User login
MDedge latest news is breaking news from medical conferences, journals, guidelines, the FDA and CDC.
Belly Fat Beats BMI in Predicting Colorectal Cancer Risk
TOPLINE:
METHODOLOGY:
- General obesity, often measured using BMI, is a recognized risk factor for colorectal cancer, but how much of this association is due to central obesity is unclear.
- Researchers assessed the associations between BMI, waist-to-hip ratio (WHR), and waist circumference (WC) with colorectal cancer risk and the degree of independence among these associations in patients aged 40-69 years recruited in the UK Biobank cohort study from 2006 to 2010.
- Anthropometric measurements were performed using standardized methods.
- Cancer registry and hospital data linkage identified colorectal cancer cases in the UK Biobank.
TAKEAWAY:
- Researchers included 460,784 participants (mean age, 56.3 years; 46.7% men), of whom 67.1% had either overweight or obesity, and 49.4% and 60.5% had high or very high WHR and WC, respectively.
- During the median 12.5-year follow-up period, 5977 participants developed colorectal cancer.
- Every SD increase in WHR (hazard ratio [HR], 1.18) showed a stronger association with colorectal cancer risk than in BMI (HR, 1.10).
- After adjustment for BMI, the association between WHR and colorectal cancer risk became slightly attenuated while still staying robust (HR, 1.15); however, after adjusting for WHR, the association between BMI and colorectal cancer risk became substantially weakened (HR, 1.04).
- WHR showed strongly significant associations with colorectal cancer risk across all BMI categories, whereas associations of BMI with colorectal cancer risk were weak and not statistically significant within all WHR categories.
- Central obesity demonstrated consistent associations with both colon and rectal cancer risks in both sexes before and after adjustment for BMI, whereas BMI showed no significant association with colorectal cancer risk in women or with rectal cancer risk after WHR adjustment.
IN PRACTICE:
“[The study] results also underline the importance of integrating additional anthropometric measures such as WHR alongside BMI into routine clinical practice for more effective prevention and management of obesity, whose prevalence is steadily increasing in many countries worldwide, in order to limit the global burden of colorectal cancer and many other obesity-related adverse health outcomes,” the authors wrote.
SOURCE:
The study was led by Fatemeh Safizadeh, German Cancer Research Center (DKFZ), Heidelberg. It was published online in The International Journal of Obesity.
LIMITATIONS:
This study relied on only one-time measurements of anthropometric measures at baseline, without considering previous lifetime history of overweight and obesity or changes during follow-up. Additionally, WHR and WC may not be the most accurate measures of central obesity, as WC includes both visceral and subcutaneous adipose tissue. The study population also showed evidence of healthy volunteer bias, with more health-conscious and socioeconomically advantaged participants being somewhat overrepresented.
DISCLOSURES:
The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- General obesity, often measured using BMI, is a recognized risk factor for colorectal cancer, but how much of this association is due to central obesity is unclear.
- Researchers assessed the associations between BMI, waist-to-hip ratio (WHR), and waist circumference (WC) with colorectal cancer risk and the degree of independence among these associations in patients aged 40-69 years recruited in the UK Biobank cohort study from 2006 to 2010.
- Anthropometric measurements were performed using standardized methods.
- Cancer registry and hospital data linkage identified colorectal cancer cases in the UK Biobank.
TAKEAWAY:
- Researchers included 460,784 participants (mean age, 56.3 years; 46.7% men), of whom 67.1% had either overweight or obesity, and 49.4% and 60.5% had high or very high WHR and WC, respectively.
- During the median 12.5-year follow-up period, 5977 participants developed colorectal cancer.
- Every SD increase in WHR (hazard ratio [HR], 1.18) showed a stronger association with colorectal cancer risk than in BMI (HR, 1.10).
- After adjustment for BMI, the association between WHR and colorectal cancer risk became slightly attenuated while still staying robust (HR, 1.15); however, after adjusting for WHR, the association between BMI and colorectal cancer risk became substantially weakened (HR, 1.04).
- WHR showed strongly significant associations with colorectal cancer risk across all BMI categories, whereas associations of BMI with colorectal cancer risk were weak and not statistically significant within all WHR categories.
- Central obesity demonstrated consistent associations with both colon and rectal cancer risks in both sexes before and after adjustment for BMI, whereas BMI showed no significant association with colorectal cancer risk in women or with rectal cancer risk after WHR adjustment.
IN PRACTICE:
“[The study] results also underline the importance of integrating additional anthropometric measures such as WHR alongside BMI into routine clinical practice for more effective prevention and management of obesity, whose prevalence is steadily increasing in many countries worldwide, in order to limit the global burden of colorectal cancer and many other obesity-related adverse health outcomes,” the authors wrote.
SOURCE:
The study was led by Fatemeh Safizadeh, German Cancer Research Center (DKFZ), Heidelberg. It was published online in The International Journal of Obesity.
LIMITATIONS:
This study relied on only one-time measurements of anthropometric measures at baseline, without considering previous lifetime history of overweight and obesity or changes during follow-up. Additionally, WHR and WC may not be the most accurate measures of central obesity, as WC includes both visceral and subcutaneous adipose tissue. The study population also showed evidence of healthy volunteer bias, with more health-conscious and socioeconomically advantaged participants being somewhat overrepresented.
DISCLOSURES:
The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- General obesity, often measured using BMI, is a recognized risk factor for colorectal cancer, but how much of this association is due to central obesity is unclear.
- Researchers assessed the associations between BMI, waist-to-hip ratio (WHR), and waist circumference (WC) with colorectal cancer risk and the degree of independence among these associations in patients aged 40-69 years recruited in the UK Biobank cohort study from 2006 to 2010.
- Anthropometric measurements were performed using standardized methods.
- Cancer registry and hospital data linkage identified colorectal cancer cases in the UK Biobank.
TAKEAWAY:
- Researchers included 460,784 participants (mean age, 56.3 years; 46.7% men), of whom 67.1% had either overweight or obesity, and 49.4% and 60.5% had high or very high WHR and WC, respectively.
- During the median 12.5-year follow-up period, 5977 participants developed colorectal cancer.
- Every SD increase in WHR (hazard ratio [HR], 1.18) showed a stronger association with colorectal cancer risk than in BMI (HR, 1.10).
- After adjustment for BMI, the association between WHR and colorectal cancer risk became slightly attenuated while still staying robust (HR, 1.15); however, after adjusting for WHR, the association between BMI and colorectal cancer risk became substantially weakened (HR, 1.04).
- WHR showed strongly significant associations with colorectal cancer risk across all BMI categories, whereas associations of BMI with colorectal cancer risk were weak and not statistically significant within all WHR categories.
- Central obesity demonstrated consistent associations with both colon and rectal cancer risks in both sexes before and after adjustment for BMI, whereas BMI showed no significant association with colorectal cancer risk in women or with rectal cancer risk after WHR adjustment.
IN PRACTICE:
“[The study] results also underline the importance of integrating additional anthropometric measures such as WHR alongside BMI into routine clinical practice for more effective prevention and management of obesity, whose prevalence is steadily increasing in many countries worldwide, in order to limit the global burden of colorectal cancer and many other obesity-related adverse health outcomes,” the authors wrote.
SOURCE:
The study was led by Fatemeh Safizadeh, German Cancer Research Center (DKFZ), Heidelberg. It was published online in The International Journal of Obesity.
LIMITATIONS:
This study relied on only one-time measurements of anthropometric measures at baseline, without considering previous lifetime history of overweight and obesity or changes during follow-up. Additionally, WHR and WC may not be the most accurate measures of central obesity, as WC includes both visceral and subcutaneous adipose tissue. The study population also showed evidence of healthy volunteer bias, with more health-conscious and socioeconomically advantaged participants being somewhat overrepresented.
DISCLOSURES:
The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Adalimumab for Psoriasis: Study Compares Biosimilars Vs. Originator
TOPLINE:
rate than those who remained on Humira.
METHODOLOGY:
- Researchers conducted a cohort study using data on patients with psoriasis who were treated with adalimumab, a tumor necrosis factor alpha inhibitor used to treat moderate to severe psoriasis, from the French National Health Data System, British Association of Dermatologists Biologics and Immunomodulators Register, and Spanish Registry of Systemic Therapy in Psoriasis.
- The analysis included 7387 adalimumab-naive patients who were new users of an adalimumab biosimilar and 3654 patients (switchers) who switched from Humira to a biosimilar. Patients were matched and compared with patients receiving Humira.
- Co-primary outcomes of the study were drug discontinuation and serious adverse events.
- Researchers assessed the following adalimumab biosimilar brands: Amgevita, Imraldi, Hyrimoz, Idacio, and Hulio.
TAKEAWAY:
- All-cause drug discontinuation rates were similar between new users of biosimilars and Humira new users (hazard ratio [HR], 0.99; 95% CI, 0.94-1.04).
- Discontinuation rates were higher among those who switched from Humira to a biosimilar (HR, 1.35; 95% CI, 1.19-1.52) than among those who stayed on Humira. Switching to Amgevita (HR, 1.25; 95% CI, 1.13-1.27), Imraldi (HR, 1.53; 95% CI, 1.33-1.76), and Hyrimoz (HR, 1.80; 95% CI, 1.29-2.52) was associated with higher discontinuation rates.
- Serious adverse events were not significantly different between new users of Humira and biosimilar new users (incidence rate ratio [IRR], 0.91; 95% CI, 0.80-1.05), and between patients who switched from a biosimilar to Humira and those who stayed on Humira (IRR, 0.92; 95% CI, 0.83-1.01).
- No significant differences in discontinuation because of ineffectiveness were found between biosimilar and Humira new users (HR, 0.97; 95% CI, 0.88-1.08). Discontinuation because of adverse events was also comparable for all biosimilars among new users, except for Hyrimoz (HR, 0.54; 95% CI, 0.35-0.85), which showed fewer discontinuations than Humira.
IN PRACTICE:
“This study found comparable drug survival and safety between adalimumab biosimilars and Humira in adalimumab-naive patients, supporting the use of biosimilars as viable alternatives for new patients,” the authors wrote. However, noting that discontinuation was more likely among those who switched from Humira to a biosimilar, they added: “Changes in treatment response, skin or injection site reactions, and nocebo effects may contribute to treatment discontinuation post-switch. Thus, patients who switch from Humira to biosimilars may require closer monitoring and support to alleviate these challenges.”
SOURCE:
The study was led by Duc Binh Phan, Dermatology Centre, Northern Care Alliance NHS Foundation Trust in Manchester, England. It was published online in The British Journal of Dermatology.
LIMITATIONS:
Unmeasured factors including psychological perceptions, regional policies, and drug availability could influence drug survival, making the results not fully reflective of treatment effectiveness or safety. Most Humira users in registries were enrolled before biosimilars became available, making it impractical to match new users on the basis of treatment initiation years. Additionally, reasons for discontinuation were not available in the French National Health Data System.
DISCLOSURES:
In the United Kingdom, the research was funded by the Psoriasis Association PhD studentship and supported by the NIHR Manchester Biomedical Research Centre. In France, the authors are employees of the French National Health Insurance, the French National Agency for the Safety of Medicines and Health Products, and the Assistance Publique — Hôpitaux de Paris and received no funding. The authors reported receiving consulting and speaker fees and clinical trial sponsorship from various pharmaceutical companies. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
rate than those who remained on Humira.
METHODOLOGY:
- Researchers conducted a cohort study using data on patients with psoriasis who were treated with adalimumab, a tumor necrosis factor alpha inhibitor used to treat moderate to severe psoriasis, from the French National Health Data System, British Association of Dermatologists Biologics and Immunomodulators Register, and Spanish Registry of Systemic Therapy in Psoriasis.
- The analysis included 7387 adalimumab-naive patients who were new users of an adalimumab biosimilar and 3654 patients (switchers) who switched from Humira to a biosimilar. Patients were matched and compared with patients receiving Humira.
- Co-primary outcomes of the study were drug discontinuation and serious adverse events.
- Researchers assessed the following adalimumab biosimilar brands: Amgevita, Imraldi, Hyrimoz, Idacio, and Hulio.
TAKEAWAY:
- All-cause drug discontinuation rates were similar between new users of biosimilars and Humira new users (hazard ratio [HR], 0.99; 95% CI, 0.94-1.04).
- Discontinuation rates were higher among those who switched from Humira to a biosimilar (HR, 1.35; 95% CI, 1.19-1.52) than among those who stayed on Humira. Switching to Amgevita (HR, 1.25; 95% CI, 1.13-1.27), Imraldi (HR, 1.53; 95% CI, 1.33-1.76), and Hyrimoz (HR, 1.80; 95% CI, 1.29-2.52) was associated with higher discontinuation rates.
- Serious adverse events were not significantly different between new users of Humira and biosimilar new users (incidence rate ratio [IRR], 0.91; 95% CI, 0.80-1.05), and between patients who switched from a biosimilar to Humira and those who stayed on Humira (IRR, 0.92; 95% CI, 0.83-1.01).
- No significant differences in discontinuation because of ineffectiveness were found between biosimilar and Humira new users (HR, 0.97; 95% CI, 0.88-1.08). Discontinuation because of adverse events was also comparable for all biosimilars among new users, except for Hyrimoz (HR, 0.54; 95% CI, 0.35-0.85), which showed fewer discontinuations than Humira.
IN PRACTICE:
“This study found comparable drug survival and safety between adalimumab biosimilars and Humira in adalimumab-naive patients, supporting the use of biosimilars as viable alternatives for new patients,” the authors wrote. However, noting that discontinuation was more likely among those who switched from Humira to a biosimilar, they added: “Changes in treatment response, skin or injection site reactions, and nocebo effects may contribute to treatment discontinuation post-switch. Thus, patients who switch from Humira to biosimilars may require closer monitoring and support to alleviate these challenges.”
SOURCE:
The study was led by Duc Binh Phan, Dermatology Centre, Northern Care Alliance NHS Foundation Trust in Manchester, England. It was published online in The British Journal of Dermatology.
LIMITATIONS:
Unmeasured factors including psychological perceptions, regional policies, and drug availability could influence drug survival, making the results not fully reflective of treatment effectiveness or safety. Most Humira users in registries were enrolled before biosimilars became available, making it impractical to match new users on the basis of treatment initiation years. Additionally, reasons for discontinuation were not available in the French National Health Data System.
DISCLOSURES:
In the United Kingdom, the research was funded by the Psoriasis Association PhD studentship and supported by the NIHR Manchester Biomedical Research Centre. In France, the authors are employees of the French National Health Insurance, the French National Agency for the Safety of Medicines and Health Products, and the Assistance Publique — Hôpitaux de Paris and received no funding. The authors reported receiving consulting and speaker fees and clinical trial sponsorship from various pharmaceutical companies. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
rate than those who remained on Humira.
METHODOLOGY:
- Researchers conducted a cohort study using data on patients with psoriasis who were treated with adalimumab, a tumor necrosis factor alpha inhibitor used to treat moderate to severe psoriasis, from the French National Health Data System, British Association of Dermatologists Biologics and Immunomodulators Register, and Spanish Registry of Systemic Therapy in Psoriasis.
- The analysis included 7387 adalimumab-naive patients who were new users of an adalimumab biosimilar and 3654 patients (switchers) who switched from Humira to a biosimilar. Patients were matched and compared with patients receiving Humira.
- Co-primary outcomes of the study were drug discontinuation and serious adverse events.
- Researchers assessed the following adalimumab biosimilar brands: Amgevita, Imraldi, Hyrimoz, Idacio, and Hulio.
TAKEAWAY:
- All-cause drug discontinuation rates were similar between new users of biosimilars and Humira new users (hazard ratio [HR], 0.99; 95% CI, 0.94-1.04).
- Discontinuation rates were higher among those who switched from Humira to a biosimilar (HR, 1.35; 95% CI, 1.19-1.52) than among those who stayed on Humira. Switching to Amgevita (HR, 1.25; 95% CI, 1.13-1.27), Imraldi (HR, 1.53; 95% CI, 1.33-1.76), and Hyrimoz (HR, 1.80; 95% CI, 1.29-2.52) was associated with higher discontinuation rates.
- Serious adverse events were not significantly different between new users of Humira and biosimilar new users (incidence rate ratio [IRR], 0.91; 95% CI, 0.80-1.05), and between patients who switched from a biosimilar to Humira and those who stayed on Humira (IRR, 0.92; 95% CI, 0.83-1.01).
- No significant differences in discontinuation because of ineffectiveness were found between biosimilar and Humira new users (HR, 0.97; 95% CI, 0.88-1.08). Discontinuation because of adverse events was also comparable for all biosimilars among new users, except for Hyrimoz (HR, 0.54; 95% CI, 0.35-0.85), which showed fewer discontinuations than Humira.
IN PRACTICE:
“This study found comparable drug survival and safety between adalimumab biosimilars and Humira in adalimumab-naive patients, supporting the use of biosimilars as viable alternatives for new patients,” the authors wrote. However, noting that discontinuation was more likely among those who switched from Humira to a biosimilar, they added: “Changes in treatment response, skin or injection site reactions, and nocebo effects may contribute to treatment discontinuation post-switch. Thus, patients who switch from Humira to biosimilars may require closer monitoring and support to alleviate these challenges.”
SOURCE:
The study was led by Duc Binh Phan, Dermatology Centre, Northern Care Alliance NHS Foundation Trust in Manchester, England. It was published online in The British Journal of Dermatology.
LIMITATIONS:
Unmeasured factors including psychological perceptions, regional policies, and drug availability could influence drug survival, making the results not fully reflective of treatment effectiveness or safety. Most Humira users in registries were enrolled before biosimilars became available, making it impractical to match new users on the basis of treatment initiation years. Additionally, reasons for discontinuation were not available in the French National Health Data System.
DISCLOSURES:
In the United Kingdom, the research was funded by the Psoriasis Association PhD studentship and supported by the NIHR Manchester Biomedical Research Centre. In France, the authors are employees of the French National Health Insurance, the French National Agency for the Safety of Medicines and Health Products, and the Assistance Publique — Hôpitaux de Paris and received no funding. The authors reported receiving consulting and speaker fees and clinical trial sponsorship from various pharmaceutical companies. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Treating Onychomycosis: Pearls from a Podiatrist
LAS VEGAS —
According to Tracey C. Vlahovic, DPM, a professor at the Samuel Merritt University College of Podiatric Medicine, Oakland, California, most cases of onychomycosis are caused by the dermatophytes Trichophyton rubrum and T mentagrophytes, although the cause can also be a mixed infection. “Dermatophytes are going to impact the nails first, and molds may come in and join the party later,” she said at the Society of Dermatology Physician Associates (SDPA) 22nd Annual Fall Dermatology Conference.
“The distal subungual onychomycosis (DSO) type is still the most common, but don’t forget that onychomycosis and nail psoriasis can happen at the same time. What we can’t lose sight of is that onychomycosis is a disease of the nail bed, which ultimately affects the nail plate; it’s not a disease of the nail plate first.”
Her diagnostic approach combines periodic acid-Schiff (PAS) staining with fungal culture “because I like to know the speciation,” she said. “PAS doesn’t give me the speciation; fungal cultures should. PCR can be expensive, but that can give me speciation.”
How Does This Happen?
Fungal DSO occurs because of exposure to a dermatophyte, which can be as simple as tinea pedis. “Perhaps it’s the environment in the shoe,” said Vlahovic, one of the authors of a textbook on onychomycosis. “That’s something I’m always concentrating on with the patient. What is your foot hygiene like? What’s your shoe and sock wear? What’s your level of physical activity? You can have trauma to the hyponychium, where the skin and the nail meet. Maybe they trim their nails too close to the skin, or maybe there’s another skin condition like psoriasis.”
The dermatophyte, she continued, enters and invades the nail at the hyponychium and uses the keratinase enzyme to digest keratin in the nail bed. Mild inflammation develops, and pH changes cause focal parakeratosis and subungual hyperkeratosis in the form of onycholysis and subungual debris. “Hyphae then invade the lamina of the nail plate, which causes brittle nails,” she said. “The compromised hyponychium creates a reservoir for molds and bacteria.”
Therapies approved by the Food and Drug Administration (FDA) for onychomycosis include the topical agents efinaconazole, tavaborole, and ciclopirox; the oral agents terbinafine and itraconazole; and laser therapy. Off-label, Vlahovic said that she sometimes uses oral fluconazole, pulsed dosing for terbinafine, and booster doses of terbinafine or any approved oral antifungal agent. Pulse dosing for itraconazole is FDA-approved for fingernails but not for toenails.
“We don’t have any oral antifungals that are approved for children, but we do have weight-based dosing,” she noted. Other off-label treatments for onychomycosis that patients may come across while browsing the internet but do not penetrate the nail plate, include products containing tolnaftate, tree oil, and undecylenic acid, “which is a very long-chain antifungal,” Vlahovic said. “It’s so huge that it can’t get through the nail plate. These products must get through the nail plate into the nail bed where the infection is.”
According to therapeutic recommendations for the treatment of toenail onychomycosis in the United States, published in 2021, terbinafine is the primary choice for oral treatment and efinaconazole 10% for topical treatment. There are no current treatment recommendations for pregnant or lactating patients. “I always defer to the obstetrician,” said Vlahovic, a coauthor of the recommendations. For pediatric patients, there are approved topical medications: Efinaconazole and tavaborole for ages 6 and up and ciclopirox for ages 12 years or older.
Treatment recommendations for adults vary based on clinical presentation and patient characteristics. Questions to consider: Are they older? Do they have diabetes? Are they able to reach their feet to apply medication? What other medications are they taking? Are there any kidney or liver issues that are cause for concern?
Another question to consider is whether they have concurrent nail psoriasis. “When I have those patients, I often treat the onychomycosis first and the nail psoriasis second,” she said.
Evidence for Lasers Weak
Though laser therapy is FDA approved for the temporary increase of clear nails in onychomycosis, Vlahovic is underwhelmed by the evidence of its use for onychomycosis. According to a systematic review of 261 studies, only 1 reported treatment success as 16.7%, and clinical cures ranged from 13% to 16%. “Many of the existing studies were so poorly done in terms of protocols; it was frustrating,” she said. “No study has reported complete cure. There’s a lack of standardization across laser companies and a lack of standardization across protocols.”
Before starting oral antifungal therapy, Vlahovic uses the Onychomycosis Severity Index to determine the number of nails involved and the proportion of nails that are affected. She also wants to know if the patient is taking any medication that might interfere with an oral antifungal and gets baseline liver function tests (LFTs) to document results in the chart. “You want to discuss the pros and cons of oral antifungal therapy, and you want to set realistic expectations,” she added. “These medications are not cosmetic products; they are meant to kill fungus. Sometimes patients lose sight of that.”
Vlahovic routinely offers pulse dosing of terbinafine, which is FDA approved at a dose of 250 mg/d for 90 days. Pulse dosing involves taking terbinafine 250 mg twice a day for 1 week, followed by a 3-week break. This cycle is repeated three or four times. A clinical trial found no significant difference in outcome between patients who received pulsed vs continuous terbinafine dosing for the treatment of dermatophyte onychomycosis.
What About Oral Antifungal Safety?
For patients who ask about the safety of oral antifungals, Vlahovic characterized them as “well tolerated and safe in an immunocompetent population.” In a meta-analysis of 122 studies of about 22,000 patients, the pooled risk for treatment discontinuation because of adverse events was 3.4% for terbinafine 250 mg/d and 4.21% for itraconazole 200 mg/d. The risk for liver injury requiring termination of treatment and the risk of having symptomatic elevation of LFTs were less than 2% for all regimens.
According to the best available published evidence, Vlahovic said, the onychomycosis recurrence rate ranges from 6% to 40%. “That’s a wild number. We really have no idea what the true recurrence rate is, and that’s a problem.”
Vlahovic disclosed having been a consultant to and an investigator for Ortho Dermatologics and Sagis Diagnostics.
A version of this article appeared on Medscape.com.
LAS VEGAS —
According to Tracey C. Vlahovic, DPM, a professor at the Samuel Merritt University College of Podiatric Medicine, Oakland, California, most cases of onychomycosis are caused by the dermatophytes Trichophyton rubrum and T mentagrophytes, although the cause can also be a mixed infection. “Dermatophytes are going to impact the nails first, and molds may come in and join the party later,” she said at the Society of Dermatology Physician Associates (SDPA) 22nd Annual Fall Dermatology Conference.
“The distal subungual onychomycosis (DSO) type is still the most common, but don’t forget that onychomycosis and nail psoriasis can happen at the same time. What we can’t lose sight of is that onychomycosis is a disease of the nail bed, which ultimately affects the nail plate; it’s not a disease of the nail plate first.”
Her diagnostic approach combines periodic acid-Schiff (PAS) staining with fungal culture “because I like to know the speciation,” she said. “PAS doesn’t give me the speciation; fungal cultures should. PCR can be expensive, but that can give me speciation.”
How Does This Happen?
Fungal DSO occurs because of exposure to a dermatophyte, which can be as simple as tinea pedis. “Perhaps it’s the environment in the shoe,” said Vlahovic, one of the authors of a textbook on onychomycosis. “That’s something I’m always concentrating on with the patient. What is your foot hygiene like? What’s your shoe and sock wear? What’s your level of physical activity? You can have trauma to the hyponychium, where the skin and the nail meet. Maybe they trim their nails too close to the skin, or maybe there’s another skin condition like psoriasis.”
The dermatophyte, she continued, enters and invades the nail at the hyponychium and uses the keratinase enzyme to digest keratin in the nail bed. Mild inflammation develops, and pH changes cause focal parakeratosis and subungual hyperkeratosis in the form of onycholysis and subungual debris. “Hyphae then invade the lamina of the nail plate, which causes brittle nails,” she said. “The compromised hyponychium creates a reservoir for molds and bacteria.”
Therapies approved by the Food and Drug Administration (FDA) for onychomycosis include the topical agents efinaconazole, tavaborole, and ciclopirox; the oral agents terbinafine and itraconazole; and laser therapy. Off-label, Vlahovic said that she sometimes uses oral fluconazole, pulsed dosing for terbinafine, and booster doses of terbinafine or any approved oral antifungal agent. Pulse dosing for itraconazole is FDA-approved for fingernails but not for toenails.
“We don’t have any oral antifungals that are approved for children, but we do have weight-based dosing,” she noted. Other off-label treatments for onychomycosis that patients may come across while browsing the internet but do not penetrate the nail plate, include products containing tolnaftate, tree oil, and undecylenic acid, “which is a very long-chain antifungal,” Vlahovic said. “It’s so huge that it can’t get through the nail plate. These products must get through the nail plate into the nail bed where the infection is.”
According to therapeutic recommendations for the treatment of toenail onychomycosis in the United States, published in 2021, terbinafine is the primary choice for oral treatment and efinaconazole 10% for topical treatment. There are no current treatment recommendations for pregnant or lactating patients. “I always defer to the obstetrician,” said Vlahovic, a coauthor of the recommendations. For pediatric patients, there are approved topical medications: Efinaconazole and tavaborole for ages 6 and up and ciclopirox for ages 12 years or older.
Treatment recommendations for adults vary based on clinical presentation and patient characteristics. Questions to consider: Are they older? Do they have diabetes? Are they able to reach their feet to apply medication? What other medications are they taking? Are there any kidney or liver issues that are cause for concern?
Another question to consider is whether they have concurrent nail psoriasis. “When I have those patients, I often treat the onychomycosis first and the nail psoriasis second,” she said.
Evidence for Lasers Weak
Though laser therapy is FDA approved for the temporary increase of clear nails in onychomycosis, Vlahovic is underwhelmed by the evidence of its use for onychomycosis. According to a systematic review of 261 studies, only 1 reported treatment success as 16.7%, and clinical cures ranged from 13% to 16%. “Many of the existing studies were so poorly done in terms of protocols; it was frustrating,” she said. “No study has reported complete cure. There’s a lack of standardization across laser companies and a lack of standardization across protocols.”
Before starting oral antifungal therapy, Vlahovic uses the Onychomycosis Severity Index to determine the number of nails involved and the proportion of nails that are affected. She also wants to know if the patient is taking any medication that might interfere with an oral antifungal and gets baseline liver function tests (LFTs) to document results in the chart. “You want to discuss the pros and cons of oral antifungal therapy, and you want to set realistic expectations,” she added. “These medications are not cosmetic products; they are meant to kill fungus. Sometimes patients lose sight of that.”
Vlahovic routinely offers pulse dosing of terbinafine, which is FDA approved at a dose of 250 mg/d for 90 days. Pulse dosing involves taking terbinafine 250 mg twice a day for 1 week, followed by a 3-week break. This cycle is repeated three or four times. A clinical trial found no significant difference in outcome between patients who received pulsed vs continuous terbinafine dosing for the treatment of dermatophyte onychomycosis.
What About Oral Antifungal Safety?
For patients who ask about the safety of oral antifungals, Vlahovic characterized them as “well tolerated and safe in an immunocompetent population.” In a meta-analysis of 122 studies of about 22,000 patients, the pooled risk for treatment discontinuation because of adverse events was 3.4% for terbinafine 250 mg/d and 4.21% for itraconazole 200 mg/d. The risk for liver injury requiring termination of treatment and the risk of having symptomatic elevation of LFTs were less than 2% for all regimens.
According to the best available published evidence, Vlahovic said, the onychomycosis recurrence rate ranges from 6% to 40%. “That’s a wild number. We really have no idea what the true recurrence rate is, and that’s a problem.”
Vlahovic disclosed having been a consultant to and an investigator for Ortho Dermatologics and Sagis Diagnostics.
A version of this article appeared on Medscape.com.
LAS VEGAS —
According to Tracey C. Vlahovic, DPM, a professor at the Samuel Merritt University College of Podiatric Medicine, Oakland, California, most cases of onychomycosis are caused by the dermatophytes Trichophyton rubrum and T mentagrophytes, although the cause can also be a mixed infection. “Dermatophytes are going to impact the nails first, and molds may come in and join the party later,” she said at the Society of Dermatology Physician Associates (SDPA) 22nd Annual Fall Dermatology Conference.
“The distal subungual onychomycosis (DSO) type is still the most common, but don’t forget that onychomycosis and nail psoriasis can happen at the same time. What we can’t lose sight of is that onychomycosis is a disease of the nail bed, which ultimately affects the nail plate; it’s not a disease of the nail plate first.”
Her diagnostic approach combines periodic acid-Schiff (PAS) staining with fungal culture “because I like to know the speciation,” she said. “PAS doesn’t give me the speciation; fungal cultures should. PCR can be expensive, but that can give me speciation.”
How Does This Happen?
Fungal DSO occurs because of exposure to a dermatophyte, which can be as simple as tinea pedis. “Perhaps it’s the environment in the shoe,” said Vlahovic, one of the authors of a textbook on onychomycosis. “That’s something I’m always concentrating on with the patient. What is your foot hygiene like? What’s your shoe and sock wear? What’s your level of physical activity? You can have trauma to the hyponychium, where the skin and the nail meet. Maybe they trim their nails too close to the skin, or maybe there’s another skin condition like psoriasis.”
The dermatophyte, she continued, enters and invades the nail at the hyponychium and uses the keratinase enzyme to digest keratin in the nail bed. Mild inflammation develops, and pH changes cause focal parakeratosis and subungual hyperkeratosis in the form of onycholysis and subungual debris. “Hyphae then invade the lamina of the nail plate, which causes brittle nails,” she said. “The compromised hyponychium creates a reservoir for molds and bacteria.”
Therapies approved by the Food and Drug Administration (FDA) for onychomycosis include the topical agents efinaconazole, tavaborole, and ciclopirox; the oral agents terbinafine and itraconazole; and laser therapy. Off-label, Vlahovic said that she sometimes uses oral fluconazole, pulsed dosing for terbinafine, and booster doses of terbinafine or any approved oral antifungal agent. Pulse dosing for itraconazole is FDA-approved for fingernails but not for toenails.
“We don’t have any oral antifungals that are approved for children, but we do have weight-based dosing,” she noted. Other off-label treatments for onychomycosis that patients may come across while browsing the internet but do not penetrate the nail plate, include products containing tolnaftate, tree oil, and undecylenic acid, “which is a very long-chain antifungal,” Vlahovic said. “It’s so huge that it can’t get through the nail plate. These products must get through the nail plate into the nail bed where the infection is.”
According to therapeutic recommendations for the treatment of toenail onychomycosis in the United States, published in 2021, terbinafine is the primary choice for oral treatment and efinaconazole 10% for topical treatment. There are no current treatment recommendations for pregnant or lactating patients. “I always defer to the obstetrician,” said Vlahovic, a coauthor of the recommendations. For pediatric patients, there are approved topical medications: Efinaconazole and tavaborole for ages 6 and up and ciclopirox for ages 12 years or older.
Treatment recommendations for adults vary based on clinical presentation and patient characteristics. Questions to consider: Are they older? Do they have diabetes? Are they able to reach their feet to apply medication? What other medications are they taking? Are there any kidney or liver issues that are cause for concern?
Another question to consider is whether they have concurrent nail psoriasis. “When I have those patients, I often treat the onychomycosis first and the nail psoriasis second,” she said.
Evidence for Lasers Weak
Though laser therapy is FDA approved for the temporary increase of clear nails in onychomycosis, Vlahovic is underwhelmed by the evidence of its use for onychomycosis. According to a systematic review of 261 studies, only 1 reported treatment success as 16.7%, and clinical cures ranged from 13% to 16%. “Many of the existing studies were so poorly done in terms of protocols; it was frustrating,” she said. “No study has reported complete cure. There’s a lack of standardization across laser companies and a lack of standardization across protocols.”
Before starting oral antifungal therapy, Vlahovic uses the Onychomycosis Severity Index to determine the number of nails involved and the proportion of nails that are affected. She also wants to know if the patient is taking any medication that might interfere with an oral antifungal and gets baseline liver function tests (LFTs) to document results in the chart. “You want to discuss the pros and cons of oral antifungal therapy, and you want to set realistic expectations,” she added. “These medications are not cosmetic products; they are meant to kill fungus. Sometimes patients lose sight of that.”
Vlahovic routinely offers pulse dosing of terbinafine, which is FDA approved at a dose of 250 mg/d for 90 days. Pulse dosing involves taking terbinafine 250 mg twice a day for 1 week, followed by a 3-week break. This cycle is repeated three or four times. A clinical trial found no significant difference in outcome between patients who received pulsed vs continuous terbinafine dosing for the treatment of dermatophyte onychomycosis.
What About Oral Antifungal Safety?
For patients who ask about the safety of oral antifungals, Vlahovic characterized them as “well tolerated and safe in an immunocompetent population.” In a meta-analysis of 122 studies of about 22,000 patients, the pooled risk for treatment discontinuation because of adverse events was 3.4% for terbinafine 250 mg/d and 4.21% for itraconazole 200 mg/d. The risk for liver injury requiring termination of treatment and the risk of having symptomatic elevation of LFTs were less than 2% for all regimens.
According to the best available published evidence, Vlahovic said, the onychomycosis recurrence rate ranges from 6% to 40%. “That’s a wild number. We really have no idea what the true recurrence rate is, and that’s a problem.”
Vlahovic disclosed having been a consultant to and an investigator for Ortho Dermatologics and Sagis Diagnostics.
A version of this article appeared on Medscape.com.
FROM SDPA 2024
Study Finds Different Survival Rates for Hidradenitis Suppurativa Treatments in Children
results from a small single-center study showed.
A previous study found that overall drug survival of adalimumab and infliximab in adults with HS at 12 and 24 months was 56.3% and 30.5%, and 58.3% and 48.6%, respectively. “They also found that older age, longer disease duration, higher body mass index (BMI), and surgery during treatment are associated with increased drug survival,” Robyn Guo, a third-year medical student at Duke University, Durham, North Carolina, told this news organization following the annual Symposium on Hidradenitis Suppurativa Advances, where the study was presented during an oral abstract session. “To our knowledge, the drug survival of biologic therapies in pediatric HS patients has not been previously investigated.”
Adalimumab and infliximab are tumor necrosis factor blockers approved for multiple indications; adalimumab is approved for treating moderate to severe HS in patients aged 12 years or older. Infliximab is not approved for HS but is used to treat the disease.
To determine the drug survival of adalimumab and infliximab in pediatric patients with HS and whether patient comorbidities and HS lesion location are associated with length of biologic survival in pediatric patients with HS, Guo and colleagues used Kaplan-Meier survival curves to calculate biologic survival at 12 and 24 months following biologic initiation and Cox proportional hazards regression to analyze potential factors associated with biologic survival. The study population included 49 pediatric patients in the adalimumab cohort and 11 in the infliximab cohort.
The researchers found that drug survival for adalimumab was 90.6% at 12 months (95% CI, 83.0%-98.8%) and 78.3% at 24 months (95% CI, 67.7%-90.6%), while drug survival for infliximab was 54.5% at 12 months (95% CI, 31.8%-93.6%) and 36.4% at 24 months, an overall difference that reached statistical significance (P = .0009). “Our data suggests that adalimumab survival is significantly higher than infliximab survival in pediatric HS patients,” Guo said.
On univariate Cox regression analysis, gluteal HS lesions were associated with shorter adalimumab survival, and obesity was associated with longer infliximab survival.
The researchers acknowledged certain limitations of their study, including the small sample size and that unadjusted Cox regression analysis did not account for baseline HS severity, biologic therapy dosing, and concomitant medication use. Also, there were patients in both cohorts who were not biologic-naive: Two in the adalimumab cohort were previously treated with infliximab, and five patients in the infliximab cohort were previously treated with adalimumab.
“We plan on conducting further analysis using adjusted Cox regression analysis to account for baseline disease severity measured by Hurley stage, BMI, medication dosing, and concomitant medication use,” Guo said.
The researchers reported having no financial disclosures.
A version of this article appeared on Medscape.com.
results from a small single-center study showed.
A previous study found that overall drug survival of adalimumab and infliximab in adults with HS at 12 and 24 months was 56.3% and 30.5%, and 58.3% and 48.6%, respectively. “They also found that older age, longer disease duration, higher body mass index (BMI), and surgery during treatment are associated with increased drug survival,” Robyn Guo, a third-year medical student at Duke University, Durham, North Carolina, told this news organization following the annual Symposium on Hidradenitis Suppurativa Advances, where the study was presented during an oral abstract session. “To our knowledge, the drug survival of biologic therapies in pediatric HS patients has not been previously investigated.”
Adalimumab and infliximab are tumor necrosis factor blockers approved for multiple indications; adalimumab is approved for treating moderate to severe HS in patients aged 12 years or older. Infliximab is not approved for HS but is used to treat the disease.
To determine the drug survival of adalimumab and infliximab in pediatric patients with HS and whether patient comorbidities and HS lesion location are associated with length of biologic survival in pediatric patients with HS, Guo and colleagues used Kaplan-Meier survival curves to calculate biologic survival at 12 and 24 months following biologic initiation and Cox proportional hazards regression to analyze potential factors associated with biologic survival. The study population included 49 pediatric patients in the adalimumab cohort and 11 in the infliximab cohort.
The researchers found that drug survival for adalimumab was 90.6% at 12 months (95% CI, 83.0%-98.8%) and 78.3% at 24 months (95% CI, 67.7%-90.6%), while drug survival for infliximab was 54.5% at 12 months (95% CI, 31.8%-93.6%) and 36.4% at 24 months, an overall difference that reached statistical significance (P = .0009). “Our data suggests that adalimumab survival is significantly higher than infliximab survival in pediatric HS patients,” Guo said.
On univariate Cox regression analysis, gluteal HS lesions were associated with shorter adalimumab survival, and obesity was associated with longer infliximab survival.
The researchers acknowledged certain limitations of their study, including the small sample size and that unadjusted Cox regression analysis did not account for baseline HS severity, biologic therapy dosing, and concomitant medication use. Also, there were patients in both cohorts who were not biologic-naive: Two in the adalimumab cohort were previously treated with infliximab, and five patients in the infliximab cohort were previously treated with adalimumab.
“We plan on conducting further analysis using adjusted Cox regression analysis to account for baseline disease severity measured by Hurley stage, BMI, medication dosing, and concomitant medication use,” Guo said.
The researchers reported having no financial disclosures.
A version of this article appeared on Medscape.com.
results from a small single-center study showed.
A previous study found that overall drug survival of adalimumab and infliximab in adults with HS at 12 and 24 months was 56.3% and 30.5%, and 58.3% and 48.6%, respectively. “They also found that older age, longer disease duration, higher body mass index (BMI), and surgery during treatment are associated with increased drug survival,” Robyn Guo, a third-year medical student at Duke University, Durham, North Carolina, told this news organization following the annual Symposium on Hidradenitis Suppurativa Advances, where the study was presented during an oral abstract session. “To our knowledge, the drug survival of biologic therapies in pediatric HS patients has not been previously investigated.”
Adalimumab and infliximab are tumor necrosis factor blockers approved for multiple indications; adalimumab is approved for treating moderate to severe HS in patients aged 12 years or older. Infliximab is not approved for HS but is used to treat the disease.
To determine the drug survival of adalimumab and infliximab in pediatric patients with HS and whether patient comorbidities and HS lesion location are associated with length of biologic survival in pediatric patients with HS, Guo and colleagues used Kaplan-Meier survival curves to calculate biologic survival at 12 and 24 months following biologic initiation and Cox proportional hazards regression to analyze potential factors associated with biologic survival. The study population included 49 pediatric patients in the adalimumab cohort and 11 in the infliximab cohort.
The researchers found that drug survival for adalimumab was 90.6% at 12 months (95% CI, 83.0%-98.8%) and 78.3% at 24 months (95% CI, 67.7%-90.6%), while drug survival for infliximab was 54.5% at 12 months (95% CI, 31.8%-93.6%) and 36.4% at 24 months, an overall difference that reached statistical significance (P = .0009). “Our data suggests that adalimumab survival is significantly higher than infliximab survival in pediatric HS patients,” Guo said.
On univariate Cox regression analysis, gluteal HS lesions were associated with shorter adalimumab survival, and obesity was associated with longer infliximab survival.
The researchers acknowledged certain limitations of their study, including the small sample size and that unadjusted Cox regression analysis did not account for baseline HS severity, biologic therapy dosing, and concomitant medication use. Also, there were patients in both cohorts who were not biologic-naive: Two in the adalimumab cohort were previously treated with infliximab, and five patients in the infliximab cohort were previously treated with adalimumab.
“We plan on conducting further analysis using adjusted Cox regression analysis to account for baseline disease severity measured by Hurley stage, BMI, medication dosing, and concomitant medication use,” Guo said.
The researchers reported having no financial disclosures.
A version of this article appeared on Medscape.com.
FROM SDPA 24
Varicella Outbreaks: 2022-2024
Practitioners providing care to children are familiar with the childhood immunization schedule and routinely administer varicella vaccine at the 12-month and 4- to 5-year visits. However, when is the last time most of us or any of the current trainees have seen a case?
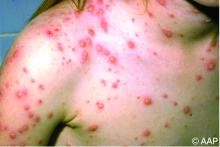
Briefly, varicella is a highly contagious disease caused by varicella-zoster virus (VZV). It is characterized by a generalized pruritic erythematous rash in various stages of development beginning as macules, progressing to papules, and ultimately becoming vesicular lesions on an erythematous base (“dewdrop on a rose petal”) and resolves with crusting of the lesion (Figure 1). It has an incubation period of 10-21 days with symptoms usually developing within 14-16 days after exposure. The vesicular rash must be differentiated from enterovirus, Staphylococcus aureus, contact dermatitis, or insect bites, which initially may be difficult. Approximately 50% of children can have symptoms including fever, malaise, anorexia, headache, and occasionally, mild abdominal pain in the 24-48 hours prior to the appearance of rash. Lesions usually first appear on the scalp, face, or trunk in successive crops over several days. A person with varicella has lesions in various stages.
In a normal host, new vesicle formation usually stops within 4 days, and most lesions have fully crusted by day 6. VZV establishes latency in sensory ganglia and may reactivate years or decades later to cause herpes zoster (HZ). Most healthy children with varicella recover without sequelae so the disease is generally regarded as benign. However, varicella can lead to serious complications and deaths in healthy as well as immunocompromised persons.
Complications of Varicella: bacterial superinfection of skin lesions most often with Streptococcus pyogenes or S aureus manifested as cellulitis, myositis, or necrotizing fasciitis; neurologic complications include cerebellar ataxia and encephalitis with the latter seen most often in adults. Pneumonia occurs most often in adults, especially those infected during pregnancy. Another concern, infection during the first 20 weeks of pregnancy can lead to fetal death or severe birth defects, including limb hypoplasia, cutaneous scarring, ocular abnormalities, and central nervous system damage (congenital varicella syndrome).
The risk for development of severe disseminated disease was first noted in the 1960s as treatments for leukemia in children improved. They were surviving their cancer only to develop severe and often fatal varicella. Today it is recognized that development of disseminated disease is a risk for all infected persons with impaired T cell function, malignancies, HIV, or receiving immunosuppressive therapy.
Reye’s syndrome is rarely seen today since taking salicylates while infected with VZV was identified as a predisposing factor for development.
VZV is only found in humans and transmission is person to person or airborne. The secondary household attack rate is approximately 90%. In contrast, the secondary attack rates in classrooms may be as low as 12%-33%. Transmission rates in the tropics for unexplained reasons are also lower.
Vaccine History: Why do we rarely see this disease anymore? Varicella, a live attenuated vaccine, was developed in 1974 by Dr. Michiaki Takahashi. It remains the only vaccine directed against a herpes group virus. In 1979, the Collaborative Varicella Vaccine Study Group was established at the National Institutes of Health (NIH) and additional safety and efficacy trials were conducted in the United States initially in leukemic patients in remission and later in healthy children, which supported Takahashi’s data. Licensure of varicella vaccine was granted in 1995. That same year, due to continuing disease and societal burden, the United States was the first country to incorporate varicella into the routine childhood immunization schedule, which resulted in significant reductions in cases. To further improve control of varicella, in 2007 vaccine recommendations were revised and a routine two-dose schedule was implemented. The impact of varicella disease pre- and post-vaccine licensure is illustrated in Figure 2. Not listed, is that in the pre-vaccine era, there were approximately 44 cases of congenital varicella syndrome annually.

As of 2023 only 23% (45/195) of nations routinely administer this vaccine and 4% (8/195) have restricted recommendations. The remaining 73% of countries do not offer the vaccine, including all countries on the African continent, and Cuba, Guatemala, Haiti, Honduras, India, Jordan, Lebanon, Philippines, Portugal, and Venezuela to list a few.
Varicella Outbreak: In October 2022, New York City (NYC) identified a varicella outbreak primarily involving persons who recently migrated from Central and South America and lived in a shelter in NYC or residential facility (n = 105); the outbreak is ongoing. As of March 8, 2024, 873 cases (53%) were among children aged 4-18 years and 91.9% had no documentation of varicella vaccine at time of symptom onset. There were 28 hospitalizations, and no deaths reported. The most common sources of transmission were the residential facilities (41.3%) and importation or possible importation (39.4%). School transmission accounted for only 1.2% of cases.
Most migrants arrived from countries where varicella vaccination is not part of the routine childhood immunization schedule. Although most cases occurred in children, almost 30% occurred in adults. Many of the migrants arrived from tropical countries where susceptibility rates are also higher in adults. This outbreak is a reminder of the importance of limiting disease transmission by maintaining high vaccination rates. To curtail this outbreak, approximately 27,000 doses of varicella vaccine were administered to the arriving migrants. In addition, MMR, COVID-19, influenza, and all routine pediatric vaccines required for school entry were administered. Temporary closure of the residential facilities were required. Education was provided to residents regarding immunizations as well as assistance to help them establish a primary care home. Multiple agencies were mobilized to successfully coordinate these efforts.
Take Home Message
1. Each country has its own routine immunization schedule. It may not include all vaccines recommended in the US schedule. When questioned I’m frequently told that immunizations are up to date, only to review records and find they are not, especially when it is related to MMR. It is often administered at 9 months and/or MR or MM is administered depending on the country. As reported here, varicella is a routine vaccine in only 45 countries.
2.
3. Once an outbreak has been identified, the infrastructure to manage and contain it must already be established. In most instances there will be a need for a rapid and often large-scale effort involving multiple agencies including local health care providers.
4. Not all diseases are reportable. Only deaths by varicella are nationally notifiable. Otherwise, cases are reported voluntarily. As of November 2, 2024, there have been 5,157 cases of varicella reported, excluding any cases from NYC.
Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
CDC. Nationally Notifiable Infectious Diseases and Conditions, United States: Weekly Tables. https://wonder.cdc.gov/nndss/nndss_weekly_tables_menu.asp.
Graham KA et al. Varicella Outbreak Among Recent Arrivals to New York City, 2022-2024. MMWR Morb Mortal Wkly Rep. 2024 May 30;73(21):478-483. doi: 10.15585/mmwr.mm7321a1.
Marin M et al. Health and Economic Impact of the United States Varicella Vaccination Program, 1996-2020. J Infect Dis. 2022 Oct 21;226(Suppl 4):S463-S469. doi: 10.1093/infdis/jiac271.
Varicella-Zoster Virus Infections in Kimberkin DW et al, eds. Red Book: 2024 Report of the Committee on Infectious Diseases, 33rd Edition. American Academy of Pediatrics, 2024:938-951. https://www.aap.org/Red-Book-2024-Report-of-the-Committee-on-Infectious-Diseases-33rd-Edition-Paperback?srsltid=AfmBOoqyF60rR9ZwQ5jA8AouNhtRRTyPLnc_r7HWw7JVYV8v33Hr2vQS.
Practitioners providing care to children are familiar with the childhood immunization schedule and routinely administer varicella vaccine at the 12-month and 4- to 5-year visits. However, when is the last time most of us or any of the current trainees have seen a case?
Briefly, varicella is a highly contagious disease caused by varicella-zoster virus (VZV). It is characterized by a generalized pruritic erythematous rash in various stages of development beginning as macules, progressing to papules, and ultimately becoming vesicular lesions on an erythematous base (“dewdrop on a rose petal”) and resolves with crusting of the lesion (Figure 1). It has an incubation period of 10-21 days with symptoms usually developing within 14-16 days after exposure. The vesicular rash must be differentiated from enterovirus, Staphylococcus aureus, contact dermatitis, or insect bites, which initially may be difficult. Approximately 50% of children can have symptoms including fever, malaise, anorexia, headache, and occasionally, mild abdominal pain in the 24-48 hours prior to the appearance of rash. Lesions usually first appear on the scalp, face, or trunk in successive crops over several days. A person with varicella has lesions in various stages.
In a normal host, new vesicle formation usually stops within 4 days, and most lesions have fully crusted by day 6. VZV establishes latency in sensory ganglia and may reactivate years or decades later to cause herpes zoster (HZ). Most healthy children with varicella recover without sequelae so the disease is generally regarded as benign. However, varicella can lead to serious complications and deaths in healthy as well as immunocompromised persons.
Complications of Varicella: bacterial superinfection of skin lesions most often with Streptococcus pyogenes or S aureus manifested as cellulitis, myositis, or necrotizing fasciitis; neurologic complications include cerebellar ataxia and encephalitis with the latter seen most often in adults. Pneumonia occurs most often in adults, especially those infected during pregnancy. Another concern, infection during the first 20 weeks of pregnancy can lead to fetal death or severe birth defects, including limb hypoplasia, cutaneous scarring, ocular abnormalities, and central nervous system damage (congenital varicella syndrome).
The risk for development of severe disseminated disease was first noted in the 1960s as treatments for leukemia in children improved. They were surviving their cancer only to develop severe and often fatal varicella. Today it is recognized that development of disseminated disease is a risk for all infected persons with impaired T cell function, malignancies, HIV, or receiving immunosuppressive therapy.
Reye’s syndrome is rarely seen today since taking salicylates while infected with VZV was identified as a predisposing factor for development.
VZV is only found in humans and transmission is person to person or airborne. The secondary household attack rate is approximately 90%. In contrast, the secondary attack rates in classrooms may be as low as 12%-33%. Transmission rates in the tropics for unexplained reasons are also lower.
Vaccine History: Why do we rarely see this disease anymore? Varicella, a live attenuated vaccine, was developed in 1974 by Dr. Michiaki Takahashi. It remains the only vaccine directed against a herpes group virus. In 1979, the Collaborative Varicella Vaccine Study Group was established at the National Institutes of Health (NIH) and additional safety and efficacy trials were conducted in the United States initially in leukemic patients in remission and later in healthy children, which supported Takahashi’s data. Licensure of varicella vaccine was granted in 1995. That same year, due to continuing disease and societal burden, the United States was the first country to incorporate varicella into the routine childhood immunization schedule, which resulted in significant reductions in cases. To further improve control of varicella, in 2007 vaccine recommendations were revised and a routine two-dose schedule was implemented. The impact of varicella disease pre- and post-vaccine licensure is illustrated in Figure 2. Not listed, is that in the pre-vaccine era, there were approximately 44 cases of congenital varicella syndrome annually.

As of 2023 only 23% (45/195) of nations routinely administer this vaccine and 4% (8/195) have restricted recommendations. The remaining 73% of countries do not offer the vaccine, including all countries on the African continent, and Cuba, Guatemala, Haiti, Honduras, India, Jordan, Lebanon, Philippines, Portugal, and Venezuela to list a few.
Varicella Outbreak: In October 2022, New York City (NYC) identified a varicella outbreak primarily involving persons who recently migrated from Central and South America and lived in a shelter in NYC or residential facility (n = 105); the outbreak is ongoing. As of March 8, 2024, 873 cases (53%) were among children aged 4-18 years and 91.9% had no documentation of varicella vaccine at time of symptom onset. There were 28 hospitalizations, and no deaths reported. The most common sources of transmission were the residential facilities (41.3%) and importation or possible importation (39.4%). School transmission accounted for only 1.2% of cases.
Most migrants arrived from countries where varicella vaccination is not part of the routine childhood immunization schedule. Although most cases occurred in children, almost 30% occurred in adults. Many of the migrants arrived from tropical countries where susceptibility rates are also higher in adults. This outbreak is a reminder of the importance of limiting disease transmission by maintaining high vaccination rates. To curtail this outbreak, approximately 27,000 doses of varicella vaccine were administered to the arriving migrants. In addition, MMR, COVID-19, influenza, and all routine pediatric vaccines required for school entry were administered. Temporary closure of the residential facilities were required. Education was provided to residents regarding immunizations as well as assistance to help them establish a primary care home. Multiple agencies were mobilized to successfully coordinate these efforts.
Take Home Message
1. Each country has its own routine immunization schedule. It may not include all vaccines recommended in the US schedule. When questioned I’m frequently told that immunizations are up to date, only to review records and find they are not, especially when it is related to MMR. It is often administered at 9 months and/or MR or MM is administered depending on the country. As reported here, varicella is a routine vaccine in only 45 countries.
2.
3. Once an outbreak has been identified, the infrastructure to manage and contain it must already be established. In most instances there will be a need for a rapid and often large-scale effort involving multiple agencies including local health care providers.
4. Not all diseases are reportable. Only deaths by varicella are nationally notifiable. Otherwise, cases are reported voluntarily. As of November 2, 2024, there have been 5,157 cases of varicella reported, excluding any cases from NYC.
Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
CDC. Nationally Notifiable Infectious Diseases and Conditions, United States: Weekly Tables. https://wonder.cdc.gov/nndss/nndss_weekly_tables_menu.asp.
Graham KA et al. Varicella Outbreak Among Recent Arrivals to New York City, 2022-2024. MMWR Morb Mortal Wkly Rep. 2024 May 30;73(21):478-483. doi: 10.15585/mmwr.mm7321a1.
Marin M et al. Health and Economic Impact of the United States Varicella Vaccination Program, 1996-2020. J Infect Dis. 2022 Oct 21;226(Suppl 4):S463-S469. doi: 10.1093/infdis/jiac271.
Varicella-Zoster Virus Infections in Kimberkin DW et al, eds. Red Book: 2024 Report of the Committee on Infectious Diseases, 33rd Edition. American Academy of Pediatrics, 2024:938-951. https://www.aap.org/Red-Book-2024-Report-of-the-Committee-on-Infectious-Diseases-33rd-Edition-Paperback?srsltid=AfmBOoqyF60rR9ZwQ5jA8AouNhtRRTyPLnc_r7HWw7JVYV8v33Hr2vQS.
Practitioners providing care to children are familiar with the childhood immunization schedule and routinely administer varicella vaccine at the 12-month and 4- to 5-year visits. However, when is the last time most of us or any of the current trainees have seen a case?
Briefly, varicella is a highly contagious disease caused by varicella-zoster virus (VZV). It is characterized by a generalized pruritic erythematous rash in various stages of development beginning as macules, progressing to papules, and ultimately becoming vesicular lesions on an erythematous base (“dewdrop on a rose petal”) and resolves with crusting of the lesion (Figure 1). It has an incubation period of 10-21 days with symptoms usually developing within 14-16 days after exposure. The vesicular rash must be differentiated from enterovirus, Staphylococcus aureus, contact dermatitis, or insect bites, which initially may be difficult. Approximately 50% of children can have symptoms including fever, malaise, anorexia, headache, and occasionally, mild abdominal pain in the 24-48 hours prior to the appearance of rash. Lesions usually first appear on the scalp, face, or trunk in successive crops over several days. A person with varicella has lesions in various stages.
In a normal host, new vesicle formation usually stops within 4 days, and most lesions have fully crusted by day 6. VZV establishes latency in sensory ganglia and may reactivate years or decades later to cause herpes zoster (HZ). Most healthy children with varicella recover without sequelae so the disease is generally regarded as benign. However, varicella can lead to serious complications and deaths in healthy as well as immunocompromised persons.
Complications of Varicella: bacterial superinfection of skin lesions most often with Streptococcus pyogenes or S aureus manifested as cellulitis, myositis, or necrotizing fasciitis; neurologic complications include cerebellar ataxia and encephalitis with the latter seen most often in adults. Pneumonia occurs most often in adults, especially those infected during pregnancy. Another concern, infection during the first 20 weeks of pregnancy can lead to fetal death or severe birth defects, including limb hypoplasia, cutaneous scarring, ocular abnormalities, and central nervous system damage (congenital varicella syndrome).
The risk for development of severe disseminated disease was first noted in the 1960s as treatments for leukemia in children improved. They were surviving their cancer only to develop severe and often fatal varicella. Today it is recognized that development of disseminated disease is a risk for all infected persons with impaired T cell function, malignancies, HIV, or receiving immunosuppressive therapy.
Reye’s syndrome is rarely seen today since taking salicylates while infected with VZV was identified as a predisposing factor for development.
VZV is only found in humans and transmission is person to person or airborne. The secondary household attack rate is approximately 90%. In contrast, the secondary attack rates in classrooms may be as low as 12%-33%. Transmission rates in the tropics for unexplained reasons are also lower.
Vaccine History: Why do we rarely see this disease anymore? Varicella, a live attenuated vaccine, was developed in 1974 by Dr. Michiaki Takahashi. It remains the only vaccine directed against a herpes group virus. In 1979, the Collaborative Varicella Vaccine Study Group was established at the National Institutes of Health (NIH) and additional safety and efficacy trials were conducted in the United States initially in leukemic patients in remission and later in healthy children, which supported Takahashi’s data. Licensure of varicella vaccine was granted in 1995. That same year, due to continuing disease and societal burden, the United States was the first country to incorporate varicella into the routine childhood immunization schedule, which resulted in significant reductions in cases. To further improve control of varicella, in 2007 vaccine recommendations were revised and a routine two-dose schedule was implemented. The impact of varicella disease pre- and post-vaccine licensure is illustrated in Figure 2. Not listed, is that in the pre-vaccine era, there were approximately 44 cases of congenital varicella syndrome annually.

As of 2023 only 23% (45/195) of nations routinely administer this vaccine and 4% (8/195) have restricted recommendations. The remaining 73% of countries do not offer the vaccine, including all countries on the African continent, and Cuba, Guatemala, Haiti, Honduras, India, Jordan, Lebanon, Philippines, Portugal, and Venezuela to list a few.
Varicella Outbreak: In October 2022, New York City (NYC) identified a varicella outbreak primarily involving persons who recently migrated from Central and South America and lived in a shelter in NYC or residential facility (n = 105); the outbreak is ongoing. As of March 8, 2024, 873 cases (53%) were among children aged 4-18 years and 91.9% had no documentation of varicella vaccine at time of symptom onset. There were 28 hospitalizations, and no deaths reported. The most common sources of transmission were the residential facilities (41.3%) and importation or possible importation (39.4%). School transmission accounted for only 1.2% of cases.
Most migrants arrived from countries where varicella vaccination is not part of the routine childhood immunization schedule. Although most cases occurred in children, almost 30% occurred in adults. Many of the migrants arrived from tropical countries where susceptibility rates are also higher in adults. This outbreak is a reminder of the importance of limiting disease transmission by maintaining high vaccination rates. To curtail this outbreak, approximately 27,000 doses of varicella vaccine were administered to the arriving migrants. In addition, MMR, COVID-19, influenza, and all routine pediatric vaccines required for school entry were administered. Temporary closure of the residential facilities were required. Education was provided to residents regarding immunizations as well as assistance to help them establish a primary care home. Multiple agencies were mobilized to successfully coordinate these efforts.
Take Home Message
1. Each country has its own routine immunization schedule. It may not include all vaccines recommended in the US schedule. When questioned I’m frequently told that immunizations are up to date, only to review records and find they are not, especially when it is related to MMR. It is often administered at 9 months and/or MR or MM is administered depending on the country. As reported here, varicella is a routine vaccine in only 45 countries.
2.
3. Once an outbreak has been identified, the infrastructure to manage and contain it must already be established. In most instances there will be a need for a rapid and often large-scale effort involving multiple agencies including local health care providers.
4. Not all diseases are reportable. Only deaths by varicella are nationally notifiable. Otherwise, cases are reported voluntarily. As of November 2, 2024, there have been 5,157 cases of varicella reported, excluding any cases from NYC.
Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
CDC. Nationally Notifiable Infectious Diseases and Conditions, United States: Weekly Tables. https://wonder.cdc.gov/nndss/nndss_weekly_tables_menu.asp.
Graham KA et al. Varicella Outbreak Among Recent Arrivals to New York City, 2022-2024. MMWR Morb Mortal Wkly Rep. 2024 May 30;73(21):478-483. doi: 10.15585/mmwr.mm7321a1.
Marin M et al. Health and Economic Impact of the United States Varicella Vaccination Program, 1996-2020. J Infect Dis. 2022 Oct 21;226(Suppl 4):S463-S469. doi: 10.1093/infdis/jiac271.
Varicella-Zoster Virus Infections in Kimberkin DW et al, eds. Red Book: 2024 Report of the Committee on Infectious Diseases, 33rd Edition. American Academy of Pediatrics, 2024:938-951. https://www.aap.org/Red-Book-2024-Report-of-the-Committee-on-Infectious-Diseases-33rd-Edition-Paperback?srsltid=AfmBOoqyF60rR9ZwQ5jA8AouNhtRRTyPLnc_r7HWw7JVYV8v33Hr2vQS.
‘Being a Doctor Isn’t Healthy’: Train Your Body to Handle It
Heather K. Schopper, MD, a head and neck surgeon at Penn State Health, Hershey, Pennsylvania, wasn’t long into her career when she began feeling its physical demands. Standing for 12 hours at a time, holding awkward positions for long periods, and working with surgical tables and instruments made for doctors much taller and larger meant severe back, shoulder, and neck pain at the end of every shift.
“You just want to lie down on the floor at the end of the day,” Schopper explained. “The wear and tear of our profession is really challenging.”
Here’s the thing: At the time Schopper wasn’t particularly out of shape. She only knew she needed to build up her body for long days and a long career. What, physically, would that look like?
This was the catalyst for what she calls a “health and fitness journey” that transformed the way she practices.
“Medicine is unique in its physical demands,” said Meghan Wieser, PT, DPT, a doctor of physical therapy at Recharge Health and Fitness in Ellicott City, Maryland. Wieser frequently works with physicians and others in high-stress career environments, and she’s observed the serious toll that physically demanding medical practice can take on the body.
It’s not just about preventing acute or chronic injury, she said. It’s about performing better for longer periods. And every doctor knows the only way to build a more functional body is training.
The Fantasy of Physical Perfection vs the Reality of, Well, Reality
Jordan D. Metzl, MD, is a sports medicine physician at Hospital for Special Surgery (HSS) in New York City. He’s also a lifelong triathlete and marathon runner and has parlayed that passion into an online fitness community of more than 10,000 people called Ironstrength. Through that, Metzl has led free exercise classes in Central Park for years. He doesn’t dabble. Three times a year he leads a boot camp class of more than 1000 people on the flight deck of the USS Intrepid on the Hudson River.
“I get it, being a doctor is all about the hours,” he said. “The time sacrifices get brutal and you have to cut something out, sometimes every day. For a lot of us, that’s exercise.”
Metzl understands it so well that he recently began leading twice-monthly boot camp classes just for his HSS physician colleagues on Wednesday mornings. He says those doctors both want and need that extra boost and will be aggressive about making time for it.
“The better shape you’re in, the better job you’ll do as a physician,” he said. “You’ll feel better when the hours get long. In my own career, I have always been a better doctor when I’m active and in shape.”
Knowledge isn’t really the issue for physicians. Reality is. And reality dictates that doctors have just as much issue with achieving consistency as any patient they prescribe exercise to.
Metzl suggests total body functional training to mimic real-world movement, particularly core and lower body to keep you upright for hours at a time. How do you schedule that? He uses early mornings and weekends to train for his races and run his fitness classes, which is why his primary advice is to focus not on the activity, but on time.
“Schedule full workouts when you can and steal the rest,” he said.
Schopper agrees. “You may not be able to fit in 60 minutes of exercise every day, but 20-30 minutes of intentional movement is key,” she explained. “When you have a day off, prioritize a longer session of something you can’t fit in on workdays.”
Those shorter bouts of exercise might include “bookending” the day with 10 minutes of burpees in the morning and then 10 minutes of bodyweight strength moves like planks, push-ups, and air squats in the evening.
“Bodyweight exercises are low-hanging fruit,” said Wieser. “If you’ve got a short window, aim for something that can shoot your heart rate up quickly.”
You can also throw in “movement snacks” throughout the day — skip the elevator and run up a flight of stairs, walk around during a quick lunch break, or throw in a set of jumping jacks between patients. (Don’t worry — you won’t be dripping sweat when they walk in.)
Remember, the rehab room in the orthopedic wing may have a few dumbbells and exercise bands you can utilize when you have 5 extra minutes in your day. “Any way you can squeeze in extra movement counts,” said Wieser.
Feats of Strength? Neighborhood Sprints? It All Matters
Kissinger Goldman, DO, a Florida-based ER physician, began his dedication to exercise 17 years ago, after a high-cholesterol diagnosis. “Did I have time to exercise in medical school and residency? Yes,” Goldman admitted. “But I didn’t have the same commitment to my health until I received that number. I set about to change everything.”
Goldman follows the approach of dividing up his exercise routine into short or long sessions, depending on his schedule. “If I’m off, I’ll aim for 30 minutes of cardio and 30 minutes of strength and core work,” he explained. “When I have to work, I’ll do a compressed version of that routine as soon as I wake up, and make sure the cardio is very intense — I’ll sprint in my neighborhood, for instance.”
Matt Klein, a doctor of physical therapy and professor at George Fox University in Newberg, Oregon, who has treated many doctors, says that, when pushed for time, just 20 minutes of “heavy” strength training can deliver good results. “The definition of heavy will vary, but aim for a weight that is challenging, whether a beginner or a more experienced exerciser,” he said. “Most doctors won’t have time to go to the gym, so a simple set of dumbbells or kettlebells will work just fine. The easier it is to access, the more likely you are to do it consistently.”
Klein is a fan of strength training with good reason: “Strength is a predictor of chronic disease, so doing some high-level strength training or power training can go a long way,” he said.
The endorphin high and overall sense of improved well-being are an extra bonus. Goldman credits it with ensuring he rarely misses a workout.
Get Hardcore About Sleep
Consider the following passage: “There are clear negative effects of sleep deprivation on performance, including reaction time, accuracy, vigor, submaximal strength, and endurance. Cognitive functions such as judgment and decision-making also suffer.”
Does that sound like how you feel on suboptimal sleep? That’s from an International Journal of Sports Medicine study on the effects of sleep deprivation on athletes.
Athletes aren’t doctors — but when you consider “reaction time, accuracy, endurance, judgment, and decision-making” — doctors could certainly benefit by thinking like athletes.
Schopper is serious about sleep and sets firm boundaries.
“It’s hard,” she admitted. “We want to work, see our families, have fun. But I work hard to say, ‘I’m done,’ and go to bed.”
“Rest is crucial for this job,” agreed Goldman. “If you don’t have adequate sleep, your cortisone levels are going to go up. When you’re exhausted and you’re working, you’re likely to miss something.” Goldman is consistent with early bedtimes around 9:00 or 9:30 PM, and he allows for a bit of “wind-down” time by reading for about 20 minutes before nodding off.
Goldman also sees a link between rest and improved interactions with patients. “There’s a direct correlation between number of hours worked in a row with respect to ‘customer service’ with patients,” he said.
But don’t aim for perfection. Allow some wiggle room for the time you spend asleep, Klein recommends. “We’ve always aimed for 8 hours, but there’s evidence that even 6 or 7 hours can be enough to allow you to recover as needed,” he said. “Optimally, you want that to be uninterrupted, but if not, a 10-minute power nap can help with mental clarity.”
Keep Searching, Keep Trying, Keep Training
Schopper was never, nor has she become, a gym rat. Still, “I knew I needed to build upper body strength,” she said. That meant expanding her fitness possibilities beyond the obvious. She discovered aerial arts — intense workouts using straps and other suspension tools to work every muscle in her body while hanging from the ceiling. Increased strength was a given, but she also seriously increased her range of motion.
For Schopper, the improvements to her lifestyle have been game changers. “I still have long days, but I’m no longer sore and tired after them,” she said. “I sleep better and have more energy. I’m proud of myself for putting the effort into this.”
A journey toward health and fitness may look different for everyone, but (as doctors frequently tell their patients) it’s a path anyone can follow.
“Being a doctor is not necessarily good for your health,” said Klein. “The body can handle the job, however, if you train for it.”
A version of this article first appeared on Medscape.com.
Heather K. Schopper, MD, a head and neck surgeon at Penn State Health, Hershey, Pennsylvania, wasn’t long into her career when she began feeling its physical demands. Standing for 12 hours at a time, holding awkward positions for long periods, and working with surgical tables and instruments made for doctors much taller and larger meant severe back, shoulder, and neck pain at the end of every shift.
“You just want to lie down on the floor at the end of the day,” Schopper explained. “The wear and tear of our profession is really challenging.”
Here’s the thing: At the time Schopper wasn’t particularly out of shape. She only knew she needed to build up her body for long days and a long career. What, physically, would that look like?
This was the catalyst for what she calls a “health and fitness journey” that transformed the way she practices.
“Medicine is unique in its physical demands,” said Meghan Wieser, PT, DPT, a doctor of physical therapy at Recharge Health and Fitness in Ellicott City, Maryland. Wieser frequently works with physicians and others in high-stress career environments, and she’s observed the serious toll that physically demanding medical practice can take on the body.
It’s not just about preventing acute or chronic injury, she said. It’s about performing better for longer periods. And every doctor knows the only way to build a more functional body is training.
The Fantasy of Physical Perfection vs the Reality of, Well, Reality
Jordan D. Metzl, MD, is a sports medicine physician at Hospital for Special Surgery (HSS) in New York City. He’s also a lifelong triathlete and marathon runner and has parlayed that passion into an online fitness community of more than 10,000 people called Ironstrength. Through that, Metzl has led free exercise classes in Central Park for years. He doesn’t dabble. Three times a year he leads a boot camp class of more than 1000 people on the flight deck of the USS Intrepid on the Hudson River.
“I get it, being a doctor is all about the hours,” he said. “The time sacrifices get brutal and you have to cut something out, sometimes every day. For a lot of us, that’s exercise.”
Metzl understands it so well that he recently began leading twice-monthly boot camp classes just for his HSS physician colleagues on Wednesday mornings. He says those doctors both want and need that extra boost and will be aggressive about making time for it.
“The better shape you’re in, the better job you’ll do as a physician,” he said. “You’ll feel better when the hours get long. In my own career, I have always been a better doctor when I’m active and in shape.”
Knowledge isn’t really the issue for physicians. Reality is. And reality dictates that doctors have just as much issue with achieving consistency as any patient they prescribe exercise to.
Metzl suggests total body functional training to mimic real-world movement, particularly core and lower body to keep you upright for hours at a time. How do you schedule that? He uses early mornings and weekends to train for his races and run his fitness classes, which is why his primary advice is to focus not on the activity, but on time.
“Schedule full workouts when you can and steal the rest,” he said.
Schopper agrees. “You may not be able to fit in 60 minutes of exercise every day, but 20-30 minutes of intentional movement is key,” she explained. “When you have a day off, prioritize a longer session of something you can’t fit in on workdays.”
Those shorter bouts of exercise might include “bookending” the day with 10 minutes of burpees in the morning and then 10 minutes of bodyweight strength moves like planks, push-ups, and air squats in the evening.
“Bodyweight exercises are low-hanging fruit,” said Wieser. “If you’ve got a short window, aim for something that can shoot your heart rate up quickly.”
You can also throw in “movement snacks” throughout the day — skip the elevator and run up a flight of stairs, walk around during a quick lunch break, or throw in a set of jumping jacks between patients. (Don’t worry — you won’t be dripping sweat when they walk in.)
Remember, the rehab room in the orthopedic wing may have a few dumbbells and exercise bands you can utilize when you have 5 extra minutes in your day. “Any way you can squeeze in extra movement counts,” said Wieser.
Feats of Strength? Neighborhood Sprints? It All Matters
Kissinger Goldman, DO, a Florida-based ER physician, began his dedication to exercise 17 years ago, after a high-cholesterol diagnosis. “Did I have time to exercise in medical school and residency? Yes,” Goldman admitted. “But I didn’t have the same commitment to my health until I received that number. I set about to change everything.”
Goldman follows the approach of dividing up his exercise routine into short or long sessions, depending on his schedule. “If I’m off, I’ll aim for 30 minutes of cardio and 30 minutes of strength and core work,” he explained. “When I have to work, I’ll do a compressed version of that routine as soon as I wake up, and make sure the cardio is very intense — I’ll sprint in my neighborhood, for instance.”
Matt Klein, a doctor of physical therapy and professor at George Fox University in Newberg, Oregon, who has treated many doctors, says that, when pushed for time, just 20 minutes of “heavy” strength training can deliver good results. “The definition of heavy will vary, but aim for a weight that is challenging, whether a beginner or a more experienced exerciser,” he said. “Most doctors won’t have time to go to the gym, so a simple set of dumbbells or kettlebells will work just fine. The easier it is to access, the more likely you are to do it consistently.”
Klein is a fan of strength training with good reason: “Strength is a predictor of chronic disease, so doing some high-level strength training or power training can go a long way,” he said.
The endorphin high and overall sense of improved well-being are an extra bonus. Goldman credits it with ensuring he rarely misses a workout.
Get Hardcore About Sleep
Consider the following passage: “There are clear negative effects of sleep deprivation on performance, including reaction time, accuracy, vigor, submaximal strength, and endurance. Cognitive functions such as judgment and decision-making also suffer.”
Does that sound like how you feel on suboptimal sleep? That’s from an International Journal of Sports Medicine study on the effects of sleep deprivation on athletes.
Athletes aren’t doctors — but when you consider “reaction time, accuracy, endurance, judgment, and decision-making” — doctors could certainly benefit by thinking like athletes.
Schopper is serious about sleep and sets firm boundaries.
“It’s hard,” she admitted. “We want to work, see our families, have fun. But I work hard to say, ‘I’m done,’ and go to bed.”
“Rest is crucial for this job,” agreed Goldman. “If you don’t have adequate sleep, your cortisone levels are going to go up. When you’re exhausted and you’re working, you’re likely to miss something.” Goldman is consistent with early bedtimes around 9:00 or 9:30 PM, and he allows for a bit of “wind-down” time by reading for about 20 minutes before nodding off.
Goldman also sees a link between rest and improved interactions with patients. “There’s a direct correlation between number of hours worked in a row with respect to ‘customer service’ with patients,” he said.
But don’t aim for perfection. Allow some wiggle room for the time you spend asleep, Klein recommends. “We’ve always aimed for 8 hours, but there’s evidence that even 6 or 7 hours can be enough to allow you to recover as needed,” he said. “Optimally, you want that to be uninterrupted, but if not, a 10-minute power nap can help with mental clarity.”
Keep Searching, Keep Trying, Keep Training
Schopper was never, nor has she become, a gym rat. Still, “I knew I needed to build upper body strength,” she said. That meant expanding her fitness possibilities beyond the obvious. She discovered aerial arts — intense workouts using straps and other suspension tools to work every muscle in her body while hanging from the ceiling. Increased strength was a given, but she also seriously increased her range of motion.
For Schopper, the improvements to her lifestyle have been game changers. “I still have long days, but I’m no longer sore and tired after them,” she said. “I sleep better and have more energy. I’m proud of myself for putting the effort into this.”
A journey toward health and fitness may look different for everyone, but (as doctors frequently tell their patients) it’s a path anyone can follow.
“Being a doctor is not necessarily good for your health,” said Klein. “The body can handle the job, however, if you train for it.”
A version of this article first appeared on Medscape.com.
Heather K. Schopper, MD, a head and neck surgeon at Penn State Health, Hershey, Pennsylvania, wasn’t long into her career when she began feeling its physical demands. Standing for 12 hours at a time, holding awkward positions for long periods, and working with surgical tables and instruments made for doctors much taller and larger meant severe back, shoulder, and neck pain at the end of every shift.
“You just want to lie down on the floor at the end of the day,” Schopper explained. “The wear and tear of our profession is really challenging.”
Here’s the thing: At the time Schopper wasn’t particularly out of shape. She only knew she needed to build up her body for long days and a long career. What, physically, would that look like?
This was the catalyst for what she calls a “health and fitness journey” that transformed the way she practices.
“Medicine is unique in its physical demands,” said Meghan Wieser, PT, DPT, a doctor of physical therapy at Recharge Health and Fitness in Ellicott City, Maryland. Wieser frequently works with physicians and others in high-stress career environments, and she’s observed the serious toll that physically demanding medical practice can take on the body.
It’s not just about preventing acute or chronic injury, she said. It’s about performing better for longer periods. And every doctor knows the only way to build a more functional body is training.
The Fantasy of Physical Perfection vs the Reality of, Well, Reality
Jordan D. Metzl, MD, is a sports medicine physician at Hospital for Special Surgery (HSS) in New York City. He’s also a lifelong triathlete and marathon runner and has parlayed that passion into an online fitness community of more than 10,000 people called Ironstrength. Through that, Metzl has led free exercise classes in Central Park for years. He doesn’t dabble. Three times a year he leads a boot camp class of more than 1000 people on the flight deck of the USS Intrepid on the Hudson River.
“I get it, being a doctor is all about the hours,” he said. “The time sacrifices get brutal and you have to cut something out, sometimes every day. For a lot of us, that’s exercise.”
Metzl understands it so well that he recently began leading twice-monthly boot camp classes just for his HSS physician colleagues on Wednesday mornings. He says those doctors both want and need that extra boost and will be aggressive about making time for it.
“The better shape you’re in, the better job you’ll do as a physician,” he said. “You’ll feel better when the hours get long. In my own career, I have always been a better doctor when I’m active and in shape.”
Knowledge isn’t really the issue for physicians. Reality is. And reality dictates that doctors have just as much issue with achieving consistency as any patient they prescribe exercise to.
Metzl suggests total body functional training to mimic real-world movement, particularly core and lower body to keep you upright for hours at a time. How do you schedule that? He uses early mornings and weekends to train for his races and run his fitness classes, which is why his primary advice is to focus not on the activity, but on time.
“Schedule full workouts when you can and steal the rest,” he said.
Schopper agrees. “You may not be able to fit in 60 minutes of exercise every day, but 20-30 minutes of intentional movement is key,” she explained. “When you have a day off, prioritize a longer session of something you can’t fit in on workdays.”
Those shorter bouts of exercise might include “bookending” the day with 10 minutes of burpees in the morning and then 10 minutes of bodyweight strength moves like planks, push-ups, and air squats in the evening.
“Bodyweight exercises are low-hanging fruit,” said Wieser. “If you’ve got a short window, aim for something that can shoot your heart rate up quickly.”
You can also throw in “movement snacks” throughout the day — skip the elevator and run up a flight of stairs, walk around during a quick lunch break, or throw in a set of jumping jacks between patients. (Don’t worry — you won’t be dripping sweat when they walk in.)
Remember, the rehab room in the orthopedic wing may have a few dumbbells and exercise bands you can utilize when you have 5 extra minutes in your day. “Any way you can squeeze in extra movement counts,” said Wieser.
Feats of Strength? Neighborhood Sprints? It All Matters
Kissinger Goldman, DO, a Florida-based ER physician, began his dedication to exercise 17 years ago, after a high-cholesterol diagnosis. “Did I have time to exercise in medical school and residency? Yes,” Goldman admitted. “But I didn’t have the same commitment to my health until I received that number. I set about to change everything.”
Goldman follows the approach of dividing up his exercise routine into short or long sessions, depending on his schedule. “If I’m off, I’ll aim for 30 minutes of cardio and 30 minutes of strength and core work,” he explained. “When I have to work, I’ll do a compressed version of that routine as soon as I wake up, and make sure the cardio is very intense — I’ll sprint in my neighborhood, for instance.”
Matt Klein, a doctor of physical therapy and professor at George Fox University in Newberg, Oregon, who has treated many doctors, says that, when pushed for time, just 20 minutes of “heavy” strength training can deliver good results. “The definition of heavy will vary, but aim for a weight that is challenging, whether a beginner or a more experienced exerciser,” he said. “Most doctors won’t have time to go to the gym, so a simple set of dumbbells or kettlebells will work just fine. The easier it is to access, the more likely you are to do it consistently.”
Klein is a fan of strength training with good reason: “Strength is a predictor of chronic disease, so doing some high-level strength training or power training can go a long way,” he said.
The endorphin high and overall sense of improved well-being are an extra bonus. Goldman credits it with ensuring he rarely misses a workout.
Get Hardcore About Sleep
Consider the following passage: “There are clear negative effects of sleep deprivation on performance, including reaction time, accuracy, vigor, submaximal strength, and endurance. Cognitive functions such as judgment and decision-making also suffer.”
Does that sound like how you feel on suboptimal sleep? That’s from an International Journal of Sports Medicine study on the effects of sleep deprivation on athletes.
Athletes aren’t doctors — but when you consider “reaction time, accuracy, endurance, judgment, and decision-making” — doctors could certainly benefit by thinking like athletes.
Schopper is serious about sleep and sets firm boundaries.
“It’s hard,” she admitted. “We want to work, see our families, have fun. But I work hard to say, ‘I’m done,’ and go to bed.”
“Rest is crucial for this job,” agreed Goldman. “If you don’t have adequate sleep, your cortisone levels are going to go up. When you’re exhausted and you’re working, you’re likely to miss something.” Goldman is consistent with early bedtimes around 9:00 or 9:30 PM, and he allows for a bit of “wind-down” time by reading for about 20 minutes before nodding off.
Goldman also sees a link between rest and improved interactions with patients. “There’s a direct correlation between number of hours worked in a row with respect to ‘customer service’ with patients,” he said.
But don’t aim for perfection. Allow some wiggle room for the time you spend asleep, Klein recommends. “We’ve always aimed for 8 hours, but there’s evidence that even 6 or 7 hours can be enough to allow you to recover as needed,” he said. “Optimally, you want that to be uninterrupted, but if not, a 10-minute power nap can help with mental clarity.”
Keep Searching, Keep Trying, Keep Training
Schopper was never, nor has she become, a gym rat. Still, “I knew I needed to build upper body strength,” she said. That meant expanding her fitness possibilities beyond the obvious. She discovered aerial arts — intense workouts using straps and other suspension tools to work every muscle in her body while hanging from the ceiling. Increased strength was a given, but she also seriously increased her range of motion.
For Schopper, the improvements to her lifestyle have been game changers. “I still have long days, but I’m no longer sore and tired after them,” she said. “I sleep better and have more energy. I’m proud of myself for putting the effort into this.”
A journey toward health and fitness may look different for everyone, but (as doctors frequently tell their patients) it’s a path anyone can follow.
“Being a doctor is not necessarily good for your health,” said Klein. “The body can handle the job, however, if you train for it.”
A version of this article first appeared on Medscape.com.
Exercise or Inactivity?
The answer one gets often depends on how one crafts the question. For example, Jeffrey D. Johnson PhD, a professor of communications at Portland State University in Oregon has found that if patients are asked “Is there something else you would like to address today?” 80% had their unmet questions addressed. However, if the question was worded “Is there anything else ...?” Very few had their unmet concerns addressed.
I recently encountered two studies that provide another striking example of how differently structured questions aimed at same topic can result in dramatically different results. In this case both studies used one database, the UK Biobank cohort study which contains “de-identified genetic, lifestyle, and health information” collected from a half million adults in the UK. A subgroup of nearly 90,000 who had undergone a week long activity measurement using a wrist accelerometer was the focus of both groups of investigators who asked the same broad question “What is the relationship between physical activity and disease?”
The first study I found has already received some publicity in the lay press and dealt with those individuals who, for a variety of reasons, pack all of their exercise into just a few days, usually the weekend, aka weekend warriors. The investigators found that when compared with generally inactive individuals those who were able to achieve activity volumes that met current guidelines were at lower risk for more than 200 diseases, particularly those that were cardiac based. I guess that shouldn’t surprise us. The finding that has received most of the publicity to date in the lay press was that “Associations were similar whether the activity followed a weekend warrior pattern or was spread out evenly through the week.”
The second study, using the same database, found that those individuals who spent more than 10.6 hours per day sitting had 60% an increased risk of heart failure and cardiovascular related death. And, here’s the real news, that risk remained even in people who were otherwise physically active.
I suspect these two groups of investigators, both associated with Harvard-related institutions, knew of each other’s work and would agree that their findings are not incompatible. However, it is interesting that, when presented with the same database, one group chose to focus its attention on the exercise end of the spectrum while the other looked at the effect of inactivity.
I have always tried to include a “healthy” amount of exercise in my day. However, more recently my professional interest has been drawn to the increasing number of studies I read that deal with the risks of inactivity and sedentarism. For example, just in the last 2 years I have written about a study in children that showed that sedentary time is responsible for 70% of the total increase in cholesterol as children advance into young adulthood. Another study in adults found that every 2-hour increase in sedentary behavior was associated with a 12% decrease in the patient’s likelihood of achieving healthy aging.
If I were asked to place relative values on these two studies, I would say that the study highlighting the risk of prolonged sitting is potentially far more relevant to the population at large, which is for the most part sedentary. Of course, while I have no data to support my contention, I see the weekend warrior population as a niche group.
So what are the take-home messages from these two studies? One is for the weekend warrior. “You can take some comfort in the results that support your exercise schedule but don’t feel too comfortable about it if most of the week you are sitting at a desk.”
For the rest of us — It’s beginning to feel like we should be including accelerometers in our regular diagnostic and therapeutic weaponry. Sending home patients with a Holter cardiac monitor has become commonplace. We should be sending more folks home with accelerometers or asking the more affluent to share the data from their smart watches. “You’ve been bragging about your “steps. Show me your sitting time.”
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
The answer one gets often depends on how one crafts the question. For example, Jeffrey D. Johnson PhD, a professor of communications at Portland State University in Oregon has found that if patients are asked “Is there something else you would like to address today?” 80% had their unmet questions addressed. However, if the question was worded “Is there anything else ...?” Very few had their unmet concerns addressed.
I recently encountered two studies that provide another striking example of how differently structured questions aimed at same topic can result in dramatically different results. In this case both studies used one database, the UK Biobank cohort study which contains “de-identified genetic, lifestyle, and health information” collected from a half million adults in the UK. A subgroup of nearly 90,000 who had undergone a week long activity measurement using a wrist accelerometer was the focus of both groups of investigators who asked the same broad question “What is the relationship between physical activity and disease?”
The first study I found has already received some publicity in the lay press and dealt with those individuals who, for a variety of reasons, pack all of their exercise into just a few days, usually the weekend, aka weekend warriors. The investigators found that when compared with generally inactive individuals those who were able to achieve activity volumes that met current guidelines were at lower risk for more than 200 diseases, particularly those that were cardiac based. I guess that shouldn’t surprise us. The finding that has received most of the publicity to date in the lay press was that “Associations were similar whether the activity followed a weekend warrior pattern or was spread out evenly through the week.”
The second study, using the same database, found that those individuals who spent more than 10.6 hours per day sitting had 60% an increased risk of heart failure and cardiovascular related death. And, here’s the real news, that risk remained even in people who were otherwise physically active.
I suspect these two groups of investigators, both associated with Harvard-related institutions, knew of each other’s work and would agree that their findings are not incompatible. However, it is interesting that, when presented with the same database, one group chose to focus its attention on the exercise end of the spectrum while the other looked at the effect of inactivity.
I have always tried to include a “healthy” amount of exercise in my day. However, more recently my professional interest has been drawn to the increasing number of studies I read that deal with the risks of inactivity and sedentarism. For example, just in the last 2 years I have written about a study in children that showed that sedentary time is responsible for 70% of the total increase in cholesterol as children advance into young adulthood. Another study in adults found that every 2-hour increase in sedentary behavior was associated with a 12% decrease in the patient’s likelihood of achieving healthy aging.
If I were asked to place relative values on these two studies, I would say that the study highlighting the risk of prolonged sitting is potentially far more relevant to the population at large, which is for the most part sedentary. Of course, while I have no data to support my contention, I see the weekend warrior population as a niche group.
So what are the take-home messages from these two studies? One is for the weekend warrior. “You can take some comfort in the results that support your exercise schedule but don’t feel too comfortable about it if most of the week you are sitting at a desk.”
For the rest of us — It’s beginning to feel like we should be including accelerometers in our regular diagnostic and therapeutic weaponry. Sending home patients with a Holter cardiac monitor has become commonplace. We should be sending more folks home with accelerometers or asking the more affluent to share the data from their smart watches. “You’ve been bragging about your “steps. Show me your sitting time.”
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
The answer one gets often depends on how one crafts the question. For example, Jeffrey D. Johnson PhD, a professor of communications at Portland State University in Oregon has found that if patients are asked “Is there something else you would like to address today?” 80% had their unmet questions addressed. However, if the question was worded “Is there anything else ...?” Very few had their unmet concerns addressed.
I recently encountered two studies that provide another striking example of how differently structured questions aimed at same topic can result in dramatically different results. In this case both studies used one database, the UK Biobank cohort study which contains “de-identified genetic, lifestyle, and health information” collected from a half million adults in the UK. A subgroup of nearly 90,000 who had undergone a week long activity measurement using a wrist accelerometer was the focus of both groups of investigators who asked the same broad question “What is the relationship between physical activity and disease?”
The first study I found has already received some publicity in the lay press and dealt with those individuals who, for a variety of reasons, pack all of their exercise into just a few days, usually the weekend, aka weekend warriors. The investigators found that when compared with generally inactive individuals those who were able to achieve activity volumes that met current guidelines were at lower risk for more than 200 diseases, particularly those that were cardiac based. I guess that shouldn’t surprise us. The finding that has received most of the publicity to date in the lay press was that “Associations were similar whether the activity followed a weekend warrior pattern or was spread out evenly through the week.”
The second study, using the same database, found that those individuals who spent more than 10.6 hours per day sitting had 60% an increased risk of heart failure and cardiovascular related death. And, here’s the real news, that risk remained even in people who were otherwise physically active.
I suspect these two groups of investigators, both associated with Harvard-related institutions, knew of each other’s work and would agree that their findings are not incompatible. However, it is interesting that, when presented with the same database, one group chose to focus its attention on the exercise end of the spectrum while the other looked at the effect of inactivity.
I have always tried to include a “healthy” amount of exercise in my day. However, more recently my professional interest has been drawn to the increasing number of studies I read that deal with the risks of inactivity and sedentarism. For example, just in the last 2 years I have written about a study in children that showed that sedentary time is responsible for 70% of the total increase in cholesterol as children advance into young adulthood. Another study in adults found that every 2-hour increase in sedentary behavior was associated with a 12% decrease in the patient’s likelihood of achieving healthy aging.
If I were asked to place relative values on these two studies, I would say that the study highlighting the risk of prolonged sitting is potentially far more relevant to the population at large, which is for the most part sedentary. Of course, while I have no data to support my contention, I see the weekend warrior population as a niche group.
So what are the take-home messages from these two studies? One is for the weekend warrior. “You can take some comfort in the results that support your exercise schedule but don’t feel too comfortable about it if most of the week you are sitting at a desk.”
For the rest of us — It’s beginning to feel like we should be including accelerometers in our regular diagnostic and therapeutic weaponry. Sending home patients with a Holter cardiac monitor has become commonplace. We should be sending more folks home with accelerometers or asking the more affluent to share the data from their smart watches. “You’ve been bragging about your “steps. Show me your sitting time.”
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
CRC Screening: Right Patient, Right Test, Right Time
It has been three and a half years since the US Preventive Services Task Force (USPSTF) lowered the age to start colorectal cancer (CRC) screening from 50 to 45. As I mentioned in a previous commentary, two major medical groups — the American Academy of Family Physicians and the American College of Physicians — felt that the evidence was insufficient to support this change.
Comparing CRC screening rates in more than 10 million adults aged 45-49 during the 20 months preceding and 20 months following the USPSTF recommendation, researchers found significant increases during the latter time period, with the greatest increases among persons of high socioeconomic status or living in metropolitan areas.
Another study addressed concerns that younger adults may be less likely to follow up on positive screening results or more likely to have false positives on a fecal immunochemical test (FIT). Patients aged 45-49 years were slightly less likely to have a positive FIT result than 50-year-olds, but they had similar rates of colonoscopy completion and similar percentages of abnormal findings on colonoscopy.
Although the sensitivity and specificity of FIT varies quite a bit across different test brands, its overall effectiveness at reducing colorectal cancer deaths is well established. In 2024, the Food and Drug Administration approved three new screening options: a blood-based screening test (Shield), a next-generation multitarget stool DNA test (Cologuard Plus), and a multitarget stool RNA test (ColoSense) with similar performance characteristics as Cologuard Plus. The latter two tests will become available early next year.
This profusion of noninvasive options for CRC screening will challenge those tasked with developing the next iteration of the USPSTF recommendations. Not only must future guidelines establish what evidence threshold is sufficient to recommend a new screening strategy, but they also will need to consider the population-level consequences of relative utilization of different tests. For example, a cost-effectiveness analysis found that more CRC deaths would occur if people who would have otherwise accepted colonoscopy or fecal tests chose to be screened with Shield instead; however, this negative outcome could be offset if for every three of these test substitutions, two other people chose Shield who would otherwise have not been screened at all.
In the meantime, it is important for primary care clinicians to be familiar with evidence-based intervals for CRC screening tests and test eligibility criteria. A troubling study of patients who completed a multitarget stool DNA test in a Midwestern health system in 2021 found that more than one in five had the test ordered inappropriately, based on USPSTF guidelines. Reasons for inappropriate testing included having had a colonoscopy within the past 10 years, a family history of CRC, symptoms suggestive of possible CRC, age younger than 45, and a prior diagnosis of colonic adenomas.
Just as a medication works best when the patient takes it as prescribed, a CRC screening test is most likely to yield more benefit than harm when it’s provided to the right patient at the right time.
Dr. Lin is Associate Director, Family Medicine Residency Program, at Lancaster General Hospital in Pennsylvania. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
It has been three and a half years since the US Preventive Services Task Force (USPSTF) lowered the age to start colorectal cancer (CRC) screening from 50 to 45. As I mentioned in a previous commentary, two major medical groups — the American Academy of Family Physicians and the American College of Physicians — felt that the evidence was insufficient to support this change.
Comparing CRC screening rates in more than 10 million adults aged 45-49 during the 20 months preceding and 20 months following the USPSTF recommendation, researchers found significant increases during the latter time period, with the greatest increases among persons of high socioeconomic status or living in metropolitan areas.
Another study addressed concerns that younger adults may be less likely to follow up on positive screening results or more likely to have false positives on a fecal immunochemical test (FIT). Patients aged 45-49 years were slightly less likely to have a positive FIT result than 50-year-olds, but they had similar rates of colonoscopy completion and similar percentages of abnormal findings on colonoscopy.
Although the sensitivity and specificity of FIT varies quite a bit across different test brands, its overall effectiveness at reducing colorectal cancer deaths is well established. In 2024, the Food and Drug Administration approved three new screening options: a blood-based screening test (Shield), a next-generation multitarget stool DNA test (Cologuard Plus), and a multitarget stool RNA test (ColoSense) with similar performance characteristics as Cologuard Plus. The latter two tests will become available early next year.
This profusion of noninvasive options for CRC screening will challenge those tasked with developing the next iteration of the USPSTF recommendations. Not only must future guidelines establish what evidence threshold is sufficient to recommend a new screening strategy, but they also will need to consider the population-level consequences of relative utilization of different tests. For example, a cost-effectiveness analysis found that more CRC deaths would occur if people who would have otherwise accepted colonoscopy or fecal tests chose to be screened with Shield instead; however, this negative outcome could be offset if for every three of these test substitutions, two other people chose Shield who would otherwise have not been screened at all.
In the meantime, it is important for primary care clinicians to be familiar with evidence-based intervals for CRC screening tests and test eligibility criteria. A troubling study of patients who completed a multitarget stool DNA test in a Midwestern health system in 2021 found that more than one in five had the test ordered inappropriately, based on USPSTF guidelines. Reasons for inappropriate testing included having had a colonoscopy within the past 10 years, a family history of CRC, symptoms suggestive of possible CRC, age younger than 45, and a prior diagnosis of colonic adenomas.
Just as a medication works best when the patient takes it as prescribed, a CRC screening test is most likely to yield more benefit than harm when it’s provided to the right patient at the right time.
Dr. Lin is Associate Director, Family Medicine Residency Program, at Lancaster General Hospital in Pennsylvania. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
It has been three and a half years since the US Preventive Services Task Force (USPSTF) lowered the age to start colorectal cancer (CRC) screening from 50 to 45. As I mentioned in a previous commentary, two major medical groups — the American Academy of Family Physicians and the American College of Physicians — felt that the evidence was insufficient to support this change.
Comparing CRC screening rates in more than 10 million adults aged 45-49 during the 20 months preceding and 20 months following the USPSTF recommendation, researchers found significant increases during the latter time period, with the greatest increases among persons of high socioeconomic status or living in metropolitan areas.
Another study addressed concerns that younger adults may be less likely to follow up on positive screening results or more likely to have false positives on a fecal immunochemical test (FIT). Patients aged 45-49 years were slightly less likely to have a positive FIT result than 50-year-olds, but they had similar rates of colonoscopy completion and similar percentages of abnormal findings on colonoscopy.
Although the sensitivity and specificity of FIT varies quite a bit across different test brands, its overall effectiveness at reducing colorectal cancer deaths is well established. In 2024, the Food and Drug Administration approved three new screening options: a blood-based screening test (Shield), a next-generation multitarget stool DNA test (Cologuard Plus), and a multitarget stool RNA test (ColoSense) with similar performance characteristics as Cologuard Plus. The latter two tests will become available early next year.
This profusion of noninvasive options for CRC screening will challenge those tasked with developing the next iteration of the USPSTF recommendations. Not only must future guidelines establish what evidence threshold is sufficient to recommend a new screening strategy, but they also will need to consider the population-level consequences of relative utilization of different tests. For example, a cost-effectiveness analysis found that more CRC deaths would occur if people who would have otherwise accepted colonoscopy or fecal tests chose to be screened with Shield instead; however, this negative outcome could be offset if for every three of these test substitutions, two other people chose Shield who would otherwise have not been screened at all.
In the meantime, it is important for primary care clinicians to be familiar with evidence-based intervals for CRC screening tests and test eligibility criteria. A troubling study of patients who completed a multitarget stool DNA test in a Midwestern health system in 2021 found that more than one in five had the test ordered inappropriately, based on USPSTF guidelines. Reasons for inappropriate testing included having had a colonoscopy within the past 10 years, a family history of CRC, symptoms suggestive of possible CRC, age younger than 45, and a prior diagnosis of colonic adenomas.
Just as a medication works best when the patient takes it as prescribed, a CRC screening test is most likely to yield more benefit than harm when it’s provided to the right patient at the right time.
Dr. Lin is Associate Director, Family Medicine Residency Program, at Lancaster General Hospital in Pennsylvania. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
CRC Screening Uptake Rises in Adults Aged 45-49 Years
TOPLINE:
but disparities by socioeconomic status and locality occurred.
METHODOLOGY:
- Researchers compared absolute and relative changes in screening uptake among average-risk adults 45-49 years between a 20-month period before and a 20-month period after the USPSTF recommendation was issued (May 1, 2018, to December 31, 2019, and May 1, 2021, to December 31, 2022). Data was evaluated bimonthly.
- They analyzed claims data from more than 10.2 million people with private Blue Cross Blue Shield (BCBS) coverage, with about three million eligible for screening during each bimonthly period, both pre- and post-recommendation.
- They used interrupted time-series analysis and autoregressive integrated moving average models to gauge changes in screening rates.
TAKEAWAY:
- Mean CRC screening uptake in average-risk adults 45-49 years increased from 0.50% in the pre-recommendation period to 1.51% post-recommendation, reflecting a significant absolute change of 1.01 percentage points but no significant relative change.
- Adults 45-49 years living in areas with the highest socioeconomic status (SES) had the largest absolute change in screening uptake compared with peers in the lowest SES areas (1.25 vs 0.75 percentage points). Relative changes were not significant.
- The absolute change in screening uptake was higher among individuals in metropolitan areas than individuals in nonmetropolitan areas (1.06 vs 0.73 percentage points). Again, relative changes were not significant.
- The screening uptake rate increased the fastest among those living in the highest SES and metropolitan areas (0.24 and 0.20 percentage points every 2 months, respectively).
- By December 2022 (the end of the post-recommendation period), CRC screening uptake among adults 45-49 years were on par with those seen in adults 50-75 years (2.37% vs 2.4%). Nonetheless, only 11.5% of average-risk adults aged 45-49 years received CRC screening during the post-recommendation period.
IN PRACTICE:
“The threefold increase in screening uptake among average-risk individuals aged 45-49 years reflects an accomplishment, yet evidence of widening disparities based on SDI [Social Deprivation Index] and locality indicate that population subgroups may not be benefiting equally from this change in CRC screening recommendation. Furthermore, given that only 11.5% of average-risk individuals aged 45-49 years during the post-recommendation period received CRC screening before the age of 50 years, targeted initiatives to improve screening in this age group are warranted to reach the national goal of screening 80% of the population in every community,” the researchers wrote.
SOURCE:
The study, with first author Sunny Siddique, MPH, with Yale School of Public Health, New Haven, Connecticut, was published online in JAMA Network Open.
LIMITATIONS:
Data on race and ethnicity were incomplete, which may have impacted the analysis of disparities. The study cohort may not be fully representative of the general US population because BCBS beneficiaries tend to be younger and more socioeconomically advantaged with employer-based insurance. Specific information on the type of coverage provided by each beneficiary’s insurance plan was not available.
DISCLOSURES:
The study was funded by the National Cancer Institute. The authors declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
but disparities by socioeconomic status and locality occurred.
METHODOLOGY:
- Researchers compared absolute and relative changes in screening uptake among average-risk adults 45-49 years between a 20-month period before and a 20-month period after the USPSTF recommendation was issued (May 1, 2018, to December 31, 2019, and May 1, 2021, to December 31, 2022). Data was evaluated bimonthly.
- They analyzed claims data from more than 10.2 million people with private Blue Cross Blue Shield (BCBS) coverage, with about three million eligible for screening during each bimonthly period, both pre- and post-recommendation.
- They used interrupted time-series analysis and autoregressive integrated moving average models to gauge changes in screening rates.
TAKEAWAY:
- Mean CRC screening uptake in average-risk adults 45-49 years increased from 0.50% in the pre-recommendation period to 1.51% post-recommendation, reflecting a significant absolute change of 1.01 percentage points but no significant relative change.
- Adults 45-49 years living in areas with the highest socioeconomic status (SES) had the largest absolute change in screening uptake compared with peers in the lowest SES areas (1.25 vs 0.75 percentage points). Relative changes were not significant.
- The absolute change in screening uptake was higher among individuals in metropolitan areas than individuals in nonmetropolitan areas (1.06 vs 0.73 percentage points). Again, relative changes were not significant.
- The screening uptake rate increased the fastest among those living in the highest SES and metropolitan areas (0.24 and 0.20 percentage points every 2 months, respectively).
- By December 2022 (the end of the post-recommendation period), CRC screening uptake among adults 45-49 years were on par with those seen in adults 50-75 years (2.37% vs 2.4%). Nonetheless, only 11.5% of average-risk adults aged 45-49 years received CRC screening during the post-recommendation period.
IN PRACTICE:
“The threefold increase in screening uptake among average-risk individuals aged 45-49 years reflects an accomplishment, yet evidence of widening disparities based on SDI [Social Deprivation Index] and locality indicate that population subgroups may not be benefiting equally from this change in CRC screening recommendation. Furthermore, given that only 11.5% of average-risk individuals aged 45-49 years during the post-recommendation period received CRC screening before the age of 50 years, targeted initiatives to improve screening in this age group are warranted to reach the national goal of screening 80% of the population in every community,” the researchers wrote.
SOURCE:
The study, with first author Sunny Siddique, MPH, with Yale School of Public Health, New Haven, Connecticut, was published online in JAMA Network Open.
LIMITATIONS:
Data on race and ethnicity were incomplete, which may have impacted the analysis of disparities. The study cohort may not be fully representative of the general US population because BCBS beneficiaries tend to be younger and more socioeconomically advantaged with employer-based insurance. Specific information on the type of coverage provided by each beneficiary’s insurance plan was not available.
DISCLOSURES:
The study was funded by the National Cancer Institute. The authors declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
but disparities by socioeconomic status and locality occurred.
METHODOLOGY:
- Researchers compared absolute and relative changes in screening uptake among average-risk adults 45-49 years between a 20-month period before and a 20-month period after the USPSTF recommendation was issued (May 1, 2018, to December 31, 2019, and May 1, 2021, to December 31, 2022). Data was evaluated bimonthly.
- They analyzed claims data from more than 10.2 million people with private Blue Cross Blue Shield (BCBS) coverage, with about three million eligible for screening during each bimonthly period, both pre- and post-recommendation.
- They used interrupted time-series analysis and autoregressive integrated moving average models to gauge changes in screening rates.
TAKEAWAY:
- Mean CRC screening uptake in average-risk adults 45-49 years increased from 0.50% in the pre-recommendation period to 1.51% post-recommendation, reflecting a significant absolute change of 1.01 percentage points but no significant relative change.
- Adults 45-49 years living in areas with the highest socioeconomic status (SES) had the largest absolute change in screening uptake compared with peers in the lowest SES areas (1.25 vs 0.75 percentage points). Relative changes were not significant.
- The absolute change in screening uptake was higher among individuals in metropolitan areas than individuals in nonmetropolitan areas (1.06 vs 0.73 percentage points). Again, relative changes were not significant.
- The screening uptake rate increased the fastest among those living in the highest SES and metropolitan areas (0.24 and 0.20 percentage points every 2 months, respectively).
- By December 2022 (the end of the post-recommendation period), CRC screening uptake among adults 45-49 years were on par with those seen in adults 50-75 years (2.37% vs 2.4%). Nonetheless, only 11.5% of average-risk adults aged 45-49 years received CRC screening during the post-recommendation period.
IN PRACTICE:
“The threefold increase in screening uptake among average-risk individuals aged 45-49 years reflects an accomplishment, yet evidence of widening disparities based on SDI [Social Deprivation Index] and locality indicate that population subgroups may not be benefiting equally from this change in CRC screening recommendation. Furthermore, given that only 11.5% of average-risk individuals aged 45-49 years during the post-recommendation period received CRC screening before the age of 50 years, targeted initiatives to improve screening in this age group are warranted to reach the national goal of screening 80% of the population in every community,” the researchers wrote.
SOURCE:
The study, with first author Sunny Siddique, MPH, with Yale School of Public Health, New Haven, Connecticut, was published online in JAMA Network Open.
LIMITATIONS:
Data on race and ethnicity were incomplete, which may have impacted the analysis of disparities. The study cohort may not be fully representative of the general US population because BCBS beneficiaries tend to be younger and more socioeconomically advantaged with employer-based insurance. Specific information on the type of coverage provided by each beneficiary’s insurance plan was not available.
DISCLOSURES:
The study was funded by the National Cancer Institute. The authors declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Diet Matters in Prostate Cancer, but It’s Complicated
Recent studies have shown that ultralow-carbohydrate diets, weight loss diets, supplementation with omega-3 fatty acids, pro- and anti-inflammatory diets, fasting, and even tea drinking may affect prostate cancer risk or risk for progression.
In October, a cohort study involving about 900 men under active surveillance for early stage prostate cancers found that those who reported eating a diet that adhered closely to the US government’s recommendations as indicated by the Healthy Eating Index (HEI) saw a lower risk for progression at a median 6.5 months follow-up.
These findings follow results from an observational study, published in May, that followed about 2000 men with locally advanced prostate tumors. Men consuming a primarily plant-based diet (one closely adhering to the plant-based diet index) had less likelihood of progression over a median 6.5 years than those consuming diets low in plant-based foods.
“There is an increasing body of literature that says your diet matters,” said urologist Stephen J. Freedland, MD, of Cedars-Sinai Medical Center in Los Angeles, California, and director of its Center for Integrated Research in Cancer and Lifestyle. “At the same time, there are a lot of things that could explain these associations. People who can afford lots of plant-based foods tend to have higher socioeconomic status, for example.”
What’s needed, Freedland said, are more randomized trials to test the hypotheses emerging from the longitudinal cohort studies. “That’s where I’m going with my own research,” he said. “I’d like to look at a study like [one of these] and design a trial. Let’s say we get half of patients to eat according to the healthy eating index, while half eat whatever they want. Can dietary modification change which genes are turned on and off in a tumor, as a start?”
Oncologist and Nutritionist Collaborate on Multiple Studies
Nutritionist Pao-Hwa Lin, PhD, of Duke University in Durham, North Carolina, has been working for several years with Freedland on trials of nutrition interventions. A longtime researcher of chronic disease and diet, she first collaborated with Freedland on a study, published in 2019, that looked at whether insulin could be driven down with diet and exercise in men treated with androgen deprivation therapy.
Not only are high levels of insulin a known contributor to prostate cancer growth, Lin said, but “insulin resistance is a very common side effect of hormone therapy. And we saw that the low carb diet was very helpful for that.” The finding led Freedland and Lin to design further trials investigating carbohydrate restriction in people with prostate cancer.
Lin said randomized trials tend to be smaller and shorter in duration than the observational cohort studies because “interventions like these can be hard to maintain, and recruitment can be hard to sustain. A very well controlled and intensive nutrition intervention is not going to be super long.” Short trial durations also mean that prostate cancer progression can be difficult to capture. Risk for progression has to be measured using surrogate markers, such as the doubling time for prostate-specific antigen (PSA).
In 2020, Freedland and Lin published results from a pilot study of 57 men who had been treated with surgery or radiation for localized prostate cancer but had a PSA recurrence and were randomized to an ultralow-carbohydrate diet or no restrictions for 6 months. The investigators saw that PSA doubling times, an intermediate measure of tumor growth rate, were slower among those consuming the low-carb diet.
Currently they are wrapping up a trial that randomizes men who have been scheduled for radical prostatectomy to daily supplementation with walnuts, a natural source of polyphenols and omega-3 acids. This time, the aim is to determine whether gene expression in tumors changes in response to supplementation.
The researchers are also recruiting for a study in men being treated for metastatic prostate cancer. This study randomizes patients to a fasting-mimicking diet, which is a type of intermittent fasting, or no dietary restrictions for 6 months.
Developed by biologist Valter Longo, PhD, of the University of Southern California, Los Angeles, the fasting-mimicking diet has been shown to boost treatment effects in women with hormone receptor–positive breast cancer. In 2023, Longo and his colleagues published results from a small pilot study of the same diet in men with prostate cancer, reporting some positive metabolic findings.
Longo, who is consulting on Lin and Freedland’s trial, “has proven that the diet is helpful in treatment outcomes for breast cancer. So we connected and decided to test it and see if it’s helpful in prostate cancer as well.”
More Than One Approach Likely to Work
Though Lin and Freedland have focused most of their investigations on carbohydrate restriction, neither dismisses the potential for other dietary approaches to show benefit.
“There are two main schools of thought in terms of the relationship between diet and prostate cancer,” Lin said. “One is the insulin angle, and that’s what we hypothesized when we first tested the low-carb diet. The other is the inflammation angle.”
Studies have shown greater adherence to the HEI — a diet quality indicator that favors grains, fruits, dairy, vegetables, beans, and seafood — or the plant-based diet index to be associated with lower biomarkers of inflammation, she noted.
Insulin resistance, Lin explained, “is also highly related to inflammation.” (Several of the diets being investigated in prostate cancer were originally studied in diabetes.)
Moreover, weight loss caused by low-carb diets — or other healthy diets — can have a positive effect on insulin resistance independent of diet composition. “So it is a very complicated picture — and that doesn’t exclude other pathways that could also be contributing,” she said.
On the surface, a low-carb diet that is heavy in eggs, cheeses, and meats would seem to have little in common with the HEI or a plant-based diet. But Freedland noted that there are commonalities among the approaches being studied. “No one’s promoting eating a lot of simple sugars. No one’s saying eat a lot of processed foods. All of these diets emphasize whole, natural foods,” he said.
Lin hopes that she and Freedland will one day be able to test a diet that is both lower carb and anti-inflammatory in men with prostate cancer. “Why not combine the approaches, have all the good features together?” she asked.
But Freeland pointed out and explained why most clinicians don’t make dietary recommendations to their newly diagnosed patients.
“A new prostate cancer patient already gets easily an hour discussion of treatment options, of pros and cons. Patients often become overwhelmed. And then to extend it further to talk about diet, they’ll end up even more overwhelmed.” Moreover, he said, current evidence offers doctors few take-home messages to deliver besides avoiding sugar and processed foods.
Multiple dietary approaches are likely to prove helpful in prostate cancer, and when the evidence for them is better established, patients and their doctors will want to consider lifestyle factors in choosing one. The best diet will depend on a patient’s philosophy, tastes, and willingness to follow it, he concluded.
“At the end of the day I’m not rooting for one diet or another. I just want to get the answers.”
Lin disclosed no financial conflicts of interest. Freedland disclosed serving as a speaker for AstraZeneca, Astellas, and Pfizer and as a consultant for Astellas, AstraZeneca, Bayer, Eli Lilly, Janssen, Merck, Novartis, Pfizer, Sanofi-Aventis, and Sumitomo.
A version of this article first appeared on Medscape.com.
Recent studies have shown that ultralow-carbohydrate diets, weight loss diets, supplementation with omega-3 fatty acids, pro- and anti-inflammatory diets, fasting, and even tea drinking may affect prostate cancer risk or risk for progression.
In October, a cohort study involving about 900 men under active surveillance for early stage prostate cancers found that those who reported eating a diet that adhered closely to the US government’s recommendations as indicated by the Healthy Eating Index (HEI) saw a lower risk for progression at a median 6.5 months follow-up.
These findings follow results from an observational study, published in May, that followed about 2000 men with locally advanced prostate tumors. Men consuming a primarily plant-based diet (one closely adhering to the plant-based diet index) had less likelihood of progression over a median 6.5 years than those consuming diets low in plant-based foods.
“There is an increasing body of literature that says your diet matters,” said urologist Stephen J. Freedland, MD, of Cedars-Sinai Medical Center in Los Angeles, California, and director of its Center for Integrated Research in Cancer and Lifestyle. “At the same time, there are a lot of things that could explain these associations. People who can afford lots of plant-based foods tend to have higher socioeconomic status, for example.”
What’s needed, Freedland said, are more randomized trials to test the hypotheses emerging from the longitudinal cohort studies. “That’s where I’m going with my own research,” he said. “I’d like to look at a study like [one of these] and design a trial. Let’s say we get half of patients to eat according to the healthy eating index, while half eat whatever they want. Can dietary modification change which genes are turned on and off in a tumor, as a start?”
Oncologist and Nutritionist Collaborate on Multiple Studies
Nutritionist Pao-Hwa Lin, PhD, of Duke University in Durham, North Carolina, has been working for several years with Freedland on trials of nutrition interventions. A longtime researcher of chronic disease and diet, she first collaborated with Freedland on a study, published in 2019, that looked at whether insulin could be driven down with diet and exercise in men treated with androgen deprivation therapy.
Not only are high levels of insulin a known contributor to prostate cancer growth, Lin said, but “insulin resistance is a very common side effect of hormone therapy. And we saw that the low carb diet was very helpful for that.” The finding led Freedland and Lin to design further trials investigating carbohydrate restriction in people with prostate cancer.
Lin said randomized trials tend to be smaller and shorter in duration than the observational cohort studies because “interventions like these can be hard to maintain, and recruitment can be hard to sustain. A very well controlled and intensive nutrition intervention is not going to be super long.” Short trial durations also mean that prostate cancer progression can be difficult to capture. Risk for progression has to be measured using surrogate markers, such as the doubling time for prostate-specific antigen (PSA).
In 2020, Freedland and Lin published results from a pilot study of 57 men who had been treated with surgery or radiation for localized prostate cancer but had a PSA recurrence and were randomized to an ultralow-carbohydrate diet or no restrictions for 6 months. The investigators saw that PSA doubling times, an intermediate measure of tumor growth rate, were slower among those consuming the low-carb diet.
Currently they are wrapping up a trial that randomizes men who have been scheduled for radical prostatectomy to daily supplementation with walnuts, a natural source of polyphenols and omega-3 acids. This time, the aim is to determine whether gene expression in tumors changes in response to supplementation.
The researchers are also recruiting for a study in men being treated for metastatic prostate cancer. This study randomizes patients to a fasting-mimicking diet, which is a type of intermittent fasting, or no dietary restrictions for 6 months.
Developed by biologist Valter Longo, PhD, of the University of Southern California, Los Angeles, the fasting-mimicking diet has been shown to boost treatment effects in women with hormone receptor–positive breast cancer. In 2023, Longo and his colleagues published results from a small pilot study of the same diet in men with prostate cancer, reporting some positive metabolic findings.
Longo, who is consulting on Lin and Freedland’s trial, “has proven that the diet is helpful in treatment outcomes for breast cancer. So we connected and decided to test it and see if it’s helpful in prostate cancer as well.”
More Than One Approach Likely to Work
Though Lin and Freedland have focused most of their investigations on carbohydrate restriction, neither dismisses the potential for other dietary approaches to show benefit.
“There are two main schools of thought in terms of the relationship between diet and prostate cancer,” Lin said. “One is the insulin angle, and that’s what we hypothesized when we first tested the low-carb diet. The other is the inflammation angle.”
Studies have shown greater adherence to the HEI — a diet quality indicator that favors grains, fruits, dairy, vegetables, beans, and seafood — or the plant-based diet index to be associated with lower biomarkers of inflammation, she noted.
Insulin resistance, Lin explained, “is also highly related to inflammation.” (Several of the diets being investigated in prostate cancer were originally studied in diabetes.)
Moreover, weight loss caused by low-carb diets — or other healthy diets — can have a positive effect on insulin resistance independent of diet composition. “So it is a very complicated picture — and that doesn’t exclude other pathways that could also be contributing,” she said.
On the surface, a low-carb diet that is heavy in eggs, cheeses, and meats would seem to have little in common with the HEI or a plant-based diet. But Freedland noted that there are commonalities among the approaches being studied. “No one’s promoting eating a lot of simple sugars. No one’s saying eat a lot of processed foods. All of these diets emphasize whole, natural foods,” he said.
Lin hopes that she and Freedland will one day be able to test a diet that is both lower carb and anti-inflammatory in men with prostate cancer. “Why not combine the approaches, have all the good features together?” she asked.
But Freeland pointed out and explained why most clinicians don’t make dietary recommendations to their newly diagnosed patients.
“A new prostate cancer patient already gets easily an hour discussion of treatment options, of pros and cons. Patients often become overwhelmed. And then to extend it further to talk about diet, they’ll end up even more overwhelmed.” Moreover, he said, current evidence offers doctors few take-home messages to deliver besides avoiding sugar and processed foods.
Multiple dietary approaches are likely to prove helpful in prostate cancer, and when the evidence for them is better established, patients and their doctors will want to consider lifestyle factors in choosing one. The best diet will depend on a patient’s philosophy, tastes, and willingness to follow it, he concluded.
“At the end of the day I’m not rooting for one diet or another. I just want to get the answers.”
Lin disclosed no financial conflicts of interest. Freedland disclosed serving as a speaker for AstraZeneca, Astellas, and Pfizer and as a consultant for Astellas, AstraZeneca, Bayer, Eli Lilly, Janssen, Merck, Novartis, Pfizer, Sanofi-Aventis, and Sumitomo.
A version of this article first appeared on Medscape.com.
Recent studies have shown that ultralow-carbohydrate diets, weight loss diets, supplementation with omega-3 fatty acids, pro- and anti-inflammatory diets, fasting, and even tea drinking may affect prostate cancer risk or risk for progression.
In October, a cohort study involving about 900 men under active surveillance for early stage prostate cancers found that those who reported eating a diet that adhered closely to the US government’s recommendations as indicated by the Healthy Eating Index (HEI) saw a lower risk for progression at a median 6.5 months follow-up.
These findings follow results from an observational study, published in May, that followed about 2000 men with locally advanced prostate tumors. Men consuming a primarily plant-based diet (one closely adhering to the plant-based diet index) had less likelihood of progression over a median 6.5 years than those consuming diets low in plant-based foods.
“There is an increasing body of literature that says your diet matters,” said urologist Stephen J. Freedland, MD, of Cedars-Sinai Medical Center in Los Angeles, California, and director of its Center for Integrated Research in Cancer and Lifestyle. “At the same time, there are a lot of things that could explain these associations. People who can afford lots of plant-based foods tend to have higher socioeconomic status, for example.”
What’s needed, Freedland said, are more randomized trials to test the hypotheses emerging from the longitudinal cohort studies. “That’s where I’m going with my own research,” he said. “I’d like to look at a study like [one of these] and design a trial. Let’s say we get half of patients to eat according to the healthy eating index, while half eat whatever they want. Can dietary modification change which genes are turned on and off in a tumor, as a start?”
Oncologist and Nutritionist Collaborate on Multiple Studies
Nutritionist Pao-Hwa Lin, PhD, of Duke University in Durham, North Carolina, has been working for several years with Freedland on trials of nutrition interventions. A longtime researcher of chronic disease and diet, she first collaborated with Freedland on a study, published in 2019, that looked at whether insulin could be driven down with diet and exercise in men treated with androgen deprivation therapy.
Not only are high levels of insulin a known contributor to prostate cancer growth, Lin said, but “insulin resistance is a very common side effect of hormone therapy. And we saw that the low carb diet was very helpful for that.” The finding led Freedland and Lin to design further trials investigating carbohydrate restriction in people with prostate cancer.
Lin said randomized trials tend to be smaller and shorter in duration than the observational cohort studies because “interventions like these can be hard to maintain, and recruitment can be hard to sustain. A very well controlled and intensive nutrition intervention is not going to be super long.” Short trial durations also mean that prostate cancer progression can be difficult to capture. Risk for progression has to be measured using surrogate markers, such as the doubling time for prostate-specific antigen (PSA).
In 2020, Freedland and Lin published results from a pilot study of 57 men who had been treated with surgery or radiation for localized prostate cancer but had a PSA recurrence and were randomized to an ultralow-carbohydrate diet or no restrictions for 6 months. The investigators saw that PSA doubling times, an intermediate measure of tumor growth rate, were slower among those consuming the low-carb diet.
Currently they are wrapping up a trial that randomizes men who have been scheduled for radical prostatectomy to daily supplementation with walnuts, a natural source of polyphenols and omega-3 acids. This time, the aim is to determine whether gene expression in tumors changes in response to supplementation.
The researchers are also recruiting for a study in men being treated for metastatic prostate cancer. This study randomizes patients to a fasting-mimicking diet, which is a type of intermittent fasting, or no dietary restrictions for 6 months.
Developed by biologist Valter Longo, PhD, of the University of Southern California, Los Angeles, the fasting-mimicking diet has been shown to boost treatment effects in women with hormone receptor–positive breast cancer. In 2023, Longo and his colleagues published results from a small pilot study of the same diet in men with prostate cancer, reporting some positive metabolic findings.
Longo, who is consulting on Lin and Freedland’s trial, “has proven that the diet is helpful in treatment outcomes for breast cancer. So we connected and decided to test it and see if it’s helpful in prostate cancer as well.”
More Than One Approach Likely to Work
Though Lin and Freedland have focused most of their investigations on carbohydrate restriction, neither dismisses the potential for other dietary approaches to show benefit.
“There are two main schools of thought in terms of the relationship between diet and prostate cancer,” Lin said. “One is the insulin angle, and that’s what we hypothesized when we first tested the low-carb diet. The other is the inflammation angle.”
Studies have shown greater adherence to the HEI — a diet quality indicator that favors grains, fruits, dairy, vegetables, beans, and seafood — or the plant-based diet index to be associated with lower biomarkers of inflammation, she noted.
Insulin resistance, Lin explained, “is also highly related to inflammation.” (Several of the diets being investigated in prostate cancer were originally studied in diabetes.)
Moreover, weight loss caused by low-carb diets — or other healthy diets — can have a positive effect on insulin resistance independent of diet composition. “So it is a very complicated picture — and that doesn’t exclude other pathways that could also be contributing,” she said.
On the surface, a low-carb diet that is heavy in eggs, cheeses, and meats would seem to have little in common with the HEI or a plant-based diet. But Freedland noted that there are commonalities among the approaches being studied. “No one’s promoting eating a lot of simple sugars. No one’s saying eat a lot of processed foods. All of these diets emphasize whole, natural foods,” he said.
Lin hopes that she and Freedland will one day be able to test a diet that is both lower carb and anti-inflammatory in men with prostate cancer. “Why not combine the approaches, have all the good features together?” she asked.
But Freeland pointed out and explained why most clinicians don’t make dietary recommendations to their newly diagnosed patients.
“A new prostate cancer patient already gets easily an hour discussion of treatment options, of pros and cons. Patients often become overwhelmed. And then to extend it further to talk about diet, they’ll end up even more overwhelmed.” Moreover, he said, current evidence offers doctors few take-home messages to deliver besides avoiding sugar and processed foods.
Multiple dietary approaches are likely to prove helpful in prostate cancer, and when the evidence for them is better established, patients and their doctors will want to consider lifestyle factors in choosing one. The best diet will depend on a patient’s philosophy, tastes, and willingness to follow it, he concluded.
“At the end of the day I’m not rooting for one diet or another. I just want to get the answers.”
Lin disclosed no financial conflicts of interest. Freedland disclosed serving as a speaker for AstraZeneca, Astellas, and Pfizer and as a consultant for Astellas, AstraZeneca, Bayer, Eli Lilly, Janssen, Merck, Novartis, Pfizer, Sanofi-Aventis, and Sumitomo.
A version of this article first appeared on Medscape.com.