User login
Pink plaque on the ear
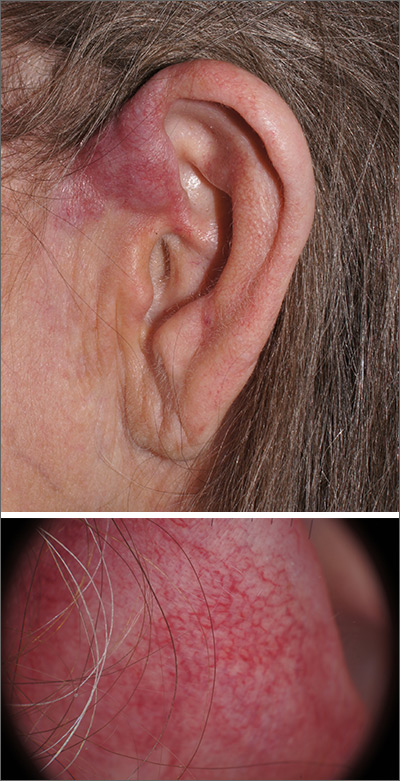
A 4-mm punch biopsy was performed and revealed B-cell lymphoma, consistent with extranodal marginal zone lymphoma. The plaque was palpated carefully and the location of branches of the superficial temporal artery, which usually course anterior to the helix, were mapped, and avoided as a biopsy site.
Marginal zone lymphoma is a relatively indolent B-cell lymphoma that occurs in adults in mucosal associated lymphoid tissue, most often in the gastrointestinal (GI) tract. Neoplasms also occur in the lungs, eyes, and skin. Initial symptoms vary according to the site of manifestation. Patients with GI tumors may present with GI bleeding, abdominal pain, and weight loss. Pulmonary lesions are often asymptomatic and picked up on chest imaging for other indications. Chronic gastritis associated with Helicobacter pylori contributes to cases and occasionally eradication of H. pylori may clear patients of disease. Autoimmune diseases, particularly Sjögren disease and Hashimoto thyroiditis, also have a causative association. Rarely, transformation into high-grade disease occurs.
On the skin, marginal zone lymphoma may be exhibited as soft salmon-colored patches with serpentine vascular markings, as seen here on dermoscopy.1 The differential diagnosis includes keloid scar, basal cell carcinoma, Merkel cell carcinoma, arthropod bites, and amelanotic melanoma.
This patient had been under the care of Medical Oncology for surveillance and was offered observation as a potential treatment strategy because of the relatively indolent nature of the tumor. However, because the tumor was painful and growing, she opted for focal palliative radiation therapy.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Geller S, Marghoob AA, Scope A, et al. Dermoscopy and the diagnosis of primary cutaneous B-cell lymphoma. J Eur Acad Dermatol Venereol. 2018;32:53-56. doi:10.1111/jdv.14549

A 4-mm punch biopsy was performed and revealed B-cell lymphoma, consistent with extranodal marginal zone lymphoma. The plaque was palpated carefully and the location of branches of the superficial temporal artery, which usually course anterior to the helix, were mapped, and avoided as a biopsy site.
Marginal zone lymphoma is a relatively indolent B-cell lymphoma that occurs in adults in mucosal associated lymphoid tissue, most often in the gastrointestinal (GI) tract. Neoplasms also occur in the lungs, eyes, and skin. Initial symptoms vary according to the site of manifestation. Patients with GI tumors may present with GI bleeding, abdominal pain, and weight loss. Pulmonary lesions are often asymptomatic and picked up on chest imaging for other indications. Chronic gastritis associated with Helicobacter pylori contributes to cases and occasionally eradication of H. pylori may clear patients of disease. Autoimmune diseases, particularly Sjögren disease and Hashimoto thyroiditis, also have a causative association. Rarely, transformation into high-grade disease occurs.
On the skin, marginal zone lymphoma may be exhibited as soft salmon-colored patches with serpentine vascular markings, as seen here on dermoscopy.1 The differential diagnosis includes keloid scar, basal cell carcinoma, Merkel cell carcinoma, arthropod bites, and amelanotic melanoma.
This patient had been under the care of Medical Oncology for surveillance and was offered observation as a potential treatment strategy because of the relatively indolent nature of the tumor. However, because the tumor was painful and growing, she opted for focal palliative radiation therapy.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

A 4-mm punch biopsy was performed and revealed B-cell lymphoma, consistent with extranodal marginal zone lymphoma. The plaque was palpated carefully and the location of branches of the superficial temporal artery, which usually course anterior to the helix, were mapped, and avoided as a biopsy site.
Marginal zone lymphoma is a relatively indolent B-cell lymphoma that occurs in adults in mucosal associated lymphoid tissue, most often in the gastrointestinal (GI) tract. Neoplasms also occur in the lungs, eyes, and skin. Initial symptoms vary according to the site of manifestation. Patients with GI tumors may present with GI bleeding, abdominal pain, and weight loss. Pulmonary lesions are often asymptomatic and picked up on chest imaging for other indications. Chronic gastritis associated with Helicobacter pylori contributes to cases and occasionally eradication of H. pylori may clear patients of disease. Autoimmune diseases, particularly Sjögren disease and Hashimoto thyroiditis, also have a causative association. Rarely, transformation into high-grade disease occurs.
On the skin, marginal zone lymphoma may be exhibited as soft salmon-colored patches with serpentine vascular markings, as seen here on dermoscopy.1 The differential diagnosis includes keloid scar, basal cell carcinoma, Merkel cell carcinoma, arthropod bites, and amelanotic melanoma.
This patient had been under the care of Medical Oncology for surveillance and was offered observation as a potential treatment strategy because of the relatively indolent nature of the tumor. However, because the tumor was painful and growing, she opted for focal palliative radiation therapy.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Geller S, Marghoob AA, Scope A, et al. Dermoscopy and the diagnosis of primary cutaneous B-cell lymphoma. J Eur Acad Dermatol Venereol. 2018;32:53-56. doi:10.1111/jdv.14549
1. Geller S, Marghoob AA, Scope A, et al. Dermoscopy and the diagnosis of primary cutaneous B-cell lymphoma. J Eur Acad Dermatol Venereol. 2018;32:53-56. doi:10.1111/jdv.14549
Itch response faster with abrocitinib in trial comparing JAK inhibitor to dupilumab
, in a multicenter, randomized trial.
In addition, in the study, those on 200-mg and 100-mg daily doses of abrocitinib experienced significantly greater reductions in signs and symptoms of AD at 12 and 16 weeks, than those on placebo, the authors reported.
The findings from the JADE COMPARE trial, published on March 25 in the New England Journal of Medicine, suggest abrocitinib will provide clinicians with another treatment option for patients who don’t get adequate relief from either topical medications or dupilumab. Abrocitinib is associated with a different set of adverse reactions than dupilumab, according to investigators.
In 2017, dupilumab (Dupixent) became the first systemic drug approved by the Food and Drug Administration specifically for AD, though systemic steroids and other immunosuppressant drugs are sometimes prescribed. A monoclonal antibody delivered by subcutaneous injection, dupilumab binds to interleukin-4 receptors to block signaling pathways involved in AD; it is now approved for treatment of patients with moderate to severe AD down to age 6 years.
“It is sort of the bar for efficacy and for safety in those patients, because that’s what we have right now,” said one of the JADE Compare investigators and study author, Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology and director of clinical research and contact dermatitis at George Washington University, Washington, said in an interview. “For any new therapy coming to market, we really do want to understand how it compares to what’s out there.”
Abrocitinib is a small molecule that inhibits JAK1, which is thought to modulate multiple cytokines involved in AD, including interleukin (IL)–4, IL-13, IL-31, IL-22, and thymic stromal lymphopoietin. Two other JAK1 inhibitors, baricitinib and upadacitinib, are also being investigated as systemic treatments for AD.
In JADE COMPARE, people with moderate to severe AD from 18 countries on four continents, entered a 28-day screening period during which they discontinued treatments. They began using emollients twice a day at least 7 days before being randomly assigned to a treatment group, and continued on topical medication once daily. Topical treatments included low- or medium-potency topical glucocorticoids, calcineurin inhibitors, and phosphodiesterase-4 inhibitors.
The researchers randomly assigned 838 to trial groups: 226 received 200 mg of abrocitinib orally once a day, 238 received 100 mg of abrocitinib once a day, 243 received a 300-mg dupilumab injection every other week, and 131 received placebo versions of both medications, for 16 weeks. The mean age of the patients overall was about 38 years; about two-thirds were White.
At 2 weeks, half of the patients on 200 mg of abrocitinib and 31.8% of those on the 100-mg dose had an itch response, defined as at least a 4-point improvement from baseline in the 0-10 Peak Pruritus Numerical Rating Scale. This was compared with 26.4% of those on dupilumab and 13.8% of those on placebo.
And at 12 weeks, more of the patients in the 200-mg abrocitinib group than in the other groups had an Investigator’s Global Assessment (IGA) response (defined as clear or almost clear) and more had an Eczema Area and Severity Index (EASI-75) response (defined as an improvement of at least 75%). (See Table) EASI-75 and IGA responses at week 12 were the primary outcomes of the study.
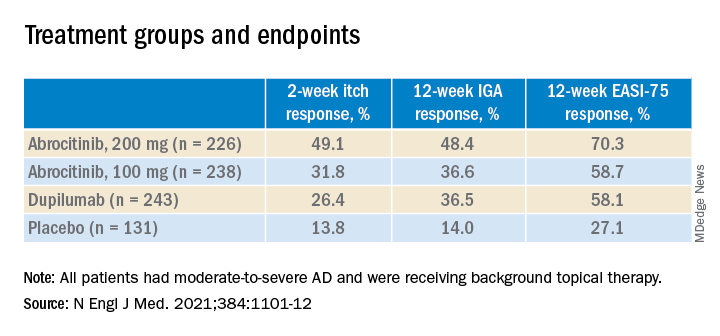
The differences between both abrocitinib groups and the placebo group were statistically significant by all these measures (P < .001). The difference between the 200-mg abrocitinib and the dupilumab group was only significant for itch at 2 weeks, and the difference in itch response between the 100-mg group and the dupilumab group at 2 weeks was not significant (P < .20).
At 16 weeks, the EASI-75 response (a secondary endpoint) among those on either dose of abrocitinib was not significantly different than among those on dupilumab (71% and 60.3% among those on 200 mg and 100 mg, respectively; and 65.5% among those on dupilumab, compared with 30.6% of those on placebo).
“The patients I have on this medicine [abrocitinib] are very happy,” said one of the study authors, Melinda Gooderham, MsC, MD, an assistant professor at Queen’s University, Kingston, Ont., an investigator in the trial. “It works very quickly for itch,” she said in an interview.
The study didn’t have sufficient statistical power to fully explore the comparison to dupilumab, and future trials will go deeper into the comparison, she added.
Still, in this trial, abrocitinib demonstrated a clear advantage in the speed and depth of efficacy, Dr. Silverberg noted. “The 100-mg dose of abrocitinib was about as effective as, or maybe slightly less effective than, dupilumab, and the 200-mg dose was more effective than dupilumab.”
The overall incidence of adverse events was higher in the 200-mg abrocitinib arm than in the other groups, but the incidence of serious or severe adverse events, and the incidence of adverse events that resulted in discontinuing the medication, were similar across the trial groups.
However, nausea affected 11.1% of the patients in the 200-mg abrocitinib group and 4.2% of those in the 100-mg abrocitinib group. Acne was also reported in these groups (6.6% and 2.9%, among those on 200 mg and 100 mg, respectively, compared with 1.2% of those on dupilumab and none of those on placebo). In a few of those on abrocitinib, herpes zoster flared up. And median platelet counts decreased among the patients taking abrocitinib, although none dropped below 75,000/mm3. Serious infections were reported in two patients on abrocitinib, but resolved.
By contrast, only 2.9% of the patients on dupilumab had nausea. But 6.2% in the dupilumab group had conjunctivitis, compared with 1.3% of patients in the 200-mg abrocitinib group and 0.8 in the 100-mg abrocitinib group.
As an oral medication, abrocitinib will appeal to patients who want to avoid injections, and dosing will be easier to adjust, Dr. Silverberg said. On the other hand, he added, dupilumab will have an advantage for patients who don’t want to take a daily medication, or who are concerned about the adverse events associated with abrocitinib, particularly those with blood-clotting disorders.
On the basis of two previous JADE phase 3 trials, Pfizer has submitted a new drug application for abrocitinib for treating moderate to severe AD in patients aged 12 and older to the FDA; a decision is expected in April, according to the company. The company has also applied to market the drug in Europe and the United Kingdom.
The study was funded by Pfizer. Dr. Silverberg’s disclosures included serving as a consultant to companies including AbbVie, Pfizer, and Regeneron. Several authors are Pfizer employees; other authors had disclosures related to Pfizer and other pharmaceutical companies.
, in a multicenter, randomized trial.
In addition, in the study, those on 200-mg and 100-mg daily doses of abrocitinib experienced significantly greater reductions in signs and symptoms of AD at 12 and 16 weeks, than those on placebo, the authors reported.
The findings from the JADE COMPARE trial, published on March 25 in the New England Journal of Medicine, suggest abrocitinib will provide clinicians with another treatment option for patients who don’t get adequate relief from either topical medications or dupilumab. Abrocitinib is associated with a different set of adverse reactions than dupilumab, according to investigators.
In 2017, dupilumab (Dupixent) became the first systemic drug approved by the Food and Drug Administration specifically for AD, though systemic steroids and other immunosuppressant drugs are sometimes prescribed. A monoclonal antibody delivered by subcutaneous injection, dupilumab binds to interleukin-4 receptors to block signaling pathways involved in AD; it is now approved for treatment of patients with moderate to severe AD down to age 6 years.
“It is sort of the bar for efficacy and for safety in those patients, because that’s what we have right now,” said one of the JADE Compare investigators and study author, Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology and director of clinical research and contact dermatitis at George Washington University, Washington, said in an interview. “For any new therapy coming to market, we really do want to understand how it compares to what’s out there.”
Abrocitinib is a small molecule that inhibits JAK1, which is thought to modulate multiple cytokines involved in AD, including interleukin (IL)–4, IL-13, IL-31, IL-22, and thymic stromal lymphopoietin. Two other JAK1 inhibitors, baricitinib and upadacitinib, are also being investigated as systemic treatments for AD.
In JADE COMPARE, people with moderate to severe AD from 18 countries on four continents, entered a 28-day screening period during which they discontinued treatments. They began using emollients twice a day at least 7 days before being randomly assigned to a treatment group, and continued on topical medication once daily. Topical treatments included low- or medium-potency topical glucocorticoids, calcineurin inhibitors, and phosphodiesterase-4 inhibitors.
The researchers randomly assigned 838 to trial groups: 226 received 200 mg of abrocitinib orally once a day, 238 received 100 mg of abrocitinib once a day, 243 received a 300-mg dupilumab injection every other week, and 131 received placebo versions of both medications, for 16 weeks. The mean age of the patients overall was about 38 years; about two-thirds were White.
At 2 weeks, half of the patients on 200 mg of abrocitinib and 31.8% of those on the 100-mg dose had an itch response, defined as at least a 4-point improvement from baseline in the 0-10 Peak Pruritus Numerical Rating Scale. This was compared with 26.4% of those on dupilumab and 13.8% of those on placebo.
And at 12 weeks, more of the patients in the 200-mg abrocitinib group than in the other groups had an Investigator’s Global Assessment (IGA) response (defined as clear or almost clear) and more had an Eczema Area and Severity Index (EASI-75) response (defined as an improvement of at least 75%). (See Table) EASI-75 and IGA responses at week 12 were the primary outcomes of the study.

The differences between both abrocitinib groups and the placebo group were statistically significant by all these measures (P < .001). The difference between the 200-mg abrocitinib and the dupilumab group was only significant for itch at 2 weeks, and the difference in itch response between the 100-mg group and the dupilumab group at 2 weeks was not significant (P < .20).
At 16 weeks, the EASI-75 response (a secondary endpoint) among those on either dose of abrocitinib was not significantly different than among those on dupilumab (71% and 60.3% among those on 200 mg and 100 mg, respectively; and 65.5% among those on dupilumab, compared with 30.6% of those on placebo).
“The patients I have on this medicine [abrocitinib] are very happy,” said one of the study authors, Melinda Gooderham, MsC, MD, an assistant professor at Queen’s University, Kingston, Ont., an investigator in the trial. “It works very quickly for itch,” she said in an interview.
The study didn’t have sufficient statistical power to fully explore the comparison to dupilumab, and future trials will go deeper into the comparison, she added.
Still, in this trial, abrocitinib demonstrated a clear advantage in the speed and depth of efficacy, Dr. Silverberg noted. “The 100-mg dose of abrocitinib was about as effective as, or maybe slightly less effective than, dupilumab, and the 200-mg dose was more effective than dupilumab.”
The overall incidence of adverse events was higher in the 200-mg abrocitinib arm than in the other groups, but the incidence of serious or severe adverse events, and the incidence of adverse events that resulted in discontinuing the medication, were similar across the trial groups.
However, nausea affected 11.1% of the patients in the 200-mg abrocitinib group and 4.2% of those in the 100-mg abrocitinib group. Acne was also reported in these groups (6.6% and 2.9%, among those on 200 mg and 100 mg, respectively, compared with 1.2% of those on dupilumab and none of those on placebo). In a few of those on abrocitinib, herpes zoster flared up. And median platelet counts decreased among the patients taking abrocitinib, although none dropped below 75,000/mm3. Serious infections were reported in two patients on abrocitinib, but resolved.
By contrast, only 2.9% of the patients on dupilumab had nausea. But 6.2% in the dupilumab group had conjunctivitis, compared with 1.3% of patients in the 200-mg abrocitinib group and 0.8 in the 100-mg abrocitinib group.
As an oral medication, abrocitinib will appeal to patients who want to avoid injections, and dosing will be easier to adjust, Dr. Silverberg said. On the other hand, he added, dupilumab will have an advantage for patients who don’t want to take a daily medication, or who are concerned about the adverse events associated with abrocitinib, particularly those with blood-clotting disorders.
On the basis of two previous JADE phase 3 trials, Pfizer has submitted a new drug application for abrocitinib for treating moderate to severe AD in patients aged 12 and older to the FDA; a decision is expected in April, according to the company. The company has also applied to market the drug in Europe and the United Kingdom.
The study was funded by Pfizer. Dr. Silverberg’s disclosures included serving as a consultant to companies including AbbVie, Pfizer, and Regeneron. Several authors are Pfizer employees; other authors had disclosures related to Pfizer and other pharmaceutical companies.
, in a multicenter, randomized trial.
In addition, in the study, those on 200-mg and 100-mg daily doses of abrocitinib experienced significantly greater reductions in signs and symptoms of AD at 12 and 16 weeks, than those on placebo, the authors reported.
The findings from the JADE COMPARE trial, published on March 25 in the New England Journal of Medicine, suggest abrocitinib will provide clinicians with another treatment option for patients who don’t get adequate relief from either topical medications or dupilumab. Abrocitinib is associated with a different set of adverse reactions than dupilumab, according to investigators.
In 2017, dupilumab (Dupixent) became the first systemic drug approved by the Food and Drug Administration specifically for AD, though systemic steroids and other immunosuppressant drugs are sometimes prescribed. A monoclonal antibody delivered by subcutaneous injection, dupilumab binds to interleukin-4 receptors to block signaling pathways involved in AD; it is now approved for treatment of patients with moderate to severe AD down to age 6 years.
“It is sort of the bar for efficacy and for safety in those patients, because that’s what we have right now,” said one of the JADE Compare investigators and study author, Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology and director of clinical research and contact dermatitis at George Washington University, Washington, said in an interview. “For any new therapy coming to market, we really do want to understand how it compares to what’s out there.”
Abrocitinib is a small molecule that inhibits JAK1, which is thought to modulate multiple cytokines involved in AD, including interleukin (IL)–4, IL-13, IL-31, IL-22, and thymic stromal lymphopoietin. Two other JAK1 inhibitors, baricitinib and upadacitinib, are also being investigated as systemic treatments for AD.
In JADE COMPARE, people with moderate to severe AD from 18 countries on four continents, entered a 28-day screening period during which they discontinued treatments. They began using emollients twice a day at least 7 days before being randomly assigned to a treatment group, and continued on topical medication once daily. Topical treatments included low- or medium-potency topical glucocorticoids, calcineurin inhibitors, and phosphodiesterase-4 inhibitors.
The researchers randomly assigned 838 to trial groups: 226 received 200 mg of abrocitinib orally once a day, 238 received 100 mg of abrocitinib once a day, 243 received a 300-mg dupilumab injection every other week, and 131 received placebo versions of both medications, for 16 weeks. The mean age of the patients overall was about 38 years; about two-thirds were White.
At 2 weeks, half of the patients on 200 mg of abrocitinib and 31.8% of those on the 100-mg dose had an itch response, defined as at least a 4-point improvement from baseline in the 0-10 Peak Pruritus Numerical Rating Scale. This was compared with 26.4% of those on dupilumab and 13.8% of those on placebo.
And at 12 weeks, more of the patients in the 200-mg abrocitinib group than in the other groups had an Investigator’s Global Assessment (IGA) response (defined as clear or almost clear) and more had an Eczema Area and Severity Index (EASI-75) response (defined as an improvement of at least 75%). (See Table) EASI-75 and IGA responses at week 12 were the primary outcomes of the study.

The differences between both abrocitinib groups and the placebo group were statistically significant by all these measures (P < .001). The difference between the 200-mg abrocitinib and the dupilumab group was only significant for itch at 2 weeks, and the difference in itch response between the 100-mg group and the dupilumab group at 2 weeks was not significant (P < .20).
At 16 weeks, the EASI-75 response (a secondary endpoint) among those on either dose of abrocitinib was not significantly different than among those on dupilumab (71% and 60.3% among those on 200 mg and 100 mg, respectively; and 65.5% among those on dupilumab, compared with 30.6% of those on placebo).
“The patients I have on this medicine [abrocitinib] are very happy,” said one of the study authors, Melinda Gooderham, MsC, MD, an assistant professor at Queen’s University, Kingston, Ont., an investigator in the trial. “It works very quickly for itch,” she said in an interview.
The study didn’t have sufficient statistical power to fully explore the comparison to dupilumab, and future trials will go deeper into the comparison, she added.
Still, in this trial, abrocitinib demonstrated a clear advantage in the speed and depth of efficacy, Dr. Silverberg noted. “The 100-mg dose of abrocitinib was about as effective as, or maybe slightly less effective than, dupilumab, and the 200-mg dose was more effective than dupilumab.”
The overall incidence of adverse events was higher in the 200-mg abrocitinib arm than in the other groups, but the incidence of serious or severe adverse events, and the incidence of adverse events that resulted in discontinuing the medication, were similar across the trial groups.
However, nausea affected 11.1% of the patients in the 200-mg abrocitinib group and 4.2% of those in the 100-mg abrocitinib group. Acne was also reported in these groups (6.6% and 2.9%, among those on 200 mg and 100 mg, respectively, compared with 1.2% of those on dupilumab and none of those on placebo). In a few of those on abrocitinib, herpes zoster flared up. And median platelet counts decreased among the patients taking abrocitinib, although none dropped below 75,000/mm3. Serious infections were reported in two patients on abrocitinib, but resolved.
By contrast, only 2.9% of the patients on dupilumab had nausea. But 6.2% in the dupilumab group had conjunctivitis, compared with 1.3% of patients in the 200-mg abrocitinib group and 0.8 in the 100-mg abrocitinib group.
As an oral medication, abrocitinib will appeal to patients who want to avoid injections, and dosing will be easier to adjust, Dr. Silverberg said. On the other hand, he added, dupilumab will have an advantage for patients who don’t want to take a daily medication, or who are concerned about the adverse events associated with abrocitinib, particularly those with blood-clotting disorders.
On the basis of two previous JADE phase 3 trials, Pfizer has submitted a new drug application for abrocitinib for treating moderate to severe AD in patients aged 12 and older to the FDA; a decision is expected in April, according to the company. The company has also applied to market the drug in Europe and the United Kingdom.
The study was funded by Pfizer. Dr. Silverberg’s disclosures included serving as a consultant to companies including AbbVie, Pfizer, and Regeneron. Several authors are Pfizer employees; other authors had disclosures related to Pfizer and other pharmaceutical companies.
Ruxolitinib cream for atopic dermatitis is in regulatory home stretch
, demonstrated a dual mechanism of action in two pivotal phase 3 trials: antipruritic and anti-inflammatory, Kim A. Papp, MD, PhD, said at Innovations in Dermatology: Virtual Spring Conference 2021. He presented a pooled analysis of the TRuE-AD1 and TRuE-AD2 trials, in which 1,249 patients with AD affecting 3%-20% of the body surface area were randomized 2:2:1 double-blind to ruxolitinib cream 0.75%, 1.5%, or vehicle twice daily for 8 weeks.
Striking evidence of the drug’s antipruritic effect comes from the finding that patient-reported itch scores separated significantly from the vehicle controls within just 12 hours after the first application. The margin of difference grew over time such that at 4 weeks, 48.5% of patients on ruxolitinib 1.5% experienced a clinically meaningful reduction in itch – defined by at least a 4-point improvement on the itch numeric rating scale – as did 30.1% of those on ruxolitinib 0.75% and 6.1% of controls. By week 8, these figures had further improved to 51.5%, 41.5%, and 15.8%, respectively, noted Dr. Papp, a dermatologist and president of Probity Medical Research in Waterloo, Ont.
Ruxolitinib’s anti-inflammatory mechanism of action was on display in the primary study endpoint, which was the proportion of patients achieving an Investigator Global Assessment score of 0 or 1 with at least a 2-grade improvement from baseline at week 8. The rates were 52.6% with ruxolitinib 1.5% and 44.7% at the lower dose, both significantly better than the 11.5% rate with vehicle.
For the secondary endpoint of at least a 75% improvement in Eczema Area and Severity Index score at week 8, the rates were 62% with ruxolitinib 1.5% and 53.8% at the 0.75% concentration, compared with 19.7% with vehicle.
The topical JAK inhibitor also showed superior efficacy in terms of improvement on the Patient-Reported Outcomes Measurement Information System Sleep Disturbance Score, with a clinically meaningful 6-point or greater improvement in 23.9% and 20.9% of patients in the high- and low-dose ruxolitinib groups, versus 14.2% in controls.
Plasma drug levels remained consistently low and near-flat throughout the study.
Session comoderator Lawrence F. Eichenfield, MD, was struck by what he termed the “incredibly low” rates of irritancy, burning, and stinging in the ruxolitinib-treated patients: 7 cases of application-site burning in 999 treated patients, compared with 11 cases in 250 vehicle-treated patients, and 4 cases of application-site pruritus in nearly 1,000 patients on ruxolitinib, versus 6 cases in one-fourth as many controls.
“If that’s really true in clinical practice, it would be tremendous to have a nonsteroid that doesn’t have stinging and burning and may have that efficacy,” said Dr. Eichenfield, professor of dermatology and pediatrics and vice-chair of dermatology at the University of California, San Diego.
“I think the fast action is an exciting aspect of this,” said comoderator Jonathan I. Silverberg, MD, PhD, MBA, director of clinical research and contact dermatitis in the department of dermatology at George Washington University in Washington.
He noted that in an earlier phase 2 study, ruxolitinib cream was at least as efficacious as 0.1% triamcinolone cream, providing dermatologists with a rough yardstick as to where the topical JAK inhibitor lies on the potency spectrum for AD treatment.
The FDA is expected to issue a decision on the application for approval of ruxolitinib cream in June. Dr. Eichenfield expects the drug to easily win approval. The big unanswered question is whether the regulatory agency will require boxed safety warnings, as it does for the oral JAK inhibitors approved for various indications, even though safety issues haven’t arisen with the topical agent in the clinical trials.
Dr. Papp reported receiving research grants from and serving as a consultant to Incyte Corp., which funded the ruxolitinib studies, as well as numerous other pharmaceutical companies. MedscapeLive and this news organization are owned by the same parent company.
, demonstrated a dual mechanism of action in two pivotal phase 3 trials: antipruritic and anti-inflammatory, Kim A. Papp, MD, PhD, said at Innovations in Dermatology: Virtual Spring Conference 2021. He presented a pooled analysis of the TRuE-AD1 and TRuE-AD2 trials, in which 1,249 patients with AD affecting 3%-20% of the body surface area were randomized 2:2:1 double-blind to ruxolitinib cream 0.75%, 1.5%, or vehicle twice daily for 8 weeks.
Striking evidence of the drug’s antipruritic effect comes from the finding that patient-reported itch scores separated significantly from the vehicle controls within just 12 hours after the first application. The margin of difference grew over time such that at 4 weeks, 48.5% of patients on ruxolitinib 1.5% experienced a clinically meaningful reduction in itch – defined by at least a 4-point improvement on the itch numeric rating scale – as did 30.1% of those on ruxolitinib 0.75% and 6.1% of controls. By week 8, these figures had further improved to 51.5%, 41.5%, and 15.8%, respectively, noted Dr. Papp, a dermatologist and president of Probity Medical Research in Waterloo, Ont.
Ruxolitinib’s anti-inflammatory mechanism of action was on display in the primary study endpoint, which was the proportion of patients achieving an Investigator Global Assessment score of 0 or 1 with at least a 2-grade improvement from baseline at week 8. The rates were 52.6% with ruxolitinib 1.5% and 44.7% at the lower dose, both significantly better than the 11.5% rate with vehicle.
For the secondary endpoint of at least a 75% improvement in Eczema Area and Severity Index score at week 8, the rates were 62% with ruxolitinib 1.5% and 53.8% at the 0.75% concentration, compared with 19.7% with vehicle.
The topical JAK inhibitor also showed superior efficacy in terms of improvement on the Patient-Reported Outcomes Measurement Information System Sleep Disturbance Score, with a clinically meaningful 6-point or greater improvement in 23.9% and 20.9% of patients in the high- and low-dose ruxolitinib groups, versus 14.2% in controls.
Plasma drug levels remained consistently low and near-flat throughout the study.
Session comoderator Lawrence F. Eichenfield, MD, was struck by what he termed the “incredibly low” rates of irritancy, burning, and stinging in the ruxolitinib-treated patients: 7 cases of application-site burning in 999 treated patients, compared with 11 cases in 250 vehicle-treated patients, and 4 cases of application-site pruritus in nearly 1,000 patients on ruxolitinib, versus 6 cases in one-fourth as many controls.
“If that’s really true in clinical practice, it would be tremendous to have a nonsteroid that doesn’t have stinging and burning and may have that efficacy,” said Dr. Eichenfield, professor of dermatology and pediatrics and vice-chair of dermatology at the University of California, San Diego.
“I think the fast action is an exciting aspect of this,” said comoderator Jonathan I. Silverberg, MD, PhD, MBA, director of clinical research and contact dermatitis in the department of dermatology at George Washington University in Washington.
He noted that in an earlier phase 2 study, ruxolitinib cream was at least as efficacious as 0.1% triamcinolone cream, providing dermatologists with a rough yardstick as to where the topical JAK inhibitor lies on the potency spectrum for AD treatment.
The FDA is expected to issue a decision on the application for approval of ruxolitinib cream in June. Dr. Eichenfield expects the drug to easily win approval. The big unanswered question is whether the regulatory agency will require boxed safety warnings, as it does for the oral JAK inhibitors approved for various indications, even though safety issues haven’t arisen with the topical agent in the clinical trials.
Dr. Papp reported receiving research grants from and serving as a consultant to Incyte Corp., which funded the ruxolitinib studies, as well as numerous other pharmaceutical companies. MedscapeLive and this news organization are owned by the same parent company.
, demonstrated a dual mechanism of action in two pivotal phase 3 trials: antipruritic and anti-inflammatory, Kim A. Papp, MD, PhD, said at Innovations in Dermatology: Virtual Spring Conference 2021. He presented a pooled analysis of the TRuE-AD1 and TRuE-AD2 trials, in which 1,249 patients with AD affecting 3%-20% of the body surface area were randomized 2:2:1 double-blind to ruxolitinib cream 0.75%, 1.5%, or vehicle twice daily for 8 weeks.
Striking evidence of the drug’s antipruritic effect comes from the finding that patient-reported itch scores separated significantly from the vehicle controls within just 12 hours after the first application. The margin of difference grew over time such that at 4 weeks, 48.5% of patients on ruxolitinib 1.5% experienced a clinically meaningful reduction in itch – defined by at least a 4-point improvement on the itch numeric rating scale – as did 30.1% of those on ruxolitinib 0.75% and 6.1% of controls. By week 8, these figures had further improved to 51.5%, 41.5%, and 15.8%, respectively, noted Dr. Papp, a dermatologist and president of Probity Medical Research in Waterloo, Ont.
Ruxolitinib’s anti-inflammatory mechanism of action was on display in the primary study endpoint, which was the proportion of patients achieving an Investigator Global Assessment score of 0 or 1 with at least a 2-grade improvement from baseline at week 8. The rates were 52.6% with ruxolitinib 1.5% and 44.7% at the lower dose, both significantly better than the 11.5% rate with vehicle.
For the secondary endpoint of at least a 75% improvement in Eczema Area and Severity Index score at week 8, the rates were 62% with ruxolitinib 1.5% and 53.8% at the 0.75% concentration, compared with 19.7% with vehicle.
The topical JAK inhibitor also showed superior efficacy in terms of improvement on the Patient-Reported Outcomes Measurement Information System Sleep Disturbance Score, with a clinically meaningful 6-point or greater improvement in 23.9% and 20.9% of patients in the high- and low-dose ruxolitinib groups, versus 14.2% in controls.
Plasma drug levels remained consistently low and near-flat throughout the study.
Session comoderator Lawrence F. Eichenfield, MD, was struck by what he termed the “incredibly low” rates of irritancy, burning, and stinging in the ruxolitinib-treated patients: 7 cases of application-site burning in 999 treated patients, compared with 11 cases in 250 vehicle-treated patients, and 4 cases of application-site pruritus in nearly 1,000 patients on ruxolitinib, versus 6 cases in one-fourth as many controls.
“If that’s really true in clinical practice, it would be tremendous to have a nonsteroid that doesn’t have stinging and burning and may have that efficacy,” said Dr. Eichenfield, professor of dermatology and pediatrics and vice-chair of dermatology at the University of California, San Diego.
“I think the fast action is an exciting aspect of this,” said comoderator Jonathan I. Silverberg, MD, PhD, MBA, director of clinical research and contact dermatitis in the department of dermatology at George Washington University in Washington.
He noted that in an earlier phase 2 study, ruxolitinib cream was at least as efficacious as 0.1% triamcinolone cream, providing dermatologists with a rough yardstick as to where the topical JAK inhibitor lies on the potency spectrum for AD treatment.
The FDA is expected to issue a decision on the application for approval of ruxolitinib cream in June. Dr. Eichenfield expects the drug to easily win approval. The big unanswered question is whether the regulatory agency will require boxed safety warnings, as it does for the oral JAK inhibitors approved for various indications, even though safety issues haven’t arisen with the topical agent in the clinical trials.
Dr. Papp reported receiving research grants from and serving as a consultant to Incyte Corp., which funded the ruxolitinib studies, as well as numerous other pharmaceutical companies. MedscapeLive and this news organization are owned by the same parent company.
FROM INNOVATIONS IN DERMATOLOGY
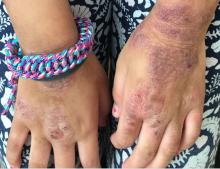
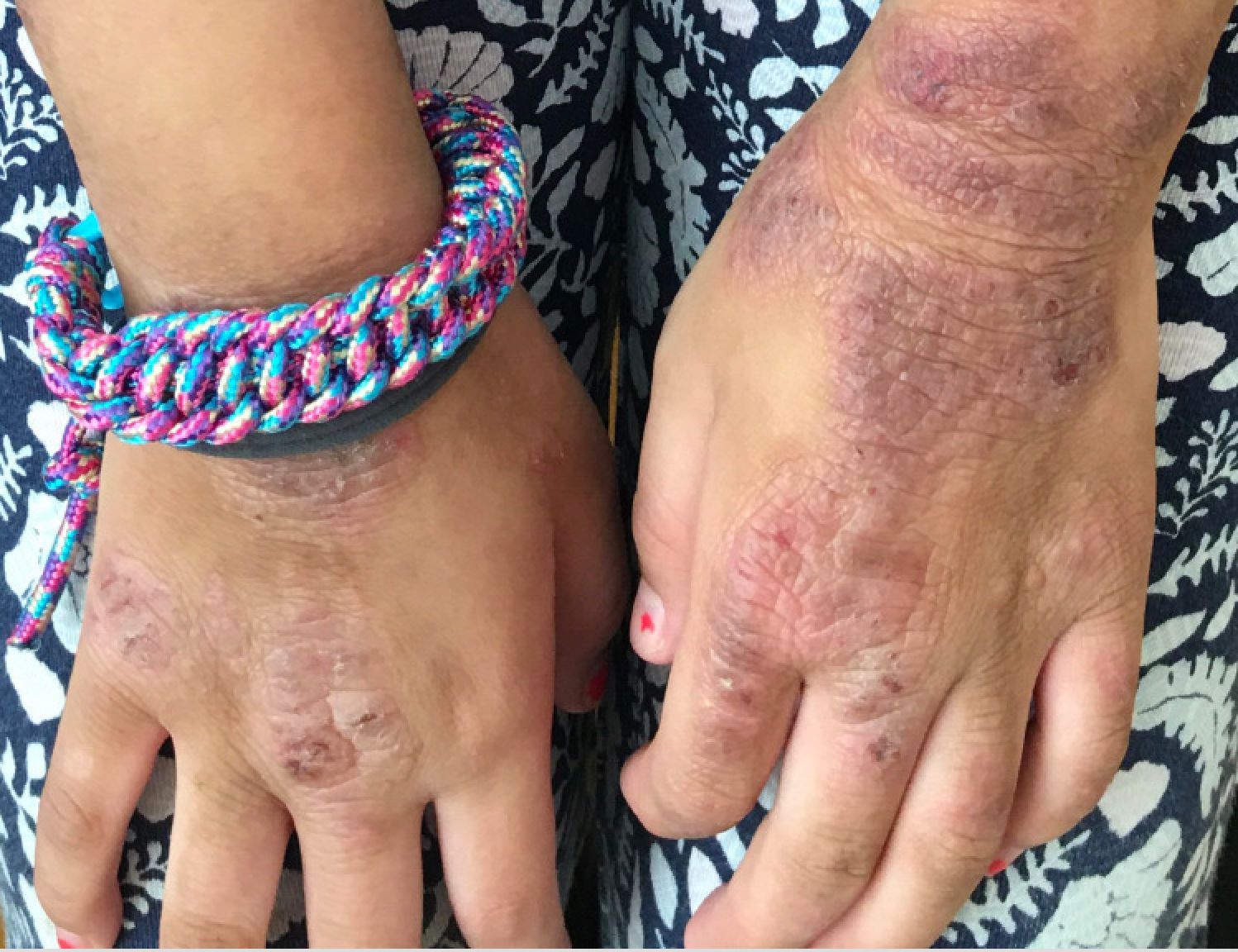
A young girl presents with ‘itchy, rashy’ hands
Given the presence of erythema, lichenification, fissuring, and scale of the hands over the course of more than 3 months with the absence of nail findings is most consistent with a diagnosis of chronic hand eczema.
Chronic hand eczema (CHE) is an inflammatory dermatitis of the hands or wrists that persists for longer than 3 months or recurs twice or more in a 12-month timespan.1,2 Hand eczema can be a manifestation of atopic dermatitis, allergic contact dermatitis, or irritant contact dermatitis. Its multifactorial pathogenesis includes epidermal injury and disturbed epidermal barrier function from exogenous factors such as irritants or contact allergens, as well as endogenous factors including atopic dermatitis.3 In pediatrics, it often presents after an acute phase of hand dermatitis with chronic pruritus, erythema, and dry skin with scale.4 Examination findings vary widely with erythema, vesicles, scale, fissures, crusting, hyperkeratosis, and/or lichenification.3,5 Diagnosis is often achieved with careful history, asking about potential exposures that may induce lesions, and physical exam of the entire skin, including the feet. Based upon clinical history or persistent dermatitis, allergic contact dermatitis patch testing should be considered.2
What’s the treatment plan?
Given that CHE is an inflammatory disease process, the goal of treatment is to reduce inflammation and allow for skin barrier repair. Unfortunately, only one study has investigated therapeutics for pediatric CHE,6 with the remainder of the literature based on adult CHE. Current CHE guidelines recommend avoidance of allergens, irritants, or other triggers of the disease as well as liberal and regular use of emollients. Because of the relative thickness of hand skin, higher-potency topical corticosteroids are often used as first-line therapy, with lower-strength topical steroids, calcineurin inhibitors, or crisaborole used as maintenance therapy. Other treatment options include phototherapy, and rarely, systemic therapies are utilized for atopic dermatitis.
What’s the differential diagnosis?
The differential diagnosis of CHE includes other scaling or hyperkeratotic skin conditions including psoriasis and tinea manuum. Other skin conditions that localize to extremities including scabies and hand-foot-and-mouth disease are discussed below.
Psoriasis can present on the hands with erythematous, well-demarcated, silver scaling plaques. However, additional plaques may be found on the elbows, knees, scalp, umbilicus, and sacrum. Nails can demonstrate pitting, oil drops, splinter hemorrhages, or onycholysis. First-line treatment includes a combination of topical steroids, topical vitamin D analogues, and keratolytics.
Tinea mannum is a dermatophyte infection of the skin of the hands. Typically, only one hand is affected with concomitant bilateral tinea pedis. It results in a white scaly plaque with dorsal hand involvement demonstrating an annular appearance, elevated edge, and central clearing. KOH prep will demonstrate septate hyphae, and cultures will grow dermatophyte colonies. Treatment includes topical antifungals or systemic antifungals for recalcitrant disease.
Scabies presents with short linear hypopigmented lesions with a black dot on one end as well as erythematous pruritic papules. These appear on the interdigital web spaces, wrists, axilla, buttocks, and genital region. Skin scraping prep with mineral oil can show mites and eggs. All individuals in an affected household should be treated with either topical permethrin or oral ivermectin to avoid reinfection or parasitic spread. All contacted linens must be cleaned with hot water and dried on high heat.
Hand-foot-and-mouth disease, classically caused by coxsackievirus, is an acute viral illness that results in an eruption of erythematous macules, papules, and vesicles on the ventral hands, soles of the feet, and oral mucosa. Diagnosis is achieved clinically and treatment is symptomatic as the lesions are self-limited.
Our patient underwent patch testing but did not return positive to any allergens. She was started on potent topical corticosteroids, educated on trigger avoidance, and gradually achieved good disease control.
Neither Mr. Haft nor Dr. Eichenfield have any relevant financial disclosures.
Michael Haft is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. He is a 4th year medical student at the University of Rochester (N.Y.). Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital.
References
1. Diepgen TL et al. Br J Dermatol. 2009;160(2):353-8.
2. Diepgen TL et al. J Dtsch Dermatol Ges. 2015;13(1):e1-22.
3. Agner T and Elsner P. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:4-12.
4. Mortz CG et al. Br J Dermatol. 2001;144(3):523-32.
5. Silvestre Salvador JF et al. Actas Dermosifiliogr. 2020;111(1):26-40.
6. Luchsinger I et al. J Eur Acad Dermatol Venereol. 2020;34(5):1037-42.
7. English J et al. Clin Exp Dermatol. 2009;34(7):761-9.
8. Elsner P and Agner T. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:13-21.
Given the presence of erythema, lichenification, fissuring, and scale of the hands over the course of more than 3 months with the absence of nail findings is most consistent with a diagnosis of chronic hand eczema.
Chronic hand eczema (CHE) is an inflammatory dermatitis of the hands or wrists that persists for longer than 3 months or recurs twice or more in a 12-month timespan.1,2 Hand eczema can be a manifestation of atopic dermatitis, allergic contact dermatitis, or irritant contact dermatitis. Its multifactorial pathogenesis includes epidermal injury and disturbed epidermal barrier function from exogenous factors such as irritants or contact allergens, as well as endogenous factors including atopic dermatitis.3 In pediatrics, it often presents after an acute phase of hand dermatitis with chronic pruritus, erythema, and dry skin with scale.4 Examination findings vary widely with erythema, vesicles, scale, fissures, crusting, hyperkeratosis, and/or lichenification.3,5 Diagnosis is often achieved with careful history, asking about potential exposures that may induce lesions, and physical exam of the entire skin, including the feet. Based upon clinical history or persistent dermatitis, allergic contact dermatitis patch testing should be considered.2
What’s the treatment plan?
Given that CHE is an inflammatory disease process, the goal of treatment is to reduce inflammation and allow for skin barrier repair. Unfortunately, only one study has investigated therapeutics for pediatric CHE,6 with the remainder of the literature based on adult CHE. Current CHE guidelines recommend avoidance of allergens, irritants, or other triggers of the disease as well as liberal and regular use of emollients. Because of the relative thickness of hand skin, higher-potency topical corticosteroids are often used as first-line therapy, with lower-strength topical steroids, calcineurin inhibitors, or crisaborole used as maintenance therapy. Other treatment options include phototherapy, and rarely, systemic therapies are utilized for atopic dermatitis.
What’s the differential diagnosis?
The differential diagnosis of CHE includes other scaling or hyperkeratotic skin conditions including psoriasis and tinea manuum. Other skin conditions that localize to extremities including scabies and hand-foot-and-mouth disease are discussed below.
Psoriasis can present on the hands with erythematous, well-demarcated, silver scaling plaques. However, additional plaques may be found on the elbows, knees, scalp, umbilicus, and sacrum. Nails can demonstrate pitting, oil drops, splinter hemorrhages, or onycholysis. First-line treatment includes a combination of topical steroids, topical vitamin D analogues, and keratolytics.
Tinea mannum is a dermatophyte infection of the skin of the hands. Typically, only one hand is affected with concomitant bilateral tinea pedis. It results in a white scaly plaque with dorsal hand involvement demonstrating an annular appearance, elevated edge, and central clearing. KOH prep will demonstrate septate hyphae, and cultures will grow dermatophyte colonies. Treatment includes topical antifungals or systemic antifungals for recalcitrant disease.
Scabies presents with short linear hypopigmented lesions with a black dot on one end as well as erythematous pruritic papules. These appear on the interdigital web spaces, wrists, axilla, buttocks, and genital region. Skin scraping prep with mineral oil can show mites and eggs. All individuals in an affected household should be treated with either topical permethrin or oral ivermectin to avoid reinfection or parasitic spread. All contacted linens must be cleaned with hot water and dried on high heat.
Hand-foot-and-mouth disease, classically caused by coxsackievirus, is an acute viral illness that results in an eruption of erythematous macules, papules, and vesicles on the ventral hands, soles of the feet, and oral mucosa. Diagnosis is achieved clinically and treatment is symptomatic as the lesions are self-limited.
Our patient underwent patch testing but did not return positive to any allergens. She was started on potent topical corticosteroids, educated on trigger avoidance, and gradually achieved good disease control.
Neither Mr. Haft nor Dr. Eichenfield have any relevant financial disclosures.
Michael Haft is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. He is a 4th year medical student at the University of Rochester (N.Y.). Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital.
References
1. Diepgen TL et al. Br J Dermatol. 2009;160(2):353-8.
2. Diepgen TL et al. J Dtsch Dermatol Ges. 2015;13(1):e1-22.
3. Agner T and Elsner P. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:4-12.
4. Mortz CG et al. Br J Dermatol. 2001;144(3):523-32.
5. Silvestre Salvador JF et al. Actas Dermosifiliogr. 2020;111(1):26-40.
6. Luchsinger I et al. J Eur Acad Dermatol Venereol. 2020;34(5):1037-42.
7. English J et al. Clin Exp Dermatol. 2009;34(7):761-9.
8. Elsner P and Agner T. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:13-21.
Given the presence of erythema, lichenification, fissuring, and scale of the hands over the course of more than 3 months with the absence of nail findings is most consistent with a diagnosis of chronic hand eczema.
Chronic hand eczema (CHE) is an inflammatory dermatitis of the hands or wrists that persists for longer than 3 months or recurs twice or more in a 12-month timespan.1,2 Hand eczema can be a manifestation of atopic dermatitis, allergic contact dermatitis, or irritant contact dermatitis. Its multifactorial pathogenesis includes epidermal injury and disturbed epidermal barrier function from exogenous factors such as irritants or contact allergens, as well as endogenous factors including atopic dermatitis.3 In pediatrics, it often presents after an acute phase of hand dermatitis with chronic pruritus, erythema, and dry skin with scale.4 Examination findings vary widely with erythema, vesicles, scale, fissures, crusting, hyperkeratosis, and/or lichenification.3,5 Diagnosis is often achieved with careful history, asking about potential exposures that may induce lesions, and physical exam of the entire skin, including the feet. Based upon clinical history or persistent dermatitis, allergic contact dermatitis patch testing should be considered.2
What’s the treatment plan?
Given that CHE is an inflammatory disease process, the goal of treatment is to reduce inflammation and allow for skin barrier repair. Unfortunately, only one study has investigated therapeutics for pediatric CHE,6 with the remainder of the literature based on adult CHE. Current CHE guidelines recommend avoidance of allergens, irritants, or other triggers of the disease as well as liberal and regular use of emollients. Because of the relative thickness of hand skin, higher-potency topical corticosteroids are often used as first-line therapy, with lower-strength topical steroids, calcineurin inhibitors, or crisaborole used as maintenance therapy. Other treatment options include phototherapy, and rarely, systemic therapies are utilized for atopic dermatitis.
What’s the differential diagnosis?
The differential diagnosis of CHE includes other scaling or hyperkeratotic skin conditions including psoriasis and tinea manuum. Other skin conditions that localize to extremities including scabies and hand-foot-and-mouth disease are discussed below.
Psoriasis can present on the hands with erythematous, well-demarcated, silver scaling plaques. However, additional plaques may be found on the elbows, knees, scalp, umbilicus, and sacrum. Nails can demonstrate pitting, oil drops, splinter hemorrhages, or onycholysis. First-line treatment includes a combination of topical steroids, topical vitamin D analogues, and keratolytics.
Tinea mannum is a dermatophyte infection of the skin of the hands. Typically, only one hand is affected with concomitant bilateral tinea pedis. It results in a white scaly plaque with dorsal hand involvement demonstrating an annular appearance, elevated edge, and central clearing. KOH prep will demonstrate septate hyphae, and cultures will grow dermatophyte colonies. Treatment includes topical antifungals or systemic antifungals for recalcitrant disease.
Scabies presents with short linear hypopigmented lesions with a black dot on one end as well as erythematous pruritic papules. These appear on the interdigital web spaces, wrists, axilla, buttocks, and genital region. Skin scraping prep with mineral oil can show mites and eggs. All individuals in an affected household should be treated with either topical permethrin or oral ivermectin to avoid reinfection or parasitic spread. All contacted linens must be cleaned with hot water and dried on high heat.
Hand-foot-and-mouth disease, classically caused by coxsackievirus, is an acute viral illness that results in an eruption of erythematous macules, papules, and vesicles on the ventral hands, soles of the feet, and oral mucosa. Diagnosis is achieved clinically and treatment is symptomatic as the lesions are self-limited.
Our patient underwent patch testing but did not return positive to any allergens. She was started on potent topical corticosteroids, educated on trigger avoidance, and gradually achieved good disease control.
Neither Mr. Haft nor Dr. Eichenfield have any relevant financial disclosures.
Michael Haft is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. He is a 4th year medical student at the University of Rochester (N.Y.). Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital.
References
1. Diepgen TL et al. Br J Dermatol. 2009;160(2):353-8.
2. Diepgen TL et al. J Dtsch Dermatol Ges. 2015;13(1):e1-22.
3. Agner T and Elsner P. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:4-12.
4. Mortz CG et al. Br J Dermatol. 2001;144(3):523-32.
5. Silvestre Salvador JF et al. Actas Dermosifiliogr. 2020;111(1):26-40.
6. Luchsinger I et al. J Eur Acad Dermatol Venereol. 2020;34(5):1037-42.
7. English J et al. Clin Exp Dermatol. 2009;34(7):761-9.
8. Elsner P and Agner T. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:13-21.
Examination findings of the bilateral hands and wrists demonstrate plaques of erythema, lichenification, and scale of the dorsal surfaces of the hands and digits. Closer inspection reveals fissuring and erythematous crust of the affected skin but normal nails. The rest of the skin exam is unremarkable.
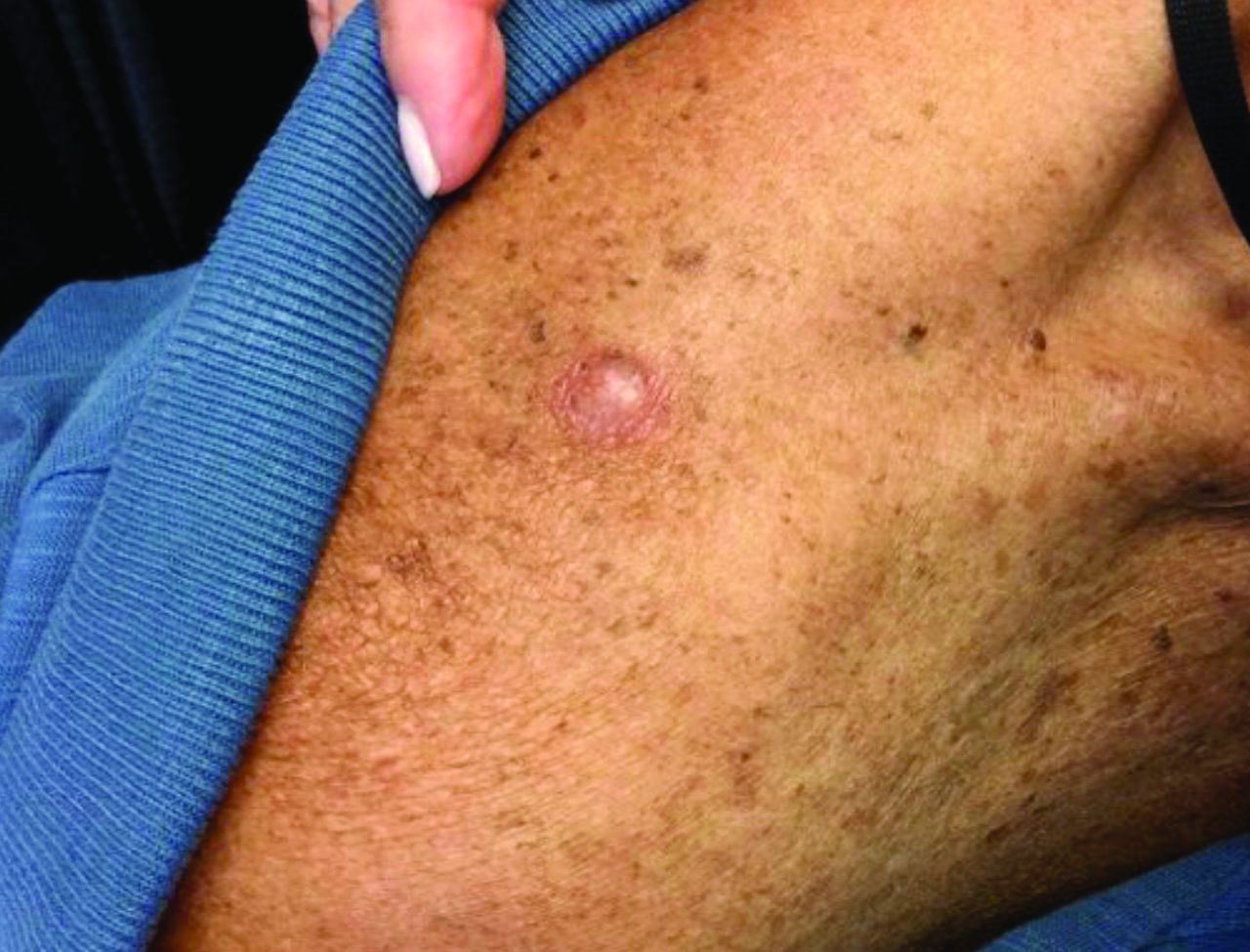
Pink papule on thigh

A deep-shave biopsy indicated that this was an inflamed/irritated solitary neurofibroma. Basal cell carcinoma, inflamed nevus, and Merkel cell carcinoma were also considered.
Most often manifesting in adults, solitary neurofibromas are common nonencapsulated, soft to firm papules that range in size from 2 mm to 2 cm. Solitary neurofibromas are benign and work-up for systemic neurofibromatosis is not indicated. However, if a patient presents with multiple neurofibromas, axillary freckling, or multiple café au lait macules, systemic disease should be considered, followed by molecular testing and/or referral to a medical geneticist or neurofibromatosis clinic.
Although both the triage amalgamated diagnostic algorithm and the 2-step dermoscopy algorithm suggested this lesion was higher risk, it was ultimately found to be benign. This case highlights areas in which dermoscopy and physical exam lack specificity, but this trade-off increases the sensitivity of an algorithmic approach. Solitary pink papules can include some subtle, but fearsome, diagnoses and deserve close attention. In this case, the biopsy not only helped confirm the diagnosis, but it also alleviated the discomfort caused by the neurofibroma.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Geller S, Pulitzer M, Brady MS, et al. Dermoscopic assessment of vascular structures in solitary small pink lesions—differentiating between good and evil. Dermatol Pract Concept. 2017;7:47-50. doi: 10.5826/dpc.0703a10

A deep-shave biopsy indicated that this was an inflamed/irritated solitary neurofibroma. Basal cell carcinoma, inflamed nevus, and Merkel cell carcinoma were also considered.
Most often manifesting in adults, solitary neurofibromas are common nonencapsulated, soft to firm papules that range in size from 2 mm to 2 cm. Solitary neurofibromas are benign and work-up for systemic neurofibromatosis is not indicated. However, if a patient presents with multiple neurofibromas, axillary freckling, or multiple café au lait macules, systemic disease should be considered, followed by molecular testing and/or referral to a medical geneticist or neurofibromatosis clinic.
Although both the triage amalgamated diagnostic algorithm and the 2-step dermoscopy algorithm suggested this lesion was higher risk, it was ultimately found to be benign. This case highlights areas in which dermoscopy and physical exam lack specificity, but this trade-off increases the sensitivity of an algorithmic approach. Solitary pink papules can include some subtle, but fearsome, diagnoses and deserve close attention. In this case, the biopsy not only helped confirm the diagnosis, but it also alleviated the discomfort caused by the neurofibroma.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

A deep-shave biopsy indicated that this was an inflamed/irritated solitary neurofibroma. Basal cell carcinoma, inflamed nevus, and Merkel cell carcinoma were also considered.
Most often manifesting in adults, solitary neurofibromas are common nonencapsulated, soft to firm papules that range in size from 2 mm to 2 cm. Solitary neurofibromas are benign and work-up for systemic neurofibromatosis is not indicated. However, if a patient presents with multiple neurofibromas, axillary freckling, or multiple café au lait macules, systemic disease should be considered, followed by molecular testing and/or referral to a medical geneticist or neurofibromatosis clinic.
Although both the triage amalgamated diagnostic algorithm and the 2-step dermoscopy algorithm suggested this lesion was higher risk, it was ultimately found to be benign. This case highlights areas in which dermoscopy and physical exam lack specificity, but this trade-off increases the sensitivity of an algorithmic approach. Solitary pink papules can include some subtle, but fearsome, diagnoses and deserve close attention. In this case, the biopsy not only helped confirm the diagnosis, but it also alleviated the discomfort caused by the neurofibroma.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Geller S, Pulitzer M, Brady MS, et al. Dermoscopic assessment of vascular structures in solitary small pink lesions—differentiating between good and evil. Dermatol Pract Concept. 2017;7:47-50. doi: 10.5826/dpc.0703a10
1. Geller S, Pulitzer M, Brady MS, et al. Dermoscopic assessment of vascular structures in solitary small pink lesions—differentiating between good and evil. Dermatol Pract Concept. 2017;7:47-50. doi: 10.5826/dpc.0703a10
An 80-year-old patient presents with an asymptomatic firm pink plaque on his shoulder
Melanoma is a type of skin cancer that arises from melanocytes. According to the American Cancer Society, about 106,110 new melanomas will be diagnosed in the United States in 2021.The risk for developing melanoma increases with age. There are multiple clinical forms of cutaneous melanoma. The four main types are superficial spreading melanoma, nodular melanoma, melanoma in situ (lentigo maligna), and acral lentiginous melanoma. Rare variants include amelanotic melanoma, nevoid melanoma, spitzoid melanoma, and desmoplastic melanoma (DM). Melanoma can also rarely affect parts of the eye and mucosa.
, according to the Memorial Sloan Kettering Cancer Center. It typically presents as a subtle pigmented, pink, red, or skin colored patch, papule or plaque on sun-exposed skin (head and neck most frequently). Chronic UV exposure has been linked to DM. It may be mistaken for a scar or dermatofibroma. DM tends to grow locally and has less risk for nodal metastasis.1
Histologic diagnosis may be challenging. Two histologic variants in desmoplastic melanoma have been described: pure and mixed, depending on the degree of desmoplasia and cellularity present in the tumor.1 Pure DM tends to have a less aggressive course. Melanocytes can appear spindled in a fibrotic stroma. Patchy lymphocyte aggregates may be seen. Perineural invasion is more common in desmoplastic melanoma. Histologically, the differential includes spindle cell carcinoma and sarcoma. Immunostaining is helpful in differentiation.
Our patient had no lymphadenopathy on physical examination. Biopsy revealed a desmoplastic melanoma, 3.6 mm in depth, no ulceration, no regression, mitotic rate 1/mm2. He was referred to surgical oncology. The patient underwent wide excision. Sentinel lymph node biopsy was deferred.
It is imperative for dermatologists to be cognizant of this challenging subtype of melanoma when evaluating patients.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Chen L et al. J Am Acad Dermatol. 2013 May;68(5):825-33.
Melanoma is a type of skin cancer that arises from melanocytes. According to the American Cancer Society, about 106,110 new melanomas will be diagnosed in the United States in 2021.The risk for developing melanoma increases with age. There are multiple clinical forms of cutaneous melanoma. The four main types are superficial spreading melanoma, nodular melanoma, melanoma in situ (lentigo maligna), and acral lentiginous melanoma. Rare variants include amelanotic melanoma, nevoid melanoma, spitzoid melanoma, and desmoplastic melanoma (DM). Melanoma can also rarely affect parts of the eye and mucosa.
, according to the Memorial Sloan Kettering Cancer Center. It typically presents as a subtle pigmented, pink, red, or skin colored patch, papule or plaque on sun-exposed skin (head and neck most frequently). Chronic UV exposure has been linked to DM. It may be mistaken for a scar or dermatofibroma. DM tends to grow locally and has less risk for nodal metastasis.1
Histologic diagnosis may be challenging. Two histologic variants in desmoplastic melanoma have been described: pure and mixed, depending on the degree of desmoplasia and cellularity present in the tumor.1 Pure DM tends to have a less aggressive course. Melanocytes can appear spindled in a fibrotic stroma. Patchy lymphocyte aggregates may be seen. Perineural invasion is more common in desmoplastic melanoma. Histologically, the differential includes spindle cell carcinoma and sarcoma. Immunostaining is helpful in differentiation.
Our patient had no lymphadenopathy on physical examination. Biopsy revealed a desmoplastic melanoma, 3.6 mm in depth, no ulceration, no regression, mitotic rate 1/mm2. He was referred to surgical oncology. The patient underwent wide excision. Sentinel lymph node biopsy was deferred.
It is imperative for dermatologists to be cognizant of this challenging subtype of melanoma when evaluating patients.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Chen L et al. J Am Acad Dermatol. 2013 May;68(5):825-33.
Melanoma is a type of skin cancer that arises from melanocytes. According to the American Cancer Society, about 106,110 new melanomas will be diagnosed in the United States in 2021.The risk for developing melanoma increases with age. There are multiple clinical forms of cutaneous melanoma. The four main types are superficial spreading melanoma, nodular melanoma, melanoma in situ (lentigo maligna), and acral lentiginous melanoma. Rare variants include amelanotic melanoma, nevoid melanoma, spitzoid melanoma, and desmoplastic melanoma (DM). Melanoma can also rarely affect parts of the eye and mucosa.
, according to the Memorial Sloan Kettering Cancer Center. It typically presents as a subtle pigmented, pink, red, or skin colored patch, papule or plaque on sun-exposed skin (head and neck most frequently). Chronic UV exposure has been linked to DM. It may be mistaken for a scar or dermatofibroma. DM tends to grow locally and has less risk for nodal metastasis.1
Histologic diagnosis may be challenging. Two histologic variants in desmoplastic melanoma have been described: pure and mixed, depending on the degree of desmoplasia and cellularity present in the tumor.1 Pure DM tends to have a less aggressive course. Melanocytes can appear spindled in a fibrotic stroma. Patchy lymphocyte aggregates may be seen. Perineural invasion is more common in desmoplastic melanoma. Histologically, the differential includes spindle cell carcinoma and sarcoma. Immunostaining is helpful in differentiation.
Our patient had no lymphadenopathy on physical examination. Biopsy revealed a desmoplastic melanoma, 3.6 mm in depth, no ulceration, no regression, mitotic rate 1/mm2. He was referred to surgical oncology. The patient underwent wide excision. Sentinel lymph node biopsy was deferred.
It is imperative for dermatologists to be cognizant of this challenging subtype of melanoma when evaluating patients.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Chen L et al. J Am Acad Dermatol. 2013 May;68(5):825-33.
AAP issues five recommendations for common dermatologic problems
The American Academy of Pediatrics recently issued five recommendations for the most common dermatologic problems in primary care pediatrics.
Topics include diagnostic and management strategies for a variety of conditions, including atopic dermatitis, fungal infections, and autoimmune conditions.
The AAP Section on Dermatology created the recommendations, which were then reviewed and approved by “more than a dozen relevant AAP committees, councils, and sections,” before final approval by the AAP executive committee and board of directors.
The final list represents a collaborative effort with the Choosing Wisely initiative of the American Board of Internal Medicine Foundation, which aims “to promote conversations between clinicians and patients by helping patients choose care that is supported by evidence, not duplicative of other tests or procedures already received, free from harm, [and] truly necessary.”
Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said that the recommendations are “a fine set of suggestions to help health care providers with some of their pediatric dermatology issues.”
• To begin, the AAP recommended against use of combination topical steroid antifungals for candida skin infections, diaper dermatitis, and tinea corporis, despite approvals for these indications.
“Many providers are unaware that the combination products contain a relatively high-potency topical steroid,” the AAP wrote, noting that “combination products are also often expensive and not covered by pharmacy plans.”
Diaper dermatitis responds best to barrier creams and ointments alone, according to the AAP. If needed, a topical, low-potency steroid may be used no more than twice a day, and tapered with improvement. Similarly, the AAP recommended a separate, low-potency steroid for tinea corporis if pruritus is severe.
• In contrast with this call for minimal treatment intensity, the AAP recommended a more intensive approach to tinea capitis, advising against topical medications alone.
“Topical treatments cannot penetrate the hair shaft itself, which is where the infection lies; thus, monotherapy with topical medications is insufficient to effectively treat the infection,” the AAP wrote. “This insufficient treatment can lead to increased health care costs resulting from multiple visits and the prescribing of ineffective medications.”
While medicated shampoos may still be used as adjunctive treatments for tinea capitis, the AAP recommended primary therapy with either griseofulvin or terbinafine, slightly favoring terbinafine because of adequate efficacy, lesser expense, and shorter regimen.
According to Dr. Eichenfield, a more thorough workup should also be considered.
“Consider culturing possible tinea capitis, so that oral antifungals can be used judiciously and not used for other scaling scalp diagnoses,” he said.
• For most cases of atopic dermatitis, the AAP advised against oral or injected corticosteroids, despite rapid efficacy, because of potential for adverse events, such as adrenal suppression, growth retardation, and disease worsening upon discontinuation. Instead, they recommended topical therapies, “good skin care practices,” and if necessary, “phototherapy and/or steroid-sparing systemic agents.”
“Systemic corticosteroids should only be prescribed for severe flares once all other treatment options have been exhausted and should be limited to a short course for the purpose of bridging to a steroid-sparing agent,” the AAP wrote.
Dr. Eichenfield emphasized this point, noting that new therapies have expanded treatment options.
“Be aware of the advances in atopic dermatitis,” he said, “with newer topical medications and with a new systemic biologic agent approved for moderate to severe refractory atopic dermatitis for ages 6 and older.”
• Turning to diagnostic strategies, the AAP recommended against routine laboratory testing for associated autoimmune diseases among patients with vitiligo, unless clinical signs and/or symptoms of such diseases are present.
“There is no convincing evidence that extensive workups in the absence of specific clinical suspicion improves outcomes for patients and may in fact beget additional costs and harms,” the AAP wrote. “Although many studies suggest ordering these tests, it is based largely on the increased cosegregation of vitiligo and thyroid disease and not on improved outcomes from having identified an abnormal laboratory test result.”
• Similarly, the AAP advised practitioners to avoid routinely testing patients with alopecia areata for other diseases if relevant symptoms and signs aren’t present.
“As in the case of vitiligo, it is more common to find thyroid autoantibodies or subclinical hypothyroidism than overt thyroid disease, unless there are clinically suspicious findings,” the AAP wrote. “Patients identified as having subclinical hypothyroidism are not currently treated and may even have resolution of the abnormal TSH.”
Before drawing blood, Dr. Eichenfield suggested that clinicians first ask the right questions.
“Be comfortable with screening questions about growth, weight, or activity changes to assist with decisions for thyroid screening in a patient with vitiligo or alopecia areata,” he said.
Choosing Wisely is an initiative of the American Board of Internal Medicine. The AAP and Dr. Eichenfield reported no conflicts of interest.
The American Academy of Pediatrics recently issued five recommendations for the most common dermatologic problems in primary care pediatrics.
Topics include diagnostic and management strategies for a variety of conditions, including atopic dermatitis, fungal infections, and autoimmune conditions.
The AAP Section on Dermatology created the recommendations, which were then reviewed and approved by “more than a dozen relevant AAP committees, councils, and sections,” before final approval by the AAP executive committee and board of directors.
The final list represents a collaborative effort with the Choosing Wisely initiative of the American Board of Internal Medicine Foundation, which aims “to promote conversations between clinicians and patients by helping patients choose care that is supported by evidence, not duplicative of other tests or procedures already received, free from harm, [and] truly necessary.”
Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said that the recommendations are “a fine set of suggestions to help health care providers with some of their pediatric dermatology issues.”
• To begin, the AAP recommended against use of combination topical steroid antifungals for candida skin infections, diaper dermatitis, and tinea corporis, despite approvals for these indications.
“Many providers are unaware that the combination products contain a relatively high-potency topical steroid,” the AAP wrote, noting that “combination products are also often expensive and not covered by pharmacy plans.”
Diaper dermatitis responds best to barrier creams and ointments alone, according to the AAP. If needed, a topical, low-potency steroid may be used no more than twice a day, and tapered with improvement. Similarly, the AAP recommended a separate, low-potency steroid for tinea corporis if pruritus is severe.
• In contrast with this call for minimal treatment intensity, the AAP recommended a more intensive approach to tinea capitis, advising against topical medications alone.
“Topical treatments cannot penetrate the hair shaft itself, which is where the infection lies; thus, monotherapy with topical medications is insufficient to effectively treat the infection,” the AAP wrote. “This insufficient treatment can lead to increased health care costs resulting from multiple visits and the prescribing of ineffective medications.”
While medicated shampoos may still be used as adjunctive treatments for tinea capitis, the AAP recommended primary therapy with either griseofulvin or terbinafine, slightly favoring terbinafine because of adequate efficacy, lesser expense, and shorter regimen.
According to Dr. Eichenfield, a more thorough workup should also be considered.
“Consider culturing possible tinea capitis, so that oral antifungals can be used judiciously and not used for other scaling scalp diagnoses,” he said.
• For most cases of atopic dermatitis, the AAP advised against oral or injected corticosteroids, despite rapid efficacy, because of potential for adverse events, such as adrenal suppression, growth retardation, and disease worsening upon discontinuation. Instead, they recommended topical therapies, “good skin care practices,” and if necessary, “phototherapy and/or steroid-sparing systemic agents.”
“Systemic corticosteroids should only be prescribed for severe flares once all other treatment options have been exhausted and should be limited to a short course for the purpose of bridging to a steroid-sparing agent,” the AAP wrote.
Dr. Eichenfield emphasized this point, noting that new therapies have expanded treatment options.
“Be aware of the advances in atopic dermatitis,” he said, “with newer topical medications and with a new systemic biologic agent approved for moderate to severe refractory atopic dermatitis for ages 6 and older.”
• Turning to diagnostic strategies, the AAP recommended against routine laboratory testing for associated autoimmune diseases among patients with vitiligo, unless clinical signs and/or symptoms of such diseases are present.
“There is no convincing evidence that extensive workups in the absence of specific clinical suspicion improves outcomes for patients and may in fact beget additional costs and harms,” the AAP wrote. “Although many studies suggest ordering these tests, it is based largely on the increased cosegregation of vitiligo and thyroid disease and not on improved outcomes from having identified an abnormal laboratory test result.”
• Similarly, the AAP advised practitioners to avoid routinely testing patients with alopecia areata for other diseases if relevant symptoms and signs aren’t present.
“As in the case of vitiligo, it is more common to find thyroid autoantibodies or subclinical hypothyroidism than overt thyroid disease, unless there are clinically suspicious findings,” the AAP wrote. “Patients identified as having subclinical hypothyroidism are not currently treated and may even have resolution of the abnormal TSH.”
Before drawing blood, Dr. Eichenfield suggested that clinicians first ask the right questions.
“Be comfortable with screening questions about growth, weight, or activity changes to assist with decisions for thyroid screening in a patient with vitiligo or alopecia areata,” he said.
Choosing Wisely is an initiative of the American Board of Internal Medicine. The AAP and Dr. Eichenfield reported no conflicts of interest.
The American Academy of Pediatrics recently issued five recommendations for the most common dermatologic problems in primary care pediatrics.
Topics include diagnostic and management strategies for a variety of conditions, including atopic dermatitis, fungal infections, and autoimmune conditions.
The AAP Section on Dermatology created the recommendations, which were then reviewed and approved by “more than a dozen relevant AAP committees, councils, and sections,” before final approval by the AAP executive committee and board of directors.
The final list represents a collaborative effort with the Choosing Wisely initiative of the American Board of Internal Medicine Foundation, which aims “to promote conversations between clinicians and patients by helping patients choose care that is supported by evidence, not duplicative of other tests or procedures already received, free from harm, [and] truly necessary.”
Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said that the recommendations are “a fine set of suggestions to help health care providers with some of their pediatric dermatology issues.”
• To begin, the AAP recommended against use of combination topical steroid antifungals for candida skin infections, diaper dermatitis, and tinea corporis, despite approvals for these indications.
“Many providers are unaware that the combination products contain a relatively high-potency topical steroid,” the AAP wrote, noting that “combination products are also often expensive and not covered by pharmacy plans.”
Diaper dermatitis responds best to barrier creams and ointments alone, according to the AAP. If needed, a topical, low-potency steroid may be used no more than twice a day, and tapered with improvement. Similarly, the AAP recommended a separate, low-potency steroid for tinea corporis if pruritus is severe.
• In contrast with this call for minimal treatment intensity, the AAP recommended a more intensive approach to tinea capitis, advising against topical medications alone.
“Topical treatments cannot penetrate the hair shaft itself, which is where the infection lies; thus, monotherapy with topical medications is insufficient to effectively treat the infection,” the AAP wrote. “This insufficient treatment can lead to increased health care costs resulting from multiple visits and the prescribing of ineffective medications.”
While medicated shampoos may still be used as adjunctive treatments for tinea capitis, the AAP recommended primary therapy with either griseofulvin or terbinafine, slightly favoring terbinafine because of adequate efficacy, lesser expense, and shorter regimen.
According to Dr. Eichenfield, a more thorough workup should also be considered.
“Consider culturing possible tinea capitis, so that oral antifungals can be used judiciously and not used for other scaling scalp diagnoses,” he said.
• For most cases of atopic dermatitis, the AAP advised against oral or injected corticosteroids, despite rapid efficacy, because of potential for adverse events, such as adrenal suppression, growth retardation, and disease worsening upon discontinuation. Instead, they recommended topical therapies, “good skin care practices,” and if necessary, “phototherapy and/or steroid-sparing systemic agents.”
“Systemic corticosteroids should only be prescribed for severe flares once all other treatment options have been exhausted and should be limited to a short course for the purpose of bridging to a steroid-sparing agent,” the AAP wrote.
Dr. Eichenfield emphasized this point, noting that new therapies have expanded treatment options.
“Be aware of the advances in atopic dermatitis,” he said, “with newer topical medications and with a new systemic biologic agent approved for moderate to severe refractory atopic dermatitis for ages 6 and older.”
• Turning to diagnostic strategies, the AAP recommended against routine laboratory testing for associated autoimmune diseases among patients with vitiligo, unless clinical signs and/or symptoms of such diseases are present.
“There is no convincing evidence that extensive workups in the absence of specific clinical suspicion improves outcomes for patients and may in fact beget additional costs and harms,” the AAP wrote. “Although many studies suggest ordering these tests, it is based largely on the increased cosegregation of vitiligo and thyroid disease and not on improved outcomes from having identified an abnormal laboratory test result.”
• Similarly, the AAP advised practitioners to avoid routinely testing patients with alopecia areata for other diseases if relevant symptoms and signs aren’t present.
“As in the case of vitiligo, it is more common to find thyroid autoantibodies or subclinical hypothyroidism than overt thyroid disease, unless there are clinically suspicious findings,” the AAP wrote. “Patients identified as having subclinical hypothyroidism are not currently treated and may even have resolution of the abnormal TSH.”
Before drawing blood, Dr. Eichenfield suggested that clinicians first ask the right questions.
“Be comfortable with screening questions about growth, weight, or activity changes to assist with decisions for thyroid screening in a patient with vitiligo or alopecia areata,” he said.
Choosing Wisely is an initiative of the American Board of Internal Medicine. The AAP and Dr. Eichenfield reported no conflicts of interest.
FROM CHOOSING WISELY AND THE AAP
This Rash Really Stinks!
ANSWER
The correct diagnosis is Darier disease (choice “d”).
DISCUSSION
Darier disease, also known as Darier-White disease or keratosis follicularis, is an inherited defect transmitted by autosomal dominant mode. The pathophysiologic process is a breakdown of cell adhesion that normally binds keratin filaments to tiny connecting fibers called desmosomes.
Darier disease manifests with a “branny” papulosquamous rash, typically arising in the third decade of life and affecting the chest, scalp, back, and intertriginous areas. The nail and intraoral findings noted in this patient are typical. In the author’s experience, the former is more commonly seen and is essentially pathognomic for the disease.
Darier disease is relatively rare, occurring in 1:30,000 to 1:100,000 population, depending on the geographic area studied. Men and women are equally affected, although it is more common in those with darker skin.
The differential outlined in the answer choices is reasonable, considering the condition’s rarity and how unlikely it is to manifest solely in the inframammary area. One could conclude that, just as with psoriasis (choice “b”) and seborrhea, intertrigo (choice “c”) is not always a primary process. And although yeast infection (choice “a”) can complicate any florid rash in this area, topical and oral anti-yeast treatment had utterly failed to help.
TREATMENT
Isotretinoin is used in cases such as this one, but it only offers temporary relief. For less severe cases, oral antibiotics (eg minocycline) or topical steroids (used with caution given the risk for atrophy in the inframammary area) often suffice. This patient’s prognosis is guarded at best, although control of the worst is certainly possible.
ANSWER
The correct diagnosis is Darier disease (choice “d”).
DISCUSSION
Darier disease, also known as Darier-White disease or keratosis follicularis, is an inherited defect transmitted by autosomal dominant mode. The pathophysiologic process is a breakdown of cell adhesion that normally binds keratin filaments to tiny connecting fibers called desmosomes.
Darier disease manifests with a “branny” papulosquamous rash, typically arising in the third decade of life and affecting the chest, scalp, back, and intertriginous areas. The nail and intraoral findings noted in this patient are typical. In the author’s experience, the former is more commonly seen and is essentially pathognomic for the disease.
Darier disease is relatively rare, occurring in 1:30,000 to 1:100,000 population, depending on the geographic area studied. Men and women are equally affected, although it is more common in those with darker skin.
The differential outlined in the answer choices is reasonable, considering the condition’s rarity and how unlikely it is to manifest solely in the inframammary area. One could conclude that, just as with psoriasis (choice “b”) and seborrhea, intertrigo (choice “c”) is not always a primary process. And although yeast infection (choice “a”) can complicate any florid rash in this area, topical and oral anti-yeast treatment had utterly failed to help.
TREATMENT
Isotretinoin is used in cases such as this one, but it only offers temporary relief. For less severe cases, oral antibiotics (eg minocycline) or topical steroids (used with caution given the risk for atrophy in the inframammary area) often suffice. This patient’s prognosis is guarded at best, although control of the worst is certainly possible.
ANSWER
The correct diagnosis is Darier disease (choice “d”).
DISCUSSION
Darier disease, also known as Darier-White disease or keratosis follicularis, is an inherited defect transmitted by autosomal dominant mode. The pathophysiologic process is a breakdown of cell adhesion that normally binds keratin filaments to tiny connecting fibers called desmosomes.
Darier disease manifests with a “branny” papulosquamous rash, typically arising in the third decade of life and affecting the chest, scalp, back, and intertriginous areas. The nail and intraoral findings noted in this patient are typical. In the author’s experience, the former is more commonly seen and is essentially pathognomic for the disease.
Darier disease is relatively rare, occurring in 1:30,000 to 1:100,000 population, depending on the geographic area studied. Men and women are equally affected, although it is more common in those with darker skin.
The differential outlined in the answer choices is reasonable, considering the condition’s rarity and how unlikely it is to manifest solely in the inframammary area. One could conclude that, just as with psoriasis (choice “b”) and seborrhea, intertrigo (choice “c”) is not always a primary process. And although yeast infection (choice “a”) can complicate any florid rash in this area, topical and oral anti-yeast treatment had utterly failed to help.
TREATMENT
Isotretinoin is used in cases such as this one, but it only offers temporary relief. For less severe cases, oral antibiotics (eg minocycline) or topical steroids (used with caution given the risk for atrophy in the inframammary area) often suffice. This patient’s prognosis is guarded at best, although control of the worst is certainly possible.

A 50-year-old woman is referred to dermatology with a “yeast” infection of several years’ duration. The condition causes considerable discomfort, especially during hot weather when the rash emits a very objectionable odor.
The florid, white, scaly rash under her breasts is a stark contrast to the patient’s type V skin. On both sides, the affected skin perfectly matches the inframammary fold. There are sharp margins and uniform moist scaling.
Looking elsewhere, 7 of 10 fingernails exhibit longitudinal white and red streaks, along with triangular nicks in the edges of several nails. The roof of the patient’s mouth is studded with fleshy nodules measuring 0.6 to 1.0 cm. Several pits are seen on her palms.
The patient is in no distress but is quite agitated by the lack of effective treatment. She reports trying a number of prescription and OTC anti-yeast creams, lotions, and oral medications, none of which resolved the problem.
History-taking reveals a family history of skin problems, although neither the patient nor anyone else in the family has ever been seen by a dermatologist. No one has ever suggested that a biopsy be done.
A punch biopsy is performed on the affected inframammary skin. The pathology report shows acantholysis with focal dyskeratotic keratinocytes. Intraepidermal separation is seen throughout the specimen.
Vasodilatory medications found protective against rosacea
.
“Our initial hypothesis was that perhaps antihypertensive agents might be associated with worsening rosacea,” one of the study authors, Jennifer G. Powers, MD, associate professor of dermatology at the University of Iowa, Iowa City, said in an interview. “What we found was exactly the opposite – that in fact their presence in a medical chart correlated with lower rates of rosacea diagnoses, as defined by ICD 9/10 codes.”
According to the researchers, who published their findings in the Journal of the American Academy of Dermatology, cases of acute vasodilator-induced rosacea have been reported, but no long-term association has been established. “In fact, many widely used antihypertensive medications modulate peripheral vascular tone,” they wrote. “Therefore, chronic use in patients with hypertension may reduce damage to peripheral vessels, and thus decrease risk of rosacea.”
To determine the correlates between vasodilator use and risk of rosacea, Dr. Powers and colleagues identified 680 hypertensive patients being treated with vasodilators or a thiazide diuretic in whom rosacea developed within 5 years of initiating therapy between June 1, 2006, and April 31, 2019. Vasodilator therapies included angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, and calcium channel blockers (CCBs). Patients on thiazide diuretics served as the control group. The researchers stratified the patients by age, gender, race, diabetes, chronic kidney disease, and coronary artery disease and calculated relative risk estimates comparing vasodilators with thiazides between strata.
Of the 680 patients, all but 40 were White; 127 were on thiazides, and the remaining 553 were on vasodilators. Overall, the researchers observed that use of vasodilators had a protective effect on the development of rosacea within 5 years, compared with thiazides (relative risk [RR], 0.56; P less than .0001). Specifically, the relative risk was 0.50 for ACE-inhibitors (P less than .0001); 0.69 for ARBs (P = .041); 0.55 for beta-blockers (P less than .0001); and 0.39 for CCBs (P less than .0001).
Dr. Powers and colleagues also observed significant inverse correlations in ACE-inhibitors, beta-blockers, and CCBs among White women aged 50 and older, but no significance was observed in non-White subgroups. The cohorts of patients with chronic kidney disease and coronary artery disease were too small for analysis.
“We were very surprised to find that many of the agents we think of as vasodilators might actually be beneficial for rosacea,” Dr. Powers said. “We would like to see these results reproduced in larger population studies. There are also potential questions about the mechanism at play. However, should these findings hold true, [it’s] all the more reason for our rosacea patients with hypertension to be managed well. They need not fear that those medications are worsening disease. Also, there might be new therapeutic options based on this data.”
The study received funding support from the National Center for Advancing Translational Sciences. The researchers reported having no financial disclosures.
One of Dr. Powers’ coauthors is her husband, Edward M. Powers, MD, a cardiology fellow at the University of Iowa. “We sometimes bounce ideas off one another and will talk about how systemic effects on the vasculature may impact skin disease,” she said, noting that they also published a report on statins and atopic dermatitis.
.
“Our initial hypothesis was that perhaps antihypertensive agents might be associated with worsening rosacea,” one of the study authors, Jennifer G. Powers, MD, associate professor of dermatology at the University of Iowa, Iowa City, said in an interview. “What we found was exactly the opposite – that in fact their presence in a medical chart correlated with lower rates of rosacea diagnoses, as defined by ICD 9/10 codes.”
According to the researchers, who published their findings in the Journal of the American Academy of Dermatology, cases of acute vasodilator-induced rosacea have been reported, but no long-term association has been established. “In fact, many widely used antihypertensive medications modulate peripheral vascular tone,” they wrote. “Therefore, chronic use in patients with hypertension may reduce damage to peripheral vessels, and thus decrease risk of rosacea.”
To determine the correlates between vasodilator use and risk of rosacea, Dr. Powers and colleagues identified 680 hypertensive patients being treated with vasodilators or a thiazide diuretic in whom rosacea developed within 5 years of initiating therapy between June 1, 2006, and April 31, 2019. Vasodilator therapies included angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, and calcium channel blockers (CCBs). Patients on thiazide diuretics served as the control group. The researchers stratified the patients by age, gender, race, diabetes, chronic kidney disease, and coronary artery disease and calculated relative risk estimates comparing vasodilators with thiazides between strata.
Of the 680 patients, all but 40 were White; 127 were on thiazides, and the remaining 553 were on vasodilators. Overall, the researchers observed that use of vasodilators had a protective effect on the development of rosacea within 5 years, compared with thiazides (relative risk [RR], 0.56; P less than .0001). Specifically, the relative risk was 0.50 for ACE-inhibitors (P less than .0001); 0.69 for ARBs (P = .041); 0.55 for beta-blockers (P less than .0001); and 0.39 for CCBs (P less than .0001).
Dr. Powers and colleagues also observed significant inverse correlations in ACE-inhibitors, beta-blockers, and CCBs among White women aged 50 and older, but no significance was observed in non-White subgroups. The cohorts of patients with chronic kidney disease and coronary artery disease were too small for analysis.
“We were very surprised to find that many of the agents we think of as vasodilators might actually be beneficial for rosacea,” Dr. Powers said. “We would like to see these results reproduced in larger population studies. There are also potential questions about the mechanism at play. However, should these findings hold true, [it’s] all the more reason for our rosacea patients with hypertension to be managed well. They need not fear that those medications are worsening disease. Also, there might be new therapeutic options based on this data.”
The study received funding support from the National Center for Advancing Translational Sciences. The researchers reported having no financial disclosures.
One of Dr. Powers’ coauthors is her husband, Edward M. Powers, MD, a cardiology fellow at the University of Iowa. “We sometimes bounce ideas off one another and will talk about how systemic effects on the vasculature may impact skin disease,” she said, noting that they also published a report on statins and atopic dermatitis.
.
“Our initial hypothesis was that perhaps antihypertensive agents might be associated with worsening rosacea,” one of the study authors, Jennifer G. Powers, MD, associate professor of dermatology at the University of Iowa, Iowa City, said in an interview. “What we found was exactly the opposite – that in fact their presence in a medical chart correlated with lower rates of rosacea diagnoses, as defined by ICD 9/10 codes.”
According to the researchers, who published their findings in the Journal of the American Academy of Dermatology, cases of acute vasodilator-induced rosacea have been reported, but no long-term association has been established. “In fact, many widely used antihypertensive medications modulate peripheral vascular tone,” they wrote. “Therefore, chronic use in patients with hypertension may reduce damage to peripheral vessels, and thus decrease risk of rosacea.”
To determine the correlates between vasodilator use and risk of rosacea, Dr. Powers and colleagues identified 680 hypertensive patients being treated with vasodilators or a thiazide diuretic in whom rosacea developed within 5 years of initiating therapy between June 1, 2006, and April 31, 2019. Vasodilator therapies included angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, and calcium channel blockers (CCBs). Patients on thiazide diuretics served as the control group. The researchers stratified the patients by age, gender, race, diabetes, chronic kidney disease, and coronary artery disease and calculated relative risk estimates comparing vasodilators with thiazides between strata.
Of the 680 patients, all but 40 were White; 127 were on thiazides, and the remaining 553 were on vasodilators. Overall, the researchers observed that use of vasodilators had a protective effect on the development of rosacea within 5 years, compared with thiazides (relative risk [RR], 0.56; P less than .0001). Specifically, the relative risk was 0.50 for ACE-inhibitors (P less than .0001); 0.69 for ARBs (P = .041); 0.55 for beta-blockers (P less than .0001); and 0.39 for CCBs (P less than .0001).
Dr. Powers and colleagues also observed significant inverse correlations in ACE-inhibitors, beta-blockers, and CCBs among White women aged 50 and older, but no significance was observed in non-White subgroups. The cohorts of patients with chronic kidney disease and coronary artery disease were too small for analysis.
“We were very surprised to find that many of the agents we think of as vasodilators might actually be beneficial for rosacea,” Dr. Powers said. “We would like to see these results reproduced in larger population studies. There are also potential questions about the mechanism at play. However, should these findings hold true, [it’s] all the more reason for our rosacea patients with hypertension to be managed well. They need not fear that those medications are worsening disease. Also, there might be new therapeutic options based on this data.”
The study received funding support from the National Center for Advancing Translational Sciences. The researchers reported having no financial disclosures.
One of Dr. Powers’ coauthors is her husband, Edward M. Powers, MD, a cardiology fellow at the University of Iowa. “We sometimes bounce ideas off one another and will talk about how systemic effects on the vasculature may impact skin disease,” she said, noting that they also published a report on statins and atopic dermatitis.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Managing hyperhidrosis, HS: Ask questions first
A wide variety of medications exists for treating hyperhidrosis, a dermatologist told colleagues, but before prescribing anything to a pediatric patient, he recommended, ask the patient a simple question: “What bothers you the most?”
The answer will provide guidance for developing a step-by-step treatment strategy and help provide the patient “a set of realistic expectations in terms of what the response will look like,” George Hightower, MD, PhD, a pediatric dermatologist at Rady Children’s Hospital and the University of California, San Diego, said at MedscapeLive’s Women’s & Pediatric Dermatology Seminar.
A similar question-based approach will help guide therapy for patients with hidradenitis suppurativa (HS), he said.
With regards to hyperhidrosis, Dr. Hightower said that patients most commonly complain that their underarms are too smelly, too sweaty, and red, itchy, or painful. Causes, he said, can include irritation/contact dermatitis, folliculitis, and seborrheic dermatitis, as well as hyperhidrosis or HS.
Primary focal axillary hyperhidrosis is defined as focal, visible, excessive sweating for at least 6 months without an apparent cause plus at least two of the following characteristics: Sweating is bilateral and relatively symmetric, it impairs daily activities, it starts before the age of 25 with at least one episode per week (many patients have it daily), a family history of idiopathic hyperhidrosis is present, and focal sweating does not occur during sleep.
Secondary hyperhidrosis can be linked to other conditions, such as a spinal column injury, Dr. Hightower noted.
The first step on the treatment ladder is topical 20% aluminum chloride, which is available over the counter. This should be applied nightly for 1 week then every 1-2 weeks, Dr. Hightower recommended. All of his patients with hyperhidrosis have had at least one trial of this treatment.
The next option is daily topical treatment with 2.4% glycopyrronium tosylate (Qbrexza) cloths, approved by the Food and Drug Administration in 2018 for primary axillary hyperhidrosis in patients aged 9 and older. According to the prescribing information, dry mouth was by far the most common treatment-associated adverse effect in clinical trials (24% versus almost 6% among those on vehicle). As for skin reactions, erythema occurred in about 17% of both the intervention and vehicle groups, and burning/stinging occurred in 14% of those on treatment and almost 17% of those on vehicle.
“If they’re not able to get access to the cloths due to [insurance] coverage issues, or they don’t allow them to reach the clinical endpoint desired, then I use an oral daily glycopyrrolate pill,” Dr. Hightower said.
He recommends 1 mg to 6 mg daily of the anticholinergic drug, which has been used off-label for hyperhidrosis for several years. A 2012 study of 31 children with hyperhidrosis, he noted, supported the use of the drug. The retrospective study found that 90% of the patients, at a mean daily dose of 2 mg, experienced improvements, reported as major in 71%. In addition, patients experienced improvement within hours of taking the medication, and benefits disappeared within a day of stopping the medication. In the study, patients were on the treatment for an average of 2.1 years, and 29% experienced side effects, which were dose related; the most common were dry mouth in 26% and dry eyes in 10%.
According to goodrx.com, a month’s supply of 2 mg of the drug costs as little as $13 with a discount or coupon.
The next steps in treatment are procedural interventions such as microwave-based therapies.
Dr. Hightower said that patients should be advised that treatment may take years, and to encourage them to return for follow-up. He suggested this helpful message: “We’re still trying to find the best treatment for you, and we’ll need to see you back in the office.”
Hidradenitis suppurativa
Dr. Hightower said that too often, HS goes undiagnosed for a significant period of time, preventing patients from seeing a dermatologist for treatment. Hallmarks of HS include inflammatory nodules, abscesses, and scarring, he said. “It can be disfiguring, painful, embarrassing, and associated with significantly decreased quality of life. Early recognition in terms of making and solidifying the diagnosis is important so we can prevent further worsening of the disease.”
The goal of treatment include preventing scars and unnecessary emergency department visits, and stopping flares from worsening, Dr. Hightower said. For specifics, he pointed to clinical management guidelines released by the United States and Canadian hidradenitis suppurativa foundations in 2019.
Make sure to set individualized treatment goals and understand the impact of treatment on the patient’s interactions with family, school, and peers, he said. And keep in mind that “parent-defined goals may be different from patient-defined goals.”
Dr. Hightower reported no relevant disclosures. MedscapeLive and this news organization are owned by the same parent company
A wide variety of medications exists for treating hyperhidrosis, a dermatologist told colleagues, but before prescribing anything to a pediatric patient, he recommended, ask the patient a simple question: “What bothers you the most?”
The answer will provide guidance for developing a step-by-step treatment strategy and help provide the patient “a set of realistic expectations in terms of what the response will look like,” George Hightower, MD, PhD, a pediatric dermatologist at Rady Children’s Hospital and the University of California, San Diego, said at MedscapeLive’s Women’s & Pediatric Dermatology Seminar.
A similar question-based approach will help guide therapy for patients with hidradenitis suppurativa (HS), he said.
With regards to hyperhidrosis, Dr. Hightower said that patients most commonly complain that their underarms are too smelly, too sweaty, and red, itchy, or painful. Causes, he said, can include irritation/contact dermatitis, folliculitis, and seborrheic dermatitis, as well as hyperhidrosis or HS.
Primary focal axillary hyperhidrosis is defined as focal, visible, excessive sweating for at least 6 months without an apparent cause plus at least two of the following characteristics: Sweating is bilateral and relatively symmetric, it impairs daily activities, it starts before the age of 25 with at least one episode per week (many patients have it daily), a family history of idiopathic hyperhidrosis is present, and focal sweating does not occur during sleep.
Secondary hyperhidrosis can be linked to other conditions, such as a spinal column injury, Dr. Hightower noted.
The first step on the treatment ladder is topical 20% aluminum chloride, which is available over the counter. This should be applied nightly for 1 week then every 1-2 weeks, Dr. Hightower recommended. All of his patients with hyperhidrosis have had at least one trial of this treatment.
The next option is daily topical treatment with 2.4% glycopyrronium tosylate (Qbrexza) cloths, approved by the Food and Drug Administration in 2018 for primary axillary hyperhidrosis in patients aged 9 and older. According to the prescribing information, dry mouth was by far the most common treatment-associated adverse effect in clinical trials (24% versus almost 6% among those on vehicle). As for skin reactions, erythema occurred in about 17% of both the intervention and vehicle groups, and burning/stinging occurred in 14% of those on treatment and almost 17% of those on vehicle.
“If they’re not able to get access to the cloths due to [insurance] coverage issues, or they don’t allow them to reach the clinical endpoint desired, then I use an oral daily glycopyrrolate pill,” Dr. Hightower said.
He recommends 1 mg to 6 mg daily of the anticholinergic drug, which has been used off-label for hyperhidrosis for several years. A 2012 study of 31 children with hyperhidrosis, he noted, supported the use of the drug. The retrospective study found that 90% of the patients, at a mean daily dose of 2 mg, experienced improvements, reported as major in 71%. In addition, patients experienced improvement within hours of taking the medication, and benefits disappeared within a day of stopping the medication. In the study, patients were on the treatment for an average of 2.1 years, and 29% experienced side effects, which were dose related; the most common were dry mouth in 26% and dry eyes in 10%.
According to goodrx.com, a month’s supply of 2 mg of the drug costs as little as $13 with a discount or coupon.
The next steps in treatment are procedural interventions such as microwave-based therapies.
Dr. Hightower said that patients should be advised that treatment may take years, and to encourage them to return for follow-up. He suggested this helpful message: “We’re still trying to find the best treatment for you, and we’ll need to see you back in the office.”
Hidradenitis suppurativa
Dr. Hightower said that too often, HS goes undiagnosed for a significant period of time, preventing patients from seeing a dermatologist for treatment. Hallmarks of HS include inflammatory nodules, abscesses, and scarring, he said. “It can be disfiguring, painful, embarrassing, and associated with significantly decreased quality of life. Early recognition in terms of making and solidifying the diagnosis is important so we can prevent further worsening of the disease.”
The goal of treatment include preventing scars and unnecessary emergency department visits, and stopping flares from worsening, Dr. Hightower said. For specifics, he pointed to clinical management guidelines released by the United States and Canadian hidradenitis suppurativa foundations in 2019.
Make sure to set individualized treatment goals and understand the impact of treatment on the patient’s interactions with family, school, and peers, he said. And keep in mind that “parent-defined goals may be different from patient-defined goals.”
Dr. Hightower reported no relevant disclosures. MedscapeLive and this news organization are owned by the same parent company
A wide variety of medications exists for treating hyperhidrosis, a dermatologist told colleagues, but before prescribing anything to a pediatric patient, he recommended, ask the patient a simple question: “What bothers you the most?”
The answer will provide guidance for developing a step-by-step treatment strategy and help provide the patient “a set of realistic expectations in terms of what the response will look like,” George Hightower, MD, PhD, a pediatric dermatologist at Rady Children’s Hospital and the University of California, San Diego, said at MedscapeLive’s Women’s & Pediatric Dermatology Seminar.
A similar question-based approach will help guide therapy for patients with hidradenitis suppurativa (HS), he said.
With regards to hyperhidrosis, Dr. Hightower said that patients most commonly complain that their underarms are too smelly, too sweaty, and red, itchy, or painful. Causes, he said, can include irritation/contact dermatitis, folliculitis, and seborrheic dermatitis, as well as hyperhidrosis or HS.
Primary focal axillary hyperhidrosis is defined as focal, visible, excessive sweating for at least 6 months without an apparent cause plus at least two of the following characteristics: Sweating is bilateral and relatively symmetric, it impairs daily activities, it starts before the age of 25 with at least one episode per week (many patients have it daily), a family history of idiopathic hyperhidrosis is present, and focal sweating does not occur during sleep.
Secondary hyperhidrosis can be linked to other conditions, such as a spinal column injury, Dr. Hightower noted.
The first step on the treatment ladder is topical 20% aluminum chloride, which is available over the counter. This should be applied nightly for 1 week then every 1-2 weeks, Dr. Hightower recommended. All of his patients with hyperhidrosis have had at least one trial of this treatment.
The next option is daily topical treatment with 2.4% glycopyrronium tosylate (Qbrexza) cloths, approved by the Food and Drug Administration in 2018 for primary axillary hyperhidrosis in patients aged 9 and older. According to the prescribing information, dry mouth was by far the most common treatment-associated adverse effect in clinical trials (24% versus almost 6% among those on vehicle). As for skin reactions, erythema occurred in about 17% of both the intervention and vehicle groups, and burning/stinging occurred in 14% of those on treatment and almost 17% of those on vehicle.
“If they’re not able to get access to the cloths due to [insurance] coverage issues, or they don’t allow them to reach the clinical endpoint desired, then I use an oral daily glycopyrrolate pill,” Dr. Hightower said.
He recommends 1 mg to 6 mg daily of the anticholinergic drug, which has been used off-label for hyperhidrosis for several years. A 2012 study of 31 children with hyperhidrosis, he noted, supported the use of the drug. The retrospective study found that 90% of the patients, at a mean daily dose of 2 mg, experienced improvements, reported as major in 71%. In addition, patients experienced improvement within hours of taking the medication, and benefits disappeared within a day of stopping the medication. In the study, patients were on the treatment for an average of 2.1 years, and 29% experienced side effects, which were dose related; the most common were dry mouth in 26% and dry eyes in 10%.
According to goodrx.com, a month’s supply of 2 mg of the drug costs as little as $13 with a discount or coupon.
The next steps in treatment are procedural interventions such as microwave-based therapies.
Dr. Hightower said that patients should be advised that treatment may take years, and to encourage them to return for follow-up. He suggested this helpful message: “We’re still trying to find the best treatment for you, and we’ll need to see you back in the office.”
Hidradenitis suppurativa
Dr. Hightower said that too often, HS goes undiagnosed for a significant period of time, preventing patients from seeing a dermatologist for treatment. Hallmarks of HS include inflammatory nodules, abscesses, and scarring, he said. “It can be disfiguring, painful, embarrassing, and associated with significantly decreased quality of life. Early recognition in terms of making and solidifying the diagnosis is important so we can prevent further worsening of the disease.”
The goal of treatment include preventing scars and unnecessary emergency department visits, and stopping flares from worsening, Dr. Hightower said. For specifics, he pointed to clinical management guidelines released by the United States and Canadian hidradenitis suppurativa foundations in 2019.
Make sure to set individualized treatment goals and understand the impact of treatment on the patient’s interactions with family, school, and peers, he said. And keep in mind that “parent-defined goals may be different from patient-defined goals.”
Dr. Hightower reported no relevant disclosures. MedscapeLive and this news organization are owned by the same parent company
FROM MEDSCAPELIVE WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR