User login
Less than half of office visits involve primary care
, according to the National Center for Health Statistics.
Primary care physicians’ share of office visits fell from 66.2% in 1980 to 49.1% in 2013, the NCHS reported in “Health, United States, 2016.” The corresponding increase among specialty care physicians gave them a total of 50.9% of all office visits in 2013, up from 33.8% in 1980.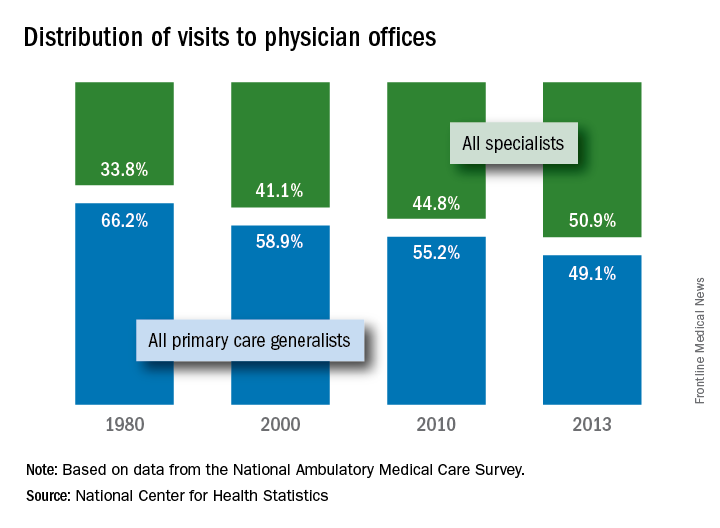
The NCHS estimates are based on data collected by the National Ambulatory Medical Care Survey, which excluded Alaska and Hawaii in 1980.
, according to the National Center for Health Statistics.
Primary care physicians’ share of office visits fell from 66.2% in 1980 to 49.1% in 2013, the NCHS reported in “Health, United States, 2016.” The corresponding increase among specialty care physicians gave them a total of 50.9% of all office visits in 2013, up from 33.8% in 1980.
The NCHS estimates are based on data collected by the National Ambulatory Medical Care Survey, which excluded Alaska and Hawaii in 1980.
, according to the National Center for Health Statistics.
Primary care physicians’ share of office visits fell from 66.2% in 1980 to 49.1% in 2013, the NCHS reported in “Health, United States, 2016.” The corresponding increase among specialty care physicians gave them a total of 50.9% of all office visits in 2013, up from 33.8% in 1980.
The NCHS estimates are based on data collected by the National Ambulatory Medical Care Survey, which excluded Alaska and Hawaii in 1980.
FDA approves faster, pangenotypic cure for hep C virus
The first pangenotypic treatment for the hepatitis C virus, which also shaves 4 weeks off current regimens, has just been approved by the Food and Drug Administration.
Manufactured by AbbVie, glecaprevir/pibrentasvir (Mavyret) combines a nonstructural protein 3/4A protease inhibitor with a next-generation NS5A protein inhibitor for a once-daily, ribavirin-free treatment for adults with any of the major genotypes of chronic hepatitis C virus (HCV) infection.
“This approval provides a shorter treatment duration for many patients, and also a treatment option for certain patients with genotype 1 infection, the most common HCV genotype in the United States, who were not successfully treated with other direct-acting antiviral treatments in the past,” Edward Cox, MD, director of the office of antimicrobial products in the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md., said in a statement.
The 8-week regimen is indicated in patients without cirrhosis or with compensated cirrhosis, who are new to treatment, and those with limited treatment options, such as patients with chronic kidney disease, including those on dialysis. The intervention also is indicated in adults with HCV genotype 1 who have been treated with either of the drugs in the combination, but not both. Glecaprevir/pibrentasvir is not recommended in patients with moderate cirrhosis and is contraindicated in patients with severe cirrhosis and in those taking the drugs atazanavir and rifampin.
The safety and efficacy of the treatment were evaluated in approximately 2,300 adults with genotype 1, 2, 3, 4, 5 or 6 HCV infection without cirrhosis or with mild cirrhosis. In the clinical trials, between 92% and 100% of patients treated with glecaprevir/pibrentasvir for 8, 12, or 16 weeks had no detectable serum levels of the virus 12 weeks after finishing treatment. The most commonly reported adverse reactions were headache, fatigue, and nausea.
The FDA directs health care professionals to test all patients for current or prior hepatitis B virus (HBV) infection prior to starting this direct-acting antiviral drug combination since HBV reactivation has been reported in adult patients coinfected with both viruses who were undergoing or had completed treatment with HCV direct-acting antivirals and who were not receiving HBV antiviral therapy.
The first pangenotypic treatment for the hepatitis C virus, which also shaves 4 weeks off current regimens, has just been approved by the Food and Drug Administration.
Manufactured by AbbVie, glecaprevir/pibrentasvir (Mavyret) combines a nonstructural protein 3/4A protease inhibitor with a next-generation NS5A protein inhibitor for a once-daily, ribavirin-free treatment for adults with any of the major genotypes of chronic hepatitis C virus (HCV) infection.
“This approval provides a shorter treatment duration for many patients, and also a treatment option for certain patients with genotype 1 infection, the most common HCV genotype in the United States, who were not successfully treated with other direct-acting antiviral treatments in the past,” Edward Cox, MD, director of the office of antimicrobial products in the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md., said in a statement.
The 8-week regimen is indicated in patients without cirrhosis or with compensated cirrhosis, who are new to treatment, and those with limited treatment options, such as patients with chronic kidney disease, including those on dialysis. The intervention also is indicated in adults with HCV genotype 1 who have been treated with either of the drugs in the combination, but not both. Glecaprevir/pibrentasvir is not recommended in patients with moderate cirrhosis and is contraindicated in patients with severe cirrhosis and in those taking the drugs atazanavir and rifampin.
The safety and efficacy of the treatment were evaluated in approximately 2,300 adults with genotype 1, 2, 3, 4, 5 or 6 HCV infection without cirrhosis or with mild cirrhosis. In the clinical trials, between 92% and 100% of patients treated with glecaprevir/pibrentasvir for 8, 12, or 16 weeks had no detectable serum levels of the virus 12 weeks after finishing treatment. The most commonly reported adverse reactions were headache, fatigue, and nausea.
The FDA directs health care professionals to test all patients for current or prior hepatitis B virus (HBV) infection prior to starting this direct-acting antiviral drug combination since HBV reactivation has been reported in adult patients coinfected with both viruses who were undergoing or had completed treatment with HCV direct-acting antivirals and who were not receiving HBV antiviral therapy.
The first pangenotypic treatment for the hepatitis C virus, which also shaves 4 weeks off current regimens, has just been approved by the Food and Drug Administration.
Manufactured by AbbVie, glecaprevir/pibrentasvir (Mavyret) combines a nonstructural protein 3/4A protease inhibitor with a next-generation NS5A protein inhibitor for a once-daily, ribavirin-free treatment for adults with any of the major genotypes of chronic hepatitis C virus (HCV) infection.
“This approval provides a shorter treatment duration for many patients, and also a treatment option for certain patients with genotype 1 infection, the most common HCV genotype in the United States, who were not successfully treated with other direct-acting antiviral treatments in the past,” Edward Cox, MD, director of the office of antimicrobial products in the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md., said in a statement.
The 8-week regimen is indicated in patients without cirrhosis or with compensated cirrhosis, who are new to treatment, and those with limited treatment options, such as patients with chronic kidney disease, including those on dialysis. The intervention also is indicated in adults with HCV genotype 1 who have been treated with either of the drugs in the combination, but not both. Glecaprevir/pibrentasvir is not recommended in patients with moderate cirrhosis and is contraindicated in patients with severe cirrhosis and in those taking the drugs atazanavir and rifampin.
The safety and efficacy of the treatment were evaluated in approximately 2,300 adults with genotype 1, 2, 3, 4, 5 or 6 HCV infection without cirrhosis or with mild cirrhosis. In the clinical trials, between 92% and 100% of patients treated with glecaprevir/pibrentasvir for 8, 12, or 16 weeks had no detectable serum levels of the virus 12 weeks after finishing treatment. The most commonly reported adverse reactions were headache, fatigue, and nausea.
The FDA directs health care professionals to test all patients for current or prior hepatitis B virus (HBV) infection prior to starting this direct-acting antiviral drug combination since HBV reactivation has been reported in adult patients coinfected with both viruses who were undergoing or had completed treatment with HCV direct-acting antivirals and who were not receiving HBV antiviral therapy.
Abilify Maintena OK’d by FDA for adults with bipolar I disorder
The Food and Drug Administration has approved a monthly injectable formulation of aripiprazole, (Abilify Maintena) , for a maintenance monotherapy treatment of bipolar I disorder for adults, Otsuka and Lundbeck have announced.
Patients treated with the injectable formulation of the atypical antipsychotic must continue to take a daily oral antipsychotic for the first 14 days. After that, however, the long-acting injectable (LAI) – which must be administered by a health care professional – can replace the daily medication.
Joseph R. Calabrese, MD, director of the mood disorders program at University Hospitals Cleveland Medical Center, said in the July 28 announcement that the LAI is a new treatment option for bipolar I patients “who have established tolerability with oral aripiprazole.”
The drug label includes a warning that elderly patients with dementia-related psychosis who are treated with antipsychotics are at a higher mortality risk. Adverse reactions that have been associated with treatment with aripiprazole include weight gain, akathisia, injection site pain, sedation, and certain compulsive behaviors.
Created by Otsuka, and marketed by Otsuka and Lundbeck, the LAI was approved in the United States for treating adults with schizophrenia in 2013.
The Food and Drug Administration has approved a monthly injectable formulation of aripiprazole, (Abilify Maintena) , for a maintenance monotherapy treatment of bipolar I disorder for adults, Otsuka and Lundbeck have announced.
Patients treated with the injectable formulation of the atypical antipsychotic must continue to take a daily oral antipsychotic for the first 14 days. After that, however, the long-acting injectable (LAI) – which must be administered by a health care professional – can replace the daily medication.
Joseph R. Calabrese, MD, director of the mood disorders program at University Hospitals Cleveland Medical Center, said in the July 28 announcement that the LAI is a new treatment option for bipolar I patients “who have established tolerability with oral aripiprazole.”
The drug label includes a warning that elderly patients with dementia-related psychosis who are treated with antipsychotics are at a higher mortality risk. Adverse reactions that have been associated with treatment with aripiprazole include weight gain, akathisia, injection site pain, sedation, and certain compulsive behaviors.
Created by Otsuka, and marketed by Otsuka and Lundbeck, the LAI was approved in the United States for treating adults with schizophrenia in 2013.
The Food and Drug Administration has approved a monthly injectable formulation of aripiprazole, (Abilify Maintena) , for a maintenance monotherapy treatment of bipolar I disorder for adults, Otsuka and Lundbeck have announced.
Patients treated with the injectable formulation of the atypical antipsychotic must continue to take a daily oral antipsychotic for the first 14 days. After that, however, the long-acting injectable (LAI) – which must be administered by a health care professional – can replace the daily medication.
Joseph R. Calabrese, MD, director of the mood disorders program at University Hospitals Cleveland Medical Center, said in the July 28 announcement that the LAI is a new treatment option for bipolar I patients “who have established tolerability with oral aripiprazole.”
The drug label includes a warning that elderly patients with dementia-related psychosis who are treated with antipsychotics are at a higher mortality risk. Adverse reactions that have been associated with treatment with aripiprazole include weight gain, akathisia, injection site pain, sedation, and certain compulsive behaviors.
Created by Otsuka, and marketed by Otsuka and Lundbeck, the LAI was approved in the United States for treating adults with schizophrenia in 2013.
Pain frequency varies by employment status
Adults who were previously employed are twice as likely to report daily or almost-daily pain than are those who are currently employed, according to the Centers for Disease Control and Prevention.
In an ongoing survey, just over 30% of adults aged 18 years and older who were previously employed reported that they experienced pain on “most days or every day” in the past 6 months, compared with 15% of those who were currently employed and 19% of those classified as never employed, investigators from the CDC estimated (MMWR. 2017 Jul 28;66[29]:796).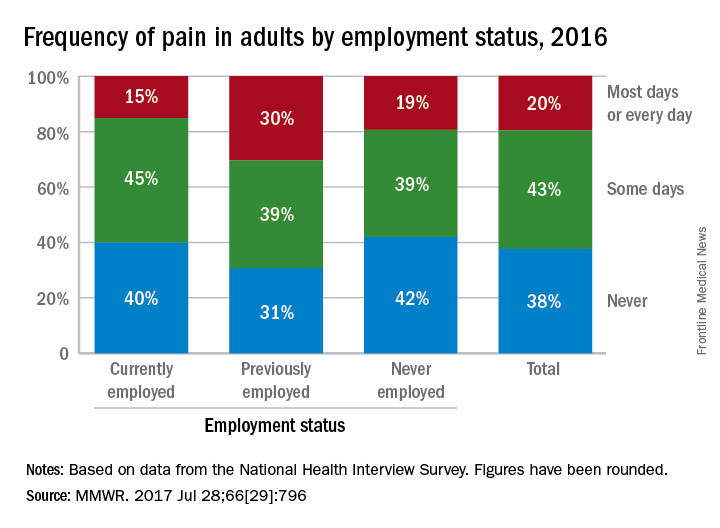
Adults who were previously employed are twice as likely to report daily or almost-daily pain than are those who are currently employed, according to the Centers for Disease Control and Prevention.
In an ongoing survey, just over 30% of adults aged 18 years and older who were previously employed reported that they experienced pain on “most days or every day” in the past 6 months, compared with 15% of those who were currently employed and 19% of those classified as never employed, investigators from the CDC estimated (MMWR. 2017 Jul 28;66[29]:796).
Adults who were previously employed are twice as likely to report daily or almost-daily pain than are those who are currently employed, according to the Centers for Disease Control and Prevention.
In an ongoing survey, just over 30% of adults aged 18 years and older who were previously employed reported that they experienced pain on “most days or every day” in the past 6 months, compared with 15% of those who were currently employed and 19% of those classified as never employed, investigators from the CDC estimated (MMWR. 2017 Jul 28;66[29]:796).
FROM MMWR
Fujifilm issues recall to update ED-530XT duodenoscopes
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
The AGA Center for GI Innovation and Technology is committed to working towards zero device-associated infections and will keep you apprised of any future updates. Learn more at www.gastro.org/CGIT.
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
The AGA Center for GI Innovation and Technology is committed to working towards zero device-associated infections and will keep you apprised of any future updates. Learn more at www.gastro.org/CGIT.
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
The AGA Center for GI Innovation and Technology is committed to working towards zero device-associated infections and will keep you apprised of any future updates. Learn more at www.gastro.org/CGIT.
Over 45% of American adults have type 2 diabetes mellitus or prediabetes
More than 114 million American adults have type 2 diabetes mellitus or prediabetes, according to estimates from the Centers for Disease Control and Prevention.
As of 2015, the combined prevalence of diabetes and prediabetes was 45.4% among adults in the United States: 11.5% (30.3 million) have diabetes and 33.9% have prediabetes, representing 84.1 million people who could develop type 2 diabetes within 5 years, the CDC said in the National Diabetes Statistics Report, 2017.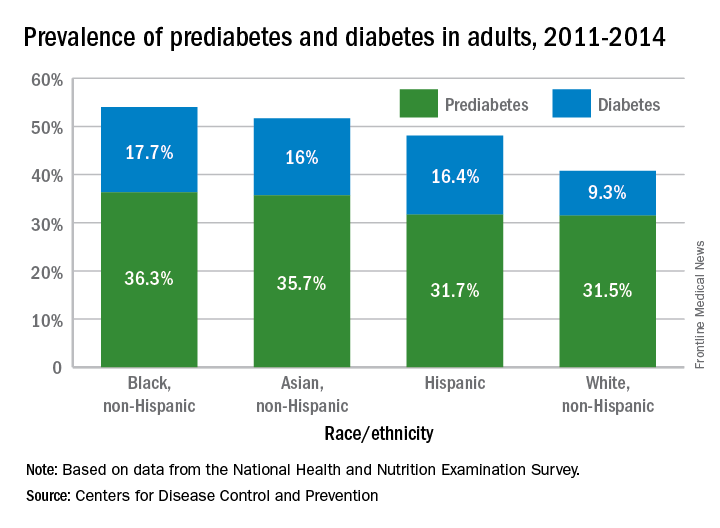
Lack of knowledge about having the disease was common: Almost a quarter (23.8%) of adults with diabetes didn’t know they had it, and the number jumped to 88% for those with prediabetes, based on data from the National Health and Nutrition Examination Survey, which included a blood test for fasting plasma glucose or hemoglobin A1c.
“More than a third of U.S. adults have prediabetes, and the majority don’t know it. Now, more than ever, we must step up our efforts to reduce the burden of this serious disease,” CDC Director Brenda Fitzgerald, MD, said in a statement.
“Diabetes and prediabetes remain serious threats for more than one-third of Americans – the statistics are staggering,” William T. Cefalu, MD, chief scientific, medical, and mission officer of the American Diabetes Association, said in a written statement.
“We must continue to innovate in scientific research and to translate findings to the clinical level to decrease the prevalence of diabetes. These data clearly confirm it is critical for us to continue to provide the education and support needed to improve health outcomes and decrease the daily burden of diabetes. We must reduce the incidence of diabetes and its enormous costs, including both the financial costs and the human toll of lost quality of life,” he said.
“Diabetes and prediabetes remain serious threats for more than one-third of Americans – the statistics are staggering,” William T. Cefalu, MD, chief scientific, medical, and mission officer of the American Diabetes Association, said in a written statement.
“We must continue to innovate in scientific research and to translate findings to the clinical level to decrease the prevalence of diabetes. These data clearly confirm it is critical for us to continue to provide the education and support needed to improve health outcomes and decrease the daily burden of diabetes. We must reduce the incidence of diabetes and its enormous costs, including both the financial costs and the human toll of lost quality of life,” he said.
“Diabetes and prediabetes remain serious threats for more than one-third of Americans – the statistics are staggering,” William T. Cefalu, MD, chief scientific, medical, and mission officer of the American Diabetes Association, said in a written statement.
“We must continue to innovate in scientific research and to translate findings to the clinical level to decrease the prevalence of diabetes. These data clearly confirm it is critical for us to continue to provide the education and support needed to improve health outcomes and decrease the daily burden of diabetes. We must reduce the incidence of diabetes and its enormous costs, including both the financial costs and the human toll of lost quality of life,” he said.
More than 114 million American adults have type 2 diabetes mellitus or prediabetes, according to estimates from the Centers for Disease Control and Prevention.
As of 2015, the combined prevalence of diabetes and prediabetes was 45.4% among adults in the United States: 11.5% (30.3 million) have diabetes and 33.9% have prediabetes, representing 84.1 million people who could develop type 2 diabetes within 5 years, the CDC said in the National Diabetes Statistics Report, 2017.
Lack of knowledge about having the disease was common: Almost a quarter (23.8%) of adults with diabetes didn’t know they had it, and the number jumped to 88% for those with prediabetes, based on data from the National Health and Nutrition Examination Survey, which included a blood test for fasting plasma glucose or hemoglobin A1c.
“More than a third of U.S. adults have prediabetes, and the majority don’t know it. Now, more than ever, we must step up our efforts to reduce the burden of this serious disease,” CDC Director Brenda Fitzgerald, MD, said in a statement.
More than 114 million American adults have type 2 diabetes mellitus or prediabetes, according to estimates from the Centers for Disease Control and Prevention.
As of 2015, the combined prevalence of diabetes and prediabetes was 45.4% among adults in the United States: 11.5% (30.3 million) have diabetes and 33.9% have prediabetes, representing 84.1 million people who could develop type 2 diabetes within 5 years, the CDC said in the National Diabetes Statistics Report, 2017.
Lack of knowledge about having the disease was common: Almost a quarter (23.8%) of adults with diabetes didn’t know they had it, and the number jumped to 88% for those with prediabetes, based on data from the National Health and Nutrition Examination Survey, which included a blood test for fasting plasma glucose or hemoglobin A1c.
“More than a third of U.S. adults have prediabetes, and the majority don’t know it. Now, more than ever, we must step up our efforts to reduce the burden of this serious disease,” CDC Director Brenda Fitzgerald, MD, said in a statement.
Substance use treatment cost $34 billion in 2014
Total spending on the treatment of substance use disorder reached $34 billion in 2014, with outpatient care taking the largest share, according to the National Center for Health Statistics.
That $34 billion represents an increase of 273% from the $9.1 billion spent in 1986 and a considerable shift in the distribution of spending over the last 30 years. In 1986, the largest share of spending – 50%, or $4.6 billion – for substance use disorder went toward inpatient care and only $2.4 billion (27%) was used for outpatient care. In 2014, outpatient treatment of substance use disorder had a 40% share ($13.6 billion) of all spending, and inpatient care was down to 19% ($6.4 billion), the NCHS reported in “Health, United States, 2016.”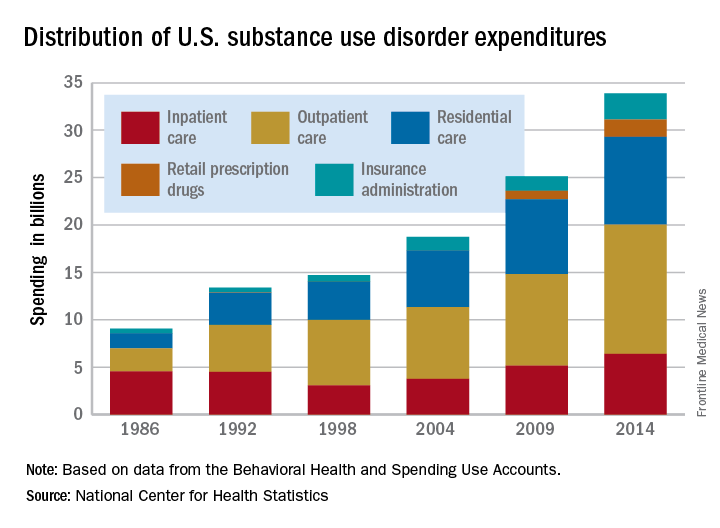
Since methadone is not considered a retail drug by SAMHSA, it is classified under specialty substance use disorder treatment centers, which are included in the estimate for outpatient care, the NCHS noted.
Total spending on the treatment of substance use disorder reached $34 billion in 2014, with outpatient care taking the largest share, according to the National Center for Health Statistics.
That $34 billion represents an increase of 273% from the $9.1 billion spent in 1986 and a considerable shift in the distribution of spending over the last 30 years. In 1986, the largest share of spending – 50%, or $4.6 billion – for substance use disorder went toward inpatient care and only $2.4 billion (27%) was used for outpatient care. In 2014, outpatient treatment of substance use disorder had a 40% share ($13.6 billion) of all spending, and inpatient care was down to 19% ($6.4 billion), the NCHS reported in “Health, United States, 2016.”
Since methadone is not considered a retail drug by SAMHSA, it is classified under specialty substance use disorder treatment centers, which are included in the estimate for outpatient care, the NCHS noted.
Total spending on the treatment of substance use disorder reached $34 billion in 2014, with outpatient care taking the largest share, according to the National Center for Health Statistics.
That $34 billion represents an increase of 273% from the $9.1 billion spent in 1986 and a considerable shift in the distribution of spending over the last 30 years. In 1986, the largest share of spending – 50%, or $4.6 billion – for substance use disorder went toward inpatient care and only $2.4 billion (27%) was used for outpatient care. In 2014, outpatient treatment of substance use disorder had a 40% share ($13.6 billion) of all spending, and inpatient care was down to 19% ($6.4 billion), the NCHS reported in “Health, United States, 2016.”
Since methadone is not considered a retail drug by SAMHSA, it is classified under specialty substance use disorder treatment centers, which are included in the estimate for outpatient care, the NCHS noted.
Vaccine reduced risk for flu visits by 42%
Last year’s influenza vaccination reduced the overall risk for flu-related medical visits by 42%, according to the Centers for Disease Control and Prevention.
In an article summarizing influenza activity in the United States during October 2016–May 2017, investigators said that most of the viral strains antigenically characterized at the CDC “were similar to the reference viruses representing the recommended components for the 2016-2017 vaccine.”
The 2017-2018 influenza vaccine has been updated to include an additional influenza A (H1N1) component. This change was recommended by the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee, based on data from global influenza virologic and epidemiologic surveillance, genetic and antigenic characterization, human serology studies, antiviral susceptibility, and the availability of candidate influenza viruses (MMWR. 2017;66[25]:668-76).
Preliminary data show that, during the 2016-2017 flu season, there were 18,184 laboratory-confirmed, flu-related hospitalizations, for an overall incidence of 65 per 100,000 population, more than double that for the 2015-2017 season (31/100,000). Broken down by age groups, the rates per 100,000 population in this past season were 44 at ages 0-4 years, 17 at ages 5-17 years, 20 at ages 18-49 years, and 65 at ages 50-64 years, compared with 291 at ages 65 years and older. Finalized estimates of the number of influenza illnesses, medical visits, and hospitalizations averted by vaccination during the 2016-2017 season will be published in December, the investigators said.
Last year’s influenza vaccination reduced the overall risk for flu-related medical visits by 42%, according to the Centers for Disease Control and Prevention.
In an article summarizing influenza activity in the United States during October 2016–May 2017, investigators said that most of the viral strains antigenically characterized at the CDC “were similar to the reference viruses representing the recommended components for the 2016-2017 vaccine.”
The 2017-2018 influenza vaccine has been updated to include an additional influenza A (H1N1) component. This change was recommended by the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee, based on data from global influenza virologic and epidemiologic surveillance, genetic and antigenic characterization, human serology studies, antiviral susceptibility, and the availability of candidate influenza viruses (MMWR. 2017;66[25]:668-76).
Preliminary data show that, during the 2016-2017 flu season, there were 18,184 laboratory-confirmed, flu-related hospitalizations, for an overall incidence of 65 per 100,000 population, more than double that for the 2015-2017 season (31/100,000). Broken down by age groups, the rates per 100,000 population in this past season were 44 at ages 0-4 years, 17 at ages 5-17 years, 20 at ages 18-49 years, and 65 at ages 50-64 years, compared with 291 at ages 65 years and older. Finalized estimates of the number of influenza illnesses, medical visits, and hospitalizations averted by vaccination during the 2016-2017 season will be published in December, the investigators said.
Last year’s influenza vaccination reduced the overall risk for flu-related medical visits by 42%, according to the Centers for Disease Control and Prevention.
In an article summarizing influenza activity in the United States during October 2016–May 2017, investigators said that most of the viral strains antigenically characterized at the CDC “were similar to the reference viruses representing the recommended components for the 2016-2017 vaccine.”
The 2017-2018 influenza vaccine has been updated to include an additional influenza A (H1N1) component. This change was recommended by the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee, based on data from global influenza virologic and epidemiologic surveillance, genetic and antigenic characterization, human serology studies, antiviral susceptibility, and the availability of candidate influenza viruses (MMWR. 2017;66[25]:668-76).
Preliminary data show that, during the 2016-2017 flu season, there were 18,184 laboratory-confirmed, flu-related hospitalizations, for an overall incidence of 65 per 100,000 population, more than double that for the 2015-2017 season (31/100,000). Broken down by age groups, the rates per 100,000 population in this past season were 44 at ages 0-4 years, 17 at ages 5-17 years, 20 at ages 18-49 years, and 65 at ages 50-64 years, compared with 291 at ages 65 years and older. Finalized estimates of the number of influenza illnesses, medical visits, and hospitalizations averted by vaccination during the 2016-2017 season will be published in December, the investigators said.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Key clinical point: Last year’s influenza vaccination reduced the overall risk for flu-related medical visits by 42%.
Major finding: During the 2016-2017 flu season, there were 18,184 laboratory-confirmed, flu-related hospitalizations, for an overall incidence of 65 per 100,000 population.
Data source: A review of data submitted to the Centers for Disease Control and Prevention regarding the 2016-2017 influenza season.
Disclosures: This study was supported by the CDC. Dr. Blanton and her associates reported having no relevant financial disclosures.
First IL-23 blocker, guselkumab, earns FDA approval for psoriasis
, based on three phase 3 studies of more than 2,000 adults, the manufacturer announced July 13.
The approved indication is for adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy, according to a press release issued by Janssen Biotech, which stated that this is the first IL-23 blocker approved for psoriasis.
Results of one of the phase 3 trials, VOYAGE 1, included a significantly greater proportion of patients treated with guselkumab achieving at least a 90% improvement in the Psoriasis Area Severity Index (PASI 90) at 16 weeks, compared with placebo (73.3% vs. 2.9%). At 16 weeks, 85.1% of those treated with guselkumab achieved an Investigator’s Global Assessment (IGA) score of 0 (cleared) or 1 (minimal disease), compared with 6.9% of those on placebo. Superior responses continued through 48 weeks.
In an active comparator arm of the study comparing guselkumab with the TNF blocker adalimumab (Humira), a significantly higher proportion of those treated with guselkumab achieved PASI 90 scores (76.3% vs. 47.9%) and IGA 0/1 scores (80.5% vs. 55.4%) at week 48. The results were published in March (J Am Acad Dermatol. 2017 Mar;76[3]:405-17).
Results of VOYAGE 2 comparing guselkumab with adalimumab included a PASI 90 rate of 66.1% at week 48 among adalimumab nonresponders who switched to guselkumab (J Am Acad Dermatol. 2017 Mar;76[3]:418-31).
The most common serious adverse effects associated with treatment included upper respiratory infections, headache, injection site reactions, arthralgias, diarrhea, gastroenteritis, fungal skin infections, and herpes simplex infections, according to the company statement.
Phase 3 studies of guselkumab for active psoriatic arthritis and in comparison with secukinumab (Cosentyx) in patients with moderate to severe plaque psoriasis are underway, according to Janssen, which is marketing guselkumab as Tremfya.
, based on three phase 3 studies of more than 2,000 adults, the manufacturer announced July 13.
The approved indication is for adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy, according to a press release issued by Janssen Biotech, which stated that this is the first IL-23 blocker approved for psoriasis.
Results of one of the phase 3 trials, VOYAGE 1, included a significantly greater proportion of patients treated with guselkumab achieving at least a 90% improvement in the Psoriasis Area Severity Index (PASI 90) at 16 weeks, compared with placebo (73.3% vs. 2.9%). At 16 weeks, 85.1% of those treated with guselkumab achieved an Investigator’s Global Assessment (IGA) score of 0 (cleared) or 1 (minimal disease), compared with 6.9% of those on placebo. Superior responses continued through 48 weeks.
In an active comparator arm of the study comparing guselkumab with the TNF blocker adalimumab (Humira), a significantly higher proportion of those treated with guselkumab achieved PASI 90 scores (76.3% vs. 47.9%) and IGA 0/1 scores (80.5% vs. 55.4%) at week 48. The results were published in March (J Am Acad Dermatol. 2017 Mar;76[3]:405-17).
Results of VOYAGE 2 comparing guselkumab with adalimumab included a PASI 90 rate of 66.1% at week 48 among adalimumab nonresponders who switched to guselkumab (J Am Acad Dermatol. 2017 Mar;76[3]:418-31).
The most common serious adverse effects associated with treatment included upper respiratory infections, headache, injection site reactions, arthralgias, diarrhea, gastroenteritis, fungal skin infections, and herpes simplex infections, according to the company statement.
Phase 3 studies of guselkumab for active psoriatic arthritis and in comparison with secukinumab (Cosentyx) in patients with moderate to severe plaque psoriasis are underway, according to Janssen, which is marketing guselkumab as Tremfya.
, based on three phase 3 studies of more than 2,000 adults, the manufacturer announced July 13.
The approved indication is for adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy, according to a press release issued by Janssen Biotech, which stated that this is the first IL-23 blocker approved for psoriasis.
Results of one of the phase 3 trials, VOYAGE 1, included a significantly greater proportion of patients treated with guselkumab achieving at least a 90% improvement in the Psoriasis Area Severity Index (PASI 90) at 16 weeks, compared with placebo (73.3% vs. 2.9%). At 16 weeks, 85.1% of those treated with guselkumab achieved an Investigator’s Global Assessment (IGA) score of 0 (cleared) or 1 (minimal disease), compared with 6.9% of those on placebo. Superior responses continued through 48 weeks.
In an active comparator arm of the study comparing guselkumab with the TNF blocker adalimumab (Humira), a significantly higher proportion of those treated with guselkumab achieved PASI 90 scores (76.3% vs. 47.9%) and IGA 0/1 scores (80.5% vs. 55.4%) at week 48. The results were published in March (J Am Acad Dermatol. 2017 Mar;76[3]:405-17).
Results of VOYAGE 2 comparing guselkumab with adalimumab included a PASI 90 rate of 66.1% at week 48 among adalimumab nonresponders who switched to guselkumab (J Am Acad Dermatol. 2017 Mar;76[3]:418-31).
The most common serious adverse effects associated with treatment included upper respiratory infections, headache, injection site reactions, arthralgias, diarrhea, gastroenteritis, fungal skin infections, and herpes simplex infections, according to the company statement.
Phase 3 studies of guselkumab for active psoriatic arthritis and in comparison with secukinumab (Cosentyx) in patients with moderate to severe plaque psoriasis are underway, according to Janssen, which is marketing guselkumab as Tremfya.
Use of Pap smears, mammography on the decline
Use of Pap smears and mammography has slowly but steadily declined since both peaked in the year 2000, according to the National Center for Health Statistics.
The age-adjusted rate of women aged 18 years and over who reported having had a Pap smear in the past 3 years dropped from 81.3% in 2000 to 70.2% in 2015. Over that same time period, the age-adjusted rate of women aged 40 years and over who had a mammogram over the previous 2 years declined from 70.4% in 2000 to 64% in 2015, the NCHS reported in “Health, United States, 2016.”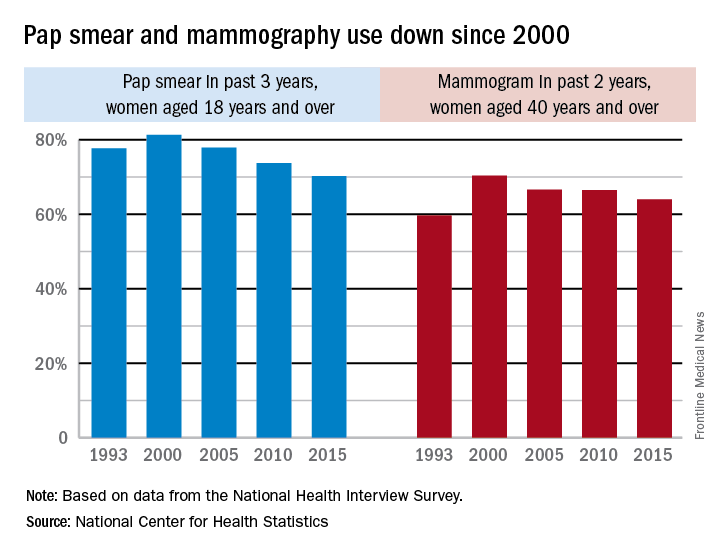
The pattern for mammography use, however, was somewhat different. Declines in use were consistent by age group, but not by race. White women were less likely to get a mammogram in 2015 (65.3%), compared with 2000 (71.4%), while Hispanic women experienced a small drop from 61.2% to 60.9%, but black, American Indian/Alaska Native, and Asian women all increased their use of mammography over that time period, the NCHS reported.
Use of Pap smears and mammography has slowly but steadily declined since both peaked in the year 2000, according to the National Center for Health Statistics.
The age-adjusted rate of women aged 18 years and over who reported having had a Pap smear in the past 3 years dropped from 81.3% in 2000 to 70.2% in 2015. Over that same time period, the age-adjusted rate of women aged 40 years and over who had a mammogram over the previous 2 years declined from 70.4% in 2000 to 64% in 2015, the NCHS reported in “Health, United States, 2016.”
The pattern for mammography use, however, was somewhat different. Declines in use were consistent by age group, but not by race. White women were less likely to get a mammogram in 2015 (65.3%), compared with 2000 (71.4%), while Hispanic women experienced a small drop from 61.2% to 60.9%, but black, American Indian/Alaska Native, and Asian women all increased their use of mammography over that time period, the NCHS reported.
Use of Pap smears and mammography has slowly but steadily declined since both peaked in the year 2000, according to the National Center for Health Statistics.
The age-adjusted rate of women aged 18 years and over who reported having had a Pap smear in the past 3 years dropped from 81.3% in 2000 to 70.2% in 2015. Over that same time period, the age-adjusted rate of women aged 40 years and over who had a mammogram over the previous 2 years declined from 70.4% in 2000 to 64% in 2015, the NCHS reported in “Health, United States, 2016.”
The pattern for mammography use, however, was somewhat different. Declines in use were consistent by age group, but not by race. White women were less likely to get a mammogram in 2015 (65.3%), compared with 2000 (71.4%), while Hispanic women experienced a small drop from 61.2% to 60.9%, but black, American Indian/Alaska Native, and Asian women all increased their use of mammography over that time period, the NCHS reported.