User login
‘Cutting for Stone’ author closes out this year’s AAD plenary session
The plenary session at the 2018 American Academy of Dermatology annual meeting in San Diego includes the Clarence S. Livingood, MD Memorial Award and Lectureship, by Mary-Margaret Chren, MD, professor in residence, department of dermatology, University of California, San Francisco, on “The State of (Measuring) the Art of Dermatology.”
Dr. Chren will be followed by the AAD president’s address, given by outgoing president Henry W. Lim, MD, chairman of the department of dermatology and Clarence S. Livingood Chair in Dermatology at Henry Ford Health System in Detroit.
Following Dr. Lim, Jan T. Vilcek, MD, PhD, will give the Eugene J. Van Scott Award for Innovative Therapy of the Skin and Phillip Frost Leadership Lecture on “How a TNF Inhibitor Advanced from Modest Beginnings to Unforeseen Therapeutic Successes.” Dr. Vilcek is research professor and professor emeritus of microbiology in the department of microbiology at New York University.
Suzanne M. Olbricht, MD, president-elect of the AAD and chief of dermatology at Beth Israel Deaconess Medical Center, Boston, will follow.
Jennifer A. Doudna, PhD, professor of chemistry, and professor of biochemistry and molecular biology at the University of California, Berkeley, will give the Lila and Murray Gruber Memorial Cancer Research Award and Lectureship. Her talk is titled, “CRISPR Systems: Nature’s Toolkit for Genome Editing.”
Finally, this year’s guest speaker is Abraham Verghese, MD, whose talk is titled: “The Pathology Within: Burnout, Wellness, and the Search for Meaning in a Professional Life.”
Dr. Verghese, professor and Linda R. Meier and Joan F. Lane Provostial Professor, and vice chair for the Theory and Practice of Medicine, Stanford (Calif.) University, is the author of several books including “Cutting for Stone,” his first novel.
The plenary session is scheduled for Sunday, Feb. 18, from 8 a.m. to 11:30 a.m.
The plenary session at the 2018 American Academy of Dermatology annual meeting in San Diego includes the Clarence S. Livingood, MD Memorial Award and Lectureship, by Mary-Margaret Chren, MD, professor in residence, department of dermatology, University of California, San Francisco, on “The State of (Measuring) the Art of Dermatology.”
Dr. Chren will be followed by the AAD president’s address, given by outgoing president Henry W. Lim, MD, chairman of the department of dermatology and Clarence S. Livingood Chair in Dermatology at Henry Ford Health System in Detroit.
Following Dr. Lim, Jan T. Vilcek, MD, PhD, will give the Eugene J. Van Scott Award for Innovative Therapy of the Skin and Phillip Frost Leadership Lecture on “How a TNF Inhibitor Advanced from Modest Beginnings to Unforeseen Therapeutic Successes.” Dr. Vilcek is research professor and professor emeritus of microbiology in the department of microbiology at New York University.
Suzanne M. Olbricht, MD, president-elect of the AAD and chief of dermatology at Beth Israel Deaconess Medical Center, Boston, will follow.
Jennifer A. Doudna, PhD, professor of chemistry, and professor of biochemistry and molecular biology at the University of California, Berkeley, will give the Lila and Murray Gruber Memorial Cancer Research Award and Lectureship. Her talk is titled, “CRISPR Systems: Nature’s Toolkit for Genome Editing.”
Finally, this year’s guest speaker is Abraham Verghese, MD, whose talk is titled: “The Pathology Within: Burnout, Wellness, and the Search for Meaning in a Professional Life.”
Dr. Verghese, professor and Linda R. Meier and Joan F. Lane Provostial Professor, and vice chair for the Theory and Practice of Medicine, Stanford (Calif.) University, is the author of several books including “Cutting for Stone,” his first novel.
The plenary session is scheduled for Sunday, Feb. 18, from 8 a.m. to 11:30 a.m.
The plenary session at the 2018 American Academy of Dermatology annual meeting in San Diego includes the Clarence S. Livingood, MD Memorial Award and Lectureship, by Mary-Margaret Chren, MD, professor in residence, department of dermatology, University of California, San Francisco, on “The State of (Measuring) the Art of Dermatology.”
Dr. Chren will be followed by the AAD president’s address, given by outgoing president Henry W. Lim, MD, chairman of the department of dermatology and Clarence S. Livingood Chair in Dermatology at Henry Ford Health System in Detroit.
Following Dr. Lim, Jan T. Vilcek, MD, PhD, will give the Eugene J. Van Scott Award for Innovative Therapy of the Skin and Phillip Frost Leadership Lecture on “How a TNF Inhibitor Advanced from Modest Beginnings to Unforeseen Therapeutic Successes.” Dr. Vilcek is research professor and professor emeritus of microbiology in the department of microbiology at New York University.
Suzanne M. Olbricht, MD, president-elect of the AAD and chief of dermatology at Beth Israel Deaconess Medical Center, Boston, will follow.
Jennifer A. Doudna, PhD, professor of chemistry, and professor of biochemistry and molecular biology at the University of California, Berkeley, will give the Lila and Murray Gruber Memorial Cancer Research Award and Lectureship. Her talk is titled, “CRISPR Systems: Nature’s Toolkit for Genome Editing.”
Finally, this year’s guest speaker is Abraham Verghese, MD, whose talk is titled: “The Pathology Within: Burnout, Wellness, and the Search for Meaning in a Professional Life.”
Dr. Verghese, professor and Linda R. Meier and Joan F. Lane Provostial Professor, and vice chair for the Theory and Practice of Medicine, Stanford (Calif.) University, is the author of several books including “Cutting for Stone,” his first novel.
The plenary session is scheduled for Sunday, Feb. 18, from 8 a.m. to 11:30 a.m.
VIDEO: Off-label dupilumab finding a home in pediatric AD
KAUAI, HAWAII – Pediatric dermatologists aren’t waiting for Food and Drug Administration approval to try dupilumab (Dupixent) for their patients with severe atopic dermatitis.
It’s not approved in children, but the possibility of good control without the side effects of cyclosporine and other alternatives is too much to resist. A phase 2, company-sponsored study reported Eczema Area and Severity Index score improvements of up to 76% in pediatric patients treated with dupilumab, an interleukin-4 and IL-13 signaling blocker approved in 2017 for moderate to severe AD in adults.
Large pediatric trials are pending, but with results like that, “many of us just feel if it was our own kid, and we could get dupilumab, we would like to do that first,” said Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego.
It’s not just dupilumab that’s causing excitement. With almost 20 biologics in the pipeline, eczema seems poised to undergo a revolution in treatment much like psoriasis has in recent years.
Dr. Eichenfield explained (Eucrisa), a topical nonsteroidal phosphodiesterase-4 inhibitor approved for mild to moderate AD for children and adults ages two and older in December 2016, which doesn’t seem to have the duration limits of steroids, he said in an interview at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Treatment of pediatric AD is “going to be a very different picture over the next few years,” he said.
Dr. Eichenfield is a consultant or investigator for many companies, including Regeneron/Sanofi, Genentech, Novartis, Pfizer, Lilly, and Allergan.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Pediatric dermatologists aren’t waiting for Food and Drug Administration approval to try dupilumab (Dupixent) for their patients with severe atopic dermatitis.
It’s not approved in children, but the possibility of good control without the side effects of cyclosporine and other alternatives is too much to resist. A phase 2, company-sponsored study reported Eczema Area and Severity Index score improvements of up to 76% in pediatric patients treated with dupilumab, an interleukin-4 and IL-13 signaling blocker approved in 2017 for moderate to severe AD in adults.
Large pediatric trials are pending, but with results like that, “many of us just feel if it was our own kid, and we could get dupilumab, we would like to do that first,” said Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego.
It’s not just dupilumab that’s causing excitement. With almost 20 biologics in the pipeline, eczema seems poised to undergo a revolution in treatment much like psoriasis has in recent years.
Dr. Eichenfield explained (Eucrisa), a topical nonsteroidal phosphodiesterase-4 inhibitor approved for mild to moderate AD for children and adults ages two and older in December 2016, which doesn’t seem to have the duration limits of steroids, he said in an interview at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Treatment of pediatric AD is “going to be a very different picture over the next few years,” he said.
Dr. Eichenfield is a consultant or investigator for many companies, including Regeneron/Sanofi, Genentech, Novartis, Pfizer, Lilly, and Allergan.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Pediatric dermatologists aren’t waiting for Food and Drug Administration approval to try dupilumab (Dupixent) for their patients with severe atopic dermatitis.
It’s not approved in children, but the possibility of good control without the side effects of cyclosporine and other alternatives is too much to resist. A phase 2, company-sponsored study reported Eczema Area and Severity Index score improvements of up to 76% in pediatric patients treated with dupilumab, an interleukin-4 and IL-13 signaling blocker approved in 2017 for moderate to severe AD in adults.
Large pediatric trials are pending, but with results like that, “many of us just feel if it was our own kid, and we could get dupilumab, we would like to do that first,” said Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego.
It’s not just dupilumab that’s causing excitement. With almost 20 biologics in the pipeline, eczema seems poised to undergo a revolution in treatment much like psoriasis has in recent years.
Dr. Eichenfield explained (Eucrisa), a topical nonsteroidal phosphodiesterase-4 inhibitor approved for mild to moderate AD for children and adults ages two and older in December 2016, which doesn’t seem to have the duration limits of steroids, he said in an interview at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Treatment of pediatric AD is “going to be a very different picture over the next few years,” he said.
Dr. Eichenfield is a consultant or investigator for many companies, including Regeneron/Sanofi, Genentech, Novartis, Pfizer, Lilly, and Allergan.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
Mobile Medical Apps for Patient Education: A Graded Review of Available Dermatology Apps
According to industry estimates, roughly 64% of US adults were smartphone users in 2015.1 Smartphones enable users to utilize mobile applications (apps) that can perform a variety of functions in many categories, including business, music, photography, entertainment, education, social networking, travel, and lifestyle. The widespread adoption and use of mobile apps has implications for medical practice. Mobile apps have the capability to serve as information sources for patients, educational tools for students, and diagnostic aids for physicians.2 Consequently, a number of medical and health care–oriented apps have already been developed3 and are increasingly utilized by patients and providers.4
Given its visual nature, dermatology is particularly amenable to the integration of mobile medical apps. A study by Brewer et al5 identified more than 229 dermatology-related apps in categories ranging from general dermatology reference, self-surveillance and diagnosis, disease guides, educational aids, sunscreen and UV recommendations, and teledermatology. Patients served as the target audience and principal consumers of more than half of these dermatology apps.5
Mobile medical and health care apps demonstrate great potential for serving as valuable information sources for patients with dermatologic conditions; however, the content, functions, accuracy, and educational value of dermatology mobile apps are not well characterized, making it difficult for patients and health care providers to select and recommend appropriate apps.6 In this study, we created a rubric to objectively grade 44 publicly available mobile dermatology apps with the primary focus of patient education.
Methods
We conducted a search of dermatology-related educational mobile apps that were publicly available via the App Store (Apple Inc) from January 2016 to November 2016. (The pricing, availability, and other features of these apps may have changed since the study period.) The following search terms were used: dermatology, dermoscopy, melanoma, skin cancer, psoriasis, rosacea, acne, eczema, dermal fillers, and Mohs surgery. We excluded apps that were not in English; had a solely commercial focus; were mobile textbooks or scientific journals; were used to provide teledermatology services with no educational purpose; were solely focused on homeopathic, alternative, and/or complementary medicine; or were intended primarily as a reference for students or health care professionals. Our search yielded 44 apps with patient education as a primary objective. The apps were divided into 6 categories based on their focus: general dermatology, cosmetic dermatology, acne, eczema, psoriasis, and skin cancer.
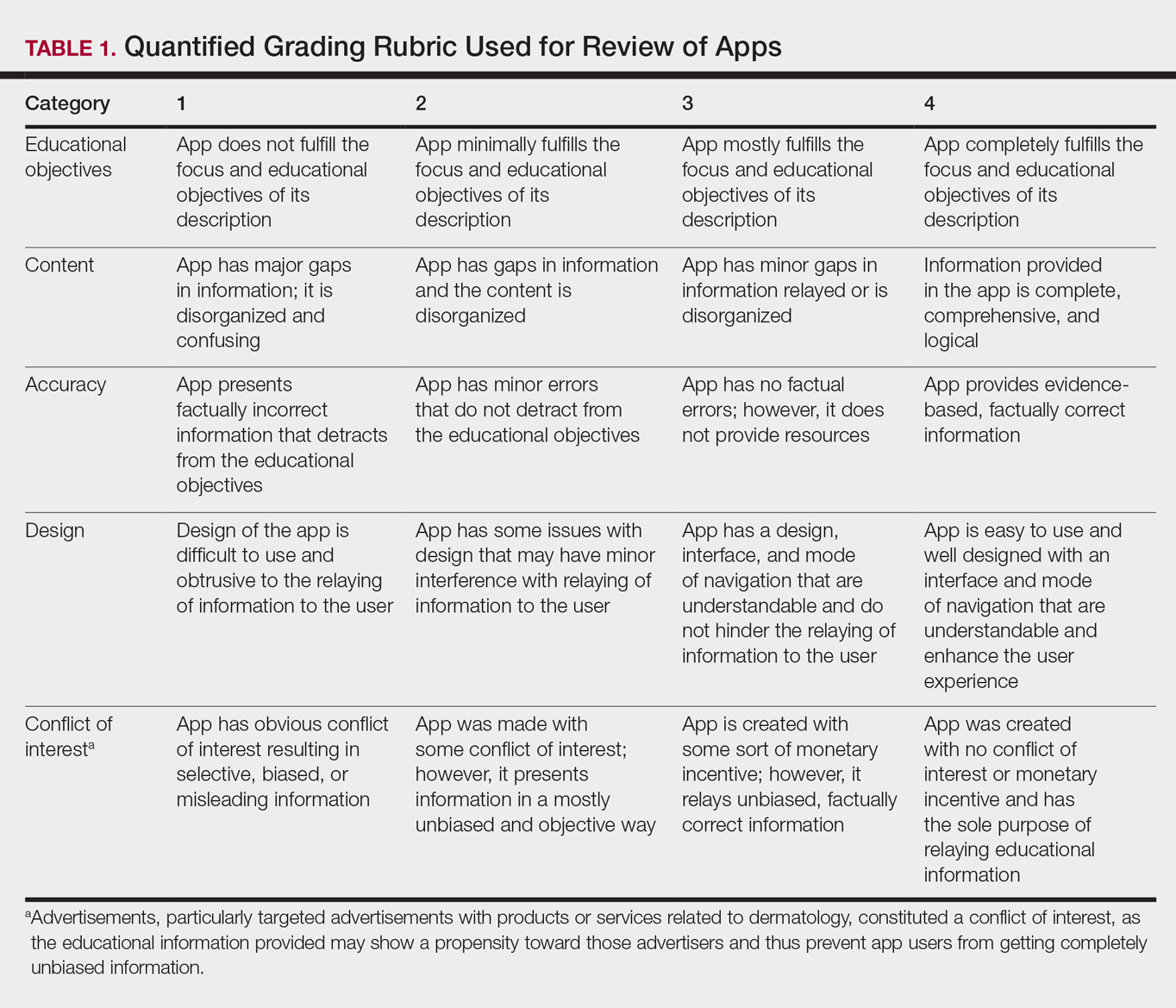
Each app was reviewed using a quantified grading rubric developed by the researchers. In a prior evaluation, Handel7 reviewed 35 health and wellness mobile apps utilizing the categories of ease of use, reliability, quality, scope of information, and aesthetics.4 These criteria were modified and adapted for the purposes of this study, and a 4-point scale was applied to each criterion. The final criteria were (1) educational objectives, (2) content, (3) accuracy, (4) design, and (5) conflict of interest. The quantified grading rubric is described in Table 1.
Results
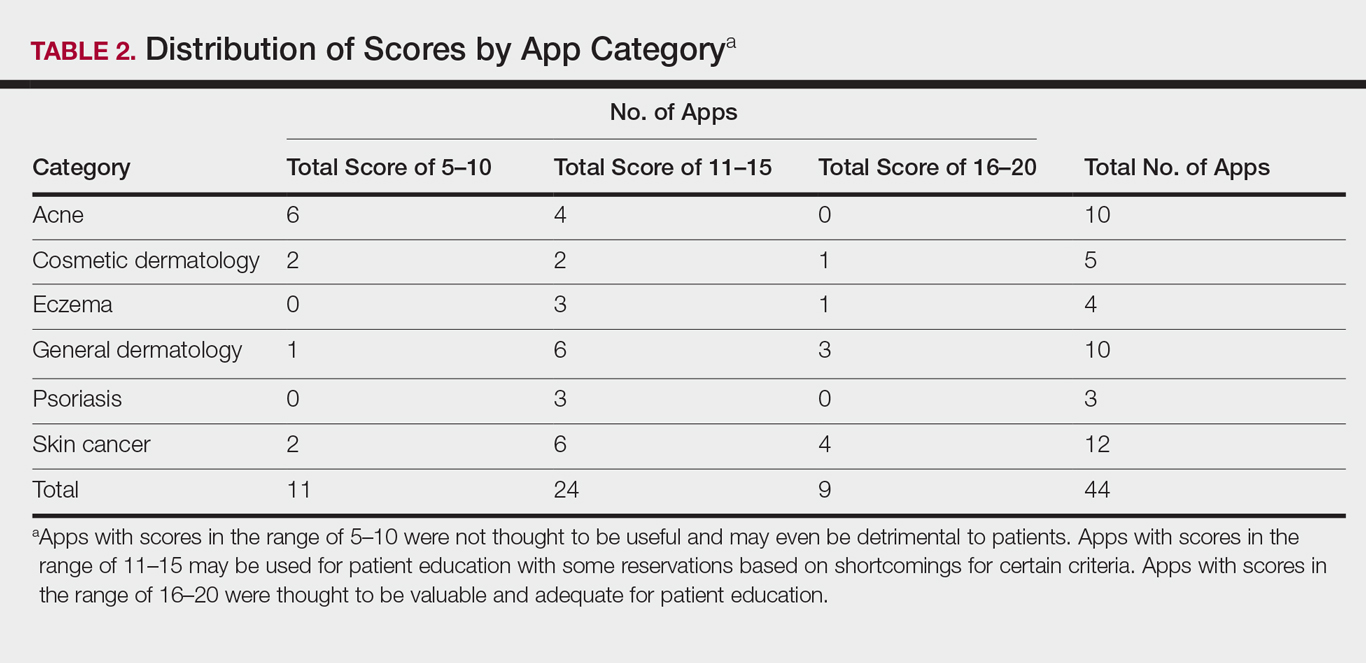
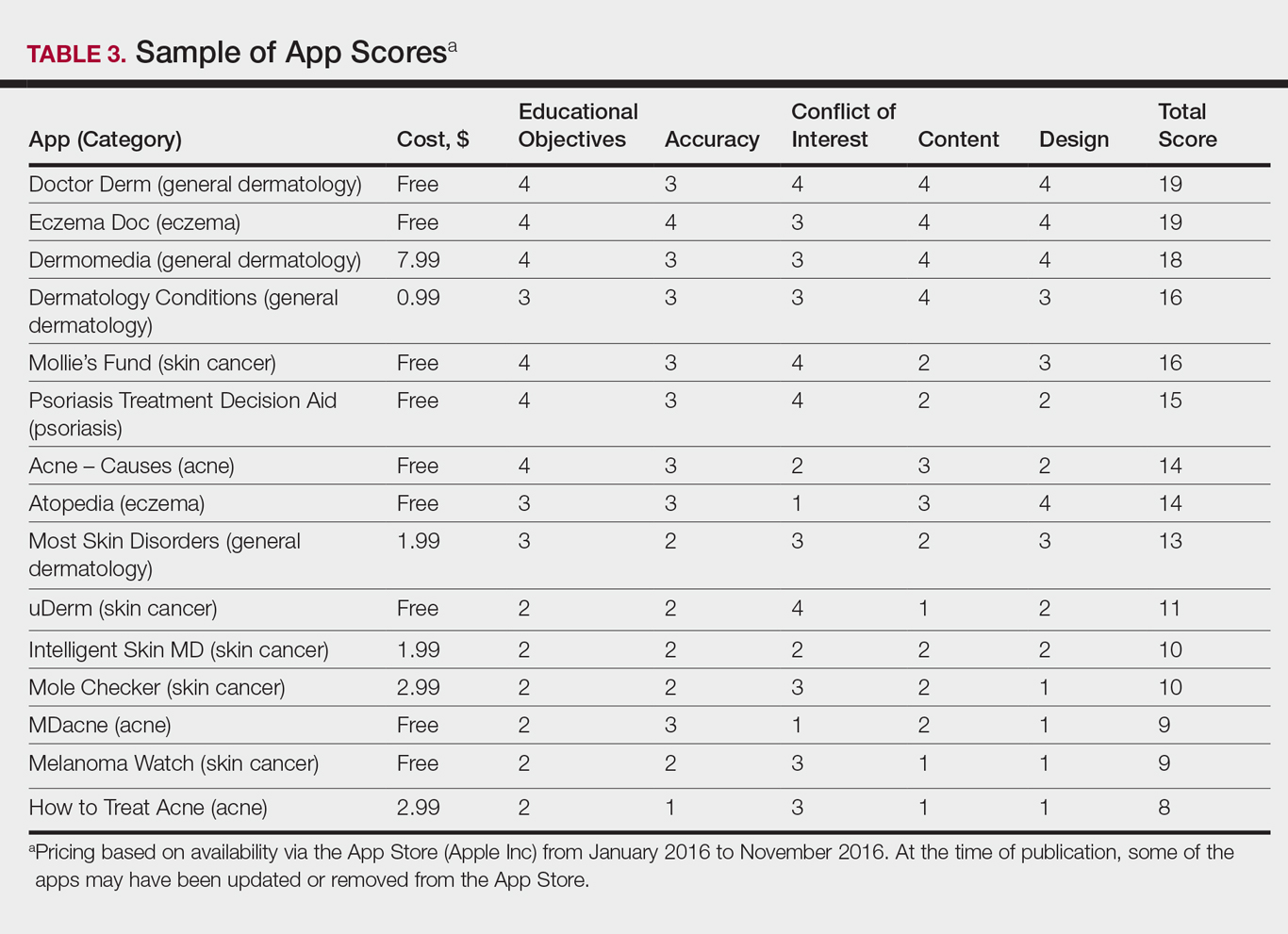
The possible range of scores based on the grading rubric was 5 to 20. The actual range of scores was 8 to 19 (Table 2). The 44 reviewed apps were categorized by topic as acne, cosmetic dermatology, eczema, general dermatology, psoriasis, or skin cancer. A sample of 15 apps selected to represent the distribution of scores and their grading on the rubric are presented in Table 3.
Comment
The number of dermatology-related apps available to mobile users continues to grow at an increasing rate.8 The apps vary in many aspects, including their purpose, scope, intended audience, and goals of the app publisher. In turn, more individuals are turning to mobile apps for medical information,4 especially in dermatology, thus it is necessary to create a systematic way to evaluate the quality and utility of each app to assist users in making informed decisions about which apps will best meet their needs in the midst of a wide array of choices.
For the purpose of this study, an objective rubric was created that can be used to evaluate the quality of medical apps for patient education in dermatology. An app’s adequacy and usefulness for patient education was thought to depend on 3 possible score ranges into which the app could fall based on the grading rubric. An app with a total score in the range of 5 to 10 was not thought to be useful and may even be detrimental to patients. An app with a total score in the range of 11 to 15 may be used for patient education with some reservations based on shortcomings for certain criteria. An app with a score in the range of 16 to 20 was thought to be valuable and adequate for patient education. For example, the How to Treat Acne app received a total score of 8 and therefore would not be recommended to patients based on the grading rubric used in this study. This particular app provided sparse and sometimes inaccurate information, had a confusing user interface, and contained many obstructive advertisements. In contrast, the Eczema Doc app received a total score of 19, which indicates a quality app deemed to be useful for patient information based on the established rubric. This app met all the objectives that it advertised, contained accurate information with verified citation of sources, and was very easy for users to navigate.
Of the 44 graded apps, only 9 (20.5%) received scores in the highest range of 16 to 20, which indicates a need for improvements in mobile dermatology apps intended for patient education. Adopting the grading rubric developed in this study as a standard in the creation of medical apps could have beneficial implications in disseminating accurate, safe, unbiased, and easy-to-understand information to patients.
- Smith A. U.S. smartphone use in 2015. Pew Research Center website. http://www.pewinternet.org/2015/04/01/us-smartphone-use-in-2015. Published April 1, 2015. Accessed August 29, 2017.
- Nilsen W, Kumar S, Shar A, et al. Advancing the science of mHealth. J Health Commun. 2012;17(suppl 1):5-10.
- West DM. How mobile devices are transforming healthcare issues in technology innovation. Issues Technol Innov. 2012;18:1-14.
- Boudreaux ED, Waring ME, Hayes RB, et al. Evaluating and selecting mobile health apps: strategies for healthcare providers and healthcare organizations. Transl Behav Med. 2014;4:363-371.
- Brewer AC, Endly DC, Henley J, et al. Mobile applications in dermatology. JAMA Dermatol. 2013;149:1300-1304.
- Cummings E, Borycki E, Roehrer E. Issues and considerations for healthcare consumers using mobile applications. Stud Health Technol Inform. 2013;183:227-231.
- Handel MJ. mHealth (mobile health)-using apps for health and wellness. Explore. 2011;7:256-261.
- Boulos MN, Brewer AC, Karimkhani C, et al. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J Public Health Inform. 2014;5:229.
According to industry estimates, roughly 64% of US adults were smartphone users in 2015.1 Smartphones enable users to utilize mobile applications (apps) that can perform a variety of functions in many categories, including business, music, photography, entertainment, education, social networking, travel, and lifestyle. The widespread adoption and use of mobile apps has implications for medical practice. Mobile apps have the capability to serve as information sources for patients, educational tools for students, and diagnostic aids for physicians.2 Consequently, a number of medical and health care–oriented apps have already been developed3 and are increasingly utilized by patients and providers.4
Given its visual nature, dermatology is particularly amenable to the integration of mobile medical apps. A study by Brewer et al5 identified more than 229 dermatology-related apps in categories ranging from general dermatology reference, self-surveillance and diagnosis, disease guides, educational aids, sunscreen and UV recommendations, and teledermatology. Patients served as the target audience and principal consumers of more than half of these dermatology apps.5
Mobile medical and health care apps demonstrate great potential for serving as valuable information sources for patients with dermatologic conditions; however, the content, functions, accuracy, and educational value of dermatology mobile apps are not well characterized, making it difficult for patients and health care providers to select and recommend appropriate apps.6 In this study, we created a rubric to objectively grade 44 publicly available mobile dermatology apps with the primary focus of patient education.
Methods
We conducted a search of dermatology-related educational mobile apps that were publicly available via the App Store (Apple Inc) from January 2016 to November 2016. (The pricing, availability, and other features of these apps may have changed since the study period.) The following search terms were used: dermatology, dermoscopy, melanoma, skin cancer, psoriasis, rosacea, acne, eczema, dermal fillers, and Mohs surgery. We excluded apps that were not in English; had a solely commercial focus; were mobile textbooks or scientific journals; were used to provide teledermatology services with no educational purpose; were solely focused on homeopathic, alternative, and/or complementary medicine; or were intended primarily as a reference for students or health care professionals. Our search yielded 44 apps with patient education as a primary objective. The apps were divided into 6 categories based on their focus: general dermatology, cosmetic dermatology, acne, eczema, psoriasis, and skin cancer.
Each app was reviewed using a quantified grading rubric developed by the researchers. In a prior evaluation, Handel7 reviewed 35 health and wellness mobile apps utilizing the categories of ease of use, reliability, quality, scope of information, and aesthetics.4 These criteria were modified and adapted for the purposes of this study, and a 4-point scale was applied to each criterion. The final criteria were (1) educational objectives, (2) content, (3) accuracy, (4) design, and (5) conflict of interest. The quantified grading rubric is described in Table 1.

Results
The possible range of scores based on the grading rubric was 5 to 20. The actual range of scores was 8 to 19 (Table 2). The 44 reviewed apps were categorized by topic as acne, cosmetic dermatology, eczema, general dermatology, psoriasis, or skin cancer. A sample of 15 apps selected to represent the distribution of scores and their grading on the rubric are presented in Table 3.


Comment
The number of dermatology-related apps available to mobile users continues to grow at an increasing rate.8 The apps vary in many aspects, including their purpose, scope, intended audience, and goals of the app publisher. In turn, more individuals are turning to mobile apps for medical information,4 especially in dermatology, thus it is necessary to create a systematic way to evaluate the quality and utility of each app to assist users in making informed decisions about which apps will best meet their needs in the midst of a wide array of choices.
For the purpose of this study, an objective rubric was created that can be used to evaluate the quality of medical apps for patient education in dermatology. An app’s adequacy and usefulness for patient education was thought to depend on 3 possible score ranges into which the app could fall based on the grading rubric. An app with a total score in the range of 5 to 10 was not thought to be useful and may even be detrimental to patients. An app with a total score in the range of 11 to 15 may be used for patient education with some reservations based on shortcomings for certain criteria. An app with a score in the range of 16 to 20 was thought to be valuable and adequate for patient education. For example, the How to Treat Acne app received a total score of 8 and therefore would not be recommended to patients based on the grading rubric used in this study. This particular app provided sparse and sometimes inaccurate information, had a confusing user interface, and contained many obstructive advertisements. In contrast, the Eczema Doc app received a total score of 19, which indicates a quality app deemed to be useful for patient information based on the established rubric. This app met all the objectives that it advertised, contained accurate information with verified citation of sources, and was very easy for users to navigate.
Of the 44 graded apps, only 9 (20.5%) received scores in the highest range of 16 to 20, which indicates a need for improvements in mobile dermatology apps intended for patient education. Adopting the grading rubric developed in this study as a standard in the creation of medical apps could have beneficial implications in disseminating accurate, safe, unbiased, and easy-to-understand information to patients.
According to industry estimates, roughly 64% of US adults were smartphone users in 2015.1 Smartphones enable users to utilize mobile applications (apps) that can perform a variety of functions in many categories, including business, music, photography, entertainment, education, social networking, travel, and lifestyle. The widespread adoption and use of mobile apps has implications for medical practice. Mobile apps have the capability to serve as information sources for patients, educational tools for students, and diagnostic aids for physicians.2 Consequently, a number of medical and health care–oriented apps have already been developed3 and are increasingly utilized by patients and providers.4
Given its visual nature, dermatology is particularly amenable to the integration of mobile medical apps. A study by Brewer et al5 identified more than 229 dermatology-related apps in categories ranging from general dermatology reference, self-surveillance and diagnosis, disease guides, educational aids, sunscreen and UV recommendations, and teledermatology. Patients served as the target audience and principal consumers of more than half of these dermatology apps.5
Mobile medical and health care apps demonstrate great potential for serving as valuable information sources for patients with dermatologic conditions; however, the content, functions, accuracy, and educational value of dermatology mobile apps are not well characterized, making it difficult for patients and health care providers to select and recommend appropriate apps.6 In this study, we created a rubric to objectively grade 44 publicly available mobile dermatology apps with the primary focus of patient education.
Methods
We conducted a search of dermatology-related educational mobile apps that were publicly available via the App Store (Apple Inc) from January 2016 to November 2016. (The pricing, availability, and other features of these apps may have changed since the study period.) The following search terms were used: dermatology, dermoscopy, melanoma, skin cancer, psoriasis, rosacea, acne, eczema, dermal fillers, and Mohs surgery. We excluded apps that were not in English; had a solely commercial focus; were mobile textbooks or scientific journals; were used to provide teledermatology services with no educational purpose; were solely focused on homeopathic, alternative, and/or complementary medicine; or were intended primarily as a reference for students or health care professionals. Our search yielded 44 apps with patient education as a primary objective. The apps were divided into 6 categories based on their focus: general dermatology, cosmetic dermatology, acne, eczema, psoriasis, and skin cancer.
Each app was reviewed using a quantified grading rubric developed by the researchers. In a prior evaluation, Handel7 reviewed 35 health and wellness mobile apps utilizing the categories of ease of use, reliability, quality, scope of information, and aesthetics.4 These criteria were modified and adapted for the purposes of this study, and a 4-point scale was applied to each criterion. The final criteria were (1) educational objectives, (2) content, (3) accuracy, (4) design, and (5) conflict of interest. The quantified grading rubric is described in Table 1.

Results
The possible range of scores based on the grading rubric was 5 to 20. The actual range of scores was 8 to 19 (Table 2). The 44 reviewed apps were categorized by topic as acne, cosmetic dermatology, eczema, general dermatology, psoriasis, or skin cancer. A sample of 15 apps selected to represent the distribution of scores and their grading on the rubric are presented in Table 3.


Comment
The number of dermatology-related apps available to mobile users continues to grow at an increasing rate.8 The apps vary in many aspects, including their purpose, scope, intended audience, and goals of the app publisher. In turn, more individuals are turning to mobile apps for medical information,4 especially in dermatology, thus it is necessary to create a systematic way to evaluate the quality and utility of each app to assist users in making informed decisions about which apps will best meet their needs in the midst of a wide array of choices.
For the purpose of this study, an objective rubric was created that can be used to evaluate the quality of medical apps for patient education in dermatology. An app’s adequacy and usefulness for patient education was thought to depend on 3 possible score ranges into which the app could fall based on the grading rubric. An app with a total score in the range of 5 to 10 was not thought to be useful and may even be detrimental to patients. An app with a total score in the range of 11 to 15 may be used for patient education with some reservations based on shortcomings for certain criteria. An app with a score in the range of 16 to 20 was thought to be valuable and adequate for patient education. For example, the How to Treat Acne app received a total score of 8 and therefore would not be recommended to patients based on the grading rubric used in this study. This particular app provided sparse and sometimes inaccurate information, had a confusing user interface, and contained many obstructive advertisements. In contrast, the Eczema Doc app received a total score of 19, which indicates a quality app deemed to be useful for patient information based on the established rubric. This app met all the objectives that it advertised, contained accurate information with verified citation of sources, and was very easy for users to navigate.
Of the 44 graded apps, only 9 (20.5%) received scores in the highest range of 16 to 20, which indicates a need for improvements in mobile dermatology apps intended for patient education. Adopting the grading rubric developed in this study as a standard in the creation of medical apps could have beneficial implications in disseminating accurate, safe, unbiased, and easy-to-understand information to patients.
- Smith A. U.S. smartphone use in 2015. Pew Research Center website. http://www.pewinternet.org/2015/04/01/us-smartphone-use-in-2015. Published April 1, 2015. Accessed August 29, 2017.
- Nilsen W, Kumar S, Shar A, et al. Advancing the science of mHealth. J Health Commun. 2012;17(suppl 1):5-10.
- West DM. How mobile devices are transforming healthcare issues in technology innovation. Issues Technol Innov. 2012;18:1-14.
- Boudreaux ED, Waring ME, Hayes RB, et al. Evaluating and selecting mobile health apps: strategies for healthcare providers and healthcare organizations. Transl Behav Med. 2014;4:363-371.
- Brewer AC, Endly DC, Henley J, et al. Mobile applications in dermatology. JAMA Dermatol. 2013;149:1300-1304.
- Cummings E, Borycki E, Roehrer E. Issues and considerations for healthcare consumers using mobile applications. Stud Health Technol Inform. 2013;183:227-231.
- Handel MJ. mHealth (mobile health)-using apps for health and wellness. Explore. 2011;7:256-261.
- Boulos MN, Brewer AC, Karimkhani C, et al. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J Public Health Inform. 2014;5:229.
- Smith A. U.S. smartphone use in 2015. Pew Research Center website. http://www.pewinternet.org/2015/04/01/us-smartphone-use-in-2015. Published April 1, 2015. Accessed August 29, 2017.
- Nilsen W, Kumar S, Shar A, et al. Advancing the science of mHealth. J Health Commun. 2012;17(suppl 1):5-10.
- West DM. How mobile devices are transforming healthcare issues in technology innovation. Issues Technol Innov. 2012;18:1-14.
- Boudreaux ED, Waring ME, Hayes RB, et al. Evaluating and selecting mobile health apps: strategies for healthcare providers and healthcare organizations. Transl Behav Med. 2014;4:363-371.
- Brewer AC, Endly DC, Henley J, et al. Mobile applications in dermatology. JAMA Dermatol. 2013;149:1300-1304.
- Cummings E, Borycki E, Roehrer E. Issues and considerations for healthcare consumers using mobile applications. Stud Health Technol Inform. 2013;183:227-231.
- Handel MJ. mHealth (mobile health)-using apps for health and wellness. Explore. 2011;7:256-261.
- Boulos MN, Brewer AC, Karimkhani C, et al. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J Public Health Inform. 2014;5:229.
Practice Points
- Mobile dermatology apps for educational purposes should be objectively reviewed before being used by patients.
- In our study, only 9 (20.5%) of the 44 dermatology apps evaluated were considered adequate for patient information based on our grading criteria.
VIDEO: Dupilumab or cyclosporine for treating atopic dermatitis?
KAUAI, HAWAII – Sometimes, older is better, according to Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland.
Dr. Simpson was a key investigator in trials that were the basis of dupilumab’s approval in 2017 for adults with moderate to severe atopic dermatitis (AD), but there’s still a role for cyclosporine and other old standbys, he said in a video interview at the Hawaii Dermatology Seminar, provided by Global Academy for Medical Education/Skin Disease Education Foundation.
He said he’s asked all the time how to pick a systemic treatment for AD when topicals aren’t doing the trick. In the interview, he explained how dupilumab (Dupixent) fits into the picture, and how to select the right systemic therapy for the right patient. There are not a lot of data yet pointing to one option over the others for first-line treatment; a lot of it comes down to clinical smarts and patient preference.
Dr. Simpson is a consultant and/or investigator for a number of companies, including Eli Lilly, Pfizer, Novartis, and dupilumab manufacturer, Regeneron.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Sometimes, older is better, according to Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland.
Dr. Simpson was a key investigator in trials that were the basis of dupilumab’s approval in 2017 for adults with moderate to severe atopic dermatitis (AD), but there’s still a role for cyclosporine and other old standbys, he said in a video interview at the Hawaii Dermatology Seminar, provided by Global Academy for Medical Education/Skin Disease Education Foundation.
He said he’s asked all the time how to pick a systemic treatment for AD when topicals aren’t doing the trick. In the interview, he explained how dupilumab (Dupixent) fits into the picture, and how to select the right systemic therapy for the right patient. There are not a lot of data yet pointing to one option over the others for first-line treatment; a lot of it comes down to clinical smarts and patient preference.
Dr. Simpson is a consultant and/or investigator for a number of companies, including Eli Lilly, Pfizer, Novartis, and dupilumab manufacturer, Regeneron.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Sometimes, older is better, according to Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland.
Dr. Simpson was a key investigator in trials that were the basis of dupilumab’s approval in 2017 for adults with moderate to severe atopic dermatitis (AD), but there’s still a role for cyclosporine and other old standbys, he said in a video interview at the Hawaii Dermatology Seminar, provided by Global Academy for Medical Education/Skin Disease Education Foundation.
He said he’s asked all the time how to pick a systemic treatment for AD when topicals aren’t doing the trick. In the interview, he explained how dupilumab (Dupixent) fits into the picture, and how to select the right systemic therapy for the right patient. There are not a lot of data yet pointing to one option over the others for first-line treatment; a lot of it comes down to clinical smarts and patient preference.
Dr. Simpson is a consultant and/or investigator for a number of companies, including Eli Lilly, Pfizer, Novartis, and dupilumab manufacturer, Regeneron.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
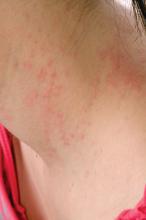
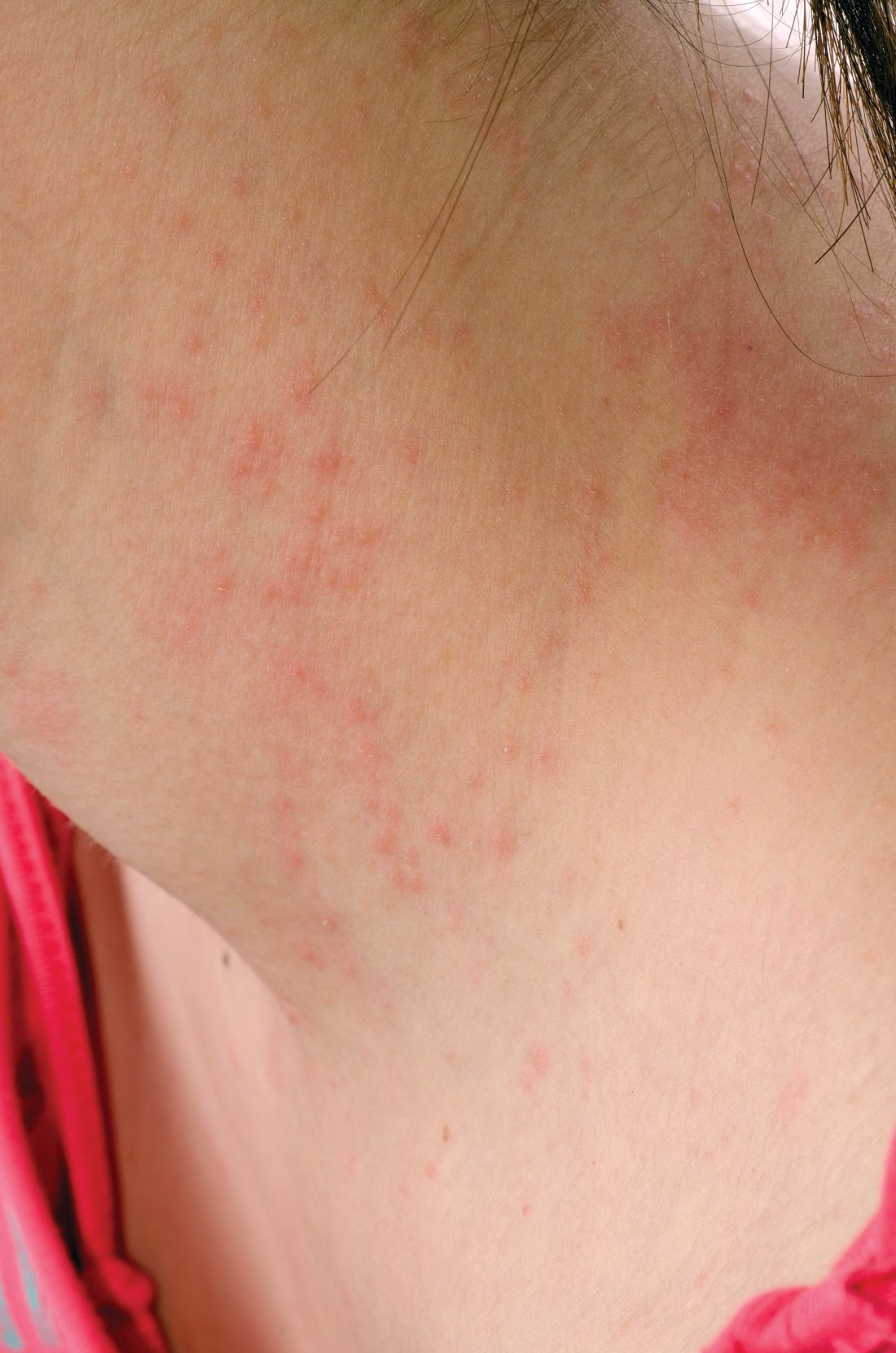
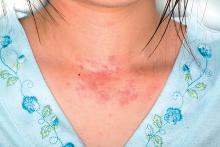
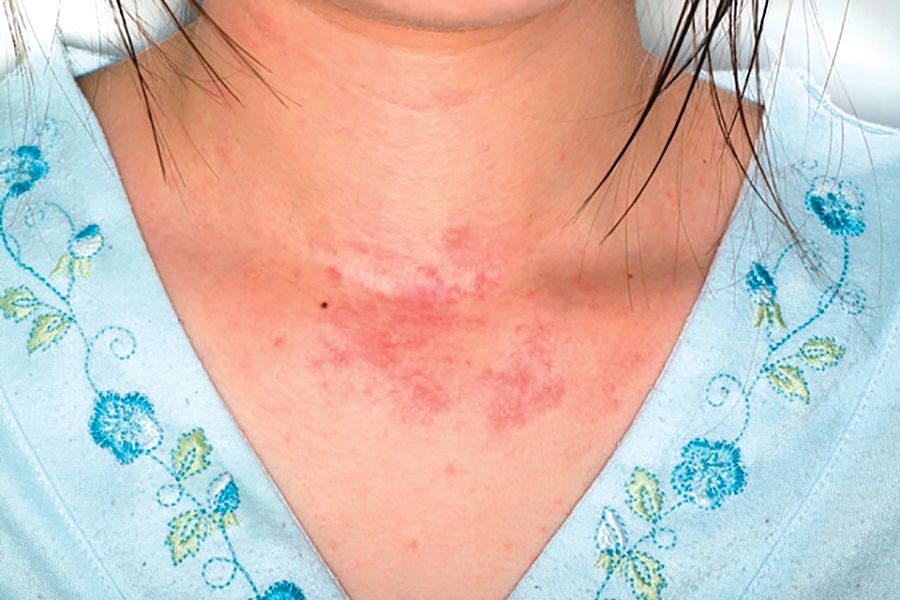
Topical steroid reduces atopic dermatitis relapse in children
according to Elena Rubio-Gomis of the Consorcio Hospital General Universitario de Valencia, Spain, and her associates.
Most atopic disease management is based on managing active disease, but atopic dermatitis is a chronic relapsing disease for which long-term maintenance therapies are being sought.
“Fluticasone 0.05% cream is a potent topical corticosteroid with a low systemic absorption and rapid metabolism and clearance,” which argues for low likelihood of causing systemic side effects and possible use in preventing atopic dermatitis relapse, the investigators wrote. And it is not very expensive.
In a randomized, double-blind, placebo-controlled study of 54 children with mild to moderate atopic dermatitis treated with twice-daily fluticasone propionate 0.05% cream up to 2 weeks, 49 children aged 2-10 years entered the second phase of the study. These children were randomly assigned to receive fluticasone propionate or vehicle twice weekly on consecutive days for 16 weeks; treatment was stopped then or at relapse.
Children treated with fluticasone propionate had a 2.7-fold lower risk of relapse than that of children treated with vehicle; 7 of 26 (27%) in the fluticasone propionate group and 13 of 23 patients (57%) in the vehicle group experienced relapse, the investigators wrote. The report was published in Allergologia et Immunopathologia. The median time to relapse with fluticasone propionate exceeded 16 weeks (the duration of the study), while in the vehicle group it was 65 days (a little over 9 weeks).
The treatment with fluticasone propionate and vehicle both were well tolerated. During the second phase of the study, 9 patients (35%) reported at least one adverse event in the fluticasone propionate group and 11 (48%) in the vehicle group. None of these were believed to be related to the drug studied, the investigators said.
SOURCE: Rubio-Gomis E et al. Allergol Immunopathol (Madr). 2018. doi: 10.1016/j.aller.2017.12.001.
according to Elena Rubio-Gomis of the Consorcio Hospital General Universitario de Valencia, Spain, and her associates.
Most atopic disease management is based on managing active disease, but atopic dermatitis is a chronic relapsing disease for which long-term maintenance therapies are being sought.
“Fluticasone 0.05% cream is a potent topical corticosteroid with a low systemic absorption and rapid metabolism and clearance,” which argues for low likelihood of causing systemic side effects and possible use in preventing atopic dermatitis relapse, the investigators wrote. And it is not very expensive.
In a randomized, double-blind, placebo-controlled study of 54 children with mild to moderate atopic dermatitis treated with twice-daily fluticasone propionate 0.05% cream up to 2 weeks, 49 children aged 2-10 years entered the second phase of the study. These children were randomly assigned to receive fluticasone propionate or vehicle twice weekly on consecutive days for 16 weeks; treatment was stopped then or at relapse.
Children treated with fluticasone propionate had a 2.7-fold lower risk of relapse than that of children treated with vehicle; 7 of 26 (27%) in the fluticasone propionate group and 13 of 23 patients (57%) in the vehicle group experienced relapse, the investigators wrote. The report was published in Allergologia et Immunopathologia. The median time to relapse with fluticasone propionate exceeded 16 weeks (the duration of the study), while in the vehicle group it was 65 days (a little over 9 weeks).
The treatment with fluticasone propionate and vehicle both were well tolerated. During the second phase of the study, 9 patients (35%) reported at least one adverse event in the fluticasone propionate group and 11 (48%) in the vehicle group. None of these were believed to be related to the drug studied, the investigators said.
SOURCE: Rubio-Gomis E et al. Allergol Immunopathol (Madr). 2018. doi: 10.1016/j.aller.2017.12.001.
according to Elena Rubio-Gomis of the Consorcio Hospital General Universitario de Valencia, Spain, and her associates.
Most atopic disease management is based on managing active disease, but atopic dermatitis is a chronic relapsing disease for which long-term maintenance therapies are being sought.
“Fluticasone 0.05% cream is a potent topical corticosteroid with a low systemic absorption and rapid metabolism and clearance,” which argues for low likelihood of causing systemic side effects and possible use in preventing atopic dermatitis relapse, the investigators wrote. And it is not very expensive.
In a randomized, double-blind, placebo-controlled study of 54 children with mild to moderate atopic dermatitis treated with twice-daily fluticasone propionate 0.05% cream up to 2 weeks, 49 children aged 2-10 years entered the second phase of the study. These children were randomly assigned to receive fluticasone propionate or vehicle twice weekly on consecutive days for 16 weeks; treatment was stopped then or at relapse.
Children treated with fluticasone propionate had a 2.7-fold lower risk of relapse than that of children treated with vehicle; 7 of 26 (27%) in the fluticasone propionate group and 13 of 23 patients (57%) in the vehicle group experienced relapse, the investigators wrote. The report was published in Allergologia et Immunopathologia. The median time to relapse with fluticasone propionate exceeded 16 weeks (the duration of the study), while in the vehicle group it was 65 days (a little over 9 weeks).
The treatment with fluticasone propionate and vehicle both were well tolerated. During the second phase of the study, 9 patients (35%) reported at least one adverse event in the fluticasone propionate group and 11 (48%) in the vehicle group. None of these were believed to be related to the drug studied, the investigators said.
SOURCE: Rubio-Gomis E et al. Allergol Immunopathol (Madr). 2018. doi: 10.1016/j.aller.2017.12.001.
FROM ALLERGOLOGIA ET IMMUNOPATHOLOGIA
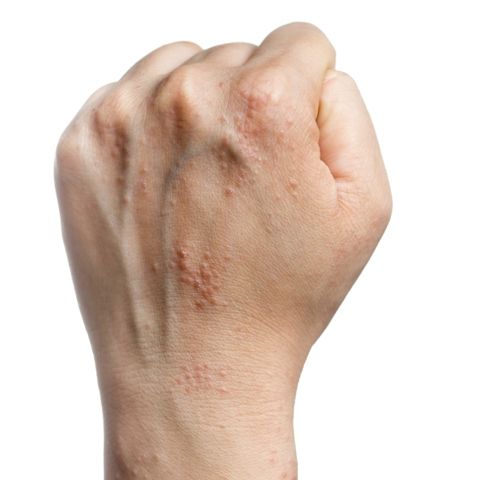
Antihistamines still are prescribed inappropriately for atopic dermatitis
said Alice He, MD, at the Center for Dermatology Research, Wake Forest University, Winston-Salem, N.C., and her associates.
Results of double-blind, randomized trials have found that oral antihistamines do not effectively treat itch associated with atopic dermatitis (AD), and American Academy of Dermatology guidelines state that intermittent short-term use of sedating antihistamines may help insomnia secondary to itch, but should not be substituted for topical treatments in atopic dermatitis management (J Am Acad Dermatol. 2014;71[2]:327-49), the investigators said. Nonetheless, physicians continue to prescribe antihistamines to AD patients.
Sedating antihistamines accounted for the majority of antihistamines prescribed by pediatricians (58%) and dermatologists (70%) for AD. Nonsedating antihistamines accounted for the majority of the antihistamines prescribed by family physicians (84%), internists (100%), and other specialists (55%), the investigators said.
(The sedating antihistamines prescribed included hydroxyzine, diphenhydramine, chlorpheniramine, brompheniramine, and cyproheptadine; nonsedating antihistamines include cetirizine, desloratadine, levocetirizine, loratadine, and fexofenadine.)
Although the AAD guidelines allow intermittent, short-term use of sedating antihistamines to treat AD-associated itch, these medications may be harmful to sleep in terms of reduced sleep quality and decreased REM sleep. Also, studies have shown that their effects can persist into the daytime and have the potential to impair cognitive function, including learning and memory.
“Physicians should consider the potential risks when prescribing sedating antihistamines as adjunctive treatment for atopic dermatitis,” Dr. He and her associates warned in the article in the Journal of the American Academy of Dermatology.
The researchers took a look at the top ten comorbidities reported among visits for AD, because nonsedating antihistamines might have been prescribed for a different diagnosis. There were three conditions for which antihistamines may be indicated: allergic rhinitis (13%), dermatitis attributable to food (8.5%), and conjunctivitis (3%). This “validates antihistamine prescriptions for a maximum of only 24.5% of patients who received antihistamines, indicating that a large proportion of antihistamine prescriptions were prescribed specifically for AD,” they said.
The authors said that, “while histamine does not play a role in AD via H1R [histamine-1 receptor], it may still be relevant in AD pathogenesis through actions on its most recently described receptor, histamine-4 receptor (H4R).”
“The advent of new targeted systemic therapies, such as T-helper 2 cytokine antagonists and H4R antagonists,” may one day help fill “the unmet need for effective treatments for atopic dermatitis,” Dr. He and her associates concluded.
The Center for Dermatology Research is supported by an unrestricted educational grant from Galderma Laboratories. Dr. He listed no other relevant financial disclosures. Her coauthors disclosed research, speaking, and/or consulting support from a variety of pharmaceutical companies; one author is an employee of Abbvie.
SOURCE: He A et al. J Amer Acad of Dermatol. 2008. doi: 10.1016/j.jaad.2017.12.077.
said Alice He, MD, at the Center for Dermatology Research, Wake Forest University, Winston-Salem, N.C., and her associates.
Results of double-blind, randomized trials have found that oral antihistamines do not effectively treat itch associated with atopic dermatitis (AD), and American Academy of Dermatology guidelines state that intermittent short-term use of sedating antihistamines may help insomnia secondary to itch, but should not be substituted for topical treatments in atopic dermatitis management (J Am Acad Dermatol. 2014;71[2]:327-49), the investigators said. Nonetheless, physicians continue to prescribe antihistamines to AD patients.
Sedating antihistamines accounted for the majority of antihistamines prescribed by pediatricians (58%) and dermatologists (70%) for AD. Nonsedating antihistamines accounted for the majority of the antihistamines prescribed by family physicians (84%), internists (100%), and other specialists (55%), the investigators said.
(The sedating antihistamines prescribed included hydroxyzine, diphenhydramine, chlorpheniramine, brompheniramine, and cyproheptadine; nonsedating antihistamines include cetirizine, desloratadine, levocetirizine, loratadine, and fexofenadine.)
Although the AAD guidelines allow intermittent, short-term use of sedating antihistamines to treat AD-associated itch, these medications may be harmful to sleep in terms of reduced sleep quality and decreased REM sleep. Also, studies have shown that their effects can persist into the daytime and have the potential to impair cognitive function, including learning and memory.
“Physicians should consider the potential risks when prescribing sedating antihistamines as adjunctive treatment for atopic dermatitis,” Dr. He and her associates warned in the article in the Journal of the American Academy of Dermatology.
The researchers took a look at the top ten comorbidities reported among visits for AD, because nonsedating antihistamines might have been prescribed for a different diagnosis. There were three conditions for which antihistamines may be indicated: allergic rhinitis (13%), dermatitis attributable to food (8.5%), and conjunctivitis (3%). This “validates antihistamine prescriptions for a maximum of only 24.5% of patients who received antihistamines, indicating that a large proportion of antihistamine prescriptions were prescribed specifically for AD,” they said.
The authors said that, “while histamine does not play a role in AD via H1R [histamine-1 receptor], it may still be relevant in AD pathogenesis through actions on its most recently described receptor, histamine-4 receptor (H4R).”
“The advent of new targeted systemic therapies, such as T-helper 2 cytokine antagonists and H4R antagonists,” may one day help fill “the unmet need for effective treatments for atopic dermatitis,” Dr. He and her associates concluded.
The Center for Dermatology Research is supported by an unrestricted educational grant from Galderma Laboratories. Dr. He listed no other relevant financial disclosures. Her coauthors disclosed research, speaking, and/or consulting support from a variety of pharmaceutical companies; one author is an employee of Abbvie.
SOURCE: He A et al. J Amer Acad of Dermatol. 2008. doi: 10.1016/j.jaad.2017.12.077.
said Alice He, MD, at the Center for Dermatology Research, Wake Forest University, Winston-Salem, N.C., and her associates.
Results of double-blind, randomized trials have found that oral antihistamines do not effectively treat itch associated with atopic dermatitis (AD), and American Academy of Dermatology guidelines state that intermittent short-term use of sedating antihistamines may help insomnia secondary to itch, but should not be substituted for topical treatments in atopic dermatitis management (J Am Acad Dermatol. 2014;71[2]:327-49), the investigators said. Nonetheless, physicians continue to prescribe antihistamines to AD patients.
Sedating antihistamines accounted for the majority of antihistamines prescribed by pediatricians (58%) and dermatologists (70%) for AD. Nonsedating antihistamines accounted for the majority of the antihistamines prescribed by family physicians (84%), internists (100%), and other specialists (55%), the investigators said.
(The sedating antihistamines prescribed included hydroxyzine, diphenhydramine, chlorpheniramine, brompheniramine, and cyproheptadine; nonsedating antihistamines include cetirizine, desloratadine, levocetirizine, loratadine, and fexofenadine.)
Although the AAD guidelines allow intermittent, short-term use of sedating antihistamines to treat AD-associated itch, these medications may be harmful to sleep in terms of reduced sleep quality and decreased REM sleep. Also, studies have shown that their effects can persist into the daytime and have the potential to impair cognitive function, including learning and memory.
“Physicians should consider the potential risks when prescribing sedating antihistamines as adjunctive treatment for atopic dermatitis,” Dr. He and her associates warned in the article in the Journal of the American Academy of Dermatology.
The researchers took a look at the top ten comorbidities reported among visits for AD, because nonsedating antihistamines might have been prescribed for a different diagnosis. There were three conditions for which antihistamines may be indicated: allergic rhinitis (13%), dermatitis attributable to food (8.5%), and conjunctivitis (3%). This “validates antihistamine prescriptions for a maximum of only 24.5% of patients who received antihistamines, indicating that a large proportion of antihistamine prescriptions were prescribed specifically for AD,” they said.
The authors said that, “while histamine does not play a role in AD via H1R [histamine-1 receptor], it may still be relevant in AD pathogenesis through actions on its most recently described receptor, histamine-4 receptor (H4R).”
“The advent of new targeted systemic therapies, such as T-helper 2 cytokine antagonists and H4R antagonists,” may one day help fill “the unmet need for effective treatments for atopic dermatitis,” Dr. He and her associates concluded.
The Center for Dermatology Research is supported by an unrestricted educational grant from Galderma Laboratories. Dr. He listed no other relevant financial disclosures. Her coauthors disclosed research, speaking, and/or consulting support from a variety of pharmaceutical companies; one author is an employee of Abbvie.
SOURCE: He A et al. J Amer Acad of Dermatol. 2008. doi: 10.1016/j.jaad.2017.12.077.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: Physicians should consider the potential risks when prescribing sedating antihistamines as adjunctive treatment for atopic dermatitis.
Major finding: Physicians from specialties other than dermatology prescribed antihistamines at a greater proportion of AD visits than dermatologists (26%-44% of visits vs. 22%), with the exception of pediatricians (16%).
Study details: Data from the National Ambulatory Medical Care Survey on 9.9 million physician visits for AD from 2003 to 2012.
Disclosures: The Center for Dermatology Research is supported by an unrestricted educational grant from Galderma Laboratories. Dr. He listed no other relevant financial disclosures. Her associates disclosed research, speaking and/or consulting support from a variety of pharmaceutical companies. One author was an employee of Abbvie.
Source: He A et al. J Amer Acad of Dermatol. 2008. doi: 10.1016/j.jaad.2017.12.077.
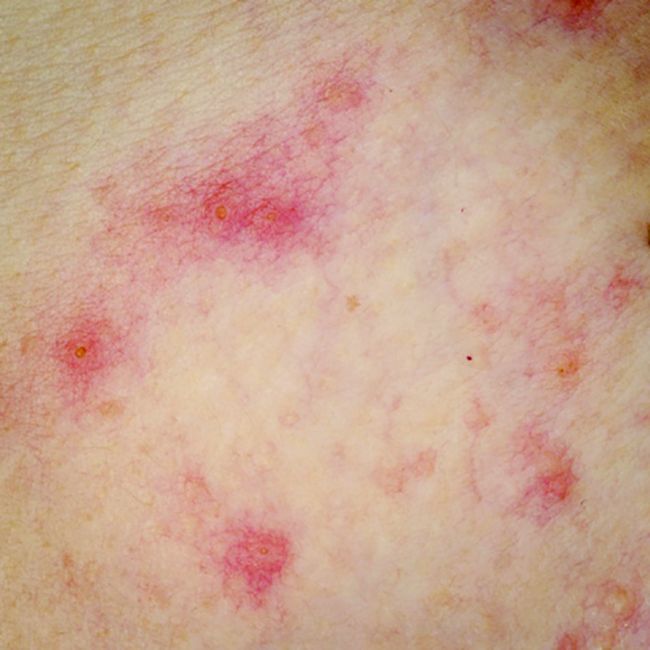
Atopic dermatitis linked to psychiatric disorders but not hospitalizations
Atopic dermatitis (AD) is associated with anxiety, depression, and suicidal thoughts but does not lead to increased psychiatric hospitalization or suicide, according to a Danish study.
While previous literature has indicated a connection between AD and depressive symptoms, these findings indicated the psychiatric burden is not severe.
A total of 9,656 patients selected from the Danish study of Functional Disorders (DanFunD) during 2011-2015 were asked to fill out a questionnaire concerning clinical diagnoses and psychiatric symptoms.
Investigators also conducted an analysis of 4,259,457 patient records gathered the Danish National Patient Register.
The 1,044 AD patients from the DanFunD were slightly younger on average, at 50 years, compared with an average of 53 years in the 8,612-person control group; there were more women (65%) in the AD group, compared with a relatively even split (53%) in the control group.
Patients with AD were more likely to have clinically diagnosed depression (odds ratio, 1.76) or anxiety (OR, 1.61) than were those in the control group.
The report was published in the journal Allergy.
Among the 568 individuals with symptoms consistent of major depressive disorder, those with AD (112) were almost twice as likely to report these symptoms on the questionnaire (OR, 1.92) and more than twice as likely to report anxiety attacks (OR, 2.32), despite differences in psychiatric hospitalizations remaining insignificant.
The investigators hypothesized the treatment methods for AD patients may be part of the reason that suicide and hospitalization rates were not higher.
“These more severe outcomes of suicide and psychiatric hospitalization could be mitigated in AD patients due to their increased general health care and psychiatric health care utilization leading to, for example, earlier and more aggressive antidepressant therapy,” according to Dr. Thyssen and his fellow investigators.
In an analysis of the general Danish population, medication was most aggressive in patients with moderate-severe AD, who were significantly more likely to use antidepressants (hazard ratio,1.24) and anxiolytic drugs (HR, 1.66), while those with milder symptoms had only a slightly increased likelihood.
Because of the observational nature of the study, investigators could not determine what the specific cause of these psychiatric symptoms were.
The DanFunD Study was supported by TrygFonden and Lundbeck Foundation. Dr. Thyssen is supported by a grant from the Lundbeck Foundation. Some of the investigators received financial support from various pharmaceutical companies or their foundations. The remaining investigators reported no relevant financial disclosures.
ezimmerman@frontlinemedcom.com
SOURCE: Thyssen JP et al. Allergy. 2018 Jan;73(1):214-20.
Atopic dermatitis (AD) is associated with anxiety, depression, and suicidal thoughts but does not lead to increased psychiatric hospitalization or suicide, according to a Danish study.
While previous literature has indicated a connection between AD and depressive symptoms, these findings indicated the psychiatric burden is not severe.
A total of 9,656 patients selected from the Danish study of Functional Disorders (DanFunD) during 2011-2015 were asked to fill out a questionnaire concerning clinical diagnoses and psychiatric symptoms.
Investigators also conducted an analysis of 4,259,457 patient records gathered the Danish National Patient Register.
The 1,044 AD patients from the DanFunD were slightly younger on average, at 50 years, compared with an average of 53 years in the 8,612-person control group; there were more women (65%) in the AD group, compared with a relatively even split (53%) in the control group.
Patients with AD were more likely to have clinically diagnosed depression (odds ratio, 1.76) or anxiety (OR, 1.61) than were those in the control group.
The report was published in the journal Allergy.
Among the 568 individuals with symptoms consistent of major depressive disorder, those with AD (112) were almost twice as likely to report these symptoms on the questionnaire (OR, 1.92) and more than twice as likely to report anxiety attacks (OR, 2.32), despite differences in psychiatric hospitalizations remaining insignificant.
The investigators hypothesized the treatment methods for AD patients may be part of the reason that suicide and hospitalization rates were not higher.
“These more severe outcomes of suicide and psychiatric hospitalization could be mitigated in AD patients due to their increased general health care and psychiatric health care utilization leading to, for example, earlier and more aggressive antidepressant therapy,” according to Dr. Thyssen and his fellow investigators.
In an analysis of the general Danish population, medication was most aggressive in patients with moderate-severe AD, who were significantly more likely to use antidepressants (hazard ratio,1.24) and anxiolytic drugs (HR, 1.66), while those with milder symptoms had only a slightly increased likelihood.
Because of the observational nature of the study, investigators could not determine what the specific cause of these psychiatric symptoms were.
The DanFunD Study was supported by TrygFonden and Lundbeck Foundation. Dr. Thyssen is supported by a grant from the Lundbeck Foundation. Some of the investigators received financial support from various pharmaceutical companies or their foundations. The remaining investigators reported no relevant financial disclosures.
ezimmerman@frontlinemedcom.com
SOURCE: Thyssen JP et al. Allergy. 2018 Jan;73(1):214-20.
Atopic dermatitis (AD) is associated with anxiety, depression, and suicidal thoughts but does not lead to increased psychiatric hospitalization or suicide, according to a Danish study.
While previous literature has indicated a connection between AD and depressive symptoms, these findings indicated the psychiatric burden is not severe.
A total of 9,656 patients selected from the Danish study of Functional Disorders (DanFunD) during 2011-2015 were asked to fill out a questionnaire concerning clinical diagnoses and psychiatric symptoms.
Investigators also conducted an analysis of 4,259,457 patient records gathered the Danish National Patient Register.
The 1,044 AD patients from the DanFunD were slightly younger on average, at 50 years, compared with an average of 53 years in the 8,612-person control group; there were more women (65%) in the AD group, compared with a relatively even split (53%) in the control group.
Patients with AD were more likely to have clinically diagnosed depression (odds ratio, 1.76) or anxiety (OR, 1.61) than were those in the control group.
The report was published in the journal Allergy.
Among the 568 individuals with symptoms consistent of major depressive disorder, those with AD (112) were almost twice as likely to report these symptoms on the questionnaire (OR, 1.92) and more than twice as likely to report anxiety attacks (OR, 2.32), despite differences in psychiatric hospitalizations remaining insignificant.
The investigators hypothesized the treatment methods for AD patients may be part of the reason that suicide and hospitalization rates were not higher.
“These more severe outcomes of suicide and psychiatric hospitalization could be mitigated in AD patients due to their increased general health care and psychiatric health care utilization leading to, for example, earlier and more aggressive antidepressant therapy,” according to Dr. Thyssen and his fellow investigators.
In an analysis of the general Danish population, medication was most aggressive in patients with moderate-severe AD, who were significantly more likely to use antidepressants (hazard ratio,1.24) and anxiolytic drugs (HR, 1.66), while those with milder symptoms had only a slightly increased likelihood.
Because of the observational nature of the study, investigators could not determine what the specific cause of these psychiatric symptoms were.
The DanFunD Study was supported by TrygFonden and Lundbeck Foundation. Dr. Thyssen is supported by a grant from the Lundbeck Foundation. Some of the investigators received financial support from various pharmaceutical companies or their foundations. The remaining investigators reported no relevant financial disclosures.
ezimmerman@frontlinemedcom.com
SOURCE: Thyssen JP et al. Allergy. 2018 Jan;73(1):214-20.
FROM ALLERGY
Key clinical point:
Major finding: Atopic dermatitis patients were at greater risk for depression (OR, 1.76) and anxiety (OR, 1.61).
Study details: Observational, controlled study of 9,656 Danish residents collected from the Danish study of Functional Disorders between 2011 and 2015, and an analysis of 4,259,457 patient records gathered the Danish National Patient Register.
Disclosures: The DanFunD Study was supported by TrygFonden and Lundbeck Foundation. Dr. Thyssen is supported by a grant from the Lundbeck Foundation. Some of the investigators received financial support from various pharmaceutical companies or their foundations. The remaining investigators reported no relevant financial disclosures.
Source: Thyssen JP et al. Allergy. 2018 Jan;73(1):214-20.
Atopic march largely attributed to genetic factors
according to a systematic review.
PhD candidate Sabria Khan and her colleagues at the Centre for Epidemiology and Biostatistics at the University of Melbourne said that the atopic march concept “asserts that allergic diseases start in early life with eczema, progress through food allergy, and culminate with hay fever and asthma.”
This systematic review of ten twin and sibling studies looked at known, measured environmental and genetic influences on the associations between the atopic phenotypes of the atopic march.
The studies of asthma and hay fever suggested that the prevalence of having both conditions was high (32%) and that they were more likely to occur together in monozygotic twins than they were in dizygotic twins. Similarly, other studies found a high phenotypic overlap between eczema and asthma and between eczema and hay fever, which was more pronounced in monozygotic twins than in dizygotic twins.
“Asthma is linked to hay fever and eczema through intermediate phenotypes like clinical measures of lung function, physiological measures of airway responsiveness and the biomarker exhaled nitric oxide, all of which are influenced by hereditary factors,” the authors said.
Overall, they concluded that genetic factors account for 75% of eczema cases, 70%-91% of asthma cases, and 72%-84% of hay fever cases, making them all highly heritable diseases.
“Our study found that the contribution of shared environmental factors to the proportion of correlation are very low (from 4% to 18%) and does not explain the familial patterns seen for asthma and hay fever,” they reported. “This finding contradicts various analyses where smoking behavior, indoor-outdoor pollution, and house dust mites were found to be significant risk factor for asthma and hay fever that are shared by siblings.”
The authors commented that preventing the onset of the atopic march, or arresting its development, could have significant public health implications. They suggested that interventions such as oral antihistamines could be introduced either before a child gets eczema or before a child with eczema goes on to develop asthma or hay fever. “Two randomized controlled trials showed moisturizing the skin can prevent mild to moderate eczema, and long-term studies are needed to see whether such intervention will prevent development of asthma and hay fever,” they said.
No conflicts of interest were declared.
SOURCE: Khan SJ et al. Allergy. 2018 Jan;73(1):17-28.
according to a systematic review.
PhD candidate Sabria Khan and her colleagues at the Centre for Epidemiology and Biostatistics at the University of Melbourne said that the atopic march concept “asserts that allergic diseases start in early life with eczema, progress through food allergy, and culminate with hay fever and asthma.”
This systematic review of ten twin and sibling studies looked at known, measured environmental and genetic influences on the associations between the atopic phenotypes of the atopic march.
The studies of asthma and hay fever suggested that the prevalence of having both conditions was high (32%) and that they were more likely to occur together in monozygotic twins than they were in dizygotic twins. Similarly, other studies found a high phenotypic overlap between eczema and asthma and between eczema and hay fever, which was more pronounced in monozygotic twins than in dizygotic twins.
“Asthma is linked to hay fever and eczema through intermediate phenotypes like clinical measures of lung function, physiological measures of airway responsiveness and the biomarker exhaled nitric oxide, all of which are influenced by hereditary factors,” the authors said.
Overall, they concluded that genetic factors account for 75% of eczema cases, 70%-91% of asthma cases, and 72%-84% of hay fever cases, making them all highly heritable diseases.
“Our study found that the contribution of shared environmental factors to the proportion of correlation are very low (from 4% to 18%) and does not explain the familial patterns seen for asthma and hay fever,” they reported. “This finding contradicts various analyses where smoking behavior, indoor-outdoor pollution, and house dust mites were found to be significant risk factor for asthma and hay fever that are shared by siblings.”
The authors commented that preventing the onset of the atopic march, or arresting its development, could have significant public health implications. They suggested that interventions such as oral antihistamines could be introduced either before a child gets eczema or before a child with eczema goes on to develop asthma or hay fever. “Two randomized controlled trials showed moisturizing the skin can prevent mild to moderate eczema, and long-term studies are needed to see whether such intervention will prevent development of asthma and hay fever,” they said.
No conflicts of interest were declared.
SOURCE: Khan SJ et al. Allergy. 2018 Jan;73(1):17-28.
according to a systematic review.
PhD candidate Sabria Khan and her colleagues at the Centre for Epidemiology and Biostatistics at the University of Melbourne said that the atopic march concept “asserts that allergic diseases start in early life with eczema, progress through food allergy, and culminate with hay fever and asthma.”
This systematic review of ten twin and sibling studies looked at known, measured environmental and genetic influences on the associations between the atopic phenotypes of the atopic march.
The studies of asthma and hay fever suggested that the prevalence of having both conditions was high (32%) and that they were more likely to occur together in monozygotic twins than they were in dizygotic twins. Similarly, other studies found a high phenotypic overlap between eczema and asthma and between eczema and hay fever, which was more pronounced in monozygotic twins than in dizygotic twins.
“Asthma is linked to hay fever and eczema through intermediate phenotypes like clinical measures of lung function, physiological measures of airway responsiveness and the biomarker exhaled nitric oxide, all of which are influenced by hereditary factors,” the authors said.
Overall, they concluded that genetic factors account for 75% of eczema cases, 70%-91% of asthma cases, and 72%-84% of hay fever cases, making them all highly heritable diseases.
“Our study found that the contribution of shared environmental factors to the proportion of correlation are very low (from 4% to 18%) and does not explain the familial patterns seen for asthma and hay fever,” they reported. “This finding contradicts various analyses where smoking behavior, indoor-outdoor pollution, and house dust mites were found to be significant risk factor for asthma and hay fever that are shared by siblings.”
The authors commented that preventing the onset of the atopic march, or arresting its development, could have significant public health implications. They suggested that interventions such as oral antihistamines could be introduced either before a child gets eczema or before a child with eczema goes on to develop asthma or hay fever. “Two randomized controlled trials showed moisturizing the skin can prevent mild to moderate eczema, and long-term studies are needed to see whether such intervention will prevent development of asthma and hay fever,” they said.
No conflicts of interest were declared.
SOURCE: Khan SJ et al. Allergy. 2018 Jan;73(1):17-28.
FROM ALLERGY
Key clinical point: Twin and sibling studies suggest that genetics play a far more significant role than environmental factors in the progression of atopic disease in childhood known as the “atopic march.”
Major finding: Genetic factors account for 75% of eczema, 70%-91% of asthma, and 72%-84% of hay fever.
Data source: Systematic review of ten twin and sibling studies.
Disclosures: No conflicts of interest were declared.
Source: Khan SJ et al. Allergy. 2018 Jan;73(1):17-28.
FDA grants ‘Breakthrough Therapy Designation’ for upadacitinib for atopic dermatitis
The Food and Drug Administration has granted “Breakthrough Therapy Designation” for the investigational, once-daily oral Janus kinase 1 (JAK1)-selective inhibitor upadacitinib (ABT-494) in adult patients with moderate to severe atopic dermatitis who are candidates for systemic therapy.
The Breakthrough Therapy Designation is based on positive phase 2b results announced in Sept. 2017. The study found that patients treated with upadacitinib achieved statistically significant improvements in the primary endpoint (greater mean percentage change from baseline in Eczema Area and Severity Index score) and in all skin- and itch-specific secondary endpoints across all doses (30 mg, 15mg, or 7.5 mg once-daily) at week 16, compared with placebo (P less than .05). Reduction in itch was observed within the first week and improvement in skin within the first 2 weeks (P less than .001 across all doses). Of patients receiving the 30 mg once-daily dose of upadacitinib, 50% had clear or almost clear skin, according to a press release. There were 42 patients in each of the three treatment groups and 41 patients in the placebo group in this randomized, double-blind, parallel-group study sponsored by AbbVie, which discovered and developed upadacitinib.
The phase 3 clinical program is expected to begin in the first half of 2018, according to AbbVie. Any additional information on the clinical trials for upadacitinib is available at clinicaltrials.gov.
SOURCE: Prnewswire.com.
The Food and Drug Administration has granted “Breakthrough Therapy Designation” for the investigational, once-daily oral Janus kinase 1 (JAK1)-selective inhibitor upadacitinib (ABT-494) in adult patients with moderate to severe atopic dermatitis who are candidates for systemic therapy.
The Breakthrough Therapy Designation is based on positive phase 2b results announced in Sept. 2017. The study found that patients treated with upadacitinib achieved statistically significant improvements in the primary endpoint (greater mean percentage change from baseline in Eczema Area and Severity Index score) and in all skin- and itch-specific secondary endpoints across all doses (30 mg, 15mg, or 7.5 mg once-daily) at week 16, compared with placebo (P less than .05). Reduction in itch was observed within the first week and improvement in skin within the first 2 weeks (P less than .001 across all doses). Of patients receiving the 30 mg once-daily dose of upadacitinib, 50% had clear or almost clear skin, according to a press release. There were 42 patients in each of the three treatment groups and 41 patients in the placebo group in this randomized, double-blind, parallel-group study sponsored by AbbVie, which discovered and developed upadacitinib.
The phase 3 clinical program is expected to begin in the first half of 2018, according to AbbVie. Any additional information on the clinical trials for upadacitinib is available at clinicaltrials.gov.
SOURCE: Prnewswire.com.
The Food and Drug Administration has granted “Breakthrough Therapy Designation” for the investigational, once-daily oral Janus kinase 1 (JAK1)-selective inhibitor upadacitinib (ABT-494) in adult patients with moderate to severe atopic dermatitis who are candidates for systemic therapy.
The Breakthrough Therapy Designation is based on positive phase 2b results announced in Sept. 2017. The study found that patients treated with upadacitinib achieved statistically significant improvements in the primary endpoint (greater mean percentage change from baseline in Eczema Area and Severity Index score) and in all skin- and itch-specific secondary endpoints across all doses (30 mg, 15mg, or 7.5 mg once-daily) at week 16, compared with placebo (P less than .05). Reduction in itch was observed within the first week and improvement in skin within the first 2 weeks (P less than .001 across all doses). Of patients receiving the 30 mg once-daily dose of upadacitinib, 50% had clear or almost clear skin, according to a press release. There were 42 patients in each of the three treatment groups and 41 patients in the placebo group in this randomized, double-blind, parallel-group study sponsored by AbbVie, which discovered and developed upadacitinib.
The phase 3 clinical program is expected to begin in the first half of 2018, according to AbbVie. Any additional information on the clinical trials for upadacitinib is available at clinicaltrials.gov.
SOURCE: Prnewswire.com.
Debunking Atopic Dermatitis Myths: Should Patients Avoid Products With Parabens?
Myth: Parabens are dangerous
Some atopic dermatitis (AD) patients may be misinformed by reports that parabens have estrogenic and antiandrogenic effects and may be involved in carcinogenesis via endocrine modulation. Although in Europe some parabens have been banned or restricted, in the United States there are no regulations against the use of parabens in cosmetics. Dermatologists must acknowledge that their AD patients may have concerns about cosmetic products and they must be prepared to dispel any myths.
Parabens such as methylparaben, propylparaben, butylparaben, and ethylparaben are common in cosmetics such as moisturizers. Parabens have protective properties to prevent the growth of harmful bacteria and mold. According to the US Food and Drug Administration, “scientists continue to review published studies on the safety of parabens. At this time, we do not have information showing that parabens as they are used in cosmetics have an effect on human health. . . . If we determine that a health hazard exists, we will advise the industry and the public.”
Here are some important facts to note for patients, based on a research article published in Cosmetics & Toiletries in June 2017:
- Parabens are not toxic at the concentrations used in personal care products
- Parabens are not genotoxic or carcinogenic
- Parabens are readily excreted in urine and do not accumulate in tissues
In patients with chronic dermatitis, the Cosmetic Ingredient Review Expert Panel reported that parabens generally induce sensitization in less than 4% of patients. The panel concluded that they can support the safety of cosmetic products in which parabens are used as preservatives.
In fact, one study published in the Journal of the American Academy of Dermatology found that AD patients were not predisposed to allergies to parabens, formaldehyde, or diazolidinyl urea, but they were more likely to have allergic reactions to formaldehyde releasers. As a result, AD patients should choose moisturizers containing parabens and should have no fears about using them.
Expert Commentary
In general I recommend paraben-containing cleansers and emollients on a daily basis in practice. However, patient concerns exist due to negative online content easily accessed and fear can prevent usage of agents. Therefore, I am also open to offering options lacking in parabens, such as coconut oil.
—Nanette B. Silverberg, MD (New York, New York)
Doyle K. Some skin creams bad news for eczema. Reuters. December 12, 2013. https://www.reuters.com/article/us-skin-creams-eczema/some-skin-creams-bad-news-for-eczema-idUSBRE9BB14720131212. Accessed January 12, 2018.
Final amended report on the safety assessment of Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben as used in cosmetic products. Int J Toxicol. 2008;27(suppl 4):1-82.
Krowka JF, Loretz L, Geis PA, et al. Preserving the facts on parabens: an overview of these important tools of the trade. Cosmetics & Toiletries. June 1, 2017. http://www.cosmeticsandtoiletries.com/research/chemistry/Preserving-the-Facts-on-Parabens-An-Overview-of-These-Important-Tools-of-the-Trade-425784294.html. Accessed January 12, 2018.
Parabens in cosmetics. US Food and Drug Administration website. https://www.fda.gov/Cosmetics/ProductsIngredients/Ingredients/ucm128042.html. Accessed January 12, 2018.
Sasseville D, Alfalah M, Lacroix JP. “Parabenoia” debunked, or “who’s afraid of parabens?” Dermatitis. 2015;26:254-259.
Shaughnessy CN, Malajian D, Belsito DV. Cutaneous delayed-type hypersensitivity in patients with atopic dermatitis: reactivity to topical preservatives. J Am Acad Dermatol. 2014;70:102-107.
Myth: Parabens are dangerous
Some atopic dermatitis (AD) patients may be misinformed by reports that parabens have estrogenic and antiandrogenic effects and may be involved in carcinogenesis via endocrine modulation. Although in Europe some parabens have been banned or restricted, in the United States there are no regulations against the use of parabens in cosmetics. Dermatologists must acknowledge that their AD patients may have concerns about cosmetic products and they must be prepared to dispel any myths.
Parabens such as methylparaben, propylparaben, butylparaben, and ethylparaben are common in cosmetics such as moisturizers. Parabens have protective properties to prevent the growth of harmful bacteria and mold. According to the US Food and Drug Administration, “scientists continue to review published studies on the safety of parabens. At this time, we do not have information showing that parabens as they are used in cosmetics have an effect on human health. . . . If we determine that a health hazard exists, we will advise the industry and the public.”
Here are some important facts to note for patients, based on a research article published in Cosmetics & Toiletries in June 2017:
- Parabens are not toxic at the concentrations used in personal care products
- Parabens are not genotoxic or carcinogenic
- Parabens are readily excreted in urine and do not accumulate in tissues
In patients with chronic dermatitis, the Cosmetic Ingredient Review Expert Panel reported that parabens generally induce sensitization in less than 4% of patients. The panel concluded that they can support the safety of cosmetic products in which parabens are used as preservatives.
In fact, one study published in the Journal of the American Academy of Dermatology found that AD patients were not predisposed to allergies to parabens, formaldehyde, or diazolidinyl urea, but they were more likely to have allergic reactions to formaldehyde releasers. As a result, AD patients should choose moisturizers containing parabens and should have no fears about using them.
Expert Commentary
In general I recommend paraben-containing cleansers and emollients on a daily basis in practice. However, patient concerns exist due to negative online content easily accessed and fear can prevent usage of agents. Therefore, I am also open to offering options lacking in parabens, such as coconut oil.
—Nanette B. Silverberg, MD (New York, New York)
Myth: Parabens are dangerous
Some atopic dermatitis (AD) patients may be misinformed by reports that parabens have estrogenic and antiandrogenic effects and may be involved in carcinogenesis via endocrine modulation. Although in Europe some parabens have been banned or restricted, in the United States there are no regulations against the use of parabens in cosmetics. Dermatologists must acknowledge that their AD patients may have concerns about cosmetic products and they must be prepared to dispel any myths.
Parabens such as methylparaben, propylparaben, butylparaben, and ethylparaben are common in cosmetics such as moisturizers. Parabens have protective properties to prevent the growth of harmful bacteria and mold. According to the US Food and Drug Administration, “scientists continue to review published studies on the safety of parabens. At this time, we do not have information showing that parabens as they are used in cosmetics have an effect on human health. . . . If we determine that a health hazard exists, we will advise the industry and the public.”
Here are some important facts to note for patients, based on a research article published in Cosmetics & Toiletries in June 2017:
- Parabens are not toxic at the concentrations used in personal care products
- Parabens are not genotoxic or carcinogenic
- Parabens are readily excreted in urine and do not accumulate in tissues
In patients with chronic dermatitis, the Cosmetic Ingredient Review Expert Panel reported that parabens generally induce sensitization in less than 4% of patients. The panel concluded that they can support the safety of cosmetic products in which parabens are used as preservatives.
In fact, one study published in the Journal of the American Academy of Dermatology found that AD patients were not predisposed to allergies to parabens, formaldehyde, or diazolidinyl urea, but they were more likely to have allergic reactions to formaldehyde releasers. As a result, AD patients should choose moisturizers containing parabens and should have no fears about using them.
Expert Commentary
In general I recommend paraben-containing cleansers and emollients on a daily basis in practice. However, patient concerns exist due to negative online content easily accessed and fear can prevent usage of agents. Therefore, I am also open to offering options lacking in parabens, such as coconut oil.
—Nanette B. Silverberg, MD (New York, New York)
Doyle K. Some skin creams bad news for eczema. Reuters. December 12, 2013. https://www.reuters.com/article/us-skin-creams-eczema/some-skin-creams-bad-news-for-eczema-idUSBRE9BB14720131212. Accessed January 12, 2018.
Final amended report on the safety assessment of Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben as used in cosmetic products. Int J Toxicol. 2008;27(suppl 4):1-82.
Krowka JF, Loretz L, Geis PA, et al. Preserving the facts on parabens: an overview of these important tools of the trade. Cosmetics & Toiletries. June 1, 2017. http://www.cosmeticsandtoiletries.com/research/chemistry/Preserving-the-Facts-on-Parabens-An-Overview-of-These-Important-Tools-of-the-Trade-425784294.html. Accessed January 12, 2018.
Parabens in cosmetics. US Food and Drug Administration website. https://www.fda.gov/Cosmetics/ProductsIngredients/Ingredients/ucm128042.html. Accessed January 12, 2018.
Sasseville D, Alfalah M, Lacroix JP. “Parabenoia” debunked, or “who’s afraid of parabens?” Dermatitis. 2015;26:254-259.
Shaughnessy CN, Malajian D, Belsito DV. Cutaneous delayed-type hypersensitivity in patients with atopic dermatitis: reactivity to topical preservatives. J Am Acad Dermatol. 2014;70:102-107.
Doyle K. Some skin creams bad news for eczema. Reuters. December 12, 2013. https://www.reuters.com/article/us-skin-creams-eczema/some-skin-creams-bad-news-for-eczema-idUSBRE9BB14720131212. Accessed January 12, 2018.
Final amended report on the safety assessment of Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben, and Benzylparaben as used in cosmetic products. Int J Toxicol. 2008;27(suppl 4):1-82.
Krowka JF, Loretz L, Geis PA, et al. Preserving the facts on parabens: an overview of these important tools of the trade. Cosmetics & Toiletries. June 1, 2017. http://www.cosmeticsandtoiletries.com/research/chemistry/Preserving-the-Facts-on-Parabens-An-Overview-of-These-Important-Tools-of-the-Trade-425784294.html. Accessed January 12, 2018.
Parabens in cosmetics. US Food and Drug Administration website. https://www.fda.gov/Cosmetics/ProductsIngredients/Ingredients/ucm128042.html. Accessed January 12, 2018.
Sasseville D, Alfalah M, Lacroix JP. “Parabenoia” debunked, or “who’s afraid of parabens?” Dermatitis. 2015;26:254-259.
Shaughnessy CN, Malajian D, Belsito DV. Cutaneous delayed-type hypersensitivity in patients with atopic dermatitis: reactivity to topical preservatives. J Am Acad Dermatol. 2014;70:102-107.