User login
To dream the maybe possible dream: A breast cancer vaccine
The Journal generally does not publish articles on topics not yet clinically relevant. But since the topic of immunization is so often in the news, and since immunotherapeutic strategies against cancer continue to be tested in clinical trials, we decided to include in this issue an edited transcript of a Medicine Grand Rounds presentation at Cleveland Clinic by Dr. Vincent Tuohy on a novel strategy to develop a vaccine against a particularly virulent form of breast cancer.
The immune system’s response to cancer is complex. Melanoma and renal cell carcinoma seem particularly susceptible to suppression by the native or augmented immune response. But most cancers seem to grow—and many metastasize—seemingly unaffected by our immune system, and sometimes even in the presence of detectable antitumor cell-directed lymphocytes and antibodies. Many attempts at devising human antitumor vaccines and immunotherapies have failed. On the other hand, we have seen the successful development of effective monoclonal antibody therapies (eg, rituximab for B cell lymphoma), immunomodulatory treatments for patients with advanced disease, and vaccines against viruses that cause cancer, ie, human papillomavirus and hepatitis B.
To fully appreciate the nuances of Dr. Tuohy’s proposed strategy, which has not yet been tested in clinical trials, and the complexities of tumor immunology, a very brief primer on the challenges is in order.
CHALLENGES TO DEVELOPING CANCER IMMUNOTHERAPY
Solid tumors can be triggered by multiple mechanisms, alone or in combination, including viruses, spontaneous mutations, overexpression of tumor promoters, and underexpression of tumor suppressors. Once growing, the solid tumor establishes its own rogue growth community, complete with a new infrastructure to supply nutrition and oxygen, potential means for expansion locally and distally, and a system to defend itself from the body’s immune system. The last of these poses specific challenges to successful spontaneous immune surveillance and to immunotherapy designed to kill cancer cells.
Microbial pathogens trigger both a nonspecific (innate) and a specific immune response in the human body. Initially, the immune system is nonspecifically “revved up,” triggered by shared “danger signals” associated with the perceived pathogen and its specific antigens. Then, specialized cells including dendritic cells locally and in proximate lymph nodes are primed to present the de novo antigens in a way that generates a specific and maturing immune response capable of getting rid of the pathogen. Tumors are also pathogenic and in some ways “foreign.” However, they are also similar to normal tissue and interact quite differently with the immune response in ways that enhance their likelihood of growth and survival. Tumor cells often do not send a danger signal to the immune system akin to what is generated by a staphylococcal or mycobacterial invader.
Tumor cells express specific antigens on their surface, such as viral proteins, cancer-associated mutated proteins, and overexpressed differentiated or undifferentiated antigens, including in some cases what Dr. Tuohy discusses as “retired proteins.” But some of these antigens may also be expressed in normal tissues, especially in an environment of resolving inflammation. Some signal the immune system to down-regulate what could otherwise be a vigorous self-destructive response every time there was inflammation.
The dendritic cell activation of the potential antitumor T-cell response and the antitumor T-cell response itself seem to be systematically blunted by many tumors. Reversal of this blunting represents one strategy currently used with very modest clinical success in treating advanced melanoma. Some newly generated tumor-antigen–recognizing T cells may in fact exert suppressor (or regulator) and not cytotoxic activity. Some tumors exhibit systemic immunosuppressive activity; this can be manifested not only by unchecked tumor growth, but also by an increased susceptibility to certain infections.
PITFALLS OF MESSING WITH THE IMMUNE SYSTEM
Messing with the immune system is not without pitfalls. Not all toxicities will be predicted by preclinical animal studies, and human immunity is not a mirror image of rodents’ or even other primates’ immune systems. Augmentation of an antitumor response, in part from the interplay of the complexities noted above, may lead to destruction of normal tissue elsewhere, or even to disruption of tolerance with the expression of autoimmunity. The toxicities will be different than the somewhat predictable toxicities from traditional antiproliferative chemotherapies—witness the striking systemic toxicity from interleukin 2-based therapies.
Whether Dr. Tuohy’s approach to developing a tumor vaccine will ultimately reach our formularies remains to be seen. The work is in an extremely preliminary phase. But the concept of immunotherapy for cancer remains an active area of research that is worth keeping an eye on.
The Journal generally does not publish articles on topics not yet clinically relevant. But since the topic of immunization is so often in the news, and since immunotherapeutic strategies against cancer continue to be tested in clinical trials, we decided to include in this issue an edited transcript of a Medicine Grand Rounds presentation at Cleveland Clinic by Dr. Vincent Tuohy on a novel strategy to develop a vaccine against a particularly virulent form of breast cancer.
The immune system’s response to cancer is complex. Melanoma and renal cell carcinoma seem particularly susceptible to suppression by the native or augmented immune response. But most cancers seem to grow—and many metastasize—seemingly unaffected by our immune system, and sometimes even in the presence of detectable antitumor cell-directed lymphocytes and antibodies. Many attempts at devising human antitumor vaccines and immunotherapies have failed. On the other hand, we have seen the successful development of effective monoclonal antibody therapies (eg, rituximab for B cell lymphoma), immunomodulatory treatments for patients with advanced disease, and vaccines against viruses that cause cancer, ie, human papillomavirus and hepatitis B.
To fully appreciate the nuances of Dr. Tuohy’s proposed strategy, which has not yet been tested in clinical trials, and the complexities of tumor immunology, a very brief primer on the challenges is in order.
CHALLENGES TO DEVELOPING CANCER IMMUNOTHERAPY
Solid tumors can be triggered by multiple mechanisms, alone or in combination, including viruses, spontaneous mutations, overexpression of tumor promoters, and underexpression of tumor suppressors. Once growing, the solid tumor establishes its own rogue growth community, complete with a new infrastructure to supply nutrition and oxygen, potential means for expansion locally and distally, and a system to defend itself from the body’s immune system. The last of these poses specific challenges to successful spontaneous immune surveillance and to immunotherapy designed to kill cancer cells.
Microbial pathogens trigger both a nonspecific (innate) and a specific immune response in the human body. Initially, the immune system is nonspecifically “revved up,” triggered by shared “danger signals” associated with the perceived pathogen and its specific antigens. Then, specialized cells including dendritic cells locally and in proximate lymph nodes are primed to present the de novo antigens in a way that generates a specific and maturing immune response capable of getting rid of the pathogen. Tumors are also pathogenic and in some ways “foreign.” However, they are also similar to normal tissue and interact quite differently with the immune response in ways that enhance their likelihood of growth and survival. Tumor cells often do not send a danger signal to the immune system akin to what is generated by a staphylococcal or mycobacterial invader.
Tumor cells express specific antigens on their surface, such as viral proteins, cancer-associated mutated proteins, and overexpressed differentiated or undifferentiated antigens, including in some cases what Dr. Tuohy discusses as “retired proteins.” But some of these antigens may also be expressed in normal tissues, especially in an environment of resolving inflammation. Some signal the immune system to down-regulate what could otherwise be a vigorous self-destructive response every time there was inflammation.
The dendritic cell activation of the potential antitumor T-cell response and the antitumor T-cell response itself seem to be systematically blunted by many tumors. Reversal of this blunting represents one strategy currently used with very modest clinical success in treating advanced melanoma. Some newly generated tumor-antigen–recognizing T cells may in fact exert suppressor (or regulator) and not cytotoxic activity. Some tumors exhibit systemic immunosuppressive activity; this can be manifested not only by unchecked tumor growth, but also by an increased susceptibility to certain infections.
PITFALLS OF MESSING WITH THE IMMUNE SYSTEM
Messing with the immune system is not without pitfalls. Not all toxicities will be predicted by preclinical animal studies, and human immunity is not a mirror image of rodents’ or even other primates’ immune systems. Augmentation of an antitumor response, in part from the interplay of the complexities noted above, may lead to destruction of normal tissue elsewhere, or even to disruption of tolerance with the expression of autoimmunity. The toxicities will be different than the somewhat predictable toxicities from traditional antiproliferative chemotherapies—witness the striking systemic toxicity from interleukin 2-based therapies.
Whether Dr. Tuohy’s approach to developing a tumor vaccine will ultimately reach our formularies remains to be seen. The work is in an extremely preliminary phase. But the concept of immunotherapy for cancer remains an active area of research that is worth keeping an eye on.
The Journal generally does not publish articles on topics not yet clinically relevant. But since the topic of immunization is so often in the news, and since immunotherapeutic strategies against cancer continue to be tested in clinical trials, we decided to include in this issue an edited transcript of a Medicine Grand Rounds presentation at Cleveland Clinic by Dr. Vincent Tuohy on a novel strategy to develop a vaccine against a particularly virulent form of breast cancer.
The immune system’s response to cancer is complex. Melanoma and renal cell carcinoma seem particularly susceptible to suppression by the native or augmented immune response. But most cancers seem to grow—and many metastasize—seemingly unaffected by our immune system, and sometimes even in the presence of detectable antitumor cell-directed lymphocytes and antibodies. Many attempts at devising human antitumor vaccines and immunotherapies have failed. On the other hand, we have seen the successful development of effective monoclonal antibody therapies (eg, rituximab for B cell lymphoma), immunomodulatory treatments for patients with advanced disease, and vaccines against viruses that cause cancer, ie, human papillomavirus and hepatitis B.
To fully appreciate the nuances of Dr. Tuohy’s proposed strategy, which has not yet been tested in clinical trials, and the complexities of tumor immunology, a very brief primer on the challenges is in order.
CHALLENGES TO DEVELOPING CANCER IMMUNOTHERAPY
Solid tumors can be triggered by multiple mechanisms, alone or in combination, including viruses, spontaneous mutations, overexpression of tumor promoters, and underexpression of tumor suppressors. Once growing, the solid tumor establishes its own rogue growth community, complete with a new infrastructure to supply nutrition and oxygen, potential means for expansion locally and distally, and a system to defend itself from the body’s immune system. The last of these poses specific challenges to successful spontaneous immune surveillance and to immunotherapy designed to kill cancer cells.
Microbial pathogens trigger both a nonspecific (innate) and a specific immune response in the human body. Initially, the immune system is nonspecifically “revved up,” triggered by shared “danger signals” associated with the perceived pathogen and its specific antigens. Then, specialized cells including dendritic cells locally and in proximate lymph nodes are primed to present the de novo antigens in a way that generates a specific and maturing immune response capable of getting rid of the pathogen. Tumors are also pathogenic and in some ways “foreign.” However, they are also similar to normal tissue and interact quite differently with the immune response in ways that enhance their likelihood of growth and survival. Tumor cells often do not send a danger signal to the immune system akin to what is generated by a staphylococcal or mycobacterial invader.
Tumor cells express specific antigens on their surface, such as viral proteins, cancer-associated mutated proteins, and overexpressed differentiated or undifferentiated antigens, including in some cases what Dr. Tuohy discusses as “retired proteins.” But some of these antigens may also be expressed in normal tissues, especially in an environment of resolving inflammation. Some signal the immune system to down-regulate what could otherwise be a vigorous self-destructive response every time there was inflammation.
The dendritic cell activation of the potential antitumor T-cell response and the antitumor T-cell response itself seem to be systematically blunted by many tumors. Reversal of this blunting represents one strategy currently used with very modest clinical success in treating advanced melanoma. Some newly generated tumor-antigen–recognizing T cells may in fact exert suppressor (or regulator) and not cytotoxic activity. Some tumors exhibit systemic immunosuppressive activity; this can be manifested not only by unchecked tumor growth, but also by an increased susceptibility to certain infections.
PITFALLS OF MESSING WITH THE IMMUNE SYSTEM
Messing with the immune system is not without pitfalls. Not all toxicities will be predicted by preclinical animal studies, and human immunity is not a mirror image of rodents’ or even other primates’ immune systems. Augmentation of an antitumor response, in part from the interplay of the complexities noted above, may lead to destruction of normal tissue elsewhere, or even to disruption of tolerance with the expression of autoimmunity. The toxicities will be different than the somewhat predictable toxicities from traditional antiproliferative chemotherapies—witness the striking systemic toxicity from interleukin 2-based therapies.
Whether Dr. Tuohy’s approach to developing a tumor vaccine will ultimately reach our formularies remains to be seen. The work is in an extremely preliminary phase. But the concept of immunotherapy for cancer remains an active area of research that is worth keeping an eye on.
Tackle the challenging shoulder dystocia emergency by practicing delivery of the posterior arm
CASE: McRobert’s maneuver fails
You are attempting an early term vaginal delivery of a 31-year-old G2P1 woman with type 2 diabetes mellitus and an estimated fetal weight of 4,100 g. The fetal head has delivered but retracted against the perineum, producing the “turtle sign.”
You call a shoulder dystocia emergency and request help. In sequence, you tell the mother to stop pushing, check for a nuchal cord, and cut a mediolateral episiotomy. Working seamlessly with your nurse, you place the patient at the edge of the bed, perform the McRobert’s maneuver, provide suprapubic pressure and apply gentle downward guidance to the fetal head. Unfortunately, with these maneuvers the baby does not deliver.
What is your next obstetric maneuver?
With alacrity, move on to an advanced maneuver. In this article, I outline your options for this advanced maneuver and describe the technique for execution. First, however, I discuss the amount of time you have to work with.
How long do you have to perform advanced maneuvers?
In managing a difficult shoulder dystocia, critical goals are to avoid permanent injury to the newborn, including brachial plexus injury, fetal asphyxia, central nervous system injury, and death. Many experts believe that the accoucheur has approximately 4 or 5 minutes to deliver the impacted fetus before the risk of these adverse outcomes rises substantially.1-3 In one study, a head-to-body delivery interval of less than 5 minutes and 5 minutes or longer were associated with rates of hypoxic ischemic encephalopathy of 0.5% and 24%, respectively.2
Stay calm, move on. Given the time pressure for management, it is important to initiate an advanced maneuver, such as rotation of the fetal body or delivery of the posterior arm, when the initial sequence of McRobert’s maneuver, suprapubic pressure, and gentle downward guidance on the fetal head do not result in delivery. Repetitively repeating these initial maneuvers will increase the risk of an adverse fetal outcome. Stay calm and quickly move on to an advanced maneuver.
Advanced maneuvers
The two advanced shoulder dystocia maneuvers that often result in a successful birth are:
- rotation of the fetal shoulders
- delivery of the posterior arm.4,5
In a prior editorial, I described in detail the Woods and Rubin rotational maneuvers.6 In this editorial, I focus on the technique for delivery of the posterior arm.
Delivery of the posterior arm
This maneuver to resolve difficult shoulder dystocia deliveries has been in the armamentarium of obstetricians since at least the mid-18th Century.7 The delivery of the posterior arm reduces the presenting fetal diameter from the larger bisacromial diameter to the smaller axilloacromial diameter. Experts estimate that this change results in a 2-cm decrease in the presenting fetal diameter, thereby facilitating delivery.8,9
In describing posterior arm delivery, it is important to clearly define the anatomy of the upper extremity. The arm is the portion of the upper extremity from the shoulder to the elbow joint. The long bone of the arm is the humerus. The forearm is the portion of the upper extremity from the elbow to the wrist. The long bones of the forearm are the radius and ulna.
Descriptions of how to deliver the posterior arm range from concise to detailed. A concise description recommends “inserting a hand in the vagina, grasping the fetal arm, and sweeping it across the chest.”9
These detailed instructions are provided by Dr. John Rodis, Chief of Obstetrics and Gynecology at St. Francis Hospital in Hartford Connecticut, in UpToDate:
Additional technical guidance. After grasping the fetal wrist and hand, pull the upper extremity against the fetal chest. Approaching the vaginal introitus, pull the wrist and hand toward the fetal ear nearest the maternal symphysis pubis.11 These maneuvers may result in a fracture to the humerus, but this complication is acceptable given the risk of fetal asphyxia and death.
Newborn injuries associated with shoulder dystocia | ||
| In a large retrospective study of 132,098 vaginal cephalic singleton births there were 2,018 cases of shoulder dystocia, representing a 1.5% rate of shoulder dystocia during vaginal birth.5A total of 101 neonatal injuries were reported in association with a shoulder dystocia, the most common being Erb’s palsy, clavicular fracture, and hypoxic ischemic encephalopathy. Some newborns incurred multiple injuries. | ||
| Type of injury | No. of newborns with injury | Rate of injury per 100 shoulder dystocias |
| Erb’s palsy | 60 | 3 |
| Clavicular fracture | 39 | 1.9 |
| Hypoxic ischemic encephalopathy | 6 | 0.3 |
| Klumpke’s palsy | 4 | 0.2 |
| Humerus fracture | 2 | 0.1 |
| Neonatal death | 0 | 0 |
Source: Hoffman, et al. Obstet Gynecol. 2011;117(6):1272–1278. | ||
Approaches to grasping the posterior arm
The posterior arm may be in one of three positions, and your approach to each position will be different.
Fetal hand near the chin. Delivery of the posterior arm is relatively easy when the fetal hand is in this position. Grasp the wrist gently and guide it out of the vagina. The fetal wrist should be pulled toward the fetal ear closest to the maternal symphysis.
Fetal hand on the abdomen. In this position, the operator can exert pressure on the antecubital fossa with the index and middle fingers, resulting in flexion of the forearm at the elbow. This will bring the fetal hand and wrist to the upper chest. The wrist then can be grasped and pronated over the fetal chest. The wrist and forearm are then pulled upward along the chest toward the fetal ear closest to the maternal symphysis.
Fetal upper extremity is extended with the hand next to the thigh. The most challenging situation is when the upper extremity of the fetus is extended along the trunk or behind the buttocks. In this situation the hand and wrist may be near the fetal thigh and very difficult to reach. In addition, the upper extremity may be tightly pinned between fetal trunk and maternal tissues, making it impossible to flex the forearm by gentle pressure on the antecubital fossa.
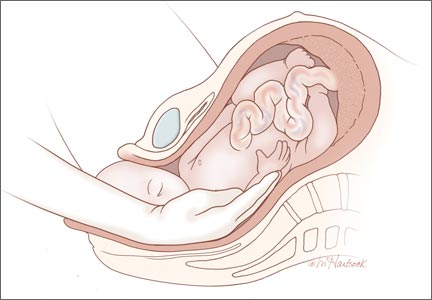
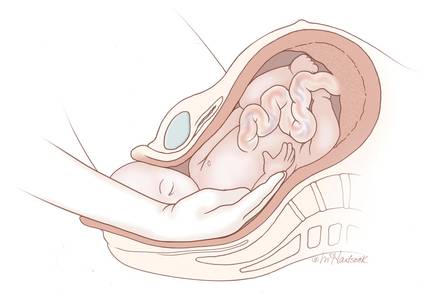
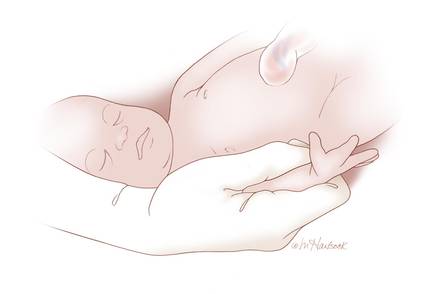
In this situation the operator’s hand must reach the fetal wrist and distal forearm, grasp these structures, and pull hard across the trunk to free the pinned upper extremity. The fetal wrist and distal forearm can be securely grasped using techniques pictured in the Figure. It can take 30 to 90 seconds for the operator to place a hand in the vagina, identify the posterior shoulder, follow the extended arm to the hand, and secure the wrist. Given the amount of time that it may take to accomplish the first steps of the maneuver, the nurse in the room should call out the time elapsed since the birth of the head at regular intervals to assist the obstetrician in pacing the speed of the intervention.
___________________________________________________________________________________________________
| 
| |
| Figure. When the fetal upper extremity is extended and the hand is near the fetal thigh the fetal upper extremity may be tightly pinned between maternal and fetal tissues. Gentle pressure in the antecubital fossa may not cause the forearm to flex toward the vaginal introitus. In this situation it may be very difficult to grasp the fetal wrist or forearm. The operator should be prepared to place their entire hand and forearm into the vagina to reach the fetal wrist (Top left). Two options for grasping the fetal wrist are with the index finger and middle finger (Top right), or by encircling the wrist with the thumb and index finger (Bottom left). For many obstetricians, the index and middle fingers extend much further from their wrist than the thumb. Consequently, when the fetal wrist and hand are against the fetal thigh it may be easier to reach the fetal wrist with the operator’s index and middle finger. However, many obstetricians find that the thumb and index finger provide a more secure grip of the fetal wrist. |
______________________________________________________________________________________________________
When the posterior arm is fully extended and pinned between fetal trunk and maternal tissues it can be very difficult to reach the fetal wrist. To help successfully complete the maneuver, the obstetrician should visualize placing his or her hand and entire forearm up to the elbow in the vagina to reach the fetal wrist. It may not be necessary to insert the entire forearm in the vagina, but the operator should visualize this step so he or she is prepared for the possibility.Surprisingly, the hollow of the sacrum often provides sufficient space for inserting the hand and entire forearm of the operator. In this process the operator’s hand and forearm may be strongly compressed by maternal and fetal tissues, cutting off circulation to the upper extremity. The operator’s upper extremity may quickly become numb, resulting in a reduction in tactile sensation and strength.
If the posterior arm is positioned behind the back of the fetus, maneuvers similar to those described above can be used to grasp the wrist and pull the arm to the anterior side of the fetal trunk, followed by delivery of the posterior arm.
Practice, practice, and practice some more
Obstetric emergencies create a rush of adrenaline and great stress for the obstetrician. This may adversely impact motor performance, decision-making, and communication skills.12 Low- and high-fidelity simulation exercises permit the obstetrics team to practice the sequence of maneuvers necessary to successfully resolve a shoulder dystocia, thereby reducing stress and improving performance when the emergency actually occurs.13 Simulating obstetric emergencies and visualizing the steps necessary to resolve an emergency are good approaches to prepare obstetricians for the most challenging emergencies. For the difficult to resolve shoulder dystocia, my recommendation is: “Deliver the posterior arm.”
Use this checklist to document a shoulder dystocia event
-Safety Checklists
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
1. Allen RH, Rosenbaum TC, Ghidini A, Poggi SH, Spong CY. Correlating head-to-body delivery intervals with neonatal depression in vaginal births that result in permanent brachial plexus injury. Am J Obstet Gynecol. 2002;187(4):839–842.
2. Leung TY, Stuart O, Sahota DS, Suen SS, Lau TK, Lao TT. Head-to-body delivery interval and risk of fetal acidosis and hypoxic ischaemic encephalopathy in shoulder dystocia: a retrospective review. BJOG. 2011;118(4):474–479.
3. Lerner H, Durlacher K, Smith S, Hamilton E. Relationship between head-to-body delivery interval in shoulder dystocia and neonatal depression. Obstet Gynecol. 2011;118(2 pt 1):318–322.
4. Leung TY, Stuart O, Suen SS, Sahota DS, Lau TK, Lao TT. Comparison of perinatal outcomes of shoulder dystocia alleviated by different type and sequence of manoeuvres: a retrospective review. BJOG. 2011;118(8):985–990.
5. Hoffman MK, Bailit KL, Branch DW, et al. A comparison of obstetric maneuvers for the acute management of should dystocia. Obstet Gynecol. 2011;117(6):1272–1278.
6. Barbieri RL. You are the second responder to a shoulder dystocia emergency. What do you do first? OBG Manag. 2013;25(????):10, 12, 15.
7. Beer E. History of extraction of the posterior arm to resolve shoulder dystocia. Obstet Gynecol Surv. 2006;61(3):149–151.
8. Kung J, Swan AV, Arulkumaran S. Delivery of the posterior arm reduces shoulder dimensions in shoulder dystocia. Int J Gynaecol Obstet. 2006;93(3):233–237.
9. Poggi SH, Spong CY, Allen RH. Prioritizing posterior arm delivery during severe shoulder dystocia. Obstet Gynecol. 2003;101(5 pt 2):1068–1072.
10. Rodis JF. Shoulder dystocia, intrapartum diagnosis, management and outcome. UpToDate, Waltham MA.
11. Mazzanti GA. Delivery of the anterior shoulder; a neglected art. Obstet Gynecol. 1959;13(5):603–607.
12. Wetzel CM, Kneebone RL, Woloshynowych M, et al. The effects of stress on surgical performance. Am J Surg. 2006;191(1):5–10.
13. Grobman WA, Miller D, Burke C, Hornbogen A, Tam K, Costello R. Outcomes associated with introduction of a shoulder dystocia protocol. Am J Obstet Gynecol. 2011;205(6):513–517.
CASE: McRobert’s maneuver fails
You are attempting an early term vaginal delivery of a 31-year-old G2P1 woman with type 2 diabetes mellitus and an estimated fetal weight of 4,100 g. The fetal head has delivered but retracted against the perineum, producing the “turtle sign.”
You call a shoulder dystocia emergency and request help. In sequence, you tell the mother to stop pushing, check for a nuchal cord, and cut a mediolateral episiotomy. Working seamlessly with your nurse, you place the patient at the edge of the bed, perform the McRobert’s maneuver, provide suprapubic pressure and apply gentle downward guidance to the fetal head. Unfortunately, with these maneuvers the baby does not deliver.
What is your next obstetric maneuver?
With alacrity, move on to an advanced maneuver. In this article, I outline your options for this advanced maneuver and describe the technique for execution. First, however, I discuss the amount of time you have to work with.
How long do you have to perform advanced maneuvers?
In managing a difficult shoulder dystocia, critical goals are to avoid permanent injury to the newborn, including brachial plexus injury, fetal asphyxia, central nervous system injury, and death. Many experts believe that the accoucheur has approximately 4 or 5 minutes to deliver the impacted fetus before the risk of these adverse outcomes rises substantially.1-3 In one study, a head-to-body delivery interval of less than 5 minutes and 5 minutes or longer were associated with rates of hypoxic ischemic encephalopathy of 0.5% and 24%, respectively.2
Stay calm, move on. Given the time pressure for management, it is important to initiate an advanced maneuver, such as rotation of the fetal body or delivery of the posterior arm, when the initial sequence of McRobert’s maneuver, suprapubic pressure, and gentle downward guidance on the fetal head do not result in delivery. Repetitively repeating these initial maneuvers will increase the risk of an adverse fetal outcome. Stay calm and quickly move on to an advanced maneuver.
Advanced maneuvers
The two advanced shoulder dystocia maneuvers that often result in a successful birth are:
- rotation of the fetal shoulders
- delivery of the posterior arm.4,5
In a prior editorial, I described in detail the Woods and Rubin rotational maneuvers.6 In this editorial, I focus on the technique for delivery of the posterior arm.
Delivery of the posterior arm
This maneuver to resolve difficult shoulder dystocia deliveries has been in the armamentarium of obstetricians since at least the mid-18th Century.7 The delivery of the posterior arm reduces the presenting fetal diameter from the larger bisacromial diameter to the smaller axilloacromial diameter. Experts estimate that this change results in a 2-cm decrease in the presenting fetal diameter, thereby facilitating delivery.8,9
In describing posterior arm delivery, it is important to clearly define the anatomy of the upper extremity. The arm is the portion of the upper extremity from the shoulder to the elbow joint. The long bone of the arm is the humerus. The forearm is the portion of the upper extremity from the elbow to the wrist. The long bones of the forearm are the radius and ulna.
Descriptions of how to deliver the posterior arm range from concise to detailed. A concise description recommends “inserting a hand in the vagina, grasping the fetal arm, and sweeping it across the chest.”9
These detailed instructions are provided by Dr. John Rodis, Chief of Obstetrics and Gynecology at St. Francis Hospital in Hartford Connecticut, in UpToDate:
Additional technical guidance. After grasping the fetal wrist and hand, pull the upper extremity against the fetal chest. Approaching the vaginal introitus, pull the wrist and hand toward the fetal ear nearest the maternal symphysis pubis.11 These maneuvers may result in a fracture to the humerus, but this complication is acceptable given the risk of fetal asphyxia and death.
Newborn injuries associated with shoulder dystocia | ||
| In a large retrospective study of 132,098 vaginal cephalic singleton births there were 2,018 cases of shoulder dystocia, representing a 1.5% rate of shoulder dystocia during vaginal birth.5A total of 101 neonatal injuries were reported in association with a shoulder dystocia, the most common being Erb’s palsy, clavicular fracture, and hypoxic ischemic encephalopathy. Some newborns incurred multiple injuries. | ||
| Type of injury | No. of newborns with injury | Rate of injury per 100 shoulder dystocias |
| Erb’s palsy | 60 | 3 |
| Clavicular fracture | 39 | 1.9 |
| Hypoxic ischemic encephalopathy | 6 | 0.3 |
| Klumpke’s palsy | 4 | 0.2 |
| Humerus fracture | 2 | 0.1 |
| Neonatal death | 0 | 0 |
Source: Hoffman, et al. Obstet Gynecol. 2011;117(6):1272–1278. | ||
Approaches to grasping the posterior arm
The posterior arm may be in one of three positions, and your approach to each position will be different.
Fetal hand near the chin. Delivery of the posterior arm is relatively easy when the fetal hand is in this position. Grasp the wrist gently and guide it out of the vagina. The fetal wrist should be pulled toward the fetal ear closest to the maternal symphysis.
Fetal hand on the abdomen. In this position, the operator can exert pressure on the antecubital fossa with the index and middle fingers, resulting in flexion of the forearm at the elbow. This will bring the fetal hand and wrist to the upper chest. The wrist then can be grasped and pronated over the fetal chest. The wrist and forearm are then pulled upward along the chest toward the fetal ear closest to the maternal symphysis.
Fetal upper extremity is extended with the hand next to the thigh. The most challenging situation is when the upper extremity of the fetus is extended along the trunk or behind the buttocks. In this situation the hand and wrist may be near the fetal thigh and very difficult to reach. In addition, the upper extremity may be tightly pinned between fetal trunk and maternal tissues, making it impossible to flex the forearm by gentle pressure on the antecubital fossa.
In this situation the operator’s hand must reach the fetal wrist and distal forearm, grasp these structures, and pull hard across the trunk to free the pinned upper extremity. The fetal wrist and distal forearm can be securely grasped using techniques pictured in the Figure. It can take 30 to 90 seconds for the operator to place a hand in the vagina, identify the posterior shoulder, follow the extended arm to the hand, and secure the wrist. Given the amount of time that it may take to accomplish the first steps of the maneuver, the nurse in the room should call out the time elapsed since the birth of the head at regular intervals to assist the obstetrician in pacing the speed of the intervention.
___________________________________________________________________________________________________

| 
| |

| Figure. When the fetal upper extremity is extended and the hand is near the fetal thigh the fetal upper extremity may be tightly pinned between maternal and fetal tissues. Gentle pressure in the antecubital fossa may not cause the forearm to flex toward the vaginal introitus. In this situation it may be very difficult to grasp the fetal wrist or forearm. The operator should be prepared to place their entire hand and forearm into the vagina to reach the fetal wrist (Top left). Two options for grasping the fetal wrist are with the index finger and middle finger (Top right), or by encircling the wrist with the thumb and index finger (Bottom left). For many obstetricians, the index and middle fingers extend much further from their wrist than the thumb. Consequently, when the fetal wrist and hand are against the fetal thigh it may be easier to reach the fetal wrist with the operator’s index and middle finger. However, many obstetricians find that the thumb and index finger provide a more secure grip of the fetal wrist. |
______________________________________________________________________________________________________
When the posterior arm is fully extended and pinned between fetal trunk and maternal tissues it can be very difficult to reach the fetal wrist. To help successfully complete the maneuver, the obstetrician should visualize placing his or her hand and entire forearm up to the elbow in the vagina to reach the fetal wrist. It may not be necessary to insert the entire forearm in the vagina, but the operator should visualize this step so he or she is prepared for the possibility.Surprisingly, the hollow of the sacrum often provides sufficient space for inserting the hand and entire forearm of the operator. In this process the operator’s hand and forearm may be strongly compressed by maternal and fetal tissues, cutting off circulation to the upper extremity. The operator’s upper extremity may quickly become numb, resulting in a reduction in tactile sensation and strength.
If the posterior arm is positioned behind the back of the fetus, maneuvers similar to those described above can be used to grasp the wrist and pull the arm to the anterior side of the fetal trunk, followed by delivery of the posterior arm.
Practice, practice, and practice some more
Obstetric emergencies create a rush of adrenaline and great stress for the obstetrician. This may adversely impact motor performance, decision-making, and communication skills.12 Low- and high-fidelity simulation exercises permit the obstetrics team to practice the sequence of maneuvers necessary to successfully resolve a shoulder dystocia, thereby reducing stress and improving performance when the emergency actually occurs.13 Simulating obstetric emergencies and visualizing the steps necessary to resolve an emergency are good approaches to prepare obstetricians for the most challenging emergencies. For the difficult to resolve shoulder dystocia, my recommendation is: “Deliver the posterior arm.”
Use this checklist to document a shoulder dystocia event
-Safety Checklists
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
CASE: McRobert’s maneuver fails
You are attempting an early term vaginal delivery of a 31-year-old G2P1 woman with type 2 diabetes mellitus and an estimated fetal weight of 4,100 g. The fetal head has delivered but retracted against the perineum, producing the “turtle sign.”
You call a shoulder dystocia emergency and request help. In sequence, you tell the mother to stop pushing, check for a nuchal cord, and cut a mediolateral episiotomy. Working seamlessly with your nurse, you place the patient at the edge of the bed, perform the McRobert’s maneuver, provide suprapubic pressure and apply gentle downward guidance to the fetal head. Unfortunately, with these maneuvers the baby does not deliver.
What is your next obstetric maneuver?
With alacrity, move on to an advanced maneuver. In this article, I outline your options for this advanced maneuver and describe the technique for execution. First, however, I discuss the amount of time you have to work with.
How long do you have to perform advanced maneuvers?
In managing a difficult shoulder dystocia, critical goals are to avoid permanent injury to the newborn, including brachial plexus injury, fetal asphyxia, central nervous system injury, and death. Many experts believe that the accoucheur has approximately 4 or 5 minutes to deliver the impacted fetus before the risk of these adverse outcomes rises substantially.1-3 In one study, a head-to-body delivery interval of less than 5 minutes and 5 minutes or longer were associated with rates of hypoxic ischemic encephalopathy of 0.5% and 24%, respectively.2
Stay calm, move on. Given the time pressure for management, it is important to initiate an advanced maneuver, such as rotation of the fetal body or delivery of the posterior arm, when the initial sequence of McRobert’s maneuver, suprapubic pressure, and gentle downward guidance on the fetal head do not result in delivery. Repetitively repeating these initial maneuvers will increase the risk of an adverse fetal outcome. Stay calm and quickly move on to an advanced maneuver.
Advanced maneuvers
The two advanced shoulder dystocia maneuvers that often result in a successful birth are:
- rotation of the fetal shoulders
- delivery of the posterior arm.4,5
In a prior editorial, I described in detail the Woods and Rubin rotational maneuvers.6 In this editorial, I focus on the technique for delivery of the posterior arm.
Delivery of the posterior arm
This maneuver to resolve difficult shoulder dystocia deliveries has been in the armamentarium of obstetricians since at least the mid-18th Century.7 The delivery of the posterior arm reduces the presenting fetal diameter from the larger bisacromial diameter to the smaller axilloacromial diameter. Experts estimate that this change results in a 2-cm decrease in the presenting fetal diameter, thereby facilitating delivery.8,9
In describing posterior arm delivery, it is important to clearly define the anatomy of the upper extremity. The arm is the portion of the upper extremity from the shoulder to the elbow joint. The long bone of the arm is the humerus. The forearm is the portion of the upper extremity from the elbow to the wrist. The long bones of the forearm are the radius and ulna.
Descriptions of how to deliver the posterior arm range from concise to detailed. A concise description recommends “inserting a hand in the vagina, grasping the fetal arm, and sweeping it across the chest.”9
These detailed instructions are provided by Dr. John Rodis, Chief of Obstetrics and Gynecology at St. Francis Hospital in Hartford Connecticut, in UpToDate:
Additional technical guidance. After grasping the fetal wrist and hand, pull the upper extremity against the fetal chest. Approaching the vaginal introitus, pull the wrist and hand toward the fetal ear nearest the maternal symphysis pubis.11 These maneuvers may result in a fracture to the humerus, but this complication is acceptable given the risk of fetal asphyxia and death.
Newborn injuries associated with shoulder dystocia | ||
| In a large retrospective study of 132,098 vaginal cephalic singleton births there were 2,018 cases of shoulder dystocia, representing a 1.5% rate of shoulder dystocia during vaginal birth.5A total of 101 neonatal injuries were reported in association with a shoulder dystocia, the most common being Erb’s palsy, clavicular fracture, and hypoxic ischemic encephalopathy. Some newborns incurred multiple injuries. | ||
| Type of injury | No. of newborns with injury | Rate of injury per 100 shoulder dystocias |
| Erb’s palsy | 60 | 3 |
| Clavicular fracture | 39 | 1.9 |
| Hypoxic ischemic encephalopathy | 6 | 0.3 |
| Klumpke’s palsy | 4 | 0.2 |
| Humerus fracture | 2 | 0.1 |
| Neonatal death | 0 | 0 |
Source: Hoffman, et al. Obstet Gynecol. 2011;117(6):1272–1278. | ||
Approaches to grasping the posterior arm
The posterior arm may be in one of three positions, and your approach to each position will be different.
Fetal hand near the chin. Delivery of the posterior arm is relatively easy when the fetal hand is in this position. Grasp the wrist gently and guide it out of the vagina. The fetal wrist should be pulled toward the fetal ear closest to the maternal symphysis.
Fetal hand on the abdomen. In this position, the operator can exert pressure on the antecubital fossa with the index and middle fingers, resulting in flexion of the forearm at the elbow. This will bring the fetal hand and wrist to the upper chest. The wrist then can be grasped and pronated over the fetal chest. The wrist and forearm are then pulled upward along the chest toward the fetal ear closest to the maternal symphysis.
Fetal upper extremity is extended with the hand next to the thigh. The most challenging situation is when the upper extremity of the fetus is extended along the trunk or behind the buttocks. In this situation the hand and wrist may be near the fetal thigh and very difficult to reach. In addition, the upper extremity may be tightly pinned between fetal trunk and maternal tissues, making it impossible to flex the forearm by gentle pressure on the antecubital fossa.
In this situation the operator’s hand must reach the fetal wrist and distal forearm, grasp these structures, and pull hard across the trunk to free the pinned upper extremity. The fetal wrist and distal forearm can be securely grasped using techniques pictured in the Figure. It can take 30 to 90 seconds for the operator to place a hand in the vagina, identify the posterior shoulder, follow the extended arm to the hand, and secure the wrist. Given the amount of time that it may take to accomplish the first steps of the maneuver, the nurse in the room should call out the time elapsed since the birth of the head at regular intervals to assist the obstetrician in pacing the speed of the intervention.
___________________________________________________________________________________________________

| 
| |

| Figure. When the fetal upper extremity is extended and the hand is near the fetal thigh the fetal upper extremity may be tightly pinned between maternal and fetal tissues. Gentle pressure in the antecubital fossa may not cause the forearm to flex toward the vaginal introitus. In this situation it may be very difficult to grasp the fetal wrist or forearm. The operator should be prepared to place their entire hand and forearm into the vagina to reach the fetal wrist (Top left). Two options for grasping the fetal wrist are with the index finger and middle finger (Top right), or by encircling the wrist with the thumb and index finger (Bottom left). For many obstetricians, the index and middle fingers extend much further from their wrist than the thumb. Consequently, when the fetal wrist and hand are against the fetal thigh it may be easier to reach the fetal wrist with the operator’s index and middle finger. However, many obstetricians find that the thumb and index finger provide a more secure grip of the fetal wrist. |
______________________________________________________________________________________________________
When the posterior arm is fully extended and pinned between fetal trunk and maternal tissues it can be very difficult to reach the fetal wrist. To help successfully complete the maneuver, the obstetrician should visualize placing his or her hand and entire forearm up to the elbow in the vagina to reach the fetal wrist. It may not be necessary to insert the entire forearm in the vagina, but the operator should visualize this step so he or she is prepared for the possibility.Surprisingly, the hollow of the sacrum often provides sufficient space for inserting the hand and entire forearm of the operator. In this process the operator’s hand and forearm may be strongly compressed by maternal and fetal tissues, cutting off circulation to the upper extremity. The operator’s upper extremity may quickly become numb, resulting in a reduction in tactile sensation and strength.
If the posterior arm is positioned behind the back of the fetus, maneuvers similar to those described above can be used to grasp the wrist and pull the arm to the anterior side of the fetal trunk, followed by delivery of the posterior arm.
Practice, practice, and practice some more
Obstetric emergencies create a rush of adrenaline and great stress for the obstetrician. This may adversely impact motor performance, decision-making, and communication skills.12 Low- and high-fidelity simulation exercises permit the obstetrics team to practice the sequence of maneuvers necessary to successfully resolve a shoulder dystocia, thereby reducing stress and improving performance when the emergency actually occurs.13 Simulating obstetric emergencies and visualizing the steps necessary to resolve an emergency are good approaches to prepare obstetricians for the most challenging emergencies. For the difficult to resolve shoulder dystocia, my recommendation is: “Deliver the posterior arm.”
Use this checklist to document a shoulder dystocia event
-Safety Checklists
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
1. Allen RH, Rosenbaum TC, Ghidini A, Poggi SH, Spong CY. Correlating head-to-body delivery intervals with neonatal depression in vaginal births that result in permanent brachial plexus injury. Am J Obstet Gynecol. 2002;187(4):839–842.
2. Leung TY, Stuart O, Sahota DS, Suen SS, Lau TK, Lao TT. Head-to-body delivery interval and risk of fetal acidosis and hypoxic ischaemic encephalopathy in shoulder dystocia: a retrospective review. BJOG. 2011;118(4):474–479.
3. Lerner H, Durlacher K, Smith S, Hamilton E. Relationship between head-to-body delivery interval in shoulder dystocia and neonatal depression. Obstet Gynecol. 2011;118(2 pt 1):318–322.
4. Leung TY, Stuart O, Suen SS, Sahota DS, Lau TK, Lao TT. Comparison of perinatal outcomes of shoulder dystocia alleviated by different type and sequence of manoeuvres: a retrospective review. BJOG. 2011;118(8):985–990.
5. Hoffman MK, Bailit KL, Branch DW, et al. A comparison of obstetric maneuvers for the acute management of should dystocia. Obstet Gynecol. 2011;117(6):1272–1278.
6. Barbieri RL. You are the second responder to a shoulder dystocia emergency. What do you do first? OBG Manag. 2013;25(????):10, 12, 15.
7. Beer E. History of extraction of the posterior arm to resolve shoulder dystocia. Obstet Gynecol Surv. 2006;61(3):149–151.
8. Kung J, Swan AV, Arulkumaran S. Delivery of the posterior arm reduces shoulder dimensions in shoulder dystocia. Int J Gynaecol Obstet. 2006;93(3):233–237.
9. Poggi SH, Spong CY, Allen RH. Prioritizing posterior arm delivery during severe shoulder dystocia. Obstet Gynecol. 2003;101(5 pt 2):1068–1072.
10. Rodis JF. Shoulder dystocia, intrapartum diagnosis, management and outcome. UpToDate, Waltham MA.
11. Mazzanti GA. Delivery of the anterior shoulder; a neglected art. Obstet Gynecol. 1959;13(5):603–607.
12. Wetzel CM, Kneebone RL, Woloshynowych M, et al. The effects of stress on surgical performance. Am J Surg. 2006;191(1):5–10.
13. Grobman WA, Miller D, Burke C, Hornbogen A, Tam K, Costello R. Outcomes associated with introduction of a shoulder dystocia protocol. Am J Obstet Gynecol. 2011;205(6):513–517.
1. Allen RH, Rosenbaum TC, Ghidini A, Poggi SH, Spong CY. Correlating head-to-body delivery intervals with neonatal depression in vaginal births that result in permanent brachial plexus injury. Am J Obstet Gynecol. 2002;187(4):839–842.
2. Leung TY, Stuart O, Sahota DS, Suen SS, Lau TK, Lao TT. Head-to-body delivery interval and risk of fetal acidosis and hypoxic ischaemic encephalopathy in shoulder dystocia: a retrospective review. BJOG. 2011;118(4):474–479.
3. Lerner H, Durlacher K, Smith S, Hamilton E. Relationship between head-to-body delivery interval in shoulder dystocia and neonatal depression. Obstet Gynecol. 2011;118(2 pt 1):318–322.
4. Leung TY, Stuart O, Suen SS, Sahota DS, Lau TK, Lao TT. Comparison of perinatal outcomes of shoulder dystocia alleviated by different type and sequence of manoeuvres: a retrospective review. BJOG. 2011;118(8):985–990.
5. Hoffman MK, Bailit KL, Branch DW, et al. A comparison of obstetric maneuvers for the acute management of should dystocia. Obstet Gynecol. 2011;117(6):1272–1278.
6. Barbieri RL. You are the second responder to a shoulder dystocia emergency. What do you do first? OBG Manag. 2013;25(????):10, 12, 15.
7. Beer E. History of extraction of the posterior arm to resolve shoulder dystocia. Obstet Gynecol Surv. 2006;61(3):149–151.
8. Kung J, Swan AV, Arulkumaran S. Delivery of the posterior arm reduces shoulder dimensions in shoulder dystocia. Int J Gynaecol Obstet. 2006;93(3):233–237.
9. Poggi SH, Spong CY, Allen RH. Prioritizing posterior arm delivery during severe shoulder dystocia. Obstet Gynecol. 2003;101(5 pt 2):1068–1072.
10. Rodis JF. Shoulder dystocia, intrapartum diagnosis, management and outcome. UpToDate, Waltham MA.
11. Mazzanti GA. Delivery of the anterior shoulder; a neglected art. Obstet Gynecol. 1959;13(5):603–607.
12. Wetzel CM, Kneebone RL, Woloshynowych M, et al. The effects of stress on surgical performance. Am J Surg. 2006;191(1):5–10.
13. Grobman WA, Miller D, Burke C, Hornbogen A, Tam K, Costello R. Outcomes associated with introduction of a shoulder dystocia protocol. Am J Obstet Gynecol. 2011;205(6):513–517.
The art and science of cancer care
Summer is winding down as we go to press with this month’s issue, and while we might well reflect a little sadly on its departure, we can also look forward to the fall season with its promise of renewal and adventure. As I settled back in to my familiar work routine after the Labor Day weekend, I was reminded of how, despite the remarkable clinical advances in oncology, we are still caregivers, involved in our patients’ everyday lives and that we can never forget our humanity. The advent of high-tech personalized medicine or precision oncology, as I prefer to call it, has given oncologists a remarkable cache of treatment options for their patients and the hope that more – and better – therapies are to come. Next-generation diagnostics are helping us identify the cellular targets we need to take aim at to kill the tumor and globally, research is yielding more and more therapeutics to subdue those targets and hence the tumor.
Click on the PDF icon at the top of this introduction to read the full article.
Summer is winding down as we go to press with this month’s issue, and while we might well reflect a little sadly on its departure, we can also look forward to the fall season with its promise of renewal and adventure. As I settled back in to my familiar work routine after the Labor Day weekend, I was reminded of how, despite the remarkable clinical advances in oncology, we are still caregivers, involved in our patients’ everyday lives and that we can never forget our humanity. The advent of high-tech personalized medicine or precision oncology, as I prefer to call it, has given oncologists a remarkable cache of treatment options for their patients and the hope that more – and better – therapies are to come. Next-generation diagnostics are helping us identify the cellular targets we need to take aim at to kill the tumor and globally, research is yielding more and more therapeutics to subdue those targets and hence the tumor.
Click on the PDF icon at the top of this introduction to read the full article.
Summer is winding down as we go to press with this month’s issue, and while we might well reflect a little sadly on its departure, we can also look forward to the fall season with its promise of renewal and adventure. As I settled back in to my familiar work routine after the Labor Day weekend, I was reminded of how, despite the remarkable clinical advances in oncology, we are still caregivers, involved in our patients’ everyday lives and that we can never forget our humanity. The advent of high-tech personalized medicine or precision oncology, as I prefer to call it, has given oncologists a remarkable cache of treatment options for their patients and the hope that more – and better – therapies are to come. Next-generation diagnostics are helping us identify the cellular targets we need to take aim at to kill the tumor and globally, research is yielding more and more therapeutics to subdue those targets and hence the tumor.
Click on the PDF icon at the top of this introduction to read the full article.
Clozapine is a vastly underutilized, unique agent with multiple applications
Since clozapine was launched in 1989, miraculous improvements and “awakenings” have been reported in many patients afflicted with severe schizophrenia and considered hopelessly refractory to antipsychotic pharmacotherapy. Not only do severely disabled patients regain their sanity and return to normal functioning, but the joy that their family and treating psychiatrist experience is priceless.
That’s why I am perplexed by how infrequently clozapine is used in the United States (in about 5% of patients)—even though approximately 25% of patients who have schizophrenia are either treatment-resistant or have refractory hallucinations or delusions.
Consider Bethany’s case. She was one of my young patients, who, after taking clozapine, recovered fully and resumed a productive life, after years of homelessness during which she was controlled by auditory hallucinations.
Bethany’s story began well…
Bethany grew up in a loving home, smart and talented, an “A” student in high school and talented violinist. She received a scholarship to a prestigious private university at 16 and left her parent’s home in Ohio to major in molecular biology. Her goal was to attend medical school. She excelled during her first 3 years of college, and even published 2 papers in top-tier science journals.
In her senior year, after returning from a trip to Africa, Bethany began to change. She neglected her studies and focused on raising money for HIV clinics in Africa. She began getting F’s instead of A’s, lost her scholarship and her residence hall room, and had to drop out of college. Soon, she began hearing voices commanding her every action.
..but took a really bad turn
Bethany became homeless for the next 4.5 years. She ate discarded food from garbage cans, had no change of clothes, and slept on a concrete slab behind a downtown church in a major city in California. Her parents lost track of her, although her mother, a retired nurse, frantically and relentlessly tried to find out what happened to her only daughter during that time.
Eventually, Bethany was arrested when she was found screaming back at the voices, at midnight in a residential area of the city. She was hospitalized on a psychiatric ward and given antipsychotics, but with only modest improvement.
Her parents were contacted; immediately, they flew to California to see her. The treating psychiatrist told them that their daughter had schizophrenia, and that they should lower their expectations because she would be totally disabled for the rest of her life. They brought Bethany back to Ohio where, after a tumultuous year of failed trials of several antipsychotics to suppress the auditory hallucinations, we gave her clozapine.
Gradually, Bethany improved, but she still could not read a book or magazine (which I urged her to do) without the voices intensifying and preventing her from reading.
Bethany recovers
After 6 to 8 months on clozapine, however, Bethany’s auditory hallucinations faded away. With my encouragement, she enrolled at the University of Cincinnati and took 1 course at a time. She began to get A’s again—in advanced courses, such as genetics, physics, and molecular biology. She completed her degree requirements and graduated with honors, with a Bachelor of Science degree in molecular biology. She also served as a marshal in the commencement ceremony procession.
Over the next year, with strong encouragement, Bethany wrote a book about her remarkable recovery from refractory psychosis.1 In addition, her mother wrote a deeply emotional book that described the gut-wrenching ordeal that she and her husband went through during the years that Bethany disappeared.2 I urge you to read these inspiring books (Figure) about the remarkable recovery from refractory psychosis and the heavy family burden of schizophrenia.
Back to clozapine
Although the package insert for clozapine contains 5 black-box warnings (for agranulocytosis, seizures, myocarditis, respiratory effects, and increased mortality in geriatric patients with psychosis associated with dementia), the drug is a useful last-resort medication for several approved indications and off-label uses. In addition to the official, evidence-based indication for treatment-resistant and refractory schizophrenia,3 clozapine is FDA-approved for suicidality in schizophrenia.4 Clinically reported, but unapproved, uses are listed in the Table.5-13
A little-known advantage of clozapine is its salutary effect on mortality. In a Finnish study of 66,881 persons who had schizophrenia,14 those taking clozapine had, overall, lower mortality during the treatment period than those taking any of the 6 most commonly used antipsychotic drugs.
No doubt, clozapine is associated with serious side effects15—but so is chemotherapy for cancer, and oncologists do not hesitate to use it to save their patients from physical death. Severe schizophrenia is like a cancer of the mind, and clozapine is its chemotherapy.
Fortunately for Bethany, she had almost no physical adverse effects from clozapine except for intense sedation, which was mitigated with modafinil.
We should use clozapine more than we do
Clozapine has the potential to have a healing effect for many patients whose schizophrenia is resistant to treatment. Most such patients, however, never receive a trial of the drug. Furthermore, few practitioners use clozapine for schizophrenia patients with suicidal tendencies, despite the high rate of suicide completion in schizophrenia.16
Clozapine remains, regrettably, an underutilized agent in psychiatry. Until other breakthrough drugs are discovered, its use ought to be double or triple what it is now because there are many people like Bethany who are not being given a chance to recover from their illness.
1. Yeiser B. Mind estranged. My journey from schizophrenia and homelessness to recovery. North Charleston, SC: CreateSpace Independent Publishing Platform; 2014.
2. Yeiser KS. Flight from reason: a mother’s story of schizophrenia, recovery and hope. North Charleston, SC: CreateSpace Independent Publishing Platform; 2014.
3. Kane J, Honigfeld G, Singer J, et al. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch Gen Psychiatry. 1988;45(9):789-796.
4. Meltzer HY, Alphs L, Green AI, et al. Clozapine treatment for suicidality in schizophrenia: International Suicide Prevention Trial (InterSePT) [Erratum in: Arch Gen Psychiatry. 2003;60(7):735.] Arch Gen Psychiatry. 2003;60(1):82-91.
5. Frogley C, Taylor D, Dickens G, et al. A systematic review of the evidence of clozapine’s anti-aggressive effects. Int J Neuropsychopharmacol. 2012;15(9):1351-1371.
6. Margetié B, Aukst-Margetié B, Zarkovié-Palijan T. Successful treatment of polydipsia, water intoxication, and delusional jealousy in an alcohol dependent patient with clozapine. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30(7):1347-1349.
7. Cupina D, Boulton M. Secondary delusional parasitosis treated successfully with a combination of clozapine and citalopram. Psychosomatics. 2012;53(3):301-302.
8. Connolly BD, Lang AE. Pharmacolgoic treatment of Parkinson disease: a review. JAMA. 2014;311(16):1670-1683.
9. Hazari N, Kate N, Grover S, et al. Clozapine and tardive movement disorders: a review. Asian J Psychiatry. 2013;6(6):439-451.
10. Zarzar T, McEvoy J. Clozapine for self-injurious behavior in individuals with borderline personality disorder. Ther Adv Psychopharmacol. 2013;3(5):272-274.
11. Vohra AK. Treatment of severe borderline personality disorder with clozapine. Indian J Psychiatry. 2010;52(3):267-269.
12. Ifteni P, Correll CU, Nielse J, et al. Rapid clozapine titration in treatment-refractory bipolar disorder. J Affect Disord. 2014;166:168-172.
13. Rogoz Z. Combined treatment with atypical antipsychotics and antidepressants in treatment-resistant depression: preclinical and clinical efficacy. Pharmacol Rep. 2013;65(6)1535-1544.
14. Tiihonen J, Lönnqvist J, Wahlbeck K, et al. 11-year follow-up of mortality in patients with schizophrenia: a population-based cohort study (FIN11 study). Lancet. 2009;374(9690):620-627.
15. Raja M, Raja S. Clozapine safety, 40 years later [published online April 28, 2014]. Curr Drug Saf. doi: 10.2174/1574886309666140428115040.
16. Siris SG. Suicide and schizophrenia. J Psychopharmacol. 2001;15(2):127-135.
Since clozapine was launched in 1989, miraculous improvements and “awakenings” have been reported in many patients afflicted with severe schizophrenia and considered hopelessly refractory to antipsychotic pharmacotherapy. Not only do severely disabled patients regain their sanity and return to normal functioning, but the joy that their family and treating psychiatrist experience is priceless.
That’s why I am perplexed by how infrequently clozapine is used in the United States (in about 5% of patients)—even though approximately 25% of patients who have schizophrenia are either treatment-resistant or have refractory hallucinations or delusions.
Consider Bethany’s case. She was one of my young patients, who, after taking clozapine, recovered fully and resumed a productive life, after years of homelessness during which she was controlled by auditory hallucinations.
Bethany’s story began well…
Bethany grew up in a loving home, smart and talented, an “A” student in high school and talented violinist. She received a scholarship to a prestigious private university at 16 and left her parent’s home in Ohio to major in molecular biology. Her goal was to attend medical school. She excelled during her first 3 years of college, and even published 2 papers in top-tier science journals.
In her senior year, after returning from a trip to Africa, Bethany began to change. She neglected her studies and focused on raising money for HIV clinics in Africa. She began getting F’s instead of A’s, lost her scholarship and her residence hall room, and had to drop out of college. Soon, she began hearing voices commanding her every action.
..but took a really bad turn
Bethany became homeless for the next 4.5 years. She ate discarded food from garbage cans, had no change of clothes, and slept on a concrete slab behind a downtown church in a major city in California. Her parents lost track of her, although her mother, a retired nurse, frantically and relentlessly tried to find out what happened to her only daughter during that time.
Eventually, Bethany was arrested when she was found screaming back at the voices, at midnight in a residential area of the city. She was hospitalized on a psychiatric ward and given antipsychotics, but with only modest improvement.
Her parents were contacted; immediately, they flew to California to see her. The treating psychiatrist told them that their daughter had schizophrenia, and that they should lower their expectations because she would be totally disabled for the rest of her life. They brought Bethany back to Ohio where, after a tumultuous year of failed trials of several antipsychotics to suppress the auditory hallucinations, we gave her clozapine.
Gradually, Bethany improved, but she still could not read a book or magazine (which I urged her to do) without the voices intensifying and preventing her from reading.
Bethany recovers
After 6 to 8 months on clozapine, however, Bethany’s auditory hallucinations faded away. With my encouragement, she enrolled at the University of Cincinnati and took 1 course at a time. She began to get A’s again—in advanced courses, such as genetics, physics, and molecular biology. She completed her degree requirements and graduated with honors, with a Bachelor of Science degree in molecular biology. She also served as a marshal in the commencement ceremony procession.
Over the next year, with strong encouragement, Bethany wrote a book about her remarkable recovery from refractory psychosis.1 In addition, her mother wrote a deeply emotional book that described the gut-wrenching ordeal that she and her husband went through during the years that Bethany disappeared.2 I urge you to read these inspiring books (Figure) about the remarkable recovery from refractory psychosis and the heavy family burden of schizophrenia.
Back to clozapine
Although the package insert for clozapine contains 5 black-box warnings (for agranulocytosis, seizures, myocarditis, respiratory effects, and increased mortality in geriatric patients with psychosis associated with dementia), the drug is a useful last-resort medication for several approved indications and off-label uses. In addition to the official, evidence-based indication for treatment-resistant and refractory schizophrenia,3 clozapine is FDA-approved for suicidality in schizophrenia.4 Clinically reported, but unapproved, uses are listed in the Table.5-13
A little-known advantage of clozapine is its salutary effect on mortality. In a Finnish study of 66,881 persons who had schizophrenia,14 those taking clozapine had, overall, lower mortality during the treatment period than those taking any of the 6 most commonly used antipsychotic drugs.
No doubt, clozapine is associated with serious side effects15—but so is chemotherapy for cancer, and oncologists do not hesitate to use it to save their patients from physical death. Severe schizophrenia is like a cancer of the mind, and clozapine is its chemotherapy.
Fortunately for Bethany, she had almost no physical adverse effects from clozapine except for intense sedation, which was mitigated with modafinil.
We should use clozapine more than we do
Clozapine has the potential to have a healing effect for many patients whose schizophrenia is resistant to treatment. Most such patients, however, never receive a trial of the drug. Furthermore, few practitioners use clozapine for schizophrenia patients with suicidal tendencies, despite the high rate of suicide completion in schizophrenia.16
Clozapine remains, regrettably, an underutilized agent in psychiatry. Until other breakthrough drugs are discovered, its use ought to be double or triple what it is now because there are many people like Bethany who are not being given a chance to recover from their illness.
Since clozapine was launched in 1989, miraculous improvements and “awakenings” have been reported in many patients afflicted with severe schizophrenia and considered hopelessly refractory to antipsychotic pharmacotherapy. Not only do severely disabled patients regain their sanity and return to normal functioning, but the joy that their family and treating psychiatrist experience is priceless.
That’s why I am perplexed by how infrequently clozapine is used in the United States (in about 5% of patients)—even though approximately 25% of patients who have schizophrenia are either treatment-resistant or have refractory hallucinations or delusions.
Consider Bethany’s case. She was one of my young patients, who, after taking clozapine, recovered fully and resumed a productive life, after years of homelessness during which she was controlled by auditory hallucinations.
Bethany’s story began well…
Bethany grew up in a loving home, smart and talented, an “A” student in high school and talented violinist. She received a scholarship to a prestigious private university at 16 and left her parent’s home in Ohio to major in molecular biology. Her goal was to attend medical school. She excelled during her first 3 years of college, and even published 2 papers in top-tier science journals.
In her senior year, after returning from a trip to Africa, Bethany began to change. She neglected her studies and focused on raising money for HIV clinics in Africa. She began getting F’s instead of A’s, lost her scholarship and her residence hall room, and had to drop out of college. Soon, she began hearing voices commanding her every action.
..but took a really bad turn
Bethany became homeless for the next 4.5 years. She ate discarded food from garbage cans, had no change of clothes, and slept on a concrete slab behind a downtown church in a major city in California. Her parents lost track of her, although her mother, a retired nurse, frantically and relentlessly tried to find out what happened to her only daughter during that time.
Eventually, Bethany was arrested when she was found screaming back at the voices, at midnight in a residential area of the city. She was hospitalized on a psychiatric ward and given antipsychotics, but with only modest improvement.
Her parents were contacted; immediately, they flew to California to see her. The treating psychiatrist told them that their daughter had schizophrenia, and that they should lower their expectations because she would be totally disabled for the rest of her life. They brought Bethany back to Ohio where, after a tumultuous year of failed trials of several antipsychotics to suppress the auditory hallucinations, we gave her clozapine.
Gradually, Bethany improved, but she still could not read a book or magazine (which I urged her to do) without the voices intensifying and preventing her from reading.
Bethany recovers
After 6 to 8 months on clozapine, however, Bethany’s auditory hallucinations faded away. With my encouragement, she enrolled at the University of Cincinnati and took 1 course at a time. She began to get A’s again—in advanced courses, such as genetics, physics, and molecular biology. She completed her degree requirements and graduated with honors, with a Bachelor of Science degree in molecular biology. She also served as a marshal in the commencement ceremony procession.
Over the next year, with strong encouragement, Bethany wrote a book about her remarkable recovery from refractory psychosis.1 In addition, her mother wrote a deeply emotional book that described the gut-wrenching ordeal that she and her husband went through during the years that Bethany disappeared.2 I urge you to read these inspiring books (Figure) about the remarkable recovery from refractory psychosis and the heavy family burden of schizophrenia.
Back to clozapine
Although the package insert for clozapine contains 5 black-box warnings (for agranulocytosis, seizures, myocarditis, respiratory effects, and increased mortality in geriatric patients with psychosis associated with dementia), the drug is a useful last-resort medication for several approved indications and off-label uses. In addition to the official, evidence-based indication for treatment-resistant and refractory schizophrenia,3 clozapine is FDA-approved for suicidality in schizophrenia.4 Clinically reported, but unapproved, uses are listed in the Table.5-13
A little-known advantage of clozapine is its salutary effect on mortality. In a Finnish study of 66,881 persons who had schizophrenia,14 those taking clozapine had, overall, lower mortality during the treatment period than those taking any of the 6 most commonly used antipsychotic drugs.
No doubt, clozapine is associated with serious side effects15—but so is chemotherapy for cancer, and oncologists do not hesitate to use it to save their patients from physical death. Severe schizophrenia is like a cancer of the mind, and clozapine is its chemotherapy.
Fortunately for Bethany, she had almost no physical adverse effects from clozapine except for intense sedation, which was mitigated with modafinil.
We should use clozapine more than we do
Clozapine has the potential to have a healing effect for many patients whose schizophrenia is resistant to treatment. Most such patients, however, never receive a trial of the drug. Furthermore, few practitioners use clozapine for schizophrenia patients with suicidal tendencies, despite the high rate of suicide completion in schizophrenia.16
Clozapine remains, regrettably, an underutilized agent in psychiatry. Until other breakthrough drugs are discovered, its use ought to be double or triple what it is now because there are many people like Bethany who are not being given a chance to recover from their illness.
1. Yeiser B. Mind estranged. My journey from schizophrenia and homelessness to recovery. North Charleston, SC: CreateSpace Independent Publishing Platform; 2014.
2. Yeiser KS. Flight from reason: a mother’s story of schizophrenia, recovery and hope. North Charleston, SC: CreateSpace Independent Publishing Platform; 2014.
3. Kane J, Honigfeld G, Singer J, et al. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch Gen Psychiatry. 1988;45(9):789-796.
4. Meltzer HY, Alphs L, Green AI, et al. Clozapine treatment for suicidality in schizophrenia: International Suicide Prevention Trial (InterSePT) [Erratum in: Arch Gen Psychiatry. 2003;60(7):735.] Arch Gen Psychiatry. 2003;60(1):82-91.
5. Frogley C, Taylor D, Dickens G, et al. A systematic review of the evidence of clozapine’s anti-aggressive effects. Int J Neuropsychopharmacol. 2012;15(9):1351-1371.
6. Margetié B, Aukst-Margetié B, Zarkovié-Palijan T. Successful treatment of polydipsia, water intoxication, and delusional jealousy in an alcohol dependent patient with clozapine. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30(7):1347-1349.
7. Cupina D, Boulton M. Secondary delusional parasitosis treated successfully with a combination of clozapine and citalopram. Psychosomatics. 2012;53(3):301-302.
8. Connolly BD, Lang AE. Pharmacolgoic treatment of Parkinson disease: a review. JAMA. 2014;311(16):1670-1683.
9. Hazari N, Kate N, Grover S, et al. Clozapine and tardive movement disorders: a review. Asian J Psychiatry. 2013;6(6):439-451.
10. Zarzar T, McEvoy J. Clozapine for self-injurious behavior in individuals with borderline personality disorder. Ther Adv Psychopharmacol. 2013;3(5):272-274.
11. Vohra AK. Treatment of severe borderline personality disorder with clozapine. Indian J Psychiatry. 2010;52(3):267-269.
12. Ifteni P, Correll CU, Nielse J, et al. Rapid clozapine titration in treatment-refractory bipolar disorder. J Affect Disord. 2014;166:168-172.
13. Rogoz Z. Combined treatment with atypical antipsychotics and antidepressants in treatment-resistant depression: preclinical and clinical efficacy. Pharmacol Rep. 2013;65(6)1535-1544.
14. Tiihonen J, Lönnqvist J, Wahlbeck K, et al. 11-year follow-up of mortality in patients with schizophrenia: a population-based cohort study (FIN11 study). Lancet. 2009;374(9690):620-627.
15. Raja M, Raja S. Clozapine safety, 40 years later [published online April 28, 2014]. Curr Drug Saf. doi: 10.2174/1574886309666140428115040.
16. Siris SG. Suicide and schizophrenia. J Psychopharmacol. 2001;15(2):127-135.
1. Yeiser B. Mind estranged. My journey from schizophrenia and homelessness to recovery. North Charleston, SC: CreateSpace Independent Publishing Platform; 2014.
2. Yeiser KS. Flight from reason: a mother’s story of schizophrenia, recovery and hope. North Charleston, SC: CreateSpace Independent Publishing Platform; 2014.
3. Kane J, Honigfeld G, Singer J, et al. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch Gen Psychiatry. 1988;45(9):789-796.
4. Meltzer HY, Alphs L, Green AI, et al. Clozapine treatment for suicidality in schizophrenia: International Suicide Prevention Trial (InterSePT) [Erratum in: Arch Gen Psychiatry. 2003;60(7):735.] Arch Gen Psychiatry. 2003;60(1):82-91.
5. Frogley C, Taylor D, Dickens G, et al. A systematic review of the evidence of clozapine’s anti-aggressive effects. Int J Neuropsychopharmacol. 2012;15(9):1351-1371.
6. Margetié B, Aukst-Margetié B, Zarkovié-Palijan T. Successful treatment of polydipsia, water intoxication, and delusional jealousy in an alcohol dependent patient with clozapine. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30(7):1347-1349.
7. Cupina D, Boulton M. Secondary delusional parasitosis treated successfully with a combination of clozapine and citalopram. Psychosomatics. 2012;53(3):301-302.
8. Connolly BD, Lang AE. Pharmacolgoic treatment of Parkinson disease: a review. JAMA. 2014;311(16):1670-1683.
9. Hazari N, Kate N, Grover S, et al. Clozapine and tardive movement disorders: a review. Asian J Psychiatry. 2013;6(6):439-451.
10. Zarzar T, McEvoy J. Clozapine for self-injurious behavior in individuals with borderline personality disorder. Ther Adv Psychopharmacol. 2013;3(5):272-274.
11. Vohra AK. Treatment of severe borderline personality disorder with clozapine. Indian J Psychiatry. 2010;52(3):267-269.
12. Ifteni P, Correll CU, Nielse J, et al. Rapid clozapine titration in treatment-refractory bipolar disorder. J Affect Disord. 2014;166:168-172.
13. Rogoz Z. Combined treatment with atypical antipsychotics and antidepressants in treatment-resistant depression: preclinical and clinical efficacy. Pharmacol Rep. 2013;65(6)1535-1544.
14. Tiihonen J, Lönnqvist J, Wahlbeck K, et al. 11-year follow-up of mortality in patients with schizophrenia: a population-based cohort study (FIN11 study). Lancet. 2009;374(9690):620-627.
15. Raja M, Raja S. Clozapine safety, 40 years later [published online April 28, 2014]. Curr Drug Saf. doi: 10.2174/1574886309666140428115040.
16. Siris SG. Suicide and schizophrenia. J Psychopharmacol. 2001;15(2):127-135.
Polycystic kidney disease: Molecular understanding dictating management
Dr. Braun is an iconic figure in Cleveland Clinic medicine. He is the consummate internist, nephrologist, and transplantation physician, but he is also a critical thinker. He strives to understand (and explain) what underpins our clinical observations and therapeutic decisions. He asks the “why” questions. As he ticked through the manifestations of PKD and the diagnostic dilemmas that arise in taking care of these patients, and then transitioned into explaining the interesting though incomplete current molecular understanding of this relatively prevalent genetic disorder, I heard many of the same questions I had asked myself 30 years ago. But this time I was getting some answers.
How can one be certain a cyst is infected? How do these cysts form and expand without apparent communication with the tubular lumens? (Intracystic bleeding and infection may not be reflected in the urinalysis, although the organism isolated from infected cysts is frequently Escherichia coli.) If renal cysts are formed from tubular epithelial cells that are preprogrammed to self-organize into lumen-like structures, how does the same genetic defect predispose to cyst formation in organs such as the liver, or to aneurysms in blood vessels in the brain? Why does the disease take so long to express itself, and why is its expression so variable?
The patient did well during his hospital stay 30 years ago. As I recall, he had staphylococcal bacteremia with an infected cyst. We discussed the clinical scenario but had no suggestions as to how to prevent the growth of what we now know are about 60 subclinical cysts for every one that we recognize. And we certainly didn’t discuss the idea that the disease process may be partially driven by dysfunctional nonmotile cilia that should respond to urine flow by appropriately directing regeneration and proliferation of renal tubular cells.
I love getting answers to questions that I didn’t know enough to ask.
Dr. Braun is an iconic figure in Cleveland Clinic medicine. He is the consummate internist, nephrologist, and transplantation physician, but he is also a critical thinker. He strives to understand (and explain) what underpins our clinical observations and therapeutic decisions. He asks the “why” questions. As he ticked through the manifestations of PKD and the diagnostic dilemmas that arise in taking care of these patients, and then transitioned into explaining the interesting though incomplete current molecular understanding of this relatively prevalent genetic disorder, I heard many of the same questions I had asked myself 30 years ago. But this time I was getting some answers.
How can one be certain a cyst is infected? How do these cysts form and expand without apparent communication with the tubular lumens? (Intracystic bleeding and infection may not be reflected in the urinalysis, although the organism isolated from infected cysts is frequently Escherichia coli.) If renal cysts are formed from tubular epithelial cells that are preprogrammed to self-organize into lumen-like structures, how does the same genetic defect predispose to cyst formation in organs such as the liver, or to aneurysms in blood vessels in the brain? Why does the disease take so long to express itself, and why is its expression so variable?
The patient did well during his hospital stay 30 years ago. As I recall, he had staphylococcal bacteremia with an infected cyst. We discussed the clinical scenario but had no suggestions as to how to prevent the growth of what we now know are about 60 subclinical cysts for every one that we recognize. And we certainly didn’t discuss the idea that the disease process may be partially driven by dysfunctional nonmotile cilia that should respond to urine flow by appropriately directing regeneration and proliferation of renal tubular cells.
I love getting answers to questions that I didn’t know enough to ask.
Dr. Braun is an iconic figure in Cleveland Clinic medicine. He is the consummate internist, nephrologist, and transplantation physician, but he is also a critical thinker. He strives to understand (and explain) what underpins our clinical observations and therapeutic decisions. He asks the “why” questions. As he ticked through the manifestations of PKD and the diagnostic dilemmas that arise in taking care of these patients, and then transitioned into explaining the interesting though incomplete current molecular understanding of this relatively prevalent genetic disorder, I heard many of the same questions I had asked myself 30 years ago. But this time I was getting some answers.
How can one be certain a cyst is infected? How do these cysts form and expand without apparent communication with the tubular lumens? (Intracystic bleeding and infection may not be reflected in the urinalysis, although the organism isolated from infected cysts is frequently Escherichia coli.) If renal cysts are formed from tubular epithelial cells that are preprogrammed to self-organize into lumen-like structures, how does the same genetic defect predispose to cyst formation in organs such as the liver, or to aneurysms in blood vessels in the brain? Why does the disease take so long to express itself, and why is its expression so variable?
The patient did well during his hospital stay 30 years ago. As I recall, he had staphylococcal bacteremia with an infected cyst. We discussed the clinical scenario but had no suggestions as to how to prevent the growth of what we now know are about 60 subclinical cysts for every one that we recognize. And we certainly didn’t discuss the idea that the disease process may be partially driven by dysfunctional nonmotile cilia that should respond to urine flow by appropriately directing regeneration and proliferation of renal tubular cells.
I love getting answers to questions that I didn’t know enough to ask.
Progression-free survival, patient-reported outcomes, and the Holy Grail
In oncology clinical trials, overall survival (OS) remains the gold standard for clinical benefit. However, because there are so many available treatment options for most types of cancer, survival analysis in clinical trials is often confounded by subsequent therapies. Progression-free survival (PFS) is an endpoint not so confounded, and one that requires fewer patients and less time to arrive at a conclusion about a new therapy. As a result, an ever-increasing number of oncology clinical trials are launched in which PFS is used as the primary endpoint. Several years ago, Pazdur1 emphasized that the time-to-progression endpoint must use the same evaluation techniques and schedules for all treatment arms and he recommended blinding of trials or at minimum, the use of an external blinded radiographic review committee. He also noted that improvement in disease-related symptoms qualifies as clinical benefit and may therefore be an appropriate endpoint for drug approval.
Click on the PDF icon at the top of this introduction to read the full article.
In oncology clinical trials, overall survival (OS) remains the gold standard for clinical benefit. However, because there are so many available treatment options for most types of cancer, survival analysis in clinical trials is often confounded by subsequent therapies. Progression-free survival (PFS) is an endpoint not so confounded, and one that requires fewer patients and less time to arrive at a conclusion about a new therapy. As a result, an ever-increasing number of oncology clinical trials are launched in which PFS is used as the primary endpoint. Several years ago, Pazdur1 emphasized that the time-to-progression endpoint must use the same evaluation techniques and schedules for all treatment arms and he recommended blinding of trials or at minimum, the use of an external blinded radiographic review committee. He also noted that improvement in disease-related symptoms qualifies as clinical benefit and may therefore be an appropriate endpoint for drug approval.
Click on the PDF icon at the top of this introduction to read the full article.
In oncology clinical trials, overall survival (OS) remains the gold standard for clinical benefit. However, because there are so many available treatment options for most types of cancer, survival analysis in clinical trials is often confounded by subsequent therapies. Progression-free survival (PFS) is an endpoint not so confounded, and one that requires fewer patients and less time to arrive at a conclusion about a new therapy. As a result, an ever-increasing number of oncology clinical trials are launched in which PFS is used as the primary endpoint. Several years ago, Pazdur1 emphasized that the time-to-progression endpoint must use the same evaluation techniques and schedules for all treatment arms and he recommended blinding of trials or at minimum, the use of an external blinded radiographic review committee. He also noted that improvement in disease-related symptoms qualifies as clinical benefit and may therefore be an appropriate endpoint for drug approval.
Click on the PDF icon at the top of this introduction to read the full article.
Farewell to indigo carmine
Suddenly, indigo carmine is in short supply throughout the United States. One manufacturer has stopped production because of a raw materials shortage; another is experiencing manufacturing delays. Neither company can estimate a resupply or product return date.1
Indigo carmine is approved by the US Food and Drug Administration (FDA) to localize ureteral orifices during cystoscopy and is commonly used in obstetrics and gynecology as a marker dye in the following additional situations:
- administered in a dilute solution via a catheter to back fill the bladder and test for bladder injury
- administered via a cannula in the uterine cavity to test the patency of the fallopian tubes
- injected into the amniotic fluid compartment to test for premature rupture of the membranes (PROM)
- injected into the amniotic fluid of a twin gestation to mark the amniotic fluid of one twin.
With this agent in short supply, we need to identify alternative marker dyes to use in our clinical practice. In this editorial, I provide a list of possible options to replace indigo carmine. Evidence supporting the use and safety of marker dyes in obstetrics and gynecology is based on small cohorts or case reports. The available evidence is of modest to low quality, and it is especially challenging to identify uncommon adverse effects. Consequently, expert opinion guides most practice recommendations.
Options to test the function of the ureters at cystoscopy
Given the lack of availability of indigo carmine, I recommend one of the following three options to test the function of the ureters at cystoscopy.
Partially fill the bladder with a solution of either sterile water or a 10% dextrose solution. (Experienced surgeons may prefer to use saline.) The turbulence of the interaction between the ureteral urine jet and instilled fluid in the bladder may permit visualization of the urine stream exiting the ureteral orifices as it swirls through the sterile water or dextrose solution. Both sterile water and a 10% dextrose solution offer a contrast in viscosity between the urine and the cystoscopy fluid, which may enhance the ability to detect the urine jet leaving the ureter.2
Administer IV methylene blue. Methylene blue is FDA approved for methemoglobinemia treatment. For this indication, it is administered intravenously at a dose of 1 to 2 mg/kgover 5 to 10 minutes. Paradoxically, when administered at a dose of >7 mg/kg, methylene blue can cause methemoglobinemia.3
Methylene blue often is provided as a 1% solution of 10 mg/mL in 10-mL vials. To use intravenous (IV) methylene blue to test ureteral function at cystoscopy, administer IV methylene blue 50 mg over 5 minutes. Use the cystoscope to view the colored urine exiting the ureteral orifices.4,5 Administering a small dose of IV furosemide may accelerate the appearance of methylene blue in the urine.
When to use caution. Methylene blue blocks serotonin metabolism by inhibiting monoamine oxidase and may precipitate a serotonin syndrome in patients taking selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), or monoamine oxidase inhibitors (MAOIs).
Common findings in the serotonin syndrome include: temperature above 38° C (100.4° F), anxiety, agitation, delirium, clonus, tremor, and hypertonia.6 I recommend that you DO NOT use IV methylene blue in patients taking these medications.7,8
Great caution should be used before administering IV methylene blue in the presence of the following clinical situations:
- renal impairment
- G6PD deficiency
- pediatric patients.
Methylene blue never should be given by a subcutaneous or intrathecal route. In pregnant women it should never be injected into the amniotic fluid compartment.
Use preoperative oral phenazo-pyridine. If the preoperative plan includes a cystoscopy procedure to test ureteral function, administering oral phenazopyridine in the preoperative holding area will result in colorization of the urine within 30 minutes, and it will persist for approximately 4 to 5 hours. To use this approach, administer one dose of phenazopyridine (Pyridium, Azo-Gesic), 100 mg or 200 mg orally, 30 minutes to 1 hour before the planned surgical start time, in the preoperative holding area.9 During cystoscopy, the urine from the ureteral jet will be colored orange.
When to use caution. Phenazopyridine should not be administered to patients with G6PD deficiency.
Option to test for bladder injury
Methylene blue. If methylene blue is used to test the integrity of the bladder, I recommend diluting 10 mg methylene blue in 1 L normal saline and then instilling the dilute solution through a catheter into the bladder.10
It is unlikely that this technique will be associated with sufficient methylene blue absorption to cause a serotonin syndrome. Therefore, this technique can be used in patients taking SSRIs, SNRIs, and MAOIs.
Option to test patency of the fallopian tubes (chromopertubation)
Methylene blue. If methylene blue is used via a cannula in the uterine cavity to test the patency of the fallopian tubes, I recommend diluting 10 mg methylene blue in 150 mL normal saline and using the dilute solution to test tubal patency.
It is unlikely that this process will lead to sufficient methylene blue absorption to cause a serotonin syndrome. Therefore, this technique can be used in patients taking SSRIs, SNRIs and MAOIs. However, if intrauterine injection of the dilute dye solution results in extravasation of the dye into the pelvic veins, a significant amount of dye can enter the circulation.11 There are case reports of anaphylaxis following intrauterine injection of methylene blue to test tubal patency.12,13
Options to diagnose PROM and to use for twin amniocentesis
None. Most experts recommend against the intra-amniotic injection of methylene blue to diagnose PROM or in twin amniocentesis procedures. Methylene blue injected into the intra-amniotic fluid during amniocentesis in multiple gestations has been reported to cause fetal bowel obstruction or atresia.14,15 Fetal death also has been reported.16 Decades ago, indocyanine green, which is FDA approved to determine cardiac output, liver blood flow, and hepatic function, was reported to be useful to mark one sac of a twin gestation during amniocentesis.17 With modern ultrasonography technology, the need to rely on a dye to mark a sac of a twin has decreased significantly.
Our only option is to cope—effectively as possible Over decades, the medical community develops patterns of patient care that are critically dependent on the availability of key pharmaceuticals and devices. When a pharmaceutical or device suddenly becomes unavailable, it can disrupt important patterns of patient care. Imagine the impact on obstetrics practice if oxytocin became unavailable due to manufacturing shortages. Likely, both market forces and government regulation are the root cause of the shortfalls. Preventing the adverse consequences of the sudden loss of key pharmaceuticals and devices is an important priority to ensure optimal care of our patients.
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
1. Indigo carmine injection. American Society of Health-System Pharmacists (ASHP) Web site. http://www.ashp.org/menu/DrugShortages/CurrentShortages/Bulletin.aspx?id=861. Updated July 29, 2014. Accessed August 21, 2014.
2. Lin BL, Iwata Y. Modified cystoscopy to evaluate unilateral traumatic injury of the ureter during pelvic surgery. Am J Obstet Gynecol. 1990;162(5):1343–1344.
3. Lee M, Sharifi R. Methylene blue versus indigo carmine. Urology. 1996;47(5):783–784.
4. Thompson JD. Operative injuries to the ureter: prevention, recognition and management. In Rock JA, Thompson JD, eds. TeLinde’s Operative Gynecology. Philadelphia, PA: Lippincott Williams & Wilkins; 1997:1155.
5. Wang AC. The techniques of trocar insertion and intraoperative urethroscopy in tension-free vaginal taping: an experience of 600 cases. Acta Obstet Gynecol Scand. 2004;83(3):293–298.
6. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112–1120.
7. Shah-Khan MG, Lovely J, Degnim AC. Safety of methylene blue dye for lymphatic mapping in patients taking selective serotonin reuptake inhibitors. Am J Surg. 2012;204(5):798–799.
8. Ng BK, Cameron AJ. The role of methylene blue in serotonin syndrome: a systematic review. Psychosomatics. 2010;51(3):194–200.
9. Hui JY, Harvey MA, Johnston SL. Confirmation of ureteric patency during cystoscopy using phenazopyridine HCl: a low-cost approach. J Obstet Gynaecol Can. 2009;31(9):845–849.
10. Moore CR, Shirodkar SP, Avallone MA, et al. Intravesical methylene blue facilitates precise identification of the diverticular neck during robot-assisted laparoscopic bladder diverticulectomy. J Laparoendosc Adv Surg Tech A. 2012;22(5):492–495.
11. Mhaskar R, Mhaskar AM. Methemoglobinemia following chromopertubation in treated pelvic tuberculosis. Int J Gynaecol Obstet. 2002;77(1):41–42.
12. Rzymski P, Wozniak J, Opala T, Wilczak M, Sajdak S. Anaphylactic reaction to methylene blue dye after laparoscopic chromopertubation. Int J Gynaecol Obstet. 2003;81(1):71–72.
13. Dewachter P, Mouton-Faivre C, Trechot P, Lieu JC, Mertes PM. Severe anaphylactic shock with methylene blue instillation. Anesth Analg. 2005;101(1):149–150.
14. McFadyen I. The dangers of intra-amniotic methylene blue. Br J Obstet Gynaecol. 1992;99(2):89–90.
15. Van der Pol JG, Wolf H, Boer K, et al. Jejunal atresia related to the use of methylene blue in genetic amniocentesis in twins. Br J Obstet Gynaecol. 1992;99(2):141–143.
16. Kidd SA, Lancaster PA, Anderson JC, et al. Fetal death after exposure to methylene blue dye during mid-trimester amniocentesis in twin pregnancy. Prenat Diagn. 1996;16(1):39–47.
17. Hobbins JC, Winsberg F, Blanchett M, et al. Section 5: fetal imaging. Prenat Diagn. 1981;1(5):35–38.
Suddenly, indigo carmine is in short supply throughout the United States. One manufacturer has stopped production because of a raw materials shortage; another is experiencing manufacturing delays. Neither company can estimate a resupply or product return date.1
Indigo carmine is approved by the US Food and Drug Administration (FDA) to localize ureteral orifices during cystoscopy and is commonly used in obstetrics and gynecology as a marker dye in the following additional situations:
- administered in a dilute solution via a catheter to back fill the bladder and test for bladder injury
- administered via a cannula in the uterine cavity to test the patency of the fallopian tubes
- injected into the amniotic fluid compartment to test for premature rupture of the membranes (PROM)
- injected into the amniotic fluid of a twin gestation to mark the amniotic fluid of one twin.
With this agent in short supply, we need to identify alternative marker dyes to use in our clinical practice. In this editorial, I provide a list of possible options to replace indigo carmine. Evidence supporting the use and safety of marker dyes in obstetrics and gynecology is based on small cohorts or case reports. The available evidence is of modest to low quality, and it is especially challenging to identify uncommon adverse effects. Consequently, expert opinion guides most practice recommendations.
Options to test the function of the ureters at cystoscopy
Given the lack of availability of indigo carmine, I recommend one of the following three options to test the function of the ureters at cystoscopy.
Partially fill the bladder with a solution of either sterile water or a 10% dextrose solution. (Experienced surgeons may prefer to use saline.) The turbulence of the interaction between the ureteral urine jet and instilled fluid in the bladder may permit visualization of the urine stream exiting the ureteral orifices as it swirls through the sterile water or dextrose solution. Both sterile water and a 10% dextrose solution offer a contrast in viscosity between the urine and the cystoscopy fluid, which may enhance the ability to detect the urine jet leaving the ureter.2
Administer IV methylene blue. Methylene blue is FDA approved for methemoglobinemia treatment. For this indication, it is administered intravenously at a dose of 1 to 2 mg/kgover 5 to 10 minutes. Paradoxically, when administered at a dose of >7 mg/kg, methylene blue can cause methemoglobinemia.3
Methylene blue often is provided as a 1% solution of 10 mg/mL in 10-mL vials. To use intravenous (IV) methylene blue to test ureteral function at cystoscopy, administer IV methylene blue 50 mg over 5 minutes. Use the cystoscope to view the colored urine exiting the ureteral orifices.4,5 Administering a small dose of IV furosemide may accelerate the appearance of methylene blue in the urine.
When to use caution. Methylene blue blocks serotonin metabolism by inhibiting monoamine oxidase and may precipitate a serotonin syndrome in patients taking selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), or monoamine oxidase inhibitors (MAOIs).
Common findings in the serotonin syndrome include: temperature above 38° C (100.4° F), anxiety, agitation, delirium, clonus, tremor, and hypertonia.6 I recommend that you DO NOT use IV methylene blue in patients taking these medications.7,8
Great caution should be used before administering IV methylene blue in the presence of the following clinical situations:
- renal impairment
- G6PD deficiency
- pediatric patients.
Methylene blue never should be given by a subcutaneous or intrathecal route. In pregnant women it should never be injected into the amniotic fluid compartment.
Use preoperative oral phenazo-pyridine. If the preoperative plan includes a cystoscopy procedure to test ureteral function, administering oral phenazopyridine in the preoperative holding area will result in colorization of the urine within 30 minutes, and it will persist for approximately 4 to 5 hours. To use this approach, administer one dose of phenazopyridine (Pyridium, Azo-Gesic), 100 mg or 200 mg orally, 30 minutes to 1 hour before the planned surgical start time, in the preoperative holding area.9 During cystoscopy, the urine from the ureteral jet will be colored orange.
When to use caution. Phenazopyridine should not be administered to patients with G6PD deficiency.
Option to test for bladder injury
Methylene blue. If methylene blue is used to test the integrity of the bladder, I recommend diluting 10 mg methylene blue in 1 L normal saline and then instilling the dilute solution through a catheter into the bladder.10
It is unlikely that this technique will be associated with sufficient methylene blue absorption to cause a serotonin syndrome. Therefore, this technique can be used in patients taking SSRIs, SNRIs, and MAOIs.
Option to test patency of the fallopian tubes (chromopertubation)
Methylene blue. If methylene blue is used via a cannula in the uterine cavity to test the patency of the fallopian tubes, I recommend diluting 10 mg methylene blue in 150 mL normal saline and using the dilute solution to test tubal patency.
It is unlikely that this process will lead to sufficient methylene blue absorption to cause a serotonin syndrome. Therefore, this technique can be used in patients taking SSRIs, SNRIs and MAOIs. However, if intrauterine injection of the dilute dye solution results in extravasation of the dye into the pelvic veins, a significant amount of dye can enter the circulation.11 There are case reports of anaphylaxis following intrauterine injection of methylene blue to test tubal patency.12,13
Options to diagnose PROM and to use for twin amniocentesis
None. Most experts recommend against the intra-amniotic injection of methylene blue to diagnose PROM or in twin amniocentesis procedures. Methylene blue injected into the intra-amniotic fluid during amniocentesis in multiple gestations has been reported to cause fetal bowel obstruction or atresia.14,15 Fetal death also has been reported.16 Decades ago, indocyanine green, which is FDA approved to determine cardiac output, liver blood flow, and hepatic function, was reported to be useful to mark one sac of a twin gestation during amniocentesis.17 With modern ultrasonography technology, the need to rely on a dye to mark a sac of a twin has decreased significantly.
Our only option is to cope—effectively as possible Over decades, the medical community develops patterns of patient care that are critically dependent on the availability of key pharmaceuticals and devices. When a pharmaceutical or device suddenly becomes unavailable, it can disrupt important patterns of patient care. Imagine the impact on obstetrics practice if oxytocin became unavailable due to manufacturing shortages. Likely, both market forces and government regulation are the root cause of the shortfalls. Preventing the adverse consequences of the sudden loss of key pharmaceuticals and devices is an important priority to ensure optimal care of our patients.
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
Suddenly, indigo carmine is in short supply throughout the United States. One manufacturer has stopped production because of a raw materials shortage; another is experiencing manufacturing delays. Neither company can estimate a resupply or product return date.1
Indigo carmine is approved by the US Food and Drug Administration (FDA) to localize ureteral orifices during cystoscopy and is commonly used in obstetrics and gynecology as a marker dye in the following additional situations:
- administered in a dilute solution via a catheter to back fill the bladder and test for bladder injury
- administered via a cannula in the uterine cavity to test the patency of the fallopian tubes
- injected into the amniotic fluid compartment to test for premature rupture of the membranes (PROM)
- injected into the amniotic fluid of a twin gestation to mark the amniotic fluid of one twin.
With this agent in short supply, we need to identify alternative marker dyes to use in our clinical practice. In this editorial, I provide a list of possible options to replace indigo carmine. Evidence supporting the use and safety of marker dyes in obstetrics and gynecology is based on small cohorts or case reports. The available evidence is of modest to low quality, and it is especially challenging to identify uncommon adverse effects. Consequently, expert opinion guides most practice recommendations.
Options to test the function of the ureters at cystoscopy
Given the lack of availability of indigo carmine, I recommend one of the following three options to test the function of the ureters at cystoscopy.
Partially fill the bladder with a solution of either sterile water or a 10% dextrose solution. (Experienced surgeons may prefer to use saline.) The turbulence of the interaction between the ureteral urine jet and instilled fluid in the bladder may permit visualization of the urine stream exiting the ureteral orifices as it swirls through the sterile water or dextrose solution. Both sterile water and a 10% dextrose solution offer a contrast in viscosity between the urine and the cystoscopy fluid, which may enhance the ability to detect the urine jet leaving the ureter.2
Administer IV methylene blue. Methylene blue is FDA approved for methemoglobinemia treatment. For this indication, it is administered intravenously at a dose of 1 to 2 mg/kgover 5 to 10 minutes. Paradoxically, when administered at a dose of >7 mg/kg, methylene blue can cause methemoglobinemia.3
Methylene blue often is provided as a 1% solution of 10 mg/mL in 10-mL vials. To use intravenous (IV) methylene blue to test ureteral function at cystoscopy, administer IV methylene blue 50 mg over 5 minutes. Use the cystoscope to view the colored urine exiting the ureteral orifices.4,5 Administering a small dose of IV furosemide may accelerate the appearance of methylene blue in the urine.
When to use caution. Methylene blue blocks serotonin metabolism by inhibiting monoamine oxidase and may precipitate a serotonin syndrome in patients taking selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), or monoamine oxidase inhibitors (MAOIs).
Common findings in the serotonin syndrome include: temperature above 38° C (100.4° F), anxiety, agitation, delirium, clonus, tremor, and hypertonia.6 I recommend that you DO NOT use IV methylene blue in patients taking these medications.7,8
Great caution should be used before administering IV methylene blue in the presence of the following clinical situations:
- renal impairment
- G6PD deficiency
- pediatric patients.
Methylene blue never should be given by a subcutaneous or intrathecal route. In pregnant women it should never be injected into the amniotic fluid compartment.
Use preoperative oral phenazo-pyridine. If the preoperative plan includes a cystoscopy procedure to test ureteral function, administering oral phenazopyridine in the preoperative holding area will result in colorization of the urine within 30 minutes, and it will persist for approximately 4 to 5 hours. To use this approach, administer one dose of phenazopyridine (Pyridium, Azo-Gesic), 100 mg or 200 mg orally, 30 minutes to 1 hour before the planned surgical start time, in the preoperative holding area.9 During cystoscopy, the urine from the ureteral jet will be colored orange.
When to use caution. Phenazopyridine should not be administered to patients with G6PD deficiency.
Option to test for bladder injury
Methylene blue. If methylene blue is used to test the integrity of the bladder, I recommend diluting 10 mg methylene blue in 1 L normal saline and then instilling the dilute solution through a catheter into the bladder.10
It is unlikely that this technique will be associated with sufficient methylene blue absorption to cause a serotonin syndrome. Therefore, this technique can be used in patients taking SSRIs, SNRIs, and MAOIs.
Option to test patency of the fallopian tubes (chromopertubation)
Methylene blue. If methylene blue is used via a cannula in the uterine cavity to test the patency of the fallopian tubes, I recommend diluting 10 mg methylene blue in 150 mL normal saline and using the dilute solution to test tubal patency.
It is unlikely that this process will lead to sufficient methylene blue absorption to cause a serotonin syndrome. Therefore, this technique can be used in patients taking SSRIs, SNRIs and MAOIs. However, if intrauterine injection of the dilute dye solution results in extravasation of the dye into the pelvic veins, a significant amount of dye can enter the circulation.11 There are case reports of anaphylaxis following intrauterine injection of methylene blue to test tubal patency.12,13
Options to diagnose PROM and to use for twin amniocentesis
None. Most experts recommend against the intra-amniotic injection of methylene blue to diagnose PROM or in twin amniocentesis procedures. Methylene blue injected into the intra-amniotic fluid during amniocentesis in multiple gestations has been reported to cause fetal bowel obstruction or atresia.14,15 Fetal death also has been reported.16 Decades ago, indocyanine green, which is FDA approved to determine cardiac output, liver blood flow, and hepatic function, was reported to be useful to mark one sac of a twin gestation during amniocentesis.17 With modern ultrasonography technology, the need to rely on a dye to mark a sac of a twin has decreased significantly.
Our only option is to cope—effectively as possible Over decades, the medical community develops patterns of patient care that are critically dependent on the availability of key pharmaceuticals and devices. When a pharmaceutical or device suddenly becomes unavailable, it can disrupt important patterns of patient care. Imagine the impact on obstetrics practice if oxytocin became unavailable due to manufacturing shortages. Likely, both market forces and government regulation are the root cause of the shortfalls. Preventing the adverse consequences of the sudden loss of key pharmaceuticals and devices is an important priority to ensure optimal care of our patients.
Share your thoughts on this article! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
1. Indigo carmine injection. American Society of Health-System Pharmacists (ASHP) Web site. http://www.ashp.org/menu/DrugShortages/CurrentShortages/Bulletin.aspx?id=861. Updated July 29, 2014. Accessed August 21, 2014.
2. Lin BL, Iwata Y. Modified cystoscopy to evaluate unilateral traumatic injury of the ureter during pelvic surgery. Am J Obstet Gynecol. 1990;162(5):1343–1344.
3. Lee M, Sharifi R. Methylene blue versus indigo carmine. Urology. 1996;47(5):783–784.
4. Thompson JD. Operative injuries to the ureter: prevention, recognition and management. In Rock JA, Thompson JD, eds. TeLinde’s Operative Gynecology. Philadelphia, PA: Lippincott Williams & Wilkins; 1997:1155.
5. Wang AC. The techniques of trocar insertion and intraoperative urethroscopy in tension-free vaginal taping: an experience of 600 cases. Acta Obstet Gynecol Scand. 2004;83(3):293–298.
6. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112–1120.
7. Shah-Khan MG, Lovely J, Degnim AC. Safety of methylene blue dye for lymphatic mapping in patients taking selective serotonin reuptake inhibitors. Am J Surg. 2012;204(5):798–799.
8. Ng BK, Cameron AJ. The role of methylene blue in serotonin syndrome: a systematic review. Psychosomatics. 2010;51(3):194–200.
9. Hui JY, Harvey MA, Johnston SL. Confirmation of ureteric patency during cystoscopy using phenazopyridine HCl: a low-cost approach. J Obstet Gynaecol Can. 2009;31(9):845–849.
10. Moore CR, Shirodkar SP, Avallone MA, et al. Intravesical methylene blue facilitates precise identification of the diverticular neck during robot-assisted laparoscopic bladder diverticulectomy. J Laparoendosc Adv Surg Tech A. 2012;22(5):492–495.
11. Mhaskar R, Mhaskar AM. Methemoglobinemia following chromopertubation in treated pelvic tuberculosis. Int J Gynaecol Obstet. 2002;77(1):41–42.
12. Rzymski P, Wozniak J, Opala T, Wilczak M, Sajdak S. Anaphylactic reaction to methylene blue dye after laparoscopic chromopertubation. Int J Gynaecol Obstet. 2003;81(1):71–72.
13. Dewachter P, Mouton-Faivre C, Trechot P, Lieu JC, Mertes PM. Severe anaphylactic shock with methylene blue instillation. Anesth Analg. 2005;101(1):149–150.
14. McFadyen I. The dangers of intra-amniotic methylene blue. Br J Obstet Gynaecol. 1992;99(2):89–90.
15. Van der Pol JG, Wolf H, Boer K, et al. Jejunal atresia related to the use of methylene blue in genetic amniocentesis in twins. Br J Obstet Gynaecol. 1992;99(2):141–143.
16. Kidd SA, Lancaster PA, Anderson JC, et al. Fetal death after exposure to methylene blue dye during mid-trimester amniocentesis in twin pregnancy. Prenat Diagn. 1996;16(1):39–47.
17. Hobbins JC, Winsberg F, Blanchett M, et al. Section 5: fetal imaging. Prenat Diagn. 1981;1(5):35–38.
1. Indigo carmine injection. American Society of Health-System Pharmacists (ASHP) Web site. http://www.ashp.org/menu/DrugShortages/CurrentShortages/Bulletin.aspx?id=861. Updated July 29, 2014. Accessed August 21, 2014.
2. Lin BL, Iwata Y. Modified cystoscopy to evaluate unilateral traumatic injury of the ureter during pelvic surgery. Am J Obstet Gynecol. 1990;162(5):1343–1344.
3. Lee M, Sharifi R. Methylene blue versus indigo carmine. Urology. 1996;47(5):783–784.
4. Thompson JD. Operative injuries to the ureter: prevention, recognition and management. In Rock JA, Thompson JD, eds. TeLinde’s Operative Gynecology. Philadelphia, PA: Lippincott Williams & Wilkins; 1997:1155.
5. Wang AC. The techniques of trocar insertion and intraoperative urethroscopy in tension-free vaginal taping: an experience of 600 cases. Acta Obstet Gynecol Scand. 2004;83(3):293–298.
6. Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352(11):1112–1120.
7. Shah-Khan MG, Lovely J, Degnim AC. Safety of methylene blue dye for lymphatic mapping in patients taking selective serotonin reuptake inhibitors. Am J Surg. 2012;204(5):798–799.
8. Ng BK, Cameron AJ. The role of methylene blue in serotonin syndrome: a systematic review. Psychosomatics. 2010;51(3):194–200.
9. Hui JY, Harvey MA, Johnston SL. Confirmation of ureteric patency during cystoscopy using phenazopyridine HCl: a low-cost approach. J Obstet Gynaecol Can. 2009;31(9):845–849.
10. Moore CR, Shirodkar SP, Avallone MA, et al. Intravesical methylene blue facilitates precise identification of the diverticular neck during robot-assisted laparoscopic bladder diverticulectomy. J Laparoendosc Adv Surg Tech A. 2012;22(5):492–495.
11. Mhaskar R, Mhaskar AM. Methemoglobinemia following chromopertubation in treated pelvic tuberculosis. Int J Gynaecol Obstet. 2002;77(1):41–42.
12. Rzymski P, Wozniak J, Opala T, Wilczak M, Sajdak S. Anaphylactic reaction to methylene blue dye after laparoscopic chromopertubation. Int J Gynaecol Obstet. 2003;81(1):71–72.
13. Dewachter P, Mouton-Faivre C, Trechot P, Lieu JC, Mertes PM. Severe anaphylactic shock with methylene blue instillation. Anesth Analg. 2005;101(1):149–150.
14. McFadyen I. The dangers of intra-amniotic methylene blue. Br J Obstet Gynaecol. 1992;99(2):89–90.
15. Van der Pol JG, Wolf H, Boer K, et al. Jejunal atresia related to the use of methylene blue in genetic amniocentesis in twins. Br J Obstet Gynaecol. 1992;99(2):141–143.
16. Kidd SA, Lancaster PA, Anderson JC, et al. Fetal death after exposure to methylene blue dye during mid-trimester amniocentesis in twin pregnancy. Prenat Diagn. 1996;16(1):39–47.
17. Hobbins JC, Winsberg F, Blanchett M, et al. Section 5: fetal imaging. Prenat Diagn. 1981;1(5):35–38.
Can philanthropy fill the unmet needs of psychiatry?
Recent examples come quickly to mind:
• $100 million from the Lieber family to fund the Lieber Institute for Brain Development at Johns Hopkins University
• $300 million from Microsoft co-founder Paul G. Allen to create the Allen Institute for Brain Science
• $650 million from Ted Stanley for the Stanley Center at the Broad Institute.
Such generosity is cause for celebration by psychiatrists and their long-suffering patients who are disabled by a brain disorder. Private money supplements research funding by the National Institutes of Health and will bolster the war against mental illness,1 which costs >$300 billion annually (Box,2page 12).
Although philanthropy will help, many needs in psychiatry are unmet, and not all can be addressed with money. Consider a number of areas of need.
Unmet clinical needs
Models of disease. Psychiatry is in desperate need of an objective, valid diagnostic system that transcends the DSM model of symptom clusters. The Research Domain Criteria3 represents the effort to find an alternative. To achieve that goal, it’s necessary to identify biomarkers and establish their utility—a task that requires a huge amount of funding.
Therapeutics. Clinicians are hungry for innovative, safe pharmaceuticals and non-drug treatments that modify disease, not just alleviate symptoms. Obsessive-compulsive disorder always has lacked such therapies; so have dementia, schizophrenia, personality disorders, dissociative disorders, and sexual pathologies. In fact, >80% of DSM disorders do not have an FDA-approved, evidence-based treatment,4 and many available medications are only partially efficacious, poorly tolerated, or unsafe.
There are more unmet needs in therapeutics:
• Development of promising non-drug therapies, such as neuromodulation, proceeds slowly.
• Research into neurobiological mechanisms of psychotherapy is in its infancy.
• A foolproof method to monitor adherence does not exist, and countless patients relapse needlessly and deteriorate functionally.
• Effective, evidence-based rehabilitation for serious psychiatric disorders is used narrowly and vastly underfunded.
• Insurers continue to thumb their nose at laws that require parity for treating mental illness—thus impeding access to, delaying, or truncating psychiatric care.
Unmet scientific needs
Translational investigators. Despite increased funding for basic neuroscientific study and breathtaking discoveries in animal molecular neurobiology, a trickle of findings has been applied to clinical medicine. This translational gap has many causes, including a shortage of translational neuroscientists (MD-PhD psychiatrists and neurologists), insufficient long-term funding to develop such clinician-researchers, and complex regulatory oversight of human research.
Stalled progress in drug development. Development of novel-mechanism therapeutics for brain disorders is languishing. Some pharmaceutical manufacturers have abandoned the development of drugs that act on the CNS in favor of less complex, more lucrative areas such as oncology and cardiology; others have reduced their investment in CNS products. Developing treatments for knotty disorders of the most complex structure in the known universe requires mammoth investment. Why are stakeholders bailing out on the greatest challenge for science and medicine for easier endeavors?
Discovering new genes for every devastating neuropsychiatric syndrome, such as schizophrenia, is cause for celebration, but the champagne won’t flow until the coding of every gene is unscrambled so that specific biological interventions can be developed. The cost of the chase might be orders of magnitude greater than what is invested in research today. Conceptualizing new models of brain disorders is a critical part of scientific progress and an antidote to the inertia of perpetual group-think. Depression, for example, is being reconceptualized as a disorder of impaired neuroplasticity and neurotropic deficiency, rather than a shortage of serotonin and norepinephrine. Rapid reversal of severe depression to euthymia—in 1 or 2 hours—with IV ketamine shattered the dogma that depression takes weeks to lift, and is ushering in unprecedented new thinking and models likely to revolutionize treatment of severe depression. We need such breakthroughs for other psychiatric brain disorders.
Unmet professional and sociopolitical needs
Broadening of training. Psychiatrists have focused on the mind but insufficiently attended to the biology of the brain. For psychiatry to rise to the next level as a medical specialty and brain discipline, training must incorporate more neurology than it does now. The converse is true in neurology.
Hospitalization not incarceration. It is unconscionable that people suffering from a medical illness that impairs their judgment and behavior are locked up as criminals. Psychiatry must forcefully lobby so that the seriously mentally ill are treated in secure hospitals staffed by physicians, nurses, and mental health professionals.
That’s right: Bring back the asylum to address this unmet medical, political, and ethical need for psychiatric patients.a A serious mental disorder must be accepted as a fault-free illness.
aTo read more about this, I recommend my March 2008 editorial, “Bring back the asylums?,” at CurrentPsychiatry.com, and Dr. George Paulson’s excellent book, Closing the asylums: Causes and consequences of the deinstitutionalization movement (Jefferson, NC: McFarland & Co. Inc; 2012).
Full integration of psychiatry into the rest of medicine remains an unmet need, despite good progress. Because almost every medical illness can cause psychiatric symptoms, DSM-5 mandates that general medical conditions be ruled out before a primary psychiatric diagnosis is made.
Along the same lines, most severely mentally ill persons suffer from medical and neurologic ailments before their first episode,5 and many die prematurely from cardiovascular causes that often are the result of unhealthy lifestyle; iatrogenic complications; and lack of primary care interventions.6 Psychiatric patients must always receive standard general medical evaluation and management, side by side with their psychiatric care.
Philanthropy for psychiatry
Philanthropic support of psychiatry is a salutary trend. Some unmet needs in psychiatry, however, require not only money but a change in attitude (such as eliminating the absurd and discriminatory stigma of mental illness), better training, and forceful political activism by all of us.
1. Licinio J, Wong ML. Launching the ‘war on mental illness’. Mol Psychiatry. 2014;19(1):1-5.
2. Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421-427.
3. Devulapalli KK, Nasrallah HA. An analysis of the high psychotropic off-label use in psychiatric disorders. The majority of psychiatric diagnoses have no approved drug. Asian J Psychiatr. 2009;2(1):29-36.
4. Cuthbert BN. The RDoC framework: facilitating transition from ICD/OSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry. 2014;13(1):28-35.
5. Sørensen HJ, Nielsen PR, Benros ME, et al. Somatic diseases and conditions before the first diagnosis of schizophrenia: a nationwide population-based cohort study in more than 900000 individuals [published online July 25, 2014]. Schizophr Bull. doi: 10.1093/schbul/sbu110.
6. Nasrallah HA, Meyer JM, Goff DC, et al. Low rates of treatment for hypertension, dyslipidemia and diabetes in schizophrenia: data from the CATIE schizophrenia trial sample at baseline. Schizophr Res. 2006;86(1-3):15-22.
Recent examples come quickly to mind:
• $100 million from the Lieber family to fund the Lieber Institute for Brain Development at Johns Hopkins University
• $300 million from Microsoft co-founder Paul G. Allen to create the Allen Institute for Brain Science
• $650 million from Ted Stanley for the Stanley Center at the Broad Institute.
Such generosity is cause for celebration by psychiatrists and their long-suffering patients who are disabled by a brain disorder. Private money supplements research funding by the National Institutes of Health and will bolster the war against mental illness,1 which costs >$300 billion annually (Box,2page 12).
Although philanthropy will help, many needs in psychiatry are unmet, and not all can be addressed with money. Consider a number of areas of need.
Unmet clinical needs
Models of disease. Psychiatry is in desperate need of an objective, valid diagnostic system that transcends the DSM model of symptom clusters. The Research Domain Criteria3 represents the effort to find an alternative. To achieve that goal, it’s necessary to identify biomarkers and establish their utility—a task that requires a huge amount of funding.
Therapeutics. Clinicians are hungry for innovative, safe pharmaceuticals and non-drug treatments that modify disease, not just alleviate symptoms. Obsessive-compulsive disorder always has lacked such therapies; so have dementia, schizophrenia, personality disorders, dissociative disorders, and sexual pathologies. In fact, >80% of DSM disorders do not have an FDA-approved, evidence-based treatment,4 and many available medications are only partially efficacious, poorly tolerated, or unsafe.
There are more unmet needs in therapeutics:
• Development of promising non-drug therapies, such as neuromodulation, proceeds slowly.
• Research into neurobiological mechanisms of psychotherapy is in its infancy.
• A foolproof method to monitor adherence does not exist, and countless patients relapse needlessly and deteriorate functionally.
• Effective, evidence-based rehabilitation for serious psychiatric disorders is used narrowly and vastly underfunded.
• Insurers continue to thumb their nose at laws that require parity for treating mental illness—thus impeding access to, delaying, or truncating psychiatric care.
Unmet scientific needs
Translational investigators. Despite increased funding for basic neuroscientific study and breathtaking discoveries in animal molecular neurobiology, a trickle of findings has been applied to clinical medicine. This translational gap has many causes, including a shortage of translational neuroscientists (MD-PhD psychiatrists and neurologists), insufficient long-term funding to develop such clinician-researchers, and complex regulatory oversight of human research.
Stalled progress in drug development. Development of novel-mechanism therapeutics for brain disorders is languishing. Some pharmaceutical manufacturers have abandoned the development of drugs that act on the CNS in favor of less complex, more lucrative areas such as oncology and cardiology; others have reduced their investment in CNS products. Developing treatments for knotty disorders of the most complex structure in the known universe requires mammoth investment. Why are stakeholders bailing out on the greatest challenge for science and medicine for easier endeavors?
Discovering new genes for every devastating neuropsychiatric syndrome, such as schizophrenia, is cause for celebration, but the champagne won’t flow until the coding of every gene is unscrambled so that specific biological interventions can be developed. The cost of the chase might be orders of magnitude greater than what is invested in research today. Conceptualizing new models of brain disorders is a critical part of scientific progress and an antidote to the inertia of perpetual group-think. Depression, for example, is being reconceptualized as a disorder of impaired neuroplasticity and neurotropic deficiency, rather than a shortage of serotonin and norepinephrine. Rapid reversal of severe depression to euthymia—in 1 or 2 hours—with IV ketamine shattered the dogma that depression takes weeks to lift, and is ushering in unprecedented new thinking and models likely to revolutionize treatment of severe depression. We need such breakthroughs for other psychiatric brain disorders.
Unmet professional and sociopolitical needs
Broadening of training. Psychiatrists have focused on the mind but insufficiently attended to the biology of the brain. For psychiatry to rise to the next level as a medical specialty and brain discipline, training must incorporate more neurology than it does now. The converse is true in neurology.
Hospitalization not incarceration. It is unconscionable that people suffering from a medical illness that impairs their judgment and behavior are locked up as criminals. Psychiatry must forcefully lobby so that the seriously mentally ill are treated in secure hospitals staffed by physicians, nurses, and mental health professionals.
That’s right: Bring back the asylum to address this unmet medical, political, and ethical need for psychiatric patients.a A serious mental disorder must be accepted as a fault-free illness.
aTo read more about this, I recommend my March 2008 editorial, “Bring back the asylums?,” at CurrentPsychiatry.com, and Dr. George Paulson’s excellent book, Closing the asylums: Causes and consequences of the deinstitutionalization movement (Jefferson, NC: McFarland & Co. Inc; 2012).
Full integration of psychiatry into the rest of medicine remains an unmet need, despite good progress. Because almost every medical illness can cause psychiatric symptoms, DSM-5 mandates that general medical conditions be ruled out before a primary psychiatric diagnosis is made.
Along the same lines, most severely mentally ill persons suffer from medical and neurologic ailments before their first episode,5 and many die prematurely from cardiovascular causes that often are the result of unhealthy lifestyle; iatrogenic complications; and lack of primary care interventions.6 Psychiatric patients must always receive standard general medical evaluation and management, side by side with their psychiatric care.
Philanthropy for psychiatry
Philanthropic support of psychiatry is a salutary trend. Some unmet needs in psychiatry, however, require not only money but a change in attitude (such as eliminating the absurd and discriminatory stigma of mental illness), better training, and forceful political activism by all of us.
Recent examples come quickly to mind:
• $100 million from the Lieber family to fund the Lieber Institute for Brain Development at Johns Hopkins University
• $300 million from Microsoft co-founder Paul G. Allen to create the Allen Institute for Brain Science
• $650 million from Ted Stanley for the Stanley Center at the Broad Institute.
Such generosity is cause for celebration by psychiatrists and their long-suffering patients who are disabled by a brain disorder. Private money supplements research funding by the National Institutes of Health and will bolster the war against mental illness,1 which costs >$300 billion annually (Box,2page 12).
Although philanthropy will help, many needs in psychiatry are unmet, and not all can be addressed with money. Consider a number of areas of need.
Unmet clinical needs
Models of disease. Psychiatry is in desperate need of an objective, valid diagnostic system that transcends the DSM model of symptom clusters. The Research Domain Criteria3 represents the effort to find an alternative. To achieve that goal, it’s necessary to identify biomarkers and establish their utility—a task that requires a huge amount of funding.
Therapeutics. Clinicians are hungry for innovative, safe pharmaceuticals and non-drug treatments that modify disease, not just alleviate symptoms. Obsessive-compulsive disorder always has lacked such therapies; so have dementia, schizophrenia, personality disorders, dissociative disorders, and sexual pathologies. In fact, >80% of DSM disorders do not have an FDA-approved, evidence-based treatment,4 and many available medications are only partially efficacious, poorly tolerated, or unsafe.
There are more unmet needs in therapeutics:
• Development of promising non-drug therapies, such as neuromodulation, proceeds slowly.
• Research into neurobiological mechanisms of psychotherapy is in its infancy.
• A foolproof method to monitor adherence does not exist, and countless patients relapse needlessly and deteriorate functionally.
• Effective, evidence-based rehabilitation for serious psychiatric disorders is used narrowly and vastly underfunded.
• Insurers continue to thumb their nose at laws that require parity for treating mental illness—thus impeding access to, delaying, or truncating psychiatric care.
Unmet scientific needs
Translational investigators. Despite increased funding for basic neuroscientific study and breathtaking discoveries in animal molecular neurobiology, a trickle of findings has been applied to clinical medicine. This translational gap has many causes, including a shortage of translational neuroscientists (MD-PhD psychiatrists and neurologists), insufficient long-term funding to develop such clinician-researchers, and complex regulatory oversight of human research.
Stalled progress in drug development. Development of novel-mechanism therapeutics for brain disorders is languishing. Some pharmaceutical manufacturers have abandoned the development of drugs that act on the CNS in favor of less complex, more lucrative areas such as oncology and cardiology; others have reduced their investment in CNS products. Developing treatments for knotty disorders of the most complex structure in the known universe requires mammoth investment. Why are stakeholders bailing out on the greatest challenge for science and medicine for easier endeavors?
Discovering new genes for every devastating neuropsychiatric syndrome, such as schizophrenia, is cause for celebration, but the champagne won’t flow until the coding of every gene is unscrambled so that specific biological interventions can be developed. The cost of the chase might be orders of magnitude greater than what is invested in research today. Conceptualizing new models of brain disorders is a critical part of scientific progress and an antidote to the inertia of perpetual group-think. Depression, for example, is being reconceptualized as a disorder of impaired neuroplasticity and neurotropic deficiency, rather than a shortage of serotonin and norepinephrine. Rapid reversal of severe depression to euthymia—in 1 or 2 hours—with IV ketamine shattered the dogma that depression takes weeks to lift, and is ushering in unprecedented new thinking and models likely to revolutionize treatment of severe depression. We need such breakthroughs for other psychiatric brain disorders.
Unmet professional and sociopolitical needs
Broadening of training. Psychiatrists have focused on the mind but insufficiently attended to the biology of the brain. For psychiatry to rise to the next level as a medical specialty and brain discipline, training must incorporate more neurology than it does now. The converse is true in neurology.
Hospitalization not incarceration. It is unconscionable that people suffering from a medical illness that impairs their judgment and behavior are locked up as criminals. Psychiatry must forcefully lobby so that the seriously mentally ill are treated in secure hospitals staffed by physicians, nurses, and mental health professionals.
That’s right: Bring back the asylum to address this unmet medical, political, and ethical need for psychiatric patients.a A serious mental disorder must be accepted as a fault-free illness.
aTo read more about this, I recommend my March 2008 editorial, “Bring back the asylums?,” at CurrentPsychiatry.com, and Dr. George Paulson’s excellent book, Closing the asylums: Causes and consequences of the deinstitutionalization movement (Jefferson, NC: McFarland & Co. Inc; 2012).
Full integration of psychiatry into the rest of medicine remains an unmet need, despite good progress. Because almost every medical illness can cause psychiatric symptoms, DSM-5 mandates that general medical conditions be ruled out before a primary psychiatric diagnosis is made.
Along the same lines, most severely mentally ill persons suffer from medical and neurologic ailments before their first episode,5 and many die prematurely from cardiovascular causes that often are the result of unhealthy lifestyle; iatrogenic complications; and lack of primary care interventions.6 Psychiatric patients must always receive standard general medical evaluation and management, side by side with their psychiatric care.
Philanthropy for psychiatry
Philanthropic support of psychiatry is a salutary trend. Some unmet needs in psychiatry, however, require not only money but a change in attitude (such as eliminating the absurd and discriminatory stigma of mental illness), better training, and forceful political activism by all of us.
1. Licinio J, Wong ML. Launching the ‘war on mental illness’. Mol Psychiatry. 2014;19(1):1-5.
2. Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421-427.
3. Devulapalli KK, Nasrallah HA. An analysis of the high psychotropic off-label use in psychiatric disorders. The majority of psychiatric diagnoses have no approved drug. Asian J Psychiatr. 2009;2(1):29-36.
4. Cuthbert BN. The RDoC framework: facilitating transition from ICD/OSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry. 2014;13(1):28-35.
5. Sørensen HJ, Nielsen PR, Benros ME, et al. Somatic diseases and conditions before the first diagnosis of schizophrenia: a nationwide population-based cohort study in more than 900000 individuals [published online July 25, 2014]. Schizophr Bull. doi: 10.1093/schbul/sbu110.
6. Nasrallah HA, Meyer JM, Goff DC, et al. Low rates of treatment for hypertension, dyslipidemia and diabetes in schizophrenia: data from the CATIE schizophrenia trial sample at baseline. Schizophr Res. 2006;86(1-3):15-22.
1. Licinio J, Wong ML. Launching the ‘war on mental illness’. Mol Psychiatry. 2014;19(1):1-5.
2. Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421-427.
3. Devulapalli KK, Nasrallah HA. An analysis of the high psychotropic off-label use in psychiatric disorders. The majority of psychiatric diagnoses have no approved drug. Asian J Psychiatr. 2009;2(1):29-36.
4. Cuthbert BN. The RDoC framework: facilitating transition from ICD/OSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry. 2014;13(1):28-35.
5. Sørensen HJ, Nielsen PR, Benros ME, et al. Somatic diseases and conditions before the first diagnosis of schizophrenia: a nationwide population-based cohort study in more than 900000 individuals [published online July 25, 2014]. Schizophr Bull. doi: 10.1093/schbul/sbu110.
6. Nasrallah HA, Meyer JM, Goff DC, et al. Low rates of treatment for hypertension, dyslipidemia and diabetes in schizophrenia: data from the CATIE schizophrenia trial sample at baseline. Schizophr Res. 2006;86(1-3):15-22.
Many eyes are better
Peer review is the filter that determines what is published in the scientific literature and what is not allowed to see the light of day. It has been considered "the gatekeeper of science." In modern times, peer review has consisted of a journal editor sending a submitted article to a limited number of experts in the field who then judge its worth for publication. Many specialists see reviewing manuscripts as a service to their profession, put considerable effort into their analysis, and provide extensive comments on revisions needed to strengthen papers.
However, the value of this venerable process as it is presently constituted has been questioned by numerous critics. Because manuscripts are reviewed by a limited number of often rival scientists in a highly specialized field, the practice is prone to bias. Innovation may be stifled when reviewers reject outlier concepts that may be correct but that do not fit into the mainstream of thought. Critiques are often superficial as they are performed by otherwise busy individuals who receive no compensation for their efforts. Finally, journal editors are given undue power in the process. Not only do they make the final decision to publish or reject a manuscript, they are also responsible for selecting reviewers and are then free to ignore or accept their recommendations.
These criticisms of this time-honored system have led to a strong impetus for change. Coincident with the motivation for modifying this essential component of the publication process have been technological advancements that are facilitating new approaches. The Internet has provided a mechanism for making peer review a more open, inclusive process with any member of the scientific community who so desires having the opportunity to contribute to the evaluation of published work. A recent incident highlights how uninvited, but valuable, input from the wider scientific community can rapidly and effectively improve the accuracy of the literature.
On July 2, 2014, National Public Radio’s Morning Edition broadcast "Easy method for making stem cells was too good to be true" and a New York Times headline proclaimed "Stem cell research papers are retracted." In January 2014, Haruko Obokata of the RIKEN Centre for Developmental Biology in Japan published an innovative and considerably simpler method of producing stem cells than extracting them from embryos or making them from skin cells in a complicated and prolonged process (Nature 2014;505: 641-7). At the time of publication, the work was viewed by many to be potentially Nobel Prize-worthy research—that is, until the stem cell research community chimed in with its extensive and detailed post-publication peer review.
A number of research groups questioned Obokata’s conclusions. Some even attempted to replicate the experiments, but with no success. Soon the critics’ findings and opinions appeared on a variety of websites and blogs, including the Nature website. The RIKEN Centre took notice and appointed a committee to investigate the research. The committee found that Obokata had manipulated her data on at least two occasions and concluded that she had participated in research misconduct. Pressure mounted, which led to the recent voluntary retraction of the article by its authors.
This case represents an extreme outcome resulting from a failure of pre-publication vetting followed by a successful post-hoc peer review. But it demonstrates how the emerging and more comprehensive means of evaluating published research is rapidly working its way into the fabric of how science and the reporting of it operate. It would be ideal if this extensive vetting of potentially important research could be done prior to rather than after its release to the general public. Physics academicians have accomplished this by posting their research papers as pre-prints on-line for their colleagues to evaluate. Only after a successful conclusion of this process is a work deemed acceptable for entry into the physics literature. Submission of biomedical research to a similar process has a significant downside in that new, possibly harmful therapies, not yet peer-reviewed, could be adopted by practitioners and/or patients before their time. However, some modification of it will likely evolve and lead to a more accurate assessment of submitted work than the present process allows.
How publications are valued is also being modified thanks to the omnibus means of rapid communication allowed by an ever-expanding Internet. Bibliometrics, most notably the number of times an article is subsequently cited in print, has been the mainstay in determining the value of individual articles. The journal impact factor, which has historically been the main measure of a journal\'s standing compared to that of others in its field, is derived from the aggregate of citations for all articles over a period of time. With the advent of the Internet, a new set of alternative metrics (altmetrics) is now contributing to the evaluation of published work. While print citations take years to accumulate, article downloads, mentions on Facebook, number of tweets on Twitter, and numerous other altmetrics have the considerable advantage of immediacy and can be logged by any reader, not only those authors who decide to subsequently cite certain publications. Although these new metrics are unlikely to replace traditional citations in assigning value to individual articles, along with them, they will be helpful in determining what must be read to maintain currency in one’s specialty.
So, readers of the surgical literature feel your newly found power and exert it. You along with your colleagues around the globe can, and in fact have an obligation to, play a role in determining what is worth reading from an ever-expanding volume of new information. Many eyes will almost certainly be better than relying solely on the opinions of a chosen few.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
Peer review is the filter that determines what is published in the scientific literature and what is not allowed to see the light of day. It has been considered "the gatekeeper of science." In modern times, peer review has consisted of a journal editor sending a submitted article to a limited number of experts in the field who then judge its worth for publication. Many specialists see reviewing manuscripts as a service to their profession, put considerable effort into their analysis, and provide extensive comments on revisions needed to strengthen papers.
However, the value of this venerable process as it is presently constituted has been questioned by numerous critics. Because manuscripts are reviewed by a limited number of often rival scientists in a highly specialized field, the practice is prone to bias. Innovation may be stifled when reviewers reject outlier concepts that may be correct but that do not fit into the mainstream of thought. Critiques are often superficial as they are performed by otherwise busy individuals who receive no compensation for their efforts. Finally, journal editors are given undue power in the process. Not only do they make the final decision to publish or reject a manuscript, they are also responsible for selecting reviewers and are then free to ignore or accept their recommendations.
These criticisms of this time-honored system have led to a strong impetus for change. Coincident with the motivation for modifying this essential component of the publication process have been technological advancements that are facilitating new approaches. The Internet has provided a mechanism for making peer review a more open, inclusive process with any member of the scientific community who so desires having the opportunity to contribute to the evaluation of published work. A recent incident highlights how uninvited, but valuable, input from the wider scientific community can rapidly and effectively improve the accuracy of the literature.
On July 2, 2014, National Public Radio’s Morning Edition broadcast "Easy method for making stem cells was too good to be true" and a New York Times headline proclaimed "Stem cell research papers are retracted." In January 2014, Haruko Obokata of the RIKEN Centre for Developmental Biology in Japan published an innovative and considerably simpler method of producing stem cells than extracting them from embryos or making them from skin cells in a complicated and prolonged process (Nature 2014;505: 641-7). At the time of publication, the work was viewed by many to be potentially Nobel Prize-worthy research—that is, until the stem cell research community chimed in with its extensive and detailed post-publication peer review.
A number of research groups questioned Obokata’s conclusions. Some even attempted to replicate the experiments, but with no success. Soon the critics’ findings and opinions appeared on a variety of websites and blogs, including the Nature website. The RIKEN Centre took notice and appointed a committee to investigate the research. The committee found that Obokata had manipulated her data on at least two occasions and concluded that she had participated in research misconduct. Pressure mounted, which led to the recent voluntary retraction of the article by its authors.
This case represents an extreme outcome resulting from a failure of pre-publication vetting followed by a successful post-hoc peer review. But it demonstrates how the emerging and more comprehensive means of evaluating published research is rapidly working its way into the fabric of how science and the reporting of it operate. It would be ideal if this extensive vetting of potentially important research could be done prior to rather than after its release to the general public. Physics academicians have accomplished this by posting their research papers as pre-prints on-line for their colleagues to evaluate. Only after a successful conclusion of this process is a work deemed acceptable for entry into the physics literature. Submission of biomedical research to a similar process has a significant downside in that new, possibly harmful therapies, not yet peer-reviewed, could be adopted by practitioners and/or patients before their time. However, some modification of it will likely evolve and lead to a more accurate assessment of submitted work than the present process allows.
How publications are valued is also being modified thanks to the omnibus means of rapid communication allowed by an ever-expanding Internet. Bibliometrics, most notably the number of times an article is subsequently cited in print, has been the mainstay in determining the value of individual articles. The journal impact factor, which has historically been the main measure of a journal\'s standing compared to that of others in its field, is derived from the aggregate of citations for all articles over a period of time. With the advent of the Internet, a new set of alternative metrics (altmetrics) is now contributing to the evaluation of published work. While print citations take years to accumulate, article downloads, mentions on Facebook, number of tweets on Twitter, and numerous other altmetrics have the considerable advantage of immediacy and can be logged by any reader, not only those authors who decide to subsequently cite certain publications. Although these new metrics are unlikely to replace traditional citations in assigning value to individual articles, along with them, they will be helpful in determining what must be read to maintain currency in one’s specialty.
So, readers of the surgical literature feel your newly found power and exert it. You along with your colleagues around the globe can, and in fact have an obligation to, play a role in determining what is worth reading from an ever-expanding volume of new information. Many eyes will almost certainly be better than relying solely on the opinions of a chosen few.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
Peer review is the filter that determines what is published in the scientific literature and what is not allowed to see the light of day. It has been considered "the gatekeeper of science." In modern times, peer review has consisted of a journal editor sending a submitted article to a limited number of experts in the field who then judge its worth for publication. Many specialists see reviewing manuscripts as a service to their profession, put considerable effort into their analysis, and provide extensive comments on revisions needed to strengthen papers.
However, the value of this venerable process as it is presently constituted has been questioned by numerous critics. Because manuscripts are reviewed by a limited number of often rival scientists in a highly specialized field, the practice is prone to bias. Innovation may be stifled when reviewers reject outlier concepts that may be correct but that do not fit into the mainstream of thought. Critiques are often superficial as they are performed by otherwise busy individuals who receive no compensation for their efforts. Finally, journal editors are given undue power in the process. Not only do they make the final decision to publish or reject a manuscript, they are also responsible for selecting reviewers and are then free to ignore or accept their recommendations.
These criticisms of this time-honored system have led to a strong impetus for change. Coincident with the motivation for modifying this essential component of the publication process have been technological advancements that are facilitating new approaches. The Internet has provided a mechanism for making peer review a more open, inclusive process with any member of the scientific community who so desires having the opportunity to contribute to the evaluation of published work. A recent incident highlights how uninvited, but valuable, input from the wider scientific community can rapidly and effectively improve the accuracy of the literature.
On July 2, 2014, National Public Radio’s Morning Edition broadcast "Easy method for making stem cells was too good to be true" and a New York Times headline proclaimed "Stem cell research papers are retracted." In January 2014, Haruko Obokata of the RIKEN Centre for Developmental Biology in Japan published an innovative and considerably simpler method of producing stem cells than extracting them from embryos or making them from skin cells in a complicated and prolonged process (Nature 2014;505: 641-7). At the time of publication, the work was viewed by many to be potentially Nobel Prize-worthy research—that is, until the stem cell research community chimed in with its extensive and detailed post-publication peer review.
A number of research groups questioned Obokata’s conclusions. Some even attempted to replicate the experiments, but with no success. Soon the critics’ findings and opinions appeared on a variety of websites and blogs, including the Nature website. The RIKEN Centre took notice and appointed a committee to investigate the research. The committee found that Obokata had manipulated her data on at least two occasions and concluded that she had participated in research misconduct. Pressure mounted, which led to the recent voluntary retraction of the article by its authors.
This case represents an extreme outcome resulting from a failure of pre-publication vetting followed by a successful post-hoc peer review. But it demonstrates how the emerging and more comprehensive means of evaluating published research is rapidly working its way into the fabric of how science and the reporting of it operate. It would be ideal if this extensive vetting of potentially important research could be done prior to rather than after its release to the general public. Physics academicians have accomplished this by posting their research papers as pre-prints on-line for their colleagues to evaluate. Only after a successful conclusion of this process is a work deemed acceptable for entry into the physics literature. Submission of biomedical research to a similar process has a significant downside in that new, possibly harmful therapies, not yet peer-reviewed, could be adopted by practitioners and/or patients before their time. However, some modification of it will likely evolve and lead to a more accurate assessment of submitted work than the present process allows.
How publications are valued is also being modified thanks to the omnibus means of rapid communication allowed by an ever-expanding Internet. Bibliometrics, most notably the number of times an article is subsequently cited in print, has been the mainstay in determining the value of individual articles. The journal impact factor, which has historically been the main measure of a journal\'s standing compared to that of others in its field, is derived from the aggregate of citations for all articles over a period of time. With the advent of the Internet, a new set of alternative metrics (altmetrics) is now contributing to the evaluation of published work. While print citations take years to accumulate, article downloads, mentions on Facebook, number of tweets on Twitter, and numerous other altmetrics have the considerable advantage of immediacy and can be logged by any reader, not only those authors who decide to subsequently cite certain publications. Although these new metrics are unlikely to replace traditional citations in assigning value to individual articles, along with them, they will be helpful in determining what must be read to maintain currency in one’s specialty.
So, readers of the surgical literature feel your newly found power and exert it. You along with your colleagues around the globe can, and in fact have an obligation to, play a role in determining what is worth reading from an ever-expanding volume of new information. Many eyes will almost certainly be better than relying solely on the opinions of a chosen few.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
When snoring is more than an annoyance
We have all seen cartoons of an unhappy wife awake in bed next to her loudly snoring husband. Casual conversations with friends, particularly female ones, indicate that this is an accurate representation of a common scenario. As I have started to probe more diligently for evidence of obstructive sleep apnea (OSA) in my patients, not just in those who complain of “fatigue” (more patients use this term with me than “sleepiness”), I see a lot of shaking of heads from the wives of men who deny that they snore or have disrupted sleep. I am not implying that this is solely a male disease. Far from it. But as in other medical scenarios, the Y chromosome seems somehow linked to denial or lack of awareness of symptoms. In any event, I was not a bit surprised to read in the review by Dr. Mehra in this issue of the Journal that 17% of adults may have OSA.
As awareness of OSA has grown and testing for it has become easier, multiple reports have documented associated comorbidities: hypertension, restless leg syndrome, gout, and neurocognitive deficits. Home devices to treat OSA have significantly improved. Technological advances have led to the development of small, quiet, smart pumps that provide continuous positive airway pressure (CPAP) via nasal or relatively comfortable full-face masks. Compliance and patient acceptance of CPAP have improved, although patient education and a bit of cajoling in the office are still necessary—less so if the bedroom partner is also present for this discussion.
Perhaps surprising is a growing pool of data showing that CPAP’s benefits extend to more than just reducing sleepiness. It can reduce nocturia, restless leg syndrome, arrhythmias including atrial fibrillation, gastric reflux, and fatal and nonfatal cardiovascular events. Snoring and thus probably sleep-partner satisfaction are also improved.
Several physiologic mechanisms may explain the benefits of CPAP, including reducing hypoxic episodes (explaining its effect on atrial fibrillation), altered atrial natriuretic factor levels (thus reducing nocturia), and changing intrathoracic pressure (thus reducing gastric reflux). It will be interesting to see if there are long-term effects of successfully applied CPAP on neurocognition and progression of neurodegenerative diseases.
While high-decibel snoring and snorting are not present in all patients with OSA, it is quite clear now that they represent far more than an annoyance. We should be vigilant about looking for OSA and strongly encourage a trial of CPAP in appropriately diagnosed patients.
We have all seen cartoons of an unhappy wife awake in bed next to her loudly snoring husband. Casual conversations with friends, particularly female ones, indicate that this is an accurate representation of a common scenario. As I have started to probe more diligently for evidence of obstructive sleep apnea (OSA) in my patients, not just in those who complain of “fatigue” (more patients use this term with me than “sleepiness”), I see a lot of shaking of heads from the wives of men who deny that they snore or have disrupted sleep. I am not implying that this is solely a male disease. Far from it. But as in other medical scenarios, the Y chromosome seems somehow linked to denial or lack of awareness of symptoms. In any event, I was not a bit surprised to read in the review by Dr. Mehra in this issue of the Journal that 17% of adults may have OSA.
As awareness of OSA has grown and testing for it has become easier, multiple reports have documented associated comorbidities: hypertension, restless leg syndrome, gout, and neurocognitive deficits. Home devices to treat OSA have significantly improved. Technological advances have led to the development of small, quiet, smart pumps that provide continuous positive airway pressure (CPAP) via nasal or relatively comfortable full-face masks. Compliance and patient acceptance of CPAP have improved, although patient education and a bit of cajoling in the office are still necessary—less so if the bedroom partner is also present for this discussion.
Perhaps surprising is a growing pool of data showing that CPAP’s benefits extend to more than just reducing sleepiness. It can reduce nocturia, restless leg syndrome, arrhythmias including atrial fibrillation, gastric reflux, and fatal and nonfatal cardiovascular events. Snoring and thus probably sleep-partner satisfaction are also improved.
Several physiologic mechanisms may explain the benefits of CPAP, including reducing hypoxic episodes (explaining its effect on atrial fibrillation), altered atrial natriuretic factor levels (thus reducing nocturia), and changing intrathoracic pressure (thus reducing gastric reflux). It will be interesting to see if there are long-term effects of successfully applied CPAP on neurocognition and progression of neurodegenerative diseases.
While high-decibel snoring and snorting are not present in all patients with OSA, it is quite clear now that they represent far more than an annoyance. We should be vigilant about looking for OSA and strongly encourage a trial of CPAP in appropriately diagnosed patients.
We have all seen cartoons of an unhappy wife awake in bed next to her loudly snoring husband. Casual conversations with friends, particularly female ones, indicate that this is an accurate representation of a common scenario. As I have started to probe more diligently for evidence of obstructive sleep apnea (OSA) in my patients, not just in those who complain of “fatigue” (more patients use this term with me than “sleepiness”), I see a lot of shaking of heads from the wives of men who deny that they snore or have disrupted sleep. I am not implying that this is solely a male disease. Far from it. But as in other medical scenarios, the Y chromosome seems somehow linked to denial or lack of awareness of symptoms. In any event, I was not a bit surprised to read in the review by Dr. Mehra in this issue of the Journal that 17% of adults may have OSA.
As awareness of OSA has grown and testing for it has become easier, multiple reports have documented associated comorbidities: hypertension, restless leg syndrome, gout, and neurocognitive deficits. Home devices to treat OSA have significantly improved. Technological advances have led to the development of small, quiet, smart pumps that provide continuous positive airway pressure (CPAP) via nasal or relatively comfortable full-face masks. Compliance and patient acceptance of CPAP have improved, although patient education and a bit of cajoling in the office are still necessary—less so if the bedroom partner is also present for this discussion.
Perhaps surprising is a growing pool of data showing that CPAP’s benefits extend to more than just reducing sleepiness. It can reduce nocturia, restless leg syndrome, arrhythmias including atrial fibrillation, gastric reflux, and fatal and nonfatal cardiovascular events. Snoring and thus probably sleep-partner satisfaction are also improved.
Several physiologic mechanisms may explain the benefits of CPAP, including reducing hypoxic episodes (explaining its effect on atrial fibrillation), altered atrial natriuretic factor levels (thus reducing nocturia), and changing intrathoracic pressure (thus reducing gastric reflux). It will be interesting to see if there are long-term effects of successfully applied CPAP on neurocognition and progression of neurodegenerative diseases.
While high-decibel snoring and snorting are not present in all patients with OSA, it is quite clear now that they represent far more than an annoyance. We should be vigilant about looking for OSA and strongly encourage a trial of CPAP in appropriately diagnosed patients.