User login
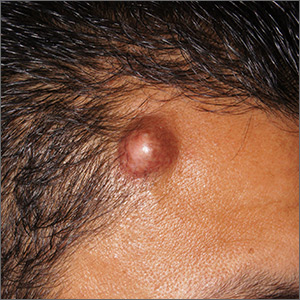
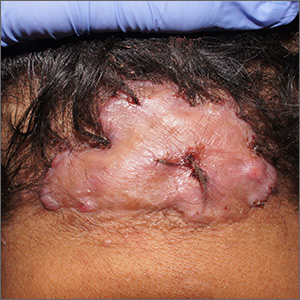
Forehead cyst

Upon palpation, the physician noted a strong pulse consistent with a traumatic arteriovenous fistula (in this case involving the superficial temporal artery). This finding, combined with the cyst’s appearance and the patient’s history, led the physician conclude that this was an epidermoid (sebaceous) cyst. (Prior to palpation, the visual differential diagnosis included dermoid cyst, lipoma, trichilemmal or epidermoid cyst, and foreign body granuloma.)
Cystic nodules on the forehead and midline deserve close scrutiny. This author has seen 3 similar cases in 10 years of daily dermatology consultative practice that have involved the superficial temporal artery and a history of head trauma. Each case had been stable for many months before presentation and had been incorrectly identified as a more common benign cyst.
In this particular case, the planned procedure in the outpatient setting was cancelled and the patient was referred to Vascular Surgery, where surgeons were planning to perform a ligation of the superficial temporal artery.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Matsumoto H, Yamaura I, Yoshida Y. Identity of growing pulsatile mass lesion of the scalp after blunt head injury: case reports and literature review. Trauma Case Rep. 2018;17:43-47.

Upon palpation, the physician noted a strong pulse consistent with a traumatic arteriovenous fistula (in this case involving the superficial temporal artery). This finding, combined with the cyst’s appearance and the patient’s history, led the physician conclude that this was an epidermoid (sebaceous) cyst. (Prior to palpation, the visual differential diagnosis included dermoid cyst, lipoma, trichilemmal or epidermoid cyst, and foreign body granuloma.)
Cystic nodules on the forehead and midline deserve close scrutiny. This author has seen 3 similar cases in 10 years of daily dermatology consultative practice that have involved the superficial temporal artery and a history of head trauma. Each case had been stable for many months before presentation and had been incorrectly identified as a more common benign cyst.
In this particular case, the planned procedure in the outpatient setting was cancelled and the patient was referred to Vascular Surgery, where surgeons were planning to perform a ligation of the superficial temporal artery.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Upon palpation, the physician noted a strong pulse consistent with a traumatic arteriovenous fistula (in this case involving the superficial temporal artery). This finding, combined with the cyst’s appearance and the patient’s history, led the physician conclude that this was an epidermoid (sebaceous) cyst. (Prior to palpation, the visual differential diagnosis included dermoid cyst, lipoma, trichilemmal or epidermoid cyst, and foreign body granuloma.)
Cystic nodules on the forehead and midline deserve close scrutiny. This author has seen 3 similar cases in 10 years of daily dermatology consultative practice that have involved the superficial temporal artery and a history of head trauma. Each case had been stable for many months before presentation and had been incorrectly identified as a more common benign cyst.
In this particular case, the planned procedure in the outpatient setting was cancelled and the patient was referred to Vascular Surgery, where surgeons were planning to perform a ligation of the superficial temporal artery.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Matsumoto H, Yamaura I, Yoshida Y. Identity of growing pulsatile mass lesion of the scalp after blunt head injury: case reports and literature review. Trauma Case Rep. 2018;17:43-47.
Matsumoto H, Yamaura I, Yoshida Y. Identity of growing pulsatile mass lesion of the scalp after blunt head injury: case reports and literature review. Trauma Case Rep. 2018;17:43-47.
Dupilumab approval sought for AD under age 12
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
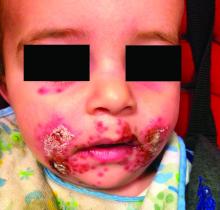
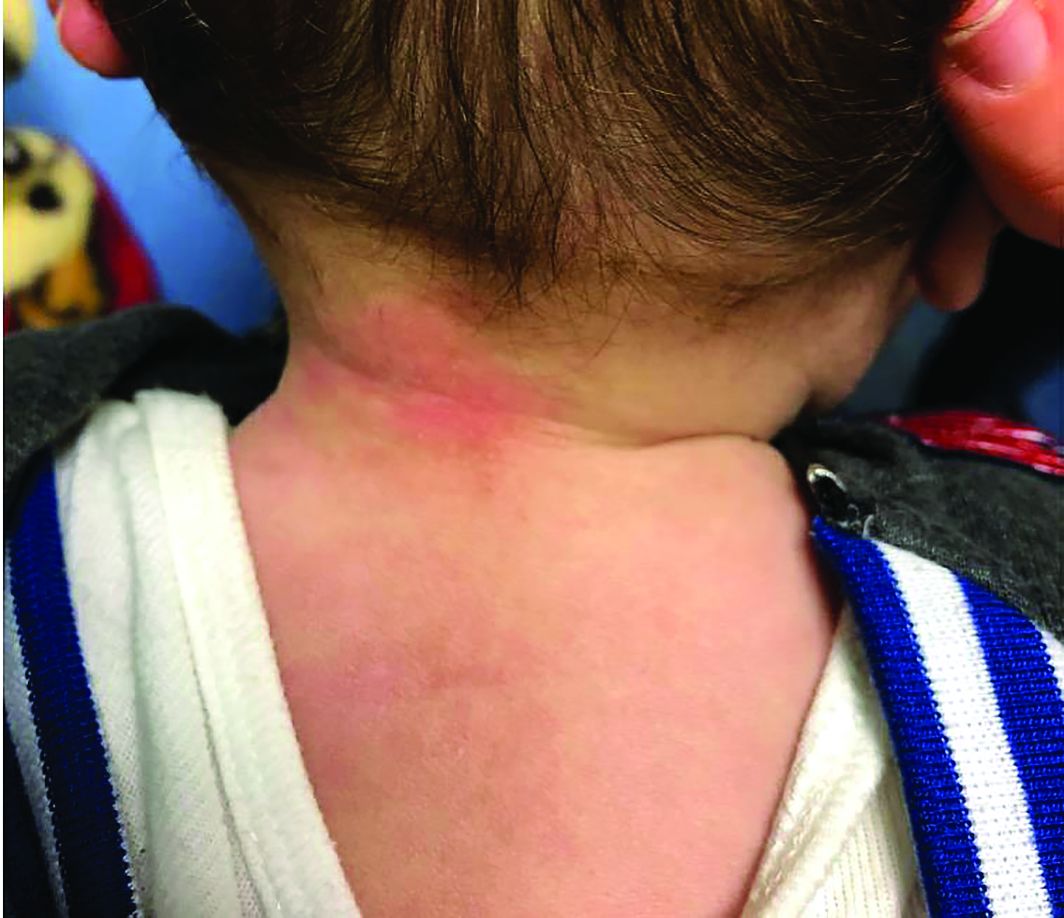
A 7-month-old male presents with perioral rash and fever
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at pdnews@mdedge.com.
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at pdnews@mdedge.com.
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at pdnews@mdedge.com.
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Enlarging scalp plaque
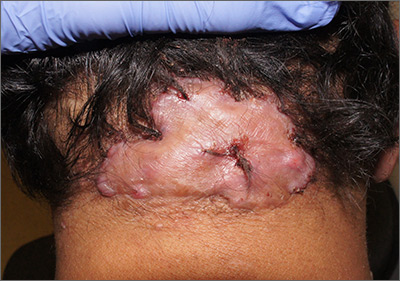
The findings of follicular-based papules, pustules, and scars led to the diagnosis of folliculitis keloidalis nuchae.
Follicular keloidalis, also called acne keloidalis nuchae, is more common in patients with darker skin types (Fitzpatrick skin types IV-VI). The pathogenesis is unclear, but the condition may arise from mechanical occlusion with a retained short hair that leads to follicular destruction. It also may be a primary disorder arising from bacterial infection and subsequent vigorous inflammation.
In the earliest stages, when only small inflammatory papules are present, patients should be instructed not to cut their hair shorter than 0.25-in long to avoid hair retention. Also, topical clindamycin lotion or topical chlorhexidine solution may serve as sufficient treatment. As the disease progresses, oral doxycycline 100 mg bid, combination rifampin 300 mg bid and clindamycin 300 mg bid for 3 months, or isotretinoin 1 mg/kg daily for 6 to 8 months are medical therapeutic options. Procedural options include radiation and laser hair removal.
In this patient, trials of intralesional triamcinolone acetonide 10 mg/mL helped to flatten the plaque, but hair loss persisted. Ultimately, he was referred to Plastic Surgery for excision and was treated for several months with doxycycline 100 mg bid and monitored for recurrence.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Chouk C, Litaiem N, Jones M, et al. Acne keloidalis nuchae: clinical and dermoscopic features. BMJ Case Rep. 2017. pii: bcr-2017-222222. doi: 10.1136/bcr-2017-222222.

The findings of follicular-based papules, pustules, and scars led to the diagnosis of folliculitis keloidalis nuchae.
Follicular keloidalis, also called acne keloidalis nuchae, is more common in patients with darker skin types (Fitzpatrick skin types IV-VI). The pathogenesis is unclear, but the condition may arise from mechanical occlusion with a retained short hair that leads to follicular destruction. It also may be a primary disorder arising from bacterial infection and subsequent vigorous inflammation.
In the earliest stages, when only small inflammatory papules are present, patients should be instructed not to cut their hair shorter than 0.25-in long to avoid hair retention. Also, topical clindamycin lotion or topical chlorhexidine solution may serve as sufficient treatment. As the disease progresses, oral doxycycline 100 mg bid, combination rifampin 300 mg bid and clindamycin 300 mg bid for 3 months, or isotretinoin 1 mg/kg daily for 6 to 8 months are medical therapeutic options. Procedural options include radiation and laser hair removal.
In this patient, trials of intralesional triamcinolone acetonide 10 mg/mL helped to flatten the plaque, but hair loss persisted. Ultimately, he was referred to Plastic Surgery for excision and was treated for several months with doxycycline 100 mg bid and monitored for recurrence.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The findings of follicular-based papules, pustules, and scars led to the diagnosis of folliculitis keloidalis nuchae.
Follicular keloidalis, also called acne keloidalis nuchae, is more common in patients with darker skin types (Fitzpatrick skin types IV-VI). The pathogenesis is unclear, but the condition may arise from mechanical occlusion with a retained short hair that leads to follicular destruction. It also may be a primary disorder arising from bacterial infection and subsequent vigorous inflammation.
In the earliest stages, when only small inflammatory papules are present, patients should be instructed not to cut their hair shorter than 0.25-in long to avoid hair retention. Also, topical clindamycin lotion or topical chlorhexidine solution may serve as sufficient treatment. As the disease progresses, oral doxycycline 100 mg bid, combination rifampin 300 mg bid and clindamycin 300 mg bid for 3 months, or isotretinoin 1 mg/kg daily for 6 to 8 months are medical therapeutic options. Procedural options include radiation and laser hair removal.
In this patient, trials of intralesional triamcinolone acetonide 10 mg/mL helped to flatten the plaque, but hair loss persisted. Ultimately, he was referred to Plastic Surgery for excision and was treated for several months with doxycycline 100 mg bid and monitored for recurrence.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Chouk C, Litaiem N, Jones M, et al. Acne keloidalis nuchae: clinical and dermoscopic features. BMJ Case Rep. 2017. pii: bcr-2017-222222. doi: 10.1136/bcr-2017-222222.
Chouk C, Litaiem N, Jones M, et al. Acne keloidalis nuchae: clinical and dermoscopic features. BMJ Case Rep. 2017. pii: bcr-2017-222222. doi: 10.1136/bcr-2017-222222.
Coronavirus outbreak prompts cancellation of AAD annual meeting
The American Academy of Dermatology annual meeting is the latest large medical conference to be canceled because of the coronavirus disease 2019 (COVID-19) outbreak.
“After carefully weighing the emerging facts, as well as our duties to the Academy, our members, other meeting attendees, and the local communities we as dermatologists serve, the AAD has made the difficult but necessary decision to cancel the AAD 2020 Annual Meeting in Denver,” AAD President George Hruza, MD, said in an announcement posted on the AAD’s website late on March 9. “We also want to respect our physicians’ need to be available to and healthy for their own patients, communities, and countries,” he added.
Earlier in the day, the American College of Cardiology announced that its annual meeting would be canceled, as did the Society of Gynecologic Oncology.
In his statement, Dr. Hruza said that the AAD is looking into “virtual meeting options” to provide content that was scheduled to be presented at the meeting.
Updates on those plans will be posted on the AAD’s website at www.aad.org.
The American Academy of Dermatology annual meeting is the latest large medical conference to be canceled because of the coronavirus disease 2019 (COVID-19) outbreak.
“After carefully weighing the emerging facts, as well as our duties to the Academy, our members, other meeting attendees, and the local communities we as dermatologists serve, the AAD has made the difficult but necessary decision to cancel the AAD 2020 Annual Meeting in Denver,” AAD President George Hruza, MD, said in an announcement posted on the AAD’s website late on March 9. “We also want to respect our physicians’ need to be available to and healthy for their own patients, communities, and countries,” he added.
Earlier in the day, the American College of Cardiology announced that its annual meeting would be canceled, as did the Society of Gynecologic Oncology.
In his statement, Dr. Hruza said that the AAD is looking into “virtual meeting options” to provide content that was scheduled to be presented at the meeting.
Updates on those plans will be posted on the AAD’s website at www.aad.org.
The American Academy of Dermatology annual meeting is the latest large medical conference to be canceled because of the coronavirus disease 2019 (COVID-19) outbreak.
“After carefully weighing the emerging facts, as well as our duties to the Academy, our members, other meeting attendees, and the local communities we as dermatologists serve, the AAD has made the difficult but necessary decision to cancel the AAD 2020 Annual Meeting in Denver,” AAD President George Hruza, MD, said in an announcement posted on the AAD’s website late on March 9. “We also want to respect our physicians’ need to be available to and healthy for their own patients, communities, and countries,” he added.
Earlier in the day, the American College of Cardiology announced that its annual meeting would be canceled, as did the Society of Gynecologic Oncology.
In his statement, Dr. Hruza said that the AAD is looking into “virtual meeting options” to provide content that was scheduled to be presented at the meeting.
Updates on those plans will be posted on the AAD’s website at www.aad.org.
Antifungal drug appears safe for pregnancy
Treatment with the according to results from a large registry study in Denmark.
Physicians have been reluctant to prescribe the drug during pregnancy because of the limited safety data. The drug has not been associated with any signs of fetal toxicity in animal studies, but only one study – in 54 pregnancies – has examined the issue in humans and did not identify an increased fetal risk, according to Niklas Worm Andersson, MD, of the department of clinical pharmacology, Copenhagen University Hospital at Bispebjerg and Frederiksberg, and coauthors.
The retrospective, nationwide cohort study analyzed exposure to oral and tropical terbinafine in a large pregnancy registry and found no increase in the risk of major malformations or spontaneous abortions in exposed versus unexposed pregnancies. The study was published in JAMA Dermatology.
Still, these results fell short of certainty, the authors noted. “Although our results may provide reassurance for pregnancies exposed to oral terbinafine by reporting no overall increased risk of major malformations, we cannot exclude a potential increased risk of a specific malformation,” they wrote.
“To our knowledge, this is by far the largest, most statistically rigorous study in the literature regarding this topic,” Jenny E. Murase, MD, of the department of dermatology at the University of California, San Francisco, and Mary Kathryn Abel, a medical student at UCSF, wrote in an accompanying editorial. They described the study as “a substantial contribution to the nearly absent literature regarding the use of terbinafine during pregnancy. Among the antifungal medications, it is possible that terbinafine is the safest one currently available for use in pregnancy, particularly of the oral formulations.”
However, since asymptomatic onychomycosis “is typically a cosmetic, rather than medical, concern, waiting until after pregnancy to initiate therapy is reasonable. ... It is important to acknowledge the uncertainty in this field and question the appropriateness of treating non–life-threatening diseases during pregnancy and lactation,” they wrote.
The Danish researchers drew from a registry of 1,650,649 pregnancies between 1997 and 2016, which included 891 pregnancies exposed to oral terbinafine, and 3,174 exposed to topical terbinafine. Matched outcome analyses compared the exposed pregnancies with up to 40,650 controls unexposed during pregnancy.
Propensity-matched comparisons showed no increased risk of major malformations for oral terbinafine exposure versus no exposure (odds ratio, 1.01; 95% confidence interval, 0.63-1.62) or topical exposure versus no exposure (OR, 1.08; 95% CI, 0.81-1.44). There was also no difference in oral versus topical exposure (OR, 1.18; 95% CI, 0.61-2.29).
With respect to spontaneous abortions, there was no significant association with oral terbinafine (hazard ratio, 1.06; 95% CI, 0.86-1.32) or topical terbinafine (HR, 1.04; 95% CI, 0.88-1.21), compared with unexposed pregnancies, or oral over topical terbinafine-exposed pregnancies (HR, 1.19; 95% CI, 0.84-1.70).
The study is limited by the fact that it was conducted in a Danish population, and the data relied on filled prescriptions for determining exposure, which did not account for adherence. Residual confounding is possible because of the retrospective nature of the study, the authors pointed out.
No source of funding was disclosed. One of the authors has received grants and personal fees from Novartis. Dr. Murase has received fees from Sanofi Genzyme, Dermira, UCB, Regeneron, Ferndale, and UpToDate.
SOURCES: Andersson NW et al. JAMA Dermatol. 2020 Mar 4. doi: 10.1001/jamadermatol.2020.0142; Murase JE, Abel MK. JAMA Dermatol. 2020 Mar 4. doi: 10.1001/jamadermatol.2019.5036.
Treatment with the according to results from a large registry study in Denmark.
Physicians have been reluctant to prescribe the drug during pregnancy because of the limited safety data. The drug has not been associated with any signs of fetal toxicity in animal studies, but only one study – in 54 pregnancies – has examined the issue in humans and did not identify an increased fetal risk, according to Niklas Worm Andersson, MD, of the department of clinical pharmacology, Copenhagen University Hospital at Bispebjerg and Frederiksberg, and coauthors.
The retrospective, nationwide cohort study analyzed exposure to oral and tropical terbinafine in a large pregnancy registry and found no increase in the risk of major malformations or spontaneous abortions in exposed versus unexposed pregnancies. The study was published in JAMA Dermatology.
Still, these results fell short of certainty, the authors noted. “Although our results may provide reassurance for pregnancies exposed to oral terbinafine by reporting no overall increased risk of major malformations, we cannot exclude a potential increased risk of a specific malformation,” they wrote.
“To our knowledge, this is by far the largest, most statistically rigorous study in the literature regarding this topic,” Jenny E. Murase, MD, of the department of dermatology at the University of California, San Francisco, and Mary Kathryn Abel, a medical student at UCSF, wrote in an accompanying editorial. They described the study as “a substantial contribution to the nearly absent literature regarding the use of terbinafine during pregnancy. Among the antifungal medications, it is possible that terbinafine is the safest one currently available for use in pregnancy, particularly of the oral formulations.”
However, since asymptomatic onychomycosis “is typically a cosmetic, rather than medical, concern, waiting until after pregnancy to initiate therapy is reasonable. ... It is important to acknowledge the uncertainty in this field and question the appropriateness of treating non–life-threatening diseases during pregnancy and lactation,” they wrote.
The Danish researchers drew from a registry of 1,650,649 pregnancies between 1997 and 2016, which included 891 pregnancies exposed to oral terbinafine, and 3,174 exposed to topical terbinafine. Matched outcome analyses compared the exposed pregnancies with up to 40,650 controls unexposed during pregnancy.
Propensity-matched comparisons showed no increased risk of major malformations for oral terbinafine exposure versus no exposure (odds ratio, 1.01; 95% confidence interval, 0.63-1.62) or topical exposure versus no exposure (OR, 1.08; 95% CI, 0.81-1.44). There was also no difference in oral versus topical exposure (OR, 1.18; 95% CI, 0.61-2.29).
With respect to spontaneous abortions, there was no significant association with oral terbinafine (hazard ratio, 1.06; 95% CI, 0.86-1.32) or topical terbinafine (HR, 1.04; 95% CI, 0.88-1.21), compared with unexposed pregnancies, or oral over topical terbinafine-exposed pregnancies (HR, 1.19; 95% CI, 0.84-1.70).
The study is limited by the fact that it was conducted in a Danish population, and the data relied on filled prescriptions for determining exposure, which did not account for adherence. Residual confounding is possible because of the retrospective nature of the study, the authors pointed out.
No source of funding was disclosed. One of the authors has received grants and personal fees from Novartis. Dr. Murase has received fees from Sanofi Genzyme, Dermira, UCB, Regeneron, Ferndale, and UpToDate.
SOURCES: Andersson NW et al. JAMA Dermatol. 2020 Mar 4. doi: 10.1001/jamadermatol.2020.0142; Murase JE, Abel MK. JAMA Dermatol. 2020 Mar 4. doi: 10.1001/jamadermatol.2019.5036.
Treatment with the according to results from a large registry study in Denmark.
Physicians have been reluctant to prescribe the drug during pregnancy because of the limited safety data. The drug has not been associated with any signs of fetal toxicity in animal studies, but only one study – in 54 pregnancies – has examined the issue in humans and did not identify an increased fetal risk, according to Niklas Worm Andersson, MD, of the department of clinical pharmacology, Copenhagen University Hospital at Bispebjerg and Frederiksberg, and coauthors.
The retrospective, nationwide cohort study analyzed exposure to oral and tropical terbinafine in a large pregnancy registry and found no increase in the risk of major malformations or spontaneous abortions in exposed versus unexposed pregnancies. The study was published in JAMA Dermatology.
Still, these results fell short of certainty, the authors noted. “Although our results may provide reassurance for pregnancies exposed to oral terbinafine by reporting no overall increased risk of major malformations, we cannot exclude a potential increased risk of a specific malformation,” they wrote.
“To our knowledge, this is by far the largest, most statistically rigorous study in the literature regarding this topic,” Jenny E. Murase, MD, of the department of dermatology at the University of California, San Francisco, and Mary Kathryn Abel, a medical student at UCSF, wrote in an accompanying editorial. They described the study as “a substantial contribution to the nearly absent literature regarding the use of terbinafine during pregnancy. Among the antifungal medications, it is possible that terbinafine is the safest one currently available for use in pregnancy, particularly of the oral formulations.”
However, since asymptomatic onychomycosis “is typically a cosmetic, rather than medical, concern, waiting until after pregnancy to initiate therapy is reasonable. ... It is important to acknowledge the uncertainty in this field and question the appropriateness of treating non–life-threatening diseases during pregnancy and lactation,” they wrote.
The Danish researchers drew from a registry of 1,650,649 pregnancies between 1997 and 2016, which included 891 pregnancies exposed to oral terbinafine, and 3,174 exposed to topical terbinafine. Matched outcome analyses compared the exposed pregnancies with up to 40,650 controls unexposed during pregnancy.
Propensity-matched comparisons showed no increased risk of major malformations for oral terbinafine exposure versus no exposure (odds ratio, 1.01; 95% confidence interval, 0.63-1.62) or topical exposure versus no exposure (OR, 1.08; 95% CI, 0.81-1.44). There was also no difference in oral versus topical exposure (OR, 1.18; 95% CI, 0.61-2.29).
With respect to spontaneous abortions, there was no significant association with oral terbinafine (hazard ratio, 1.06; 95% CI, 0.86-1.32) or topical terbinafine (HR, 1.04; 95% CI, 0.88-1.21), compared with unexposed pregnancies, or oral over topical terbinafine-exposed pregnancies (HR, 1.19; 95% CI, 0.84-1.70).
The study is limited by the fact that it was conducted in a Danish population, and the data relied on filled prescriptions for determining exposure, which did not account for adherence. Residual confounding is possible because of the retrospective nature of the study, the authors pointed out.
No source of funding was disclosed. One of the authors has received grants and personal fees from Novartis. Dr. Murase has received fees from Sanofi Genzyme, Dermira, UCB, Regeneron, Ferndale, and UpToDate.
SOURCES: Andersson NW et al. JAMA Dermatol. 2020 Mar 4. doi: 10.1001/jamadermatol.2020.0142; Murase JE, Abel MK. JAMA Dermatol. 2020 Mar 4. doi: 10.1001/jamadermatol.2019.5036.
FROM JAMA DERMATOLOGY
Breaking bacterial communication may heal EB wounds
LONDON – Disrupting how microorganisms communicate with each other could be a way to overcome antibiotic resistance and to help heal chronic wounds in patients with epidermolysis bullosa (EB), according to presenters at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA).
The majority of chronic wounds in patients with EB are colonized with microorganisms, with a predominance of Staphylococcus species, said Erik Gerner, an industrial PhD student at Mölnlycke Health Care in Gothenburg, Sweden, and Gothenburg University.
Because of the growing problem of antibiotic resistance, alternative treatments are needed, and one possible alternative for treating infected wounds could be interfering with quorum sensing, the cell-to-cell communication used by bacteria, he said. He is hoping to explore this possibility as a novel treatment strategy for infected wounds.
“Quorum sensing is defined as the ability to detect and respond to population density,” Mr. Gerner said, noting that, when there is a sufficient density of bacteria, “they start to communicate with each other.” This enables them to act as a community and perform actions that they could not do as individual cells. Such actions include forming biofilms, which helps protect bacteria from their environment, such as the immune system. Other actions include collectively switching on the production of virulence factors and becoming resistant to treatments.
“Bacteria use quorum sensing to act collectively,” Mr. Gerner said. “If we could shut down this quorum sensing system, it would be very beneficial … and increase the chances to heal the wound.”
The quorum sensing system is based on the production of signaling molecules called AHL (N-acyl homoserine lactones), which are constantly produced at a low rate. This isn’t a problem until the level of bacteria increases and the level of quorum sensing breaches a threshold, he explained.
There are several benefits of inhibiting bacterial communication through disrupting quorum sensing, namely, “a low risk of resistance,” Mr. Gerner said. There is also potentially less toxin production by bacteria, and this could help the immune system in killing the invading bacteria.
One approach to disrupting quorum testing that Mr. Gerner has been investigating is the use of sodium salicylate (NaSa). So far, preclinical work shows that NaSa can reduce toxin production but not the growth rate of bacteria. The advantage of using NaSa is that it is nontoxic to human dermal fibroblasts, with similar results seen in human keratinocytes and immune cells. His work to date has shown that NaSa reduced activity of NF-kB (a proinflammatory signaling pathway) in differentiated and lipopolysaccharide-stimulated monocytes; NF-kB activated production of proinflammatory cytokines (such as interleukin-1 beta and IL-6) are elevated in EB wounds. “My studies support the bodies of evidence that bacteria use quorum sensing to coordinate … and to produce a large number of toxic factors,” Mr. Gerner concluded. Future studies will look at the potential of NaSa to disrupt this activity.
Skin microbiome of EB wounds
Liat Samuelov, MD, of the department of molecular dermatology at Tel Aviv (Israel) Sourasky Medical Center, presented data on skin microbiome characteristics in eight patients with recessive dystrophic EB (RDEB). This showed that there was reduced bacterial diversity in wounds, and a “progressive development of dysbiosis across different stages of DEB wound formation.”
The skin microbiome has been implicated in several skin diseases, Dr. Samuelov and associates observed in a poster presentation. That includes the autoimmune blistering disease bullous pemphigoid (Exp Dermatol. 2017 Dec;26[12]:1221-7). “Colonization of DEB chronic wounds may lead to systemic infections, result in delayed healing, and possibly be involved in the development of squamous cell carcinoma,” they noted in the poster, “thus accurate delineation of the dysbiotic profile … may point to corrective measures of great therapeutic potential.”
The aim was to see what microorganisms were present in the chronic wounds of the patients. To be included in the study, patients must not have had any antibiotic treatment – oral or topical – in the past 6 months. Samples were taken from an untreated wound, around the wound, and from uninvolved skin, which were compared with samples taken from similar areas in age-matched controls.
Reduced bacterial diversity was observed in RDEB wounds, compared with uninvolved or perilesional areas and the skin of control subjects, Dr. Samuelov said in an oral presentation of the study results. There was increased abundance of Staphylococcus epidermidis and decreased Cutibacterium acnes, which she noted was in contrast to other studies where S. aureus was the most common colonizer in RDEB wounds.
Bacterial composition in each group was calculated using the beta-diversity score, while control samples showed similar microbial composition, the DEB samples had no microbial similarities among different samples. These data “suggest the need to ascertain the potential therapeutic benefit of interventions aimed at restoring normal microbiome composition in DEB,” Dr. Samuelov concluded.
Wound colonization and squamous cell carcinoma
Other research on wound microbiology was presented by Laura E. Levin, MD, a dermatologist at New York–Presbyterian, and associates. “Given the potential role of bacteria-induced inflammation in the development of wound-associated SCC [squamous cell carcinoma] in a subset of patients, we sought to improve our understanding of what microbes colonize and infect the wounds of patients with epidermolysis bullosa,” they explained in their poster.
The researchers, from New York–Presbyterian Morgan Stanley Children’s Hospital and Columbia University Irvine Medical Center, New York, presented data from a retrospective analysis of 739 wound cultures taken between 2001 and 2017 from 158 patients enrolled in the Epidermolysis Bullosa Clinical Characterization and Outcomes Database. In the analysis, just under 70% of patients had DEB, of which 90% were of the RDEB subtype; 13% had EB simplex, 14% had junctional EB, and 3% had an unknown EB subtype.
At least one organism grew in 87% of cultures, with the most common microorganism isolated being Staphylococcus aureus (84% of cultures). Other commonly isolated microbes were Pseudomonas aeruginosa in 35% of cultures, Streptococcus group A in 34% of cultures (of which 22% were Streptococcus pyogenes), Corynebacterium species in 31% of cultures, and Proteus species in 18% of cultures.
“Improved understanding of what microbes are colonizing the wounds of our patients may help improve antibiotic stewardship,” the researchers stated.
Looking at the antibiotic susceptibilities, Dr. Levin and associates found that 68% of 115 cultures were sensitive to methicillin and 60% of 15 cultures were sensitive to mupirocin. “Resistance to many systemic and topical antibiotic agents in EB patients supports surveillance cultures with routine testing for mupirocin susceptibility,” they suggested.
A total of 23 patients developed SCC of whom 10 had cultures that grew S. aureus (90%) and P. aeruginosa (50%), and Proteus species (20%). Among the patients who did not develop SCC, the respective cultures positive for each of those microorganisms were 83%, 34%, and 11%. Perhaps “gram-negative and flagellated organisms may be more common in wounds of patients at risk for SCC,” they observed, adding that further studies were needed to determine if “wound microbiome interventions inhibit the risk of development of SCC and improve outcomes.”
Mr. Gerner’s research is supported by Mölnlycke Health Care. Dr. Samuelov had no disclosures. The work by Dr. Levin and associates is supported by the Pediatric Dermatology Research Alliance, EB Research Partnership, and the Epidermolysis Bullosa Medical Research Foundation.
LONDON – Disrupting how microorganisms communicate with each other could be a way to overcome antibiotic resistance and to help heal chronic wounds in patients with epidermolysis bullosa (EB), according to presenters at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA).
The majority of chronic wounds in patients with EB are colonized with microorganisms, with a predominance of Staphylococcus species, said Erik Gerner, an industrial PhD student at Mölnlycke Health Care in Gothenburg, Sweden, and Gothenburg University.
Because of the growing problem of antibiotic resistance, alternative treatments are needed, and one possible alternative for treating infected wounds could be interfering with quorum sensing, the cell-to-cell communication used by bacteria, he said. He is hoping to explore this possibility as a novel treatment strategy for infected wounds.
“Quorum sensing is defined as the ability to detect and respond to population density,” Mr. Gerner said, noting that, when there is a sufficient density of bacteria, “they start to communicate with each other.” This enables them to act as a community and perform actions that they could not do as individual cells. Such actions include forming biofilms, which helps protect bacteria from their environment, such as the immune system. Other actions include collectively switching on the production of virulence factors and becoming resistant to treatments.
“Bacteria use quorum sensing to act collectively,” Mr. Gerner said. “If we could shut down this quorum sensing system, it would be very beneficial … and increase the chances to heal the wound.”
The quorum sensing system is based on the production of signaling molecules called AHL (N-acyl homoserine lactones), which are constantly produced at a low rate. This isn’t a problem until the level of bacteria increases and the level of quorum sensing breaches a threshold, he explained.
There are several benefits of inhibiting bacterial communication through disrupting quorum sensing, namely, “a low risk of resistance,” Mr. Gerner said. There is also potentially less toxin production by bacteria, and this could help the immune system in killing the invading bacteria.
One approach to disrupting quorum testing that Mr. Gerner has been investigating is the use of sodium salicylate (NaSa). So far, preclinical work shows that NaSa can reduce toxin production but not the growth rate of bacteria. The advantage of using NaSa is that it is nontoxic to human dermal fibroblasts, with similar results seen in human keratinocytes and immune cells. His work to date has shown that NaSa reduced activity of NF-kB (a proinflammatory signaling pathway) in differentiated and lipopolysaccharide-stimulated monocytes; NF-kB activated production of proinflammatory cytokines (such as interleukin-1 beta and IL-6) are elevated in EB wounds. “My studies support the bodies of evidence that bacteria use quorum sensing to coordinate … and to produce a large number of toxic factors,” Mr. Gerner concluded. Future studies will look at the potential of NaSa to disrupt this activity.
Skin microbiome of EB wounds
Liat Samuelov, MD, of the department of molecular dermatology at Tel Aviv (Israel) Sourasky Medical Center, presented data on skin microbiome characteristics in eight patients with recessive dystrophic EB (RDEB). This showed that there was reduced bacterial diversity in wounds, and a “progressive development of dysbiosis across different stages of DEB wound formation.”
The skin microbiome has been implicated in several skin diseases, Dr. Samuelov and associates observed in a poster presentation. That includes the autoimmune blistering disease bullous pemphigoid (Exp Dermatol. 2017 Dec;26[12]:1221-7). “Colonization of DEB chronic wounds may lead to systemic infections, result in delayed healing, and possibly be involved in the development of squamous cell carcinoma,” they noted in the poster, “thus accurate delineation of the dysbiotic profile … may point to corrective measures of great therapeutic potential.”
The aim was to see what microorganisms were present in the chronic wounds of the patients. To be included in the study, patients must not have had any antibiotic treatment – oral or topical – in the past 6 months. Samples were taken from an untreated wound, around the wound, and from uninvolved skin, which were compared with samples taken from similar areas in age-matched controls.
Reduced bacterial diversity was observed in RDEB wounds, compared with uninvolved or perilesional areas and the skin of control subjects, Dr. Samuelov said in an oral presentation of the study results. There was increased abundance of Staphylococcus epidermidis and decreased Cutibacterium acnes, which she noted was in contrast to other studies where S. aureus was the most common colonizer in RDEB wounds.
Bacterial composition in each group was calculated using the beta-diversity score, while control samples showed similar microbial composition, the DEB samples had no microbial similarities among different samples. These data “suggest the need to ascertain the potential therapeutic benefit of interventions aimed at restoring normal microbiome composition in DEB,” Dr. Samuelov concluded.
Wound colonization and squamous cell carcinoma
Other research on wound microbiology was presented by Laura E. Levin, MD, a dermatologist at New York–Presbyterian, and associates. “Given the potential role of bacteria-induced inflammation in the development of wound-associated SCC [squamous cell carcinoma] in a subset of patients, we sought to improve our understanding of what microbes colonize and infect the wounds of patients with epidermolysis bullosa,” they explained in their poster.
The researchers, from New York–Presbyterian Morgan Stanley Children’s Hospital and Columbia University Irvine Medical Center, New York, presented data from a retrospective analysis of 739 wound cultures taken between 2001 and 2017 from 158 patients enrolled in the Epidermolysis Bullosa Clinical Characterization and Outcomes Database. In the analysis, just under 70% of patients had DEB, of which 90% were of the RDEB subtype; 13% had EB simplex, 14% had junctional EB, and 3% had an unknown EB subtype.
At least one organism grew in 87% of cultures, with the most common microorganism isolated being Staphylococcus aureus (84% of cultures). Other commonly isolated microbes were Pseudomonas aeruginosa in 35% of cultures, Streptococcus group A in 34% of cultures (of which 22% were Streptococcus pyogenes), Corynebacterium species in 31% of cultures, and Proteus species in 18% of cultures.
“Improved understanding of what microbes are colonizing the wounds of our patients may help improve antibiotic stewardship,” the researchers stated.
Looking at the antibiotic susceptibilities, Dr. Levin and associates found that 68% of 115 cultures were sensitive to methicillin and 60% of 15 cultures were sensitive to mupirocin. “Resistance to many systemic and topical antibiotic agents in EB patients supports surveillance cultures with routine testing for mupirocin susceptibility,” they suggested.
A total of 23 patients developed SCC of whom 10 had cultures that grew S. aureus (90%) and P. aeruginosa (50%), and Proteus species (20%). Among the patients who did not develop SCC, the respective cultures positive for each of those microorganisms were 83%, 34%, and 11%. Perhaps “gram-negative and flagellated organisms may be more common in wounds of patients at risk for SCC,” they observed, adding that further studies were needed to determine if “wound microbiome interventions inhibit the risk of development of SCC and improve outcomes.”
Mr. Gerner’s research is supported by Mölnlycke Health Care. Dr. Samuelov had no disclosures. The work by Dr. Levin and associates is supported by the Pediatric Dermatology Research Alliance, EB Research Partnership, and the Epidermolysis Bullosa Medical Research Foundation.
LONDON – Disrupting how microorganisms communicate with each other could be a way to overcome antibiotic resistance and to help heal chronic wounds in patients with epidermolysis bullosa (EB), according to presenters at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA).
The majority of chronic wounds in patients with EB are colonized with microorganisms, with a predominance of Staphylococcus species, said Erik Gerner, an industrial PhD student at Mölnlycke Health Care in Gothenburg, Sweden, and Gothenburg University.
Because of the growing problem of antibiotic resistance, alternative treatments are needed, and one possible alternative for treating infected wounds could be interfering with quorum sensing, the cell-to-cell communication used by bacteria, he said. He is hoping to explore this possibility as a novel treatment strategy for infected wounds.
“Quorum sensing is defined as the ability to detect and respond to population density,” Mr. Gerner said, noting that, when there is a sufficient density of bacteria, “they start to communicate with each other.” This enables them to act as a community and perform actions that they could not do as individual cells. Such actions include forming biofilms, which helps protect bacteria from their environment, such as the immune system. Other actions include collectively switching on the production of virulence factors and becoming resistant to treatments.
“Bacteria use quorum sensing to act collectively,” Mr. Gerner said. “If we could shut down this quorum sensing system, it would be very beneficial … and increase the chances to heal the wound.”
The quorum sensing system is based on the production of signaling molecules called AHL (N-acyl homoserine lactones), which are constantly produced at a low rate. This isn’t a problem until the level of bacteria increases and the level of quorum sensing breaches a threshold, he explained.
There are several benefits of inhibiting bacterial communication through disrupting quorum sensing, namely, “a low risk of resistance,” Mr. Gerner said. There is also potentially less toxin production by bacteria, and this could help the immune system in killing the invading bacteria.
One approach to disrupting quorum testing that Mr. Gerner has been investigating is the use of sodium salicylate (NaSa). So far, preclinical work shows that NaSa can reduce toxin production but not the growth rate of bacteria. The advantage of using NaSa is that it is nontoxic to human dermal fibroblasts, with similar results seen in human keratinocytes and immune cells. His work to date has shown that NaSa reduced activity of NF-kB (a proinflammatory signaling pathway) in differentiated and lipopolysaccharide-stimulated monocytes; NF-kB activated production of proinflammatory cytokines (such as interleukin-1 beta and IL-6) are elevated in EB wounds. “My studies support the bodies of evidence that bacteria use quorum sensing to coordinate … and to produce a large number of toxic factors,” Mr. Gerner concluded. Future studies will look at the potential of NaSa to disrupt this activity.
Skin microbiome of EB wounds
Liat Samuelov, MD, of the department of molecular dermatology at Tel Aviv (Israel) Sourasky Medical Center, presented data on skin microbiome characteristics in eight patients with recessive dystrophic EB (RDEB). This showed that there was reduced bacterial diversity in wounds, and a “progressive development of dysbiosis across different stages of DEB wound formation.”
The skin microbiome has been implicated in several skin diseases, Dr. Samuelov and associates observed in a poster presentation. That includes the autoimmune blistering disease bullous pemphigoid (Exp Dermatol. 2017 Dec;26[12]:1221-7). “Colonization of DEB chronic wounds may lead to systemic infections, result in delayed healing, and possibly be involved in the development of squamous cell carcinoma,” they noted in the poster, “thus accurate delineation of the dysbiotic profile … may point to corrective measures of great therapeutic potential.”
The aim was to see what microorganisms were present in the chronic wounds of the patients. To be included in the study, patients must not have had any antibiotic treatment – oral or topical – in the past 6 months. Samples were taken from an untreated wound, around the wound, and from uninvolved skin, which were compared with samples taken from similar areas in age-matched controls.
Reduced bacterial diversity was observed in RDEB wounds, compared with uninvolved or perilesional areas and the skin of control subjects, Dr. Samuelov said in an oral presentation of the study results. There was increased abundance of Staphylococcus epidermidis and decreased Cutibacterium acnes, which she noted was in contrast to other studies where S. aureus was the most common colonizer in RDEB wounds.
Bacterial composition in each group was calculated using the beta-diversity score, while control samples showed similar microbial composition, the DEB samples had no microbial similarities among different samples. These data “suggest the need to ascertain the potential therapeutic benefit of interventions aimed at restoring normal microbiome composition in DEB,” Dr. Samuelov concluded.
Wound colonization and squamous cell carcinoma
Other research on wound microbiology was presented by Laura E. Levin, MD, a dermatologist at New York–Presbyterian, and associates. “Given the potential role of bacteria-induced inflammation in the development of wound-associated SCC [squamous cell carcinoma] in a subset of patients, we sought to improve our understanding of what microbes colonize and infect the wounds of patients with epidermolysis bullosa,” they explained in their poster.
The researchers, from New York–Presbyterian Morgan Stanley Children’s Hospital and Columbia University Irvine Medical Center, New York, presented data from a retrospective analysis of 739 wound cultures taken between 2001 and 2017 from 158 patients enrolled in the Epidermolysis Bullosa Clinical Characterization and Outcomes Database. In the analysis, just under 70% of patients had DEB, of which 90% were of the RDEB subtype; 13% had EB simplex, 14% had junctional EB, and 3% had an unknown EB subtype.
At least one organism grew in 87% of cultures, with the most common microorganism isolated being Staphylococcus aureus (84% of cultures). Other commonly isolated microbes were Pseudomonas aeruginosa in 35% of cultures, Streptococcus group A in 34% of cultures (of which 22% were Streptococcus pyogenes), Corynebacterium species in 31% of cultures, and Proteus species in 18% of cultures.
“Improved understanding of what microbes are colonizing the wounds of our patients may help improve antibiotic stewardship,” the researchers stated.
Looking at the antibiotic susceptibilities, Dr. Levin and associates found that 68% of 115 cultures were sensitive to methicillin and 60% of 15 cultures were sensitive to mupirocin. “Resistance to many systemic and topical antibiotic agents in EB patients supports surveillance cultures with routine testing for mupirocin susceptibility,” they suggested.
A total of 23 patients developed SCC of whom 10 had cultures that grew S. aureus (90%) and P. aeruginosa (50%), and Proteus species (20%). Among the patients who did not develop SCC, the respective cultures positive for each of those microorganisms were 83%, 34%, and 11%. Perhaps “gram-negative and flagellated organisms may be more common in wounds of patients at risk for SCC,” they observed, adding that further studies were needed to determine if “wound microbiome interventions inhibit the risk of development of SCC and improve outcomes.”
Mr. Gerner’s research is supported by Mölnlycke Health Care. Dr. Samuelov had no disclosures. The work by Dr. Levin and associates is supported by the Pediatric Dermatology Research Alliance, EB Research Partnership, and the Epidermolysis Bullosa Medical Research Foundation.
REPORTING FROM EB 2020
‘Natural is not always good’ when it comes to treatments for alopecia
ORLANDO – Biotin is the most popular consumer supplement for alopecia and highly popular online, but should patients be taking it?
Patients may want something “natural” to treat their hair loss, but “natural is not always good,” Amy McMichael, MD, professor and chair of the department of dermatology at Wake Forest Baptist Health, Winston-Salem, N.C., said at the ODAC Dermatology, Aesthetic, & Surgical Conference.
Dosages of commercially available biotin supplements can vary significantly, with some doses as high as 10,000 mcg, making supraphysiological dosing possible. Dr. McMichael said that, not only is biotin unlikely to help with a patient’s alopecia, but a high intake of biotin can interfere with certain assays that use streptavidin-biotin capture techniques (Clin Chem Lab Med. 2017 May 1;55[6]:817-25). This could present a problem for a patient who experiences an MI or has a thyroid disorder where a high level of biotin could affect lab results, she noted.
Dr. McMichael advises patients that there is a
Questioning side effects of 5-alpha reductase inhibitors
Research in the early 2010s associated the 5-alpha reductase inhibitor finasteride with persistent sexual side effects and depression. But a later meta-analysis of the Prostate Cancer Prevention Trial and other trials did not find evidence of persistent sexual side effects or depression in men on finasteride, and the authors said that double-blind, placebo-controlled studies were needed (J Clin Aesthet Dermatol. 2014 Dec;7[12]:51-5).
However, two meta-analyses of 34 clinical trials published in 2015 found that none of the clinical trials evaluating finasteride treatment in patients with androgenic alopecia had accurate safety reporting (JAMA Dermatol. 2015 Jun;151[6]:600-6). Another study published by the same group in 2017 found that 0.8% of men aged 16-42 years to exposed to a 5-alpha reductase inhibitor developed persistent erectile dysfunction after a longer duration of exposure (median, 1,534 days). compared with a shorter duration of exposure (PeerJ. 2017 Mar 9;5:e3020).
The bottom line when considering use of 5-alpha reductase inhibitors in men is to discuss the outlier data on persistent sexual dysfunction in the studies, ask patients whether they have a history of sexual dysfunction and depression, and then only treat appropriate patients with no such history, Dr. McMichael said.
Use of 5-alpha reductase inhibitors also appears to be related to an increased risk of type 2 diabetes in men with benign prostatic hyperplasia, according to more recent data. In a study of patients in the U.K. Clinical Practice Research Datalink and Taiwanese National Health Insurance Research Database who received dutasteride, finasteride, or the alpha blocker tamsulosin, there was a slightly increased risk of type 2 diabetes among those who took the two 5-alpha reductase inhibitors, compared with those on tamsulosin (BMJ. 2019;365:l1204). In light of these results, Dr. McMichael advised clinicians to be aware of these risks and to consider screening patients for type 2 diabetes and ask them about their family history, but the results shouldn’t affect patients at risk for type 2 diabetes or metabolic disorder.
JAK inhibitors for alopecia areata
Within the past few years, promising results for Janus kinase (JAK) inhibitors like tofacitinib and ruxolitinib for alopecia areata have been reported, but they are currently not a first-line therapy, Dr. McMichael said. Consider methotrexate first in older adolescents and adults with more than 50% hair loss, followed by a JAK inhibitor if there is no improvement or methotrexate is not tolerated well.
Clinicians can consider enrolling their patients in a clinical trial to give them access to JAK inhibitors as a treatment option, she noted, and if a trial is not available, it may be worth appealing to an insurance company using an article titled “Alopecia areata is a medical disease” coauthored by Dr. McMichael and others (Am Acad Dermatol. 2018 Apr;78[4]:832-4). After two denials by insurance, the manufacturer’s patient assistance program (Xelsource) may be helpful in obtaining tofacitinib (Xeljanz) through a letter and references. There are adolescent data on JAK inhibitors for alopecia, but “absolutely no data” in very young children, so prior to adolescence, she would not recommend this treatment. In a study of 13 adolescents with alopecia areata, totalis, or universalis, those treated with tofacitinib for a mean of 6.5 months, 9 had significant hair regrowth with treatment and adverse events were mild (J Am Acad Dermatol. 2017 Jan;76[1]:29-32).
Dr. McMichael reports being an investigator for Allergan, Intendis, Proctor & Gamble, Samumed, Cassiopia, Concert, Aclaris, and Incyte; and is a consultant for Johnson & Johnson, Proctor & Gamble, Allergan, Bayer, Galderma, Incyte, Samumed, Aclaris, Anacor, Pfizer, Nutrafol, Bioniz, and Almirall.
ORLANDO – Biotin is the most popular consumer supplement for alopecia and highly popular online, but should patients be taking it?
Patients may want something “natural” to treat their hair loss, but “natural is not always good,” Amy McMichael, MD, professor and chair of the department of dermatology at Wake Forest Baptist Health, Winston-Salem, N.C., said at the ODAC Dermatology, Aesthetic, & Surgical Conference.
Dosages of commercially available biotin supplements can vary significantly, with some doses as high as 10,000 mcg, making supraphysiological dosing possible. Dr. McMichael said that, not only is biotin unlikely to help with a patient’s alopecia, but a high intake of biotin can interfere with certain assays that use streptavidin-biotin capture techniques (Clin Chem Lab Med. 2017 May 1;55[6]:817-25). This could present a problem for a patient who experiences an MI or has a thyroid disorder where a high level of biotin could affect lab results, she noted.
Dr. McMichael advises patients that there is a
Questioning side effects of 5-alpha reductase inhibitors
Research in the early 2010s associated the 5-alpha reductase inhibitor finasteride with persistent sexual side effects and depression. But a later meta-analysis of the Prostate Cancer Prevention Trial and other trials did not find evidence of persistent sexual side effects or depression in men on finasteride, and the authors said that double-blind, placebo-controlled studies were needed (J Clin Aesthet Dermatol. 2014 Dec;7[12]:51-5).
However, two meta-analyses of 34 clinical trials published in 2015 found that none of the clinical trials evaluating finasteride treatment in patients with androgenic alopecia had accurate safety reporting (JAMA Dermatol. 2015 Jun;151[6]:600-6). Another study published by the same group in 2017 found that 0.8% of men aged 16-42 years to exposed to a 5-alpha reductase inhibitor developed persistent erectile dysfunction after a longer duration of exposure (median, 1,534 days). compared with a shorter duration of exposure (PeerJ. 2017 Mar 9;5:e3020).
The bottom line when considering use of 5-alpha reductase inhibitors in men is to discuss the outlier data on persistent sexual dysfunction in the studies, ask patients whether they have a history of sexual dysfunction and depression, and then only treat appropriate patients with no such history, Dr. McMichael said.
Use of 5-alpha reductase inhibitors also appears to be related to an increased risk of type 2 diabetes in men with benign prostatic hyperplasia, according to more recent data. In a study of patients in the U.K. Clinical Practice Research Datalink and Taiwanese National Health Insurance Research Database who received dutasteride, finasteride, or the alpha blocker tamsulosin, there was a slightly increased risk of type 2 diabetes among those who took the two 5-alpha reductase inhibitors, compared with those on tamsulosin (BMJ. 2019;365:l1204). In light of these results, Dr. McMichael advised clinicians to be aware of these risks and to consider screening patients for type 2 diabetes and ask them about their family history, but the results shouldn’t affect patients at risk for type 2 diabetes or metabolic disorder.
JAK inhibitors for alopecia areata
Within the past few years, promising results for Janus kinase (JAK) inhibitors like tofacitinib and ruxolitinib for alopecia areata have been reported, but they are currently not a first-line therapy, Dr. McMichael said. Consider methotrexate first in older adolescents and adults with more than 50% hair loss, followed by a JAK inhibitor if there is no improvement or methotrexate is not tolerated well.
Clinicians can consider enrolling their patients in a clinical trial to give them access to JAK inhibitors as a treatment option, she noted, and if a trial is not available, it may be worth appealing to an insurance company using an article titled “Alopecia areata is a medical disease” coauthored by Dr. McMichael and others (Am Acad Dermatol. 2018 Apr;78[4]:832-4). After two denials by insurance, the manufacturer’s patient assistance program (Xelsource) may be helpful in obtaining tofacitinib (Xeljanz) through a letter and references. There are adolescent data on JAK inhibitors for alopecia, but “absolutely no data” in very young children, so prior to adolescence, she would not recommend this treatment. In a study of 13 adolescents with alopecia areata, totalis, or universalis, those treated with tofacitinib for a mean of 6.5 months, 9 had significant hair regrowth with treatment and adverse events were mild (J Am Acad Dermatol. 2017 Jan;76[1]:29-32).
Dr. McMichael reports being an investigator for Allergan, Intendis, Proctor & Gamble, Samumed, Cassiopia, Concert, Aclaris, and Incyte; and is a consultant for Johnson & Johnson, Proctor & Gamble, Allergan, Bayer, Galderma, Incyte, Samumed, Aclaris, Anacor, Pfizer, Nutrafol, Bioniz, and Almirall.
ORLANDO – Biotin is the most popular consumer supplement for alopecia and highly popular online, but should patients be taking it?
Patients may want something “natural” to treat their hair loss, but “natural is not always good,” Amy McMichael, MD, professor and chair of the department of dermatology at Wake Forest Baptist Health, Winston-Salem, N.C., said at the ODAC Dermatology, Aesthetic, & Surgical Conference.
Dosages of commercially available biotin supplements can vary significantly, with some doses as high as 10,000 mcg, making supraphysiological dosing possible. Dr. McMichael said that, not only is biotin unlikely to help with a patient’s alopecia, but a high intake of biotin can interfere with certain assays that use streptavidin-biotin capture techniques (Clin Chem Lab Med. 2017 May 1;55[6]:817-25). This could present a problem for a patient who experiences an MI or has a thyroid disorder where a high level of biotin could affect lab results, she noted.
Dr. McMichael advises patients that there is a
Questioning side effects of 5-alpha reductase inhibitors
Research in the early 2010s associated the 5-alpha reductase inhibitor finasteride with persistent sexual side effects and depression. But a later meta-analysis of the Prostate Cancer Prevention Trial and other trials did not find evidence of persistent sexual side effects or depression in men on finasteride, and the authors said that double-blind, placebo-controlled studies were needed (J Clin Aesthet Dermatol. 2014 Dec;7[12]:51-5).
However, two meta-analyses of 34 clinical trials published in 2015 found that none of the clinical trials evaluating finasteride treatment in patients with androgenic alopecia had accurate safety reporting (JAMA Dermatol. 2015 Jun;151[6]:600-6). Another study published by the same group in 2017 found that 0.8% of men aged 16-42 years to exposed to a 5-alpha reductase inhibitor developed persistent erectile dysfunction after a longer duration of exposure (median, 1,534 days). compared with a shorter duration of exposure (PeerJ. 2017 Mar 9;5:e3020).
The bottom line when considering use of 5-alpha reductase inhibitors in men is to discuss the outlier data on persistent sexual dysfunction in the studies, ask patients whether they have a history of sexual dysfunction and depression, and then only treat appropriate patients with no such history, Dr. McMichael said.
Use of 5-alpha reductase inhibitors also appears to be related to an increased risk of type 2 diabetes in men with benign prostatic hyperplasia, according to more recent data. In a study of patients in the U.K. Clinical Practice Research Datalink and Taiwanese National Health Insurance Research Database who received dutasteride, finasteride, or the alpha blocker tamsulosin, there was a slightly increased risk of type 2 diabetes among those who took the two 5-alpha reductase inhibitors, compared with those on tamsulosin (BMJ. 2019;365:l1204). In light of these results, Dr. McMichael advised clinicians to be aware of these risks and to consider screening patients for type 2 diabetes and ask them about their family history, but the results shouldn’t affect patients at risk for type 2 diabetes or metabolic disorder.
JAK inhibitors for alopecia areata
Within the past few years, promising results for Janus kinase (JAK) inhibitors like tofacitinib and ruxolitinib for alopecia areata have been reported, but they are currently not a first-line therapy, Dr. McMichael said. Consider methotrexate first in older adolescents and adults with more than 50% hair loss, followed by a JAK inhibitor if there is no improvement or methotrexate is not tolerated well.
Clinicians can consider enrolling their patients in a clinical trial to give them access to JAK inhibitors as a treatment option, she noted, and if a trial is not available, it may be worth appealing to an insurance company using an article titled “Alopecia areata is a medical disease” coauthored by Dr. McMichael and others (Am Acad Dermatol. 2018 Apr;78[4]:832-4). After two denials by insurance, the manufacturer’s patient assistance program (Xelsource) may be helpful in obtaining tofacitinib (Xeljanz) through a letter and references. There are adolescent data on JAK inhibitors for alopecia, but “absolutely no data” in very young children, so prior to adolescence, she would not recommend this treatment. In a study of 13 adolescents with alopecia areata, totalis, or universalis, those treated with tofacitinib for a mean of 6.5 months, 9 had significant hair regrowth with treatment and adverse events were mild (J Am Acad Dermatol. 2017 Jan;76[1]:29-32).
Dr. McMichael reports being an investigator for Allergan, Intendis, Proctor & Gamble, Samumed, Cassiopia, Concert, Aclaris, and Incyte; and is a consultant for Johnson & Johnson, Proctor & Gamble, Allergan, Bayer, Galderma, Incyte, Samumed, Aclaris, Anacor, Pfizer, Nutrafol, Bioniz, and Almirall.
EXPERT ANALYSIS FROM ODAC 2020
Emollients didn’t prevent atopic dermatitis in high-risk infants
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
FROM THE LANCET
Nail growth
The treatment failure with the terbinafine made onychomycosis unlikely, and the appearance of the finger did not suggest that this was a wart. So, the physician opted for a 4-mm punch biopsy of the lateral nail fold, which confirmed that this was a well-differentiated squamous cell carcinoma (SCC) of the fingertip.
Periungual SCC is twice as common in men as women. Lesions tend to appear as hyperkeratotic plaques or nodules, pushing the nail plate away from the nail bed. With onychomycosis, one would expect the nail to be more thickened and discolored. A wart would not be as keratotic as this lesion was, and there would be thrombosed capillaries on closer inspection.
SCC is the second most common skin cancer in humans. It is most common on sun-exposed areas but may present in sites not exposed to the sun. It has been hypothesized that high risk human papillomavirus (HPV) types—particularly HPV 16—may contribute to diseases of the fingertips and nail unit in older adults. (There was no known history of HPV in this patient.)
A surgical approach often is curative. Achieving appropriate margins occasionally requires partial amputation. Mohs micrographic surgery (MMS) offers the highest cure rate and spares as much uninvolved tissue as possible. Radiation therapy is another tissue-sparing technique. It requires 15 to 30 sessions over 3 to 6 weeks and has a lower cure rate than MMS.
In this case, the patient underwent MMS. Follow-up skin surveillance exams revealed other small nonmelanoma skin cancers at other sites. The patient also developed a dystrophic nail spicule near the surgical site that was re-excised and deemed benign.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Riddel C, Rashid R, Thomas V. Ungual and periungual human papillomavirus-associated squamous cell carcinoma: a review. J Am Acad Dermatol. 2011 Jun;64:1147-1153.

The treatment failure with the terbinafine made onychomycosis unlikely, and the appearance of the finger did not suggest that this was a wart. So, the physician opted for a 4-mm punch biopsy of the lateral nail fold, which confirmed that this was a well-differentiated squamous cell carcinoma (SCC) of the fingertip.
Periungual SCC is twice as common in men as women. Lesions tend to appear as hyperkeratotic plaques or nodules, pushing the nail plate away from the nail bed. With onychomycosis, one would expect the nail to be more thickened and discolored. A wart would not be as keratotic as this lesion was, and there would be thrombosed capillaries on closer inspection.
SCC is the second most common skin cancer in humans. It is most common on sun-exposed areas but may present in sites not exposed to the sun. It has been hypothesized that high risk human papillomavirus (HPV) types—particularly HPV 16—may contribute to diseases of the fingertips and nail unit in older adults. (There was no known history of HPV in this patient.)
A surgical approach often is curative. Achieving appropriate margins occasionally requires partial amputation. Mohs micrographic surgery (MMS) offers the highest cure rate and spares as much uninvolved tissue as possible. Radiation therapy is another tissue-sparing technique. It requires 15 to 30 sessions over 3 to 6 weeks and has a lower cure rate than MMS.
In this case, the patient underwent MMS. Follow-up skin surveillance exams revealed other small nonmelanoma skin cancers at other sites. The patient also developed a dystrophic nail spicule near the surgical site that was re-excised and deemed benign.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The treatment failure with the terbinafine made onychomycosis unlikely, and the appearance of the finger did not suggest that this was a wart. So, the physician opted for a 4-mm punch biopsy of the lateral nail fold, which confirmed that this was a well-differentiated squamous cell carcinoma (SCC) of the fingertip.
Periungual SCC is twice as common in men as women. Lesions tend to appear as hyperkeratotic plaques or nodules, pushing the nail plate away from the nail bed. With onychomycosis, one would expect the nail to be more thickened and discolored. A wart would not be as keratotic as this lesion was, and there would be thrombosed capillaries on closer inspection.
SCC is the second most common skin cancer in humans. It is most common on sun-exposed areas but may present in sites not exposed to the sun. It has been hypothesized that high risk human papillomavirus (HPV) types—particularly HPV 16—may contribute to diseases of the fingertips and nail unit in older adults. (There was no known history of HPV in this patient.)
A surgical approach often is curative. Achieving appropriate margins occasionally requires partial amputation. Mohs micrographic surgery (MMS) offers the highest cure rate and spares as much uninvolved tissue as possible. Radiation therapy is another tissue-sparing technique. It requires 15 to 30 sessions over 3 to 6 weeks and has a lower cure rate than MMS.
In this case, the patient underwent MMS. Follow-up skin surveillance exams revealed other small nonmelanoma skin cancers at other sites. The patient also developed a dystrophic nail spicule near the surgical site that was re-excised and deemed benign.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Riddel C, Rashid R, Thomas V. Ungual and periungual human papillomavirus-associated squamous cell carcinoma: a review. J Am Acad Dermatol. 2011 Jun;64:1147-1153.
Riddel C, Rashid R, Thomas V. Ungual and periungual human papillomavirus-associated squamous cell carcinoma: a review. J Am Acad Dermatol. 2011 Jun;64:1147-1153.