User login
MDedge latest news is breaking news from medical conferences, journals, guidelines, the FDA and CDC.
Model May Predict Which UC Patients Risk Rehospitalization
a preliminary modeling study suggests.
“Absence of a gastroenterologist consultation within the year prior to admission, male sex, shorter length of hospital stay, and narcotic prescription at the time of discharge were independently associated with the risk for 90-day rehospitalization for a UC-related indication,” study author Sanjay Murthy, MD, associate professor of gastroenterology at the University of Ottawa, Ontario, Canada, and staff gastroenterologist at the Inflammatory Bowel Disease Centre at The Ottawa Hospital, said in an interview.
“While some hospital readmissions are likely unavoidable, a subset of them, particularly readmissions that occur soon after discharge, may be preventable with early and intensive postdischarge outpatient management,” he said. “Identifying those who are at high risk for early readmission is a rational first step toward applying targeted outpatient interventions that reduce this risk.”
The study was published in The Journal of the Canadian Association of Gastroenterology.
Major Predictor Variables
The researchers conducted a retrospective study in adults with UC who were admitted to The Ottawa Hospital between 2009 and 2016 for a UC flare or UC-related complication, excluding bowel cancer. Using medical records and administrative health databases, they derived and validated a multivariable logistic regression model of 90-day UC-related rehospitalization risk.
Participants’ mean age at UC diagnosis was 35.3 years and 50.4% were men. In the year before the index hospitalization, 138 (55.6%) participants had a gastroenterologist visit, whereas 41 (16.5%) were hospitalized.
During the index hospitalization, 42 (16.9%) patients were newly diagnosed with UC, and 25 (10.1%) underwent intra-abdominal surgery. At discharge, 34 (13.7%) patients were prescribed an outpatient narcotic. The mean length of hospital stay was 9.97 days. Twenty-seven individuals (10.9%) were rehospitalized within 90 days of discharge.
Out of 35 variables, the model identified the following four as significant predictors of 90-day rehospitalization: gastroenterologist consultation within the prior year (adjusted odds ratio [aOR], 0.09), male sex (aOR, 3.77), length of hospital stay (aOR, 0.93), and discharge with narcotics prescription (aOR, 5.94).
The model had 77.8% sensitivity, 80.9% specificity, 33% positive predictive value, and 96.7% negative predictive value for predicting high vs low risk for 90-day hospital readmission.
The researchers noted several study limitations. The cohort was relatively small, which limited the statistical power for model building and identifying variable associations with the outcome. In addition, the study was conducted in a single tertiary care center, which limits its generalizability. Retrospective data may have affected the accuracy of the measurements, and information on some relevant variables was not available.
Nevertheless, Murthy said, “optimally applying our prediction model at the point of hospital discharge would have classified only about a quarter of individuals in our cohort as being at high-risk for 90-day readmission and potentially needing targeted early outpatient intervention, and this would have captured close to 80% of individuals who were destined for early readmission.”
“However, our research is still preliminary and requires considerably more work to ensure that the findings are suitable for application to clinical practice,” he added. “In the meantime, practitioners may reflect on the potential importance of the major predictor variables identified in our study within their practices.”
Careful Follow-Up Key
Rajiv Bhuta, MD, assistant professor of clinical gastroenterology and hepatology at Temple University and a gastroenterologist at Temple University Hospital, both in Philadelphia, Pennsylvania, commented on the study but was not involved in it.
“The model performed fairly well (c-statistic of 0.78) using four variables: Gastroenterologist consultation within the prior year (protective), male sex (higher risk), length of stay (marginally protective), and narcotic prescription at discharge (higher risk). These are intuitive predictors that align with prior literature on UC hospitalizations,” said Bhuta.
“From a clinical perspective, this type of tool could be useful for targeting high-risk patients for early outpatient interventions (eg, close gastroenterology follow-up and pain management strategies). The negative predictive value (96.7%) suggests that it is particularly good at identifying patients at low risk for rehospitalization, which may help prioritize resource allocation more efficiently. However, practical implementation will require external validation and integration into electronic medical records to automatically flag high-risk patients at discharge.”
In addition, Bhuta noted, “the study only examines patient data through 2016. Why have the last 8 years been excluded? Given the small sample size and the sea change in available inflammatory bowel disease therapies since 2016, there could be significantly different findings with more current data.”
Furthermore, there is a lack of specific data supporting the protective effect of a gastroenterology visit in the previous year, and the readmission rate was lower than that reported by others (10% vs 20%), which, he said “may skew their findings.”
“The strong protective effect of prior gastroenterologist visits underscores the importance of specialty proactive disease management in these complex patients,” Bhuta continued. “Narcotic prescriptions at discharge may indicate inadequate disease activity control, thus making these patients important targets for close follow-up. Narcotics are generally not required once successful disease control has been achieved with steroids or biologics.
“While promising, this tool should not yet replace clinical judgment until it undergoes external validation,” he concluded. “In the meantime, clinicians should focus on structured outpatient follow-up and careful discharge planning to minimize UC-related rehospitalizations.”
This study was funded by a grant provided to Murthy by the department of medicine at the University of Ottawa. Murthy and Bhuta declared having no relevant financial relationships.
A version of this article appeared on Medscape.com .
a preliminary modeling study suggests.
“Absence of a gastroenterologist consultation within the year prior to admission, male sex, shorter length of hospital stay, and narcotic prescription at the time of discharge were independently associated with the risk for 90-day rehospitalization for a UC-related indication,” study author Sanjay Murthy, MD, associate professor of gastroenterology at the University of Ottawa, Ontario, Canada, and staff gastroenterologist at the Inflammatory Bowel Disease Centre at The Ottawa Hospital, said in an interview.
“While some hospital readmissions are likely unavoidable, a subset of them, particularly readmissions that occur soon after discharge, may be preventable with early and intensive postdischarge outpatient management,” he said. “Identifying those who are at high risk for early readmission is a rational first step toward applying targeted outpatient interventions that reduce this risk.”
The study was published in The Journal of the Canadian Association of Gastroenterology.
Major Predictor Variables
The researchers conducted a retrospective study in adults with UC who were admitted to The Ottawa Hospital between 2009 and 2016 for a UC flare or UC-related complication, excluding bowel cancer. Using medical records and administrative health databases, they derived and validated a multivariable logistic regression model of 90-day UC-related rehospitalization risk.
Participants’ mean age at UC diagnosis was 35.3 years and 50.4% were men. In the year before the index hospitalization, 138 (55.6%) participants had a gastroenterologist visit, whereas 41 (16.5%) were hospitalized.
During the index hospitalization, 42 (16.9%) patients were newly diagnosed with UC, and 25 (10.1%) underwent intra-abdominal surgery. At discharge, 34 (13.7%) patients were prescribed an outpatient narcotic. The mean length of hospital stay was 9.97 days. Twenty-seven individuals (10.9%) were rehospitalized within 90 days of discharge.
Out of 35 variables, the model identified the following four as significant predictors of 90-day rehospitalization: gastroenterologist consultation within the prior year (adjusted odds ratio [aOR], 0.09), male sex (aOR, 3.77), length of hospital stay (aOR, 0.93), and discharge with narcotics prescription (aOR, 5.94).
The model had 77.8% sensitivity, 80.9% specificity, 33% positive predictive value, and 96.7% negative predictive value for predicting high vs low risk for 90-day hospital readmission.
The researchers noted several study limitations. The cohort was relatively small, which limited the statistical power for model building and identifying variable associations with the outcome. In addition, the study was conducted in a single tertiary care center, which limits its generalizability. Retrospective data may have affected the accuracy of the measurements, and information on some relevant variables was not available.
Nevertheless, Murthy said, “optimally applying our prediction model at the point of hospital discharge would have classified only about a quarter of individuals in our cohort as being at high-risk for 90-day readmission and potentially needing targeted early outpatient intervention, and this would have captured close to 80% of individuals who were destined for early readmission.”
“However, our research is still preliminary and requires considerably more work to ensure that the findings are suitable for application to clinical practice,” he added. “In the meantime, practitioners may reflect on the potential importance of the major predictor variables identified in our study within their practices.”
Careful Follow-Up Key
Rajiv Bhuta, MD, assistant professor of clinical gastroenterology and hepatology at Temple University and a gastroenterologist at Temple University Hospital, both in Philadelphia, Pennsylvania, commented on the study but was not involved in it.
“The model performed fairly well (c-statistic of 0.78) using four variables: Gastroenterologist consultation within the prior year (protective), male sex (higher risk), length of stay (marginally protective), and narcotic prescription at discharge (higher risk). These are intuitive predictors that align with prior literature on UC hospitalizations,” said Bhuta.
“From a clinical perspective, this type of tool could be useful for targeting high-risk patients for early outpatient interventions (eg, close gastroenterology follow-up and pain management strategies). The negative predictive value (96.7%) suggests that it is particularly good at identifying patients at low risk for rehospitalization, which may help prioritize resource allocation more efficiently. However, practical implementation will require external validation and integration into electronic medical records to automatically flag high-risk patients at discharge.”
In addition, Bhuta noted, “the study only examines patient data through 2016. Why have the last 8 years been excluded? Given the small sample size and the sea change in available inflammatory bowel disease therapies since 2016, there could be significantly different findings with more current data.”
Furthermore, there is a lack of specific data supporting the protective effect of a gastroenterology visit in the previous year, and the readmission rate was lower than that reported by others (10% vs 20%), which, he said “may skew their findings.”
“The strong protective effect of prior gastroenterologist visits underscores the importance of specialty proactive disease management in these complex patients,” Bhuta continued. “Narcotic prescriptions at discharge may indicate inadequate disease activity control, thus making these patients important targets for close follow-up. Narcotics are generally not required once successful disease control has been achieved with steroids or biologics.
“While promising, this tool should not yet replace clinical judgment until it undergoes external validation,” he concluded. “In the meantime, clinicians should focus on structured outpatient follow-up and careful discharge planning to minimize UC-related rehospitalizations.”
This study was funded by a grant provided to Murthy by the department of medicine at the University of Ottawa. Murthy and Bhuta declared having no relevant financial relationships.
A version of this article appeared on Medscape.com .
a preliminary modeling study suggests.
“Absence of a gastroenterologist consultation within the year prior to admission, male sex, shorter length of hospital stay, and narcotic prescription at the time of discharge were independently associated with the risk for 90-day rehospitalization for a UC-related indication,” study author Sanjay Murthy, MD, associate professor of gastroenterology at the University of Ottawa, Ontario, Canada, and staff gastroenterologist at the Inflammatory Bowel Disease Centre at The Ottawa Hospital, said in an interview.
“While some hospital readmissions are likely unavoidable, a subset of them, particularly readmissions that occur soon after discharge, may be preventable with early and intensive postdischarge outpatient management,” he said. “Identifying those who are at high risk for early readmission is a rational first step toward applying targeted outpatient interventions that reduce this risk.”
The study was published in The Journal of the Canadian Association of Gastroenterology.
Major Predictor Variables
The researchers conducted a retrospective study in adults with UC who were admitted to The Ottawa Hospital between 2009 and 2016 for a UC flare or UC-related complication, excluding bowel cancer. Using medical records and administrative health databases, they derived and validated a multivariable logistic regression model of 90-day UC-related rehospitalization risk.
Participants’ mean age at UC diagnosis was 35.3 years and 50.4% were men. In the year before the index hospitalization, 138 (55.6%) participants had a gastroenterologist visit, whereas 41 (16.5%) were hospitalized.
During the index hospitalization, 42 (16.9%) patients were newly diagnosed with UC, and 25 (10.1%) underwent intra-abdominal surgery. At discharge, 34 (13.7%) patients were prescribed an outpatient narcotic. The mean length of hospital stay was 9.97 days. Twenty-seven individuals (10.9%) were rehospitalized within 90 days of discharge.
Out of 35 variables, the model identified the following four as significant predictors of 90-day rehospitalization: gastroenterologist consultation within the prior year (adjusted odds ratio [aOR], 0.09), male sex (aOR, 3.77), length of hospital stay (aOR, 0.93), and discharge with narcotics prescription (aOR, 5.94).
The model had 77.8% sensitivity, 80.9% specificity, 33% positive predictive value, and 96.7% negative predictive value for predicting high vs low risk for 90-day hospital readmission.
The researchers noted several study limitations. The cohort was relatively small, which limited the statistical power for model building and identifying variable associations with the outcome. In addition, the study was conducted in a single tertiary care center, which limits its generalizability. Retrospective data may have affected the accuracy of the measurements, and information on some relevant variables was not available.
Nevertheless, Murthy said, “optimally applying our prediction model at the point of hospital discharge would have classified only about a quarter of individuals in our cohort as being at high-risk for 90-day readmission and potentially needing targeted early outpatient intervention, and this would have captured close to 80% of individuals who were destined for early readmission.”
“However, our research is still preliminary and requires considerably more work to ensure that the findings are suitable for application to clinical practice,” he added. “In the meantime, practitioners may reflect on the potential importance of the major predictor variables identified in our study within their practices.”
Careful Follow-Up Key
Rajiv Bhuta, MD, assistant professor of clinical gastroenterology and hepatology at Temple University and a gastroenterologist at Temple University Hospital, both in Philadelphia, Pennsylvania, commented on the study but was not involved in it.
“The model performed fairly well (c-statistic of 0.78) using four variables: Gastroenterologist consultation within the prior year (protective), male sex (higher risk), length of stay (marginally protective), and narcotic prescription at discharge (higher risk). These are intuitive predictors that align with prior literature on UC hospitalizations,” said Bhuta.
“From a clinical perspective, this type of tool could be useful for targeting high-risk patients for early outpatient interventions (eg, close gastroenterology follow-up and pain management strategies). The negative predictive value (96.7%) suggests that it is particularly good at identifying patients at low risk for rehospitalization, which may help prioritize resource allocation more efficiently. However, practical implementation will require external validation and integration into electronic medical records to automatically flag high-risk patients at discharge.”
In addition, Bhuta noted, “the study only examines patient data through 2016. Why have the last 8 years been excluded? Given the small sample size and the sea change in available inflammatory bowel disease therapies since 2016, there could be significantly different findings with more current data.”
Furthermore, there is a lack of specific data supporting the protective effect of a gastroenterology visit in the previous year, and the readmission rate was lower than that reported by others (10% vs 20%), which, he said “may skew their findings.”
“The strong protective effect of prior gastroenterologist visits underscores the importance of specialty proactive disease management in these complex patients,” Bhuta continued. “Narcotic prescriptions at discharge may indicate inadequate disease activity control, thus making these patients important targets for close follow-up. Narcotics are generally not required once successful disease control has been achieved with steroids or biologics.
“While promising, this tool should not yet replace clinical judgment until it undergoes external validation,” he concluded. “In the meantime, clinicians should focus on structured outpatient follow-up and careful discharge planning to minimize UC-related rehospitalizations.”
This study was funded by a grant provided to Murthy by the department of medicine at the University of Ottawa. Murthy and Bhuta declared having no relevant financial relationships.
A version of this article appeared on Medscape.com .
FROM THE JOURNAL OF THE CANADIAN ASSOCIATION OF GASTROENTEROLOGY
More Layoffs at VA and Other Health Agencies
The large-scale layoffs in the federal government that began in January continue, as the US Department of Veterans Affairs (VA) announced the dismissal of > 1400 employees in “non-mission critical roles,” including those “related to DEI” (diversity, equity, inclusion) on Feb. 24. According to VA, those fired are bargaining-unit probationary employees who have served > 1 year in a competitive service appointment or who have served > 2 years in an excepted service appointment.
The agency says the “personnel moves” will save > $83 million annually, which will be redirected back toward health care, benefits and services for VA beneficiaries.
Of the nearly 40,000 probationary employees in the department, the majority were exempt, the VA says, because they serve in mission-critical positions—primarily those supporting benefits and services for VA beneficiaries, such as Veterans Crisis Line responders. VA employees who elected to participate in the Office of Personnel Management’s (OPM) deferred resignation program are also exempt. As an “additional safeguard,” the VA says the first Senior Executive Service (SES) or SES-equivalent leader in a dismissed employee’s chain of command can request the employee be exempted from removal.
The latest cuts follow the dismissal of > 1000 employees announced Feb. 13. In that case, the VA expected to save > $98 million annually, also to be “redirected back” toward health care, benefits, and services. VA insists it continues to hire for mission-critical positions that are exempt from the federal hiring freeze.
Layoffs are also impacting other federal public health agencies. Although the White House has not released figures, a ProPublica investigation details the impact of the layoffs on organ transplant and maternal mortality programs. Other layoffs that have been reported include :
- About 750 workers at the Centers for Disease Control and Prevention
- More than 1000 staffers at the National Institutes of Health
- “Dozens” at the Centers for Medicare and Medicaid Services
- “Scores” at the US Food and Drug Administration
- Downsizing at the VA EHR Modernization Integration Office
“By gutting essential health staff, hiding vital public health data, and silencing health experts, these actions have left every American family more vulnerable to deadly disease outbreaks, unsafe food and water, and preventable deaths,” the American Public Health Association said in a press release. “This is also not just an attack on federal institutions – it's a direct attack on every parent trying to protect their child from disease, every worker relying on public health safeguards and every family depending on rapid responses to outbreaks and emergencies.” American Public Health Association also announced that is suing the Department of Government Efficiency for violating federal transparency laws. “It is unfathomable that anybody thinks these cuts have value and are doing anything other than being performative.”
In 2024, the VA had planned to trim its 458,000-member workforce by about 2%, or 10,000 employees, through attrition (with most of the reduction coming from VHA). VHA Chief Financial Officer Laura Duke told reporters in March 2024 that the reduction was needed because the agency had far exceeded its hiring goals last year, and was also seeing higher-than-expected retention rates.
“These and other recent personnel decisions are extraordinarily difficult, but VA is focused on allocating its resources to help as many veterans, families, caregivers, and survivors as possible,” VA Secretary Doug Collins said. “These moves will not hurt VA health care, benefits or beneficiaries. In fact, veterans are going to notice a change for the better. In the coming weeks and months, VA will be announcing plans to put these resources to work helping the department fulfill its core mission: providing the best possible care and benefits to veterans, their families, caregivers and survivors.”
Senate Veterans’ Affairs Committee Ranking Member Richard Blumenthal (D-CT) and a group of 35 Democratic senators signed a letter earlier in February calling for Sec. Collins to immediately reinstate the terminated VA employees. “[W]e were outraged,” the letter said, “by the Administration’s abrupt and indiscriminate termination of tens of thousands of workers across almost every government agency, including more than 1000 Department of Veterans Affairs (VA) employees. We were further disturbed by the manner in which you publicly celebrated this reprehensible announcement—a clear departure from the assurances provided throughout your confirmation process to never ‘balance budgets on the back of veterans’ benefits’ and to always ‘put the veteran first.’”
Blumenthal also notes that the “continued mass terminations” come at a time when the VA faces critical staffing shortages and increased demand for its services. The senators detailed the effects the cuts were having, including how openings for new clinics were delayed because the VA cannot hire the necessary staff to open their doors; service lines at VA hospitals and clinics halted; beds and operating rooms at VA facilities suspended; support lines for caregivers reduced; Veterans Crisis Line employees fired; and suicide prevention training sessions postponed or canceled.
The large-scale layoffs in the federal government that began in January continue, as the US Department of Veterans Affairs (VA) announced the dismissal of > 1400 employees in “non-mission critical roles,” including those “related to DEI” (diversity, equity, inclusion) on Feb. 24. According to VA, those fired are bargaining-unit probationary employees who have served > 1 year in a competitive service appointment or who have served > 2 years in an excepted service appointment.
The agency says the “personnel moves” will save > $83 million annually, which will be redirected back toward health care, benefits and services for VA beneficiaries.
Of the nearly 40,000 probationary employees in the department, the majority were exempt, the VA says, because they serve in mission-critical positions—primarily those supporting benefits and services for VA beneficiaries, such as Veterans Crisis Line responders. VA employees who elected to participate in the Office of Personnel Management’s (OPM) deferred resignation program are also exempt. As an “additional safeguard,” the VA says the first Senior Executive Service (SES) or SES-equivalent leader in a dismissed employee’s chain of command can request the employee be exempted from removal.
The latest cuts follow the dismissal of > 1000 employees announced Feb. 13. In that case, the VA expected to save > $98 million annually, also to be “redirected back” toward health care, benefits, and services. VA insists it continues to hire for mission-critical positions that are exempt from the federal hiring freeze.
Layoffs are also impacting other federal public health agencies. Although the White House has not released figures, a ProPublica investigation details the impact of the layoffs on organ transplant and maternal mortality programs. Other layoffs that have been reported include :
- About 750 workers at the Centers for Disease Control and Prevention
- More than 1000 staffers at the National Institutes of Health
- “Dozens” at the Centers for Medicare and Medicaid Services
- “Scores” at the US Food and Drug Administration
- Downsizing at the VA EHR Modernization Integration Office
“By gutting essential health staff, hiding vital public health data, and silencing health experts, these actions have left every American family more vulnerable to deadly disease outbreaks, unsafe food and water, and preventable deaths,” the American Public Health Association said in a press release. “This is also not just an attack on federal institutions – it's a direct attack on every parent trying to protect their child from disease, every worker relying on public health safeguards and every family depending on rapid responses to outbreaks and emergencies.” American Public Health Association also announced that is suing the Department of Government Efficiency for violating federal transparency laws. “It is unfathomable that anybody thinks these cuts have value and are doing anything other than being performative.”
In 2024, the VA had planned to trim its 458,000-member workforce by about 2%, or 10,000 employees, through attrition (with most of the reduction coming from VHA). VHA Chief Financial Officer Laura Duke told reporters in March 2024 that the reduction was needed because the agency had far exceeded its hiring goals last year, and was also seeing higher-than-expected retention rates.
“These and other recent personnel decisions are extraordinarily difficult, but VA is focused on allocating its resources to help as many veterans, families, caregivers, and survivors as possible,” VA Secretary Doug Collins said. “These moves will not hurt VA health care, benefits or beneficiaries. In fact, veterans are going to notice a change for the better. In the coming weeks and months, VA will be announcing plans to put these resources to work helping the department fulfill its core mission: providing the best possible care and benefits to veterans, their families, caregivers and survivors.”
Senate Veterans’ Affairs Committee Ranking Member Richard Blumenthal (D-CT) and a group of 35 Democratic senators signed a letter earlier in February calling for Sec. Collins to immediately reinstate the terminated VA employees. “[W]e were outraged,” the letter said, “by the Administration’s abrupt and indiscriminate termination of tens of thousands of workers across almost every government agency, including more than 1000 Department of Veterans Affairs (VA) employees. We were further disturbed by the manner in which you publicly celebrated this reprehensible announcement—a clear departure from the assurances provided throughout your confirmation process to never ‘balance budgets on the back of veterans’ benefits’ and to always ‘put the veteran first.’”
Blumenthal also notes that the “continued mass terminations” come at a time when the VA faces critical staffing shortages and increased demand for its services. The senators detailed the effects the cuts were having, including how openings for new clinics were delayed because the VA cannot hire the necessary staff to open their doors; service lines at VA hospitals and clinics halted; beds and operating rooms at VA facilities suspended; support lines for caregivers reduced; Veterans Crisis Line employees fired; and suicide prevention training sessions postponed or canceled.
The large-scale layoffs in the federal government that began in January continue, as the US Department of Veterans Affairs (VA) announced the dismissal of > 1400 employees in “non-mission critical roles,” including those “related to DEI” (diversity, equity, inclusion) on Feb. 24. According to VA, those fired are bargaining-unit probationary employees who have served > 1 year in a competitive service appointment or who have served > 2 years in an excepted service appointment.
The agency says the “personnel moves” will save > $83 million annually, which will be redirected back toward health care, benefits and services for VA beneficiaries.
Of the nearly 40,000 probationary employees in the department, the majority were exempt, the VA says, because they serve in mission-critical positions—primarily those supporting benefits and services for VA beneficiaries, such as Veterans Crisis Line responders. VA employees who elected to participate in the Office of Personnel Management’s (OPM) deferred resignation program are also exempt. As an “additional safeguard,” the VA says the first Senior Executive Service (SES) or SES-equivalent leader in a dismissed employee’s chain of command can request the employee be exempted from removal.
The latest cuts follow the dismissal of > 1000 employees announced Feb. 13. In that case, the VA expected to save > $98 million annually, also to be “redirected back” toward health care, benefits, and services. VA insists it continues to hire for mission-critical positions that are exempt from the federal hiring freeze.
Layoffs are also impacting other federal public health agencies. Although the White House has not released figures, a ProPublica investigation details the impact of the layoffs on organ transplant and maternal mortality programs. Other layoffs that have been reported include :
- About 750 workers at the Centers for Disease Control and Prevention
- More than 1000 staffers at the National Institutes of Health
- “Dozens” at the Centers for Medicare and Medicaid Services
- “Scores” at the US Food and Drug Administration
- Downsizing at the VA EHR Modernization Integration Office
“By gutting essential health staff, hiding vital public health data, and silencing health experts, these actions have left every American family more vulnerable to deadly disease outbreaks, unsafe food and water, and preventable deaths,” the American Public Health Association said in a press release. “This is also not just an attack on federal institutions – it's a direct attack on every parent trying to protect their child from disease, every worker relying on public health safeguards and every family depending on rapid responses to outbreaks and emergencies.” American Public Health Association also announced that is suing the Department of Government Efficiency for violating federal transparency laws. “It is unfathomable that anybody thinks these cuts have value and are doing anything other than being performative.”
In 2024, the VA had planned to trim its 458,000-member workforce by about 2%, or 10,000 employees, through attrition (with most of the reduction coming from VHA). VHA Chief Financial Officer Laura Duke told reporters in March 2024 that the reduction was needed because the agency had far exceeded its hiring goals last year, and was also seeing higher-than-expected retention rates.
“These and other recent personnel decisions are extraordinarily difficult, but VA is focused on allocating its resources to help as many veterans, families, caregivers, and survivors as possible,” VA Secretary Doug Collins said. “These moves will not hurt VA health care, benefits or beneficiaries. In fact, veterans are going to notice a change for the better. In the coming weeks and months, VA will be announcing plans to put these resources to work helping the department fulfill its core mission: providing the best possible care and benefits to veterans, their families, caregivers and survivors.”
Senate Veterans’ Affairs Committee Ranking Member Richard Blumenthal (D-CT) and a group of 35 Democratic senators signed a letter earlier in February calling for Sec. Collins to immediately reinstate the terminated VA employees. “[W]e were outraged,” the letter said, “by the Administration’s abrupt and indiscriminate termination of tens of thousands of workers across almost every government agency, including more than 1000 Department of Veterans Affairs (VA) employees. We were further disturbed by the manner in which you publicly celebrated this reprehensible announcement—a clear departure from the assurances provided throughout your confirmation process to never ‘balance budgets on the back of veterans’ benefits’ and to always ‘put the veteran first.’”
Blumenthal also notes that the “continued mass terminations” come at a time when the VA faces critical staffing shortages and increased demand for its services. The senators detailed the effects the cuts were having, including how openings for new clinics were delayed because the VA cannot hire the necessary staff to open their doors; service lines at VA hospitals and clinics halted; beds and operating rooms at VA facilities suspended; support lines for caregivers reduced; Veterans Crisis Line employees fired; and suicide prevention training sessions postponed or canceled.
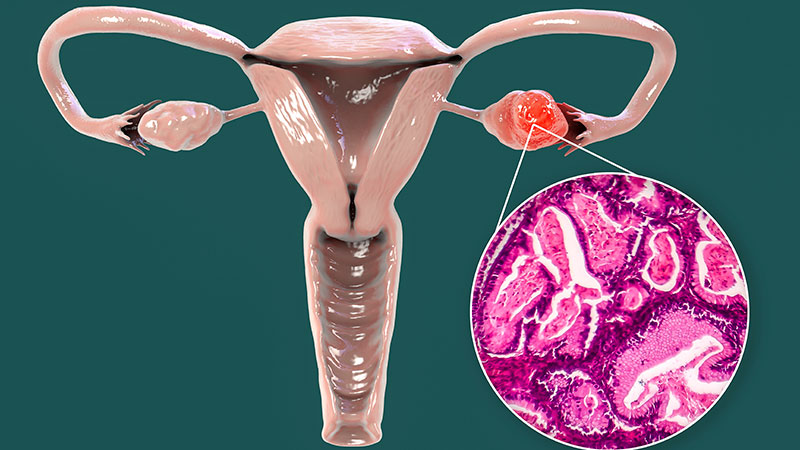
ASCO Updates Treatment Guidance for Newly Diagnosed, Advanced Ovarian Cancer
The American Society of Clinical Oncology (ASCO) has released updated guidelines for neoadjuvant chemotherapy in newly diagnosed advanced ovarian cancer, introducing changes in patient selection and treatment strategies. The changes reflect emerging evidence on racial disparities, treatment outcomes, and quality of life considerations.
The publication of the new guidance follows dramatic shifts in treatment patterns over the past decade.
“There had been a big shift in how we were treating patients in the United States,” explained Stephanie Gaillard, MD, PhD, one of the authors of the updated guidelines. “We saw a substantial drop in the number of patients undergoing primary cytoreductive surgery for ovarian cancer from about 70% of patients in 2010 to only about 37% in 2021.”
The new guidelines maintain the recommendation for platinum/taxane-based neoadjuvant chemotherapy but introduce modifications regarding timing and duration.
“It’s still a recommendation that gynecologic oncologists are involved in determining whether someone is eligible for primary cytoreductive surgery or should undergo neoadjuvant chemotherapy first,” Gaillard noted. “We emphasize that patients who are eligible for primary cytoreductive surgery should undergo surgery as opposed to receiving neoadjuvant chemotherapy.”
Alexander Melamed, MD, MPH, a gynecologic oncologist at Massachusetts General Hospital, Boston, who was not involved in authoring the updated guidelines, noted that additional evidence-based guidance is needed to individualize treatment plans. He pointed to four completed trials comparing neoadjuvant chemotherapy with cytoreductive surgery, noting: “When these trials have been pooled together in meta-analyses, there was a higher risk of mortality associated with primary cytoreductive surgery and a higher risk of severe complications.”
The updated guidelines take this higher risk for mortality with primary cytoreductive surgery into consideration, and patients who are not eligible for primary surgery would receive neoadjuvant chemotherapy, Gaillard noted.
Changes in Patient Selection
The 2025 guidelines describe a more nuanced approach for selecting patients for neoadjuvant chemotherapy vs primary cytoreductive surgery. While the 2016 ASCO guidelines primarily focused on disease burden and surgical resectability when selecting patients for neoadjuvant chemotherapy, the new recommendations incorporate additional factors.
The guidelines discuss recent findings showing that Black patients experience a 38% lower likelihood of undergoing cytoreductive surgery than non-Black patients. In addition, compared with non-Hispanic White women, Asian and Black women more frequently receive neoadjuvant chemotherapy with interval debulking surgery rather than primary cytoreductive surgery. According to the authors, these differences persist even after accounting for clinical factors, suggesting that structural barriers to healthcare access may play a role.
The guidelines discuss how affordability, availability, and accessibility mediate racial disparities in ovarian cancer care. According to the authors, structural inequities in healthcare access influence treatment quality for minority patients. Non-White patients face greater challenges in accessing gynecologic oncology consultations and standard-of-care combination therapy, leading to poorer survival outcomes, the guidelines say.
According to Melamed, the guidelines serve as an important tool for promoting healthcare equity. “Having recommendations and standards is incredibly important for achieving equity because once there is consensus on a best practice, it doesn’t matter if you’re rich, poor, or a patient of a particular racial or ethnic group — if you have the disease, you ought to have access to that standard,” he said.
The 2016 ASCO guidelines focused primarily on disease burden and surgical resectability, whereas the 2024 National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines for ovarian cancer focus more on oncologic outcomes and surgical considerations. Based on the NCCN guidelines, treatment selection for ovarian cancer is primarily determined by the histologic subtype, stage of disease, and whether the patient is a candidate for primary surgery. The 2025 ASCO guidelines, on the other hand, emphasize the importance of quality-of-life outcomes during treatment selection. The authors of the updated ASCO guidelines acknowledged that treatment decisions should consider both the duration and quality of life, particularly for elderly patients or those with multiple comorbidities.
Treatment Timing and Duration
The guidelines maintain the recommendations for platinum/taxane-based neoadjuvant chemotherapy described in the previous ASCO guidelines but introduce modifications regarding treatment timing and duration. The optimal window for interval cytoreductive surgery now falls after three to four chemotherapy cycles, allowing more individualized approaches based on patient response and tolerance.
In addition, postsurgical chemotherapy protocols have become more flexible. Rather than mandating a fixed number of cycles, the guidelines encourage tailoring treatment duration to individual patient factors including response assessment, performance status, and quality-of-life considerations.
The updated guidelines also emphasize the importance of genetic and molecular testing at diagnosis, which Melamed identifies as “absolutely central to treatment and deciding who receives maintenance therapy.” This is also recommended by the NCCN guidelines.
However, he highlighted the following practical challenge in molecular testing after neoadjuvant chemotherapy. “Probably 20% of patients have an exceptional response to neoadjuvant therapy, such that there is insufficient tissue at the time of their cytoreduction to do somatic testing,” he said.
Hyperthermic Intraperitoneal Chemotherapy (HIPEC)
A notable difference between the 2016 and 2025 guidelines is the inclusion of HIPEC in the updated guidelines.
Commenting to this news organization, Gaillard explained the nuanced approach to HIPEC: “The committee discussed HIPEC extensively. We recognize that it may not be available at many centers and requires specially trained staff and dedicated resources. The reason for including HIPEC in the guidelines is to highlight that there have been studies that show a potential overall survival benefit.”
Melamed considers the recommendation of HIPEC to be one of the strongest aspects of the updated guidelines. “There have been two large trials and one smaller one that have shown that for patients treated with neoadjuvant chemotherapy, the addition of HIPEC appears to improve overall survival,” he explained.
Implementation Strategies
The authors acknowledged that barriers to healthcare delivery present significant challenges to the implementation of the guidelines. Limited access to gynecologic oncologists in rural areas, insurance coverage gaps, and varying surgical expertise across institutions complicate the delivery of optimal care. The guidelines also emphasize the need for solutions to ensure equitable access to recommended treatments.
Melamed noted that the decentralized structure of the healthcare system in the United States complicates the uniform adoption of guidelines, particularly in resource-limited settings, adding that “geographic region and local resources and expertise influence both access to treatment and outcomes.”
Although both the updated ASCO guidelines and NCCN guidelines emphasize the importance of evaluation by a gynecologic oncologist for determining the most appropriate treatment strategy, the scarcity of gynecologic oncologists is one of the most significant barriers to accessing optimal care, according to Gaillard. She proposes telemedicine consultations and enhanced communication between medical oncologists and gynecologic oncologists to ensure equitable access.
Gaillard also commented on the challenges in implementing a multidisciplinary treatment approach, the importance of which is emphasized in the updated guidelines.
“There can be a limited availability of the multidisciplinary team to be involved in this decision-making,” she said. “Ideally, patient assessment by a gynecologic oncologist would happen in person, but recognizing that availability is limited, it doesn’t necessarily have to. Sometimes, it can just be a conversation between a medical oncologist and a gynecologic oncologist detailing a treatment plan together.”
Looking Ahead
Gaillard noted that ovarian cancer is a very active field of research and that the guidelines may need to be updated again in the near future to incorporate novel treatment approaches.
“Newer and more effective targeted therapies based on tumor profiling are being developed,” she said. “These will hopefully move earlier in the treatment course for patients. Maybe we will not use chemotherapy in the future because we will have more directed and targeted therapies.”
She also emphasized the importance of early diagnosis in shaping future treatment guidelines for ovarian cancer.
“Neoadjuvant chemotherapy is predominantly used in situations where patients have very advanced disease and may not benefit from primary cytoreductive surgery,” she noted. “If we develop better diagnostic tools that will allow us to diagnose patients earlier, then we may not need to use neoadjuvant chemotherapy.”
All funding for the administration of the guideline development project was provided by ASCO. Gaillard reported receiving consulting or advisory fees from Verastem, Merck, AstraZeneca, and Compugen; research funding from AstraZeneca, Tesaro, Compugen, Genentech/Roche, Clovis Oncology, Tempest Therapeutics, Blueprint Pharmaceutic, Immunogen, Volastra Therapeutics, and Beigene; and patents, royalties, or other intellectual property from US Patent Nos 10,258,604 and 10,905,659, licensed by Duke University to Sermonix. Melamed reported receiving research funding from the National Cancer Institute and the National Institutes of Health.
A version of this article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) has released updated guidelines for neoadjuvant chemotherapy in newly diagnosed advanced ovarian cancer, introducing changes in patient selection and treatment strategies. The changes reflect emerging evidence on racial disparities, treatment outcomes, and quality of life considerations.
The publication of the new guidance follows dramatic shifts in treatment patterns over the past decade.
“There had been a big shift in how we were treating patients in the United States,” explained Stephanie Gaillard, MD, PhD, one of the authors of the updated guidelines. “We saw a substantial drop in the number of patients undergoing primary cytoreductive surgery for ovarian cancer from about 70% of patients in 2010 to only about 37% in 2021.”
The new guidelines maintain the recommendation for platinum/taxane-based neoadjuvant chemotherapy but introduce modifications regarding timing and duration.
“It’s still a recommendation that gynecologic oncologists are involved in determining whether someone is eligible for primary cytoreductive surgery or should undergo neoadjuvant chemotherapy first,” Gaillard noted. “We emphasize that patients who are eligible for primary cytoreductive surgery should undergo surgery as opposed to receiving neoadjuvant chemotherapy.”
Alexander Melamed, MD, MPH, a gynecologic oncologist at Massachusetts General Hospital, Boston, who was not involved in authoring the updated guidelines, noted that additional evidence-based guidance is needed to individualize treatment plans. He pointed to four completed trials comparing neoadjuvant chemotherapy with cytoreductive surgery, noting: “When these trials have been pooled together in meta-analyses, there was a higher risk of mortality associated with primary cytoreductive surgery and a higher risk of severe complications.”
The updated guidelines take this higher risk for mortality with primary cytoreductive surgery into consideration, and patients who are not eligible for primary surgery would receive neoadjuvant chemotherapy, Gaillard noted.
Changes in Patient Selection
The 2025 guidelines describe a more nuanced approach for selecting patients for neoadjuvant chemotherapy vs primary cytoreductive surgery. While the 2016 ASCO guidelines primarily focused on disease burden and surgical resectability when selecting patients for neoadjuvant chemotherapy, the new recommendations incorporate additional factors.
The guidelines discuss recent findings showing that Black patients experience a 38% lower likelihood of undergoing cytoreductive surgery than non-Black patients. In addition, compared with non-Hispanic White women, Asian and Black women more frequently receive neoadjuvant chemotherapy with interval debulking surgery rather than primary cytoreductive surgery. According to the authors, these differences persist even after accounting for clinical factors, suggesting that structural barriers to healthcare access may play a role.
The guidelines discuss how affordability, availability, and accessibility mediate racial disparities in ovarian cancer care. According to the authors, structural inequities in healthcare access influence treatment quality for minority patients. Non-White patients face greater challenges in accessing gynecologic oncology consultations and standard-of-care combination therapy, leading to poorer survival outcomes, the guidelines say.
According to Melamed, the guidelines serve as an important tool for promoting healthcare equity. “Having recommendations and standards is incredibly important for achieving equity because once there is consensus on a best practice, it doesn’t matter if you’re rich, poor, or a patient of a particular racial or ethnic group — if you have the disease, you ought to have access to that standard,” he said.
The 2016 ASCO guidelines focused primarily on disease burden and surgical resectability, whereas the 2024 National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines for ovarian cancer focus more on oncologic outcomes and surgical considerations. Based on the NCCN guidelines, treatment selection for ovarian cancer is primarily determined by the histologic subtype, stage of disease, and whether the patient is a candidate for primary surgery. The 2025 ASCO guidelines, on the other hand, emphasize the importance of quality-of-life outcomes during treatment selection. The authors of the updated ASCO guidelines acknowledged that treatment decisions should consider both the duration and quality of life, particularly for elderly patients or those with multiple comorbidities.
Treatment Timing and Duration
The guidelines maintain the recommendations for platinum/taxane-based neoadjuvant chemotherapy described in the previous ASCO guidelines but introduce modifications regarding treatment timing and duration. The optimal window for interval cytoreductive surgery now falls after three to four chemotherapy cycles, allowing more individualized approaches based on patient response and tolerance.
In addition, postsurgical chemotherapy protocols have become more flexible. Rather than mandating a fixed number of cycles, the guidelines encourage tailoring treatment duration to individual patient factors including response assessment, performance status, and quality-of-life considerations.
The updated guidelines also emphasize the importance of genetic and molecular testing at diagnosis, which Melamed identifies as “absolutely central to treatment and deciding who receives maintenance therapy.” This is also recommended by the NCCN guidelines.
However, he highlighted the following practical challenge in molecular testing after neoadjuvant chemotherapy. “Probably 20% of patients have an exceptional response to neoadjuvant therapy, such that there is insufficient tissue at the time of their cytoreduction to do somatic testing,” he said.
Hyperthermic Intraperitoneal Chemotherapy (HIPEC)
A notable difference between the 2016 and 2025 guidelines is the inclusion of HIPEC in the updated guidelines.
Commenting to this news organization, Gaillard explained the nuanced approach to HIPEC: “The committee discussed HIPEC extensively. We recognize that it may not be available at many centers and requires specially trained staff and dedicated resources. The reason for including HIPEC in the guidelines is to highlight that there have been studies that show a potential overall survival benefit.”
Melamed considers the recommendation of HIPEC to be one of the strongest aspects of the updated guidelines. “There have been two large trials and one smaller one that have shown that for patients treated with neoadjuvant chemotherapy, the addition of HIPEC appears to improve overall survival,” he explained.
Implementation Strategies
The authors acknowledged that barriers to healthcare delivery present significant challenges to the implementation of the guidelines. Limited access to gynecologic oncologists in rural areas, insurance coverage gaps, and varying surgical expertise across institutions complicate the delivery of optimal care. The guidelines also emphasize the need for solutions to ensure equitable access to recommended treatments.
Melamed noted that the decentralized structure of the healthcare system in the United States complicates the uniform adoption of guidelines, particularly in resource-limited settings, adding that “geographic region and local resources and expertise influence both access to treatment and outcomes.”
Although both the updated ASCO guidelines and NCCN guidelines emphasize the importance of evaluation by a gynecologic oncologist for determining the most appropriate treatment strategy, the scarcity of gynecologic oncologists is one of the most significant barriers to accessing optimal care, according to Gaillard. She proposes telemedicine consultations and enhanced communication between medical oncologists and gynecologic oncologists to ensure equitable access.
Gaillard also commented on the challenges in implementing a multidisciplinary treatment approach, the importance of which is emphasized in the updated guidelines.
“There can be a limited availability of the multidisciplinary team to be involved in this decision-making,” she said. “Ideally, patient assessment by a gynecologic oncologist would happen in person, but recognizing that availability is limited, it doesn’t necessarily have to. Sometimes, it can just be a conversation between a medical oncologist and a gynecologic oncologist detailing a treatment plan together.”
Looking Ahead
Gaillard noted that ovarian cancer is a very active field of research and that the guidelines may need to be updated again in the near future to incorporate novel treatment approaches.
“Newer and more effective targeted therapies based on tumor profiling are being developed,” she said. “These will hopefully move earlier in the treatment course for patients. Maybe we will not use chemotherapy in the future because we will have more directed and targeted therapies.”
She also emphasized the importance of early diagnosis in shaping future treatment guidelines for ovarian cancer.
“Neoadjuvant chemotherapy is predominantly used in situations where patients have very advanced disease and may not benefit from primary cytoreductive surgery,” she noted. “If we develop better diagnostic tools that will allow us to diagnose patients earlier, then we may not need to use neoadjuvant chemotherapy.”
All funding for the administration of the guideline development project was provided by ASCO. Gaillard reported receiving consulting or advisory fees from Verastem, Merck, AstraZeneca, and Compugen; research funding from AstraZeneca, Tesaro, Compugen, Genentech/Roche, Clovis Oncology, Tempest Therapeutics, Blueprint Pharmaceutic, Immunogen, Volastra Therapeutics, and Beigene; and patents, royalties, or other intellectual property from US Patent Nos 10,258,604 and 10,905,659, licensed by Duke University to Sermonix. Melamed reported receiving research funding from the National Cancer Institute and the National Institutes of Health.
A version of this article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) has released updated guidelines for neoadjuvant chemotherapy in newly diagnosed advanced ovarian cancer, introducing changes in patient selection and treatment strategies. The changes reflect emerging evidence on racial disparities, treatment outcomes, and quality of life considerations.
The publication of the new guidance follows dramatic shifts in treatment patterns over the past decade.
“There had been a big shift in how we were treating patients in the United States,” explained Stephanie Gaillard, MD, PhD, one of the authors of the updated guidelines. “We saw a substantial drop in the number of patients undergoing primary cytoreductive surgery for ovarian cancer from about 70% of patients in 2010 to only about 37% in 2021.”
The new guidelines maintain the recommendation for platinum/taxane-based neoadjuvant chemotherapy but introduce modifications regarding timing and duration.
“It’s still a recommendation that gynecologic oncologists are involved in determining whether someone is eligible for primary cytoreductive surgery or should undergo neoadjuvant chemotherapy first,” Gaillard noted. “We emphasize that patients who are eligible for primary cytoreductive surgery should undergo surgery as opposed to receiving neoadjuvant chemotherapy.”
Alexander Melamed, MD, MPH, a gynecologic oncologist at Massachusetts General Hospital, Boston, who was not involved in authoring the updated guidelines, noted that additional evidence-based guidance is needed to individualize treatment plans. He pointed to four completed trials comparing neoadjuvant chemotherapy with cytoreductive surgery, noting: “When these trials have been pooled together in meta-analyses, there was a higher risk of mortality associated with primary cytoreductive surgery and a higher risk of severe complications.”
The updated guidelines take this higher risk for mortality with primary cytoreductive surgery into consideration, and patients who are not eligible for primary surgery would receive neoadjuvant chemotherapy, Gaillard noted.
Changes in Patient Selection
The 2025 guidelines describe a more nuanced approach for selecting patients for neoadjuvant chemotherapy vs primary cytoreductive surgery. While the 2016 ASCO guidelines primarily focused on disease burden and surgical resectability when selecting patients for neoadjuvant chemotherapy, the new recommendations incorporate additional factors.
The guidelines discuss recent findings showing that Black patients experience a 38% lower likelihood of undergoing cytoreductive surgery than non-Black patients. In addition, compared with non-Hispanic White women, Asian and Black women more frequently receive neoadjuvant chemotherapy with interval debulking surgery rather than primary cytoreductive surgery. According to the authors, these differences persist even after accounting for clinical factors, suggesting that structural barriers to healthcare access may play a role.
The guidelines discuss how affordability, availability, and accessibility mediate racial disparities in ovarian cancer care. According to the authors, structural inequities in healthcare access influence treatment quality for minority patients. Non-White patients face greater challenges in accessing gynecologic oncology consultations and standard-of-care combination therapy, leading to poorer survival outcomes, the guidelines say.
According to Melamed, the guidelines serve as an important tool for promoting healthcare equity. “Having recommendations and standards is incredibly important for achieving equity because once there is consensus on a best practice, it doesn’t matter if you’re rich, poor, or a patient of a particular racial or ethnic group — if you have the disease, you ought to have access to that standard,” he said.
The 2016 ASCO guidelines focused primarily on disease burden and surgical resectability, whereas the 2024 National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines for ovarian cancer focus more on oncologic outcomes and surgical considerations. Based on the NCCN guidelines, treatment selection for ovarian cancer is primarily determined by the histologic subtype, stage of disease, and whether the patient is a candidate for primary surgery. The 2025 ASCO guidelines, on the other hand, emphasize the importance of quality-of-life outcomes during treatment selection. The authors of the updated ASCO guidelines acknowledged that treatment decisions should consider both the duration and quality of life, particularly for elderly patients or those with multiple comorbidities.
Treatment Timing and Duration
The guidelines maintain the recommendations for platinum/taxane-based neoadjuvant chemotherapy described in the previous ASCO guidelines but introduce modifications regarding treatment timing and duration. The optimal window for interval cytoreductive surgery now falls after three to four chemotherapy cycles, allowing more individualized approaches based on patient response and tolerance.
In addition, postsurgical chemotherapy protocols have become more flexible. Rather than mandating a fixed number of cycles, the guidelines encourage tailoring treatment duration to individual patient factors including response assessment, performance status, and quality-of-life considerations.
The updated guidelines also emphasize the importance of genetic and molecular testing at diagnosis, which Melamed identifies as “absolutely central to treatment and deciding who receives maintenance therapy.” This is also recommended by the NCCN guidelines.
However, he highlighted the following practical challenge in molecular testing after neoadjuvant chemotherapy. “Probably 20% of patients have an exceptional response to neoadjuvant therapy, such that there is insufficient tissue at the time of their cytoreduction to do somatic testing,” he said.
Hyperthermic Intraperitoneal Chemotherapy (HIPEC)
A notable difference between the 2016 and 2025 guidelines is the inclusion of HIPEC in the updated guidelines.
Commenting to this news organization, Gaillard explained the nuanced approach to HIPEC: “The committee discussed HIPEC extensively. We recognize that it may not be available at many centers and requires specially trained staff and dedicated resources. The reason for including HIPEC in the guidelines is to highlight that there have been studies that show a potential overall survival benefit.”
Melamed considers the recommendation of HIPEC to be one of the strongest aspects of the updated guidelines. “There have been two large trials and one smaller one that have shown that for patients treated with neoadjuvant chemotherapy, the addition of HIPEC appears to improve overall survival,” he explained.
Implementation Strategies
The authors acknowledged that barriers to healthcare delivery present significant challenges to the implementation of the guidelines. Limited access to gynecologic oncologists in rural areas, insurance coverage gaps, and varying surgical expertise across institutions complicate the delivery of optimal care. The guidelines also emphasize the need for solutions to ensure equitable access to recommended treatments.
Melamed noted that the decentralized structure of the healthcare system in the United States complicates the uniform adoption of guidelines, particularly in resource-limited settings, adding that “geographic region and local resources and expertise influence both access to treatment and outcomes.”
Although both the updated ASCO guidelines and NCCN guidelines emphasize the importance of evaluation by a gynecologic oncologist for determining the most appropriate treatment strategy, the scarcity of gynecologic oncologists is one of the most significant barriers to accessing optimal care, according to Gaillard. She proposes telemedicine consultations and enhanced communication between medical oncologists and gynecologic oncologists to ensure equitable access.
Gaillard also commented on the challenges in implementing a multidisciplinary treatment approach, the importance of which is emphasized in the updated guidelines.
“There can be a limited availability of the multidisciplinary team to be involved in this decision-making,” she said. “Ideally, patient assessment by a gynecologic oncologist would happen in person, but recognizing that availability is limited, it doesn’t necessarily have to. Sometimes, it can just be a conversation between a medical oncologist and a gynecologic oncologist detailing a treatment plan together.”
Looking Ahead
Gaillard noted that ovarian cancer is a very active field of research and that the guidelines may need to be updated again in the near future to incorporate novel treatment approaches.
“Newer and more effective targeted therapies based on tumor profiling are being developed,” she said. “These will hopefully move earlier in the treatment course for patients. Maybe we will not use chemotherapy in the future because we will have more directed and targeted therapies.”
She also emphasized the importance of early diagnosis in shaping future treatment guidelines for ovarian cancer.
“Neoadjuvant chemotherapy is predominantly used in situations where patients have very advanced disease and may not benefit from primary cytoreductive surgery,” she noted. “If we develop better diagnostic tools that will allow us to diagnose patients earlier, then we may not need to use neoadjuvant chemotherapy.”
All funding for the administration of the guideline development project was provided by ASCO. Gaillard reported receiving consulting or advisory fees from Verastem, Merck, AstraZeneca, and Compugen; research funding from AstraZeneca, Tesaro, Compugen, Genentech/Roche, Clovis Oncology, Tempest Therapeutics, Blueprint Pharmaceutic, Immunogen, Volastra Therapeutics, and Beigene; and patents, royalties, or other intellectual property from US Patent Nos 10,258,604 and 10,905,659, licensed by Duke University to Sermonix. Melamed reported receiving research funding from the National Cancer Institute and the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Choosing the Ideal Endoscopic Enteral Access Method: AGA Practice Update
At least 250,000 US hospitalized patients a year require enteral support using an artificial pathway into the gastrointestinal (GI) tract to deliver nutrition or medication. In light of this,
Covering indications, placement techniques, and management, the comprehensive document is a response to the increasing use of enteral access devices in chronic GI conditions. The update, published in Gastroenterology, addresses patient factors complicating placement decision-making such as thrombocytopenia, use of dual antiplatelet therapy, or performance of percutaneous access in the setting of cirrhosis.
“We provide clinical recommendations in these various scenarios understanding that the final decision-making is in the hands of the provider and care team,” said first author Dejan Micic, MD, a gastroenterologist and associate professor at University of Chicago Medical Center in Illinois at the time of the update (since relocated to Loyola University Medical Center in Chicago). “We hope this can serve a day-to-day purpose for clinical gastroenterologists and can be referenced as they encounter individuals with or needing an enteral access device.”
Traditionally, enteral access was reserved for patients with severe malnutrition or those unable to maintain oral intake. Recent recommendations emphasize early nutritional intervention including prehabilitation before major surgery, adjunctive therapy for oncology patients, and in specific inflammatory conditions such as Crohn’s disease. “These shifts recognize the role of enteral nutrition not only in preventing malnutrition but also as a therapeutic strategy,” Micic said in an interview.
There is, however, variability in the use of devices including the selection of appropriate units, technical aspects of placement, and subsequent management. “Such variability can lead to complications, suboptimal patient outcomes, and inefficiencies in care delivery,” Micic said.
He added that enteral access has been historically underemphasized in GI endoscopic training. “While procedural skill in placing devices such as percutaneous endoscopic gastrostomy, or PEG, tubes is often taught, a comprehensive understanding of the broader clinical context — such as proper patient selection, prevention of complications, and postplacement care — is not always thoroughly covered.”
The current update aims to bridge knowledge gaps with evidence-based-guidance. “It also underscores the importance of interdisciplinary collaboration with dietitians, nurses, and care givers to achieve the best outcomes for patients,” Micic said.
Commenting on the update but not involved with creating it, Shirley C. Paski, MD, MS, a gastroenterologist at the Cleveland Clinic, Ohio, called it timely, adding: “As GI training is becoming more subspecialized and interventional radiology has been able to provide enteral access, gastroenterology training in enteral access has declined to where some fellows are graduating with limited enteral access experience.”
Yet malnutrition remains a common consequence when GI disease is severe, chronic, or refractory to treatment, or in the setting of postsurgical anatomy, she added. “Enteral nutrition is increasingly being considered a therapeutic or adjunct treatment in some cases of Crohn’s disease or small intestinal bacterial overgrowth. Gastroenterologists need the endoscopic skill to secure enteral access tubes, particularly in more challenging anatomy.”
Also commenting on the document but not involved in it, Steven Shamah, MD, director of Endoscopy at Northwell Lenox Hill Hospital in New York City, said: “This should serve as a concise review for any general hospitalist or gastroenterologist to understand what we have and when we should offer the proper feeding tube options.” He stressed, however, that all gastroenterologists should be trained in the placing of all of tube options.
“The axiom ‘If the gut works, we should use it’ is something that I was taught when I was a medical student and it still holds true,” Shamah continued. “There’s been a jump in interventional procedures to assure continuity of the GI tract even in progressive malignancy. So there’s a rise in moving away from intravenous nutrition and a rise in tube-delivered enteral nutrition.” Options for reducing reflux and aspiration will likely take on more importance, he said.
Tubing Options
According to Micic and colleagues, recent data suggest a favorable safety profile of enteral feeding tubes placed endoscopically compared with surgical or radiologic placement. The illustrated AGA document outlines such approaches as synthetic flexible tubes placed into the stomach or small bowel via the oral (orogastric and oroenteral) or nasal routes (nasogastric [NG] and nasojejunal [NJ]) and percutaneous tubes accessing the stomach. The choice of tube, access point, delivery site, and feeding method varies with indication, expected duration of use, and patient anatomy, the authors stressed.
The update notes that NG and NJ tubes can be used immediately after confirmation of placement, most often with abdominal radiography. PEG tubes can be used immediately for medications and after 4 hours for tube feedings. A multidisciplinary team approach after placement provides improved patient care. “Dietitians assist with formula choice, volume, free water needs, and delivery method, and nurses and advanced practice clinicians assist with tube site assessment and troubleshooting,” the authors wrote.
Complications can occur but should be infrequent, Micic said. “Frankly, most complications can be predicted based on the duration of use and prevented with appropriate monitoring.” Common complications include tube dislodgement, clogging, site infections, buried bumper syndrome, and aspiration. “Minimizing these risks requires a thorough understanding of patient-specific factors, careful technique during placement, and ongoing monitoring after the device is in use,” he added.
Paski said the update aligns with established guidelines for enteral access but also offers suggestions to mitigate the risk of tube placement in patients in whom placement has traditionally been more challenging. “This is a helpful addition to the literature because if enteral access cannot be obtained in a patient unable to meet their needs orally, total paternal nutrition is the next and much more invasive step for nutrition support.”
She called the practice update a concise, comprehensive reference for trainees and experienced gastroenterologists to optimize placement conditions and reduce complication risk, noting that training in nutrition is suboptimal in many GI fellowships.
Becoming familiar with common and advanced enteral access techniques is within the armamentarium of all practicing gastroenterologists, the authors stated. Because malnutrition affects nearly all GI disorders, “understanding common routes of enteral access and the basic principles of nutrition support promotes the initiation of optimal enteral nutrition, mitigating the impact of malnutrition, and improving prognosis for patients at nutritional risk,” they wrote.
Micic served on the advisory board for Ironwood Pharmaceuticals and is on the speaker’s bureau for Takeda Pharmaceuticals. One coauthor served as a consultant for Merit Medical, Circa Scientific, and Aspero Medical. Paski and Shamah had disclosed no competing interests relevant to their comments.
A version of this article appeared on Medscape.com.
At least 250,000 US hospitalized patients a year require enteral support using an artificial pathway into the gastrointestinal (GI) tract to deliver nutrition or medication. In light of this,
Covering indications, placement techniques, and management, the comprehensive document is a response to the increasing use of enteral access devices in chronic GI conditions. The update, published in Gastroenterology, addresses patient factors complicating placement decision-making such as thrombocytopenia, use of dual antiplatelet therapy, or performance of percutaneous access in the setting of cirrhosis.
“We provide clinical recommendations in these various scenarios understanding that the final decision-making is in the hands of the provider and care team,” said first author Dejan Micic, MD, a gastroenterologist and associate professor at University of Chicago Medical Center in Illinois at the time of the update (since relocated to Loyola University Medical Center in Chicago). “We hope this can serve a day-to-day purpose for clinical gastroenterologists and can be referenced as they encounter individuals with or needing an enteral access device.”
Traditionally, enteral access was reserved for patients with severe malnutrition or those unable to maintain oral intake. Recent recommendations emphasize early nutritional intervention including prehabilitation before major surgery, adjunctive therapy for oncology patients, and in specific inflammatory conditions such as Crohn’s disease. “These shifts recognize the role of enteral nutrition not only in preventing malnutrition but also as a therapeutic strategy,” Micic said in an interview.
There is, however, variability in the use of devices including the selection of appropriate units, technical aspects of placement, and subsequent management. “Such variability can lead to complications, suboptimal patient outcomes, and inefficiencies in care delivery,” Micic said.
He added that enteral access has been historically underemphasized in GI endoscopic training. “While procedural skill in placing devices such as percutaneous endoscopic gastrostomy, or PEG, tubes is often taught, a comprehensive understanding of the broader clinical context — such as proper patient selection, prevention of complications, and postplacement care — is not always thoroughly covered.”
The current update aims to bridge knowledge gaps with evidence-based-guidance. “It also underscores the importance of interdisciplinary collaboration with dietitians, nurses, and care givers to achieve the best outcomes for patients,” Micic said.
Commenting on the update but not involved with creating it, Shirley C. Paski, MD, MS, a gastroenterologist at the Cleveland Clinic, Ohio, called it timely, adding: “As GI training is becoming more subspecialized and interventional radiology has been able to provide enteral access, gastroenterology training in enteral access has declined to where some fellows are graduating with limited enteral access experience.”
Yet malnutrition remains a common consequence when GI disease is severe, chronic, or refractory to treatment, or in the setting of postsurgical anatomy, she added. “Enteral nutrition is increasingly being considered a therapeutic or adjunct treatment in some cases of Crohn’s disease or small intestinal bacterial overgrowth. Gastroenterologists need the endoscopic skill to secure enteral access tubes, particularly in more challenging anatomy.”
Also commenting on the document but not involved in it, Steven Shamah, MD, director of Endoscopy at Northwell Lenox Hill Hospital in New York City, said: “This should serve as a concise review for any general hospitalist or gastroenterologist to understand what we have and when we should offer the proper feeding tube options.” He stressed, however, that all gastroenterologists should be trained in the placing of all of tube options.
“The axiom ‘If the gut works, we should use it’ is something that I was taught when I was a medical student and it still holds true,” Shamah continued. “There’s been a jump in interventional procedures to assure continuity of the GI tract even in progressive malignancy. So there’s a rise in moving away from intravenous nutrition and a rise in tube-delivered enteral nutrition.” Options for reducing reflux and aspiration will likely take on more importance, he said.
Tubing Options
According to Micic and colleagues, recent data suggest a favorable safety profile of enteral feeding tubes placed endoscopically compared with surgical or radiologic placement. The illustrated AGA document outlines such approaches as synthetic flexible tubes placed into the stomach or small bowel via the oral (orogastric and oroenteral) or nasal routes (nasogastric [NG] and nasojejunal [NJ]) and percutaneous tubes accessing the stomach. The choice of tube, access point, delivery site, and feeding method varies with indication, expected duration of use, and patient anatomy, the authors stressed.
The update notes that NG and NJ tubes can be used immediately after confirmation of placement, most often with abdominal radiography. PEG tubes can be used immediately for medications and after 4 hours for tube feedings. A multidisciplinary team approach after placement provides improved patient care. “Dietitians assist with formula choice, volume, free water needs, and delivery method, and nurses and advanced practice clinicians assist with tube site assessment and troubleshooting,” the authors wrote.
Complications can occur but should be infrequent, Micic said. “Frankly, most complications can be predicted based on the duration of use and prevented with appropriate monitoring.” Common complications include tube dislodgement, clogging, site infections, buried bumper syndrome, and aspiration. “Minimizing these risks requires a thorough understanding of patient-specific factors, careful technique during placement, and ongoing monitoring after the device is in use,” he added.
Paski said the update aligns with established guidelines for enteral access but also offers suggestions to mitigate the risk of tube placement in patients in whom placement has traditionally been more challenging. “This is a helpful addition to the literature because if enteral access cannot be obtained in a patient unable to meet their needs orally, total paternal nutrition is the next and much more invasive step for nutrition support.”
She called the practice update a concise, comprehensive reference for trainees and experienced gastroenterologists to optimize placement conditions and reduce complication risk, noting that training in nutrition is suboptimal in many GI fellowships.
Becoming familiar with common and advanced enteral access techniques is within the armamentarium of all practicing gastroenterologists, the authors stated. Because malnutrition affects nearly all GI disorders, “understanding common routes of enteral access and the basic principles of nutrition support promotes the initiation of optimal enteral nutrition, mitigating the impact of malnutrition, and improving prognosis for patients at nutritional risk,” they wrote.
Micic served on the advisory board for Ironwood Pharmaceuticals and is on the speaker’s bureau for Takeda Pharmaceuticals. One coauthor served as a consultant for Merit Medical, Circa Scientific, and Aspero Medical. Paski and Shamah had disclosed no competing interests relevant to their comments.
A version of this article appeared on Medscape.com.
At least 250,000 US hospitalized patients a year require enteral support using an artificial pathway into the gastrointestinal (GI) tract to deliver nutrition or medication. In light of this,
Covering indications, placement techniques, and management, the comprehensive document is a response to the increasing use of enteral access devices in chronic GI conditions. The update, published in Gastroenterology, addresses patient factors complicating placement decision-making such as thrombocytopenia, use of dual antiplatelet therapy, or performance of percutaneous access in the setting of cirrhosis.
“We provide clinical recommendations in these various scenarios understanding that the final decision-making is in the hands of the provider and care team,” said first author Dejan Micic, MD, a gastroenterologist and associate professor at University of Chicago Medical Center in Illinois at the time of the update (since relocated to Loyola University Medical Center in Chicago). “We hope this can serve a day-to-day purpose for clinical gastroenterologists and can be referenced as they encounter individuals with or needing an enteral access device.”
Traditionally, enteral access was reserved for patients with severe malnutrition or those unable to maintain oral intake. Recent recommendations emphasize early nutritional intervention including prehabilitation before major surgery, adjunctive therapy for oncology patients, and in specific inflammatory conditions such as Crohn’s disease. “These shifts recognize the role of enteral nutrition not only in preventing malnutrition but also as a therapeutic strategy,” Micic said in an interview.
There is, however, variability in the use of devices including the selection of appropriate units, technical aspects of placement, and subsequent management. “Such variability can lead to complications, suboptimal patient outcomes, and inefficiencies in care delivery,” Micic said.
He added that enteral access has been historically underemphasized in GI endoscopic training. “While procedural skill in placing devices such as percutaneous endoscopic gastrostomy, or PEG, tubes is often taught, a comprehensive understanding of the broader clinical context — such as proper patient selection, prevention of complications, and postplacement care — is not always thoroughly covered.”
The current update aims to bridge knowledge gaps with evidence-based-guidance. “It also underscores the importance of interdisciplinary collaboration with dietitians, nurses, and care givers to achieve the best outcomes for patients,” Micic said.
Commenting on the update but not involved with creating it, Shirley C. Paski, MD, MS, a gastroenterologist at the Cleveland Clinic, Ohio, called it timely, adding: “As GI training is becoming more subspecialized and interventional radiology has been able to provide enteral access, gastroenterology training in enteral access has declined to where some fellows are graduating with limited enteral access experience.”
Yet malnutrition remains a common consequence when GI disease is severe, chronic, or refractory to treatment, or in the setting of postsurgical anatomy, she added. “Enteral nutrition is increasingly being considered a therapeutic or adjunct treatment in some cases of Crohn’s disease or small intestinal bacterial overgrowth. Gastroenterologists need the endoscopic skill to secure enteral access tubes, particularly in more challenging anatomy.”
Also commenting on the document but not involved in it, Steven Shamah, MD, director of Endoscopy at Northwell Lenox Hill Hospital in New York City, said: “This should serve as a concise review for any general hospitalist or gastroenterologist to understand what we have and when we should offer the proper feeding tube options.” He stressed, however, that all gastroenterologists should be trained in the placing of all of tube options.
“The axiom ‘If the gut works, we should use it’ is something that I was taught when I was a medical student and it still holds true,” Shamah continued. “There’s been a jump in interventional procedures to assure continuity of the GI tract even in progressive malignancy. So there’s a rise in moving away from intravenous nutrition and a rise in tube-delivered enteral nutrition.” Options for reducing reflux and aspiration will likely take on more importance, he said.
Tubing Options
According to Micic and colleagues, recent data suggest a favorable safety profile of enteral feeding tubes placed endoscopically compared with surgical or radiologic placement. The illustrated AGA document outlines such approaches as synthetic flexible tubes placed into the stomach or small bowel via the oral (orogastric and oroenteral) or nasal routes (nasogastric [NG] and nasojejunal [NJ]) and percutaneous tubes accessing the stomach. The choice of tube, access point, delivery site, and feeding method varies with indication, expected duration of use, and patient anatomy, the authors stressed.
The update notes that NG and NJ tubes can be used immediately after confirmation of placement, most often with abdominal radiography. PEG tubes can be used immediately for medications and after 4 hours for tube feedings. A multidisciplinary team approach after placement provides improved patient care. “Dietitians assist with formula choice, volume, free water needs, and delivery method, and nurses and advanced practice clinicians assist with tube site assessment and troubleshooting,” the authors wrote.
Complications can occur but should be infrequent, Micic said. “Frankly, most complications can be predicted based on the duration of use and prevented with appropriate monitoring.” Common complications include tube dislodgement, clogging, site infections, buried bumper syndrome, and aspiration. “Minimizing these risks requires a thorough understanding of patient-specific factors, careful technique during placement, and ongoing monitoring after the device is in use,” he added.
Paski said the update aligns with established guidelines for enteral access but also offers suggestions to mitigate the risk of tube placement in patients in whom placement has traditionally been more challenging. “This is a helpful addition to the literature because if enteral access cannot be obtained in a patient unable to meet their needs orally, total paternal nutrition is the next and much more invasive step for nutrition support.”
She called the practice update a concise, comprehensive reference for trainees and experienced gastroenterologists to optimize placement conditions and reduce complication risk, noting that training in nutrition is suboptimal in many GI fellowships.
Becoming familiar with common and advanced enteral access techniques is within the armamentarium of all practicing gastroenterologists, the authors stated. Because malnutrition affects nearly all GI disorders, “understanding common routes of enteral access and the basic principles of nutrition support promotes the initiation of optimal enteral nutrition, mitigating the impact of malnutrition, and improving prognosis for patients at nutritional risk,” they wrote.
Micic served on the advisory board for Ironwood Pharmaceuticals and is on the speaker’s bureau for Takeda Pharmaceuticals. One coauthor served as a consultant for Merit Medical, Circa Scientific, and Aspero Medical. Paski and Shamah had disclosed no competing interests relevant to their comments.
A version of this article appeared on Medscape.com.
FROM GASTROENTEROLOGY
Open Clinical Trials for Patients With Chronic Obstructive Pulmonary Disease
The clinical trials listed below are open as of February 21, 2025; have ≥ 1 US Department of Veterans Affairs (VA) medical center (VAMC) or US Department of Defense (DoD) military treatment facility location recruiting patients; and are focused on treatments for chronic obstructive pulmonary disease (COPD). For additional information and full inclusion/exclusion criteria, please consult clinicaltrials.gov.
Actively Recruiting
The Effect of Interval Exercise on Functional Outcomes in Veterans With COPD and OSA
The term overlap syndrome (OS) is used to describe the presence of both COPD and obstructive sleep apnea (OSA) in a single patient. Due to premature aging, patients with OS are prone to developing functional decline up to 20 years earlier than the general population. The International Classification of Functioning, Disability and Health (ICF) evaluates functional status in chronic pulmonary disease globally in 5 domains. The investigators propose to study validated outcomes in 3 of these domains: (1) participation in life situations; (2) physical activity; and (3) cardiovascular health. The investigators’ long-term goal is to develop an exercise strategy tailored to veterans with OS which will reduce the risk of functional decline through increased physical activity.
ID: NCT05254431
Sponsor; Collaborator: VA Office of Research and Development; Madalina Macrea, MD, PhD
Location: Salem VA Medical Center, Virginia
The Development of an Integrated Physical Activity and Mental Health Intervention for Veterans With COPD, Emotion Distress, and Low Physical Activity
COPD is a prevalent and debilitating chronic disease in veterans. COPD is highly comorbid with depression and anxiety, conferring greater morbidity and mortality risk. Physical activity is a modifiable behavior that can improve COPD outcomes. However, to date, interventions targeting physical activity have not addressed the high comorbidity between COPD and depression and/or anxiety symptoms (emotional distress) despite emotional distress predicting poorer response to physical activity interventions. This CDA-2 proposal will develop and test the acceptability and feasibility of an integrative physical activity and mental health intervention for veterans with COPD, emotional distress, and low physical activity. The intervention will be delivered via VA Video Connect enabling access to care among veterans with substantial barriers to hospital-based outpatient care.
ID: NCT04953806
Sponsor; Collaborator: VA Office of Research and Development; Patricia Bamonti, PhD
Location: VA Boston Healthcare System, Jamaica Plain Campus
Neurocognitive and Health Impact of Sleep Apnea in Elderly Veterans With Comorbid COPD
Cognitive dysfunction in the aging veteran population is a growing health concern in the Veterans Health System. It is not known whether OSA coexisting with COPD will enhance the risk for cognitive dysfunction. The investigators sought to investigate whether these two highly prevalent diseases that often coexist as 'overlap syndrome' combine to enhance cognitive impairment in the elderly veteran population. Thus, the investigators will study whether elderly patients with overlap syndrome have increased cognitive deficits compared with OSA or COPD alone. Additionally, treatment of OSA with positive airway pressure (PAP) has been shown to improve neurocognitive function in moderate-to-severe OSA while cognitive decline in COPD may be reversible through treatment with long-term oxygen therapy. The investigators will also study whether treatment with PAP and supplemental oxygen vs PAP alone will improve cognitive function and improve quality of life of elderly veterans.
ID: NCT02703207
Sponsor; Investigators: VA Office of Research and Development; Susmita Chowdhuri, MD, MS
Locations: John D. Dingell VA Medical Center, Detroit
The Effect of a Technology-Mediated Integrated Walking and Tai Chi Intervention on Physical Function in Veterans With COPD and Chronic Musculoskeletal Pain (WATCH for Pain)
Persons with COPD benefit from being physically active, but they are often limited by chronic musculoskeletal pain. This project will determine whether a non-pharmacologic, integrated, technology-mediated walking and tai chi mindfulness intervention can improve physical function in veterans with COPD and chronic musculoskeletal pain. The proposed research addresses VA Rehabilitation R&D Service's high priority area of improving health-related quality of life by reducing disease burden and maximizing function in veterans with chronic disease.
ID: NCT05701982
Sponsor; Investigator: VA Office of Research and Development; Marilyn L. Moy, MD; University of Michigan, Beth Israel Deaconess Medical Center
Location: VA Boston Healthcare System
Internet-based Cognitive-behavioral Treatment for Insomnia in COPD Patients Undergoing Pulmonary Rehabilitation
This study is a randomized controlled trial (RCT) to compare sleep and health-related functioning in veterans with COPD and insomnia receiving an Internet-based behavioral treatment for insomnia vs online insomnia patient education. Participants will undergo a sleep and health assessment that will be performed at baseline, post-treatment, and 3 months later. Participants will be randomly assigned to either Internet-based behavioral treatment for insomnia or online insomnia patient education.
ID: NCT04700098
Sponsor; Collaborators: VA Office of Research and Development; Faith S. Luyster, PhD
Locations: VA Pittsburgh Healthcare System; John D. Dingell VA Medical Center, Detroit
Breathe Easier With Tadalafil Therapy for Dyspnea in COPD-PH (BETTER COPD-PH)
The investigators will study whether the drug tadalafil improves shortness of breath in 126 veterans with COPD and high blood pressure in the lungs. The investigators will also assess whether tadalafil improves quality of life, home daily physical activity, exercise endurance, the frequency of acute flares of COPD, blood pressure in the lungs, and lung function. Veterans who enroll in the trial will be allocated by chance to either active tadalafil or an inactive identical capsule (placebo). Neither the veteran nor the investigator will know whether the veteran is taking tadalafil or placebo. Veterans will be followed closely in clinic or by telephone at 1, 2, 3, 4, 5, and 6 months, with attention to side effects and safety. At 1,3, and 6 months the investigators will repeat the questionnaires and testing of blood pressures in the lung and lung function. The investigators anticipate that the results of this study will determine whether tadalafil improves shortness of breath when added to usual medications for COPD.
ID: NCT05937854
Sponsor; Collaborator: VA Office of Research and Development; Sharon I. Rounds, MD
Locations: Rocky Mountain Regional VA Medical Center, Colorado; Joseph Maxwell Cleland Atlanta VA Medical Center ; VA Boston Healthcare System Jamaica Plain Campus; VA Nebraska-Western Iowa Health Care System; Providence VA Medical Center
Impact of Positive Airway Pressure Therapy on Clinical Outcomes in Older Veterans With Chronic Obstructive Pulmonary Disease and Comorbid Obstructive Sleep Apnea (Overlap Syndrome)
Obstructive sleep apnea (OSA) and COPD are highly prevalent chronic respiratory diseases in the veteran population. OSA co-occurring with COPD, known as overlap syndrome (OVS), is a complex chronic medical condition associated with grave consequences. OVS is highly prevalent in veterans. Veterans with OVS may be at increased risk for cognitive deficits, poor sleep quality as well as a reduced quality of life (QoL). The overall objective is to study the effects of positive airway pressure therapy on clinical outcomes in patients with OVS.
ID: NCT04179981
Sponsor; Investigator: VA Office of Research and Development; Susmita Chowdhuri, MD, MS
Locations: VA Ann Arbor Healthcare System; John D. Dingell VA Medical Center, Detroit
Developing an Intervention to Optimize Virtual Care Adoption for COPD Management (VC-OPTIONS)
VA is a leader in virtual care (VC), including the patient portal, mobile apps, and telehealth programs. VC has great utility for managing chronic conditions like COPD. However, adoption of many VC services has been slow. Lack of awareness about these services is one of the most prominent patient- and health care team-facing barriers to adopting VC. This study will develop, refine, and pilot a stakeholder-informed multicomponent implementation strategy to support adoption of VC, referred to as VC-OPTIONS (Virtual Care for Chronic Obstructive Pulmonary Disease Adoption Support). This feasibility trial will pilot the VC-OPTIONS implementation strategy to assess feasibility and acceptability and gather preliminary effectiveness data to inform a larger hybrid effectiveness-implementation trial. The core component of VC-OPTIONS will be the provision of information via VA's Annie texting program to empower patients with knowledge about the array of VC services and how they can be used to support COPD management. It is hypothesized that this strategy will be acceptable and feasible. This work will improve patient and team awareness of and communication about VC services, and support patient access to VC services for COPD management.
ID: NCT05986214
Sponsor; Collaborators: VA Office of Research and Development; Stephanie Robinson, PhD
Location: VA Bedford Healthcare System, Massachusetts; VA Boston Healthcare System Jamaica Plain Campus
Chronic Lung Disease and COVID-19: Understanding Severity, Recovery and Rehabilitation Needs (LAUREL)
This study is comprised of 3 approaches. First, the investigators will conduct a retrospective cohort study to determine factors associated with COVID-19 severity and complications and understand COVID-19 outcomes, including all-cause mortality, postdischarge events, and impacts of rehabilitation services (third aim). The second aim is a mixed-method study and follows COVID-19 patients with repeated surveys to determine patient-reported functional outcomes, health recovery, and rehabilitation needs after COVID-19. The investigators will recruit patients and their informal caregivers for interviews to assess their function and rehabilitation needs.
ID: NCT04628039
Sponsor; Collaborators: VA Office of Research and Development; Kristina A. Crothers, MD
Locations: VA Ann Arbor Healthcare System; VA Puget Sound Health Care System, Washington
Accessing Mobility Using Wearable Sensors
This study will examine whether wearable sensors can be used to track changes in cognitive-motor performance in response to a disease or an intervention. The investigators specific aims are twofold, first aim to explore whether and how a clinical condition such as chronic obstructive pulmonary disease (COPD) or congestive heart failure (CHF) may impact motor-cognitive performance measurable using validated wearable devices (eg, LEGSys, BalanSENS, and Frailty Meter). Second, the investigators will explore whether an exercise intervention provided via telemedicine (telerehabilitation) can enhance motor-cognitive performance.
ID: NCT04306588
Sponsor; Collaborators: Baylor College of Medicine, Bijan Najafi, PhD
Locations: Michael E. DeBakey Veterans Affairs Medical Center, Houston
The clinical trials listed below are open as of February 21, 2025; have ≥ 1 US Department of Veterans Affairs (VA) medical center (VAMC) or US Department of Defense (DoD) military treatment facility location recruiting patients; and are focused on treatments for chronic obstructive pulmonary disease (COPD). For additional information and full inclusion/exclusion criteria, please consult clinicaltrials.gov.
Actively Recruiting
The Effect of Interval Exercise on Functional Outcomes in Veterans With COPD and OSA
The term overlap syndrome (OS) is used to describe the presence of both COPD and obstructive sleep apnea (OSA) in a single patient. Due to premature aging, patients with OS are prone to developing functional decline up to 20 years earlier than the general population. The International Classification of Functioning, Disability and Health (ICF) evaluates functional status in chronic pulmonary disease globally in 5 domains. The investigators propose to study validated outcomes in 3 of these domains: (1) participation in life situations; (2) physical activity; and (3) cardiovascular health. The investigators’ long-term goal is to develop an exercise strategy tailored to veterans with OS which will reduce the risk of functional decline through increased physical activity.
ID: NCT05254431
Sponsor; Collaborator: VA Office of Research and Development; Madalina Macrea, MD, PhD
Location: Salem VA Medical Center, Virginia
The Development of an Integrated Physical Activity and Mental Health Intervention for Veterans With COPD, Emotion Distress, and Low Physical Activity
COPD is a prevalent and debilitating chronic disease in veterans. COPD is highly comorbid with depression and anxiety, conferring greater morbidity and mortality risk. Physical activity is a modifiable behavior that can improve COPD outcomes. However, to date, interventions targeting physical activity have not addressed the high comorbidity between COPD and depression and/or anxiety symptoms (emotional distress) despite emotional distress predicting poorer response to physical activity interventions. This CDA-2 proposal will develop and test the acceptability and feasibility of an integrative physical activity and mental health intervention for veterans with COPD, emotional distress, and low physical activity. The intervention will be delivered via VA Video Connect enabling access to care among veterans with substantial barriers to hospital-based outpatient care.
ID: NCT04953806
Sponsor; Collaborator: VA Office of Research and Development; Patricia Bamonti, PhD
Location: VA Boston Healthcare System, Jamaica Plain Campus
Neurocognitive and Health Impact of Sleep Apnea in Elderly Veterans With Comorbid COPD
Cognitive dysfunction in the aging veteran population is a growing health concern in the Veterans Health System. It is not known whether OSA coexisting with COPD will enhance the risk for cognitive dysfunction. The investigators sought to investigate whether these two highly prevalent diseases that often coexist as 'overlap syndrome' combine to enhance cognitive impairment in the elderly veteran population. Thus, the investigators will study whether elderly patients with overlap syndrome have increased cognitive deficits compared with OSA or COPD alone. Additionally, treatment of OSA with positive airway pressure (PAP) has been shown to improve neurocognitive function in moderate-to-severe OSA while cognitive decline in COPD may be reversible through treatment with long-term oxygen therapy. The investigators will also study whether treatment with PAP and supplemental oxygen vs PAP alone will improve cognitive function and improve quality of life of elderly veterans.
ID: NCT02703207
Sponsor; Investigators: VA Office of Research and Development; Susmita Chowdhuri, MD, MS
Locations: John D. Dingell VA Medical Center, Detroit
The Effect of a Technology-Mediated Integrated Walking and Tai Chi Intervention on Physical Function in Veterans With COPD and Chronic Musculoskeletal Pain (WATCH for Pain)
Persons with COPD benefit from being physically active, but they are often limited by chronic musculoskeletal pain. This project will determine whether a non-pharmacologic, integrated, technology-mediated walking and tai chi mindfulness intervention can improve physical function in veterans with COPD and chronic musculoskeletal pain. The proposed research addresses VA Rehabilitation R&D Service's high priority area of improving health-related quality of life by reducing disease burden and maximizing function in veterans with chronic disease.
ID: NCT05701982
Sponsor; Investigator: VA Office of Research and Development; Marilyn L. Moy, MD; University of Michigan, Beth Israel Deaconess Medical Center
Location: VA Boston Healthcare System
Internet-based Cognitive-behavioral Treatment for Insomnia in COPD Patients Undergoing Pulmonary Rehabilitation
This study is a randomized controlled trial (RCT) to compare sleep and health-related functioning in veterans with COPD and insomnia receiving an Internet-based behavioral treatment for insomnia vs online insomnia patient education. Participants will undergo a sleep and health assessment that will be performed at baseline, post-treatment, and 3 months later. Participants will be randomly assigned to either Internet-based behavioral treatment for insomnia or online insomnia patient education.
ID: NCT04700098
Sponsor; Collaborators: VA Office of Research and Development; Faith S. Luyster, PhD
Locations: VA Pittsburgh Healthcare System; John D. Dingell VA Medical Center, Detroit
Breathe Easier With Tadalafil Therapy for Dyspnea in COPD-PH (BETTER COPD-PH)
The investigators will study whether the drug tadalafil improves shortness of breath in 126 veterans with COPD and high blood pressure in the lungs. The investigators will also assess whether tadalafil improves quality of life, home daily physical activity, exercise endurance, the frequency of acute flares of COPD, blood pressure in the lungs, and lung function. Veterans who enroll in the trial will be allocated by chance to either active tadalafil or an inactive identical capsule (placebo). Neither the veteran nor the investigator will know whether the veteran is taking tadalafil or placebo. Veterans will be followed closely in clinic or by telephone at 1, 2, 3, 4, 5, and 6 months, with attention to side effects and safety. At 1,3, and 6 months the investigators will repeat the questionnaires and testing of blood pressures in the lung and lung function. The investigators anticipate that the results of this study will determine whether tadalafil improves shortness of breath when added to usual medications for COPD.
ID: NCT05937854
Sponsor; Collaborator: VA Office of Research and Development; Sharon I. Rounds, MD
Locations: Rocky Mountain Regional VA Medical Center, Colorado; Joseph Maxwell Cleland Atlanta VA Medical Center ; VA Boston Healthcare System Jamaica Plain Campus; VA Nebraska-Western Iowa Health Care System; Providence VA Medical Center
Impact of Positive Airway Pressure Therapy on Clinical Outcomes in Older Veterans With Chronic Obstructive Pulmonary Disease and Comorbid Obstructive Sleep Apnea (Overlap Syndrome)
Obstructive sleep apnea (OSA) and COPD are highly prevalent chronic respiratory diseases in the veteran population. OSA co-occurring with COPD, known as overlap syndrome (OVS), is a complex chronic medical condition associated with grave consequences. OVS is highly prevalent in veterans. Veterans with OVS may be at increased risk for cognitive deficits, poor sleep quality as well as a reduced quality of life (QoL). The overall objective is to study the effects of positive airway pressure therapy on clinical outcomes in patients with OVS.
ID: NCT04179981
Sponsor; Investigator: VA Office of Research and Development; Susmita Chowdhuri, MD, MS
Locations: VA Ann Arbor Healthcare System; John D. Dingell VA Medical Center, Detroit
Developing an Intervention to Optimize Virtual Care Adoption for COPD Management (VC-OPTIONS)
VA is a leader in virtual care (VC), including the patient portal, mobile apps, and telehealth programs. VC has great utility for managing chronic conditions like COPD. However, adoption of many VC services has been slow. Lack of awareness about these services is one of the most prominent patient- and health care team-facing barriers to adopting VC. This study will develop, refine, and pilot a stakeholder-informed multicomponent implementation strategy to support adoption of VC, referred to as VC-OPTIONS (Virtual Care for Chronic Obstructive Pulmonary Disease Adoption Support). This feasibility trial will pilot the VC-OPTIONS implementation strategy to assess feasibility and acceptability and gather preliminary effectiveness data to inform a larger hybrid effectiveness-implementation trial. The core component of VC-OPTIONS will be the provision of information via VA's Annie texting program to empower patients with knowledge about the array of VC services and how they can be used to support COPD management. It is hypothesized that this strategy will be acceptable and feasible. This work will improve patient and team awareness of and communication about VC services, and support patient access to VC services for COPD management.
ID: NCT05986214
Sponsor; Collaborators: VA Office of Research and Development; Stephanie Robinson, PhD
Location: VA Bedford Healthcare System, Massachusetts; VA Boston Healthcare System Jamaica Plain Campus
Chronic Lung Disease and COVID-19: Understanding Severity, Recovery and Rehabilitation Needs (LAUREL)
This study is comprised of 3 approaches. First, the investigators will conduct a retrospective cohort study to determine factors associated with COVID-19 severity and complications and understand COVID-19 outcomes, including all-cause mortality, postdischarge events, and impacts of rehabilitation services (third aim). The second aim is a mixed-method study and follows COVID-19 patients with repeated surveys to determine patient-reported functional outcomes, health recovery, and rehabilitation needs after COVID-19. The investigators will recruit patients and their informal caregivers for interviews to assess their function and rehabilitation needs.
ID: NCT04628039
Sponsor; Collaborators: VA Office of Research and Development; Kristina A. Crothers, MD
Locations: VA Ann Arbor Healthcare System; VA Puget Sound Health Care System, Washington
Accessing Mobility Using Wearable Sensors
This study will examine whether wearable sensors can be used to track changes in cognitive-motor performance in response to a disease or an intervention. The investigators specific aims are twofold, first aim to explore whether and how a clinical condition such as chronic obstructive pulmonary disease (COPD) or congestive heart failure (CHF) may impact motor-cognitive performance measurable using validated wearable devices (eg, LEGSys, BalanSENS, and Frailty Meter). Second, the investigators will explore whether an exercise intervention provided via telemedicine (telerehabilitation) can enhance motor-cognitive performance.
ID: NCT04306588
Sponsor; Collaborators: Baylor College of Medicine, Bijan Najafi, PhD
Locations: Michael E. DeBakey Veterans Affairs Medical Center, Houston
The clinical trials listed below are open as of February 21, 2025; have ≥ 1 US Department of Veterans Affairs (VA) medical center (VAMC) or US Department of Defense (DoD) military treatment facility location recruiting patients; and are focused on treatments for chronic obstructive pulmonary disease (COPD). For additional information and full inclusion/exclusion criteria, please consult clinicaltrials.gov.
Actively Recruiting
The Effect of Interval Exercise on Functional Outcomes in Veterans With COPD and OSA
The term overlap syndrome (OS) is used to describe the presence of both COPD and obstructive sleep apnea (OSA) in a single patient. Due to premature aging, patients with OS are prone to developing functional decline up to 20 years earlier than the general population. The International Classification of Functioning, Disability and Health (ICF) evaluates functional status in chronic pulmonary disease globally in 5 domains. The investigators propose to study validated outcomes in 3 of these domains: (1) participation in life situations; (2) physical activity; and (3) cardiovascular health. The investigators’ long-term goal is to develop an exercise strategy tailored to veterans with OS which will reduce the risk of functional decline through increased physical activity.
ID: NCT05254431
Sponsor; Collaborator: VA Office of Research and Development; Madalina Macrea, MD, PhD
Location: Salem VA Medical Center, Virginia
The Development of an Integrated Physical Activity and Mental Health Intervention for Veterans With COPD, Emotion Distress, and Low Physical Activity
COPD is a prevalent and debilitating chronic disease in veterans. COPD is highly comorbid with depression and anxiety, conferring greater morbidity and mortality risk. Physical activity is a modifiable behavior that can improve COPD outcomes. However, to date, interventions targeting physical activity have not addressed the high comorbidity between COPD and depression and/or anxiety symptoms (emotional distress) despite emotional distress predicting poorer response to physical activity interventions. This CDA-2 proposal will develop and test the acceptability and feasibility of an integrative physical activity and mental health intervention for veterans with COPD, emotional distress, and low physical activity. The intervention will be delivered via VA Video Connect enabling access to care among veterans with substantial barriers to hospital-based outpatient care.
ID: NCT04953806
Sponsor; Collaborator: VA Office of Research and Development; Patricia Bamonti, PhD
Location: VA Boston Healthcare System, Jamaica Plain Campus
Neurocognitive and Health Impact of Sleep Apnea in Elderly Veterans With Comorbid COPD
Cognitive dysfunction in the aging veteran population is a growing health concern in the Veterans Health System. It is not known whether OSA coexisting with COPD will enhance the risk for cognitive dysfunction. The investigators sought to investigate whether these two highly prevalent diseases that often coexist as 'overlap syndrome' combine to enhance cognitive impairment in the elderly veteran population. Thus, the investigators will study whether elderly patients with overlap syndrome have increased cognitive deficits compared with OSA or COPD alone. Additionally, treatment of OSA with positive airway pressure (PAP) has been shown to improve neurocognitive function in moderate-to-severe OSA while cognitive decline in COPD may be reversible through treatment with long-term oxygen therapy. The investigators will also study whether treatment with PAP and supplemental oxygen vs PAP alone will improve cognitive function and improve quality of life of elderly veterans.
ID: NCT02703207
Sponsor; Investigators: VA Office of Research and Development; Susmita Chowdhuri, MD, MS
Locations: John D. Dingell VA Medical Center, Detroit
The Effect of a Technology-Mediated Integrated Walking and Tai Chi Intervention on Physical Function in Veterans With COPD and Chronic Musculoskeletal Pain (WATCH for Pain)
Persons with COPD benefit from being physically active, but they are often limited by chronic musculoskeletal pain. This project will determine whether a non-pharmacologic, integrated, technology-mediated walking and tai chi mindfulness intervention can improve physical function in veterans with COPD and chronic musculoskeletal pain. The proposed research addresses VA Rehabilitation R&D Service's high priority area of improving health-related quality of life by reducing disease burden and maximizing function in veterans with chronic disease.
ID: NCT05701982
Sponsor; Investigator: VA Office of Research and Development; Marilyn L. Moy, MD; University of Michigan, Beth Israel Deaconess Medical Center
Location: VA Boston Healthcare System
Internet-based Cognitive-behavioral Treatment for Insomnia in COPD Patients Undergoing Pulmonary Rehabilitation
This study is a randomized controlled trial (RCT) to compare sleep and health-related functioning in veterans with COPD and insomnia receiving an Internet-based behavioral treatment for insomnia vs online insomnia patient education. Participants will undergo a sleep and health assessment that will be performed at baseline, post-treatment, and 3 months later. Participants will be randomly assigned to either Internet-based behavioral treatment for insomnia or online insomnia patient education.
ID: NCT04700098
Sponsor; Collaborators: VA Office of Research and Development; Faith S. Luyster, PhD
Locations: VA Pittsburgh Healthcare System; John D. Dingell VA Medical Center, Detroit
Breathe Easier With Tadalafil Therapy for Dyspnea in COPD-PH (BETTER COPD-PH)
The investigators will study whether the drug tadalafil improves shortness of breath in 126 veterans with COPD and high blood pressure in the lungs. The investigators will also assess whether tadalafil improves quality of life, home daily physical activity, exercise endurance, the frequency of acute flares of COPD, blood pressure in the lungs, and lung function. Veterans who enroll in the trial will be allocated by chance to either active tadalafil or an inactive identical capsule (placebo). Neither the veteran nor the investigator will know whether the veteran is taking tadalafil or placebo. Veterans will be followed closely in clinic or by telephone at 1, 2, 3, 4, 5, and 6 months, with attention to side effects and safety. At 1,3, and 6 months the investigators will repeat the questionnaires and testing of blood pressures in the lung and lung function. The investigators anticipate that the results of this study will determine whether tadalafil improves shortness of breath when added to usual medications for COPD.
ID: NCT05937854
Sponsor; Collaborator: VA Office of Research and Development; Sharon I. Rounds, MD
Locations: Rocky Mountain Regional VA Medical Center, Colorado; Joseph Maxwell Cleland Atlanta VA Medical Center ; VA Boston Healthcare System Jamaica Plain Campus; VA Nebraska-Western Iowa Health Care System; Providence VA Medical Center
Impact of Positive Airway Pressure Therapy on Clinical Outcomes in Older Veterans With Chronic Obstructive Pulmonary Disease and Comorbid Obstructive Sleep Apnea (Overlap Syndrome)
Obstructive sleep apnea (OSA) and COPD are highly prevalent chronic respiratory diseases in the veteran population. OSA co-occurring with COPD, known as overlap syndrome (OVS), is a complex chronic medical condition associated with grave consequences. OVS is highly prevalent in veterans. Veterans with OVS may be at increased risk for cognitive deficits, poor sleep quality as well as a reduced quality of life (QoL). The overall objective is to study the effects of positive airway pressure therapy on clinical outcomes in patients with OVS.
ID: NCT04179981
Sponsor; Investigator: VA Office of Research and Development; Susmita Chowdhuri, MD, MS
Locations: VA Ann Arbor Healthcare System; John D. Dingell VA Medical Center, Detroit
Developing an Intervention to Optimize Virtual Care Adoption for COPD Management (VC-OPTIONS)
VA is a leader in virtual care (VC), including the patient portal, mobile apps, and telehealth programs. VC has great utility for managing chronic conditions like COPD. However, adoption of many VC services has been slow. Lack of awareness about these services is one of the most prominent patient- and health care team-facing barriers to adopting VC. This study will develop, refine, and pilot a stakeholder-informed multicomponent implementation strategy to support adoption of VC, referred to as VC-OPTIONS (Virtual Care for Chronic Obstructive Pulmonary Disease Adoption Support). This feasibility trial will pilot the VC-OPTIONS implementation strategy to assess feasibility and acceptability and gather preliminary effectiveness data to inform a larger hybrid effectiveness-implementation trial. The core component of VC-OPTIONS will be the provision of information via VA's Annie texting program to empower patients with knowledge about the array of VC services and how they can be used to support COPD management. It is hypothesized that this strategy will be acceptable and feasible. This work will improve patient and team awareness of and communication about VC services, and support patient access to VC services for COPD management.
ID: NCT05986214
Sponsor; Collaborators: VA Office of Research and Development; Stephanie Robinson, PhD
Location: VA Bedford Healthcare System, Massachusetts; VA Boston Healthcare System Jamaica Plain Campus
Chronic Lung Disease and COVID-19: Understanding Severity, Recovery and Rehabilitation Needs (LAUREL)
This study is comprised of 3 approaches. First, the investigators will conduct a retrospective cohort study to determine factors associated with COVID-19 severity and complications and understand COVID-19 outcomes, including all-cause mortality, postdischarge events, and impacts of rehabilitation services (third aim). The second aim is a mixed-method study and follows COVID-19 patients with repeated surveys to determine patient-reported functional outcomes, health recovery, and rehabilitation needs after COVID-19. The investigators will recruit patients and their informal caregivers for interviews to assess their function and rehabilitation needs.
ID: NCT04628039
Sponsor; Collaborators: VA Office of Research and Development; Kristina A. Crothers, MD
Locations: VA Ann Arbor Healthcare System; VA Puget Sound Health Care System, Washington
Accessing Mobility Using Wearable Sensors
This study will examine whether wearable sensors can be used to track changes in cognitive-motor performance in response to a disease or an intervention. The investigators specific aims are twofold, first aim to explore whether and how a clinical condition such as chronic obstructive pulmonary disease (COPD) or congestive heart failure (CHF) may impact motor-cognitive performance measurable using validated wearable devices (eg, LEGSys, BalanSENS, and Frailty Meter). Second, the investigators will explore whether an exercise intervention provided via telemedicine (telerehabilitation) can enhance motor-cognitive performance.
ID: NCT04306588
Sponsor; Collaborators: Baylor College of Medicine, Bijan Najafi, PhD
Locations: Michael E. DeBakey Veterans Affairs Medical Center, Houston
Patients With Asthma and COPD At Increased Cancer Risk From Microplastics
Individuals with asthma and chronic obstructive pulmonary disease (COPD) were more vulnerable than healthy controls to epithelial cell changes caused by microplastics exposure, based on data from a new simulation study.
Microplastic fibers present in the ambient air can be inhaled into the lungs and promote a range of complications including oxidative stress, local injury, and cytotoxicity, but data on the effects of microplastic fibers on individuals with obstructive lung diseases are limited, wrote Magdalena Poplinska-Goryca, MD, of the Medical University of Warsaw, Warsaw, Poland, and colleagues.
In a study published in Scientific Reports, the researchers identified 10 adults aged ≥ 18 years with asthma, eight adults aged ≥ 40 years with COPD, and 11 healthy adult controls. Individuals with more serious conditions such as severe asthma or COPD, unstable or uncontrolled disease, concomitant malignancies, or chronic or acute lung disease were excluded.
The researchers obtained nasal epithelial cells from all participants, and exposed these cells to microplastic fibers created by the researchers in a laboratory setting. Overall, asthmatic and COPD airway epithelial cells showed a different reaction to microplastic fibers stimulation compared to healthy epithelial cells. The most significant response was associated with Th2 inflammation, modulation of stress response, and carcinogenesis. No differences in cytotoxic or minor inflammatory effects on epithelial cells of patients with asthma or COPD were noted compared with healthy controls.
In addition, flow cytometric analysis showed increased CD24+ epithelial cells in asthma patients compared to controls after microplastics exposure.
“Many of the gene candidates selected from RNA-Seq analysis are related to cancer (upregulated in many cancer types according to the literature), and the activation of CD24 on primarily ciliated asthmatic epithelial cells after microplastic stimulation further supports this theory,” the researchers wrote.
The findings were limited by several factors including the use of nasal rather than bronchial epithelial cells, which would have yielded more information, the researchers noted. Also, patients with severe asthma and COPD were excluded, they said, because of the impact of oral steroid and antibiotic use by this patient group on epithelial cell immunology that could bias the results of epithelial response to microplastic fiber exposure.
However, the results suggest that “the structural impairment of the airway epithelium in obstructive diseases enhances the impact of microplastic particles compared to healthy epithelium,” the researchers concluded.
Current and Future Implications
The current study is important in addressing the increasing environmental presence of microplastics and their potential impact on respiratory health, said Seyedmohammad Pourshahid, MD, assistant professor of thoracic medicine and surgery at the Lewis Katz School of Medicine at Temple University, Philadelphia, in an interview.
“By examining how microplastics interact with airway epithelial cells, particularly in individuals with asthma and COPD, the research aims to elucidate mechanisms that could contribute to disease progression or exacerbation,” he said.
“The study’s findings that microplastics did not induce a strong inflammatory response, unlike other pollutants such as PM2.5, were unexpected; instead, microplastics appeared to influence pathways related to airway remodeling and oxidative stress,” Pourshahid noted. “This suggests that microplastics may affect respiratory health through mechanisms distinct from traditional pollutants,” he said.
“While preliminary, this research highlights the potential role of environmental microplastic exposure in respiratory diseases,” Pourshahid told this news organization. “Clinicians should be aware of emerging environmental factors that could impact patient health, especially in individuals with asthma and COPD. This awareness may inform patient education and advocacy for reducing exposure to airborne microplastics,” he said.
More studies are needed to explore the long-term effects of microplastic exposure on respiratory health, particularly in vulnerable populations, said Pourshahid. Research with in vivo models is necessary to confirm the findings and assess potential clinical implications to confirm these findings and assess potential clinical implications, he said. “Understanding the prevalence and sources of daily microplastic exposure can inform public health strategies to mitigate risks,” he added.
The study was supported by the Jakub Potocki Foundation. Paplińska-Goryca and Pourshahid had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Individuals with asthma and chronic obstructive pulmonary disease (COPD) were more vulnerable than healthy controls to epithelial cell changes caused by microplastics exposure, based on data from a new simulation study.
Microplastic fibers present in the ambient air can be inhaled into the lungs and promote a range of complications including oxidative stress, local injury, and cytotoxicity, but data on the effects of microplastic fibers on individuals with obstructive lung diseases are limited, wrote Magdalena Poplinska-Goryca, MD, of the Medical University of Warsaw, Warsaw, Poland, and colleagues.
In a study published in Scientific Reports, the researchers identified 10 adults aged ≥ 18 years with asthma, eight adults aged ≥ 40 years with COPD, and 11 healthy adult controls. Individuals with more serious conditions such as severe asthma or COPD, unstable or uncontrolled disease, concomitant malignancies, or chronic or acute lung disease were excluded.
The researchers obtained nasal epithelial cells from all participants, and exposed these cells to microplastic fibers created by the researchers in a laboratory setting. Overall, asthmatic and COPD airway epithelial cells showed a different reaction to microplastic fibers stimulation compared to healthy epithelial cells. The most significant response was associated with Th2 inflammation, modulation of stress response, and carcinogenesis. No differences in cytotoxic or minor inflammatory effects on epithelial cells of patients with asthma or COPD were noted compared with healthy controls.
In addition, flow cytometric analysis showed increased CD24+ epithelial cells in asthma patients compared to controls after microplastics exposure.
“Many of the gene candidates selected from RNA-Seq analysis are related to cancer (upregulated in many cancer types according to the literature), and the activation of CD24 on primarily ciliated asthmatic epithelial cells after microplastic stimulation further supports this theory,” the researchers wrote.
The findings were limited by several factors including the use of nasal rather than bronchial epithelial cells, which would have yielded more information, the researchers noted. Also, patients with severe asthma and COPD were excluded, they said, because of the impact of oral steroid and antibiotic use by this patient group on epithelial cell immunology that could bias the results of epithelial response to microplastic fiber exposure.
However, the results suggest that “the structural impairment of the airway epithelium in obstructive diseases enhances the impact of microplastic particles compared to healthy epithelium,” the researchers concluded.
Current and Future Implications
The current study is important in addressing the increasing environmental presence of microplastics and their potential impact on respiratory health, said Seyedmohammad Pourshahid, MD, assistant professor of thoracic medicine and surgery at the Lewis Katz School of Medicine at Temple University, Philadelphia, in an interview.
“By examining how microplastics interact with airway epithelial cells, particularly in individuals with asthma and COPD, the research aims to elucidate mechanisms that could contribute to disease progression or exacerbation,” he said.
“The study’s findings that microplastics did not induce a strong inflammatory response, unlike other pollutants such as PM2.5, were unexpected; instead, microplastics appeared to influence pathways related to airway remodeling and oxidative stress,” Pourshahid noted. “This suggests that microplastics may affect respiratory health through mechanisms distinct from traditional pollutants,” he said.
“While preliminary, this research highlights the potential role of environmental microplastic exposure in respiratory diseases,” Pourshahid told this news organization. “Clinicians should be aware of emerging environmental factors that could impact patient health, especially in individuals with asthma and COPD. This awareness may inform patient education and advocacy for reducing exposure to airborne microplastics,” he said.
More studies are needed to explore the long-term effects of microplastic exposure on respiratory health, particularly in vulnerable populations, said Pourshahid. Research with in vivo models is necessary to confirm the findings and assess potential clinical implications to confirm these findings and assess potential clinical implications, he said. “Understanding the prevalence and sources of daily microplastic exposure can inform public health strategies to mitigate risks,” he added.
The study was supported by the Jakub Potocki Foundation. Paplińska-Goryca and Pourshahid had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Individuals with asthma and chronic obstructive pulmonary disease (COPD) were more vulnerable than healthy controls to epithelial cell changes caused by microplastics exposure, based on data from a new simulation study.
Microplastic fibers present in the ambient air can be inhaled into the lungs and promote a range of complications including oxidative stress, local injury, and cytotoxicity, but data on the effects of microplastic fibers on individuals with obstructive lung diseases are limited, wrote Magdalena Poplinska-Goryca, MD, of the Medical University of Warsaw, Warsaw, Poland, and colleagues.
In a study published in Scientific Reports, the researchers identified 10 adults aged ≥ 18 years with asthma, eight adults aged ≥ 40 years with COPD, and 11 healthy adult controls. Individuals with more serious conditions such as severe asthma or COPD, unstable or uncontrolled disease, concomitant malignancies, or chronic or acute lung disease were excluded.
The researchers obtained nasal epithelial cells from all participants, and exposed these cells to microplastic fibers created by the researchers in a laboratory setting. Overall, asthmatic and COPD airway epithelial cells showed a different reaction to microplastic fibers stimulation compared to healthy epithelial cells. The most significant response was associated with Th2 inflammation, modulation of stress response, and carcinogenesis. No differences in cytotoxic or minor inflammatory effects on epithelial cells of patients with asthma or COPD were noted compared with healthy controls.
In addition, flow cytometric analysis showed increased CD24+ epithelial cells in asthma patients compared to controls after microplastics exposure.
“Many of the gene candidates selected from RNA-Seq analysis are related to cancer (upregulated in many cancer types according to the literature), and the activation of CD24 on primarily ciliated asthmatic epithelial cells after microplastic stimulation further supports this theory,” the researchers wrote.
The findings were limited by several factors including the use of nasal rather than bronchial epithelial cells, which would have yielded more information, the researchers noted. Also, patients with severe asthma and COPD were excluded, they said, because of the impact of oral steroid and antibiotic use by this patient group on epithelial cell immunology that could bias the results of epithelial response to microplastic fiber exposure.
However, the results suggest that “the structural impairment of the airway epithelium in obstructive diseases enhances the impact of microplastic particles compared to healthy epithelium,” the researchers concluded.
Current and Future Implications
The current study is important in addressing the increasing environmental presence of microplastics and their potential impact on respiratory health, said Seyedmohammad Pourshahid, MD, assistant professor of thoracic medicine and surgery at the Lewis Katz School of Medicine at Temple University, Philadelphia, in an interview.
“By examining how microplastics interact with airway epithelial cells, particularly in individuals with asthma and COPD, the research aims to elucidate mechanisms that could contribute to disease progression or exacerbation,” he said.
“The study’s findings that microplastics did not induce a strong inflammatory response, unlike other pollutants such as PM2.5, were unexpected; instead, microplastics appeared to influence pathways related to airway remodeling and oxidative stress,” Pourshahid noted. “This suggests that microplastics may affect respiratory health through mechanisms distinct from traditional pollutants,” he said.
“While preliminary, this research highlights the potential role of environmental microplastic exposure in respiratory diseases,” Pourshahid told this news organization. “Clinicians should be aware of emerging environmental factors that could impact patient health, especially in individuals with asthma and COPD. This awareness may inform patient education and advocacy for reducing exposure to airborne microplastics,” he said.
More studies are needed to explore the long-term effects of microplastic exposure on respiratory health, particularly in vulnerable populations, said Pourshahid. Research with in vivo models is necessary to confirm the findings and assess potential clinical implications to confirm these findings and assess potential clinical implications, he said. “Understanding the prevalence and sources of daily microplastic exposure can inform public health strategies to mitigate risks,” he added.
The study was supported by the Jakub Potocki Foundation. Paplińska-Goryca and Pourshahid had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Trump Administration Review of Psychiatric Meds Raises Concerns
The Trump administration’s plans to study the “threat” posed by psychiatric medications in children have medical societies and mental health professionals concerned that the administration may be considering restrictions on the use of psychotropic drugs in pediatric patients.
An executive order signed last week created the “Make American Healthy Again Commission” to investigate the nation’s “escalating health crisis,” particularly in child health. Recently confirmed Secretary of the US Department of Health and Human Services Robert F. Kennedy Jr. will chair the effort.
As part of its investigation, the executive order directed the commission to assess “the prevalence of and threat posed by the prescription of selective serotonin reuptake inhibitors (SSRIs), antipsychotics, mood stabilizers, stimulants, and weight-loss drugs.”
A report on the commission’s findings is due in a little less than 100 days. Eighty days later, the commission must submit recommendations for federal action.
Although who the commission members are and the scope of its work is unclear, the language in the executive order — namely the implication that the Trump administration views psychotropic medication as a “threat” to children — was enough to prompt psychiatrists from across the country to contact the American Psychiatric Association (APA) about possible limitations on the use of psychotropic medications in pediatric patients.
“It’s concerning and surprising that some of our nation’s most vulnerable children who need these treatments to participate fully in life would be under scrutiny in this way,” Marketa Wills, MD, MBA, chief executive officer and medical director for the APA, told this news organization.
“If these medications are under threat and children decompensate that would not be good from a public health perspective, for the healthcare system or for the families we serve,” Wills said.
Past Comments Fuel Distress
Past comments by the commission chair have only fueled distress over the commission’s goals. Kennedy has long expressed skepticism about antidepressants, especially (SSRIs), questioning their safety and suggesting they are as addictive as heroin.
“I know people, including members of my family, who’ve had a much worse time getting off of SSRIs than they have getting off of heroin,” Kennedy said during his Senate confirmation hearing in late January.
But there is no evidence to suggest SSRIs or other antidepressants are addictive, Leslie A. Hulvershorn, MD, chair and associate professor of psychiatry at Indiana University School of Medicine, told this news organization.
“They don’t work in the systems of the brain that drive addiction. A large amount of research suggests that they are safe to take for a long time,” she said. “I suspect the confusion comes from the difference between it not being wise to come off of the medication, because of a concern for relapse of a psychiatric illness, and some transient discomfort from abruptly stopping SSRIs without tapering them off versus being addicted to it, like heroin.”
During the hearing, Kennedy was also asked to respond to comments he made during a 2023 livestream on X in which he claimed that the use of antidepressants have contributed to the increase in school shootings in the United States.
“I am also going to look very closely at the role of psychiatric drugs in these events and there are no good studies right now that should have been done years ago on this issue because there is a tremendous circumstantial evidence that SSRIs and benzos and other drugs are doing this,” he said in the livestream.
Research has shown that there is no link between school shootings and antidepressant use.
In a 2024 interview on the Latino Capitalist podcast, Kennedy said that he wanted create “wellness farms” for adults addicted to illicit drugs and children who take antidepressants or stimulants for ADHD could be “reparented.”
“The views on those wellness farms are concerning for us here at the American Psychiatric Association. It remains to be seen if he brings that back up in his new role at HHS. There is currently no evidence of their efficacy,” Wills said.
Fear Is a ‘Real Concern’
These controversial comments, combined with the commission’s charge to investigate the potential “threat” psychotropic medications pose to children, worry clinicians and families fear that access to medication could be restricted.
“Psychiatrists and patients are very concerned about the risk these statements may pose,” Hulvershorn said.
“Certainly, there is evidence that psychotropic medications are overprescribed, particularly in children who are in state care — like wards of the state — and who are part of Medicaid programs, but there is tremendous overall benefit associated with psychotropic medications in youth and adults. They are lifesaving and game changing in many instances,” she added.
Psychiatrists who’ve contacted the APA since last week’s announcement echo Hulvershorn’s comments.
“The fear is the real concern,” Wills said. “No parent takes the decision lightly to put their child on medication. With all interventions, particularly with children, there are risks and benefits that must be carefully weighed. The best person to weigh those risks and benefits is the child and adolescent psychiatrist, in conjunction with the child’s parents.”
The focus on medication also overlooks the fact that psychosocial interventions — not medication — are first-line treatment for children with mental health issues and that guidelines recommend medication be used alongside nonpharmacological therapy.
“Extensive research, including large national multi-site studies, have examined the most effective ways to reduce psychological symptoms among youth, including anxiety, depression, and ADHD. Results consistently reveal that both psychotropic medications and psychological interventions can offer significant improvements, often in combination,” Mitch Prinstein, PhD, chief of psychology strategy and integration at the American Psychological Association, told this news organization.
“Given the substantial challenges for many in gaining access to psychotherapy and a national shortage of licensed psychologists, reducing access to medications would undoubtedly have a debilitating effect of the already concerning youth mental health crisis,” Prinstein said.
A Seat at The Table
While the launch of the commission has left some feeling uneasy, experts agree that a national focus on children’s mental health is needed.
The APA would “welcome an opportunity to be part of this national conversation following the evidence base, following settled science that shows when and how these medications are effective and helpful for children and families,” said Wills. “We also think it’s very important that child and adolescent psychiatrists be at the table for this national conversation on behalf of the families they serve.”
In a joint letter with the APA, officials with the American Academy of Child and Adolescent Psychiatry also expressed interest in playing a role in the commission’s work.
“We are in the middle of a mental health crisis, with a record number of Americans struggling with mental health and substance use disorders. We strongly urge you to prioritize strengthening the ability to respond to an increasing demand for psychiatric services, especially for children,” the letter stated.
Indeed, looking beyond just the use of psychotropic medications is vital to the success of any strategy to address the youth mental health crisis, Hulvershorn noted.
“There are already many programs underway to examine the overprescribing. In my view, the lack of supports by payors for behavioral interventions, such as evidence-based family interventions, psychotherapies, etc., is the major driver for overuse of medications,” she said.
“Every pediatrician and child psychiatrist I know would rather try a behavioral intervention with a family first, but those are services that our systems do not financially support well and are, thus, underdeveloped, and very difficult to access,” Hulvershorn added.
More funding for evidence-based interventions — both behavioral and pharmacological — is desperately needed, she said. Support for workforce development should also be a part of any proposed solution.
“Adequate and responsible funding in all of those areas is needed, but we have some low hanging fruit in terms of figuring out how to just deliver the interventions that science has shown us do work,” Hulvershorn said. “Many of those interventions don’t involve medication and I think every expert in the field would be glad to see more effort put into system reform to better deliver interventions that work to youth and their families.”
A version of this article first appeared on Medscape.com.
The Trump administration’s plans to study the “threat” posed by psychiatric medications in children have medical societies and mental health professionals concerned that the administration may be considering restrictions on the use of psychotropic drugs in pediatric patients.
An executive order signed last week created the “Make American Healthy Again Commission” to investigate the nation’s “escalating health crisis,” particularly in child health. Recently confirmed Secretary of the US Department of Health and Human Services Robert F. Kennedy Jr. will chair the effort.
As part of its investigation, the executive order directed the commission to assess “the prevalence of and threat posed by the prescription of selective serotonin reuptake inhibitors (SSRIs), antipsychotics, mood stabilizers, stimulants, and weight-loss drugs.”
A report on the commission’s findings is due in a little less than 100 days. Eighty days later, the commission must submit recommendations for federal action.
Although who the commission members are and the scope of its work is unclear, the language in the executive order — namely the implication that the Trump administration views psychotropic medication as a “threat” to children — was enough to prompt psychiatrists from across the country to contact the American Psychiatric Association (APA) about possible limitations on the use of psychotropic medications in pediatric patients.
“It’s concerning and surprising that some of our nation’s most vulnerable children who need these treatments to participate fully in life would be under scrutiny in this way,” Marketa Wills, MD, MBA, chief executive officer and medical director for the APA, told this news organization.
“If these medications are under threat and children decompensate that would not be good from a public health perspective, for the healthcare system or for the families we serve,” Wills said.
Past Comments Fuel Distress
Past comments by the commission chair have only fueled distress over the commission’s goals. Kennedy has long expressed skepticism about antidepressants, especially (SSRIs), questioning their safety and suggesting they are as addictive as heroin.
“I know people, including members of my family, who’ve had a much worse time getting off of SSRIs than they have getting off of heroin,” Kennedy said during his Senate confirmation hearing in late January.
But there is no evidence to suggest SSRIs or other antidepressants are addictive, Leslie A. Hulvershorn, MD, chair and associate professor of psychiatry at Indiana University School of Medicine, told this news organization.
“They don’t work in the systems of the brain that drive addiction. A large amount of research suggests that they are safe to take for a long time,” she said. “I suspect the confusion comes from the difference between it not being wise to come off of the medication, because of a concern for relapse of a psychiatric illness, and some transient discomfort from abruptly stopping SSRIs without tapering them off versus being addicted to it, like heroin.”
During the hearing, Kennedy was also asked to respond to comments he made during a 2023 livestream on X in which he claimed that the use of antidepressants have contributed to the increase in school shootings in the United States.
“I am also going to look very closely at the role of psychiatric drugs in these events and there are no good studies right now that should have been done years ago on this issue because there is a tremendous circumstantial evidence that SSRIs and benzos and other drugs are doing this,” he said in the livestream.
Research has shown that there is no link between school shootings and antidepressant use.
In a 2024 interview on the Latino Capitalist podcast, Kennedy said that he wanted create “wellness farms” for adults addicted to illicit drugs and children who take antidepressants or stimulants for ADHD could be “reparented.”
“The views on those wellness farms are concerning for us here at the American Psychiatric Association. It remains to be seen if he brings that back up in his new role at HHS. There is currently no evidence of their efficacy,” Wills said.
Fear Is a ‘Real Concern’
These controversial comments, combined with the commission’s charge to investigate the potential “threat” psychotropic medications pose to children, worry clinicians and families fear that access to medication could be restricted.
“Psychiatrists and patients are very concerned about the risk these statements may pose,” Hulvershorn said.
“Certainly, there is evidence that psychotropic medications are overprescribed, particularly in children who are in state care — like wards of the state — and who are part of Medicaid programs, but there is tremendous overall benefit associated with psychotropic medications in youth and adults. They are lifesaving and game changing in many instances,” she added.
Psychiatrists who’ve contacted the APA since last week’s announcement echo Hulvershorn’s comments.
“The fear is the real concern,” Wills said. “No parent takes the decision lightly to put their child on medication. With all interventions, particularly with children, there are risks and benefits that must be carefully weighed. The best person to weigh those risks and benefits is the child and adolescent psychiatrist, in conjunction with the child’s parents.”
The focus on medication also overlooks the fact that psychosocial interventions — not medication — are first-line treatment for children with mental health issues and that guidelines recommend medication be used alongside nonpharmacological therapy.
“Extensive research, including large national multi-site studies, have examined the most effective ways to reduce psychological symptoms among youth, including anxiety, depression, and ADHD. Results consistently reveal that both psychotropic medications and psychological interventions can offer significant improvements, often in combination,” Mitch Prinstein, PhD, chief of psychology strategy and integration at the American Psychological Association, told this news organization.
“Given the substantial challenges for many in gaining access to psychotherapy and a national shortage of licensed psychologists, reducing access to medications would undoubtedly have a debilitating effect of the already concerning youth mental health crisis,” Prinstein said.
A Seat at The Table
While the launch of the commission has left some feeling uneasy, experts agree that a national focus on children’s mental health is needed.
The APA would “welcome an opportunity to be part of this national conversation following the evidence base, following settled science that shows when and how these medications are effective and helpful for children and families,” said Wills. “We also think it’s very important that child and adolescent psychiatrists be at the table for this national conversation on behalf of the families they serve.”
In a joint letter with the APA, officials with the American Academy of Child and Adolescent Psychiatry also expressed interest in playing a role in the commission’s work.
“We are in the middle of a mental health crisis, with a record number of Americans struggling with mental health and substance use disorders. We strongly urge you to prioritize strengthening the ability to respond to an increasing demand for psychiatric services, especially for children,” the letter stated.
Indeed, looking beyond just the use of psychotropic medications is vital to the success of any strategy to address the youth mental health crisis, Hulvershorn noted.
“There are already many programs underway to examine the overprescribing. In my view, the lack of supports by payors for behavioral interventions, such as evidence-based family interventions, psychotherapies, etc., is the major driver for overuse of medications,” she said.
“Every pediatrician and child psychiatrist I know would rather try a behavioral intervention with a family first, but those are services that our systems do not financially support well and are, thus, underdeveloped, and very difficult to access,” Hulvershorn added.
More funding for evidence-based interventions — both behavioral and pharmacological — is desperately needed, she said. Support for workforce development should also be a part of any proposed solution.
“Adequate and responsible funding in all of those areas is needed, but we have some low hanging fruit in terms of figuring out how to just deliver the interventions that science has shown us do work,” Hulvershorn said. “Many of those interventions don’t involve medication and I think every expert in the field would be glad to see more effort put into system reform to better deliver interventions that work to youth and their families.”
A version of this article first appeared on Medscape.com.
The Trump administration’s plans to study the “threat” posed by psychiatric medications in children have medical societies and mental health professionals concerned that the administration may be considering restrictions on the use of psychotropic drugs in pediatric patients.
An executive order signed last week created the “Make American Healthy Again Commission” to investigate the nation’s “escalating health crisis,” particularly in child health. Recently confirmed Secretary of the US Department of Health and Human Services Robert F. Kennedy Jr. will chair the effort.
As part of its investigation, the executive order directed the commission to assess “the prevalence of and threat posed by the prescription of selective serotonin reuptake inhibitors (SSRIs), antipsychotics, mood stabilizers, stimulants, and weight-loss drugs.”
A report on the commission’s findings is due in a little less than 100 days. Eighty days later, the commission must submit recommendations for federal action.
Although who the commission members are and the scope of its work is unclear, the language in the executive order — namely the implication that the Trump administration views psychotropic medication as a “threat” to children — was enough to prompt psychiatrists from across the country to contact the American Psychiatric Association (APA) about possible limitations on the use of psychotropic medications in pediatric patients.
“It’s concerning and surprising that some of our nation’s most vulnerable children who need these treatments to participate fully in life would be under scrutiny in this way,” Marketa Wills, MD, MBA, chief executive officer and medical director for the APA, told this news organization.
“If these medications are under threat and children decompensate that would not be good from a public health perspective, for the healthcare system or for the families we serve,” Wills said.
Past Comments Fuel Distress
Past comments by the commission chair have only fueled distress over the commission’s goals. Kennedy has long expressed skepticism about antidepressants, especially (SSRIs), questioning their safety and suggesting they are as addictive as heroin.
“I know people, including members of my family, who’ve had a much worse time getting off of SSRIs than they have getting off of heroin,” Kennedy said during his Senate confirmation hearing in late January.
But there is no evidence to suggest SSRIs or other antidepressants are addictive, Leslie A. Hulvershorn, MD, chair and associate professor of psychiatry at Indiana University School of Medicine, told this news organization.
“They don’t work in the systems of the brain that drive addiction. A large amount of research suggests that they are safe to take for a long time,” she said. “I suspect the confusion comes from the difference between it not being wise to come off of the medication, because of a concern for relapse of a psychiatric illness, and some transient discomfort from abruptly stopping SSRIs without tapering them off versus being addicted to it, like heroin.”
During the hearing, Kennedy was also asked to respond to comments he made during a 2023 livestream on X in which he claimed that the use of antidepressants have contributed to the increase in school shootings in the United States.
“I am also going to look very closely at the role of psychiatric drugs in these events and there are no good studies right now that should have been done years ago on this issue because there is a tremendous circumstantial evidence that SSRIs and benzos and other drugs are doing this,” he said in the livestream.
Research has shown that there is no link between school shootings and antidepressant use.
In a 2024 interview on the Latino Capitalist podcast, Kennedy said that he wanted create “wellness farms” for adults addicted to illicit drugs and children who take antidepressants or stimulants for ADHD could be “reparented.”
“The views on those wellness farms are concerning for us here at the American Psychiatric Association. It remains to be seen if he brings that back up in his new role at HHS. There is currently no evidence of their efficacy,” Wills said.
Fear Is a ‘Real Concern’
These controversial comments, combined with the commission’s charge to investigate the potential “threat” psychotropic medications pose to children, worry clinicians and families fear that access to medication could be restricted.
“Psychiatrists and patients are very concerned about the risk these statements may pose,” Hulvershorn said.
“Certainly, there is evidence that psychotropic medications are overprescribed, particularly in children who are in state care — like wards of the state — and who are part of Medicaid programs, but there is tremendous overall benefit associated with psychotropic medications in youth and adults. They are lifesaving and game changing in many instances,” she added.
Psychiatrists who’ve contacted the APA since last week’s announcement echo Hulvershorn’s comments.
“The fear is the real concern,” Wills said. “No parent takes the decision lightly to put their child on medication. With all interventions, particularly with children, there are risks and benefits that must be carefully weighed. The best person to weigh those risks and benefits is the child and adolescent psychiatrist, in conjunction with the child’s parents.”
The focus on medication also overlooks the fact that psychosocial interventions — not medication — are first-line treatment for children with mental health issues and that guidelines recommend medication be used alongside nonpharmacological therapy.
“Extensive research, including large national multi-site studies, have examined the most effective ways to reduce psychological symptoms among youth, including anxiety, depression, and ADHD. Results consistently reveal that both psychotropic medications and psychological interventions can offer significant improvements, often in combination,” Mitch Prinstein, PhD, chief of psychology strategy and integration at the American Psychological Association, told this news organization.
“Given the substantial challenges for many in gaining access to psychotherapy and a national shortage of licensed psychologists, reducing access to medications would undoubtedly have a debilitating effect of the already concerning youth mental health crisis,” Prinstein said.
A Seat at The Table
While the launch of the commission has left some feeling uneasy, experts agree that a national focus on children’s mental health is needed.
The APA would “welcome an opportunity to be part of this national conversation following the evidence base, following settled science that shows when and how these medications are effective and helpful for children and families,” said Wills. “We also think it’s very important that child and adolescent psychiatrists be at the table for this national conversation on behalf of the families they serve.”
In a joint letter with the APA, officials with the American Academy of Child and Adolescent Psychiatry also expressed interest in playing a role in the commission’s work.
“We are in the middle of a mental health crisis, with a record number of Americans struggling with mental health and substance use disorders. We strongly urge you to prioritize strengthening the ability to respond to an increasing demand for psychiatric services, especially for children,” the letter stated.
Indeed, looking beyond just the use of psychotropic medications is vital to the success of any strategy to address the youth mental health crisis, Hulvershorn noted.
“There are already many programs underway to examine the overprescribing. In my view, the lack of supports by payors for behavioral interventions, such as evidence-based family interventions, psychotherapies, etc., is the major driver for overuse of medications,” she said.
“Every pediatrician and child psychiatrist I know would rather try a behavioral intervention with a family first, but those are services that our systems do not financially support well and are, thus, underdeveloped, and very difficult to access,” Hulvershorn added.
More funding for evidence-based interventions — both behavioral and pharmacological — is desperately needed, she said. Support for workforce development should also be a part of any proposed solution.
“Adequate and responsible funding in all of those areas is needed, but we have some low hanging fruit in terms of figuring out how to just deliver the interventions that science has shown us do work,” Hulvershorn said. “Many of those interventions don’t involve medication and I think every expert in the field would be glad to see more effort put into system reform to better deliver interventions that work to youth and their families.”
A version of this article first appeared on Medscape.com.
Diet Changes Show Promise in Early Prostate Cancer
A diet high in omega-3 and low in omega-6 fatty acids, alongside fish oil supplements, may curb the growth of prostate cancer cells in men with early-stage disease, new data showed.
Among men on active surveillance for prostate cancer, consuming this diet for a year led to a significant decrease in the prostate cancer tissue Ki-67 index, a biomarker for prostate cancer progression, metastasis, and death, according to findings from the phase 2 CAPFISH-3 study presented at the 2025 American Society of Clinical Oncology Genitourinary Cancers Symposium.
“This data is certainly intriguing and supports studies looking at this further in prostate cancer,” Bradley Alexander McGregor, MD, from Dana-Farber Cancer Institute, Boston, Massachusetts, who wasn’t involved in the study, told this news organization. But, McGregor noted, patients were on the diet for 1 year, and the long-term implications of this diet are not known.
Growing Evidence on Diet
Diets that include fried and processed foods tend to be high in omega 6s, while those that include salmon and tuna are higher in omega 3s.
Research has shown that consuming more omega-3 fatty acids is associated with a lower risk for mortality from prostate cancer, explained study investigator William Aronson, MD, with David Geffen School of Medicine at University of California, Los Angeles (UCLA). Research suggests that ingesting more omega-6 accelerates the growth of human tumors in mice, while raising omega-3 levels lowers it. High omega-3 and low omega-6 are also known to have an inhibitory effect on M2-like macrophages, which are the predominant immune cell type in prostate cancer metastasis.
To investigate the impact of these fatty acids on early-stage prostate cancer, Aronson and colleagues conducted a single-center, phase 2, randomized, open-label study in 100 men with grade 1/2 prostate cancer who elected active surveillance.
Patients were randomly allocated 1:1 to a control group that continued their normal diet (minus fish oil) or to an intervention group that followed a low omega-6/high omega-3 diet, supplemented with fish oil (2.2 g/d), for 1 year.
The primary endpoint was the change in Ki-67 index from baseline to 1 year from same-site biopsies between the groups.
For the primary endpoint, the Ki-67 index decreased in the intervention group by 15% from baseline to 1 year and increased in the control group by 24%. The difference between groups was statistically significant (P = .043).
For the secondary endpoints, the intervention led to a reduction in triglyceride levels and macrophage colony stimulating factor but no change in tumor volume grade group, PSA level, or Decipher 22 gene score.
Aronson said the findings support future phase 3 trials incorporating this intervention among men on active surveillance for prostate cancer.
McGregor said it’s important to note that this was “an aggressive intervention with dietary changes and addition of fish oil and patients need to be highly motivated.” Four men discontinued due to adverse effects — primarily gastrointestinal adverse effects such as diarrhea and nausea — larger sample sizes will be key to better understand the tolerability.
Bottom line, said McGregor, “based on this data alone, it should not be recommended but can be considered for highly motivated patients after discussion of the limitations of available data and side effects.”
The study was funded in part by the National Cancer Institute, the UCLA Health Jonsson Comprehensive Cancer Center, Howard B. Klein, and the Seafood Industry Research Fund. Aronson disclosed relationships with AstraZeneca, Bayer, Blue Earth Diagnostics, Janssen Oncology, and Pfizer/Astellas. McGregor disclosed relationships with Arcus Biosciences, Astellas Pharma, Bristol Myers Squibb, Daiichi Sankyo/AstraZeneca, Eisai, Exelixis, Genmab, Gilead Sciences, Loxo/Lilly, Pfizer, and Seattle Genetics/Astellas.
A version of this article first appeared on Medscape.com.
A diet high in omega-3 and low in omega-6 fatty acids, alongside fish oil supplements, may curb the growth of prostate cancer cells in men with early-stage disease, new data showed.
Among men on active surveillance for prostate cancer, consuming this diet for a year led to a significant decrease in the prostate cancer tissue Ki-67 index, a biomarker for prostate cancer progression, metastasis, and death, according to findings from the phase 2 CAPFISH-3 study presented at the 2025 American Society of Clinical Oncology Genitourinary Cancers Symposium.
“This data is certainly intriguing and supports studies looking at this further in prostate cancer,” Bradley Alexander McGregor, MD, from Dana-Farber Cancer Institute, Boston, Massachusetts, who wasn’t involved in the study, told this news organization. But, McGregor noted, patients were on the diet for 1 year, and the long-term implications of this diet are not known.
Growing Evidence on Diet
Diets that include fried and processed foods tend to be high in omega 6s, while those that include salmon and tuna are higher in omega 3s.
Research has shown that consuming more omega-3 fatty acids is associated with a lower risk for mortality from prostate cancer, explained study investigator William Aronson, MD, with David Geffen School of Medicine at University of California, Los Angeles (UCLA). Research suggests that ingesting more omega-6 accelerates the growth of human tumors in mice, while raising omega-3 levels lowers it. High omega-3 and low omega-6 are also known to have an inhibitory effect on M2-like macrophages, which are the predominant immune cell type in prostate cancer metastasis.
To investigate the impact of these fatty acids on early-stage prostate cancer, Aronson and colleagues conducted a single-center, phase 2, randomized, open-label study in 100 men with grade 1/2 prostate cancer who elected active surveillance.
Patients were randomly allocated 1:1 to a control group that continued their normal diet (minus fish oil) or to an intervention group that followed a low omega-6/high omega-3 diet, supplemented with fish oil (2.2 g/d), for 1 year.
The primary endpoint was the change in Ki-67 index from baseline to 1 year from same-site biopsies between the groups.
For the primary endpoint, the Ki-67 index decreased in the intervention group by 15% from baseline to 1 year and increased in the control group by 24%. The difference between groups was statistically significant (P = .043).
For the secondary endpoints, the intervention led to a reduction in triglyceride levels and macrophage colony stimulating factor but no change in tumor volume grade group, PSA level, or Decipher 22 gene score.
Aronson said the findings support future phase 3 trials incorporating this intervention among men on active surveillance for prostate cancer.
McGregor said it’s important to note that this was “an aggressive intervention with dietary changes and addition of fish oil and patients need to be highly motivated.” Four men discontinued due to adverse effects — primarily gastrointestinal adverse effects such as diarrhea and nausea — larger sample sizes will be key to better understand the tolerability.
Bottom line, said McGregor, “based on this data alone, it should not be recommended but can be considered for highly motivated patients after discussion of the limitations of available data and side effects.”
The study was funded in part by the National Cancer Institute, the UCLA Health Jonsson Comprehensive Cancer Center, Howard B. Klein, and the Seafood Industry Research Fund. Aronson disclosed relationships with AstraZeneca, Bayer, Blue Earth Diagnostics, Janssen Oncology, and Pfizer/Astellas. McGregor disclosed relationships with Arcus Biosciences, Astellas Pharma, Bristol Myers Squibb, Daiichi Sankyo/AstraZeneca, Eisai, Exelixis, Genmab, Gilead Sciences, Loxo/Lilly, Pfizer, and Seattle Genetics/Astellas.
A version of this article first appeared on Medscape.com.
A diet high in omega-3 and low in omega-6 fatty acids, alongside fish oil supplements, may curb the growth of prostate cancer cells in men with early-stage disease, new data showed.
Among men on active surveillance for prostate cancer, consuming this diet for a year led to a significant decrease in the prostate cancer tissue Ki-67 index, a biomarker for prostate cancer progression, metastasis, and death, according to findings from the phase 2 CAPFISH-3 study presented at the 2025 American Society of Clinical Oncology Genitourinary Cancers Symposium.
“This data is certainly intriguing and supports studies looking at this further in prostate cancer,” Bradley Alexander McGregor, MD, from Dana-Farber Cancer Institute, Boston, Massachusetts, who wasn’t involved in the study, told this news organization. But, McGregor noted, patients were on the diet for 1 year, and the long-term implications of this diet are not known.
Growing Evidence on Diet
Diets that include fried and processed foods tend to be high in omega 6s, while those that include salmon and tuna are higher in omega 3s.
Research has shown that consuming more omega-3 fatty acids is associated with a lower risk for mortality from prostate cancer, explained study investigator William Aronson, MD, with David Geffen School of Medicine at University of California, Los Angeles (UCLA). Research suggests that ingesting more omega-6 accelerates the growth of human tumors in mice, while raising omega-3 levels lowers it. High omega-3 and low omega-6 are also known to have an inhibitory effect on M2-like macrophages, which are the predominant immune cell type in prostate cancer metastasis.
To investigate the impact of these fatty acids on early-stage prostate cancer, Aronson and colleagues conducted a single-center, phase 2, randomized, open-label study in 100 men with grade 1/2 prostate cancer who elected active surveillance.
Patients were randomly allocated 1:1 to a control group that continued their normal diet (minus fish oil) or to an intervention group that followed a low omega-6/high omega-3 diet, supplemented with fish oil (2.2 g/d), for 1 year.
The primary endpoint was the change in Ki-67 index from baseline to 1 year from same-site biopsies between the groups.
For the primary endpoint, the Ki-67 index decreased in the intervention group by 15% from baseline to 1 year and increased in the control group by 24%. The difference between groups was statistically significant (P = .043).
For the secondary endpoints, the intervention led to a reduction in triglyceride levels and macrophage colony stimulating factor but no change in tumor volume grade group, PSA level, or Decipher 22 gene score.
Aronson said the findings support future phase 3 trials incorporating this intervention among men on active surveillance for prostate cancer.
McGregor said it’s important to note that this was “an aggressive intervention with dietary changes and addition of fish oil and patients need to be highly motivated.” Four men discontinued due to adverse effects — primarily gastrointestinal adverse effects such as diarrhea and nausea — larger sample sizes will be key to better understand the tolerability.
Bottom line, said McGregor, “based on this data alone, it should not be recommended but can be considered for highly motivated patients after discussion of the limitations of available data and side effects.”
The study was funded in part by the National Cancer Institute, the UCLA Health Jonsson Comprehensive Cancer Center, Howard B. Klein, and the Seafood Industry Research Fund. Aronson disclosed relationships with AstraZeneca, Bayer, Blue Earth Diagnostics, Janssen Oncology, and Pfizer/Astellas. McGregor disclosed relationships with Arcus Biosciences, Astellas Pharma, Bristol Myers Squibb, Daiichi Sankyo/AstraZeneca, Eisai, Exelixis, Genmab, Gilead Sciences, Loxo/Lilly, Pfizer, and Seattle Genetics/Astellas.
A version of this article first appeared on Medscape.com.
FROM GUCS 2025
New Clues to Links Between Gulf War Illness and Anthrax Vaccine
TOPLINE: Gulf War Illness (GWI) symptom severity shows negative correlation with predicted binding affinity of anthrax vaccine antigen to Human Leukocyte Antigen (HLA) Class II molecules. Stronger binding affinity is associated with weaker symptoms, with correlation coefficient r = -0.356 (P < .001).
METHODOLOGY:
- Researchers analyzed 458 Gulf War veterans: 397 men, 61 women, mean (SD) age 56.3 (0.5) years.
- The aim was to determine the association between GWI symptom severity and binding affinity of anthrax Protective Antigen to HLA Class II molecules.
- Analysis included in silico estimation of predicted binding affinity between 750 15-amino acid length subsequences of protective antigen and specific HLA Class II alleles carried by each participant.
- Investigators assessed GWI symptom severity across 6 domains: fatigue, pain, neurological/cognitive/mood, respiratory, gastrointestinal, and dermatologic symptoms that began during or after Gulf War and lasted > 6 months.
TAKEAWAY:
- GWI symptom severity demonstrated significant negative correlation with strength of predicted binding affinity of protective antigen peptides to HLA-II molecules (correlation coefficient [r], −0.356; P < .001), independent of age (partial correlation, −0.376; P < .001).
- Researchers identified 180 of 750 (24%) 15-mer epitopes with strong binding affinities to HLA-II molecules, suggesting good potential for CD4+ lymphocyte engagement.
- Analysis revealed that DPB1 (15/31, 48%) and DRB1 (13/44, 30%) alleles showed strong binding affinity with Protective Antigen epitopes, while all DQB1 alleles (18/18, 100%) showed no strong binding.
- The number of strong binding hits per individual ranged from 3 to 168, indicating wide variability in potential antibody production capability across participants.
IN PRACTICE: "The current findings, demonstrating a robust negative association between HLAanthrax vaccine PA binding and GWI symptom severity, strongly support the hypothesized role of reduced antibody production against anthrax vaccine PA in GWI that most probably underlies the findings supporting anthrax antigen persistence in GWI, in the broader context of antigen persistence in other diseases," Lisa M. James and Apostolos P. Georgopoulos write.
SOURCE: The study was led by Lisa M. James and Apostolos P. Georgopoulos of the Brain Sciences Center at the Minneapolis Veterans Affairs Health Care System. It was published online on January 18 in Vaccines.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Gulf War Illness (GWI) symptom severity shows negative correlation with predicted binding affinity of anthrax vaccine antigen to Human Leukocyte Antigen (HLA) Class II molecules. Stronger binding affinity is associated with weaker symptoms, with correlation coefficient r = -0.356 (P < .001).
METHODOLOGY:
- Researchers analyzed 458 Gulf War veterans: 397 men, 61 women, mean (SD) age 56.3 (0.5) years.
- The aim was to determine the association between GWI symptom severity and binding affinity of anthrax Protective Antigen to HLA Class II molecules.
- Analysis included in silico estimation of predicted binding affinity between 750 15-amino acid length subsequences of protective antigen and specific HLA Class II alleles carried by each participant.
- Investigators assessed GWI symptom severity across 6 domains: fatigue, pain, neurological/cognitive/mood, respiratory, gastrointestinal, and dermatologic symptoms that began during or after Gulf War and lasted > 6 months.
TAKEAWAY:
- GWI symptom severity demonstrated significant negative correlation with strength of predicted binding affinity of protective antigen peptides to HLA-II molecules (correlation coefficient [r], −0.356; P < .001), independent of age (partial correlation, −0.376; P < .001).
- Researchers identified 180 of 750 (24%) 15-mer epitopes with strong binding affinities to HLA-II molecules, suggesting good potential for CD4+ lymphocyte engagement.
- Analysis revealed that DPB1 (15/31, 48%) and DRB1 (13/44, 30%) alleles showed strong binding affinity with Protective Antigen epitopes, while all DQB1 alleles (18/18, 100%) showed no strong binding.
- The number of strong binding hits per individual ranged from 3 to 168, indicating wide variability in potential antibody production capability across participants.
IN PRACTICE: "The current findings, demonstrating a robust negative association between HLAanthrax vaccine PA binding and GWI symptom severity, strongly support the hypothesized role of reduced antibody production against anthrax vaccine PA in GWI that most probably underlies the findings supporting anthrax antigen persistence in GWI, in the broader context of antigen persistence in other diseases," Lisa M. James and Apostolos P. Georgopoulos write.
SOURCE: The study was led by Lisa M. James and Apostolos P. Georgopoulos of the Brain Sciences Center at the Minneapolis Veterans Affairs Health Care System. It was published online on January 18 in Vaccines.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Gulf War Illness (GWI) symptom severity shows negative correlation with predicted binding affinity of anthrax vaccine antigen to Human Leukocyte Antigen (HLA) Class II molecules. Stronger binding affinity is associated with weaker symptoms, with correlation coefficient r = -0.356 (P < .001).
METHODOLOGY:
- Researchers analyzed 458 Gulf War veterans: 397 men, 61 women, mean (SD) age 56.3 (0.5) years.
- The aim was to determine the association between GWI symptom severity and binding affinity of anthrax Protective Antigen to HLA Class II molecules.
- Analysis included in silico estimation of predicted binding affinity between 750 15-amino acid length subsequences of protective antigen and specific HLA Class II alleles carried by each participant.
- Investigators assessed GWI symptom severity across 6 domains: fatigue, pain, neurological/cognitive/mood, respiratory, gastrointestinal, and dermatologic symptoms that began during or after Gulf War and lasted > 6 months.
TAKEAWAY:
- GWI symptom severity demonstrated significant negative correlation with strength of predicted binding affinity of protective antigen peptides to HLA-II molecules (correlation coefficient [r], −0.356; P < .001), independent of age (partial correlation, −0.376; P < .001).
- Researchers identified 180 of 750 (24%) 15-mer epitopes with strong binding affinities to HLA-II molecules, suggesting good potential for CD4+ lymphocyte engagement.
- Analysis revealed that DPB1 (15/31, 48%) and DRB1 (13/44, 30%) alleles showed strong binding affinity with Protective Antigen epitopes, while all DQB1 alleles (18/18, 100%) showed no strong binding.
- The number of strong binding hits per individual ranged from 3 to 168, indicating wide variability in potential antibody production capability across participants.
IN PRACTICE: "The current findings, demonstrating a robust negative association between HLAanthrax vaccine PA binding and GWI symptom severity, strongly support the hypothesized role of reduced antibody production against anthrax vaccine PA in GWI that most probably underlies the findings supporting anthrax antigen persistence in GWI, in the broader context of antigen persistence in other diseases," Lisa M. James and Apostolos P. Georgopoulos write.
SOURCE: The study was led by Lisa M. James and Apostolos P. Georgopoulos of the Brain Sciences Center at the Minneapolis Veterans Affairs Health Care System. It was published online on January 18 in Vaccines.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Vasculitis Patients Need Multiple COVID Vaccine Boosters
People with vasculitis may need at least three or four vaccinations for COVID-19 before they start to show an immune response against SARS-CoV-2 infection, new research has suggested.
In a longitudinal retrospective study, serum antibody neutralization against the Omicron variant of the virus and its descendants was found to be “largely absent” after the first two doses of COVID-19 vaccine had been given to patients. But increasing neutralizing antibody titers were seen after both the third and fourth vaccine boosters had been administered.
Results also showed that the more recently people had been treated with the B cell–depleting therapy rituximab, the lower the levels of immunogenicity that were achieved, and thus protection against SARS-CoV-2.
“Our results have significant implications for individuals treated with rituximab in the post-Omicron era, highlighting the value of additive boosters in affirming increasing protection in clinically vulnerable populations,” the team behind the work at the University of Cambridge in England, has reported in Science Advances.
Moreover, because the use of rituximab reduced the neutralization of not just wild-type (WT) Omicron but also the Omicron-descendant variants BA.1, BA.2, BA.4, and XBB, this highlights “the urgent need for additional adjunctive strategies to enhance vaccine-induced immunity as well as preferential access for such patients to updated vaccines using spike from now circulating Omicron lineages,” the team added.
Studying Humoral Responses to SARS-CoV-2 Vaccines
Corresponding author Ravindra K. Gupta, BMBCh, MA, MPH, PhD, told this news organization that studying humoral responses to SARS-CoV-2 vaccines in immunocompromised individuals such as those with vasculitis was important for two main reasons.
“It is really important at individual level for their own health, of course, but also because we know that variants of concern have often evolved and developed within patients and can then spread in wider populations,” he said.
Gupta, who is professor of clinical microbiology at the Cambridge Institute for Therapeutic Immunology & Infectious Disease added: “We believe that the variants of concern that we’re having to deal with right now, including Omicron, have come from such [immunocompromised] individuals.”
Omicron “was a big shift,” Gupta noted. “It had a lot of new mutations on it, so it was almost like a new strain of the virus.” Few studies have looked at the longitudinal immunogenicity proffered by COVID vaccines in the post-Omicron era, particularly in those with vasculitis who are often treated with immunosuppressive drugs, including rituximab.
Two-Pronged Study Approach
For the study, a population of immunocompromised individuals diagnosed with vasculitis who had been treated with rituximab in the past 5 years was identified. Just over half (58%) had received adenovirus-based AZD1222/ChAdOx1 nCoV-19 (AstraZeneca-Oxford; AZN) and 37% BNT162b2 (Pfizer-BioNTech; mRNA) as their primary vaccines. Patients with antineutrophil cytoplasmic antibody–associated vasculitis comprised the majority of those who received rituximab (83%), compared with less than half of those who did not take rituximab (48%).
A two-pronged approach was taken with the researchers first measuring neutralizing antibody titers before and 30 days after four successive COVID vaccinations in a group of 32 individuals with available samples. They then performed a cross-sectional, case-control study in 95 individuals to look at neutralizing antibody titers and antibody-dependent cell-mediated cytotoxicity (ADCC) in individuals who had (n = 64) and had not (n = 31) been treated with rituximab in the past 5 years and had samples available after their third and fourth COVID vaccinations.
The first analysis was done to see how people were responding to vaccination over time. “That told us that there was a problem with the first two doses and that we got some response after doses three and four, but the response was uniformly quite poor against the new variants of concern,” Gupta said.
A human embryonic kidney cell model had been used to determine individuals’ neutralizing antibody titers in response to WT, BA.1, BA.2, BA.4, and XBB pseudotyped viruses. After the first and second COVID vaccinations, the geometric mean titer (GMT) against each variant barely increased from a baseline of 40.0. The greatest increases in GMT was seen with the WT virus, at 43.7, 90.7, 256.3, and 394.2, after the first, second, third and fourth doses, respectively. The lowest increases in GMT were seen with the XBB variant, with respective values of 40.0, 40.8, 45.7, and 53.9.
Incremental Benefit Offers Some ‘Reassurance’
Vasculitis specialist Rona Smith, MA, MB BChir, MD, who was one of the authors of the paper, told this news organization separately that the results showed there was “an incremental benefit of having COVID vaccinations,” which “offers a little bit of reassurance” that there can be an immune response in people with vasculitis.
Although results of the cross-sectional study showed that there was a significant dampening effect of rituximab treatment on the immune response, “I don’t think it’s an isolated effect in our [vasculitis] patients,” Smith suggested, adding the results were “probably still relevant to patients who receive routine dosing of rituximab for other conditions.”
Neutralizing antibody titers were consistently lower among individuals who had been treated with rituximab vs those who had not, with treatment in the past 18 months found to significantly impair immunogenicity.
The ADCC response was better preserved than the neutralizing antibody response, Gupta said, although it was still significantly lower in the rituximab-treated than in the non–rituximab-treated patients.
When to Vaccinate in Vasculitis?
Regarding when to give vaccines to people with vasculitis, Smith said: “Current recommendations are that patients should receive any vaccines that they’re offered routinely, whether that be COVID vaccines, flu vaccines, pneumococcal vaccines.”
As for the timing of those vaccinations, she observed that the current thinking was that vaccinations should “ideally be at least 1 month before a rituximab treatment, and ideally 3-4 [months] after their last dose. However, as many patients are on a 6-month dosing cycle, it can be difficult for some of them to find a suitable time window to have the COVID vaccine when it is offered.”
Additional precautions, such as wearing masks in crowded places and avoiding visits to acutely unwell friends or relatives, may still be prudent, Smith acknowledged, but he was clear that people should not be locking themselves away as they did during the COVID-19 pandemic.
When advising patients, “our general recommendation is that it is better to have a vaccine than not, but we can’t guarantee how well you will respond to it, but some response is better than none,” Smith said.
The study was independently supported. Gupta had no relevant financial relationships to disclose. Smith was a coauthor of the paper and has received research grant funding from Union Therapeutics, GlaxoSmithKline/Vir Biotechnology, Addenbrooke’s Charitable Trust, and Vasculitis UK. Another coauthor reported receiving research grants from CSL Vifor, Roche, and GlaxoSmithKline and advisory board, consultancy, and lecture fees from Roche and CSL Vifor.
A version of this article first appeared on Medscape.com.
People with vasculitis may need at least three or four vaccinations for COVID-19 before they start to show an immune response against SARS-CoV-2 infection, new research has suggested.
In a longitudinal retrospective study, serum antibody neutralization against the Omicron variant of the virus and its descendants was found to be “largely absent” after the first two doses of COVID-19 vaccine had been given to patients. But increasing neutralizing antibody titers were seen after both the third and fourth vaccine boosters had been administered.
Results also showed that the more recently people had been treated with the B cell–depleting therapy rituximab, the lower the levels of immunogenicity that were achieved, and thus protection against SARS-CoV-2.
“Our results have significant implications for individuals treated with rituximab in the post-Omicron era, highlighting the value of additive boosters in affirming increasing protection in clinically vulnerable populations,” the team behind the work at the University of Cambridge in England, has reported in Science Advances.
Moreover, because the use of rituximab reduced the neutralization of not just wild-type (WT) Omicron but also the Omicron-descendant variants BA.1, BA.2, BA.4, and XBB, this highlights “the urgent need for additional adjunctive strategies to enhance vaccine-induced immunity as well as preferential access for such patients to updated vaccines using spike from now circulating Omicron lineages,” the team added.
Studying Humoral Responses to SARS-CoV-2 Vaccines
Corresponding author Ravindra K. Gupta, BMBCh, MA, MPH, PhD, told this news organization that studying humoral responses to SARS-CoV-2 vaccines in immunocompromised individuals such as those with vasculitis was important for two main reasons.
“It is really important at individual level for their own health, of course, but also because we know that variants of concern have often evolved and developed within patients and can then spread in wider populations,” he said.
Gupta, who is professor of clinical microbiology at the Cambridge Institute for Therapeutic Immunology & Infectious Disease added: “We believe that the variants of concern that we’re having to deal with right now, including Omicron, have come from such [immunocompromised] individuals.”
Omicron “was a big shift,” Gupta noted. “It had a lot of new mutations on it, so it was almost like a new strain of the virus.” Few studies have looked at the longitudinal immunogenicity proffered by COVID vaccines in the post-Omicron era, particularly in those with vasculitis who are often treated with immunosuppressive drugs, including rituximab.
Two-Pronged Study Approach
For the study, a population of immunocompromised individuals diagnosed with vasculitis who had been treated with rituximab in the past 5 years was identified. Just over half (58%) had received adenovirus-based AZD1222/ChAdOx1 nCoV-19 (AstraZeneca-Oxford; AZN) and 37% BNT162b2 (Pfizer-BioNTech; mRNA) as their primary vaccines. Patients with antineutrophil cytoplasmic antibody–associated vasculitis comprised the majority of those who received rituximab (83%), compared with less than half of those who did not take rituximab (48%).
A two-pronged approach was taken with the researchers first measuring neutralizing antibody titers before and 30 days after four successive COVID vaccinations in a group of 32 individuals with available samples. They then performed a cross-sectional, case-control study in 95 individuals to look at neutralizing antibody titers and antibody-dependent cell-mediated cytotoxicity (ADCC) in individuals who had (n = 64) and had not (n = 31) been treated with rituximab in the past 5 years and had samples available after their third and fourth COVID vaccinations.
The first analysis was done to see how people were responding to vaccination over time. “That told us that there was a problem with the first two doses and that we got some response after doses three and four, but the response was uniformly quite poor against the new variants of concern,” Gupta said.
A human embryonic kidney cell model had been used to determine individuals’ neutralizing antibody titers in response to WT, BA.1, BA.2, BA.4, and XBB pseudotyped viruses. After the first and second COVID vaccinations, the geometric mean titer (GMT) against each variant barely increased from a baseline of 40.0. The greatest increases in GMT was seen with the WT virus, at 43.7, 90.7, 256.3, and 394.2, after the first, second, third and fourth doses, respectively. The lowest increases in GMT were seen with the XBB variant, with respective values of 40.0, 40.8, 45.7, and 53.9.
Incremental Benefit Offers Some ‘Reassurance’
Vasculitis specialist Rona Smith, MA, MB BChir, MD, who was one of the authors of the paper, told this news organization separately that the results showed there was “an incremental benefit of having COVID vaccinations,” which “offers a little bit of reassurance” that there can be an immune response in people with vasculitis.
Although results of the cross-sectional study showed that there was a significant dampening effect of rituximab treatment on the immune response, “I don’t think it’s an isolated effect in our [vasculitis] patients,” Smith suggested, adding the results were “probably still relevant to patients who receive routine dosing of rituximab for other conditions.”
Neutralizing antibody titers were consistently lower among individuals who had been treated with rituximab vs those who had not, with treatment in the past 18 months found to significantly impair immunogenicity.
The ADCC response was better preserved than the neutralizing antibody response, Gupta said, although it was still significantly lower in the rituximab-treated than in the non–rituximab-treated patients.
When to Vaccinate in Vasculitis?
Regarding when to give vaccines to people with vasculitis, Smith said: “Current recommendations are that patients should receive any vaccines that they’re offered routinely, whether that be COVID vaccines, flu vaccines, pneumococcal vaccines.”
As for the timing of those vaccinations, she observed that the current thinking was that vaccinations should “ideally be at least 1 month before a rituximab treatment, and ideally 3-4 [months] after their last dose. However, as many patients are on a 6-month dosing cycle, it can be difficult for some of them to find a suitable time window to have the COVID vaccine when it is offered.”
Additional precautions, such as wearing masks in crowded places and avoiding visits to acutely unwell friends or relatives, may still be prudent, Smith acknowledged, but he was clear that people should not be locking themselves away as they did during the COVID-19 pandemic.
When advising patients, “our general recommendation is that it is better to have a vaccine than not, but we can’t guarantee how well you will respond to it, but some response is better than none,” Smith said.
The study was independently supported. Gupta had no relevant financial relationships to disclose. Smith was a coauthor of the paper and has received research grant funding from Union Therapeutics, GlaxoSmithKline/Vir Biotechnology, Addenbrooke’s Charitable Trust, and Vasculitis UK. Another coauthor reported receiving research grants from CSL Vifor, Roche, and GlaxoSmithKline and advisory board, consultancy, and lecture fees from Roche and CSL Vifor.
A version of this article first appeared on Medscape.com.
People with vasculitis may need at least three or four vaccinations for COVID-19 before they start to show an immune response against SARS-CoV-2 infection, new research has suggested.
In a longitudinal retrospective study, serum antibody neutralization against the Omicron variant of the virus and its descendants was found to be “largely absent” after the first two doses of COVID-19 vaccine had been given to patients. But increasing neutralizing antibody titers were seen after both the third and fourth vaccine boosters had been administered.
Results also showed that the more recently people had been treated with the B cell–depleting therapy rituximab, the lower the levels of immunogenicity that were achieved, and thus protection against SARS-CoV-2.
“Our results have significant implications for individuals treated with rituximab in the post-Omicron era, highlighting the value of additive boosters in affirming increasing protection in clinically vulnerable populations,” the team behind the work at the University of Cambridge in England, has reported in Science Advances.
Moreover, because the use of rituximab reduced the neutralization of not just wild-type (WT) Omicron but also the Omicron-descendant variants BA.1, BA.2, BA.4, and XBB, this highlights “the urgent need for additional adjunctive strategies to enhance vaccine-induced immunity as well as preferential access for such patients to updated vaccines using spike from now circulating Omicron lineages,” the team added.
Studying Humoral Responses to SARS-CoV-2 Vaccines
Corresponding author Ravindra K. Gupta, BMBCh, MA, MPH, PhD, told this news organization that studying humoral responses to SARS-CoV-2 vaccines in immunocompromised individuals such as those with vasculitis was important for two main reasons.
“It is really important at individual level for their own health, of course, but also because we know that variants of concern have often evolved and developed within patients and can then spread in wider populations,” he said.
Gupta, who is professor of clinical microbiology at the Cambridge Institute for Therapeutic Immunology & Infectious Disease added: “We believe that the variants of concern that we’re having to deal with right now, including Omicron, have come from such [immunocompromised] individuals.”
Omicron “was a big shift,” Gupta noted. “It had a lot of new mutations on it, so it was almost like a new strain of the virus.” Few studies have looked at the longitudinal immunogenicity proffered by COVID vaccines in the post-Omicron era, particularly in those with vasculitis who are often treated with immunosuppressive drugs, including rituximab.
Two-Pronged Study Approach
For the study, a population of immunocompromised individuals diagnosed with vasculitis who had been treated with rituximab in the past 5 years was identified. Just over half (58%) had received adenovirus-based AZD1222/ChAdOx1 nCoV-19 (AstraZeneca-Oxford; AZN) and 37% BNT162b2 (Pfizer-BioNTech; mRNA) as their primary vaccines. Patients with antineutrophil cytoplasmic antibody–associated vasculitis comprised the majority of those who received rituximab (83%), compared with less than half of those who did not take rituximab (48%).
A two-pronged approach was taken with the researchers first measuring neutralizing antibody titers before and 30 days after four successive COVID vaccinations in a group of 32 individuals with available samples. They then performed a cross-sectional, case-control study in 95 individuals to look at neutralizing antibody titers and antibody-dependent cell-mediated cytotoxicity (ADCC) in individuals who had (n = 64) and had not (n = 31) been treated with rituximab in the past 5 years and had samples available after their third and fourth COVID vaccinations.
The first analysis was done to see how people were responding to vaccination over time. “That told us that there was a problem with the first two doses and that we got some response after doses three and four, but the response was uniformly quite poor against the new variants of concern,” Gupta said.
A human embryonic kidney cell model had been used to determine individuals’ neutralizing antibody titers in response to WT, BA.1, BA.2, BA.4, and XBB pseudotyped viruses. After the first and second COVID vaccinations, the geometric mean titer (GMT) against each variant barely increased from a baseline of 40.0. The greatest increases in GMT was seen with the WT virus, at 43.7, 90.7, 256.3, and 394.2, after the first, second, third and fourth doses, respectively. The lowest increases in GMT were seen with the XBB variant, with respective values of 40.0, 40.8, 45.7, and 53.9.
Incremental Benefit Offers Some ‘Reassurance’
Vasculitis specialist Rona Smith, MA, MB BChir, MD, who was one of the authors of the paper, told this news organization separately that the results showed there was “an incremental benefit of having COVID vaccinations,” which “offers a little bit of reassurance” that there can be an immune response in people with vasculitis.
Although results of the cross-sectional study showed that there was a significant dampening effect of rituximab treatment on the immune response, “I don’t think it’s an isolated effect in our [vasculitis] patients,” Smith suggested, adding the results were “probably still relevant to patients who receive routine dosing of rituximab for other conditions.”
Neutralizing antibody titers were consistently lower among individuals who had been treated with rituximab vs those who had not, with treatment in the past 18 months found to significantly impair immunogenicity.
The ADCC response was better preserved than the neutralizing antibody response, Gupta said, although it was still significantly lower in the rituximab-treated than in the non–rituximab-treated patients.
When to Vaccinate in Vasculitis?
Regarding when to give vaccines to people with vasculitis, Smith said: “Current recommendations are that patients should receive any vaccines that they’re offered routinely, whether that be COVID vaccines, flu vaccines, pneumococcal vaccines.”
As for the timing of those vaccinations, she observed that the current thinking was that vaccinations should “ideally be at least 1 month before a rituximab treatment, and ideally 3-4 [months] after their last dose. However, as many patients are on a 6-month dosing cycle, it can be difficult for some of them to find a suitable time window to have the COVID vaccine when it is offered.”
Additional precautions, such as wearing masks in crowded places and avoiding visits to acutely unwell friends or relatives, may still be prudent, Smith acknowledged, but he was clear that people should not be locking themselves away as they did during the COVID-19 pandemic.
When advising patients, “our general recommendation is that it is better to have a vaccine than not, but we can’t guarantee how well you will respond to it, but some response is better than none,” Smith said.
The study was independently supported. Gupta had no relevant financial relationships to disclose. Smith was a coauthor of the paper and has received research grant funding from Union Therapeutics, GlaxoSmithKline/Vir Biotechnology, Addenbrooke’s Charitable Trust, and Vasculitis UK. Another coauthor reported receiving research grants from CSL Vifor, Roche, and GlaxoSmithKline and advisory board, consultancy, and lecture fees from Roche and CSL Vifor.
A version of this article first appeared on Medscape.com.
FROM SCIENCE ADVANCES