User login
Botulinum Toxin: Less Is More in Lower Face
ORLANDO – Dose is the most important consideration when injecting botulinum toxin in the lower face – even more important than during treatment of the upper face, according to Dr. Doris Hexsel.
"You should always use the lowest effective dose in the lower face," said Dr. Hexsel. This strategy reduces the risk for asymmetry, muscle dysfunction, and temporary oral paralysis. "These are dose-related and technique-related side effects."
Botulinum toxin can treat perioral wrinkles and marionette lines, as well as improve the appearance of a patient’s chin or gummy smile. However, only treat one or two areas in the lower face during the same session to minimize the risk of "sum of effect," she said. In other words, the effects of multiple, simultaneous injections around the mouth can be cumulative.
Also consider a combination of the botulinum toxin and filler injections, Dr. Hexsel said at the annual meeting of the Florida Society of Dermatologic Surgeons.
Dr. Hexsel shared her expertise with botulinum toxin for the following indications:
• Perioral wrinkles. Injections should be superficial and at least 1.5 cm from the corners of the mouth. Any closer and you increase the risk of undesirable relaxation of the depressor anguli oris muscle, the zygomaticus major muscle, and the risorius muscle, said Dr. Hexsel of the department of dermatology at Pontificia Universidade Católica do Rio Grande do Sul, Brazil.
She said that she typically injects 1.25 U-2.5 U abobotulinumtoxinA (Dysport, Ipsen/Medicis) per point, or 0.5 U-1.0 U onabotulinumtoxinA (Botox, Allergan). With two to six injection points, the total dose varies from 4 U-18 U for abobotulinum toxin or 4 U-10 U for onabotulinumtoxinA.
As a bonus, she said, "We observe a slight increase in the volume of the lips due to just the relaxation."
• "Cellulitic chin." Loss of collagen and subcutaneous fat, along with action of the jaw muscles, can cause a chin to have a cellulitic or "peau d’orange" appearance. Botulinum toxin can treat this area as well, said Dr. Hexsel. "I prefer two point injections – bilaterally at the most distal point of the mentalis muscle.
Again, keep the injections superficial and avoid high doses to minimize undesirable relaxation of the depressor labii inferioris muscle, she said. The total dose for a dimpled chin ranges from 15 U to 20 U of abobotulinumtoxinA and from 5 U to 10 U of onabotulinumtoxinA.
"I touch up patients 15 to 30 days later, if necessary," she added.
• Marionette lines. Botulinum toxin can improve the appearance of patients when the corners of their mouth appear permanently turned down. Better results may be obtained by combining toxin with fillers, she said.
For mild presentations, treat the mentalis muscle first, she advised. Treatment of this muscle also recruits the depressor anguli oris (DAO) muscle less (the mentalis is the agonist muscle to the DAO). Dr. Hexsel said she generally uses a total dose of 10 U-20 U of abobotulinumtoxinA or 3 U-6 U of onabotulinumtoxinA for this indication.
• Gummy smile. Consider botulinum toxin when a patient complains of a gummy smile. To foster a more natural look, also treat any natural asymmetries or posterior gummy smile, Dr. Hexsel said.
To treat posterior gingival exposure, inject two points on either side of the malar region. Inject in the nasolabial fold at the point of greatest lateral contraction during a smile.
Total doses vary from 5 U to 15 U of abobotulinumtoxinA or from 4 U to 10 U of onabotulinumtoxinA.
A final tip is to take before and after clinical photos, both during movement and at rest, when injecting the lower face.
Dr. Hexsel reported receiving grants and research support from Allergan, Galderma, and Ipsen. She also is a consultant for Allergan and Ipsen.
ORLANDO – Dose is the most important consideration when injecting botulinum toxin in the lower face – even more important than during treatment of the upper face, according to Dr. Doris Hexsel.
"You should always use the lowest effective dose in the lower face," said Dr. Hexsel. This strategy reduces the risk for asymmetry, muscle dysfunction, and temporary oral paralysis. "These are dose-related and technique-related side effects."
Botulinum toxin can treat perioral wrinkles and marionette lines, as well as improve the appearance of a patient’s chin or gummy smile. However, only treat one or two areas in the lower face during the same session to minimize the risk of "sum of effect," she said. In other words, the effects of multiple, simultaneous injections around the mouth can be cumulative.
Also consider a combination of the botulinum toxin and filler injections, Dr. Hexsel said at the annual meeting of the Florida Society of Dermatologic Surgeons.
Dr. Hexsel shared her expertise with botulinum toxin for the following indications:
• Perioral wrinkles. Injections should be superficial and at least 1.5 cm from the corners of the mouth. Any closer and you increase the risk of undesirable relaxation of the depressor anguli oris muscle, the zygomaticus major muscle, and the risorius muscle, said Dr. Hexsel of the department of dermatology at Pontificia Universidade Católica do Rio Grande do Sul, Brazil.
She said that she typically injects 1.25 U-2.5 U abobotulinumtoxinA (Dysport, Ipsen/Medicis) per point, or 0.5 U-1.0 U onabotulinumtoxinA (Botox, Allergan). With two to six injection points, the total dose varies from 4 U-18 U for abobotulinum toxin or 4 U-10 U for onabotulinumtoxinA.
As a bonus, she said, "We observe a slight increase in the volume of the lips due to just the relaxation."
• "Cellulitic chin." Loss of collagen and subcutaneous fat, along with action of the jaw muscles, can cause a chin to have a cellulitic or "peau d’orange" appearance. Botulinum toxin can treat this area as well, said Dr. Hexsel. "I prefer two point injections – bilaterally at the most distal point of the mentalis muscle.
Again, keep the injections superficial and avoid high doses to minimize undesirable relaxation of the depressor labii inferioris muscle, she said. The total dose for a dimpled chin ranges from 15 U to 20 U of abobotulinumtoxinA and from 5 U to 10 U of onabotulinumtoxinA.
"I touch up patients 15 to 30 days later, if necessary," she added.
• Marionette lines. Botulinum toxin can improve the appearance of patients when the corners of their mouth appear permanently turned down. Better results may be obtained by combining toxin with fillers, she said.
For mild presentations, treat the mentalis muscle first, she advised. Treatment of this muscle also recruits the depressor anguli oris (DAO) muscle less (the mentalis is the agonist muscle to the DAO). Dr. Hexsel said she generally uses a total dose of 10 U-20 U of abobotulinumtoxinA or 3 U-6 U of onabotulinumtoxinA for this indication.
• Gummy smile. Consider botulinum toxin when a patient complains of a gummy smile. To foster a more natural look, also treat any natural asymmetries or posterior gummy smile, Dr. Hexsel said.
To treat posterior gingival exposure, inject two points on either side of the malar region. Inject in the nasolabial fold at the point of greatest lateral contraction during a smile.
Total doses vary from 5 U to 15 U of abobotulinumtoxinA or from 4 U to 10 U of onabotulinumtoxinA.
A final tip is to take before and after clinical photos, both during movement and at rest, when injecting the lower face.
Dr. Hexsel reported receiving grants and research support from Allergan, Galderma, and Ipsen. She also is a consultant for Allergan and Ipsen.
ORLANDO – Dose is the most important consideration when injecting botulinum toxin in the lower face – even more important than during treatment of the upper face, according to Dr. Doris Hexsel.
"You should always use the lowest effective dose in the lower face," said Dr. Hexsel. This strategy reduces the risk for asymmetry, muscle dysfunction, and temporary oral paralysis. "These are dose-related and technique-related side effects."
Botulinum toxin can treat perioral wrinkles and marionette lines, as well as improve the appearance of a patient’s chin or gummy smile. However, only treat one or two areas in the lower face during the same session to minimize the risk of "sum of effect," she said. In other words, the effects of multiple, simultaneous injections around the mouth can be cumulative.
Also consider a combination of the botulinum toxin and filler injections, Dr. Hexsel said at the annual meeting of the Florida Society of Dermatologic Surgeons.
Dr. Hexsel shared her expertise with botulinum toxin for the following indications:
• Perioral wrinkles. Injections should be superficial and at least 1.5 cm from the corners of the mouth. Any closer and you increase the risk of undesirable relaxation of the depressor anguli oris muscle, the zygomaticus major muscle, and the risorius muscle, said Dr. Hexsel of the department of dermatology at Pontificia Universidade Católica do Rio Grande do Sul, Brazil.
She said that she typically injects 1.25 U-2.5 U abobotulinumtoxinA (Dysport, Ipsen/Medicis) per point, or 0.5 U-1.0 U onabotulinumtoxinA (Botox, Allergan). With two to six injection points, the total dose varies from 4 U-18 U for abobotulinum toxin or 4 U-10 U for onabotulinumtoxinA.
As a bonus, she said, "We observe a slight increase in the volume of the lips due to just the relaxation."
• "Cellulitic chin." Loss of collagen and subcutaneous fat, along with action of the jaw muscles, can cause a chin to have a cellulitic or "peau d’orange" appearance. Botulinum toxin can treat this area as well, said Dr. Hexsel. "I prefer two point injections – bilaterally at the most distal point of the mentalis muscle.
Again, keep the injections superficial and avoid high doses to minimize undesirable relaxation of the depressor labii inferioris muscle, she said. The total dose for a dimpled chin ranges from 15 U to 20 U of abobotulinumtoxinA and from 5 U to 10 U of onabotulinumtoxinA.
"I touch up patients 15 to 30 days later, if necessary," she added.
• Marionette lines. Botulinum toxin can improve the appearance of patients when the corners of their mouth appear permanently turned down. Better results may be obtained by combining toxin with fillers, she said.
For mild presentations, treat the mentalis muscle first, she advised. Treatment of this muscle also recruits the depressor anguli oris (DAO) muscle less (the mentalis is the agonist muscle to the DAO). Dr. Hexsel said she generally uses a total dose of 10 U-20 U of abobotulinumtoxinA or 3 U-6 U of onabotulinumtoxinA for this indication.
• Gummy smile. Consider botulinum toxin when a patient complains of a gummy smile. To foster a more natural look, also treat any natural asymmetries or posterior gummy smile, Dr. Hexsel said.
To treat posterior gingival exposure, inject two points on either side of the malar region. Inject in the nasolabial fold at the point of greatest lateral contraction during a smile.
Total doses vary from 5 U to 15 U of abobotulinumtoxinA or from 4 U to 10 U of onabotulinumtoxinA.
A final tip is to take before and after clinical photos, both during movement and at rest, when injecting the lower face.
Dr. Hexsel reported receiving grants and research support from Allergan, Galderma, and Ipsen. She also is a consultant for Allergan and Ipsen.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGIC SURGEONS
Approved Botulinum Toxins All 'Work About the Same'
LAS VEGAS – Manufacturers of the approved botulinum toxins may claim that their product is superior, but there is likely little difference between them for cosmetic dermatology purposes, according to Dr. Michael H. Gold.
"The reality is, they all work about the same," said Dr. Gold at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery.
Unfortunately, there are hundreds of other botulinum toxin products available on the Internet that have not been tested or approved. "This is where it gets scary," added Dr. Gold, a dermatologist in private practice in Nashville, Tenn.
There is little difference between the three botulinum toxins approved by the Food and Drug Administration – Botox (onabotulinumtoxinA, Allergan), Dysport (abobotulinumtoxinA, Medicis/Ipsen), and Xeomin (incobotulinumtoxinA, Merz) – and a fourth that is likely to be approved, he said. Phase III clinical data for PurTox (Mentor), are being reviewed by the FDA.
"It’s going to get approved eventually, and will have the same approval as all the others," Dr. Gold predicted. He said that his office was the first in the United States to have done research on all four neurotoxins. All are approved in Europe, where they sell under different names.
There are well-known dosing differences between Botox and Dysport. Dosing charts are easily available on the Internet. "If you use both of these, you should have these charts in your office," he said.
Some company-sponsored studies claim differences in efficacy between the neurotoxins, but the industry-sponsored trials should be read skeptically, he said.
A 20-patient study, sponsored by Medicis, showed that Dysport reached full effect faster than Botox when injected into the frontalis muscle, which was already known from the pivotal clinical trials, Dr. Gold said (J. Drugs Dermatol. 2011;10:1148-57).
A separate Medicis-sponsored study of 90 patients treated for lateral orbital rhytids reported that Dysport worked better than Botox (Arch. Facial Plast. Surg. 2011;13:380-6[doi:10.1001/archfacial.2011.37]). On average, the full effect was achieved in 1-2 days with Dysport, compared with 3 days for Botox. "But again, this is a sponsored study," not an independent evaluation, he noted.
Noninferiority studies comparing Xeomin (approved by the FDA in August 2011) and Botox showed that "they work exactly the same," Dr. Gold said. Xeomin’s approval is nearly identical to the approvals of the other botulinum toxins, and its duration is equivalent – "about 3 months," he said.
Xeomin does not contain hemagglutinin and, therefore, should have a lower risk for an antigen effect than Botox and Dysport. Whether or not this is significant is unclear, because there have been no antigen effects reported from the use of Botox or Dysport, he said.
PurTox also is a pure neurotoxin, "which means no resistance," but again it’s not clear if containing hemagglutinin is a risk, he said.
Another difference between the approved toxins is that the FDA required a two-point satisfaction grading scale in the Xeomin clinical studies, compared with a one-point satisfaction grading scale in the studies of Botox and Dysport. To show an effect from Xeomin, the satisfaction rating had to change from 3 to 1 or from 2 to 0.
In comparing the products, "the numbers and how you put them together are hard," Dr. Gold said. "Both Allergan and Medicis are going to tell us how great their product is, and the poor folks from Merz are going to have to tell us the difference between a one-point and two-point satisfaction" rating change.
Data for botulinum toxins that have not been FDA approved, however, are even harder to find.
Makers of neurotoxins available in other parts of the world have their own sponsored studies claiming equivalence to Botox. In China, for example, the search engine Alibaba turns up hundreds of botulinum toxin products for sale from at least 24 suppliers, said Dr. Gold. Most of them have no data to back them up, are sold illegally, and put the buyer at risk of a jail sentence.
An Allergan researcher analyzed a product from Nanfeng, China, and found that it contained 240 times the dose of botulinum toxin available in a vial of Botox. It killed every animal on which it was tested until it was diluted in a 1:240 ratio (J. Am. Acad. Dermatol. 2009;61:149-50).
Several well-manufactured neurotoxins are available in China that have gone through extensive Chinese regulatory processes, but caution is needed even with these, Dr. Gold said. In one open-label study comparing ChinaTox with Botox for the treatment of spasms and dystonia, five patients developed rash, even though the neurotoxin is less powerful than Botox.
"You’ve got to be careful what you do," he said, and suggested that dermatologists stick with branded and approved neurotoxins.
Dr. Gold has been a consultant or researcher for Allergan, Galderma, Medicis, Mentor (Johnson & Johnson), and Merz Aesthetics.
LAS VEGAS – Manufacturers of the approved botulinum toxins may claim that their product is superior, but there is likely little difference between them for cosmetic dermatology purposes, according to Dr. Michael H. Gold.
"The reality is, they all work about the same," said Dr. Gold at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery.
Unfortunately, there are hundreds of other botulinum toxin products available on the Internet that have not been tested or approved. "This is where it gets scary," added Dr. Gold, a dermatologist in private practice in Nashville, Tenn.
There is little difference between the three botulinum toxins approved by the Food and Drug Administration – Botox (onabotulinumtoxinA, Allergan), Dysport (abobotulinumtoxinA, Medicis/Ipsen), and Xeomin (incobotulinumtoxinA, Merz) – and a fourth that is likely to be approved, he said. Phase III clinical data for PurTox (Mentor), are being reviewed by the FDA.
"It’s going to get approved eventually, and will have the same approval as all the others," Dr. Gold predicted. He said that his office was the first in the United States to have done research on all four neurotoxins. All are approved in Europe, where they sell under different names.
There are well-known dosing differences between Botox and Dysport. Dosing charts are easily available on the Internet. "If you use both of these, you should have these charts in your office," he said.
Some company-sponsored studies claim differences in efficacy between the neurotoxins, but the industry-sponsored trials should be read skeptically, he said.
A 20-patient study, sponsored by Medicis, showed that Dysport reached full effect faster than Botox when injected into the frontalis muscle, which was already known from the pivotal clinical trials, Dr. Gold said (J. Drugs Dermatol. 2011;10:1148-57).
A separate Medicis-sponsored study of 90 patients treated for lateral orbital rhytids reported that Dysport worked better than Botox (Arch. Facial Plast. Surg. 2011;13:380-6[doi:10.1001/archfacial.2011.37]). On average, the full effect was achieved in 1-2 days with Dysport, compared with 3 days for Botox. "But again, this is a sponsored study," not an independent evaluation, he noted.
Noninferiority studies comparing Xeomin (approved by the FDA in August 2011) and Botox showed that "they work exactly the same," Dr. Gold said. Xeomin’s approval is nearly identical to the approvals of the other botulinum toxins, and its duration is equivalent – "about 3 months," he said.
Xeomin does not contain hemagglutinin and, therefore, should have a lower risk for an antigen effect than Botox and Dysport. Whether or not this is significant is unclear, because there have been no antigen effects reported from the use of Botox or Dysport, he said.
PurTox also is a pure neurotoxin, "which means no resistance," but again it’s not clear if containing hemagglutinin is a risk, he said.
Another difference between the approved toxins is that the FDA required a two-point satisfaction grading scale in the Xeomin clinical studies, compared with a one-point satisfaction grading scale in the studies of Botox and Dysport. To show an effect from Xeomin, the satisfaction rating had to change from 3 to 1 or from 2 to 0.
In comparing the products, "the numbers and how you put them together are hard," Dr. Gold said. "Both Allergan and Medicis are going to tell us how great their product is, and the poor folks from Merz are going to have to tell us the difference between a one-point and two-point satisfaction" rating change.
Data for botulinum toxins that have not been FDA approved, however, are even harder to find.
Makers of neurotoxins available in other parts of the world have their own sponsored studies claiming equivalence to Botox. In China, for example, the search engine Alibaba turns up hundreds of botulinum toxin products for sale from at least 24 suppliers, said Dr. Gold. Most of them have no data to back them up, are sold illegally, and put the buyer at risk of a jail sentence.
An Allergan researcher analyzed a product from Nanfeng, China, and found that it contained 240 times the dose of botulinum toxin available in a vial of Botox. It killed every animal on which it was tested until it was diluted in a 1:240 ratio (J. Am. Acad. Dermatol. 2009;61:149-50).
Several well-manufactured neurotoxins are available in China that have gone through extensive Chinese regulatory processes, but caution is needed even with these, Dr. Gold said. In one open-label study comparing ChinaTox with Botox for the treatment of spasms and dystonia, five patients developed rash, even though the neurotoxin is less powerful than Botox.
"You’ve got to be careful what you do," he said, and suggested that dermatologists stick with branded and approved neurotoxins.
Dr. Gold has been a consultant or researcher for Allergan, Galderma, Medicis, Mentor (Johnson & Johnson), and Merz Aesthetics.
LAS VEGAS – Manufacturers of the approved botulinum toxins may claim that their product is superior, but there is likely little difference between them for cosmetic dermatology purposes, according to Dr. Michael H. Gold.
"The reality is, they all work about the same," said Dr. Gold at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery.
Unfortunately, there are hundreds of other botulinum toxin products available on the Internet that have not been tested or approved. "This is where it gets scary," added Dr. Gold, a dermatologist in private practice in Nashville, Tenn.
There is little difference between the three botulinum toxins approved by the Food and Drug Administration – Botox (onabotulinumtoxinA, Allergan), Dysport (abobotulinumtoxinA, Medicis/Ipsen), and Xeomin (incobotulinumtoxinA, Merz) – and a fourth that is likely to be approved, he said. Phase III clinical data for PurTox (Mentor), are being reviewed by the FDA.
"It’s going to get approved eventually, and will have the same approval as all the others," Dr. Gold predicted. He said that his office was the first in the United States to have done research on all four neurotoxins. All are approved in Europe, where they sell under different names.
There are well-known dosing differences between Botox and Dysport. Dosing charts are easily available on the Internet. "If you use both of these, you should have these charts in your office," he said.
Some company-sponsored studies claim differences in efficacy between the neurotoxins, but the industry-sponsored trials should be read skeptically, he said.
A 20-patient study, sponsored by Medicis, showed that Dysport reached full effect faster than Botox when injected into the frontalis muscle, which was already known from the pivotal clinical trials, Dr. Gold said (J. Drugs Dermatol. 2011;10:1148-57).
A separate Medicis-sponsored study of 90 patients treated for lateral orbital rhytids reported that Dysport worked better than Botox (Arch. Facial Plast. Surg. 2011;13:380-6[doi:10.1001/archfacial.2011.37]). On average, the full effect was achieved in 1-2 days with Dysport, compared with 3 days for Botox. "But again, this is a sponsored study," not an independent evaluation, he noted.
Noninferiority studies comparing Xeomin (approved by the FDA in August 2011) and Botox showed that "they work exactly the same," Dr. Gold said. Xeomin’s approval is nearly identical to the approvals of the other botulinum toxins, and its duration is equivalent – "about 3 months," he said.
Xeomin does not contain hemagglutinin and, therefore, should have a lower risk for an antigen effect than Botox and Dysport. Whether or not this is significant is unclear, because there have been no antigen effects reported from the use of Botox or Dysport, he said.
PurTox also is a pure neurotoxin, "which means no resistance," but again it’s not clear if containing hemagglutinin is a risk, he said.
Another difference between the approved toxins is that the FDA required a two-point satisfaction grading scale in the Xeomin clinical studies, compared with a one-point satisfaction grading scale in the studies of Botox and Dysport. To show an effect from Xeomin, the satisfaction rating had to change from 3 to 1 or from 2 to 0.
In comparing the products, "the numbers and how you put them together are hard," Dr. Gold said. "Both Allergan and Medicis are going to tell us how great their product is, and the poor folks from Merz are going to have to tell us the difference between a one-point and two-point satisfaction" rating change.
Data for botulinum toxins that have not been FDA approved, however, are even harder to find.
Makers of neurotoxins available in other parts of the world have their own sponsored studies claiming equivalence to Botox. In China, for example, the search engine Alibaba turns up hundreds of botulinum toxin products for sale from at least 24 suppliers, said Dr. Gold. Most of them have no data to back them up, are sold illegally, and put the buyer at risk of a jail sentence.
An Allergan researcher analyzed a product from Nanfeng, China, and found that it contained 240 times the dose of botulinum toxin available in a vial of Botox. It killed every animal on which it was tested until it was diluted in a 1:240 ratio (J. Am. Acad. Dermatol. 2009;61:149-50).
Several well-manufactured neurotoxins are available in China that have gone through extensive Chinese regulatory processes, but caution is needed even with these, Dr. Gold said. In one open-label study comparing ChinaTox with Botox for the treatment of spasms and dystonia, five patients developed rash, even though the neurotoxin is less powerful than Botox.
"You’ve got to be careful what you do," he said, and suggested that dermatologists stick with branded and approved neurotoxins.
Dr. Gold has been a consultant or researcher for Allergan, Galderma, Medicis, Mentor (Johnson & Johnson), and Merz Aesthetics.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF COSMETIC DERMATOLOGY AND AESTHETIC SURGERY
Blog: Cosmeceutical Experts Agree on Best Anti-Aging Product
Three of the top experts on cosmeceuticals agreed that the best over-the-counter, anti-aging products come down to two simple words: "moisturizer" and "sunscreen."
There is oh, so much more out there being sold in the $9 billion/year skin care market, much of which are anti-aging products. But the skin regimen that provides the biggest bang for the buck: moisturizer and sunscreen. Spend $100 on a product, and chances are its moisturizer and sunscreen anyway.
Speaking in separate presentations at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery (ASCDAS), Dr. Zoe D. Draelos, Dr. Ellen S. Marmur and Dr. Michael H. Gold agreed that there is little science to back up claims made by cosmeceutical companies.
Cosmeceuticals fall somewhere between cosmetics and pharmaceuticals; therefore, they are not regulated, and manufacturers are not required to show evidence of anti-aging effectiveness. When evidence does exist, it generally points back to – you guessed it – moisturizer and sunscreen.
Dr. Draelos a dermatologist in High Point, N.C., and a consulting professor of dermatology at Duke University, studied the ingredients in over-the-counter skin care products. She found that 80% of products are moisturizers that serve as a vehicle to deliver whichever high-profile ingredient is being touted by the manufacturer (Plast. Reconst. Surg. 2010;125:719-24).
"Hands down, it's the moisturizer" that's the most important cosmeceutical choice, she said. "All the products that make anti-aging claims are making moisturizer claims," and most anti-aging products also have sunscreen ingredients in them.
When Dr. Gold first spoke at a medical conference many years ago about selling cosmeceutical products in his practice, most dermatologists frowned on the idea. He said he has been dispensing cosmeceuticals for 22 years and has seen the market explode. "There is now a $700 moisturizer," he said. "There may be no difference" between that and a $10 moisturizer, "but some people will pay the $700 because it's $700."
Dr. Gold, who is based in Nashville, urged his colleagues, "If you dispense, do it ethically."
Dermatologists have an important role to play by not just listening to marketing claims of cosmeceuticals, but assessing them. "Even though we don't have a lot of good research right now, patients want something their doctor feels good about," said Dr. Marmur of Mount Sinai School of Medicine, New York, and president of the ASCDAS.
She took that approach in her book, written with Gina Way, Simple Skin Beauty: Every Woman’s Guide to a Lifetime of Healthy, Gorgeous Skin (Atria Books, 2009).
The book "perhaps put me at odds with some of the industry, but we're challenging them to produce better science," she said.
Dr. Marmur and her associates are conducting the first randomized, controlled trial comparing over-the-counter anti-aging creams. With more than 130 subjects enrolled, the study will evaluate 28 biomarkers in seven lines of products, with the patients who apply the products blinded to the product being used.
Not all anti-aging cream manufacturers are included, however. "Some brands refused to participate," perhaps because they were afraid that the study would show that their product is effective but is a pharmaceutical, she said.
Dr. Draelos echoed that idea during the question-and-answer session: "The industry doesn't want closer scrutiny," she said.
I also heard Dr. Draelos speak recently at the Skin Disease and Education Foundation's Women's and Pediatric Dermatology Seminar. Her recommendations for cosmeceuticals include starting with a moisturizer containing dimethicone, glycerin, and/or petrolatum. "The most robust moisturizer known to man has these three ingredients," she said. For anti-aging, she suggests a sunscreen containing avobenzone, oxybenzone, octocrylene, reflecting spheres, or antioxidant botanicals.
How does that translate into over-the-counter products? For hands, Dr. Draelos favors Neutrogena Norwegian Formula Hand Cream. For the face, she recommends Johnson & Johnson's Aveeno Positively Radiant Daily Moisturizer SPF 30. And for the body, Galderma's Cetaphil cream.
Dr. Draelos has been a consultant and researcher for Johnson & Johnson, L'Oreal, Procter & Gamble, Nu Skin, Avon, Stiefel, and Dial Corp. Dr. Marmur disclosed financial relationships with Allergan, DUSA Pharmaceuticals, Genentech, Medicis, Merz, and Sanofi Aventis. Dr. Gold has been a consultant or researcher for Allergan, Medicis, Mentor (Johnson & Johnson), Merz, Galderma, and numerous other companies.
SDEF and this news organization are owned by Elsevier.
Three of the top experts on cosmeceuticals agreed that the best over-the-counter, anti-aging products come down to two simple words: "moisturizer" and "sunscreen."
There is oh, so much more out there being sold in the $9 billion/year skin care market, much of which are anti-aging products. But the skin regimen that provides the biggest bang for the buck: moisturizer and sunscreen. Spend $100 on a product, and chances are its moisturizer and sunscreen anyway.
Speaking in separate presentations at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery (ASCDAS), Dr. Zoe D. Draelos, Dr. Ellen S. Marmur and Dr. Michael H. Gold agreed that there is little science to back up claims made by cosmeceutical companies.
Cosmeceuticals fall somewhere between cosmetics and pharmaceuticals; therefore, they are not regulated, and manufacturers are not required to show evidence of anti-aging effectiveness. When evidence does exist, it generally points back to – you guessed it – moisturizer and sunscreen.
Dr. Draelos a dermatologist in High Point, N.C., and a consulting professor of dermatology at Duke University, studied the ingredients in over-the-counter skin care products. She found that 80% of products are moisturizers that serve as a vehicle to deliver whichever high-profile ingredient is being touted by the manufacturer (Plast. Reconst. Surg. 2010;125:719-24).
"Hands down, it's the moisturizer" that's the most important cosmeceutical choice, she said. "All the products that make anti-aging claims are making moisturizer claims," and most anti-aging products also have sunscreen ingredients in them.
When Dr. Gold first spoke at a medical conference many years ago about selling cosmeceutical products in his practice, most dermatologists frowned on the idea. He said he has been dispensing cosmeceuticals for 22 years and has seen the market explode. "There is now a $700 moisturizer," he said. "There may be no difference" between that and a $10 moisturizer, "but some people will pay the $700 because it's $700."
Dr. Gold, who is based in Nashville, urged his colleagues, "If you dispense, do it ethically."
Dermatologists have an important role to play by not just listening to marketing claims of cosmeceuticals, but assessing them. "Even though we don't have a lot of good research right now, patients want something their doctor feels good about," said Dr. Marmur of Mount Sinai School of Medicine, New York, and president of the ASCDAS.
She took that approach in her book, written with Gina Way, Simple Skin Beauty: Every Woman’s Guide to a Lifetime of Healthy, Gorgeous Skin (Atria Books, 2009).
The book "perhaps put me at odds with some of the industry, but we're challenging them to produce better science," she said.
Dr. Marmur and her associates are conducting the first randomized, controlled trial comparing over-the-counter anti-aging creams. With more than 130 subjects enrolled, the study will evaluate 28 biomarkers in seven lines of products, with the patients who apply the products blinded to the product being used.
Not all anti-aging cream manufacturers are included, however. "Some brands refused to participate," perhaps because they were afraid that the study would show that their product is effective but is a pharmaceutical, she said.
Dr. Draelos echoed that idea during the question-and-answer session: "The industry doesn't want closer scrutiny," she said.
I also heard Dr. Draelos speak recently at the Skin Disease and Education Foundation's Women's and Pediatric Dermatology Seminar. Her recommendations for cosmeceuticals include starting with a moisturizer containing dimethicone, glycerin, and/or petrolatum. "The most robust moisturizer known to man has these three ingredients," she said. For anti-aging, she suggests a sunscreen containing avobenzone, oxybenzone, octocrylene, reflecting spheres, or antioxidant botanicals.
How does that translate into over-the-counter products? For hands, Dr. Draelos favors Neutrogena Norwegian Formula Hand Cream. For the face, she recommends Johnson & Johnson's Aveeno Positively Radiant Daily Moisturizer SPF 30. And for the body, Galderma's Cetaphil cream.
Dr. Draelos has been a consultant and researcher for Johnson & Johnson, L'Oreal, Procter & Gamble, Nu Skin, Avon, Stiefel, and Dial Corp. Dr. Marmur disclosed financial relationships with Allergan, DUSA Pharmaceuticals, Genentech, Medicis, Merz, and Sanofi Aventis. Dr. Gold has been a consultant or researcher for Allergan, Medicis, Mentor (Johnson & Johnson), Merz, Galderma, and numerous other companies.
SDEF and this news organization are owned by Elsevier.
Three of the top experts on cosmeceuticals agreed that the best over-the-counter, anti-aging products come down to two simple words: "moisturizer" and "sunscreen."
There is oh, so much more out there being sold in the $9 billion/year skin care market, much of which are anti-aging products. But the skin regimen that provides the biggest bang for the buck: moisturizer and sunscreen. Spend $100 on a product, and chances are its moisturizer and sunscreen anyway.
Speaking in separate presentations at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery (ASCDAS), Dr. Zoe D. Draelos, Dr. Ellen S. Marmur and Dr. Michael H. Gold agreed that there is little science to back up claims made by cosmeceutical companies.
Cosmeceuticals fall somewhere between cosmetics and pharmaceuticals; therefore, they are not regulated, and manufacturers are not required to show evidence of anti-aging effectiveness. When evidence does exist, it generally points back to – you guessed it – moisturizer and sunscreen.
Dr. Draelos a dermatologist in High Point, N.C., and a consulting professor of dermatology at Duke University, studied the ingredients in over-the-counter skin care products. She found that 80% of products are moisturizers that serve as a vehicle to deliver whichever high-profile ingredient is being touted by the manufacturer (Plast. Reconst. Surg. 2010;125:719-24).
"Hands down, it's the moisturizer" that's the most important cosmeceutical choice, she said. "All the products that make anti-aging claims are making moisturizer claims," and most anti-aging products also have sunscreen ingredients in them.
When Dr. Gold first spoke at a medical conference many years ago about selling cosmeceutical products in his practice, most dermatologists frowned on the idea. He said he has been dispensing cosmeceuticals for 22 years and has seen the market explode. "There is now a $700 moisturizer," he said. "There may be no difference" between that and a $10 moisturizer, "but some people will pay the $700 because it's $700."
Dr. Gold, who is based in Nashville, urged his colleagues, "If you dispense, do it ethically."
Dermatologists have an important role to play by not just listening to marketing claims of cosmeceuticals, but assessing them. "Even though we don't have a lot of good research right now, patients want something their doctor feels good about," said Dr. Marmur of Mount Sinai School of Medicine, New York, and president of the ASCDAS.
She took that approach in her book, written with Gina Way, Simple Skin Beauty: Every Woman’s Guide to a Lifetime of Healthy, Gorgeous Skin (Atria Books, 2009).
The book "perhaps put me at odds with some of the industry, but we're challenging them to produce better science," she said.
Dr. Marmur and her associates are conducting the first randomized, controlled trial comparing over-the-counter anti-aging creams. With more than 130 subjects enrolled, the study will evaluate 28 biomarkers in seven lines of products, with the patients who apply the products blinded to the product being used.
Not all anti-aging cream manufacturers are included, however. "Some brands refused to participate," perhaps because they were afraid that the study would show that their product is effective but is a pharmaceutical, she said.
Dr. Draelos echoed that idea during the question-and-answer session: "The industry doesn't want closer scrutiny," she said.
I also heard Dr. Draelos speak recently at the Skin Disease and Education Foundation's Women's and Pediatric Dermatology Seminar. Her recommendations for cosmeceuticals include starting with a moisturizer containing dimethicone, glycerin, and/or petrolatum. "The most robust moisturizer known to man has these three ingredients," she said. For anti-aging, she suggests a sunscreen containing avobenzone, oxybenzone, octocrylene, reflecting spheres, or antioxidant botanicals.
How does that translate into over-the-counter products? For hands, Dr. Draelos favors Neutrogena Norwegian Formula Hand Cream. For the face, she recommends Johnson & Johnson's Aveeno Positively Radiant Daily Moisturizer SPF 30. And for the body, Galderma's Cetaphil cream.
Dr. Draelos has been a consultant and researcher for Johnson & Johnson, L'Oreal, Procter & Gamble, Nu Skin, Avon, Stiefel, and Dial Corp. Dr. Marmur disclosed financial relationships with Allergan, DUSA Pharmaceuticals, Genentech, Medicis, Merz, and Sanofi Aventis. Dr. Gold has been a consultant or researcher for Allergan, Medicis, Mentor (Johnson & Johnson), Merz, Galderma, and numerous other companies.
SDEF and this news organization are owned by Elsevier.
Endovenous Lasers Have Revolutionized Leg Vein Treatment
LAS VEGAS – Endovenous laser therapy is replacing ligation and vein stripping for many patients with superficial venous incompetence, especially in the legs.
"The endovenous laser has been a major revolutionary advance in the treatment of medical varicose veins of the lower extremities," Dr. Neil S. Sadick said at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery.
Endovenous laser fibers are inserted in the vessel under ultrasound guidance to eradicate truncal varicosities in the short and lower saphenous veins. The laser heat transfer causes shrinkage of the vein wall collagen and decreased lumen, with the shrinkage proportional to the delivered linear endovenous energy density.
"This is an extremely easy procedure," that takes about 15 minutes, said Dr. Sadick, a dermatologist at Weill Cornell Medical Center in New York. "It’s almost bloodless. Patients can go back to work that day" wearing light compression hose, and "there’s very little discomfort after" the procedure.
Recurrence rates with endovenous laser therapy also are lower, compared with conventional invasive surgical ligation and stripping procedures, he added. Studies by Dr. Sadick and his associates showed that recurrence rates after treatment of superficial venous incompetence with a combination of endovascular laser and ambulatory phlebectomy were approximately 6% at 1 year, 4% at 2 years, 3% at 3 years, and 4% at 4 years.
Endovenous laser therapy "induces an endothelial type of thrombosis, and then the vein gets dissolved by the body," he said.
Treatment Algorithm
Dr. Sadick developed an algorithm for treatment based on the type of leg varicosity. He uses endovenous laser therapy or endovenous radiofrequency technology to treat large varicose veins of the axial junctions, such as in the long or short saphenous veins. Most intermediate-size varicose veins, such as truncal varicosities or perforations, can be treated by either ambulatory phlebectomy or foam sclerotherapy.
For reticular veins, he prefers to treat with an external 1064-nm Nd:YAG (neodymium YAG) laser or sclerotherapy with or without foam. Microtelangiectasia, or "very, very small vessels," can be treated with microsclerotherapy via very dilute concentrations of sclerosant, "but this is where an external 1064-nm Nd:YAG laser plays an important role" and may suffice without microsclerotherapy, he said.
"Not all leg veins are treated equally," he added. Red telangiectasias measuring less than 1 mm in tiny, oxygenated, red vessels usually are superficial and are "hit hard" with short pulse durations of high-fluence external laser energy in small spot sizes of 1-2 mm.
For more bluish veins that measure 1-2 mm (which he called blue venulectasia) and for larger reticular varicosities that are 2-4 mm, spot sizes and pulse durations increase but with more moderate fluences. External laser settings for blue venulectasia would be a spot size of 2-4 mm with a medium pulse width and moderately high fluence. For reticular veins, he uses an external spot size of 4-6 mm with a long pulse width and moderate fluence.
"You can see excellent results if you use this paradigm of variable pulse moding for treating telangiectasia without even injecting patients," he said.
Lasers are indicated for the cosmetic treatment of leg veins in patients whose veins are too small to cannulize for sclerotherapy, or for the small subset of patients who do not respond to sclerotherapy. Patients with needle phobia or those who have multiple sclerosant allergies also are candidates for cosmetic laser treatment of leg veins.
Leg veins require different treatment than do facial telangiectasias, Dr. Sadick added. Hydrostatic pressure is greater in the legs. Lower extremity vessels are larger, with increased basal lamina, compared with facial telangiectasias. The deeper location of many lower-extremity vessels makes access more difficult.
Caution Is Advised
The longer wavelengths and higher fluences of external laser treatment for leg veins cause a greater inflammatory reaction. "The worst thing you can do is treat patients on a weekly or biweekly basis" with external laser, he said. "You need to wait 6-8 weeks after each laser treatment so all the inflammation can resolve before the next treatment session" in order to avoid complications.
Dr. Sadick cautioned that the external 1064-nm Nd:YAG lasers are "sort of like weapons. They need to be handled very gently and understood, particularly when you’re treating the legs." Complications can include ulceration, purpura, blisters, and hyper- or hypopigmentation. These most commonly are caused by stacking pulses in a given area (especially with longer wavelengths), treating tanned skin, improperly matching wavelength to skin type, and failing to address hydrostatic pressure.
"If you use a short wavelength laser like a pulsed dye laser for a dark-skinned individual, for sure you’re going to get hypopigmentation and potential scarring," he said. Avoid laser treatment in patients who have larger vessels feeding the spider telangiectasia, because the hydrostatic pressure increases the risk for side effects.
The clinical goals of laser therapy for leg telangiectasia are to treat vasospasm, erythema, and urticaria. "It’s important to understand the end points of therapy. You don’t need to treat these veins until the vessels go away," he said.
Dr. Sadick has consulted for or received research grants from Cutera, Cynosure, Palomar, Solta Medical, and Syneron.
LAS VEGAS – Endovenous laser therapy is replacing ligation and vein stripping for many patients with superficial venous incompetence, especially in the legs.
"The endovenous laser has been a major revolutionary advance in the treatment of medical varicose veins of the lower extremities," Dr. Neil S. Sadick said at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery.
Endovenous laser fibers are inserted in the vessel under ultrasound guidance to eradicate truncal varicosities in the short and lower saphenous veins. The laser heat transfer causes shrinkage of the vein wall collagen and decreased lumen, with the shrinkage proportional to the delivered linear endovenous energy density.
"This is an extremely easy procedure," that takes about 15 minutes, said Dr. Sadick, a dermatologist at Weill Cornell Medical Center in New York. "It’s almost bloodless. Patients can go back to work that day" wearing light compression hose, and "there’s very little discomfort after" the procedure.
Recurrence rates with endovenous laser therapy also are lower, compared with conventional invasive surgical ligation and stripping procedures, he added. Studies by Dr. Sadick and his associates showed that recurrence rates after treatment of superficial venous incompetence with a combination of endovascular laser and ambulatory phlebectomy were approximately 6% at 1 year, 4% at 2 years, 3% at 3 years, and 4% at 4 years.
Endovenous laser therapy "induces an endothelial type of thrombosis, and then the vein gets dissolved by the body," he said.
Treatment Algorithm
Dr. Sadick developed an algorithm for treatment based on the type of leg varicosity. He uses endovenous laser therapy or endovenous radiofrequency technology to treat large varicose veins of the axial junctions, such as in the long or short saphenous veins. Most intermediate-size varicose veins, such as truncal varicosities or perforations, can be treated by either ambulatory phlebectomy or foam sclerotherapy.
For reticular veins, he prefers to treat with an external 1064-nm Nd:YAG (neodymium YAG) laser or sclerotherapy with or without foam. Microtelangiectasia, or "very, very small vessels," can be treated with microsclerotherapy via very dilute concentrations of sclerosant, "but this is where an external 1064-nm Nd:YAG laser plays an important role" and may suffice without microsclerotherapy, he said.
"Not all leg veins are treated equally," he added. Red telangiectasias measuring less than 1 mm in tiny, oxygenated, red vessels usually are superficial and are "hit hard" with short pulse durations of high-fluence external laser energy in small spot sizes of 1-2 mm.
For more bluish veins that measure 1-2 mm (which he called blue venulectasia) and for larger reticular varicosities that are 2-4 mm, spot sizes and pulse durations increase but with more moderate fluences. External laser settings for blue venulectasia would be a spot size of 2-4 mm with a medium pulse width and moderately high fluence. For reticular veins, he uses an external spot size of 4-6 mm with a long pulse width and moderate fluence.
"You can see excellent results if you use this paradigm of variable pulse moding for treating telangiectasia without even injecting patients," he said.
Lasers are indicated for the cosmetic treatment of leg veins in patients whose veins are too small to cannulize for sclerotherapy, or for the small subset of patients who do not respond to sclerotherapy. Patients with needle phobia or those who have multiple sclerosant allergies also are candidates for cosmetic laser treatment of leg veins.
Leg veins require different treatment than do facial telangiectasias, Dr. Sadick added. Hydrostatic pressure is greater in the legs. Lower extremity vessels are larger, with increased basal lamina, compared with facial telangiectasias. The deeper location of many lower-extremity vessels makes access more difficult.
Caution Is Advised
The longer wavelengths and higher fluences of external laser treatment for leg veins cause a greater inflammatory reaction. "The worst thing you can do is treat patients on a weekly or biweekly basis" with external laser, he said. "You need to wait 6-8 weeks after each laser treatment so all the inflammation can resolve before the next treatment session" in order to avoid complications.
Dr. Sadick cautioned that the external 1064-nm Nd:YAG lasers are "sort of like weapons. They need to be handled very gently and understood, particularly when you’re treating the legs." Complications can include ulceration, purpura, blisters, and hyper- or hypopigmentation. These most commonly are caused by stacking pulses in a given area (especially with longer wavelengths), treating tanned skin, improperly matching wavelength to skin type, and failing to address hydrostatic pressure.
"If you use a short wavelength laser like a pulsed dye laser for a dark-skinned individual, for sure you’re going to get hypopigmentation and potential scarring," he said. Avoid laser treatment in patients who have larger vessels feeding the spider telangiectasia, because the hydrostatic pressure increases the risk for side effects.
The clinical goals of laser therapy for leg telangiectasia are to treat vasospasm, erythema, and urticaria. "It’s important to understand the end points of therapy. You don’t need to treat these veins until the vessels go away," he said.
Dr. Sadick has consulted for or received research grants from Cutera, Cynosure, Palomar, Solta Medical, and Syneron.
LAS VEGAS – Endovenous laser therapy is replacing ligation and vein stripping for many patients with superficial venous incompetence, especially in the legs.
"The endovenous laser has been a major revolutionary advance in the treatment of medical varicose veins of the lower extremities," Dr. Neil S. Sadick said at the annual meeting of the American Society of Cosmetic Dermatology and Aesthetic Surgery.
Endovenous laser fibers are inserted in the vessel under ultrasound guidance to eradicate truncal varicosities in the short and lower saphenous veins. The laser heat transfer causes shrinkage of the vein wall collagen and decreased lumen, with the shrinkage proportional to the delivered linear endovenous energy density.
"This is an extremely easy procedure," that takes about 15 minutes, said Dr. Sadick, a dermatologist at Weill Cornell Medical Center in New York. "It’s almost bloodless. Patients can go back to work that day" wearing light compression hose, and "there’s very little discomfort after" the procedure.
Recurrence rates with endovenous laser therapy also are lower, compared with conventional invasive surgical ligation and stripping procedures, he added. Studies by Dr. Sadick and his associates showed that recurrence rates after treatment of superficial venous incompetence with a combination of endovascular laser and ambulatory phlebectomy were approximately 6% at 1 year, 4% at 2 years, 3% at 3 years, and 4% at 4 years.
Endovenous laser therapy "induces an endothelial type of thrombosis, and then the vein gets dissolved by the body," he said.
Treatment Algorithm
Dr. Sadick developed an algorithm for treatment based on the type of leg varicosity. He uses endovenous laser therapy or endovenous radiofrequency technology to treat large varicose veins of the axial junctions, such as in the long or short saphenous veins. Most intermediate-size varicose veins, such as truncal varicosities or perforations, can be treated by either ambulatory phlebectomy or foam sclerotherapy.
For reticular veins, he prefers to treat with an external 1064-nm Nd:YAG (neodymium YAG) laser or sclerotherapy with or without foam. Microtelangiectasia, or "very, very small vessels," can be treated with microsclerotherapy via very dilute concentrations of sclerosant, "but this is where an external 1064-nm Nd:YAG laser plays an important role" and may suffice without microsclerotherapy, he said.
"Not all leg veins are treated equally," he added. Red telangiectasias measuring less than 1 mm in tiny, oxygenated, red vessels usually are superficial and are "hit hard" with short pulse durations of high-fluence external laser energy in small spot sizes of 1-2 mm.
For more bluish veins that measure 1-2 mm (which he called blue venulectasia) and for larger reticular varicosities that are 2-4 mm, spot sizes and pulse durations increase but with more moderate fluences. External laser settings for blue venulectasia would be a spot size of 2-4 mm with a medium pulse width and moderately high fluence. For reticular veins, he uses an external spot size of 4-6 mm with a long pulse width and moderate fluence.
"You can see excellent results if you use this paradigm of variable pulse moding for treating telangiectasia without even injecting patients," he said.
Lasers are indicated for the cosmetic treatment of leg veins in patients whose veins are too small to cannulize for sclerotherapy, or for the small subset of patients who do not respond to sclerotherapy. Patients with needle phobia or those who have multiple sclerosant allergies also are candidates for cosmetic laser treatment of leg veins.
Leg veins require different treatment than do facial telangiectasias, Dr. Sadick added. Hydrostatic pressure is greater in the legs. Lower extremity vessels are larger, with increased basal lamina, compared with facial telangiectasias. The deeper location of many lower-extremity vessels makes access more difficult.
Caution Is Advised
The longer wavelengths and higher fluences of external laser treatment for leg veins cause a greater inflammatory reaction. "The worst thing you can do is treat patients on a weekly or biweekly basis" with external laser, he said. "You need to wait 6-8 weeks after each laser treatment so all the inflammation can resolve before the next treatment session" in order to avoid complications.
Dr. Sadick cautioned that the external 1064-nm Nd:YAG lasers are "sort of like weapons. They need to be handled very gently and understood, particularly when you’re treating the legs." Complications can include ulceration, purpura, blisters, and hyper- or hypopigmentation. These most commonly are caused by stacking pulses in a given area (especially with longer wavelengths), treating tanned skin, improperly matching wavelength to skin type, and failing to address hydrostatic pressure.
"If you use a short wavelength laser like a pulsed dye laser for a dark-skinned individual, for sure you’re going to get hypopigmentation and potential scarring," he said. Avoid laser treatment in patients who have larger vessels feeding the spider telangiectasia, because the hydrostatic pressure increases the risk for side effects.
The clinical goals of laser therapy for leg telangiectasia are to treat vasospasm, erythema, and urticaria. "It’s important to understand the end points of therapy. You don’t need to treat these veins until the vessels go away," he said.
Dr. Sadick has consulted for or received research grants from Cutera, Cynosure, Palomar, Solta Medical, and Syneron.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF COSMETIC DERMATOLOGY AND AESTHETIC SURGERY
Chronic Inflammation Implies Perineural Invasion
SAN DIEGO – Evidence of chronic inflammation noted during Mohs surgery is a telltale sign of perineural invasion.
"If you see chronic inflammatory infiltrate within or proximal to a neurovascular bundle, look for perineural tumor," said Dr. Alexander Miller, a dermatologic surgeon in private practice in Yorba Linda, Calif. Histologically, that means abundant lymphocytes and perhaps histiocytes.
An abundance of neutrophils, however, is likely indicative of an acute inflammatory response. Perhaps even a response resulting from electrocautery during a Mohs procedure.
Neutrophils might also be present if a keratinizing tumor has ruptured into stroma, generating a microabscess. "But then it’s pretty darned obvious what you have," said Dr. Miller at a meeting sponsored by the American Society for Mohs Surgery.
Finding perineural tumor cells within a cluster of inflammatory cells can be like hunting for the proverbial needle in a haystack. A low-power view might miss them, he said. On medium or higher power views, tumor cells may appear as minute dots, a tiny stripe, or a sliver along one edge of a nerve.
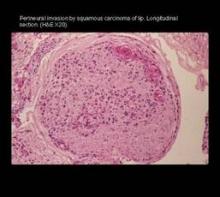
To demonstrate, Dr. Miller displayed a slide depicting voluminous chronic inflammation surrounding an artery, vessel, and nerve.
"No tumor," he said.
"But if one looks carefully, two sections down, same slide, same patient, lo and behold there’s the tumor. Complacency should not be had here. One has to ensure that particularly when there’s inflammation, one needs to look very carefully at all sections of the slide."
Dr. Miller reported having no financial disclosures relevant to his talk.
SAN DIEGO – Evidence of chronic inflammation noted during Mohs surgery is a telltale sign of perineural invasion.
"If you see chronic inflammatory infiltrate within or proximal to a neurovascular bundle, look for perineural tumor," said Dr. Alexander Miller, a dermatologic surgeon in private practice in Yorba Linda, Calif. Histologically, that means abundant lymphocytes and perhaps histiocytes.
An abundance of neutrophils, however, is likely indicative of an acute inflammatory response. Perhaps even a response resulting from electrocautery during a Mohs procedure.
Neutrophils might also be present if a keratinizing tumor has ruptured into stroma, generating a microabscess. "But then it’s pretty darned obvious what you have," said Dr. Miller at a meeting sponsored by the American Society for Mohs Surgery.
Finding perineural tumor cells within a cluster of inflammatory cells can be like hunting for the proverbial needle in a haystack. A low-power view might miss them, he said. On medium or higher power views, tumor cells may appear as minute dots, a tiny stripe, or a sliver along one edge of a nerve.
To demonstrate, Dr. Miller displayed a slide depicting voluminous chronic inflammation surrounding an artery, vessel, and nerve.
"No tumor," he said.
"But if one looks carefully, two sections down, same slide, same patient, lo and behold there’s the tumor. Complacency should not be had here. One has to ensure that particularly when there’s inflammation, one needs to look very carefully at all sections of the slide."
Dr. Miller reported having no financial disclosures relevant to his talk.
SAN DIEGO – Evidence of chronic inflammation noted during Mohs surgery is a telltale sign of perineural invasion.
"If you see chronic inflammatory infiltrate within or proximal to a neurovascular bundle, look for perineural tumor," said Dr. Alexander Miller, a dermatologic surgeon in private practice in Yorba Linda, Calif. Histologically, that means abundant lymphocytes and perhaps histiocytes.
An abundance of neutrophils, however, is likely indicative of an acute inflammatory response. Perhaps even a response resulting from electrocautery during a Mohs procedure.
Neutrophils might also be present if a keratinizing tumor has ruptured into stroma, generating a microabscess. "But then it’s pretty darned obvious what you have," said Dr. Miller at a meeting sponsored by the American Society for Mohs Surgery.
Finding perineural tumor cells within a cluster of inflammatory cells can be like hunting for the proverbial needle in a haystack. A low-power view might miss them, he said. On medium or higher power views, tumor cells may appear as minute dots, a tiny stripe, or a sliver along one edge of a nerve.
To demonstrate, Dr. Miller displayed a slide depicting voluminous chronic inflammation surrounding an artery, vessel, and nerve.
"No tumor," he said.
"But if one looks carefully, two sections down, same slide, same patient, lo and behold there’s the tumor. Complacency should not be had here. One has to ensure that particularly when there’s inflammation, one needs to look very carefully at all sections of the slide."
Dr. Miller reported having no financial disclosures relevant to his talk.
EXPERT ANALYSIS FROM A MEETING SPONSORED BY THE AMERICAN SOCIETY FOR MOHS SURGERY