User login
Spironolactone not linked to increased cancer risk in systematic review and meta-analysis
covering seven observational studies and a total population of over 4.5 million people.
The data, published in JAMA Dermatology, are “reassuring,” the authors reported, considering that the spironolactone label carries a Food and Drug Administration warning regarding possible tumorigenicity, which is based on animal studies of doses up to 150-fold greater than doses used for humans. The drug’s antiandrogenic properties have driven its off-label use as a treatment for acne, hidradenitis, androgenetic alopecia, and hirsutism.
Spironolactone, a synthetic 17-lactone steroid, is approved for the treatment of heart failure, edema and ascites, hypertension, and primary hyperaldosteronism. Off label, it is also frequently used in gender-affirming care and is included in Endocrine Society guidelines as part of hormonal regimens for transgender women, the authors noted.
The seven eligible studies looked at the occurrence of cancer in men and women who had any exposure to the drug, regardless of the primary indication. Sample sizes ranged from 18,035 to 2.3 million, and the mean age across all studies was 62.6-72 years.
The researchers synthesized the studies, mostly of European individuals, using random effects meta-analysis and found no statistically significant association between spironolactone use and risk of breast cancer (risk ratio, 1.04; 95% confidence interval, 0.86-1.22). Three of the seven studies investigated breast cancer.
There was also no significant association between spironolactone use and risk of ovarian cancer (two studies), bladder cancer (three studies), kidney cancer (two studies), gastric cancer (two studies), or esophageal cancer (two studies).
For prostate cancer, investigated in four studies, use of the drug was associated with decreased risk (RR, 0.79, 95% CI, 0.68-0.90).
Kanthi Bommareddy, MD, of the University of Miami and coauthors concluded that all studies were at low risk of bias after appraising each one using a scale that looks at selection bias, confounding bias, and detection and outcome bias.
In dermatology, the results should “help us to take a collective sigh of relief,” said Julie C. Harper, MD, of the Dermatology and Skin Care Center of Birmingham, Ala., who was asked to comment on the study. The drug has been “safe and effective in our clinics and it is affordable and accessible to our patients,” she said, but with the FDA’s warning and the drug’s antiandrogen capacity, “there has been concern that we might be putting our patients at increased risk of breast cancer [in particular].”
The pooling of seven large studies together and the finding of no substantive increased risk of cancer “gives us evidence and comfort that spironolactone does not increase the risk of cancer in our dermatology patients,” said Dr. Harper, a past president of the American Acne & Rosacea Society.
“With every passing year,” she noted, “dermatologists are prescribing more and more spironolactone for acne, hidradenitis, androgenetic alopecia, and hirsutism.”
Four of the seven studies stratified analyses by sex, and in those without stratification by sex, women accounted for 17.2%-54.4% of the samples.
The studies had long follow-up periods of 5-20 years, but certainty of the evidence was low and since many of the studies included mostly older individuals, “they may not generalize to younger populations, such as those treated with spironolactone for acne,” the investigators wrote.
The authors also noted they were unable to look for dose-dependent associations between spironolactone and cancer risk, and that confidence intervals for rarer cancers like ovarian cancer were wide. “We cannot entirely exclude the potential for a meaningful increase in cancer risk,” and future studies are needed, in populations that include younger patients and those with acne or hirsutism.
Dr. Bommareddy reported no disclosures. Other coauthors reported grants from the National Cancer Institute outside the submitted work, and personal fees as a Cancer Prevention and Research Institute of Texas Scholar in Cancer Research. There was no funding reported for the study. Dr. Harper said she had no disclosures.
covering seven observational studies and a total population of over 4.5 million people.
The data, published in JAMA Dermatology, are “reassuring,” the authors reported, considering that the spironolactone label carries a Food and Drug Administration warning regarding possible tumorigenicity, which is based on animal studies of doses up to 150-fold greater than doses used for humans. The drug’s antiandrogenic properties have driven its off-label use as a treatment for acne, hidradenitis, androgenetic alopecia, and hirsutism.
Spironolactone, a synthetic 17-lactone steroid, is approved for the treatment of heart failure, edema and ascites, hypertension, and primary hyperaldosteronism. Off label, it is also frequently used in gender-affirming care and is included in Endocrine Society guidelines as part of hormonal regimens for transgender women, the authors noted.
The seven eligible studies looked at the occurrence of cancer in men and women who had any exposure to the drug, regardless of the primary indication. Sample sizes ranged from 18,035 to 2.3 million, and the mean age across all studies was 62.6-72 years.
The researchers synthesized the studies, mostly of European individuals, using random effects meta-analysis and found no statistically significant association between spironolactone use and risk of breast cancer (risk ratio, 1.04; 95% confidence interval, 0.86-1.22). Three of the seven studies investigated breast cancer.
There was also no significant association between spironolactone use and risk of ovarian cancer (two studies), bladder cancer (three studies), kidney cancer (two studies), gastric cancer (two studies), or esophageal cancer (two studies).
For prostate cancer, investigated in four studies, use of the drug was associated with decreased risk (RR, 0.79, 95% CI, 0.68-0.90).
Kanthi Bommareddy, MD, of the University of Miami and coauthors concluded that all studies were at low risk of bias after appraising each one using a scale that looks at selection bias, confounding bias, and detection and outcome bias.
In dermatology, the results should “help us to take a collective sigh of relief,” said Julie C. Harper, MD, of the Dermatology and Skin Care Center of Birmingham, Ala., who was asked to comment on the study. The drug has been “safe and effective in our clinics and it is affordable and accessible to our patients,” she said, but with the FDA’s warning and the drug’s antiandrogen capacity, “there has been concern that we might be putting our patients at increased risk of breast cancer [in particular].”
The pooling of seven large studies together and the finding of no substantive increased risk of cancer “gives us evidence and comfort that spironolactone does not increase the risk of cancer in our dermatology patients,” said Dr. Harper, a past president of the American Acne & Rosacea Society.
“With every passing year,” she noted, “dermatologists are prescribing more and more spironolactone for acne, hidradenitis, androgenetic alopecia, and hirsutism.”
Four of the seven studies stratified analyses by sex, and in those without stratification by sex, women accounted for 17.2%-54.4% of the samples.
The studies had long follow-up periods of 5-20 years, but certainty of the evidence was low and since many of the studies included mostly older individuals, “they may not generalize to younger populations, such as those treated with spironolactone for acne,” the investigators wrote.
The authors also noted they were unable to look for dose-dependent associations between spironolactone and cancer risk, and that confidence intervals for rarer cancers like ovarian cancer were wide. “We cannot entirely exclude the potential for a meaningful increase in cancer risk,” and future studies are needed, in populations that include younger patients and those with acne or hirsutism.
Dr. Bommareddy reported no disclosures. Other coauthors reported grants from the National Cancer Institute outside the submitted work, and personal fees as a Cancer Prevention and Research Institute of Texas Scholar in Cancer Research. There was no funding reported for the study. Dr. Harper said she had no disclosures.
covering seven observational studies and a total population of over 4.5 million people.
The data, published in JAMA Dermatology, are “reassuring,” the authors reported, considering that the spironolactone label carries a Food and Drug Administration warning regarding possible tumorigenicity, which is based on animal studies of doses up to 150-fold greater than doses used for humans. The drug’s antiandrogenic properties have driven its off-label use as a treatment for acne, hidradenitis, androgenetic alopecia, and hirsutism.
Spironolactone, a synthetic 17-lactone steroid, is approved for the treatment of heart failure, edema and ascites, hypertension, and primary hyperaldosteronism. Off label, it is also frequently used in gender-affirming care and is included in Endocrine Society guidelines as part of hormonal regimens for transgender women, the authors noted.
The seven eligible studies looked at the occurrence of cancer in men and women who had any exposure to the drug, regardless of the primary indication. Sample sizes ranged from 18,035 to 2.3 million, and the mean age across all studies was 62.6-72 years.
The researchers synthesized the studies, mostly of European individuals, using random effects meta-analysis and found no statistically significant association between spironolactone use and risk of breast cancer (risk ratio, 1.04; 95% confidence interval, 0.86-1.22). Three of the seven studies investigated breast cancer.
There was also no significant association between spironolactone use and risk of ovarian cancer (two studies), bladder cancer (three studies), kidney cancer (two studies), gastric cancer (two studies), or esophageal cancer (two studies).
For prostate cancer, investigated in four studies, use of the drug was associated with decreased risk (RR, 0.79, 95% CI, 0.68-0.90).
Kanthi Bommareddy, MD, of the University of Miami and coauthors concluded that all studies were at low risk of bias after appraising each one using a scale that looks at selection bias, confounding bias, and detection and outcome bias.
In dermatology, the results should “help us to take a collective sigh of relief,” said Julie C. Harper, MD, of the Dermatology and Skin Care Center of Birmingham, Ala., who was asked to comment on the study. The drug has been “safe and effective in our clinics and it is affordable and accessible to our patients,” she said, but with the FDA’s warning and the drug’s antiandrogen capacity, “there has been concern that we might be putting our patients at increased risk of breast cancer [in particular].”
The pooling of seven large studies together and the finding of no substantive increased risk of cancer “gives us evidence and comfort that spironolactone does not increase the risk of cancer in our dermatology patients,” said Dr. Harper, a past president of the American Acne & Rosacea Society.
“With every passing year,” she noted, “dermatologists are prescribing more and more spironolactone for acne, hidradenitis, androgenetic alopecia, and hirsutism.”
Four of the seven studies stratified analyses by sex, and in those without stratification by sex, women accounted for 17.2%-54.4% of the samples.
The studies had long follow-up periods of 5-20 years, but certainty of the evidence was low and since many of the studies included mostly older individuals, “they may not generalize to younger populations, such as those treated with spironolactone for acne,” the investigators wrote.
The authors also noted they were unable to look for dose-dependent associations between spironolactone and cancer risk, and that confidence intervals for rarer cancers like ovarian cancer were wide. “We cannot entirely exclude the potential for a meaningful increase in cancer risk,” and future studies are needed, in populations that include younger patients and those with acne or hirsutism.
Dr. Bommareddy reported no disclosures. Other coauthors reported grants from the National Cancer Institute outside the submitted work, and personal fees as a Cancer Prevention and Research Institute of Texas Scholar in Cancer Research. There was no funding reported for the study. Dr. Harper said she had no disclosures.
FROM JAMA DERMATOLOGY
Oral tofacitinib produces hair regrowth in children with alopecia areata
and published in Pediatric Dermatology.
The 11 pediatric patients, ages 8-18 years, all with a diagnosis of AA, were treated with tofacitinib. Eight patients, or nearly 73%, experienced hair regrowth, while the other three (27.3%) did not, as the investigators reported in the retrospective chart review.
“A success rate of 73% is very good,” said lead author Cory A. Dunnick, MD, professor of dermatology and director of clinical trials at the University of Colorado at Denver, Aurora. No serious adverse events occurred, and adverse events of any kind were limited, the researchers found.
“It is important to get information into the literature about potential treatments for severe alopecia areata because there is no [Food and Drug Administration]–approved therapy at the present time,” Dr. Dunnick told this news organization. Patients’ insurance plans often deny non–FDA-approved therapies unless there are data to support their use.
The researchers found no correlation between the dose, duration of treatment, or the presence of comorbidities and clinical response.
Oral tofacitinib has been shown to be effective and well tolerated for AA in adults, the researchers said. They referred to recent studies that have used JAK inhibitors, including tofacitinib, “in an effort to inhibit T-cell activation and halt disease progression in adult and pediatric patients” with AA.
Study details
Of the 11 patients evaluated, 6 had alopecia universalis, 1 had alopecia totalis, and 4 had patchy AA. Concomitant medical conditions known to be associated with AA affected four patients. These included atopic dermatitis, autoimmune hypothyroidism, and asthma. One patient reported having two brothers with AA.
The median disease duration was 6 years. “In my experience, JAK inhibitors are less effective for patients with longstanding – more than 10 years – alopecia totalis or alopecia universalis,” Dr. Dunnick said.
Previously, patients had been given methotrexate, oral prednisone, intralesional triamcinolone, topical corticosteroids, and topical diphenylcyclopropenone. During treatment with tofacitinib, 5 of the 11 patients also received topical steroid treatment.
The study was a retrospective chart review, so dosing was not standardized, the researchers said. Most took 5-10 mg twice daily. Median treatment time was 32 months, with a range of 5-39 months.
Patients with a complete or near complete clinical response were categorized as responders; subjectively, these were the patients who had persistent hair regrowth over more than 50% of affected areas. Five patients had complete regrowth of hair on the scalp, eyebrows, and body during treatment. Others had incomplete responses. For instance, one patient had improved growth of eyelashes and eyebrows but not on the scalp. Once the medication was increased to 15 mg daily, the patient had complete regrowth of body hair, eyelashes, and eyebrows but slow regrowth on the scalp after 1 year of treatment.
“Patients are very happy with regrowth of their hair,” Dr. Dunnick said, noting that severe AA affects self-esteem and quality of life.
Other research
In a retrospective study that looked at the effects of oral tofacitinib given to 14 preadolescent patients with AA, 9 experienced “clinically significant improvement” in their Severity of Alopecia Tool score. Three had complete remission, and seven (63.6%) had more than a 50% improvement in the score.
Mechanisms, concerns
The researchers of the current study explained that interferon signaling activity through the JAK pathways is a key mediator of the inflammation and cytotoxic T-cell response in AA. That modulation of the signaling may decrease disease progression, as the results of the current chart review suggest.
A main concern, the researchers wrote, is the potential for significant adverse events. Although this chart review did not find any, the researchers did see some transient lab abnormalities. One study found lab abnormalities in such measures as triglycerides and cholesterol.
Asked to comment on the study results, Brett King, MD, PHD, associate professor of dermatology at Yale University, New Haven, Conn., said that the study “is an important addition to a series of articles dating back to 2017 showing efficacy of tofacitinib in the pediatric age group.” The results are similar to those of previous studies, “showing that severe AA can be treated effectively with tofacitinib. Cumulatively, there is significant data to support treatment of this age group with JAK inhibitors,” he said.
At the 2021 European Academy of Dermatology and Venereology meeting, Dr. King presented the results of two phase 3 studies, which found that treatment with the oral JAK inhibitor baricitinib resulted in substantial hair growth in adults with AA. He and colleagues have also reported positive results of tofacitinib in treating AA in four children ages 8-10, with alopecia totalis and alopecia universalis, and in adolescents with AA.
Currently, three large, randomized, phase 3 clinical trials of other JAK inhibitors for AA are underway – ritlecitinib, baricitinib, and ruxolitinib – and the ritlecitinib trial includes adolescents (ages 12 years and older). “It is the results of these trials that we eagerly await, because FDA approval will bring greater access to these treatments,” Dr. King said.
Dr. Dunnick has disclosed no relevant financial relationships. Dr. King has served on advisory boards and/or is a consultant and/or a clinical trial investigator for AbbVie, Bristol-Myers Squibb, Concert Pharmaceuticals, Eli Lilly, Incyte, Pfizer, and others. He is on speaker bureaus for AbbVie, Incyte, Pfizer, and others.
A version of this article first appeared on Medscape.com.
and published in Pediatric Dermatology.
The 11 pediatric patients, ages 8-18 years, all with a diagnosis of AA, were treated with tofacitinib. Eight patients, or nearly 73%, experienced hair regrowth, while the other three (27.3%) did not, as the investigators reported in the retrospective chart review.
“A success rate of 73% is very good,” said lead author Cory A. Dunnick, MD, professor of dermatology and director of clinical trials at the University of Colorado at Denver, Aurora. No serious adverse events occurred, and adverse events of any kind were limited, the researchers found.
“It is important to get information into the literature about potential treatments for severe alopecia areata because there is no [Food and Drug Administration]–approved therapy at the present time,” Dr. Dunnick told this news organization. Patients’ insurance plans often deny non–FDA-approved therapies unless there are data to support their use.
The researchers found no correlation between the dose, duration of treatment, or the presence of comorbidities and clinical response.
Oral tofacitinib has been shown to be effective and well tolerated for AA in adults, the researchers said. They referred to recent studies that have used JAK inhibitors, including tofacitinib, “in an effort to inhibit T-cell activation and halt disease progression in adult and pediatric patients” with AA.
Study details
Of the 11 patients evaluated, 6 had alopecia universalis, 1 had alopecia totalis, and 4 had patchy AA. Concomitant medical conditions known to be associated with AA affected four patients. These included atopic dermatitis, autoimmune hypothyroidism, and asthma. One patient reported having two brothers with AA.
The median disease duration was 6 years. “In my experience, JAK inhibitors are less effective for patients with longstanding – more than 10 years – alopecia totalis or alopecia universalis,” Dr. Dunnick said.
Previously, patients had been given methotrexate, oral prednisone, intralesional triamcinolone, topical corticosteroids, and topical diphenylcyclopropenone. During treatment with tofacitinib, 5 of the 11 patients also received topical steroid treatment.
The study was a retrospective chart review, so dosing was not standardized, the researchers said. Most took 5-10 mg twice daily. Median treatment time was 32 months, with a range of 5-39 months.
Patients with a complete or near complete clinical response were categorized as responders; subjectively, these were the patients who had persistent hair regrowth over more than 50% of affected areas. Five patients had complete regrowth of hair on the scalp, eyebrows, and body during treatment. Others had incomplete responses. For instance, one patient had improved growth of eyelashes and eyebrows but not on the scalp. Once the medication was increased to 15 mg daily, the patient had complete regrowth of body hair, eyelashes, and eyebrows but slow regrowth on the scalp after 1 year of treatment.
“Patients are very happy with regrowth of their hair,” Dr. Dunnick said, noting that severe AA affects self-esteem and quality of life.
Other research
In a retrospective study that looked at the effects of oral tofacitinib given to 14 preadolescent patients with AA, 9 experienced “clinically significant improvement” in their Severity of Alopecia Tool score. Three had complete remission, and seven (63.6%) had more than a 50% improvement in the score.
Mechanisms, concerns
The researchers of the current study explained that interferon signaling activity through the JAK pathways is a key mediator of the inflammation and cytotoxic T-cell response in AA. That modulation of the signaling may decrease disease progression, as the results of the current chart review suggest.
A main concern, the researchers wrote, is the potential for significant adverse events. Although this chart review did not find any, the researchers did see some transient lab abnormalities. One study found lab abnormalities in such measures as triglycerides and cholesterol.
Asked to comment on the study results, Brett King, MD, PHD, associate professor of dermatology at Yale University, New Haven, Conn., said that the study “is an important addition to a series of articles dating back to 2017 showing efficacy of tofacitinib in the pediatric age group.” The results are similar to those of previous studies, “showing that severe AA can be treated effectively with tofacitinib. Cumulatively, there is significant data to support treatment of this age group with JAK inhibitors,” he said.
At the 2021 European Academy of Dermatology and Venereology meeting, Dr. King presented the results of two phase 3 studies, which found that treatment with the oral JAK inhibitor baricitinib resulted in substantial hair growth in adults with AA. He and colleagues have also reported positive results of tofacitinib in treating AA in four children ages 8-10, with alopecia totalis and alopecia universalis, and in adolescents with AA.
Currently, three large, randomized, phase 3 clinical trials of other JAK inhibitors for AA are underway – ritlecitinib, baricitinib, and ruxolitinib – and the ritlecitinib trial includes adolescents (ages 12 years and older). “It is the results of these trials that we eagerly await, because FDA approval will bring greater access to these treatments,” Dr. King said.
Dr. Dunnick has disclosed no relevant financial relationships. Dr. King has served on advisory boards and/or is a consultant and/or a clinical trial investigator for AbbVie, Bristol-Myers Squibb, Concert Pharmaceuticals, Eli Lilly, Incyte, Pfizer, and others. He is on speaker bureaus for AbbVie, Incyte, Pfizer, and others.
A version of this article first appeared on Medscape.com.
and published in Pediatric Dermatology.
The 11 pediatric patients, ages 8-18 years, all with a diagnosis of AA, were treated with tofacitinib. Eight patients, or nearly 73%, experienced hair regrowth, while the other three (27.3%) did not, as the investigators reported in the retrospective chart review.
“A success rate of 73% is very good,” said lead author Cory A. Dunnick, MD, professor of dermatology and director of clinical trials at the University of Colorado at Denver, Aurora. No serious adverse events occurred, and adverse events of any kind were limited, the researchers found.
“It is important to get information into the literature about potential treatments for severe alopecia areata because there is no [Food and Drug Administration]–approved therapy at the present time,” Dr. Dunnick told this news organization. Patients’ insurance plans often deny non–FDA-approved therapies unless there are data to support their use.
The researchers found no correlation between the dose, duration of treatment, or the presence of comorbidities and clinical response.
Oral tofacitinib has been shown to be effective and well tolerated for AA in adults, the researchers said. They referred to recent studies that have used JAK inhibitors, including tofacitinib, “in an effort to inhibit T-cell activation and halt disease progression in adult and pediatric patients” with AA.
Study details
Of the 11 patients evaluated, 6 had alopecia universalis, 1 had alopecia totalis, and 4 had patchy AA. Concomitant medical conditions known to be associated with AA affected four patients. These included atopic dermatitis, autoimmune hypothyroidism, and asthma. One patient reported having two brothers with AA.
The median disease duration was 6 years. “In my experience, JAK inhibitors are less effective for patients with longstanding – more than 10 years – alopecia totalis or alopecia universalis,” Dr. Dunnick said.
Previously, patients had been given methotrexate, oral prednisone, intralesional triamcinolone, topical corticosteroids, and topical diphenylcyclopropenone. During treatment with tofacitinib, 5 of the 11 patients also received topical steroid treatment.
The study was a retrospective chart review, so dosing was not standardized, the researchers said. Most took 5-10 mg twice daily. Median treatment time was 32 months, with a range of 5-39 months.
Patients with a complete or near complete clinical response were categorized as responders; subjectively, these were the patients who had persistent hair regrowth over more than 50% of affected areas. Five patients had complete regrowth of hair on the scalp, eyebrows, and body during treatment. Others had incomplete responses. For instance, one patient had improved growth of eyelashes and eyebrows but not on the scalp. Once the medication was increased to 15 mg daily, the patient had complete regrowth of body hair, eyelashes, and eyebrows but slow regrowth on the scalp after 1 year of treatment.
“Patients are very happy with regrowth of their hair,” Dr. Dunnick said, noting that severe AA affects self-esteem and quality of life.
Other research
In a retrospective study that looked at the effects of oral tofacitinib given to 14 preadolescent patients with AA, 9 experienced “clinically significant improvement” in their Severity of Alopecia Tool score. Three had complete remission, and seven (63.6%) had more than a 50% improvement in the score.
Mechanisms, concerns
The researchers of the current study explained that interferon signaling activity through the JAK pathways is a key mediator of the inflammation and cytotoxic T-cell response in AA. That modulation of the signaling may decrease disease progression, as the results of the current chart review suggest.
A main concern, the researchers wrote, is the potential for significant adverse events. Although this chart review did not find any, the researchers did see some transient lab abnormalities. One study found lab abnormalities in such measures as triglycerides and cholesterol.
Asked to comment on the study results, Brett King, MD, PHD, associate professor of dermatology at Yale University, New Haven, Conn., said that the study “is an important addition to a series of articles dating back to 2017 showing efficacy of tofacitinib in the pediatric age group.” The results are similar to those of previous studies, “showing that severe AA can be treated effectively with tofacitinib. Cumulatively, there is significant data to support treatment of this age group with JAK inhibitors,” he said.
At the 2021 European Academy of Dermatology and Venereology meeting, Dr. King presented the results of two phase 3 studies, which found that treatment with the oral JAK inhibitor baricitinib resulted in substantial hair growth in adults with AA. He and colleagues have also reported positive results of tofacitinib in treating AA in four children ages 8-10, with alopecia totalis and alopecia universalis, and in adolescents with AA.
Currently, three large, randomized, phase 3 clinical trials of other JAK inhibitors for AA are underway – ritlecitinib, baricitinib, and ruxolitinib – and the ritlecitinib trial includes adolescents (ages 12 years and older). “It is the results of these trials that we eagerly await, because FDA approval will bring greater access to these treatments,” Dr. King said.
Dr. Dunnick has disclosed no relevant financial relationships. Dr. King has served on advisory boards and/or is a consultant and/or a clinical trial investigator for AbbVie, Bristol-Myers Squibb, Concert Pharmaceuticals, Eli Lilly, Incyte, Pfizer, and others. He is on speaker bureaus for AbbVie, Incyte, Pfizer, and others.
A version of this article first appeared on Medscape.com.
FROM PEDIATRIC DERMATOLOGY
Urticaria: An elusive disorder with ‘a high unmet need for treatment’
In the clinical experience of Adam Friedman, MD, when patients present with acute urticaria, the culprit is usually food, a drug, or a bug.
But in some cases, the trigger remains elusive. “We don’t always find the source, but don’t beat yourself up about it,” Dr. Friedman, professor and chair of dermatology at George Washington University, Washington, said at the ODAC Dermatology, Aesthetic, and Surgical Conference. “The basic rule is to treat patients to clearance and keep them clear.”
Chronic urticaria is characterized by plaques with a burning/itch sensation that often “move” to different locations on the body over minutes to hours, and they typically last for less than 24 hours. The plaques are often oval, round, or irregular in shape and they typically leave no postinflammatory pigmentary alteration or scarring other than from scratching.
Urticaria affects an estimated 20% of the population, Dr. Friedman said, and is more common in females than males. More than two-thirds of cases are self-limiting but 10% can persist longer than 5 years. Acute episodes are more likely to have an identifiable trigger, while chronic episodes, which last more than six weeks, typically do not. The longer the duration, the lower the chance of identifying the root cause. The foods/food products most commonly affecting children with acute urticaria include milk, egg, peanut, wheat, and soy, while the common culprits in adults are tree nuts, peanuts, and shellfish. Other triggers include the yellow food dye annatto, the red food dye carmine, contact with raw fruits or vegetables, animal saliva, and certain detergents or perfume.
“When you have no idea what the cause is for acute urticaria, I think about viral or bacterial infections, especially in children,” Dr. Friedman said, particularly mycoplasma, adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus. COVID-19 has also been a new etiologic source for a recent rise in acute urticaria cases.
Other causes include certain medications such as antibiotics, opiates, muscle relaxants, salicylates, and NSAIDs; stinging insects; and exposure to latex products, which can cross react with passion fruit, banana, avocado, chestnut, and kiwi. Alcohol consumption can also trigger urticaria.
“Ask patients if they have joint discomfort or pain,” Dr. Friedman advised, referring to urticaria arthritis syndrome that is typically seen more often in women than in men. “It’s rare but important, because that may distinguish for you what is needed to get those patients under control.”
which spontaneously occurs after an exposure to an external force. “The distinguishing feature here is that it doesn’t last long – 30 minutes or so – and is typically unresponsive to corticosteroids,” Dr. Friedman said. “It comes on quickly but disappears quickly whereas with chronic spontaneous urticaria, someone might be getting those wheals of flare for hours and hours.”
The most common form of physical urticaria is dermatographism, while other examples include physical urticaria resulting from exposure to cholinergic agents, heat, exercise, cold, water, sunlight, and pressure on the skin.
About half of patients with chronic urticaria are disease free within 1 year, but 20% continue to experience episodes for more than 10 years. One study found that patients with chronic spontaneous urticaria who were diagnosed at a younger age trended toward a longer disease course, and rates were higher in women, compared with men. “Perceived stress can make this worse,” Dr. Friedman added.
According to Dr. Friedman, it’s more important to ask patients targeted questions during office visits than it is to do a full workup. “I ask patients to keep a diary, which can help them identify triggers if there are any,” he said. “I also ask them to take a picture of the papules with their smartphone. There can be a genetic association, so it’s important to ask if anyone else in the family has urticaria. No routine lab tests are required unless there’s something in the history that suggests it’s worthwhile. Let the patient guide the diagnostic workup; don’t just order a million tests.”
That said, known comorbidities associated with urticaria include autoimmune disease, atopy, infections, metabolic conditions, and neoplastic disorders. “Biopsies are typically useless because this is an invisible dermatosis,” he said. “They’re useful when it’s urticarial, not urticaria, when you’re trying to figure out what it is.”
According to recently published international guidelines on urticaria, published in September 2021, the recommended first line of treatment for urticaria is with second-generation nonsedating antihistamines such as cetirizine and loratadine, up to four times the recommended dose.
Second-generation derivatives include desloratadine, levocetirizine, and fexofenadine. “I like using fexofenadine in the morning for folks who don’t tolerate cetirizine, then I’ll recommend something a little more sedating at night,” Dr. Friedman said. “We max out [the dose] by week 4. If it works, great. If not, we move on to something else.”
In late 2021, the British Association of Dermatologists also published guidelines on the treatment of chronic urticaria.
As for markers of treatment success, a study of 240 children with chronic spontaneous urticaria found that risk factors for a poor response included longer duration of disease, higher treatment step until initial disease control, and food sensitization.
Vitamin D supplementation may also add some benefit. One study of 42 adults with urticaria found that low and high doses of vitamin D added to antihistamine therapy can boost effectiveness. “This may be because vitamin D could be a marker of severity,” Dr. Friedman said. “The reality is, however, that a lot of patients don’t do well.”
Data from the large, prospective study known as AWARE (A World-Wide Antihistamine-Refractory Chronic Urticaria Patient Evaluation) found that 23% patients treated with nonsedating H1-antihistamines and 42% patients treated with up-dosed nonsedating H1-antihistamines had uncontrolled chronic spontaneous urticaria at month 24.
A second-line treatment option for patients aged 6 and older is the anti-IgE antibody omalizumab, 150-300 mg by subcutaneous injection every 4 weeks. Dr. Friedman typically uses only the 300-mg dose. “You do not need to take pretreatment serum IgE levels,” he said. “The most significant adverse event is anaphylaxis, which only affects 0.2% of patients.”
A third-line option is cyclosporine A. A dose of 3-5mg/kg per day appears to benefit about two-thirds of patients with antihistamine recalcitrant chronic urticaria. “It works fast but you can’t keep patients on it for very long,” he said.
Another third-line option is mycophenolate mofetil, which may work by inhibiting the production of autoantibodies to the high-affinity IgE receptor and/or IgE. “It does work well, especially in conjunction with antihistamines; it’s kind of a softer immunosuppressant,” he said. Methotrexate can also be used as an add-on therapy to H1 antihistamine therapy in difficult-to-treat cases.
“It’s great we have [a Food and Drug Administration]–approved biologic therapy in omalizumab and access to over-the-counter antihistamines, but there is a high unmet need for treatment,” and a need for new therapies, Dr. Friedman said. “Only about 39% achieve symptomatic control with conventional dosing of antihistamines, and 63% of nonresponders achieve symptom control with a fourfold increased dosing of antihistamines.” In addition, about 20% of patients will not respond to either standard or increased doses of antihistamines and are eligible for treatment with omalizumab. However, more than 50% of such patients experience a delay or lack of response to omalizumab. “We need innovation; we need to understand the disease better,” he said.
Dr. Friedman disclosed that he serves as a consultant and/or adviser for Loreal, La Roche Posay, Cerave, Galderma, Aveeno, Microcures, Pfizer, Novartis, Dermira, Brickell Biotech, Incyte, UCB, Janssen, Pfizer, Bristol-Myers Squibb, Almirall, Zylo Therapeutics, Hoth Therapeutics, Corbus, Greenway Therapeutics, TruPotency, and Dermavant. He is a speaker for Regeneron/Sanofi, AbbVie, Janssen, Brickell Biotech, and Incyte, and has received grants from Pfizer, the Dermatology Foundation, Incyte, and Galderma.
In the clinical experience of Adam Friedman, MD, when patients present with acute urticaria, the culprit is usually food, a drug, or a bug.
But in some cases, the trigger remains elusive. “We don’t always find the source, but don’t beat yourself up about it,” Dr. Friedman, professor and chair of dermatology at George Washington University, Washington, said at the ODAC Dermatology, Aesthetic, and Surgical Conference. “The basic rule is to treat patients to clearance and keep them clear.”
Chronic urticaria is characterized by plaques with a burning/itch sensation that often “move” to different locations on the body over minutes to hours, and they typically last for less than 24 hours. The plaques are often oval, round, or irregular in shape and they typically leave no postinflammatory pigmentary alteration or scarring other than from scratching.
Urticaria affects an estimated 20% of the population, Dr. Friedman said, and is more common in females than males. More than two-thirds of cases are self-limiting but 10% can persist longer than 5 years. Acute episodes are more likely to have an identifiable trigger, while chronic episodes, which last more than six weeks, typically do not. The longer the duration, the lower the chance of identifying the root cause. The foods/food products most commonly affecting children with acute urticaria include milk, egg, peanut, wheat, and soy, while the common culprits in adults are tree nuts, peanuts, and shellfish. Other triggers include the yellow food dye annatto, the red food dye carmine, contact with raw fruits or vegetables, animal saliva, and certain detergents or perfume.
“When you have no idea what the cause is for acute urticaria, I think about viral or bacterial infections, especially in children,” Dr. Friedman said, particularly mycoplasma, adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus. COVID-19 has also been a new etiologic source for a recent rise in acute urticaria cases.
Other causes include certain medications such as antibiotics, opiates, muscle relaxants, salicylates, and NSAIDs; stinging insects; and exposure to latex products, which can cross react with passion fruit, banana, avocado, chestnut, and kiwi. Alcohol consumption can also trigger urticaria.
“Ask patients if they have joint discomfort or pain,” Dr. Friedman advised, referring to urticaria arthritis syndrome that is typically seen more often in women than in men. “It’s rare but important, because that may distinguish for you what is needed to get those patients under control.”
which spontaneously occurs after an exposure to an external force. “The distinguishing feature here is that it doesn’t last long – 30 minutes or so – and is typically unresponsive to corticosteroids,” Dr. Friedman said. “It comes on quickly but disappears quickly whereas with chronic spontaneous urticaria, someone might be getting those wheals of flare for hours and hours.”
The most common form of physical urticaria is dermatographism, while other examples include physical urticaria resulting from exposure to cholinergic agents, heat, exercise, cold, water, sunlight, and pressure on the skin.
About half of patients with chronic urticaria are disease free within 1 year, but 20% continue to experience episodes for more than 10 years. One study found that patients with chronic spontaneous urticaria who were diagnosed at a younger age trended toward a longer disease course, and rates were higher in women, compared with men. “Perceived stress can make this worse,” Dr. Friedman added.
According to Dr. Friedman, it’s more important to ask patients targeted questions during office visits than it is to do a full workup. “I ask patients to keep a diary, which can help them identify triggers if there are any,” he said. “I also ask them to take a picture of the papules with their smartphone. There can be a genetic association, so it’s important to ask if anyone else in the family has urticaria. No routine lab tests are required unless there’s something in the history that suggests it’s worthwhile. Let the patient guide the diagnostic workup; don’t just order a million tests.”
That said, known comorbidities associated with urticaria include autoimmune disease, atopy, infections, metabolic conditions, and neoplastic disorders. “Biopsies are typically useless because this is an invisible dermatosis,” he said. “They’re useful when it’s urticarial, not urticaria, when you’re trying to figure out what it is.”
According to recently published international guidelines on urticaria, published in September 2021, the recommended first line of treatment for urticaria is with second-generation nonsedating antihistamines such as cetirizine and loratadine, up to four times the recommended dose.
Second-generation derivatives include desloratadine, levocetirizine, and fexofenadine. “I like using fexofenadine in the morning for folks who don’t tolerate cetirizine, then I’ll recommend something a little more sedating at night,” Dr. Friedman said. “We max out [the dose] by week 4. If it works, great. If not, we move on to something else.”
In late 2021, the British Association of Dermatologists also published guidelines on the treatment of chronic urticaria.
As for markers of treatment success, a study of 240 children with chronic spontaneous urticaria found that risk factors for a poor response included longer duration of disease, higher treatment step until initial disease control, and food sensitization.
Vitamin D supplementation may also add some benefit. One study of 42 adults with urticaria found that low and high doses of vitamin D added to antihistamine therapy can boost effectiveness. “This may be because vitamin D could be a marker of severity,” Dr. Friedman said. “The reality is, however, that a lot of patients don’t do well.”
Data from the large, prospective study known as AWARE (A World-Wide Antihistamine-Refractory Chronic Urticaria Patient Evaluation) found that 23% patients treated with nonsedating H1-antihistamines and 42% patients treated with up-dosed nonsedating H1-antihistamines had uncontrolled chronic spontaneous urticaria at month 24.
A second-line treatment option for patients aged 6 and older is the anti-IgE antibody omalizumab, 150-300 mg by subcutaneous injection every 4 weeks. Dr. Friedman typically uses only the 300-mg dose. “You do not need to take pretreatment serum IgE levels,” he said. “The most significant adverse event is anaphylaxis, which only affects 0.2% of patients.”
A third-line option is cyclosporine A. A dose of 3-5mg/kg per day appears to benefit about two-thirds of patients with antihistamine recalcitrant chronic urticaria. “It works fast but you can’t keep patients on it for very long,” he said.
Another third-line option is mycophenolate mofetil, which may work by inhibiting the production of autoantibodies to the high-affinity IgE receptor and/or IgE. “It does work well, especially in conjunction with antihistamines; it’s kind of a softer immunosuppressant,” he said. Methotrexate can also be used as an add-on therapy to H1 antihistamine therapy in difficult-to-treat cases.
“It’s great we have [a Food and Drug Administration]–approved biologic therapy in omalizumab and access to over-the-counter antihistamines, but there is a high unmet need for treatment,” and a need for new therapies, Dr. Friedman said. “Only about 39% achieve symptomatic control with conventional dosing of antihistamines, and 63% of nonresponders achieve symptom control with a fourfold increased dosing of antihistamines.” In addition, about 20% of patients will not respond to either standard or increased doses of antihistamines and are eligible for treatment with omalizumab. However, more than 50% of such patients experience a delay or lack of response to omalizumab. “We need innovation; we need to understand the disease better,” he said.
Dr. Friedman disclosed that he serves as a consultant and/or adviser for Loreal, La Roche Posay, Cerave, Galderma, Aveeno, Microcures, Pfizer, Novartis, Dermira, Brickell Biotech, Incyte, UCB, Janssen, Pfizer, Bristol-Myers Squibb, Almirall, Zylo Therapeutics, Hoth Therapeutics, Corbus, Greenway Therapeutics, TruPotency, and Dermavant. He is a speaker for Regeneron/Sanofi, AbbVie, Janssen, Brickell Biotech, and Incyte, and has received grants from Pfizer, the Dermatology Foundation, Incyte, and Galderma.
In the clinical experience of Adam Friedman, MD, when patients present with acute urticaria, the culprit is usually food, a drug, or a bug.
But in some cases, the trigger remains elusive. “We don’t always find the source, but don’t beat yourself up about it,” Dr. Friedman, professor and chair of dermatology at George Washington University, Washington, said at the ODAC Dermatology, Aesthetic, and Surgical Conference. “The basic rule is to treat patients to clearance and keep them clear.”
Chronic urticaria is characterized by plaques with a burning/itch sensation that often “move” to different locations on the body over minutes to hours, and they typically last for less than 24 hours. The plaques are often oval, round, or irregular in shape and they typically leave no postinflammatory pigmentary alteration or scarring other than from scratching.
Urticaria affects an estimated 20% of the population, Dr. Friedman said, and is more common in females than males. More than two-thirds of cases are self-limiting but 10% can persist longer than 5 years. Acute episodes are more likely to have an identifiable trigger, while chronic episodes, which last more than six weeks, typically do not. The longer the duration, the lower the chance of identifying the root cause. The foods/food products most commonly affecting children with acute urticaria include milk, egg, peanut, wheat, and soy, while the common culprits in adults are tree nuts, peanuts, and shellfish. Other triggers include the yellow food dye annatto, the red food dye carmine, contact with raw fruits or vegetables, animal saliva, and certain detergents or perfume.
“When you have no idea what the cause is for acute urticaria, I think about viral or bacterial infections, especially in children,” Dr. Friedman said, particularly mycoplasma, adenovirus, enterovirus, rotavirus, respiratory syncytial virus, Epstein-Barr virus, and cytomegalovirus. COVID-19 has also been a new etiologic source for a recent rise in acute urticaria cases.
Other causes include certain medications such as antibiotics, opiates, muscle relaxants, salicylates, and NSAIDs; stinging insects; and exposure to latex products, which can cross react with passion fruit, banana, avocado, chestnut, and kiwi. Alcohol consumption can also trigger urticaria.
“Ask patients if they have joint discomfort or pain,” Dr. Friedman advised, referring to urticaria arthritis syndrome that is typically seen more often in women than in men. “It’s rare but important, because that may distinguish for you what is needed to get those patients under control.”
which spontaneously occurs after an exposure to an external force. “The distinguishing feature here is that it doesn’t last long – 30 minutes or so – and is typically unresponsive to corticosteroids,” Dr. Friedman said. “It comes on quickly but disappears quickly whereas with chronic spontaneous urticaria, someone might be getting those wheals of flare for hours and hours.”
The most common form of physical urticaria is dermatographism, while other examples include physical urticaria resulting from exposure to cholinergic agents, heat, exercise, cold, water, sunlight, and pressure on the skin.
About half of patients with chronic urticaria are disease free within 1 year, but 20% continue to experience episodes for more than 10 years. One study found that patients with chronic spontaneous urticaria who were diagnosed at a younger age trended toward a longer disease course, and rates were higher in women, compared with men. “Perceived stress can make this worse,” Dr. Friedman added.
According to Dr. Friedman, it’s more important to ask patients targeted questions during office visits than it is to do a full workup. “I ask patients to keep a diary, which can help them identify triggers if there are any,” he said. “I also ask them to take a picture of the papules with their smartphone. There can be a genetic association, so it’s important to ask if anyone else in the family has urticaria. No routine lab tests are required unless there’s something in the history that suggests it’s worthwhile. Let the patient guide the diagnostic workup; don’t just order a million tests.”
That said, known comorbidities associated with urticaria include autoimmune disease, atopy, infections, metabolic conditions, and neoplastic disorders. “Biopsies are typically useless because this is an invisible dermatosis,” he said. “They’re useful when it’s urticarial, not urticaria, when you’re trying to figure out what it is.”
According to recently published international guidelines on urticaria, published in September 2021, the recommended first line of treatment for urticaria is with second-generation nonsedating antihistamines such as cetirizine and loratadine, up to four times the recommended dose.
Second-generation derivatives include desloratadine, levocetirizine, and fexofenadine. “I like using fexofenadine in the morning for folks who don’t tolerate cetirizine, then I’ll recommend something a little more sedating at night,” Dr. Friedman said. “We max out [the dose] by week 4. If it works, great. If not, we move on to something else.”
In late 2021, the British Association of Dermatologists also published guidelines on the treatment of chronic urticaria.
As for markers of treatment success, a study of 240 children with chronic spontaneous urticaria found that risk factors for a poor response included longer duration of disease, higher treatment step until initial disease control, and food sensitization.
Vitamin D supplementation may also add some benefit. One study of 42 adults with urticaria found that low and high doses of vitamin D added to antihistamine therapy can boost effectiveness. “This may be because vitamin D could be a marker of severity,” Dr. Friedman said. “The reality is, however, that a lot of patients don’t do well.”
Data from the large, prospective study known as AWARE (A World-Wide Antihistamine-Refractory Chronic Urticaria Patient Evaluation) found that 23% patients treated with nonsedating H1-antihistamines and 42% patients treated with up-dosed nonsedating H1-antihistamines had uncontrolled chronic spontaneous urticaria at month 24.
A second-line treatment option for patients aged 6 and older is the anti-IgE antibody omalizumab, 150-300 mg by subcutaneous injection every 4 weeks. Dr. Friedman typically uses only the 300-mg dose. “You do not need to take pretreatment serum IgE levels,” he said. “The most significant adverse event is anaphylaxis, which only affects 0.2% of patients.”
A third-line option is cyclosporine A. A dose of 3-5mg/kg per day appears to benefit about two-thirds of patients with antihistamine recalcitrant chronic urticaria. “It works fast but you can’t keep patients on it for very long,” he said.
Another third-line option is mycophenolate mofetil, which may work by inhibiting the production of autoantibodies to the high-affinity IgE receptor and/or IgE. “It does work well, especially in conjunction with antihistamines; it’s kind of a softer immunosuppressant,” he said. Methotrexate can also be used as an add-on therapy to H1 antihistamine therapy in difficult-to-treat cases.
“It’s great we have [a Food and Drug Administration]–approved biologic therapy in omalizumab and access to over-the-counter antihistamines, but there is a high unmet need for treatment,” and a need for new therapies, Dr. Friedman said. “Only about 39% achieve symptomatic control with conventional dosing of antihistamines, and 63% of nonresponders achieve symptom control with a fourfold increased dosing of antihistamines.” In addition, about 20% of patients will not respond to either standard or increased doses of antihistamines and are eligible for treatment with omalizumab. However, more than 50% of such patients experience a delay or lack of response to omalizumab. “We need innovation; we need to understand the disease better,” he said.
Dr. Friedman disclosed that he serves as a consultant and/or adviser for Loreal, La Roche Posay, Cerave, Galderma, Aveeno, Microcures, Pfizer, Novartis, Dermira, Brickell Biotech, Incyte, UCB, Janssen, Pfizer, Bristol-Myers Squibb, Almirall, Zylo Therapeutics, Hoth Therapeutics, Corbus, Greenway Therapeutics, TruPotency, and Dermavant. He is a speaker for Regeneron/Sanofi, AbbVie, Janssen, Brickell Biotech, and Incyte, and has received grants from Pfizer, the Dermatology Foundation, Incyte, and Galderma.
FROM ODAC 2022
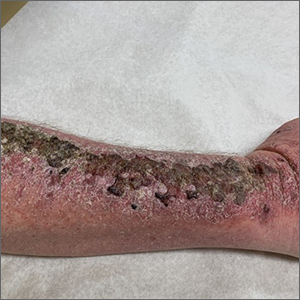
Right arm rash
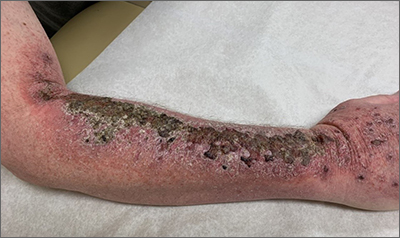
Common skin reactions to treatment with topical 5-FU for actinic keratosis include erythema, ulceration, and burning. In this case, however, the skin disruption opened the door to secondary impetigo.
Secondary skin infections are a known risk of treatment with topical 5-FU. The agent inhibits thymidylate synthetase, an enzyme involved in the synthesis and repair of DNA. The inhibition of this enzyme can lead to skin disruption with erosion, desquamation, and the risk of superimposed skin infections, as was seen with this patient.1
Impetigo is a common skin infection affecting the superficial layers of the epidermis and is most commonly caused by gram-positive bacteria, such as Staphylococcus aureus or Streptococcus pyogenes.2 Secondary impetigo, also known as impetiginization, is an infection of previously disrupted skin due to eczema, trauma, insect bites, and other conditions. This contrasts with primary impetigo, which results from a direct bacterial invasion of intact healthy skin. While impetigo predominantly affects children between the ages of 2 and 5 years, people of any age can be affected.2 Impetigo characteristically manifests with painful erosions, classically covered by honey-colored crusts. Thin-walled vesicles often appear and subsequently rupture.
Treatment options for impetigo include both topical and systemic antibiotics. Topical therapy is preferred for patients with limited skin involvement, while systemic therapy is indicated for patients with numerous lesions. Mupirocin and retapamulin are first-line topical treatments. Systemic antibiotic therapy should provide coverage for both S. aureus and streptococcal infections; cephalexin and dicloxacillin are preferred. Doxycycline, trimethoprim-sulfamethoxazole, or clindamycin can be used if methicillin-resistant Staphylococcus aureus is suspected.3
This patient was advised to try warm soaks (to reduce the crusting) and to follow that with the application of white petrolatum bid. The patient was also prescribed doxycycline 100 mg orally bid for 10 days. At the 1-month follow-up, there was some residual erythema, but the impetigo and crusting had resolved. The actinic keratoses had resolved, as well.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Tess Pei Lemon, BA, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque
1. Chughtai K, Gupta R, Upadhaya S, et al. Topical 5-Fluorouracil associated skin reaction. Oxf Med Case Rep. 2017;2017(8):omx043. doi:10.1093/omcr/omx043
2. Hartman-Adams H, Banvard C, Juckett G. Impetigo: diagnosis and treatment. Am Fam Physician. 2014;90:229-235.
3. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59:147-159. doi:10.1093/cid/ciu296

Common skin reactions to treatment with topical 5-FU for actinic keratosis include erythema, ulceration, and burning. In this case, however, the skin disruption opened the door to secondary impetigo.
Secondary skin infections are a known risk of treatment with topical 5-FU. The agent inhibits thymidylate synthetase, an enzyme involved in the synthesis and repair of DNA. The inhibition of this enzyme can lead to skin disruption with erosion, desquamation, and the risk of superimposed skin infections, as was seen with this patient.1
Impetigo is a common skin infection affecting the superficial layers of the epidermis and is most commonly caused by gram-positive bacteria, such as Staphylococcus aureus or Streptococcus pyogenes.2 Secondary impetigo, also known as impetiginization, is an infection of previously disrupted skin due to eczema, trauma, insect bites, and other conditions. This contrasts with primary impetigo, which results from a direct bacterial invasion of intact healthy skin. While impetigo predominantly affects children between the ages of 2 and 5 years, people of any age can be affected.2 Impetigo characteristically manifests with painful erosions, classically covered by honey-colored crusts. Thin-walled vesicles often appear and subsequently rupture.
Treatment options for impetigo include both topical and systemic antibiotics. Topical therapy is preferred for patients with limited skin involvement, while systemic therapy is indicated for patients with numerous lesions. Mupirocin and retapamulin are first-line topical treatments. Systemic antibiotic therapy should provide coverage for both S. aureus and streptococcal infections; cephalexin and dicloxacillin are preferred. Doxycycline, trimethoprim-sulfamethoxazole, or clindamycin can be used if methicillin-resistant Staphylococcus aureus is suspected.3
This patient was advised to try warm soaks (to reduce the crusting) and to follow that with the application of white petrolatum bid. The patient was also prescribed doxycycline 100 mg orally bid for 10 days. At the 1-month follow-up, there was some residual erythema, but the impetigo and crusting had resolved. The actinic keratoses had resolved, as well.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Tess Pei Lemon, BA, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque

Common skin reactions to treatment with topical 5-FU for actinic keratosis include erythema, ulceration, and burning. In this case, however, the skin disruption opened the door to secondary impetigo.
Secondary skin infections are a known risk of treatment with topical 5-FU. The agent inhibits thymidylate synthetase, an enzyme involved in the synthesis and repair of DNA. The inhibition of this enzyme can lead to skin disruption with erosion, desquamation, and the risk of superimposed skin infections, as was seen with this patient.1
Impetigo is a common skin infection affecting the superficial layers of the epidermis and is most commonly caused by gram-positive bacteria, such as Staphylococcus aureus or Streptococcus pyogenes.2 Secondary impetigo, also known as impetiginization, is an infection of previously disrupted skin due to eczema, trauma, insect bites, and other conditions. This contrasts with primary impetigo, which results from a direct bacterial invasion of intact healthy skin. While impetigo predominantly affects children between the ages of 2 and 5 years, people of any age can be affected.2 Impetigo characteristically manifests with painful erosions, classically covered by honey-colored crusts. Thin-walled vesicles often appear and subsequently rupture.
Treatment options for impetigo include both topical and systemic antibiotics. Topical therapy is preferred for patients with limited skin involvement, while systemic therapy is indicated for patients with numerous lesions. Mupirocin and retapamulin are first-line topical treatments. Systemic antibiotic therapy should provide coverage for both S. aureus and streptococcal infections; cephalexin and dicloxacillin are preferred. Doxycycline, trimethoprim-sulfamethoxazole, or clindamycin can be used if methicillin-resistant Staphylococcus aureus is suspected.3
This patient was advised to try warm soaks (to reduce the crusting) and to follow that with the application of white petrolatum bid. The patient was also prescribed doxycycline 100 mg orally bid for 10 days. At the 1-month follow-up, there was some residual erythema, but the impetigo and crusting had resolved. The actinic keratoses had resolved, as well.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Tess Pei Lemon, BA, University of New Mexico School of Medicine and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque
1. Chughtai K, Gupta R, Upadhaya S, et al. Topical 5-Fluorouracil associated skin reaction. Oxf Med Case Rep. 2017;2017(8):omx043. doi:10.1093/omcr/omx043
2. Hartman-Adams H, Banvard C, Juckett G. Impetigo: diagnosis and treatment. Am Fam Physician. 2014;90:229-235.
3. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59:147-159. doi:10.1093/cid/ciu296
1. Chughtai K, Gupta R, Upadhaya S, et al. Topical 5-Fluorouracil associated skin reaction. Oxf Med Case Rep. 2017;2017(8):omx043. doi:10.1093/omcr/omx043
2. Hartman-Adams H, Banvard C, Juckett G. Impetigo: diagnosis and treatment. Am Fam Physician. 2014;90:229-235.
3. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59:147-159. doi:10.1093/cid/ciu296
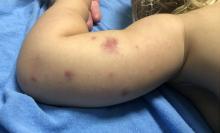
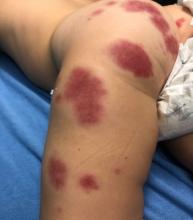
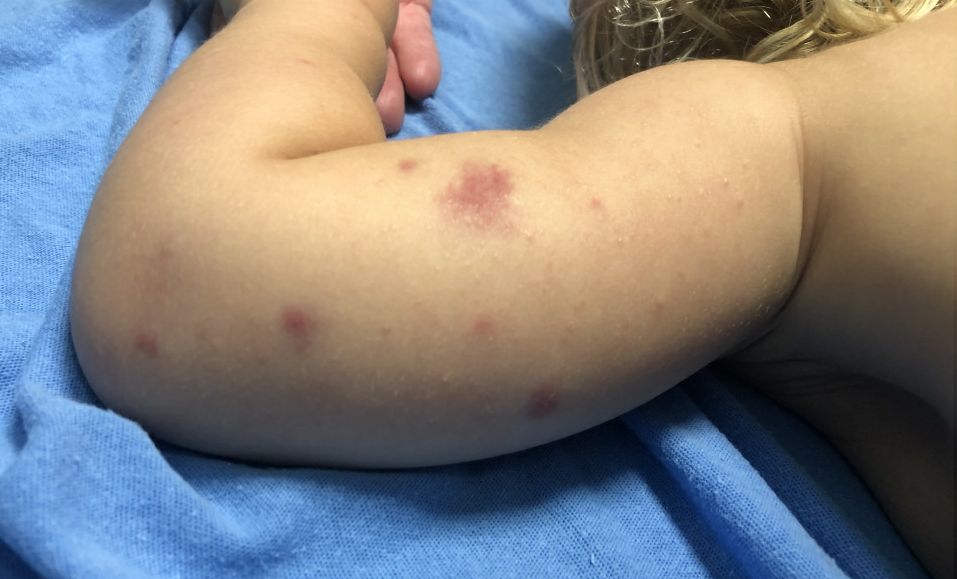
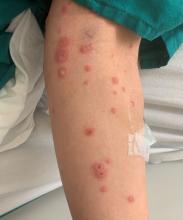
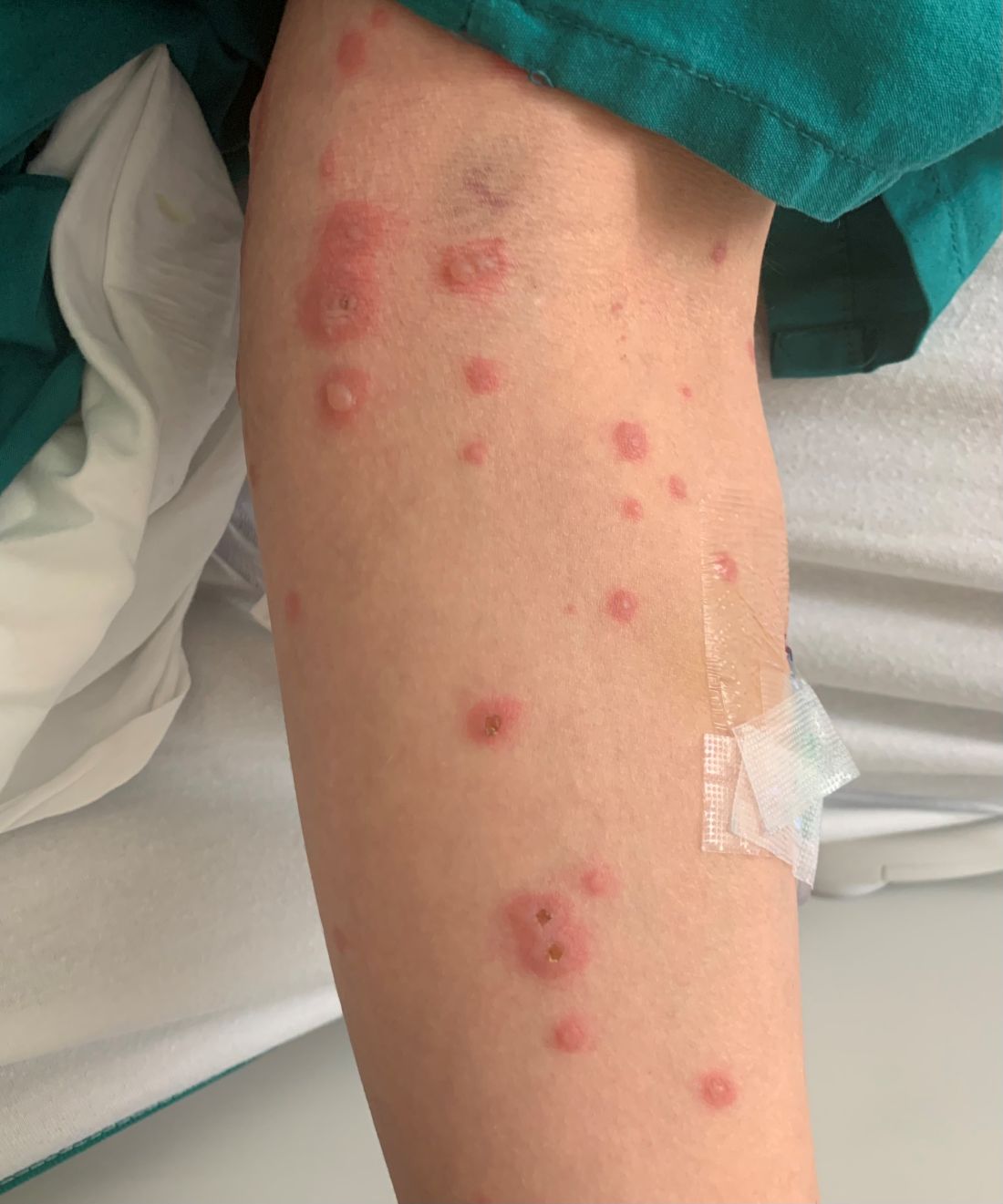
A 19-month-old vaccinated female with 2 days of rash
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
A 34-year-old male presented with 10 days of a pruritic rash
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Review finds anti-staphylococcus treatments have little impact on eczema
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published in Clinical and Experimental Allergy.
Eczema remains a huge disease burden worldwide, and colonization with S. aureus in eczema patients is common, but no standard intervention exists to relieve symptoms, wrote Nandini Banerjee, MD, of Addenbrooke’s Hospital, Cambridge, England. “While antibiotic treatment of clinically obvious infections such as cellulitis is beneficial, it is not clear whether antibiotic treatment of eczema influences eczema severity,” Dr. Banerjee noted.
The 41 studies included 1,753 participants and 10 treatment categories. Most of the studies were conducted in secondary care centers in Western Europe, North America, and the Far East. Twelve studies included children, four included only adults, 19 included children and adults, and in six studies, the participant age range was unclear. Among the studies with reported ages, the mean age ranged from 1.1 to 34.6 years. Eczema severity ranged from mild to severe, and treatment durations ranged from 10 minutes to 3 months.
The review presented comparisons of topical steroid/antibiotic combinations, oral antibiotics, and bleach baths. In 14 studies that compared topical steroid/antibiotic combinations to topical steroids alone, patients showed slightly greater global improvement in symptoms with the combination, but the impact on quality of life was not significantly different. Severe adverse events, including flare of dermatitis, worsening of eczema, and folliculitis, were reported by the patients who received the combination and the topical steroid–only patients. One study reported similar rates of antibiotic resistance in children treated with steroid only and with an antibiotic/steroid combination at 3 months’ follow-up.
In four studies, oral antibiotics “may make no difference in terms of good or excellent global improvement in infants and children at 14 to 28 days follow-up compared to placebo,” according to the review. The reviewers said that there was likely little or no difference in quality of life for infants and children given oral antibiotics, although they noted the low quality of evidence on this topic.
Five studies evaluated the impact of bleach baths on eczema patients with and without S. aureus infections. These studies showed no difference in global improvement measures compared with placebo and little or no difference in quality of life. Also, patients who underwent bleach baths compared with placebo patients reported similar adverse events of burning/stinging or dry skin at 2 months’ follow-up.
“Low-quality evidence, due to risk of bias, imprecise effect estimates, and heterogeneity, made pooling of results difficult,” Dr. Banerjee wrote. “Topical steroid/antibiotic combinations may be associated with possible small improvements in good or excellent signs/symptoms compared with topical steroid alone. High-quality trials evaluating efficacy, QOL, and antibiotic resistance are required,” she concluded.
In a commentary section after the review, Dr. Banerjee and colleagues noted that the United Kingdom’s NICE guidelines for managing atopic eczema in children younger than 12 years of age, published in March 2021, include evidence from the current updated Cochrane Review. The NICE guidelines emphasize that “in people who are not systemically unwell, clinicians should not routinely offer either a topical or oral antibiotic for secondary bacterial infection of eczema,” the Cochrane authors said. They added in their commentary that the use of antibiotics in cases of nonsevere infections can worsen eczema. Also, “the risk of antimicrobial resistance is high with topical antibiotics, and therefore extended doses of the same antibiotics should be avoided to prevent resistance,” they said. However, the authors acknowledged a role for antibiotics in certain situations. “In patients with systemic signs of infection such as cellulitis, systemic antibiotics have an important role in helping clear infection,” they noted.
Reasons for varying disease severity elude research
The current study is important because of the abundance of preclinical and clinical data that implicate S. aureus in atopic dermatitis pathogenesis, Brian Kim, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview.
Dr. Kim said that he was surprised by some of the study findings but not others. “On the one hand, I thought there would be data supporting antimicrobial therapy, albeit not strong support,” he said. “However, AD is a very complex disease, and understanding what a disease modifier does to it is hard to capture across studies of various different designs,” he said.
“The data supporting antimicrobial therapy for S. aureus in AD is not as clear as our clinical impressions may indicate,” said Dr. Kim. “We need to understand the relationship better, perhaps in particular subsets of patients,” he emphasized. In addition, “We need a better understanding of why some people are colonized with S. aureus, yet with little effect on AD itself, while others experience severe exacerbation of disease,” said Dr. Kim. Therefore, a key research question for future studies is whether the exacerbation is caused by the particular strain of the bug, the host susceptibility, or both, he said.
The review received no outside funding. Dr. Banerjee and Dr. Kim have disclosed that they had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CLINICAL AND EXPERIMENTAL ALLERGY
Male alopecia agents ranked by efficacy in meta-analysis
While up to 90% of men experience AGA in their lifetime, only three therapies are currently approved for treatment of the condition by the Food and Drug Administration – topical minoxidil, oral finasteride 1 mg, and low-level light therapy.
However, with common use of off-label oral minoxidil, as well as oral dutasteride and higher doses of oral finasteride, the latter two being 5-alpha reductase inhibitors, Aditya K. Gupta, MD, PhD, of Mediprobe Research, in London, Ont., and colleagues sought to compare the data on the three agents. Their results were published in JAMA Dermatology.
They note that, while there have been recent comparisons between oral and topical minoxidil, “to our knowledge no study has determined the comparative effectiveness of these 2 [formulations] with that of local and systemic dutasteride and finasteride.”
For the meta-analysis, the authors identified 23 studies meeting their criteria, involving patients with mean ages ranging from 22.8 to 41.8 years.
For the primary endpoint of the greatest increases in total hair count at 24 weeks, the analysis showed the 0.5-mg/day dose of dutasteride topped the list, with significantly greater efficacy, compared with 1 mg/day of finasteride (mean difference, 7.1 hairs per cm2).
The 0.5-mg/d dutasteride dose also showed higher efficacy than oral minoxidil at 0.25 mg/day (mean difference, 23.7 hairs per cm2) and 5 mg/day (mean difference, 15.0 hairs per cm2) and topical minoxidil at 2% (mean difference, 8.5 hairs per cm2).
For the secondary endpoint of the greatest increase in terminal hair count at 24 weeks, the 5-mg/day dose of minoxidil had significantly greater efficacy compared with the 0.25-mg/day dose of the drug, as well as with minoxidil’s 2% and 5% topical formulations.
The minoxidil 5-mg/day dose was also significantly more effective than 1 mg/day of finasteride for terminal hair count at 24 weeks.
In longer-term outcomes at 48 weeks, the greatest increase in total hair count at 48 weeks was observed with 5 mg/day of finasteride, which was significantly more effective, compared with 2% topical minoxidil.
And the greatest increase in terminal hair count at 48 weeks was observed with 1 mg/day of oral finasteride, which was significantly more effective than 2% as well as 5% topical minoxidil.
Based on the results, the authors ranked the agents in a decreasing order of efficacy: 0.5 mg/day of oral dutasteride, 5 mg/day of oral finasteride, 5 mg/day of oral minoxidil, 1 mg/day of oral finasteride, 5% topical minoxidil, 2% topical minoxidil, and 0.25 mg/day of oral minoxidil.
Commenting on the analysis in an accompanying editorial, Kathie P. Huang, MD, of the department of dermatology, Brigham and Women’s Hospital, Boston, and Maryanne M. Senna, MD, of the department of dermatology, Massachusetts General Hospital, Boston, said the results, in general, are consistent with their experiences, noting that 2% minoxidil is typically not used in men.
They noted that, “although topical minoxidil ranked higher than the very-low-dose 0.25 mg oral minoxidil, our personal experience is that oral minoxidil at doses of 1.25 mg to 5 mg are far superior to topical minoxidil for treating AGA.”
Adverse event considerations important
Importantly, however, strong consideration needs to be given to adverse-event profiles, as well as patient comorbidities in selecting agents, the editorial authors asserted.
With 1 mg finasteride, for instance, potential adverse events include decreased libido, erectile dysfunction, decreased ejaculatory volume, reduction in sperm count, testicular pain, depression, and gynecomastia, they noted.
And while finasteride appears to be associated with a decreased risk of prostate cancer, those receiving the drug who do develop prostate cancer may be diagnosed with higher-grade prostate cancer; however, that “might be related to tissue sampling artifact,” the editorial authors said.
Less has been published on dutasteride’s adverse-event profile, and that, in itself, is a concern.
Overall, “as more direct-to-consumer companies treating male AGA emerge, it is especially important that the potential risks of these medications be made clear to patients,” they added.
Further commenting on the analysis to this news organization, Antonella Tosti, MD, the Fredric Brandt Endowed Professor of Dermatology and Cutaneous Surgery at the University of Miami, said the study offers some important insights – and caveats.
“I think this is a very interesting study, but you have to consider what works for your patients,” she said.
Dr. Tosti noted that the 5-mg dose of minoxidil is a concern in terms of side effects. “That dose is pretty high and could feasibly cause some hypertrichosis, which can be a concern to men as well as women.”
She agrees that the lack of data on side effects with dutasteride is also a concern, especially in light of some of the known side effects with other agents.
“That’s why I don’t use it very much in younger patients – because I’m afraid it could potentially affect their fertility,” Dr. Tosti said.
In general, Dr. Tosti said she finds a combination of agents provides the best results, as many clinicians use.
“I find dutasteride (0.5 mg/day) plus oral minoxidil (1-2.5 mg/day) plus topical 5% minoxidil is the best combination,” she said.
The authors and Dr. Tosti disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While up to 90% of men experience AGA in their lifetime, only three therapies are currently approved for treatment of the condition by the Food and Drug Administration – topical minoxidil, oral finasteride 1 mg, and low-level light therapy.
However, with common use of off-label oral minoxidil, as well as oral dutasteride and higher doses of oral finasteride, the latter two being 5-alpha reductase inhibitors, Aditya K. Gupta, MD, PhD, of Mediprobe Research, in London, Ont., and colleagues sought to compare the data on the three agents. Their results were published in JAMA Dermatology.
They note that, while there have been recent comparisons between oral and topical minoxidil, “to our knowledge no study has determined the comparative effectiveness of these 2 [formulations] with that of local and systemic dutasteride and finasteride.”
For the meta-analysis, the authors identified 23 studies meeting their criteria, involving patients with mean ages ranging from 22.8 to 41.8 years.
For the primary endpoint of the greatest increases in total hair count at 24 weeks, the analysis showed the 0.5-mg/day dose of dutasteride topped the list, with significantly greater efficacy, compared with 1 mg/day of finasteride (mean difference, 7.1 hairs per cm2).
The 0.5-mg/d dutasteride dose also showed higher efficacy than oral minoxidil at 0.25 mg/day (mean difference, 23.7 hairs per cm2) and 5 mg/day (mean difference, 15.0 hairs per cm2) and topical minoxidil at 2% (mean difference, 8.5 hairs per cm2).
For the secondary endpoint of the greatest increase in terminal hair count at 24 weeks, the 5-mg/day dose of minoxidil had significantly greater efficacy compared with the 0.25-mg/day dose of the drug, as well as with minoxidil’s 2% and 5% topical formulations.
The minoxidil 5-mg/day dose was also significantly more effective than 1 mg/day of finasteride for terminal hair count at 24 weeks.
In longer-term outcomes at 48 weeks, the greatest increase in total hair count at 48 weeks was observed with 5 mg/day of finasteride, which was significantly more effective, compared with 2% topical minoxidil.
And the greatest increase in terminal hair count at 48 weeks was observed with 1 mg/day of oral finasteride, which was significantly more effective than 2% as well as 5% topical minoxidil.
Based on the results, the authors ranked the agents in a decreasing order of efficacy: 0.5 mg/day of oral dutasteride, 5 mg/day of oral finasteride, 5 mg/day of oral minoxidil, 1 mg/day of oral finasteride, 5% topical minoxidil, 2% topical minoxidil, and 0.25 mg/day of oral minoxidil.
Commenting on the analysis in an accompanying editorial, Kathie P. Huang, MD, of the department of dermatology, Brigham and Women’s Hospital, Boston, and Maryanne M. Senna, MD, of the department of dermatology, Massachusetts General Hospital, Boston, said the results, in general, are consistent with their experiences, noting that 2% minoxidil is typically not used in men.
They noted that, “although topical minoxidil ranked higher than the very-low-dose 0.25 mg oral minoxidil, our personal experience is that oral minoxidil at doses of 1.25 mg to 5 mg are far superior to topical minoxidil for treating AGA.”
Adverse event considerations important
Importantly, however, strong consideration needs to be given to adverse-event profiles, as well as patient comorbidities in selecting agents, the editorial authors asserted.
With 1 mg finasteride, for instance, potential adverse events include decreased libido, erectile dysfunction, decreased ejaculatory volume, reduction in sperm count, testicular pain, depression, and gynecomastia, they noted.
And while finasteride appears to be associated with a decreased risk of prostate cancer, those receiving the drug who do develop prostate cancer may be diagnosed with higher-grade prostate cancer; however, that “might be related to tissue sampling artifact,” the editorial authors said.
Less has been published on dutasteride’s adverse-event profile, and that, in itself, is a concern.
Overall, “as more direct-to-consumer companies treating male AGA emerge, it is especially important that the potential risks of these medications be made clear to patients,” they added.
Further commenting on the analysis to this news organization, Antonella Tosti, MD, the Fredric Brandt Endowed Professor of Dermatology and Cutaneous Surgery at the University of Miami, said the study offers some important insights – and caveats.
“I think this is a very interesting study, but you have to consider what works for your patients,” she said.
Dr. Tosti noted that the 5-mg dose of minoxidil is a concern in terms of side effects. “That dose is pretty high and could feasibly cause some hypertrichosis, which can be a concern to men as well as women.”
She agrees that the lack of data on side effects with dutasteride is also a concern, especially in light of some of the known side effects with other agents.
“That’s why I don’t use it very much in younger patients – because I’m afraid it could potentially affect their fertility,” Dr. Tosti said.
In general, Dr. Tosti said she finds a combination of agents provides the best results, as many clinicians use.
“I find dutasteride (0.5 mg/day) plus oral minoxidil (1-2.5 mg/day) plus topical 5% minoxidil is the best combination,” she said.
The authors and Dr. Tosti disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While up to 90% of men experience AGA in their lifetime, only three therapies are currently approved for treatment of the condition by the Food and Drug Administration – topical minoxidil, oral finasteride 1 mg, and low-level light therapy.
However, with common use of off-label oral minoxidil, as well as oral dutasteride and higher doses of oral finasteride, the latter two being 5-alpha reductase inhibitors, Aditya K. Gupta, MD, PhD, of Mediprobe Research, in London, Ont., and colleagues sought to compare the data on the three agents. Their results were published in JAMA Dermatology.
They note that, while there have been recent comparisons between oral and topical minoxidil, “to our knowledge no study has determined the comparative effectiveness of these 2 [formulations] with that of local and systemic dutasteride and finasteride.”
For the meta-analysis, the authors identified 23 studies meeting their criteria, involving patients with mean ages ranging from 22.8 to 41.8 years.
For the primary endpoint of the greatest increases in total hair count at 24 weeks, the analysis showed the 0.5-mg/day dose of dutasteride topped the list, with significantly greater efficacy, compared with 1 mg/day of finasteride (mean difference, 7.1 hairs per cm2).
The 0.5-mg/d dutasteride dose also showed higher efficacy than oral minoxidil at 0.25 mg/day (mean difference, 23.7 hairs per cm2) and 5 mg/day (mean difference, 15.0 hairs per cm2) and topical minoxidil at 2% (mean difference, 8.5 hairs per cm2).
For the secondary endpoint of the greatest increase in terminal hair count at 24 weeks, the 5-mg/day dose of minoxidil had significantly greater efficacy compared with the 0.25-mg/day dose of the drug, as well as with minoxidil’s 2% and 5% topical formulations.
The minoxidil 5-mg/day dose was also significantly more effective than 1 mg/day of finasteride for terminal hair count at 24 weeks.
In longer-term outcomes at 48 weeks, the greatest increase in total hair count at 48 weeks was observed with 5 mg/day of finasteride, which was significantly more effective, compared with 2% topical minoxidil.
And the greatest increase in terminal hair count at 48 weeks was observed with 1 mg/day of oral finasteride, which was significantly more effective than 2% as well as 5% topical minoxidil.
Based on the results, the authors ranked the agents in a decreasing order of efficacy: 0.5 mg/day of oral dutasteride, 5 mg/day of oral finasteride, 5 mg/day of oral minoxidil, 1 mg/day of oral finasteride, 5% topical minoxidil, 2% topical minoxidil, and 0.25 mg/day of oral minoxidil.
Commenting on the analysis in an accompanying editorial, Kathie P. Huang, MD, of the department of dermatology, Brigham and Women’s Hospital, Boston, and Maryanne M. Senna, MD, of the department of dermatology, Massachusetts General Hospital, Boston, said the results, in general, are consistent with their experiences, noting that 2% minoxidil is typically not used in men.
They noted that, “although topical minoxidil ranked higher than the very-low-dose 0.25 mg oral minoxidil, our personal experience is that oral minoxidil at doses of 1.25 mg to 5 mg are far superior to topical minoxidil for treating AGA.”
Adverse event considerations important
Importantly, however, strong consideration needs to be given to adverse-event profiles, as well as patient comorbidities in selecting agents, the editorial authors asserted.
With 1 mg finasteride, for instance, potential adverse events include decreased libido, erectile dysfunction, decreased ejaculatory volume, reduction in sperm count, testicular pain, depression, and gynecomastia, they noted.
And while finasteride appears to be associated with a decreased risk of prostate cancer, those receiving the drug who do develop prostate cancer may be diagnosed with higher-grade prostate cancer; however, that “might be related to tissue sampling artifact,” the editorial authors said.
Less has been published on dutasteride’s adverse-event profile, and that, in itself, is a concern.
Overall, “as more direct-to-consumer companies treating male AGA emerge, it is especially important that the potential risks of these medications be made clear to patients,” they added.
Further commenting on the analysis to this news organization, Antonella Tosti, MD, the Fredric Brandt Endowed Professor of Dermatology and Cutaneous Surgery at the University of Miami, said the study offers some important insights – and caveats.
“I think this is a very interesting study, but you have to consider what works for your patients,” she said.
Dr. Tosti noted that the 5-mg dose of minoxidil is a concern in terms of side effects. “That dose is pretty high and could feasibly cause some hypertrichosis, which can be a concern to men as well as women.”
She agrees that the lack of data on side effects with dutasteride is also a concern, especially in light of some of the known side effects with other agents.
“That’s why I don’t use it very much in younger patients – because I’m afraid it could potentially affect their fertility,” Dr. Tosti said.
In general, Dr. Tosti said she finds a combination of agents provides the best results, as many clinicians use.
“I find dutasteride (0.5 mg/day) plus oral minoxidil (1-2.5 mg/day) plus topical 5% minoxidil is the best combination,” she said.
The authors and Dr. Tosti disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Dupilumab under FDA review for atopic dermatitis in children aged 6 months to 5 years
The and Sanofi.
If approved, dupilumab would be the first biologic approved for children in this age group in the United States, according to the statement. The proposed indication is as add-on therapy for children with moderate to severe AD not adequately controlled with topical prescription therapies or for whom topical therapies are not advised. The FDA granted breakthrough therapy designation for dupilumab for the treatment of severe AD in children aged 6 months to 11 years in 2016.
Approximately 85%-95% of atopic dermatitis patients develop symptoms before 5 years of age, and these symptoms often continue into adulthood, with an increased risk of skin infections and a significant impact on quality of life, according to the statement.
The sBLA is based on data from a phase 3 pivotal study of 162 children aged 6 months to 5 years in which dupilumab was added to standard-of-care topical corticosteroids, presented in December 2021. In the study, dupilumab plus standard of care significantly improved skin clearance and reduced overall disease severity and itch at 16 weeks compared with standard of care alone. Overall, 28% of the children randomized to dupilumab achieved the primary endpoint of clear or almost-clear skin, compared with 4% with those on standard of care alone (P < .0001), according to the manufacturers. Patients in the dupilumab group received either 200 mg (for children weighing ≥ 5 to < 15 kg) or 300 mg (for children weighing ≥ 15 to < 30 kg) every 4 weeks. Safety results were similar to those seen with dupilumab for children aged 6 years and older.
Conjunctivitis and herpes infections were among the most common adverse events associated with dupilumab in the study, according to the statement.
The target action date for the FDA decision on this application is June 9, 2022.
The and Sanofi.
If approved, dupilumab would be the first biologic approved for children in this age group in the United States, according to the statement. The proposed indication is as add-on therapy for children with moderate to severe AD not adequately controlled with topical prescription therapies or for whom topical therapies are not advised. The FDA granted breakthrough therapy designation for dupilumab for the treatment of severe AD in children aged 6 months to 11 years in 2016.
Approximately 85%-95% of atopic dermatitis patients develop symptoms before 5 years of age, and these symptoms often continue into adulthood, with an increased risk of skin infections and a significant impact on quality of life, according to the statement.
The sBLA is based on data from a phase 3 pivotal study of 162 children aged 6 months to 5 years in which dupilumab was added to standard-of-care topical corticosteroids, presented in December 2021. In the study, dupilumab plus standard of care significantly improved skin clearance and reduced overall disease severity and itch at 16 weeks compared with standard of care alone. Overall, 28% of the children randomized to dupilumab achieved the primary endpoint of clear or almost-clear skin, compared with 4% with those on standard of care alone (P < .0001), according to the manufacturers. Patients in the dupilumab group received either 200 mg (for children weighing ≥ 5 to < 15 kg) or 300 mg (for children weighing ≥ 15 to < 30 kg) every 4 weeks. Safety results were similar to those seen with dupilumab for children aged 6 years and older.
Conjunctivitis and herpes infections were among the most common adverse events associated with dupilumab in the study, according to the statement.
The target action date for the FDA decision on this application is June 9, 2022.
The and Sanofi.
If approved, dupilumab would be the first biologic approved for children in this age group in the United States, according to the statement. The proposed indication is as add-on therapy for children with moderate to severe AD not adequately controlled with topical prescription therapies or for whom topical therapies are not advised. The FDA granted breakthrough therapy designation for dupilumab for the treatment of severe AD in children aged 6 months to 11 years in 2016.
Approximately 85%-95% of atopic dermatitis patients develop symptoms before 5 years of age, and these symptoms often continue into adulthood, with an increased risk of skin infections and a significant impact on quality of life, according to the statement.
The sBLA is based on data from a phase 3 pivotal study of 162 children aged 6 months to 5 years in which dupilumab was added to standard-of-care topical corticosteroids, presented in December 2021. In the study, dupilumab plus standard of care significantly improved skin clearance and reduced overall disease severity and itch at 16 weeks compared with standard of care alone. Overall, 28% of the children randomized to dupilumab achieved the primary endpoint of clear or almost-clear skin, compared with 4% with those on standard of care alone (P < .0001), according to the manufacturers. Patients in the dupilumab group received either 200 mg (for children weighing ≥ 5 to < 15 kg) or 300 mg (for children weighing ≥ 15 to < 30 kg) every 4 weeks. Safety results were similar to those seen with dupilumab for children aged 6 years and older.
Conjunctivitis and herpes infections were among the most common adverse events associated with dupilumab in the study, according to the statement.
The target action date for the FDA decision on this application is June 9, 2022.
FROM THE FDA
Chronic truncal rash
The patient was given a diagnosis of tinea versicolor (TV), also known as pityriasis versicolor, after a potassium hydroxide (KOH) prep test on a skin scraping confirmed the spaghetti and meatballs pattern of Malassezia Furfur (see image above). Note that in most cases, a KOH prep is not required for the diagnosis. Experienced clinicians will usually make the diagnosis based on the appearance of hyper- or hypopigmented macules or patches with fine scale on the trunk of adults. KOH prep is useful if the diagnosis is uncertain.
TV is a common fungal infection that’s seen more frequently in tropical climates and occurs equally in men and women.1 M. Furfur thrives on the lipids in the skin of sebum-rich areas, which explains its truncal distribution and rare occurrence in children (who have much lower sebum production).
Usually, topical antifungal medications are considered first-line treatment, but since large areas of skin are involved, adequate amounts need to be used. One of the most common and inexpensive treatments is to apply selenium sulfide shampoo (Selsun Blue) undiluted to the entire trunk, then allow to dry and remain in place overnight before showering. A repeat application should be done 1 week later. Topical terbinafine cream applied bid for 2 weeks is another option, as is oral itraconazole in a single 400 mg dose.
This patient declined the selenium sulfide topical treatment and requested systemic therapy, so he was prescribed itraconazole 400 mg orally as a single dose. He was advised that it might take a few weeks to clear up, and to use the selenium sulfide application if the itraconazole was not effective. Follow-up was not planned due to the high success rate of these therapies.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Saunte DML, Gaitanis G, Hay RJ. Malassezia-associated skin diseases, the use of diagnostics and treatment. Front Cell Infect Microbiol. 2020;10:112. doi: 10.3389/fcimb.2020.00112

The patient was given a diagnosis of tinea versicolor (TV), also known as pityriasis versicolor, after a potassium hydroxide (KOH) prep test on a skin scraping confirmed the spaghetti and meatballs pattern of Malassezia Furfur (see image above). Note that in most cases, a KOH prep is not required for the diagnosis. Experienced clinicians will usually make the diagnosis based on the appearance of hyper- or hypopigmented macules or patches with fine scale on the trunk of adults. KOH prep is useful if the diagnosis is uncertain.
TV is a common fungal infection that’s seen more frequently in tropical climates and occurs equally in men and women.1 M. Furfur thrives on the lipids in the skin of sebum-rich areas, which explains its truncal distribution and rare occurrence in children (who have much lower sebum production).
Usually, topical antifungal medications are considered first-line treatment, but since large areas of skin are involved, adequate amounts need to be used. One of the most common and inexpensive treatments is to apply selenium sulfide shampoo (Selsun Blue) undiluted to the entire trunk, then allow to dry and remain in place overnight before showering. A repeat application should be done 1 week later. Topical terbinafine cream applied bid for 2 weeks is another option, as is oral itraconazole in a single 400 mg dose.
This patient declined the selenium sulfide topical treatment and requested systemic therapy, so he was prescribed itraconazole 400 mg orally as a single dose. He was advised that it might take a few weeks to clear up, and to use the selenium sulfide application if the itraconazole was not effective. Follow-up was not planned due to the high success rate of these therapies.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The patient was given a diagnosis of tinea versicolor (TV), also known as pityriasis versicolor, after a potassium hydroxide (KOH) prep test on a skin scraping confirmed the spaghetti and meatballs pattern of Malassezia Furfur (see image above). Note that in most cases, a KOH prep is not required for the diagnosis. Experienced clinicians will usually make the diagnosis based on the appearance of hyper- or hypopigmented macules or patches with fine scale on the trunk of adults. KOH prep is useful if the diagnosis is uncertain.
TV is a common fungal infection that’s seen more frequently in tropical climates and occurs equally in men and women.1 M. Furfur thrives on the lipids in the skin of sebum-rich areas, which explains its truncal distribution and rare occurrence in children (who have much lower sebum production).
Usually, topical antifungal medications are considered first-line treatment, but since large areas of skin are involved, adequate amounts need to be used. One of the most common and inexpensive treatments is to apply selenium sulfide shampoo (Selsun Blue) undiluted to the entire trunk, then allow to dry and remain in place overnight before showering. A repeat application should be done 1 week later. Topical terbinafine cream applied bid for 2 weeks is another option, as is oral itraconazole in a single 400 mg dose.
This patient declined the selenium sulfide topical treatment and requested systemic therapy, so he was prescribed itraconazole 400 mg orally as a single dose. He was advised that it might take a few weeks to clear up, and to use the selenium sulfide application if the itraconazole was not effective. Follow-up was not planned due to the high success rate of these therapies.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Saunte DML, Gaitanis G, Hay RJ. Malassezia-associated skin diseases, the use of diagnostics and treatment. Front Cell Infect Microbiol. 2020;10:112. doi: 10.3389/fcimb.2020.00112
1. Saunte DML, Gaitanis G, Hay RJ. Malassezia-associated skin diseases, the use of diagnostics and treatment. Front Cell Infect Microbiol. 2020;10:112. doi: 10.3389/fcimb.2020.00112