User login
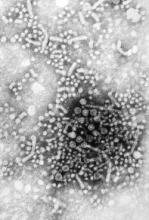
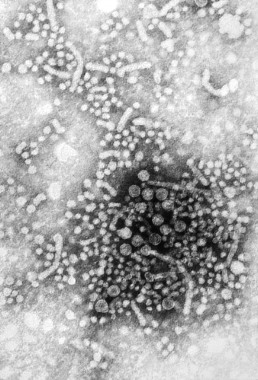
Sofosbuvir combo effective in unresponsive HCV
A 6-month course of the antiviral drug sofosbuvir plus low-dose ribavirin was effective against chronic hepatitis C type 1 in a phase II study predominantly involving patients who had unfavorable predictors of treatment response.
The combination therapy yielded sustained virologic response rates ranging from 48% to 68% in a study population with high prevalences of black race, HCV genotype 1a, advanced liver fibrosis, and obesity, said Dr. Anuoluwapo Osinusi of the National Institute of Allergy and Infectious Diseases, Bethesda, Md., and her associates.
The findings of the small, single-center, open-label study, published online Aug. 27 in JAMA, "are preliminary in nature and require further evaluation in larger studies," the investigators noted.
Several recent studies have demonstrated that interferon-free drug regimens composed solely of directly acting antiviral agents can be effective against chronic HCV type 1, but patients who have certain unfavorable host and viral traits have been underrepresented in these studies.
Noting that it is crucial for such research to involve the "populations most affected by the disease," Dr. Osinusi and her colleagues assessed the safety and efficacy of sofosbuvir in a two-part randomized, controlled trial involving patients who had newly diagnosed, liver biopsy–proven chronic HCV type 1 for which they had never been treated (JAMA 2013 Aug. 27 [doi:10.1001/jama.2013.109309]).
In the study’s proof-of-concept portion, 10 patients who had early to moderate liver fibrosis were treated for 24 weeks with 400 mg/day of sofosbuvir plus weight-based daily doses of ribavirin. In the second, randomized portion of the study, 50 patients with any stage of fibrosis, including compensated cirrhosis, were randomly assigned to receive the same dose of sofosbuvir plus either weight-based ribavirin or low-dose ribavirin for 24 weeks.
Fifty patients (83%) were black, and 29 (48%) were obese. Fourteen (23%) had advanced liver disease and 37 (62%) had high HCV RNA levels at baseline. In comparison, in previous studies the prevalence of black race was 7%-15%, that of obesity was 22%, and that of advanced liver disease was 9%-20%, the researchers said.
All the patients showed a rapid decline in HCV RNA with treatment, and 24 patients in each group achieved total viral suppression within 1 month of beginning treatment. That was accompanied by a rapid improvement in alanine aminotransferase levels, with 77% of patients showing normalized levels within 1 week and 98% showing normalized levels within 2 weeks. The pattern was similar for aspartate aminotransferase levels.
The primary study endpoint was the proportion of patients who had an undetectable HCV viral load 6 months after completing treatment. That endpoint was reached by 68% of patients who took sofosbuvir plus weight-based doses of ribavirin, and by 48% of those who took sofosbuvir plus low doses of ribavirin, the investigators said.
Twenty-nine patients underwent liver biopsy before and after treatment, and 27 of them (93%) showed a large improvement in inflammation. On a 15-point scale of inflammation, the median decline was 5 points.
There were no cases of viral breakthrough during therapy, and HCV deep sequencing in a subgroup of patients detected none of the genetic mutations known to be associated with resistance to sofosbuvir.
"The combination of sofosbuvir and ribavirin was safe and well tolerated, with no death or discontinuation of treatment due to adverse events. The most frequent adverse events were headache, anemia, fatigue, and nausea, the severity of which ranged from mild to moderate," Dr. Osinusi and her associates said.
In an exploratory analysis of viral kinetics-pharmacodynamics, patients who subsequently relapsed showed a significantly slower loss rate of infectious virus at the beginning of treatment. "The mechanism of viral relapse in these participants remains elusive, and future research will be focused on identifying the biological basis for incomplete clearance of HCV in [such] patients," they added.
"This study demonstrates the efficacy of an interferon-free regimen in a traditionally difficult-to-treat population," Dr. Osinusi and her colleagues said. That is encouraging, because HCV treatment is now evolving from interferon-based combination therapy to "an all-oral, interferon-free, directly acting antiviral agent regimen," they added.
The National Cancer Institute, the National Institute of Allergy and Infectious Diseases, and the German Research Foundation funded the study. Pharmasset Pharmaceuticals and Gilead Sciences provided the sofosbuvir and scientific assistance. Dr. Osinusi reported no financial conflicts of interest; her associates reported ties to Abbott, Gilead, Merck, Novartis, Roche Pharma, and Vertex Pharmaceuticals.
A 6-month course of the antiviral drug sofosbuvir plus low-dose ribavirin was effective against chronic hepatitis C type 1 in a phase II study predominantly involving patients who had unfavorable predictors of treatment response.
The combination therapy yielded sustained virologic response rates ranging from 48% to 68% in a study population with high prevalences of black race, HCV genotype 1a, advanced liver fibrosis, and obesity, said Dr. Anuoluwapo Osinusi of the National Institute of Allergy and Infectious Diseases, Bethesda, Md., and her associates.
The findings of the small, single-center, open-label study, published online Aug. 27 in JAMA, "are preliminary in nature and require further evaluation in larger studies," the investigators noted.
Several recent studies have demonstrated that interferon-free drug regimens composed solely of directly acting antiviral agents can be effective against chronic HCV type 1, but patients who have certain unfavorable host and viral traits have been underrepresented in these studies.
Noting that it is crucial for such research to involve the "populations most affected by the disease," Dr. Osinusi and her colleagues assessed the safety and efficacy of sofosbuvir in a two-part randomized, controlled trial involving patients who had newly diagnosed, liver biopsy–proven chronic HCV type 1 for which they had never been treated (JAMA 2013 Aug. 27 [doi:10.1001/jama.2013.109309]).
In the study’s proof-of-concept portion, 10 patients who had early to moderate liver fibrosis were treated for 24 weeks with 400 mg/day of sofosbuvir plus weight-based daily doses of ribavirin. In the second, randomized portion of the study, 50 patients with any stage of fibrosis, including compensated cirrhosis, were randomly assigned to receive the same dose of sofosbuvir plus either weight-based ribavirin or low-dose ribavirin for 24 weeks.
Fifty patients (83%) were black, and 29 (48%) were obese. Fourteen (23%) had advanced liver disease and 37 (62%) had high HCV RNA levels at baseline. In comparison, in previous studies the prevalence of black race was 7%-15%, that of obesity was 22%, and that of advanced liver disease was 9%-20%, the researchers said.
All the patients showed a rapid decline in HCV RNA with treatment, and 24 patients in each group achieved total viral suppression within 1 month of beginning treatment. That was accompanied by a rapid improvement in alanine aminotransferase levels, with 77% of patients showing normalized levels within 1 week and 98% showing normalized levels within 2 weeks. The pattern was similar for aspartate aminotransferase levels.
The primary study endpoint was the proportion of patients who had an undetectable HCV viral load 6 months after completing treatment. That endpoint was reached by 68% of patients who took sofosbuvir plus weight-based doses of ribavirin, and by 48% of those who took sofosbuvir plus low doses of ribavirin, the investigators said.
Twenty-nine patients underwent liver biopsy before and after treatment, and 27 of them (93%) showed a large improvement in inflammation. On a 15-point scale of inflammation, the median decline was 5 points.
There were no cases of viral breakthrough during therapy, and HCV deep sequencing in a subgroup of patients detected none of the genetic mutations known to be associated with resistance to sofosbuvir.
"The combination of sofosbuvir and ribavirin was safe and well tolerated, with no death or discontinuation of treatment due to adverse events. The most frequent adverse events were headache, anemia, fatigue, and nausea, the severity of which ranged from mild to moderate," Dr. Osinusi and her associates said.
In an exploratory analysis of viral kinetics-pharmacodynamics, patients who subsequently relapsed showed a significantly slower loss rate of infectious virus at the beginning of treatment. "The mechanism of viral relapse in these participants remains elusive, and future research will be focused on identifying the biological basis for incomplete clearance of HCV in [such] patients," they added.
"This study demonstrates the efficacy of an interferon-free regimen in a traditionally difficult-to-treat population," Dr. Osinusi and her colleagues said. That is encouraging, because HCV treatment is now evolving from interferon-based combination therapy to "an all-oral, interferon-free, directly acting antiviral agent regimen," they added.
The National Cancer Institute, the National Institute of Allergy and Infectious Diseases, and the German Research Foundation funded the study. Pharmasset Pharmaceuticals and Gilead Sciences provided the sofosbuvir and scientific assistance. Dr. Osinusi reported no financial conflicts of interest; her associates reported ties to Abbott, Gilead, Merck, Novartis, Roche Pharma, and Vertex Pharmaceuticals.
A 6-month course of the antiviral drug sofosbuvir plus low-dose ribavirin was effective against chronic hepatitis C type 1 in a phase II study predominantly involving patients who had unfavorable predictors of treatment response.
The combination therapy yielded sustained virologic response rates ranging from 48% to 68% in a study population with high prevalences of black race, HCV genotype 1a, advanced liver fibrosis, and obesity, said Dr. Anuoluwapo Osinusi of the National Institute of Allergy and Infectious Diseases, Bethesda, Md., and her associates.
The findings of the small, single-center, open-label study, published online Aug. 27 in JAMA, "are preliminary in nature and require further evaluation in larger studies," the investigators noted.
Several recent studies have demonstrated that interferon-free drug regimens composed solely of directly acting antiviral agents can be effective against chronic HCV type 1, but patients who have certain unfavorable host and viral traits have been underrepresented in these studies.
Noting that it is crucial for such research to involve the "populations most affected by the disease," Dr. Osinusi and her colleagues assessed the safety and efficacy of sofosbuvir in a two-part randomized, controlled trial involving patients who had newly diagnosed, liver biopsy–proven chronic HCV type 1 for which they had never been treated (JAMA 2013 Aug. 27 [doi:10.1001/jama.2013.109309]).
In the study’s proof-of-concept portion, 10 patients who had early to moderate liver fibrosis were treated for 24 weeks with 400 mg/day of sofosbuvir plus weight-based daily doses of ribavirin. In the second, randomized portion of the study, 50 patients with any stage of fibrosis, including compensated cirrhosis, were randomly assigned to receive the same dose of sofosbuvir plus either weight-based ribavirin or low-dose ribavirin for 24 weeks.
Fifty patients (83%) were black, and 29 (48%) were obese. Fourteen (23%) had advanced liver disease and 37 (62%) had high HCV RNA levels at baseline. In comparison, in previous studies the prevalence of black race was 7%-15%, that of obesity was 22%, and that of advanced liver disease was 9%-20%, the researchers said.
All the patients showed a rapid decline in HCV RNA with treatment, and 24 patients in each group achieved total viral suppression within 1 month of beginning treatment. That was accompanied by a rapid improvement in alanine aminotransferase levels, with 77% of patients showing normalized levels within 1 week and 98% showing normalized levels within 2 weeks. The pattern was similar for aspartate aminotransferase levels.
The primary study endpoint was the proportion of patients who had an undetectable HCV viral load 6 months after completing treatment. That endpoint was reached by 68% of patients who took sofosbuvir plus weight-based doses of ribavirin, and by 48% of those who took sofosbuvir plus low doses of ribavirin, the investigators said.
Twenty-nine patients underwent liver biopsy before and after treatment, and 27 of them (93%) showed a large improvement in inflammation. On a 15-point scale of inflammation, the median decline was 5 points.
There were no cases of viral breakthrough during therapy, and HCV deep sequencing in a subgroup of patients detected none of the genetic mutations known to be associated with resistance to sofosbuvir.
"The combination of sofosbuvir and ribavirin was safe and well tolerated, with no death or discontinuation of treatment due to adverse events. The most frequent adverse events were headache, anemia, fatigue, and nausea, the severity of which ranged from mild to moderate," Dr. Osinusi and her associates said.
In an exploratory analysis of viral kinetics-pharmacodynamics, patients who subsequently relapsed showed a significantly slower loss rate of infectious virus at the beginning of treatment. "The mechanism of viral relapse in these participants remains elusive, and future research will be focused on identifying the biological basis for incomplete clearance of HCV in [such] patients," they added.
"This study demonstrates the efficacy of an interferon-free regimen in a traditionally difficult-to-treat population," Dr. Osinusi and her colleagues said. That is encouraging, because HCV treatment is now evolving from interferon-based combination therapy to "an all-oral, interferon-free, directly acting antiviral agent regimen," they added.
The National Cancer Institute, the National Institute of Allergy and Infectious Diseases, and the German Research Foundation funded the study. Pharmasset Pharmaceuticals and Gilead Sciences provided the sofosbuvir and scientific assistance. Dr. Osinusi reported no financial conflicts of interest; her associates reported ties to Abbott, Gilead, Merck, Novartis, Roche Pharma, and Vertex Pharmaceuticals.
FROM JAMA
Major Finding: Sixty-eight percent of patients who took sofosbuvir plus weight-based doses of ribavirin and 48% of those who took sofosbuvir plus low doses of ribavirin had an undetectable HCV viral load 6 months after completing treatment.
Data Source: A single-center, randomized, open-label, phase II clinical trial involving 60 treatment-naive patients who had chronic hepatitis C type 1 and were treated with sofosbuvir for 6 months.
Disclosures: The National Cancer Institute, the National Institute of Allergy and Infectious Diseases, and the German Research Foundation funded the study. Pharmasset Pharmaceuticals and Gilead Sciences provided the sofosbuvir and scientific assistance. Dr. Osinusi reported no financial conflicts of interest; her associates reported ties to Abbott, Gilead, Merck, Novartis, Roche Pharma, and Vertex Pharmaceuticals.
Interferon-free regimens effective for hepatitis C virus genotype 1
An interferon-free regimen of faldaprevir, deleobuvir, and ribavirin proved to be effective in a phase II clinical trial involving 362 patients with previously untreated chronic hepatitis C virus genotype 1 infection, according to a report published in the New England Journal of Medicine.
The new combination therapy achieved sustained virologic response rates of 52%-69% 12 weeks after treatment was completed, reported Dr. Stefan Zeuzem of Johann Wolfgang Goethe University Medical Center, Frankfurt am Main (Germany), and his associates (N. Engl. J. Med. 2013;369:630-9).
Those sustained virologic response rates compare well with rates of 68%-75% reported in phase III trials of the regimen of pegylated interferon, ribavirin, and telaprevir or boceprevir that is the current standard of care, the investigators noted.
The new, interferon-free regimen may offer an advantage over the standard of care by avoiding interferon’s detrimental effects on white-cell and platelet counts, they said.
The open-label trial, sponsored by Boehringer Ingelheim, enrolled patients at 48 medical centers in Europe, Australia, and New Zealand. They were randomly assigned to one of five treatment groups with various dosages of the protease inhibitor faldaprevir and the nonnucleoside polymerase inhibitor deleobuvir, with or without the addition of daily ribavirin. Treatment intervals of 16, 28, and 40 weeks were tested.
The primary efficacy endpoint was a sustained virologic response (an undetectable plasma level of hepatitis C virus [HCV] RNA) 12 weeks after the completion of treatment. That endpoint was achieved in 52%-69% of patients with several combinations of faldaprevir, deleobuvir, and ribavirin, and with all treatment durations, Dr. Zeuzem and his colleagues said.
The treatment response rate was not affected by the duration of therapy in most groups. However, the relapse rate was markedly higher in the subgroup of patients with HCV genotype 1a who received only 16 weeks of therapy (41%), compared with those who received 28 weeks (0%) or 40 weeks (6%).
In contrast, relapse rates among patients with HCV genotype 1b were consistently low across all treatment durations, suggesting that 16 weeks of treatment may be sufficient for this subgroup of patients, the researchers said.
The groups who did not receive ribavirin showed high rates of both virologic breakthrough during treatment and relapse after treatment was completed. This has been reported in other studies of interferon-free regimens without ribavirin, and indicates that ribavirin is a necessary component of such regimens, they said.
There were small, nonsignificant differences in response rates according to the dosage of deleobuvir. However, the rate of premature discontinuation among patients who did not have a sustained virologic response 12 weeks after the completion of therapy was significantly higher among those who took deleobuvir three times per day (15%) than in those who took it twice per day (4%).
Adverse events were extremely common, affecting 94% of patients. Nine percent reported severe adverse events, including rash (six patients) and anemia (two patients).
Gastrointestinal effects (nausea, diarrhea, and vomiting) and dermatologic effects (pruritus, maculopapular rash, photosensitivity reaction, and dry skin) were the most frequently reported, and they were particularly common during the first week of treatment. That may be because patients received high loading doses at the start of therapy.
Jaundice also was reported and was attributed to increased levels of bilirubin. Faldaprevir is known to inhibit bilirubin metabolism and likely contributed to the 67 cases in the study.
"Substantial reductions in red-cell, white-cell, and platelet counts are the most prohibitive side effects of interferon-based treatments for HCV infection," but were uncommon in the study, Dr. Zeuzem and his associates said.
However, the study was limited because it didn’t include an interferon-receiving control group, and the finding regarding cell counts should be interpreted with caution, they added.
Boehringer Ingelheim funded the trial, monitored the study, collected data, performed the statistical analyses, and helped write the report.
An interferon-free regimen of faldaprevir, deleobuvir, and ribavirin proved to be effective in a phase II clinical trial involving 362 patients with previously untreated chronic hepatitis C virus genotype 1 infection, according to a report published in the New England Journal of Medicine.
The new combination therapy achieved sustained virologic response rates of 52%-69% 12 weeks after treatment was completed, reported Dr. Stefan Zeuzem of Johann Wolfgang Goethe University Medical Center, Frankfurt am Main (Germany), and his associates (N. Engl. J. Med. 2013;369:630-9).
Those sustained virologic response rates compare well with rates of 68%-75% reported in phase III trials of the regimen of pegylated interferon, ribavirin, and telaprevir or boceprevir that is the current standard of care, the investigators noted.
The new, interferon-free regimen may offer an advantage over the standard of care by avoiding interferon’s detrimental effects on white-cell and platelet counts, they said.
The open-label trial, sponsored by Boehringer Ingelheim, enrolled patients at 48 medical centers in Europe, Australia, and New Zealand. They were randomly assigned to one of five treatment groups with various dosages of the protease inhibitor faldaprevir and the nonnucleoside polymerase inhibitor deleobuvir, with or without the addition of daily ribavirin. Treatment intervals of 16, 28, and 40 weeks were tested.
The primary efficacy endpoint was a sustained virologic response (an undetectable plasma level of hepatitis C virus [HCV] RNA) 12 weeks after the completion of treatment. That endpoint was achieved in 52%-69% of patients with several combinations of faldaprevir, deleobuvir, and ribavirin, and with all treatment durations, Dr. Zeuzem and his colleagues said.
The treatment response rate was not affected by the duration of therapy in most groups. However, the relapse rate was markedly higher in the subgroup of patients with HCV genotype 1a who received only 16 weeks of therapy (41%), compared with those who received 28 weeks (0%) or 40 weeks (6%).
In contrast, relapse rates among patients with HCV genotype 1b were consistently low across all treatment durations, suggesting that 16 weeks of treatment may be sufficient for this subgroup of patients, the researchers said.
The groups who did not receive ribavirin showed high rates of both virologic breakthrough during treatment and relapse after treatment was completed. This has been reported in other studies of interferon-free regimens without ribavirin, and indicates that ribavirin is a necessary component of such regimens, they said.
There were small, nonsignificant differences in response rates according to the dosage of deleobuvir. However, the rate of premature discontinuation among patients who did not have a sustained virologic response 12 weeks after the completion of therapy was significantly higher among those who took deleobuvir three times per day (15%) than in those who took it twice per day (4%).
Adverse events were extremely common, affecting 94% of patients. Nine percent reported severe adverse events, including rash (six patients) and anemia (two patients).
Gastrointestinal effects (nausea, diarrhea, and vomiting) and dermatologic effects (pruritus, maculopapular rash, photosensitivity reaction, and dry skin) were the most frequently reported, and they were particularly common during the first week of treatment. That may be because patients received high loading doses at the start of therapy.
Jaundice also was reported and was attributed to increased levels of bilirubin. Faldaprevir is known to inhibit bilirubin metabolism and likely contributed to the 67 cases in the study.
"Substantial reductions in red-cell, white-cell, and platelet counts are the most prohibitive side effects of interferon-based treatments for HCV infection," but were uncommon in the study, Dr. Zeuzem and his associates said.
However, the study was limited because it didn’t include an interferon-receiving control group, and the finding regarding cell counts should be interpreted with caution, they added.
Boehringer Ingelheim funded the trial, monitored the study, collected data, performed the statistical analyses, and helped write the report.
An interferon-free regimen of faldaprevir, deleobuvir, and ribavirin proved to be effective in a phase II clinical trial involving 362 patients with previously untreated chronic hepatitis C virus genotype 1 infection, according to a report published in the New England Journal of Medicine.
The new combination therapy achieved sustained virologic response rates of 52%-69% 12 weeks after treatment was completed, reported Dr. Stefan Zeuzem of Johann Wolfgang Goethe University Medical Center, Frankfurt am Main (Germany), and his associates (N. Engl. J. Med. 2013;369:630-9).
Those sustained virologic response rates compare well with rates of 68%-75% reported in phase III trials of the regimen of pegylated interferon, ribavirin, and telaprevir or boceprevir that is the current standard of care, the investigators noted.
The new, interferon-free regimen may offer an advantage over the standard of care by avoiding interferon’s detrimental effects on white-cell and platelet counts, they said.
The open-label trial, sponsored by Boehringer Ingelheim, enrolled patients at 48 medical centers in Europe, Australia, and New Zealand. They were randomly assigned to one of five treatment groups with various dosages of the protease inhibitor faldaprevir and the nonnucleoside polymerase inhibitor deleobuvir, with or without the addition of daily ribavirin. Treatment intervals of 16, 28, and 40 weeks were tested.
The primary efficacy endpoint was a sustained virologic response (an undetectable plasma level of hepatitis C virus [HCV] RNA) 12 weeks after the completion of treatment. That endpoint was achieved in 52%-69% of patients with several combinations of faldaprevir, deleobuvir, and ribavirin, and with all treatment durations, Dr. Zeuzem and his colleagues said.
The treatment response rate was not affected by the duration of therapy in most groups. However, the relapse rate was markedly higher in the subgroup of patients with HCV genotype 1a who received only 16 weeks of therapy (41%), compared with those who received 28 weeks (0%) or 40 weeks (6%).
In contrast, relapse rates among patients with HCV genotype 1b were consistently low across all treatment durations, suggesting that 16 weeks of treatment may be sufficient for this subgroup of patients, the researchers said.
The groups who did not receive ribavirin showed high rates of both virologic breakthrough during treatment and relapse after treatment was completed. This has been reported in other studies of interferon-free regimens without ribavirin, and indicates that ribavirin is a necessary component of such regimens, they said.
There were small, nonsignificant differences in response rates according to the dosage of deleobuvir. However, the rate of premature discontinuation among patients who did not have a sustained virologic response 12 weeks after the completion of therapy was significantly higher among those who took deleobuvir three times per day (15%) than in those who took it twice per day (4%).
Adverse events were extremely common, affecting 94% of patients. Nine percent reported severe adverse events, including rash (six patients) and anemia (two patients).
Gastrointestinal effects (nausea, diarrhea, and vomiting) and dermatologic effects (pruritus, maculopapular rash, photosensitivity reaction, and dry skin) were the most frequently reported, and they were particularly common during the first week of treatment. That may be because patients received high loading doses at the start of therapy.
Jaundice also was reported and was attributed to increased levels of bilirubin. Faldaprevir is known to inhibit bilirubin metabolism and likely contributed to the 67 cases in the study.
"Substantial reductions in red-cell, white-cell, and platelet counts are the most prohibitive side effects of interferon-based treatments for HCV infection," but were uncommon in the study, Dr. Zeuzem and his associates said.
However, the study was limited because it didn’t include an interferon-receiving control group, and the finding regarding cell counts should be interpreted with caution, they added.
Boehringer Ingelheim funded the trial, monitored the study, collected data, performed the statistical analyses, and helped write the report.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The primary efficacy endpoint – a sustained virologic response (an undetectable plasma level of HCV RNA) 12 weeks after the completion of treatment – was achieved in 52%-69% of patients with several combinations of faldaprevir, deleobuvir, and ribavirin, and with all treatment durations.
Data source: A phase II randomized, open-label clinical trial comparing various regimens of faldaprevir plus deleobuvir with or without ribavirin in 362 treatment-naive patients who had chronic HCV-1.
Disclosures: Boehringer Ingelheim funded the trial, monitored the study, collected data, performed the statistical analyses, and helped write the report.
New estimates show lack of progress in liver mortality
Deaths from liver-related causes remained static between 1979 and 2008, according to updated estimates from the Rochester Epidemiology Project and the National Death Index.
The finding represents a dramatic correction to previous estimates from the National Center for Health Statistics (NCHS) at the Centers for Disease Control and Prevention, which used a very narrow definition of liver-related mortality, Dr. Sumeet K. Asrani and colleagues reported. The results were published in the August issue of Gastroenterology.
Courtesy: American Gastroenterological Association
According to Dr. Asrani of the Mayo Clinic, Rochester, Minn., "current estimates [from the NCHS] are solely based on one diagnostic category, namely chronic liver disease and cirrhosis, which fails to capture deaths attributed to other uniquely liver-related descriptors, such as hepatic encephalopathy or hepatorenal syndrome."
Deaths due to viral hepatitis or liver cancer are also not included.
"For example, the current estimates of liver-related deaths might not include the demise of a person with hepatitis C cirrhosis who died of hepatorenal syndrome."
In their updated assessment, the researchers looked at data from the Rochester Epidemiology Project, in which death records of Olmsted County residents are tracked from multiple sources, including county and state vital records, as well as individual medical charts.
The researchers found that when the CDC definition was used, which includes causes attributable to a single ICD-9 code of 571 and ICD-10 codes of K70, K73, and K74, there were 71 liver-related deaths among Olmsted County residents between 1999 and 2008.
In contrast, when the updated definition was applied, which included additional diagnoses specific to liver disease yet excluded in the CDC definition, as well as viral hepatitis and malignant neoplasm of the liver and intrahepatic bile ducts, there were 261 liver-related deaths in the county between 1999 and 2008.
That included 85 deaths (32.6%) from viral hepatitis and 70 deaths (26.8%) from hepatobiliary malignancies.
The researchers then looked at national mortality rates.
According to the restrictive CDC estimates, in 2008, there were 29,951 deaths due to liver disease in the United States, for a death rate of 11.7 per 100,000 persons (16.2 for men and 7.6 for women).
Using the updated definition, however, Dr. Asrani tallied 66,007 deaths, including 10,256 from the expanded liver disease diagnosis, 7,625 from viral hepatitis, and 18,175 from hepatobiliary malignancies, for a death rate of 25.7 per 100,000 persons (35.7 for men and 16.8 for women).
Finally, the investigators compared longitudinal trends in death rates.
They found that when the CDC definition was used, deaths due to liver disease (per 100,000 persons) decreased from 16.5 in the era encompassing the years 1979 to 1988 to 11.7 in the years between 1999 and 2008, a reduction of 38%.
"However, when the updated definition was applied, the downward trend disappeared," they wrote.
Indeed, liver-related mortality per 100,000 persons was basically unchanged over 30 years: 23.9 for the era between 1979 and1988 and 24.4 for the period from 1999 to 2008.
"This discrepancy was accounted for by deaths due to viral hepatitis, which increased from 0.5 to 2.5 per 100,000 and hepatobiliary cancer, which increased from 3.1 to 6.4 per 100,000," wrote the authors.
"These data support that deaths due to viral hepatitis and hepatobiliary cancers should be included in the enumeration of liver-related deaths to accurately represent the burden of chronic liver disease."
"Underappreciation of the prevalence and natural history of liver disease can lead to suboptimal care," they concluded.
The authors disclosed no conflicts of interest related to this study, which was partially supported by grants from the National Institutes of Health.
Deaths from liver-related causes remained static between 1979 and 2008, according to updated estimates from the Rochester Epidemiology Project and the National Death Index.
The finding represents a dramatic correction to previous estimates from the National Center for Health Statistics (NCHS) at the Centers for Disease Control and Prevention, which used a very narrow definition of liver-related mortality, Dr. Sumeet K. Asrani and colleagues reported. The results were published in the August issue of Gastroenterology.
Courtesy: American Gastroenterological Association
According to Dr. Asrani of the Mayo Clinic, Rochester, Minn., "current estimates [from the NCHS] are solely based on one diagnostic category, namely chronic liver disease and cirrhosis, which fails to capture deaths attributed to other uniquely liver-related descriptors, such as hepatic encephalopathy or hepatorenal syndrome."
Deaths due to viral hepatitis or liver cancer are also not included.
"For example, the current estimates of liver-related deaths might not include the demise of a person with hepatitis C cirrhosis who died of hepatorenal syndrome."
In their updated assessment, the researchers looked at data from the Rochester Epidemiology Project, in which death records of Olmsted County residents are tracked from multiple sources, including county and state vital records, as well as individual medical charts.
The researchers found that when the CDC definition was used, which includes causes attributable to a single ICD-9 code of 571 and ICD-10 codes of K70, K73, and K74, there were 71 liver-related deaths among Olmsted County residents between 1999 and 2008.
In contrast, when the updated definition was applied, which included additional diagnoses specific to liver disease yet excluded in the CDC definition, as well as viral hepatitis and malignant neoplasm of the liver and intrahepatic bile ducts, there were 261 liver-related deaths in the county between 1999 and 2008.
That included 85 deaths (32.6%) from viral hepatitis and 70 deaths (26.8%) from hepatobiliary malignancies.
The researchers then looked at national mortality rates.
According to the restrictive CDC estimates, in 2008, there were 29,951 deaths due to liver disease in the United States, for a death rate of 11.7 per 100,000 persons (16.2 for men and 7.6 for women).
Using the updated definition, however, Dr. Asrani tallied 66,007 deaths, including 10,256 from the expanded liver disease diagnosis, 7,625 from viral hepatitis, and 18,175 from hepatobiliary malignancies, for a death rate of 25.7 per 100,000 persons (35.7 for men and 16.8 for women).
Finally, the investigators compared longitudinal trends in death rates.
They found that when the CDC definition was used, deaths due to liver disease (per 100,000 persons) decreased from 16.5 in the era encompassing the years 1979 to 1988 to 11.7 in the years between 1999 and 2008, a reduction of 38%.
"However, when the updated definition was applied, the downward trend disappeared," they wrote.
Indeed, liver-related mortality per 100,000 persons was basically unchanged over 30 years: 23.9 for the era between 1979 and1988 and 24.4 for the period from 1999 to 2008.
"This discrepancy was accounted for by deaths due to viral hepatitis, which increased from 0.5 to 2.5 per 100,000 and hepatobiliary cancer, which increased from 3.1 to 6.4 per 100,000," wrote the authors.
"These data support that deaths due to viral hepatitis and hepatobiliary cancers should be included in the enumeration of liver-related deaths to accurately represent the burden of chronic liver disease."
"Underappreciation of the prevalence and natural history of liver disease can lead to suboptimal care," they concluded.
The authors disclosed no conflicts of interest related to this study, which was partially supported by grants from the National Institutes of Health.
Deaths from liver-related causes remained static between 1979 and 2008, according to updated estimates from the Rochester Epidemiology Project and the National Death Index.
The finding represents a dramatic correction to previous estimates from the National Center for Health Statistics (NCHS) at the Centers for Disease Control and Prevention, which used a very narrow definition of liver-related mortality, Dr. Sumeet K. Asrani and colleagues reported. The results were published in the August issue of Gastroenterology.
Courtesy: American Gastroenterological Association
According to Dr. Asrani of the Mayo Clinic, Rochester, Minn., "current estimates [from the NCHS] are solely based on one diagnostic category, namely chronic liver disease and cirrhosis, which fails to capture deaths attributed to other uniquely liver-related descriptors, such as hepatic encephalopathy or hepatorenal syndrome."
Deaths due to viral hepatitis or liver cancer are also not included.
"For example, the current estimates of liver-related deaths might not include the demise of a person with hepatitis C cirrhosis who died of hepatorenal syndrome."
In their updated assessment, the researchers looked at data from the Rochester Epidemiology Project, in which death records of Olmsted County residents are tracked from multiple sources, including county and state vital records, as well as individual medical charts.
The researchers found that when the CDC definition was used, which includes causes attributable to a single ICD-9 code of 571 and ICD-10 codes of K70, K73, and K74, there were 71 liver-related deaths among Olmsted County residents between 1999 and 2008.
In contrast, when the updated definition was applied, which included additional diagnoses specific to liver disease yet excluded in the CDC definition, as well as viral hepatitis and malignant neoplasm of the liver and intrahepatic bile ducts, there were 261 liver-related deaths in the county between 1999 and 2008.
That included 85 deaths (32.6%) from viral hepatitis and 70 deaths (26.8%) from hepatobiliary malignancies.
The researchers then looked at national mortality rates.
According to the restrictive CDC estimates, in 2008, there were 29,951 deaths due to liver disease in the United States, for a death rate of 11.7 per 100,000 persons (16.2 for men and 7.6 for women).
Using the updated definition, however, Dr. Asrani tallied 66,007 deaths, including 10,256 from the expanded liver disease diagnosis, 7,625 from viral hepatitis, and 18,175 from hepatobiliary malignancies, for a death rate of 25.7 per 100,000 persons (35.7 for men and 16.8 for women).
Finally, the investigators compared longitudinal trends in death rates.
They found that when the CDC definition was used, deaths due to liver disease (per 100,000 persons) decreased from 16.5 in the era encompassing the years 1979 to 1988 to 11.7 in the years between 1999 and 2008, a reduction of 38%.
"However, when the updated definition was applied, the downward trend disappeared," they wrote.
Indeed, liver-related mortality per 100,000 persons was basically unchanged over 30 years: 23.9 for the era between 1979 and1988 and 24.4 for the period from 1999 to 2008.
"This discrepancy was accounted for by deaths due to viral hepatitis, which increased from 0.5 to 2.5 per 100,000 and hepatobiliary cancer, which increased from 3.1 to 6.4 per 100,000," wrote the authors.
"These data support that deaths due to viral hepatitis and hepatobiliary cancers should be included in the enumeration of liver-related deaths to accurately represent the burden of chronic liver disease."
"Underappreciation of the prevalence and natural history of liver disease can lead to suboptimal care," they concluded.
The authors disclosed no conflicts of interest related to this study, which was partially supported by grants from the National Institutes of Health.
FROM GASTROENTEROLOGY
Major finding: An updated definition of liver-related mortality shows that death rates are unchanged over 30 years: 23.9 per 100,000 persons for the era between 1979 and 1988 and 24.4 for the period from 1999 to 2008.
Data source: Records from the Rochester Epidemiology Project and the National Death Registry.
Disclosures: The authors disclosed no conflicts of interest related to this study, which was partially supported by grants from the National Institutes of Health.
Triple therapy underutilized in HCV
Fewer than 20% of patients with hepatitis C virus genotype 1 receive triple therapy with boceprevir or telaprevir, according to Dr. Emerson Y. Chen and colleagues. The results are in the August issue of Clinical Gastroenterology and Hepatology.
The "disappointingly low use of the new therapies, even after a decade without novel medications," suggests that real-world use of these drugs is hampered by factors including safety and the low predicted response rates in prior nonresponders, they wrote.
Dr. Chen of the University of Texas Southwestern Medical School, Dallas, looked at 487 patients with HCV genotype 1 presenting to one of two academic outpatient hepatology practices in Dallas and Miami.
The majority of patients were between 50 and 60 years of age, male, and white. More than two-thirds had private insurance, and half of the patients were treatment naive.
Overall, the authors found that 91 patients (18.7%) were started on triple therapy (boceprevir or telaprevir plus pegylated interferon and ribavirin) while the remaining 396 patients remained untreated.
Dr. Chen then assessed the reasons for treatment deferral. The most common, he found, was contraindication (50.5%), including complications of liver disease and medical comorbidities. The next biggest reason cited for treatment deferral was "patient choice" (22.5%), which included concerns about side effects, limited success rates, financial issues, or inability to commit time for treatment. Finally, the presence of less advanced liver disease (17.4%) and the anticipation (among providers and patients alike) of better future therapies (9.6%) were also factors in triple therapy refusal.
"Although several expert panels for treatment recommendations are available, there exists an uncertainty among providers regarding selecting patients without major contraindications for triple therapy now over newer direct-acting antiviral therapy later," they added.
Next, the researchers compared the triple therapy patients with therapy deferrals.
In univariate analysis, they found that about three-fourths of patients who chose to initiate triple therapy had fibrosis stage 3 or were cirrhotic, although less than 10% had a history of overt decompensation.
"In contrast, among those who deferred treatment, more than 20% had decompensated liver disease, and 46% had early [an] fibrosis stage," they wrote.
Looking at patients’ mean Model for End-Stage Liver Disease scores, cirrhotic patients who deferred treatment registered a 9.3, compared with 7.3 for patients who elected to undergo triple therapy treatment (P less than .001). Additionally, 90% of cirrhotic patients on treatment were Child-Pugh class A compared with 63% in the nontreatment group (P = .003).
Commenting on the findings, Dr. Chen wrote that "although patients with mild fibrosis or persistently normal liver function tests have often not been recommended for repeat biopsy or treatment, they would likely benefit from treatment before they become older or develop contraindications.
"Nevertheless, many hepatologists appear to be recommending deferral of treatment for patients with mild to moderate fibrosis, waiting for the second-generation direct-acting antivirals with even higher sustained virologic response rates" and fewer side effects.
"Newly diagnosed treatment-naive patients along with those with less advanced liver disease will benefit from society-wide consensus regarding therapy initiation now or at a later time."
The authors disclosed funding from the Doris Duke Charitable Foundation and the University of Miami. Two authors disclosed ties to Merck, maker of boceprevir and ribavirin, and Vertex, which makes telaprevir.
Fewer than 20% of patients with hepatitis C virus genotype 1 receive triple therapy with boceprevir or telaprevir, according to Dr. Emerson Y. Chen and colleagues. The results are in the August issue of Clinical Gastroenterology and Hepatology.
The "disappointingly low use of the new therapies, even after a decade without novel medications," suggests that real-world use of these drugs is hampered by factors including safety and the low predicted response rates in prior nonresponders, they wrote.
Dr. Chen of the University of Texas Southwestern Medical School, Dallas, looked at 487 patients with HCV genotype 1 presenting to one of two academic outpatient hepatology practices in Dallas and Miami.
The majority of patients were between 50 and 60 years of age, male, and white. More than two-thirds had private insurance, and half of the patients were treatment naive.
Overall, the authors found that 91 patients (18.7%) were started on triple therapy (boceprevir or telaprevir plus pegylated interferon and ribavirin) while the remaining 396 patients remained untreated.
Dr. Chen then assessed the reasons for treatment deferral. The most common, he found, was contraindication (50.5%), including complications of liver disease and medical comorbidities. The next biggest reason cited for treatment deferral was "patient choice" (22.5%), which included concerns about side effects, limited success rates, financial issues, or inability to commit time for treatment. Finally, the presence of less advanced liver disease (17.4%) and the anticipation (among providers and patients alike) of better future therapies (9.6%) were also factors in triple therapy refusal.
"Although several expert panels for treatment recommendations are available, there exists an uncertainty among providers regarding selecting patients without major contraindications for triple therapy now over newer direct-acting antiviral therapy later," they added.
Next, the researchers compared the triple therapy patients with therapy deferrals.
In univariate analysis, they found that about three-fourths of patients who chose to initiate triple therapy had fibrosis stage 3 or were cirrhotic, although less than 10% had a history of overt decompensation.
"In contrast, among those who deferred treatment, more than 20% had decompensated liver disease, and 46% had early [an] fibrosis stage," they wrote.
Looking at patients’ mean Model for End-Stage Liver Disease scores, cirrhotic patients who deferred treatment registered a 9.3, compared with 7.3 for patients who elected to undergo triple therapy treatment (P less than .001). Additionally, 90% of cirrhotic patients on treatment were Child-Pugh class A compared with 63% in the nontreatment group (P = .003).
Commenting on the findings, Dr. Chen wrote that "although patients with mild fibrosis or persistently normal liver function tests have often not been recommended for repeat biopsy or treatment, they would likely benefit from treatment before they become older or develop contraindications.
"Nevertheless, many hepatologists appear to be recommending deferral of treatment for patients with mild to moderate fibrosis, waiting for the second-generation direct-acting antivirals with even higher sustained virologic response rates" and fewer side effects.
"Newly diagnosed treatment-naive patients along with those with less advanced liver disease will benefit from society-wide consensus regarding therapy initiation now or at a later time."
The authors disclosed funding from the Doris Duke Charitable Foundation and the University of Miami. Two authors disclosed ties to Merck, maker of boceprevir and ribavirin, and Vertex, which makes telaprevir.
Fewer than 20% of patients with hepatitis C virus genotype 1 receive triple therapy with boceprevir or telaprevir, according to Dr. Emerson Y. Chen and colleagues. The results are in the August issue of Clinical Gastroenterology and Hepatology.
The "disappointingly low use of the new therapies, even after a decade without novel medications," suggests that real-world use of these drugs is hampered by factors including safety and the low predicted response rates in prior nonresponders, they wrote.
Dr. Chen of the University of Texas Southwestern Medical School, Dallas, looked at 487 patients with HCV genotype 1 presenting to one of two academic outpatient hepatology practices in Dallas and Miami.
The majority of patients were between 50 and 60 years of age, male, and white. More than two-thirds had private insurance, and half of the patients were treatment naive.
Overall, the authors found that 91 patients (18.7%) were started on triple therapy (boceprevir or telaprevir plus pegylated interferon and ribavirin) while the remaining 396 patients remained untreated.
Dr. Chen then assessed the reasons for treatment deferral. The most common, he found, was contraindication (50.5%), including complications of liver disease and medical comorbidities. The next biggest reason cited for treatment deferral was "patient choice" (22.5%), which included concerns about side effects, limited success rates, financial issues, or inability to commit time for treatment. Finally, the presence of less advanced liver disease (17.4%) and the anticipation (among providers and patients alike) of better future therapies (9.6%) were also factors in triple therapy refusal.
"Although several expert panels for treatment recommendations are available, there exists an uncertainty among providers regarding selecting patients without major contraindications for triple therapy now over newer direct-acting antiviral therapy later," they added.
Next, the researchers compared the triple therapy patients with therapy deferrals.
In univariate analysis, they found that about three-fourths of patients who chose to initiate triple therapy had fibrosis stage 3 or were cirrhotic, although less than 10% had a history of overt decompensation.
"In contrast, among those who deferred treatment, more than 20% had decompensated liver disease, and 46% had early [an] fibrosis stage," they wrote.
Looking at patients’ mean Model for End-Stage Liver Disease scores, cirrhotic patients who deferred treatment registered a 9.3, compared with 7.3 for patients who elected to undergo triple therapy treatment (P less than .001). Additionally, 90% of cirrhotic patients on treatment were Child-Pugh class A compared with 63% in the nontreatment group (P = .003).
Commenting on the findings, Dr. Chen wrote that "although patients with mild fibrosis or persistently normal liver function tests have often not been recommended for repeat biopsy or treatment, they would likely benefit from treatment before they become older or develop contraindications.
"Nevertheless, many hepatologists appear to be recommending deferral of treatment for patients with mild to moderate fibrosis, waiting for the second-generation direct-acting antivirals with even higher sustained virologic response rates" and fewer side effects.
"Newly diagnosed treatment-naive patients along with those with less advanced liver disease will benefit from society-wide consensus regarding therapy initiation now or at a later time."
The authors disclosed funding from the Doris Duke Charitable Foundation and the University of Miami. Two authors disclosed ties to Merck, maker of boceprevir and ribavirin, and Vertex, which makes telaprevir.
Major finding: From a cohort of nearly 500 hepatitis C patients, 91 (18.7%) were started on triple therapy.
Data source: A retrospective, 1-year, cross-sectional study of adults with HCV infection presenting to two outpatient hepatology practices in Dallas and Miami.
Disclosures: The authors disclosed funding from the Doris Duke Charitable Foundation and the University of Miami. Two authors disclosed ties to Merck, maker of boceprevir and ribavirin, and Vertex, which makes telaprevir.
High discontinuation rate noted for direct-acting antiviral therapy
Despite high rates of early clinical response, 30% of veterans taking direct-acting antivirals for hepatitis C discontinued treatment by week 24.
The findings highlight "the challenges associated with maintenance of these regimens" in a real-world cohort, wrote Pamela S. Belperio, Pharm.D., and colleagues. The study appears in the August issue of Clinical Gastroenterology and Hepatology.
Dr. Belperio of Veterans Affairs Palo Alto (Calif.) Health Care System, looked at 859 patients registered with the VA’s Clinical Case Registry for HCV who initiated triple therapy with pegylated interferon, ribavirin, and either boceprevir (n = 661) or telaprevir (n = 198) before Jan. 1, 2012, at 94 different VA facilities.
The authors determined how long the patient took the drug by looking at medication dispensed dates, and patients were classified as having response rates of "undetectable" if HCV RNA levels at their most recent assay were undetectable.
Patients’ mean age was 57 years; most of the patients were male. Patients with HIV or hepatitis B virus coinfection, hepatocellular carcinoma, or liver transplantation were excluded.
Even so, "Up to 13% of boceprevir-treated veterans and 24% of telaprevir-treated veterans would have been excluded from the phase III trials [of these therapies] based on hematologic exclusion criteria used in the telaprevir trials," Dr. Belperio wrote.
Despite this, the authors found that by week 12, 76% of all treatment-naive, noncirrhotic patients taking boceprevir had achieved undetectable viral loads, as had 78% of telaprevir patients.
By 24 weeks, those numbers were 74% for boceprevir patients and 60% for telaprevir patients.
However, the researchers also found that about one-third of patients discontinued treatment with either boceprevir or telaprevir before 24 weeks (30% and 34%, respectively; P = .37), with an incidence of hematologic adverse events that was both higher and more pronounced than has been reported in clinical trials of direct-acting antivirals (DAAs), wrote Dr. Belperio.
Indeed, anemia of at least grade 1 (hemoglobin below normal limits but greater than or equal to 10 g/dL) occurred in 50% of patients in both DAA groups, "which is up to a 15% increased incidence of anemia over what has been reported elsewhere."
There was also significant thrombocytopenia for both drugs, which Dr. Belperio said was four times higher than in clinical trials. In fact, her group reported that 66% of patients taking boceprevir had grade 1 thrombocytopenia. (platelets below normal limits but greater than or equal to 75,000/mm3), as did 59% of patients taking telaprevir.
"This may reflect the greater proportion of cirrhotic patients in our cohort compared with the clinical trials; however, it is concerning given the limited strategies currently available to manage thrombocytopenia and the unknown effect that PEG dose reductions may have on sustained virologic response in the context of DAA-based therapies," they wrote.
There also was a portion of patients for whom treatment was determined to be futile, according to Food and Drug Administration specifications: 9% of patients receiving boceprevir at week 12 and 5% at week 24, as well as 6% of telaprevir patients at week 4, 4% at week 12, and 7% at week 24.
The authors conceded several limitations to this study, including that they were unable to assess reasons for early treatment discontinuation.
Nevertheless, the data "offer clinicians a perspective on expectations of early response and safety of DAA regimens in routine clinical practice, allowing a more nuanced discussion between providers and patients regarding the risks and benefits of embarking on DAA-based treatment."
The authors disclosed that one coinvestigator has previously received grant support from Gilead Sciences, maker of HCV therapies. They disclosed no other conflicts of interest.
Despite high rates of early clinical response, 30% of veterans taking direct-acting antivirals for hepatitis C discontinued treatment by week 24.
The findings highlight "the challenges associated with maintenance of these regimens" in a real-world cohort, wrote Pamela S. Belperio, Pharm.D., and colleagues. The study appears in the August issue of Clinical Gastroenterology and Hepatology.
Dr. Belperio of Veterans Affairs Palo Alto (Calif.) Health Care System, looked at 859 patients registered with the VA’s Clinical Case Registry for HCV who initiated triple therapy with pegylated interferon, ribavirin, and either boceprevir (n = 661) or telaprevir (n = 198) before Jan. 1, 2012, at 94 different VA facilities.
The authors determined how long the patient took the drug by looking at medication dispensed dates, and patients were classified as having response rates of "undetectable" if HCV RNA levels at their most recent assay were undetectable.
Patients’ mean age was 57 years; most of the patients were male. Patients with HIV or hepatitis B virus coinfection, hepatocellular carcinoma, or liver transplantation were excluded.
Even so, "Up to 13% of boceprevir-treated veterans and 24% of telaprevir-treated veterans would have been excluded from the phase III trials [of these therapies] based on hematologic exclusion criteria used in the telaprevir trials," Dr. Belperio wrote.
Despite this, the authors found that by week 12, 76% of all treatment-naive, noncirrhotic patients taking boceprevir had achieved undetectable viral loads, as had 78% of telaprevir patients.
By 24 weeks, those numbers were 74% for boceprevir patients and 60% for telaprevir patients.
However, the researchers also found that about one-third of patients discontinued treatment with either boceprevir or telaprevir before 24 weeks (30% and 34%, respectively; P = .37), with an incidence of hematologic adverse events that was both higher and more pronounced than has been reported in clinical trials of direct-acting antivirals (DAAs), wrote Dr. Belperio.
Indeed, anemia of at least grade 1 (hemoglobin below normal limits but greater than or equal to 10 g/dL) occurred in 50% of patients in both DAA groups, "which is up to a 15% increased incidence of anemia over what has been reported elsewhere."
There was also significant thrombocytopenia for both drugs, which Dr. Belperio said was four times higher than in clinical trials. In fact, her group reported that 66% of patients taking boceprevir had grade 1 thrombocytopenia. (platelets below normal limits but greater than or equal to 75,000/mm3), as did 59% of patients taking telaprevir.
"This may reflect the greater proportion of cirrhotic patients in our cohort compared with the clinical trials; however, it is concerning given the limited strategies currently available to manage thrombocytopenia and the unknown effect that PEG dose reductions may have on sustained virologic response in the context of DAA-based therapies," they wrote.
There also was a portion of patients for whom treatment was determined to be futile, according to Food and Drug Administration specifications: 9% of patients receiving boceprevir at week 12 and 5% at week 24, as well as 6% of telaprevir patients at week 4, 4% at week 12, and 7% at week 24.
The authors conceded several limitations to this study, including that they were unable to assess reasons for early treatment discontinuation.
Nevertheless, the data "offer clinicians a perspective on expectations of early response and safety of DAA regimens in routine clinical practice, allowing a more nuanced discussion between providers and patients regarding the risks and benefits of embarking on DAA-based treatment."
The authors disclosed that one coinvestigator has previously received grant support from Gilead Sciences, maker of HCV therapies. They disclosed no other conflicts of interest.
Despite high rates of early clinical response, 30% of veterans taking direct-acting antivirals for hepatitis C discontinued treatment by week 24.
The findings highlight "the challenges associated with maintenance of these regimens" in a real-world cohort, wrote Pamela S. Belperio, Pharm.D., and colleagues. The study appears in the August issue of Clinical Gastroenterology and Hepatology.
Dr. Belperio of Veterans Affairs Palo Alto (Calif.) Health Care System, looked at 859 patients registered with the VA’s Clinical Case Registry for HCV who initiated triple therapy with pegylated interferon, ribavirin, and either boceprevir (n = 661) or telaprevir (n = 198) before Jan. 1, 2012, at 94 different VA facilities.
The authors determined how long the patient took the drug by looking at medication dispensed dates, and patients were classified as having response rates of "undetectable" if HCV RNA levels at their most recent assay were undetectable.
Patients’ mean age was 57 years; most of the patients were male. Patients with HIV or hepatitis B virus coinfection, hepatocellular carcinoma, or liver transplantation were excluded.
Even so, "Up to 13% of boceprevir-treated veterans and 24% of telaprevir-treated veterans would have been excluded from the phase III trials [of these therapies] based on hematologic exclusion criteria used in the telaprevir trials," Dr. Belperio wrote.
Despite this, the authors found that by week 12, 76% of all treatment-naive, noncirrhotic patients taking boceprevir had achieved undetectable viral loads, as had 78% of telaprevir patients.
By 24 weeks, those numbers were 74% for boceprevir patients and 60% for telaprevir patients.
However, the researchers also found that about one-third of patients discontinued treatment with either boceprevir or telaprevir before 24 weeks (30% and 34%, respectively; P = .37), with an incidence of hematologic adverse events that was both higher and more pronounced than has been reported in clinical trials of direct-acting antivirals (DAAs), wrote Dr. Belperio.
Indeed, anemia of at least grade 1 (hemoglobin below normal limits but greater than or equal to 10 g/dL) occurred in 50% of patients in both DAA groups, "which is up to a 15% increased incidence of anemia over what has been reported elsewhere."
There was also significant thrombocytopenia for both drugs, which Dr. Belperio said was four times higher than in clinical trials. In fact, her group reported that 66% of patients taking boceprevir had grade 1 thrombocytopenia. (platelets below normal limits but greater than or equal to 75,000/mm3), as did 59% of patients taking telaprevir.
"This may reflect the greater proportion of cirrhotic patients in our cohort compared with the clinical trials; however, it is concerning given the limited strategies currently available to manage thrombocytopenia and the unknown effect that PEG dose reductions may have on sustained virologic response in the context of DAA-based therapies," they wrote.
There also was a portion of patients for whom treatment was determined to be futile, according to Food and Drug Administration specifications: 9% of patients receiving boceprevir at week 12 and 5% at week 24, as well as 6% of telaprevir patients at week 4, 4% at week 12, and 7% at week 24.
The authors conceded several limitations to this study, including that they were unable to assess reasons for early treatment discontinuation.
Nevertheless, the data "offer clinicians a perspective on expectations of early response and safety of DAA regimens in routine clinical practice, allowing a more nuanced discussion between providers and patients regarding the risks and benefits of embarking on DAA-based treatment."
The authors disclosed that one coinvestigator has previously received grant support from Gilead Sciences, maker of HCV therapies. They disclosed no other conflicts of interest.
Major finding: By 24 weeks, 74% of hepatitis C patients taking boceprevir and 60% taking telaprevir had an early virologic response, but nearly one-third had discontinued treatment.
Data source: A cohort of 859 HCV patients from the Veterans Affairs Clinical Case Registry for HCV.
Disclosures: The authors disclosed that one coinvestigator has previously received grant support from Gilead Sciences, maker of HCV therapies. They disclosed no other conflicts of interest.
‘HCC-4 risk score’ IDs hepatitis C patients likely to develop HCC
A risk score derived from four simple test results readily obtained during routine care may help identify the patients with chronic hepatitis C who are most at risk for developing hepatocellular carcinoma, according to a retrospective study published online July 12 in the European Journal of Internal Medicine.
The score could enable physicians to target only the highest-risk patients for annual surveillance for malignant hepatic nodules, which is crucial because current screening methods are too invasive, too expensive, and too low-yield to be applied broadly across all risk groups.
The new risk score also may help identify patients with chronic hepatitis C who are at lowest risk for developing HCC, who can then be reassured that they can safely forgo invasive and expensive surveillance, reported Dr. Juan Carlos Gavilan and his associates at University Hospital Virgin de la Victoria, Malaga (Spain).
The investigators reviewed data from a 17-year longitudinal cohort study involving 829 patients with chronic hepatitis C. These subjects were assessed every 6 months for the development of HCC using serum alpha-fetoprotein (AFP) levels and ultrasound imaging to detect new focal hepatic lesions.
A total of 58 subjects (7%) developed HCC during follow-up.
An initial univariate analysis identified numerous clinical and epidemiologic factors associated with elevated risk for HCC. The researchers constructed a formula for predicting risk using the four independent factors that were most predictive of HCC in this cohort: patient age, platelet count, gamma-globulin level, and AFP level at baseline.
By dividing the study population into tertiles, Dr. Gavilan and his colleagues established cutoff ranges for low, medium, and high risk. They then classified each study participant as belonging to one of these three categories, to see how well this risk score correlated with the actual rates of HCC.
The annual incidence of HCC was 0.06% in the group designated as low risk, 0.5% in the group designated as medium risk, and 2.6% in the group designated as high risk, indicating that this "HCC-4 risk score" was indeed highly predictive, Dr. Gavilan and his associates said (Eur. J. Intern. Med. 2013 July 12 [doi: 10.1016/j.ejim.2013.06.010]).
In fact, the score was more accurate at predicting HCC than was the commonly used fibrosis index, they noted.
According to recently published recommendations, surveillance is only justified in populations with an HCC incidence of 1.5% or more per year. Thus, patients found to be high risk using this HCC-4 risk score would be appropriate for such surveillance, while those at medium or low risk would not be.
"These results must be confirmed in other studies," the investigators said.
There was no external funding source for this study, and no financial conflicts of interest were reported.
A risk score derived from four simple test results readily obtained during routine care may help identify the patients with chronic hepatitis C who are most at risk for developing hepatocellular carcinoma, according to a retrospective study published online July 12 in the European Journal of Internal Medicine.
The score could enable physicians to target only the highest-risk patients for annual surveillance for malignant hepatic nodules, which is crucial because current screening methods are too invasive, too expensive, and too low-yield to be applied broadly across all risk groups.
The new risk score also may help identify patients with chronic hepatitis C who are at lowest risk for developing HCC, who can then be reassured that they can safely forgo invasive and expensive surveillance, reported Dr. Juan Carlos Gavilan and his associates at University Hospital Virgin de la Victoria, Malaga (Spain).
The investigators reviewed data from a 17-year longitudinal cohort study involving 829 patients with chronic hepatitis C. These subjects were assessed every 6 months for the development of HCC using serum alpha-fetoprotein (AFP) levels and ultrasound imaging to detect new focal hepatic lesions.
A total of 58 subjects (7%) developed HCC during follow-up.
An initial univariate analysis identified numerous clinical and epidemiologic factors associated with elevated risk for HCC. The researchers constructed a formula for predicting risk using the four independent factors that were most predictive of HCC in this cohort: patient age, platelet count, gamma-globulin level, and AFP level at baseline.
By dividing the study population into tertiles, Dr. Gavilan and his colleagues established cutoff ranges for low, medium, and high risk. They then classified each study participant as belonging to one of these three categories, to see how well this risk score correlated with the actual rates of HCC.
The annual incidence of HCC was 0.06% in the group designated as low risk, 0.5% in the group designated as medium risk, and 2.6% in the group designated as high risk, indicating that this "HCC-4 risk score" was indeed highly predictive, Dr. Gavilan and his associates said (Eur. J. Intern. Med. 2013 July 12 [doi: 10.1016/j.ejim.2013.06.010]).
In fact, the score was more accurate at predicting HCC than was the commonly used fibrosis index, they noted.
According to recently published recommendations, surveillance is only justified in populations with an HCC incidence of 1.5% or more per year. Thus, patients found to be high risk using this HCC-4 risk score would be appropriate for such surveillance, while those at medium or low risk would not be.
"These results must be confirmed in other studies," the investigators said.
There was no external funding source for this study, and no financial conflicts of interest were reported.
A risk score derived from four simple test results readily obtained during routine care may help identify the patients with chronic hepatitis C who are most at risk for developing hepatocellular carcinoma, according to a retrospective study published online July 12 in the European Journal of Internal Medicine.
The score could enable physicians to target only the highest-risk patients for annual surveillance for malignant hepatic nodules, which is crucial because current screening methods are too invasive, too expensive, and too low-yield to be applied broadly across all risk groups.
The new risk score also may help identify patients with chronic hepatitis C who are at lowest risk for developing HCC, who can then be reassured that they can safely forgo invasive and expensive surveillance, reported Dr. Juan Carlos Gavilan and his associates at University Hospital Virgin de la Victoria, Malaga (Spain).
The investigators reviewed data from a 17-year longitudinal cohort study involving 829 patients with chronic hepatitis C. These subjects were assessed every 6 months for the development of HCC using serum alpha-fetoprotein (AFP) levels and ultrasound imaging to detect new focal hepatic lesions.
A total of 58 subjects (7%) developed HCC during follow-up.
An initial univariate analysis identified numerous clinical and epidemiologic factors associated with elevated risk for HCC. The researchers constructed a formula for predicting risk using the four independent factors that were most predictive of HCC in this cohort: patient age, platelet count, gamma-globulin level, and AFP level at baseline.
By dividing the study population into tertiles, Dr. Gavilan and his colleagues established cutoff ranges for low, medium, and high risk. They then classified each study participant as belonging to one of these three categories, to see how well this risk score correlated with the actual rates of HCC.
The annual incidence of HCC was 0.06% in the group designated as low risk, 0.5% in the group designated as medium risk, and 2.6% in the group designated as high risk, indicating that this "HCC-4 risk score" was indeed highly predictive, Dr. Gavilan and his associates said (Eur. J. Intern. Med. 2013 July 12 [doi: 10.1016/j.ejim.2013.06.010]).
In fact, the score was more accurate at predicting HCC than was the commonly used fibrosis index, they noted.
According to recently published recommendations, surveillance is only justified in populations with an HCC incidence of 1.5% or more per year. Thus, patients found to be high risk using this HCC-4 risk score would be appropriate for such surveillance, while those at medium or low risk would not be.
"These results must be confirmed in other studies," the investigators said.
There was no external funding source for this study, and no financial conflicts of interest were reported.
FROM THE EUROPEAN JOURNAL OF INTERNAL MEDICINE
Major finding: The annual incidence of HCC was 0.06% in the group designated as low risk, 0.5% in the group designated as medium risk, and 2.6% in the group designated as high risk, indicating that the HCC-4 risk score was highly predictive of actual outcomes.
Data source: A retrospective study of data collected in a 17-year longitudinal cohort study involving 829 adults with chronic hepatitis C, of whom 58 developed HCC.
Disclosures: There was no external funding source for this study, and no financial conflicts of interest were reported.
FDA issues strong warning about oral ketoconazole
Ketoconazole tablets should not be used as a first-line treatment for fungal infections because treatment has been associated with an increased risk of adrenal insufficiency, potentially fatal hepatotoxicity, and drug interactions, the Food and Drug Administration has announced.
Marketed as Nizoral, oral ketoconazole is no longer indicated for the treatment of Candida and dermatophyte infections and "should be used only for the treatment of certain life-threatening mycoses when the potential benefits outweigh the risks and alternative therapeutic options are not available or tolerated," according to the MedWatch safety alert issued on July 26.
In addition, oral ketoconazole should not be used to treat fungal infections of the skin and nails, and is only indicated for the treatment of blastomycosis, coccidioidomycosis, histoplasmosis, chromomycosis, and paracoccidioidomycosis "in patients in whom other treatments have failed or who are intolerant to other therapies," according to the label, which has been modified to reflect these risks and recommendations.
There is now a Medication Guide that will be provided to patients with each filled prescription of oral ketoconazole, explaining the risks.
Because oral ketoconazole has been associated with hepatoxicity that can result in liver transplantation or death, it is now contraindicated in patients with acute or chronic liver disease. The label also now recommends that patients be assessed and monitored for liver toxicity. Monitoring of adrenal function also is now recommended in patients who take the oral formulation of the drug and have adrenal problems or "are under prolonged periods of stress such as those who have had a recent major surgery or who are under intensive care in the hospital."
In addition, coadministration of ketoconazole – a potent inhibitor of the cytochrome P450 3A4 isoenzyme (CYP3A4) – with certain drugs is either restricted or contraindicated because of the increase in drug concentrations and increased risk of QT prolongation and other serious reactions. Contraindicated drugs include dofetilide, quinidine, pimozide, and cisapride.
The FDA changes are based on risk-benefit analyses of data that include reports made to the FDA’s Adverse Events Reporting System.
On July 26, the European Medicines Agency’s Committee on Medicinal Products for Human Use (CHMP) announced that it has concluded that the risk of hepatoxicity with oral ketoconazole products was greater than the benefits in treating fungal infections and recommended that these products no longer be marketed in the European Union.
Creams, shampoos, and other topical ketoconazole formulations have not been associated with these problems, according to the FDA.
The updated label is available here. Serious adverse events associated with ketoconazole should be reported to the FDA at 800-332-1088 or MedWatch.
Ketoconazole tablets should not be used as a first-line treatment for fungal infections because treatment has been associated with an increased risk of adrenal insufficiency, potentially fatal hepatotoxicity, and drug interactions, the Food and Drug Administration has announced.
Marketed as Nizoral, oral ketoconazole is no longer indicated for the treatment of Candida and dermatophyte infections and "should be used only for the treatment of certain life-threatening mycoses when the potential benefits outweigh the risks and alternative therapeutic options are not available or tolerated," according to the MedWatch safety alert issued on July 26.
In addition, oral ketoconazole should not be used to treat fungal infections of the skin and nails, and is only indicated for the treatment of blastomycosis, coccidioidomycosis, histoplasmosis, chromomycosis, and paracoccidioidomycosis "in patients in whom other treatments have failed or who are intolerant to other therapies," according to the label, which has been modified to reflect these risks and recommendations.
There is now a Medication Guide that will be provided to patients with each filled prescription of oral ketoconazole, explaining the risks.
Because oral ketoconazole has been associated with hepatoxicity that can result in liver transplantation or death, it is now contraindicated in patients with acute or chronic liver disease. The label also now recommends that patients be assessed and monitored for liver toxicity. Monitoring of adrenal function also is now recommended in patients who take the oral formulation of the drug and have adrenal problems or "are under prolonged periods of stress such as those who have had a recent major surgery or who are under intensive care in the hospital."
In addition, coadministration of ketoconazole – a potent inhibitor of the cytochrome P450 3A4 isoenzyme (CYP3A4) – with certain drugs is either restricted or contraindicated because of the increase in drug concentrations and increased risk of QT prolongation and other serious reactions. Contraindicated drugs include dofetilide, quinidine, pimozide, and cisapride.
The FDA changes are based on risk-benefit analyses of data that include reports made to the FDA’s Adverse Events Reporting System.
On July 26, the European Medicines Agency’s Committee on Medicinal Products for Human Use (CHMP) announced that it has concluded that the risk of hepatoxicity with oral ketoconazole products was greater than the benefits in treating fungal infections and recommended that these products no longer be marketed in the European Union.
Creams, shampoos, and other topical ketoconazole formulations have not been associated with these problems, according to the FDA.
The updated label is available here. Serious adverse events associated with ketoconazole should be reported to the FDA at 800-332-1088 or MedWatch.
Ketoconazole tablets should not be used as a first-line treatment for fungal infections because treatment has been associated with an increased risk of adrenal insufficiency, potentially fatal hepatotoxicity, and drug interactions, the Food and Drug Administration has announced.
Marketed as Nizoral, oral ketoconazole is no longer indicated for the treatment of Candida and dermatophyte infections and "should be used only for the treatment of certain life-threatening mycoses when the potential benefits outweigh the risks and alternative therapeutic options are not available or tolerated," according to the MedWatch safety alert issued on July 26.
In addition, oral ketoconazole should not be used to treat fungal infections of the skin and nails, and is only indicated for the treatment of blastomycosis, coccidioidomycosis, histoplasmosis, chromomycosis, and paracoccidioidomycosis "in patients in whom other treatments have failed or who are intolerant to other therapies," according to the label, which has been modified to reflect these risks and recommendations.
There is now a Medication Guide that will be provided to patients with each filled prescription of oral ketoconazole, explaining the risks.
Because oral ketoconazole has been associated with hepatoxicity that can result in liver transplantation or death, it is now contraindicated in patients with acute or chronic liver disease. The label also now recommends that patients be assessed and monitored for liver toxicity. Monitoring of adrenal function also is now recommended in patients who take the oral formulation of the drug and have adrenal problems or "are under prolonged periods of stress such as those who have had a recent major surgery or who are under intensive care in the hospital."
In addition, coadministration of ketoconazole – a potent inhibitor of the cytochrome P450 3A4 isoenzyme (CYP3A4) – with certain drugs is either restricted or contraindicated because of the increase in drug concentrations and increased risk of QT prolongation and other serious reactions. Contraindicated drugs include dofetilide, quinidine, pimozide, and cisapride.
The FDA changes are based on risk-benefit analyses of data that include reports made to the FDA’s Adverse Events Reporting System.
On July 26, the European Medicines Agency’s Committee on Medicinal Products for Human Use (CHMP) announced that it has concluded that the risk of hepatoxicity with oral ketoconazole products was greater than the benefits in treating fungal infections and recommended that these products no longer be marketed in the European Union.
Creams, shampoos, and other topical ketoconazole formulations have not been associated with these problems, according to the FDA.
The updated label is available here. Serious adverse events associated with ketoconazole should be reported to the FDA at 800-332-1088 or MedWatch.
Coinfection with chronic hepatitis C and occult hepatitis B related to poor outcomes
Chronic hepatitis C patients who are coinfected with occult hepatitis B virus face a high risk of progression toward cirrhosis, the development of hepatocellular carcinoma, and lower survival, an observational study demonstrated.
To evaluate the clinical evolution of chronic hepatitis C (CHC) patients according to their occult hepatitis B virus infection (OBI) status, 326 hepatitis B surface antigen–negative CHC patients who had undergone needle liver biopsy in the liver unit at the University of Messina (Italy) between 1991 and 2000 were tested for OBI by analysis of liver biopsy DNA extracts. More than half of the patients (65%) were male, and their median age was 52 years, researchers led by Dr. Giovanni Squadrito and Dr. Giovanni Raimondo of the University of Messina reported in an article in press from the Journal of Hepatology (2013 June 10).
None of them had received any antiviral therapy before liver biopsy was performed, none was infected with HIV, and none had evidence of alcoholic or autoimmune liver disease.
Of the 326 patients, 128 (39%) were OBI positive while the remaining 198 were OBI negative. A total of 94 patients (37 OBI positive and 57 OBI negative) were followed for a median of 11 years. Among these 94 patients, 79 underwent anti-HCV treatments and 26 achieved a sustained virologic response that occurred independently of their OBI status.
The researchers reported that 18 of the 94 patients (19%) developed hepatocellular carcinoma (HCC). Of these, 13 were among the 37 who were OBI positive and 5 were among the 57 who were OBI negative, a difference that was statistically significant (P less than .01). Among the 76 patients who did not develop HCC, 15 (20%) developed advanced forms of cirrhosis. Of these, 8 were among the 24 who were OBI positive and 7 were among the 52 who were OBI negative, a difference that was statistically significant (P less than .05).
During the follow-up period, 18 patients died (19%) and 2 underwent liver transplantation (2%). Of the 18 deaths, 14 occurred among HCC patients and 4, among patients with clear worsening of liver disease. The researchers found that the cumulative survival rate was significantly shorter among OBI-positive patients compared with OBI-negative patients (P = .003), while liver-related deaths occurred significantly more frequently in OBI-positive patients than in OBI-negative patients (P less than .01). In addition, nonresponse to anti-HCV therapy was significantly associated with lower survival (P = .02).
"The pro-oncogenic role of OBI is not surprising, considering that HBV is a major causative agent of liver cancer worldwide and the potential mechanisms whereby overt HBV might induce tumor formation are mostly maintained in the occult status," the researchers wrote. "In addition, when patients who developed HCC were excluded from the analysis, OBI still appears to play a negative role in the CHC outcome since it was significantly associated with the progression toward the severe deterioration and decompensation of the liver disease. Although this association is in accordance with the results of many cross-sectional studies performed in different areas of the world, how OBI may favor (or accelerate) the progression toward cirrhosis of CHC patients is far from being clearly understood."
The investigators noted that there is evidence "both in humans and in animal models that intrahepatic persistence of occult hepadnavirus genomes may produce a very mild but constant liver necroinflammation, and recent reports have shown an association between phases of a rise in ALT levels and reappearance of circulating HBV DNA in patients with chronic hepatitis C and combined occult HBV infection, thus suggesting an active role of transient reactivation of HBV replication in liver cell injury."
The study was supported by grants from the Associazione Italiana per la Ricerca sul Cancro. The researchers said they had no relevant financial disclosures.
Chronic hepatitis C patients who are coinfected with occult hepatitis B virus face a high risk of progression toward cirrhosis, the development of hepatocellular carcinoma, and lower survival, an observational study demonstrated.
To evaluate the clinical evolution of chronic hepatitis C (CHC) patients according to their occult hepatitis B virus infection (OBI) status, 326 hepatitis B surface antigen–negative CHC patients who had undergone needle liver biopsy in the liver unit at the University of Messina (Italy) between 1991 and 2000 were tested for OBI by analysis of liver biopsy DNA extracts. More than half of the patients (65%) were male, and their median age was 52 years, researchers led by Dr. Giovanni Squadrito and Dr. Giovanni Raimondo of the University of Messina reported in an article in press from the Journal of Hepatology (2013 June 10).
None of them had received any antiviral therapy before liver biopsy was performed, none was infected with HIV, and none had evidence of alcoholic or autoimmune liver disease.
Of the 326 patients, 128 (39%) were OBI positive while the remaining 198 were OBI negative. A total of 94 patients (37 OBI positive and 57 OBI negative) were followed for a median of 11 years. Among these 94 patients, 79 underwent anti-HCV treatments and 26 achieved a sustained virologic response that occurred independently of their OBI status.
The researchers reported that 18 of the 94 patients (19%) developed hepatocellular carcinoma (HCC). Of these, 13 were among the 37 who were OBI positive and 5 were among the 57 who were OBI negative, a difference that was statistically significant (P less than .01). Among the 76 patients who did not develop HCC, 15 (20%) developed advanced forms of cirrhosis. Of these, 8 were among the 24 who were OBI positive and 7 were among the 52 who were OBI negative, a difference that was statistically significant (P less than .05).
During the follow-up period, 18 patients died (19%) and 2 underwent liver transplantation (2%). Of the 18 deaths, 14 occurred among HCC patients and 4, among patients with clear worsening of liver disease. The researchers found that the cumulative survival rate was significantly shorter among OBI-positive patients compared with OBI-negative patients (P = .003), while liver-related deaths occurred significantly more frequently in OBI-positive patients than in OBI-negative patients (P less than .01). In addition, nonresponse to anti-HCV therapy was significantly associated with lower survival (P = .02).
"The pro-oncogenic role of OBI is not surprising, considering that HBV is a major causative agent of liver cancer worldwide and the potential mechanisms whereby overt HBV might induce tumor formation are mostly maintained in the occult status," the researchers wrote. "In addition, when patients who developed HCC were excluded from the analysis, OBI still appears to play a negative role in the CHC outcome since it was significantly associated with the progression toward the severe deterioration and decompensation of the liver disease. Although this association is in accordance with the results of many cross-sectional studies performed in different areas of the world, how OBI may favor (or accelerate) the progression toward cirrhosis of CHC patients is far from being clearly understood."
The investigators noted that there is evidence "both in humans and in animal models that intrahepatic persistence of occult hepadnavirus genomes may produce a very mild but constant liver necroinflammation, and recent reports have shown an association between phases of a rise in ALT levels and reappearance of circulating HBV DNA in patients with chronic hepatitis C and combined occult HBV infection, thus suggesting an active role of transient reactivation of HBV replication in liver cell injury."
The study was supported by grants from the Associazione Italiana per la Ricerca sul Cancro. The researchers said they had no relevant financial disclosures.
Chronic hepatitis C patients who are coinfected with occult hepatitis B virus face a high risk of progression toward cirrhosis, the development of hepatocellular carcinoma, and lower survival, an observational study demonstrated.
To evaluate the clinical evolution of chronic hepatitis C (CHC) patients according to their occult hepatitis B virus infection (OBI) status, 326 hepatitis B surface antigen–negative CHC patients who had undergone needle liver biopsy in the liver unit at the University of Messina (Italy) between 1991 and 2000 were tested for OBI by analysis of liver biopsy DNA extracts. More than half of the patients (65%) were male, and their median age was 52 years, researchers led by Dr. Giovanni Squadrito and Dr. Giovanni Raimondo of the University of Messina reported in an article in press from the Journal of Hepatology (2013 June 10).
None of them had received any antiviral therapy before liver biopsy was performed, none was infected with HIV, and none had evidence of alcoholic or autoimmune liver disease.
Of the 326 patients, 128 (39%) were OBI positive while the remaining 198 were OBI negative. A total of 94 patients (37 OBI positive and 57 OBI negative) were followed for a median of 11 years. Among these 94 patients, 79 underwent anti-HCV treatments and 26 achieved a sustained virologic response that occurred independently of their OBI status.
The researchers reported that 18 of the 94 patients (19%) developed hepatocellular carcinoma (HCC). Of these, 13 were among the 37 who were OBI positive and 5 were among the 57 who were OBI negative, a difference that was statistically significant (P less than .01). Among the 76 patients who did not develop HCC, 15 (20%) developed advanced forms of cirrhosis. Of these, 8 were among the 24 who were OBI positive and 7 were among the 52 who were OBI negative, a difference that was statistically significant (P less than .05).
During the follow-up period, 18 patients died (19%) and 2 underwent liver transplantation (2%). Of the 18 deaths, 14 occurred among HCC patients and 4, among patients with clear worsening of liver disease. The researchers found that the cumulative survival rate was significantly shorter among OBI-positive patients compared with OBI-negative patients (P = .003), while liver-related deaths occurred significantly more frequently in OBI-positive patients than in OBI-negative patients (P less than .01). In addition, nonresponse to anti-HCV therapy was significantly associated with lower survival (P = .02).
"The pro-oncogenic role of OBI is not surprising, considering that HBV is a major causative agent of liver cancer worldwide and the potential mechanisms whereby overt HBV might induce tumor formation are mostly maintained in the occult status," the researchers wrote. "In addition, when patients who developed HCC were excluded from the analysis, OBI still appears to play a negative role in the CHC outcome since it was significantly associated with the progression toward the severe deterioration and decompensation of the liver disease. Although this association is in accordance with the results of many cross-sectional studies performed in different areas of the world, how OBI may favor (or accelerate) the progression toward cirrhosis of CHC patients is far from being clearly understood."
The investigators noted that there is evidence "both in humans and in animal models that intrahepatic persistence of occult hepadnavirus genomes may produce a very mild but constant liver necroinflammation, and recent reports have shown an association between phases of a rise in ALT levels and reappearance of circulating HBV DNA in patients with chronic hepatitis C and combined occult HBV infection, thus suggesting an active role of transient reactivation of HBV replication in liver cell injury."
The study was supported by grants from the Associazione Italiana per la Ricerca sul Cancro. The researchers said they had no relevant financial disclosures.
FROM THE JOURNAL OF HEPATOLOGY
Major finding: Among chronic hepatitis patients followed for a median of 11 years, 19% developed hepatocellular carcinoma. A significantly greater proportion of patients who were positive for occult hepatitis virus infection developed HCC compared with those who were occult hepatitis B negative (P value of less than .01).
Data source: An observational study of 326 hepatitis B surface antigen–negative chronic hepatitis patients who were tested for occult hepatitis B virus infection by the analysis of liver biopsy DNA extracts.
Disclosures: The study was supported by grants from the Associazione Italiana per la Ricerca sul Cancro. The researchers said they had no relevant financial disclosures.
Boceprevir an option for patients with HIV and HCV
Among adults infected with both HIV and hepatitis C virus, the addition of boceprevir to a combination of peginterferon and ribavirin significantly increased the rate of sustained virological response compared with those who received placebo, results from a 44-week, multicenter phase II study demonstrated.
"Rates of SVR at follow-up week 24 were increased with boceprevir in all subgroups of patients, irrespective of demographic or baseline characteristics," researchers led by Dr. Mark S. Sulkowski of Johns Hopkins University, Baltimore, reported in the July issue of the Lancet Infectious Diseases. Although SVR after treatment with peginterferon and ribavirin has been associated with improved survival, "this regimen has been relatively ineffective (SVR, 25%-30%) in patients with HIV and HCV genotype 1 infection," they wrote.
"Addition of an HCV protease inhibitor (boceprevir or telaprevir) to peginterferon-ribavirin has emerged as the standard of care for the treatment of HCV genotype 1 infection alone. Use of HCV protease inhibitors in patients infected with HIV and HCV has been restricted by the dearth of available safety and efficacy data."
For the study, which was conducted between Jan. 15 and Dec. 29, 2010, at 30 academic and nonacademic study sites, 99 adults with untreated HCV type 1 genotype infection and controlled HIV were randomized in 1:2 fashion to receive peginterferon 1.5 mcg/kg per week with weight-based ribavirin (600-1,400 mg/day) for 4 weeks, followed by peginterferon-ribavirin plus either placebo (control group) or 800 mg of boceprevir three times daily (boceprevir group) for 44 weeks (Lancet Infect. Dis. 2013;13:597-605).
The primary endpoint of interest was sustained virological response (defined as undetectable plasma HCV RNA) at follow-up week 24 after the end of treatment.
Of the 99 patients, 98 received at least one treatment dose. Of these, 64 were in the boceprevir group and 34 were in the control group. The researchers reported that at follow-up week 24, 63% of patients in the boceprevir group achieved SVR, compared with 29% of controls, a difference of 33.1% that reached significance (P = .0008). Compared with controls, a higher proportion of the boceprevir group experienced adverse events, including anemia (41% vs. 26%, respectively), pyrexia (36% vs. 21%), decreased appetite (34% vs. 18%), dysgeusia (28% vs. 15%), vomiting (28% vs. 15%), and neutropenia (19% vs. 6%). In addition, 4 patients in the control group and 3 in the boceprevir group had HIV viral breakthrough, defined as two consecutive HIV RNA values of at least 50 copies/mL.
"Boceprevir did not seem to add significant risk when used in combination with peginterferon-ribavirin therapy, and no new adverse events were reported," the researchers wrote. "HIV control was well maintained in patients taking boceprevir and HIV protease inhibitors. Drug interactions between boceprevir and HIV protease inhibitors did not have a clinically significant effect on HCV response or HIV control in this study. In view of the hepatic and extrahepatic benefits associated with SVR at follow-up week 24 in patients infected with HIV and HCV, boceprevir in combination with peginterferon-ribavirin might be an important therapeutic option for patients with such coinfection."
Merck markets boceprevir under the brand name Victrelis. Merck sponsored and funded the study. Dr. Sulkowski and numerous other coauthors disclosed having received consulting fees and research grants from the company.
Among adults infected with both HIV and hepatitis C virus, the addition of boceprevir to a combination of peginterferon and ribavirin significantly increased the rate of sustained virological response compared with those who received placebo, results from a 44-week, multicenter phase II study demonstrated.
"Rates of SVR at follow-up week 24 were increased with boceprevir in all subgroups of patients, irrespective of demographic or baseline characteristics," researchers led by Dr. Mark S. Sulkowski of Johns Hopkins University, Baltimore, reported in the July issue of the Lancet Infectious Diseases. Although SVR after treatment with peginterferon and ribavirin has been associated with improved survival, "this regimen has been relatively ineffective (SVR, 25%-30%) in patients with HIV and HCV genotype 1 infection," they wrote.
"Addition of an HCV protease inhibitor (boceprevir or telaprevir) to peginterferon-ribavirin has emerged as the standard of care for the treatment of HCV genotype 1 infection alone. Use of HCV protease inhibitors in patients infected with HIV and HCV has been restricted by the dearth of available safety and efficacy data."
For the study, which was conducted between Jan. 15 and Dec. 29, 2010, at 30 academic and nonacademic study sites, 99 adults with untreated HCV type 1 genotype infection and controlled HIV were randomized in 1:2 fashion to receive peginterferon 1.5 mcg/kg per week with weight-based ribavirin (600-1,400 mg/day) for 4 weeks, followed by peginterferon-ribavirin plus either placebo (control group) or 800 mg of boceprevir three times daily (boceprevir group) for 44 weeks (Lancet Infect. Dis. 2013;13:597-605).
The primary endpoint of interest was sustained virological response (defined as undetectable plasma HCV RNA) at follow-up week 24 after the end of treatment.
Of the 99 patients, 98 received at least one treatment dose. Of these, 64 were in the boceprevir group and 34 were in the control group. The researchers reported that at follow-up week 24, 63% of patients in the boceprevir group achieved SVR, compared with 29% of controls, a difference of 33.1% that reached significance (P = .0008). Compared with controls, a higher proportion of the boceprevir group experienced adverse events, including anemia (41% vs. 26%, respectively), pyrexia (36% vs. 21%), decreased appetite (34% vs. 18%), dysgeusia (28% vs. 15%), vomiting (28% vs. 15%), and neutropenia (19% vs. 6%). In addition, 4 patients in the control group and 3 in the boceprevir group had HIV viral breakthrough, defined as two consecutive HIV RNA values of at least 50 copies/mL.
"Boceprevir did not seem to add significant risk when used in combination with peginterferon-ribavirin therapy, and no new adverse events were reported," the researchers wrote. "HIV control was well maintained in patients taking boceprevir and HIV protease inhibitors. Drug interactions between boceprevir and HIV protease inhibitors did not have a clinically significant effect on HCV response or HIV control in this study. In view of the hepatic and extrahepatic benefits associated with SVR at follow-up week 24 in patients infected with HIV and HCV, boceprevir in combination with peginterferon-ribavirin might be an important therapeutic option for patients with such coinfection."
Merck markets boceprevir under the brand name Victrelis. Merck sponsored and funded the study. Dr. Sulkowski and numerous other coauthors disclosed having received consulting fees and research grants from the company.
Among adults infected with both HIV and hepatitis C virus, the addition of boceprevir to a combination of peginterferon and ribavirin significantly increased the rate of sustained virological response compared with those who received placebo, results from a 44-week, multicenter phase II study demonstrated.
"Rates of SVR at follow-up week 24 were increased with boceprevir in all subgroups of patients, irrespective of demographic or baseline characteristics," researchers led by Dr. Mark S. Sulkowski of Johns Hopkins University, Baltimore, reported in the July issue of the Lancet Infectious Diseases. Although SVR after treatment with peginterferon and ribavirin has been associated with improved survival, "this regimen has been relatively ineffective (SVR, 25%-30%) in patients with HIV and HCV genotype 1 infection," they wrote.
"Addition of an HCV protease inhibitor (boceprevir or telaprevir) to peginterferon-ribavirin has emerged as the standard of care for the treatment of HCV genotype 1 infection alone. Use of HCV protease inhibitors in patients infected with HIV and HCV has been restricted by the dearth of available safety and efficacy data."
For the study, which was conducted between Jan. 15 and Dec. 29, 2010, at 30 academic and nonacademic study sites, 99 adults with untreated HCV type 1 genotype infection and controlled HIV were randomized in 1:2 fashion to receive peginterferon 1.5 mcg/kg per week with weight-based ribavirin (600-1,400 mg/day) for 4 weeks, followed by peginterferon-ribavirin plus either placebo (control group) or 800 mg of boceprevir three times daily (boceprevir group) for 44 weeks (Lancet Infect. Dis. 2013;13:597-605).
The primary endpoint of interest was sustained virological response (defined as undetectable plasma HCV RNA) at follow-up week 24 after the end of treatment.
Of the 99 patients, 98 received at least one treatment dose. Of these, 64 were in the boceprevir group and 34 were in the control group. The researchers reported that at follow-up week 24, 63% of patients in the boceprevir group achieved SVR, compared with 29% of controls, a difference of 33.1% that reached significance (P = .0008). Compared with controls, a higher proportion of the boceprevir group experienced adverse events, including anemia (41% vs. 26%, respectively), pyrexia (36% vs. 21%), decreased appetite (34% vs. 18%), dysgeusia (28% vs. 15%), vomiting (28% vs. 15%), and neutropenia (19% vs. 6%). In addition, 4 patients in the control group and 3 in the boceprevir group had HIV viral breakthrough, defined as two consecutive HIV RNA values of at least 50 copies/mL.
"Boceprevir did not seem to add significant risk when used in combination with peginterferon-ribavirin therapy, and no new adverse events were reported," the researchers wrote. "HIV control was well maintained in patients taking boceprevir and HIV protease inhibitors. Drug interactions between boceprevir and HIV protease inhibitors did not have a clinically significant effect on HCV response or HIV control in this study. In view of the hepatic and extrahepatic benefits associated with SVR at follow-up week 24 in patients infected with HIV and HCV, boceprevir in combination with peginterferon-ribavirin might be an important therapeutic option for patients with such coinfection."
Merck markets boceprevir under the brand name Victrelis. Merck sponsored and funded the study. Dr. Sulkowski and numerous other coauthors disclosed having received consulting fees and research grants from the company.
FROM THE LANCET INFECTIOUS DISEASES
Major finding: At follow-up week 24, 63% of patients in the boceprevir group achieved sustained virological response, compared with 29% of controls, a difference of 33.1% that reached significance (P = .0008).
Data source: A phase II multicenter trial of 99 patients coinfected with HIV and HCV who were randomized to receive peginterferon 1.5 mcg/kg per week with weight-based ribavirin (600-1,400 mg/day) for 4 weeks, followed by peginterferon-ribavirin plus either placebo (control group) or 800 mg of boceprevir three times daily (boceprevir group) for 44 weeks, with 24 weeks of follow-up after treatment.
Disclosures: Merck markets boceprevir under the brand name Victrelis. Merck sponsored and funded the study. Dr. Sulkowski and numerous other coauthors disclosed having received consulting fees and research grants from the company.
FDA approves first hepatitis C genotype test
The Food and Drug Administration has approved a rapid test to determine the genotype of an infected patient’s hepatitis C virus, allowing physicians to better tailor therapy.
The RealTime HCV Genotype II can differentiate between genotypes 1, 1a, 1b, 2, 3, 4, and 5.
"Along with other clinical factors, the particular type of HCV is an important consideration in aiding health care professionals in determining if and when to initiate treatment and the appropriate type of treatment," said Alberto Gutierrez, Ph.D., director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health, in a statement.
The diagnostic, manufactured by Abbott Molecular, is approved only for individuals known to be chronically infected with HCV. It is not meant to be used as a screening test or to detect HCV in blood, blood products, or tissue donors, said the FDA.
The agency also noted that the RealTime test has not been evaluated in newborns or pediatric patients, or in the immunocompromised.
The Centers for Disease Control and Prevention estimates that 3.2 million Americans are chronically infected with HCV. It is the most common chronic bloodborne infection and the leading cause of liver transplants, according to the CDC.
The CDC recently urged HCV testing for all Americans born between 1945 and 1965. The FDA said it based its approval of the Abbott test partly by assessing its accuracy in differentiating specific HCV viral genotypes, compared with a validated gene sequencing method. The agency said it also reviewed data that demonstrated the relationship between HCV genotype and effectiveness of drug therapy.
The Abbott diagnostic would be ordered after an initial HCV confirmatory test, said the company. The RealTime test runs on an automated platform, "which provides laboratories substantial improvements in workflow efficiency to meet the increased demand," the company said in a statement.
"When patients are identified, determining their specific genotype is important to ensuring they receive the treatment that will prove to be most effective," said Dr. Carol Brosgart of the division of global health at the University of California, San Francisco, in the Abbott statement. "The introduction of this test for broad use in the United States is a significant advancement in helping to address an important public health issue."
On Twitter @aliciaault
The Food and Drug Administration has approved a rapid test to determine the genotype of an infected patient’s hepatitis C virus, allowing physicians to better tailor therapy.
The RealTime HCV Genotype II can differentiate between genotypes 1, 1a, 1b, 2, 3, 4, and 5.
"Along with other clinical factors, the particular type of HCV is an important consideration in aiding health care professionals in determining if and when to initiate treatment and the appropriate type of treatment," said Alberto Gutierrez, Ph.D., director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health, in a statement.
The diagnostic, manufactured by Abbott Molecular, is approved only for individuals known to be chronically infected with HCV. It is not meant to be used as a screening test or to detect HCV in blood, blood products, or tissue donors, said the FDA.
The agency also noted that the RealTime test has not been evaluated in newborns or pediatric patients, or in the immunocompromised.
The Centers for Disease Control and Prevention estimates that 3.2 million Americans are chronically infected with HCV. It is the most common chronic bloodborne infection and the leading cause of liver transplants, according to the CDC.
The CDC recently urged HCV testing for all Americans born between 1945 and 1965. The FDA said it based its approval of the Abbott test partly by assessing its accuracy in differentiating specific HCV viral genotypes, compared with a validated gene sequencing method. The agency said it also reviewed data that demonstrated the relationship between HCV genotype and effectiveness of drug therapy.
The Abbott diagnostic would be ordered after an initial HCV confirmatory test, said the company. The RealTime test runs on an automated platform, "which provides laboratories substantial improvements in workflow efficiency to meet the increased demand," the company said in a statement.
"When patients are identified, determining their specific genotype is important to ensuring they receive the treatment that will prove to be most effective," said Dr. Carol Brosgart of the division of global health at the University of California, San Francisco, in the Abbott statement. "The introduction of this test for broad use in the United States is a significant advancement in helping to address an important public health issue."
On Twitter @aliciaault
The Food and Drug Administration has approved a rapid test to determine the genotype of an infected patient’s hepatitis C virus, allowing physicians to better tailor therapy.
The RealTime HCV Genotype II can differentiate between genotypes 1, 1a, 1b, 2, 3, 4, and 5.
"Along with other clinical factors, the particular type of HCV is an important consideration in aiding health care professionals in determining if and when to initiate treatment and the appropriate type of treatment," said Alberto Gutierrez, Ph.D., director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health, in a statement.
The diagnostic, manufactured by Abbott Molecular, is approved only for individuals known to be chronically infected with HCV. It is not meant to be used as a screening test or to detect HCV in blood, blood products, or tissue donors, said the FDA.
The agency also noted that the RealTime test has not been evaluated in newborns or pediatric patients, or in the immunocompromised.
The Centers for Disease Control and Prevention estimates that 3.2 million Americans are chronically infected with HCV. It is the most common chronic bloodborne infection and the leading cause of liver transplants, according to the CDC.
The CDC recently urged HCV testing for all Americans born between 1945 and 1965. The FDA said it based its approval of the Abbott test partly by assessing its accuracy in differentiating specific HCV viral genotypes, compared with a validated gene sequencing method. The agency said it also reviewed data that demonstrated the relationship between HCV genotype and effectiveness of drug therapy.
The Abbott diagnostic would be ordered after an initial HCV confirmatory test, said the company. The RealTime test runs on an automated platform, "which provides laboratories substantial improvements in workflow efficiency to meet the increased demand," the company said in a statement.
"When patients are identified, determining their specific genotype is important to ensuring they receive the treatment that will prove to be most effective," said Dr. Carol Brosgart of the division of global health at the University of California, San Francisco, in the Abbott statement. "The introduction of this test for broad use in the United States is a significant advancement in helping to address an important public health issue."
On Twitter @aliciaault