User login
Blog: Could Sublative Rejuvenation Revolutionize Laser Field?
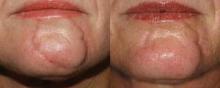
Sublation appears to be the new "it" word in skin laser rejuvenation. Studies have found that a device utilizing sublative technology was safe and effective for skin tightening and treating acne scars, even in darker skinned patients.
Dr. David J. Goldberg, director of dermatologic laser research at Mount Sinai School of Medicine, New York, presented early trial results at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
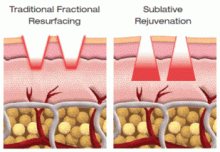
Besides being safe, effective, and offering minimal downtime, an immediate impact was seen after sublation through histopathology imaging. The small, fractionated, bipolar, radio-frequency device (Syneron eMatrix system) delivers energy deep into the dermis with limited skin surface involvement. Dr. Goldberg described the energy as entering an upside down cone. The energy spreads out as it goes deeper into the skin.
Compared with ablative resurfacing, sublative rejuvenation has the same healing physiological pathway (healing cascade) and the same initiating event (inflammation), said Dr. Goldberg. However, sublation has different perdominant events and there is a different proportion of dermal/epidermal involvement. Once again, said Dr. Goldberg, for delivering energy, think upside down cone, compared with the cone of ablation.
Initial studies at four sites in the United States and Canada of about 80 patients, with an average age of 52 and Fitzpatrick skin types I-IV, found that 83% of patients had improved skin brightness, 87% had improved skin tightness, and 90% had wrinkle improvement. Full data will be presented in April at the American Society for Laser Medicine and Surgery meeting in San Antonio.
In another study of patients with Fitzpatrick skin types IV-VI treated with the sublative laser, early results found improved photo damage and acne scarring after treatment. There was little erythema after treatment, said Dr. Goldberg.
He stressed that the results are early, and "only time will tell" if this device will live up to its early hype.
Dr. Goldberg disclosed receiving research grants from Syneron. SDEF and this news organization are owned by Elsevier.
-Amy Pfeiffer
Sublation appears to be the new "it" word in skin laser rejuvenation. Studies have found that a device utilizing sublative technology was safe and effective for skin tightening and treating acne scars, even in darker skinned patients.
Dr. David J. Goldberg, director of dermatologic laser research at Mount Sinai School of Medicine, New York, presented early trial results at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Besides being safe, effective, and offering minimal downtime, an immediate impact was seen after sublation through histopathology imaging. The small, fractionated, bipolar, radio-frequency device (Syneron eMatrix system) delivers energy deep into the dermis with limited skin surface involvement. Dr. Goldberg described the energy as entering an upside down cone. The energy spreads out as it goes deeper into the skin.
Compared with ablative resurfacing, sublative rejuvenation has the same healing physiological pathway (healing cascade) and the same initiating event (inflammation), said Dr. Goldberg. However, sublation has different perdominant events and there is a different proportion of dermal/epidermal involvement. Once again, said Dr. Goldberg, for delivering energy, think upside down cone, compared with the cone of ablation.
Initial studies at four sites in the United States and Canada of about 80 patients, with an average age of 52 and Fitzpatrick skin types I-IV, found that 83% of patients had improved skin brightness, 87% had improved skin tightness, and 90% had wrinkle improvement. Full data will be presented in April at the American Society for Laser Medicine and Surgery meeting in San Antonio.
In another study of patients with Fitzpatrick skin types IV-VI treated with the sublative laser, early results found improved photo damage and acne scarring after treatment. There was little erythema after treatment, said Dr. Goldberg.
He stressed that the results are early, and "only time will tell" if this device will live up to its early hype.
Dr. Goldberg disclosed receiving research grants from Syneron. SDEF and this news organization are owned by Elsevier.
-Amy Pfeiffer
Sublation appears to be the new "it" word in skin laser rejuvenation. Studies have found that a device utilizing sublative technology was safe and effective for skin tightening and treating acne scars, even in darker skinned patients.
Dr. David J. Goldberg, director of dermatologic laser research at Mount Sinai School of Medicine, New York, presented early trial results at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
Besides being safe, effective, and offering minimal downtime, an immediate impact was seen after sublation through histopathology imaging. The small, fractionated, bipolar, radio-frequency device (Syneron eMatrix system) delivers energy deep into the dermis with limited skin surface involvement. Dr. Goldberg described the energy as entering an upside down cone. The energy spreads out as it goes deeper into the skin.
Compared with ablative resurfacing, sublative rejuvenation has the same healing physiological pathway (healing cascade) and the same initiating event (inflammation), said Dr. Goldberg. However, sublation has different perdominant events and there is a different proportion of dermal/epidermal involvement. Once again, said Dr. Goldberg, for delivering energy, think upside down cone, compared with the cone of ablation.
Initial studies at four sites in the United States and Canada of about 80 patients, with an average age of 52 and Fitzpatrick skin types I-IV, found that 83% of patients had improved skin brightness, 87% had improved skin tightness, and 90% had wrinkle improvement. Full data will be presented in April at the American Society for Laser Medicine and Surgery meeting in San Antonio.
In another study of patients with Fitzpatrick skin types IV-VI treated with the sublative laser, early results found improved photo damage and acne scarring after treatment. There was little erythema after treatment, said Dr. Goldberg.
He stressed that the results are early, and "only time will tell" if this device will live up to its early hype.
Dr. Goldberg disclosed receiving research grants from Syneron. SDEF and this news organization are owned by Elsevier.
-Amy Pfeiffer
SDEF: Fractional Lasers a "Go To" Device for Scar Treatment
Partially or fully ablative lasers are safe and effective tools to treat many types of scars and keloids, Dr. Suzanne L. Kilmer reported.
In treating scars, "the challenge is to normalize the color and the texture and to somehow repopulate that area with normal collagen and return it to normal skin color for that individual," she said in an interview.
A key challenge to treatment is managing patient expectations. She described three treatment techniques to improve the appearance of scars and keloids at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF): pulsed dye lasers, fractional lasers, and fully ablative erbium/CO2 laser resurfacing.
Each of these lasers has its pros and cons for scar treatment, Dr. Kilmer explained. "In most cases now I will go straight to a fractional device, and depending on location, patient's tolerance for downtime, pain, and budget constraints, I will proceed with the best choice."
Pulsed-dye lasers help improve erythema by targeting hemoglobin, and they can also aid in collagen remodeling, reported Dr. Kilmer of the Laser & Skin Surgery Center of Northern California, Sacramento.
Fractional lasers heat up or even ablate thousands of tiny 120-mcm columns of tissue, allowing tissue remodeling and the regeneration of more normal collagen. This normal collagen gradually takes over the area with scar tissue and helps to return the skin to its prescar appearance. This is also helpful for striae (stretch marks), she noted.
Fractional resurfacing is often effective for pigment, texture, or vascular changes. The fractional laser produces a microthermal zone up to 1,500 mcm deep that creates lateral reepithelialization and promotes skin healing. A Fraxel 1550-nm erbium laser (Solta) or a StarLux 1540-nm laser (Palomar) is a good choice for these procedures.
Most patients find the Fraxel 1550-nm laser more painful, and anesthesia is needed for the entire procedure, she reported. However, the Fraxel is faster, so it is a better choice for procedures involving the full face, and provides a more even treatment.
By contrast, the StarLux 1,540-nm laser often allows the clinician to treat small areas without the need for anesthesia, and these areas blend easily into nontreated skin, Dr. Kilmer pointed out. Small areas of the skin can be treated quickly, but the StarLux can be slow and uneven when used on larger areas.
Swelling is common – but not usually painful – after nonablative fractional resurfacing, she explained, and most patients need only minimal aftercare. Usually ice or hydration is sufficient.
Fractional ablative laser resurfacing carries more risk and requires more downtime than do other scar treatments, but it offers "the greatest efficacy for a single treatment," noted Dr. Kilmer. This treatment allows the clinician to sculpt more deeply with less thermal damage, and provides the most predictable results, including skin tightening and smoothing of acne scar ridges. The fractional ablative component allows for deeper scar remodeling and can be done at the same time, but with caution, she noted.
Dr. Kilmer emphasized that clinicians who are new to laser scar treatments should go slowly while they become familiar with the device. "And go lighter when you are off the face," she added.
Dr. Kilmer has received research support from Palomar and Solta. SDEF and this news organization are owned by Elsevier.
Partially or fully ablative lasers are safe and effective tools to treat many types of scars and keloids, Dr. Suzanne L. Kilmer reported.
In treating scars, "the challenge is to normalize the color and the texture and to somehow repopulate that area with normal collagen and return it to normal skin color for that individual," she said in an interview.
A key challenge to treatment is managing patient expectations. She described three treatment techniques to improve the appearance of scars and keloids at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF): pulsed dye lasers, fractional lasers, and fully ablative erbium/CO2 laser resurfacing.
Each of these lasers has its pros and cons for scar treatment, Dr. Kilmer explained. "In most cases now I will go straight to a fractional device, and depending on location, patient's tolerance for downtime, pain, and budget constraints, I will proceed with the best choice."
Pulsed-dye lasers help improve erythema by targeting hemoglobin, and they can also aid in collagen remodeling, reported Dr. Kilmer of the Laser & Skin Surgery Center of Northern California, Sacramento.
Fractional lasers heat up or even ablate thousands of tiny 120-mcm columns of tissue, allowing tissue remodeling and the regeneration of more normal collagen. This normal collagen gradually takes over the area with scar tissue and helps to return the skin to its prescar appearance. This is also helpful for striae (stretch marks), she noted.
Fractional resurfacing is often effective for pigment, texture, or vascular changes. The fractional laser produces a microthermal zone up to 1,500 mcm deep that creates lateral reepithelialization and promotes skin healing. A Fraxel 1550-nm erbium laser (Solta) or a StarLux 1540-nm laser (Palomar) is a good choice for these procedures.
Most patients find the Fraxel 1550-nm laser more painful, and anesthesia is needed for the entire procedure, she reported. However, the Fraxel is faster, so it is a better choice for procedures involving the full face, and provides a more even treatment.
By contrast, the StarLux 1,540-nm laser often allows the clinician to treat small areas without the need for anesthesia, and these areas blend easily into nontreated skin, Dr. Kilmer pointed out. Small areas of the skin can be treated quickly, but the StarLux can be slow and uneven when used on larger areas.
Swelling is common – but not usually painful – after nonablative fractional resurfacing, she explained, and most patients need only minimal aftercare. Usually ice or hydration is sufficient.
Fractional ablative laser resurfacing carries more risk and requires more downtime than do other scar treatments, but it offers "the greatest efficacy for a single treatment," noted Dr. Kilmer. This treatment allows the clinician to sculpt more deeply with less thermal damage, and provides the most predictable results, including skin tightening and smoothing of acne scar ridges. The fractional ablative component allows for deeper scar remodeling and can be done at the same time, but with caution, she noted.
Dr. Kilmer emphasized that clinicians who are new to laser scar treatments should go slowly while they become familiar with the device. "And go lighter when you are off the face," she added.
Dr. Kilmer has received research support from Palomar and Solta. SDEF and this news organization are owned by Elsevier.
Partially or fully ablative lasers are safe and effective tools to treat many types of scars and keloids, Dr. Suzanne L. Kilmer reported.
In treating scars, "the challenge is to normalize the color and the texture and to somehow repopulate that area with normal collagen and return it to normal skin color for that individual," she said in an interview.
A key challenge to treatment is managing patient expectations. She described three treatment techniques to improve the appearance of scars and keloids at the Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF): pulsed dye lasers, fractional lasers, and fully ablative erbium/CO2 laser resurfacing.
Each of these lasers has its pros and cons for scar treatment, Dr. Kilmer explained. "In most cases now I will go straight to a fractional device, and depending on location, patient's tolerance for downtime, pain, and budget constraints, I will proceed with the best choice."
Pulsed-dye lasers help improve erythema by targeting hemoglobin, and they can also aid in collagen remodeling, reported Dr. Kilmer of the Laser & Skin Surgery Center of Northern California, Sacramento.
Fractional lasers heat up or even ablate thousands of tiny 120-mcm columns of tissue, allowing tissue remodeling and the regeneration of more normal collagen. This normal collagen gradually takes over the area with scar tissue and helps to return the skin to its prescar appearance. This is also helpful for striae (stretch marks), she noted.
Fractional resurfacing is often effective for pigment, texture, or vascular changes. The fractional laser produces a microthermal zone up to 1,500 mcm deep that creates lateral reepithelialization and promotes skin healing. A Fraxel 1550-nm erbium laser (Solta) or a StarLux 1540-nm laser (Palomar) is a good choice for these procedures.
Most patients find the Fraxel 1550-nm laser more painful, and anesthesia is needed for the entire procedure, she reported. However, the Fraxel is faster, so it is a better choice for procedures involving the full face, and provides a more even treatment.
By contrast, the StarLux 1,540-nm laser often allows the clinician to treat small areas without the need for anesthesia, and these areas blend easily into nontreated skin, Dr. Kilmer pointed out. Small areas of the skin can be treated quickly, but the StarLux can be slow and uneven when used on larger areas.
Swelling is common – but not usually painful – after nonablative fractional resurfacing, she explained, and most patients need only minimal aftercare. Usually ice or hydration is sufficient.
Fractional ablative laser resurfacing carries more risk and requires more downtime than do other scar treatments, but it offers "the greatest efficacy for a single treatment," noted Dr. Kilmer. This treatment allows the clinician to sculpt more deeply with less thermal damage, and provides the most predictable results, including skin tightening and smoothing of acne scar ridges. The fractional ablative component allows for deeper scar remodeling and can be done at the same time, but with caution, she noted.
Dr. Kilmer emphasized that clinicians who are new to laser scar treatments should go slowly while they become familiar with the device. "And go lighter when you are off the face," she added.
Dr. Kilmer has received research support from Palomar and Solta. SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Foam Sclerotherapy Appears as Effective as Surgery for Varicose Veins
SAN DIEGO – At 5 years of follow-up, patients who had ultrasound-guided foam sclerotherapy combined with saphenofemoral ligation had equally good clinical results compared with patients who underwent surgical treatment of varicose veins, based on a randomized controlled trial.
"Since surgery may not provide a definitive treatment, foam sclerotherapy could be offered like a dental care treatment model: treating as and when the problem appears," Dr. Evi Kalodiki said at the annual meeting of the American Venous Forum.
Dr. Kalodiki, a vascular surgeon at Ealing Hospital and Imperial College, London, and her associates evaluated 82 legs that were treated in 73 patients. The mean age of the patients was 48 years, and 75% were female. The researchers randomized the cases to two treatments: 39 legs underwent saphenofemoral ligation, stripping, and multiple phlebectomies under general anesthesia (surgery group) and 43 underwent saphenofemoral ligation under local anesthesia with concurrent foam sclerotherapy (foam group).
Assessments included ultrasound, the CEAP (clinical, etiologic, anatomical, pathophysiologic) classification, the Venous Clinical Severity Score, the Aberdeen Varicose Veins Questionnaire (AVVQ), and the Short Form 36 Health Survey (SF-36). Follow-ups were conducted annually until a median of 5.12 years.
Dr. Kalodiki reported that the CEAP classification was similar for the two groups, as was the percentage of legs that required additional foam sessions (40% of legs in the surgery group, with a mean volume of 11 mL for each case, vs. 48% of legs in the foam group, with a mean volume of 9 mL for each case).
Preoperative Venous Clinical Severity Scores (VCSS) were similar for the two groups (median of 5 in the surgery group vs. 4 in the foam group), as were post-treatment VCSS, at a median of 1 in each group. Median changes in VCSS from baseline to 5 years were similar in the two groups: 3 in the surgery group vs. 3.5 in the foam group. Absolute VCSS values at 5 years were a median of 1 in each group.
The AVVQ score also improved in both groups. In the surgery group, the median AVVQ score improved from 16.32 preoperatively to 8.94 at 3 years, while in the foam group the median AVVQ score improved from 12.28 preoperatively to 4.97 at 3 years. These results were maintained at 5 years.
Scores on the mental component of the SF-36 worsened in the surgery group. There was no change in the physical scores of the SF-36 in either group over 3 or 5 years.
There were no significant differences between the groups or within the groups regarding obliteration or reflux above the knee or below the knee at 3 or 5 years. In the majority of cases, the reflux was asymptomatic and was only detected because of the follow-up duplex examination.
The study was funded by STD Pharmaceutical. Dr. Kalodiki said that she had no relevant financial disclosures to make.
SAN DIEGO – At 5 years of follow-up, patients who had ultrasound-guided foam sclerotherapy combined with saphenofemoral ligation had equally good clinical results compared with patients who underwent surgical treatment of varicose veins, based on a randomized controlled trial.
"Since surgery may not provide a definitive treatment, foam sclerotherapy could be offered like a dental care treatment model: treating as and when the problem appears," Dr. Evi Kalodiki said at the annual meeting of the American Venous Forum.
Dr. Kalodiki, a vascular surgeon at Ealing Hospital and Imperial College, London, and her associates evaluated 82 legs that were treated in 73 patients. The mean age of the patients was 48 years, and 75% were female. The researchers randomized the cases to two treatments: 39 legs underwent saphenofemoral ligation, stripping, and multiple phlebectomies under general anesthesia (surgery group) and 43 underwent saphenofemoral ligation under local anesthesia with concurrent foam sclerotherapy (foam group).
Assessments included ultrasound, the CEAP (clinical, etiologic, anatomical, pathophysiologic) classification, the Venous Clinical Severity Score, the Aberdeen Varicose Veins Questionnaire (AVVQ), and the Short Form 36 Health Survey (SF-36). Follow-ups were conducted annually until a median of 5.12 years.
Dr. Kalodiki reported that the CEAP classification was similar for the two groups, as was the percentage of legs that required additional foam sessions (40% of legs in the surgery group, with a mean volume of 11 mL for each case, vs. 48% of legs in the foam group, with a mean volume of 9 mL for each case).
Preoperative Venous Clinical Severity Scores (VCSS) were similar for the two groups (median of 5 in the surgery group vs. 4 in the foam group), as were post-treatment VCSS, at a median of 1 in each group. Median changes in VCSS from baseline to 5 years were similar in the two groups: 3 in the surgery group vs. 3.5 in the foam group. Absolute VCSS values at 5 years were a median of 1 in each group.
The AVVQ score also improved in both groups. In the surgery group, the median AVVQ score improved from 16.32 preoperatively to 8.94 at 3 years, while in the foam group the median AVVQ score improved from 12.28 preoperatively to 4.97 at 3 years. These results were maintained at 5 years.
Scores on the mental component of the SF-36 worsened in the surgery group. There was no change in the physical scores of the SF-36 in either group over 3 or 5 years.
There were no significant differences between the groups or within the groups regarding obliteration or reflux above the knee or below the knee at 3 or 5 years. In the majority of cases, the reflux was asymptomatic and was only detected because of the follow-up duplex examination.
The study was funded by STD Pharmaceutical. Dr. Kalodiki said that she had no relevant financial disclosures to make.
SAN DIEGO – At 5 years of follow-up, patients who had ultrasound-guided foam sclerotherapy combined with saphenofemoral ligation had equally good clinical results compared with patients who underwent surgical treatment of varicose veins, based on a randomized controlled trial.
"Since surgery may not provide a definitive treatment, foam sclerotherapy could be offered like a dental care treatment model: treating as and when the problem appears," Dr. Evi Kalodiki said at the annual meeting of the American Venous Forum.
Dr. Kalodiki, a vascular surgeon at Ealing Hospital and Imperial College, London, and her associates evaluated 82 legs that were treated in 73 patients. The mean age of the patients was 48 years, and 75% were female. The researchers randomized the cases to two treatments: 39 legs underwent saphenofemoral ligation, stripping, and multiple phlebectomies under general anesthesia (surgery group) and 43 underwent saphenofemoral ligation under local anesthesia with concurrent foam sclerotherapy (foam group).
Assessments included ultrasound, the CEAP (clinical, etiologic, anatomical, pathophysiologic) classification, the Venous Clinical Severity Score, the Aberdeen Varicose Veins Questionnaire (AVVQ), and the Short Form 36 Health Survey (SF-36). Follow-ups were conducted annually until a median of 5.12 years.
Dr. Kalodiki reported that the CEAP classification was similar for the two groups, as was the percentage of legs that required additional foam sessions (40% of legs in the surgery group, with a mean volume of 11 mL for each case, vs. 48% of legs in the foam group, with a mean volume of 9 mL for each case).
Preoperative Venous Clinical Severity Scores (VCSS) were similar for the two groups (median of 5 in the surgery group vs. 4 in the foam group), as were post-treatment VCSS, at a median of 1 in each group. Median changes in VCSS from baseline to 5 years were similar in the two groups: 3 in the surgery group vs. 3.5 in the foam group. Absolute VCSS values at 5 years were a median of 1 in each group.
The AVVQ score also improved in both groups. In the surgery group, the median AVVQ score improved from 16.32 preoperatively to 8.94 at 3 years, while in the foam group the median AVVQ score improved from 12.28 preoperatively to 4.97 at 3 years. These results were maintained at 5 years.
Scores on the mental component of the SF-36 worsened in the surgery group. There was no change in the physical scores of the SF-36 in either group over 3 or 5 years.
There were no significant differences between the groups or within the groups regarding obliteration or reflux above the knee or below the knee at 3 or 5 years. In the majority of cases, the reflux was asymptomatic and was only detected because of the follow-up duplex examination.
The study was funded by STD Pharmaceutical. Dr. Kalodiki said that she had no relevant financial disclosures to make.
FROM THE ANNUAL MEETING OF THE AMERICAN VENOUS FORUM
Major Finding: Improvements in venous clinical severity scores at 3 and 5 years were similar among patients who underwent saphenofemoral ligation, stripping, and multiple phlebectomies under general anesthesia and those who underwent saphenofemoral ligation under local anesthesia with concurrent foam sclerotherapy.
Data Source: A randomized, controlled trial of 82 legs that were treated in 73 patients with a mean age of 48 years.
Disclosures: The study was funded by STD Pharmaceutical. Dr. Kalodiki said that she had no relevant financial disclosures to make.
Two Sclerotherapy Methods Yield Similar 6-Month Outcomes
SAN DIEGO – Foam sclerotherapy alone was just as effective as ultrasound-guided foam sclerotherapy for the treatment of great saphenous vein reflux, results from a single-center study showed.
Dr. Takashi Yamaki, a vascular surgeon, and his associates in the department of plastic and reconstructive surgery at Tokyo Women's Medical University conducted a randomized study between January and July 2010 in which 51 limbs in 48 patients were treated with ultrasound-guided foam sclerotherapy and 52 limbs in 49 patients were treated with foam sclerotherapy alone.
Patients in the combined-treatment group received less than 0.5 mL of 1% polidocanol foam per injection to minimize any foam migration beyond the target vein, said Dr. Yamaki at the annual meeting of the American Venous Forum.
Patients in the foam-only treatment group received more than 0.5 mL of 1% polidocanol foam per injection, but the amount never exceeded 1.0 mL.
The researchers conducted ultrasonographic inspection of the foam in the greater saphenous vein for 5 minutes before compression was applied. They also conducted postsclerotherapy surveillance at 1, 3, and 6 months. The study's primary end point was elimination of greater saphenous vein reflux at 6 months.
The mean age of patients was 66 years, and 71% were female. There were no differences between the groups in terms of CEAP clinical classification (which is based on clinical severity, etiology or cause, anatomy, and pathophysiology) or venous clinical severity scores.
Dr. Yamaki reported that the mean greater saphenous vein diameter was 0.6 cm in the combined-treatment group and 0.57 cm in the foam-only treatment group, a difference that was not significant. The mean injected volume of foam for varicose tributary veins was significantly lower in the combined-treatment group than in the foam-only group (3.9 mL vs. 5.8 mL, respectively). However, the mean total amount of injected foam was significantly higher in the combined-treatment group than in the foam-only group (6.5 mL vs. 5.8 mL).
Ultrasonic evaluation revealed complete spasm of the greater saphenous vein in 73% of limbs that underwent combined treatment, compared with 56% of limbs that underwent foam-only treatment, a difference that was not significant.
At 6 months the researchers observed absence of reflux in 59% of limbs that underwent combined treatment, compared with 71% of limbs that underwent foam-only treatment, a difference that was not significant.
No serious complications occurred in either group.
"Further studies are required in order to define the predictive factors for successful outcome, especially in patients with uncomplicated varicose veins, including factors such as foam production, the optimal volume of foam, the use of physiologic gases, injection techniques, and the optimal period of use of compression stockings," said Dr. Yamaki.
Zeria Pharmaceutical provided the 1% polidocanol used in the study. Dr. Yamaki said that he had no relevant financial disclosures.
SAN DIEGO – Foam sclerotherapy alone was just as effective as ultrasound-guided foam sclerotherapy for the treatment of great saphenous vein reflux, results from a single-center study showed.
Dr. Takashi Yamaki, a vascular surgeon, and his associates in the department of plastic and reconstructive surgery at Tokyo Women's Medical University conducted a randomized study between January and July 2010 in which 51 limbs in 48 patients were treated with ultrasound-guided foam sclerotherapy and 52 limbs in 49 patients were treated with foam sclerotherapy alone.
Patients in the combined-treatment group received less than 0.5 mL of 1% polidocanol foam per injection to minimize any foam migration beyond the target vein, said Dr. Yamaki at the annual meeting of the American Venous Forum.
Patients in the foam-only treatment group received more than 0.5 mL of 1% polidocanol foam per injection, but the amount never exceeded 1.0 mL.
The researchers conducted ultrasonographic inspection of the foam in the greater saphenous vein for 5 minutes before compression was applied. They also conducted postsclerotherapy surveillance at 1, 3, and 6 months. The study's primary end point was elimination of greater saphenous vein reflux at 6 months.
The mean age of patients was 66 years, and 71% were female. There were no differences between the groups in terms of CEAP clinical classification (which is based on clinical severity, etiology or cause, anatomy, and pathophysiology) or venous clinical severity scores.
Dr. Yamaki reported that the mean greater saphenous vein diameter was 0.6 cm in the combined-treatment group and 0.57 cm in the foam-only treatment group, a difference that was not significant. The mean injected volume of foam for varicose tributary veins was significantly lower in the combined-treatment group than in the foam-only group (3.9 mL vs. 5.8 mL, respectively). However, the mean total amount of injected foam was significantly higher in the combined-treatment group than in the foam-only group (6.5 mL vs. 5.8 mL).
Ultrasonic evaluation revealed complete spasm of the greater saphenous vein in 73% of limbs that underwent combined treatment, compared with 56% of limbs that underwent foam-only treatment, a difference that was not significant.
At 6 months the researchers observed absence of reflux in 59% of limbs that underwent combined treatment, compared with 71% of limbs that underwent foam-only treatment, a difference that was not significant.
No serious complications occurred in either group.
"Further studies are required in order to define the predictive factors for successful outcome, especially in patients with uncomplicated varicose veins, including factors such as foam production, the optimal volume of foam, the use of physiologic gases, injection techniques, and the optimal period of use of compression stockings," said Dr. Yamaki.
Zeria Pharmaceutical provided the 1% polidocanol used in the study. Dr. Yamaki said that he had no relevant financial disclosures.
SAN DIEGO – Foam sclerotherapy alone was just as effective as ultrasound-guided foam sclerotherapy for the treatment of great saphenous vein reflux, results from a single-center study showed.
Dr. Takashi Yamaki, a vascular surgeon, and his associates in the department of plastic and reconstructive surgery at Tokyo Women's Medical University conducted a randomized study between January and July 2010 in which 51 limbs in 48 patients were treated with ultrasound-guided foam sclerotherapy and 52 limbs in 49 patients were treated with foam sclerotherapy alone.
Patients in the combined-treatment group received less than 0.5 mL of 1% polidocanol foam per injection to minimize any foam migration beyond the target vein, said Dr. Yamaki at the annual meeting of the American Venous Forum.
Patients in the foam-only treatment group received more than 0.5 mL of 1% polidocanol foam per injection, but the amount never exceeded 1.0 mL.
The researchers conducted ultrasonographic inspection of the foam in the greater saphenous vein for 5 minutes before compression was applied. They also conducted postsclerotherapy surveillance at 1, 3, and 6 months. The study's primary end point was elimination of greater saphenous vein reflux at 6 months.
The mean age of patients was 66 years, and 71% were female. There were no differences between the groups in terms of CEAP clinical classification (which is based on clinical severity, etiology or cause, anatomy, and pathophysiology) or venous clinical severity scores.
Dr. Yamaki reported that the mean greater saphenous vein diameter was 0.6 cm in the combined-treatment group and 0.57 cm in the foam-only treatment group, a difference that was not significant. The mean injected volume of foam for varicose tributary veins was significantly lower in the combined-treatment group than in the foam-only group (3.9 mL vs. 5.8 mL, respectively). However, the mean total amount of injected foam was significantly higher in the combined-treatment group than in the foam-only group (6.5 mL vs. 5.8 mL).
Ultrasonic evaluation revealed complete spasm of the greater saphenous vein in 73% of limbs that underwent combined treatment, compared with 56% of limbs that underwent foam-only treatment, a difference that was not significant.
At 6 months the researchers observed absence of reflux in 59% of limbs that underwent combined treatment, compared with 71% of limbs that underwent foam-only treatment, a difference that was not significant.
No serious complications occurred in either group.
"Further studies are required in order to define the predictive factors for successful outcome, especially in patients with uncomplicated varicose veins, including factors such as foam production, the optimal volume of foam, the use of physiologic gases, injection techniques, and the optimal period of use of compression stockings," said Dr. Yamaki.
Zeria Pharmaceutical provided the 1% polidocanol used in the study. Dr. Yamaki said that he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN VENOUS FORUM
Major Finding: At 6 months, absence of greater saphenous vein reflux was observed in 59% of limbs that underwent ultrasound-guided foam sclerotherapy, compared with 71% of limbs that underwent foam sclerotherapy only, a difference that was not significant.
Data Source: A single-center study of 97 patients.
Disclosures: Zeria Pharmaceutical provided the 1% polidocanol used in the study. Dr. Yamaki said that he had no relevant financial disclosures.
Voluma Facial Filler Expected to Hit Market This Year
MIAMI BEACH – You may soon be able to "pump up the volume" with a new facial filler, according to Dr. Nowell Solish.
Dr. Solish reported his experience with a low-molecular-weight, hyaluronic acid filler at the South Beach Symposium.
Allergan Inc.’s Voluma is already available in Canada and is expected to be available in the United States this year. This viscous formulation of the Juvéderm product line "is really for deeper injections and really for facial volume."
Appropriate use includes injections of the cheek, submalar region, and chin. "This is really for volume replacement in patients whose faces have become ‘bottom heavy,’ " said Dr. Solish of the division of dermatology at the University of Toronto.
"I use this in the midface mostly. It all depends on the age of the patient," he said. He tends to inject more filler medially for older patients and more laterally on the face for his younger patients.
Dr. Solish recommended injection planning using markings to ensure symmetrical placement of the filler. For example, he explained how a triangle on the midface created by the intersection of three straight lines could be used as a guide. One line would go from the lateral canthus to the corner of the patient’s mouth, the second from the upper part of the tragus to the alar lobule, and the third from the lateral canthus to the lower tragus.
Subcutaneous injections are made to the deep dermis and above the periosteum. Do not overcorrect your patient and always massage the area post injection to ensure even distribution, he said.
Voluma is expected to contain lidocaine when it is marketed in the United States. "I mix this filler with lidocaine. Voluma does not come with lidocaine in Canada," Dr. Solish said. He said that the addition of lidocaine with epinephrine makes the filler less viscous and easier to inject through a 28-gauge needle.
Results can last up to a year or longer. His patients typically return for subsequent treatment in 9-12 months. "We are finding they need about half as much product as their initial treatment," he said.
Dr. Solish reported side effects similar to other hyaluronic acid fillers, including bruising, swelling, and discomfort.
"But some patients have a foreign body–like reaction that requires draining and hyaluronidase," Dr. Solish said. "We’ve seen a few cases in Canada."
The low-molecular-weight hyaluronic acid is composed of 90% short molecular chains and 10% of the chains "you are used to with regular Juvéderm," Dr. Solish said.
The term "molecular weight" refers to the length of the molecular chains (number of repeating chains). Greater cross-linking with these shorter chains increases the product’s viscosity.
Dr. Solish disclosed being a consultant and researcher for Allergan.
MIAMI BEACH – You may soon be able to "pump up the volume" with a new facial filler, according to Dr. Nowell Solish.
Dr. Solish reported his experience with a low-molecular-weight, hyaluronic acid filler at the South Beach Symposium.
Allergan Inc.’s Voluma is already available in Canada and is expected to be available in the United States this year. This viscous formulation of the Juvéderm product line "is really for deeper injections and really for facial volume."
Appropriate use includes injections of the cheek, submalar region, and chin. "This is really for volume replacement in patients whose faces have become ‘bottom heavy,’ " said Dr. Solish of the division of dermatology at the University of Toronto.
"I use this in the midface mostly. It all depends on the age of the patient," he said. He tends to inject more filler medially for older patients and more laterally on the face for his younger patients.
Dr. Solish recommended injection planning using markings to ensure symmetrical placement of the filler. For example, he explained how a triangle on the midface created by the intersection of three straight lines could be used as a guide. One line would go from the lateral canthus to the corner of the patient’s mouth, the second from the upper part of the tragus to the alar lobule, and the third from the lateral canthus to the lower tragus.
Subcutaneous injections are made to the deep dermis and above the periosteum. Do not overcorrect your patient and always massage the area post injection to ensure even distribution, he said.
Voluma is expected to contain lidocaine when it is marketed in the United States. "I mix this filler with lidocaine. Voluma does not come with lidocaine in Canada," Dr. Solish said. He said that the addition of lidocaine with epinephrine makes the filler less viscous and easier to inject through a 28-gauge needle.
Results can last up to a year or longer. His patients typically return for subsequent treatment in 9-12 months. "We are finding they need about half as much product as their initial treatment," he said.
Dr. Solish reported side effects similar to other hyaluronic acid fillers, including bruising, swelling, and discomfort.
"But some patients have a foreign body–like reaction that requires draining and hyaluronidase," Dr. Solish said. "We’ve seen a few cases in Canada."
The low-molecular-weight hyaluronic acid is composed of 90% short molecular chains and 10% of the chains "you are used to with regular Juvéderm," Dr. Solish said.
The term "molecular weight" refers to the length of the molecular chains (number of repeating chains). Greater cross-linking with these shorter chains increases the product’s viscosity.
Dr. Solish disclosed being a consultant and researcher for Allergan.
MIAMI BEACH – You may soon be able to "pump up the volume" with a new facial filler, according to Dr. Nowell Solish.
Dr. Solish reported his experience with a low-molecular-weight, hyaluronic acid filler at the South Beach Symposium.
Allergan Inc.’s Voluma is already available in Canada and is expected to be available in the United States this year. This viscous formulation of the Juvéderm product line "is really for deeper injections and really for facial volume."
Appropriate use includes injections of the cheek, submalar region, and chin. "This is really for volume replacement in patients whose faces have become ‘bottom heavy,’ " said Dr. Solish of the division of dermatology at the University of Toronto.
"I use this in the midface mostly. It all depends on the age of the patient," he said. He tends to inject more filler medially for older patients and more laterally on the face for his younger patients.
Dr. Solish recommended injection planning using markings to ensure symmetrical placement of the filler. For example, he explained how a triangle on the midface created by the intersection of three straight lines could be used as a guide. One line would go from the lateral canthus to the corner of the patient’s mouth, the second from the upper part of the tragus to the alar lobule, and the third from the lateral canthus to the lower tragus.
Subcutaneous injections are made to the deep dermis and above the periosteum. Do not overcorrect your patient and always massage the area post injection to ensure even distribution, he said.
Voluma is expected to contain lidocaine when it is marketed in the United States. "I mix this filler with lidocaine. Voluma does not come with lidocaine in Canada," Dr. Solish said. He said that the addition of lidocaine with epinephrine makes the filler less viscous and easier to inject through a 28-gauge needle.
Results can last up to a year or longer. His patients typically return for subsequent treatment in 9-12 months. "We are finding they need about half as much product as their initial treatment," he said.
Dr. Solish reported side effects similar to other hyaluronic acid fillers, including bruising, swelling, and discomfort.
"But some patients have a foreign body–like reaction that requires draining and hyaluronidase," Dr. Solish said. "We’ve seen a few cases in Canada."
The low-molecular-weight hyaluronic acid is composed of 90% short molecular chains and 10% of the chains "you are used to with regular Juvéderm," Dr. Solish said.
The term "molecular weight" refers to the length of the molecular chains (number of repeating chains). Greater cross-linking with these shorter chains increases the product’s viscosity.
Dr. Solish disclosed being a consultant and researcher for Allergan.
EXPERT ANALYSIS FROM THE SOUTH BEACH SYMPOSIUM
Cannulas Less Traumatic Than Needles for Administering Fillers
MIAMI BEACH – It's time to consider being blunt with your aesthetic patients, at least in terms of how you administer filler products for facial and hand rejuvenation.
"The blunt cannula is really an exciting tool for us," Dr. Susan H. Weinkle said at the South Beach Symposium.
Instead of multiple puncture wounds with needles, Dr. Weinkle creates one entry point with a percutaneous stick of a 26-gauge needle. Then she inserts a cannula. "You can treat the midface [and then] you can turn the cannula and treat all along the cheekbone and the zygomatic arch. Then, through the same injection point, you can turn the cannula south and treat the nasolabial fold."
Less trauma, lower risk for bruising, and quicker downtime are among the advantages, compared with multiple needle injections, Dr. Weinkle said.
Less precise delivery of the filler – because the cannula holes are not at the tip like a needle – is a drawback, but not a significant one, she added.
Cannulas already have gained popularity in Europe and South America. "We are not learning this as quickly as our colleagues," she said.
"A year ago, I went to an exciting meeting in Paris where I heard a little about cannulas." Dr. Weinkle brought some back to her private practice in Bradenton, Fla., with the best intentions, but did not use them. "This year I went back to the same meeting in January, and I decided I did not want to be left behind."
Filler augmentation of the dorsal side of the hands is another procedure that is well suited to the use of these blunt cannulas, Dr. Weinkle said.
Patience is advised when the technique is tried for the first time. "It's not always easy. You're not going to love it right away. You have to finesse the cannula through the skin." However, she added, "Don't get discouraged."
These are not cannulas that are used for liposuction, but newer products that are specifically designed for use in soft tissue. Only one such cannula – CosmoFrance Inc.'s DermaSculpt microcannula, a 1.5 inch, 27 gauge cannula with a nonbruising blunt tip – is currently approved by the Food and Drug Administration in the United States.
"More are coming. We will have shorter, fatter, thicker, thinner, and different gauge cannulas," Dr. Weinkle said. "We're going to see some of the companies that provide fillers for us adopting this. Maybe we'll find one needle and one cannula in the future in our packaging."
"This is the wave of the future. We really want to embrace new and exciting things," she said.
Dr. Weinkle said she had no relevant financial disclosures.
MIAMI BEACH – It's time to consider being blunt with your aesthetic patients, at least in terms of how you administer filler products for facial and hand rejuvenation.
"The blunt cannula is really an exciting tool for us," Dr. Susan H. Weinkle said at the South Beach Symposium.
Instead of multiple puncture wounds with needles, Dr. Weinkle creates one entry point with a percutaneous stick of a 26-gauge needle. Then she inserts a cannula. "You can treat the midface [and then] you can turn the cannula and treat all along the cheekbone and the zygomatic arch. Then, through the same injection point, you can turn the cannula south and treat the nasolabial fold."
Less trauma, lower risk for bruising, and quicker downtime are among the advantages, compared with multiple needle injections, Dr. Weinkle said.
Less precise delivery of the filler – because the cannula holes are not at the tip like a needle – is a drawback, but not a significant one, she added.
Cannulas already have gained popularity in Europe and South America. "We are not learning this as quickly as our colleagues," she said.
"A year ago, I went to an exciting meeting in Paris where I heard a little about cannulas." Dr. Weinkle brought some back to her private practice in Bradenton, Fla., with the best intentions, but did not use them. "This year I went back to the same meeting in January, and I decided I did not want to be left behind."
Filler augmentation of the dorsal side of the hands is another procedure that is well suited to the use of these blunt cannulas, Dr. Weinkle said.
Patience is advised when the technique is tried for the first time. "It's not always easy. You're not going to love it right away. You have to finesse the cannula through the skin." However, she added, "Don't get discouraged."
These are not cannulas that are used for liposuction, but newer products that are specifically designed for use in soft tissue. Only one such cannula – CosmoFrance Inc.'s DermaSculpt microcannula, a 1.5 inch, 27 gauge cannula with a nonbruising blunt tip – is currently approved by the Food and Drug Administration in the United States.
"More are coming. We will have shorter, fatter, thicker, thinner, and different gauge cannulas," Dr. Weinkle said. "We're going to see some of the companies that provide fillers for us adopting this. Maybe we'll find one needle and one cannula in the future in our packaging."
"This is the wave of the future. We really want to embrace new and exciting things," she said.
Dr. Weinkle said she had no relevant financial disclosures.
MIAMI BEACH – It's time to consider being blunt with your aesthetic patients, at least in terms of how you administer filler products for facial and hand rejuvenation.
"The blunt cannula is really an exciting tool for us," Dr. Susan H. Weinkle said at the South Beach Symposium.
Instead of multiple puncture wounds with needles, Dr. Weinkle creates one entry point with a percutaneous stick of a 26-gauge needle. Then she inserts a cannula. "You can treat the midface [and then] you can turn the cannula and treat all along the cheekbone and the zygomatic arch. Then, through the same injection point, you can turn the cannula south and treat the nasolabial fold."
Less trauma, lower risk for bruising, and quicker downtime are among the advantages, compared with multiple needle injections, Dr. Weinkle said.
Less precise delivery of the filler – because the cannula holes are not at the tip like a needle – is a drawback, but not a significant one, she added.
Cannulas already have gained popularity in Europe and South America. "We are not learning this as quickly as our colleagues," she said.
"A year ago, I went to an exciting meeting in Paris where I heard a little about cannulas." Dr. Weinkle brought some back to her private practice in Bradenton, Fla., with the best intentions, but did not use them. "This year I went back to the same meeting in January, and I decided I did not want to be left behind."
Filler augmentation of the dorsal side of the hands is another procedure that is well suited to the use of these blunt cannulas, Dr. Weinkle said.
Patience is advised when the technique is tried for the first time. "It's not always easy. You're not going to love it right away. You have to finesse the cannula through the skin." However, she added, "Don't get discouraged."
These are not cannulas that are used for liposuction, but newer products that are specifically designed for use in soft tissue. Only one such cannula – CosmoFrance Inc.'s DermaSculpt microcannula, a 1.5 inch, 27 gauge cannula with a nonbruising blunt tip – is currently approved by the Food and Drug Administration in the United States.
"More are coming. We will have shorter, fatter, thicker, thinner, and different gauge cannulas," Dr. Weinkle said. "We're going to see some of the companies that provide fillers for us adopting this. Maybe we'll find one needle and one cannula in the future in our packaging."
"This is the wave of the future. We really want to embrace new and exciting things," she said.
Dr. Weinkle said she had no relevant financial disclosures.
EXPERT ANALYSIS FROM THE SOUTH BEACH SYMPOSIUM
Plastic Surgery Groups Remove Breast Implant Webinar
Two plastic surgery professional organizations have removed a members-only webinar in the wake of complaints by an advocacy group that the program downplayed the risk of anaplastic large-cell lymphoma in women who have breast implants.
Public Citizens Health Research Group wrote to the Food and Drug Administration on Feb. 17 to urge the agency to take action against the American Society of Plastic Surgeons (ASPS) and the American Society of Aesthetic Plastic Surgery (ASAPS).
The Washington-based nonprofit said that the groups held the webinar in the wake of the FDA's Jan. 26 announcement that there were a growing number of cases of anaplastic large-cell lymphoma (ALCL) in women with implants.
Dr. Jeffrey Shuren, director of the FDA's Center for Devices and Radiological Health, said in a Feb. 28 letter to Public Citizen that it had viewed the webinar and "spoke with representatives of both organizations." Dr. Shuren added that, "They informed us of their plans to remove the webinar from their Web site."
Both organizations said that they were not instructed by the FDA to take the webinar down, but that it was a voluntary decision.
In a March 2 statement, the ASAPS said that it removed the webinar "as newer information became available a week ago." That information, according to ASAPS president Felmont F. Eaves III, is "an independent, systematic review of ALCL, which will be published in an upcoming edition of Plastic and Reconstructive Surgery."
Dr. Eaves said in an interview that the Rand Corp. conducted the review and that it is his understanding that the article will be available some time in June. For the time being, the advanced copy of the article is available only to ASAPS members.
The ASPS said in a March 2 statement, "It was never our intention to downplay the risk of a very rarely occurring cancer associated with breast implants." Rather, said the Society, "We did not want to unnecessarily alarm patients when the risk of ALCL associated with breast implants is so low."
According to Public Citizen, ASPS president Phil Haeck explained in the webinar that ALCL should not be referred to as a tumor or a malignancy, but as a "condition." Dr. Haeck said, "I would recommend that you use the same terms with your patients rather than disturb them by saying this is a cancer, this is a malignancy. The best word is this is a condition," according to Public Citizen. Dr. Haeck added, "And I think you are certainly justified, with what we know now, in downplaying the malignant potential of these."
Public Citizen also objected to the webinar telling members that "surgery was curative," for ALCL.
In his response to Public Citizen, the FDA's Dr. Shuren said, "the FDA believes the optimal treatment regimen has not been established and that additional data collection is needed to fully understand the possible relationship between ALCL and breast implants, as well as the risk factors, optimal treatment plan, and prognosis."
The FDA is asking health care providers to report confirmed cases of ALCL. The agency also notes that ASPS and others are collaborating with the agency to develop a registry tracking ALCL and implants. ASAPS said in late January that it also is supporting the registry. Details are still being worked out.
Two plastic surgery professional organizations have removed a members-only webinar in the wake of complaints by an advocacy group that the program downplayed the risk of anaplastic large-cell lymphoma in women who have breast implants.
Public Citizens Health Research Group wrote to the Food and Drug Administration on Feb. 17 to urge the agency to take action against the American Society of Plastic Surgeons (ASPS) and the American Society of Aesthetic Plastic Surgery (ASAPS).
The Washington-based nonprofit said that the groups held the webinar in the wake of the FDA's Jan. 26 announcement that there were a growing number of cases of anaplastic large-cell lymphoma (ALCL) in women with implants.
Dr. Jeffrey Shuren, director of the FDA's Center for Devices and Radiological Health, said in a Feb. 28 letter to Public Citizen that it had viewed the webinar and "spoke with representatives of both organizations." Dr. Shuren added that, "They informed us of their plans to remove the webinar from their Web site."
Both organizations said that they were not instructed by the FDA to take the webinar down, but that it was a voluntary decision.
In a March 2 statement, the ASAPS said that it removed the webinar "as newer information became available a week ago." That information, according to ASAPS president Felmont F. Eaves III, is "an independent, systematic review of ALCL, which will be published in an upcoming edition of Plastic and Reconstructive Surgery."
Dr. Eaves said in an interview that the Rand Corp. conducted the review and that it is his understanding that the article will be available some time in June. For the time being, the advanced copy of the article is available only to ASAPS members.
The ASPS said in a March 2 statement, "It was never our intention to downplay the risk of a very rarely occurring cancer associated with breast implants." Rather, said the Society, "We did not want to unnecessarily alarm patients when the risk of ALCL associated with breast implants is so low."
According to Public Citizen, ASPS president Phil Haeck explained in the webinar that ALCL should not be referred to as a tumor or a malignancy, but as a "condition." Dr. Haeck said, "I would recommend that you use the same terms with your patients rather than disturb them by saying this is a cancer, this is a malignancy. The best word is this is a condition," according to Public Citizen. Dr. Haeck added, "And I think you are certainly justified, with what we know now, in downplaying the malignant potential of these."
Public Citizen also objected to the webinar telling members that "surgery was curative," for ALCL.
In his response to Public Citizen, the FDA's Dr. Shuren said, "the FDA believes the optimal treatment regimen has not been established and that additional data collection is needed to fully understand the possible relationship between ALCL and breast implants, as well as the risk factors, optimal treatment plan, and prognosis."
The FDA is asking health care providers to report confirmed cases of ALCL. The agency also notes that ASPS and others are collaborating with the agency to develop a registry tracking ALCL and implants. ASAPS said in late January that it also is supporting the registry. Details are still being worked out.
Two plastic surgery professional organizations have removed a members-only webinar in the wake of complaints by an advocacy group that the program downplayed the risk of anaplastic large-cell lymphoma in women who have breast implants.
Public Citizens Health Research Group wrote to the Food and Drug Administration on Feb. 17 to urge the agency to take action against the American Society of Plastic Surgeons (ASPS) and the American Society of Aesthetic Plastic Surgery (ASAPS).
The Washington-based nonprofit said that the groups held the webinar in the wake of the FDA's Jan. 26 announcement that there were a growing number of cases of anaplastic large-cell lymphoma (ALCL) in women with implants.
Dr. Jeffrey Shuren, director of the FDA's Center for Devices and Radiological Health, said in a Feb. 28 letter to Public Citizen that it had viewed the webinar and "spoke with representatives of both organizations." Dr. Shuren added that, "They informed us of their plans to remove the webinar from their Web site."
Both organizations said that they were not instructed by the FDA to take the webinar down, but that it was a voluntary decision.
In a March 2 statement, the ASAPS said that it removed the webinar "as newer information became available a week ago." That information, according to ASAPS president Felmont F. Eaves III, is "an independent, systematic review of ALCL, which will be published in an upcoming edition of Plastic and Reconstructive Surgery."
Dr. Eaves said in an interview that the Rand Corp. conducted the review and that it is his understanding that the article will be available some time in June. For the time being, the advanced copy of the article is available only to ASAPS members.
The ASPS said in a March 2 statement, "It was never our intention to downplay the risk of a very rarely occurring cancer associated with breast implants." Rather, said the Society, "We did not want to unnecessarily alarm patients when the risk of ALCL associated with breast implants is so low."
According to Public Citizen, ASPS president Phil Haeck explained in the webinar that ALCL should not be referred to as a tumor or a malignancy, but as a "condition." Dr. Haeck said, "I would recommend that you use the same terms with your patients rather than disturb them by saying this is a cancer, this is a malignancy. The best word is this is a condition," according to Public Citizen. Dr. Haeck added, "And I think you are certainly justified, with what we know now, in downplaying the malignant potential of these."
Public Citizen also objected to the webinar telling members that "surgery was curative," for ALCL.
In his response to Public Citizen, the FDA's Dr. Shuren said, "the FDA believes the optimal treatment regimen has not been established and that additional data collection is needed to fully understand the possible relationship between ALCL and breast implants, as well as the risk factors, optimal treatment plan, and prognosis."
The FDA is asking health care providers to report confirmed cases of ALCL. The agency also notes that ASPS and others are collaborating with the agency to develop a registry tracking ALCL and implants. ASAPS said in late January that it also is supporting the registry. Details are still being worked out.
Do Lasers or Topicals Really Work for Nonmelanoma Skin Cancers?
Lori Brightman, MD, Melanie Warycha, MD, Robert Anolik, MD, and Roy Geronemus, MD*
Novel strategies are urgently needed to address the millions of nonmelanoma skin cancers treated in the United States annually. The need is greatest for those patients who are poor surgical candidates or those prone to numerous nonmelanoma skin cancers and therefore at risk for marked disfigurement. Traditional treatment strategies include electrosurgery with curettage, radiation therapy, cryotherapy, excision, and Mohs micrographic surgery. Alternatives to traditional treatment, including topical medications and light or laser therapies, are becoming popular; however, there are various degrees of efficacy among these alternative tactics. These alternatives include topical retinoids, peels, 5-fluorouracil, imiquimod, photodynamic therapy, and lasers. The purpose of this paper is to review the available data regarding these alternative strategies and permit the reader to have a sense of which therapies are reasonable options for care.
*For a PDF of the full article, click on the link to the left of this introduction.
Lori Brightman, MD, Melanie Warycha, MD, Robert Anolik, MD, and Roy Geronemus, MD*
Novel strategies are urgently needed to address the millions of nonmelanoma skin cancers treated in the United States annually. The need is greatest for those patients who are poor surgical candidates or those prone to numerous nonmelanoma skin cancers and therefore at risk for marked disfigurement. Traditional treatment strategies include electrosurgery with curettage, radiation therapy, cryotherapy, excision, and Mohs micrographic surgery. Alternatives to traditional treatment, including topical medications and light or laser therapies, are becoming popular; however, there are various degrees of efficacy among these alternative tactics. These alternatives include topical retinoids, peels, 5-fluorouracil, imiquimod, photodynamic therapy, and lasers. The purpose of this paper is to review the available data regarding these alternative strategies and permit the reader to have a sense of which therapies are reasonable options for care.
*For a PDF of the full article, click on the link to the left of this introduction.
Lori Brightman, MD, Melanie Warycha, MD, Robert Anolik, MD, and Roy Geronemus, MD*
Novel strategies are urgently needed to address the millions of nonmelanoma skin cancers treated in the United States annually. The need is greatest for those patients who are poor surgical candidates or those prone to numerous nonmelanoma skin cancers and therefore at risk for marked disfigurement. Traditional treatment strategies include electrosurgery with curettage, radiation therapy, cryotherapy, excision, and Mohs micrographic surgery. Alternatives to traditional treatment, including topical medications and light or laser therapies, are becoming popular; however, there are various degrees of efficacy among these alternative tactics. These alternatives include topical retinoids, peels, 5-fluorouracil, imiquimod, photodynamic therapy, and lasers. The purpose of this paper is to review the available data regarding these alternative strategies and permit the reader to have a sense of which therapies are reasonable options for care.
*For a PDF of the full article, click on the link to the left of this introduction.