User login
Survey shines new light on weighty comorbidity burden in adult atopic dermatitis
VIENNA – Newly enhanced appreciation of the profound burden of comorbidities associated with adult atopic dermatitis (AD) is provided by the Liberty AD-AWARE study, investigators said at a joint program of the International Eczema Council and the International Psoriasis Council held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“I think the only reason we thought psoriasis is a systemic disease and atopic dermatitis is not is because people were researching it much more in psoriasis. I think atopic dermatitis will emerge as potentially more systemic than psoriasis, including the comorbidities. It’s just a matter of time before the evidence is put forth for atopic dermatitis,” predicted Emma Guttman-Yassky, MD, PhD, professor and vice chair of the department of dermatology at Mount Sinai School of Medicine in New York.
Dr. Guttman-Yassky noted that 85% of cases of AD begin before 5 years of age. Many cases resolve later in childhood, but for others it becomes a chronic lifelong condition. And while the burden of AD has been well characterized in the pediatric population, that’s not so in affected adults. This was the impetus for the Liberty AD-AWARE (Adults With Atopic Dermatitis Reporting on their Experience) study, an Internet-based cross-sectional survey of more than 1,500 adults with AD receiving their care from dermatologists at eight major U.S. academic medical centers.
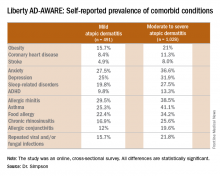
Eric L. Simpson, MD, a coinvestigator with Dr. Guttman-Yassky in Liberty AD-AWARE, observed that the study documented self-reported high rates of a range of psychiatric, cardiovascular, allergic, respiratory, and infectious diseases in participants. And while a cross-sectional study can’t establish causality, it’s important to appreciate that rates of these comorbidities were across the board significantly higher in the 1,028 patients with moderate to severe AD over the prior 12 months than in the 491 classified as having mild AD.
These associations between AD and mental health problems have been confirmed in other studies. For example, a recent analysis of data on more than 354,000 children and nearly 35,000 adults in the United States demonstrated that AD was independently associated with a 14% increased likelihood of attention-deficit/hyperactivity disorder in children and a 61% increased risk in adults. Those risks of ADHD rose far higher in individuals with severe AD and sleep disruption (Br J Dermatol. 2016 Nov;175[5]:920-9).
A number of theories have been put forth to explain these associations, including altered brain development stemming from early exposure to inflammatory cytokines or perhaps shared genetic predisposition, but Dr. Simpson proposed a simpler explanation which carries more optimistic implications.
“I suspect the mental health problems associated with adult atopic dermatitis are probably nonspecific sequelae of any chronic skin disorder involving severe itch and sleep disturbances,” said Dr. Simpson, professor of dermatology at Oregon Health & Science University, Portland.
Moreover, there is good reason to believe that novel therapies targeting inflammation more effectively than what’s been available to date may help improve mental health outcomes, as well as asthma in affected adults with AD, he added. He cited a phase IIb, randomized, double-blind, placebo-controlled study for which he was lead investigator. In this trial, 16 weeks of treatment with dupilumab, a first-in-class investigational blocker of the interleukin-4/interleukin-13 signaling pathway, not only resulted in significant reductions in itch and sleep problems, it also decreased anxiety and depression symptoms and improved multiple validated measures of health-related quality of life (J Am Acad Dermatol. 2016 Sep;75[3]:506-15).
Liberty AD-AWARE provides hints of the profound cumulative negative impact moderate to severe AD can have on a patient’s life course. Among the group with moderate to severe disease, 7.5% said AD had a large negative effect on their pursuit of an education, 10.7% said their disease had influenced their career choice “a lot/very much,” 13.3% were unemployed for reasons other than being retired or a student, and 17.1% reported an annual family income of less than $25,000. All these rates were multifold higher than in patients with mild AD in the study, which didn’t include a non-AD control group.
Dr. Guttman-Yassky observed that 42% of the moderate to severe AD group in Liberty AD-AWARE reported their current treatments were ineffective at controlling their disease, even though study participants were presumably receiving high-quality care at academic medical centers. Twenty-eight percent of patients with inadequately controlled AD had used phototherapy or an immunomodulatory drug within the past 7 days, underscoring the limitations of those forms of therapy in patients with more severe AD as well as the need for new and better treatments.
Dr. Guttman-Yassky has played a key role in the paradigm shift regarding understanding of the pathogenesis of AD as involving not just disordered skin barrier function but also immunologic impairment. She was senior author of a study that showed the nonlesional skin of patients with AD is characterized by high-level expression of inflammatory cytokines, whereas the nonlesional skin of psoriasis patients is not, an observation that serves to highlight the need for proactive treatments for AD (J Allergy Clin Immunol. 2011 Apr;127[4]:954-64.e1-4). Later, she and her coworkers demonstrated that AD is characterized by greater levels of T-cell activation among central and effector CD4+ and CD8+CLA+ and CD8+CLA– memory cell subsets (J Allergy Clin Immunol. 2015 Jul;136[1]:208-11).
More recently, she was also senior author of a landmark study that provides a mechanism to account for the reason AD patients would potentially have more comorbid illnesses than psoriasis patients. The investigators demonstrated that AD is accompanied by systemic expansion of transitional and chronically activated memory B cells, plasmablasts, and IgE-expressing memory B cells in both skin and blood. In other words, AD is characterized by a greater level of systemic immune activation, compared with psoriasis, where activated T cells are largely confined to the skin, and activated central memory B cells don’t figure prominently (J Allergy Clin Immunol. 2016 Jan;137[1]:118-29.e5).
The Liberty AD-AWARE study was sponsored by Sanofi and Regeneron. Dr. Simpson and Dr. Guttman-Yassky reported receiving research grants from and serving as consultants to those and other pharmaceutical companies.
VIENNA – Newly enhanced appreciation of the profound burden of comorbidities associated with adult atopic dermatitis (AD) is provided by the Liberty AD-AWARE study, investigators said at a joint program of the International Eczema Council and the International Psoriasis Council held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“I think the only reason we thought psoriasis is a systemic disease and atopic dermatitis is not is because people were researching it much more in psoriasis. I think atopic dermatitis will emerge as potentially more systemic than psoriasis, including the comorbidities. It’s just a matter of time before the evidence is put forth for atopic dermatitis,” predicted Emma Guttman-Yassky, MD, PhD, professor and vice chair of the department of dermatology at Mount Sinai School of Medicine in New York.
Dr. Guttman-Yassky noted that 85% of cases of AD begin before 5 years of age. Many cases resolve later in childhood, but for others it becomes a chronic lifelong condition. And while the burden of AD has been well characterized in the pediatric population, that’s not so in affected adults. This was the impetus for the Liberty AD-AWARE (Adults With Atopic Dermatitis Reporting on their Experience) study, an Internet-based cross-sectional survey of more than 1,500 adults with AD receiving their care from dermatologists at eight major U.S. academic medical centers.
Eric L. Simpson, MD, a coinvestigator with Dr. Guttman-Yassky in Liberty AD-AWARE, observed that the study documented self-reported high rates of a range of psychiatric, cardiovascular, allergic, respiratory, and infectious diseases in participants. And while a cross-sectional study can’t establish causality, it’s important to appreciate that rates of these comorbidities were across the board significantly higher in the 1,028 patients with moderate to severe AD over the prior 12 months than in the 491 classified as having mild AD.
These associations between AD and mental health problems have been confirmed in other studies. For example, a recent analysis of data on more than 354,000 children and nearly 35,000 adults in the United States demonstrated that AD was independently associated with a 14% increased likelihood of attention-deficit/hyperactivity disorder in children and a 61% increased risk in adults. Those risks of ADHD rose far higher in individuals with severe AD and sleep disruption (Br J Dermatol. 2016 Nov;175[5]:920-9).
A number of theories have been put forth to explain these associations, including altered brain development stemming from early exposure to inflammatory cytokines or perhaps shared genetic predisposition, but Dr. Simpson proposed a simpler explanation which carries more optimistic implications.
“I suspect the mental health problems associated with adult atopic dermatitis are probably nonspecific sequelae of any chronic skin disorder involving severe itch and sleep disturbances,” said Dr. Simpson, professor of dermatology at Oregon Health & Science University, Portland.
Moreover, there is good reason to believe that novel therapies targeting inflammation more effectively than what’s been available to date may help improve mental health outcomes, as well as asthma in affected adults with AD, he added. He cited a phase IIb, randomized, double-blind, placebo-controlled study for which he was lead investigator. In this trial, 16 weeks of treatment with dupilumab, a first-in-class investigational blocker of the interleukin-4/interleukin-13 signaling pathway, not only resulted in significant reductions in itch and sleep problems, it also decreased anxiety and depression symptoms and improved multiple validated measures of health-related quality of life (J Am Acad Dermatol. 2016 Sep;75[3]:506-15).
Liberty AD-AWARE provides hints of the profound cumulative negative impact moderate to severe AD can have on a patient’s life course. Among the group with moderate to severe disease, 7.5% said AD had a large negative effect on their pursuit of an education, 10.7% said their disease had influenced their career choice “a lot/very much,” 13.3% were unemployed for reasons other than being retired or a student, and 17.1% reported an annual family income of less than $25,000. All these rates were multifold higher than in patients with mild AD in the study, which didn’t include a non-AD control group.
Dr. Guttman-Yassky observed that 42% of the moderate to severe AD group in Liberty AD-AWARE reported their current treatments were ineffective at controlling their disease, even though study participants were presumably receiving high-quality care at academic medical centers. Twenty-eight percent of patients with inadequately controlled AD had used phototherapy or an immunomodulatory drug within the past 7 days, underscoring the limitations of those forms of therapy in patients with more severe AD as well as the need for new and better treatments.
Dr. Guttman-Yassky has played a key role in the paradigm shift regarding understanding of the pathogenesis of AD as involving not just disordered skin barrier function but also immunologic impairment. She was senior author of a study that showed the nonlesional skin of patients with AD is characterized by high-level expression of inflammatory cytokines, whereas the nonlesional skin of psoriasis patients is not, an observation that serves to highlight the need for proactive treatments for AD (J Allergy Clin Immunol. 2011 Apr;127[4]:954-64.e1-4). Later, she and her coworkers demonstrated that AD is characterized by greater levels of T-cell activation among central and effector CD4+ and CD8+CLA+ and CD8+CLA– memory cell subsets (J Allergy Clin Immunol. 2015 Jul;136[1]:208-11).
More recently, she was also senior author of a landmark study that provides a mechanism to account for the reason AD patients would potentially have more comorbid illnesses than psoriasis patients. The investigators demonstrated that AD is accompanied by systemic expansion of transitional and chronically activated memory B cells, plasmablasts, and IgE-expressing memory B cells in both skin and blood. In other words, AD is characterized by a greater level of systemic immune activation, compared with psoriasis, where activated T cells are largely confined to the skin, and activated central memory B cells don’t figure prominently (J Allergy Clin Immunol. 2016 Jan;137[1]:118-29.e5).
The Liberty AD-AWARE study was sponsored by Sanofi and Regeneron. Dr. Simpson and Dr. Guttman-Yassky reported receiving research grants from and serving as consultants to those and other pharmaceutical companies.
VIENNA – Newly enhanced appreciation of the profound burden of comorbidities associated with adult atopic dermatitis (AD) is provided by the Liberty AD-AWARE study, investigators said at a joint program of the International Eczema Council and the International Psoriasis Council held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“I think the only reason we thought psoriasis is a systemic disease and atopic dermatitis is not is because people were researching it much more in psoriasis. I think atopic dermatitis will emerge as potentially more systemic than psoriasis, including the comorbidities. It’s just a matter of time before the evidence is put forth for atopic dermatitis,” predicted Emma Guttman-Yassky, MD, PhD, professor and vice chair of the department of dermatology at Mount Sinai School of Medicine in New York.
Dr. Guttman-Yassky noted that 85% of cases of AD begin before 5 years of age. Many cases resolve later in childhood, but for others it becomes a chronic lifelong condition. And while the burden of AD has been well characterized in the pediatric population, that’s not so in affected adults. This was the impetus for the Liberty AD-AWARE (Adults With Atopic Dermatitis Reporting on their Experience) study, an Internet-based cross-sectional survey of more than 1,500 adults with AD receiving their care from dermatologists at eight major U.S. academic medical centers.
Eric L. Simpson, MD, a coinvestigator with Dr. Guttman-Yassky in Liberty AD-AWARE, observed that the study documented self-reported high rates of a range of psychiatric, cardiovascular, allergic, respiratory, and infectious diseases in participants. And while a cross-sectional study can’t establish causality, it’s important to appreciate that rates of these comorbidities were across the board significantly higher in the 1,028 patients with moderate to severe AD over the prior 12 months than in the 491 classified as having mild AD.
These associations between AD and mental health problems have been confirmed in other studies. For example, a recent analysis of data on more than 354,000 children and nearly 35,000 adults in the United States demonstrated that AD was independently associated with a 14% increased likelihood of attention-deficit/hyperactivity disorder in children and a 61% increased risk in adults. Those risks of ADHD rose far higher in individuals with severe AD and sleep disruption (Br J Dermatol. 2016 Nov;175[5]:920-9).
A number of theories have been put forth to explain these associations, including altered brain development stemming from early exposure to inflammatory cytokines or perhaps shared genetic predisposition, but Dr. Simpson proposed a simpler explanation which carries more optimistic implications.
“I suspect the mental health problems associated with adult atopic dermatitis are probably nonspecific sequelae of any chronic skin disorder involving severe itch and sleep disturbances,” said Dr. Simpson, professor of dermatology at Oregon Health & Science University, Portland.
Moreover, there is good reason to believe that novel therapies targeting inflammation more effectively than what’s been available to date may help improve mental health outcomes, as well as asthma in affected adults with AD, he added. He cited a phase IIb, randomized, double-blind, placebo-controlled study for which he was lead investigator. In this trial, 16 weeks of treatment with dupilumab, a first-in-class investigational blocker of the interleukin-4/interleukin-13 signaling pathway, not only resulted in significant reductions in itch and sleep problems, it also decreased anxiety and depression symptoms and improved multiple validated measures of health-related quality of life (J Am Acad Dermatol. 2016 Sep;75[3]:506-15).
Liberty AD-AWARE provides hints of the profound cumulative negative impact moderate to severe AD can have on a patient’s life course. Among the group with moderate to severe disease, 7.5% said AD had a large negative effect on their pursuit of an education, 10.7% said their disease had influenced their career choice “a lot/very much,” 13.3% were unemployed for reasons other than being retired or a student, and 17.1% reported an annual family income of less than $25,000. All these rates were multifold higher than in patients with mild AD in the study, which didn’t include a non-AD control group.
Dr. Guttman-Yassky observed that 42% of the moderate to severe AD group in Liberty AD-AWARE reported their current treatments were ineffective at controlling their disease, even though study participants were presumably receiving high-quality care at academic medical centers. Twenty-eight percent of patients with inadequately controlled AD had used phototherapy or an immunomodulatory drug within the past 7 days, underscoring the limitations of those forms of therapy in patients with more severe AD as well as the need for new and better treatments.
Dr. Guttman-Yassky has played a key role in the paradigm shift regarding understanding of the pathogenesis of AD as involving not just disordered skin barrier function but also immunologic impairment. She was senior author of a study that showed the nonlesional skin of patients with AD is characterized by high-level expression of inflammatory cytokines, whereas the nonlesional skin of psoriasis patients is not, an observation that serves to highlight the need for proactive treatments for AD (J Allergy Clin Immunol. 2011 Apr;127[4]:954-64.e1-4). Later, she and her coworkers demonstrated that AD is characterized by greater levels of T-cell activation among central and effector CD4+ and CD8+CLA+ and CD8+CLA– memory cell subsets (J Allergy Clin Immunol. 2015 Jul;136[1]:208-11).
More recently, she was also senior author of a landmark study that provides a mechanism to account for the reason AD patients would potentially have more comorbid illnesses than psoriasis patients. The investigators demonstrated that AD is accompanied by systemic expansion of transitional and chronically activated memory B cells, plasmablasts, and IgE-expressing memory B cells in both skin and blood. In other words, AD is characterized by a greater level of systemic immune activation, compared with psoriasis, where activated T cells are largely confined to the skin, and activated central memory B cells don’t figure prominently (J Allergy Clin Immunol. 2016 Jan;137[1]:118-29.e5).
The Liberty AD-AWARE study was sponsored by Sanofi and Regeneron. Dr. Simpson and Dr. Guttman-Yassky reported receiving research grants from and serving as consultants to those and other pharmaceutical companies.
EXPERT ANALYSIS FROM THE EADV CONGRESS
FDA gives nod to crisaborole for atopic dermatitis
The Food and Drug Administration has approved crisaborole topical ointment, 2%, to treat mild to moderate atopic dermatitis in patients 2 years of age and older. The boron-based phosphodiesterase 4 inhibitor, which will be marketed as Eucrisa, was developed by Anacor Pharmaceuticals, which Pfizer acquired in May of 2016.
“Today’s approval provides another treatment option for patients dealing with mild to moderate atopic dermatitis,” Amy Egan, deputy director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research, said in a prepared statement. Safety and efficacy of the drug were established in two placebo-controlled trials with a total of 1,522 participants aged 2-79 years with mild to moderate atopic dermatitis.
At the annual meeting of the Pacific Dermatologic Association in August 2016, Dr. Kelly Cordoro, a pediatric dermatologist at the University of California, San Francisco, described crisaborole as an anti-inflammatory agent that modifies inflammation by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signaling pathways. “Crisaborole has shown promising results from four clinical studies in patients 2 years of age and older, with notable improvements in all atopic dermatitis parameters,” she said.
The results of the two phase III studies were recently published in the Journal of the American Academy of Dermatology (2016 Sept;75[3]:494-503).
The Food and Drug Administration has approved crisaborole topical ointment, 2%, to treat mild to moderate atopic dermatitis in patients 2 years of age and older. The boron-based phosphodiesterase 4 inhibitor, which will be marketed as Eucrisa, was developed by Anacor Pharmaceuticals, which Pfizer acquired in May of 2016.
“Today’s approval provides another treatment option for patients dealing with mild to moderate atopic dermatitis,” Amy Egan, deputy director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research, said in a prepared statement. Safety and efficacy of the drug were established in two placebo-controlled trials with a total of 1,522 participants aged 2-79 years with mild to moderate atopic dermatitis.
At the annual meeting of the Pacific Dermatologic Association in August 2016, Dr. Kelly Cordoro, a pediatric dermatologist at the University of California, San Francisco, described crisaborole as an anti-inflammatory agent that modifies inflammation by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signaling pathways. “Crisaborole has shown promising results from four clinical studies in patients 2 years of age and older, with notable improvements in all atopic dermatitis parameters,” she said.
The results of the two phase III studies were recently published in the Journal of the American Academy of Dermatology (2016 Sept;75[3]:494-503).
The Food and Drug Administration has approved crisaborole topical ointment, 2%, to treat mild to moderate atopic dermatitis in patients 2 years of age and older. The boron-based phosphodiesterase 4 inhibitor, which will be marketed as Eucrisa, was developed by Anacor Pharmaceuticals, which Pfizer acquired in May of 2016.
“Today’s approval provides another treatment option for patients dealing with mild to moderate atopic dermatitis,” Amy Egan, deputy director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research, said in a prepared statement. Safety and efficacy of the drug were established in two placebo-controlled trials with a total of 1,522 participants aged 2-79 years with mild to moderate atopic dermatitis.
At the annual meeting of the Pacific Dermatologic Association in August 2016, Dr. Kelly Cordoro, a pediatric dermatologist at the University of California, San Francisco, described crisaborole as an anti-inflammatory agent that modifies inflammation by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signaling pathways. “Crisaborole has shown promising results from four clinical studies in patients 2 years of age and older, with notable improvements in all atopic dermatitis parameters,” she said.
The results of the two phase III studies were recently published in the Journal of the American Academy of Dermatology (2016 Sept;75[3]:494-503).
Daily moisturizing to prevent AD found cost effective
Daily full-body moisturizing of babies from birth to 6 months of age was cost effective and may prove to be a simple preventive strategy to reduce the burden of atopic dermatitis (AD), according to a report published online on Dec. 5 in JAMA Pediatrics.
The annual cost of AD in the United States is estimated at $364 million to $3.8 billion. Preliminary studies have suggested that applying moisturizers every day for the first several months of life to babies at high risk of developing AD reduces the cumulative incidence of the disorder by approximately 50%, said Shuai Xu, MD, of the department of dermatology, Northwestern University, Chicago, and his associates.
The average amount of moisturizer needed was 3.6 g/day at birth, increasing to 6.6 g/day at age 6 months. The cost for these amounts ranged from $0.13 per ounce to $2.96 per ounce for the seven moisturizers. Petroleum jelly was the most affordable product, costing just $7.30 for a 6-month supply, and Vaniply ointment was the most expensive, costing $173.39 for a 6-month supply. The costs of Aveeno Eczema Therapy moisturizing cream, Cetaphil moisturizing cream, CeraVe moisturizing cream, Aquaphor Baby Healing ointment, and sunflower-seed oil fell between the costs of these two products.
For preventing AD, petroleum jelly was the most cost-effective product at $353 per QALY and Vaniply ointment was the least cost effective at $8,386 per QALY. All the moisturizers easily met the widely accepted threshold for cost effectiveness of $38,000 per QALY, Dr. Xu and his associates said. “Beyond the direct cost savings in preventing atopic dermatitis, preserving the skin barrier early in life for high-risk individuals may theoretically reduce the risk of developing other atopic diseases. For instance, neonatal skin barrier dysfunction is associated with food allergies at 2 years of age,” they noted.
“Furthermore, prophylactic moisturization may mitigate the risk of the occurrence of a growing list of atopic dermatitis comorbidities, which include sleep disturbances, obesity, anemia, and attention-deficit/hyperactivity disorder.”
This study was limited in that it did not include any human participants and did not measure the actual development of AD throughout childhood, but instead relied on mathematical estimates and predictions. “Larger-scale studies with longer follow-up will determine whether prophylactic moisturization simply delays the onset of atopic dermatitis or alters the actual disease course,” Dr. Xu and his associates wrote.
No sponsor was cited for this study. Dr. Xu reported being the founder and an equity owner of a website providing safe product recommendations for patients with AD, which has no financial relationships with makers of any skin products. He also reported receiving a one-time travel award from Aquaphor manufacturer Beiersdorf to present research at a medical conference. One of his coauthors reported being a consultant and/or advisor for Anacor/Pfizer, Exeltis, Galderma, Johnson & Johnson, Pierre Fabre, Regeneron, Sanofi, Theraplex, and Valeant.
Daily full-body moisturizing of babies from birth to 6 months of age was cost effective and may prove to be a simple preventive strategy to reduce the burden of atopic dermatitis (AD), according to a report published online on Dec. 5 in JAMA Pediatrics.
The annual cost of AD in the United States is estimated at $364 million to $3.8 billion. Preliminary studies have suggested that applying moisturizers every day for the first several months of life to babies at high risk of developing AD reduces the cumulative incidence of the disorder by approximately 50%, said Shuai Xu, MD, of the department of dermatology, Northwestern University, Chicago, and his associates.
The average amount of moisturizer needed was 3.6 g/day at birth, increasing to 6.6 g/day at age 6 months. The cost for these amounts ranged from $0.13 per ounce to $2.96 per ounce for the seven moisturizers. Petroleum jelly was the most affordable product, costing just $7.30 for a 6-month supply, and Vaniply ointment was the most expensive, costing $173.39 for a 6-month supply. The costs of Aveeno Eczema Therapy moisturizing cream, Cetaphil moisturizing cream, CeraVe moisturizing cream, Aquaphor Baby Healing ointment, and sunflower-seed oil fell between the costs of these two products.
For preventing AD, petroleum jelly was the most cost-effective product at $353 per QALY and Vaniply ointment was the least cost effective at $8,386 per QALY. All the moisturizers easily met the widely accepted threshold for cost effectiveness of $38,000 per QALY, Dr. Xu and his associates said. “Beyond the direct cost savings in preventing atopic dermatitis, preserving the skin barrier early in life for high-risk individuals may theoretically reduce the risk of developing other atopic diseases. For instance, neonatal skin barrier dysfunction is associated with food allergies at 2 years of age,” they noted.
“Furthermore, prophylactic moisturization may mitigate the risk of the occurrence of a growing list of atopic dermatitis comorbidities, which include sleep disturbances, obesity, anemia, and attention-deficit/hyperactivity disorder.”
This study was limited in that it did not include any human participants and did not measure the actual development of AD throughout childhood, but instead relied on mathematical estimates and predictions. “Larger-scale studies with longer follow-up will determine whether prophylactic moisturization simply delays the onset of atopic dermatitis or alters the actual disease course,” Dr. Xu and his associates wrote.
No sponsor was cited for this study. Dr. Xu reported being the founder and an equity owner of a website providing safe product recommendations for patients with AD, which has no financial relationships with makers of any skin products. He also reported receiving a one-time travel award from Aquaphor manufacturer Beiersdorf to present research at a medical conference. One of his coauthors reported being a consultant and/or advisor for Anacor/Pfizer, Exeltis, Galderma, Johnson & Johnson, Pierre Fabre, Regeneron, Sanofi, Theraplex, and Valeant.
Daily full-body moisturizing of babies from birth to 6 months of age was cost effective and may prove to be a simple preventive strategy to reduce the burden of atopic dermatitis (AD), according to a report published online on Dec. 5 in JAMA Pediatrics.
The annual cost of AD in the United States is estimated at $364 million to $3.8 billion. Preliminary studies have suggested that applying moisturizers every day for the first several months of life to babies at high risk of developing AD reduces the cumulative incidence of the disorder by approximately 50%, said Shuai Xu, MD, of the department of dermatology, Northwestern University, Chicago, and his associates.
The average amount of moisturizer needed was 3.6 g/day at birth, increasing to 6.6 g/day at age 6 months. The cost for these amounts ranged from $0.13 per ounce to $2.96 per ounce for the seven moisturizers. Petroleum jelly was the most affordable product, costing just $7.30 for a 6-month supply, and Vaniply ointment was the most expensive, costing $173.39 for a 6-month supply. The costs of Aveeno Eczema Therapy moisturizing cream, Cetaphil moisturizing cream, CeraVe moisturizing cream, Aquaphor Baby Healing ointment, and sunflower-seed oil fell between the costs of these two products.
For preventing AD, petroleum jelly was the most cost-effective product at $353 per QALY and Vaniply ointment was the least cost effective at $8,386 per QALY. All the moisturizers easily met the widely accepted threshold for cost effectiveness of $38,000 per QALY, Dr. Xu and his associates said. “Beyond the direct cost savings in preventing atopic dermatitis, preserving the skin barrier early in life for high-risk individuals may theoretically reduce the risk of developing other atopic diseases. For instance, neonatal skin barrier dysfunction is associated with food allergies at 2 years of age,” they noted.
“Furthermore, prophylactic moisturization may mitigate the risk of the occurrence of a growing list of atopic dermatitis comorbidities, which include sleep disturbances, obesity, anemia, and attention-deficit/hyperactivity disorder.”
This study was limited in that it did not include any human participants and did not measure the actual development of AD throughout childhood, but instead relied on mathematical estimates and predictions. “Larger-scale studies with longer follow-up will determine whether prophylactic moisturization simply delays the onset of atopic dermatitis or alters the actual disease course,” Dr. Xu and his associates wrote.
No sponsor was cited for this study. Dr. Xu reported being the founder and an equity owner of a website providing safe product recommendations for patients with AD, which has no financial relationships with makers of any skin products. He also reported receiving a one-time travel award from Aquaphor manufacturer Beiersdorf to present research at a medical conference. One of his coauthors reported being a consultant and/or advisor for Anacor/Pfizer, Exeltis, Galderma, Johnson & Johnson, Pierre Fabre, Regeneron, Sanofi, Theraplex, and Valeant.
FROM JAMA PEDIATRICS
Key clinical point: Daily full-body moisturizing from birth to 6 months of age was cost effective and may prove to be a simple preventive strategy for atopic dermatitis (AD).
Major finding: For preventing AD, the seven moisturizers easily met the accepted threshold for cost effectiveness of $38,000 per QALY.
Data source: A cost-effectiveness analysis based on calculations of the body surface area of hypothetical babies, the price of seven common moisturizers, and previously reported estimates of risk reduction for AD.
Disclosures: No sponsor was cited for this study. Dr. Xu reported being the founder and an equity owner of a website providing safe product recommendations for patients with atopic dermatitis, which has no financial relationships with makers of any skin products. He also reported receiving a one-time travel award from Beiersdorf to present research at a medical conference. One of his coauthors reported being a consultant and/or advisor to Anacor/Pfizer, Exeltis, Galderma, Johnson & Johnson, Pierre Fabre, Regeneron, Sanofi, Theraplex, and Valeant.
Parents look online for atopic dermatitis advice
Many caregivers of children with atopic dermatitis do not receive clear instructions on how to properly use topical corticosteroids and seek advice on online forums, according to Emma Teasdale, PhD, and her associates.
The investigators analyzed 27 forum discussions involving 95 participants from 2003 to 2015 and found that parents expressed a range of beliefs regarding the use of topical corticosteroids. Some parents expressed positive views, but many were cautious and perceived topical corticosteroids as unnatural or too strong. Notably, parents said they believed that topical corticosteroids thinned or weakened the skin.
Parents also expressed uncertainty over how to use topical corticosteroids. Common questions involved duration of use, where and when to apply, and confusion over the strength of different preparations. Parents also noted that they received conflicting physician instructions regarding duration, dosage, tapering, and safety.
“Given the prevalence of concerns about potential adverse effects of topical-corticosteroids, it would seem prudent to signpost parents/carers towards convenient, consistent, evidence-based information to ensure that the potential negative impacts of seeking (unsubstantiated) medical advice online are minimized. In the absence of such information they are likely to turn to online discussion forums as their sole resource where, although much useful support and advice can be found, some is of questionable validity,” the investigators concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.15130).
Many caregivers of children with atopic dermatitis do not receive clear instructions on how to properly use topical corticosteroids and seek advice on online forums, according to Emma Teasdale, PhD, and her associates.
The investigators analyzed 27 forum discussions involving 95 participants from 2003 to 2015 and found that parents expressed a range of beliefs regarding the use of topical corticosteroids. Some parents expressed positive views, but many were cautious and perceived topical corticosteroids as unnatural or too strong. Notably, parents said they believed that topical corticosteroids thinned or weakened the skin.
Parents also expressed uncertainty over how to use topical corticosteroids. Common questions involved duration of use, where and when to apply, and confusion over the strength of different preparations. Parents also noted that they received conflicting physician instructions regarding duration, dosage, tapering, and safety.
“Given the prevalence of concerns about potential adverse effects of topical-corticosteroids, it would seem prudent to signpost parents/carers towards convenient, consistent, evidence-based information to ensure that the potential negative impacts of seeking (unsubstantiated) medical advice online are minimized. In the absence of such information they are likely to turn to online discussion forums as their sole resource where, although much useful support and advice can be found, some is of questionable validity,” the investigators concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.15130).
Many caregivers of children with atopic dermatitis do not receive clear instructions on how to properly use topical corticosteroids and seek advice on online forums, according to Emma Teasdale, PhD, and her associates.
The investigators analyzed 27 forum discussions involving 95 participants from 2003 to 2015 and found that parents expressed a range of beliefs regarding the use of topical corticosteroids. Some parents expressed positive views, but many were cautious and perceived topical corticosteroids as unnatural or too strong. Notably, parents said they believed that topical corticosteroids thinned or weakened the skin.
Parents also expressed uncertainty over how to use topical corticosteroids. Common questions involved duration of use, where and when to apply, and confusion over the strength of different preparations. Parents also noted that they received conflicting physician instructions regarding duration, dosage, tapering, and safety.
“Given the prevalence of concerns about potential adverse effects of topical-corticosteroids, it would seem prudent to signpost parents/carers towards convenient, consistent, evidence-based information to ensure that the potential negative impacts of seeking (unsubstantiated) medical advice online are minimized. In the absence of such information they are likely to turn to online discussion forums as their sole resource where, although much useful support and advice can be found, some is of questionable validity,” the investigators concluded.
Find the full study in the British Journal of Dermatology (doi: 10.1111/bjd.15130).
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Topical tofacitinib shows promise in atopic dermatitis
Topical tofacitinib showed significant improvements across all endpoints and for pruritus at week 4, compared with vehicle, results of a phase IIa trial have shown.
Tofacitinib is a Janus kinase inhibitor that affects the interleukin (IL)–4, IL-5, and IL-31 signaling pathways, interfering with the immune response that leads to inflammation.
The study could mean “that inhibition of the JAK-STAT pathway may be a new therapeutic target for AD,” wrote the study’s lead author, Robert Bissonnette, MD, president of Innovaderm Research in Montreal. The study was published in the British Journal of Dermatology (2016 Nov;175[5]:902-11).
In the multicenter, double-blind, controlled study of 69 adults with mild to moderate atopic dermatitis randomly assigned to either 2% tofacitinib or vehicle ointment twice daily, the study group achieved an 81.7% mean reduction in baseline Eczema Area and Severity Index (EASI) score, compared with 29.9% of controls over the 4-week study period (P less than .001). EASI scores in the study group were about 80% at a score of 50, 60% at a score of 75, and 40% at a score of 90.
By week 4, about three-quarters of the study group were either clear or almost clear of their skin condition, according to the physician global assessment scale, compared with 22% of controls (P less than .05).
There also was a rapid reduction in patient-reported pruritus in the tofacitinib group per the Itch Severity Item scale, compared with controls, at weeks 2 and 4 (P less than .001 for each time point).
Tolerability was similar across the study, and treatment-related adverse effects were mild, although 44% of the tofacitinib group did report experiencing some form of infection, infestation, or other complication. Two people in the study group dropped out because of the severity of their treatment-emergent adverse events. There were no reported severe or serious infections.
Dr. Bissonnette has numerous pharmaceutical industry relationships, including with Pfizer, the study’s sponsor.
With the discovery of how cytokines such as IL-4, IL-5, IL-13, and IL-31 drive inflammatory disease pathogenesis, more targeted therapies are possible for dermatologic conditions such as atopic dermatitis.
The promise of such pathogenesis-based treatments gives hope to patients with atopic dermatitis, for whom new treatments have not been brought to market in more than 15 years.
While this is reason for excitement, the emergence of several new promising therapies calls for comparison trials, according to Brett A. King, MD, and William Damsky, MD, PhD, both of Yale University, New Haven, Conn.
“Further studies will be needed to address long-term efficacy and safety,” Dr. King and Dr. Damsky noted. The results of Dr. Bissonnette’s phase IIa trial of the topical Janus kinase inhibitor tofacitinib mean there is potentially a third targeted topical agent to emerge as a treatment for atopic dermatitis. The others include the phosphodiesterase-4 inhibitor crisaborole, and dupilumab, a monoclonal antibody that targets IL-4 and IL-13.
“Head-to-head trials involving these agents and superpotent topical steroids would be useful in establishing their place in AD treatment algorithms,” Dr. King and Dr. Damsky said.
Dr. King is an assistant professor of dermatology at Yale University. His coauthor, Dr. Damsky, is a second-year resident in dermatology at Yale University. These remarks are taken from an editorial accompanying Dr. Bissonnette’s study (Br J Dermatol. 2016 Nov;175[5]:861-2). Dr. King disclosed he has industry ties with Eli Lilly and Pfizer, among others. Dr. Damsky had no relevant disclosures.
With the discovery of how cytokines such as IL-4, IL-5, IL-13, and IL-31 drive inflammatory disease pathogenesis, more targeted therapies are possible for dermatologic conditions such as atopic dermatitis.
The promise of such pathogenesis-based treatments gives hope to patients with atopic dermatitis, for whom new treatments have not been brought to market in more than 15 years.
While this is reason for excitement, the emergence of several new promising therapies calls for comparison trials, according to Brett A. King, MD, and William Damsky, MD, PhD, both of Yale University, New Haven, Conn.
“Further studies will be needed to address long-term efficacy and safety,” Dr. King and Dr. Damsky noted. The results of Dr. Bissonnette’s phase IIa trial of the topical Janus kinase inhibitor tofacitinib mean there is potentially a third targeted topical agent to emerge as a treatment for atopic dermatitis. The others include the phosphodiesterase-4 inhibitor crisaborole, and dupilumab, a monoclonal antibody that targets IL-4 and IL-13.
“Head-to-head trials involving these agents and superpotent topical steroids would be useful in establishing their place in AD treatment algorithms,” Dr. King and Dr. Damsky said.
Dr. King is an assistant professor of dermatology at Yale University. His coauthor, Dr. Damsky, is a second-year resident in dermatology at Yale University. These remarks are taken from an editorial accompanying Dr. Bissonnette’s study (Br J Dermatol. 2016 Nov;175[5]:861-2). Dr. King disclosed he has industry ties with Eli Lilly and Pfizer, among others. Dr. Damsky had no relevant disclosures.
With the discovery of how cytokines such as IL-4, IL-5, IL-13, and IL-31 drive inflammatory disease pathogenesis, more targeted therapies are possible for dermatologic conditions such as atopic dermatitis.
The promise of such pathogenesis-based treatments gives hope to patients with atopic dermatitis, for whom new treatments have not been brought to market in more than 15 years.
While this is reason for excitement, the emergence of several new promising therapies calls for comparison trials, according to Brett A. King, MD, and William Damsky, MD, PhD, both of Yale University, New Haven, Conn.
“Further studies will be needed to address long-term efficacy and safety,” Dr. King and Dr. Damsky noted. The results of Dr. Bissonnette’s phase IIa trial of the topical Janus kinase inhibitor tofacitinib mean there is potentially a third targeted topical agent to emerge as a treatment for atopic dermatitis. The others include the phosphodiesterase-4 inhibitor crisaborole, and dupilumab, a monoclonal antibody that targets IL-4 and IL-13.
“Head-to-head trials involving these agents and superpotent topical steroids would be useful in establishing their place in AD treatment algorithms,” Dr. King and Dr. Damsky said.
Dr. King is an assistant professor of dermatology at Yale University. His coauthor, Dr. Damsky, is a second-year resident in dermatology at Yale University. These remarks are taken from an editorial accompanying Dr. Bissonnette’s study (Br J Dermatol. 2016 Nov;175[5]:861-2). Dr. King disclosed he has industry ties with Eli Lilly and Pfizer, among others. Dr. Damsky had no relevant disclosures.
Topical tofacitinib showed significant improvements across all endpoints and for pruritus at week 4, compared with vehicle, results of a phase IIa trial have shown.
Tofacitinib is a Janus kinase inhibitor that affects the interleukin (IL)–4, IL-5, and IL-31 signaling pathways, interfering with the immune response that leads to inflammation.
The study could mean “that inhibition of the JAK-STAT pathway may be a new therapeutic target for AD,” wrote the study’s lead author, Robert Bissonnette, MD, president of Innovaderm Research in Montreal. The study was published in the British Journal of Dermatology (2016 Nov;175[5]:902-11).
In the multicenter, double-blind, controlled study of 69 adults with mild to moderate atopic dermatitis randomly assigned to either 2% tofacitinib or vehicle ointment twice daily, the study group achieved an 81.7% mean reduction in baseline Eczema Area and Severity Index (EASI) score, compared with 29.9% of controls over the 4-week study period (P less than .001). EASI scores in the study group were about 80% at a score of 50, 60% at a score of 75, and 40% at a score of 90.
By week 4, about three-quarters of the study group were either clear or almost clear of their skin condition, according to the physician global assessment scale, compared with 22% of controls (P less than .05).
There also was a rapid reduction in patient-reported pruritus in the tofacitinib group per the Itch Severity Item scale, compared with controls, at weeks 2 and 4 (P less than .001 for each time point).
Tolerability was similar across the study, and treatment-related adverse effects were mild, although 44% of the tofacitinib group did report experiencing some form of infection, infestation, or other complication. Two people in the study group dropped out because of the severity of their treatment-emergent adverse events. There were no reported severe or serious infections.
Dr. Bissonnette has numerous pharmaceutical industry relationships, including with Pfizer, the study’s sponsor.
Topical tofacitinib showed significant improvements across all endpoints and for pruritus at week 4, compared with vehicle, results of a phase IIa trial have shown.
Tofacitinib is a Janus kinase inhibitor that affects the interleukin (IL)–4, IL-5, and IL-31 signaling pathways, interfering with the immune response that leads to inflammation.
The study could mean “that inhibition of the JAK-STAT pathway may be a new therapeutic target for AD,” wrote the study’s lead author, Robert Bissonnette, MD, president of Innovaderm Research in Montreal. The study was published in the British Journal of Dermatology (2016 Nov;175[5]:902-11).
In the multicenter, double-blind, controlled study of 69 adults with mild to moderate atopic dermatitis randomly assigned to either 2% tofacitinib or vehicle ointment twice daily, the study group achieved an 81.7% mean reduction in baseline Eczema Area and Severity Index (EASI) score, compared with 29.9% of controls over the 4-week study period (P less than .001). EASI scores in the study group were about 80% at a score of 50, 60% at a score of 75, and 40% at a score of 90.
By week 4, about three-quarters of the study group were either clear or almost clear of their skin condition, according to the physician global assessment scale, compared with 22% of controls (P less than .05).
There also was a rapid reduction in patient-reported pruritus in the tofacitinib group per the Itch Severity Item scale, compared with controls, at weeks 2 and 4 (P less than .001 for each time point).
Tolerability was similar across the study, and treatment-related adverse effects were mild, although 44% of the tofacitinib group did report experiencing some form of infection, infestation, or other complication. Two people in the study group dropped out because of the severity of their treatment-emergent adverse events. There were no reported severe or serious infections.
Dr. Bissonnette has numerous pharmaceutical industry relationships, including with Pfizer, the study’s sponsor.
FROM BRITISH JOURNAL OF DERMATOLOGY
Key clinical point:
Major finding: Tofacitinib showed significant improvements across all endpoints and for pruritus at week 4, compared with vehicle (P less than .001)
Data source: Multicenter, phase IIa, 4-week, double-blind, controlled study of 69 adults with mild to moderate atopic dermatitis randomly assigned to either 2% tofacitinib or vehicle ointment twice daily.
Disclosures: Dr. Bissonnette has numerous pharmaceutical industry relationships, including with Pfizer, the sponsor of this study.
Bioactive lipid shows promise in atopic dermatitis
VIENNA – A novel oral agent known as DS107 showed promise as a safe and effective treatment for moderate-to-severe atopic dermatitis in a phase IIa proof-of-concept study, Diamant Thaçi, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Clear efficacy signals in the reduction of clinical symptoms of atopic dermatitis were detected within 2 weeks of treatment, with the maximum improvement in the endpoints observed between weeks 4 and 8,” said Dr. Thaçi, professor of dermatology and head of the Comprehensive Center for Inflammation Medicine at University Hospital Schleswig-Holstein, in Lübeck, Germany.
Dr. Thaçi presented the results of an 8-week, double-blind, randomized, placebo-controlled, multicenter phase IIa study that included 102 patients with moderate-to-severe AD. Participants averaged a baseline Investigator Global Assessment (IGA) score of 3.5 on the 0-5 scale. They were randomized to 2 g of oral DS107 once daily or an equal quantity of mineral oil as a placebo control.
Based upon the encouraging findings, a 300-patient phase IIb study will get underway soon. It will examine the effects of 1 g of DS107 per day as well as 2 g in the hope that the lower dose will cut down on the high rate of minor GI side effects seen at 2 g/day while preserving the efficacy of the higher dose.
The primary efficacy endpoint in the phase IIa study was at least a 2-point drop from baseline in IGA plus an end-of-treatment IGA of 0 or 1, meaning clear or almost clear. In the intent-to-treat analysis, this was achieved in 21.6% of the group assigned to DS107, compared with 11.8% of controls. In the 71 participants who actually completed 8 weeks of treatment – 35 in the DS107 arm, 36 controls – the composite efficacy endpoint was achieved in 31.4% of those on active treatment and 16.7% on mineral oil.
Significant separation between the active treatment and control arms in terms of itch visual analog scores was seen by week 4. This was a particularly encouraging finding, since patients report pruritis to be the most troublesome symptom of AD, Dr. Thaçi noted.
Significantly greater improvement in quality of life as measured by the Patient-Oriented Eczema Measure (POEM) was also seen by week 4 in the DS107 group as compared with controls.
No severe adverse events occurred in the study. However, more than one-quarter of subjects interrupted or discontinued participation because of mild nausea, loose stools, and/or abdominal pain. These issues were equally common in the DS107 and mineral oil groups, and Dr. Thaçi and his coinvestigators suspect that for many patients it was simply a matter of too much oil in the stomach. The GI symptoms resolved quickly without intervention after a brief halt of therapy, but some patients never returned to participation.
“This problem can be solved in the future with a different dosing design or even a different method of delivering the DGLA,” the dermatologist added.
Asked about the significant placebo response seen in the study, Dr. Thaçi shrugged it off as “quite understandable.”
“Placebo is not always a placebo. There is the feeling of fullness in the stomach, there is some emollient effect, the continuous contact with the physician. You see this in all the clinical trials in atopic dermatitis: in the beginning, the first 2-3 weeks, you have some influence of placebo,” he said.
The placebo effect was greatly diminished in patients with more severe AD. In the subset with a baseline IGA of 4 or 5, none of the control subjects achieved the primary efficacy endpoint, while more than 20% on DS107 did, Dr. Thaçi noted.
The mechanism of benefit of DGLA is not fully understood as yet. Animal studies point to an antibacterial effect, and DGLA also reduces levels of inflammatory cytokines. Eosinophilia was reduced to a much greater extent in the DS107 group than controls.
Dr. Thaçi reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
bjancin@frontlinemedcom.com
VIENNA – A novel oral agent known as DS107 showed promise as a safe and effective treatment for moderate-to-severe atopic dermatitis in a phase IIa proof-of-concept study, Diamant Thaçi, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Clear efficacy signals in the reduction of clinical symptoms of atopic dermatitis were detected within 2 weeks of treatment, with the maximum improvement in the endpoints observed between weeks 4 and 8,” said Dr. Thaçi, professor of dermatology and head of the Comprehensive Center for Inflammation Medicine at University Hospital Schleswig-Holstein, in Lübeck, Germany.
Dr. Thaçi presented the results of an 8-week, double-blind, randomized, placebo-controlled, multicenter phase IIa study that included 102 patients with moderate-to-severe AD. Participants averaged a baseline Investigator Global Assessment (IGA) score of 3.5 on the 0-5 scale. They were randomized to 2 g of oral DS107 once daily or an equal quantity of mineral oil as a placebo control.
Based upon the encouraging findings, a 300-patient phase IIb study will get underway soon. It will examine the effects of 1 g of DS107 per day as well as 2 g in the hope that the lower dose will cut down on the high rate of minor GI side effects seen at 2 g/day while preserving the efficacy of the higher dose.
The primary efficacy endpoint in the phase IIa study was at least a 2-point drop from baseline in IGA plus an end-of-treatment IGA of 0 or 1, meaning clear or almost clear. In the intent-to-treat analysis, this was achieved in 21.6% of the group assigned to DS107, compared with 11.8% of controls. In the 71 participants who actually completed 8 weeks of treatment – 35 in the DS107 arm, 36 controls – the composite efficacy endpoint was achieved in 31.4% of those on active treatment and 16.7% on mineral oil.
Significant separation between the active treatment and control arms in terms of itch visual analog scores was seen by week 4. This was a particularly encouraging finding, since patients report pruritis to be the most troublesome symptom of AD, Dr. Thaçi noted.
Significantly greater improvement in quality of life as measured by the Patient-Oriented Eczema Measure (POEM) was also seen by week 4 in the DS107 group as compared with controls.
No severe adverse events occurred in the study. However, more than one-quarter of subjects interrupted or discontinued participation because of mild nausea, loose stools, and/or abdominal pain. These issues were equally common in the DS107 and mineral oil groups, and Dr. Thaçi and his coinvestigators suspect that for many patients it was simply a matter of too much oil in the stomach. The GI symptoms resolved quickly without intervention after a brief halt of therapy, but some patients never returned to participation.
“This problem can be solved in the future with a different dosing design or even a different method of delivering the DGLA,” the dermatologist added.
Asked about the significant placebo response seen in the study, Dr. Thaçi shrugged it off as “quite understandable.”
“Placebo is not always a placebo. There is the feeling of fullness in the stomach, there is some emollient effect, the continuous contact with the physician. You see this in all the clinical trials in atopic dermatitis: in the beginning, the first 2-3 weeks, you have some influence of placebo,” he said.
The placebo effect was greatly diminished in patients with more severe AD. In the subset with a baseline IGA of 4 or 5, none of the control subjects achieved the primary efficacy endpoint, while more than 20% on DS107 did, Dr. Thaçi noted.
The mechanism of benefit of DGLA is not fully understood as yet. Animal studies point to an antibacterial effect, and DGLA also reduces levels of inflammatory cytokines. Eosinophilia was reduced to a much greater extent in the DS107 group than controls.
Dr. Thaçi reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
bjancin@frontlinemedcom.com
VIENNA – A novel oral agent known as DS107 showed promise as a safe and effective treatment for moderate-to-severe atopic dermatitis in a phase IIa proof-of-concept study, Diamant Thaçi, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Clear efficacy signals in the reduction of clinical symptoms of atopic dermatitis were detected within 2 weeks of treatment, with the maximum improvement in the endpoints observed between weeks 4 and 8,” said Dr. Thaçi, professor of dermatology and head of the Comprehensive Center for Inflammation Medicine at University Hospital Schleswig-Holstein, in Lübeck, Germany.
Dr. Thaçi presented the results of an 8-week, double-blind, randomized, placebo-controlled, multicenter phase IIa study that included 102 patients with moderate-to-severe AD. Participants averaged a baseline Investigator Global Assessment (IGA) score of 3.5 on the 0-5 scale. They were randomized to 2 g of oral DS107 once daily or an equal quantity of mineral oil as a placebo control.
Based upon the encouraging findings, a 300-patient phase IIb study will get underway soon. It will examine the effects of 1 g of DS107 per day as well as 2 g in the hope that the lower dose will cut down on the high rate of minor GI side effects seen at 2 g/day while preserving the efficacy of the higher dose.
The primary efficacy endpoint in the phase IIa study was at least a 2-point drop from baseline in IGA plus an end-of-treatment IGA of 0 or 1, meaning clear or almost clear. In the intent-to-treat analysis, this was achieved in 21.6% of the group assigned to DS107, compared with 11.8% of controls. In the 71 participants who actually completed 8 weeks of treatment – 35 in the DS107 arm, 36 controls – the composite efficacy endpoint was achieved in 31.4% of those on active treatment and 16.7% on mineral oil.
Significant separation between the active treatment and control arms in terms of itch visual analog scores was seen by week 4. This was a particularly encouraging finding, since patients report pruritis to be the most troublesome symptom of AD, Dr. Thaçi noted.
Significantly greater improvement in quality of life as measured by the Patient-Oriented Eczema Measure (POEM) was also seen by week 4 in the DS107 group as compared with controls.
No severe adverse events occurred in the study. However, more than one-quarter of subjects interrupted or discontinued participation because of mild nausea, loose stools, and/or abdominal pain. These issues were equally common in the DS107 and mineral oil groups, and Dr. Thaçi and his coinvestigators suspect that for many patients it was simply a matter of too much oil in the stomach. The GI symptoms resolved quickly without intervention after a brief halt of therapy, but some patients never returned to participation.
“This problem can be solved in the future with a different dosing design or even a different method of delivering the DGLA,” the dermatologist added.
Asked about the significant placebo response seen in the study, Dr. Thaçi shrugged it off as “quite understandable.”
“Placebo is not always a placebo. There is the feeling of fullness in the stomach, there is some emollient effect, the continuous contact with the physician. You see this in all the clinical trials in atopic dermatitis: in the beginning, the first 2-3 weeks, you have some influence of placebo,” he said.
The placebo effect was greatly diminished in patients with more severe AD. In the subset with a baseline IGA of 4 or 5, none of the control subjects achieved the primary efficacy endpoint, while more than 20% on DS107 did, Dr. Thaçi noted.
The mechanism of benefit of DGLA is not fully understood as yet. Animal studies point to an antibacterial effect, and DGLA also reduces levels of inflammatory cytokines. Eosinophilia was reduced to a much greater extent in the DS107 group than controls.
Dr. Thaçi reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
bjancin@frontlinemedcom.com
AT THE EADV CONGRESS
Key clinical point:
Major finding: Once-daily oral DS107, which contains a bioactive lipid, achieved significant improvement in moderate-to-severe atopic dermatitis in 31.4% of patients, compared with 16.7% on placebo.
Data source: This phase IIa, double-blind, randomized, placebo-controlled, multicenter 8-week trial included 102 adults with moderate-to-severe atopic dermatitis.
Disclosures: The study presenter reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
Topical crisaborole boosts quality of life in atopic dermatitis
VIENNA – Topical crisaborole 2% ointment administered twice a day was consistently associated with clinically meaningful quality of life improvement scores on multiple measures in the two pivotal phase III, randomized, controlled trials of atopic dermatitis (AD) patients aged 2 years old through adulthood, Amy S. Paller, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
AD in children and adolescents is infamous for the adverse quality of life impact it imposes upon the patients’ parents, family, and caregivers. So the significant improvement seen with crisaborole, compared with its vehicle on the Dermatitis Family Impact (DFI) Questionnaire was particularly gratifying. The DFI questionnaire assesses quality of life in key domains, including family, parent, and caregiver sleep, emotional distress, relationships, family leisure, and ability to do housework or go shopping.
“If approved, crisaborole ... could improve the quality of life for patients with mild to moderate atopic dermatitis and, very importantly, for their families as well,” declared Dr. Paller, professor of dermatology and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Crisaborole’s developer, Anacor Pharmaceuticals, has filed an application for approval for treating mild to moderate AD in patients aged 2 years and older, now under review at the Food and Drug Administration.
In a separate presentation at the EADV congress, Lawrence F. Eichenfield, MD, presented the results of a long-term, open-label crisaborole safety study of 48-52 weeks duration. The long-term study involved 517 participants in the two pivotal phase III trials. There were no serious adverse events and no long-term cutaneous adverse events such as the skin atrophy or telangiectasias that can occur with topical steroids. The safety profile was favorable for long-term treatment of patients 2 years of age or older with mild to moderate AD.
Crisaborole 2% topical ointment is a novel, boron-based, nonsteroidal inhibitor of phosphodiesterase 4 (PDE-4). AD is marked by overactivity of PDE-4, which results in decreased levels of cyclic AMP and resultant increased release of inflammatory cytokines.
Dr. Paller noted that, in the previously reported efficacy results of the two pivotal, double-blind, 28-day, phase III trials, crisaborole treatment reduced global disease severity and provided early and sustained improvement in itch severity. She presented the prespecified quality of life results for the two identically designed, parallel pivotal trials, which totaled 1,016 patients on crisaborole and 506 on its vehicle. At baseline, 39% of subjects had mild AD, and 61% had moderate AD. The mean body surface area affected was 18%. Participants’ mean age was 12.3 years, and 14% were aged 8 years old or older.
The structure of the long-term safety study suggests how crisaborole might be used in clinical practice. During the year-long, open-label study, patients were evaluated every 28 days. If their skin was deemed clear or almost clear on the basis of an Investigator’s Static Global Assessment (ISGA) score of 0 or 1, they were taken off crisaborole and could use only emollients for the next 28 days, at which time they would be reevaluated. At that point, if they had an ISGA of 2 or more, they went back on crisaborole twice a day for 28 days until their next evaluation.
Dr. Eichenfield reported that 10.2% of participants in the long-term safety study reported treatment-related adverse events, which were mild to moderate. The most frequently reported of these were mild to moderate flares of AD during a 28-day off-treatment period in 1.3% of patients, application site pain in 2.3%, and application site infection in 1.2%.
Dr. Paller and Dr. Eichenfield reported serving as consultants to Anacor Pharmaceuticals.
The FDA review of crisaborole is expected to be completed by early January 2017, according to Anacor.
VIENNA – Topical crisaborole 2% ointment administered twice a day was consistently associated with clinically meaningful quality of life improvement scores on multiple measures in the two pivotal phase III, randomized, controlled trials of atopic dermatitis (AD) patients aged 2 years old through adulthood, Amy S. Paller, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
AD in children and adolescents is infamous for the adverse quality of life impact it imposes upon the patients’ parents, family, and caregivers. So the significant improvement seen with crisaborole, compared with its vehicle on the Dermatitis Family Impact (DFI) Questionnaire was particularly gratifying. The DFI questionnaire assesses quality of life in key domains, including family, parent, and caregiver sleep, emotional distress, relationships, family leisure, and ability to do housework or go shopping.
“If approved, crisaborole ... could improve the quality of life for patients with mild to moderate atopic dermatitis and, very importantly, for their families as well,” declared Dr. Paller, professor of dermatology and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Crisaborole’s developer, Anacor Pharmaceuticals, has filed an application for approval for treating mild to moderate AD in patients aged 2 years and older, now under review at the Food and Drug Administration.
In a separate presentation at the EADV congress, Lawrence F. Eichenfield, MD, presented the results of a long-term, open-label crisaborole safety study of 48-52 weeks duration. The long-term study involved 517 participants in the two pivotal phase III trials. There were no serious adverse events and no long-term cutaneous adverse events such as the skin atrophy or telangiectasias that can occur with topical steroids. The safety profile was favorable for long-term treatment of patients 2 years of age or older with mild to moderate AD.
Crisaborole 2% topical ointment is a novel, boron-based, nonsteroidal inhibitor of phosphodiesterase 4 (PDE-4). AD is marked by overactivity of PDE-4, which results in decreased levels of cyclic AMP and resultant increased release of inflammatory cytokines.
Dr. Paller noted that, in the previously reported efficacy results of the two pivotal, double-blind, 28-day, phase III trials, crisaborole treatment reduced global disease severity and provided early and sustained improvement in itch severity. She presented the prespecified quality of life results for the two identically designed, parallel pivotal trials, which totaled 1,016 patients on crisaborole and 506 on its vehicle. At baseline, 39% of subjects had mild AD, and 61% had moderate AD. The mean body surface area affected was 18%. Participants’ mean age was 12.3 years, and 14% were aged 8 years old or older.
The structure of the long-term safety study suggests how crisaborole might be used in clinical practice. During the year-long, open-label study, patients were evaluated every 28 days. If their skin was deemed clear or almost clear on the basis of an Investigator’s Static Global Assessment (ISGA) score of 0 or 1, they were taken off crisaborole and could use only emollients for the next 28 days, at which time they would be reevaluated. At that point, if they had an ISGA of 2 or more, they went back on crisaborole twice a day for 28 days until their next evaluation.
Dr. Eichenfield reported that 10.2% of participants in the long-term safety study reported treatment-related adverse events, which were mild to moderate. The most frequently reported of these were mild to moderate flares of AD during a 28-day off-treatment period in 1.3% of patients, application site pain in 2.3%, and application site infection in 1.2%.
Dr. Paller and Dr. Eichenfield reported serving as consultants to Anacor Pharmaceuticals.
The FDA review of crisaborole is expected to be completed by early January 2017, according to Anacor.
VIENNA – Topical crisaborole 2% ointment administered twice a day was consistently associated with clinically meaningful quality of life improvement scores on multiple measures in the two pivotal phase III, randomized, controlled trials of atopic dermatitis (AD) patients aged 2 years old through adulthood, Amy S. Paller, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
AD in children and adolescents is infamous for the adverse quality of life impact it imposes upon the patients’ parents, family, and caregivers. So the significant improvement seen with crisaborole, compared with its vehicle on the Dermatitis Family Impact (DFI) Questionnaire was particularly gratifying. The DFI questionnaire assesses quality of life in key domains, including family, parent, and caregiver sleep, emotional distress, relationships, family leisure, and ability to do housework or go shopping.
“If approved, crisaborole ... could improve the quality of life for patients with mild to moderate atopic dermatitis and, very importantly, for their families as well,” declared Dr. Paller, professor of dermatology and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Crisaborole’s developer, Anacor Pharmaceuticals, has filed an application for approval for treating mild to moderate AD in patients aged 2 years and older, now under review at the Food and Drug Administration.
In a separate presentation at the EADV congress, Lawrence F. Eichenfield, MD, presented the results of a long-term, open-label crisaborole safety study of 48-52 weeks duration. The long-term study involved 517 participants in the two pivotal phase III trials. There were no serious adverse events and no long-term cutaneous adverse events such as the skin atrophy or telangiectasias that can occur with topical steroids. The safety profile was favorable for long-term treatment of patients 2 years of age or older with mild to moderate AD.
Crisaborole 2% topical ointment is a novel, boron-based, nonsteroidal inhibitor of phosphodiesterase 4 (PDE-4). AD is marked by overactivity of PDE-4, which results in decreased levels of cyclic AMP and resultant increased release of inflammatory cytokines.
Dr. Paller noted that, in the previously reported efficacy results of the two pivotal, double-blind, 28-day, phase III trials, crisaborole treatment reduced global disease severity and provided early and sustained improvement in itch severity. She presented the prespecified quality of life results for the two identically designed, parallel pivotal trials, which totaled 1,016 patients on crisaborole and 506 on its vehicle. At baseline, 39% of subjects had mild AD, and 61% had moderate AD. The mean body surface area affected was 18%. Participants’ mean age was 12.3 years, and 14% were aged 8 years old or older.
The structure of the long-term safety study suggests how crisaborole might be used in clinical practice. During the year-long, open-label study, patients were evaluated every 28 days. If their skin was deemed clear or almost clear on the basis of an Investigator’s Static Global Assessment (ISGA) score of 0 or 1, they were taken off crisaborole and could use only emollients for the next 28 days, at which time they would be reevaluated. At that point, if they had an ISGA of 2 or more, they went back on crisaborole twice a day for 28 days until their next evaluation.
Dr. Eichenfield reported that 10.2% of participants in the long-term safety study reported treatment-related adverse events, which were mild to moderate. The most frequently reported of these were mild to moderate flares of AD during a 28-day off-treatment period in 1.3% of patients, application site pain in 2.3%, and application site infection in 1.2%.
Dr. Paller and Dr. Eichenfield reported serving as consultants to Anacor Pharmaceuticals.
The FDA review of crisaborole is expected to be completed by early January 2017, according to Anacor.
EXPERT ANALYSIS FROM THE EADV CONGRESS
Disease severity, QOL outcome measures need standardizing in atopic dermatitis research
The number of outcome measures used to assess disease severity and quality of life (QOL) in randomized controlled trials of patients with atopic dermatitis (AD) has risen in recent years, according to a systematic review reports.
An overall lack of standardization of these outcome measures, however, is hindering the synthesis and translation of research into clinical practice, reported Mary K. Hill of the University of Colorado, Aurora, and her associates.
“Standardization of disease severity and QOL outcome instruments is essential for comparability among studies and improved quality of research evidence,” they wrote (J Am Acad Dermatol. 2016 Nov;75[5]:906-17. doi: 10.1016/j.jaad.2016.07.002).
Their systematic review of 135 randomized controlled trials (RCTs) identified 62 disease-severity and 28 quality-of-life instruments used in studies of patients with AD between July 2010 and July 2015.
This was a drastic increase from the 20 disease severity scales and 14 QOL indices identified in a previous systematic review of 382 RCTs of AD therapies conducted between 1985 and 2010, they noted.
In their review, the most frequently used disease severity scale was the Scoring Atopic Dermatitis (SCORAD) index, which was used in 79 studies. That was followed by the visual analogue scale (VAS) for pruritus, used in 30 studies; the Investigator’s Global Assessment (IGA) tool, used in 29 studies; and the Eczema Area and Severity Index (EASI), used in 28 studies.
But despite the well-documented burden of AD, the researchers noted that only 33% of the RCTs they reviewed assessed QOL. “This is up from the 18% of RCTs on AD that reported QOL outcomes between 1985 and July 2010, perhaps signifying gradually increased attention to patient emotional well-being,” Ms. Hill and her associates wrote.
A trend described by the authors, however, as “perhaps the most disconcerting” was that 75% of identified QOL instruments were used only once. “Continued increases in the reporting of QOL outcomes will be of limited benefit for interstudy comparisons if the diversity of measures used also continues to rise,” they said.
Adding to the confusion, the researchers found frequent overlap in the naming and content of instruments used in studies, making it even more challenging to identify meaningful comparisons.
There was no funding source, and the authors had no conflicts of interest to declare.
The number of outcome measures used to assess disease severity and quality of life (QOL) in randomized controlled trials of patients with atopic dermatitis (AD) has risen in recent years, according to a systematic review reports.
An overall lack of standardization of these outcome measures, however, is hindering the synthesis and translation of research into clinical practice, reported Mary K. Hill of the University of Colorado, Aurora, and her associates.
“Standardization of disease severity and QOL outcome instruments is essential for comparability among studies and improved quality of research evidence,” they wrote (J Am Acad Dermatol. 2016 Nov;75[5]:906-17. doi: 10.1016/j.jaad.2016.07.002).
Their systematic review of 135 randomized controlled trials (RCTs) identified 62 disease-severity and 28 quality-of-life instruments used in studies of patients with AD between July 2010 and July 2015.
This was a drastic increase from the 20 disease severity scales and 14 QOL indices identified in a previous systematic review of 382 RCTs of AD therapies conducted between 1985 and 2010, they noted.
In their review, the most frequently used disease severity scale was the Scoring Atopic Dermatitis (SCORAD) index, which was used in 79 studies. That was followed by the visual analogue scale (VAS) for pruritus, used in 30 studies; the Investigator’s Global Assessment (IGA) tool, used in 29 studies; and the Eczema Area and Severity Index (EASI), used in 28 studies.
But despite the well-documented burden of AD, the researchers noted that only 33% of the RCTs they reviewed assessed QOL. “This is up from the 18% of RCTs on AD that reported QOL outcomes between 1985 and July 2010, perhaps signifying gradually increased attention to patient emotional well-being,” Ms. Hill and her associates wrote.
A trend described by the authors, however, as “perhaps the most disconcerting” was that 75% of identified QOL instruments were used only once. “Continued increases in the reporting of QOL outcomes will be of limited benefit for interstudy comparisons if the diversity of measures used also continues to rise,” they said.
Adding to the confusion, the researchers found frequent overlap in the naming and content of instruments used in studies, making it even more challenging to identify meaningful comparisons.
There was no funding source, and the authors had no conflicts of interest to declare.
The number of outcome measures used to assess disease severity and quality of life (QOL) in randomized controlled trials of patients with atopic dermatitis (AD) has risen in recent years, according to a systematic review reports.
An overall lack of standardization of these outcome measures, however, is hindering the synthesis and translation of research into clinical practice, reported Mary K. Hill of the University of Colorado, Aurora, and her associates.
“Standardization of disease severity and QOL outcome instruments is essential for comparability among studies and improved quality of research evidence,” they wrote (J Am Acad Dermatol. 2016 Nov;75[5]:906-17. doi: 10.1016/j.jaad.2016.07.002).
Their systematic review of 135 randomized controlled trials (RCTs) identified 62 disease-severity and 28 quality-of-life instruments used in studies of patients with AD between July 2010 and July 2015.
This was a drastic increase from the 20 disease severity scales and 14 QOL indices identified in a previous systematic review of 382 RCTs of AD therapies conducted between 1985 and 2010, they noted.
In their review, the most frequently used disease severity scale was the Scoring Atopic Dermatitis (SCORAD) index, which was used in 79 studies. That was followed by the visual analogue scale (VAS) for pruritus, used in 30 studies; the Investigator’s Global Assessment (IGA) tool, used in 29 studies; and the Eczema Area and Severity Index (EASI), used in 28 studies.
But despite the well-documented burden of AD, the researchers noted that only 33% of the RCTs they reviewed assessed QOL. “This is up from the 18% of RCTs on AD that reported QOL outcomes between 1985 and July 2010, perhaps signifying gradually increased attention to patient emotional well-being,” Ms. Hill and her associates wrote.
A trend described by the authors, however, as “perhaps the most disconcerting” was that 75% of identified QOL instruments were used only once. “Continued increases in the reporting of QOL outcomes will be of limited benefit for interstudy comparisons if the diversity of measures used also continues to rise,” they said.
Adding to the confusion, the researchers found frequent overlap in the naming and content of instruments used in studies, making it even more challenging to identify meaningful comparisons.
There was no funding source, and the authors had no conflicts of interest to declare.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: Disease severity and quality-of-life measures used in randomized controlled trials (RCTs) of atopic dermatitis (AD) need to be standardized so that study outcomes can be meaningfully compared and translated into clinical practice.
Major finding: The use of outcome measures used in atopic dermatitis RCTs has increased recently but still fall short in measuring quality-of-life outcomes.
Data source: A systematic review of 135 RCTs published between 2010-2015 that involved patients with AD.
Disclosures: There was no funding source, and the authors had no conflicts of interest to declare.
Atopic dermatitis prevention strategies under study
VIENNA – Diverse strategies aimed at preventing childhood atopic dermatitis (AD) now under study include installation of home water softeners, daily use of emollients starting at birth, and maternal consumption of probiotics beginning late in pregnancy, Carsten Flohr, PhD, said at a joint program of the International Eczema Council and the International Psoriasis Council held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
To date there is no effective method for preventing AD. Preventive strategies are needed sorely because the prevalence of pediatric AD worldwide is expected to increase substantially. It appears to have stabilized at roughly 20% in many affluent countries, but the global burden of the disease will climb as low-income countries – where AD is historically uncommon – become more developed and urbanized. This trend has been well documented via the International Study of Asthma and Allergies in Childhood (ISAAC), which in several phases has studied nearly 2 million children in more than 100 countries, noted Dr. Flohr of St. John’s Institute of Dermatology at King’s College London.
Dr. Flohr and coinvestigators in the Enquiring About Tolerance (EAT) study recently documented a significant association between water hardness and the risk of infant-onset AD. The investigators took advantage of the considerable variation in the amount of bedrock limestone across England, which enabled them to study the relationship between domestic water calcium carbonate concentrations and the presence of AD in 1,303 babies at 3 monthd of age drawn from the general population across the country. Filaggrin skin barrier gene mutation status was determined in all infants.
Infants whose water supply contained a calcium carbonate level above the median value were at an adjusted 46% greater risk of having visible AD at age 3 months than those whose household water calcium carbonate level was below the median. The AD risk rose by 1% for each 1 mg/L increase in calcium carbonate concentration above the median. This increased risk was confined to infants with a filaggrin skin barrier gene mutation; hard water didn’t increase early AD risk in children with the normal, wild-type version of the filaggrin gene (J Allergy Clin Immunol. 2016 Aug;138[2]:509-16).
As a result of these findings, a UK prevention trial is underway in which home water softeners are provided to families at high risk of having a baby with AD in water districts with high calcium carbonate concentrations. An earlier UK study found that installation of home water softeners didn’t reduce AD severity in children with established disease (PLoS Med. 2011 Feb 15;8[2]:e1000395), but disease prevention may be another story.
The role of the gut microbiota in development of childhood AD is an active area of investigation. Dr. Flohr said “there is a signal” that maternal intake of probiotics including lactobacilli and bifidobacteria in the third trimester and postnatally may reduce a child’s risk of developing AD by encouraging establishment of a more diverse gut microflora. He cited a meta-analysis of 14 published studies of probiotics which provided evidence of a 21% reduction in the incidence of AD in young children (Epidemiology 2012 May;23[3]:402-14). The studies have methodologic shortcomings, so multiple research groups are continuing to pursue the signal of an AD preventive effect.
Dr. Flor reported having no financial conflicts of interest regarding his presentation.
VIENNA – Diverse strategies aimed at preventing childhood atopic dermatitis (AD) now under study include installation of home water softeners, daily use of emollients starting at birth, and maternal consumption of probiotics beginning late in pregnancy, Carsten Flohr, PhD, said at a joint program of the International Eczema Council and the International Psoriasis Council held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
To date there is no effective method for preventing AD. Preventive strategies are needed sorely because the prevalence of pediatric AD worldwide is expected to increase substantially. It appears to have stabilized at roughly 20% in many affluent countries, but the global burden of the disease will climb as low-income countries – where AD is historically uncommon – become more developed and urbanized. This trend has been well documented via the International Study of Asthma and Allergies in Childhood (ISAAC), which in several phases has studied nearly 2 million children in more than 100 countries, noted Dr. Flohr of St. John’s Institute of Dermatology at King’s College London.
Dr. Flohr and coinvestigators in the Enquiring About Tolerance (EAT) study recently documented a significant association between water hardness and the risk of infant-onset AD. The investigators took advantage of the considerable variation in the amount of bedrock limestone across England, which enabled them to study the relationship between domestic water calcium carbonate concentrations and the presence of AD in 1,303 babies at 3 monthd of age drawn from the general population across the country. Filaggrin skin barrier gene mutation status was determined in all infants.
Infants whose water supply contained a calcium carbonate level above the median value were at an adjusted 46% greater risk of having visible AD at age 3 months than those whose household water calcium carbonate level was below the median. The AD risk rose by 1% for each 1 mg/L increase in calcium carbonate concentration above the median. This increased risk was confined to infants with a filaggrin skin barrier gene mutation; hard water didn’t increase early AD risk in children with the normal, wild-type version of the filaggrin gene (J Allergy Clin Immunol. 2016 Aug;138[2]:509-16).
As a result of these findings, a UK prevention trial is underway in which home water softeners are provided to families at high risk of having a baby with AD in water districts with high calcium carbonate concentrations. An earlier UK study found that installation of home water softeners didn’t reduce AD severity in children with established disease (PLoS Med. 2011 Feb 15;8[2]:e1000395), but disease prevention may be another story.
The role of the gut microbiota in development of childhood AD is an active area of investigation. Dr. Flohr said “there is a signal” that maternal intake of probiotics including lactobacilli and bifidobacteria in the third trimester and postnatally may reduce a child’s risk of developing AD by encouraging establishment of a more diverse gut microflora. He cited a meta-analysis of 14 published studies of probiotics which provided evidence of a 21% reduction in the incidence of AD in young children (Epidemiology 2012 May;23[3]:402-14). The studies have methodologic shortcomings, so multiple research groups are continuing to pursue the signal of an AD preventive effect.
Dr. Flor reported having no financial conflicts of interest regarding his presentation.
VIENNA – Diverse strategies aimed at preventing childhood atopic dermatitis (AD) now under study include installation of home water softeners, daily use of emollients starting at birth, and maternal consumption of probiotics beginning late in pregnancy, Carsten Flohr, PhD, said at a joint program of the International Eczema Council and the International Psoriasis Council held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
To date there is no effective method for preventing AD. Preventive strategies are needed sorely because the prevalence of pediatric AD worldwide is expected to increase substantially. It appears to have stabilized at roughly 20% in many affluent countries, but the global burden of the disease will climb as low-income countries – where AD is historically uncommon – become more developed and urbanized. This trend has been well documented via the International Study of Asthma and Allergies in Childhood (ISAAC), which in several phases has studied nearly 2 million children in more than 100 countries, noted Dr. Flohr of St. John’s Institute of Dermatology at King’s College London.
Dr. Flohr and coinvestigators in the Enquiring About Tolerance (EAT) study recently documented a significant association between water hardness and the risk of infant-onset AD. The investigators took advantage of the considerable variation in the amount of bedrock limestone across England, which enabled them to study the relationship between domestic water calcium carbonate concentrations and the presence of AD in 1,303 babies at 3 monthd of age drawn from the general population across the country. Filaggrin skin barrier gene mutation status was determined in all infants.
Infants whose water supply contained a calcium carbonate level above the median value were at an adjusted 46% greater risk of having visible AD at age 3 months than those whose household water calcium carbonate level was below the median. The AD risk rose by 1% for each 1 mg/L increase in calcium carbonate concentration above the median. This increased risk was confined to infants with a filaggrin skin barrier gene mutation; hard water didn’t increase early AD risk in children with the normal, wild-type version of the filaggrin gene (J Allergy Clin Immunol. 2016 Aug;138[2]:509-16).
As a result of these findings, a UK prevention trial is underway in which home water softeners are provided to families at high risk of having a baby with AD in water districts with high calcium carbonate concentrations. An earlier UK study found that installation of home water softeners didn’t reduce AD severity in children with established disease (PLoS Med. 2011 Feb 15;8[2]:e1000395), but disease prevention may be another story.
The role of the gut microbiota in development of childhood AD is an active area of investigation. Dr. Flohr said “there is a signal” that maternal intake of probiotics including lactobacilli and bifidobacteria in the third trimester and postnatally may reduce a child’s risk of developing AD by encouraging establishment of a more diverse gut microflora. He cited a meta-analysis of 14 published studies of probiotics which provided evidence of a 21% reduction in the incidence of AD in young children (Epidemiology 2012 May;23[3]:402-14). The studies have methodologic shortcomings, so multiple research groups are continuing to pursue the signal of an AD preventive effect.
Dr. Flor reported having no financial conflicts of interest regarding his presentation.
EXPERT ANALYSIS FROM THE EADV CONGRESS
Lebrikizumab opens new door in atopic dermatitis therapy
VIENNA – The success of the interleukin-13 blocker lebrikizumab in the TREBLE trial provides a new avenue in the treatment of moderate-to-severe atopic dermatitis.
“This is another promising molecule in atopic dermatitis,” Eric L. Simpson, MD, declared in presenting the phase II TREBLE data at the annual congress of the European Academy of Dermatology and Venereology.
Interleukin-13 is known to be an especially potent promoter of type 1, IgE-mediated inflammation. For this reason, lebrikizumab is also under investigation in the treatment of severe asthma, where large phase III trials have been completed, as well as in idiopathic pulmonary fibrosis.
In mouse models of AD, topical anti-IL-13 therapy markedly reduces skin inflammation. This observation helped provide the rationale for investigating lebrikizumab as a novel therapy for AD.
TREBLE was a double-blind, dose-ranging study involving 209 adults with moderate-to-severe AD despite intensive topical corticosteroid therapy. Indeed, enrollment was restricted to patients who still had moderate or severe disease after 2 weeks of triamcinolone 0.1% cream BID. Continuation of that twice-daily topical steroid regimen was mandatory for the full 12-week study period that followed. Patients were randomized 1:1:1:1 to triamcinolone 0.1% BID plus either a single 125-mg subcutaneous dose of lebrikizumab at week 0, a single 250-mg dose of the biologic, 125 mg every 4 weeks, or placebo injections.
At baseline, after their 2 weeks of topical steroid monotherapy, patients had a median Eczema Area and Severity Index (EASI) score of 22 and a median SCORing Atopic Dermatitis (SCORAD) of about 56. They averaged 36 years of age. Slightly over 40% of their body surface area was affected.
The primary endpoint in TREBLE was the percentage of patients who achieved at least a 50% reduction from baseline on the EASI, or EASI 50. A dose-response effect was apparent: the EASI 50 rate was 62.3% in patients on placebo plus daily topical steroids, 69.2% with a single 125-mg dose of lebrikizumab, 69.8% with a single 250-mg dose, and 82.4% with 125 mg of lebrikizumab at weeks 0, 4, 8, and 12. Only the group with monthly dosing of the biologic plus daily triamcinolone 0.1% BID had an EASI 50 response rate significantly better than the controls on placebo plus topical steroid therapy.
A key secondary endpoint was the SCORAD 50 response. The rate was 26.4% in the control group, 34.6% in patients who received one 125-mg dose of lebrikizumab, 47.2% in those who got a single 250-mg dose, and 51% with monthly dosing. The SCORAD 50 rate was significantly greater than placebo in the latter two lebrikizumab arms.
The EASI 75 rate was significantly greater than placebo plus triamcinolone only in the group on monthly lebrikizumab plus topical steroids.
The EASI 50 and 75 response rates in the group on 125 mg of lebrikizumab every 4 weeks was still climbing with no plateau evident when the trial ended at 12 weeks. This suggests greater benefit might be achieved with longer treatment duration and/or a higher dosage, Dr. Simpson observed.
Turning to safety issues, he pointed out that the total number of adverse events and serious adverse events were similar across all four treatment arms. Herpes infections occurred in 2%-6% of patients who received lebrikizumab but in no controls. There was also a nonsignificant trend for more cases of conjunctivitis in the groups that received the biologic.
Audience member Andrew Blauvelt, MD, rose to take issue with the study design.
“This study is almost an advertisement for not using topical steroids in a biologic trial for atopic dermatitis. I think it just confuses everything. It especially doesn’t make sense when one of the inclusion criteria is inadequate control with topical steroids. Shouldn’t we be doing monotherapy trials in the new era of biologics for atopic dermatitis?” commented Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Dr. Simpson replied that when TREBLE was designed, the thinking was that in real-world clinical practice, physicians often prescribe a biologic for AD on top of topical therapy. The goal was to conduct a trial that mimics that situation. That being said, he agreed with Dr. Blauvelt’s critique.
“I think we’re realizing that topical steroids have a very strong effect, especially if you use them all the way through a trial or ad lib,” he said. “Topical steroids bring too many confounders. If you can remove potent topical steroid therapy I think you’re going to have a clearer result.”
“We’re not where psoriasis is,” Dr. Simpson added. “We don’t have any standardized methodologies in atopic dermatitis clinical trials yet, but I think we’re getting there.”
The TREBLE trial was sponsored by Genentech/Roche. Dr. Simpson reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
VIENNA – The success of the interleukin-13 blocker lebrikizumab in the TREBLE trial provides a new avenue in the treatment of moderate-to-severe atopic dermatitis.
“This is another promising molecule in atopic dermatitis,” Eric L. Simpson, MD, declared in presenting the phase II TREBLE data at the annual congress of the European Academy of Dermatology and Venereology.
Interleukin-13 is known to be an especially potent promoter of type 1, IgE-mediated inflammation. For this reason, lebrikizumab is also under investigation in the treatment of severe asthma, where large phase III trials have been completed, as well as in idiopathic pulmonary fibrosis.
In mouse models of AD, topical anti-IL-13 therapy markedly reduces skin inflammation. This observation helped provide the rationale for investigating lebrikizumab as a novel therapy for AD.
TREBLE was a double-blind, dose-ranging study involving 209 adults with moderate-to-severe AD despite intensive topical corticosteroid therapy. Indeed, enrollment was restricted to patients who still had moderate or severe disease after 2 weeks of triamcinolone 0.1% cream BID. Continuation of that twice-daily topical steroid regimen was mandatory for the full 12-week study period that followed. Patients were randomized 1:1:1:1 to triamcinolone 0.1% BID plus either a single 125-mg subcutaneous dose of lebrikizumab at week 0, a single 250-mg dose of the biologic, 125 mg every 4 weeks, or placebo injections.
At baseline, after their 2 weeks of topical steroid monotherapy, patients had a median Eczema Area and Severity Index (EASI) score of 22 and a median SCORing Atopic Dermatitis (SCORAD) of about 56. They averaged 36 years of age. Slightly over 40% of their body surface area was affected.
The primary endpoint in TREBLE was the percentage of patients who achieved at least a 50% reduction from baseline on the EASI, or EASI 50. A dose-response effect was apparent: the EASI 50 rate was 62.3% in patients on placebo plus daily topical steroids, 69.2% with a single 125-mg dose of lebrikizumab, 69.8% with a single 250-mg dose, and 82.4% with 125 mg of lebrikizumab at weeks 0, 4, 8, and 12. Only the group with monthly dosing of the biologic plus daily triamcinolone 0.1% BID had an EASI 50 response rate significantly better than the controls on placebo plus topical steroid therapy.
A key secondary endpoint was the SCORAD 50 response. The rate was 26.4% in the control group, 34.6% in patients who received one 125-mg dose of lebrikizumab, 47.2% in those who got a single 250-mg dose, and 51% with monthly dosing. The SCORAD 50 rate was significantly greater than placebo in the latter two lebrikizumab arms.
The EASI 75 rate was significantly greater than placebo plus triamcinolone only in the group on monthly lebrikizumab plus topical steroids.
The EASI 50 and 75 response rates in the group on 125 mg of lebrikizumab every 4 weeks was still climbing with no plateau evident when the trial ended at 12 weeks. This suggests greater benefit might be achieved with longer treatment duration and/or a higher dosage, Dr. Simpson observed.
Turning to safety issues, he pointed out that the total number of adverse events and serious adverse events were similar across all four treatment arms. Herpes infections occurred in 2%-6% of patients who received lebrikizumab but in no controls. There was also a nonsignificant trend for more cases of conjunctivitis in the groups that received the biologic.
Audience member Andrew Blauvelt, MD, rose to take issue with the study design.
“This study is almost an advertisement for not using topical steroids in a biologic trial for atopic dermatitis. I think it just confuses everything. It especially doesn’t make sense when one of the inclusion criteria is inadequate control with topical steroids. Shouldn’t we be doing monotherapy trials in the new era of biologics for atopic dermatitis?” commented Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Dr. Simpson replied that when TREBLE was designed, the thinking was that in real-world clinical practice, physicians often prescribe a biologic for AD on top of topical therapy. The goal was to conduct a trial that mimics that situation. That being said, he agreed with Dr. Blauvelt’s critique.
“I think we’re realizing that topical steroids have a very strong effect, especially if you use them all the way through a trial or ad lib,” he said. “Topical steroids bring too many confounders. If you can remove potent topical steroid therapy I think you’re going to have a clearer result.”
“We’re not where psoriasis is,” Dr. Simpson added. “We don’t have any standardized methodologies in atopic dermatitis clinical trials yet, but I think we’re getting there.”
The TREBLE trial was sponsored by Genentech/Roche. Dr. Simpson reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
VIENNA – The success of the interleukin-13 blocker lebrikizumab in the TREBLE trial provides a new avenue in the treatment of moderate-to-severe atopic dermatitis.
“This is another promising molecule in atopic dermatitis,” Eric L. Simpson, MD, declared in presenting the phase II TREBLE data at the annual congress of the European Academy of Dermatology and Venereology.
Interleukin-13 is known to be an especially potent promoter of type 1, IgE-mediated inflammation. For this reason, lebrikizumab is also under investigation in the treatment of severe asthma, where large phase III trials have been completed, as well as in idiopathic pulmonary fibrosis.
In mouse models of AD, topical anti-IL-13 therapy markedly reduces skin inflammation. This observation helped provide the rationale for investigating lebrikizumab as a novel therapy for AD.
TREBLE was a double-blind, dose-ranging study involving 209 adults with moderate-to-severe AD despite intensive topical corticosteroid therapy. Indeed, enrollment was restricted to patients who still had moderate or severe disease after 2 weeks of triamcinolone 0.1% cream BID. Continuation of that twice-daily topical steroid regimen was mandatory for the full 12-week study period that followed. Patients were randomized 1:1:1:1 to triamcinolone 0.1% BID plus either a single 125-mg subcutaneous dose of lebrikizumab at week 0, a single 250-mg dose of the biologic, 125 mg every 4 weeks, or placebo injections.
At baseline, after their 2 weeks of topical steroid monotherapy, patients had a median Eczema Area and Severity Index (EASI) score of 22 and a median SCORing Atopic Dermatitis (SCORAD) of about 56. They averaged 36 years of age. Slightly over 40% of their body surface area was affected.
The primary endpoint in TREBLE was the percentage of patients who achieved at least a 50% reduction from baseline on the EASI, or EASI 50. A dose-response effect was apparent: the EASI 50 rate was 62.3% in patients on placebo plus daily topical steroids, 69.2% with a single 125-mg dose of lebrikizumab, 69.8% with a single 250-mg dose, and 82.4% with 125 mg of lebrikizumab at weeks 0, 4, 8, and 12. Only the group with monthly dosing of the biologic plus daily triamcinolone 0.1% BID had an EASI 50 response rate significantly better than the controls on placebo plus topical steroid therapy.
A key secondary endpoint was the SCORAD 50 response. The rate was 26.4% in the control group, 34.6% in patients who received one 125-mg dose of lebrikizumab, 47.2% in those who got a single 250-mg dose, and 51% with monthly dosing. The SCORAD 50 rate was significantly greater than placebo in the latter two lebrikizumab arms.
The EASI 75 rate was significantly greater than placebo plus triamcinolone only in the group on monthly lebrikizumab plus topical steroids.
The EASI 50 and 75 response rates in the group on 125 mg of lebrikizumab every 4 weeks was still climbing with no plateau evident when the trial ended at 12 weeks. This suggests greater benefit might be achieved with longer treatment duration and/or a higher dosage, Dr. Simpson observed.
Turning to safety issues, he pointed out that the total number of adverse events and serious adverse events were similar across all four treatment arms. Herpes infections occurred in 2%-6% of patients who received lebrikizumab but in no controls. There was also a nonsignificant trend for more cases of conjunctivitis in the groups that received the biologic.
Audience member Andrew Blauvelt, MD, rose to take issue with the study design.
“This study is almost an advertisement for not using topical steroids in a biologic trial for atopic dermatitis. I think it just confuses everything. It especially doesn’t make sense when one of the inclusion criteria is inadequate control with topical steroids. Shouldn’t we be doing monotherapy trials in the new era of biologics for atopic dermatitis?” commented Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Dr. Simpson replied that when TREBLE was designed, the thinking was that in real-world clinical practice, physicians often prescribe a biologic for AD on top of topical therapy. The goal was to conduct a trial that mimics that situation. That being said, he agreed with Dr. Blauvelt’s critique.
“I think we’re realizing that topical steroids have a very strong effect, especially if you use them all the way through a trial or ad lib,” he said. “Topical steroids bring too many confounders. If you can remove potent topical steroid therapy I think you’re going to have a clearer result.”
“We’re not where psoriasis is,” Dr. Simpson added. “We don’t have any standardized methodologies in atopic dermatitis clinical trials yet, but I think we’re getting there.”
The TREBLE trial was sponsored by Genentech/Roche. Dr. Simpson reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point:
Major finding: Of atopic dermatitis patients randomized to 125 mg of lebrikizumab every 4 weeks on top of twice-daily potent topical steroids, 82% experienced at least a 50% reduction in Eczema Area and Severity Index scores at 12 weeks, compared with 62% on the same topical steroid regimen plus placebo injections.
Data source: The TREBLE trial was a phase II, double-blind, placebo-controlled, 12-week study including 209 adults with moderate-to-severe atopic dermatitis inadequately controlled by intensive topical steroid therapy.
Disclosures: The study was sponsored by Genentech/Roche. The presenter reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.