User login
Reassurance on general anesthesia in young kids
LAHAINA, HAWAII – Two recent large, well-conducted, and persuasive Jessica Sprague, MD, said at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“These two studies can be cited in conversation with parents and are very reassuring for a single episode of general anesthesia,” observed Dr. Sprague, a dermatologist at Rady Children’s Hospital and the University of California, both in San Diego.
“As a take home, I think we can feel pretty confident that single exposure to short-duration general anesthesia does not have any adverse neurocognitive effects,” she added.
In 2016, the Food and Drug Administration issued a drug safety communication that general anesthesia lasting for more than 3 hours in children aged less than 3 years, or repeated shorter-duration general anesthesia, may affect the development of children’s brains. This edict caused considerable turmoil among both physicians and parents. The warning was based upon animal studies suggesting adverse effects, including abnormal axon formation and other structural changes, impaired learning and memory, and heightened emotional reactivity to threats. Preliminary human cohort studies generated conflicting results, but were tough to interpret because of potential confounding issues, most prominently the distinct possibility that the very reason the child was undergoing general anesthesia might inherently predispose to neurodevelopmental problems, the dermatologist explained.
Enter the GAS trial, a multinational, assessor-blinded study in which 722 generally healthy infants undergoing hernia repair at 28 centers in the United States and six other countries were randomized to general anesthesia for a median of 54 minutes or awake regional anesthesia. Assessment via a detailed neuropsychological test battery and parent questionnaires at age 2 and 5 years showed no between-group differences at all. Of note, the GAS trial was funded by the FDA, the National Institutes of Health, and similar national health care agencies in the other participating countries (Lancet. 2019 Feb 16;393[10172]:664-77).
The other major recent research contribution was a province-wide Ontario study led by investigators at the Hospital for Sick Children in Toronto. This retrospective study included 2,346 sibling pairs aged 4-5 years in which one child in each pair received general anesthesia as a preschooler. All participants underwent testing using the comprehensive Early Development Instrument. Reassuringly, no between-group differences were found in any of the five domains assessed by the testing: language and cognitive development, physical health and well-being, emotional health and maturity, social knowledge and competence, and communication skills and general knowledge (JAMA Pediatr. 2019 Jan 1;173[1]:29-36).
These two studies address a pressing issue, since 10% of children in the United States and other developed countries receive general anesthesia within their first 3 years of life. Common indications in dermatology include excisional surgery, laser therapy for extensive port wine birthmarks, and diagnostic MRIs.
Dr. Sprague advised that, based upon the new data, “you definitely do not want to delay necessary imaging studies or surgeries, but MRIs can often be done without general anesthesia in infants less than 2 months old. If you have an infant who needs an MRI for something like PHACE syndrome [posterior fossa brain malformations, hemangioma, arterial lesions, cardiac abnormalities, and eye abnormalities], if you can get them in before 2 months of age sometimes you can avoid the general anesthesia if you wrap them tight enough. But once they get over 2 months ,there’s too much wiggle and it’s pretty impossible.”
Her other suggestions:
- Consider delaying nonurgent surgeries and imaging until at least age 6 months and ideally 3 years. “Parents will eventually want surgery to be done for a benign-appearing congenital nevus on the cheek, but it doesn’t necessarily need to be done before 6 months. The same with a residual hemangioma. I would recommend doing it before they go to kindergarten and before they get a sort of sense of what their self looks like, but you have some time between ages 3 and 5 to do that,” Dr. Sprague said.
- Seek out an anesthesiologist who has extensive experience with infants and young children, as is common at a dedicated children’s hospital. “If you live somewhere where the anesthesiologists are primarily seeing adult patients, they’re just not as good,” according to the pediatric dermatologist.
- Definitely consider a topical anesthesia strategy in infants who require multiple procedures, because there remains some unresolved concern about the potential neurodevelopmental impact of multiple bouts of general anesthesia.
Dr. Sprague reported having no financial conflicts regarding her presentation.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Two recent large, well-conducted, and persuasive Jessica Sprague, MD, said at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“These two studies can be cited in conversation with parents and are very reassuring for a single episode of general anesthesia,” observed Dr. Sprague, a dermatologist at Rady Children’s Hospital and the University of California, both in San Diego.
“As a take home, I think we can feel pretty confident that single exposure to short-duration general anesthesia does not have any adverse neurocognitive effects,” she added.
In 2016, the Food and Drug Administration issued a drug safety communication that general anesthesia lasting for more than 3 hours in children aged less than 3 years, or repeated shorter-duration general anesthesia, may affect the development of children’s brains. This edict caused considerable turmoil among both physicians and parents. The warning was based upon animal studies suggesting adverse effects, including abnormal axon formation and other structural changes, impaired learning and memory, and heightened emotional reactivity to threats. Preliminary human cohort studies generated conflicting results, but were tough to interpret because of potential confounding issues, most prominently the distinct possibility that the very reason the child was undergoing general anesthesia might inherently predispose to neurodevelopmental problems, the dermatologist explained.
Enter the GAS trial, a multinational, assessor-blinded study in which 722 generally healthy infants undergoing hernia repair at 28 centers in the United States and six other countries were randomized to general anesthesia for a median of 54 minutes or awake regional anesthesia. Assessment via a detailed neuropsychological test battery and parent questionnaires at age 2 and 5 years showed no between-group differences at all. Of note, the GAS trial was funded by the FDA, the National Institutes of Health, and similar national health care agencies in the other participating countries (Lancet. 2019 Feb 16;393[10172]:664-77).
The other major recent research contribution was a province-wide Ontario study led by investigators at the Hospital for Sick Children in Toronto. This retrospective study included 2,346 sibling pairs aged 4-5 years in which one child in each pair received general anesthesia as a preschooler. All participants underwent testing using the comprehensive Early Development Instrument. Reassuringly, no between-group differences were found in any of the five domains assessed by the testing: language and cognitive development, physical health and well-being, emotional health and maturity, social knowledge and competence, and communication skills and general knowledge (JAMA Pediatr. 2019 Jan 1;173[1]:29-36).
These two studies address a pressing issue, since 10% of children in the United States and other developed countries receive general anesthesia within their first 3 years of life. Common indications in dermatology include excisional surgery, laser therapy for extensive port wine birthmarks, and diagnostic MRIs.
Dr. Sprague advised that, based upon the new data, “you definitely do not want to delay necessary imaging studies or surgeries, but MRIs can often be done without general anesthesia in infants less than 2 months old. If you have an infant who needs an MRI for something like PHACE syndrome [posterior fossa brain malformations, hemangioma, arterial lesions, cardiac abnormalities, and eye abnormalities], if you can get them in before 2 months of age sometimes you can avoid the general anesthesia if you wrap them tight enough. But once they get over 2 months ,there’s too much wiggle and it’s pretty impossible.”
Her other suggestions:
- Consider delaying nonurgent surgeries and imaging until at least age 6 months and ideally 3 years. “Parents will eventually want surgery to be done for a benign-appearing congenital nevus on the cheek, but it doesn’t necessarily need to be done before 6 months. The same with a residual hemangioma. I would recommend doing it before they go to kindergarten and before they get a sort of sense of what their self looks like, but you have some time between ages 3 and 5 to do that,” Dr. Sprague said.
- Seek out an anesthesiologist who has extensive experience with infants and young children, as is common at a dedicated children’s hospital. “If you live somewhere where the anesthesiologists are primarily seeing adult patients, they’re just not as good,” according to the pediatric dermatologist.
- Definitely consider a topical anesthesia strategy in infants who require multiple procedures, because there remains some unresolved concern about the potential neurodevelopmental impact of multiple bouts of general anesthesia.
Dr. Sprague reported having no financial conflicts regarding her presentation.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Two recent large, well-conducted, and persuasive Jessica Sprague, MD, said at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“These two studies can be cited in conversation with parents and are very reassuring for a single episode of general anesthesia,” observed Dr. Sprague, a dermatologist at Rady Children’s Hospital and the University of California, both in San Diego.
“As a take home, I think we can feel pretty confident that single exposure to short-duration general anesthesia does not have any adverse neurocognitive effects,” she added.
In 2016, the Food and Drug Administration issued a drug safety communication that general anesthesia lasting for more than 3 hours in children aged less than 3 years, or repeated shorter-duration general anesthesia, may affect the development of children’s brains. This edict caused considerable turmoil among both physicians and parents. The warning was based upon animal studies suggesting adverse effects, including abnormal axon formation and other structural changes, impaired learning and memory, and heightened emotional reactivity to threats. Preliminary human cohort studies generated conflicting results, but were tough to interpret because of potential confounding issues, most prominently the distinct possibility that the very reason the child was undergoing general anesthesia might inherently predispose to neurodevelopmental problems, the dermatologist explained.
Enter the GAS trial, a multinational, assessor-blinded study in which 722 generally healthy infants undergoing hernia repair at 28 centers in the United States and six other countries were randomized to general anesthesia for a median of 54 minutes or awake regional anesthesia. Assessment via a detailed neuropsychological test battery and parent questionnaires at age 2 and 5 years showed no between-group differences at all. Of note, the GAS trial was funded by the FDA, the National Institutes of Health, and similar national health care agencies in the other participating countries (Lancet. 2019 Feb 16;393[10172]:664-77).
The other major recent research contribution was a province-wide Ontario study led by investigators at the Hospital for Sick Children in Toronto. This retrospective study included 2,346 sibling pairs aged 4-5 years in which one child in each pair received general anesthesia as a preschooler. All participants underwent testing using the comprehensive Early Development Instrument. Reassuringly, no between-group differences were found in any of the five domains assessed by the testing: language and cognitive development, physical health and well-being, emotional health and maturity, social knowledge and competence, and communication skills and general knowledge (JAMA Pediatr. 2019 Jan 1;173[1]:29-36).
These two studies address a pressing issue, since 10% of children in the United States and other developed countries receive general anesthesia within their first 3 years of life. Common indications in dermatology include excisional surgery, laser therapy for extensive port wine birthmarks, and diagnostic MRIs.
Dr. Sprague advised that, based upon the new data, “you definitely do not want to delay necessary imaging studies or surgeries, but MRIs can often be done without general anesthesia in infants less than 2 months old. If you have an infant who needs an MRI for something like PHACE syndrome [posterior fossa brain malformations, hemangioma, arterial lesions, cardiac abnormalities, and eye abnormalities], if you can get them in before 2 months of age sometimes you can avoid the general anesthesia if you wrap them tight enough. But once they get over 2 months ,there’s too much wiggle and it’s pretty impossible.”
Her other suggestions:
- Consider delaying nonurgent surgeries and imaging until at least age 6 months and ideally 3 years. “Parents will eventually want surgery to be done for a benign-appearing congenital nevus on the cheek, but it doesn’t necessarily need to be done before 6 months. The same with a residual hemangioma. I would recommend doing it before they go to kindergarten and before they get a sort of sense of what their self looks like, but you have some time between ages 3 and 5 to do that,” Dr. Sprague said.
- Seek out an anesthesiologist who has extensive experience with infants and young children, as is common at a dedicated children’s hospital. “If you live somewhere where the anesthesiologists are primarily seeing adult patients, they’re just not as good,” according to the pediatric dermatologist.
- Definitely consider a topical anesthesia strategy in infants who require multiple procedures, because there remains some unresolved concern about the potential neurodevelopmental impact of multiple bouts of general anesthesia.
Dr. Sprague reported having no financial conflicts regarding her presentation.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Nail dystrophy and nail plate thinning
At a follow-up visit, a biopsy of the skin on the fingertips was performed, which showed lichenoid lymphocytic inflammatory infiltrate with associated hyperkeratosis, hypergranulosis, and acanthosis.
No fungal elements were seen. The findings were consistent with lichen planus.
The patient was started on hydroxychloroquine. It was recommended she start a 6-week course of oral prednisone, but the mother was opposed to systemic treatment because of potential side effects.
She continued topical betamethasone without much change. Topical tacrolimus later was recommended to use on off days of betamethasone, which led to no improvement. Narrow-band UVB also was started with minimal improvement. Unfortunately,
Nail lichen planus (NLP) in children is not a common condition.1 In a recent series from Chiheb et al., NLP was reported in 90 patients, of which 40% were children; a quarter of the patients reported having extracutaneous involvement as well.2 In another childhood LP series,14 % of the children presented with nail disease.3 It can be a severe disease that, if not treated aggressively, may lead to destruction of the nail bed. This condition seems to be more prevalent in boys than girls and more prevalent in African American children.3 Unfortunately, in this patient’s case, the mother was hesitant to use systemic therapy and aggressive treatment was delayed.
Possible but not clear associations with autoimmune conditions such as vitiligo, autoimmune thyroiditis, myasthenia gravis, alopecia areata, thymoma, autoimmune polyendocrinopathy, atopic dermatitis, and lichen nitidus have been described in children with LP.
The clinical characteristics of NLP include nail plate thinning with longitudinal ridging and fissuring, with or without pterygium; trachyonychia; and erythema of the lunula when the nail matrix is involved. When the nail bed is affected, the patient can present with onycholysis with or without subungual hyperkeratosis and violaceous hue of the nail bed.4 NLP can have three different clinical presentations described by Tosti et al., which include typical NLP, 20‐nail dystrophy (trachyonychia), and idiopathic nail atrophy. Idiopathic nail atrophy is described solely in children as an acute and rapid progression that leads to destruction of the nail within months, which appears to be the clinical presentation in our patient.
The differential diagnosis of nail dystrophy in children includes infectious processes such as onychomycosis, especially when children present with onycholysis and subungual hyperkeratosis. Because of this, it is recommended to perform a nail culture or submit a sample of nail clippings for microscopic evaluation to confirm the diagnosis of onychomycosis prior to starting systemic therapy in children. Fingernail involvement without toenail involvement is an unusual presentation of onychomycosis.
Twenty-nail dystrophy – also known as trachyonychia – can be caused by several inflammatory skin conditions such as lichen planus, psoriasis, eczema, pemphigus vulgaris, and alopecia areata. Clinically, there is uniformly monomorphic thinning of the nail plate with longitudinal ridging without splitting or pterygium.1 This is a benign condition and should not cause scarring. About 10% of the cases of 20-nail dystrophy are caused by lichen planus.
Nail psoriasis is characterized by nail pitting, oil spots on the nail plate, leukonychia, subungual hyperkeratosis, and onycholysis, as well as nail crumbling, which were not seen in our patient. Although her initial presentation was of 20-nail dystrophy, which also can be a presentation of nail psoriasis, its rapid evolution with associated nail atrophy and pterygium make it unlikely to be psoriasis in this particular patient.
Patients with pachyonychia congenita – which is a genetic disorder or keratinization caused by mutations on several genes encoding keratin such as K6a, K16, K17, K6b, and possibly K6c – present with nail thickening (pachyonychia) and discoloration of the nails, as well as pincer nails. These patients also present with oral leukokeratosis and focal palmoplantar keratoderma.
The main treatment of lichen planus is potent topical corticosteroids.
For nail disease, topical treatment may not be effective and systemic treatment may be necessary. Systemic corticosteroids have been used in several pediatric series varying from a short course given at a dose of 1- 2 mg/kg per day for 2 weeks to a longer 3-month course followed by tapering.3 There are several protocols of intramuscular triamcinolone at a dose of 0.5 mg/kg in children in once a month injections for about 3 months that have been reported successful with minimal side effects.1 Other medications reported useful in patients with NLP include dapsone and acitretin. Other treatment options include narrow-band UVB and PUVA.3
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Email her at pdnews@mdedge.com.
References
1. Arch Dermatol. 2001 Aug;137(8):1027-32.
2. Ann Dermatol Venereol. 2015 Jan;142(1):21-5.
3. Pediatr Dermatol. 2014 Jan-Feb;31(1):59-67.
4. Dermatological diseases, in “Nails: Diagnosis, Therapy, and Surgery,” 3rd ed. (Oxford: Elsevier Saunders, 2005, p. 105).
At a follow-up visit, a biopsy of the skin on the fingertips was performed, which showed lichenoid lymphocytic inflammatory infiltrate with associated hyperkeratosis, hypergranulosis, and acanthosis.
No fungal elements were seen. The findings were consistent with lichen planus.
The patient was started on hydroxychloroquine. It was recommended she start a 6-week course of oral prednisone, but the mother was opposed to systemic treatment because of potential side effects.
She continued topical betamethasone without much change. Topical tacrolimus later was recommended to use on off days of betamethasone, which led to no improvement. Narrow-band UVB also was started with minimal improvement. Unfortunately,
Nail lichen planus (NLP) in children is not a common condition.1 In a recent series from Chiheb et al., NLP was reported in 90 patients, of which 40% were children; a quarter of the patients reported having extracutaneous involvement as well.2 In another childhood LP series,14 % of the children presented with nail disease.3 It can be a severe disease that, if not treated aggressively, may lead to destruction of the nail bed. This condition seems to be more prevalent in boys than girls and more prevalent in African American children.3 Unfortunately, in this patient’s case, the mother was hesitant to use systemic therapy and aggressive treatment was delayed.
Possible but not clear associations with autoimmune conditions such as vitiligo, autoimmune thyroiditis, myasthenia gravis, alopecia areata, thymoma, autoimmune polyendocrinopathy, atopic dermatitis, and lichen nitidus have been described in children with LP.
The clinical characteristics of NLP include nail plate thinning with longitudinal ridging and fissuring, with or without pterygium; trachyonychia; and erythema of the lunula when the nail matrix is involved. When the nail bed is affected, the patient can present with onycholysis with or without subungual hyperkeratosis and violaceous hue of the nail bed.4 NLP can have three different clinical presentations described by Tosti et al., which include typical NLP, 20‐nail dystrophy (trachyonychia), and idiopathic nail atrophy. Idiopathic nail atrophy is described solely in children as an acute and rapid progression that leads to destruction of the nail within months, which appears to be the clinical presentation in our patient.
The differential diagnosis of nail dystrophy in children includes infectious processes such as onychomycosis, especially when children present with onycholysis and subungual hyperkeratosis. Because of this, it is recommended to perform a nail culture or submit a sample of nail clippings for microscopic evaluation to confirm the diagnosis of onychomycosis prior to starting systemic therapy in children. Fingernail involvement without toenail involvement is an unusual presentation of onychomycosis.
Twenty-nail dystrophy – also known as trachyonychia – can be caused by several inflammatory skin conditions such as lichen planus, psoriasis, eczema, pemphigus vulgaris, and alopecia areata. Clinically, there is uniformly monomorphic thinning of the nail plate with longitudinal ridging without splitting or pterygium.1 This is a benign condition and should not cause scarring. About 10% of the cases of 20-nail dystrophy are caused by lichen planus.
Nail psoriasis is characterized by nail pitting, oil spots on the nail plate, leukonychia, subungual hyperkeratosis, and onycholysis, as well as nail crumbling, which were not seen in our patient. Although her initial presentation was of 20-nail dystrophy, which also can be a presentation of nail psoriasis, its rapid evolution with associated nail atrophy and pterygium make it unlikely to be psoriasis in this particular patient.
Patients with pachyonychia congenita – which is a genetic disorder or keratinization caused by mutations on several genes encoding keratin such as K6a, K16, K17, K6b, and possibly K6c – present with nail thickening (pachyonychia) and discoloration of the nails, as well as pincer nails. These patients also present with oral leukokeratosis and focal palmoplantar keratoderma.
The main treatment of lichen planus is potent topical corticosteroids.
For nail disease, topical treatment may not be effective and systemic treatment may be necessary. Systemic corticosteroids have been used in several pediatric series varying from a short course given at a dose of 1- 2 mg/kg per day for 2 weeks to a longer 3-month course followed by tapering.3 There are several protocols of intramuscular triamcinolone at a dose of 0.5 mg/kg in children in once a month injections for about 3 months that have been reported successful with minimal side effects.1 Other medications reported useful in patients with NLP include dapsone and acitretin. Other treatment options include narrow-band UVB and PUVA.3
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Email her at pdnews@mdedge.com.
References
1. Arch Dermatol. 2001 Aug;137(8):1027-32.
2. Ann Dermatol Venereol. 2015 Jan;142(1):21-5.
3. Pediatr Dermatol. 2014 Jan-Feb;31(1):59-67.
4. Dermatological diseases, in “Nails: Diagnosis, Therapy, and Surgery,” 3rd ed. (Oxford: Elsevier Saunders, 2005, p. 105).
At a follow-up visit, a biopsy of the skin on the fingertips was performed, which showed lichenoid lymphocytic inflammatory infiltrate with associated hyperkeratosis, hypergranulosis, and acanthosis.
No fungal elements were seen. The findings were consistent with lichen planus.
The patient was started on hydroxychloroquine. It was recommended she start a 6-week course of oral prednisone, but the mother was opposed to systemic treatment because of potential side effects.
She continued topical betamethasone without much change. Topical tacrolimus later was recommended to use on off days of betamethasone, which led to no improvement. Narrow-band UVB also was started with minimal improvement. Unfortunately,
Nail lichen planus (NLP) in children is not a common condition.1 In a recent series from Chiheb et al., NLP was reported in 90 patients, of which 40% were children; a quarter of the patients reported having extracutaneous involvement as well.2 In another childhood LP series,14 % of the children presented with nail disease.3 It can be a severe disease that, if not treated aggressively, may lead to destruction of the nail bed. This condition seems to be more prevalent in boys than girls and more prevalent in African American children.3 Unfortunately, in this patient’s case, the mother was hesitant to use systemic therapy and aggressive treatment was delayed.
Possible but not clear associations with autoimmune conditions such as vitiligo, autoimmune thyroiditis, myasthenia gravis, alopecia areata, thymoma, autoimmune polyendocrinopathy, atopic dermatitis, and lichen nitidus have been described in children with LP.
The clinical characteristics of NLP include nail plate thinning with longitudinal ridging and fissuring, with or without pterygium; trachyonychia; and erythema of the lunula when the nail matrix is involved. When the nail bed is affected, the patient can present with onycholysis with or without subungual hyperkeratosis and violaceous hue of the nail bed.4 NLP can have three different clinical presentations described by Tosti et al., which include typical NLP, 20‐nail dystrophy (trachyonychia), and idiopathic nail atrophy. Idiopathic nail atrophy is described solely in children as an acute and rapid progression that leads to destruction of the nail within months, which appears to be the clinical presentation in our patient.
The differential diagnosis of nail dystrophy in children includes infectious processes such as onychomycosis, especially when children present with onycholysis and subungual hyperkeratosis. Because of this, it is recommended to perform a nail culture or submit a sample of nail clippings for microscopic evaluation to confirm the diagnosis of onychomycosis prior to starting systemic therapy in children. Fingernail involvement without toenail involvement is an unusual presentation of onychomycosis.
Twenty-nail dystrophy – also known as trachyonychia – can be caused by several inflammatory skin conditions such as lichen planus, psoriasis, eczema, pemphigus vulgaris, and alopecia areata. Clinically, there is uniformly monomorphic thinning of the nail plate with longitudinal ridging without splitting or pterygium.1 This is a benign condition and should not cause scarring. About 10% of the cases of 20-nail dystrophy are caused by lichen planus.
Nail psoriasis is characterized by nail pitting, oil spots on the nail plate, leukonychia, subungual hyperkeratosis, and onycholysis, as well as nail crumbling, which were not seen in our patient. Although her initial presentation was of 20-nail dystrophy, which also can be a presentation of nail psoriasis, its rapid evolution with associated nail atrophy and pterygium make it unlikely to be psoriasis in this particular patient.
Patients with pachyonychia congenita – which is a genetic disorder or keratinization caused by mutations on several genes encoding keratin such as K6a, K16, K17, K6b, and possibly K6c – present with nail thickening (pachyonychia) and discoloration of the nails, as well as pincer nails. These patients also present with oral leukokeratosis and focal palmoplantar keratoderma.
The main treatment of lichen planus is potent topical corticosteroids.
For nail disease, topical treatment may not be effective and systemic treatment may be necessary. Systemic corticosteroids have been used in several pediatric series varying from a short course given at a dose of 1- 2 mg/kg per day for 2 weeks to a longer 3-month course followed by tapering.3 There are several protocols of intramuscular triamcinolone at a dose of 0.5 mg/kg in children in once a month injections for about 3 months that have been reported successful with minimal side effects.1 Other medications reported useful in patients with NLP include dapsone and acitretin. Other treatment options include narrow-band UVB and PUVA.3
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Email her at pdnews@mdedge.com.
References
1. Arch Dermatol. 2001 Aug;137(8):1027-32.
2. Ann Dermatol Venereol. 2015 Jan;142(1):21-5.
3. Pediatr Dermatol. 2014 Jan-Feb;31(1):59-67.
4. Dermatological diseases, in “Nails: Diagnosis, Therapy, and Surgery,” 3rd ed. (Oxford: Elsevier Saunders, 2005, p. 105).
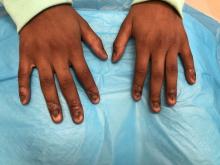
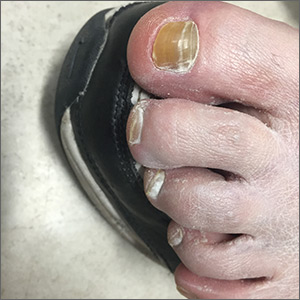
An 8-year-old female child comes to our pediatric dermatology clinic for evaluation of onychomycosis on her fingernails. The mother stated the child started developing funny-looking nails 1 year prior to the visit. It started with only two fingernails affected and now has spread to all her fingernails. Her toenails are not involved.
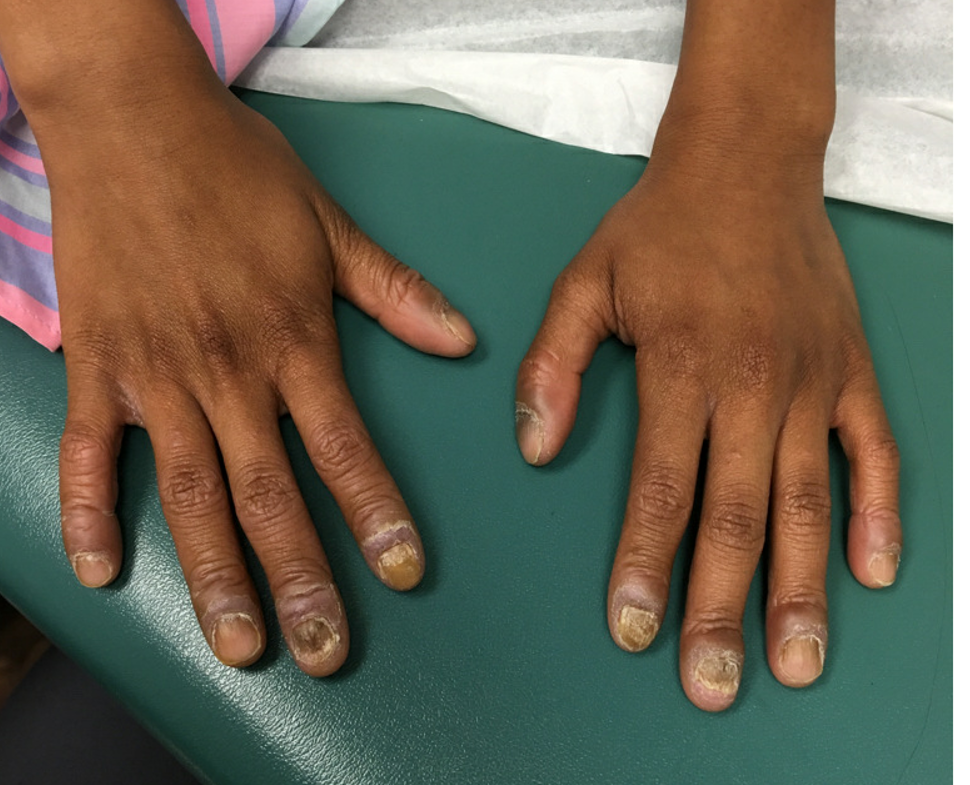
She denied any pain or itching. She initially was treated with topical antifungal medications as well as tea tree oil, apple cider vinegar, and a 6-week course of oral griseofulvin without any improvement. Her nails progressively have gotten much worse. She has no history of atopic dermatitis or any other skin conditions. She denied any joint pain, sun sensitivity, hair loss, or any other symptoms. The mother denied any family history of nail fungus, ringworm, psoriasis, or eczema.
She likes to play basketball and enjoys arts and crafts. She has a cat and a dog; neither of them have any skin problems.
On physical examination, there is nail dystrophy with nail plate thinning and longitudinal fissuring of all fingernails but not of the toenails. She also has hyperpigmented violaceous plaques on the surrounding periungual skin. There are no other skin lesions, and there are no oral or genital lesions. There is no scalp involvement or hair loss. At follow-up several months later, she had complete destruction of the nail plate with scar formation.
A fungal culture was performed, as well as microscopic analysis of the nail with periodic acid fast and giemsa stains, which showed no fungal organisms.
She initially was treated with topical betamethasone twice a day for 6 weeks and then 2 weeks on and 2 weeks off without much change.
Psoriasis elevates cancer risk
Psoriasis patients are at increased risk for several types of cancer, notably lymphoma and keratinocyte cancer, based on data from a systematic review and meta-analysis of more than 2 million patients.
Previous studies have identified an increased overall cancer risk in psoriasis patients, compared with the general population or controls without psoriasis, and both lymphomas and keratinocyte cancers occur more often in psoriasis patients, compared with controls, but additional larger studies have been conducted since the last meta-analysis was published in 2013, wrote Sofie Vaengebjerg, MD, of the University of Copenhagen and colleagues.
To better identify the risk of cancer in psoriasis and psoriatic arthritis patients and to explore the impact of biologics, the researchers reviewed data from 112 studies totaling 2,053,932 patients in a study published in JAMA Dermatology.
Overall, the risk of any cancer was slightly higher in psoriasis patients (risk ratio, 1.21; 95% confidence interval, 1.11-1.33), compared with controls, with a prevalence of 4.78% and an incidence rate of 11.75 per 1,000 person-years. The most common cancer among psoriasis patients was keratinocyte cancer, with a risk ratio of 2.28 (95% CI, 1.73-3.01), a prevalence of 2.55%, and an incidence rate of 4.35 per 1,000 person-years.
Other cancers with significantly elevated risk among psoriasis patients were lymphomas (RR, 1.56; 95% CI, 1.37-1.78), lung cancer (RR, 1.26; 95% CI, 1.13-1.40), and bladder cancer (RR, 1.12; 95% CI, 1.04-1.19).
No increased risk of cancer was noted among psoriasis patients who were treated with biologics. “However, patients receiving biologic agents are selected and the results might be reliant on selection bias, and studies investigating long-term safety of these drugs are still limited,” the researchers wrote.
In addition, psoriatic arthritis was not associated with any overall increase in cancer risk, with the exception of three studies showing an increased risk for breast cancer, the researchers noted. The overall cancer prevalence for psoriatic arthritis patients was 5.74%, with an incidence rate of 6.44 per 1,000 person-years.
The study findings were limited by several factors, including the inconsistencies in study design and characteristics and the small amount of data on biologic agents and psoriatic arthritis, the researchers noted. However, the results were strengthened by the large number of patients, real-world study settings, inclusion of biologics, and analysis of cancer in psoriatic arthritis patients.
“Clinicians treating patients with psoriasis should be aware of this increased risk, especially for lymphomas, as immunogenic treatment might be associated with exacerbations,” and should be aware that more research is needed to assess cancer risk associated with biologics, they concluded.
The study received no outside funding. Lead author Dr. Vaengebjerg had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple companies, including AbbVie, Janssen, Novartis, Eli Lilly, LEO Pharma, UCB, Almirall, and Sanofi.
SOURCE: Vaengebjerg S et al. JAMA Dermatol. 2020 Feb 19. doi:10.1001/jamadermatol.2020.0024.
Psoriasis patients are at increased risk for several types of cancer, notably lymphoma and keratinocyte cancer, based on data from a systematic review and meta-analysis of more than 2 million patients.
Previous studies have identified an increased overall cancer risk in psoriasis patients, compared with the general population or controls without psoriasis, and both lymphomas and keratinocyte cancers occur more often in psoriasis patients, compared with controls, but additional larger studies have been conducted since the last meta-analysis was published in 2013, wrote Sofie Vaengebjerg, MD, of the University of Copenhagen and colleagues.
To better identify the risk of cancer in psoriasis and psoriatic arthritis patients and to explore the impact of biologics, the researchers reviewed data from 112 studies totaling 2,053,932 patients in a study published in JAMA Dermatology.
Overall, the risk of any cancer was slightly higher in psoriasis patients (risk ratio, 1.21; 95% confidence interval, 1.11-1.33), compared with controls, with a prevalence of 4.78% and an incidence rate of 11.75 per 1,000 person-years. The most common cancer among psoriasis patients was keratinocyte cancer, with a risk ratio of 2.28 (95% CI, 1.73-3.01), a prevalence of 2.55%, and an incidence rate of 4.35 per 1,000 person-years.
Other cancers with significantly elevated risk among psoriasis patients were lymphomas (RR, 1.56; 95% CI, 1.37-1.78), lung cancer (RR, 1.26; 95% CI, 1.13-1.40), and bladder cancer (RR, 1.12; 95% CI, 1.04-1.19).
No increased risk of cancer was noted among psoriasis patients who were treated with biologics. “However, patients receiving biologic agents are selected and the results might be reliant on selection bias, and studies investigating long-term safety of these drugs are still limited,” the researchers wrote.
In addition, psoriatic arthritis was not associated with any overall increase in cancer risk, with the exception of three studies showing an increased risk for breast cancer, the researchers noted. The overall cancer prevalence for psoriatic arthritis patients was 5.74%, with an incidence rate of 6.44 per 1,000 person-years.
The study findings were limited by several factors, including the inconsistencies in study design and characteristics and the small amount of data on biologic agents and psoriatic arthritis, the researchers noted. However, the results were strengthened by the large number of patients, real-world study settings, inclusion of biologics, and analysis of cancer in psoriatic arthritis patients.
“Clinicians treating patients with psoriasis should be aware of this increased risk, especially for lymphomas, as immunogenic treatment might be associated with exacerbations,” and should be aware that more research is needed to assess cancer risk associated with biologics, they concluded.
The study received no outside funding. Lead author Dr. Vaengebjerg had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple companies, including AbbVie, Janssen, Novartis, Eli Lilly, LEO Pharma, UCB, Almirall, and Sanofi.
SOURCE: Vaengebjerg S et al. JAMA Dermatol. 2020 Feb 19. doi:10.1001/jamadermatol.2020.0024.
Psoriasis patients are at increased risk for several types of cancer, notably lymphoma and keratinocyte cancer, based on data from a systematic review and meta-analysis of more than 2 million patients.
Previous studies have identified an increased overall cancer risk in psoriasis patients, compared with the general population or controls without psoriasis, and both lymphomas and keratinocyte cancers occur more often in psoriasis patients, compared with controls, but additional larger studies have been conducted since the last meta-analysis was published in 2013, wrote Sofie Vaengebjerg, MD, of the University of Copenhagen and colleagues.
To better identify the risk of cancer in psoriasis and psoriatic arthritis patients and to explore the impact of biologics, the researchers reviewed data from 112 studies totaling 2,053,932 patients in a study published in JAMA Dermatology.
Overall, the risk of any cancer was slightly higher in psoriasis patients (risk ratio, 1.21; 95% confidence interval, 1.11-1.33), compared with controls, with a prevalence of 4.78% and an incidence rate of 11.75 per 1,000 person-years. The most common cancer among psoriasis patients was keratinocyte cancer, with a risk ratio of 2.28 (95% CI, 1.73-3.01), a prevalence of 2.55%, and an incidence rate of 4.35 per 1,000 person-years.
Other cancers with significantly elevated risk among psoriasis patients were lymphomas (RR, 1.56; 95% CI, 1.37-1.78), lung cancer (RR, 1.26; 95% CI, 1.13-1.40), and bladder cancer (RR, 1.12; 95% CI, 1.04-1.19).
No increased risk of cancer was noted among psoriasis patients who were treated with biologics. “However, patients receiving biologic agents are selected and the results might be reliant on selection bias, and studies investigating long-term safety of these drugs are still limited,” the researchers wrote.
In addition, psoriatic arthritis was not associated with any overall increase in cancer risk, with the exception of three studies showing an increased risk for breast cancer, the researchers noted. The overall cancer prevalence for psoriatic arthritis patients was 5.74%, with an incidence rate of 6.44 per 1,000 person-years.
The study findings were limited by several factors, including the inconsistencies in study design and characteristics and the small amount of data on biologic agents and psoriatic arthritis, the researchers noted. However, the results were strengthened by the large number of patients, real-world study settings, inclusion of biologics, and analysis of cancer in psoriatic arthritis patients.
“Clinicians treating patients with psoriasis should be aware of this increased risk, especially for lymphomas, as immunogenic treatment might be associated with exacerbations,” and should be aware that more research is needed to assess cancer risk associated with biologics, they concluded.
The study received no outside funding. Lead author Dr. Vaengebjerg had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple companies, including AbbVie, Janssen, Novartis, Eli Lilly, LEO Pharma, UCB, Almirall, and Sanofi.
SOURCE: Vaengebjerg S et al. JAMA Dermatol. 2020 Feb 19. doi:10.1001/jamadermatol.2020.0024.
FROM JAMA DERMATOLOGY
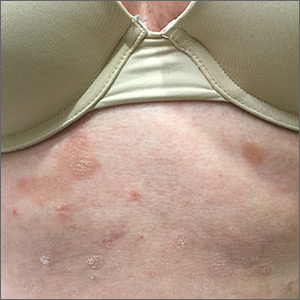
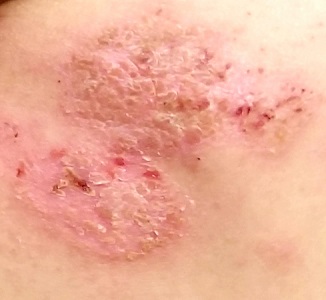
Pruritic rash on trunk
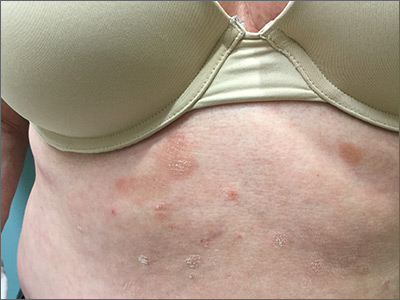
The patient underwent a skin biopsy, which was consistent with mycosis fungoides (MF) a form of cutaneous T cell lymphoma. Common MF complaints are a persistent pruritic rash that slowly progresses in size and shape. Nodules, papules, alopecia, and excoriations are often seen and can become secondarily infected. Sun spared areas are most affected. Itching and erythroderma can be quite intense, especially in Sezary Syndrome (SS)—a rare subtype of cutaneous T cell lymphoma that has a worse prognosis than localized MF.
It is a common for many patients with MF to go undiagnosed or incorrectly diagnosed for years. The differential on initial presentation can include eczema, dermatitis, psoriasis, or a drug reaction. Clues to this patient’s diagnosis were that the rash involved sun-spared areas and didn’t improve with a change to her oral medications or a course of topical steroids. Other clues that pointed to the diagnosis were that she had no history of prior scaling skin disease or psoriasis, her age (usual age of onset for MF is between 50 and 60 years), and the observation that the rash, although it looked like eczema or tinea, presented in atypical and multiple locations.
MF and SS are the most common cutaneous T cell lymphomas. Although these disorders initially involve the skin, later stages can spread to internal organs. Early MF can be difficult to diagnose on histology and can require multiple biopsies. The most accurate biopsy practice involves stopping topical medications for 4 weeks, then taking multiple biopsies of clinically different areas to confirm the diagnosis and clonality (the same cell line in more than one location). SS typically presents with a larger area of involvement and may be associated with lymphadenopathy.
The patient was seen by Dermatology. Due to the extent of the disease, which was suggestive of SS, she underwent peripheral blood flow cytometry, which revealed > 1000 Sezary cells/mcL. This confirmed the diagnosis of SS. The patient was started on photophoresis, interferon, high-potency topical steroids for the local symptoms, and bexarotene, which blocks abnormal cell growth by binding to retinoid receptors.
Photos and text courtesy of John Durkin, MD, Pigmented Lesions Clinic, University of New Mexico, and Kirill Balatsky, MS II, University of New Mexico School of Medicine.
Submitted for publication by Dr. Daniel Stulberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Hoppe RT, Kim YH. Clinical manifestations, pathologic features, and diagnosis of mycosis fungoides. UpToDate Web site. https://www.uptodate.com/contents/clinical-manifestations-pathologic-features-and-diagnosis-of-mycosis-fungoides. Updated February 27, 2019. Accessed January 29, 2020.

The patient underwent a skin biopsy, which was consistent with mycosis fungoides (MF) a form of cutaneous T cell lymphoma. Common MF complaints are a persistent pruritic rash that slowly progresses in size and shape. Nodules, papules, alopecia, and excoriations are often seen and can become secondarily infected. Sun spared areas are most affected. Itching and erythroderma can be quite intense, especially in Sezary Syndrome (SS)—a rare subtype of cutaneous T cell lymphoma that has a worse prognosis than localized MF.
It is a common for many patients with MF to go undiagnosed or incorrectly diagnosed for years. The differential on initial presentation can include eczema, dermatitis, psoriasis, or a drug reaction. Clues to this patient’s diagnosis were that the rash involved sun-spared areas and didn’t improve with a change to her oral medications or a course of topical steroids. Other clues that pointed to the diagnosis were that she had no history of prior scaling skin disease or psoriasis, her age (usual age of onset for MF is between 50 and 60 years), and the observation that the rash, although it looked like eczema or tinea, presented in atypical and multiple locations.
MF and SS are the most common cutaneous T cell lymphomas. Although these disorders initially involve the skin, later stages can spread to internal organs. Early MF can be difficult to diagnose on histology and can require multiple biopsies. The most accurate biopsy practice involves stopping topical medications for 4 weeks, then taking multiple biopsies of clinically different areas to confirm the diagnosis and clonality (the same cell line in more than one location). SS typically presents with a larger area of involvement and may be associated with lymphadenopathy.
The patient was seen by Dermatology. Due to the extent of the disease, which was suggestive of SS, she underwent peripheral blood flow cytometry, which revealed > 1000 Sezary cells/mcL. This confirmed the diagnosis of SS. The patient was started on photophoresis, interferon, high-potency topical steroids for the local symptoms, and bexarotene, which blocks abnormal cell growth by binding to retinoid receptors.
Photos and text courtesy of John Durkin, MD, Pigmented Lesions Clinic, University of New Mexico, and Kirill Balatsky, MS II, University of New Mexico School of Medicine.
Submitted for publication by Dr. Daniel Stulberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The patient underwent a skin biopsy, which was consistent with mycosis fungoides (MF) a form of cutaneous T cell lymphoma. Common MF complaints are a persistent pruritic rash that slowly progresses in size and shape. Nodules, papules, alopecia, and excoriations are often seen and can become secondarily infected. Sun spared areas are most affected. Itching and erythroderma can be quite intense, especially in Sezary Syndrome (SS)—a rare subtype of cutaneous T cell lymphoma that has a worse prognosis than localized MF.
It is a common for many patients with MF to go undiagnosed or incorrectly diagnosed for years. The differential on initial presentation can include eczema, dermatitis, psoriasis, or a drug reaction. Clues to this patient’s diagnosis were that the rash involved sun-spared areas and didn’t improve with a change to her oral medications or a course of topical steroids. Other clues that pointed to the diagnosis were that she had no history of prior scaling skin disease or psoriasis, her age (usual age of onset for MF is between 50 and 60 years), and the observation that the rash, although it looked like eczema or tinea, presented in atypical and multiple locations.
MF and SS are the most common cutaneous T cell lymphomas. Although these disorders initially involve the skin, later stages can spread to internal organs. Early MF can be difficult to diagnose on histology and can require multiple biopsies. The most accurate biopsy practice involves stopping topical medications for 4 weeks, then taking multiple biopsies of clinically different areas to confirm the diagnosis and clonality (the same cell line in more than one location). SS typically presents with a larger area of involvement and may be associated with lymphadenopathy.
The patient was seen by Dermatology. Due to the extent of the disease, which was suggestive of SS, she underwent peripheral blood flow cytometry, which revealed > 1000 Sezary cells/mcL. This confirmed the diagnosis of SS. The patient was started on photophoresis, interferon, high-potency topical steroids for the local symptoms, and bexarotene, which blocks abnormal cell growth by binding to retinoid receptors.
Photos and text courtesy of John Durkin, MD, Pigmented Lesions Clinic, University of New Mexico, and Kirill Balatsky, MS II, University of New Mexico School of Medicine.
Submitted for publication by Dr. Daniel Stulberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Hoppe RT, Kim YH. Clinical manifestations, pathologic features, and diagnosis of mycosis fungoides. UpToDate Web site. https://www.uptodate.com/contents/clinical-manifestations-pathologic-features-and-diagnosis-of-mycosis-fungoides. Updated February 27, 2019. Accessed January 29, 2020.
Hoppe RT, Kim YH. Clinical manifestations, pathologic features, and diagnosis of mycosis fungoides. UpToDate Web site. https://www.uptodate.com/contents/clinical-manifestations-pathologic-features-and-diagnosis-of-mycosis-fungoides. Updated February 27, 2019. Accessed January 29, 2020.
Rosacea: Target treatment to pathogenic pathway
LAHAINA, HAWAII – The pathophysiology of rosacea is complicated, which is why “we try to target our treatments to various areas in this pathogenic pathway” to achieve optimal results, according to Linda Stein Gold, MD, director of dermatology research at the Henry Ford Health System in Detroit.
For example, in a patient with papules and pustules, a topical or oral anti-inflammatory agent is needed “to calm that down.” If background erythema is present, separate from papules and pustules, use a topical alpha-adrenergic agonist, she advised. For telangiectasias, consider a device-based treatment, and for a phyma, a surgical approach, she recommended at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
For the background erythema of rosacea, which she described as “that pink face that’s always there,” the two alpha-adrenergic receptor agonists available, brimonidine gel 0.33%, approved by the Food and Drug Administration in 2013, and oxymetazoline cream 1%, approved in 2017, both work on the neurovascular junction, but on different receptors.
Brimonidine “kicks in very, very rapidly,” with a significant decrease in background erythema evident within 30 minutes and improvements that last over a 12-hour day, she said. It is effective over a year, but in longterm and postmarketing studies, about 20% of patients experienced exacerbation of erythema, with two peaks of redness. “One occurs at 3-6 hours,” and the other peak occurs when the drug is wearing off later in the day, Dr. Stein Gold said.
A study that sought to identify factors that might make patients more prone to this adverse effect found that “less is better” regarding brimonidine application, with an optimal application of one to three pea-sized dollops on the face, not five as instructed in the package insert. In addition, patients with more than five flushing episodes a week, particularly women, “tend to have more labile disease and [are] more likely to get that rebound erythema,” the study found.
Oxymetazoline 1% in a cream formulation has a “slightly more gentle onset of action and a more gentle offset of action,” without exacerbation of erythema and has been shown to have sustained efficacy over 52 weeks. In a yearlong safety study, there were “no new red flags and we weren’t seeing that redness at hours 3 to 6, or even when you take the patient off the drug,” she noted.
Dr. Stein Gold reported that she has served as a consultant, investigator, or speaker for Galderma, Dermira, Foamix Pharmaceuticals, Valeant, Allergan, Actavis, and Roche.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – The pathophysiology of rosacea is complicated, which is why “we try to target our treatments to various areas in this pathogenic pathway” to achieve optimal results, according to Linda Stein Gold, MD, director of dermatology research at the Henry Ford Health System in Detroit.
For example, in a patient with papules and pustules, a topical or oral anti-inflammatory agent is needed “to calm that down.” If background erythema is present, separate from papules and pustules, use a topical alpha-adrenergic agonist, she advised. For telangiectasias, consider a device-based treatment, and for a phyma, a surgical approach, she recommended at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
For the background erythema of rosacea, which she described as “that pink face that’s always there,” the two alpha-adrenergic receptor agonists available, brimonidine gel 0.33%, approved by the Food and Drug Administration in 2013, and oxymetazoline cream 1%, approved in 2017, both work on the neurovascular junction, but on different receptors.
Brimonidine “kicks in very, very rapidly,” with a significant decrease in background erythema evident within 30 minutes and improvements that last over a 12-hour day, she said. It is effective over a year, but in longterm and postmarketing studies, about 20% of patients experienced exacerbation of erythema, with two peaks of redness. “One occurs at 3-6 hours,” and the other peak occurs when the drug is wearing off later in the day, Dr. Stein Gold said.
A study that sought to identify factors that might make patients more prone to this adverse effect found that “less is better” regarding brimonidine application, with an optimal application of one to three pea-sized dollops on the face, not five as instructed in the package insert. In addition, patients with more than five flushing episodes a week, particularly women, “tend to have more labile disease and [are] more likely to get that rebound erythema,” the study found.
Oxymetazoline 1% in a cream formulation has a “slightly more gentle onset of action and a more gentle offset of action,” without exacerbation of erythema and has been shown to have sustained efficacy over 52 weeks. In a yearlong safety study, there were “no new red flags and we weren’t seeing that redness at hours 3 to 6, or even when you take the patient off the drug,” she noted.
Dr. Stein Gold reported that she has served as a consultant, investigator, or speaker for Galderma, Dermira, Foamix Pharmaceuticals, Valeant, Allergan, Actavis, and Roche.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – The pathophysiology of rosacea is complicated, which is why “we try to target our treatments to various areas in this pathogenic pathway” to achieve optimal results, according to Linda Stein Gold, MD, director of dermatology research at the Henry Ford Health System in Detroit.
For example, in a patient with papules and pustules, a topical or oral anti-inflammatory agent is needed “to calm that down.” If background erythema is present, separate from papules and pustules, use a topical alpha-adrenergic agonist, she advised. For telangiectasias, consider a device-based treatment, and for a phyma, a surgical approach, she recommended at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
For the background erythema of rosacea, which she described as “that pink face that’s always there,” the two alpha-adrenergic receptor agonists available, brimonidine gel 0.33%, approved by the Food and Drug Administration in 2013, and oxymetazoline cream 1%, approved in 2017, both work on the neurovascular junction, but on different receptors.
Brimonidine “kicks in very, very rapidly,” with a significant decrease in background erythema evident within 30 minutes and improvements that last over a 12-hour day, she said. It is effective over a year, but in longterm and postmarketing studies, about 20% of patients experienced exacerbation of erythema, with two peaks of redness. “One occurs at 3-6 hours,” and the other peak occurs when the drug is wearing off later in the day, Dr. Stein Gold said.
A study that sought to identify factors that might make patients more prone to this adverse effect found that “less is better” regarding brimonidine application, with an optimal application of one to three pea-sized dollops on the face, not five as instructed in the package insert. In addition, patients with more than five flushing episodes a week, particularly women, “tend to have more labile disease and [are] more likely to get that rebound erythema,” the study found.
Oxymetazoline 1% in a cream formulation has a “slightly more gentle onset of action and a more gentle offset of action,” without exacerbation of erythema and has been shown to have sustained efficacy over 52 weeks. In a yearlong safety study, there were “no new red flags and we weren’t seeing that redness at hours 3 to 6, or even when you take the patient off the drug,” she noted.
Dr. Stein Gold reported that she has served as a consultant, investigator, or speaker for Galderma, Dermira, Foamix Pharmaceuticals, Valeant, Allergan, Actavis, and Roche.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
Dupilumab for severe AD: Expert advocates continuous treatment
LAHAINA, HAWAII – rather than treatment on an as-needed basis, Andrew Blauvelt, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I view atopic dermatitis as a chronic disease requiring chronic treatment. So be very careful about stopping. We know that if you start and stop biologics you’re going to be far more prone to develop antidrug antibodies resulting in drug resistance than with continual dosing,” said Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
He said dupilumab (Dupixent) seldom induces disease remission as defined by clear skin while off all drugs for at least 1 year, although he has a few patients who seem to be exceptions. Yet clearly dupilumab doesn’t change an individual’s predisposing genetics or environmental allergen exposure pattern, so it’s best to think of it as a treatment for the long haul.
Dr. Blauvelt considers dupilumab far and away the best medication for treatment of adults and teenagers whose atopic dermatitis (AD) is uncontrolled with topical therapy. Payers often balk at authorizing dupilumab unless a patient has first undergone an unsuccessful trial of cyclosporine or methotrexate, which are far less expensive. But that’s not what the expert consensus guidelines recommend (Ann Allergy Asthma Immunol. 2018 Jan;120[1]:10-22.e2).
“The guidelines don’t suggest that failure on methotrexate or cyclosporine should be a prerequisite for dupilumab. So if you’re having problems with an insurance company and you really want to use dupilumab, you can point to this paper and say, ‘Look, the experts do not recommend step therapy, we can go directly to dupilumab.’ And the dupilumab label says simply that failure of topical therapy is required before being allowed to use dupilumab. So both the label and the experts say you don’t have to go through a bunch of steps in order to get to what I consider the very best drug for our patients,” he explained.
Both cyclosporine and methotrexate are far more broadly immunosuppressive and hence less safe than dupilumab. Both require laboratory monitoring. In contrast, blood work isn’t required in patients on dupilumab; in fact, Dr. Blauvelt considers it an unwise use of resources. Nor is tuberculosis testing advised prior to starting dupilumab.
When he can’t get authorization for dupilumab, Dr. Blauvelt’s go-to drug is methotrexate at 15-25 mg/week. It’s not as effective as cyclosporine for rapid clearing, but it’s safer for long-term use.
“Methotrexate is the devil we know – we know how to use it, and we know how to monitor for it,” he commented, adding that he reserves cyclosporine for a maximum of a month or 2 of acute crisis management, or as a bridge in getting patients off of systemic corticosteroids.
Set realistic efficacy expectations
Dermatologists who prescribe the newest biologics for psoriasis are accustomed to routinely seeing PASI 90 responses and even complete disease clearing. However, AD is a more challenging disease. In the landmark dupilumab phase 3 randomized trials, roughly two-thirds of patients achieved an Eczema Area and Severity Index (EASI) 75 response, with a mean 80% improvement in EASI symptom scores over baseline. Roughly 20% of dupilumab-treated adults with AD achieve disease clearance, and a similar percentage become almost clear. The improvements are durable in long-term follow-up studies.
“Dupilumab doesn’t get a lot of people to zero. They’re not going to be completely clearing their eczema. So they shouldn’t be freaking out if they still have eczema. What they can expect is diminution of the disease to much lower levels,” Dr. Blauvelt said.
The marked improvement in quality of life that occurs with dupilumab therapy isn’t adequately captured by EASI scores. “In my experience, more than 80%-90% of patients are happy on this drug,” Dr. Blauvelt said.
Conference codirector Linda Stein Gold, MD, agreed, commenting that she has found dupilumab to be “absolutely life altering” for her patients with severe AD.
“They know they still have AD, but now they can go whole days without thinking about it,” said Dr. Stein Gold, director of dermatology research and head of the division of dermatology at the Henry Ford Health System in Detroit.
Dr. Blauvelt noted that most of his patients on dupilumab remain on topical therapy, typically with triamcinolone on the body and hydrocortisone on the face. What he terms “miniflares” in patients on dupilumab are not at all unusual, but they’re readily manageable.
“Flares that used to last for weeks now last for a day or 2, maybe 3, and then it’s back to normal in patients on dupilumab,” Dr. Blauvelt said.
Safety
Dupilumab is a targeted inhibitor of interleukins-4 and -13, cytokines involved in allergy-mediated inflammation and the control of parasitic infections, but which have no bearing on control of bacterial or viral infections or malignancies. Indeed, the randomized trials have demonstrated that the incidence of skin infections is actually lower with dupilumab than with placebo.
“You’re improving the skin barrier so much that they’re not going to be getting staph or herpes simplex,” he explained.
The main side effect consists of dupilumab-associated eye issues. These occur in up to 20% of treated patients and encompass a spectrum ranging from dry eye to nonallergic conjunctivitis, inflammation of the eyelid, and keratitis. The mechanism is unknown. The condition is not infectious and doesn’t affect vision. Intriguingly, it doesn’t occur in patients with asthma, a disease for which dupilumab is also approved.
“Ask about eye issues at every office visit,” the dermatologist urged.
He sends all of his AD patients with dupilumab-associated eye issues to a single trusted local ophthalmologist and lets him manage the condition, which is generally mild to moderate. Eye issues have resulted in discontinuation of dupilumab in only 2 of the roughly 150 AD patients Dr. Blauvelt has placed on the biologic. The ophthalmologist generally relies upon lubricating eye drops and a couple of weeks of steroid eye drops or, in some cases, topical cyclosporine 0.05% ophthalmic emulsion, followed by episodic use of the steroid eye drops on an as-needed basis.
Residual facial disease in AD patients on dupilumab can be caused by a variety of causes, including breakthrough AD, rosacea, allergic contact dermatitis, steroid withdrawal, or photosensitivity, with Demodex thought to play a role in some cases.
Dr. Blauvelt reported serving as a scientific adviser to and paid clinical trial investigator for several dozen pharmaceutical companies. SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – rather than treatment on an as-needed basis, Andrew Blauvelt, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I view atopic dermatitis as a chronic disease requiring chronic treatment. So be very careful about stopping. We know that if you start and stop biologics you’re going to be far more prone to develop antidrug antibodies resulting in drug resistance than with continual dosing,” said Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
He said dupilumab (Dupixent) seldom induces disease remission as defined by clear skin while off all drugs for at least 1 year, although he has a few patients who seem to be exceptions. Yet clearly dupilumab doesn’t change an individual’s predisposing genetics or environmental allergen exposure pattern, so it’s best to think of it as a treatment for the long haul.
Dr. Blauvelt considers dupilumab far and away the best medication for treatment of adults and teenagers whose atopic dermatitis (AD) is uncontrolled with topical therapy. Payers often balk at authorizing dupilumab unless a patient has first undergone an unsuccessful trial of cyclosporine or methotrexate, which are far less expensive. But that’s not what the expert consensus guidelines recommend (Ann Allergy Asthma Immunol. 2018 Jan;120[1]:10-22.e2).
“The guidelines don’t suggest that failure on methotrexate or cyclosporine should be a prerequisite for dupilumab. So if you’re having problems with an insurance company and you really want to use dupilumab, you can point to this paper and say, ‘Look, the experts do not recommend step therapy, we can go directly to dupilumab.’ And the dupilumab label says simply that failure of topical therapy is required before being allowed to use dupilumab. So both the label and the experts say you don’t have to go through a bunch of steps in order to get to what I consider the very best drug for our patients,” he explained.
Both cyclosporine and methotrexate are far more broadly immunosuppressive and hence less safe than dupilumab. Both require laboratory monitoring. In contrast, blood work isn’t required in patients on dupilumab; in fact, Dr. Blauvelt considers it an unwise use of resources. Nor is tuberculosis testing advised prior to starting dupilumab.
When he can’t get authorization for dupilumab, Dr. Blauvelt’s go-to drug is methotrexate at 15-25 mg/week. It’s not as effective as cyclosporine for rapid clearing, but it’s safer for long-term use.
“Methotrexate is the devil we know – we know how to use it, and we know how to monitor for it,” he commented, adding that he reserves cyclosporine for a maximum of a month or 2 of acute crisis management, or as a bridge in getting patients off of systemic corticosteroids.
Set realistic efficacy expectations
Dermatologists who prescribe the newest biologics for psoriasis are accustomed to routinely seeing PASI 90 responses and even complete disease clearing. However, AD is a more challenging disease. In the landmark dupilumab phase 3 randomized trials, roughly two-thirds of patients achieved an Eczema Area and Severity Index (EASI) 75 response, with a mean 80% improvement in EASI symptom scores over baseline. Roughly 20% of dupilumab-treated adults with AD achieve disease clearance, and a similar percentage become almost clear. The improvements are durable in long-term follow-up studies.
“Dupilumab doesn’t get a lot of people to zero. They’re not going to be completely clearing their eczema. So they shouldn’t be freaking out if they still have eczema. What they can expect is diminution of the disease to much lower levels,” Dr. Blauvelt said.
The marked improvement in quality of life that occurs with dupilumab therapy isn’t adequately captured by EASI scores. “In my experience, more than 80%-90% of patients are happy on this drug,” Dr. Blauvelt said.
Conference codirector Linda Stein Gold, MD, agreed, commenting that she has found dupilumab to be “absolutely life altering” for her patients with severe AD.
“They know they still have AD, but now they can go whole days without thinking about it,” said Dr. Stein Gold, director of dermatology research and head of the division of dermatology at the Henry Ford Health System in Detroit.
Dr. Blauvelt noted that most of his patients on dupilumab remain on topical therapy, typically with triamcinolone on the body and hydrocortisone on the face. What he terms “miniflares” in patients on dupilumab are not at all unusual, but they’re readily manageable.
“Flares that used to last for weeks now last for a day or 2, maybe 3, and then it’s back to normal in patients on dupilumab,” Dr. Blauvelt said.
Safety
Dupilumab is a targeted inhibitor of interleukins-4 and -13, cytokines involved in allergy-mediated inflammation and the control of parasitic infections, but which have no bearing on control of bacterial or viral infections or malignancies. Indeed, the randomized trials have demonstrated that the incidence of skin infections is actually lower with dupilumab than with placebo.
“You’re improving the skin barrier so much that they’re not going to be getting staph or herpes simplex,” he explained.
The main side effect consists of dupilumab-associated eye issues. These occur in up to 20% of treated patients and encompass a spectrum ranging from dry eye to nonallergic conjunctivitis, inflammation of the eyelid, and keratitis. The mechanism is unknown. The condition is not infectious and doesn’t affect vision. Intriguingly, it doesn’t occur in patients with asthma, a disease for which dupilumab is also approved.
“Ask about eye issues at every office visit,” the dermatologist urged.
He sends all of his AD patients with dupilumab-associated eye issues to a single trusted local ophthalmologist and lets him manage the condition, which is generally mild to moderate. Eye issues have resulted in discontinuation of dupilumab in only 2 of the roughly 150 AD patients Dr. Blauvelt has placed on the biologic. The ophthalmologist generally relies upon lubricating eye drops and a couple of weeks of steroid eye drops or, in some cases, topical cyclosporine 0.05% ophthalmic emulsion, followed by episodic use of the steroid eye drops on an as-needed basis.
Residual facial disease in AD patients on dupilumab can be caused by a variety of causes, including breakthrough AD, rosacea, allergic contact dermatitis, steroid withdrawal, or photosensitivity, with Demodex thought to play a role in some cases.
Dr. Blauvelt reported serving as a scientific adviser to and paid clinical trial investigator for several dozen pharmaceutical companies. SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – rather than treatment on an as-needed basis, Andrew Blauvelt, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I view atopic dermatitis as a chronic disease requiring chronic treatment. So be very careful about stopping. We know that if you start and stop biologics you’re going to be far more prone to develop antidrug antibodies resulting in drug resistance than with continual dosing,” said Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
He said dupilumab (Dupixent) seldom induces disease remission as defined by clear skin while off all drugs for at least 1 year, although he has a few patients who seem to be exceptions. Yet clearly dupilumab doesn’t change an individual’s predisposing genetics or environmental allergen exposure pattern, so it’s best to think of it as a treatment for the long haul.
Dr. Blauvelt considers dupilumab far and away the best medication for treatment of adults and teenagers whose atopic dermatitis (AD) is uncontrolled with topical therapy. Payers often balk at authorizing dupilumab unless a patient has first undergone an unsuccessful trial of cyclosporine or methotrexate, which are far less expensive. But that’s not what the expert consensus guidelines recommend (Ann Allergy Asthma Immunol. 2018 Jan;120[1]:10-22.e2).
“The guidelines don’t suggest that failure on methotrexate or cyclosporine should be a prerequisite for dupilumab. So if you’re having problems with an insurance company and you really want to use dupilumab, you can point to this paper and say, ‘Look, the experts do not recommend step therapy, we can go directly to dupilumab.’ And the dupilumab label says simply that failure of topical therapy is required before being allowed to use dupilumab. So both the label and the experts say you don’t have to go through a bunch of steps in order to get to what I consider the very best drug for our patients,” he explained.
Both cyclosporine and methotrexate are far more broadly immunosuppressive and hence less safe than dupilumab. Both require laboratory monitoring. In contrast, blood work isn’t required in patients on dupilumab; in fact, Dr. Blauvelt considers it an unwise use of resources. Nor is tuberculosis testing advised prior to starting dupilumab.
When he can’t get authorization for dupilumab, Dr. Blauvelt’s go-to drug is methotrexate at 15-25 mg/week. It’s not as effective as cyclosporine for rapid clearing, but it’s safer for long-term use.
“Methotrexate is the devil we know – we know how to use it, and we know how to monitor for it,” he commented, adding that he reserves cyclosporine for a maximum of a month or 2 of acute crisis management, or as a bridge in getting patients off of systemic corticosteroids.
Set realistic efficacy expectations
Dermatologists who prescribe the newest biologics for psoriasis are accustomed to routinely seeing PASI 90 responses and even complete disease clearing. However, AD is a more challenging disease. In the landmark dupilumab phase 3 randomized trials, roughly two-thirds of patients achieved an Eczema Area and Severity Index (EASI) 75 response, with a mean 80% improvement in EASI symptom scores over baseline. Roughly 20% of dupilumab-treated adults with AD achieve disease clearance, and a similar percentage become almost clear. The improvements are durable in long-term follow-up studies.
“Dupilumab doesn’t get a lot of people to zero. They’re not going to be completely clearing their eczema. So they shouldn’t be freaking out if they still have eczema. What they can expect is diminution of the disease to much lower levels,” Dr. Blauvelt said.
The marked improvement in quality of life that occurs with dupilumab therapy isn’t adequately captured by EASI scores. “In my experience, more than 80%-90% of patients are happy on this drug,” Dr. Blauvelt said.
Conference codirector Linda Stein Gold, MD, agreed, commenting that she has found dupilumab to be “absolutely life altering” for her patients with severe AD.
“They know they still have AD, but now they can go whole days without thinking about it,” said Dr. Stein Gold, director of dermatology research and head of the division of dermatology at the Henry Ford Health System in Detroit.
Dr. Blauvelt noted that most of his patients on dupilumab remain on topical therapy, typically with triamcinolone on the body and hydrocortisone on the face. What he terms “miniflares” in patients on dupilumab are not at all unusual, but they’re readily manageable.
“Flares that used to last for weeks now last for a day or 2, maybe 3, and then it’s back to normal in patients on dupilumab,” Dr. Blauvelt said.
Safety
Dupilumab is a targeted inhibitor of interleukins-4 and -13, cytokines involved in allergy-mediated inflammation and the control of parasitic infections, but which have no bearing on control of bacterial or viral infections or malignancies. Indeed, the randomized trials have demonstrated that the incidence of skin infections is actually lower with dupilumab than with placebo.
“You’re improving the skin barrier so much that they’re not going to be getting staph or herpes simplex,” he explained.
The main side effect consists of dupilumab-associated eye issues. These occur in up to 20% of treated patients and encompass a spectrum ranging from dry eye to nonallergic conjunctivitis, inflammation of the eyelid, and keratitis. The mechanism is unknown. The condition is not infectious and doesn’t affect vision. Intriguingly, it doesn’t occur in patients with asthma, a disease for which dupilumab is also approved.
“Ask about eye issues at every office visit,” the dermatologist urged.
He sends all of his AD patients with dupilumab-associated eye issues to a single trusted local ophthalmologist and lets him manage the condition, which is generally mild to moderate. Eye issues have resulted in discontinuation of dupilumab in only 2 of the roughly 150 AD patients Dr. Blauvelt has placed on the biologic. The ophthalmologist generally relies upon lubricating eye drops and a couple of weeks of steroid eye drops or, in some cases, topical cyclosporine 0.05% ophthalmic emulsion, followed by episodic use of the steroid eye drops on an as-needed basis.
Residual facial disease in AD patients on dupilumab can be caused by a variety of causes, including breakthrough AD, rosacea, allergic contact dermatitis, steroid withdrawal, or photosensitivity, with Demodex thought to play a role in some cases.
Dr. Blauvelt reported serving as a scientific adviser to and paid clinical trial investigator for several dozen pharmaceutical companies. SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT OPINION FROM SDEF HAWAII DERMATOLOGY SEMINAR
Toenail thickening
The FP suspected onychauxis, more commonly called hypertrophic nail. The patient’s toenail had the characteristic features of onychauxis, which include discoloration (usually yellow or brown) and a dull appearance. Often, there is an increased curvature or deviation of the nail and a “clam shell” appearance with transverse lines or a lamellar pattern like a ram’s horn. This is in contrast to the longitudinal lines and furrows that one would see with brittle nails associated with old age or the longitudinal melanonychia (hyperpigmented lines) seen in melanoma. Trauma to the nailbed, including trauma from ill-fitting shoes, is the most common cause of onychauxis.
Although nail discoloration and thickening raise the concern for onychomycosis, not all thickened and discolored nails are due to fungal infection. In this case, the thickening of the nail itself (as opposed to the subungal hyperkeratosis typical of onychomycosis) and a lack of improvement with antifungal treatment prompted the FP to consider other causes of nail dystrophy besides onychomycosis.
Nail trimming and filing can minimize discomfort and limit nail margin trauma caused by the nail’s abnormal shape. If this does not provide relief, the curative treatment for onychauxis is toenail removal and matrix ablation. In this case, the patient chose to defer nail removal and resection of the matrix. She said she would consider this treatment option if her nail became more bothersome.
Photos and text courtesy of Sabrina Gill, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Abdullah L, Abba O. Common nail changes and disorders in older people: Diagnosis and management. Can Fam Physician. 2011;57:173–181.

The FP suspected onychauxis, more commonly called hypertrophic nail. The patient’s toenail had the characteristic features of onychauxis, which include discoloration (usually yellow or brown) and a dull appearance. Often, there is an increased curvature or deviation of the nail and a “clam shell” appearance with transverse lines or a lamellar pattern like a ram’s horn. This is in contrast to the longitudinal lines and furrows that one would see with brittle nails associated with old age or the longitudinal melanonychia (hyperpigmented lines) seen in melanoma. Trauma to the nailbed, including trauma from ill-fitting shoes, is the most common cause of onychauxis.
Although nail discoloration and thickening raise the concern for onychomycosis, not all thickened and discolored nails are due to fungal infection. In this case, the thickening of the nail itself (as opposed to the subungal hyperkeratosis typical of onychomycosis) and a lack of improvement with antifungal treatment prompted the FP to consider other causes of nail dystrophy besides onychomycosis.
Nail trimming and filing can minimize discomfort and limit nail margin trauma caused by the nail’s abnormal shape. If this does not provide relief, the curative treatment for onychauxis is toenail removal and matrix ablation. In this case, the patient chose to defer nail removal and resection of the matrix. She said she would consider this treatment option if her nail became more bothersome.
Photos and text courtesy of Sabrina Gill, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The FP suspected onychauxis, more commonly called hypertrophic nail. The patient’s toenail had the characteristic features of onychauxis, which include discoloration (usually yellow or brown) and a dull appearance. Often, there is an increased curvature or deviation of the nail and a “clam shell” appearance with transverse lines or a lamellar pattern like a ram’s horn. This is in contrast to the longitudinal lines and furrows that one would see with brittle nails associated with old age or the longitudinal melanonychia (hyperpigmented lines) seen in melanoma. Trauma to the nailbed, including trauma from ill-fitting shoes, is the most common cause of onychauxis.
Although nail discoloration and thickening raise the concern for onychomycosis, not all thickened and discolored nails are due to fungal infection. In this case, the thickening of the nail itself (as opposed to the subungal hyperkeratosis typical of onychomycosis) and a lack of improvement with antifungal treatment prompted the FP to consider other causes of nail dystrophy besides onychomycosis.
Nail trimming and filing can minimize discomfort and limit nail margin trauma caused by the nail’s abnormal shape. If this does not provide relief, the curative treatment for onychauxis is toenail removal and matrix ablation. In this case, the patient chose to defer nail removal and resection of the matrix. She said she would consider this treatment option if her nail became more bothersome.
Photos and text courtesy of Sabrina Gill, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Abdullah L, Abba O. Common nail changes and disorders in older people: Diagnosis and management. Can Fam Physician. 2011;57:173–181.
Abdullah L, Abba O. Common nail changes and disorders in older people: Diagnosis and management. Can Fam Physician. 2011;57:173–181.
Acne treatment may vary based on race, gender, insurance
based on findings from a retrospective, cohort study of 29,928 individuals with acne.
“Our findings suggest the presence of racial/ethnic, sex, and insurance-based disparities in health care use and treatment for acne and raise particular concern for undertreatment among racial/ethnic minority and female patients,” John S. Barbieri, MD, a dermatology research fellow at the University of Pennsylvania, Philadelphia, and colleagues wrote in a study published in JAMA Dermatology.
Data from previous studies have suggested racial disparities in the management of several dermatologic conditions, including atopic dermatitis and psoriasis, but associations between social demographics and prescribing patterns have not been well studied for acne treatment, the authors noted.
For the current study, the researchers used deidentified data from the Optum electronic health record from Jan. 1, 2007 to June 30, 2017. In all, 29,928 patients aged 15-35 years and who were being treated for acne were included in the study. Of that total, 64% were women, 8% were non-Hispanic black and 68% were white, with the remaining patients grouped as non-Hispanic Asian, Hispanic, or other.
Non-Hispanic black patients were significantly more likely to be seen by a dermatologist, compared with non-Hispanic white patients, who were designated as the reference (odds ratio, 1.20). However, the black patients were less likely to receive prescriptions for any acne medication (incidence rate ratio, 0.89).
Non-Hispanic black patients were more likely than non-Hispanic white patients to be prescribed topical retinoids or topical antibiotics (OR, 1.25 and 1.35, respectively). They were also were less likely than their white counterparts to be prescribed oral antibiotics, spironolactone, and isotretinoin (OR, 0.80, 0.68, and 0.39, respectively).
Overall, men were more than twice as likely as women to receive prescriptions for isotretinoin (OR, 2.44). They were also more likely to receive prescriptions for the other treatments, but the differences were not as high as those for isotretinoin.
In addition, patients with Medicaid insurance were significantly less likely than those with commercial insurance (the reference) to see a dermatologist (OR, 0.46). Medicaid patients also were less likely to be prescribed topical retinoids, oral antibiotics, spironolactone, or isotretinoin (OR, 0.82, 0.87, 0.50, and 0.43, respectively).
The study findings were limited by several factors, among them, the use of automated pharmacy data without confirmation that patients had picked up the medications they had been prescribed, the researchers said. The study also lacked data on acne severity, clinical outcomes, and the use of over-the-counter acne treatments.
“Further study is needed to confirm our findings, provide understanding of the reasons for these potential disparities, and develop strategies to ensure equitable care for patients with acne,” the researchers concluded.
The study was supported in part by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health, and by a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Barbieri had no financial conflicts to disclose. One of the study coauthors disclosed relationships with Pfizer, Eli Lilly, and Novartis.
SOURCE: Barbieri JS et al. JAMA Dermatol. 2020 Feb 5. doi: 10.1001/jamadermatol.2019.4818.
based on findings from a retrospective, cohort study of 29,928 individuals with acne.
“Our findings suggest the presence of racial/ethnic, sex, and insurance-based disparities in health care use and treatment for acne and raise particular concern for undertreatment among racial/ethnic minority and female patients,” John S. Barbieri, MD, a dermatology research fellow at the University of Pennsylvania, Philadelphia, and colleagues wrote in a study published in JAMA Dermatology.
Data from previous studies have suggested racial disparities in the management of several dermatologic conditions, including atopic dermatitis and psoriasis, but associations between social demographics and prescribing patterns have not been well studied for acne treatment, the authors noted.
For the current study, the researchers used deidentified data from the Optum electronic health record from Jan. 1, 2007 to June 30, 2017. In all, 29,928 patients aged 15-35 years and who were being treated for acne were included in the study. Of that total, 64% were women, 8% were non-Hispanic black and 68% were white, with the remaining patients grouped as non-Hispanic Asian, Hispanic, or other.
Non-Hispanic black patients were significantly more likely to be seen by a dermatologist, compared with non-Hispanic white patients, who were designated as the reference (odds ratio, 1.20). However, the black patients were less likely to receive prescriptions for any acne medication (incidence rate ratio, 0.89).
Non-Hispanic black patients were more likely than non-Hispanic white patients to be prescribed topical retinoids or topical antibiotics (OR, 1.25 and 1.35, respectively). They were also were less likely than their white counterparts to be prescribed oral antibiotics, spironolactone, and isotretinoin (OR, 0.80, 0.68, and 0.39, respectively).
Overall, men were more than twice as likely as women to receive prescriptions for isotretinoin (OR, 2.44). They were also more likely to receive prescriptions for the other treatments, but the differences were not as high as those for isotretinoin.
In addition, patients with Medicaid insurance were significantly less likely than those with commercial insurance (the reference) to see a dermatologist (OR, 0.46). Medicaid patients also were less likely to be prescribed topical retinoids, oral antibiotics, spironolactone, or isotretinoin (OR, 0.82, 0.87, 0.50, and 0.43, respectively).
The study findings were limited by several factors, among them, the use of automated pharmacy data without confirmation that patients had picked up the medications they had been prescribed, the researchers said. The study also lacked data on acne severity, clinical outcomes, and the use of over-the-counter acne treatments.
“Further study is needed to confirm our findings, provide understanding of the reasons for these potential disparities, and develop strategies to ensure equitable care for patients with acne,” the researchers concluded.
The study was supported in part by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health, and by a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Barbieri had no financial conflicts to disclose. One of the study coauthors disclosed relationships with Pfizer, Eli Lilly, and Novartis.
SOURCE: Barbieri JS et al. JAMA Dermatol. 2020 Feb 5. doi: 10.1001/jamadermatol.2019.4818.
based on findings from a retrospective, cohort study of 29,928 individuals with acne.
“Our findings suggest the presence of racial/ethnic, sex, and insurance-based disparities in health care use and treatment for acne and raise particular concern for undertreatment among racial/ethnic minority and female patients,” John S. Barbieri, MD, a dermatology research fellow at the University of Pennsylvania, Philadelphia, and colleagues wrote in a study published in JAMA Dermatology.
Data from previous studies have suggested racial disparities in the management of several dermatologic conditions, including atopic dermatitis and psoriasis, but associations between social demographics and prescribing patterns have not been well studied for acne treatment, the authors noted.
For the current study, the researchers used deidentified data from the Optum electronic health record from Jan. 1, 2007 to June 30, 2017. In all, 29,928 patients aged 15-35 years and who were being treated for acne were included in the study. Of that total, 64% were women, 8% were non-Hispanic black and 68% were white, with the remaining patients grouped as non-Hispanic Asian, Hispanic, or other.
Non-Hispanic black patients were significantly more likely to be seen by a dermatologist, compared with non-Hispanic white patients, who were designated as the reference (odds ratio, 1.20). However, the black patients were less likely to receive prescriptions for any acne medication (incidence rate ratio, 0.89).
Non-Hispanic black patients were more likely than non-Hispanic white patients to be prescribed topical retinoids or topical antibiotics (OR, 1.25 and 1.35, respectively). They were also were less likely than their white counterparts to be prescribed oral antibiotics, spironolactone, and isotretinoin (OR, 0.80, 0.68, and 0.39, respectively).
Overall, men were more than twice as likely as women to receive prescriptions for isotretinoin (OR, 2.44). They were also more likely to receive prescriptions for the other treatments, but the differences were not as high as those for isotretinoin.
In addition, patients with Medicaid insurance were significantly less likely than those with commercial insurance (the reference) to see a dermatologist (OR, 0.46). Medicaid patients also were less likely to be prescribed topical retinoids, oral antibiotics, spironolactone, or isotretinoin (OR, 0.82, 0.87, 0.50, and 0.43, respectively).
The study findings were limited by several factors, among them, the use of automated pharmacy data without confirmation that patients had picked up the medications they had been prescribed, the researchers said. The study also lacked data on acne severity, clinical outcomes, and the use of over-the-counter acne treatments.
“Further study is needed to confirm our findings, provide understanding of the reasons for these potential disparities, and develop strategies to ensure equitable care for patients with acne,” the researchers concluded.
The study was supported in part by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health, and by a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Barbieri had no financial conflicts to disclose. One of the study coauthors disclosed relationships with Pfizer, Eli Lilly, and Novartis.
SOURCE: Barbieri JS et al. JAMA Dermatol. 2020 Feb 5. doi: 10.1001/jamadermatol.2019.4818.
FROM JAMA DERMATOLOGY
Baby’s Got Back Rash
ANSWER
The correct answer is psoriasis vulgaris (choice “a”).
DISCUSSION
At least 30% of patients with psoriasis have a family history of the disease—a meaningful clue in developing a differential. Besides asking about the history, always look for corroborating signs in areas where the disease is commonly seen (eg, the fingernails). In this case, further corroboration was provided by the history of illness at the time of the rash’s onset; what was initially strep-driven guttate psoriasis morphed into full-blown psoriasis vulgaris.
The heavy scales, with their salmon-pink base, tipped the scales in favor of psoriasis as the diagnosis. The pinpoint bleeding (known as the Auspitz sign), although not pathognomic for psoriasis, is certainly suggestive of it.
In adults, these findings would probably have been sufficient to settle on psoriasis. But before labeling a young child with a serious, lifelong diagnosis, it was necessary to be sure. For one thing, advanced psoriasis is very unusual in children as young as this patient, and for another, treatment would likely be problematic. Fortunately for clarity’s sake, the biopsy was consistent with psoriasis and inconsistent with the other items in the differential.
TREATMENT
The patient was prescribed a topical steroid cream to apply every other day, alternating with vitamin D–derived ointment. In addition, he was advised to increase his exposure to natural sunlight. Phototherapy with narrow-band ultraviolet light B would be a superior option, but his family lives too far from the clinic to make 3 roundtrips per week for such treatment.
If these measures fail, a biologic agent may be appropriate. Unfortunately, the patient’s insurance carrier requires the failure of several other modalities before it will approve use of such therapy.
ANSWER
The correct answer is psoriasis vulgaris (choice “a”).
DISCUSSION
At least 30% of patients with psoriasis have a family history of the disease—a meaningful clue in developing a differential. Besides asking about the history, always look for corroborating signs in areas where the disease is commonly seen (eg, the fingernails). In this case, further corroboration was provided by the history of illness at the time of the rash’s onset; what was initially strep-driven guttate psoriasis morphed into full-blown psoriasis vulgaris.
The heavy scales, with their salmon-pink base, tipped the scales in favor of psoriasis as the diagnosis. The pinpoint bleeding (known as the Auspitz sign), although not pathognomic for psoriasis, is certainly suggestive of it.
In adults, these findings would probably have been sufficient to settle on psoriasis. But before labeling a young child with a serious, lifelong diagnosis, it was necessary to be sure. For one thing, advanced psoriasis is very unusual in children as young as this patient, and for another, treatment would likely be problematic. Fortunately for clarity’s sake, the biopsy was consistent with psoriasis and inconsistent with the other items in the differential.
TREATMENT
The patient was prescribed a topical steroid cream to apply every other day, alternating with vitamin D–derived ointment. In addition, he was advised to increase his exposure to natural sunlight. Phototherapy with narrow-band ultraviolet light B would be a superior option, but his family lives too far from the clinic to make 3 roundtrips per week for such treatment.
If these measures fail, a biologic agent may be appropriate. Unfortunately, the patient’s insurance carrier requires the failure of several other modalities before it will approve use of such therapy.
ANSWER
The correct answer is psoriasis vulgaris (choice “a”).
DISCUSSION
At least 30% of patients with psoriasis have a family history of the disease—a meaningful clue in developing a differential. Besides asking about the history, always look for corroborating signs in areas where the disease is commonly seen (eg, the fingernails). In this case, further corroboration was provided by the history of illness at the time of the rash’s onset; what was initially strep-driven guttate psoriasis morphed into full-blown psoriasis vulgaris.
The heavy scales, with their salmon-pink base, tipped the scales in favor of psoriasis as the diagnosis. The pinpoint bleeding (known as the Auspitz sign), although not pathognomic for psoriasis, is certainly suggestive of it.
In adults, these findings would probably have been sufficient to settle on psoriasis. But before labeling a young child with a serious, lifelong diagnosis, it was necessary to be sure. For one thing, advanced psoriasis is very unusual in children as young as this patient, and for another, treatment would likely be problematic. Fortunately for clarity’s sake, the biopsy was consistent with psoriasis and inconsistent with the other items in the differential.
TREATMENT
The patient was prescribed a topical steroid cream to apply every other day, alternating with vitamin D–derived ointment. In addition, he was advised to increase his exposure to natural sunlight. Phototherapy with narrow-band ultraviolet light B would be a superior option, but his family lives too far from the clinic to make 3 roundtrips per week for such treatment.
If these measures fail, a biologic agent may be appropriate. Unfortunately, the patient’s insurance carrier requires the failure of several other modalities before it will approve use of such therapy.
Several months ago, a rash of numerous small, red, scaly papules and patches manifested on this 3-year-old boy’s back and shoulders. At the time, he had been ill for about a week, and his primary care provider diagnosed chickenpox—even though the child had been immunized.
Although the patient’s health soon improved, the appearance of the rash worsened. Treatment with various products—including calamine lotion, OTC tolnaftate and miconazole, and a 2-week course of oral antibiotics—was of no help. Finally, the patient was referred to dermatology.
Family history is positive for psoriasis. However, the parents are quick to note that the boy’s rash appears far different from that of affected family members, and previous providers have dismissed this diagnosis from the differential. There is no family or personal history of atopy.
Examination reveals a dense papulosquamous rash mainly confined to the child’s back and posterior shoulders (the area over the scapula). No other areas are similarly affected, but 1 fingernail is mildly pitted.
A #10 blade lifts the edge of one of the scales gently (and painlessly) until there is pinpoint bleeding from 2 tiny foci. A 5-mm full-thickness punch biopsy with primary closure shows marked parakeratosis, collections of neutrophils on the crests of dermal papillae, and fusing of rete ridges, which effectively obscure the normal wave-like pattern of the dermoepidermal junction.
Psoriasis ointment helped with itch, healing in phase 2 EB study
LONDON – , in a small, placebo-controlled, phase 2 study.
More importantly, use of the ointment promoted wound healing in those with the severe skin-blistering condition. Indeed, compared with placebo, a greater reduction in wound size was observed after 2 weeks when the ointment was applied (a mean reduction of 65.5% vs. 88.4%; P less than .006). However, at 1 month, no significant differences were seen in the size of the wounds between the two treatment arms.
“Calcipotriol is a vitamin D analog and it is well known that vitamin D is a very critical factor for skin homeostasis and proper wound healing,” Christina Guttmann-Gruber, PhD, said at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA). Dr. Guttmann-Gruber, a group lead researcher for EB House Austria, which is based at the Salzburg (Austria) University Clinic for Dermatology, noted that vitamin D also helps with tissue repair and immune modulation, and enhances local antimicrobial activity.
During an oral poster presentation at the meeting, Dr. Guttmann-Gruber explained that in previous in vitro studies, it was found that low concentrations (100 nmol) of calcipotriol inhibited proliferation of RDEB tumor cells (Sci Rep. 2018 Sep 7;8:13430). Calcipotriol (also known as calcipotriene) also was found to improve the expression of antimicrobial peptides and promote wound closure. “Therefore, we thought that applying calcipotriol at the site of injury, on chronic wounds prone to superinfection where it is needed, might be beneficial for our patients.”
She and her associates designed a two-arm, randomized, double-blind crossover study to assess the effects of an existing calcipotriol-containing ointment on wound healing in patients with RDEB. The ointment used in the study is approved for treating psoriasis but was adapted by the in-house pharmacy team to reduce the concentration of calcipotriol to about 0.05 mcg/g, or around 121 nmol. The reason for the reduction was that, at higher doses, keratinocyte proliferation was reduced, which would be detrimental in RDEB patients.
Nine patients were included in the study and were randomized to either apply 1 g of the active or placebo ointment to each of two designated wounds, of at least 6 cm2 in size, every day for 4 weeks. A 2-month washout period then followed before the groups switched to use the other ointment for 1 month. Six out of the nine patients completed both treatment phases. The reasons for the patients not completing both intervention phases were not related to the drug.
Calcipotriol treatment resulted in a significant and steady reduction in itch over the entire course of treatment, which was not seen among those on placebo, Dr. Guttmann-Gruber observed. The reduction in itch was “striking,” but only while the treatment was being used, she said. Results for pain were less clear, with a significant reduction in pain after 2 weeks seen only in the placebo group, while both treatments reduced pain to the same degree by 1 month.
No serious adverse events were observed at any time point and topical use of the low-dose calcipotriol did not significantly change serum levels of calcium or vitamin D in the two patients in which this was studied, Dr. Guttmann-Gruber said.
“This is an approved drug; it’s used in psoriasis, but at a very high concentration. We were able to use it off label and make a diluted version,” she observed. “Any pharmacy can do it.” Although it was applied topically, it could be done by applying it to the dressing rather directly onto the wounded skin, she said.
Data on the skin microbiome response to treatment were also collected but were not available to analyze in time for presentation, but it appeared that there was improvement with the low-dose calcipotriol treatment, Dr. Guttmann-Gruber said. “When the wounds are healing, the microbial flora is improving.”
The next step will probably be to plan a multicenter trial of this treatment, Dr. Guttmann-Gruber said in an interview. The questions is whether such a trial would get the financial backing it needed, but if an orphan drug designation could be obtained for calcipotriol for EB, then it would be possible to conduct such a trial.
The study was funded by DEBRA Austria. The presenting author, Dr. Guttmann-Gruber, had no conflicts of interest to disclose.
SOURCE: Guttmann-Gruber C et al. EB World Congress 2020. Poster 34.
LONDON – , in a small, placebo-controlled, phase 2 study.
More importantly, use of the ointment promoted wound healing in those with the severe skin-blistering condition. Indeed, compared with placebo, a greater reduction in wound size was observed after 2 weeks when the ointment was applied (a mean reduction of 65.5% vs. 88.4%; P less than .006). However, at 1 month, no significant differences were seen in the size of the wounds between the two treatment arms.
“Calcipotriol is a vitamin D analog and it is well known that vitamin D is a very critical factor for skin homeostasis and proper wound healing,” Christina Guttmann-Gruber, PhD, said at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA). Dr. Guttmann-Gruber, a group lead researcher for EB House Austria, which is based at the Salzburg (Austria) University Clinic for Dermatology, noted that vitamin D also helps with tissue repair and immune modulation, and enhances local antimicrobial activity.
During an oral poster presentation at the meeting, Dr. Guttmann-Gruber explained that in previous in vitro studies, it was found that low concentrations (100 nmol) of calcipotriol inhibited proliferation of RDEB tumor cells (Sci Rep. 2018 Sep 7;8:13430). Calcipotriol (also known as calcipotriene) also was found to improve the expression of antimicrobial peptides and promote wound closure. “Therefore, we thought that applying calcipotriol at the site of injury, on chronic wounds prone to superinfection where it is needed, might be beneficial for our patients.”
She and her associates designed a two-arm, randomized, double-blind crossover study to assess the effects of an existing calcipotriol-containing ointment on wound healing in patients with RDEB. The ointment used in the study is approved for treating psoriasis but was adapted by the in-house pharmacy team to reduce the concentration of calcipotriol to about 0.05 mcg/g, or around 121 nmol. The reason for the reduction was that, at higher doses, keratinocyte proliferation was reduced, which would be detrimental in RDEB patients.
Nine patients were included in the study and were randomized to either apply 1 g of the active or placebo ointment to each of two designated wounds, of at least 6 cm2 in size, every day for 4 weeks. A 2-month washout period then followed before the groups switched to use the other ointment for 1 month. Six out of the nine patients completed both treatment phases. The reasons for the patients not completing both intervention phases were not related to the drug.
Calcipotriol treatment resulted in a significant and steady reduction in itch over the entire course of treatment, which was not seen among those on placebo, Dr. Guttmann-Gruber observed. The reduction in itch was “striking,” but only while the treatment was being used, she said. Results for pain were less clear, with a significant reduction in pain after 2 weeks seen only in the placebo group, while both treatments reduced pain to the same degree by 1 month.
No serious adverse events were observed at any time point and topical use of the low-dose calcipotriol did not significantly change serum levels of calcium or vitamin D in the two patients in which this was studied, Dr. Guttmann-Gruber said.
“This is an approved drug; it’s used in psoriasis, but at a very high concentration. We were able to use it off label and make a diluted version,” she observed. “Any pharmacy can do it.” Although it was applied topically, it could be done by applying it to the dressing rather directly onto the wounded skin, she said.
Data on the skin microbiome response to treatment were also collected but were not available to analyze in time for presentation, but it appeared that there was improvement with the low-dose calcipotriol treatment, Dr. Guttmann-Gruber said. “When the wounds are healing, the microbial flora is improving.”
The next step will probably be to plan a multicenter trial of this treatment, Dr. Guttmann-Gruber said in an interview. The questions is whether such a trial would get the financial backing it needed, but if an orphan drug designation could be obtained for calcipotriol for EB, then it would be possible to conduct such a trial.
The study was funded by DEBRA Austria. The presenting author, Dr. Guttmann-Gruber, had no conflicts of interest to disclose.
SOURCE: Guttmann-Gruber C et al. EB World Congress 2020. Poster 34.
LONDON – , in a small, placebo-controlled, phase 2 study.
More importantly, use of the ointment promoted wound healing in those with the severe skin-blistering condition. Indeed, compared with placebo, a greater reduction in wound size was observed after 2 weeks when the ointment was applied (a mean reduction of 65.5% vs. 88.4%; P less than .006). However, at 1 month, no significant differences were seen in the size of the wounds between the two treatment arms.
“Calcipotriol is a vitamin D analog and it is well known that vitamin D is a very critical factor for skin homeostasis and proper wound healing,” Christina Guttmann-Gruber, PhD, said at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA). Dr. Guttmann-Gruber, a group lead researcher for EB House Austria, which is based at the Salzburg (Austria) University Clinic for Dermatology, noted that vitamin D also helps with tissue repair and immune modulation, and enhances local antimicrobial activity.
During an oral poster presentation at the meeting, Dr. Guttmann-Gruber explained that in previous in vitro studies, it was found that low concentrations (100 nmol) of calcipotriol inhibited proliferation of RDEB tumor cells (Sci Rep. 2018 Sep 7;8:13430). Calcipotriol (also known as calcipotriene) also was found to improve the expression of antimicrobial peptides and promote wound closure. “Therefore, we thought that applying calcipotriol at the site of injury, on chronic wounds prone to superinfection where it is needed, might be beneficial for our patients.”
She and her associates designed a two-arm, randomized, double-blind crossover study to assess the effects of an existing calcipotriol-containing ointment on wound healing in patients with RDEB. The ointment used in the study is approved for treating psoriasis but was adapted by the in-house pharmacy team to reduce the concentration of calcipotriol to about 0.05 mcg/g, or around 121 nmol. The reason for the reduction was that, at higher doses, keratinocyte proliferation was reduced, which would be detrimental in RDEB patients.
Nine patients were included in the study and were randomized to either apply 1 g of the active or placebo ointment to each of two designated wounds, of at least 6 cm2 in size, every day for 4 weeks. A 2-month washout period then followed before the groups switched to use the other ointment for 1 month. Six out of the nine patients completed both treatment phases. The reasons for the patients not completing both intervention phases were not related to the drug.
Calcipotriol treatment resulted in a significant and steady reduction in itch over the entire course of treatment, which was not seen among those on placebo, Dr. Guttmann-Gruber observed. The reduction in itch was “striking,” but only while the treatment was being used, she said. Results for pain were less clear, with a significant reduction in pain after 2 weeks seen only in the placebo group, while both treatments reduced pain to the same degree by 1 month.
No serious adverse events were observed at any time point and topical use of the low-dose calcipotriol did not significantly change serum levels of calcium or vitamin D in the two patients in which this was studied, Dr. Guttmann-Gruber said.
“This is an approved drug; it’s used in psoriasis, but at a very high concentration. We were able to use it off label and make a diluted version,” she observed. “Any pharmacy can do it.” Although it was applied topically, it could be done by applying it to the dressing rather directly onto the wounded skin, she said.
Data on the skin microbiome response to treatment were also collected but were not available to analyze in time for presentation, but it appeared that there was improvement with the low-dose calcipotriol treatment, Dr. Guttmann-Gruber said. “When the wounds are healing, the microbial flora is improving.”
The next step will probably be to plan a multicenter trial of this treatment, Dr. Guttmann-Gruber said in an interview. The questions is whether such a trial would get the financial backing it needed, but if an orphan drug designation could be obtained for calcipotriol for EB, then it would be possible to conduct such a trial.
The study was funded by DEBRA Austria. The presenting author, Dr. Guttmann-Gruber, had no conflicts of interest to disclose.
SOURCE: Guttmann-Gruber C et al. EB World Congress 2020. Poster 34.
REPORTING FROM EB 2020