User login
Write an exercise Rx to improve patients' cardiorespiratory fitness
It is well-known that per capita health care spending in the United States is more than twice the average in other developed countries1; nevertheless, the overall health care ranking of the US is near the bottom compared to other countries in this group.2 Much of the reason for this poor relative showing lies in the fact that the US has employed a somewhat traditional fee-for-service health care model that does not incentivize efforts to promote health and wellness or prevent chronic disease. The paradigm of promoting physical activity for its disease-preventing and treatment benefits has not been well-integrated in the US health care system.
In this article, we endeavor to provide better understanding of the barriers that keep family physicians from routinely promoting physical activity in clinical practice; define tools and resources that can be used in the clinical setting to promote physical activity; and delineate areas for future work.
Glaring hole in US physical activity education
Many primary care physicians feel underprepared to prescribe or motivate patients to exercise. The reason for that lack of preparedness likely relates to a medical education system that does not spend time preparing physicians to perform this critical task. A study showed that, on average, medical schools require only 8 hours of physical activity education in their curriculum during the 4 years of schooling.3 Likewise, the average primary care residency program offers only 3 hours of didactic training on physical activity, nutrition, and obesity.4 The problem extends to sports medicine fellowship training, in which a 2019 survey showed that 63% of fellows were never taught how to write an exercise prescription in their training program.5
Without education on physical activity, medical students, residents, and fellows are woefully underprepared to realize the therapeutic value of physical activity in patient care, comprehend current physical activity guidelines, appropriately motivate patients to engage in exercise, and competently discuss exercise prescriptions in different disease states. Throughout their training, it is imperative for medical professionals to be educated on the social determinants of health, which include the conditions in which people live, work, and play. These environmental variables can contribute to health inequities that create additional barriers to improvement in physical fitness.6
National guidelines on physical activity
The 2018 National Physical Activity Guidelines detail recommendations for children, adolescents, adults, and special populations.7 The guidelines define physical activity as bodily movement produced by skeletal muscles that result in energy expenditure above resting baseline levels, and includes all types, intensities, and domains of activity. Exercise is a subset of physical activity characterized as planned, structured, repetitive, and designed to improve or maintain physical fitness, physical performance, or health.
Highlights from the 2018 guidelines include7:
- Preschool-aged children (3 to 5 years of age) should be physically active throughout the day, with as much as 3 hours per day of physical activity of all intensities—light, moderate, and vigorous.
- Older children and adolescents (6 to 17 years) should accumulate 60 minutes per day of moderate-to-vigorous physical activity, including aerobic, muscle-strengthening, and bone-strengthening activities.
- Adults of all ages should achieve approximately 150 to 300 minutes of moderate or 75 to 150 minutes of vigorous physical activity (or an equivalent combination) per week, along with at least 2 days per week of muscle-strengthening activities. Other types of physical activity include flexibility, balance, bone-strengthening, and mind–body exercises.
3-step framework for enhancing physical activity counseling
Merely knowing that physical activity is healthy is not enough, during a patient encounter, to increase the level of physical activity. Therefore, it is imperative to learn and adopt a framework that has proved to yield successful outcomes. The Screening, Brief Intervention, and Referral to Treatment (SBIRT) framework, which has predominantly been used to change patient behavior related to alcohol and substance use, is now being utilized by some providers to promote physical activity.8 We apply the SBIRT approach in this article, although research is lacking on its clinical utility and outcome measures.
Continue to: SBIRT
SBIRT: Screening
An office visit provides an opportunity to understand a patient’s level of physical activity. Often, understanding a patient’s baseline level of activity is only asked during a thorough social history, which might not be performed during patient encounters. As physical activity is the primary determinant of cardiorespiratory fitness (CRF), some health care systems have begun delineating physical activity levels as a vital sign to ensure that the assessment of physical activity is a standard part of every clinical encounter. At a minimum, this serves as a prompt and provides an opportunity to start a conversation around improving physical activity levels when guidelines are not being met.
The exercise vital sign. Assessment and documentation of physical activity in the electronic health record are not yet standardized; however, Kaiser Permanente health plans have implemented the exercise vital sign, or EVS, in its HealthConnect (Epic Systems) electronic health record. The EVS incorporates information about a patient’s:
- days per week of moderate-to-strenuous exercise (eg, a brisk walk)
- minutes per day, on average, of exercise at this level.
The physical activity vital sign. Intermountain Healthcare implemented the physical activity vital sign, or PAVS, in its iCentra (Cerner Corp.) electronic health record. The 3-question PAVS assessment asks:
- On average, how many days of the week do you perform physical activity or exercise?
- On average, how many total minutes of physical activity or exercise do you perform on those days?
- How would you describe the intensity of your physical activity or exercise: Light (ie, a casual walk)? Moderate (a brisk walk)? Or vigorous (jogging)?
PAVS includes a fourth data point: The physician–user documents whether the patient was counseled to start, increase, maintain, or modify physical activity or exercise.
EVS and the PAVS have demonstrated validity.9-11
Continue to: Cardiorespiratory fitness as a vital sign
Cardiorespiratory fitness as a vital sign. In 2016, the American Heart Association (AHA) asserted the importance of assessing CRF as a clinical vital sign.12 CRF is commonly expressed as maximal oxygen consumption (VO2max = O2 mL/kg/min) and measured through cardiopulmonary exercise testing (CPET), considered the gold standard by combining conventional graded exercise testing with ventilatory expired gas analysis. CPET is more objective and precise than equations estimating CRF that are derived from peak work rate. AHA recommended that efforts to improve CRF should become standard in clinical encounters, explaining that even a small increase in CRF (eg, 1 or 2 metabolic equivalentsa [METs]) is associated with a considerably (10% to 30%) lower rate of adverse cardiovascular events.12
De Souza de Silva and colleagues revealed an association between each 1-MET increase in CRF and per-person annual health care cost savings (adjusted for age and presence of cardiovascular disease) of $3272 (normal-weight patients), $4252 (overweight), and $6103 (obese).13 In its 2016 scientific statement on CRF as a vital sign, AHA listed several methods of estimating CRF and concluded that, although CPET involves a higher level of training, proficiency, equipment, and, therefore, cost, the independent and additive information obtained justifies its use in many patients.12
CASE
Mary Q, 68 years of age, presents for an annual well-woman examination. Body mass index is 32; resting heart rate (HR), 73 bpm; and blood pressure, 126/74 mm Hg. She reports being inactive, except for light walking every day with her dog around the neighborhood, which takes them approximately 15 minutes. She denies any history or signs and symptoms of cardiovascular, metabolic, or renal disease.
You consider 3 questions before taking next steps regarding increasing Ms. Q’s activity level:
- What is her PAVS?
- Does she need medical clearance before starting an exercise program?
- What would an evidence-based cardiovascular exercise prescription for Ms. Q look like?
SBIRT: Brief intervention
When a patient does not meet the recommended level of physical activity, you have an opportunity to deliver a brief intervention. To do this effectively, you must have adequate understanding of the patient’s receptivity for change. The transtheoretical, or Stages of Change, model proposes that a person typically goes through 5 stages of growth—pre-contemplation, contemplation, preparation, action, and maintenance—in the process of lifestyle modification. This model highlights the different approaches to exercise adoption and maintenance that need to be taken, based on a given patient’s stage at the moment.
Continue to: Using this framework...
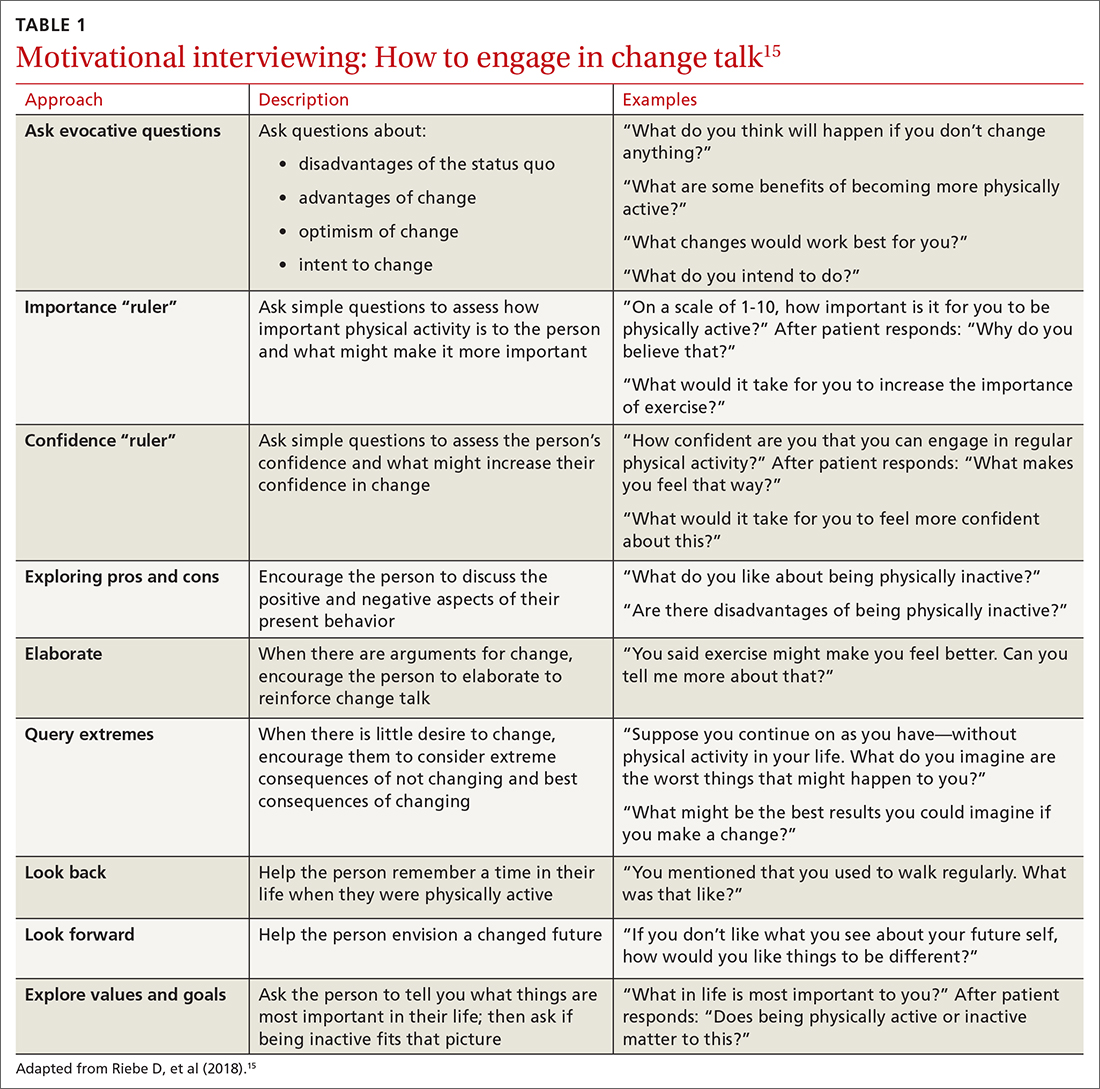
Using this framework, you can help patients realize intrinsic motivation that can facilitate progression through each stage, utilizing techniques such as motivational interviewing—so-called change talk—to increase self-efficacy.14TABLE 115 provides examples of motivational interviewing techniques that can be used during a patient encounter to improve health behaviors, such as physical activity.
Writing the exercise prescription
A patient who wants to increase their level of physical activity should be offered a formal exercise prescription, which has been shown to increase the level of physical activity, particularly in older patients. In fact, a study conducted in Spain in the practices of family physicians found that older patients who received a physical activity prescription increased their activity by 131 minutes per week; and compared to control patients, they doubled the minutes per week devoted to moderate or vigorous physical activity.16
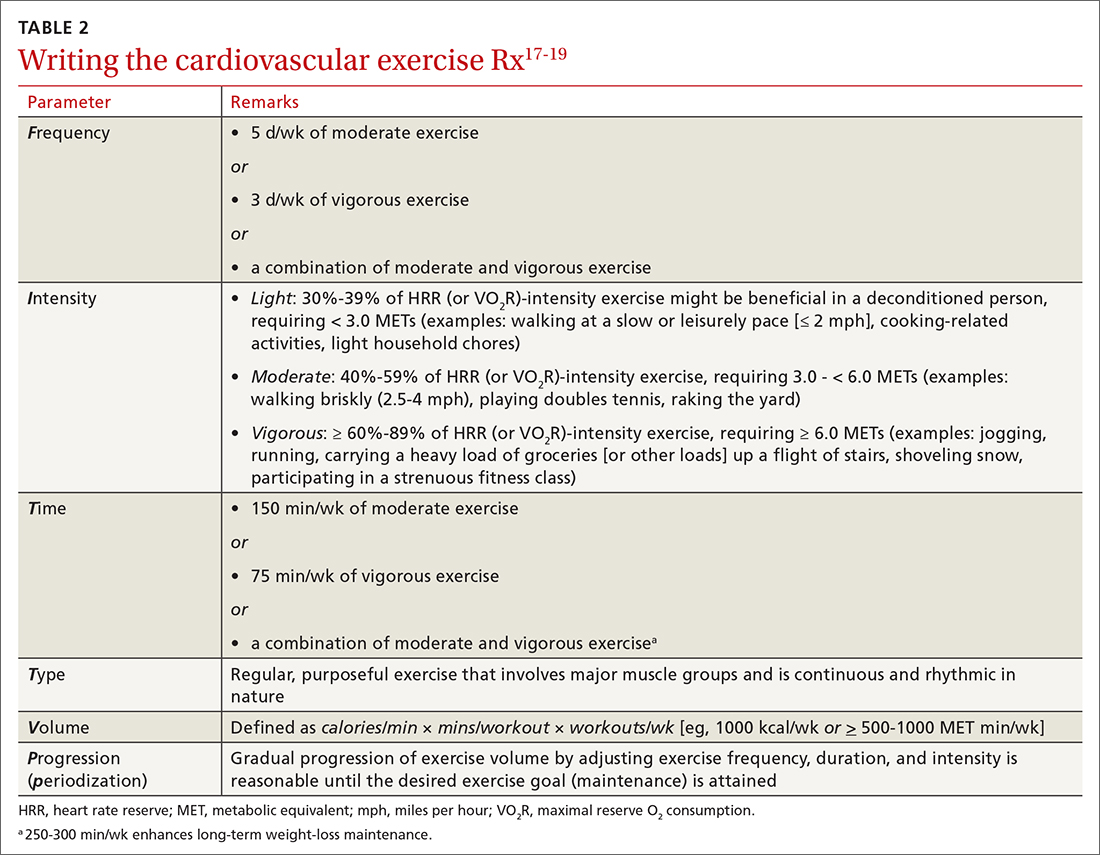
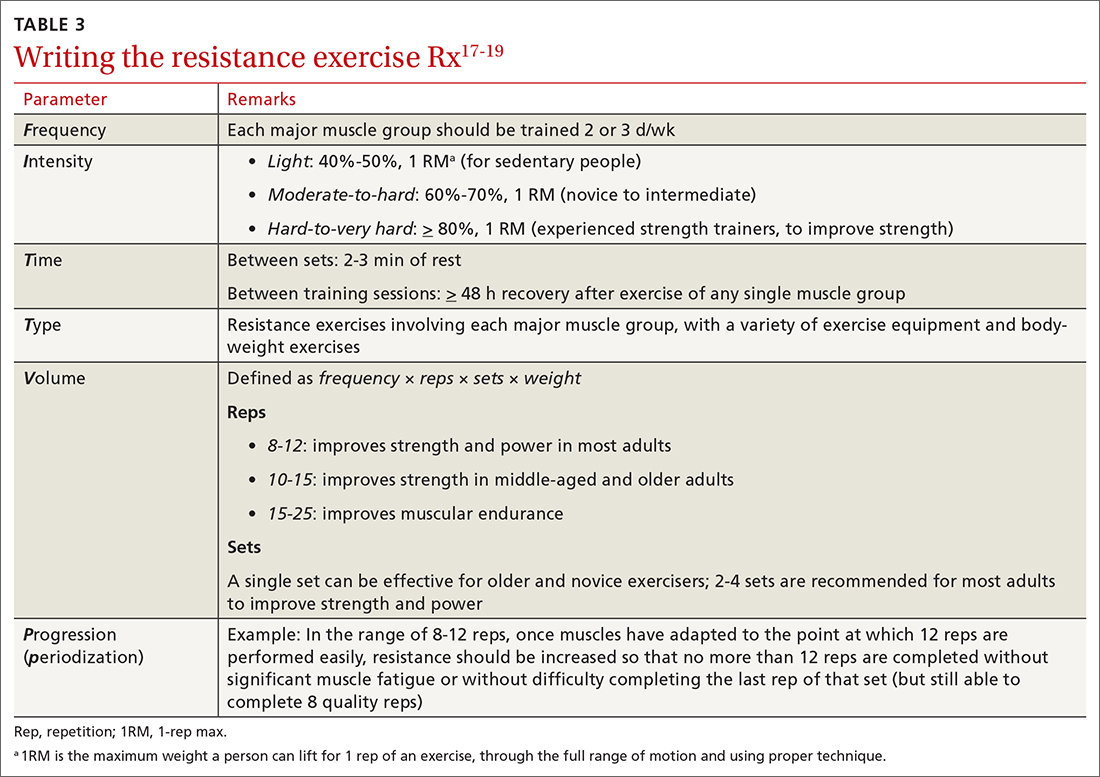
FITT-VP. The basics of a cardiovascular exercise prescription can be found in the FITT-VP (Frequency, Intensity, Time, Type, Volume, and [monitoring of] Progression) framework (TABLE 217-19). For most patients, this model includes 3 to 5 days per week of moderate-to-vigorous physical activity for 30 to 60 minutes per session. For patients with established chronic disease, physical activity provides health benefits but might require modification. Disease-specific patient handouts for exercise can be downloaded, at no cost, through the American College of Sports Medicine (ACSM) “Exercise Is Medicine” program, which can be found at: www.exerciseismedicine.org/support_page.php/rx-for-health-series.
Determining intensity level. Although CPET is the gold standard for determining a patient’s target intensity level, such a test might be impracticable for a given patient. Surrogate markers of target intensity level can be obtained by measuring maximum HR (HRmax), using a well-known equation20:
HRmax = 220 – age
which is then multiplied by intensity range:
- light: 30%-39%
- moderate: 40%-59%
- vigorous: 60%-89%
or, more preferably, by calculating the HR training zone while accounting for HR at rest (HRrest). This is accomplished by calculating the HR reserve (HRR) (ie, HRR = HRmax – HRrest) and then calculating the target heart rate (THR)21:
THR = [HRR × %intensity] + HRrest
Continue to: The THR calculation...
The THR calculation is performed twice, once with a lower %intensity and again with a higher %intensity to develop a training zone based on HRR.
The HRR equation is more accurate than calculating HRmax from 220 – age, because HRR accounts for resting HR, which is often lower in people who are better conditioned.
Another method of calculating intensity for patients who are beginning a physical activity program is the rating of perceived exertion (RPE), which is graded on a scale of 6 to 20: Moderate exercise correlates with an RPE of 12 to 13 (“somewhat hard”); vigorous exercise correlates with an RPE of 14 to 16 (“hard”). By adding a zero to the rating on the RPE scale, the corresponding HR in a healthy adult can be estimated when they are performing an activity at that perceived intensity.22 Moderate exercise therefore correlates with a HR of 120 and 130 bpm.
The so-called talk test can also guide exercise intensity: Light-intensity activity correlates with an ability to sing; moderate-intensity physical activity likely allows the patient to still hold a conversation; and vigorous-intensity activity correlates with an inability to carry on a conversation while exercising.
An exercise prescription should be accompanied by a patient-derived goal, which can be reassessed during a follow-up visit. So-called SMART goals (Specific, Measurable, Achievable, Relevant, and Time-bound) are tools to help patients set personalized and realistic expectations for physical activity. Meeting the goal of approximately 150 to 300 minutes of moderate or 75 to 150 minutes of vigorous physical activity (or an equivalent combination) per week is ideal, but a patient needs to start where they are, at the moment, and gradually increase activity by setting what for them are realistic and sustainable goals.
Continue to: CASE
CASE
With a PAVS of 105 minutes (ie, 15 minutes per day × 7 days) of weekly light-to-moderate exercise walking her dog, Ms. Q does not satisfy current physical activity guidelines. She needs an exercise prescription to incorporate into her lifestyle (see “Cardiovascular exercise prescription,” at left).
First, based on ACSM pre-participation guidelines, Ms. Q does not need medical clearance before initiating light-to-moderate exercise and gradually progressing to vigorous-intensity exercise.
Second, in addition to walking the dog for 105 minutes a week, you:
- advise her to start walking for 10 minutes, 3 times per week, at a pace that keeps her HR at 97-104 bpm.
- encourage her to gradually increase the frequency or duration of her walks by no more than 10% per week.

SBIRT: Referral for treatment
When referring a patient to a fitness program or professional, it is essential to consider their preferences, resources, and environment.23 Community fitness partners are often an excellent referral option for a patient seeking guidance or structure for their exercise program. Using the ACSM ProFinder service, (www.acsm.org/get-stay-certified/find-a-pro) you can search for exercise professionals who have achieved the College’s Gold Standard credential.
Gym memberships or fitness programs might be part of the extra coverage offered by Medicare Advantage Plans, other Medicare health plans, or Medicare Supplement Insurance (Medigap) plans.24
Continue to: CASE
CASE
After providing Ms. Q with her exercise prescription, you refer her to a local gym that participates in the Silver Sneakers fitness and wellness program (for adults ≥ 65 years of age in eligible Medicare plans) to determine whether she qualifies to begin resistance and flexibility training, for which you will write a second exercise prescription (TABLE 317-19).
Pre-participation screening
Updated 2015 ACSM exercise pre-participation health screening recommendations attempt to decrease possible barriers to people who are becoming more physically active, by minimizing unnecessary referral to health care providers before they change their level of physical activity. ACSM recommendations on exercise clearance include this guidance25:
- For a patient who is asymptomatic and already physically active—regardless of whether they have known cardiovascular, metabolic, or renal disease—medical clearance is unnecessary for moderate-intensity exercise.
- Any patient who has been physically active and asymptomatic but who becomes symptomatic during exercise should immediately discontinue such activity and undergo medical evaluation.
- For a patient who is inactive, asymptomatic, and who does not have known cardiovascular, metabolic, or renal disease, medical clearance for light- or moderate-intensity exercise is unnecessary.
- For inactive, asymptomatic patients who have known cardiovascular, metabolic, or renal disease, medical clearance is recommended.
Digital health
Smartwatches and health apps (eg, CardioCoach, Fitbit, Garmin Connect, Nike Training Club, Strava, and Training Peaks) can provide workouts and offer patients the ability to collect information and even connect with other users through social media platforms. This information can be synced to Apple Health platforms for iPhones (www.apple.com/ios/health/) or through Google Fit (www.google.com/fit/) on Android devices. Primary care physicians who become familiar with health apps might find them useful for select patients who want to use technology to improve their physical activity level.
However, data on the value of using digital apps for increasing physical activity, in relation to their cost, are limited. Additional research is needed to assess their validity.
Billing and coding
For most patients, the physical activity assessment, prescription, and referral are performed in the context of treating another condition (eg, hypertension, type 2 diabetes, obesity, depression) or during a preventive health examination, and are typically covered without additional charge to the patient. An evaluation and management visit for an established patient could be used to bill if > 50% of the office visit was spent face-to-face with a physician, with patient counseling and coordination of care.
Continue to: Physicians and physical therapists...
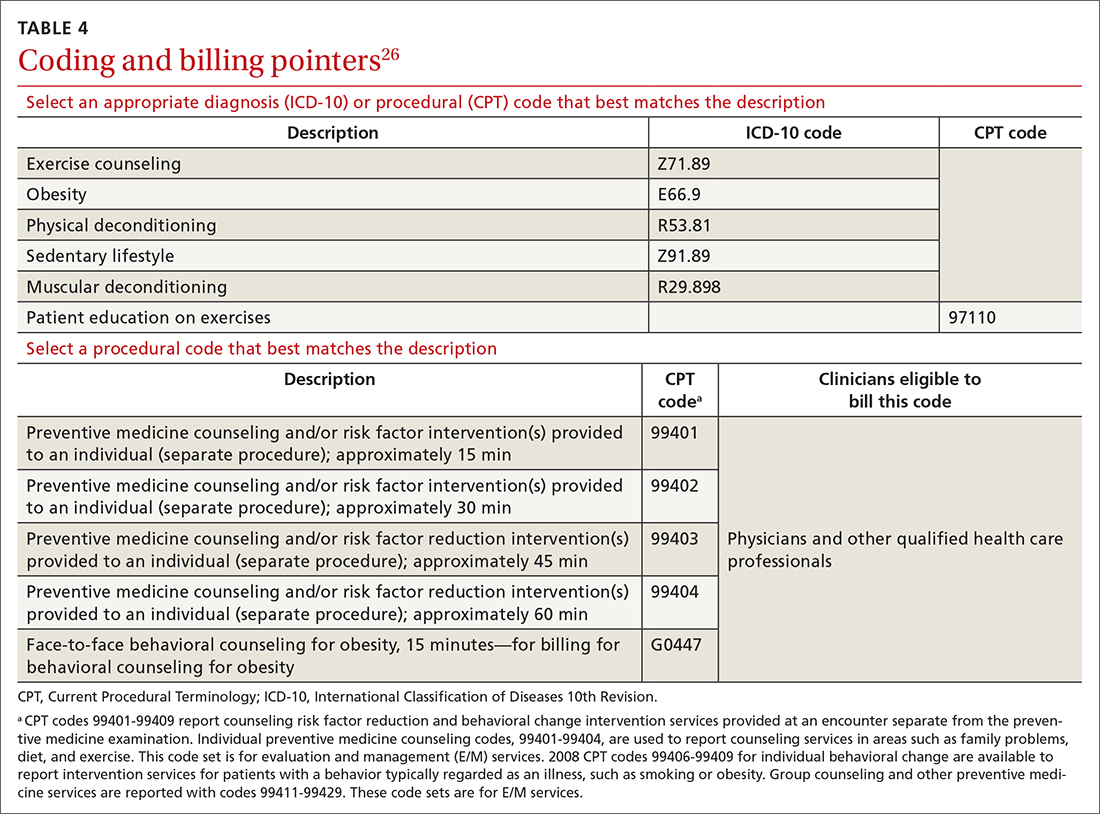
Physicians and physical therapists can use the therapeutic exercise code (Current Procedural Terminology code 97110) when teaching patients exercises to develop muscle strength and endurance, joint range of motion, and flexibility26 (TABLE 426).
Conclusion
Physical activity and CRF are strong predictors of premature mortality, even compared to other risk factors, such as cigarette smoking, hypertension, hypercholesterolemia, and type 2 diabetes.27 Brief physical activity assessment and counseling is an efficient, effective, and cost-effective means to increase physical activity, and presents a unique opportunity for you to encourage lifestyle-based strategies for reducing cardiovascular risk.28
However, it is essential to meet patients where they are before trying to have them progress; it is therefore imperative to assess the individual patient’s level of activity using PAVS. With that information in hand, you can personalize physical activity advice; determine readiness for change and potential barriers for change; assist the patient in setting SMART goals; and arrange follow-up to assess adherence to the exercise prescription. Encourage the patient to call their health insurance plan to determine whether a gym membership or fitness program is covered.
Research is needed to evaluate the value of using digital apps, in light of their cost, to increase physical activity and improve CRF in a clinical setting. Prospective trials should be initiated to determine how routine implementation of CRF assessment in primary care alters the trajectory of clinical care. It is hoped that future research will answer the question: Would such an approach improve clinical outcomes and reduce health care expenditures?12
a Defined as O2 consumed while sitting at rest; equivalent to 3.5 mL of O2 × kg of body weight × min.
CORRESPONDENCE
Matthew Kampert, DO, MS, Sports Medicine, 5555 Transportation Boulevard, Cleveland, OH 44125; kamperm@ccf.org
1. Papanicolas I, Woskie LR, Jha AK. Health care spending in the United States and other high-income countries. JAMA. 2018;319:1024-1039. doi: 10.1001/jama.2018.1150
2. Tikkanen R, Abrams MK. U.S. health care from a global perspective, 2019: higher spending, worse outcomes? The Commonwealth Fund Website. January 30, 2020. Accessed November 16, 2021. www.commonwealthfund.org/publications/issue-briefs/2020/jan/us-health-care-global-perspective-2019
3. Stoutenberg M, Stasi S, Stamatakis E, et al. Physical activity training in US medical schools: preparing future physicians to engage in primary prevention. Phys Sportsmed. 2015;43:388-394. doi: 10.1080/00913847.2015.1084868
4. Antognoli EL, Seeholzer EL, Gullett H, et al. Primary care resident training for obesity, nutrition, and physical activity counseling: a mixed-methods study. Health Promot Pract. 2017;18:672-680. doi: 10.1177/1524839916658025
5. Asif IM, Drezner JA. Sports and exercise medicine education in the USA: call to action. Br J Sports Med. 2020;54:195-196. doi: 10.1136/bjsports-2019-101104
6. Douglas JA, Briones MD, Bauer EZ, et al. Social and environmental determinants of physical activity in urban parks: testing a neighborhood disorder model. Prev Med. 2018;109:119-124. doi: 10.1016/j.ypmed.2018.01.013
7. 2018 Physical Activity Guidelines Advisory Committee. 2018 Physical Activity Guidelines Advisory Committee Scientific Report. Washington, DC: US Department of Health & Human Services; 2018. Accessed November 15, 2021. https://health.gov/sites/default/files/2019-09/PAG_Advisory_Committee_Report.pdf
8. Avis JL, Cave AL, Donaldson S, et al. Working with parents to prevent childhood obesity: protocol for a primary care-based ehealth study. JMIR Res Protoc. 2015;4:e35. doi:10.2196/resprot.4147
9. Ball TJ, Joy EA, Gren LH, et al. Concurrent validity of a self-reported physical activity ‘vital sign’ questionnaire with adult primary care patients. Prev Chronic Dis. 2016;13:e16. doi: 10.5888/pcd13.150228
10. Ball TJ, Joy EA, Gren LH, et al. Predictive validity of an adult physical activity “vital sign” recorded in electronic health records. J Phys Act Health. 2016;13:403-408. doi: 10.1123/jpah.2015-0210
11. Coleman KJ, Ngor E, Reynolds K, et al. Initial validation of an exercise “vital sign” in electronic medical records. Med Sci Sports Exerc. 2012;44:2071-2076. doi: 10.1249/MSS.0b013e3182630ec1
12. Ross R, Blair SN, Arena R, et al; ; ; ; ; ; . Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation. 2016;134:e653-e699. doi: 10.1161/CIR.0000000000000461
13. de Souza de Silva CG, Kokkinos PP, Doom R, et al. Association between cardiorespiratory fitness, obesity, and health care costs: The Veterans Exercise Testing Study. Int J Obes (Lond). 2019;43:2225-2232. doi: 10.1038/s41366-018-0257-0
14. Prochaska JO, Velicer WF. The transtheoretical model of health behavior change. Am J Health Promot. 1997;12:38-48. doi: 10.4278/0890-1171-12.1.38
15. Riebe D, Ehrman JK, Liguori G, et al. Methods for evoking change talk. In: ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Wolters Kluwer; 2018.
16. Grandes G, Sanchez A, Sanchez-Pinilla RO, et al. Effectiveness of physical activity advice and prescription by physicians in routine primary care: a cluster randomized trial. Arch Intern Med. 2009;169:694-701. doi: 10.1001/archinternmed.2009.23
17. McNeill LH, Kreuter MW, Subramanian SV. Social environment and physical activity: a review of concepts and evidence. Soc Sci Med. 2006;63:1011-1022. doi: 10.1016/j.socscimed.2006.03.012
18. Garber CE, Blissmer BE, Deschenes MR, et al; American College of Sports Medicine. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Position stand. Med Sci Sport Exerc. 2011;43:1334-1359. doi: 10.1249/MSS.0b013e318213fefb
19. Donnelly JE, Blair SN, Jakicic JM, et al; American College of Sports Medicine. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Position stand. Med Sci Sport Exerc. 2009;41:459-471. doi: 10.1249/MSS.0b013e3181949333
20. Fox SM 3rd, Naughton JP, Haskell WL. Physical activity and the prevention of coronary heart disease. Ann Clin Res. 1971;3:404-432.
21. Karvonen MJ, Kentala E, Mustala O. The effects of training on heart rate; a longitudinal study. Ann Med Exp Biol Fenn. 1957;35:307-315.
22. The Borg RPE scale. In: Borg G. Borg’s Perceived Exertion and Pain Scales. Human Kinetics; 1998:29-38.
23. Ratamess NA, Alvar BA, Evetoch TK, et al; American College of Sports Medicine. Progression models in resistance training for healthy adults. Position stand. Med Sci Sport Exerc. 2009;41:687-708. doi: 10.1249/MSS.0b013e3181915670
24. Gym memberships & fitness programs. Medicare.gov. Baltimore, MD: US Centers for Medicare and Medicaid Services. Accessed November 16, 2021. www.medicare.gov/coverage/gym-memberships-fitness-programs
25. Riebe D, Franklin BA, Thompson PD, et al. Updating ACSM’s recommendations for exercise preparticipation health screening. Med Sci Sports Exerc. 2015;47:2473-2479. doi: 10.1249/MSS.0000000000000664
26. Physical Activity Related Current Procedural Terminology (CPT®) Codes. Physical Activity Alliance website. Accessed November 16, 2021. https://paamovewithus.org/wp-content/uploads/2020/11/PAA-Physical-Activity-CPT-Codes-Nov-2020-AMA-Approved-Final-1.pdf
27. Blair SN. Physical inactivity: the biggest public health problem of the 21st century Br J Sports Med. 2009;43:1-2.
28. Vuori IM, Lavie CJ, Blair SN. Physical activity promotion in the health care system. Mayo Clin Proc. 2013;88:1446-1461. doi: 10.1016/j.mayocp.2013.08.020
It is well-known that per capita health care spending in the United States is more than twice the average in other developed countries1; nevertheless, the overall health care ranking of the US is near the bottom compared to other countries in this group.2 Much of the reason for this poor relative showing lies in the fact that the US has employed a somewhat traditional fee-for-service health care model that does not incentivize efforts to promote health and wellness or prevent chronic disease. The paradigm of promoting physical activity for its disease-preventing and treatment benefits has not been well-integrated in the US health care system.
In this article, we endeavor to provide better understanding of the barriers that keep family physicians from routinely promoting physical activity in clinical practice; define tools and resources that can be used in the clinical setting to promote physical activity; and delineate areas for future work.
Glaring hole in US physical activity education
Many primary care physicians feel underprepared to prescribe or motivate patients to exercise. The reason for that lack of preparedness likely relates to a medical education system that does not spend time preparing physicians to perform this critical task. A study showed that, on average, medical schools require only 8 hours of physical activity education in their curriculum during the 4 years of schooling.3 Likewise, the average primary care residency program offers only 3 hours of didactic training on physical activity, nutrition, and obesity.4 The problem extends to sports medicine fellowship training, in which a 2019 survey showed that 63% of fellows were never taught how to write an exercise prescription in their training program.5
Without education on physical activity, medical students, residents, and fellows are woefully underprepared to realize the therapeutic value of physical activity in patient care, comprehend current physical activity guidelines, appropriately motivate patients to engage in exercise, and competently discuss exercise prescriptions in different disease states. Throughout their training, it is imperative for medical professionals to be educated on the social determinants of health, which include the conditions in which people live, work, and play. These environmental variables can contribute to health inequities that create additional barriers to improvement in physical fitness.6
National guidelines on physical activity
The 2018 National Physical Activity Guidelines detail recommendations for children, adolescents, adults, and special populations.7 The guidelines define physical activity as bodily movement produced by skeletal muscles that result in energy expenditure above resting baseline levels, and includes all types, intensities, and domains of activity. Exercise is a subset of physical activity characterized as planned, structured, repetitive, and designed to improve or maintain physical fitness, physical performance, or health.
Highlights from the 2018 guidelines include7:
- Preschool-aged children (3 to 5 years of age) should be physically active throughout the day, with as much as 3 hours per day of physical activity of all intensities—light, moderate, and vigorous.
- Older children and adolescents (6 to 17 years) should accumulate 60 minutes per day of moderate-to-vigorous physical activity, including aerobic, muscle-strengthening, and bone-strengthening activities.
- Adults of all ages should achieve approximately 150 to 300 minutes of moderate or 75 to 150 minutes of vigorous physical activity (or an equivalent combination) per week, along with at least 2 days per week of muscle-strengthening activities. Other types of physical activity include flexibility, balance, bone-strengthening, and mind–body exercises.
3-step framework for enhancing physical activity counseling
Merely knowing that physical activity is healthy is not enough, during a patient encounter, to increase the level of physical activity. Therefore, it is imperative to learn and adopt a framework that has proved to yield successful outcomes. The Screening, Brief Intervention, and Referral to Treatment (SBIRT) framework, which has predominantly been used to change patient behavior related to alcohol and substance use, is now being utilized by some providers to promote physical activity.8 We apply the SBIRT approach in this article, although research is lacking on its clinical utility and outcome measures.
Continue to: SBIRT
SBIRT: Screening
An office visit provides an opportunity to understand a patient’s level of physical activity. Often, understanding a patient’s baseline level of activity is only asked during a thorough social history, which might not be performed during patient encounters. As physical activity is the primary determinant of cardiorespiratory fitness (CRF), some health care systems have begun delineating physical activity levels as a vital sign to ensure that the assessment of physical activity is a standard part of every clinical encounter. At a minimum, this serves as a prompt and provides an opportunity to start a conversation around improving physical activity levels when guidelines are not being met.
The exercise vital sign. Assessment and documentation of physical activity in the electronic health record are not yet standardized; however, Kaiser Permanente health plans have implemented the exercise vital sign, or EVS, in its HealthConnect (Epic Systems) electronic health record. The EVS incorporates information about a patient’s:
- days per week of moderate-to-strenuous exercise (eg, a brisk walk)
- minutes per day, on average, of exercise at this level.
The physical activity vital sign. Intermountain Healthcare implemented the physical activity vital sign, or PAVS, in its iCentra (Cerner Corp.) electronic health record. The 3-question PAVS assessment asks:
- On average, how many days of the week do you perform physical activity or exercise?
- On average, how many total minutes of physical activity or exercise do you perform on those days?
- How would you describe the intensity of your physical activity or exercise: Light (ie, a casual walk)? Moderate (a brisk walk)? Or vigorous (jogging)?
PAVS includes a fourth data point: The physician–user documents whether the patient was counseled to start, increase, maintain, or modify physical activity or exercise.
EVS and the PAVS have demonstrated validity.9-11
Continue to: Cardiorespiratory fitness as a vital sign
Cardiorespiratory fitness as a vital sign. In 2016, the American Heart Association (AHA) asserted the importance of assessing CRF as a clinical vital sign.12 CRF is commonly expressed as maximal oxygen consumption (VO2max = O2 mL/kg/min) and measured through cardiopulmonary exercise testing (CPET), considered the gold standard by combining conventional graded exercise testing with ventilatory expired gas analysis. CPET is more objective and precise than equations estimating CRF that are derived from peak work rate. AHA recommended that efforts to improve CRF should become standard in clinical encounters, explaining that even a small increase in CRF (eg, 1 or 2 metabolic equivalentsa [METs]) is associated with a considerably (10% to 30%) lower rate of adverse cardiovascular events.12
De Souza de Silva and colleagues revealed an association between each 1-MET increase in CRF and per-person annual health care cost savings (adjusted for age and presence of cardiovascular disease) of $3272 (normal-weight patients), $4252 (overweight), and $6103 (obese).13 In its 2016 scientific statement on CRF as a vital sign, AHA listed several methods of estimating CRF and concluded that, although CPET involves a higher level of training, proficiency, equipment, and, therefore, cost, the independent and additive information obtained justifies its use in many patients.12
CASE
Mary Q, 68 years of age, presents for an annual well-woman examination. Body mass index is 32; resting heart rate (HR), 73 bpm; and blood pressure, 126/74 mm Hg. She reports being inactive, except for light walking every day with her dog around the neighborhood, which takes them approximately 15 minutes. She denies any history or signs and symptoms of cardiovascular, metabolic, or renal disease.
You consider 3 questions before taking next steps regarding increasing Ms. Q’s activity level:
- What is her PAVS?
- Does she need medical clearance before starting an exercise program?
- What would an evidence-based cardiovascular exercise prescription for Ms. Q look like?
SBIRT: Brief intervention
When a patient does not meet the recommended level of physical activity, you have an opportunity to deliver a brief intervention. To do this effectively, you must have adequate understanding of the patient’s receptivity for change. The transtheoretical, or Stages of Change, model proposes that a person typically goes through 5 stages of growth—pre-contemplation, contemplation, preparation, action, and maintenance—in the process of lifestyle modification. This model highlights the different approaches to exercise adoption and maintenance that need to be taken, based on a given patient’s stage at the moment.
Continue to: Using this framework...
Using this framework, you can help patients realize intrinsic motivation that can facilitate progression through each stage, utilizing techniques such as motivational interviewing—so-called change talk—to increase self-efficacy.14TABLE 115 provides examples of motivational interviewing techniques that can be used during a patient encounter to improve health behaviors, such as physical activity.

Writing the exercise prescription
A patient who wants to increase their level of physical activity should be offered a formal exercise prescription, which has been shown to increase the level of physical activity, particularly in older patients. In fact, a study conducted in Spain in the practices of family physicians found that older patients who received a physical activity prescription increased their activity by 131 minutes per week; and compared to control patients, they doubled the minutes per week devoted to moderate or vigorous physical activity.16
FITT-VP. The basics of a cardiovascular exercise prescription can be found in the FITT-VP (Frequency, Intensity, Time, Type, Volume, and [monitoring of] Progression) framework (TABLE 217-19). For most patients, this model includes 3 to 5 days per week of moderate-to-vigorous physical activity for 30 to 60 minutes per session. For patients with established chronic disease, physical activity provides health benefits but might require modification. Disease-specific patient handouts for exercise can be downloaded, at no cost, through the American College of Sports Medicine (ACSM) “Exercise Is Medicine” program, which can be found at: www.exerciseismedicine.org/support_page.php/rx-for-health-series.

Determining intensity level. Although CPET is the gold standard for determining a patient’s target intensity level, such a test might be impracticable for a given patient. Surrogate markers of target intensity level can be obtained by measuring maximum HR (HRmax), using a well-known equation20:
HRmax = 220 – age
which is then multiplied by intensity range:
- light: 30%-39%
- moderate: 40%-59%
- vigorous: 60%-89%
or, more preferably, by calculating the HR training zone while accounting for HR at rest (HRrest). This is accomplished by calculating the HR reserve (HRR) (ie, HRR = HRmax – HRrest) and then calculating the target heart rate (THR)21:
THR = [HRR × %intensity] + HRrest
Continue to: The THR calculation...
The THR calculation is performed twice, once with a lower %intensity and again with a higher %intensity to develop a training zone based on HRR.
The HRR equation is more accurate than calculating HRmax from 220 – age, because HRR accounts for resting HR, which is often lower in people who are better conditioned.
Another method of calculating intensity for patients who are beginning a physical activity program is the rating of perceived exertion (RPE), which is graded on a scale of 6 to 20: Moderate exercise correlates with an RPE of 12 to 13 (“somewhat hard”); vigorous exercise correlates with an RPE of 14 to 16 (“hard”). By adding a zero to the rating on the RPE scale, the corresponding HR in a healthy adult can be estimated when they are performing an activity at that perceived intensity.22 Moderate exercise therefore correlates with a HR of 120 and 130 bpm.
The so-called talk test can also guide exercise intensity: Light-intensity activity correlates with an ability to sing; moderate-intensity physical activity likely allows the patient to still hold a conversation; and vigorous-intensity activity correlates with an inability to carry on a conversation while exercising.
An exercise prescription should be accompanied by a patient-derived goal, which can be reassessed during a follow-up visit. So-called SMART goals (Specific, Measurable, Achievable, Relevant, and Time-bound) are tools to help patients set personalized and realistic expectations for physical activity. Meeting the goal of approximately 150 to 300 minutes of moderate or 75 to 150 minutes of vigorous physical activity (or an equivalent combination) per week is ideal, but a patient needs to start where they are, at the moment, and gradually increase activity by setting what for them are realistic and sustainable goals.
Continue to: CASE
CASE
With a PAVS of 105 minutes (ie, 15 minutes per day × 7 days) of weekly light-to-moderate exercise walking her dog, Ms. Q does not satisfy current physical activity guidelines. She needs an exercise prescription to incorporate into her lifestyle (see “Cardiovascular exercise prescription,” at left).
First, based on ACSM pre-participation guidelines, Ms. Q does not need medical clearance before initiating light-to-moderate exercise and gradually progressing to vigorous-intensity exercise.
Second, in addition to walking the dog for 105 minutes a week, you:
- advise her to start walking for 10 minutes, 3 times per week, at a pace that keeps her HR at 97-104 bpm.
- encourage her to gradually increase the frequency or duration of her walks by no more than 10% per week.

SBIRT: Referral for treatment
When referring a patient to a fitness program or professional, it is essential to consider their preferences, resources, and environment.23 Community fitness partners are often an excellent referral option for a patient seeking guidance or structure for their exercise program. Using the ACSM ProFinder service, (www.acsm.org/get-stay-certified/find-a-pro) you can search for exercise professionals who have achieved the College’s Gold Standard credential.
Gym memberships or fitness programs might be part of the extra coverage offered by Medicare Advantage Plans, other Medicare health plans, or Medicare Supplement Insurance (Medigap) plans.24
Continue to: CASE
CASE
After providing Ms. Q with her exercise prescription, you refer her to a local gym that participates in the Silver Sneakers fitness and wellness program (for adults ≥ 65 years of age in eligible Medicare plans) to determine whether she qualifies to begin resistance and flexibility training, for which you will write a second exercise prescription (TABLE 317-19).

Pre-participation screening
Updated 2015 ACSM exercise pre-participation health screening recommendations attempt to decrease possible barriers to people who are becoming more physically active, by minimizing unnecessary referral to health care providers before they change their level of physical activity. ACSM recommendations on exercise clearance include this guidance25:
- For a patient who is asymptomatic and already physically active—regardless of whether they have known cardiovascular, metabolic, or renal disease—medical clearance is unnecessary for moderate-intensity exercise.
- Any patient who has been physically active and asymptomatic but who becomes symptomatic during exercise should immediately discontinue such activity and undergo medical evaluation.
- For a patient who is inactive, asymptomatic, and who does not have known cardiovascular, metabolic, or renal disease, medical clearance for light- or moderate-intensity exercise is unnecessary.
- For inactive, asymptomatic patients who have known cardiovascular, metabolic, or renal disease, medical clearance is recommended.
Digital health
Smartwatches and health apps (eg, CardioCoach, Fitbit, Garmin Connect, Nike Training Club, Strava, and Training Peaks) can provide workouts and offer patients the ability to collect information and even connect with other users through social media platforms. This information can be synced to Apple Health platforms for iPhones (www.apple.com/ios/health/) or through Google Fit (www.google.com/fit/) on Android devices. Primary care physicians who become familiar with health apps might find them useful for select patients who want to use technology to improve their physical activity level.
However, data on the value of using digital apps for increasing physical activity, in relation to their cost, are limited. Additional research is needed to assess their validity.
Billing and coding
For most patients, the physical activity assessment, prescription, and referral are performed in the context of treating another condition (eg, hypertension, type 2 diabetes, obesity, depression) or during a preventive health examination, and are typically covered without additional charge to the patient. An evaluation and management visit for an established patient could be used to bill if > 50% of the office visit was spent face-to-face with a physician, with patient counseling and coordination of care.
Continue to: Physicians and physical therapists...
Physicians and physical therapists can use the therapeutic exercise code (Current Procedural Terminology code 97110) when teaching patients exercises to develop muscle strength and endurance, joint range of motion, and flexibility26 (TABLE 426).

Conclusion
Physical activity and CRF are strong predictors of premature mortality, even compared to other risk factors, such as cigarette smoking, hypertension, hypercholesterolemia, and type 2 diabetes.27 Brief physical activity assessment and counseling is an efficient, effective, and cost-effective means to increase physical activity, and presents a unique opportunity for you to encourage lifestyle-based strategies for reducing cardiovascular risk.28
However, it is essential to meet patients where they are before trying to have them progress; it is therefore imperative to assess the individual patient’s level of activity using PAVS. With that information in hand, you can personalize physical activity advice; determine readiness for change and potential barriers for change; assist the patient in setting SMART goals; and arrange follow-up to assess adherence to the exercise prescription. Encourage the patient to call their health insurance plan to determine whether a gym membership or fitness program is covered.
Research is needed to evaluate the value of using digital apps, in light of their cost, to increase physical activity and improve CRF in a clinical setting. Prospective trials should be initiated to determine how routine implementation of CRF assessment in primary care alters the trajectory of clinical care. It is hoped that future research will answer the question: Would such an approach improve clinical outcomes and reduce health care expenditures?12
a Defined as O2 consumed while sitting at rest; equivalent to 3.5 mL of O2 × kg of body weight × min.
CORRESPONDENCE
Matthew Kampert, DO, MS, Sports Medicine, 5555 Transportation Boulevard, Cleveland, OH 44125; kamperm@ccf.org
It is well-known that per capita health care spending in the United States is more than twice the average in other developed countries1; nevertheless, the overall health care ranking of the US is near the bottom compared to other countries in this group.2 Much of the reason for this poor relative showing lies in the fact that the US has employed a somewhat traditional fee-for-service health care model that does not incentivize efforts to promote health and wellness or prevent chronic disease. The paradigm of promoting physical activity for its disease-preventing and treatment benefits has not been well-integrated in the US health care system.
In this article, we endeavor to provide better understanding of the barriers that keep family physicians from routinely promoting physical activity in clinical practice; define tools and resources that can be used in the clinical setting to promote physical activity; and delineate areas for future work.
Glaring hole in US physical activity education
Many primary care physicians feel underprepared to prescribe or motivate patients to exercise. The reason for that lack of preparedness likely relates to a medical education system that does not spend time preparing physicians to perform this critical task. A study showed that, on average, medical schools require only 8 hours of physical activity education in their curriculum during the 4 years of schooling.3 Likewise, the average primary care residency program offers only 3 hours of didactic training on physical activity, nutrition, and obesity.4 The problem extends to sports medicine fellowship training, in which a 2019 survey showed that 63% of fellows were never taught how to write an exercise prescription in their training program.5
Without education on physical activity, medical students, residents, and fellows are woefully underprepared to realize the therapeutic value of physical activity in patient care, comprehend current physical activity guidelines, appropriately motivate patients to engage in exercise, and competently discuss exercise prescriptions in different disease states. Throughout their training, it is imperative for medical professionals to be educated on the social determinants of health, which include the conditions in which people live, work, and play. These environmental variables can contribute to health inequities that create additional barriers to improvement in physical fitness.6
National guidelines on physical activity
The 2018 National Physical Activity Guidelines detail recommendations for children, adolescents, adults, and special populations.7 The guidelines define physical activity as bodily movement produced by skeletal muscles that result in energy expenditure above resting baseline levels, and includes all types, intensities, and domains of activity. Exercise is a subset of physical activity characterized as planned, structured, repetitive, and designed to improve or maintain physical fitness, physical performance, or health.
Highlights from the 2018 guidelines include7:
- Preschool-aged children (3 to 5 years of age) should be physically active throughout the day, with as much as 3 hours per day of physical activity of all intensities—light, moderate, and vigorous.
- Older children and adolescents (6 to 17 years) should accumulate 60 minutes per day of moderate-to-vigorous physical activity, including aerobic, muscle-strengthening, and bone-strengthening activities.
- Adults of all ages should achieve approximately 150 to 300 minutes of moderate or 75 to 150 minutes of vigorous physical activity (or an equivalent combination) per week, along with at least 2 days per week of muscle-strengthening activities. Other types of physical activity include flexibility, balance, bone-strengthening, and mind–body exercises.
3-step framework for enhancing physical activity counseling
Merely knowing that physical activity is healthy is not enough, during a patient encounter, to increase the level of physical activity. Therefore, it is imperative to learn and adopt a framework that has proved to yield successful outcomes. The Screening, Brief Intervention, and Referral to Treatment (SBIRT) framework, which has predominantly been used to change patient behavior related to alcohol and substance use, is now being utilized by some providers to promote physical activity.8 We apply the SBIRT approach in this article, although research is lacking on its clinical utility and outcome measures.
Continue to: SBIRT
SBIRT: Screening
An office visit provides an opportunity to understand a patient’s level of physical activity. Often, understanding a patient’s baseline level of activity is only asked during a thorough social history, which might not be performed during patient encounters. As physical activity is the primary determinant of cardiorespiratory fitness (CRF), some health care systems have begun delineating physical activity levels as a vital sign to ensure that the assessment of physical activity is a standard part of every clinical encounter. At a minimum, this serves as a prompt and provides an opportunity to start a conversation around improving physical activity levels when guidelines are not being met.
The exercise vital sign. Assessment and documentation of physical activity in the electronic health record are not yet standardized; however, Kaiser Permanente health plans have implemented the exercise vital sign, or EVS, in its HealthConnect (Epic Systems) electronic health record. The EVS incorporates information about a patient’s:
- days per week of moderate-to-strenuous exercise (eg, a brisk walk)
- minutes per day, on average, of exercise at this level.
The physical activity vital sign. Intermountain Healthcare implemented the physical activity vital sign, or PAVS, in its iCentra (Cerner Corp.) electronic health record. The 3-question PAVS assessment asks:
- On average, how many days of the week do you perform physical activity or exercise?
- On average, how many total minutes of physical activity or exercise do you perform on those days?
- How would you describe the intensity of your physical activity or exercise: Light (ie, a casual walk)? Moderate (a brisk walk)? Or vigorous (jogging)?
PAVS includes a fourth data point: The physician–user documents whether the patient was counseled to start, increase, maintain, or modify physical activity or exercise.
EVS and the PAVS have demonstrated validity.9-11
Continue to: Cardiorespiratory fitness as a vital sign
Cardiorespiratory fitness as a vital sign. In 2016, the American Heart Association (AHA) asserted the importance of assessing CRF as a clinical vital sign.12 CRF is commonly expressed as maximal oxygen consumption (VO2max = O2 mL/kg/min) and measured through cardiopulmonary exercise testing (CPET), considered the gold standard by combining conventional graded exercise testing with ventilatory expired gas analysis. CPET is more objective and precise than equations estimating CRF that are derived from peak work rate. AHA recommended that efforts to improve CRF should become standard in clinical encounters, explaining that even a small increase in CRF (eg, 1 or 2 metabolic equivalentsa [METs]) is associated with a considerably (10% to 30%) lower rate of adverse cardiovascular events.12
De Souza de Silva and colleagues revealed an association between each 1-MET increase in CRF and per-person annual health care cost savings (adjusted for age and presence of cardiovascular disease) of $3272 (normal-weight patients), $4252 (overweight), and $6103 (obese).13 In its 2016 scientific statement on CRF as a vital sign, AHA listed several methods of estimating CRF and concluded that, although CPET involves a higher level of training, proficiency, equipment, and, therefore, cost, the independent and additive information obtained justifies its use in many patients.12
CASE
Mary Q, 68 years of age, presents for an annual well-woman examination. Body mass index is 32; resting heart rate (HR), 73 bpm; and blood pressure, 126/74 mm Hg. She reports being inactive, except for light walking every day with her dog around the neighborhood, which takes them approximately 15 minutes. She denies any history or signs and symptoms of cardiovascular, metabolic, or renal disease.
You consider 3 questions before taking next steps regarding increasing Ms. Q’s activity level:
- What is her PAVS?
- Does she need medical clearance before starting an exercise program?
- What would an evidence-based cardiovascular exercise prescription for Ms. Q look like?
SBIRT: Brief intervention
When a patient does not meet the recommended level of physical activity, you have an opportunity to deliver a brief intervention. To do this effectively, you must have adequate understanding of the patient’s receptivity for change. The transtheoretical, or Stages of Change, model proposes that a person typically goes through 5 stages of growth—pre-contemplation, contemplation, preparation, action, and maintenance—in the process of lifestyle modification. This model highlights the different approaches to exercise adoption and maintenance that need to be taken, based on a given patient’s stage at the moment.
Continue to: Using this framework...
Using this framework, you can help patients realize intrinsic motivation that can facilitate progression through each stage, utilizing techniques such as motivational interviewing—so-called change talk—to increase self-efficacy.14TABLE 115 provides examples of motivational interviewing techniques that can be used during a patient encounter to improve health behaviors, such as physical activity.

Writing the exercise prescription
A patient who wants to increase their level of physical activity should be offered a formal exercise prescription, which has been shown to increase the level of physical activity, particularly in older patients. In fact, a study conducted in Spain in the practices of family physicians found that older patients who received a physical activity prescription increased their activity by 131 minutes per week; and compared to control patients, they doubled the minutes per week devoted to moderate or vigorous physical activity.16
FITT-VP. The basics of a cardiovascular exercise prescription can be found in the FITT-VP (Frequency, Intensity, Time, Type, Volume, and [monitoring of] Progression) framework (TABLE 217-19). For most patients, this model includes 3 to 5 days per week of moderate-to-vigorous physical activity for 30 to 60 minutes per session. For patients with established chronic disease, physical activity provides health benefits but might require modification. Disease-specific patient handouts for exercise can be downloaded, at no cost, through the American College of Sports Medicine (ACSM) “Exercise Is Medicine” program, which can be found at: www.exerciseismedicine.org/support_page.php/rx-for-health-series.

Determining intensity level. Although CPET is the gold standard for determining a patient’s target intensity level, such a test might be impracticable for a given patient. Surrogate markers of target intensity level can be obtained by measuring maximum HR (HRmax), using a well-known equation20:
HRmax = 220 – age
which is then multiplied by intensity range:
- light: 30%-39%
- moderate: 40%-59%
- vigorous: 60%-89%
or, more preferably, by calculating the HR training zone while accounting for HR at rest (HRrest). This is accomplished by calculating the HR reserve (HRR) (ie, HRR = HRmax – HRrest) and then calculating the target heart rate (THR)21:
THR = [HRR × %intensity] + HRrest
Continue to: The THR calculation...
The THR calculation is performed twice, once with a lower %intensity and again with a higher %intensity to develop a training zone based on HRR.
The HRR equation is more accurate than calculating HRmax from 220 – age, because HRR accounts for resting HR, which is often lower in people who are better conditioned.
Another method of calculating intensity for patients who are beginning a physical activity program is the rating of perceived exertion (RPE), which is graded on a scale of 6 to 20: Moderate exercise correlates with an RPE of 12 to 13 (“somewhat hard”); vigorous exercise correlates with an RPE of 14 to 16 (“hard”). By adding a zero to the rating on the RPE scale, the corresponding HR in a healthy adult can be estimated when they are performing an activity at that perceived intensity.22 Moderate exercise therefore correlates with a HR of 120 and 130 bpm.
The so-called talk test can also guide exercise intensity: Light-intensity activity correlates with an ability to sing; moderate-intensity physical activity likely allows the patient to still hold a conversation; and vigorous-intensity activity correlates with an inability to carry on a conversation while exercising.
An exercise prescription should be accompanied by a patient-derived goal, which can be reassessed during a follow-up visit. So-called SMART goals (Specific, Measurable, Achievable, Relevant, and Time-bound) are tools to help patients set personalized and realistic expectations for physical activity. Meeting the goal of approximately 150 to 300 minutes of moderate or 75 to 150 minutes of vigorous physical activity (or an equivalent combination) per week is ideal, but a patient needs to start where they are, at the moment, and gradually increase activity by setting what for them are realistic and sustainable goals.
Continue to: CASE
CASE
With a PAVS of 105 minutes (ie, 15 minutes per day × 7 days) of weekly light-to-moderate exercise walking her dog, Ms. Q does not satisfy current physical activity guidelines. She needs an exercise prescription to incorporate into her lifestyle (see “Cardiovascular exercise prescription,” at left).
First, based on ACSM pre-participation guidelines, Ms. Q does not need medical clearance before initiating light-to-moderate exercise and gradually progressing to vigorous-intensity exercise.
Second, in addition to walking the dog for 105 minutes a week, you:
- advise her to start walking for 10 minutes, 3 times per week, at a pace that keeps her HR at 97-104 bpm.
- encourage her to gradually increase the frequency or duration of her walks by no more than 10% per week.

SBIRT: Referral for treatment
When referring a patient to a fitness program or professional, it is essential to consider their preferences, resources, and environment.23 Community fitness partners are often an excellent referral option for a patient seeking guidance or structure for their exercise program. Using the ACSM ProFinder service, (www.acsm.org/get-stay-certified/find-a-pro) you can search for exercise professionals who have achieved the College’s Gold Standard credential.
Gym memberships or fitness programs might be part of the extra coverage offered by Medicare Advantage Plans, other Medicare health plans, or Medicare Supplement Insurance (Medigap) plans.24
Continue to: CASE
CASE
After providing Ms. Q with her exercise prescription, you refer her to a local gym that participates in the Silver Sneakers fitness and wellness program (for adults ≥ 65 years of age in eligible Medicare plans) to determine whether she qualifies to begin resistance and flexibility training, for which you will write a second exercise prescription (TABLE 317-19).

Pre-participation screening
Updated 2015 ACSM exercise pre-participation health screening recommendations attempt to decrease possible barriers to people who are becoming more physically active, by minimizing unnecessary referral to health care providers before they change their level of physical activity. ACSM recommendations on exercise clearance include this guidance25:
- For a patient who is asymptomatic and already physically active—regardless of whether they have known cardiovascular, metabolic, or renal disease—medical clearance is unnecessary for moderate-intensity exercise.
- Any patient who has been physically active and asymptomatic but who becomes symptomatic during exercise should immediately discontinue such activity and undergo medical evaluation.
- For a patient who is inactive, asymptomatic, and who does not have known cardiovascular, metabolic, or renal disease, medical clearance for light- or moderate-intensity exercise is unnecessary.
- For inactive, asymptomatic patients who have known cardiovascular, metabolic, or renal disease, medical clearance is recommended.
Digital health
Smartwatches and health apps (eg, CardioCoach, Fitbit, Garmin Connect, Nike Training Club, Strava, and Training Peaks) can provide workouts and offer patients the ability to collect information and even connect with other users through social media platforms. This information can be synced to Apple Health platforms for iPhones (www.apple.com/ios/health/) or through Google Fit (www.google.com/fit/) on Android devices. Primary care physicians who become familiar with health apps might find them useful for select patients who want to use technology to improve their physical activity level.
However, data on the value of using digital apps for increasing physical activity, in relation to their cost, are limited. Additional research is needed to assess their validity.
Billing and coding
For most patients, the physical activity assessment, prescription, and referral are performed in the context of treating another condition (eg, hypertension, type 2 diabetes, obesity, depression) or during a preventive health examination, and are typically covered without additional charge to the patient. An evaluation and management visit for an established patient could be used to bill if > 50% of the office visit was spent face-to-face with a physician, with patient counseling and coordination of care.
Continue to: Physicians and physical therapists...
Physicians and physical therapists can use the therapeutic exercise code (Current Procedural Terminology code 97110) when teaching patients exercises to develop muscle strength and endurance, joint range of motion, and flexibility26 (TABLE 426).

Conclusion
Physical activity and CRF are strong predictors of premature mortality, even compared to other risk factors, such as cigarette smoking, hypertension, hypercholesterolemia, and type 2 diabetes.27 Brief physical activity assessment and counseling is an efficient, effective, and cost-effective means to increase physical activity, and presents a unique opportunity for you to encourage lifestyle-based strategies for reducing cardiovascular risk.28
However, it is essential to meet patients where they are before trying to have them progress; it is therefore imperative to assess the individual patient’s level of activity using PAVS. With that information in hand, you can personalize physical activity advice; determine readiness for change and potential barriers for change; assist the patient in setting SMART goals; and arrange follow-up to assess adherence to the exercise prescription. Encourage the patient to call their health insurance plan to determine whether a gym membership or fitness program is covered.
Research is needed to evaluate the value of using digital apps, in light of their cost, to increase physical activity and improve CRF in a clinical setting. Prospective trials should be initiated to determine how routine implementation of CRF assessment in primary care alters the trajectory of clinical care. It is hoped that future research will answer the question: Would such an approach improve clinical outcomes and reduce health care expenditures?12
a Defined as O2 consumed while sitting at rest; equivalent to 3.5 mL of O2 × kg of body weight × min.
CORRESPONDENCE
Matthew Kampert, DO, MS, Sports Medicine, 5555 Transportation Boulevard, Cleveland, OH 44125; kamperm@ccf.org
1. Papanicolas I, Woskie LR, Jha AK. Health care spending in the United States and other high-income countries. JAMA. 2018;319:1024-1039. doi: 10.1001/jama.2018.1150
2. Tikkanen R, Abrams MK. U.S. health care from a global perspective, 2019: higher spending, worse outcomes? The Commonwealth Fund Website. January 30, 2020. Accessed November 16, 2021. www.commonwealthfund.org/publications/issue-briefs/2020/jan/us-health-care-global-perspective-2019
3. Stoutenberg M, Stasi S, Stamatakis E, et al. Physical activity training in US medical schools: preparing future physicians to engage in primary prevention. Phys Sportsmed. 2015;43:388-394. doi: 10.1080/00913847.2015.1084868
4. Antognoli EL, Seeholzer EL, Gullett H, et al. Primary care resident training for obesity, nutrition, and physical activity counseling: a mixed-methods study. Health Promot Pract. 2017;18:672-680. doi: 10.1177/1524839916658025
5. Asif IM, Drezner JA. Sports and exercise medicine education in the USA: call to action. Br J Sports Med. 2020;54:195-196. doi: 10.1136/bjsports-2019-101104
6. Douglas JA, Briones MD, Bauer EZ, et al. Social and environmental determinants of physical activity in urban parks: testing a neighborhood disorder model. Prev Med. 2018;109:119-124. doi: 10.1016/j.ypmed.2018.01.013
7. 2018 Physical Activity Guidelines Advisory Committee. 2018 Physical Activity Guidelines Advisory Committee Scientific Report. Washington, DC: US Department of Health & Human Services; 2018. Accessed November 15, 2021. https://health.gov/sites/default/files/2019-09/PAG_Advisory_Committee_Report.pdf
8. Avis JL, Cave AL, Donaldson S, et al. Working with parents to prevent childhood obesity: protocol for a primary care-based ehealth study. JMIR Res Protoc. 2015;4:e35. doi:10.2196/resprot.4147
9. Ball TJ, Joy EA, Gren LH, et al. Concurrent validity of a self-reported physical activity ‘vital sign’ questionnaire with adult primary care patients. Prev Chronic Dis. 2016;13:e16. doi: 10.5888/pcd13.150228
10. Ball TJ, Joy EA, Gren LH, et al. Predictive validity of an adult physical activity “vital sign” recorded in electronic health records. J Phys Act Health. 2016;13:403-408. doi: 10.1123/jpah.2015-0210
11. Coleman KJ, Ngor E, Reynolds K, et al. Initial validation of an exercise “vital sign” in electronic medical records. Med Sci Sports Exerc. 2012;44:2071-2076. doi: 10.1249/MSS.0b013e3182630ec1
12. Ross R, Blair SN, Arena R, et al; ; ; ; ; ; . Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation. 2016;134:e653-e699. doi: 10.1161/CIR.0000000000000461
13. de Souza de Silva CG, Kokkinos PP, Doom R, et al. Association between cardiorespiratory fitness, obesity, and health care costs: The Veterans Exercise Testing Study. Int J Obes (Lond). 2019;43:2225-2232. doi: 10.1038/s41366-018-0257-0
14. Prochaska JO, Velicer WF. The transtheoretical model of health behavior change. Am J Health Promot. 1997;12:38-48. doi: 10.4278/0890-1171-12.1.38
15. Riebe D, Ehrman JK, Liguori G, et al. Methods for evoking change talk. In: ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Wolters Kluwer; 2018.
16. Grandes G, Sanchez A, Sanchez-Pinilla RO, et al. Effectiveness of physical activity advice and prescription by physicians in routine primary care: a cluster randomized trial. Arch Intern Med. 2009;169:694-701. doi: 10.1001/archinternmed.2009.23
17. McNeill LH, Kreuter MW, Subramanian SV. Social environment and physical activity: a review of concepts and evidence. Soc Sci Med. 2006;63:1011-1022. doi: 10.1016/j.socscimed.2006.03.012
18. Garber CE, Blissmer BE, Deschenes MR, et al; American College of Sports Medicine. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Position stand. Med Sci Sport Exerc. 2011;43:1334-1359. doi: 10.1249/MSS.0b013e318213fefb
19. Donnelly JE, Blair SN, Jakicic JM, et al; American College of Sports Medicine. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Position stand. Med Sci Sport Exerc. 2009;41:459-471. doi: 10.1249/MSS.0b013e3181949333
20. Fox SM 3rd, Naughton JP, Haskell WL. Physical activity and the prevention of coronary heart disease. Ann Clin Res. 1971;3:404-432.
21. Karvonen MJ, Kentala E, Mustala O. The effects of training on heart rate; a longitudinal study. Ann Med Exp Biol Fenn. 1957;35:307-315.
22. The Borg RPE scale. In: Borg G. Borg’s Perceived Exertion and Pain Scales. Human Kinetics; 1998:29-38.
23. Ratamess NA, Alvar BA, Evetoch TK, et al; American College of Sports Medicine. Progression models in resistance training for healthy adults. Position stand. Med Sci Sport Exerc. 2009;41:687-708. doi: 10.1249/MSS.0b013e3181915670
24. Gym memberships & fitness programs. Medicare.gov. Baltimore, MD: US Centers for Medicare and Medicaid Services. Accessed November 16, 2021. www.medicare.gov/coverage/gym-memberships-fitness-programs
25. Riebe D, Franklin BA, Thompson PD, et al. Updating ACSM’s recommendations for exercise preparticipation health screening. Med Sci Sports Exerc. 2015;47:2473-2479. doi: 10.1249/MSS.0000000000000664
26. Physical Activity Related Current Procedural Terminology (CPT®) Codes. Physical Activity Alliance website. Accessed November 16, 2021. https://paamovewithus.org/wp-content/uploads/2020/11/PAA-Physical-Activity-CPT-Codes-Nov-2020-AMA-Approved-Final-1.pdf
27. Blair SN. Physical inactivity: the biggest public health problem of the 21st century Br J Sports Med. 2009;43:1-2.
28. Vuori IM, Lavie CJ, Blair SN. Physical activity promotion in the health care system. Mayo Clin Proc. 2013;88:1446-1461. doi: 10.1016/j.mayocp.2013.08.020
1. Papanicolas I, Woskie LR, Jha AK. Health care spending in the United States and other high-income countries. JAMA. 2018;319:1024-1039. doi: 10.1001/jama.2018.1150
2. Tikkanen R, Abrams MK. U.S. health care from a global perspective, 2019: higher spending, worse outcomes? The Commonwealth Fund Website. January 30, 2020. Accessed November 16, 2021. www.commonwealthfund.org/publications/issue-briefs/2020/jan/us-health-care-global-perspective-2019
3. Stoutenberg M, Stasi S, Stamatakis E, et al. Physical activity training in US medical schools: preparing future physicians to engage in primary prevention. Phys Sportsmed. 2015;43:388-394. doi: 10.1080/00913847.2015.1084868
4. Antognoli EL, Seeholzer EL, Gullett H, et al. Primary care resident training for obesity, nutrition, and physical activity counseling: a mixed-methods study. Health Promot Pract. 2017;18:672-680. doi: 10.1177/1524839916658025
5. Asif IM, Drezner JA. Sports and exercise medicine education in the USA: call to action. Br J Sports Med. 2020;54:195-196. doi: 10.1136/bjsports-2019-101104
6. Douglas JA, Briones MD, Bauer EZ, et al. Social and environmental determinants of physical activity in urban parks: testing a neighborhood disorder model. Prev Med. 2018;109:119-124. doi: 10.1016/j.ypmed.2018.01.013
7. 2018 Physical Activity Guidelines Advisory Committee. 2018 Physical Activity Guidelines Advisory Committee Scientific Report. Washington, DC: US Department of Health & Human Services; 2018. Accessed November 15, 2021. https://health.gov/sites/default/files/2019-09/PAG_Advisory_Committee_Report.pdf
8. Avis JL, Cave AL, Donaldson S, et al. Working with parents to prevent childhood obesity: protocol for a primary care-based ehealth study. JMIR Res Protoc. 2015;4:e35. doi:10.2196/resprot.4147
9. Ball TJ, Joy EA, Gren LH, et al. Concurrent validity of a self-reported physical activity ‘vital sign’ questionnaire with adult primary care patients. Prev Chronic Dis. 2016;13:e16. doi: 10.5888/pcd13.150228
10. Ball TJ, Joy EA, Gren LH, et al. Predictive validity of an adult physical activity “vital sign” recorded in electronic health records. J Phys Act Health. 2016;13:403-408. doi: 10.1123/jpah.2015-0210
11. Coleman KJ, Ngor E, Reynolds K, et al. Initial validation of an exercise “vital sign” in electronic medical records. Med Sci Sports Exerc. 2012;44:2071-2076. doi: 10.1249/MSS.0b013e3182630ec1
12. Ross R, Blair SN, Arena R, et al; ; ; ; ; ; . Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation. 2016;134:e653-e699. doi: 10.1161/CIR.0000000000000461
13. de Souza de Silva CG, Kokkinos PP, Doom R, et al. Association between cardiorespiratory fitness, obesity, and health care costs: The Veterans Exercise Testing Study. Int J Obes (Lond). 2019;43:2225-2232. doi: 10.1038/s41366-018-0257-0
14. Prochaska JO, Velicer WF. The transtheoretical model of health behavior change. Am J Health Promot. 1997;12:38-48. doi: 10.4278/0890-1171-12.1.38
15. Riebe D, Ehrman JK, Liguori G, et al. Methods for evoking change talk. In: ACSM’s Guidelines for Exercise Testing and Prescription. 10th ed. Wolters Kluwer; 2018.
16. Grandes G, Sanchez A, Sanchez-Pinilla RO, et al. Effectiveness of physical activity advice and prescription by physicians in routine primary care: a cluster randomized trial. Arch Intern Med. 2009;169:694-701. doi: 10.1001/archinternmed.2009.23
17. McNeill LH, Kreuter MW, Subramanian SV. Social environment and physical activity: a review of concepts and evidence. Soc Sci Med. 2006;63:1011-1022. doi: 10.1016/j.socscimed.2006.03.012
18. Garber CE, Blissmer BE, Deschenes MR, et al; American College of Sports Medicine. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Position stand. Med Sci Sport Exerc. 2011;43:1334-1359. doi: 10.1249/MSS.0b013e318213fefb
19. Donnelly JE, Blair SN, Jakicic JM, et al; American College of Sports Medicine. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Position stand. Med Sci Sport Exerc. 2009;41:459-471. doi: 10.1249/MSS.0b013e3181949333
20. Fox SM 3rd, Naughton JP, Haskell WL. Physical activity and the prevention of coronary heart disease. Ann Clin Res. 1971;3:404-432.
21. Karvonen MJ, Kentala E, Mustala O. The effects of training on heart rate; a longitudinal study. Ann Med Exp Biol Fenn. 1957;35:307-315.
22. The Borg RPE scale. In: Borg G. Borg’s Perceived Exertion and Pain Scales. Human Kinetics; 1998:29-38.
23. Ratamess NA, Alvar BA, Evetoch TK, et al; American College of Sports Medicine. Progression models in resistance training for healthy adults. Position stand. Med Sci Sport Exerc. 2009;41:687-708. doi: 10.1249/MSS.0b013e3181915670
24. Gym memberships & fitness programs. Medicare.gov. Baltimore, MD: US Centers for Medicare and Medicaid Services. Accessed November 16, 2021. www.medicare.gov/coverage/gym-memberships-fitness-programs
25. Riebe D, Franklin BA, Thompson PD, et al. Updating ACSM’s recommendations for exercise preparticipation health screening. Med Sci Sports Exerc. 2015;47:2473-2479. doi: 10.1249/MSS.0000000000000664
26. Physical Activity Related Current Procedural Terminology (CPT®) Codes. Physical Activity Alliance website. Accessed November 16, 2021. https://paamovewithus.org/wp-content/uploads/2020/11/PAA-Physical-Activity-CPT-Codes-Nov-2020-AMA-Approved-Final-1.pdf
27. Blair SN. Physical inactivity: the biggest public health problem of the 21st century Br J Sports Med. 2009;43:1-2.
28. Vuori IM, Lavie CJ, Blair SN. Physical activity promotion in the health care system. Mayo Clin Proc. 2013;88:1446-1461. doi: 10.1016/j.mayocp.2013.08.020
PRACTICE RECOMMENDATIONS
› Encourage children and adolescents (6 to 17 years of age) to engage in 60 min of moderate-to-vigorous physical activity, including aerobic, muscle-strengthening, and bone-strengthening endeavors on most, if not all, days of the week. A
› Encourage adults to perform approximately 150 to 300 min of moderate or 75 to 150 min of vigorous physical activity (or an equivalent combination) per week, along with moderate-intensity muscle-strengthening activities on ≥ 2 days per week. A
› Counsel patients that even a small (eg, 1-2 metabolic equivalents) increase in cardiorespiratory fitness is associated with a 10% to 30% lower rate of adverse events. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Could Viagra help prevent Alzheimer’s?
published in the journal Nature Aging.
Patients who used the drug sildenafil, the generic name for Viagra, were 69% less likely to develop the disease than were nonusers.
“Sildenafil, which has been shown to significantly improve cognition and memory in preclinical models, presented as the best drug candidate,” Feixiong Cheng, PhD, the lead study author in the Cleveland Clinic’s Genomic Medicine Institute, said in a statement.
“Notably, we found that sildenafil use reduced the likelihood of Alzheimer’s in individuals with coronary artery disease, hypertension, and type 2 diabetes, all of which are comorbidities significantly associated with risk of the disease, as well as in those without,” he said.
Alzheimer’s, which is the most common form of age-related dementia, affects hundreds of millions of people worldwide. The disease is expected to affect nearly 14 million Americans by 2050. There is no approved treatment for it.
Dr. Cheng and colleagues at the Cleveland Clinic used a large gene-mapping network to analyze whether more than 1,600 Food and Drug Administration–approved drugs could work against Alzheimer’s. They gave higher scores to drugs that target both amyloid and tau proteins in the brain, which are two hallmarks of the disease. Sildenafil appeared at the top of the list.
Then the researchers used a database of health insurance claims for more than 7 million people in the U.S. to understand the relationship between sildenafil and Alzheimer’s disease outcomes. They compared sildenafil users to nonusers and found that those who used the drug were 69% less likely to have the neurodegenerative disease, even after 6 years of follow-up.
After that, the research team came up with a lab model that showed the sildenafil increased brain cell growth and targeted tau proteins. The lab model could indicate how the drug influences disease-related brain changes.
But Dr. Cheng cautioned against drawing strong conclusions. The study doesn’t demonstrate a causal relationship between sildenafil and Alzheimer’s disease. Researchers will need to conduct clinical trials with a placebo control to see how well the drug works.
Other researchers said the findings offer a new avenue for research but don’t yet provide solid answers.
“Being able to repurpose a drug already licensed for health conditions could help speed up the drug discovery process and bring about life-changing dementia treatments sooner,” Susan Kohlhaas, PhD, director of research at Alzheimer’s Research UK, told the Science Media Centre.
“Importantly, this research doesn’t prove that sildenafil is responsible for reducing dementia risk, or that it slows or stops the disease,” she continued. “If you want to discuss any treatments you are receiving, the first port of call is to speak to your doctor.”
And doctors won’t likely recommend it as a treatment just yet either.
“While these data are interesting scientifically, based on this study, I would not rush out to start taking sildenafil as a prevention for Alzheimer’s disease,” Tara Spires-Jones, PhD, deputy director of the Centre for Discovery Brain Sciences at the University of Edinburgh, told the Science Media Centre.
A version of this article first appeared on WebMD.com.
published in the journal Nature Aging.
Patients who used the drug sildenafil, the generic name for Viagra, were 69% less likely to develop the disease than were nonusers.
“Sildenafil, which has been shown to significantly improve cognition and memory in preclinical models, presented as the best drug candidate,” Feixiong Cheng, PhD, the lead study author in the Cleveland Clinic’s Genomic Medicine Institute, said in a statement.
“Notably, we found that sildenafil use reduced the likelihood of Alzheimer’s in individuals with coronary artery disease, hypertension, and type 2 diabetes, all of which are comorbidities significantly associated with risk of the disease, as well as in those without,” he said.
Alzheimer’s, which is the most common form of age-related dementia, affects hundreds of millions of people worldwide. The disease is expected to affect nearly 14 million Americans by 2050. There is no approved treatment for it.
Dr. Cheng and colleagues at the Cleveland Clinic used a large gene-mapping network to analyze whether more than 1,600 Food and Drug Administration–approved drugs could work against Alzheimer’s. They gave higher scores to drugs that target both amyloid and tau proteins in the brain, which are two hallmarks of the disease. Sildenafil appeared at the top of the list.
Then the researchers used a database of health insurance claims for more than 7 million people in the U.S. to understand the relationship between sildenafil and Alzheimer’s disease outcomes. They compared sildenafil users to nonusers and found that those who used the drug were 69% less likely to have the neurodegenerative disease, even after 6 years of follow-up.
After that, the research team came up with a lab model that showed the sildenafil increased brain cell growth and targeted tau proteins. The lab model could indicate how the drug influences disease-related brain changes.
But Dr. Cheng cautioned against drawing strong conclusions. The study doesn’t demonstrate a causal relationship between sildenafil and Alzheimer’s disease. Researchers will need to conduct clinical trials with a placebo control to see how well the drug works.
Other researchers said the findings offer a new avenue for research but don’t yet provide solid answers.
“Being able to repurpose a drug already licensed for health conditions could help speed up the drug discovery process and bring about life-changing dementia treatments sooner,” Susan Kohlhaas, PhD, director of research at Alzheimer’s Research UK, told the Science Media Centre.
“Importantly, this research doesn’t prove that sildenafil is responsible for reducing dementia risk, or that it slows or stops the disease,” she continued. “If you want to discuss any treatments you are receiving, the first port of call is to speak to your doctor.”
And doctors won’t likely recommend it as a treatment just yet either.
“While these data are interesting scientifically, based on this study, I would not rush out to start taking sildenafil as a prevention for Alzheimer’s disease,” Tara Spires-Jones, PhD, deputy director of the Centre for Discovery Brain Sciences at the University of Edinburgh, told the Science Media Centre.
A version of this article first appeared on WebMD.com.
published in the journal Nature Aging.
Patients who used the drug sildenafil, the generic name for Viagra, were 69% less likely to develop the disease than were nonusers.
“Sildenafil, which has been shown to significantly improve cognition and memory in preclinical models, presented as the best drug candidate,” Feixiong Cheng, PhD, the lead study author in the Cleveland Clinic’s Genomic Medicine Institute, said in a statement.
“Notably, we found that sildenafil use reduced the likelihood of Alzheimer’s in individuals with coronary artery disease, hypertension, and type 2 diabetes, all of which are comorbidities significantly associated with risk of the disease, as well as in those without,” he said.
Alzheimer’s, which is the most common form of age-related dementia, affects hundreds of millions of people worldwide. The disease is expected to affect nearly 14 million Americans by 2050. There is no approved treatment for it.
Dr. Cheng and colleagues at the Cleveland Clinic used a large gene-mapping network to analyze whether more than 1,600 Food and Drug Administration–approved drugs could work against Alzheimer’s. They gave higher scores to drugs that target both amyloid and tau proteins in the brain, which are two hallmarks of the disease. Sildenafil appeared at the top of the list.
Then the researchers used a database of health insurance claims for more than 7 million people in the U.S. to understand the relationship between sildenafil and Alzheimer’s disease outcomes. They compared sildenafil users to nonusers and found that those who used the drug were 69% less likely to have the neurodegenerative disease, even after 6 years of follow-up.
After that, the research team came up with a lab model that showed the sildenafil increased brain cell growth and targeted tau proteins. The lab model could indicate how the drug influences disease-related brain changes.
But Dr. Cheng cautioned against drawing strong conclusions. The study doesn’t demonstrate a causal relationship between sildenafil and Alzheimer’s disease. Researchers will need to conduct clinical trials with a placebo control to see how well the drug works.
Other researchers said the findings offer a new avenue for research but don’t yet provide solid answers.
“Being able to repurpose a drug already licensed for health conditions could help speed up the drug discovery process and bring about life-changing dementia treatments sooner,” Susan Kohlhaas, PhD, director of research at Alzheimer’s Research UK, told the Science Media Centre.
“Importantly, this research doesn’t prove that sildenafil is responsible for reducing dementia risk, or that it slows or stops the disease,” she continued. “If you want to discuss any treatments you are receiving, the first port of call is to speak to your doctor.”
And doctors won’t likely recommend it as a treatment just yet either.
“While these data are interesting scientifically, based on this study, I would not rush out to start taking sildenafil as a prevention for Alzheimer’s disease,” Tara Spires-Jones, PhD, deputy director of the Centre for Discovery Brain Sciences at the University of Edinburgh, told the Science Media Centre.
A version of this article first appeared on WebMD.com.
FROM NATURE AGING
Higher resting heart rate tied to increased dementia risk
independent of the presence of cardiovascular disease (CVD) risk factors, new research shows.
“RHR is easy to measure and might be used to identify older people potentially at high risk of dementia and cognitive decline for early interventions,” Yume Imahori, MD, PhD, with the Aging Research Center, Karolinska Institutet, Stockholm, said in an interview.
“Health care professionals should be aware of potential cognitive consequences associated with elevated RHR in older people and may advise older people with high RHR to have a follow-up assessment of cognitive function,” Dr. Imahori said.
The study was published online Dec. 3, 2021, in Alzheimer’s & Dementia.
Heart-brain connection
The findings are based on 2,147 adults (62% women) aged 60 years and older (mean age, 70.6 years) from the population-based Swedish National Aging and Care in Kungsholmen (SNAC-K) study. All were free of dementia at baseline and were followed regularly from 2001-2004 to 2013-2016.
The average RHR at baseline was 65.7 bpm. Individuals in higher RHR groups were older, less educated, and were more likely to be smokers and sedentary and to have hypertension. There were no differences among RHR groups in the prevalence of CVD at baseline.
During a median follow-up of 11.4 years, 289 participants were diagnosed with dementia.
In the fully adjusted model, participants with RHR of 80 bpm or higher had a 55% increased risk of developing dementia, compared with peers with lower RHR of 60 to 69 bpm (hazard ratio, 1.55; 95% CI, 1.06-2.27).
“This association was not due to underlying cardiovascular diseases such as atrial fibrillation and heart failure, which is important because elevated RHR is often related to heart disease,” Dr. Imahori said in an interview.
Regarding cognitive function, Mini-Mental State Examination scores declined over time during the follow-up period in all RHR groups, but participants with RHR 70-79 and 80+ bpm had a greater decline, compared with those with lower RHR of 60-69 bpm.
Dr. Imahori said these findings are in line with data from the U.S. Atherosclerosis Risk in Communities study linking elevated RHR of 80+ bpm in midlife to dementia and cognitive decline in late life.
Public health implications
Reached for comment, Claire Sexton, DPhil, Alzheimer’s Association director of scientific programs and outreach, said this study adds to the “growing body of research showing the health of the heart and brain are closely connected. However, this study only shows a correlation between resting heart rate and cognition, not causation. More research is needed.
“Evidence shows that other risk factors for cardiovascular disease and stroke – obesity, high blood pressure, and diabetes – negatively impact your cognitive health,” Dr. Sexton said in an interview.
“The Alzheimer’s Association believes the conversation about heart health management is something everyone should be having with their doctor,” she said.
“There are things you can do today to lower your risk for cardiovascular disease, including regular exercise and maintaining a healthy diet. Improving your heart health is an important step to maintaining your brain health as you age,” Dr. Sexton added.
SNAC-K is supported by the Swedish Ministry of Health and Social Affairs and the participating county councils and municipalities and in part by additional grants from the Swedish Research Council and the Swedish Research Council for Health, Working Life and Welfare. Dr. Imahori and Dr. Sexton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
independent of the presence of cardiovascular disease (CVD) risk factors, new research shows.
“RHR is easy to measure and might be used to identify older people potentially at high risk of dementia and cognitive decline for early interventions,” Yume Imahori, MD, PhD, with the Aging Research Center, Karolinska Institutet, Stockholm, said in an interview.
“Health care professionals should be aware of potential cognitive consequences associated with elevated RHR in older people and may advise older people with high RHR to have a follow-up assessment of cognitive function,” Dr. Imahori said.
The study was published online Dec. 3, 2021, in Alzheimer’s & Dementia.
Heart-brain connection
The findings are based on 2,147 adults (62% women) aged 60 years and older (mean age, 70.6 years) from the population-based Swedish National Aging and Care in Kungsholmen (SNAC-K) study. All were free of dementia at baseline and were followed regularly from 2001-2004 to 2013-2016.
The average RHR at baseline was 65.7 bpm. Individuals in higher RHR groups were older, less educated, and were more likely to be smokers and sedentary and to have hypertension. There were no differences among RHR groups in the prevalence of CVD at baseline.
During a median follow-up of 11.4 years, 289 participants were diagnosed with dementia.
In the fully adjusted model, participants with RHR of 80 bpm or higher had a 55% increased risk of developing dementia, compared with peers with lower RHR of 60 to 69 bpm (hazard ratio, 1.55; 95% CI, 1.06-2.27).
“This association was not due to underlying cardiovascular diseases such as atrial fibrillation and heart failure, which is important because elevated RHR is often related to heart disease,” Dr. Imahori said in an interview.
Regarding cognitive function, Mini-Mental State Examination scores declined over time during the follow-up period in all RHR groups, but participants with RHR 70-79 and 80+ bpm had a greater decline, compared with those with lower RHR of 60-69 bpm.
Dr. Imahori said these findings are in line with data from the U.S. Atherosclerosis Risk in Communities study linking elevated RHR of 80+ bpm in midlife to dementia and cognitive decline in late life.
Public health implications
Reached for comment, Claire Sexton, DPhil, Alzheimer’s Association director of scientific programs and outreach, said this study adds to the “growing body of research showing the health of the heart and brain are closely connected. However, this study only shows a correlation between resting heart rate and cognition, not causation. More research is needed.
“Evidence shows that other risk factors for cardiovascular disease and stroke – obesity, high blood pressure, and diabetes – negatively impact your cognitive health,” Dr. Sexton said in an interview.
“The Alzheimer’s Association believes the conversation about heart health management is something everyone should be having with their doctor,” she said.
“There are things you can do today to lower your risk for cardiovascular disease, including regular exercise and maintaining a healthy diet. Improving your heart health is an important step to maintaining your brain health as you age,” Dr. Sexton added.
SNAC-K is supported by the Swedish Ministry of Health and Social Affairs and the participating county councils and municipalities and in part by additional grants from the Swedish Research Council and the Swedish Research Council for Health, Working Life and Welfare. Dr. Imahori and Dr. Sexton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
independent of the presence of cardiovascular disease (CVD) risk factors, new research shows.
“RHR is easy to measure and might be used to identify older people potentially at high risk of dementia and cognitive decline for early interventions,” Yume Imahori, MD, PhD, with the Aging Research Center, Karolinska Institutet, Stockholm, said in an interview.
“Health care professionals should be aware of potential cognitive consequences associated with elevated RHR in older people and may advise older people with high RHR to have a follow-up assessment of cognitive function,” Dr. Imahori said.
The study was published online Dec. 3, 2021, in Alzheimer’s & Dementia.
Heart-brain connection
The findings are based on 2,147 adults (62% women) aged 60 years and older (mean age, 70.6 years) from the population-based Swedish National Aging and Care in Kungsholmen (SNAC-K) study. All were free of dementia at baseline and were followed regularly from 2001-2004 to 2013-2016.
The average RHR at baseline was 65.7 bpm. Individuals in higher RHR groups were older, less educated, and were more likely to be smokers and sedentary and to have hypertension. There were no differences among RHR groups in the prevalence of CVD at baseline.
During a median follow-up of 11.4 years, 289 participants were diagnosed with dementia.
In the fully adjusted model, participants with RHR of 80 bpm or higher had a 55% increased risk of developing dementia, compared with peers with lower RHR of 60 to 69 bpm (hazard ratio, 1.55; 95% CI, 1.06-2.27).
“This association was not due to underlying cardiovascular diseases such as atrial fibrillation and heart failure, which is important because elevated RHR is often related to heart disease,” Dr. Imahori said in an interview.
Regarding cognitive function, Mini-Mental State Examination scores declined over time during the follow-up period in all RHR groups, but participants with RHR 70-79 and 80+ bpm had a greater decline, compared with those with lower RHR of 60-69 bpm.
Dr. Imahori said these findings are in line with data from the U.S. Atherosclerosis Risk in Communities study linking elevated RHR of 80+ bpm in midlife to dementia and cognitive decline in late life.
Public health implications
Reached for comment, Claire Sexton, DPhil, Alzheimer’s Association director of scientific programs and outreach, said this study adds to the “growing body of research showing the health of the heart and brain are closely connected. However, this study only shows a correlation between resting heart rate and cognition, not causation. More research is needed.
“Evidence shows that other risk factors for cardiovascular disease and stroke – obesity, high blood pressure, and diabetes – negatively impact your cognitive health,” Dr. Sexton said in an interview.
“The Alzheimer’s Association believes the conversation about heart health management is something everyone should be having with their doctor,” she said.
“There are things you can do today to lower your risk for cardiovascular disease, including regular exercise and maintaining a healthy diet. Improving your heart health is an important step to maintaining your brain health as you age,” Dr. Sexton added.
SNAC-K is supported by the Swedish Ministry of Health and Social Affairs and the participating county councils and municipalities and in part by additional grants from the Swedish Research Council and the Swedish Research Council for Health, Working Life and Welfare. Dr. Imahori and Dr. Sexton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALZHEIMER’S & DEMENTIA
No serious CV risks for elderly after Pfizer COVID-19 vaccine
A French population-based study provides further evidence that the BNT162b2 Pfizer-BioNTech mRNA COVID-19 vaccine does not increase the short-term risk for serious cardiovascular adverse events in older people.
The study showed no increased risk of myocardial infarction (MI), stroke, or pulmonary embolism (PE) following vaccination in adults aged 75 years or older in the 14 days following vaccination.
“These findings regarding the BNT162b2 vaccine’s short-term cardiovascular safety profile in older people are reassuring. They should be taken into account by doctors when considering implementing a third dose of the vaccine in older people,” Marie Joelle Jabagi, PharmD, PhD, with the French National Agency for Medicines and Health Products Safety, Saint-Denis, France, said in an interview.
The study was published as a research letter online Nov. 22 in JAMA.
The Pfizer-BioNTech mRNA vaccine was the first SARS-CoV-2 vaccine authorized in France and has been widely used in older people. The phase 3 trials of the vaccine showed no increase in cardiovascular events, but older people were underrepresented in the trials.
As of April 30, 2021, nearly 3.9 million French adults aged 75 or older had received at least one dose of the Pfizer COVID-19 vaccine and 3.2 million had received two doses.
Using the French National Health Data System linked to the national COVID-19 vaccination database, Dr. Jabagi and her colleagues identified all unvaccinated or vaccinated adults aged 75 and older who were hospitalized between Dec. 15, 2020, and April 30, 2021, for acute MI, hemorrhagic or ischemic stroke, or PE.
During the 4.5-month study period, 11,113 elderly were hospitalized for acute MI, 17,014 for ischemic stroke, 4,804 for hemorrhagic stroke, and 7,221 for PE. Of these, 58.6%, 54.0%, 42.7%, and 55.3%, respectively, had received at least one dose of vaccine.
In the 14 days following receipt of either dose, no significant increased risk was found for any outcome, the investigators report.
The relative incidence (RI) for MI after the first and second dose was 0.97 (95% CI, 0.88-1.06) and 1.04 (95% CI, 0.93-1.16), respectively.
For ischemic stroke, the RI was 0.90 after the first dose (95% CI, 0.84-0.98) and 0.92 (95% CI, 0.84-1.02) after the second; for hemorrhagic stroke, the RI was 0.90 (95% CI, 0.78-1.04) and 0.97 (95% CI, 0.81-1.15), respectively.
For PE, the RI was 0.85 (95% CI, 0.75-0.96) after the first dose and 1.10 (95% CI, 0.95-1.26) after the second dose.
There was also no significant increase for any of the cardiovascular events when the exposure risk window was subdivided into 1 to 7 days and 8 to 14 days.
“Evaluating the short-term risk of hospitalization for severe cardiovascular events after the BNT162b2 mRNA vaccine in older people was a priority, especially after signals for hypertension and cardiovascular, thromboembolic, and hemorrhagic events have been issued from spontaneous notification data,” Dr. Jabagi said in an interview.
“The results of this nationwide study provide further solid evidence regarding the lack of increase of serious cardiovascular adverse events in older people in the 14 days following both doses of the vaccine,” Dr. Jabagi said.
The French study supports a recent U.S. study of more than 6 million people demonstrating that serious health risks were no more common in the first 3 weeks after Pfizer/BioNTech or Moderna COVID-19 vaccination compared with 22 to 42 days later.
As previously reported by this news organization, mRNA vaccination was not associated with greater risks for Guillain-Barré syndrome, myocarditis/pericarditis, stroke, or 20 other serious outcomes.
The current study had no specific funding. Dr. Jabagi and colleagues have declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
A French population-based study provides further evidence that the BNT162b2 Pfizer-BioNTech mRNA COVID-19 vaccine does not increase the short-term risk for serious cardiovascular adverse events in older people.
The study showed no increased risk of myocardial infarction (MI), stroke, or pulmonary embolism (PE) following vaccination in adults aged 75 years or older in the 14 days following vaccination.
“These findings regarding the BNT162b2 vaccine’s short-term cardiovascular safety profile in older people are reassuring. They should be taken into account by doctors when considering implementing a third dose of the vaccine in older people,” Marie Joelle Jabagi, PharmD, PhD, with the French National Agency for Medicines and Health Products Safety, Saint-Denis, France, said in an interview.
The study was published as a research letter online Nov. 22 in JAMA.
The Pfizer-BioNTech mRNA vaccine was the first SARS-CoV-2 vaccine authorized in France and has been widely used in older people. The phase 3 trials of the vaccine showed no increase in cardiovascular events, but older people were underrepresented in the trials.
As of April 30, 2021, nearly 3.9 million French adults aged 75 or older had received at least one dose of the Pfizer COVID-19 vaccine and 3.2 million had received two doses.
Using the French National Health Data System linked to the national COVID-19 vaccination database, Dr. Jabagi and her colleagues identified all unvaccinated or vaccinated adults aged 75 and older who were hospitalized between Dec. 15, 2020, and April 30, 2021, for acute MI, hemorrhagic or ischemic stroke, or PE.
During the 4.5-month study period, 11,113 elderly were hospitalized for acute MI, 17,014 for ischemic stroke, 4,804 for hemorrhagic stroke, and 7,221 for PE. Of these, 58.6%, 54.0%, 42.7%, and 55.3%, respectively, had received at least one dose of vaccine.
In the 14 days following receipt of either dose, no significant increased risk was found for any outcome, the investigators report.
The relative incidence (RI) for MI after the first and second dose was 0.97 (95% CI, 0.88-1.06) and 1.04 (95% CI, 0.93-1.16), respectively.
For ischemic stroke, the RI was 0.90 after the first dose (95% CI, 0.84-0.98) and 0.92 (95% CI, 0.84-1.02) after the second; for hemorrhagic stroke, the RI was 0.90 (95% CI, 0.78-1.04) and 0.97 (95% CI, 0.81-1.15), respectively.
For PE, the RI was 0.85 (95% CI, 0.75-0.96) after the first dose and 1.10 (95% CI, 0.95-1.26) after the second dose.
There was also no significant increase for any of the cardiovascular events when the exposure risk window was subdivided into 1 to 7 days and 8 to 14 days.
“Evaluating the short-term risk of hospitalization for severe cardiovascular events after the BNT162b2 mRNA vaccine in older people was a priority, especially after signals for hypertension and cardiovascular, thromboembolic, and hemorrhagic events have been issued from spontaneous notification data,” Dr. Jabagi said in an interview.
“The results of this nationwide study provide further solid evidence regarding the lack of increase of serious cardiovascular adverse events in older people in the 14 days following both doses of the vaccine,” Dr. Jabagi said.
The French study supports a recent U.S. study of more than 6 million people demonstrating that serious health risks were no more common in the first 3 weeks after Pfizer/BioNTech or Moderna COVID-19 vaccination compared with 22 to 42 days later.
As previously reported by this news organization, mRNA vaccination was not associated with greater risks for Guillain-Barré syndrome, myocarditis/pericarditis, stroke, or 20 other serious outcomes.
The current study had no specific funding. Dr. Jabagi and colleagues have declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
A French population-based study provides further evidence that the BNT162b2 Pfizer-BioNTech mRNA COVID-19 vaccine does not increase the short-term risk for serious cardiovascular adverse events in older people.
The study showed no increased risk of myocardial infarction (MI), stroke, or pulmonary embolism (PE) following vaccination in adults aged 75 years or older in the 14 days following vaccination.
“These findings regarding the BNT162b2 vaccine’s short-term cardiovascular safety profile in older people are reassuring. They should be taken into account by doctors when considering implementing a third dose of the vaccine in older people,” Marie Joelle Jabagi, PharmD, PhD, with the French National Agency for Medicines and Health Products Safety, Saint-Denis, France, said in an interview.
The study was published as a research letter online Nov. 22 in JAMA.
The Pfizer-BioNTech mRNA vaccine was the first SARS-CoV-2 vaccine authorized in France and has been widely used in older people. The phase 3 trials of the vaccine showed no increase in cardiovascular events, but older people were underrepresented in the trials.
As of April 30, 2021, nearly 3.9 million French adults aged 75 or older had received at least one dose of the Pfizer COVID-19 vaccine and 3.2 million had received two doses.
Using the French National Health Data System linked to the national COVID-19 vaccination database, Dr. Jabagi and her colleagues identified all unvaccinated or vaccinated adults aged 75 and older who were hospitalized between Dec. 15, 2020, and April 30, 2021, for acute MI, hemorrhagic or ischemic stroke, or PE.
During the 4.5-month study period, 11,113 elderly were hospitalized for acute MI, 17,014 for ischemic stroke, 4,804 for hemorrhagic stroke, and 7,221 for PE. Of these, 58.6%, 54.0%, 42.7%, and 55.3%, respectively, had received at least one dose of vaccine.
In the 14 days following receipt of either dose, no significant increased risk was found for any outcome, the investigators report.
The relative incidence (RI) for MI after the first and second dose was 0.97 (95% CI, 0.88-1.06) and 1.04 (95% CI, 0.93-1.16), respectively.
For ischemic stroke, the RI was 0.90 after the first dose (95% CI, 0.84-0.98) and 0.92 (95% CI, 0.84-1.02) after the second; for hemorrhagic stroke, the RI was 0.90 (95% CI, 0.78-1.04) and 0.97 (95% CI, 0.81-1.15), respectively.
For PE, the RI was 0.85 (95% CI, 0.75-0.96) after the first dose and 1.10 (95% CI, 0.95-1.26) after the second dose.
There was also no significant increase for any of the cardiovascular events when the exposure risk window was subdivided into 1 to 7 days and 8 to 14 days.
“Evaluating the short-term risk of hospitalization for severe cardiovascular events after the BNT162b2 mRNA vaccine in older people was a priority, especially after signals for hypertension and cardiovascular, thromboembolic, and hemorrhagic events have been issued from spontaneous notification data,” Dr. Jabagi said in an interview.
“The results of this nationwide study provide further solid evidence regarding the lack of increase of serious cardiovascular adverse events in older people in the 14 days following both doses of the vaccine,” Dr. Jabagi said.
The French study supports a recent U.S. study of more than 6 million people demonstrating that serious health risks were no more common in the first 3 weeks after Pfizer/BioNTech or Moderna COVID-19 vaccination compared with 22 to 42 days later.
As previously reported by this news organization, mRNA vaccination was not associated with greater risks for Guillain-Barré syndrome, myocarditis/pericarditis, stroke, or 20 other serious outcomes.
The current study had no specific funding. Dr. Jabagi and colleagues have declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
For older adults, smelling the roses may be more difficult
Young and old alike are affected – more than 80%-90% of those diagnosed with the virus, according to some estimates. While most people recover in a few months, 16% take half a year or longer to do so, research has found. According to new estimates, up to 1.6 million Americans have chronic olfactory dysfunction due to COVID-19.
Seniors are especially vulnerable, experts suggest. “We know that many older adults have a compromised sense of smell to begin with. Add to that the insult of COVID, and it made these problems worse,” said Dr. Jayant Pinto, professor of surgery and a specialist in sinus and nasal diseases at the University of Chicago Medical Center.
Recent data highlight the interaction between COVID-19, advanced age, and loss of smell. When Italian researchers evaluated 101 patients who’d been hospitalized for mild to moderate COVID-19, 50 showed objective signs of smell impairment 6 months later. Those 65 or older were nearly twice as likely to be impaired; those 75 or older were more than 2½ times as likely.
Most people aren’t aware of the extent to which smell can be diminished in later life. More than half of 65- to 80-year-olds have some degree of smell loss, or olfactory dysfunction, as it’s known in the scientific literature. That rises to as high as 80% for those even older. People affected often report concerns about safety, less enjoyment eating, and an impaired quality of life.
But because the ability to detect, identify, and discriminate among odors declines gradually, most older adults – up to 75% of those with some degree of olfactory dysfunction – don’t realize they’re affected.
A host of factors are believed to contribute to age-related smell loss, including a reduction in the number of olfactory sensory neurons in the nose, which are essential for detecting odors; changes in stem cells that replenish these neurons every few months; atrophy of the processing center for smell in the brain, called the olfactory bulb; and the shrinkage of brain centers closely connected with the olfactory bulb, such as the hippocampus, a region central to learning and memory.
Also, environmental toxic substances such as air pollution play a part, research shows. “Olfactory neurons in your nose are basically little pieces of your brain hanging out in the outside world,” and exposure to them over time damages those neurons and the tissues that support them, explained Pamela Dalton, PhD, a principal investigator at the Monell Chemical Senses Center, a smell and taste research institute in Philadelphia.
Still, the complex workings of the olfactory system have not been mapped in detail yet, and much remains unknown, said Dr. Sandeep Robert Datta, professor of neurobiology at Harvard Medical School, Boston.
“We tend to think of our sense of smell as primarily aesthetic,” he said. “What’s very clear is that it’s far more important. The olfactory system plays a key role in maintaining our emotional well-being and connecting us with the world.”
Dr. Datta experienced this after having a bone marrow transplant followed by chemotherapy years ago. Unable to smell or taste food, he said, he felt “very disoriented” in his environment.
Common consequences of smell loss include a loss of appetite (without smell, taste is deeply compromised), difficulty monitoring personal hygiene, depression, and an inability to detect noxious fumes. In older adults, this can lead to weight loss, malnutrition, frailty, inadequate personal care, and accidents caused by gas leaks or fires.
Jerome Pisano, 75, of Bloomington, Ill., has been living with smell loss for 5 years. Repeated tests and consultations with physicians haven’t pinpointed a reason for this ailment, and sometimes he feels “hopeless,” he admitted.
Before he became smell-impaired, Mr. Pisano was certified as a wine specialist. He has an 800-bottle wine cellar. “I can’t appreciate that as much as I’d like. I miss the smell of cut grass. Flowers. My wife’s cooking,” he said. “It certainly does decrease my quality of life.”
Smell loss is also associated in various research studies with a higher risk of death for older adults. One study, authored by Dr. Pinto and colleagues, found that older adults with olfactory dysfunction were nearly three times as likely to die over a period of 5 years as were seniors whose sense of smell remained intact.
“Our sense of smell signals how our nervous system is doing and how well our brain is doing overall,” Dr. Pinto said. According to a review published earlier this year, 90% of people with early-stage Parkinson’s disease and more than 80% of people with Alzheimer’s disease have olfactory dysfunction – a symptom that can precede other symptoms by many years.
There is no treatment for smell loss associated with neurological illness or head trauma, but if someone has persistent sinus problems or allergies that cause congestion, an over-the-counter antihistamine or nasal steroid spray can help. Usually, smell returns in a few weeks.
For smell loss following a viral infection, the picture is less clear. It’s not known, yet, which viruses are associated with olfactory dysfunction, why they damage smell, and what trajectory recovery takes. COVID-19 may help shine a light on this since it has inspired a wave of research on olfaction loss around the world.
“What characteristics make people more vulnerable to a persistent loss of smell after a virus? We don’t know that, but I think we will because that research is underway and we’ve never had a cohort [of people with smell loss] this large to study,” said Dr. Dalton, of the Monell center.
Some experts recommend smell training, noting evidence of efficacy and no indication of harm. This involves sniffing four distinct scents (often eucalyptus, lemon, rose, and cloves) twice a day for 30 seconds each, usually for 4 weeks. Sometimes the practice is combined with pictures of the items being smelled, a form of visual reinforcement.
The theory is that “practice, practice, practice” will stimulate the olfactory system, said Charles Greer, PhD, professor of neurosurgery and neuroscience at Yale University, New Haven, Conn. Although scientific support isn’t well established, he said, he often recommends that people who think their smell is declining “get a shelf full of spices and smell them on a regular basis.”
Richard Doty, PhD, director of the University of Pennsylvania’s Smell and Taste Center, remains skeptical. He’s writing a review of smell training and notes that 20%-30% of people with viral infections and smell loss recover in a relatively short time, whether or not they pursue this therapy.
“The main thing we recommend is avoid polluted environments and get your full complement of vitamins,” since several vitamins play an important role in maintaining the olfactory system, he said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Young and old alike are affected – more than 80%-90% of those diagnosed with the virus, according to some estimates. While most people recover in a few months, 16% take half a year or longer to do so, research has found. According to new estimates, up to 1.6 million Americans have chronic olfactory dysfunction due to COVID-19.
Seniors are especially vulnerable, experts suggest. “We know that many older adults have a compromised sense of smell to begin with. Add to that the insult of COVID, and it made these problems worse,” said Dr. Jayant Pinto, professor of surgery and a specialist in sinus and nasal diseases at the University of Chicago Medical Center.
Recent data highlight the interaction between COVID-19, advanced age, and loss of smell. When Italian researchers evaluated 101 patients who’d been hospitalized for mild to moderate COVID-19, 50 showed objective signs of smell impairment 6 months later. Those 65 or older were nearly twice as likely to be impaired; those 75 or older were more than 2½ times as likely.
Most people aren’t aware of the extent to which smell can be diminished in later life. More than half of 65- to 80-year-olds have some degree of smell loss, or olfactory dysfunction, as it’s known in the scientific literature. That rises to as high as 80% for those even older. People affected often report concerns about safety, less enjoyment eating, and an impaired quality of life.
But because the ability to detect, identify, and discriminate among odors declines gradually, most older adults – up to 75% of those with some degree of olfactory dysfunction – don’t realize they’re affected.
A host of factors are believed to contribute to age-related smell loss, including a reduction in the number of olfactory sensory neurons in the nose, which are essential for detecting odors; changes in stem cells that replenish these neurons every few months; atrophy of the processing center for smell in the brain, called the olfactory bulb; and the shrinkage of brain centers closely connected with the olfactory bulb, such as the hippocampus, a region central to learning and memory.
Also, environmental toxic substances such as air pollution play a part, research shows. “Olfactory neurons in your nose are basically little pieces of your brain hanging out in the outside world,” and exposure to them over time damages those neurons and the tissues that support them, explained Pamela Dalton, PhD, a principal investigator at the Monell Chemical Senses Center, a smell and taste research institute in Philadelphia.
Still, the complex workings of the olfactory system have not been mapped in detail yet, and much remains unknown, said Dr. Sandeep Robert Datta, professor of neurobiology at Harvard Medical School, Boston.
“We tend to think of our sense of smell as primarily aesthetic,” he said. “What’s very clear is that it’s far more important. The olfactory system plays a key role in maintaining our emotional well-being and connecting us with the world.”
Dr. Datta experienced this after having a bone marrow transplant followed by chemotherapy years ago. Unable to smell or taste food, he said, he felt “very disoriented” in his environment.
Common consequences of smell loss include a loss of appetite (without smell, taste is deeply compromised), difficulty monitoring personal hygiene, depression, and an inability to detect noxious fumes. In older adults, this can lead to weight loss, malnutrition, frailty, inadequate personal care, and accidents caused by gas leaks or fires.
Jerome Pisano, 75, of Bloomington, Ill., has been living with smell loss for 5 years. Repeated tests and consultations with physicians haven’t pinpointed a reason for this ailment, and sometimes he feels “hopeless,” he admitted.
Before he became smell-impaired, Mr. Pisano was certified as a wine specialist. He has an 800-bottle wine cellar. “I can’t appreciate that as much as I’d like. I miss the smell of cut grass. Flowers. My wife’s cooking,” he said. “It certainly does decrease my quality of life.”
Smell loss is also associated in various research studies with a higher risk of death for older adults. One study, authored by Dr. Pinto and colleagues, found that older adults with olfactory dysfunction were nearly three times as likely to die over a period of 5 years as were seniors whose sense of smell remained intact.
“Our sense of smell signals how our nervous system is doing and how well our brain is doing overall,” Dr. Pinto said. According to a review published earlier this year, 90% of people with early-stage Parkinson’s disease and more than 80% of people with Alzheimer’s disease have olfactory dysfunction – a symptom that can precede other symptoms by many years.
There is no treatment for smell loss associated with neurological illness or head trauma, but if someone has persistent sinus problems or allergies that cause congestion, an over-the-counter antihistamine or nasal steroid spray can help. Usually, smell returns in a few weeks.
For smell loss following a viral infection, the picture is less clear. It’s not known, yet, which viruses are associated with olfactory dysfunction, why they damage smell, and what trajectory recovery takes. COVID-19 may help shine a light on this since it has inspired a wave of research on olfaction loss around the world.
“What characteristics make people more vulnerable to a persistent loss of smell after a virus? We don’t know that, but I think we will because that research is underway and we’ve never had a cohort [of people with smell loss] this large to study,” said Dr. Dalton, of the Monell center.
Some experts recommend smell training, noting evidence of efficacy and no indication of harm. This involves sniffing four distinct scents (often eucalyptus, lemon, rose, and cloves) twice a day for 30 seconds each, usually for 4 weeks. Sometimes the practice is combined with pictures of the items being smelled, a form of visual reinforcement.
The theory is that “practice, practice, practice” will stimulate the olfactory system, said Charles Greer, PhD, professor of neurosurgery and neuroscience at Yale University, New Haven, Conn. Although scientific support isn’t well established, he said, he often recommends that people who think their smell is declining “get a shelf full of spices and smell them on a regular basis.”
Richard Doty, PhD, director of the University of Pennsylvania’s Smell and Taste Center, remains skeptical. He’s writing a review of smell training and notes that 20%-30% of people with viral infections and smell loss recover in a relatively short time, whether or not they pursue this therapy.
“The main thing we recommend is avoid polluted environments and get your full complement of vitamins,” since several vitamins play an important role in maintaining the olfactory system, he said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Young and old alike are affected – more than 80%-90% of those diagnosed with the virus, according to some estimates. While most people recover in a few months, 16% take half a year or longer to do so, research has found. According to new estimates, up to 1.6 million Americans have chronic olfactory dysfunction due to COVID-19.
Seniors are especially vulnerable, experts suggest. “We know that many older adults have a compromised sense of smell to begin with. Add to that the insult of COVID, and it made these problems worse,” said Dr. Jayant Pinto, professor of surgery and a specialist in sinus and nasal diseases at the University of Chicago Medical Center.
Recent data highlight the interaction between COVID-19, advanced age, and loss of smell. When Italian researchers evaluated 101 patients who’d been hospitalized for mild to moderate COVID-19, 50 showed objective signs of smell impairment 6 months later. Those 65 or older were nearly twice as likely to be impaired; those 75 or older were more than 2½ times as likely.
Most people aren’t aware of the extent to which smell can be diminished in later life. More than half of 65- to 80-year-olds have some degree of smell loss, or olfactory dysfunction, as it’s known in the scientific literature. That rises to as high as 80% for those even older. People affected often report concerns about safety, less enjoyment eating, and an impaired quality of life.
But because the ability to detect, identify, and discriminate among odors declines gradually, most older adults – up to 75% of those with some degree of olfactory dysfunction – don’t realize they’re affected.
A host of factors are believed to contribute to age-related smell loss, including a reduction in the number of olfactory sensory neurons in the nose, which are essential for detecting odors; changes in stem cells that replenish these neurons every few months; atrophy of the processing center for smell in the brain, called the olfactory bulb; and the shrinkage of brain centers closely connected with the olfactory bulb, such as the hippocampus, a region central to learning and memory.
Also, environmental toxic substances such as air pollution play a part, research shows. “Olfactory neurons in your nose are basically little pieces of your brain hanging out in the outside world,” and exposure to them over time damages those neurons and the tissues that support them, explained Pamela Dalton, PhD, a principal investigator at the Monell Chemical Senses Center, a smell and taste research institute in Philadelphia.
Still, the complex workings of the olfactory system have not been mapped in detail yet, and much remains unknown, said Dr. Sandeep Robert Datta, professor of neurobiology at Harvard Medical School, Boston.
“We tend to think of our sense of smell as primarily aesthetic,” he said. “What’s very clear is that it’s far more important. The olfactory system plays a key role in maintaining our emotional well-being and connecting us with the world.”
Dr. Datta experienced this after having a bone marrow transplant followed by chemotherapy years ago. Unable to smell or taste food, he said, he felt “very disoriented” in his environment.
Common consequences of smell loss include a loss of appetite (without smell, taste is deeply compromised), difficulty monitoring personal hygiene, depression, and an inability to detect noxious fumes. In older adults, this can lead to weight loss, malnutrition, frailty, inadequate personal care, and accidents caused by gas leaks or fires.
Jerome Pisano, 75, of Bloomington, Ill., has been living with smell loss for 5 years. Repeated tests and consultations with physicians haven’t pinpointed a reason for this ailment, and sometimes he feels “hopeless,” he admitted.
Before he became smell-impaired, Mr. Pisano was certified as a wine specialist. He has an 800-bottle wine cellar. “I can’t appreciate that as much as I’d like. I miss the smell of cut grass. Flowers. My wife’s cooking,” he said. “It certainly does decrease my quality of life.”
Smell loss is also associated in various research studies with a higher risk of death for older adults. One study, authored by Dr. Pinto and colleagues, found that older adults with olfactory dysfunction were nearly three times as likely to die over a period of 5 years as were seniors whose sense of smell remained intact.
“Our sense of smell signals how our nervous system is doing and how well our brain is doing overall,” Dr. Pinto said. According to a review published earlier this year, 90% of people with early-stage Parkinson’s disease and more than 80% of people with Alzheimer’s disease have olfactory dysfunction – a symptom that can precede other symptoms by many years.
There is no treatment for smell loss associated with neurological illness or head trauma, but if someone has persistent sinus problems or allergies that cause congestion, an over-the-counter antihistamine or nasal steroid spray can help. Usually, smell returns in a few weeks.
For smell loss following a viral infection, the picture is less clear. It’s not known, yet, which viruses are associated with olfactory dysfunction, why they damage smell, and what trajectory recovery takes. COVID-19 may help shine a light on this since it has inspired a wave of research on olfaction loss around the world.
“What characteristics make people more vulnerable to a persistent loss of smell after a virus? We don’t know that, but I think we will because that research is underway and we’ve never had a cohort [of people with smell loss] this large to study,” said Dr. Dalton, of the Monell center.
Some experts recommend smell training, noting evidence of efficacy and no indication of harm. This involves sniffing four distinct scents (often eucalyptus, lemon, rose, and cloves) twice a day for 30 seconds each, usually for 4 weeks. Sometimes the practice is combined with pictures of the items being smelled, a form of visual reinforcement.
The theory is that “practice, practice, practice” will stimulate the olfactory system, said Charles Greer, PhD, professor of neurosurgery and neuroscience at Yale University, New Haven, Conn. Although scientific support isn’t well established, he said, he often recommends that people who think their smell is declining “get a shelf full of spices and smell them on a regular basis.”
Richard Doty, PhD, director of the University of Pennsylvania’s Smell and Taste Center, remains skeptical. He’s writing a review of smell training and notes that 20%-30% of people with viral infections and smell loss recover in a relatively short time, whether or not they pursue this therapy.
“The main thing we recommend is avoid polluted environments and get your full complement of vitamins,” since several vitamins play an important role in maintaining the olfactory system, he said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
CBT prevents depression in up to 50% of patients with insomnia
Cognitive-behavioral therapy (CBT) is linked to a significantly reduced risk of depression in patients with insomnia, new research shows.
Insomnia affects over 50% of older adults, and insomnia contributes to a twofold greater risk for major depression, investigators noted.
“We show that by treating insomnia with a simple behavioral approach called Cognitive Behavioral Therapy for Insomnia, or CBT-I, you can reduce the likelihood of developing depression by over 50%,” lead author Michael R. Irwin, MD, Cousins Distinguished Professor of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, said in an interview.
The study is unique in that the treatment “is not just reducing depression, it’s preventing depression,” Dr. Irwin added.
The findings were published online Nov. 24 in JAMA Psychiatry.
Primary outcome met
within the previous 12 months.
All were randomly assigned to receive either CBT-I or Sleep Education Therapy (SET).
CBT-I is a first-line treatment for insomnia that includes five components: cognitive therapy targeting dysfunctional thoughts about sleep, stimulus control, sleep restriction, sleep hygiene, and relaxation.
SET provides information on behavioral and environmental factors contributing to poor sleep. While sleep education provides tips on improving sleep, CBT-I helps patients implement those changes and behaviors, Dr. Irwin noted.
Both interventions were delivered by trained personnel in weekly 120-minute group sessions for 2 months, consistent with the format and duration of most CBT-I trials.
The primary outcome was time to incident or recurrent major depressive disorder as diagnosed by the Structured Clinical Interview of the DSM-5 every 6 months during 36 months of follow-up. A monthly Patient Health Questionnaire 9 (PHQ-9) was used to screen for depressive symptoms.
Results showed depression occurred in 12.2% of the CBT-I group versus 25.9% of the SET group. The hazard ratio (HR) for depression in the CBT-I group compared with the SET group was 0.51 (95% confidence interval, 0.29-0.88; P = .02). The number needed to treat to prevent incident or recurrent depression was 7.3.
After adjustment for factors affecting depression risk such as sex, educational level, income, comorbidity, and history of depression, the HR for depression in the CBT-I group versus the SET group was 0.45 (95% CI, 0.23-0.86; P = .02).
Treatment with CBT-I yielded an annual 4.1% incidence of depression, which is similar to the population rate and half the rate in SET, which was 8.6%.
‘Remission is key’
The secondary outcome was sustained remission of insomnia disorder. The investigators found a greater proportion of the CBT-I group than the SET group achieved remission after treatment (50.7% vs. 37.7%; 95% CI, 0.10-0.93; P = .02).
“Remission is really key to the benefits that we’re seeing,” said Dr. Irwin.
Inflammation may explain why insomnia raises the risk for depression, he noted. “We know sleep disturbance can lead to inflammation and we also know inflammation can produce depression,” Dr. Irwin said.
It is also possible insomnia leads to an impaired pleasure or reward system, which is linked to depression, he added.
The authors noted that because insomnia is associated with suicidal ideation and dementia, CBT-I may reduce risk for suicide or cognitive decline.
While 8-week CBT-I treatments are readily available, “unfortunately, most clinicians will prescribe medications,” said Dr. Irwin. He noted that in older adults, drugs are linked to adverse events such as falls and cognitive problems.
These new results “really argue that psychology and psychiatry need to be fully integrated into what we call collaborative care models,” Dr. Irwin said.
There were no adverse events during treatment, and none of the serious events that occurred during follow-up were attributed to the trial.
Convincing argument?
Commenting on the findings for this news organization, Philip R. Muskin, MD, professor of psychiatry at Columbia University Irving Medical Center, New York, said the study was “nicely written” and the authors put forward “a very convincing argument” for CBT-I to prevent depression.
“It’s eye opening in that it’s a robust study; it’s carefully done; subjects were followed for a long period of time, and it’s an accessible treatment,” said Dr. Muskin, who was not involved with the research.
The study also shows “it’s possible to intervene in something we know is a risk factor in elderly people,” he added. “We think of older people as being less malleable to these kinds of things, but they’re not. They clearly participated, and there wasn’t a huge dropout rate.”
Dr. Muskin noted that less than half of the older participants were married or had a partner. He would have liked more information on this status because being widowed or divorced, as well as when this life change occurred, could affect vulnerability to depression.
The authors of an accompanying editorial called the study “seminal,” and noted that insomnia treatment possibly preventing depressive disorders is a “major finding.”
Proving this preventive strategy is effective in older adults will be important because “insomnia and depression are highly prevalent in this population and the uptake of both preventive and treatment services is low,” wrote Pim Cuijpers, PhD, department of clinical, neuro, and developmental psychology, Amsterdam Public Health Research Institute, and Charles F. Reynolds III, MD, department of psychiatry, University of Pittsburgh.
If the reduced rates of depression observed in the study could be generalized to the total population with insomnia, “the incidence of major depression could be reduced considerably,” they wrote.
“Can we prevent depression through interventions aimed at procrastination in college students, interventions aimed at perfectionism in perinatal women, stress management training for employees, social skills training in adolescents?” they asked.
This approach to preventing depressive disorders “offers all kinds of new opportunities to develop and test indirect interventions” for problems that are significantly associated with the onset of depression, the editorialists wrote.
The study was funded by a grant from the National Institute on Aging to the University of California, which partially supported the authors’ salaries. Dr. Irwin, Dr. Muskin, and Dr. Cuijpers have reported no relevant financial relationships. Dr. Reynolds reported being coinventor of the Pittsburgh Sleep Quality Index, for which he receives royalties.
A version of this article first appeared on Medscape.com.
Cognitive-behavioral therapy (CBT) is linked to a significantly reduced risk of depression in patients with insomnia, new research shows.
Insomnia affects over 50% of older adults, and insomnia contributes to a twofold greater risk for major depression, investigators noted.
“We show that by treating insomnia with a simple behavioral approach called Cognitive Behavioral Therapy for Insomnia, or CBT-I, you can reduce the likelihood of developing depression by over 50%,” lead author Michael R. Irwin, MD, Cousins Distinguished Professor of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, said in an interview.
The study is unique in that the treatment “is not just reducing depression, it’s preventing depression,” Dr. Irwin added.
The findings were published online Nov. 24 in JAMA Psychiatry.
Primary outcome met
within the previous 12 months.
All were randomly assigned to receive either CBT-I or Sleep Education Therapy (SET).
CBT-I is a first-line treatment for insomnia that includes five components: cognitive therapy targeting dysfunctional thoughts about sleep, stimulus control, sleep restriction, sleep hygiene, and relaxation.
SET provides information on behavioral and environmental factors contributing to poor sleep. While sleep education provides tips on improving sleep, CBT-I helps patients implement those changes and behaviors, Dr. Irwin noted.
Both interventions were delivered by trained personnel in weekly 120-minute group sessions for 2 months, consistent with the format and duration of most CBT-I trials.
The primary outcome was time to incident or recurrent major depressive disorder as diagnosed by the Structured Clinical Interview of the DSM-5 every 6 months during 36 months of follow-up. A monthly Patient Health Questionnaire 9 (PHQ-9) was used to screen for depressive symptoms.
Results showed depression occurred in 12.2% of the CBT-I group versus 25.9% of the SET group. The hazard ratio (HR) for depression in the CBT-I group compared with the SET group was 0.51 (95% confidence interval, 0.29-0.88; P = .02). The number needed to treat to prevent incident or recurrent depression was 7.3.
After adjustment for factors affecting depression risk such as sex, educational level, income, comorbidity, and history of depression, the HR for depression in the CBT-I group versus the SET group was 0.45 (95% CI, 0.23-0.86; P = .02).
Treatment with CBT-I yielded an annual 4.1% incidence of depression, which is similar to the population rate and half the rate in SET, which was 8.6%.
‘Remission is key’
The secondary outcome was sustained remission of insomnia disorder. The investigators found a greater proportion of the CBT-I group than the SET group achieved remission after treatment (50.7% vs. 37.7%; 95% CI, 0.10-0.93; P = .02).
“Remission is really key to the benefits that we’re seeing,” said Dr. Irwin.
Inflammation may explain why insomnia raises the risk for depression, he noted. “We know sleep disturbance can lead to inflammation and we also know inflammation can produce depression,” Dr. Irwin said.
It is also possible insomnia leads to an impaired pleasure or reward system, which is linked to depression, he added.
The authors noted that because insomnia is associated with suicidal ideation and dementia, CBT-I may reduce risk for suicide or cognitive decline.
While 8-week CBT-I treatments are readily available, “unfortunately, most clinicians will prescribe medications,” said Dr. Irwin. He noted that in older adults, drugs are linked to adverse events such as falls and cognitive problems.
These new results “really argue that psychology and psychiatry need to be fully integrated into what we call collaborative care models,” Dr. Irwin said.
There were no adverse events during treatment, and none of the serious events that occurred during follow-up were attributed to the trial.
Convincing argument?
Commenting on the findings for this news organization, Philip R. Muskin, MD, professor of psychiatry at Columbia University Irving Medical Center, New York, said the study was “nicely written” and the authors put forward “a very convincing argument” for CBT-I to prevent depression.
“It’s eye opening in that it’s a robust study; it’s carefully done; subjects were followed for a long period of time, and it’s an accessible treatment,” said Dr. Muskin, who was not involved with the research.
The study also shows “it’s possible to intervene in something we know is a risk factor in elderly people,” he added. “We think of older people as being less malleable to these kinds of things, but they’re not. They clearly participated, and there wasn’t a huge dropout rate.”
Dr. Muskin noted that less than half of the older participants were married or had a partner. He would have liked more information on this status because being widowed or divorced, as well as when this life change occurred, could affect vulnerability to depression.
The authors of an accompanying editorial called the study “seminal,” and noted that insomnia treatment possibly preventing depressive disorders is a “major finding.”
Proving this preventive strategy is effective in older adults will be important because “insomnia and depression are highly prevalent in this population and the uptake of both preventive and treatment services is low,” wrote Pim Cuijpers, PhD, department of clinical, neuro, and developmental psychology, Amsterdam Public Health Research Institute, and Charles F. Reynolds III, MD, department of psychiatry, University of Pittsburgh.
If the reduced rates of depression observed in the study could be generalized to the total population with insomnia, “the incidence of major depression could be reduced considerably,” they wrote.
“Can we prevent depression through interventions aimed at procrastination in college students, interventions aimed at perfectionism in perinatal women, stress management training for employees, social skills training in adolescents?” they asked.
This approach to preventing depressive disorders “offers all kinds of new opportunities to develop and test indirect interventions” for problems that are significantly associated with the onset of depression, the editorialists wrote.
The study was funded by a grant from the National Institute on Aging to the University of California, which partially supported the authors’ salaries. Dr. Irwin, Dr. Muskin, and Dr. Cuijpers have reported no relevant financial relationships. Dr. Reynolds reported being coinventor of the Pittsburgh Sleep Quality Index, for which he receives royalties.
A version of this article first appeared on Medscape.com.
Cognitive-behavioral therapy (CBT) is linked to a significantly reduced risk of depression in patients with insomnia, new research shows.
Insomnia affects over 50% of older adults, and insomnia contributes to a twofold greater risk for major depression, investigators noted.
“We show that by treating insomnia with a simple behavioral approach called Cognitive Behavioral Therapy for Insomnia, or CBT-I, you can reduce the likelihood of developing depression by over 50%,” lead author Michael R. Irwin, MD, Cousins Distinguished Professor of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, said in an interview.
The study is unique in that the treatment “is not just reducing depression, it’s preventing depression,” Dr. Irwin added.
The findings were published online Nov. 24 in JAMA Psychiatry.
Primary outcome met
within the previous 12 months.
All were randomly assigned to receive either CBT-I or Sleep Education Therapy (SET).
CBT-I is a first-line treatment for insomnia that includes five components: cognitive therapy targeting dysfunctional thoughts about sleep, stimulus control, sleep restriction, sleep hygiene, and relaxation.
SET provides information on behavioral and environmental factors contributing to poor sleep. While sleep education provides tips on improving sleep, CBT-I helps patients implement those changes and behaviors, Dr. Irwin noted.
Both interventions were delivered by trained personnel in weekly 120-minute group sessions for 2 months, consistent with the format and duration of most CBT-I trials.
The primary outcome was time to incident or recurrent major depressive disorder as diagnosed by the Structured Clinical Interview of the DSM-5 every 6 months during 36 months of follow-up. A monthly Patient Health Questionnaire 9 (PHQ-9) was used to screen for depressive symptoms.
Results showed depression occurred in 12.2% of the CBT-I group versus 25.9% of the SET group. The hazard ratio (HR) for depression in the CBT-I group compared with the SET group was 0.51 (95% confidence interval, 0.29-0.88; P = .02). The number needed to treat to prevent incident or recurrent depression was 7.3.
After adjustment for factors affecting depression risk such as sex, educational level, income, comorbidity, and history of depression, the HR for depression in the CBT-I group versus the SET group was 0.45 (95% CI, 0.23-0.86; P = .02).
Treatment with CBT-I yielded an annual 4.1% incidence of depression, which is similar to the population rate and half the rate in SET, which was 8.6%.
‘Remission is key’
The secondary outcome was sustained remission of insomnia disorder. The investigators found a greater proportion of the CBT-I group than the SET group achieved remission after treatment (50.7% vs. 37.7%; 95% CI, 0.10-0.93; P = .02).
“Remission is really key to the benefits that we’re seeing,” said Dr. Irwin.
Inflammation may explain why insomnia raises the risk for depression, he noted. “We know sleep disturbance can lead to inflammation and we also know inflammation can produce depression,” Dr. Irwin said.
It is also possible insomnia leads to an impaired pleasure or reward system, which is linked to depression, he added.
The authors noted that because insomnia is associated with suicidal ideation and dementia, CBT-I may reduce risk for suicide or cognitive decline.
While 8-week CBT-I treatments are readily available, “unfortunately, most clinicians will prescribe medications,” said Dr. Irwin. He noted that in older adults, drugs are linked to adverse events such as falls and cognitive problems.
These new results “really argue that psychology and psychiatry need to be fully integrated into what we call collaborative care models,” Dr. Irwin said.
There were no adverse events during treatment, and none of the serious events that occurred during follow-up were attributed to the trial.
Convincing argument?
Commenting on the findings for this news organization, Philip R. Muskin, MD, professor of psychiatry at Columbia University Irving Medical Center, New York, said the study was “nicely written” and the authors put forward “a very convincing argument” for CBT-I to prevent depression.
“It’s eye opening in that it’s a robust study; it’s carefully done; subjects were followed for a long period of time, and it’s an accessible treatment,” said Dr. Muskin, who was not involved with the research.
The study also shows “it’s possible to intervene in something we know is a risk factor in elderly people,” he added. “We think of older people as being less malleable to these kinds of things, but they’re not. They clearly participated, and there wasn’t a huge dropout rate.”
Dr. Muskin noted that less than half of the older participants were married or had a partner. He would have liked more information on this status because being widowed or divorced, as well as when this life change occurred, could affect vulnerability to depression.
The authors of an accompanying editorial called the study “seminal,” and noted that insomnia treatment possibly preventing depressive disorders is a “major finding.”
Proving this preventive strategy is effective in older adults will be important because “insomnia and depression are highly prevalent in this population and the uptake of both preventive and treatment services is low,” wrote Pim Cuijpers, PhD, department of clinical, neuro, and developmental psychology, Amsterdam Public Health Research Institute, and Charles F. Reynolds III, MD, department of psychiatry, University of Pittsburgh.
If the reduced rates of depression observed in the study could be generalized to the total population with insomnia, “the incidence of major depression could be reduced considerably,” they wrote.
“Can we prevent depression through interventions aimed at procrastination in college students, interventions aimed at perfectionism in perinatal women, stress management training for employees, social skills training in adolescents?” they asked.
This approach to preventing depressive disorders “offers all kinds of new opportunities to develop and test indirect interventions” for problems that are significantly associated with the onset of depression, the editorialists wrote.
The study was funded by a grant from the National Institute on Aging to the University of California, which partially supported the authors’ salaries. Dr. Irwin, Dr. Muskin, and Dr. Cuijpers have reported no relevant financial relationships. Dr. Reynolds reported being coinventor of the Pittsburgh Sleep Quality Index, for which he receives royalties.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Preoperative Code Status Discussion in Older Adults: Are We Doing Enough?
Study Overview
Objective. The objective of this study was to evaluate orders and documentation describing perioperative management of code status in adults.
Design. A retrospective case series of all adult inpatients admitted to hospitals at 1 academic health system in the US.
Setting and participants. This retrospective case series was conducted at 5 hospitals within the University of Pennsylvania Health System. Cases included all adult inpatients admitted to hospitals between March 2017 and September 2018 who had a Do-Not-Resuscitate (DNR) order placed in their medical record during admission and subsequently underwent a surgical procedure that required anesthesia care.
Main outcome measures. Medical records of included cases were manually reviewed by the authors to verify whether a DNR order was in place at the time surgical intervention was discussed with a patient. Clinical notes and DNR orders of eligible cases were reviewed to identify documentation and outcome of goals of care discussions that were conducted within 48 hours prior to the surgical procedure. Collected data included patient demographics (age, sex, race); case characteristics (American Society of Anesthesiologists [ASA] physical status score, anesthesia type [general vs others such as regional], emergency status [emergent vs elective surgery], procedures by service [surgical including hip fracture repair, gastrostomy or jejunostomy, or exploratory laparotomy vs medical including endoscopy, bronchoscopy, or transesophageal echocardiogram]); and hospital policy for perioperative management of DNR orders (written policy encouraging discussion vs written policy plus additional initiatives, including procedure-specific DNR form). The primary outcome was the presence of a preoperative order or note documenting code status discussion or change. Data were analyzed using χ2 and Fisher exact tests and the threshold for statistical significance was P < .05.
Main results. Of the 27 665 inpatient procedures identified across 5 hospitals, 444 (1.6%) cases met the inclusion criteria. Patients from these cases aged 75 (SD 13) years (95% CI, 72-77 years); 247 (56%, 95% CI, 55%-57%) were women; and 300 (68%, 95% CI, 65%-71%) were White. A total of 426 patients (96%, 95% CI, 90%-100%) had an ASA physical status score of 3 or higher and 237 (53%, 95% CI, 51%-56%) received general anesthesia. The most common procedures performed were endoscopy (148 [33%]), hip fracture repair (43 [10%]), and gastrostomy or jejunostomy (28 [6%]). Reevaluation of code status was documented in 126 cases (28%, 95% CI, 25%-31%); code status orders were changed in 20 of 126 cases (16%, 95% CI, 7%-24%); and a note was filed without a corresponding order for 106 of 126 cases (84%, 95% CI, 75%-95%). In the majority of cases (109 of 126 [87%], 95% CI, 78%-95%) in which documented discussion occurred, DNR orders were suspended. Of 126 cases in which a discussion was documented, participants of these discussions included surgeons 10% of the time (13 cases, 95% CI, 8%-13%), members of the anesthesia team 51% of the time (64 cases, 95% CI, 49%-53%), and medicine or palliative care clinicians 39% of the time (49 cases, 95% CI, 37%-41%).
The rate of documented preoperative code status discussion was higher in patients with higher ASA physical status score (35% in patients with an ASA physical status score ≥ 4 [55 of 155] vs 25% in those with an ASA physical status score ≤ 3 [71 of 289]; P = .02). The rates of documented preoperative code status discussion were similar by anesthesia type (29% for general anesthesia [69 of 237 cases] vs 28% [57 of 207 cases] for other modalities; P = .70). The hospitals involved in this study all had a written policy encouraging rediscussion of code status before surgery. However, only 1 hospital reported added measures (eg, provision of a procedure-specific DNR form) to increase documentation of preoperative code status discussions. In this specific hospital, documentation of preoperative code status discussions was higher compared to other hospitals (67% [37 of 55 cases] vs 23% [89 of 389 cases]; P < .01).
Conclusion. In a retrospective case series conducted at 5 hospitals within 1 academic health system in the US, fewer than 1 in 5 patients with preexisting DNR orders had a documented discussion of code status prior to undergoing surgery. Additional strategies including the development of institutional protocols that facilitate perioperative management of advance directives, identification of local champions, and patient education, should be explored as means to improve preoperative code status reevaulation per guideline recommendations.
Commentary
It is not unusual that patients with a DNR order may require and undergo surgical interventions to treat reversible conditions, prevent progression of underlying disease, or mitigate distressing symptoms such as pain. For instance, intubation, mechanical ventilation, and administration of vasoactive drugs are resuscitative measures that may be needed to safely anesthetize and sedate a patient. As such, the American College of Surgeons1 has provided a statement on advance directives by patients with an existing DNR order to guide management. Specifically, the statement indicates that the best approach for these patients is a policy of “required reconsideration” of the existing DNR order. Required reconsideration means that “the patient or designated surrogate and the physicians who will be responsible for the patient’s care should, when possible, discuss the new intraoperative and perioperative risks associated with the surgical procedure, the patient’s treatment goals, and an approach for potentially life-threatening problems consistent with the patient’s values and preferences.” Moreover, the required reconsideration discussion needs to occur as early as it is practical once a decision is made to have surgery because the discussion “may result in the patient agreeing to suspend the DNR order during surgery and the perioperative period, retaining the original DNR order, or modifying the DNR order.” Given that surgical patients with DNR orders have significant comorbidities, many sustain postoperative complications, and nearly 1 in 4 die within 30 days of surgery, preoperative advance care planning (ACP) and code status discussions are particularly essential to delivering high quality surgical care.2
In the current study, Hadler et al3 conducted a retrospective analysis to evaluate orders and documentation describing perioperative management of code status in patients with existing DNR order at an academic health system in the US. The authors reported that fewer than 20% of patients with existing DNR orders had a documented discussion of code status prior to undergoing surgery. These findings add to the notion that compliance with such guidance on required reconsideration discussion is suboptimal in perioperative care in the US.4,5 A recently published study focused on patients aged more than 60 years undergoing high-risk oncologic or vascular surgeries similarly showed that the frequency of ACP discussions or advance directive documentations among older patients was low.6 This growing body of evidence is highly clinically relevant in that preoperative discussion on code status is highly relevant to the care of older adults, a population group that accounts for the majority of surgeries and is most vulnerable to poor surgical outcomes. Additionally, it highlights a disconnect between the shared recognition by surgeons and patients that ACP discussion is important in perioperative care and its low implementation rates.
Unsurprisingly, Hadler et al3 reported that added measures such as the provision of a procedure-specific DNR form led to an increase in the documentation of preoperative code status discussions in 1 of the hospitals studied. The authors suggested that strategies such as the development of institutional protocols aimed to facilitate perioperative advance directive discussions, identify local champions, and educate patients may be ways to improve preoperative code status reevaulation. The idea that institutional value and culture are key factors impacting surgeon behavior and may influence the practice of ACP discussion is not new. Thus, creative and adaptable strategies, resources, and trainings that are required by medical institutions and hospitals to support preoperative ACP discussions with patients undergoing surgeries need to be identified, validated, and implemented to optimize perioperative care in vulnerable patients.
Applications for Clinical Practice
The findings from the current study indicate that less than 20% of patients with preexisting DNR orders have a documented discussion of code status prior to undergoing surgery. Physicians and health care institutions need to identify barriers to, and implement strategies that, facilitate and optimize preoperative ACP discussions in order to provide patient-centered care in vulnerable surgical patients.
Financial disclosures: None.
1. American College of Surgeons Board of Regents. Statement on Advance Directives by Patients: “Do Not Resuscitate” in the Operating Room. American College of Surgeons. January 3, 2014. Accessed November 6, 2021. https://www.facs.org/about-acs/statements/19-advance-directives
2. Kazaure H, Roman S, Sosa JA. High mortality in surgical patients with do-not-resuscitate orders: analysis of 8256 patients. Arch Surg. 2011;146(8):922-928. doi:10.1001/archsurg.2011.69
3. Hadler RA, Fatuzzo M, Sahota G, Neuman MD. Perioperative Management of Do-Not-Resuscitate Orders at a Large Academic Health System. JAMA Surg. 2021;e214135. doi:10.1001/jamasurg.2021.4135
4. Coopmans VC, Gries CA. CRNA awareness and experience with perioperative DNR orders. AANA J. 2000;68(3):247-256.
5. Urman RD, Lilley EJ, Changala M, Lindvall C, Hepner DL, Bader AM. A Pilot Study to Evaluate Compliance with Guidelines for Preprocedural Reconsideration of Code Status Limitations. J Palliat Med. 2018;21(8):1152-1156. doi:10.1089/jpm.2017.0601
6. Kalbfell E, Kata A, Buffington AS, et al. Frequency of Preoperative Advance Care Planning for Older Adults Undergoing High-risk Surgery: A Secondary Analysis of a Randomized Clinical Trial. JAMA Surg. 2021;156(7):e211521. doi:10.1001/jamasurg.2021.1521
Study Overview
Objective. The objective of this study was to evaluate orders and documentation describing perioperative management of code status in adults.
Design. A retrospective case series of all adult inpatients admitted to hospitals at 1 academic health system in the US.
Setting and participants. This retrospective case series was conducted at 5 hospitals within the University of Pennsylvania Health System. Cases included all adult inpatients admitted to hospitals between March 2017 and September 2018 who had a Do-Not-Resuscitate (DNR) order placed in their medical record during admission and subsequently underwent a surgical procedure that required anesthesia care.
Main outcome measures. Medical records of included cases were manually reviewed by the authors to verify whether a DNR order was in place at the time surgical intervention was discussed with a patient. Clinical notes and DNR orders of eligible cases were reviewed to identify documentation and outcome of goals of care discussions that were conducted within 48 hours prior to the surgical procedure. Collected data included patient demographics (age, sex, race); case characteristics (American Society of Anesthesiologists [ASA] physical status score, anesthesia type [general vs others such as regional], emergency status [emergent vs elective surgery], procedures by service [surgical including hip fracture repair, gastrostomy or jejunostomy, or exploratory laparotomy vs medical including endoscopy, bronchoscopy, or transesophageal echocardiogram]); and hospital policy for perioperative management of DNR orders (written policy encouraging discussion vs written policy plus additional initiatives, including procedure-specific DNR form). The primary outcome was the presence of a preoperative order or note documenting code status discussion or change. Data were analyzed using χ2 and Fisher exact tests and the threshold for statistical significance was P < .05.
Main results. Of the 27 665 inpatient procedures identified across 5 hospitals, 444 (1.6%) cases met the inclusion criteria. Patients from these cases aged 75 (SD 13) years (95% CI, 72-77 years); 247 (56%, 95% CI, 55%-57%) were women; and 300 (68%, 95% CI, 65%-71%) were White. A total of 426 patients (96%, 95% CI, 90%-100%) had an ASA physical status score of 3 or higher and 237 (53%, 95% CI, 51%-56%) received general anesthesia. The most common procedures performed were endoscopy (148 [33%]), hip fracture repair (43 [10%]), and gastrostomy or jejunostomy (28 [6%]). Reevaluation of code status was documented in 126 cases (28%, 95% CI, 25%-31%); code status orders were changed in 20 of 126 cases (16%, 95% CI, 7%-24%); and a note was filed without a corresponding order for 106 of 126 cases (84%, 95% CI, 75%-95%). In the majority of cases (109 of 126 [87%], 95% CI, 78%-95%) in which documented discussion occurred, DNR orders were suspended. Of 126 cases in which a discussion was documented, participants of these discussions included surgeons 10% of the time (13 cases, 95% CI, 8%-13%), members of the anesthesia team 51% of the time (64 cases, 95% CI, 49%-53%), and medicine or palliative care clinicians 39% of the time (49 cases, 95% CI, 37%-41%).
The rate of documented preoperative code status discussion was higher in patients with higher ASA physical status score (35% in patients with an ASA physical status score ≥ 4 [55 of 155] vs 25% in those with an ASA physical status score ≤ 3 [71 of 289]; P = .02). The rates of documented preoperative code status discussion were similar by anesthesia type (29% for general anesthesia [69 of 237 cases] vs 28% [57 of 207 cases] for other modalities; P = .70). The hospitals involved in this study all had a written policy encouraging rediscussion of code status before surgery. However, only 1 hospital reported added measures (eg, provision of a procedure-specific DNR form) to increase documentation of preoperative code status discussions. In this specific hospital, documentation of preoperative code status discussions was higher compared to other hospitals (67% [37 of 55 cases] vs 23% [89 of 389 cases]; P < .01).
Conclusion. In a retrospective case series conducted at 5 hospitals within 1 academic health system in the US, fewer than 1 in 5 patients with preexisting DNR orders had a documented discussion of code status prior to undergoing surgery. Additional strategies including the development of institutional protocols that facilitate perioperative management of advance directives, identification of local champions, and patient education, should be explored as means to improve preoperative code status reevaulation per guideline recommendations.
Commentary
It is not unusual that patients with a DNR order may require and undergo surgical interventions to treat reversible conditions, prevent progression of underlying disease, or mitigate distressing symptoms such as pain. For instance, intubation, mechanical ventilation, and administration of vasoactive drugs are resuscitative measures that may be needed to safely anesthetize and sedate a patient. As such, the American College of Surgeons1 has provided a statement on advance directives by patients with an existing DNR order to guide management. Specifically, the statement indicates that the best approach for these patients is a policy of “required reconsideration” of the existing DNR order. Required reconsideration means that “the patient or designated surrogate and the physicians who will be responsible for the patient’s care should, when possible, discuss the new intraoperative and perioperative risks associated with the surgical procedure, the patient’s treatment goals, and an approach for potentially life-threatening problems consistent with the patient’s values and preferences.” Moreover, the required reconsideration discussion needs to occur as early as it is practical once a decision is made to have surgery because the discussion “may result in the patient agreeing to suspend the DNR order during surgery and the perioperative period, retaining the original DNR order, or modifying the DNR order.” Given that surgical patients with DNR orders have significant comorbidities, many sustain postoperative complications, and nearly 1 in 4 die within 30 days of surgery, preoperative advance care planning (ACP) and code status discussions are particularly essential to delivering high quality surgical care.2
In the current study, Hadler et al3 conducted a retrospective analysis to evaluate orders and documentation describing perioperative management of code status in patients with existing DNR order at an academic health system in the US. The authors reported that fewer than 20% of patients with existing DNR orders had a documented discussion of code status prior to undergoing surgery. These findings add to the notion that compliance with such guidance on required reconsideration discussion is suboptimal in perioperative care in the US.4,5 A recently published study focused on patients aged more than 60 years undergoing high-risk oncologic or vascular surgeries similarly showed that the frequency of ACP discussions or advance directive documentations among older patients was low.6 This growing body of evidence is highly clinically relevant in that preoperative discussion on code status is highly relevant to the care of older adults, a population group that accounts for the majority of surgeries and is most vulnerable to poor surgical outcomes. Additionally, it highlights a disconnect between the shared recognition by surgeons and patients that ACP discussion is important in perioperative care and its low implementation rates.
Unsurprisingly, Hadler et al3 reported that added measures such as the provision of a procedure-specific DNR form led to an increase in the documentation of preoperative code status discussions in 1 of the hospitals studied. The authors suggested that strategies such as the development of institutional protocols aimed to facilitate perioperative advance directive discussions, identify local champions, and educate patients may be ways to improve preoperative code status reevaulation. The idea that institutional value and culture are key factors impacting surgeon behavior and may influence the practice of ACP discussion is not new. Thus, creative and adaptable strategies, resources, and trainings that are required by medical institutions and hospitals to support preoperative ACP discussions with patients undergoing surgeries need to be identified, validated, and implemented to optimize perioperative care in vulnerable patients.
Applications for Clinical Practice
The findings from the current study indicate that less than 20% of patients with preexisting DNR orders have a documented discussion of code status prior to undergoing surgery. Physicians and health care institutions need to identify barriers to, and implement strategies that, facilitate and optimize preoperative ACP discussions in order to provide patient-centered care in vulnerable surgical patients.
Financial disclosures: None.
Study Overview
Objective. The objective of this study was to evaluate orders and documentation describing perioperative management of code status in adults.
Design. A retrospective case series of all adult inpatients admitted to hospitals at 1 academic health system in the US.
Setting and participants. This retrospective case series was conducted at 5 hospitals within the University of Pennsylvania Health System. Cases included all adult inpatients admitted to hospitals between March 2017 and September 2018 who had a Do-Not-Resuscitate (DNR) order placed in their medical record during admission and subsequently underwent a surgical procedure that required anesthesia care.
Main outcome measures. Medical records of included cases were manually reviewed by the authors to verify whether a DNR order was in place at the time surgical intervention was discussed with a patient. Clinical notes and DNR orders of eligible cases were reviewed to identify documentation and outcome of goals of care discussions that were conducted within 48 hours prior to the surgical procedure. Collected data included patient demographics (age, sex, race); case characteristics (American Society of Anesthesiologists [ASA] physical status score, anesthesia type [general vs others such as regional], emergency status [emergent vs elective surgery], procedures by service [surgical including hip fracture repair, gastrostomy or jejunostomy, or exploratory laparotomy vs medical including endoscopy, bronchoscopy, or transesophageal echocardiogram]); and hospital policy for perioperative management of DNR orders (written policy encouraging discussion vs written policy plus additional initiatives, including procedure-specific DNR form). The primary outcome was the presence of a preoperative order or note documenting code status discussion or change. Data were analyzed using χ2 and Fisher exact tests and the threshold for statistical significance was P < .05.
Main results. Of the 27 665 inpatient procedures identified across 5 hospitals, 444 (1.6%) cases met the inclusion criteria. Patients from these cases aged 75 (SD 13) years (95% CI, 72-77 years); 247 (56%, 95% CI, 55%-57%) were women; and 300 (68%, 95% CI, 65%-71%) were White. A total of 426 patients (96%, 95% CI, 90%-100%) had an ASA physical status score of 3 or higher and 237 (53%, 95% CI, 51%-56%) received general anesthesia. The most common procedures performed were endoscopy (148 [33%]), hip fracture repair (43 [10%]), and gastrostomy or jejunostomy (28 [6%]). Reevaluation of code status was documented in 126 cases (28%, 95% CI, 25%-31%); code status orders were changed in 20 of 126 cases (16%, 95% CI, 7%-24%); and a note was filed without a corresponding order for 106 of 126 cases (84%, 95% CI, 75%-95%). In the majority of cases (109 of 126 [87%], 95% CI, 78%-95%) in which documented discussion occurred, DNR orders were suspended. Of 126 cases in which a discussion was documented, participants of these discussions included surgeons 10% of the time (13 cases, 95% CI, 8%-13%), members of the anesthesia team 51% of the time (64 cases, 95% CI, 49%-53%), and medicine or palliative care clinicians 39% of the time (49 cases, 95% CI, 37%-41%).
The rate of documented preoperative code status discussion was higher in patients with higher ASA physical status score (35% in patients with an ASA physical status score ≥ 4 [55 of 155] vs 25% in those with an ASA physical status score ≤ 3 [71 of 289]; P = .02). The rates of documented preoperative code status discussion were similar by anesthesia type (29% for general anesthesia [69 of 237 cases] vs 28% [57 of 207 cases] for other modalities; P = .70). The hospitals involved in this study all had a written policy encouraging rediscussion of code status before surgery. However, only 1 hospital reported added measures (eg, provision of a procedure-specific DNR form) to increase documentation of preoperative code status discussions. In this specific hospital, documentation of preoperative code status discussions was higher compared to other hospitals (67% [37 of 55 cases] vs 23% [89 of 389 cases]; P < .01).
Conclusion. In a retrospective case series conducted at 5 hospitals within 1 academic health system in the US, fewer than 1 in 5 patients with preexisting DNR orders had a documented discussion of code status prior to undergoing surgery. Additional strategies including the development of institutional protocols that facilitate perioperative management of advance directives, identification of local champions, and patient education, should be explored as means to improve preoperative code status reevaulation per guideline recommendations.
Commentary
It is not unusual that patients with a DNR order may require and undergo surgical interventions to treat reversible conditions, prevent progression of underlying disease, or mitigate distressing symptoms such as pain. For instance, intubation, mechanical ventilation, and administration of vasoactive drugs are resuscitative measures that may be needed to safely anesthetize and sedate a patient. As such, the American College of Surgeons1 has provided a statement on advance directives by patients with an existing DNR order to guide management. Specifically, the statement indicates that the best approach for these patients is a policy of “required reconsideration” of the existing DNR order. Required reconsideration means that “the patient or designated surrogate and the physicians who will be responsible for the patient’s care should, when possible, discuss the new intraoperative and perioperative risks associated with the surgical procedure, the patient’s treatment goals, and an approach for potentially life-threatening problems consistent with the patient’s values and preferences.” Moreover, the required reconsideration discussion needs to occur as early as it is practical once a decision is made to have surgery because the discussion “may result in the patient agreeing to suspend the DNR order during surgery and the perioperative period, retaining the original DNR order, or modifying the DNR order.” Given that surgical patients with DNR orders have significant comorbidities, many sustain postoperative complications, and nearly 1 in 4 die within 30 days of surgery, preoperative advance care planning (ACP) and code status discussions are particularly essential to delivering high quality surgical care.2
In the current study, Hadler et al3 conducted a retrospective analysis to evaluate orders and documentation describing perioperative management of code status in patients with existing DNR order at an academic health system in the US. The authors reported that fewer than 20% of patients with existing DNR orders had a documented discussion of code status prior to undergoing surgery. These findings add to the notion that compliance with such guidance on required reconsideration discussion is suboptimal in perioperative care in the US.4,5 A recently published study focused on patients aged more than 60 years undergoing high-risk oncologic or vascular surgeries similarly showed that the frequency of ACP discussions or advance directive documentations among older patients was low.6 This growing body of evidence is highly clinically relevant in that preoperative discussion on code status is highly relevant to the care of older adults, a population group that accounts for the majority of surgeries and is most vulnerable to poor surgical outcomes. Additionally, it highlights a disconnect between the shared recognition by surgeons and patients that ACP discussion is important in perioperative care and its low implementation rates.
Unsurprisingly, Hadler et al3 reported that added measures such as the provision of a procedure-specific DNR form led to an increase in the documentation of preoperative code status discussions in 1 of the hospitals studied. The authors suggested that strategies such as the development of institutional protocols aimed to facilitate perioperative advance directive discussions, identify local champions, and educate patients may be ways to improve preoperative code status reevaulation. The idea that institutional value and culture are key factors impacting surgeon behavior and may influence the practice of ACP discussion is not new. Thus, creative and adaptable strategies, resources, and trainings that are required by medical institutions and hospitals to support preoperative ACP discussions with patients undergoing surgeries need to be identified, validated, and implemented to optimize perioperative care in vulnerable patients.
Applications for Clinical Practice
The findings from the current study indicate that less than 20% of patients with preexisting DNR orders have a documented discussion of code status prior to undergoing surgery. Physicians and health care institutions need to identify barriers to, and implement strategies that, facilitate and optimize preoperative ACP discussions in order to provide patient-centered care in vulnerable surgical patients.
Financial disclosures: None.
1. American College of Surgeons Board of Regents. Statement on Advance Directives by Patients: “Do Not Resuscitate” in the Operating Room. American College of Surgeons. January 3, 2014. Accessed November 6, 2021. https://www.facs.org/about-acs/statements/19-advance-directives
2. Kazaure H, Roman S, Sosa JA. High mortality in surgical patients with do-not-resuscitate orders: analysis of 8256 patients. Arch Surg. 2011;146(8):922-928. doi:10.1001/archsurg.2011.69
3. Hadler RA, Fatuzzo M, Sahota G, Neuman MD. Perioperative Management of Do-Not-Resuscitate Orders at a Large Academic Health System. JAMA Surg. 2021;e214135. doi:10.1001/jamasurg.2021.4135
4. Coopmans VC, Gries CA. CRNA awareness and experience with perioperative DNR orders. AANA J. 2000;68(3):247-256.
5. Urman RD, Lilley EJ, Changala M, Lindvall C, Hepner DL, Bader AM. A Pilot Study to Evaluate Compliance with Guidelines for Preprocedural Reconsideration of Code Status Limitations. J Palliat Med. 2018;21(8):1152-1156. doi:10.1089/jpm.2017.0601
6. Kalbfell E, Kata A, Buffington AS, et al. Frequency of Preoperative Advance Care Planning for Older Adults Undergoing High-risk Surgery: A Secondary Analysis of a Randomized Clinical Trial. JAMA Surg. 2021;156(7):e211521. doi:10.1001/jamasurg.2021.1521
1. American College of Surgeons Board of Regents. Statement on Advance Directives by Patients: “Do Not Resuscitate” in the Operating Room. American College of Surgeons. January 3, 2014. Accessed November 6, 2021. https://www.facs.org/about-acs/statements/19-advance-directives
2. Kazaure H, Roman S, Sosa JA. High mortality in surgical patients with do-not-resuscitate orders: analysis of 8256 patients. Arch Surg. 2011;146(8):922-928. doi:10.1001/archsurg.2011.69
3. Hadler RA, Fatuzzo M, Sahota G, Neuman MD. Perioperative Management of Do-Not-Resuscitate Orders at a Large Academic Health System. JAMA Surg. 2021;e214135. doi:10.1001/jamasurg.2021.4135
4. Coopmans VC, Gries CA. CRNA awareness and experience with perioperative DNR orders. AANA J. 2000;68(3):247-256.
5. Urman RD, Lilley EJ, Changala M, Lindvall C, Hepner DL, Bader AM. A Pilot Study to Evaluate Compliance with Guidelines for Preprocedural Reconsideration of Code Status Limitations. J Palliat Med. 2018;21(8):1152-1156. doi:10.1089/jpm.2017.0601
6. Kalbfell E, Kata A, Buffington AS, et al. Frequency of Preoperative Advance Care Planning for Older Adults Undergoing High-risk Surgery: A Secondary Analysis of a Randomized Clinical Trial. JAMA Surg. 2021;156(7):e211521. doi:10.1001/jamasurg.2021.1521
Positive Outcomes Following a Multidisciplinary Approach in the Diagnosis and Prevention of Hospital Delirium
From the Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA (Drs. Ching, Darwish, Li, Wong, Simpson, and Funk), the Department of Anesthesia, Cedars-Sinai Medical Center, Los Angeles, CA (Keith Siegel), and the Department of Psychiatry, Cedars-Sinai Medical Center, Los Angeles, CA (Dr. Bamgbose).
Objectives: To reduce the incidence and duration of delirium among patients in a hospital ward through standardized delirium screening tools and nonpharmacologic interventions. To advance nursing-focused education on delirium-prevention strategies. To measure the efficacy of the interventions with the aim of reproducing best practices.
Background: Delirium is associated with poor patient outcomes but may be preventable in a significant percentage of hospitalized patients.
Methods: Following nursing-focused education to prevent delirium, we prospectively evaluated patient care outcomes in a consecutive series of patients who were admitted to a hospital medical-surgical ward within a 25-week period. All patients who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria (N = 353). Standards for Quality Improvement Reporting Excellence guidelines were adhered to.
Results: There were 187 patients in the control group, and 166 in the postintervention group. Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days), mean length of stay (8.5 days vs 5.9 days), and use of an indwelling urinary catheter (9.1% vs 2.4%).
Conclusion: A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs.
Delirium is a disorder characterized by inattention and acute changes in cognition. It is defined by the American Psychiatric Association’s fifth edition of the Diagnostic and Statistical Manual of Mental Disorders as a disturbance in attention, awareness, and cognition over hours to a few days that is not better explained by a preexisting, established, or other evolving neurocognitive disorder.1 Delirium is common yet often under-recognized among hospitalized patients, particularly in the elderly. The incidence of delirium in elderly patients on admission is estimated to be 11% to 25%, and an additional 29% to 31% of elderly patients will develop delirium during the hospitalization.2 Delirium costs the health care system an estimated $38 billion to $152 billion per year.3 It is associated with negative outcomes, such as increased new placements to nursing homes, increased mortality, increased risk of dementia, and further cognitive deterioration among patients with dementia.4-6
Despite its prevalence, delirium may be preventable in a significant percentage of hospitalized patients. Targeted intervention strategies, such as frequent reorientation, maximizing sleep, early mobilization, restricting use of psychoactive medications, and addressing hearing or vision impairment, have been demonstrated to significantly reduce the incidence of hospital delirium.7,8 To achieve these goals, we explored the use of a multimodal strategy centered on nursing education. We integrated consistent, standardized delirium screening and nonpharmacologic interventions as part of a preventative protocol to reduce the incidence of delirium in the hospital ward.
Methods
We evaluated a consecutive series of patients who were admitted to a designated hospital medical-surgical ward within a 25-week period between October 2019 and April 2020. All patients during this period who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria. Patients who did not have a CAM documented were excluded from the analysis. Delirium was defined according to the CAM diagnostic algorithm.9
Core nursing staff regularly assigned to the ward completed a multimodal training program designed to improve recognition, documentation, and prevention of hospital delirium. Prior to the training, the nurses completed a 5-point Likert scale survey assessing their level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium. Nurses completed the same survey after the study period ended.
The training curriculum for nurses began with an online module reviewing the epidemiology and risk factors for delirium. Nurses then participated in a series of in-service training sessions led by a team of physicians, during which the CAM and nonpharmacologic delirium prevention measures were reviewed then practiced first-hand. Nursing staff attended an in-person lecture reviewing the current body of literature on delirium risk factors and effective nursing interventions. After formal training was completed, nurses were instructed to document CAM screens
Patients admitted to the hospital unit from the start of the training program (week 1) until the order set was made available (week 15) constituted our control group. The postintervention study group consisted of patients admitted for 10 weeks after the completion of the interventions (weeks 16-25). A timeline of the study events is shown in Figure 1.
Patient demographics and hospital-stay metrics determined a priori were attained via the Cedars-Sinai Enterprise Information Services core. Age, sex, medical history, and incidence of surgery with anesthesia during hospitalization were recorded. The Charlson Comorbidity Index was calculated from patients’ listed diagnoses following discharge. Primary outcomes included incidence of patients with delirium during hospitalization, percentage of tested shifts with positive CAM screens, length of hospital stay, and survival. Secondary outcomes included measures associated with delirium, including the use of chemical restraints, physical restraints, sitters, indwelling urinary catheters, and new psychiatry and neurology consults. Chemical restraints were defined as administration of a new antipsychotic medication or benzodiazepine for the specific indication of hyperactive delirium or agitation.
Statistical analysis was conducted by a statistician, using R version 3.6.3.10P values of < .05 were considered significant. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed with Welch’s t-test or, for highly skewed continuous variables, with Wilcoxon rank-sum test or Mood’s median test. All patient data were anonymized and stored securely in accordance with institutional guidelines.
Our project was deemed to represent nonhuman subject research and therefore did not require Institutional Review Board (IRB) approval upon review by our institution’s IRB committee and Office of Research Compliance and Quality Improvement. Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) guidelines were adhered to (Supplementary File can be found at mdedge.com/jcomjournal).
Results
We evaluated 353 patients who met our inclusion criteria: 187 in the control group, and 166 in the postintervention group. Ten patients were readmitted to the ward after their initial discharge; only the initial admission encounters were included in our analysis. Median age, sex, median Charlson Comorbidity Index, and incidence of surgery with anesthesia during hospitalization were comparable between the control and postintervention groups and are summarized in Table 2.
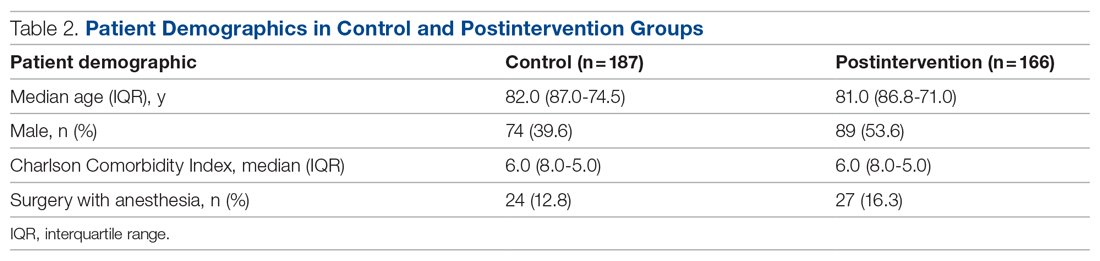
In the control group, 1572 CAMs were performed, with 74 positive CAMs recorded among 27 patients with delirium. In the postintervention group, 1298 CAMs were performed, with 12 positive CAMs recorded among 7 patients with delirium (Figure 2). Primary and secondary outcomes, as well as CAM compliance measures, are summarized in Table 3.
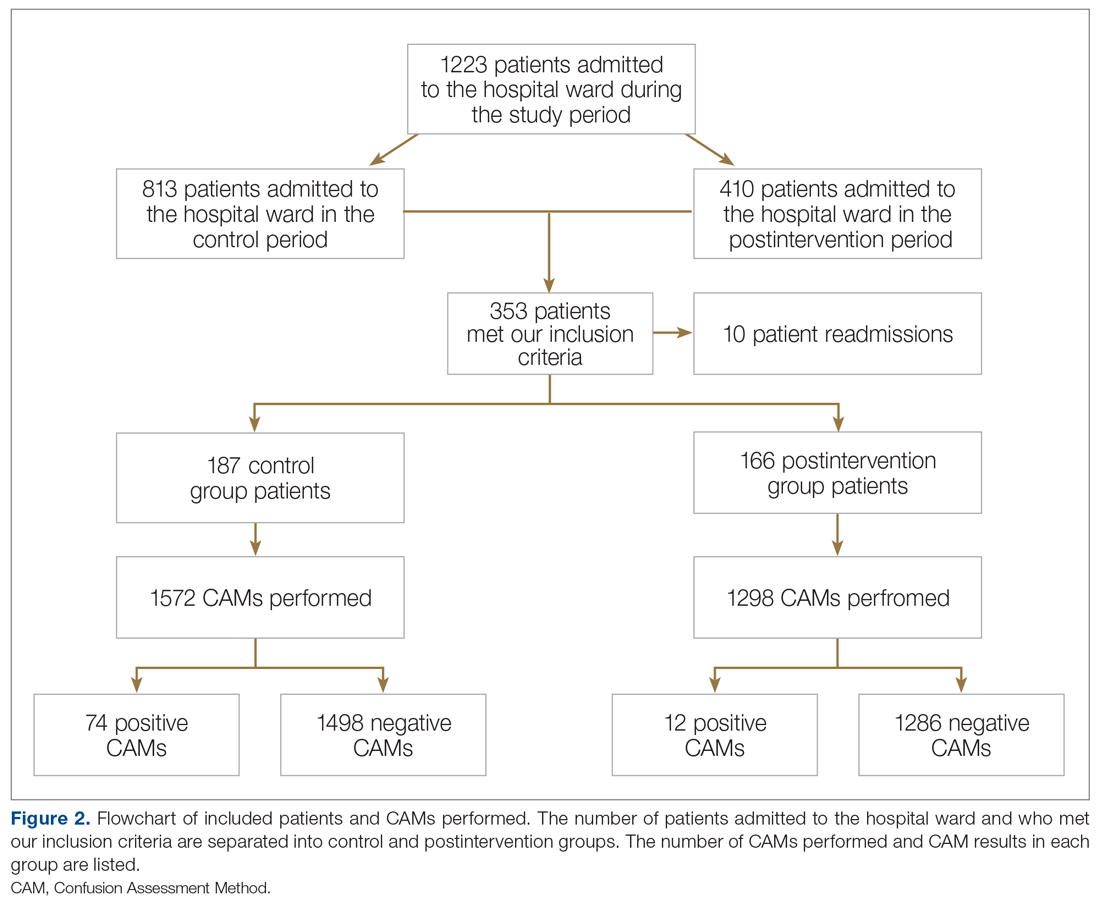
Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%, P = .002) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%, P = .002). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days, P = .004), mean length of stay (8.5 days vs 5.9 days, P = .003), and use of an indwelling urinary catheter (9.1% vs 2.4%, P = .012). There was a trend towards decreased incidence of chemical restraints and psychiatry consults, which did not reach statistical significance. Differences in mortality during hospitalization, physical restraint use, and sitter use could not be assessed due to low incidence.
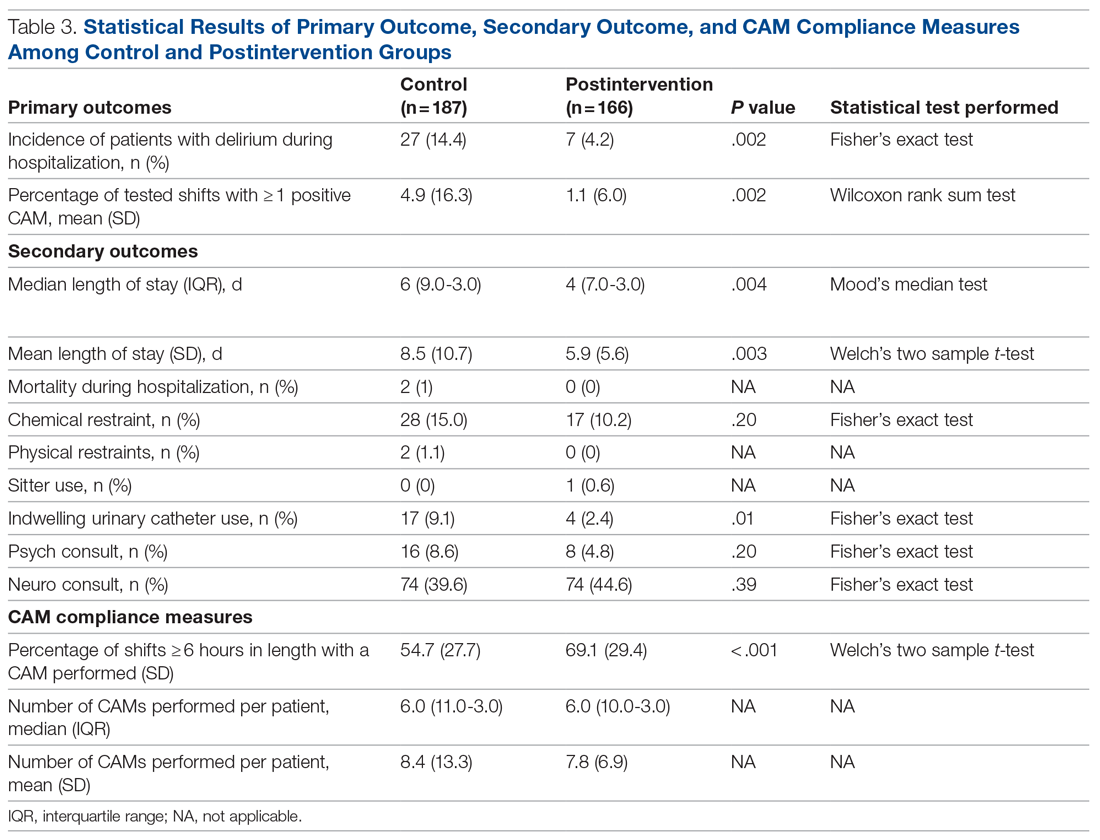
Compliance with nursing CAM assessments was evaluated. Compared to the control group, the postintervention group saw a significant increase in the percentage of shifts with a CAM performed (54.7% vs 69.1%, P < .001). The median and mean number of CAMs performed per patient were similar between the control and postintervention groups.
Results of nursing surveys completed before and after the training program are listed in Table 4. After training, nurses had a greater level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium.
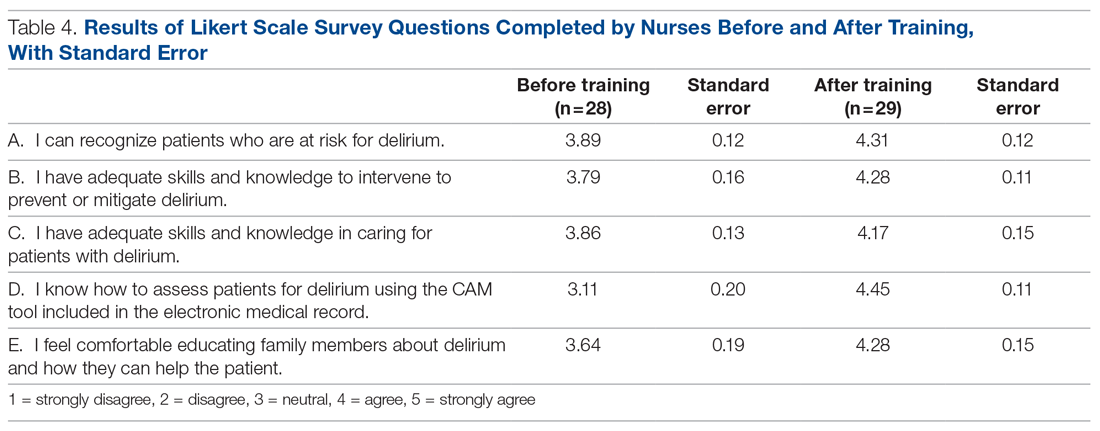
Discussion
Our study utilized a standardized delirium assessment tool to compare patient cohorts before and after nurse-targeted training interventions on delirium recognition and prevention. Our interventions emphasized nonpharmacologic intervention strategies, which are recommended as first-line in the management of patients with delirium.11 Patients were not excluded from the analysis based on preexisting medical conditions or recent surgery with anesthesia, to allow for conditions that are representative of community hospitals. We also did not use an inclusion criterion based on age; however, the majority of our patients were greater than 70 years old, representing those at highest risk for delirium.2 Significant outcomes among patients in the postintervention group include decreased incidence of delirium, lower average length of stay, decreased indwelling urinary catheter use, and increased compliance with delirium screening by nursing staff.
While the study’s focus was primarily on delirium prevention rather than treatment, these strategies may also have conferred the benefit of reversing delirium symptoms. In addition to measuring incidence of delirium, our primary outcome of percentage of tested shifts with 1 or more positive CAM was intended to assess the overall duration in which patients had delirium during their hospitalization. The reduction in shifts with positive CAMs observed in the postintervention group is notable, given that a significant percentage of patients with hospital delirium have the potential for symptom reversibility.12
Multiple studies have shown that admitted patients who develop delirium experience prolonged hospital stays, often up to 5 to 10 days longer.12-14 The decreased incidence and duration of delirium in our postintervention group is a reasonable explanation for the observed decrease in average length of stay. Our study is in line with previously documented initiatives that show that nonpharmacologic interventions can effectively address downstream health and fiscal sequelae of hospital delirium. For example, a volunteer-based initiative named the Hospital Elder Life Program, from which elements in our order set were modeled after, demonstrated significant reductions in delirium incidence, length of stay, and health care costs.14-16 Other initiatives that focused on educational training for nurses to assess and prevent delirium have also demonstrated similar positive results.17-19 Our study provides a model for effective nursing-focused education that can be reproduced in the hospital setting.
Unlike some other studies, which identified delirium based only on physician assessments, our initiative utilized the CAM performed by floor nurses to identify delirium. While this method
Our study demonstrated an increase in the overall compliance with the CAM screening during the postintervention period, which is significant given the under-recognition of delirium by health care professionals.20 We attribute this increase to greater realized importance and a higher level of confidence from nursing staff in recognizing and addressing delirium, as supported by survey data. While the increased screening of patients should be considered a positive outcome, it also poses the possibility that the observed decrease in delirium incidence in the postintervention group was in fact due to more CAMs performed on patients without delirium. Likewise, nurses may have become more adept at recognizing true delirium, as opposed to delirium mimics, in the latter period of the study.
Perhaps the greatest limitation of our study is the variability in performing and recording CAMs, as some patients had multiple CAMs recorded while others did not have any CAMs recorded. This may have been affected in part by the increase in COVID-19 cases in our hospital towards the latter half of the study, which resulted in changes in nursing assignments as well as patient comorbidities in ways that cannot be easily quantified. Given the limited size of our patient cohorts, certain outcomes, such as the use of sitters, physical restraints, and in-hospital mortality, were unable to be assessed for changes statistically. Causative relationships between our interventions and associated outcome measures are necessarily limited in a binary comparison between control and postintervention groups.
Within these limitations, our study demonstrates promising results in core dimensions of patient care. We anticipate further quality improvement initiatives involving greater numbers of nursing staff and patients to better quantify the impact of nonpharmacologic nursing-centered interventions for preventing hospital delirium.
Conclusion
A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs. Education and tools to equip nurses to perform standardized delirium screening and interventions should be prioritized.
Acknowledgment: The authors thanks Olena Svetlov, NP, Oscar Abarca, Jose Chavez, and Jenita Gutierrez.
Corresponding author: Jason Ching, MD, Department of Neurology, Cedars-Sinai Medical Center, 8700 Beverly Blvd, Los Angeles, CA 90048; jason.ching@cshs.org.
Financial disclosures: None.
Funding: This research was supported by NIH National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR001881.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th edition. American Psychiatric Association; 2013.
2. Vasilevskis EE, Han JH, Hughes CG, et al. Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol. 2012;26(3):277-287. doi:10.1016/j.bpa.2012.07003
3. Leslie DL, Marcantonio ER, Zhang Y, et al. One-year health care costs associated with delirium in the elderly population. Arch Intern Med. 2008;168(1):27-32. doi:10.1001/archinternmed.2007.4
4. McCusker J, Cole M, Abrahamowicz M, et al. Delirium predicts 12-month mortality. Arch Intern Med. 2002;162(4):457-463. doi:10.1001/archinte.162.4.457
5. Witlox J, Eurelings LS, de Jonghe JF, et al. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304(4):443-451. doi:10.1001/jama.2010.1013
6. Gross AL, Jones RN, Habtemariam DA, et al. Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324-1331. doi:10.1001/archinternmed.2012.3203
7. Inouye SK. Prevention of delirium in hospitalized older patients: risk factors and targeted intervention strategies. Ann Med. 2000;32(4):257-263. doi:10.3109/07853890009011770
8. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. doi:10.7326/0003-4819-113-12-941
10. R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2017.
11. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220. doi:10.1038/nrneurol.2009.24
12. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364. doi:10.1093/ageing/afl005
13. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291(14):1753-1762. doi:10.1001/jama.291.14.1753
14. Chen CC, Lin MT, Tien YW, et al. Modified Hospital Elder Life Program: effects on abdominal surgery patients. J Am Coll Surg. 2011;213(2):245-252. doi:10.1016/j.jamcollsurg.2011.05.004
15. Zaubler TS, Murphy K, Rizzuto L, et al. Quality improvement and cost savings with multicomponent delirium interventions: replication of the Hospital Elder Life Program in a community hospital. Psychosomatics. 2013;54(3):219-226. doi:10.1016/j.psym.2013.01.010
16. Rubin FH, Neal K, Fenlon K, et al. Sustainability and scalability of the Hospital Elder Life Program at a community hospital. J Am Geriatr Soc. 2011;59(2):359-365. doi:10.1111/j.1532-5415.2010.03243.x
17. Milisen K, Foreman MD, Abraham IL, et al. A nurse-led interdisciplinary intervention program for delirium in elderly hip-fracture patients. J Am Geriatr Soc. 2001;49(5):523-532. doi:10.1046/j.1532-5415.2001.49109.x
18. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628. doi:10.1111/j.1532-5415.2005.53210.x
19. Tabet N, Hudson S, Sweeney V, et al. An educational intervention can prevent delirium on acute medical wards. Age Ageing. 2005;34(2):152-156. doi:10.1093/ageing/afi0320. Han JH, Zimmerman EE, Cutler N, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009;16(3):193-200. doi:10.1111/j.1553-2712.2008.00339.x
From the Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA (Drs. Ching, Darwish, Li, Wong, Simpson, and Funk), the Department of Anesthesia, Cedars-Sinai Medical Center, Los Angeles, CA (Keith Siegel), and the Department of Psychiatry, Cedars-Sinai Medical Center, Los Angeles, CA (Dr. Bamgbose).
Objectives: To reduce the incidence and duration of delirium among patients in a hospital ward through standardized delirium screening tools and nonpharmacologic interventions. To advance nursing-focused education on delirium-prevention strategies. To measure the efficacy of the interventions with the aim of reproducing best practices.
Background: Delirium is associated with poor patient outcomes but may be preventable in a significant percentage of hospitalized patients.
Methods: Following nursing-focused education to prevent delirium, we prospectively evaluated patient care outcomes in a consecutive series of patients who were admitted to a hospital medical-surgical ward within a 25-week period. All patients who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria (N = 353). Standards for Quality Improvement Reporting Excellence guidelines were adhered to.
Results: There were 187 patients in the control group, and 166 in the postintervention group. Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days), mean length of stay (8.5 days vs 5.9 days), and use of an indwelling urinary catheter (9.1% vs 2.4%).
Conclusion: A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs.
Delirium is a disorder characterized by inattention and acute changes in cognition. It is defined by the American Psychiatric Association’s fifth edition of the Diagnostic and Statistical Manual of Mental Disorders as a disturbance in attention, awareness, and cognition over hours to a few days that is not better explained by a preexisting, established, or other evolving neurocognitive disorder.1 Delirium is common yet often under-recognized among hospitalized patients, particularly in the elderly. The incidence of delirium in elderly patients on admission is estimated to be 11% to 25%, and an additional 29% to 31% of elderly patients will develop delirium during the hospitalization.2 Delirium costs the health care system an estimated $38 billion to $152 billion per year.3 It is associated with negative outcomes, such as increased new placements to nursing homes, increased mortality, increased risk of dementia, and further cognitive deterioration among patients with dementia.4-6
Despite its prevalence, delirium may be preventable in a significant percentage of hospitalized patients. Targeted intervention strategies, such as frequent reorientation, maximizing sleep, early mobilization, restricting use of psychoactive medications, and addressing hearing or vision impairment, have been demonstrated to significantly reduce the incidence of hospital delirium.7,8 To achieve these goals, we explored the use of a multimodal strategy centered on nursing education. We integrated consistent, standardized delirium screening and nonpharmacologic interventions as part of a preventative protocol to reduce the incidence of delirium in the hospital ward.
Methods
We evaluated a consecutive series of patients who were admitted to a designated hospital medical-surgical ward within a 25-week period between October 2019 and April 2020. All patients during this period who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria. Patients who did not have a CAM documented were excluded from the analysis. Delirium was defined according to the CAM diagnostic algorithm.9
Core nursing staff regularly assigned to the ward completed a multimodal training program designed to improve recognition, documentation, and prevention of hospital delirium. Prior to the training, the nurses completed a 5-point Likert scale survey assessing their level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium. Nurses completed the same survey after the study period ended.
The training curriculum for nurses began with an online module reviewing the epidemiology and risk factors for delirium. Nurses then participated in a series of in-service training sessions led by a team of physicians, during which the CAM and nonpharmacologic delirium prevention measures were reviewed then practiced first-hand. Nursing staff attended an in-person lecture reviewing the current body of literature on delirium risk factors and effective nursing interventions. After formal training was completed, nurses were instructed to document CAM screens

Patients admitted to the hospital unit from the start of the training program (week 1) until the order set was made available (week 15) constituted our control group. The postintervention study group consisted of patients admitted for 10 weeks after the completion of the interventions (weeks 16-25). A timeline of the study events is shown in Figure 1.

Patient demographics and hospital-stay metrics determined a priori were attained via the Cedars-Sinai Enterprise Information Services core. Age, sex, medical history, and incidence of surgery with anesthesia during hospitalization were recorded. The Charlson Comorbidity Index was calculated from patients’ listed diagnoses following discharge. Primary outcomes included incidence of patients with delirium during hospitalization, percentage of tested shifts with positive CAM screens, length of hospital stay, and survival. Secondary outcomes included measures associated with delirium, including the use of chemical restraints, physical restraints, sitters, indwelling urinary catheters, and new psychiatry and neurology consults. Chemical restraints were defined as administration of a new antipsychotic medication or benzodiazepine for the specific indication of hyperactive delirium or agitation.
Statistical analysis was conducted by a statistician, using R version 3.6.3.10P values of < .05 were considered significant. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed with Welch’s t-test or, for highly skewed continuous variables, with Wilcoxon rank-sum test or Mood’s median test. All patient data were anonymized and stored securely in accordance with institutional guidelines.
Our project was deemed to represent nonhuman subject research and therefore did not require Institutional Review Board (IRB) approval upon review by our institution’s IRB committee and Office of Research Compliance and Quality Improvement. Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) guidelines were adhered to (Supplementary File can be found at mdedge.com/jcomjournal).
Results
We evaluated 353 patients who met our inclusion criteria: 187 in the control group, and 166 in the postintervention group. Ten patients were readmitted to the ward after their initial discharge; only the initial admission encounters were included in our analysis. Median age, sex, median Charlson Comorbidity Index, and incidence of surgery with anesthesia during hospitalization were comparable between the control and postintervention groups and are summarized in Table 2.

In the control group, 1572 CAMs were performed, with 74 positive CAMs recorded among 27 patients with delirium. In the postintervention group, 1298 CAMs were performed, with 12 positive CAMs recorded among 7 patients with delirium (Figure 2). Primary and secondary outcomes, as well as CAM compliance measures, are summarized in Table 3.

Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%, P = .002) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%, P = .002). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days, P = .004), mean length of stay (8.5 days vs 5.9 days, P = .003), and use of an indwelling urinary catheter (9.1% vs 2.4%, P = .012). There was a trend towards decreased incidence of chemical restraints and psychiatry consults, which did not reach statistical significance. Differences in mortality during hospitalization, physical restraint use, and sitter use could not be assessed due to low incidence.

Compliance with nursing CAM assessments was evaluated. Compared to the control group, the postintervention group saw a significant increase in the percentage of shifts with a CAM performed (54.7% vs 69.1%, P < .001). The median and mean number of CAMs performed per patient were similar between the control and postintervention groups.
Results of nursing surveys completed before and after the training program are listed in Table 4. After training, nurses had a greater level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium.

Discussion
Our study utilized a standardized delirium assessment tool to compare patient cohorts before and after nurse-targeted training interventions on delirium recognition and prevention. Our interventions emphasized nonpharmacologic intervention strategies, which are recommended as first-line in the management of patients with delirium.11 Patients were not excluded from the analysis based on preexisting medical conditions or recent surgery with anesthesia, to allow for conditions that are representative of community hospitals. We also did not use an inclusion criterion based on age; however, the majority of our patients were greater than 70 years old, representing those at highest risk for delirium.2 Significant outcomes among patients in the postintervention group include decreased incidence of delirium, lower average length of stay, decreased indwelling urinary catheter use, and increased compliance with delirium screening by nursing staff.
While the study’s focus was primarily on delirium prevention rather than treatment, these strategies may also have conferred the benefit of reversing delirium symptoms. In addition to measuring incidence of delirium, our primary outcome of percentage of tested shifts with 1 or more positive CAM was intended to assess the overall duration in which patients had delirium during their hospitalization. The reduction in shifts with positive CAMs observed in the postintervention group is notable, given that a significant percentage of patients with hospital delirium have the potential for symptom reversibility.12
Multiple studies have shown that admitted patients who develop delirium experience prolonged hospital stays, often up to 5 to 10 days longer.12-14 The decreased incidence and duration of delirium in our postintervention group is a reasonable explanation for the observed decrease in average length of stay. Our study is in line with previously documented initiatives that show that nonpharmacologic interventions can effectively address downstream health and fiscal sequelae of hospital delirium. For example, a volunteer-based initiative named the Hospital Elder Life Program, from which elements in our order set were modeled after, demonstrated significant reductions in delirium incidence, length of stay, and health care costs.14-16 Other initiatives that focused on educational training for nurses to assess and prevent delirium have also demonstrated similar positive results.17-19 Our study provides a model for effective nursing-focused education that can be reproduced in the hospital setting.
Unlike some other studies, which identified delirium based only on physician assessments, our initiative utilized the CAM performed by floor nurses to identify delirium. While this method
Our study demonstrated an increase in the overall compliance with the CAM screening during the postintervention period, which is significant given the under-recognition of delirium by health care professionals.20 We attribute this increase to greater realized importance and a higher level of confidence from nursing staff in recognizing and addressing delirium, as supported by survey data. While the increased screening of patients should be considered a positive outcome, it also poses the possibility that the observed decrease in delirium incidence in the postintervention group was in fact due to more CAMs performed on patients without delirium. Likewise, nurses may have become more adept at recognizing true delirium, as opposed to delirium mimics, in the latter period of the study.
Perhaps the greatest limitation of our study is the variability in performing and recording CAMs, as some patients had multiple CAMs recorded while others did not have any CAMs recorded. This may have been affected in part by the increase in COVID-19 cases in our hospital towards the latter half of the study, which resulted in changes in nursing assignments as well as patient comorbidities in ways that cannot be easily quantified. Given the limited size of our patient cohorts, certain outcomes, such as the use of sitters, physical restraints, and in-hospital mortality, were unable to be assessed for changes statistically. Causative relationships between our interventions and associated outcome measures are necessarily limited in a binary comparison between control and postintervention groups.
Within these limitations, our study demonstrates promising results in core dimensions of patient care. We anticipate further quality improvement initiatives involving greater numbers of nursing staff and patients to better quantify the impact of nonpharmacologic nursing-centered interventions for preventing hospital delirium.
Conclusion
A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs. Education and tools to equip nurses to perform standardized delirium screening and interventions should be prioritized.
Acknowledgment: The authors thanks Olena Svetlov, NP, Oscar Abarca, Jose Chavez, and Jenita Gutierrez.
Corresponding author: Jason Ching, MD, Department of Neurology, Cedars-Sinai Medical Center, 8700 Beverly Blvd, Los Angeles, CA 90048; jason.ching@cshs.org.
Financial disclosures: None.
Funding: This research was supported by NIH National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR001881.
From the Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA (Drs. Ching, Darwish, Li, Wong, Simpson, and Funk), the Department of Anesthesia, Cedars-Sinai Medical Center, Los Angeles, CA (Keith Siegel), and the Department of Psychiatry, Cedars-Sinai Medical Center, Los Angeles, CA (Dr. Bamgbose).
Objectives: To reduce the incidence and duration of delirium among patients in a hospital ward through standardized delirium screening tools and nonpharmacologic interventions. To advance nursing-focused education on delirium-prevention strategies. To measure the efficacy of the interventions with the aim of reproducing best practices.
Background: Delirium is associated with poor patient outcomes but may be preventable in a significant percentage of hospitalized patients.
Methods: Following nursing-focused education to prevent delirium, we prospectively evaluated patient care outcomes in a consecutive series of patients who were admitted to a hospital medical-surgical ward within a 25-week period. All patients who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria (N = 353). Standards for Quality Improvement Reporting Excellence guidelines were adhered to.
Results: There were 187 patients in the control group, and 166 in the postintervention group. Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days), mean length of stay (8.5 days vs 5.9 days), and use of an indwelling urinary catheter (9.1% vs 2.4%).
Conclusion: A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs.
Delirium is a disorder characterized by inattention and acute changes in cognition. It is defined by the American Psychiatric Association’s fifth edition of the Diagnostic and Statistical Manual of Mental Disorders as a disturbance in attention, awareness, and cognition over hours to a few days that is not better explained by a preexisting, established, or other evolving neurocognitive disorder.1 Delirium is common yet often under-recognized among hospitalized patients, particularly in the elderly. The incidence of delirium in elderly patients on admission is estimated to be 11% to 25%, and an additional 29% to 31% of elderly patients will develop delirium during the hospitalization.2 Delirium costs the health care system an estimated $38 billion to $152 billion per year.3 It is associated with negative outcomes, such as increased new placements to nursing homes, increased mortality, increased risk of dementia, and further cognitive deterioration among patients with dementia.4-6
Despite its prevalence, delirium may be preventable in a significant percentage of hospitalized patients. Targeted intervention strategies, such as frequent reorientation, maximizing sleep, early mobilization, restricting use of psychoactive medications, and addressing hearing or vision impairment, have been demonstrated to significantly reduce the incidence of hospital delirium.7,8 To achieve these goals, we explored the use of a multimodal strategy centered on nursing education. We integrated consistent, standardized delirium screening and nonpharmacologic interventions as part of a preventative protocol to reduce the incidence of delirium in the hospital ward.
Methods
We evaluated a consecutive series of patients who were admitted to a designated hospital medical-surgical ward within a 25-week period between October 2019 and April 2020. All patients during this period who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria. Patients who did not have a CAM documented were excluded from the analysis. Delirium was defined according to the CAM diagnostic algorithm.9
Core nursing staff regularly assigned to the ward completed a multimodal training program designed to improve recognition, documentation, and prevention of hospital delirium. Prior to the training, the nurses completed a 5-point Likert scale survey assessing their level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium. Nurses completed the same survey after the study period ended.
The training curriculum for nurses began with an online module reviewing the epidemiology and risk factors for delirium. Nurses then participated in a series of in-service training sessions led by a team of physicians, during which the CAM and nonpharmacologic delirium prevention measures were reviewed then practiced first-hand. Nursing staff attended an in-person lecture reviewing the current body of literature on delirium risk factors and effective nursing interventions. After formal training was completed, nurses were instructed to document CAM screens

Patients admitted to the hospital unit from the start of the training program (week 1) until the order set was made available (week 15) constituted our control group. The postintervention study group consisted of patients admitted for 10 weeks after the completion of the interventions (weeks 16-25). A timeline of the study events is shown in Figure 1.

Patient demographics and hospital-stay metrics determined a priori were attained via the Cedars-Sinai Enterprise Information Services core. Age, sex, medical history, and incidence of surgery with anesthesia during hospitalization were recorded. The Charlson Comorbidity Index was calculated from patients’ listed diagnoses following discharge. Primary outcomes included incidence of patients with delirium during hospitalization, percentage of tested shifts with positive CAM screens, length of hospital stay, and survival. Secondary outcomes included measures associated with delirium, including the use of chemical restraints, physical restraints, sitters, indwelling urinary catheters, and new psychiatry and neurology consults. Chemical restraints were defined as administration of a new antipsychotic medication or benzodiazepine for the specific indication of hyperactive delirium or agitation.
Statistical analysis was conducted by a statistician, using R version 3.6.3.10P values of < .05 were considered significant. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed with Welch’s t-test or, for highly skewed continuous variables, with Wilcoxon rank-sum test or Mood’s median test. All patient data were anonymized and stored securely in accordance with institutional guidelines.
Our project was deemed to represent nonhuman subject research and therefore did not require Institutional Review Board (IRB) approval upon review by our institution’s IRB committee and Office of Research Compliance and Quality Improvement. Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) guidelines were adhered to (Supplementary File can be found at mdedge.com/jcomjournal).
Results
We evaluated 353 patients who met our inclusion criteria: 187 in the control group, and 166 in the postintervention group. Ten patients were readmitted to the ward after their initial discharge; only the initial admission encounters were included in our analysis. Median age, sex, median Charlson Comorbidity Index, and incidence of surgery with anesthesia during hospitalization were comparable between the control and postintervention groups and are summarized in Table 2.

In the control group, 1572 CAMs were performed, with 74 positive CAMs recorded among 27 patients with delirium. In the postintervention group, 1298 CAMs were performed, with 12 positive CAMs recorded among 7 patients with delirium (Figure 2). Primary and secondary outcomes, as well as CAM compliance measures, are summarized in Table 3.

Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%, P = .002) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%, P = .002). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days, P = .004), mean length of stay (8.5 days vs 5.9 days, P = .003), and use of an indwelling urinary catheter (9.1% vs 2.4%, P = .012). There was a trend towards decreased incidence of chemical restraints and psychiatry consults, which did not reach statistical significance. Differences in mortality during hospitalization, physical restraint use, and sitter use could not be assessed due to low incidence.

Compliance with nursing CAM assessments was evaluated. Compared to the control group, the postintervention group saw a significant increase in the percentage of shifts with a CAM performed (54.7% vs 69.1%, P < .001). The median and mean number of CAMs performed per patient were similar between the control and postintervention groups.
Results of nursing surveys completed before and after the training program are listed in Table 4. After training, nurses had a greater level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium.

Discussion
Our study utilized a standardized delirium assessment tool to compare patient cohorts before and after nurse-targeted training interventions on delirium recognition and prevention. Our interventions emphasized nonpharmacologic intervention strategies, which are recommended as first-line in the management of patients with delirium.11 Patients were not excluded from the analysis based on preexisting medical conditions or recent surgery with anesthesia, to allow for conditions that are representative of community hospitals. We also did not use an inclusion criterion based on age; however, the majority of our patients were greater than 70 years old, representing those at highest risk for delirium.2 Significant outcomes among patients in the postintervention group include decreased incidence of delirium, lower average length of stay, decreased indwelling urinary catheter use, and increased compliance with delirium screening by nursing staff.
While the study’s focus was primarily on delirium prevention rather than treatment, these strategies may also have conferred the benefit of reversing delirium symptoms. In addition to measuring incidence of delirium, our primary outcome of percentage of tested shifts with 1 or more positive CAM was intended to assess the overall duration in which patients had delirium during their hospitalization. The reduction in shifts with positive CAMs observed in the postintervention group is notable, given that a significant percentage of patients with hospital delirium have the potential for symptom reversibility.12
Multiple studies have shown that admitted patients who develop delirium experience prolonged hospital stays, often up to 5 to 10 days longer.12-14 The decreased incidence and duration of delirium in our postintervention group is a reasonable explanation for the observed decrease in average length of stay. Our study is in line with previously documented initiatives that show that nonpharmacologic interventions can effectively address downstream health and fiscal sequelae of hospital delirium. For example, a volunteer-based initiative named the Hospital Elder Life Program, from which elements in our order set were modeled after, demonstrated significant reductions in delirium incidence, length of stay, and health care costs.14-16 Other initiatives that focused on educational training for nurses to assess and prevent delirium have also demonstrated similar positive results.17-19 Our study provides a model for effective nursing-focused education that can be reproduced in the hospital setting.
Unlike some other studies, which identified delirium based only on physician assessments, our initiative utilized the CAM performed by floor nurses to identify delirium. While this method
Our study demonstrated an increase in the overall compliance with the CAM screening during the postintervention period, which is significant given the under-recognition of delirium by health care professionals.20 We attribute this increase to greater realized importance and a higher level of confidence from nursing staff in recognizing and addressing delirium, as supported by survey data. While the increased screening of patients should be considered a positive outcome, it also poses the possibility that the observed decrease in delirium incidence in the postintervention group was in fact due to more CAMs performed on patients without delirium. Likewise, nurses may have become more adept at recognizing true delirium, as opposed to delirium mimics, in the latter period of the study.
Perhaps the greatest limitation of our study is the variability in performing and recording CAMs, as some patients had multiple CAMs recorded while others did not have any CAMs recorded. This may have been affected in part by the increase in COVID-19 cases in our hospital towards the latter half of the study, which resulted in changes in nursing assignments as well as patient comorbidities in ways that cannot be easily quantified. Given the limited size of our patient cohorts, certain outcomes, such as the use of sitters, physical restraints, and in-hospital mortality, were unable to be assessed for changes statistically. Causative relationships between our interventions and associated outcome measures are necessarily limited in a binary comparison between control and postintervention groups.
Within these limitations, our study demonstrates promising results in core dimensions of patient care. We anticipate further quality improvement initiatives involving greater numbers of nursing staff and patients to better quantify the impact of nonpharmacologic nursing-centered interventions for preventing hospital delirium.
Conclusion
A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs. Education and tools to equip nurses to perform standardized delirium screening and interventions should be prioritized.
Acknowledgment: The authors thanks Olena Svetlov, NP, Oscar Abarca, Jose Chavez, and Jenita Gutierrez.
Corresponding author: Jason Ching, MD, Department of Neurology, Cedars-Sinai Medical Center, 8700 Beverly Blvd, Los Angeles, CA 90048; jason.ching@cshs.org.
Financial disclosures: None.
Funding: This research was supported by NIH National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR001881.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th edition. American Psychiatric Association; 2013.
2. Vasilevskis EE, Han JH, Hughes CG, et al. Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol. 2012;26(3):277-287. doi:10.1016/j.bpa.2012.07003
3. Leslie DL, Marcantonio ER, Zhang Y, et al. One-year health care costs associated with delirium in the elderly population. Arch Intern Med. 2008;168(1):27-32. doi:10.1001/archinternmed.2007.4
4. McCusker J, Cole M, Abrahamowicz M, et al. Delirium predicts 12-month mortality. Arch Intern Med. 2002;162(4):457-463. doi:10.1001/archinte.162.4.457
5. Witlox J, Eurelings LS, de Jonghe JF, et al. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304(4):443-451. doi:10.1001/jama.2010.1013
6. Gross AL, Jones RN, Habtemariam DA, et al. Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324-1331. doi:10.1001/archinternmed.2012.3203
7. Inouye SK. Prevention of delirium in hospitalized older patients: risk factors and targeted intervention strategies. Ann Med. 2000;32(4):257-263. doi:10.3109/07853890009011770
8. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. doi:10.7326/0003-4819-113-12-941
10. R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2017.
11. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220. doi:10.1038/nrneurol.2009.24
12. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364. doi:10.1093/ageing/afl005
13. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291(14):1753-1762. doi:10.1001/jama.291.14.1753
14. Chen CC, Lin MT, Tien YW, et al. Modified Hospital Elder Life Program: effects on abdominal surgery patients. J Am Coll Surg. 2011;213(2):245-252. doi:10.1016/j.jamcollsurg.2011.05.004
15. Zaubler TS, Murphy K, Rizzuto L, et al. Quality improvement and cost savings with multicomponent delirium interventions: replication of the Hospital Elder Life Program in a community hospital. Psychosomatics. 2013;54(3):219-226. doi:10.1016/j.psym.2013.01.010
16. Rubin FH, Neal K, Fenlon K, et al. Sustainability and scalability of the Hospital Elder Life Program at a community hospital. J Am Geriatr Soc. 2011;59(2):359-365. doi:10.1111/j.1532-5415.2010.03243.x
17. Milisen K, Foreman MD, Abraham IL, et al. A nurse-led interdisciplinary intervention program for delirium in elderly hip-fracture patients. J Am Geriatr Soc. 2001;49(5):523-532. doi:10.1046/j.1532-5415.2001.49109.x
18. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628. doi:10.1111/j.1532-5415.2005.53210.x
19. Tabet N, Hudson S, Sweeney V, et al. An educational intervention can prevent delirium on acute medical wards. Age Ageing. 2005;34(2):152-156. doi:10.1093/ageing/afi0320. Han JH, Zimmerman EE, Cutler N, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009;16(3):193-200. doi:10.1111/j.1553-2712.2008.00339.x
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th edition. American Psychiatric Association; 2013.
2. Vasilevskis EE, Han JH, Hughes CG, et al. Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol. 2012;26(3):277-287. doi:10.1016/j.bpa.2012.07003
3. Leslie DL, Marcantonio ER, Zhang Y, et al. One-year health care costs associated with delirium in the elderly population. Arch Intern Med. 2008;168(1):27-32. doi:10.1001/archinternmed.2007.4
4. McCusker J, Cole M, Abrahamowicz M, et al. Delirium predicts 12-month mortality. Arch Intern Med. 2002;162(4):457-463. doi:10.1001/archinte.162.4.457
5. Witlox J, Eurelings LS, de Jonghe JF, et al. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304(4):443-451. doi:10.1001/jama.2010.1013
6. Gross AL, Jones RN, Habtemariam DA, et al. Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324-1331. doi:10.1001/archinternmed.2012.3203
7. Inouye SK. Prevention of delirium in hospitalized older patients: risk factors and targeted intervention strategies. Ann Med. 2000;32(4):257-263. doi:10.3109/07853890009011770
8. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. doi:10.7326/0003-4819-113-12-941
10. R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2017.
11. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220. doi:10.1038/nrneurol.2009.24
12. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364. doi:10.1093/ageing/afl005
13. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291(14):1753-1762. doi:10.1001/jama.291.14.1753
14. Chen CC, Lin MT, Tien YW, et al. Modified Hospital Elder Life Program: effects on abdominal surgery patients. J Am Coll Surg. 2011;213(2):245-252. doi:10.1016/j.jamcollsurg.2011.05.004
15. Zaubler TS, Murphy K, Rizzuto L, et al. Quality improvement and cost savings with multicomponent delirium interventions: replication of the Hospital Elder Life Program in a community hospital. Psychosomatics. 2013;54(3):219-226. doi:10.1016/j.psym.2013.01.010
16. Rubin FH, Neal K, Fenlon K, et al. Sustainability and scalability of the Hospital Elder Life Program at a community hospital. J Am Geriatr Soc. 2011;59(2):359-365. doi:10.1111/j.1532-5415.2010.03243.x
17. Milisen K, Foreman MD, Abraham IL, et al. A nurse-led interdisciplinary intervention program for delirium in elderly hip-fracture patients. J Am Geriatr Soc. 2001;49(5):523-532. doi:10.1046/j.1532-5415.2001.49109.x
18. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628. doi:10.1111/j.1532-5415.2005.53210.x
19. Tabet N, Hudson S, Sweeney V, et al. An educational intervention can prevent delirium on acute medical wards. Age Ageing. 2005;34(2):152-156. doi:10.1093/ageing/afi0320. Han JH, Zimmerman EE, Cutler N, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009;16(3):193-200. doi:10.1111/j.1553-2712.2008.00339.x
Beware of private equity–owned nursing homes: study
When you have to help a parent choose a nursing home or you need nursing home care yourself, you can consult a health care professional, talk to friends, or look at the Nursing Home Compare website of the Centers for Medicare and Medicaid Services (CMS). The CMS website includes star ratings for each nursing home, both overall and on health inspections, staffing and certain quality measures.
But what you might not know is what financial incentives a particular nursing home might have to provide high-quality care, depending on what kind of entity owns the facility.
According to the study, you can expect a somewhat lower level of quality in a PE-owned nursing home than in other for-profit facilities.
The researchers compared CMS data on 302 nursing homes owned by 79 PE firms to data on 9,562 for-profit facilities not owned by such companies from 2013 to 2017. Among fee-for-service Medicare patients in long-term care, private equity acquisitions of nursing homes were associated with an 11.1% increase in ambulatory-care-sensitive (ACS) visits to the emergency department (ED) and an 8.7% increase in ACS hospitalizations per quarter, compared to the changes that occurred in the non-PE-owned facilities, they found.
What’s more, Medicare costs per beneficiary increased 3.9% more – or about $1,000 a year – in the PE-owned nursing homes than they did in the other cohort during the study period.
And when the acquired nursing homes were compared to the nursing homes prior to their acquisition by PE firms, there were no statistically significant differences in unadjusted outcomes, the researchers found. That means the two cohorts were broadly comparable.
The researchers adjusted the numbers in their study for various characteristics of the facilities and their residents. For example, the PE-acquired nursing homes were likely to have a higher percentage of patients covered by Medicare and a lower percentage covered by Medicaid than their non-PE counterparts.
The mean percentages of Black residents, female residents, and residents aged 85 or older were 12.4%, 65.4%, and 36.2%, respectively, for the PE-owned nursing homes and 15.7%, 67.8%, and 39%, respectively, for the non–PE-owned facilities.
Less than optimal outcomes
On average, the residents of non–PE-owned nursing homes had better outcomes than did the patients in the PE-owned facilities. But that doesn’t mean that the average for-profit nursing home had terrific outcomes.
For all the nursing homes in the study, the mean quarterly rate of ACS emergency department visits was 14.1%, and the mean quarterly rate of ACS hospitalizations was 17.3%.
“These events should be largely, although not completely, preventable with appropriate care,” the researchers pointed out.
To date, PE firms have invested about $750 billion in U.S. health care, with nursing homes being a major target of these companies, which currently own 5% of skilled nursing facilities, per the study. PE companies seek annual returns of 20% or more, the paper says, and thus feel pressure to generate high short-term profits. That could lead to reduced staffing, services, supplies, or equipment in their facilities.
Some nursing homes purchased by PE firms may be responsible for the debt incurred in their own leveraged buyouts, the researchers noted. There is also concern that PE firms may focus their properties disproportionately on short-term post-acute care, which is reimbursed at a higher rate than long-term care, the study says.
For all these reasons, some health policy makers are concerned about the long-term impact of private-equity nursing home acquisitions, according to the study.
A version of this article first appeared on WebMD.com.
When you have to help a parent choose a nursing home or you need nursing home care yourself, you can consult a health care professional, talk to friends, or look at the Nursing Home Compare website of the Centers for Medicare and Medicaid Services (CMS). The CMS website includes star ratings for each nursing home, both overall and on health inspections, staffing and certain quality measures.
But what you might not know is what financial incentives a particular nursing home might have to provide high-quality care, depending on what kind of entity owns the facility.
According to the study, you can expect a somewhat lower level of quality in a PE-owned nursing home than in other for-profit facilities.
The researchers compared CMS data on 302 nursing homes owned by 79 PE firms to data on 9,562 for-profit facilities not owned by such companies from 2013 to 2017. Among fee-for-service Medicare patients in long-term care, private equity acquisitions of nursing homes were associated with an 11.1% increase in ambulatory-care-sensitive (ACS) visits to the emergency department (ED) and an 8.7% increase in ACS hospitalizations per quarter, compared to the changes that occurred in the non-PE-owned facilities, they found.
What’s more, Medicare costs per beneficiary increased 3.9% more – or about $1,000 a year – in the PE-owned nursing homes than they did in the other cohort during the study period.
And when the acquired nursing homes were compared to the nursing homes prior to their acquisition by PE firms, there were no statistically significant differences in unadjusted outcomes, the researchers found. That means the two cohorts were broadly comparable.
The researchers adjusted the numbers in their study for various characteristics of the facilities and their residents. For example, the PE-acquired nursing homes were likely to have a higher percentage of patients covered by Medicare and a lower percentage covered by Medicaid than their non-PE counterparts.
The mean percentages of Black residents, female residents, and residents aged 85 or older were 12.4%, 65.4%, and 36.2%, respectively, for the PE-owned nursing homes and 15.7%, 67.8%, and 39%, respectively, for the non–PE-owned facilities.
Less than optimal outcomes
On average, the residents of non–PE-owned nursing homes had better outcomes than did the patients in the PE-owned facilities. But that doesn’t mean that the average for-profit nursing home had terrific outcomes.
For all the nursing homes in the study, the mean quarterly rate of ACS emergency department visits was 14.1%, and the mean quarterly rate of ACS hospitalizations was 17.3%.
“These events should be largely, although not completely, preventable with appropriate care,” the researchers pointed out.
To date, PE firms have invested about $750 billion in U.S. health care, with nursing homes being a major target of these companies, which currently own 5% of skilled nursing facilities, per the study. PE companies seek annual returns of 20% or more, the paper says, and thus feel pressure to generate high short-term profits. That could lead to reduced staffing, services, supplies, or equipment in their facilities.
Some nursing homes purchased by PE firms may be responsible for the debt incurred in their own leveraged buyouts, the researchers noted. There is also concern that PE firms may focus their properties disproportionately on short-term post-acute care, which is reimbursed at a higher rate than long-term care, the study says.
For all these reasons, some health policy makers are concerned about the long-term impact of private-equity nursing home acquisitions, according to the study.
A version of this article first appeared on WebMD.com.
When you have to help a parent choose a nursing home or you need nursing home care yourself, you can consult a health care professional, talk to friends, or look at the Nursing Home Compare website of the Centers for Medicare and Medicaid Services (CMS). The CMS website includes star ratings for each nursing home, both overall and on health inspections, staffing and certain quality measures.
But what you might not know is what financial incentives a particular nursing home might have to provide high-quality care, depending on what kind of entity owns the facility.
According to the study, you can expect a somewhat lower level of quality in a PE-owned nursing home than in other for-profit facilities.
The researchers compared CMS data on 302 nursing homes owned by 79 PE firms to data on 9,562 for-profit facilities not owned by such companies from 2013 to 2017. Among fee-for-service Medicare patients in long-term care, private equity acquisitions of nursing homes were associated with an 11.1% increase in ambulatory-care-sensitive (ACS) visits to the emergency department (ED) and an 8.7% increase in ACS hospitalizations per quarter, compared to the changes that occurred in the non-PE-owned facilities, they found.
What’s more, Medicare costs per beneficiary increased 3.9% more – or about $1,000 a year – in the PE-owned nursing homes than they did in the other cohort during the study period.
And when the acquired nursing homes were compared to the nursing homes prior to their acquisition by PE firms, there were no statistically significant differences in unadjusted outcomes, the researchers found. That means the two cohorts were broadly comparable.
The researchers adjusted the numbers in their study for various characteristics of the facilities and their residents. For example, the PE-acquired nursing homes were likely to have a higher percentage of patients covered by Medicare and a lower percentage covered by Medicaid than their non-PE counterparts.
The mean percentages of Black residents, female residents, and residents aged 85 or older were 12.4%, 65.4%, and 36.2%, respectively, for the PE-owned nursing homes and 15.7%, 67.8%, and 39%, respectively, for the non–PE-owned facilities.
Less than optimal outcomes
On average, the residents of non–PE-owned nursing homes had better outcomes than did the patients in the PE-owned facilities. But that doesn’t mean that the average for-profit nursing home had terrific outcomes.
For all the nursing homes in the study, the mean quarterly rate of ACS emergency department visits was 14.1%, and the mean quarterly rate of ACS hospitalizations was 17.3%.
“These events should be largely, although not completely, preventable with appropriate care,” the researchers pointed out.
To date, PE firms have invested about $750 billion in U.S. health care, with nursing homes being a major target of these companies, which currently own 5% of skilled nursing facilities, per the study. PE companies seek annual returns of 20% or more, the paper says, and thus feel pressure to generate high short-term profits. That could lead to reduced staffing, services, supplies, or equipment in their facilities.
Some nursing homes purchased by PE firms may be responsible for the debt incurred in their own leveraged buyouts, the researchers noted. There is also concern that PE firms may focus their properties disproportionately on short-term post-acute care, which is reimbursed at a higher rate than long-term care, the study says.
For all these reasons, some health policy makers are concerned about the long-term impact of private-equity nursing home acquisitions, according to the study.
A version of this article first appeared on WebMD.com.
Case: Older patient with T2D has recurrent flushing
He has had no other symptoms. His only abnormalities on physical exam are a blood pressure of 160/100 and mild peripheral edema.
His current medications include: Famotidine 20 mg b.i.d., Pseudoephedrine/guaifenesin SR b.i.d., Metformin 1,000 mg twice a day, Nifedipine 60 mg XL once a day, and Atorvastatin 20 mg once a day.
His laboratory work up includes: blood urea nitrogen: 20, creatinine: 1.3, sodium: 140, Chloride: 104, potassium: 3.9, glucose: 205, white blood cell count: 6,000, hematocrit: 41, 24-hour urine 5-hydroxyindoleacetic acid (5HIAA) test: 12 mg/day (normal 2-8 mg/day), free catecholamines: 80 mg/24 hours (normal less than 100 mg/24 hours).
What is the most likely diagnosis?
A. Drug effect
B. Pheochromocytoma
C. Carcinoid syndrome
D. Mastocytosis
E. Medullary thyroid cancer
The most likely diagnosis is a drug effect. His flushing is likely caused by nifedipine.
Flushing is one of the most common side effects of this drug.1 This patient had lab testing done for carcinoid (urine 5HIAA), presumably because he had flushing. This lab test result was a false positive, likely because of guaifenesin ingestion, which can cause false-positive 5HIAA results.2
Carcinoid syndrome is very rare (estimates from less than 1 patient/100,000), and the vast majority of patients who have it present with metastatic disease at presentation. Drug side effects are common, and usually are much more likely than rare diseases.
Four principles for assisting with making a diagnosis
This case points out the following four principles that I will touch on to help us make diagnoses in challenging cases.
1. Trigger symptoms: These are symptoms that make us think of a rare disease. In this case, the symptom is flushing, which may make you think of carcinoid syndrome.
Another good example of a trigger symptom is night sweats, where you may think of tuberculosis or lymphoma. These symptoms almost always have a much more common and likely cause, which in this case is a common drug side effect.
Trigger symptoms are great to pay attention to, but do not jump to working up the rare diagnosis without more evidence that it is a plausible diagnosis. Working up rare diseases without a reasonable pretest probability will lead to significant false-positive results.
2. Distinguishing features: These are findings, or combinations of findings, that make rarer diseases more likely. For example, flushing, although seen in many patients with carcinoid syndrome, is much more commonly caused by rosacea, medications, or estrogen/testosterone deficiency.
If a patient presents with flushing plus diarrhea, carcinoid syndrome becomes more likely in differentials. An example of a specific distinguishing feature is transient visual obstructions in patients with idiopathic intracranial hypertension (IIH or pseudotumor cerebri).
Sudden transient visual loss is not a symptom we see often, but headaches and obesity are problems we see every day. A patient with headaches and obesity is very likely to have IIH if they have transient visual obstructions along with headaches and obesity.
3. Intentional physical exams: Do the physical exam focusing on what findings will change your diagnostic probabilities. For example, in this case, if you are considering carcinoid, do a careful abdominal exam, with close attention to the liver, as 75% of patients with carcinoid syndrome have liver metastases.
If you are thinking about IIH, a fundoscopic exam is mandatory, as papilledema is a key feature of this diagnosis.
Read about the rare diagnosis you are considering, this will help with targeting your exam.
4. Remember the unusual presentation of a common disease is more common than the common presentation of a rare disease: Good examples of this are sleep apnea and gastroesophageal reflux disease causing night sweats more commonly than finding lymphomas or active tuberculosis (in the United States) as the cause.3
Pearl: Trigger symptoms help us think of rare diseases, but distinguishing features are most helpful in including or excluding the diagnosis.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Gueret P et al. Drugs. 1990;39 Suppl 2:67-72.
2. Corcuff J et al. Endocr Connect. 2017;6:R87.
3. Smith CS and Paauw DS. J Am Board Fam Pract. 2000;13:424-9.
He has had no other symptoms. His only abnormalities on physical exam are a blood pressure of 160/100 and mild peripheral edema.
His current medications include: Famotidine 20 mg b.i.d., Pseudoephedrine/guaifenesin SR b.i.d., Metformin 1,000 mg twice a day, Nifedipine 60 mg XL once a day, and Atorvastatin 20 mg once a day.
His laboratory work up includes: blood urea nitrogen: 20, creatinine: 1.3, sodium: 140, Chloride: 104, potassium: 3.9, glucose: 205, white blood cell count: 6,000, hematocrit: 41, 24-hour urine 5-hydroxyindoleacetic acid (5HIAA) test: 12 mg/day (normal 2-8 mg/day), free catecholamines: 80 mg/24 hours (normal less than 100 mg/24 hours).
What is the most likely diagnosis?
A. Drug effect
B. Pheochromocytoma
C. Carcinoid syndrome
D. Mastocytosis
E. Medullary thyroid cancer
The most likely diagnosis is a drug effect. His flushing is likely caused by nifedipine.
Flushing is one of the most common side effects of this drug.1 This patient had lab testing done for carcinoid (urine 5HIAA), presumably because he had flushing. This lab test result was a false positive, likely because of guaifenesin ingestion, which can cause false-positive 5HIAA results.2
Carcinoid syndrome is very rare (estimates from less than 1 patient/100,000), and the vast majority of patients who have it present with metastatic disease at presentation. Drug side effects are common, and usually are much more likely than rare diseases.
Four principles for assisting with making a diagnosis
This case points out the following four principles that I will touch on to help us make diagnoses in challenging cases.
1. Trigger symptoms: These are symptoms that make us think of a rare disease. In this case, the symptom is flushing, which may make you think of carcinoid syndrome.
Another good example of a trigger symptom is night sweats, where you may think of tuberculosis or lymphoma. These symptoms almost always have a much more common and likely cause, which in this case is a common drug side effect.
Trigger symptoms are great to pay attention to, but do not jump to working up the rare diagnosis without more evidence that it is a plausible diagnosis. Working up rare diseases without a reasonable pretest probability will lead to significant false-positive results.
2. Distinguishing features: These are findings, or combinations of findings, that make rarer diseases more likely. For example, flushing, although seen in many patients with carcinoid syndrome, is much more commonly caused by rosacea, medications, or estrogen/testosterone deficiency.
If a patient presents with flushing plus diarrhea, carcinoid syndrome becomes more likely in differentials. An example of a specific distinguishing feature is transient visual obstructions in patients with idiopathic intracranial hypertension (IIH or pseudotumor cerebri).
Sudden transient visual loss is not a symptom we see often, but headaches and obesity are problems we see every day. A patient with headaches and obesity is very likely to have IIH if they have transient visual obstructions along with headaches and obesity.
3. Intentional physical exams: Do the physical exam focusing on what findings will change your diagnostic probabilities. For example, in this case, if you are considering carcinoid, do a careful abdominal exam, with close attention to the liver, as 75% of patients with carcinoid syndrome have liver metastases.
If you are thinking about IIH, a fundoscopic exam is mandatory, as papilledema is a key feature of this diagnosis.
Read about the rare diagnosis you are considering, this will help with targeting your exam.
4. Remember the unusual presentation of a common disease is more common than the common presentation of a rare disease: Good examples of this are sleep apnea and gastroesophageal reflux disease causing night sweats more commonly than finding lymphomas or active tuberculosis (in the United States) as the cause.3
Pearl: Trigger symptoms help us think of rare diseases, but distinguishing features are most helpful in including or excluding the diagnosis.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Gueret P et al. Drugs. 1990;39 Suppl 2:67-72.
2. Corcuff J et al. Endocr Connect. 2017;6:R87.
3. Smith CS and Paauw DS. J Am Board Fam Pract. 2000;13:424-9.
He has had no other symptoms. His only abnormalities on physical exam are a blood pressure of 160/100 and mild peripheral edema.
His current medications include: Famotidine 20 mg b.i.d., Pseudoephedrine/guaifenesin SR b.i.d., Metformin 1,000 mg twice a day, Nifedipine 60 mg XL once a day, and Atorvastatin 20 mg once a day.
His laboratory work up includes: blood urea nitrogen: 20, creatinine: 1.3, sodium: 140, Chloride: 104, potassium: 3.9, glucose: 205, white blood cell count: 6,000, hematocrit: 41, 24-hour urine 5-hydroxyindoleacetic acid (5HIAA) test: 12 mg/day (normal 2-8 mg/day), free catecholamines: 80 mg/24 hours (normal less than 100 mg/24 hours).
What is the most likely diagnosis?
A. Drug effect
B. Pheochromocytoma
C. Carcinoid syndrome
D. Mastocytosis
E. Medullary thyroid cancer
The most likely diagnosis is a drug effect. His flushing is likely caused by nifedipine.
Flushing is one of the most common side effects of this drug.1 This patient had lab testing done for carcinoid (urine 5HIAA), presumably because he had flushing. This lab test result was a false positive, likely because of guaifenesin ingestion, which can cause false-positive 5HIAA results.2
Carcinoid syndrome is very rare (estimates from less than 1 patient/100,000), and the vast majority of patients who have it present with metastatic disease at presentation. Drug side effects are common, and usually are much more likely than rare diseases.
Four principles for assisting with making a diagnosis
This case points out the following four principles that I will touch on to help us make diagnoses in challenging cases.
1. Trigger symptoms: These are symptoms that make us think of a rare disease. In this case, the symptom is flushing, which may make you think of carcinoid syndrome.
Another good example of a trigger symptom is night sweats, where you may think of tuberculosis or lymphoma. These symptoms almost always have a much more common and likely cause, which in this case is a common drug side effect.
Trigger symptoms are great to pay attention to, but do not jump to working up the rare diagnosis without more evidence that it is a plausible diagnosis. Working up rare diseases without a reasonable pretest probability will lead to significant false-positive results.
2. Distinguishing features: These are findings, or combinations of findings, that make rarer diseases more likely. For example, flushing, although seen in many patients with carcinoid syndrome, is much more commonly caused by rosacea, medications, or estrogen/testosterone deficiency.
If a patient presents with flushing plus diarrhea, carcinoid syndrome becomes more likely in differentials. An example of a specific distinguishing feature is transient visual obstructions in patients with idiopathic intracranial hypertension (IIH or pseudotumor cerebri).
Sudden transient visual loss is not a symptom we see often, but headaches and obesity are problems we see every day. A patient with headaches and obesity is very likely to have IIH if they have transient visual obstructions along with headaches and obesity.
3. Intentional physical exams: Do the physical exam focusing on what findings will change your diagnostic probabilities. For example, in this case, if you are considering carcinoid, do a careful abdominal exam, with close attention to the liver, as 75% of patients with carcinoid syndrome have liver metastases.
If you are thinking about IIH, a fundoscopic exam is mandatory, as papilledema is a key feature of this diagnosis.
Read about the rare diagnosis you are considering, this will help with targeting your exam.
4. Remember the unusual presentation of a common disease is more common than the common presentation of a rare disease: Good examples of this are sleep apnea and gastroesophageal reflux disease causing night sweats more commonly than finding lymphomas or active tuberculosis (in the United States) as the cause.3
Pearl: Trigger symptoms help us think of rare diseases, but distinguishing features are most helpful in including or excluding the diagnosis.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Gueret P et al. Drugs. 1990;39 Suppl 2:67-72.
2. Corcuff J et al. Endocr Connect. 2017;6:R87.
3. Smith CS and Paauw DS. J Am Board Fam Pract. 2000;13:424-9.