User login
New test for Lp(a) allows more accurate LDL-cholesterol results
A new study has drawn attention to inaccurate measurement of LDL-cholesterol levels in some patients with current assays, which could lead to incorrect therapeutic approaches.
The patient groups most affected are those with high levels of the lipoprotein Lp(a), in whom LDL-cholesterol levels are being overestimated in current laboratory tests, the authors say.
“Current laboratory assays all have the limitation that they cannot measure or report LDL cholesterol accurately. They are actually measuring the combination of LDL and Lp(a),” senior study author Sotirios Tsimikas, MD, University of California, San Diego, explained to this news organization.
“While this may not matter much in individuals with normal Lp(a) levels, in those with elevated Lp(a), the Lp(a) cholesterol may constitute a substantial proportion of the reported LDL cholesterol, and the actual LDL-cholesterol levels could be much lower that the value the lab is telling us,” he said.
Dr. Tsimikas gave the example of a patient with an LDL-cholesterol lab measurement of 75 mg/dL. “If that patient has an Lp(a) level of zero, then they do actually have an LDL level of 75. But as the Lp(a) increases, then the proportion of the result accounted for by LDL cholesterol decreases. So, if a patient with a measured LDL cholesterol of 75 has an Lp(a)-cholesterol level of 20, then their actual LDL level is 55.”
Dr. Tsimikas said it is important to know levels of both lipoproteins individually, so the correct therapeutic approach is used in situations where the Lp(a) cholesterol might be elevated.
“By understanding the actual values of LDL cholesterol and Lp(a) cholesterol, this will allow us to personalize the use of cholesterol-lowering medications and decide where to focus treatment. In the patient with a high level of Lp(a), their residual risk could be coming from Lp(a) cholesterol and less so from LDL cholesterol,” he added. “As we develop drugs to lower Lp(a), this patient might be better off on one of these rather than increasing efforts to lower LDL cholesterol, which might already be at goal.”
The study was published in the March 22 issue of the Journal of the American College of Cardiology.
Dr. Tsimikas noted that Lp(a) is now accepted as a genetic, independent, causal risk factor for cardiovascular disease, but current LDL-lowering drugs do not have much effect on Lp(a).
“Lp(a) can be lowered a little with niacin and PCSK9 inhibitors, but both have a quite a weak effect, and statins increase Lp(a). However, there are now multiple RNA-based therapeutics specifically targeting Lp(a) in clinical development,” he said.
At present, Lp(a) cholesterol has to be mathematically estimated, most commonly with the Dahlén formula, because of the lack of a validated, quantitative method to measure Lp(a) cholesterol, Dr. Tsimikas says.
For the current study, the researchers used a novel, quantitative, sensitive method to directly measure Lp(a) cholesterol, then applied this method to data from a recent study with the one of the new Lp(a)-lowering drugs in development – pelacarsen – which was conducted in patients with elevated Lp(a) levels.
Results showed that direct Lp(a)-cholesterol assessment, and subtracting this value from the laboratory LDL-cholesterol value, provides a more accurate reflection of the baseline and change in LDL cholesterol, the authors report. In the current study, corrected LDL cholesterol was 13 to 16 mg/dL lower than laboratory-reported LDL cholesterol.
Using the corrected LDL-cholesterol results, the study showed that pelacarsen significantly decreases Lp(a) cholesterol, with neutral to modest effects on LDL.
The study also suggests that the current method of calculating Lp(a) cholesterol, and then deriving a corrected LDL cholesterol – the Dahlén formula – is not accurate.
“The Dahlén formula relies on the assumption that Lp(a) cholesterol is universally a fixed 30% of Lp(a) mass, but this usually isn’t the case. The Dahlén formula needs to be discontinued. It can be highly inaccurate,” Dr. Tsimikas said.
Important implications
In an accompanying editorial, Guillaume Paré, MD, Michael Chong, PhD student, and Pedrum Mohammadi-Shemirani, BSc, all of McMaster University, Hamilton, Ont., say the current findings have three important clinical implications.
“First, they provide further proof that in individuals with elevated Lp(a), the contribution of Lp(a)-cholesterol to LDL-cholesterol is non-negligible using standard assays, with 13-16 mg/dL lower LDL-cholesterol post-correction.”
Secondly, the editorialists point out that these new findings confirm that the effect of Lp(a) inhibitors is likely to be mostly confined to Lp(a), “as would be expected.”
Finally, “and perhaps more importantly, the authors highlight the need to improve clinical reporting of lipid fractions to properly treat LDL-cholesterol and Lp(a) in high-risk patients,” they note.
“The report paves the way for future studies investigating the clinical utility of these additional measurements to initiate and monitor lipid-lowering therapy,” they conclude.
The clinical trial was funded by Ionis Pharmaceuticals, and the direct Lp(a)-cholesterol measurements were funded by Novartis through a research grant to the University of California, San Diego. Dr. Tsimikas is an employee of Ionis Pharmaceuticals and of the University of California, San Diego, and he is a cofounder of Covicept Therapeutics. He is also a coinventor and receives royalties from patents owned by UCSD on oxidation-specific antibodies and on biomarkers related to oxidized lipoproteins, as well as a cofounder and has equity interest in Oxitope and Kleanthi Diagnostics.
A version of this article first appeared on Medscape.com.
A new study has drawn attention to inaccurate measurement of LDL-cholesterol levels in some patients with current assays, which could lead to incorrect therapeutic approaches.
The patient groups most affected are those with high levels of the lipoprotein Lp(a), in whom LDL-cholesterol levels are being overestimated in current laboratory tests, the authors say.
“Current laboratory assays all have the limitation that they cannot measure or report LDL cholesterol accurately. They are actually measuring the combination of LDL and Lp(a),” senior study author Sotirios Tsimikas, MD, University of California, San Diego, explained to this news organization.
“While this may not matter much in individuals with normal Lp(a) levels, in those with elevated Lp(a), the Lp(a) cholesterol may constitute a substantial proportion of the reported LDL cholesterol, and the actual LDL-cholesterol levels could be much lower that the value the lab is telling us,” he said.
Dr. Tsimikas gave the example of a patient with an LDL-cholesterol lab measurement of 75 mg/dL. “If that patient has an Lp(a) level of zero, then they do actually have an LDL level of 75. But as the Lp(a) increases, then the proportion of the result accounted for by LDL cholesterol decreases. So, if a patient with a measured LDL cholesterol of 75 has an Lp(a)-cholesterol level of 20, then their actual LDL level is 55.”
Dr. Tsimikas said it is important to know levels of both lipoproteins individually, so the correct therapeutic approach is used in situations where the Lp(a) cholesterol might be elevated.
“By understanding the actual values of LDL cholesterol and Lp(a) cholesterol, this will allow us to personalize the use of cholesterol-lowering medications and decide where to focus treatment. In the patient with a high level of Lp(a), their residual risk could be coming from Lp(a) cholesterol and less so from LDL cholesterol,” he added. “As we develop drugs to lower Lp(a), this patient might be better off on one of these rather than increasing efforts to lower LDL cholesterol, which might already be at goal.”
The study was published in the March 22 issue of the Journal of the American College of Cardiology.
Dr. Tsimikas noted that Lp(a) is now accepted as a genetic, independent, causal risk factor for cardiovascular disease, but current LDL-lowering drugs do not have much effect on Lp(a).
“Lp(a) can be lowered a little with niacin and PCSK9 inhibitors, but both have a quite a weak effect, and statins increase Lp(a). However, there are now multiple RNA-based therapeutics specifically targeting Lp(a) in clinical development,” he said.
At present, Lp(a) cholesterol has to be mathematically estimated, most commonly with the Dahlén formula, because of the lack of a validated, quantitative method to measure Lp(a) cholesterol, Dr. Tsimikas says.
For the current study, the researchers used a novel, quantitative, sensitive method to directly measure Lp(a) cholesterol, then applied this method to data from a recent study with the one of the new Lp(a)-lowering drugs in development – pelacarsen – which was conducted in patients with elevated Lp(a) levels.
Results showed that direct Lp(a)-cholesterol assessment, and subtracting this value from the laboratory LDL-cholesterol value, provides a more accurate reflection of the baseline and change in LDL cholesterol, the authors report. In the current study, corrected LDL cholesterol was 13 to 16 mg/dL lower than laboratory-reported LDL cholesterol.
Using the corrected LDL-cholesterol results, the study showed that pelacarsen significantly decreases Lp(a) cholesterol, with neutral to modest effects on LDL.
The study also suggests that the current method of calculating Lp(a) cholesterol, and then deriving a corrected LDL cholesterol – the Dahlén formula – is not accurate.
“The Dahlén formula relies on the assumption that Lp(a) cholesterol is universally a fixed 30% of Lp(a) mass, but this usually isn’t the case. The Dahlén formula needs to be discontinued. It can be highly inaccurate,” Dr. Tsimikas said.
Important implications
In an accompanying editorial, Guillaume Paré, MD, Michael Chong, PhD student, and Pedrum Mohammadi-Shemirani, BSc, all of McMaster University, Hamilton, Ont., say the current findings have three important clinical implications.
“First, they provide further proof that in individuals with elevated Lp(a), the contribution of Lp(a)-cholesterol to LDL-cholesterol is non-negligible using standard assays, with 13-16 mg/dL lower LDL-cholesterol post-correction.”
Secondly, the editorialists point out that these new findings confirm that the effect of Lp(a) inhibitors is likely to be mostly confined to Lp(a), “as would be expected.”
Finally, “and perhaps more importantly, the authors highlight the need to improve clinical reporting of lipid fractions to properly treat LDL-cholesterol and Lp(a) in high-risk patients,” they note.
“The report paves the way for future studies investigating the clinical utility of these additional measurements to initiate and monitor lipid-lowering therapy,” they conclude.
The clinical trial was funded by Ionis Pharmaceuticals, and the direct Lp(a)-cholesterol measurements were funded by Novartis through a research grant to the University of California, San Diego. Dr. Tsimikas is an employee of Ionis Pharmaceuticals and of the University of California, San Diego, and he is a cofounder of Covicept Therapeutics. He is also a coinventor and receives royalties from patents owned by UCSD on oxidation-specific antibodies and on biomarkers related to oxidized lipoproteins, as well as a cofounder and has equity interest in Oxitope and Kleanthi Diagnostics.
A version of this article first appeared on Medscape.com.
A new study has drawn attention to inaccurate measurement of LDL-cholesterol levels in some patients with current assays, which could lead to incorrect therapeutic approaches.
The patient groups most affected are those with high levels of the lipoprotein Lp(a), in whom LDL-cholesterol levels are being overestimated in current laboratory tests, the authors say.
“Current laboratory assays all have the limitation that they cannot measure or report LDL cholesterol accurately. They are actually measuring the combination of LDL and Lp(a),” senior study author Sotirios Tsimikas, MD, University of California, San Diego, explained to this news organization.
“While this may not matter much in individuals with normal Lp(a) levels, in those with elevated Lp(a), the Lp(a) cholesterol may constitute a substantial proportion of the reported LDL cholesterol, and the actual LDL-cholesterol levels could be much lower that the value the lab is telling us,” he said.
Dr. Tsimikas gave the example of a patient with an LDL-cholesterol lab measurement of 75 mg/dL. “If that patient has an Lp(a) level of zero, then they do actually have an LDL level of 75. But as the Lp(a) increases, then the proportion of the result accounted for by LDL cholesterol decreases. So, if a patient with a measured LDL cholesterol of 75 has an Lp(a)-cholesterol level of 20, then their actual LDL level is 55.”
Dr. Tsimikas said it is important to know levels of both lipoproteins individually, so the correct therapeutic approach is used in situations where the Lp(a) cholesterol might be elevated.
“By understanding the actual values of LDL cholesterol and Lp(a) cholesterol, this will allow us to personalize the use of cholesterol-lowering medications and decide where to focus treatment. In the patient with a high level of Lp(a), their residual risk could be coming from Lp(a) cholesterol and less so from LDL cholesterol,” he added. “As we develop drugs to lower Lp(a), this patient might be better off on one of these rather than increasing efforts to lower LDL cholesterol, which might already be at goal.”
The study was published in the March 22 issue of the Journal of the American College of Cardiology.
Dr. Tsimikas noted that Lp(a) is now accepted as a genetic, independent, causal risk factor for cardiovascular disease, but current LDL-lowering drugs do not have much effect on Lp(a).
“Lp(a) can be lowered a little with niacin and PCSK9 inhibitors, but both have a quite a weak effect, and statins increase Lp(a). However, there are now multiple RNA-based therapeutics specifically targeting Lp(a) in clinical development,” he said.
At present, Lp(a) cholesterol has to be mathematically estimated, most commonly with the Dahlén formula, because of the lack of a validated, quantitative method to measure Lp(a) cholesterol, Dr. Tsimikas says.
For the current study, the researchers used a novel, quantitative, sensitive method to directly measure Lp(a) cholesterol, then applied this method to data from a recent study with the one of the new Lp(a)-lowering drugs in development – pelacarsen – which was conducted in patients with elevated Lp(a) levels.
Results showed that direct Lp(a)-cholesterol assessment, and subtracting this value from the laboratory LDL-cholesterol value, provides a more accurate reflection of the baseline and change in LDL cholesterol, the authors report. In the current study, corrected LDL cholesterol was 13 to 16 mg/dL lower than laboratory-reported LDL cholesterol.
Using the corrected LDL-cholesterol results, the study showed that pelacarsen significantly decreases Lp(a) cholesterol, with neutral to modest effects on LDL.
The study also suggests that the current method of calculating Lp(a) cholesterol, and then deriving a corrected LDL cholesterol – the Dahlén formula – is not accurate.
“The Dahlén formula relies on the assumption that Lp(a) cholesterol is universally a fixed 30% of Lp(a) mass, but this usually isn’t the case. The Dahlén formula needs to be discontinued. It can be highly inaccurate,” Dr. Tsimikas said.
Important implications
In an accompanying editorial, Guillaume Paré, MD, Michael Chong, PhD student, and Pedrum Mohammadi-Shemirani, BSc, all of McMaster University, Hamilton, Ont., say the current findings have three important clinical implications.
“First, they provide further proof that in individuals with elevated Lp(a), the contribution of Lp(a)-cholesterol to LDL-cholesterol is non-negligible using standard assays, with 13-16 mg/dL lower LDL-cholesterol post-correction.”
Secondly, the editorialists point out that these new findings confirm that the effect of Lp(a) inhibitors is likely to be mostly confined to Lp(a), “as would be expected.”
Finally, “and perhaps more importantly, the authors highlight the need to improve clinical reporting of lipid fractions to properly treat LDL-cholesterol and Lp(a) in high-risk patients,” they note.
“The report paves the way for future studies investigating the clinical utility of these additional measurements to initiate and monitor lipid-lowering therapy,” they conclude.
The clinical trial was funded by Ionis Pharmaceuticals, and the direct Lp(a)-cholesterol measurements were funded by Novartis through a research grant to the University of California, San Diego. Dr. Tsimikas is an employee of Ionis Pharmaceuticals and of the University of California, San Diego, and he is a cofounder of Covicept Therapeutics. He is also a coinventor and receives royalties from patents owned by UCSD on oxidation-specific antibodies and on biomarkers related to oxidized lipoproteins, as well as a cofounder and has equity interest in Oxitope and Kleanthi Diagnostics.
A version of this article first appeared on Medscape.com.
Acute STEMI During the COVID-19 Pandemic at a Regional Hospital: Incidence, Clinical Characteristics, and Outcomes
From the Department of Medicine, Medical College of Georgia at the Augusta University-University of Georgia Medical Partnership, Athens, GA (Syed H. Ali, Syed Hyder, and Dr. Murrow), and the Department of Cardiology, Piedmont Heart Institute, Piedmont Athens Regional, Athens, GA (Dr. Murrow and Mrs. Davis).
Abstract
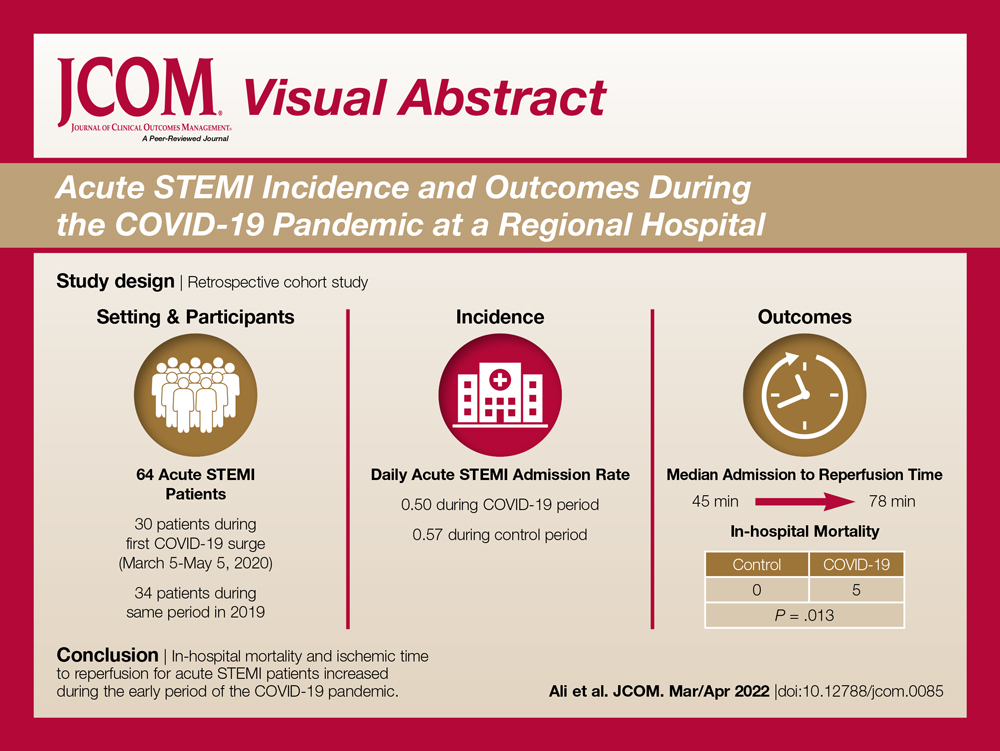
Objectives: The aim of this study was to describe the characteristics and in-hospital outcomes of patients with acute ST-segment elevation myocardial infarction (STEMI) during the early COVID-19 pandemic at Piedmont Athens Regional (PAR), a 330-bed tertiary referral center in Northeast Georgia.
Methods: A retrospective study was conducted at PAR to evaluate patients with acute STEMI admitted over an 8-week period during the initial COVID-19 outbreak. This study group was compared to patients admitted during the corresponding period in 2019. The primary endpoint of this study was defined as a composite of sustained ventricular arrhythmia, congestive heart failure (CHF) with pulmonary congestion, and/or in-hospital mortality.
Results: This study cohort was composed of 64 patients with acute STEMI; 30 patients (46.9%) were hospitalized during the COVID-19 pandemic. Patients with STEMI in both the COVID-19 and control groups had similar comorbidities, Killip classification score, and clinical presentations. The median (interquartile range) time from symptom onset to reperfusion (total ischemic time) increased from 99.5 minutes (84.8-132) in 2019 to 149 minutes (96.3-231.8; P = .032) in 2020. Hospitalization during the COVID-19 period was associated with an increased risk for combined in-hospital outcome (odds ratio, 3.96; P = .046).
Conclusion: Patients with STEMI admitted during the first wave of the COVID-19 outbreak experienced longer total ischemic time and increased risk for combined in-hospital outcomes compared to patients admitted during the corresponding period in 2019.
Keywords: myocardial infarction, acute coronary syndrome, hospitalization, outcomes.
The emergence of the SARS-Cov-2 virus in December 2019 caused a worldwide shift in resource allocation and the restructuring of health care systems within the span of a few months. With the rapid spread of infection, the World Health Organization officially declared a pandemic in March 2020. The pandemic led to the deferral and cancellation of in-person patient visits, routine diagnostic studies, and nonessential surgeries and procedures. This response occurred secondary to a joint effort to reduce transmission via stay-at-home mandates and appropriate social distancing.1
Alongside the reduction in elective procedures and health care visits, significant reductions in hospitalization rates due to decreases in acute ST-segment elevation myocardial infarction (STEMI) and catheterization laboratory utilization have been reported in many studies from around the world.2-7 Comprehensive data demonstrating the impact of the COVID-19 pandemic on acute STEMI patient characteristics, clinical presentation, and in-hospital outcomes are lacking. Although patients with previously diagnosed cardiovascular disease are more likely to encounter worse outcomes in the setting of COVID-19, there may also be an indirect impact of the pandemic on high-risk patients, including those without the infection.8 Several theories have been hypothesized to explain this phenomenon. One theory postulates that the fear of contracting the virus during hospitalization is great enough to prevent patients from seeking care.2 Another theory suggests that the increased utilization of telemedicine prevents exacerbation of chronic conditions and the need for hospitalization.9 Contrary to this trend, previous studies have shown an increased incidence of acute STEMI following stressful events such as natural disasters.10
The aim of this study was to describe trends pertaining to clinical characteristics and in-hospital outcomes of patients with acute STEMI during the early COVID-19 pandemic at Piedmont Athens Regional (PAR), a 330-bed tertiary referral center in Northeast Georgia.
Methods
A retrospective cohort study was conducted at PAR to evaluate patients with STEMI admitted to the cardiovascular intensive care unit over an 8-week period (March 5 to May 5, 2020) during the COVID-19 outbreak. COVID-19 was declared a national emergency on March 13, 2020, in the United States. The institutional review board at PAR approved the study; the need for individual consent was waived under the condition that participant data would undergo de-identification and be strictly safeguarded.
Data Collection
Because there are seasonal variations in cardiovascular admissions, patient data from a control period (March 9 to May 9, 2019) were obtained to compare with data from the 2020 period. The number of patients with the diagnosis of acute STEMI during the COVID-19 period was recorded. Demographic data, clinical characteristics, and primary angiographic findings were gathered for all patients. Time from symptom onset to hospital admission and time from hospital admission to reperfusion (defined as door-to-balloon time) were documented for each patient. Killip classification was used to assess patients’ clinical status on admission. Length of stay was determined as days from hospital admission to discharge or death (if occurring during the same hospitalization).
Adverse in-hospital complications were also recorded. These were selected based on inclusion of the following categories of acute STEMI complications: ischemic, mechanical, arrhythmic, embolic, and inflammatory. The following complications occurred in our patient cohort: sustained ventricular arrhythmia, congestive heart failure (CHF) defined as congestion requiring intravenous diuretics, re-infarction, mechanical complications (free-wall rupture, ventricular septal defect, or mitral regurgitation), second- or third-degree atrioventricular block, atrial fibrillation, stroke, mechanical ventilation, major bleeding, pericarditis, cardiogenic shock, cardiac arrest, and in-hospital mortality. The primary outcome of this study was defined as a composite of sustained ventricular arrhythmia, CHF with congestion requiring intravenous diuretics, and/or in-hospital mortality. Ventricular arrythmia and CHF were included in the composite outcome because they are defined as the 2 most common causes of sudden cardiac death following acute STEMI.11,12
Statistical Analysis
Normally distributed continuous variables and categorical variables were compared using the paired t-test. A 2-sided P value <.05 was considered to be statistically significant. Mean admission rates for acute STEMI hospitalizations were determined by dividing the number of admissions by the number of days in each time period. The daily rate of COVID-19 cases per 100,000 individuals was obtained from the Centers for Disease Control and Prevention COVID-19 database. All data analyses were performed using Microsoft Excel.
Results
The study cohort consisted of 64 patients, of whom 30 (46.9%) were hospitalized between March 5 and May 5, 2020, and 34 (53.1%) who were admitted during the analogous time period in 2019. This reflected a 6% decrease in STEMI admissions at PAR in the COVID-19 cohort.
Acute STEMI Hospitalization Rates and COVID-19 Incidence
The mean daily acute STEMI admission rate was 0.50 during the study period compared to 0.57 during the control period. During the study period in 2020 in the state of Georgia, the daily rate of newly confirmed COVID-19 cases ranged from 0.194 per 100,000 on March 5 to 8.778 per 100,000 on May 5. Results of COVID-19 testing were available for 9 STEMI patients, and of these 0 tests were positive.
Baseline Characteristics
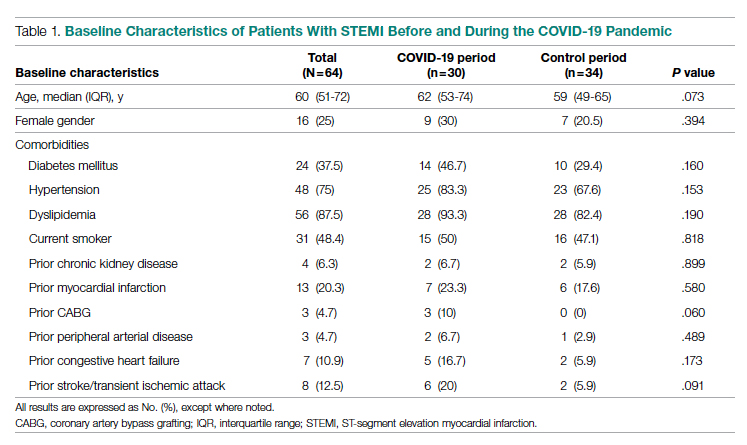
Baseline characteristics of the acute STEMI cohorts are presented in Table 1. Approximately 75% were male; median (interquartile range [IQR]) age was 60 (51-72) years. There were no significant differences in age and gender between the study periods. Three-quarters of patients had a history of hypertension, and 87.5% had a history of dyslipidemia. There was no significant difference in baseline comorbidity profiles between the 2 study periods; therefore, our sample populations shared similar characteristics.
Clinical Presentation
Significant differences were observed regarding the time intervals of STEMI patients in the COVID-19 period and the control period (Table 2). Median time from symptom onset to hospital admission (patient delay) was extended from 57.5 minutes (IQR, 40.3-106) in 2019 to 93 minutes (IQR, 48.8-132) in 2020; however, this difference was not statistically significant (P = .697). Median time from hospital admission to reperfusion (system delay) was prolonged from 45 minutes (IQR, 28-61) in 2019 to 78 minutes (IQR, 50-110) in 2020 (P < .001). Overall time from symptom onset to reperfusion (total ischemic time) increased from 99.5 minutes (IQR, 84.8-132) in 2019 to 149 minutes (IQR, 96.3-231.8) in 2020 (P = .032).
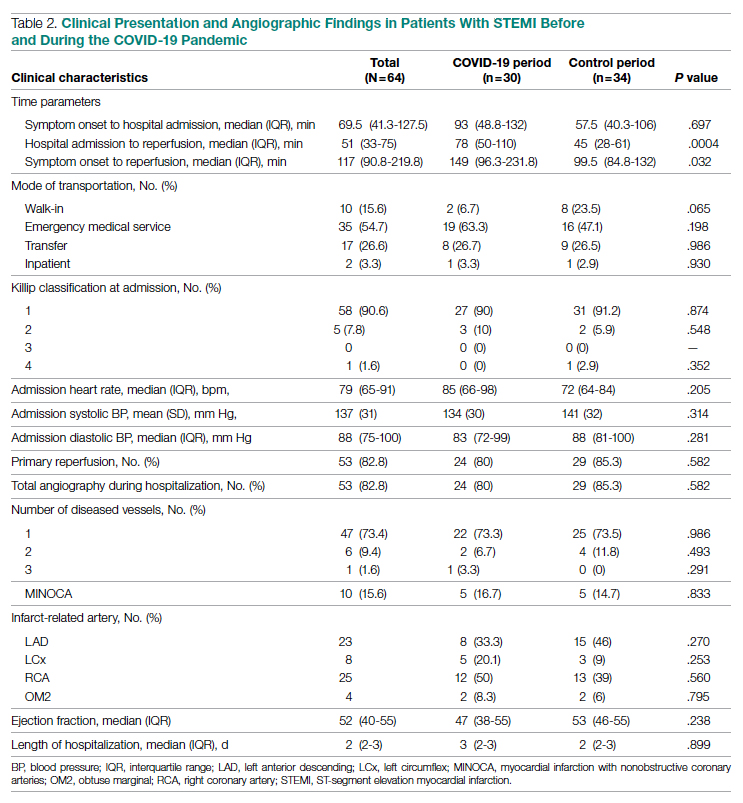
Regarding mode of transportation, 23.5% of patients in 2019 were walk-in admissions to the emergency department. During the COVID-19 period, walk-in admissions decreased to 6.7% (P = .065). There were no significant differences between emergency medical service, transfer, or in-patient admissions for STEMI cases between the 2 study periods.
Killip classification scores were calculated for all patients on admission; 90.6% of patients were classified as Killip Class 1. There was no significant difference between hemodynamic presentations during the COVID-19 period compared to the control period.
Angiographic Data
Overall, 53 (82.8%) patients admitted with acute STEMI underwent coronary angiography during their hospital stay. The proportion of patients who underwent primary reperfusion was greater in the control period than in the COVID-19 period (85.3% vs 80%; P = .582). Angiographic characteristics and findings were similar between the 2 study groups (Table 2).
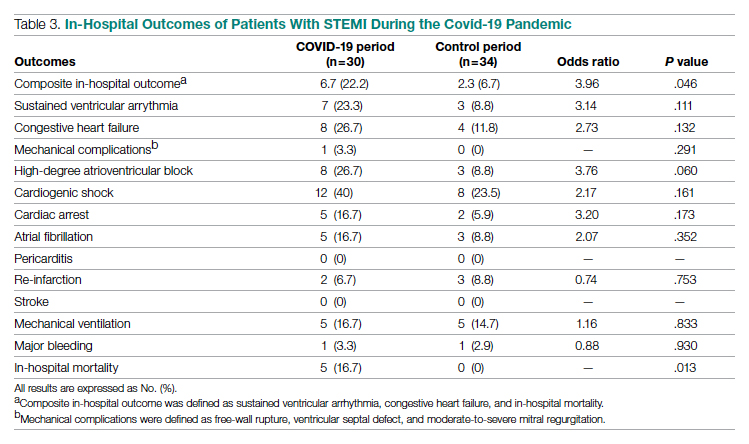
In-Hospital Outcomes
In-hospital outcome data were available for all patients. As shown in Table 3, hospitalization during the COVID-19 period was independently associated with an increased risk for combined in-hospital outcome (odds ratio, 3.96; P = .046). The rate of in-hospital mortality was greater in the COVID-19 period (P = .013). We found no significant difference when comparing secondary outcomes from admissions during the COVID-19 period and the control period in 2019. For the 5 patients who died during the study period, the primary diagnosis at death was acute STEMI complicated by CHF (3 patients) or cardiogenic shock (2 patients).
Discussion
This single-center retrospective study at PAR looks at the impact of COVID-19 on hospitalizations for acute STEMI during the initial peak of the pandemic. The key findings of this study show a significant increase in ischemic time parameters (symptom onset to reperfusion, hospital admission to reperfusion), in-hospital mortality, and combined in-hospital outcomes.
There was a 49.5-minute increase in total ischemic time noted in this study (P = .032). Though there was a numerical increase in time of symptom onset to hospital admission by 23.5 minutes, this difference was not statistically significant (P = .697). However, this study observed a statistically significant 33-minute increase in ischemic time from hospital admission to reperfusion (P < .001). Multiple studies globally have found a similar increase in total ischemic times, including those conducted in China and Europe.13-15 Every level of potential delay must be considered, including pre-hospital, triage and emergency department, and/or reperfusion team. Pre-hospital sources of delays that have been suggested include “stay-at-home” orders and apprehension to seek medical care due to concern about contracting the virus or overwhelming the health care facilities. There was a clinically significant 4-fold decrease in the number of walk-in acute STEMI cases in the study period. In 2019, there were 8 walk-in cases compared to 2 cases in 2020 (P = .065). However, this change was not statistically significant. In-hospital/systemic sources of delays have been mentioned in other studies; they include increased time taken to rule out COVID-19 (nasopharyngeal swab/chest x-ray) and increased time due to the need for intensive gowning and gloving procedures by staff. It was difficult to objectively determine the sources of system delay by the reperfusion team due to a lack of quantitative data.
In the current study, we found a significant increase in in-hospital mortality during the COVID-19 period compared to a parallel time frame in 2019. This finding is contrary to a multicenter study from Spain that reported no difference in in-hospital outcomes or mortality rates among all acute coronary syndrome cases.16 The worsening outcomes and prognosis may simply be a result of increased ischemic time; however, the virus that causes COVID-19 itself may play a role as well. Studies have found that SARS-Cov-2 infection places patients at greater risk for cardiovascular conditions such as hypercoagulability, myocarditis, and arrhythmias.17 In our study, however, there were no acute STEMI patients who tested positive for COVID-19. Therefore, we cannot discuss the impact of increased thrombus burden in patients with COVID-19. Piedmont Healthcare published a STEMI treatment protocol in May 2020 that advised increased use of tissue plasminogen activator (tPA) in COVID-19-positive cases; during the study period, however, there were no occasions when tPA use was deemed appropriate based on clinical judgment.
Our findings align with previous studies that describe an increase in combined in-hospital adverse outcomes during the COVID-19 era. Previous studies detected a higher rate of complications in the COVID-19 cohort, but in the current study, the adverse in-hospital course is unrelated to underlying infection.18,19 This study reports a higher incidence of major in-hospital outcomes, including a 65% increase in the rate of combined in-hospital outcomes, which is similar to a multicenter study conducted in Israel.19 There was a 2.3-fold numerical increase in sustained ventricular arrhythmias and a 2.5-fold numerical increase in the incidence of cardiac arrest in the study period. This phenomenon was observed despite a similar rate of reperfusion procedures in both groups.
Acute STEMI is a highly fatal condition with an incidence of 8.5 in 10,000 annually in the United States. While studies across the world have shown a 25% to 40% reduction in the rate of hospitalized acute coronary syndrome cases during the COVID-19 pandemic, the decrease from 34 to 30 STEMI admissions at PAR is not statistically significant.20 Possible reasons for the reduction globally include increased out-of-hospital mortality and decreased incidence of acute STEMI across the general population as a result of improved access to telemedicine or decreased levels of life stressors.20
In summary, there was an increase in ischemic time to reperfusion, in-hospital mortality, and combined in-hospital outcomes for acute STEMI patients at PAR during the COVID period.
Limitations
This study has several limitations. This is a single-center study, so the sample size is small and may not be generalizable to a larger population. This is a retrospective observational study, so causation cannot be inferred. This study analyzed ischemic time parameters as average rates over time rather than in an interrupted time series. Post-reperfusion outcomes were limited to hospital stay. Post-hospital follow-up would provide a better picture of the effects of STEMI intervention. There is no account of patients who died out-of-hospital secondary to acute STEMI. COVID-19 testing was not introduced until midway in our study period. Therefore, we cannot rule out the possibility of the SARS-Cov-2 virus inciting acute STEMI and subsequently leading to worse outcomes and poor prognosis.
Conclusions
This study provides an analysis of the incidence, characteristics, and clinical outcomes of patients presenting with acute STEMI during the early period of the COVID-19 pandemic. In-hospital mortality and ischemic time to reperfusion increased while combined in-hospital outcomes worsened.
Acknowledgment: The authors thank Piedmont Athens Regional IRB for approving this project and allowing access to patient data.
Corresponding author: Syed H. Ali; Department of Medicine, Medical College of Georgia at the Augusta University-University of Georgia Medical Partnership, 30606, Athens, GA; syedha.ali@gmail.com
Disclosures: None reported.
doi:10.12788/jcom.0085
1. Bhatt AS, Moscone A, McElrath EE, et al. Fewer hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J Am Coll Cardiol. 2020;76(3):280-288. doi:10.1016/j.jacc.2020.05.038
2. Metzler B, Siostrzonek P, Binder RK, Bauer A, Reinstadler SJR. Decline of acute coronary syndrome admissions in Austria since the outbreak of Covid-19: the pandemic response causes cardiac collateral damage. Eur Heart J. 2020;41:1852-1853. doi:10.1093/eurheartj/ehaa314
3. De Rosa S, Spaccarotella C, Basso C, et al. Reduction of hospitalizations for myocardial infarction in Italy in the Covid-19 era. Eur Heart J. 2020;41(22):2083-2088.
4. Wilson SJ, Connolly MJ, Elghamry Z, et al. Effect of the COVID-19 pandemic on ST-segment-elevation myocardial infarction presentations and in-hospital outcomes. Circ Cardiovasc Interv. 2020; 13(7):e009438. doi:10.1161/CIRCINTERVENTIONS.120.009438
5. Mafham MM, Spata E, Goldacre R, et al. Covid-19 pandemic and admission rates for and management of acute coronary syndromes in England. Lancet. 2020;396 (10248):381-389. doi:10.1016/S0140-6736(20)31356-8
6. Bhatt AS, Moscone A, McElrath EE, et al. Fewer Hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J Am Coll Cardiol. 2020;76(3):280-288. doi:10.1016/j.jacc.2020.05.038
7. Tam CF, Cheung KS, Lam S, et al. Impact of Coronavirus disease 2019 (Covid-19) outbreak on ST-segment elevation myocardial infarction care in Hong Kong, China. Circ Cardiovasc Qual Outcomes. 2020;13(4):e006631. doi:10.1161/CIRCOUTCOMES.120.006631
8. Clerkin KJ, Fried JA, Raikhelkar J, et al. Coronavirus disease 2019 (COVID-19) and cardiovascular disease. Circulation. 2020;141:1648-1655. doi:10.1161/CIRCULATIONAHA.120.046941
9. Ebinger JE, Shah PK. Declining admissions for acute cardiovascular illness: The Covid-19 paradox. J Am Coll Cardiol. 2020;76(3):289-291. doi:10.1016/j.jacc.2020.05.039
10 Leor J, Poole WK, Kloner RA. Sudden cardiac death triggered by an earthquake. N Engl J Med. 1996;334(7):413-419. doi:10.1056/NEJM199602153340701
11. Hiramori K. Major causes of death from acute myocardial infarction in a coronary care unit. Jpn Circ J. 1987;51(9):1041-1047. doi:10.1253/jcj.51.1041
12. Bui AH, Waks JW. Risk stratification of sudden cardiac death after acute myocardial infarction. J Innov Card Rhythm Manag. 2018;9(2):3035-3049. doi:10.19102/icrm.2018.090201
13. Xiang D, Xiang X, Zhang W, et al. Management and outcomes of patients with STEMI during the COVID-19 pandemic in China. J Am Coll Cardiol. 2020;76(11):1318-1324. doi:10.1016/j.jacc.2020.06.039
14. Hakim R, Motreff P, Rangé G. COVID-19 and STEMI. [Article in French]. Ann Cardiol Angeiol (Paris). 2020;69(6):355-359. doi:10.1016/j.ancard.2020.09.034
15. Soylu K, Coksevim M, Yanık A, Bugra Cerik I, Aksan G. Effect of Covid-19 pandemic process on STEMI patients timeline. Int J Clin Pract. 2021;75(5):e14005. doi:10.1111/ijcp.14005
16. Salinas P, Travieso A, Vergara-Uzcategui C, et al. Clinical profile and 30-day mortality of invasively managed patients with suspected acute coronary syndrome during the COVID-19 outbreak. Int Heart J. 2021;62(2):274-281. doi:10.1536/ihj.20-574
17. Hu Y, Sun J, Dai Z, et al. Prevalence and severity of corona virus disease 2019 (Covid-19): a systematic review and meta-analysis. J Clin Virol. 2020;127:104371. doi:10.1016/j.jcv.2020.104371
18. Rodriguez-Leor O, Cid Alvarez AB, Perez de Prado A, et al. In-hospital outcomes of COVID-19 ST-elevation myocardial infarction patients. EuroIntervention. 2021;16(17):1426-1433. doi:10.4244/EIJ-D-20-00935
19. Fardman A, Zahger D, Orvin K, et al. Acute myocardial infarction in the Covid-19 era: incidence, clinical characteristics and in-hospital outcomes—A multicenter registry. PLoS ONE. 2021;16(6): e0253524. doi:10.1371/journal.pone.0253524
20. Pessoa-Amorim G, Camm CF, Gajendragadkar P, et al. Admission of patients with STEMI since the outbreak of the COVID-19 pandemic: a survey by the European Society of Cardiology. Eur Heart J Qual Care Clin Outcomes. 2020;6(3):210-216. doi:10.1093/ehjqcco/qcaa046
From the Department of Medicine, Medical College of Georgia at the Augusta University-University of Georgia Medical Partnership, Athens, GA (Syed H. Ali, Syed Hyder, and Dr. Murrow), and the Department of Cardiology, Piedmont Heart Institute, Piedmont Athens Regional, Athens, GA (Dr. Murrow and Mrs. Davis).
Abstract
Objectives: The aim of this study was to describe the characteristics and in-hospital outcomes of patients with acute ST-segment elevation myocardial infarction (STEMI) during the early COVID-19 pandemic at Piedmont Athens Regional (PAR), a 330-bed tertiary referral center in Northeast Georgia.
Methods: A retrospective study was conducted at PAR to evaluate patients with acute STEMI admitted over an 8-week period during the initial COVID-19 outbreak. This study group was compared to patients admitted during the corresponding period in 2019. The primary endpoint of this study was defined as a composite of sustained ventricular arrhythmia, congestive heart failure (CHF) with pulmonary congestion, and/or in-hospital mortality.
Results: This study cohort was composed of 64 patients with acute STEMI; 30 patients (46.9%) were hospitalized during the COVID-19 pandemic. Patients with STEMI in both the COVID-19 and control groups had similar comorbidities, Killip classification score, and clinical presentations. The median (interquartile range) time from symptom onset to reperfusion (total ischemic time) increased from 99.5 minutes (84.8-132) in 2019 to 149 minutes (96.3-231.8; P = .032) in 2020. Hospitalization during the COVID-19 period was associated with an increased risk for combined in-hospital outcome (odds ratio, 3.96; P = .046).
Conclusion: Patients with STEMI admitted during the first wave of the COVID-19 outbreak experienced longer total ischemic time and increased risk for combined in-hospital outcomes compared to patients admitted during the corresponding period in 2019.
Keywords: myocardial infarction, acute coronary syndrome, hospitalization, outcomes.

The emergence of the SARS-Cov-2 virus in December 2019 caused a worldwide shift in resource allocation and the restructuring of health care systems within the span of a few months. With the rapid spread of infection, the World Health Organization officially declared a pandemic in March 2020. The pandemic led to the deferral and cancellation of in-person patient visits, routine diagnostic studies, and nonessential surgeries and procedures. This response occurred secondary to a joint effort to reduce transmission via stay-at-home mandates and appropriate social distancing.1
Alongside the reduction in elective procedures and health care visits, significant reductions in hospitalization rates due to decreases in acute ST-segment elevation myocardial infarction (STEMI) and catheterization laboratory utilization have been reported in many studies from around the world.2-7 Comprehensive data demonstrating the impact of the COVID-19 pandemic on acute STEMI patient characteristics, clinical presentation, and in-hospital outcomes are lacking. Although patients with previously diagnosed cardiovascular disease are more likely to encounter worse outcomes in the setting of COVID-19, there may also be an indirect impact of the pandemic on high-risk patients, including those without the infection.8 Several theories have been hypothesized to explain this phenomenon. One theory postulates that the fear of contracting the virus during hospitalization is great enough to prevent patients from seeking care.2 Another theory suggests that the increased utilization of telemedicine prevents exacerbation of chronic conditions and the need for hospitalization.9 Contrary to this trend, previous studies have shown an increased incidence of acute STEMI following stressful events such as natural disasters.10
The aim of this study was to describe trends pertaining to clinical characteristics and in-hospital outcomes of patients with acute STEMI during the early COVID-19 pandemic at Piedmont Athens Regional (PAR), a 330-bed tertiary referral center in Northeast Georgia.
Methods
A retrospective cohort study was conducted at PAR to evaluate patients with STEMI admitted to the cardiovascular intensive care unit over an 8-week period (March 5 to May 5, 2020) during the COVID-19 outbreak. COVID-19 was declared a national emergency on March 13, 2020, in the United States. The institutional review board at PAR approved the study; the need for individual consent was waived under the condition that participant data would undergo de-identification and be strictly safeguarded.
Data Collection
Because there are seasonal variations in cardiovascular admissions, patient data from a control period (March 9 to May 9, 2019) were obtained to compare with data from the 2020 period. The number of patients with the diagnosis of acute STEMI during the COVID-19 period was recorded. Demographic data, clinical characteristics, and primary angiographic findings were gathered for all patients. Time from symptom onset to hospital admission and time from hospital admission to reperfusion (defined as door-to-balloon time) were documented for each patient. Killip classification was used to assess patients’ clinical status on admission. Length of stay was determined as days from hospital admission to discharge or death (if occurring during the same hospitalization).
Adverse in-hospital complications were also recorded. These were selected based on inclusion of the following categories of acute STEMI complications: ischemic, mechanical, arrhythmic, embolic, and inflammatory. The following complications occurred in our patient cohort: sustained ventricular arrhythmia, congestive heart failure (CHF) defined as congestion requiring intravenous diuretics, re-infarction, mechanical complications (free-wall rupture, ventricular septal defect, or mitral regurgitation), second- or third-degree atrioventricular block, atrial fibrillation, stroke, mechanical ventilation, major bleeding, pericarditis, cardiogenic shock, cardiac arrest, and in-hospital mortality. The primary outcome of this study was defined as a composite of sustained ventricular arrhythmia, CHF with congestion requiring intravenous diuretics, and/or in-hospital mortality. Ventricular arrythmia and CHF were included in the composite outcome because they are defined as the 2 most common causes of sudden cardiac death following acute STEMI.11,12
Statistical Analysis
Normally distributed continuous variables and categorical variables were compared using the paired t-test. A 2-sided P value <.05 was considered to be statistically significant. Mean admission rates for acute STEMI hospitalizations were determined by dividing the number of admissions by the number of days in each time period. The daily rate of COVID-19 cases per 100,000 individuals was obtained from the Centers for Disease Control and Prevention COVID-19 database. All data analyses were performed using Microsoft Excel.
Results
The study cohort consisted of 64 patients, of whom 30 (46.9%) were hospitalized between March 5 and May 5, 2020, and 34 (53.1%) who were admitted during the analogous time period in 2019. This reflected a 6% decrease in STEMI admissions at PAR in the COVID-19 cohort.
Acute STEMI Hospitalization Rates and COVID-19 Incidence
The mean daily acute STEMI admission rate was 0.50 during the study period compared to 0.57 during the control period. During the study period in 2020 in the state of Georgia, the daily rate of newly confirmed COVID-19 cases ranged from 0.194 per 100,000 on March 5 to 8.778 per 100,000 on May 5. Results of COVID-19 testing were available for 9 STEMI patients, and of these 0 tests were positive.
Baseline Characteristics
Baseline characteristics of the acute STEMI cohorts are presented in Table 1. Approximately 75% were male; median (interquartile range [IQR]) age was 60 (51-72) years. There were no significant differences in age and gender between the study periods. Three-quarters of patients had a history of hypertension, and 87.5% had a history of dyslipidemia. There was no significant difference in baseline comorbidity profiles between the 2 study periods; therefore, our sample populations shared similar characteristics.

Clinical Presentation
Significant differences were observed regarding the time intervals of STEMI patients in the COVID-19 period and the control period (Table 2). Median time from symptom onset to hospital admission (patient delay) was extended from 57.5 minutes (IQR, 40.3-106) in 2019 to 93 minutes (IQR, 48.8-132) in 2020; however, this difference was not statistically significant (P = .697). Median time from hospital admission to reperfusion (system delay) was prolonged from 45 minutes (IQR, 28-61) in 2019 to 78 minutes (IQR, 50-110) in 2020 (P < .001). Overall time from symptom onset to reperfusion (total ischemic time) increased from 99.5 minutes (IQR, 84.8-132) in 2019 to 149 minutes (IQR, 96.3-231.8) in 2020 (P = .032).

Regarding mode of transportation, 23.5% of patients in 2019 were walk-in admissions to the emergency department. During the COVID-19 period, walk-in admissions decreased to 6.7% (P = .065). There were no significant differences between emergency medical service, transfer, or in-patient admissions for STEMI cases between the 2 study periods.
Killip classification scores were calculated for all patients on admission; 90.6% of patients were classified as Killip Class 1. There was no significant difference between hemodynamic presentations during the COVID-19 period compared to the control period.
Angiographic Data
Overall, 53 (82.8%) patients admitted with acute STEMI underwent coronary angiography during their hospital stay. The proportion of patients who underwent primary reperfusion was greater in the control period than in the COVID-19 period (85.3% vs 80%; P = .582). Angiographic characteristics and findings were similar between the 2 study groups (Table 2).
In-Hospital Outcomes
In-hospital outcome data were available for all patients. As shown in Table 3, hospitalization during the COVID-19 period was independently associated with an increased risk for combined in-hospital outcome (odds ratio, 3.96; P = .046). The rate of in-hospital mortality was greater in the COVID-19 period (P = .013). We found no significant difference when comparing secondary outcomes from admissions during the COVID-19 period and the control period in 2019. For the 5 patients who died during the study period, the primary diagnosis at death was acute STEMI complicated by CHF (3 patients) or cardiogenic shock (2 patients).

Discussion
This single-center retrospective study at PAR looks at the impact of COVID-19 on hospitalizations for acute STEMI during the initial peak of the pandemic. The key findings of this study show a significant increase in ischemic time parameters (symptom onset to reperfusion, hospital admission to reperfusion), in-hospital mortality, and combined in-hospital outcomes.
There was a 49.5-minute increase in total ischemic time noted in this study (P = .032). Though there was a numerical increase in time of symptom onset to hospital admission by 23.5 minutes, this difference was not statistically significant (P = .697). However, this study observed a statistically significant 33-minute increase in ischemic time from hospital admission to reperfusion (P < .001). Multiple studies globally have found a similar increase in total ischemic times, including those conducted in China and Europe.13-15 Every level of potential delay must be considered, including pre-hospital, triage and emergency department, and/or reperfusion team. Pre-hospital sources of delays that have been suggested include “stay-at-home” orders and apprehension to seek medical care due to concern about contracting the virus or overwhelming the health care facilities. There was a clinically significant 4-fold decrease in the number of walk-in acute STEMI cases in the study period. In 2019, there were 8 walk-in cases compared to 2 cases in 2020 (P = .065). However, this change was not statistically significant. In-hospital/systemic sources of delays have been mentioned in other studies; they include increased time taken to rule out COVID-19 (nasopharyngeal swab/chest x-ray) and increased time due to the need for intensive gowning and gloving procedures by staff. It was difficult to objectively determine the sources of system delay by the reperfusion team due to a lack of quantitative data.
In the current study, we found a significant increase in in-hospital mortality during the COVID-19 period compared to a parallel time frame in 2019. This finding is contrary to a multicenter study from Spain that reported no difference in in-hospital outcomes or mortality rates among all acute coronary syndrome cases.16 The worsening outcomes and prognosis may simply be a result of increased ischemic time; however, the virus that causes COVID-19 itself may play a role as well. Studies have found that SARS-Cov-2 infection places patients at greater risk for cardiovascular conditions such as hypercoagulability, myocarditis, and arrhythmias.17 In our study, however, there were no acute STEMI patients who tested positive for COVID-19. Therefore, we cannot discuss the impact of increased thrombus burden in patients with COVID-19. Piedmont Healthcare published a STEMI treatment protocol in May 2020 that advised increased use of tissue plasminogen activator (tPA) in COVID-19-positive cases; during the study period, however, there were no occasions when tPA use was deemed appropriate based on clinical judgment.
Our findings align with previous studies that describe an increase in combined in-hospital adverse outcomes during the COVID-19 era. Previous studies detected a higher rate of complications in the COVID-19 cohort, but in the current study, the adverse in-hospital course is unrelated to underlying infection.18,19 This study reports a higher incidence of major in-hospital outcomes, including a 65% increase in the rate of combined in-hospital outcomes, which is similar to a multicenter study conducted in Israel.19 There was a 2.3-fold numerical increase in sustained ventricular arrhythmias and a 2.5-fold numerical increase in the incidence of cardiac arrest in the study period. This phenomenon was observed despite a similar rate of reperfusion procedures in both groups.
Acute STEMI is a highly fatal condition with an incidence of 8.5 in 10,000 annually in the United States. While studies across the world have shown a 25% to 40% reduction in the rate of hospitalized acute coronary syndrome cases during the COVID-19 pandemic, the decrease from 34 to 30 STEMI admissions at PAR is not statistically significant.20 Possible reasons for the reduction globally include increased out-of-hospital mortality and decreased incidence of acute STEMI across the general population as a result of improved access to telemedicine or decreased levels of life stressors.20
In summary, there was an increase in ischemic time to reperfusion, in-hospital mortality, and combined in-hospital outcomes for acute STEMI patients at PAR during the COVID period.
Limitations
This study has several limitations. This is a single-center study, so the sample size is small and may not be generalizable to a larger population. This is a retrospective observational study, so causation cannot be inferred. This study analyzed ischemic time parameters as average rates over time rather than in an interrupted time series. Post-reperfusion outcomes were limited to hospital stay. Post-hospital follow-up would provide a better picture of the effects of STEMI intervention. There is no account of patients who died out-of-hospital secondary to acute STEMI. COVID-19 testing was not introduced until midway in our study period. Therefore, we cannot rule out the possibility of the SARS-Cov-2 virus inciting acute STEMI and subsequently leading to worse outcomes and poor prognosis.
Conclusions
This study provides an analysis of the incidence, characteristics, and clinical outcomes of patients presenting with acute STEMI during the early period of the COVID-19 pandemic. In-hospital mortality and ischemic time to reperfusion increased while combined in-hospital outcomes worsened.
Acknowledgment: The authors thank Piedmont Athens Regional IRB for approving this project and allowing access to patient data.
Corresponding author: Syed H. Ali; Department of Medicine, Medical College of Georgia at the Augusta University-University of Georgia Medical Partnership, 30606, Athens, GA; syedha.ali@gmail.com
Disclosures: None reported.
doi:10.12788/jcom.0085
From the Department of Medicine, Medical College of Georgia at the Augusta University-University of Georgia Medical Partnership, Athens, GA (Syed H. Ali, Syed Hyder, and Dr. Murrow), and the Department of Cardiology, Piedmont Heart Institute, Piedmont Athens Regional, Athens, GA (Dr. Murrow and Mrs. Davis).
Abstract
Objectives: The aim of this study was to describe the characteristics and in-hospital outcomes of patients with acute ST-segment elevation myocardial infarction (STEMI) during the early COVID-19 pandemic at Piedmont Athens Regional (PAR), a 330-bed tertiary referral center in Northeast Georgia.
Methods: A retrospective study was conducted at PAR to evaluate patients with acute STEMI admitted over an 8-week period during the initial COVID-19 outbreak. This study group was compared to patients admitted during the corresponding period in 2019. The primary endpoint of this study was defined as a composite of sustained ventricular arrhythmia, congestive heart failure (CHF) with pulmonary congestion, and/or in-hospital mortality.
Results: This study cohort was composed of 64 patients with acute STEMI; 30 patients (46.9%) were hospitalized during the COVID-19 pandemic. Patients with STEMI in both the COVID-19 and control groups had similar comorbidities, Killip classification score, and clinical presentations. The median (interquartile range) time from symptom onset to reperfusion (total ischemic time) increased from 99.5 minutes (84.8-132) in 2019 to 149 minutes (96.3-231.8; P = .032) in 2020. Hospitalization during the COVID-19 period was associated with an increased risk for combined in-hospital outcome (odds ratio, 3.96; P = .046).
Conclusion: Patients with STEMI admitted during the first wave of the COVID-19 outbreak experienced longer total ischemic time and increased risk for combined in-hospital outcomes compared to patients admitted during the corresponding period in 2019.
Keywords: myocardial infarction, acute coronary syndrome, hospitalization, outcomes.

The emergence of the SARS-Cov-2 virus in December 2019 caused a worldwide shift in resource allocation and the restructuring of health care systems within the span of a few months. With the rapid spread of infection, the World Health Organization officially declared a pandemic in March 2020. The pandemic led to the deferral and cancellation of in-person patient visits, routine diagnostic studies, and nonessential surgeries and procedures. This response occurred secondary to a joint effort to reduce transmission via stay-at-home mandates and appropriate social distancing.1
Alongside the reduction in elective procedures and health care visits, significant reductions in hospitalization rates due to decreases in acute ST-segment elevation myocardial infarction (STEMI) and catheterization laboratory utilization have been reported in many studies from around the world.2-7 Comprehensive data demonstrating the impact of the COVID-19 pandemic on acute STEMI patient characteristics, clinical presentation, and in-hospital outcomes are lacking. Although patients with previously diagnosed cardiovascular disease are more likely to encounter worse outcomes in the setting of COVID-19, there may also be an indirect impact of the pandemic on high-risk patients, including those without the infection.8 Several theories have been hypothesized to explain this phenomenon. One theory postulates that the fear of contracting the virus during hospitalization is great enough to prevent patients from seeking care.2 Another theory suggests that the increased utilization of telemedicine prevents exacerbation of chronic conditions and the need for hospitalization.9 Contrary to this trend, previous studies have shown an increased incidence of acute STEMI following stressful events such as natural disasters.10
The aim of this study was to describe trends pertaining to clinical characteristics and in-hospital outcomes of patients with acute STEMI during the early COVID-19 pandemic at Piedmont Athens Regional (PAR), a 330-bed tertiary referral center in Northeast Georgia.
Methods
A retrospective cohort study was conducted at PAR to evaluate patients with STEMI admitted to the cardiovascular intensive care unit over an 8-week period (March 5 to May 5, 2020) during the COVID-19 outbreak. COVID-19 was declared a national emergency on March 13, 2020, in the United States. The institutional review board at PAR approved the study; the need for individual consent was waived under the condition that participant data would undergo de-identification and be strictly safeguarded.
Data Collection
Because there are seasonal variations in cardiovascular admissions, patient data from a control period (March 9 to May 9, 2019) were obtained to compare with data from the 2020 period. The number of patients with the diagnosis of acute STEMI during the COVID-19 period was recorded. Demographic data, clinical characteristics, and primary angiographic findings were gathered for all patients. Time from symptom onset to hospital admission and time from hospital admission to reperfusion (defined as door-to-balloon time) were documented for each patient. Killip classification was used to assess patients’ clinical status on admission. Length of stay was determined as days from hospital admission to discharge or death (if occurring during the same hospitalization).
Adverse in-hospital complications were also recorded. These were selected based on inclusion of the following categories of acute STEMI complications: ischemic, mechanical, arrhythmic, embolic, and inflammatory. The following complications occurred in our patient cohort: sustained ventricular arrhythmia, congestive heart failure (CHF) defined as congestion requiring intravenous diuretics, re-infarction, mechanical complications (free-wall rupture, ventricular septal defect, or mitral regurgitation), second- or third-degree atrioventricular block, atrial fibrillation, stroke, mechanical ventilation, major bleeding, pericarditis, cardiogenic shock, cardiac arrest, and in-hospital mortality. The primary outcome of this study was defined as a composite of sustained ventricular arrhythmia, CHF with congestion requiring intravenous diuretics, and/or in-hospital mortality. Ventricular arrythmia and CHF were included in the composite outcome because they are defined as the 2 most common causes of sudden cardiac death following acute STEMI.11,12
Statistical Analysis
Normally distributed continuous variables and categorical variables were compared using the paired t-test. A 2-sided P value <.05 was considered to be statistically significant. Mean admission rates for acute STEMI hospitalizations were determined by dividing the number of admissions by the number of days in each time period. The daily rate of COVID-19 cases per 100,000 individuals was obtained from the Centers for Disease Control and Prevention COVID-19 database. All data analyses were performed using Microsoft Excel.
Results
The study cohort consisted of 64 patients, of whom 30 (46.9%) were hospitalized between March 5 and May 5, 2020, and 34 (53.1%) who were admitted during the analogous time period in 2019. This reflected a 6% decrease in STEMI admissions at PAR in the COVID-19 cohort.
Acute STEMI Hospitalization Rates and COVID-19 Incidence
The mean daily acute STEMI admission rate was 0.50 during the study period compared to 0.57 during the control period. During the study period in 2020 in the state of Georgia, the daily rate of newly confirmed COVID-19 cases ranged from 0.194 per 100,000 on March 5 to 8.778 per 100,000 on May 5. Results of COVID-19 testing were available for 9 STEMI patients, and of these 0 tests were positive.
Baseline Characteristics
Baseline characteristics of the acute STEMI cohorts are presented in Table 1. Approximately 75% were male; median (interquartile range [IQR]) age was 60 (51-72) years. There were no significant differences in age and gender between the study periods. Three-quarters of patients had a history of hypertension, and 87.5% had a history of dyslipidemia. There was no significant difference in baseline comorbidity profiles between the 2 study periods; therefore, our sample populations shared similar characteristics.

Clinical Presentation
Significant differences were observed regarding the time intervals of STEMI patients in the COVID-19 period and the control period (Table 2). Median time from symptom onset to hospital admission (patient delay) was extended from 57.5 minutes (IQR, 40.3-106) in 2019 to 93 minutes (IQR, 48.8-132) in 2020; however, this difference was not statistically significant (P = .697). Median time from hospital admission to reperfusion (system delay) was prolonged from 45 minutes (IQR, 28-61) in 2019 to 78 minutes (IQR, 50-110) in 2020 (P < .001). Overall time from symptom onset to reperfusion (total ischemic time) increased from 99.5 minutes (IQR, 84.8-132) in 2019 to 149 minutes (IQR, 96.3-231.8) in 2020 (P = .032).

Regarding mode of transportation, 23.5% of patients in 2019 were walk-in admissions to the emergency department. During the COVID-19 period, walk-in admissions decreased to 6.7% (P = .065). There were no significant differences between emergency medical service, transfer, or in-patient admissions for STEMI cases between the 2 study periods.
Killip classification scores were calculated for all patients on admission; 90.6% of patients were classified as Killip Class 1. There was no significant difference between hemodynamic presentations during the COVID-19 period compared to the control period.
Angiographic Data
Overall, 53 (82.8%) patients admitted with acute STEMI underwent coronary angiography during their hospital stay. The proportion of patients who underwent primary reperfusion was greater in the control period than in the COVID-19 period (85.3% vs 80%; P = .582). Angiographic characteristics and findings were similar between the 2 study groups (Table 2).
In-Hospital Outcomes
In-hospital outcome data were available for all patients. As shown in Table 3, hospitalization during the COVID-19 period was independently associated with an increased risk for combined in-hospital outcome (odds ratio, 3.96; P = .046). The rate of in-hospital mortality was greater in the COVID-19 period (P = .013). We found no significant difference when comparing secondary outcomes from admissions during the COVID-19 period and the control period in 2019. For the 5 patients who died during the study period, the primary diagnosis at death was acute STEMI complicated by CHF (3 patients) or cardiogenic shock (2 patients).

Discussion
This single-center retrospective study at PAR looks at the impact of COVID-19 on hospitalizations for acute STEMI during the initial peak of the pandemic. The key findings of this study show a significant increase in ischemic time parameters (symptom onset to reperfusion, hospital admission to reperfusion), in-hospital mortality, and combined in-hospital outcomes.
There was a 49.5-minute increase in total ischemic time noted in this study (P = .032). Though there was a numerical increase in time of symptom onset to hospital admission by 23.5 minutes, this difference was not statistically significant (P = .697). However, this study observed a statistically significant 33-minute increase in ischemic time from hospital admission to reperfusion (P < .001). Multiple studies globally have found a similar increase in total ischemic times, including those conducted in China and Europe.13-15 Every level of potential delay must be considered, including pre-hospital, triage and emergency department, and/or reperfusion team. Pre-hospital sources of delays that have been suggested include “stay-at-home” orders and apprehension to seek medical care due to concern about contracting the virus or overwhelming the health care facilities. There was a clinically significant 4-fold decrease in the number of walk-in acute STEMI cases in the study period. In 2019, there were 8 walk-in cases compared to 2 cases in 2020 (P = .065). However, this change was not statistically significant. In-hospital/systemic sources of delays have been mentioned in other studies; they include increased time taken to rule out COVID-19 (nasopharyngeal swab/chest x-ray) and increased time due to the need for intensive gowning and gloving procedures by staff. It was difficult to objectively determine the sources of system delay by the reperfusion team due to a lack of quantitative data.
In the current study, we found a significant increase in in-hospital mortality during the COVID-19 period compared to a parallel time frame in 2019. This finding is contrary to a multicenter study from Spain that reported no difference in in-hospital outcomes or mortality rates among all acute coronary syndrome cases.16 The worsening outcomes and prognosis may simply be a result of increased ischemic time; however, the virus that causes COVID-19 itself may play a role as well. Studies have found that SARS-Cov-2 infection places patients at greater risk for cardiovascular conditions such as hypercoagulability, myocarditis, and arrhythmias.17 In our study, however, there were no acute STEMI patients who tested positive for COVID-19. Therefore, we cannot discuss the impact of increased thrombus burden in patients with COVID-19. Piedmont Healthcare published a STEMI treatment protocol in May 2020 that advised increased use of tissue plasminogen activator (tPA) in COVID-19-positive cases; during the study period, however, there were no occasions when tPA use was deemed appropriate based on clinical judgment.
Our findings align with previous studies that describe an increase in combined in-hospital adverse outcomes during the COVID-19 era. Previous studies detected a higher rate of complications in the COVID-19 cohort, but in the current study, the adverse in-hospital course is unrelated to underlying infection.18,19 This study reports a higher incidence of major in-hospital outcomes, including a 65% increase in the rate of combined in-hospital outcomes, which is similar to a multicenter study conducted in Israel.19 There was a 2.3-fold numerical increase in sustained ventricular arrhythmias and a 2.5-fold numerical increase in the incidence of cardiac arrest in the study period. This phenomenon was observed despite a similar rate of reperfusion procedures in both groups.
Acute STEMI is a highly fatal condition with an incidence of 8.5 in 10,000 annually in the United States. While studies across the world have shown a 25% to 40% reduction in the rate of hospitalized acute coronary syndrome cases during the COVID-19 pandemic, the decrease from 34 to 30 STEMI admissions at PAR is not statistically significant.20 Possible reasons for the reduction globally include increased out-of-hospital mortality and decreased incidence of acute STEMI across the general population as a result of improved access to telemedicine or decreased levels of life stressors.20
In summary, there was an increase in ischemic time to reperfusion, in-hospital mortality, and combined in-hospital outcomes for acute STEMI patients at PAR during the COVID period.
Limitations
This study has several limitations. This is a single-center study, so the sample size is small and may not be generalizable to a larger population. This is a retrospective observational study, so causation cannot be inferred. This study analyzed ischemic time parameters as average rates over time rather than in an interrupted time series. Post-reperfusion outcomes were limited to hospital stay. Post-hospital follow-up would provide a better picture of the effects of STEMI intervention. There is no account of patients who died out-of-hospital secondary to acute STEMI. COVID-19 testing was not introduced until midway in our study period. Therefore, we cannot rule out the possibility of the SARS-Cov-2 virus inciting acute STEMI and subsequently leading to worse outcomes and poor prognosis.
Conclusions
This study provides an analysis of the incidence, characteristics, and clinical outcomes of patients presenting with acute STEMI during the early period of the COVID-19 pandemic. In-hospital mortality and ischemic time to reperfusion increased while combined in-hospital outcomes worsened.
Acknowledgment: The authors thank Piedmont Athens Regional IRB for approving this project and allowing access to patient data.
Corresponding author: Syed H. Ali; Department of Medicine, Medical College of Georgia at the Augusta University-University of Georgia Medical Partnership, 30606, Athens, GA; syedha.ali@gmail.com
Disclosures: None reported.
doi:10.12788/jcom.0085
1. Bhatt AS, Moscone A, McElrath EE, et al. Fewer hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J Am Coll Cardiol. 2020;76(3):280-288. doi:10.1016/j.jacc.2020.05.038
2. Metzler B, Siostrzonek P, Binder RK, Bauer A, Reinstadler SJR. Decline of acute coronary syndrome admissions in Austria since the outbreak of Covid-19: the pandemic response causes cardiac collateral damage. Eur Heart J. 2020;41:1852-1853. doi:10.1093/eurheartj/ehaa314
3. De Rosa S, Spaccarotella C, Basso C, et al. Reduction of hospitalizations for myocardial infarction in Italy in the Covid-19 era. Eur Heart J. 2020;41(22):2083-2088.
4. Wilson SJ, Connolly MJ, Elghamry Z, et al. Effect of the COVID-19 pandemic on ST-segment-elevation myocardial infarction presentations and in-hospital outcomes. Circ Cardiovasc Interv. 2020; 13(7):e009438. doi:10.1161/CIRCINTERVENTIONS.120.009438
5. Mafham MM, Spata E, Goldacre R, et al. Covid-19 pandemic and admission rates for and management of acute coronary syndromes in England. Lancet. 2020;396 (10248):381-389. doi:10.1016/S0140-6736(20)31356-8
6. Bhatt AS, Moscone A, McElrath EE, et al. Fewer Hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J Am Coll Cardiol. 2020;76(3):280-288. doi:10.1016/j.jacc.2020.05.038
7. Tam CF, Cheung KS, Lam S, et al. Impact of Coronavirus disease 2019 (Covid-19) outbreak on ST-segment elevation myocardial infarction care in Hong Kong, China. Circ Cardiovasc Qual Outcomes. 2020;13(4):e006631. doi:10.1161/CIRCOUTCOMES.120.006631
8. Clerkin KJ, Fried JA, Raikhelkar J, et al. Coronavirus disease 2019 (COVID-19) and cardiovascular disease. Circulation. 2020;141:1648-1655. doi:10.1161/CIRCULATIONAHA.120.046941
9. Ebinger JE, Shah PK. Declining admissions for acute cardiovascular illness: The Covid-19 paradox. J Am Coll Cardiol. 2020;76(3):289-291. doi:10.1016/j.jacc.2020.05.039
10 Leor J, Poole WK, Kloner RA. Sudden cardiac death triggered by an earthquake. N Engl J Med. 1996;334(7):413-419. doi:10.1056/NEJM199602153340701
11. Hiramori K. Major causes of death from acute myocardial infarction in a coronary care unit. Jpn Circ J. 1987;51(9):1041-1047. doi:10.1253/jcj.51.1041
12. Bui AH, Waks JW. Risk stratification of sudden cardiac death after acute myocardial infarction. J Innov Card Rhythm Manag. 2018;9(2):3035-3049. doi:10.19102/icrm.2018.090201
13. Xiang D, Xiang X, Zhang W, et al. Management and outcomes of patients with STEMI during the COVID-19 pandemic in China. J Am Coll Cardiol. 2020;76(11):1318-1324. doi:10.1016/j.jacc.2020.06.039
14. Hakim R, Motreff P, Rangé G. COVID-19 and STEMI. [Article in French]. Ann Cardiol Angeiol (Paris). 2020;69(6):355-359. doi:10.1016/j.ancard.2020.09.034
15. Soylu K, Coksevim M, Yanık A, Bugra Cerik I, Aksan G. Effect of Covid-19 pandemic process on STEMI patients timeline. Int J Clin Pract. 2021;75(5):e14005. doi:10.1111/ijcp.14005
16. Salinas P, Travieso A, Vergara-Uzcategui C, et al. Clinical profile and 30-day mortality of invasively managed patients with suspected acute coronary syndrome during the COVID-19 outbreak. Int Heart J. 2021;62(2):274-281. doi:10.1536/ihj.20-574
17. Hu Y, Sun J, Dai Z, et al. Prevalence and severity of corona virus disease 2019 (Covid-19): a systematic review and meta-analysis. J Clin Virol. 2020;127:104371. doi:10.1016/j.jcv.2020.104371
18. Rodriguez-Leor O, Cid Alvarez AB, Perez de Prado A, et al. In-hospital outcomes of COVID-19 ST-elevation myocardial infarction patients. EuroIntervention. 2021;16(17):1426-1433. doi:10.4244/EIJ-D-20-00935
19. Fardman A, Zahger D, Orvin K, et al. Acute myocardial infarction in the Covid-19 era: incidence, clinical characteristics and in-hospital outcomes—A multicenter registry. PLoS ONE. 2021;16(6): e0253524. doi:10.1371/journal.pone.0253524
20. Pessoa-Amorim G, Camm CF, Gajendragadkar P, et al. Admission of patients with STEMI since the outbreak of the COVID-19 pandemic: a survey by the European Society of Cardiology. Eur Heart J Qual Care Clin Outcomes. 2020;6(3):210-216. doi:10.1093/ehjqcco/qcaa046
1. Bhatt AS, Moscone A, McElrath EE, et al. Fewer hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J Am Coll Cardiol. 2020;76(3):280-288. doi:10.1016/j.jacc.2020.05.038
2. Metzler B, Siostrzonek P, Binder RK, Bauer A, Reinstadler SJR. Decline of acute coronary syndrome admissions in Austria since the outbreak of Covid-19: the pandemic response causes cardiac collateral damage. Eur Heart J. 2020;41:1852-1853. doi:10.1093/eurheartj/ehaa314
3. De Rosa S, Spaccarotella C, Basso C, et al. Reduction of hospitalizations for myocardial infarction in Italy in the Covid-19 era. Eur Heart J. 2020;41(22):2083-2088.
4. Wilson SJ, Connolly MJ, Elghamry Z, et al. Effect of the COVID-19 pandemic on ST-segment-elevation myocardial infarction presentations and in-hospital outcomes. Circ Cardiovasc Interv. 2020; 13(7):e009438. doi:10.1161/CIRCINTERVENTIONS.120.009438
5. Mafham MM, Spata E, Goldacre R, et al. Covid-19 pandemic and admission rates for and management of acute coronary syndromes in England. Lancet. 2020;396 (10248):381-389. doi:10.1016/S0140-6736(20)31356-8
6. Bhatt AS, Moscone A, McElrath EE, et al. Fewer Hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J Am Coll Cardiol. 2020;76(3):280-288. doi:10.1016/j.jacc.2020.05.038
7. Tam CF, Cheung KS, Lam S, et al. Impact of Coronavirus disease 2019 (Covid-19) outbreak on ST-segment elevation myocardial infarction care in Hong Kong, China. Circ Cardiovasc Qual Outcomes. 2020;13(4):e006631. doi:10.1161/CIRCOUTCOMES.120.006631
8. Clerkin KJ, Fried JA, Raikhelkar J, et al. Coronavirus disease 2019 (COVID-19) and cardiovascular disease. Circulation. 2020;141:1648-1655. doi:10.1161/CIRCULATIONAHA.120.046941
9. Ebinger JE, Shah PK. Declining admissions for acute cardiovascular illness: The Covid-19 paradox. J Am Coll Cardiol. 2020;76(3):289-291. doi:10.1016/j.jacc.2020.05.039
10 Leor J, Poole WK, Kloner RA. Sudden cardiac death triggered by an earthquake. N Engl J Med. 1996;334(7):413-419. doi:10.1056/NEJM199602153340701
11. Hiramori K. Major causes of death from acute myocardial infarction in a coronary care unit. Jpn Circ J. 1987;51(9):1041-1047. doi:10.1253/jcj.51.1041
12. Bui AH, Waks JW. Risk stratification of sudden cardiac death after acute myocardial infarction. J Innov Card Rhythm Manag. 2018;9(2):3035-3049. doi:10.19102/icrm.2018.090201
13. Xiang D, Xiang X, Zhang W, et al. Management and outcomes of patients with STEMI during the COVID-19 pandemic in China. J Am Coll Cardiol. 2020;76(11):1318-1324. doi:10.1016/j.jacc.2020.06.039
14. Hakim R, Motreff P, Rangé G. COVID-19 and STEMI. [Article in French]. Ann Cardiol Angeiol (Paris). 2020;69(6):355-359. doi:10.1016/j.ancard.2020.09.034
15. Soylu K, Coksevim M, Yanık A, Bugra Cerik I, Aksan G. Effect of Covid-19 pandemic process on STEMI patients timeline. Int J Clin Pract. 2021;75(5):e14005. doi:10.1111/ijcp.14005
16. Salinas P, Travieso A, Vergara-Uzcategui C, et al. Clinical profile and 30-day mortality of invasively managed patients with suspected acute coronary syndrome during the COVID-19 outbreak. Int Heart J. 2021;62(2):274-281. doi:10.1536/ihj.20-574
17. Hu Y, Sun J, Dai Z, et al. Prevalence and severity of corona virus disease 2019 (Covid-19): a systematic review and meta-analysis. J Clin Virol. 2020;127:104371. doi:10.1016/j.jcv.2020.104371
18. Rodriguez-Leor O, Cid Alvarez AB, Perez de Prado A, et al. In-hospital outcomes of COVID-19 ST-elevation myocardial infarction patients. EuroIntervention. 2021;16(17):1426-1433. doi:10.4244/EIJ-D-20-00935
19. Fardman A, Zahger D, Orvin K, et al. Acute myocardial infarction in the Covid-19 era: incidence, clinical characteristics and in-hospital outcomes—A multicenter registry. PLoS ONE. 2021;16(6): e0253524. doi:10.1371/journal.pone.0253524
20. Pessoa-Amorim G, Camm CF, Gajendragadkar P, et al. Admission of patients with STEMI since the outbreak of the COVID-19 pandemic: a survey by the European Society of Cardiology. Eur Heart J Qual Care Clin Outcomes. 2020;6(3):210-216. doi:10.1093/ehjqcco/qcaa046
Early Hospital Discharge Following PCI for Patients With STEMI
Study Overview
Objective: To assess the safety and efficacy of early hospital discharge (EHD) for selected low-risk patients with ST-segment elevation myocardial infarction (STEMI) after primary percutaneous coronary intervention (PCI).
Design: Single-center retrospective analysis of prospectively collected data.
Setting and participants: An EHD group comprised of 600 patients who were discharged at <48 hours between April 2020 and June 2021 was compared to a control group of 700 patients who met EHD criteria but were discharged at >48 hour between October 2018 and June 2021. Patients were selected into the EHD group based on the following criteria, in accordance with recommendations from the European Society of Cardiology, and all patients had close follow-up with a combination of structured telephone follow-up at 48 hours post discharge and virtual visits at 2, 6, and 8 weeks and at 3 months:
- Left ventricular ejection fraction ≥40%
- Successful primary PCI (that achieved thrombolysis in myocardial infarction flow grade 3)
- Absence of severe nonculprit disease requiring further inpatient revascularization
- Absence of ischemic symptoms post PCI
- Absence of heart failure or hemodynamic instability
- Absence of significant arrhythmia (ventricular fibrillation, ventricular tachycardia, or atrial fibrillation or atrial flutter requiring prolonged stay)
- Mobility with suitable social circumstances for discharge
Main outcome measures: The outcomes measured were length of hospitalization and a composite primary endpoint of cardiovascular mortality and major adverse cardiovascular event (MACE) rates, defined as a composite of all-cause mortality, recurrent MI, and target lesion revascularization.
Main results: The median length of stay of hospitalization in the EHD group was 24.6 hours compared to 56.1 hours in the >48-hour historical control group. On median follow-up of 271 days, the EHD group demonstrated 0% cardiovascular mortality and a MACE rate of 1.2%. This was shown to be noninferior compared to the >48-hour historical control group, which had mortality of 0.7% and a MACE rate of 1.9%.
Conclusion: Selected low-risk STEMI patients can be safely discharged early with appropriate follow-up after primary PCI.
Commentary
Patients with STEMI have a higher risk of postprocedural adverse events such as MI, arrhythmia, or acute heart failure compared to patients with stable ischemic heart disease, and thus are monitored after primary PCI. Although patients were traditionally monitored for 5 to 7 days a few decades ago,1 with improvements in PCI techniques, devices, and pharmacotherapy as well as in door-to-balloon time, the in-hospital complication rates for patients with STEMI have been decreasing, leading to earlier discharge. Currently in the United States, patients are most commonly monitored for 48 to 72 hours post PCI.2 The current guidelines support this practice, recommending early discharge within 48 to 72 hours in selected low-risk patients if adequate follow-up and rehabilitation are arranged.3
Given the COVID-19 pandemic and decreased hospital bed availability, Rathod et al took one step further on the question of whether low-risk STEMI patients with primary PCI can be discharged safely within 48 hours with adequate follow-up. They found that at a median follow-up of 271 days, EHD patients had 2 COVID-related deaths, with 0% cardiovascular mortality and a MACE rate of 1.2%, including deaths, MI, and ischemic revascularization. The median time to discharge was 25 hours. This was noninferior to the >48-hour historical control group, which had mortality of 0.7% (P = 0.349) and a MACE rate of 1.9% (P = .674). The results remained similar after propensity matching for mortality (0.34% vs 0.69%, P = .410) or MACE (1.2% vs 1.9%, P = .342).
This is the first prospective study to systematically assess the safety and feasibility of discharge of low-risk STEMI patients with primary PCI within 48 hours. This study is unique in that it involved the use of telemedicine, including a virtual platform to collect data such as heart rate, blood pressure, and blood glucose, and virtual visits to facilitate follow-up and reduce clinic travel, cost, and potential COVID-19 exposure. The investigators’ protocol included virtual follow-up by cardiology advanced practitioners at 2, 6, and 8 weeks and by an interventional cardiologist at 12 weeks. This protocol led to an increase in patient satisfaction. The study’s main limitation is that it is a single-center trial with a smaller sample size. Further studies are necessary to confirm the safety and feasibility of this approach. In addition, further refinement of the patient selection criteria for EHD should be considered.
Applications for Clinical Practice
In low-risk STEMI patients after primary PCI, discharge within 48 hours may be considered if close follow-up is arranged. However, further studies are necessary to confirm this finding.
—Thai Nguyen, MD, Albert Chan, MD, and Taishi Hirai MD
1. Grines CL, Marsalese DL, Brodie B, et al. Safety and cost-effectiveness of early discharge after primary angioplasty in low risk patients with acute myocardial infarction. PAMI-II Investigators. Primary Angioplasty in Myocardial Infarction. J Am Coll Cardiol. 1998;31:967-72. doi:10.1016/s0735-1097(98)00031-x
2. Seto AH, Shroff A, Abu-Fadel M, et al. Length of stay following percutaneous coronary intervention: An expert consensus document update from the society for cardiovascular angiography and interventions. Catheter Cardiovasc Interv. 2018;92:717-731. doi:10.1002/ccd.27637
3. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2018;39:119-177. doi:10.1093/eurheartj/ehx393
Study Overview
Objective: To assess the safety and efficacy of early hospital discharge (EHD) for selected low-risk patients with ST-segment elevation myocardial infarction (STEMI) after primary percutaneous coronary intervention (PCI).
Design: Single-center retrospective analysis of prospectively collected data.
Setting and participants: An EHD group comprised of 600 patients who were discharged at <48 hours between April 2020 and June 2021 was compared to a control group of 700 patients who met EHD criteria but were discharged at >48 hour between October 2018 and June 2021. Patients were selected into the EHD group based on the following criteria, in accordance with recommendations from the European Society of Cardiology, and all patients had close follow-up with a combination of structured telephone follow-up at 48 hours post discharge and virtual visits at 2, 6, and 8 weeks and at 3 months:
- Left ventricular ejection fraction ≥40%
- Successful primary PCI (that achieved thrombolysis in myocardial infarction flow grade 3)
- Absence of severe nonculprit disease requiring further inpatient revascularization
- Absence of ischemic symptoms post PCI
- Absence of heart failure or hemodynamic instability
- Absence of significant arrhythmia (ventricular fibrillation, ventricular tachycardia, or atrial fibrillation or atrial flutter requiring prolonged stay)
- Mobility with suitable social circumstances for discharge
Main outcome measures: The outcomes measured were length of hospitalization and a composite primary endpoint of cardiovascular mortality and major adverse cardiovascular event (MACE) rates, defined as a composite of all-cause mortality, recurrent MI, and target lesion revascularization.
Main results: The median length of stay of hospitalization in the EHD group was 24.6 hours compared to 56.1 hours in the >48-hour historical control group. On median follow-up of 271 days, the EHD group demonstrated 0% cardiovascular mortality and a MACE rate of 1.2%. This was shown to be noninferior compared to the >48-hour historical control group, which had mortality of 0.7% and a MACE rate of 1.9%.
Conclusion: Selected low-risk STEMI patients can be safely discharged early with appropriate follow-up after primary PCI.
Commentary
Patients with STEMI have a higher risk of postprocedural adverse events such as MI, arrhythmia, or acute heart failure compared to patients with stable ischemic heart disease, and thus are monitored after primary PCI. Although patients were traditionally monitored for 5 to 7 days a few decades ago,1 with improvements in PCI techniques, devices, and pharmacotherapy as well as in door-to-balloon time, the in-hospital complication rates for patients with STEMI have been decreasing, leading to earlier discharge. Currently in the United States, patients are most commonly monitored for 48 to 72 hours post PCI.2 The current guidelines support this practice, recommending early discharge within 48 to 72 hours in selected low-risk patients if adequate follow-up and rehabilitation are arranged.3
Given the COVID-19 pandemic and decreased hospital bed availability, Rathod et al took one step further on the question of whether low-risk STEMI patients with primary PCI can be discharged safely within 48 hours with adequate follow-up. They found that at a median follow-up of 271 days, EHD patients had 2 COVID-related deaths, with 0% cardiovascular mortality and a MACE rate of 1.2%, including deaths, MI, and ischemic revascularization. The median time to discharge was 25 hours. This was noninferior to the >48-hour historical control group, which had mortality of 0.7% (P = 0.349) and a MACE rate of 1.9% (P = .674). The results remained similar after propensity matching for mortality (0.34% vs 0.69%, P = .410) or MACE (1.2% vs 1.9%, P = .342).
This is the first prospective study to systematically assess the safety and feasibility of discharge of low-risk STEMI patients with primary PCI within 48 hours. This study is unique in that it involved the use of telemedicine, including a virtual platform to collect data such as heart rate, blood pressure, and blood glucose, and virtual visits to facilitate follow-up and reduce clinic travel, cost, and potential COVID-19 exposure. The investigators’ protocol included virtual follow-up by cardiology advanced practitioners at 2, 6, and 8 weeks and by an interventional cardiologist at 12 weeks. This protocol led to an increase in patient satisfaction. The study’s main limitation is that it is a single-center trial with a smaller sample size. Further studies are necessary to confirm the safety and feasibility of this approach. In addition, further refinement of the patient selection criteria for EHD should be considered.
Applications for Clinical Practice
In low-risk STEMI patients after primary PCI, discharge within 48 hours may be considered if close follow-up is arranged. However, further studies are necessary to confirm this finding.
—Thai Nguyen, MD, Albert Chan, MD, and Taishi Hirai MD
Study Overview
Objective: To assess the safety and efficacy of early hospital discharge (EHD) for selected low-risk patients with ST-segment elevation myocardial infarction (STEMI) after primary percutaneous coronary intervention (PCI).
Design: Single-center retrospective analysis of prospectively collected data.
Setting and participants: An EHD group comprised of 600 patients who were discharged at <48 hours between April 2020 and June 2021 was compared to a control group of 700 patients who met EHD criteria but were discharged at >48 hour between October 2018 and June 2021. Patients were selected into the EHD group based on the following criteria, in accordance with recommendations from the European Society of Cardiology, and all patients had close follow-up with a combination of structured telephone follow-up at 48 hours post discharge and virtual visits at 2, 6, and 8 weeks and at 3 months:
- Left ventricular ejection fraction ≥40%
- Successful primary PCI (that achieved thrombolysis in myocardial infarction flow grade 3)
- Absence of severe nonculprit disease requiring further inpatient revascularization
- Absence of ischemic symptoms post PCI
- Absence of heart failure or hemodynamic instability
- Absence of significant arrhythmia (ventricular fibrillation, ventricular tachycardia, or atrial fibrillation or atrial flutter requiring prolonged stay)
- Mobility with suitable social circumstances for discharge
Main outcome measures: The outcomes measured were length of hospitalization and a composite primary endpoint of cardiovascular mortality and major adverse cardiovascular event (MACE) rates, defined as a composite of all-cause mortality, recurrent MI, and target lesion revascularization.
Main results: The median length of stay of hospitalization in the EHD group was 24.6 hours compared to 56.1 hours in the >48-hour historical control group. On median follow-up of 271 days, the EHD group demonstrated 0% cardiovascular mortality and a MACE rate of 1.2%. This was shown to be noninferior compared to the >48-hour historical control group, which had mortality of 0.7% and a MACE rate of 1.9%.
Conclusion: Selected low-risk STEMI patients can be safely discharged early with appropriate follow-up after primary PCI.
Commentary
Patients with STEMI have a higher risk of postprocedural adverse events such as MI, arrhythmia, or acute heart failure compared to patients with stable ischemic heart disease, and thus are monitored after primary PCI. Although patients were traditionally monitored for 5 to 7 days a few decades ago,1 with improvements in PCI techniques, devices, and pharmacotherapy as well as in door-to-balloon time, the in-hospital complication rates for patients with STEMI have been decreasing, leading to earlier discharge. Currently in the United States, patients are most commonly monitored for 48 to 72 hours post PCI.2 The current guidelines support this practice, recommending early discharge within 48 to 72 hours in selected low-risk patients if adequate follow-up and rehabilitation are arranged.3
Given the COVID-19 pandemic and decreased hospital bed availability, Rathod et al took one step further on the question of whether low-risk STEMI patients with primary PCI can be discharged safely within 48 hours with adequate follow-up. They found that at a median follow-up of 271 days, EHD patients had 2 COVID-related deaths, with 0% cardiovascular mortality and a MACE rate of 1.2%, including deaths, MI, and ischemic revascularization. The median time to discharge was 25 hours. This was noninferior to the >48-hour historical control group, which had mortality of 0.7% (P = 0.349) and a MACE rate of 1.9% (P = .674). The results remained similar after propensity matching for mortality (0.34% vs 0.69%, P = .410) or MACE (1.2% vs 1.9%, P = .342).
This is the first prospective study to systematically assess the safety and feasibility of discharge of low-risk STEMI patients with primary PCI within 48 hours. This study is unique in that it involved the use of telemedicine, including a virtual platform to collect data such as heart rate, blood pressure, and blood glucose, and virtual visits to facilitate follow-up and reduce clinic travel, cost, and potential COVID-19 exposure. The investigators’ protocol included virtual follow-up by cardiology advanced practitioners at 2, 6, and 8 weeks and by an interventional cardiologist at 12 weeks. This protocol led to an increase in patient satisfaction. The study’s main limitation is that it is a single-center trial with a smaller sample size. Further studies are necessary to confirm the safety and feasibility of this approach. In addition, further refinement of the patient selection criteria for EHD should be considered.
Applications for Clinical Practice
In low-risk STEMI patients after primary PCI, discharge within 48 hours may be considered if close follow-up is arranged. However, further studies are necessary to confirm this finding.
—Thai Nguyen, MD, Albert Chan, MD, and Taishi Hirai MD
1. Grines CL, Marsalese DL, Brodie B, et al. Safety and cost-effectiveness of early discharge after primary angioplasty in low risk patients with acute myocardial infarction. PAMI-II Investigators. Primary Angioplasty in Myocardial Infarction. J Am Coll Cardiol. 1998;31:967-72. doi:10.1016/s0735-1097(98)00031-x
2. Seto AH, Shroff A, Abu-Fadel M, et al. Length of stay following percutaneous coronary intervention: An expert consensus document update from the society for cardiovascular angiography and interventions. Catheter Cardiovasc Interv. 2018;92:717-731. doi:10.1002/ccd.27637
3. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2018;39:119-177. doi:10.1093/eurheartj/ehx393
1. Grines CL, Marsalese DL, Brodie B, et al. Safety and cost-effectiveness of early discharge after primary angioplasty in low risk patients with acute myocardial infarction. PAMI-II Investigators. Primary Angioplasty in Myocardial Infarction. J Am Coll Cardiol. 1998;31:967-72. doi:10.1016/s0735-1097(98)00031-x
2. Seto AH, Shroff A, Abu-Fadel M, et al. Length of stay following percutaneous coronary intervention: An expert consensus document update from the society for cardiovascular angiography and interventions. Catheter Cardiovasc Interv. 2018;92:717-731. doi:10.1002/ccd.27637
3. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2018;39:119-177. doi:10.1093/eurheartj/ehx393
Walking 10,000 steps a day: Desirable goal or urban myth?
Some myths never die. The idea of taking 10,000 steps a day is one of them. What started as a catchy marketing slogan has become a mantra for anyone promoting physical activity.
It all began in 1965 when the Japanese company Yamasa Tokei began selling a new step-counter which they called manpo-kei (ten-thousand steps meter). They coupled the product launch with an ad campaign – “Let’s walk 10,000 steps a day!” – in a bid to encourage physical activity. The threshold was always somewhat arbitrary, but the idea of 10,000 steps cemented itself in the public consciousness from that point forward.
To be fair, there is nothing wrong with taking 10,000 steps a day, and it does roughly correlate with the generally recommended amount of physical activity. Most people will take somewhere between 5,000 and 7,500 steps a day even if they lead largely sedentary lives. If you add 30 minutes of walking to your daily routine, that will account for an extra 3,000-4,000 steps and bring you close to that 10,000-step threshold. As such, setting a 10,000-step target is a potentially useful shorthand for people aspiring to achieve ideal levels of physical activity.
But walking fewer steps still has a benefit. A study in JAMA Network Open followed a cohort of 2,110 adults from the CARDIA study and found, rather unsurprisingly, that those with more steps per day had lower rates of all-cause mortality. But interestingly, those who averaged 7,000-10,000 steps per day did just as well as those who walked more than 10,000 steps, suggesting that the lower threshold was probably the inflection point.
Other research has shown that improving your step count is probably more important than achieving any specific threshold. In one Canadian study, patients with diabetes were randomized to usual care or to an exercise prescription from their physicians. The intervention group improved their daily step count from around 5,000 steps per day to about 6,200 steps per day. While the increase was less than the researchers had hoped for, it still resulted in improvements in blood sugar control. In another study, a 24-week walking program reduced blood pressure by 11 points in postmenopausal women, even though their increased daily step counts fell shy of the 10,000 goal at about 9,000 steps. Similarly, a small Japanese study found that enrolling postmenopausal women in a weekly exercise program helped improve their lipid profile even though they only increased their daily step count from 6,800 to 8,500 steps per day. And an analysis of U.S. NHANES data showed a mortality benefit when individuals taking more than 8,000 steps were compared with those taking fewer than 4,000 steps per day. The benefits largely plateaued beyond 9,000-10,000 steps.
The reality is that walking 10,000 steps a day is a laudable goal and is almost certainly beneficial. But even lower levels of physical activity have benefits. The trick is not so much to aim for some theoretical ideal but to improve upon your current baseline. Encouraging patients to get into the habit of taking a daily walk (be it in the morning, during lunchtime, or in the evening) is going to pay dividends regardless of their daily step count. The point is that when it comes to physical activity, the greatest benefit seems to be when we go from doing nothing to doing something.
Dr. Labos is a cardiologist at Queen Elizabeth Health Complex, Montreal. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Some myths never die. The idea of taking 10,000 steps a day is one of them. What started as a catchy marketing slogan has become a mantra for anyone promoting physical activity.
It all began in 1965 when the Japanese company Yamasa Tokei began selling a new step-counter which they called manpo-kei (ten-thousand steps meter). They coupled the product launch with an ad campaign – “Let’s walk 10,000 steps a day!” – in a bid to encourage physical activity. The threshold was always somewhat arbitrary, but the idea of 10,000 steps cemented itself in the public consciousness from that point forward.
To be fair, there is nothing wrong with taking 10,000 steps a day, and it does roughly correlate with the generally recommended amount of physical activity. Most people will take somewhere between 5,000 and 7,500 steps a day even if they lead largely sedentary lives. If you add 30 minutes of walking to your daily routine, that will account for an extra 3,000-4,000 steps and bring you close to that 10,000-step threshold. As such, setting a 10,000-step target is a potentially useful shorthand for people aspiring to achieve ideal levels of physical activity.
But walking fewer steps still has a benefit. A study in JAMA Network Open followed a cohort of 2,110 adults from the CARDIA study and found, rather unsurprisingly, that those with more steps per day had lower rates of all-cause mortality. But interestingly, those who averaged 7,000-10,000 steps per day did just as well as those who walked more than 10,000 steps, suggesting that the lower threshold was probably the inflection point.
Other research has shown that improving your step count is probably more important than achieving any specific threshold. In one Canadian study, patients with diabetes were randomized to usual care or to an exercise prescription from their physicians. The intervention group improved their daily step count from around 5,000 steps per day to about 6,200 steps per day. While the increase was less than the researchers had hoped for, it still resulted in improvements in blood sugar control. In another study, a 24-week walking program reduced blood pressure by 11 points in postmenopausal women, even though their increased daily step counts fell shy of the 10,000 goal at about 9,000 steps. Similarly, a small Japanese study found that enrolling postmenopausal women in a weekly exercise program helped improve their lipid profile even though they only increased their daily step count from 6,800 to 8,500 steps per day. And an analysis of U.S. NHANES data showed a mortality benefit when individuals taking more than 8,000 steps were compared with those taking fewer than 4,000 steps per day. The benefits largely plateaued beyond 9,000-10,000 steps.
The reality is that walking 10,000 steps a day is a laudable goal and is almost certainly beneficial. But even lower levels of physical activity have benefits. The trick is not so much to aim for some theoretical ideal but to improve upon your current baseline. Encouraging patients to get into the habit of taking a daily walk (be it in the morning, during lunchtime, or in the evening) is going to pay dividends regardless of their daily step count. The point is that when it comes to physical activity, the greatest benefit seems to be when we go from doing nothing to doing something.
Dr. Labos is a cardiologist at Queen Elizabeth Health Complex, Montreal. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Some myths never die. The idea of taking 10,000 steps a day is one of them. What started as a catchy marketing slogan has become a mantra for anyone promoting physical activity.
It all began in 1965 when the Japanese company Yamasa Tokei began selling a new step-counter which they called manpo-kei (ten-thousand steps meter). They coupled the product launch with an ad campaign – “Let’s walk 10,000 steps a day!” – in a bid to encourage physical activity. The threshold was always somewhat arbitrary, but the idea of 10,000 steps cemented itself in the public consciousness from that point forward.
To be fair, there is nothing wrong with taking 10,000 steps a day, and it does roughly correlate with the generally recommended amount of physical activity. Most people will take somewhere between 5,000 and 7,500 steps a day even if they lead largely sedentary lives. If you add 30 minutes of walking to your daily routine, that will account for an extra 3,000-4,000 steps and bring you close to that 10,000-step threshold. As such, setting a 10,000-step target is a potentially useful shorthand for people aspiring to achieve ideal levels of physical activity.
But walking fewer steps still has a benefit. A study in JAMA Network Open followed a cohort of 2,110 adults from the CARDIA study and found, rather unsurprisingly, that those with more steps per day had lower rates of all-cause mortality. But interestingly, those who averaged 7,000-10,000 steps per day did just as well as those who walked more than 10,000 steps, suggesting that the lower threshold was probably the inflection point.
Other research has shown that improving your step count is probably more important than achieving any specific threshold. In one Canadian study, patients with diabetes were randomized to usual care or to an exercise prescription from their physicians. The intervention group improved their daily step count from around 5,000 steps per day to about 6,200 steps per day. While the increase was less than the researchers had hoped for, it still resulted in improvements in blood sugar control. In another study, a 24-week walking program reduced blood pressure by 11 points in postmenopausal women, even though their increased daily step counts fell shy of the 10,000 goal at about 9,000 steps. Similarly, a small Japanese study found that enrolling postmenopausal women in a weekly exercise program helped improve their lipid profile even though they only increased their daily step count from 6,800 to 8,500 steps per day. And an analysis of U.S. NHANES data showed a mortality benefit when individuals taking more than 8,000 steps were compared with those taking fewer than 4,000 steps per day. The benefits largely plateaued beyond 9,000-10,000 steps.
The reality is that walking 10,000 steps a day is a laudable goal and is almost certainly beneficial. But even lower levels of physical activity have benefits. The trick is not so much to aim for some theoretical ideal but to improve upon your current baseline. Encouraging patients to get into the habit of taking a daily walk (be it in the morning, during lunchtime, or in the evening) is going to pay dividends regardless of their daily step count. The point is that when it comes to physical activity, the greatest benefit seems to be when we go from doing nothing to doing something.
Dr. Labos is a cardiologist at Queen Elizabeth Health Complex, Montreal. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Kawasaki disease guideline highlights rheumatology angles
All Kawasaki disease (KD) patients should be treated first with intravenous immunoglobulin, according to an updated guideline issued jointly by the American College of Rheumatology and the Vasculitis Foundation.
KD has low mortality when treated appropriately, guideline first author Mark Gorelik, MD, assistant professor of pediatrics at Columbia University, New York, and colleagues wrote.
The update is important at this time because new evidence continues to emerge in the clinical management of KD, Dr. Gorelik said in an interview.
“In addition, this guideline approaches Kawasaki disease from a perspective of acting as an adjunct to the already existing and excellent American Heart Association guidelines by adding information in areas that rheumatologists may play a role,” Dr. Gorelik said. “This is specifically regarding patients who may require additional therapy beyond standard IVIg, such as patients who may be at higher risk of morbidity from disease and patients who have refractory disease,” he explained.
The guideline, published in Arthritis & Rheumatology, includes 11 recommendations, 1 good practice statement, and 1 ungraded position statement. The good practice statement emphasizes that all patients with KD should be initially treated with IVIg.
The position statement advises that either nonglucocorticoid immunosuppressive therapy or glucocorticoids may be used for patients with acute KD whose fever persists despite repeated IVIg treatment. No clinical evidence currently supports the superiority of either nonglucocorticoid immunosuppressive therapy or glucocorticoids; therefore, the authors support the use of either based on what is appropriate in any given clinical situation. Although optimal dosage and duration of glucocorticoids have yet to be determined in a U.S. population, the authors described a typical glucocorticoid dosage as starting prednisone at 2 mg/kg per day, with a maximum of 60 mg/day, and dose tapering over 15 days.
The 11 recommendations consist of 7 strong and 4 conditional recommendations. The strong recommendations focus on prompt treatment of incomplete KD, treatment with aspirin, and obtaining an echocardiogram in patients with unexplained macrophage activation syndrome or shock. The conditional recommendations support using established therapy promptly at disease onset, then identifying cases in which additional therapy is needed.
Dr. Gorelik highlighted four clinical takeaways from the guideline. First, “patients with higher risk for complications do exist in Kawasaki disease, and that these patients can be treated more aggressively,” he said. “Specifically, patients with aneurysms seen at first ultrasound, and patients who are under 6 months, are more likely to have progressive and/or refractory disease; these patients can be treated with an adjunctive short course of corticosteroids.”
Second, “the use of high-dose aspirin for patients with Kawasaki disease does not have strong basis in evidence. While aspirin itself of some dose is necessary for patients with Kawasaki disease, use of either high- or low-dose aspirin has the same outcome for patients, and a physician may choose either of these in practice,” he said.
Third, “we continue to recommend that refractory patients with Kawasaki disease be treated with a second dose of IVIg; however, there are many scenarios in which a physician may choose either corticosteroids [either a single high dose of >10 mg/kg, or a short moderate-dose course of 2 mg/kg per day for 5-7 days] or a biologic agent such as infliximab. ... These are valid choices for therapy in patients with refractory Kawasaki disease,” he emphasized.
Fourth, “physicians should discard the idea of treating before [and conversely, not treating after] 10 days of fever,” Dr. Gorelik said. “Patients with Kawasaki disease should be treated as soon as the diagnosis is made, regardless of whether this patient is on day 5, day 12, or day 20 of symptoms.”
Update incorporates emerging evidence
Potential barriers to implementing the guideline in practice include the challenge of weaning doctors from practices that are habitual in medicine, Dr. Gorelik said. “One of these is the use of high-dose aspirin for Kawasaki disease; a number of studies have shown over the past decade or more that high-dose aspirin has no greater effect than lower-dose aspirin for Kawasaki disease. Despite all of these studies, the use of high-dose aspirin continued. High-dose aspirin for Kawasaki disease was used in the era prior to use of IVIg as an anti-inflammatory agent. However, it has poor efficacy in this regard, and the true benefit for aspirin is for anticoagulation for patients at risk of a clot, and this is just as effective in lower doses. Expressing this in a guideline could help to change practices by helping physicians understand not only what they are guided to do, but why.”
Additional research is needed to better identify high-risk patients in non-Japanese populations, he noted. “While studies from Japan suggest that higher-risk patients can be identified based on various parameters, these have not been well replicated in non-Japanese populations. Good research that identifies which patients may be more at risk in other populations would be helpful to more precisely target high-risk therapy.”
Other research needs include a clearer understanding of the best therapies for refractory patients, Dr. Gorelik said. “One area of the most difficulty was determining whether patients with refractory disease should have repeated IVIg or a switch to glucocorticoids and biologic agents. Some of this research is underway, and some was published just as these guidelines were being drawn, and this particular area is one that is likely to change significantly. While currently we recommend a repeated dose of IVIg, it is likely that over the very near term, the use of repeated IVIg in KD will be curtailed” because of concerns such as the relatively high rate of hemolysis. Research to identify which therapy has a noninferior effect with a superior risk profile is needed; such research “will likely result in a future iteration of these guidelines specifically related to this question,” he concluded.
The KD guideline is the final companion to three additional ACR/VF vasculitis guidelines that were released in July 2021. The guideline research received no outside funding. The researchers had no financial conflicts to disclose.
All Kawasaki disease (KD) patients should be treated first with intravenous immunoglobulin, according to an updated guideline issued jointly by the American College of Rheumatology and the Vasculitis Foundation.
KD has low mortality when treated appropriately, guideline first author Mark Gorelik, MD, assistant professor of pediatrics at Columbia University, New York, and colleagues wrote.
The update is important at this time because new evidence continues to emerge in the clinical management of KD, Dr. Gorelik said in an interview.
“In addition, this guideline approaches Kawasaki disease from a perspective of acting as an adjunct to the already existing and excellent American Heart Association guidelines by adding information in areas that rheumatologists may play a role,” Dr. Gorelik said. “This is specifically regarding patients who may require additional therapy beyond standard IVIg, such as patients who may be at higher risk of morbidity from disease and patients who have refractory disease,” he explained.
The guideline, published in Arthritis & Rheumatology, includes 11 recommendations, 1 good practice statement, and 1 ungraded position statement. The good practice statement emphasizes that all patients with KD should be initially treated with IVIg.
The position statement advises that either nonglucocorticoid immunosuppressive therapy or glucocorticoids may be used for patients with acute KD whose fever persists despite repeated IVIg treatment. No clinical evidence currently supports the superiority of either nonglucocorticoid immunosuppressive therapy or glucocorticoids; therefore, the authors support the use of either based on what is appropriate in any given clinical situation. Although optimal dosage and duration of glucocorticoids have yet to be determined in a U.S. population, the authors described a typical glucocorticoid dosage as starting prednisone at 2 mg/kg per day, with a maximum of 60 mg/day, and dose tapering over 15 days.
The 11 recommendations consist of 7 strong and 4 conditional recommendations. The strong recommendations focus on prompt treatment of incomplete KD, treatment with aspirin, and obtaining an echocardiogram in patients with unexplained macrophage activation syndrome or shock. The conditional recommendations support using established therapy promptly at disease onset, then identifying cases in which additional therapy is needed.
Dr. Gorelik highlighted four clinical takeaways from the guideline. First, “patients with higher risk for complications do exist in Kawasaki disease, and that these patients can be treated more aggressively,” he said. “Specifically, patients with aneurysms seen at first ultrasound, and patients who are under 6 months, are more likely to have progressive and/or refractory disease; these patients can be treated with an adjunctive short course of corticosteroids.”
Second, “the use of high-dose aspirin for patients with Kawasaki disease does not have strong basis in evidence. While aspirin itself of some dose is necessary for patients with Kawasaki disease, use of either high- or low-dose aspirin has the same outcome for patients, and a physician may choose either of these in practice,” he said.
Third, “we continue to recommend that refractory patients with Kawasaki disease be treated with a second dose of IVIg; however, there are many scenarios in which a physician may choose either corticosteroids [either a single high dose of >10 mg/kg, or a short moderate-dose course of 2 mg/kg per day for 5-7 days] or a biologic agent such as infliximab. ... These are valid choices for therapy in patients with refractory Kawasaki disease,” he emphasized.
Fourth, “physicians should discard the idea of treating before [and conversely, not treating after] 10 days of fever,” Dr. Gorelik said. “Patients with Kawasaki disease should be treated as soon as the diagnosis is made, regardless of whether this patient is on day 5, day 12, or day 20 of symptoms.”
Update incorporates emerging evidence
Potential barriers to implementing the guideline in practice include the challenge of weaning doctors from practices that are habitual in medicine, Dr. Gorelik said. “One of these is the use of high-dose aspirin for Kawasaki disease; a number of studies have shown over the past decade or more that high-dose aspirin has no greater effect than lower-dose aspirin for Kawasaki disease. Despite all of these studies, the use of high-dose aspirin continued. High-dose aspirin for Kawasaki disease was used in the era prior to use of IVIg as an anti-inflammatory agent. However, it has poor efficacy in this regard, and the true benefit for aspirin is for anticoagulation for patients at risk of a clot, and this is just as effective in lower doses. Expressing this in a guideline could help to change practices by helping physicians understand not only what they are guided to do, but why.”
Additional research is needed to better identify high-risk patients in non-Japanese populations, he noted. “While studies from Japan suggest that higher-risk patients can be identified based on various parameters, these have not been well replicated in non-Japanese populations. Good research that identifies which patients may be more at risk in other populations would be helpful to more precisely target high-risk therapy.”
Other research needs include a clearer understanding of the best therapies for refractory patients, Dr. Gorelik said. “One area of the most difficulty was determining whether patients with refractory disease should have repeated IVIg or a switch to glucocorticoids and biologic agents. Some of this research is underway, and some was published just as these guidelines were being drawn, and this particular area is one that is likely to change significantly. While currently we recommend a repeated dose of IVIg, it is likely that over the very near term, the use of repeated IVIg in KD will be curtailed” because of concerns such as the relatively high rate of hemolysis. Research to identify which therapy has a noninferior effect with a superior risk profile is needed; such research “will likely result in a future iteration of these guidelines specifically related to this question,” he concluded.
The KD guideline is the final companion to three additional ACR/VF vasculitis guidelines that were released in July 2021. The guideline research received no outside funding. The researchers had no financial conflicts to disclose.
All Kawasaki disease (KD) patients should be treated first with intravenous immunoglobulin, according to an updated guideline issued jointly by the American College of Rheumatology and the Vasculitis Foundation.
KD has low mortality when treated appropriately, guideline first author Mark Gorelik, MD, assistant professor of pediatrics at Columbia University, New York, and colleagues wrote.
The update is important at this time because new evidence continues to emerge in the clinical management of KD, Dr. Gorelik said in an interview.
“In addition, this guideline approaches Kawasaki disease from a perspective of acting as an adjunct to the already existing and excellent American Heart Association guidelines by adding information in areas that rheumatologists may play a role,” Dr. Gorelik said. “This is specifically regarding patients who may require additional therapy beyond standard IVIg, such as patients who may be at higher risk of morbidity from disease and patients who have refractory disease,” he explained.
The guideline, published in Arthritis & Rheumatology, includes 11 recommendations, 1 good practice statement, and 1 ungraded position statement. The good practice statement emphasizes that all patients with KD should be initially treated with IVIg.
The position statement advises that either nonglucocorticoid immunosuppressive therapy or glucocorticoids may be used for patients with acute KD whose fever persists despite repeated IVIg treatment. No clinical evidence currently supports the superiority of either nonglucocorticoid immunosuppressive therapy or glucocorticoids; therefore, the authors support the use of either based on what is appropriate in any given clinical situation. Although optimal dosage and duration of glucocorticoids have yet to be determined in a U.S. population, the authors described a typical glucocorticoid dosage as starting prednisone at 2 mg/kg per day, with a maximum of 60 mg/day, and dose tapering over 15 days.
The 11 recommendations consist of 7 strong and 4 conditional recommendations. The strong recommendations focus on prompt treatment of incomplete KD, treatment with aspirin, and obtaining an echocardiogram in patients with unexplained macrophage activation syndrome or shock. The conditional recommendations support using established therapy promptly at disease onset, then identifying cases in which additional therapy is needed.
Dr. Gorelik highlighted four clinical takeaways from the guideline. First, “patients with higher risk for complications do exist in Kawasaki disease, and that these patients can be treated more aggressively,” he said. “Specifically, patients with aneurysms seen at first ultrasound, and patients who are under 6 months, are more likely to have progressive and/or refractory disease; these patients can be treated with an adjunctive short course of corticosteroids.”
Second, “the use of high-dose aspirin for patients with Kawasaki disease does not have strong basis in evidence. While aspirin itself of some dose is necessary for patients with Kawasaki disease, use of either high- or low-dose aspirin has the same outcome for patients, and a physician may choose either of these in practice,” he said.
Third, “we continue to recommend that refractory patients with Kawasaki disease be treated with a second dose of IVIg; however, there are many scenarios in which a physician may choose either corticosteroids [either a single high dose of >10 mg/kg, or a short moderate-dose course of 2 mg/kg per day for 5-7 days] or a biologic agent such as infliximab. ... These are valid choices for therapy in patients with refractory Kawasaki disease,” he emphasized.
Fourth, “physicians should discard the idea of treating before [and conversely, not treating after] 10 days of fever,” Dr. Gorelik said. “Patients with Kawasaki disease should be treated as soon as the diagnosis is made, regardless of whether this patient is on day 5, day 12, or day 20 of symptoms.”
Update incorporates emerging evidence
Potential barriers to implementing the guideline in practice include the challenge of weaning doctors from practices that are habitual in medicine, Dr. Gorelik said. “One of these is the use of high-dose aspirin for Kawasaki disease; a number of studies have shown over the past decade or more that high-dose aspirin has no greater effect than lower-dose aspirin for Kawasaki disease. Despite all of these studies, the use of high-dose aspirin continued. High-dose aspirin for Kawasaki disease was used in the era prior to use of IVIg as an anti-inflammatory agent. However, it has poor efficacy in this regard, and the true benefit for aspirin is for anticoagulation for patients at risk of a clot, and this is just as effective in lower doses. Expressing this in a guideline could help to change practices by helping physicians understand not only what they are guided to do, but why.”
Additional research is needed to better identify high-risk patients in non-Japanese populations, he noted. “While studies from Japan suggest that higher-risk patients can be identified based on various parameters, these have not been well replicated in non-Japanese populations. Good research that identifies which patients may be more at risk in other populations would be helpful to more precisely target high-risk therapy.”
Other research needs include a clearer understanding of the best therapies for refractory patients, Dr. Gorelik said. “One area of the most difficulty was determining whether patients with refractory disease should have repeated IVIg or a switch to glucocorticoids and biologic agents. Some of this research is underway, and some was published just as these guidelines were being drawn, and this particular area is one that is likely to change significantly. While currently we recommend a repeated dose of IVIg, it is likely that over the very near term, the use of repeated IVIg in KD will be curtailed” because of concerns such as the relatively high rate of hemolysis. Research to identify which therapy has a noninferior effect with a superior risk profile is needed; such research “will likely result in a future iteration of these guidelines specifically related to this question,” he concluded.
The KD guideline is the final companion to three additional ACR/VF vasculitis guidelines that were released in July 2021. The guideline research received no outside funding. The researchers had no financial conflicts to disclose.
FROM ARTHRITIS & RHEUMATOLOGY
Doctors treat osteoporosis with hormone therapy against guidelines
This type of hormone therapy (HT) can be given as estrogen or a combination of hormones including estrogen. The physicians interviewed for this piece who prescribe HT for osteoporosis suggest the benefits outweigh the downsides to its use for some of their patients. But such doctors may be a minority group, suggests Michael R. McClung, MD, founding director of the Oregon Osteoporosis Center, Portland.
According to Dr. McClung, HT is now rarely prescribed as treatment – as opposed to prevention – for osteoporosis in the absence of additional benefits such as reducing vasomotor symptoms.
Researchers’ findings on HT use in women with osteoporosis are complex. While HT is approved for menopausal prevention of osteoporosis, it is not indicated as a treatment for the disease by the Food and Drug Administration. See the prescribing information for Premarin tablets, which contain a mixture of estrogen hormones, for an example of the FDA’s indications and usage for the type of HT addressed in this article.
Women’s Health Initiative findings
The Women’s Health Initiative (WHI) hormone therapy trials showed that HT reduces the incidence of all osteoporosis-related fractures in postmenopausal women, even those at low risk of fracture, but osteoporosis-related fractures was not a study endpoint. These trials also revealed that HT was associated with increased risks of cardiovascular and cerebrovascular events, an increased risk of breast cancer, and other adverse health outcomes.
The release of the interim results of the WHI trials in 2002 led to a fair amount of fear and confusion about the use of HT after menopause. After the WHI findings were published, estrogen use dropped dramatically, but for everything, including for vasomotor symptoms and the prevention and treatment of osteoporosis.
Prior to the WHI study, it was very common for hormone therapy to be prescribed as women neared or entered menopause, said Risa Kagan MD, clinical professor of obstetrics, gynecology, and reproductive sciences, University of California, San Francisco.
“When a woman turned 50, that was one of the first things we did – was to put her on hormone therapy. All that changed with the WHI, but now we are coming full circle,” noted Dr. Kagan, who currently prescribes HT as first line treatment for osteoporosis to some women.
Hormone therapy’s complex history
HT’s ability to reduce bone loss in postmenopausal women is well-documented in many papers, including one published March 8, 2018, in Osteoporosis International, by Dr. Kagan and colleagues. This reduced bone loss has been shown to significantly reduce fractures in patients with low bone mass and osteoporosis.
While a growing number of therapies are now available to treat osteoporosis, HT was traditionally viewed as a standard method of preventing fractures in this population. It was also widely used to prevent other types of symptoms associated with the menopause, such as hot flashes, night sweats, and sleep disturbances, and multiple observational studies had demonstrated that its use appeared to reduce the incidence of cardiovascular disease (CVD) in symptomatic menopausal women who initiated HT in early menopause.
Even though the WHI studies were the largest randomized trials ever performed in postmenopausal women, they had notable limitations, according to Dr. Kagan.
“The women were older – the average age was 63 years,” she said. “And they only investigated one route and one dose of estrogen.”
Since then, many different formulations and routes of administration with more favorable safety profiles than what was used in the WHI have become available.
It’s both scientifically and clinically unsound to extrapolate the unfavorable risk-benefit profile of HT seen in the WHI trials to all women regardless of age, HT dosage or formulation, or the length of time they’re on it, she added.
Today’s use of HT in women with osteoporosis
Re-analyses and follow-up studies from the WHI trials, along with data from other studies, have suggested that the benefit-risk profiles of HT are affected by a variety of factors. These include the timing of use in relation to menopause and chronological age and the type of hormone regimen.
“Clinically, many advocate for [hormone therapy] use, especially in the newer younger postmenopausal women to prevent bone loss, but also in younger women who are diagnosed with osteoporosis and then as they get older transition to more bone specific agents,” noted Dr. Kagan.
“Some advocate preserving bone mass and preventing osteoporosis and even treating the younger newly postmenopausal women who have no contraindications with hormone therapy initially, and then gradually transitioning them to a bone specific agent as they get older and at risk for fracture.
“If a woman is already fractured and/or has very low bone density with no other obvious secondary metabolic reason, we also often advocate anabolic agents for 1-2 years then consider estrogen for maintenance – again, if [there is] no contraindication to using HT,” she added.
Thus, an individualized approach is recommended to determine a woman’s risk-benefit ratio of HT use based on the absolute risk of adverse effects, Dr. Kagan noted.
“Transdermal and low/ultra-low doses of HT, have a favorable risk profile, and are effective in preserving bone mineral density and bone quality in many women,” she said.
According to Dr. McClung, HT “is most often used for treatment in women in whom hormone therapy was begun for hot flashes and then, when osteoporosis was found later, was simply continued.
“Society guidelines are cautious about recommending hormone therapy for osteoporosis treatment since estrogen is not approved for treatment, despite the clear fracture protection benefit observed in the WHI study,” he said. “Since [women in the WHI trials] were not recruited as having osteoporosis, those results do not meet the FDA requirement for treatment approval, namely the reduction in fracture risk in patients with osteoporosis. However, knowing what we know about the salutary skeletal effects of estrogen, many of us do use them in our patients with osteoporosis – although not prescribed for that purpose.”
Additional scenarios when doctors may advise HT
“I often recommend – and I think colleagues do as well – that women with recent menopause and menopausal symptoms who also have low bone mineral density or even scores showing osteoporosis see their gynecologist to discuss HT for a few years, perhaps until age 60 if no contraindications, and if it is well tolerated,” said Ethel S. Siris, MD, professor of medicine at Columbia University Medical Center in New York.
“Once they stop it we can then give one of our other bone drugs, but it delays the need to start them since on adequate estrogen the bone density should remain stable while they take it,” added Dr. Siris, an endocrinologist and internist, and director of the Toni Stabile Osteoporosis Center in New York. “They may need a bisphosphonate or another bone drug to further protect them from bone loss and future fracture [after stopping HT].”
Victor L. Roberts, MD, founder of Endocrine Associates of Florida, Lake Mary, pointed out that women now have many options for treatment of osteoporosis.
“If a woman is in early menopause and is having other symptoms, then estrogen is warranted,” he said. “If she has osteoporosis, then it’s a bonus.”
“We have better agents that are bone specific,” for a patient who presents with osteoporosis and no other symptoms, he said.
“If a woman is intolerant of alendronate or other similar drugs, or chooses not to have an injectable, then estrogen or a SERM [selective estrogen receptor modulator] would be an option.”
Dr. Roberts added that HT would be more of a niche drug.
“It has a role and documented benefit and works,” he said. “There is good scientific data for the use of estrogen.”
Dr. Kagan is a consultant for Pfizer, Therapeutics MD, Amgen, on the Medical and Scientific Advisory Board of American Bone Health. The other experts interviewed for this piece reported no conflicts.
This type of hormone therapy (HT) can be given as estrogen or a combination of hormones including estrogen. The physicians interviewed for this piece who prescribe HT for osteoporosis suggest the benefits outweigh the downsides to its use for some of their patients. But such doctors may be a minority group, suggests Michael R. McClung, MD, founding director of the Oregon Osteoporosis Center, Portland.
According to Dr. McClung, HT is now rarely prescribed as treatment – as opposed to prevention – for osteoporosis in the absence of additional benefits such as reducing vasomotor symptoms.
Researchers’ findings on HT use in women with osteoporosis are complex. While HT is approved for menopausal prevention of osteoporosis, it is not indicated as a treatment for the disease by the Food and Drug Administration. See the prescribing information for Premarin tablets, which contain a mixture of estrogen hormones, for an example of the FDA’s indications and usage for the type of HT addressed in this article.
Women’s Health Initiative findings
The Women’s Health Initiative (WHI) hormone therapy trials showed that HT reduces the incidence of all osteoporosis-related fractures in postmenopausal women, even those at low risk of fracture, but osteoporosis-related fractures was not a study endpoint. These trials also revealed that HT was associated with increased risks of cardiovascular and cerebrovascular events, an increased risk of breast cancer, and other adverse health outcomes.
The release of the interim results of the WHI trials in 2002 led to a fair amount of fear and confusion about the use of HT after menopause. After the WHI findings were published, estrogen use dropped dramatically, but for everything, including for vasomotor symptoms and the prevention and treatment of osteoporosis.
Prior to the WHI study, it was very common for hormone therapy to be prescribed as women neared or entered menopause, said Risa Kagan MD, clinical professor of obstetrics, gynecology, and reproductive sciences, University of California, San Francisco.
“When a woman turned 50, that was one of the first things we did – was to put her on hormone therapy. All that changed with the WHI, but now we are coming full circle,” noted Dr. Kagan, who currently prescribes HT as first line treatment for osteoporosis to some women.
Hormone therapy’s complex history
HT’s ability to reduce bone loss in postmenopausal women is well-documented in many papers, including one published March 8, 2018, in Osteoporosis International, by Dr. Kagan and colleagues. This reduced bone loss has been shown to significantly reduce fractures in patients with low bone mass and osteoporosis.
While a growing number of therapies are now available to treat osteoporosis, HT was traditionally viewed as a standard method of preventing fractures in this population. It was also widely used to prevent other types of symptoms associated with the menopause, such as hot flashes, night sweats, and sleep disturbances, and multiple observational studies had demonstrated that its use appeared to reduce the incidence of cardiovascular disease (CVD) in symptomatic menopausal women who initiated HT in early menopause.
Even though the WHI studies were the largest randomized trials ever performed in postmenopausal women, they had notable limitations, according to Dr. Kagan.
“The women were older – the average age was 63 years,” she said. “And they only investigated one route and one dose of estrogen.”
Since then, many different formulations and routes of administration with more favorable safety profiles than what was used in the WHI have become available.
It’s both scientifically and clinically unsound to extrapolate the unfavorable risk-benefit profile of HT seen in the WHI trials to all women regardless of age, HT dosage or formulation, or the length of time they’re on it, she added.
Today’s use of HT in women with osteoporosis
Re-analyses and follow-up studies from the WHI trials, along with data from other studies, have suggested that the benefit-risk profiles of HT are affected by a variety of factors. These include the timing of use in relation to menopause and chronological age and the type of hormone regimen.
“Clinically, many advocate for [hormone therapy] use, especially in the newer younger postmenopausal women to prevent bone loss, but also in younger women who are diagnosed with osteoporosis and then as they get older transition to more bone specific agents,” noted Dr. Kagan.
“Some advocate preserving bone mass and preventing osteoporosis and even treating the younger newly postmenopausal women who have no contraindications with hormone therapy initially, and then gradually transitioning them to a bone specific agent as they get older and at risk for fracture.
“If a woman is already fractured and/or has very low bone density with no other obvious secondary metabolic reason, we also often advocate anabolic agents for 1-2 years then consider estrogen for maintenance – again, if [there is] no contraindication to using HT,” she added.
Thus, an individualized approach is recommended to determine a woman’s risk-benefit ratio of HT use based on the absolute risk of adverse effects, Dr. Kagan noted.
“Transdermal and low/ultra-low doses of HT, have a favorable risk profile, and are effective in preserving bone mineral density and bone quality in many women,” she said.
According to Dr. McClung, HT “is most often used for treatment in women in whom hormone therapy was begun for hot flashes and then, when osteoporosis was found later, was simply continued.
“Society guidelines are cautious about recommending hormone therapy for osteoporosis treatment since estrogen is not approved for treatment, despite the clear fracture protection benefit observed in the WHI study,” he said. “Since [women in the WHI trials] were not recruited as having osteoporosis, those results do not meet the FDA requirement for treatment approval, namely the reduction in fracture risk in patients with osteoporosis. However, knowing what we know about the salutary skeletal effects of estrogen, many of us do use them in our patients with osteoporosis – although not prescribed for that purpose.”
Additional scenarios when doctors may advise HT
“I often recommend – and I think colleagues do as well – that women with recent menopause and menopausal symptoms who also have low bone mineral density or even scores showing osteoporosis see their gynecologist to discuss HT for a few years, perhaps until age 60 if no contraindications, and if it is well tolerated,” said Ethel S. Siris, MD, professor of medicine at Columbia University Medical Center in New York.
“Once they stop it we can then give one of our other bone drugs, but it delays the need to start them since on adequate estrogen the bone density should remain stable while they take it,” added Dr. Siris, an endocrinologist and internist, and director of the Toni Stabile Osteoporosis Center in New York. “They may need a bisphosphonate or another bone drug to further protect them from bone loss and future fracture [after stopping HT].”
Victor L. Roberts, MD, founder of Endocrine Associates of Florida, Lake Mary, pointed out that women now have many options for treatment of osteoporosis.
“If a woman is in early menopause and is having other symptoms, then estrogen is warranted,” he said. “If she has osteoporosis, then it’s a bonus.”
“We have better agents that are bone specific,” for a patient who presents with osteoporosis and no other symptoms, he said.
“If a woman is intolerant of alendronate or other similar drugs, or chooses not to have an injectable, then estrogen or a SERM [selective estrogen receptor modulator] would be an option.”
Dr. Roberts added that HT would be more of a niche drug.
“It has a role and documented benefit and works,” he said. “There is good scientific data for the use of estrogen.”
Dr. Kagan is a consultant for Pfizer, Therapeutics MD, Amgen, on the Medical and Scientific Advisory Board of American Bone Health. The other experts interviewed for this piece reported no conflicts.
This type of hormone therapy (HT) can be given as estrogen or a combination of hormones including estrogen. The physicians interviewed for this piece who prescribe HT for osteoporosis suggest the benefits outweigh the downsides to its use for some of their patients. But such doctors may be a minority group, suggests Michael R. McClung, MD, founding director of the Oregon Osteoporosis Center, Portland.
According to Dr. McClung, HT is now rarely prescribed as treatment – as opposed to prevention – for osteoporosis in the absence of additional benefits such as reducing vasomotor symptoms.
Researchers’ findings on HT use in women with osteoporosis are complex. While HT is approved for menopausal prevention of osteoporosis, it is not indicated as a treatment for the disease by the Food and Drug Administration. See the prescribing information for Premarin tablets, which contain a mixture of estrogen hormones, for an example of the FDA’s indications and usage for the type of HT addressed in this article.
Women’s Health Initiative findings
The Women’s Health Initiative (WHI) hormone therapy trials showed that HT reduces the incidence of all osteoporosis-related fractures in postmenopausal women, even those at low risk of fracture, but osteoporosis-related fractures was not a study endpoint. These trials also revealed that HT was associated with increased risks of cardiovascular and cerebrovascular events, an increased risk of breast cancer, and other adverse health outcomes.
The release of the interim results of the WHI trials in 2002 led to a fair amount of fear and confusion about the use of HT after menopause. After the WHI findings were published, estrogen use dropped dramatically, but for everything, including for vasomotor symptoms and the prevention and treatment of osteoporosis.
Prior to the WHI study, it was very common for hormone therapy to be prescribed as women neared or entered menopause, said Risa Kagan MD, clinical professor of obstetrics, gynecology, and reproductive sciences, University of California, San Francisco.
“When a woman turned 50, that was one of the first things we did – was to put her on hormone therapy. All that changed with the WHI, but now we are coming full circle,” noted Dr. Kagan, who currently prescribes HT as first line treatment for osteoporosis to some women.
Hormone therapy’s complex history
HT’s ability to reduce bone loss in postmenopausal women is well-documented in many papers, including one published March 8, 2018, in Osteoporosis International, by Dr. Kagan and colleagues. This reduced bone loss has been shown to significantly reduce fractures in patients with low bone mass and osteoporosis.
While a growing number of therapies are now available to treat osteoporosis, HT was traditionally viewed as a standard method of preventing fractures in this population. It was also widely used to prevent other types of symptoms associated with the menopause, such as hot flashes, night sweats, and sleep disturbances, and multiple observational studies had demonstrated that its use appeared to reduce the incidence of cardiovascular disease (CVD) in symptomatic menopausal women who initiated HT in early menopause.
Even though the WHI studies were the largest randomized trials ever performed in postmenopausal women, they had notable limitations, according to Dr. Kagan.
“The women were older – the average age was 63 years,” she said. “And they only investigated one route and one dose of estrogen.”
Since then, many different formulations and routes of administration with more favorable safety profiles than what was used in the WHI have become available.
It’s both scientifically and clinically unsound to extrapolate the unfavorable risk-benefit profile of HT seen in the WHI trials to all women regardless of age, HT dosage or formulation, or the length of time they’re on it, she added.
Today’s use of HT in women with osteoporosis
Re-analyses and follow-up studies from the WHI trials, along with data from other studies, have suggested that the benefit-risk profiles of HT are affected by a variety of factors. These include the timing of use in relation to menopause and chronological age and the type of hormone regimen.
“Clinically, many advocate for [hormone therapy] use, especially in the newer younger postmenopausal women to prevent bone loss, but also in younger women who are diagnosed with osteoporosis and then as they get older transition to more bone specific agents,” noted Dr. Kagan.
“Some advocate preserving bone mass and preventing osteoporosis and even treating the younger newly postmenopausal women who have no contraindications with hormone therapy initially, and then gradually transitioning them to a bone specific agent as they get older and at risk for fracture.
“If a woman is already fractured and/or has very low bone density with no other obvious secondary metabolic reason, we also often advocate anabolic agents for 1-2 years then consider estrogen for maintenance – again, if [there is] no contraindication to using HT,” she added.
Thus, an individualized approach is recommended to determine a woman’s risk-benefit ratio of HT use based on the absolute risk of adverse effects, Dr. Kagan noted.
“Transdermal and low/ultra-low doses of HT, have a favorable risk profile, and are effective in preserving bone mineral density and bone quality in many women,” she said.
According to Dr. McClung, HT “is most often used for treatment in women in whom hormone therapy was begun for hot flashes and then, when osteoporosis was found later, was simply continued.
“Society guidelines are cautious about recommending hormone therapy for osteoporosis treatment since estrogen is not approved for treatment, despite the clear fracture protection benefit observed in the WHI study,” he said. “Since [women in the WHI trials] were not recruited as having osteoporosis, those results do not meet the FDA requirement for treatment approval, namely the reduction in fracture risk in patients with osteoporosis. However, knowing what we know about the salutary skeletal effects of estrogen, many of us do use them in our patients with osteoporosis – although not prescribed for that purpose.”
Additional scenarios when doctors may advise HT
“I often recommend – and I think colleagues do as well – that women with recent menopause and menopausal symptoms who also have low bone mineral density or even scores showing osteoporosis see their gynecologist to discuss HT for a few years, perhaps until age 60 if no contraindications, and if it is well tolerated,” said Ethel S. Siris, MD, professor of medicine at Columbia University Medical Center in New York.
“Once they stop it we can then give one of our other bone drugs, but it delays the need to start them since on adequate estrogen the bone density should remain stable while they take it,” added Dr. Siris, an endocrinologist and internist, and director of the Toni Stabile Osteoporosis Center in New York. “They may need a bisphosphonate or another bone drug to further protect them from bone loss and future fracture [after stopping HT].”
Victor L. Roberts, MD, founder of Endocrine Associates of Florida, Lake Mary, pointed out that women now have many options for treatment of osteoporosis.
“If a woman is in early menopause and is having other symptoms, then estrogen is warranted,” he said. “If she has osteoporosis, then it’s a bonus.”
“We have better agents that are bone specific,” for a patient who presents with osteoporosis and no other symptoms, he said.
“If a woman is intolerant of alendronate or other similar drugs, or chooses not to have an injectable, then estrogen or a SERM [selective estrogen receptor modulator] would be an option.”
Dr. Roberts added that HT would be more of a niche drug.
“It has a role and documented benefit and works,” he said. “There is good scientific data for the use of estrogen.”
Dr. Kagan is a consultant for Pfizer, Therapeutics MD, Amgen, on the Medical and Scientific Advisory Board of American Bone Health. The other experts interviewed for this piece reported no conflicts.
New ACC guidance on cardiovascular consequences of COVID-19
The American College of Cardiology has issued an expert consensus clinical guidance document for the evaluation and management of adults with key cardiovascular consequences of COVID-19.
The document makes recommendations on how to evaluate and manage COVID-associated myocarditis and long COVID and gives advice on resumption of exercise following COVID-19 infection.
The clinical guidance was published online March 16 in the Journal of the American College of Cardiology.
“The best means to diagnose and treat myocarditis and long COVID following SARS-CoV-2 infection continues to evolve,” said Ty Gluckman, MD, MHA, cochair of the expert consensus decision pathway. “This document attempts to provide key recommendations for how to evaluate and manage adults with these conditions, including guidance for safe return to play for both competitive and noncompetitive athletes.”
The authors of the guidance note that COVID-19 can be associated with various abnormalities in cardiac testing and a wide range of cardiovascular complications. For some patients, cardiac symptoms such as chest pain, shortness of breath, fatigue, and palpitations persist, lasting months after the initial illness, and evidence of myocardial injury has also been observed in both symptomatic and asymptomatic individuals, as well as after receipt of the COVID-19 mRNA vaccine.
“For clinicians treating these individuals, a growing number of questions exist related to evaluation and management of these conditions, as well as safe resumption of physical activity,” they say. This report is intended to provide practical guidance on these issues.
Myocarditis
The report states that myocarditis has been recognized as a rare but serious complication of SARS-CoV-2 infection as well as COVID-19 mRNA vaccination.
It defines myocarditis as: 1.cardiac symptoms such as chest pain, dyspnea, palpitations, or syncope; 2. elevated cardiac troponin; and 3. abnormal electrocardiographic, echocardiographic, cardiac MRI, and/or histopathologic findings on biopsy.
The document makes the following recommendations in regard to COVID-related myocarditis:
When there is increased suspicion for cardiac involvement with COVID-19, initial testing should consist of an ECG, measurement of cardiac troponin, and an echocardiogram. Cardiology consultation is recommended for those with a rising cardiac troponin and/or echocardiographic abnormalities. Cardiac MRI is recommended in hemodynamically stable patients with suspected myocarditis.
Hospitalization is recommended for patients with definite myocarditis, ideally at an advanced heart failure center. Patients with fulminant myocarditis should be managed at centers with an expertise in advanced heart failure, mechanical circulatory support, and other advanced therapies.
Patients with myocarditis and COVID-19 pneumonia (with an ongoing need for supplemental oxygen) should be treated with corticosteroids. For patients with suspected pericardial involvement, treatment with NSAIDs, colchicine, and/or prednisone is reasonable. Intravenous corticosteroids may be considered in those with suspected or confirmed COVID-19 myocarditis with hemodynamic compromise or MIS-A (multisystem inflammatory syndrome in adults). Empiric use of corticosteroids may also be considered in those with biopsy evidence of severe myocardial infiltrates or fulminant myocarditis, balanced against infection risk.
As appropriate, guideline-directed medical therapy for heart failure should be initiated and continued after discharge.
The document notes that myocarditis following COVID-19 mRNA vaccination is rare, with highest rates seen in young males after the second vaccine dose. As of May 22, 2021, the U.S. Vaccine Adverse Event Reporting System noted rates of 40.6 cases per million after the second vaccine dose among male individuals aged 12-29 years and 2.4 cases per million among male individuals aged 30 and older. Corresponding rates in female individuals were 4.2 and 1 cases per million, respectively.
But the report says that COVID-19 vaccination is associated with “a very favorable benefit-to-risk ratio” for all age and sex groups evaluated thus far.
In general, vaccine-associated myocarditis should be diagnosed, categorized, and treated in a manner analogous to myocarditis following SARS-CoV-2 infection, the guidance advises.
Long COVID
The document refers to long COVID as postacute sequelae of SARS-CoV-2 infection (PASC), and reports that this condition is experienced by up to 10%-30% of infected individuals. It is defined by a constellation of new, returning, or persistent health problems experienced by individuals 4 or more weeks after COVID-19 infection.
Although individuals with this condition may experience wide-ranging symptoms, the symptoms that draw increased attention to the cardiovascular system include tachycardia, exercise intolerance, chest pain, and shortness of breath.
Nicole Bhave, MD, cochair of the expert consensus decision pathway, says: “There appears to be a ‘downward spiral’ for long-COVID patients. Fatigue and decreased exercise capacity lead to diminished activity and bed rest, in turn leading to worsening symptoms and decreased quality of life.” She adds that “the writing committee recommends a basic cardiopulmonary evaluation performed up front to determine if further specialty care and formalized medical therapy is needed for these patients.”
The authors propose two terms to better understand potential etiologies for those with cardiovascular symptoms:
PASC-CVD, or PASC-cardiovascular disease, refers to a broad group of cardiovascular conditions (including myocarditis) that manifest at least 4 weeks after COVID-19 infection.
PASC-CVS, or PASC-cardiovascular syndrome, includes a wide range of cardiovascular symptoms without objective evidence of cardiovascular disease following standard diagnostic testing.
The document makes the following recommendations for the management of PASC-CVD and PASC-CVS.
For patients with cardiovascular symptoms and suspected PASC, the authors suggest that a reasonable initial testing approach includes basic laboratory testing, including cardiac troponin, an ECG, an echocardiogram, an ambulatory rhythm monitor, chest imaging, and/or pulmonary function tests.
Cardiology consultation is recommended for patients with PASC who have abnormal cardiac test results, known cardiovascular disease with new or worsening symptoms, documented cardiac complications during SARS-CoV-2 infection, and/or persistent cardiopulmonary symptoms that are not otherwise explained.
Recumbent or semirecumbent exercise (for example, rowing, swimming, or cycling) is recommended initially for PASC-CVS patients with tachycardia, exercise/orthostatic intolerance, and/or deconditioning, with transition to upright exercise as orthostatic intolerance improves. Exercise duration should also be short (5-10 minutes/day) initially, with gradual increases as functional capacity improves.
Salt and fluid loading represent nonpharmacologic interventions that may provide symptomatic relief for patients with tachycardia, palpitations, and/or orthostatic hypotension.
Beta-blockers, nondihydropyridine calcium-channel blockers, ivabradine, fludrocortisone, and midodrine may be used empirically as well.
Return to play for athletes
The authors note that concerns about possible cardiac injury after COVID-19 fueled early apprehension regarding the safety of competitive sports for athletes recovering from the infection.
But they say that subsequent data from large registries have demonstrated an overall low prevalence of clinical myocarditis, without a rise in the rate of adverse cardiac events. Based on this, updated guidance is provided with a practical, evidence-based framework to guide resumption of athletics and intense exercise training.
They make the following recommendations:
- For athletes recovering from COVID-19 with ongoing cardiopulmonary symptoms (chest pain, shortness of breath, palpitations, lightheadedness) or those requiring hospitalization with increased suspicion for cardiac involvement, further evaluation with triad testing – an ECG, measurement of cardiac troponin, and an echocardiogram – should be performed.
- For those with abnormal test results, further evaluation with cardiac MRI should be considered. Individuals diagnosed with clinical myocarditis should abstain from exercise for 3-6 months.
- Cardiac testing is not recommended for asymptomatic individuals following COVID-19 infection. Individuals should abstain from training for 3 days to ensure that symptoms do not develop.
- For those with mild or moderate noncardiopulmonary symptoms (fever, lethargy, muscle aches), training may resume after symptom resolution.
- For those with remote infection (≥3 months) without ongoing cardiopulmonary symptoms, a gradual increase in exercise is recommended without the need for cardiac testing.
Based on the low prevalence of myocarditis observed in competitive athletes with COVID-19, the authors note that these recommendations can be reasonably applied to high-school athletes (aged 14 and older) along with adult recreational exercise enthusiasts.
Future study is needed, however, to better understand how long cardiac abnormalities persist following COVID-19 infection and the role of exercise training in long COVID.
The authors conclude that the current guidance is intended to help clinicians understand not only when testing may be warranted, but also when it is not.
“Given that it reflects the current state of knowledge through early 2022, it is anticipated that recommendations will change over time as our understanding evolves,” they say.
The 2022 ACC Expert Consensus Decision Pathway on Cardiovascular Sequelae of COVID-19: Myocarditis, Post-Acute Sequelae of SARS-CoV-2 Infection (PASC), and Return to Play will be discussed in a session at the American College of Cardiology’s annual scientific session meeting in Washington in April.
A version of this article first appeared on Medscape.com.
The American College of Cardiology has issued an expert consensus clinical guidance document for the evaluation and management of adults with key cardiovascular consequences of COVID-19.
The document makes recommendations on how to evaluate and manage COVID-associated myocarditis and long COVID and gives advice on resumption of exercise following COVID-19 infection.
The clinical guidance was published online March 16 in the Journal of the American College of Cardiology.
“The best means to diagnose and treat myocarditis and long COVID following SARS-CoV-2 infection continues to evolve,” said Ty Gluckman, MD, MHA, cochair of the expert consensus decision pathway. “This document attempts to provide key recommendations for how to evaluate and manage adults with these conditions, including guidance for safe return to play for both competitive and noncompetitive athletes.”
The authors of the guidance note that COVID-19 can be associated with various abnormalities in cardiac testing and a wide range of cardiovascular complications. For some patients, cardiac symptoms such as chest pain, shortness of breath, fatigue, and palpitations persist, lasting months after the initial illness, and evidence of myocardial injury has also been observed in both symptomatic and asymptomatic individuals, as well as after receipt of the COVID-19 mRNA vaccine.
“For clinicians treating these individuals, a growing number of questions exist related to evaluation and management of these conditions, as well as safe resumption of physical activity,” they say. This report is intended to provide practical guidance on these issues.
Myocarditis
The report states that myocarditis has been recognized as a rare but serious complication of SARS-CoV-2 infection as well as COVID-19 mRNA vaccination.
It defines myocarditis as: 1.cardiac symptoms such as chest pain, dyspnea, palpitations, or syncope; 2. elevated cardiac troponin; and 3. abnormal electrocardiographic, echocardiographic, cardiac MRI, and/or histopathologic findings on biopsy.
The document makes the following recommendations in regard to COVID-related myocarditis:
When there is increased suspicion for cardiac involvement with COVID-19, initial testing should consist of an ECG, measurement of cardiac troponin, and an echocardiogram. Cardiology consultation is recommended for those with a rising cardiac troponin and/or echocardiographic abnormalities. Cardiac MRI is recommended in hemodynamically stable patients with suspected myocarditis.
Hospitalization is recommended for patients with definite myocarditis, ideally at an advanced heart failure center. Patients with fulminant myocarditis should be managed at centers with an expertise in advanced heart failure, mechanical circulatory support, and other advanced therapies.
Patients with myocarditis and COVID-19 pneumonia (with an ongoing need for supplemental oxygen) should be treated with corticosteroids. For patients with suspected pericardial involvement, treatment with NSAIDs, colchicine, and/or prednisone is reasonable. Intravenous corticosteroids may be considered in those with suspected or confirmed COVID-19 myocarditis with hemodynamic compromise or MIS-A (multisystem inflammatory syndrome in adults). Empiric use of corticosteroids may also be considered in those with biopsy evidence of severe myocardial infiltrates or fulminant myocarditis, balanced against infection risk.
As appropriate, guideline-directed medical therapy for heart failure should be initiated and continued after discharge.
The document notes that myocarditis following COVID-19 mRNA vaccination is rare, with highest rates seen in young males after the second vaccine dose. As of May 22, 2021, the U.S. Vaccine Adverse Event Reporting System noted rates of 40.6 cases per million after the second vaccine dose among male individuals aged 12-29 years and 2.4 cases per million among male individuals aged 30 and older. Corresponding rates in female individuals were 4.2 and 1 cases per million, respectively.
But the report says that COVID-19 vaccination is associated with “a very favorable benefit-to-risk ratio” for all age and sex groups evaluated thus far.
In general, vaccine-associated myocarditis should be diagnosed, categorized, and treated in a manner analogous to myocarditis following SARS-CoV-2 infection, the guidance advises.
Long COVID
The document refers to long COVID as postacute sequelae of SARS-CoV-2 infection (PASC), and reports that this condition is experienced by up to 10%-30% of infected individuals. It is defined by a constellation of new, returning, or persistent health problems experienced by individuals 4 or more weeks after COVID-19 infection.
Although individuals with this condition may experience wide-ranging symptoms, the symptoms that draw increased attention to the cardiovascular system include tachycardia, exercise intolerance, chest pain, and shortness of breath.
Nicole Bhave, MD, cochair of the expert consensus decision pathway, says: “There appears to be a ‘downward spiral’ for long-COVID patients. Fatigue and decreased exercise capacity lead to diminished activity and bed rest, in turn leading to worsening symptoms and decreased quality of life.” She adds that “the writing committee recommends a basic cardiopulmonary evaluation performed up front to determine if further specialty care and formalized medical therapy is needed for these patients.”
The authors propose two terms to better understand potential etiologies for those with cardiovascular symptoms:
PASC-CVD, or PASC-cardiovascular disease, refers to a broad group of cardiovascular conditions (including myocarditis) that manifest at least 4 weeks after COVID-19 infection.
PASC-CVS, or PASC-cardiovascular syndrome, includes a wide range of cardiovascular symptoms without objective evidence of cardiovascular disease following standard diagnostic testing.
The document makes the following recommendations for the management of PASC-CVD and PASC-CVS.
For patients with cardiovascular symptoms and suspected PASC, the authors suggest that a reasonable initial testing approach includes basic laboratory testing, including cardiac troponin, an ECG, an echocardiogram, an ambulatory rhythm monitor, chest imaging, and/or pulmonary function tests.
Cardiology consultation is recommended for patients with PASC who have abnormal cardiac test results, known cardiovascular disease with new or worsening symptoms, documented cardiac complications during SARS-CoV-2 infection, and/or persistent cardiopulmonary symptoms that are not otherwise explained.
Recumbent or semirecumbent exercise (for example, rowing, swimming, or cycling) is recommended initially for PASC-CVS patients with tachycardia, exercise/orthostatic intolerance, and/or deconditioning, with transition to upright exercise as orthostatic intolerance improves. Exercise duration should also be short (5-10 minutes/day) initially, with gradual increases as functional capacity improves.
Salt and fluid loading represent nonpharmacologic interventions that may provide symptomatic relief for patients with tachycardia, palpitations, and/or orthostatic hypotension.
Beta-blockers, nondihydropyridine calcium-channel blockers, ivabradine, fludrocortisone, and midodrine may be used empirically as well.
Return to play for athletes
The authors note that concerns about possible cardiac injury after COVID-19 fueled early apprehension regarding the safety of competitive sports for athletes recovering from the infection.
But they say that subsequent data from large registries have demonstrated an overall low prevalence of clinical myocarditis, without a rise in the rate of adverse cardiac events. Based on this, updated guidance is provided with a practical, evidence-based framework to guide resumption of athletics and intense exercise training.
They make the following recommendations:
- For athletes recovering from COVID-19 with ongoing cardiopulmonary symptoms (chest pain, shortness of breath, palpitations, lightheadedness) or those requiring hospitalization with increased suspicion for cardiac involvement, further evaluation with triad testing – an ECG, measurement of cardiac troponin, and an echocardiogram – should be performed.
- For those with abnormal test results, further evaluation with cardiac MRI should be considered. Individuals diagnosed with clinical myocarditis should abstain from exercise for 3-6 months.
- Cardiac testing is not recommended for asymptomatic individuals following COVID-19 infection. Individuals should abstain from training for 3 days to ensure that symptoms do not develop.
- For those with mild or moderate noncardiopulmonary symptoms (fever, lethargy, muscle aches), training may resume after symptom resolution.
- For those with remote infection (≥3 months) without ongoing cardiopulmonary symptoms, a gradual increase in exercise is recommended without the need for cardiac testing.
Based on the low prevalence of myocarditis observed in competitive athletes with COVID-19, the authors note that these recommendations can be reasonably applied to high-school athletes (aged 14 and older) along with adult recreational exercise enthusiasts.
Future study is needed, however, to better understand how long cardiac abnormalities persist following COVID-19 infection and the role of exercise training in long COVID.
The authors conclude that the current guidance is intended to help clinicians understand not only when testing may be warranted, but also when it is not.
“Given that it reflects the current state of knowledge through early 2022, it is anticipated that recommendations will change over time as our understanding evolves,” they say.
The 2022 ACC Expert Consensus Decision Pathway on Cardiovascular Sequelae of COVID-19: Myocarditis, Post-Acute Sequelae of SARS-CoV-2 Infection (PASC), and Return to Play will be discussed in a session at the American College of Cardiology’s annual scientific session meeting in Washington in April.
A version of this article first appeared on Medscape.com.
The American College of Cardiology has issued an expert consensus clinical guidance document for the evaluation and management of adults with key cardiovascular consequences of COVID-19.
The document makes recommendations on how to evaluate and manage COVID-associated myocarditis and long COVID and gives advice on resumption of exercise following COVID-19 infection.
The clinical guidance was published online March 16 in the Journal of the American College of Cardiology.
“The best means to diagnose and treat myocarditis and long COVID following SARS-CoV-2 infection continues to evolve,” said Ty Gluckman, MD, MHA, cochair of the expert consensus decision pathway. “This document attempts to provide key recommendations for how to evaluate and manage adults with these conditions, including guidance for safe return to play for both competitive and noncompetitive athletes.”
The authors of the guidance note that COVID-19 can be associated with various abnormalities in cardiac testing and a wide range of cardiovascular complications. For some patients, cardiac symptoms such as chest pain, shortness of breath, fatigue, and palpitations persist, lasting months after the initial illness, and evidence of myocardial injury has also been observed in both symptomatic and asymptomatic individuals, as well as after receipt of the COVID-19 mRNA vaccine.
“For clinicians treating these individuals, a growing number of questions exist related to evaluation and management of these conditions, as well as safe resumption of physical activity,” they say. This report is intended to provide practical guidance on these issues.
Myocarditis
The report states that myocarditis has been recognized as a rare but serious complication of SARS-CoV-2 infection as well as COVID-19 mRNA vaccination.
It defines myocarditis as: 1.cardiac symptoms such as chest pain, dyspnea, palpitations, or syncope; 2. elevated cardiac troponin; and 3. abnormal electrocardiographic, echocardiographic, cardiac MRI, and/or histopathologic findings on biopsy.
The document makes the following recommendations in regard to COVID-related myocarditis:
When there is increased suspicion for cardiac involvement with COVID-19, initial testing should consist of an ECG, measurement of cardiac troponin, and an echocardiogram. Cardiology consultation is recommended for those with a rising cardiac troponin and/or echocardiographic abnormalities. Cardiac MRI is recommended in hemodynamically stable patients with suspected myocarditis.
Hospitalization is recommended for patients with definite myocarditis, ideally at an advanced heart failure center. Patients with fulminant myocarditis should be managed at centers with an expertise in advanced heart failure, mechanical circulatory support, and other advanced therapies.
Patients with myocarditis and COVID-19 pneumonia (with an ongoing need for supplemental oxygen) should be treated with corticosteroids. For patients with suspected pericardial involvement, treatment with NSAIDs, colchicine, and/or prednisone is reasonable. Intravenous corticosteroids may be considered in those with suspected or confirmed COVID-19 myocarditis with hemodynamic compromise or MIS-A (multisystem inflammatory syndrome in adults). Empiric use of corticosteroids may also be considered in those with biopsy evidence of severe myocardial infiltrates or fulminant myocarditis, balanced against infection risk.
As appropriate, guideline-directed medical therapy for heart failure should be initiated and continued after discharge.
The document notes that myocarditis following COVID-19 mRNA vaccination is rare, with highest rates seen in young males after the second vaccine dose. As of May 22, 2021, the U.S. Vaccine Adverse Event Reporting System noted rates of 40.6 cases per million after the second vaccine dose among male individuals aged 12-29 years and 2.4 cases per million among male individuals aged 30 and older. Corresponding rates in female individuals were 4.2 and 1 cases per million, respectively.
But the report says that COVID-19 vaccination is associated with “a very favorable benefit-to-risk ratio” for all age and sex groups evaluated thus far.
In general, vaccine-associated myocarditis should be diagnosed, categorized, and treated in a manner analogous to myocarditis following SARS-CoV-2 infection, the guidance advises.
Long COVID
The document refers to long COVID as postacute sequelae of SARS-CoV-2 infection (PASC), and reports that this condition is experienced by up to 10%-30% of infected individuals. It is defined by a constellation of new, returning, or persistent health problems experienced by individuals 4 or more weeks after COVID-19 infection.
Although individuals with this condition may experience wide-ranging symptoms, the symptoms that draw increased attention to the cardiovascular system include tachycardia, exercise intolerance, chest pain, and shortness of breath.
Nicole Bhave, MD, cochair of the expert consensus decision pathway, says: “There appears to be a ‘downward spiral’ for long-COVID patients. Fatigue and decreased exercise capacity lead to diminished activity and bed rest, in turn leading to worsening symptoms and decreased quality of life.” She adds that “the writing committee recommends a basic cardiopulmonary evaluation performed up front to determine if further specialty care and formalized medical therapy is needed for these patients.”
The authors propose two terms to better understand potential etiologies for those with cardiovascular symptoms:
PASC-CVD, or PASC-cardiovascular disease, refers to a broad group of cardiovascular conditions (including myocarditis) that manifest at least 4 weeks after COVID-19 infection.
PASC-CVS, or PASC-cardiovascular syndrome, includes a wide range of cardiovascular symptoms without objective evidence of cardiovascular disease following standard diagnostic testing.
The document makes the following recommendations for the management of PASC-CVD and PASC-CVS.
For patients with cardiovascular symptoms and suspected PASC, the authors suggest that a reasonable initial testing approach includes basic laboratory testing, including cardiac troponin, an ECG, an echocardiogram, an ambulatory rhythm monitor, chest imaging, and/or pulmonary function tests.
Cardiology consultation is recommended for patients with PASC who have abnormal cardiac test results, known cardiovascular disease with new or worsening symptoms, documented cardiac complications during SARS-CoV-2 infection, and/or persistent cardiopulmonary symptoms that are not otherwise explained.
Recumbent or semirecumbent exercise (for example, rowing, swimming, or cycling) is recommended initially for PASC-CVS patients with tachycardia, exercise/orthostatic intolerance, and/or deconditioning, with transition to upright exercise as orthostatic intolerance improves. Exercise duration should also be short (5-10 minutes/day) initially, with gradual increases as functional capacity improves.
Salt and fluid loading represent nonpharmacologic interventions that may provide symptomatic relief for patients with tachycardia, palpitations, and/or orthostatic hypotension.
Beta-blockers, nondihydropyridine calcium-channel blockers, ivabradine, fludrocortisone, and midodrine may be used empirically as well.
Return to play for athletes
The authors note that concerns about possible cardiac injury after COVID-19 fueled early apprehension regarding the safety of competitive sports for athletes recovering from the infection.
But they say that subsequent data from large registries have demonstrated an overall low prevalence of clinical myocarditis, without a rise in the rate of adverse cardiac events. Based on this, updated guidance is provided with a practical, evidence-based framework to guide resumption of athletics and intense exercise training.
They make the following recommendations:
- For athletes recovering from COVID-19 with ongoing cardiopulmonary symptoms (chest pain, shortness of breath, palpitations, lightheadedness) or those requiring hospitalization with increased suspicion for cardiac involvement, further evaluation with triad testing – an ECG, measurement of cardiac troponin, and an echocardiogram – should be performed.
- For those with abnormal test results, further evaluation with cardiac MRI should be considered. Individuals diagnosed with clinical myocarditis should abstain from exercise for 3-6 months.
- Cardiac testing is not recommended for asymptomatic individuals following COVID-19 infection. Individuals should abstain from training for 3 days to ensure that symptoms do not develop.
- For those with mild or moderate noncardiopulmonary symptoms (fever, lethargy, muscle aches), training may resume after symptom resolution.
- For those with remote infection (≥3 months) without ongoing cardiopulmonary symptoms, a gradual increase in exercise is recommended without the need for cardiac testing.
Based on the low prevalence of myocarditis observed in competitive athletes with COVID-19, the authors note that these recommendations can be reasonably applied to high-school athletes (aged 14 and older) along with adult recreational exercise enthusiasts.
Future study is needed, however, to better understand how long cardiac abnormalities persist following COVID-19 infection and the role of exercise training in long COVID.
The authors conclude that the current guidance is intended to help clinicians understand not only when testing may be warranted, but also when it is not.
“Given that it reflects the current state of knowledge through early 2022, it is anticipated that recommendations will change over time as our understanding evolves,” they say.
The 2022 ACC Expert Consensus Decision Pathway on Cardiovascular Sequelae of COVID-19: Myocarditis, Post-Acute Sequelae of SARS-CoV-2 Infection (PASC), and Return to Play will be discussed in a session at the American College of Cardiology’s annual scientific session meeting in Washington in April.
A version of this article first appeared on Medscape.com.
High-intensity exercise vs. omega-3s for heart failure risk reduction
A year of high-intensity interval training seemed to benefit obese middle-aged adults at a high risk of heart failure, but omega-3 fatty acid supplementation didn’t have any effect on cardiac biomarkers measured in a small, single-center, prospective study.
“One year of HIIT training reduces adiposity but had no consistent effect on myocardial triglyceride content or visceral adiposity,” wrote lead author Christopher M. Hearon Jr., PhD, and colleagues in JACC: Heart Failure. “However, long-duration HIIT improves fitness and induces favorable cardiac remodeling.” Omega-3 supplementation, however, had “no independent or additive effect.” Dr. Hearon is an instructor of applied clinical research at University of Texas Southwestern Medical Center in Dallas.
Investigators there and at the Institute for Exercise and Environmental Medicine at Texas Health Presbyterian Hospital Dallas studied 80 patients aged 40-55 years classified as high risk for HF and obese, randomizing them to a year of high-intensity interval training (HIIT) with supplementation of either 1.6 g omega-3 FA or placebo daily; or to a control group split between supplementation or placebo. Fifty-six patients completed the 1-year study, with a compliance rate of 90% in the HIIT group and 92% in those assigned omega-3 FA supplementation.
Carl J. “Chip” Lavie, MD, of the John Ochsner Heart and Vascular Institute in New Orleans, commented that, although the study was “extremely well done from an excellent research group,” it was limited by its small population and relatively short follow-up. Future research should evaluate HIIT and moderate exercise on clinical events over a longer term as well as different doses of omega-3 “There is tremendous potential for omega-3 in heart failure prevention and treatment.”
HIIT boosts exercise capacity, more
In the study, the HIIT group showed improvement in a number of cardiac markers: around a 22% improvement in exercise capacity as measured by absolute peak and relative peak oxygen uptake (VO2), even without significant weight loss. They improved an average of 0.43 L/min (0.32-0.53; P < .0001) and 4.46 mL/kg per minute (3.18-5.56; P < .0001), respectively.
The researchers attributed the increase in peak VO2 to an increase in peak cardiac output averaging 2.15 L/min (95% confidence interval, 0.90-3.39; P = .001) and stroke volume averaging 9.46 mL (95% CI, 0.65-18.27; P = .04). A year of exercise training also resulted in changes in cardiac remodeling, including increases in left ventricle mass and LV end diastolic volume, averaging 9.4 g (95% CI, 4.36-14.44; P < .001) and 12.33 mL (95% CI, 5.61-19.05; P < .001), respectively.
The study also found that neither intervention had any appreciable impact on body weight, body mass index, body surface area or lean mass, or markers of arterial or local carotid stiffness. The exercise group had a modest decrease in fat mass, averaging 2.63 kg (95% CI,–4.81 to –0.46; P = .02), but without any effect from omega-3 supplementation.
The study acknowledged that high-dose omega-3 supplements have been found to lower triglyceride levels in people with severe hypertriglyceridemia, and hypothesized that HIIT alone or with omega-3 supplementation would improve fitness and biomarkers in people with stage A HF. “Contrary to our hypothesis, we found that one year of n-3FA [omega-3 FA] supplementation had no detectable effect on any parameter related to cardiopulmonary fitness, cardiovascular remodeling/stiffness, visceral adiposity, or myocardial triglyceride content,” Dr. Hearon and colleagues wrote.
The study “shows that obese middle-aged patients with heart failure with preserved ejection fraction [HFpEF] can markedly improve their fitness with HIIT and, generally, fitness is one of the strongest if not the strongest predictor of prognosis and survival,” said Dr. Lavie.
“Studies are needed on exercise that improves fitness in both HF with reduced ejection fraction and HFpEF, but especially HFpEF,” he said.
The study received funding from the American Heart Association Strategically Focused Research Network. Dr. Hearon and coauthors have no relevant disclosures. Dr. Lavie is a speaker and consultant for PAI Health, the Global Organization for EPA and DHA Omega-3s and DSM Nutritional Products.
A year of high-intensity interval training seemed to benefit obese middle-aged adults at a high risk of heart failure, but omega-3 fatty acid supplementation didn’t have any effect on cardiac biomarkers measured in a small, single-center, prospective study.
“One year of HIIT training reduces adiposity but had no consistent effect on myocardial triglyceride content or visceral adiposity,” wrote lead author Christopher M. Hearon Jr., PhD, and colleagues in JACC: Heart Failure. “However, long-duration HIIT improves fitness and induces favorable cardiac remodeling.” Omega-3 supplementation, however, had “no independent or additive effect.” Dr. Hearon is an instructor of applied clinical research at University of Texas Southwestern Medical Center in Dallas.
Investigators there and at the Institute for Exercise and Environmental Medicine at Texas Health Presbyterian Hospital Dallas studied 80 patients aged 40-55 years classified as high risk for HF and obese, randomizing them to a year of high-intensity interval training (HIIT) with supplementation of either 1.6 g omega-3 FA or placebo daily; or to a control group split between supplementation or placebo. Fifty-six patients completed the 1-year study, with a compliance rate of 90% in the HIIT group and 92% in those assigned omega-3 FA supplementation.
Carl J. “Chip” Lavie, MD, of the John Ochsner Heart and Vascular Institute in New Orleans, commented that, although the study was “extremely well done from an excellent research group,” it was limited by its small population and relatively short follow-up. Future research should evaluate HIIT and moderate exercise on clinical events over a longer term as well as different doses of omega-3 “There is tremendous potential for omega-3 in heart failure prevention and treatment.”
HIIT boosts exercise capacity, more
In the study, the HIIT group showed improvement in a number of cardiac markers: around a 22% improvement in exercise capacity as measured by absolute peak and relative peak oxygen uptake (VO2), even without significant weight loss. They improved an average of 0.43 L/min (0.32-0.53; P < .0001) and 4.46 mL/kg per minute (3.18-5.56; P < .0001), respectively.
The researchers attributed the increase in peak VO2 to an increase in peak cardiac output averaging 2.15 L/min (95% confidence interval, 0.90-3.39; P = .001) and stroke volume averaging 9.46 mL (95% CI, 0.65-18.27; P = .04). A year of exercise training also resulted in changes in cardiac remodeling, including increases in left ventricle mass and LV end diastolic volume, averaging 9.4 g (95% CI, 4.36-14.44; P < .001) and 12.33 mL (95% CI, 5.61-19.05; P < .001), respectively.
The study also found that neither intervention had any appreciable impact on body weight, body mass index, body surface area or lean mass, or markers of arterial or local carotid stiffness. The exercise group had a modest decrease in fat mass, averaging 2.63 kg (95% CI,–4.81 to –0.46; P = .02), but without any effect from omega-3 supplementation.
The study acknowledged that high-dose omega-3 supplements have been found to lower triglyceride levels in people with severe hypertriglyceridemia, and hypothesized that HIIT alone or with omega-3 supplementation would improve fitness and biomarkers in people with stage A HF. “Contrary to our hypothesis, we found that one year of n-3FA [omega-3 FA] supplementation had no detectable effect on any parameter related to cardiopulmonary fitness, cardiovascular remodeling/stiffness, visceral adiposity, or myocardial triglyceride content,” Dr. Hearon and colleagues wrote.
The study “shows that obese middle-aged patients with heart failure with preserved ejection fraction [HFpEF] can markedly improve their fitness with HIIT and, generally, fitness is one of the strongest if not the strongest predictor of prognosis and survival,” said Dr. Lavie.
“Studies are needed on exercise that improves fitness in both HF with reduced ejection fraction and HFpEF, but especially HFpEF,” he said.
The study received funding from the American Heart Association Strategically Focused Research Network. Dr. Hearon and coauthors have no relevant disclosures. Dr. Lavie is a speaker and consultant for PAI Health, the Global Organization for EPA and DHA Omega-3s and DSM Nutritional Products.
A year of high-intensity interval training seemed to benefit obese middle-aged adults at a high risk of heart failure, but omega-3 fatty acid supplementation didn’t have any effect on cardiac biomarkers measured in a small, single-center, prospective study.
“One year of HIIT training reduces adiposity but had no consistent effect on myocardial triglyceride content or visceral adiposity,” wrote lead author Christopher M. Hearon Jr., PhD, and colleagues in JACC: Heart Failure. “However, long-duration HIIT improves fitness and induces favorable cardiac remodeling.” Omega-3 supplementation, however, had “no independent or additive effect.” Dr. Hearon is an instructor of applied clinical research at University of Texas Southwestern Medical Center in Dallas.
Investigators there and at the Institute for Exercise and Environmental Medicine at Texas Health Presbyterian Hospital Dallas studied 80 patients aged 40-55 years classified as high risk for HF and obese, randomizing them to a year of high-intensity interval training (HIIT) with supplementation of either 1.6 g omega-3 FA or placebo daily; or to a control group split between supplementation or placebo. Fifty-six patients completed the 1-year study, with a compliance rate of 90% in the HIIT group and 92% in those assigned omega-3 FA supplementation.
Carl J. “Chip” Lavie, MD, of the John Ochsner Heart and Vascular Institute in New Orleans, commented that, although the study was “extremely well done from an excellent research group,” it was limited by its small population and relatively short follow-up. Future research should evaluate HIIT and moderate exercise on clinical events over a longer term as well as different doses of omega-3 “There is tremendous potential for omega-3 in heart failure prevention and treatment.”
HIIT boosts exercise capacity, more
In the study, the HIIT group showed improvement in a number of cardiac markers: around a 22% improvement in exercise capacity as measured by absolute peak and relative peak oxygen uptake (VO2), even without significant weight loss. They improved an average of 0.43 L/min (0.32-0.53; P < .0001) and 4.46 mL/kg per minute (3.18-5.56; P < .0001), respectively.
The researchers attributed the increase in peak VO2 to an increase in peak cardiac output averaging 2.15 L/min (95% confidence interval, 0.90-3.39; P = .001) and stroke volume averaging 9.46 mL (95% CI, 0.65-18.27; P = .04). A year of exercise training also resulted in changes in cardiac remodeling, including increases in left ventricle mass and LV end diastolic volume, averaging 9.4 g (95% CI, 4.36-14.44; P < .001) and 12.33 mL (95% CI, 5.61-19.05; P < .001), respectively.
The study also found that neither intervention had any appreciable impact on body weight, body mass index, body surface area or lean mass, or markers of arterial or local carotid stiffness. The exercise group had a modest decrease in fat mass, averaging 2.63 kg (95% CI,–4.81 to –0.46; P = .02), but without any effect from omega-3 supplementation.
The study acknowledged that high-dose omega-3 supplements have been found to lower triglyceride levels in people with severe hypertriglyceridemia, and hypothesized that HIIT alone or with omega-3 supplementation would improve fitness and biomarkers in people with stage A HF. “Contrary to our hypothesis, we found that one year of n-3FA [omega-3 FA] supplementation had no detectable effect on any parameter related to cardiopulmonary fitness, cardiovascular remodeling/stiffness, visceral adiposity, or myocardial triglyceride content,” Dr. Hearon and colleagues wrote.
The study “shows that obese middle-aged patients with heart failure with preserved ejection fraction [HFpEF] can markedly improve their fitness with HIIT and, generally, fitness is one of the strongest if not the strongest predictor of prognosis and survival,” said Dr. Lavie.
“Studies are needed on exercise that improves fitness in both HF with reduced ejection fraction and HFpEF, but especially HFpEF,” he said.
The study received funding from the American Heart Association Strategically Focused Research Network. Dr. Hearon and coauthors have no relevant disclosures. Dr. Lavie is a speaker and consultant for PAI Health, the Global Organization for EPA and DHA Omega-3s and DSM Nutritional Products.
FROM JACC: HEART FAILURE
Cardiologists say rights to maternity leave violated
A survey of 323 women cardiologists who were working while they were pregnant showed that nearly 75% experienced discriminatory maternity-leave practices, some of which were likely violations of the Family and Medical Leave Act (FMLA).
More than 40% saw their salaries decreased during their year of pregnancy, 38% were required to perform extra service or call before taking maternity leave, exposing them to occupational hazards such as radiation, and 40% experienced a pregnancy complication, significantly higher than the general population and other medical specialties.
Additionally, of those who performed extra service or call, 18% were placed on bedrest before delivery, compared with 7.4% who did not perform extra service or call.
More than half of respondents reported that pregnancy negatively impacted their careers, and 42.4% said they experienced pressure to return to work and a delay in promotions, both illegal practices under the FMLA.
The survey is published in the Journal of the American College of Cardiology.
“Childbearing is difficult for women in cardiology with more than double the rate of gestational complications of the U.S. population, frequent income loss out of proportion to reduced productivity, and for nearly half, has an adverse impact on their career,” lead author Martha Gulati, MD, University of Arizona, Phoenix, said in a statement.
“While many professions struggle to create environments supportive of pregnancy and child-rearing, the prevalence of illegal behavior in cardiology is quite high and presents substantial legal risk for employers,” Dr. Gulati added.
C. Noel Bairey Merz, MD, professor of cardiology at Cedars-Sinai Smidt Heart Institute, Los Angeles, and a coauthor of the survey, told this news organization that it’s not surprising that such a situation exists, even “in this day and age.”
“I’m not surprised as a woman in cardiology myself. I was told by my training director that if I took off more than my allowed sick leave when I had my first and second children, I would have to repeat the year of training, so not surprised at all. I hear this from colleagues all the time,” Dr. Bairey Merz said.
The exchange left her feeling fearful for her career.
“Who wants to repeat a year? It pushes you back from a career standpoint, financially, everything. It also made me angry. I had a colleague who busted his leg in a motorcycle accident. He was unable to do any procedures for 16 weeks, and he didn’t have to repeat the year,” she pointed out.
The challenge that pregnancy represents is frequently cited by women as a deterrent for applying for a cardiology fellowship, Laxmi S. Mehta, MD, Ohio State University, Columbus, and colleagues wrote in an accompanying editorial.
The findings from the survey “reveal restrictive maternity leave data in a profession that has historically and currently continues to have a diversity problem,” they wrote.
“Maternity and pregnancy issues are a thing in cardiology,” Dr. Mehta said in an interview. “It’s one of the reasons why women get deterred from going into the field. It makes it challenging to choose cardiology if you perceive that the culture is negative, that it’s hard to be pregnant, or to bear children, or to take care of them post partum. It is problematic and it should not be occurring now.”
Leadership that condones such restrictive policies or even promotes them through ignorance and inaction needs to be held accountable, she added.
“We need to move forward from this negativity and make it more warm and welcoming to have families, whether you are a trainee or a practicing cardiologist, male or female. We need transparent and consistent parental leave policies and things like lactation support when a woman returns to work. That is a big issue,” Dr. Mehta said.
Having cardiovascular leaders champion the cause of adequate maternity and paternity leave are crucial to creating a newer, inclusive environment in cardiology.
As an example, Dr. Mehta recounted her own experience when she was in training 17 years ago.
“When I interviewed for a cardiology fellowship, one of the female program directors asked me if I was planning to have children, because if I did, the other fellows wouldn’t like it if they had to cover for me,” she said. “I ended up doing my fellowship where the chief of cardiology encouraged me to have children. He said: ‘Have your children during training, we will support you.’ And he did. I still had to do all of the call make-up and that stuff, but I worked in a supportive environment, and it made all the difference.”
“It’s about allyship,” she added. “You will have some people who are supportive and some who are not, but when you have the chief supporting you, you have a strong ally.”
The researchers suggest that one strategy is to temporarily replace cardiologists on maternity leave with locums, or “deepen the bench of coverage for clinical work, as is done for other absences. Given the expanding coverage of parental and family medical leaves, and awareness of these issues nationally, the need for this is likely to become less of an exception and more the rule.”
For example, nine states and Washington, D.C. now provide paid parental leave, they wrote, “and there is pending legislation in others.”
Dr. Bairey Merz and Dr. Mehta reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A survey of 323 women cardiologists who were working while they were pregnant showed that nearly 75% experienced discriminatory maternity-leave practices, some of which were likely violations of the Family and Medical Leave Act (FMLA).
More than 40% saw their salaries decreased during their year of pregnancy, 38% were required to perform extra service or call before taking maternity leave, exposing them to occupational hazards such as radiation, and 40% experienced a pregnancy complication, significantly higher than the general population and other medical specialties.
Additionally, of those who performed extra service or call, 18% were placed on bedrest before delivery, compared with 7.4% who did not perform extra service or call.
More than half of respondents reported that pregnancy negatively impacted their careers, and 42.4% said they experienced pressure to return to work and a delay in promotions, both illegal practices under the FMLA.
The survey is published in the Journal of the American College of Cardiology.
“Childbearing is difficult for women in cardiology with more than double the rate of gestational complications of the U.S. population, frequent income loss out of proportion to reduced productivity, and for nearly half, has an adverse impact on their career,” lead author Martha Gulati, MD, University of Arizona, Phoenix, said in a statement.
“While many professions struggle to create environments supportive of pregnancy and child-rearing, the prevalence of illegal behavior in cardiology is quite high and presents substantial legal risk for employers,” Dr. Gulati added.
C. Noel Bairey Merz, MD, professor of cardiology at Cedars-Sinai Smidt Heart Institute, Los Angeles, and a coauthor of the survey, told this news organization that it’s not surprising that such a situation exists, even “in this day and age.”
“I’m not surprised as a woman in cardiology myself. I was told by my training director that if I took off more than my allowed sick leave when I had my first and second children, I would have to repeat the year of training, so not surprised at all. I hear this from colleagues all the time,” Dr. Bairey Merz said.
The exchange left her feeling fearful for her career.
“Who wants to repeat a year? It pushes you back from a career standpoint, financially, everything. It also made me angry. I had a colleague who busted his leg in a motorcycle accident. He was unable to do any procedures for 16 weeks, and he didn’t have to repeat the year,” she pointed out.
The challenge that pregnancy represents is frequently cited by women as a deterrent for applying for a cardiology fellowship, Laxmi S. Mehta, MD, Ohio State University, Columbus, and colleagues wrote in an accompanying editorial.
The findings from the survey “reveal restrictive maternity leave data in a profession that has historically and currently continues to have a diversity problem,” they wrote.
“Maternity and pregnancy issues are a thing in cardiology,” Dr. Mehta said in an interview. “It’s one of the reasons why women get deterred from going into the field. It makes it challenging to choose cardiology if you perceive that the culture is negative, that it’s hard to be pregnant, or to bear children, or to take care of them post partum. It is problematic and it should not be occurring now.”
Leadership that condones such restrictive policies or even promotes them through ignorance and inaction needs to be held accountable, she added.
“We need to move forward from this negativity and make it more warm and welcoming to have families, whether you are a trainee or a practicing cardiologist, male or female. We need transparent and consistent parental leave policies and things like lactation support when a woman returns to work. That is a big issue,” Dr. Mehta said.
Having cardiovascular leaders champion the cause of adequate maternity and paternity leave are crucial to creating a newer, inclusive environment in cardiology.
As an example, Dr. Mehta recounted her own experience when she was in training 17 years ago.
“When I interviewed for a cardiology fellowship, one of the female program directors asked me if I was planning to have children, because if I did, the other fellows wouldn’t like it if they had to cover for me,” she said. “I ended up doing my fellowship where the chief of cardiology encouraged me to have children. He said: ‘Have your children during training, we will support you.’ And he did. I still had to do all of the call make-up and that stuff, but I worked in a supportive environment, and it made all the difference.”
“It’s about allyship,” she added. “You will have some people who are supportive and some who are not, but when you have the chief supporting you, you have a strong ally.”
The researchers suggest that one strategy is to temporarily replace cardiologists on maternity leave with locums, or “deepen the bench of coverage for clinical work, as is done for other absences. Given the expanding coverage of parental and family medical leaves, and awareness of these issues nationally, the need for this is likely to become less of an exception and more the rule.”
For example, nine states and Washington, D.C. now provide paid parental leave, they wrote, “and there is pending legislation in others.”
Dr. Bairey Merz and Dr. Mehta reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A survey of 323 women cardiologists who were working while they were pregnant showed that nearly 75% experienced discriminatory maternity-leave practices, some of which were likely violations of the Family and Medical Leave Act (FMLA).
More than 40% saw their salaries decreased during their year of pregnancy, 38% were required to perform extra service or call before taking maternity leave, exposing them to occupational hazards such as radiation, and 40% experienced a pregnancy complication, significantly higher than the general population and other medical specialties.
Additionally, of those who performed extra service or call, 18% were placed on bedrest before delivery, compared with 7.4% who did not perform extra service or call.
More than half of respondents reported that pregnancy negatively impacted their careers, and 42.4% said they experienced pressure to return to work and a delay in promotions, both illegal practices under the FMLA.
The survey is published in the Journal of the American College of Cardiology.
“Childbearing is difficult for women in cardiology with more than double the rate of gestational complications of the U.S. population, frequent income loss out of proportion to reduced productivity, and for nearly half, has an adverse impact on their career,” lead author Martha Gulati, MD, University of Arizona, Phoenix, said in a statement.
“While many professions struggle to create environments supportive of pregnancy and child-rearing, the prevalence of illegal behavior in cardiology is quite high and presents substantial legal risk for employers,” Dr. Gulati added.
C. Noel Bairey Merz, MD, professor of cardiology at Cedars-Sinai Smidt Heart Institute, Los Angeles, and a coauthor of the survey, told this news organization that it’s not surprising that such a situation exists, even “in this day and age.”
“I’m not surprised as a woman in cardiology myself. I was told by my training director that if I took off more than my allowed sick leave when I had my first and second children, I would have to repeat the year of training, so not surprised at all. I hear this from colleagues all the time,” Dr. Bairey Merz said.
The exchange left her feeling fearful for her career.
“Who wants to repeat a year? It pushes you back from a career standpoint, financially, everything. It also made me angry. I had a colleague who busted his leg in a motorcycle accident. He was unable to do any procedures for 16 weeks, and he didn’t have to repeat the year,” she pointed out.
The challenge that pregnancy represents is frequently cited by women as a deterrent for applying for a cardiology fellowship, Laxmi S. Mehta, MD, Ohio State University, Columbus, and colleagues wrote in an accompanying editorial.
The findings from the survey “reveal restrictive maternity leave data in a profession that has historically and currently continues to have a diversity problem,” they wrote.
“Maternity and pregnancy issues are a thing in cardiology,” Dr. Mehta said in an interview. “It’s one of the reasons why women get deterred from going into the field. It makes it challenging to choose cardiology if you perceive that the culture is negative, that it’s hard to be pregnant, or to bear children, or to take care of them post partum. It is problematic and it should not be occurring now.”
Leadership that condones such restrictive policies or even promotes them through ignorance and inaction needs to be held accountable, she added.
“We need to move forward from this negativity and make it more warm and welcoming to have families, whether you are a trainee or a practicing cardiologist, male or female. We need transparent and consistent parental leave policies and things like lactation support when a woman returns to work. That is a big issue,” Dr. Mehta said.
Having cardiovascular leaders champion the cause of adequate maternity and paternity leave are crucial to creating a newer, inclusive environment in cardiology.
As an example, Dr. Mehta recounted her own experience when she was in training 17 years ago.
“When I interviewed for a cardiology fellowship, one of the female program directors asked me if I was planning to have children, because if I did, the other fellows wouldn’t like it if they had to cover for me,” she said. “I ended up doing my fellowship where the chief of cardiology encouraged me to have children. He said: ‘Have your children during training, we will support you.’ And he did. I still had to do all of the call make-up and that stuff, but I worked in a supportive environment, and it made all the difference.”
“It’s about allyship,” she added. “You will have some people who are supportive and some who are not, but when you have the chief supporting you, you have a strong ally.”
The researchers suggest that one strategy is to temporarily replace cardiologists on maternity leave with locums, or “deepen the bench of coverage for clinical work, as is done for other absences. Given the expanding coverage of parental and family medical leaves, and awareness of these issues nationally, the need for this is likely to become less of an exception and more the rule.”
For example, nine states and Washington, D.C. now provide paid parental leave, they wrote, “and there is pending legislation in others.”
Dr. Bairey Merz and Dr. Mehta reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Food insecurity linked to metabolic syndrome in Hispanic/Latino youth
Severe food insecurity was associated with metabolic syndrome and unfavorable cardiometabolic markers in Hispanic/Latino youth, researchers report.
The findings, published March 16 in Pediatrics, highlight the need to investigate interventions that address food insecurity among Hispanic/Latino youth, a segment of the U.S. population at high risk of cardiometabolic complications.
“Among Hispanic/Latino youth, no study, to our knowledge has evaluated food insecurity’s role in metabolic syndrome and metabolic syndrome–relevant cardiometabolic markers in this population,” lead author Luis E. Maldonado, PhD, of the University of North Carolina at Chapel Hill, and colleagues explained.
The researchers conducted a cross-sectional study to evaluate the associations between lower household and child food security and metabolic syndrome, as well as clinically measured cardiometabolic markers, including fasting plasma glucose, waist circumference, triglycerides, systolic and diastolic blood pressure, and high-density lipoprotein cholesterol (HDL-C).
Household food security (high, marginal, low, very low) and child food security (high, marginal, low/very low) measures were evaluated separately, and were adjusted for participant age, sex, site, parental education, and poverty-income ratio.
Data were obtained from the Hispanic Community Children’s Health Study/Study of Latino Youth, a study of offspring of adults enrolled in the Hispanic Community Health Survey/Study of Latinos.
Results
The study cohort included 1,325 Hispanic/Latino youth aged 8-16 years. For both household food security and child food security, youth in the lowest food security category had significantly lower HDL-C compared with youth with high food security (household food security, –3.17; 95% confidence interval, –5.65 to –0.70; child food security, –1.81; 95% CI, –3.54 to –0.09).
In addition, low/very low compared with high child food security was associated with higher triglycerides (beta, 8.68; 95% CI, 1.75-15.61), higher fasting plasma glucose (beta, 1.37; 95% CI, 0.08-2.65), and metabolic syndrome composite variable expected log counts (beta, 2.12; 95% CI, 0.02-0.45).
Furthermore, the researchers found statistically significant interactions between each of the two food security measures and receipt of any food assistance in the previous year in models of triglycerides (P for interactions: household food security, .03 and child food security, .005) and HDL-C (P for interactions: household food security, .01 and child food security, .04).
After evaluating the effect of parental place of birth, they found a statistically significant association for triglycerides only (P for interactions: household food security, .05 and child food security, .008).
“Our study is among the first to document adverse associations between household and child food security measures with a metabolic syndrome score variable and several metabolic syndrome–relevant cardiometabolic markers among US Hispanic/Latino youth,” the researchers wrote.
The researchers acknowledged that the cross-sectional nature of the study was a key limitation; thus, causality could not be inferred.
“In the future, we plan to conduct more qualitative work to better understand how Hispanic/Latino families respond to food insecurity, which may identify the factors that shape their response,” study author Sandra S. Albrecht, PhD, of Columbia University, New York, NY, said in an interview.
Recommendations for pediatricians
Food insecurity researcher Yankun Wang, PhD candidate at Indiana University, Bloomington, commented: “I would recommend pediatricians pay more attention to children from low-income households since they are more likely to have mental and physical health issues due to food insecurity.
“It can be very helpful if pediatricians could help families obtain SNAP benefits, enroll youth in the school breakfast and lunch programs, and promote nutrition education in schools,” Mr. Wang added.
This study was supported by grant funding from the National Heart, Lung, and Blood Institute. The authors reported no relevant disclosures.
Severe food insecurity was associated with metabolic syndrome and unfavorable cardiometabolic markers in Hispanic/Latino youth, researchers report.
The findings, published March 16 in Pediatrics, highlight the need to investigate interventions that address food insecurity among Hispanic/Latino youth, a segment of the U.S. population at high risk of cardiometabolic complications.
“Among Hispanic/Latino youth, no study, to our knowledge has evaluated food insecurity’s role in metabolic syndrome and metabolic syndrome–relevant cardiometabolic markers in this population,” lead author Luis E. Maldonado, PhD, of the University of North Carolina at Chapel Hill, and colleagues explained.
The researchers conducted a cross-sectional study to evaluate the associations between lower household and child food security and metabolic syndrome, as well as clinically measured cardiometabolic markers, including fasting plasma glucose, waist circumference, triglycerides, systolic and diastolic blood pressure, and high-density lipoprotein cholesterol (HDL-C).
Household food security (high, marginal, low, very low) and child food security (high, marginal, low/very low) measures were evaluated separately, and were adjusted for participant age, sex, site, parental education, and poverty-income ratio.
Data were obtained from the Hispanic Community Children’s Health Study/Study of Latino Youth, a study of offspring of adults enrolled in the Hispanic Community Health Survey/Study of Latinos.
Results
The study cohort included 1,325 Hispanic/Latino youth aged 8-16 years. For both household food security and child food security, youth in the lowest food security category had significantly lower HDL-C compared with youth with high food security (household food security, –3.17; 95% confidence interval, –5.65 to –0.70; child food security, –1.81; 95% CI, –3.54 to –0.09).
In addition, low/very low compared with high child food security was associated with higher triglycerides (beta, 8.68; 95% CI, 1.75-15.61), higher fasting plasma glucose (beta, 1.37; 95% CI, 0.08-2.65), and metabolic syndrome composite variable expected log counts (beta, 2.12; 95% CI, 0.02-0.45).
Furthermore, the researchers found statistically significant interactions between each of the two food security measures and receipt of any food assistance in the previous year in models of triglycerides (P for interactions: household food security, .03 and child food security, .005) and HDL-C (P for interactions: household food security, .01 and child food security, .04).
After evaluating the effect of parental place of birth, they found a statistically significant association for triglycerides only (P for interactions: household food security, .05 and child food security, .008).
“Our study is among the first to document adverse associations between household and child food security measures with a metabolic syndrome score variable and several metabolic syndrome–relevant cardiometabolic markers among US Hispanic/Latino youth,” the researchers wrote.
The researchers acknowledged that the cross-sectional nature of the study was a key limitation; thus, causality could not be inferred.
“In the future, we plan to conduct more qualitative work to better understand how Hispanic/Latino families respond to food insecurity, which may identify the factors that shape their response,” study author Sandra S. Albrecht, PhD, of Columbia University, New York, NY, said in an interview.
Recommendations for pediatricians
Food insecurity researcher Yankun Wang, PhD candidate at Indiana University, Bloomington, commented: “I would recommend pediatricians pay more attention to children from low-income households since they are more likely to have mental and physical health issues due to food insecurity.
“It can be very helpful if pediatricians could help families obtain SNAP benefits, enroll youth in the school breakfast and lunch programs, and promote nutrition education in schools,” Mr. Wang added.
This study was supported by grant funding from the National Heart, Lung, and Blood Institute. The authors reported no relevant disclosures.
Severe food insecurity was associated with metabolic syndrome and unfavorable cardiometabolic markers in Hispanic/Latino youth, researchers report.
The findings, published March 16 in Pediatrics, highlight the need to investigate interventions that address food insecurity among Hispanic/Latino youth, a segment of the U.S. population at high risk of cardiometabolic complications.
“Among Hispanic/Latino youth, no study, to our knowledge has evaluated food insecurity’s role in metabolic syndrome and metabolic syndrome–relevant cardiometabolic markers in this population,” lead author Luis E. Maldonado, PhD, of the University of North Carolina at Chapel Hill, and colleagues explained.
The researchers conducted a cross-sectional study to evaluate the associations between lower household and child food security and metabolic syndrome, as well as clinically measured cardiometabolic markers, including fasting plasma glucose, waist circumference, triglycerides, systolic and diastolic blood pressure, and high-density lipoprotein cholesterol (HDL-C).
Household food security (high, marginal, low, very low) and child food security (high, marginal, low/very low) measures were evaluated separately, and were adjusted for participant age, sex, site, parental education, and poverty-income ratio.
Data were obtained from the Hispanic Community Children’s Health Study/Study of Latino Youth, a study of offspring of adults enrolled in the Hispanic Community Health Survey/Study of Latinos.
Results
The study cohort included 1,325 Hispanic/Latino youth aged 8-16 years. For both household food security and child food security, youth in the lowest food security category had significantly lower HDL-C compared with youth with high food security (household food security, –3.17; 95% confidence interval, –5.65 to –0.70; child food security, –1.81; 95% CI, –3.54 to –0.09).
In addition, low/very low compared with high child food security was associated with higher triglycerides (beta, 8.68; 95% CI, 1.75-15.61), higher fasting plasma glucose (beta, 1.37; 95% CI, 0.08-2.65), and metabolic syndrome composite variable expected log counts (beta, 2.12; 95% CI, 0.02-0.45).
Furthermore, the researchers found statistically significant interactions between each of the two food security measures and receipt of any food assistance in the previous year in models of triglycerides (P for interactions: household food security, .03 and child food security, .005) and HDL-C (P for interactions: household food security, .01 and child food security, .04).
After evaluating the effect of parental place of birth, they found a statistically significant association for triglycerides only (P for interactions: household food security, .05 and child food security, .008).
“Our study is among the first to document adverse associations between household and child food security measures with a metabolic syndrome score variable and several metabolic syndrome–relevant cardiometabolic markers among US Hispanic/Latino youth,” the researchers wrote.
The researchers acknowledged that the cross-sectional nature of the study was a key limitation; thus, causality could not be inferred.
“In the future, we plan to conduct more qualitative work to better understand how Hispanic/Latino families respond to food insecurity, which may identify the factors that shape their response,” study author Sandra S. Albrecht, PhD, of Columbia University, New York, NY, said in an interview.
Recommendations for pediatricians
Food insecurity researcher Yankun Wang, PhD candidate at Indiana University, Bloomington, commented: “I would recommend pediatricians pay more attention to children from low-income households since they are more likely to have mental and physical health issues due to food insecurity.
“It can be very helpful if pediatricians could help families obtain SNAP benefits, enroll youth in the school breakfast and lunch programs, and promote nutrition education in schools,” Mr. Wang added.
This study was supported by grant funding from the National Heart, Lung, and Blood Institute. The authors reported no relevant disclosures.
FROM PEDIATRICS