User login
Lifestyle changes can lead to remission, but not a cure, in type 2 diabetes
Whenever I get a new patient with type 2 diabetes, who is generally on metformin, one of the very first questions they ask me is, “Can I get off my medication?” Everybody, it seems, who gets diabetes wants to not have diabetes.
So, what does this really mean? What does this mean to me as a clinician? And what does this mean to my patients? The American Diabetes Association recently came out with a consensus statement that defines and interprets the definition of remission in people with type 2 diabetes. Basically, if the hemoglobin A1c is less than 6.5% without diabetes medications for at least 3 months, that’s considered remission.
There are other considerations, such as metabolic surgery, that can lead to remission. But again, such patients should be 3 months post surgery and at least 3 months off diabetes medication. As for a lifestyle intervention, the authors state that remission really happens within about 6 months.
That leads me to wonder: What is remission? Remission really means temporary recovery, so it doesn’t mean a cure. Now, I’m not against curing diabetes. In fact, I’m all for it. But when somebody gets diabetes – and honestly, it doesn’t matter whether it’s type 1 or type 2 – the first thing I think of, and I think the first thing that my patients are taught, is how important it is to have a healthy lifestyle. This healthy lifestyle isn’t just for people with diabetes; it largely means the healthy lifestyle that all of us should follow, one where we eat fewer simple carbs, less processed food, more vegetables, more lean proteins and meats – all of the things that we know we should do. And all of the things that keep us healthy. To some degree, I don’t think you can ever get “remission” from diabetes, because if having diabetes points an individual toward having a healthier lifestyle, I think that’s great.
I think people should exercise more. When it comes to treating diabetes, exercise is key. When you think about obesity, we want to help people who are overweight or obese lose weight as part of their treatment for diabetes. And that doesn’t go away, either.
So, no, people who are diagnosed with diabetes don’t really go into remission if they keep their same old habits and don’t lose weight and don’t exercise. But many people with diabetes can get off medication if they do those things.
However, it’s not true for everybody, and I don’t want people to get unrealistic expectations because I think there are probably about a thousand different subtypes of type 2 diabetes. And I’ve definitely seen people who are lean with type 2 diabetes who don’t respond as well to a lifestyle intervention, or people who are more insulin deficient, who also need medication.
I think it’s really important to frame the expectation that, if remission means going back to the way it was before, when they didn’t have to think about what they ate or whether or not they exercised, that’s not going to happen. I think diabetes should really be a wake-up call that people need to be healthier in terms of their lifestyle habits.
The issue of medication is really an individual one, and I think we need to help patients look for what’s best for the individual, what their targets are, what their goals are. But we also have to think that diabetes isn’t just about glucose.
So remission in terms of the ADA’s definition looks at glucose, but I look at more than glucose. You have to look at lipids and blood pressure. And, as I mentioned earlier, you have to look at whether or not a person has preexisting cardiovascular disease or has the presence of microvascular complications that need to be screened for and treated.
I actually think that, in some ways, the diagnosis of diabetes is helpful simply because it helps put people on a better path to health. I don’t want people to think that remission means that they can go back to unhealthy habits. I really encourage all people to live a healthier lifestyle, and if it leads to improvements in glucose levels and getting off medication, I think that’s wonderful and a worthy goal. But remember, health and meeting one’s targets remain key in the treatment of type 2 diabetes.
Dr. Peters is a professor in the department of clinical medicine at the University of Southern California, Los Angeles. She reported serving on the advisory board or speakers’ bureau of Medscape and several pharmaceutical companies, and has received research support from Dexcom, MannKind, and AstraZeneca. This perspective and an accompanying video first appeared on Medscape.com.
Whenever I get a new patient with type 2 diabetes, who is generally on metformin, one of the very first questions they ask me is, “Can I get off my medication?” Everybody, it seems, who gets diabetes wants to not have diabetes.
So, what does this really mean? What does this mean to me as a clinician? And what does this mean to my patients? The American Diabetes Association recently came out with a consensus statement that defines and interprets the definition of remission in people with type 2 diabetes. Basically, if the hemoglobin A1c is less than 6.5% without diabetes medications for at least 3 months, that’s considered remission.
There are other considerations, such as metabolic surgery, that can lead to remission. But again, such patients should be 3 months post surgery and at least 3 months off diabetes medication. As for a lifestyle intervention, the authors state that remission really happens within about 6 months.
That leads me to wonder: What is remission? Remission really means temporary recovery, so it doesn’t mean a cure. Now, I’m not against curing diabetes. In fact, I’m all for it. But when somebody gets diabetes – and honestly, it doesn’t matter whether it’s type 1 or type 2 – the first thing I think of, and I think the first thing that my patients are taught, is how important it is to have a healthy lifestyle. This healthy lifestyle isn’t just for people with diabetes; it largely means the healthy lifestyle that all of us should follow, one where we eat fewer simple carbs, less processed food, more vegetables, more lean proteins and meats – all of the things that we know we should do. And all of the things that keep us healthy. To some degree, I don’t think you can ever get “remission” from diabetes, because if having diabetes points an individual toward having a healthier lifestyle, I think that’s great.
I think people should exercise more. When it comes to treating diabetes, exercise is key. When you think about obesity, we want to help people who are overweight or obese lose weight as part of their treatment for diabetes. And that doesn’t go away, either.
So, no, people who are diagnosed with diabetes don’t really go into remission if they keep their same old habits and don’t lose weight and don’t exercise. But many people with diabetes can get off medication if they do those things.
However, it’s not true for everybody, and I don’t want people to get unrealistic expectations because I think there are probably about a thousand different subtypes of type 2 diabetes. And I’ve definitely seen people who are lean with type 2 diabetes who don’t respond as well to a lifestyle intervention, or people who are more insulin deficient, who also need medication.
I think it’s really important to frame the expectation that, if remission means going back to the way it was before, when they didn’t have to think about what they ate or whether or not they exercised, that’s not going to happen. I think diabetes should really be a wake-up call that people need to be healthier in terms of their lifestyle habits.
The issue of medication is really an individual one, and I think we need to help patients look for what’s best for the individual, what their targets are, what their goals are. But we also have to think that diabetes isn’t just about glucose.
So remission in terms of the ADA’s definition looks at glucose, but I look at more than glucose. You have to look at lipids and blood pressure. And, as I mentioned earlier, you have to look at whether or not a person has preexisting cardiovascular disease or has the presence of microvascular complications that need to be screened for and treated.
I actually think that, in some ways, the diagnosis of diabetes is helpful simply because it helps put people on a better path to health. I don’t want people to think that remission means that they can go back to unhealthy habits. I really encourage all people to live a healthier lifestyle, and if it leads to improvements in glucose levels and getting off medication, I think that’s wonderful and a worthy goal. But remember, health and meeting one’s targets remain key in the treatment of type 2 diabetes.
Dr. Peters is a professor in the department of clinical medicine at the University of Southern California, Los Angeles. She reported serving on the advisory board or speakers’ bureau of Medscape and several pharmaceutical companies, and has received research support from Dexcom, MannKind, and AstraZeneca. This perspective and an accompanying video first appeared on Medscape.com.
Whenever I get a new patient with type 2 diabetes, who is generally on metformin, one of the very first questions they ask me is, “Can I get off my medication?” Everybody, it seems, who gets diabetes wants to not have diabetes.
So, what does this really mean? What does this mean to me as a clinician? And what does this mean to my patients? The American Diabetes Association recently came out with a consensus statement that defines and interprets the definition of remission in people with type 2 diabetes. Basically, if the hemoglobin A1c is less than 6.5% without diabetes medications for at least 3 months, that’s considered remission.
There are other considerations, such as metabolic surgery, that can lead to remission. But again, such patients should be 3 months post surgery and at least 3 months off diabetes medication. As for a lifestyle intervention, the authors state that remission really happens within about 6 months.
That leads me to wonder: What is remission? Remission really means temporary recovery, so it doesn’t mean a cure. Now, I’m not against curing diabetes. In fact, I’m all for it. But when somebody gets diabetes – and honestly, it doesn’t matter whether it’s type 1 or type 2 – the first thing I think of, and I think the first thing that my patients are taught, is how important it is to have a healthy lifestyle. This healthy lifestyle isn’t just for people with diabetes; it largely means the healthy lifestyle that all of us should follow, one where we eat fewer simple carbs, less processed food, more vegetables, more lean proteins and meats – all of the things that we know we should do. And all of the things that keep us healthy. To some degree, I don’t think you can ever get “remission” from diabetes, because if having diabetes points an individual toward having a healthier lifestyle, I think that’s great.
I think people should exercise more. When it comes to treating diabetes, exercise is key. When you think about obesity, we want to help people who are overweight or obese lose weight as part of their treatment for diabetes. And that doesn’t go away, either.
So, no, people who are diagnosed with diabetes don’t really go into remission if they keep their same old habits and don’t lose weight and don’t exercise. But many people with diabetes can get off medication if they do those things.
However, it’s not true for everybody, and I don’t want people to get unrealistic expectations because I think there are probably about a thousand different subtypes of type 2 diabetes. And I’ve definitely seen people who are lean with type 2 diabetes who don’t respond as well to a lifestyle intervention, or people who are more insulin deficient, who also need medication.
I think it’s really important to frame the expectation that, if remission means going back to the way it was before, when they didn’t have to think about what they ate or whether or not they exercised, that’s not going to happen. I think diabetes should really be a wake-up call that people need to be healthier in terms of their lifestyle habits.
The issue of medication is really an individual one, and I think we need to help patients look for what’s best for the individual, what their targets are, what their goals are. But we also have to think that diabetes isn’t just about glucose.
So remission in terms of the ADA’s definition looks at glucose, but I look at more than glucose. You have to look at lipids and blood pressure. And, as I mentioned earlier, you have to look at whether or not a person has preexisting cardiovascular disease or has the presence of microvascular complications that need to be screened for and treated.
I actually think that, in some ways, the diagnosis of diabetes is helpful simply because it helps put people on a better path to health. I don’t want people to think that remission means that they can go back to unhealthy habits. I really encourage all people to live a healthier lifestyle, and if it leads to improvements in glucose levels and getting off medication, I think that’s wonderful and a worthy goal. But remember, health and meeting one’s targets remain key in the treatment of type 2 diabetes.
Dr. Peters is a professor in the department of clinical medicine at the University of Southern California, Los Angeles. She reported serving on the advisory board or speakers’ bureau of Medscape and several pharmaceutical companies, and has received research support from Dexcom, MannKind, and AstraZeneca. This perspective and an accompanying video first appeared on Medscape.com.
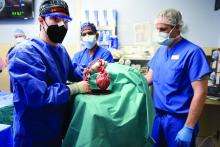
What does a pig-to-human heart transplant mean for medicine?
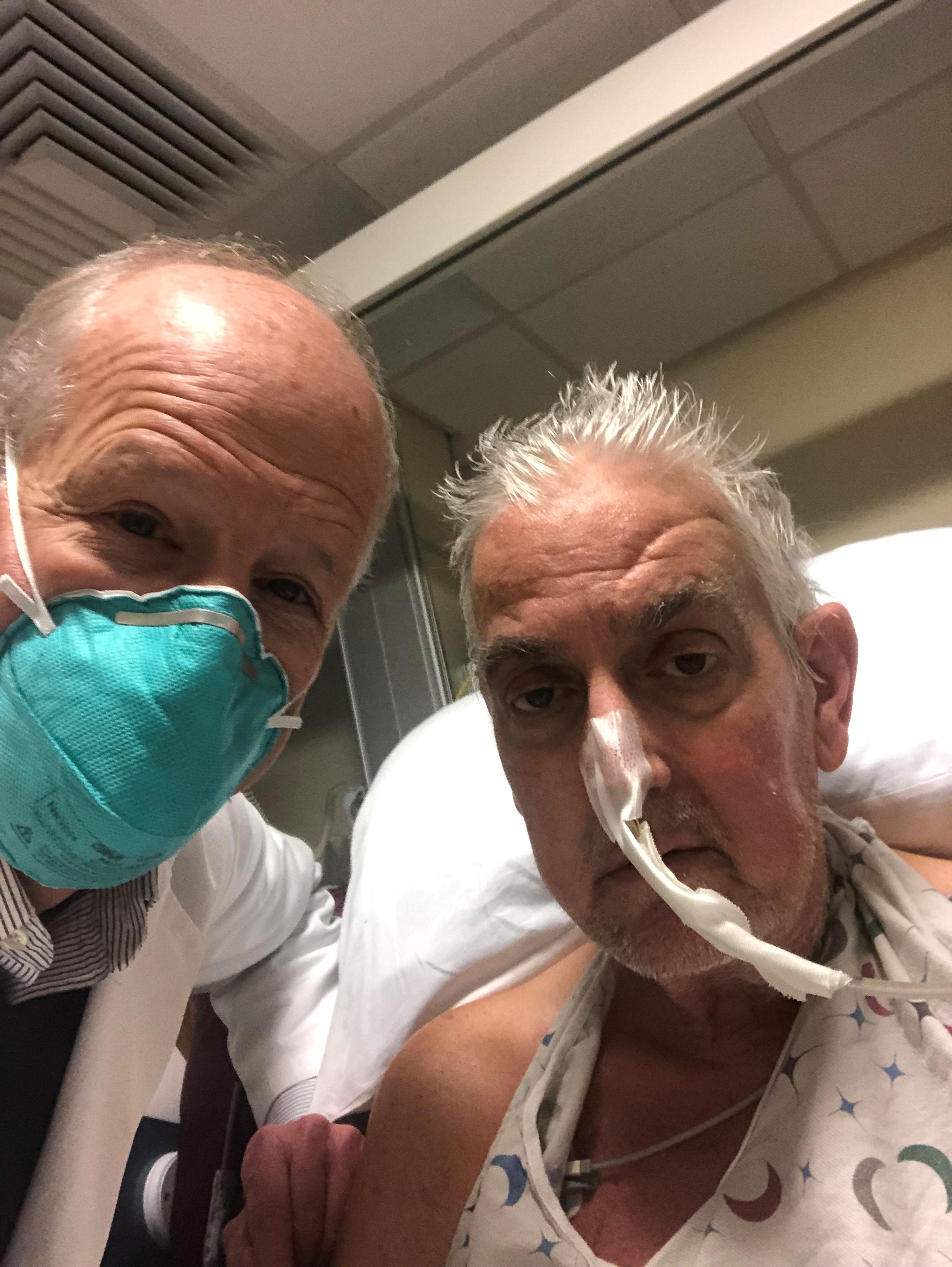
Scientific achievements usually raise big new questions, and the remarkable surgery that took place on Jan. 7, when Maryland resident David Bennett was transplanted with a genetically modified heart from a pig, has been no different.
The 57-year-old with end-stage heart failure had been repeatedly turned down for a standard transplant and was judged a poor candidate for a ventricular assist device. Now his new heart is beating soundly and apparently accepted by his immune system as Mr. Bennett, his physicians at the University of Maryland where the procedure took place, and indeed the world set out on a journey with far more unknowns than knowns.
“I think even just a couple of years ago, people felt that xenotransplantation for the heart and other organs was still a long way off. And it seems like it’s started to move very quickly,” Larry A. Allen, MD, University of Colorado, Aurora, said in an interview.
Demand for donor hearts far outstrips supply, and despite advances in the development of ventricular assist pumps and artificial hearts, “there are still significant limitations to them in terms of clotting, stroke, and infection. We’ve seen the use of those devices plateau,” Dr. Allen said. “So, the concept of a nonhuman source of organs is exciting and very much in need, if people can get it to work.”
“I really credit the surgeons at the University of Maryland for courageous clinical work and a brilliant scientific innovation,” Clyde W. Yancy, MD, MSc, Northwestern University, Chicago, said in an interview. “But it’s always in the implementation that we have to hold our breath.” Heart xenotransplantation is an old idea that “has never before been successful,” he said. And standard heart transplantation has set a high bar, with a 1-year survival of about 90% and low 1-year risk for rejection. Whether the new procedure can meet that standard is unknown, as is its potential for complications, such as chronic rejection or cancers due to long-term immunosuppression. Those are “major questions requiring more time and careful follow-up.”
‘Still a nascent technology’
“This is an exciting and courageous step forward in heart transplantation, and kudos to the team at the University of Maryland,” said Mandeep R. Mehra, MD, Brigham and Woman’s Hospital, Boston. But “there are many challenges here.”
The procedure’s 10 gene modifications were reportedly aimed at preventing hyperacute rejection of the heart and its excessive growth after transplantation, and making the organ less immunogenic, Dr. Mehra said in an interview. But even if those goals are met, could the same changes potentially impede the heart’s adaptation to human physiology, such as during ambulation or stress?
That kind of adaptation may become important. For example, Dr. Mehra observed, normally a pig heart “provides flow in a four-footed configuration, and pig temperature is inherently higher than humans by several degrees, so it will be functioning in a relatively hypothermic environment.”
Transplantation remains the gold standard for patients with advanced heart failure despite modern medical and device therapy, Dr. Allen agreed. But “if we can raise pig hearts that provide the organ, and it can be implanted with a surgery that’s been done for 50 years, and rejection can be managed with gene editing and tailored immunosuppression, then it’s not hard to think about this very rapidly replacing a lot of what we do in the advanced heart failure and transplantation world.”
Certainly, it would be a major advance if the gene editing technique successfully improves the heart’s immunologic compatibility, Dr. Yancy noted. But do we have enough genomic knowledge to select gene deletions and insertions in the safest way for a successful outcome? “We have to appreciate that this is still a nascent technology, and we should be careful that there might be consequences that we haven’t anticipated.”
For example, he said, the xenotransplantation and gene-modifying techniques should be explored in a range of patients, including older and younger people, women and men, and people of different ethnicities and races.
“There may be some differences based on ancestry, based on gender, based on aging, that will influence the way in which these engineered donor hearts are experienced clinically,” Dr. Yancy said.
The xenotransplantation technique’s potential impact on health equity should also be considered, as it “almost assuredly will be a very expensive technology that will be utilized in a very select population,” he noted. “We need to have a really wide lens to think about all of the potential ramifications.”
‘This field needs to evolve’
Dr. Mehra also flagged the procedure’s potential cost should it become mainstream. Perhaps that would promote dialogue on how to primarily use it “after legitimately exhausting all available options, such as total artificial heart support.”
It might also teach the field to take greater advantage of the many donated hearts discarded as suboptimal. “The general usage rate for offered organs is around a third,” despite opportunities to expand use of those that are “less than perfect,” Dr. Mehra said. “I think that the field will grow with the community focusing on reduced discards of current available heart organs, and not necessarily grow because of the availability of ‘xeno-organs.’ ”
“This field needs to evolve because we’re actively transplanting patients today. But in my mind, the real future is to have such a sufficient understanding of the biology of left ventricular dysfunction that transplantation is a rare event,” Dr. Yancy proposed.
“I’m not certain that heart transplantation per se is the endgame. I think the avoidance of transplantation is the real endgame,” he said. “This may be controversial, but my vision of the future is not one where we have a supply of animals that we can use for transplantation. My vision of the future is that heart transplantation becomes obsolete.”
A version of this article first appeared on Medscape.com.
Scientific achievements usually raise big new questions, and the remarkable surgery that took place on Jan. 7, when Maryland resident David Bennett was transplanted with a genetically modified heart from a pig, has been no different.
The 57-year-old with end-stage heart failure had been repeatedly turned down for a standard transplant and was judged a poor candidate for a ventricular assist device. Now his new heart is beating soundly and apparently accepted by his immune system as Mr. Bennett, his physicians at the University of Maryland where the procedure took place, and indeed the world set out on a journey with far more unknowns than knowns.
“I think even just a couple of years ago, people felt that xenotransplantation for the heart and other organs was still a long way off. And it seems like it’s started to move very quickly,” Larry A. Allen, MD, University of Colorado, Aurora, said in an interview.
Demand for donor hearts far outstrips supply, and despite advances in the development of ventricular assist pumps and artificial hearts, “there are still significant limitations to them in terms of clotting, stroke, and infection. We’ve seen the use of those devices plateau,” Dr. Allen said. “So, the concept of a nonhuman source of organs is exciting and very much in need, if people can get it to work.”
“I really credit the surgeons at the University of Maryland for courageous clinical work and a brilliant scientific innovation,” Clyde W. Yancy, MD, MSc, Northwestern University, Chicago, said in an interview. “But it’s always in the implementation that we have to hold our breath.” Heart xenotransplantation is an old idea that “has never before been successful,” he said. And standard heart transplantation has set a high bar, with a 1-year survival of about 90% and low 1-year risk for rejection. Whether the new procedure can meet that standard is unknown, as is its potential for complications, such as chronic rejection or cancers due to long-term immunosuppression. Those are “major questions requiring more time and careful follow-up.”
‘Still a nascent technology’
“This is an exciting and courageous step forward in heart transplantation, and kudos to the team at the University of Maryland,” said Mandeep R. Mehra, MD, Brigham and Woman’s Hospital, Boston. But “there are many challenges here.”
The procedure’s 10 gene modifications were reportedly aimed at preventing hyperacute rejection of the heart and its excessive growth after transplantation, and making the organ less immunogenic, Dr. Mehra said in an interview. But even if those goals are met, could the same changes potentially impede the heart’s adaptation to human physiology, such as during ambulation or stress?
That kind of adaptation may become important. For example, Dr. Mehra observed, normally a pig heart “provides flow in a four-footed configuration, and pig temperature is inherently higher than humans by several degrees, so it will be functioning in a relatively hypothermic environment.”
Transplantation remains the gold standard for patients with advanced heart failure despite modern medical and device therapy, Dr. Allen agreed. But “if we can raise pig hearts that provide the organ, and it can be implanted with a surgery that’s been done for 50 years, and rejection can be managed with gene editing and tailored immunosuppression, then it’s not hard to think about this very rapidly replacing a lot of what we do in the advanced heart failure and transplantation world.”
Certainly, it would be a major advance if the gene editing technique successfully improves the heart’s immunologic compatibility, Dr. Yancy noted. But do we have enough genomic knowledge to select gene deletions and insertions in the safest way for a successful outcome? “We have to appreciate that this is still a nascent technology, and we should be careful that there might be consequences that we haven’t anticipated.”
For example, he said, the xenotransplantation and gene-modifying techniques should be explored in a range of patients, including older and younger people, women and men, and people of different ethnicities and races.
“There may be some differences based on ancestry, based on gender, based on aging, that will influence the way in which these engineered donor hearts are experienced clinically,” Dr. Yancy said.
The xenotransplantation technique’s potential impact on health equity should also be considered, as it “almost assuredly will be a very expensive technology that will be utilized in a very select population,” he noted. “We need to have a really wide lens to think about all of the potential ramifications.”
‘This field needs to evolve’
Dr. Mehra also flagged the procedure’s potential cost should it become mainstream. Perhaps that would promote dialogue on how to primarily use it “after legitimately exhausting all available options, such as total artificial heart support.”
It might also teach the field to take greater advantage of the many donated hearts discarded as suboptimal. “The general usage rate for offered organs is around a third,” despite opportunities to expand use of those that are “less than perfect,” Dr. Mehra said. “I think that the field will grow with the community focusing on reduced discards of current available heart organs, and not necessarily grow because of the availability of ‘xeno-organs.’ ”
“This field needs to evolve because we’re actively transplanting patients today. But in my mind, the real future is to have such a sufficient understanding of the biology of left ventricular dysfunction that transplantation is a rare event,” Dr. Yancy proposed.
“I’m not certain that heart transplantation per se is the endgame. I think the avoidance of transplantation is the real endgame,” he said. “This may be controversial, but my vision of the future is not one where we have a supply of animals that we can use for transplantation. My vision of the future is that heart transplantation becomes obsolete.”
A version of this article first appeared on Medscape.com.
Scientific achievements usually raise big new questions, and the remarkable surgery that took place on Jan. 7, when Maryland resident David Bennett was transplanted with a genetically modified heart from a pig, has been no different.
The 57-year-old with end-stage heart failure had been repeatedly turned down for a standard transplant and was judged a poor candidate for a ventricular assist device. Now his new heart is beating soundly and apparently accepted by his immune system as Mr. Bennett, his physicians at the University of Maryland where the procedure took place, and indeed the world set out on a journey with far more unknowns than knowns.
“I think even just a couple of years ago, people felt that xenotransplantation for the heart and other organs was still a long way off. And it seems like it’s started to move very quickly,” Larry A. Allen, MD, University of Colorado, Aurora, said in an interview.
Demand for donor hearts far outstrips supply, and despite advances in the development of ventricular assist pumps and artificial hearts, “there are still significant limitations to them in terms of clotting, stroke, and infection. We’ve seen the use of those devices plateau,” Dr. Allen said. “So, the concept of a nonhuman source of organs is exciting and very much in need, if people can get it to work.”
“I really credit the surgeons at the University of Maryland for courageous clinical work and a brilliant scientific innovation,” Clyde W. Yancy, MD, MSc, Northwestern University, Chicago, said in an interview. “But it’s always in the implementation that we have to hold our breath.” Heart xenotransplantation is an old idea that “has never before been successful,” he said. And standard heart transplantation has set a high bar, with a 1-year survival of about 90% and low 1-year risk for rejection. Whether the new procedure can meet that standard is unknown, as is its potential for complications, such as chronic rejection or cancers due to long-term immunosuppression. Those are “major questions requiring more time and careful follow-up.”
‘Still a nascent technology’
“This is an exciting and courageous step forward in heart transplantation, and kudos to the team at the University of Maryland,” said Mandeep R. Mehra, MD, Brigham and Woman’s Hospital, Boston. But “there are many challenges here.”
The procedure’s 10 gene modifications were reportedly aimed at preventing hyperacute rejection of the heart and its excessive growth after transplantation, and making the organ less immunogenic, Dr. Mehra said in an interview. But even if those goals are met, could the same changes potentially impede the heart’s adaptation to human physiology, such as during ambulation or stress?
That kind of adaptation may become important. For example, Dr. Mehra observed, normally a pig heart “provides flow in a four-footed configuration, and pig temperature is inherently higher than humans by several degrees, so it will be functioning in a relatively hypothermic environment.”
Transplantation remains the gold standard for patients with advanced heart failure despite modern medical and device therapy, Dr. Allen agreed. But “if we can raise pig hearts that provide the organ, and it can be implanted with a surgery that’s been done for 50 years, and rejection can be managed with gene editing and tailored immunosuppression, then it’s not hard to think about this very rapidly replacing a lot of what we do in the advanced heart failure and transplantation world.”
Certainly, it would be a major advance if the gene editing technique successfully improves the heart’s immunologic compatibility, Dr. Yancy noted. But do we have enough genomic knowledge to select gene deletions and insertions in the safest way for a successful outcome? “We have to appreciate that this is still a nascent technology, and we should be careful that there might be consequences that we haven’t anticipated.”
For example, he said, the xenotransplantation and gene-modifying techniques should be explored in a range of patients, including older and younger people, women and men, and people of different ethnicities and races.
“There may be some differences based on ancestry, based on gender, based on aging, that will influence the way in which these engineered donor hearts are experienced clinically,” Dr. Yancy said.
The xenotransplantation technique’s potential impact on health equity should also be considered, as it “almost assuredly will be a very expensive technology that will be utilized in a very select population,” he noted. “We need to have a really wide lens to think about all of the potential ramifications.”
‘This field needs to evolve’
Dr. Mehra also flagged the procedure’s potential cost should it become mainstream. Perhaps that would promote dialogue on how to primarily use it “after legitimately exhausting all available options, such as total artificial heart support.”
It might also teach the field to take greater advantage of the many donated hearts discarded as suboptimal. “The general usage rate for offered organs is around a third,” despite opportunities to expand use of those that are “less than perfect,” Dr. Mehra said. “I think that the field will grow with the community focusing on reduced discards of current available heart organs, and not necessarily grow because of the availability of ‘xeno-organs.’ ”
“This field needs to evolve because we’re actively transplanting patients today. But in my mind, the real future is to have such a sufficient understanding of the biology of left ventricular dysfunction that transplantation is a rare event,” Dr. Yancy proposed.
“I’m not certain that heart transplantation per se is the endgame. I think the avoidance of transplantation is the real endgame,” he said. “This may be controversial, but my vision of the future is not one where we have a supply of animals that we can use for transplantation. My vision of the future is that heart transplantation becomes obsolete.”
A version of this article first appeared on Medscape.com.
Olive oil intake tied to reduced mortality
Compared with men and women who rarely or never consumed olive oil (the lowest intake), those who consumed greater than 0.5 tablespoon/day or more than 7 g/day (the highest intake) had a 19% lower mortality risk over a 28-year follow-up, starting from an average age of 56 years.
Moreover, compared with those with the lowest olive oil intake, those with the highest intake had a 19% lower cardiovascular disease (CVD) mortality, a 17% lower risk of dying from cancer, a 29% lower risk of dying from neurodegenerative disease, and an 18% lower risk of dying from respiratory disease during follow-up.
The researchers estimate that replacing 10 g/day of margarine, butter, mayonnaise, or dairy fat with the same amount of olive oil is associated with an 8%-34% lower risk of death from various causes.
The study by Marta Guasch-Ferré, PhD, and colleagues was published online Jan. 10 in the Journal of the American College of Cardiology.
Results support plant-based dietary fat recommendations
“Our results support current dietary recommendations to increase the intake of olive oil and other unsaturated vegetable oils in place of other fats to improve overall health and longevity,” the researchers summarize.
However, “I wouldn’t say that olive oil is the only way to help you live longer,” Dr. Guasch-Ferré, a senior research scientist in the department of nutrition, Harvard T.H. Chan School of Public Health, Boston, cautioned in an interview with this news organization.
“Other things are very important, such as not smoking, doing physical activity, etc., but one recommendation could be to try to eat more plant-based food including olive oil and healthy fat,” she added, and to use it for cooking, salad dressing, and baking, and substitute it for saturated fat or animal fat, especially for cooking.
The study suggests that people should “consume a more plant-based diet and prioritize fatty acids such as olive oil because they have a better nutritional composition (high in phenols and antioxidants), instead of using butter or margarines or other animal fats that have been shown to have detrimental effects for health,” she added, which is consistent with recommendations in the Dietary Guidelines for Americans.
“That said,” Dr. Guasch-Ferré summarized, “replication is needed in other cohorts and populations to see if the results are similar.”
In an accompanying editorial, Susanna C. Larsson, PhD, writes that “this was a well-designed study, with long-term follow-up and repeated measurements of dietary intake and other risk factors for diseases.”
“However, the difference in olive oil consumption between those with the highest and those with the lowest/no olive oil consumption was very low (0.5 tablespoon) and a [12%] reduced mortality risk was observed already at a much lower intake (0.5 teaspoon, about 1.5 g/day) of olive oil,” she noted in an email to this news organization.
“It’s a bit hard to believe that such a small amount could have an independent effect on mortality risk,” Dr. Larsson, associate professor of epidemiology at the Karolinska Institutet, Stockholm, cautioned.
Like Dr. Guasch-Ferré, she noted that “just adding one or two teaspoons of olive oil to the diet each day will likely not change the risk of mortality.”
Rather, “people may need to make larger changes in the whole diet, not focus on fat only. An overall healthier diet, rich in nonrefined plant-based foods (vegetables, whole grains, nuts), low/no intake of processed foods, and a switch to healthier fat (eg, olive oil) is needed.”
Importantly, “this study cannot say anything about causality, that is, whether it’s olive oil specifically that reduces mortality risk or if there are many other beneficial factors that act together to reduce mortality rate among those with high olive oil consumption.”
The researchers acknowledge this observational study limitation and that the findings may not be generalizable to other populations.
Novel findings regarding Alzheimer’s and respiratory disease
Dr. Larsson highlights two novel findings of this study.
First, it showed a 27% reduction in risk of dementia-related mortality for those in the highest versus lowest category of olive oil consumption. “Considering the lack of preventive strategies for Alzheimer’s disease and the high morbidity and mortality related to this disease, this finding, if confirmed, is of great public health importance,” she said.
Second, the study reported an inverse association of olive oil consumption with risk of respiratory disease mortality. “Because residual confounding from smoking cannot be ruled out,” Dr. Larsson said, “this finding is tentative and requires confirmation in a study that is less susceptible to confounding, such as a randomized trial.”
And although the current study and previous studies have found that consumption of olive oil may have health benefits, she identified several remaining questions.
“Are the associations causal or spurious?” she noted. Is olive oil consumption protective for certain cardiovascular diseases like stroke or atrial fibrillation only, as has been shown in other studies, or also for other major diseases and causes of death, she added. What is the amount of olive oil required for a protective effect?
Further, is the potential effect related to monounsaturated fatty acids (MUFAs) or phenolic compounds; that is, “is the protective effect confined to polyphenol-rich extra-virgin olive oil or are refined olive oil and other vegetable oils as beneficial? More research is needed to address these questions,” she concludes.
“Further studies are needed,” the researchers agree, “to confirm the association of olive oil consumption with reduced mortality, clarify the mechanisms responsible, and quantify the dose/volume boundaries around this effect.”
Virgin olive oil has more polyphenols
Olive oil, a key component of the Mediterranean diet, is high in MUFAs, especially oleic acid, as well as vitamin E and polyphenols, which contribute to its anti-inflammatory and antioxidant properties, the researchers explain.
Virgin olive oil, produced by mechanically pressing ripe olives, contains multiple bioactive and antioxidant components and has an acidity of less than 1.5%. And extra-virgin olive oil is produced the same way but has a higher quality, more intense taste, and lower acidity (less than 1%).
Refined or processed olive oil contains less phytochemicals, as some are lost during processing; it usually contains more than 80% refined oil, plus virgin oil added back to enhance flavor, and may also be labeled “pure” or “light.” However, refined olive oil “still has a good amount of healthy fatty acids but less bioactive compounds,” Dr. Guasch-Ferré noted.
Until now, no large prospective study has examined the link between olive oil intake and all-cause and cause-specific mortality in a U.S. population, where olive oil consumption is limited, compared with Mediterranean countries.
The researchers identified 60,582 women in the Nurses’ Health Study and 31,801 men in the Health Professionals Follow-up Study who were free of CVD or cancer in 1990, the first year that food frequency questionnaires in these studies asked about olive oil.
Participants replied to questionnaires every 4 years that asked about use of olive oil (for salad dressing, baking, frying, sautéing, and spreading on bread), other vegetable oils (for example, corn, safflower, soybean, canola oil), margarine, butter, and dairy fat. The researchers averaged the consumption of these fats during the follow-up years.
From 1990 to 2019, the average consumption of olive oil increased from 1.6 g/day to 4 g/day. Margarine in the 1990s contained saturated fat and trans fats, whereas more recently margarine contains beneficial olive oil or vegetable fat, Dr. Guasch-Ferré noted.
Baseline olive oil consumption in this U.S. population “differed remarkably” from that in the Spanish population in the PREDIMED (Prevención con Dieta Mediterránea) trial, which was, on average, 20-22 g/day of extra-virgin olive oil and 16-18 g/day of refined/mixed olive oil, Larsson pointed out.
Because olive oil consumption was so low in this U.S. study, the researchers did not distinguish between virgin/extra-virgin olive oil and refined/processed olive oil.
The participants were almost all White (99%) and were generally healthier than the average U.S. population; on average, they had a body mass index of 25.3-25.8 kg/m2 and ate 4.8-7.2 fruits and vegetables/day.
Those with the highest olive oil consumption were more physically active, had a healthier diet, were more likely to have Southern European or Mediterranean ancestry, and were less likely to smoke.
During 28 years of follow-up, 36,856 participants died. The researchers classified the deaths into five categories: CVD, cancer, neurodegenerative disease (including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis), respiratory disease (such as chronic obstructive pulmonary disease), and all other causes (including suicide, injury, infections, diabetes, and kidney disease).
After adjusting for multiple confounders, compared with participants who rarely or never consumed olive oil, those in the highest quartile for olive oil consumption had a decreased risk of death from all causes (hazard ratio, 0.81; 95% confidence interval, 0.78 - 0.84) and from CVD (HR, 0.81; 95% CI, 0.75-0.87), cancer (HR, 0.83; 95% CI, 0.78-0.89), neurodegenerative disease (HR, 0.71; 95% CI, 0.64-0.78), and respiratory disease (HR, 0.82; 95% CI, 0.72-0.93).
There was no decrease in mortality in models where the researchers substituted olive oil for vegetable oil, suggesting that vegetable oils may provide similar health benefits as olive oil.
The research was supported by grants from the National Institutes of Health. Dr. Guasch-Ferré was supported by the American Diabetes Association. Coauthor Salas-Salvadó is partially supported by the Catalan Institution for Research and Advanced Studies and received the virgin olive oil that was used in the PREDIMED and PREDIMED-Plus studies from the Patrimonio Communal Olivalero and Hojiblanca (Málaga, Spain). The other study authors and Dr. Larsson have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Compared with men and women who rarely or never consumed olive oil (the lowest intake), those who consumed greater than 0.5 tablespoon/day or more than 7 g/day (the highest intake) had a 19% lower mortality risk over a 28-year follow-up, starting from an average age of 56 years.
Moreover, compared with those with the lowest olive oil intake, those with the highest intake had a 19% lower cardiovascular disease (CVD) mortality, a 17% lower risk of dying from cancer, a 29% lower risk of dying from neurodegenerative disease, and an 18% lower risk of dying from respiratory disease during follow-up.
The researchers estimate that replacing 10 g/day of margarine, butter, mayonnaise, or dairy fat with the same amount of olive oil is associated with an 8%-34% lower risk of death from various causes.
The study by Marta Guasch-Ferré, PhD, and colleagues was published online Jan. 10 in the Journal of the American College of Cardiology.
Results support plant-based dietary fat recommendations
“Our results support current dietary recommendations to increase the intake of olive oil and other unsaturated vegetable oils in place of other fats to improve overall health and longevity,” the researchers summarize.
However, “I wouldn’t say that olive oil is the only way to help you live longer,” Dr. Guasch-Ferré, a senior research scientist in the department of nutrition, Harvard T.H. Chan School of Public Health, Boston, cautioned in an interview with this news organization.
“Other things are very important, such as not smoking, doing physical activity, etc., but one recommendation could be to try to eat more plant-based food including olive oil and healthy fat,” she added, and to use it for cooking, salad dressing, and baking, and substitute it for saturated fat or animal fat, especially for cooking.
The study suggests that people should “consume a more plant-based diet and prioritize fatty acids such as olive oil because they have a better nutritional composition (high in phenols and antioxidants), instead of using butter or margarines or other animal fats that have been shown to have detrimental effects for health,” she added, which is consistent with recommendations in the Dietary Guidelines for Americans.
“That said,” Dr. Guasch-Ferré summarized, “replication is needed in other cohorts and populations to see if the results are similar.”
In an accompanying editorial, Susanna C. Larsson, PhD, writes that “this was a well-designed study, with long-term follow-up and repeated measurements of dietary intake and other risk factors for diseases.”
“However, the difference in olive oil consumption between those with the highest and those with the lowest/no olive oil consumption was very low (0.5 tablespoon) and a [12%] reduced mortality risk was observed already at a much lower intake (0.5 teaspoon, about 1.5 g/day) of olive oil,” she noted in an email to this news organization.
“It’s a bit hard to believe that such a small amount could have an independent effect on mortality risk,” Dr. Larsson, associate professor of epidemiology at the Karolinska Institutet, Stockholm, cautioned.
Like Dr. Guasch-Ferré, she noted that “just adding one or two teaspoons of olive oil to the diet each day will likely not change the risk of mortality.”
Rather, “people may need to make larger changes in the whole diet, not focus on fat only. An overall healthier diet, rich in nonrefined plant-based foods (vegetables, whole grains, nuts), low/no intake of processed foods, and a switch to healthier fat (eg, olive oil) is needed.”
Importantly, “this study cannot say anything about causality, that is, whether it’s olive oil specifically that reduces mortality risk or if there are many other beneficial factors that act together to reduce mortality rate among those with high olive oil consumption.”
The researchers acknowledge this observational study limitation and that the findings may not be generalizable to other populations.
Novel findings regarding Alzheimer’s and respiratory disease
Dr. Larsson highlights two novel findings of this study.
First, it showed a 27% reduction in risk of dementia-related mortality for those in the highest versus lowest category of olive oil consumption. “Considering the lack of preventive strategies for Alzheimer’s disease and the high morbidity and mortality related to this disease, this finding, if confirmed, is of great public health importance,” she said.
Second, the study reported an inverse association of olive oil consumption with risk of respiratory disease mortality. “Because residual confounding from smoking cannot be ruled out,” Dr. Larsson said, “this finding is tentative and requires confirmation in a study that is less susceptible to confounding, such as a randomized trial.”
And although the current study and previous studies have found that consumption of olive oil may have health benefits, she identified several remaining questions.
“Are the associations causal or spurious?” she noted. Is olive oil consumption protective for certain cardiovascular diseases like stroke or atrial fibrillation only, as has been shown in other studies, or also for other major diseases and causes of death, she added. What is the amount of olive oil required for a protective effect?
Further, is the potential effect related to monounsaturated fatty acids (MUFAs) or phenolic compounds; that is, “is the protective effect confined to polyphenol-rich extra-virgin olive oil or are refined olive oil and other vegetable oils as beneficial? More research is needed to address these questions,” she concludes.
“Further studies are needed,” the researchers agree, “to confirm the association of olive oil consumption with reduced mortality, clarify the mechanisms responsible, and quantify the dose/volume boundaries around this effect.”
Virgin olive oil has more polyphenols
Olive oil, a key component of the Mediterranean diet, is high in MUFAs, especially oleic acid, as well as vitamin E and polyphenols, which contribute to its anti-inflammatory and antioxidant properties, the researchers explain.
Virgin olive oil, produced by mechanically pressing ripe olives, contains multiple bioactive and antioxidant components and has an acidity of less than 1.5%. And extra-virgin olive oil is produced the same way but has a higher quality, more intense taste, and lower acidity (less than 1%).
Refined or processed olive oil contains less phytochemicals, as some are lost during processing; it usually contains more than 80% refined oil, plus virgin oil added back to enhance flavor, and may also be labeled “pure” or “light.” However, refined olive oil “still has a good amount of healthy fatty acids but less bioactive compounds,” Dr. Guasch-Ferré noted.
Until now, no large prospective study has examined the link between olive oil intake and all-cause and cause-specific mortality in a U.S. population, where olive oil consumption is limited, compared with Mediterranean countries.
The researchers identified 60,582 women in the Nurses’ Health Study and 31,801 men in the Health Professionals Follow-up Study who were free of CVD or cancer in 1990, the first year that food frequency questionnaires in these studies asked about olive oil.
Participants replied to questionnaires every 4 years that asked about use of olive oil (for salad dressing, baking, frying, sautéing, and spreading on bread), other vegetable oils (for example, corn, safflower, soybean, canola oil), margarine, butter, and dairy fat. The researchers averaged the consumption of these fats during the follow-up years.
From 1990 to 2019, the average consumption of olive oil increased from 1.6 g/day to 4 g/day. Margarine in the 1990s contained saturated fat and trans fats, whereas more recently margarine contains beneficial olive oil or vegetable fat, Dr. Guasch-Ferré noted.
Baseline olive oil consumption in this U.S. population “differed remarkably” from that in the Spanish population in the PREDIMED (Prevención con Dieta Mediterránea) trial, which was, on average, 20-22 g/day of extra-virgin olive oil and 16-18 g/day of refined/mixed olive oil, Larsson pointed out.
Because olive oil consumption was so low in this U.S. study, the researchers did not distinguish between virgin/extra-virgin olive oil and refined/processed olive oil.
The participants were almost all White (99%) and were generally healthier than the average U.S. population; on average, they had a body mass index of 25.3-25.8 kg/m2 and ate 4.8-7.2 fruits and vegetables/day.
Those with the highest olive oil consumption were more physically active, had a healthier diet, were more likely to have Southern European or Mediterranean ancestry, and were less likely to smoke.
During 28 years of follow-up, 36,856 participants died. The researchers classified the deaths into five categories: CVD, cancer, neurodegenerative disease (including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis), respiratory disease (such as chronic obstructive pulmonary disease), and all other causes (including suicide, injury, infections, diabetes, and kidney disease).
After adjusting for multiple confounders, compared with participants who rarely or never consumed olive oil, those in the highest quartile for olive oil consumption had a decreased risk of death from all causes (hazard ratio, 0.81; 95% confidence interval, 0.78 - 0.84) and from CVD (HR, 0.81; 95% CI, 0.75-0.87), cancer (HR, 0.83; 95% CI, 0.78-0.89), neurodegenerative disease (HR, 0.71; 95% CI, 0.64-0.78), and respiratory disease (HR, 0.82; 95% CI, 0.72-0.93).
There was no decrease in mortality in models where the researchers substituted olive oil for vegetable oil, suggesting that vegetable oils may provide similar health benefits as olive oil.
The research was supported by grants from the National Institutes of Health. Dr. Guasch-Ferré was supported by the American Diabetes Association. Coauthor Salas-Salvadó is partially supported by the Catalan Institution for Research and Advanced Studies and received the virgin olive oil that was used in the PREDIMED and PREDIMED-Plus studies from the Patrimonio Communal Olivalero and Hojiblanca (Málaga, Spain). The other study authors and Dr. Larsson have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Compared with men and women who rarely or never consumed olive oil (the lowest intake), those who consumed greater than 0.5 tablespoon/day or more than 7 g/day (the highest intake) had a 19% lower mortality risk over a 28-year follow-up, starting from an average age of 56 years.
Moreover, compared with those with the lowest olive oil intake, those with the highest intake had a 19% lower cardiovascular disease (CVD) mortality, a 17% lower risk of dying from cancer, a 29% lower risk of dying from neurodegenerative disease, and an 18% lower risk of dying from respiratory disease during follow-up.
The researchers estimate that replacing 10 g/day of margarine, butter, mayonnaise, or dairy fat with the same amount of olive oil is associated with an 8%-34% lower risk of death from various causes.
The study by Marta Guasch-Ferré, PhD, and colleagues was published online Jan. 10 in the Journal of the American College of Cardiology.
Results support plant-based dietary fat recommendations
“Our results support current dietary recommendations to increase the intake of olive oil and other unsaturated vegetable oils in place of other fats to improve overall health and longevity,” the researchers summarize.
However, “I wouldn’t say that olive oil is the only way to help you live longer,” Dr. Guasch-Ferré, a senior research scientist in the department of nutrition, Harvard T.H. Chan School of Public Health, Boston, cautioned in an interview with this news organization.
“Other things are very important, such as not smoking, doing physical activity, etc., but one recommendation could be to try to eat more plant-based food including olive oil and healthy fat,” she added, and to use it for cooking, salad dressing, and baking, and substitute it for saturated fat or animal fat, especially for cooking.
The study suggests that people should “consume a more plant-based diet and prioritize fatty acids such as olive oil because they have a better nutritional composition (high in phenols and antioxidants), instead of using butter or margarines or other animal fats that have been shown to have detrimental effects for health,” she added, which is consistent with recommendations in the Dietary Guidelines for Americans.
“That said,” Dr. Guasch-Ferré summarized, “replication is needed in other cohorts and populations to see if the results are similar.”
In an accompanying editorial, Susanna C. Larsson, PhD, writes that “this was a well-designed study, with long-term follow-up and repeated measurements of dietary intake and other risk factors for diseases.”
“However, the difference in olive oil consumption between those with the highest and those with the lowest/no olive oil consumption was very low (0.5 tablespoon) and a [12%] reduced mortality risk was observed already at a much lower intake (0.5 teaspoon, about 1.5 g/day) of olive oil,” she noted in an email to this news organization.
“It’s a bit hard to believe that such a small amount could have an independent effect on mortality risk,” Dr. Larsson, associate professor of epidemiology at the Karolinska Institutet, Stockholm, cautioned.
Like Dr. Guasch-Ferré, she noted that “just adding one or two teaspoons of olive oil to the diet each day will likely not change the risk of mortality.”
Rather, “people may need to make larger changes in the whole diet, not focus on fat only. An overall healthier diet, rich in nonrefined plant-based foods (vegetables, whole grains, nuts), low/no intake of processed foods, and a switch to healthier fat (eg, olive oil) is needed.”
Importantly, “this study cannot say anything about causality, that is, whether it’s olive oil specifically that reduces mortality risk or if there are many other beneficial factors that act together to reduce mortality rate among those with high olive oil consumption.”
The researchers acknowledge this observational study limitation and that the findings may not be generalizable to other populations.
Novel findings regarding Alzheimer’s and respiratory disease
Dr. Larsson highlights two novel findings of this study.
First, it showed a 27% reduction in risk of dementia-related mortality for those in the highest versus lowest category of olive oil consumption. “Considering the lack of preventive strategies for Alzheimer’s disease and the high morbidity and mortality related to this disease, this finding, if confirmed, is of great public health importance,” she said.
Second, the study reported an inverse association of olive oil consumption with risk of respiratory disease mortality. “Because residual confounding from smoking cannot be ruled out,” Dr. Larsson said, “this finding is tentative and requires confirmation in a study that is less susceptible to confounding, such as a randomized trial.”
And although the current study and previous studies have found that consumption of olive oil may have health benefits, she identified several remaining questions.
“Are the associations causal or spurious?” she noted. Is olive oil consumption protective for certain cardiovascular diseases like stroke or atrial fibrillation only, as has been shown in other studies, or also for other major diseases and causes of death, she added. What is the amount of olive oil required for a protective effect?
Further, is the potential effect related to monounsaturated fatty acids (MUFAs) or phenolic compounds; that is, “is the protective effect confined to polyphenol-rich extra-virgin olive oil or are refined olive oil and other vegetable oils as beneficial? More research is needed to address these questions,” she concludes.
“Further studies are needed,” the researchers agree, “to confirm the association of olive oil consumption with reduced mortality, clarify the mechanisms responsible, and quantify the dose/volume boundaries around this effect.”
Virgin olive oil has more polyphenols
Olive oil, a key component of the Mediterranean diet, is high in MUFAs, especially oleic acid, as well as vitamin E and polyphenols, which contribute to its anti-inflammatory and antioxidant properties, the researchers explain.
Virgin olive oil, produced by mechanically pressing ripe olives, contains multiple bioactive and antioxidant components and has an acidity of less than 1.5%. And extra-virgin olive oil is produced the same way but has a higher quality, more intense taste, and lower acidity (less than 1%).
Refined or processed olive oil contains less phytochemicals, as some are lost during processing; it usually contains more than 80% refined oil, plus virgin oil added back to enhance flavor, and may also be labeled “pure” or “light.” However, refined olive oil “still has a good amount of healthy fatty acids but less bioactive compounds,” Dr. Guasch-Ferré noted.
Until now, no large prospective study has examined the link between olive oil intake and all-cause and cause-specific mortality in a U.S. population, where olive oil consumption is limited, compared with Mediterranean countries.
The researchers identified 60,582 women in the Nurses’ Health Study and 31,801 men in the Health Professionals Follow-up Study who were free of CVD or cancer in 1990, the first year that food frequency questionnaires in these studies asked about olive oil.
Participants replied to questionnaires every 4 years that asked about use of olive oil (for salad dressing, baking, frying, sautéing, and spreading on bread), other vegetable oils (for example, corn, safflower, soybean, canola oil), margarine, butter, and dairy fat. The researchers averaged the consumption of these fats during the follow-up years.
From 1990 to 2019, the average consumption of olive oil increased from 1.6 g/day to 4 g/day. Margarine in the 1990s contained saturated fat and trans fats, whereas more recently margarine contains beneficial olive oil or vegetable fat, Dr. Guasch-Ferré noted.
Baseline olive oil consumption in this U.S. population “differed remarkably” from that in the Spanish population in the PREDIMED (Prevención con Dieta Mediterránea) trial, which was, on average, 20-22 g/day of extra-virgin olive oil and 16-18 g/day of refined/mixed olive oil, Larsson pointed out.
Because olive oil consumption was so low in this U.S. study, the researchers did not distinguish between virgin/extra-virgin olive oil and refined/processed olive oil.
The participants were almost all White (99%) and were generally healthier than the average U.S. population; on average, they had a body mass index of 25.3-25.8 kg/m2 and ate 4.8-7.2 fruits and vegetables/day.
Those with the highest olive oil consumption were more physically active, had a healthier diet, were more likely to have Southern European or Mediterranean ancestry, and were less likely to smoke.
During 28 years of follow-up, 36,856 participants died. The researchers classified the deaths into five categories: CVD, cancer, neurodegenerative disease (including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis), respiratory disease (such as chronic obstructive pulmonary disease), and all other causes (including suicide, injury, infections, diabetes, and kidney disease).
After adjusting for multiple confounders, compared with participants who rarely or never consumed olive oil, those in the highest quartile for olive oil consumption had a decreased risk of death from all causes (hazard ratio, 0.81; 95% confidence interval, 0.78 - 0.84) and from CVD (HR, 0.81; 95% CI, 0.75-0.87), cancer (HR, 0.83; 95% CI, 0.78-0.89), neurodegenerative disease (HR, 0.71; 95% CI, 0.64-0.78), and respiratory disease (HR, 0.82; 95% CI, 0.72-0.93).
There was no decrease in mortality in models where the researchers substituted olive oil for vegetable oil, suggesting that vegetable oils may provide similar health benefits as olive oil.
The research was supported by grants from the National Institutes of Health. Dr. Guasch-Ferré was supported by the American Diabetes Association. Coauthor Salas-Salvadó is partially supported by the Catalan Institution for Research and Advanced Studies and received the virgin olive oil that was used in the PREDIMED and PREDIMED-Plus studies from the Patrimonio Communal Olivalero and Hojiblanca (Málaga, Spain). The other study authors and Dr. Larsson have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Health issues in women midlife linked with health decline at 65
Having specific health issues, including depressive symptoms and cardiovascular disease, as a middle-aged woman was associated with experiencing clinically important declines in health later in life, a new study finds.
The most predictive parameters of poorer health at age 65 were cardiovascular disease, clinically significant depressive symptoms, and current smoking. Osteoarthritis, lower education level, and higher body mass index (BMI) also were associated with poorer health status 10 years on, Daniel H. Solomon, MD, MPH and colleagues wrote in their observational study, which was published in JAMA Network Open.
Determining a patient’s score on a health-related quality of life measure based on these variables might be useful in clinical practice to recognize midlife patients at increased risk for later health deterioration, Dr. Solomon, of the division of rheumatology, inflammation, and immunity at Brigham and Women’s Hospital, Boston, said in a statement. This measure is called the Short Form 36 (SF-36), and the researchers specifically focused on the physical component summary score (PCS) of this measure. The SF-36 is similar to the Framingham 10-year coronary heart disease risk prediction score, according to Dr. Solomon, who is a professor of medicine at Harvard Medical School, also in Boston.
Based on their risk scores, women could preemptively target modifiable risk factors before they enter old age, the investigators wrote.
“Age 55-65 may be a critical decade. A person’s health and factors during this period may set them on a path for their later adult years,” Dr. Solomon said in a statement. “The good news is that a large proportion of women at midlife are very stable and will not go on to experience declines. But being able to identify women at higher risk could help lead to interventions targeted to them.”
Study details
The study included a cohort of 1,091 women drawn from the 3,302-participant Study of Women’s Health Across the Nation (SWAN), a racially and ethnically diverse group enrolled from six U.S. sites at or immediately before transition to menopause and followed for 10 years from age 55 to 65. The study sample, consisting of 24.6% Black, 24% Japanese or Chinese, and 51.9% White, had a median baseline age of 54.8 years and median BMI of 27 kg/m2 at entry. The median baseline PCS score was 53.1 (interquartile range, 46.8-56.7).
Over 10 years, 206 (18.9%) of the women in the study experienced clinically important declines of at least 8 points in baseline characteristics at around age 55. The following were significantly associated with these declines:
- Having a higher BMI.
- Having osteoarthritis.
- Having a lower educational level.
- Being a current smoker.
- Having clinically significant depressive symptoms.
- Having cardiovascular disease.
- Having better (or higher) physical health and function score on the PCS.
The association between a higher PCS score and a greater decline might seem like an anomaly, Dr. Solomon said in an interview, but one interpretation of this finding is that women with higher or better scores at baseline have further to fall once other risk factors take effect.
With data analyzed from October 2020 to March 2021, the median 10-year change in PCS was –1.02 points, but 206 women experienced declines of 8 points or more.
Those with health declines were more likely to be Black and less likely to be Japanese. They were also more likely to have other comorbidities such as diabetes, hypertension, and osteoporosis, and to report less physical activity.
Scoring system should not replace individualized evaluation, outside expert said
Commenting on the findings, Margaret J. Nachtigall, MD, a clinical associate professor in the department of obstetrics and gynecology at New York University Langone Health, cautioned that a generalized scoring system should not replace individualized evaluation of women at midlife.
“I assess women around age 55 on a daily basis for health risk factors going forward. And while a number such as BMI can be helpful, I worry that reliance on a score could miss treating the individual,” Dr. Nachtigall said an interview. For instance, one woman might have a high BMI owing to greater muscle mass, which is heavy, while another may have a lower BMI but more fat-related weight, as well as exacerbating conditions such as hypertension that would elevate her risk. “You have to make the calculation for each person.”
Dr. Nachtigall, who was not involved in the SWAN analysis, noted, however, that a big-data scoring system might be a useful adjunct to individual patient evaluation in that “it would make physicians look at all these many risk factors to identify those prone to decline.”
Study includes racially diverse population
According to the authors, while other studies have identified similar and other risk factors such as poor sleep, most have not included such a racially diverse population and have focused on women already in their senior years when the window of opportunity may already have closed.
“As a clinician and epidemiologist, I often think about the window of opportunity at midlife, when people are vital, engaged, and resilient,” said Dr. Solomon in the statement. “If we can identify risk factors and determine who is at risk, we may be able to find interventions that can stave off health declines and help put people on a better health trajectory.”
Eric M. Ascher, DO, who practices family medicine at Lenox Hill Hospital in New York and was not involved in the SWAN research, agreed with Dr. Solomon.
“Doctors who treat chronic conditions often meet patients when they are already suffering from a medical problem,” he said in an interview. “It is key to decrease your risk factors before it is too late.”
Dr. Ascher added that many primary care providers already rely heavily on scoring systems when determining level of risk and type of intervention. “Any additional risk factor-scoring systems that are easy to implement and will prevent chronic diseases would be something providers would want to use with their patients.”
Detailed analyses of larger at-risk populations are needed to validate these risk factors and identify others, the authors said.
SWAN is supported by the National Institute on Aging, the National Institute of Nursing Research, and the National Institutes of Heath’s Office of Research on Women’s Health. Dr. Solomon reported financial ties to Amgen, AbbVie and Moderna, UpToDate, and Arthritis & Rheumatology; as well as serving on the board of directors for the Childhood Arthritis and Rheumatology Research Alliance and an advisory committee for the Food and Drug Administration outside of this work. Dr. Nachtigall and Dr. Ascher disclosed no conflicts of interest with regard to their comments.
Having specific health issues, including depressive symptoms and cardiovascular disease, as a middle-aged woman was associated with experiencing clinically important declines in health later in life, a new study finds.
The most predictive parameters of poorer health at age 65 were cardiovascular disease, clinically significant depressive symptoms, and current smoking. Osteoarthritis, lower education level, and higher body mass index (BMI) also were associated with poorer health status 10 years on, Daniel H. Solomon, MD, MPH and colleagues wrote in their observational study, which was published in JAMA Network Open.
Determining a patient’s score on a health-related quality of life measure based on these variables might be useful in clinical practice to recognize midlife patients at increased risk for later health deterioration, Dr. Solomon, of the division of rheumatology, inflammation, and immunity at Brigham and Women’s Hospital, Boston, said in a statement. This measure is called the Short Form 36 (SF-36), and the researchers specifically focused on the physical component summary score (PCS) of this measure. The SF-36 is similar to the Framingham 10-year coronary heart disease risk prediction score, according to Dr. Solomon, who is a professor of medicine at Harvard Medical School, also in Boston.
Based on their risk scores, women could preemptively target modifiable risk factors before they enter old age, the investigators wrote.
“Age 55-65 may be a critical decade. A person’s health and factors during this period may set them on a path for their later adult years,” Dr. Solomon said in a statement. “The good news is that a large proportion of women at midlife are very stable and will not go on to experience declines. But being able to identify women at higher risk could help lead to interventions targeted to them.”
Study details
The study included a cohort of 1,091 women drawn from the 3,302-participant Study of Women’s Health Across the Nation (SWAN), a racially and ethnically diverse group enrolled from six U.S. sites at or immediately before transition to menopause and followed for 10 years from age 55 to 65. The study sample, consisting of 24.6% Black, 24% Japanese or Chinese, and 51.9% White, had a median baseline age of 54.8 years and median BMI of 27 kg/m2 at entry. The median baseline PCS score was 53.1 (interquartile range, 46.8-56.7).
Over 10 years, 206 (18.9%) of the women in the study experienced clinically important declines of at least 8 points in baseline characteristics at around age 55. The following were significantly associated with these declines:
- Having a higher BMI.
- Having osteoarthritis.
- Having a lower educational level.
- Being a current smoker.
- Having clinically significant depressive symptoms.
- Having cardiovascular disease.
- Having better (or higher) physical health and function score on the PCS.
The association between a higher PCS score and a greater decline might seem like an anomaly, Dr. Solomon said in an interview, but one interpretation of this finding is that women with higher or better scores at baseline have further to fall once other risk factors take effect.
With data analyzed from October 2020 to March 2021, the median 10-year change in PCS was –1.02 points, but 206 women experienced declines of 8 points or more.
Those with health declines were more likely to be Black and less likely to be Japanese. They were also more likely to have other comorbidities such as diabetes, hypertension, and osteoporosis, and to report less physical activity.
Scoring system should not replace individualized evaluation, outside expert said
Commenting on the findings, Margaret J. Nachtigall, MD, a clinical associate professor in the department of obstetrics and gynecology at New York University Langone Health, cautioned that a generalized scoring system should not replace individualized evaluation of women at midlife.
“I assess women around age 55 on a daily basis for health risk factors going forward. And while a number such as BMI can be helpful, I worry that reliance on a score could miss treating the individual,” Dr. Nachtigall said an interview. For instance, one woman might have a high BMI owing to greater muscle mass, which is heavy, while another may have a lower BMI but more fat-related weight, as well as exacerbating conditions such as hypertension that would elevate her risk. “You have to make the calculation for each person.”
Dr. Nachtigall, who was not involved in the SWAN analysis, noted, however, that a big-data scoring system might be a useful adjunct to individual patient evaluation in that “it would make physicians look at all these many risk factors to identify those prone to decline.”
Study includes racially diverse population
According to the authors, while other studies have identified similar and other risk factors such as poor sleep, most have not included such a racially diverse population and have focused on women already in their senior years when the window of opportunity may already have closed.
“As a clinician and epidemiologist, I often think about the window of opportunity at midlife, when people are vital, engaged, and resilient,” said Dr. Solomon in the statement. “If we can identify risk factors and determine who is at risk, we may be able to find interventions that can stave off health declines and help put people on a better health trajectory.”
Eric M. Ascher, DO, who practices family medicine at Lenox Hill Hospital in New York and was not involved in the SWAN research, agreed with Dr. Solomon.
“Doctors who treat chronic conditions often meet patients when they are already suffering from a medical problem,” he said in an interview. “It is key to decrease your risk factors before it is too late.”
Dr. Ascher added that many primary care providers already rely heavily on scoring systems when determining level of risk and type of intervention. “Any additional risk factor-scoring systems that are easy to implement and will prevent chronic diseases would be something providers would want to use with their patients.”
Detailed analyses of larger at-risk populations are needed to validate these risk factors and identify others, the authors said.
SWAN is supported by the National Institute on Aging, the National Institute of Nursing Research, and the National Institutes of Heath’s Office of Research on Women’s Health. Dr. Solomon reported financial ties to Amgen, AbbVie and Moderna, UpToDate, and Arthritis & Rheumatology; as well as serving on the board of directors for the Childhood Arthritis and Rheumatology Research Alliance and an advisory committee for the Food and Drug Administration outside of this work. Dr. Nachtigall and Dr. Ascher disclosed no conflicts of interest with regard to their comments.
Having specific health issues, including depressive symptoms and cardiovascular disease, as a middle-aged woman was associated with experiencing clinically important declines in health later in life, a new study finds.
The most predictive parameters of poorer health at age 65 were cardiovascular disease, clinically significant depressive symptoms, and current smoking. Osteoarthritis, lower education level, and higher body mass index (BMI) also were associated with poorer health status 10 years on, Daniel H. Solomon, MD, MPH and colleagues wrote in their observational study, which was published in JAMA Network Open.
Determining a patient’s score on a health-related quality of life measure based on these variables might be useful in clinical practice to recognize midlife patients at increased risk for later health deterioration, Dr. Solomon, of the division of rheumatology, inflammation, and immunity at Brigham and Women’s Hospital, Boston, said in a statement. This measure is called the Short Form 36 (SF-36), and the researchers specifically focused on the physical component summary score (PCS) of this measure. The SF-36 is similar to the Framingham 10-year coronary heart disease risk prediction score, according to Dr. Solomon, who is a professor of medicine at Harvard Medical School, also in Boston.
Based on their risk scores, women could preemptively target modifiable risk factors before they enter old age, the investigators wrote.
“Age 55-65 may be a critical decade. A person’s health and factors during this period may set them on a path for their later adult years,” Dr. Solomon said in a statement. “The good news is that a large proportion of women at midlife are very stable and will not go on to experience declines. But being able to identify women at higher risk could help lead to interventions targeted to them.”
Study details
The study included a cohort of 1,091 women drawn from the 3,302-participant Study of Women’s Health Across the Nation (SWAN), a racially and ethnically diverse group enrolled from six U.S. sites at or immediately before transition to menopause and followed for 10 years from age 55 to 65. The study sample, consisting of 24.6% Black, 24% Japanese or Chinese, and 51.9% White, had a median baseline age of 54.8 years and median BMI of 27 kg/m2 at entry. The median baseline PCS score was 53.1 (interquartile range, 46.8-56.7).
Over 10 years, 206 (18.9%) of the women in the study experienced clinically important declines of at least 8 points in baseline characteristics at around age 55. The following were significantly associated with these declines:
- Having a higher BMI.
- Having osteoarthritis.
- Having a lower educational level.
- Being a current smoker.
- Having clinically significant depressive symptoms.
- Having cardiovascular disease.
- Having better (or higher) physical health and function score on the PCS.
The association between a higher PCS score and a greater decline might seem like an anomaly, Dr. Solomon said in an interview, but one interpretation of this finding is that women with higher or better scores at baseline have further to fall once other risk factors take effect.
With data analyzed from October 2020 to March 2021, the median 10-year change in PCS was –1.02 points, but 206 women experienced declines of 8 points or more.
Those with health declines were more likely to be Black and less likely to be Japanese. They were also more likely to have other comorbidities such as diabetes, hypertension, and osteoporosis, and to report less physical activity.
Scoring system should not replace individualized evaluation, outside expert said
Commenting on the findings, Margaret J. Nachtigall, MD, a clinical associate professor in the department of obstetrics and gynecology at New York University Langone Health, cautioned that a generalized scoring system should not replace individualized evaluation of women at midlife.
“I assess women around age 55 on a daily basis for health risk factors going forward. And while a number such as BMI can be helpful, I worry that reliance on a score could miss treating the individual,” Dr. Nachtigall said an interview. For instance, one woman might have a high BMI owing to greater muscle mass, which is heavy, while another may have a lower BMI but more fat-related weight, as well as exacerbating conditions such as hypertension that would elevate her risk. “You have to make the calculation for each person.”
Dr. Nachtigall, who was not involved in the SWAN analysis, noted, however, that a big-data scoring system might be a useful adjunct to individual patient evaluation in that “it would make physicians look at all these many risk factors to identify those prone to decline.”
Study includes racially diverse population
According to the authors, while other studies have identified similar and other risk factors such as poor sleep, most have not included such a racially diverse population and have focused on women already in their senior years when the window of opportunity may already have closed.
“As a clinician and epidemiologist, I often think about the window of opportunity at midlife, when people are vital, engaged, and resilient,” said Dr. Solomon in the statement. “If we can identify risk factors and determine who is at risk, we may be able to find interventions that can stave off health declines and help put people on a better health trajectory.”
Eric M. Ascher, DO, who practices family medicine at Lenox Hill Hospital in New York and was not involved in the SWAN research, agreed with Dr. Solomon.
“Doctors who treat chronic conditions often meet patients when they are already suffering from a medical problem,” he said in an interview. “It is key to decrease your risk factors before it is too late.”
Dr. Ascher added that many primary care providers already rely heavily on scoring systems when determining level of risk and type of intervention. “Any additional risk factor-scoring systems that are easy to implement and will prevent chronic diseases would be something providers would want to use with their patients.”
Detailed analyses of larger at-risk populations are needed to validate these risk factors and identify others, the authors said.
SWAN is supported by the National Institute on Aging, the National Institute of Nursing Research, and the National Institutes of Heath’s Office of Research on Women’s Health. Dr. Solomon reported financial ties to Amgen, AbbVie and Moderna, UpToDate, and Arthritis & Rheumatology; as well as serving on the board of directors for the Childhood Arthritis and Rheumatology Research Alliance and an advisory committee for the Food and Drug Administration outside of this work. Dr. Nachtigall and Dr. Ascher disclosed no conflicts of interest with regard to their comments.
FROM JAMA NETWORK OPEN
Pig heart successfully transplanted to man
A genetically modified pig heart has been successfully transplanted into a 57-year-old man who had no other treatment options but is “doing well” 3 days after the procedure, officials at the University of Maryland Medical Center (UMMC), Baltimore, announced Jan. 10.
“This organ transplant demonstrated for the first time that a genetically modified animal heart can function like a human heart without immediate rejection by the body,” they said.
Three genes associated with antibody-mediated rejection had been knocked out in the pig supplying the transplanted heart, and six human genes associated with immune acceptance of the organ had been inserted into the pig’s genome, notes a UMMC press release.
“Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue, which totaled 10 unique gene edits made in the donor pig,” the release states.
The patient, Maryland resident David Bennett, had required mechanical circulatory support to stay alive but was rejected for standard heart transplantation at UMMC and other centers. He was ineligible for an implanted ventricular assist device due to ventricular arrhythmias.
Mr. Bennett “is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits,” the announcement says.
“We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” notes a quote from Bartley P. Griffith, MD, the UMMC surgeon who performed the procedure.
The pig supplying the heart was provided to the center by Revivicor (Blacksburg, Virginia), a regenerative medicine company. An experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Massachusetts) was also used, in addition to standard immunosuppressants.
A version of this article first appeared on Medscape.com.
A genetically modified pig heart has been successfully transplanted into a 57-year-old man who had no other treatment options but is “doing well” 3 days after the procedure, officials at the University of Maryland Medical Center (UMMC), Baltimore, announced Jan. 10.
“This organ transplant demonstrated for the first time that a genetically modified animal heart can function like a human heart without immediate rejection by the body,” they said.
Three genes associated with antibody-mediated rejection had been knocked out in the pig supplying the transplanted heart, and six human genes associated with immune acceptance of the organ had been inserted into the pig’s genome, notes a UMMC press release.
“Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue, which totaled 10 unique gene edits made in the donor pig,” the release states.
The patient, Maryland resident David Bennett, had required mechanical circulatory support to stay alive but was rejected for standard heart transplantation at UMMC and other centers. He was ineligible for an implanted ventricular assist device due to ventricular arrhythmias.
Mr. Bennett “is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits,” the announcement says.
“We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” notes a quote from Bartley P. Griffith, MD, the UMMC surgeon who performed the procedure.
The pig supplying the heart was provided to the center by Revivicor (Blacksburg, Virginia), a regenerative medicine company. An experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Massachusetts) was also used, in addition to standard immunosuppressants.
A version of this article first appeared on Medscape.com.
A genetically modified pig heart has been successfully transplanted into a 57-year-old man who had no other treatment options but is “doing well” 3 days after the procedure, officials at the University of Maryland Medical Center (UMMC), Baltimore, announced Jan. 10.
“This organ transplant demonstrated for the first time that a genetically modified animal heart can function like a human heart without immediate rejection by the body,” they said.
Three genes associated with antibody-mediated rejection had been knocked out in the pig supplying the transplanted heart, and six human genes associated with immune acceptance of the organ had been inserted into the pig’s genome, notes a UMMC press release.
“Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue, which totaled 10 unique gene edits made in the donor pig,” the release states.
The patient, Maryland resident David Bennett, had required mechanical circulatory support to stay alive but was rejected for standard heart transplantation at UMMC and other centers. He was ineligible for an implanted ventricular assist device due to ventricular arrhythmias.
Mr. Bennett “is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits,” the announcement says.
“We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” notes a quote from Bartley P. Griffith, MD, the UMMC surgeon who performed the procedure.
The pig supplying the heart was provided to the center by Revivicor (Blacksburg, Virginia), a regenerative medicine company. An experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Massachusetts) was also used, in addition to standard immunosuppressants.
A version of this article first appeared on Medscape.com.
Is outpatient care as safe as inpatient for TIA, minor stroke?
In a meta-analysis of more than 200,000 patients with TIA or mIS, risk for subsequent stroke within 90 days was 2.1% for those treated in a TIA clinic versus 2.8% for patients treated in inpatient settings, which was not significantly different. The risk for patients treated in an emergency department was higher, at 3.5%.
“The message is that if you do the correct risk stratification and then triage patients based on their risk profile, you can safely discharge and have a timely follow-up for the patients who have low risk for a subsequent event,” said coinvestigator Ramin Zand, MD, vascular neurologist and stroke attending physician at Geisinger Health System, Danville, Pennsylvania.
The findings were published online Jan. 5 in JAMA Network Open.
Higher risk in EDs
There is currently no consensus on the care protocol for patients with TIA or mIS, and the rate at which these patients are hospitalized varies by region, hospital, and practitioner, the investigators noted.
Previous studies have indicated that outpatient management of certain individuals with TIA can be safe and cost-effective.
The current researchers searched for retrospective and prospective studies of adult patients that provided information about ischemic stroke after TIA or mIS. Studies that used time- and tissue-based definitions of TIA were included, as well as studies that used various definitions of mIS.
The investigators examined care provided at TIA clinics, inpatient settings (such as medical-surgical units, stroke units, or observation units), EDs, and unspecified settings. Their main aim was to compare outcomes between TIA clinics and inpatient settings.
In all, 226,683 patients (recruited between 1981 and 2018) from 71 studies were included in the meta-analysis. The studies examined 101 cohorts, 24 of which were studied prospectively. Among the 5,636 patients who received care in TIA clinics, the mean age was 65.7 years, and 50.8% of this group were men. Among the 130,139 inpatients, the mean age was 78.3 years, and 61.6% of the group were women.
Results showed no significant difference in risk for subsequent stroke between patients treated in the inpatient and outpatient settings.
Among patients treated in a TIA clinic, risk for subsequent stroke following a TIA or mIS was 0.3% within 2 days, 1.0% within 7 days, 1.3% within 30 days, and 2.1% within 90 days. Among those treated as inpatients, risk for subsequent stroke was 0.5% within 2 days, 1.2% within 7 days, 1.6% within 30 days, and 2.8% within 90 days.
Risk for subsequent stroke was higher among patients treated in the ED and in unspecified settings. At the EDs, the risk was 1.9% within 2 days, 3.4% within 7 days, 3.5% within 30 days, and 3.5% within 90 days. Among those treated in unspecified settings, the risk was 2.2% within 2 days, 3.4% within 7 days, 4.2% within 30 days, and 6.0% within 90 days.
Patients treated in the ED also had a significantly higher risk for subsequent stroke at 2 and 7 days, compared with those treated in inpatient settings and a significantly higher risk for subsequent stroke at 2, 7, and 90 days, compared with those treated in TIA clinics.
‘Most comprehensive look’
“This is the most comprehensive look at all the studies to try and answer this research question,” said Dr. Zand. The results were similar to what was expected, he added.
The infrastructure and resources differed among the sites at which the various studies were conducted, and the investigators adjusted for these differences as much as possible, Dr. Zand noted. A certain amount of selection bias may remain, but it does not affect the overall conclusion, he added.
“Timely outpatient care among low-risk TIA patients is both feasible and safe,” he said.
Dr. Zand noted that the findings have implications not only for patient management but also for the management of the health system. “It’s not feasible nor desirable to admit all the TIA patients, especially with the lessons that we learned from COVID, the burden on the health systems, and the fact that many hospitals are operating at full capacity right now,” he said.
The recommendation is to hospitalize high-risk patients and provide outpatient evaluation and workup to low-risk patients, he added. “This is exactly what we saw in this study,” Dr. Zand said.
Selection bias?
Commenting on the research, Louis R. Caplan, MD, professor of neurology at Harvard Medical School, Boston, noted that evaluation of patients with TIA or mIS “can be done very well as an outpatient” if clinicians have experienced personnel, the outpatient facilities to do the studies necessary, and criteria in place for deciding who to admit or not admit.
However, the decision on whether to choose an inpatient or outpatient approach for a particular patient is complicated, said Dr. Caplan, who was not involved with the research.
Clinicians must consider factors such as whether the patient is mobile, has a car, or has a significant other. The patient’s symptoms and past illnesses also influence the decision, he added.
Dr. Caplan noted that in the meta-analysis, far fewer patients were seen in the TIA clinics than were seen in the inpatient setting. In addition, none of the studies used uniform criteria to determine which patients should undergo workup as outpatients and which as inpatients. “There was a lot of selection bias that may have had nothing to do with how sick the person was,” Dr. Caplan said.
In addition, few hospitals in the United States have an outpatient TIA clinic, he noted. Most of the studies of TIA clinics that the researchers examined were conducted in Europe. “It’s easier to do [that] in Europe because of their socialized medicine,” said Dr. Caplan.
But TIA clinics should be more widespread in the U.S., he added. “Insurance companies should be willing to pay for comparable facilities, inpatient and outpatient,” he said.
The study was conducted without external funding. Dr. Zand reported no relevant financial relationships. Dr. Caplan was an investigator for TIAregistry.org, which analyzed the outcomes of treatment in TIA clinics in Europe.
A version of this article first appeared on Medscape.com.
In a meta-analysis of more than 200,000 patients with TIA or mIS, risk for subsequent stroke within 90 days was 2.1% for those treated in a TIA clinic versus 2.8% for patients treated in inpatient settings, which was not significantly different. The risk for patients treated in an emergency department was higher, at 3.5%.
“The message is that if you do the correct risk stratification and then triage patients based on their risk profile, you can safely discharge and have a timely follow-up for the patients who have low risk for a subsequent event,” said coinvestigator Ramin Zand, MD, vascular neurologist and stroke attending physician at Geisinger Health System, Danville, Pennsylvania.
The findings were published online Jan. 5 in JAMA Network Open.
Higher risk in EDs
There is currently no consensus on the care protocol for patients with TIA or mIS, and the rate at which these patients are hospitalized varies by region, hospital, and practitioner, the investigators noted.
Previous studies have indicated that outpatient management of certain individuals with TIA can be safe and cost-effective.
The current researchers searched for retrospective and prospective studies of adult patients that provided information about ischemic stroke after TIA or mIS. Studies that used time- and tissue-based definitions of TIA were included, as well as studies that used various definitions of mIS.
The investigators examined care provided at TIA clinics, inpatient settings (such as medical-surgical units, stroke units, or observation units), EDs, and unspecified settings. Their main aim was to compare outcomes between TIA clinics and inpatient settings.
In all, 226,683 patients (recruited between 1981 and 2018) from 71 studies were included in the meta-analysis. The studies examined 101 cohorts, 24 of which were studied prospectively. Among the 5,636 patients who received care in TIA clinics, the mean age was 65.7 years, and 50.8% of this group were men. Among the 130,139 inpatients, the mean age was 78.3 years, and 61.6% of the group were women.
Results showed no significant difference in risk for subsequent stroke between patients treated in the inpatient and outpatient settings.
Among patients treated in a TIA clinic, risk for subsequent stroke following a TIA or mIS was 0.3% within 2 days, 1.0% within 7 days, 1.3% within 30 days, and 2.1% within 90 days. Among those treated as inpatients, risk for subsequent stroke was 0.5% within 2 days, 1.2% within 7 days, 1.6% within 30 days, and 2.8% within 90 days.
Risk for subsequent stroke was higher among patients treated in the ED and in unspecified settings. At the EDs, the risk was 1.9% within 2 days, 3.4% within 7 days, 3.5% within 30 days, and 3.5% within 90 days. Among those treated in unspecified settings, the risk was 2.2% within 2 days, 3.4% within 7 days, 4.2% within 30 days, and 6.0% within 90 days.
Patients treated in the ED also had a significantly higher risk for subsequent stroke at 2 and 7 days, compared with those treated in inpatient settings and a significantly higher risk for subsequent stroke at 2, 7, and 90 days, compared with those treated in TIA clinics.
‘Most comprehensive look’
“This is the most comprehensive look at all the studies to try and answer this research question,” said Dr. Zand. The results were similar to what was expected, he added.
The infrastructure and resources differed among the sites at which the various studies were conducted, and the investigators adjusted for these differences as much as possible, Dr. Zand noted. A certain amount of selection bias may remain, but it does not affect the overall conclusion, he added.
“Timely outpatient care among low-risk TIA patients is both feasible and safe,” he said.
Dr. Zand noted that the findings have implications not only for patient management but also for the management of the health system. “It’s not feasible nor desirable to admit all the TIA patients, especially with the lessons that we learned from COVID, the burden on the health systems, and the fact that many hospitals are operating at full capacity right now,” he said.
The recommendation is to hospitalize high-risk patients and provide outpatient evaluation and workup to low-risk patients, he added. “This is exactly what we saw in this study,” Dr. Zand said.
Selection bias?
Commenting on the research, Louis R. Caplan, MD, professor of neurology at Harvard Medical School, Boston, noted that evaluation of patients with TIA or mIS “can be done very well as an outpatient” if clinicians have experienced personnel, the outpatient facilities to do the studies necessary, and criteria in place for deciding who to admit or not admit.
However, the decision on whether to choose an inpatient or outpatient approach for a particular patient is complicated, said Dr. Caplan, who was not involved with the research.
Clinicians must consider factors such as whether the patient is mobile, has a car, or has a significant other. The patient’s symptoms and past illnesses also influence the decision, he added.
Dr. Caplan noted that in the meta-analysis, far fewer patients were seen in the TIA clinics than were seen in the inpatient setting. In addition, none of the studies used uniform criteria to determine which patients should undergo workup as outpatients and which as inpatients. “There was a lot of selection bias that may have had nothing to do with how sick the person was,” Dr. Caplan said.
In addition, few hospitals in the United States have an outpatient TIA clinic, he noted. Most of the studies of TIA clinics that the researchers examined were conducted in Europe. “It’s easier to do [that] in Europe because of their socialized medicine,” said Dr. Caplan.
But TIA clinics should be more widespread in the U.S., he added. “Insurance companies should be willing to pay for comparable facilities, inpatient and outpatient,” he said.
The study was conducted without external funding. Dr. Zand reported no relevant financial relationships. Dr. Caplan was an investigator for TIAregistry.org, which analyzed the outcomes of treatment in TIA clinics in Europe.
A version of this article first appeared on Medscape.com.
In a meta-analysis of more than 200,000 patients with TIA or mIS, risk for subsequent stroke within 90 days was 2.1% for those treated in a TIA clinic versus 2.8% for patients treated in inpatient settings, which was not significantly different. The risk for patients treated in an emergency department was higher, at 3.5%.
“The message is that if you do the correct risk stratification and then triage patients based on their risk profile, you can safely discharge and have a timely follow-up for the patients who have low risk for a subsequent event,” said coinvestigator Ramin Zand, MD, vascular neurologist and stroke attending physician at Geisinger Health System, Danville, Pennsylvania.
The findings were published online Jan. 5 in JAMA Network Open.
Higher risk in EDs
There is currently no consensus on the care protocol for patients with TIA or mIS, and the rate at which these patients are hospitalized varies by region, hospital, and practitioner, the investigators noted.
Previous studies have indicated that outpatient management of certain individuals with TIA can be safe and cost-effective.
The current researchers searched for retrospective and prospective studies of adult patients that provided information about ischemic stroke after TIA or mIS. Studies that used time- and tissue-based definitions of TIA were included, as well as studies that used various definitions of mIS.
The investigators examined care provided at TIA clinics, inpatient settings (such as medical-surgical units, stroke units, or observation units), EDs, and unspecified settings. Their main aim was to compare outcomes between TIA clinics and inpatient settings.
In all, 226,683 patients (recruited between 1981 and 2018) from 71 studies were included in the meta-analysis. The studies examined 101 cohorts, 24 of which were studied prospectively. Among the 5,636 patients who received care in TIA clinics, the mean age was 65.7 years, and 50.8% of this group were men. Among the 130,139 inpatients, the mean age was 78.3 years, and 61.6% of the group were women.
Results showed no significant difference in risk for subsequent stroke between patients treated in the inpatient and outpatient settings.
Among patients treated in a TIA clinic, risk for subsequent stroke following a TIA or mIS was 0.3% within 2 days, 1.0% within 7 days, 1.3% within 30 days, and 2.1% within 90 days. Among those treated as inpatients, risk for subsequent stroke was 0.5% within 2 days, 1.2% within 7 days, 1.6% within 30 days, and 2.8% within 90 days.
Risk for subsequent stroke was higher among patients treated in the ED and in unspecified settings. At the EDs, the risk was 1.9% within 2 days, 3.4% within 7 days, 3.5% within 30 days, and 3.5% within 90 days. Among those treated in unspecified settings, the risk was 2.2% within 2 days, 3.4% within 7 days, 4.2% within 30 days, and 6.0% within 90 days.
Patients treated in the ED also had a significantly higher risk for subsequent stroke at 2 and 7 days, compared with those treated in inpatient settings and a significantly higher risk for subsequent stroke at 2, 7, and 90 days, compared with those treated in TIA clinics.
‘Most comprehensive look’
“This is the most comprehensive look at all the studies to try and answer this research question,” said Dr. Zand. The results were similar to what was expected, he added.
The infrastructure and resources differed among the sites at which the various studies were conducted, and the investigators adjusted for these differences as much as possible, Dr. Zand noted. A certain amount of selection bias may remain, but it does not affect the overall conclusion, he added.
“Timely outpatient care among low-risk TIA patients is both feasible and safe,” he said.
Dr. Zand noted that the findings have implications not only for patient management but also for the management of the health system. “It’s not feasible nor desirable to admit all the TIA patients, especially with the lessons that we learned from COVID, the burden on the health systems, and the fact that many hospitals are operating at full capacity right now,” he said.
The recommendation is to hospitalize high-risk patients and provide outpatient evaluation and workup to low-risk patients, he added. “This is exactly what we saw in this study,” Dr. Zand said.
Selection bias?
Commenting on the research, Louis R. Caplan, MD, professor of neurology at Harvard Medical School, Boston, noted that evaluation of patients with TIA or mIS “can be done very well as an outpatient” if clinicians have experienced personnel, the outpatient facilities to do the studies necessary, and criteria in place for deciding who to admit or not admit.
However, the decision on whether to choose an inpatient or outpatient approach for a particular patient is complicated, said Dr. Caplan, who was not involved with the research.
Clinicians must consider factors such as whether the patient is mobile, has a car, or has a significant other. The patient’s symptoms and past illnesses also influence the decision, he added.
Dr. Caplan noted that in the meta-analysis, far fewer patients were seen in the TIA clinics than were seen in the inpatient setting. In addition, none of the studies used uniform criteria to determine which patients should undergo workup as outpatients and which as inpatients. “There was a lot of selection bias that may have had nothing to do with how sick the person was,” Dr. Caplan said.
In addition, few hospitals in the United States have an outpatient TIA clinic, he noted. Most of the studies of TIA clinics that the researchers examined were conducted in Europe. “It’s easier to do [that] in Europe because of their socialized medicine,” said Dr. Caplan.
But TIA clinics should be more widespread in the U.S., he added. “Insurance companies should be willing to pay for comparable facilities, inpatient and outpatient,” he said.
The study was conducted without external funding. Dr. Zand reported no relevant financial relationships. Dr. Caplan was an investigator for TIAregistry.org, which analyzed the outcomes of treatment in TIA clinics in Europe.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
COVID-vaccine myocarditis: Rare, mild, and usually in young men
The risk of myocarditis after immunization with mRNA-based vaccines against SARS-CoV-2 raised concerns when it came to light in early 2021. But as report after report showed such cases to be rare and usually mild and self-limited, focus has turned to the “how and why.”
The mechanism linking the BNT162b2 (Pfizer-BioNTech) and especially mRNA-1273 (Moderna) vaccines to the occurrence of myocarditis is unclear for now, but one potential driver may be tied to a peculiarity that became apparent early: It occurs overwhelmingly in younger males, from 16 to perhaps 40 or 50 years of age. Excess risk has not been consistently seen among women, girls, and older men.
That observation has led to speculation that higher testosterone levels in adolescent boys and young men may somehow promote the adverse vaccine effect, whereas greater levels of estrogen among girls and women in the same age range may be cardioprotective.
Unlikely, brief, and ‘benign’
“Most of the myocarditis is benign, by which I mean that maybe the patients are admitted due to chest pain, but without reduction in ventricular function,” Enrico Ammirati, MD, PhD, a myocarditis expert at De Gasperis Cardio Center and Transplant Center, Niguarda Hospital, Milan, said in an interview.
In a Nov. 14 address on this topic at the annual scientific sessions of the American Heart Association, Dror Mevorach, MD, described the typical case presentation as “mild” and one that clears in fairly short order based on resolution of “clinical symptoms, inflammatory markers and troponin decline, EKG normalization, echo normalization, and a relatively short length of hospital stay.”
Dr. Mevorach, of Hadassah Hebrew University Medical Center, Jerusalem, subsequently published the findings in a report in the New England Journal of Medicine that described 136 confirmed myocarditis cases among more than 5 million people in Israel immunized with the Pfizer-BioNTech vaccine. Myocarditis was considered “mild” in 129 cases, or 95%.
And the risk is tiny, compared with myocarditis from infection by SARS-CoV-2, not to mention the possibility of nasty clinical COVID-19 complications such as pneumonia and pulmonary embolism, Dr. Mevorach observed.
Many other reports agree that the incidence is minimal, especially given the rewards of vaccination. In a separate NEJM publication in September 2021 – from Noam Barda, MD, Clalit (Israel) Research Institute, and colleagues on 1.7 million people in that country, about half unvaccinated and half given the Pfizer-BioNTech vaccine – there were an estimated 2.7 cases of myocarditis per 100,000 vaccinated persons. There were also 11 cases of myocarditis per 100,000 persons who were positive for SARS-CoV-2 infection.
And in a recent case series of vaccinated people aged 16 or older, the myocarditis rate after a first or second Pfizer-BioNTech or Moderna injection was estimated at 1 or fewer per 100,000. The corresponding estimate was 4 such cases per 100,000 after a positive SARS-CoV-2 test among the same population, notes a report published Dec.14, 2021, in Nature Medicine.
In general, “the risk of any kind of cardiac injury is vastly lower with a vaccine than it is with the actual viral infection,” Leslie T. Cooper Jr., MD, a myocarditis expert and clinical trialist at the Mayo Clinic, Jacksonville, Fla., said in an interview. With the mRNA-based vaccines, “we do not have any conceivable danger signal that would outweigh the benefit of vaccination.”
Males of a certain age
Evidence that such myocarditis predominates in young adult men and adolescent boys, especially following a second vaccine dose, is remarkably consistent.
The risk was elevated only among mRNA-based vaccine recipients who were younger than 40 in the recent Nature Medicine analysis. Among that group, estimates after a second dose numbered fewer than 1 case per 100,000 for Pfizer-BioNTech and 1.5 per 100,000 for Moderna.
In a third analysis from Israel – also in NEJM, from Guy Witberg, MD, Rabin Medical Center, Petah Tikva, and colleagues, based on 2.5 million people aged 16 and older with at least one Pfizer-BioNTech injection – 2.1 cases per 100,000 were estimated overall, but the number rose to 10.7 per 100,000 among those aged 16-29 years.
In Dr. Mevorach’s NEJM report, estimates after a second Pfizer-BioNTech vaccine dose were 1 per 26,000 males versus 1 in 218,000 females, compared with 1 myocarditis case in 10,857 persons among “the general unvaccinated population.”
Most recipients of a first vaccine dose were younger than 50, and 16- to 29-year-olds accounted for most who completed two doses, noted Dr. Mevorach. Younger males bore the brunt of any myocarditis: the estimated prevalence after a second dose among males aged 16-19 was 1 per 6,637, compared with 1 per 99,853 females in the same age range, the group reported.
In the BMJ report, based on about 5 million people 12 years of age or older in Denmark, the estimated rates of myocarditis or pericarditis associated with Moderna immunization were 2 per 100,000 among women but 6.3 per 100,000 for men. The incidence and sex difference was much lower among those getting the Pfizer-BioNTech vaccine: 1.3 per 100,000 and 1.5 per 100,000 in women and men, respectively.
Sex hormones may be key
The predominance of vaccine-associated myocarditis among adolescent and young adult males is probably more about the myocarditis itself than the vaccines, observed Biykem Bozkurt, MD, PhD, who has been studying COVID-related myocarditis at Baylor College of Medicine, Houston.
Male sex historically is associated in both epidemiologic studies and experimental models with a greater propensity for most any form of myocarditis, Dr. Bozkurt said in an interview. Given that males aged 16-19 or so appear to be at highest risk of myocarditis as a complication of SARS-CoV-2 vaccination, the mechanism may well be related to sex hormones.
“Therefore, testosterone is implicated as a player in their higher risk of inflammation and injury and lack of adaptive response in terms of healing, and in terms of prevention of injury,” Dr. Bozkurt said. For its part, estrogen inhibits proinflammatory processes and, in particular, “blunts cell-mediated immune responses.”
“We don’t know the mechanism, but a theory that attributes a protective role to estrogen, or a risk associated with testosterone, is reasonable. It makes sense, at least based on epidemiological data,” Dr. Ammirati agreed. Still, “we do not have any direct evidence in human beings.”
Sex-associated differences in experimental myocarditis have been reported in the journals for at least 70 years, but “the testosterone literature and the estrogen literature have not been evaluated in detail in vaccine-associated myocarditis,” Dr. Cooper said.
Most myocarditis in the laboratory is viral, Dr. Cooper observed, and “the links between testosterone, viruses, and inflammation have been pretty well worked out, I would say, if you’re a mouse. If you’re a human, I think it’s still a bit uncertain.”
Were it to apply in humans, greater testosterone levels might independently promote myocarditis, “and if estrogen is cardioprotective, it would be another mechanism,” Dr. Cooper said. “That would translate to slight male predominance in most kinds of myocarditis.”
In males, compared with females, “the heart can be more vulnerable to events such as arrhythmias or to immune-mediated phenomena. So, probably there is also higher vulnerability to myocarditis in men,” Dr. Ammirati noted.
Male predominance in vaccine-related myocarditis is provocative, so it’s worth considering whether testosterone is part of the mechanism as well as the possibility of estrogen cardioprotection, Dr. Ammirati said. But given limitations of the animal models, “we don’t really have robust data to support any part of that.”
Although myocarditis is in some way immune mediated, “and hormones can modulate the response,” the mechanism has to be more than just sex hormones, he said. “They probably cannot explain the specificity for the heart. It’s not a systemic response, it’s an organ-specific response.”
Modulation of immune responses
Details about the immune processes underlying mRNA-vaccine myocarditis, hormone modulated or not, have been elusive. The complication doesn’t resemble serum sickness, nor does it seem to be a reaction to infection by other cardiotropic viruses, such as coxsackie virus B, a cause of viral myocarditis, Dr. Bozkurt said. The latter had been a compelling possibility because such hypersensitivity to smallpox vaccination is well recognized.
“We don’t know the mechanism, that’s the short answer. But there are many hypotheses,” she said. One candidate widely proposed in the literature: autoantibodies driven by molecular mimicry between the SARS-CoV-2 spike protein targeted by the mRNA vaccines and a structurally similar myocardial protein, possibly alpha-myosin, noted Dr. Bozkurt and colleagues in a recent publication.
But elevations in specific “antiheart antibodies” have not been documented in recipients of the two mRNA-based vaccines, said Dr. Cooper. “So, I would say that – although molecular mimicry is a well-established mechanism of, for example, rheumatic carditis after a streptococcal A infection – that has not been demonstrated yet for COVID-19 mRNA vaccination–related myocarditis.”
“We probably won’t know, ever, with a huge level of certainty, the exact mechanisms,” Dr. Cooper added. There is no animal model for vaccine-induced myocarditis, and “We’re still talking very, very small numbers of patients. The vast majority of them recover,” and so don’t generally provide mechanistic clues.
Prospects for younger children
Vaccination against SARS-CoV-2 has now been authorized by the Centers for Disease Control and Prevention for kids as young as 5-11 years, using the Pfizer-BioNTech vaccine. Experience so far suggests the immunization is safe in that age group with negligible risk of myocarditis or other complications. But with prospects of possible authorization in children younger than 5, should myocarditis be a concern for them?
Probably not, if the complication is driven primarily by sex hormones, Dr. Cooper proposed. “One would predict that before puberty you would have a lower – much, much lower – rate of myocarditis in males than you would in the 16- to 19-year-old range, and that it would be roughly equal to females.” Dr. Ammirati and Dr. Bozkurt largely agreed.
It remains to be seen whether the vaccine-related myocarditis risk applies to children younger than 12, “but I doubt it. I think it’s going to be puberty-related,” Dr. Bozkurt said. Still, “I don’t want to hypothesize without data.”
A version of this article first appeared on Medscape.com.
The risk of myocarditis after immunization with mRNA-based vaccines against SARS-CoV-2 raised concerns when it came to light in early 2021. But as report after report showed such cases to be rare and usually mild and self-limited, focus has turned to the “how and why.”
The mechanism linking the BNT162b2 (Pfizer-BioNTech) and especially mRNA-1273 (Moderna) vaccines to the occurrence of myocarditis is unclear for now, but one potential driver may be tied to a peculiarity that became apparent early: It occurs overwhelmingly in younger males, from 16 to perhaps 40 or 50 years of age. Excess risk has not been consistently seen among women, girls, and older men.
That observation has led to speculation that higher testosterone levels in adolescent boys and young men may somehow promote the adverse vaccine effect, whereas greater levels of estrogen among girls and women in the same age range may be cardioprotective.
Unlikely, brief, and ‘benign’
“Most of the myocarditis is benign, by which I mean that maybe the patients are admitted due to chest pain, but without reduction in ventricular function,” Enrico Ammirati, MD, PhD, a myocarditis expert at De Gasperis Cardio Center and Transplant Center, Niguarda Hospital, Milan, said in an interview.
In a Nov. 14 address on this topic at the annual scientific sessions of the American Heart Association, Dror Mevorach, MD, described the typical case presentation as “mild” and one that clears in fairly short order based on resolution of “clinical symptoms, inflammatory markers and troponin decline, EKG normalization, echo normalization, and a relatively short length of hospital stay.”
Dr. Mevorach, of Hadassah Hebrew University Medical Center, Jerusalem, subsequently published the findings in a report in the New England Journal of Medicine that described 136 confirmed myocarditis cases among more than 5 million people in Israel immunized with the Pfizer-BioNTech vaccine. Myocarditis was considered “mild” in 129 cases, or 95%.
And the risk is tiny, compared with myocarditis from infection by SARS-CoV-2, not to mention the possibility of nasty clinical COVID-19 complications such as pneumonia and pulmonary embolism, Dr. Mevorach observed.
Many other reports agree that the incidence is minimal, especially given the rewards of vaccination. In a separate NEJM publication in September 2021 – from Noam Barda, MD, Clalit (Israel) Research Institute, and colleagues on 1.7 million people in that country, about half unvaccinated and half given the Pfizer-BioNTech vaccine – there were an estimated 2.7 cases of myocarditis per 100,000 vaccinated persons. There were also 11 cases of myocarditis per 100,000 persons who were positive for SARS-CoV-2 infection.
And in a recent case series of vaccinated people aged 16 or older, the myocarditis rate after a first or second Pfizer-BioNTech or Moderna injection was estimated at 1 or fewer per 100,000. The corresponding estimate was 4 such cases per 100,000 after a positive SARS-CoV-2 test among the same population, notes a report published Dec.14, 2021, in Nature Medicine.
In general, “the risk of any kind of cardiac injury is vastly lower with a vaccine than it is with the actual viral infection,” Leslie T. Cooper Jr., MD, a myocarditis expert and clinical trialist at the Mayo Clinic, Jacksonville, Fla., said in an interview. With the mRNA-based vaccines, “we do not have any conceivable danger signal that would outweigh the benefit of vaccination.”
Males of a certain age
Evidence that such myocarditis predominates in young adult men and adolescent boys, especially following a second vaccine dose, is remarkably consistent.
The risk was elevated only among mRNA-based vaccine recipients who were younger than 40 in the recent Nature Medicine analysis. Among that group, estimates after a second dose numbered fewer than 1 case per 100,000 for Pfizer-BioNTech and 1.5 per 100,000 for Moderna.
In a third analysis from Israel – also in NEJM, from Guy Witberg, MD, Rabin Medical Center, Petah Tikva, and colleagues, based on 2.5 million people aged 16 and older with at least one Pfizer-BioNTech injection – 2.1 cases per 100,000 were estimated overall, but the number rose to 10.7 per 100,000 among those aged 16-29 years.
In Dr. Mevorach’s NEJM report, estimates after a second Pfizer-BioNTech vaccine dose were 1 per 26,000 males versus 1 in 218,000 females, compared with 1 myocarditis case in 10,857 persons among “the general unvaccinated population.”
Most recipients of a first vaccine dose were younger than 50, and 16- to 29-year-olds accounted for most who completed two doses, noted Dr. Mevorach. Younger males bore the brunt of any myocarditis: the estimated prevalence after a second dose among males aged 16-19 was 1 per 6,637, compared with 1 per 99,853 females in the same age range, the group reported.
In the BMJ report, based on about 5 million people 12 years of age or older in Denmark, the estimated rates of myocarditis or pericarditis associated with Moderna immunization were 2 per 100,000 among women but 6.3 per 100,000 for men. The incidence and sex difference was much lower among those getting the Pfizer-BioNTech vaccine: 1.3 per 100,000 and 1.5 per 100,000 in women and men, respectively.
Sex hormones may be key
The predominance of vaccine-associated myocarditis among adolescent and young adult males is probably more about the myocarditis itself than the vaccines, observed Biykem Bozkurt, MD, PhD, who has been studying COVID-related myocarditis at Baylor College of Medicine, Houston.
Male sex historically is associated in both epidemiologic studies and experimental models with a greater propensity for most any form of myocarditis, Dr. Bozkurt said in an interview. Given that males aged 16-19 or so appear to be at highest risk of myocarditis as a complication of SARS-CoV-2 vaccination, the mechanism may well be related to sex hormones.
“Therefore, testosterone is implicated as a player in their higher risk of inflammation and injury and lack of adaptive response in terms of healing, and in terms of prevention of injury,” Dr. Bozkurt said. For its part, estrogen inhibits proinflammatory processes and, in particular, “blunts cell-mediated immune responses.”
“We don’t know the mechanism, but a theory that attributes a protective role to estrogen, or a risk associated with testosterone, is reasonable. It makes sense, at least based on epidemiological data,” Dr. Ammirati agreed. Still, “we do not have any direct evidence in human beings.”
Sex-associated differences in experimental myocarditis have been reported in the journals for at least 70 years, but “the testosterone literature and the estrogen literature have not been evaluated in detail in vaccine-associated myocarditis,” Dr. Cooper said.
Most myocarditis in the laboratory is viral, Dr. Cooper observed, and “the links between testosterone, viruses, and inflammation have been pretty well worked out, I would say, if you’re a mouse. If you’re a human, I think it’s still a bit uncertain.”
Were it to apply in humans, greater testosterone levels might independently promote myocarditis, “and if estrogen is cardioprotective, it would be another mechanism,” Dr. Cooper said. “That would translate to slight male predominance in most kinds of myocarditis.”
In males, compared with females, “the heart can be more vulnerable to events such as arrhythmias or to immune-mediated phenomena. So, probably there is also higher vulnerability to myocarditis in men,” Dr. Ammirati noted.
Male predominance in vaccine-related myocarditis is provocative, so it’s worth considering whether testosterone is part of the mechanism as well as the possibility of estrogen cardioprotection, Dr. Ammirati said. But given limitations of the animal models, “we don’t really have robust data to support any part of that.”
Although myocarditis is in some way immune mediated, “and hormones can modulate the response,” the mechanism has to be more than just sex hormones, he said. “They probably cannot explain the specificity for the heart. It’s not a systemic response, it’s an organ-specific response.”
Modulation of immune responses
Details about the immune processes underlying mRNA-vaccine myocarditis, hormone modulated or not, have been elusive. The complication doesn’t resemble serum sickness, nor does it seem to be a reaction to infection by other cardiotropic viruses, such as coxsackie virus B, a cause of viral myocarditis, Dr. Bozkurt said. The latter had been a compelling possibility because such hypersensitivity to smallpox vaccination is well recognized.
“We don’t know the mechanism, that’s the short answer. But there are many hypotheses,” she said. One candidate widely proposed in the literature: autoantibodies driven by molecular mimicry between the SARS-CoV-2 spike protein targeted by the mRNA vaccines and a structurally similar myocardial protein, possibly alpha-myosin, noted Dr. Bozkurt and colleagues in a recent publication.
But elevations in specific “antiheart antibodies” have not been documented in recipients of the two mRNA-based vaccines, said Dr. Cooper. “So, I would say that – although molecular mimicry is a well-established mechanism of, for example, rheumatic carditis after a streptococcal A infection – that has not been demonstrated yet for COVID-19 mRNA vaccination–related myocarditis.”
“We probably won’t know, ever, with a huge level of certainty, the exact mechanisms,” Dr. Cooper added. There is no animal model for vaccine-induced myocarditis, and “We’re still talking very, very small numbers of patients. The vast majority of them recover,” and so don’t generally provide mechanistic clues.
Prospects for younger children
Vaccination against SARS-CoV-2 has now been authorized by the Centers for Disease Control and Prevention for kids as young as 5-11 years, using the Pfizer-BioNTech vaccine. Experience so far suggests the immunization is safe in that age group with negligible risk of myocarditis or other complications. But with prospects of possible authorization in children younger than 5, should myocarditis be a concern for them?
Probably not, if the complication is driven primarily by sex hormones, Dr. Cooper proposed. “One would predict that before puberty you would have a lower – much, much lower – rate of myocarditis in males than you would in the 16- to 19-year-old range, and that it would be roughly equal to females.” Dr. Ammirati and Dr. Bozkurt largely agreed.
It remains to be seen whether the vaccine-related myocarditis risk applies to children younger than 12, “but I doubt it. I think it’s going to be puberty-related,” Dr. Bozkurt said. Still, “I don’t want to hypothesize without data.”
A version of this article first appeared on Medscape.com.
The risk of myocarditis after immunization with mRNA-based vaccines against SARS-CoV-2 raised concerns when it came to light in early 2021. But as report after report showed such cases to be rare and usually mild and self-limited, focus has turned to the “how and why.”
The mechanism linking the BNT162b2 (Pfizer-BioNTech) and especially mRNA-1273 (Moderna) vaccines to the occurrence of myocarditis is unclear for now, but one potential driver may be tied to a peculiarity that became apparent early: It occurs overwhelmingly in younger males, from 16 to perhaps 40 or 50 years of age. Excess risk has not been consistently seen among women, girls, and older men.
That observation has led to speculation that higher testosterone levels in adolescent boys and young men may somehow promote the adverse vaccine effect, whereas greater levels of estrogen among girls and women in the same age range may be cardioprotective.
Unlikely, brief, and ‘benign’
“Most of the myocarditis is benign, by which I mean that maybe the patients are admitted due to chest pain, but without reduction in ventricular function,” Enrico Ammirati, MD, PhD, a myocarditis expert at De Gasperis Cardio Center and Transplant Center, Niguarda Hospital, Milan, said in an interview.
In a Nov. 14 address on this topic at the annual scientific sessions of the American Heart Association, Dror Mevorach, MD, described the typical case presentation as “mild” and one that clears in fairly short order based on resolution of “clinical symptoms, inflammatory markers and troponin decline, EKG normalization, echo normalization, and a relatively short length of hospital stay.”
Dr. Mevorach, of Hadassah Hebrew University Medical Center, Jerusalem, subsequently published the findings in a report in the New England Journal of Medicine that described 136 confirmed myocarditis cases among more than 5 million people in Israel immunized with the Pfizer-BioNTech vaccine. Myocarditis was considered “mild” in 129 cases, or 95%.
And the risk is tiny, compared with myocarditis from infection by SARS-CoV-2, not to mention the possibility of nasty clinical COVID-19 complications such as pneumonia and pulmonary embolism, Dr. Mevorach observed.
Many other reports agree that the incidence is minimal, especially given the rewards of vaccination. In a separate NEJM publication in September 2021 – from Noam Barda, MD, Clalit (Israel) Research Institute, and colleagues on 1.7 million people in that country, about half unvaccinated and half given the Pfizer-BioNTech vaccine – there were an estimated 2.7 cases of myocarditis per 100,000 vaccinated persons. There were also 11 cases of myocarditis per 100,000 persons who were positive for SARS-CoV-2 infection.
And in a recent case series of vaccinated people aged 16 or older, the myocarditis rate after a first or second Pfizer-BioNTech or Moderna injection was estimated at 1 or fewer per 100,000. The corresponding estimate was 4 such cases per 100,000 after a positive SARS-CoV-2 test among the same population, notes a report published Dec.14, 2021, in Nature Medicine.
In general, “the risk of any kind of cardiac injury is vastly lower with a vaccine than it is with the actual viral infection,” Leslie T. Cooper Jr., MD, a myocarditis expert and clinical trialist at the Mayo Clinic, Jacksonville, Fla., said in an interview. With the mRNA-based vaccines, “we do not have any conceivable danger signal that would outweigh the benefit of vaccination.”
Males of a certain age
Evidence that such myocarditis predominates in young adult men and adolescent boys, especially following a second vaccine dose, is remarkably consistent.
The risk was elevated only among mRNA-based vaccine recipients who were younger than 40 in the recent Nature Medicine analysis. Among that group, estimates after a second dose numbered fewer than 1 case per 100,000 for Pfizer-BioNTech and 1.5 per 100,000 for Moderna.
In a third analysis from Israel – also in NEJM, from Guy Witberg, MD, Rabin Medical Center, Petah Tikva, and colleagues, based on 2.5 million people aged 16 and older with at least one Pfizer-BioNTech injection – 2.1 cases per 100,000 were estimated overall, but the number rose to 10.7 per 100,000 among those aged 16-29 years.
In Dr. Mevorach’s NEJM report, estimates after a second Pfizer-BioNTech vaccine dose were 1 per 26,000 males versus 1 in 218,000 females, compared with 1 myocarditis case in 10,857 persons among “the general unvaccinated population.”
Most recipients of a first vaccine dose were younger than 50, and 16- to 29-year-olds accounted for most who completed two doses, noted Dr. Mevorach. Younger males bore the brunt of any myocarditis: the estimated prevalence after a second dose among males aged 16-19 was 1 per 6,637, compared with 1 per 99,853 females in the same age range, the group reported.
In the BMJ report, based on about 5 million people 12 years of age or older in Denmark, the estimated rates of myocarditis or pericarditis associated with Moderna immunization were 2 per 100,000 among women but 6.3 per 100,000 for men. The incidence and sex difference was much lower among those getting the Pfizer-BioNTech vaccine: 1.3 per 100,000 and 1.5 per 100,000 in women and men, respectively.
Sex hormones may be key
The predominance of vaccine-associated myocarditis among adolescent and young adult males is probably more about the myocarditis itself than the vaccines, observed Biykem Bozkurt, MD, PhD, who has been studying COVID-related myocarditis at Baylor College of Medicine, Houston.
Male sex historically is associated in both epidemiologic studies and experimental models with a greater propensity for most any form of myocarditis, Dr. Bozkurt said in an interview. Given that males aged 16-19 or so appear to be at highest risk of myocarditis as a complication of SARS-CoV-2 vaccination, the mechanism may well be related to sex hormones.
“Therefore, testosterone is implicated as a player in their higher risk of inflammation and injury and lack of adaptive response in terms of healing, and in terms of prevention of injury,” Dr. Bozkurt said. For its part, estrogen inhibits proinflammatory processes and, in particular, “blunts cell-mediated immune responses.”
“We don’t know the mechanism, but a theory that attributes a protective role to estrogen, or a risk associated with testosterone, is reasonable. It makes sense, at least based on epidemiological data,” Dr. Ammirati agreed. Still, “we do not have any direct evidence in human beings.”
Sex-associated differences in experimental myocarditis have been reported in the journals for at least 70 years, but “the testosterone literature and the estrogen literature have not been evaluated in detail in vaccine-associated myocarditis,” Dr. Cooper said.
Most myocarditis in the laboratory is viral, Dr. Cooper observed, and “the links between testosterone, viruses, and inflammation have been pretty well worked out, I would say, if you’re a mouse. If you’re a human, I think it’s still a bit uncertain.”
Were it to apply in humans, greater testosterone levels might independently promote myocarditis, “and if estrogen is cardioprotective, it would be another mechanism,” Dr. Cooper said. “That would translate to slight male predominance in most kinds of myocarditis.”
In males, compared with females, “the heart can be more vulnerable to events such as arrhythmias or to immune-mediated phenomena. So, probably there is also higher vulnerability to myocarditis in men,” Dr. Ammirati noted.
Male predominance in vaccine-related myocarditis is provocative, so it’s worth considering whether testosterone is part of the mechanism as well as the possibility of estrogen cardioprotection, Dr. Ammirati said. But given limitations of the animal models, “we don’t really have robust data to support any part of that.”
Although myocarditis is in some way immune mediated, “and hormones can modulate the response,” the mechanism has to be more than just sex hormones, he said. “They probably cannot explain the specificity for the heart. It’s not a systemic response, it’s an organ-specific response.”
Modulation of immune responses
Details about the immune processes underlying mRNA-vaccine myocarditis, hormone modulated or not, have been elusive. The complication doesn’t resemble serum sickness, nor does it seem to be a reaction to infection by other cardiotropic viruses, such as coxsackie virus B, a cause of viral myocarditis, Dr. Bozkurt said. The latter had been a compelling possibility because such hypersensitivity to smallpox vaccination is well recognized.
“We don’t know the mechanism, that’s the short answer. But there are many hypotheses,” she said. One candidate widely proposed in the literature: autoantibodies driven by molecular mimicry between the SARS-CoV-2 spike protein targeted by the mRNA vaccines and a structurally similar myocardial protein, possibly alpha-myosin, noted Dr. Bozkurt and colleagues in a recent publication.
But elevations in specific “antiheart antibodies” have not been documented in recipients of the two mRNA-based vaccines, said Dr. Cooper. “So, I would say that – although molecular mimicry is a well-established mechanism of, for example, rheumatic carditis after a streptococcal A infection – that has not been demonstrated yet for COVID-19 mRNA vaccination–related myocarditis.”
“We probably won’t know, ever, with a huge level of certainty, the exact mechanisms,” Dr. Cooper added. There is no animal model for vaccine-induced myocarditis, and “We’re still talking very, very small numbers of patients. The vast majority of them recover,” and so don’t generally provide mechanistic clues.
Prospects for younger children
Vaccination against SARS-CoV-2 has now been authorized by the Centers for Disease Control and Prevention for kids as young as 5-11 years, using the Pfizer-BioNTech vaccine. Experience so far suggests the immunization is safe in that age group with negligible risk of myocarditis or other complications. But with prospects of possible authorization in children younger than 5, should myocarditis be a concern for them?
Probably not, if the complication is driven primarily by sex hormones, Dr. Cooper proposed. “One would predict that before puberty you would have a lower – much, much lower – rate of myocarditis in males than you would in the 16- to 19-year-old range, and that it would be roughly equal to females.” Dr. Ammirati and Dr. Bozkurt largely agreed.
It remains to be seen whether the vaccine-related myocarditis risk applies to children younger than 12, “but I doubt it. I think it’s going to be puberty-related,” Dr. Bozkurt said. Still, “I don’t want to hypothesize without data.”
A version of this article first appeared on Medscape.com.
AHA advice for diabetes patients to stay heart healthy
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A high-risk medical device didn’t meet federal standards. The government paid millions for more
In 2014, when the Food and Drug Administration found serious problems with a life-sustaining heart pump, its warning letter to the manufacturer threatened to notify other federal health agencies about the inspection’s findings.
But for years, no such alert ever went out. Instead, the agency added the warning letter to an online database alongside thousands of others, following its typical procedures, an FDA spokesperson said.
Agencies such as the Centers for Medicare & Medicaid Services and the U.S. Department of Veterans Affairs went on paying to implant the HeartWare Ventricular Assist Device, or HVAD, in new patients even though federal inspectors had found problems with the device linked to patient deaths and injuries.
Taxpayer dollars continued to flow to the original device maker, HeartWare, and then to the company that acquired it in 2016, Medtronic, for 7 years while the issues raised in the warning letter remained unresolved.
If crucial safety information in FDA warning letters doesn’t make it to other arms of the government responsible for deciding which medical devices to pay for, experts said patients are the ones put at risk.
“It’s clearly a breakdown of communication,” said Dr. Rita Redberg, a cardiologist at the University of California, San Francisco, who researches medical device safety and regulation. “It’s not just the money, obviously. It’s people’s lives.”
The FDA acknowledged that it doesn’t directly notify other agencies when it issues warning letters, pointing instead to its online database, which is accessible to both government officials and the public. “The FDA’s decisions are intended to be patient-centric with the health and safety of device users as our highest priority,” the agency spokesperson said in an email.
The HeartWare letter was removed from the public database about 2 years ago, even though the problems remained unresolved and patients were still receiving implants. The database clears out letters that are more than 5 years old.
CMS, which oversees the Medicare and Medicaid programs, would not say why it continued paying for a device that didn’t meet government standards. It directed questions about the HeartWare warning letter to the FDA. “CMS does not have oversight of the manufacturing and related safety assessments of a medical device manufacturer,” a spokesperson said in an email.
The spokesperson noted that CMS requires heart pump patients to have specialized medical teams managing their care, which should monitor FDA communications regarding safety of devices.
CMS doesn’t track data on devices by manufacturer, so it’s essentially impossible to calculate its total spending on HVADs. One 2018 medical journal study found that Medicare and Medicaid paid for more than half the cost of all heart pump implants from 2009 to 2014. If that rate of spending continued, CMS may have spent more than $400 million on implanting HVADs since 2014.
A spokesperson for the VA said his agency was never notified about the HeartWare warning letter. The VA paid HeartWare and Medtronic more than $3 million after the FDA issued the letter in 2014. It offered this explanation for why: “It’s important to note that FDA Warning Letters are notifications issued to manufacturers found to be in significant violation of federal regulations. They are not product recalls.”
In the case of the HVAD, the FDA’s failure to make sure its warning reached beyond the manufacturer may have had life-and-death consequences.
In August, ProPublica reported that federal inspectors continued finding problems at the HVAD’s manufacturing plant for years. Meanwhile, the FDA received thousands of reports of suspicious deaths and injuries and more than a dozen high-risk safety alerts from the manufacturer.
The documents detailed one horrifying device failure after another. A father of four died after his device suddenly failed and his teenage daughter couldn’t resuscitate him. Another patient’s heart tissue was charred after a pump short-circuited and overheated. A teenager died after vomiting blood as his mother struggled to restart a defective pump.
In June, Medtronic ended sales and implants of the device, citing new data that showed patients with HVADs had a higher rate of deaths and strokes than those with a competing heart pump.
Medtronic declined to comment for this story. It has previously said it believed that after the 2014 warning letter the benefits of the HVAD still outweighed the risks for patients with severe heart failure.
Experts said the lack of communication between federal agencies when serious device problems are found is baffling but not surprising. It fits a broader trend of device regulators focusing more on evaluating new products than monitoring the ones already on the market.
“The priority is to get more medical devices out there, paid for and getting used,” said Dr. Joseph Ross, a professor of medicine and public health at Yale University who studies medical device regulation.
Other U.S. health care regulators move more forcefully when providers and suppliers don’t meet the government’s minimum safety requirements for an extended period, putting patients at risk.
Take hospitals. When inspectors find a facility is not meeting safety standards, CMS can issue an immediate jeopardy citation and, if problems aren’t fixed, move to withhold federal payments, which make up substantial portions of most hospitals’ revenues. In the rare cases when hospitals don’t take sufficient action, CMS follows through and revokes funding.
Redberg, the UCSF cardiologist, said the lack of similar action for medical devices offers a clear “opportunity for improvement.” At minimum, the FDA could establish processes to directly inform other agencies when it issues warning letters and finds serious problems with devices being sold in the United States.
“If the agency’s mission is to protect public health, they would want to do these things and move quickly,” she said.
This story was originally published on ProPublica. ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive their biggest stories as soon as they’re published.
In 2014, when the Food and Drug Administration found serious problems with a life-sustaining heart pump, its warning letter to the manufacturer threatened to notify other federal health agencies about the inspection’s findings.
But for years, no such alert ever went out. Instead, the agency added the warning letter to an online database alongside thousands of others, following its typical procedures, an FDA spokesperson said.
Agencies such as the Centers for Medicare & Medicaid Services and the U.S. Department of Veterans Affairs went on paying to implant the HeartWare Ventricular Assist Device, or HVAD, in new patients even though federal inspectors had found problems with the device linked to patient deaths and injuries.
Taxpayer dollars continued to flow to the original device maker, HeartWare, and then to the company that acquired it in 2016, Medtronic, for 7 years while the issues raised in the warning letter remained unresolved.
If crucial safety information in FDA warning letters doesn’t make it to other arms of the government responsible for deciding which medical devices to pay for, experts said patients are the ones put at risk.
“It’s clearly a breakdown of communication,” said Dr. Rita Redberg, a cardiologist at the University of California, San Francisco, who researches medical device safety and regulation. “It’s not just the money, obviously. It’s people’s lives.”
The FDA acknowledged that it doesn’t directly notify other agencies when it issues warning letters, pointing instead to its online database, which is accessible to both government officials and the public. “The FDA’s decisions are intended to be patient-centric with the health and safety of device users as our highest priority,” the agency spokesperson said in an email.
The HeartWare letter was removed from the public database about 2 years ago, even though the problems remained unresolved and patients were still receiving implants. The database clears out letters that are more than 5 years old.
CMS, which oversees the Medicare and Medicaid programs, would not say why it continued paying for a device that didn’t meet government standards. It directed questions about the HeartWare warning letter to the FDA. “CMS does not have oversight of the manufacturing and related safety assessments of a medical device manufacturer,” a spokesperson said in an email.
The spokesperson noted that CMS requires heart pump patients to have specialized medical teams managing their care, which should monitor FDA communications regarding safety of devices.
CMS doesn’t track data on devices by manufacturer, so it’s essentially impossible to calculate its total spending on HVADs. One 2018 medical journal study found that Medicare and Medicaid paid for more than half the cost of all heart pump implants from 2009 to 2014. If that rate of spending continued, CMS may have spent more than $400 million on implanting HVADs since 2014.
A spokesperson for the VA said his agency was never notified about the HeartWare warning letter. The VA paid HeartWare and Medtronic more than $3 million after the FDA issued the letter in 2014. It offered this explanation for why: “It’s important to note that FDA Warning Letters are notifications issued to manufacturers found to be in significant violation of federal regulations. They are not product recalls.”
In the case of the HVAD, the FDA’s failure to make sure its warning reached beyond the manufacturer may have had life-and-death consequences.
In August, ProPublica reported that federal inspectors continued finding problems at the HVAD’s manufacturing plant for years. Meanwhile, the FDA received thousands of reports of suspicious deaths and injuries and more than a dozen high-risk safety alerts from the manufacturer.
The documents detailed one horrifying device failure after another. A father of four died after his device suddenly failed and his teenage daughter couldn’t resuscitate him. Another patient’s heart tissue was charred after a pump short-circuited and overheated. A teenager died after vomiting blood as his mother struggled to restart a defective pump.
In June, Medtronic ended sales and implants of the device, citing new data that showed patients with HVADs had a higher rate of deaths and strokes than those with a competing heart pump.
Medtronic declined to comment for this story. It has previously said it believed that after the 2014 warning letter the benefits of the HVAD still outweighed the risks for patients with severe heart failure.
Experts said the lack of communication between federal agencies when serious device problems are found is baffling but not surprising. It fits a broader trend of device regulators focusing more on evaluating new products than monitoring the ones already on the market.
“The priority is to get more medical devices out there, paid for and getting used,” said Dr. Joseph Ross, a professor of medicine and public health at Yale University who studies medical device regulation.
Other U.S. health care regulators move more forcefully when providers and suppliers don’t meet the government’s minimum safety requirements for an extended period, putting patients at risk.
Take hospitals. When inspectors find a facility is not meeting safety standards, CMS can issue an immediate jeopardy citation and, if problems aren’t fixed, move to withhold federal payments, which make up substantial portions of most hospitals’ revenues. In the rare cases when hospitals don’t take sufficient action, CMS follows through and revokes funding.
Redberg, the UCSF cardiologist, said the lack of similar action for medical devices offers a clear “opportunity for improvement.” At minimum, the FDA could establish processes to directly inform other agencies when it issues warning letters and finds serious problems with devices being sold in the United States.
“If the agency’s mission is to protect public health, they would want to do these things and move quickly,” she said.
This story was originally published on ProPublica. ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive their biggest stories as soon as they’re published.
In 2014, when the Food and Drug Administration found serious problems with a life-sustaining heart pump, its warning letter to the manufacturer threatened to notify other federal health agencies about the inspection’s findings.
But for years, no such alert ever went out. Instead, the agency added the warning letter to an online database alongside thousands of others, following its typical procedures, an FDA spokesperson said.
Agencies such as the Centers for Medicare & Medicaid Services and the U.S. Department of Veterans Affairs went on paying to implant the HeartWare Ventricular Assist Device, or HVAD, in new patients even though federal inspectors had found problems with the device linked to patient deaths and injuries.
Taxpayer dollars continued to flow to the original device maker, HeartWare, and then to the company that acquired it in 2016, Medtronic, for 7 years while the issues raised in the warning letter remained unresolved.
If crucial safety information in FDA warning letters doesn’t make it to other arms of the government responsible for deciding which medical devices to pay for, experts said patients are the ones put at risk.
“It’s clearly a breakdown of communication,” said Dr. Rita Redberg, a cardiologist at the University of California, San Francisco, who researches medical device safety and regulation. “It’s not just the money, obviously. It’s people’s lives.”
The FDA acknowledged that it doesn’t directly notify other agencies when it issues warning letters, pointing instead to its online database, which is accessible to both government officials and the public. “The FDA’s decisions are intended to be patient-centric with the health and safety of device users as our highest priority,” the agency spokesperson said in an email.
The HeartWare letter was removed from the public database about 2 years ago, even though the problems remained unresolved and patients were still receiving implants. The database clears out letters that are more than 5 years old.
CMS, which oversees the Medicare and Medicaid programs, would not say why it continued paying for a device that didn’t meet government standards. It directed questions about the HeartWare warning letter to the FDA. “CMS does not have oversight of the manufacturing and related safety assessments of a medical device manufacturer,” a spokesperson said in an email.
The spokesperson noted that CMS requires heart pump patients to have specialized medical teams managing their care, which should monitor FDA communications regarding safety of devices.
CMS doesn’t track data on devices by manufacturer, so it’s essentially impossible to calculate its total spending on HVADs. One 2018 medical journal study found that Medicare and Medicaid paid for more than half the cost of all heart pump implants from 2009 to 2014. If that rate of spending continued, CMS may have spent more than $400 million on implanting HVADs since 2014.
A spokesperson for the VA said his agency was never notified about the HeartWare warning letter. The VA paid HeartWare and Medtronic more than $3 million after the FDA issued the letter in 2014. It offered this explanation for why: “It’s important to note that FDA Warning Letters are notifications issued to manufacturers found to be in significant violation of federal regulations. They are not product recalls.”
In the case of the HVAD, the FDA’s failure to make sure its warning reached beyond the manufacturer may have had life-and-death consequences.
In August, ProPublica reported that federal inspectors continued finding problems at the HVAD’s manufacturing plant for years. Meanwhile, the FDA received thousands of reports of suspicious deaths and injuries and more than a dozen high-risk safety alerts from the manufacturer.
The documents detailed one horrifying device failure after another. A father of four died after his device suddenly failed and his teenage daughter couldn’t resuscitate him. Another patient’s heart tissue was charred after a pump short-circuited and overheated. A teenager died after vomiting blood as his mother struggled to restart a defective pump.
In June, Medtronic ended sales and implants of the device, citing new data that showed patients with HVADs had a higher rate of deaths and strokes than those with a competing heart pump.
Medtronic declined to comment for this story. It has previously said it believed that after the 2014 warning letter the benefits of the HVAD still outweighed the risks for patients with severe heart failure.
Experts said the lack of communication between federal agencies when serious device problems are found is baffling but not surprising. It fits a broader trend of device regulators focusing more on evaluating new products than monitoring the ones already on the market.
“The priority is to get more medical devices out there, paid for and getting used,” said Dr. Joseph Ross, a professor of medicine and public health at Yale University who studies medical device regulation.
Other U.S. health care regulators move more forcefully when providers and suppliers don’t meet the government’s minimum safety requirements for an extended period, putting patients at risk.
Take hospitals. When inspectors find a facility is not meeting safety standards, CMS can issue an immediate jeopardy citation and, if problems aren’t fixed, move to withhold federal payments, which make up substantial portions of most hospitals’ revenues. In the rare cases when hospitals don’t take sufficient action, CMS follows through and revokes funding.
Redberg, the UCSF cardiologist, said the lack of similar action for medical devices offers a clear “opportunity for improvement.” At minimum, the FDA could establish processes to directly inform other agencies when it issues warning letters and finds serious problems with devices being sold in the United States.
“If the agency’s mission is to protect public health, they would want to do these things and move quickly,” she said.
This story was originally published on ProPublica. ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive their biggest stories as soon as they’re published.
Bleeding after reperfusion contributes to cardiac injury in MI
The damage to the heart caused by a myocardial infarction is not just a result of ischemia caused by the blocked artery but is also brought about by bleeding in the myocardium after the artery has been opened, a new study suggests.
This observation is leading to new approaches to limiting infarct size and treating MI.
“In MI treatment, we have always focused on opening up the artery as quickly as possible to limit the myocardial damage caused by ischemia,” the study’s senior author, Rohan Dharmakumar, PhD, Indiana University, Indianapolis, told this news organization.
“We are pursuing a completely new approach focusing on limiting the damage after revascularization,” he said. “We are totally rethinking what a myocardial infarction is – what causes the injury and the time course of the injury – our results suggest that it’s not just ischemic damage and a lot of the harm is caused by hemorrhage after reperfusion.”
It has been known for many years that hemorrhage is often seen in the myocardium in large MIs, but it has not been established before now whether it contributes to the injury or not, Dr. Dharmakumar explained.
“This study was done to look at that – and we found that the hemorrhage drives a second layer of injury on top of the ischemia.”
Dr. Dharmakumar said this hemorrhage is part of the phenomenon known as reperfusion injury. “This has been known to exist for many years, but we haven’t fully understood all the factors contributing to it. Our results suggest that hemorrhage is a major component of reperfusion injury – probably the dominant factor,” he said.
The researchers are now working on therapeutic approaches to try to prevent this hemorrhage and/or to minimize its effect.
“We are studying how hemorrhage drives damage and how to block these biological processes,” Dr. Dharmakumar said. “Our studies suggest that hemorrhage could account for up to half of the damage caused by a myocardial infarction. If we can limit that, we should be able to reduce the size of the infarct and this should translate into better long-term outcomes.
“I’m very excited about these results,” he added. “We are already seeing a remarkable improvement in animal models with some of the potential therapeutic approaches we are working on.”
The current study is published in the January 2022 issue of the Journal of the American College of Cardiology (JACC).
The authors explain that it is now recognized that reperfusion injury can contribute to increasing infarct size, which they refer to as “infarct surge.” Previous studies have also shown that reperfusion injury can contribute to as much as 50% of the final infarct size, but the factors contributing to the observed variability are not known, and previous attempts to limit infarct surge from reperfusion injury have failed.
They noted that after reperfusion, microvessels can remain obstructed, resulting in intramyocardial hemorrhage. They conducted the current study to investigate whether such hemorrhage causes expansion of the infarct.
They studied 70 patients with ST-segment elevation MI who were categorized with cardiovascular MRI to have intramyocardial hemorrhage or not following primary PCI, and for whom serial cardiac troponin measures were used to assess infarct size.
Results showed that while troponin levels were not different before reperfusion, patients with intramyocardial hemorrhage had significantly higher cardiac troponin levels after reperfusion and these levels peaked earlier than in patients without hemorrhage.
In animal models, those with intramyocardial hemorrhage had a more rapid expansion of myocardial necrosis than did those without hemorrhage, and within 72 hours of reperfusion, a fourfold greater loss in salvageable myocardium was evident in hemorrhagic MIs.
“We have shown that damage to the heart continues after revascularization as measured by rapidly increasing troponin levels in the hearts that have had a hemorrhage,” Dr. Dharmakumar said.
“Hemorrhage in the myocardium was associated with larger infarctions, and in infarcts causing the same area of myocardium to be at risk, those with hemorrhage after revascularization lost a lot more of the salvageable myocardium than those without hemorrhage,” he added.
Dr. Dharmakumar estimates that such hemorrhage occurs in about half of MIs after revascularization, with risk factors including male gender, anterior wall MIs, and smoking.
He pointed out that previous attempts to treat or prevent reperfusion injury have not been successful, probably because they have not been addressing the key mechanism. “We have not been looking at hemorrhage in this regard until now. This is because it is only recently that we have had the tools to be able to identify hemorrhage in the heart with the use of cardiac MRI.”
Final frontier
In an accompanying editorial, Colin Berry, MBChB, University of Glasgow, and Borja Ibáñez, MD, Jiménez Díaz Foundation University Hospital, Madrid, said they applaud the investigators for providing new, mechanistic insights into a difficult clinical problem that has an unmet therapeutic need.
But they pointed out that it is difficult to completely dissect the impact of hemorrhage versus MI size on adverse remodeling, noting that it might be the case that more severe ischemia/reperfusion events are associated with large MI sizes and higher degree of hemorrhage.
However, they concluded that: “Intramyocardial hemorrhage represents the final frontier for preventing heart failure post-MI. It is readily detected using CMR, and clinical research of novel therapeutic approaches merits prioritization.”
This work was supported by grants from National Institutes of Health/National Heart, Lung, and Blood Institute. Dr. Dharmakumar and coauthor Robert Finney, PhD, have ownership interest in Cardiotheranostics. Dr. Berry is employed by the University of Glasgow, which holds consultancy and research agreements for his work with Abbott Vascular, AstraZeneca, Boehringer Ingelheim, Causeway Therapeutics, Coroventis, Genentech, GlaxoSmithKline, HeartFlow, Menarini, Neovasc, Siemens Healthcare, and Valo Health.
A version of this article first appeared on Medscape.com.
The damage to the heart caused by a myocardial infarction is not just a result of ischemia caused by the blocked artery but is also brought about by bleeding in the myocardium after the artery has been opened, a new study suggests.
This observation is leading to new approaches to limiting infarct size and treating MI.
“In MI treatment, we have always focused on opening up the artery as quickly as possible to limit the myocardial damage caused by ischemia,” the study’s senior author, Rohan Dharmakumar, PhD, Indiana University, Indianapolis, told this news organization.
“We are pursuing a completely new approach focusing on limiting the damage after revascularization,” he said. “We are totally rethinking what a myocardial infarction is – what causes the injury and the time course of the injury – our results suggest that it’s not just ischemic damage and a lot of the harm is caused by hemorrhage after reperfusion.”
It has been known for many years that hemorrhage is often seen in the myocardium in large MIs, but it has not been established before now whether it contributes to the injury or not, Dr. Dharmakumar explained.
“This study was done to look at that – and we found that the hemorrhage drives a second layer of injury on top of the ischemia.”
Dr. Dharmakumar said this hemorrhage is part of the phenomenon known as reperfusion injury. “This has been known to exist for many years, but we haven’t fully understood all the factors contributing to it. Our results suggest that hemorrhage is a major component of reperfusion injury – probably the dominant factor,” he said.
The researchers are now working on therapeutic approaches to try to prevent this hemorrhage and/or to minimize its effect.
“We are studying how hemorrhage drives damage and how to block these biological processes,” Dr. Dharmakumar said. “Our studies suggest that hemorrhage could account for up to half of the damage caused by a myocardial infarction. If we can limit that, we should be able to reduce the size of the infarct and this should translate into better long-term outcomes.
“I’m very excited about these results,” he added. “We are already seeing a remarkable improvement in animal models with some of the potential therapeutic approaches we are working on.”
The current study is published in the January 2022 issue of the Journal of the American College of Cardiology (JACC).
The authors explain that it is now recognized that reperfusion injury can contribute to increasing infarct size, which they refer to as “infarct surge.” Previous studies have also shown that reperfusion injury can contribute to as much as 50% of the final infarct size, but the factors contributing to the observed variability are not known, and previous attempts to limit infarct surge from reperfusion injury have failed.
They noted that after reperfusion, microvessels can remain obstructed, resulting in intramyocardial hemorrhage. They conducted the current study to investigate whether such hemorrhage causes expansion of the infarct.
They studied 70 patients with ST-segment elevation MI who were categorized with cardiovascular MRI to have intramyocardial hemorrhage or not following primary PCI, and for whom serial cardiac troponin measures were used to assess infarct size.
Results showed that while troponin levels were not different before reperfusion, patients with intramyocardial hemorrhage had significantly higher cardiac troponin levels after reperfusion and these levels peaked earlier than in patients without hemorrhage.
In animal models, those with intramyocardial hemorrhage had a more rapid expansion of myocardial necrosis than did those without hemorrhage, and within 72 hours of reperfusion, a fourfold greater loss in salvageable myocardium was evident in hemorrhagic MIs.
“We have shown that damage to the heart continues after revascularization as measured by rapidly increasing troponin levels in the hearts that have had a hemorrhage,” Dr. Dharmakumar said.
“Hemorrhage in the myocardium was associated with larger infarctions, and in infarcts causing the same area of myocardium to be at risk, those with hemorrhage after revascularization lost a lot more of the salvageable myocardium than those without hemorrhage,” he added.
Dr. Dharmakumar estimates that such hemorrhage occurs in about half of MIs after revascularization, with risk factors including male gender, anterior wall MIs, and smoking.
He pointed out that previous attempts to treat or prevent reperfusion injury have not been successful, probably because they have not been addressing the key mechanism. “We have not been looking at hemorrhage in this regard until now. This is because it is only recently that we have had the tools to be able to identify hemorrhage in the heart with the use of cardiac MRI.”
Final frontier
In an accompanying editorial, Colin Berry, MBChB, University of Glasgow, and Borja Ibáñez, MD, Jiménez Díaz Foundation University Hospital, Madrid, said they applaud the investigators for providing new, mechanistic insights into a difficult clinical problem that has an unmet therapeutic need.
But they pointed out that it is difficult to completely dissect the impact of hemorrhage versus MI size on adverse remodeling, noting that it might be the case that more severe ischemia/reperfusion events are associated with large MI sizes and higher degree of hemorrhage.
However, they concluded that: “Intramyocardial hemorrhage represents the final frontier for preventing heart failure post-MI. It is readily detected using CMR, and clinical research of novel therapeutic approaches merits prioritization.”
This work was supported by grants from National Institutes of Health/National Heart, Lung, and Blood Institute. Dr. Dharmakumar and coauthor Robert Finney, PhD, have ownership interest in Cardiotheranostics. Dr. Berry is employed by the University of Glasgow, which holds consultancy and research agreements for his work with Abbott Vascular, AstraZeneca, Boehringer Ingelheim, Causeway Therapeutics, Coroventis, Genentech, GlaxoSmithKline, HeartFlow, Menarini, Neovasc, Siemens Healthcare, and Valo Health.
A version of this article first appeared on Medscape.com.
The damage to the heart caused by a myocardial infarction is not just a result of ischemia caused by the blocked artery but is also brought about by bleeding in the myocardium after the artery has been opened, a new study suggests.
This observation is leading to new approaches to limiting infarct size and treating MI.
“In MI treatment, we have always focused on opening up the artery as quickly as possible to limit the myocardial damage caused by ischemia,” the study’s senior author, Rohan Dharmakumar, PhD, Indiana University, Indianapolis, told this news organization.
“We are pursuing a completely new approach focusing on limiting the damage after revascularization,” he said. “We are totally rethinking what a myocardial infarction is – what causes the injury and the time course of the injury – our results suggest that it’s not just ischemic damage and a lot of the harm is caused by hemorrhage after reperfusion.”
It has been known for many years that hemorrhage is often seen in the myocardium in large MIs, but it has not been established before now whether it contributes to the injury or not, Dr. Dharmakumar explained.
“This study was done to look at that – and we found that the hemorrhage drives a second layer of injury on top of the ischemia.”
Dr. Dharmakumar said this hemorrhage is part of the phenomenon known as reperfusion injury. “This has been known to exist for many years, but we haven’t fully understood all the factors contributing to it. Our results suggest that hemorrhage is a major component of reperfusion injury – probably the dominant factor,” he said.
The researchers are now working on therapeutic approaches to try to prevent this hemorrhage and/or to minimize its effect.
“We are studying how hemorrhage drives damage and how to block these biological processes,” Dr. Dharmakumar said. “Our studies suggest that hemorrhage could account for up to half of the damage caused by a myocardial infarction. If we can limit that, we should be able to reduce the size of the infarct and this should translate into better long-term outcomes.
“I’m very excited about these results,” he added. “We are already seeing a remarkable improvement in animal models with some of the potential therapeutic approaches we are working on.”
The current study is published in the January 2022 issue of the Journal of the American College of Cardiology (JACC).
The authors explain that it is now recognized that reperfusion injury can contribute to increasing infarct size, which they refer to as “infarct surge.” Previous studies have also shown that reperfusion injury can contribute to as much as 50% of the final infarct size, but the factors contributing to the observed variability are not known, and previous attempts to limit infarct surge from reperfusion injury have failed.
They noted that after reperfusion, microvessels can remain obstructed, resulting in intramyocardial hemorrhage. They conducted the current study to investigate whether such hemorrhage causes expansion of the infarct.
They studied 70 patients with ST-segment elevation MI who were categorized with cardiovascular MRI to have intramyocardial hemorrhage or not following primary PCI, and for whom serial cardiac troponin measures were used to assess infarct size.
Results showed that while troponin levels were not different before reperfusion, patients with intramyocardial hemorrhage had significantly higher cardiac troponin levels after reperfusion and these levels peaked earlier than in patients without hemorrhage.
In animal models, those with intramyocardial hemorrhage had a more rapid expansion of myocardial necrosis than did those without hemorrhage, and within 72 hours of reperfusion, a fourfold greater loss in salvageable myocardium was evident in hemorrhagic MIs.
“We have shown that damage to the heart continues after revascularization as measured by rapidly increasing troponin levels in the hearts that have had a hemorrhage,” Dr. Dharmakumar said.
“Hemorrhage in the myocardium was associated with larger infarctions, and in infarcts causing the same area of myocardium to be at risk, those with hemorrhage after revascularization lost a lot more of the salvageable myocardium than those without hemorrhage,” he added.
Dr. Dharmakumar estimates that such hemorrhage occurs in about half of MIs after revascularization, with risk factors including male gender, anterior wall MIs, and smoking.
He pointed out that previous attempts to treat or prevent reperfusion injury have not been successful, probably because they have not been addressing the key mechanism. “We have not been looking at hemorrhage in this regard until now. This is because it is only recently that we have had the tools to be able to identify hemorrhage in the heart with the use of cardiac MRI.”
Final frontier
In an accompanying editorial, Colin Berry, MBChB, University of Glasgow, and Borja Ibáñez, MD, Jiménez Díaz Foundation University Hospital, Madrid, said they applaud the investigators for providing new, mechanistic insights into a difficult clinical problem that has an unmet therapeutic need.
But they pointed out that it is difficult to completely dissect the impact of hemorrhage versus MI size on adverse remodeling, noting that it might be the case that more severe ischemia/reperfusion events are associated with large MI sizes and higher degree of hemorrhage.
However, they concluded that: “Intramyocardial hemorrhage represents the final frontier for preventing heart failure post-MI. It is readily detected using CMR, and clinical research of novel therapeutic approaches merits prioritization.”
This work was supported by grants from National Institutes of Health/National Heart, Lung, and Blood Institute. Dr. Dharmakumar and coauthor Robert Finney, PhD, have ownership interest in Cardiotheranostics. Dr. Berry is employed by the University of Glasgow, which holds consultancy and research agreements for his work with Abbott Vascular, AstraZeneca, Boehringer Ingelheim, Causeway Therapeutics, Coroventis, Genentech, GlaxoSmithKline, HeartFlow, Menarini, Neovasc, Siemens Healthcare, and Valo Health.
A version of this article first appeared on Medscape.com.