User login
Atopic dermatitis: Pivotal dupilumab results create sensation
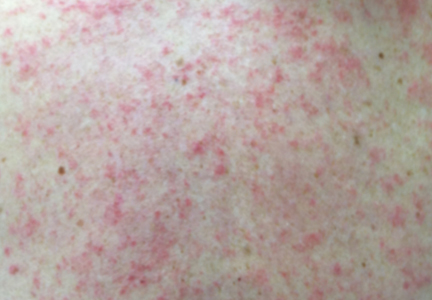
VIENNA – The marquee event at this year’s annual congress of the European Academy of Dermatology and Venereology – the one everyone was eagerly awaiting – was the first presentation of two large, international, pivotal phase III randomized trials of dupilumab for treatment of inadequately controlled moderate to severe atopic dermatitis in adults.
Attendees at EADV 2016 understood that, if positive, these studies, known as SOLO 1 and SOLO 2, would be transformative. They would herald a new era of highly effective targeted biologic therapy for this common and often debilitating chronic relapsing skin disease, akin to what occurred in psoriasis therapy well over a decade ago.
The results did not disappoint.
“Dual targeting of interleukin-4 and -13 represents a therapeutic option for patients with moderate to severe atopic dermatitis,” added Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
These results have implications extending beyond atopic dermatitis. Asthma, chronic sinusitis with nasal polyposis, and eosinophilic esophagitis are other conditions where the type 2 inflammatory cytokines IL-4 and -13 are believed to be important drivers of disease activity. Clinical trials of dupilumab in those diseases are underway.
Dupilumab, a fully human monoclonal antibody that binds specifically to the shared alpha chain subunit of the IL-4 and -13 receptors, hit all of its primary and secondary outcome measures in SOLO 1 and SOLO 2. Moreover, some of these “secondary” endpoints are consistently reported in patient surveys to be among what they consider to be the most troublesome aspects of atopic dermatitis, including intense itching, disrupted sleep, clinically significant anxiety and/or depression, and generally diminished quality of life.
SOLO 1 and SOLO 2 were identically designed, independent, randomized, double-blind, placebo-controlled clinical trials of 16 weeks’ duration. Conducted in North America, Europe, and Asia, they included a total of 1,379 patients, split roughly 50/50 between those with moderate or severe atopic dermatitis. Their average disease duration was 26 years. Participants were randomized to subcutaneous injection of dupilumab at 300 mg once weekly or every 2 weeks or to matching placebo.
The primary endpoint was a score of clear or almost clear – 0 or 1 – on the Investigator’s Global Assessment (IGA) at week 16 accompanied by a reduction of at least 2 points from baseline. A key secondary endpoint was at least a 75% improvement in the Eczema Area and Severity Index (EASI-75), considered a coprimary endpoint by regulators in Japan and the European Union.
The use of topical agents for atopic dermatitis was not permitted except as rescue therapy for uncontrolled symptoms. An IGA of 0 or 1 with at least a 2-point drop from baseline was a high bar to reach, given that a median of 50% of participants’ body surface area was affected. But in SOLO 1, that target was achieved in 37.9% of subjects on dupilumab every other week, 37.2% with weekly therapy, and just 10.3% of placebo-treated controls. Similarly, in SOLO 2, the rates were 36.1%, 36.4%, and 8.5%, respectively.
Of note, there were essentially no differences in outcomes across the board with weekly versus biweekly dosing of dupilumab.
From a median baseline EASI score of 30, an EASI-75 was achieved at 16 weeks in 51.3% of patients on dupilumab every other week, 52.5% on weekly injections, and 14.7% of controls in SOLO 1. In SOLO 2, the corresponding EASI-75 rates were 44.2%, 48.1%, and 11.9%, respectively.
Itch is described by most patients with moderate to severe atopic dermatitis as their No. 1 issue. From a baseline median peak score of 7.7 on a 0-10 numerical rating scale for pruritus, week 16 scores dropped by a median of 51% in patients on dupilumab every 2 weeks, 48.9% with weekly therapy, and 26.1% with placebo in SOLO 1. Results in SOLO 2 mirrored those in SOLO 1.
Particularly noteworthy was the finding that a significant reduction in itch severity was documented by week 2 in both dupilumab treatment arms, Dr. Simpson observed.
Just under half of study participants had a baseline score of 8 or more on the Hospital Anxiety and Depression Scale Anxiety subscale or HADS Depression subscale, considered the cutoff for a clinically significant mood disorder. Among affected patients, a score of less than 8 was achieved at 16 weeks without the use of psychotropic medications in 12.4% of SOLO 1 participants on placebo, 41% on biweekly dupilumab, and 36.3% with weekly dupilumab. In SOLO 2, the rates were 6.1% with placebo, 39.5% with biweekly dupilumab, and 41.2% with once-weekly dupilumab.
The median baseline Dermatology Life Quality Index score was 15 across the two parallel trials. The collective proportion of patients who experienced at least a 4-point improvement, which is considered a clinically meaningful response, was 29.1% in controls, compared with 68.6% in patients dupilumab every other week and 60.2% with weekly dupilumab.
On the Patient-Oriented Eczema Measure, a composite yardstick that emphasizes sleep symptoms, the median baseline score was 22 out of a possible 28. An improvement of 4 points or more, defined as a minimal clinically important difference, was achieved in a collective 25.6% of controls, 69.6% of patients on biweekly dupilumab, and 63.6% on weekly dupilumab.
Regarding safety, no increase in infections was seen with dupilumab. In fact, only two adverse events were more frequent than with placebo. One was injection-site reactions, which were two- to threefold more common than in controls, and all of which were mild to moderate. The other safety issue was conjunctivitis, which occurred in three patients in the control arms of SOLO 1 and 2, compared with 36 in the dupilumab arms.
Asked about the mechanism of this conjunctivitis, Dr. Simpson said it remains unknown. There was no signal of an issue in the phase II studies.
“Ongoing studies are attempting to further characterize the affected patients. I would say the comforting thing is that most cases have been mild to moderate and have responded to topical steroids or topical cyclosporine. Only one patient had to discontinue dupilumab,” according to the dermatologist.
In any event, 16 weeks of treatment is not sufficient to determine the safety of long-term therapy. Long-term extension studies of SOLO 1 and 2 are well underway, as are earlier stage clinical trials in pediatric patients with moderate to severe atopic dermatitis.
In response to another audience question, Dr. Simpson said he and his coinvestigators plan to drill down into the data to see if patients with severe atopic dermatitis obtained significantly more benefits from weekly as compared with biweekly therapy, or if treatment every 2 weeks was as good as weekly therapy across the board. It’s an important question, but the study finished so recently that the investigators haven’t yet had time to conduct the analysis.
The pivotal phase III dupilumab findings met with an enthusiastic reception.
“Biologic therapy for atopic dermatitis is the light at the end of the tunnel,” declared session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
“Seminal work,” commented David M. Pariser, MD, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Dr. Simpson’s presentation of the pivotal dupilumab studies was but one of the highlights of a horn-of-plenty late-breaking clinical trials session held on the final full day of EADV 2016. As attendees mingled in the hall afterward, a palpable sense of pride in their profession was evident. It was borne of the knowledge that their field not only includes basic and translational scientists capable of unraveling the inflammatory pathways involved in a challenging disease like atopic dermatitis, where there is a long-standing unmet need for new therapies, but also that their specialty includes experienced clinical trialists who can put those novel targeted therapies to the test.
There was also a sense of satisfaction that, although dermatology is a small specialty, these accomplishments are drawing favorable attention throughout the broader medical community. Pivotal trials of novel treatments for important dermatologic diseases are regularly getting published in prominent nondermatology journals. For instance, simultaneous with Dr. Simpson’s presentation in Vienna at EADV 2016, the SOLO 1 and 2 results were published online in the New England Journal of Medicine (doi. 10.1056/NEJMoa1610020).
“The online publication occurred a few minutes ago, at the start of my presentation. I didn’t say anything then because I didn’t want everybody looking at their cell phones,” he quipped.
The Food and Drug Administration has granted dupilumab a breakthrough therapy designation; a decision on the application for approval is expected by March 29, 2017.
The phase III dupilumab trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Simpson reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
VIENNA – The marquee event at this year’s annual congress of the European Academy of Dermatology and Venereology – the one everyone was eagerly awaiting – was the first presentation of two large, international, pivotal phase III randomized trials of dupilumab for treatment of inadequately controlled moderate to severe atopic dermatitis in adults.
Attendees at EADV 2016 understood that, if positive, these studies, known as SOLO 1 and SOLO 2, would be transformative. They would herald a new era of highly effective targeted biologic therapy for this common and often debilitating chronic relapsing skin disease, akin to what occurred in psoriasis therapy well over a decade ago.
The results did not disappoint.
“Dual targeting of interleukin-4 and -13 represents a therapeutic option for patients with moderate to severe atopic dermatitis,” added Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
These results have implications extending beyond atopic dermatitis. Asthma, chronic sinusitis with nasal polyposis, and eosinophilic esophagitis are other conditions where the type 2 inflammatory cytokines IL-4 and -13 are believed to be important drivers of disease activity. Clinical trials of dupilumab in those diseases are underway.
Dupilumab, a fully human monoclonal antibody that binds specifically to the shared alpha chain subunit of the IL-4 and -13 receptors, hit all of its primary and secondary outcome measures in SOLO 1 and SOLO 2. Moreover, some of these “secondary” endpoints are consistently reported in patient surveys to be among what they consider to be the most troublesome aspects of atopic dermatitis, including intense itching, disrupted sleep, clinically significant anxiety and/or depression, and generally diminished quality of life.
SOLO 1 and SOLO 2 were identically designed, independent, randomized, double-blind, placebo-controlled clinical trials of 16 weeks’ duration. Conducted in North America, Europe, and Asia, they included a total of 1,379 patients, split roughly 50/50 between those with moderate or severe atopic dermatitis. Their average disease duration was 26 years. Participants were randomized to subcutaneous injection of dupilumab at 300 mg once weekly or every 2 weeks or to matching placebo.
The primary endpoint was a score of clear or almost clear – 0 or 1 – on the Investigator’s Global Assessment (IGA) at week 16 accompanied by a reduction of at least 2 points from baseline. A key secondary endpoint was at least a 75% improvement in the Eczema Area and Severity Index (EASI-75), considered a coprimary endpoint by regulators in Japan and the European Union.
The use of topical agents for atopic dermatitis was not permitted except as rescue therapy for uncontrolled symptoms. An IGA of 0 or 1 with at least a 2-point drop from baseline was a high bar to reach, given that a median of 50% of participants’ body surface area was affected. But in SOLO 1, that target was achieved in 37.9% of subjects on dupilumab every other week, 37.2% with weekly therapy, and just 10.3% of placebo-treated controls. Similarly, in SOLO 2, the rates were 36.1%, 36.4%, and 8.5%, respectively.
Of note, there were essentially no differences in outcomes across the board with weekly versus biweekly dosing of dupilumab.
From a median baseline EASI score of 30, an EASI-75 was achieved at 16 weeks in 51.3% of patients on dupilumab every other week, 52.5% on weekly injections, and 14.7% of controls in SOLO 1. In SOLO 2, the corresponding EASI-75 rates were 44.2%, 48.1%, and 11.9%, respectively.
Itch is described by most patients with moderate to severe atopic dermatitis as their No. 1 issue. From a baseline median peak score of 7.7 on a 0-10 numerical rating scale for pruritus, week 16 scores dropped by a median of 51% in patients on dupilumab every 2 weeks, 48.9% with weekly therapy, and 26.1% with placebo in SOLO 1. Results in SOLO 2 mirrored those in SOLO 1.
Particularly noteworthy was the finding that a significant reduction in itch severity was documented by week 2 in both dupilumab treatment arms, Dr. Simpson observed.
Just under half of study participants had a baseline score of 8 or more on the Hospital Anxiety and Depression Scale Anxiety subscale or HADS Depression subscale, considered the cutoff for a clinically significant mood disorder. Among affected patients, a score of less than 8 was achieved at 16 weeks without the use of psychotropic medications in 12.4% of SOLO 1 participants on placebo, 41% on biweekly dupilumab, and 36.3% with weekly dupilumab. In SOLO 2, the rates were 6.1% with placebo, 39.5% with biweekly dupilumab, and 41.2% with once-weekly dupilumab.
The median baseline Dermatology Life Quality Index score was 15 across the two parallel trials. The collective proportion of patients who experienced at least a 4-point improvement, which is considered a clinically meaningful response, was 29.1% in controls, compared with 68.6% in patients dupilumab every other week and 60.2% with weekly dupilumab.
On the Patient-Oriented Eczema Measure, a composite yardstick that emphasizes sleep symptoms, the median baseline score was 22 out of a possible 28. An improvement of 4 points or more, defined as a minimal clinically important difference, was achieved in a collective 25.6% of controls, 69.6% of patients on biweekly dupilumab, and 63.6% on weekly dupilumab.
Regarding safety, no increase in infections was seen with dupilumab. In fact, only two adverse events were more frequent than with placebo. One was injection-site reactions, which were two- to threefold more common than in controls, and all of which were mild to moderate. The other safety issue was conjunctivitis, which occurred in three patients in the control arms of SOLO 1 and 2, compared with 36 in the dupilumab arms.
Asked about the mechanism of this conjunctivitis, Dr. Simpson said it remains unknown. There was no signal of an issue in the phase II studies.
“Ongoing studies are attempting to further characterize the affected patients. I would say the comforting thing is that most cases have been mild to moderate and have responded to topical steroids or topical cyclosporine. Only one patient had to discontinue dupilumab,” according to the dermatologist.
In any event, 16 weeks of treatment is not sufficient to determine the safety of long-term therapy. Long-term extension studies of SOLO 1 and 2 are well underway, as are earlier stage clinical trials in pediatric patients with moderate to severe atopic dermatitis.
In response to another audience question, Dr. Simpson said he and his coinvestigators plan to drill down into the data to see if patients with severe atopic dermatitis obtained significantly more benefits from weekly as compared with biweekly therapy, or if treatment every 2 weeks was as good as weekly therapy across the board. It’s an important question, but the study finished so recently that the investigators haven’t yet had time to conduct the analysis.
The pivotal phase III dupilumab findings met with an enthusiastic reception.
“Biologic therapy for atopic dermatitis is the light at the end of the tunnel,” declared session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
“Seminal work,” commented David M. Pariser, MD, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Dr. Simpson’s presentation of the pivotal dupilumab studies was but one of the highlights of a horn-of-plenty late-breaking clinical trials session held on the final full day of EADV 2016. As attendees mingled in the hall afterward, a palpable sense of pride in their profession was evident. It was borne of the knowledge that their field not only includes basic and translational scientists capable of unraveling the inflammatory pathways involved in a challenging disease like atopic dermatitis, where there is a long-standing unmet need for new therapies, but also that their specialty includes experienced clinical trialists who can put those novel targeted therapies to the test.
There was also a sense of satisfaction that, although dermatology is a small specialty, these accomplishments are drawing favorable attention throughout the broader medical community. Pivotal trials of novel treatments for important dermatologic diseases are regularly getting published in prominent nondermatology journals. For instance, simultaneous with Dr. Simpson’s presentation in Vienna at EADV 2016, the SOLO 1 and 2 results were published online in the New England Journal of Medicine (doi. 10.1056/NEJMoa1610020).
“The online publication occurred a few minutes ago, at the start of my presentation. I didn’t say anything then because I didn’t want everybody looking at their cell phones,” he quipped.
The Food and Drug Administration has granted dupilumab a breakthrough therapy designation; a decision on the application for approval is expected by March 29, 2017.
The phase III dupilumab trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Simpson reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
VIENNA – The marquee event at this year’s annual congress of the European Academy of Dermatology and Venereology – the one everyone was eagerly awaiting – was the first presentation of two large, international, pivotal phase III randomized trials of dupilumab for treatment of inadequately controlled moderate to severe atopic dermatitis in adults.
Attendees at EADV 2016 understood that, if positive, these studies, known as SOLO 1 and SOLO 2, would be transformative. They would herald a new era of highly effective targeted biologic therapy for this common and often debilitating chronic relapsing skin disease, akin to what occurred in psoriasis therapy well over a decade ago.
The results did not disappoint.
“Dual targeting of interleukin-4 and -13 represents a therapeutic option for patients with moderate to severe atopic dermatitis,” added Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
These results have implications extending beyond atopic dermatitis. Asthma, chronic sinusitis with nasal polyposis, and eosinophilic esophagitis are other conditions where the type 2 inflammatory cytokines IL-4 and -13 are believed to be important drivers of disease activity. Clinical trials of dupilumab in those diseases are underway.
Dupilumab, a fully human monoclonal antibody that binds specifically to the shared alpha chain subunit of the IL-4 and -13 receptors, hit all of its primary and secondary outcome measures in SOLO 1 and SOLO 2. Moreover, some of these “secondary” endpoints are consistently reported in patient surveys to be among what they consider to be the most troublesome aspects of atopic dermatitis, including intense itching, disrupted sleep, clinically significant anxiety and/or depression, and generally diminished quality of life.
SOLO 1 and SOLO 2 were identically designed, independent, randomized, double-blind, placebo-controlled clinical trials of 16 weeks’ duration. Conducted in North America, Europe, and Asia, they included a total of 1,379 patients, split roughly 50/50 between those with moderate or severe atopic dermatitis. Their average disease duration was 26 years. Participants were randomized to subcutaneous injection of dupilumab at 300 mg once weekly or every 2 weeks or to matching placebo.
The primary endpoint was a score of clear or almost clear – 0 or 1 – on the Investigator’s Global Assessment (IGA) at week 16 accompanied by a reduction of at least 2 points from baseline. A key secondary endpoint was at least a 75% improvement in the Eczema Area and Severity Index (EASI-75), considered a coprimary endpoint by regulators in Japan and the European Union.
The use of topical agents for atopic dermatitis was not permitted except as rescue therapy for uncontrolled symptoms. An IGA of 0 or 1 with at least a 2-point drop from baseline was a high bar to reach, given that a median of 50% of participants’ body surface area was affected. But in SOLO 1, that target was achieved in 37.9% of subjects on dupilumab every other week, 37.2% with weekly therapy, and just 10.3% of placebo-treated controls. Similarly, in SOLO 2, the rates were 36.1%, 36.4%, and 8.5%, respectively.
Of note, there were essentially no differences in outcomes across the board with weekly versus biweekly dosing of dupilumab.
From a median baseline EASI score of 30, an EASI-75 was achieved at 16 weeks in 51.3% of patients on dupilumab every other week, 52.5% on weekly injections, and 14.7% of controls in SOLO 1. In SOLO 2, the corresponding EASI-75 rates were 44.2%, 48.1%, and 11.9%, respectively.
Itch is described by most patients with moderate to severe atopic dermatitis as their No. 1 issue. From a baseline median peak score of 7.7 on a 0-10 numerical rating scale for pruritus, week 16 scores dropped by a median of 51% in patients on dupilumab every 2 weeks, 48.9% with weekly therapy, and 26.1% with placebo in SOLO 1. Results in SOLO 2 mirrored those in SOLO 1.
Particularly noteworthy was the finding that a significant reduction in itch severity was documented by week 2 in both dupilumab treatment arms, Dr. Simpson observed.
Just under half of study participants had a baseline score of 8 or more on the Hospital Anxiety and Depression Scale Anxiety subscale or HADS Depression subscale, considered the cutoff for a clinically significant mood disorder. Among affected patients, a score of less than 8 was achieved at 16 weeks without the use of psychotropic medications in 12.4% of SOLO 1 participants on placebo, 41% on biweekly dupilumab, and 36.3% with weekly dupilumab. In SOLO 2, the rates were 6.1% with placebo, 39.5% with biweekly dupilumab, and 41.2% with once-weekly dupilumab.
The median baseline Dermatology Life Quality Index score was 15 across the two parallel trials. The collective proportion of patients who experienced at least a 4-point improvement, which is considered a clinically meaningful response, was 29.1% in controls, compared with 68.6% in patients dupilumab every other week and 60.2% with weekly dupilumab.
On the Patient-Oriented Eczema Measure, a composite yardstick that emphasizes sleep symptoms, the median baseline score was 22 out of a possible 28. An improvement of 4 points or more, defined as a minimal clinically important difference, was achieved in a collective 25.6% of controls, 69.6% of patients on biweekly dupilumab, and 63.6% on weekly dupilumab.
Regarding safety, no increase in infections was seen with dupilumab. In fact, only two adverse events were more frequent than with placebo. One was injection-site reactions, which were two- to threefold more common than in controls, and all of which were mild to moderate. The other safety issue was conjunctivitis, which occurred in three patients in the control arms of SOLO 1 and 2, compared with 36 in the dupilumab arms.
Asked about the mechanism of this conjunctivitis, Dr. Simpson said it remains unknown. There was no signal of an issue in the phase II studies.
“Ongoing studies are attempting to further characterize the affected patients. I would say the comforting thing is that most cases have been mild to moderate and have responded to topical steroids or topical cyclosporine. Only one patient had to discontinue dupilumab,” according to the dermatologist.
In any event, 16 weeks of treatment is not sufficient to determine the safety of long-term therapy. Long-term extension studies of SOLO 1 and 2 are well underway, as are earlier stage clinical trials in pediatric patients with moderate to severe atopic dermatitis.
In response to another audience question, Dr. Simpson said he and his coinvestigators plan to drill down into the data to see if patients with severe atopic dermatitis obtained significantly more benefits from weekly as compared with biweekly therapy, or if treatment every 2 weeks was as good as weekly therapy across the board. It’s an important question, but the study finished so recently that the investigators haven’t yet had time to conduct the analysis.
The pivotal phase III dupilumab findings met with an enthusiastic reception.
“Biologic therapy for atopic dermatitis is the light at the end of the tunnel,” declared session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
“Seminal work,” commented David M. Pariser, MD, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Dr. Simpson’s presentation of the pivotal dupilumab studies was but one of the highlights of a horn-of-plenty late-breaking clinical trials session held on the final full day of EADV 2016. As attendees mingled in the hall afterward, a palpable sense of pride in their profession was evident. It was borne of the knowledge that their field not only includes basic and translational scientists capable of unraveling the inflammatory pathways involved in a challenging disease like atopic dermatitis, where there is a long-standing unmet need for new therapies, but also that their specialty includes experienced clinical trialists who can put those novel targeted therapies to the test.
There was also a sense of satisfaction that, although dermatology is a small specialty, these accomplishments are drawing favorable attention throughout the broader medical community. Pivotal trials of novel treatments for important dermatologic diseases are regularly getting published in prominent nondermatology journals. For instance, simultaneous with Dr. Simpson’s presentation in Vienna at EADV 2016, the SOLO 1 and 2 results were published online in the New England Journal of Medicine (doi. 10.1056/NEJMoa1610020).
“The online publication occurred a few minutes ago, at the start of my presentation. I didn’t say anything then because I didn’t want everybody looking at their cell phones,” he quipped.
The Food and Drug Administration has granted dupilumab a breakthrough therapy designation; a decision on the application for approval is expected by March 29, 2017.
The phase III dupilumab trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Simpson reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
Key clinical point:
Major finding: After 16 weeks of weekly or biweekly subcutaneous injections of dupilumab, 36%-38% of patients with baseline moderate or severe atopic dermatitis were clear or almost clear, compared with 8%-10% of placebo-treated controls.
Data source: The SOLO 1 and SOLO 2 pivotal phase III randomized, double-blind, placebo-controlled clinical trials included a total of 1,379 adults with inadequately controlled moderate or severe atopic dermatitis on three continents.
Disclosures: The trials were funded by Sanofi and Regeneron Pharmaceuticals. The presenter reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
New-onset pediatric AD phenotype differs from adult AD
The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, according to an assessment of biopsy findings in infants and children.
The study findings have important therapeutic implications, especially in light of the fact that much of the work in this area has been based on adult biomarkers, reflecting “decades of disease activity and chronic use of immunosuppressants in adults,” the investigators reported. Little is known about alterations in early lesions in children, which limits the advancement of targeted therapies, Hitokazu Esaki, MD, of the Icahn School of Medicine at Mount Sinai in New York City, and colleagues reported online in the Journal of Allergy and Clinical Immunology (2016 Sep 22. doi: 10.1016/j.jaci.2016.07.013).
To characterize early pediatric AD skin phenotype, the investigators assessed lesional and nonlesional biopsies from 19 children under age 5 years (mean, 1.3 years) within 6 months of moderate to severe disease onset, as well as those from age-matched controls and adults, and found that, compared with adult AD, early AD involves comparable or greater epidermal hyperplasia and cellular infiltration, similar strong activation of Th2 and Th22 axes, and some Th1 skewing.
In addition, early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD. Expression of filaggrin – an abundant barrier differentiation protein – was similar in AD and healthy children, whereas down-regulation is characteristic in adult AD, the investigators noted.
Nonlesional skin biopsies from the children showed both higher levels of inflammation and epidermal proliferation markers, they said.
The “surprising findings” of an early multicytokine response in new-onset pediatric AD, characterized by marked Th17, Th9, Th2, and Th22 activation, suggest that targeting of multiple cytokine axes may be needed in children with early-onset AD, one of the lead authors on the study, Emma Guttman-Yassky, MD, also of the Icahn School of Medicine at Mount Sinai, said in an interview.
Dr. Guttman-Yassky, who noted that the study was conducted in close collaboration with Amy S. Paller, MD, of Northwestern University, Chicago, explained that early AD, compared with adult AD, involves differential immune skewing and barrier responses with features that are in some ways comparable to those of psoriasis – particularly with respect to the consistently higher levels of Th17-related mediators in childhood AD, as psoriasis is considered a Th17-centered disease.
Further, the findings with respect to filaggrin represent another important aspect of the study, she said, noting that they represent a possible challenge to the notion that filaggrin is integral to disease elicitation and instigation of the “atopic march.”
The study findings may suggest novel targets for pediatric AD, and they also suggest a need for early immune intervention, not only to treat the AD, but also to prevent the atopic march, she said.
“These findings are likely to result in both different understanding of AD onset and distinct treatment approaches for infants and children,” she and her colleagues concluded.
This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
Atopic dermatitis (AD) is the most common chronic inflammatory skin disease. Severe AD places a huge burden on patients, their families, and society in terms of health care dollars spent and lost work days. Considering the prevalence of AD, both families and dermatologists find it understandably frustrating that we have limited and often ineffective tools to treat severe AD in children. Change may be around the corner.
This study by Esaki et al. sheds critical light on the pathogenesis of early onset AD in children and we hope it will set the stage to revolutionize the treatment of AD using the paradigm of psoriasis as a model. Using lesional and nonlesional biopsies from 19 children under age 5 obtained during the first 6 months of onset of AD, Esaki et al. have demonstrated that children with AD have a multicytokine inflammatory infiltrate with Th17 predominance. This sets the stage for biologics focused on the Th17 pathway in these children, although multimodal therapy to address different cytokines may ultimately be required.
The investigators also found that children with AD had similar filaggrin expression compared to control children, implying that atopic dermatitis is at its heart an immunologic disorder rather than a barrier defect although we will likely continue to learn more about this fine balance.
As pediatric dermatologists on the front line caring for patients with severe AD, we welcome further studies and especially look forward to effective treatments for our patients who might finally experience relief of itch, clear skin, and a good night’s sleep.
A. Yasmine Kirkorian, MD, and Kalyani Marathe, MD, are pediatric dermatologists at Children’s National Health System, in the departments of dermatology and pediatrics at George Washington University, Washington, DC. They are on the editorial advisory board of Dermatology News. They had no disclosures.
Atopic dermatitis (AD) is the most common chronic inflammatory skin disease. Severe AD places a huge burden on patients, their families, and society in terms of health care dollars spent and lost work days. Considering the prevalence of AD, both families and dermatologists find it understandably frustrating that we have limited and often ineffective tools to treat severe AD in children. Change may be around the corner.
This study by Esaki et al. sheds critical light on the pathogenesis of early onset AD in children and we hope it will set the stage to revolutionize the treatment of AD using the paradigm of psoriasis as a model. Using lesional and nonlesional biopsies from 19 children under age 5 obtained during the first 6 months of onset of AD, Esaki et al. have demonstrated that children with AD have a multicytokine inflammatory infiltrate with Th17 predominance. This sets the stage for biologics focused on the Th17 pathway in these children, although multimodal therapy to address different cytokines may ultimately be required.
The investigators also found that children with AD had similar filaggrin expression compared to control children, implying that atopic dermatitis is at its heart an immunologic disorder rather than a barrier defect although we will likely continue to learn more about this fine balance.
As pediatric dermatologists on the front line caring for patients with severe AD, we welcome further studies and especially look forward to effective treatments for our patients who might finally experience relief of itch, clear skin, and a good night’s sleep.
A. Yasmine Kirkorian, MD, and Kalyani Marathe, MD, are pediatric dermatologists at Children’s National Health System, in the departments of dermatology and pediatrics at George Washington University, Washington, DC. They are on the editorial advisory board of Dermatology News. They had no disclosures.
Atopic dermatitis (AD) is the most common chronic inflammatory skin disease. Severe AD places a huge burden on patients, their families, and society in terms of health care dollars spent and lost work days. Considering the prevalence of AD, both families and dermatologists find it understandably frustrating that we have limited and often ineffective tools to treat severe AD in children. Change may be around the corner.
This study by Esaki et al. sheds critical light on the pathogenesis of early onset AD in children and we hope it will set the stage to revolutionize the treatment of AD using the paradigm of psoriasis as a model. Using lesional and nonlesional biopsies from 19 children under age 5 obtained during the first 6 months of onset of AD, Esaki et al. have demonstrated that children with AD have a multicytokine inflammatory infiltrate with Th17 predominance. This sets the stage for biologics focused on the Th17 pathway in these children, although multimodal therapy to address different cytokines may ultimately be required.
The investigators also found that children with AD had similar filaggrin expression compared to control children, implying that atopic dermatitis is at its heart an immunologic disorder rather than a barrier defect although we will likely continue to learn more about this fine balance.
As pediatric dermatologists on the front line caring for patients with severe AD, we welcome further studies and especially look forward to effective treatments for our patients who might finally experience relief of itch, clear skin, and a good night’s sleep.
A. Yasmine Kirkorian, MD, and Kalyani Marathe, MD, are pediatric dermatologists at Children’s National Health System, in the departments of dermatology and pediatrics at George Washington University, Washington, DC. They are on the editorial advisory board of Dermatology News. They had no disclosures.
The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, according to an assessment of biopsy findings in infants and children.
The study findings have important therapeutic implications, especially in light of the fact that much of the work in this area has been based on adult biomarkers, reflecting “decades of disease activity and chronic use of immunosuppressants in adults,” the investigators reported. Little is known about alterations in early lesions in children, which limits the advancement of targeted therapies, Hitokazu Esaki, MD, of the Icahn School of Medicine at Mount Sinai in New York City, and colleagues reported online in the Journal of Allergy and Clinical Immunology (2016 Sep 22. doi: 10.1016/j.jaci.2016.07.013).
To characterize early pediatric AD skin phenotype, the investigators assessed lesional and nonlesional biopsies from 19 children under age 5 years (mean, 1.3 years) within 6 months of moderate to severe disease onset, as well as those from age-matched controls and adults, and found that, compared with adult AD, early AD involves comparable or greater epidermal hyperplasia and cellular infiltration, similar strong activation of Th2 and Th22 axes, and some Th1 skewing.
In addition, early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD. Expression of filaggrin – an abundant barrier differentiation protein – was similar in AD and healthy children, whereas down-regulation is characteristic in adult AD, the investigators noted.
Nonlesional skin biopsies from the children showed both higher levels of inflammation and epidermal proliferation markers, they said.
The “surprising findings” of an early multicytokine response in new-onset pediatric AD, characterized by marked Th17, Th9, Th2, and Th22 activation, suggest that targeting of multiple cytokine axes may be needed in children with early-onset AD, one of the lead authors on the study, Emma Guttman-Yassky, MD, also of the Icahn School of Medicine at Mount Sinai, said in an interview.
Dr. Guttman-Yassky, who noted that the study was conducted in close collaboration with Amy S. Paller, MD, of Northwestern University, Chicago, explained that early AD, compared with adult AD, involves differential immune skewing and barrier responses with features that are in some ways comparable to those of psoriasis – particularly with respect to the consistently higher levels of Th17-related mediators in childhood AD, as psoriasis is considered a Th17-centered disease.
Further, the findings with respect to filaggrin represent another important aspect of the study, she said, noting that they represent a possible challenge to the notion that filaggrin is integral to disease elicitation and instigation of the “atopic march.”
The study findings may suggest novel targets for pediatric AD, and they also suggest a need for early immune intervention, not only to treat the AD, but also to prevent the atopic march, she said.
“These findings are likely to result in both different understanding of AD onset and distinct treatment approaches for infants and children,” she and her colleagues concluded.
This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, according to an assessment of biopsy findings in infants and children.
The study findings have important therapeutic implications, especially in light of the fact that much of the work in this area has been based on adult biomarkers, reflecting “decades of disease activity and chronic use of immunosuppressants in adults,” the investigators reported. Little is known about alterations in early lesions in children, which limits the advancement of targeted therapies, Hitokazu Esaki, MD, of the Icahn School of Medicine at Mount Sinai in New York City, and colleagues reported online in the Journal of Allergy and Clinical Immunology (2016 Sep 22. doi: 10.1016/j.jaci.2016.07.013).
To characterize early pediatric AD skin phenotype, the investigators assessed lesional and nonlesional biopsies from 19 children under age 5 years (mean, 1.3 years) within 6 months of moderate to severe disease onset, as well as those from age-matched controls and adults, and found that, compared with adult AD, early AD involves comparable or greater epidermal hyperplasia and cellular infiltration, similar strong activation of Th2 and Th22 axes, and some Th1 skewing.
In addition, early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD. Expression of filaggrin – an abundant barrier differentiation protein – was similar in AD and healthy children, whereas down-regulation is characteristic in adult AD, the investigators noted.
Nonlesional skin biopsies from the children showed both higher levels of inflammation and epidermal proliferation markers, they said.
The “surprising findings” of an early multicytokine response in new-onset pediatric AD, characterized by marked Th17, Th9, Th2, and Th22 activation, suggest that targeting of multiple cytokine axes may be needed in children with early-onset AD, one of the lead authors on the study, Emma Guttman-Yassky, MD, also of the Icahn School of Medicine at Mount Sinai, said in an interview.
Dr. Guttman-Yassky, who noted that the study was conducted in close collaboration with Amy S. Paller, MD, of Northwestern University, Chicago, explained that early AD, compared with adult AD, involves differential immune skewing and barrier responses with features that are in some ways comparable to those of psoriasis – particularly with respect to the consistently higher levels of Th17-related mediators in childhood AD, as psoriasis is considered a Th17-centered disease.
Further, the findings with respect to filaggrin represent another important aspect of the study, she said, noting that they represent a possible challenge to the notion that filaggrin is integral to disease elicitation and instigation of the “atopic march.”
The study findings may suggest novel targets for pediatric AD, and they also suggest a need for early immune intervention, not only to treat the AD, but also to prevent the atopic march, she said.
“These findings are likely to result in both different understanding of AD onset and distinct treatment approaches for infants and children,” she and her colleagues concluded.
This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Key clinical point: The skin phenotype of new-onset pediatric atopic dermatitis differs substantially from that in adult AD, which has important therapeutic implications, according to a study of biopsy findings in infants and children.
Major finding: Early AD involves significantly higher induction of Th17-related cytokines, compared with adult AD.
Data source: An analysis of biopsies from 19 children with AD.
Disclosures: This work was funded by a research grant from the LEO Foundation. Individual authors were supported by grants from the National Center for Advancing Translational Sciences and the National Institutes of Health Clinical and Translational Science Award program. Dr. Esaki reported having no disclosures. Dr. Guttman-Yassky reported financial relationships with numerous pharmaceutical companies.
Influence of Diet in Acne Vulgaris and Atopic Dermatitis
When I am in clinic, I often get at least 3 to 4 inquiries each day from patients about the necessity for dietary restrictions or alterations as well as the benefits of these changes in limiting their dermatological disease processes. I usually am restricted in my response because the research rarely indicates benefits of one diet versus another; however, this discussion has recently become a heavily researched area as patients have come to value natural nonpharmaceutical approaches to their holistic care. In this article, a few dietary restrictions and supplements are reviewed that may have a beneficial effect in managing patients with acne vulgaris and atopic dermatitis.
Acne Vulgaris
In 1969 Fulton et al1 conducted one of the first few trials on acne and diet management. In this crossover, patient-blinded, interventional study, patients were divided into 2 subgroups (N=65): 1 adolescent patient with moderate acne was compared to 1 male prisoner given a chocolate bar for 4 weeks or a control bar with equivalent caloric index. The results indicated no change in acne vulgaris lesions based on either intervention; however, there were obvious deficiencies in the study including small sample size, inappropriate grouping of an adolescent patient versus a prisoner, and limited study period.1
Since then, multiple studies have been conducted with parallel participants, large sample sizes, and at least a 12-week study period. In 2005, Adebamowo et al2 studied 47,355 women using a validated food frequency questionnaire that determined the amount of dairy consumed, specifically skim milk. The study showed a positive link between increased dairy consumption and acne formation; however, again due to the retrospective analysis and recall bias, it is difficult to determine if a link can truly be noted between acne and dairy in this study.2
More recently, LaRosa et al3 conducted a study that included 225 participants aged 14 to 19 years. Excluding participants with lactose intolerance and current use of oral contraceptives and isotretinoin, the study placed 120 participants in the test group versus 105 participants in the control group. The study was conducted using 3 telephone interviews and a 24-hour diet recall technique. The results supported a link between acne and skim milk consumption. Again, although the studied relied on participant self-reports of diet and followed a case-control design, a possible association was suspected but not validated.3 A longitudinal, questionnaire-based population study performed by Ulvestad et al4 included 2489 patients. This study further evaluated recall of dairy product consumption at 15 to 16 years of age and then 3 years later acne severity was self-assessed and reported at 18 to 19 years of age. Overall, this evaluation indicated that a high intake of dairy products and acne in adolescence have been positively associated. However, it was another retrospective study with recall bias.4 In 2009 Melnick and Schmitz5 concluded that milk causes the body to elevate both insulin and insulinlike growth factor 1 levels. In another study by Melnick6 in 2011, a definitive link between increased insulin and insulinlike growth factor 1 signaling in promoting comedogenesis was reported. Given the few studies that show the potential link between dairy products and acne, this dairy-free diet can be considered as a diet recommendation for acne patients.
Atopic Dermatitis
A Cochrane review conducted in 2012 regarding dietary supplements as a treatment of atopic dermatitis evaluated randomized controlled trials (N=596). Supplementation with vitamin D, fish oil, olive oil, zinc sulfate, selenium, vitamin E, pyridoxine, sea buckthorn seed oil, hempseed oil, sunflower oil (linoleic acid), and docosahexaenoic acid were evaluated among all the studies reviewed for atopic dermatitis.7 Bronsnick et al8 conducted a review of evidence supporting vitamin supplementation and atopic dermatitis, and for the most part determined that the studies had insufficient evidence. The only positive correlation was noted with prebiotics and probiotics in another Cochrane review in 2013, which evaluated 4 studies with 1428 infants showing prebiotic supplementation reduced atopic dermatitis.9 In 2014 Panduru et al10 evaluated 16 studies in a meta-analysis that showed how probiotics were possibly beneficial in both general and high-risk atopic populations. Specifically, a subgroup analysis showed that Lactobacillus and Lactobacillus with Bifidobacterium also can be protective against atopic dermatitis.10 Lastly, diet avoidance in pregnancy or during lactation in infants up to 18 months of age did not have any effect on improving the infant’s atopic dermatitis based on a 2012 Cochrane review that included 952 participants.11
Conclusion
Overall, there are some benefits to dietary restrictions and supplementation as indicated by the studies reviewed here; however, the extent to which these changes contribute to disease manifestation has only been linked, not definitively proven. Randomized controlled trials with large sample sizes, double-blind studies, and appropriately controlled studies with comparative patient populations are difficult to obtain, as diet cannot be completely restrictive for every patient. Patients should be provided with the latest data supporting a possible link between dairy consumption and acne production as well as prebiotics or probiotics during pregnancy and at infancy to reduce the risk for atopic dermatitis with the caveat of association. That said, future studies might prove that dietary and environmental alterations may prevent disease progression or appearance far more than previously assumed.
- Fulton JE Jr, Plewaig G, Kligman AM. Effect of chocolate on acne vulgaris. JAMA. 1969;210: 2071-2074.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. High school dietary diary intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- LaRosa CL, Quach KA, Koons K, et al. Consumption of dairy in teenagers with and without acne. J Am Acad Dermatol. 2016;75:318-322.
- Ulvestad M, Bjertness E, Dalgard F, et al. Acne and dairy products in adolescence: results from a Norwegian longitudinal study [published online ahead of print July 16, 2016]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.13835.
- Melnick BC, Schmitz G. Role of insulin, insulin like growth factor 1, hyperglycemic food and milk consumption in the pathogenesis of acne vulgaris. Exp Dermatol. 2009;18:833-841.
- Melnick BC. Evidence for acne-promoting effect of milk and other insulinotropic dairy products. Nestle Nutr Worksop
Ser Pediatr Program. 2011;67:131-145. - Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;2:CD005205.
- Bronsnick T, Murzaku EC, Rao BK. Diet in dermatology: part i. atopic dermatitis, acne, and nonmelanoma skin cancer [published online November 15, 2014]. J Am Acad Dermatol. 2014;71:1039.e1-1039.e12.
- Osborn DA, Sinn JKH. Prebiotics in infants for prevention of allergy. Cochrane Database Syst Rev. 2013;2:CD006474.
- Panduru M, Panduru NM, Saˇlaˇvaˇstru CM, et al. Probiotics and primary prevention of atopic dermatitis: a meta-analysis of randomized controlled studies [published online April 4, 2014]. J Eur Acad Dermatol Venereol. 2015;29:232-242.
- Kramer MS, Kakuma R. Maternal dietary antigen avoidance during pregnancy or lactation, or both, for preventing or treating atopic disease in the child. Cochrane Database Syst Rev. 2012;9:CD000133.
When I am in clinic, I often get at least 3 to 4 inquiries each day from patients about the necessity for dietary restrictions or alterations as well as the benefits of these changes in limiting their dermatological disease processes. I usually am restricted in my response because the research rarely indicates benefits of one diet versus another; however, this discussion has recently become a heavily researched area as patients have come to value natural nonpharmaceutical approaches to their holistic care. In this article, a few dietary restrictions and supplements are reviewed that may have a beneficial effect in managing patients with acne vulgaris and atopic dermatitis.
Acne Vulgaris
In 1969 Fulton et al1 conducted one of the first few trials on acne and diet management. In this crossover, patient-blinded, interventional study, patients were divided into 2 subgroups (N=65): 1 adolescent patient with moderate acne was compared to 1 male prisoner given a chocolate bar for 4 weeks or a control bar with equivalent caloric index. The results indicated no change in acne vulgaris lesions based on either intervention; however, there were obvious deficiencies in the study including small sample size, inappropriate grouping of an adolescent patient versus a prisoner, and limited study period.1
Since then, multiple studies have been conducted with parallel participants, large sample sizes, and at least a 12-week study period. In 2005, Adebamowo et al2 studied 47,355 women using a validated food frequency questionnaire that determined the amount of dairy consumed, specifically skim milk. The study showed a positive link between increased dairy consumption and acne formation; however, again due to the retrospective analysis and recall bias, it is difficult to determine if a link can truly be noted between acne and dairy in this study.2
More recently, LaRosa et al3 conducted a study that included 225 participants aged 14 to 19 years. Excluding participants with lactose intolerance and current use of oral contraceptives and isotretinoin, the study placed 120 participants in the test group versus 105 participants in the control group. The study was conducted using 3 telephone interviews and a 24-hour diet recall technique. The results supported a link between acne and skim milk consumption. Again, although the studied relied on participant self-reports of diet and followed a case-control design, a possible association was suspected but not validated.3 A longitudinal, questionnaire-based population study performed by Ulvestad et al4 included 2489 patients. This study further evaluated recall of dairy product consumption at 15 to 16 years of age and then 3 years later acne severity was self-assessed and reported at 18 to 19 years of age. Overall, this evaluation indicated that a high intake of dairy products and acne in adolescence have been positively associated. However, it was another retrospective study with recall bias.4 In 2009 Melnick and Schmitz5 concluded that milk causes the body to elevate both insulin and insulinlike growth factor 1 levels. In another study by Melnick6 in 2011, a definitive link between increased insulin and insulinlike growth factor 1 signaling in promoting comedogenesis was reported. Given the few studies that show the potential link between dairy products and acne, this dairy-free diet can be considered as a diet recommendation for acne patients.
Atopic Dermatitis
A Cochrane review conducted in 2012 regarding dietary supplements as a treatment of atopic dermatitis evaluated randomized controlled trials (N=596). Supplementation with vitamin D, fish oil, olive oil, zinc sulfate, selenium, vitamin E, pyridoxine, sea buckthorn seed oil, hempseed oil, sunflower oil (linoleic acid), and docosahexaenoic acid were evaluated among all the studies reviewed for atopic dermatitis.7 Bronsnick et al8 conducted a review of evidence supporting vitamin supplementation and atopic dermatitis, and for the most part determined that the studies had insufficient evidence. The only positive correlation was noted with prebiotics and probiotics in another Cochrane review in 2013, which evaluated 4 studies with 1428 infants showing prebiotic supplementation reduced atopic dermatitis.9 In 2014 Panduru et al10 evaluated 16 studies in a meta-analysis that showed how probiotics were possibly beneficial in both general and high-risk atopic populations. Specifically, a subgroup analysis showed that Lactobacillus and Lactobacillus with Bifidobacterium also can be protective against atopic dermatitis.10 Lastly, diet avoidance in pregnancy or during lactation in infants up to 18 months of age did not have any effect on improving the infant’s atopic dermatitis based on a 2012 Cochrane review that included 952 participants.11
Conclusion
Overall, there are some benefits to dietary restrictions and supplementation as indicated by the studies reviewed here; however, the extent to which these changes contribute to disease manifestation has only been linked, not definitively proven. Randomized controlled trials with large sample sizes, double-blind studies, and appropriately controlled studies with comparative patient populations are difficult to obtain, as diet cannot be completely restrictive for every patient. Patients should be provided with the latest data supporting a possible link between dairy consumption and acne production as well as prebiotics or probiotics during pregnancy and at infancy to reduce the risk for atopic dermatitis with the caveat of association. That said, future studies might prove that dietary and environmental alterations may prevent disease progression or appearance far more than previously assumed.
When I am in clinic, I often get at least 3 to 4 inquiries each day from patients about the necessity for dietary restrictions or alterations as well as the benefits of these changes in limiting their dermatological disease processes. I usually am restricted in my response because the research rarely indicates benefits of one diet versus another; however, this discussion has recently become a heavily researched area as patients have come to value natural nonpharmaceutical approaches to their holistic care. In this article, a few dietary restrictions and supplements are reviewed that may have a beneficial effect in managing patients with acne vulgaris and atopic dermatitis.
Acne Vulgaris
In 1969 Fulton et al1 conducted one of the first few trials on acne and diet management. In this crossover, patient-blinded, interventional study, patients were divided into 2 subgroups (N=65): 1 adolescent patient with moderate acne was compared to 1 male prisoner given a chocolate bar for 4 weeks or a control bar with equivalent caloric index. The results indicated no change in acne vulgaris lesions based on either intervention; however, there were obvious deficiencies in the study including small sample size, inappropriate grouping of an adolescent patient versus a prisoner, and limited study period.1
Since then, multiple studies have been conducted with parallel participants, large sample sizes, and at least a 12-week study period. In 2005, Adebamowo et al2 studied 47,355 women using a validated food frequency questionnaire that determined the amount of dairy consumed, specifically skim milk. The study showed a positive link between increased dairy consumption and acne formation; however, again due to the retrospective analysis and recall bias, it is difficult to determine if a link can truly be noted between acne and dairy in this study.2
More recently, LaRosa et al3 conducted a study that included 225 participants aged 14 to 19 years. Excluding participants with lactose intolerance and current use of oral contraceptives and isotretinoin, the study placed 120 participants in the test group versus 105 participants in the control group. The study was conducted using 3 telephone interviews and a 24-hour diet recall technique. The results supported a link between acne and skim milk consumption. Again, although the studied relied on participant self-reports of diet and followed a case-control design, a possible association was suspected but not validated.3 A longitudinal, questionnaire-based population study performed by Ulvestad et al4 included 2489 patients. This study further evaluated recall of dairy product consumption at 15 to 16 years of age and then 3 years later acne severity was self-assessed and reported at 18 to 19 years of age. Overall, this evaluation indicated that a high intake of dairy products and acne in adolescence have been positively associated. However, it was another retrospective study with recall bias.4 In 2009 Melnick and Schmitz5 concluded that milk causes the body to elevate both insulin and insulinlike growth factor 1 levels. In another study by Melnick6 in 2011, a definitive link between increased insulin and insulinlike growth factor 1 signaling in promoting comedogenesis was reported. Given the few studies that show the potential link between dairy products and acne, this dairy-free diet can be considered as a diet recommendation for acne patients.
Atopic Dermatitis
A Cochrane review conducted in 2012 regarding dietary supplements as a treatment of atopic dermatitis evaluated randomized controlled trials (N=596). Supplementation with vitamin D, fish oil, olive oil, zinc sulfate, selenium, vitamin E, pyridoxine, sea buckthorn seed oil, hempseed oil, sunflower oil (linoleic acid), and docosahexaenoic acid were evaluated among all the studies reviewed for atopic dermatitis.7 Bronsnick et al8 conducted a review of evidence supporting vitamin supplementation and atopic dermatitis, and for the most part determined that the studies had insufficient evidence. The only positive correlation was noted with prebiotics and probiotics in another Cochrane review in 2013, which evaluated 4 studies with 1428 infants showing prebiotic supplementation reduced atopic dermatitis.9 In 2014 Panduru et al10 evaluated 16 studies in a meta-analysis that showed how probiotics were possibly beneficial in both general and high-risk atopic populations. Specifically, a subgroup analysis showed that Lactobacillus and Lactobacillus with Bifidobacterium also can be protective against atopic dermatitis.10 Lastly, diet avoidance in pregnancy or during lactation in infants up to 18 months of age did not have any effect on improving the infant’s atopic dermatitis based on a 2012 Cochrane review that included 952 participants.11
Conclusion
Overall, there are some benefits to dietary restrictions and supplementation as indicated by the studies reviewed here; however, the extent to which these changes contribute to disease manifestation has only been linked, not definitively proven. Randomized controlled trials with large sample sizes, double-blind studies, and appropriately controlled studies with comparative patient populations are difficult to obtain, as diet cannot be completely restrictive for every patient. Patients should be provided with the latest data supporting a possible link between dairy consumption and acne production as well as prebiotics or probiotics during pregnancy and at infancy to reduce the risk for atopic dermatitis with the caveat of association. That said, future studies might prove that dietary and environmental alterations may prevent disease progression or appearance far more than previously assumed.
- Fulton JE Jr, Plewaig G, Kligman AM. Effect of chocolate on acne vulgaris. JAMA. 1969;210: 2071-2074.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. High school dietary diary intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- LaRosa CL, Quach KA, Koons K, et al. Consumption of dairy in teenagers with and without acne. J Am Acad Dermatol. 2016;75:318-322.
- Ulvestad M, Bjertness E, Dalgard F, et al. Acne and dairy products in adolescence: results from a Norwegian longitudinal study [published online ahead of print July 16, 2016]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.13835.
- Melnick BC, Schmitz G. Role of insulin, insulin like growth factor 1, hyperglycemic food and milk consumption in the pathogenesis of acne vulgaris. Exp Dermatol. 2009;18:833-841.
- Melnick BC. Evidence for acne-promoting effect of milk and other insulinotropic dairy products. Nestle Nutr Worksop
Ser Pediatr Program. 2011;67:131-145. - Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;2:CD005205.
- Bronsnick T, Murzaku EC, Rao BK. Diet in dermatology: part i. atopic dermatitis, acne, and nonmelanoma skin cancer [published online November 15, 2014]. J Am Acad Dermatol. 2014;71:1039.e1-1039.e12.
- Osborn DA, Sinn JKH. Prebiotics in infants for prevention of allergy. Cochrane Database Syst Rev. 2013;2:CD006474.
- Panduru M, Panduru NM, Saˇlaˇvaˇstru CM, et al. Probiotics and primary prevention of atopic dermatitis: a meta-analysis of randomized controlled studies [published online April 4, 2014]. J Eur Acad Dermatol Venereol. 2015;29:232-242.
- Kramer MS, Kakuma R. Maternal dietary antigen avoidance during pregnancy or lactation, or both, for preventing or treating atopic disease in the child. Cochrane Database Syst Rev. 2012;9:CD000133.
- Fulton JE Jr, Plewaig G, Kligman AM. Effect of chocolate on acne vulgaris. JAMA. 1969;210: 2071-2074.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. High school dietary diary intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- LaRosa CL, Quach KA, Koons K, et al. Consumption of dairy in teenagers with and without acne. J Am Acad Dermatol. 2016;75:318-322.
- Ulvestad M, Bjertness E, Dalgard F, et al. Acne and dairy products in adolescence: results from a Norwegian longitudinal study [published online ahead of print July 16, 2016]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.13835.
- Melnick BC, Schmitz G. Role of insulin, insulin like growth factor 1, hyperglycemic food and milk consumption in the pathogenesis of acne vulgaris. Exp Dermatol. 2009;18:833-841.
- Melnick BC. Evidence for acne-promoting effect of milk and other insulinotropic dairy products. Nestle Nutr Worksop
Ser Pediatr Program. 2011;67:131-145. - Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;2:CD005205.
- Bronsnick T, Murzaku EC, Rao BK. Diet in dermatology: part i. atopic dermatitis, acne, and nonmelanoma skin cancer [published online November 15, 2014]. J Am Acad Dermatol. 2014;71:1039.e1-1039.e12.
- Osborn DA, Sinn JKH. Prebiotics in infants for prevention of allergy. Cochrane Database Syst Rev. 2013;2:CD006474.
- Panduru M, Panduru NM, Saˇlaˇvaˇstru CM, et al. Probiotics and primary prevention of atopic dermatitis: a meta-analysis of randomized controlled studies [published online April 4, 2014]. J Eur Acad Dermatol Venereol. 2015;29:232-242.
- Kramer MS, Kakuma R. Maternal dietary antigen avoidance during pregnancy or lactation, or both, for preventing or treating atopic disease in the child. Cochrane Database Syst Rev. 2012;9:CD000133.
EpiPen cost increases far exceed overall medical inflation
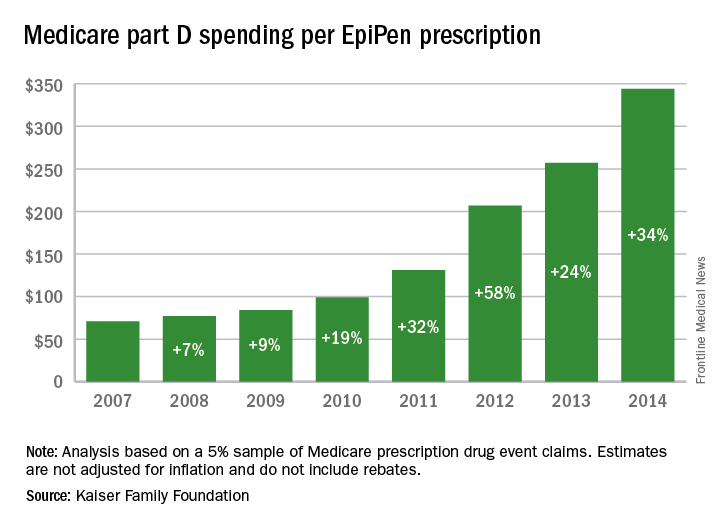
Total Medicare part D spending on EpiPen auto-injectors rose from $7.0 million in 2007 to $87.9 million in 2014 – an increase of 1,151%, according to an analysis released Sept. 20 by the Kaiser Family Foundation.
The number of EpiPen users also increased over that time, however, bringing with it a commensurate 159% rise in the number of prescriptions. Those two trends took the average cost of a single EpiPen prescription from $71 in 2007 to $344 in 2014, the Kaiser analysis showed.
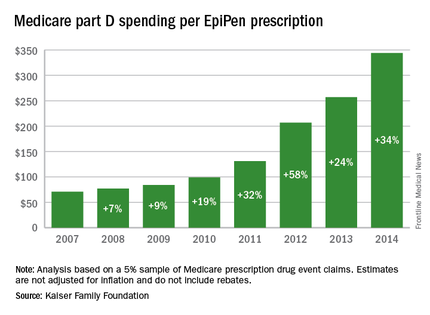
That increase in cost per prescription did not fail to at least double overall medical care price inflation for each year from 2008 to 2014. In 2008, when the two trends were closest together, the EpiPen cost per prescription rose 7.4% from the year before, compared with 3.7% for overall medical spending. In 2014, Medicare part D’s cost for an EpiPen prescription rose 34% from the year before, which was 14 times higher than the 2.4% increase in total medical spending, Kaiser noted.
The analysis was based on a 5% sample of Medicare prescription drug event claims and included beneficiaries who had a least 1 month of part D coverage and one EpiPen prescription during the year. Estimates are not adjusted for inflation and do not include any possible manufacturer discounts, Kaiser said.
Total Medicare part D spending on EpiPen auto-injectors rose from $7.0 million in 2007 to $87.9 million in 2014 – an increase of 1,151%, according to an analysis released Sept. 20 by the Kaiser Family Foundation.
The number of EpiPen users also increased over that time, however, bringing with it a commensurate 159% rise in the number of prescriptions. Those two trends took the average cost of a single EpiPen prescription from $71 in 2007 to $344 in 2014, the Kaiser analysis showed.

That increase in cost per prescription did not fail to at least double overall medical care price inflation for each year from 2008 to 2014. In 2008, when the two trends were closest together, the EpiPen cost per prescription rose 7.4% from the year before, compared with 3.7% for overall medical spending. In 2014, Medicare part D’s cost for an EpiPen prescription rose 34% from the year before, which was 14 times higher than the 2.4% increase in total medical spending, Kaiser noted.
The analysis was based on a 5% sample of Medicare prescription drug event claims and included beneficiaries who had a least 1 month of part D coverage and one EpiPen prescription during the year. Estimates are not adjusted for inflation and do not include any possible manufacturer discounts, Kaiser said.
Total Medicare part D spending on EpiPen auto-injectors rose from $7.0 million in 2007 to $87.9 million in 2014 – an increase of 1,151%, according to an analysis released Sept. 20 by the Kaiser Family Foundation.
The number of EpiPen users also increased over that time, however, bringing with it a commensurate 159% rise in the number of prescriptions. Those two trends took the average cost of a single EpiPen prescription from $71 in 2007 to $344 in 2014, the Kaiser analysis showed.

That increase in cost per prescription did not fail to at least double overall medical care price inflation for each year from 2008 to 2014. In 2008, when the two trends were closest together, the EpiPen cost per prescription rose 7.4% from the year before, compared with 3.7% for overall medical spending. In 2014, Medicare part D’s cost for an EpiPen prescription rose 34% from the year before, which was 14 times higher than the 2.4% increase in total medical spending, Kaiser noted.
The analysis was based on a 5% sample of Medicare prescription drug event claims and included beneficiaries who had a least 1 month of part D coverage and one EpiPen prescription during the year. Estimates are not adjusted for inflation and do not include any possible manufacturer discounts, Kaiser said.
Drugs in the pipeline hold promise for atopic dermatitis
NEWPORT BEACH, CALIF. – In the clinical opinion of Kelly M. Cordoro, MD, anyone who cares for patients with severe atopic dermatitis understands the sense of misery that can ensue.
“Atopic dermatitis patients don’t sleep well; they have poor school and work performance,” she said at the annual meeting of the Pacific Dermatologic Association. “They have absences. They’re unable to play; they can’t exercise. This leads to social disability; isolation from peers, and it goes on and on. The patients are miserable, the whole family is miserable, and we as physicians trying to sort out how to optimally treat them are miserable trying to figure out what the next best step is.”
The good news is, several drugs in the pipeline hold promise for atopic dermatitis patients, thanks largely to emerging data on its pathophysiology. In addition, mechanisms of itch, which are not yet fully understood, are also being unraveled. “It’s exciting to read the literature about the interaction of the skin, the immune system, and the nervous system,” said Dr. Cordoro, a pediatric dermatologist at the University of California, San Francisco. “Many of the mediators of itch are being identified. That has allowed for the development of targeted therapies against many of them.”
One of the promising treatments on the horizon for atopic dermatitis patients is phosphodiesterase-4 (PDE4) inhibitors. PDE-4 is a predominant cAMP-degrading enzyme in keratinocytes and inflammatory cells. “It’s really a candidate for not only atopic dermatitis but for psoriasis,” she said.
Oral PDE-4 inhibitors are already approved for psoriasis. Apremilast (Otezla) was approved by the Food and Drug Administration in 2014 for psoriasis and psoriatic arthritis, and a phase II trial of topical apremilast in adults with AD has been completed and the results are pending. “I look forward to seeing if this can help our patients,” Dr. Cordoro said.
Another promising agent for atopic dermatitis is 2% crisaborole topical ointment, a boron-based PDE-4 inhibitor developed by Anacor Pharmaceuticals. Dr. Cordoro described this compound as an anti-inflammatory agent that modifies inflammation by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signaling pathways.
“Crisaborole has shown promising results from four clinical studies in patients 2 years of age and older, with notable improvements in all atopic dermatitis parameters,” she said (J Am Acad Dermatol. 2016 Sept;75[3]:494-503.e). The FDA review of crisaborole for the treatment of mild to moderate atopic dermatitis in children and adults is currently underway, and is expected to be completed by early January 2017.
An especially favorable drug in development for atopic dermatitis is dupilumab, a fully human monoclonal antibody that targets the interleukin (IL)–4 receptor, and inhibits IL-4 and IL-13 signaling. A published trial of its use in adults with moderate to severe atopic dermatitis showed rapid improvements in all atopic dermatitis clinical indices (N Engl J Med. 2014;371[2]:130-9). The most common side effects were headache and pharyngitis, and skin infections and flares were more common in the placebo group, compared with the treatment group.
Dupilumab “has the potential to shift the treatment landscape of atopic dermatitis, because it can actually change the molecular signature of dermatitic skin, reducing inflammatory and proliferative markers,” Dr. Cordoro said. There are ongoing trials in adult and pediatric populations and FDA approval is anticipated in early 2017.
Published reports also suggest a role for the IL-12/23 pathway inhibitor ustekinumab in severe refractory adult atopic dermatitis (Int J Dermatol. 2012;51[1]:115-6 and JAAD Case Reports 2015;1:25-6). Additional studies are ongoing.
Therapies for itch that have completed phase II trials include the anti-IL31R monoclonal antibody nemolizumab (CIM331); the neurokinin-1R antagonist VLY-686; and the neurokinin-1R antagonist aprepitant gel.
Dr. Cordoro disclosed that she is a consultant for Celgene Corporation, Valeant, and Anacor Pharmaceuticals.
NEWPORT BEACH, CALIF. – In the clinical opinion of Kelly M. Cordoro, MD, anyone who cares for patients with severe atopic dermatitis understands the sense of misery that can ensue.
“Atopic dermatitis patients don’t sleep well; they have poor school and work performance,” she said at the annual meeting of the Pacific Dermatologic Association. “They have absences. They’re unable to play; they can’t exercise. This leads to social disability; isolation from peers, and it goes on and on. The patients are miserable, the whole family is miserable, and we as physicians trying to sort out how to optimally treat them are miserable trying to figure out what the next best step is.”
The good news is, several drugs in the pipeline hold promise for atopic dermatitis patients, thanks largely to emerging data on its pathophysiology. In addition, mechanisms of itch, which are not yet fully understood, are also being unraveled. “It’s exciting to read the literature about the interaction of the skin, the immune system, and the nervous system,” said Dr. Cordoro, a pediatric dermatologist at the University of California, San Francisco. “Many of the mediators of itch are being identified. That has allowed for the development of targeted therapies against many of them.”
One of the promising treatments on the horizon for atopic dermatitis patients is phosphodiesterase-4 (PDE4) inhibitors. PDE-4 is a predominant cAMP-degrading enzyme in keratinocytes and inflammatory cells. “It’s really a candidate for not only atopic dermatitis but for psoriasis,” she said.
Oral PDE-4 inhibitors are already approved for psoriasis. Apremilast (Otezla) was approved by the Food and Drug Administration in 2014 for psoriasis and psoriatic arthritis, and a phase II trial of topical apremilast in adults with AD has been completed and the results are pending. “I look forward to seeing if this can help our patients,” Dr. Cordoro said.
Another promising agent for atopic dermatitis is 2% crisaborole topical ointment, a boron-based PDE-4 inhibitor developed by Anacor Pharmaceuticals. Dr. Cordoro described this compound as an anti-inflammatory agent that modifies inflammation by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signaling pathways.
“Crisaborole has shown promising results from four clinical studies in patients 2 years of age and older, with notable improvements in all atopic dermatitis parameters,” she said (J Am Acad Dermatol. 2016 Sept;75[3]:494-503.e). The FDA review of crisaborole for the treatment of mild to moderate atopic dermatitis in children and adults is currently underway, and is expected to be completed by early January 2017.
An especially favorable drug in development for atopic dermatitis is dupilumab, a fully human monoclonal antibody that targets the interleukin (IL)–4 receptor, and inhibits IL-4 and IL-13 signaling. A published trial of its use in adults with moderate to severe atopic dermatitis showed rapid improvements in all atopic dermatitis clinical indices (N Engl J Med. 2014;371[2]:130-9). The most common side effects were headache and pharyngitis, and skin infections and flares were more common in the placebo group, compared with the treatment group.
Dupilumab “has the potential to shift the treatment landscape of atopic dermatitis, because it can actually change the molecular signature of dermatitic skin, reducing inflammatory and proliferative markers,” Dr. Cordoro said. There are ongoing trials in adult and pediatric populations and FDA approval is anticipated in early 2017.
Published reports also suggest a role for the IL-12/23 pathway inhibitor ustekinumab in severe refractory adult atopic dermatitis (Int J Dermatol. 2012;51[1]:115-6 and JAAD Case Reports 2015;1:25-6). Additional studies are ongoing.
Therapies for itch that have completed phase II trials include the anti-IL31R monoclonal antibody nemolizumab (CIM331); the neurokinin-1R antagonist VLY-686; and the neurokinin-1R antagonist aprepitant gel.
Dr. Cordoro disclosed that she is a consultant for Celgene Corporation, Valeant, and Anacor Pharmaceuticals.
NEWPORT BEACH, CALIF. – In the clinical opinion of Kelly M. Cordoro, MD, anyone who cares for patients with severe atopic dermatitis understands the sense of misery that can ensue.
“Atopic dermatitis patients don’t sleep well; they have poor school and work performance,” she said at the annual meeting of the Pacific Dermatologic Association. “They have absences. They’re unable to play; they can’t exercise. This leads to social disability; isolation from peers, and it goes on and on. The patients are miserable, the whole family is miserable, and we as physicians trying to sort out how to optimally treat them are miserable trying to figure out what the next best step is.”
The good news is, several drugs in the pipeline hold promise for atopic dermatitis patients, thanks largely to emerging data on its pathophysiology. In addition, mechanisms of itch, which are not yet fully understood, are also being unraveled. “It’s exciting to read the literature about the interaction of the skin, the immune system, and the nervous system,” said Dr. Cordoro, a pediatric dermatologist at the University of California, San Francisco. “Many of the mediators of itch are being identified. That has allowed for the development of targeted therapies against many of them.”
One of the promising treatments on the horizon for atopic dermatitis patients is phosphodiesterase-4 (PDE4) inhibitors. PDE-4 is a predominant cAMP-degrading enzyme in keratinocytes and inflammatory cells. “It’s really a candidate for not only atopic dermatitis but for psoriasis,” she said.
Oral PDE-4 inhibitors are already approved for psoriasis. Apremilast (Otezla) was approved by the Food and Drug Administration in 2014 for psoriasis and psoriatic arthritis, and a phase II trial of topical apremilast in adults with AD has been completed and the results are pending. “I look forward to seeing if this can help our patients,” Dr. Cordoro said.
Another promising agent for atopic dermatitis is 2% crisaborole topical ointment, a boron-based PDE-4 inhibitor developed by Anacor Pharmaceuticals. Dr. Cordoro described this compound as an anti-inflammatory agent that modifies inflammation by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signaling pathways.
“Crisaborole has shown promising results from four clinical studies in patients 2 years of age and older, with notable improvements in all atopic dermatitis parameters,” she said (J Am Acad Dermatol. 2016 Sept;75[3]:494-503.e). The FDA review of crisaborole for the treatment of mild to moderate atopic dermatitis in children and adults is currently underway, and is expected to be completed by early January 2017.
An especially favorable drug in development for atopic dermatitis is dupilumab, a fully human monoclonal antibody that targets the interleukin (IL)–4 receptor, and inhibits IL-4 and IL-13 signaling. A published trial of its use in adults with moderate to severe atopic dermatitis showed rapid improvements in all atopic dermatitis clinical indices (N Engl J Med. 2014;371[2]:130-9). The most common side effects were headache and pharyngitis, and skin infections and flares were more common in the placebo group, compared with the treatment group.
Dupilumab “has the potential to shift the treatment landscape of atopic dermatitis, because it can actually change the molecular signature of dermatitic skin, reducing inflammatory and proliferative markers,” Dr. Cordoro said. There are ongoing trials in adult and pediatric populations and FDA approval is anticipated in early 2017.
Published reports also suggest a role for the IL-12/23 pathway inhibitor ustekinumab in severe refractory adult atopic dermatitis (Int J Dermatol. 2012;51[1]:115-6 and JAAD Case Reports 2015;1:25-6). Additional studies are ongoing.
Therapies for itch that have completed phase II trials include the anti-IL31R monoclonal antibody nemolizumab (CIM331); the neurokinin-1R antagonist VLY-686; and the neurokinin-1R antagonist aprepitant gel.
Dr. Cordoro disclosed that she is a consultant for Celgene Corporation, Valeant, and Anacor Pharmaceuticals.
EXPERT ANALYSIS FROM PDA 2016
The Translational Revolution in Atopic Dermatitis, and How It Also Translates to Other Inflammatory Skin Diseases
Atopic dermatitis (AD) is the most common inflammatory skin disease in both adults and children.1 Unfortunately, the current treatment armamentarium is largely confined to topical calcineurin inhibitors, topical and systemic steroids, phototherapy, cyclosporine (not approved by the US Food and Drug Administration for AD), and other oral immunosuppressants.2 The availability of partially helpful and highly toxic treatments creates a huge unmet need for more effective and safer treatments, particularly for patients with moderate to severe AD who often require systemic approaches.
Recent extensive translational (bench top to bedside and back) investigations in skin of AD patients has shown that skin phenotype is characterized by increased T-cell infiltration and related inflammatory cytokines as well as epidermal abnormalities (eg, hyperplasia, aberrant differentiation).3 Clinical improvement of AD has been demonstrated with broad T-cell targeted therapeutics, such as cyclosporine and narrowband UVB, coupled with decreases of T-cell infiltrates and inflammatory gene products as well as improvement of the pathologic epidermal phenotype.4,5
In the past, AD was conceptualized as a T helper cell TH2 (acute disease)/TH1 (chronic disease) bipolar cytokine disorder.6 Acute lesions are characterized by high TH2, TH22, and some TH17 signals, with intensification of these axes and TH1 augmentation orchestrating the chronic phenotype.7 The identification of the inflammatory pathways underlying AD has led to the development and testing of more than 10 broad or targeted therapeutics (Table).8 Phase 1 and phase 2 studies of dupilumab (targeting IL-4Rα) have shown not only tremendous AD improvement (~70%) but also tissue reversal of the immune and barrier abnormalities, including inflammatory cytokines and epidermal hyperplasia.9-11 As a result, other TH2 axis inhibitors (anti–IL-13/tralokinumab, anti–IL-31RA/CIM 331) are now in clinical trials. The identification of IL-22 in AD lesions has prompted trials with an anti–IL-22 (ILV 094) and an IL-12/IL-23p40 (ustekinumab) inhibitor.12 For psoriasis, ustekinumab showed 75% improvement in approximately 70% of patients,13 but for AD, despite clear clinical and molecular effects, differences compared to placebo were not statistically significant,12 probably due to underdosing of the drug in an excessively immune-activated disease14 as well as allowing topical steroids in patients, which may minimize the differences in treatment effect between drug and placebo.

The developments seen in AD are now moving into other inflammatory skin diseases, particularly alopecia areata (AA), a T-cell–mediated disease that shares phenotypic similarities with AD and often is associated with it.15 There is a paucity of effective, remission-sustaining treatments of AA, particularly for patients with severe disease who rarely experience spontaneous hair regrowth and who have a limited response to topical interventions.16,17 Our clinical experience showed that successfully treating patients with concurrent AD and AA has led to hair regrowth. Inspired by these clinical observations and by results obtained in AD,9-12 studying AA skin showed an upregulation of not only the traditionally suspected culprit TH1 but also TH2 and TH9 axes, IL-23 cytokines, and phosphodiesterase 4.18 Subsequently, a pilot study of 3 patients with extensive AA treated with ustekinumab showed that all 3 patients not only experienced hair regrowth but also had a reduction in inflammatory markers in scalp lesions.19 Although these results are promising, AA is an immunologically complex disease and it is yet to be determined if therapeutically targeting 1 (eg, IL-4) rather than a wide array of cytokines can reverse disease phenotype. There are ongoing clinical trials directed at different pathogenic targets (eg, Jak inhibitors, IL-13 antagonist, IL-17 antagonist, phosphodiesterase 4 antagonist); some showed some efficacy in small studies.20,21
The finding of a commonly upregulated TH2 pathway in both AD and AA will pave the way for studies with TH2 antagonists in AA patients. Future targeted therapeutic studies will shed light on the pathogenic pathways of this devastating skin disease and answer the extensive unmet therapeutic need it presents.
- Czarnowicki T, Krueger JG, Guttman-Yassky E. Skin barrier and immune dysregulation in atopic dermatitis: an evolving story with important clinical implications. J Allergy Clin Immunol Pract. 2014;2:371-379; quiz 380-381.
- Roekevisch E, Spuls PI, Kuester D, et al. Efficacy and safety of systemic treatments for moderate-to-severe atopic dermatitis: a systematic review. J Allergy Clin Immunol. 2014;133:429-438.
- Guttman-Yassky E, Nograles KE, Krueger JG. Contrasting pathogenesis of atopic dermatitis and psoriasis—part
I: clinical and pathologic concepts. J Allergy Clin Immunol. 2011;127:1110-1118. - Khattri S, Shemer A, Rozenblit M, et al. Cyclosporine in patients with atopic dermatitis modulates activated inflammatory pathways and reverses epidermal pathology. J Allergy Clin Immunol. 2014;133:1626-1634.
- Tintle S, Shemer A, Suárez-Fariñas M, et al. Reversal of atopic dermatitis with narrow-band UVB phototherapy and biomarkers for therapeutic response [published online July 16, 2011]. J Allergy Clin Immunol. 2011;128:583-593.
- Eyerich K, Novak N. Immunology of atopic eczema: overcoming the Th1/Th2 paradigm. Allergy. 2013;68:974-982.
- Gittler JK, Shemer A, Suárez-Fariñas M, et al. Progressive activation of T(H)2/T(H)22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis [published online August 27, 2012]. J Allergy Clin Immunol. 2012;130:1344-1354.
- Noda S, Krueger JG, Guttman-Yassky E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J Allergy Clin Immunol. 2015;135:324-336.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Hamilton JD, Suárez-Fariñas M, Dhingra N, et al. Dupilumab improves the molecular signature in skin of patients with moderate-to-severe atopic dermatitis. J Allergy Clin Immunol. 2014;134:1293-1300.
- Hamilton J, Ren H, Weinstein SP, et al. Dupilumab improved all domains of Eczema Area and Severity Index (EASI) and 5-D pruritus scale in adults with atopic dermatitis in a phase 2 study. J Invest Dermatol. 2014;134:S104.
- Khattri S, Brunner PM, Garcet S, et al. Efficacy and safety of ustekinumab treatment in adults with moderate-to-severe atopic dermatitis [published online June 15, 2016]. Exp Dermatol. doi:10.1111/exd.13112.
- Griffiths CEM, Strober BE, van de Kerkhof P, et al. Comparison of ustekinumab and etanercept for moderate-to-severe psoriasis. N Engl J Med. 2010;362:118-128.
- Czarnowicki T, Malajian D, Shemer A, et al. Skin-homing and systemic T-cell subsets show higher activation in atopic dermatitis versus psoriasis. J Allergy Clin Immunol. 2015;136:208-211.
- Barahmani N, Schabath MB, Duvic M. History of atopy or autoimmunity increases risk of alopecia areata. J Am Acad Dermatol. 2009;61:581-591.
- Price VH, Hordinsky MK, Olsen EA, et al. Subcutaneous efalizumab is not effective in the treatment of alopecia areata. J Am Acad Dermatol. 2008;58:395-402.
- Alkhalifah A, Alsantali A, Wang E, et al. Alopecia areata update part II. treatment. J Am Acad Dermatol. 2010;62:191-202.
- Suárez-Fariñas M, Ungar B, Noda S, et al. Alopecia areata profiling shows TH1, TH2, and IL-23 cytokine activation without parallel TH17/TH22 skewing. J Allergy Clin Immunol. 2015;136:1277-1287.
- Guttman-Yassky E, Ungar B, Noda S, et al. Extensive alopecia areata is reversed by IL-12/IL-23p40 cytokine antagonism. J Allergy Clin Immunol. 2016;137:301-304.
- Xing LZ, Dai ZP, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043-1049.
- Castela E, Le Duff F, Butori C, et al. Effects of low-dose recombinant interleukin 2 to promote T-regulatory cells in alopecia areata. JAMA Dermatol. 2014;150:748-751.
Atopic dermatitis (AD) is the most common inflammatory skin disease in both adults and children.1 Unfortunately, the current treatment armamentarium is largely confined to topical calcineurin inhibitors, topical and systemic steroids, phototherapy, cyclosporine (not approved by the US Food and Drug Administration for AD), and other oral immunosuppressants.2 The availability of partially helpful and highly toxic treatments creates a huge unmet need for more effective and safer treatments, particularly for patients with moderate to severe AD who often require systemic approaches.
Recent extensive translational (bench top to bedside and back) investigations in skin of AD patients has shown that skin phenotype is characterized by increased T-cell infiltration and related inflammatory cytokines as well as epidermal abnormalities (eg, hyperplasia, aberrant differentiation).3 Clinical improvement of AD has been demonstrated with broad T-cell targeted therapeutics, such as cyclosporine and narrowband UVB, coupled with decreases of T-cell infiltrates and inflammatory gene products as well as improvement of the pathologic epidermal phenotype.4,5
In the past, AD was conceptualized as a T helper cell TH2 (acute disease)/TH1 (chronic disease) bipolar cytokine disorder.6 Acute lesions are characterized by high TH2, TH22, and some TH17 signals, with intensification of these axes and TH1 augmentation orchestrating the chronic phenotype.7 The identification of the inflammatory pathways underlying AD has led to the development and testing of more than 10 broad or targeted therapeutics (Table).8 Phase 1 and phase 2 studies of dupilumab (targeting IL-4Rα) have shown not only tremendous AD improvement (~70%) but also tissue reversal of the immune and barrier abnormalities, including inflammatory cytokines and epidermal hyperplasia.9-11 As a result, other TH2 axis inhibitors (anti–IL-13/tralokinumab, anti–IL-31RA/CIM 331) are now in clinical trials. The identification of IL-22 in AD lesions has prompted trials with an anti–IL-22 (ILV 094) and an IL-12/IL-23p40 (ustekinumab) inhibitor.12 For psoriasis, ustekinumab showed 75% improvement in approximately 70% of patients,13 but for AD, despite clear clinical and molecular effects, differences compared to placebo were not statistically significant,12 probably due to underdosing of the drug in an excessively immune-activated disease14 as well as allowing topical steroids in patients, which may minimize the differences in treatment effect between drug and placebo.

The developments seen in AD are now moving into other inflammatory skin diseases, particularly alopecia areata (AA), a T-cell–mediated disease that shares phenotypic similarities with AD and often is associated with it.15 There is a paucity of effective, remission-sustaining treatments of AA, particularly for patients with severe disease who rarely experience spontaneous hair regrowth and who have a limited response to topical interventions.16,17 Our clinical experience showed that successfully treating patients with concurrent AD and AA has led to hair regrowth. Inspired by these clinical observations and by results obtained in AD,9-12 studying AA skin showed an upregulation of not only the traditionally suspected culprit TH1 but also TH2 and TH9 axes, IL-23 cytokines, and phosphodiesterase 4.18 Subsequently, a pilot study of 3 patients with extensive AA treated with ustekinumab showed that all 3 patients not only experienced hair regrowth but also had a reduction in inflammatory markers in scalp lesions.19 Although these results are promising, AA is an immunologically complex disease and it is yet to be determined if therapeutically targeting 1 (eg, IL-4) rather than a wide array of cytokines can reverse disease phenotype. There are ongoing clinical trials directed at different pathogenic targets (eg, Jak inhibitors, IL-13 antagonist, IL-17 antagonist, phosphodiesterase 4 antagonist); some showed some efficacy in small studies.20,21
The finding of a commonly upregulated TH2 pathway in both AD and AA will pave the way for studies with TH2 antagonists in AA patients. Future targeted therapeutic studies will shed light on the pathogenic pathways of this devastating skin disease and answer the extensive unmet therapeutic need it presents.
Atopic dermatitis (AD) is the most common inflammatory skin disease in both adults and children.1 Unfortunately, the current treatment armamentarium is largely confined to topical calcineurin inhibitors, topical and systemic steroids, phototherapy, cyclosporine (not approved by the US Food and Drug Administration for AD), and other oral immunosuppressants.2 The availability of partially helpful and highly toxic treatments creates a huge unmet need for more effective and safer treatments, particularly for patients with moderate to severe AD who often require systemic approaches.
Recent extensive translational (bench top to bedside and back) investigations in skin of AD patients has shown that skin phenotype is characterized by increased T-cell infiltration and related inflammatory cytokines as well as epidermal abnormalities (eg, hyperplasia, aberrant differentiation).3 Clinical improvement of AD has been demonstrated with broad T-cell targeted therapeutics, such as cyclosporine and narrowband UVB, coupled with decreases of T-cell infiltrates and inflammatory gene products as well as improvement of the pathologic epidermal phenotype.4,5
In the past, AD was conceptualized as a T helper cell TH2 (acute disease)/TH1 (chronic disease) bipolar cytokine disorder.6 Acute lesions are characterized by high TH2, TH22, and some TH17 signals, with intensification of these axes and TH1 augmentation orchestrating the chronic phenotype.7 The identification of the inflammatory pathways underlying AD has led to the development and testing of more than 10 broad or targeted therapeutics (Table).8 Phase 1 and phase 2 studies of dupilumab (targeting IL-4Rα) have shown not only tremendous AD improvement (~70%) but also tissue reversal of the immune and barrier abnormalities, including inflammatory cytokines and epidermal hyperplasia.9-11 As a result, other TH2 axis inhibitors (anti–IL-13/tralokinumab, anti–IL-31RA/CIM 331) are now in clinical trials. The identification of IL-22 in AD lesions has prompted trials with an anti–IL-22 (ILV 094) and an IL-12/IL-23p40 (ustekinumab) inhibitor.12 For psoriasis, ustekinumab showed 75% improvement in approximately 70% of patients,13 but for AD, despite clear clinical and molecular effects, differences compared to placebo were not statistically significant,12 probably due to underdosing of the drug in an excessively immune-activated disease14 as well as allowing topical steroids in patients, which may minimize the differences in treatment effect between drug and placebo.

The developments seen in AD are now moving into other inflammatory skin diseases, particularly alopecia areata (AA), a T-cell–mediated disease that shares phenotypic similarities with AD and often is associated with it.15 There is a paucity of effective, remission-sustaining treatments of AA, particularly for patients with severe disease who rarely experience spontaneous hair regrowth and who have a limited response to topical interventions.16,17 Our clinical experience showed that successfully treating patients with concurrent AD and AA has led to hair regrowth. Inspired by these clinical observations and by results obtained in AD,9-12 studying AA skin showed an upregulation of not only the traditionally suspected culprit TH1 but also TH2 and TH9 axes, IL-23 cytokines, and phosphodiesterase 4.18 Subsequently, a pilot study of 3 patients with extensive AA treated with ustekinumab showed that all 3 patients not only experienced hair regrowth but also had a reduction in inflammatory markers in scalp lesions.19 Although these results are promising, AA is an immunologically complex disease and it is yet to be determined if therapeutically targeting 1 (eg, IL-4) rather than a wide array of cytokines can reverse disease phenotype. There are ongoing clinical trials directed at different pathogenic targets (eg, Jak inhibitors, IL-13 antagonist, IL-17 antagonist, phosphodiesterase 4 antagonist); some showed some efficacy in small studies.20,21
The finding of a commonly upregulated TH2 pathway in both AD and AA will pave the way for studies with TH2 antagonists in AA patients. Future targeted therapeutic studies will shed light on the pathogenic pathways of this devastating skin disease and answer the extensive unmet therapeutic need it presents.
- Czarnowicki T, Krueger JG, Guttman-Yassky E. Skin barrier and immune dysregulation in atopic dermatitis: an evolving story with important clinical implications. J Allergy Clin Immunol Pract. 2014;2:371-379; quiz 380-381.
- Roekevisch E, Spuls PI, Kuester D, et al. Efficacy and safety of systemic treatments for moderate-to-severe atopic dermatitis: a systematic review. J Allergy Clin Immunol. 2014;133:429-438.
- Guttman-Yassky E, Nograles KE, Krueger JG. Contrasting pathogenesis of atopic dermatitis and psoriasis—part
I: clinical and pathologic concepts. J Allergy Clin Immunol. 2011;127:1110-1118. - Khattri S, Shemer A, Rozenblit M, et al. Cyclosporine in patients with atopic dermatitis modulates activated inflammatory pathways and reverses epidermal pathology. J Allergy Clin Immunol. 2014;133:1626-1634.
- Tintle S, Shemer A, Suárez-Fariñas M, et al. Reversal of atopic dermatitis with narrow-band UVB phototherapy and biomarkers for therapeutic response [published online July 16, 2011]. J Allergy Clin Immunol. 2011;128:583-593.
- Eyerich K, Novak N. Immunology of atopic eczema: overcoming the Th1/Th2 paradigm. Allergy. 2013;68:974-982.
- Gittler JK, Shemer A, Suárez-Fariñas M, et al. Progressive activation of T(H)2/T(H)22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis [published online August 27, 2012]. J Allergy Clin Immunol. 2012;130:1344-1354.
- Noda S, Krueger JG, Guttman-Yassky E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J Allergy Clin Immunol. 2015;135:324-336.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Hamilton JD, Suárez-Fariñas M, Dhingra N, et al. Dupilumab improves the molecular signature in skin of patients with moderate-to-severe atopic dermatitis. J Allergy Clin Immunol. 2014;134:1293-1300.
- Hamilton J, Ren H, Weinstein SP, et al. Dupilumab improved all domains of Eczema Area and Severity Index (EASI) and 5-D pruritus scale in adults with atopic dermatitis in a phase 2 study. J Invest Dermatol. 2014;134:S104.
- Khattri S, Brunner PM, Garcet S, et al. Efficacy and safety of ustekinumab treatment in adults with moderate-to-severe atopic dermatitis [published online June 15, 2016]. Exp Dermatol. doi:10.1111/exd.13112.
- Griffiths CEM, Strober BE, van de Kerkhof P, et al. Comparison of ustekinumab and etanercept for moderate-to-severe psoriasis. N Engl J Med. 2010;362:118-128.
- Czarnowicki T, Malajian D, Shemer A, et al. Skin-homing and systemic T-cell subsets show higher activation in atopic dermatitis versus psoriasis. J Allergy Clin Immunol. 2015;136:208-211.
- Barahmani N, Schabath MB, Duvic M. History of atopy or autoimmunity increases risk of alopecia areata. J Am Acad Dermatol. 2009;61:581-591.
- Price VH, Hordinsky MK, Olsen EA, et al. Subcutaneous efalizumab is not effective in the treatment of alopecia areata. J Am Acad Dermatol. 2008;58:395-402.
- Alkhalifah A, Alsantali A, Wang E, et al. Alopecia areata update part II. treatment. J Am Acad Dermatol. 2010;62:191-202.
- Suárez-Fariñas M, Ungar B, Noda S, et al. Alopecia areata profiling shows TH1, TH2, and IL-23 cytokine activation without parallel TH17/TH22 skewing. J Allergy Clin Immunol. 2015;136:1277-1287.
- Guttman-Yassky E, Ungar B, Noda S, et al. Extensive alopecia areata is reversed by IL-12/IL-23p40 cytokine antagonism. J Allergy Clin Immunol. 2016;137:301-304.
- Xing LZ, Dai ZP, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043-1049.
- Castela E, Le Duff F, Butori C, et al. Effects of low-dose recombinant interleukin 2 to promote T-regulatory cells in alopecia areata. JAMA Dermatol. 2014;150:748-751.
- Czarnowicki T, Krueger JG, Guttman-Yassky E. Skin barrier and immune dysregulation in atopic dermatitis: an evolving story with important clinical implications. J Allergy Clin Immunol Pract. 2014;2:371-379; quiz 380-381.
- Roekevisch E, Spuls PI, Kuester D, et al. Efficacy and safety of systemic treatments for moderate-to-severe atopic dermatitis: a systematic review. J Allergy Clin Immunol. 2014;133:429-438.
- Guttman-Yassky E, Nograles KE, Krueger JG. Contrasting pathogenesis of atopic dermatitis and psoriasis—part
I: clinical and pathologic concepts. J Allergy Clin Immunol. 2011;127:1110-1118. - Khattri S, Shemer A, Rozenblit M, et al. Cyclosporine in patients with atopic dermatitis modulates activated inflammatory pathways and reverses epidermal pathology. J Allergy Clin Immunol. 2014;133:1626-1634.
- Tintle S, Shemer A, Suárez-Fariñas M, et al. Reversal of atopic dermatitis with narrow-band UVB phototherapy and biomarkers for therapeutic response [published online July 16, 2011]. J Allergy Clin Immunol. 2011;128:583-593.
- Eyerich K, Novak N. Immunology of atopic eczema: overcoming the Th1/Th2 paradigm. Allergy. 2013;68:974-982.
- Gittler JK, Shemer A, Suárez-Fariñas M, et al. Progressive activation of T(H)2/T(H)22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis [published online August 27, 2012]. J Allergy Clin Immunol. 2012;130:1344-1354.
- Noda S, Krueger JG, Guttman-Yassky E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J Allergy Clin Immunol. 2015;135:324-336.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Hamilton JD, Suárez-Fariñas M, Dhingra N, et al. Dupilumab improves the molecular signature in skin of patients with moderate-to-severe atopic dermatitis. J Allergy Clin Immunol. 2014;134:1293-1300.
- Hamilton J, Ren H, Weinstein SP, et al. Dupilumab improved all domains of Eczema Area and Severity Index (EASI) and 5-D pruritus scale in adults with atopic dermatitis in a phase 2 study. J Invest Dermatol. 2014;134:S104.
- Khattri S, Brunner PM, Garcet S, et al. Efficacy and safety of ustekinumab treatment in adults with moderate-to-severe atopic dermatitis [published online June 15, 2016]. Exp Dermatol. doi:10.1111/exd.13112.
- Griffiths CEM, Strober BE, van de Kerkhof P, et al. Comparison of ustekinumab and etanercept for moderate-to-severe psoriasis. N Engl J Med. 2010;362:118-128.
- Czarnowicki T, Malajian D, Shemer A, et al. Skin-homing and systemic T-cell subsets show higher activation in atopic dermatitis versus psoriasis. J Allergy Clin Immunol. 2015;136:208-211.
- Barahmani N, Schabath MB, Duvic M. History of atopy or autoimmunity increases risk of alopecia areata. J Am Acad Dermatol. 2009;61:581-591.
- Price VH, Hordinsky MK, Olsen EA, et al. Subcutaneous efalizumab is not effective in the treatment of alopecia areata. J Am Acad Dermatol. 2008;58:395-402.
- Alkhalifah A, Alsantali A, Wang E, et al. Alopecia areata update part II. treatment. J Am Acad Dermatol. 2010;62:191-202.
- Suárez-Fariñas M, Ungar B, Noda S, et al. Alopecia areata profiling shows TH1, TH2, and IL-23 cytokine activation without parallel TH17/TH22 skewing. J Allergy Clin Immunol. 2015;136:1277-1287.
- Guttman-Yassky E, Ungar B, Noda S, et al. Extensive alopecia areata is reversed by IL-12/IL-23p40 cytokine antagonism. J Allergy Clin Immunol. 2016;137:301-304.
- Xing LZ, Dai ZP, Jabbari A, et al. Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat Med. 2014;20:1043-1049.
- Castela E, Le Duff F, Butori C, et al. Effects of low-dose recombinant interleukin 2 to promote T-regulatory cells in alopecia areata. JAMA Dermatol. 2014;150:748-751.
Nonpharmacologic AD therapy: Strongest evidence supports moisturizers
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2016
Anastrozole-Induced Subacute Cutaneous Lupus Erythematosus
Drug-induced subacute cutaneous lupus erythematosus (DI-SCLE) was first described in 1985 in 5 patients who had been taking hydrochlorothiazide.1 The skin lesions in these patients were identical to those seen in idiopathic subacute cutaneous lupus erythematosus (SCLE) and were accompanied by the same autoantibodies (anti-Ro/Sjögren syndrome antigen A [SS-A] and anti-La/Sjögren syndrome antigen B [SS-B]) and HLA type (HLA-DR2/DR3) that are known to be associated with idiopathic SCLE. The skin lesions of SCLE in these 5 patients resolved spontaneously after discontinuing hydrochlorothiazide; however, anti-Ro/SS-A antibodies persisted in all except 1 patient.1 Over the last decade, an increasing number of drugs from different classes have been implicated to be associated with DI-SCLE. Since the concept of DI-SCLE was introduced, it has been reported to look identical to idiopathic SCLE, both clinically and histopathologically; however, one report suggested that the 2 entities can be distinguished based on clinical variations.2 In general, patients with DI-SCLE develop the same anti-Ro antibodies as seen in idiopathic SCLE. In addition, although the rash in DI-SCLE typically resolves with withdrawal of the offending drug, the antibodies tend to persist. Herein, we report a case of a patient being treated with an aromatase inhibitor who presented with clinical, serologic, and histopathologic evidence of DI-SCLE.
Case Report
A 69-year-old woman diagnosed with breast cancer 4 years prior to her presentation to dermatology initially underwent a lumpectomy and radiation treatment. She was subsequently started on anastrozole 2 years later. After 16 months of treatment with anastrozole, she developed an erythematous scaly rash on sun-exposed areas of the skin. The patient was seen by an outside dermatologist who treated her for a patient-perceived drug rash based on biopsy results that simply demonstrated interface dermatitis. She was treated with both topical and oral steroids with little improvement and therefore presented to our office approximately 6 months after starting treatment seeking a second opinion.
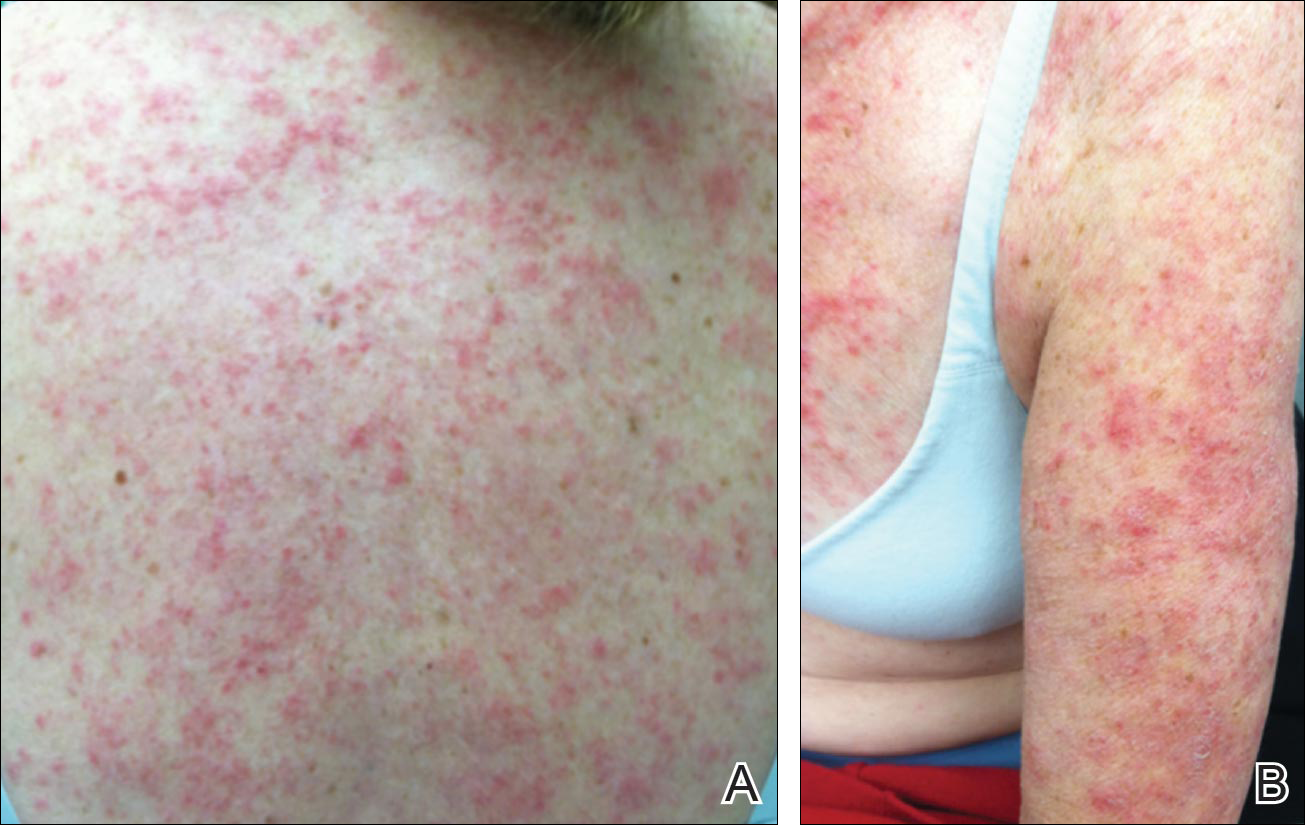
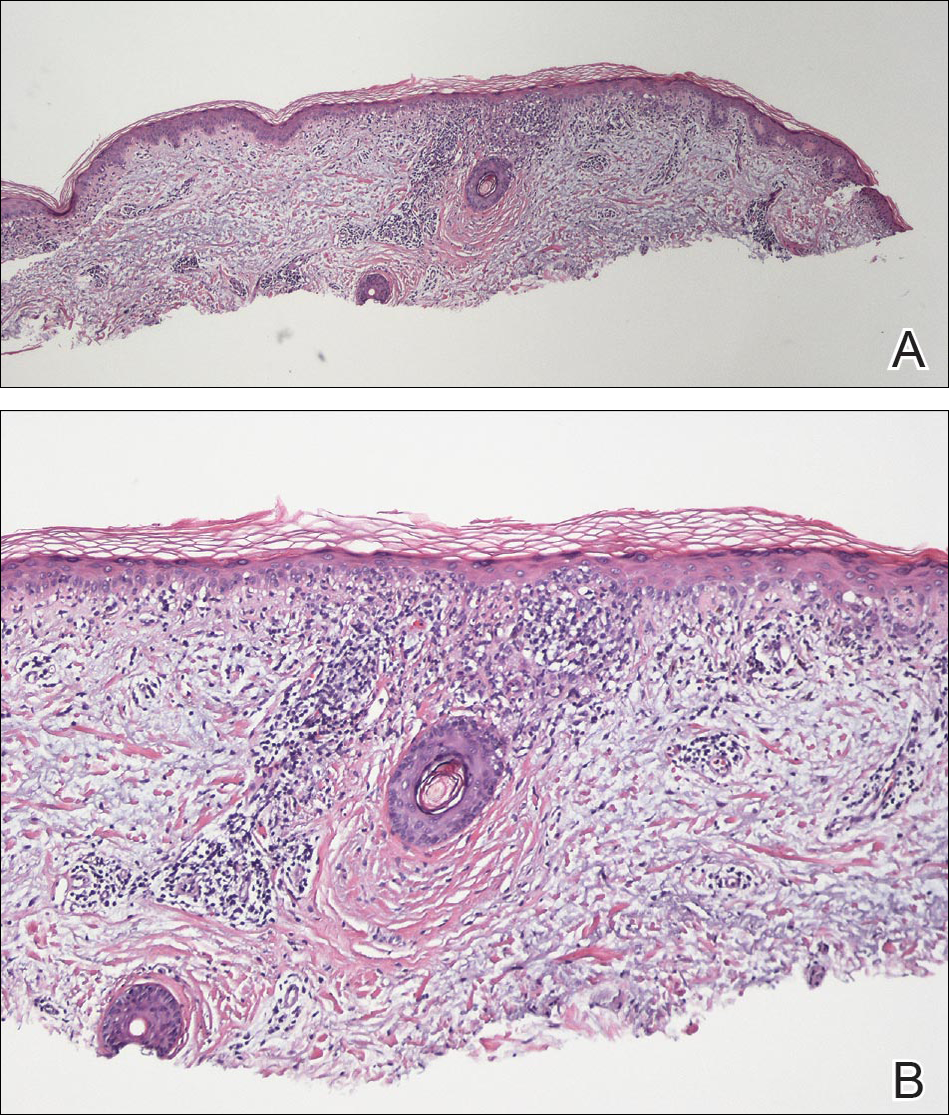
Physical examination revealed numerous erythematous scaly papules and plaques in a photodistributed pattern on the chest, back, legs, and arms (Figure 1). On further questioning, the patient noted that the rash became worse when she was at the beach or playing tennis outside as well as under indoor lights. A repeat biopsy was performed, revealing interface and perivascular dermatitis with an infiltrate composed of lymphocytes, histiocytes, and scattered pigment-laden macrophages (Figure 2). Given the appearance and distribution of the rash as well as the clinical scenario, drug-induced lupus was suspected. Anastrozole was the only medication being taken. Laboratory evaluation was performed and was negative for antinuclear antibodies, antihistone antibodies, and anti-La/SS-B antibodies but was positive for anti-Ro/SS-A antibodies (>8.0 U [reference range, <1.0 U]). Based on these findings, anastrozole-induced SCLE was the most likely explanation for this presentation. The patient was started on a sun-protective regimen (ie, wide-brimmed hat, daily sunscreen) and anastrozole was discontinued by her oncologist; the combination led to moderate improvement in symptoms. One week later, oral hydroxychloroquine 200 mg twice daily was started, which led to notable improvement (Figure 3). The patient was seen for 2 additional follow-up visits, each time with sustained resolution of the rash. The hydroxychloroquine was then stopped at her last visit 3 months after diagnosis. The patient was subsequently lost to follow-up.
Comment
Presentation of SCLE
Subacute cutaneous lupus erythematosus is a form of lupus erythematosus characterized by nonscarring, annular, scaly, erythematous plaques that occur on sun-exposed skin. The lesions are classically distributed on the upper back, chest, dorsal arms, and lateral neck but also can be found in other locations.3,4 Subacute cutaneous lupus erythematosus may be idiopathic; may occur in patients with systemic lupus erythematosus, Sjögren syndrome, or deficiency of the second component of complement (C2d); or may be drug induced.5 On histology SCLE presents as a lichenoid tissue reaction with focal vacuolization of the epidermal basal layer and perivascular lymphocytic infiltrate. On direct immunofluorescence, both idiopathic and drug-induced SCLE present with granular deposition of IgM, IgG, and C3 in a bandlike array at the dermoepidermal junction and circulating anti-Ro/SS-A antibodies. Therefore, histopathologically and immunologically, DI-SCLE is indistinguishable from idiopathic cases.6
Differential Diagnosis
It was previously thought that the clinical presentation of DI-SCLE and idiopathic SCLE were indistinguishable; however, Marzano et al2 described remarkable differences in the cutaneous manifestations of the 2 diseases. Drug-induced SCLE lesions are more widespread, occur more frequently on the legs, and may be bullous or erythema multiforme–like versus the idiopathic lesions, which tend to be more concentrated on the upper body and classically present as scaly erythematous plaques. Additionally, malar rash and vasculitic lesions, such as purpura and necrotic-ulcerative lesions, are seen more often in DI-SCLE.
Drug-induced systemic lupus erythematosus (DI-SLE) is a lupuslike syndrome that can be differentiated from DI-SCLE by virtue of its clinical and serological presentation. It differs from DI-SCLE in that DI-SLE typically does not present with skin symptoms; rather, systemic symptoms such as fever, weight loss, arthralgia, polyarthritis, pericarditis, and pleuritis are more commonly seen. Additionally, it has been associated with antihistone antibodies.4 More than 80 drugs have been reported to cause DI-SLE, including procainamide, hydralazine, and quinidine.7
To be classified as either DI-SCLE or DI-SLE, symptoms need to present after administration of the triggering drug and must resolve after the drug is discontinued.7 The drugs most commonly associated with DI-SCLE are thiazides, calcium channel blockers, tumor necrosis factor α inhibitors, angiotensin-converting enzyme inhibitors, and terbinafine, with few cases citing anastrozole as the inciting agent.4,6,8,9 The incubation period for DI-SCLE varies substantially. Thiazide diuretics and calcium channel blockers typically have the longest incubation period, ranging from 6 months to 5 years for thiazides,1,6,10,11 while calcium channel blockers have an average incubation period of 3 years.12 Drug-induced SCLE associated with antifungals, however, usually is much more rapid in onset; the incubation period on average is 5 weeks for terbinafine and 2 weeks for griseofulvin.13-15
Antiestrogen Drugs and SCLE
Anastrozole, the inciting agent in our case, is a third-generation, selective, nonsteroidal, aromatase inhibitor with no progestogenic, androgenic, or estrogenic activity. Anastrozole, when taken at its recommended dosage of 1 mg daily, will suppress estradiol. It is used as an adjuvant treatment of estrogen-sensitive breast cancer in postmenopausal women. In contrast to a prior case of DI-SCLE secondary to anastrozole in which the incubation period was approximately 1 month,8 our patient had an incubation period of approximately 16 months. Tamoxifen, another antiestrogen drug, also has been associated with DI-SCLE.9 In cases of tamoxifen-induced SCLE, the incubation period was several years, which is more similar to our patient. Although these drugs do not have the same mechanism of action, they both have antiestrogen properties.9 A systemic review of DI-SCLE reported that incubation periods between drug exposure and appearance of DI-SCLE varied greatly and were drug class dependent. It is possible that reactions associated with antiestrogen medications have a delayed presentation; however, given there are limited cases of anastrozole-induced DI-SCLE, we cannot make a clear statement on incubation periods.6
Reports of DI-SCLE caused by antiestrogen drugs are particularly interesting because sex hormones in relation to lupus disease activity have been the subject of debate for decades. Women are considerably more likely to develop autoimmune diseases than men, suggesting that steroid hormones, especially estrogen and progesterone, influence the immune system.16 Estrogen actions are proinflammatory, while the actions of progesterone, androgens, and glucocorticoids are anti-inflammatory.17 Studies in women with lupus revealed an increased rate of mild- to moderate-intensity disease flares associated with estrogen-containing hormone replace-ment therapy.18-20
Over the years, several antiestrogen therapies have been used in murine models, which showed remarkable clinical improvement in the course of SLE. The precise mechanisms involved in disease immunomodulation by these therapies have not been elucidated.21-23 It is thought that estrogen plays a role in the synthesis and expression of Ro antigens on the surface of keratinocytes, increasing the fixation of anti-Ro antibodies in keratinocytes and provoking the appearance of a cutaneous eruption in patients with a susceptible HLA profile.6
Conclusion
We report a rare case of SCLE induced by anastrozole use. Cases such as ours and others that implicate antiestrogen drugs in association with DI-SCLE are particularly noteworthy, considering many studies are looking at the potential usefulness of antiestrogen therapy in the treatment of SLE. Further research on this relationship is warranted.
- Reed B, Huff J, Jones S, et al. Subacute cutaneous lupus erythematosus associated with hydrochlorothiazide therapy. Ann Intern Med. 1985;103:49-51.
- Marzano A, Lazzari R, Polloni I, et al. Drug-induced subacute cutaneous lupus erythematosus: evidence for differences from its idiopathic counterpart. Br J Dermatol. 2011;165:335-341.
- Bonsmann G, Schiller M, Luger T, et al. Terbinafine-induced subacute cutaneous lupus erythematosus. J Am Acad Dermatol. 2001;44:925-931.
- Callen J. Review: drug induced subacute cutaneous lupus erythematosus. Lupus. 2010;19:1107-1111.
- Lin J, Callen JP. Subacute cutaneous lupus erythematosus (SCLE). Medscape website. http://emedicine.medscape.com/article/1065657-overview. Updated March 7, 2016. Accessed April 29, 2016.
- Lowe GC, Henderson CL, Grau RH, et al. A systematic review of drug-induced subacute cutaneous lupus erythematosus. Br J Dermatol. 2011;164:465-472.
- Vedove C, Giglio M, Schena D, et al. Drug-induced lupus erythematosus. Arch Dermatol Res. 2009;301:99-105.
- Trancart M, Cavailhes A, Balme B, et al. Anastrozole-induced subacute cutaneous lupus erythematosus [published online December 6, 2007]. Br J Dermatol. 2008;158:628-629.
- Fumal I, Danchin A, Cosserat F, et al. Subacute cutaneous lupus erythematosus associated with tamoxifen therapy: two cases. Dermatology. 2005;210:251-252.
- Brown C, Deng J. Thiazide diuretics induce cutaneous lupus-like adverse reaction. J Toxicol Clin Toxicol. 1995;33:729-733.
- Sontheimer R. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev. 2005;4:253-263.
- Crowson A, Magro C. Subacute cutaneous lupus erythematosus arising in the setting of calcium channel blocker therapy. Hum Pathol. 1997;28:67-73.
- Lorentz K, Booken N, Goerdt S, et al. Subacute cutaneous lupus erythematosus induced by terbinafine: case report and review of literature. J Dtsch Dermatol Ges. 2008;6:823-837.
- Kasperkiewicz M, Anemüller W, Angelova-Fischer I, et al. Subacute cutaneous lupus erythematosus associated with terbinafine. Clin Exp Dermatol. 2009;34:403-404.
- Miyagawa S, Okuchi T, Shiomi Y, et al. Subacute cutaneous lupus erythematosus lesions precipitated by griseofulvin. J Am Acad Dermatol. 1989;21:343-346.
- Inman RD. Immunologic sex differences and the female predominance in systemic lupus erythematosus. Arthritis Rheum. 1978;21:849-854.
- Cutolo M, Wilder RL. Different roles of androgens and estrogens in the susceptibility to autoimmune rheumatic diseases. Rheum Dis Clin North Am. 2000;26:825-839.
- Petri M. Sex hormones and systemic lupus erythematosus. Lupus. 2008;17:412-415.
- Lateef A, Petri M. Hormone replacement and contraceptive therapy in autoimmune diseases [published online January 18, 2012]. J Autoimmun. 2012;38:J170-J176.
- Buyon JP, Petri M, Kim MY, et al. The effect of combined estrogen and progesterone hormone replacement therapy on disease activity in systemic lupus erythematosus: a randomized trial. Ann Intern Med. 2005;142:954-962.
- Wu W, Suen J, Lin B, et al. Tamoxifen alleviates disease severity and decreases double negative T cells in autoimmune MRL-lpr/lpr mice. Immunology. 2000;100:110-118.
- Dayan M, Zinger H, Kalush F, et al. The beneficial effects of treatment with tamoxifen and anti-oestradiol antibody on experimental systemic lupus erythematosus are associated with cytokine modulations. Immunology. 1997;90:101-108.
- Sthoeger Z, Zinger H, Mozes E. Beneficial effects of the anti-oestrogen tamoxifen on systemic lupus erythematosus of (NZBxNZW)F1 female mice are associated with specific reduction of IgG3 autoantibodies. Ann Rheum Dis. 2003;62:341-346.
Drug-induced subacute cutaneous lupus erythematosus (DI-SCLE) was first described in 1985 in 5 patients who had been taking hydrochlorothiazide.1 The skin lesions in these patients were identical to those seen in idiopathic subacute cutaneous lupus erythematosus (SCLE) and were accompanied by the same autoantibodies (anti-Ro/Sjögren syndrome antigen A [SS-A] and anti-La/Sjögren syndrome antigen B [SS-B]) and HLA type (HLA-DR2/DR3) that are known to be associated with idiopathic SCLE. The skin lesions of SCLE in these 5 patients resolved spontaneously after discontinuing hydrochlorothiazide; however, anti-Ro/SS-A antibodies persisted in all except 1 patient.1 Over the last decade, an increasing number of drugs from different classes have been implicated to be associated with DI-SCLE. Since the concept of DI-SCLE was introduced, it has been reported to look identical to idiopathic SCLE, both clinically and histopathologically; however, one report suggested that the 2 entities can be distinguished based on clinical variations.2 In general, patients with DI-SCLE develop the same anti-Ro antibodies as seen in idiopathic SCLE. In addition, although the rash in DI-SCLE typically resolves with withdrawal of the offending drug, the antibodies tend to persist. Herein, we report a case of a patient being treated with an aromatase inhibitor who presented with clinical, serologic, and histopathologic evidence of DI-SCLE.
Case Report
A 69-year-old woman diagnosed with breast cancer 4 years prior to her presentation to dermatology initially underwent a lumpectomy and radiation treatment. She was subsequently started on anastrozole 2 years later. After 16 months of treatment with anastrozole, she developed an erythematous scaly rash on sun-exposed areas of the skin. The patient was seen by an outside dermatologist who treated her for a patient-perceived drug rash based on biopsy results that simply demonstrated interface dermatitis. She was treated with both topical and oral steroids with little improvement and therefore presented to our office approximately 6 months after starting treatment seeking a second opinion.



Physical examination revealed numerous erythematous scaly papules and plaques in a photodistributed pattern on the chest, back, legs, and arms (Figure 1). On further questioning, the patient noted that the rash became worse when she was at the beach or playing tennis outside as well as under indoor lights. A repeat biopsy was performed, revealing interface and perivascular dermatitis with an infiltrate composed of lymphocytes, histiocytes, and scattered pigment-laden macrophages (Figure 2). Given the appearance and distribution of the rash as well as the clinical scenario, drug-induced lupus was suspected. Anastrozole was the only medication being taken. Laboratory evaluation was performed and was negative for antinuclear antibodies, antihistone antibodies, and anti-La/SS-B antibodies but was positive for anti-Ro/SS-A antibodies (>8.0 U [reference range, <1.0 U]). Based on these findings, anastrozole-induced SCLE was the most likely explanation for this presentation. The patient was started on a sun-protective regimen (ie, wide-brimmed hat, daily sunscreen) and anastrozole was discontinued by her oncologist; the combination led to moderate improvement in symptoms. One week later, oral hydroxychloroquine 200 mg twice daily was started, which led to notable improvement (Figure 3). The patient was seen for 2 additional follow-up visits, each time with sustained resolution of the rash. The hydroxychloroquine was then stopped at her last visit 3 months after diagnosis. The patient was subsequently lost to follow-up.
Comment
Presentation of SCLE
Subacute cutaneous lupus erythematosus is a form of lupus erythematosus characterized by nonscarring, annular, scaly, erythematous plaques that occur on sun-exposed skin. The lesions are classically distributed on the upper back, chest, dorsal arms, and lateral neck but also can be found in other locations.3,4 Subacute cutaneous lupus erythematosus may be idiopathic; may occur in patients with systemic lupus erythematosus, Sjögren syndrome, or deficiency of the second component of complement (C2d); or may be drug induced.5 On histology SCLE presents as a lichenoid tissue reaction with focal vacuolization of the epidermal basal layer and perivascular lymphocytic infiltrate. On direct immunofluorescence, both idiopathic and drug-induced SCLE present with granular deposition of IgM, IgG, and C3 in a bandlike array at the dermoepidermal junction and circulating anti-Ro/SS-A antibodies. Therefore, histopathologically and immunologically, DI-SCLE is indistinguishable from idiopathic cases.6
Differential Diagnosis
It was previously thought that the clinical presentation of DI-SCLE and idiopathic SCLE were indistinguishable; however, Marzano et al2 described remarkable differences in the cutaneous manifestations of the 2 diseases. Drug-induced SCLE lesions are more widespread, occur more frequently on the legs, and may be bullous or erythema multiforme–like versus the idiopathic lesions, which tend to be more concentrated on the upper body and classically present as scaly erythematous plaques. Additionally, malar rash and vasculitic lesions, such as purpura and necrotic-ulcerative lesions, are seen more often in DI-SCLE.
Drug-induced systemic lupus erythematosus (DI-SLE) is a lupuslike syndrome that can be differentiated from DI-SCLE by virtue of its clinical and serological presentation. It differs from DI-SCLE in that DI-SLE typically does not present with skin symptoms; rather, systemic symptoms such as fever, weight loss, arthralgia, polyarthritis, pericarditis, and pleuritis are more commonly seen. Additionally, it has been associated with antihistone antibodies.4 More than 80 drugs have been reported to cause DI-SLE, including procainamide, hydralazine, and quinidine.7
To be classified as either DI-SCLE or DI-SLE, symptoms need to present after administration of the triggering drug and must resolve after the drug is discontinued.7 The drugs most commonly associated with DI-SCLE are thiazides, calcium channel blockers, tumor necrosis factor α inhibitors, angiotensin-converting enzyme inhibitors, and terbinafine, with few cases citing anastrozole as the inciting agent.4,6,8,9 The incubation period for DI-SCLE varies substantially. Thiazide diuretics and calcium channel blockers typically have the longest incubation period, ranging from 6 months to 5 years for thiazides,1,6,10,11 while calcium channel blockers have an average incubation period of 3 years.12 Drug-induced SCLE associated with antifungals, however, usually is much more rapid in onset; the incubation period on average is 5 weeks for terbinafine and 2 weeks for griseofulvin.13-15
Antiestrogen Drugs and SCLE
Anastrozole, the inciting agent in our case, is a third-generation, selective, nonsteroidal, aromatase inhibitor with no progestogenic, androgenic, or estrogenic activity. Anastrozole, when taken at its recommended dosage of 1 mg daily, will suppress estradiol. It is used as an adjuvant treatment of estrogen-sensitive breast cancer in postmenopausal women. In contrast to a prior case of DI-SCLE secondary to anastrozole in which the incubation period was approximately 1 month,8 our patient had an incubation period of approximately 16 months. Tamoxifen, another antiestrogen drug, also has been associated with DI-SCLE.9 In cases of tamoxifen-induced SCLE, the incubation period was several years, which is more similar to our patient. Although these drugs do not have the same mechanism of action, they both have antiestrogen properties.9 A systemic review of DI-SCLE reported that incubation periods between drug exposure and appearance of DI-SCLE varied greatly and were drug class dependent. It is possible that reactions associated with antiestrogen medications have a delayed presentation; however, given there are limited cases of anastrozole-induced DI-SCLE, we cannot make a clear statement on incubation periods.6
Reports of DI-SCLE caused by antiestrogen drugs are particularly interesting because sex hormones in relation to lupus disease activity have been the subject of debate for decades. Women are considerably more likely to develop autoimmune diseases than men, suggesting that steroid hormones, especially estrogen and progesterone, influence the immune system.16 Estrogen actions are proinflammatory, while the actions of progesterone, androgens, and glucocorticoids are anti-inflammatory.17 Studies in women with lupus revealed an increased rate of mild- to moderate-intensity disease flares associated with estrogen-containing hormone replace-ment therapy.18-20
Over the years, several antiestrogen therapies have been used in murine models, which showed remarkable clinical improvement in the course of SLE. The precise mechanisms involved in disease immunomodulation by these therapies have not been elucidated.21-23 It is thought that estrogen plays a role in the synthesis and expression of Ro antigens on the surface of keratinocytes, increasing the fixation of anti-Ro antibodies in keratinocytes and provoking the appearance of a cutaneous eruption in patients with a susceptible HLA profile.6
Conclusion
We report a rare case of SCLE induced by anastrozole use. Cases such as ours and others that implicate antiestrogen drugs in association with DI-SCLE are particularly noteworthy, considering many studies are looking at the potential usefulness of antiestrogen therapy in the treatment of SLE. Further research on this relationship is warranted.
Drug-induced subacute cutaneous lupus erythematosus (DI-SCLE) was first described in 1985 in 5 patients who had been taking hydrochlorothiazide.1 The skin lesions in these patients were identical to those seen in idiopathic subacute cutaneous lupus erythematosus (SCLE) and were accompanied by the same autoantibodies (anti-Ro/Sjögren syndrome antigen A [SS-A] and anti-La/Sjögren syndrome antigen B [SS-B]) and HLA type (HLA-DR2/DR3) that are known to be associated with idiopathic SCLE. The skin lesions of SCLE in these 5 patients resolved spontaneously after discontinuing hydrochlorothiazide; however, anti-Ro/SS-A antibodies persisted in all except 1 patient.1 Over the last decade, an increasing number of drugs from different classes have been implicated to be associated with DI-SCLE. Since the concept of DI-SCLE was introduced, it has been reported to look identical to idiopathic SCLE, both clinically and histopathologically; however, one report suggested that the 2 entities can be distinguished based on clinical variations.2 In general, patients with DI-SCLE develop the same anti-Ro antibodies as seen in idiopathic SCLE. In addition, although the rash in DI-SCLE typically resolves with withdrawal of the offending drug, the antibodies tend to persist. Herein, we report a case of a patient being treated with an aromatase inhibitor who presented with clinical, serologic, and histopathologic evidence of DI-SCLE.
Case Report
A 69-year-old woman diagnosed with breast cancer 4 years prior to her presentation to dermatology initially underwent a lumpectomy and radiation treatment. She was subsequently started on anastrozole 2 years later. After 16 months of treatment with anastrozole, she developed an erythematous scaly rash on sun-exposed areas of the skin. The patient was seen by an outside dermatologist who treated her for a patient-perceived drug rash based on biopsy results that simply demonstrated interface dermatitis. She was treated with both topical and oral steroids with little improvement and therefore presented to our office approximately 6 months after starting treatment seeking a second opinion.



Physical examination revealed numerous erythematous scaly papules and plaques in a photodistributed pattern on the chest, back, legs, and arms (Figure 1). On further questioning, the patient noted that the rash became worse when she was at the beach or playing tennis outside as well as under indoor lights. A repeat biopsy was performed, revealing interface and perivascular dermatitis with an infiltrate composed of lymphocytes, histiocytes, and scattered pigment-laden macrophages (Figure 2). Given the appearance and distribution of the rash as well as the clinical scenario, drug-induced lupus was suspected. Anastrozole was the only medication being taken. Laboratory evaluation was performed and was negative for antinuclear antibodies, antihistone antibodies, and anti-La/SS-B antibodies but was positive for anti-Ro/SS-A antibodies (>8.0 U [reference range, <1.0 U]). Based on these findings, anastrozole-induced SCLE was the most likely explanation for this presentation. The patient was started on a sun-protective regimen (ie, wide-brimmed hat, daily sunscreen) and anastrozole was discontinued by her oncologist; the combination led to moderate improvement in symptoms. One week later, oral hydroxychloroquine 200 mg twice daily was started, which led to notable improvement (Figure 3). The patient was seen for 2 additional follow-up visits, each time with sustained resolution of the rash. The hydroxychloroquine was then stopped at her last visit 3 months after diagnosis. The patient was subsequently lost to follow-up.
Comment
Presentation of SCLE
Subacute cutaneous lupus erythematosus is a form of lupus erythematosus characterized by nonscarring, annular, scaly, erythematous plaques that occur on sun-exposed skin. The lesions are classically distributed on the upper back, chest, dorsal arms, and lateral neck but also can be found in other locations.3,4 Subacute cutaneous lupus erythematosus may be idiopathic; may occur in patients with systemic lupus erythematosus, Sjögren syndrome, or deficiency of the second component of complement (C2d); or may be drug induced.5 On histology SCLE presents as a lichenoid tissue reaction with focal vacuolization of the epidermal basal layer and perivascular lymphocytic infiltrate. On direct immunofluorescence, both idiopathic and drug-induced SCLE present with granular deposition of IgM, IgG, and C3 in a bandlike array at the dermoepidermal junction and circulating anti-Ro/SS-A antibodies. Therefore, histopathologically and immunologically, DI-SCLE is indistinguishable from idiopathic cases.6
Differential Diagnosis
It was previously thought that the clinical presentation of DI-SCLE and idiopathic SCLE were indistinguishable; however, Marzano et al2 described remarkable differences in the cutaneous manifestations of the 2 diseases. Drug-induced SCLE lesions are more widespread, occur more frequently on the legs, and may be bullous or erythema multiforme–like versus the idiopathic lesions, which tend to be more concentrated on the upper body and classically present as scaly erythematous plaques. Additionally, malar rash and vasculitic lesions, such as purpura and necrotic-ulcerative lesions, are seen more often in DI-SCLE.
Drug-induced systemic lupus erythematosus (DI-SLE) is a lupuslike syndrome that can be differentiated from DI-SCLE by virtue of its clinical and serological presentation. It differs from DI-SCLE in that DI-SLE typically does not present with skin symptoms; rather, systemic symptoms such as fever, weight loss, arthralgia, polyarthritis, pericarditis, and pleuritis are more commonly seen. Additionally, it has been associated with antihistone antibodies.4 More than 80 drugs have been reported to cause DI-SLE, including procainamide, hydralazine, and quinidine.7
To be classified as either DI-SCLE or DI-SLE, symptoms need to present after administration of the triggering drug and must resolve after the drug is discontinued.7 The drugs most commonly associated with DI-SCLE are thiazides, calcium channel blockers, tumor necrosis factor α inhibitors, angiotensin-converting enzyme inhibitors, and terbinafine, with few cases citing anastrozole as the inciting agent.4,6,8,9 The incubation period for DI-SCLE varies substantially. Thiazide diuretics and calcium channel blockers typically have the longest incubation period, ranging from 6 months to 5 years for thiazides,1,6,10,11 while calcium channel blockers have an average incubation period of 3 years.12 Drug-induced SCLE associated with antifungals, however, usually is much more rapid in onset; the incubation period on average is 5 weeks for terbinafine and 2 weeks for griseofulvin.13-15
Antiestrogen Drugs and SCLE
Anastrozole, the inciting agent in our case, is a third-generation, selective, nonsteroidal, aromatase inhibitor with no progestogenic, androgenic, or estrogenic activity. Anastrozole, when taken at its recommended dosage of 1 mg daily, will suppress estradiol. It is used as an adjuvant treatment of estrogen-sensitive breast cancer in postmenopausal women. In contrast to a prior case of DI-SCLE secondary to anastrozole in which the incubation period was approximately 1 month,8 our patient had an incubation period of approximately 16 months. Tamoxifen, another antiestrogen drug, also has been associated with DI-SCLE.9 In cases of tamoxifen-induced SCLE, the incubation period was several years, which is more similar to our patient. Although these drugs do not have the same mechanism of action, they both have antiestrogen properties.9 A systemic review of DI-SCLE reported that incubation periods between drug exposure and appearance of DI-SCLE varied greatly and were drug class dependent. It is possible that reactions associated with antiestrogen medications have a delayed presentation; however, given there are limited cases of anastrozole-induced DI-SCLE, we cannot make a clear statement on incubation periods.6
Reports of DI-SCLE caused by antiestrogen drugs are particularly interesting because sex hormones in relation to lupus disease activity have been the subject of debate for decades. Women are considerably more likely to develop autoimmune diseases than men, suggesting that steroid hormones, especially estrogen and progesterone, influence the immune system.16 Estrogen actions are proinflammatory, while the actions of progesterone, androgens, and glucocorticoids are anti-inflammatory.17 Studies in women with lupus revealed an increased rate of mild- to moderate-intensity disease flares associated with estrogen-containing hormone replace-ment therapy.18-20
Over the years, several antiestrogen therapies have been used in murine models, which showed remarkable clinical improvement in the course of SLE. The precise mechanisms involved in disease immunomodulation by these therapies have not been elucidated.21-23 It is thought that estrogen plays a role in the synthesis and expression of Ro antigens on the surface of keratinocytes, increasing the fixation of anti-Ro antibodies in keratinocytes and provoking the appearance of a cutaneous eruption in patients with a susceptible HLA profile.6
Conclusion
We report a rare case of SCLE induced by anastrozole use. Cases such as ours and others that implicate antiestrogen drugs in association with DI-SCLE are particularly noteworthy, considering many studies are looking at the potential usefulness of antiestrogen therapy in the treatment of SLE. Further research on this relationship is warranted.
- Reed B, Huff J, Jones S, et al. Subacute cutaneous lupus erythematosus associated with hydrochlorothiazide therapy. Ann Intern Med. 1985;103:49-51.
- Marzano A, Lazzari R, Polloni I, et al. Drug-induced subacute cutaneous lupus erythematosus: evidence for differences from its idiopathic counterpart. Br J Dermatol. 2011;165:335-341.
- Bonsmann G, Schiller M, Luger T, et al. Terbinafine-induced subacute cutaneous lupus erythematosus. J Am Acad Dermatol. 2001;44:925-931.
- Callen J. Review: drug induced subacute cutaneous lupus erythematosus. Lupus. 2010;19:1107-1111.
- Lin J, Callen JP. Subacute cutaneous lupus erythematosus (SCLE). Medscape website. http://emedicine.medscape.com/article/1065657-overview. Updated March 7, 2016. Accessed April 29, 2016.
- Lowe GC, Henderson CL, Grau RH, et al. A systematic review of drug-induced subacute cutaneous lupus erythematosus. Br J Dermatol. 2011;164:465-472.
- Vedove C, Giglio M, Schena D, et al. Drug-induced lupus erythematosus. Arch Dermatol Res. 2009;301:99-105.
- Trancart M, Cavailhes A, Balme B, et al. Anastrozole-induced subacute cutaneous lupus erythematosus [published online December 6, 2007]. Br J Dermatol. 2008;158:628-629.
- Fumal I, Danchin A, Cosserat F, et al. Subacute cutaneous lupus erythematosus associated with tamoxifen therapy: two cases. Dermatology. 2005;210:251-252.
- Brown C, Deng J. Thiazide diuretics induce cutaneous lupus-like adverse reaction. J Toxicol Clin Toxicol. 1995;33:729-733.
- Sontheimer R. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev. 2005;4:253-263.
- Crowson A, Magro C. Subacute cutaneous lupus erythematosus arising in the setting of calcium channel blocker therapy. Hum Pathol. 1997;28:67-73.
- Lorentz K, Booken N, Goerdt S, et al. Subacute cutaneous lupus erythematosus induced by terbinafine: case report and review of literature. J Dtsch Dermatol Ges. 2008;6:823-837.
- Kasperkiewicz M, Anemüller W, Angelova-Fischer I, et al. Subacute cutaneous lupus erythematosus associated with terbinafine. Clin Exp Dermatol. 2009;34:403-404.
- Miyagawa S, Okuchi T, Shiomi Y, et al. Subacute cutaneous lupus erythematosus lesions precipitated by griseofulvin. J Am Acad Dermatol. 1989;21:343-346.
- Inman RD. Immunologic sex differences and the female predominance in systemic lupus erythematosus. Arthritis Rheum. 1978;21:849-854.
- Cutolo M, Wilder RL. Different roles of androgens and estrogens in the susceptibility to autoimmune rheumatic diseases. Rheum Dis Clin North Am. 2000;26:825-839.
- Petri M. Sex hormones and systemic lupus erythematosus. Lupus. 2008;17:412-415.
- Lateef A, Petri M. Hormone replacement and contraceptive therapy in autoimmune diseases [published online January 18, 2012]. J Autoimmun. 2012;38:J170-J176.
- Buyon JP, Petri M, Kim MY, et al. The effect of combined estrogen and progesterone hormone replacement therapy on disease activity in systemic lupus erythematosus: a randomized trial. Ann Intern Med. 2005;142:954-962.
- Wu W, Suen J, Lin B, et al. Tamoxifen alleviates disease severity and decreases double negative T cells in autoimmune MRL-lpr/lpr mice. Immunology. 2000;100:110-118.
- Dayan M, Zinger H, Kalush F, et al. The beneficial effects of treatment with tamoxifen and anti-oestradiol antibody on experimental systemic lupus erythematosus are associated with cytokine modulations. Immunology. 1997;90:101-108.
- Sthoeger Z, Zinger H, Mozes E. Beneficial effects of the anti-oestrogen tamoxifen on systemic lupus erythematosus of (NZBxNZW)F1 female mice are associated with specific reduction of IgG3 autoantibodies. Ann Rheum Dis. 2003;62:341-346.
- Reed B, Huff J, Jones S, et al. Subacute cutaneous lupus erythematosus associated with hydrochlorothiazide therapy. Ann Intern Med. 1985;103:49-51.
- Marzano A, Lazzari R, Polloni I, et al. Drug-induced subacute cutaneous lupus erythematosus: evidence for differences from its idiopathic counterpart. Br J Dermatol. 2011;165:335-341.
- Bonsmann G, Schiller M, Luger T, et al. Terbinafine-induced subacute cutaneous lupus erythematosus. J Am Acad Dermatol. 2001;44:925-931.
- Callen J. Review: drug induced subacute cutaneous lupus erythematosus. Lupus. 2010;19:1107-1111.
- Lin J, Callen JP. Subacute cutaneous lupus erythematosus (SCLE). Medscape website. http://emedicine.medscape.com/article/1065657-overview. Updated March 7, 2016. Accessed April 29, 2016.
- Lowe GC, Henderson CL, Grau RH, et al. A systematic review of drug-induced subacute cutaneous lupus erythematosus. Br J Dermatol. 2011;164:465-472.
- Vedove C, Giglio M, Schena D, et al. Drug-induced lupus erythematosus. Arch Dermatol Res. 2009;301:99-105.
- Trancart M, Cavailhes A, Balme B, et al. Anastrozole-induced subacute cutaneous lupus erythematosus [published online December 6, 2007]. Br J Dermatol. 2008;158:628-629.
- Fumal I, Danchin A, Cosserat F, et al. Subacute cutaneous lupus erythematosus associated with tamoxifen therapy: two cases. Dermatology. 2005;210:251-252.
- Brown C, Deng J. Thiazide diuretics induce cutaneous lupus-like adverse reaction. J Toxicol Clin Toxicol. 1995;33:729-733.
- Sontheimer R. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev. 2005;4:253-263.
- Crowson A, Magro C. Subacute cutaneous lupus erythematosus arising in the setting of calcium channel blocker therapy. Hum Pathol. 1997;28:67-73.
- Lorentz K, Booken N, Goerdt S, et al. Subacute cutaneous lupus erythematosus induced by terbinafine: case report and review of literature. J Dtsch Dermatol Ges. 2008;6:823-837.
- Kasperkiewicz M, Anemüller W, Angelova-Fischer I, et al. Subacute cutaneous lupus erythematosus associated with terbinafine. Clin Exp Dermatol. 2009;34:403-404.
- Miyagawa S, Okuchi T, Shiomi Y, et al. Subacute cutaneous lupus erythematosus lesions precipitated by griseofulvin. J Am Acad Dermatol. 1989;21:343-346.
- Inman RD. Immunologic sex differences and the female predominance in systemic lupus erythematosus. Arthritis Rheum. 1978;21:849-854.
- Cutolo M, Wilder RL. Different roles of androgens and estrogens in the susceptibility to autoimmune rheumatic diseases. Rheum Dis Clin North Am. 2000;26:825-839.
- Petri M. Sex hormones and systemic lupus erythematosus. Lupus. 2008;17:412-415.
- Lateef A, Petri M. Hormone replacement and contraceptive therapy in autoimmune diseases [published online January 18, 2012]. J Autoimmun. 2012;38:J170-J176.
- Buyon JP, Petri M, Kim MY, et al. The effect of combined estrogen and progesterone hormone replacement therapy on disease activity in systemic lupus erythematosus: a randomized trial. Ann Intern Med. 2005;142:954-962.
- Wu W, Suen J, Lin B, et al. Tamoxifen alleviates disease severity and decreases double negative T cells in autoimmune MRL-lpr/lpr mice. Immunology. 2000;100:110-118.
- Dayan M, Zinger H, Kalush F, et al. The beneficial effects of treatment with tamoxifen and anti-oestradiol antibody on experimental systemic lupus erythematosus are associated with cytokine modulations. Immunology. 1997;90:101-108.
- Sthoeger Z, Zinger H, Mozes E. Beneficial effects of the anti-oestrogen tamoxifen on systemic lupus erythematosus of (NZBxNZW)F1 female mice are associated with specific reduction of IgG3 autoantibodies. Ann Rheum Dis. 2003;62:341-346.
Practice Points
- There are numerous cases of drug-induced subacute cutaneous lupus erythematosus (DI-SCLE) published in the literature; however, there are limited reports with anastrozole implicated as the causative agent.
- Cases of DI-SCLE are clinically and histologically indistinguishable from idiopathic cases. It is important to recognize and withdraw the offending agent.
Food allergy testing only rarely needed for AD patients
BOSTON – Between 15% and 30% of children with moderate to severe atopic dermatitis also have food allergies, but the allergies are a trigger for AD in only a small subset of patients, according to Mercedes E. Gonzalez, MD.
In most cases, allergy testing is not indicated, she said at the American Academy of Dermatology summer meeting.
She described a scenario involving a parent who is concerned that a food allergy is causing her child’s AD. The child has had no hives, no lip swelling, and no other signs of immediate hypersensitivity. In such a case, the best approach is to treat with topical therapies and follow the patient clinically.
“Allergy testing independent of history is not recommended,” she said.
However, in cases involving a significant concern about food allergy, such as the presence of hives or urticaria, or when the child has severe dermatitis that is not improving with optimized topical therapies, an assessment can be undertaken, said Dr. Gonzalez of the University of Miami.
She recommended limited food allergy testing – for common culprits such as cow’s milk, eggs, wheat, soy, and peanuts – in children younger than age 5 years with moderate to severe AD, if the AD persists despite optimized topical treatment and/or a history of immediate and reproducible reaction after ingestion of a specific food.
Food elimination diets based solely on the findings of food allergy test results are not recommended for managing AD, she noted.
If a patient has true immunoglobulin E–mediated allergy they should practice avoidance to prevent potential serious health sequelae, Dr. Gonzalez said.
When testing is done, keep in mind that skin prick tests and serum-specific IgE levels have high negative predictive values above 95%, but low specificity and positive predictive values of 40%-60%, she pointed out. Positive tests should be verified with a food elimination diet or oral food challenge.
Also, most children develop tolerance to the foods over time and should be retested, Dr. Gonzalez said.
Early peanut introduction advised in infants with AD
There is no need to delay the introduction of peanuts into the diet of an infant at high risk for atopic dermatitis, Dr. Gonzalez said.
A 2015 consensus communication from the American Academy of Pediatrics and numerous other organizations, including the American Academy of Allergy, Asthma & Immunology and the Society of Pediatric Dermatology, offering interim guidance on the topic calls for introduction of peanut products into the diets of high-risk infants in countries where peanut allergy is present, she said.
High-risk infants were defined in the study as those with egg allergy and/or severe eczema.
The guidance, which the AAP “endorses and accepts as its policy” pending more formal guidelines currently in development, was based largely on findings from the LEAP (Learn Early About Peanut Allergy) trial – a 5-year randomized, controlled trial of 640 high-risk infants aged 4-11 months. The trial showed that 17.2% of infants who avoided peanuts had peanut allergy at 5 years, compared with 3.2% of those with peanut consumption three times weekly, a relative risk reduction of 81% (N Engl J Med. 2015; 372:803-13).
In infants with egg allergy or severe eczema, an evaluation by an allergist or dermatologist familiar with the guidance may be warranted to assist in implementing the suggestions, Dr. Gonzalez said.
Dr. Gonzalez reported receiving honoraria for serving as a speaker and/or advisory board member for Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Between 15% and 30% of children with moderate to severe atopic dermatitis also have food allergies, but the allergies are a trigger for AD in only a small subset of patients, according to Mercedes E. Gonzalez, MD.
In most cases, allergy testing is not indicated, she said at the American Academy of Dermatology summer meeting.
She described a scenario involving a parent who is concerned that a food allergy is causing her child’s AD. The child has had no hives, no lip swelling, and no other signs of immediate hypersensitivity. In such a case, the best approach is to treat with topical therapies and follow the patient clinically.
“Allergy testing independent of history is not recommended,” she said.
However, in cases involving a significant concern about food allergy, such as the presence of hives or urticaria, or when the child has severe dermatitis that is not improving with optimized topical therapies, an assessment can be undertaken, said Dr. Gonzalez of the University of Miami.
She recommended limited food allergy testing – for common culprits such as cow’s milk, eggs, wheat, soy, and peanuts – in children younger than age 5 years with moderate to severe AD, if the AD persists despite optimized topical treatment and/or a history of immediate and reproducible reaction after ingestion of a specific food.
Food elimination diets based solely on the findings of food allergy test results are not recommended for managing AD, she noted.
If a patient has true immunoglobulin E–mediated allergy they should practice avoidance to prevent potential serious health sequelae, Dr. Gonzalez said.
When testing is done, keep in mind that skin prick tests and serum-specific IgE levels have high negative predictive values above 95%, but low specificity and positive predictive values of 40%-60%, she pointed out. Positive tests should be verified with a food elimination diet or oral food challenge.
Also, most children develop tolerance to the foods over time and should be retested, Dr. Gonzalez said.
Early peanut introduction advised in infants with AD
There is no need to delay the introduction of peanuts into the diet of an infant at high risk for atopic dermatitis, Dr. Gonzalez said.
A 2015 consensus communication from the American Academy of Pediatrics and numerous other organizations, including the American Academy of Allergy, Asthma & Immunology and the Society of Pediatric Dermatology, offering interim guidance on the topic calls for introduction of peanut products into the diets of high-risk infants in countries where peanut allergy is present, she said.
High-risk infants were defined in the study as those with egg allergy and/or severe eczema.
The guidance, which the AAP “endorses and accepts as its policy” pending more formal guidelines currently in development, was based largely on findings from the LEAP (Learn Early About Peanut Allergy) trial – a 5-year randomized, controlled trial of 640 high-risk infants aged 4-11 months. The trial showed that 17.2% of infants who avoided peanuts had peanut allergy at 5 years, compared with 3.2% of those with peanut consumption three times weekly, a relative risk reduction of 81% (N Engl J Med. 2015; 372:803-13).
In infants with egg allergy or severe eczema, an evaluation by an allergist or dermatologist familiar with the guidance may be warranted to assist in implementing the suggestions, Dr. Gonzalez said.
Dr. Gonzalez reported receiving honoraria for serving as a speaker and/or advisory board member for Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
BOSTON – Between 15% and 30% of children with moderate to severe atopic dermatitis also have food allergies, but the allergies are a trigger for AD in only a small subset of patients, according to Mercedes E. Gonzalez, MD.
In most cases, allergy testing is not indicated, she said at the American Academy of Dermatology summer meeting.
She described a scenario involving a parent who is concerned that a food allergy is causing her child’s AD. The child has had no hives, no lip swelling, and no other signs of immediate hypersensitivity. In such a case, the best approach is to treat with topical therapies and follow the patient clinically.
“Allergy testing independent of history is not recommended,” she said.
However, in cases involving a significant concern about food allergy, such as the presence of hives or urticaria, or when the child has severe dermatitis that is not improving with optimized topical therapies, an assessment can be undertaken, said Dr. Gonzalez of the University of Miami.
She recommended limited food allergy testing – for common culprits such as cow’s milk, eggs, wheat, soy, and peanuts – in children younger than age 5 years with moderate to severe AD, if the AD persists despite optimized topical treatment and/or a history of immediate and reproducible reaction after ingestion of a specific food.
Food elimination diets based solely on the findings of food allergy test results are not recommended for managing AD, she noted.
If a patient has true immunoglobulin E–mediated allergy they should practice avoidance to prevent potential serious health sequelae, Dr. Gonzalez said.
When testing is done, keep in mind that skin prick tests and serum-specific IgE levels have high negative predictive values above 95%, but low specificity and positive predictive values of 40%-60%, she pointed out. Positive tests should be verified with a food elimination diet or oral food challenge.
Also, most children develop tolerance to the foods over time and should be retested, Dr. Gonzalez said.
Early peanut introduction advised in infants with AD
There is no need to delay the introduction of peanuts into the diet of an infant at high risk for atopic dermatitis, Dr. Gonzalez said.
A 2015 consensus communication from the American Academy of Pediatrics and numerous other organizations, including the American Academy of Allergy, Asthma & Immunology and the Society of Pediatric Dermatology, offering interim guidance on the topic calls for introduction of peanut products into the diets of high-risk infants in countries where peanut allergy is present, she said.
High-risk infants were defined in the study as those with egg allergy and/or severe eczema.
The guidance, which the AAP “endorses and accepts as its policy” pending more formal guidelines currently in development, was based largely on findings from the LEAP (Learn Early About Peanut Allergy) trial – a 5-year randomized, controlled trial of 640 high-risk infants aged 4-11 months. The trial showed that 17.2% of infants who avoided peanuts had peanut allergy at 5 years, compared with 3.2% of those with peanut consumption three times weekly, a relative risk reduction of 81% (N Engl J Med. 2015; 372:803-13).
In infants with egg allergy or severe eczema, an evaluation by an allergist or dermatologist familiar with the guidance may be warranted to assist in implementing the suggestions, Dr. Gonzalez said.
Dr. Gonzalez reported receiving honoraria for serving as a speaker and/or advisory board member for Pierre Fabre Dermatologie, Anacor Pharmaceuticals, Encore Dermatology, and PuraCap Pharmaceutical.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
Common allergic dermatitis culprits are hiding in plain sight
MINNEAPOLIS – When it comes to allergic contact dermatitis in children, the answer is sometimes hiding in plain sight. Cleansers, moisturizers, shampoos, detergents – all can contain ingredients that provoke significant reactions, yet many of these ingredients are not on the most common testing panels.
Erin Warshaw, MD, professor of dermatology at the University of Minnesota, reviewed common but often unsuspected causes of allergic dermatitis in the pediatric population at the annual meeting of the Society for Pediatric Dermatology.
Even some hypoallergenic and frequently recommended products can contain preservatives and other ingredients that provoke allergic reactions, according to Dr. Warshaw. A chief culprit is methylisothiazolinone (MI), a preservative that came into common use as formaldehyde has been gradually phased out.
“If there’s anything I could emphasize from this talk, it’s MI, MI, MI. This is the major epidemic of our time in the contact dermatitis world,” Dr. Warshaw said. Upcoming publications, she added, will place MI in the top five most common contact allergens. “MI is in everything, including things you would think would be hypoallergenic,” she said. She recommended looking at ingredient labels with a keen eye when making testing decisions.
Despite MI’s status as a frequent culprit, it’s not an allergen that appears on common test kits, Dr. Warshaw pointed out. For example, it’s absent from one of the most commonly used test kits, the Thin-Layer Rapid Use Epicutaneous Patch (T.R.U.E. test).
The T.R.U.E. test, said Dr. Warshaw, has reasonable sensitivity – it can detect 71% of relevant positive patch tests (RPPTs) in children. However, she added, a recent study showed that about 23% of children reacted to a supplemental allergen. “That’s significant. One quarter of these individuals only reacted to a preservative … or a sunscreen, or an acrylate. These aren’t on the T.R.U.E. test.”
Decyl glucoside is another frequent culprit that is not included in commercial patch test kits. “It’s really an important emerging allergen,” said Dr. Warshaw, noting that it commonly cross-reacts with coco and lauryl glucoside, frequently found in fragrance-free products. “It’s always humbling when we find the allergen in the product we’ve recommended to our patients.”
Other important allergens not on the T.R.U.E. test include propolis, tocopherol, oxybenzone, and many surfactants and botanicals.
In order to avoid a confounding reaction to aluminum, Dr. Warshaw recommends testing using plastic-backed test chambers, such as IQ chambers, rather than Finn chambers, which are aluminum backed.
When working with families to track down allergens in the pediatric population, Dr. Warshaw adjusts her approach from what she would use for adults.
“What do I do differently in kids? First of all, I set expectations for children and parents,” she said. Some of the most frequent parental questions deal with food allergies, so she allots time to explain the rationale for not testing for food allergens when allergic contact dermatitis is suspected.
For many patients, “I try and frame that there is probably baseline eczema, and our goal is to try to figure out if there is an allergy in addition to that that is contributing to the flares,” she said. She makes sure to convey that “all it takes is one exposure every 3 weeks; that will keep this reaction going.”
However, she’s judicious in interpreting equivocal results. “I feel a responsibility not to label children with an allergy” if results are unclear. Finally, providing enough time is key, said Dr. Warshaw, who allots an hour for reviewing final testing results.
The take-home points? It’s worthwhile to patch test children, since over half of children will have at least one RPPT. Also, contact dermatitis can be an overlay on preexisting allergic dermatitis, so patch testing can still be helpful for these children. Supplemental allergens are important in patch testing, “especially in children with a negative test to a screening series,” Dr. Warshaw said.
She recommended accessing the Contact Allergen Management Program (CAMP) database, found on the American Contact Dermatitis Society website. The list is a searchable database that generates a list of “safe” products that don’t contain a given allergen. This resource is available for society members, but a member’s access code can be shared among faculty members at academic institutions, she said. Patients can also be given unique codes that will give them access for life, so they can use the CAMP database on a computer or via a smartphone app.
Dr. Warshaw reported no relevant financial disclosures.
On Twitter @karioakes
MINNEAPOLIS – When it comes to allergic contact dermatitis in children, the answer is sometimes hiding in plain sight. Cleansers, moisturizers, shampoos, detergents – all can contain ingredients that provoke significant reactions, yet many of these ingredients are not on the most common testing panels.
Erin Warshaw, MD, professor of dermatology at the University of Minnesota, reviewed common but often unsuspected causes of allergic dermatitis in the pediatric population at the annual meeting of the Society for Pediatric Dermatology.
Even some hypoallergenic and frequently recommended products can contain preservatives and other ingredients that provoke allergic reactions, according to Dr. Warshaw. A chief culprit is methylisothiazolinone (MI), a preservative that came into common use as formaldehyde has been gradually phased out.
“If there’s anything I could emphasize from this talk, it’s MI, MI, MI. This is the major epidemic of our time in the contact dermatitis world,” Dr. Warshaw said. Upcoming publications, she added, will place MI in the top five most common contact allergens. “MI is in everything, including things you would think would be hypoallergenic,” she said. She recommended looking at ingredient labels with a keen eye when making testing decisions.
Despite MI’s status as a frequent culprit, it’s not an allergen that appears on common test kits, Dr. Warshaw pointed out. For example, it’s absent from one of the most commonly used test kits, the Thin-Layer Rapid Use Epicutaneous Patch (T.R.U.E. test).
The T.R.U.E. test, said Dr. Warshaw, has reasonable sensitivity – it can detect 71% of relevant positive patch tests (RPPTs) in children. However, she added, a recent study showed that about 23% of children reacted to a supplemental allergen. “That’s significant. One quarter of these individuals only reacted to a preservative … or a sunscreen, or an acrylate. These aren’t on the T.R.U.E. test.”
Decyl glucoside is another frequent culprit that is not included in commercial patch test kits. “It’s really an important emerging allergen,” said Dr. Warshaw, noting that it commonly cross-reacts with coco and lauryl glucoside, frequently found in fragrance-free products. “It’s always humbling when we find the allergen in the product we’ve recommended to our patients.”
Other important allergens not on the T.R.U.E. test include propolis, tocopherol, oxybenzone, and many surfactants and botanicals.
In order to avoid a confounding reaction to aluminum, Dr. Warshaw recommends testing using plastic-backed test chambers, such as IQ chambers, rather than Finn chambers, which are aluminum backed.
When working with families to track down allergens in the pediatric population, Dr. Warshaw adjusts her approach from what she would use for adults.
“What do I do differently in kids? First of all, I set expectations for children and parents,” she said. Some of the most frequent parental questions deal with food allergies, so she allots time to explain the rationale for not testing for food allergens when allergic contact dermatitis is suspected.
For many patients, “I try and frame that there is probably baseline eczema, and our goal is to try to figure out if there is an allergy in addition to that that is contributing to the flares,” she said. She makes sure to convey that “all it takes is one exposure every 3 weeks; that will keep this reaction going.”
However, she’s judicious in interpreting equivocal results. “I feel a responsibility not to label children with an allergy” if results are unclear. Finally, providing enough time is key, said Dr. Warshaw, who allots an hour for reviewing final testing results.
The take-home points? It’s worthwhile to patch test children, since over half of children will have at least one RPPT. Also, contact dermatitis can be an overlay on preexisting allergic dermatitis, so patch testing can still be helpful for these children. Supplemental allergens are important in patch testing, “especially in children with a negative test to a screening series,” Dr. Warshaw said.
She recommended accessing the Contact Allergen Management Program (CAMP) database, found on the American Contact Dermatitis Society website. The list is a searchable database that generates a list of “safe” products that don’t contain a given allergen. This resource is available for society members, but a member’s access code can be shared among faculty members at academic institutions, she said. Patients can also be given unique codes that will give them access for life, so they can use the CAMP database on a computer or via a smartphone app.
Dr. Warshaw reported no relevant financial disclosures.
On Twitter @karioakes
MINNEAPOLIS – When it comes to allergic contact dermatitis in children, the answer is sometimes hiding in plain sight. Cleansers, moisturizers, shampoos, detergents – all can contain ingredients that provoke significant reactions, yet many of these ingredients are not on the most common testing panels.
Erin Warshaw, MD, professor of dermatology at the University of Minnesota, reviewed common but often unsuspected causes of allergic dermatitis in the pediatric population at the annual meeting of the Society for Pediatric Dermatology.
Even some hypoallergenic and frequently recommended products can contain preservatives and other ingredients that provoke allergic reactions, according to Dr. Warshaw. A chief culprit is methylisothiazolinone (MI), a preservative that came into common use as formaldehyde has been gradually phased out.
“If there’s anything I could emphasize from this talk, it’s MI, MI, MI. This is the major epidemic of our time in the contact dermatitis world,” Dr. Warshaw said. Upcoming publications, she added, will place MI in the top five most common contact allergens. “MI is in everything, including things you would think would be hypoallergenic,” she said. She recommended looking at ingredient labels with a keen eye when making testing decisions.
Despite MI’s status as a frequent culprit, it’s not an allergen that appears on common test kits, Dr. Warshaw pointed out. For example, it’s absent from one of the most commonly used test kits, the Thin-Layer Rapid Use Epicutaneous Patch (T.R.U.E. test).
The T.R.U.E. test, said Dr. Warshaw, has reasonable sensitivity – it can detect 71% of relevant positive patch tests (RPPTs) in children. However, she added, a recent study showed that about 23% of children reacted to a supplemental allergen. “That’s significant. One quarter of these individuals only reacted to a preservative … or a sunscreen, or an acrylate. These aren’t on the T.R.U.E. test.”
Decyl glucoside is another frequent culprit that is not included in commercial patch test kits. “It’s really an important emerging allergen,” said Dr. Warshaw, noting that it commonly cross-reacts with coco and lauryl glucoside, frequently found in fragrance-free products. “It’s always humbling when we find the allergen in the product we’ve recommended to our patients.”
Other important allergens not on the T.R.U.E. test include propolis, tocopherol, oxybenzone, and many surfactants and botanicals.
In order to avoid a confounding reaction to aluminum, Dr. Warshaw recommends testing using plastic-backed test chambers, such as IQ chambers, rather than Finn chambers, which are aluminum backed.
When working with families to track down allergens in the pediatric population, Dr. Warshaw adjusts her approach from what she would use for adults.
“What do I do differently in kids? First of all, I set expectations for children and parents,” she said. Some of the most frequent parental questions deal with food allergies, so she allots time to explain the rationale for not testing for food allergens when allergic contact dermatitis is suspected.
For many patients, “I try and frame that there is probably baseline eczema, and our goal is to try to figure out if there is an allergy in addition to that that is contributing to the flares,” she said. She makes sure to convey that “all it takes is one exposure every 3 weeks; that will keep this reaction going.”
However, she’s judicious in interpreting equivocal results. “I feel a responsibility not to label children with an allergy” if results are unclear. Finally, providing enough time is key, said Dr. Warshaw, who allots an hour for reviewing final testing results.
The take-home points? It’s worthwhile to patch test children, since over half of children will have at least one RPPT. Also, contact dermatitis can be an overlay on preexisting allergic dermatitis, so patch testing can still be helpful for these children. Supplemental allergens are important in patch testing, “especially in children with a negative test to a screening series,” Dr. Warshaw said.
She recommended accessing the Contact Allergen Management Program (CAMP) database, found on the American Contact Dermatitis Society website. The list is a searchable database that generates a list of “safe” products that don’t contain a given allergen. This resource is available for society members, but a member’s access code can be shared among faculty members at academic institutions, she said. Patients can also be given unique codes that will give them access for life, so they can use the CAMP database on a computer or via a smartphone app.
Dr. Warshaw reported no relevant financial disclosures.
On Twitter @karioakes
EXPERT ANALYSIS FROM THE SPD ANNUAL MEETING