User login
Which AK treatment has the best long-term efficacy? A study reviews the data
The four results from a systemic review and meta-analysis suggest.
To date, many studies have reported that “most interventions are superior to placebo in terms of lesion clearance and improving the cosmetic image,” corresponding author Markus V. Heppt, MD, MSc, and colleagues wrote in a study published online Aug. 4, 2021, in JAMA Dermatology.
“However, most randomized clinical trials (RCTs) and meta-analyses focused on short-term outcomes that are evaluated within 3-6 months after treatment, although AK is increasingly being considered a chronic condition and reducing the incidence of cSCC [cutaneous squamous cell carcinoma] should be the ultimate goal of treatment,” they said. In addition, most treatments have been compared with placebo “and head-to-head comparisons are widely lacking, limiting the possibility to cross compare distinct active treatments. To this end, no evidence-based recommendation regarding the long-term efficacy of interventions for AK exists.”
To determine the long-term clearance rates of treatments used in adults with AK, a precursor of cSCC, Dr. Heppt, of the department of dermatology at University Hospital Erlangen (Germany), and colleagues drew from 15 randomized clinical trials that reported sustained clearance rates after at least 12 months of treatment and were published up to April 6, 2020. They conducted the review by following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guideline and its extension for network meta-analyses (PRIMSA-NMA) and using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) process to determine the certainty of the evidence for network meta-analyses.
The study population included 4,252 patients. Among 10 studies included in a network meta-analysis for the outcome of participant complete clearance, ALA-PDT showed the most favorable risk ratio profile, compared with placebo (RR, 8.06; moderate-quality evidence on GRADE), followed by imiquimod, 5% (RR, 5.98; very-low-quality evidence on GRADE); MAL-PDT (RR, 5.95; low-quality evidence on GRADE); and cryosurgery (RR, 4.76; very-low-quality evidence on GRADE).
ALA-PDT had the highest RR in the network meta-analyses for lesion-specific clearance (RR, 5.08; moderate-quality evidence on GRADE).
“Although ALA-PDT showed the most favorable RR and was ranked best among all interventions, the relative efficacy values and treatment rankings must be interpreted with caution,” because of the low certainty of evidence and few direct, head-to-head comparisons, the authors emphasized. “In particular, it remains elusive how to translate the distinct RR values into clinical relevance. We are hesitant to derive hierarchical or algorithmic treatment recommendations from our results.”
“The current meta-analysis notes that there are conflicting results in different studies,” said Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn. who was asked to comment on the study. “Sustained participant complete clearance of actinic keratoses at 12 months is used as an outcome measure, although the authors comment that prevention/reduction of squamous cell carcinoma might be the more valid outcome measure.”
In her clinical experience, Dr. Ko said that patients often have good, sustained clearance of AKs with field treatment using a topical medication like 5-fluorouracil. “Patients can also have a good result with photodynamic therapy,” she said. “The paper’s results therefore do reflect what I have seen in my own practice. I also agree with the authors that, while it is difficult to measure, a meaningful outcome for patients is reduction/prevention of squamous cell carcinoma. It would be useful to have data on which treatment of actinic keratosis is best to reduce/prevent squamous cell carcinoma.”
The authors acknowledged limitations of the study, including the fact that field-directed treatments such as imiquimod, PDT, and fluorouracil were compared with lesion-directed approaches such as cryosurgery, “which may limit the generalizability of our results.” They concluded that their analysis “provides data that might contribute to an evidence-based framework to guide the selection of interventions for AK with proven long-term efficacy and sustained AK clearance.”
The analysis did not include data on tirbanibulin, a first-in-class dual Src kinase and tubulin polymerization inhibitor that was approved by the FDA for the topical treatment of AKs on the face or scalp in December 2020.
Dr. Heppt disclosed that he has been a member of the advisory boards of Almirall Hermal and Sanofi-Aventis and has received speaker’s honoraria from Galderma and Biofrontera. Many of his coauthors also reported having relevant financial disclosures. Dr. Ko reported having no relevant disclosures.
The four results from a systemic review and meta-analysis suggest.
To date, many studies have reported that “most interventions are superior to placebo in terms of lesion clearance and improving the cosmetic image,” corresponding author Markus V. Heppt, MD, MSc, and colleagues wrote in a study published online Aug. 4, 2021, in JAMA Dermatology.
“However, most randomized clinical trials (RCTs) and meta-analyses focused on short-term outcomes that are evaluated within 3-6 months after treatment, although AK is increasingly being considered a chronic condition and reducing the incidence of cSCC [cutaneous squamous cell carcinoma] should be the ultimate goal of treatment,” they said. In addition, most treatments have been compared with placebo “and head-to-head comparisons are widely lacking, limiting the possibility to cross compare distinct active treatments. To this end, no evidence-based recommendation regarding the long-term efficacy of interventions for AK exists.”
To determine the long-term clearance rates of treatments used in adults with AK, a precursor of cSCC, Dr. Heppt, of the department of dermatology at University Hospital Erlangen (Germany), and colleagues drew from 15 randomized clinical trials that reported sustained clearance rates after at least 12 months of treatment and were published up to April 6, 2020. They conducted the review by following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guideline and its extension for network meta-analyses (PRIMSA-NMA) and using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) process to determine the certainty of the evidence for network meta-analyses.
The study population included 4,252 patients. Among 10 studies included in a network meta-analysis for the outcome of participant complete clearance, ALA-PDT showed the most favorable risk ratio profile, compared with placebo (RR, 8.06; moderate-quality evidence on GRADE), followed by imiquimod, 5% (RR, 5.98; very-low-quality evidence on GRADE); MAL-PDT (RR, 5.95; low-quality evidence on GRADE); and cryosurgery (RR, 4.76; very-low-quality evidence on GRADE).
ALA-PDT had the highest RR in the network meta-analyses for lesion-specific clearance (RR, 5.08; moderate-quality evidence on GRADE).
“Although ALA-PDT showed the most favorable RR and was ranked best among all interventions, the relative efficacy values and treatment rankings must be interpreted with caution,” because of the low certainty of evidence and few direct, head-to-head comparisons, the authors emphasized. “In particular, it remains elusive how to translate the distinct RR values into clinical relevance. We are hesitant to derive hierarchical or algorithmic treatment recommendations from our results.”
“The current meta-analysis notes that there are conflicting results in different studies,” said Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn. who was asked to comment on the study. “Sustained participant complete clearance of actinic keratoses at 12 months is used as an outcome measure, although the authors comment that prevention/reduction of squamous cell carcinoma might be the more valid outcome measure.”
In her clinical experience, Dr. Ko said that patients often have good, sustained clearance of AKs with field treatment using a topical medication like 5-fluorouracil. “Patients can also have a good result with photodynamic therapy,” she said. “The paper’s results therefore do reflect what I have seen in my own practice. I also agree with the authors that, while it is difficult to measure, a meaningful outcome for patients is reduction/prevention of squamous cell carcinoma. It would be useful to have data on which treatment of actinic keratosis is best to reduce/prevent squamous cell carcinoma.”
The authors acknowledged limitations of the study, including the fact that field-directed treatments such as imiquimod, PDT, and fluorouracil were compared with lesion-directed approaches such as cryosurgery, “which may limit the generalizability of our results.” They concluded that their analysis “provides data that might contribute to an evidence-based framework to guide the selection of interventions for AK with proven long-term efficacy and sustained AK clearance.”
The analysis did not include data on tirbanibulin, a first-in-class dual Src kinase and tubulin polymerization inhibitor that was approved by the FDA for the topical treatment of AKs on the face or scalp in December 2020.
Dr. Heppt disclosed that he has been a member of the advisory boards of Almirall Hermal and Sanofi-Aventis and has received speaker’s honoraria from Galderma and Biofrontera. Many of his coauthors also reported having relevant financial disclosures. Dr. Ko reported having no relevant disclosures.
The four results from a systemic review and meta-analysis suggest.
To date, many studies have reported that “most interventions are superior to placebo in terms of lesion clearance and improving the cosmetic image,” corresponding author Markus V. Heppt, MD, MSc, and colleagues wrote in a study published online Aug. 4, 2021, in JAMA Dermatology.
“However, most randomized clinical trials (RCTs) and meta-analyses focused on short-term outcomes that are evaluated within 3-6 months after treatment, although AK is increasingly being considered a chronic condition and reducing the incidence of cSCC [cutaneous squamous cell carcinoma] should be the ultimate goal of treatment,” they said. In addition, most treatments have been compared with placebo “and head-to-head comparisons are widely lacking, limiting the possibility to cross compare distinct active treatments. To this end, no evidence-based recommendation regarding the long-term efficacy of interventions for AK exists.”
To determine the long-term clearance rates of treatments used in adults with AK, a precursor of cSCC, Dr. Heppt, of the department of dermatology at University Hospital Erlangen (Germany), and colleagues drew from 15 randomized clinical trials that reported sustained clearance rates after at least 12 months of treatment and were published up to April 6, 2020. They conducted the review by following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guideline and its extension for network meta-analyses (PRIMSA-NMA) and using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) process to determine the certainty of the evidence for network meta-analyses.
The study population included 4,252 patients. Among 10 studies included in a network meta-analysis for the outcome of participant complete clearance, ALA-PDT showed the most favorable risk ratio profile, compared with placebo (RR, 8.06; moderate-quality evidence on GRADE), followed by imiquimod, 5% (RR, 5.98; very-low-quality evidence on GRADE); MAL-PDT (RR, 5.95; low-quality evidence on GRADE); and cryosurgery (RR, 4.76; very-low-quality evidence on GRADE).
ALA-PDT had the highest RR in the network meta-analyses for lesion-specific clearance (RR, 5.08; moderate-quality evidence on GRADE).
“Although ALA-PDT showed the most favorable RR and was ranked best among all interventions, the relative efficacy values and treatment rankings must be interpreted with caution,” because of the low certainty of evidence and few direct, head-to-head comparisons, the authors emphasized. “In particular, it remains elusive how to translate the distinct RR values into clinical relevance. We are hesitant to derive hierarchical or algorithmic treatment recommendations from our results.”
“The current meta-analysis notes that there are conflicting results in different studies,” said Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn. who was asked to comment on the study. “Sustained participant complete clearance of actinic keratoses at 12 months is used as an outcome measure, although the authors comment that prevention/reduction of squamous cell carcinoma might be the more valid outcome measure.”
In her clinical experience, Dr. Ko said that patients often have good, sustained clearance of AKs with field treatment using a topical medication like 5-fluorouracil. “Patients can also have a good result with photodynamic therapy,” she said. “The paper’s results therefore do reflect what I have seen in my own practice. I also agree with the authors that, while it is difficult to measure, a meaningful outcome for patients is reduction/prevention of squamous cell carcinoma. It would be useful to have data on which treatment of actinic keratosis is best to reduce/prevent squamous cell carcinoma.”
The authors acknowledged limitations of the study, including the fact that field-directed treatments such as imiquimod, PDT, and fluorouracil were compared with lesion-directed approaches such as cryosurgery, “which may limit the generalizability of our results.” They concluded that their analysis “provides data that might contribute to an evidence-based framework to guide the selection of interventions for AK with proven long-term efficacy and sustained AK clearance.”
The analysis did not include data on tirbanibulin, a first-in-class dual Src kinase and tubulin polymerization inhibitor that was approved by the FDA for the topical treatment of AKs on the face or scalp in December 2020.
Dr. Heppt disclosed that he has been a member of the advisory boards of Almirall Hermal and Sanofi-Aventis and has received speaker’s honoraria from Galderma and Biofrontera. Many of his coauthors also reported having relevant financial disclosures. Dr. Ko reported having no relevant disclosures.
FROM JAMA DERMATOLOGY
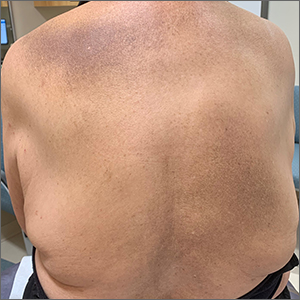
Multiple pigmented patches
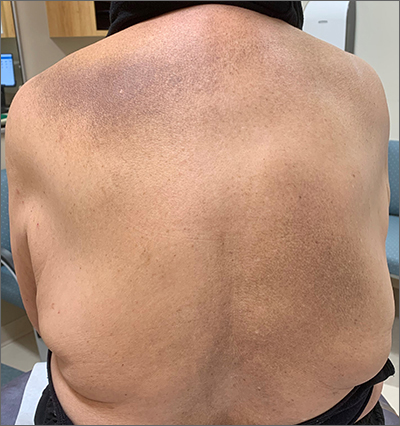
This patient was given a diagnosis of erythema dyschromicum perstans (EDP), also known as ashy dermatosis because of the hyperpigmented macules that come together into confluent patches that look like burned wood. There is often an inflammatory erythematous aspect to EDP.
The etiology of EDP is unknown. It is not related to sun exposure and occurs most commonly on the trunk. Although there are case reports implicating medications or infections, no clear connection has been found. This patient’s chemotherapy may have been an inciting factor, based on her history, but it is not likely that cancer caused the EDP.
EDP tends to be chronic and difficult to treat. Fortunately, other than the itching and skin discoloration, it is usually asymptomatic and benign. Large-scale trials are lacking, but there are case reports showing benefit from narrow beam UVB treatments and topical tacrolimus.1 Laser has not proven very helpful, and the hyperpigmentation can recur.
Based on the clinical appearance of this patient’s lesion, and the fact that a previous biopsy in the same location was consistent with her diagnosis, no further testing was performed. The patient was advised to apply topical diphenhydramine to her back 4 times daily for a 2-week trial. If the diphenhydramine failed to provide relief, the next step in her treatment would have been topical tacrolimus.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Leung N, Oliveira M, Selim MA, et al. Erythema dyschromicum perstans: a case report and systematic review of histologic presentation and treatment. Int J Womens Dermatol. 2018;4:216-222. doi: 10.1016/j.ijwd.2018.08.003

This patient was given a diagnosis of erythema dyschromicum perstans (EDP), also known as ashy dermatosis because of the hyperpigmented macules that come together into confluent patches that look like burned wood. There is often an inflammatory erythematous aspect to EDP.
The etiology of EDP is unknown. It is not related to sun exposure and occurs most commonly on the trunk. Although there are case reports implicating medications or infections, no clear connection has been found. This patient’s chemotherapy may have been an inciting factor, based on her history, but it is not likely that cancer caused the EDP.
EDP tends to be chronic and difficult to treat. Fortunately, other than the itching and skin discoloration, it is usually asymptomatic and benign. Large-scale trials are lacking, but there are case reports showing benefit from narrow beam UVB treatments and topical tacrolimus.1 Laser has not proven very helpful, and the hyperpigmentation can recur.
Based on the clinical appearance of this patient’s lesion, and the fact that a previous biopsy in the same location was consistent with her diagnosis, no further testing was performed. The patient was advised to apply topical diphenhydramine to her back 4 times daily for a 2-week trial. If the diphenhydramine failed to provide relief, the next step in her treatment would have been topical tacrolimus.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This patient was given a diagnosis of erythema dyschromicum perstans (EDP), also known as ashy dermatosis because of the hyperpigmented macules that come together into confluent patches that look like burned wood. There is often an inflammatory erythematous aspect to EDP.
The etiology of EDP is unknown. It is not related to sun exposure and occurs most commonly on the trunk. Although there are case reports implicating medications or infections, no clear connection has been found. This patient’s chemotherapy may have been an inciting factor, based on her history, but it is not likely that cancer caused the EDP.
EDP tends to be chronic and difficult to treat. Fortunately, other than the itching and skin discoloration, it is usually asymptomatic and benign. Large-scale trials are lacking, but there are case reports showing benefit from narrow beam UVB treatments and topical tacrolimus.1 Laser has not proven very helpful, and the hyperpigmentation can recur.
Based on the clinical appearance of this patient’s lesion, and the fact that a previous biopsy in the same location was consistent with her diagnosis, no further testing was performed. The patient was advised to apply topical diphenhydramine to her back 4 times daily for a 2-week trial. If the diphenhydramine failed to provide relief, the next step in her treatment would have been topical tacrolimus.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Leung N, Oliveira M, Selim MA, et al. Erythema dyschromicum perstans: a case report and systematic review of histologic presentation and treatment. Int J Womens Dermatol. 2018;4:216-222. doi: 10.1016/j.ijwd.2018.08.003
1. Leung N, Oliveira M, Selim MA, et al. Erythema dyschromicum perstans: a case report and systematic review of histologic presentation and treatment. Int J Womens Dermatol. 2018;4:216-222. doi: 10.1016/j.ijwd.2018.08.003
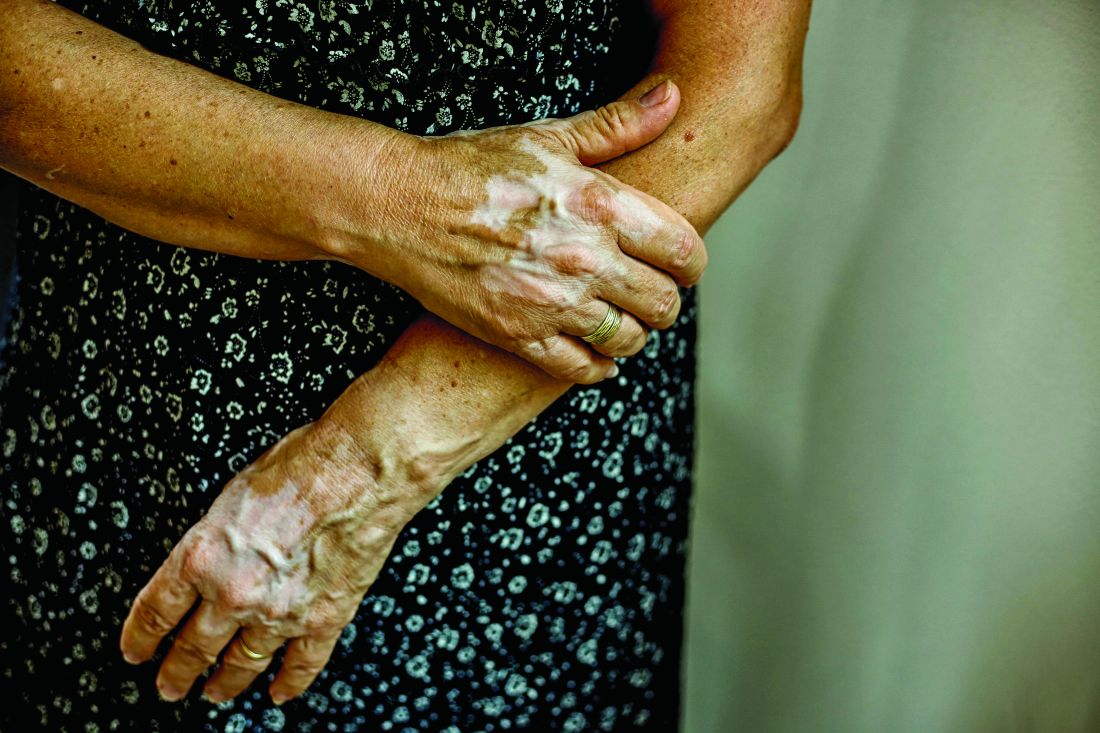
Insurance coverage for vitiligo varies widely in the U.S., analysis finds
, which may disproportionately affect patients of color.
Those are the conclusions from an analysis of vitiligo treatment coverage policies across major health insurers in the United States.
“Vitiligo can be less noticeable in patients with lighter skin types, becoming apparent only when affected patches fail to tan,” first authors Andrew Blundell, MD, MSc, and Moniyka Sachar, MD, wrote in a study published online on July 16 in Pediatric Dermatology. However, they pointed out that, in patients with darker skin types, “vitiligo can be far more evident due to the stark contrast of involved versus uninvolved skin, and as such can lead to a significant impact on quality of life, as well as heightened stigmatization.”
Nevertheless, they noted many health care insurers consider vitiligo as a cosmetic condition, and do not cover treatments, and for the 1%-2% of the general population with vitiligo, “this lack of recognition from health care insurers makes treatments both less accessible and affordable, and only further marginalizes patients with this condition.”
Dr. Blundell, of San Juan Bautista School of Medicine, Caguas, P.R., and Dr. Sachar, of the department of dermatology at Brown University, Providence, R.I., and colleagues surveyed 15 commercial health care insurers, 50 BlueCross BlueShield plans, Medicare, Medicaid, and Veterans Affairs to determine the level of treatment coverage for vitiligo. They looked at office visits, medications (the topical calcineurin inhibitors [TCIs] pimecrolimus, and tacrolimus), excimer laser therapy, and phototherapy (psoralen with UVA [PUVA] and narrow-band UVB [nbUVB]). They collected information from medical policies available online or by direct contact with the plans in 2018.
The researchers reported data from 17 organizations with regional or national coverage policies for vitiligo treatment and two others – BlueCross BlueShield and Medicaid – which had policies that differed by state and plan. Of the 17 organizations, only 12% did not cover TCIs, 56% did not cover nbUVB phototherapy, 53% did not cover PUVA phototherapy, and 41% did not cover laser therapy.
As for BlueCross BlueShield, the health plan did not cover pimecrolimus and tacrolimus in 39% and 35% of states, respectively. At the same time, NbUVB and PUVA therapy were not covered in 20% and 10% of states, respectively, while excimer laser therapy was not covered in 82% of states.
Of accessible Medicaid information from 32 states, 11 did not cover topicals, 5 did not cover nbUVB, 4 did not cover PUVA, and 7 did not cover laser therapy. “The two most commonly cited reasons for denial of coverage were (a) vitiligo is considered a cosmetic condition and (b) certain therapies are not FDA-approved for vitiligo, though they may be approved for other skin conditions,” the study authors wrote.
While the analysis revealed that topical TCI therapy is more widely covered by insurance companies, compared with phototherapy, “multiple studies have shown that a combination of both topical and phototherapy is more effective in treating vitiligo than either alone,” they noted. “Vitiligo treatments can delay the progression of the disease and result in better outcomes when started early, furthering the need for insurance coverage of these treatments. If all proven and accepted vitiligo treatments were covered by their health insurers, patients would have better access, as well as timely and affordable ways by which to limit depigmentation and to repigment affected areas.”
In addition, lack of access to treatments “may increase health disparities among already-marginalized groups, such as children and adults of darker skin phototypes,” they wrote.
Seemal R. Desai, MD, who was asked to comment on the study, said that the findings resonate with him based on his clinical experience as a dermatologist at the University of Texas Southwestern Medical Center in Dallas and in clinical practice. “Vitiligo has a high psychological impact, continues to increase in its prevalence, and has been shown to be an autoimmune, chronic, inflammatory skin disease, yet we’re still having challenges with treatment,” said Dr. Desai, who is also a member of the board of directors for the American Academy of Dermatology and the Global Vitiligo Foundation (GVF).
He said that he is working with the AAD, the GVF, and other stakeholders to improve treatment coverage. For example, in Massachusetts, the Tufts Health Plan had stopped covering treatment for vitiligo. “Through a series of advocacy efforts, that was reversed a couple of years ago,” said Dr. Desai, who is also a past president of the Skin of Color Society. “We also have seen isolated reports of Medicaid and Medicare coverage where local contractors aren’t following national Centers for Medicare and Medicaid Service directive guidance. The challenge becomes, how do you get consistency in treatment coverage, and how do you make sure patients continue to get access to treatment?”
Turning the tide will require “a concerted effort” by dermatologists to engage with the payers, he added. “I’ve had to get on the phone with countless insurance companies on behalf of my patients and make them understand the comorbidities associated with vitiligo, sending them copies of studies that show it’s an autoimmune disease linked to thyroid issues,” Dr. Desai continued. “We talk a lot about the psychological burden and quality of life. There’s still a lot of work to be done in this sphere, but I think we’re making progress.”
With hopes that Janus kinase (JAK) inhibitors and other new products being investigated will soon be approved as a treatment option for vitiligo, Dr. Desai said that now is the time to standardize coverage for patients. “It’s important that we start talking about insurance coverage and denial issues now and get ahead of it, so that when we get those JAK inhibitors available, we don’t fight coverage decisions then.”
The researchers acknowledged certain limitations of the study, including the fact that it was based on insurance coverage from 2017 to 2018 and the lack of easily available state Medicaid policies.
The study coauthors were Colleen K. Gabel, MD, of the University of Massachusetts, Worcester, and Lionel G. Bercovitch, MD, of Brown University. None of the study authors reported financial disclosures.
Dr. Desai disclosed that he has conducted vitiligo research trials and has done consulting work for several pharmaceutical companies.
, which may disproportionately affect patients of color.
Those are the conclusions from an analysis of vitiligo treatment coverage policies across major health insurers in the United States.
“Vitiligo can be less noticeable in patients with lighter skin types, becoming apparent only when affected patches fail to tan,” first authors Andrew Blundell, MD, MSc, and Moniyka Sachar, MD, wrote in a study published online on July 16 in Pediatric Dermatology. However, they pointed out that, in patients with darker skin types, “vitiligo can be far more evident due to the stark contrast of involved versus uninvolved skin, and as such can lead to a significant impact on quality of life, as well as heightened stigmatization.”
Nevertheless, they noted many health care insurers consider vitiligo as a cosmetic condition, and do not cover treatments, and for the 1%-2% of the general population with vitiligo, “this lack of recognition from health care insurers makes treatments both less accessible and affordable, and only further marginalizes patients with this condition.”
Dr. Blundell, of San Juan Bautista School of Medicine, Caguas, P.R., and Dr. Sachar, of the department of dermatology at Brown University, Providence, R.I., and colleagues surveyed 15 commercial health care insurers, 50 BlueCross BlueShield plans, Medicare, Medicaid, and Veterans Affairs to determine the level of treatment coverage for vitiligo. They looked at office visits, medications (the topical calcineurin inhibitors [TCIs] pimecrolimus, and tacrolimus), excimer laser therapy, and phototherapy (psoralen with UVA [PUVA] and narrow-band UVB [nbUVB]). They collected information from medical policies available online or by direct contact with the plans in 2018.
The researchers reported data from 17 organizations with regional or national coverage policies for vitiligo treatment and two others – BlueCross BlueShield and Medicaid – which had policies that differed by state and plan. Of the 17 organizations, only 12% did not cover TCIs, 56% did not cover nbUVB phototherapy, 53% did not cover PUVA phototherapy, and 41% did not cover laser therapy.
As for BlueCross BlueShield, the health plan did not cover pimecrolimus and tacrolimus in 39% and 35% of states, respectively. At the same time, NbUVB and PUVA therapy were not covered in 20% and 10% of states, respectively, while excimer laser therapy was not covered in 82% of states.
Of accessible Medicaid information from 32 states, 11 did not cover topicals, 5 did not cover nbUVB, 4 did not cover PUVA, and 7 did not cover laser therapy. “The two most commonly cited reasons for denial of coverage were (a) vitiligo is considered a cosmetic condition and (b) certain therapies are not FDA-approved for vitiligo, though they may be approved for other skin conditions,” the study authors wrote.
While the analysis revealed that topical TCI therapy is more widely covered by insurance companies, compared with phototherapy, “multiple studies have shown that a combination of both topical and phototherapy is more effective in treating vitiligo than either alone,” they noted. “Vitiligo treatments can delay the progression of the disease and result in better outcomes when started early, furthering the need for insurance coverage of these treatments. If all proven and accepted vitiligo treatments were covered by their health insurers, patients would have better access, as well as timely and affordable ways by which to limit depigmentation and to repigment affected areas.”
In addition, lack of access to treatments “may increase health disparities among already-marginalized groups, such as children and adults of darker skin phototypes,” they wrote.
Seemal R. Desai, MD, who was asked to comment on the study, said that the findings resonate with him based on his clinical experience as a dermatologist at the University of Texas Southwestern Medical Center in Dallas and in clinical practice. “Vitiligo has a high psychological impact, continues to increase in its prevalence, and has been shown to be an autoimmune, chronic, inflammatory skin disease, yet we’re still having challenges with treatment,” said Dr. Desai, who is also a member of the board of directors for the American Academy of Dermatology and the Global Vitiligo Foundation (GVF).
He said that he is working with the AAD, the GVF, and other stakeholders to improve treatment coverage. For example, in Massachusetts, the Tufts Health Plan had stopped covering treatment for vitiligo. “Through a series of advocacy efforts, that was reversed a couple of years ago,” said Dr. Desai, who is also a past president of the Skin of Color Society. “We also have seen isolated reports of Medicaid and Medicare coverage where local contractors aren’t following national Centers for Medicare and Medicaid Service directive guidance. The challenge becomes, how do you get consistency in treatment coverage, and how do you make sure patients continue to get access to treatment?”
Turning the tide will require “a concerted effort” by dermatologists to engage with the payers, he added. “I’ve had to get on the phone with countless insurance companies on behalf of my patients and make them understand the comorbidities associated with vitiligo, sending them copies of studies that show it’s an autoimmune disease linked to thyroid issues,” Dr. Desai continued. “We talk a lot about the psychological burden and quality of life. There’s still a lot of work to be done in this sphere, but I think we’re making progress.”
With hopes that Janus kinase (JAK) inhibitors and other new products being investigated will soon be approved as a treatment option for vitiligo, Dr. Desai said that now is the time to standardize coverage for patients. “It’s important that we start talking about insurance coverage and denial issues now and get ahead of it, so that when we get those JAK inhibitors available, we don’t fight coverage decisions then.”
The researchers acknowledged certain limitations of the study, including the fact that it was based on insurance coverage from 2017 to 2018 and the lack of easily available state Medicaid policies.
The study coauthors were Colleen K. Gabel, MD, of the University of Massachusetts, Worcester, and Lionel G. Bercovitch, MD, of Brown University. None of the study authors reported financial disclosures.
Dr. Desai disclosed that he has conducted vitiligo research trials and has done consulting work for several pharmaceutical companies.
, which may disproportionately affect patients of color.
Those are the conclusions from an analysis of vitiligo treatment coverage policies across major health insurers in the United States.
“Vitiligo can be less noticeable in patients with lighter skin types, becoming apparent only when affected patches fail to tan,” first authors Andrew Blundell, MD, MSc, and Moniyka Sachar, MD, wrote in a study published online on July 16 in Pediatric Dermatology. However, they pointed out that, in patients with darker skin types, “vitiligo can be far more evident due to the stark contrast of involved versus uninvolved skin, and as such can lead to a significant impact on quality of life, as well as heightened stigmatization.”
Nevertheless, they noted many health care insurers consider vitiligo as a cosmetic condition, and do not cover treatments, and for the 1%-2% of the general population with vitiligo, “this lack of recognition from health care insurers makes treatments both less accessible and affordable, and only further marginalizes patients with this condition.”
Dr. Blundell, of San Juan Bautista School of Medicine, Caguas, P.R., and Dr. Sachar, of the department of dermatology at Brown University, Providence, R.I., and colleagues surveyed 15 commercial health care insurers, 50 BlueCross BlueShield plans, Medicare, Medicaid, and Veterans Affairs to determine the level of treatment coverage for vitiligo. They looked at office visits, medications (the topical calcineurin inhibitors [TCIs] pimecrolimus, and tacrolimus), excimer laser therapy, and phototherapy (psoralen with UVA [PUVA] and narrow-band UVB [nbUVB]). They collected information from medical policies available online or by direct contact with the plans in 2018.
The researchers reported data from 17 organizations with regional or national coverage policies for vitiligo treatment and two others – BlueCross BlueShield and Medicaid – which had policies that differed by state and plan. Of the 17 organizations, only 12% did not cover TCIs, 56% did not cover nbUVB phototherapy, 53% did not cover PUVA phototherapy, and 41% did not cover laser therapy.
As for BlueCross BlueShield, the health plan did not cover pimecrolimus and tacrolimus in 39% and 35% of states, respectively. At the same time, NbUVB and PUVA therapy were not covered in 20% and 10% of states, respectively, while excimer laser therapy was not covered in 82% of states.
Of accessible Medicaid information from 32 states, 11 did not cover topicals, 5 did not cover nbUVB, 4 did not cover PUVA, and 7 did not cover laser therapy. “The two most commonly cited reasons for denial of coverage were (a) vitiligo is considered a cosmetic condition and (b) certain therapies are not FDA-approved for vitiligo, though they may be approved for other skin conditions,” the study authors wrote.
While the analysis revealed that topical TCI therapy is more widely covered by insurance companies, compared with phototherapy, “multiple studies have shown that a combination of both topical and phototherapy is more effective in treating vitiligo than either alone,” they noted. “Vitiligo treatments can delay the progression of the disease and result in better outcomes when started early, furthering the need for insurance coverage of these treatments. If all proven and accepted vitiligo treatments were covered by their health insurers, patients would have better access, as well as timely and affordable ways by which to limit depigmentation and to repigment affected areas.”
In addition, lack of access to treatments “may increase health disparities among already-marginalized groups, such as children and adults of darker skin phototypes,” they wrote.
Seemal R. Desai, MD, who was asked to comment on the study, said that the findings resonate with him based on his clinical experience as a dermatologist at the University of Texas Southwestern Medical Center in Dallas and in clinical practice. “Vitiligo has a high psychological impact, continues to increase in its prevalence, and has been shown to be an autoimmune, chronic, inflammatory skin disease, yet we’re still having challenges with treatment,” said Dr. Desai, who is also a member of the board of directors for the American Academy of Dermatology and the Global Vitiligo Foundation (GVF).
He said that he is working with the AAD, the GVF, and other stakeholders to improve treatment coverage. For example, in Massachusetts, the Tufts Health Plan had stopped covering treatment for vitiligo. “Through a series of advocacy efforts, that was reversed a couple of years ago,” said Dr. Desai, who is also a past president of the Skin of Color Society. “We also have seen isolated reports of Medicaid and Medicare coverage where local contractors aren’t following national Centers for Medicare and Medicaid Service directive guidance. The challenge becomes, how do you get consistency in treatment coverage, and how do you make sure patients continue to get access to treatment?”
Turning the tide will require “a concerted effort” by dermatologists to engage with the payers, he added. “I’ve had to get on the phone with countless insurance companies on behalf of my patients and make them understand the comorbidities associated with vitiligo, sending them copies of studies that show it’s an autoimmune disease linked to thyroid issues,” Dr. Desai continued. “We talk a lot about the psychological burden and quality of life. There’s still a lot of work to be done in this sphere, but I think we’re making progress.”
With hopes that Janus kinase (JAK) inhibitors and other new products being investigated will soon be approved as a treatment option for vitiligo, Dr. Desai said that now is the time to standardize coverage for patients. “It’s important that we start talking about insurance coverage and denial issues now and get ahead of it, so that when we get those JAK inhibitors available, we don’t fight coverage decisions then.”
The researchers acknowledged certain limitations of the study, including the fact that it was based on insurance coverage from 2017 to 2018 and the lack of easily available state Medicaid policies.
The study coauthors were Colleen K. Gabel, MD, of the University of Massachusetts, Worcester, and Lionel G. Bercovitch, MD, of Brown University. None of the study authors reported financial disclosures.
Dr. Desai disclosed that he has conducted vitiligo research trials and has done consulting work for several pharmaceutical companies.
FROM PEDIATRIC DERMATOLOGY
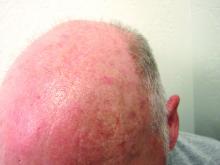
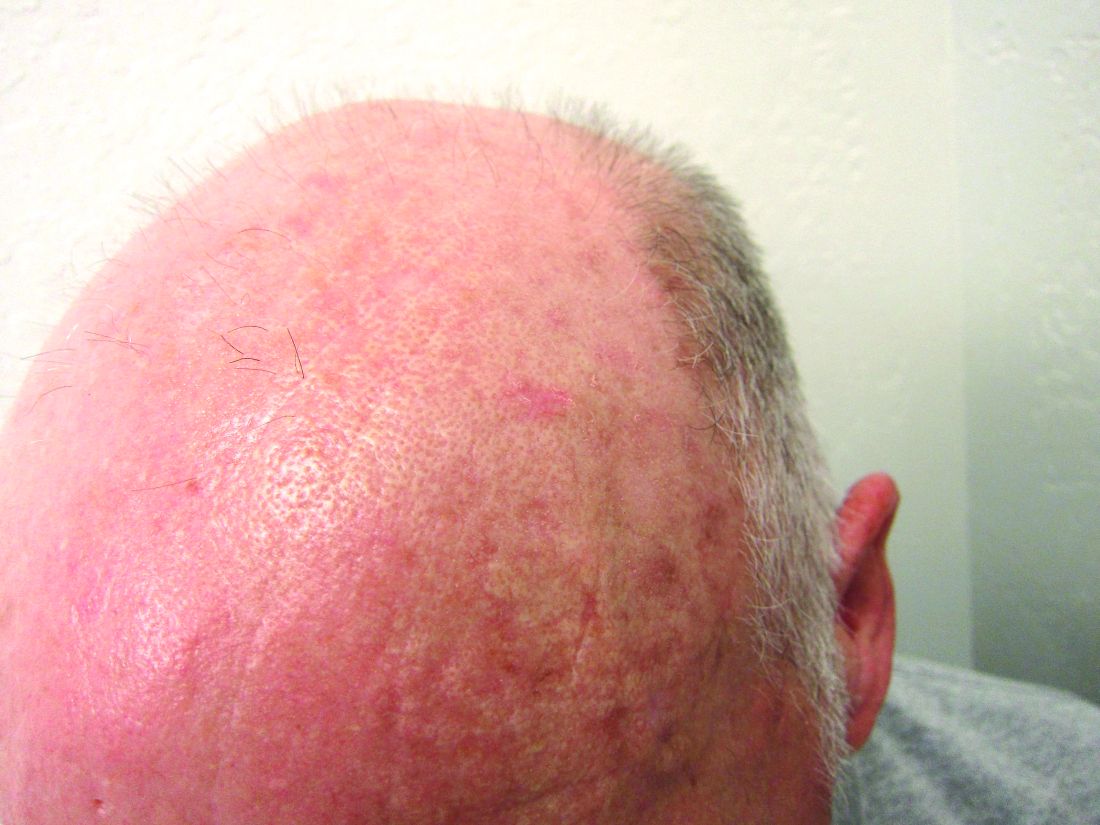
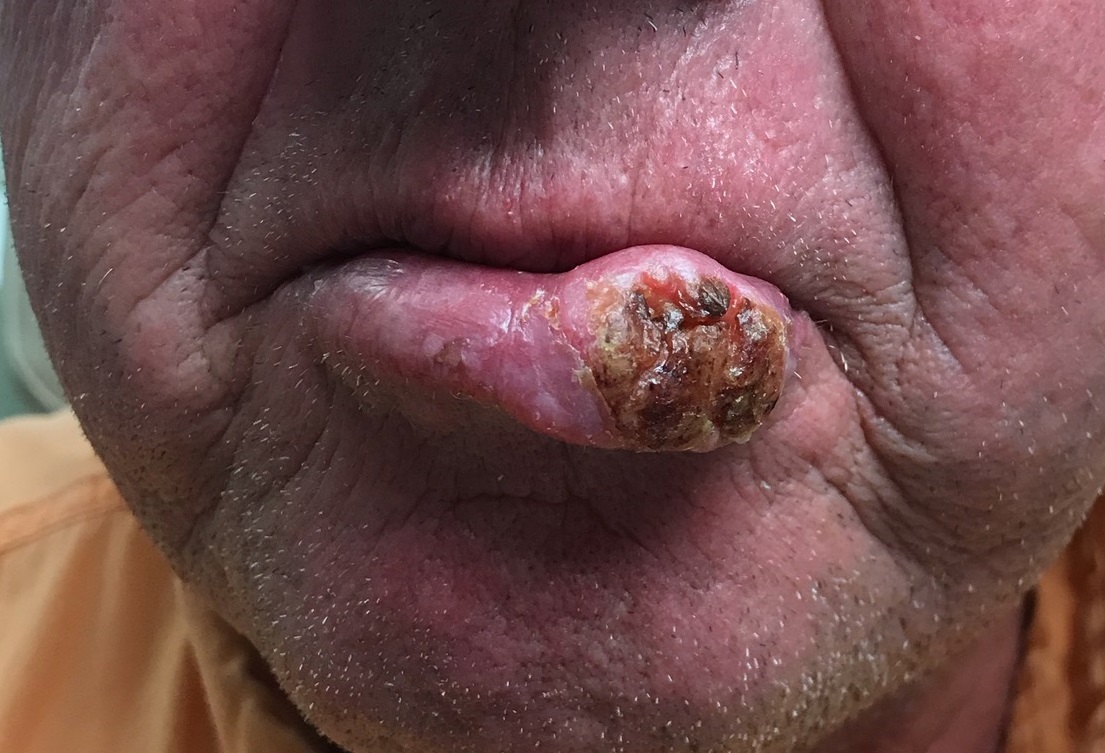
He Needs More Than Lip Service for This Lesion
ANSWER
The correct answer is all of the above (choice “g”).
DISCUSSION
Squamous cell carcinoma (SCC) of the lip—almost always the lower lip—is quite common and appears to be caused by several factors. These can include exposure to ultraviolet light, ionizing radiation, arsenic (through contaminated groundwater or certain medications), tobacco, and human papillomavirus.
Early on in its manifestation, this patient’s SCC could have been excised with margins, producing an excellent prognosis. But with the extended delay in diagnosis and apparent related adenopathy, the patient’s future looked much less certain. At the very least, he would face extensive surgery, possible lymph node dissection, and maybe chemo and radiation therapies. Metastasis to the brain and lung(s) were very real possibilities.
The history associated with this case is far from uncommon, since these cancers are often mistaken for infection, which further delays correct diagnosis and treatment. Compounding this problem—and for reasons unclear to this author—affected patients are often referred to the wrong specialist. The critical missing piece of information was a diagnosis, which could only have been obtained through an incisional biopsy. Only then could the patient be referred appropriately.
Fortunately, in this case, the patient ended up under the care of a head and neck surgeon who planned to address this tumor after imaging of the head, neck, and lungs was completed.
ANSWER
The correct answer is all of the above (choice “g”).
DISCUSSION
Squamous cell carcinoma (SCC) of the lip—almost always the lower lip—is quite common and appears to be caused by several factors. These can include exposure to ultraviolet light, ionizing radiation, arsenic (through contaminated groundwater or certain medications), tobacco, and human papillomavirus.
Early on in its manifestation, this patient’s SCC could have been excised with margins, producing an excellent prognosis. But with the extended delay in diagnosis and apparent related adenopathy, the patient’s future looked much less certain. At the very least, he would face extensive surgery, possible lymph node dissection, and maybe chemo and radiation therapies. Metastasis to the brain and lung(s) were very real possibilities.
The history associated with this case is far from uncommon, since these cancers are often mistaken for infection, which further delays correct diagnosis and treatment. Compounding this problem—and for reasons unclear to this author—affected patients are often referred to the wrong specialist. The critical missing piece of information was a diagnosis, which could only have been obtained through an incisional biopsy. Only then could the patient be referred appropriately.
Fortunately, in this case, the patient ended up under the care of a head and neck surgeon who planned to address this tumor after imaging of the head, neck, and lungs was completed.
ANSWER
The correct answer is all of the above (choice “g”).
DISCUSSION
Squamous cell carcinoma (SCC) of the lip—almost always the lower lip—is quite common and appears to be caused by several factors. These can include exposure to ultraviolet light, ionizing radiation, arsenic (through contaminated groundwater or certain medications), tobacco, and human papillomavirus.
Early on in its manifestation, this patient’s SCC could have been excised with margins, producing an excellent prognosis. But with the extended delay in diagnosis and apparent related adenopathy, the patient’s future looked much less certain. At the very least, he would face extensive surgery, possible lymph node dissection, and maybe chemo and radiation therapies. Metastasis to the brain and lung(s) were very real possibilities.
The history associated with this case is far from uncommon, since these cancers are often mistaken for infection, which further delays correct diagnosis and treatment. Compounding this problem—and for reasons unclear to this author—affected patients are often referred to the wrong specialist. The critical missing piece of information was a diagnosis, which could only have been obtained through an incisional biopsy. Only then could the patient be referred appropriately.
Fortunately, in this case, the patient ended up under the care of a head and neck surgeon who planned to address this tumor after imaging of the head, neck, and lungs was completed.
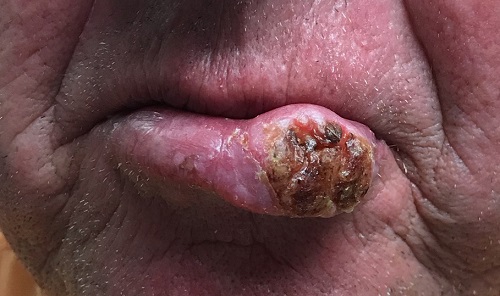
Several years ago, the patient developed what he thought was a cold sore, which waxed and waned but never quite healed. It grew considerably over time but caused no pain, so the patient and his wife assumed it was benign. A few times over the years, his primary care provider (PCP) prescribed oral antibiotics, then later acyclovir, to treat the lesion. When it became large enough to interfere with normal speech, the patient’s PCP referred him to a dentist, who in turn arranged an urgent consultation with dermatology.
The patient was retired but had spent more than 50 years working and recreating outdoors without any protection. For 40 of those adult years, he had smoked. His health was good in other respects.
The 3-cm warty mass on the vermillion surface of the lower lip, just to the left of center, was focally eroded and quite firm to touch. Palpation of local nodal locations revealed several fixed nontender nodes, most notably in the submental area. His sun-exposed skin was quite sun damaged, with stellate scarring, abundant dyschromia, telangiectasias, and marked solar atrophy.
A 5-mm punch biopsy was performed, showing poorly differentiated squamous cell carcinoma.
Physicians question the future of TNF inhibitors for psoriasis, PsA
Tumor necrosis factor inhibitors have long been the go-to treatment of choice for patients with psoriasis and psoriatic arthritis (PsA). They’ve served patients well since etanercept was first approved for PsA in 2002, but today, with the availability of more attractive interleukin-17 and IL-23 inhibitors, dermatologists and rheumatologists are asking whether it’s time to reconsider the use of TNF inhibitors as first-line therapy in psoriasis and PsA.
“TNF inhibitors have served psoriasis patients well for many years. The question is, ‘Is it time to move on from them as first-line agents for psoriasis?’ ” said April W. Armstrong, MD, MPH, a dermatologist and associate dean for clinical research at the University of Southern California, Los Angeles. Dr. Armstrong participated in a point/counterpoint debate about the merits of IL-17 and IL-23 inhibitors over TNF inhibitors at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. “For the majority of our patients, IL-17 and IL-23 inhibitors are probably rationally better than TNF inhibitors as first-line agents for moderate to severe plaque psoriasis,” she said.
In this debate, dermatologists and rheumatologists cited studies showing the safety and efficacy of IL-17 and IL-23 inhibitors over TNF inhibitors. TNF inhibitors include etanercept (Enbrel and biosimilars), infliximab (Remicade and biosimilars), adalimumab (Humira and biosimilars), certolizumab pegol (Cimzia), and golimumab (Simponi). IL-12/23 inhibitors are limited to ustekinumab (Stelara). IL-17 inhibitors include secukinumab (Cosentyx), ixekizumab (Taltz), and brodalumab (Siliq). IL-23 inhibitors include guselkumab (Tremfya), tildrakizumab (Ilumya), and risankizumab (Skyrizi).
TNF inhibitors are recommended by the American College of Rheumatology as first-line therapy for treatment-naive patients with active PsA, and they, along with IL-12/23, IL-17, and IL-23 inhibitors are all recommended by the American Academy of Dermatology as monotherapy treatment options in adult patients with moderate to severe plaque psoriasis. However, some studies have shown that non–TNF-inhibitor biologics have a higher efficacy than TNF inhibitors in some cases for some patients, such as those with moderate to severe psoriasis alone or for musculoskeletal efficacy in patients with PsA who have peripheral arthritis, enthesitis, dactylitis, or axial manifestations.
Favorable characteristics of non–TNF-inhibitor biologics
Dr. Armstrong cited a number of head-to-head trials to support her view that IL-17 and IL-23 inhibitors are better than TNF inhibitors as first-line agents for patients with moderate to severe plaque psoriasis. In the first head-to-head study of its kind in patients with moderate to severe psoriasis, ustekinumab proved superior to etanercept. Guselkumab was shown to be superior to adalimumab for patients with moderate to severe psoriasis. Tildrakizumab also proved superior to etanercept for patients with psoriasis. Risankizumab bested adalimumab in patients with moderate to severe psoriasis. Ixekizumab proved superior to etanercept in two pivotal studies of patients with widespread moderate-to-severe psoriasis.
IL-23 and IL-17 inhibitors tend to have less frequent maintenance dosing, with IL-17 inhibitors being once every 2 or 4 weeks and IL-23 inhibitors once every 8 or 12 weeks, compared with frequencies ranging from every week to every 8 weeks with TNF inhibitors, Dr. Armstrong said.
IL-17 and IL-23 inhibitors also appear to have fewer safety concerns than TNF inhibitors, although there is less long-term data for them overall and there are some notable exceptions in certain patient populations. TNF inhibitors should be avoided in patients with a history of demyelinating disease or hepatitis B virus infection, and they are not preferred in patients who have a history of latent tuberculosis or advanced heart failure. IL-17 inhibitors should not be used in patients with a history of inflammatory bowel disease, and their use is associated with a higher rate of oral candidiasis. IL-23 inhibitors have a good safety profile overall, she said.
“The IL-17/23 axis is very important to psoriatic arthritis and should be the focus of our treatments” for PsA, said Deepak Jadon, MBBCh, MRCP, PhD, a rheumatologist and director of the rheumatology research unit at Addenbrooke’s Hospital, Cambridge (England) University Hospitals NHS Foundation Trust. In his presentation, he proposed that IL-17 inhibitors and IL-23 inhibitors be used as first-line therapies in PsA ahead of TNF inhibitors.
One reason to go with IL-17 and IL-23 inhibitors may be to ”get it right immunologically the first time,” Dr. Jadon said. He cited evidence showing substantially better response to guselkumab when given to biologic-naive patients with PsA versus those who had a inadequate response to TNF inhibitors, as well as data indicating better response with secukinumab regardless of previous TNF inhibitor use.
IL-17 inhibitors target more domains of psoriatic disease than do TNF inhibitors, he said, noting that “they have excellent musculoskeletal efficacy in patients with moderate skin psoriasis, not just those with severe psoriasis.” Ixekizumab proved superior to adalimumab in biologic-naive patients with PsA. The results of this study also indicated that IL-17 inhibitors should not be reserved only for patients with severe psoriasis since a higher percentage of patients with moderate psoriasis who were taking ixekizumab achieved very low PsA activity. Secukinumab also beat adalimumab in a head-to-head comparison and showed a greater impact on some measures of health-related quality of life.
IL-17 inhibitors also do not require concomitant methotrexate, he said, “which is a major bonus for our patients. All of my patients wish to stop methotrexate even if tolerated. Not having to cope with prescribed methotrexate improves risk of adverse events and frequency of blood test monitoring.”
IL-17 and IL-23 inhibitors appear to have good efficacy against axial disease in patients with PsA. Randomized trial results for secukinumab versus placebo show high percentages of patients improving either 20% or 40% in Assessment in Spondyloarthritis International Society response criteria and reduced inflammatory MRI lesions in the spine and sacroiliac joints. Analyses of trial results in guselkumab-treated patients with axial manifestations of PsA have shown the IL-23 inhibitor’s efficacy versus placebo across different measures of disease activity.
Dr. Jadon also cited real-world data showing that patients stay longer on IL-17 and IL-12/23 inhibitors versus TNF inhibitors. A 2016 study of patients with psoriasis in the PSOLAR registry showed that patients persisted on treatment longer with ustekinumab than with adalimumab, etanercept, or infliximab. Similarly, a 2020 study of patients with psoriasis from the British Association of Dermatologists Biologics and Immunomodulators Register found that both ustekinumab and secukinumab had better sustained drug survival than did adalimumab.
Accessibility weighs heavily in using TNF inhibitor first
Clinical trials data show that IL-17 inhibitors outperform TNF inhibitors for psoriasis, but in clinical practice, TNF inhibitors still perform very well in individual patients and are well tolerated, said Amit Garg, MD, founding chair of the department of dermatology at Hofstra University, Hempstead, N.Y.
He argued in favor of TNF inhibitors as first-line therapy over IL-17 inhibitors for psoriasis. In this case, treatment decisions often come down to accessibility, Dr. Garg said. Not all insurance companies cover the cost of the newer IL-23 inhibitors. Plus, access to TNF inhibitors is widespread and costs are generally lower.
“As a physician, I don’t have complete autonomy in prescribing what I want. The reality is whether it be because of cross indication or discount pricing, [TNF inhibitors] – in particular adalimumab – is widely available on all plans and is usually the preferred treatment plan, at least in our area,” he said. “I’m not a big fan of plans that allow drugs at low or no cost for a year or 2, and then abandon the patients at that point thereafter. I like to use something that insurance will cover sustainably, and, quite frankly, TNFs have served well in that regard.”
However, TNF inhibitors are associated with more safety signals, plus they carry a greater risk of infection, leading to tolerability and persistence issues with patients.
“Psoriasis is a lifelong disease. I wish I could tell you that every drug is going to work well forever for individual patients, but I don’t think we know that yet. From my perspective, for efficacy, general well tolerance, convenience, and access, TNFs are still an important part of our ability to treat psoriasis effectively. I have no problem starting there and transitioning as needed for individual patients.
“In my experience, I think patients on TNFs generally do well. We don’t always get the patients clear and certainly there’s drop off of efficacy over time, but I’m not sure that’s a rationale for [changing treatment],” Dr. Garg said.
Ying Ying (Katy) Leung, MD, a rheumatologist with Singapore General Hospital, and a member of the GRAPPA peripheral arthritis working group, argued against the use of IL-17 and IL-23 inhibitors as first-line treatment for PsA over TNF inhibitors. She reasoned that TNF blockers are more accessible, have more long-term safety data (including data indicating safety during pregnancy), and have better cardiovascular protection. She also noted that GRAPPA treatment recommendations strongly advise using TNF blockers (or IL-17 inhibitors) for treatment-naive patients with PsA.
“Accessibility is very important as I learned along the way of leading the peripheral arthritis [GRAPPA] working group. Accessibility [issues] can be coming from a lot of sources, but if you don’t take good care of accessibility, you might be developing a guideline that is way out of reality and nobody is going to use it,” she said.
In her native Singapore, Dr. Leung said that patients pay for biologics out of pocket, so cost is a key factor for her patients. She stated that adalimumab is available as a biosimilar at about $200 monthly for patients with PsA in Singapore, while the average monthly costs are $1,400 for originator infliximab and $1,500 for originator etanercept. By comparison, secukinumab sells for about $750 monthly, ixekizumab $540 monthly, and guselkumab $2,000 monthly.
Treatment choices should be aligned with the disease manifestations of PsA, Dr. Leung said, keeping in mind that accessibility and individual patient needs and preferences should be considered as well. She conducted an informal comparison that found TNF inhibitors are most effective for patients with uveitis or inflammatory bowel disease. Evidence from head-to-head studies indicates that TNF inhibitors and IL-17 inhibitors have similar efficacy for peripheral arthritis, enthesitis, and dactylitis. But caution is warranted, she suggested, for determining the best biologics for axial disease because no head-to-head comparison trials have been conducted for IL-17 or IL-23 inhibitors versus TNF inhibitors.
Dr. Armstrong has been a consultant to AbbVie, Bristol-Myers Squibb, Dermira, Genzyme, Incyte, Janssen, Leo Pharma, Eli Lilly, Novartis, Pfizer, and UCB. Dr. Jadon has been a consultant to, has been on speakers bureaus for, and has received grant/research support from AbbVie, Amgen, Celgene, Celltrion, Gilead, Janssen, Eli Lilly, MSD, Novartis, Pfizer, Roche, Sandoz, and UCB. Dr. Garg has consulted for AbbVie, Boehringer Ingelheim, Janssen, and UCB. Dr. Leung has been a consultant to AbbVie, Boehringer Ingelheim, Janssen, Eli Lilly, Novartis, and Pfizer. She has been on speakers bureaus for AbbVie, Janssen Eli Lilly, and Novartis. She has received grant/research support from Pfizer and conference support from AbbVie,
Tumor necrosis factor inhibitors have long been the go-to treatment of choice for patients with psoriasis and psoriatic arthritis (PsA). They’ve served patients well since etanercept was first approved for PsA in 2002, but today, with the availability of more attractive interleukin-17 and IL-23 inhibitors, dermatologists and rheumatologists are asking whether it’s time to reconsider the use of TNF inhibitors as first-line therapy in psoriasis and PsA.
“TNF inhibitors have served psoriasis patients well for many years. The question is, ‘Is it time to move on from them as first-line agents for psoriasis?’ ” said April W. Armstrong, MD, MPH, a dermatologist and associate dean for clinical research at the University of Southern California, Los Angeles. Dr. Armstrong participated in a point/counterpoint debate about the merits of IL-17 and IL-23 inhibitors over TNF inhibitors at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. “For the majority of our patients, IL-17 and IL-23 inhibitors are probably rationally better than TNF inhibitors as first-line agents for moderate to severe plaque psoriasis,” she said.
In this debate, dermatologists and rheumatologists cited studies showing the safety and efficacy of IL-17 and IL-23 inhibitors over TNF inhibitors. TNF inhibitors include etanercept (Enbrel and biosimilars), infliximab (Remicade and biosimilars), adalimumab (Humira and biosimilars), certolizumab pegol (Cimzia), and golimumab (Simponi). IL-12/23 inhibitors are limited to ustekinumab (Stelara). IL-17 inhibitors include secukinumab (Cosentyx), ixekizumab (Taltz), and brodalumab (Siliq). IL-23 inhibitors include guselkumab (Tremfya), tildrakizumab (Ilumya), and risankizumab (Skyrizi).
TNF inhibitors are recommended by the American College of Rheumatology as first-line therapy for treatment-naive patients with active PsA, and they, along with IL-12/23, IL-17, and IL-23 inhibitors are all recommended by the American Academy of Dermatology as monotherapy treatment options in adult patients with moderate to severe plaque psoriasis. However, some studies have shown that non–TNF-inhibitor biologics have a higher efficacy than TNF inhibitors in some cases for some patients, such as those with moderate to severe psoriasis alone or for musculoskeletal efficacy in patients with PsA who have peripheral arthritis, enthesitis, dactylitis, or axial manifestations.
Favorable characteristics of non–TNF-inhibitor biologics
Dr. Armstrong cited a number of head-to-head trials to support her view that IL-17 and IL-23 inhibitors are better than TNF inhibitors as first-line agents for patients with moderate to severe plaque psoriasis. In the first head-to-head study of its kind in patients with moderate to severe psoriasis, ustekinumab proved superior to etanercept. Guselkumab was shown to be superior to adalimumab for patients with moderate to severe psoriasis. Tildrakizumab also proved superior to etanercept for patients with psoriasis. Risankizumab bested adalimumab in patients with moderate to severe psoriasis. Ixekizumab proved superior to etanercept in two pivotal studies of patients with widespread moderate-to-severe psoriasis.
IL-23 and IL-17 inhibitors tend to have less frequent maintenance dosing, with IL-17 inhibitors being once every 2 or 4 weeks and IL-23 inhibitors once every 8 or 12 weeks, compared with frequencies ranging from every week to every 8 weeks with TNF inhibitors, Dr. Armstrong said.
IL-17 and IL-23 inhibitors also appear to have fewer safety concerns than TNF inhibitors, although there is less long-term data for them overall and there are some notable exceptions in certain patient populations. TNF inhibitors should be avoided in patients with a history of demyelinating disease or hepatitis B virus infection, and they are not preferred in patients who have a history of latent tuberculosis or advanced heart failure. IL-17 inhibitors should not be used in patients with a history of inflammatory bowel disease, and their use is associated with a higher rate of oral candidiasis. IL-23 inhibitors have a good safety profile overall, she said.
“The IL-17/23 axis is very important to psoriatic arthritis and should be the focus of our treatments” for PsA, said Deepak Jadon, MBBCh, MRCP, PhD, a rheumatologist and director of the rheumatology research unit at Addenbrooke’s Hospital, Cambridge (England) University Hospitals NHS Foundation Trust. In his presentation, he proposed that IL-17 inhibitors and IL-23 inhibitors be used as first-line therapies in PsA ahead of TNF inhibitors.
One reason to go with IL-17 and IL-23 inhibitors may be to ”get it right immunologically the first time,” Dr. Jadon said. He cited evidence showing substantially better response to guselkumab when given to biologic-naive patients with PsA versus those who had a inadequate response to TNF inhibitors, as well as data indicating better response with secukinumab regardless of previous TNF inhibitor use.
IL-17 inhibitors target more domains of psoriatic disease than do TNF inhibitors, he said, noting that “they have excellent musculoskeletal efficacy in patients with moderate skin psoriasis, not just those with severe psoriasis.” Ixekizumab proved superior to adalimumab in biologic-naive patients with PsA. The results of this study also indicated that IL-17 inhibitors should not be reserved only for patients with severe psoriasis since a higher percentage of patients with moderate psoriasis who were taking ixekizumab achieved very low PsA activity. Secukinumab also beat adalimumab in a head-to-head comparison and showed a greater impact on some measures of health-related quality of life.
IL-17 inhibitors also do not require concomitant methotrexate, he said, “which is a major bonus for our patients. All of my patients wish to stop methotrexate even if tolerated. Not having to cope with prescribed methotrexate improves risk of adverse events and frequency of blood test monitoring.”
IL-17 and IL-23 inhibitors appear to have good efficacy against axial disease in patients with PsA. Randomized trial results for secukinumab versus placebo show high percentages of patients improving either 20% or 40% in Assessment in Spondyloarthritis International Society response criteria and reduced inflammatory MRI lesions in the spine and sacroiliac joints. Analyses of trial results in guselkumab-treated patients with axial manifestations of PsA have shown the IL-23 inhibitor’s efficacy versus placebo across different measures of disease activity.
Dr. Jadon also cited real-world data showing that patients stay longer on IL-17 and IL-12/23 inhibitors versus TNF inhibitors. A 2016 study of patients with psoriasis in the PSOLAR registry showed that patients persisted on treatment longer with ustekinumab than with adalimumab, etanercept, or infliximab. Similarly, a 2020 study of patients with psoriasis from the British Association of Dermatologists Biologics and Immunomodulators Register found that both ustekinumab and secukinumab had better sustained drug survival than did adalimumab.
Accessibility weighs heavily in using TNF inhibitor first
Clinical trials data show that IL-17 inhibitors outperform TNF inhibitors for psoriasis, but in clinical practice, TNF inhibitors still perform very well in individual patients and are well tolerated, said Amit Garg, MD, founding chair of the department of dermatology at Hofstra University, Hempstead, N.Y.
He argued in favor of TNF inhibitors as first-line therapy over IL-17 inhibitors for psoriasis. In this case, treatment decisions often come down to accessibility, Dr. Garg said. Not all insurance companies cover the cost of the newer IL-23 inhibitors. Plus, access to TNF inhibitors is widespread and costs are generally lower.
“As a physician, I don’t have complete autonomy in prescribing what I want. The reality is whether it be because of cross indication or discount pricing, [TNF inhibitors] – in particular adalimumab – is widely available on all plans and is usually the preferred treatment plan, at least in our area,” he said. “I’m not a big fan of plans that allow drugs at low or no cost for a year or 2, and then abandon the patients at that point thereafter. I like to use something that insurance will cover sustainably, and, quite frankly, TNFs have served well in that regard.”
However, TNF inhibitors are associated with more safety signals, plus they carry a greater risk of infection, leading to tolerability and persistence issues with patients.
“Psoriasis is a lifelong disease. I wish I could tell you that every drug is going to work well forever for individual patients, but I don’t think we know that yet. From my perspective, for efficacy, general well tolerance, convenience, and access, TNFs are still an important part of our ability to treat psoriasis effectively. I have no problem starting there and transitioning as needed for individual patients.
“In my experience, I think patients on TNFs generally do well. We don’t always get the patients clear and certainly there’s drop off of efficacy over time, but I’m not sure that’s a rationale for [changing treatment],” Dr. Garg said.
Ying Ying (Katy) Leung, MD, a rheumatologist with Singapore General Hospital, and a member of the GRAPPA peripheral arthritis working group, argued against the use of IL-17 and IL-23 inhibitors as first-line treatment for PsA over TNF inhibitors. She reasoned that TNF blockers are more accessible, have more long-term safety data (including data indicating safety during pregnancy), and have better cardiovascular protection. She also noted that GRAPPA treatment recommendations strongly advise using TNF blockers (or IL-17 inhibitors) for treatment-naive patients with PsA.
“Accessibility is very important as I learned along the way of leading the peripheral arthritis [GRAPPA] working group. Accessibility [issues] can be coming from a lot of sources, but if you don’t take good care of accessibility, you might be developing a guideline that is way out of reality and nobody is going to use it,” she said.
In her native Singapore, Dr. Leung said that patients pay for biologics out of pocket, so cost is a key factor for her patients. She stated that adalimumab is available as a biosimilar at about $200 monthly for patients with PsA in Singapore, while the average monthly costs are $1,400 for originator infliximab and $1,500 for originator etanercept. By comparison, secukinumab sells for about $750 monthly, ixekizumab $540 monthly, and guselkumab $2,000 monthly.
Treatment choices should be aligned with the disease manifestations of PsA, Dr. Leung said, keeping in mind that accessibility and individual patient needs and preferences should be considered as well. She conducted an informal comparison that found TNF inhibitors are most effective for patients with uveitis or inflammatory bowel disease. Evidence from head-to-head studies indicates that TNF inhibitors and IL-17 inhibitors have similar efficacy for peripheral arthritis, enthesitis, and dactylitis. But caution is warranted, she suggested, for determining the best biologics for axial disease because no head-to-head comparison trials have been conducted for IL-17 or IL-23 inhibitors versus TNF inhibitors.
Dr. Armstrong has been a consultant to AbbVie, Bristol-Myers Squibb, Dermira, Genzyme, Incyte, Janssen, Leo Pharma, Eli Lilly, Novartis, Pfizer, and UCB. Dr. Jadon has been a consultant to, has been on speakers bureaus for, and has received grant/research support from AbbVie, Amgen, Celgene, Celltrion, Gilead, Janssen, Eli Lilly, MSD, Novartis, Pfizer, Roche, Sandoz, and UCB. Dr. Garg has consulted for AbbVie, Boehringer Ingelheim, Janssen, and UCB. Dr. Leung has been a consultant to AbbVie, Boehringer Ingelheim, Janssen, Eli Lilly, Novartis, and Pfizer. She has been on speakers bureaus for AbbVie, Janssen Eli Lilly, and Novartis. She has received grant/research support from Pfizer and conference support from AbbVie,
Tumor necrosis factor inhibitors have long been the go-to treatment of choice for patients with psoriasis and psoriatic arthritis (PsA). They’ve served patients well since etanercept was first approved for PsA in 2002, but today, with the availability of more attractive interleukin-17 and IL-23 inhibitors, dermatologists and rheumatologists are asking whether it’s time to reconsider the use of TNF inhibitors as first-line therapy in psoriasis and PsA.
“TNF inhibitors have served psoriasis patients well for many years. The question is, ‘Is it time to move on from them as first-line agents for psoriasis?’ ” said April W. Armstrong, MD, MPH, a dermatologist and associate dean for clinical research at the University of Southern California, Los Angeles. Dr. Armstrong participated in a point/counterpoint debate about the merits of IL-17 and IL-23 inhibitors over TNF inhibitors at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. “For the majority of our patients, IL-17 and IL-23 inhibitors are probably rationally better than TNF inhibitors as first-line agents for moderate to severe plaque psoriasis,” she said.
In this debate, dermatologists and rheumatologists cited studies showing the safety and efficacy of IL-17 and IL-23 inhibitors over TNF inhibitors. TNF inhibitors include etanercept (Enbrel and biosimilars), infliximab (Remicade and biosimilars), adalimumab (Humira and biosimilars), certolizumab pegol (Cimzia), and golimumab (Simponi). IL-12/23 inhibitors are limited to ustekinumab (Stelara). IL-17 inhibitors include secukinumab (Cosentyx), ixekizumab (Taltz), and brodalumab (Siliq). IL-23 inhibitors include guselkumab (Tremfya), tildrakizumab (Ilumya), and risankizumab (Skyrizi).
TNF inhibitors are recommended by the American College of Rheumatology as first-line therapy for treatment-naive patients with active PsA, and they, along with IL-12/23, IL-17, and IL-23 inhibitors are all recommended by the American Academy of Dermatology as monotherapy treatment options in adult patients with moderate to severe plaque psoriasis. However, some studies have shown that non–TNF-inhibitor biologics have a higher efficacy than TNF inhibitors in some cases for some patients, such as those with moderate to severe psoriasis alone or for musculoskeletal efficacy in patients with PsA who have peripheral arthritis, enthesitis, dactylitis, or axial manifestations.
Favorable characteristics of non–TNF-inhibitor biologics
Dr. Armstrong cited a number of head-to-head trials to support her view that IL-17 and IL-23 inhibitors are better than TNF inhibitors as first-line agents for patients with moderate to severe plaque psoriasis. In the first head-to-head study of its kind in patients with moderate to severe psoriasis, ustekinumab proved superior to etanercept. Guselkumab was shown to be superior to adalimumab for patients with moderate to severe psoriasis. Tildrakizumab also proved superior to etanercept for patients with psoriasis. Risankizumab bested adalimumab in patients with moderate to severe psoriasis. Ixekizumab proved superior to etanercept in two pivotal studies of patients with widespread moderate-to-severe psoriasis.
IL-23 and IL-17 inhibitors tend to have less frequent maintenance dosing, with IL-17 inhibitors being once every 2 or 4 weeks and IL-23 inhibitors once every 8 or 12 weeks, compared with frequencies ranging from every week to every 8 weeks with TNF inhibitors, Dr. Armstrong said.
IL-17 and IL-23 inhibitors also appear to have fewer safety concerns than TNF inhibitors, although there is less long-term data for them overall and there are some notable exceptions in certain patient populations. TNF inhibitors should be avoided in patients with a history of demyelinating disease or hepatitis B virus infection, and they are not preferred in patients who have a history of latent tuberculosis or advanced heart failure. IL-17 inhibitors should not be used in patients with a history of inflammatory bowel disease, and their use is associated with a higher rate of oral candidiasis. IL-23 inhibitors have a good safety profile overall, she said.
“The IL-17/23 axis is very important to psoriatic arthritis and should be the focus of our treatments” for PsA, said Deepak Jadon, MBBCh, MRCP, PhD, a rheumatologist and director of the rheumatology research unit at Addenbrooke’s Hospital, Cambridge (England) University Hospitals NHS Foundation Trust. In his presentation, he proposed that IL-17 inhibitors and IL-23 inhibitors be used as first-line therapies in PsA ahead of TNF inhibitors.
One reason to go with IL-17 and IL-23 inhibitors may be to ”get it right immunologically the first time,” Dr. Jadon said. He cited evidence showing substantially better response to guselkumab when given to biologic-naive patients with PsA versus those who had a inadequate response to TNF inhibitors, as well as data indicating better response with secukinumab regardless of previous TNF inhibitor use.
IL-17 inhibitors target more domains of psoriatic disease than do TNF inhibitors, he said, noting that “they have excellent musculoskeletal efficacy in patients with moderate skin psoriasis, not just those with severe psoriasis.” Ixekizumab proved superior to adalimumab in biologic-naive patients with PsA. The results of this study also indicated that IL-17 inhibitors should not be reserved only for patients with severe psoriasis since a higher percentage of patients with moderate psoriasis who were taking ixekizumab achieved very low PsA activity. Secukinumab also beat adalimumab in a head-to-head comparison and showed a greater impact on some measures of health-related quality of life.
IL-17 inhibitors also do not require concomitant methotrexate, he said, “which is a major bonus for our patients. All of my patients wish to stop methotrexate even if tolerated. Not having to cope with prescribed methotrexate improves risk of adverse events and frequency of blood test monitoring.”
IL-17 and IL-23 inhibitors appear to have good efficacy against axial disease in patients with PsA. Randomized trial results for secukinumab versus placebo show high percentages of patients improving either 20% or 40% in Assessment in Spondyloarthritis International Society response criteria and reduced inflammatory MRI lesions in the spine and sacroiliac joints. Analyses of trial results in guselkumab-treated patients with axial manifestations of PsA have shown the IL-23 inhibitor’s efficacy versus placebo across different measures of disease activity.
Dr. Jadon also cited real-world data showing that patients stay longer on IL-17 and IL-12/23 inhibitors versus TNF inhibitors. A 2016 study of patients with psoriasis in the PSOLAR registry showed that patients persisted on treatment longer with ustekinumab than with adalimumab, etanercept, or infliximab. Similarly, a 2020 study of patients with psoriasis from the British Association of Dermatologists Biologics and Immunomodulators Register found that both ustekinumab and secukinumab had better sustained drug survival than did adalimumab.
Accessibility weighs heavily in using TNF inhibitor first
Clinical trials data show that IL-17 inhibitors outperform TNF inhibitors for psoriasis, but in clinical practice, TNF inhibitors still perform very well in individual patients and are well tolerated, said Amit Garg, MD, founding chair of the department of dermatology at Hofstra University, Hempstead, N.Y.
He argued in favor of TNF inhibitors as first-line therapy over IL-17 inhibitors for psoriasis. In this case, treatment decisions often come down to accessibility, Dr. Garg said. Not all insurance companies cover the cost of the newer IL-23 inhibitors. Plus, access to TNF inhibitors is widespread and costs are generally lower.
“As a physician, I don’t have complete autonomy in prescribing what I want. The reality is whether it be because of cross indication or discount pricing, [TNF inhibitors] – in particular adalimumab – is widely available on all plans and is usually the preferred treatment plan, at least in our area,” he said. “I’m not a big fan of plans that allow drugs at low or no cost for a year or 2, and then abandon the patients at that point thereafter. I like to use something that insurance will cover sustainably, and, quite frankly, TNFs have served well in that regard.”
However, TNF inhibitors are associated with more safety signals, plus they carry a greater risk of infection, leading to tolerability and persistence issues with patients.
“Psoriasis is a lifelong disease. I wish I could tell you that every drug is going to work well forever for individual patients, but I don’t think we know that yet. From my perspective, for efficacy, general well tolerance, convenience, and access, TNFs are still an important part of our ability to treat psoriasis effectively. I have no problem starting there and transitioning as needed for individual patients.
“In my experience, I think patients on TNFs generally do well. We don’t always get the patients clear and certainly there’s drop off of efficacy over time, but I’m not sure that’s a rationale for [changing treatment],” Dr. Garg said.
Ying Ying (Katy) Leung, MD, a rheumatologist with Singapore General Hospital, and a member of the GRAPPA peripheral arthritis working group, argued against the use of IL-17 and IL-23 inhibitors as first-line treatment for PsA over TNF inhibitors. She reasoned that TNF blockers are more accessible, have more long-term safety data (including data indicating safety during pregnancy), and have better cardiovascular protection. She also noted that GRAPPA treatment recommendations strongly advise using TNF blockers (or IL-17 inhibitors) for treatment-naive patients with PsA.
“Accessibility is very important as I learned along the way of leading the peripheral arthritis [GRAPPA] working group. Accessibility [issues] can be coming from a lot of sources, but if you don’t take good care of accessibility, you might be developing a guideline that is way out of reality and nobody is going to use it,” she said.
In her native Singapore, Dr. Leung said that patients pay for biologics out of pocket, so cost is a key factor for her patients. She stated that adalimumab is available as a biosimilar at about $200 monthly for patients with PsA in Singapore, while the average monthly costs are $1,400 for originator infliximab and $1,500 for originator etanercept. By comparison, secukinumab sells for about $750 monthly, ixekizumab $540 monthly, and guselkumab $2,000 monthly.
Treatment choices should be aligned with the disease manifestations of PsA, Dr. Leung said, keeping in mind that accessibility and individual patient needs and preferences should be considered as well. She conducted an informal comparison that found TNF inhibitors are most effective for patients with uveitis or inflammatory bowel disease. Evidence from head-to-head studies indicates that TNF inhibitors and IL-17 inhibitors have similar efficacy for peripheral arthritis, enthesitis, and dactylitis. But caution is warranted, she suggested, for determining the best biologics for axial disease because no head-to-head comparison trials have been conducted for IL-17 or IL-23 inhibitors versus TNF inhibitors.
Dr. Armstrong has been a consultant to AbbVie, Bristol-Myers Squibb, Dermira, Genzyme, Incyte, Janssen, Leo Pharma, Eli Lilly, Novartis, Pfizer, and UCB. Dr. Jadon has been a consultant to, has been on speakers bureaus for, and has received grant/research support from AbbVie, Amgen, Celgene, Celltrion, Gilead, Janssen, Eli Lilly, MSD, Novartis, Pfizer, Roche, Sandoz, and UCB. Dr. Garg has consulted for AbbVie, Boehringer Ingelheim, Janssen, and UCB. Dr. Leung has been a consultant to AbbVie, Boehringer Ingelheim, Janssen, Eli Lilly, Novartis, and Pfizer. She has been on speakers bureaus for AbbVie, Janssen Eli Lilly, and Novartis. She has received grant/research support from Pfizer and conference support from AbbVie,
FROM THE GRAPPA 2021 ANNUAL MEETING
Androgenetic alopecia fuels negative emotions and poor quality of life
and meta-analysis of 41 studies.
“Hair loss affects self-image, causes trichodynia, and plays a role in emotions and social activity, which may be associated with psychiatric problems and impaired health-related quality of life,” wrote Chun-Hsien Huang, MD, of Chang Gung Memorial Hospital, Linkou, Taiwan, and colleagues. However, systematic reviews of the associations between androgenetic alopecia (AGA) and health-related quality of life (HRQOL) are lacking, they said.
In a study published in JAMA Dermatology, the researchers reviewed data from a total of 7,995 AGA patients in 41 studies. The studies included 11 tools for HRQOL assessment and 29 tools for psychological assessment. Of these, the Dermatology Life Quality Index (DLQI) and the Hair-Specific Skindex-29 were used to assess quality of life, and the Center for Epidemiologic Studies Depression Scale (CES-D) was used for psychological assessment in the meta-analysis.
Overall, 27 studies identified 18 factors associated with HRQOL; those with an inverse effect were higher self-rated hair loss severity, lower VAS score, and higher educational level. Of note, neither physician-rated hair loss severity nor treatment response were factors in HRQOL, the researchers said.
The pooled DLQI score across studies was 8.16, and subgroup analysis showed no differences in HRQOL between men and women or between patients from European vs. Asian countries. However, five studies showed significant differences in HRQOL between men and women when different assessment tools were used, which emphasized the need for more studies to examine the association of AGA with HRQOL by sex, the researchers said.
The meta-analysis of the Hair-Specific Skindex-29 scores showed pooled averages of 21.95 for symptom dimension, 18.52 in function dimension, and 29.22 in emotion dimension. Of these, the emotion dimension scores indicated moderate emotional impairment.
The average pooled score on the CES-D in the meta-analysis was 14.98, indicating no association between AGA and depression, the researchers said. However, “depression accounts for only a part of the emotion dimension,” they said. “Therefore, emotion dimension could be impaired even if no depressive symptoms were noted.”
The pooled DLQI scores for AGA (8.16) were higher than scores for other skin conditions including alopecia areata (6.3), contact dermatitis (7.35), and acne vulgaris (7.45), but lower than the pooled scores for vitiligo (9.11), urticaria (9.8), psoriasis (10.53), and atopic dermatitis (11.2), the researchers noted. “However, additional head-to-head studies are needed for direct comparisons of HRQOL in patients with various dermatoses,” they said.
The study findings were limited by the cross-sectional design of many of the included studies, and the limited number of assessment tools included in the analysis, the researchers noted. Other limitations were the lack of specific domain scores and the inclusion of only three studies from China, they said.
However, the results are consistent with findings from previous studies, and suggest that patients with AGA may benefit from psychological and psychosocial support, the researchers said.
Quality of life issues deserve attention
“Studies of the quality-of-life impact of various conditions are becoming more common in the medical literature,” Jamie B. MacKelfresh, MD, associate professor of dermatology, Emory University, Atlanta, said in an interview.
“Androgenetic alopecia is the most common type of hair loss in men and women,” she noted. “Hair loss can be labeled as a cosmetic concern, so it is important that providers understand the significant quality-of-life impact androgenetic alopecia has on the many people with this diagnosis,” she emphasized.
Dr. MacKelfresh, who was asked to comment on the study, said she was surprised that the subgroup analysis of the DLQI showed no significant difference between men and women. “This surprised me because a number of past studies have highlighted the relatively greater quality-of-life impact of hair loss on women compared to men,” she noted.
However, she added, “I was not surprised to see that androgenetic alopecia has a significant quality-of-life impact on many patients, and that physician objective assessments of the hair loss do not always correlate with the amount of quality-of-life impact,” said Dr. MacKelfresh. “In the patients I see, I find hair loss very often has a significant quality-of-life impact on patients, regardless of gender, and the amount of quality-of-life impact definitely does not always correlate with the objective amount of hair loss,” she noted.
A takeaway message for clinicians is to be aware that androgenetic alopecia frequently has a significant impact on patients, “particularly in the emotional dimension,” and can affect both men and women, Dr. MacKelfresh said. “Objective assessments of hair loss severity by providers may not accurately predict the degree of quality-of-life impact a patient may experience; therefore providers should include quality-of-life questions as part of their standard evaluation of patients with androgenetic alopecia,” she said. In addition to treating the hair loss, providers can help these patients by guiding them to psychological support resources, she emphasized.
More research is needed to assess the impact of androgenetic alopecia on “men, women, and the non-binary gender population,” as well as the relationship between self-esteem and hair loss, she said. “Finally, it would be helpful to understand what interventions can best help improve androgenetic alopecia patients’ quality of life,” she noted.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. MacKelfresh had no financial conflicts to disclose.
and meta-analysis of 41 studies.
“Hair loss affects self-image, causes trichodynia, and plays a role in emotions and social activity, which may be associated with psychiatric problems and impaired health-related quality of life,” wrote Chun-Hsien Huang, MD, of Chang Gung Memorial Hospital, Linkou, Taiwan, and colleagues. However, systematic reviews of the associations between androgenetic alopecia (AGA) and health-related quality of life (HRQOL) are lacking, they said.
In a study published in JAMA Dermatology, the researchers reviewed data from a total of 7,995 AGA patients in 41 studies. The studies included 11 tools for HRQOL assessment and 29 tools for psychological assessment. Of these, the Dermatology Life Quality Index (DLQI) and the Hair-Specific Skindex-29 were used to assess quality of life, and the Center for Epidemiologic Studies Depression Scale (CES-D) was used for psychological assessment in the meta-analysis.
Overall, 27 studies identified 18 factors associated with HRQOL; those with an inverse effect were higher self-rated hair loss severity, lower VAS score, and higher educational level. Of note, neither physician-rated hair loss severity nor treatment response were factors in HRQOL, the researchers said.
The pooled DLQI score across studies was 8.16, and subgroup analysis showed no differences in HRQOL between men and women or between patients from European vs. Asian countries. However, five studies showed significant differences in HRQOL between men and women when different assessment tools were used, which emphasized the need for more studies to examine the association of AGA with HRQOL by sex, the researchers said.
The meta-analysis of the Hair-Specific Skindex-29 scores showed pooled averages of 21.95 for symptom dimension, 18.52 in function dimension, and 29.22 in emotion dimension. Of these, the emotion dimension scores indicated moderate emotional impairment.
The average pooled score on the CES-D in the meta-analysis was 14.98, indicating no association between AGA and depression, the researchers said. However, “depression accounts for only a part of the emotion dimension,” they said. “Therefore, emotion dimension could be impaired even if no depressive symptoms were noted.”
The pooled DLQI scores for AGA (8.16) were higher than scores for other skin conditions including alopecia areata (6.3), contact dermatitis (7.35), and acne vulgaris (7.45), but lower than the pooled scores for vitiligo (9.11), urticaria (9.8), psoriasis (10.53), and atopic dermatitis (11.2), the researchers noted. “However, additional head-to-head studies are needed for direct comparisons of HRQOL in patients with various dermatoses,” they said.
The study findings were limited by the cross-sectional design of many of the included studies, and the limited number of assessment tools included in the analysis, the researchers noted. Other limitations were the lack of specific domain scores and the inclusion of only three studies from China, they said.
However, the results are consistent with findings from previous studies, and suggest that patients with AGA may benefit from psychological and psychosocial support, the researchers said.
Quality of life issues deserve attention
“Studies of the quality-of-life impact of various conditions are becoming more common in the medical literature,” Jamie B. MacKelfresh, MD, associate professor of dermatology, Emory University, Atlanta, said in an interview.
“Androgenetic alopecia is the most common type of hair loss in men and women,” she noted. “Hair loss can be labeled as a cosmetic concern, so it is important that providers understand the significant quality-of-life impact androgenetic alopecia has on the many people with this diagnosis,” she emphasized.
Dr. MacKelfresh, who was asked to comment on the study, said she was surprised that the subgroup analysis of the DLQI showed no significant difference between men and women. “This surprised me because a number of past studies have highlighted the relatively greater quality-of-life impact of hair loss on women compared to men,” she noted.
However, she added, “I was not surprised to see that androgenetic alopecia has a significant quality-of-life impact on many patients, and that physician objective assessments of the hair loss do not always correlate with the amount of quality-of-life impact,” said Dr. MacKelfresh. “In the patients I see, I find hair loss very often has a significant quality-of-life impact on patients, regardless of gender, and the amount of quality-of-life impact definitely does not always correlate with the objective amount of hair loss,” she noted.
A takeaway message for clinicians is to be aware that androgenetic alopecia frequently has a significant impact on patients, “particularly in the emotional dimension,” and can affect both men and women, Dr. MacKelfresh said. “Objective assessments of hair loss severity by providers may not accurately predict the degree of quality-of-life impact a patient may experience; therefore providers should include quality-of-life questions as part of their standard evaluation of patients with androgenetic alopecia,” she said. In addition to treating the hair loss, providers can help these patients by guiding them to psychological support resources, she emphasized.
More research is needed to assess the impact of androgenetic alopecia on “men, women, and the non-binary gender population,” as well as the relationship between self-esteem and hair loss, she said. “Finally, it would be helpful to understand what interventions can best help improve androgenetic alopecia patients’ quality of life,” she noted.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. MacKelfresh had no financial conflicts to disclose.
and meta-analysis of 41 studies.
“Hair loss affects self-image, causes trichodynia, and plays a role in emotions and social activity, which may be associated with psychiatric problems and impaired health-related quality of life,” wrote Chun-Hsien Huang, MD, of Chang Gung Memorial Hospital, Linkou, Taiwan, and colleagues. However, systematic reviews of the associations between androgenetic alopecia (AGA) and health-related quality of life (HRQOL) are lacking, they said.
In a study published in JAMA Dermatology, the researchers reviewed data from a total of 7,995 AGA patients in 41 studies. The studies included 11 tools for HRQOL assessment and 29 tools for psychological assessment. Of these, the Dermatology Life Quality Index (DLQI) and the Hair-Specific Skindex-29 were used to assess quality of life, and the Center for Epidemiologic Studies Depression Scale (CES-D) was used for psychological assessment in the meta-analysis.
Overall, 27 studies identified 18 factors associated with HRQOL; those with an inverse effect were higher self-rated hair loss severity, lower VAS score, and higher educational level. Of note, neither physician-rated hair loss severity nor treatment response were factors in HRQOL, the researchers said.
The pooled DLQI score across studies was 8.16, and subgroup analysis showed no differences in HRQOL between men and women or between patients from European vs. Asian countries. However, five studies showed significant differences in HRQOL between men and women when different assessment tools were used, which emphasized the need for more studies to examine the association of AGA with HRQOL by sex, the researchers said.
The meta-analysis of the Hair-Specific Skindex-29 scores showed pooled averages of 21.95 for symptom dimension, 18.52 in function dimension, and 29.22 in emotion dimension. Of these, the emotion dimension scores indicated moderate emotional impairment.
The average pooled score on the CES-D in the meta-analysis was 14.98, indicating no association between AGA and depression, the researchers said. However, “depression accounts for only a part of the emotion dimension,” they said. “Therefore, emotion dimension could be impaired even if no depressive symptoms were noted.”
The pooled DLQI scores for AGA (8.16) were higher than scores for other skin conditions including alopecia areata (6.3), contact dermatitis (7.35), and acne vulgaris (7.45), but lower than the pooled scores for vitiligo (9.11), urticaria (9.8), psoriasis (10.53), and atopic dermatitis (11.2), the researchers noted. “However, additional head-to-head studies are needed for direct comparisons of HRQOL in patients with various dermatoses,” they said.
The study findings were limited by the cross-sectional design of many of the included studies, and the limited number of assessment tools included in the analysis, the researchers noted. Other limitations were the lack of specific domain scores and the inclusion of only three studies from China, they said.
However, the results are consistent with findings from previous studies, and suggest that patients with AGA may benefit from psychological and psychosocial support, the researchers said.
Quality of life issues deserve attention
“Studies of the quality-of-life impact of various conditions are becoming more common in the medical literature,” Jamie B. MacKelfresh, MD, associate professor of dermatology, Emory University, Atlanta, said in an interview.
“Androgenetic alopecia is the most common type of hair loss in men and women,” she noted. “Hair loss can be labeled as a cosmetic concern, so it is important that providers understand the significant quality-of-life impact androgenetic alopecia has on the many people with this diagnosis,” she emphasized.
Dr. MacKelfresh, who was asked to comment on the study, said she was surprised that the subgroup analysis of the DLQI showed no significant difference between men and women. “This surprised me because a number of past studies have highlighted the relatively greater quality-of-life impact of hair loss on women compared to men,” she noted.
However, she added, “I was not surprised to see that androgenetic alopecia has a significant quality-of-life impact on many patients, and that physician objective assessments of the hair loss do not always correlate with the amount of quality-of-life impact,” said Dr. MacKelfresh. “In the patients I see, I find hair loss very often has a significant quality-of-life impact on patients, regardless of gender, and the amount of quality-of-life impact definitely does not always correlate with the objective amount of hair loss,” she noted.
A takeaway message for clinicians is to be aware that androgenetic alopecia frequently has a significant impact on patients, “particularly in the emotional dimension,” and can affect both men and women, Dr. MacKelfresh said. “Objective assessments of hair loss severity by providers may not accurately predict the degree of quality-of-life impact a patient may experience; therefore providers should include quality-of-life questions as part of their standard evaluation of patients with androgenetic alopecia,” she said. In addition to treating the hair loss, providers can help these patients by guiding them to psychological support resources, she emphasized.
More research is needed to assess the impact of androgenetic alopecia on “men, women, and the non-binary gender population,” as well as the relationship between self-esteem and hair loss, she said. “Finally, it would be helpful to understand what interventions can best help improve androgenetic alopecia patients’ quality of life,” she noted.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. MacKelfresh had no financial conflicts to disclose.
FROM JAMA DERMATOLOGY
Pruritic welts
The patient’s history and the classic morphology of raised plaques was consistent with chronic spontaneous urticaria. Erythema may be subtle or not visible in patients with skin of color.
Individual urticarial lesions last less than 24 hours with itching and burning. Angioedema also occurs, and manifests as a deeper edema with itching, burning, and pain. Symptoms typically last for 2 to 3 days and involve the eyelids, lips, and tongue.
Chronic urticaria (CU) is defined as episodes of spontaneous wheals or angioedema lasting for more than 6 weeks and is subdivided into chronic spontaneous urticaria or inducible urticaria (if there is a known cause). Triggers include infection, emotional stressors, or medications (eg, angiotensin-converting enzyme inhibitors, nonsteroidal anti-inflammatory drugs). Autoimmunity is seen in one-third of CU patients.1
Only a limited routine-diagnostic work-up is necessary for CU and it may include a complete blood count with differential and erythrocyte sedimentation rate and C-reactive protein testing. Further diagnostic testing is based on history and may include functional autoantibodies, thyroid stimulating hormone, and thyroid autoantibodies.
The initial step in treatment involves identification and elimination of triggers. If this is not possible, first-line therapy includes second-generation antihistamines. If symptoms persist beyond 2 weeks, the dose of the antihistamine is increased up to 4-fold. Patients should be counseled that the lowest necessary dose needs to be taken habitually to prevent urticaria. If symptoms persist, omalizumab, a recombinant humanized IgG monoclonal antibody against serum IgE, cyclosporine, or montelukast, may be added. Leukotriene-receptor antagonists such as montelukast were previously second-line therapy, but they have had mixed results in randomized controlled trials. With a much lower cost than omalizumab, leukotriene-receptor antagonists are a reasonable alternative for patients who are refractory to antihistamines.1
This patient was given high-dose nonsedating antihistamines; she experienced sedation and inadequate relief. A prior authorization was successfully obtained, and she was prescribed omalizumab.
Image courtesy of Kriti Mishra, MD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kriti Mishra, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Antia C, Baquerizo K, Korman A, et al. Urticaria: a comprehensive review: treatment of chronic urticaria, special populations, and disease outcomes. J Am Acad Dermatol. 2018;79:617-633. doi: 10.1016/j.jaad.2018.01.023

The patient’s history and the classic morphology of raised plaques was consistent with chronic spontaneous urticaria. Erythema may be subtle or not visible in patients with skin of color.
Individual urticarial lesions last less than 24 hours with itching and burning. Angioedema also occurs, and manifests as a deeper edema with itching, burning, and pain. Symptoms typically last for 2 to 3 days and involve the eyelids, lips, and tongue.
Chronic urticaria (CU) is defined as episodes of spontaneous wheals or angioedema lasting for more than 6 weeks and is subdivided into chronic spontaneous urticaria or inducible urticaria (if there is a known cause). Triggers include infection, emotional stressors, or medications (eg, angiotensin-converting enzyme inhibitors, nonsteroidal anti-inflammatory drugs). Autoimmunity is seen in one-third of CU patients.1
Only a limited routine-diagnostic work-up is necessary for CU and it may include a complete blood count with differential and erythrocyte sedimentation rate and C-reactive protein testing. Further diagnostic testing is based on history and may include functional autoantibodies, thyroid stimulating hormone, and thyroid autoantibodies.
The initial step in treatment involves identification and elimination of triggers. If this is not possible, first-line therapy includes second-generation antihistamines. If symptoms persist beyond 2 weeks, the dose of the antihistamine is increased up to 4-fold. Patients should be counseled that the lowest necessary dose needs to be taken habitually to prevent urticaria. If symptoms persist, omalizumab, a recombinant humanized IgG monoclonal antibody against serum IgE, cyclosporine, or montelukast, may be added. Leukotriene-receptor antagonists such as montelukast were previously second-line therapy, but they have had mixed results in randomized controlled trials. With a much lower cost than omalizumab, leukotriene-receptor antagonists are a reasonable alternative for patients who are refractory to antihistamines.1
This patient was given high-dose nonsedating antihistamines; she experienced sedation and inadequate relief. A prior authorization was successfully obtained, and she was prescribed omalizumab.
Image courtesy of Kriti Mishra, MD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kriti Mishra, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The patient’s history and the classic morphology of raised plaques was consistent with chronic spontaneous urticaria. Erythema may be subtle or not visible in patients with skin of color.
Individual urticarial lesions last less than 24 hours with itching and burning. Angioedema also occurs, and manifests as a deeper edema with itching, burning, and pain. Symptoms typically last for 2 to 3 days and involve the eyelids, lips, and tongue.
Chronic urticaria (CU) is defined as episodes of spontaneous wheals or angioedema lasting for more than 6 weeks and is subdivided into chronic spontaneous urticaria or inducible urticaria (if there is a known cause). Triggers include infection, emotional stressors, or medications (eg, angiotensin-converting enzyme inhibitors, nonsteroidal anti-inflammatory drugs). Autoimmunity is seen in one-third of CU patients.1
Only a limited routine-diagnostic work-up is necessary for CU and it may include a complete blood count with differential and erythrocyte sedimentation rate and C-reactive protein testing. Further diagnostic testing is based on history and may include functional autoantibodies, thyroid stimulating hormone, and thyroid autoantibodies.
The initial step in treatment involves identification and elimination of triggers. If this is not possible, first-line therapy includes second-generation antihistamines. If symptoms persist beyond 2 weeks, the dose of the antihistamine is increased up to 4-fold. Patients should be counseled that the lowest necessary dose needs to be taken habitually to prevent urticaria. If symptoms persist, omalizumab, a recombinant humanized IgG monoclonal antibody against serum IgE, cyclosporine, or montelukast, may be added. Leukotriene-receptor antagonists such as montelukast were previously second-line therapy, but they have had mixed results in randomized controlled trials. With a much lower cost than omalizumab, leukotriene-receptor antagonists are a reasonable alternative for patients who are refractory to antihistamines.1
This patient was given high-dose nonsedating antihistamines; she experienced sedation and inadequate relief. A prior authorization was successfully obtained, and she was prescribed omalizumab.
Image courtesy of Kriti Mishra, MD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kriti Mishra, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Antia C, Baquerizo K, Korman A, et al. Urticaria: a comprehensive review: treatment of chronic urticaria, special populations, and disease outcomes. J Am Acad Dermatol. 2018;79:617-633. doi: 10.1016/j.jaad.2018.01.023
1. Antia C, Baquerizo K, Korman A, et al. Urticaria: a comprehensive review: treatment of chronic urticaria, special populations, and disease outcomes. J Am Acad Dermatol. 2018;79:617-633. doi: 10.1016/j.jaad.2018.01.023
Study highlights impact of acne in adult women on quality of life, mental health
results from a qualitative study demonstrated.
“Nearly 50% of women experience acne in their 20s, and 35% experience acne in their 30s,” the study’s corresponding author, John S. Barbieri, MD, MBA, formerly of the department of dermatology at the University of Pennsylvania, Philadelphia, told this news organization. “While several qualitative studies have examined acne in adolescence, the lived experience of adult female acne has not been explored in detail and prior studies have included relatively few patients. As a result, we conducted a series of semistructured interviews among adult women with acne to examine the lived experience of adult acne and its treatment.”
For the study, published online July 28, 2021, in JAMA Dermatology, Dr. Barbieri and colleagues conducted voluntary, confidential phone interviews with 50 women aged between 18 and 40 years with moderate to severe acne who were recruited from the University of Pennsylvania Health System and from a private dermatology clinic in Cincinnati. They used free listing and open-ended, semistructured interviews to elicit opinions from the women on how acne affected their lives; their experience with acne treatments, dermatologists, and health care systems; as well as their views on treatment success.
The mean age of the participants was 28 years and 48% were white (10% were Black, 8% were Asian, 4% were more than one race, and the rest abstained from answering this question; 10% said they were Hispanic).
More than three-quarters (78%) reported prior treatment with topical retinoids, followed by spironolactone (70%), topical antibiotics (43%), combined oral contraceptives (43%), and isotretinoin (41%). During the free-listing part of interviews, where the women reported the first words that came to their mind when asked about success of treatment and adverse effects, the most important terms expressed related to treatment success were clear skin, no scarring, and no acne. The most important terms related to treatment adverse effects were dryness, redness, and burning.
In the semistructured interview portion of the study, the main themes expressed were acne-related concerns about appearance, including feeling less confident at work; mental and emotional health, including feelings of depression, anxiety, depression, and low self-worth during acne breakouts; and everyday life impact, including the notion that acne affected how other people perceived them. The other main themes included successful treatment, with clear skin and having a manageable number of lesions being desirable outcomes; and interactions with health care, including varied experiences with dermatologists. The researchers observed that most participants did not think oral antibiotics were appropriate treatments for their acne, specifically because of limited long-term effectiveness.
“Many patients described frustration with finding a dermatologist with whom they were comfortable and with identifying effective treatments for their acne,” the authors wrote. “In contrast, those who thought their dermatologist listened to their concerns and individualized their treatment plan reported higher levels of satisfaction.”
In an interview, Dr. Barbieri, who is now with the department of dermatology at Brigham and Women’s Hospital, Boston, said that he was surprised by how many patients expressed interest in nonantibiotic treatments for acne, “given that oral antibiotics are by far the most commonly prescribed systemic treatment for acne.”
Moreover, he added, “although I have experienced many patients being hesitant about isotretinoin, I was surprised by how strong patients’ concerns were about isotretinoin side effects. Unfortunately, there are many misconceptions about isotretinoin that limit use of this treatment that can be highly effective and safe for the appropriate patient.”
In an accompanying editorial, dermatologists Diane M. Thiboutot, MD and Andrea L. Zaenglein, MD, with Penn State University, Hershey, and Alison M. Layton, MB, ChB, with the Harrogate Foundation Trust, Harrogate, North Yorkshire, England, wrote that the findings from the study “resonate with those recently reported in several international studies that examine the impacts of acne, how patients assess treatment success, and what is important to measure from a patient and health care professional perspective in a clinical trial for acne.”
A large systematic review on the impact of acne on patients, conducted by the Acne Core Outcomes Research Network (ACORN), found that “appearance-related concerns and negative psychosocial effects were found to be a major impact of acne,” they noted. “Surprisingly, only 22 of the 473 studies identified in this review included qualitative data gathered from patient interviews. It is encouraging to see the concordance between the concerns voiced by the participants in the current study and those identified from the literature review, wherein a variety of methods were used to assess acne impacts.”
For his part, Dr. Barbieri said that the study findings “justify the importance of having a discussion with patients about their unique lived experience of acne and individualizing treatment to their specific needs. Patient reported outcome measures could be a useful adjunctive tool to capture these impacts on quality of life.”
This study was funded by grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Barbieri disclosed that he received partial salary support through a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Thiboutot reported receiving consultant fees from Galderma and Novartis outside the submitted work. Dr. Layton reported receiving unrestricted educational presentation, advisory board, and consultancy fees from Galderma Honoraria; unrestricted educational presentation and advisory board honoraria from Leo; advisory board honoraria from Novartis and Mylan; consultancy honoraria from Procter and Gamble and Meda; grants from Galderma; and consultancy and advisory board honoraria from Origimm outside the submitted work.
results from a qualitative study demonstrated.
“Nearly 50% of women experience acne in their 20s, and 35% experience acne in their 30s,” the study’s corresponding author, John S. Barbieri, MD, MBA, formerly of the department of dermatology at the University of Pennsylvania, Philadelphia, told this news organization. “While several qualitative studies have examined acne in adolescence, the lived experience of adult female acne has not been explored in detail and prior studies have included relatively few patients. As a result, we conducted a series of semistructured interviews among adult women with acne to examine the lived experience of adult acne and its treatment.”
For the study, published online July 28, 2021, in JAMA Dermatology, Dr. Barbieri and colleagues conducted voluntary, confidential phone interviews with 50 women aged between 18 and 40 years with moderate to severe acne who were recruited from the University of Pennsylvania Health System and from a private dermatology clinic in Cincinnati. They used free listing and open-ended, semistructured interviews to elicit opinions from the women on how acne affected their lives; their experience with acne treatments, dermatologists, and health care systems; as well as their views on treatment success.
The mean age of the participants was 28 years and 48% were white (10% were Black, 8% were Asian, 4% were more than one race, and the rest abstained from answering this question; 10% said they were Hispanic).
More than three-quarters (78%) reported prior treatment with topical retinoids, followed by spironolactone (70%), topical antibiotics (43%), combined oral contraceptives (43%), and isotretinoin (41%). During the free-listing part of interviews, where the women reported the first words that came to their mind when asked about success of treatment and adverse effects, the most important terms expressed related to treatment success were clear skin, no scarring, and no acne. The most important terms related to treatment adverse effects were dryness, redness, and burning.
In the semistructured interview portion of the study, the main themes expressed were acne-related concerns about appearance, including feeling less confident at work; mental and emotional health, including feelings of depression, anxiety, depression, and low self-worth during acne breakouts; and everyday life impact, including the notion that acne affected how other people perceived them. The other main themes included successful treatment, with clear skin and having a manageable number of lesions being desirable outcomes; and interactions with health care, including varied experiences with dermatologists. The researchers observed that most participants did not think oral antibiotics were appropriate treatments for their acne, specifically because of limited long-term effectiveness.
“Many patients described frustration with finding a dermatologist with whom they were comfortable and with identifying effective treatments for their acne,” the authors wrote. “In contrast, those who thought their dermatologist listened to their concerns and individualized their treatment plan reported higher levels of satisfaction.”
In an interview, Dr. Barbieri, who is now with the department of dermatology at Brigham and Women’s Hospital, Boston, said that he was surprised by how many patients expressed interest in nonantibiotic treatments for acne, “given that oral antibiotics are by far the most commonly prescribed systemic treatment for acne.”
Moreover, he added, “although I have experienced many patients being hesitant about isotretinoin, I was surprised by how strong patients’ concerns were about isotretinoin side effects. Unfortunately, there are many misconceptions about isotretinoin that limit use of this treatment that can be highly effective and safe for the appropriate patient.”
In an accompanying editorial, dermatologists Diane M. Thiboutot, MD and Andrea L. Zaenglein, MD, with Penn State University, Hershey, and Alison M. Layton, MB, ChB, with the Harrogate Foundation Trust, Harrogate, North Yorkshire, England, wrote that the findings from the study “resonate with those recently reported in several international studies that examine the impacts of acne, how patients assess treatment success, and what is important to measure from a patient and health care professional perspective in a clinical trial for acne.”
A large systematic review on the impact of acne on patients, conducted by the Acne Core Outcomes Research Network (ACORN), found that “appearance-related concerns and negative psychosocial effects were found to be a major impact of acne,” they noted. “Surprisingly, only 22 of the 473 studies identified in this review included qualitative data gathered from patient interviews. It is encouraging to see the concordance between the concerns voiced by the participants in the current study and those identified from the literature review, wherein a variety of methods were used to assess acne impacts.”
For his part, Dr. Barbieri said that the study findings “justify the importance of having a discussion with patients about their unique lived experience of acne and individualizing treatment to their specific needs. Patient reported outcome measures could be a useful adjunctive tool to capture these impacts on quality of life.”
This study was funded by grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Barbieri disclosed that he received partial salary support through a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Thiboutot reported receiving consultant fees from Galderma and Novartis outside the submitted work. Dr. Layton reported receiving unrestricted educational presentation, advisory board, and consultancy fees from Galderma Honoraria; unrestricted educational presentation and advisory board honoraria from Leo; advisory board honoraria from Novartis and Mylan; consultancy honoraria from Procter and Gamble and Meda; grants from Galderma; and consultancy and advisory board honoraria from Origimm outside the submitted work.
results from a qualitative study demonstrated.
“Nearly 50% of women experience acne in their 20s, and 35% experience acne in their 30s,” the study’s corresponding author, John S. Barbieri, MD, MBA, formerly of the department of dermatology at the University of Pennsylvania, Philadelphia, told this news organization. “While several qualitative studies have examined acne in adolescence, the lived experience of adult female acne has not been explored in detail and prior studies have included relatively few patients. As a result, we conducted a series of semistructured interviews among adult women with acne to examine the lived experience of adult acne and its treatment.”
For the study, published online July 28, 2021, in JAMA Dermatology, Dr. Barbieri and colleagues conducted voluntary, confidential phone interviews with 50 women aged between 18 and 40 years with moderate to severe acne who were recruited from the University of Pennsylvania Health System and from a private dermatology clinic in Cincinnati. They used free listing and open-ended, semistructured interviews to elicit opinions from the women on how acne affected their lives; their experience with acne treatments, dermatologists, and health care systems; as well as their views on treatment success.
The mean age of the participants was 28 years and 48% were white (10% were Black, 8% were Asian, 4% were more than one race, and the rest abstained from answering this question; 10% said they were Hispanic).
More than three-quarters (78%) reported prior treatment with topical retinoids, followed by spironolactone (70%), topical antibiotics (43%), combined oral contraceptives (43%), and isotretinoin (41%). During the free-listing part of interviews, where the women reported the first words that came to their mind when asked about success of treatment and adverse effects, the most important terms expressed related to treatment success were clear skin, no scarring, and no acne. The most important terms related to treatment adverse effects were dryness, redness, and burning.
In the semistructured interview portion of the study, the main themes expressed were acne-related concerns about appearance, including feeling less confident at work; mental and emotional health, including feelings of depression, anxiety, depression, and low self-worth during acne breakouts; and everyday life impact, including the notion that acne affected how other people perceived them. The other main themes included successful treatment, with clear skin and having a manageable number of lesions being desirable outcomes; and interactions with health care, including varied experiences with dermatologists. The researchers observed that most participants did not think oral antibiotics were appropriate treatments for their acne, specifically because of limited long-term effectiveness.
“Many patients described frustration with finding a dermatologist with whom they were comfortable and with identifying effective treatments for their acne,” the authors wrote. “In contrast, those who thought their dermatologist listened to their concerns and individualized their treatment plan reported higher levels of satisfaction.”
In an interview, Dr. Barbieri, who is now with the department of dermatology at Brigham and Women’s Hospital, Boston, said that he was surprised by how many patients expressed interest in nonantibiotic treatments for acne, “given that oral antibiotics are by far the most commonly prescribed systemic treatment for acne.”
Moreover, he added, “although I have experienced many patients being hesitant about isotretinoin, I was surprised by how strong patients’ concerns were about isotretinoin side effects. Unfortunately, there are many misconceptions about isotretinoin that limit use of this treatment that can be highly effective and safe for the appropriate patient.”
In an accompanying editorial, dermatologists Diane M. Thiboutot, MD and Andrea L. Zaenglein, MD, with Penn State University, Hershey, and Alison M. Layton, MB, ChB, with the Harrogate Foundation Trust, Harrogate, North Yorkshire, England, wrote that the findings from the study “resonate with those recently reported in several international studies that examine the impacts of acne, how patients assess treatment success, and what is important to measure from a patient and health care professional perspective in a clinical trial for acne.”
A large systematic review on the impact of acne on patients, conducted by the Acne Core Outcomes Research Network (ACORN), found that “appearance-related concerns and negative psychosocial effects were found to be a major impact of acne,” they noted. “Surprisingly, only 22 of the 473 studies identified in this review included qualitative data gathered from patient interviews. It is encouraging to see the concordance between the concerns voiced by the participants in the current study and those identified from the literature review, wherein a variety of methods were used to assess acne impacts.”
For his part, Dr. Barbieri said that the study findings “justify the importance of having a discussion with patients about their unique lived experience of acne and individualizing treatment to their specific needs. Patient reported outcome measures could be a useful adjunctive tool to capture these impacts on quality of life.”
This study was funded by grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Barbieri disclosed that he received partial salary support through a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Thiboutot reported receiving consultant fees from Galderma and Novartis outside the submitted work. Dr. Layton reported receiving unrestricted educational presentation, advisory board, and consultancy fees from Galderma Honoraria; unrestricted educational presentation and advisory board honoraria from Leo; advisory board honoraria from Novartis and Mylan; consultancy honoraria from Procter and Gamble and Meda; grants from Galderma; and consultancy and advisory board honoraria from Origimm outside the submitted work.
FROM JAMA DERMATOLOGY
Leg rash
The appearance and location of this rash are classic signs for necrobiosis lipoidica, a chronic granulomatous skin disease commonly associated with diabetes. The patient’s initial hemoglobin A1c was 12.4%, confirming a diagnosis of type 2 diabetes, and a punch biopsy of the lesion demonstrated a broad zone of necrobiosis in the mid to lower dermis and a chronic inflammatory infiltrate, including plasma cells.
Necrobiosis lipoidica is rare, typically affects middle-aged adults, and is more common in women than in men.1 Although commonly associated with diabetes (hence the historical name necrobiosis lipoidica diabeticorum), a significant number of cases occur in patients without diabetes.2 The pathogenesis is not fully understood. The condition first appears as asymptomatic yellow to red-brown papules and plaques, most commonly on the anterior legs. The lesions then flatten over time, forming broad, yellow-pink patches and plaques.1,2
Generally, the diagnosis can be made clinically but, if uncertain, a punch biopsy is the preferred technique for confirmation. The differential diagnosis includes chronic cutaneous lupus erythematosus (LE), sarcoidosis, and dermatophytosis. Although the appearance of lesions associated with LE or sarcoidosis can vary, neither one manifests with the yellow coloring seen here. Dermatophytosis typically demonstrates scale and pruritus with an active border; viewing a potassium hydroxide preparation of a skin scraping is usually sufficient to make a diagnosis.
Treatment for necrobiosis lipoidica includes counseling patients to avoid trauma to the affected areas, high-potency topical corticosteroids, and photodynamic therapy.3 Often, lesions are permanent.
For this patient’s diabetes treatment, she was prescribed metformin and insulin glargine and counseled extensively on weight loss, regular exercise, and appropriate diet adjustments. The rash was treated topically with triamcinolone 0.1% cream bid. At her 4-month follow-up, the patient’s hemoglobin A1c value had dropped to 5.4%, and the rash had become less prominent and widespread. The patient was pleased with the cosmetic outcome and declined referral to a dermatologist for further treatment.
Photo and text courtesy of Samuel Dickmann, MD, and James Medley, MD, University of Florida College of Medicine, Gainesville.
1. Hashemi DA, Brown-Joel ZO, Tkachenko E, et al. Clinical features and comorbidities of patients with necrobiosis lipoidica with or without diabetes. JAMA Dermatol. 2019;155:455-459. doi: 10.1001/jamadermatol.2018.5635
2. O’Toole EA, Kennedy U, Nolan JJ, et al. Necrobiosis lipoidica: only a minority of patients have diabetes mellitus. Br J Dermatol. 1999;140:283-286. doi: 10.1046/j.1365-2133.1999.02663.x
3. Heidenheim M, Jemec GBE. Successful treatment of necrobiosis lipoidica diabeticorum with photodynamic therapy. Arch Dermatol. 2006;142:1548-1550. doi: 10.1001/archderm.142.12.1548

The appearance and location of this rash are classic signs for necrobiosis lipoidica, a chronic granulomatous skin disease commonly associated with diabetes. The patient’s initial hemoglobin A1c was 12.4%, confirming a diagnosis of type 2 diabetes, and a punch biopsy of the lesion demonstrated a broad zone of necrobiosis in the mid to lower dermis and a chronic inflammatory infiltrate, including plasma cells.
Necrobiosis lipoidica is rare, typically affects middle-aged adults, and is more common in women than in men.1 Although commonly associated with diabetes (hence the historical name necrobiosis lipoidica diabeticorum), a significant number of cases occur in patients without diabetes.2 The pathogenesis is not fully understood. The condition first appears as asymptomatic yellow to red-brown papules and plaques, most commonly on the anterior legs. The lesions then flatten over time, forming broad, yellow-pink patches and plaques.1,2
Generally, the diagnosis can be made clinically but, if uncertain, a punch biopsy is the preferred technique for confirmation. The differential diagnosis includes chronic cutaneous lupus erythematosus (LE), sarcoidosis, and dermatophytosis. Although the appearance of lesions associated with LE or sarcoidosis can vary, neither one manifests with the yellow coloring seen here. Dermatophytosis typically demonstrates scale and pruritus with an active border; viewing a potassium hydroxide preparation of a skin scraping is usually sufficient to make a diagnosis.
Treatment for necrobiosis lipoidica includes counseling patients to avoid trauma to the affected areas, high-potency topical corticosteroids, and photodynamic therapy.3 Often, lesions are permanent.
For this patient’s diabetes treatment, she was prescribed metformin and insulin glargine and counseled extensively on weight loss, regular exercise, and appropriate diet adjustments. The rash was treated topically with triamcinolone 0.1% cream bid. At her 4-month follow-up, the patient’s hemoglobin A1c value had dropped to 5.4%, and the rash had become less prominent and widespread. The patient was pleased with the cosmetic outcome and declined referral to a dermatologist for further treatment.
Photo and text courtesy of Samuel Dickmann, MD, and James Medley, MD, University of Florida College of Medicine, Gainesville.

The appearance and location of this rash are classic signs for necrobiosis lipoidica, a chronic granulomatous skin disease commonly associated with diabetes. The patient’s initial hemoglobin A1c was 12.4%, confirming a diagnosis of type 2 diabetes, and a punch biopsy of the lesion demonstrated a broad zone of necrobiosis in the mid to lower dermis and a chronic inflammatory infiltrate, including plasma cells.
Necrobiosis lipoidica is rare, typically affects middle-aged adults, and is more common in women than in men.1 Although commonly associated with diabetes (hence the historical name necrobiosis lipoidica diabeticorum), a significant number of cases occur in patients without diabetes.2 The pathogenesis is not fully understood. The condition first appears as asymptomatic yellow to red-brown papules and plaques, most commonly on the anterior legs. The lesions then flatten over time, forming broad, yellow-pink patches and plaques.1,2
Generally, the diagnosis can be made clinically but, if uncertain, a punch biopsy is the preferred technique for confirmation. The differential diagnosis includes chronic cutaneous lupus erythematosus (LE), sarcoidosis, and dermatophytosis. Although the appearance of lesions associated with LE or sarcoidosis can vary, neither one manifests with the yellow coloring seen here. Dermatophytosis typically demonstrates scale and pruritus with an active border; viewing a potassium hydroxide preparation of a skin scraping is usually sufficient to make a diagnosis.
Treatment for necrobiosis lipoidica includes counseling patients to avoid trauma to the affected areas, high-potency topical corticosteroids, and photodynamic therapy.3 Often, lesions are permanent.
For this patient’s diabetes treatment, she was prescribed metformin and insulin glargine and counseled extensively on weight loss, regular exercise, and appropriate diet adjustments. The rash was treated topically with triamcinolone 0.1% cream bid. At her 4-month follow-up, the patient’s hemoglobin A1c value had dropped to 5.4%, and the rash had become less prominent and widespread. The patient was pleased with the cosmetic outcome and declined referral to a dermatologist for further treatment.
Photo and text courtesy of Samuel Dickmann, MD, and James Medley, MD, University of Florida College of Medicine, Gainesville.
1. Hashemi DA, Brown-Joel ZO, Tkachenko E, et al. Clinical features and comorbidities of patients with necrobiosis lipoidica with or without diabetes. JAMA Dermatol. 2019;155:455-459. doi: 10.1001/jamadermatol.2018.5635
2. O’Toole EA, Kennedy U, Nolan JJ, et al. Necrobiosis lipoidica: only a minority of patients have diabetes mellitus. Br J Dermatol. 1999;140:283-286. doi: 10.1046/j.1365-2133.1999.02663.x
3. Heidenheim M, Jemec GBE. Successful treatment of necrobiosis lipoidica diabeticorum with photodynamic therapy. Arch Dermatol. 2006;142:1548-1550. doi: 10.1001/archderm.142.12.1548
1. Hashemi DA, Brown-Joel ZO, Tkachenko E, et al. Clinical features and comorbidities of patients with necrobiosis lipoidica with or without diabetes. JAMA Dermatol. 2019;155:455-459. doi: 10.1001/jamadermatol.2018.5635
2. O’Toole EA, Kennedy U, Nolan JJ, et al. Necrobiosis lipoidica: only a minority of patients have diabetes mellitus. Br J Dermatol. 1999;140:283-286. doi: 10.1046/j.1365-2133.1999.02663.x
3. Heidenheim M, Jemec GBE. Successful treatment of necrobiosis lipoidica diabeticorum with photodynamic therapy. Arch Dermatol. 2006;142:1548-1550. doi: 10.1001/archderm.142.12.1548
When is MRI useful in the management of congenital melanocytic nevi?
When used for appropriate patients, results from a small multi-institutional study showed.
“The majority of congenital nevi are considered low risk for cutaneous and/or systemic complications,” Holly Neale said at the annual meeting of the Society for Pediatric Dermatology. “However, a subset of children born with higher-risk congenital nevi require close monitoring, as some features of congenital nevi have been associated with cutaneous melanoma, central nervous system melanoma, melanin in the brain or spine, and structural irregularities in the brain or spine. It’s important to understand which congenital nevi are considered higher risk in order to guide management and counseling decisions.”
One major management decision is to do a screening magnetic resonance image of the CNS to evaluate for neurologic involvement, said Ms. Neale, a fourth-year medical student at the University of Massachusetts, Worcester. Prior studies have shown that congenital nevi that are bigger than 20 cm, posterior axial location, and having more than one congenital nevus may predict CNS abnormalities, while recent guidelines from experts in the field suggest that any child with more than one congenital nevus at birth undergo screening MRI.
“However, guidelines are evolving, and more data is required to better understand the CNS abnormalities and patient outcomes for children with congenital nevi,” said Ms. Neale, who spent the past year as a pediatric dermatology research fellow at Massachusetts General Hospital, Boston.
To address this knowledge gap, she and colleagues at the University of Massachusetts, Massachusetts General Hospital, and Boston Children’s Hospital performed a retrospective chart review between Jan. 1, 2009, and Dec. 31, 2019, of individuals ages 18 and younger who had an MRI of the brain or spine with at least one dermatologist-diagnosed nevus as identified via key words in the medical record. Of the 909 patients screened, 46 met inclusion criteria, evenly split between males and females.
The most common location of the largest nevus was the trunk (in 41% of patients), followed by lesions that spanned multiple regions. More than one-third of patients had giant nevi (greater than 40 cm).
“The majority of images were considered nonconcerning, which includes normal, benign, or other findings such as trauma related, infectious, or orthopedic, which we did not classify as abnormal as it did not guide our study question,” Ms. Neale said. Specifically, 8% of spine images and 27% of brain images were considered “concerning,” defined as any finding that prompted further workup or monitoring, which includes findings concerning for melanin.
The most common brain finding was melanin (in eight children), and one child with brain melanin also had findings suggestive of melanin in the thoracic spine. The most common finding in spine MRIs was fatty filum (in four children), requiring intervention for tethering in only one individual. No cases of cutaneous melanoma developed during the study period, and only one patient with abnormal imaging had CNS melanoma, which was fatal.
All patients with findings suggestive of CNS melanin had more than four nevi present at birth, which is in line with current imaging screening guidelines. In addition, children with concerning imaging had higher rates of death, neurodevelopmental problems, seizures, and neurosurgery, compared with their counterparts with unremarkable imaging findings. Describing preliminary analyses, Ms. Neale said that a chi square analysis was performed to test statistical significance of these differences, “and neurosurgery was the only variable that children with concerning imaging were significantly more likely to experience, although sample size limits detection for the other variables.”
The authors concluded that MRI is a helpful tool when used in the appropriate clinical context for the management of congenital nevi. “As more children undergo imaging, we may discover more nonmelanin abnormalities,” she said.
Joseph M. Lam, MD, who was asked to comment on the study, said that the increased risk of CNS melanin in patients with larger lesions and in those with multiple lesions confirms previous reports.
“It is interesting to note that some patients with nonconcerning imaging results still had neurodevelopmental problems and seizures, albeit at a lower rate than those with concerning imaging results,” said Dr. Lam, a pediatric dermatologist at British Columbia Children’s Hospital, Vancouver. “The lack of a control group for comparison of rates of neurological sequelae, such as NDP, seizures and nonmelanin structural anomalies, limits the generalizability of the findings. However, this is a nice study that helps us understand better the CNS anomalies in CMN.”
Ms. Neale acknowledged certain limitations of the study, including the lack of a control group without CMN, the small number of patients, the potential for referral bias, and its retrospective design. Also, the proximity of the study period does not allow for chronic follow-up and detection of the development of melanoma or other problems in the future.
Ms. Neale and associates reported having no relevant financial disclosures. Dr. Lam disclosed that he has received speaker fees from Pierre Fabre.
When used for appropriate patients, results from a small multi-institutional study showed.
“The majority of congenital nevi are considered low risk for cutaneous and/or systemic complications,” Holly Neale said at the annual meeting of the Society for Pediatric Dermatology. “However, a subset of children born with higher-risk congenital nevi require close monitoring, as some features of congenital nevi have been associated with cutaneous melanoma, central nervous system melanoma, melanin in the brain or spine, and structural irregularities in the brain or spine. It’s important to understand which congenital nevi are considered higher risk in order to guide management and counseling decisions.”
One major management decision is to do a screening magnetic resonance image of the CNS to evaluate for neurologic involvement, said Ms. Neale, a fourth-year medical student at the University of Massachusetts, Worcester. Prior studies have shown that congenital nevi that are bigger than 20 cm, posterior axial location, and having more than one congenital nevus may predict CNS abnormalities, while recent guidelines from experts in the field suggest that any child with more than one congenital nevus at birth undergo screening MRI.
“However, guidelines are evolving, and more data is required to better understand the CNS abnormalities and patient outcomes for children with congenital nevi,” said Ms. Neale, who spent the past year as a pediatric dermatology research fellow at Massachusetts General Hospital, Boston.
To address this knowledge gap, she and colleagues at the University of Massachusetts, Massachusetts General Hospital, and Boston Children’s Hospital performed a retrospective chart review between Jan. 1, 2009, and Dec. 31, 2019, of individuals ages 18 and younger who had an MRI of the brain or spine with at least one dermatologist-diagnosed nevus as identified via key words in the medical record. Of the 909 patients screened, 46 met inclusion criteria, evenly split between males and females.
The most common location of the largest nevus was the trunk (in 41% of patients), followed by lesions that spanned multiple regions. More than one-third of patients had giant nevi (greater than 40 cm).
“The majority of images were considered nonconcerning, which includes normal, benign, or other findings such as trauma related, infectious, or orthopedic, which we did not classify as abnormal as it did not guide our study question,” Ms. Neale said. Specifically, 8% of spine images and 27% of brain images were considered “concerning,” defined as any finding that prompted further workup or monitoring, which includes findings concerning for melanin.
The most common brain finding was melanin (in eight children), and one child with brain melanin also had findings suggestive of melanin in the thoracic spine. The most common finding in spine MRIs was fatty filum (in four children), requiring intervention for tethering in only one individual. No cases of cutaneous melanoma developed during the study period, and only one patient with abnormal imaging had CNS melanoma, which was fatal.
All patients with findings suggestive of CNS melanin had more than four nevi present at birth, which is in line with current imaging screening guidelines. In addition, children with concerning imaging had higher rates of death, neurodevelopmental problems, seizures, and neurosurgery, compared with their counterparts with unremarkable imaging findings. Describing preliminary analyses, Ms. Neale said that a chi square analysis was performed to test statistical significance of these differences, “and neurosurgery was the only variable that children with concerning imaging were significantly more likely to experience, although sample size limits detection for the other variables.”
The authors concluded that MRI is a helpful tool when used in the appropriate clinical context for the management of congenital nevi. “As more children undergo imaging, we may discover more nonmelanin abnormalities,” she said.
Joseph M. Lam, MD, who was asked to comment on the study, said that the increased risk of CNS melanin in patients with larger lesions and in those with multiple lesions confirms previous reports.
“It is interesting to note that some patients with nonconcerning imaging results still had neurodevelopmental problems and seizures, albeit at a lower rate than those with concerning imaging results,” said Dr. Lam, a pediatric dermatologist at British Columbia Children’s Hospital, Vancouver. “The lack of a control group for comparison of rates of neurological sequelae, such as NDP, seizures and nonmelanin structural anomalies, limits the generalizability of the findings. However, this is a nice study that helps us understand better the CNS anomalies in CMN.”
Ms. Neale acknowledged certain limitations of the study, including the lack of a control group without CMN, the small number of patients, the potential for referral bias, and its retrospective design. Also, the proximity of the study period does not allow for chronic follow-up and detection of the development of melanoma or other problems in the future.
Ms. Neale and associates reported having no relevant financial disclosures. Dr. Lam disclosed that he has received speaker fees from Pierre Fabre.
When used for appropriate patients, results from a small multi-institutional study showed.
“The majority of congenital nevi are considered low risk for cutaneous and/or systemic complications,” Holly Neale said at the annual meeting of the Society for Pediatric Dermatology. “However, a subset of children born with higher-risk congenital nevi require close monitoring, as some features of congenital nevi have been associated with cutaneous melanoma, central nervous system melanoma, melanin in the brain or spine, and structural irregularities in the brain or spine. It’s important to understand which congenital nevi are considered higher risk in order to guide management and counseling decisions.”
One major management decision is to do a screening magnetic resonance image of the CNS to evaluate for neurologic involvement, said Ms. Neale, a fourth-year medical student at the University of Massachusetts, Worcester. Prior studies have shown that congenital nevi that are bigger than 20 cm, posterior axial location, and having more than one congenital nevus may predict CNS abnormalities, while recent guidelines from experts in the field suggest that any child with more than one congenital nevus at birth undergo screening MRI.
“However, guidelines are evolving, and more data is required to better understand the CNS abnormalities and patient outcomes for children with congenital nevi,” said Ms. Neale, who spent the past year as a pediatric dermatology research fellow at Massachusetts General Hospital, Boston.
To address this knowledge gap, she and colleagues at the University of Massachusetts, Massachusetts General Hospital, and Boston Children’s Hospital performed a retrospective chart review between Jan. 1, 2009, and Dec. 31, 2019, of individuals ages 18 and younger who had an MRI of the brain or spine with at least one dermatologist-diagnosed nevus as identified via key words in the medical record. Of the 909 patients screened, 46 met inclusion criteria, evenly split between males and females.
The most common location of the largest nevus was the trunk (in 41% of patients), followed by lesions that spanned multiple regions. More than one-third of patients had giant nevi (greater than 40 cm).
“The majority of images were considered nonconcerning, which includes normal, benign, or other findings such as trauma related, infectious, or orthopedic, which we did not classify as abnormal as it did not guide our study question,” Ms. Neale said. Specifically, 8% of spine images and 27% of brain images were considered “concerning,” defined as any finding that prompted further workup or monitoring, which includes findings concerning for melanin.
The most common brain finding was melanin (in eight children), and one child with brain melanin also had findings suggestive of melanin in the thoracic spine. The most common finding in spine MRIs was fatty filum (in four children), requiring intervention for tethering in only one individual. No cases of cutaneous melanoma developed during the study period, and only one patient with abnormal imaging had CNS melanoma, which was fatal.
All patients with findings suggestive of CNS melanin had more than four nevi present at birth, which is in line with current imaging screening guidelines. In addition, children with concerning imaging had higher rates of death, neurodevelopmental problems, seizures, and neurosurgery, compared with their counterparts with unremarkable imaging findings. Describing preliminary analyses, Ms. Neale said that a chi square analysis was performed to test statistical significance of these differences, “and neurosurgery was the only variable that children with concerning imaging were significantly more likely to experience, although sample size limits detection for the other variables.”
The authors concluded that MRI is a helpful tool when used in the appropriate clinical context for the management of congenital nevi. “As more children undergo imaging, we may discover more nonmelanin abnormalities,” she said.
Joseph M. Lam, MD, who was asked to comment on the study, said that the increased risk of CNS melanin in patients with larger lesions and in those with multiple lesions confirms previous reports.
“It is interesting to note that some patients with nonconcerning imaging results still had neurodevelopmental problems and seizures, albeit at a lower rate than those with concerning imaging results,” said Dr. Lam, a pediatric dermatologist at British Columbia Children’s Hospital, Vancouver. “The lack of a control group for comparison of rates of neurological sequelae, such as NDP, seizures and nonmelanin structural anomalies, limits the generalizability of the findings. However, this is a nice study that helps us understand better the CNS anomalies in CMN.”
Ms. Neale acknowledged certain limitations of the study, including the lack of a control group without CMN, the small number of patients, the potential for referral bias, and its retrospective design. Also, the proximity of the study period does not allow for chronic follow-up and detection of the development of melanoma or other problems in the future.
Ms. Neale and associates reported having no relevant financial disclosures. Dr. Lam disclosed that he has received speaker fees from Pierre Fabre.
FROM SPD 2021