those findings indicate that PD-1 blockade activates T-cell and IFN-gamma signaling pathways, which provides “a compelling rationale for the further development of PD-1 blockade in HL,” they concluded.
The most common treatment-related adverse effects were hypothyroidism (16%), diarrhea (16%), nausea (13%), and pneumonitis (10%). Five patients (16%) developed grade 3 treatment-related adverse events, including elevated hepatic transaminases, axillary pain, back pain, joint swelling, colitis, and nephrotic syndrome. Two patients stopped treatment because of adverse effects, but there were no grade 4 events or deaths related to treatment, and no treatment-induced cases of hepatitis, hypophysitis, or uveitis, the researchers noted.
Merck, maker of pembrolizumab, funded the study. Dr. Armand reported financial ties to Merck, Bristol-Myers Squibb, Infinity Pharmaceuticals, Sequenta, Tensha Therapeutics, and Sigma-Tau. Senior author Craig Moskowitz, MD, and five coinvestigators disclosed consulting or advisory relationships or employment with Merck.
PD-1 checkpoint blockade via pembrolizumab is a potential option for classical Hodgkin’s lymphoma that progressed despite brentuximab vedotin therapy, based on a phase Ib, single-arm, open-label, industry-sponsored study of 31 patients.
After a median follow-up of 17 months, five (16%) patients achieved complete remission (90% confidence interval, 7%-31%), and 15 (48%) patients achieved partial remission (90% CI, 33%-64%), for an overall response rate of 65% (48%-79%). Furthermore, 70% of responses lasted at least 24 weeks, reported Philippe Armand, MD, of Dana-Farber Cancer Institute, Boston, and his associates.
“Since the time of study design, it has become apparent that complete responses are not commonly achieved with checkpoint blockade in solid tumors or hematologic malignancies,” they added. “Yet partial responses can be durable, suggesting that the achievement of complete response with checkpoint blockade is not necessary to derive significant clinical benefit.”
Pembrolizumab (Keytruda) is a humanized, highly selective IgG4 anti-PD-1 (programmed death-1) monoclonal antibody approved in the United States for patients with unresectable or metastatic melanoma or metastatic PDL-1 expressing non–small cell lung cancer. Tumor cells in classical Hodgkin lymphoma (HL) often overexpress PD-1 ligands, which “strongly suggests” that HL is PD-1 dependent, the researchers noted (J Clin Oncol. 2016 Jun 27. doi: 10.1200/JCO.2016.67.3467).
The researchers analyzed data for one group of patients within a phase Ib study of pembrolizumab in hematologic malignancies (KEYNOTE-013; NCT01953692). These 31 adults (median age, 32 years; 58% male) all had heavily pretreated classical HL – more than half had received at least five prior lines of therapy, and all had progressed on or after brentuximab vedotin therapy. Most patients (71%) also had received autologous stem cell transplantation. All 31 patients in the study received intravenous pembrolizumab (10 mg/kg) every other week.
Rates of progression-free survival were 69% at 24 weeks and 46% at 52 weeks, the researchers said. “Biomarker analyses demonstrated a high prevalence of PD-L1 and PD-L2 expression, treatment-induced expansion of T cells and natural killer cells, and activation of interferon-gamma, T-cell receptor, and expanded immune-related signaling pathways,” they reported. Those findings indicate that PD-1 blockade activates T-cell and IFN-gamma signaling pathways, which provides “a compelling rationale for the further development of PD-1 blockade in HL,” they concluded.
The most common treatment-related adverse effects were hypothyroidism (16%), diarrhea (16%), nausea (13%), and pneumonitis (10%). Five patients (16%) developed grade 3 treatment-related adverse events, including elevated hepatic transaminases, axillary pain, back pain, joint swelling, colitis, and nephrotic syndrome. Two patients stopped treatment because of adverse effects, but there were no grade 4 events or deaths related to treatment, and no treatment-induced cases of hepatitis, hypophysitis, or uveitis, the researchers noted.
Merck, maker of pembrolizumab, funded the study. Dr. Armand reported financial ties to Merck, Bristol-Myers Squibb, Infinity Pharmaceuticals, Sequenta, Tensha Therapeutics, and Sigma-Tau. Senior author Craig Moskowitz, MD, and five coinvestigators disclosed consulting or advisory relationships or employment with Merck.
PD-1 checkpoint blockade via pembrolizumab is a potential option for classical Hodgkin’s lymphoma that progressed despite brentuximab vedotin therapy, based on a phase Ib, single-arm, open-label, industry-sponsored study of 31 patients.
After a median follow-up of 17 months, five (16%) patients achieved complete remission (90% confidence interval, 7%-31%), and 15 (48%) patients achieved partial remission (90% CI, 33%-64%), for an overall response rate of 65% (48%-79%). Furthermore, 70% of responses lasted at least 24 weeks, reported Philippe Armand, MD, of Dana-Farber Cancer Institute, Boston, and his associates.
“Since the time of study design, it has become apparent that complete responses are not commonly achieved with checkpoint blockade in solid tumors or hematologic malignancies,” they added. “Yet partial responses can be durable, suggesting that the achievement of complete response with checkpoint blockade is not necessary to derive significant clinical benefit.”
Pembrolizumab (Keytruda) is a humanized, highly selective IgG4 anti-PD-1 (programmed death-1) monoclonal antibody approved in the United States for patients with unresectable or metastatic melanoma or metastatic PDL-1 expressing non–small cell lung cancer. Tumor cells in classical Hodgkin lymphoma (HL) often overexpress PD-1 ligands, which “strongly suggests” that HL is PD-1 dependent, the researchers noted (J Clin Oncol. 2016 Jun 27. doi: 10.1200/JCO.2016.67.3467).
The researchers analyzed data for one group of patients within a phase Ib study of pembrolizumab in hematologic malignancies (KEYNOTE-013; NCT01953692). These 31 adults (median age, 32 years; 58% male) all had heavily pretreated classical HL – more than half had received at least five prior lines of therapy, and all had progressed on or after brentuximab vedotin therapy. Most patients (71%) also had received autologous stem cell transplantation. All 31 patients in the study received intravenous pembrolizumab (10 mg/kg) every other week.
Rates of progression-free survival were 69% at 24 weeks and 46% at 52 weeks, the researchers said. “Biomarker analyses demonstrated a high prevalence of PD-L1 and PD-L2 expression, treatment-induced expansion of T cells and natural killer cells, and activation of interferon-gamma, T-cell receptor, and expanded immune-related signaling pathways,” they reported. Those findings indicate that PD-1 blockade activates T-cell and IFN-gamma signaling pathways, which provides “a compelling rationale for the further development of PD-1 blockade in HL,” they concluded.
The most common treatment-related adverse effects were hypothyroidism (16%), diarrhea (16%), nausea (13%), and pneumonitis (10%). Five patients (16%) developed grade 3 treatment-related adverse events, including elevated hepatic transaminases, axillary pain, back pain, joint swelling, colitis, and nephrotic syndrome. Two patients stopped treatment because of adverse effects, but there were no grade 4 events or deaths related to treatment, and no treatment-induced cases of hepatitis, hypophysitis, or uveitis, the researchers noted.
Merck, maker of pembrolizumab, funded the study. Dr. Armand reported financial ties to Merck, Bristol-Myers Squibb, Infinity Pharmaceuticals, Sequenta, Tensha Therapeutics, and Sigma-Tau. Senior author Craig Moskowitz, MD, and five coinvestigators disclosed consulting or advisory relationships or employment with Merck.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Pembrolizumab is a potential treatment option for relapsed/refractory classical Hodgkin lymphoma.
Major finding: The complete response rate was 16%, the overall response rate was 65%, and 70% of responses lasted at least 24 weeks.
Data source: A single-arm, open-label, phase Ib study of pembrolizumab (10 mg/kg every other week) in 31 patients with heavily pretreated classical Hodgkin lymphoma that had progressed on or after brentuximab vedotin.
Disclosures: Merck, maker of pembrolizumab, funded the study. Dr. Armand reported financial ties to Merck, Bristol-Myers Squibb, Infinity Pharmaceuticals, Sequenta, Tensha Therapeutics, and Sigma-Tau. Senior author Craig Moskowitz, MD, and five coinvestigators disclosed consulting or advisory relationships or employment with Merck.
New type of CAR T cells can produce responses in NHL

Image courtesy of NIAID
Results of a phase 1 study suggest that chimeric antigen receptor T cells specific for the κ light chain (κ.CAR T cells) can produce responses in patients with relapsed or refractory B-cell malignancies, largely without side effects.
The therapy induced complete and partial responses in some patients with non-Hodgkin lymphoma (NHL), and it allowed other patients with chronic lymphocytic leukemia (CLL) or multiple myeloma (MM) to maintain stable disease.
There was 1 adverse event considered possibly related to the treatment.
The researchers reported these results in The Journal of Clinical Investigation.
The κ.CAR T cells were designed to recognize κ-restricted cells and spare normal B cells expressing the nontargeted λ light chain.
“We reasoned that targeting the light chain expressed by malignant B cells should efficiently kill tumor cells while sparing normal B cells expressing the other type of light chain,” said study author Carlos Ramos, MD, of Baylor College of Medicine in Houston, Texas.
He and his colleagues tested the κ.CAR T cells in 16 patients with relapsed or refractory κ+ NHL (n=7), CLL (n=2), or MM (n=7).
The team isolated T cells from these patients and modified the cells so they could target the κ light chain on the surface of malignant B cells. The modified T cells were infused back into the patients, and each patient was monitored for disease progression and side effects.
Eleven patients stopped receiving other treatments at least 4 weeks prior to T-cell infusion. Six patients without lymphopenia received cyclophosphamide at 12.5 mg/kg 4 days before κ.CAR T-cell infusion (0.2×108 to 2×108 κ.CAR T cells/m2).
“We found the treatment to be feasible and safe at all the dose levels studied,” Dr Ramos said.
One MM patient had grade 3 lymphopenia that was deemed possibly related to treatment, but none of the other adverse events were thought to result from the κ.CAR T cells.
Two patients with NHL achieved a complete response to treatment, 1 lasting more than 32 months and the other lasting 6 weeks. A third NHL patient had a partial response lasting 3 months, and a CLL patient had stable disease lasting 6 weeks.
Five of the 7 MM patients had stable disease, 3 lasting 6 weeks, 1 lasting 17 months, and 1 lasting 24 months.
“Our approach, although we are still optimizing it, offers a new possibility for patients in whom other treatments have not been successful,” Dr Ramos concluded. ![]()

Image courtesy of NIAID
Results of a phase 1 study suggest that chimeric antigen receptor T cells specific for the κ light chain (κ.CAR T cells) can produce responses in patients with relapsed or refractory B-cell malignancies, largely without side effects.
The therapy induced complete and partial responses in some patients with non-Hodgkin lymphoma (NHL), and it allowed other patients with chronic lymphocytic leukemia (CLL) or multiple myeloma (MM) to maintain stable disease.
There was 1 adverse event considered possibly related to the treatment.
The researchers reported these results in The Journal of Clinical Investigation.
The κ.CAR T cells were designed to recognize κ-restricted cells and spare normal B cells expressing the nontargeted λ light chain.
“We reasoned that targeting the light chain expressed by malignant B cells should efficiently kill tumor cells while sparing normal B cells expressing the other type of light chain,” said study author Carlos Ramos, MD, of Baylor College of Medicine in Houston, Texas.
He and his colleagues tested the κ.CAR T cells in 16 patients with relapsed or refractory κ+ NHL (n=7), CLL (n=2), or MM (n=7).
The team isolated T cells from these patients and modified the cells so they could target the κ light chain on the surface of malignant B cells. The modified T cells were infused back into the patients, and each patient was monitored for disease progression and side effects.
Eleven patients stopped receiving other treatments at least 4 weeks prior to T-cell infusion. Six patients without lymphopenia received cyclophosphamide at 12.5 mg/kg 4 days before κ.CAR T-cell infusion (0.2×108 to 2×108 κ.CAR T cells/m2).
“We found the treatment to be feasible and safe at all the dose levels studied,” Dr Ramos said.
One MM patient had grade 3 lymphopenia that was deemed possibly related to treatment, but none of the other adverse events were thought to result from the κ.CAR T cells.
Two patients with NHL achieved a complete response to treatment, 1 lasting more than 32 months and the other lasting 6 weeks. A third NHL patient had a partial response lasting 3 months, and a CLL patient had stable disease lasting 6 weeks.
Five of the 7 MM patients had stable disease, 3 lasting 6 weeks, 1 lasting 17 months, and 1 lasting 24 months.
“Our approach, although we are still optimizing it, offers a new possibility for patients in whom other treatments have not been successful,” Dr Ramos concluded. ![]()

Image courtesy of NIAID
Results of a phase 1 study suggest that chimeric antigen receptor T cells specific for the κ light chain (κ.CAR T cells) can produce responses in patients with relapsed or refractory B-cell malignancies, largely without side effects.
The therapy induced complete and partial responses in some patients with non-Hodgkin lymphoma (NHL), and it allowed other patients with chronic lymphocytic leukemia (CLL) or multiple myeloma (MM) to maintain stable disease.
There was 1 adverse event considered possibly related to the treatment.
The researchers reported these results in The Journal of Clinical Investigation.
The κ.CAR T cells were designed to recognize κ-restricted cells and spare normal B cells expressing the nontargeted λ light chain.
“We reasoned that targeting the light chain expressed by malignant B cells should efficiently kill tumor cells while sparing normal B cells expressing the other type of light chain,” said study author Carlos Ramos, MD, of Baylor College of Medicine in Houston, Texas.
He and his colleagues tested the κ.CAR T cells in 16 patients with relapsed or refractory κ+ NHL (n=7), CLL (n=2), or MM (n=7).
The team isolated T cells from these patients and modified the cells so they could target the κ light chain on the surface of malignant B cells. The modified T cells were infused back into the patients, and each patient was monitored for disease progression and side effects.
Eleven patients stopped receiving other treatments at least 4 weeks prior to T-cell infusion. Six patients without lymphopenia received cyclophosphamide at 12.5 mg/kg 4 days before κ.CAR T-cell infusion (0.2×108 to 2×108 κ.CAR T cells/m2).
“We found the treatment to be feasible and safe at all the dose levels studied,” Dr Ramos said.
One MM patient had grade 3 lymphopenia that was deemed possibly related to treatment, but none of the other adverse events were thought to result from the κ.CAR T cells.
Two patients with NHL achieved a complete response to treatment, 1 lasting more than 32 months and the other lasting 6 weeks. A third NHL patient had a partial response lasting 3 months, and a CLL patient had stable disease lasting 6 weeks.
Five of the 7 MM patients had stable disease, 3 lasting 6 weeks, 1 lasting 17 months, and 1 lasting 24 months.
“Our approach, although we are still optimizing it, offers a new possibility for patients in whom other treatments have not been successful,” Dr Ramos concluded. ![]()
Combo shows promise for treating DLBCL

The mTOR inhibitor everolimus may provide an additional benefit when combined with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) to treat patients with newly diagnosed diffuse large B-cell lymphoma (DLBCL), according to researchers.
The combination was considered well-tolerated in a phase 1 trial, and 96% of patients responded to the treatment.
“There is an unmet need to develop new therapies based on R-CHOP to try to increase the cure rate for diffuse large B-cell lymphoma,” said Patrick Johnston, MD, PhD, of the Mayo Clinic in Rochester, Minnesota.
“This pilot study suggests that adding mTOR inhibitors to standard therapy could improve outcomes, though it needs to be validated in a larger clinical trial.”
Results from this study were published in The Lancet Haematology.
Patients and treatment
Dr Johnston and his colleagues conducted this study in 24 previously untreated DLBCL patients. Their median age was 58.5 (range, 49.5-71.5), and 58% were male. Most patients had stage IV disease (54%), followed by stage II (25%), and stage III (21%). Five patients (21%) had bulky disease.
The patients received standard R-CHOP-21 (rituximab at 375 mg/m2, cyclophosphamide at 750 mg/m2, doxorubicin at 50 mg/m2, and vincristine at 1.4 mg/m2—all on day 1 of the 21-day cycle—as well as oral prednisone at 100 mg/m2 each day on days 1–5 of the cycle) for 6 cycles, with scheduled pegfilgrastim at 6 mg on day 2 of each cycle.
They also received everolimus at 10 mg/day on 2 different schedules. Nine patients were enrolled initially—3 given everolimus on days 1–10 and 6 receiving it on days 1–14. As there were no dose-limiting toxicities in these patients, another 15 patients went on to receive everolimus on days 1–14.
Results
The median follow-up was 21.5 months. Twenty-three patients (96%) achieved an overall response and a complete metabolic response as assessed by PET. The remaining patient withdrew consent during cycle 1 and achieved a complete response with R-CHOP alone.
The 12-month event-free survival rate was 100%. Nine patients had sufficient follow-up and were event-free at 24 months. At last follow-up (March 30, 2016), no deaths or relapses had occurred.
The most common adverse events were hematologic, such as grade 4 neutropenia (75%) and grade 3 febrile neutropenia (21%).
Three patients experienced “significant” toxicity, according to the researchers. One patient had a treatment delay of 12 days due to grade 3 hypokalemia, which was considered possibly related to everolimus.
A second patient had grade 4 sepsis that was possibly related to treatment, and a third patient had a treatment delay of 10 days due to grade 3 infection that was possibly related to everolimus.
Ten patients (42%) had their dose of everolimus reduced, 2 patients permanently discontinued the drug after cycles 3 and 4, respectively, and 2 patients omitted everolimus for 1 and 2 cycles, respectively, then resumed everolimus for subsequent cycles.
“This study is the first to integrate a P13K-mTOR agent with standard R-CHOP,” Dr Johnston said.
“The encouraging outcome results and toxicity profile of this new regimen, along with the worldwide availability of everolimus, make it potentially applicable to the large population of DLBCL patients.” ![]()

The mTOR inhibitor everolimus may provide an additional benefit when combined with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) to treat patients with newly diagnosed diffuse large B-cell lymphoma (DLBCL), according to researchers.
The combination was considered well-tolerated in a phase 1 trial, and 96% of patients responded to the treatment.
“There is an unmet need to develop new therapies based on R-CHOP to try to increase the cure rate for diffuse large B-cell lymphoma,” said Patrick Johnston, MD, PhD, of the Mayo Clinic in Rochester, Minnesota.
“This pilot study suggests that adding mTOR inhibitors to standard therapy could improve outcomes, though it needs to be validated in a larger clinical trial.”
Results from this study were published in The Lancet Haematology.
Patients and treatment
Dr Johnston and his colleagues conducted this study in 24 previously untreated DLBCL patients. Their median age was 58.5 (range, 49.5-71.5), and 58% were male. Most patients had stage IV disease (54%), followed by stage II (25%), and stage III (21%). Five patients (21%) had bulky disease.
The patients received standard R-CHOP-21 (rituximab at 375 mg/m2, cyclophosphamide at 750 mg/m2, doxorubicin at 50 mg/m2, and vincristine at 1.4 mg/m2—all on day 1 of the 21-day cycle—as well as oral prednisone at 100 mg/m2 each day on days 1–5 of the cycle) for 6 cycles, with scheduled pegfilgrastim at 6 mg on day 2 of each cycle.
They also received everolimus at 10 mg/day on 2 different schedules. Nine patients were enrolled initially—3 given everolimus on days 1–10 and 6 receiving it on days 1–14. As there were no dose-limiting toxicities in these patients, another 15 patients went on to receive everolimus on days 1–14.
Results
The median follow-up was 21.5 months. Twenty-three patients (96%) achieved an overall response and a complete metabolic response as assessed by PET. The remaining patient withdrew consent during cycle 1 and achieved a complete response with R-CHOP alone.
The 12-month event-free survival rate was 100%. Nine patients had sufficient follow-up and were event-free at 24 months. At last follow-up (March 30, 2016), no deaths or relapses had occurred.
The most common adverse events were hematologic, such as grade 4 neutropenia (75%) and grade 3 febrile neutropenia (21%).
Three patients experienced “significant” toxicity, according to the researchers. One patient had a treatment delay of 12 days due to grade 3 hypokalemia, which was considered possibly related to everolimus.
A second patient had grade 4 sepsis that was possibly related to treatment, and a third patient had a treatment delay of 10 days due to grade 3 infection that was possibly related to everolimus.
Ten patients (42%) had their dose of everolimus reduced, 2 patients permanently discontinued the drug after cycles 3 and 4, respectively, and 2 patients omitted everolimus for 1 and 2 cycles, respectively, then resumed everolimus for subsequent cycles.
“This study is the first to integrate a P13K-mTOR agent with standard R-CHOP,” Dr Johnston said.
“The encouraging outcome results and toxicity profile of this new regimen, along with the worldwide availability of everolimus, make it potentially applicable to the large population of DLBCL patients.” ![]()

The mTOR inhibitor everolimus may provide an additional benefit when combined with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) to treat patients with newly diagnosed diffuse large B-cell lymphoma (DLBCL), according to researchers.
The combination was considered well-tolerated in a phase 1 trial, and 96% of patients responded to the treatment.
“There is an unmet need to develop new therapies based on R-CHOP to try to increase the cure rate for diffuse large B-cell lymphoma,” said Patrick Johnston, MD, PhD, of the Mayo Clinic in Rochester, Minnesota.
“This pilot study suggests that adding mTOR inhibitors to standard therapy could improve outcomes, though it needs to be validated in a larger clinical trial.”
Results from this study were published in The Lancet Haematology.
Patients and treatment
Dr Johnston and his colleagues conducted this study in 24 previously untreated DLBCL patients. Their median age was 58.5 (range, 49.5-71.5), and 58% were male. Most patients had stage IV disease (54%), followed by stage II (25%), and stage III (21%). Five patients (21%) had bulky disease.
The patients received standard R-CHOP-21 (rituximab at 375 mg/m2, cyclophosphamide at 750 mg/m2, doxorubicin at 50 mg/m2, and vincristine at 1.4 mg/m2—all on day 1 of the 21-day cycle—as well as oral prednisone at 100 mg/m2 each day on days 1–5 of the cycle) for 6 cycles, with scheduled pegfilgrastim at 6 mg on day 2 of each cycle.
They also received everolimus at 10 mg/day on 2 different schedules. Nine patients were enrolled initially—3 given everolimus on days 1–10 and 6 receiving it on days 1–14. As there were no dose-limiting toxicities in these patients, another 15 patients went on to receive everolimus on days 1–14.
Results
The median follow-up was 21.5 months. Twenty-three patients (96%) achieved an overall response and a complete metabolic response as assessed by PET. The remaining patient withdrew consent during cycle 1 and achieved a complete response with R-CHOP alone.
The 12-month event-free survival rate was 100%. Nine patients had sufficient follow-up and were event-free at 24 months. At last follow-up (March 30, 2016), no deaths or relapses had occurred.
The most common adverse events were hematologic, such as grade 4 neutropenia (75%) and grade 3 febrile neutropenia (21%).
Three patients experienced “significant” toxicity, according to the researchers. One patient had a treatment delay of 12 days due to grade 3 hypokalemia, which was considered possibly related to everolimus.
A second patient had grade 4 sepsis that was possibly related to treatment, and a third patient had a treatment delay of 10 days due to grade 3 infection that was possibly related to everolimus.
Ten patients (42%) had their dose of everolimus reduced, 2 patients permanently discontinued the drug after cycles 3 and 4, respectively, and 2 patients omitted everolimus for 1 and 2 cycles, respectively, then resumed everolimus for subsequent cycles.
“This study is the first to integrate a P13K-mTOR agent with standard R-CHOP,” Dr Johnston said.
“The encouraging outcome results and toxicity profile of this new regimen, along with the worldwide availability of everolimus, make it potentially applicable to the large population of DLBCL patients.” ![]()
Adding obinutuzumab to bendamustine boosts progression-free survival in rituximab-refractory indolent non-Hodgkin lymphoma
Obinutuzumab and bendamustine followed by obinutuzumab maintenance therapy was superior to bendamustine monotherapy based on progression-free survival in rituximab-refractory patients with indolent non-Hodgkin lymphoma, based on a study published online in the Lancet Oncology.
After a median follow-up of 22 months in the obinutuzumab plus bendamustine group and 20 months in the bendamustine monotherapy group, progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% confidence interval, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range, 12.8-16.6; hazard ratio, 0.55; 95% CI 0.40-0.74; P = .0001). About two-thirds of the nearly 400 patients in both study arms had grade 3-5 adverse events.
The anti-CD20 monoclonal antibody obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment, wrote Laurie H. Sehn, MD, of the British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues.
In an open-label, randomized, phase III study called GADOLIN, patients with CD20-positive indolent non-Hodgkin lymphoma were stratified by indolent non-Hodgkin lymphoma subtype, rituximab-refractory type, number of previous therapies, and geographic region.
For the study, 194 patients were assigned to obinutuzumab plus bendamustine and 202 to bendamustine monotherapy. Trial participants received six 28-day cycles with either obinutuzumab plus bendamustine (obinutuzumab 1,000 mg on days 1, 8, and 15, cycle 1; and on day 1, cycles 2-6) plus bendamustine (90 mg/m2 per day on days 1 and 2, cycles 1-6) or bendamustine monotherapy (120 mg/m2 per day on days 1 and 2 of all cycles). Patients in the obinutuzumab plus bendamustine group whose disease did not progress received obinutuzumab maintenance therapy of 1,000 mg once every 2 months for up to 2 years.
Grade 3-5 adverse events occurred in 68% of 194 patients in the obinutuzumab plus bendamustine group and in 62% of 198 patients in the bendamustine monotherapy group. Grade 3 or worse neutropenia affected 33% of the obinutuzumab plus bendamustine group and 26% of the bendamustine monotherapy group. Other grade 3 or worse events included thrombocytopenia in 11% and 16%, anemia in 8% and 10%, and infusion-related reactions in 11% and 6%. Serious adverse events occurred in 38% in the obinutuzumab plus bendamustine group and in 33% in the bendamustine monotherapy group. Adverse events resulted in death in 6% of patients in each group.
The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or advisor to Genentech as well as several other drug companies.
On Twitter @maryjodales
Obinutuzumab and bendamustine followed by obinutuzumab maintenance therapy was superior to bendamustine monotherapy based on progression-free survival in rituximab-refractory patients with indolent non-Hodgkin lymphoma, based on a study published online in the Lancet Oncology.
After a median follow-up of 22 months in the obinutuzumab plus bendamustine group and 20 months in the bendamustine monotherapy group, progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% confidence interval, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range, 12.8-16.6; hazard ratio, 0.55; 95% CI 0.40-0.74; P = .0001). About two-thirds of the nearly 400 patients in both study arms had grade 3-5 adverse events.
The anti-CD20 monoclonal antibody obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment, wrote Laurie H. Sehn, MD, of the British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues.
In an open-label, randomized, phase III study called GADOLIN, patients with CD20-positive indolent non-Hodgkin lymphoma were stratified by indolent non-Hodgkin lymphoma subtype, rituximab-refractory type, number of previous therapies, and geographic region.
For the study, 194 patients were assigned to obinutuzumab plus bendamustine and 202 to bendamustine monotherapy. Trial participants received six 28-day cycles with either obinutuzumab plus bendamustine (obinutuzumab 1,000 mg on days 1, 8, and 15, cycle 1; and on day 1, cycles 2-6) plus bendamustine (90 mg/m2 per day on days 1 and 2, cycles 1-6) or bendamustine monotherapy (120 mg/m2 per day on days 1 and 2 of all cycles). Patients in the obinutuzumab plus bendamustine group whose disease did not progress received obinutuzumab maintenance therapy of 1,000 mg once every 2 months for up to 2 years.
Grade 3-5 adverse events occurred in 68% of 194 patients in the obinutuzumab plus bendamustine group and in 62% of 198 patients in the bendamustine monotherapy group. Grade 3 or worse neutropenia affected 33% of the obinutuzumab plus bendamustine group and 26% of the bendamustine monotherapy group. Other grade 3 or worse events included thrombocytopenia in 11% and 16%, anemia in 8% and 10%, and infusion-related reactions in 11% and 6%. Serious adverse events occurred in 38% in the obinutuzumab plus bendamustine group and in 33% in the bendamustine monotherapy group. Adverse events resulted in death in 6% of patients in each group.
The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or advisor to Genentech as well as several other drug companies.
On Twitter @maryjodales
Obinutuzumab and bendamustine followed by obinutuzumab maintenance therapy was superior to bendamustine monotherapy based on progression-free survival in rituximab-refractory patients with indolent non-Hodgkin lymphoma, based on a study published online in the Lancet Oncology.
After a median follow-up of 22 months in the obinutuzumab plus bendamustine group and 20 months in the bendamustine monotherapy group, progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% confidence interval, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range, 12.8-16.6; hazard ratio, 0.55; 95% CI 0.40-0.74; P = .0001). About two-thirds of the nearly 400 patients in both study arms had grade 3-5 adverse events.
The anti-CD20 monoclonal antibody obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment, wrote Laurie H. Sehn, MD, of the British Columbia Cancer Agency and the University of British Columbia, Vancouver, and her colleagues.
In an open-label, randomized, phase III study called GADOLIN, patients with CD20-positive indolent non-Hodgkin lymphoma were stratified by indolent non-Hodgkin lymphoma subtype, rituximab-refractory type, number of previous therapies, and geographic region.
For the study, 194 patients were assigned to obinutuzumab plus bendamustine and 202 to bendamustine monotherapy. Trial participants received six 28-day cycles with either obinutuzumab plus bendamustine (obinutuzumab 1,000 mg on days 1, 8, and 15, cycle 1; and on day 1, cycles 2-6) plus bendamustine (90 mg/m2 per day on days 1 and 2, cycles 1-6) or bendamustine monotherapy (120 mg/m2 per day on days 1 and 2 of all cycles). Patients in the obinutuzumab plus bendamustine group whose disease did not progress received obinutuzumab maintenance therapy of 1,000 mg once every 2 months for up to 2 years.
Grade 3-5 adverse events occurred in 68% of 194 patients in the obinutuzumab plus bendamustine group and in 62% of 198 patients in the bendamustine monotherapy group. Grade 3 or worse neutropenia affected 33% of the obinutuzumab plus bendamustine group and 26% of the bendamustine monotherapy group. Other grade 3 or worse events included thrombocytopenia in 11% and 16%, anemia in 8% and 10%, and infusion-related reactions in 11% and 6%. Serious adverse events occurred in 38% in the obinutuzumab plus bendamustine group and in 33% in the bendamustine monotherapy group. Adverse events resulted in death in 6% of patients in each group.
The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or advisor to Genentech as well as several other drug companies.
On Twitter @maryjodales
FROM THE LANCET ONCOLOGY
Key clinical point: Obinutuzumab is an option when patients with indolent non-Hodgkin lymphoma relapse or don’t achieve adequate disease control with rituximab-based treatment.
Major finding: Progression-free survival was significantly longer with obinutuzumab plus bendamustine (median not reached; 95% CI, 22.5 months – not estimable) than with bendamustine monotherapy (14.9 months, range,12.8-16.6 months; hazard ratio, 0.55; 95% CI, 0.40-0.74; P = ·0001).
Data source: An open-label, randomized, phase III study of 396 patients.
Disclosures: The study was funded by Hoffmann-La Roche. Genentech, the maker of obinutuzumab (Gazyva) in the United States, is a wholly owned member of the Roche Group. Dr. Sehn receives honoraria and is a consultant or adviser to Genentech as well as several other drug companies.
Team maps chromatin landscape in CLL

Researchers say they have performed the first large-scale analysis of the chromatin landscape in chronic lymphocytic leukemia (CLL).
And, in doing so, they have identified shared gene regulatory networks as well as heterogeneity between patients and CLL subtypes.
The group says this work should enable deeper investigation into chromatin regulation in CLL and the identification of therapeutically relevant mechanisms of disease.
The work has been published in Nature Communications.
The researchers performed chromatin accessibility mapping—via the assay for transposase-accessible chromatin using sequencing (ATAC-seq)—on 88 CLL samples from 55 patients.
For 10 of the samples, the team also established histone profiles using ChIPmentation for 3 histone marks (H3K4me1, H3K27ac, and H3K27me3) and transcriptome profiles using RNA sequencing.
The researchers then developed a bioinformatic method for linking the chromatin profiles to clinical annotations and molecular diagnostics data, and they analyzed gene regulatory networks that underlie the major disease subtypes of CLL.
The work revealed a “shared core” of regulatory regions in CLL patients as well as variations between the samples.
Furthermore, the chromatin profiles and gene regulatory networks accurately predicted IGHV mutation status and pinpointed differences between IGVH-mutated and IGVH-unmutated CLL.
“Our study has been able to dissect the variability that exists in the epigenome of CLL patients and helped to identify disease-specific changes, which will hopefully be informative for distinguishing disease subtypes or identifying suitable treatments,” said study author Jonathan Strefford, PhD, of the University of Southampton in the UK.
“Epigenetics can offer a useful doorway into ways of improving disease diagnosis and more personalized treatment choices for patients.” ![]()

Researchers say they have performed the first large-scale analysis of the chromatin landscape in chronic lymphocytic leukemia (CLL).
And, in doing so, they have identified shared gene regulatory networks as well as heterogeneity between patients and CLL subtypes.
The group says this work should enable deeper investigation into chromatin regulation in CLL and the identification of therapeutically relevant mechanisms of disease.
The work has been published in Nature Communications.
The researchers performed chromatin accessibility mapping—via the assay for transposase-accessible chromatin using sequencing (ATAC-seq)—on 88 CLL samples from 55 patients.
For 10 of the samples, the team also established histone profiles using ChIPmentation for 3 histone marks (H3K4me1, H3K27ac, and H3K27me3) and transcriptome profiles using RNA sequencing.
The researchers then developed a bioinformatic method for linking the chromatin profiles to clinical annotations and molecular diagnostics data, and they analyzed gene regulatory networks that underlie the major disease subtypes of CLL.
The work revealed a “shared core” of regulatory regions in CLL patients as well as variations between the samples.
Furthermore, the chromatin profiles and gene regulatory networks accurately predicted IGHV mutation status and pinpointed differences between IGVH-mutated and IGVH-unmutated CLL.
“Our study has been able to dissect the variability that exists in the epigenome of CLL patients and helped to identify disease-specific changes, which will hopefully be informative for distinguishing disease subtypes or identifying suitable treatments,” said study author Jonathan Strefford, PhD, of the University of Southampton in the UK.
“Epigenetics can offer a useful doorway into ways of improving disease diagnosis and more personalized treatment choices for patients.” ![]()

Researchers say they have performed the first large-scale analysis of the chromatin landscape in chronic lymphocytic leukemia (CLL).
And, in doing so, they have identified shared gene regulatory networks as well as heterogeneity between patients and CLL subtypes.
The group says this work should enable deeper investigation into chromatin regulation in CLL and the identification of therapeutically relevant mechanisms of disease.
The work has been published in Nature Communications.
The researchers performed chromatin accessibility mapping—via the assay for transposase-accessible chromatin using sequencing (ATAC-seq)—on 88 CLL samples from 55 patients.
For 10 of the samples, the team also established histone profiles using ChIPmentation for 3 histone marks (H3K4me1, H3K27ac, and H3K27me3) and transcriptome profiles using RNA sequencing.
The researchers then developed a bioinformatic method for linking the chromatin profiles to clinical annotations and molecular diagnostics data, and they analyzed gene regulatory networks that underlie the major disease subtypes of CLL.
The work revealed a “shared core” of regulatory regions in CLL patients as well as variations between the samples.
Furthermore, the chromatin profiles and gene regulatory networks accurately predicted IGHV mutation status and pinpointed differences between IGVH-mutated and IGVH-unmutated CLL.
“Our study has been able to dissect the variability that exists in the epigenome of CLL patients and helped to identify disease-specific changes, which will hopefully be informative for distinguishing disease subtypes or identifying suitable treatments,” said study author Jonathan Strefford, PhD, of the University of Southampton in the UK.
“Epigenetics can offer a useful doorway into ways of improving disease diagnosis and more personalized treatment choices for patients.” ![]()
Immunotherapy drugs linked to rheumatic diseases

Photo by Bill Branson
Several case reports have suggested that cancer patients taking the immunotherapy drugs nivolumab and ipilimumab may have a higher-than-normal risk of developing rheumatic diseases.
Between 2012 and 2016, 13 patients at the Johns Hopkins Kimmel Cancer Center who were taking one or both drugs developed inflammatory arthritis or sicca syndrome, a set of autoimmune conditions causing dry eyes and mouth.
The cases were described in Annals of Rheumatic Diseases.
Nivolumab and ipilimumab are both designed to turn off the molecular “checkpoints” some cancers—including lymphoma—use to evade the immune system. When the drugs work, they allow the immune system to detect and attack tumor cells. However, they also turn up the activity of the immune system as a whole and can therefore trigger immune-related side effects.
Clinical trials of ipilimumab and nivolumab have indicated that the drugs confer an increased risk of inflammatory bowel diseases, lung inflammation, autoimmune thyroid disease, and pituitary gland inflammation.
However, those trials were designed primarily to determine efficacy against cancer and not to fully examine all features of rheumatologic side effects, said Laura C. Cappelli, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
With this in mind, she and her colleagues decided to take a closer look at 13 adults (older than 18) who were treated at the Johns Hopkins Kimmel Cancer Center and reported rheumatologic symptoms after their treatment with nivolumab and/or ipilimumab.
Eight patients were taking both ipilimumab and nivolumab, and 5 were taking 1 of the 2 drugs. They were receiving the drugs to treat melanoma (n=6), non-small-cell lung cancer (n=5), small-cell lung cancer (n=1), and renal cell carcinoma (n=1).
Nine of the patients developed inflammatory arthritis—4 with synovitis confirmed via imaging and 4 with inflammatory synovial fluid—and the remaining 4 patients were diagnosed with sicca syndrome. Other immune-related adverse events included pneumonitis, colitis, interstitial nephritis, and thyroiditis.
The researchers said this is the largest published case series showing a link between checkpoint inhibitors and rheumatic diseases.
The patients described in this case report make up about 1.3% of all patients treated with the drugs—singly or in combination—at The Johns Hopkins Hospital from 2012 to 2016. However, the researchers believe that rate is likely an underestimation of how common rheumatic diseases are in patients taking immune checkpoint inhibitors.
“We keep having referrals coming in from our oncologists as more patients are treated with these drugs,” said Clifton Bingham, MD, of the Johns Hopkins University School of Medicine.
“In particular, as more patients are treated with combinations of multiple immunotherapies, we expect the rate to go up.”
Dr Cappelli said she wants the case report to raise awareness among patients and clinicians that rheumatologic side effects may occur with checkpoint inhibitors.
“It is important when weighing the risk-benefit ratio of prescribing these drugs,” she said. “And it’s important for people to be on the lookout for symptoms so they can see a rheumatologist early in an effort to prevent or limit joint damage.”
Drs Cappelli and Bingham and their colleagues are planning further collaboration with Johns Hopkins oncologists to better track the incidence of rheumatic disease in patients taking immunotherapy drugs and determine whether any particular characteristics put cancer patients at higher risk of such complications. ![]()

Photo by Bill Branson
Several case reports have suggested that cancer patients taking the immunotherapy drugs nivolumab and ipilimumab may have a higher-than-normal risk of developing rheumatic diseases.
Between 2012 and 2016, 13 patients at the Johns Hopkins Kimmel Cancer Center who were taking one or both drugs developed inflammatory arthritis or sicca syndrome, a set of autoimmune conditions causing dry eyes and mouth.
The cases were described in Annals of Rheumatic Diseases.
Nivolumab and ipilimumab are both designed to turn off the molecular “checkpoints” some cancers—including lymphoma—use to evade the immune system. When the drugs work, they allow the immune system to detect and attack tumor cells. However, they also turn up the activity of the immune system as a whole and can therefore trigger immune-related side effects.
Clinical trials of ipilimumab and nivolumab have indicated that the drugs confer an increased risk of inflammatory bowel diseases, lung inflammation, autoimmune thyroid disease, and pituitary gland inflammation.
However, those trials were designed primarily to determine efficacy against cancer and not to fully examine all features of rheumatologic side effects, said Laura C. Cappelli, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
With this in mind, she and her colleagues decided to take a closer look at 13 adults (older than 18) who were treated at the Johns Hopkins Kimmel Cancer Center and reported rheumatologic symptoms after their treatment with nivolumab and/or ipilimumab.
Eight patients were taking both ipilimumab and nivolumab, and 5 were taking 1 of the 2 drugs. They were receiving the drugs to treat melanoma (n=6), non-small-cell lung cancer (n=5), small-cell lung cancer (n=1), and renal cell carcinoma (n=1).
Nine of the patients developed inflammatory arthritis—4 with synovitis confirmed via imaging and 4 with inflammatory synovial fluid—and the remaining 4 patients were diagnosed with sicca syndrome. Other immune-related adverse events included pneumonitis, colitis, interstitial nephritis, and thyroiditis.
The researchers said this is the largest published case series showing a link between checkpoint inhibitors and rheumatic diseases.
The patients described in this case report make up about 1.3% of all patients treated with the drugs—singly or in combination—at The Johns Hopkins Hospital from 2012 to 2016. However, the researchers believe that rate is likely an underestimation of how common rheumatic diseases are in patients taking immune checkpoint inhibitors.
“We keep having referrals coming in from our oncologists as more patients are treated with these drugs,” said Clifton Bingham, MD, of the Johns Hopkins University School of Medicine.
“In particular, as more patients are treated with combinations of multiple immunotherapies, we expect the rate to go up.”
Dr Cappelli said she wants the case report to raise awareness among patients and clinicians that rheumatologic side effects may occur with checkpoint inhibitors.
“It is important when weighing the risk-benefit ratio of prescribing these drugs,” she said. “And it’s important for people to be on the lookout for symptoms so they can see a rheumatologist early in an effort to prevent or limit joint damage.”
Drs Cappelli and Bingham and their colleagues are planning further collaboration with Johns Hopkins oncologists to better track the incidence of rheumatic disease in patients taking immunotherapy drugs and determine whether any particular characteristics put cancer patients at higher risk of such complications. ![]()

Photo by Bill Branson
Several case reports have suggested that cancer patients taking the immunotherapy drugs nivolumab and ipilimumab may have a higher-than-normal risk of developing rheumatic diseases.
Between 2012 and 2016, 13 patients at the Johns Hopkins Kimmel Cancer Center who were taking one or both drugs developed inflammatory arthritis or sicca syndrome, a set of autoimmune conditions causing dry eyes and mouth.
The cases were described in Annals of Rheumatic Diseases.
Nivolumab and ipilimumab are both designed to turn off the molecular “checkpoints” some cancers—including lymphoma—use to evade the immune system. When the drugs work, they allow the immune system to detect and attack tumor cells. However, they also turn up the activity of the immune system as a whole and can therefore trigger immune-related side effects.
Clinical trials of ipilimumab and nivolumab have indicated that the drugs confer an increased risk of inflammatory bowel diseases, lung inflammation, autoimmune thyroid disease, and pituitary gland inflammation.
However, those trials were designed primarily to determine efficacy against cancer and not to fully examine all features of rheumatologic side effects, said Laura C. Cappelli, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
With this in mind, she and her colleagues decided to take a closer look at 13 adults (older than 18) who were treated at the Johns Hopkins Kimmel Cancer Center and reported rheumatologic symptoms after their treatment with nivolumab and/or ipilimumab.
Eight patients were taking both ipilimumab and nivolumab, and 5 were taking 1 of the 2 drugs. They were receiving the drugs to treat melanoma (n=6), non-small-cell lung cancer (n=5), small-cell lung cancer (n=1), and renal cell carcinoma (n=1).
Nine of the patients developed inflammatory arthritis—4 with synovitis confirmed via imaging and 4 with inflammatory synovial fluid—and the remaining 4 patients were diagnosed with sicca syndrome. Other immune-related adverse events included pneumonitis, colitis, interstitial nephritis, and thyroiditis.
The researchers said this is the largest published case series showing a link between checkpoint inhibitors and rheumatic diseases.
The patients described in this case report make up about 1.3% of all patients treated with the drugs—singly or in combination—at The Johns Hopkins Hospital from 2012 to 2016. However, the researchers believe that rate is likely an underestimation of how common rheumatic diseases are in patients taking immune checkpoint inhibitors.
“We keep having referrals coming in from our oncologists as more patients are treated with these drugs,” said Clifton Bingham, MD, of the Johns Hopkins University School of Medicine.
“In particular, as more patients are treated with combinations of multiple immunotherapies, we expect the rate to go up.”
Dr Cappelli said she wants the case report to raise awareness among patients and clinicians that rheumatologic side effects may occur with checkpoint inhibitors.
“It is important when weighing the risk-benefit ratio of prescribing these drugs,” she said. “And it’s important for people to be on the lookout for symptoms so they can see a rheumatologist early in an effort to prevent or limit joint damage.”
Drs Cappelli and Bingham and their colleagues are planning further collaboration with Johns Hopkins oncologists to better track the incidence of rheumatic disease in patients taking immunotherapy drugs and determine whether any particular characteristics put cancer patients at higher risk of such complications. ![]()
CHMP rejects ofatumumab as maintenance

Photo courtesy of GSK
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended against expanding the approved indication for ofatumumab (Arzerra).
Novartis, which is developing ofatumumab in cooperation with Genmab, had submitted an application requesting that ofatumumab be authorized as maintenance therapy for patients with relapsed chronic lymphocytic leukemia (CLL).
But the CHMP has advised the European Commission (EC) not to grant this authorization.
The CHMP noted that, in the phase 3 PROLONG trial, ofatumumab maintenance improved progression-free survival (PFS) in CLL patients.
However, the committee said the importance of this improvement is not clear because the PFS results were not supported by other measures, such as overall survival or a significant improvement in patients’ quality of life.
The CHMP also said the use of ofatumumab for maintenance treatment should be seen in the context of its side effects. Common side effects of ofatumumab in the PROLONG trial were infusion reactions, neutropenia, and upper respiratory tract infections.
In the end, the CHMP decided that the PROLONG data were not sufficient to conclude that maintenance treatment with ofatumumab is of more benefit than no treatment. So the committee recommended against expanding the drug’s marketing authorization.
This decision does not have any impact on ongoing clinical trials with ofatumumab.
About ofatumumab
Ofatumumab has been authorized for use in the European Union since April 2010.
The EC first granted ofatumumab conditional approval to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Then, in 2014, the EC granted ofatumumab conditional approval for use in combination with chlorambucil or bendamustine in CLL patients who have not received prior therapy and are not eligible for fludarabine-based therapy.
Ofatumumab received conditional approval because the drug’s benefits appear to outweigh the risks it poses in the aforementioned indications. Ofatumumab will not receive full approval until the drug’s developers submit results of additional research to the EC.
About the PROLONG trial
The PROLONG trial was designed to compare ofatumumab maintenance to no further treatment in patients with a complete or partial response after second- or third-line treatment for CLL. Interim results of the study were presented at ASH 2014.
These results—in 474 patients—suggested that ofatumumab can significantly improve PFS. The median PFS was about 29 months in patients who received ofatumumab and about 15 months for patients who did not receive maintenance therapy (P<0.0001).
There was no significant difference in the median overall survival, which was not reached in either treatment arm.
The researchers said there were no unexpected safety findings. The most common adverse events (≥10%) were infusion reactions, neutropenia, and upper respiratory tract infection. ![]()

Photo courtesy of GSK
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended against expanding the approved indication for ofatumumab (Arzerra).
Novartis, which is developing ofatumumab in cooperation with Genmab, had submitted an application requesting that ofatumumab be authorized as maintenance therapy for patients with relapsed chronic lymphocytic leukemia (CLL).
But the CHMP has advised the European Commission (EC) not to grant this authorization.
The CHMP noted that, in the phase 3 PROLONG trial, ofatumumab maintenance improved progression-free survival (PFS) in CLL patients.
However, the committee said the importance of this improvement is not clear because the PFS results were not supported by other measures, such as overall survival or a significant improvement in patients’ quality of life.
The CHMP also said the use of ofatumumab for maintenance treatment should be seen in the context of its side effects. Common side effects of ofatumumab in the PROLONG trial were infusion reactions, neutropenia, and upper respiratory tract infections.
In the end, the CHMP decided that the PROLONG data were not sufficient to conclude that maintenance treatment with ofatumumab is of more benefit than no treatment. So the committee recommended against expanding the drug’s marketing authorization.
This decision does not have any impact on ongoing clinical trials with ofatumumab.
About ofatumumab
Ofatumumab has been authorized for use in the European Union since April 2010.
The EC first granted ofatumumab conditional approval to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Then, in 2014, the EC granted ofatumumab conditional approval for use in combination with chlorambucil or bendamustine in CLL patients who have not received prior therapy and are not eligible for fludarabine-based therapy.
Ofatumumab received conditional approval because the drug’s benefits appear to outweigh the risks it poses in the aforementioned indications. Ofatumumab will not receive full approval until the drug’s developers submit results of additional research to the EC.
About the PROLONG trial
The PROLONG trial was designed to compare ofatumumab maintenance to no further treatment in patients with a complete or partial response after second- or third-line treatment for CLL. Interim results of the study were presented at ASH 2014.
These results—in 474 patients—suggested that ofatumumab can significantly improve PFS. The median PFS was about 29 months in patients who received ofatumumab and about 15 months for patients who did not receive maintenance therapy (P<0.0001).
There was no significant difference in the median overall survival, which was not reached in either treatment arm.
The researchers said there were no unexpected safety findings. The most common adverse events (≥10%) were infusion reactions, neutropenia, and upper respiratory tract infection. ![]()

Photo courtesy of GSK
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended against expanding the approved indication for ofatumumab (Arzerra).
Novartis, which is developing ofatumumab in cooperation with Genmab, had submitted an application requesting that ofatumumab be authorized as maintenance therapy for patients with relapsed chronic lymphocytic leukemia (CLL).
But the CHMP has advised the European Commission (EC) not to grant this authorization.
The CHMP noted that, in the phase 3 PROLONG trial, ofatumumab maintenance improved progression-free survival (PFS) in CLL patients.
However, the committee said the importance of this improvement is not clear because the PFS results were not supported by other measures, such as overall survival or a significant improvement in patients’ quality of life.
The CHMP also said the use of ofatumumab for maintenance treatment should be seen in the context of its side effects. Common side effects of ofatumumab in the PROLONG trial were infusion reactions, neutropenia, and upper respiratory tract infections.
In the end, the CHMP decided that the PROLONG data were not sufficient to conclude that maintenance treatment with ofatumumab is of more benefit than no treatment. So the committee recommended against expanding the drug’s marketing authorization.
This decision does not have any impact on ongoing clinical trials with ofatumumab.
About ofatumumab
Ofatumumab has been authorized for use in the European Union since April 2010.
The EC first granted ofatumumab conditional approval to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Then, in 2014, the EC granted ofatumumab conditional approval for use in combination with chlorambucil or bendamustine in CLL patients who have not received prior therapy and are not eligible for fludarabine-based therapy.
Ofatumumab received conditional approval because the drug’s benefits appear to outweigh the risks it poses in the aforementioned indications. Ofatumumab will not receive full approval until the drug’s developers submit results of additional research to the EC.
About the PROLONG trial
The PROLONG trial was designed to compare ofatumumab maintenance to no further treatment in patients with a complete or partial response after second- or third-line treatment for CLL. Interim results of the study were presented at ASH 2014.
These results—in 474 patients—suggested that ofatumumab can significantly improve PFS. The median PFS was about 29 months in patients who received ofatumumab and about 15 months for patients who did not receive maintenance therapy (P<0.0001).
There was no significant difference in the median overall survival, which was not reached in either treatment arm.
The researchers said there were no unexpected safety findings. The most common adverse events (≥10%) were infusion reactions, neutropenia, and upper respiratory tract infection. ![]()
Interim PET-CT can spare HL patients intensive chemo
The use of interim PET-CT scans can spare some advanced Hodgkin lymphoma (HL) patients the toxicity associated with bleomycin, according to researchers.
The team found that patients with negative PET-CT scans after 2 cycles of ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) could go on to receive AVD (doxorubicin, vinblastine, and dacarbazine) without experiencing a significant decrease in progression-free survival (PFS) or overall survival (OS).
Peter Johnson, MD, of the University of Southampton in the UK, and his colleagues reported these findings in NEJM.
“The good news is that the majority of people diagnosed with Hodgkin lymphoma can be cured,” Dr Johnson said. “In this trial, more than 95% of patients are alive after 3 years.”
“But we worry about the long-term side effects from the treatments we use. As we’ve done in this trial, personalizing treatment based on how well it works is a major development for patients with Hodgkin lymphoma and sets a new standard of care.”
Patients and treatment
For this study, Dr Johnson and his colleagues enrolled 1214 patients with newly diagnosed, advanced, classic HL. The patients’ median age was 33 (range, 18 to 79), and 54.5% were male. More patients had stage II disease (41.6%) than stage III (30.2%) or IV (28.3%).
A total of 1119 patients underwent a baseline PET-CT scan, received 2 cycles of ABVD, and underwent an interim PET-CT scan.
The patients with negative interim scans were then randomized to continue treatment with ABVD (n=470) or with AVD (n=465) in cycles 3 through 6.
Patients with positive interim scans (n=182) went on to receive BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone, n=172), salvage treatments (n=6), or ABVD (n=4).
Results
The study’s primary outcome was the difference in 3-year PFS between the randomized groups of PET-CT-negative patients.
With a median follow-up of 41 months, the 3-year PFS was 85.7% in the ABVD group and 84.4% in the AVD group. The hazard ratio was 1.13 (95% CI, 0.81 to 1.57; P=0.48) in the intention-to-treat analysis and 1.10 (95% CI, 0.79 to 1.53; P=0.58) in the per-protocol analysis.
The absolute difference in 3-year PFS (ABVD minus AVD) was 1.6 percentage points (95% CI, −3.2 to 5.3).
The OS rates were 97.2% in the ABVD group and 97.6% in the AVD group. The hazard ratio in the intention-to-treat analysis was 0.90 (95% CI, 0.47 to 1.74; P=0.76).
Patients in the ABVD group had a significantly higher rate of clinical adverse events than patients in the AVD group—31% and 21%, respectively (P<0.005).
Patients in the ABVD group also had significantly (P<0.05) higher rates of fatigue (3% vs 1%), febrile neutropenia (5% vs 2%), pulmonary/upper respiratory events (3% vs 1%), and dyspnea (2% vs <0.5%). But patients in the AVD group had a significantly higher rate of thrombocytopenia (3% vs 1%).
For patients who had positive interim PET-CT scans, the 3-year PFS was 67.5%, and the OS was 87.8%. Among the 172 patients who went on to receive BEACOPP, 74.4% had negative findings on a third PET-CT scan.
Overall, 62 patients died during the trial—24 from HL. So, for the entire study cohort, the 3-year PFS was 82.6%, and the OS was 95.8%. ![]()

The use of interim PET-CT scans can spare some advanced Hodgkin lymphoma (HL) patients the toxicity associated with bleomycin, according to researchers.
The team found that patients with negative PET-CT scans after 2 cycles of ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) could go on to receive AVD (doxorubicin, vinblastine, and dacarbazine) without experiencing a significant decrease in progression-free survival (PFS) or overall survival (OS).
Peter Johnson, MD, of the University of Southampton in the UK, and his colleagues reported these findings in NEJM.
“The good news is that the majority of people diagnosed with Hodgkin lymphoma can be cured,” Dr Johnson said. “In this trial, more than 95% of patients are alive after 3 years.”
“But we worry about the long-term side effects from the treatments we use. As we’ve done in this trial, personalizing treatment based on how well it works is a major development for patients with Hodgkin lymphoma and sets a new standard of care.”
Patients and treatment
For this study, Dr Johnson and his colleagues enrolled 1214 patients with newly diagnosed, advanced, classic HL. The patients’ median age was 33 (range, 18 to 79), and 54.5% were male. More patients had stage II disease (41.6%) than stage III (30.2%) or IV (28.3%).
A total of 1119 patients underwent a baseline PET-CT scan, received 2 cycles of ABVD, and underwent an interim PET-CT scan.
The patients with negative interim scans were then randomized to continue treatment with ABVD (n=470) or with AVD (n=465) in cycles 3 through 6.
Patients with positive interim scans (n=182) went on to receive BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone, n=172), salvage treatments (n=6), or ABVD (n=4).
Results
The study’s primary outcome was the difference in 3-year PFS between the randomized groups of PET-CT-negative patients.
With a median follow-up of 41 months, the 3-year PFS was 85.7% in the ABVD group and 84.4% in the AVD group. The hazard ratio was 1.13 (95% CI, 0.81 to 1.57; P=0.48) in the intention-to-treat analysis and 1.10 (95% CI, 0.79 to 1.53; P=0.58) in the per-protocol analysis.
The absolute difference in 3-year PFS (ABVD minus AVD) was 1.6 percentage points (95% CI, −3.2 to 5.3).
The OS rates were 97.2% in the ABVD group and 97.6% in the AVD group. The hazard ratio in the intention-to-treat analysis was 0.90 (95% CI, 0.47 to 1.74; P=0.76).
Patients in the ABVD group had a significantly higher rate of clinical adverse events than patients in the AVD group—31% and 21%, respectively (P<0.005).
Patients in the ABVD group also had significantly (P<0.05) higher rates of fatigue (3% vs 1%), febrile neutropenia (5% vs 2%), pulmonary/upper respiratory events (3% vs 1%), and dyspnea (2% vs <0.5%). But patients in the AVD group had a significantly higher rate of thrombocytopenia (3% vs 1%).
For patients who had positive interim PET-CT scans, the 3-year PFS was 67.5%, and the OS was 87.8%. Among the 172 patients who went on to receive BEACOPP, 74.4% had negative findings on a third PET-CT scan.
Overall, 62 patients died during the trial—24 from HL. So, for the entire study cohort, the 3-year PFS was 82.6%, and the OS was 95.8%. ![]()

The use of interim PET-CT scans can spare some advanced Hodgkin lymphoma (HL) patients the toxicity associated with bleomycin, according to researchers.
The team found that patients with negative PET-CT scans after 2 cycles of ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) could go on to receive AVD (doxorubicin, vinblastine, and dacarbazine) without experiencing a significant decrease in progression-free survival (PFS) or overall survival (OS).
Peter Johnson, MD, of the University of Southampton in the UK, and his colleagues reported these findings in NEJM.
“The good news is that the majority of people diagnosed with Hodgkin lymphoma can be cured,” Dr Johnson said. “In this trial, more than 95% of patients are alive after 3 years.”
“But we worry about the long-term side effects from the treatments we use. As we’ve done in this trial, personalizing treatment based on how well it works is a major development for patients with Hodgkin lymphoma and sets a new standard of care.”
Patients and treatment
For this study, Dr Johnson and his colleagues enrolled 1214 patients with newly diagnosed, advanced, classic HL. The patients’ median age was 33 (range, 18 to 79), and 54.5% were male. More patients had stage II disease (41.6%) than stage III (30.2%) or IV (28.3%).
A total of 1119 patients underwent a baseline PET-CT scan, received 2 cycles of ABVD, and underwent an interim PET-CT scan.
The patients with negative interim scans were then randomized to continue treatment with ABVD (n=470) or with AVD (n=465) in cycles 3 through 6.
Patients with positive interim scans (n=182) went on to receive BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone, n=172), salvage treatments (n=6), or ABVD (n=4).
Results
The study’s primary outcome was the difference in 3-year PFS between the randomized groups of PET-CT-negative patients.
With a median follow-up of 41 months, the 3-year PFS was 85.7% in the ABVD group and 84.4% in the AVD group. The hazard ratio was 1.13 (95% CI, 0.81 to 1.57; P=0.48) in the intention-to-treat analysis and 1.10 (95% CI, 0.79 to 1.53; P=0.58) in the per-protocol analysis.
The absolute difference in 3-year PFS (ABVD minus AVD) was 1.6 percentage points (95% CI, −3.2 to 5.3).
The OS rates were 97.2% in the ABVD group and 97.6% in the AVD group. The hazard ratio in the intention-to-treat analysis was 0.90 (95% CI, 0.47 to 1.74; P=0.76).
Patients in the ABVD group had a significantly higher rate of clinical adverse events than patients in the AVD group—31% and 21%, respectively (P<0.005).
Patients in the ABVD group also had significantly (P<0.05) higher rates of fatigue (3% vs 1%), febrile neutropenia (5% vs 2%), pulmonary/upper respiratory events (3% vs 1%), and dyspnea (2% vs <0.5%). But patients in the AVD group had a significantly higher rate of thrombocytopenia (3% vs 1%).
For patients who had positive interim PET-CT scans, the 3-year PFS was 67.5%, and the OS was 87.8%. Among the 172 patients who went on to receive BEACOPP, 74.4% had negative findings on a third PET-CT scan.
Overall, 62 patients died during the trial—24 from HL. So, for the entire study cohort, the 3-year PFS was 82.6%, and the OS was 95.8%.