User login
DISCHARGE: CTA shows safety edge versus cath in intermediate-risk stable chest pain
Clinical outcomes over several years in the randomized trial – called DISCHARGE, with an enrollment of more than 3,500 – were statistically similar whether the patients were assigned to CTA or invasive coronary angiography (ICA) as their initial evaluation. Symptoms and quality-of-life measures were also similar.
But the patients assigned to the initial-CTA strategy, of whom fewer than a fourth went on to cardiac cath, showed far fewer procedure-related complications and less often went to coronary revascularization during the median follow-up of 3.5 years, the group reported March 4 in the New England Journal of Medicine.
Based on the findings, CTA “is a safe alternative to cardiac catheterization for patients with suspected CAD [coronary artery disease] that will likely change clinical practice worldwide by replacing invasive testing in patients with stable chest pain who can be expected to benefit” those with an intermediate pretest probability for obstructive disease, principal investigator Marc Dewey, MD, Charité – Universitätsmedizin Berlin, told this news organization.
None of the patient subgroups explored in the trial showed a significant clinical benefit from one strategy over the other, Dr. Dewey commented in an email.
The trial’s results don’t apply to patients unlike those entered, and in particular, he said, “ICA should remain the first test option in patients with high clinical pretest probability of obstructive CAD.”
Dr. Dewey is senior author on the study’s publication, which was timed to coincide with his presentation of the results at ECR 2022 Overture, an all-virtual scientific session of the European Congress of Radiology.
“This is the definitive study,” Matthew Budoff, MD, Lundquist Institute at Harbor-UCLA, Torrance, California, said in an interview. It suggests in a large population that the initial CTA strategy “is as good and maybe safer” in stable patients at intermediate risk compared with initial ICA. “I would say close to 75% or 80% of the patients that we see would fall into that range of risk” and be suitable for the testing algorithm used in the study, said Dr. Budoff, who was not part of the trial.
Invasive angiography would generally still be the initial approach for patients at greater than intermediate risk, such as those with breakthrough angina or electrocardiographic changes, he said. “I still think there’s a huge role for invasive angiography. It’s just a bit smaller now than it used to be for the lower-risk patient.”
The DISCHARGE trial, agreed cardiothoracic radiology specialist Rozemarijn Vliegenthart, MD, PhD, University of Groningen, the Netherlands, “shows that in patients with intermediate pretest probability, CTA should be used as a gatekeeper before invasive coronary angiography, instead of directly referring for invasive coronary angiography.”
It shows that “a CT-first approach” is both safe and clinically effective and even a trend suggesting better clinical outcomes, compared with ICA. And it demonstrates that “still, many diagnostic invasive coronary angiographies are performed unnecessarily,” Dr. Vliegenthart said as the invited discussant following Dr. Dewey’s presentation.
DISCOVER is only the latest in a series of major studies to explore how CTA best fits in with ICA, stress imaging, and other tests for evaluating patients with chest pain. For example, “the PROMISE trial and the SCOT-HEART trial found that CT was as good as or even better than functional testing. DISCHARGE, I think, confirms the safety of the CT strategy” and reaffirms that it is “at least as good” as an ICA-first approach, cardiologist Klaus F. Kofoed, MD, PhD, DMSc, Rigshospitalet University of Copenhagen, said when co-presenting the trial’s results with Dr. Dewey.
“We can now say CT may be suitable in intermediate-risk patients referred for ICA, particularly those with a clinical constellation suggesting a higher event risk, with abnormal or inconclusive functional test results, or with persistent symptoms despite medical treatment,” said Dr. Kofoed, who is on the DISCOVER steering committee.
The trial’s 3,561 patients with stable chest pain – at 26 experienced centers in 16 countries – were randomly assigned to undergo CTA or ICA as their initial diagnostic imaging approach. Entry required them to be at intermediate risk, defined as an estimated 10% to 60% probability of having obstructive CAD. Of note, women made up about 56% of both groups.
Imaging was positive for obstructive disease in 26% of the 1,808 patients in the CTA group and in the same proportion of the 1,753 who were assigned to ICA. Nonobstructive CAD was identified in 36% and 22%, respectively.
Importantly, 404 (22.3%) patients in the CTA group then underwent ICA, which identified obstructive CAD in 293 (72.5%).
With a complete follow-up in about 99% of patients, the report notes, the rate of the primary endpoint of major adverse cardiac events, or MACE (cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) was 2.1% in the CTA group and in 3.0% in the ICA group. The adjusted hazard ratio of 0.70 (95% confidence interval, 0.46-1.07; P = .10) fell short of significance.
The corresponding HR for an “expanded primary outcome” that also included transient ischemic attack or major procedure-related complications was 0.60 (95% CI, 0.42-0.85) in favor of the CTA group.
As a “pragmatic trial,” DISCHARGE relied on clinically identified events for the endpoint assessments and did not require, for example, laboratory biomarker or neurologic imaging for confirmation, the report notes.
Major procedure-related complications during the initial management phase occurred in 0.5% of the CTA group, and 1.9% of those assigned to initial ICA (HR, 0.26; 95% CI, 0.13-0.55).
Coronary revascularization was less common in the CTA group during the trial’s follow-up, 14.2% versus 18.0% for those assigned to ICA (HR, 0.76; 95% CI, 0.65-0.90).
But the prevalences of angina during the final 4 weeks of follow-up, the group reported, were statistically similar at 8.8% and 7.5% for patients assigned to CTA and ICA, respectively.
The trial showed “no material difference” between the initial CTA versus ICA strategies for its MACE primary endpoint, observed Joseph Loscalzo, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School, Boston, Mass., in an accompanying editorial.
“This result is probably a consequence of the lack of effect of revascularization on cardiovascular events among most patients with stable angina and the limited number of those with high-risk anatomy who would benefit from revascularization in the trial,” he writes.
That CTA was performed “significantly earlier than angiography, 3 days versus 12 days after enrollment,” may have led to earlier coronary revascularization in that group, and therefore is “a better outcome in patients whose anatomy would benefit from it.”
Dr. Loscalzo questioned several aspects of the trial design, which, for example, led to a more than 35% prevalence of patients with nonanginal chest pain among those randomized. Different criteria for classifying patients as “intermediate risk” might also have contributed to the fairly low prevalence of patients in either group ultimately identified with obstructive CAD, he proposes. That low prevalence “suggests that the overall trial population had a low risk of obstructive CAD rather than an intermediate risk.”
DISCHARGE was supported by grants from the European Union Seventh Framework Program, the Berlin Institute of Health, Rigshospitalet of the University of Copenhagen, the British Heart Foundation, and the German Research Foundation. Disclosures for the authors and editorialist are available at NEJM.org. Dr. Budoff has disclosed receiving grant support from General Electric. Dr. Vliegenthart discloses receiving grants from Siemens Healthineers and honorarium for speaking from Siemens Healthineers and Bayer.
A version of this article first appeared on Medscape.com.
Clinical outcomes over several years in the randomized trial – called DISCHARGE, with an enrollment of more than 3,500 – were statistically similar whether the patients were assigned to CTA or invasive coronary angiography (ICA) as their initial evaluation. Symptoms and quality-of-life measures were also similar.
But the patients assigned to the initial-CTA strategy, of whom fewer than a fourth went on to cardiac cath, showed far fewer procedure-related complications and less often went to coronary revascularization during the median follow-up of 3.5 years, the group reported March 4 in the New England Journal of Medicine.
Based on the findings, CTA “is a safe alternative to cardiac catheterization for patients with suspected CAD [coronary artery disease] that will likely change clinical practice worldwide by replacing invasive testing in patients with stable chest pain who can be expected to benefit” those with an intermediate pretest probability for obstructive disease, principal investigator Marc Dewey, MD, Charité – Universitätsmedizin Berlin, told this news organization.
None of the patient subgroups explored in the trial showed a significant clinical benefit from one strategy over the other, Dr. Dewey commented in an email.
The trial’s results don’t apply to patients unlike those entered, and in particular, he said, “ICA should remain the first test option in patients with high clinical pretest probability of obstructive CAD.”
Dr. Dewey is senior author on the study’s publication, which was timed to coincide with his presentation of the results at ECR 2022 Overture, an all-virtual scientific session of the European Congress of Radiology.
“This is the definitive study,” Matthew Budoff, MD, Lundquist Institute at Harbor-UCLA, Torrance, California, said in an interview. It suggests in a large population that the initial CTA strategy “is as good and maybe safer” in stable patients at intermediate risk compared with initial ICA. “I would say close to 75% or 80% of the patients that we see would fall into that range of risk” and be suitable for the testing algorithm used in the study, said Dr. Budoff, who was not part of the trial.
Invasive angiography would generally still be the initial approach for patients at greater than intermediate risk, such as those with breakthrough angina or electrocardiographic changes, he said. “I still think there’s a huge role for invasive angiography. It’s just a bit smaller now than it used to be for the lower-risk patient.”
The DISCHARGE trial, agreed cardiothoracic radiology specialist Rozemarijn Vliegenthart, MD, PhD, University of Groningen, the Netherlands, “shows that in patients with intermediate pretest probability, CTA should be used as a gatekeeper before invasive coronary angiography, instead of directly referring for invasive coronary angiography.”
It shows that “a CT-first approach” is both safe and clinically effective and even a trend suggesting better clinical outcomes, compared with ICA. And it demonstrates that “still, many diagnostic invasive coronary angiographies are performed unnecessarily,” Dr. Vliegenthart said as the invited discussant following Dr. Dewey’s presentation.
DISCOVER is only the latest in a series of major studies to explore how CTA best fits in with ICA, stress imaging, and other tests for evaluating patients with chest pain. For example, “the PROMISE trial and the SCOT-HEART trial found that CT was as good as or even better than functional testing. DISCHARGE, I think, confirms the safety of the CT strategy” and reaffirms that it is “at least as good” as an ICA-first approach, cardiologist Klaus F. Kofoed, MD, PhD, DMSc, Rigshospitalet University of Copenhagen, said when co-presenting the trial’s results with Dr. Dewey.
“We can now say CT may be suitable in intermediate-risk patients referred for ICA, particularly those with a clinical constellation suggesting a higher event risk, with abnormal or inconclusive functional test results, or with persistent symptoms despite medical treatment,” said Dr. Kofoed, who is on the DISCOVER steering committee.
The trial’s 3,561 patients with stable chest pain – at 26 experienced centers in 16 countries – were randomly assigned to undergo CTA or ICA as their initial diagnostic imaging approach. Entry required them to be at intermediate risk, defined as an estimated 10% to 60% probability of having obstructive CAD. Of note, women made up about 56% of both groups.
Imaging was positive for obstructive disease in 26% of the 1,808 patients in the CTA group and in the same proportion of the 1,753 who were assigned to ICA. Nonobstructive CAD was identified in 36% and 22%, respectively.
Importantly, 404 (22.3%) patients in the CTA group then underwent ICA, which identified obstructive CAD in 293 (72.5%).
With a complete follow-up in about 99% of patients, the report notes, the rate of the primary endpoint of major adverse cardiac events, or MACE (cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) was 2.1% in the CTA group and in 3.0% in the ICA group. The adjusted hazard ratio of 0.70 (95% confidence interval, 0.46-1.07; P = .10) fell short of significance.
The corresponding HR for an “expanded primary outcome” that also included transient ischemic attack or major procedure-related complications was 0.60 (95% CI, 0.42-0.85) in favor of the CTA group.
As a “pragmatic trial,” DISCHARGE relied on clinically identified events for the endpoint assessments and did not require, for example, laboratory biomarker or neurologic imaging for confirmation, the report notes.
Major procedure-related complications during the initial management phase occurred in 0.5% of the CTA group, and 1.9% of those assigned to initial ICA (HR, 0.26; 95% CI, 0.13-0.55).
Coronary revascularization was less common in the CTA group during the trial’s follow-up, 14.2% versus 18.0% for those assigned to ICA (HR, 0.76; 95% CI, 0.65-0.90).
But the prevalences of angina during the final 4 weeks of follow-up, the group reported, were statistically similar at 8.8% and 7.5% for patients assigned to CTA and ICA, respectively.
The trial showed “no material difference” between the initial CTA versus ICA strategies for its MACE primary endpoint, observed Joseph Loscalzo, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School, Boston, Mass., in an accompanying editorial.
“This result is probably a consequence of the lack of effect of revascularization on cardiovascular events among most patients with stable angina and the limited number of those with high-risk anatomy who would benefit from revascularization in the trial,” he writes.
That CTA was performed “significantly earlier than angiography, 3 days versus 12 days after enrollment,” may have led to earlier coronary revascularization in that group, and therefore is “a better outcome in patients whose anatomy would benefit from it.”
Dr. Loscalzo questioned several aspects of the trial design, which, for example, led to a more than 35% prevalence of patients with nonanginal chest pain among those randomized. Different criteria for classifying patients as “intermediate risk” might also have contributed to the fairly low prevalence of patients in either group ultimately identified with obstructive CAD, he proposes. That low prevalence “suggests that the overall trial population had a low risk of obstructive CAD rather than an intermediate risk.”
DISCHARGE was supported by grants from the European Union Seventh Framework Program, the Berlin Institute of Health, Rigshospitalet of the University of Copenhagen, the British Heart Foundation, and the German Research Foundation. Disclosures for the authors and editorialist are available at NEJM.org. Dr. Budoff has disclosed receiving grant support from General Electric. Dr. Vliegenthart discloses receiving grants from Siemens Healthineers and honorarium for speaking from Siemens Healthineers and Bayer.
A version of this article first appeared on Medscape.com.
Clinical outcomes over several years in the randomized trial – called DISCHARGE, with an enrollment of more than 3,500 – were statistically similar whether the patients were assigned to CTA or invasive coronary angiography (ICA) as their initial evaluation. Symptoms and quality-of-life measures were also similar.
But the patients assigned to the initial-CTA strategy, of whom fewer than a fourth went on to cardiac cath, showed far fewer procedure-related complications and less often went to coronary revascularization during the median follow-up of 3.5 years, the group reported March 4 in the New England Journal of Medicine.
Based on the findings, CTA “is a safe alternative to cardiac catheterization for patients with suspected CAD [coronary artery disease] that will likely change clinical practice worldwide by replacing invasive testing in patients with stable chest pain who can be expected to benefit” those with an intermediate pretest probability for obstructive disease, principal investigator Marc Dewey, MD, Charité – Universitätsmedizin Berlin, told this news organization.
None of the patient subgroups explored in the trial showed a significant clinical benefit from one strategy over the other, Dr. Dewey commented in an email.
The trial’s results don’t apply to patients unlike those entered, and in particular, he said, “ICA should remain the first test option in patients with high clinical pretest probability of obstructive CAD.”
Dr. Dewey is senior author on the study’s publication, which was timed to coincide with his presentation of the results at ECR 2022 Overture, an all-virtual scientific session of the European Congress of Radiology.
“This is the definitive study,” Matthew Budoff, MD, Lundquist Institute at Harbor-UCLA, Torrance, California, said in an interview. It suggests in a large population that the initial CTA strategy “is as good and maybe safer” in stable patients at intermediate risk compared with initial ICA. “I would say close to 75% or 80% of the patients that we see would fall into that range of risk” and be suitable for the testing algorithm used in the study, said Dr. Budoff, who was not part of the trial.
Invasive angiography would generally still be the initial approach for patients at greater than intermediate risk, such as those with breakthrough angina or electrocardiographic changes, he said. “I still think there’s a huge role for invasive angiography. It’s just a bit smaller now than it used to be for the lower-risk patient.”
The DISCHARGE trial, agreed cardiothoracic radiology specialist Rozemarijn Vliegenthart, MD, PhD, University of Groningen, the Netherlands, “shows that in patients with intermediate pretest probability, CTA should be used as a gatekeeper before invasive coronary angiography, instead of directly referring for invasive coronary angiography.”
It shows that “a CT-first approach” is both safe and clinically effective and even a trend suggesting better clinical outcomes, compared with ICA. And it demonstrates that “still, many diagnostic invasive coronary angiographies are performed unnecessarily,” Dr. Vliegenthart said as the invited discussant following Dr. Dewey’s presentation.
DISCOVER is only the latest in a series of major studies to explore how CTA best fits in with ICA, stress imaging, and other tests for evaluating patients with chest pain. For example, “the PROMISE trial and the SCOT-HEART trial found that CT was as good as or even better than functional testing. DISCHARGE, I think, confirms the safety of the CT strategy” and reaffirms that it is “at least as good” as an ICA-first approach, cardiologist Klaus F. Kofoed, MD, PhD, DMSc, Rigshospitalet University of Copenhagen, said when co-presenting the trial’s results with Dr. Dewey.
“We can now say CT may be suitable in intermediate-risk patients referred for ICA, particularly those with a clinical constellation suggesting a higher event risk, with abnormal or inconclusive functional test results, or with persistent symptoms despite medical treatment,” said Dr. Kofoed, who is on the DISCOVER steering committee.
The trial’s 3,561 patients with stable chest pain – at 26 experienced centers in 16 countries – were randomly assigned to undergo CTA or ICA as their initial diagnostic imaging approach. Entry required them to be at intermediate risk, defined as an estimated 10% to 60% probability of having obstructive CAD. Of note, women made up about 56% of both groups.
Imaging was positive for obstructive disease in 26% of the 1,808 patients in the CTA group and in the same proportion of the 1,753 who were assigned to ICA. Nonobstructive CAD was identified in 36% and 22%, respectively.
Importantly, 404 (22.3%) patients in the CTA group then underwent ICA, which identified obstructive CAD in 293 (72.5%).
With a complete follow-up in about 99% of patients, the report notes, the rate of the primary endpoint of major adverse cardiac events, or MACE (cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) was 2.1% in the CTA group and in 3.0% in the ICA group. The adjusted hazard ratio of 0.70 (95% confidence interval, 0.46-1.07; P = .10) fell short of significance.
The corresponding HR for an “expanded primary outcome” that also included transient ischemic attack or major procedure-related complications was 0.60 (95% CI, 0.42-0.85) in favor of the CTA group.
As a “pragmatic trial,” DISCHARGE relied on clinically identified events for the endpoint assessments and did not require, for example, laboratory biomarker or neurologic imaging for confirmation, the report notes.
Major procedure-related complications during the initial management phase occurred in 0.5% of the CTA group, and 1.9% of those assigned to initial ICA (HR, 0.26; 95% CI, 0.13-0.55).
Coronary revascularization was less common in the CTA group during the trial’s follow-up, 14.2% versus 18.0% for those assigned to ICA (HR, 0.76; 95% CI, 0.65-0.90).
But the prevalences of angina during the final 4 weeks of follow-up, the group reported, were statistically similar at 8.8% and 7.5% for patients assigned to CTA and ICA, respectively.
The trial showed “no material difference” between the initial CTA versus ICA strategies for its MACE primary endpoint, observed Joseph Loscalzo, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School, Boston, Mass., in an accompanying editorial.
“This result is probably a consequence of the lack of effect of revascularization on cardiovascular events among most patients with stable angina and the limited number of those with high-risk anatomy who would benefit from revascularization in the trial,” he writes.
That CTA was performed “significantly earlier than angiography, 3 days versus 12 days after enrollment,” may have led to earlier coronary revascularization in that group, and therefore is “a better outcome in patients whose anatomy would benefit from it.”
Dr. Loscalzo questioned several aspects of the trial design, which, for example, led to a more than 35% prevalence of patients with nonanginal chest pain among those randomized. Different criteria for classifying patients as “intermediate risk” might also have contributed to the fairly low prevalence of patients in either group ultimately identified with obstructive CAD, he proposes. That low prevalence “suggests that the overall trial population had a low risk of obstructive CAD rather than an intermediate risk.”
DISCHARGE was supported by grants from the European Union Seventh Framework Program, the Berlin Institute of Health, Rigshospitalet of the University of Copenhagen, the British Heart Foundation, and the German Research Foundation. Disclosures for the authors and editorialist are available at NEJM.org. Dr. Budoff has disclosed receiving grant support from General Electric. Dr. Vliegenthart discloses receiving grants from Siemens Healthineers and honorarium for speaking from Siemens Healthineers and Bayer.
A version of this article first appeared on Medscape.com.
A-fib prevention, treatment, and screening: Where does the evidence lead us?
Atrial fibrillation (AF) is a common problem confronting family physicians. In this issue of JFP, we offer 2 articles about AF: one on prevention and one on treatment. Both provide evidence-based guidance to help you refine your care. But gaps remain. I’ll get to that in a bit.
Prevention. This month’s PURL1 discusses a randomized controlled trial (RCT) that enrolled moderate alcohol drinkers with AF.2 Compared to those who continued to drink moderately, those who reduced their alcohol consumption to 2 drinks per week had a significant reduction in recurrent AF (73% vs 53%), fewer hospitalizations (20% vs 9%), and less moderate or severe symptoms (32% vs 10%). Although previous studies of moderate alcohol consumption have shown positive effects on heart disease, this study and other more recent studies cast serious doubt on this assertion.3
Treatment. In their applied evidence article, Osayande and Sharma4 pose the question: When is catheter ablation a sound option for your patient with AF? They give us an excellent, evidence-based answer and remind us that we must focus on the treatment goals: to prevent stroke and to control symptoms. They recommend a stepwise approach, starting with rate control, progressing to rhythm control, and saving catheter ablation for resistant cases. In nearly all cases, anticoagulation to prevent stroke must be a part of treatment, with the exception of those with very low risk (so-called “lone atrial fibrillation”).
Screening. And what about screening for asymptomatic AF? The US Preventive Services Task Force recently reaffirmed its conclusion that there is insufficient evidence for screening for asymptomatic AF (a topic discussed in an online Practice Alert Brief5).6 Since wearable exercise-monitoring devices can detect heart arrhythmias (and are advertised for this purpose), a patient may present after receiving a notification about asymptomatic AF. What shall we do in these cases? The dilemma is that your patient will know she has a potentially dangerous condition, but there is no evidence that treating it will result in more benefit than harm.
A recently published study suggests that we should be very cautious in recommending treatment. In an RCT of patients ages 70 to 90 years, 1501 patients received an implantable loop recorder, while 4503 received routine health care; median follow-up was 64.5 months.7 Although more cases of AF were detected (32% in the monitored group vs 12% in the usual care group), and oral anticoagulation treatment was started more frequently (30% vs 13%, respectively), there was no significant difference in the proportion of patients who had a stroke or systemic arterial embolism (4.5% vs 5.6%).7 Until we have more data, reassurance seems to be the best recommendation for asymptomatic AF.
1. Thiel DJ, Marshall RC, Rogers TS. Alcohol abstinence reduces A-fib burden in drinkers. J Fam Pract. 2022;71:85-87.
2. Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28. doi: 10.1056/NEJMoa1817591
3. Hoek AG, van Oort S, Mukamal KJ, et al. Alcohol consumption and cardiovascular disease risk: placing new data in context [published online ahead of print, 2022 Feb 7]. Curr Atheroscler Rep. doi: 10.1007/s11883-022-00992-1
4. Osayande AS, Sharma N. When is catheter ablation a sound option for your patient with A-fib? J Fam Pract. 2022;71:54-62.
5. Campos-Outcalt D. USPSTF releases updated guidance on asymptomatic A-fb. J Fam Pract. 2022;3. Accessed February 18, 2022. www.mdedge.com/familymedicine/article/251911/cardiology/uspstf-releases-updated-guidance-asymptomatic-fib
6. USPSTF. Screening for atrial fibrillation: US Preventive Services Task Force recommendation statement. JAMA. 2022;327:360-367. doi: 10.1001/jama.2021.23732
7. Svendsen JH, Diederichsen SZ, Hojberg S, et al. Implantable loop recorder detection of atrial fibrillation to prevent stroke (The LOOP Study): a randomised controlled trial. Lancet. 2021;398:1507-1516.
Atrial fibrillation (AF) is a common problem confronting family physicians. In this issue of JFP, we offer 2 articles about AF: one on prevention and one on treatment. Both provide evidence-based guidance to help you refine your care. But gaps remain. I’ll get to that in a bit.
Prevention. This month’s PURL1 discusses a randomized controlled trial (RCT) that enrolled moderate alcohol drinkers with AF.2 Compared to those who continued to drink moderately, those who reduced their alcohol consumption to 2 drinks per week had a significant reduction in recurrent AF (73% vs 53%), fewer hospitalizations (20% vs 9%), and less moderate or severe symptoms (32% vs 10%). Although previous studies of moderate alcohol consumption have shown positive effects on heart disease, this study and other more recent studies cast serious doubt on this assertion.3
Treatment. In their applied evidence article, Osayande and Sharma4 pose the question: When is catheter ablation a sound option for your patient with AF? They give us an excellent, evidence-based answer and remind us that we must focus on the treatment goals: to prevent stroke and to control symptoms. They recommend a stepwise approach, starting with rate control, progressing to rhythm control, and saving catheter ablation for resistant cases. In nearly all cases, anticoagulation to prevent stroke must be a part of treatment, with the exception of those with very low risk (so-called “lone atrial fibrillation”).
Screening. And what about screening for asymptomatic AF? The US Preventive Services Task Force recently reaffirmed its conclusion that there is insufficient evidence for screening for asymptomatic AF (a topic discussed in an online Practice Alert Brief5).6 Since wearable exercise-monitoring devices can detect heart arrhythmias (and are advertised for this purpose), a patient may present after receiving a notification about asymptomatic AF. What shall we do in these cases? The dilemma is that your patient will know she has a potentially dangerous condition, but there is no evidence that treating it will result in more benefit than harm.
A recently published study suggests that we should be very cautious in recommending treatment. In an RCT of patients ages 70 to 90 years, 1501 patients received an implantable loop recorder, while 4503 received routine health care; median follow-up was 64.5 months.7 Although more cases of AF were detected (32% in the monitored group vs 12% in the usual care group), and oral anticoagulation treatment was started more frequently (30% vs 13%, respectively), there was no significant difference in the proportion of patients who had a stroke or systemic arterial embolism (4.5% vs 5.6%).7 Until we have more data, reassurance seems to be the best recommendation for asymptomatic AF.
Atrial fibrillation (AF) is a common problem confronting family physicians. In this issue of JFP, we offer 2 articles about AF: one on prevention and one on treatment. Both provide evidence-based guidance to help you refine your care. But gaps remain. I’ll get to that in a bit.
Prevention. This month’s PURL1 discusses a randomized controlled trial (RCT) that enrolled moderate alcohol drinkers with AF.2 Compared to those who continued to drink moderately, those who reduced their alcohol consumption to 2 drinks per week had a significant reduction in recurrent AF (73% vs 53%), fewer hospitalizations (20% vs 9%), and less moderate or severe symptoms (32% vs 10%). Although previous studies of moderate alcohol consumption have shown positive effects on heart disease, this study and other more recent studies cast serious doubt on this assertion.3
Treatment. In their applied evidence article, Osayande and Sharma4 pose the question: When is catheter ablation a sound option for your patient with AF? They give us an excellent, evidence-based answer and remind us that we must focus on the treatment goals: to prevent stroke and to control symptoms. They recommend a stepwise approach, starting with rate control, progressing to rhythm control, and saving catheter ablation for resistant cases. In nearly all cases, anticoagulation to prevent stroke must be a part of treatment, with the exception of those with very low risk (so-called “lone atrial fibrillation”).
Screening. And what about screening for asymptomatic AF? The US Preventive Services Task Force recently reaffirmed its conclusion that there is insufficient evidence for screening for asymptomatic AF (a topic discussed in an online Practice Alert Brief5).6 Since wearable exercise-monitoring devices can detect heart arrhythmias (and are advertised for this purpose), a patient may present after receiving a notification about asymptomatic AF. What shall we do in these cases? The dilemma is that your patient will know she has a potentially dangerous condition, but there is no evidence that treating it will result in more benefit than harm.
A recently published study suggests that we should be very cautious in recommending treatment. In an RCT of patients ages 70 to 90 years, 1501 patients received an implantable loop recorder, while 4503 received routine health care; median follow-up was 64.5 months.7 Although more cases of AF were detected (32% in the monitored group vs 12% in the usual care group), and oral anticoagulation treatment was started more frequently (30% vs 13%, respectively), there was no significant difference in the proportion of patients who had a stroke or systemic arterial embolism (4.5% vs 5.6%).7 Until we have more data, reassurance seems to be the best recommendation for asymptomatic AF.
1. Thiel DJ, Marshall RC, Rogers TS. Alcohol abstinence reduces A-fib burden in drinkers. J Fam Pract. 2022;71:85-87.
2. Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28. doi: 10.1056/NEJMoa1817591
3. Hoek AG, van Oort S, Mukamal KJ, et al. Alcohol consumption and cardiovascular disease risk: placing new data in context [published online ahead of print, 2022 Feb 7]. Curr Atheroscler Rep. doi: 10.1007/s11883-022-00992-1
4. Osayande AS, Sharma N. When is catheter ablation a sound option for your patient with A-fib? J Fam Pract. 2022;71:54-62.
5. Campos-Outcalt D. USPSTF releases updated guidance on asymptomatic A-fb. J Fam Pract. 2022;3. Accessed February 18, 2022. www.mdedge.com/familymedicine/article/251911/cardiology/uspstf-releases-updated-guidance-asymptomatic-fib
6. USPSTF. Screening for atrial fibrillation: US Preventive Services Task Force recommendation statement. JAMA. 2022;327:360-367. doi: 10.1001/jama.2021.23732
7. Svendsen JH, Diederichsen SZ, Hojberg S, et al. Implantable loop recorder detection of atrial fibrillation to prevent stroke (The LOOP Study): a randomised controlled trial. Lancet. 2021;398:1507-1516.
1. Thiel DJ, Marshall RC, Rogers TS. Alcohol abstinence reduces A-fib burden in drinkers. J Fam Pract. 2022;71:85-87.
2. Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28. doi: 10.1056/NEJMoa1817591
3. Hoek AG, van Oort S, Mukamal KJ, et al. Alcohol consumption and cardiovascular disease risk: placing new data in context [published online ahead of print, 2022 Feb 7]. Curr Atheroscler Rep. doi: 10.1007/s11883-022-00992-1
4. Osayande AS, Sharma N. When is catheter ablation a sound option for your patient with A-fib? J Fam Pract. 2022;71:54-62.
5. Campos-Outcalt D. USPSTF releases updated guidance on asymptomatic A-fb. J Fam Pract. 2022;3. Accessed February 18, 2022. www.mdedge.com/familymedicine/article/251911/cardiology/uspstf-releases-updated-guidance-asymptomatic-fib
6. USPSTF. Screening for atrial fibrillation: US Preventive Services Task Force recommendation statement. JAMA. 2022;327:360-367. doi: 10.1001/jama.2021.23732
7. Svendsen JH, Diederichsen SZ, Hojberg S, et al. Implantable loop recorder detection of atrial fibrillation to prevent stroke (The LOOP Study): a randomised controlled trial. Lancet. 2021;398:1507-1516.
When is catheter ablation a sound option for your patient with A-fib?
CASE
Jack Z, a 75-year-old man with well-controlled hypertension, diabetes controlled by diet, and atrial fibrillation (AF) presents to the family medicine clinic to establish care with you after moving to the community from out of town.
The patient describes a 1-year history of AF. He provides you with an echocardiography report from 6 months ago that shows no evidence of structural heart disease. He takes lisinopril, to control blood pressure; an anticoagulant; a beta-blocker; and amiodarone for rhythm control. Initially, he took flecainide, which was ineffective for rhythm control, before being switched to amiodarone. He had 2 cardioversion procedures, each time after episodes of symptoms. He does not smoke or drink alcohol.
Mr. Z describes worsening palpitations and shortness of breath over the past 9 months. Symptoms now include episodes of exertional fatigue, even when he is not having palpitations. Prior to the episodes of worsening symptoms, he tells you that he lived a “fairly active” life, golfing twice a week.
The patient’s previous primary care physician had encouraged him to talk to his cardiologist about “other options” for managing AF, because levels of his liver enzymes had started to rise (a known adverse effect of amiodarone1) when measured 3 months ago. He did not undertake that conversation, but asks you now about other treatments for AF.
Atrial fibrillation is the most common sustained cardiac arrhythmia, characterized by discordant electrical activation of the atria due to structural or electrophysiological abnormalities, or both. The disorder is associated with an increased rate of stroke and heart failure and is independently associated with a 1.5- to 2-fold risk of all-cause mortality.2
In this article, we review the pathophysiology of AF; management, including the role of, and indications for, catheter ablation; and patient- and disease-related factors associated with ablation (including odds of success, complications, risk of recurrence, and continuing need for thromboprophylaxis) that family physicians should consider when contemplating referral to a cardiologist or electrophysiologist for catheter ablation for AF.
What provokes AF?
AF is thought to occur as a result of an interaction among 3 phenomena:
- enhanced automaticity of abnormal atrial tissue
- triggered activity of ectopic foci within 1 or more pulmonary veins, lying within the left atrium
- re-entry, in which there is propagation of electrical impulses from an ectopic beat through another pathway.
Continue to: In patients who progress...
In patients who progress from paroxysmal to persistent AF (see “Subtypes,” below), 2 distinct pathways, facilitated by the presence of abnormal tissue, continuously activate one another, thus maintaining the arrhythmia. Myocardial tissue in the pulmonary veins is responsible for most ectopic electrical impulses in patients with drug-refractory AF (see “Rhythm control”).
Subtypes. For the purpose of planning treatment, AF is classified as:
- Paroxysmal. Terminates spontaneously or with intervention ≤ 7 days after onset.
- Persistent. Continuous and sustained for > 7 days.
- Longstanding persistent. Continuous for > 12 months.
- Permanent. The patient and physician accept that there will be no further attempt to restore or maintain sinus rhythm.
Goals of treatment
Primary management goals in patients with AF are 2-fold: control of symptoms and prevention of thromboembolism. A patient with new-onset AF who presents acutely with inadequate rate control and hemodynamic compromise requires urgent assessment to determine the cause of the arrhythmia and need for cardioversion.3 A symptomatic patient with AF who does not have high-risk features (eg, valvular heart disease, mechanical valves) might be a candidate for rhythm control in addition to rate control.3,4
Rate control. After evaluation in the hospital, a patient who has a rapid ventricular response but remains hemodynamically stable, without evidence of heart failure, should be initiated on a rate-controlling medication, such as a beta-blocker or nondihydropyridine calcium-channel blocker. A resting heart rate goal of < 80 beats per minute (bpm) is recommended for a symptomatic patient with AF. The heart rate goal can be relaxed, to < 110 bpm, in an asymptomatic patient with preserved left ventricular function.5,6
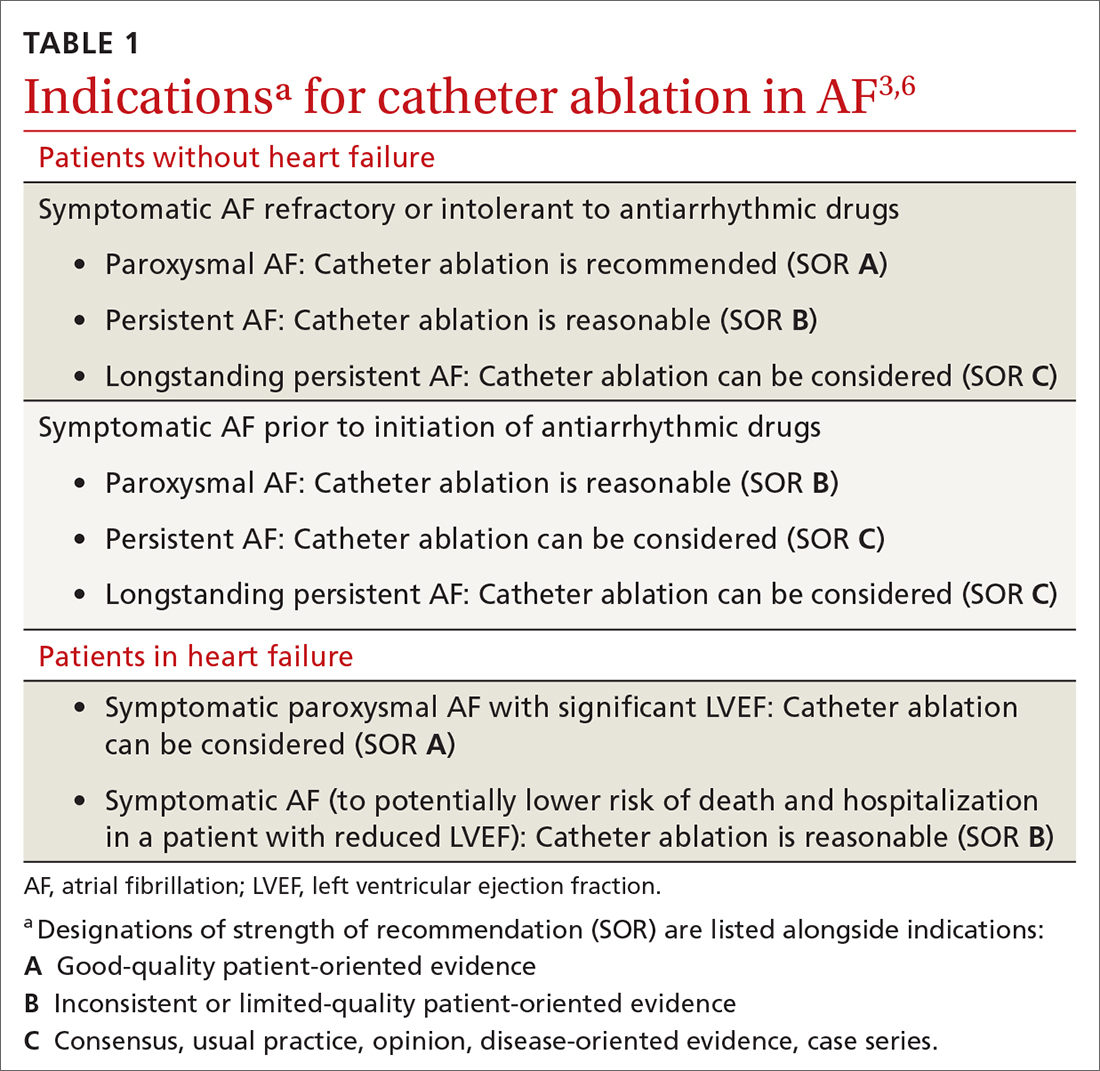
Rhythm control, indicated in patients who remain symptomatic on rate-controlling medication, can be achieved either with an antiarrhythmic drug (AAD) or by catheter ablation.4,5 In stable patients, rhythm control should be considered only after a thorough work-up for a reversible cause of AF, and can be achieved with an oral AAD or, in select patients, through catheter ablation (TABLE 13,6). Other indications for chronic rhythm control include treatment of patients with tachycardia-induced cardiomyopathy.5
A major study that documented the benefit of early rhythm control evaluated long-term outcomes in 2789 patients with AF who were undergoing catheter ablation.7 Patients were randomized to early rhythm control (catheter ablation or AAD) or “usual care”—ie, in this study, rhythm control limited to symptomatic patients. Primary outcomes were death from cardiovascular causes, stroke, and hospitalization with worsening heart failure or acute coronary syndrome. A first primary outcome event occurred in 249 patients (3.9/100 person-years) assigned to early rhythm control, compared to 316 (5.0 per 100 person-years) in the group assigned to usual care.
The study was terminated early (after 5.1 years) because of overwhelming evidence of efficacy (number need to treat = 7). Although early rhythm control was obtained through both catheter ablation and AAD (hazard ratio [HR] = 0.79; 96% CI, 0.66-0.94; P = .005), success was attributed to the use of catheter ablation for a rhythm-control strategy and its use among patients whose AF was present for < 1 year. Most patients in both treatment groups continued to receive anticoagulation, rate control, and optimization of cardiovascular risk.7
Continue to: Notably, direct studies...
Notably, direct studies comparing ablation and AAD have not confirmed the benefit of ablation over AAD in outcomes of all-cause mortality, bleeding, stroke, or cardiac arrest over a 5-year period.8
Adverse effects and mortality outcomes with AAD. Concern over using AAD for rhythm control is based mostly on adverse effects and long-term (1-year) mortality outcomes. Long-term AAD therapy has been shown to decrease the recurrence of AF—but without evidence to suggest other mortality benefits.
A meta-analysis of 59 randomized controlled trials reviewed 20,981 patients receiving AAD (including quinidine, disopyramide, propafenone, flecainide, metoprolol, amiodarone, dofetilide, dronedarone, and sotalol) for long-term effects on death, stroke, adverse reactions, and recurrence of AF.9 Findings at 10 months suggest that:
- Compared to placebo, amiodarone and sotalol increased the risk of all-cause mortality during the study period.
- There was minimal difference in mortality among patients taking dofetilide or dronedarone, compared to placebo.
- There were insufficient data to draw conclusions about the effect of disopyramide, flecainide, and propafenone on mortality.
Before starting a patient on AAD, the risk of arrhythmias and the potential for these agents to cause toxicity and adverse events should always be discussed.
CASE
You tell Mr. Z that you need to know the status of his comorbidities to make a recommendation about “other” management options, and proceed to take a detailed history.
Recent history. Mr. Z reveals that “today is a good day”: He has had “only 1” episode of palpitations, which resolved on its own. The previous episode, he explains, was 3 days ago, when palpitations were associated with lightheadedness and shortness of breath. He denies chest pains or swelling of the legs.
Physical exam. The patient appears spry, comfortable, and in no acute distress. Vital signs are within normal limits. A body mass index of 28.4 puts him in the “overweight” category. His blood pressure is 118/75 mm Hg.
Continue to: Cardiac examination...
Cardiac examination is significant for an irregular rhythm without murmurs, rubs, or gallops. His lungs are clear bilaterally; his abdomen is soft and nondistended. His extremities show no edema.
Testing. You obtain an electrocardiogram, which demonstrates a controlled ventricular rate of 88 bpm and AF. You order a complete blood count, comprehensive metabolic panel, tests of hemoglobin A1C and thyroid-stimulating hormone, lipid panel, echocardiogram, and a chest radiograph.
Results. The chest radiograph is negative for an acute cardiopulmonary process; cardiac size is normal. Aspartate aminotransferase and alanine aminotransferase levels are higher than twice the normal limit. The echocardiogram reveals an estimated left ventricular ejection fraction of 55% to 60%; no structural abnormalities are noted.
In which AF patients is catheter ablation indicated?
Ablation is recommended for select patients (TABLE 13,6) with symptomatic paroxysmal AF that is refractory to AAD or who are intolerant of AAD.3,6 It is a reasonable first-line therapy for high-performing athletes in whom AAD would affect athletic performance.3,10 It is also a reasonable option in select patients > 75 years and as an alternative to AAD therapy.3 Finally, catheter ablation should be considered in symptomatic patients with longstanding persistent AF and congestive heart failure, with or without reduced left ventricular ejection fraction.3
CASE
You inform Mr. Z that his symptoms are likely a result of symptomatic paroxysmal AF, which was refractory to flecainide and amiodarone, and that his abnormal liver function test results preclude continued use of amiodarone. You propose Holter monitoring to correlate timing of symptoms with the arrhythmia, but he reports this has been done, and the correlation confirmed, by his previous physician.
You explain that, because the diagnosis of symptomatic paroxysmal AF refractory to AADs has been confirmed, he is categorized as a patient who might benefit from catheter ablation, based on:
- the type of AF (ie, paroxysmal AF is associated with better ablation outcomes)
- persistent symptoms that are refractory to AADs
- his intolerance of AAD
- the length of time since onset of symptoms.
Mr. Z agrees to consider your recommendation.
Continue to: What are the benefits of catheter ablation?
What are the benefits of catheter ablation?
Ablation can be achieved through radiofrequency (RF) ablation, cryoablation, or newer, laser-based balloon ablation. Primary outcomes used to determine the success of any options for performing ablation include mortality, stroke, and hospitalization. Other endpoints include maintenance of sinus rhythm, freedom from AF, reduction in AF burden (estimated through patients’ report of symptoms, recurrence rate, need for a second ablation procedure, and serial long-term monitoring through an implantable cardiac monitoring device), quality of life, and prevention of AF progression.3
Patient and disease variables (TABLE 211-13). The success rate of catheter ablation, defined as freedom from either symptomatic or asymptomatic episodes of AF, is dependent on several factors,3,14 including:
- type of AF (paroxysmal or persistent)
- duration and degree of symptoms
- age
- sex
- comorbidities, including heart failure and structural heart or lung disease.
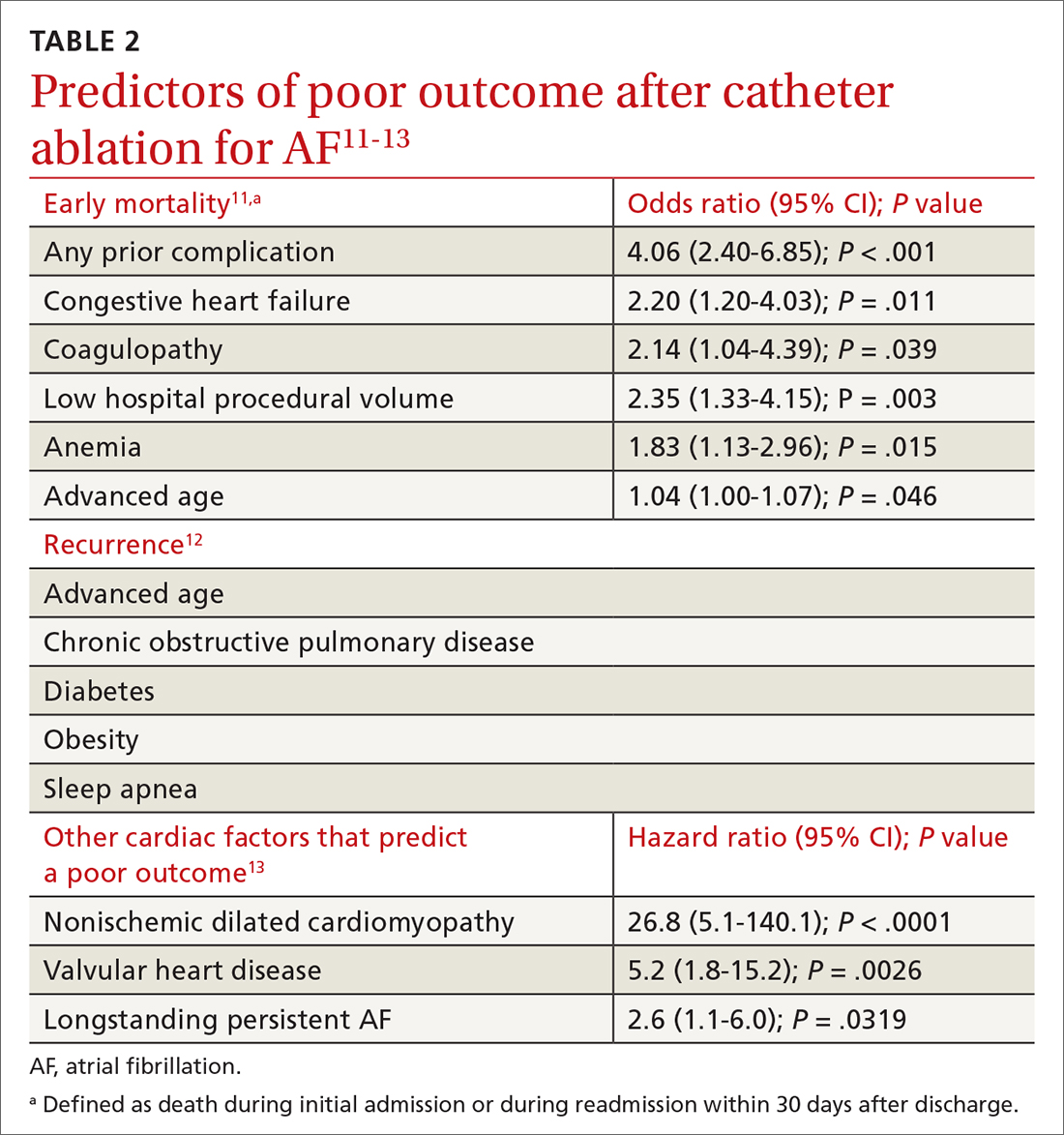
Overall, in patients with paroxysmal AF, an estimated 75% are symptom free 1 year after ablation.15 Patients with persistent and longstanding persistent AF experience a lower success rate.
RF catheter ablation has demonstrated superiority to AAD in reducing the need for cardioversion (relative risk [RR] = 0.62; 95% CI, 0.47-0.82) and cardiac-related hospitalization (RR = 0.27; 95% CI, 0.10-0.72;) at 12 months in patients with nonparoxysmal AF (persistent or longstanding persistent).16
Effect on mortality. Among patients with heart failure with reduced ejection fraction, long-term studies of cardiovascular outcomes 5 years post ablation concluded that ablation is associated with a decrease in all-cause mortality (RR = 0.69; 95% CI, 0.54-0.88; P = .003) and a reduction in hospitalization (RR = 0.62; 95% CI, 0.47-0.82; P = .0006); younger (< 65 years) and male patients derive greater benefit.6,17 Indications for ablation in patients with heart failure are similar to those in patients without heart failure; ablation can therefore be considered for select heart failure patients who remain symptomatic or for whom AAD has failed.3
Older patients. Ablation can be considered for patients > 75 years with symptomatic paroxysmal AF refractory to AAD or who are intolerant of AAD.3 A study that assessed the benefit of catheter ablation reviewed 587 older (> 75 years) patients with AF, of whom 324 were eligible for ablation. Endpoints were maintenance of sinus rhythm, stroke, death, and major bleeding. Return to normal sinus rhythm was an independent factor, associated with a decrease in the risk of mortality among all patient groups that underwent ablation (HR = 0.36; 95% CI, 0.2-0.63; P = .0005). Age > 75 years (HR = 1.09; 95% CI, 1.01-1.16; P > .02) and depressed ejection fraction < 40% (HR = 2.38; 95% CI, 1.28-4.4; P = .006) were determined to be unfavorable parameters for survival.18
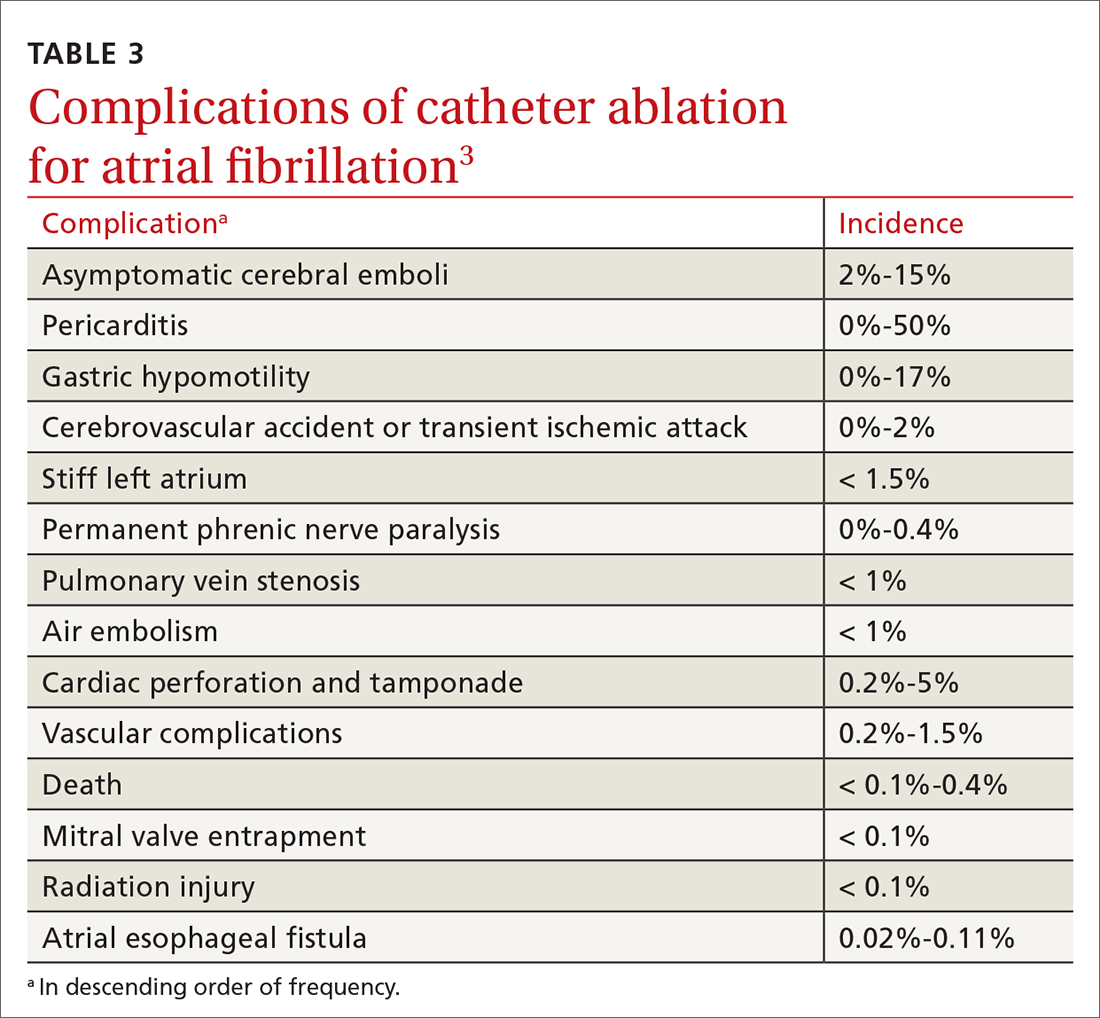
Complications and risks
Complications of catheter ablation for AF, although infrequent, can be severe (TABLE 33). Early mortality, defined as death during initial admission or 30-day readmission, occurs in approximately 0.5% of cases; half of deaths take place during readmission.11
Continue to: Complications vary...
Complications vary, based on the type and site of ablation.19,20 Cardiac tamponade or perforation, the most life-threatening complications, taken together occur in an estimated 1.9% of patients (odds ratio [OR] = 2.98; 95% CI, 1.36-6.56; P = .007).11 Other in-hospital complications independently predictive of death include any cardiac complications (OR = 12.8; 95% CI, 6.86 to 23.8; P < .001) and neurologic complications (cerebrovascular accident and transient ischemic attack) (OR = 8.72; 95% CI, 2.71-28.1; P < .001).
Other complications that do not cause death but might prolong the hospital stay include pericarditis without effusion, anesthesia-related complications, and vascular-access complications. Patients whose ablation is performed at an institution where the volume of ablations is low are also at higher risk of early mortality (OR = 2.35; 95% CI, 1.33-4.15; P = .003).16
Recurrence is common (TABLE 211-13). Risk of recurrence following ablation is significant; early (within 3 months after ablation) recurrence is seen in 50% of patients.21,22 However, this is a so-called "blanking period"—ie, a temporary period of inflammatory and proarrhythmic changes that are not predictors of later recurrence. The 5-year post-ablation recurrence rate is approximately 25.5%; longstanding persistent and persistent AF and the presence of comorbidities are major risk factors for recurrence.13,23
Recurrence is also associated with the type of procedure; pulmonary vein isolation, alone or in combination with another type of procedure, results in higher long-term success.21,23
Other variables affect outcome (TABLE 211-13). Following AF ablation, patients with nonparoxysmal AF at baseline, advanced age, sleep apnea and obesity, left atrial enlargement, and any structural heart disease tend to have a poorer long-term (5-year) outcome (ie, freedom from extended episodes of AF).3,13,23,24
Patients who undergo repeat procedures have higher arrhythmia-free survival; the highest ablation success rate is for patients with paroxysmal AF.13,23
Exposure to ionizing radiation. Fluoroscopy is required for multiple components of atrial mapping and ablation during RF ablation, including navigation, visualization, and monitoring of catheter placement. Patients undergoing this particular procedure therefore receive significant exposure to ionizing radiation. A reduction in, even complete elimination of, fluoroscopy has been achieved with:
- nonfluoroscopic 3-dimensional mapping systems25
- intracardiac echocardiography, which utilizes ultrasonographic imaging as the primary visual mode for tracking and manipulating the catheter
- robotic guided navigation.26-28
Continue to: CASE
CASE
At his return visit, Mr. Z says that he is concerned about, first, undergoing catheter ablation at his age and, second, the risks associated with the procedure. You explain that it is true that ablation is ideal in younger patients who have minimal comorbidities and that the risk of complications increases with age—but that there is no cutoff or absolute age contraindication to ablation.
You tell Mr. Z that you will work with him on risk-factor modification in anticipation of ablation. You also assure him that the decision whether to ablate must be a joint one—between him and a cardiologist experienced both in electrophysiology and in performing this highly technical procedure. And you explain that a highly practiced specialist can identify Mr. Z’s risk factors that might make ablation more difficult to perform and affect the long-term outcome.
With Mr. Z’s agreement, you screen for sleep apnea and start him on a lifestyle modification plan to achieve a more ideal weight, explaining that the risk of recurrence of AF after catheter ablation is increased by obesity and sleep apnea, in addition to age. You explain that, based on his CHA2DS2–VASc (congestive heart failure; hypertension; age, ≥ 75 years; diabetes; prior stroke, transient ischemic attack, or thromboembolism; vascular disease; age, 65 to 74 years; sex category) score of 3, he will remain on anticoagulation whether or not he has the ablation.
You refer the patient to the nearest high-volume cardiac ablation center.
Last, you caution Mr. Z that, based on his lipid levels, his 10-year risk of heart disease or stroke is elevated. You recommend treatment with a statin agent while he continues his other medications.
Delivering energy to myocardium
Myocardial tissue in pulmonary veins is responsible for most ectopic electrical impulses in patients with drug-refractory AF. The goal of catheter ablation in AF is destruction (scarring) of tissue that is the source of abnormal vein potentials.15
How RF ablation works. Ablation is most commonly performed using RF energy, a high-frequency form of electrical energy. Electrophysiology studies are carried out at the time of ablation by percutaneous, fluoroscopically guided insertion of 2 to 5 catheters, usually through the femoral or internal jugular vein, which are then positioned within several areas of the heart—usually, the right atrium, bundle of His, right ventricle, and coronary sinus.
Continue to: Electrical current...
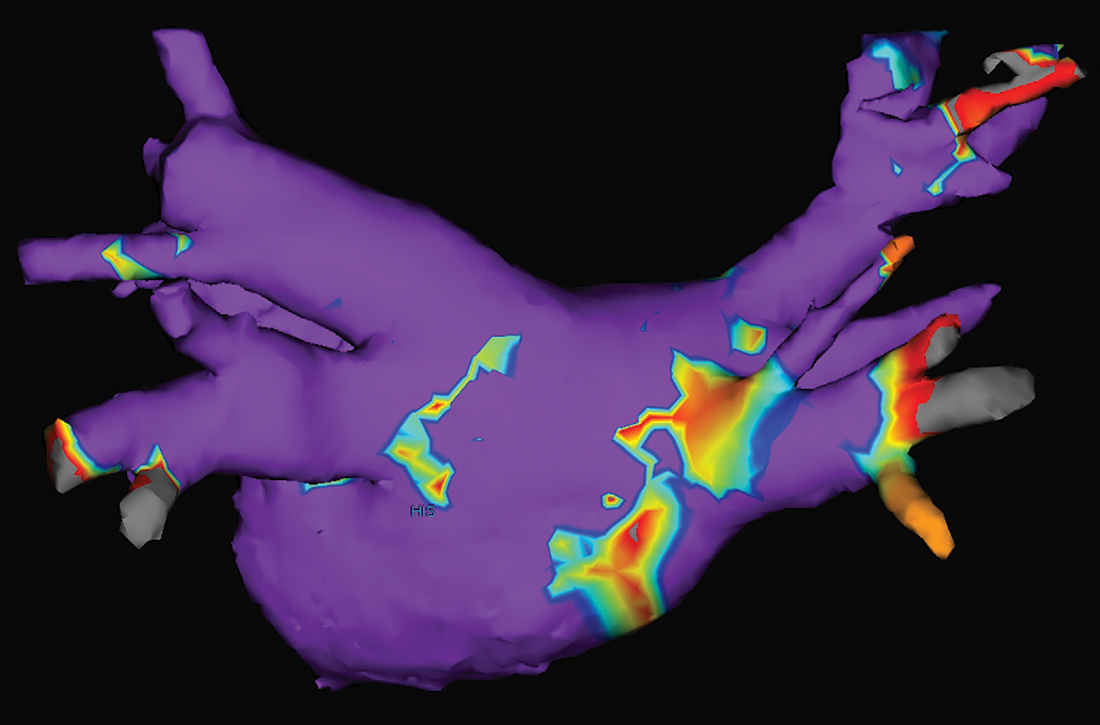
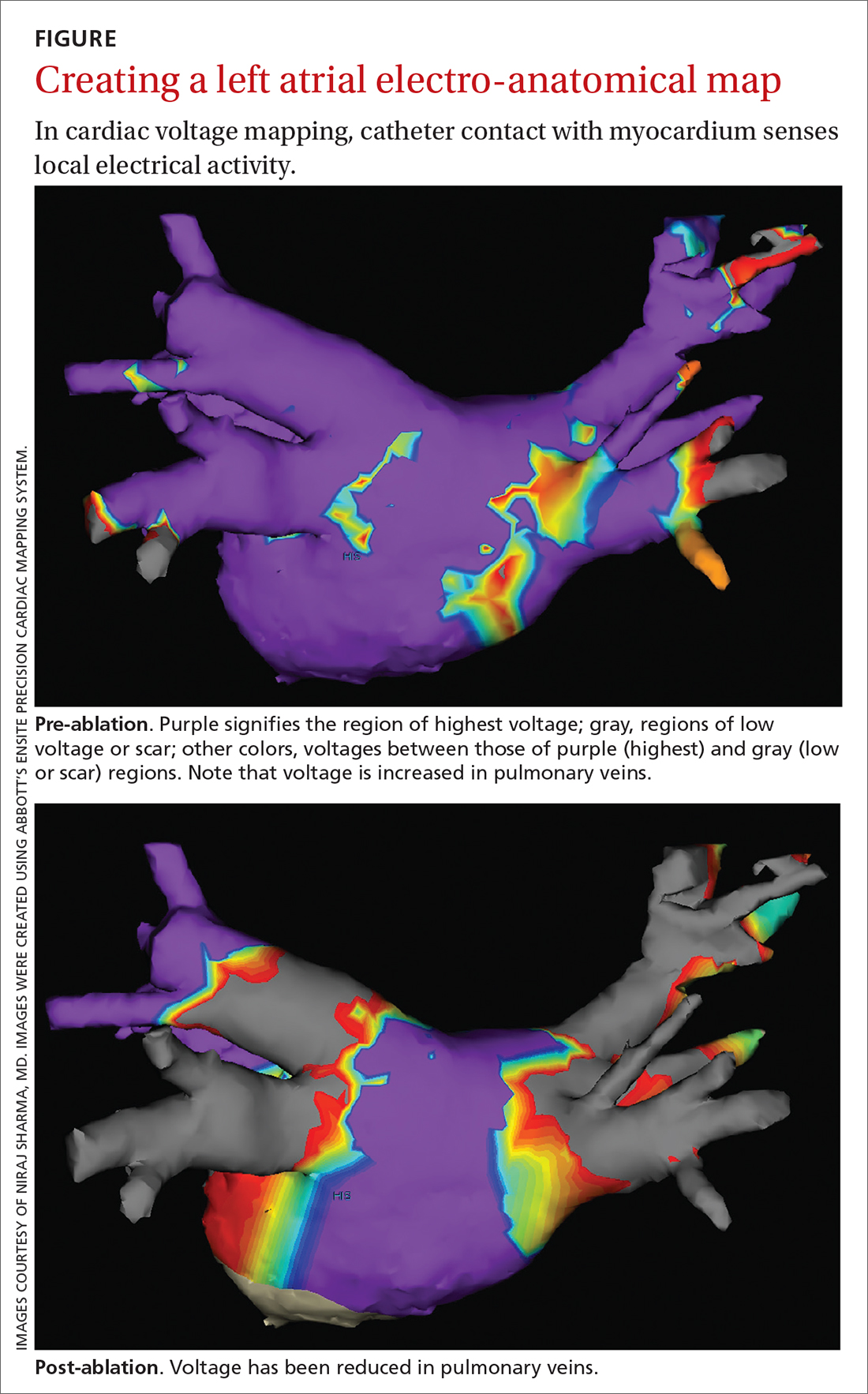
Electrical current is applied through the catheters from an external generator to stimulate the myocardium and thus determine its electrophysiologic properties. The anatomic and electrical activity of the left atrium and pulmonary veins is then identified, a technique known as electro-anatomical mapping (FIGURE). After arrhythmogenic myocardial tissue is mapped, ablation is carried out with RF energy through the catheter to the pathogenic myocardium from which arrhythmias are initiated or conducted. The result is thermal destruction of tissue and creation of small, shallow lesions that vary in size with the type of catheter and the force of contact pressure applied.3,29
Other energy sources used in catheter ablation include cryothermal energy, which utilizes liquid nitrous oxide under pressure through a cryocatheter or cryoballoon catheter. Application of cryothermal energy freezes tissue and disrupts cell membranes and any electrical activity. Cryoballoon ablation has been shown to be similarly safe and efficacious as RF ablation in patients with paroxysmal AF.30,31
Newer laser-based balloon ablations are performed under ultrasonographic guidance and utilize arcs of laser energy delivered to the pathogenic myocardium.3
Thromboembolism prophylaxis
Oral anticoagulation to decrease the risk of stroke is initiated in all patients with AF, based on a thromboembolic risk profile determined by their CHA2DS2–VASc score, with anticoagulation recommended when the score is ≥ 2 in men and ≥ 3 in women. Options for anticoagulation include warfarin and one of the novel oral anticoagulants dabigatran, apixaban, rivaroxaban, and edoxaban.4 Recommendations are as follows3:
- For patients with a CHA2DS2–VASc score of ≥ 2 (men) or ≥ 3 (women), anticoagulation should be continued indefinitely, regardless of how successful the ablation procedure is.
- When patients choose to discontinue anticoagulation, they should be counseled in detail about the risk of doing so. The continued need for frequent arrhythmia monitoring should be emphasized.
The route from primary careto catheter ablation
Perform a thorough evaluation. Patients who present to you with palpitations should first undergo a routine workup for AF, followed by confirmation of the diagnosis. Exclude structural heart disease with echocardiography. Undertake monitoring, which is essential to determine whether symptoms are a reflection of the arrhythmia, using noncontinuous or continuous electrocardiographic (EKG) monitoring. Noncontinuous detection devices include:
- scheduled or symptom-initiated EKG
- a Holter monitor, worn for at least 24 hours and as long as 7 days
- trans-telephonic recordings and patient- or automatically activated devices
- an external loop recorder.32
Continuous EKG monitoring is more permanent (≥ 12 months). This is usually achieved through an implantable loop powered by a battery that lasts as long as 3 years.3
Ablation: Yes or no? Ablation is not recommended to avoid anticoagulation or when anticoagulation is contraindicated.5 With regard to specific patient criteria, the ideal patient:
- is symptomatic
- has failed AAD therapy
- does not have pulmonary disease
- has a normal or mildly dilated left atrium or normal or mildly reduced left ventricular ejection fraction.5
Continue to: There is no absolute age...
There is no absolute age or comorbidity contraindication to ablation. The patient should be referred to a cardiologist who has received appropriate training in electrophysiology, to identify comorbidities that (1) increase the technical difficulty of the procedure and baseline risk and (2) affect long-term outcome,12 and who performs the procedure in a center that has considerable experience with catheter ablation.33
Once the decision is made to perform ablation, you can provide strategies that optimize the outcome (freedom from AF episodes). Those tactics include weight loss and screening evaluation and, if indicated, treatment for sleep apnea.3
Protocol. Prior to the procedure, the patient fasts overnight; they might be asked to taper or discontinue cardiac medications that have electrophysiologic effects. Studies suggest a low risk of bleeding associated with catheter ablation; anticoagulation should therefore continue uninterrupted for patients undergoing catheter ablation for AF3,4,34,35; however, this practice varies with the cardiologist or electrophysiologist performing ablation.
Because of the length and complexity of the procedure, electro-anatomical mapping and ablation are conducted with the patient under general anesthesia.3 The patient is kept supine, and remains so for 2 to 4 hours afterward to allow for hemostasis at puncture sites.3
Patients might be monitored overnight, although same-day catheter ablation has been shown to be safe and cost-effective in select patients.36,37 Post ablation, patients follow up with the cardiologist and electrophysiologist. Long-term arrhythmia monitoring is required.3 Anticoagulation is continued for at least 2 months, and is discontinued based on the patient’s risk for stroke, utilizing their CHA2DS2–VASc score.3,4
CASE
At Mr. Z’s 6-month primary care follow-up, he confirms what has been reported to you as the referring physician: He had a successful catheter ablation and continues to have regular follow-up monitoring with the cardiologist. He is no longer taking amiodarone.
At this visit, he reports no recurrence of AF-associated symptoms or detectable AF on cardiac monitoring. He has lost 8 lbs. You counsel to him to continue to maintain a healthy lifestyle.
CORRESPONDENCE
Amimi S. Osayande MD, FAAFP, Northside-Gwinnett Family Medicine Residency Program, Strickland Family Medicine Center, 665 Duluth Highway, Suite 501, Lawrenceville, GA 30046; amimi.osayande@northside.com
1. Amiodarone hydrochloride (marketed as Cordarone and Pacerone) information. Silver Spring, Md.: US Food & Drug Administration. Reviewed March 23, 2015. Accessed January 16, 2022. www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/amiodarone-hydrochloride-marketed-cordarone-and-pacerone-information
2. Gómez-Outes A, Suárez-Gea ML,García-Pinilla JM. Causes of death in atrial fibrillation: challenges and opportunities. Trends Cardiovasc Med. 2017;27:494-503. doi: 10.1016/j.tcm.2017.05.002
3. Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/ APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: executive summary. J Arrhythm. 2017;33:369-409. doi: 10.1016/j.joa.2017.08.001
4. Camm AJ, Lip GYH, De Caterina R, et al; ESC Committee for Practice Guidelines-CPG; Document Reviewers. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation—developed with the special contribution of the European Heart Rhythm Association. Europace. 2012;14:1385-1413. doi: 10.1093/europace/eus305
5. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: a report of the American College of Cardiology/ American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Circulation. 2014;130:2071-2104. doi: 10.1161/CIR.0000000000000040
6. January CT, Wann LS, Calkins H, et al; Writing Group Members. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/ HRS guideline for the management of patients with atrial fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2019;16:e66-e93. doi: 10.1016/j.hrthm.2019.01.024
7. Kirchhof P, Camm AJ, Goette A, et al; EAST-AFNET 4 Trial Investigators. Early rhythm-control therapy in patients with atrial fibrillation. N Engl J Med. 2020;383:1305-1316. doi: 10.1056/ NEJMoa2019422
8. Packer DL, Mark DB, Robb RA, et al; CABANA Investigators. Effect of catheter ablation vs antiarrhythmic drug therapy on mortality, stroke, bleeding, and cardiac arrest among patients with atrial fibrillation: the CABANA randomized clinical trial. JAMA. 2019;321:1261-1274. doi: 10.1001/jama.2019.0693
9. Valembois L, Audureau E, Takeda A, et al. Antiarrhythmics for maintaining sinus rhythm after cardioversion of atrial fibrillation. Cochrane Database Syst Rev. 2019;9:CD005049. doi: 10.1002/14651858.CD005049
10. Koopman P, Nuyens D, Garweg C, et al. Efficacy of radiofrequency catheter ablation in athletes with atrial fibrillation. Europace. 2011;13:1386-1393. doi: 10.1093/europace/eur142
11. Hakalahti A, Biancari F, Nielsen JC, et al. Radiofrequency ablation vs. antiarrhythmic drug therapy as first line treatment of symptomatic atrial fibrillation: systematic review and meta-analysis. Europace. 2015;17:370-378. doi: 10.1093/europace/euu376
12. Nyong J, Amit G, Adler AJ, et al. Efficacy and safety of ablation for people with non-paroxysmal atrial fibrillation. Cochrane Database Syst Rev. 2016;11:CD012088. doi: 10.1002/14651858. CD012088.pub2
13. Andrade JG, Champagne J, Dubuc M, et al; CIRCA-DOSE Study Investigators. Cryoballoon or radiofrequency ablation for atrial fibrillation assessed by continuous monitoring: a randomized clinical trial. Circulation. 2019;140:1779-1788. doi: 10.1161/ CIRCULATIONAHA.119.042622
14. Asad ZUA, Yousif A, Khan MS, et al. Catheter ablation versus medical therapy for atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Circ Arrhythm Electrophysiol. 2019;12:e007414. doi: 10.1161/ CIRCEP.119.007414
15. Nademanee K, Amnueypol M, Lee F, et al. Benefits and risks of catheter ablation in elderly patients with atrial fibrillation. Heart Rhythm. 2015;12:44-51. doi: 10.1016/j.hrthm.2014.09.049
16. Cheng EP, Liu CF, Yeo I, et al. Risk of mortality following catheter ablation of atrial fibrillation. J Am Coll Cardiol. 2019;74: 2254-2264. doi: 10.1016/j.jacc.2019.08.1036
17. Brugada J, Katritsis DG, Arbelo E, et al; ESC Scientific Document Group. 2019 ESC Guidelines for the management of patients with supraventricular tachycardia. The Task Force for the management of patients with supraventricular tachycardia of the European Society of Cardiology (ESC). Developed in collaboration with the Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2020;41:655-720. doi: 10.1093/eurheartj/ehz467
18. Hosseini SM, Rozen G, Saleh A, et al. Catheter ablation for cardiac arrhythmias: utilization and in-hospital complications, 2000 to 2013. JACC Clin Electrophysiol. 2017;3:1240-1248. doi: 10.1016/j.jacep.2017.05.005
19. Andrade JG, Macle L, Khairy P, et al. Incidence and significance of early recurrences associated with different ablation strategies for AF: a STAR-AF substudy. J Cardiovasc Electrophysiol. 2012;23:1295-1301. doi: 10.1111/j.1540-8167.2012.02399.x
20. Joshi S, Choi AD, Kamath GS, et al. Prevalence, predictors, and prognosis of atrial fibrillation early after pulmonary vein isolation: findings from 3 months of continuous automatic ECG loop recordings. J Cardiovasc Electrophysiol. 2009;20:1089-1094. doi: 10.1111/j.1540-8167.2009.01506.x
21. Weerasooriya R, Khairy P, Litalien J, et al. Catheter ablation for atrial fibrillation: are results maintained at 5 years of follow-up? J Am Coll Cardiol. 2011;57:160-166. doi: 10.1016/j.jacc.2010.05.061
22. Ouyang F, Tilz R, Chun J, et al. Long-term results of catheter ablation in paroxysmal atrial fibrillation: lessons from a 5-year follow-up. Circulation. 2010;122:2368-2377. doi: 10.1161/ CIRCULATIONAHA.110.946806
23. Tilz RR, Rillig A, Thum A-M, et al. Catheter ablation of long-standing persistent atrial fibrillation: 5-year outcomes of the Hamburg Sequential Ablation Strategy. J Am Coll Cardiol. 2012;60: 1921-1929. doi: 10.1016/j.jacc.2012.04.060
24. Forkmann M, Schwab C, Busch S. [Catheter ablation of supraventricular tachycardia]. Herzschrittmacherther Elektrophysiol. 2019;30:336-342. doi: 10.1007/s00399-019-00654-x
25. Bulava A, Hanis J, Eisenberger M. Catheter ablation of atrial fibrillation using zero-fluoroscopy technique: a randomized trial. Pacing Clin Electrophysiol. 2015;38:797-806. doi: 10.1111/pace.12634
26. Haegeli LM, Stutz L, Mohsen M, et al. Feasibility of zero or near zero fluoroscopy during catheter ablation procedures. Cardiol J. 2019;26:226-232. doi: 10.5603/CJ.a2018.0029
27. Steven D, Servatius H, Rostock T, et al. Reduced fluoroscopy during atrial fibrillation ablation: benefits of robotic guided navigation. J Cardiovasc Electrophysiol. 2010;21:6-12. doi: 10.1111/j.1540-8167.2009.01592.x
28. General therapy for cardiac arrhythmias. In: Zipes DP, Libby P, Bonow RO, et al. Braunwald’s Heart Disease: A Textbook of Cardiovascular Medicine. 11th ed. Elsevier; 2019.
29. Kuck K-H, Brugada J, Albenque J-P. Cryoballoon or radiofrequency ablation for atrial fibrillation. N Engl J Med. 2016;375: 1100-1101. doi: 10.1056/NEJMc1609160
30. Chen Y-H, Lu Z-Y, Xiang Y, et al. Cryoablation vs. radiofrequency ablation for treatment of paroxysmal atrial fibrillation: a systematic review and meta-analysis. Europace. 2017;19:784-794. doi: 10.1093/europace/euw330
31. Locati ET, Vecchi AM, Vargiu S, et al. Role of extended external loop recorders for the diagnosis of unexplained syncope, presyncope, and sustained palpitations. Europace. 2014;16:914-922. doi: 10.1093/europace/eut337
32. Calkins H, Kuck KH, Cappato R, et al; Heart Rhythm Society Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. Heart Rhythm. 2012;9:632-696.e21. doi: 10.1016/j.hrthm.2011.12.016
33. Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace. 2016;18:1609-1678. doi: 10.1093/ europace/euw295
34. Nairooz R, Sardar P, Payne J, et al. Meta-analysis of major bleeding with uninterrupted warfarin compared to interrupted warfarin and heparin bridging in ablation of atrial fibrillation. Int J Cardiol. 2015;187:426-429. doi: 10.1016/j.ijcard.2015.03.376
35. Romero J, Cerrud-Rodriguez RC, Diaz JC, et al. Uninterrupted direct oral anticoagulants vs. uninterrupted vitamin K antagonists during catheter ablation of non-valvular atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Europace. 2018;20:1612-1620. doi: 10.1093/europace/euy133
36. Deyell MW, Leather RA, Macle L, et al. Efficacy and safety of same-day discharge for atrial fibrillation ablation. JACC Clin Electrophysiol. 2020;6:609-619. doi: 10.1016/j.jacep.2020.02.009
37. Theodoreson MD, Chohan BC, McAloon CJ, et al. Same-day cardiac catheter ablation is safe and cost-effective: experience from a UK tertiary center. Heart Rhythm. 2015;12:1756-1761. doi: 10.1016/j.hrthm.2015.05.006
CASE
Jack Z, a 75-year-old man with well-controlled hypertension, diabetes controlled by diet, and atrial fibrillation (AF) presents to the family medicine clinic to establish care with you after moving to the community from out of town.
The patient describes a 1-year history of AF. He provides you with an echocardiography report from 6 months ago that shows no evidence of structural heart disease. He takes lisinopril, to control blood pressure; an anticoagulant; a beta-blocker; and amiodarone for rhythm control. Initially, he took flecainide, which was ineffective for rhythm control, before being switched to amiodarone. He had 2 cardioversion procedures, each time after episodes of symptoms. He does not smoke or drink alcohol.
Mr. Z describes worsening palpitations and shortness of breath over the past 9 months. Symptoms now include episodes of exertional fatigue, even when he is not having palpitations. Prior to the episodes of worsening symptoms, he tells you that he lived a “fairly active” life, golfing twice a week.
The patient’s previous primary care physician had encouraged him to talk to his cardiologist about “other options” for managing AF, because levels of his liver enzymes had started to rise (a known adverse effect of amiodarone1) when measured 3 months ago. He did not undertake that conversation, but asks you now about other treatments for AF.
Atrial fibrillation is the most common sustained cardiac arrhythmia, characterized by discordant electrical activation of the atria due to structural or electrophysiological abnormalities, or both. The disorder is associated with an increased rate of stroke and heart failure and is independently associated with a 1.5- to 2-fold risk of all-cause mortality.2
In this article, we review the pathophysiology of AF; management, including the role of, and indications for, catheter ablation; and patient- and disease-related factors associated with ablation (including odds of success, complications, risk of recurrence, and continuing need for thromboprophylaxis) that family physicians should consider when contemplating referral to a cardiologist or electrophysiologist for catheter ablation for AF.
What provokes AF?
AF is thought to occur as a result of an interaction among 3 phenomena:
- enhanced automaticity of abnormal atrial tissue
- triggered activity of ectopic foci within 1 or more pulmonary veins, lying within the left atrium
- re-entry, in which there is propagation of electrical impulses from an ectopic beat through another pathway.
Continue to: In patients who progress...
In patients who progress from paroxysmal to persistent AF (see “Subtypes,” below), 2 distinct pathways, facilitated by the presence of abnormal tissue, continuously activate one another, thus maintaining the arrhythmia. Myocardial tissue in the pulmonary veins is responsible for most ectopic electrical impulses in patients with drug-refractory AF (see “Rhythm control”).
Subtypes. For the purpose of planning treatment, AF is classified as:
- Paroxysmal. Terminates spontaneously or with intervention ≤ 7 days after onset.
- Persistent. Continuous and sustained for > 7 days.
- Longstanding persistent. Continuous for > 12 months.
- Permanent. The patient and physician accept that there will be no further attempt to restore or maintain sinus rhythm.
Goals of treatment
Primary management goals in patients with AF are 2-fold: control of symptoms and prevention of thromboembolism. A patient with new-onset AF who presents acutely with inadequate rate control and hemodynamic compromise requires urgent assessment to determine the cause of the arrhythmia and need for cardioversion.3 A symptomatic patient with AF who does not have high-risk features (eg, valvular heart disease, mechanical valves) might be a candidate for rhythm control in addition to rate control.3,4
Rate control. After evaluation in the hospital, a patient who has a rapid ventricular response but remains hemodynamically stable, without evidence of heart failure, should be initiated on a rate-controlling medication, such as a beta-blocker or nondihydropyridine calcium-channel blocker. A resting heart rate goal of < 80 beats per minute (bpm) is recommended for a symptomatic patient with AF. The heart rate goal can be relaxed, to < 110 bpm, in an asymptomatic patient with preserved left ventricular function.5,6
Rhythm control, indicated in patients who remain symptomatic on rate-controlling medication, can be achieved either with an antiarrhythmic drug (AAD) or by catheter ablation.4,5 In stable patients, rhythm control should be considered only after a thorough work-up for a reversible cause of AF, and can be achieved with an oral AAD or, in select patients, through catheter ablation (TABLE 13,6). Other indications for chronic rhythm control include treatment of patients with tachycardia-induced cardiomyopathy.5

A major study that documented the benefit of early rhythm control evaluated long-term outcomes in 2789 patients with AF who were undergoing catheter ablation.7 Patients were randomized to early rhythm control (catheter ablation or AAD) or “usual care”—ie, in this study, rhythm control limited to symptomatic patients. Primary outcomes were death from cardiovascular causes, stroke, and hospitalization with worsening heart failure or acute coronary syndrome. A first primary outcome event occurred in 249 patients (3.9/100 person-years) assigned to early rhythm control, compared to 316 (5.0 per 100 person-years) in the group assigned to usual care.
The study was terminated early (after 5.1 years) because of overwhelming evidence of efficacy (number need to treat = 7). Although early rhythm control was obtained through both catheter ablation and AAD (hazard ratio [HR] = 0.79; 96% CI, 0.66-0.94; P = .005), success was attributed to the use of catheter ablation for a rhythm-control strategy and its use among patients whose AF was present for < 1 year. Most patients in both treatment groups continued to receive anticoagulation, rate control, and optimization of cardiovascular risk.7
Continue to: Notably, direct studies...
Notably, direct studies comparing ablation and AAD have not confirmed the benefit of ablation over AAD in outcomes of all-cause mortality, bleeding, stroke, or cardiac arrest over a 5-year period.8
Adverse effects and mortality outcomes with AAD. Concern over using AAD for rhythm control is based mostly on adverse effects and long-term (1-year) mortality outcomes. Long-term AAD therapy has been shown to decrease the recurrence of AF—but without evidence to suggest other mortality benefits.
A meta-analysis of 59 randomized controlled trials reviewed 20,981 patients receiving AAD (including quinidine, disopyramide, propafenone, flecainide, metoprolol, amiodarone, dofetilide, dronedarone, and sotalol) for long-term effects on death, stroke, adverse reactions, and recurrence of AF.9 Findings at 10 months suggest that:
- Compared to placebo, amiodarone and sotalol increased the risk of all-cause mortality during the study period.
- There was minimal difference in mortality among patients taking dofetilide or dronedarone, compared to placebo.
- There were insufficient data to draw conclusions about the effect of disopyramide, flecainide, and propafenone on mortality.
Before starting a patient on AAD, the risk of arrhythmias and the potential for these agents to cause toxicity and adverse events should always be discussed.
CASE
You tell Mr. Z that you need to know the status of his comorbidities to make a recommendation about “other” management options, and proceed to take a detailed history.
Recent history. Mr. Z reveals that “today is a good day”: He has had “only 1” episode of palpitations, which resolved on its own. The previous episode, he explains, was 3 days ago, when palpitations were associated with lightheadedness and shortness of breath. He denies chest pains or swelling of the legs.
Physical exam. The patient appears spry, comfortable, and in no acute distress. Vital signs are within normal limits. A body mass index of 28.4 puts him in the “overweight” category. His blood pressure is 118/75 mm Hg.
Continue to: Cardiac examination...
Cardiac examination is significant for an irregular rhythm without murmurs, rubs, or gallops. His lungs are clear bilaterally; his abdomen is soft and nondistended. His extremities show no edema.
Testing. You obtain an electrocardiogram, which demonstrates a controlled ventricular rate of 88 bpm and AF. You order a complete blood count, comprehensive metabolic panel, tests of hemoglobin A1C and thyroid-stimulating hormone, lipid panel, echocardiogram, and a chest radiograph.
Results. The chest radiograph is negative for an acute cardiopulmonary process; cardiac size is normal. Aspartate aminotransferase and alanine aminotransferase levels are higher than twice the normal limit. The echocardiogram reveals an estimated left ventricular ejection fraction of 55% to 60%; no structural abnormalities are noted.
In which AF patients is catheter ablation indicated?
Ablation is recommended for select patients (TABLE 13,6) with symptomatic paroxysmal AF that is refractory to AAD or who are intolerant of AAD.3,6 It is a reasonable first-line therapy for high-performing athletes in whom AAD would affect athletic performance.3,10 It is also a reasonable option in select patients > 75 years and as an alternative to AAD therapy.3 Finally, catheter ablation should be considered in symptomatic patients with longstanding persistent AF and congestive heart failure, with or without reduced left ventricular ejection fraction.3
CASE
You inform Mr. Z that his symptoms are likely a result of symptomatic paroxysmal AF, which was refractory to flecainide and amiodarone, and that his abnormal liver function test results preclude continued use of amiodarone. You propose Holter monitoring to correlate timing of symptoms with the arrhythmia, but he reports this has been done, and the correlation confirmed, by his previous physician.
You explain that, because the diagnosis of symptomatic paroxysmal AF refractory to AADs has been confirmed, he is categorized as a patient who might benefit from catheter ablation, based on:
- the type of AF (ie, paroxysmal AF is associated with better ablation outcomes)
- persistent symptoms that are refractory to AADs
- his intolerance of AAD
- the length of time since onset of symptoms.
Mr. Z agrees to consider your recommendation.
Continue to: What are the benefits of catheter ablation?
What are the benefits of catheter ablation?
Ablation can be achieved through radiofrequency (RF) ablation, cryoablation, or newer, laser-based balloon ablation. Primary outcomes used to determine the success of any options for performing ablation include mortality, stroke, and hospitalization. Other endpoints include maintenance of sinus rhythm, freedom from AF, reduction in AF burden (estimated through patients’ report of symptoms, recurrence rate, need for a second ablation procedure, and serial long-term monitoring through an implantable cardiac monitoring device), quality of life, and prevention of AF progression.3
Patient and disease variables (TABLE 211-13). The success rate of catheter ablation, defined as freedom from either symptomatic or asymptomatic episodes of AF, is dependent on several factors,3,14 including:
- type of AF (paroxysmal or persistent)
- duration and degree of symptoms
- age
- sex
- comorbidities, including heart failure and structural heart or lung disease.

Overall, in patients with paroxysmal AF, an estimated 75% are symptom free 1 year after ablation.15 Patients with persistent and longstanding persistent AF experience a lower success rate.
RF catheter ablation has demonstrated superiority to AAD in reducing the need for cardioversion (relative risk [RR] = 0.62; 95% CI, 0.47-0.82) and cardiac-related hospitalization (RR = 0.27; 95% CI, 0.10-0.72;) at 12 months in patients with nonparoxysmal AF (persistent or longstanding persistent).16
Effect on mortality. Among patients with heart failure with reduced ejection fraction, long-term studies of cardiovascular outcomes 5 years post ablation concluded that ablation is associated with a decrease in all-cause mortality (RR = 0.69; 95% CI, 0.54-0.88; P = .003) and a reduction in hospitalization (RR = 0.62; 95% CI, 0.47-0.82; P = .0006); younger (< 65 years) and male patients derive greater benefit.6,17 Indications for ablation in patients with heart failure are similar to those in patients without heart failure; ablation can therefore be considered for select heart failure patients who remain symptomatic or for whom AAD has failed.3
Older patients. Ablation can be considered for patients > 75 years with symptomatic paroxysmal AF refractory to AAD or who are intolerant of AAD.3 A study that assessed the benefit of catheter ablation reviewed 587 older (> 75 years) patients with AF, of whom 324 were eligible for ablation. Endpoints were maintenance of sinus rhythm, stroke, death, and major bleeding. Return to normal sinus rhythm was an independent factor, associated with a decrease in the risk of mortality among all patient groups that underwent ablation (HR = 0.36; 95% CI, 0.2-0.63; P = .0005). Age > 75 years (HR = 1.09; 95% CI, 1.01-1.16; P > .02) and depressed ejection fraction < 40% (HR = 2.38; 95% CI, 1.28-4.4; P = .006) were determined to be unfavorable parameters for survival.18
Complications and risks
Complications of catheter ablation for AF, although infrequent, can be severe (TABLE 33). Early mortality, defined as death during initial admission or 30-day readmission, occurs in approximately 0.5% of cases; half of deaths take place during readmission.11

Continue to: Complications vary...
Complications vary, based on the type and site of ablation.19,20 Cardiac tamponade or perforation, the most life-threatening complications, taken together occur in an estimated 1.9% of patients (odds ratio [OR] = 2.98; 95% CI, 1.36-6.56; P = .007).11 Other in-hospital complications independently predictive of death include any cardiac complications (OR = 12.8; 95% CI, 6.86 to 23.8; P < .001) and neurologic complications (cerebrovascular accident and transient ischemic attack) (OR = 8.72; 95% CI, 2.71-28.1; P < .001).
Other complications that do not cause death but might prolong the hospital stay include pericarditis without effusion, anesthesia-related complications, and vascular-access complications. Patients whose ablation is performed at an institution where the volume of ablations is low are also at higher risk of early mortality (OR = 2.35; 95% CI, 1.33-4.15; P = .003).16
Recurrence is common (TABLE 211-13). Risk of recurrence following ablation is significant; early (within 3 months after ablation) recurrence is seen in 50% of patients.21,22 However, this is a so-called "blanking period"—ie, a temporary period of inflammatory and proarrhythmic changes that are not predictors of later recurrence. The 5-year post-ablation recurrence rate is approximately 25.5%; longstanding persistent and persistent AF and the presence of comorbidities are major risk factors for recurrence.13,23
Recurrence is also associated with the type of procedure; pulmonary vein isolation, alone or in combination with another type of procedure, results in higher long-term success.21,23
Other variables affect outcome (TABLE 211-13). Following AF ablation, patients with nonparoxysmal AF at baseline, advanced age, sleep apnea and obesity, left atrial enlargement, and any structural heart disease tend to have a poorer long-term (5-year) outcome (ie, freedom from extended episodes of AF).3,13,23,24
Patients who undergo repeat procedures have higher arrhythmia-free survival; the highest ablation success rate is for patients with paroxysmal AF.13,23
Exposure to ionizing radiation. Fluoroscopy is required for multiple components of atrial mapping and ablation during RF ablation, including navigation, visualization, and monitoring of catheter placement. Patients undergoing this particular procedure therefore receive significant exposure to ionizing radiation. A reduction in, even complete elimination of, fluoroscopy has been achieved with:
- nonfluoroscopic 3-dimensional mapping systems25
- intracardiac echocardiography, which utilizes ultrasonographic imaging as the primary visual mode for tracking and manipulating the catheter
- robotic guided navigation.26-28
Continue to: CASE
CASE
At his return visit, Mr. Z says that he is concerned about, first, undergoing catheter ablation at his age and, second, the risks associated with the procedure. You explain that it is true that ablation is ideal in younger patients who have minimal comorbidities and that the risk of complications increases with age—but that there is no cutoff or absolute age contraindication to ablation.
You tell Mr. Z that you will work with him on risk-factor modification in anticipation of ablation. You also assure him that the decision whether to ablate must be a joint one—between him and a cardiologist experienced both in electrophysiology and in performing this highly technical procedure. And you explain that a highly practiced specialist can identify Mr. Z’s risk factors that might make ablation more difficult to perform and affect the long-term outcome.
With Mr. Z’s agreement, you screen for sleep apnea and start him on a lifestyle modification plan to achieve a more ideal weight, explaining that the risk of recurrence of AF after catheter ablation is increased by obesity and sleep apnea, in addition to age. You explain that, based on his CHA2DS2–VASc (congestive heart failure; hypertension; age, ≥ 75 years; diabetes; prior stroke, transient ischemic attack, or thromboembolism; vascular disease; age, 65 to 74 years; sex category) score of 3, he will remain on anticoagulation whether or not he has the ablation.
You refer the patient to the nearest high-volume cardiac ablation center.
Last, you caution Mr. Z that, based on his lipid levels, his 10-year risk of heart disease or stroke is elevated. You recommend treatment with a statin agent while he continues his other medications.
Delivering energy to myocardium
Myocardial tissue in pulmonary veins is responsible for most ectopic electrical impulses in patients with drug-refractory AF. The goal of catheter ablation in AF is destruction (scarring) of tissue that is the source of abnormal vein potentials.15
How RF ablation works. Ablation is most commonly performed using RF energy, a high-frequency form of electrical energy. Electrophysiology studies are carried out at the time of ablation by percutaneous, fluoroscopically guided insertion of 2 to 5 catheters, usually through the femoral or internal jugular vein, which are then positioned within several areas of the heart—usually, the right atrium, bundle of His, right ventricle, and coronary sinus.
Continue to: Electrical current...
Electrical current is applied through the catheters from an external generator to stimulate the myocardium and thus determine its electrophysiologic properties. The anatomic and electrical activity of the left atrium and pulmonary veins is then identified, a technique known as electro-anatomical mapping (FIGURE). After arrhythmogenic myocardial tissue is mapped, ablation is carried out with RF energy through the catheter to the pathogenic myocardium from which arrhythmias are initiated or conducted. The result is thermal destruction of tissue and creation of small, shallow lesions that vary in size with the type of catheter and the force of contact pressure applied.3,29

Other energy sources used in catheter ablation include cryothermal energy, which utilizes liquid nitrous oxide under pressure through a cryocatheter or cryoballoon catheter. Application of cryothermal energy freezes tissue and disrupts cell membranes and any electrical activity. Cryoballoon ablation has been shown to be similarly safe and efficacious as RF ablation in patients with paroxysmal AF.30,31
Newer laser-based balloon ablations are performed under ultrasonographic guidance and utilize arcs of laser energy delivered to the pathogenic myocardium.3
Thromboembolism prophylaxis
Oral anticoagulation to decrease the risk of stroke is initiated in all patients with AF, based on a thromboembolic risk profile determined by their CHA2DS2–VASc score, with anticoagulation recommended when the score is ≥ 2 in men and ≥ 3 in women. Options for anticoagulation include warfarin and one of the novel oral anticoagulants dabigatran, apixaban, rivaroxaban, and edoxaban.4 Recommendations are as follows3:
- For patients with a CHA2DS2–VASc score of ≥ 2 (men) or ≥ 3 (women), anticoagulation should be continued indefinitely, regardless of how successful the ablation procedure is.
- When patients choose to discontinue anticoagulation, they should be counseled in detail about the risk of doing so. The continued need for frequent arrhythmia monitoring should be emphasized.
The route from primary careto catheter ablation
Perform a thorough evaluation. Patients who present to you with palpitations should first undergo a routine workup for AF, followed by confirmation of the diagnosis. Exclude structural heart disease with echocardiography. Undertake monitoring, which is essential to determine whether symptoms are a reflection of the arrhythmia, using noncontinuous or continuous electrocardiographic (EKG) monitoring. Noncontinuous detection devices include:
- scheduled or symptom-initiated EKG
- a Holter monitor, worn for at least 24 hours and as long as 7 days
- trans-telephonic recordings and patient- or automatically activated devices
- an external loop recorder.32
Continuous EKG monitoring is more permanent (≥ 12 months). This is usually achieved through an implantable loop powered by a battery that lasts as long as 3 years.3
Ablation: Yes or no? Ablation is not recommended to avoid anticoagulation or when anticoagulation is contraindicated.5 With regard to specific patient criteria, the ideal patient:
- is symptomatic
- has failed AAD therapy
- does not have pulmonary disease
- has a normal or mildly dilated left atrium or normal or mildly reduced left ventricular ejection fraction.5
Continue to: There is no absolute age...
There is no absolute age or comorbidity contraindication to ablation. The patient should be referred to a cardiologist who has received appropriate training in electrophysiology, to identify comorbidities that (1) increase the technical difficulty of the procedure and baseline risk and (2) affect long-term outcome,12 and who performs the procedure in a center that has considerable experience with catheter ablation.33
Once the decision is made to perform ablation, you can provide strategies that optimize the outcome (freedom from AF episodes). Those tactics include weight loss and screening evaluation and, if indicated, treatment for sleep apnea.3
Protocol. Prior to the procedure, the patient fasts overnight; they might be asked to taper or discontinue cardiac medications that have electrophysiologic effects. Studies suggest a low risk of bleeding associated with catheter ablation; anticoagulation should therefore continue uninterrupted for patients undergoing catheter ablation for AF3,4,34,35; however, this practice varies with the cardiologist or electrophysiologist performing ablation.
Because of the length and complexity of the procedure, electro-anatomical mapping and ablation are conducted with the patient under general anesthesia.3 The patient is kept supine, and remains so for 2 to 4 hours afterward to allow for hemostasis at puncture sites.3
Patients might be monitored overnight, although same-day catheter ablation has been shown to be safe and cost-effective in select patients.36,37 Post ablation, patients follow up with the cardiologist and electrophysiologist. Long-term arrhythmia monitoring is required.3 Anticoagulation is continued for at least 2 months, and is discontinued based on the patient’s risk for stroke, utilizing their CHA2DS2–VASc score.3,4
CASE
At Mr. Z’s 6-month primary care follow-up, he confirms what has been reported to you as the referring physician: He had a successful catheter ablation and continues to have regular follow-up monitoring with the cardiologist. He is no longer taking amiodarone.
At this visit, he reports no recurrence of AF-associated symptoms or detectable AF on cardiac monitoring. He has lost 8 lbs. You counsel to him to continue to maintain a healthy lifestyle.
CORRESPONDENCE
Amimi S. Osayande MD, FAAFP, Northside-Gwinnett Family Medicine Residency Program, Strickland Family Medicine Center, 665 Duluth Highway, Suite 501, Lawrenceville, GA 30046; amimi.osayande@northside.com
CASE
Jack Z, a 75-year-old man with well-controlled hypertension, diabetes controlled by diet, and atrial fibrillation (AF) presents to the family medicine clinic to establish care with you after moving to the community from out of town.
The patient describes a 1-year history of AF. He provides you with an echocardiography report from 6 months ago that shows no evidence of structural heart disease. He takes lisinopril, to control blood pressure; an anticoagulant; a beta-blocker; and amiodarone for rhythm control. Initially, he took flecainide, which was ineffective for rhythm control, before being switched to amiodarone. He had 2 cardioversion procedures, each time after episodes of symptoms. He does not smoke or drink alcohol.
Mr. Z describes worsening palpitations and shortness of breath over the past 9 months. Symptoms now include episodes of exertional fatigue, even when he is not having palpitations. Prior to the episodes of worsening symptoms, he tells you that he lived a “fairly active” life, golfing twice a week.
The patient’s previous primary care physician had encouraged him to talk to his cardiologist about “other options” for managing AF, because levels of his liver enzymes had started to rise (a known adverse effect of amiodarone1) when measured 3 months ago. He did not undertake that conversation, but asks you now about other treatments for AF.
Atrial fibrillation is the most common sustained cardiac arrhythmia, characterized by discordant electrical activation of the atria due to structural or electrophysiological abnormalities, or both. The disorder is associated with an increased rate of stroke and heart failure and is independently associated with a 1.5- to 2-fold risk of all-cause mortality.2
In this article, we review the pathophysiology of AF; management, including the role of, and indications for, catheter ablation; and patient- and disease-related factors associated with ablation (including odds of success, complications, risk of recurrence, and continuing need for thromboprophylaxis) that family physicians should consider when contemplating referral to a cardiologist or electrophysiologist for catheter ablation for AF.
What provokes AF?
AF is thought to occur as a result of an interaction among 3 phenomena:
- enhanced automaticity of abnormal atrial tissue
- triggered activity of ectopic foci within 1 or more pulmonary veins, lying within the left atrium
- re-entry, in which there is propagation of electrical impulses from an ectopic beat through another pathway.
Continue to: In patients who progress...
In patients who progress from paroxysmal to persistent AF (see “Subtypes,” below), 2 distinct pathways, facilitated by the presence of abnormal tissue, continuously activate one another, thus maintaining the arrhythmia. Myocardial tissue in the pulmonary veins is responsible for most ectopic electrical impulses in patients with drug-refractory AF (see “Rhythm control”).
Subtypes. For the purpose of planning treatment, AF is classified as:
- Paroxysmal. Terminates spontaneously or with intervention ≤ 7 days after onset.
- Persistent. Continuous and sustained for > 7 days.
- Longstanding persistent. Continuous for > 12 months.
- Permanent. The patient and physician accept that there will be no further attempt to restore or maintain sinus rhythm.
Goals of treatment
Primary management goals in patients with AF are 2-fold: control of symptoms and prevention of thromboembolism. A patient with new-onset AF who presents acutely with inadequate rate control and hemodynamic compromise requires urgent assessment to determine the cause of the arrhythmia and need for cardioversion.3 A symptomatic patient with AF who does not have high-risk features (eg, valvular heart disease, mechanical valves) might be a candidate for rhythm control in addition to rate control.3,4
Rate control. After evaluation in the hospital, a patient who has a rapid ventricular response but remains hemodynamically stable, without evidence of heart failure, should be initiated on a rate-controlling medication, such as a beta-blocker or nondihydropyridine calcium-channel blocker. A resting heart rate goal of < 80 beats per minute (bpm) is recommended for a symptomatic patient with AF. The heart rate goal can be relaxed, to < 110 bpm, in an asymptomatic patient with preserved left ventricular function.5,6
Rhythm control, indicated in patients who remain symptomatic on rate-controlling medication, can be achieved either with an antiarrhythmic drug (AAD) or by catheter ablation.4,5 In stable patients, rhythm control should be considered only after a thorough work-up for a reversible cause of AF, and can be achieved with an oral AAD or, in select patients, through catheter ablation (TABLE 13,6). Other indications for chronic rhythm control include treatment of patients with tachycardia-induced cardiomyopathy.5

A major study that documented the benefit of early rhythm control evaluated long-term outcomes in 2789 patients with AF who were undergoing catheter ablation.7 Patients were randomized to early rhythm control (catheter ablation or AAD) or “usual care”—ie, in this study, rhythm control limited to symptomatic patients. Primary outcomes were death from cardiovascular causes, stroke, and hospitalization with worsening heart failure or acute coronary syndrome. A first primary outcome event occurred in 249 patients (3.9/100 person-years) assigned to early rhythm control, compared to 316 (5.0 per 100 person-years) in the group assigned to usual care.
The study was terminated early (after 5.1 years) because of overwhelming evidence of efficacy (number need to treat = 7). Although early rhythm control was obtained through both catheter ablation and AAD (hazard ratio [HR] = 0.79; 96% CI, 0.66-0.94; P = .005), success was attributed to the use of catheter ablation for a rhythm-control strategy and its use among patients whose AF was present for < 1 year. Most patients in both treatment groups continued to receive anticoagulation, rate control, and optimization of cardiovascular risk.7
Continue to: Notably, direct studies...
Notably, direct studies comparing ablation and AAD have not confirmed the benefit of ablation over AAD in outcomes of all-cause mortality, bleeding, stroke, or cardiac arrest over a 5-year period.8
Adverse effects and mortality outcomes with AAD. Concern over using AAD for rhythm control is based mostly on adverse effects and long-term (1-year) mortality outcomes. Long-term AAD therapy has been shown to decrease the recurrence of AF—but without evidence to suggest other mortality benefits.
A meta-analysis of 59 randomized controlled trials reviewed 20,981 patients receiving AAD (including quinidine, disopyramide, propafenone, flecainide, metoprolol, amiodarone, dofetilide, dronedarone, and sotalol) for long-term effects on death, stroke, adverse reactions, and recurrence of AF.9 Findings at 10 months suggest that:
- Compared to placebo, amiodarone and sotalol increased the risk of all-cause mortality during the study period.
- There was minimal difference in mortality among patients taking dofetilide or dronedarone, compared to placebo.
- There were insufficient data to draw conclusions about the effect of disopyramide, flecainide, and propafenone on mortality.
Before starting a patient on AAD, the risk of arrhythmias and the potential for these agents to cause toxicity and adverse events should always be discussed.
CASE
You tell Mr. Z that you need to know the status of his comorbidities to make a recommendation about “other” management options, and proceed to take a detailed history.
Recent history. Mr. Z reveals that “today is a good day”: He has had “only 1” episode of palpitations, which resolved on its own. The previous episode, he explains, was 3 days ago, when palpitations were associated with lightheadedness and shortness of breath. He denies chest pains or swelling of the legs.
Physical exam. The patient appears spry, comfortable, and in no acute distress. Vital signs are within normal limits. A body mass index of 28.4 puts him in the “overweight” category. His blood pressure is 118/75 mm Hg.
Continue to: Cardiac examination...
Cardiac examination is significant for an irregular rhythm without murmurs, rubs, or gallops. His lungs are clear bilaterally; his abdomen is soft and nondistended. His extremities show no edema.
Testing. You obtain an electrocardiogram, which demonstrates a controlled ventricular rate of 88 bpm and AF. You order a complete blood count, comprehensive metabolic panel, tests of hemoglobin A1C and thyroid-stimulating hormone, lipid panel, echocardiogram, and a chest radiograph.
Results. The chest radiograph is negative for an acute cardiopulmonary process; cardiac size is normal. Aspartate aminotransferase and alanine aminotransferase levels are higher than twice the normal limit. The echocardiogram reveals an estimated left ventricular ejection fraction of 55% to 60%; no structural abnormalities are noted.
In which AF patients is catheter ablation indicated?
Ablation is recommended for select patients (TABLE 13,6) with symptomatic paroxysmal AF that is refractory to AAD or who are intolerant of AAD.3,6 It is a reasonable first-line therapy for high-performing athletes in whom AAD would affect athletic performance.3,10 It is also a reasonable option in select patients > 75 years and as an alternative to AAD therapy.3 Finally, catheter ablation should be considered in symptomatic patients with longstanding persistent AF and congestive heart failure, with or without reduced left ventricular ejection fraction.3
CASE
You inform Mr. Z that his symptoms are likely a result of symptomatic paroxysmal AF, which was refractory to flecainide and amiodarone, and that his abnormal liver function test results preclude continued use of amiodarone. You propose Holter monitoring to correlate timing of symptoms with the arrhythmia, but he reports this has been done, and the correlation confirmed, by his previous physician.
You explain that, because the diagnosis of symptomatic paroxysmal AF refractory to AADs has been confirmed, he is categorized as a patient who might benefit from catheter ablation, based on:
- the type of AF (ie, paroxysmal AF is associated with better ablation outcomes)
- persistent symptoms that are refractory to AADs
- his intolerance of AAD
- the length of time since onset of symptoms.
Mr. Z agrees to consider your recommendation.
Continue to: What are the benefits of catheter ablation?
What are the benefits of catheter ablation?
Ablation can be achieved through radiofrequency (RF) ablation, cryoablation, or newer, laser-based balloon ablation. Primary outcomes used to determine the success of any options for performing ablation include mortality, stroke, and hospitalization. Other endpoints include maintenance of sinus rhythm, freedom from AF, reduction in AF burden (estimated through patients’ report of symptoms, recurrence rate, need for a second ablation procedure, and serial long-term monitoring through an implantable cardiac monitoring device), quality of life, and prevention of AF progression.3
Patient and disease variables (TABLE 211-13). The success rate of catheter ablation, defined as freedom from either symptomatic or asymptomatic episodes of AF, is dependent on several factors,3,14 including:
- type of AF (paroxysmal or persistent)
- duration and degree of symptoms
- age
- sex
- comorbidities, including heart failure and structural heart or lung disease.

Overall, in patients with paroxysmal AF, an estimated 75% are symptom free 1 year after ablation.15 Patients with persistent and longstanding persistent AF experience a lower success rate.
RF catheter ablation has demonstrated superiority to AAD in reducing the need for cardioversion (relative risk [RR] = 0.62; 95% CI, 0.47-0.82) and cardiac-related hospitalization (RR = 0.27; 95% CI, 0.10-0.72;) at 12 months in patients with nonparoxysmal AF (persistent or longstanding persistent).16
Effect on mortality. Among patients with heart failure with reduced ejection fraction, long-term studies of cardiovascular outcomes 5 years post ablation concluded that ablation is associated with a decrease in all-cause mortality (RR = 0.69; 95% CI, 0.54-0.88; P = .003) and a reduction in hospitalization (RR = 0.62; 95% CI, 0.47-0.82; P = .0006); younger (< 65 years) and male patients derive greater benefit.6,17 Indications for ablation in patients with heart failure are similar to those in patients without heart failure; ablation can therefore be considered for select heart failure patients who remain symptomatic or for whom AAD has failed.3
Older patients. Ablation can be considered for patients > 75 years with symptomatic paroxysmal AF refractory to AAD or who are intolerant of AAD.3 A study that assessed the benefit of catheter ablation reviewed 587 older (> 75 years) patients with AF, of whom 324 were eligible for ablation. Endpoints were maintenance of sinus rhythm, stroke, death, and major bleeding. Return to normal sinus rhythm was an independent factor, associated with a decrease in the risk of mortality among all patient groups that underwent ablation (HR = 0.36; 95% CI, 0.2-0.63; P = .0005). Age > 75 years (HR = 1.09; 95% CI, 1.01-1.16; P > .02) and depressed ejection fraction < 40% (HR = 2.38; 95% CI, 1.28-4.4; P = .006) were determined to be unfavorable parameters for survival.18
Complications and risks
Complications of catheter ablation for AF, although infrequent, can be severe (TABLE 33). Early mortality, defined as death during initial admission or 30-day readmission, occurs in approximately 0.5% of cases; half of deaths take place during readmission.11

Continue to: Complications vary...
Complications vary, based on the type and site of ablation.19,20 Cardiac tamponade or perforation, the most life-threatening complications, taken together occur in an estimated 1.9% of patients (odds ratio [OR] = 2.98; 95% CI, 1.36-6.56; P = .007).11 Other in-hospital complications independently predictive of death include any cardiac complications (OR = 12.8; 95% CI, 6.86 to 23.8; P < .001) and neurologic complications (cerebrovascular accident and transient ischemic attack) (OR = 8.72; 95% CI, 2.71-28.1; P < .001).
Other complications that do not cause death but might prolong the hospital stay include pericarditis without effusion, anesthesia-related complications, and vascular-access complications. Patients whose ablation is performed at an institution where the volume of ablations is low are also at higher risk of early mortality (OR = 2.35; 95% CI, 1.33-4.15; P = .003).16
Recurrence is common (TABLE 211-13). Risk of recurrence following ablation is significant; early (within 3 months after ablation) recurrence is seen in 50% of patients.21,22 However, this is a so-called "blanking period"—ie, a temporary period of inflammatory and proarrhythmic changes that are not predictors of later recurrence. The 5-year post-ablation recurrence rate is approximately 25.5%; longstanding persistent and persistent AF and the presence of comorbidities are major risk factors for recurrence.13,23
Recurrence is also associated with the type of procedure; pulmonary vein isolation, alone or in combination with another type of procedure, results in higher long-term success.21,23
Other variables affect outcome (TABLE 211-13). Following AF ablation, patients with nonparoxysmal AF at baseline, advanced age, sleep apnea and obesity, left atrial enlargement, and any structural heart disease tend to have a poorer long-term (5-year) outcome (ie, freedom from extended episodes of AF).3,13,23,24
Patients who undergo repeat procedures have higher arrhythmia-free survival; the highest ablation success rate is for patients with paroxysmal AF.13,23
Exposure to ionizing radiation. Fluoroscopy is required for multiple components of atrial mapping and ablation during RF ablation, including navigation, visualization, and monitoring of catheter placement. Patients undergoing this particular procedure therefore receive significant exposure to ionizing radiation. A reduction in, even complete elimination of, fluoroscopy has been achieved with:
- nonfluoroscopic 3-dimensional mapping systems25
- intracardiac echocardiography, which utilizes ultrasonographic imaging as the primary visual mode for tracking and manipulating the catheter
- robotic guided navigation.26-28
Continue to: CASE
CASE
At his return visit, Mr. Z says that he is concerned about, first, undergoing catheter ablation at his age and, second, the risks associated with the procedure. You explain that it is true that ablation is ideal in younger patients who have minimal comorbidities and that the risk of complications increases with age—but that there is no cutoff or absolute age contraindication to ablation.
You tell Mr. Z that you will work with him on risk-factor modification in anticipation of ablation. You also assure him that the decision whether to ablate must be a joint one—between him and a cardiologist experienced both in electrophysiology and in performing this highly technical procedure. And you explain that a highly practiced specialist can identify Mr. Z’s risk factors that might make ablation more difficult to perform and affect the long-term outcome.
With Mr. Z’s agreement, you screen for sleep apnea and start him on a lifestyle modification plan to achieve a more ideal weight, explaining that the risk of recurrence of AF after catheter ablation is increased by obesity and sleep apnea, in addition to age. You explain that, based on his CHA2DS2–VASc (congestive heart failure; hypertension; age, ≥ 75 years; diabetes; prior stroke, transient ischemic attack, or thromboembolism; vascular disease; age, 65 to 74 years; sex category) score of 3, he will remain on anticoagulation whether or not he has the ablation.
You refer the patient to the nearest high-volume cardiac ablation center.
Last, you caution Mr. Z that, based on his lipid levels, his 10-year risk of heart disease or stroke is elevated. You recommend treatment with a statin agent while he continues his other medications.
Delivering energy to myocardium
Myocardial tissue in pulmonary veins is responsible for most ectopic electrical impulses in patients with drug-refractory AF. The goal of catheter ablation in AF is destruction (scarring) of tissue that is the source of abnormal vein potentials.15
How RF ablation works. Ablation is most commonly performed using RF energy, a high-frequency form of electrical energy. Electrophysiology studies are carried out at the time of ablation by percutaneous, fluoroscopically guided insertion of 2 to 5 catheters, usually through the femoral or internal jugular vein, which are then positioned within several areas of the heart—usually, the right atrium, bundle of His, right ventricle, and coronary sinus.
Continue to: Electrical current...
Electrical current is applied through the catheters from an external generator to stimulate the myocardium and thus determine its electrophysiologic properties. The anatomic and electrical activity of the left atrium and pulmonary veins is then identified, a technique known as electro-anatomical mapping (FIGURE). After arrhythmogenic myocardial tissue is mapped, ablation is carried out with RF energy through the catheter to the pathogenic myocardium from which arrhythmias are initiated or conducted. The result is thermal destruction of tissue and creation of small, shallow lesions that vary in size with the type of catheter and the force of contact pressure applied.3,29

Other energy sources used in catheter ablation include cryothermal energy, which utilizes liquid nitrous oxide under pressure through a cryocatheter or cryoballoon catheter. Application of cryothermal energy freezes tissue and disrupts cell membranes and any electrical activity. Cryoballoon ablation has been shown to be similarly safe and efficacious as RF ablation in patients with paroxysmal AF.30,31
Newer laser-based balloon ablations are performed under ultrasonographic guidance and utilize arcs of laser energy delivered to the pathogenic myocardium.3
Thromboembolism prophylaxis
Oral anticoagulation to decrease the risk of stroke is initiated in all patients with AF, based on a thromboembolic risk profile determined by their CHA2DS2–VASc score, with anticoagulation recommended when the score is ≥ 2 in men and ≥ 3 in women. Options for anticoagulation include warfarin and one of the novel oral anticoagulants dabigatran, apixaban, rivaroxaban, and edoxaban.4 Recommendations are as follows3:
- For patients with a CHA2DS2–VASc score of ≥ 2 (men) or ≥ 3 (women), anticoagulation should be continued indefinitely, regardless of how successful the ablation procedure is.
- When patients choose to discontinue anticoagulation, they should be counseled in detail about the risk of doing so. The continued need for frequent arrhythmia monitoring should be emphasized.
The route from primary careto catheter ablation
Perform a thorough evaluation. Patients who present to you with palpitations should first undergo a routine workup for AF, followed by confirmation of the diagnosis. Exclude structural heart disease with echocardiography. Undertake monitoring, which is essential to determine whether symptoms are a reflection of the arrhythmia, using noncontinuous or continuous electrocardiographic (EKG) monitoring. Noncontinuous detection devices include:
- scheduled or symptom-initiated EKG
- a Holter monitor, worn for at least 24 hours and as long as 7 days
- trans-telephonic recordings and patient- or automatically activated devices
- an external loop recorder.32
Continuous EKG monitoring is more permanent (≥ 12 months). This is usually achieved through an implantable loop powered by a battery that lasts as long as 3 years.3
Ablation: Yes or no? Ablation is not recommended to avoid anticoagulation or when anticoagulation is contraindicated.5 With regard to specific patient criteria, the ideal patient:
- is symptomatic
- has failed AAD therapy
- does not have pulmonary disease
- has a normal or mildly dilated left atrium or normal or mildly reduced left ventricular ejection fraction.5
Continue to: There is no absolute age...
There is no absolute age or comorbidity contraindication to ablation. The patient should be referred to a cardiologist who has received appropriate training in electrophysiology, to identify comorbidities that (1) increase the technical difficulty of the procedure and baseline risk and (2) affect long-term outcome,12 and who performs the procedure in a center that has considerable experience with catheter ablation.33
Once the decision is made to perform ablation, you can provide strategies that optimize the outcome (freedom from AF episodes). Those tactics include weight loss and screening evaluation and, if indicated, treatment for sleep apnea.3
Protocol. Prior to the procedure, the patient fasts overnight; they might be asked to taper or discontinue cardiac medications that have electrophysiologic effects. Studies suggest a low risk of bleeding associated with catheter ablation; anticoagulation should therefore continue uninterrupted for patients undergoing catheter ablation for AF3,4,34,35; however, this practice varies with the cardiologist or electrophysiologist performing ablation.
Because of the length and complexity of the procedure, electro-anatomical mapping and ablation are conducted with the patient under general anesthesia.3 The patient is kept supine, and remains so for 2 to 4 hours afterward to allow for hemostasis at puncture sites.3
Patients might be monitored overnight, although same-day catheter ablation has been shown to be safe and cost-effective in select patients.36,37 Post ablation, patients follow up with the cardiologist and electrophysiologist. Long-term arrhythmia monitoring is required.3 Anticoagulation is continued for at least 2 months, and is discontinued based on the patient’s risk for stroke, utilizing their CHA2DS2–VASc score.3,4
CASE
At Mr. Z’s 6-month primary care follow-up, he confirms what has been reported to you as the referring physician: He had a successful catheter ablation and continues to have regular follow-up monitoring with the cardiologist. He is no longer taking amiodarone.
At this visit, he reports no recurrence of AF-associated symptoms or detectable AF on cardiac monitoring. He has lost 8 lbs. You counsel to him to continue to maintain a healthy lifestyle.
CORRESPONDENCE
Amimi S. Osayande MD, FAAFP, Northside-Gwinnett Family Medicine Residency Program, Strickland Family Medicine Center, 665 Duluth Highway, Suite 501, Lawrenceville, GA 30046; amimi.osayande@northside.com
1. Amiodarone hydrochloride (marketed as Cordarone and Pacerone) information. Silver Spring, Md.: US Food & Drug Administration. Reviewed March 23, 2015. Accessed January 16, 2022. www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/amiodarone-hydrochloride-marketed-cordarone-and-pacerone-information
2. Gómez-Outes A, Suárez-Gea ML,García-Pinilla JM. Causes of death in atrial fibrillation: challenges and opportunities. Trends Cardiovasc Med. 2017;27:494-503. doi: 10.1016/j.tcm.2017.05.002
3. Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/ APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: executive summary. J Arrhythm. 2017;33:369-409. doi: 10.1016/j.joa.2017.08.001
4. Camm AJ, Lip GYH, De Caterina R, et al; ESC Committee for Practice Guidelines-CPG; Document Reviewers. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation—developed with the special contribution of the European Heart Rhythm Association. Europace. 2012;14:1385-1413. doi: 10.1093/europace/eus305
5. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: a report of the American College of Cardiology/ American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Circulation. 2014;130:2071-2104. doi: 10.1161/CIR.0000000000000040
6. January CT, Wann LS, Calkins H, et al; Writing Group Members. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/ HRS guideline for the management of patients with atrial fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2019;16:e66-e93. doi: 10.1016/j.hrthm.2019.01.024
7. Kirchhof P, Camm AJ, Goette A, et al; EAST-AFNET 4 Trial Investigators. Early rhythm-control therapy in patients with atrial fibrillation. N Engl J Med. 2020;383:1305-1316. doi: 10.1056/ NEJMoa2019422
8. Packer DL, Mark DB, Robb RA, et al; CABANA Investigators. Effect of catheter ablation vs antiarrhythmic drug therapy on mortality, stroke, bleeding, and cardiac arrest among patients with atrial fibrillation: the CABANA randomized clinical trial. JAMA. 2019;321:1261-1274. doi: 10.1001/jama.2019.0693
9. Valembois L, Audureau E, Takeda A, et al. Antiarrhythmics for maintaining sinus rhythm after cardioversion of atrial fibrillation. Cochrane Database Syst Rev. 2019;9:CD005049. doi: 10.1002/14651858.CD005049
10. Koopman P, Nuyens D, Garweg C, et al. Efficacy of radiofrequency catheter ablation in athletes with atrial fibrillation. Europace. 2011;13:1386-1393. doi: 10.1093/europace/eur142
11. Hakalahti A, Biancari F, Nielsen JC, et al. Radiofrequency ablation vs. antiarrhythmic drug therapy as first line treatment of symptomatic atrial fibrillation: systematic review and meta-analysis. Europace. 2015;17:370-378. doi: 10.1093/europace/euu376
12. Nyong J, Amit G, Adler AJ, et al. Efficacy and safety of ablation for people with non-paroxysmal atrial fibrillation. Cochrane Database Syst Rev. 2016;11:CD012088. doi: 10.1002/14651858. CD012088.pub2
13. Andrade JG, Champagne J, Dubuc M, et al; CIRCA-DOSE Study Investigators. Cryoballoon or radiofrequency ablation for atrial fibrillation assessed by continuous monitoring: a randomized clinical trial. Circulation. 2019;140:1779-1788. doi: 10.1161/ CIRCULATIONAHA.119.042622
14. Asad ZUA, Yousif A, Khan MS, et al. Catheter ablation versus medical therapy for atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Circ Arrhythm Electrophysiol. 2019;12:e007414. doi: 10.1161/ CIRCEP.119.007414
15. Nademanee K, Amnueypol M, Lee F, et al. Benefits and risks of catheter ablation in elderly patients with atrial fibrillation. Heart Rhythm. 2015;12:44-51. doi: 10.1016/j.hrthm.2014.09.049
16. Cheng EP, Liu CF, Yeo I, et al. Risk of mortality following catheter ablation of atrial fibrillation. J Am Coll Cardiol. 2019;74: 2254-2264. doi: 10.1016/j.jacc.2019.08.1036
17. Brugada J, Katritsis DG, Arbelo E, et al; ESC Scientific Document Group. 2019 ESC Guidelines for the management of patients with supraventricular tachycardia. The Task Force for the management of patients with supraventricular tachycardia of the European Society of Cardiology (ESC). Developed in collaboration with the Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2020;41:655-720. doi: 10.1093/eurheartj/ehz467
18. Hosseini SM, Rozen G, Saleh A, et al. Catheter ablation for cardiac arrhythmias: utilization and in-hospital complications, 2000 to 2013. JACC Clin Electrophysiol. 2017;3:1240-1248. doi: 10.1016/j.jacep.2017.05.005
19. Andrade JG, Macle L, Khairy P, et al. Incidence and significance of early recurrences associated with different ablation strategies for AF: a STAR-AF substudy. J Cardiovasc Electrophysiol. 2012;23:1295-1301. doi: 10.1111/j.1540-8167.2012.02399.x
20. Joshi S, Choi AD, Kamath GS, et al. Prevalence, predictors, and prognosis of atrial fibrillation early after pulmonary vein isolation: findings from 3 months of continuous automatic ECG loop recordings. J Cardiovasc Electrophysiol. 2009;20:1089-1094. doi: 10.1111/j.1540-8167.2009.01506.x
21. Weerasooriya R, Khairy P, Litalien J, et al. Catheter ablation for atrial fibrillation: are results maintained at 5 years of follow-up? J Am Coll Cardiol. 2011;57:160-166. doi: 10.1016/j.jacc.2010.05.061
22. Ouyang F, Tilz R, Chun J, et al. Long-term results of catheter ablation in paroxysmal atrial fibrillation: lessons from a 5-year follow-up. Circulation. 2010;122:2368-2377. doi: 10.1161/ CIRCULATIONAHA.110.946806
23. Tilz RR, Rillig A, Thum A-M, et al. Catheter ablation of long-standing persistent atrial fibrillation: 5-year outcomes of the Hamburg Sequential Ablation Strategy. J Am Coll Cardiol. 2012;60: 1921-1929. doi: 10.1016/j.jacc.2012.04.060
24. Forkmann M, Schwab C, Busch S. [Catheter ablation of supraventricular tachycardia]. Herzschrittmacherther Elektrophysiol. 2019;30:336-342. doi: 10.1007/s00399-019-00654-x
25. Bulava A, Hanis J, Eisenberger M. Catheter ablation of atrial fibrillation using zero-fluoroscopy technique: a randomized trial. Pacing Clin Electrophysiol. 2015;38:797-806. doi: 10.1111/pace.12634
26. Haegeli LM, Stutz L, Mohsen M, et al. Feasibility of zero or near zero fluoroscopy during catheter ablation procedures. Cardiol J. 2019;26:226-232. doi: 10.5603/CJ.a2018.0029
27. Steven D, Servatius H, Rostock T, et al. Reduced fluoroscopy during atrial fibrillation ablation: benefits of robotic guided navigation. J Cardiovasc Electrophysiol. 2010;21:6-12. doi: 10.1111/j.1540-8167.2009.01592.x
28. General therapy for cardiac arrhythmias. In: Zipes DP, Libby P, Bonow RO, et al. Braunwald’s Heart Disease: A Textbook of Cardiovascular Medicine. 11th ed. Elsevier; 2019.
29. Kuck K-H, Brugada J, Albenque J-P. Cryoballoon or radiofrequency ablation for atrial fibrillation. N Engl J Med. 2016;375: 1100-1101. doi: 10.1056/NEJMc1609160
30. Chen Y-H, Lu Z-Y, Xiang Y, et al. Cryoablation vs. radiofrequency ablation for treatment of paroxysmal atrial fibrillation: a systematic review and meta-analysis. Europace. 2017;19:784-794. doi: 10.1093/europace/euw330
31. Locati ET, Vecchi AM, Vargiu S, et al. Role of extended external loop recorders for the diagnosis of unexplained syncope, presyncope, and sustained palpitations. Europace. 2014;16:914-922. doi: 10.1093/europace/eut337
32. Calkins H, Kuck KH, Cappato R, et al; Heart Rhythm Society Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. Heart Rhythm. 2012;9:632-696.e21. doi: 10.1016/j.hrthm.2011.12.016
33. Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace. 2016;18:1609-1678. doi: 10.1093/ europace/euw295
34. Nairooz R, Sardar P, Payne J, et al. Meta-analysis of major bleeding with uninterrupted warfarin compared to interrupted warfarin and heparin bridging in ablation of atrial fibrillation. Int J Cardiol. 2015;187:426-429. doi: 10.1016/j.ijcard.2015.03.376
35. Romero J, Cerrud-Rodriguez RC, Diaz JC, et al. Uninterrupted direct oral anticoagulants vs. uninterrupted vitamin K antagonists during catheter ablation of non-valvular atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Europace. 2018;20:1612-1620. doi: 10.1093/europace/euy133
36. Deyell MW, Leather RA, Macle L, et al. Efficacy and safety of same-day discharge for atrial fibrillation ablation. JACC Clin Electrophysiol. 2020;6:609-619. doi: 10.1016/j.jacep.2020.02.009
37. Theodoreson MD, Chohan BC, McAloon CJ, et al. Same-day cardiac catheter ablation is safe and cost-effective: experience from a UK tertiary center. Heart Rhythm. 2015;12:1756-1761. doi: 10.1016/j.hrthm.2015.05.006
1. Amiodarone hydrochloride (marketed as Cordarone and Pacerone) information. Silver Spring, Md.: US Food & Drug Administration. Reviewed March 23, 2015. Accessed January 16, 2022. www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/amiodarone-hydrochloride-marketed-cordarone-and-pacerone-information
2. Gómez-Outes A, Suárez-Gea ML,García-Pinilla JM. Causes of death in atrial fibrillation: challenges and opportunities. Trends Cardiovasc Med. 2017;27:494-503. doi: 10.1016/j.tcm.2017.05.002
3. Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/ APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: executive summary. J Arrhythm. 2017;33:369-409. doi: 10.1016/j.joa.2017.08.001
4. Camm AJ, Lip GYH, De Caterina R, et al; ESC Committee for Practice Guidelines-CPG; Document Reviewers. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation—developed with the special contribution of the European Heart Rhythm Association. Europace. 2012;14:1385-1413. doi: 10.1093/europace/eus305
5. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: a report of the American College of Cardiology/ American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Circulation. 2014;130:2071-2104. doi: 10.1161/CIR.0000000000000040
6. January CT, Wann LS, Calkins H, et al; Writing Group Members. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/ HRS guideline for the management of patients with atrial fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2019;16:e66-e93. doi: 10.1016/j.hrthm.2019.01.024
7. Kirchhof P, Camm AJ, Goette A, et al; EAST-AFNET 4 Trial Investigators. Early rhythm-control therapy in patients with atrial fibrillation. N Engl J Med. 2020;383:1305-1316. doi: 10.1056/ NEJMoa2019422
8. Packer DL, Mark DB, Robb RA, et al; CABANA Investigators. Effect of catheter ablation vs antiarrhythmic drug therapy on mortality, stroke, bleeding, and cardiac arrest among patients with atrial fibrillation: the CABANA randomized clinical trial. JAMA. 2019;321:1261-1274. doi: 10.1001/jama.2019.0693
9. Valembois L, Audureau E, Takeda A, et al. Antiarrhythmics for maintaining sinus rhythm after cardioversion of atrial fibrillation. Cochrane Database Syst Rev. 2019;9:CD005049. doi: 10.1002/14651858.CD005049
10. Koopman P, Nuyens D, Garweg C, et al. Efficacy of radiofrequency catheter ablation in athletes with atrial fibrillation. Europace. 2011;13:1386-1393. doi: 10.1093/europace/eur142
11. Hakalahti A, Biancari F, Nielsen JC, et al. Radiofrequency ablation vs. antiarrhythmic drug therapy as first line treatment of symptomatic atrial fibrillation: systematic review and meta-analysis. Europace. 2015;17:370-378. doi: 10.1093/europace/euu376
12. Nyong J, Amit G, Adler AJ, et al. Efficacy and safety of ablation for people with non-paroxysmal atrial fibrillation. Cochrane Database Syst Rev. 2016;11:CD012088. doi: 10.1002/14651858. CD012088.pub2
13. Andrade JG, Champagne J, Dubuc M, et al; CIRCA-DOSE Study Investigators. Cryoballoon or radiofrequency ablation for atrial fibrillation assessed by continuous monitoring: a randomized clinical trial. Circulation. 2019;140:1779-1788. doi: 10.1161/ CIRCULATIONAHA.119.042622
14. Asad ZUA, Yousif A, Khan MS, et al. Catheter ablation versus medical therapy for atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Circ Arrhythm Electrophysiol. 2019;12:e007414. doi: 10.1161/ CIRCEP.119.007414
15. Nademanee K, Amnueypol M, Lee F, et al. Benefits and risks of catheter ablation in elderly patients with atrial fibrillation. Heart Rhythm. 2015;12:44-51. doi: 10.1016/j.hrthm.2014.09.049
16. Cheng EP, Liu CF, Yeo I, et al. Risk of mortality following catheter ablation of atrial fibrillation. J Am Coll Cardiol. 2019;74: 2254-2264. doi: 10.1016/j.jacc.2019.08.1036
17. Brugada J, Katritsis DG, Arbelo E, et al; ESC Scientific Document Group. 2019 ESC Guidelines for the management of patients with supraventricular tachycardia. The Task Force for the management of patients with supraventricular tachycardia of the European Society of Cardiology (ESC). Developed in collaboration with the Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2020;41:655-720. doi: 10.1093/eurheartj/ehz467
18. Hosseini SM, Rozen G, Saleh A, et al. Catheter ablation for cardiac arrhythmias: utilization and in-hospital complications, 2000 to 2013. JACC Clin Electrophysiol. 2017;3:1240-1248. doi: 10.1016/j.jacep.2017.05.005
19. Andrade JG, Macle L, Khairy P, et al. Incidence and significance of early recurrences associated with different ablation strategies for AF: a STAR-AF substudy. J Cardiovasc Electrophysiol. 2012;23:1295-1301. doi: 10.1111/j.1540-8167.2012.02399.x
20. Joshi S, Choi AD, Kamath GS, et al. Prevalence, predictors, and prognosis of atrial fibrillation early after pulmonary vein isolation: findings from 3 months of continuous automatic ECG loop recordings. J Cardiovasc Electrophysiol. 2009;20:1089-1094. doi: 10.1111/j.1540-8167.2009.01506.x
21. Weerasooriya R, Khairy P, Litalien J, et al. Catheter ablation for atrial fibrillation: are results maintained at 5 years of follow-up? J Am Coll Cardiol. 2011;57:160-166. doi: 10.1016/j.jacc.2010.05.061
22. Ouyang F, Tilz R, Chun J, et al. Long-term results of catheter ablation in paroxysmal atrial fibrillation: lessons from a 5-year follow-up. Circulation. 2010;122:2368-2377. doi: 10.1161/ CIRCULATIONAHA.110.946806
23. Tilz RR, Rillig A, Thum A-M, et al. Catheter ablation of long-standing persistent atrial fibrillation: 5-year outcomes of the Hamburg Sequential Ablation Strategy. J Am Coll Cardiol. 2012;60: 1921-1929. doi: 10.1016/j.jacc.2012.04.060
24. Forkmann M, Schwab C, Busch S. [Catheter ablation of supraventricular tachycardia]. Herzschrittmacherther Elektrophysiol. 2019;30:336-342. doi: 10.1007/s00399-019-00654-x
25. Bulava A, Hanis J, Eisenberger M. Catheter ablation of atrial fibrillation using zero-fluoroscopy technique: a randomized trial. Pacing Clin Electrophysiol. 2015;38:797-806. doi: 10.1111/pace.12634
26. Haegeli LM, Stutz L, Mohsen M, et al. Feasibility of zero or near zero fluoroscopy during catheter ablation procedures. Cardiol J. 2019;26:226-232. doi: 10.5603/CJ.a2018.0029
27. Steven D, Servatius H, Rostock T, et al. Reduced fluoroscopy during atrial fibrillation ablation: benefits of robotic guided navigation. J Cardiovasc Electrophysiol. 2010;21:6-12. doi: 10.1111/j.1540-8167.2009.01592.x
28. General therapy for cardiac arrhythmias. In: Zipes DP, Libby P, Bonow RO, et al. Braunwald’s Heart Disease: A Textbook of Cardiovascular Medicine. 11th ed. Elsevier; 2019.
29. Kuck K-H, Brugada J, Albenque J-P. Cryoballoon or radiofrequency ablation for atrial fibrillation. N Engl J Med. 2016;375: 1100-1101. doi: 10.1056/NEJMc1609160
30. Chen Y-H, Lu Z-Y, Xiang Y, et al. Cryoablation vs. radiofrequency ablation for treatment of paroxysmal atrial fibrillation: a systematic review and meta-analysis. Europace. 2017;19:784-794. doi: 10.1093/europace/euw330
31. Locati ET, Vecchi AM, Vargiu S, et al. Role of extended external loop recorders for the diagnosis of unexplained syncope, presyncope, and sustained palpitations. Europace. 2014;16:914-922. doi: 10.1093/europace/eut337
32. Calkins H, Kuck KH, Cappato R, et al; Heart Rhythm Society Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. Heart Rhythm. 2012;9:632-696.e21. doi: 10.1016/j.hrthm.2011.12.016
33. Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace. 2016;18:1609-1678. doi: 10.1093/ europace/euw295
34. Nairooz R, Sardar P, Payne J, et al. Meta-analysis of major bleeding with uninterrupted warfarin compared to interrupted warfarin and heparin bridging in ablation of atrial fibrillation. Int J Cardiol. 2015;187:426-429. doi: 10.1016/j.ijcard.2015.03.376
35. Romero J, Cerrud-Rodriguez RC, Diaz JC, et al. Uninterrupted direct oral anticoagulants vs. uninterrupted vitamin K antagonists during catheter ablation of non-valvular atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Europace. 2018;20:1612-1620. doi: 10.1093/europace/euy133
36. Deyell MW, Leather RA, Macle L, et al. Efficacy and safety of same-day discharge for atrial fibrillation ablation. JACC Clin Electrophysiol. 2020;6:609-619. doi: 10.1016/j.jacep.2020.02.009
37. Theodoreson MD, Chohan BC, McAloon CJ, et al. Same-day cardiac catheter ablation is safe and cost-effective: experience from a UK tertiary center. Heart Rhythm. 2015;12:1756-1761. doi: 10.1016/j.hrthm.2015.05.006
PRACTICE RECOMMENDATIONS
› Refer patients with atrial fibrillation (AF) to Cardiology for consideration of catheter ablation, a recommended treatment in select cases of (1) symptomatic paroxysmal AF in the setting of intolerance of antiarrhythmic drug therapy and (2) persistence of symptoms despite antiarrhythmic drug therapy. A
› Continue long-term oral anticoagulation therapy post ablation in patients with paroxysmal AF who have undergone catheter ablation if their CHA2DS2–VASc score is ≥ 2 (men) or ≥ 3 (women). C
› Regard catheter ablation as a reasonable alternative to antiarrhythmic drug therapy in select older patients with AF, and refer to a cardiologist as appropriate. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Fewer than half with severe aortic stenosis get new valves
The chance that patients with severe aortic stenosis (AS) will receive aortic valve replacement (AVR) is worse than the flip of a coin, even a decade after the gamechanging transcatheter option became available, a new study suggests.
Of the study’s 6,150 patients with an indication or potential indication for AVR, 48% received the procedure at Massachusetts General Hospital and its partner institution Brigham and Women’s Hospital, both in Boston – both of which have active, high-volume transcatheter and surgical AVR (TAVR/SAVR) programs.
“Essentially, this is a best-case scenario. So, unfortunately, I think on the national level we are likely to see rates that are far worse than what we observed here,” senior author Sammy Elmariah, MD, PhD, Massachusetts General Hospital, told this news organization.
The volume of AVR increased more than 10-fold over the 18-year study period (2000 to 2017), driven by the exponential growth of TAVR, he noted. However, the graying of America led to an even greater increase in the number of patients with severe AS and an indication for AVR.
The study, led by Shawn X. Li, MD, MBA, of Mass General, was published in the March 8 issue of the Journal of the American College of Cardiology.
Previous research has provided equally compelling data on the undertreatment of AS, including a 2021 study using natural language processing (NLP) that found AVR use was just 35.6% within 1 year of diagnosis and varied wildly among managing cardiologists.
The present study used NLP tools to identify symptoms consistent with severe AS in the medical record coupled with echocardiographic data from 10,795 patients with severe AS (valve area <1 cm2). Patients were divided into four AS subtypes and then classified as having a class 1 indication (high-gradient AS with symptoms or reduced ejection fraction [EF]) or a potential class 2a indication (low-gradient AS with symptoms) for AVR.
Among patients with high-gradient AS and class 1 indication for AVR, 1 in 3 did not receive AVR over the study period, including 30% with a normal EF and 47% with a low EF.
In those with low-gradient AS, 67% with a normal EF and 62% with a low EF did not receive AVR. The low-gradient groups were significantly less likely to receive AVR both in the entire study period and in the more contemporary period from 2014 to 2017, despite the valvular heart disease guideline 2014 update indicating AVR was “reasonable” in patients with low-gradient AS – a 2a recommendation upgraded to class 1 in the most recent 2020 update.
Better survival
In patients with a class 1 or potential class 2a indication, AVR was associated with a significantly lower risk of mortality in all four AS subgroups:
- High gradient/normal EF: 3% vs. 15%; adjusted hazard ratio, 0.42
- High-gradient/low EF: 16% vs. 72%; aHR, 0.28
- Low-gradient/normal EF: 5% vs. 14%; aHR, 0.73
- Low-gradient/low EF: 11% vs. 34%; aHR, 0.48; P < .001 for all
“I think what we need to do is change the paradigm, such that patients with a valve area that is less than or equal to 1 [cm2] is severe aortic stenosis until proven otherwise, and that essentially establishes a premise by which we default to treat these patients unless we can prove that it is in fact moderate,” Dr. Elmariah said.
Unfortunately, the opposite is currently true today, he said, and the default is not to treat and put patients through surgery or an invasive TAVR procedure unless physicians can definitively prove that it is severe AS. But they’re not always correct and don’t always have the ability to truly differentiate moderate from severe disease.
“The question, therefore, is ‘What do we do with those patients?’” Dr. Elmariah asked. “I think if a patient has symptoms, then we are obligated to intervene, given the stark difference in mortality that one sees when these patients go undertreated.”
Sounding the alarm
Robert Bonow, MD, a professor of cardiology at Northwestern University in Chicago and a writing committee member for the 2014 guideline update, said the study is a “big wake-up call” and “the take-home message is that we are missing some patients who have treatable aortic stenosis.”
The sheer magnitude of the problem, however, can be difficult to fully ascertain from administrative data like this, he said. Notably, patients who did not receive AVR were significantly older, with 37% aged 81-90 years and 12% over age 90, and had a lower hematocrit and lower estimated glomerular filtration rate. But it’s not clear how many had cancer, end-stage renal disease, or severe lung disease, which could have factored into the decision to undergo AVR.
“What’s also an issue is that over 50% of patients had low gradient disease, which is very problematic and takes careful assessment in an individual patient,” said Dr. Bonow, who is also editor-in-chief of JAMA Cardiology. “That’s all being generated by a low valve area of less than 1 cm2 from echo reports, so that’s not necessarily a careful prospective echo assessment ... so some of the patients with low-gradient disease may not have true severe aortic stenosis.”
Dr. Elmariah agreed that echocardiogram reports are not always clear cut and pointed out that referral to a valve specialist was highly predictive of whether or not a patient underwent AVR, supporting the class 1 guideline recommendation.
He also noted that Mass General is launching the DETECT-AS trial to determine whether electronic physician notifications highlighting clinical practice guideline recommendations will improve AVR utilization over standard care in 940 patients with severe AS on echocardiogram, defined by a valve area less than 1 cm2.
Reached for comment, Catherine Otto, MD, director of the Heart Valve Clinic at the University of Washington, Seattle, and a fellow member of the 2014 guideline writing committee, said “this adds to the data [that] we’re undertreating severe aortic stenosis, and it continues to be surprising given the availability of transcatheter options.”
The biggest challenge is trying to find out why it persists, which is difficult to determine from these data, she said. Whether that’s because the diagnosis is being missed or whether there are barriers to access because cardiologists aren’t understanding the indications or patients aren’t understanding what’s being offered, isn’t clear.
“The other [issue], of course, is are there inappropriate inequities in care? Is it fewer women, age-related, ethnic/racial-related; is it financial? Do people have coverage to get the treatment they need in our country?” Dr. Otto said. “All of those issues are areas that need to be addressed, and I think that is a concern we all have.”
An accompanying editorial points out that the “key lever” in combating undertreatment of AS is getting patients seen by a multidisciplinary heart team and details other possible solutions, such as adding process metrics regarding evaluation and treatment of AS to hospital performance.
“We track quality when AVR is performed (desirable), but how a hospital system performs in getting individuals treated who would benefit from AVR remains a complete blind spot,” write Brian Lindman, MD, MSc, and Angela Lowenstern, MD, MHS, both of Vanderbilt University Medical Center, Nashville, Tenn.
“Is it appropriate to consider the hospital ‘high performing’ when data from Li et al. show a 2-year absolute mortality difference from 9% to 56% based on treatment versus nontreatment with AVR for various AS patient subgroups?” they add.
Dr. Lindman and Dr. Lowenstern observe that having a 50% utilization rate for an effective therapy for a deadly cancer or stenting of ST-segment elevation myocardial infarction (STEMI) would generate negative headlines and a collective commitment to swift action by multiple stakeholders to address what would be “incontrovertibly unacceptable.”
“In one of America’s leading health care systems, there was evidence of an overwhelming reduction in the risk of death with AVR in all AS subgroups examined, but <50% of patients with AS with an indication or potential indication for AVR were treated with an AVR. Let that set in; hear and internalize the alarm. The status quo is unacceptable. What will you do? What will we do?” they conclude.
The study was funded by Edwards Lifesciences. Dr. Elmariah has received research grants from the American Heart Association, National Institutes of Health, Edwards Lifesciences, Svelte Medical, Abbott Vascular, and Medtronic, and has received consulting fees from Edwards Lifesciences. Dr. Bonow and Dr. Otto have disclosed no relevant financial relationships. Dr. Lindman has received investigator-initiated research grants from Edwards. Dr. Lowenstern has received consulting fees from Edwards.
A version of this article first appeared on Medscape.com.
The chance that patients with severe aortic stenosis (AS) will receive aortic valve replacement (AVR) is worse than the flip of a coin, even a decade after the gamechanging transcatheter option became available, a new study suggests.
Of the study’s 6,150 patients with an indication or potential indication for AVR, 48% received the procedure at Massachusetts General Hospital and its partner institution Brigham and Women’s Hospital, both in Boston – both of which have active, high-volume transcatheter and surgical AVR (TAVR/SAVR) programs.
“Essentially, this is a best-case scenario. So, unfortunately, I think on the national level we are likely to see rates that are far worse than what we observed here,” senior author Sammy Elmariah, MD, PhD, Massachusetts General Hospital, told this news organization.
The volume of AVR increased more than 10-fold over the 18-year study period (2000 to 2017), driven by the exponential growth of TAVR, he noted. However, the graying of America led to an even greater increase in the number of patients with severe AS and an indication for AVR.
The study, led by Shawn X. Li, MD, MBA, of Mass General, was published in the March 8 issue of the Journal of the American College of Cardiology.
Previous research has provided equally compelling data on the undertreatment of AS, including a 2021 study using natural language processing (NLP) that found AVR use was just 35.6% within 1 year of diagnosis and varied wildly among managing cardiologists.
The present study used NLP tools to identify symptoms consistent with severe AS in the medical record coupled with echocardiographic data from 10,795 patients with severe AS (valve area <1 cm2). Patients were divided into four AS subtypes and then classified as having a class 1 indication (high-gradient AS with symptoms or reduced ejection fraction [EF]) or a potential class 2a indication (low-gradient AS with symptoms) for AVR.
Among patients with high-gradient AS and class 1 indication for AVR, 1 in 3 did not receive AVR over the study period, including 30% with a normal EF and 47% with a low EF.
In those with low-gradient AS, 67% with a normal EF and 62% with a low EF did not receive AVR. The low-gradient groups were significantly less likely to receive AVR both in the entire study period and in the more contemporary period from 2014 to 2017, despite the valvular heart disease guideline 2014 update indicating AVR was “reasonable” in patients with low-gradient AS – a 2a recommendation upgraded to class 1 in the most recent 2020 update.
Better survival
In patients with a class 1 or potential class 2a indication, AVR was associated with a significantly lower risk of mortality in all four AS subgroups:
- High gradient/normal EF: 3% vs. 15%; adjusted hazard ratio, 0.42
- High-gradient/low EF: 16% vs. 72%; aHR, 0.28
- Low-gradient/normal EF: 5% vs. 14%; aHR, 0.73
- Low-gradient/low EF: 11% vs. 34%; aHR, 0.48; P < .001 for all
“I think what we need to do is change the paradigm, such that patients with a valve area that is less than or equal to 1 [cm2] is severe aortic stenosis until proven otherwise, and that essentially establishes a premise by which we default to treat these patients unless we can prove that it is in fact moderate,” Dr. Elmariah said.
Unfortunately, the opposite is currently true today, he said, and the default is not to treat and put patients through surgery or an invasive TAVR procedure unless physicians can definitively prove that it is severe AS. But they’re not always correct and don’t always have the ability to truly differentiate moderate from severe disease.
“The question, therefore, is ‘What do we do with those patients?’” Dr. Elmariah asked. “I think if a patient has symptoms, then we are obligated to intervene, given the stark difference in mortality that one sees when these patients go undertreated.”
Sounding the alarm
Robert Bonow, MD, a professor of cardiology at Northwestern University in Chicago and a writing committee member for the 2014 guideline update, said the study is a “big wake-up call” and “the take-home message is that we are missing some patients who have treatable aortic stenosis.”
The sheer magnitude of the problem, however, can be difficult to fully ascertain from administrative data like this, he said. Notably, patients who did not receive AVR were significantly older, with 37% aged 81-90 years and 12% over age 90, and had a lower hematocrit and lower estimated glomerular filtration rate. But it’s not clear how many had cancer, end-stage renal disease, or severe lung disease, which could have factored into the decision to undergo AVR.
“What’s also an issue is that over 50% of patients had low gradient disease, which is very problematic and takes careful assessment in an individual patient,” said Dr. Bonow, who is also editor-in-chief of JAMA Cardiology. “That’s all being generated by a low valve area of less than 1 cm2 from echo reports, so that’s not necessarily a careful prospective echo assessment ... so some of the patients with low-gradient disease may not have true severe aortic stenosis.”
Dr. Elmariah agreed that echocardiogram reports are not always clear cut and pointed out that referral to a valve specialist was highly predictive of whether or not a patient underwent AVR, supporting the class 1 guideline recommendation.
He also noted that Mass General is launching the DETECT-AS trial to determine whether electronic physician notifications highlighting clinical practice guideline recommendations will improve AVR utilization over standard care in 940 patients with severe AS on echocardiogram, defined by a valve area less than 1 cm2.
Reached for comment, Catherine Otto, MD, director of the Heart Valve Clinic at the University of Washington, Seattle, and a fellow member of the 2014 guideline writing committee, said “this adds to the data [that] we’re undertreating severe aortic stenosis, and it continues to be surprising given the availability of transcatheter options.”
The biggest challenge is trying to find out why it persists, which is difficult to determine from these data, she said. Whether that’s because the diagnosis is being missed or whether there are barriers to access because cardiologists aren’t understanding the indications or patients aren’t understanding what’s being offered, isn’t clear.
“The other [issue], of course, is are there inappropriate inequities in care? Is it fewer women, age-related, ethnic/racial-related; is it financial? Do people have coverage to get the treatment they need in our country?” Dr. Otto said. “All of those issues are areas that need to be addressed, and I think that is a concern we all have.”
An accompanying editorial points out that the “key lever” in combating undertreatment of AS is getting patients seen by a multidisciplinary heart team and details other possible solutions, such as adding process metrics regarding evaluation and treatment of AS to hospital performance.
“We track quality when AVR is performed (desirable), but how a hospital system performs in getting individuals treated who would benefit from AVR remains a complete blind spot,” write Brian Lindman, MD, MSc, and Angela Lowenstern, MD, MHS, both of Vanderbilt University Medical Center, Nashville, Tenn.
“Is it appropriate to consider the hospital ‘high performing’ when data from Li et al. show a 2-year absolute mortality difference from 9% to 56% based on treatment versus nontreatment with AVR for various AS patient subgroups?” they add.
Dr. Lindman and Dr. Lowenstern observe that having a 50% utilization rate for an effective therapy for a deadly cancer or stenting of ST-segment elevation myocardial infarction (STEMI) would generate negative headlines and a collective commitment to swift action by multiple stakeholders to address what would be “incontrovertibly unacceptable.”
“In one of America’s leading health care systems, there was evidence of an overwhelming reduction in the risk of death with AVR in all AS subgroups examined, but <50% of patients with AS with an indication or potential indication for AVR were treated with an AVR. Let that set in; hear and internalize the alarm. The status quo is unacceptable. What will you do? What will we do?” they conclude.
The study was funded by Edwards Lifesciences. Dr. Elmariah has received research grants from the American Heart Association, National Institutes of Health, Edwards Lifesciences, Svelte Medical, Abbott Vascular, and Medtronic, and has received consulting fees from Edwards Lifesciences. Dr. Bonow and Dr. Otto have disclosed no relevant financial relationships. Dr. Lindman has received investigator-initiated research grants from Edwards. Dr. Lowenstern has received consulting fees from Edwards.
A version of this article first appeared on Medscape.com.
The chance that patients with severe aortic stenosis (AS) will receive aortic valve replacement (AVR) is worse than the flip of a coin, even a decade after the gamechanging transcatheter option became available, a new study suggests.
Of the study’s 6,150 patients with an indication or potential indication for AVR, 48% received the procedure at Massachusetts General Hospital and its partner institution Brigham and Women’s Hospital, both in Boston – both of which have active, high-volume transcatheter and surgical AVR (TAVR/SAVR) programs.
“Essentially, this is a best-case scenario. So, unfortunately, I think on the national level we are likely to see rates that are far worse than what we observed here,” senior author Sammy Elmariah, MD, PhD, Massachusetts General Hospital, told this news organization.
The volume of AVR increased more than 10-fold over the 18-year study period (2000 to 2017), driven by the exponential growth of TAVR, he noted. However, the graying of America led to an even greater increase in the number of patients with severe AS and an indication for AVR.
The study, led by Shawn X. Li, MD, MBA, of Mass General, was published in the March 8 issue of the Journal of the American College of Cardiology.
Previous research has provided equally compelling data on the undertreatment of AS, including a 2021 study using natural language processing (NLP) that found AVR use was just 35.6% within 1 year of diagnosis and varied wildly among managing cardiologists.
The present study used NLP tools to identify symptoms consistent with severe AS in the medical record coupled with echocardiographic data from 10,795 patients with severe AS (valve area <1 cm2). Patients were divided into four AS subtypes and then classified as having a class 1 indication (high-gradient AS with symptoms or reduced ejection fraction [EF]) or a potential class 2a indication (low-gradient AS with symptoms) for AVR.
Among patients with high-gradient AS and class 1 indication for AVR, 1 in 3 did not receive AVR over the study period, including 30% with a normal EF and 47% with a low EF.
In those with low-gradient AS, 67% with a normal EF and 62% with a low EF did not receive AVR. The low-gradient groups were significantly less likely to receive AVR both in the entire study period and in the more contemporary period from 2014 to 2017, despite the valvular heart disease guideline 2014 update indicating AVR was “reasonable” in patients with low-gradient AS – a 2a recommendation upgraded to class 1 in the most recent 2020 update.
Better survival
In patients with a class 1 or potential class 2a indication, AVR was associated with a significantly lower risk of mortality in all four AS subgroups:
- High gradient/normal EF: 3% vs. 15%; adjusted hazard ratio, 0.42
- High-gradient/low EF: 16% vs. 72%; aHR, 0.28
- Low-gradient/normal EF: 5% vs. 14%; aHR, 0.73
- Low-gradient/low EF: 11% vs. 34%; aHR, 0.48; P < .001 for all
“I think what we need to do is change the paradigm, such that patients with a valve area that is less than or equal to 1 [cm2] is severe aortic stenosis until proven otherwise, and that essentially establishes a premise by which we default to treat these patients unless we can prove that it is in fact moderate,” Dr. Elmariah said.
Unfortunately, the opposite is currently true today, he said, and the default is not to treat and put patients through surgery or an invasive TAVR procedure unless physicians can definitively prove that it is severe AS. But they’re not always correct and don’t always have the ability to truly differentiate moderate from severe disease.
“The question, therefore, is ‘What do we do with those patients?’” Dr. Elmariah asked. “I think if a patient has symptoms, then we are obligated to intervene, given the stark difference in mortality that one sees when these patients go undertreated.”
Sounding the alarm
Robert Bonow, MD, a professor of cardiology at Northwestern University in Chicago and a writing committee member for the 2014 guideline update, said the study is a “big wake-up call” and “the take-home message is that we are missing some patients who have treatable aortic stenosis.”
The sheer magnitude of the problem, however, can be difficult to fully ascertain from administrative data like this, he said. Notably, patients who did not receive AVR were significantly older, with 37% aged 81-90 years and 12% over age 90, and had a lower hematocrit and lower estimated glomerular filtration rate. But it’s not clear how many had cancer, end-stage renal disease, or severe lung disease, which could have factored into the decision to undergo AVR.
“What’s also an issue is that over 50% of patients had low gradient disease, which is very problematic and takes careful assessment in an individual patient,” said Dr. Bonow, who is also editor-in-chief of JAMA Cardiology. “That’s all being generated by a low valve area of less than 1 cm2 from echo reports, so that’s not necessarily a careful prospective echo assessment ... so some of the patients with low-gradient disease may not have true severe aortic stenosis.”
Dr. Elmariah agreed that echocardiogram reports are not always clear cut and pointed out that referral to a valve specialist was highly predictive of whether or not a patient underwent AVR, supporting the class 1 guideline recommendation.
He also noted that Mass General is launching the DETECT-AS trial to determine whether electronic physician notifications highlighting clinical practice guideline recommendations will improve AVR utilization over standard care in 940 patients with severe AS on echocardiogram, defined by a valve area less than 1 cm2.
Reached for comment, Catherine Otto, MD, director of the Heart Valve Clinic at the University of Washington, Seattle, and a fellow member of the 2014 guideline writing committee, said “this adds to the data [that] we’re undertreating severe aortic stenosis, and it continues to be surprising given the availability of transcatheter options.”
The biggest challenge is trying to find out why it persists, which is difficult to determine from these data, she said. Whether that’s because the diagnosis is being missed or whether there are barriers to access because cardiologists aren’t understanding the indications or patients aren’t understanding what’s being offered, isn’t clear.
“The other [issue], of course, is are there inappropriate inequities in care? Is it fewer women, age-related, ethnic/racial-related; is it financial? Do people have coverage to get the treatment they need in our country?” Dr. Otto said. “All of those issues are areas that need to be addressed, and I think that is a concern we all have.”
An accompanying editorial points out that the “key lever” in combating undertreatment of AS is getting patients seen by a multidisciplinary heart team and details other possible solutions, such as adding process metrics regarding evaluation and treatment of AS to hospital performance.
“We track quality when AVR is performed (desirable), but how a hospital system performs in getting individuals treated who would benefit from AVR remains a complete blind spot,” write Brian Lindman, MD, MSc, and Angela Lowenstern, MD, MHS, both of Vanderbilt University Medical Center, Nashville, Tenn.
“Is it appropriate to consider the hospital ‘high performing’ when data from Li et al. show a 2-year absolute mortality difference from 9% to 56% based on treatment versus nontreatment with AVR for various AS patient subgroups?” they add.
Dr. Lindman and Dr. Lowenstern observe that having a 50% utilization rate for an effective therapy for a deadly cancer or stenting of ST-segment elevation myocardial infarction (STEMI) would generate negative headlines and a collective commitment to swift action by multiple stakeholders to address what would be “incontrovertibly unacceptable.”
“In one of America’s leading health care systems, there was evidence of an overwhelming reduction in the risk of death with AVR in all AS subgroups examined, but <50% of patients with AS with an indication or potential indication for AVR were treated with an AVR. Let that set in; hear and internalize the alarm. The status quo is unacceptable. What will you do? What will we do?” they conclude.
The study was funded by Edwards Lifesciences. Dr. Elmariah has received research grants from the American Heart Association, National Institutes of Health, Edwards Lifesciences, Svelte Medical, Abbott Vascular, and Medtronic, and has received consulting fees from Edwards Lifesciences. Dr. Bonow and Dr. Otto have disclosed no relevant financial relationships. Dr. Lindman has received investigator-initiated research grants from Edwards. Dr. Lowenstern has received consulting fees from Edwards.
A version of this article first appeared on Medscape.com.
Boosting daily exercise after age 70 tied to lower CVD risk
Increasingly active patterns of physical activity were linked with reduced rates of overall mortality and cardiovascular disease (CVD), but early rather than later in late life, in a 20-year follow-up cohort study.
In this population of people older than 65 years, researchers found that physical activity overall was associated with lower rates of incident CVD, particularly among men, and the association was strongest in people 70 to 75 years of age, rather than in older age groups.
They also looked at “trajectories,” or changes in activity over time, and found that a stable-high trajectory of activity was associated with a significantly lower risk for cardiovascular outcomes in men than in those with a stable-low trajectory. For women, more physical activity was consistently associated with lower CVD outcomes, although not statistically significantly so, except for overall mortality, which did reach significance.
Notably, the greatest reduction in cardiovascular risk was reported in people who did more than 20 minutes of physical exercise each day, and it was more pronounced in those 70 years of age.
Physical activity was also associated with a lower incidence of heart failure and coronary heart disease in older people, again especially early on in late life, reported Claudio Barbiellini Amidei, MD, University of Padua, Italy, and colleagues.
The data suggest that physical activity is more effective in preventing CVD onset when implemented early rather than later in life, noted Dr. Amidei in an email.
“The findings of our study are suggestive of a protective effect of physical activity in late-life on cardiovascular health. WHO recommendations for adults and older adults are to practice at least 20 minutes of moderate to vigorous physical activity per day. I believe this is a realistic target, and policy makers should raise awareness on the importance of achieving this goal at all ages, including in late-life,” Dr. Amidei said.
The study was published online Feb. 14 in Heart.
Previous research has demonstrated that the most benefit of high physical activity, compared with low, begins at about 60 years of age, and that is because younger people are at much lower risk, noted Carl “Chip” Lavie MD, FACC, medical director of cardiac rehabilitation and prevention, Ochsner Clinical School–The University of Queensland School of Medicine, New Orleans, who was not involved in the study.
“At quite old ages, for example over age 80, resistance exercise or weight training and balance training may be even more important than aerobic training,” he added.
Activity ‘trajectories’
The benefits of physical activity on cardiovascular risk are well established, the researchers note. Less clear is the role that trajectories of activity over time play, although research to date suggests a reduction in risk with increasing activity from mid-life to early old age, they write.
For the current analysis, the researchers assessed 3,099 Italian participants. Mean age was about 75 years, and baseline data were collected from 1995 to 1997.
Follow-up visits were conducted after 4 years and again after 7 years. Using hospital medical records and mortality data, the researchers were able to collect surveillance data through 2018. Hospital records, surveys, and clinical assessments helped them identify incident and prevalent cardiovascular diseases, such as stroke, coronary heart disease, and heart failure.
Participants’ physical activity patterns were classified as stable-high, low-increasing, high-decreasing, and stable-low. Exposure was evaluated at 70, 75, 80, and 85 years of age.
“In our analyses, we focused on moderate to vigorous physical activity, and these include a broad range of exercises, such as walking very briskly, playing tennis, [and] jogging, but comprise also other activities, such as gardening or doing household chores,” said Dr. Amidei.
Patterns of stable-low physical activity were linked to a significantly greater risk for cardiovascular outcomes in men than patterns of stable-high physical activity (hazard ratio, 0.48; 95% confidence interval, 0.27-0.86; P for trend = .002).
No significant relation was found between physical activity and stroke, the researchers note.
“The benefits of physical activity seem to lessen above the age of 75 years and seem more important in men,” noted Dr. Lavie. “This may be partly due to the higher risk of CVD in men. Women typically lag 13 to 15 years behind men for CVD but start catching up in older years.”
Limitations of the study include lack of information regarding physical activity during mid-life, the limited number of stroke events, the relatively few participants older than 85 years, and potential recall bias, the researchers note.
Another limitation was that the physical activity data were based on patient surveys collected 3 years apart and did not involve the use of an accelerometer, the researchers add.
“Future observational studies are required to confirm our findings and pathophysiological studies are warranted to examine the underlying biological mechanisms. Physical activity is likely to be beneficial at any age, but to summarize our findings, we could say that when it comes to being physically active, the sooner the better,” concluded Dr. Amidei.
Dr. Amidei reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Increasingly active patterns of physical activity were linked with reduced rates of overall mortality and cardiovascular disease (CVD), but early rather than later in late life, in a 20-year follow-up cohort study.
In this population of people older than 65 years, researchers found that physical activity overall was associated with lower rates of incident CVD, particularly among men, and the association was strongest in people 70 to 75 years of age, rather than in older age groups.
They also looked at “trajectories,” or changes in activity over time, and found that a stable-high trajectory of activity was associated with a significantly lower risk for cardiovascular outcomes in men than in those with a stable-low trajectory. For women, more physical activity was consistently associated with lower CVD outcomes, although not statistically significantly so, except for overall mortality, which did reach significance.
Notably, the greatest reduction in cardiovascular risk was reported in people who did more than 20 minutes of physical exercise each day, and it was more pronounced in those 70 years of age.
Physical activity was also associated with a lower incidence of heart failure and coronary heart disease in older people, again especially early on in late life, reported Claudio Barbiellini Amidei, MD, University of Padua, Italy, and colleagues.
The data suggest that physical activity is more effective in preventing CVD onset when implemented early rather than later in life, noted Dr. Amidei in an email.
“The findings of our study are suggestive of a protective effect of physical activity in late-life on cardiovascular health. WHO recommendations for adults and older adults are to practice at least 20 minutes of moderate to vigorous physical activity per day. I believe this is a realistic target, and policy makers should raise awareness on the importance of achieving this goal at all ages, including in late-life,” Dr. Amidei said.
The study was published online Feb. 14 in Heart.
Previous research has demonstrated that the most benefit of high physical activity, compared with low, begins at about 60 years of age, and that is because younger people are at much lower risk, noted Carl “Chip” Lavie MD, FACC, medical director of cardiac rehabilitation and prevention, Ochsner Clinical School–The University of Queensland School of Medicine, New Orleans, who was not involved in the study.
“At quite old ages, for example over age 80, resistance exercise or weight training and balance training may be even more important than aerobic training,” he added.
Activity ‘trajectories’
The benefits of physical activity on cardiovascular risk are well established, the researchers note. Less clear is the role that trajectories of activity over time play, although research to date suggests a reduction in risk with increasing activity from mid-life to early old age, they write.
For the current analysis, the researchers assessed 3,099 Italian participants. Mean age was about 75 years, and baseline data were collected from 1995 to 1997.
Follow-up visits were conducted after 4 years and again after 7 years. Using hospital medical records and mortality data, the researchers were able to collect surveillance data through 2018. Hospital records, surveys, and clinical assessments helped them identify incident and prevalent cardiovascular diseases, such as stroke, coronary heart disease, and heart failure.
Participants’ physical activity patterns were classified as stable-high, low-increasing, high-decreasing, and stable-low. Exposure was evaluated at 70, 75, 80, and 85 years of age.
“In our analyses, we focused on moderate to vigorous physical activity, and these include a broad range of exercises, such as walking very briskly, playing tennis, [and] jogging, but comprise also other activities, such as gardening or doing household chores,” said Dr. Amidei.
Patterns of stable-low physical activity were linked to a significantly greater risk for cardiovascular outcomes in men than patterns of stable-high physical activity (hazard ratio, 0.48; 95% confidence interval, 0.27-0.86; P for trend = .002).
No significant relation was found between physical activity and stroke, the researchers note.
“The benefits of physical activity seem to lessen above the age of 75 years and seem more important in men,” noted Dr. Lavie. “This may be partly due to the higher risk of CVD in men. Women typically lag 13 to 15 years behind men for CVD but start catching up in older years.”
Limitations of the study include lack of information regarding physical activity during mid-life, the limited number of stroke events, the relatively few participants older than 85 years, and potential recall bias, the researchers note.
Another limitation was that the physical activity data were based on patient surveys collected 3 years apart and did not involve the use of an accelerometer, the researchers add.
“Future observational studies are required to confirm our findings and pathophysiological studies are warranted to examine the underlying biological mechanisms. Physical activity is likely to be beneficial at any age, but to summarize our findings, we could say that when it comes to being physically active, the sooner the better,” concluded Dr. Amidei.
Dr. Amidei reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Increasingly active patterns of physical activity were linked with reduced rates of overall mortality and cardiovascular disease (CVD), but early rather than later in late life, in a 20-year follow-up cohort study.
In this population of people older than 65 years, researchers found that physical activity overall was associated with lower rates of incident CVD, particularly among men, and the association was strongest in people 70 to 75 years of age, rather than in older age groups.
They also looked at “trajectories,” or changes in activity over time, and found that a stable-high trajectory of activity was associated with a significantly lower risk for cardiovascular outcomes in men than in those with a stable-low trajectory. For women, more physical activity was consistently associated with lower CVD outcomes, although not statistically significantly so, except for overall mortality, which did reach significance.
Notably, the greatest reduction in cardiovascular risk was reported in people who did more than 20 minutes of physical exercise each day, and it was more pronounced in those 70 years of age.
Physical activity was also associated with a lower incidence of heart failure and coronary heart disease in older people, again especially early on in late life, reported Claudio Barbiellini Amidei, MD, University of Padua, Italy, and colleagues.
The data suggest that physical activity is more effective in preventing CVD onset when implemented early rather than later in life, noted Dr. Amidei in an email.
“The findings of our study are suggestive of a protective effect of physical activity in late-life on cardiovascular health. WHO recommendations for adults and older adults are to practice at least 20 minutes of moderate to vigorous physical activity per day. I believe this is a realistic target, and policy makers should raise awareness on the importance of achieving this goal at all ages, including in late-life,” Dr. Amidei said.
The study was published online Feb. 14 in Heart.
Previous research has demonstrated that the most benefit of high physical activity, compared with low, begins at about 60 years of age, and that is because younger people are at much lower risk, noted Carl “Chip” Lavie MD, FACC, medical director of cardiac rehabilitation and prevention, Ochsner Clinical School–The University of Queensland School of Medicine, New Orleans, who was not involved in the study.
“At quite old ages, for example over age 80, resistance exercise or weight training and balance training may be even more important than aerobic training,” he added.
Activity ‘trajectories’
The benefits of physical activity on cardiovascular risk are well established, the researchers note. Less clear is the role that trajectories of activity over time play, although research to date suggests a reduction in risk with increasing activity from mid-life to early old age, they write.
For the current analysis, the researchers assessed 3,099 Italian participants. Mean age was about 75 years, and baseline data were collected from 1995 to 1997.
Follow-up visits were conducted after 4 years and again after 7 years. Using hospital medical records and mortality data, the researchers were able to collect surveillance data through 2018. Hospital records, surveys, and clinical assessments helped them identify incident and prevalent cardiovascular diseases, such as stroke, coronary heart disease, and heart failure.
Participants’ physical activity patterns were classified as stable-high, low-increasing, high-decreasing, and stable-low. Exposure was evaluated at 70, 75, 80, and 85 years of age.
“In our analyses, we focused on moderate to vigorous physical activity, and these include a broad range of exercises, such as walking very briskly, playing tennis, [and] jogging, but comprise also other activities, such as gardening or doing household chores,” said Dr. Amidei.
Patterns of stable-low physical activity were linked to a significantly greater risk for cardiovascular outcomes in men than patterns of stable-high physical activity (hazard ratio, 0.48; 95% confidence interval, 0.27-0.86; P for trend = .002).
No significant relation was found between physical activity and stroke, the researchers note.
“The benefits of physical activity seem to lessen above the age of 75 years and seem more important in men,” noted Dr. Lavie. “This may be partly due to the higher risk of CVD in men. Women typically lag 13 to 15 years behind men for CVD but start catching up in older years.”
Limitations of the study include lack of information regarding physical activity during mid-life, the limited number of stroke events, the relatively few participants older than 85 years, and potential recall bias, the researchers note.
Another limitation was that the physical activity data were based on patient surveys collected 3 years apart and did not involve the use of an accelerometer, the researchers add.
“Future observational studies are required to confirm our findings and pathophysiological studies are warranted to examine the underlying biological mechanisms. Physical activity is likely to be beneficial at any age, but to summarize our findings, we could say that when it comes to being physically active, the sooner the better,” concluded Dr. Amidei.
Dr. Amidei reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
3-year-old girl • fever • cervical lymphadenopathy • leukocytosis • Dx?
THE CASE
A previously healthy 3-year-old girl presented to the emergency department with 4 days of fever and 2 days of right-side neck pain. The maximum temperature at home was 103 °F. The patient was irritable and vomited once. There were no other apparent or reported symptoms.
The neck exam was notable for nonfluctuant, swollen, and tender lymph nodes on the right side. Her sclera and conjunctiva were clear, and her oropharynx was unremarkable. Lab work revealed leukocytosis, with a white blood cell (WBC) count of 15.5 × 103/µL (normal range, 4.0-10.0 × 103/µL). She was given one 20 cc/kg normal saline bolus, started on intravenous clindamycin for presumed cervical lymphadenitis, and admitted to the hospital.
On Day 2, the patient developed a fine maculopapular rash on her chest, abdomen, and back. She had spiking fevers—as high as 102.2 °F—despite being on antibiotics for more than 24 hours. The erythrocyte sedimentation rate (ESR) was 39 mm/h (0-20 mm/h), and C-reactive protein (CRP) was 71.4 mg/L (0.0-4.9 mg/L). Due to concern for abscess, a neck ultrasound was performed; it showed a chain of enlarged lymph nodes in the right neck (largest, 2.3 × 1.1 × 1.4 cm) and no abscess.
On Day 3, clindamycin was switched to intravenous ampicillin/sulbactam because a nasal swab for methicillin-resistant Staphylococcus aureus was negative. A swab for respiratory viral infections was also negative. The patient then developed notable facial swelling, bilateral bulbar conjunctival injection with limbic sparing, and swelling of her hands and feet.
THE DIAGNOSIS
By the end of Day 3, the patient’s lab studies demonstrated microcytic anemia and low albumin (2.5 g/dL), but no transaminitis, thrombocytosis, or sterile pyuria. An electrocardiogram was unremarkable. A pediatric echocardiogram revealed hyperemic coronaries, indicating inflammation. The coronary arteries were measured in the upper limits of normal, and the patient’s Z-scores were < 2.5. (A Z-score < 2 indicates no involvement, 2 to < 2.5 indicates dilation, and ≥ 2.5 indicates aneurysm abnormality.1) An ultrasound of the right upper quadrant revealed an enlarged/elongated gallbladder. The patient therefore met clinical criteria for Kawasaki disease.
DISCUSSION
Kawasaki disease is a self-limited vasculitis of childhood and the leading cause of acquired heart disease in children in developed countries.1 The annual incidence of Kawasaki disease in North America is about 25 cases per 100,000 children < 5 years of age.1 In the United States, incidence is highest in Asian and Pacific Islander populations (30 per 100,000) and is particularly high among those of Japanese ancestry (~200 per 100,000).2 Disease prevalence is also noteworthy in Non-Hispanic African American (17 per 100,000) and Hispanic (16 per 100,000) populations.2
Diagnosis of Kawasaki disease requires presence of fever lasting at least 5 days and at least 4 of the following: bilateral bulbar conjunctival injection, oral mucous membrane changes (erythematous or cracked lips, erythematous pharynx, strawberry tongue), peripheral extremity changes (erythema of palms or soles, edema of hands or feet, and/or periungual desquamation), diffuse maculopapular rash, and cervical lymphadenopathy (≥ 1.5 cm, often unilateral). If ≥ 4 criteria are met, Kawasaki disease can be diagnosed on the fourth day of illness.1
Continue to: Laboratory findings suggesting...
Laboratory findings suggesting Kawasaki disease include a WBC count ≥ 15,000/mcL, normocytic, normochromic anemia, platelets ≥ 450,000/mcL after 7 days of illness, sterile pyuria (≥ 10 WBCs/high-power field), serum alanine aminotransferase level > 50 U/L, and serum albumin ≤ 3 g/dL.
Cardiac abnormalities are not included in the diagnostic criteria for Kawasaki disease but provide evidence in cases of incomplete Kawasaki disease if ≥ 4 criteria are not met and there is strong clinical suspicion.1 Incomplete Kawasaki disease should be considered in a patient with a CRP level ≥ 3 mg/dL and/or ESR ≥ 40 mm/h, ≥ 3 supplemental laboratory criteria, or a positive echocardiogram.1
Ultrasound imaging may reveal cervical lymph nodes resembling a “cluster of grapes.”3 The case patient’s imaging showed a “chain of enlarged lymph nodes.” She likely had gallbladder “hydrops” due to its increased longitudinal and horizontal diameter and lack of other anatomic changes.4
Prompt treatment is essential
Treatment for complete and incomplete Kawasaki disease is a single high dose of intravenous immunoglobulin (IVIG) along with aspirin. Patients meeting criteria should be treated as soon as the diagnosis is established.5 A single high dose of IVIG (2 g/kg), administered over 10 to 12 hours, should be initiated within 5 to 10 days of disease onset. Administering IVIG in the acute phase of Kawasaki disease reduces the prevalence of coronary artery abnormalities.6 Corticosteroids may be used as adjunctive therapy for patients with high risk of IVIG resistance.1,7-9
Our patient was not deemed to be at high risk for IVIG resistance (Non-Japanese patient, age at fever onset > 6 months, absence of coronary artery aneurysm9) and was administered IVIG on Day 4. She was also given moderate-dose aspirin, then later transitioned to low-dose aspirin. The patient’s fevers improved, she was less irritable, and she had periods of playfulness. Physical exam then showed erythematous and cracked lips with peeling skin.
Continue to: The patient was discharged...
The patient was discharged home on Day 8, after her fever resolved, with instructions to continue low-dose aspirin and to obtain a repeat echocardiogram, gallbladder ultrasound, and lab work in 2 weeks. At her follow-up appointment, periungual desquamation was noted, and ultrasound showed continued enlarged/elongated gallbladder. A repeat echocardiogram was not available. Overall, the patient recovered from Kawasaki disease after therapeutic intervention.
THE TAKEAWAY
Kawasaki disease can be relatively rare in North American populations, but it is important for physicians to be able to recognize and treat it. Untreated children have a 25% chance of developing coronary artery aneurysms.1,10,11 Early treatment with IVIG can decrease risk to 5%, resulting in an excellent medium- to long-term prognosis for patients.10 Thorough physical examination and an appropriate degree of clinical suspicion was key in this case of Kawasaki disease.
Taisha Doo, MD, 1401 Madison Street, Suite #100, Seattle, WA 98104; taisha.doo@swedish.org
1. McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. 2017;135:e927-e999. doi: 10.1161/CIR.0000000000000484
2. Holman RC, Belay ED, Christensen KY, et al. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis. 2010;29:483-488. doi: 10.1097/INF.0b013e3181cf8705
3. Tashiro N, Matsubara T, Uchida M, et al. Ultrasonographic evaluation of cervical lymph nodes in Kawasaki disease. Pediatrics. 2002;109:e77. doi: 10.1542/peds.109.5.e77
4. Chen CJ, Huang FC, Taio MM, et al. Sonographic gallbladder abnormality is associated with intravenous immunoglobulin resistance in Kawasaki disease. Scientific World J. 2012;2012:485758. doi: 10.1100/2012/485758
5. Dominguez SR, Anderson MS, El-Adawy M, et al. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012;31:1217-1220. doi: 10.1097/INF.0b013e318266bcf9
6. Kuo HC. Preventing coronary artery lesions in Kawasaki disease. Biomed J. 2017;40:141-146. doi: 10.1016/j.bj.2017.04.002
7. Chen S, Dong Y, Yin Y, et al. Intravenous immunoglobulin plus corticosteroid to prevent coronary artery abnormalities in Kawasaki disease: a meta-analysis. Heart. 2013;99:76-82. doi: 10.1136/heartjnl-2012-302126
8. Chantasiriwan N, Silvilairat S, Makonkawkeyoon K, et al. Predictors of intravenous immunoglobulin resistance and coronary artery aneurysm in patients with Kawasaki disease, Paediatr Int Child Health. 2018;38:209-212. doi: 10.1080/20469047.2018.1471381
9. Son MBF, Gauvreau K, Tremoulet AH, et al. Risk model development and validation for prediction of coronary artery aneurysms in Kawasaki disease in a North American population. J Am Heart Assoc. 2019;8:e011319. doi: 10.1161/JAHA.118.011319
10. de La Harpe M, di Bernardo S, Hofer M, et al. Thirty years of Kawasaki disease: a single-center study at the University Hospital of Lausanne. Front Pediatr. 2019;7:11. doi: 10.3389/fped.2019.00011
11. Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110:2747-2771. doi: 10.1161/01.CIR.0000145143.19711.78
THE CASE
A previously healthy 3-year-old girl presented to the emergency department with 4 days of fever and 2 days of right-side neck pain. The maximum temperature at home was 103 °F. The patient was irritable and vomited once. There were no other apparent or reported symptoms.
The neck exam was notable for nonfluctuant, swollen, and tender lymph nodes on the right side. Her sclera and conjunctiva were clear, and her oropharynx was unremarkable. Lab work revealed leukocytosis, with a white blood cell (WBC) count of 15.5 × 103/µL (normal range, 4.0-10.0 × 103/µL). She was given one 20 cc/kg normal saline bolus, started on intravenous clindamycin for presumed cervical lymphadenitis, and admitted to the hospital.
On Day 2, the patient developed a fine maculopapular rash on her chest, abdomen, and back. She had spiking fevers—as high as 102.2 °F—despite being on antibiotics for more than 24 hours. The erythrocyte sedimentation rate (ESR) was 39 mm/h (0-20 mm/h), and C-reactive protein (CRP) was 71.4 mg/L (0.0-4.9 mg/L). Due to concern for abscess, a neck ultrasound was performed; it showed a chain of enlarged lymph nodes in the right neck (largest, 2.3 × 1.1 × 1.4 cm) and no abscess.
On Day 3, clindamycin was switched to intravenous ampicillin/sulbactam because a nasal swab for methicillin-resistant Staphylococcus aureus was negative. A swab for respiratory viral infections was also negative. The patient then developed notable facial swelling, bilateral bulbar conjunctival injection with limbic sparing, and swelling of her hands and feet.
THE DIAGNOSIS
By the end of Day 3, the patient’s lab studies demonstrated microcytic anemia and low albumin (2.5 g/dL), but no transaminitis, thrombocytosis, or sterile pyuria. An electrocardiogram was unremarkable. A pediatric echocardiogram revealed hyperemic coronaries, indicating inflammation. The coronary arteries were measured in the upper limits of normal, and the patient’s Z-scores were < 2.5. (A Z-score < 2 indicates no involvement, 2 to < 2.5 indicates dilation, and ≥ 2.5 indicates aneurysm abnormality.1) An ultrasound of the right upper quadrant revealed an enlarged/elongated gallbladder. The patient therefore met clinical criteria for Kawasaki disease.
DISCUSSION
Kawasaki disease is a self-limited vasculitis of childhood and the leading cause of acquired heart disease in children in developed countries.1 The annual incidence of Kawasaki disease in North America is about 25 cases per 100,000 children < 5 years of age.1 In the United States, incidence is highest in Asian and Pacific Islander populations (30 per 100,000) and is particularly high among those of Japanese ancestry (~200 per 100,000).2 Disease prevalence is also noteworthy in Non-Hispanic African American (17 per 100,000) and Hispanic (16 per 100,000) populations.2
Diagnosis of Kawasaki disease requires presence of fever lasting at least 5 days and at least 4 of the following: bilateral bulbar conjunctival injection, oral mucous membrane changes (erythematous or cracked lips, erythematous pharynx, strawberry tongue), peripheral extremity changes (erythema of palms or soles, edema of hands or feet, and/or periungual desquamation), diffuse maculopapular rash, and cervical lymphadenopathy (≥ 1.5 cm, often unilateral). If ≥ 4 criteria are met, Kawasaki disease can be diagnosed on the fourth day of illness.1
Continue to: Laboratory findings suggesting...
Laboratory findings suggesting Kawasaki disease include a WBC count ≥ 15,000/mcL, normocytic, normochromic anemia, platelets ≥ 450,000/mcL after 7 days of illness, sterile pyuria (≥ 10 WBCs/high-power field), serum alanine aminotransferase level > 50 U/L, and serum albumin ≤ 3 g/dL.
Cardiac abnormalities are not included in the diagnostic criteria for Kawasaki disease but provide evidence in cases of incomplete Kawasaki disease if ≥ 4 criteria are not met and there is strong clinical suspicion.1 Incomplete Kawasaki disease should be considered in a patient with a CRP level ≥ 3 mg/dL and/or ESR ≥ 40 mm/h, ≥ 3 supplemental laboratory criteria, or a positive echocardiogram.1
Ultrasound imaging may reveal cervical lymph nodes resembling a “cluster of grapes.”3 The case patient’s imaging showed a “chain of enlarged lymph nodes.” She likely had gallbladder “hydrops” due to its increased longitudinal and horizontal diameter and lack of other anatomic changes.4
Prompt treatment is essential
Treatment for complete and incomplete Kawasaki disease is a single high dose of intravenous immunoglobulin (IVIG) along with aspirin. Patients meeting criteria should be treated as soon as the diagnosis is established.5 A single high dose of IVIG (2 g/kg), administered over 10 to 12 hours, should be initiated within 5 to 10 days of disease onset. Administering IVIG in the acute phase of Kawasaki disease reduces the prevalence of coronary artery abnormalities.6 Corticosteroids may be used as adjunctive therapy for patients with high risk of IVIG resistance.1,7-9
Our patient was not deemed to be at high risk for IVIG resistance (Non-Japanese patient, age at fever onset > 6 months, absence of coronary artery aneurysm9) and was administered IVIG on Day 4. She was also given moderate-dose aspirin, then later transitioned to low-dose aspirin. The patient’s fevers improved, she was less irritable, and she had periods of playfulness. Physical exam then showed erythematous and cracked lips with peeling skin.
Continue to: The patient was discharged...
The patient was discharged home on Day 8, after her fever resolved, with instructions to continue low-dose aspirin and to obtain a repeat echocardiogram, gallbladder ultrasound, and lab work in 2 weeks. At her follow-up appointment, periungual desquamation was noted, and ultrasound showed continued enlarged/elongated gallbladder. A repeat echocardiogram was not available. Overall, the patient recovered from Kawasaki disease after therapeutic intervention.
THE TAKEAWAY
Kawasaki disease can be relatively rare in North American populations, but it is important for physicians to be able to recognize and treat it. Untreated children have a 25% chance of developing coronary artery aneurysms.1,10,11 Early treatment with IVIG can decrease risk to 5%, resulting in an excellent medium- to long-term prognosis for patients.10 Thorough physical examination and an appropriate degree of clinical suspicion was key in this case of Kawasaki disease.
Taisha Doo, MD, 1401 Madison Street, Suite #100, Seattle, WA 98104; taisha.doo@swedish.org
THE CASE
A previously healthy 3-year-old girl presented to the emergency department with 4 days of fever and 2 days of right-side neck pain. The maximum temperature at home was 103 °F. The patient was irritable and vomited once. There were no other apparent or reported symptoms.
The neck exam was notable for nonfluctuant, swollen, and tender lymph nodes on the right side. Her sclera and conjunctiva were clear, and her oropharynx was unremarkable. Lab work revealed leukocytosis, with a white blood cell (WBC) count of 15.5 × 103/µL (normal range, 4.0-10.0 × 103/µL). She was given one 20 cc/kg normal saline bolus, started on intravenous clindamycin for presumed cervical lymphadenitis, and admitted to the hospital.
On Day 2, the patient developed a fine maculopapular rash on her chest, abdomen, and back. She had spiking fevers—as high as 102.2 °F—despite being on antibiotics for more than 24 hours. The erythrocyte sedimentation rate (ESR) was 39 mm/h (0-20 mm/h), and C-reactive protein (CRP) was 71.4 mg/L (0.0-4.9 mg/L). Due to concern for abscess, a neck ultrasound was performed; it showed a chain of enlarged lymph nodes in the right neck (largest, 2.3 × 1.1 × 1.4 cm) and no abscess.
On Day 3, clindamycin was switched to intravenous ampicillin/sulbactam because a nasal swab for methicillin-resistant Staphylococcus aureus was negative. A swab for respiratory viral infections was also negative. The patient then developed notable facial swelling, bilateral bulbar conjunctival injection with limbic sparing, and swelling of her hands and feet.
THE DIAGNOSIS
By the end of Day 3, the patient’s lab studies demonstrated microcytic anemia and low albumin (2.5 g/dL), but no transaminitis, thrombocytosis, or sterile pyuria. An electrocardiogram was unremarkable. A pediatric echocardiogram revealed hyperemic coronaries, indicating inflammation. The coronary arteries were measured in the upper limits of normal, and the patient’s Z-scores were < 2.5. (A Z-score < 2 indicates no involvement, 2 to < 2.5 indicates dilation, and ≥ 2.5 indicates aneurysm abnormality.1) An ultrasound of the right upper quadrant revealed an enlarged/elongated gallbladder. The patient therefore met clinical criteria for Kawasaki disease.
DISCUSSION
Kawasaki disease is a self-limited vasculitis of childhood and the leading cause of acquired heart disease in children in developed countries.1 The annual incidence of Kawasaki disease in North America is about 25 cases per 100,000 children < 5 years of age.1 In the United States, incidence is highest in Asian and Pacific Islander populations (30 per 100,000) and is particularly high among those of Japanese ancestry (~200 per 100,000).2 Disease prevalence is also noteworthy in Non-Hispanic African American (17 per 100,000) and Hispanic (16 per 100,000) populations.2
Diagnosis of Kawasaki disease requires presence of fever lasting at least 5 days and at least 4 of the following: bilateral bulbar conjunctival injection, oral mucous membrane changes (erythematous or cracked lips, erythematous pharynx, strawberry tongue), peripheral extremity changes (erythema of palms or soles, edema of hands or feet, and/or periungual desquamation), diffuse maculopapular rash, and cervical lymphadenopathy (≥ 1.5 cm, often unilateral). If ≥ 4 criteria are met, Kawasaki disease can be diagnosed on the fourth day of illness.1
Continue to: Laboratory findings suggesting...
Laboratory findings suggesting Kawasaki disease include a WBC count ≥ 15,000/mcL, normocytic, normochromic anemia, platelets ≥ 450,000/mcL after 7 days of illness, sterile pyuria (≥ 10 WBCs/high-power field), serum alanine aminotransferase level > 50 U/L, and serum albumin ≤ 3 g/dL.
Cardiac abnormalities are not included in the diagnostic criteria for Kawasaki disease but provide evidence in cases of incomplete Kawasaki disease if ≥ 4 criteria are not met and there is strong clinical suspicion.1 Incomplete Kawasaki disease should be considered in a patient with a CRP level ≥ 3 mg/dL and/or ESR ≥ 40 mm/h, ≥ 3 supplemental laboratory criteria, or a positive echocardiogram.1
Ultrasound imaging may reveal cervical lymph nodes resembling a “cluster of grapes.”3 The case patient’s imaging showed a “chain of enlarged lymph nodes.” She likely had gallbladder “hydrops” due to its increased longitudinal and horizontal diameter and lack of other anatomic changes.4
Prompt treatment is essential
Treatment for complete and incomplete Kawasaki disease is a single high dose of intravenous immunoglobulin (IVIG) along with aspirin. Patients meeting criteria should be treated as soon as the diagnosis is established.5 A single high dose of IVIG (2 g/kg), administered over 10 to 12 hours, should be initiated within 5 to 10 days of disease onset. Administering IVIG in the acute phase of Kawasaki disease reduces the prevalence of coronary artery abnormalities.6 Corticosteroids may be used as adjunctive therapy for patients with high risk of IVIG resistance.1,7-9
Our patient was not deemed to be at high risk for IVIG resistance (Non-Japanese patient, age at fever onset > 6 months, absence of coronary artery aneurysm9) and was administered IVIG on Day 4. She was also given moderate-dose aspirin, then later transitioned to low-dose aspirin. The patient’s fevers improved, she was less irritable, and she had periods of playfulness. Physical exam then showed erythematous and cracked lips with peeling skin.
Continue to: The patient was discharged...
The patient was discharged home on Day 8, after her fever resolved, with instructions to continue low-dose aspirin and to obtain a repeat echocardiogram, gallbladder ultrasound, and lab work in 2 weeks. At her follow-up appointment, periungual desquamation was noted, and ultrasound showed continued enlarged/elongated gallbladder. A repeat echocardiogram was not available. Overall, the patient recovered from Kawasaki disease after therapeutic intervention.
THE TAKEAWAY
Kawasaki disease can be relatively rare in North American populations, but it is important for physicians to be able to recognize and treat it. Untreated children have a 25% chance of developing coronary artery aneurysms.1,10,11 Early treatment with IVIG can decrease risk to 5%, resulting in an excellent medium- to long-term prognosis for patients.10 Thorough physical examination and an appropriate degree of clinical suspicion was key in this case of Kawasaki disease.
Taisha Doo, MD, 1401 Madison Street, Suite #100, Seattle, WA 98104; taisha.doo@swedish.org
1. McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. 2017;135:e927-e999. doi: 10.1161/CIR.0000000000000484
2. Holman RC, Belay ED, Christensen KY, et al. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis. 2010;29:483-488. doi: 10.1097/INF.0b013e3181cf8705
3. Tashiro N, Matsubara T, Uchida M, et al. Ultrasonographic evaluation of cervical lymph nodes in Kawasaki disease. Pediatrics. 2002;109:e77. doi: 10.1542/peds.109.5.e77
4. Chen CJ, Huang FC, Taio MM, et al. Sonographic gallbladder abnormality is associated with intravenous immunoglobulin resistance in Kawasaki disease. Scientific World J. 2012;2012:485758. doi: 10.1100/2012/485758
5. Dominguez SR, Anderson MS, El-Adawy M, et al. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012;31:1217-1220. doi: 10.1097/INF.0b013e318266bcf9
6. Kuo HC. Preventing coronary artery lesions in Kawasaki disease. Biomed J. 2017;40:141-146. doi: 10.1016/j.bj.2017.04.002
7. Chen S, Dong Y, Yin Y, et al. Intravenous immunoglobulin plus corticosteroid to prevent coronary artery abnormalities in Kawasaki disease: a meta-analysis. Heart. 2013;99:76-82. doi: 10.1136/heartjnl-2012-302126
8. Chantasiriwan N, Silvilairat S, Makonkawkeyoon K, et al. Predictors of intravenous immunoglobulin resistance and coronary artery aneurysm in patients with Kawasaki disease, Paediatr Int Child Health. 2018;38:209-212. doi: 10.1080/20469047.2018.1471381
9. Son MBF, Gauvreau K, Tremoulet AH, et al. Risk model development and validation for prediction of coronary artery aneurysms in Kawasaki disease in a North American population. J Am Heart Assoc. 2019;8:e011319. doi: 10.1161/JAHA.118.011319
10. de La Harpe M, di Bernardo S, Hofer M, et al. Thirty years of Kawasaki disease: a single-center study at the University Hospital of Lausanne. Front Pediatr. 2019;7:11. doi: 10.3389/fped.2019.00011
11. Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110:2747-2771. doi: 10.1161/01.CIR.0000145143.19711.78
1. McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. 2017;135:e927-e999. doi: 10.1161/CIR.0000000000000484
2. Holman RC, Belay ED, Christensen KY, et al. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis. 2010;29:483-488. doi: 10.1097/INF.0b013e3181cf8705
3. Tashiro N, Matsubara T, Uchida M, et al. Ultrasonographic evaluation of cervical lymph nodes in Kawasaki disease. Pediatrics. 2002;109:e77. doi: 10.1542/peds.109.5.e77
4. Chen CJ, Huang FC, Taio MM, et al. Sonographic gallbladder abnormality is associated with intravenous immunoglobulin resistance in Kawasaki disease. Scientific World J. 2012;2012:485758. doi: 10.1100/2012/485758
5. Dominguez SR, Anderson MS, El-Adawy M, et al. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012;31:1217-1220. doi: 10.1097/INF.0b013e318266bcf9
6. Kuo HC. Preventing coronary artery lesions in Kawasaki disease. Biomed J. 2017;40:141-146. doi: 10.1016/j.bj.2017.04.002
7. Chen S, Dong Y, Yin Y, et al. Intravenous immunoglobulin plus corticosteroid to prevent coronary artery abnormalities in Kawasaki disease: a meta-analysis. Heart. 2013;99:76-82. doi: 10.1136/heartjnl-2012-302126
8. Chantasiriwan N, Silvilairat S, Makonkawkeyoon K, et al. Predictors of intravenous immunoglobulin resistance and coronary artery aneurysm in patients with Kawasaki disease, Paediatr Int Child Health. 2018;38:209-212. doi: 10.1080/20469047.2018.1471381
9. Son MBF, Gauvreau K, Tremoulet AH, et al. Risk model development and validation for prediction of coronary artery aneurysms in Kawasaki disease in a North American population. J Am Heart Assoc. 2019;8:e011319. doi: 10.1161/JAHA.118.011319
10. de La Harpe M, di Bernardo S, Hofer M, et al. Thirty years of Kawasaki disease: a single-center study at the University Hospital of Lausanne. Front Pediatr. 2019;7:11. doi: 10.3389/fped.2019.00011
11. Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110:2747-2771. doi: 10.1161/01.CIR.0000145143.19711.78
Alcohol abstinence reduces A-fib burden in drinkers
ILLUSTRATIVE CASE
A 61-year-old man with hypertension and paroxysmal AF presents to your office shortly after experiencing his third episode of AF in the past 6 months. He describes these episodes, which last for several days, as “just awful,” noting that when he experiences AF, he has fatigue, palpitations, and shortness of breath and “can’t stop paying attention to my heart.” The patient, who has a body mass index of 32, consumes more than 15 alcoholic drinks per week. What can you recommend to him that will decrease his likelihood of experiencing more episodes of AF?
AF is the most common sustained cardiac arrhythmia. It is associated with significant morbidity and mortality. Known risk factors include obesity, physical inactivity, sleep apnea, diabetes, and hypertension.2
According to the Centers for Disease Control and Prevention, an estimated 12.1 million people in the United States will have AF by 2030. In 2018, AF was mentioned on more than 183,000 death certificates and was the underlying cause of more than 26,000 of those deaths.3 AF is the primary diagnosis in 450,000 hospitalizations annually,4 and the death rate from AF as the primary or contributing cause of death has been rising for more than 2 decades.3
More than 50% of Americans report alcohol consumption within the past month.5 Although alcohol use is associated with new and recurrent AF, only limited prospective data show a clear and causal association between abstaining from alcohol and decreasing AF recurrence.
STUDY SUMMARY
Reduction in AF recurrence and total AF burden following alcohol abstinence
This multicenter, prospective, open-label, randomized controlled trial (N = 140) from 6 sites in Australia evaluated the impact of alcohol abstinence on both the recurrence of AF and the amount of time in AF. Study participants were ages 18 to 85 years, consumed 10 or more standard alcohol-containing drinks per week, had paroxysmal or persistent AF, and were in sinus rhythm at the time of enrollment, regardless of antiarrhythmic therapy. Exclusion criteria included alcohol dependence or abuse, severe left ventricular systolic dysfunction (ejection fraction < 35%), clinically significant noncardiac illness, and/or coexisting psychiatric disorder.1
After a 4-week run-in period, patients were randomized to either an abstinence or a control group in a 1:1 fashion. Patients enrolled in the abstinence group were encouraged to abstain from alcohol consumption for 6 months and were provided with written and oral instructions to assist with abstaining. Control group patients continued their same level of alcohol consumption. Comprehensive rhythm monitoring occurred for all patients after randomization.
Alcohol consumption was reported by both groups using a weekly alcohol diary, supplemented with a visual guide showing pictures of standard alcohol drinks. For the abstinence group, random urine testing for ethyl glucuronide (an alcohol metabolite) was possible if no alcohol intake was reported. Primary outcomes during the 6-month study included recurrence of AF and total AF burden (percentage of time in AF).
Continue to: Secondary outcomes included hospitalizations...
Secondary outcomes included hospitalizations for AF, AF symptom severity, and change in weight. Blood pressure, quality-of-life, and depression scores were missing for > 35% of patients.1
Patients were randomized evenly to the control and abstinence groups. The typical patient was an overweight male in his early 60s with paroxysmal AF, who was taking an antiarrhythmic agent. Patients in the abstinence group decreased their alcohol consumption from 16.8 to 2.1 drinks per week (87.5% reduction; mean difference = –14.7; 95% CI, –12.7 to –16.7). Patients in the control group reduced their intake from 16.4 to 13.2 drinks per week (19.5% reduction; mean difference = –3.2; 95% CI, –1.9 to –4.4).1
AF recurred in 53% vs 73% of the abstinence and control groups, respectively, with a longer period before recurrence in the abstinence group than in the control group (hazard ratio = 0.55; 95% CI, 0.36-0.84; P = .005; number needed to treat = 5). The AF burden was also lower in the abstinence group (0.5%; interquartile range [IQR] = 0.0-3.0) than in the control group (1.2%; IQR = 0.0-10.3; P = .01). The abstinence group had a lower percentage of AF hospitalizations compared with the control group (9% vs 20%), and fewer patients reporting moderate or severe AF symptoms (10% vs 32%). In addition, the abstinence group lost 3.7 kg more weight than did the control group at 6 months.1
WHAT’S NEW
Objective new evidence for effective patient counseling
Alcohol consumption and its association with the onset and recurrence of AF has been documented previously.6 This study was the first to prospectively examine if abstaining from alcohol reduces paroxysmal AF episodes in moderate drinkers.
The study identified clinically meaningful findings among those who abstained from alcohol, including decreased AF recurrence rates, increased time to recurrence, and lower overall AF burden. This provides objective evidence that can be used for motivational interviewing in patients with paroxysmal AF who may be receptive to reducing or abstaining from alcohol consumption.
Continue to: CAVEATS
CAVEATS
The narrow study population may not be widely applicable
The study population was predominantly male, in their seventh decade of life (mean age, 61), and living in Australia. Rates of AF and symptomatology differ by gender and age, making this information challenging to apply to women or older populations. The study excluded patients with alcohol dependence or abuse, left ventricular systolic dysfunction (ejection fraction < 35%), coexisting psychiatric disorders, and clinically significant noncardiac illnesses, limiting the study’s generalizability to these patient populations. Overall, AF recurrence was low in both groups despite the intervention, and the study did not evaluate the efficacy of the counseling method for abstinence.
Since publication of this article, a prospective cohort study of approximately 3800 Swiss patients with AF evaluated the effect of alcohol consumption on the rate of stroke and embolic events. That study did not find statistically significant correlations between patients who drank no alcohol per day, > 0 to < 1, 1 to < 2, or ≥ 2 drinks per day and their rate of stroke.7 However, this study did not specifically evaluate the rate of AF recurrence or time spent in AF among the cohort, which is clinically meaningful for patient morbidity.1
CHALLENGES TO IMPLEMENTATION
Patient willingness to cut alcohol consumption may be limited
The largest challenge to implementation of this intervention is most likely the willingness of patients to cut their alcohol consumption. In this study population, 697 patients were screened for enrollment and met inclusion criteria; however, 491 patients (70.4%) were not willing to consider abstinence from alcohol, and after the run-in phase, another 17 declined randomization. Many primary care physicians would likely agree that while it is easy to encourage patients to drink less, patient adherence to these recommendations, particularly abstaining, is likely to be limited.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28. doi: 10.1056/NEJMoa1817591
2. Chung MK, Eckhardt LL, Chen LY, et al. Lifestyle and risk factor modification for reduction of atrial fibrillation: a scientific statement from the American Heart Association. Circulation. 2020;141:e750-e772. doi: 10.1161/CIR.0000000000000748
3. Atrial fibrillation. Centers for Disease Control and Prevention. Last reviewed September 27, 2021. Accessed February 9, 2022. www.cdc.gov/heartdisease/atrial_fibrillation.htm
4. Benjamin EJ, Muntner P, Alonso A, et al. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139:e56-e528. doi: 10.1161/CIR.0000000000000659
5. Alcohol facts and statistics. National Institute on Alcohol Abuse and Alcoholism. Updated June 2021. Accessed February 9, 2022. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/alcohol-facts-and-statistics
6. Kodama S, Saito K, Tanaka S, et al. Alcohol consumption and risk of atrial fibrillation: a meta-analysis. J Am Coll Cardiol. 2011;57:427-436. doi: 10.1016/j.jacc.2010.08.641
7. Reddiess P, Aeschbacher S, Meyre P, et al. Alcohol consumption and risk of cardiovascular outcomes and bleeding in patients with established atrial fibrillation. CMAJ. 2021;193:E117-E123. doi: 10.1503/cmaj.200778
ILLUSTRATIVE CASE
A 61-year-old man with hypertension and paroxysmal AF presents to your office shortly after experiencing his third episode of AF in the past 6 months. He describes these episodes, which last for several days, as “just awful,” noting that when he experiences AF, he has fatigue, palpitations, and shortness of breath and “can’t stop paying attention to my heart.” The patient, who has a body mass index of 32, consumes more than 15 alcoholic drinks per week. What can you recommend to him that will decrease his likelihood of experiencing more episodes of AF?
AF is the most common sustained cardiac arrhythmia. It is associated with significant morbidity and mortality. Known risk factors include obesity, physical inactivity, sleep apnea, diabetes, and hypertension.2
According to the Centers for Disease Control and Prevention, an estimated 12.1 million people in the United States will have AF by 2030. In 2018, AF was mentioned on more than 183,000 death certificates and was the underlying cause of more than 26,000 of those deaths.3 AF is the primary diagnosis in 450,000 hospitalizations annually,4 and the death rate from AF as the primary or contributing cause of death has been rising for more than 2 decades.3
More than 50% of Americans report alcohol consumption within the past month.5 Although alcohol use is associated with new and recurrent AF, only limited prospective data show a clear and causal association between abstaining from alcohol and decreasing AF recurrence.
STUDY SUMMARY
Reduction in AF recurrence and total AF burden following alcohol abstinence
This multicenter, prospective, open-label, randomized controlled trial (N = 140) from 6 sites in Australia evaluated the impact of alcohol abstinence on both the recurrence of AF and the amount of time in AF. Study participants were ages 18 to 85 years, consumed 10 or more standard alcohol-containing drinks per week, had paroxysmal or persistent AF, and were in sinus rhythm at the time of enrollment, regardless of antiarrhythmic therapy. Exclusion criteria included alcohol dependence or abuse, severe left ventricular systolic dysfunction (ejection fraction < 35%), clinically significant noncardiac illness, and/or coexisting psychiatric disorder.1
After a 4-week run-in period, patients were randomized to either an abstinence or a control group in a 1:1 fashion. Patients enrolled in the abstinence group were encouraged to abstain from alcohol consumption for 6 months and were provided with written and oral instructions to assist with abstaining. Control group patients continued their same level of alcohol consumption. Comprehensive rhythm monitoring occurred for all patients after randomization.
Alcohol consumption was reported by both groups using a weekly alcohol diary, supplemented with a visual guide showing pictures of standard alcohol drinks. For the abstinence group, random urine testing for ethyl glucuronide (an alcohol metabolite) was possible if no alcohol intake was reported. Primary outcomes during the 6-month study included recurrence of AF and total AF burden (percentage of time in AF).
Continue to: Secondary outcomes included hospitalizations...
Secondary outcomes included hospitalizations for AF, AF symptom severity, and change in weight. Blood pressure, quality-of-life, and depression scores were missing for > 35% of patients.1
Patients were randomized evenly to the control and abstinence groups. The typical patient was an overweight male in his early 60s with paroxysmal AF, who was taking an antiarrhythmic agent. Patients in the abstinence group decreased their alcohol consumption from 16.8 to 2.1 drinks per week (87.5% reduction; mean difference = –14.7; 95% CI, –12.7 to –16.7). Patients in the control group reduced their intake from 16.4 to 13.2 drinks per week (19.5% reduction; mean difference = –3.2; 95% CI, –1.9 to –4.4).1
AF recurred in 53% vs 73% of the abstinence and control groups, respectively, with a longer period before recurrence in the abstinence group than in the control group (hazard ratio = 0.55; 95% CI, 0.36-0.84; P = .005; number needed to treat = 5). The AF burden was also lower in the abstinence group (0.5%; interquartile range [IQR] = 0.0-3.0) than in the control group (1.2%; IQR = 0.0-10.3; P = .01). The abstinence group had a lower percentage of AF hospitalizations compared with the control group (9% vs 20%), and fewer patients reporting moderate or severe AF symptoms (10% vs 32%). In addition, the abstinence group lost 3.7 kg more weight than did the control group at 6 months.1
WHAT’S NEW
Objective new evidence for effective patient counseling
Alcohol consumption and its association with the onset and recurrence of AF has been documented previously.6 This study was the first to prospectively examine if abstaining from alcohol reduces paroxysmal AF episodes in moderate drinkers.
The study identified clinically meaningful findings among those who abstained from alcohol, including decreased AF recurrence rates, increased time to recurrence, and lower overall AF burden. This provides objective evidence that can be used for motivational interviewing in patients with paroxysmal AF who may be receptive to reducing or abstaining from alcohol consumption.
Continue to: CAVEATS
CAVEATS
The narrow study population may not be widely applicable
The study population was predominantly male, in their seventh decade of life (mean age, 61), and living in Australia. Rates of AF and symptomatology differ by gender and age, making this information challenging to apply to women or older populations. The study excluded patients with alcohol dependence or abuse, left ventricular systolic dysfunction (ejection fraction < 35%), coexisting psychiatric disorders, and clinically significant noncardiac illnesses, limiting the study’s generalizability to these patient populations. Overall, AF recurrence was low in both groups despite the intervention, and the study did not evaluate the efficacy of the counseling method for abstinence.
Since publication of this article, a prospective cohort study of approximately 3800 Swiss patients with AF evaluated the effect of alcohol consumption on the rate of stroke and embolic events. That study did not find statistically significant correlations between patients who drank no alcohol per day, > 0 to < 1, 1 to < 2, or ≥ 2 drinks per day and their rate of stroke.7 However, this study did not specifically evaluate the rate of AF recurrence or time spent in AF among the cohort, which is clinically meaningful for patient morbidity.1
CHALLENGES TO IMPLEMENTATION
Patient willingness to cut alcohol consumption may be limited
The largest challenge to implementation of this intervention is most likely the willingness of patients to cut their alcohol consumption. In this study population, 697 patients were screened for enrollment and met inclusion criteria; however, 491 patients (70.4%) were not willing to consider abstinence from alcohol, and after the run-in phase, another 17 declined randomization. Many primary care physicians would likely agree that while it is easy to encourage patients to drink less, patient adherence to these recommendations, particularly abstaining, is likely to be limited.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 61-year-old man with hypertension and paroxysmal AF presents to your office shortly after experiencing his third episode of AF in the past 6 months. He describes these episodes, which last for several days, as “just awful,” noting that when he experiences AF, he has fatigue, palpitations, and shortness of breath and “can’t stop paying attention to my heart.” The patient, who has a body mass index of 32, consumes more than 15 alcoholic drinks per week. What can you recommend to him that will decrease his likelihood of experiencing more episodes of AF?
AF is the most common sustained cardiac arrhythmia. It is associated with significant morbidity and mortality. Known risk factors include obesity, physical inactivity, sleep apnea, diabetes, and hypertension.2
According to the Centers for Disease Control and Prevention, an estimated 12.1 million people in the United States will have AF by 2030. In 2018, AF was mentioned on more than 183,000 death certificates and was the underlying cause of more than 26,000 of those deaths.3 AF is the primary diagnosis in 450,000 hospitalizations annually,4 and the death rate from AF as the primary or contributing cause of death has been rising for more than 2 decades.3
More than 50% of Americans report alcohol consumption within the past month.5 Although alcohol use is associated with new and recurrent AF, only limited prospective data show a clear and causal association between abstaining from alcohol and decreasing AF recurrence.
STUDY SUMMARY
Reduction in AF recurrence and total AF burden following alcohol abstinence
This multicenter, prospective, open-label, randomized controlled trial (N = 140) from 6 sites in Australia evaluated the impact of alcohol abstinence on both the recurrence of AF and the amount of time in AF. Study participants were ages 18 to 85 years, consumed 10 or more standard alcohol-containing drinks per week, had paroxysmal or persistent AF, and were in sinus rhythm at the time of enrollment, regardless of antiarrhythmic therapy. Exclusion criteria included alcohol dependence or abuse, severe left ventricular systolic dysfunction (ejection fraction < 35%), clinically significant noncardiac illness, and/or coexisting psychiatric disorder.1
After a 4-week run-in period, patients were randomized to either an abstinence or a control group in a 1:1 fashion. Patients enrolled in the abstinence group were encouraged to abstain from alcohol consumption for 6 months and were provided with written and oral instructions to assist with abstaining. Control group patients continued their same level of alcohol consumption. Comprehensive rhythm monitoring occurred for all patients after randomization.
Alcohol consumption was reported by both groups using a weekly alcohol diary, supplemented with a visual guide showing pictures of standard alcohol drinks. For the abstinence group, random urine testing for ethyl glucuronide (an alcohol metabolite) was possible if no alcohol intake was reported. Primary outcomes during the 6-month study included recurrence of AF and total AF burden (percentage of time in AF).
Continue to: Secondary outcomes included hospitalizations...
Secondary outcomes included hospitalizations for AF, AF symptom severity, and change in weight. Blood pressure, quality-of-life, and depression scores were missing for > 35% of patients.1
Patients were randomized evenly to the control and abstinence groups. The typical patient was an overweight male in his early 60s with paroxysmal AF, who was taking an antiarrhythmic agent. Patients in the abstinence group decreased their alcohol consumption from 16.8 to 2.1 drinks per week (87.5% reduction; mean difference = –14.7; 95% CI, –12.7 to –16.7). Patients in the control group reduced their intake from 16.4 to 13.2 drinks per week (19.5% reduction; mean difference = –3.2; 95% CI, –1.9 to –4.4).1
AF recurred in 53% vs 73% of the abstinence and control groups, respectively, with a longer period before recurrence in the abstinence group than in the control group (hazard ratio = 0.55; 95% CI, 0.36-0.84; P = .005; number needed to treat = 5). The AF burden was also lower in the abstinence group (0.5%; interquartile range [IQR] = 0.0-3.0) than in the control group (1.2%; IQR = 0.0-10.3; P = .01). The abstinence group had a lower percentage of AF hospitalizations compared with the control group (9% vs 20%), and fewer patients reporting moderate or severe AF symptoms (10% vs 32%). In addition, the abstinence group lost 3.7 kg more weight than did the control group at 6 months.1
WHAT’S NEW
Objective new evidence for effective patient counseling
Alcohol consumption and its association with the onset and recurrence of AF has been documented previously.6 This study was the first to prospectively examine if abstaining from alcohol reduces paroxysmal AF episodes in moderate drinkers.
The study identified clinically meaningful findings among those who abstained from alcohol, including decreased AF recurrence rates, increased time to recurrence, and lower overall AF burden. This provides objective evidence that can be used for motivational interviewing in patients with paroxysmal AF who may be receptive to reducing or abstaining from alcohol consumption.
Continue to: CAVEATS
CAVEATS
The narrow study population may not be widely applicable
The study population was predominantly male, in their seventh decade of life (mean age, 61), and living in Australia. Rates of AF and symptomatology differ by gender and age, making this information challenging to apply to women or older populations. The study excluded patients with alcohol dependence or abuse, left ventricular systolic dysfunction (ejection fraction < 35%), coexisting psychiatric disorders, and clinically significant noncardiac illnesses, limiting the study’s generalizability to these patient populations. Overall, AF recurrence was low in both groups despite the intervention, and the study did not evaluate the efficacy of the counseling method for abstinence.
Since publication of this article, a prospective cohort study of approximately 3800 Swiss patients with AF evaluated the effect of alcohol consumption on the rate of stroke and embolic events. That study did not find statistically significant correlations between patients who drank no alcohol per day, > 0 to < 1, 1 to < 2, or ≥ 2 drinks per day and their rate of stroke.7 However, this study did not specifically evaluate the rate of AF recurrence or time spent in AF among the cohort, which is clinically meaningful for patient morbidity.1
CHALLENGES TO IMPLEMENTATION
Patient willingness to cut alcohol consumption may be limited
The largest challenge to implementation of this intervention is most likely the willingness of patients to cut their alcohol consumption. In this study population, 697 patients were screened for enrollment and met inclusion criteria; however, 491 patients (70.4%) were not willing to consider abstinence from alcohol, and after the run-in phase, another 17 declined randomization. Many primary care physicians would likely agree that while it is easy to encourage patients to drink less, patient adherence to these recommendations, particularly abstaining, is likely to be limited.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28. doi: 10.1056/NEJMoa1817591
2. Chung MK, Eckhardt LL, Chen LY, et al. Lifestyle and risk factor modification for reduction of atrial fibrillation: a scientific statement from the American Heart Association. Circulation. 2020;141:e750-e772. doi: 10.1161/CIR.0000000000000748
3. Atrial fibrillation. Centers for Disease Control and Prevention. Last reviewed September 27, 2021. Accessed February 9, 2022. www.cdc.gov/heartdisease/atrial_fibrillation.htm
4. Benjamin EJ, Muntner P, Alonso A, et al. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139:e56-e528. doi: 10.1161/CIR.0000000000000659
5. Alcohol facts and statistics. National Institute on Alcohol Abuse and Alcoholism. Updated June 2021. Accessed February 9, 2022. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/alcohol-facts-and-statistics
6. Kodama S, Saito K, Tanaka S, et al. Alcohol consumption and risk of atrial fibrillation: a meta-analysis. J Am Coll Cardiol. 2011;57:427-436. doi: 10.1016/j.jacc.2010.08.641
7. Reddiess P, Aeschbacher S, Meyre P, et al. Alcohol consumption and risk of cardiovascular outcomes and bleeding in patients with established atrial fibrillation. CMAJ. 2021;193:E117-E123. doi: 10.1503/cmaj.200778
1. Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28. doi: 10.1056/NEJMoa1817591
2. Chung MK, Eckhardt LL, Chen LY, et al. Lifestyle and risk factor modification for reduction of atrial fibrillation: a scientific statement from the American Heart Association. Circulation. 2020;141:e750-e772. doi: 10.1161/CIR.0000000000000748
3. Atrial fibrillation. Centers for Disease Control and Prevention. Last reviewed September 27, 2021. Accessed February 9, 2022. www.cdc.gov/heartdisease/atrial_fibrillation.htm
4. Benjamin EJ, Muntner P, Alonso A, et al. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139:e56-e528. doi: 10.1161/CIR.0000000000000659
5. Alcohol facts and statistics. National Institute on Alcohol Abuse and Alcoholism. Updated June 2021. Accessed February 9, 2022. www.niaaa.nih.gov/publications/brochures-and-fact-sheets/alcohol-facts-and-statistics
6. Kodama S, Saito K, Tanaka S, et al. Alcohol consumption and risk of atrial fibrillation: a meta-analysis. J Am Coll Cardiol. 2011;57:427-436. doi: 10.1016/j.jacc.2010.08.641
7. Reddiess P, Aeschbacher S, Meyre P, et al. Alcohol consumption and risk of cardiovascular outcomes and bleeding in patients with established atrial fibrillation. CMAJ. 2021;193:E117-E123. doi: 10.1503/cmaj.200778
PRACTICE CHANGER
Counsel patients with paroxysmal or persistent atrial fibrillation (AF) who drink moderately (≥ 10 drinks per week) that they can reduce their time in AF, as well as their overall recurrence of AF, by decreasing their alcohol consumption by half or more.
STRENGTH OF RECOMMENDATION
B: Based on a well-performed randomized controlled trial1
Voskoboinik A, Kalman JM, De Silva A, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. 2020;382:20-28.
Early treatment may delay atherosclerosis in familial hypercholesterolemia
Patients with familial hypercholesterolemia (FH) who start lipid-lowering treatment earlier in life may reduce their cardiovascular risk, compared with those who don’t begin treatment early, according to results of a recent meta-analysis.
They showed a difference in the carotid intima-media thickness (IMT) between patients with and without FH that increased with age, but there was also a difference in IMT seen among patients with FH who started treatment early, compared with untreated patients with FH, Kika van Bergen en Henegouwen, of the departments of pediatrics and epidemiology and data science at Amsterdam University Medical Center, and colleagues wrote in their report, published in the Journal of Clinical Lipidology.
“The fact that the difference in IMT increases with age between FH patients and unaffected controls, and is more pronounced in studies with untreated FH patients than in studies with treated patients, suggests that starting treatment already at a young age in patients with FH is preferred,” the researchers wrote. “However, despite treatment, IMT in treated FH patients is still thicker in comparison to subjects without FH.”
The researchers identified 42 studies with among patients with FH and healthy control groups across the MEDLINE, EMBASE and Trials.gov databases up to a cutoff date of April 2020, with 39 studies specifically examining carotid IMT, 2 studies evaluating carotid and femoral IMT, and 1 study evaluating femoral IMT alone. Overall, the researchers examined IMT measurements in 3,796 patients with FH and 2,363 control group participants.
Although data on age and gender for FH and control groups were not available in 6 studies, the mean age ranged from 9 to 57 years for patients with FH and from 8 to 61 years in the control group. Men comprised just under half of both the FH and control groups.
The mean between-group difference in carotid IMT in 34 studies was 0.11 mm (95% confidence interval, 0.06-0.15 mm; P < .001) for patients with FH, compared with the control group, while the mean difference in femoral IMT in three studies was 0.47 mm (95% CI, 0.19-0.74 mm; P < .001) between FH and control groups.
In 13 studies in which data on differences between partly treated and untreated FH were available, there was a significant between-group difference in carotid IMT with partly treated patients with FH, compared with the control group (0.05 mm; 95% CI, 0.03-0.08 mm; P < .001), but a larger mean between-group difference in carotid IMT among untreated patients with FH, compared with a control group (0.12 mm; 95% CI, 0.03-0.21 mm; P = .009).
The researchers also analyzed how age impacts carotid IMT, and they found patients with FH had a mean increase of 0.0018 mm (95% CI, –0.0007 to 0.0042 mm) over a control group in 34 studies. For patients with partly treated FH, compared with patients with untreated FH, the mean between-group increase per year was smaller (0.0023 mm; 95% CI, 0.0021-0.0025 mm), compared with the control group (0.0104 mm; 95% CI, 0.0100-0.0108 mm).
“This sign of residual risk might suggest that more robust cholesterol-lowering treatment and achieving treatment targets, or earlier treatment initiation, is needed to reduce IMT progression to non-FH conditions,” the researchers said. “Therefore, we must find and diagnose these patients, and treat them according to current guidelines.”
Limitations of the authors’ meta-analyses include heterogeneity among studies, differences in IMT measurement protocols, and inclusion of studies with an open-label design. Although randomized clinical trials would be preferable to compare treatment effect, “since statin therapy is indicated in FH patients to reduce [cardiovascular disease], it would be unethical to have a placebo group,” they said.
The authors reported no relevant financial disclosures.
Patients with familial hypercholesterolemia (FH) who start lipid-lowering treatment earlier in life may reduce their cardiovascular risk, compared with those who don’t begin treatment early, according to results of a recent meta-analysis.
They showed a difference in the carotid intima-media thickness (IMT) between patients with and without FH that increased with age, but there was also a difference in IMT seen among patients with FH who started treatment early, compared with untreated patients with FH, Kika van Bergen en Henegouwen, of the departments of pediatrics and epidemiology and data science at Amsterdam University Medical Center, and colleagues wrote in their report, published in the Journal of Clinical Lipidology.
“The fact that the difference in IMT increases with age between FH patients and unaffected controls, and is more pronounced in studies with untreated FH patients than in studies with treated patients, suggests that starting treatment already at a young age in patients with FH is preferred,” the researchers wrote. “However, despite treatment, IMT in treated FH patients is still thicker in comparison to subjects without FH.”
The researchers identified 42 studies with among patients with FH and healthy control groups across the MEDLINE, EMBASE and Trials.gov databases up to a cutoff date of April 2020, with 39 studies specifically examining carotid IMT, 2 studies evaluating carotid and femoral IMT, and 1 study evaluating femoral IMT alone. Overall, the researchers examined IMT measurements in 3,796 patients with FH and 2,363 control group participants.
Although data on age and gender for FH and control groups were not available in 6 studies, the mean age ranged from 9 to 57 years for patients with FH and from 8 to 61 years in the control group. Men comprised just under half of both the FH and control groups.
The mean between-group difference in carotid IMT in 34 studies was 0.11 mm (95% confidence interval, 0.06-0.15 mm; P < .001) for patients with FH, compared with the control group, while the mean difference in femoral IMT in three studies was 0.47 mm (95% CI, 0.19-0.74 mm; P < .001) between FH and control groups.
In 13 studies in which data on differences between partly treated and untreated FH were available, there was a significant between-group difference in carotid IMT with partly treated patients with FH, compared with the control group (0.05 mm; 95% CI, 0.03-0.08 mm; P < .001), but a larger mean between-group difference in carotid IMT among untreated patients with FH, compared with a control group (0.12 mm; 95% CI, 0.03-0.21 mm; P = .009).
The researchers also analyzed how age impacts carotid IMT, and they found patients with FH had a mean increase of 0.0018 mm (95% CI, –0.0007 to 0.0042 mm) over a control group in 34 studies. For patients with partly treated FH, compared with patients with untreated FH, the mean between-group increase per year was smaller (0.0023 mm; 95% CI, 0.0021-0.0025 mm), compared with the control group (0.0104 mm; 95% CI, 0.0100-0.0108 mm).
“This sign of residual risk might suggest that more robust cholesterol-lowering treatment and achieving treatment targets, or earlier treatment initiation, is needed to reduce IMT progression to non-FH conditions,” the researchers said. “Therefore, we must find and diagnose these patients, and treat them according to current guidelines.”
Limitations of the authors’ meta-analyses include heterogeneity among studies, differences in IMT measurement protocols, and inclusion of studies with an open-label design. Although randomized clinical trials would be preferable to compare treatment effect, “since statin therapy is indicated in FH patients to reduce [cardiovascular disease], it would be unethical to have a placebo group,” they said.
The authors reported no relevant financial disclosures.
Patients with familial hypercholesterolemia (FH) who start lipid-lowering treatment earlier in life may reduce their cardiovascular risk, compared with those who don’t begin treatment early, according to results of a recent meta-analysis.
They showed a difference in the carotid intima-media thickness (IMT) between patients with and without FH that increased with age, but there was also a difference in IMT seen among patients with FH who started treatment early, compared with untreated patients with FH, Kika van Bergen en Henegouwen, of the departments of pediatrics and epidemiology and data science at Amsterdam University Medical Center, and colleagues wrote in their report, published in the Journal of Clinical Lipidology.
“The fact that the difference in IMT increases with age between FH patients and unaffected controls, and is more pronounced in studies with untreated FH patients than in studies with treated patients, suggests that starting treatment already at a young age in patients with FH is preferred,” the researchers wrote. “However, despite treatment, IMT in treated FH patients is still thicker in comparison to subjects without FH.”
The researchers identified 42 studies with among patients with FH and healthy control groups across the MEDLINE, EMBASE and Trials.gov databases up to a cutoff date of April 2020, with 39 studies specifically examining carotid IMT, 2 studies evaluating carotid and femoral IMT, and 1 study evaluating femoral IMT alone. Overall, the researchers examined IMT measurements in 3,796 patients with FH and 2,363 control group participants.
Although data on age and gender for FH and control groups were not available in 6 studies, the mean age ranged from 9 to 57 years for patients with FH and from 8 to 61 years in the control group. Men comprised just under half of both the FH and control groups.
The mean between-group difference in carotid IMT in 34 studies was 0.11 mm (95% confidence interval, 0.06-0.15 mm; P < .001) for patients with FH, compared with the control group, while the mean difference in femoral IMT in three studies was 0.47 mm (95% CI, 0.19-0.74 mm; P < .001) between FH and control groups.
In 13 studies in which data on differences between partly treated and untreated FH were available, there was a significant between-group difference in carotid IMT with partly treated patients with FH, compared with the control group (0.05 mm; 95% CI, 0.03-0.08 mm; P < .001), but a larger mean between-group difference in carotid IMT among untreated patients with FH, compared with a control group (0.12 mm; 95% CI, 0.03-0.21 mm; P = .009).
The researchers also analyzed how age impacts carotid IMT, and they found patients with FH had a mean increase of 0.0018 mm (95% CI, –0.0007 to 0.0042 mm) over a control group in 34 studies. For patients with partly treated FH, compared with patients with untreated FH, the mean between-group increase per year was smaller (0.0023 mm; 95% CI, 0.0021-0.0025 mm), compared with the control group (0.0104 mm; 95% CI, 0.0100-0.0108 mm).
“This sign of residual risk might suggest that more robust cholesterol-lowering treatment and achieving treatment targets, or earlier treatment initiation, is needed to reduce IMT progression to non-FH conditions,” the researchers said. “Therefore, we must find and diagnose these patients, and treat them according to current guidelines.”
Limitations of the authors’ meta-analyses include heterogeneity among studies, differences in IMT measurement protocols, and inclusion of studies with an open-label design. Although randomized clinical trials would be preferable to compare treatment effect, “since statin therapy is indicated in FH patients to reduce [cardiovascular disease], it would be unethical to have a placebo group,” they said.
The authors reported no relevant financial disclosures.
FROM THE JOURNAL OF CLINICAL LIPIDOLOGY
ARBs and cancer risk: New meta-analysis raises questions again
The debate on whether the popular class of antihypertensive drugs, angiotensin receptor blockers (ARBs), may be associated with an increased risk for cancer has been reopened with the publication of a new meta-analysis.
The analysis found an increasing risk for cancer, and specifically lung cancer, with increasing cumulative exposure to these drugs.
The findings are reported in a study published online in PLOS ONE.
The author of this new meta-analysis is Ilke Sipahi, MD, a cardiologist from Acibadem University Medical School, Istanbul, who previously raised this issue in an initial meta-analysis published in 2010.
“The new meta-analysis is important because it is the first study to investigate whether there is a dose response in the association between ARBs and cancer,” Dr. Sipahi told this news organization.
“I found a clear signal of increased risk of cancer as exposure to ARBs increased, and the association started to become significant when the maximum dose was taken for 3 years,” he added.
Dr. Sipahi explained that in the first meta-analysis published in Lancet Oncology, he and his colleagues reported an increased cancer risk with ARBs based on observations from high-exposure trials – those that included higher doses of ARBs with a long duration of follow-up.
Following this publication, an investigation by the U.S. Food and Drug Administration refuted the risk, and a collaboration of ARB trial investigators also performed an analysis published in the Journal of Hypertension (2011. doi: 10.1097/HJH.0b013e328344a7de), which again did not show an increased risk for cancer with use of ARBs.
Dr. Sipahi claims that those analyses by the FDA and the ARB Trialists Collaboration, which were all trial-level meta-analyses, diluted the “high exposure” data (including higher doses taken for longer periods of time) with a large amount of other data on much lower exposures (lower doses and/or shorter time periods).
“The overall risk would then inevitably become nonsignificant. These analyses also did not look at different exposure levels,” he says.
“For cancer, the degree of exposure is obviously very important. The risk associated with smoking 2 or 3 cigarettes a day for a year is very different from that of smoking 2 packs a day for 40 years. The same principle applies to taking a medication,” Dr. Sipahi asserts.
From these latest data, he estimates that 120 patients needed to be treated with the maximal daily dose of an ARB for 4.7 years for one excess cancer diagnosis, and 464 patients needed to be treated for one excess lung cancer.
“Given that at least 200 million individuals are being treated with an ARB globally, approximately 1.7 million excess cancers (and 430,000 lung cancers) in 4.6 years could be potentially caused by this class of drugs,” he suggests.
For the current analysis, Dr. Sipahi used trial-level data taken from the paper by the ARB Trialists Collaboration and investigated the effect of exposure to ARBs – including both the dose taken and the length of treatment – on risk for cancer. He performed metaregression analyses that he says has not been done before.
“I mathematically quantitated the degree of exposure in each trial. And when the degree of exposure was correlated with risk of cancer, there was a significant association.”
The new meta-analysis includes 15 randomized controlled trials. The two coprimary outcomes were the relationship between cumulative exposure to ARBs and risk for all cancers combined and the relationship between cumulative exposure and risk for lung cancer.
In the trials, 74,021 patients were randomly assigned to an ARB, resulting in a total cumulative exposure of 172,389 person-years of exposure to daily high dose (or equivalent), and 61,197 patients were randomly assigned to control.
Results showed a highly significant correlation between the degree of cumulative exposure to ARBs and risk for all cancers combined (slope = 0.07; 95% confidence interval, 0.03-0.11; P < .001) and also lung cancer (slope = 0.16; 95% CI, 0.05-0.27; P = .003).
In trials where the cumulative exposure was greater than 3 years of exposure to daily high dose, there was a statistically significant increase in risk for all cancers combined (risk ratio, 1.11; 95% CI, 1.03-1.19; P = .006).
There was also a statistically significant increase in risk for lung cancers in trials where the cumulative exposure was greater than 2.5 years (RR, 1.21; 95% CI, 1.02-1.44; P = .03).
In trials with lower cumulative exposure to ARBs, there was no increased risk either for all cancers combined or lung cancer.
Dr. Sipahi reports that the cumulative exposure-risk relationship with ARBs was independent of background angiotensin-converting enzyme (ACE) inhibitor treatment or the type of control (placebo or nonplacebo control).
But he acknowledges that since this is a trial-level analysis, the effects of patient characteristics such as age and smoking status could not be examined because of lack of patient-level data.
Dr. Sipahi says he does not know the mechanism behind these findings, but he draws attention to the recent withdrawal of several thousand lots of ARB formulations because of the presence of potentially carcinogenic impurities that have been suggested to be a byproduct of ARB synthesis.
He also claims that unlike some other classes of antihypertensives, ARBs have not been shown to reduce the risk for MI, leading him to conclude that “other classes of antihypertensives with good safety and efficacy data (such as ACE-inhibitors, calcium-channel blockers or others) should become the preferred first-line agents in the treatment of hypertension.”
Dr. Sipahi wants the FDA to reinvestigate the issue of ARBs and cancer risk using individual patient data. “They already have the patient-level data from the trials. They should look at it more carefully and look at exposure levels and how they relate to cancer risk,” he said. “And the fact that there have been studies linking high ARB exposure levels to increased cancer risk should at least get a warning on the drug labels.”
A ‘clear increase’ in risk
Dr. Sipahi also points out that a link between ARBs and cancer has been found in another meta-analysis performed in 2013 by senior FDA analyst Thomas Marciniak, MD.
“Because he worked at the FDA, [Dr.] Marciniak had access to individual patent data. This is the best type of analysis and generally produces more accurate results than a trial-level meta-analysis,” Dr. Sipahi commented.
Dr. Marciniak’s analysis, which is available on the FDA website as part of another document, was not officially published elsewhere, and no further action has been taken on the issue.
Contacted by this news organization, Dr. Marciniak, who has now retired from the FDA, said he not only conducted a patient-level meta-analysis but also followed up adverse effects reported in the trials that could have been a symptom of cancer to establish further whether the patient was later diagnosed with cancer or not.
“I used every scrap of information sent in, including serious adverse event reports. I saw a clear increase in lung cancer risk with the ARBs,” Dr. Marciniak said. He did not, however, perform a dose-response relationship analysis.
Asked why his analysis and those from Dr. Sipahi reach different conclusions to those from the ARB Trialists Collaboration and the official FDA investigations, Dr. Marciniak said: “It may be that there were too many low-exposure trials that just washed out the difference. But trial data generally do not capture adverse events such as cancer, which takes a long time to develop, very well, and if you’re not really looking for it, you’re probably not going to find it.”
Dr. Marciniak said that Dr. Sipahi’s current findings are in line with his results. “Finding a dose response, to me, is extremely compelling, and I think the signal here is real,” he commented. “I think this new paper from Dr. Sipahi verifies what I found. I think the FDA should now release all individual patient data it has.”
Contacted for comment, an FDA spokesperson said, “Generally the FDA does not comment on specific studies but evaluates them as part of the body of evidence to further our understanding about a particular issue and assist in our mission to protect public health.”
They added: “The FDA has ongoing assessment, surveillance, compliance, and pharmaceutical quality efforts across every product area, and we will continue to work with drug manufacturers to ensure safe, effective, and high-quality drugs for the American public. When we identify new and previously unrecognized risks to safety and quality, we react swiftly to resolve the problem, as we have done in responding to the recent findings of nitrosamines in certain medicines.”
Analysis ‘should be taken seriously’
Commenting on this new study, Steve Nissen, MD, a key figure in analyzing such complex data and who has himself uncovered problems with high-profile drugs in the past, says the current analysis should be taken seriously.
Dr. Nissen, who was Dr. Sipahi’s senior during his post-doc position at the Cleveland Clinic, wrote an editorial accompanying Dr. Sipahi’s first paper and calling for urgent regulatory review of the evidence.
He says the new findings add to previous evidence suggesting a possible risk for cancer with ARBs.
“[Dr.] Sipahi is a capable researcher, and this analysis needs to be taken seriously, but it needs to be verified. It is not possible to draw a strong conclusion on this analysis, as it is not based on individual patient data, but I don’t think it should be ignored,” Dr. Nissen stated.
“I will say again what I said 12 years ago – that the regulatory agencies need to carefully review all their data in a very detailed way. The FDA and EMA have access to the individual patient data and are both very capable of doing the required analyses.”
Limitations of trial-level analysis
Asked to evaluate the statistics in the current paper, Andrew Althouse, PhD, an assistant professor of medicine at the University of Pittsburgh, and a clinical trial statistician, explained that the best way to do a thorough analysis of the relationship between ARB exposure and risk for incident cancer would involve the use of patient-level data.
“As such data were not available to Dr. Sipahi, I believe he is doing as well as he can. But without full access to individual patient-level data from the respective trials, it is difficult to support any firm conclusions,” Dr. Althouse said in an interview.
He suggested that the meta-regression analyses used in the paper were unable to properly estimate the relationship between ARB exposure and risk for incident cancer.
“Taken at face value, the current analysis suggests that [in] trials with longer follow-up duration (and therefore greater cumulative exposure to ARB for the treatment group), the risk of developing cancer for patients in the ARB group versus the non-ARB group was progressively higher. But this study doesn’t take into account the actual amount of follow-up time for individual patients or potential differences in the amount of follow-up time between the two groups in each trial,” he noted.
Dr. Althouse says this raises the possibility of “competing risks” or the idea that if ARBs reduce cardiovascular disease and cardiovascular death, then there would be more patients remaining in that arm who could go on to develop cancer. “So a crude count of the number of cancer cases may look as though patients receiving ARBs are ‘more likely’ to develop cancer, but this is a mirage.”
He added: “When there are some patients dying during the study, the only way to tell whether the intervention actually increased the risk of other health-related complications is to have an analysis that properly accounts for each patient’s time-at-risk of the outcome. Unfortunately, properly analyzing this requires the use of patient-level data.”
Cardiologists skeptical?
Cardiology experts asked for thoughts on the new meta-analysis were also cautious to read too much into the findings.
Franz Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “Perhaps one would simply ignore this rambling, cherrypicking-based condemnation of ARBs if it were not for the powerful negative connotation of the word cancer. Thus, the meta-analysis of Dr. Sipahi purporting that ARBs could be increasing the development of cancers in a cumulative way is of concern to both physicians and patients.”
But, raising a similar point to Dr. Althouse about competing risks, Dr. Messerli said: “We have to consider that as one gets older, the cardiovascular disease state and cancer state will compete with each other for the outcome of death. The better that therapies protect against cardiovascular death, the more they will increase life expectancy and thus the risk of cancer.”
He also added that “in head-to-head comparisons with ACE inhibitors, ARBs showed similar efficacy in terms of death, CV mortality, MI, stroke, and end-stage kidney disease, so can we agree that the attempt of Dr. Sipahi to disparage ARBs as a class is much ado about nothing?”
Dr. Nissen, however, said he views the idea of competing risk as “a bit of a stretch” in this case. “Although ARBs are effective antihypertensive drugs, I would say there is very little evidence that they would prolong survival versus other antihypertensives.”
Dr. Sipahi also claims that this argument is not relevant to the current analysis. “ARBs did not increase survival in any of the high-exposure trials that showed an excess in cancers. Therefore, competing outcomes, or ‘survival bias’ to be more specific, is not a possibility here,” he says.
George Bakris, MD, professor of medicine at the University of Chicago Medicine, noted that while the current study shows a slight increase in cancer incidence, especially lung cancer, among those taking ARBs for more than 3 years, it “totally ignores the overwhelming cardiovascular risk reduction seen in the trials.”
“Moreover,” he adds, “the author notes that the findings were independent of ACE-inhibitors, but he can’t rule out smoking and age as factors, two major risk factors for cancer and lung cancer, specifically. Thus, as typical of these types of analyses, the associations are probably true/true unrelated or, at best, partially related.”
Dr. Bakris referred to the potentially carcinogenic nitrosamine and azido compounds found in several ARB formulations that have resulted in recalls.
“At any stage of drug synthesis throughout each product’s lifetime, these impurities may evolve if an amine reacts with a nitrosating agent coexisting under appropriate conditions,” he said. “Drug regulatory authorities worldwide have established stringent guidelines on nitrosamine contamination for all drug products. The studies noted in the author’s analysis were done well before these guidelines were implemented. Hence, many of the issues raised by the authors using trials from 10-20 years ago are not of significant concern.”
Still, the cardiology experts all agreed on one thing – that patients should continue to take ARBs as prescribed.
Noting that worldwide authorities are now addressing the issue of possible carcinogen contamination, Dr. Bakris stressed that patients “should not panic and should not stop their meds.”
Dr. Nissen added: “What we don’t want is for patents who are taking ARBs to stop taking these medications – hypertension is a deadly disorder, and these drugs have proven cardiovascular benefits.”
Dr. Sipahi received no specific funding for this work. He reports receiving lecture honoraria from Novartis, Boehringer Ingelheim, Sanofi, Sandoz, Bristol-Myers Squibb, Bayer, Pfizer, Ranbaxy, Servier, and ARIS and served on advisory boards for Novartis, Sanofi, Servier, Bristol-Myers Squibb, Pfizer, Bayer and I.E. Ulagay. The other commenters do not report any relevant disclosures.
A version of this article first appeared on Medscape.com.
The debate on whether the popular class of antihypertensive drugs, angiotensin receptor blockers (ARBs), may be associated with an increased risk for cancer has been reopened with the publication of a new meta-analysis.
The analysis found an increasing risk for cancer, and specifically lung cancer, with increasing cumulative exposure to these drugs.
The findings are reported in a study published online in PLOS ONE.
The author of this new meta-analysis is Ilke Sipahi, MD, a cardiologist from Acibadem University Medical School, Istanbul, who previously raised this issue in an initial meta-analysis published in 2010.
“The new meta-analysis is important because it is the first study to investigate whether there is a dose response in the association between ARBs and cancer,” Dr. Sipahi told this news organization.
“I found a clear signal of increased risk of cancer as exposure to ARBs increased, and the association started to become significant when the maximum dose was taken for 3 years,” he added.
Dr. Sipahi explained that in the first meta-analysis published in Lancet Oncology, he and his colleagues reported an increased cancer risk with ARBs based on observations from high-exposure trials – those that included higher doses of ARBs with a long duration of follow-up.
Following this publication, an investigation by the U.S. Food and Drug Administration refuted the risk, and a collaboration of ARB trial investigators also performed an analysis published in the Journal of Hypertension (2011. doi: 10.1097/HJH.0b013e328344a7de), which again did not show an increased risk for cancer with use of ARBs.
Dr. Sipahi claims that those analyses by the FDA and the ARB Trialists Collaboration, which were all trial-level meta-analyses, diluted the “high exposure” data (including higher doses taken for longer periods of time) with a large amount of other data on much lower exposures (lower doses and/or shorter time periods).
“The overall risk would then inevitably become nonsignificant. These analyses also did not look at different exposure levels,” he says.
“For cancer, the degree of exposure is obviously very important. The risk associated with smoking 2 or 3 cigarettes a day for a year is very different from that of smoking 2 packs a day for 40 years. The same principle applies to taking a medication,” Dr. Sipahi asserts.
From these latest data, he estimates that 120 patients needed to be treated with the maximal daily dose of an ARB for 4.7 years for one excess cancer diagnosis, and 464 patients needed to be treated for one excess lung cancer.
“Given that at least 200 million individuals are being treated with an ARB globally, approximately 1.7 million excess cancers (and 430,000 lung cancers) in 4.6 years could be potentially caused by this class of drugs,” he suggests.
For the current analysis, Dr. Sipahi used trial-level data taken from the paper by the ARB Trialists Collaboration and investigated the effect of exposure to ARBs – including both the dose taken and the length of treatment – on risk for cancer. He performed metaregression analyses that he says has not been done before.
“I mathematically quantitated the degree of exposure in each trial. And when the degree of exposure was correlated with risk of cancer, there was a significant association.”
The new meta-analysis includes 15 randomized controlled trials. The two coprimary outcomes were the relationship between cumulative exposure to ARBs and risk for all cancers combined and the relationship between cumulative exposure and risk for lung cancer.
In the trials, 74,021 patients were randomly assigned to an ARB, resulting in a total cumulative exposure of 172,389 person-years of exposure to daily high dose (or equivalent), and 61,197 patients were randomly assigned to control.
Results showed a highly significant correlation between the degree of cumulative exposure to ARBs and risk for all cancers combined (slope = 0.07; 95% confidence interval, 0.03-0.11; P < .001) and also lung cancer (slope = 0.16; 95% CI, 0.05-0.27; P = .003).
In trials where the cumulative exposure was greater than 3 years of exposure to daily high dose, there was a statistically significant increase in risk for all cancers combined (risk ratio, 1.11; 95% CI, 1.03-1.19; P = .006).
There was also a statistically significant increase in risk for lung cancers in trials where the cumulative exposure was greater than 2.5 years (RR, 1.21; 95% CI, 1.02-1.44; P = .03).
In trials with lower cumulative exposure to ARBs, there was no increased risk either for all cancers combined or lung cancer.
Dr. Sipahi reports that the cumulative exposure-risk relationship with ARBs was independent of background angiotensin-converting enzyme (ACE) inhibitor treatment or the type of control (placebo or nonplacebo control).
But he acknowledges that since this is a trial-level analysis, the effects of patient characteristics such as age and smoking status could not be examined because of lack of patient-level data.
Dr. Sipahi says he does not know the mechanism behind these findings, but he draws attention to the recent withdrawal of several thousand lots of ARB formulations because of the presence of potentially carcinogenic impurities that have been suggested to be a byproduct of ARB synthesis.
He also claims that unlike some other classes of antihypertensives, ARBs have not been shown to reduce the risk for MI, leading him to conclude that “other classes of antihypertensives with good safety and efficacy data (such as ACE-inhibitors, calcium-channel blockers or others) should become the preferred first-line agents in the treatment of hypertension.”
Dr. Sipahi wants the FDA to reinvestigate the issue of ARBs and cancer risk using individual patient data. “They already have the patient-level data from the trials. They should look at it more carefully and look at exposure levels and how they relate to cancer risk,” he said. “And the fact that there have been studies linking high ARB exposure levels to increased cancer risk should at least get a warning on the drug labels.”
A ‘clear increase’ in risk
Dr. Sipahi also points out that a link between ARBs and cancer has been found in another meta-analysis performed in 2013 by senior FDA analyst Thomas Marciniak, MD.
“Because he worked at the FDA, [Dr.] Marciniak had access to individual patent data. This is the best type of analysis and generally produces more accurate results than a trial-level meta-analysis,” Dr. Sipahi commented.
Dr. Marciniak’s analysis, which is available on the FDA website as part of another document, was not officially published elsewhere, and no further action has been taken on the issue.
Contacted by this news organization, Dr. Marciniak, who has now retired from the FDA, said he not only conducted a patient-level meta-analysis but also followed up adverse effects reported in the trials that could have been a symptom of cancer to establish further whether the patient was later diagnosed with cancer or not.
“I used every scrap of information sent in, including serious adverse event reports. I saw a clear increase in lung cancer risk with the ARBs,” Dr. Marciniak said. He did not, however, perform a dose-response relationship analysis.
Asked why his analysis and those from Dr. Sipahi reach different conclusions to those from the ARB Trialists Collaboration and the official FDA investigations, Dr. Marciniak said: “It may be that there were too many low-exposure trials that just washed out the difference. But trial data generally do not capture adverse events such as cancer, which takes a long time to develop, very well, and if you’re not really looking for it, you’re probably not going to find it.”
Dr. Marciniak said that Dr. Sipahi’s current findings are in line with his results. “Finding a dose response, to me, is extremely compelling, and I think the signal here is real,” he commented. “I think this new paper from Dr. Sipahi verifies what I found. I think the FDA should now release all individual patient data it has.”
Contacted for comment, an FDA spokesperson said, “Generally the FDA does not comment on specific studies but evaluates them as part of the body of evidence to further our understanding about a particular issue and assist in our mission to protect public health.”
They added: “The FDA has ongoing assessment, surveillance, compliance, and pharmaceutical quality efforts across every product area, and we will continue to work with drug manufacturers to ensure safe, effective, and high-quality drugs for the American public. When we identify new and previously unrecognized risks to safety and quality, we react swiftly to resolve the problem, as we have done in responding to the recent findings of nitrosamines in certain medicines.”
Analysis ‘should be taken seriously’
Commenting on this new study, Steve Nissen, MD, a key figure in analyzing such complex data and who has himself uncovered problems with high-profile drugs in the past, says the current analysis should be taken seriously.
Dr. Nissen, who was Dr. Sipahi’s senior during his post-doc position at the Cleveland Clinic, wrote an editorial accompanying Dr. Sipahi’s first paper and calling for urgent regulatory review of the evidence.
He says the new findings add to previous evidence suggesting a possible risk for cancer with ARBs.
“[Dr.] Sipahi is a capable researcher, and this analysis needs to be taken seriously, but it needs to be verified. It is not possible to draw a strong conclusion on this analysis, as it is not based on individual patient data, but I don’t think it should be ignored,” Dr. Nissen stated.
“I will say again what I said 12 years ago – that the regulatory agencies need to carefully review all their data in a very detailed way. The FDA and EMA have access to the individual patient data and are both very capable of doing the required analyses.”
Limitations of trial-level analysis
Asked to evaluate the statistics in the current paper, Andrew Althouse, PhD, an assistant professor of medicine at the University of Pittsburgh, and a clinical trial statistician, explained that the best way to do a thorough analysis of the relationship between ARB exposure and risk for incident cancer would involve the use of patient-level data.
“As such data were not available to Dr. Sipahi, I believe he is doing as well as he can. But without full access to individual patient-level data from the respective trials, it is difficult to support any firm conclusions,” Dr. Althouse said in an interview.
He suggested that the meta-regression analyses used in the paper were unable to properly estimate the relationship between ARB exposure and risk for incident cancer.
“Taken at face value, the current analysis suggests that [in] trials with longer follow-up duration (and therefore greater cumulative exposure to ARB for the treatment group), the risk of developing cancer for patients in the ARB group versus the non-ARB group was progressively higher. But this study doesn’t take into account the actual amount of follow-up time for individual patients or potential differences in the amount of follow-up time between the two groups in each trial,” he noted.
Dr. Althouse says this raises the possibility of “competing risks” or the idea that if ARBs reduce cardiovascular disease and cardiovascular death, then there would be more patients remaining in that arm who could go on to develop cancer. “So a crude count of the number of cancer cases may look as though patients receiving ARBs are ‘more likely’ to develop cancer, but this is a mirage.”
He added: “When there are some patients dying during the study, the only way to tell whether the intervention actually increased the risk of other health-related complications is to have an analysis that properly accounts for each patient’s time-at-risk of the outcome. Unfortunately, properly analyzing this requires the use of patient-level data.”
Cardiologists skeptical?
Cardiology experts asked for thoughts on the new meta-analysis were also cautious to read too much into the findings.
Franz Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “Perhaps one would simply ignore this rambling, cherrypicking-based condemnation of ARBs if it were not for the powerful negative connotation of the word cancer. Thus, the meta-analysis of Dr. Sipahi purporting that ARBs could be increasing the development of cancers in a cumulative way is of concern to both physicians and patients.”
But, raising a similar point to Dr. Althouse about competing risks, Dr. Messerli said: “We have to consider that as one gets older, the cardiovascular disease state and cancer state will compete with each other for the outcome of death. The better that therapies protect against cardiovascular death, the more they will increase life expectancy and thus the risk of cancer.”
He also added that “in head-to-head comparisons with ACE inhibitors, ARBs showed similar efficacy in terms of death, CV mortality, MI, stroke, and end-stage kidney disease, so can we agree that the attempt of Dr. Sipahi to disparage ARBs as a class is much ado about nothing?”
Dr. Nissen, however, said he views the idea of competing risk as “a bit of a stretch” in this case. “Although ARBs are effective antihypertensive drugs, I would say there is very little evidence that they would prolong survival versus other antihypertensives.”
Dr. Sipahi also claims that this argument is not relevant to the current analysis. “ARBs did not increase survival in any of the high-exposure trials that showed an excess in cancers. Therefore, competing outcomes, or ‘survival bias’ to be more specific, is not a possibility here,” he says.
George Bakris, MD, professor of medicine at the University of Chicago Medicine, noted that while the current study shows a slight increase in cancer incidence, especially lung cancer, among those taking ARBs for more than 3 years, it “totally ignores the overwhelming cardiovascular risk reduction seen in the trials.”
“Moreover,” he adds, “the author notes that the findings were independent of ACE-inhibitors, but he can’t rule out smoking and age as factors, two major risk factors for cancer and lung cancer, specifically. Thus, as typical of these types of analyses, the associations are probably true/true unrelated or, at best, partially related.”
Dr. Bakris referred to the potentially carcinogenic nitrosamine and azido compounds found in several ARB formulations that have resulted in recalls.
“At any stage of drug synthesis throughout each product’s lifetime, these impurities may evolve if an amine reacts with a nitrosating agent coexisting under appropriate conditions,” he said. “Drug regulatory authorities worldwide have established stringent guidelines on nitrosamine contamination for all drug products. The studies noted in the author’s analysis were done well before these guidelines were implemented. Hence, many of the issues raised by the authors using trials from 10-20 years ago are not of significant concern.”
Still, the cardiology experts all agreed on one thing – that patients should continue to take ARBs as prescribed.
Noting that worldwide authorities are now addressing the issue of possible carcinogen contamination, Dr. Bakris stressed that patients “should not panic and should not stop their meds.”
Dr. Nissen added: “What we don’t want is for patents who are taking ARBs to stop taking these medications – hypertension is a deadly disorder, and these drugs have proven cardiovascular benefits.”
Dr. Sipahi received no specific funding for this work. He reports receiving lecture honoraria from Novartis, Boehringer Ingelheim, Sanofi, Sandoz, Bristol-Myers Squibb, Bayer, Pfizer, Ranbaxy, Servier, and ARIS and served on advisory boards for Novartis, Sanofi, Servier, Bristol-Myers Squibb, Pfizer, Bayer and I.E. Ulagay. The other commenters do not report any relevant disclosures.
A version of this article first appeared on Medscape.com.
The debate on whether the popular class of antihypertensive drugs, angiotensin receptor blockers (ARBs), may be associated with an increased risk for cancer has been reopened with the publication of a new meta-analysis.
The analysis found an increasing risk for cancer, and specifically lung cancer, with increasing cumulative exposure to these drugs.
The findings are reported in a study published online in PLOS ONE.
The author of this new meta-analysis is Ilke Sipahi, MD, a cardiologist from Acibadem University Medical School, Istanbul, who previously raised this issue in an initial meta-analysis published in 2010.
“The new meta-analysis is important because it is the first study to investigate whether there is a dose response in the association between ARBs and cancer,” Dr. Sipahi told this news organization.
“I found a clear signal of increased risk of cancer as exposure to ARBs increased, and the association started to become significant when the maximum dose was taken for 3 years,” he added.
Dr. Sipahi explained that in the first meta-analysis published in Lancet Oncology, he and his colleagues reported an increased cancer risk with ARBs based on observations from high-exposure trials – those that included higher doses of ARBs with a long duration of follow-up.
Following this publication, an investigation by the U.S. Food and Drug Administration refuted the risk, and a collaboration of ARB trial investigators also performed an analysis published in the Journal of Hypertension (2011. doi: 10.1097/HJH.0b013e328344a7de), which again did not show an increased risk for cancer with use of ARBs.
Dr. Sipahi claims that those analyses by the FDA and the ARB Trialists Collaboration, which were all trial-level meta-analyses, diluted the “high exposure” data (including higher doses taken for longer periods of time) with a large amount of other data on much lower exposures (lower doses and/or shorter time periods).
“The overall risk would then inevitably become nonsignificant. These analyses also did not look at different exposure levels,” he says.
“For cancer, the degree of exposure is obviously very important. The risk associated with smoking 2 or 3 cigarettes a day for a year is very different from that of smoking 2 packs a day for 40 years. The same principle applies to taking a medication,” Dr. Sipahi asserts.
From these latest data, he estimates that 120 patients needed to be treated with the maximal daily dose of an ARB for 4.7 years for one excess cancer diagnosis, and 464 patients needed to be treated for one excess lung cancer.
“Given that at least 200 million individuals are being treated with an ARB globally, approximately 1.7 million excess cancers (and 430,000 lung cancers) in 4.6 years could be potentially caused by this class of drugs,” he suggests.
For the current analysis, Dr. Sipahi used trial-level data taken from the paper by the ARB Trialists Collaboration and investigated the effect of exposure to ARBs – including both the dose taken and the length of treatment – on risk for cancer. He performed metaregression analyses that he says has not been done before.
“I mathematically quantitated the degree of exposure in each trial. And when the degree of exposure was correlated with risk of cancer, there was a significant association.”
The new meta-analysis includes 15 randomized controlled trials. The two coprimary outcomes were the relationship between cumulative exposure to ARBs and risk for all cancers combined and the relationship between cumulative exposure and risk for lung cancer.
In the trials, 74,021 patients were randomly assigned to an ARB, resulting in a total cumulative exposure of 172,389 person-years of exposure to daily high dose (or equivalent), and 61,197 patients were randomly assigned to control.
Results showed a highly significant correlation between the degree of cumulative exposure to ARBs and risk for all cancers combined (slope = 0.07; 95% confidence interval, 0.03-0.11; P < .001) and also lung cancer (slope = 0.16; 95% CI, 0.05-0.27; P = .003).
In trials where the cumulative exposure was greater than 3 years of exposure to daily high dose, there was a statistically significant increase in risk for all cancers combined (risk ratio, 1.11; 95% CI, 1.03-1.19; P = .006).
There was also a statistically significant increase in risk for lung cancers in trials where the cumulative exposure was greater than 2.5 years (RR, 1.21; 95% CI, 1.02-1.44; P = .03).
In trials with lower cumulative exposure to ARBs, there was no increased risk either for all cancers combined or lung cancer.
Dr. Sipahi reports that the cumulative exposure-risk relationship with ARBs was independent of background angiotensin-converting enzyme (ACE) inhibitor treatment or the type of control (placebo or nonplacebo control).
But he acknowledges that since this is a trial-level analysis, the effects of patient characteristics such as age and smoking status could not be examined because of lack of patient-level data.
Dr. Sipahi says he does not know the mechanism behind these findings, but he draws attention to the recent withdrawal of several thousand lots of ARB formulations because of the presence of potentially carcinogenic impurities that have been suggested to be a byproduct of ARB synthesis.
He also claims that unlike some other classes of antihypertensives, ARBs have not been shown to reduce the risk for MI, leading him to conclude that “other classes of antihypertensives with good safety and efficacy data (such as ACE-inhibitors, calcium-channel blockers or others) should become the preferred first-line agents in the treatment of hypertension.”
Dr. Sipahi wants the FDA to reinvestigate the issue of ARBs and cancer risk using individual patient data. “They already have the patient-level data from the trials. They should look at it more carefully and look at exposure levels and how they relate to cancer risk,” he said. “And the fact that there have been studies linking high ARB exposure levels to increased cancer risk should at least get a warning on the drug labels.”
A ‘clear increase’ in risk
Dr. Sipahi also points out that a link between ARBs and cancer has been found in another meta-analysis performed in 2013 by senior FDA analyst Thomas Marciniak, MD.
“Because he worked at the FDA, [Dr.] Marciniak had access to individual patent data. This is the best type of analysis and generally produces more accurate results than a trial-level meta-analysis,” Dr. Sipahi commented.
Dr. Marciniak’s analysis, which is available on the FDA website as part of another document, was not officially published elsewhere, and no further action has been taken on the issue.
Contacted by this news organization, Dr. Marciniak, who has now retired from the FDA, said he not only conducted a patient-level meta-analysis but also followed up adverse effects reported in the trials that could have been a symptom of cancer to establish further whether the patient was later diagnosed with cancer or not.
“I used every scrap of information sent in, including serious adverse event reports. I saw a clear increase in lung cancer risk with the ARBs,” Dr. Marciniak said. He did not, however, perform a dose-response relationship analysis.
Asked why his analysis and those from Dr. Sipahi reach different conclusions to those from the ARB Trialists Collaboration and the official FDA investigations, Dr. Marciniak said: “It may be that there were too many low-exposure trials that just washed out the difference. But trial data generally do not capture adverse events such as cancer, which takes a long time to develop, very well, and if you’re not really looking for it, you’re probably not going to find it.”
Dr. Marciniak said that Dr. Sipahi’s current findings are in line with his results. “Finding a dose response, to me, is extremely compelling, and I think the signal here is real,” he commented. “I think this new paper from Dr. Sipahi verifies what I found. I think the FDA should now release all individual patient data it has.”
Contacted for comment, an FDA spokesperson said, “Generally the FDA does not comment on specific studies but evaluates them as part of the body of evidence to further our understanding about a particular issue and assist in our mission to protect public health.”
They added: “The FDA has ongoing assessment, surveillance, compliance, and pharmaceutical quality efforts across every product area, and we will continue to work with drug manufacturers to ensure safe, effective, and high-quality drugs for the American public. When we identify new and previously unrecognized risks to safety and quality, we react swiftly to resolve the problem, as we have done in responding to the recent findings of nitrosamines in certain medicines.”
Analysis ‘should be taken seriously’
Commenting on this new study, Steve Nissen, MD, a key figure in analyzing such complex data and who has himself uncovered problems with high-profile drugs in the past, says the current analysis should be taken seriously.
Dr. Nissen, who was Dr. Sipahi’s senior during his post-doc position at the Cleveland Clinic, wrote an editorial accompanying Dr. Sipahi’s first paper and calling for urgent regulatory review of the evidence.
He says the new findings add to previous evidence suggesting a possible risk for cancer with ARBs.
“[Dr.] Sipahi is a capable researcher, and this analysis needs to be taken seriously, but it needs to be verified. It is not possible to draw a strong conclusion on this analysis, as it is not based on individual patient data, but I don’t think it should be ignored,” Dr. Nissen stated.
“I will say again what I said 12 years ago – that the regulatory agencies need to carefully review all their data in a very detailed way. The FDA and EMA have access to the individual patient data and are both very capable of doing the required analyses.”
Limitations of trial-level analysis
Asked to evaluate the statistics in the current paper, Andrew Althouse, PhD, an assistant professor of medicine at the University of Pittsburgh, and a clinical trial statistician, explained that the best way to do a thorough analysis of the relationship between ARB exposure and risk for incident cancer would involve the use of patient-level data.
“As such data were not available to Dr. Sipahi, I believe he is doing as well as he can. But without full access to individual patient-level data from the respective trials, it is difficult to support any firm conclusions,” Dr. Althouse said in an interview.
He suggested that the meta-regression analyses used in the paper were unable to properly estimate the relationship between ARB exposure and risk for incident cancer.
“Taken at face value, the current analysis suggests that [in] trials with longer follow-up duration (and therefore greater cumulative exposure to ARB for the treatment group), the risk of developing cancer for patients in the ARB group versus the non-ARB group was progressively higher. But this study doesn’t take into account the actual amount of follow-up time for individual patients or potential differences in the amount of follow-up time between the two groups in each trial,” he noted.
Dr. Althouse says this raises the possibility of “competing risks” or the idea that if ARBs reduce cardiovascular disease and cardiovascular death, then there would be more patients remaining in that arm who could go on to develop cancer. “So a crude count of the number of cancer cases may look as though patients receiving ARBs are ‘more likely’ to develop cancer, but this is a mirage.”
He added: “When there are some patients dying during the study, the only way to tell whether the intervention actually increased the risk of other health-related complications is to have an analysis that properly accounts for each patient’s time-at-risk of the outcome. Unfortunately, properly analyzing this requires the use of patient-level data.”
Cardiologists skeptical?
Cardiology experts asked for thoughts on the new meta-analysis were also cautious to read too much into the findings.
Franz Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “Perhaps one would simply ignore this rambling, cherrypicking-based condemnation of ARBs if it were not for the powerful negative connotation of the word cancer. Thus, the meta-analysis of Dr. Sipahi purporting that ARBs could be increasing the development of cancers in a cumulative way is of concern to both physicians and patients.”
But, raising a similar point to Dr. Althouse about competing risks, Dr. Messerli said: “We have to consider that as one gets older, the cardiovascular disease state and cancer state will compete with each other for the outcome of death. The better that therapies protect against cardiovascular death, the more they will increase life expectancy and thus the risk of cancer.”
He also added that “in head-to-head comparisons with ACE inhibitors, ARBs showed similar efficacy in terms of death, CV mortality, MI, stroke, and end-stage kidney disease, so can we agree that the attempt of Dr. Sipahi to disparage ARBs as a class is much ado about nothing?”
Dr. Nissen, however, said he views the idea of competing risk as “a bit of a stretch” in this case. “Although ARBs are effective antihypertensive drugs, I would say there is very little evidence that they would prolong survival versus other antihypertensives.”
Dr. Sipahi also claims that this argument is not relevant to the current analysis. “ARBs did not increase survival in any of the high-exposure trials that showed an excess in cancers. Therefore, competing outcomes, or ‘survival bias’ to be more specific, is not a possibility here,” he says.
George Bakris, MD, professor of medicine at the University of Chicago Medicine, noted that while the current study shows a slight increase in cancer incidence, especially lung cancer, among those taking ARBs for more than 3 years, it “totally ignores the overwhelming cardiovascular risk reduction seen in the trials.”
“Moreover,” he adds, “the author notes that the findings were independent of ACE-inhibitors, but he can’t rule out smoking and age as factors, two major risk factors for cancer and lung cancer, specifically. Thus, as typical of these types of analyses, the associations are probably true/true unrelated or, at best, partially related.”
Dr. Bakris referred to the potentially carcinogenic nitrosamine and azido compounds found in several ARB formulations that have resulted in recalls.
“At any stage of drug synthesis throughout each product’s lifetime, these impurities may evolve if an amine reacts with a nitrosating agent coexisting under appropriate conditions,” he said. “Drug regulatory authorities worldwide have established stringent guidelines on nitrosamine contamination for all drug products. The studies noted in the author’s analysis were done well before these guidelines were implemented. Hence, many of the issues raised by the authors using trials from 10-20 years ago are not of significant concern.”
Still, the cardiology experts all agreed on one thing – that patients should continue to take ARBs as prescribed.
Noting that worldwide authorities are now addressing the issue of possible carcinogen contamination, Dr. Bakris stressed that patients “should not panic and should not stop their meds.”
Dr. Nissen added: “What we don’t want is for patents who are taking ARBs to stop taking these medications – hypertension is a deadly disorder, and these drugs have proven cardiovascular benefits.”
Dr. Sipahi received no specific funding for this work. He reports receiving lecture honoraria from Novartis, Boehringer Ingelheim, Sanofi, Sandoz, Bristol-Myers Squibb, Bayer, Pfizer, Ranbaxy, Servier, and ARIS and served on advisory boards for Novartis, Sanofi, Servier, Bristol-Myers Squibb, Pfizer, Bayer and I.E. Ulagay. The other commenters do not report any relevant disclosures.
A version of this article first appeared on Medscape.com.
Cardiac arrest survival lower in COVID-19 inpatients
Survival after in-hospital cardiac arrest was roughly one-third lower in patients with COVID-19 infections compared to uninfected patients, based on data from nearly 25,000 individuals.
Survival rates of less than 3% were reported in the United States and China for patients who suffered in-hospital cardiac arrest (IHCA) while infected with COVID-19 early in the pandemic, but the data came from small, single-center studies in overwhelmed hospitals, wrote Saket Girotra, MD, of the University of Iowa, Iowa City, and fellow American Heart Association Get With the Guidelines–Resuscitation Investigators. Whether these early reports reflect the broader experience of patients with COVID-19 in hospitals in the United States remains unknown.
In a study published as a research letter in JAMA Network Open, the researchers reviewed data from the American Heart Association Get With the Guidelines–Resuscitation registry. The registry collects detailed information on patients aged 18 years and older who experience cardiac arrest at participating hospitals in the United States. The study population included 24,915 patients aged 18 years and older from 286 hospitals who experienced IHCA during March–December 2020. The mean age of the patients was 64.7 years; 61.1% were White, 24.8% were Black, 3.8% were of other race or ethnicity, and 10.3% were of unknown race or ethnicity.
The primary outcomes were survival to discharge and return of spontaneous circulation (ROSC) for at least 20 minutes.
A total of 5,916 patients (23.7%) had suspected or confirmed COVID-19 infections, and infected patients were more likely to be younger, male, and Black. Patients with COVID-19 infections also were significantly more likely than noninfected patients to have nonshockable rhythm, pneumonia, respiratory insufficiency, or sepsis, and to be on mechanical ventilation or vasopressors when the IHCA occurred, the researchers noted.
Survival rates to hospital discharge were 11.9% for COVID-19 patients, compared with 23.5% for noninfected patients (adjusted relative risk, 0.65; P < .001). ROSC was 53.7% and 63.6%, for infected and noninfected patients, respectively (aRR, 0.86; P < .001).
COVID-19 patients also were more likely than noninfected patients to receive delayed defibrillation, the researchers said. “Although delays in resuscitation, especially defibrillation, may have contributed to lower survival, the negative association of COVID-19 with survival in this study was consistent across subgroups, including patients who received timely treatment with defibrillation and epinephrine.”
The extremely low survival rate in early pandemic studies likely reflected the overwhelming burden on health systems at the time, the researchers said in their discussion.
The study findings were limited by several factors, including potential confounding from unmeasured variables, the use of a quality improvement registry that may not reflect nonparticipating hospitals, and potential false-positive COVID-19 cases. However, the result support findings from recent studies of multiple centers and extend clinical knowledge by comparing infected and noninfected patients from a larger group of hospitals than previously studied, the researchers said.
“We believe that these data will be relevant to health care providers and hospital administrators as the COVID-19 pandemic continues,” they concluded.
Think beyond COVID-19 for cardiac care
“Early during the pandemic, questions were raised whether COVID-19 patients should be treated with CPR,” Dr. Girotra said in an interview. “This was because initial studies had found a dismal survival of 0%-3% in COVID patients treated with CPR. The potential of transmitting the virus to health care professionals during CPR further heightened these concerns. We wanted to know whether the poor survival reported in these initial studies were broadly representative.”
Dr. Girotra said that some of the study findings were surprising. “We found that of all patients with IHCA in 2020 in our study, one in four were suspected or confirmed to have COVID-19 infection. We were surprised by the magnitude of COVID’s impact on the cardiac arrest incidence.”
The implications for clinical decision-making are to think outside of COVID-19 infection, said Dr. Girotra. In the current study, “Although overall survival of cardiac arrest in COVID-positive patients was 30% lower, compared to non-COVID patients, it was not as poor as previously reported. COVID-19 infection alone should not be considered the sole factor for making decisions regarding CPR.
“Over the past 2 decades, we have experienced large gains in survival for in-hospital cardiac arrest. However, the COVID-19 pandemic has eroded these gains,” said Dr. Girotra. “Future studies are needed to monitor the impact of any new variants on cardiac arrest care,” as well as studies “to see whether we return to the prepandemic levels of IHCA survival once the pandemic recedes.”
Dr. Girotra has no relevant financial disclosures.
Survival after in-hospital cardiac arrest was roughly one-third lower in patients with COVID-19 infections compared to uninfected patients, based on data from nearly 25,000 individuals.
Survival rates of less than 3% were reported in the United States and China for patients who suffered in-hospital cardiac arrest (IHCA) while infected with COVID-19 early in the pandemic, but the data came from small, single-center studies in overwhelmed hospitals, wrote Saket Girotra, MD, of the University of Iowa, Iowa City, and fellow American Heart Association Get With the Guidelines–Resuscitation Investigators. Whether these early reports reflect the broader experience of patients with COVID-19 in hospitals in the United States remains unknown.
In a study published as a research letter in JAMA Network Open, the researchers reviewed data from the American Heart Association Get With the Guidelines–Resuscitation registry. The registry collects detailed information on patients aged 18 years and older who experience cardiac arrest at participating hospitals in the United States. The study population included 24,915 patients aged 18 years and older from 286 hospitals who experienced IHCA during March–December 2020. The mean age of the patients was 64.7 years; 61.1% were White, 24.8% were Black, 3.8% were of other race or ethnicity, and 10.3% were of unknown race or ethnicity.
The primary outcomes were survival to discharge and return of spontaneous circulation (ROSC) for at least 20 minutes.
A total of 5,916 patients (23.7%) had suspected or confirmed COVID-19 infections, and infected patients were more likely to be younger, male, and Black. Patients with COVID-19 infections also were significantly more likely than noninfected patients to have nonshockable rhythm, pneumonia, respiratory insufficiency, or sepsis, and to be on mechanical ventilation or vasopressors when the IHCA occurred, the researchers noted.
Survival rates to hospital discharge were 11.9% for COVID-19 patients, compared with 23.5% for noninfected patients (adjusted relative risk, 0.65; P < .001). ROSC was 53.7% and 63.6%, for infected and noninfected patients, respectively (aRR, 0.86; P < .001).
COVID-19 patients also were more likely than noninfected patients to receive delayed defibrillation, the researchers said. “Although delays in resuscitation, especially defibrillation, may have contributed to lower survival, the negative association of COVID-19 with survival in this study was consistent across subgroups, including patients who received timely treatment with defibrillation and epinephrine.”
The extremely low survival rate in early pandemic studies likely reflected the overwhelming burden on health systems at the time, the researchers said in their discussion.
The study findings were limited by several factors, including potential confounding from unmeasured variables, the use of a quality improvement registry that may not reflect nonparticipating hospitals, and potential false-positive COVID-19 cases. However, the result support findings from recent studies of multiple centers and extend clinical knowledge by comparing infected and noninfected patients from a larger group of hospitals than previously studied, the researchers said.
“We believe that these data will be relevant to health care providers and hospital administrators as the COVID-19 pandemic continues,” they concluded.
Think beyond COVID-19 for cardiac care
“Early during the pandemic, questions were raised whether COVID-19 patients should be treated with CPR,” Dr. Girotra said in an interview. “This was because initial studies had found a dismal survival of 0%-3% in COVID patients treated with CPR. The potential of transmitting the virus to health care professionals during CPR further heightened these concerns. We wanted to know whether the poor survival reported in these initial studies were broadly representative.”
Dr. Girotra said that some of the study findings were surprising. “We found that of all patients with IHCA in 2020 in our study, one in four were suspected or confirmed to have COVID-19 infection. We were surprised by the magnitude of COVID’s impact on the cardiac arrest incidence.”
The implications for clinical decision-making are to think outside of COVID-19 infection, said Dr. Girotra. In the current study, “Although overall survival of cardiac arrest in COVID-positive patients was 30% lower, compared to non-COVID patients, it was not as poor as previously reported. COVID-19 infection alone should not be considered the sole factor for making decisions regarding CPR.
“Over the past 2 decades, we have experienced large gains in survival for in-hospital cardiac arrest. However, the COVID-19 pandemic has eroded these gains,” said Dr. Girotra. “Future studies are needed to monitor the impact of any new variants on cardiac arrest care,” as well as studies “to see whether we return to the prepandemic levels of IHCA survival once the pandemic recedes.”
Dr. Girotra has no relevant financial disclosures.
Survival after in-hospital cardiac arrest was roughly one-third lower in patients with COVID-19 infections compared to uninfected patients, based on data from nearly 25,000 individuals.
Survival rates of less than 3% were reported in the United States and China for patients who suffered in-hospital cardiac arrest (IHCA) while infected with COVID-19 early in the pandemic, but the data came from small, single-center studies in overwhelmed hospitals, wrote Saket Girotra, MD, of the University of Iowa, Iowa City, and fellow American Heart Association Get With the Guidelines–Resuscitation Investigators. Whether these early reports reflect the broader experience of patients with COVID-19 in hospitals in the United States remains unknown.
In a study published as a research letter in JAMA Network Open, the researchers reviewed data from the American Heart Association Get With the Guidelines–Resuscitation registry. The registry collects detailed information on patients aged 18 years and older who experience cardiac arrest at participating hospitals in the United States. The study population included 24,915 patients aged 18 years and older from 286 hospitals who experienced IHCA during March–December 2020. The mean age of the patients was 64.7 years; 61.1% were White, 24.8% were Black, 3.8% were of other race or ethnicity, and 10.3% were of unknown race or ethnicity.
The primary outcomes were survival to discharge and return of spontaneous circulation (ROSC) for at least 20 minutes.
A total of 5,916 patients (23.7%) had suspected or confirmed COVID-19 infections, and infected patients were more likely to be younger, male, and Black. Patients with COVID-19 infections also were significantly more likely than noninfected patients to have nonshockable rhythm, pneumonia, respiratory insufficiency, or sepsis, and to be on mechanical ventilation or vasopressors when the IHCA occurred, the researchers noted.
Survival rates to hospital discharge were 11.9% for COVID-19 patients, compared with 23.5% for noninfected patients (adjusted relative risk, 0.65; P < .001). ROSC was 53.7% and 63.6%, for infected and noninfected patients, respectively (aRR, 0.86; P < .001).
COVID-19 patients also were more likely than noninfected patients to receive delayed defibrillation, the researchers said. “Although delays in resuscitation, especially defibrillation, may have contributed to lower survival, the negative association of COVID-19 with survival in this study was consistent across subgroups, including patients who received timely treatment with defibrillation and epinephrine.”
The extremely low survival rate in early pandemic studies likely reflected the overwhelming burden on health systems at the time, the researchers said in their discussion.
The study findings were limited by several factors, including potential confounding from unmeasured variables, the use of a quality improvement registry that may not reflect nonparticipating hospitals, and potential false-positive COVID-19 cases. However, the result support findings from recent studies of multiple centers and extend clinical knowledge by comparing infected and noninfected patients from a larger group of hospitals than previously studied, the researchers said.
“We believe that these data will be relevant to health care providers and hospital administrators as the COVID-19 pandemic continues,” they concluded.
Think beyond COVID-19 for cardiac care
“Early during the pandemic, questions were raised whether COVID-19 patients should be treated with CPR,” Dr. Girotra said in an interview. “This was because initial studies had found a dismal survival of 0%-3% in COVID patients treated with CPR. The potential of transmitting the virus to health care professionals during CPR further heightened these concerns. We wanted to know whether the poor survival reported in these initial studies were broadly representative.”
Dr. Girotra said that some of the study findings were surprising. “We found that of all patients with IHCA in 2020 in our study, one in four were suspected or confirmed to have COVID-19 infection. We were surprised by the magnitude of COVID’s impact on the cardiac arrest incidence.”
The implications for clinical decision-making are to think outside of COVID-19 infection, said Dr. Girotra. In the current study, “Although overall survival of cardiac arrest in COVID-positive patients was 30% lower, compared to non-COVID patients, it was not as poor as previously reported. COVID-19 infection alone should not be considered the sole factor for making decisions regarding CPR.
“Over the past 2 decades, we have experienced large gains in survival for in-hospital cardiac arrest. However, the COVID-19 pandemic has eroded these gains,” said Dr. Girotra. “Future studies are needed to monitor the impact of any new variants on cardiac arrest care,” as well as studies “to see whether we return to the prepandemic levels of IHCA survival once the pandemic recedes.”
Dr. Girotra has no relevant financial disclosures.
FROM JAMA NETWORK OPEN