User login
More eczema in children exposed to toxic metals in utero
published Oct. 27, 2021, in JAMA Network Open.
In this multicenter cohort study, led by epidemiologist Shu-Li Wang, PhD, of the National Institute of Environmental Health Sciences, in Taiwan, each twofold increase in prenatal arsenic level correlated with a 2.4-fold higher rate of atopic dermatitis in 4-year-olds.
Atopic diseases have been on the rise. Eczema (atopic dermatitis) is the first stage of the so-called atopic march, followed by food allergies, allergic rhinitis, and asthma later in childhood. Previous research has linked heavy metal exposure to allergic diseases in adults. In another study by Dr. Wang and colleagues that was published in 2021, prenatal and early-life arsenic exposure was found to correlate with higher rates of allergic rhinitis and asthma in children. In that study, the participants were followed every 2-3 years through the age of 14 as part of the Taiwan Maternal and Infant Cohort Study.
The new study included 370 mother and child pairs who were enrolled in that birth cohort study between October 2012 and May 2015. During their third trimester of pregnancy, women completed questionnaires about their lifestyle, diet, and living environment. In addition, their height, weight, and blood pressure were recorded, and urine samples were taken. In follow-up interviews 3-4 years later, the mothers were asked whether their child had ever been diagnosed with atopic dermatitis.
The researchers used an inductively coupled plasma mass spectrometer to analyze the participants’ urine samples. They assessed for exposures in utero to eight metals: arsenic, cadmium, lead, cobalt, copper, nickel, thallium, and zinc.
Each unit increase of an index that estimates the combined exposure to these metals during pregnancy was associated with 63% higher odds of atopic dermatitis in the children by age 4. The researchers adjusted for parental allergies (yes or no), mother’s educational level (<12 years, 13-16 years, or >16 years), geographic area (central or eastern Taiwan), exposure to tobacco smoke during pregnancy, and the child’s gender. Arsenic (40.1%) and cadmium (20.5%) accounted for most of the metal coexposure index.
A wealth of previous research links arsenic exposure during adulthood to skin disease and immune dysfunction. Early-life arsenic exposure has been linked with elevated risk for various adult disorders, including cancer, diabetes, and heart disease, years later. In light of such research, “the findings in this paper are not surprising,” J. Christopher States, PhD, director of the Center for Integrative Environmental Health Science at the University of Louisville (Ky.), told this news organization. “Low-level arsenic exposure does not cause disease immediately, but it does appear to have long-lasting effects, making individuals susceptible to ‘second hits’ with another environmental agent.”
Research into the molecular mechanisms for these links has shown that arsenic and cadmium exposure can promote allergic phenotypes in immune cells. “We think the toxic metals activate the alarmin pathway, thus inducing innate lymphoid cells, then activating T-helper 2 cells, which drive immunoglobulin E production and breakdown of the epithelium and promotion of allergies,” said Kari Nadeau, MD, PhD, director of the Sean N. Parker Center for Allergy and Asthma Research at Stanford University. Dr. Nadeau led that study, published in 2017 in PLOS One, along with epidemiologist Margaret Karagas, PhD, of Geisel School of Medicine at Dartmouth, Hanover, N.H.
As for what pregnant women can do to minimize their exposure to heavy metals, “that is a difficult problem and primarily a function of where one lives,” said Dr. States.
Drinking water and food are major sources of arsenic exposure. Groundwater is naturally contaminated with arsenic deposits that seep in from bedrock, said Dr. States. The U.S. Environmental Protection Agency regulates arsenic levels in public drinking water that is supplied to more than a few thousand people. However, small water supplies and private wells are unregulated, he said, and having these water sources tested for arsenic or fitted with systems to reduce arsenic can be very expensive.
Among foods, rice can have high concentrations of arsenic, Dr. Karagas told this news organization. To minimize arsenic exposure through the diet, women can limit rice-based foods, according to a web-based tool developed by her and coworkers.
In addition, tobacco smoke is a major source of cadmium exposure and a moderate source of arsenic exposure, Dr. States noted. Women can reduce their exposure to these metals by avoiding tobacco and secondhand smoke.
The study was supported by grants from the National Health Research Institutes, Chung Shan Medical University Hospital, Taiwan Ministry of Science and Technology, and the Taiwan Environmental Protection Administration. The authors and quoted experts report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published Oct. 27, 2021, in JAMA Network Open.
In this multicenter cohort study, led by epidemiologist Shu-Li Wang, PhD, of the National Institute of Environmental Health Sciences, in Taiwan, each twofold increase in prenatal arsenic level correlated with a 2.4-fold higher rate of atopic dermatitis in 4-year-olds.
Atopic diseases have been on the rise. Eczema (atopic dermatitis) is the first stage of the so-called atopic march, followed by food allergies, allergic rhinitis, and asthma later in childhood. Previous research has linked heavy metal exposure to allergic diseases in adults. In another study by Dr. Wang and colleagues that was published in 2021, prenatal and early-life arsenic exposure was found to correlate with higher rates of allergic rhinitis and asthma in children. In that study, the participants were followed every 2-3 years through the age of 14 as part of the Taiwan Maternal and Infant Cohort Study.
The new study included 370 mother and child pairs who were enrolled in that birth cohort study between October 2012 and May 2015. During their third trimester of pregnancy, women completed questionnaires about their lifestyle, diet, and living environment. In addition, their height, weight, and blood pressure were recorded, and urine samples were taken. In follow-up interviews 3-4 years later, the mothers were asked whether their child had ever been diagnosed with atopic dermatitis.
The researchers used an inductively coupled plasma mass spectrometer to analyze the participants’ urine samples. They assessed for exposures in utero to eight metals: arsenic, cadmium, lead, cobalt, copper, nickel, thallium, and zinc.
Each unit increase of an index that estimates the combined exposure to these metals during pregnancy was associated with 63% higher odds of atopic dermatitis in the children by age 4. The researchers adjusted for parental allergies (yes or no), mother’s educational level (<12 years, 13-16 years, or >16 years), geographic area (central or eastern Taiwan), exposure to tobacco smoke during pregnancy, and the child’s gender. Arsenic (40.1%) and cadmium (20.5%) accounted for most of the metal coexposure index.
A wealth of previous research links arsenic exposure during adulthood to skin disease and immune dysfunction. Early-life arsenic exposure has been linked with elevated risk for various adult disorders, including cancer, diabetes, and heart disease, years later. In light of such research, “the findings in this paper are not surprising,” J. Christopher States, PhD, director of the Center for Integrative Environmental Health Science at the University of Louisville (Ky.), told this news organization. “Low-level arsenic exposure does not cause disease immediately, but it does appear to have long-lasting effects, making individuals susceptible to ‘second hits’ with another environmental agent.”
Research into the molecular mechanisms for these links has shown that arsenic and cadmium exposure can promote allergic phenotypes in immune cells. “We think the toxic metals activate the alarmin pathway, thus inducing innate lymphoid cells, then activating T-helper 2 cells, which drive immunoglobulin E production and breakdown of the epithelium and promotion of allergies,” said Kari Nadeau, MD, PhD, director of the Sean N. Parker Center for Allergy and Asthma Research at Stanford University. Dr. Nadeau led that study, published in 2017 in PLOS One, along with epidemiologist Margaret Karagas, PhD, of Geisel School of Medicine at Dartmouth, Hanover, N.H.
As for what pregnant women can do to minimize their exposure to heavy metals, “that is a difficult problem and primarily a function of where one lives,” said Dr. States.
Drinking water and food are major sources of arsenic exposure. Groundwater is naturally contaminated with arsenic deposits that seep in from bedrock, said Dr. States. The U.S. Environmental Protection Agency regulates arsenic levels in public drinking water that is supplied to more than a few thousand people. However, small water supplies and private wells are unregulated, he said, and having these water sources tested for arsenic or fitted with systems to reduce arsenic can be very expensive.
Among foods, rice can have high concentrations of arsenic, Dr. Karagas told this news organization. To minimize arsenic exposure through the diet, women can limit rice-based foods, according to a web-based tool developed by her and coworkers.
In addition, tobacco smoke is a major source of cadmium exposure and a moderate source of arsenic exposure, Dr. States noted. Women can reduce their exposure to these metals by avoiding tobacco and secondhand smoke.
The study was supported by grants from the National Health Research Institutes, Chung Shan Medical University Hospital, Taiwan Ministry of Science and Technology, and the Taiwan Environmental Protection Administration. The authors and quoted experts report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published Oct. 27, 2021, in JAMA Network Open.
In this multicenter cohort study, led by epidemiologist Shu-Li Wang, PhD, of the National Institute of Environmental Health Sciences, in Taiwan, each twofold increase in prenatal arsenic level correlated with a 2.4-fold higher rate of atopic dermatitis in 4-year-olds.
Atopic diseases have been on the rise. Eczema (atopic dermatitis) is the first stage of the so-called atopic march, followed by food allergies, allergic rhinitis, and asthma later in childhood. Previous research has linked heavy metal exposure to allergic diseases in adults. In another study by Dr. Wang and colleagues that was published in 2021, prenatal and early-life arsenic exposure was found to correlate with higher rates of allergic rhinitis and asthma in children. In that study, the participants were followed every 2-3 years through the age of 14 as part of the Taiwan Maternal and Infant Cohort Study.
The new study included 370 mother and child pairs who were enrolled in that birth cohort study between October 2012 and May 2015. During their third trimester of pregnancy, women completed questionnaires about their lifestyle, diet, and living environment. In addition, their height, weight, and blood pressure were recorded, and urine samples were taken. In follow-up interviews 3-4 years later, the mothers were asked whether their child had ever been diagnosed with atopic dermatitis.
The researchers used an inductively coupled plasma mass spectrometer to analyze the participants’ urine samples. They assessed for exposures in utero to eight metals: arsenic, cadmium, lead, cobalt, copper, nickel, thallium, and zinc.
Each unit increase of an index that estimates the combined exposure to these metals during pregnancy was associated with 63% higher odds of atopic dermatitis in the children by age 4. The researchers adjusted for parental allergies (yes or no), mother’s educational level (<12 years, 13-16 years, or >16 years), geographic area (central or eastern Taiwan), exposure to tobacco smoke during pregnancy, and the child’s gender. Arsenic (40.1%) and cadmium (20.5%) accounted for most of the metal coexposure index.
A wealth of previous research links arsenic exposure during adulthood to skin disease and immune dysfunction. Early-life arsenic exposure has been linked with elevated risk for various adult disorders, including cancer, diabetes, and heart disease, years later. In light of such research, “the findings in this paper are not surprising,” J. Christopher States, PhD, director of the Center for Integrative Environmental Health Science at the University of Louisville (Ky.), told this news organization. “Low-level arsenic exposure does not cause disease immediately, but it does appear to have long-lasting effects, making individuals susceptible to ‘second hits’ with another environmental agent.”
Research into the molecular mechanisms for these links has shown that arsenic and cadmium exposure can promote allergic phenotypes in immune cells. “We think the toxic metals activate the alarmin pathway, thus inducing innate lymphoid cells, then activating T-helper 2 cells, which drive immunoglobulin E production and breakdown of the epithelium and promotion of allergies,” said Kari Nadeau, MD, PhD, director of the Sean N. Parker Center for Allergy and Asthma Research at Stanford University. Dr. Nadeau led that study, published in 2017 in PLOS One, along with epidemiologist Margaret Karagas, PhD, of Geisel School of Medicine at Dartmouth, Hanover, N.H.
As for what pregnant women can do to minimize their exposure to heavy metals, “that is a difficult problem and primarily a function of where one lives,” said Dr. States.
Drinking water and food are major sources of arsenic exposure. Groundwater is naturally contaminated with arsenic deposits that seep in from bedrock, said Dr. States. The U.S. Environmental Protection Agency regulates arsenic levels in public drinking water that is supplied to more than a few thousand people. However, small water supplies and private wells are unregulated, he said, and having these water sources tested for arsenic or fitted with systems to reduce arsenic can be very expensive.
Among foods, rice can have high concentrations of arsenic, Dr. Karagas told this news organization. To minimize arsenic exposure through the diet, women can limit rice-based foods, according to a web-based tool developed by her and coworkers.
In addition, tobacco smoke is a major source of cadmium exposure and a moderate source of arsenic exposure, Dr. States noted. Women can reduce their exposure to these metals by avoiding tobacco and secondhand smoke.
The study was supported by grants from the National Health Research Institutes, Chung Shan Medical University Hospital, Taiwan Ministry of Science and Technology, and the Taiwan Environmental Protection Administration. The authors and quoted experts report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Early peanut feeding guidelines still not reaching families
Four years after new infant feeding guidelines were issued to prevent allergies to peanut and other foods, 70% of surveyed parents and caregivers in the United States said they had never heard about the new recommendation.
Food allergies in developed countries have doubled in each of the last decades and now affect 7.6% of U.S. children. About 1 in 50 are allergic to peanut. Data from the 2015 LEAP study and other research has convincingly shown that early, sustained feeding of peanuts, eggs, and other allergens can prevent babies from developing allergies to these foods.
Based on those findings, the National Institute of Allergy and Infectious Diseases (NIAID) updated its feeding guidelines in 2017, urging parents to introduce these foods to babies around 4-6 months of age rather than wait until 1-3 years of age, as previously recommended. The American Academy of Pediatrics approved those guidelines too, and in 2019 changed its own feeding recommendations.
To assess awareness of this new guidance and to what extent these recommendations are being translated into clinical practice, researchers surveyed a demographically representative U.S. sample of 3,062 parents and caregivers with children between 7 months and 3½ years old. The survey was conducted in English and Spanish over the web or by phone.
More than one-third reported that their child’s primary care physician never discussed when to start feeding peanut-containing foods. And among those whose doctors did offer guidance, fewer than 1 in 4 specifically recommended introducing peanut by 6 months of age.
These data show that “despite strong evidence that early introduction of peanut within the first year of life can prevent the development of peanut allergy, this evidence is simply not making its way to parents of infants,” said Christopher Warren, PhD, assistant professor of preventive medicine at the Northwestern University Feinberg School of Medicine, Chicago. Dr. Warren led the study and presented the findings on a poster at this year’s American College of Allergy, Asthma & Immunology annual meeting in New Orleans.
In addition to caregivers, the Northwestern team surveyed U.S. allergists and pediatricians about the new feeding guidelines. Uptake was fairly good among allergists, with 65% reporting full implementation. On the other hand, while most pediatricians seemed familiar with the 2017 recommendations, fewer than one-third said they were following them.
“What’s unique about this challenge is that it’s not just a guideline change – it’s a guideline reversal,” said Wendy Sue Swanson, MD, chief medical officer for SpoonfulONE, a company that makes mix-ins and other products for multi-allergen feeding. After telling families for years to avoid these allergens in early life because food allergies were rising, “it’s harder advice to say, actually, we were wrong. Not only should you not wait, you should get peanut in while your baby’s immune system has this critical moment to learn and develop, and you should keep getting it in,” Dr. Swanson said in an interview.
Making matters worse, pediatricians are time pressed. Typically, at 4- to 6-month-old well-check visits, “they’re talking about sleep and development and feeding and milestones,” said Ruchi Gupta, MD, MPH, professor of pediatrics and medicine at Northwestern Feinberg, who led the allergist and pediatrician analyses.
Another challenge: Guidelines differ depending on the child’s level of food allergy risk, so it’s hard to explain them clearly and quickly. Babies at highest risk – as judged by having severe eczema, egg allergy, or both – should get peanut IgE blood testing and, if negative, begin regular consumption of peanut by 4-6 months. Intermediate-risk babies who have mild-to-moderate eczema are recommended to start peanut-containing foods by 6 months. And for low-risk babies with no eczema or known food allergies, the guidance is simply to introduce peanut-containing foods “in accordance with family preferences and cultural practices.”
As for pediatricians who say it’s hard to distinguish mild-to-moderate from severe eczema, “any eczema puts you at some risk,” Dr. Gupta told this news organization. “If they’ve required steroid creams to clear up their skin, or if you look at their skin, and you think it’s severe, don’t hesitate. Go ahead and draw the IgE and send them to an allergist.”
Australia, which has the highest rate of confirmed food allergy, has had more success implementing early feeding guidelines, said Dr. Swanson. Unlike the United States’ tiered approach, she said, they “had a national guideline that very crisply, years ago, told parents what to do.” Australia also has nurse educators that follow up with new moms to make sure they understand and follow the recommendations.
Dr. Gupta receives research support from the National Institutes of Health, Food Allergy Research and Education, the Melchiorre Family Foundation, the Sunshine Charitable Foundation, the Walder Foundation, the UnitedHealth Group, Thermo Fisher Scientific, and Genentech. She serves as a medical consultant/advisor for Genentech, Novartis, and Food Allergy Research and Education. Dr. Swanson serves as chief medical officer for SpoonfulONE.
A version of this article first appeared on Medscape.com.
Four years after new infant feeding guidelines were issued to prevent allergies to peanut and other foods, 70% of surveyed parents and caregivers in the United States said they had never heard about the new recommendation.
Food allergies in developed countries have doubled in each of the last decades and now affect 7.6% of U.S. children. About 1 in 50 are allergic to peanut. Data from the 2015 LEAP study and other research has convincingly shown that early, sustained feeding of peanuts, eggs, and other allergens can prevent babies from developing allergies to these foods.
Based on those findings, the National Institute of Allergy and Infectious Diseases (NIAID) updated its feeding guidelines in 2017, urging parents to introduce these foods to babies around 4-6 months of age rather than wait until 1-3 years of age, as previously recommended. The American Academy of Pediatrics approved those guidelines too, and in 2019 changed its own feeding recommendations.
To assess awareness of this new guidance and to what extent these recommendations are being translated into clinical practice, researchers surveyed a demographically representative U.S. sample of 3,062 parents and caregivers with children between 7 months and 3½ years old. The survey was conducted in English and Spanish over the web or by phone.
More than one-third reported that their child’s primary care physician never discussed when to start feeding peanut-containing foods. And among those whose doctors did offer guidance, fewer than 1 in 4 specifically recommended introducing peanut by 6 months of age.
These data show that “despite strong evidence that early introduction of peanut within the first year of life can prevent the development of peanut allergy, this evidence is simply not making its way to parents of infants,” said Christopher Warren, PhD, assistant professor of preventive medicine at the Northwestern University Feinberg School of Medicine, Chicago. Dr. Warren led the study and presented the findings on a poster at this year’s American College of Allergy, Asthma & Immunology annual meeting in New Orleans.
In addition to caregivers, the Northwestern team surveyed U.S. allergists and pediatricians about the new feeding guidelines. Uptake was fairly good among allergists, with 65% reporting full implementation. On the other hand, while most pediatricians seemed familiar with the 2017 recommendations, fewer than one-third said they were following them.
“What’s unique about this challenge is that it’s not just a guideline change – it’s a guideline reversal,” said Wendy Sue Swanson, MD, chief medical officer for SpoonfulONE, a company that makes mix-ins and other products for multi-allergen feeding. After telling families for years to avoid these allergens in early life because food allergies were rising, “it’s harder advice to say, actually, we were wrong. Not only should you not wait, you should get peanut in while your baby’s immune system has this critical moment to learn and develop, and you should keep getting it in,” Dr. Swanson said in an interview.
Making matters worse, pediatricians are time pressed. Typically, at 4- to 6-month-old well-check visits, “they’re talking about sleep and development and feeding and milestones,” said Ruchi Gupta, MD, MPH, professor of pediatrics and medicine at Northwestern Feinberg, who led the allergist and pediatrician analyses.
Another challenge: Guidelines differ depending on the child’s level of food allergy risk, so it’s hard to explain them clearly and quickly. Babies at highest risk – as judged by having severe eczema, egg allergy, or both – should get peanut IgE blood testing and, if negative, begin regular consumption of peanut by 4-6 months. Intermediate-risk babies who have mild-to-moderate eczema are recommended to start peanut-containing foods by 6 months. And for low-risk babies with no eczema or known food allergies, the guidance is simply to introduce peanut-containing foods “in accordance with family preferences and cultural practices.”
As for pediatricians who say it’s hard to distinguish mild-to-moderate from severe eczema, “any eczema puts you at some risk,” Dr. Gupta told this news organization. “If they’ve required steroid creams to clear up their skin, or if you look at their skin, and you think it’s severe, don’t hesitate. Go ahead and draw the IgE and send them to an allergist.”
Australia, which has the highest rate of confirmed food allergy, has had more success implementing early feeding guidelines, said Dr. Swanson. Unlike the United States’ tiered approach, she said, they “had a national guideline that very crisply, years ago, told parents what to do.” Australia also has nurse educators that follow up with new moms to make sure they understand and follow the recommendations.
Dr. Gupta receives research support from the National Institutes of Health, Food Allergy Research and Education, the Melchiorre Family Foundation, the Sunshine Charitable Foundation, the Walder Foundation, the UnitedHealth Group, Thermo Fisher Scientific, and Genentech. She serves as a medical consultant/advisor for Genentech, Novartis, and Food Allergy Research and Education. Dr. Swanson serves as chief medical officer for SpoonfulONE.
A version of this article first appeared on Medscape.com.
Four years after new infant feeding guidelines were issued to prevent allergies to peanut and other foods, 70% of surveyed parents and caregivers in the United States said they had never heard about the new recommendation.
Food allergies in developed countries have doubled in each of the last decades and now affect 7.6% of U.S. children. About 1 in 50 are allergic to peanut. Data from the 2015 LEAP study and other research has convincingly shown that early, sustained feeding of peanuts, eggs, and other allergens can prevent babies from developing allergies to these foods.
Based on those findings, the National Institute of Allergy and Infectious Diseases (NIAID) updated its feeding guidelines in 2017, urging parents to introduce these foods to babies around 4-6 months of age rather than wait until 1-3 years of age, as previously recommended. The American Academy of Pediatrics approved those guidelines too, and in 2019 changed its own feeding recommendations.
To assess awareness of this new guidance and to what extent these recommendations are being translated into clinical practice, researchers surveyed a demographically representative U.S. sample of 3,062 parents and caregivers with children between 7 months and 3½ years old. The survey was conducted in English and Spanish over the web or by phone.
More than one-third reported that their child’s primary care physician never discussed when to start feeding peanut-containing foods. And among those whose doctors did offer guidance, fewer than 1 in 4 specifically recommended introducing peanut by 6 months of age.
These data show that “despite strong evidence that early introduction of peanut within the first year of life can prevent the development of peanut allergy, this evidence is simply not making its way to parents of infants,” said Christopher Warren, PhD, assistant professor of preventive medicine at the Northwestern University Feinberg School of Medicine, Chicago. Dr. Warren led the study and presented the findings on a poster at this year’s American College of Allergy, Asthma & Immunology annual meeting in New Orleans.
In addition to caregivers, the Northwestern team surveyed U.S. allergists and pediatricians about the new feeding guidelines. Uptake was fairly good among allergists, with 65% reporting full implementation. On the other hand, while most pediatricians seemed familiar with the 2017 recommendations, fewer than one-third said they were following them.
“What’s unique about this challenge is that it’s not just a guideline change – it’s a guideline reversal,” said Wendy Sue Swanson, MD, chief medical officer for SpoonfulONE, a company that makes mix-ins and other products for multi-allergen feeding. After telling families for years to avoid these allergens in early life because food allergies were rising, “it’s harder advice to say, actually, we were wrong. Not only should you not wait, you should get peanut in while your baby’s immune system has this critical moment to learn and develop, and you should keep getting it in,” Dr. Swanson said in an interview.
Making matters worse, pediatricians are time pressed. Typically, at 4- to 6-month-old well-check visits, “they’re talking about sleep and development and feeding and milestones,” said Ruchi Gupta, MD, MPH, professor of pediatrics and medicine at Northwestern Feinberg, who led the allergist and pediatrician analyses.
Another challenge: Guidelines differ depending on the child’s level of food allergy risk, so it’s hard to explain them clearly and quickly. Babies at highest risk – as judged by having severe eczema, egg allergy, or both – should get peanut IgE blood testing and, if negative, begin regular consumption of peanut by 4-6 months. Intermediate-risk babies who have mild-to-moderate eczema are recommended to start peanut-containing foods by 6 months. And for low-risk babies with no eczema or known food allergies, the guidance is simply to introduce peanut-containing foods “in accordance with family preferences and cultural practices.”
As for pediatricians who say it’s hard to distinguish mild-to-moderate from severe eczema, “any eczema puts you at some risk,” Dr. Gupta told this news organization. “If they’ve required steroid creams to clear up their skin, or if you look at their skin, and you think it’s severe, don’t hesitate. Go ahead and draw the IgE and send them to an allergist.”
Australia, which has the highest rate of confirmed food allergy, has had more success implementing early feeding guidelines, said Dr. Swanson. Unlike the United States’ tiered approach, she said, they “had a national guideline that very crisply, years ago, told parents what to do.” Australia also has nurse educators that follow up with new moms to make sure they understand and follow the recommendations.
Dr. Gupta receives research support from the National Institutes of Health, Food Allergy Research and Education, the Melchiorre Family Foundation, the Sunshine Charitable Foundation, the Walder Foundation, the UnitedHealth Group, Thermo Fisher Scientific, and Genentech. She serves as a medical consultant/advisor for Genentech, Novartis, and Food Allergy Research and Education. Dr. Swanson serves as chief medical officer for SpoonfulONE.
A version of this article first appeared on Medscape.com.
Rituximab improves systemic sclerosis skin, lung symptoms
Rituximab effectively reduced skin sclerosis and appeared to have a beneficial effect on interstitial lung disease (ILD) for patients with systemic sclerosis (SSc) in a randomized, clinical trial.
At 24 weeks’ follow-up, there was significant improvement in total skin thickness scores among patients who received four once-weekly rituximab infusions, compared with patients who received placebo infusions. Among patients who received rituximab, there were also small but significant improvements in percentage of forced vital capacity (FVC). Among patients who received placebo, FVC worsened, reported Ayumi Yoshizaki, MD, of the University of Tokyo and colleagues.
“Systemic sclerosis is considered to have high unmet medical needs because of its poor prognosis and the lack of satisfactory and effective treatments,” he said at the virtual annual meeting of the American College of Rheumatology.
“Several clinical studies have suggested that B-cell depletion therapy with rituximab anti-CD20 antibody is effective in treating skin and lung fibrosis of SSc. However, no randomized, placebo-controlled trial has been able to confirm the efficacy of rituximab in SSc,” Dr. Yoshizaki said.
A rheumatologist who is currently conducting an investigator-initiated trial in which patients with SSC are undergoing treatment with rituximab followed by belimumab (Benlysta) said in an interview that he found the data to be “super interesting.”
“There are a lot of reasons to think that B cells might be important in systemic sclerosis, and actually that’s why our group had previously done an investigator-initiated trial with belimumab years ago,” said Robert Spiera, MD, director of the Scleroderma, Vasculitis, and Myositis Center at the Hospital for Special Surgery in New York.
Randomized trial
Dr. Yoshizaki and colleagues conducted the randomized, placebo-controlled DESIRES trial in four hospitals in Japan to evaluate the safety and efficacy of rituximab for the treatment of SSc.
In the investigator-initiated trial, patients aged 20-79 years who fulfilled ACR and European Alliance of Associations for Rheumatology classification criteria for systemic sclerosis and who had a modified Rodnan Skin Score (mRSS) of 10 or more and a life expectancy of at least 6 months were randomly assigned to receive infusions with either rituximab 375 mg/m2 or placebo once weekly for 4 weeks. Patients and clinicians were masked to treatment allocation.
The trial included 56 patients (51 women, 5 men). Of all patients enrolled, 27 of 28 who were allocated to receive rituximab and 22 of 28 who were allocated to receive placebo underwent at least one infusion and completed 24 weeks of follow-up.
The absolute change in mRSS at 24 weeks after the start of therapy, the primary endpoint, was –6.30 in the rituximab group, compared with +2.14 in the placebo group, a difference of –8.44 (P < .0001).
In a subgroup analysis, rituximab was superior to placebo regardless of disease duration, disease type (diffuse cutaneous or limited cutaneous SSc), prior receipt of systemic corticosteroids or immunosuppressants, or having C-reactive protein levels less than 0.3 mg/dL or at least 0.3 mg/dL.
However, there was no significant benefit with rituximab for patients with baseline mRSS of at least 20 or for those without ILD at baseline.
There was also evidence that rituximab reduced lung fibrosis. For patients assigned to the active drug, the absolute change in FVC at 24 weeks was +0.09% of the predicted value, compared with –3.56% for patients who received placebo (P = .044).
The researchers also observed radiographic evidence of lung improvement. The absolute change in the percentage of lung field occupied with interstitial shadows was –0.32% in the rituximab arm versus +2.39% in the placebo arm (P = .034). There was no significant between-group difference in the absolute change in diffusing capacity of lung for carbon monoxide, however.
Adverse events that occurred more frequently with rituximab included oral mucositis, diarrhea, and decreased neutrophil and white blood cell counts.
Convincing results
“What I thought the Japanese study did was to give a much more convincing proof of concept than has been out there,” Dr. Spiera said in an interview.
“There have been some preliminary experiences that have been encouraging with rituximab in scleroderma, most of which has been open label,” he said.
He also referred to a retrospective study by EUSTAR, the European Scleroderma Trials and Research group, which indicated that patients who had previously received rituximab seemed to have had better outcomes than patients who had been treated with other therapies.
Dr. Spiera added that, although he was glad to see the data from a randomized, placebo-controlled trial in this population, he was uncomfortable with the idea of leaving patients untreated for 6 months.
“From the standpoint of somebody wanting to know what strategies might be promising, this is great for us, but I would not have designed the trial that way,” he said.
The study results were previously published in the Lancet Rheumatology.
The study was supported by grants from the Japan Agency for Medical Research and Development and Zenyaku Kogyo. Dr. Yoshizaki disclosed no relevant financial relationships. Dr. Spiera has received grant/research support from and has consulted for Roche/Genentech, maker of rituximab, and has received compensation from other companies.
A version of this article first appeared on Medscape.com.
Rituximab effectively reduced skin sclerosis and appeared to have a beneficial effect on interstitial lung disease (ILD) for patients with systemic sclerosis (SSc) in a randomized, clinical trial.
At 24 weeks’ follow-up, there was significant improvement in total skin thickness scores among patients who received four once-weekly rituximab infusions, compared with patients who received placebo infusions. Among patients who received rituximab, there were also small but significant improvements in percentage of forced vital capacity (FVC). Among patients who received placebo, FVC worsened, reported Ayumi Yoshizaki, MD, of the University of Tokyo and colleagues.
“Systemic sclerosis is considered to have high unmet medical needs because of its poor prognosis and the lack of satisfactory and effective treatments,” he said at the virtual annual meeting of the American College of Rheumatology.
“Several clinical studies have suggested that B-cell depletion therapy with rituximab anti-CD20 antibody is effective in treating skin and lung fibrosis of SSc. However, no randomized, placebo-controlled trial has been able to confirm the efficacy of rituximab in SSc,” Dr. Yoshizaki said.
A rheumatologist who is currently conducting an investigator-initiated trial in which patients with SSC are undergoing treatment with rituximab followed by belimumab (Benlysta) said in an interview that he found the data to be “super interesting.”
“There are a lot of reasons to think that B cells might be important in systemic sclerosis, and actually that’s why our group had previously done an investigator-initiated trial with belimumab years ago,” said Robert Spiera, MD, director of the Scleroderma, Vasculitis, and Myositis Center at the Hospital for Special Surgery in New York.
Randomized trial
Dr. Yoshizaki and colleagues conducted the randomized, placebo-controlled DESIRES trial in four hospitals in Japan to evaluate the safety and efficacy of rituximab for the treatment of SSc.
In the investigator-initiated trial, patients aged 20-79 years who fulfilled ACR and European Alliance of Associations for Rheumatology classification criteria for systemic sclerosis and who had a modified Rodnan Skin Score (mRSS) of 10 or more and a life expectancy of at least 6 months were randomly assigned to receive infusions with either rituximab 375 mg/m2 or placebo once weekly for 4 weeks. Patients and clinicians were masked to treatment allocation.
The trial included 56 patients (51 women, 5 men). Of all patients enrolled, 27 of 28 who were allocated to receive rituximab and 22 of 28 who were allocated to receive placebo underwent at least one infusion and completed 24 weeks of follow-up.
The absolute change in mRSS at 24 weeks after the start of therapy, the primary endpoint, was –6.30 in the rituximab group, compared with +2.14 in the placebo group, a difference of –8.44 (P < .0001).
In a subgroup analysis, rituximab was superior to placebo regardless of disease duration, disease type (diffuse cutaneous or limited cutaneous SSc), prior receipt of systemic corticosteroids or immunosuppressants, or having C-reactive protein levels less than 0.3 mg/dL or at least 0.3 mg/dL.
However, there was no significant benefit with rituximab for patients with baseline mRSS of at least 20 or for those without ILD at baseline.
There was also evidence that rituximab reduced lung fibrosis. For patients assigned to the active drug, the absolute change in FVC at 24 weeks was +0.09% of the predicted value, compared with –3.56% for patients who received placebo (P = .044).
The researchers also observed radiographic evidence of lung improvement. The absolute change in the percentage of lung field occupied with interstitial shadows was –0.32% in the rituximab arm versus +2.39% in the placebo arm (P = .034). There was no significant between-group difference in the absolute change in diffusing capacity of lung for carbon monoxide, however.
Adverse events that occurred more frequently with rituximab included oral mucositis, diarrhea, and decreased neutrophil and white blood cell counts.
Convincing results
“What I thought the Japanese study did was to give a much more convincing proof of concept than has been out there,” Dr. Spiera said in an interview.
“There have been some preliminary experiences that have been encouraging with rituximab in scleroderma, most of which has been open label,” he said.
He also referred to a retrospective study by EUSTAR, the European Scleroderma Trials and Research group, which indicated that patients who had previously received rituximab seemed to have had better outcomes than patients who had been treated with other therapies.
Dr. Spiera added that, although he was glad to see the data from a randomized, placebo-controlled trial in this population, he was uncomfortable with the idea of leaving patients untreated for 6 months.
“From the standpoint of somebody wanting to know what strategies might be promising, this is great for us, but I would not have designed the trial that way,” he said.
The study results were previously published in the Lancet Rheumatology.
The study was supported by grants from the Japan Agency for Medical Research and Development and Zenyaku Kogyo. Dr. Yoshizaki disclosed no relevant financial relationships. Dr. Spiera has received grant/research support from and has consulted for Roche/Genentech, maker of rituximab, and has received compensation from other companies.
A version of this article first appeared on Medscape.com.
Rituximab effectively reduced skin sclerosis and appeared to have a beneficial effect on interstitial lung disease (ILD) for patients with systemic sclerosis (SSc) in a randomized, clinical trial.
At 24 weeks’ follow-up, there was significant improvement in total skin thickness scores among patients who received four once-weekly rituximab infusions, compared with patients who received placebo infusions. Among patients who received rituximab, there were also small but significant improvements in percentage of forced vital capacity (FVC). Among patients who received placebo, FVC worsened, reported Ayumi Yoshizaki, MD, of the University of Tokyo and colleagues.
“Systemic sclerosis is considered to have high unmet medical needs because of its poor prognosis and the lack of satisfactory and effective treatments,” he said at the virtual annual meeting of the American College of Rheumatology.
“Several clinical studies have suggested that B-cell depletion therapy with rituximab anti-CD20 antibody is effective in treating skin and lung fibrosis of SSc. However, no randomized, placebo-controlled trial has been able to confirm the efficacy of rituximab in SSc,” Dr. Yoshizaki said.
A rheumatologist who is currently conducting an investigator-initiated trial in which patients with SSC are undergoing treatment with rituximab followed by belimumab (Benlysta) said in an interview that he found the data to be “super interesting.”
“There are a lot of reasons to think that B cells might be important in systemic sclerosis, and actually that’s why our group had previously done an investigator-initiated trial with belimumab years ago,” said Robert Spiera, MD, director of the Scleroderma, Vasculitis, and Myositis Center at the Hospital for Special Surgery in New York.
Randomized trial
Dr. Yoshizaki and colleagues conducted the randomized, placebo-controlled DESIRES trial in four hospitals in Japan to evaluate the safety and efficacy of rituximab for the treatment of SSc.
In the investigator-initiated trial, patients aged 20-79 years who fulfilled ACR and European Alliance of Associations for Rheumatology classification criteria for systemic sclerosis and who had a modified Rodnan Skin Score (mRSS) of 10 or more and a life expectancy of at least 6 months were randomly assigned to receive infusions with either rituximab 375 mg/m2 or placebo once weekly for 4 weeks. Patients and clinicians were masked to treatment allocation.
The trial included 56 patients (51 women, 5 men). Of all patients enrolled, 27 of 28 who were allocated to receive rituximab and 22 of 28 who were allocated to receive placebo underwent at least one infusion and completed 24 weeks of follow-up.
The absolute change in mRSS at 24 weeks after the start of therapy, the primary endpoint, was –6.30 in the rituximab group, compared with +2.14 in the placebo group, a difference of –8.44 (P < .0001).
In a subgroup analysis, rituximab was superior to placebo regardless of disease duration, disease type (diffuse cutaneous or limited cutaneous SSc), prior receipt of systemic corticosteroids or immunosuppressants, or having C-reactive protein levels less than 0.3 mg/dL or at least 0.3 mg/dL.
However, there was no significant benefit with rituximab for patients with baseline mRSS of at least 20 or for those without ILD at baseline.
There was also evidence that rituximab reduced lung fibrosis. For patients assigned to the active drug, the absolute change in FVC at 24 weeks was +0.09% of the predicted value, compared with –3.56% for patients who received placebo (P = .044).
The researchers also observed radiographic evidence of lung improvement. The absolute change in the percentage of lung field occupied with interstitial shadows was –0.32% in the rituximab arm versus +2.39% in the placebo arm (P = .034). There was no significant between-group difference in the absolute change in diffusing capacity of lung for carbon monoxide, however.
Adverse events that occurred more frequently with rituximab included oral mucositis, diarrhea, and decreased neutrophil and white blood cell counts.
Convincing results
“What I thought the Japanese study did was to give a much more convincing proof of concept than has been out there,” Dr. Spiera said in an interview.
“There have been some preliminary experiences that have been encouraging with rituximab in scleroderma, most of which has been open label,” he said.
He also referred to a retrospective study by EUSTAR, the European Scleroderma Trials and Research group, which indicated that patients who had previously received rituximab seemed to have had better outcomes than patients who had been treated with other therapies.
Dr. Spiera added that, although he was glad to see the data from a randomized, placebo-controlled trial in this population, he was uncomfortable with the idea of leaving patients untreated for 6 months.
“From the standpoint of somebody wanting to know what strategies might be promising, this is great for us, but I would not have designed the trial that way,” he said.
The study results were previously published in the Lancet Rheumatology.
The study was supported by grants from the Japan Agency for Medical Research and Development and Zenyaku Kogyo. Dr. Yoshizaki disclosed no relevant financial relationships. Dr. Spiera has received grant/research support from and has consulted for Roche/Genentech, maker of rituximab, and has received compensation from other companies.
A version of this article first appeared on Medscape.com.
FROM ACR 2021
Alopecia tied to a threefold increased risk for dementia
Alopecia areata (AA) has been linked to a significantly increased risk for dementia, new research shows.
After controlling for an array of potential confounders, investigators found a threefold higher risk of developing any form of dementia and a fourfold higher risk of developing Alzheimer’s disease (AD) in those with AA versus the controls.
“AA shares a similar inflammatory signature with dementia and has great psychological impacts that lead to poor social engagement,” lead author Cheng-Yuan Li, MD, MSc, of the department of dermatology, Taipei (Taiwan) Veterans General Hospital.
“Poor social engagement and shared inflammatory cytokines might both be important links between AA and dementia,” said Dr. Li, who is also affiliated with the School of Medicine and the Institute of Brain Science at National Yang Ming Chiao Tung University, Taipei.
The study was published online Oct. 26, 2021, in the Journal of Clinical Psychiatry (doi: 10.4088/JCP.21m13931).
Significant psychological impact
Patients with AA often experience anxiety and depression, possibly caused by the negative emotional and psychological impact of the hair loss and partial or even complete baldness associated with the disease, the authors noted.
However, AA is also associated with an array of other atopic and autoimmune diseases, including psoriasis and systemic lupus erythematosus (SLE).
Epidemiologic research has suggested a link between dementia and autoimmune diseases such as psoriasis and SLE, with some evidence suggesting that autoimmune and inflammatory mechanisms may “play a role” in the development of AD.
Dementia in general and AD in particular, “have been shown to include an inflammatory component” that may share some of the same mediators seen in AA (eg, IL-1 beta, IL-6, and tumor necrosis factor–alpha).
Moreover, “the great negative psychosocial impact of AA might result in poor social engagement, a typical risk factor for dementia,” said Dr. Li. The investigators sought to investigate whether patients with AA actually do have a higher dementia risk than individuals without AA.
The researchers used data from the Taiwan National Health Insurance Research Database, comparing 2,534 patients with AA against 25,340 controls matched for age, sex, residence, income, dementia-related comorbidities, systemic steroid use, and annual outpatient visits. Participants were enrolled between 1998 and 2011 and followed to the end of 2013.
The mean age of the cohort was 53.9 years, and a little over half (57.6%) were female. The most common comorbidity was hypertension (32.3%), followed by dyslipidemia (27%) and diabetes (15.4%).
Dual intervention
After adjusting for potential confounders, those with AA were more likely to develop dementia, AD, and unspecified dementia, compared with controls. They also had a numerically higher risk for vascular dementia, compared with controls, but it was not statistically significant.
When participants were stratified by age, investigators found a significant association between AA and higher risk for any dementia as well as unspecified dementia in individuals of all ages and an increased risk for AD in patients with dementia age at onset of 65 years and older.
The mean age of dementia diagnosis was considerably younger in patients with AA versus controls (73.4 vs. 78.9 years, P = .002). The risk for any dementia and unspecified dementia was higher in patients of both sexes, but the risk for AD was higher only in male patients.
Sensitivity analyses that excluded the first year or first 3 years of observation yielded similar and consistent findings.
“Intervention targeting poor social engagement and inflammatory cytokines may be beneficial to AA-associated dementia,” said Dr. Li.
“Physicians should be more aware of this possible association, help reduce disease discrimination among the public, and encourage more social engagement for AA patients,” he said.
“Further studies are needed to elucidate the underlying pathophysiology between AA and dementia risk,” he added.
No cause and effect
Commenting on the study, Heather M. Snyder, PhD, vice president of medical and scientific affairs, Alzheimer’s Association, said, “We continue to learn about and better understand factors that may increase or decrease a person’s risk of dementia.”
“While we know the immune system plays a role in Alzheimer’s and other dementia, we are still investigating links between, and impact of, autoimmune diseases – like alopecia areata, rheumatoid arthritis, and others – on our overall health and our brains, [which] may eventually give us important information on risk reduction strategies as well,” said Dr. Snyder, who was not involved in the research.
She cautioned that although the study did show a correlation between AA and dementia risk, this does not equate to a demonstration of cause and effect.
At present, “the message for clinicians is that when a patient comes to your office with complaints about their memory, they should, No. 1, be taken seriously; and, No. 2, receive a thorough evaluation that takes into account the many factors that may lead to cognitive decline,” Dr. Snyder said.
The study was supported by a grant from Taipei Veterans General Hospital and the Ministry of Science and Technology, Taiwan. Dr. Li, coauthors, and Dr. Snyder disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alopecia areata (AA) has been linked to a significantly increased risk for dementia, new research shows.
After controlling for an array of potential confounders, investigators found a threefold higher risk of developing any form of dementia and a fourfold higher risk of developing Alzheimer’s disease (AD) in those with AA versus the controls.
“AA shares a similar inflammatory signature with dementia and has great psychological impacts that lead to poor social engagement,” lead author Cheng-Yuan Li, MD, MSc, of the department of dermatology, Taipei (Taiwan) Veterans General Hospital.
“Poor social engagement and shared inflammatory cytokines might both be important links between AA and dementia,” said Dr. Li, who is also affiliated with the School of Medicine and the Institute of Brain Science at National Yang Ming Chiao Tung University, Taipei.
The study was published online Oct. 26, 2021, in the Journal of Clinical Psychiatry (doi: 10.4088/JCP.21m13931).
Significant psychological impact
Patients with AA often experience anxiety and depression, possibly caused by the negative emotional and psychological impact of the hair loss and partial or even complete baldness associated with the disease, the authors noted.
However, AA is also associated with an array of other atopic and autoimmune diseases, including psoriasis and systemic lupus erythematosus (SLE).
Epidemiologic research has suggested a link between dementia and autoimmune diseases such as psoriasis and SLE, with some evidence suggesting that autoimmune and inflammatory mechanisms may “play a role” in the development of AD.
Dementia in general and AD in particular, “have been shown to include an inflammatory component” that may share some of the same mediators seen in AA (eg, IL-1 beta, IL-6, and tumor necrosis factor–alpha).
Moreover, “the great negative psychosocial impact of AA might result in poor social engagement, a typical risk factor for dementia,” said Dr. Li. The investigators sought to investigate whether patients with AA actually do have a higher dementia risk than individuals without AA.
The researchers used data from the Taiwan National Health Insurance Research Database, comparing 2,534 patients with AA against 25,340 controls matched for age, sex, residence, income, dementia-related comorbidities, systemic steroid use, and annual outpatient visits. Participants were enrolled between 1998 and 2011 and followed to the end of 2013.
The mean age of the cohort was 53.9 years, and a little over half (57.6%) were female. The most common comorbidity was hypertension (32.3%), followed by dyslipidemia (27%) and diabetes (15.4%).
Dual intervention
After adjusting for potential confounders, those with AA were more likely to develop dementia, AD, and unspecified dementia, compared with controls. They also had a numerically higher risk for vascular dementia, compared with controls, but it was not statistically significant.
When participants were stratified by age, investigators found a significant association between AA and higher risk for any dementia as well as unspecified dementia in individuals of all ages and an increased risk for AD in patients with dementia age at onset of 65 years and older.
The mean age of dementia diagnosis was considerably younger in patients with AA versus controls (73.4 vs. 78.9 years, P = .002). The risk for any dementia and unspecified dementia was higher in patients of both sexes, but the risk for AD was higher only in male patients.
Sensitivity analyses that excluded the first year or first 3 years of observation yielded similar and consistent findings.
“Intervention targeting poor social engagement and inflammatory cytokines may be beneficial to AA-associated dementia,” said Dr. Li.
“Physicians should be more aware of this possible association, help reduce disease discrimination among the public, and encourage more social engagement for AA patients,” he said.
“Further studies are needed to elucidate the underlying pathophysiology between AA and dementia risk,” he added.
No cause and effect
Commenting on the study, Heather M. Snyder, PhD, vice president of medical and scientific affairs, Alzheimer’s Association, said, “We continue to learn about and better understand factors that may increase or decrease a person’s risk of dementia.”
“While we know the immune system plays a role in Alzheimer’s and other dementia, we are still investigating links between, and impact of, autoimmune diseases – like alopecia areata, rheumatoid arthritis, and others – on our overall health and our brains, [which] may eventually give us important information on risk reduction strategies as well,” said Dr. Snyder, who was not involved in the research.
She cautioned that although the study did show a correlation between AA and dementia risk, this does not equate to a demonstration of cause and effect.
At present, “the message for clinicians is that when a patient comes to your office with complaints about their memory, they should, No. 1, be taken seriously; and, No. 2, receive a thorough evaluation that takes into account the many factors that may lead to cognitive decline,” Dr. Snyder said.
The study was supported by a grant from Taipei Veterans General Hospital and the Ministry of Science and Technology, Taiwan. Dr. Li, coauthors, and Dr. Snyder disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alopecia areata (AA) has been linked to a significantly increased risk for dementia, new research shows.
After controlling for an array of potential confounders, investigators found a threefold higher risk of developing any form of dementia and a fourfold higher risk of developing Alzheimer’s disease (AD) in those with AA versus the controls.
“AA shares a similar inflammatory signature with dementia and has great psychological impacts that lead to poor social engagement,” lead author Cheng-Yuan Li, MD, MSc, of the department of dermatology, Taipei (Taiwan) Veterans General Hospital.
“Poor social engagement and shared inflammatory cytokines might both be important links between AA and dementia,” said Dr. Li, who is also affiliated with the School of Medicine and the Institute of Brain Science at National Yang Ming Chiao Tung University, Taipei.
The study was published online Oct. 26, 2021, in the Journal of Clinical Psychiatry (doi: 10.4088/JCP.21m13931).
Significant psychological impact
Patients with AA often experience anxiety and depression, possibly caused by the negative emotional and psychological impact of the hair loss and partial or even complete baldness associated with the disease, the authors noted.
However, AA is also associated with an array of other atopic and autoimmune diseases, including psoriasis and systemic lupus erythematosus (SLE).
Epidemiologic research has suggested a link between dementia and autoimmune diseases such as psoriasis and SLE, with some evidence suggesting that autoimmune and inflammatory mechanisms may “play a role” in the development of AD.
Dementia in general and AD in particular, “have been shown to include an inflammatory component” that may share some of the same mediators seen in AA (eg, IL-1 beta, IL-6, and tumor necrosis factor–alpha).
Moreover, “the great negative psychosocial impact of AA might result in poor social engagement, a typical risk factor for dementia,” said Dr. Li. The investigators sought to investigate whether patients with AA actually do have a higher dementia risk than individuals without AA.
The researchers used data from the Taiwan National Health Insurance Research Database, comparing 2,534 patients with AA against 25,340 controls matched for age, sex, residence, income, dementia-related comorbidities, systemic steroid use, and annual outpatient visits. Participants were enrolled between 1998 and 2011 and followed to the end of 2013.
The mean age of the cohort was 53.9 years, and a little over half (57.6%) were female. The most common comorbidity was hypertension (32.3%), followed by dyslipidemia (27%) and diabetes (15.4%).
Dual intervention
After adjusting for potential confounders, those with AA were more likely to develop dementia, AD, and unspecified dementia, compared with controls. They also had a numerically higher risk for vascular dementia, compared with controls, but it was not statistically significant.
When participants were stratified by age, investigators found a significant association between AA and higher risk for any dementia as well as unspecified dementia in individuals of all ages and an increased risk for AD in patients with dementia age at onset of 65 years and older.
The mean age of dementia diagnosis was considerably younger in patients with AA versus controls (73.4 vs. 78.9 years, P = .002). The risk for any dementia and unspecified dementia was higher in patients of both sexes, but the risk for AD was higher only in male patients.
Sensitivity analyses that excluded the first year or first 3 years of observation yielded similar and consistent findings.
“Intervention targeting poor social engagement and inflammatory cytokines may be beneficial to AA-associated dementia,” said Dr. Li.
“Physicians should be more aware of this possible association, help reduce disease discrimination among the public, and encourage more social engagement for AA patients,” he said.
“Further studies are needed to elucidate the underlying pathophysiology between AA and dementia risk,” he added.
No cause and effect
Commenting on the study, Heather M. Snyder, PhD, vice president of medical and scientific affairs, Alzheimer’s Association, said, “We continue to learn about and better understand factors that may increase or decrease a person’s risk of dementia.”
“While we know the immune system plays a role in Alzheimer’s and other dementia, we are still investigating links between, and impact of, autoimmune diseases – like alopecia areata, rheumatoid arthritis, and others – on our overall health and our brains, [which] may eventually give us important information on risk reduction strategies as well,” said Dr. Snyder, who was not involved in the research.
She cautioned that although the study did show a correlation between AA and dementia risk, this does not equate to a demonstration of cause and effect.
At present, “the message for clinicians is that when a patient comes to your office with complaints about their memory, they should, No. 1, be taken seriously; and, No. 2, receive a thorough evaluation that takes into account the many factors that may lead to cognitive decline,” Dr. Snyder said.
The study was supported by a grant from Taipei Veterans General Hospital and the Ministry of Science and Technology, Taiwan. Dr. Li, coauthors, and Dr. Snyder disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
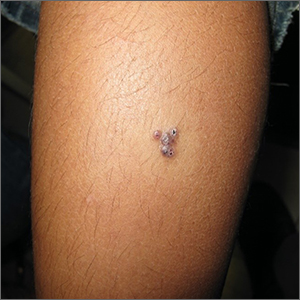
Enlarging purple plaque on leg
Clinical and dermoscopic features were consistent with a sporadic angiokeratoma, a benign ectasia of vessels associated with keratinization. Diagnosis was confirmed with shave biopsy.
Sporadic angiokeratomas are common and increase with age. They may occur anywhere on the skin, including mucous membranes. Dermoscopy of an angiokeratoma will reveal vascular lacunae—small pools of red or near black blood—as well as keratin scale, which appears as a white veil or shiny white lines.1
Other subtypes of angiokeratomas may be more widespread, including angiokeratoma of Fordyce, which manifests on the vulva and scrotum (usually in adulthood), or angiokeratoma circumscriptum, which occurs congenitally. There are some rare syndromic conditions associated with widespread angiokeratomas, such as Fabry disease.
The differential diagnosis for this solitary, vascular-appearing papule or plaque includes cherry angioma, pyogenic granuloma, and melanoma. Over time, the keratinization may increase, and lesions may look like a wart. A punch or shave biopsy will distinguish an angiokeratoma from other types of lesions. When a lesion is small enough, it may also be curative.
Angiokeratomas do not require treatment. However, because they may occur in cosmetically sensitive areas or bleed when traumatized, remedies are often sought. Excision, cryotherapy, electrocautery, and vascular laser are all possible treatments.
In this case, the patient was treated with curettage and light electrocautery, which left a small scar.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318-325. doi: 10.1001/archderm.143.3.318

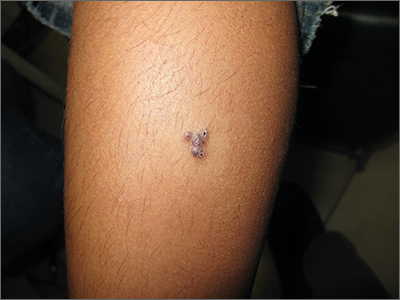
Clinical and dermoscopic features were consistent with a sporadic angiokeratoma, a benign ectasia of vessels associated with keratinization. Diagnosis was confirmed with shave biopsy.
Sporadic angiokeratomas are common and increase with age. They may occur anywhere on the skin, including mucous membranes. Dermoscopy of an angiokeratoma will reveal vascular lacunae—small pools of red or near black blood—as well as keratin scale, which appears as a white veil or shiny white lines.1
Other subtypes of angiokeratomas may be more widespread, including angiokeratoma of Fordyce, which manifests on the vulva and scrotum (usually in adulthood), or angiokeratoma circumscriptum, which occurs congenitally. There are some rare syndromic conditions associated with widespread angiokeratomas, such as Fabry disease.
The differential diagnosis for this solitary, vascular-appearing papule or plaque includes cherry angioma, pyogenic granuloma, and melanoma. Over time, the keratinization may increase, and lesions may look like a wart. A punch or shave biopsy will distinguish an angiokeratoma from other types of lesions. When a lesion is small enough, it may also be curative.
Angiokeratomas do not require treatment. However, because they may occur in cosmetically sensitive areas or bleed when traumatized, remedies are often sought. Excision, cryotherapy, electrocautery, and vascular laser are all possible treatments.
In this case, the patient was treated with curettage and light electrocautery, which left a small scar.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Clinical and dermoscopic features were consistent with a sporadic angiokeratoma, a benign ectasia of vessels associated with keratinization. Diagnosis was confirmed with shave biopsy.
Sporadic angiokeratomas are common and increase with age. They may occur anywhere on the skin, including mucous membranes. Dermoscopy of an angiokeratoma will reveal vascular lacunae—small pools of red or near black blood—as well as keratin scale, which appears as a white veil or shiny white lines.1
Other subtypes of angiokeratomas may be more widespread, including angiokeratoma of Fordyce, which manifests on the vulva and scrotum (usually in adulthood), or angiokeratoma circumscriptum, which occurs congenitally. There are some rare syndromic conditions associated with widespread angiokeratomas, such as Fabry disease.
The differential diagnosis for this solitary, vascular-appearing papule or plaque includes cherry angioma, pyogenic granuloma, and melanoma. Over time, the keratinization may increase, and lesions may look like a wart. A punch or shave biopsy will distinguish an angiokeratoma from other types of lesions. When a lesion is small enough, it may also be curative.
Angiokeratomas do not require treatment. However, because they may occur in cosmetically sensitive areas or bleed when traumatized, remedies are often sought. Excision, cryotherapy, electrocautery, and vascular laser are all possible treatments.
In this case, the patient was treated with curettage and light electrocautery, which left a small scar.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318-325. doi: 10.1001/archderm.143.3.318
1. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318-325. doi: 10.1001/archderm.143.3.318
Skin checks reduce all-cause but not melanoma-specific deaths
In Australia, where they know a thing or two about skin cancer, authors of .
Among patients in New South Wales diagnosed with melanoma in 2006 or 2007 and followed for nearly 12 years, there was no significant difference in the rate of melanoma-specific death associated with either patient-detected or clinician-detected melanomas in an analysis adjusted for prognostic factors.
Although melanomas found through routine clinician-performed skin checks were associated with a 25% reduction in all-cause mortality compared with patient-detected lesions (P = .006), this difference may have been due to the tendency of health-oriented patients to participate in screening programs.
The study – one of the largest to date and performed in an area of the world where there is a high incidence of skin cancer and high degree of public awareness of the risks of too much sun exposure – could not fully answer its central question: Can routine skin checks, a proxy for skin cancer screening, significantly decrease the incidence of melanoma-related deaths?
“A large randomized clinical trial is needed to provide definitive evidence that screening for skin cancer reduces melanoma-specific and all-cause mortality among people invited (vs. not invited) to screen, but there are concerns about feasibility. Our findings could be used to estimate the sample size for a future trial,” wrote Caroline G. Watts, PhD, of the University of Sydney, Australia, and colleagues. Their study was published online Nov. 3 in JAMA Dermatology.
In an editorial accompanying the study, dermatologists Allan C. Halpern, MD, and Michael A. Marchetti, MD, of Memorial Sloan-Kettering Cancer Center in New York, point out that “there has never been a randomized clinical trial of melanoma screening, nor is there one currently ongoing or planned. Even if one were to be initiated immediately, such a trial would take well over a decade to conduct.
“Thus, for the foreseeable future, our approaches to melanoma secondary prevention need to be based on indirect evidence and our understanding of biology and epidemiology,” they wrote.
A dermatology researcher who was not involved in the study said that while it doesn’t solve the screening conundrum, it does highlight the value of public health campaigns.
“The way that I interpret the data, especially the fact that it’s coming out of Australia, is that if education about self-examination is done properly, that can also be effective in terms of detecting these skin cancers,” said Shawn Demehri, MD, PhD, principal investigator at the Cutaneous Biology Research Center at Massachusetts General Hospital, Boston. Dr. Demehri was asked to comment on the study.
“I would argue that the results would probably have been different if the study had been conducted in the U.S. rather than Australia, because the education in terms of self-examination is much more advanced and organized in Australia,” he said in an interview.
Study details
To assess melanoma-specific and all-cause mortality associated with melanoma identified through routine skin checks, Dr. Watts and colleagues followed patients diagnosed with melanoma from October 2006 through October 2007 who were enrolled in the Melanoma Patterns of Care Study. The patients were followed until 2018 (mean follow-up 11.9 years).
Of the 2,452 patients for whom data were available, 291 had an initial diagnosis of primary melanoma in situ (MIS), and 2,161 were diagnosed with invasive cutaneous melanoma.
The median age at diagnosis was 65 years, ranging from 16 to 98 years. Nearly two-thirds of the patients (61%) were men.
Among all patients, 858 (35%) had melanoma detected during a routine skin check, 1,148 (47%) detected the lesions themselves, 293 (12%) had incidentally-detected melanomas, and 153 (6%) had lesions detected by other, unspecified means.
In analyses adjusted for age and sex, the investigators found that compared with patient-detected lesions, melanomas detected during routine skin checks were associated with a 59% lower risk for melanoma-specific mortality (subhazard ratio, 0.41, P < .001) and 36% lower risk for all-cause mortality (hazard ratio, 0.64, P < .001).
But after adjustment for melanoma prognostic factors such as ulceration and mitotic rate, the association of skin check–detected lesions with melanoma-specific mortality was no longer statistically significant. The association with lower all-cause mortality was somewhat attenuated, but remained significant (HR, 0.75, P = .006).
Factors associated with a higher likelihood of melanoma detection during routine skin checks included males vs. females, a history of melanoma, having multiple moles, age 50 or older, and residence in a urban vs. rural areas.
Screen with care
In their editorial, Dr. Halpern and Dr. Marchetti propose methods for screening that find a balance between detection of significant disease and potential harm to patients from unnecessary biopsy or invasive procedures.
“For many lesions, we could use serial photography and dermoscopy in lieu of tissue biopsy to identify those that are truly dynamic outliers and likely to be of greater risk to the patient. An analogous approach is already used for the management of small lung nodules detected incidentally and through screening,” they wrote.
They also raise the issue of potential overdiagnosis and overtreatment of MIS, and recommend an approach similar to that used for some older patients with prostate cancer, for example.
“The consequences of MIS treatment differ greatly based on the type, anatomic location, and size of the tumor; these factors should be considered in shared decision-making with patients. Options such as active surveillance and topical therapy should be discussed, particularly in those with significant comorbidities or advanced age,” they wrote.
The study was supported by grants from the Australian National Health and Medical Research Council, Cancer Institute New South Wales, and the New South Wales State Government. Dr. Watts, Dr. Halpern, Dr. Marchetti, and Dr. Demehri reported having no conflicts of interest.
In Australia, where they know a thing or two about skin cancer, authors of .
Among patients in New South Wales diagnosed with melanoma in 2006 or 2007 and followed for nearly 12 years, there was no significant difference in the rate of melanoma-specific death associated with either patient-detected or clinician-detected melanomas in an analysis adjusted for prognostic factors.
Although melanomas found through routine clinician-performed skin checks were associated with a 25% reduction in all-cause mortality compared with patient-detected lesions (P = .006), this difference may have been due to the tendency of health-oriented patients to participate in screening programs.
The study – one of the largest to date and performed in an area of the world where there is a high incidence of skin cancer and high degree of public awareness of the risks of too much sun exposure – could not fully answer its central question: Can routine skin checks, a proxy for skin cancer screening, significantly decrease the incidence of melanoma-related deaths?
“A large randomized clinical trial is needed to provide definitive evidence that screening for skin cancer reduces melanoma-specific and all-cause mortality among people invited (vs. not invited) to screen, but there are concerns about feasibility. Our findings could be used to estimate the sample size for a future trial,” wrote Caroline G. Watts, PhD, of the University of Sydney, Australia, and colleagues. Their study was published online Nov. 3 in JAMA Dermatology.
In an editorial accompanying the study, dermatologists Allan C. Halpern, MD, and Michael A. Marchetti, MD, of Memorial Sloan-Kettering Cancer Center in New York, point out that “there has never been a randomized clinical trial of melanoma screening, nor is there one currently ongoing or planned. Even if one were to be initiated immediately, such a trial would take well over a decade to conduct.
“Thus, for the foreseeable future, our approaches to melanoma secondary prevention need to be based on indirect evidence and our understanding of biology and epidemiology,” they wrote.
A dermatology researcher who was not involved in the study said that while it doesn’t solve the screening conundrum, it does highlight the value of public health campaigns.
“The way that I interpret the data, especially the fact that it’s coming out of Australia, is that if education about self-examination is done properly, that can also be effective in terms of detecting these skin cancers,” said Shawn Demehri, MD, PhD, principal investigator at the Cutaneous Biology Research Center at Massachusetts General Hospital, Boston. Dr. Demehri was asked to comment on the study.
“I would argue that the results would probably have been different if the study had been conducted in the U.S. rather than Australia, because the education in terms of self-examination is much more advanced and organized in Australia,” he said in an interview.
Study details
To assess melanoma-specific and all-cause mortality associated with melanoma identified through routine skin checks, Dr. Watts and colleagues followed patients diagnosed with melanoma from October 2006 through October 2007 who were enrolled in the Melanoma Patterns of Care Study. The patients were followed until 2018 (mean follow-up 11.9 years).
Of the 2,452 patients for whom data were available, 291 had an initial diagnosis of primary melanoma in situ (MIS), and 2,161 were diagnosed with invasive cutaneous melanoma.
The median age at diagnosis was 65 years, ranging from 16 to 98 years. Nearly two-thirds of the patients (61%) were men.
Among all patients, 858 (35%) had melanoma detected during a routine skin check, 1,148 (47%) detected the lesions themselves, 293 (12%) had incidentally-detected melanomas, and 153 (6%) had lesions detected by other, unspecified means.
In analyses adjusted for age and sex, the investigators found that compared with patient-detected lesions, melanomas detected during routine skin checks were associated with a 59% lower risk for melanoma-specific mortality (subhazard ratio, 0.41, P < .001) and 36% lower risk for all-cause mortality (hazard ratio, 0.64, P < .001).
But after adjustment for melanoma prognostic factors such as ulceration and mitotic rate, the association of skin check–detected lesions with melanoma-specific mortality was no longer statistically significant. The association with lower all-cause mortality was somewhat attenuated, but remained significant (HR, 0.75, P = .006).
Factors associated with a higher likelihood of melanoma detection during routine skin checks included males vs. females, a history of melanoma, having multiple moles, age 50 or older, and residence in a urban vs. rural areas.
Screen with care
In their editorial, Dr. Halpern and Dr. Marchetti propose methods for screening that find a balance between detection of significant disease and potential harm to patients from unnecessary biopsy or invasive procedures.
“For many lesions, we could use serial photography and dermoscopy in lieu of tissue biopsy to identify those that are truly dynamic outliers and likely to be of greater risk to the patient. An analogous approach is already used for the management of small lung nodules detected incidentally and through screening,” they wrote.
They also raise the issue of potential overdiagnosis and overtreatment of MIS, and recommend an approach similar to that used for some older patients with prostate cancer, for example.
“The consequences of MIS treatment differ greatly based on the type, anatomic location, and size of the tumor; these factors should be considered in shared decision-making with patients. Options such as active surveillance and topical therapy should be discussed, particularly in those with significant comorbidities or advanced age,” they wrote.
The study was supported by grants from the Australian National Health and Medical Research Council, Cancer Institute New South Wales, and the New South Wales State Government. Dr. Watts, Dr. Halpern, Dr. Marchetti, and Dr. Demehri reported having no conflicts of interest.
In Australia, where they know a thing or two about skin cancer, authors of .
Among patients in New South Wales diagnosed with melanoma in 2006 or 2007 and followed for nearly 12 years, there was no significant difference in the rate of melanoma-specific death associated with either patient-detected or clinician-detected melanomas in an analysis adjusted for prognostic factors.
Although melanomas found through routine clinician-performed skin checks were associated with a 25% reduction in all-cause mortality compared with patient-detected lesions (P = .006), this difference may have been due to the tendency of health-oriented patients to participate in screening programs.
The study – one of the largest to date and performed in an area of the world where there is a high incidence of skin cancer and high degree of public awareness of the risks of too much sun exposure – could not fully answer its central question: Can routine skin checks, a proxy for skin cancer screening, significantly decrease the incidence of melanoma-related deaths?
“A large randomized clinical trial is needed to provide definitive evidence that screening for skin cancer reduces melanoma-specific and all-cause mortality among people invited (vs. not invited) to screen, but there are concerns about feasibility. Our findings could be used to estimate the sample size for a future trial,” wrote Caroline G. Watts, PhD, of the University of Sydney, Australia, and colleagues. Their study was published online Nov. 3 in JAMA Dermatology.
In an editorial accompanying the study, dermatologists Allan C. Halpern, MD, and Michael A. Marchetti, MD, of Memorial Sloan-Kettering Cancer Center in New York, point out that “there has never been a randomized clinical trial of melanoma screening, nor is there one currently ongoing or planned. Even if one were to be initiated immediately, such a trial would take well over a decade to conduct.
“Thus, for the foreseeable future, our approaches to melanoma secondary prevention need to be based on indirect evidence and our understanding of biology and epidemiology,” they wrote.
A dermatology researcher who was not involved in the study said that while it doesn’t solve the screening conundrum, it does highlight the value of public health campaigns.
“The way that I interpret the data, especially the fact that it’s coming out of Australia, is that if education about self-examination is done properly, that can also be effective in terms of detecting these skin cancers,” said Shawn Demehri, MD, PhD, principal investigator at the Cutaneous Biology Research Center at Massachusetts General Hospital, Boston. Dr. Demehri was asked to comment on the study.
“I would argue that the results would probably have been different if the study had been conducted in the U.S. rather than Australia, because the education in terms of self-examination is much more advanced and organized in Australia,” he said in an interview.
Study details
To assess melanoma-specific and all-cause mortality associated with melanoma identified through routine skin checks, Dr. Watts and colleagues followed patients diagnosed with melanoma from October 2006 through October 2007 who were enrolled in the Melanoma Patterns of Care Study. The patients were followed until 2018 (mean follow-up 11.9 years).
Of the 2,452 patients for whom data were available, 291 had an initial diagnosis of primary melanoma in situ (MIS), and 2,161 were diagnosed with invasive cutaneous melanoma.
The median age at diagnosis was 65 years, ranging from 16 to 98 years. Nearly two-thirds of the patients (61%) were men.
Among all patients, 858 (35%) had melanoma detected during a routine skin check, 1,148 (47%) detected the lesions themselves, 293 (12%) had incidentally-detected melanomas, and 153 (6%) had lesions detected by other, unspecified means.
In analyses adjusted for age and sex, the investigators found that compared with patient-detected lesions, melanomas detected during routine skin checks were associated with a 59% lower risk for melanoma-specific mortality (subhazard ratio, 0.41, P < .001) and 36% lower risk for all-cause mortality (hazard ratio, 0.64, P < .001).
But after adjustment for melanoma prognostic factors such as ulceration and mitotic rate, the association of skin check–detected lesions with melanoma-specific mortality was no longer statistically significant. The association with lower all-cause mortality was somewhat attenuated, but remained significant (HR, 0.75, P = .006).
Factors associated with a higher likelihood of melanoma detection during routine skin checks included males vs. females, a history of melanoma, having multiple moles, age 50 or older, and residence in a urban vs. rural areas.
Screen with care
In their editorial, Dr. Halpern and Dr. Marchetti propose methods for screening that find a balance between detection of significant disease and potential harm to patients from unnecessary biopsy or invasive procedures.
“For many lesions, we could use serial photography and dermoscopy in lieu of tissue biopsy to identify those that are truly dynamic outliers and likely to be of greater risk to the patient. An analogous approach is already used for the management of small lung nodules detected incidentally and through screening,” they wrote.
They also raise the issue of potential overdiagnosis and overtreatment of MIS, and recommend an approach similar to that used for some older patients with prostate cancer, for example.
“The consequences of MIS treatment differ greatly based on the type, anatomic location, and size of the tumor; these factors should be considered in shared decision-making with patients. Options such as active surveillance and topical therapy should be discussed, particularly in those with significant comorbidities or advanced age,” they wrote.
The study was supported by grants from the Australian National Health and Medical Research Council, Cancer Institute New South Wales, and the New South Wales State Government. Dr. Watts, Dr. Halpern, Dr. Marchetti, and Dr. Demehri reported having no conflicts of interest.
FROM JAMA DERMATOLOGY
Hair follicle miniaturization common in persistent chemo-induced alopecia, case series suggests
and treatment with minoxidil (sometimes with antiandrogen therapy) was associated with improved hair density, according to a recently published retrospective case series.
“An improvement in hair density was observed in most of the patients treated with topical minoxidil or LDOM [low-dose oral minoxidil], with a more favorable outcome seen with LDOM with or without antiandrogens,” reported Bevin Bhoyrul, MBBS, of Sinclair Dermatology in Melbourne and coauthors from the United Kingdom and Germany.
The findings, published in JAMA Dermatology, suggest that pCIA “may be at least partly reversible,” they wrote.
The investigators analyzed the clinicopathologic characteristics of pCIA in 100 patients presenting to the hair clinics, as well as the results of trichoscopy performed in 90 of the patients and biopsies in 18. The researchers also assessed the effectiveness of treatment in 49 of these patients who met their criteria of completing at least 6 months of therapy with minoxidil.
Almost all patients in their series – 92% – were treated with taxanes and had more severe alopecia than those who weren’t exposed to taxanes (a median Sinclair scale grade of 4 vs. 2). Defined as absent or incomplete hair regrowth 6 months or more after completion of chemotherapy, pCIA has been increasingly reported in the literature, the authors note.
Of the 100 patients, all but one of whom were women, 39 had globally-reduced hair density that also involved the occipital area (diffuse alopecia), and 55 patients had thinning of the centroparietal scalp hair in a female pattern hair loss (FPHL) distribution. Patients presented between November 2011 and February 2020 and had a mean age of 54. The Sinclair scale, which grades from 1 to 5, was used to assess the severity of hair loss in these patients.
Five female patients had bitemporal recession or balding of the crown in a male pattern hair loss (MPHL) distribution, and the one male patient had extensive baldness resembling Hamilton-Norwood type VII.
The vast majority of patients who had trichoscopy performed – 88% – had trichoscopic features that were “indistinguishable from those of androgenetic alopecia,” most commonly hair shaft diameter variability, increased vellus hairs, and predominant single-hair follicular units, the authors reported.
Of the 18 patients who had biopsies, 14 had androgenetic alopecia-like features with decreased terminal hairs, increased vellus hairs, and fibrous streamers. The reduced terminal-to-vellus ratio characterizes hair follicle miniaturization, a hallmark of androgenetic alopecia, they said. (Two patients had cicatricial alopecia, and two had features of both.)
“The predominant phenotypes of pCIA show prominent vellus hairs both clinically and histologically, suggesting that terminal hair follicles undergo miniaturization,” Dr. Bhoyrul and coauthors wrote. Among the 49 patients who completed 6 months or more of treatment, the median Sinclair grade improved from 4 to 3 in 21 patients who received topical minoxidil for a median duration of 17 months; from 4 to 2.5 in 18 patients who received LDOM for a median duration of 29 months; and from 5 to 3 in 10 patients who received LDOM combined with an antiandrogen, such as spironolactone, for a median of 33 months.
Almost three-quarters of the patients in the series received adjuvant hormone therapy, which is independently associated with hair loss, the authors noted. However, there was no statistically significant difference in the pattern or severity of alopecia between patients who were treated with endocrine therapy and those who weren’t.
Asked to comment on the study and on the care of patients with pCIA, Maria K. Hordinsky, MD, professor and chair of dermatology at the University of Minnesota, Minneapolis, and an expert in hair diseases, said the case series points to the value of biopsies in patients with pCIA.
“Some patients really do have a loss of hair follicles,” she said. “But if you do a biopsy and see this miniaturization of the hair follicles, then we have tools to stimulate the hair follicles to become more normal. ... These patients can be successfully treated.”
For patients who do not want to do a biopsy, a therapeutic trial is acceptable. “But knowing helps set expectations for people,” she said. “If the follicles are really small, it will take months [of therapy].”
In addition to topical minoxidil, which she said “is always a good tool,” and LDOM, which is “becoming very popular,” Dr. Hordinsky has used low-level laser light successfully. She cautioned against the use of spironolactone and other hair-growth promoting therapies with potentially significant hormonal impacts unless there is discussion between the dermatologist, oncologist, and patient.
The authors of the case series called in their conclusion for wider use of hair-protective strategies such as scalp hypothermia. But Dr. Hordinsky said that, in the United States, there are divergent opinions among oncologists and among cancer centers on the use of scalp cooling and whether or not it might lessen response to chemotherapy.
More research is needed, she noted, on chemotherapy-induced hair loss in patients of different races and ethnicities. Of the 100 patients in the case series, 91 were European; others were Afro Caribbean, Middle Eastern, and South Asian.
Dr. Bhoyrul is supported by the Geoffrey Dowling Fellowship from the British Association of Dermatologists. One coauthor disclosed serving as a principal investigator and/or scientific board member for various pharmaceutical companies, outside of the submitted study. There were no other disclosures reported. Dr. Hordinsky, the immediate past president of the American Hair Research Society and a section editor for hair diseases in UpToDate, had no relevant disclosures.
and treatment with minoxidil (sometimes with antiandrogen therapy) was associated with improved hair density, according to a recently published retrospective case series.
“An improvement in hair density was observed in most of the patients treated with topical minoxidil or LDOM [low-dose oral minoxidil], with a more favorable outcome seen with LDOM with or without antiandrogens,” reported Bevin Bhoyrul, MBBS, of Sinclair Dermatology in Melbourne and coauthors from the United Kingdom and Germany.
The findings, published in JAMA Dermatology, suggest that pCIA “may be at least partly reversible,” they wrote.
The investigators analyzed the clinicopathologic characteristics of pCIA in 100 patients presenting to the hair clinics, as well as the results of trichoscopy performed in 90 of the patients and biopsies in 18. The researchers also assessed the effectiveness of treatment in 49 of these patients who met their criteria of completing at least 6 months of therapy with minoxidil.
Almost all patients in their series – 92% – were treated with taxanes and had more severe alopecia than those who weren’t exposed to taxanes (a median Sinclair scale grade of 4 vs. 2). Defined as absent or incomplete hair regrowth 6 months or more after completion of chemotherapy, pCIA has been increasingly reported in the literature, the authors note.
Of the 100 patients, all but one of whom were women, 39 had globally-reduced hair density that also involved the occipital area (diffuse alopecia), and 55 patients had thinning of the centroparietal scalp hair in a female pattern hair loss (FPHL) distribution. Patients presented between November 2011 and February 2020 and had a mean age of 54. The Sinclair scale, which grades from 1 to 5, was used to assess the severity of hair loss in these patients.
Five female patients had bitemporal recession or balding of the crown in a male pattern hair loss (MPHL) distribution, and the one male patient had extensive baldness resembling Hamilton-Norwood type VII.
The vast majority of patients who had trichoscopy performed – 88% – had trichoscopic features that were “indistinguishable from those of androgenetic alopecia,” most commonly hair shaft diameter variability, increased vellus hairs, and predominant single-hair follicular units, the authors reported.
Of the 18 patients who had biopsies, 14 had androgenetic alopecia-like features with decreased terminal hairs, increased vellus hairs, and fibrous streamers. The reduced terminal-to-vellus ratio characterizes hair follicle miniaturization, a hallmark of androgenetic alopecia, they said. (Two patients had cicatricial alopecia, and two had features of both.)
“The predominant phenotypes of pCIA show prominent vellus hairs both clinically and histologically, suggesting that terminal hair follicles undergo miniaturization,” Dr. Bhoyrul and coauthors wrote. Among the 49 patients who completed 6 months or more of treatment, the median Sinclair grade improved from 4 to 3 in 21 patients who received topical minoxidil for a median duration of 17 months; from 4 to 2.5 in 18 patients who received LDOM for a median duration of 29 months; and from 5 to 3 in 10 patients who received LDOM combined with an antiandrogen, such as spironolactone, for a median of 33 months.
Almost three-quarters of the patients in the series received adjuvant hormone therapy, which is independently associated with hair loss, the authors noted. However, there was no statistically significant difference in the pattern or severity of alopecia between patients who were treated with endocrine therapy and those who weren’t.
Asked to comment on the study and on the care of patients with pCIA, Maria K. Hordinsky, MD, professor and chair of dermatology at the University of Minnesota, Minneapolis, and an expert in hair diseases, said the case series points to the value of biopsies in patients with pCIA.
“Some patients really do have a loss of hair follicles,” she said. “But if you do a biopsy and see this miniaturization of the hair follicles, then we have tools to stimulate the hair follicles to become more normal. ... These patients can be successfully treated.”
For patients who do not want to do a biopsy, a therapeutic trial is acceptable. “But knowing helps set expectations for people,” she said. “If the follicles are really small, it will take months [of therapy].”
In addition to topical minoxidil, which she said “is always a good tool,” and LDOM, which is “becoming very popular,” Dr. Hordinsky has used low-level laser light successfully. She cautioned against the use of spironolactone and other hair-growth promoting therapies with potentially significant hormonal impacts unless there is discussion between the dermatologist, oncologist, and patient.
The authors of the case series called in their conclusion for wider use of hair-protective strategies such as scalp hypothermia. But Dr. Hordinsky said that, in the United States, there are divergent opinions among oncologists and among cancer centers on the use of scalp cooling and whether or not it might lessen response to chemotherapy.
More research is needed, she noted, on chemotherapy-induced hair loss in patients of different races and ethnicities. Of the 100 patients in the case series, 91 were European; others were Afro Caribbean, Middle Eastern, and South Asian.
Dr. Bhoyrul is supported by the Geoffrey Dowling Fellowship from the British Association of Dermatologists. One coauthor disclosed serving as a principal investigator and/or scientific board member for various pharmaceutical companies, outside of the submitted study. There were no other disclosures reported. Dr. Hordinsky, the immediate past president of the American Hair Research Society and a section editor for hair diseases in UpToDate, had no relevant disclosures.
and treatment with minoxidil (sometimes with antiandrogen therapy) was associated with improved hair density, according to a recently published retrospective case series.
“An improvement in hair density was observed in most of the patients treated with topical minoxidil or LDOM [low-dose oral minoxidil], with a more favorable outcome seen with LDOM with or without antiandrogens,” reported Bevin Bhoyrul, MBBS, of Sinclair Dermatology in Melbourne and coauthors from the United Kingdom and Germany.
The findings, published in JAMA Dermatology, suggest that pCIA “may be at least partly reversible,” they wrote.
The investigators analyzed the clinicopathologic characteristics of pCIA in 100 patients presenting to the hair clinics, as well as the results of trichoscopy performed in 90 of the patients and biopsies in 18. The researchers also assessed the effectiveness of treatment in 49 of these patients who met their criteria of completing at least 6 months of therapy with minoxidil.
Almost all patients in their series – 92% – were treated with taxanes and had more severe alopecia than those who weren’t exposed to taxanes (a median Sinclair scale grade of 4 vs. 2). Defined as absent or incomplete hair regrowth 6 months or more after completion of chemotherapy, pCIA has been increasingly reported in the literature, the authors note.
Of the 100 patients, all but one of whom were women, 39 had globally-reduced hair density that also involved the occipital area (diffuse alopecia), and 55 patients had thinning of the centroparietal scalp hair in a female pattern hair loss (FPHL) distribution. Patients presented between November 2011 and February 2020 and had a mean age of 54. The Sinclair scale, which grades from 1 to 5, was used to assess the severity of hair loss in these patients.
Five female patients had bitemporal recession or balding of the crown in a male pattern hair loss (MPHL) distribution, and the one male patient had extensive baldness resembling Hamilton-Norwood type VII.
The vast majority of patients who had trichoscopy performed – 88% – had trichoscopic features that were “indistinguishable from those of androgenetic alopecia,” most commonly hair shaft diameter variability, increased vellus hairs, and predominant single-hair follicular units, the authors reported.
Of the 18 patients who had biopsies, 14 had androgenetic alopecia-like features with decreased terminal hairs, increased vellus hairs, and fibrous streamers. The reduced terminal-to-vellus ratio characterizes hair follicle miniaturization, a hallmark of androgenetic alopecia, they said. (Two patients had cicatricial alopecia, and two had features of both.)
“The predominant phenotypes of pCIA show prominent vellus hairs both clinically and histologically, suggesting that terminal hair follicles undergo miniaturization,” Dr. Bhoyrul and coauthors wrote. Among the 49 patients who completed 6 months or more of treatment, the median Sinclair grade improved from 4 to 3 in 21 patients who received topical minoxidil for a median duration of 17 months; from 4 to 2.5 in 18 patients who received LDOM for a median duration of 29 months; and from 5 to 3 in 10 patients who received LDOM combined with an antiandrogen, such as spironolactone, for a median of 33 months.
Almost three-quarters of the patients in the series received adjuvant hormone therapy, which is independently associated with hair loss, the authors noted. However, there was no statistically significant difference in the pattern or severity of alopecia between patients who were treated with endocrine therapy and those who weren’t.
Asked to comment on the study and on the care of patients with pCIA, Maria K. Hordinsky, MD, professor and chair of dermatology at the University of Minnesota, Minneapolis, and an expert in hair diseases, said the case series points to the value of biopsies in patients with pCIA.
“Some patients really do have a loss of hair follicles,” she said. “But if you do a biopsy and see this miniaturization of the hair follicles, then we have tools to stimulate the hair follicles to become more normal. ... These patients can be successfully treated.”
For patients who do not want to do a biopsy, a therapeutic trial is acceptable. “But knowing helps set expectations for people,” she said. “If the follicles are really small, it will take months [of therapy].”
In addition to topical minoxidil, which she said “is always a good tool,” and LDOM, which is “becoming very popular,” Dr. Hordinsky has used low-level laser light successfully. She cautioned against the use of spironolactone and other hair-growth promoting therapies with potentially significant hormonal impacts unless there is discussion between the dermatologist, oncologist, and patient.
The authors of the case series called in their conclusion for wider use of hair-protective strategies such as scalp hypothermia. But Dr. Hordinsky said that, in the United States, there are divergent opinions among oncologists and among cancer centers on the use of scalp cooling and whether or not it might lessen response to chemotherapy.
More research is needed, she noted, on chemotherapy-induced hair loss in patients of different races and ethnicities. Of the 100 patients in the case series, 91 were European; others were Afro Caribbean, Middle Eastern, and South Asian.
Dr. Bhoyrul is supported by the Geoffrey Dowling Fellowship from the British Association of Dermatologists. One coauthor disclosed serving as a principal investigator and/or scientific board member for various pharmaceutical companies, outside of the submitted study. There were no other disclosures reported. Dr. Hordinsky, the immediate past president of the American Hair Research Society and a section editor for hair diseases in UpToDate, had no relevant disclosures.
FROM JAMA DERMATOLOGY
Evaluations of novel approaches to treating NF-1 tumors are underway
In the clinical experience of R. Rox Anderson, MD, currently available treatment options for benign tumors caused by neurofibromatosis type 1 (NF-1) are not acceptable.
“Simply removing the tumors with surgery is not the answer,” Dr. Anderson, a dermatologist who is the director of the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, said during a virtual course on laser and aesthetic skin therapy. “We need a way to inhibit the cutaneous neurofibromatosis early in life and prevent disfigurement that occurs when kids become adults.
“Kids with NF-1 are born looking normal,” he said. “They have café au lait macules and Lisch nodules in their eye, but they’re normal-looking kids. By early adulthood, many will grow hundreds of tumors that are disfiguring.”
In patients with NF-1, surgical excision works for cutaneous tumors but is expensive and not widely available, and is usually not covered by health insurance. “Plus, you have these adults who have already been through a lot of trauma, with the disfigurement in their lives, who have to be put under general anesthesia to remove a large number of tumors,” Dr. Anderson said at the meeting, which was named What’s the Truth and was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. Cryotherapy is a minimally invasive way to treat cutaneous neurofibroma tumors, “but this destroys the overlying skin, so you get unwanted destruction,” he said. “I like the idea of selecting heating, but we don’t know yet by what method.”
Dr. Anderson and his colleagues just launched a comparative clinical They plan to perform one or more treatment methods per patient in a single treatment session, then follow up at least 6 months later. Baseline and untreated cutaneous NF lesions will serve as controls. The researchers plan to conduct three-dimensional imaging, clinical assessments, and evaluate pain and other subjective measures.
Use of deoxycholate in a pilot trial was well tolerated and induced tumor regression in adults with cutaneous NF, he said.
Dr. Anderson noted that other researchers are studying the potential role of topical or local mitogen-activated protein kinase (MEK) inhibitors for these tumors. “Systemic MEK inhibitors are effective for plexiform neuromas, but cause significant cutaneous side effects,” he said. A “soft” MEK inhibitor, NFX-179 is rapidly metabolized such that high drug levels are achieved in skin without systemic drug levels. However, Dr. Anderson said that it remains unclear if this approach will prevent cutaneous NF tumors from forming, arrest their growth, or induce their regression.
Dr. Anderson reported having received research funding and/or consulting fees from numerous device and pharmaceutical companies.
Commentary by Lawrence F. Eichenfield, MD
Neurofibromatosis type 1 (NF1) is a common genodermatosis, associated with the development of neurofibromas derived from nerves, soft tissue, and skin. Cutaneous NFs often develop in later childhood onward and may be deforming, associated with pruritus, pain, and significant effect on quality of life. Dr. Anderson is a world leader in laser treatment, having developed the theories behind laser development for medical usage, as well as the laser technology used for vascular birthmarks and hair removal, laser and cooling techniques targeting fat, and “fractionating” laser energy, which has revolutionized scar management. We look forward to his group’s insights into better management of NF1 lesions!
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
In the clinical experience of R. Rox Anderson, MD, currently available treatment options for benign tumors caused by neurofibromatosis type 1 (NF-1) are not acceptable.
“Simply removing the tumors with surgery is not the answer,” Dr. Anderson, a dermatologist who is the director of the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, said during a virtual course on laser and aesthetic skin therapy. “We need a way to inhibit the cutaneous neurofibromatosis early in life and prevent disfigurement that occurs when kids become adults.
“Kids with NF-1 are born looking normal,” he said. “They have café au lait macules and Lisch nodules in their eye, but they’re normal-looking kids. By early adulthood, many will grow hundreds of tumors that are disfiguring.”
In patients with NF-1, surgical excision works for cutaneous tumors but is expensive and not widely available, and is usually not covered by health insurance. “Plus, you have these adults who have already been through a lot of trauma, with the disfigurement in their lives, who have to be put under general anesthesia to remove a large number of tumors,” Dr. Anderson said at the meeting, which was named What’s the Truth and was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. Cryotherapy is a minimally invasive way to treat cutaneous neurofibroma tumors, “but this destroys the overlying skin, so you get unwanted destruction,” he said. “I like the idea of selecting heating, but we don’t know yet by what method.”
Dr. Anderson and his colleagues just launched a comparative clinical They plan to perform one or more treatment methods per patient in a single treatment session, then follow up at least 6 months later. Baseline and untreated cutaneous NF lesions will serve as controls. The researchers plan to conduct three-dimensional imaging, clinical assessments, and evaluate pain and other subjective measures.
Use of deoxycholate in a pilot trial was well tolerated and induced tumor regression in adults with cutaneous NF, he said.
Dr. Anderson noted that other researchers are studying the potential role of topical or local mitogen-activated protein kinase (MEK) inhibitors for these tumors. “Systemic MEK inhibitors are effective for plexiform neuromas, but cause significant cutaneous side effects,” he said. A “soft” MEK inhibitor, NFX-179 is rapidly metabolized such that high drug levels are achieved in skin without systemic drug levels. However, Dr. Anderson said that it remains unclear if this approach will prevent cutaneous NF tumors from forming, arrest their growth, or induce their regression.
Dr. Anderson reported having received research funding and/or consulting fees from numerous device and pharmaceutical companies.
Commentary by Lawrence F. Eichenfield, MD
Neurofibromatosis type 1 (NF1) is a common genodermatosis, associated with the development of neurofibromas derived from nerves, soft tissue, and skin. Cutaneous NFs often develop in later childhood onward and may be deforming, associated with pruritus, pain, and significant effect on quality of life. Dr. Anderson is a world leader in laser treatment, having developed the theories behind laser development for medical usage, as well as the laser technology used for vascular birthmarks and hair removal, laser and cooling techniques targeting fat, and “fractionating” laser energy, which has revolutionized scar management. We look forward to his group’s insights into better management of NF1 lesions!
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
In the clinical experience of R. Rox Anderson, MD, currently available treatment options for benign tumors caused by neurofibromatosis type 1 (NF-1) are not acceptable.
“Simply removing the tumors with surgery is not the answer,” Dr. Anderson, a dermatologist who is the director of the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, said during a virtual course on laser and aesthetic skin therapy. “We need a way to inhibit the cutaneous neurofibromatosis early in life and prevent disfigurement that occurs when kids become adults.
“Kids with NF-1 are born looking normal,” he said. “They have café au lait macules and Lisch nodules in their eye, but they’re normal-looking kids. By early adulthood, many will grow hundreds of tumors that are disfiguring.”
In patients with NF-1, surgical excision works for cutaneous tumors but is expensive and not widely available, and is usually not covered by health insurance. “Plus, you have these adults who have already been through a lot of trauma, with the disfigurement in their lives, who have to be put under general anesthesia to remove a large number of tumors,” Dr. Anderson said at the meeting, which was named What’s the Truth and was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. Cryotherapy is a minimally invasive way to treat cutaneous neurofibroma tumors, “but this destroys the overlying skin, so you get unwanted destruction,” he said. “I like the idea of selecting heating, but we don’t know yet by what method.”
Dr. Anderson and his colleagues just launched a comparative clinical They plan to perform one or more treatment methods per patient in a single treatment session, then follow up at least 6 months later. Baseline and untreated cutaneous NF lesions will serve as controls. The researchers plan to conduct three-dimensional imaging, clinical assessments, and evaluate pain and other subjective measures.
Use of deoxycholate in a pilot trial was well tolerated and induced tumor regression in adults with cutaneous NF, he said.
Dr. Anderson noted that other researchers are studying the potential role of topical or local mitogen-activated protein kinase (MEK) inhibitors for these tumors. “Systemic MEK inhibitors are effective for plexiform neuromas, but cause significant cutaneous side effects,” he said. A “soft” MEK inhibitor, NFX-179 is rapidly metabolized such that high drug levels are achieved in skin without systemic drug levels. However, Dr. Anderson said that it remains unclear if this approach will prevent cutaneous NF tumors from forming, arrest their growth, or induce their regression.
Dr. Anderson reported having received research funding and/or consulting fees from numerous device and pharmaceutical companies.
Commentary by Lawrence F. Eichenfield, MD
Neurofibromatosis type 1 (NF1) is a common genodermatosis, associated with the development of neurofibromas derived from nerves, soft tissue, and skin. Cutaneous NFs often develop in later childhood onward and may be deforming, associated with pruritus, pain, and significant effect on quality of life. Dr. Anderson is a world leader in laser treatment, having developed the theories behind laser development for medical usage, as well as the laser technology used for vascular birthmarks and hair removal, laser and cooling techniques targeting fat, and “fractionating” laser energy, which has revolutionized scar management. We look forward to his group’s insights into better management of NF1 lesions!
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM A LASER & AESTHETIC SKIN THERAPY COURSE
Sunscreen, other sun-protective habits not linked with poorer bone health, fractures
Using , according to a new study that included more than 3,000 men and women.
“We have objective data for the first time, and in a large-scale representative population of the U.S. adults, to indicate sun protection is not associated with negative bone-related outcomes,” said study lead author Mohsen Afarideh, MD, MPH, a postdoctoral research fellow at the autoimmune skin diseases unit at the University of Pennsylvania, Philadelphia.
The study, published online in JAMA Dermatology, goes a step further than previous research by others that has found sunscreen use does not compromise vitamin D synthesis and has little effect on circulating 25-hydroxyvitamin D levels.
In the new study, researchers looked at three sun-protective behaviors – sunscreen use, staying in the shade, wearing long sleeves – and their effects on bone mineral density and the risk of fractures.
While the effects of sun-protective habits on blood levels of vitamin D and BMD scores are important, ‘’what we are more interested to know is if the sun-protective behaviors actually cause or increase the risk of fracture,” Dr. Afarideh said in an interview. “The answer to that is a firm ‘No.’ These data are very reassuring and will help clinicians to keep recommending sun protection to the public.”
Study details
Dr. Afarideh and his colleagues from the Mayo Clinic in Rochester, Minn., looked at data from the National Health and Nutrition Examination Survey (NHANES) from 2017 to 2018, obtaining final information on 3,403 men and women, ages 20-59, who completed a dermatology questionnaire The men and women reported on the three sun-protective habits, and noted whether they followed these practices always or most of the time, sometimes, or never or rarely.
The frequency of the three behaviors was not widespread. Frequent staying in the shade was reported by 31.6% of the sample, wearing long sleeves by 11.8%, and sunscreen use by 26.1%.
The researchers also had data on the participants’ bone mineral density (BMD) scores along with dietary information such as milk consumption, vitamin D supplement use, taking steroid drugs, and exercise activity.
“Moderate sunscreen use was linked with a slightly lower lumbar BMD score,” Dr. Afarideh said, which was “the only significant association that could be interpreted as concerning.” And this was more likely to be seen in older respondents, he said.
However, otherwise they found the practice of the three behaviors was not associated with lower total or site-specific BMD z scores, nor was it linked with an increased risk of osteoporotic fractures. (The BMD z score compares an individual’s bone density to the average bone density of someone their same age and gender.)
The focus on fracture risk is the more important outcome, Dr. Afarideh said. And they found no increased risk overall of osteoporotic fractures in those who practiced sun-protective behaviors.
Moderate to frequent staying in the shade was actually linked with a reduced prevalence of spine fractures in the multivariate model (odds ratio, 0.19; 95% confidence interval, 0.04-0.86, P = .02). The researchers say that may be attributable to these respondents also being careful in other areas of life, such as avoiding falls and not participating in high-risk activities that would increase the chance of fractures. “However, this is just an assumption,” Dr. Afarideh said.
Expert perspectives
Other dermatologists not involved in the new research said the study results provide some “real-world” information that’s valuable for clinicians to share with patients.
“I think this is an important study on multiple levels,” said Henry W. Lim, MD, a former president of the American Academy of Dermatology who is a member of the department of dermatology and senior vice president of academic affairs at Henry Ford Health System, Detroit. “It is a well-done study, involving a large number. It is a real-life situation, asking people their photo protective behaviors and then looking at their bone mineral density.” The bottom line, he said: “Bone health is not affected by photo protection habits in real life.”
The findings are important but not surprising, said Antony R. Young, PhD, emeritus professor of experimental photobiology at St. John’s Institute of Dermatology, King’s College, London, who has researched sunscreens and vitamin D status. “My study showed that correct sunscreen use, albeit with a relatively low SPF of 15, did prevent sunburn in a high UVR [ultraviolet radiation] environment but did allow very good vitamin D synthesis. I think this is because the necessary dose of UVB is very low.”
Michele Green, MD, a New York dermatologist and clinical staff member at Lenox Hill Hospital there, said she often hears concerns about bone health from patients. “Every week, patients ask, ‘Why would I wear sunblock? Don’t I need sun for bone health? Don’t I need it for vitamin D?’’’
Now, she said, ‘’Dermatologists can point to the study and say ‘Don’t worry.’ It clarifies that using sunscreen won’t cause you to have osteoporosis.’’
Dr. Afarideh, who was a postdoctoral research fellow at the Mayo Clinic, and his coauthors, Megha M. Tollefson, MD, and Julio C. Sartori-Valinotti, of the Mayo Clinic, and Dr. Green had no disclosures. Dr. Lim and Dr. Young consult for the sunscreen industry.
Using , according to a new study that included more than 3,000 men and women.
“We have objective data for the first time, and in a large-scale representative population of the U.S. adults, to indicate sun protection is not associated with negative bone-related outcomes,” said study lead author Mohsen Afarideh, MD, MPH, a postdoctoral research fellow at the autoimmune skin diseases unit at the University of Pennsylvania, Philadelphia.
The study, published online in JAMA Dermatology, goes a step further than previous research by others that has found sunscreen use does not compromise vitamin D synthesis and has little effect on circulating 25-hydroxyvitamin D levels.
In the new study, researchers looked at three sun-protective behaviors – sunscreen use, staying in the shade, wearing long sleeves – and their effects on bone mineral density and the risk of fractures.
While the effects of sun-protective habits on blood levels of vitamin D and BMD scores are important, ‘’what we are more interested to know is if the sun-protective behaviors actually cause or increase the risk of fracture,” Dr. Afarideh said in an interview. “The answer to that is a firm ‘No.’ These data are very reassuring and will help clinicians to keep recommending sun protection to the public.”
Study details
Dr. Afarideh and his colleagues from the Mayo Clinic in Rochester, Minn., looked at data from the National Health and Nutrition Examination Survey (NHANES) from 2017 to 2018, obtaining final information on 3,403 men and women, ages 20-59, who completed a dermatology questionnaire The men and women reported on the three sun-protective habits, and noted whether they followed these practices always or most of the time, sometimes, or never or rarely.
The frequency of the three behaviors was not widespread. Frequent staying in the shade was reported by 31.6% of the sample, wearing long sleeves by 11.8%, and sunscreen use by 26.1%.
The researchers also had data on the participants’ bone mineral density (BMD) scores along with dietary information such as milk consumption, vitamin D supplement use, taking steroid drugs, and exercise activity.
“Moderate sunscreen use was linked with a slightly lower lumbar BMD score,” Dr. Afarideh said, which was “the only significant association that could be interpreted as concerning.” And this was more likely to be seen in older respondents, he said.
However, otherwise they found the practice of the three behaviors was not associated with lower total or site-specific BMD z scores, nor was it linked with an increased risk of osteoporotic fractures. (The BMD z score compares an individual’s bone density to the average bone density of someone their same age and gender.)
The focus on fracture risk is the more important outcome, Dr. Afarideh said. And they found no increased risk overall of osteoporotic fractures in those who practiced sun-protective behaviors.
Moderate to frequent staying in the shade was actually linked with a reduced prevalence of spine fractures in the multivariate model (odds ratio, 0.19; 95% confidence interval, 0.04-0.86, P = .02). The researchers say that may be attributable to these respondents also being careful in other areas of life, such as avoiding falls and not participating in high-risk activities that would increase the chance of fractures. “However, this is just an assumption,” Dr. Afarideh said.
Expert perspectives
Other dermatologists not involved in the new research said the study results provide some “real-world” information that’s valuable for clinicians to share with patients.
“I think this is an important study on multiple levels,” said Henry W. Lim, MD, a former president of the American Academy of Dermatology who is a member of the department of dermatology and senior vice president of academic affairs at Henry Ford Health System, Detroit. “It is a well-done study, involving a large number. It is a real-life situation, asking people their photo protective behaviors and then looking at their bone mineral density.” The bottom line, he said: “Bone health is not affected by photo protection habits in real life.”
The findings are important but not surprising, said Antony R. Young, PhD, emeritus professor of experimental photobiology at St. John’s Institute of Dermatology, King’s College, London, who has researched sunscreens and vitamin D status. “My study showed that correct sunscreen use, albeit with a relatively low SPF of 15, did prevent sunburn in a high UVR [ultraviolet radiation] environment but did allow very good vitamin D synthesis. I think this is because the necessary dose of UVB is very low.”
Michele Green, MD, a New York dermatologist and clinical staff member at Lenox Hill Hospital there, said she often hears concerns about bone health from patients. “Every week, patients ask, ‘Why would I wear sunblock? Don’t I need sun for bone health? Don’t I need it for vitamin D?’’’
Now, she said, ‘’Dermatologists can point to the study and say ‘Don’t worry.’ It clarifies that using sunscreen won’t cause you to have osteoporosis.’’
Dr. Afarideh, who was a postdoctoral research fellow at the Mayo Clinic, and his coauthors, Megha M. Tollefson, MD, and Julio C. Sartori-Valinotti, of the Mayo Clinic, and Dr. Green had no disclosures. Dr. Lim and Dr. Young consult for the sunscreen industry.
Using , according to a new study that included more than 3,000 men and women.
“We have objective data for the first time, and in a large-scale representative population of the U.S. adults, to indicate sun protection is not associated with negative bone-related outcomes,” said study lead author Mohsen Afarideh, MD, MPH, a postdoctoral research fellow at the autoimmune skin diseases unit at the University of Pennsylvania, Philadelphia.
The study, published online in JAMA Dermatology, goes a step further than previous research by others that has found sunscreen use does not compromise vitamin D synthesis and has little effect on circulating 25-hydroxyvitamin D levels.
In the new study, researchers looked at three sun-protective behaviors – sunscreen use, staying in the shade, wearing long sleeves – and their effects on bone mineral density and the risk of fractures.
While the effects of sun-protective habits on blood levels of vitamin D and BMD scores are important, ‘’what we are more interested to know is if the sun-protective behaviors actually cause or increase the risk of fracture,” Dr. Afarideh said in an interview. “The answer to that is a firm ‘No.’ These data are very reassuring and will help clinicians to keep recommending sun protection to the public.”
Study details
Dr. Afarideh and his colleagues from the Mayo Clinic in Rochester, Minn., looked at data from the National Health and Nutrition Examination Survey (NHANES) from 2017 to 2018, obtaining final information on 3,403 men and women, ages 20-59, who completed a dermatology questionnaire The men and women reported on the three sun-protective habits, and noted whether they followed these practices always or most of the time, sometimes, or never or rarely.
The frequency of the three behaviors was not widespread. Frequent staying in the shade was reported by 31.6% of the sample, wearing long sleeves by 11.8%, and sunscreen use by 26.1%.
The researchers also had data on the participants’ bone mineral density (BMD) scores along with dietary information such as milk consumption, vitamin D supplement use, taking steroid drugs, and exercise activity.
“Moderate sunscreen use was linked with a slightly lower lumbar BMD score,” Dr. Afarideh said, which was “the only significant association that could be interpreted as concerning.” And this was more likely to be seen in older respondents, he said.
However, otherwise they found the practice of the three behaviors was not associated with lower total or site-specific BMD z scores, nor was it linked with an increased risk of osteoporotic fractures. (The BMD z score compares an individual’s bone density to the average bone density of someone their same age and gender.)
The focus on fracture risk is the more important outcome, Dr. Afarideh said. And they found no increased risk overall of osteoporotic fractures in those who practiced sun-protective behaviors.
Moderate to frequent staying in the shade was actually linked with a reduced prevalence of spine fractures in the multivariate model (odds ratio, 0.19; 95% confidence interval, 0.04-0.86, P = .02). The researchers say that may be attributable to these respondents also being careful in other areas of life, such as avoiding falls and not participating in high-risk activities that would increase the chance of fractures. “However, this is just an assumption,” Dr. Afarideh said.
Expert perspectives
Other dermatologists not involved in the new research said the study results provide some “real-world” information that’s valuable for clinicians to share with patients.
“I think this is an important study on multiple levels,” said Henry W. Lim, MD, a former president of the American Academy of Dermatology who is a member of the department of dermatology and senior vice president of academic affairs at Henry Ford Health System, Detroit. “It is a well-done study, involving a large number. It is a real-life situation, asking people their photo protective behaviors and then looking at their bone mineral density.” The bottom line, he said: “Bone health is not affected by photo protection habits in real life.”
The findings are important but not surprising, said Antony R. Young, PhD, emeritus professor of experimental photobiology at St. John’s Institute of Dermatology, King’s College, London, who has researched sunscreens and vitamin D status. “My study showed that correct sunscreen use, albeit with a relatively low SPF of 15, did prevent sunburn in a high UVR [ultraviolet radiation] environment but did allow very good vitamin D synthesis. I think this is because the necessary dose of UVB is very low.”
Michele Green, MD, a New York dermatologist and clinical staff member at Lenox Hill Hospital there, said she often hears concerns about bone health from patients. “Every week, patients ask, ‘Why would I wear sunblock? Don’t I need sun for bone health? Don’t I need it for vitamin D?’’’
Now, she said, ‘’Dermatologists can point to the study and say ‘Don’t worry.’ It clarifies that using sunscreen won’t cause you to have osteoporosis.’’
Dr. Afarideh, who was a postdoctoral research fellow at the Mayo Clinic, and his coauthors, Megha M. Tollefson, MD, and Julio C. Sartori-Valinotti, of the Mayo Clinic, and Dr. Green had no disclosures. Dr. Lim and Dr. Young consult for the sunscreen industry.
FROM JAMA DERMATOLOGY
One of the keys to success on social media? Entertain and educate the public
Social media isn’t everyone’s cup of tea, but
“I admit that I’m somewhat obsessed with it. I kind of blame it on my work as a dermatologist, that I’m trying to grow my social media as well. It’s interesting to me, fascinating, and I want to understand it more. I think that’s the mindset you need to approach it with.”
Perhaps no other public figure in dermatology has enjoyed success in social media more than Dr. Lee, a board-certified dermatologist who practices in Upland, Calif. In the fall of 2014, she started using Instagram to provide followers a glimpse into her life as a dermatologist, everything from Mohs surgery and Botox to keloid removals and ear lobe repair surgeries. From this she formed her alter ego, “Dr. Pimple Popper,” and became a YouTube sensation, building 7.1 million subscribers over the course of a few years, amounting to 4.5 billion lifetime views. She also grew 12 million followers on TikTok, 4.4 million followers on Instagram, 3 million on Facebook, and more than 139,000 on Twitter. About 80% of her followers are women who range between 18 and 40 years of age.
During the meeting she offered five social media marketing tips for busy clinicians:
You have to ‘play’ to ‘win.’ Active participation in social media is required to develop followers. “You cannot delegate this content,” Dr. Lee said. “You can hire people to help you or leave the task to a social media-savvy medical assistant in your office, but the content should be your responsibility ultimately, because you are the physician,” she added. Not everyone chooses to participate in social media, but it’s also something not to shy away from out of intimidation. “There is some talent associated with it, but it takes a lot of persistence as well,” she said.
Patients come first. Protect them at all costs. Dr. Lee rarely posts the faces of patients she cares for unless they grant consent in advance. “I try to show the work that I do and the beauty of dermatology,” she said during the meeting, which was named “Laser & Aesthetic Skin Therapy: What’s the Truth?” and was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. She added that taking part in social media can help you improve communication skills by engaging with followers who like, share, or respond to the material posted. “When you look back at your posts objectively, you learn about yourself and how you relate to your patients,” she said. “It helps to hone my bedside manner and my skills as a dermatologist.”
Show that you are human. Many dermatologists and other “skin influencers” have established their presence on the Internet and may be direct competitors for patients, but that doesn’t mean you can’t establish your own identity. One way to stand out is by posting content related to your authentic self, such as a photo or video that shows you engaged in a hobby, dining at a favorite restaurant, or visiting a beloved vacation spot. “Your followers don’t want a robot, someone who thinks they’re amazing and can do everything,” said Dr. Lee, who stars in her own TV reality show on TLC. “Show that you have a funny side. You want them to fall in love with you and see a little bit of your world, whatever it might be. Charm the socks off of them.”
Entertain first, educate a close second. The main way you’re going to get people to follow and watch you is to provide some entertainment, “not at the expense of a patient or your practice, though,” she said. “Then you’re going to educate people. We dermatologists have something to teach the world because we are experts on skin, hair, and nails. You want to impart this knowledge in a way that captivates people.” It’s like the sense of accomplishment that comes from learning something new after reading a book or watching a movie, she explained. “You feel good about it, and you can take that knowledge with you somewhere else. I love it when kids come up to me and tell me they know what a lipoma is, what a cyst is, and what psoriasis is because they’ve seen my show, or because they follow me on social media. It’s wonderful because I can see that I’ve educated them.”
Be kind and don’t activate the trolls. Dr. Lee allows positivity and kindness to rule the day on her social media content. “This is what I try to relay to followers, but I also do not engage with the negativity,” she said. “Every now and then, there will be someone who tries to insult what you do or who insults you personally. If you engage with them, it almost invites them to do it more. It almost gives them the ability to fight with you. Try to stay above that; just put out goodness and kindness.”
Several years ago, YouTube and Instagram temporarily shut down Dr. Lee’s accounts because she posted graphic images of skin lesions and procedures – a practice that wasn’t so commonplace at the time. “Don’t just post a graphic image just to be graphic,” she advised. “Make sure it has an educational message associated with it. That will help to validate your content. Posting a warning sign that some images may be graphic could help, too.”
Dr. Lee reported having no relevant financial disclosures.
Social media isn’t everyone’s cup of tea, but
“I admit that I’m somewhat obsessed with it. I kind of blame it on my work as a dermatologist, that I’m trying to grow my social media as well. It’s interesting to me, fascinating, and I want to understand it more. I think that’s the mindset you need to approach it with.”
Perhaps no other public figure in dermatology has enjoyed success in social media more than Dr. Lee, a board-certified dermatologist who practices in Upland, Calif. In the fall of 2014, she started using Instagram to provide followers a glimpse into her life as a dermatologist, everything from Mohs surgery and Botox to keloid removals and ear lobe repair surgeries. From this she formed her alter ego, “Dr. Pimple Popper,” and became a YouTube sensation, building 7.1 million subscribers over the course of a few years, amounting to 4.5 billion lifetime views. She also grew 12 million followers on TikTok, 4.4 million followers on Instagram, 3 million on Facebook, and more than 139,000 on Twitter. About 80% of her followers are women who range between 18 and 40 years of age.
During the meeting she offered five social media marketing tips for busy clinicians:
You have to ‘play’ to ‘win.’ Active participation in social media is required to develop followers. “You cannot delegate this content,” Dr. Lee said. “You can hire people to help you or leave the task to a social media-savvy medical assistant in your office, but the content should be your responsibility ultimately, because you are the physician,” she added. Not everyone chooses to participate in social media, but it’s also something not to shy away from out of intimidation. “There is some talent associated with it, but it takes a lot of persistence as well,” she said.
Patients come first. Protect them at all costs. Dr. Lee rarely posts the faces of patients she cares for unless they grant consent in advance. “I try to show the work that I do and the beauty of dermatology,” she said during the meeting, which was named “Laser & Aesthetic Skin Therapy: What’s the Truth?” and was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. She added that taking part in social media can help you improve communication skills by engaging with followers who like, share, or respond to the material posted. “When you look back at your posts objectively, you learn about yourself and how you relate to your patients,” she said. “It helps to hone my bedside manner and my skills as a dermatologist.”
Show that you are human. Many dermatologists and other “skin influencers” have established their presence on the Internet and may be direct competitors for patients, but that doesn’t mean you can’t establish your own identity. One way to stand out is by posting content related to your authentic self, such as a photo or video that shows you engaged in a hobby, dining at a favorite restaurant, or visiting a beloved vacation spot. “Your followers don’t want a robot, someone who thinks they’re amazing and can do everything,” said Dr. Lee, who stars in her own TV reality show on TLC. “Show that you have a funny side. You want them to fall in love with you and see a little bit of your world, whatever it might be. Charm the socks off of them.”
Entertain first, educate a close second. The main way you’re going to get people to follow and watch you is to provide some entertainment, “not at the expense of a patient or your practice, though,” she said. “Then you’re going to educate people. We dermatologists have something to teach the world because we are experts on skin, hair, and nails. You want to impart this knowledge in a way that captivates people.” It’s like the sense of accomplishment that comes from learning something new after reading a book or watching a movie, she explained. “You feel good about it, and you can take that knowledge with you somewhere else. I love it when kids come up to me and tell me they know what a lipoma is, what a cyst is, and what psoriasis is because they’ve seen my show, or because they follow me on social media. It’s wonderful because I can see that I’ve educated them.”
Be kind and don’t activate the trolls. Dr. Lee allows positivity and kindness to rule the day on her social media content. “This is what I try to relay to followers, but I also do not engage with the negativity,” she said. “Every now and then, there will be someone who tries to insult what you do or who insults you personally. If you engage with them, it almost invites them to do it more. It almost gives them the ability to fight with you. Try to stay above that; just put out goodness and kindness.”
Several years ago, YouTube and Instagram temporarily shut down Dr. Lee’s accounts because she posted graphic images of skin lesions and procedures – a practice that wasn’t so commonplace at the time. “Don’t just post a graphic image just to be graphic,” she advised. “Make sure it has an educational message associated with it. That will help to validate your content. Posting a warning sign that some images may be graphic could help, too.”
Dr. Lee reported having no relevant financial disclosures.
Social media isn’t everyone’s cup of tea, but
“I admit that I’m somewhat obsessed with it. I kind of blame it on my work as a dermatologist, that I’m trying to grow my social media as well. It’s interesting to me, fascinating, and I want to understand it more. I think that’s the mindset you need to approach it with.”
Perhaps no other public figure in dermatology has enjoyed success in social media more than Dr. Lee, a board-certified dermatologist who practices in Upland, Calif. In the fall of 2014, she started using Instagram to provide followers a glimpse into her life as a dermatologist, everything from Mohs surgery and Botox to keloid removals and ear lobe repair surgeries. From this she formed her alter ego, “Dr. Pimple Popper,” and became a YouTube sensation, building 7.1 million subscribers over the course of a few years, amounting to 4.5 billion lifetime views. She also grew 12 million followers on TikTok, 4.4 million followers on Instagram, 3 million on Facebook, and more than 139,000 on Twitter. About 80% of her followers are women who range between 18 and 40 years of age.
During the meeting she offered five social media marketing tips for busy clinicians:
You have to ‘play’ to ‘win.’ Active participation in social media is required to develop followers. “You cannot delegate this content,” Dr. Lee said. “You can hire people to help you or leave the task to a social media-savvy medical assistant in your office, but the content should be your responsibility ultimately, because you are the physician,” she added. Not everyone chooses to participate in social media, but it’s also something not to shy away from out of intimidation. “There is some talent associated with it, but it takes a lot of persistence as well,” she said.
Patients come first. Protect them at all costs. Dr. Lee rarely posts the faces of patients she cares for unless they grant consent in advance. “I try to show the work that I do and the beauty of dermatology,” she said during the meeting, which was named “Laser & Aesthetic Skin Therapy: What’s the Truth?” and was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. She added that taking part in social media can help you improve communication skills by engaging with followers who like, share, or respond to the material posted. “When you look back at your posts objectively, you learn about yourself and how you relate to your patients,” she said. “It helps to hone my bedside manner and my skills as a dermatologist.”
Show that you are human. Many dermatologists and other “skin influencers” have established their presence on the Internet and may be direct competitors for patients, but that doesn’t mean you can’t establish your own identity. One way to stand out is by posting content related to your authentic self, such as a photo or video that shows you engaged in a hobby, dining at a favorite restaurant, or visiting a beloved vacation spot. “Your followers don’t want a robot, someone who thinks they’re amazing and can do everything,” said Dr. Lee, who stars in her own TV reality show on TLC. “Show that you have a funny side. You want them to fall in love with you and see a little bit of your world, whatever it might be. Charm the socks off of them.”
Entertain first, educate a close second. The main way you’re going to get people to follow and watch you is to provide some entertainment, “not at the expense of a patient or your practice, though,” she said. “Then you’re going to educate people. We dermatologists have something to teach the world because we are experts on skin, hair, and nails. You want to impart this knowledge in a way that captivates people.” It’s like the sense of accomplishment that comes from learning something new after reading a book or watching a movie, she explained. “You feel good about it, and you can take that knowledge with you somewhere else. I love it when kids come up to me and tell me they know what a lipoma is, what a cyst is, and what psoriasis is because they’ve seen my show, or because they follow me on social media. It’s wonderful because I can see that I’ve educated them.”
Be kind and don’t activate the trolls. Dr. Lee allows positivity and kindness to rule the day on her social media content. “This is what I try to relay to followers, but I also do not engage with the negativity,” she said. “Every now and then, there will be someone who tries to insult what you do or who insults you personally. If you engage with them, it almost invites them to do it more. It almost gives them the ability to fight with you. Try to stay above that; just put out goodness and kindness.”
Several years ago, YouTube and Instagram temporarily shut down Dr. Lee’s accounts because she posted graphic images of skin lesions and procedures – a practice that wasn’t so commonplace at the time. “Don’t just post a graphic image just to be graphic,” she advised. “Make sure it has an educational message associated with it. That will help to validate your content. Posting a warning sign that some images may be graphic could help, too.”
Dr. Lee reported having no relevant financial disclosures.
FROM A LASER & AESTHETIC SKIN THERAPY COURSE