User login
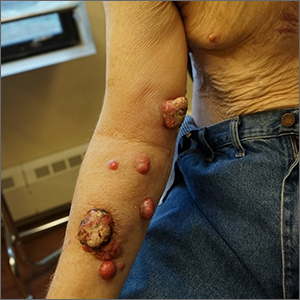
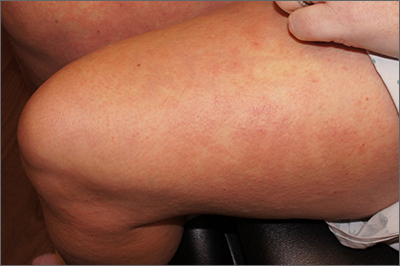
Nodules on the arm
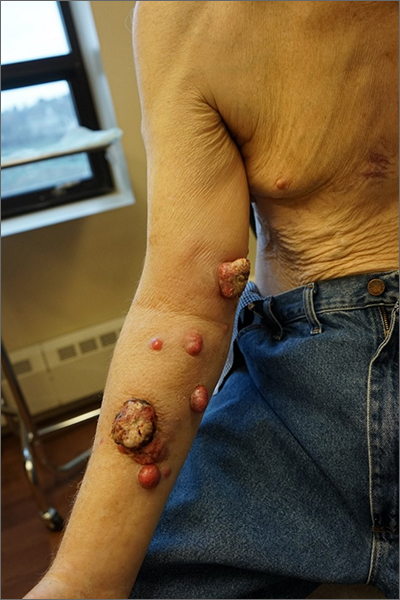
Excisional biopsy was performed on a small tumor and immune staining suggested lung cancer as the primary source.
Metastatic cancer of an unknown primary source accounts for 3% to 5% of invasive cancers.1 Poorly differentiated SCC, as in this case, may arise from skin, lung, or head and neck SCC. Risks of metastatic disease in cutaneous SCC include size greater than 2 cm, depth greater than 2 mm, and location on the ear, hand, lip, or within a burn.2 Recurrent tumors, poorly differentiated tumors, and tumors demonstrating perineural invasion also increase the risk of metastasis.
Having failed both previous treatments, further tissue characterization offered some hope of effective immunotherapy. Specifically, SCC from lung cancer as a primary source may express programmed-death ligand 1 (PD-L1), a transmembrane protein that suppresses adaptive immune responses. Pembrolizumab inhibits this molecule leading to a more aggressive immune response to tumor cells. Additionally, tumor profiling of specific oncogenes can highlight potentially beneficial therapies.
In this case, gene profiling identified several active tumor oncogenes, but PD-L1 was not strongly expressed. Despite the effort, this process did not uncover practical or novel treatment options. The patient was offered an empiric trial of immunotherapy but opted to pursue palliative therapy alone and passed away 2 months later.
This case highlights the role of multidisciplinary care of metastatic disease to the skin and the potential role, and limitations, of skin biopsy in tumor profiling and treatment guidance. Even though the most likely primary site was lung, cutaneous metastases can be equally devastating.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379:1428-1435. doi: 10.1016/S0140-6736(11)61178-1
2. Brougham NDLS, Dennett ER, Cameron R, et al. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811-815. doi: 10.1002/jso.23155

Excisional biopsy was performed on a small tumor and immune staining suggested lung cancer as the primary source.
Metastatic cancer of an unknown primary source accounts for 3% to 5% of invasive cancers.1 Poorly differentiated SCC, as in this case, may arise from skin, lung, or head and neck SCC. Risks of metastatic disease in cutaneous SCC include size greater than 2 cm, depth greater than 2 mm, and location on the ear, hand, lip, or within a burn.2 Recurrent tumors, poorly differentiated tumors, and tumors demonstrating perineural invasion also increase the risk of metastasis.
Having failed both previous treatments, further tissue characterization offered some hope of effective immunotherapy. Specifically, SCC from lung cancer as a primary source may express programmed-death ligand 1 (PD-L1), a transmembrane protein that suppresses adaptive immune responses. Pembrolizumab inhibits this molecule leading to a more aggressive immune response to tumor cells. Additionally, tumor profiling of specific oncogenes can highlight potentially beneficial therapies.
In this case, gene profiling identified several active tumor oncogenes, but PD-L1 was not strongly expressed. Despite the effort, this process did not uncover practical or novel treatment options. The patient was offered an empiric trial of immunotherapy but opted to pursue palliative therapy alone and passed away 2 months later.
This case highlights the role of multidisciplinary care of metastatic disease to the skin and the potential role, and limitations, of skin biopsy in tumor profiling and treatment guidance. Even though the most likely primary site was lung, cutaneous metastases can be equally devastating.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Excisional biopsy was performed on a small tumor and immune staining suggested lung cancer as the primary source.
Metastatic cancer of an unknown primary source accounts for 3% to 5% of invasive cancers.1 Poorly differentiated SCC, as in this case, may arise from skin, lung, or head and neck SCC. Risks of metastatic disease in cutaneous SCC include size greater than 2 cm, depth greater than 2 mm, and location on the ear, hand, lip, or within a burn.2 Recurrent tumors, poorly differentiated tumors, and tumors demonstrating perineural invasion also increase the risk of metastasis.
Having failed both previous treatments, further tissue characterization offered some hope of effective immunotherapy. Specifically, SCC from lung cancer as a primary source may express programmed-death ligand 1 (PD-L1), a transmembrane protein that suppresses adaptive immune responses. Pembrolizumab inhibits this molecule leading to a more aggressive immune response to tumor cells. Additionally, tumor profiling of specific oncogenes can highlight potentially beneficial therapies.
In this case, gene profiling identified several active tumor oncogenes, but PD-L1 was not strongly expressed. Despite the effort, this process did not uncover practical or novel treatment options. The patient was offered an empiric trial of immunotherapy but opted to pursue palliative therapy alone and passed away 2 months later.
This case highlights the role of multidisciplinary care of metastatic disease to the skin and the potential role, and limitations, of skin biopsy in tumor profiling and treatment guidance. Even though the most likely primary site was lung, cutaneous metastases can be equally devastating.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379:1428-1435. doi: 10.1016/S0140-6736(11)61178-1
2. Brougham NDLS, Dennett ER, Cameron R, et al. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811-815. doi: 10.1002/jso.23155
1. Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379:1428-1435. doi: 10.1016/S0140-6736(11)61178-1
2. Brougham NDLS, Dennett ER, Cameron R, et al. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811-815. doi: 10.1002/jso.23155
New analysis puts U.S. psoriasis prevalence at 3%
, according to an analysis of national survey data from 2011 to 2014.
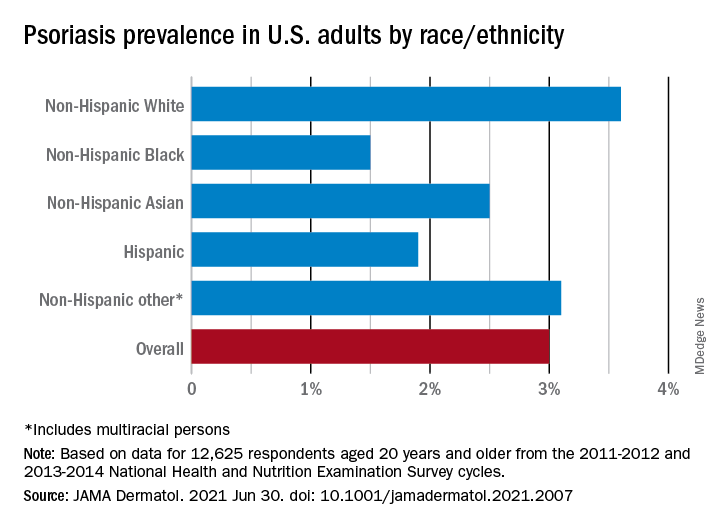
“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
, according to an analysis of national survey data from 2011 to 2014.

“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
, according to an analysis of national survey data from 2011 to 2014.

“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
FROM JAMA DERMATOLOGY
Expert shares practical considerations when prescribing dupilumab
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
FROM REVOLUTIONIZING AD 2021
Rash after a medication change
A punch biopsy revealed lichenoid interface dermatitis with eosinophils and mild spongiosis—consistent with, but not conclusive for, a drug eruption. A complete blood count (CBC) and comprehensive metabolic panel revealed elevated levels of eosinophils and transaminases which raised the possibility of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. In this patient’s case, the transaminitis suggested some mild hepatitis; the liver is the most common organ involved in DRESS.
Diagnostic criteria for DRESS syndrome need not always be met but can include fever, lymphadenopathy, facial edema, and a morbilliform rash presenting 2 to 3 weeks after drug exposure. In this case, a lack of fever, facial edema, and other systemic symptoms favored a less severe drug eruption or what has been described as mini-DRESS.1 DRESS syndrome often merits hospitalization, and in 10% of cases it can be fatal.1
The patient in this case was started on prednisone 60 mg/d and her OCPs were discontinued. One week later, a repeat CBC showed normalized levels of eosinophils and transaminases. However, shortly after a 3-week taper of the prednisone, her levels of eosinophils and transaminases rose again. A repeat prednisone taper finally led to complete resolution of rash and sustained normalization of eosinophil and transaminase levels.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102:322-326.

A punch biopsy revealed lichenoid interface dermatitis with eosinophils and mild spongiosis—consistent with, but not conclusive for, a drug eruption. A complete blood count (CBC) and comprehensive metabolic panel revealed elevated levels of eosinophils and transaminases which raised the possibility of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. In this patient’s case, the transaminitis suggested some mild hepatitis; the liver is the most common organ involved in DRESS.
Diagnostic criteria for DRESS syndrome need not always be met but can include fever, lymphadenopathy, facial edema, and a morbilliform rash presenting 2 to 3 weeks after drug exposure. In this case, a lack of fever, facial edema, and other systemic symptoms favored a less severe drug eruption or what has been described as mini-DRESS.1 DRESS syndrome often merits hospitalization, and in 10% of cases it can be fatal.1
The patient in this case was started on prednisone 60 mg/d and her OCPs were discontinued. One week later, a repeat CBC showed normalized levels of eosinophils and transaminases. However, shortly after a 3-week taper of the prednisone, her levels of eosinophils and transaminases rose again. A repeat prednisone taper finally led to complete resolution of rash and sustained normalization of eosinophil and transaminase levels.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

A punch biopsy revealed lichenoid interface dermatitis with eosinophils and mild spongiosis—consistent with, but not conclusive for, a drug eruption. A complete blood count (CBC) and comprehensive metabolic panel revealed elevated levels of eosinophils and transaminases which raised the possibility of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. In this patient’s case, the transaminitis suggested some mild hepatitis; the liver is the most common organ involved in DRESS.
Diagnostic criteria for DRESS syndrome need not always be met but can include fever, lymphadenopathy, facial edema, and a morbilliform rash presenting 2 to 3 weeks after drug exposure. In this case, a lack of fever, facial edema, and other systemic symptoms favored a less severe drug eruption or what has been described as mini-DRESS.1 DRESS syndrome often merits hospitalization, and in 10% of cases it can be fatal.1
The patient in this case was started on prednisone 60 mg/d and her OCPs were discontinued. One week later, a repeat CBC showed normalized levels of eosinophils and transaminases. However, shortly after a 3-week taper of the prednisone, her levels of eosinophils and transaminases rose again. A repeat prednisone taper finally led to complete resolution of rash and sustained normalization of eosinophil and transaminase levels.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102:322-326.
Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102:322-326.
Baricitinib found effective for moderate to severe AD out to 52 weeks
“With long-term therapy, the baricitinib 2 mg response remains stable or slightly improved, compared with week 16 for skin inflammation, itch, sleep, and quality of life,” presenting study author Eric L. Simpson, MD, said during the Revolutionizing Atopic Dermatitis symposium.
Baricitinib is an oral selective Janus kinase 1/JAK2 inhibitor being developed for the treatment of moderate to severe AD in adults who are candidates for systemic therapy. The drug is already approved for AD in Europe at the 2-mg and 4-mg doses. A 16-week placebo-controlled study conducted in North America known as BREEZE-AD5 found that 2 mg of baricitinib improved disease in adults with moderate to severe AD.
For the current analysis, Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland, and colleagues integrated data from BREEZE-AD5 and BREEZE-AD6, an ongoing, open-label study of BREEZE-AD5, to evaluate the long-term efficacy and safety of baricitinib 2 mg in patients with moderate to severe AD.
At week 16, patients from BREEZE-AD5 who were on baricitinib 2 mg could either continue the trial out to week 52, or they could transition to BREEZE-AD6 if they were nonresponders. The use of low-potency corticosteroids was permitted after week 16 in BREEZE-AD5 and throughout BREEZE-AD6. Endpoints of interest at week 52 in both trials were the proportions of patients with 75% or greater improvement from baseline in the Eczema and Severity Index (EASI75), a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1, a Dermatology Life Quality Index (DLQI) score of 5 or less, as well as mean SCORing AD (SCORAD) visual analog scales of itch and sleeplessness scores, and the mean percent change from baseline in EASI score.
Dr. Simpson presented data on 146 patients from both trials who were randomized to baricitinib 2 mg. Their mean age was 40 years, 53% were female, 58% were White, 21% were Black, 15% were Asian, and the remainder were from other backgrounds. Their mean duration of AD was 16 years and their average EASI score was 26.6. At weeks 16, 32, and 52, the proportion of patients who achieved an EASI75 response was 40%, 51%, and 49%, respectively, while the mean percent change from baseline in EASI score was –50%, –59%, and –57%.
At weeks 16, 32, and 52, the vIGA-AD responses of 0 or 1 were observed in 27%, 38%, and 31% of patients. The mean SCORAD pruritus score improved from 7.7 at baseline to 4.8 at week 16 and was maintained at weeks 32 (3.8) and 52 (4.3). The mean SCORAD sleeplessness score also improved from 6.5 at baseline to 3.9 at week 16 and remained stable through weeks 32 (3.4) and 52 (3.7).
Finally, among 129 patients who had a baseline DLQI of greater than 5, 39% had DLQI scores of 5 or lower at week 16, compared with 49% at week 32 and 45% at week 52, indicating a small or no effect of AD on quality of life.
The study was sponsored by Eli Lilly, which is developing baricitinib. Dr. Simpson disclosed that he is a consultant to and/or an investigator for several pharmaceutical companies, including Eli Lilly.
“With long-term therapy, the baricitinib 2 mg response remains stable or slightly improved, compared with week 16 for skin inflammation, itch, sleep, and quality of life,” presenting study author Eric L. Simpson, MD, said during the Revolutionizing Atopic Dermatitis symposium.
Baricitinib is an oral selective Janus kinase 1/JAK2 inhibitor being developed for the treatment of moderate to severe AD in adults who are candidates for systemic therapy. The drug is already approved for AD in Europe at the 2-mg and 4-mg doses. A 16-week placebo-controlled study conducted in North America known as BREEZE-AD5 found that 2 mg of baricitinib improved disease in adults with moderate to severe AD.
For the current analysis, Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland, and colleagues integrated data from BREEZE-AD5 and BREEZE-AD6, an ongoing, open-label study of BREEZE-AD5, to evaluate the long-term efficacy and safety of baricitinib 2 mg in patients with moderate to severe AD.
At week 16, patients from BREEZE-AD5 who were on baricitinib 2 mg could either continue the trial out to week 52, or they could transition to BREEZE-AD6 if they were nonresponders. The use of low-potency corticosteroids was permitted after week 16 in BREEZE-AD5 and throughout BREEZE-AD6. Endpoints of interest at week 52 in both trials were the proportions of patients with 75% or greater improvement from baseline in the Eczema and Severity Index (EASI75), a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1, a Dermatology Life Quality Index (DLQI) score of 5 or less, as well as mean SCORing AD (SCORAD) visual analog scales of itch and sleeplessness scores, and the mean percent change from baseline in EASI score.
Dr. Simpson presented data on 146 patients from both trials who were randomized to baricitinib 2 mg. Their mean age was 40 years, 53% were female, 58% were White, 21% were Black, 15% were Asian, and the remainder were from other backgrounds. Their mean duration of AD was 16 years and their average EASI score was 26.6. At weeks 16, 32, and 52, the proportion of patients who achieved an EASI75 response was 40%, 51%, and 49%, respectively, while the mean percent change from baseline in EASI score was –50%, –59%, and –57%.
At weeks 16, 32, and 52, the vIGA-AD responses of 0 or 1 were observed in 27%, 38%, and 31% of patients. The mean SCORAD pruritus score improved from 7.7 at baseline to 4.8 at week 16 and was maintained at weeks 32 (3.8) and 52 (4.3). The mean SCORAD sleeplessness score also improved from 6.5 at baseline to 3.9 at week 16 and remained stable through weeks 32 (3.4) and 52 (3.7).
Finally, among 129 patients who had a baseline DLQI of greater than 5, 39% had DLQI scores of 5 or lower at week 16, compared with 49% at week 32 and 45% at week 52, indicating a small or no effect of AD on quality of life.
The study was sponsored by Eli Lilly, which is developing baricitinib. Dr. Simpson disclosed that he is a consultant to and/or an investigator for several pharmaceutical companies, including Eli Lilly.
“With long-term therapy, the baricitinib 2 mg response remains stable or slightly improved, compared with week 16 for skin inflammation, itch, sleep, and quality of life,” presenting study author Eric L. Simpson, MD, said during the Revolutionizing Atopic Dermatitis symposium.
Baricitinib is an oral selective Janus kinase 1/JAK2 inhibitor being developed for the treatment of moderate to severe AD in adults who are candidates for systemic therapy. The drug is already approved for AD in Europe at the 2-mg and 4-mg doses. A 16-week placebo-controlled study conducted in North America known as BREEZE-AD5 found that 2 mg of baricitinib improved disease in adults with moderate to severe AD.
For the current analysis, Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland, and colleagues integrated data from BREEZE-AD5 and BREEZE-AD6, an ongoing, open-label study of BREEZE-AD5, to evaluate the long-term efficacy and safety of baricitinib 2 mg in patients with moderate to severe AD.
At week 16, patients from BREEZE-AD5 who were on baricitinib 2 mg could either continue the trial out to week 52, or they could transition to BREEZE-AD6 if they were nonresponders. The use of low-potency corticosteroids was permitted after week 16 in BREEZE-AD5 and throughout BREEZE-AD6. Endpoints of interest at week 52 in both trials were the proportions of patients with 75% or greater improvement from baseline in the Eczema and Severity Index (EASI75), a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1, a Dermatology Life Quality Index (DLQI) score of 5 or less, as well as mean SCORing AD (SCORAD) visual analog scales of itch and sleeplessness scores, and the mean percent change from baseline in EASI score.
Dr. Simpson presented data on 146 patients from both trials who were randomized to baricitinib 2 mg. Their mean age was 40 years, 53% were female, 58% were White, 21% were Black, 15% were Asian, and the remainder were from other backgrounds. Their mean duration of AD was 16 years and their average EASI score was 26.6. At weeks 16, 32, and 52, the proportion of patients who achieved an EASI75 response was 40%, 51%, and 49%, respectively, while the mean percent change from baseline in EASI score was –50%, –59%, and –57%.
At weeks 16, 32, and 52, the vIGA-AD responses of 0 or 1 were observed in 27%, 38%, and 31% of patients. The mean SCORAD pruritus score improved from 7.7 at baseline to 4.8 at week 16 and was maintained at weeks 32 (3.8) and 52 (4.3). The mean SCORAD sleeplessness score also improved from 6.5 at baseline to 3.9 at week 16 and remained stable through weeks 32 (3.4) and 52 (3.7).
Finally, among 129 patients who had a baseline DLQI of greater than 5, 39% had DLQI scores of 5 or lower at week 16, compared with 49% at week 32 and 45% at week 52, indicating a small or no effect of AD on quality of life.
The study was sponsored by Eli Lilly, which is developing baricitinib. Dr. Simpson disclosed that he is a consultant to and/or an investigator for several pharmaceutical companies, including Eli Lilly.
FROM REVOLUTIONIZING AD 2021
In Black patients, acne scarring might not mean what you think
Treating the needs of patients of color requires an understanding of differences that may not be readily apparent, a dermatologist told colleagues. For example, of the term that may be misinterpreted in the doctor’s office.
“Scarring is not usually what they’re talking about, although they may have some of that as well. They’re [typically] talking about what we know as postinflammatory hyperpigmentation, not scarring. So right away, you have to clarify,” Amy McMichael, MD, professor and chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in a presentation at the Inaugural Symposium for Inflammatory Skin Disease. “When you’re talking about scarring, do you mean the dark spots? What exactly are you concerned about?”
Dr. McMichael highlighted a 2014 study that reported the results of a survey of 208 women (51% were White; 49% were non-White), which included 51 Black, 23 Hispanic, and 16 Asian women aged 25-45 (mean age, 35) with 25 or more lesions. White women were more troubled by facial acne than were women of color (89% vs. 76%, respectively, P < .05), and they were more likely to say lesion clearance was most important to them (58% vs. 32%, respectively, P < .001).
Meanwhile, non-White women were much more likely than were White women to say that clearance of postinflammatory hyperpigmentation was most important to them (42% vs. 8%, respectively, P < .0001).
“Seventy percent of [non-White women] felt that their race and ethnicity required targeted attention [in treatment], and two-thirds desired acne treatment that was designed to meet the needs of their skin type,” Dr. McMichael said. “If you don’t address the issues, if you don’t talk about the pigmentation with them or explain how you’re going to address it, people don’t feel heard. They don’t feel like they’ve really seen a dermatologist who understands their needs.”
She added that it’s crucial to ask about over-the-counter products. “If you don’t discuss them, they’ll assume that what they’re doing is okay.” She warns her patients against using and exposing their skin and face to cocoa butter and oils such as tea tree oil.
Research has suggested that among people of color, Blacks and Hispanics are most likely to experience dyspigmentation and scarring, Dr. McMichael said. She advised colleagues to be aware of pomade acne in these two groups of patients. Pomade acne appears along the hair line and is caused by the use of hair products. She also cautioned about acne cosmetica, which can be triggered by products such as makeup, used to cover up acne and postinflammatory hyperpigmentation.
As for acne treatments, Dr. McMichael highlighted a long list of familiar topical and oral agents and procedural options. Less familiar strategies include laser and light-based therapies, she said.
As for up-and-coming options, she pointed to topical minocycline, “which allows us to use an anti-inflammatory agent topically rather than orally when we’re trying to get away from using a lot of oral antibiotics.”
Also consider whether female patients have polycystic ovary syndrome, she said. “Then you might consider spironolactone. I certainly use a lot more of that these days to try to avoid long-term oral antibiotics.”
She recommended earlier use of isotretinoin in patients overall, and she urged colleagues to proceed with their standard retinoid approaches. However, she noted that she lets patients know that she’ll focus first on treating the acne itself and then work on the dark spots in later treatments. “If you give people a bleaching agent in the beginning, they’re going to stop using their main products, and they’re going to chase those dark spots. That’s just something that they can’t help doing.”
Dr. McMichael disclosed investigator and consultant relationships with multiple drug makers.
Treating the needs of patients of color requires an understanding of differences that may not be readily apparent, a dermatologist told colleagues. For example, of the term that may be misinterpreted in the doctor’s office.
“Scarring is not usually what they’re talking about, although they may have some of that as well. They’re [typically] talking about what we know as postinflammatory hyperpigmentation, not scarring. So right away, you have to clarify,” Amy McMichael, MD, professor and chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in a presentation at the Inaugural Symposium for Inflammatory Skin Disease. “When you’re talking about scarring, do you mean the dark spots? What exactly are you concerned about?”
Dr. McMichael highlighted a 2014 study that reported the results of a survey of 208 women (51% were White; 49% were non-White), which included 51 Black, 23 Hispanic, and 16 Asian women aged 25-45 (mean age, 35) with 25 or more lesions. White women were more troubled by facial acne than were women of color (89% vs. 76%, respectively, P < .05), and they were more likely to say lesion clearance was most important to them (58% vs. 32%, respectively, P < .001).
Meanwhile, non-White women were much more likely than were White women to say that clearance of postinflammatory hyperpigmentation was most important to them (42% vs. 8%, respectively, P < .0001).
“Seventy percent of [non-White women] felt that their race and ethnicity required targeted attention [in treatment], and two-thirds desired acne treatment that was designed to meet the needs of their skin type,” Dr. McMichael said. “If you don’t address the issues, if you don’t talk about the pigmentation with them or explain how you’re going to address it, people don’t feel heard. They don’t feel like they’ve really seen a dermatologist who understands their needs.”
She added that it’s crucial to ask about over-the-counter products. “If you don’t discuss them, they’ll assume that what they’re doing is okay.” She warns her patients against using and exposing their skin and face to cocoa butter and oils such as tea tree oil.
Research has suggested that among people of color, Blacks and Hispanics are most likely to experience dyspigmentation and scarring, Dr. McMichael said. She advised colleagues to be aware of pomade acne in these two groups of patients. Pomade acne appears along the hair line and is caused by the use of hair products. She also cautioned about acne cosmetica, which can be triggered by products such as makeup, used to cover up acne and postinflammatory hyperpigmentation.
As for acne treatments, Dr. McMichael highlighted a long list of familiar topical and oral agents and procedural options. Less familiar strategies include laser and light-based therapies, she said.
As for up-and-coming options, she pointed to topical minocycline, “which allows us to use an anti-inflammatory agent topically rather than orally when we’re trying to get away from using a lot of oral antibiotics.”
Also consider whether female patients have polycystic ovary syndrome, she said. “Then you might consider spironolactone. I certainly use a lot more of that these days to try to avoid long-term oral antibiotics.”
She recommended earlier use of isotretinoin in patients overall, and she urged colleagues to proceed with their standard retinoid approaches. However, she noted that she lets patients know that she’ll focus first on treating the acne itself and then work on the dark spots in later treatments. “If you give people a bleaching agent in the beginning, they’re going to stop using their main products, and they’re going to chase those dark spots. That’s just something that they can’t help doing.”
Dr. McMichael disclosed investigator and consultant relationships with multiple drug makers.
Treating the needs of patients of color requires an understanding of differences that may not be readily apparent, a dermatologist told colleagues. For example, of the term that may be misinterpreted in the doctor’s office.
“Scarring is not usually what they’re talking about, although they may have some of that as well. They’re [typically] talking about what we know as postinflammatory hyperpigmentation, not scarring. So right away, you have to clarify,” Amy McMichael, MD, professor and chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in a presentation at the Inaugural Symposium for Inflammatory Skin Disease. “When you’re talking about scarring, do you mean the dark spots? What exactly are you concerned about?”
Dr. McMichael highlighted a 2014 study that reported the results of a survey of 208 women (51% were White; 49% were non-White), which included 51 Black, 23 Hispanic, and 16 Asian women aged 25-45 (mean age, 35) with 25 or more lesions. White women were more troubled by facial acne than were women of color (89% vs. 76%, respectively, P < .05), and they were more likely to say lesion clearance was most important to them (58% vs. 32%, respectively, P < .001).
Meanwhile, non-White women were much more likely than were White women to say that clearance of postinflammatory hyperpigmentation was most important to them (42% vs. 8%, respectively, P < .0001).
“Seventy percent of [non-White women] felt that their race and ethnicity required targeted attention [in treatment], and two-thirds desired acne treatment that was designed to meet the needs of their skin type,” Dr. McMichael said. “If you don’t address the issues, if you don’t talk about the pigmentation with them or explain how you’re going to address it, people don’t feel heard. They don’t feel like they’ve really seen a dermatologist who understands their needs.”
She added that it’s crucial to ask about over-the-counter products. “If you don’t discuss them, they’ll assume that what they’re doing is okay.” She warns her patients against using and exposing their skin and face to cocoa butter and oils such as tea tree oil.
Research has suggested that among people of color, Blacks and Hispanics are most likely to experience dyspigmentation and scarring, Dr. McMichael said. She advised colleagues to be aware of pomade acne in these two groups of patients. Pomade acne appears along the hair line and is caused by the use of hair products. She also cautioned about acne cosmetica, which can be triggered by products such as makeup, used to cover up acne and postinflammatory hyperpigmentation.
As for acne treatments, Dr. McMichael highlighted a long list of familiar topical and oral agents and procedural options. Less familiar strategies include laser and light-based therapies, she said.
As for up-and-coming options, she pointed to topical minocycline, “which allows us to use an anti-inflammatory agent topically rather than orally when we’re trying to get away from using a lot of oral antibiotics.”
Also consider whether female patients have polycystic ovary syndrome, she said. “Then you might consider spironolactone. I certainly use a lot more of that these days to try to avoid long-term oral antibiotics.”
She recommended earlier use of isotretinoin in patients overall, and she urged colleagues to proceed with their standard retinoid approaches. However, she noted that she lets patients know that she’ll focus first on treating the acne itself and then work on the dark spots in later treatments. “If you give people a bleaching agent in the beginning, they’re going to stop using their main products, and they’re going to chase those dark spots. That’s just something that they can’t help doing.”
Dr. McMichael disclosed investigator and consultant relationships with multiple drug makers.
FROM SISD 2021
Ruxolitinib cream for atopic dermatitis found to be effective, safe up to 52 weeks
results from a long-term analysis of clinical trial data showed.
“The incidence of application-site reactions was low, and there were no clinically meaningful changes or trends in hematologic parameters,” Kim Papp, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium.
Ruxolitinib cream is a selective Janus kinase 1/JAK2 inhibitor being developed by Incyte for the treatment of atopic dermatitis (AD).
According to a press release from the company, the Food and Drug Administration has extended the New Drug Application review period for the agent by 3 months to September 2021. If approved, it would become first topical JAK inhibitor for use in dermatology.
In two phase 3, randomized studies of identical design involving 1,249 patients aged 12 and older with AD – TRuE-AD1 and TRuE-AD2 – ruxolitinib cream demonstrated anti-inflammatory activity, with rapid and sustained antipruritic action, compared with vehicle. To be eligible for the trials patients with an Investigator’s Global Assessment (IGA) score of 2 or 3 and 3%-20% of affected body surface area (BSA) were randomized (2:2:1) to twice-daily 0.75% ruxolitinib cream, 1.5% ruxolitinib cream, or vehicle cream for 8 continuous weeks.
A recently published report found that significantly more patients in TRuE-AD1 and TRuE-AD2 achieved IGA treatment success with 0.75% (50% vs. 39%, respectively) and 1.5% ruxolitinib cream (53.8% vs. 51.3%), compared with vehicle (15.1% vs. 7.6%; P < .0001) at week 8. In addition, significant reductions in itch, compared with vehicle, were reported within 12 hours of first applying 1.5% ruxolitinib cream (P < .05).
Longterm data
During the symposium, Dr. Papp presented long-term safety data of ruxolitinib cream in patients who were followed for an additional 44 weeks. Those initially randomized to vehicle were rerandomized 1:1 (blinded) to either ruxolitinib cream regimen. They were instructed to treat skin areas with active AD only and to stop treatment 3 days after clearance of lesions, and to restart treatment with ruxolitinib cream at the first sign of recurrence. Safety and tolerability were assessed by frequency and severity of adverse events, while disease control was measured by the proportion of patients with an IGA score of 0 or 1 and the affected BSA.
Dr. Papp, a dermatologist and founder of Probity Medical Research, Waterloo, Ont., reported that 543 patients from TRuE-AD1 and 530 from TRuE-AD2 entered the long-term analysis and that about 78% of these patients completed the study. From weeks 12 to 52, the proportion of patients with an IGA score of 0 or 1 with 0.75% and 1.5% ruxolitinib cream ranged from 62%-77% and 67%-77%, respectively, in TRuE-AD1 to 60%-77% and 72%-80% in TRuE-AD2.
The measured mean total affected BSA was less than 3% throughout the follow-up period in the 1.5% ruxolitinib cream arm in TRuE-AD1 and TRuE-AD2 and was less than 3% in the 0.75% ruxolitinib cream arm during most of the study period.
In a pooled safety analysis, treatment-emergent adverse events (TEAEs) were reported in 60% and 54% of patients who applied 0.75% and 1.5% ruxolitinib cream, respectively, over 44 weeks. The frequency of application-site reactions remained low. Specifically, treatment-related adverse events were reported in 5% of patients who applied 0.75% ruxolitinib cream and in 3% of patients who applied 1.5% ruxolitinib cream; none were serious. TEAEs led to discontinuation in 2% of patients in the 0.75% ruxolitinib cream group, and no patients in the 1.5% ruxolitinib cream group.
“The most common treatment adverse events were upper respiratory tract infections and nasopharyngitis,” Dr. Papp said. “When looking at exposure-adjusted adverse events, we see that there is a high degree of similarity between any of the TEAEs across all of the treatment groups in both studies. We also see that it was patients on the vehicle who experienced the greatest number of application-site reactions.”
Dr. Papp disclosed that he has received honoraria or clinical research grants as a consultant, speaker, scientific officer, advisory board member, and/or steering committee member for several pharmaceutical companies, including Incyte.
results from a long-term analysis of clinical trial data showed.
“The incidence of application-site reactions was low, and there were no clinically meaningful changes or trends in hematologic parameters,” Kim Papp, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium.
Ruxolitinib cream is a selective Janus kinase 1/JAK2 inhibitor being developed by Incyte for the treatment of atopic dermatitis (AD).
According to a press release from the company, the Food and Drug Administration has extended the New Drug Application review period for the agent by 3 months to September 2021. If approved, it would become first topical JAK inhibitor for use in dermatology.
In two phase 3, randomized studies of identical design involving 1,249 patients aged 12 and older with AD – TRuE-AD1 and TRuE-AD2 – ruxolitinib cream demonstrated anti-inflammatory activity, with rapid and sustained antipruritic action, compared with vehicle. To be eligible for the trials patients with an Investigator’s Global Assessment (IGA) score of 2 or 3 and 3%-20% of affected body surface area (BSA) were randomized (2:2:1) to twice-daily 0.75% ruxolitinib cream, 1.5% ruxolitinib cream, or vehicle cream for 8 continuous weeks.
A recently published report found that significantly more patients in TRuE-AD1 and TRuE-AD2 achieved IGA treatment success with 0.75% (50% vs. 39%, respectively) and 1.5% ruxolitinib cream (53.8% vs. 51.3%), compared with vehicle (15.1% vs. 7.6%; P < .0001) at week 8. In addition, significant reductions in itch, compared with vehicle, were reported within 12 hours of first applying 1.5% ruxolitinib cream (P < .05).
Longterm data
During the symposium, Dr. Papp presented long-term safety data of ruxolitinib cream in patients who were followed for an additional 44 weeks. Those initially randomized to vehicle were rerandomized 1:1 (blinded) to either ruxolitinib cream regimen. They were instructed to treat skin areas with active AD only and to stop treatment 3 days after clearance of lesions, and to restart treatment with ruxolitinib cream at the first sign of recurrence. Safety and tolerability were assessed by frequency and severity of adverse events, while disease control was measured by the proportion of patients with an IGA score of 0 or 1 and the affected BSA.
Dr. Papp, a dermatologist and founder of Probity Medical Research, Waterloo, Ont., reported that 543 patients from TRuE-AD1 and 530 from TRuE-AD2 entered the long-term analysis and that about 78% of these patients completed the study. From weeks 12 to 52, the proportion of patients with an IGA score of 0 or 1 with 0.75% and 1.5% ruxolitinib cream ranged from 62%-77% and 67%-77%, respectively, in TRuE-AD1 to 60%-77% and 72%-80% in TRuE-AD2.
The measured mean total affected BSA was less than 3% throughout the follow-up period in the 1.5% ruxolitinib cream arm in TRuE-AD1 and TRuE-AD2 and was less than 3% in the 0.75% ruxolitinib cream arm during most of the study period.
In a pooled safety analysis, treatment-emergent adverse events (TEAEs) were reported in 60% and 54% of patients who applied 0.75% and 1.5% ruxolitinib cream, respectively, over 44 weeks. The frequency of application-site reactions remained low. Specifically, treatment-related adverse events were reported in 5% of patients who applied 0.75% ruxolitinib cream and in 3% of patients who applied 1.5% ruxolitinib cream; none were serious. TEAEs led to discontinuation in 2% of patients in the 0.75% ruxolitinib cream group, and no patients in the 1.5% ruxolitinib cream group.
“The most common treatment adverse events were upper respiratory tract infections and nasopharyngitis,” Dr. Papp said. “When looking at exposure-adjusted adverse events, we see that there is a high degree of similarity between any of the TEAEs across all of the treatment groups in both studies. We also see that it was patients on the vehicle who experienced the greatest number of application-site reactions.”
Dr. Papp disclosed that he has received honoraria or clinical research grants as a consultant, speaker, scientific officer, advisory board member, and/or steering committee member for several pharmaceutical companies, including Incyte.
results from a long-term analysis of clinical trial data showed.
“The incidence of application-site reactions was low, and there were no clinically meaningful changes or trends in hematologic parameters,” Kim Papp, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium.
Ruxolitinib cream is a selective Janus kinase 1/JAK2 inhibitor being developed by Incyte for the treatment of atopic dermatitis (AD).
According to a press release from the company, the Food and Drug Administration has extended the New Drug Application review period for the agent by 3 months to September 2021. If approved, it would become first topical JAK inhibitor for use in dermatology.
In two phase 3, randomized studies of identical design involving 1,249 patients aged 12 and older with AD – TRuE-AD1 and TRuE-AD2 – ruxolitinib cream demonstrated anti-inflammatory activity, with rapid and sustained antipruritic action, compared with vehicle. To be eligible for the trials patients with an Investigator’s Global Assessment (IGA) score of 2 or 3 and 3%-20% of affected body surface area (BSA) were randomized (2:2:1) to twice-daily 0.75% ruxolitinib cream, 1.5% ruxolitinib cream, or vehicle cream for 8 continuous weeks.
A recently published report found that significantly more patients in TRuE-AD1 and TRuE-AD2 achieved IGA treatment success with 0.75% (50% vs. 39%, respectively) and 1.5% ruxolitinib cream (53.8% vs. 51.3%), compared with vehicle (15.1% vs. 7.6%; P < .0001) at week 8. In addition, significant reductions in itch, compared with vehicle, were reported within 12 hours of first applying 1.5% ruxolitinib cream (P < .05).
Longterm data
During the symposium, Dr. Papp presented long-term safety data of ruxolitinib cream in patients who were followed for an additional 44 weeks. Those initially randomized to vehicle were rerandomized 1:1 (blinded) to either ruxolitinib cream regimen. They were instructed to treat skin areas with active AD only and to stop treatment 3 days after clearance of lesions, and to restart treatment with ruxolitinib cream at the first sign of recurrence. Safety and tolerability were assessed by frequency and severity of adverse events, while disease control was measured by the proportion of patients with an IGA score of 0 or 1 and the affected BSA.
Dr. Papp, a dermatologist and founder of Probity Medical Research, Waterloo, Ont., reported that 543 patients from TRuE-AD1 and 530 from TRuE-AD2 entered the long-term analysis and that about 78% of these patients completed the study. From weeks 12 to 52, the proportion of patients with an IGA score of 0 or 1 with 0.75% and 1.5% ruxolitinib cream ranged from 62%-77% and 67%-77%, respectively, in TRuE-AD1 to 60%-77% and 72%-80% in TRuE-AD2.
The measured mean total affected BSA was less than 3% throughout the follow-up period in the 1.5% ruxolitinib cream arm in TRuE-AD1 and TRuE-AD2 and was less than 3% in the 0.75% ruxolitinib cream arm during most of the study period.
In a pooled safety analysis, treatment-emergent adverse events (TEAEs) were reported in 60% and 54% of patients who applied 0.75% and 1.5% ruxolitinib cream, respectively, over 44 weeks. The frequency of application-site reactions remained low. Specifically, treatment-related adverse events were reported in 5% of patients who applied 0.75% ruxolitinib cream and in 3% of patients who applied 1.5% ruxolitinib cream; none were serious. TEAEs led to discontinuation in 2% of patients in the 0.75% ruxolitinib cream group, and no patients in the 1.5% ruxolitinib cream group.
“The most common treatment adverse events were upper respiratory tract infections and nasopharyngitis,” Dr. Papp said. “When looking at exposure-adjusted adverse events, we see that there is a high degree of similarity between any of the TEAEs across all of the treatment groups in both studies. We also see that it was patients on the vehicle who experienced the greatest number of application-site reactions.”
Dr. Papp disclosed that he has received honoraria or clinical research grants as a consultant, speaker, scientific officer, advisory board member, and/or steering committee member for several pharmaceutical companies, including Incyte.
FROM REVOLUTIONIZING AD 2021
Survey spotlights the out-of-pocket burden on Blacks with atopic dermatitis
They also have significantly poorer disease control and an increased rate of comorbid skin infections.
Those are among the key findings from a 25-question survey administered to members of the National Eczema Association.
“Black individuals with AD have a unique sociodemographic and disease profile,” lead study investigator Raj Chovatiya, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium. “Out-of-pocket expenses are just one component of the real-world burden faced by this population.”
According to Dr. Chovatiya, of the department of dermatology at Northwestern University, Chicago, the clinical phenotype and burden of AD can vary across racial and ethnic groups. Black race, for example, is associated with a higher prevalence of AD, a higher burden of moderate to severe disease, increased rates of allergic comorbidities, greater AD-related impact on health-related quality of life, and more treatment-resistant AD.
“These features can make long-term AD control very difficult,” he said. “Given the variable long-term efficacy and safety of current treatments, health care providers and patients often have to combine therapies, seek new treatments, and consider adjunctive approaches – all of which can contribute to increased costs.”
AD is also associated with a considerable financial burden, he continued, including direct health care costs, lost work productivity and out-of-pocket health care expenses. “Previous population-based studies suggest that there are multifactorial increases in overall out-of-pocket health expenses in AD,” Dr. Chovatiya said. “Black race in particular is thought to be associated with increased health care utilization in AD, but little is known about the out-of-pocket health care expenses.”
To characterize the categories and impact of out-of-pocket health care expenses associated with AD management among Black individuals, he and his colleagues administered a 25-question voluntary survey to 113,502 members of the NEA between Nov. 14 and Dec. 21, 2019. They included adults with a self-reported diagnosis of AD or children, teens, or young adults who had a caregiver responding for them. In all, 1,118 respondents met inclusion criteria. Questions included those about out-of-pocket expenses for AD over the past 30 days and over the past year, as well as the disease impact on household finances.
The cohort included 75% of individuals with AD; 25% were primary caregivers of children, teens, and young adults with AD. More than three-quarters of respondents (77%) were female, 73% were White, 11% were Black, 6% were Asian, and the remainder were from other ethnic backgrounds. More than half of respondents (58%) had employer-sponsored insurance coverage and the median annual household income was between $50,000 and $75,000.
Nearly three-quarters of respondents (74%) classified their AD severity as moderate or severe, and 63% reported minimally controlled or somewhat-controlled AD. Black respondents were significantly more likely to be younger, have lower household incomes, live in an urban setting, use Medicaid or state assistance, have poor disease control, and frequent skin infections (P ≤ .02). “A numerically higher proportion of Black respondents also had increased AD severity and reported the use of step-up therapy with systemic agents, prescription polypharmacy with three or more prescriptions, and a higher monthly out-of-pocket cost,” Dr. Chovatiya said.
Compared with their non-Black counterparts, Black survey respondents reported more out-of-pocket costs for prescription medications covered by insurance (74.2% vs. 63.6%, P = .04), prescription medications not covered by insurance (65.1% vs. 46.5%, P = .0004), ED visits (22.1% vs. 11.8%, P = .005), and outpatient laboratory testing (33.3% vs. 21.8%, P = .01). Black race was associated with increased household financial impact from out-of-pocket expenses (P = .0009), and predictors of financial impact included minimally controlled AD (adjusted odds ratio, 13.88; P = .02), comorbid anxiety and/or depression (aOR, 4.34; P = .01), systemic therapy (aOR, 4.34; P = .003), out-of-pocket costs that exceeded $200 per month (aOR, 14.28; P = .0003), and Medicaid insurance (aOR, 4.02; P = .03). Blacks with Medicaid had higher odds of harmful financial impact (aOR, 3.32; P = .0002) than respondents who were Black (aOR, 1.81; P = .04) or those with Medicaid alone (aOR, 1.39; P = .04).
“I looked at some of the findings from recent studies that have talked about this burden, including an increased prevalence among Black children, a higher likelihood of moderate to severe disease, higher rates of ED visits and hospitalizations, and increased prescription medications,” Dr. Chovatiya said.“Our findings reflect these racial and socioeconomic disparities and provide another piece of evidence for increased financial burden among Black individuals with AD and support the need for targeted strategies to address these inequities.”
The study received funding support from the NEA. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Incyte, and Regeneron/Sanofi-Genzyme.
They also have significantly poorer disease control and an increased rate of comorbid skin infections.
Those are among the key findings from a 25-question survey administered to members of the National Eczema Association.
“Black individuals with AD have a unique sociodemographic and disease profile,” lead study investigator Raj Chovatiya, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium. “Out-of-pocket expenses are just one component of the real-world burden faced by this population.”
According to Dr. Chovatiya, of the department of dermatology at Northwestern University, Chicago, the clinical phenotype and burden of AD can vary across racial and ethnic groups. Black race, for example, is associated with a higher prevalence of AD, a higher burden of moderate to severe disease, increased rates of allergic comorbidities, greater AD-related impact on health-related quality of life, and more treatment-resistant AD.
“These features can make long-term AD control very difficult,” he said. “Given the variable long-term efficacy and safety of current treatments, health care providers and patients often have to combine therapies, seek new treatments, and consider adjunctive approaches – all of which can contribute to increased costs.”
AD is also associated with a considerable financial burden, he continued, including direct health care costs, lost work productivity and out-of-pocket health care expenses. “Previous population-based studies suggest that there are multifactorial increases in overall out-of-pocket health expenses in AD,” Dr. Chovatiya said. “Black race in particular is thought to be associated with increased health care utilization in AD, but little is known about the out-of-pocket health care expenses.”
To characterize the categories and impact of out-of-pocket health care expenses associated with AD management among Black individuals, he and his colleagues administered a 25-question voluntary survey to 113,502 members of the NEA between Nov. 14 and Dec. 21, 2019. They included adults with a self-reported diagnosis of AD or children, teens, or young adults who had a caregiver responding for them. In all, 1,118 respondents met inclusion criteria. Questions included those about out-of-pocket expenses for AD over the past 30 days and over the past year, as well as the disease impact on household finances.
The cohort included 75% of individuals with AD; 25% were primary caregivers of children, teens, and young adults with AD. More than three-quarters of respondents (77%) were female, 73% were White, 11% were Black, 6% were Asian, and the remainder were from other ethnic backgrounds. More than half of respondents (58%) had employer-sponsored insurance coverage and the median annual household income was between $50,000 and $75,000.
Nearly three-quarters of respondents (74%) classified their AD severity as moderate or severe, and 63% reported minimally controlled or somewhat-controlled AD. Black respondents were significantly more likely to be younger, have lower household incomes, live in an urban setting, use Medicaid or state assistance, have poor disease control, and frequent skin infections (P ≤ .02). “A numerically higher proportion of Black respondents also had increased AD severity and reported the use of step-up therapy with systemic agents, prescription polypharmacy with three or more prescriptions, and a higher monthly out-of-pocket cost,” Dr. Chovatiya said.
Compared with their non-Black counterparts, Black survey respondents reported more out-of-pocket costs for prescription medications covered by insurance (74.2% vs. 63.6%, P = .04), prescription medications not covered by insurance (65.1% vs. 46.5%, P = .0004), ED visits (22.1% vs. 11.8%, P = .005), and outpatient laboratory testing (33.3% vs. 21.8%, P = .01). Black race was associated with increased household financial impact from out-of-pocket expenses (P = .0009), and predictors of financial impact included minimally controlled AD (adjusted odds ratio, 13.88; P = .02), comorbid anxiety and/or depression (aOR, 4.34; P = .01), systemic therapy (aOR, 4.34; P = .003), out-of-pocket costs that exceeded $200 per month (aOR, 14.28; P = .0003), and Medicaid insurance (aOR, 4.02; P = .03). Blacks with Medicaid had higher odds of harmful financial impact (aOR, 3.32; P = .0002) than respondents who were Black (aOR, 1.81; P = .04) or those with Medicaid alone (aOR, 1.39; P = .04).
“I looked at some of the findings from recent studies that have talked about this burden, including an increased prevalence among Black children, a higher likelihood of moderate to severe disease, higher rates of ED visits and hospitalizations, and increased prescription medications,” Dr. Chovatiya said.“Our findings reflect these racial and socioeconomic disparities and provide another piece of evidence for increased financial burden among Black individuals with AD and support the need for targeted strategies to address these inequities.”
The study received funding support from the NEA. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Incyte, and Regeneron/Sanofi-Genzyme.
They also have significantly poorer disease control and an increased rate of comorbid skin infections.
Those are among the key findings from a 25-question survey administered to members of the National Eczema Association.
“Black individuals with AD have a unique sociodemographic and disease profile,” lead study investigator Raj Chovatiya, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium. “Out-of-pocket expenses are just one component of the real-world burden faced by this population.”
According to Dr. Chovatiya, of the department of dermatology at Northwestern University, Chicago, the clinical phenotype and burden of AD can vary across racial and ethnic groups. Black race, for example, is associated with a higher prevalence of AD, a higher burden of moderate to severe disease, increased rates of allergic comorbidities, greater AD-related impact on health-related quality of life, and more treatment-resistant AD.
“These features can make long-term AD control very difficult,” he said. “Given the variable long-term efficacy and safety of current treatments, health care providers and patients often have to combine therapies, seek new treatments, and consider adjunctive approaches – all of which can contribute to increased costs.”
AD is also associated with a considerable financial burden, he continued, including direct health care costs, lost work productivity and out-of-pocket health care expenses. “Previous population-based studies suggest that there are multifactorial increases in overall out-of-pocket health expenses in AD,” Dr. Chovatiya said. “Black race in particular is thought to be associated with increased health care utilization in AD, but little is known about the out-of-pocket health care expenses.”
To characterize the categories and impact of out-of-pocket health care expenses associated with AD management among Black individuals, he and his colleagues administered a 25-question voluntary survey to 113,502 members of the NEA between Nov. 14 and Dec. 21, 2019. They included adults with a self-reported diagnosis of AD or children, teens, or young adults who had a caregiver responding for them. In all, 1,118 respondents met inclusion criteria. Questions included those about out-of-pocket expenses for AD over the past 30 days and over the past year, as well as the disease impact on household finances.
The cohort included 75% of individuals with AD; 25% were primary caregivers of children, teens, and young adults with AD. More than three-quarters of respondents (77%) were female, 73% were White, 11% were Black, 6% were Asian, and the remainder were from other ethnic backgrounds. More than half of respondents (58%) had employer-sponsored insurance coverage and the median annual household income was between $50,000 and $75,000.
Nearly three-quarters of respondents (74%) classified their AD severity as moderate or severe, and 63% reported minimally controlled or somewhat-controlled AD. Black respondents were significantly more likely to be younger, have lower household incomes, live in an urban setting, use Medicaid or state assistance, have poor disease control, and frequent skin infections (P ≤ .02). “A numerically higher proportion of Black respondents also had increased AD severity and reported the use of step-up therapy with systemic agents, prescription polypharmacy with three or more prescriptions, and a higher monthly out-of-pocket cost,” Dr. Chovatiya said.
Compared with their non-Black counterparts, Black survey respondents reported more out-of-pocket costs for prescription medications covered by insurance (74.2% vs. 63.6%, P = .04), prescription medications not covered by insurance (65.1% vs. 46.5%, P = .0004), ED visits (22.1% vs. 11.8%, P = .005), and outpatient laboratory testing (33.3% vs. 21.8%, P = .01). Black race was associated with increased household financial impact from out-of-pocket expenses (P = .0009), and predictors of financial impact included minimally controlled AD (adjusted odds ratio, 13.88; P = .02), comorbid anxiety and/or depression (aOR, 4.34; P = .01), systemic therapy (aOR, 4.34; P = .003), out-of-pocket costs that exceeded $200 per month (aOR, 14.28; P = .0003), and Medicaid insurance (aOR, 4.02; P = .03). Blacks with Medicaid had higher odds of harmful financial impact (aOR, 3.32; P = .0002) than respondents who were Black (aOR, 1.81; P = .04) or those with Medicaid alone (aOR, 1.39; P = .04).
“I looked at some of the findings from recent studies that have talked about this burden, including an increased prevalence among Black children, a higher likelihood of moderate to severe disease, higher rates of ED visits and hospitalizations, and increased prescription medications,” Dr. Chovatiya said.“Our findings reflect these racial and socioeconomic disparities and provide another piece of evidence for increased financial burden among Black individuals with AD and support the need for targeted strategies to address these inequities.”
The study received funding support from the NEA. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Incyte, and Regeneron/Sanofi-Genzyme.
FROM REVOLUTIONIZING AD 2021
Scaly beard rash
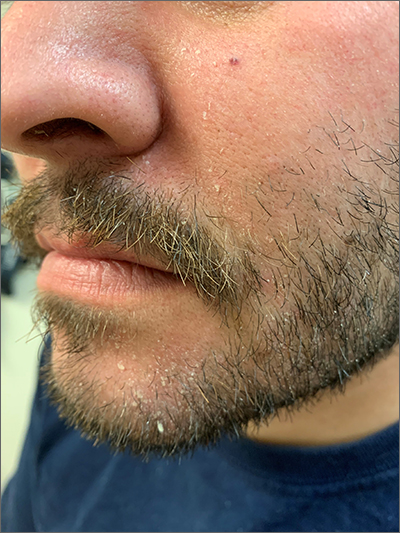
Waxy loose scale with associated erythema on the face and scalp is a classic sign of seborrheic dermatitis (SD).
SD is caused by inflammation related to the presence of Malassezia, which proliferates on sebum-rich areas of skin. Malassezia is normally present on the skin, but some individuals have a heightened sensitivity to it, leading to erythema and scale. It is prudent to examine the scalp, nasolabial folds, and around the ears where it often occurs concomitantly.
There are multiple topical and systemic options which treat the fungal involvement, the subsequent inflammation, and reduce the scale.1 Topical azole antifungals are effective for reducing the amount of Malassezia present. Topical steroids work well to reduce the erythema. Fortunately, low-potency steroids, including hydrocortisone and desonide, are adequate. This is important since SD frequently involves the face and higher-potency steroids can cause skin atrophy or rebound erythema.
Salicylic acid products exfoliate the scale and topical tar products suppress the scale, both leading to clinical improvement. Sunlight and narrow beam UVB light therapy are also effective treatments. As was true with this patient, SD often improves during the summer months (when there is more sunlight) and when patients shave, as this allows for additional sun exposure to the skin.
The patient in this case was told to use ketoconazole shampoo for his scalp, beard, and mustache. He was instructed to use it at least 3 times per week, applying it to the scalp as the first part of his bathing routine and then waiting until the end to rinse it off. This technique maximizes the antifungal shampoo’s contact time on the skin. He was also given a prescription for ketoconazole cream to apply twice daily to the areas of facial erythema and scale. He was counseled that shaving his beard and mustache might help reduce the SD in those areas.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque
Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169. doi: 10.1080/09546634.2018.1473554

Waxy loose scale with associated erythema on the face and scalp is a classic sign of seborrheic dermatitis (SD).
SD is caused by inflammation related to the presence of Malassezia, which proliferates on sebum-rich areas of skin. Malassezia is normally present on the skin, but some individuals have a heightened sensitivity to it, leading to erythema and scale. It is prudent to examine the scalp, nasolabial folds, and around the ears where it often occurs concomitantly.
There are multiple topical and systemic options which treat the fungal involvement, the subsequent inflammation, and reduce the scale.1 Topical azole antifungals are effective for reducing the amount of Malassezia present. Topical steroids work well to reduce the erythema. Fortunately, low-potency steroids, including hydrocortisone and desonide, are adequate. This is important since SD frequently involves the face and higher-potency steroids can cause skin atrophy or rebound erythema.
Salicylic acid products exfoliate the scale and topical tar products suppress the scale, both leading to clinical improvement. Sunlight and narrow beam UVB light therapy are also effective treatments. As was true with this patient, SD often improves during the summer months (when there is more sunlight) and when patients shave, as this allows for additional sun exposure to the skin.
The patient in this case was told to use ketoconazole shampoo for his scalp, beard, and mustache. He was instructed to use it at least 3 times per week, applying it to the scalp as the first part of his bathing routine and then waiting until the end to rinse it off. This technique maximizes the antifungal shampoo’s contact time on the skin. He was also given a prescription for ketoconazole cream to apply twice daily to the areas of facial erythema and scale. He was counseled that shaving his beard and mustache might help reduce the SD in those areas.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque

Waxy loose scale with associated erythema on the face and scalp is a classic sign of seborrheic dermatitis (SD).
SD is caused by inflammation related to the presence of Malassezia, which proliferates on sebum-rich areas of skin. Malassezia is normally present on the skin, but some individuals have a heightened sensitivity to it, leading to erythema and scale. It is prudent to examine the scalp, nasolabial folds, and around the ears where it often occurs concomitantly.
There are multiple topical and systemic options which treat the fungal involvement, the subsequent inflammation, and reduce the scale.1 Topical azole antifungals are effective for reducing the amount of Malassezia present. Topical steroids work well to reduce the erythema. Fortunately, low-potency steroids, including hydrocortisone and desonide, are adequate. This is important since SD frequently involves the face and higher-potency steroids can cause skin atrophy or rebound erythema.
Salicylic acid products exfoliate the scale and topical tar products suppress the scale, both leading to clinical improvement. Sunlight and narrow beam UVB light therapy are also effective treatments. As was true with this patient, SD often improves during the summer months (when there is more sunlight) and when patients shave, as this allows for additional sun exposure to the skin.
The patient in this case was told to use ketoconazole shampoo for his scalp, beard, and mustache. He was instructed to use it at least 3 times per week, applying it to the scalp as the first part of his bathing routine and then waiting until the end to rinse it off. This technique maximizes the antifungal shampoo’s contact time on the skin. He was also given a prescription for ketoconazole cream to apply twice daily to the areas of facial erythema and scale. He was counseled that shaving his beard and mustache might help reduce the SD in those areas.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque
Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169. doi: 10.1080/09546634.2018.1473554
Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169. doi: 10.1080/09546634.2018.1473554
Allergic conjunctivitis severely affects children’s quality of life
Allergic conjunctivitis harms quality of life for children and their parents, apparently causing greater day-to-day worries than potentially blinding diseases, researchers report.
Parents worry especially that treatments might not be effective, according to Shi-yao Zhang, MD, and colleagues from Sun Yat-sen University, Guangzhou, China. “This finding suggests that more communication with parents regarding treatment and prognosis is needed,” they write in an article published online June 10 in JAMA Ophthalmology.
One of the most prevalent eye disorders in children, allergic conjunctivitis is often chronic, leading patients to ask repeatedly for help from physicians. It can take an emotional toll and can cause children to miss school.
“With any sign of a slightly pink eye [or a] runny nose, which are very common with allergies, children are being sent home, because everyone’s concerned about COVID,” said Yi Ning J. Strube, MD, an associate professor of ophthalmology and pediatrics at Queen’s University, Kingston, Canada, whose commentary appears in the same issue of JAMA Ophthalmology.
Adolescents are also sometimes accused of smoking cannabis because of their red eyes, she said.
However, little research has examined the effects of allergic conjunctivitis on the quality of life of children and their guardians, Dr. Zhang and colleagues write. To fill that gap, the researchers administered the Pediatric Quality of Life Inventory (PedsQL) to 92 children with allergic conjunctivitis and their parents. The children were aged 5 to 18 years.
The researchers administered the same questionnaire to 96 healthy children of the same ages, along with their parents. These participants served as a control group.
On a scale of 0 to 100, in which a higher score signifies a better quality of life, the median total PedsQL score was 69.6 for children with allergic conjunctivitis versus 96.7 for the control group.
Subscores of physical, emotional, social, and especially school functioning were all significantly lower for the children with allergic conjunctivitis than for the control persons. “Because children generally spend most of their time in the school environment, this outcome raises an issue regarding whether children have a poorer performance in their education,” Dr. Zhang and colleagues write.
Dr. Strube recommends that physicians educate their patients about allergic conjunctivitis using handouts or high-quality websites. She often refers patients and their families to the allergic conjunctivitis webpage of the American Academy of Pediatric Ophthalmology and Strabismus.
She tells parents to have their child “take a shower and wash their hair when they get home before they rub their pollen-filled hair on their pillowcase and make their allergy symptoms worse.”
Parents and schools should try to filter pollen and other allergens from indoor air, she added.
Parents of the children with allergic conjunctivitis in the study also reported lower quality of life; they scored 68.8, versus 96.5 for parents of children in the control group. The differences for both parents and children were statistically significant (P < .001). Overall, the parents’ quality-of-life scores correlated with their children’s (correlation coefficient, r = 0.59; P < .001).
Children with vernal or atopic keratoconjunctivitis scored 3.3 points lower on health-related quality of life than those with seasonal allergic conjunctivitis.
Children with higher corneal fluorescein staining scores also had lower quality-of-life scores. Parents whose children had higher corneal fluorescein staining scores and also those who had multiple consultations with health care practitioners also reported lower quality of life.
The quality-of-life scores of the children with allergic conjunctivitis were lower than scores in previous studies for children with vision-threatening diseases, such as glaucoma and congenital cataract. This may be because glaucoma and cataracts do not typically cause discomfort even if they impair the patient’s vision, said Dr. Strube.
She pointed out one potential flaw in the study: In the cohort with allergic conjunctivitis, 83.7% were boys, compared to 42.7% of the control group. Vernal keratoconjunctivitis affects more boys than girls, and not controlling for this factor could have confounded the data, Dr. Strube said.
It could also be useful to replicate the study in other countries to see whether geographic or cultural factors affected the results, she said. “A lot of these big centers around the world, including in China, have poor air quality, so that may be contributing to patients’ symptoms,” she said. “With regards to reported health quality of life and impact on education, results from different parts of the world may be different, due to parenting styles and education styles,” she said.
The study was supported by the National Natural Science Foundation of China and the Science Foundation of Guangdong Province. Dr. Zhang and colleagues reported no relevant financial relationships. Dr. Strube reported receiving personal fees from Santen Canada Advisory Board Consultant outside the submitted work.
A version of this article first appeared on Medscape.com.
Allergic conjunctivitis harms quality of life for children and their parents, apparently causing greater day-to-day worries than potentially blinding diseases, researchers report.
Parents worry especially that treatments might not be effective, according to Shi-yao Zhang, MD, and colleagues from Sun Yat-sen University, Guangzhou, China. “This finding suggests that more communication with parents regarding treatment and prognosis is needed,” they write in an article published online June 10 in JAMA Ophthalmology.
One of the most prevalent eye disorders in children, allergic conjunctivitis is often chronic, leading patients to ask repeatedly for help from physicians. It can take an emotional toll and can cause children to miss school.
“With any sign of a slightly pink eye [or a] runny nose, which are very common with allergies, children are being sent home, because everyone’s concerned about COVID,” said Yi Ning J. Strube, MD, an associate professor of ophthalmology and pediatrics at Queen’s University, Kingston, Canada, whose commentary appears in the same issue of JAMA Ophthalmology.
Adolescents are also sometimes accused of smoking cannabis because of their red eyes, she said.
However, little research has examined the effects of allergic conjunctivitis on the quality of life of children and their guardians, Dr. Zhang and colleagues write. To fill that gap, the researchers administered the Pediatric Quality of Life Inventory (PedsQL) to 92 children with allergic conjunctivitis and their parents. The children were aged 5 to 18 years.
The researchers administered the same questionnaire to 96 healthy children of the same ages, along with their parents. These participants served as a control group.
On a scale of 0 to 100, in which a higher score signifies a better quality of life, the median total PedsQL score was 69.6 for children with allergic conjunctivitis versus 96.7 for the control group.
Subscores of physical, emotional, social, and especially school functioning were all significantly lower for the children with allergic conjunctivitis than for the control persons. “Because children generally spend most of their time in the school environment, this outcome raises an issue regarding whether children have a poorer performance in their education,” Dr. Zhang and colleagues write.
Dr. Strube recommends that physicians educate their patients about allergic conjunctivitis using handouts or high-quality websites. She often refers patients and their families to the allergic conjunctivitis webpage of the American Academy of Pediatric Ophthalmology and Strabismus.
She tells parents to have their child “take a shower and wash their hair when they get home before they rub their pollen-filled hair on their pillowcase and make their allergy symptoms worse.”
Parents and schools should try to filter pollen and other allergens from indoor air, she added.
Parents of the children with allergic conjunctivitis in the study also reported lower quality of life; they scored 68.8, versus 96.5 for parents of children in the control group. The differences for both parents and children were statistically significant (P < .001). Overall, the parents’ quality-of-life scores correlated with their children’s (correlation coefficient, r = 0.59; P < .001).
Children with vernal or atopic keratoconjunctivitis scored 3.3 points lower on health-related quality of life than those with seasonal allergic conjunctivitis.
Children with higher corneal fluorescein staining scores also had lower quality-of-life scores. Parents whose children had higher corneal fluorescein staining scores and also those who had multiple consultations with health care practitioners also reported lower quality of life.
The quality-of-life scores of the children with allergic conjunctivitis were lower than scores in previous studies for children with vision-threatening diseases, such as glaucoma and congenital cataract. This may be because glaucoma and cataracts do not typically cause discomfort even if they impair the patient’s vision, said Dr. Strube.
She pointed out one potential flaw in the study: In the cohort with allergic conjunctivitis, 83.7% were boys, compared to 42.7% of the control group. Vernal keratoconjunctivitis affects more boys than girls, and not controlling for this factor could have confounded the data, Dr. Strube said.
It could also be useful to replicate the study in other countries to see whether geographic or cultural factors affected the results, she said. “A lot of these big centers around the world, including in China, have poor air quality, so that may be contributing to patients’ symptoms,” she said. “With regards to reported health quality of life and impact on education, results from different parts of the world may be different, due to parenting styles and education styles,” she said.
The study was supported by the National Natural Science Foundation of China and the Science Foundation of Guangdong Province. Dr. Zhang and colleagues reported no relevant financial relationships. Dr. Strube reported receiving personal fees from Santen Canada Advisory Board Consultant outside the submitted work.
A version of this article first appeared on Medscape.com.
Allergic conjunctivitis harms quality of life for children and their parents, apparently causing greater day-to-day worries than potentially blinding diseases, researchers report.
Parents worry especially that treatments might not be effective, according to Shi-yao Zhang, MD, and colleagues from Sun Yat-sen University, Guangzhou, China. “This finding suggests that more communication with parents regarding treatment and prognosis is needed,” they write in an article published online June 10 in JAMA Ophthalmology.
One of the most prevalent eye disorders in children, allergic conjunctivitis is often chronic, leading patients to ask repeatedly for help from physicians. It can take an emotional toll and can cause children to miss school.
“With any sign of a slightly pink eye [or a] runny nose, which are very common with allergies, children are being sent home, because everyone’s concerned about COVID,” said Yi Ning J. Strube, MD, an associate professor of ophthalmology and pediatrics at Queen’s University, Kingston, Canada, whose commentary appears in the same issue of JAMA Ophthalmology.
Adolescents are also sometimes accused of smoking cannabis because of their red eyes, she said.
However, little research has examined the effects of allergic conjunctivitis on the quality of life of children and their guardians, Dr. Zhang and colleagues write. To fill that gap, the researchers administered the Pediatric Quality of Life Inventory (PedsQL) to 92 children with allergic conjunctivitis and their parents. The children were aged 5 to 18 years.
The researchers administered the same questionnaire to 96 healthy children of the same ages, along with their parents. These participants served as a control group.
On a scale of 0 to 100, in which a higher score signifies a better quality of life, the median total PedsQL score was 69.6 for children with allergic conjunctivitis versus 96.7 for the control group.
Subscores of physical, emotional, social, and especially school functioning were all significantly lower for the children with allergic conjunctivitis than for the control persons. “Because children generally spend most of their time in the school environment, this outcome raises an issue regarding whether children have a poorer performance in their education,” Dr. Zhang and colleagues write.
Dr. Strube recommends that physicians educate their patients about allergic conjunctivitis using handouts or high-quality websites. She often refers patients and their families to the allergic conjunctivitis webpage of the American Academy of Pediatric Ophthalmology and Strabismus.
She tells parents to have their child “take a shower and wash their hair when they get home before they rub their pollen-filled hair on their pillowcase and make their allergy symptoms worse.”
Parents and schools should try to filter pollen and other allergens from indoor air, she added.
Parents of the children with allergic conjunctivitis in the study also reported lower quality of life; they scored 68.8, versus 96.5 for parents of children in the control group. The differences for both parents and children were statistically significant (P < .001). Overall, the parents’ quality-of-life scores correlated with their children’s (correlation coefficient, r = 0.59; P < .001).
Children with vernal or atopic keratoconjunctivitis scored 3.3 points lower on health-related quality of life than those with seasonal allergic conjunctivitis.
Children with higher corneal fluorescein staining scores also had lower quality-of-life scores. Parents whose children had higher corneal fluorescein staining scores and also those who had multiple consultations with health care practitioners also reported lower quality of life.
The quality-of-life scores of the children with allergic conjunctivitis were lower than scores in previous studies for children with vision-threatening diseases, such as glaucoma and congenital cataract. This may be because glaucoma and cataracts do not typically cause discomfort even if they impair the patient’s vision, said Dr. Strube.
She pointed out one potential flaw in the study: In the cohort with allergic conjunctivitis, 83.7% were boys, compared to 42.7% of the control group. Vernal keratoconjunctivitis affects more boys than girls, and not controlling for this factor could have confounded the data, Dr. Strube said.
It could also be useful to replicate the study in other countries to see whether geographic or cultural factors affected the results, she said. “A lot of these big centers around the world, including in China, have poor air quality, so that may be contributing to patients’ symptoms,” she said. “With regards to reported health quality of life and impact on education, results from different parts of the world may be different, due to parenting styles and education styles,” she said.
The study was supported by the National Natural Science Foundation of China and the Science Foundation of Guangdong Province. Dr. Zhang and colleagues reported no relevant financial relationships. Dr. Strube reported receiving personal fees from Santen Canada Advisory Board Consultant outside the submitted work.
A version of this article first appeared on Medscape.com.