User login
Hepatitis B infection in pregnancy: Essentials of antiviral therapy and immunoprophylaxis
Hepatitis B is one of the more common infections encountered in the daily practice of obstetrics. It is responsible for 40% to 45% of all cases of viral hepatitis.1,2 Hepatitis B may cause serious complications in both the infected mother and neonate.
In this article, I review the virology, epidemiology, and clinical presentation of hepatitis B and then discuss the key diagnostic tests and, subsequently, the clinical management for both the mother and neonate. I focus particular attention on relatively new information about the value of specific antiviral medication to enhance the protective effect of conventional neonatal immunoprophylaxis.
To set the framework for the discussion, consider the following 2 case studies.
CASE 1 Undetectable level of hepatitis B surface antibody in a pregnant woman
A 25-year-old healthy primigravid woman at 10 weeks’ gestation had a series of laboratory studies that included a test for hepatitis B surface antigen (HBsAg) and hepatitis B surface antibody (HBsAb). The test for the surface antigen was negative. The test for the surface antibody was below the level of detection. Upon questioning, the patient indicates that she received the 3-dose hepatitis B vaccine when she was age 13 years.
- What treatment, if any, is indicated for this patient?
- What treatment is indicated for her neonate?
CASE 2 Pregnant woman tests positive for hepatitis B surface antigen
A 31-year-old woman (G3P2002) at 12 weeks’ gestation tested positive for HBsAg. She indicates that she never has had symptomatic hepatitis and that she considers herself to be in excellent health.
- What additional laboratory tests are indicated at this time?
- What additional laboratory test should be performed at the end of the second trimester?
- What treatment is indicated for the mother and neonate?
Virology and epidemiology of hepatitis B
Hepatitis B is caused by a double-stranded, enveloped DNA virus. The virus has 10 genotypes and 24 subtypes.3 The organism contains 3 major antigens. Detection of these antigens and their corresponding antibodies is an essential step in the diagnostic workup of patients who may be infected.
The surface antigen (HBsAg) confers infectivity and is the most valuable serologic marker of infection. The e antigen (HBeAg) is not present in every infected patient. It is secreted from infected cells, but it is not incorporated into the viral particle. When present, it denotes a high level of viral replication and exceptionally high infectivity. The core antigen (HBcAg) is a valuable serologic marker for distinguishing between acute and chronic infection.1-3
Hepatitis B is highly infectious, much more so than HIV or hepatitis C. The virus has an incubation period of 4 weeks to 6 months, and the duration of incubation is inversely related to the size of the viral inoculum. The virus is transmitted in 3 principal ways: sexual contact with contaminated genital tract secretions, contact with infected blood from sharing contaminated drug-injecting paraphernalia or from receiving a blood transfusion (extremely rare today), and transmission from an infected mother to her neonate. Perinatal transmission occurs primarily during the delivery process as opposed to transplacental infection. Transmission also can occur by more casual household contact, such as sharing eating utensils, kissing, and handling an infant.1,2,4,5
Worldwide, more than 400 million people have chronic hepatitis B infection. In the United States, approximately 1.25 to 1.5 million individuals are infected. Several groups are at particularly high risk for being infected, including1-3:
- Asians
- Alaska Natives
- sub-Saharan Africans
- sex workers
- intravenous drug users
- individuals with hemophilia
- international travelers
- staff and residents of long-term care facilities
- tattoo recipients.
Continue to: Clinical presentation...
Clinical presentation
Approximately 90% of adult patients who contract hepatitis B, either symptomatically or asymptomatically, will develop protective levels of antibody and clear the virus from their system. They will then have lifelong immunity to reinfection. Approximately 10% of patients will fail to develop protective levels of antibody and will become chronically infected, posing a risk to their household members, sexual contacts, and their fetus if they become pregnant. Persistence of the surface antigen in the patient’s serum for more than 6 months denotes chronic infection. A very small number of individuals—less than 1%—will develop acute liver failure and experience a fatal outcome.1-3,5
In the United States, the prevalence of acute hepatitis B in pregnancy is 1 to 2 per 1,000. Clinical manifestations typically include anorexia, nausea, low-grade fever, right upper quadrant pain and tenderness, passage of clay-colored stools, and jaundice.
The prevalence of chronic infection in pregnancy is significantly higher, approximately 5 to 15 per 1,000. Over the long term, patients with chronic infection are at risk for progressive liver injury, including cirrhosis and even hepatocellular carcinoma. These serious sequelae are particularly likely to occur when the patient is co-infected with hepatitis C, D, or both. The overall risk of progression to chronic cirrhosis is approximately 15% to 30%. In patients who progress to cirrhosis, the annual incidence of hepatocellular carcinoma is 10%.1-3
Diagnosis of hepatitis B infection
Patients with acute hepatitis B will test positive for HBsAg and immunoglobulin M (IgM) antibody to the core antigen. Some patients will also test positive for HBeAg. Assessment of the patient’s serum by polymerase chain reaction (PCR) allows quantitation of the viral load, which often is expressed as viral copies per milliliter. Alternatively, the quantitative hepatitis B DNA concentration may be expressed as international units per milliliter (IU/mL). The World Health Organization recommends this latter quantitative method. Multiplying the DNA in IU/mL by 5.6 provides the conversion to viral copies per milliliter.
Patients with chronic hepatitis B infection will test positive for the HBsAg and for immunoglobulin G (IgG) antibody to the core antigen. They may also have a positive test for the HBeAg, and PCR may be used to quantify the viral load.1-3
Managing hepatitis B infection in pregnancy
General supportive measures. All pregnant patients should be tested for the HBsAg and HBsAb at the time of the first prenatal appointment. The tests should be repeated at the beginning of the third trimester in high-risk patients. Seropositive patients should have a hepatitis B genotype, a test for the e antigen, and tests for other sexually transmissible infections (gonorrhea, chlamydia, syphilis, HIV) and for hepatitis C and D. Liver function tests should be performed to assess for elevations in the alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels. Patients with elevated transaminase enzymes should have a coagulation profile to be certain they are not at risk for a coagulopathy.
At the end of the second trimester, patients should have a PCR assessment to determine the viral load. This assessment will be important for deciding if specific antiviral therapy is indicated during the third trimester to enhance the effects of neonatal immunoprophylaxis (see below). Of note, patients who are positive for the e antigen may have a very high viral load and yet have normal or near-normal transaminase levels. This seemingly paradoxical finding reflects the non-cytopathic nature of hepatitis B.
The patient should optimize her nutrition and sleep. She should avoid, or at least minimize, medications such as acetaminophen that could cause further liver injury. Without question, she should refrain from consuming even small amounts of alcohol. She should be tested for immunity to hepatitis A; if found to be susceptible, she should be vaccinated with the hepatitis A vaccine. This agent is an inactivated vaccine and is safe for administration at any time in pregnancy.1,2,5
Household contacts. In addition to the measures outlined above, the patient’s household contacts, particularly her sexual partner(s), should be tested for immunity to hepatitis B. If they do not have immunity by virtue of natural infection or previous vaccination, they should receive the hepatitis B vaccine series. It is also prudent to provide the sexual partner(s) with an initial dose of hepatitis B immune globulin (HBIG) to provide a temporary level of passive immunity.
Postdelivery care. After delivery, the patient should be referred to an infectious disease specialist or hepatologist for consideration of long-term treatment with antiviral agents, such as interferon alfa, pegylated interferon alfa, lamivudine, adefovir, entecavir, telbivudine, or tenofovir.6 The principal candidates for treatment are those who have cirrhosis and detectable levels of hepatitis B DNA. The ultimate goal of treatment is to reduce the serum hepatitis B DNA concentration to an undetectable level. Once the surface antigenemia is cleared, treatment can be stopped. A cure is defined when the absence of hepa-titis B DNA in the serum is sustained.
- Hepatitis B is a DNA virus that is transmitted via sexual contact, exposure to infected blood, and from an infected mother to her fetus.
- Most patients in our practice will most likely have chronic, asymptomatic infection, and the diagnosis will be established by detection of HBsAg in the patient’s serum.
- All obstetric patients should be tested for both HBsAg and HBsAb.
- Patients who are positive for the surface antigen should be tested for HIV infection and hepatitis C and D. They also should have a determination of the hepatitis B genotype and viral load and assessment of liver function (ALT, AST).
- Patients who are chronically infected with hepatitis B should be vaccinated against hepatitis A to prevent further liver injury. They also should avoid medications that might cause hepatic injury.
- Patients who have a viral DNA concentration greater than 200,000 IU/mL or a viral load greater than 1,120,000 million copies/mL should be treated with tenofovir, 300 mg daily, from week 28 until 4 to 8 weeks after delivery.
- Infants delivered to infected mothers should receive HBIG within 12 hours of birth and then begin the 3-dose hepatitis B vaccine series. The first dose should be administered prior to hospital discharge.
- Infants delivered to mothers who are negative for the surface antigen should begin the hepatitis B vaccine series prior to discharge from the hospital.
- Mothers who test negative for HBsAb should be questioned about prior vaccination. If they have never been vaccinated, they should receive the 3-dose vaccine series. If they have been vaccinated, they should receive a single hepatitis B vaccine booster. The vaccine is safe for administration at any time during pregnancy.
- Infected mothers may breastfeed as long as they do not have cracked or bleeding nipples or exudative skin lesions near the nipple(s).
Neonatal immunoprophylaxis
The Centers for Disease Control and Prevention recommends universal hepatitis B vaccination for all newborns. The first dose of the vaccine should be administered prior to hospital discharge. The second and third doses should be administered 1 and 6 months later.1,2,5 There are few, if any, medical contraindications to neonatal vaccination. For the vast majority of infants, the immunity induced by vaccination is lifelong. For a small number, immunity may wane over time. Thus, reassessment of the HBsAb concentration is indicated in selected situations, for example, acute high-risk exposure to an infected person, development of an immunosuppressive disorder, or pregnancy.
Infants delivered to mothers who are infected with hepatitis B also should receive HBIG in addition to the vaccine. HBIG provides passive immunization to counteract the high viral inoculum encountered by the neonate during delivery. This preparation should be administered within 12 hours of birth.1,2,5
In the absence of immunoprophylaxis, a neonate delivered to a mother who is seropositive for HBsAg has a 20% to 30% probability of becoming chronically infected. If the mother is positive for both the surface antigen and the e antigen, the risk of chronic infection increases to almost 90%. Approximately 90% of infants who are infected in the perinatal period subsequently develop chronic infection. However, with appropriate immunoprophylaxis in the neonatal period, the risk of perinatal transmission is reduced by 85% to 95%.1,2,5
Cesarean delivery offers no additional protection beyond that provided by immunoprophylaxis. Moreover, because immunoprophylaxis is so effective, infected mothers may breastfeed without fear of transmitting infection to their infant. Shi and colleagues published a systematic review and meta-analysis of the risk associated with breastfeeding in hepatitis B–infected mothers.7 Infants who breastfed did not have a higher rate of mother-to-child transmission, regardless of whether they received combined immunoprophylaxis or only hepatitis B vaccine and regardless of whether the HBsAg was detected in the mother’s breast milk. The only precaution is the need to avoid breastfeeding if the nipples are cracked or bleeding or if exudative lesions are present on the skin near the nipple.
Continue to: Maternal antiviral therapy...
Maternal antiviral therapy
As noted above, neonatal immunoprophylaxis is 85% to 95% effective in preventing perinatal transmission of hepatitis B infection. Failures of prophylaxis are primarily due to antenatal transmission in patients who have exceptionally high viral loads. Several cutoffs have been used to define “high viral load,” including greater than 1 to 2 million copies/mL and a hepatitis B DNA concentration greater than 200,000 IU/mL. There is not a perfect consensus on the appropriate cutoff.
In essence, 2 different approaches have been tried to further reduce the risk of perinatal transmission in these high-risk patients.8 The first major initiative was administration of HBIG (100–200 IU) intramuscularly to the patient at 28, 32, and 36 weeks. The outcomes with this approach have been inconsistent, due, at least in part, to varying doses of the agent and various cutoffs for defining “high risk,” and this intervention is no longer recommended.1,2
The second major approach is administration of specific antiviral drugs to the mother during the third trimester. The first agent widely used in clinical practice was lamivudine. In a systematic review and meta-analysis, Shi and colleagues reported that, in infants whose mothers received lamivudine plus conventional neonatal immunuprophylaxis, the risk of perinatal infection was significantly reduced compared with infants who received only immunoprophylaxis.9
Although lamivudine is effective, there is considerable concern about the rapid development of viral resistance to the medication. Accordingly, most attention today is focused on the use of tenofovir to prevent perinatal transmission.
In an important early investigation, Pan and colleagues reported the results of a randomized controlled trial conducted in China in women with a hepatitis B DNA concentration greater than 200,000 IU/mL (viral load > 1,120,000 copies/mL).10 Patients also were positive for the e antigen. Ninety-two patients were assigned to tenofovir disoproxil fumarate (TDF), 300 mg daily, from 30 to 32 weeks until postpartum week 4 plus conventional neonatal immunoprophylaxis, and 100 patients were assigned to immunoprophylaxis alone. In the intention-to-treat analysis, 18 neonates in the control group were infected compared with 5 in the treatment group (P = .007). In the per-protocol analysis, 7 neonates in the control group were infected compared with 0 in the treatment group (P = .01). No clinically significant adverse maternal or neonatal effects occurred in the treatment group.
Subsequently, Jourdain and colleagues reported a multicenter, double-blind trial conducted in 17 public health hospitals in Thailand.11 TDF (300 mg daily) or placebo was administered from 28 weeks’ gestation until 8 weeks postpartum. Patients in both arms of the study were positive for the e antigen; 87% to 90% of the patients had a serum hepatitis B DNA concentration greater than 200,000 IU/mL.Following birth, infants in both groups received an injection of HBIG and then 4 doses of hepatitis B vaccine (0, 1, 2, 4, and 6 months). Both the HBIG and hepatitis B vaccine were administered very promptly after birth (median time, 1.2–1.3 hours).
At 6 months after delivery, 2% of infants in the placebo group (3 of 147) were HBsAg-positive compared with none of the infants in the treatment arm.11 No serious adverse effects occurred in infants in the TDF group. This difference in outcome was not statistically significant, but the overall rate of infection was so low in both groups that the sample size was definitely too small to exclude a type 2 statistical error. Moreover, the fourth dose of neonatal hepatitis B vaccine may have contributed to the surprisingly low rate of perinatal transmission. Of note, the serum hepatitis B DNA concentration in the TDF group declined from a mean of 7.6 log10 IU/mL to a mean of 4.0 log10 IU/mL at delivery.
In the most recent report, Wang and colleagues reported the results of a prospective cohort study in patients with a hepatitis B virus DNA concentration greater than 200,000 IU/mL.12 Beginning at either 24 or 32 weeks, patients were assigned to treatment with either oral TDF (300 mg daily) or oral telbivudine (LdT, 600 mg daily). The medications were continued for 4 weeks postpartum. In the intention-to-treat analysis, the rates of perinatal transmission were comparable, 1.5% versus 1.8%. In the per-protocol analysis, no infants in either group were infected. However, the predelivery decline in hepatitis Bvirus DNA concentration was greater in the TDF group. The ALT elevation rate was also lower in the TDF group. Patients in the LdT group had fewer problems with anorexia but more instances of arthralgia compared with those in the TDF group.
Based primarily on these 3 investigations, I recommend that all infected patients with a hepatitis B DNA concentration greater than 200,000 IU/mL or a viral load greater than 1,120,000 million copies/mL receive oral TDF, 300 mg daily, from 28 weeks until at least 4 to 8 weeks postpartum. The decision about duration of postpartum treatment should be made in consultation with an infectious disease specialist or hepatologist.
Case studies resolved
CASE 1 No protective level of surface antibody
This patient should promptly receive a single booster dose of the hepatitis B vaccine. The vaccine is an inactivated agent and is safe for administration at any time in pregnancy. Following delivery and prior to discharge from the hospital, the neonate should receive the first dose of the hepatitis B vaccine. A second dose should be administered 1 month later, and a third dose should be administered 6 months after the first dose.
CASE 2 Mother is seropositive for HBsAg
This patient should be tested immediately for HIV infection and hepatitis C and D. The hepatitis B viral genotype should be determined. She also should have a panel of liver function tests. If any of these tests are abnormal, a coagulation profile should be obtained to be certain that the patient is not at risk for a coagulopathy. Near the end of the second trimester, a hepatitis B viral load should be obtained. If the viral DNA concentration is greater than 200,000 IU/mLor a viral load greater than 1,120,000 million copies/mL, the patient should be treated with tenofovir, 300 mg daily, from week 28 until at least 4 weeks after delivery. The neonate should receive an injection of HBIG within 12 hours of birth and the first dose of the hepatitis B vaccine prior to discharge from the hospital. Two additional doses of the vaccine should be administered 1 and 6 months later. ●
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al, eds. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862.
- Bernstein HB, Lee MJ. Maternal and perinatal infection in pregnancy: viral. In: Landon MB, Galan HL, Jauniaux ERM, et al, eds. Gabbe’s Obstetrics. Normal and Problem Pregnancies. 8th ed. Elsevier; 2021;1092.
- Dusheiko G, Agarwal K, Maini MK. New approaches to chronic hepatitis B. N Engl J Med. 2023;388:55-69.
- Ma L, Alla NR, Li X, et al. Mother to child transmission of HBV: review of current clinical management and prevention strategies. Rev Med Virol. 2014; 24: 396-406.
- Society for Maternal-Fetal Medicine; Dionne-Odom J, Tita ATN, Silverman NS. SMFM consult: preventing vertical transmission of hepatitis B. Contemporary OB/GYN. September 22, 2015. Accessed August 21, 2023. https://www .contemporaryobgyn.net/view/smfm-consult-preventing -vertical-transmission-hepatitis-b
- Lok ASF. The maze of treatments for hepatitis B. N Engl J Med. 2005;352:2743-2746.
- Shi Z, Yang Y, Wang H, et al. Breastfeeding of newborns by mothers carrying hepatitis B virus: a meta-analysis and systematic review. Arch Pediatr Adolesc Med. 2011;165:837-846.
- Dusheiko G. A shift in thinking to reduce mother-to-infant transmission of hepatitis B. N Engl J Med. 2018;378:952-953.
- Shi Z, Yang Y, Ma L, et al. Lamivudine in late pregnancy to interrupt in utero transmission of hepatitis B virus: a systematic review and meta-analysis. Obstet Gynecol. 2010;116:147-159.
- Pan C, Duan Z, Dai E, et al; China Study Group for the Motherto-Child Transmission of Hepatitis B. Tenofovir to prevent hepatitis B transmission in mothers with high viral load. N Engl J Med. 2016;374:2324-2334.
- Jourdain G, Ngo-Giang-Huong N, Harrison L, et al. Tenofovir versus placebo to prevent perinatal transmission of hepatitis B. N Engl J Med. 2018;378:911-923.
- Wang M, Ran R, Zhu Y, et al. Comparison of tenofovir disoproxil fumarate and telbivudine in preventing hepatitis B transmission in mothers with high viral load. Int J Gynaecol Obstet. 2023:160:646-652.

Hepatitis B is one of the more common infections encountered in the daily practice of obstetrics. It is responsible for 40% to 45% of all cases of viral hepatitis.1,2 Hepatitis B may cause serious complications in both the infected mother and neonate.
In this article, I review the virology, epidemiology, and clinical presentation of hepatitis B and then discuss the key diagnostic tests and, subsequently, the clinical management for both the mother and neonate. I focus particular attention on relatively new information about the value of specific antiviral medication to enhance the protective effect of conventional neonatal immunoprophylaxis.
To set the framework for the discussion, consider the following 2 case studies.
CASE 1 Undetectable level of hepatitis B surface antibody in a pregnant woman
A 25-year-old healthy primigravid woman at 10 weeks’ gestation had a series of laboratory studies that included a test for hepatitis B surface antigen (HBsAg) and hepatitis B surface antibody (HBsAb). The test for the surface antigen was negative. The test for the surface antibody was below the level of detection. Upon questioning, the patient indicates that she received the 3-dose hepatitis B vaccine when she was age 13 years.
- What treatment, if any, is indicated for this patient?
- What treatment is indicated for her neonate?
CASE 2 Pregnant woman tests positive for hepatitis B surface antigen
A 31-year-old woman (G3P2002) at 12 weeks’ gestation tested positive for HBsAg. She indicates that she never has had symptomatic hepatitis and that she considers herself to be in excellent health.
- What additional laboratory tests are indicated at this time?
- What additional laboratory test should be performed at the end of the second trimester?
- What treatment is indicated for the mother and neonate?
Virology and epidemiology of hepatitis B
Hepatitis B is caused by a double-stranded, enveloped DNA virus. The virus has 10 genotypes and 24 subtypes.3 The organism contains 3 major antigens. Detection of these antigens and their corresponding antibodies is an essential step in the diagnostic workup of patients who may be infected.
The surface antigen (HBsAg) confers infectivity and is the most valuable serologic marker of infection. The e antigen (HBeAg) is not present in every infected patient. It is secreted from infected cells, but it is not incorporated into the viral particle. When present, it denotes a high level of viral replication and exceptionally high infectivity. The core antigen (HBcAg) is a valuable serologic marker for distinguishing between acute and chronic infection.1-3
Hepatitis B is highly infectious, much more so than HIV or hepatitis C. The virus has an incubation period of 4 weeks to 6 months, and the duration of incubation is inversely related to the size of the viral inoculum. The virus is transmitted in 3 principal ways: sexual contact with contaminated genital tract secretions, contact with infected blood from sharing contaminated drug-injecting paraphernalia or from receiving a blood transfusion (extremely rare today), and transmission from an infected mother to her neonate. Perinatal transmission occurs primarily during the delivery process as opposed to transplacental infection. Transmission also can occur by more casual household contact, such as sharing eating utensils, kissing, and handling an infant.1,2,4,5
Worldwide, more than 400 million people have chronic hepatitis B infection. In the United States, approximately 1.25 to 1.5 million individuals are infected. Several groups are at particularly high risk for being infected, including1-3:
- Asians
- Alaska Natives
- sub-Saharan Africans
- sex workers
- intravenous drug users
- individuals with hemophilia
- international travelers
- staff and residents of long-term care facilities
- tattoo recipients.
Continue to: Clinical presentation...
Clinical presentation
Approximately 90% of adult patients who contract hepatitis B, either symptomatically or asymptomatically, will develop protective levels of antibody and clear the virus from their system. They will then have lifelong immunity to reinfection. Approximately 10% of patients will fail to develop protective levels of antibody and will become chronically infected, posing a risk to their household members, sexual contacts, and their fetus if they become pregnant. Persistence of the surface antigen in the patient’s serum for more than 6 months denotes chronic infection. A very small number of individuals—less than 1%—will develop acute liver failure and experience a fatal outcome.1-3,5
In the United States, the prevalence of acute hepatitis B in pregnancy is 1 to 2 per 1,000. Clinical manifestations typically include anorexia, nausea, low-grade fever, right upper quadrant pain and tenderness, passage of clay-colored stools, and jaundice.
The prevalence of chronic infection in pregnancy is significantly higher, approximately 5 to 15 per 1,000. Over the long term, patients with chronic infection are at risk for progressive liver injury, including cirrhosis and even hepatocellular carcinoma. These serious sequelae are particularly likely to occur when the patient is co-infected with hepatitis C, D, or both. The overall risk of progression to chronic cirrhosis is approximately 15% to 30%. In patients who progress to cirrhosis, the annual incidence of hepatocellular carcinoma is 10%.1-3
Diagnosis of hepatitis B infection
Patients with acute hepatitis B will test positive for HBsAg and immunoglobulin M (IgM) antibody to the core antigen. Some patients will also test positive for HBeAg. Assessment of the patient’s serum by polymerase chain reaction (PCR) allows quantitation of the viral load, which often is expressed as viral copies per milliliter. Alternatively, the quantitative hepatitis B DNA concentration may be expressed as international units per milliliter (IU/mL). The World Health Organization recommends this latter quantitative method. Multiplying the DNA in IU/mL by 5.6 provides the conversion to viral copies per milliliter.
Patients with chronic hepatitis B infection will test positive for the HBsAg and for immunoglobulin G (IgG) antibody to the core antigen. They may also have a positive test for the HBeAg, and PCR may be used to quantify the viral load.1-3
Managing hepatitis B infection in pregnancy
General supportive measures. All pregnant patients should be tested for the HBsAg and HBsAb at the time of the first prenatal appointment. The tests should be repeated at the beginning of the third trimester in high-risk patients. Seropositive patients should have a hepatitis B genotype, a test for the e antigen, and tests for other sexually transmissible infections (gonorrhea, chlamydia, syphilis, HIV) and for hepatitis C and D. Liver function tests should be performed to assess for elevations in the alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels. Patients with elevated transaminase enzymes should have a coagulation profile to be certain they are not at risk for a coagulopathy.
At the end of the second trimester, patients should have a PCR assessment to determine the viral load. This assessment will be important for deciding if specific antiviral therapy is indicated during the third trimester to enhance the effects of neonatal immunoprophylaxis (see below). Of note, patients who are positive for the e antigen may have a very high viral load and yet have normal or near-normal transaminase levels. This seemingly paradoxical finding reflects the non-cytopathic nature of hepatitis B.
The patient should optimize her nutrition and sleep. She should avoid, or at least minimize, medications such as acetaminophen that could cause further liver injury. Without question, she should refrain from consuming even small amounts of alcohol. She should be tested for immunity to hepatitis A; if found to be susceptible, she should be vaccinated with the hepatitis A vaccine. This agent is an inactivated vaccine and is safe for administration at any time in pregnancy.1,2,5
Household contacts. In addition to the measures outlined above, the patient’s household contacts, particularly her sexual partner(s), should be tested for immunity to hepatitis B. If they do not have immunity by virtue of natural infection or previous vaccination, they should receive the hepatitis B vaccine series. It is also prudent to provide the sexual partner(s) with an initial dose of hepatitis B immune globulin (HBIG) to provide a temporary level of passive immunity.
Postdelivery care. After delivery, the patient should be referred to an infectious disease specialist or hepatologist for consideration of long-term treatment with antiviral agents, such as interferon alfa, pegylated interferon alfa, lamivudine, adefovir, entecavir, telbivudine, or tenofovir.6 The principal candidates for treatment are those who have cirrhosis and detectable levels of hepatitis B DNA. The ultimate goal of treatment is to reduce the serum hepatitis B DNA concentration to an undetectable level. Once the surface antigenemia is cleared, treatment can be stopped. A cure is defined when the absence of hepa-titis B DNA in the serum is sustained.
- Hepatitis B is a DNA virus that is transmitted via sexual contact, exposure to infected blood, and from an infected mother to her fetus.
- Most patients in our practice will most likely have chronic, asymptomatic infection, and the diagnosis will be established by detection of HBsAg in the patient’s serum.
- All obstetric patients should be tested for both HBsAg and HBsAb.
- Patients who are positive for the surface antigen should be tested for HIV infection and hepatitis C and D. They also should have a determination of the hepatitis B genotype and viral load and assessment of liver function (ALT, AST).
- Patients who are chronically infected with hepatitis B should be vaccinated against hepatitis A to prevent further liver injury. They also should avoid medications that might cause hepatic injury.
- Patients who have a viral DNA concentration greater than 200,000 IU/mL or a viral load greater than 1,120,000 million copies/mL should be treated with tenofovir, 300 mg daily, from week 28 until 4 to 8 weeks after delivery.
- Infants delivered to infected mothers should receive HBIG within 12 hours of birth and then begin the 3-dose hepatitis B vaccine series. The first dose should be administered prior to hospital discharge.
- Infants delivered to mothers who are negative for the surface antigen should begin the hepatitis B vaccine series prior to discharge from the hospital.
- Mothers who test negative for HBsAb should be questioned about prior vaccination. If they have never been vaccinated, they should receive the 3-dose vaccine series. If they have been vaccinated, they should receive a single hepatitis B vaccine booster. The vaccine is safe for administration at any time during pregnancy.
- Infected mothers may breastfeed as long as they do not have cracked or bleeding nipples or exudative skin lesions near the nipple(s).
Neonatal immunoprophylaxis
The Centers for Disease Control and Prevention recommends universal hepatitis B vaccination for all newborns. The first dose of the vaccine should be administered prior to hospital discharge. The second and third doses should be administered 1 and 6 months later.1,2,5 There are few, if any, medical contraindications to neonatal vaccination. For the vast majority of infants, the immunity induced by vaccination is lifelong. For a small number, immunity may wane over time. Thus, reassessment of the HBsAb concentration is indicated in selected situations, for example, acute high-risk exposure to an infected person, development of an immunosuppressive disorder, or pregnancy.
Infants delivered to mothers who are infected with hepatitis B also should receive HBIG in addition to the vaccine. HBIG provides passive immunization to counteract the high viral inoculum encountered by the neonate during delivery. This preparation should be administered within 12 hours of birth.1,2,5
In the absence of immunoprophylaxis, a neonate delivered to a mother who is seropositive for HBsAg has a 20% to 30% probability of becoming chronically infected. If the mother is positive for both the surface antigen and the e antigen, the risk of chronic infection increases to almost 90%. Approximately 90% of infants who are infected in the perinatal period subsequently develop chronic infection. However, with appropriate immunoprophylaxis in the neonatal period, the risk of perinatal transmission is reduced by 85% to 95%.1,2,5
Cesarean delivery offers no additional protection beyond that provided by immunoprophylaxis. Moreover, because immunoprophylaxis is so effective, infected mothers may breastfeed without fear of transmitting infection to their infant. Shi and colleagues published a systematic review and meta-analysis of the risk associated with breastfeeding in hepatitis B–infected mothers.7 Infants who breastfed did not have a higher rate of mother-to-child transmission, regardless of whether they received combined immunoprophylaxis or only hepatitis B vaccine and regardless of whether the HBsAg was detected in the mother’s breast milk. The only precaution is the need to avoid breastfeeding if the nipples are cracked or bleeding or if exudative lesions are present on the skin near the nipple.
Continue to: Maternal antiviral therapy...
Maternal antiviral therapy
As noted above, neonatal immunoprophylaxis is 85% to 95% effective in preventing perinatal transmission of hepatitis B infection. Failures of prophylaxis are primarily due to antenatal transmission in patients who have exceptionally high viral loads. Several cutoffs have been used to define “high viral load,” including greater than 1 to 2 million copies/mL and a hepatitis B DNA concentration greater than 200,000 IU/mL. There is not a perfect consensus on the appropriate cutoff.
In essence, 2 different approaches have been tried to further reduce the risk of perinatal transmission in these high-risk patients.8 The first major initiative was administration of HBIG (100–200 IU) intramuscularly to the patient at 28, 32, and 36 weeks. The outcomes with this approach have been inconsistent, due, at least in part, to varying doses of the agent and various cutoffs for defining “high risk,” and this intervention is no longer recommended.1,2
The second major approach is administration of specific antiviral drugs to the mother during the third trimester. The first agent widely used in clinical practice was lamivudine. In a systematic review and meta-analysis, Shi and colleagues reported that, in infants whose mothers received lamivudine plus conventional neonatal immunuprophylaxis, the risk of perinatal infection was significantly reduced compared with infants who received only immunoprophylaxis.9
Although lamivudine is effective, there is considerable concern about the rapid development of viral resistance to the medication. Accordingly, most attention today is focused on the use of tenofovir to prevent perinatal transmission.
In an important early investigation, Pan and colleagues reported the results of a randomized controlled trial conducted in China in women with a hepatitis B DNA concentration greater than 200,000 IU/mL (viral load > 1,120,000 copies/mL).10 Patients also were positive for the e antigen. Ninety-two patients were assigned to tenofovir disoproxil fumarate (TDF), 300 mg daily, from 30 to 32 weeks until postpartum week 4 plus conventional neonatal immunoprophylaxis, and 100 patients were assigned to immunoprophylaxis alone. In the intention-to-treat analysis, 18 neonates in the control group were infected compared with 5 in the treatment group (P = .007). In the per-protocol analysis, 7 neonates in the control group were infected compared with 0 in the treatment group (P = .01). No clinically significant adverse maternal or neonatal effects occurred in the treatment group.
Subsequently, Jourdain and colleagues reported a multicenter, double-blind trial conducted in 17 public health hospitals in Thailand.11 TDF (300 mg daily) or placebo was administered from 28 weeks’ gestation until 8 weeks postpartum. Patients in both arms of the study were positive for the e antigen; 87% to 90% of the patients had a serum hepatitis B DNA concentration greater than 200,000 IU/mL.Following birth, infants in both groups received an injection of HBIG and then 4 doses of hepatitis B vaccine (0, 1, 2, 4, and 6 months). Both the HBIG and hepatitis B vaccine were administered very promptly after birth (median time, 1.2–1.3 hours).
At 6 months after delivery, 2% of infants in the placebo group (3 of 147) were HBsAg-positive compared with none of the infants in the treatment arm.11 No serious adverse effects occurred in infants in the TDF group. This difference in outcome was not statistically significant, but the overall rate of infection was so low in both groups that the sample size was definitely too small to exclude a type 2 statistical error. Moreover, the fourth dose of neonatal hepatitis B vaccine may have contributed to the surprisingly low rate of perinatal transmission. Of note, the serum hepatitis B DNA concentration in the TDF group declined from a mean of 7.6 log10 IU/mL to a mean of 4.0 log10 IU/mL at delivery.
In the most recent report, Wang and colleagues reported the results of a prospective cohort study in patients with a hepatitis B virus DNA concentration greater than 200,000 IU/mL.12 Beginning at either 24 or 32 weeks, patients were assigned to treatment with either oral TDF (300 mg daily) or oral telbivudine (LdT, 600 mg daily). The medications were continued for 4 weeks postpartum. In the intention-to-treat analysis, the rates of perinatal transmission were comparable, 1.5% versus 1.8%. In the per-protocol analysis, no infants in either group were infected. However, the predelivery decline in hepatitis Bvirus DNA concentration was greater in the TDF group. The ALT elevation rate was also lower in the TDF group. Patients in the LdT group had fewer problems with anorexia but more instances of arthralgia compared with those in the TDF group.
Based primarily on these 3 investigations, I recommend that all infected patients with a hepatitis B DNA concentration greater than 200,000 IU/mL or a viral load greater than 1,120,000 million copies/mL receive oral TDF, 300 mg daily, from 28 weeks until at least 4 to 8 weeks postpartum. The decision about duration of postpartum treatment should be made in consultation with an infectious disease specialist or hepatologist.
Case studies resolved
CASE 1 No protective level of surface antibody
This patient should promptly receive a single booster dose of the hepatitis B vaccine. The vaccine is an inactivated agent and is safe for administration at any time in pregnancy. Following delivery and prior to discharge from the hospital, the neonate should receive the first dose of the hepatitis B vaccine. A second dose should be administered 1 month later, and a third dose should be administered 6 months after the first dose.
CASE 2 Mother is seropositive for HBsAg
This patient should be tested immediately for HIV infection and hepatitis C and D. The hepatitis B viral genotype should be determined. She also should have a panel of liver function tests. If any of these tests are abnormal, a coagulation profile should be obtained to be certain that the patient is not at risk for a coagulopathy. Near the end of the second trimester, a hepatitis B viral load should be obtained. If the viral DNA concentration is greater than 200,000 IU/mLor a viral load greater than 1,120,000 million copies/mL, the patient should be treated with tenofovir, 300 mg daily, from week 28 until at least 4 weeks after delivery. The neonate should receive an injection of HBIG within 12 hours of birth and the first dose of the hepatitis B vaccine prior to discharge from the hospital. Two additional doses of the vaccine should be administered 1 and 6 months later. ●

Hepatitis B is one of the more common infections encountered in the daily practice of obstetrics. It is responsible for 40% to 45% of all cases of viral hepatitis.1,2 Hepatitis B may cause serious complications in both the infected mother and neonate.
In this article, I review the virology, epidemiology, and clinical presentation of hepatitis B and then discuss the key diagnostic tests and, subsequently, the clinical management for both the mother and neonate. I focus particular attention on relatively new information about the value of specific antiviral medication to enhance the protective effect of conventional neonatal immunoprophylaxis.
To set the framework for the discussion, consider the following 2 case studies.
CASE 1 Undetectable level of hepatitis B surface antibody in a pregnant woman
A 25-year-old healthy primigravid woman at 10 weeks’ gestation had a series of laboratory studies that included a test for hepatitis B surface antigen (HBsAg) and hepatitis B surface antibody (HBsAb). The test for the surface antigen was negative. The test for the surface antibody was below the level of detection. Upon questioning, the patient indicates that she received the 3-dose hepatitis B vaccine when she was age 13 years.
- What treatment, if any, is indicated for this patient?
- What treatment is indicated for her neonate?
CASE 2 Pregnant woman tests positive for hepatitis B surface antigen
A 31-year-old woman (G3P2002) at 12 weeks’ gestation tested positive for HBsAg. She indicates that she never has had symptomatic hepatitis and that she considers herself to be in excellent health.
- What additional laboratory tests are indicated at this time?
- What additional laboratory test should be performed at the end of the second trimester?
- What treatment is indicated for the mother and neonate?
Virology and epidemiology of hepatitis B
Hepatitis B is caused by a double-stranded, enveloped DNA virus. The virus has 10 genotypes and 24 subtypes.3 The organism contains 3 major antigens. Detection of these antigens and their corresponding antibodies is an essential step in the diagnostic workup of patients who may be infected.
The surface antigen (HBsAg) confers infectivity and is the most valuable serologic marker of infection. The e antigen (HBeAg) is not present in every infected patient. It is secreted from infected cells, but it is not incorporated into the viral particle. When present, it denotes a high level of viral replication and exceptionally high infectivity. The core antigen (HBcAg) is a valuable serologic marker for distinguishing between acute and chronic infection.1-3
Hepatitis B is highly infectious, much more so than HIV or hepatitis C. The virus has an incubation period of 4 weeks to 6 months, and the duration of incubation is inversely related to the size of the viral inoculum. The virus is transmitted in 3 principal ways: sexual contact with contaminated genital tract secretions, contact with infected blood from sharing contaminated drug-injecting paraphernalia or from receiving a blood transfusion (extremely rare today), and transmission from an infected mother to her neonate. Perinatal transmission occurs primarily during the delivery process as opposed to transplacental infection. Transmission also can occur by more casual household contact, such as sharing eating utensils, kissing, and handling an infant.1,2,4,5
Worldwide, more than 400 million people have chronic hepatitis B infection. In the United States, approximately 1.25 to 1.5 million individuals are infected. Several groups are at particularly high risk for being infected, including1-3:
- Asians
- Alaska Natives
- sub-Saharan Africans
- sex workers
- intravenous drug users
- individuals with hemophilia
- international travelers
- staff and residents of long-term care facilities
- tattoo recipients.
Continue to: Clinical presentation...
Clinical presentation
Approximately 90% of adult patients who contract hepatitis B, either symptomatically or asymptomatically, will develop protective levels of antibody and clear the virus from their system. They will then have lifelong immunity to reinfection. Approximately 10% of patients will fail to develop protective levels of antibody and will become chronically infected, posing a risk to their household members, sexual contacts, and their fetus if they become pregnant. Persistence of the surface antigen in the patient’s serum for more than 6 months denotes chronic infection. A very small number of individuals—less than 1%—will develop acute liver failure and experience a fatal outcome.1-3,5
In the United States, the prevalence of acute hepatitis B in pregnancy is 1 to 2 per 1,000. Clinical manifestations typically include anorexia, nausea, low-grade fever, right upper quadrant pain and tenderness, passage of clay-colored stools, and jaundice.
The prevalence of chronic infection in pregnancy is significantly higher, approximately 5 to 15 per 1,000. Over the long term, patients with chronic infection are at risk for progressive liver injury, including cirrhosis and even hepatocellular carcinoma. These serious sequelae are particularly likely to occur when the patient is co-infected with hepatitis C, D, or both. The overall risk of progression to chronic cirrhosis is approximately 15% to 30%. In patients who progress to cirrhosis, the annual incidence of hepatocellular carcinoma is 10%.1-3
Diagnosis of hepatitis B infection
Patients with acute hepatitis B will test positive for HBsAg and immunoglobulin M (IgM) antibody to the core antigen. Some patients will also test positive for HBeAg. Assessment of the patient’s serum by polymerase chain reaction (PCR) allows quantitation of the viral load, which often is expressed as viral copies per milliliter. Alternatively, the quantitative hepatitis B DNA concentration may be expressed as international units per milliliter (IU/mL). The World Health Organization recommends this latter quantitative method. Multiplying the DNA in IU/mL by 5.6 provides the conversion to viral copies per milliliter.
Patients with chronic hepatitis B infection will test positive for the HBsAg and for immunoglobulin G (IgG) antibody to the core antigen. They may also have a positive test for the HBeAg, and PCR may be used to quantify the viral load.1-3
Managing hepatitis B infection in pregnancy
General supportive measures. All pregnant patients should be tested for the HBsAg and HBsAb at the time of the first prenatal appointment. The tests should be repeated at the beginning of the third trimester in high-risk patients. Seropositive patients should have a hepatitis B genotype, a test for the e antigen, and tests for other sexually transmissible infections (gonorrhea, chlamydia, syphilis, HIV) and for hepatitis C and D. Liver function tests should be performed to assess for elevations in the alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels. Patients with elevated transaminase enzymes should have a coagulation profile to be certain they are not at risk for a coagulopathy.
At the end of the second trimester, patients should have a PCR assessment to determine the viral load. This assessment will be important for deciding if specific antiviral therapy is indicated during the third trimester to enhance the effects of neonatal immunoprophylaxis (see below). Of note, patients who are positive for the e antigen may have a very high viral load and yet have normal or near-normal transaminase levels. This seemingly paradoxical finding reflects the non-cytopathic nature of hepatitis B.
The patient should optimize her nutrition and sleep. She should avoid, or at least minimize, medications such as acetaminophen that could cause further liver injury. Without question, she should refrain from consuming even small amounts of alcohol. She should be tested for immunity to hepatitis A; if found to be susceptible, she should be vaccinated with the hepatitis A vaccine. This agent is an inactivated vaccine and is safe for administration at any time in pregnancy.1,2,5
Household contacts. In addition to the measures outlined above, the patient’s household contacts, particularly her sexual partner(s), should be tested for immunity to hepatitis B. If they do not have immunity by virtue of natural infection or previous vaccination, they should receive the hepatitis B vaccine series. It is also prudent to provide the sexual partner(s) with an initial dose of hepatitis B immune globulin (HBIG) to provide a temporary level of passive immunity.
Postdelivery care. After delivery, the patient should be referred to an infectious disease specialist or hepatologist for consideration of long-term treatment with antiviral agents, such as interferon alfa, pegylated interferon alfa, lamivudine, adefovir, entecavir, telbivudine, or tenofovir.6 The principal candidates for treatment are those who have cirrhosis and detectable levels of hepatitis B DNA. The ultimate goal of treatment is to reduce the serum hepatitis B DNA concentration to an undetectable level. Once the surface antigenemia is cleared, treatment can be stopped. A cure is defined when the absence of hepa-titis B DNA in the serum is sustained.
- Hepatitis B is a DNA virus that is transmitted via sexual contact, exposure to infected blood, and from an infected mother to her fetus.
- Most patients in our practice will most likely have chronic, asymptomatic infection, and the diagnosis will be established by detection of HBsAg in the patient’s serum.
- All obstetric patients should be tested for both HBsAg and HBsAb.
- Patients who are positive for the surface antigen should be tested for HIV infection and hepatitis C and D. They also should have a determination of the hepatitis B genotype and viral load and assessment of liver function (ALT, AST).
- Patients who are chronically infected with hepatitis B should be vaccinated against hepatitis A to prevent further liver injury. They also should avoid medications that might cause hepatic injury.
- Patients who have a viral DNA concentration greater than 200,000 IU/mL or a viral load greater than 1,120,000 million copies/mL should be treated with tenofovir, 300 mg daily, from week 28 until 4 to 8 weeks after delivery.
- Infants delivered to infected mothers should receive HBIG within 12 hours of birth and then begin the 3-dose hepatitis B vaccine series. The first dose should be administered prior to hospital discharge.
- Infants delivered to mothers who are negative for the surface antigen should begin the hepatitis B vaccine series prior to discharge from the hospital.
- Mothers who test negative for HBsAb should be questioned about prior vaccination. If they have never been vaccinated, they should receive the 3-dose vaccine series. If they have been vaccinated, they should receive a single hepatitis B vaccine booster. The vaccine is safe for administration at any time during pregnancy.
- Infected mothers may breastfeed as long as they do not have cracked or bleeding nipples or exudative skin lesions near the nipple(s).
Neonatal immunoprophylaxis
The Centers for Disease Control and Prevention recommends universal hepatitis B vaccination for all newborns. The first dose of the vaccine should be administered prior to hospital discharge. The second and third doses should be administered 1 and 6 months later.1,2,5 There are few, if any, medical contraindications to neonatal vaccination. For the vast majority of infants, the immunity induced by vaccination is lifelong. For a small number, immunity may wane over time. Thus, reassessment of the HBsAb concentration is indicated in selected situations, for example, acute high-risk exposure to an infected person, development of an immunosuppressive disorder, or pregnancy.
Infants delivered to mothers who are infected with hepatitis B also should receive HBIG in addition to the vaccine. HBIG provides passive immunization to counteract the high viral inoculum encountered by the neonate during delivery. This preparation should be administered within 12 hours of birth.1,2,5
In the absence of immunoprophylaxis, a neonate delivered to a mother who is seropositive for HBsAg has a 20% to 30% probability of becoming chronically infected. If the mother is positive for both the surface antigen and the e antigen, the risk of chronic infection increases to almost 90%. Approximately 90% of infants who are infected in the perinatal period subsequently develop chronic infection. However, with appropriate immunoprophylaxis in the neonatal period, the risk of perinatal transmission is reduced by 85% to 95%.1,2,5
Cesarean delivery offers no additional protection beyond that provided by immunoprophylaxis. Moreover, because immunoprophylaxis is so effective, infected mothers may breastfeed without fear of transmitting infection to their infant. Shi and colleagues published a systematic review and meta-analysis of the risk associated with breastfeeding in hepatitis B–infected mothers.7 Infants who breastfed did not have a higher rate of mother-to-child transmission, regardless of whether they received combined immunoprophylaxis or only hepatitis B vaccine and regardless of whether the HBsAg was detected in the mother’s breast milk. The only precaution is the need to avoid breastfeeding if the nipples are cracked or bleeding or if exudative lesions are present on the skin near the nipple.
Continue to: Maternal antiviral therapy...
Maternal antiviral therapy
As noted above, neonatal immunoprophylaxis is 85% to 95% effective in preventing perinatal transmission of hepatitis B infection. Failures of prophylaxis are primarily due to antenatal transmission in patients who have exceptionally high viral loads. Several cutoffs have been used to define “high viral load,” including greater than 1 to 2 million copies/mL and a hepatitis B DNA concentration greater than 200,000 IU/mL. There is not a perfect consensus on the appropriate cutoff.
In essence, 2 different approaches have been tried to further reduce the risk of perinatal transmission in these high-risk patients.8 The first major initiative was administration of HBIG (100–200 IU) intramuscularly to the patient at 28, 32, and 36 weeks. The outcomes with this approach have been inconsistent, due, at least in part, to varying doses of the agent and various cutoffs for defining “high risk,” and this intervention is no longer recommended.1,2
The second major approach is administration of specific antiviral drugs to the mother during the third trimester. The first agent widely used in clinical practice was lamivudine. In a systematic review and meta-analysis, Shi and colleagues reported that, in infants whose mothers received lamivudine plus conventional neonatal immunuprophylaxis, the risk of perinatal infection was significantly reduced compared with infants who received only immunoprophylaxis.9
Although lamivudine is effective, there is considerable concern about the rapid development of viral resistance to the medication. Accordingly, most attention today is focused on the use of tenofovir to prevent perinatal transmission.
In an important early investigation, Pan and colleagues reported the results of a randomized controlled trial conducted in China in women with a hepatitis B DNA concentration greater than 200,000 IU/mL (viral load > 1,120,000 copies/mL).10 Patients also were positive for the e antigen. Ninety-two patients were assigned to tenofovir disoproxil fumarate (TDF), 300 mg daily, from 30 to 32 weeks until postpartum week 4 plus conventional neonatal immunoprophylaxis, and 100 patients were assigned to immunoprophylaxis alone. In the intention-to-treat analysis, 18 neonates in the control group were infected compared with 5 in the treatment group (P = .007). In the per-protocol analysis, 7 neonates in the control group were infected compared with 0 in the treatment group (P = .01). No clinically significant adverse maternal or neonatal effects occurred in the treatment group.
Subsequently, Jourdain and colleagues reported a multicenter, double-blind trial conducted in 17 public health hospitals in Thailand.11 TDF (300 mg daily) or placebo was administered from 28 weeks’ gestation until 8 weeks postpartum. Patients in both arms of the study were positive for the e antigen; 87% to 90% of the patients had a serum hepatitis B DNA concentration greater than 200,000 IU/mL.Following birth, infants in both groups received an injection of HBIG and then 4 doses of hepatitis B vaccine (0, 1, 2, 4, and 6 months). Both the HBIG and hepatitis B vaccine were administered very promptly after birth (median time, 1.2–1.3 hours).
At 6 months after delivery, 2% of infants in the placebo group (3 of 147) were HBsAg-positive compared with none of the infants in the treatment arm.11 No serious adverse effects occurred in infants in the TDF group. This difference in outcome was not statistically significant, but the overall rate of infection was so low in both groups that the sample size was definitely too small to exclude a type 2 statistical error. Moreover, the fourth dose of neonatal hepatitis B vaccine may have contributed to the surprisingly low rate of perinatal transmission. Of note, the serum hepatitis B DNA concentration in the TDF group declined from a mean of 7.6 log10 IU/mL to a mean of 4.0 log10 IU/mL at delivery.
In the most recent report, Wang and colleagues reported the results of a prospective cohort study in patients with a hepatitis B virus DNA concentration greater than 200,000 IU/mL.12 Beginning at either 24 or 32 weeks, patients were assigned to treatment with either oral TDF (300 mg daily) or oral telbivudine (LdT, 600 mg daily). The medications were continued for 4 weeks postpartum. In the intention-to-treat analysis, the rates of perinatal transmission were comparable, 1.5% versus 1.8%. In the per-protocol analysis, no infants in either group were infected. However, the predelivery decline in hepatitis Bvirus DNA concentration was greater in the TDF group. The ALT elevation rate was also lower in the TDF group. Patients in the LdT group had fewer problems with anorexia but more instances of arthralgia compared with those in the TDF group.
Based primarily on these 3 investigations, I recommend that all infected patients with a hepatitis B DNA concentration greater than 200,000 IU/mL or a viral load greater than 1,120,000 million copies/mL receive oral TDF, 300 mg daily, from 28 weeks until at least 4 to 8 weeks postpartum. The decision about duration of postpartum treatment should be made in consultation with an infectious disease specialist or hepatologist.
Case studies resolved
CASE 1 No protective level of surface antibody
This patient should promptly receive a single booster dose of the hepatitis B vaccine. The vaccine is an inactivated agent and is safe for administration at any time in pregnancy. Following delivery and prior to discharge from the hospital, the neonate should receive the first dose of the hepatitis B vaccine. A second dose should be administered 1 month later, and a third dose should be administered 6 months after the first dose.
CASE 2 Mother is seropositive for HBsAg
This patient should be tested immediately for HIV infection and hepatitis C and D. The hepatitis B viral genotype should be determined. She also should have a panel of liver function tests. If any of these tests are abnormal, a coagulation profile should be obtained to be certain that the patient is not at risk for a coagulopathy. Near the end of the second trimester, a hepatitis B viral load should be obtained. If the viral DNA concentration is greater than 200,000 IU/mLor a viral load greater than 1,120,000 million copies/mL, the patient should be treated with tenofovir, 300 mg daily, from week 28 until at least 4 weeks after delivery. The neonate should receive an injection of HBIG within 12 hours of birth and the first dose of the hepatitis B vaccine prior to discharge from the hospital. Two additional doses of the vaccine should be administered 1 and 6 months later. ●
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al, eds. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862.
- Bernstein HB, Lee MJ. Maternal and perinatal infection in pregnancy: viral. In: Landon MB, Galan HL, Jauniaux ERM, et al, eds. Gabbe’s Obstetrics. Normal and Problem Pregnancies. 8th ed. Elsevier; 2021;1092.
- Dusheiko G, Agarwal K, Maini MK. New approaches to chronic hepatitis B. N Engl J Med. 2023;388:55-69.
- Ma L, Alla NR, Li X, et al. Mother to child transmission of HBV: review of current clinical management and prevention strategies. Rev Med Virol. 2014; 24: 396-406.
- Society for Maternal-Fetal Medicine; Dionne-Odom J, Tita ATN, Silverman NS. SMFM consult: preventing vertical transmission of hepatitis B. Contemporary OB/GYN. September 22, 2015. Accessed August 21, 2023. https://www .contemporaryobgyn.net/view/smfm-consult-preventing -vertical-transmission-hepatitis-b
- Lok ASF. The maze of treatments for hepatitis B. N Engl J Med. 2005;352:2743-2746.
- Shi Z, Yang Y, Wang H, et al. Breastfeeding of newborns by mothers carrying hepatitis B virus: a meta-analysis and systematic review. Arch Pediatr Adolesc Med. 2011;165:837-846.
- Dusheiko G. A shift in thinking to reduce mother-to-infant transmission of hepatitis B. N Engl J Med. 2018;378:952-953.
- Shi Z, Yang Y, Ma L, et al. Lamivudine in late pregnancy to interrupt in utero transmission of hepatitis B virus: a systematic review and meta-analysis. Obstet Gynecol. 2010;116:147-159.
- Pan C, Duan Z, Dai E, et al; China Study Group for the Motherto-Child Transmission of Hepatitis B. Tenofovir to prevent hepatitis B transmission in mothers with high viral load. N Engl J Med. 2016;374:2324-2334.
- Jourdain G, Ngo-Giang-Huong N, Harrison L, et al. Tenofovir versus placebo to prevent perinatal transmission of hepatitis B. N Engl J Med. 2018;378:911-923.
- Wang M, Ran R, Zhu Y, et al. Comparison of tenofovir disoproxil fumarate and telbivudine in preventing hepatitis B transmission in mothers with high viral load. Int J Gynaecol Obstet. 2023:160:646-652.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al, eds. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862.
- Bernstein HB, Lee MJ. Maternal and perinatal infection in pregnancy: viral. In: Landon MB, Galan HL, Jauniaux ERM, et al, eds. Gabbe’s Obstetrics. Normal and Problem Pregnancies. 8th ed. Elsevier; 2021;1092.
- Dusheiko G, Agarwal K, Maini MK. New approaches to chronic hepatitis B. N Engl J Med. 2023;388:55-69.
- Ma L, Alla NR, Li X, et al. Mother to child transmission of HBV: review of current clinical management and prevention strategies. Rev Med Virol. 2014; 24: 396-406.
- Society for Maternal-Fetal Medicine; Dionne-Odom J, Tita ATN, Silverman NS. SMFM consult: preventing vertical transmission of hepatitis B. Contemporary OB/GYN. September 22, 2015. Accessed August 21, 2023. https://www .contemporaryobgyn.net/view/smfm-consult-preventing -vertical-transmission-hepatitis-b
- Lok ASF. The maze of treatments for hepatitis B. N Engl J Med. 2005;352:2743-2746.
- Shi Z, Yang Y, Wang H, et al. Breastfeeding of newborns by mothers carrying hepatitis B virus: a meta-analysis and systematic review. Arch Pediatr Adolesc Med. 2011;165:837-846.
- Dusheiko G. A shift in thinking to reduce mother-to-infant transmission of hepatitis B. N Engl J Med. 2018;378:952-953.
- Shi Z, Yang Y, Ma L, et al. Lamivudine in late pregnancy to interrupt in utero transmission of hepatitis B virus: a systematic review and meta-analysis. Obstet Gynecol. 2010;116:147-159.
- Pan C, Duan Z, Dai E, et al; China Study Group for the Motherto-Child Transmission of Hepatitis B. Tenofovir to prevent hepatitis B transmission in mothers with high viral load. N Engl J Med. 2016;374:2324-2334.
- Jourdain G, Ngo-Giang-Huong N, Harrison L, et al. Tenofovir versus placebo to prevent perinatal transmission of hepatitis B. N Engl J Med. 2018;378:911-923.
- Wang M, Ran R, Zhu Y, et al. Comparison of tenofovir disoproxil fumarate and telbivudine in preventing hepatitis B transmission in mothers with high viral load. Int J Gynaecol Obstet. 2023:160:646-652.
Freezing the biological clock: A 2023 update on preserving fertility
Throughout the 20th century, the management of ectopic pregnancy evolved from preserving the life of the mother to preserving fertility by utilizing the conservative treatment of methotrexate and/or tubal surgery. I make this, seemingly obscure, reference to managing ectopic pregnancy to consider an analogous shift over time in the management of patients with cancer. Over the next decade, the number of people who have lived 5 or more years after their cancer diagnosis is projected to increase approximately 30%, to 16.3 million. Due to the improved survival rates following a cancer diagnosis,1 revolutionary developments have been made in fertility preservation to obviate the impact of gonadotoxic therapy. We have evolved, however, from shielding and transposing ovaries to ovarian tissue cryopreservation,2 with rapid implementation.
While advances in reproductive cryopreservation have allowed for the delay, or even potential “prevention” of infertility, assisted reproductive technology (ART) cannot yet claim a “cure” in ensuring procreation. Nevertheless, fertility preservation is a burgeoning field that has transitioned from an experimental label to a standard of care in 2012, as designated by the American Society for Reproductive Medicine (ASRM).3 From the original intention of offering oocyte cryopreservation to women at risk of ovarian failure from impending gonadotoxic cancer treatment, fertility preservation has accelerated to include freezing for nonmedical reasons—eg, planned oocyte cryopreservation (POC), or “social” egg freezing, to ovarian tissue cryopreservation to accommodate the expediency needed for the treatment of certain cancer treatments. Additionally, across the United States, the number of donor egg banks, which allow women an easily accessible option, is rivaling enduring sperm banks. Due to the advanced methodology of vitrification and growing demand for the technology due to increasing IVF cycles, cryopreservation has become a specialized area of reproductive medicine, and a target of venture capital and private equity commercialization. This article will review the latest techniques, appropriate counseling, and cost/benefit ratio of fertility preservation, with an emphasis on POC.
CASE 1 Fertility preservation options for patient with breast cancer
A 37-year-old woman with newly diagnosed hormone receptor−positive breast cancer is referred for a fertility preservation consultation prior to initiating treatment. Her oncologist plans chemotherapy, followed by radiation and a minimum of 5 years of tamoxifen therapy.
What is the best consultation approach for this patient?
Consultation involves understanding several factors
The consultation approach to this patient involves ascertaining her medical, social, and family history, along with her reproductive plans.
Medical history. For the medical component, we must focus on her diagnosis, anticipated treatment with timeline, risks of gonadal toxicity with planned treatments, her current medical stability, and prognosis for expected survival.
Social history. Her age, relationship status, and desired family size address her social history.
Family history. Given that her cancer affects the breast, there is the risk of genetic susceptibility and potential for embryo testing for the BRCA gene.
Reproductive plans. These include her and her partner’s, if applicable, number of desired children and their risk factors for infertility.
Regarding the reproductive timeline, the antihormonal therapy that may be required for her treatment may improve overall survival, but it would delay the time to pregnancy. Consequently, the pursuit of fertility preservation prior to cancer treatment is a multidisciplinary approach that can involve medical oncology, radiation oncology, REI, medical genetics, and often, psychology. Fortunately, evidence continues to support fertility preservation, with or without hormonal ovarian stimulation, for patients with breast cancer. Data, with up to 5 years of follow-up, has indicated that it is safe.4
Continue to: Oncofertility...
Oncofertility
To address the need to maximize the reproductive potential of patients with newly diagnosed cancer, the field of oncofertility combines the specialties of oncology and reproductive medicine. The reproductive risk of cancer treatment is gonadotoxicity, with subsequent iatrogenic primary ovarian insufficiency (POI) and infertility. Alkylating agents (including cyclosphosphamide) have the highest risk for amenorrhea, while antimetabolites (including methotrexate, 5–fluorouracil) have the lowest risk.5 Treating bone marrow/stem cell transplantation using high-dose alkylating agents, with or without whole body irradiation, results in ≥80% amenorrhea. The minimum radiation dose to induce ovarian failure decreases with advancing age, from 18.4 Gy at age 10 years to 6 Gy at age 40 years, due to biologically diminishing ovarian reserve and an increase in the radiosensitivity of oocytes.6 An online tool—using varying factors including age, chemotherapy dose, prior treatment, smoking, and baseline diminished ovarian reserve—is available to help predict the chance of ovarian failure following chemotherapy.7
Since 2006, the American Society of Clinical Oncology recommended, as part of the consent prior to therapy, oncologists should address the possibility of infertility with patients “as early in treatment planning as possible” and “...Fertility preservation is an important, if not necessary, consideration when planning cancer treatment in reproductive-age patients.”
Reference
1. Lee SJ, Schover LR, Partridge AH, et al. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J Clin Oncol. 2006;24:2917-2931.
Cryopreservation to the rescue
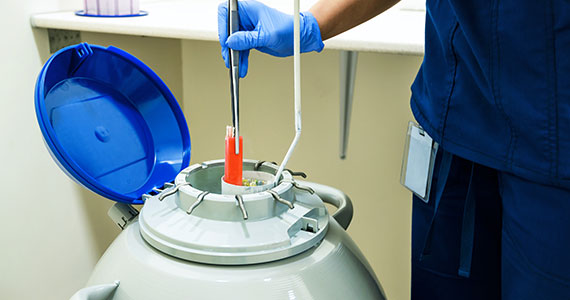
Since 2012, when ASRM removed the experimental designation on oocyte cryopreservation (OC), the number of cycles offered for fertility preservation has increased dramatically (FIGURE),8 initially being used for patients with cancer and now also including women desiring POC.
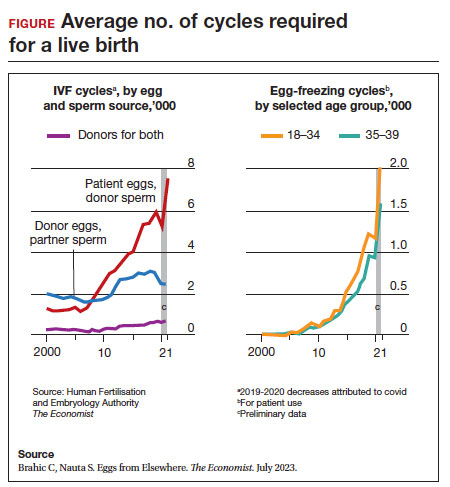
Ovarian and embryo cryopreservation. Ovarian stimulation and egg retrieval for OC can now occur within 2 weeks due to a random start protocol whereby women can begin ovarian stimulation any day in their cycle (ie, preovulation or postovulation).9
OC followed by thawing for subsequent fertilization and embryo transfer is employed as a matter of routine when patients with infertility utilize frozen eggs from a donor. While there remains debate over better live birth rates with frozen eggs versus fresh eggs, clinic experience may be a critical factor.10
Ovarian tissue cryopreservation. In addition to the fertility preservation procedures of oocytes and embryo cryopreservation, ovarian tissue cryopreservation became a standard option in 2019 when ASRM removed its experimental designation.11 Given the potential time constraints of urgent cancer treatment, ovarian tissue cryopreservation has the advantage of not requiring ovarian stimulation or sexual maturity and is able to be performed while patients are receiving chemotherapy. If successful, ovarian tissue cryopreservation followed by orthotopic transplantation has the potential to restore natural ovarian function and natural conceptions.12 However, despite first successfully being described in 2004, ovarian tissue cryopreservation, which does require subsequent thawing and tissue transplantation, remains less available to patients due to low usage rates, which have resulted in few clinics having adequate proficiency.13,14
Ovarian tissue cryopreservation involves obtaining ovarian cortical tissue, dissecting the tissue into small fragments, and cryopreserving it using either a slow-cool technique or vitrification. Orthotopic transplantation has been the most successful method for using ovarian tissue in humans. Live birth rates are modest.15 In all cancer survivors, particularly those with leukemia, autologous ovarian tissue transplantation may contain malignant cells that could lead to the reintroduction of cancer as the tissue is removed prior to treatment.16
Pregnancy outcomes using embryos created from ovaries recently exposed to chemotherapy in humans is not known, but animal studies suggest that there may be higher rates of miscarriage and birth defects given the severe DNA damage to oocytes of developing follicles.17 Hence, ovarian stimulation should be initiated and completed before the start of chemotherapy.
Continue to: Planned oocyte cryopreservation...
Planned oocyte cryopreservation
With advances in ART, POC offers patients the opportunity to preserve fertility until desired. However, despite its potential benefits, POC compels the discussion of various considerations in addition to oncofertility, such as ethical concerns and insurance coverage.
CASE 2 Woman plans for elective egg freezing
A 32-year-old single, professional woman is advancing in her career and wishes to delay childbearing. She is concerned about the potential for age-related fertility decline and wants to explore the option of elective egg freezing. Emily has no medical conditions that would impair her fertility, but she wants to ensure that she has the option of having biological children in the future. She is unsure about the potential financial burden of the procedure and whether her employer’s insurance covers such elective procedures.
How do you counsel her about her options?
Medical considerations
Approximately 25% of reproductive-aged women have considered POC.18 An analysis revealed POC was more cost-effective than delaying procreation and undergoing IVF with preimplantation genetic testing for aneuploidies at an advanced reproductive age.19
The process of planned oocyte cryopreservation. POC involves ovarian stimulation, usually with parenteral gonadotropins, to produce multiple mature oocytes for same-day cryopreservation following transvaginal retrieval, typically in an office-based surgery center as an outpatient procedure while the patient is under IV sedation. While the procedure has been proven effective, there are inherent risks and limitations. The success rates of subsequent fertility treatments using the cryopreserved eggs are influenced by the woman’s age at the time of freezing, the number of mature oocytes retrieved and vitrified, and the quality of the oocytes following thaw. A recent study reported a 70% live-birth rate in women aged less than 38 years who cryopreserved ≥ 20 mature eggs.20 To increase the number of cryopreserved oocytes, multiple egg retrievals or “batching” may be of benefit for women with diminished ovarian reserve.21
It is important for clinicians to thoroughly assess a patient’s medical history, ovarian reserve (by antral follicle count and levels of anti-müllerian hormone [AMH]), and reproductive goals before recommending proceeding with POC. Of note, AMH is a useful marker for ovarian reserve but has not been shown to predict natural fertility. Its value is in providing a guide to the dosage of ovarian stimulation and an estimation of the number of oocytes to be retrieved. Per ASRM, “Extremely low AMH values should not be used to refuse treatment in IVF.” AMH levels and antral follicle count have only a weak association with such qualitative outcomes as oocyte quality, clinical pregnancy rates, and live birth rates. Complications from egg retrieval, both short and long term, are rare. The inherent risk from POC is the lack of a guaranteed subsequent live birth.22
Ethical and social considerations
POC raises several ethical considerations, including concerns of perpetuating societal pressure on women to defer procreation to prioritize their careers over family planning.23 Despite controversies, POC appears as a chosen strategy against age-related infertility and may allow women to feel that they are more socially, psychologically, and financially stable before pursuing motherhood.24 Open and honest discussions between clinicians and patients are crucial to ensure informed decision making and address these ethical concerns.
Per an ACOG statement from February 2023 (https://www.acog.org/womens-health/faqs/having-a-baby-after-age-35-how-aging-affects-fertility-and-pregnancy) “...egg freezing is recommended mainly for patients having cancer treatment that will affect their future fertility. There is not enough research to recommend routine egg freezing for the sole purpose of delaying childbearing.”
A recent survey of patients who had elected egg freezing at some point included more than 80% who were aged 35 or older, and revealed that 93% of the survey participants had not yet returned to use their frozen oocytes.25 The most common reason cited in the survey for a delay in attempted procreation was lack of a partner. Another reason was undergoing oocyte cryopreservation after an optimal reproductive age, with participants concluding that they felt they had improved their reproductive future after undergoing oocyte cryopreservation and feeling empowered by the process. As part of counseling, women should be informed of the possibility of not utilizing their frozen eggs in the future, whether due to natural conception or other personal reasons.
Continue to: Employer insurance coverage...
Employer insurance coverage
Access to elective egg freezing is largely influenced by insurance coverage. Currently, employer-provided insurance coverage for this procedure varies widely. While some companies offer comprehensive coverage, others provide limited or no coverage at all. The cost of elective egg freezing can range from $10,000 to $15,000, excluding additional expenses such as medications and annual storage fees. The financial burden can create a gap between patients who desire POC and those with an ability to implement the process. The cost can be a significant barrier for many patients considering this option and perpetuates the lack of universal diversity, equity, and inclusion.
CASE 3 Gender dysphoria and fertility preservation
A 22-year-old transgender man is preparing to undergo gender-affirming hormone therapy and surgery. He is concerned about the potential impact of testosterone therapy on his oocytes and wishes to explore options for fertility preservation prior to oophorectomy.26
What are the patient’s options for fertility preservation?
The patient has the fertility preservation options of OC following ovarian stimulation or ovarian tissue cryopreservation at the time of oophorectomy. Preliminary evidence does not demonstrate impairment of ovarian stimulation and oocyte retrieval number with concurrent testosterone exposure. Ethical considerations, in this case, involve respecting the patient’s autonomy, addressing potential conflicts between gender-affirming care and fertility preservation (eg, a risk of dysphoria in transgender patients preserving biological gametes from a prior assigned gender), and ensuring access to fertility preservation services without discrimination. It is essential to provide the patient in this case with comprehensive information regarding the impact of hormone therapy on fertility, the available options, and the potential financial costs involved. Supportive counseling should also be offered to address any psychological or emotional aspects related to fertility preservation for all patients considering this option.
A call for diversity, equity, and inclusion
To improve access to POC, advocating for employer-offered insurance coverage is paramount. Women’s health providers can encourage dialogue between employers, insurers, and policymakers, which can lead to policy changes that prioritize coverage for fertilitypreservation options. This could include mandating coverage for POC as part of comprehensive health care plans or providing tax incentives to employers who offer coverage for these procedures. Furthermore, public awareness campaigns and advocacy efforts can help educate employers about the importance of including fertility preservation coverage in their employee benefits packages.
Conclusion
Just as physicians must recognize their responsibility to patients to distinguish unproven yet promising science from evidence-based and clinically established science, so too must they advise their patients to consider fertility preservation services in a way that is both clinically justified and ethically appropriate. Informed decisions must be made by appropriate counseling of evidence-based medicine to protect the interest of patients. POC provides patients with an opportunity to preserve their fertility and exercise reproductive autonomy. However, access to this procedure is often hindered by limited or nonexistent employer insurance coverage. By recognizing the medical, ethical, and social implications of POC and implementing strategies to improve coverage, collaborative efforts may increase accessibility and defray costs to provide patients with the option of deferring childbearing and preserving their reproductive potential. ●
1. Promptly offer fertility preservation treatment options with sensitivity and clarity.
2. Dedicate ample time and exercise patience during the consultation.
3. Provide education using multiple modalities to help patients assimilate information.
4. Encourage consultation with mental health professionals.
Special considerations for hematologic malignancies:
- Treatment can be associated with significant gonadal toxicity and premature ovarian failure.
- Patients are frequently ill at the time of presentation and ineligible for certain fertility preservation options.
References
1. Ethics Committee of the American Society for Reproductive Medicine. Fertility preservation and reproduction in patients facing gonadotoxic therapies: a committee opinion. Fertil Steril. 2018;110:380-386. doi:10.1016/j.fertnstert.2018.06.012
2. Kim SS, Klemp J, Fabian C. Breast cancer and fertility preservation. Fertil Steril. 2011;95:15351543. doi: 10.1016/j.fertnstert.2011.01.003
- American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2022-2024. Atlanta, Georgia: American Cancer Society; 2022.
- Oktay K, Karlikaya G. Ovarian function after autologous transplantation of frozen-banked human ovarian tissue. N Engl J Med. 2000;342:1919
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. Mature oocyte cryopreservation: a guideline. Fertil Steril. 2013;99:37-43. doi: 10.1016 /j.fertnstert.2012.09.028
- Marklund A, Lekberg T, Hedayati E, et al. Relapse rates and diseasespecific mortality following procedures for fertility preservation at time of breast cancer diagnosis. JAMA Oncol. 2022;8:1438-1446. doi:10.1001 /jamaoncol.2022.3677
- Zhao J, Liu J, Chen K, et al. What lies behind chemotherapy-induced amenorrhea for breast cancer patients: a meta-analysis. Breast Cancer Res Treat. 2014;145:113-128. https://doi.org/10.1007/s10549-014-2914-x
- Wallace WH, Thomson AB, Saran F, et al. Predicting age of ovarian failure after radiation to a field that includes the ovaries. Int J Radiat Oncol Biol Phys. 2005;62:738-744. http://doi.org10.1016/j.ijrobp.2004.11.038
- Chung EH, Acharya CR, Harris BS, et al. Development of a fertility risk calculator to predict individualized chance of hovarian failure after chemotherapy. J Assist Reprod Genetics. 2021;38:3047-3055. https://doi .org/10.1007/s10815-021-02311-0
- Brahic C, Nauta S. Eggs From Elsewhere. The Economist. July 2023.
- Cakmak H, Rosen MP. Random-start ovarian stimulation in patients with cancer. Curr Opin Obstet Gynecol. 2015;27:215-221. doi: 10.1097/ GCO.0000000000000180
- Eaton JL, Truong T, Li YJ, et al. Prevalence of a good perinatal outcome with cryopreserved compared with fresh donor oocytes. Obstet Gynecol. 2020;135:709-716. doi: 10.1097/AOG.0000000000003695
- Practice Committee of the American Society for Reproductive Medicine. Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril. 2019;112:1022-1033. doi: 10.1016/j.fertnstert.2019.09.013
- Oktay K, Marin L, Bedoschi G, et al. Ovarian transplantation with robotic surgery and a neovascularizing human extracellular matrix scaffold: a case series in comparison to meta-analytic data. Fertil Steril. 2021. doi:https ://doi.org/10.1016/j.fertnstert.2021.08.034
- Donnez J, Dolmans MM, Demylle D, et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004;364:1405-1410.
- Hoekman EJ, Louwe LA, Rooijers M, et al. Ovarian tissue cryopreservation: low usage rates and high live-birth rate after transplantation. Acta Obstet Gynecol Scand. 2020;99:213-221. doi: 10.1111/aogs.13735
- Donnez J, Dolmans MM, Diaz C, et al. Ovarian cortex transplantation: time to move on from experimental studies to open clinical application. Fertil Steril. 2015;104:1097-1098. doi: 10.1016/j.fertnstert.2015.08.005
- Rosendahl M, Greve T, Andersen CY. The safety of transplanting cryopreserved ovarian tissue in cancer patients: a review of the literature. J Assist Reprod Genet. 2013;30, 11-24. https://doi.org/10.1007/s10815-012-9912-x
- Soleimani R, Heytens E, Darzynkiewicz Z, et al. Mechanisms of chemotherapyinduced human ovarian aging: double strand DNA breaks and microvascular compromise. Aging (Albany NY). 2011;3:782-793.
- Milman LW, Senapati S, Sammel MD, et al. Assessing reproductive choices of women and the likelihood of oocyte cryopreservation in the era of elective oocyte freezing. Fertil Steril. 2017;107:1214-1222.e3. doi: 10.1016 /j.fertnstert.2017.03.010
- Bakkensen JB, Flannagan KSJ, Mumford SL, et al. A SART data cost-effectiveness analysis of planned oocyte cryopreservation versus in vitro fertilization with preimplantation genetic testing for aneuploidy considering ideal family size. Fertil Steril. 2022;118:875-884. https://doi.org/10.1016/j.fertnstert.2022.07.022
- Cascante SD, Blakemore JK, DeVore S. Fifteen years of autologous oocyte thaw outcomes from a large university-based fertility center. Fertil Steril. 2022;118:158-166. doi: 10.1016/j.fertnstert.2022.04.013
- Cobo A, Garrido N, Crespo J, et al. Accumulation of oocytes: a new strategy for managing low-responder patients. Reprod BioMedicine Online. 2018;37:669675. doi:10.1016/j.rbmo.2018.07.004
- Practice Committee of the American Society for Reproductive Medicine. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertil Steril. 2020;114:1151-1157. doi: 10.1016/j.fertnstert.2020.09
- What you need to know about egg-freezing, the hot new perk at Google, Apple, and Facebook. Business Insider. September 17, 2017. Accessed August 9, 2023. https://www.businessinsider.com/egg-freezing-at-facebook-apple -google-hot-new-perk-2017-9
- Varlas VN, Bors RG, Albu D, et al. Social freezing: pressing pause on fertility. Int J Environ Res Public Health. 2021;18:8088. doi: 10.3390/ijerph18158088
- Hodes-Wertz B, Druckenmiller S, Smith M, et al. What do reproductive-age women who undergo oocyte cryopreservation think about the process as a means to preserve fertility? Fertil Steril. 2013;100:1343-1349. doi: 10.1016 /j.fertnstert.2013.07.201
- Moravek MB, Dixon M, Pena SM, et al. Management of testosterone around ovarian stimulation in transmasculine patients: challenging common practices to meet patient needs-2 case reports. Hum Reprod. 2023;38:482-488. doi: 10.1093/humrep/dead003
Throughout the 20th century, the management of ectopic pregnancy evolved from preserving the life of the mother to preserving fertility by utilizing the conservative treatment of methotrexate and/or tubal surgery. I make this, seemingly obscure, reference to managing ectopic pregnancy to consider an analogous shift over time in the management of patients with cancer. Over the next decade, the number of people who have lived 5 or more years after their cancer diagnosis is projected to increase approximately 30%, to 16.3 million. Due to the improved survival rates following a cancer diagnosis,1 revolutionary developments have been made in fertility preservation to obviate the impact of gonadotoxic therapy. We have evolved, however, from shielding and transposing ovaries to ovarian tissue cryopreservation,2 with rapid implementation.
While advances in reproductive cryopreservation have allowed for the delay, or even potential “prevention” of infertility, assisted reproductive technology (ART) cannot yet claim a “cure” in ensuring procreation. Nevertheless, fertility preservation is a burgeoning field that has transitioned from an experimental label to a standard of care in 2012, as designated by the American Society for Reproductive Medicine (ASRM).3 From the original intention of offering oocyte cryopreservation to women at risk of ovarian failure from impending gonadotoxic cancer treatment, fertility preservation has accelerated to include freezing for nonmedical reasons—eg, planned oocyte cryopreservation (POC), or “social” egg freezing, to ovarian tissue cryopreservation to accommodate the expediency needed for the treatment of certain cancer treatments. Additionally, across the United States, the number of donor egg banks, which allow women an easily accessible option, is rivaling enduring sperm banks. Due to the advanced methodology of vitrification and growing demand for the technology due to increasing IVF cycles, cryopreservation has become a specialized area of reproductive medicine, and a target of venture capital and private equity commercialization. This article will review the latest techniques, appropriate counseling, and cost/benefit ratio of fertility preservation, with an emphasis on POC.
CASE 1 Fertility preservation options for patient with breast cancer
A 37-year-old woman with newly diagnosed hormone receptor−positive breast cancer is referred for a fertility preservation consultation prior to initiating treatment. Her oncologist plans chemotherapy, followed by radiation and a minimum of 5 years of tamoxifen therapy.
What is the best consultation approach for this patient?
Consultation involves understanding several factors
The consultation approach to this patient involves ascertaining her medical, social, and family history, along with her reproductive plans.
Medical history. For the medical component, we must focus on her diagnosis, anticipated treatment with timeline, risks of gonadal toxicity with planned treatments, her current medical stability, and prognosis for expected survival.
Social history. Her age, relationship status, and desired family size address her social history.
Family history. Given that her cancer affects the breast, there is the risk of genetic susceptibility and potential for embryo testing for the BRCA gene.
Reproductive plans. These include her and her partner’s, if applicable, number of desired children and their risk factors for infertility.
Regarding the reproductive timeline, the antihormonal therapy that may be required for her treatment may improve overall survival, but it would delay the time to pregnancy. Consequently, the pursuit of fertility preservation prior to cancer treatment is a multidisciplinary approach that can involve medical oncology, radiation oncology, REI, medical genetics, and often, psychology. Fortunately, evidence continues to support fertility preservation, with or without hormonal ovarian stimulation, for patients with breast cancer. Data, with up to 5 years of follow-up, has indicated that it is safe.4
Continue to: Oncofertility...
Oncofertility
To address the need to maximize the reproductive potential of patients with newly diagnosed cancer, the field of oncofertility combines the specialties of oncology and reproductive medicine. The reproductive risk of cancer treatment is gonadotoxicity, with subsequent iatrogenic primary ovarian insufficiency (POI) and infertility. Alkylating agents (including cyclosphosphamide) have the highest risk for amenorrhea, while antimetabolites (including methotrexate, 5–fluorouracil) have the lowest risk.5 Treating bone marrow/stem cell transplantation using high-dose alkylating agents, with or without whole body irradiation, results in ≥80% amenorrhea. The minimum radiation dose to induce ovarian failure decreases with advancing age, from 18.4 Gy at age 10 years to 6 Gy at age 40 years, due to biologically diminishing ovarian reserve and an increase in the radiosensitivity of oocytes.6 An online tool—using varying factors including age, chemotherapy dose, prior treatment, smoking, and baseline diminished ovarian reserve—is available to help predict the chance of ovarian failure following chemotherapy.7
Since 2006, the American Society of Clinical Oncology recommended, as part of the consent prior to therapy, oncologists should address the possibility of infertility with patients “as early in treatment planning as possible” and “...Fertility preservation is an important, if not necessary, consideration when planning cancer treatment in reproductive-age patients.”
Reference
1. Lee SJ, Schover LR, Partridge AH, et al. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J Clin Oncol. 2006;24:2917-2931.
Cryopreservation to the rescue
Since 2012, when ASRM removed the experimental designation on oocyte cryopreservation (OC), the number of cycles offered for fertility preservation has increased dramatically (FIGURE),8 initially being used for patients with cancer and now also including women desiring POC.

Ovarian and embryo cryopreservation. Ovarian stimulation and egg retrieval for OC can now occur within 2 weeks due to a random start protocol whereby women can begin ovarian stimulation any day in their cycle (ie, preovulation or postovulation).9
OC followed by thawing for subsequent fertilization and embryo transfer is employed as a matter of routine when patients with infertility utilize frozen eggs from a donor. While there remains debate over better live birth rates with frozen eggs versus fresh eggs, clinic experience may be a critical factor.10
Ovarian tissue cryopreservation. In addition to the fertility preservation procedures of oocytes and embryo cryopreservation, ovarian tissue cryopreservation became a standard option in 2019 when ASRM removed its experimental designation.11 Given the potential time constraints of urgent cancer treatment, ovarian tissue cryopreservation has the advantage of not requiring ovarian stimulation or sexual maturity and is able to be performed while patients are receiving chemotherapy. If successful, ovarian tissue cryopreservation followed by orthotopic transplantation has the potential to restore natural ovarian function and natural conceptions.12 However, despite first successfully being described in 2004, ovarian tissue cryopreservation, which does require subsequent thawing and tissue transplantation, remains less available to patients due to low usage rates, which have resulted in few clinics having adequate proficiency.13,14
Ovarian tissue cryopreservation involves obtaining ovarian cortical tissue, dissecting the tissue into small fragments, and cryopreserving it using either a slow-cool technique or vitrification. Orthotopic transplantation has been the most successful method for using ovarian tissue in humans. Live birth rates are modest.15 In all cancer survivors, particularly those with leukemia, autologous ovarian tissue transplantation may contain malignant cells that could lead to the reintroduction of cancer as the tissue is removed prior to treatment.16
Pregnancy outcomes using embryos created from ovaries recently exposed to chemotherapy in humans is not known, but animal studies suggest that there may be higher rates of miscarriage and birth defects given the severe DNA damage to oocytes of developing follicles.17 Hence, ovarian stimulation should be initiated and completed before the start of chemotherapy.
Continue to: Planned oocyte cryopreservation...
Planned oocyte cryopreservation
With advances in ART, POC offers patients the opportunity to preserve fertility until desired. However, despite its potential benefits, POC compels the discussion of various considerations in addition to oncofertility, such as ethical concerns and insurance coverage.
CASE 2 Woman plans for elective egg freezing
A 32-year-old single, professional woman is advancing in her career and wishes to delay childbearing. She is concerned about the potential for age-related fertility decline and wants to explore the option of elective egg freezing. Emily has no medical conditions that would impair her fertility, but she wants to ensure that she has the option of having biological children in the future. She is unsure about the potential financial burden of the procedure and whether her employer’s insurance covers such elective procedures.
How do you counsel her about her options?
Medical considerations
Approximately 25% of reproductive-aged women have considered POC.18 An analysis revealed POC was more cost-effective than delaying procreation and undergoing IVF with preimplantation genetic testing for aneuploidies at an advanced reproductive age.19
The process of planned oocyte cryopreservation. POC involves ovarian stimulation, usually with parenteral gonadotropins, to produce multiple mature oocytes for same-day cryopreservation following transvaginal retrieval, typically in an office-based surgery center as an outpatient procedure while the patient is under IV sedation. While the procedure has been proven effective, there are inherent risks and limitations. The success rates of subsequent fertility treatments using the cryopreserved eggs are influenced by the woman’s age at the time of freezing, the number of mature oocytes retrieved and vitrified, and the quality of the oocytes following thaw. A recent study reported a 70% live-birth rate in women aged less than 38 years who cryopreserved ≥ 20 mature eggs.20 To increase the number of cryopreserved oocytes, multiple egg retrievals or “batching” may be of benefit for women with diminished ovarian reserve.21
It is important for clinicians to thoroughly assess a patient’s medical history, ovarian reserve (by antral follicle count and levels of anti-müllerian hormone [AMH]), and reproductive goals before recommending proceeding with POC. Of note, AMH is a useful marker for ovarian reserve but has not been shown to predict natural fertility. Its value is in providing a guide to the dosage of ovarian stimulation and an estimation of the number of oocytes to be retrieved. Per ASRM, “Extremely low AMH values should not be used to refuse treatment in IVF.” AMH levels and antral follicle count have only a weak association with such qualitative outcomes as oocyte quality, clinical pregnancy rates, and live birth rates. Complications from egg retrieval, both short and long term, are rare. The inherent risk from POC is the lack of a guaranteed subsequent live birth.22
Ethical and social considerations
POC raises several ethical considerations, including concerns of perpetuating societal pressure on women to defer procreation to prioritize their careers over family planning.23 Despite controversies, POC appears as a chosen strategy against age-related infertility and may allow women to feel that they are more socially, psychologically, and financially stable before pursuing motherhood.24 Open and honest discussions between clinicians and patients are crucial to ensure informed decision making and address these ethical concerns.
Per an ACOG statement from February 2023 (https://www.acog.org/womens-health/faqs/having-a-baby-after-age-35-how-aging-affects-fertility-and-pregnancy) “...egg freezing is recommended mainly for patients having cancer treatment that will affect their future fertility. There is not enough research to recommend routine egg freezing for the sole purpose of delaying childbearing.”
A recent survey of patients who had elected egg freezing at some point included more than 80% who were aged 35 or older, and revealed that 93% of the survey participants had not yet returned to use their frozen oocytes.25 The most common reason cited in the survey for a delay in attempted procreation was lack of a partner. Another reason was undergoing oocyte cryopreservation after an optimal reproductive age, with participants concluding that they felt they had improved their reproductive future after undergoing oocyte cryopreservation and feeling empowered by the process. As part of counseling, women should be informed of the possibility of not utilizing their frozen eggs in the future, whether due to natural conception or other personal reasons.
Continue to: Employer insurance coverage...
Employer insurance coverage
Access to elective egg freezing is largely influenced by insurance coverage. Currently, employer-provided insurance coverage for this procedure varies widely. While some companies offer comprehensive coverage, others provide limited or no coverage at all. The cost of elective egg freezing can range from $10,000 to $15,000, excluding additional expenses such as medications and annual storage fees. The financial burden can create a gap between patients who desire POC and those with an ability to implement the process. The cost can be a significant barrier for many patients considering this option and perpetuates the lack of universal diversity, equity, and inclusion.
CASE 3 Gender dysphoria and fertility preservation
A 22-year-old transgender man is preparing to undergo gender-affirming hormone therapy and surgery. He is concerned about the potential impact of testosterone therapy on his oocytes and wishes to explore options for fertility preservation prior to oophorectomy.26
What are the patient’s options for fertility preservation?
The patient has the fertility preservation options of OC following ovarian stimulation or ovarian tissue cryopreservation at the time of oophorectomy. Preliminary evidence does not demonstrate impairment of ovarian stimulation and oocyte retrieval number with concurrent testosterone exposure. Ethical considerations, in this case, involve respecting the patient’s autonomy, addressing potential conflicts between gender-affirming care and fertility preservation (eg, a risk of dysphoria in transgender patients preserving biological gametes from a prior assigned gender), and ensuring access to fertility preservation services without discrimination. It is essential to provide the patient in this case with comprehensive information regarding the impact of hormone therapy on fertility, the available options, and the potential financial costs involved. Supportive counseling should also be offered to address any psychological or emotional aspects related to fertility preservation for all patients considering this option.
A call for diversity, equity, and inclusion
To improve access to POC, advocating for employer-offered insurance coverage is paramount. Women’s health providers can encourage dialogue between employers, insurers, and policymakers, which can lead to policy changes that prioritize coverage for fertilitypreservation options. This could include mandating coverage for POC as part of comprehensive health care plans or providing tax incentives to employers who offer coverage for these procedures. Furthermore, public awareness campaigns and advocacy efforts can help educate employers about the importance of including fertility preservation coverage in their employee benefits packages.
Conclusion
Just as physicians must recognize their responsibility to patients to distinguish unproven yet promising science from evidence-based and clinically established science, so too must they advise their patients to consider fertility preservation services in a way that is both clinically justified and ethically appropriate. Informed decisions must be made by appropriate counseling of evidence-based medicine to protect the interest of patients. POC provides patients with an opportunity to preserve their fertility and exercise reproductive autonomy. However, access to this procedure is often hindered by limited or nonexistent employer insurance coverage. By recognizing the medical, ethical, and social implications of POC and implementing strategies to improve coverage, collaborative efforts may increase accessibility and defray costs to provide patients with the option of deferring childbearing and preserving their reproductive potential. ●
1. Promptly offer fertility preservation treatment options with sensitivity and clarity.
2. Dedicate ample time and exercise patience during the consultation.
3. Provide education using multiple modalities to help patients assimilate information.
4. Encourage consultation with mental health professionals.
Special considerations for hematologic malignancies:
- Treatment can be associated with significant gonadal toxicity and premature ovarian failure.
- Patients are frequently ill at the time of presentation and ineligible for certain fertility preservation options.
References
1. Ethics Committee of the American Society for Reproductive Medicine. Fertility preservation and reproduction in patients facing gonadotoxic therapies: a committee opinion. Fertil Steril. 2018;110:380-386. doi:10.1016/j.fertnstert.2018.06.012
2. Kim SS, Klemp J, Fabian C. Breast cancer and fertility preservation. Fertil Steril. 2011;95:15351543. doi: 10.1016/j.fertnstert.2011.01.003
Throughout the 20th century, the management of ectopic pregnancy evolved from preserving the life of the mother to preserving fertility by utilizing the conservative treatment of methotrexate and/or tubal surgery. I make this, seemingly obscure, reference to managing ectopic pregnancy to consider an analogous shift over time in the management of patients with cancer. Over the next decade, the number of people who have lived 5 or more years after their cancer diagnosis is projected to increase approximately 30%, to 16.3 million. Due to the improved survival rates following a cancer diagnosis,1 revolutionary developments have been made in fertility preservation to obviate the impact of gonadotoxic therapy. We have evolved, however, from shielding and transposing ovaries to ovarian tissue cryopreservation,2 with rapid implementation.
While advances in reproductive cryopreservation have allowed for the delay, or even potential “prevention” of infertility, assisted reproductive technology (ART) cannot yet claim a “cure” in ensuring procreation. Nevertheless, fertility preservation is a burgeoning field that has transitioned from an experimental label to a standard of care in 2012, as designated by the American Society for Reproductive Medicine (ASRM).3 From the original intention of offering oocyte cryopreservation to women at risk of ovarian failure from impending gonadotoxic cancer treatment, fertility preservation has accelerated to include freezing for nonmedical reasons—eg, planned oocyte cryopreservation (POC), or “social” egg freezing, to ovarian tissue cryopreservation to accommodate the expediency needed for the treatment of certain cancer treatments. Additionally, across the United States, the number of donor egg banks, which allow women an easily accessible option, is rivaling enduring sperm banks. Due to the advanced methodology of vitrification and growing demand for the technology due to increasing IVF cycles, cryopreservation has become a specialized area of reproductive medicine, and a target of venture capital and private equity commercialization. This article will review the latest techniques, appropriate counseling, and cost/benefit ratio of fertility preservation, with an emphasis on POC.
CASE 1 Fertility preservation options for patient with breast cancer
A 37-year-old woman with newly diagnosed hormone receptor−positive breast cancer is referred for a fertility preservation consultation prior to initiating treatment. Her oncologist plans chemotherapy, followed by radiation and a minimum of 5 years of tamoxifen therapy.
What is the best consultation approach for this patient?
Consultation involves understanding several factors
The consultation approach to this patient involves ascertaining her medical, social, and family history, along with her reproductive plans.
Medical history. For the medical component, we must focus on her diagnosis, anticipated treatment with timeline, risks of gonadal toxicity with planned treatments, her current medical stability, and prognosis for expected survival.
Social history. Her age, relationship status, and desired family size address her social history.
Family history. Given that her cancer affects the breast, there is the risk of genetic susceptibility and potential for embryo testing for the BRCA gene.
Reproductive plans. These include her and her partner’s, if applicable, number of desired children and their risk factors for infertility.
Regarding the reproductive timeline, the antihormonal therapy that may be required for her treatment may improve overall survival, but it would delay the time to pregnancy. Consequently, the pursuit of fertility preservation prior to cancer treatment is a multidisciplinary approach that can involve medical oncology, radiation oncology, REI, medical genetics, and often, psychology. Fortunately, evidence continues to support fertility preservation, with or without hormonal ovarian stimulation, for patients with breast cancer. Data, with up to 5 years of follow-up, has indicated that it is safe.4
Continue to: Oncofertility...
Oncofertility
To address the need to maximize the reproductive potential of patients with newly diagnosed cancer, the field of oncofertility combines the specialties of oncology and reproductive medicine. The reproductive risk of cancer treatment is gonadotoxicity, with subsequent iatrogenic primary ovarian insufficiency (POI) and infertility. Alkylating agents (including cyclosphosphamide) have the highest risk for amenorrhea, while antimetabolites (including methotrexate, 5–fluorouracil) have the lowest risk.5 Treating bone marrow/stem cell transplantation using high-dose alkylating agents, with or without whole body irradiation, results in ≥80% amenorrhea. The minimum radiation dose to induce ovarian failure decreases with advancing age, from 18.4 Gy at age 10 years to 6 Gy at age 40 years, due to biologically diminishing ovarian reserve and an increase in the radiosensitivity of oocytes.6 An online tool—using varying factors including age, chemotherapy dose, prior treatment, smoking, and baseline diminished ovarian reserve—is available to help predict the chance of ovarian failure following chemotherapy.7
Since 2006, the American Society of Clinical Oncology recommended, as part of the consent prior to therapy, oncologists should address the possibility of infertility with patients “as early in treatment planning as possible” and “...Fertility preservation is an important, if not necessary, consideration when planning cancer treatment in reproductive-age patients.”
Reference
1. Lee SJ, Schover LR, Partridge AH, et al. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J Clin Oncol. 2006;24:2917-2931.
Cryopreservation to the rescue
Since 2012, when ASRM removed the experimental designation on oocyte cryopreservation (OC), the number of cycles offered for fertility preservation has increased dramatically (FIGURE),8 initially being used for patients with cancer and now also including women desiring POC.

Ovarian and embryo cryopreservation. Ovarian stimulation and egg retrieval for OC can now occur within 2 weeks due to a random start protocol whereby women can begin ovarian stimulation any day in their cycle (ie, preovulation or postovulation).9
OC followed by thawing for subsequent fertilization and embryo transfer is employed as a matter of routine when patients with infertility utilize frozen eggs from a donor. While there remains debate over better live birth rates with frozen eggs versus fresh eggs, clinic experience may be a critical factor.10
Ovarian tissue cryopreservation. In addition to the fertility preservation procedures of oocytes and embryo cryopreservation, ovarian tissue cryopreservation became a standard option in 2019 when ASRM removed its experimental designation.11 Given the potential time constraints of urgent cancer treatment, ovarian tissue cryopreservation has the advantage of not requiring ovarian stimulation or sexual maturity and is able to be performed while patients are receiving chemotherapy. If successful, ovarian tissue cryopreservation followed by orthotopic transplantation has the potential to restore natural ovarian function and natural conceptions.12 However, despite first successfully being described in 2004, ovarian tissue cryopreservation, which does require subsequent thawing and tissue transplantation, remains less available to patients due to low usage rates, which have resulted in few clinics having adequate proficiency.13,14
Ovarian tissue cryopreservation involves obtaining ovarian cortical tissue, dissecting the tissue into small fragments, and cryopreserving it using either a slow-cool technique or vitrification. Orthotopic transplantation has been the most successful method for using ovarian tissue in humans. Live birth rates are modest.15 In all cancer survivors, particularly those with leukemia, autologous ovarian tissue transplantation may contain malignant cells that could lead to the reintroduction of cancer as the tissue is removed prior to treatment.16
Pregnancy outcomes using embryos created from ovaries recently exposed to chemotherapy in humans is not known, but animal studies suggest that there may be higher rates of miscarriage and birth defects given the severe DNA damage to oocytes of developing follicles.17 Hence, ovarian stimulation should be initiated and completed before the start of chemotherapy.
Continue to: Planned oocyte cryopreservation...
Planned oocyte cryopreservation
With advances in ART, POC offers patients the opportunity to preserve fertility until desired. However, despite its potential benefits, POC compels the discussion of various considerations in addition to oncofertility, such as ethical concerns and insurance coverage.
CASE 2 Woman plans for elective egg freezing
A 32-year-old single, professional woman is advancing in her career and wishes to delay childbearing. She is concerned about the potential for age-related fertility decline and wants to explore the option of elective egg freezing. Emily has no medical conditions that would impair her fertility, but she wants to ensure that she has the option of having biological children in the future. She is unsure about the potential financial burden of the procedure and whether her employer’s insurance covers such elective procedures.
How do you counsel her about her options?
Medical considerations
Approximately 25% of reproductive-aged women have considered POC.18 An analysis revealed POC was more cost-effective than delaying procreation and undergoing IVF with preimplantation genetic testing for aneuploidies at an advanced reproductive age.19
The process of planned oocyte cryopreservation. POC involves ovarian stimulation, usually with parenteral gonadotropins, to produce multiple mature oocytes for same-day cryopreservation following transvaginal retrieval, typically in an office-based surgery center as an outpatient procedure while the patient is under IV sedation. While the procedure has been proven effective, there are inherent risks and limitations. The success rates of subsequent fertility treatments using the cryopreserved eggs are influenced by the woman’s age at the time of freezing, the number of mature oocytes retrieved and vitrified, and the quality of the oocytes following thaw. A recent study reported a 70% live-birth rate in women aged less than 38 years who cryopreserved ≥ 20 mature eggs.20 To increase the number of cryopreserved oocytes, multiple egg retrievals or “batching” may be of benefit for women with diminished ovarian reserve.21
It is important for clinicians to thoroughly assess a patient’s medical history, ovarian reserve (by antral follicle count and levels of anti-müllerian hormone [AMH]), and reproductive goals before recommending proceeding with POC. Of note, AMH is a useful marker for ovarian reserve but has not been shown to predict natural fertility. Its value is in providing a guide to the dosage of ovarian stimulation and an estimation of the number of oocytes to be retrieved. Per ASRM, “Extremely low AMH values should not be used to refuse treatment in IVF.” AMH levels and antral follicle count have only a weak association with such qualitative outcomes as oocyte quality, clinical pregnancy rates, and live birth rates. Complications from egg retrieval, both short and long term, are rare. The inherent risk from POC is the lack of a guaranteed subsequent live birth.22
Ethical and social considerations
POC raises several ethical considerations, including concerns of perpetuating societal pressure on women to defer procreation to prioritize their careers over family planning.23 Despite controversies, POC appears as a chosen strategy against age-related infertility and may allow women to feel that they are more socially, psychologically, and financially stable before pursuing motherhood.24 Open and honest discussions between clinicians and patients are crucial to ensure informed decision making and address these ethical concerns.
Per an ACOG statement from February 2023 (https://www.acog.org/womens-health/faqs/having-a-baby-after-age-35-how-aging-affects-fertility-and-pregnancy) “...egg freezing is recommended mainly for patients having cancer treatment that will affect their future fertility. There is not enough research to recommend routine egg freezing for the sole purpose of delaying childbearing.”
A recent survey of patients who had elected egg freezing at some point included more than 80% who were aged 35 or older, and revealed that 93% of the survey participants had not yet returned to use their frozen oocytes.25 The most common reason cited in the survey for a delay in attempted procreation was lack of a partner. Another reason was undergoing oocyte cryopreservation after an optimal reproductive age, with participants concluding that they felt they had improved their reproductive future after undergoing oocyte cryopreservation and feeling empowered by the process. As part of counseling, women should be informed of the possibility of not utilizing their frozen eggs in the future, whether due to natural conception or other personal reasons.
Continue to: Employer insurance coverage...
Employer insurance coverage
Access to elective egg freezing is largely influenced by insurance coverage. Currently, employer-provided insurance coverage for this procedure varies widely. While some companies offer comprehensive coverage, others provide limited or no coverage at all. The cost of elective egg freezing can range from $10,000 to $15,000, excluding additional expenses such as medications and annual storage fees. The financial burden can create a gap between patients who desire POC and those with an ability to implement the process. The cost can be a significant barrier for many patients considering this option and perpetuates the lack of universal diversity, equity, and inclusion.
CASE 3 Gender dysphoria and fertility preservation
A 22-year-old transgender man is preparing to undergo gender-affirming hormone therapy and surgery. He is concerned about the potential impact of testosterone therapy on his oocytes and wishes to explore options for fertility preservation prior to oophorectomy.26
What are the patient’s options for fertility preservation?
The patient has the fertility preservation options of OC following ovarian stimulation or ovarian tissue cryopreservation at the time of oophorectomy. Preliminary evidence does not demonstrate impairment of ovarian stimulation and oocyte retrieval number with concurrent testosterone exposure. Ethical considerations, in this case, involve respecting the patient’s autonomy, addressing potential conflicts between gender-affirming care and fertility preservation (eg, a risk of dysphoria in transgender patients preserving biological gametes from a prior assigned gender), and ensuring access to fertility preservation services without discrimination. It is essential to provide the patient in this case with comprehensive information regarding the impact of hormone therapy on fertility, the available options, and the potential financial costs involved. Supportive counseling should also be offered to address any psychological or emotional aspects related to fertility preservation for all patients considering this option.
A call for diversity, equity, and inclusion
To improve access to POC, advocating for employer-offered insurance coverage is paramount. Women’s health providers can encourage dialogue between employers, insurers, and policymakers, which can lead to policy changes that prioritize coverage for fertilitypreservation options. This could include mandating coverage for POC as part of comprehensive health care plans or providing tax incentives to employers who offer coverage for these procedures. Furthermore, public awareness campaigns and advocacy efforts can help educate employers about the importance of including fertility preservation coverage in their employee benefits packages.
Conclusion
Just as physicians must recognize their responsibility to patients to distinguish unproven yet promising science from evidence-based and clinically established science, so too must they advise their patients to consider fertility preservation services in a way that is both clinically justified and ethically appropriate. Informed decisions must be made by appropriate counseling of evidence-based medicine to protect the interest of patients. POC provides patients with an opportunity to preserve their fertility and exercise reproductive autonomy. However, access to this procedure is often hindered by limited or nonexistent employer insurance coverage. By recognizing the medical, ethical, and social implications of POC and implementing strategies to improve coverage, collaborative efforts may increase accessibility and defray costs to provide patients with the option of deferring childbearing and preserving their reproductive potential. ●
1. Promptly offer fertility preservation treatment options with sensitivity and clarity.
2. Dedicate ample time and exercise patience during the consultation.
3. Provide education using multiple modalities to help patients assimilate information.
4. Encourage consultation with mental health professionals.
Special considerations for hematologic malignancies:
- Treatment can be associated with significant gonadal toxicity and premature ovarian failure.
- Patients are frequently ill at the time of presentation and ineligible for certain fertility preservation options.
References
1. Ethics Committee of the American Society for Reproductive Medicine. Fertility preservation and reproduction in patients facing gonadotoxic therapies: a committee opinion. Fertil Steril. 2018;110:380-386. doi:10.1016/j.fertnstert.2018.06.012
2. Kim SS, Klemp J, Fabian C. Breast cancer and fertility preservation. Fertil Steril. 2011;95:15351543. doi: 10.1016/j.fertnstert.2011.01.003
- American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2022-2024. Atlanta, Georgia: American Cancer Society; 2022.
- Oktay K, Karlikaya G. Ovarian function after autologous transplantation of frozen-banked human ovarian tissue. N Engl J Med. 2000;342:1919
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. Mature oocyte cryopreservation: a guideline. Fertil Steril. 2013;99:37-43. doi: 10.1016 /j.fertnstert.2012.09.028
- Marklund A, Lekberg T, Hedayati E, et al. Relapse rates and diseasespecific mortality following procedures for fertility preservation at time of breast cancer diagnosis. JAMA Oncol. 2022;8:1438-1446. doi:10.1001 /jamaoncol.2022.3677
- Zhao J, Liu J, Chen K, et al. What lies behind chemotherapy-induced amenorrhea for breast cancer patients: a meta-analysis. Breast Cancer Res Treat. 2014;145:113-128. https://doi.org/10.1007/s10549-014-2914-x
- Wallace WH, Thomson AB, Saran F, et al. Predicting age of ovarian failure after radiation to a field that includes the ovaries. Int J Radiat Oncol Biol Phys. 2005;62:738-744. http://doi.org10.1016/j.ijrobp.2004.11.038
- Chung EH, Acharya CR, Harris BS, et al. Development of a fertility risk calculator to predict individualized chance of hovarian failure after chemotherapy. J Assist Reprod Genetics. 2021;38:3047-3055. https://doi .org/10.1007/s10815-021-02311-0
- Brahic C, Nauta S. Eggs From Elsewhere. The Economist. July 2023.
- Cakmak H, Rosen MP. Random-start ovarian stimulation in patients with cancer. Curr Opin Obstet Gynecol. 2015;27:215-221. doi: 10.1097/ GCO.0000000000000180
- Eaton JL, Truong T, Li YJ, et al. Prevalence of a good perinatal outcome with cryopreserved compared with fresh donor oocytes. Obstet Gynecol. 2020;135:709-716. doi: 10.1097/AOG.0000000000003695
- Practice Committee of the American Society for Reproductive Medicine. Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril. 2019;112:1022-1033. doi: 10.1016/j.fertnstert.2019.09.013
- Oktay K, Marin L, Bedoschi G, et al. Ovarian transplantation with robotic surgery and a neovascularizing human extracellular matrix scaffold: a case series in comparison to meta-analytic data. Fertil Steril. 2021. doi:https ://doi.org/10.1016/j.fertnstert.2021.08.034
- Donnez J, Dolmans MM, Demylle D, et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004;364:1405-1410.
- Hoekman EJ, Louwe LA, Rooijers M, et al. Ovarian tissue cryopreservation: low usage rates and high live-birth rate after transplantation. Acta Obstet Gynecol Scand. 2020;99:213-221. doi: 10.1111/aogs.13735
- Donnez J, Dolmans MM, Diaz C, et al. Ovarian cortex transplantation: time to move on from experimental studies to open clinical application. Fertil Steril. 2015;104:1097-1098. doi: 10.1016/j.fertnstert.2015.08.005
- Rosendahl M, Greve T, Andersen CY. The safety of transplanting cryopreserved ovarian tissue in cancer patients: a review of the literature. J Assist Reprod Genet. 2013;30, 11-24. https://doi.org/10.1007/s10815-012-9912-x
- Soleimani R, Heytens E, Darzynkiewicz Z, et al. Mechanisms of chemotherapyinduced human ovarian aging: double strand DNA breaks and microvascular compromise. Aging (Albany NY). 2011;3:782-793.
- Milman LW, Senapati S, Sammel MD, et al. Assessing reproductive choices of women and the likelihood of oocyte cryopreservation in the era of elective oocyte freezing. Fertil Steril. 2017;107:1214-1222.e3. doi: 10.1016 /j.fertnstert.2017.03.010
- Bakkensen JB, Flannagan KSJ, Mumford SL, et al. A SART data cost-effectiveness analysis of planned oocyte cryopreservation versus in vitro fertilization with preimplantation genetic testing for aneuploidy considering ideal family size. Fertil Steril. 2022;118:875-884. https://doi.org/10.1016/j.fertnstert.2022.07.022
- Cascante SD, Blakemore JK, DeVore S. Fifteen years of autologous oocyte thaw outcomes from a large university-based fertility center. Fertil Steril. 2022;118:158-166. doi: 10.1016/j.fertnstert.2022.04.013
- Cobo A, Garrido N, Crespo J, et al. Accumulation of oocytes: a new strategy for managing low-responder patients. Reprod BioMedicine Online. 2018;37:669675. doi:10.1016/j.rbmo.2018.07.004
- Practice Committee of the American Society for Reproductive Medicine. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertil Steril. 2020;114:1151-1157. doi: 10.1016/j.fertnstert.2020.09
- What you need to know about egg-freezing, the hot new perk at Google, Apple, and Facebook. Business Insider. September 17, 2017. Accessed August 9, 2023. https://www.businessinsider.com/egg-freezing-at-facebook-apple -google-hot-new-perk-2017-9
- Varlas VN, Bors RG, Albu D, et al. Social freezing: pressing pause on fertility. Int J Environ Res Public Health. 2021;18:8088. doi: 10.3390/ijerph18158088
- Hodes-Wertz B, Druckenmiller S, Smith M, et al. What do reproductive-age women who undergo oocyte cryopreservation think about the process as a means to preserve fertility? Fertil Steril. 2013;100:1343-1349. doi: 10.1016 /j.fertnstert.2013.07.201
- Moravek MB, Dixon M, Pena SM, et al. Management of testosterone around ovarian stimulation in transmasculine patients: challenging common practices to meet patient needs-2 case reports. Hum Reprod. 2023;38:482-488. doi: 10.1093/humrep/dead003
- American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2022-2024. Atlanta, Georgia: American Cancer Society; 2022.
- Oktay K, Karlikaya G. Ovarian function after autologous transplantation of frozen-banked human ovarian tissue. N Engl J Med. 2000;342:1919
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. Mature oocyte cryopreservation: a guideline. Fertil Steril. 2013;99:37-43. doi: 10.1016 /j.fertnstert.2012.09.028
- Marklund A, Lekberg T, Hedayati E, et al. Relapse rates and diseasespecific mortality following procedures for fertility preservation at time of breast cancer diagnosis. JAMA Oncol. 2022;8:1438-1446. doi:10.1001 /jamaoncol.2022.3677
- Zhao J, Liu J, Chen K, et al. What lies behind chemotherapy-induced amenorrhea for breast cancer patients: a meta-analysis. Breast Cancer Res Treat. 2014;145:113-128. https://doi.org/10.1007/s10549-014-2914-x
- Wallace WH, Thomson AB, Saran F, et al. Predicting age of ovarian failure after radiation to a field that includes the ovaries. Int J Radiat Oncol Biol Phys. 2005;62:738-744. http://doi.org10.1016/j.ijrobp.2004.11.038
- Chung EH, Acharya CR, Harris BS, et al. Development of a fertility risk calculator to predict individualized chance of hovarian failure after chemotherapy. J Assist Reprod Genetics. 2021;38:3047-3055. https://doi .org/10.1007/s10815-021-02311-0
- Brahic C, Nauta S. Eggs From Elsewhere. The Economist. July 2023.
- Cakmak H, Rosen MP. Random-start ovarian stimulation in patients with cancer. Curr Opin Obstet Gynecol. 2015;27:215-221. doi: 10.1097/ GCO.0000000000000180
- Eaton JL, Truong T, Li YJ, et al. Prevalence of a good perinatal outcome with cryopreserved compared with fresh donor oocytes. Obstet Gynecol. 2020;135:709-716. doi: 10.1097/AOG.0000000000003695
- Practice Committee of the American Society for Reproductive Medicine. Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril. 2019;112:1022-1033. doi: 10.1016/j.fertnstert.2019.09.013
- Oktay K, Marin L, Bedoschi G, et al. Ovarian transplantation with robotic surgery and a neovascularizing human extracellular matrix scaffold: a case series in comparison to meta-analytic data. Fertil Steril. 2021. doi:https ://doi.org/10.1016/j.fertnstert.2021.08.034
- Donnez J, Dolmans MM, Demylle D, et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004;364:1405-1410.
- Hoekman EJ, Louwe LA, Rooijers M, et al. Ovarian tissue cryopreservation: low usage rates and high live-birth rate after transplantation. Acta Obstet Gynecol Scand. 2020;99:213-221. doi: 10.1111/aogs.13735
- Donnez J, Dolmans MM, Diaz C, et al. Ovarian cortex transplantation: time to move on from experimental studies to open clinical application. Fertil Steril. 2015;104:1097-1098. doi: 10.1016/j.fertnstert.2015.08.005
- Rosendahl M, Greve T, Andersen CY. The safety of transplanting cryopreserved ovarian tissue in cancer patients: a review of the literature. J Assist Reprod Genet. 2013;30, 11-24. https://doi.org/10.1007/s10815-012-9912-x
- Soleimani R, Heytens E, Darzynkiewicz Z, et al. Mechanisms of chemotherapyinduced human ovarian aging: double strand DNA breaks and microvascular compromise. Aging (Albany NY). 2011;3:782-793.
- Milman LW, Senapati S, Sammel MD, et al. Assessing reproductive choices of women and the likelihood of oocyte cryopreservation in the era of elective oocyte freezing. Fertil Steril. 2017;107:1214-1222.e3. doi: 10.1016 /j.fertnstert.2017.03.010
- Bakkensen JB, Flannagan KSJ, Mumford SL, et al. A SART data cost-effectiveness analysis of planned oocyte cryopreservation versus in vitro fertilization with preimplantation genetic testing for aneuploidy considering ideal family size. Fertil Steril. 2022;118:875-884. https://doi.org/10.1016/j.fertnstert.2022.07.022
- Cascante SD, Blakemore JK, DeVore S. Fifteen years of autologous oocyte thaw outcomes from a large university-based fertility center. Fertil Steril. 2022;118:158-166. doi: 10.1016/j.fertnstert.2022.04.013
- Cobo A, Garrido N, Crespo J, et al. Accumulation of oocytes: a new strategy for managing low-responder patients. Reprod BioMedicine Online. 2018;37:669675. doi:10.1016/j.rbmo.2018.07.004
- Practice Committee of the American Society for Reproductive Medicine. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertil Steril. 2020;114:1151-1157. doi: 10.1016/j.fertnstert.2020.09
- What you need to know about egg-freezing, the hot new perk at Google, Apple, and Facebook. Business Insider. September 17, 2017. Accessed August 9, 2023. https://www.businessinsider.com/egg-freezing-at-facebook-apple -google-hot-new-perk-2017-9
- Varlas VN, Bors RG, Albu D, et al. Social freezing: pressing pause on fertility. Int J Environ Res Public Health. 2021;18:8088. doi: 10.3390/ijerph18158088
- Hodes-Wertz B, Druckenmiller S, Smith M, et al. What do reproductive-age women who undergo oocyte cryopreservation think about the process as a means to preserve fertility? Fertil Steril. 2013;100:1343-1349. doi: 10.1016 /j.fertnstert.2013.07.201
- Moravek MB, Dixon M, Pena SM, et al. Management of testosterone around ovarian stimulation in transmasculine patients: challenging common practices to meet patient needs-2 case reports. Hum Reprod. 2023;38:482-488. doi: 10.1093/humrep/dead003
From Breakouts to Bargains: Strategies for Patient-Centered, Cost-effective Acne Care
In the United States, acne affects 85% of adolescents and can persist into adulthood at a prevalence of 30% to 50% in adult women. 1,2 The pathogenesis of acne is multifactorial and involves hyperkeratinization of the follicle, bacterial colonization with Cutibacterium acnes , and increased androgen-induced sebum production, which together lead to inflammation. 3,4 A wide range of treatment guideline–recommended options are available, including benzoyl peroxide (BPO), topical retinoids, topical and oral antibiotics, antiandrogens, and isotretinoin. 5 However, these options vary widely in their clinical uses, effectiveness, and costs.
Why Cost-effective Acne Care Matters
Out-of-pocket spending by patients on acne treatments can be substantial, with surveys finding that acne patients often spend hundreds to thousands of dollars per year.6,7 In a poll conducted in 2019 by the Kaiser Family Foundation, 3 in 10 patients said they had not taken their medicine as prescribed because of costs.8 A mixed methods study by Ryskina et al9 found that 65% (17/26) of participants who reported primary nonadherence—intended to fill prescriptions but were unable to do so—cited cost or coverage-related barriers as the reason. With the continued rise of dermatologic drug prices and increased prevalence of high-deductible health plans, cost-effective treatment continues to grow in importance. Failure to consider cost-effective, patient-centered care may lead to increased financial toxicity, reduced adherence, and ultimately worse outcomes and patient satisfaction. We aim to review the cost-effectiveness of current prescription therapies for acne management and highlight the most cost-effective approaches to patients with mild to moderate acne as well as moderate to severe acne.
In this review, we will take a value-oriented framework.10 Value can be defined as the cost per outcome of interest. Therefore, a treatment does not necessarily need to be inexpensive to provide high value if it delivers outstanding clinical outcomes. In addition, we will focus on incremental cost-effectiveness relative to common alternatives (eg, a retinoid could deliver high value relative to a vehicle but still provide limited value compared to other available retinoids if it is more expensive but not more efficacious). When possible, we present data from cost-effectiveness studies.11,12 We also use recent available price data obtained from GoodRx on August 11, 2023, to guide this discussion.13 However, as comparative-effectiveness and cost-effectiveness studies rarely are performed for acne medications, much of this discussion will be based on expert opinion.
Treatment Categories
Topical Retinoids—There currently are 4 topical retinoids that are approved by the US Food and Drug Administration (FDA) for the treatment of acne: tretinoin, tazarotene, trifarotene, and adapalene. These drugs are vitamin A derivatives that bind retinoic acid receptors and function as comedolytic and anti-inflammatory agents.5 In general, generic tretinoin and adapalene products have the lowest cost (Table).
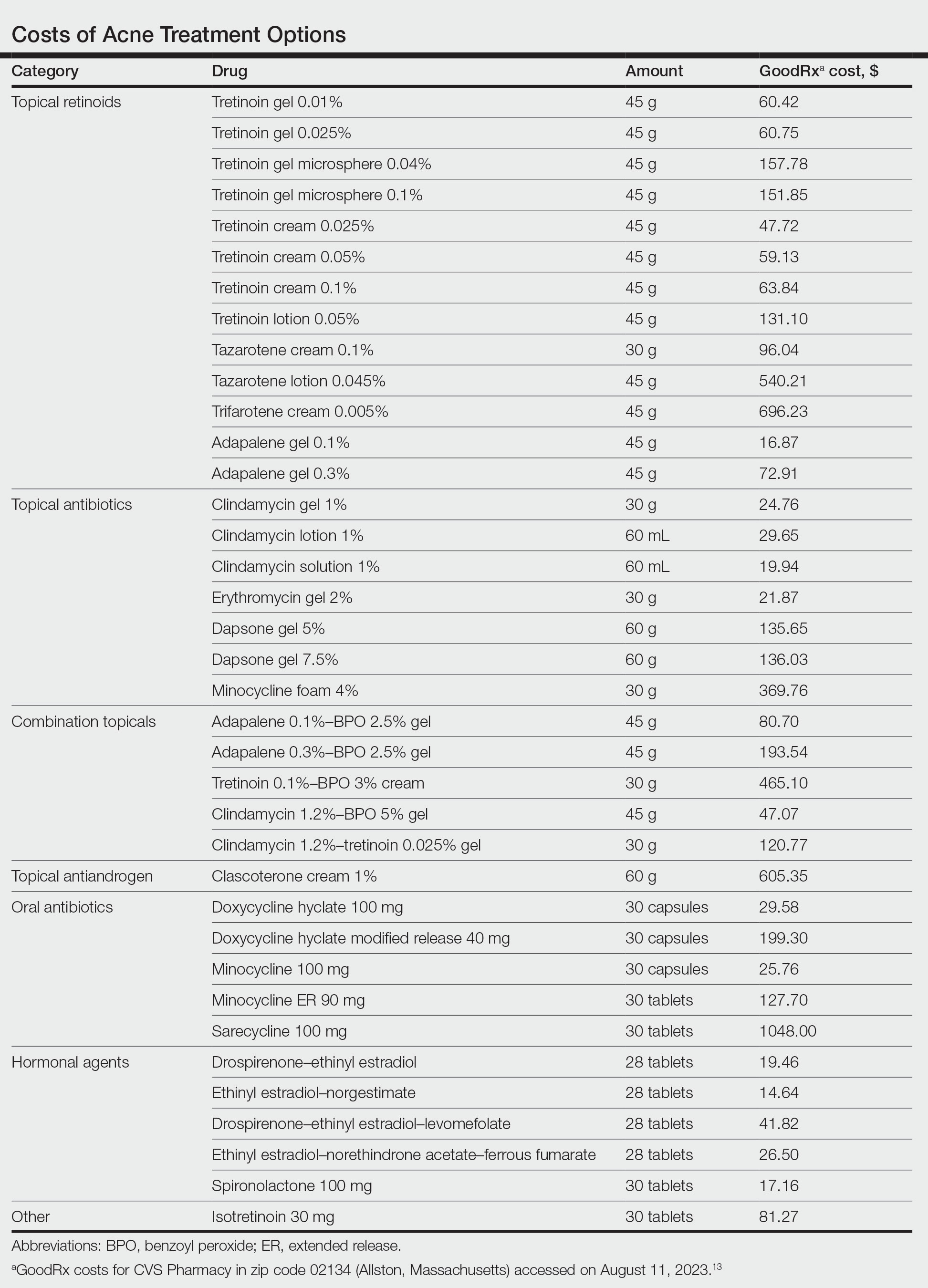
In network meta-analyses, tretinoin and adapalene often are highly ranked topical treatment options with respect to efficacy.14 Combined with their low cost, generic tretinoin and adapalene likely are excellent initial options for topical therapy from the standpoint of cost-effectiveness.15 Adapalene may be preferred in many situations because of its better photostability and compatibility with BPO.
Due to the importance of the vehicle in determining retinoid tolerability, efforts have been made to use encapsulation and polymeric emulsion technology to improve tolerability. Recently, polymeric lotion formulations of tretinoin and tazarotene have become available. In a phase 2 study, tazarotene lotion 0.045% was found to have equivalent efficacy and superior tolerability to tazarotene cream 0.1%.16 Although head-to-head data are not available, it is likely that tretinoin lotion may offer similar tolerability improvements.17 Although these formulations currently are more costly, this improved tolerability may be critical for some patients to be able to use topical retinoids, and the additional cost may be worthwhile. In addition, as these products lose market exclusivity, they may become more affordable and similarly priced to other topical retinoids. It is important to keep in mind that in clinical trials of tretinoin and adapalene, rates of dropout due to adverse events typically were 1% to 2%; therefore, because many patients can tolerate generic tretinoin and adapalene, at current prices the lotion formulations of retinoids may not be cost-effective relative to these generics.14
Trifarotene cream 0.005%, a fourth-generation topical retinoid that is highly sensitive for retinoic acid receptor γ, recently was FDA approved for the treatment of acne. Although trifarotene is efficacious for both facial and truncal acne, there is a lack of active comparator data compared to other topical retinoids.18 In a 2023 network meta-analysis, trifarotene was found to be both less efficacious and less tolerable compared to other topical retinoids.19 Thus, it is unclear if trifarotene offers any improved efficacy compared to other options, and it comes at a much higher cost (Table). In a tolerability study, trifarotene was found to be significantly more irritating than tazarotene lotion 0.045% and adapalene gel 0.3% (P<.05).20 Therefore, trifarotene cream 0.005% is unlikely to be a cost-effective option; in fact, it may be overall inferior to other topical retinoids, given its potentially lower tolerability.
Topical Antibiotics—There are 4 commonly prescribed topical antibiotics that are approved by the FDA for the treatment of acne: clindamycin, erythromycin, dapsone, and minocycline. The American Academy of Dermatology guidelines for the treatment of acne recommend concomitant use of BPO to prevent antibiotic resistance.5 Clindamycin is favored over erythromycin because of increasing antibiotic resistance to erythromycin.21 Inexpensive generic options in multiple vehicles (eg, solution, foam, gel) make clindamycin a highly cost-effective option when antibiotic therapy is desired as part of a topical regimen (Table).
The cost-effectiveness of dapsone gel and minocycline foam relative to clindamycin are less certain. Rates of resistance to minocycline are lower than clindamycin, and minocycline foam may be a reasonable alternative in patients who have not had success with other topical antibiotics, such as clindamycin.22 However, given the absence of comparative effectiveness data to suggest minocycline is more effective than clindamycin, it is difficult to justify the substantially higher cost for the typical patient. Although dapsone gel has been suggested as an option for adult women with acne, there are no data to support that it is any more effective than other topical antibiotics in this patient population.23 As generic dapsone prices decrease, it may become a reasonable alternative to clindamycin. In addition, the antineutrophil properties of dapsone may be useful in other acneform and inflammatory eruptions, such as scalp folliculitis and folliculitis decalvans.24
Combination Topicals—Current combination topical products include antibiotic and BPO, antibiotic and retinoid, and retinoid and BPO. Use of combination agents is recommended to reduce the risk for resistance and to enhance effectiveness. Combination products offer improved convenience, which is associated with better adherence and outcomes.25 Generic fixed-dose adapalene-BPO can be a highly cost-effective option that can sometimes be less expensive than the individual component products (Table). Similarly, fixed-dose clindamycin-BPO also is likely to be highly cost-effective. A network meta-analysis found fixed-dose adapalene-BPO to be the most efficacious topical treatment, though it also was found to be the most irritating—more so than fixed-dose clindamycin-BPO, which may have similar efficacy.14,26,27 Generic fixed-dose tretinoin-clindamycin offers improved convenience and adherence compared to the individual components, but it is more expensive, and its cost-effectiveness may be influenced by the importance of convenience for the patient.25 An encapsulated, fixed-dose tretinoin 0.1%–BPO 3% cream is FDA approved for acne, but the cost is high and there is a lack of comparative effectiveness data demonstrating advantages over generic fixed-dose adapalene-BPO products.
Topical Antiandrogen—Clascoterone was introduced in 2020 as the first FDA-approved topical medication to target the hormonal pathogenesis of acne, inhibiting the androgen receptors in the sebaceous gland.28 Because it is rapidly metabolized to cortexolone and does not have systemic antiandrogen effects, clascoterone can be used in both men and women with acne. In clinical trials, it had minimal side effects, including no evidence of irritability, which is an advantage over topical retinoids and BPO.29 In addition, a phase 2 study found that clascoterone may have similar to superior efficacy to tretinoin cream 0.05%.30 Although clascoterone has several strengths, including its efficacy, tolerability, and unique mechanism of action, its cost-effectiveness is limited due to its high cost (Table) and the need for twice-daily application, which reduces convenience. Clascoterone likely is best reserved for patients with a strong hormonal pathogenesis of their acne or difficulty tolerating other topicals, or as an additional therapy to complement other topicals.
Oral Antibiotics—Oral antibiotics are the most commonly prescribed systemic treatments for acne, particularly tetracyclines such as doxycycline, minocycline, and sarecycline.31-34 Doxycycline and minocycline are considered first-line oral antibiotic therapy in the United States and are inexpensive and easily accessible.5 Doxycycline generally is recommended over minocycline given lack of evidence of superior efficacy of minocycline and concerns about severe adverse cutaneous reactions and drug-induced lupus with minocycline.35
In recent years, there has been growing concern of the development of antibiotic resistance.5 Sarecycline is a narrow-spectrum tetracycline that was FDA approved for acne in 2018. In vitro studies demonstrate sarecycline maintains high efficacy against C acnes with less activity against other bacteria, particularly gram-negative enterobes.36 The selectivity of sarecycline may lessen alterations of the gut microbiome seen with other oral antibiotics and reduce gastrointestinal tract side effects. Although comparative effectiveness studies are lacking, sarecycline was efficacious in phase 3 trials with few side effects compared with placebo.37 However, at this time, given the absence of comparative effectiveness data and its high cost (Table), sarecycline likely is best reserved for patients with comorbidities (eg, gastrointestinal disease), those requiring long-term antibiotic therapy, or those with acne that has failed to respond to other oral antibiotics.
Hormonal Treatments—Hormonal treatments such as combined oral contraceptives (COCs) and spironolactone often are considered second-line options, though they may represent cost-effective and safe alternatives to oral antibiotics for women with moderate to severe acne.38-41 There currently are 4 COCs approved by the FDA for the treatment of moderate acne in postmenarcheal females: drospirenone-ethinyl estradiol (Yaz [Bayer HealthCare Pharmaceuticals, Inc]), ethinyl estradiol-norgestimate (Ortho Tri-Cyclen [Ortho-McNeil Pharmaceuticals, Inc]), drospirenone-ethinyl estradiol-levomefolate (Beyaz [Bayer HealthCare Pharmaceuticals, Inc]), and ethinyl estradiol-norethindrone acetate-ferrous fumarate (Estrostep Fe [Allergan USA, Inc]).5 Treatment with COCs has been shown to cause substantial reductions in lesion counts across all lesion types compared to placebo, and a meta-analysis of 24 randomized trials conducted by Arowojolu et al42 demonstrated no consistent differences in acne reduction among different COCs.43,44 Although oral antibiotics are associated with faster improvement than COCs, there is some evidence that they have similar efficacy at 6 months of therapy.45 Combined oral contraceptives are inexpensive and likely reflect a highly cost-effective option (Table).
Spironolactone is an aldosterone inhibitor and androgen receptor blocker that is used off label to treat acne. It is one of the least expensive systemic medications for acne (Table). Although randomized controlled trials are lacking, several large case series support the effectiveness of spironolactone for women with acne.38,46 In addition, observational data suggest spironolactone may have similar effectiveness to oral antibiotics.41 Spironolactone generally is well tolerated, with the most common adverse effects being menstrual irregularities, breast tenderness, and diuresis.47,48 Many of these adverse effects are dose dependent and less likely with the dosing used in acne care. Additionally, menstrual irregularities can be reduced by concomitant use of a COC.48
Although frequent potassium monitoring remains common among patients being treated with spironolactone, there is growing evidence to suggest that potassium monitoring is of low value in young healthy women with acne.49-51 Reducing this laboratory monitoring likely represents an opportunity to provide higher-value care to patients being treated with spironolactone. However, laboratory monitoring should be considered if risk factors for hyperkalemia are present (eg, older age, comorbidities, medications).51
Isotretinoin—Isotretinoin is the most efficacious treatment available for acne and has the unique property of being able to induce a remission of acne activity for many patients.5 Although it remains modestly expensive (Table), it may be less costly overall relative to other treatments that may need continued use over many years because it can induce a remission of acne activity. As with spironolactone, frequent laboratory monitoring remains common among patients being treated with isotretinoin. There is no evidence to support checking complete blood cell counts.52 Several observational studies and a Delphi consensus support reduced monitoring, such as checking lipids and alanine aminotransferase at baseline and peak dose in otherwise young healthy patients.53,54 A recent critically appraised topic published in the British Journal of Dermatology has proposed eliminating laboratory monitoring entirely.55 Reducing laboratory monitoring for patients being treated with isotretinoin has been estimated to potentially save $100 million to $200 million per year in the United States.52-54
Other Strategies to Reduce Patient Costs
Although choosing a cost-effective treatment approach is critical to preventing financial toxicity given poor coverage for acne care and the growth of high-deductible insurance plans, some patients may still experience high treatment costs.56 Because pharmacy costs often are inflated, potentially related to practices of pharmacy benefit managers, it often is possible to find better prices than the presented list price, either by using platforms such as GoodRx or through direct-to-patient mail-order pharmacies such as Cost Plus Drug.57 For branded medications, some patients may be eligible for patient-assistance programs, though they typically are not available for those with public insurance such as Medicare or Medicaid. Compounding pharmacies offer another approach to reduce cost and improve convenience for patients, but because the vehicle can influence the efficacy and tolerability of some topical medications, it is possible that these compounded formulations may not perform similarly to the original FDA-approved products.
Conclusion
For mild to moderate acne, multimodal topical therapy often is required. Fixed-dose combination adapalene-BPO and clindamycin-BPO are highly cost-effective options for most patients. Lotion formulations of topical retinoids may be useful in patients with difficulty tolerating other formulations. Clascoterone is a novel topical antiandrogen that is more expensive than other topical therapies but can complement other topical therapies and is well tolerated.
For moderate to severe acne, doxycycline or hormonal therapy (ie, COCs, spironolactone) are highly cost-effective options. Isotretinoin is recommended for severe or scarring acne. Reduced laboratory monitoring for spironolactone and isotretinoin is an opportunity to provide higher-value care.
- Bhate K, Williams HC. Epidemiology of acne vulgaris. Br J Dermatol. 2013;168:474-485. doi:10.1111/bjd.12149
- Collier CN, Harper JC, Cafardi JA, et al. The prevalence of acne in adults 20 years and older. J Am Acad Dermatol. 2008;58:56-59. doi:10.1016/j.jaad.2007.06.045
- Webster GF. The pathophysiology of acne. Cutis. 2005;76(2 suppl):4-7.
- Degitz K, Placzek M, Borelli C, et al. Pathophysiology of acne. J Dtsch Dermatol Ges. 2007;5:316-323. doi:10.1111/j.1610-0387.2007.06274.x
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.e33. doi:10.1016/j.jaad.2015.12.037
- Felmingham C, Kerr A, Veysey E. Costs incurred by patients with acne prior to dermatological consultation and their relation to patient income. Australas J Dermatol. 2020;61:384-386. doi:10.1111/ajd.13324
- Perche P, Singh R, Feldman S. Patient preferences for acne vulgaris treatment and barriers to care: a survey study. J Drugs Dermatol. 2022;21:1191-1195. doi:10.36849/JDD.6940
- KFF Health Tracking Poll—February 2019. Accessed August 9, 2023. https://files.kff.org/attachment/Topline-KFF-Health-Tracking-Poll-February-2019
- Ryskina KL, Goldberg E, Lott B, et al. The role of the physician in patient perceptions of barriers to primary adherence with acne medications. JAMA Dermatol. 2018;154:456-459. doi:10.1001/jamadermatol.2017.6144
- Porter ME. What is value in health care? N Engl J Med. 2010;363:2477-2481. doi:10.1056/NEJMp1011024
- Barbieri JS, Tan JKL, Adamson AS. Active comparator trial designs used to promote development of innovative new medications. Cutis. 2020;106:E4-E6. doi:10.12788/cutis.0067
- Miller J, Ly S, Mostaghimi A, et al. Use of active comparator trials for topical medications in dermatology. JAMA Dermatol. 2021;157:597-599. doi:10.1001/jamadermatol.2021.0356
- GoodRx. Accessed August 11, 2023. https://www.goodrx.com
- Stuart B, Maund E, Wilcox C, et al. Topical preparations for the treatment of mild‐to‐moderate acne vulgaris: systematic review and network meta‐analysis. Br J Dermatol. 2021;185:512-525. doi:10.1111/bjd.20080
- Mavranezouli I, Welton NJ, Daly CH, et al. Cost-effectiveness of topical pharmacological, oral pharmacological, physical and combined treatments for acne vulgaris. Clin Exp Dermatol. 2022;47:2176-2187. doi:10.1111/ced.15356
- Tanghetti E, Werschler W, Lain T, et al. Tazarotene 0.045% lotion for once-daily treatment of moderate-to-severe acne vulgaris: results from two phase 3 trials. J Drugs Dermatol. 2020;19:70-77. doi:10.36849/JDD.2020.3977
- Tyring SK, Kircik LH, Pariser DM, et al. Novel tretinoin 0.05% lotion for the once-daily treatment of moderate-to-severe acne vulgaris: assessment of efficacy and safety in patients aged 9 years and older. J Drugs Dermatol. 2018;17:1084-1091.
- Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 μg/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80:1691-1699. doi:10.1016/j.jaad.2019.02.044
- Huang CY, Chang IJ, Bolick N, et al. Comparative efficacy of pharmacological treatments for acne vulgaris: a network meta-analysis of 221 randomized controlled trials. Ann Fam Med. 2023;21:358-369. doi:10.1370/afm.2995
- Draelos ZD. Low irritation potential of tazarotene 0.045% lotion: head-to-head comparison to adapalene 0.3% gel and trifarotene 0.005% cream in two studies. J Dermatolog Treat. 2023;34:2166346. doi:10.1080/09546634.2023.2166346
- Dessinioti C, Katsambas A. Antibiotics and antimicrobial resistance in acne: epidemiological trends and clinical practice considerations. Yale J Biol Med. 2022;95:429-443.
- Gold LS, Dhawan S, Weiss J, et al. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80:168-177. doi:10.1016/j.jaad.2018.08.020
- Wang X, Wang Z, Sun L, et al. Efficacy and safety of dapsone gel for acne: a systematic review and meta-analysis. Ann Palliat Med. 2022;11:611-620. doi:10.21037/apm-21-3935
- Melián-Olivera A, Burgos-Blasco P, Selda-Enríquez G, et al. Topical dapsone for folliculitis decalvans: a retrospective cohort study. J Am Acad Dermatol. 2022;87:150-151. doi:10.1016/j.jaad.2021.07.004
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86:103-108.
- Ting W. Randomized, observer-blind, split-face study to compare the irritation potential of 2 topical acne formulations over a 14-day treatment period. Cutis. 2012;90:91-96.
- Aschoff R, Möller S, Haase R, et al. Tolerability and efficacy ofclindamycin/tretinoin versus adapalene/benzoyl peroxide in the treatment of acne vulgaris. J Drugs Dermatol. 2021;20:295-301. doi:10.36849/JDD.2021.5641
- Rosette C, Agan FJ, Mazzetti A, et al. Cortexolone 17α-propionate (clascoterone) is a novel androgen receptor antagonist that inhibits production of lipids and inflammatory cytokines from sebocytes in vitro. J Drugs Dermatol. 2019;18:412-418.
- Hebert A, Thiboutot D, Stein Gold L, et al. Efficacy and safety of topical clascoterone cream, 1%, for treatment in patients with facial acne: two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156:621-630. doi:10.1001/jamadermatol.2020.0465
- Trifu V, Tiplica GS, Naumescu E, et al. Cortexolone 17α-propionate 1% cream, a new potent antiandrogen for topical treatment of acne vulgaris. a pilot randomized, double-blind comparative study vs. placebo and tretinoin 0·05% cream. Br J Dermatol. 2011;165:177-183. doi:10.1111/j.1365-2133.2011.10332.x
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Guzman AK, Barbieri JS. Comparative analysis of prescribing patterns of tetracycline class antibiotics and spironolactone between advanced practice providers and physicians in the treatment of acne vulgaris. J Am Acad Dermatol. 2021;84:1119-1121. doi:10.1016/j.jaad.2020.06.044
- Barbieri JS, James WD, Margolis DJ. Trends in prescribing behavior of systemic agents used in the treatment of acne among dermatologists and nondermatologists: a retrospective analysis, 2004-2013. J Am Acad Dermatol. 2017;77:456-463.e4. doi:10.1016/j.jaad.2017.04.016
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016. JAMA Dermatol. 2019;155:290-297. doi:10.1001/jamadermatol.2018.4944
- Garner SE, Eady A, Bennett C, et al. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012;2012:CD002086. doi:10.1002/14651858.CD002086.pub2
- Zhanel G, Critchley I, Lin LY, et al. Microbiological profile of sarecycline, a novel targeted spectrum tetracycline for the treatment of acne vulgaris. Antimicrob Agents Chemother. 2018;63:e01297-18. doi:10.1128/AAC.01297-18
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987-996.
- Garg V, Choi JK, James WD, et al. Long-term use of spironolactone for acne in women: a case series of 403 patients. J Am Acad Dermatol. 2021;84:1348-1355. doi:10.1016/j.jaad.2020.12.071
- Barbieri JS, Choi JK, James WD, et al. Real-world drug usage survival of spironolactone versus oral antibiotics for the management of female patients with acne. J Am Acad Dermatol. 2019;81:848-851. doi:10.1016/j.jaad.2019.03.036
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. 2019;80:538-549. doi:10.1016/j.jaad.2018.09.055
- Barbieri JS, Choi JK, Mitra N, et al. Frequency of treatment switching for spironolactone compared to oral tetracycline-class antibiotics for women with acne: a retrospective cohort study 2010-2016. J Drugs Dermatol. 2018;17:632-638.
- Arowojolu AO, Gallo MF, Lopez LM, et al. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;7:CD004425. doi:10.1002/14651858.CD004425.pub6
- Maloney JM, Dietze P, Watson D, et al. Treatment of acne using a 3-milligram drospirenone/20-microgram ethinyl estradiol oral contraceptive administered in a 24/4 regimen. Obstet Gynecol. 2008;112:773-781. doi:10.1097/AOG.0b013e318187e1c5
- Lucky AW, Koltun W, Thiboutot D, et al. A combined oral contraceptive containing 3-mg drospirenone/20-microg ethinyl estradiol in the treatment of acne vulgaris: a randomized, double-blind, placebo-controlled study evaluating lesion counts and participant self-assessment. Cutis. 2008;82:143-150.
- Koo EB, Petersen TD, Kimball AB. Meta-analysis comparing efficacy of antibiotics versus oral contraceptives in acne vulgaris. J Am Acad Dermatol. 2014;71:450-459. doi:10.1016/j.jaad.2014.03.051
- Roberts EE, Nowsheen S, Davis DMR, et al. Use of spironolactone to treat acne in adolescent females. Pediatr Dermatol. 2021;38:72-76. doi:10.1111/pde.14391
- Shaw JC. Low-dose adjunctive spironolactone in the treatment of acne in women: a retrospective analysis of 85 consecutively treated patients. J Am Acad Dermatol. 2000;43:498-502. doi:10.1067/mjd.2000.105557
- Layton AM, Eady EA, Whitehouse H, et al. Oral spironolactone for acne vulgaris in adult females: a hybrid systematic review. Am J Clin Dermatol. 2017;18:169-191. doi:10.1007/s40257-016-0245-x
- Barbieri JS, Margolis DJ, Mostaghimi A. Temporal trends and clinician variability in potassium monitoring of healthy young women treated for acne with spironolactone. JAMA Dermatol. 2021;157:296-300. doi:10.1001/jamadermatol.2020.5468
- Plovanich M, Weng QY, Mostaghimi A. Low usefulness of potassium monitoring among healthy young women taking spironolactone for acne. JAMA Dermatol. 2015;151:941-944. doi:10.1001/jamadermatol.2015.34
- Thiede RM, Rastogi S, Nardone B, et al. Hyperkalemia in women with acne exposed to oral spironolactone: a retrospective study from the RADAR (Research on Adverse Drug Events and Reports) program. Int J Womens Dermatol. 2019;5:155-157. doi:10.1016/j.ijwd.2019.04.024
- Barbieri JS, Shin DB, Wang S, et al. The clinical utility of laboratory monitoring during isotretinoin therapy for acne and changes to monitoring practices over time. J Am Acad Dermatol. 2020;82:72-79. doi:10.1016/j.jaad.2019.06.025
- Lee YH, Scharnitz TP, Muscat J, et al. Laboratory monitoring during isotretinoin therapy for acne: a systematic review and meta-analysis. JAMA Dermatol. 2016;152:35-44. doi:10.1001/jamadermatol.2015.3091
- Xia E, Han J, Faletsky A, et al. Isotretinoin laboratory monitoring in acne treatment: a Delphi consensus study. JAMA Dermatol. 2022;158:942-948. doi:10.1001/jamadermatol.2022.2044
- Affleck A, Jackson D, Williams HC, et al. Is routine laboratory testing in healthy young patients taking isotretinoin necessary: a critically appraised topic. Br J Dermatol. 2022;187:857-865. doi:10.1111/bjd.21840
- Barbieri JS, LaChance A, Albrecht J. Double standards and inconsistencies in access to care-what constitutes a cosmetic treatment? JAMA Dermatol. 2023;159:245-246. doi:10.1001/jamadermatol.2022.6322
- Trish E, Van Nuys K, Popovian R. US consumers overpay for generic drugs. Schaeffer Center White Paper Series. May 31, 2022. doi:10.25549/m589-2268
In the United States, acne affects 85% of adolescents and can persist into adulthood at a prevalence of 30% to 50% in adult women. 1,2 The pathogenesis of acne is multifactorial and involves hyperkeratinization of the follicle, bacterial colonization with Cutibacterium acnes , and increased androgen-induced sebum production, which together lead to inflammation. 3,4 A wide range of treatment guideline–recommended options are available, including benzoyl peroxide (BPO), topical retinoids, topical and oral antibiotics, antiandrogens, and isotretinoin. 5 However, these options vary widely in their clinical uses, effectiveness, and costs.
Why Cost-effective Acne Care Matters
Out-of-pocket spending by patients on acne treatments can be substantial, with surveys finding that acne patients often spend hundreds to thousands of dollars per year.6,7 In a poll conducted in 2019 by the Kaiser Family Foundation, 3 in 10 patients said they had not taken their medicine as prescribed because of costs.8 A mixed methods study by Ryskina et al9 found that 65% (17/26) of participants who reported primary nonadherence—intended to fill prescriptions but were unable to do so—cited cost or coverage-related barriers as the reason. With the continued rise of dermatologic drug prices and increased prevalence of high-deductible health plans, cost-effective treatment continues to grow in importance. Failure to consider cost-effective, patient-centered care may lead to increased financial toxicity, reduced adherence, and ultimately worse outcomes and patient satisfaction. We aim to review the cost-effectiveness of current prescription therapies for acne management and highlight the most cost-effective approaches to patients with mild to moderate acne as well as moderate to severe acne.
In this review, we will take a value-oriented framework.10 Value can be defined as the cost per outcome of interest. Therefore, a treatment does not necessarily need to be inexpensive to provide high value if it delivers outstanding clinical outcomes. In addition, we will focus on incremental cost-effectiveness relative to common alternatives (eg, a retinoid could deliver high value relative to a vehicle but still provide limited value compared to other available retinoids if it is more expensive but not more efficacious). When possible, we present data from cost-effectiveness studies.11,12 We also use recent available price data obtained from GoodRx on August 11, 2023, to guide this discussion.13 However, as comparative-effectiveness and cost-effectiveness studies rarely are performed for acne medications, much of this discussion will be based on expert opinion.
Treatment Categories
Topical Retinoids—There currently are 4 topical retinoids that are approved by the US Food and Drug Administration (FDA) for the treatment of acne: tretinoin, tazarotene, trifarotene, and adapalene. These drugs are vitamin A derivatives that bind retinoic acid receptors and function as comedolytic and anti-inflammatory agents.5 In general, generic tretinoin and adapalene products have the lowest cost (Table).

In network meta-analyses, tretinoin and adapalene often are highly ranked topical treatment options with respect to efficacy.14 Combined with their low cost, generic tretinoin and adapalene likely are excellent initial options for topical therapy from the standpoint of cost-effectiveness.15 Adapalene may be preferred in many situations because of its better photostability and compatibility with BPO.
Due to the importance of the vehicle in determining retinoid tolerability, efforts have been made to use encapsulation and polymeric emulsion technology to improve tolerability. Recently, polymeric lotion formulations of tretinoin and tazarotene have become available. In a phase 2 study, tazarotene lotion 0.045% was found to have equivalent efficacy and superior tolerability to tazarotene cream 0.1%.16 Although head-to-head data are not available, it is likely that tretinoin lotion may offer similar tolerability improvements.17 Although these formulations currently are more costly, this improved tolerability may be critical for some patients to be able to use topical retinoids, and the additional cost may be worthwhile. In addition, as these products lose market exclusivity, they may become more affordable and similarly priced to other topical retinoids. It is important to keep in mind that in clinical trials of tretinoin and adapalene, rates of dropout due to adverse events typically were 1% to 2%; therefore, because many patients can tolerate generic tretinoin and adapalene, at current prices the lotion formulations of retinoids may not be cost-effective relative to these generics.14
Trifarotene cream 0.005%, a fourth-generation topical retinoid that is highly sensitive for retinoic acid receptor γ, recently was FDA approved for the treatment of acne. Although trifarotene is efficacious for both facial and truncal acne, there is a lack of active comparator data compared to other topical retinoids.18 In a 2023 network meta-analysis, trifarotene was found to be both less efficacious and less tolerable compared to other topical retinoids.19 Thus, it is unclear if trifarotene offers any improved efficacy compared to other options, and it comes at a much higher cost (Table). In a tolerability study, trifarotene was found to be significantly more irritating than tazarotene lotion 0.045% and adapalene gel 0.3% (P<.05).20 Therefore, trifarotene cream 0.005% is unlikely to be a cost-effective option; in fact, it may be overall inferior to other topical retinoids, given its potentially lower tolerability.
Topical Antibiotics—There are 4 commonly prescribed topical antibiotics that are approved by the FDA for the treatment of acne: clindamycin, erythromycin, dapsone, and minocycline. The American Academy of Dermatology guidelines for the treatment of acne recommend concomitant use of BPO to prevent antibiotic resistance.5 Clindamycin is favored over erythromycin because of increasing antibiotic resistance to erythromycin.21 Inexpensive generic options in multiple vehicles (eg, solution, foam, gel) make clindamycin a highly cost-effective option when antibiotic therapy is desired as part of a topical regimen (Table).
The cost-effectiveness of dapsone gel and minocycline foam relative to clindamycin are less certain. Rates of resistance to minocycline are lower than clindamycin, and minocycline foam may be a reasonable alternative in patients who have not had success with other topical antibiotics, such as clindamycin.22 However, given the absence of comparative effectiveness data to suggest minocycline is more effective than clindamycin, it is difficult to justify the substantially higher cost for the typical patient. Although dapsone gel has been suggested as an option for adult women with acne, there are no data to support that it is any more effective than other topical antibiotics in this patient population.23 As generic dapsone prices decrease, it may become a reasonable alternative to clindamycin. In addition, the antineutrophil properties of dapsone may be useful in other acneform and inflammatory eruptions, such as scalp folliculitis and folliculitis decalvans.24
Combination Topicals—Current combination topical products include antibiotic and BPO, antibiotic and retinoid, and retinoid and BPO. Use of combination agents is recommended to reduce the risk for resistance and to enhance effectiveness. Combination products offer improved convenience, which is associated with better adherence and outcomes.25 Generic fixed-dose adapalene-BPO can be a highly cost-effective option that can sometimes be less expensive than the individual component products (Table). Similarly, fixed-dose clindamycin-BPO also is likely to be highly cost-effective. A network meta-analysis found fixed-dose adapalene-BPO to be the most efficacious topical treatment, though it also was found to be the most irritating—more so than fixed-dose clindamycin-BPO, which may have similar efficacy.14,26,27 Generic fixed-dose tretinoin-clindamycin offers improved convenience and adherence compared to the individual components, but it is more expensive, and its cost-effectiveness may be influenced by the importance of convenience for the patient.25 An encapsulated, fixed-dose tretinoin 0.1%–BPO 3% cream is FDA approved for acne, but the cost is high and there is a lack of comparative effectiveness data demonstrating advantages over generic fixed-dose adapalene-BPO products.
Topical Antiandrogen—Clascoterone was introduced in 2020 as the first FDA-approved topical medication to target the hormonal pathogenesis of acne, inhibiting the androgen receptors in the sebaceous gland.28 Because it is rapidly metabolized to cortexolone and does not have systemic antiandrogen effects, clascoterone can be used in both men and women with acne. In clinical trials, it had minimal side effects, including no evidence of irritability, which is an advantage over topical retinoids and BPO.29 In addition, a phase 2 study found that clascoterone may have similar to superior efficacy to tretinoin cream 0.05%.30 Although clascoterone has several strengths, including its efficacy, tolerability, and unique mechanism of action, its cost-effectiveness is limited due to its high cost (Table) and the need for twice-daily application, which reduces convenience. Clascoterone likely is best reserved for patients with a strong hormonal pathogenesis of their acne or difficulty tolerating other topicals, or as an additional therapy to complement other topicals.
Oral Antibiotics—Oral antibiotics are the most commonly prescribed systemic treatments for acne, particularly tetracyclines such as doxycycline, minocycline, and sarecycline.31-34 Doxycycline and minocycline are considered first-line oral antibiotic therapy in the United States and are inexpensive and easily accessible.5 Doxycycline generally is recommended over minocycline given lack of evidence of superior efficacy of minocycline and concerns about severe adverse cutaneous reactions and drug-induced lupus with minocycline.35
In recent years, there has been growing concern of the development of antibiotic resistance.5 Sarecycline is a narrow-spectrum tetracycline that was FDA approved for acne in 2018. In vitro studies demonstrate sarecycline maintains high efficacy against C acnes with less activity against other bacteria, particularly gram-negative enterobes.36 The selectivity of sarecycline may lessen alterations of the gut microbiome seen with other oral antibiotics and reduce gastrointestinal tract side effects. Although comparative effectiveness studies are lacking, sarecycline was efficacious in phase 3 trials with few side effects compared with placebo.37 However, at this time, given the absence of comparative effectiveness data and its high cost (Table), sarecycline likely is best reserved for patients with comorbidities (eg, gastrointestinal disease), those requiring long-term antibiotic therapy, or those with acne that has failed to respond to other oral antibiotics.
Hormonal Treatments—Hormonal treatments such as combined oral contraceptives (COCs) and spironolactone often are considered second-line options, though they may represent cost-effective and safe alternatives to oral antibiotics for women with moderate to severe acne.38-41 There currently are 4 COCs approved by the FDA for the treatment of moderate acne in postmenarcheal females: drospirenone-ethinyl estradiol (Yaz [Bayer HealthCare Pharmaceuticals, Inc]), ethinyl estradiol-norgestimate (Ortho Tri-Cyclen [Ortho-McNeil Pharmaceuticals, Inc]), drospirenone-ethinyl estradiol-levomefolate (Beyaz [Bayer HealthCare Pharmaceuticals, Inc]), and ethinyl estradiol-norethindrone acetate-ferrous fumarate (Estrostep Fe [Allergan USA, Inc]).5 Treatment with COCs has been shown to cause substantial reductions in lesion counts across all lesion types compared to placebo, and a meta-analysis of 24 randomized trials conducted by Arowojolu et al42 demonstrated no consistent differences in acne reduction among different COCs.43,44 Although oral antibiotics are associated with faster improvement than COCs, there is some evidence that they have similar efficacy at 6 months of therapy.45 Combined oral contraceptives are inexpensive and likely reflect a highly cost-effective option (Table).
Spironolactone is an aldosterone inhibitor and androgen receptor blocker that is used off label to treat acne. It is one of the least expensive systemic medications for acne (Table). Although randomized controlled trials are lacking, several large case series support the effectiveness of spironolactone for women with acne.38,46 In addition, observational data suggest spironolactone may have similar effectiveness to oral antibiotics.41 Spironolactone generally is well tolerated, with the most common adverse effects being menstrual irregularities, breast tenderness, and diuresis.47,48 Many of these adverse effects are dose dependent and less likely with the dosing used in acne care. Additionally, menstrual irregularities can be reduced by concomitant use of a COC.48
Although frequent potassium monitoring remains common among patients being treated with spironolactone, there is growing evidence to suggest that potassium monitoring is of low value in young healthy women with acne.49-51 Reducing this laboratory monitoring likely represents an opportunity to provide higher-value care to patients being treated with spironolactone. However, laboratory monitoring should be considered if risk factors for hyperkalemia are present (eg, older age, comorbidities, medications).51
Isotretinoin—Isotretinoin is the most efficacious treatment available for acne and has the unique property of being able to induce a remission of acne activity for many patients.5 Although it remains modestly expensive (Table), it may be less costly overall relative to other treatments that may need continued use over many years because it can induce a remission of acne activity. As with spironolactone, frequent laboratory monitoring remains common among patients being treated with isotretinoin. There is no evidence to support checking complete blood cell counts.52 Several observational studies and a Delphi consensus support reduced monitoring, such as checking lipids and alanine aminotransferase at baseline and peak dose in otherwise young healthy patients.53,54 A recent critically appraised topic published in the British Journal of Dermatology has proposed eliminating laboratory monitoring entirely.55 Reducing laboratory monitoring for patients being treated with isotretinoin has been estimated to potentially save $100 million to $200 million per year in the United States.52-54
Other Strategies to Reduce Patient Costs
Although choosing a cost-effective treatment approach is critical to preventing financial toxicity given poor coverage for acne care and the growth of high-deductible insurance plans, some patients may still experience high treatment costs.56 Because pharmacy costs often are inflated, potentially related to practices of pharmacy benefit managers, it often is possible to find better prices than the presented list price, either by using platforms such as GoodRx or through direct-to-patient mail-order pharmacies such as Cost Plus Drug.57 For branded medications, some patients may be eligible for patient-assistance programs, though they typically are not available for those with public insurance such as Medicare or Medicaid. Compounding pharmacies offer another approach to reduce cost and improve convenience for patients, but because the vehicle can influence the efficacy and tolerability of some topical medications, it is possible that these compounded formulations may not perform similarly to the original FDA-approved products.
Conclusion
For mild to moderate acne, multimodal topical therapy often is required. Fixed-dose combination adapalene-BPO and clindamycin-BPO are highly cost-effective options for most patients. Lotion formulations of topical retinoids may be useful in patients with difficulty tolerating other formulations. Clascoterone is a novel topical antiandrogen that is more expensive than other topical therapies but can complement other topical therapies and is well tolerated.
For moderate to severe acne, doxycycline or hormonal therapy (ie, COCs, spironolactone) are highly cost-effective options. Isotretinoin is recommended for severe or scarring acne. Reduced laboratory monitoring for spironolactone and isotretinoin is an opportunity to provide higher-value care.
In the United States, acne affects 85% of adolescents and can persist into adulthood at a prevalence of 30% to 50% in adult women. 1,2 The pathogenesis of acne is multifactorial and involves hyperkeratinization of the follicle, bacterial colonization with Cutibacterium acnes , and increased androgen-induced sebum production, which together lead to inflammation. 3,4 A wide range of treatment guideline–recommended options are available, including benzoyl peroxide (BPO), topical retinoids, topical and oral antibiotics, antiandrogens, and isotretinoin. 5 However, these options vary widely in their clinical uses, effectiveness, and costs.
Why Cost-effective Acne Care Matters
Out-of-pocket spending by patients on acne treatments can be substantial, with surveys finding that acne patients often spend hundreds to thousands of dollars per year.6,7 In a poll conducted in 2019 by the Kaiser Family Foundation, 3 in 10 patients said they had not taken their medicine as prescribed because of costs.8 A mixed methods study by Ryskina et al9 found that 65% (17/26) of participants who reported primary nonadherence—intended to fill prescriptions but were unable to do so—cited cost or coverage-related barriers as the reason. With the continued rise of dermatologic drug prices and increased prevalence of high-deductible health plans, cost-effective treatment continues to grow in importance. Failure to consider cost-effective, patient-centered care may lead to increased financial toxicity, reduced adherence, and ultimately worse outcomes and patient satisfaction. We aim to review the cost-effectiveness of current prescription therapies for acne management and highlight the most cost-effective approaches to patients with mild to moderate acne as well as moderate to severe acne.
In this review, we will take a value-oriented framework.10 Value can be defined as the cost per outcome of interest. Therefore, a treatment does not necessarily need to be inexpensive to provide high value if it delivers outstanding clinical outcomes. In addition, we will focus on incremental cost-effectiveness relative to common alternatives (eg, a retinoid could deliver high value relative to a vehicle but still provide limited value compared to other available retinoids if it is more expensive but not more efficacious). When possible, we present data from cost-effectiveness studies.11,12 We also use recent available price data obtained from GoodRx on August 11, 2023, to guide this discussion.13 However, as comparative-effectiveness and cost-effectiveness studies rarely are performed for acne medications, much of this discussion will be based on expert opinion.
Treatment Categories
Topical Retinoids—There currently are 4 topical retinoids that are approved by the US Food and Drug Administration (FDA) for the treatment of acne: tretinoin, tazarotene, trifarotene, and adapalene. These drugs are vitamin A derivatives that bind retinoic acid receptors and function as comedolytic and anti-inflammatory agents.5 In general, generic tretinoin and adapalene products have the lowest cost (Table).

In network meta-analyses, tretinoin and adapalene often are highly ranked topical treatment options with respect to efficacy.14 Combined with their low cost, generic tretinoin and adapalene likely are excellent initial options for topical therapy from the standpoint of cost-effectiveness.15 Adapalene may be preferred in many situations because of its better photostability and compatibility with BPO.
Due to the importance of the vehicle in determining retinoid tolerability, efforts have been made to use encapsulation and polymeric emulsion technology to improve tolerability. Recently, polymeric lotion formulations of tretinoin and tazarotene have become available. In a phase 2 study, tazarotene lotion 0.045% was found to have equivalent efficacy and superior tolerability to tazarotene cream 0.1%.16 Although head-to-head data are not available, it is likely that tretinoin lotion may offer similar tolerability improvements.17 Although these formulations currently are more costly, this improved tolerability may be critical for some patients to be able to use topical retinoids, and the additional cost may be worthwhile. In addition, as these products lose market exclusivity, they may become more affordable and similarly priced to other topical retinoids. It is important to keep in mind that in clinical trials of tretinoin and adapalene, rates of dropout due to adverse events typically were 1% to 2%; therefore, because many patients can tolerate generic tretinoin and adapalene, at current prices the lotion formulations of retinoids may not be cost-effective relative to these generics.14
Trifarotene cream 0.005%, a fourth-generation topical retinoid that is highly sensitive for retinoic acid receptor γ, recently was FDA approved for the treatment of acne. Although trifarotene is efficacious for both facial and truncal acne, there is a lack of active comparator data compared to other topical retinoids.18 In a 2023 network meta-analysis, trifarotene was found to be both less efficacious and less tolerable compared to other topical retinoids.19 Thus, it is unclear if trifarotene offers any improved efficacy compared to other options, and it comes at a much higher cost (Table). In a tolerability study, trifarotene was found to be significantly more irritating than tazarotene lotion 0.045% and adapalene gel 0.3% (P<.05).20 Therefore, trifarotene cream 0.005% is unlikely to be a cost-effective option; in fact, it may be overall inferior to other topical retinoids, given its potentially lower tolerability.
Topical Antibiotics—There are 4 commonly prescribed topical antibiotics that are approved by the FDA for the treatment of acne: clindamycin, erythromycin, dapsone, and minocycline. The American Academy of Dermatology guidelines for the treatment of acne recommend concomitant use of BPO to prevent antibiotic resistance.5 Clindamycin is favored over erythromycin because of increasing antibiotic resistance to erythromycin.21 Inexpensive generic options in multiple vehicles (eg, solution, foam, gel) make clindamycin a highly cost-effective option when antibiotic therapy is desired as part of a topical regimen (Table).
The cost-effectiveness of dapsone gel and minocycline foam relative to clindamycin are less certain. Rates of resistance to minocycline are lower than clindamycin, and minocycline foam may be a reasonable alternative in patients who have not had success with other topical antibiotics, such as clindamycin.22 However, given the absence of comparative effectiveness data to suggest minocycline is more effective than clindamycin, it is difficult to justify the substantially higher cost for the typical patient. Although dapsone gel has been suggested as an option for adult women with acne, there are no data to support that it is any more effective than other topical antibiotics in this patient population.23 As generic dapsone prices decrease, it may become a reasonable alternative to clindamycin. In addition, the antineutrophil properties of dapsone may be useful in other acneform and inflammatory eruptions, such as scalp folliculitis and folliculitis decalvans.24
Combination Topicals—Current combination topical products include antibiotic and BPO, antibiotic and retinoid, and retinoid and BPO. Use of combination agents is recommended to reduce the risk for resistance and to enhance effectiveness. Combination products offer improved convenience, which is associated with better adherence and outcomes.25 Generic fixed-dose adapalene-BPO can be a highly cost-effective option that can sometimes be less expensive than the individual component products (Table). Similarly, fixed-dose clindamycin-BPO also is likely to be highly cost-effective. A network meta-analysis found fixed-dose adapalene-BPO to be the most efficacious topical treatment, though it also was found to be the most irritating—more so than fixed-dose clindamycin-BPO, which may have similar efficacy.14,26,27 Generic fixed-dose tretinoin-clindamycin offers improved convenience and adherence compared to the individual components, but it is more expensive, and its cost-effectiveness may be influenced by the importance of convenience for the patient.25 An encapsulated, fixed-dose tretinoin 0.1%–BPO 3% cream is FDA approved for acne, but the cost is high and there is a lack of comparative effectiveness data demonstrating advantages over generic fixed-dose adapalene-BPO products.
Topical Antiandrogen—Clascoterone was introduced in 2020 as the first FDA-approved topical medication to target the hormonal pathogenesis of acne, inhibiting the androgen receptors in the sebaceous gland.28 Because it is rapidly metabolized to cortexolone and does not have systemic antiandrogen effects, clascoterone can be used in both men and women with acne. In clinical trials, it had minimal side effects, including no evidence of irritability, which is an advantage over topical retinoids and BPO.29 In addition, a phase 2 study found that clascoterone may have similar to superior efficacy to tretinoin cream 0.05%.30 Although clascoterone has several strengths, including its efficacy, tolerability, and unique mechanism of action, its cost-effectiveness is limited due to its high cost (Table) and the need for twice-daily application, which reduces convenience. Clascoterone likely is best reserved for patients with a strong hormonal pathogenesis of their acne or difficulty tolerating other topicals, or as an additional therapy to complement other topicals.
Oral Antibiotics—Oral antibiotics are the most commonly prescribed systemic treatments for acne, particularly tetracyclines such as doxycycline, minocycline, and sarecycline.31-34 Doxycycline and minocycline are considered first-line oral antibiotic therapy in the United States and are inexpensive and easily accessible.5 Doxycycline generally is recommended over minocycline given lack of evidence of superior efficacy of minocycline and concerns about severe adverse cutaneous reactions and drug-induced lupus with minocycline.35
In recent years, there has been growing concern of the development of antibiotic resistance.5 Sarecycline is a narrow-spectrum tetracycline that was FDA approved for acne in 2018. In vitro studies demonstrate sarecycline maintains high efficacy against C acnes with less activity against other bacteria, particularly gram-negative enterobes.36 The selectivity of sarecycline may lessen alterations of the gut microbiome seen with other oral antibiotics and reduce gastrointestinal tract side effects. Although comparative effectiveness studies are lacking, sarecycline was efficacious in phase 3 trials with few side effects compared with placebo.37 However, at this time, given the absence of comparative effectiveness data and its high cost (Table), sarecycline likely is best reserved for patients with comorbidities (eg, gastrointestinal disease), those requiring long-term antibiotic therapy, or those with acne that has failed to respond to other oral antibiotics.
Hormonal Treatments—Hormonal treatments such as combined oral contraceptives (COCs) and spironolactone often are considered second-line options, though they may represent cost-effective and safe alternatives to oral antibiotics for women with moderate to severe acne.38-41 There currently are 4 COCs approved by the FDA for the treatment of moderate acne in postmenarcheal females: drospirenone-ethinyl estradiol (Yaz [Bayer HealthCare Pharmaceuticals, Inc]), ethinyl estradiol-norgestimate (Ortho Tri-Cyclen [Ortho-McNeil Pharmaceuticals, Inc]), drospirenone-ethinyl estradiol-levomefolate (Beyaz [Bayer HealthCare Pharmaceuticals, Inc]), and ethinyl estradiol-norethindrone acetate-ferrous fumarate (Estrostep Fe [Allergan USA, Inc]).5 Treatment with COCs has been shown to cause substantial reductions in lesion counts across all lesion types compared to placebo, and a meta-analysis of 24 randomized trials conducted by Arowojolu et al42 demonstrated no consistent differences in acne reduction among different COCs.43,44 Although oral antibiotics are associated with faster improvement than COCs, there is some evidence that they have similar efficacy at 6 months of therapy.45 Combined oral contraceptives are inexpensive and likely reflect a highly cost-effective option (Table).
Spironolactone is an aldosterone inhibitor and androgen receptor blocker that is used off label to treat acne. It is one of the least expensive systemic medications for acne (Table). Although randomized controlled trials are lacking, several large case series support the effectiveness of spironolactone for women with acne.38,46 In addition, observational data suggest spironolactone may have similar effectiveness to oral antibiotics.41 Spironolactone generally is well tolerated, with the most common adverse effects being menstrual irregularities, breast tenderness, and diuresis.47,48 Many of these adverse effects are dose dependent and less likely with the dosing used in acne care. Additionally, menstrual irregularities can be reduced by concomitant use of a COC.48
Although frequent potassium monitoring remains common among patients being treated with spironolactone, there is growing evidence to suggest that potassium monitoring is of low value in young healthy women with acne.49-51 Reducing this laboratory monitoring likely represents an opportunity to provide higher-value care to patients being treated with spironolactone. However, laboratory monitoring should be considered if risk factors for hyperkalemia are present (eg, older age, comorbidities, medications).51
Isotretinoin—Isotretinoin is the most efficacious treatment available for acne and has the unique property of being able to induce a remission of acne activity for many patients.5 Although it remains modestly expensive (Table), it may be less costly overall relative to other treatments that may need continued use over many years because it can induce a remission of acne activity. As with spironolactone, frequent laboratory monitoring remains common among patients being treated with isotretinoin. There is no evidence to support checking complete blood cell counts.52 Several observational studies and a Delphi consensus support reduced monitoring, such as checking lipids and alanine aminotransferase at baseline and peak dose in otherwise young healthy patients.53,54 A recent critically appraised topic published in the British Journal of Dermatology has proposed eliminating laboratory monitoring entirely.55 Reducing laboratory monitoring for patients being treated with isotretinoin has been estimated to potentially save $100 million to $200 million per year in the United States.52-54
Other Strategies to Reduce Patient Costs
Although choosing a cost-effective treatment approach is critical to preventing financial toxicity given poor coverage for acne care and the growth of high-deductible insurance plans, some patients may still experience high treatment costs.56 Because pharmacy costs often are inflated, potentially related to practices of pharmacy benefit managers, it often is possible to find better prices than the presented list price, either by using platforms such as GoodRx or through direct-to-patient mail-order pharmacies such as Cost Plus Drug.57 For branded medications, some patients may be eligible for patient-assistance programs, though they typically are not available for those with public insurance such as Medicare or Medicaid. Compounding pharmacies offer another approach to reduce cost and improve convenience for patients, but because the vehicle can influence the efficacy and tolerability of some topical medications, it is possible that these compounded formulations may not perform similarly to the original FDA-approved products.
Conclusion
For mild to moderate acne, multimodal topical therapy often is required. Fixed-dose combination adapalene-BPO and clindamycin-BPO are highly cost-effective options for most patients. Lotion formulations of topical retinoids may be useful in patients with difficulty tolerating other formulations. Clascoterone is a novel topical antiandrogen that is more expensive than other topical therapies but can complement other topical therapies and is well tolerated.
For moderate to severe acne, doxycycline or hormonal therapy (ie, COCs, spironolactone) are highly cost-effective options. Isotretinoin is recommended for severe or scarring acne. Reduced laboratory monitoring for spironolactone and isotretinoin is an opportunity to provide higher-value care.
- Bhate K, Williams HC. Epidemiology of acne vulgaris. Br J Dermatol. 2013;168:474-485. doi:10.1111/bjd.12149
- Collier CN, Harper JC, Cafardi JA, et al. The prevalence of acne in adults 20 years and older. J Am Acad Dermatol. 2008;58:56-59. doi:10.1016/j.jaad.2007.06.045
- Webster GF. The pathophysiology of acne. Cutis. 2005;76(2 suppl):4-7.
- Degitz K, Placzek M, Borelli C, et al. Pathophysiology of acne. J Dtsch Dermatol Ges. 2007;5:316-323. doi:10.1111/j.1610-0387.2007.06274.x
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.e33. doi:10.1016/j.jaad.2015.12.037
- Felmingham C, Kerr A, Veysey E. Costs incurred by patients with acne prior to dermatological consultation and their relation to patient income. Australas J Dermatol. 2020;61:384-386. doi:10.1111/ajd.13324
- Perche P, Singh R, Feldman S. Patient preferences for acne vulgaris treatment and barriers to care: a survey study. J Drugs Dermatol. 2022;21:1191-1195. doi:10.36849/JDD.6940
- KFF Health Tracking Poll—February 2019. Accessed August 9, 2023. https://files.kff.org/attachment/Topline-KFF-Health-Tracking-Poll-February-2019
- Ryskina KL, Goldberg E, Lott B, et al. The role of the physician in patient perceptions of barriers to primary adherence with acne medications. JAMA Dermatol. 2018;154:456-459. doi:10.1001/jamadermatol.2017.6144
- Porter ME. What is value in health care? N Engl J Med. 2010;363:2477-2481. doi:10.1056/NEJMp1011024
- Barbieri JS, Tan JKL, Adamson AS. Active comparator trial designs used to promote development of innovative new medications. Cutis. 2020;106:E4-E6. doi:10.12788/cutis.0067
- Miller J, Ly S, Mostaghimi A, et al. Use of active comparator trials for topical medications in dermatology. JAMA Dermatol. 2021;157:597-599. doi:10.1001/jamadermatol.2021.0356
- GoodRx. Accessed August 11, 2023. https://www.goodrx.com
- Stuart B, Maund E, Wilcox C, et al. Topical preparations for the treatment of mild‐to‐moderate acne vulgaris: systematic review and network meta‐analysis. Br J Dermatol. 2021;185:512-525. doi:10.1111/bjd.20080
- Mavranezouli I, Welton NJ, Daly CH, et al. Cost-effectiveness of topical pharmacological, oral pharmacological, physical and combined treatments for acne vulgaris. Clin Exp Dermatol. 2022;47:2176-2187. doi:10.1111/ced.15356
- Tanghetti E, Werschler W, Lain T, et al. Tazarotene 0.045% lotion for once-daily treatment of moderate-to-severe acne vulgaris: results from two phase 3 trials. J Drugs Dermatol. 2020;19:70-77. doi:10.36849/JDD.2020.3977
- Tyring SK, Kircik LH, Pariser DM, et al. Novel tretinoin 0.05% lotion for the once-daily treatment of moderate-to-severe acne vulgaris: assessment of efficacy and safety in patients aged 9 years and older. J Drugs Dermatol. 2018;17:1084-1091.
- Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 μg/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80:1691-1699. doi:10.1016/j.jaad.2019.02.044
- Huang CY, Chang IJ, Bolick N, et al. Comparative efficacy of pharmacological treatments for acne vulgaris: a network meta-analysis of 221 randomized controlled trials. Ann Fam Med. 2023;21:358-369. doi:10.1370/afm.2995
- Draelos ZD. Low irritation potential of tazarotene 0.045% lotion: head-to-head comparison to adapalene 0.3% gel and trifarotene 0.005% cream in two studies. J Dermatolog Treat. 2023;34:2166346. doi:10.1080/09546634.2023.2166346
- Dessinioti C, Katsambas A. Antibiotics and antimicrobial resistance in acne: epidemiological trends and clinical practice considerations. Yale J Biol Med. 2022;95:429-443.
- Gold LS, Dhawan S, Weiss J, et al. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80:168-177. doi:10.1016/j.jaad.2018.08.020
- Wang X, Wang Z, Sun L, et al. Efficacy and safety of dapsone gel for acne: a systematic review and meta-analysis. Ann Palliat Med. 2022;11:611-620. doi:10.21037/apm-21-3935
- Melián-Olivera A, Burgos-Blasco P, Selda-Enríquez G, et al. Topical dapsone for folliculitis decalvans: a retrospective cohort study. J Am Acad Dermatol. 2022;87:150-151. doi:10.1016/j.jaad.2021.07.004
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86:103-108.
- Ting W. Randomized, observer-blind, split-face study to compare the irritation potential of 2 topical acne formulations over a 14-day treatment period. Cutis. 2012;90:91-96.
- Aschoff R, Möller S, Haase R, et al. Tolerability and efficacy ofclindamycin/tretinoin versus adapalene/benzoyl peroxide in the treatment of acne vulgaris. J Drugs Dermatol. 2021;20:295-301. doi:10.36849/JDD.2021.5641
- Rosette C, Agan FJ, Mazzetti A, et al. Cortexolone 17α-propionate (clascoterone) is a novel androgen receptor antagonist that inhibits production of lipids and inflammatory cytokines from sebocytes in vitro. J Drugs Dermatol. 2019;18:412-418.
- Hebert A, Thiboutot D, Stein Gold L, et al. Efficacy and safety of topical clascoterone cream, 1%, for treatment in patients with facial acne: two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156:621-630. doi:10.1001/jamadermatol.2020.0465
- Trifu V, Tiplica GS, Naumescu E, et al. Cortexolone 17α-propionate 1% cream, a new potent antiandrogen for topical treatment of acne vulgaris. a pilot randomized, double-blind comparative study vs. placebo and tretinoin 0·05% cream. Br J Dermatol. 2011;165:177-183. doi:10.1111/j.1365-2133.2011.10332.x
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Guzman AK, Barbieri JS. Comparative analysis of prescribing patterns of tetracycline class antibiotics and spironolactone between advanced practice providers and physicians in the treatment of acne vulgaris. J Am Acad Dermatol. 2021;84:1119-1121. doi:10.1016/j.jaad.2020.06.044
- Barbieri JS, James WD, Margolis DJ. Trends in prescribing behavior of systemic agents used in the treatment of acne among dermatologists and nondermatologists: a retrospective analysis, 2004-2013. J Am Acad Dermatol. 2017;77:456-463.e4. doi:10.1016/j.jaad.2017.04.016
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016. JAMA Dermatol. 2019;155:290-297. doi:10.1001/jamadermatol.2018.4944
- Garner SE, Eady A, Bennett C, et al. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012;2012:CD002086. doi:10.1002/14651858.CD002086.pub2
- Zhanel G, Critchley I, Lin LY, et al. Microbiological profile of sarecycline, a novel targeted spectrum tetracycline for the treatment of acne vulgaris. Antimicrob Agents Chemother. 2018;63:e01297-18. doi:10.1128/AAC.01297-18
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987-996.
- Garg V, Choi JK, James WD, et al. Long-term use of spironolactone for acne in women: a case series of 403 patients. J Am Acad Dermatol. 2021;84:1348-1355. doi:10.1016/j.jaad.2020.12.071
- Barbieri JS, Choi JK, James WD, et al. Real-world drug usage survival of spironolactone versus oral antibiotics for the management of female patients with acne. J Am Acad Dermatol. 2019;81:848-851. doi:10.1016/j.jaad.2019.03.036
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. 2019;80:538-549. doi:10.1016/j.jaad.2018.09.055
- Barbieri JS, Choi JK, Mitra N, et al. Frequency of treatment switching for spironolactone compared to oral tetracycline-class antibiotics for women with acne: a retrospective cohort study 2010-2016. J Drugs Dermatol. 2018;17:632-638.
- Arowojolu AO, Gallo MF, Lopez LM, et al. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;7:CD004425. doi:10.1002/14651858.CD004425.pub6
- Maloney JM, Dietze P, Watson D, et al. Treatment of acne using a 3-milligram drospirenone/20-microgram ethinyl estradiol oral contraceptive administered in a 24/4 regimen. Obstet Gynecol. 2008;112:773-781. doi:10.1097/AOG.0b013e318187e1c5
- Lucky AW, Koltun W, Thiboutot D, et al. A combined oral contraceptive containing 3-mg drospirenone/20-microg ethinyl estradiol in the treatment of acne vulgaris: a randomized, double-blind, placebo-controlled study evaluating lesion counts and participant self-assessment. Cutis. 2008;82:143-150.
- Koo EB, Petersen TD, Kimball AB. Meta-analysis comparing efficacy of antibiotics versus oral contraceptives in acne vulgaris. J Am Acad Dermatol. 2014;71:450-459. doi:10.1016/j.jaad.2014.03.051
- Roberts EE, Nowsheen S, Davis DMR, et al. Use of spironolactone to treat acne in adolescent females. Pediatr Dermatol. 2021;38:72-76. doi:10.1111/pde.14391
- Shaw JC. Low-dose adjunctive spironolactone in the treatment of acne in women: a retrospective analysis of 85 consecutively treated patients. J Am Acad Dermatol. 2000;43:498-502. doi:10.1067/mjd.2000.105557
- Layton AM, Eady EA, Whitehouse H, et al. Oral spironolactone for acne vulgaris in adult females: a hybrid systematic review. Am J Clin Dermatol. 2017;18:169-191. doi:10.1007/s40257-016-0245-x
- Barbieri JS, Margolis DJ, Mostaghimi A. Temporal trends and clinician variability in potassium monitoring of healthy young women treated for acne with spironolactone. JAMA Dermatol. 2021;157:296-300. doi:10.1001/jamadermatol.2020.5468
- Plovanich M, Weng QY, Mostaghimi A. Low usefulness of potassium monitoring among healthy young women taking spironolactone for acne. JAMA Dermatol. 2015;151:941-944. doi:10.1001/jamadermatol.2015.34
- Thiede RM, Rastogi S, Nardone B, et al. Hyperkalemia in women with acne exposed to oral spironolactone: a retrospective study from the RADAR (Research on Adverse Drug Events and Reports) program. Int J Womens Dermatol. 2019;5:155-157. doi:10.1016/j.ijwd.2019.04.024
- Barbieri JS, Shin DB, Wang S, et al. The clinical utility of laboratory monitoring during isotretinoin therapy for acne and changes to monitoring practices over time. J Am Acad Dermatol. 2020;82:72-79. doi:10.1016/j.jaad.2019.06.025
- Lee YH, Scharnitz TP, Muscat J, et al. Laboratory monitoring during isotretinoin therapy for acne: a systematic review and meta-analysis. JAMA Dermatol. 2016;152:35-44. doi:10.1001/jamadermatol.2015.3091
- Xia E, Han J, Faletsky A, et al. Isotretinoin laboratory monitoring in acne treatment: a Delphi consensus study. JAMA Dermatol. 2022;158:942-948. doi:10.1001/jamadermatol.2022.2044
- Affleck A, Jackson D, Williams HC, et al. Is routine laboratory testing in healthy young patients taking isotretinoin necessary: a critically appraised topic. Br J Dermatol. 2022;187:857-865. doi:10.1111/bjd.21840
- Barbieri JS, LaChance A, Albrecht J. Double standards and inconsistencies in access to care-what constitutes a cosmetic treatment? JAMA Dermatol. 2023;159:245-246. doi:10.1001/jamadermatol.2022.6322
- Trish E, Van Nuys K, Popovian R. US consumers overpay for generic drugs. Schaeffer Center White Paper Series. May 31, 2022. doi:10.25549/m589-2268
- Bhate K, Williams HC. Epidemiology of acne vulgaris. Br J Dermatol. 2013;168:474-485. doi:10.1111/bjd.12149
- Collier CN, Harper JC, Cafardi JA, et al. The prevalence of acne in adults 20 years and older. J Am Acad Dermatol. 2008;58:56-59. doi:10.1016/j.jaad.2007.06.045
- Webster GF. The pathophysiology of acne. Cutis. 2005;76(2 suppl):4-7.
- Degitz K, Placzek M, Borelli C, et al. Pathophysiology of acne. J Dtsch Dermatol Ges. 2007;5:316-323. doi:10.1111/j.1610-0387.2007.06274.x
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-973.e33. doi:10.1016/j.jaad.2015.12.037
- Felmingham C, Kerr A, Veysey E. Costs incurred by patients with acne prior to dermatological consultation and their relation to patient income. Australas J Dermatol. 2020;61:384-386. doi:10.1111/ajd.13324
- Perche P, Singh R, Feldman S. Patient preferences for acne vulgaris treatment and barriers to care: a survey study. J Drugs Dermatol. 2022;21:1191-1195. doi:10.36849/JDD.6940
- KFF Health Tracking Poll—February 2019. Accessed August 9, 2023. https://files.kff.org/attachment/Topline-KFF-Health-Tracking-Poll-February-2019
- Ryskina KL, Goldberg E, Lott B, et al. The role of the physician in patient perceptions of barriers to primary adherence with acne medications. JAMA Dermatol. 2018;154:456-459. doi:10.1001/jamadermatol.2017.6144
- Porter ME. What is value in health care? N Engl J Med. 2010;363:2477-2481. doi:10.1056/NEJMp1011024
- Barbieri JS, Tan JKL, Adamson AS. Active comparator trial designs used to promote development of innovative new medications. Cutis. 2020;106:E4-E6. doi:10.12788/cutis.0067
- Miller J, Ly S, Mostaghimi A, et al. Use of active comparator trials for topical medications in dermatology. JAMA Dermatol. 2021;157:597-599. doi:10.1001/jamadermatol.2021.0356
- GoodRx. Accessed August 11, 2023. https://www.goodrx.com
- Stuart B, Maund E, Wilcox C, et al. Topical preparations for the treatment of mild‐to‐moderate acne vulgaris: systematic review and network meta‐analysis. Br J Dermatol. 2021;185:512-525. doi:10.1111/bjd.20080
- Mavranezouli I, Welton NJ, Daly CH, et al. Cost-effectiveness of topical pharmacological, oral pharmacological, physical and combined treatments for acne vulgaris. Clin Exp Dermatol. 2022;47:2176-2187. doi:10.1111/ced.15356
- Tanghetti E, Werschler W, Lain T, et al. Tazarotene 0.045% lotion for once-daily treatment of moderate-to-severe acne vulgaris: results from two phase 3 trials. J Drugs Dermatol. 2020;19:70-77. doi:10.36849/JDD.2020.3977
- Tyring SK, Kircik LH, Pariser DM, et al. Novel tretinoin 0.05% lotion for the once-daily treatment of moderate-to-severe acne vulgaris: assessment of efficacy and safety in patients aged 9 years and older. J Drugs Dermatol. 2018;17:1084-1091.
- Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 μg/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80:1691-1699. doi:10.1016/j.jaad.2019.02.044
- Huang CY, Chang IJ, Bolick N, et al. Comparative efficacy of pharmacological treatments for acne vulgaris: a network meta-analysis of 221 randomized controlled trials. Ann Fam Med. 2023;21:358-369. doi:10.1370/afm.2995
- Draelos ZD. Low irritation potential of tazarotene 0.045% lotion: head-to-head comparison to adapalene 0.3% gel and trifarotene 0.005% cream in two studies. J Dermatolog Treat. 2023;34:2166346. doi:10.1080/09546634.2023.2166346
- Dessinioti C, Katsambas A. Antibiotics and antimicrobial resistance in acne: epidemiological trends and clinical practice considerations. Yale J Biol Med. 2022;95:429-443.
- Gold LS, Dhawan S, Weiss J, et al. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80:168-177. doi:10.1016/j.jaad.2018.08.020
- Wang X, Wang Z, Sun L, et al. Efficacy and safety of dapsone gel for acne: a systematic review and meta-analysis. Ann Palliat Med. 2022;11:611-620. doi:10.21037/apm-21-3935
- Melián-Olivera A, Burgos-Blasco P, Selda-Enríquez G, et al. Topical dapsone for folliculitis decalvans: a retrospective cohort study. J Am Acad Dermatol. 2022;87:150-151. doi:10.1016/j.jaad.2021.07.004
- Yentzer BA, Ade RA, Fountain JM, et al. Simplifying regimens promotes greater adherence and outcomes with topical acne medications: a randomized controlled trial. Cutis. 2010;86:103-108.
- Ting W. Randomized, observer-blind, split-face study to compare the irritation potential of 2 topical acne formulations over a 14-day treatment period. Cutis. 2012;90:91-96.
- Aschoff R, Möller S, Haase R, et al. Tolerability and efficacy ofclindamycin/tretinoin versus adapalene/benzoyl peroxide in the treatment of acne vulgaris. J Drugs Dermatol. 2021;20:295-301. doi:10.36849/JDD.2021.5641
- Rosette C, Agan FJ, Mazzetti A, et al. Cortexolone 17α-propionate (clascoterone) is a novel androgen receptor antagonist that inhibits production of lipids and inflammatory cytokines from sebocytes in vitro. J Drugs Dermatol. 2019;18:412-418.
- Hebert A, Thiboutot D, Stein Gold L, et al. Efficacy and safety of topical clascoterone cream, 1%, for treatment in patients with facial acne: two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156:621-630. doi:10.1001/jamadermatol.2020.0465
- Trifu V, Tiplica GS, Naumescu E, et al. Cortexolone 17α-propionate 1% cream, a new potent antiandrogen for topical treatment of acne vulgaris. a pilot randomized, double-blind comparative study vs. placebo and tretinoin 0·05% cream. Br J Dermatol. 2011;165:177-183. doi:10.1111/j.1365-2133.2011.10332.x
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Guzman AK, Barbieri JS. Comparative analysis of prescribing patterns of tetracycline class antibiotics and spironolactone between advanced practice providers and physicians in the treatment of acne vulgaris. J Am Acad Dermatol. 2021;84:1119-1121. doi:10.1016/j.jaad.2020.06.044
- Barbieri JS, James WD, Margolis DJ. Trends in prescribing behavior of systemic agents used in the treatment of acne among dermatologists and nondermatologists: a retrospective analysis, 2004-2013. J Am Acad Dermatol. 2017;77:456-463.e4. doi:10.1016/j.jaad.2017.04.016
- Barbieri JS, Bhate K, Hartnett KP, et al. Trends in oral antibiotic prescription in dermatology, 2008 to 2016. JAMA Dermatol. 2019;155:290-297. doi:10.1001/jamadermatol.2018.4944
- Garner SE, Eady A, Bennett C, et al. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012;2012:CD002086. doi:10.1002/14651858.CD002086.pub2
- Zhanel G, Critchley I, Lin LY, et al. Microbiological profile of sarecycline, a novel targeted spectrum tetracycline for the treatment of acne vulgaris. Antimicrob Agents Chemother. 2018;63:e01297-18. doi:10.1128/AAC.01297-18
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17:987-996.
- Garg V, Choi JK, James WD, et al. Long-term use of spironolactone for acne in women: a case series of 403 patients. J Am Acad Dermatol. 2021;84:1348-1355. doi:10.1016/j.jaad.2020.12.071
- Barbieri JS, Choi JK, James WD, et al. Real-world drug usage survival of spironolactone versus oral antibiotics for the management of female patients with acne. J Am Acad Dermatol. 2019;81:848-851. doi:10.1016/j.jaad.2019.03.036
- Barbieri JS, Spaccarelli N, Margolis DJ, et al. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. 2019;80:538-549. doi:10.1016/j.jaad.2018.09.055
- Barbieri JS, Choi JK, Mitra N, et al. Frequency of treatment switching for spironolactone compared to oral tetracycline-class antibiotics for women with acne: a retrospective cohort study 2010-2016. J Drugs Dermatol. 2018;17:632-638.
- Arowojolu AO, Gallo MF, Lopez LM, et al. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;7:CD004425. doi:10.1002/14651858.CD004425.pub6
- Maloney JM, Dietze P, Watson D, et al. Treatment of acne using a 3-milligram drospirenone/20-microgram ethinyl estradiol oral contraceptive administered in a 24/4 regimen. Obstet Gynecol. 2008;112:773-781. doi:10.1097/AOG.0b013e318187e1c5
- Lucky AW, Koltun W, Thiboutot D, et al. A combined oral contraceptive containing 3-mg drospirenone/20-microg ethinyl estradiol in the treatment of acne vulgaris: a randomized, double-blind, placebo-controlled study evaluating lesion counts and participant self-assessment. Cutis. 2008;82:143-150.
- Koo EB, Petersen TD, Kimball AB. Meta-analysis comparing efficacy of antibiotics versus oral contraceptives in acne vulgaris. J Am Acad Dermatol. 2014;71:450-459. doi:10.1016/j.jaad.2014.03.051
- Roberts EE, Nowsheen S, Davis DMR, et al. Use of spironolactone to treat acne in adolescent females. Pediatr Dermatol. 2021;38:72-76. doi:10.1111/pde.14391
- Shaw JC. Low-dose adjunctive spironolactone in the treatment of acne in women: a retrospective analysis of 85 consecutively treated patients. J Am Acad Dermatol. 2000;43:498-502. doi:10.1067/mjd.2000.105557
- Layton AM, Eady EA, Whitehouse H, et al. Oral spironolactone for acne vulgaris in adult females: a hybrid systematic review. Am J Clin Dermatol. 2017;18:169-191. doi:10.1007/s40257-016-0245-x
- Barbieri JS, Margolis DJ, Mostaghimi A. Temporal trends and clinician variability in potassium monitoring of healthy young women treated for acne with spironolactone. JAMA Dermatol. 2021;157:296-300. doi:10.1001/jamadermatol.2020.5468
- Plovanich M, Weng QY, Mostaghimi A. Low usefulness of potassium monitoring among healthy young women taking spironolactone for acne. JAMA Dermatol. 2015;151:941-944. doi:10.1001/jamadermatol.2015.34
- Thiede RM, Rastogi S, Nardone B, et al. Hyperkalemia in women with acne exposed to oral spironolactone: a retrospective study from the RADAR (Research on Adverse Drug Events and Reports) program. Int J Womens Dermatol. 2019;5:155-157. doi:10.1016/j.ijwd.2019.04.024
- Barbieri JS, Shin DB, Wang S, et al. The clinical utility of laboratory monitoring during isotretinoin therapy for acne and changes to monitoring practices over time. J Am Acad Dermatol. 2020;82:72-79. doi:10.1016/j.jaad.2019.06.025
- Lee YH, Scharnitz TP, Muscat J, et al. Laboratory monitoring during isotretinoin therapy for acne: a systematic review and meta-analysis. JAMA Dermatol. 2016;152:35-44. doi:10.1001/jamadermatol.2015.3091
- Xia E, Han J, Faletsky A, et al. Isotretinoin laboratory monitoring in acne treatment: a Delphi consensus study. JAMA Dermatol. 2022;158:942-948. doi:10.1001/jamadermatol.2022.2044
- Affleck A, Jackson D, Williams HC, et al. Is routine laboratory testing in healthy young patients taking isotretinoin necessary: a critically appraised topic. Br J Dermatol. 2022;187:857-865. doi:10.1111/bjd.21840
- Barbieri JS, LaChance A, Albrecht J. Double standards and inconsistencies in access to care-what constitutes a cosmetic treatment? JAMA Dermatol. 2023;159:245-246. doi:10.1001/jamadermatol.2022.6322
- Trish E, Van Nuys K, Popovian R. US consumers overpay for generic drugs. Schaeffer Center White Paper Series. May 31, 2022. doi:10.25549/m589-2268
Practice Points
- For mild to moderate acne, fixed-dose combination adapalene–benzoyl peroxide and clindamycin–benzoyl peroxide are highly cost-effective options for most patients.
- For moderate to severe acne, doxycycline or hormonal therapy (ie, combined oral contraceptives, spironolactone) are highly cost-effective options.
- Reduction of laboratory monitoring for spironolactone and isotretinoin is an opportunity to provide higher-value care.
Complications of Body Piercings: A Systematic Review
The practice of body piercing has been present in cultures around the world for centuries. Piercings may be performed for religious or spiritual reasons or as a form of self-expression. In recent years, body piercings have become increasingly popular in all genders, with the most common sites being the ears, mouth, nose, eyebrows, nipples, navel, and genitals.1 The prevalence of body piercing in the general population is estimated to be as high as 50%.2 With the rising popularity of piercings, there also has been an increase in their associated complications, with one study noting that up to 35% of individuals with pierced ears and 30% of all pierced sites developed a complication.3 Common problems following piercing include infections, keloid formation, allergic contact dermatitis, site deformation, and tooth fractures.4 It is of utmost importance that health care professionals are aware of the potential complications associated with such a common practice. A comprehensive review of complications associated with cutaneous and mucosal piercings is lacking. We conducted a systematic review to summarize the clinical characteristics, complication types and frequency, and treatments reported for cutaneous and mucosal piercings.
METHODS
We conducted a systematic review of the literature adhering to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) reporting guidelines.5
Search Strategy, Study Eligibility Criteria, and Study Selection
A literature search of the Embase, MEDLINE, and PubMed databases was performed on June 20, 2022, using search terms related to body piercing and possible piercing-induced complications (Supplemental Information online). All studies reporting complications following body piercing were included. In vitro and animal studies were excluded. Title and abstract screening were completed by 6 independent researchers (S.C., K.K., M.M-B., K.A., T.S., I.M.M.) using Covidence online systematic review software (www.covidence.org). Six reviewers (S.C., K.K., M.M-B., K.A., T.S., I.M.M.) independently evaluated titles, abstracts, and full texts to identify relevant studies. Conflicts were resolved by the senior reviewer (I.M.M.).
Data Extraction and Synthesis
Five reviewers (S.C., K.K., M.M-B., K.A., T.S.) independently extracted data from eligible studies using a standardized extraction form that included title; authors; year of publication; sample size; and key findings, including mean age, sex, piercing location, complication type, and treatment received.
Treatment type was placed into the following categories: surgical treatments, antimicrobials, medical treatments, direct-target therapy, oral procedures, avoidance, miscellaneous therapies, and no treatment. (Data regarding treatments can be found in the Supplemental Information online.)
RESULTS
The combined search yielded 2679 studies, 617 of which underwent full-text review; 319 studies were included (Figure). Studies were published from 1950 to June 2022 and included both adult and pediatric populations.
Patient Characteristics
In total, our pooled analysis included data on 30,090 complications across 36,803 pierced sites in 30,231 patients (Table 1). Demographic data are available for 55% (n=30,231) of patients. Overall, 74% (22,247/30,231) of the individuals included in our analysis were female. The mean age was 27.8 years (range, 0–76 years).
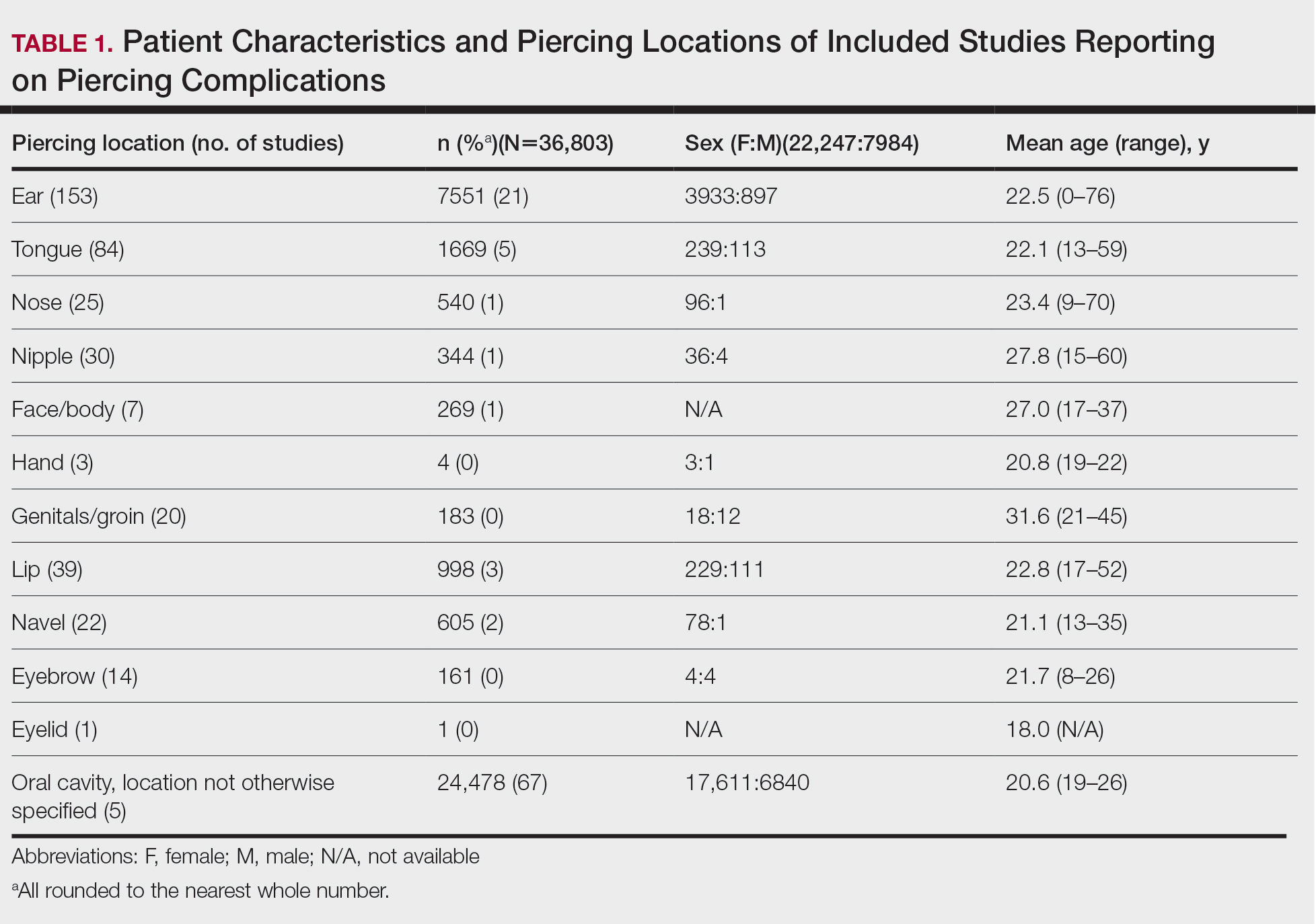
Piercing Location
Overall, 36,803 pierced sites had a reported complication. The oral cavity, location not otherwise specified, was the most common site associated with a complication, accounting for 67% (n=24,478) of complications (Table 1). Other reported sites included (in decreasing frequency) the ears (21%, n=7551), tongue (5%, n=1669), lip (3%, n=998), navel (2%, n=605), nose (1%, n=540), nipple (1%, n=344), face/body (1%, n=269), genitals/groin (0%, n=183), eyebrow (0%, n=161), hand (0%, n=4), and eyelid (0%, n=1). Piercing complications were more commonly reported among females across all piercing locations except for the eyebrow, which was equal in both sexes.
Complications
Local Infections—Local infections accounted for 36% of reported complication types (n=10,872/30,090): perichondritis (1%, n=85); abscesses (0%, n=25); bacterial colonization (1%, n=106); and local infections, not otherwise specified (98%, n=10,648)(Table 2). The majority of local infections were found to be secondary to piercings of the ear and oral cavity. The nipple was found to be a common site for abscesses (40%, n=10), whereas the tongue was found to be the most common site for bacterial colonization (69%, n=73).
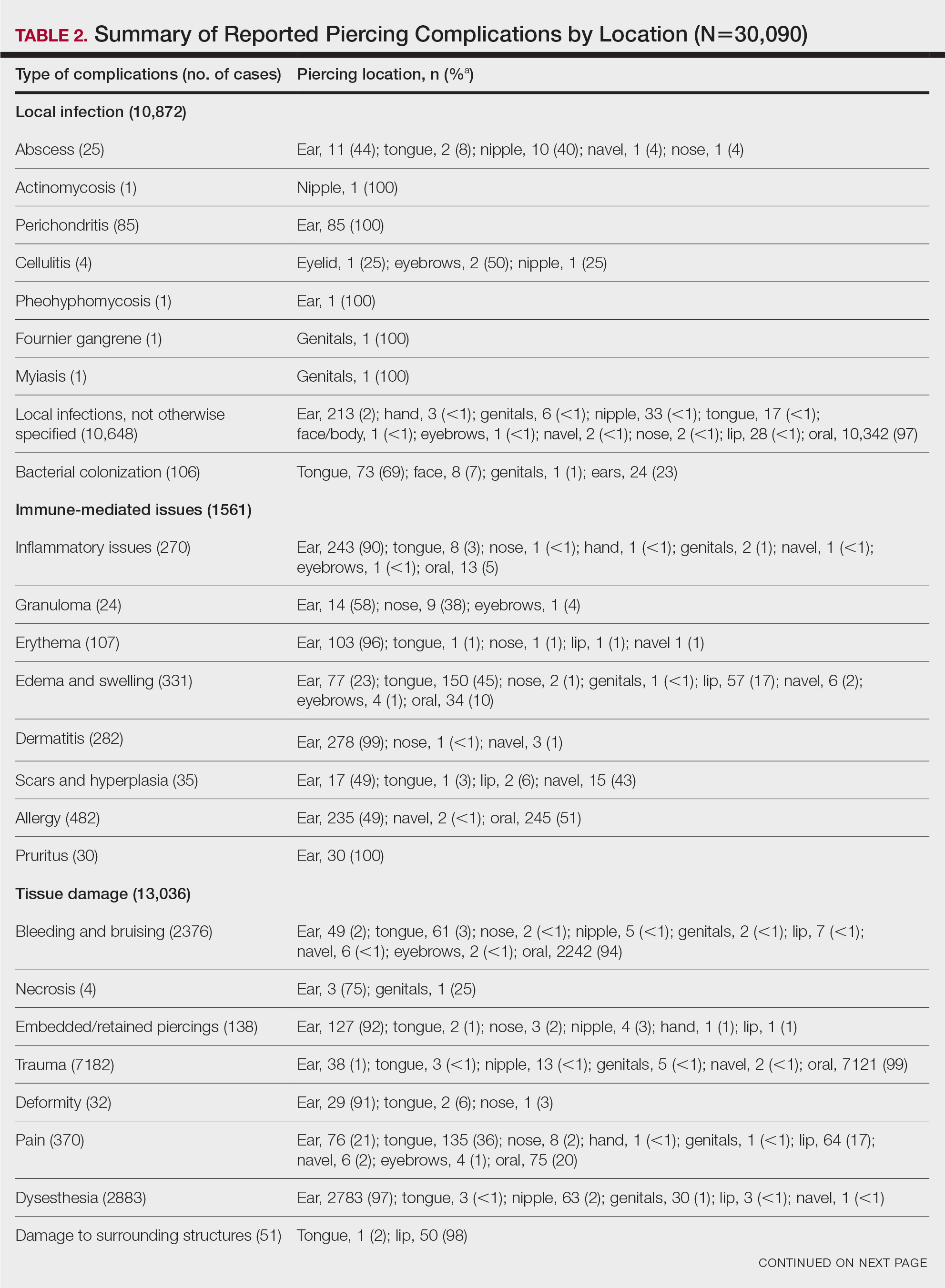
Immune-Mediated Issues—Immune-mediated issues encompassed 5% of the total reported complications (n=1561/30,090). The most commonly reported immune-mediated complications included allergies (31%, n=482), edema and swelling (21%, n=331), dermatitis (18%, n=282), and inflammatory lesions (17%, n=270). The majority were found to occur secondary to ear piercings, with the exception of edema, which mainly occurred secondary to tongue piercings (45%, n=150), and allergy, which primarily was associated with oral piercings (51%, n=245)(Table 2).
Tissue Damage—Tissue damage accounted for 43% of all complications (n=13,036/30,090). The most common forms of tissue damage were trauma (55%, n=7182), dysesthesia (22%, n=2883), bleeding and bruising (18%, n=2376), and pain (3%, n=370)(Table 2). Trauma was mainly found to be a complication in the context of oral piercings (99%, n=7121). Similarly, 94% (n=2242) of bleeding and bruising occurred secondary to oral piercings. Embedded piercings (92%, n=127), deformity (91%, n=29), and necrosis (75%, n=3) mostly occurred following ear piercings. Lip piercings were found to be the most common cause of damage to surrounding structures (98%, n=50).
Oral—Overall, 3193 intraoral complications were reported, constituting 11% of the total complications (Table 2). Oral complications included dental damage (86%, n=2732), gum recession (14%, n=459), and gingivitis (0%, n=2). Dental damage was mostly reported following oral piercings (90%, n=2453), whereas gum recession was mostly reported following lip piercings (59%, n=272).
Proliferations—Proliferations accounted for 795 (3%) of reported piercing complications. The majority (97%, n=772) were keloids, 2% (n=16) were other benign growths, and 1% (n=7) were malignancies. These complications mostly occurred secondary to ear piercings, which resulted in 741 (96%) keloids, 6 (38%) benign growths, and 4 (57%) malignancies.
Systemic—Overall, 2% (n=633) of the total complications were classified as systemic issues, including functional impairment (45%, n=282), secondary organ involvement (24%, n=150), cardiac issues (3%, n=21), and aspiration/inhalation (1%, n=8). Nonlocalized infections such as hepatitis or an increased risk thereof (17%, n=107), tetanus (8%, n=52), chlamydia (1%, n=9), HIV (0%, n=1), herpes simplex virus (0%, n=1), gonorrhea (0%, n=1), and bacterial vaginosis (0%, n=1) also were included in this category. The tongue, ear, and genitals were the locations most involved in these complications (Table 2). Secondary organ involvement mostly occurred after ear (36%, n=54) and genital piercings (27%, n=41). A total of 8 cases of piercing aspiration and/or inhalation were reported in association with piercings of the head and neck (Table 2).
COMMENT
Piercing Complications
Overall, the ear, tongue, and oral cavity were found to be the sites with the most associated complications recorded in the literature, and local infection and tissue damage were found to be the most prevalent types of complications. A plethora of treatments were used to manage piercing-induced complications, including surgical or medical treatments and avoidance (Supplemental Information). Reports by Metts6 and Escudero-Castaño et al7 provide detailed protocols and photographs of piercings.
Infections
Our review found that local infections were commonly reported complications associated with body piercings, which is consistent with other studies.1 The initial trauma inherent in the piercing process followed by the presence of an ongoing foreign body lends itself to an increased risk for developing these complications. Wound healing after piercing also varies based on the piercing location.
The rate and severity of the infection are influenced by the anatomic location of the piercing, hygiene, method of piercing, types of materials used, and aftercare.8 Piercing cartilage sites, such as the helix, concha, or nose, increases susceptibility to infections and permanent deformities. Cartilage is particularly at risk because of its avascular nature.9 Other studies have reported similar incidences of superficial localized infections; infectious complications were seen in 10% to 30% of body piercings in one study,3 while 45% of American and Australian college students reported infection at a piercing site in a second study.10
Systemic Issues
Systemic issues are potentially the most dangerous piercing-induced complications, though they were rarer in our analysis. Some serious complications included septic emboli, fatal staphylococcal toxic shock syndrome, and death. Although some systemic issues, such as staphylococcal toxic shock syndrome and septic sacroiliitis, required extensive hospital stays and complex treatment, others had lifelong repercussions, such as hepatitis and HIV. One report showed an increased incidence of endocarditis associated with body piercing, including staphylococcal endocarditis following nasal piercings, Neisseria endocarditis following tongue piercings, and Staphylococcus epidermidis endocarditis following nipple piercings.11 Moreover, Mariano et al12—who noted a case of endocarditis and meningitis associated with a nape piercing in a young female in 2015—reinforced the notion that information pertaining to the risks associated with body piercing must be better disseminated, given the potential for morbid or fatal outcomes. Finally, nonsterile piercing techniques and poor hygiene were found to contribute substantially to the increased risk for infection, so it is of utmost importance to reinforce proper practices in piercing salons.4
Immune-Mediated Issues
Because piercings are foreign bodies, they are susceptible to both acute and chronic immune responses. Our study found that allergies and dermatitis made up almost half of the immune-mediated piercing complications. It is especially important to emphasize that costume jewelry exposes our skin to a variety of contact allergens, most prominently nickel, heightening the risk for developing allergic contact dermatitis.13 Moreover, a study conducted by Brandão et al14 found that patients with pierced ears were significantly more likely to react to nickel than those without pierced ears (P=.031). Although other studies have found that allergy to metals ranges from 8.3% to 20% in the general population,15 we were not able to quantify the incidence in our study due to a lack of reporting of common benign complications, such as contact dermatitis.
Tissue Damage and Local Problems
Our review found that tissue and oral damage also were commonly reported piercing complications, with the most common pathologies being trauma, dysesthesia, bleeding/bruising, and dental damage. Laumann and Derick16 reported that bleeding, tissue trauma, and local problems were common physical health problems associated with body piercing. Severe complications, such as abscesses, toxic shock syndrome, and endocarditis, also have been reported in association with intraoral piercings.17 Moreover, other studies have shown that oral piercings are associated with several adverse oral and systemic conditions. A meta-analysis of individuals with oral piercings found a similar prevalence of dental fracture, gingival recession, and tooth wear (34%), as well as unspecified dental damage (27%) and tooth chipping (22%). Additionally, this meta-analysis reported a 3-fold increased risk for dental fracture and 7-fold increased risk for gingival recession with oral piercings.18 Another meta-analysis of oral piercing complications found a similar prevalence of dental fracture (34%), tooth wear (34%), gingival recession (33%), unspecified dental damage (27%), and tooth chipping (22%).19 Considering the extensive amount of cumulative damage, wearers of oral jewelry require periodic periodontal evaluations to monitor for dental damage and gingival recession.20 There are limited data on treatments for complications of oral piercings, and further research in this area is warranted.
Proliferations and Scars
Although proliferations and scarring were among the least common complications reported in the literature, they are some of the most cosmetically disfiguring for patients. Keloids, the most common type of growth associated with piercings, do not naturally regress and thus require some form of intervention. Given the multimodal approach used to treat keloids, as described by the evidence-based algorithm by Ogawa,21 it is not surprising that keloids also represented the complication most treated with medical therapies, such as steroids, and also with direct-target therapy, such as liquid nitrogen therapy (Supplemental Information).
Other proliferations reported in the literature include benign pyogenic granulomas22 and much less commonly malignant neoplasms such as basal cell carcinoma23 and squamous cell carcinoma.24 Although rare, treatment of piercing-associated malignancies include surgical removal, chemotherapy, and radiation therapy (Supplemental Information).
Limitations
There are several limitations to our systematic review. First, heterogeneity in study designs, patient populations, treatment interventions, and outcome measures of included studies may have affected the quality and generalizability of our results. Moreover, because the studies included in this systematic review focused on specific complications, we could not compare our results to the literature that analyzes incidence rates of piercing complications. Furthermore, not all studies included the data that we hoped to extract, and thus only available data were reported in these instances. Finally, the articles we reviewed may have included publication bias, with positive findings being more frequently published, potentially inflating certain types and sites of complications and treatment choices. Despite these limitations, our review provides essential information that must be interpreted in a clinical context.
CONCLUSION
Given that cutaneous and mucosal piercing has become more prevalent in recent years, along with an increase in the variety of piercing-induced complications, it is of utmost importance that piercing salons have proper hygiene practices in place and that patients are aware of the multitude of potential complications that can arise—whether common and benign or rare but life-threatening.
- Preslar D, Borger J. Body piercing infections. In: StatPearls. StatPearls Publishing; 2022.
- Antoszewski B, Jedrzejczak M, Kruk-Jeromin J. Complications after body piercing in patient suffering from type 1 diabetes mellitus. Int J Dermatol. 2007;46:1250-1252.
- Simplot TC, Hoffman HT. Comparison between cartilage and soft tissue ear piercing complications. Am J Otolaryngol. 1998;19:305-310.
- Meltzer DI. Complications of body piercing. Am Fam Physician. 2005;72:2029-2034.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
- Metts J. Common complications of body piercing. West J Med. 2002;176:85-86.
- Escudero-Castaño N, Perea-García MA, Campo-Trapero J, et al. Oral and perioral piercing complications. Open Dent J. 2008;2:133-136.
- Tweeten SS, Rickman LS. Infectious complications of body piercing. Clin Infect Dis. 1998;26:735-740.
- Gabriel OT, Anthony OO, Paul EA, et al. Trends and complications of ear piercing among selected Nigerian population. J Family Med Prim Care. 2017;6:517-521.
- Armstrong ML, Koch JR, Saunders JC, et al. The hole picture: risks, decision making, purpose, regulations, and the future of body piercing. Clin Dermatol. 2007;25:398-406.
- Millar BC, Moore JE. Antibiotic prophylaxis, body piercing and infective endocarditis. J Antimicrob Chemother. 2004;53:123-126.
- Mariano A, Pisapia R, Abdeddaim A, et al. Endocarditis and meningitis associated to nape piercing in a young female: a case report. Infez Med. 2015;23:275-279.
- Ivey LA, Limone BA, Jacob SE. Approach to the jewelry aficionado. Pediatr Dermatol. 2018;35:274-275.
- Brandão MH, Gontijo B, Girundi MA, et al. Ear piercing as a risk factor for contact allergy to nickel. J Pediatr (Rio J). 2010;86:149-154.
- Schuttelaar MLA, Ofenloch RF, Bruze M, et al. Prevalence of contact allergy to metals in the European general population with a focus on nickel and piercings: The EDEN Fragrance Study. Contact Dermatitis. 2018;79:1-9.
- Laumann AE, Derick AJ. Tattoos and body piercings in the United States: a national data set. J Am Acad Dermatol. 2006;55:413-421.
- De Moor RJ, De Witte AM, Delmé KI, et al. Dental and oral complications of lip and tongue piercings. Br Dent J. 2005;199:506-509.
- Offen E, Allison JR. Do oral piercings cause problems in the mouth? Evid Based Dent. 2022;23:126-127.
- Passos PF, Pintor AVB, Marañón-Vásquez GA, et al. Oral manifestations arising from oral piercings: A systematic review and meta-analyses. Oral Surg Oral Med Oral Pathol Oral Radiol. 2022;134:327-341.
- Covello F, Salerno C, Giovannini V, et al. Piercing and oral health: a study on the knowledge of risks and complications. Int J Environ Res Public Health. 2020;17:613.
- Ogawa R. The most current algorithms for the treatment and prevention of hypertrophic scars and keloids: a 2020 update of the algorithms published 10 years ago. Plast Reconstr Surg. 2022;149:E79-E94.
- Kumar Ghosh S, Bandyopadhyay D. Granuloma pyogenicum as a complication of decorative nose piercing: report of eight cases from eastern India. J Cutan Med Surg. 2012;16:197-200.
- Dreher K, Kern M, Rush L, et al. Basal cell carcinoma invasion of an ear piercing. Dermatol Online J. 2022;28.
- Stanko P, Poruban D, Mracna J, et al. Squamous cell carcinoma and piercing of the tongue—a case report. J Craniomaxillofac Surg. 2012;40:329-331.
The practice of body piercing has been present in cultures around the world for centuries. Piercings may be performed for religious or spiritual reasons or as a form of self-expression. In recent years, body piercings have become increasingly popular in all genders, with the most common sites being the ears, mouth, nose, eyebrows, nipples, navel, and genitals.1 The prevalence of body piercing in the general population is estimated to be as high as 50%.2 With the rising popularity of piercings, there also has been an increase in their associated complications, with one study noting that up to 35% of individuals with pierced ears and 30% of all pierced sites developed a complication.3 Common problems following piercing include infections, keloid formation, allergic contact dermatitis, site deformation, and tooth fractures.4 It is of utmost importance that health care professionals are aware of the potential complications associated with such a common practice. A comprehensive review of complications associated with cutaneous and mucosal piercings is lacking. We conducted a systematic review to summarize the clinical characteristics, complication types and frequency, and treatments reported for cutaneous and mucosal piercings.
METHODS
We conducted a systematic review of the literature adhering to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) reporting guidelines.5
Search Strategy, Study Eligibility Criteria, and Study Selection
A literature search of the Embase, MEDLINE, and PubMed databases was performed on June 20, 2022, using search terms related to body piercing and possible piercing-induced complications (Supplemental Information online). All studies reporting complications following body piercing were included. In vitro and animal studies were excluded. Title and abstract screening were completed by 6 independent researchers (S.C., K.K., M.M-B., K.A., T.S., I.M.M.) using Covidence online systematic review software (www.covidence.org). Six reviewers (S.C., K.K., M.M-B., K.A., T.S., I.M.M.) independently evaluated titles, abstracts, and full texts to identify relevant studies. Conflicts were resolved by the senior reviewer (I.M.M.).
Data Extraction and Synthesis
Five reviewers (S.C., K.K., M.M-B., K.A., T.S.) independently extracted data from eligible studies using a standardized extraction form that included title; authors; year of publication; sample size; and key findings, including mean age, sex, piercing location, complication type, and treatment received.
Treatment type was placed into the following categories: surgical treatments, antimicrobials, medical treatments, direct-target therapy, oral procedures, avoidance, miscellaneous therapies, and no treatment. (Data regarding treatments can be found in the Supplemental Information online.)
RESULTS
The combined search yielded 2679 studies, 617 of which underwent full-text review; 319 studies were included (Figure). Studies were published from 1950 to June 2022 and included both adult and pediatric populations.

Patient Characteristics
In total, our pooled analysis included data on 30,090 complications across 36,803 pierced sites in 30,231 patients (Table 1). Demographic data are available for 55% (n=30,231) of patients. Overall, 74% (22,247/30,231) of the individuals included in our analysis were female. The mean age was 27.8 years (range, 0–76 years).

Piercing Location
Overall, 36,803 pierced sites had a reported complication. The oral cavity, location not otherwise specified, was the most common site associated with a complication, accounting for 67% (n=24,478) of complications (Table 1). Other reported sites included (in decreasing frequency) the ears (21%, n=7551), tongue (5%, n=1669), lip (3%, n=998), navel (2%, n=605), nose (1%, n=540), nipple (1%, n=344), face/body (1%, n=269), genitals/groin (0%, n=183), eyebrow (0%, n=161), hand (0%, n=4), and eyelid (0%, n=1). Piercing complications were more commonly reported among females across all piercing locations except for the eyebrow, which was equal in both sexes.
Complications
Local Infections—Local infections accounted for 36% of reported complication types (n=10,872/30,090): perichondritis (1%, n=85); abscesses (0%, n=25); bacterial colonization (1%, n=106); and local infections, not otherwise specified (98%, n=10,648)(Table 2). The majority of local infections were found to be secondary to piercings of the ear and oral cavity. The nipple was found to be a common site for abscesses (40%, n=10), whereas the tongue was found to be the most common site for bacterial colonization (69%, n=73).


Immune-Mediated Issues—Immune-mediated issues encompassed 5% of the total reported complications (n=1561/30,090). The most commonly reported immune-mediated complications included allergies (31%, n=482), edema and swelling (21%, n=331), dermatitis (18%, n=282), and inflammatory lesions (17%, n=270). The majority were found to occur secondary to ear piercings, with the exception of edema, which mainly occurred secondary to tongue piercings (45%, n=150), and allergy, which primarily was associated with oral piercings (51%, n=245)(Table 2).
Tissue Damage—Tissue damage accounted for 43% of all complications (n=13,036/30,090). The most common forms of tissue damage were trauma (55%, n=7182), dysesthesia (22%, n=2883), bleeding and bruising (18%, n=2376), and pain (3%, n=370)(Table 2). Trauma was mainly found to be a complication in the context of oral piercings (99%, n=7121). Similarly, 94% (n=2242) of bleeding and bruising occurred secondary to oral piercings. Embedded piercings (92%, n=127), deformity (91%, n=29), and necrosis (75%, n=3) mostly occurred following ear piercings. Lip piercings were found to be the most common cause of damage to surrounding structures (98%, n=50).
Oral—Overall, 3193 intraoral complications were reported, constituting 11% of the total complications (Table 2). Oral complications included dental damage (86%, n=2732), gum recession (14%, n=459), and gingivitis (0%, n=2). Dental damage was mostly reported following oral piercings (90%, n=2453), whereas gum recession was mostly reported following lip piercings (59%, n=272).
Proliferations—Proliferations accounted for 795 (3%) of reported piercing complications. The majority (97%, n=772) were keloids, 2% (n=16) were other benign growths, and 1% (n=7) were malignancies. These complications mostly occurred secondary to ear piercings, which resulted in 741 (96%) keloids, 6 (38%) benign growths, and 4 (57%) malignancies.
Systemic—Overall, 2% (n=633) of the total complications were classified as systemic issues, including functional impairment (45%, n=282), secondary organ involvement (24%, n=150), cardiac issues (3%, n=21), and aspiration/inhalation (1%, n=8). Nonlocalized infections such as hepatitis or an increased risk thereof (17%, n=107), tetanus (8%, n=52), chlamydia (1%, n=9), HIV (0%, n=1), herpes simplex virus (0%, n=1), gonorrhea (0%, n=1), and bacterial vaginosis (0%, n=1) also were included in this category. The tongue, ear, and genitals were the locations most involved in these complications (Table 2). Secondary organ involvement mostly occurred after ear (36%, n=54) and genital piercings (27%, n=41). A total of 8 cases of piercing aspiration and/or inhalation were reported in association with piercings of the head and neck (Table 2).
COMMENT
Piercing Complications
Overall, the ear, tongue, and oral cavity were found to be the sites with the most associated complications recorded in the literature, and local infection and tissue damage were found to be the most prevalent types of complications. A plethora of treatments were used to manage piercing-induced complications, including surgical or medical treatments and avoidance (Supplemental Information). Reports by Metts6 and Escudero-Castaño et al7 provide detailed protocols and photographs of piercings.
Infections
Our review found that local infections were commonly reported complications associated with body piercings, which is consistent with other studies.1 The initial trauma inherent in the piercing process followed by the presence of an ongoing foreign body lends itself to an increased risk for developing these complications. Wound healing after piercing also varies based on the piercing location.
The rate and severity of the infection are influenced by the anatomic location of the piercing, hygiene, method of piercing, types of materials used, and aftercare.8 Piercing cartilage sites, such as the helix, concha, or nose, increases susceptibility to infections and permanent deformities. Cartilage is particularly at risk because of its avascular nature.9 Other studies have reported similar incidences of superficial localized infections; infectious complications were seen in 10% to 30% of body piercings in one study,3 while 45% of American and Australian college students reported infection at a piercing site in a second study.10
Systemic Issues
Systemic issues are potentially the most dangerous piercing-induced complications, though they were rarer in our analysis. Some serious complications included septic emboli, fatal staphylococcal toxic shock syndrome, and death. Although some systemic issues, such as staphylococcal toxic shock syndrome and septic sacroiliitis, required extensive hospital stays and complex treatment, others had lifelong repercussions, such as hepatitis and HIV. One report showed an increased incidence of endocarditis associated with body piercing, including staphylococcal endocarditis following nasal piercings, Neisseria endocarditis following tongue piercings, and Staphylococcus epidermidis endocarditis following nipple piercings.11 Moreover, Mariano et al12—who noted a case of endocarditis and meningitis associated with a nape piercing in a young female in 2015—reinforced the notion that information pertaining to the risks associated with body piercing must be better disseminated, given the potential for morbid or fatal outcomes. Finally, nonsterile piercing techniques and poor hygiene were found to contribute substantially to the increased risk for infection, so it is of utmost importance to reinforce proper practices in piercing salons.4
Immune-Mediated Issues
Because piercings are foreign bodies, they are susceptible to both acute and chronic immune responses. Our study found that allergies and dermatitis made up almost half of the immune-mediated piercing complications. It is especially important to emphasize that costume jewelry exposes our skin to a variety of contact allergens, most prominently nickel, heightening the risk for developing allergic contact dermatitis.13 Moreover, a study conducted by Brandão et al14 found that patients with pierced ears were significantly more likely to react to nickel than those without pierced ears (P=.031). Although other studies have found that allergy to metals ranges from 8.3% to 20% in the general population,15 we were not able to quantify the incidence in our study due to a lack of reporting of common benign complications, such as contact dermatitis.
Tissue Damage and Local Problems
Our review found that tissue and oral damage also were commonly reported piercing complications, with the most common pathologies being trauma, dysesthesia, bleeding/bruising, and dental damage. Laumann and Derick16 reported that bleeding, tissue trauma, and local problems were common physical health problems associated with body piercing. Severe complications, such as abscesses, toxic shock syndrome, and endocarditis, also have been reported in association with intraoral piercings.17 Moreover, other studies have shown that oral piercings are associated with several adverse oral and systemic conditions. A meta-analysis of individuals with oral piercings found a similar prevalence of dental fracture, gingival recession, and tooth wear (34%), as well as unspecified dental damage (27%) and tooth chipping (22%). Additionally, this meta-analysis reported a 3-fold increased risk for dental fracture and 7-fold increased risk for gingival recession with oral piercings.18 Another meta-analysis of oral piercing complications found a similar prevalence of dental fracture (34%), tooth wear (34%), gingival recession (33%), unspecified dental damage (27%), and tooth chipping (22%).19 Considering the extensive amount of cumulative damage, wearers of oral jewelry require periodic periodontal evaluations to monitor for dental damage and gingival recession.20 There are limited data on treatments for complications of oral piercings, and further research in this area is warranted.
Proliferations and Scars
Although proliferations and scarring were among the least common complications reported in the literature, they are some of the most cosmetically disfiguring for patients. Keloids, the most common type of growth associated with piercings, do not naturally regress and thus require some form of intervention. Given the multimodal approach used to treat keloids, as described by the evidence-based algorithm by Ogawa,21 it is not surprising that keloids also represented the complication most treated with medical therapies, such as steroids, and also with direct-target therapy, such as liquid nitrogen therapy (Supplemental Information).
Other proliferations reported in the literature include benign pyogenic granulomas22 and much less commonly malignant neoplasms such as basal cell carcinoma23 and squamous cell carcinoma.24 Although rare, treatment of piercing-associated malignancies include surgical removal, chemotherapy, and radiation therapy (Supplemental Information).
Limitations
There are several limitations to our systematic review. First, heterogeneity in study designs, patient populations, treatment interventions, and outcome measures of included studies may have affected the quality and generalizability of our results. Moreover, because the studies included in this systematic review focused on specific complications, we could not compare our results to the literature that analyzes incidence rates of piercing complications. Furthermore, not all studies included the data that we hoped to extract, and thus only available data were reported in these instances. Finally, the articles we reviewed may have included publication bias, with positive findings being more frequently published, potentially inflating certain types and sites of complications and treatment choices. Despite these limitations, our review provides essential information that must be interpreted in a clinical context.
CONCLUSION
Given that cutaneous and mucosal piercing has become more prevalent in recent years, along with an increase in the variety of piercing-induced complications, it is of utmost importance that piercing salons have proper hygiene practices in place and that patients are aware of the multitude of potential complications that can arise—whether common and benign or rare but life-threatening.
The practice of body piercing has been present in cultures around the world for centuries. Piercings may be performed for religious or spiritual reasons or as a form of self-expression. In recent years, body piercings have become increasingly popular in all genders, with the most common sites being the ears, mouth, nose, eyebrows, nipples, navel, and genitals.1 The prevalence of body piercing in the general population is estimated to be as high as 50%.2 With the rising popularity of piercings, there also has been an increase in their associated complications, with one study noting that up to 35% of individuals with pierced ears and 30% of all pierced sites developed a complication.3 Common problems following piercing include infections, keloid formation, allergic contact dermatitis, site deformation, and tooth fractures.4 It is of utmost importance that health care professionals are aware of the potential complications associated with such a common practice. A comprehensive review of complications associated with cutaneous and mucosal piercings is lacking. We conducted a systematic review to summarize the clinical characteristics, complication types and frequency, and treatments reported for cutaneous and mucosal piercings.
METHODS
We conducted a systematic review of the literature adhering to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) reporting guidelines.5
Search Strategy, Study Eligibility Criteria, and Study Selection
A literature search of the Embase, MEDLINE, and PubMed databases was performed on June 20, 2022, using search terms related to body piercing and possible piercing-induced complications (Supplemental Information online). All studies reporting complications following body piercing were included. In vitro and animal studies were excluded. Title and abstract screening were completed by 6 independent researchers (S.C., K.K., M.M-B., K.A., T.S., I.M.M.) using Covidence online systematic review software (www.covidence.org). Six reviewers (S.C., K.K., M.M-B., K.A., T.S., I.M.M.) independently evaluated titles, abstracts, and full texts to identify relevant studies. Conflicts were resolved by the senior reviewer (I.M.M.).
Data Extraction and Synthesis
Five reviewers (S.C., K.K., M.M-B., K.A., T.S.) independently extracted data from eligible studies using a standardized extraction form that included title; authors; year of publication; sample size; and key findings, including mean age, sex, piercing location, complication type, and treatment received.
Treatment type was placed into the following categories: surgical treatments, antimicrobials, medical treatments, direct-target therapy, oral procedures, avoidance, miscellaneous therapies, and no treatment. (Data regarding treatments can be found in the Supplemental Information online.)
RESULTS
The combined search yielded 2679 studies, 617 of which underwent full-text review; 319 studies were included (Figure). Studies were published from 1950 to June 2022 and included both adult and pediatric populations.

Patient Characteristics
In total, our pooled analysis included data on 30,090 complications across 36,803 pierced sites in 30,231 patients (Table 1). Demographic data are available for 55% (n=30,231) of patients. Overall, 74% (22,247/30,231) of the individuals included in our analysis were female. The mean age was 27.8 years (range, 0–76 years).

Piercing Location
Overall, 36,803 pierced sites had a reported complication. The oral cavity, location not otherwise specified, was the most common site associated with a complication, accounting for 67% (n=24,478) of complications (Table 1). Other reported sites included (in decreasing frequency) the ears (21%, n=7551), tongue (5%, n=1669), lip (3%, n=998), navel (2%, n=605), nose (1%, n=540), nipple (1%, n=344), face/body (1%, n=269), genitals/groin (0%, n=183), eyebrow (0%, n=161), hand (0%, n=4), and eyelid (0%, n=1). Piercing complications were more commonly reported among females across all piercing locations except for the eyebrow, which was equal in both sexes.
Complications
Local Infections—Local infections accounted for 36% of reported complication types (n=10,872/30,090): perichondritis (1%, n=85); abscesses (0%, n=25); bacterial colonization (1%, n=106); and local infections, not otherwise specified (98%, n=10,648)(Table 2). The majority of local infections were found to be secondary to piercings of the ear and oral cavity. The nipple was found to be a common site for abscesses (40%, n=10), whereas the tongue was found to be the most common site for bacterial colonization (69%, n=73).


Immune-Mediated Issues—Immune-mediated issues encompassed 5% of the total reported complications (n=1561/30,090). The most commonly reported immune-mediated complications included allergies (31%, n=482), edema and swelling (21%, n=331), dermatitis (18%, n=282), and inflammatory lesions (17%, n=270). The majority were found to occur secondary to ear piercings, with the exception of edema, which mainly occurred secondary to tongue piercings (45%, n=150), and allergy, which primarily was associated with oral piercings (51%, n=245)(Table 2).
Tissue Damage—Tissue damage accounted for 43% of all complications (n=13,036/30,090). The most common forms of tissue damage were trauma (55%, n=7182), dysesthesia (22%, n=2883), bleeding and bruising (18%, n=2376), and pain (3%, n=370)(Table 2). Trauma was mainly found to be a complication in the context of oral piercings (99%, n=7121). Similarly, 94% (n=2242) of bleeding and bruising occurred secondary to oral piercings. Embedded piercings (92%, n=127), deformity (91%, n=29), and necrosis (75%, n=3) mostly occurred following ear piercings. Lip piercings were found to be the most common cause of damage to surrounding structures (98%, n=50).
Oral—Overall, 3193 intraoral complications were reported, constituting 11% of the total complications (Table 2). Oral complications included dental damage (86%, n=2732), gum recession (14%, n=459), and gingivitis (0%, n=2). Dental damage was mostly reported following oral piercings (90%, n=2453), whereas gum recession was mostly reported following lip piercings (59%, n=272).
Proliferations—Proliferations accounted for 795 (3%) of reported piercing complications. The majority (97%, n=772) were keloids, 2% (n=16) were other benign growths, and 1% (n=7) were malignancies. These complications mostly occurred secondary to ear piercings, which resulted in 741 (96%) keloids, 6 (38%) benign growths, and 4 (57%) malignancies.
Systemic—Overall, 2% (n=633) of the total complications were classified as systemic issues, including functional impairment (45%, n=282), secondary organ involvement (24%, n=150), cardiac issues (3%, n=21), and aspiration/inhalation (1%, n=8). Nonlocalized infections such as hepatitis or an increased risk thereof (17%, n=107), tetanus (8%, n=52), chlamydia (1%, n=9), HIV (0%, n=1), herpes simplex virus (0%, n=1), gonorrhea (0%, n=1), and bacterial vaginosis (0%, n=1) also were included in this category. The tongue, ear, and genitals were the locations most involved in these complications (Table 2). Secondary organ involvement mostly occurred after ear (36%, n=54) and genital piercings (27%, n=41). A total of 8 cases of piercing aspiration and/or inhalation were reported in association with piercings of the head and neck (Table 2).
COMMENT
Piercing Complications
Overall, the ear, tongue, and oral cavity were found to be the sites with the most associated complications recorded in the literature, and local infection and tissue damage were found to be the most prevalent types of complications. A plethora of treatments were used to manage piercing-induced complications, including surgical or medical treatments and avoidance (Supplemental Information). Reports by Metts6 and Escudero-Castaño et al7 provide detailed protocols and photographs of piercings.
Infections
Our review found that local infections were commonly reported complications associated with body piercings, which is consistent with other studies.1 The initial trauma inherent in the piercing process followed by the presence of an ongoing foreign body lends itself to an increased risk for developing these complications. Wound healing after piercing also varies based on the piercing location.
The rate and severity of the infection are influenced by the anatomic location of the piercing, hygiene, method of piercing, types of materials used, and aftercare.8 Piercing cartilage sites, such as the helix, concha, or nose, increases susceptibility to infections and permanent deformities. Cartilage is particularly at risk because of its avascular nature.9 Other studies have reported similar incidences of superficial localized infections; infectious complications were seen in 10% to 30% of body piercings in one study,3 while 45% of American and Australian college students reported infection at a piercing site in a second study.10
Systemic Issues
Systemic issues are potentially the most dangerous piercing-induced complications, though they were rarer in our analysis. Some serious complications included septic emboli, fatal staphylococcal toxic shock syndrome, and death. Although some systemic issues, such as staphylococcal toxic shock syndrome and septic sacroiliitis, required extensive hospital stays and complex treatment, others had lifelong repercussions, such as hepatitis and HIV. One report showed an increased incidence of endocarditis associated with body piercing, including staphylococcal endocarditis following nasal piercings, Neisseria endocarditis following tongue piercings, and Staphylococcus epidermidis endocarditis following nipple piercings.11 Moreover, Mariano et al12—who noted a case of endocarditis and meningitis associated with a nape piercing in a young female in 2015—reinforced the notion that information pertaining to the risks associated with body piercing must be better disseminated, given the potential for morbid or fatal outcomes. Finally, nonsterile piercing techniques and poor hygiene were found to contribute substantially to the increased risk for infection, so it is of utmost importance to reinforce proper practices in piercing salons.4
Immune-Mediated Issues
Because piercings are foreign bodies, they are susceptible to both acute and chronic immune responses. Our study found that allergies and dermatitis made up almost half of the immune-mediated piercing complications. It is especially important to emphasize that costume jewelry exposes our skin to a variety of contact allergens, most prominently nickel, heightening the risk for developing allergic contact dermatitis.13 Moreover, a study conducted by Brandão et al14 found that patients with pierced ears were significantly more likely to react to nickel than those without pierced ears (P=.031). Although other studies have found that allergy to metals ranges from 8.3% to 20% in the general population,15 we were not able to quantify the incidence in our study due to a lack of reporting of common benign complications, such as contact dermatitis.
Tissue Damage and Local Problems
Our review found that tissue and oral damage also were commonly reported piercing complications, with the most common pathologies being trauma, dysesthesia, bleeding/bruising, and dental damage. Laumann and Derick16 reported that bleeding, tissue trauma, and local problems were common physical health problems associated with body piercing. Severe complications, such as abscesses, toxic shock syndrome, and endocarditis, also have been reported in association with intraoral piercings.17 Moreover, other studies have shown that oral piercings are associated with several adverse oral and systemic conditions. A meta-analysis of individuals with oral piercings found a similar prevalence of dental fracture, gingival recession, and tooth wear (34%), as well as unspecified dental damage (27%) and tooth chipping (22%). Additionally, this meta-analysis reported a 3-fold increased risk for dental fracture and 7-fold increased risk for gingival recession with oral piercings.18 Another meta-analysis of oral piercing complications found a similar prevalence of dental fracture (34%), tooth wear (34%), gingival recession (33%), unspecified dental damage (27%), and tooth chipping (22%).19 Considering the extensive amount of cumulative damage, wearers of oral jewelry require periodic periodontal evaluations to monitor for dental damage and gingival recession.20 There are limited data on treatments for complications of oral piercings, and further research in this area is warranted.
Proliferations and Scars
Although proliferations and scarring were among the least common complications reported in the literature, they are some of the most cosmetically disfiguring for patients. Keloids, the most common type of growth associated with piercings, do not naturally regress and thus require some form of intervention. Given the multimodal approach used to treat keloids, as described by the evidence-based algorithm by Ogawa,21 it is not surprising that keloids also represented the complication most treated with medical therapies, such as steroids, and also with direct-target therapy, such as liquid nitrogen therapy (Supplemental Information).
Other proliferations reported in the literature include benign pyogenic granulomas22 and much less commonly malignant neoplasms such as basal cell carcinoma23 and squamous cell carcinoma.24 Although rare, treatment of piercing-associated malignancies include surgical removal, chemotherapy, and radiation therapy (Supplemental Information).
Limitations
There are several limitations to our systematic review. First, heterogeneity in study designs, patient populations, treatment interventions, and outcome measures of included studies may have affected the quality and generalizability of our results. Moreover, because the studies included in this systematic review focused on specific complications, we could not compare our results to the literature that analyzes incidence rates of piercing complications. Furthermore, not all studies included the data that we hoped to extract, and thus only available data were reported in these instances. Finally, the articles we reviewed may have included publication bias, with positive findings being more frequently published, potentially inflating certain types and sites of complications and treatment choices. Despite these limitations, our review provides essential information that must be interpreted in a clinical context.
CONCLUSION
Given that cutaneous and mucosal piercing has become more prevalent in recent years, along with an increase in the variety of piercing-induced complications, it is of utmost importance that piercing salons have proper hygiene practices in place and that patients are aware of the multitude of potential complications that can arise—whether common and benign or rare but life-threatening.
- Preslar D, Borger J. Body piercing infections. In: StatPearls. StatPearls Publishing; 2022.
- Antoszewski B, Jedrzejczak M, Kruk-Jeromin J. Complications after body piercing in patient suffering from type 1 diabetes mellitus. Int J Dermatol. 2007;46:1250-1252.
- Simplot TC, Hoffman HT. Comparison between cartilage and soft tissue ear piercing complications. Am J Otolaryngol. 1998;19:305-310.
- Meltzer DI. Complications of body piercing. Am Fam Physician. 2005;72:2029-2034.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
- Metts J. Common complications of body piercing. West J Med. 2002;176:85-86.
- Escudero-Castaño N, Perea-García MA, Campo-Trapero J, et al. Oral and perioral piercing complications. Open Dent J. 2008;2:133-136.
- Tweeten SS, Rickman LS. Infectious complications of body piercing. Clin Infect Dis. 1998;26:735-740.
- Gabriel OT, Anthony OO, Paul EA, et al. Trends and complications of ear piercing among selected Nigerian population. J Family Med Prim Care. 2017;6:517-521.
- Armstrong ML, Koch JR, Saunders JC, et al. The hole picture: risks, decision making, purpose, regulations, and the future of body piercing. Clin Dermatol. 2007;25:398-406.
- Millar BC, Moore JE. Antibiotic prophylaxis, body piercing and infective endocarditis. J Antimicrob Chemother. 2004;53:123-126.
- Mariano A, Pisapia R, Abdeddaim A, et al. Endocarditis and meningitis associated to nape piercing in a young female: a case report. Infez Med. 2015;23:275-279.
- Ivey LA, Limone BA, Jacob SE. Approach to the jewelry aficionado. Pediatr Dermatol. 2018;35:274-275.
- Brandão MH, Gontijo B, Girundi MA, et al. Ear piercing as a risk factor for contact allergy to nickel. J Pediatr (Rio J). 2010;86:149-154.
- Schuttelaar MLA, Ofenloch RF, Bruze M, et al. Prevalence of contact allergy to metals in the European general population with a focus on nickel and piercings: The EDEN Fragrance Study. Contact Dermatitis. 2018;79:1-9.
- Laumann AE, Derick AJ. Tattoos and body piercings in the United States: a national data set. J Am Acad Dermatol. 2006;55:413-421.
- De Moor RJ, De Witte AM, Delmé KI, et al. Dental and oral complications of lip and tongue piercings. Br Dent J. 2005;199:506-509.
- Offen E, Allison JR. Do oral piercings cause problems in the mouth? Evid Based Dent. 2022;23:126-127.
- Passos PF, Pintor AVB, Marañón-Vásquez GA, et al. Oral manifestations arising from oral piercings: A systematic review and meta-analyses. Oral Surg Oral Med Oral Pathol Oral Radiol. 2022;134:327-341.
- Covello F, Salerno C, Giovannini V, et al. Piercing and oral health: a study on the knowledge of risks and complications. Int J Environ Res Public Health. 2020;17:613.
- Ogawa R. The most current algorithms for the treatment and prevention of hypertrophic scars and keloids: a 2020 update of the algorithms published 10 years ago. Plast Reconstr Surg. 2022;149:E79-E94.
- Kumar Ghosh S, Bandyopadhyay D. Granuloma pyogenicum as a complication of decorative nose piercing: report of eight cases from eastern India. J Cutan Med Surg. 2012;16:197-200.
- Dreher K, Kern M, Rush L, et al. Basal cell carcinoma invasion of an ear piercing. Dermatol Online J. 2022;28.
- Stanko P, Poruban D, Mracna J, et al. Squamous cell carcinoma and piercing of the tongue—a case report. J Craniomaxillofac Surg. 2012;40:329-331.
- Preslar D, Borger J. Body piercing infections. In: StatPearls. StatPearls Publishing; 2022.
- Antoszewski B, Jedrzejczak M, Kruk-Jeromin J. Complications after body piercing in patient suffering from type 1 diabetes mellitus. Int J Dermatol. 2007;46:1250-1252.
- Simplot TC, Hoffman HT. Comparison between cartilage and soft tissue ear piercing complications. Am J Otolaryngol. 1998;19:305-310.
- Meltzer DI. Complications of body piercing. Am Fam Physician. 2005;72:2029-2034.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
- Metts J. Common complications of body piercing. West J Med. 2002;176:85-86.
- Escudero-Castaño N, Perea-García MA, Campo-Trapero J, et al. Oral and perioral piercing complications. Open Dent J. 2008;2:133-136.
- Tweeten SS, Rickman LS. Infectious complications of body piercing. Clin Infect Dis. 1998;26:735-740.
- Gabriel OT, Anthony OO, Paul EA, et al. Trends and complications of ear piercing among selected Nigerian population. J Family Med Prim Care. 2017;6:517-521.
- Armstrong ML, Koch JR, Saunders JC, et al. The hole picture: risks, decision making, purpose, regulations, and the future of body piercing. Clin Dermatol. 2007;25:398-406.
- Millar BC, Moore JE. Antibiotic prophylaxis, body piercing and infective endocarditis. J Antimicrob Chemother. 2004;53:123-126.
- Mariano A, Pisapia R, Abdeddaim A, et al. Endocarditis and meningitis associated to nape piercing in a young female: a case report. Infez Med. 2015;23:275-279.
- Ivey LA, Limone BA, Jacob SE. Approach to the jewelry aficionado. Pediatr Dermatol. 2018;35:274-275.
- Brandão MH, Gontijo B, Girundi MA, et al. Ear piercing as a risk factor for contact allergy to nickel. J Pediatr (Rio J). 2010;86:149-154.
- Schuttelaar MLA, Ofenloch RF, Bruze M, et al. Prevalence of contact allergy to metals in the European general population with a focus on nickel and piercings: The EDEN Fragrance Study. Contact Dermatitis. 2018;79:1-9.
- Laumann AE, Derick AJ. Tattoos and body piercings in the United States: a national data set. J Am Acad Dermatol. 2006;55:413-421.
- De Moor RJ, De Witte AM, Delmé KI, et al. Dental and oral complications of lip and tongue piercings. Br Dent J. 2005;199:506-509.
- Offen E, Allison JR. Do oral piercings cause problems in the mouth? Evid Based Dent. 2022;23:126-127.
- Passos PF, Pintor AVB, Marañón-Vásquez GA, et al. Oral manifestations arising from oral piercings: A systematic review and meta-analyses. Oral Surg Oral Med Oral Pathol Oral Radiol. 2022;134:327-341.
- Covello F, Salerno C, Giovannini V, et al. Piercing and oral health: a study on the knowledge of risks and complications. Int J Environ Res Public Health. 2020;17:613.
- Ogawa R. The most current algorithms for the treatment and prevention of hypertrophic scars and keloids: a 2020 update of the algorithms published 10 years ago. Plast Reconstr Surg. 2022;149:E79-E94.
- Kumar Ghosh S, Bandyopadhyay D. Granuloma pyogenicum as a complication of decorative nose piercing: report of eight cases from eastern India. J Cutan Med Surg. 2012;16:197-200.
- Dreher K, Kern M, Rush L, et al. Basal cell carcinoma invasion of an ear piercing. Dermatol Online J. 2022;28.
- Stanko P, Poruban D, Mracna J, et al. Squamous cell carcinoma and piercing of the tongue—a case report. J Craniomaxillofac Surg. 2012;40:329-331.
Practice Points
- Intraoral piercings of the tongue, lip, gingiva, or mucosa are associated with the most acute and chronic complications.
- Tissue damage is a common complication associated with cutaneous and mucocutaneous piercings, including trauma, bleeding and bruising, or dysesthesia.
- Given the rapid rise in the popularity of piercings, general practitioners and dermatologists should be aware of the multitude of acute or chronic complications associated with body piercings as well as effective treatment modalities.
Adenomyosis: Why we need to reassess our understanding of this condition
CASE Painful, heavy menstruation and recurrent pregnancy loss
A 37-year-old woman (G3P0030) with a history of recurrent pregnancy loss presents for evaluation. She had 3 losses—most recently a miscarriage at 22 weeks with a cerclage in place. She did not undergo any surgical procedures for these losses. Hormonal and thrombophilia workup is negative and semen analysis is normal. She reports a history of painful, heavy periods for many years, as well as dyspareunia and occasional post-coital bleeding. Past medical history was otherwise unremarkable. Pelvic magnetic resonance imaging (MRI) revealed focal thickening of the junctional zone up to 15 mm with 2 foci of T2 hyperintensities suggesting adenomyosis (FIGURE 1).
How do you counsel this patient regarding the MRI findings and their impact on her fertility?
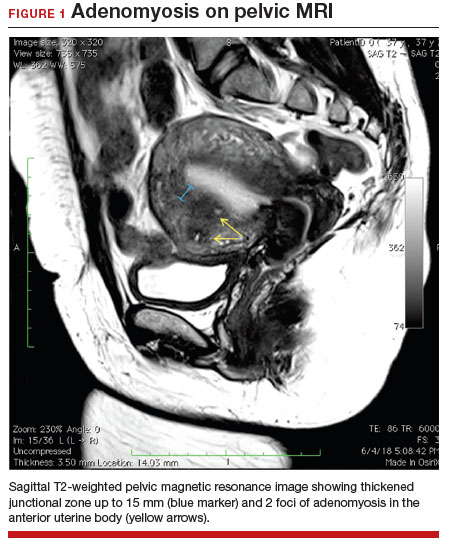
Adenomyosis is a condition in which endometrial glands and stroma are abnormally present in the uterine myometrium, resulting in smooth muscle hypertrophy and abnormal uterine contractility. Traditional teaching describes a woman in her 40s with heavy and painful menses, a “boggy uterus” on examination, who has completed childbearing and desires definitive treatment. Histologic diagnosis of adenomyosis is made from the uterine specimen at the time of hysterectomy, invariably confounding our understanding of the epidemiology of adenomyosis.
More recently, however, we are beginning to learn that this narrative is misguided. Imaging changes of adenomyosis can be seen in women who desire future fertility and in adolescents with severe dysmenorrhea, suggesting an earlier age of incidence.1 In a recent systematic review, prevalence estimates ranged from 15% to 67%, owing to varying diagnostic methods and patient inclusion criteria.2 It is increasingly being recognized as a primary contributor to infertility, with one study estimating a 30% prevalence of infertility in women with adenomyosis.3 Moreover, treatment with gonadotropin-releasing hormone agonists and/or surgical excision may improve fertility outcomes.4
As we learn more about this prevalent and life-altering condition, we owe it to our patients to consider this diagnosis when counseling on dysmenorrhea, heavy menstrual bleeding, or infertility.
Anatomy of the myometrium
The myometrium is composed of the inner and outer myometrium: the inner myometrium (IM) and endometrium are of Müllerian origin, and the outer myometrium (OM) is of mesenchymal origin. The IM thickens in response to steroid hormones during the menstrual cycle with metaplasia of endometrial stromal cells into myocytes and back again, whereas the OM is not responsive to hormones.5 Emerging literature suggests the OM is further divided into a middle and outer section based on different histologic morphologies, though the clinical implications of this are not understood.6 The term “junctional zone” (JZ) refers to the imaging appearance of what is thought to be the IM. Interestingly it cannot be identified on traditional hematoxylin and eosin staining. When the JZ is thickened or demonstrates irregular borders, it is used as a diagnostic marker for adenomyosis and is postulated to play an important role in adenomyosis pathophysiology, particularly heavy menstrual bleeding and infertility.7
Continue to: Subtypes of adenomyosis...
Subtypes of adenomyosis
While various disease classifications have been suggested for adenomyosis, to date there is no international consensus. Adenomyosis is typically described in 3 forms: diffuse, focal, or adenomyoma.8 As implied, the term focal adenomyosis refers to discrete lesions surrounded by normal myometrium, whereas abnormal glandular changes are pervasive throughout the myometrium in diffuse disease. Adenomyomas are a subgroup of focal adenomyosis that are thought to be surrounded by leiomyomatous smooth muscle and may be well demarcated on imaging.9
Recent research uses novel histologic imaging techniques to explore adenomyotic growth patterns in 3-dimensional (3D) reconstructions. Combining tissue-clearing methods with light-sheet fluorescence microscopy enables highly detailed 3D representations of the protein and nucleic acid structure of organs.10 For example, Yamaguchi and colleagues used this technology to explore the 3D morphological features of adenomyotic tissue and observed direct invasion of the endometrial glands into the myometrium and an “ant colony ̶ like network” of ectopic endometrial glands in the myometrium (FIGURE 2).11 These abnormal glandular networks have been visualized beyond the IM, which may not be captured on ultrasonography or MRI. While this work is still in its infancy, it has the potential to provide important insight into disease pathogenesis and to inform future therapy.
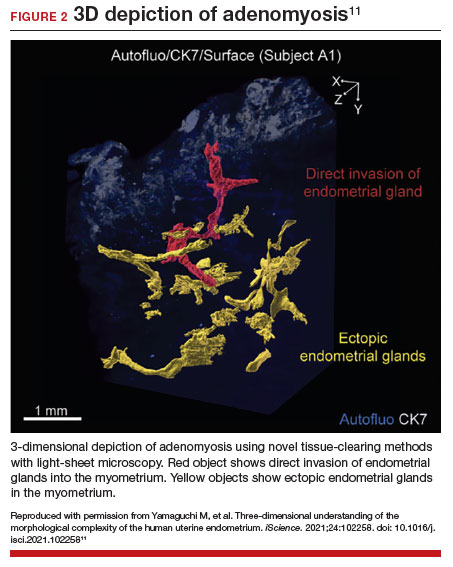
Pathogenesis
Proposed mechanisms for the development of adenomyosis include endometrial invasion, tissue injury and repair (TIAR) mechanisms, and the stem cell theory.12 According to the endometrial invasion theory, glandular epithelial cells from the basalis layer invaginate through an altered IM, slipping through weak muscle fibers and attracted by certain growth factors. In the TIAR mechanism theory, micro- or macro-trauma to the IM (whether from pregnancy, surgery, or infection) results in chronic proliferation and inflammation leading to the development of adenomyosis. Finally, the stem cell theory proposes that adenomyosis might develop from de novo ectopic endometrial tissue.
While the exact pathogenesis of adenomyosis is largely unknown, it has been associated with predictable molecular changes in the endometrium and surrounding myometrium.12 Myometrial hypercontractility is seen in patients with adenomyosis and dysmenorrhea, whereas neovascularization, high microvessel density, and abnormal uterine contractility are seen in those with abnormal uterine bleeding.13 In patients with infertility, increased inflammation, abnormal endometrial receptivity, and alterations in the myometrial architecture have been suggested to impair contractility and sperm transport.12,14
Differential growth factor expression and abnormal estrogen and progesterone signaling pathways have been observed in the IM in patients with adenomyosis, along with dysregulation of immune factors and increased inflammatory oxidative stress.12 This in turn results in myometrial hypertrophy and fibrosis, impairing normal uterine contractility patterns. This abnormal contractility may alter sperm transport and embryo implantation, and animal models that target pathways leading to fibrosis may improve endometrial receptivity.14,15 Further research is needed to elucidate specific molecular pathways and their complex interplay in this disease.
Continue to: Diagnosis...
Diagnosis
The gold standard for diagnosis of adenomyosis is histopathology from hysterectomy specimens, but specific definitions vary. Published criteria include endometrial glands within the myometrial layer greater than 0.5 to 1 low power field from the basal layer of the endometrium, endometrial glands extending deeper than 25% of the myometrial thickness, or endometrial glands a certain distance (ranging from 1-3 mm) from the basalis layer of the endometrium.16 Various methods of non-hysterectomy tissue sampling have been proposed for diagnosis, including needle, hysteroscopic, or laparoscopic sampling, but the sensitivity of these methods is poor.17 Limiting the diagnosis of adenomyosis to specimen pathology relies on invasive methods and clearly we cannot confirm the diagnosis by hysterectomy in patients with a desire for future fertility. It is for this reason that the prevalence of the disease is widely unknown.
The alternative to pathologic diagnosis is to identify radiologic changes that are associated with adenomyosis via either transvaginal ultrasound (TVUS) or MRI. Features suggestive of adenomyosis on MRI overlap with TVUS features, including uterine enlargement, anteroposterior myometrial asymmetry, T1- or T2-intense myometrial cysts or foci, and a thickened JZ.18 A JZ thicker than 12 mm has been thought to be predictive of adenomyosis, whereas a thickness of less than 8 mm is predictive of its absence, although the JZ may vary in thickness with the menstrual cycle.19,20 A 2021 systematic review and meta-analysis comparing MRI diagnosis with histopathologic findings reported a pooled sensitivity and specificity of 60% and 96%, respectively.21 The reported range for sensitivity and specificity is wide: 70% to 93% for sensitivity and 67% to 93% for specificity.22-24
Key TVUS features associated with adenomyosis were defined in 2015 in a consensus statement released by the Morphological Uterus Sonographic Assessment (MUSA) group.25 These include a globally enlarged uterus, anteroposterior myometrial asymmetry, myometrial cysts, fan-shaped shadowing, mixed myometrial echogenicity, translesional vascularity, echogenic subendometrial lines and buds, and a thickened, irregular or discontinuous JZ (FIGURES 3 and 4).25 The accuracy of ultrasonographic diagnosis of adenomyosis using these features has been investigated in multiple systematic reviews and meta-analyses, most recently by Liu and colleagues who found a pooled sensitivity of TVUS of 81% and pooled specificity of 87%.23 The range for ultrasonographic sensitivity and specificity is wide, however, ranging from 33% to 84% for sensitivity and 64% to 100% for specificity.22 Consensus is lacking as to which TVUS features are most predictive of adenomyosis, but in general, the combination of multiple MUSA criteria (particularly myometrial cysts and irregular JZ on 3D imaging) appears to be more accurate than any one feature alone.23 The presence of fibroids may decrease the sensitivity of TVUS, and one study suggested elastography may increase the accuracy of TVUS.24,26 Moreover, given that most radiologists receive limited training on the MUSA criteria, it behooves gynecologists to become familiar with these sonographic features to be able to identify adenomyosis in our patients.
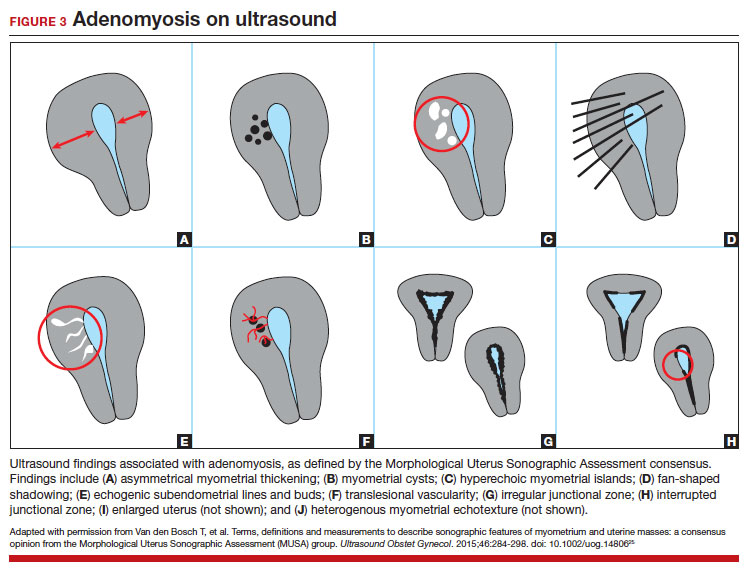

Adenomyosis also may be suspected based on hysteroscopic findings, although a normal hysteroscopy cannot rule out the disease and data are lacking to support these markers as diagnostic. Visual findings can include a “strawberry” pattern, mucosal elevation, cystic hemorrhagic lesions, localized vascularity, or endometrial defects.27 Hysteroscopy may be effective in the treatment of localized lesions, although that discussion is beyond the scope of this review.
Clinical presentation
While many women who are later diagnosed with adenomyosis are asymptomatic, the disease can present with heavy menstrual bleeding and dysmenorrhea, which occur in 50% and 30% of patients, respectively.28 Other symptoms include dyspareunia and infertility. Symptoms were previously reported to develop between the ages of 40 and 50 years; however, this is biased by diagnosis at the time of hysterectomy and the fact that younger patients are less likely to undergo definitive surgery. When using imaging criteria for diagnosis, adenomyosis might be more responsible for dysmenorrhea and chronic pelvic pain in younger patients than previously appreciated.1,29 In a recent study reviewing TVUS in 270 adolescents for any reason, adenomyosis was present in 5% of cases and this increased up to 44% in the presence of endometriosis.30
Adenomyosis often co-exists and shares similar clinical presentations with other gynecologic pathologies such as endometriosis and fibroids, making diagnosis on symptomatology alone challenging. Concurrent adenomyosis has been found in up to 73% and 57% of patients with suspected or diagnosed endometriosis and fibroids, respectively.31,32 Accumulating evidence suggests that pelvic pain previously attributed to endometriosis may in fact be a result of adenomyosis; for example, persistent pelvic pain after optimal resection of endometriosis may be confounded by the presence of adenomyosis.29 In one study of 155 patients with complete resection of deep infiltrating endometriosis, persistent pelvic pain was significantly associated with the presence of adenomyosis on imaging.33
Adenomyosis is increasingly being recognized at the time of infertility evaluation with an estimated prevalence of 30% in women with infertility.3 Among women with infertility, adenomyosis has been associated with a lower clinical pregnancy rate, higher miscarriage rate, and lower live birth rate, as well as obstetric complications such as abnormal placentation.34-36 A study of 37 baboons found the histologic diagnosis of adenomyosis alone at necropsy was associated with a 20-fold increased risk of lifelong infertility (odds ratio [OR], 20.1; 95% CI, 2.1-921), whereas presence of endometriosis was associated with a nonsignificant 3-fold risk of lifelong infertility (OR, 3.6; 95% CI, 0.9-15.8).37
In women with endometriosis and infertility, co-existing adenomyosis portends worse fertility outcomes. In a retrospective study of 244 women who underwent endometriosis surgery, more than five features of adenomyosis on imaging was associated with higher rates of infertility, in vitro fertilization treatments, and a higher number of in vitro fertilization cycles.31 Moreover, in women who underwent surgery for deep infiltrating endometriosis, the presence of adenomyosis on imaging was associated with a 68% reduction in likelihood of pregnancy after surgery.38
Conclusion
As we begin to learn about adenomyosis, our misconceptions become more evident. The notion that it largely affects women at the end of their reproductive lives is biased by using histopathology at hysterectomy as the gold standard for diagnosis. Lack of definitive histologic or imaging criteria and biopsy techniques add to the diagnostic challenge. This in turn leads to inaccurate estimates of incidence and prevalence, as we assume patients’ symptoms must be attributable to what we can see at the time of surgery (for example, Stage I or II endometriosis), rather than what we cannot see. We now know that adenomyosis is present in women of all ages, including adolescents, and can significantly contribute to reduced fertility and quality of life. We owe it to our patients to consider this condition in the differential diagnosis of dysmenorrhea, heavy menstrual bleeding, dyspareunia, and infertility.
CASE Resolved
The patient underwent targeted hysteroscopic resection of adenomyosis (FIGURE 5) and conceived spontaneously the following year. ●
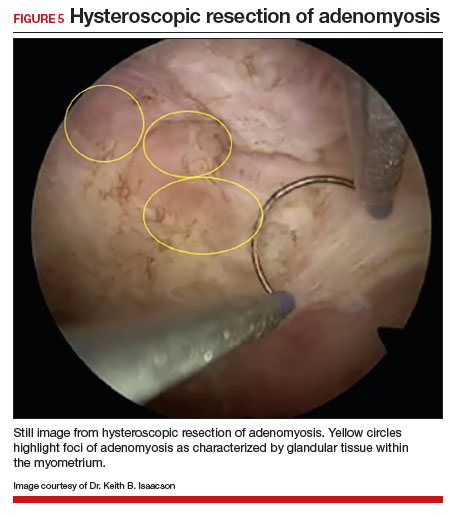
- Exacoustos C, Lazzeri L, Martire FG, et al. Ultrasound findings of adenomyosis in adolescents: type and grade of the disease. J Minim Invasive Gynecol. 2021;29:291.e1-299.e1. doi: 10.1016/j.jmig.2021.08.023
- Loring M, Chen TY, Isaacson KB. A systematic review of adenomyosis: it is time to reassess what we thought we knew about the disease. J Minim Invasive Gynecol. 2021;28:644655. doi: 10.1016/j.jmig.2020.10.012
- Bourdon M, Santulli P, Oliveira J, et al. Focal adenomyosis is associated with primary infertility. Fertil Steril. 2020;114:1271-1277. doi: 10.1016/j.fertnstert.2020.06.018
- Lan J, Wu Y, Wu Z, et al. Ultra-long GnRH agonist protocol during IVF/ICSI improves pregnancy outcomes in women with adenomyosis: a retrospective cohort study. Front Endocrinol (Lausanne). 2021;12:609771. doi: 10.3389 /fendo.2021.609771
- Gnecco JS, Brown AT, Kan EL, et al. Physiomimetic models of adenomyosis. Semin Reprod Med. 2020;38:179-196. doi: 10.1055/s-0040-1719084
- Harmsen MJ, Trommelen LM, de Leeuw RA, et al. Uterine junctional zone and adenomyosis: comparison of MRI, transvaginal ultrasound and histology. Ultrasound Obstet Gynecol. 2023;62:42-60. doi: 10.1002/uog.26117
- Xie T, Xu X, Yang Y, et al. The role of abnormal uterine junction zone in the occurrence and development of adenomyosis. Reprod Sci. 2022;29:2719-2730. doi: 10.1007/s43032-021 -00684-2
- Lazzeri L, Morosetti G, Centini G, et al. A sonographic classification of adenomyosis: interobserver reproducibility in the evaluation of type and degree of the myometrial involvement. Fertil Steril. 2018;110:1154-1161.e3. doi: 10.1016 /j.fertnstert.2018.06.031
- Tahlan A, Nanda A, Mohan H. Uterine adenomyoma: a clinicopathologic review of 26 cases and a review of the literature. Int J Gynecol Pathol. 2006;25:361-365. doi: 10.1097/01.pgp.0000209570.08716.b3
- Chung K, Wallace J, Kim S-Y, et al. Structural and molecular interrogation of intact biological systems. Nature. 2013;497:332-337. doi: 10.1038/nature12107
- Yamaguchi M, Yoshihara K, Suda K, et al. Three-dimensional understanding of the morphological complexity of the human uterine endometrium. iScience. 2021;24:102258. doi: 10.1016/j.isci.2021.102258
- Vannuccini S, Tosti C, Carmona F, et al. Pathogenesis of adenomyosis: an update on molecular mechanisms. Reprod Biomed Online. 2017;35:592-601. doi: 10.1016 /j.rbmo.2017.06.016
- Zhai J, Vannuccini S, Petraglia F, et al. Adenomyosis: mechanisms and pathogenesis. Semin Reprod Med. 2020;38:129-143. doi: 10.1055/s-0040-1716687
- Munro MG. Uterine polyps, adenomyosis, leiomyomas, and endometrial receptivity. Fertil Steril. 2019;111:629-640. doi: 10.1016/j.fertnstert.2019.02.008
- Kay N, Huang C-Y, Shiu L-Y, et al. TGF-β1 neutralization improves pregnancy outcomes by restoring endometrial receptivity in mice with adenomyosis. Reprod Sci. 2021;28:877-887. doi: 10.1007/s43032-020-00308-1
- Habiba M, Benagiano G. Classifying adenomyosis: progress and challenges. Int J Environ Res Public Health. 2021;18:12386. doi: 10.3390/ijerph182312386
- Movilla P, Morris S, Isaacson K. A systematic review of tissue sampling techniques for the diagnosis of adenomyosis. J Minim Invasive Gynecol. 2020;27:344-351. doi: 10.1016 /j.jmig.2019.09.001
- Agostinho L, Cruz R, Osório F, et al. MRI for adenomyosis: a pictorial review. Insights Imaging. 2017;8:549-556. doi: 10.1007/s13244-017-0576-z
- Bazot M, Cortez A, Darai E, et al. Ultrasonography compared with magnetic resonance imaging for the diagnosis of adenomyosis: correlation with histopathology. Hum Reprod. 2001;16:2427-2433. doi: 10.1093/humrep/16.11.2427
- Reinhold C, Tafazoli F, Mehio A, et al. Uterine adenomyosis: endovaginal US and MR imaging features with histopathologic correlation. Radiographics. 1999;19:S147-S160. doi: 10.1148 /radiographics.19.suppl_1.g99oc13s147
- Rees CO, Nederend J, Mischi M, et al. Objective measures of adenomyosis on MRI and their diagnostic accuracy—a systematic review & meta-analysis. Acta Obstet Gynecol Scand. 2021;100:1377-1391.
- Chapron C, Vannuccini S, Santulli P, et al. Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum Reprod Update. 2020;26:392-411. doi: 10.1093 /humupd/dmz049
- Liu L, Li W, Leonardi M, et al. Diagnostic accuracy of transvaginal ultrasound and magnetic resonance imaging for adenomyosis: systematic review and meta-analysis and review of sonographic diagnostic criteria. J Ultrasound Med. 2021;40:2289-2306. doi: 10.1002/jum.15635
- Bazot M, Daraï E. Role of transvaginal sonography and magnetic resonance imaging in the diagnosis of uterine adenomyosis. Fertil Steril. 2018;109:389-397. doi: 10.1016 /j.fertnstert.2018.01.024
- Van den Bosch T, Dueholm M, Leone FPG, et al. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: a consensus opinion from the Morphological Uterus Sonographic Assessment (MUSA) group. Ultrasound Obstet Gynecol. 2015;46:284-298. doi: 10.1002/uog.14806
- Săsăran V, Turdean S, Gliga M, et al. Value of strainratio elastography in the diagnosis and differentiation of uterine fibroids and adenomyosis. J Pers Med. 2021;11:824. doi: 10.3390/jpm11080824
- Di Spiezio Sardo A, Calagna G, Santangelo F, et al. The role of hysteroscopy in the diagnosis and treatment of adenomyosis. Biomed Res Int. 2017;2017:2518396. doi: 10.1155/2017/2518396
- Azzi R. Adenomyosis: current perspectives. Obstet Gynecol Clin North Am. 1989;16:221-235.
- Parker JD, Leondires M, Sinaii N, et al. Persistence of dysmenorrhea and nonmenstrual pain after optimal endometriosis surgery may indicate adenomyosis. Fertil Steril. 2006;86:711-715. doi: 10.1016/j.fertnstert.2006.01.030
- Martire FG, Lazzeri L, Conway F, et al. Adolescence and endometriosis: symptoms, ultrasound signs and early diagnosis. Fertil Steril. 2020;114:1049-1057. doi: 10.1016 /j.fertnstert.2020.06.012
- Decter D, Arbib N, Markovitz H, et al. Sonographic signs of adenomyosis in women with endometriosis are associated with infertility. J Clin Med. 2021;10:2355. doi: 10.3390 /jcm10112355
- Brucker SY, Huebner M, Wallwiener M, et al. Clinical characteristics indicating adenomyosis coexisting with leiomyomas: a retrospective, questionnaire-based study. Fertil Steril. 2014;101:237-241.e1. doi: 10.1016 /j.fertnstert.2013.09.038
- Perelló MF, Martínez-Zamora MÁ, Torres X, et al. Endometriotic pain is associated with adenomyosis but not with the compartments affected by deep infiltrating endometriosis. Gynecol Obstet Invest. 2017;82:240-246. doi: 10.1159/000447633
- Younes G, Tulandi T. Effects of adenomyosis on in vitro fertilization treatment outcomes: a metaanalysis. Fertil Steril. 2017;108:483-490.e3. doi: 10.1016 /j.fertnstert.2017.06.025
- Nirgianakis K, Kalaitzopoulos DR, Schwartz ASK, et al. Fertility, pregnancy and neonatal outcomes of patients with adenomyosis: a systematic review and meta-analysis. Reprod BioMed Online. 2021;42:185-206. doi: 10.1016 /j.rbmo.2020.09.023
- Ono Y, Ota H, Takimoto K, et al. Perinatal outcomes associated with the positional relationship between the placenta and the adenomyosis lesion. J Gynecol Obstet Hum Reprod. 2021;50:102114. doi: 10.1016/j.jogoh.2021.102114
- Barrier BF, Malinowski MJ, Dick EJ Jr, et al. Adenomyosis in the baboon is associated with primary infertility. Fertil Steril. 2004;82(suppl 3):1091-1094. doi: 10.1016 /j.fertnstert.2003.11.065
- Vercellini P, Consonni D, Barbara G, et al. Adenomyosis and reproductive performance after surgery for rectovaginal and colorectal endometriosis: a systematic review and meta-analysis. Reprod Biomed Online. 2014;28:704-713. doi: 10.1016/j.rbmo.2014.02.006

CASE Painful, heavy menstruation and recurrent pregnancy loss
A 37-year-old woman (G3P0030) with a history of recurrent pregnancy loss presents for evaluation. She had 3 losses—most recently a miscarriage at 22 weeks with a cerclage in place. She did not undergo any surgical procedures for these losses. Hormonal and thrombophilia workup is negative and semen analysis is normal. She reports a history of painful, heavy periods for many years, as well as dyspareunia and occasional post-coital bleeding. Past medical history was otherwise unremarkable. Pelvic magnetic resonance imaging (MRI) revealed focal thickening of the junctional zone up to 15 mm with 2 foci of T2 hyperintensities suggesting adenomyosis (FIGURE 1).
How do you counsel this patient regarding the MRI findings and their impact on her fertility?

Adenomyosis is a condition in which endometrial glands and stroma are abnormally present in the uterine myometrium, resulting in smooth muscle hypertrophy and abnormal uterine contractility. Traditional teaching describes a woman in her 40s with heavy and painful menses, a “boggy uterus” on examination, who has completed childbearing and desires definitive treatment. Histologic diagnosis of adenomyosis is made from the uterine specimen at the time of hysterectomy, invariably confounding our understanding of the epidemiology of adenomyosis.
More recently, however, we are beginning to learn that this narrative is misguided. Imaging changes of adenomyosis can be seen in women who desire future fertility and in adolescents with severe dysmenorrhea, suggesting an earlier age of incidence.1 In a recent systematic review, prevalence estimates ranged from 15% to 67%, owing to varying diagnostic methods and patient inclusion criteria.2 It is increasingly being recognized as a primary contributor to infertility, with one study estimating a 30% prevalence of infertility in women with adenomyosis.3 Moreover, treatment with gonadotropin-releasing hormone agonists and/or surgical excision may improve fertility outcomes.4
As we learn more about this prevalent and life-altering condition, we owe it to our patients to consider this diagnosis when counseling on dysmenorrhea, heavy menstrual bleeding, or infertility.
Anatomy of the myometrium
The myometrium is composed of the inner and outer myometrium: the inner myometrium (IM) and endometrium are of Müllerian origin, and the outer myometrium (OM) is of mesenchymal origin. The IM thickens in response to steroid hormones during the menstrual cycle with metaplasia of endometrial stromal cells into myocytes and back again, whereas the OM is not responsive to hormones.5 Emerging literature suggests the OM is further divided into a middle and outer section based on different histologic morphologies, though the clinical implications of this are not understood.6 The term “junctional zone” (JZ) refers to the imaging appearance of what is thought to be the IM. Interestingly it cannot be identified on traditional hematoxylin and eosin staining. When the JZ is thickened or demonstrates irregular borders, it is used as a diagnostic marker for adenomyosis and is postulated to play an important role in adenomyosis pathophysiology, particularly heavy menstrual bleeding and infertility.7
Continue to: Subtypes of adenomyosis...
Subtypes of adenomyosis
While various disease classifications have been suggested for adenomyosis, to date there is no international consensus. Adenomyosis is typically described in 3 forms: diffuse, focal, or adenomyoma.8 As implied, the term focal adenomyosis refers to discrete lesions surrounded by normal myometrium, whereas abnormal glandular changes are pervasive throughout the myometrium in diffuse disease. Adenomyomas are a subgroup of focal adenomyosis that are thought to be surrounded by leiomyomatous smooth muscle and may be well demarcated on imaging.9
Recent research uses novel histologic imaging techniques to explore adenomyotic growth patterns in 3-dimensional (3D) reconstructions. Combining tissue-clearing methods with light-sheet fluorescence microscopy enables highly detailed 3D representations of the protein and nucleic acid structure of organs.10 For example, Yamaguchi and colleagues used this technology to explore the 3D morphological features of adenomyotic tissue and observed direct invasion of the endometrial glands into the myometrium and an “ant colony ̶ like network” of ectopic endometrial glands in the myometrium (FIGURE 2).11 These abnormal glandular networks have been visualized beyond the IM, which may not be captured on ultrasonography or MRI. While this work is still in its infancy, it has the potential to provide important insight into disease pathogenesis and to inform future therapy.

Pathogenesis
Proposed mechanisms for the development of adenomyosis include endometrial invasion, tissue injury and repair (TIAR) mechanisms, and the stem cell theory.12 According to the endometrial invasion theory, glandular epithelial cells from the basalis layer invaginate through an altered IM, slipping through weak muscle fibers and attracted by certain growth factors. In the TIAR mechanism theory, micro- or macro-trauma to the IM (whether from pregnancy, surgery, or infection) results in chronic proliferation and inflammation leading to the development of adenomyosis. Finally, the stem cell theory proposes that adenomyosis might develop from de novo ectopic endometrial tissue.
While the exact pathogenesis of adenomyosis is largely unknown, it has been associated with predictable molecular changes in the endometrium and surrounding myometrium.12 Myometrial hypercontractility is seen in patients with adenomyosis and dysmenorrhea, whereas neovascularization, high microvessel density, and abnormal uterine contractility are seen in those with abnormal uterine bleeding.13 In patients with infertility, increased inflammation, abnormal endometrial receptivity, and alterations in the myometrial architecture have been suggested to impair contractility and sperm transport.12,14
Differential growth factor expression and abnormal estrogen and progesterone signaling pathways have been observed in the IM in patients with adenomyosis, along with dysregulation of immune factors and increased inflammatory oxidative stress.12 This in turn results in myometrial hypertrophy and fibrosis, impairing normal uterine contractility patterns. This abnormal contractility may alter sperm transport and embryo implantation, and animal models that target pathways leading to fibrosis may improve endometrial receptivity.14,15 Further research is needed to elucidate specific molecular pathways and their complex interplay in this disease.
Continue to: Diagnosis...
Diagnosis
The gold standard for diagnosis of adenomyosis is histopathology from hysterectomy specimens, but specific definitions vary. Published criteria include endometrial glands within the myometrial layer greater than 0.5 to 1 low power field from the basal layer of the endometrium, endometrial glands extending deeper than 25% of the myometrial thickness, or endometrial glands a certain distance (ranging from 1-3 mm) from the basalis layer of the endometrium.16 Various methods of non-hysterectomy tissue sampling have been proposed for diagnosis, including needle, hysteroscopic, or laparoscopic sampling, but the sensitivity of these methods is poor.17 Limiting the diagnosis of adenomyosis to specimen pathology relies on invasive methods and clearly we cannot confirm the diagnosis by hysterectomy in patients with a desire for future fertility. It is for this reason that the prevalence of the disease is widely unknown.
The alternative to pathologic diagnosis is to identify radiologic changes that are associated with adenomyosis via either transvaginal ultrasound (TVUS) or MRI. Features suggestive of adenomyosis on MRI overlap with TVUS features, including uterine enlargement, anteroposterior myometrial asymmetry, T1- or T2-intense myometrial cysts or foci, and a thickened JZ.18 A JZ thicker than 12 mm has been thought to be predictive of adenomyosis, whereas a thickness of less than 8 mm is predictive of its absence, although the JZ may vary in thickness with the menstrual cycle.19,20 A 2021 systematic review and meta-analysis comparing MRI diagnosis with histopathologic findings reported a pooled sensitivity and specificity of 60% and 96%, respectively.21 The reported range for sensitivity and specificity is wide: 70% to 93% for sensitivity and 67% to 93% for specificity.22-24
Key TVUS features associated with adenomyosis were defined in 2015 in a consensus statement released by the Morphological Uterus Sonographic Assessment (MUSA) group.25 These include a globally enlarged uterus, anteroposterior myometrial asymmetry, myometrial cysts, fan-shaped shadowing, mixed myometrial echogenicity, translesional vascularity, echogenic subendometrial lines and buds, and a thickened, irregular or discontinuous JZ (FIGURES 3 and 4).25 The accuracy of ultrasonographic diagnosis of adenomyosis using these features has been investigated in multiple systematic reviews and meta-analyses, most recently by Liu and colleagues who found a pooled sensitivity of TVUS of 81% and pooled specificity of 87%.23 The range for ultrasonographic sensitivity and specificity is wide, however, ranging from 33% to 84% for sensitivity and 64% to 100% for specificity.22 Consensus is lacking as to which TVUS features are most predictive of adenomyosis, but in general, the combination of multiple MUSA criteria (particularly myometrial cysts and irregular JZ on 3D imaging) appears to be more accurate than any one feature alone.23 The presence of fibroids may decrease the sensitivity of TVUS, and one study suggested elastography may increase the accuracy of TVUS.24,26 Moreover, given that most radiologists receive limited training on the MUSA criteria, it behooves gynecologists to become familiar with these sonographic features to be able to identify adenomyosis in our patients.


Adenomyosis also may be suspected based on hysteroscopic findings, although a normal hysteroscopy cannot rule out the disease and data are lacking to support these markers as diagnostic. Visual findings can include a “strawberry” pattern, mucosal elevation, cystic hemorrhagic lesions, localized vascularity, or endometrial defects.27 Hysteroscopy may be effective in the treatment of localized lesions, although that discussion is beyond the scope of this review.
Clinical presentation
While many women who are later diagnosed with adenomyosis are asymptomatic, the disease can present with heavy menstrual bleeding and dysmenorrhea, which occur in 50% and 30% of patients, respectively.28 Other symptoms include dyspareunia and infertility. Symptoms were previously reported to develop between the ages of 40 and 50 years; however, this is biased by diagnosis at the time of hysterectomy and the fact that younger patients are less likely to undergo definitive surgery. When using imaging criteria for diagnosis, adenomyosis might be more responsible for dysmenorrhea and chronic pelvic pain in younger patients than previously appreciated.1,29 In a recent study reviewing TVUS in 270 adolescents for any reason, adenomyosis was present in 5% of cases and this increased up to 44% in the presence of endometriosis.30
Adenomyosis often co-exists and shares similar clinical presentations with other gynecologic pathologies such as endometriosis and fibroids, making diagnosis on symptomatology alone challenging. Concurrent adenomyosis has been found in up to 73% and 57% of patients with suspected or diagnosed endometriosis and fibroids, respectively.31,32 Accumulating evidence suggests that pelvic pain previously attributed to endometriosis may in fact be a result of adenomyosis; for example, persistent pelvic pain after optimal resection of endometriosis may be confounded by the presence of adenomyosis.29 In one study of 155 patients with complete resection of deep infiltrating endometriosis, persistent pelvic pain was significantly associated with the presence of adenomyosis on imaging.33
Adenomyosis is increasingly being recognized at the time of infertility evaluation with an estimated prevalence of 30% in women with infertility.3 Among women with infertility, adenomyosis has been associated with a lower clinical pregnancy rate, higher miscarriage rate, and lower live birth rate, as well as obstetric complications such as abnormal placentation.34-36 A study of 37 baboons found the histologic diagnosis of adenomyosis alone at necropsy was associated with a 20-fold increased risk of lifelong infertility (odds ratio [OR], 20.1; 95% CI, 2.1-921), whereas presence of endometriosis was associated with a nonsignificant 3-fold risk of lifelong infertility (OR, 3.6; 95% CI, 0.9-15.8).37
In women with endometriosis and infertility, co-existing adenomyosis portends worse fertility outcomes. In a retrospective study of 244 women who underwent endometriosis surgery, more than five features of adenomyosis on imaging was associated with higher rates of infertility, in vitro fertilization treatments, and a higher number of in vitro fertilization cycles.31 Moreover, in women who underwent surgery for deep infiltrating endometriosis, the presence of adenomyosis on imaging was associated with a 68% reduction in likelihood of pregnancy after surgery.38
Conclusion
As we begin to learn about adenomyosis, our misconceptions become more evident. The notion that it largely affects women at the end of their reproductive lives is biased by using histopathology at hysterectomy as the gold standard for diagnosis. Lack of definitive histologic or imaging criteria and biopsy techniques add to the diagnostic challenge. This in turn leads to inaccurate estimates of incidence and prevalence, as we assume patients’ symptoms must be attributable to what we can see at the time of surgery (for example, Stage I or II endometriosis), rather than what we cannot see. We now know that adenomyosis is present in women of all ages, including adolescents, and can significantly contribute to reduced fertility and quality of life. We owe it to our patients to consider this condition in the differential diagnosis of dysmenorrhea, heavy menstrual bleeding, dyspareunia, and infertility.
CASE Resolved
The patient underwent targeted hysteroscopic resection of adenomyosis (FIGURE 5) and conceived spontaneously the following year. ●


CASE Painful, heavy menstruation and recurrent pregnancy loss
A 37-year-old woman (G3P0030) with a history of recurrent pregnancy loss presents for evaluation. She had 3 losses—most recently a miscarriage at 22 weeks with a cerclage in place. She did not undergo any surgical procedures for these losses. Hormonal and thrombophilia workup is negative and semen analysis is normal. She reports a history of painful, heavy periods for many years, as well as dyspareunia and occasional post-coital bleeding. Past medical history was otherwise unremarkable. Pelvic magnetic resonance imaging (MRI) revealed focal thickening of the junctional zone up to 15 mm with 2 foci of T2 hyperintensities suggesting adenomyosis (FIGURE 1).
How do you counsel this patient regarding the MRI findings and their impact on her fertility?

Adenomyosis is a condition in which endometrial glands and stroma are abnormally present in the uterine myometrium, resulting in smooth muscle hypertrophy and abnormal uterine contractility. Traditional teaching describes a woman in her 40s with heavy and painful menses, a “boggy uterus” on examination, who has completed childbearing and desires definitive treatment. Histologic diagnosis of adenomyosis is made from the uterine specimen at the time of hysterectomy, invariably confounding our understanding of the epidemiology of adenomyosis.
More recently, however, we are beginning to learn that this narrative is misguided. Imaging changes of adenomyosis can be seen in women who desire future fertility and in adolescents with severe dysmenorrhea, suggesting an earlier age of incidence.1 In a recent systematic review, prevalence estimates ranged from 15% to 67%, owing to varying diagnostic methods and patient inclusion criteria.2 It is increasingly being recognized as a primary contributor to infertility, with one study estimating a 30% prevalence of infertility in women with adenomyosis.3 Moreover, treatment with gonadotropin-releasing hormone agonists and/or surgical excision may improve fertility outcomes.4
As we learn more about this prevalent and life-altering condition, we owe it to our patients to consider this diagnosis when counseling on dysmenorrhea, heavy menstrual bleeding, or infertility.
Anatomy of the myometrium
The myometrium is composed of the inner and outer myometrium: the inner myometrium (IM) and endometrium are of Müllerian origin, and the outer myometrium (OM) is of mesenchymal origin. The IM thickens in response to steroid hormones during the menstrual cycle with metaplasia of endometrial stromal cells into myocytes and back again, whereas the OM is not responsive to hormones.5 Emerging literature suggests the OM is further divided into a middle and outer section based on different histologic morphologies, though the clinical implications of this are not understood.6 The term “junctional zone” (JZ) refers to the imaging appearance of what is thought to be the IM. Interestingly it cannot be identified on traditional hematoxylin and eosin staining. When the JZ is thickened or demonstrates irregular borders, it is used as a diagnostic marker for adenomyosis and is postulated to play an important role in adenomyosis pathophysiology, particularly heavy menstrual bleeding and infertility.7
Continue to: Subtypes of adenomyosis...
Subtypes of adenomyosis
While various disease classifications have been suggested for adenomyosis, to date there is no international consensus. Adenomyosis is typically described in 3 forms: diffuse, focal, or adenomyoma.8 As implied, the term focal adenomyosis refers to discrete lesions surrounded by normal myometrium, whereas abnormal glandular changes are pervasive throughout the myometrium in diffuse disease. Adenomyomas are a subgroup of focal adenomyosis that are thought to be surrounded by leiomyomatous smooth muscle and may be well demarcated on imaging.9
Recent research uses novel histologic imaging techniques to explore adenomyotic growth patterns in 3-dimensional (3D) reconstructions. Combining tissue-clearing methods with light-sheet fluorescence microscopy enables highly detailed 3D representations of the protein and nucleic acid structure of organs.10 For example, Yamaguchi and colleagues used this technology to explore the 3D morphological features of adenomyotic tissue and observed direct invasion of the endometrial glands into the myometrium and an “ant colony ̶ like network” of ectopic endometrial glands in the myometrium (FIGURE 2).11 These abnormal glandular networks have been visualized beyond the IM, which may not be captured on ultrasonography or MRI. While this work is still in its infancy, it has the potential to provide important insight into disease pathogenesis and to inform future therapy.

Pathogenesis
Proposed mechanisms for the development of adenomyosis include endometrial invasion, tissue injury and repair (TIAR) mechanisms, and the stem cell theory.12 According to the endometrial invasion theory, glandular epithelial cells from the basalis layer invaginate through an altered IM, slipping through weak muscle fibers and attracted by certain growth factors. In the TIAR mechanism theory, micro- or macro-trauma to the IM (whether from pregnancy, surgery, or infection) results in chronic proliferation and inflammation leading to the development of adenomyosis. Finally, the stem cell theory proposes that adenomyosis might develop from de novo ectopic endometrial tissue.
While the exact pathogenesis of adenomyosis is largely unknown, it has been associated with predictable molecular changes in the endometrium and surrounding myometrium.12 Myometrial hypercontractility is seen in patients with adenomyosis and dysmenorrhea, whereas neovascularization, high microvessel density, and abnormal uterine contractility are seen in those with abnormal uterine bleeding.13 In patients with infertility, increased inflammation, abnormal endometrial receptivity, and alterations in the myometrial architecture have been suggested to impair contractility and sperm transport.12,14
Differential growth factor expression and abnormal estrogen and progesterone signaling pathways have been observed in the IM in patients with adenomyosis, along with dysregulation of immune factors and increased inflammatory oxidative stress.12 This in turn results in myometrial hypertrophy and fibrosis, impairing normal uterine contractility patterns. This abnormal contractility may alter sperm transport and embryo implantation, and animal models that target pathways leading to fibrosis may improve endometrial receptivity.14,15 Further research is needed to elucidate specific molecular pathways and their complex interplay in this disease.
Continue to: Diagnosis...
Diagnosis
The gold standard for diagnosis of adenomyosis is histopathology from hysterectomy specimens, but specific definitions vary. Published criteria include endometrial glands within the myometrial layer greater than 0.5 to 1 low power field from the basal layer of the endometrium, endometrial glands extending deeper than 25% of the myometrial thickness, or endometrial glands a certain distance (ranging from 1-3 mm) from the basalis layer of the endometrium.16 Various methods of non-hysterectomy tissue sampling have been proposed for diagnosis, including needle, hysteroscopic, or laparoscopic sampling, but the sensitivity of these methods is poor.17 Limiting the diagnosis of adenomyosis to specimen pathology relies on invasive methods and clearly we cannot confirm the diagnosis by hysterectomy in patients with a desire for future fertility. It is for this reason that the prevalence of the disease is widely unknown.
The alternative to pathologic diagnosis is to identify radiologic changes that are associated with adenomyosis via either transvaginal ultrasound (TVUS) or MRI. Features suggestive of adenomyosis on MRI overlap with TVUS features, including uterine enlargement, anteroposterior myometrial asymmetry, T1- or T2-intense myometrial cysts or foci, and a thickened JZ.18 A JZ thicker than 12 mm has been thought to be predictive of adenomyosis, whereas a thickness of less than 8 mm is predictive of its absence, although the JZ may vary in thickness with the menstrual cycle.19,20 A 2021 systematic review and meta-analysis comparing MRI diagnosis with histopathologic findings reported a pooled sensitivity and specificity of 60% and 96%, respectively.21 The reported range for sensitivity and specificity is wide: 70% to 93% for sensitivity and 67% to 93% for specificity.22-24
Key TVUS features associated with adenomyosis were defined in 2015 in a consensus statement released by the Morphological Uterus Sonographic Assessment (MUSA) group.25 These include a globally enlarged uterus, anteroposterior myometrial asymmetry, myometrial cysts, fan-shaped shadowing, mixed myometrial echogenicity, translesional vascularity, echogenic subendometrial lines and buds, and a thickened, irregular or discontinuous JZ (FIGURES 3 and 4).25 The accuracy of ultrasonographic diagnosis of adenomyosis using these features has been investigated in multiple systematic reviews and meta-analyses, most recently by Liu and colleagues who found a pooled sensitivity of TVUS of 81% and pooled specificity of 87%.23 The range for ultrasonographic sensitivity and specificity is wide, however, ranging from 33% to 84% for sensitivity and 64% to 100% for specificity.22 Consensus is lacking as to which TVUS features are most predictive of adenomyosis, but in general, the combination of multiple MUSA criteria (particularly myometrial cysts and irregular JZ on 3D imaging) appears to be more accurate than any one feature alone.23 The presence of fibroids may decrease the sensitivity of TVUS, and one study suggested elastography may increase the accuracy of TVUS.24,26 Moreover, given that most radiologists receive limited training on the MUSA criteria, it behooves gynecologists to become familiar with these sonographic features to be able to identify adenomyosis in our patients.


Adenomyosis also may be suspected based on hysteroscopic findings, although a normal hysteroscopy cannot rule out the disease and data are lacking to support these markers as diagnostic. Visual findings can include a “strawberry” pattern, mucosal elevation, cystic hemorrhagic lesions, localized vascularity, or endometrial defects.27 Hysteroscopy may be effective in the treatment of localized lesions, although that discussion is beyond the scope of this review.
Clinical presentation
While many women who are later diagnosed with adenomyosis are asymptomatic, the disease can present with heavy menstrual bleeding and dysmenorrhea, which occur in 50% and 30% of patients, respectively.28 Other symptoms include dyspareunia and infertility. Symptoms were previously reported to develop between the ages of 40 and 50 years; however, this is biased by diagnosis at the time of hysterectomy and the fact that younger patients are less likely to undergo definitive surgery. When using imaging criteria for diagnosis, adenomyosis might be more responsible for dysmenorrhea and chronic pelvic pain in younger patients than previously appreciated.1,29 In a recent study reviewing TVUS in 270 adolescents for any reason, adenomyosis was present in 5% of cases and this increased up to 44% in the presence of endometriosis.30
Adenomyosis often co-exists and shares similar clinical presentations with other gynecologic pathologies such as endometriosis and fibroids, making diagnosis on symptomatology alone challenging. Concurrent adenomyosis has been found in up to 73% and 57% of patients with suspected or diagnosed endometriosis and fibroids, respectively.31,32 Accumulating evidence suggests that pelvic pain previously attributed to endometriosis may in fact be a result of adenomyosis; for example, persistent pelvic pain after optimal resection of endometriosis may be confounded by the presence of adenomyosis.29 In one study of 155 patients with complete resection of deep infiltrating endometriosis, persistent pelvic pain was significantly associated with the presence of adenomyosis on imaging.33
Adenomyosis is increasingly being recognized at the time of infertility evaluation with an estimated prevalence of 30% in women with infertility.3 Among women with infertility, adenomyosis has been associated with a lower clinical pregnancy rate, higher miscarriage rate, and lower live birth rate, as well as obstetric complications such as abnormal placentation.34-36 A study of 37 baboons found the histologic diagnosis of adenomyosis alone at necropsy was associated with a 20-fold increased risk of lifelong infertility (odds ratio [OR], 20.1; 95% CI, 2.1-921), whereas presence of endometriosis was associated with a nonsignificant 3-fold risk of lifelong infertility (OR, 3.6; 95% CI, 0.9-15.8).37
In women with endometriosis and infertility, co-existing adenomyosis portends worse fertility outcomes. In a retrospective study of 244 women who underwent endometriosis surgery, more than five features of adenomyosis on imaging was associated with higher rates of infertility, in vitro fertilization treatments, and a higher number of in vitro fertilization cycles.31 Moreover, in women who underwent surgery for deep infiltrating endometriosis, the presence of adenomyosis on imaging was associated with a 68% reduction in likelihood of pregnancy after surgery.38
Conclusion
As we begin to learn about adenomyosis, our misconceptions become more evident. The notion that it largely affects women at the end of their reproductive lives is biased by using histopathology at hysterectomy as the gold standard for diagnosis. Lack of definitive histologic or imaging criteria and biopsy techniques add to the diagnostic challenge. This in turn leads to inaccurate estimates of incidence and prevalence, as we assume patients’ symptoms must be attributable to what we can see at the time of surgery (for example, Stage I or II endometriosis), rather than what we cannot see. We now know that adenomyosis is present in women of all ages, including adolescents, and can significantly contribute to reduced fertility and quality of life. We owe it to our patients to consider this condition in the differential diagnosis of dysmenorrhea, heavy menstrual bleeding, dyspareunia, and infertility.
CASE Resolved
The patient underwent targeted hysteroscopic resection of adenomyosis (FIGURE 5) and conceived spontaneously the following year. ●

- Exacoustos C, Lazzeri L, Martire FG, et al. Ultrasound findings of adenomyosis in adolescents: type and grade of the disease. J Minim Invasive Gynecol. 2021;29:291.e1-299.e1. doi: 10.1016/j.jmig.2021.08.023
- Loring M, Chen TY, Isaacson KB. A systematic review of adenomyosis: it is time to reassess what we thought we knew about the disease. J Minim Invasive Gynecol. 2021;28:644655. doi: 10.1016/j.jmig.2020.10.012
- Bourdon M, Santulli P, Oliveira J, et al. Focal adenomyosis is associated with primary infertility. Fertil Steril. 2020;114:1271-1277. doi: 10.1016/j.fertnstert.2020.06.018
- Lan J, Wu Y, Wu Z, et al. Ultra-long GnRH agonist protocol during IVF/ICSI improves pregnancy outcomes in women with adenomyosis: a retrospective cohort study. Front Endocrinol (Lausanne). 2021;12:609771. doi: 10.3389 /fendo.2021.609771
- Gnecco JS, Brown AT, Kan EL, et al. Physiomimetic models of adenomyosis. Semin Reprod Med. 2020;38:179-196. doi: 10.1055/s-0040-1719084
- Harmsen MJ, Trommelen LM, de Leeuw RA, et al. Uterine junctional zone and adenomyosis: comparison of MRI, transvaginal ultrasound and histology. Ultrasound Obstet Gynecol. 2023;62:42-60. doi: 10.1002/uog.26117
- Xie T, Xu X, Yang Y, et al. The role of abnormal uterine junction zone in the occurrence and development of adenomyosis. Reprod Sci. 2022;29:2719-2730. doi: 10.1007/s43032-021 -00684-2
- Lazzeri L, Morosetti G, Centini G, et al. A sonographic classification of adenomyosis: interobserver reproducibility in the evaluation of type and degree of the myometrial involvement. Fertil Steril. 2018;110:1154-1161.e3. doi: 10.1016 /j.fertnstert.2018.06.031
- Tahlan A, Nanda A, Mohan H. Uterine adenomyoma: a clinicopathologic review of 26 cases and a review of the literature. Int J Gynecol Pathol. 2006;25:361-365. doi: 10.1097/01.pgp.0000209570.08716.b3
- Chung K, Wallace J, Kim S-Y, et al. Structural and molecular interrogation of intact biological systems. Nature. 2013;497:332-337. doi: 10.1038/nature12107
- Yamaguchi M, Yoshihara K, Suda K, et al. Three-dimensional understanding of the morphological complexity of the human uterine endometrium. iScience. 2021;24:102258. doi: 10.1016/j.isci.2021.102258
- Vannuccini S, Tosti C, Carmona F, et al. Pathogenesis of adenomyosis: an update on molecular mechanisms. Reprod Biomed Online. 2017;35:592-601. doi: 10.1016 /j.rbmo.2017.06.016
- Zhai J, Vannuccini S, Petraglia F, et al. Adenomyosis: mechanisms and pathogenesis. Semin Reprod Med. 2020;38:129-143. doi: 10.1055/s-0040-1716687
- Munro MG. Uterine polyps, adenomyosis, leiomyomas, and endometrial receptivity. Fertil Steril. 2019;111:629-640. doi: 10.1016/j.fertnstert.2019.02.008
- Kay N, Huang C-Y, Shiu L-Y, et al. TGF-β1 neutralization improves pregnancy outcomes by restoring endometrial receptivity in mice with adenomyosis. Reprod Sci. 2021;28:877-887. doi: 10.1007/s43032-020-00308-1
- Habiba M, Benagiano G. Classifying adenomyosis: progress and challenges. Int J Environ Res Public Health. 2021;18:12386. doi: 10.3390/ijerph182312386
- Movilla P, Morris S, Isaacson K. A systematic review of tissue sampling techniques for the diagnosis of adenomyosis. J Minim Invasive Gynecol. 2020;27:344-351. doi: 10.1016 /j.jmig.2019.09.001
- Agostinho L, Cruz R, Osório F, et al. MRI for adenomyosis: a pictorial review. Insights Imaging. 2017;8:549-556. doi: 10.1007/s13244-017-0576-z
- Bazot M, Cortez A, Darai E, et al. Ultrasonography compared with magnetic resonance imaging for the diagnosis of adenomyosis: correlation with histopathology. Hum Reprod. 2001;16:2427-2433. doi: 10.1093/humrep/16.11.2427
- Reinhold C, Tafazoli F, Mehio A, et al. Uterine adenomyosis: endovaginal US and MR imaging features with histopathologic correlation. Radiographics. 1999;19:S147-S160. doi: 10.1148 /radiographics.19.suppl_1.g99oc13s147
- Rees CO, Nederend J, Mischi M, et al. Objective measures of adenomyosis on MRI and their diagnostic accuracy—a systematic review & meta-analysis. Acta Obstet Gynecol Scand. 2021;100:1377-1391.
- Chapron C, Vannuccini S, Santulli P, et al. Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum Reprod Update. 2020;26:392-411. doi: 10.1093 /humupd/dmz049
- Liu L, Li W, Leonardi M, et al. Diagnostic accuracy of transvaginal ultrasound and magnetic resonance imaging for adenomyosis: systematic review and meta-analysis and review of sonographic diagnostic criteria. J Ultrasound Med. 2021;40:2289-2306. doi: 10.1002/jum.15635
- Bazot M, Daraï E. Role of transvaginal sonography and magnetic resonance imaging in the diagnosis of uterine adenomyosis. Fertil Steril. 2018;109:389-397. doi: 10.1016 /j.fertnstert.2018.01.024
- Van den Bosch T, Dueholm M, Leone FPG, et al. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: a consensus opinion from the Morphological Uterus Sonographic Assessment (MUSA) group. Ultrasound Obstet Gynecol. 2015;46:284-298. doi: 10.1002/uog.14806
- Săsăran V, Turdean S, Gliga M, et al. Value of strainratio elastography in the diagnosis and differentiation of uterine fibroids and adenomyosis. J Pers Med. 2021;11:824. doi: 10.3390/jpm11080824
- Di Spiezio Sardo A, Calagna G, Santangelo F, et al. The role of hysteroscopy in the diagnosis and treatment of adenomyosis. Biomed Res Int. 2017;2017:2518396. doi: 10.1155/2017/2518396
- Azzi R. Adenomyosis: current perspectives. Obstet Gynecol Clin North Am. 1989;16:221-235.
- Parker JD, Leondires M, Sinaii N, et al. Persistence of dysmenorrhea and nonmenstrual pain after optimal endometriosis surgery may indicate adenomyosis. Fertil Steril. 2006;86:711-715. doi: 10.1016/j.fertnstert.2006.01.030
- Martire FG, Lazzeri L, Conway F, et al. Adolescence and endometriosis: symptoms, ultrasound signs and early diagnosis. Fertil Steril. 2020;114:1049-1057. doi: 10.1016 /j.fertnstert.2020.06.012
- Decter D, Arbib N, Markovitz H, et al. Sonographic signs of adenomyosis in women with endometriosis are associated with infertility. J Clin Med. 2021;10:2355. doi: 10.3390 /jcm10112355
- Brucker SY, Huebner M, Wallwiener M, et al. Clinical characteristics indicating adenomyosis coexisting with leiomyomas: a retrospective, questionnaire-based study. Fertil Steril. 2014;101:237-241.e1. doi: 10.1016 /j.fertnstert.2013.09.038
- Perelló MF, Martínez-Zamora MÁ, Torres X, et al. Endometriotic pain is associated with adenomyosis but not with the compartments affected by deep infiltrating endometriosis. Gynecol Obstet Invest. 2017;82:240-246. doi: 10.1159/000447633
- Younes G, Tulandi T. Effects of adenomyosis on in vitro fertilization treatment outcomes: a metaanalysis. Fertil Steril. 2017;108:483-490.e3. doi: 10.1016 /j.fertnstert.2017.06.025
- Nirgianakis K, Kalaitzopoulos DR, Schwartz ASK, et al. Fertility, pregnancy and neonatal outcomes of patients with adenomyosis: a systematic review and meta-analysis. Reprod BioMed Online. 2021;42:185-206. doi: 10.1016 /j.rbmo.2020.09.023
- Ono Y, Ota H, Takimoto K, et al. Perinatal outcomes associated with the positional relationship between the placenta and the adenomyosis lesion. J Gynecol Obstet Hum Reprod. 2021;50:102114. doi: 10.1016/j.jogoh.2021.102114
- Barrier BF, Malinowski MJ, Dick EJ Jr, et al. Adenomyosis in the baboon is associated with primary infertility. Fertil Steril. 2004;82(suppl 3):1091-1094. doi: 10.1016 /j.fertnstert.2003.11.065
- Vercellini P, Consonni D, Barbara G, et al. Adenomyosis and reproductive performance after surgery for rectovaginal and colorectal endometriosis: a systematic review and meta-analysis. Reprod Biomed Online. 2014;28:704-713. doi: 10.1016/j.rbmo.2014.02.006
- Exacoustos C, Lazzeri L, Martire FG, et al. Ultrasound findings of adenomyosis in adolescents: type and grade of the disease. J Minim Invasive Gynecol. 2021;29:291.e1-299.e1. doi: 10.1016/j.jmig.2021.08.023
- Loring M, Chen TY, Isaacson KB. A systematic review of adenomyosis: it is time to reassess what we thought we knew about the disease. J Minim Invasive Gynecol. 2021;28:644655. doi: 10.1016/j.jmig.2020.10.012
- Bourdon M, Santulli P, Oliveira J, et al. Focal adenomyosis is associated with primary infertility. Fertil Steril. 2020;114:1271-1277. doi: 10.1016/j.fertnstert.2020.06.018
- Lan J, Wu Y, Wu Z, et al. Ultra-long GnRH agonist protocol during IVF/ICSI improves pregnancy outcomes in women with adenomyosis: a retrospective cohort study. Front Endocrinol (Lausanne). 2021;12:609771. doi: 10.3389 /fendo.2021.609771
- Gnecco JS, Brown AT, Kan EL, et al. Physiomimetic models of adenomyosis. Semin Reprod Med. 2020;38:179-196. doi: 10.1055/s-0040-1719084
- Harmsen MJ, Trommelen LM, de Leeuw RA, et al. Uterine junctional zone and adenomyosis: comparison of MRI, transvaginal ultrasound and histology. Ultrasound Obstet Gynecol. 2023;62:42-60. doi: 10.1002/uog.26117
- Xie T, Xu X, Yang Y, et al. The role of abnormal uterine junction zone in the occurrence and development of adenomyosis. Reprod Sci. 2022;29:2719-2730. doi: 10.1007/s43032-021 -00684-2
- Lazzeri L, Morosetti G, Centini G, et al. A sonographic classification of adenomyosis: interobserver reproducibility in the evaluation of type and degree of the myometrial involvement. Fertil Steril. 2018;110:1154-1161.e3. doi: 10.1016 /j.fertnstert.2018.06.031
- Tahlan A, Nanda A, Mohan H. Uterine adenomyoma: a clinicopathologic review of 26 cases and a review of the literature. Int J Gynecol Pathol. 2006;25:361-365. doi: 10.1097/01.pgp.0000209570.08716.b3
- Chung K, Wallace J, Kim S-Y, et al. Structural and molecular interrogation of intact biological systems. Nature. 2013;497:332-337. doi: 10.1038/nature12107
- Yamaguchi M, Yoshihara K, Suda K, et al. Three-dimensional understanding of the morphological complexity of the human uterine endometrium. iScience. 2021;24:102258. doi: 10.1016/j.isci.2021.102258
- Vannuccini S, Tosti C, Carmona F, et al. Pathogenesis of adenomyosis: an update on molecular mechanisms. Reprod Biomed Online. 2017;35:592-601. doi: 10.1016 /j.rbmo.2017.06.016
- Zhai J, Vannuccini S, Petraglia F, et al. Adenomyosis: mechanisms and pathogenesis. Semin Reprod Med. 2020;38:129-143. doi: 10.1055/s-0040-1716687
- Munro MG. Uterine polyps, adenomyosis, leiomyomas, and endometrial receptivity. Fertil Steril. 2019;111:629-640. doi: 10.1016/j.fertnstert.2019.02.008
- Kay N, Huang C-Y, Shiu L-Y, et al. TGF-β1 neutralization improves pregnancy outcomes by restoring endometrial receptivity in mice with adenomyosis. Reprod Sci. 2021;28:877-887. doi: 10.1007/s43032-020-00308-1
- Habiba M, Benagiano G. Classifying adenomyosis: progress and challenges. Int J Environ Res Public Health. 2021;18:12386. doi: 10.3390/ijerph182312386
- Movilla P, Morris S, Isaacson K. A systematic review of tissue sampling techniques for the diagnosis of adenomyosis. J Minim Invasive Gynecol. 2020;27:344-351. doi: 10.1016 /j.jmig.2019.09.001
- Agostinho L, Cruz R, Osório F, et al. MRI for adenomyosis: a pictorial review. Insights Imaging. 2017;8:549-556. doi: 10.1007/s13244-017-0576-z
- Bazot M, Cortez A, Darai E, et al. Ultrasonography compared with magnetic resonance imaging for the diagnosis of adenomyosis: correlation with histopathology. Hum Reprod. 2001;16:2427-2433. doi: 10.1093/humrep/16.11.2427
- Reinhold C, Tafazoli F, Mehio A, et al. Uterine adenomyosis: endovaginal US and MR imaging features with histopathologic correlation. Radiographics. 1999;19:S147-S160. doi: 10.1148 /radiographics.19.suppl_1.g99oc13s147
- Rees CO, Nederend J, Mischi M, et al. Objective measures of adenomyosis on MRI and their diagnostic accuracy—a systematic review & meta-analysis. Acta Obstet Gynecol Scand. 2021;100:1377-1391.
- Chapron C, Vannuccini S, Santulli P, et al. Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum Reprod Update. 2020;26:392-411. doi: 10.1093 /humupd/dmz049
- Liu L, Li W, Leonardi M, et al. Diagnostic accuracy of transvaginal ultrasound and magnetic resonance imaging for adenomyosis: systematic review and meta-analysis and review of sonographic diagnostic criteria. J Ultrasound Med. 2021;40:2289-2306. doi: 10.1002/jum.15635
- Bazot M, Daraï E. Role of transvaginal sonography and magnetic resonance imaging in the diagnosis of uterine adenomyosis. Fertil Steril. 2018;109:389-397. doi: 10.1016 /j.fertnstert.2018.01.024
- Van den Bosch T, Dueholm M, Leone FPG, et al. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: a consensus opinion from the Morphological Uterus Sonographic Assessment (MUSA) group. Ultrasound Obstet Gynecol. 2015;46:284-298. doi: 10.1002/uog.14806
- Săsăran V, Turdean S, Gliga M, et al. Value of strainratio elastography in the diagnosis and differentiation of uterine fibroids and adenomyosis. J Pers Med. 2021;11:824. doi: 10.3390/jpm11080824
- Di Spiezio Sardo A, Calagna G, Santangelo F, et al. The role of hysteroscopy in the diagnosis and treatment of adenomyosis. Biomed Res Int. 2017;2017:2518396. doi: 10.1155/2017/2518396
- Azzi R. Adenomyosis: current perspectives. Obstet Gynecol Clin North Am. 1989;16:221-235.
- Parker JD, Leondires M, Sinaii N, et al. Persistence of dysmenorrhea and nonmenstrual pain after optimal endometriosis surgery may indicate adenomyosis. Fertil Steril. 2006;86:711-715. doi: 10.1016/j.fertnstert.2006.01.030
- Martire FG, Lazzeri L, Conway F, et al. Adolescence and endometriosis: symptoms, ultrasound signs and early diagnosis. Fertil Steril. 2020;114:1049-1057. doi: 10.1016 /j.fertnstert.2020.06.012
- Decter D, Arbib N, Markovitz H, et al. Sonographic signs of adenomyosis in women with endometriosis are associated with infertility. J Clin Med. 2021;10:2355. doi: 10.3390 /jcm10112355
- Brucker SY, Huebner M, Wallwiener M, et al. Clinical characteristics indicating adenomyosis coexisting with leiomyomas: a retrospective, questionnaire-based study. Fertil Steril. 2014;101:237-241.e1. doi: 10.1016 /j.fertnstert.2013.09.038
- Perelló MF, Martínez-Zamora MÁ, Torres X, et al. Endometriotic pain is associated with adenomyosis but not with the compartments affected by deep infiltrating endometriosis. Gynecol Obstet Invest. 2017;82:240-246. doi: 10.1159/000447633
- Younes G, Tulandi T. Effects of adenomyosis on in vitro fertilization treatment outcomes: a metaanalysis. Fertil Steril. 2017;108:483-490.e3. doi: 10.1016 /j.fertnstert.2017.06.025
- Nirgianakis K, Kalaitzopoulos DR, Schwartz ASK, et al. Fertility, pregnancy and neonatal outcomes of patients with adenomyosis: a systematic review and meta-analysis. Reprod BioMed Online. 2021;42:185-206. doi: 10.1016 /j.rbmo.2020.09.023
- Ono Y, Ota H, Takimoto K, et al. Perinatal outcomes associated with the positional relationship between the placenta and the adenomyosis lesion. J Gynecol Obstet Hum Reprod. 2021;50:102114. doi: 10.1016/j.jogoh.2021.102114
- Barrier BF, Malinowski MJ, Dick EJ Jr, et al. Adenomyosis in the baboon is associated with primary infertility. Fertil Steril. 2004;82(suppl 3):1091-1094. doi: 10.1016 /j.fertnstert.2003.11.065
- Vercellini P, Consonni D, Barbara G, et al. Adenomyosis and reproductive performance after surgery for rectovaginal and colorectal endometriosis: a systematic review and meta-analysis. Reprod Biomed Online. 2014;28:704-713. doi: 10.1016/j.rbmo.2014.02.006
Managing intrahepatic cholestasis of pregnancy
CASE Pregnant woman with intense itching
A 28-year-old woman (G1P0) is seen for a routine prenatal visit at 32 3/7 weeks’ gestation. She reports having generalized intense itching, including on her palms and soles, that is most intense at night and has been present for approximately 1 week. Her pregnancy is otherwise uncomplicated to date. Physical exam is within normal limits, with no evidence of a skin rash. Cholestasis of pregnancy is suspected, and laboratory tests are ordered, including bile acids and liver transaminases. Test results show that her aspartate transaminase (AST) and alanine transaminase (ALT) levels are mildly elevated at 55 IU/L and 41 IU/L, respectively, and several days later her bile acid level result is 21 µmol/L.
How should this patient be managed?
Intrahepatic cholestasis of pregnancy (ICP) affects 0.5% to 0.7% of pregnant individuals and results in maternal pruritus and elevated serum bile acid levels.1-3 Pruritus in ICP typically is generalized, including occurrence on the palms of the hands and soles of the feet, and it often is reported to be worse at night.4 Up to 25% of pregnant women will develop pruritus during pregnancy but the majority will not have ICP.2,5 Patients with ICP have no associated rash, but clinicians may note excoriations on exam. ICP typically presents in the third trimester of pregnancy but has been reported to occur earlier in gestation.6
Making a diagnosis of ICP
The presence of maternal pruritus in the absence of a skin condition along with elevated levels of serum bile acids are required for the diagnosis of ICP.7 Thus, a thorough history and physical exam is recommended to rule out another skin condition that could potentially explain the patient’s pruritus.
Some controversy exists regarding the bile acid level cutoff that should be used to make a diagnosis of ICP.8 It has been noted that nonfasting serum bile acid levels in pregnancy are considerably higher than those in in the nonpregnant state, and an upper limit of 18 µmol/L has been proposed as a cutoff in pregnancy.9 However, nonfasting total serum bile acids also have been shown to vary considerably by race, with levels 25.8% higher in Black women compared with those in White women and 24.3% higher in Black women compared with those in south Asian women.9 This raises the question of whether we should be using race-specific bile acid values to make a diagnosis of ICP.
Bile acid levels also vary based on whether a patient is in a fasting or postprandial state.10 Despite this variation, most guidelines do not recommend testing fasting bile acid levels as the postprandial state effect overall is small.7,9,11 The Society for Maternal-Fetal Medicine (SMFM) recommends that if a pregnancy-specific bile acid range is available from the laboratory, then the upper limit of normal for pregnancy should be used when making a diagnosis of ICP.7 The SMFM guidelines also acknowledge, however, that pregnancy-specific values rarely are available, and in this case, levels above the upper limit of normal—often 10 µmol/L should be considered diagnostic for ICP until further evidence regarding optimal bile acid cutoff levels in pregnancy becomes available.7
For patients with suspected ICP, liver transaminase levels should be measured in addition to nonfasting serum bile acid levels.7 A thorough history should include assessment for additional symptoms of liver disease, such as changes in weight, appetite, jaundice, excessive fatigue, malaise, and abdominal pain.7 Elevated transaminases levels may be associated with ICP, but they are not necessary for diagnosis. In the absence of additional clinical symptoms that suggest underlying liver disease or severe early onset ICP, additional evaluation beyond nonfasting serum bile acids and liver transaminase levels, such as liver ultrasonography or evaluation for viral or autoimmune hepatitis, is not recommended.7 Obstetric care clinicians should be aware that there is an increased incidence of preeclampsia among patients with ICP, although no specific guidance regarding further recommendations for screening is provided.7

Continue to: Risks associated with ICP...
Risks associated with ICP
For both patients and clinicians, the greatest concern among patients with ICP is the increased risk of stillbirth. Stillbirth risk in ICP appears to be related to serum bile acid levels and has been reported to be highest in patients with bile acid levels greater than 100 µmol/L. A systematic review and meta-analysis of ICP studies demonstrated no increased risk of stillbirth among patients with bile acid levels less than 100 µmol/L.12 These results, however, must be interpreted with extreme caution as the majority of studies included patients with ICP who were actively managed with attempts to mitigate the risk of stillbirth.7
In the absence of additional pregnancy risk factors, the risk of stillbirth among patients with ICP and serum bile acid levels between 19 and 39 µmol/L does not appear to be elevated above their baseline risk.11 The same is true for pregnant individuals with ICP and no additional pregnancy risk factors with serum bile acid levels between 40 and 99 µmol/L until approximately 38 weeks’ gestation, when the risk of stillbirth is elevated.11 The risk of stillbirth is elevated in ICP with peak bile acid levels greater than 100 µmol/L, with an absolute risk of 3.44%.11
Management of patients with ICP
Laboratory evaluation
There is no consensus on the need for repeat testing of bile acid levels in patients with ICP. SMFM advises that follow-up testing of bile acid levels may help to guide delivery timing, especially in cases of severe ICP, but the society recommends against serial testing.7 By contrast, the Royal College of Obstetricians and Gynaecologists (RCOG) provides a detailed algorithm regarding time intervals between serum bile acid level testing to guide delivery timing.11 The TABLE lists the strategy for reassessment of serum bile acid levels in ICP as recommended by the RCOG.11

In the United States, bile acid testing traditionally takes several days as the testing is commonly performed at reference laboratories. We therefore suggest that clinicians consider repeating bile acid level testing in situations in which the timing of delivery may be altered if further elevations of bile acid levels were noted. This is particularly relevant for patients diagnosed with ICP early in the third trimester when repeat bile acid levels would still allow for an adjustment in delivery timing.
Antepartum fetal surveillance
Unfortunately, antepartum fetal testing for pregnant patients with ICP does not appear to reliably predict or prevent stillbirth as several studies have reported stillbirths within days of normal fetal testing.13-16 It is therefore important to counsel pregnant patients regarding monitoring of fetal movements and advise them to present for evaluation if concerns arise.
Currently, SMFM recommends that patients with ICP should begin antenatal fetal surveillance at a gestational age when abnormal fetal testing would result in delivery.7 Patients should be counseled, however, regarding the unpredictability of stillbirth with ICP in the setting of a low absolute risk of such.
Medications
While SMFM recommends a starting dose of ursodeoxycholic acid 10 to 15 mg/kg per day divided into 2 or 3 daily doses as first-line therapy for the treatment of maternal symptoms of ICP, it is important to acknowledge that the goal of treatment is to alleviate maternal symptoms as there is no evidence that ursodeoxycholic acid improves either maternal serum bile acid levels or perinatal outcomes.7,17,18 Since publication of the guidelines, ursodeoxycholic acid’s lack of benefit has been further confirmed in a meta-analysis, and thus discontinuation is not unreasonable in the absence of any improvement in maternal symptoms.18
Timing of delivery
The optimal management of ICP remains unknown. SMFM recommends delivery based on peak serum bile acid levels. Delivery is recommended at 36 weeks’ gestation with ICP and total bile acid levels greater than 100 µmol/L as these patients have the greatest risk of stillbirth.7 For patients with ICP and bile acid levels less than 100 µmol/L, delivery is recommended between 36 0/7 and 39 0/7 weeks’ gestation.7 This is a wide gestational age window for clinicians to consider timing of delivery, and certainly the risks of stillbirth should be carefully balanced with the morbidity associated with a preterm or early term delivery.
For patients with ICP who have bile acid levels greater than 40 µmol/L, it is reasonable to consider delivery earlier in the gestational age window, given an evidence of increased risk of stillbirth after 38 weeks.7,12 For patients with ICP who have bile acid levels less than 40 µmol/L, delivery closer to 39 weeks’ gestation is recommended, as the risk of stillbirth does not appear to be increased above the baseline risk.7,12 Clinicians should be aware that the presence of concomitant morbidities, such as preeclampsia and gestational diabetes, are associated with an increased risk of stillbirth and should be considered for delivery planning.19
Postpartum follow-up
Routine laboratory evaluation following delivery is not recommended.7 However, in the presence of persistent pruritus or other signs and symptoms of hepatobiliary disease, liver function tests should be repeated with referral to hepatology if results are persistently abnormal 4 to 6 weeks postpartum.7
CASE Patient follow-up and outcomes
- Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37.
- Kenyon AP, Tribe RM, Nelson-Piercy C, et al. Pruritus in pregnancy: a study of anatomical distribution and prevalence in relation to the development of obstetric cholestasis. Obstet Med. 2010;3:25-29.
- Wikstrom Shemer E, Marschall HU, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated adverse pregnancy and fetal outcomes: a 12-year population-based cohort study. BJOG. 2013;120:717-723.
- Ambros-Rudolph CM, Glatz M, Trauner M, et al. The importance of serum bile acid level analysis and treatment with ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a case series from central Europe. Arch Dermatol. 2007;143:757-762.
- Szczech J, Wiatrowski A, Hirnle L, et al. Prevalence and relevance of pruritus in pregnancy. Biomed Res Int. 2017;2017:4238139.
- Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049-2066.
- Society for Maternal-Fetal Medicine; Lee RH, Greenberg M, Metz TD, et al. Society for Maternal-Fetal Medicine Consult Series #53: intrahepatic cholestasis of pregnancy: replaces Consult #13, April 2011. Am J Obstet Gynecol. 2021;224:B2-B9.
- Horgan R, Bitas C, Abuhamad A. Intrahepatic cholestasis of pregnancy: a comparison of Society for Maternal-Fetal Medicine and the Royal College of Obstetricians and Gynaecologists’ guidelines. Am J Obstet Gynecol MFM. 2023;5:100838.
- Mitchell AL, Ovadia C, Syngelaki A, et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: case-control and cohort study. BJOG. 2021;128:1635-1644.
- Adams A, Jacobs K, Vogel RI, et al. Bile acid determination after standardized glucose load in pregnant women. AJP Rep. 2015;5:e168-e171.
- Girling J, Knight CL, Chappell L; Royal College of Obstetricians and Gynaecologists. Intrahepatic cholestasis of pregnancy: Green-top guideline no. 43, June 2022. BJOG. 2022;129:e95-e114.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393:899-909.
- Alsulyman OM, Ouzounian JG, Ames-Castro M, et al. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960.
- Herrera CA, Manuck TA, Stoddard GJ, et al. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2018;31:1913-1920.
- Lee RH, Incerpi MH, Miller DA, et al. Sudden fetal death in intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2009;113:528-531.
- Sentilhes L, Verspyck E, Pia P, et al. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460.
- Chappell LC, Bell JL, Smith A, et al; PITCHES Study Group. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial. Lancet. 2019;394:849-860.
- Ovadia C, Sajous J, Seed PT, et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a systematic review and individual participant data meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:547-558.
- Geenes V, Chappell LC, Seed PT, et al. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology. 2014;59:1482-1491.
CASE Pregnant woman with intense itching
A 28-year-old woman (G1P0) is seen for a routine prenatal visit at 32 3/7 weeks’ gestation. She reports having generalized intense itching, including on her palms and soles, that is most intense at night and has been present for approximately 1 week. Her pregnancy is otherwise uncomplicated to date. Physical exam is within normal limits, with no evidence of a skin rash. Cholestasis of pregnancy is suspected, and laboratory tests are ordered, including bile acids and liver transaminases. Test results show that her aspartate transaminase (AST) and alanine transaminase (ALT) levels are mildly elevated at 55 IU/L and 41 IU/L, respectively, and several days later her bile acid level result is 21 µmol/L.
How should this patient be managed?
Intrahepatic cholestasis of pregnancy (ICP) affects 0.5% to 0.7% of pregnant individuals and results in maternal pruritus and elevated serum bile acid levels.1-3 Pruritus in ICP typically is generalized, including occurrence on the palms of the hands and soles of the feet, and it often is reported to be worse at night.4 Up to 25% of pregnant women will develop pruritus during pregnancy but the majority will not have ICP.2,5 Patients with ICP have no associated rash, but clinicians may note excoriations on exam. ICP typically presents in the third trimester of pregnancy but has been reported to occur earlier in gestation.6
Making a diagnosis of ICP
The presence of maternal pruritus in the absence of a skin condition along with elevated levels of serum bile acids are required for the diagnosis of ICP.7 Thus, a thorough history and physical exam is recommended to rule out another skin condition that could potentially explain the patient’s pruritus.
Some controversy exists regarding the bile acid level cutoff that should be used to make a diagnosis of ICP.8 It has been noted that nonfasting serum bile acid levels in pregnancy are considerably higher than those in in the nonpregnant state, and an upper limit of 18 µmol/L has been proposed as a cutoff in pregnancy.9 However, nonfasting total serum bile acids also have been shown to vary considerably by race, with levels 25.8% higher in Black women compared with those in White women and 24.3% higher in Black women compared with those in south Asian women.9 This raises the question of whether we should be using race-specific bile acid values to make a diagnosis of ICP.
Bile acid levels also vary based on whether a patient is in a fasting or postprandial state.10 Despite this variation, most guidelines do not recommend testing fasting bile acid levels as the postprandial state effect overall is small.7,9,11 The Society for Maternal-Fetal Medicine (SMFM) recommends that if a pregnancy-specific bile acid range is available from the laboratory, then the upper limit of normal for pregnancy should be used when making a diagnosis of ICP.7 The SMFM guidelines also acknowledge, however, that pregnancy-specific values rarely are available, and in this case, levels above the upper limit of normal—often 10 µmol/L should be considered diagnostic for ICP until further evidence regarding optimal bile acid cutoff levels in pregnancy becomes available.7
For patients with suspected ICP, liver transaminase levels should be measured in addition to nonfasting serum bile acid levels.7 A thorough history should include assessment for additional symptoms of liver disease, such as changes in weight, appetite, jaundice, excessive fatigue, malaise, and abdominal pain.7 Elevated transaminases levels may be associated with ICP, but they are not necessary for diagnosis. In the absence of additional clinical symptoms that suggest underlying liver disease or severe early onset ICP, additional evaluation beyond nonfasting serum bile acids and liver transaminase levels, such as liver ultrasonography or evaluation for viral or autoimmune hepatitis, is not recommended.7 Obstetric care clinicians should be aware that there is an increased incidence of preeclampsia among patients with ICP, although no specific guidance regarding further recommendations for screening is provided.7

Continue to: Risks associated with ICP...
Risks associated with ICP
For both patients and clinicians, the greatest concern among patients with ICP is the increased risk of stillbirth. Stillbirth risk in ICP appears to be related to serum bile acid levels and has been reported to be highest in patients with bile acid levels greater than 100 µmol/L. A systematic review and meta-analysis of ICP studies demonstrated no increased risk of stillbirth among patients with bile acid levels less than 100 µmol/L.12 These results, however, must be interpreted with extreme caution as the majority of studies included patients with ICP who were actively managed with attempts to mitigate the risk of stillbirth.7
In the absence of additional pregnancy risk factors, the risk of stillbirth among patients with ICP and serum bile acid levels between 19 and 39 µmol/L does not appear to be elevated above their baseline risk.11 The same is true for pregnant individuals with ICP and no additional pregnancy risk factors with serum bile acid levels between 40 and 99 µmol/L until approximately 38 weeks’ gestation, when the risk of stillbirth is elevated.11 The risk of stillbirth is elevated in ICP with peak bile acid levels greater than 100 µmol/L, with an absolute risk of 3.44%.11
Management of patients with ICP
Laboratory evaluation
There is no consensus on the need for repeat testing of bile acid levels in patients with ICP. SMFM advises that follow-up testing of bile acid levels may help to guide delivery timing, especially in cases of severe ICP, but the society recommends against serial testing.7 By contrast, the Royal College of Obstetricians and Gynaecologists (RCOG) provides a detailed algorithm regarding time intervals between serum bile acid level testing to guide delivery timing.11 The TABLE lists the strategy for reassessment of serum bile acid levels in ICP as recommended by the RCOG.11

In the United States, bile acid testing traditionally takes several days as the testing is commonly performed at reference laboratories. We therefore suggest that clinicians consider repeating bile acid level testing in situations in which the timing of delivery may be altered if further elevations of bile acid levels were noted. This is particularly relevant for patients diagnosed with ICP early in the third trimester when repeat bile acid levels would still allow for an adjustment in delivery timing.
Antepartum fetal surveillance
Unfortunately, antepartum fetal testing for pregnant patients with ICP does not appear to reliably predict or prevent stillbirth as several studies have reported stillbirths within days of normal fetal testing.13-16 It is therefore important to counsel pregnant patients regarding monitoring of fetal movements and advise them to present for evaluation if concerns arise.
Currently, SMFM recommends that patients with ICP should begin antenatal fetal surveillance at a gestational age when abnormal fetal testing would result in delivery.7 Patients should be counseled, however, regarding the unpredictability of stillbirth with ICP in the setting of a low absolute risk of such.
Medications
While SMFM recommends a starting dose of ursodeoxycholic acid 10 to 15 mg/kg per day divided into 2 or 3 daily doses as first-line therapy for the treatment of maternal symptoms of ICP, it is important to acknowledge that the goal of treatment is to alleviate maternal symptoms as there is no evidence that ursodeoxycholic acid improves either maternal serum bile acid levels or perinatal outcomes.7,17,18 Since publication of the guidelines, ursodeoxycholic acid’s lack of benefit has been further confirmed in a meta-analysis, and thus discontinuation is not unreasonable in the absence of any improvement in maternal symptoms.18
Timing of delivery
The optimal management of ICP remains unknown. SMFM recommends delivery based on peak serum bile acid levels. Delivery is recommended at 36 weeks’ gestation with ICP and total bile acid levels greater than 100 µmol/L as these patients have the greatest risk of stillbirth.7 For patients with ICP and bile acid levels less than 100 µmol/L, delivery is recommended between 36 0/7 and 39 0/7 weeks’ gestation.7 This is a wide gestational age window for clinicians to consider timing of delivery, and certainly the risks of stillbirth should be carefully balanced with the morbidity associated with a preterm or early term delivery.
For patients with ICP who have bile acid levels greater than 40 µmol/L, it is reasonable to consider delivery earlier in the gestational age window, given an evidence of increased risk of stillbirth after 38 weeks.7,12 For patients with ICP who have bile acid levels less than 40 µmol/L, delivery closer to 39 weeks’ gestation is recommended, as the risk of stillbirth does not appear to be increased above the baseline risk.7,12 Clinicians should be aware that the presence of concomitant morbidities, such as preeclampsia and gestational diabetes, are associated with an increased risk of stillbirth and should be considered for delivery planning.19
Postpartum follow-up
Routine laboratory evaluation following delivery is not recommended.7 However, in the presence of persistent pruritus or other signs and symptoms of hepatobiliary disease, liver function tests should be repeated with referral to hepatology if results are persistently abnormal 4 to 6 weeks postpartum.7
CASE Patient follow-up and outcomes
CASE Pregnant woman with intense itching
A 28-year-old woman (G1P0) is seen for a routine prenatal visit at 32 3/7 weeks’ gestation. She reports having generalized intense itching, including on her palms and soles, that is most intense at night and has been present for approximately 1 week. Her pregnancy is otherwise uncomplicated to date. Physical exam is within normal limits, with no evidence of a skin rash. Cholestasis of pregnancy is suspected, and laboratory tests are ordered, including bile acids and liver transaminases. Test results show that her aspartate transaminase (AST) and alanine transaminase (ALT) levels are mildly elevated at 55 IU/L and 41 IU/L, respectively, and several days later her bile acid level result is 21 µmol/L.
How should this patient be managed?
Intrahepatic cholestasis of pregnancy (ICP) affects 0.5% to 0.7% of pregnant individuals and results in maternal pruritus and elevated serum bile acid levels.1-3 Pruritus in ICP typically is generalized, including occurrence on the palms of the hands and soles of the feet, and it often is reported to be worse at night.4 Up to 25% of pregnant women will develop pruritus during pregnancy but the majority will not have ICP.2,5 Patients with ICP have no associated rash, but clinicians may note excoriations on exam. ICP typically presents in the third trimester of pregnancy but has been reported to occur earlier in gestation.6
Making a diagnosis of ICP
The presence of maternal pruritus in the absence of a skin condition along with elevated levels of serum bile acids are required for the diagnosis of ICP.7 Thus, a thorough history and physical exam is recommended to rule out another skin condition that could potentially explain the patient’s pruritus.
Some controversy exists regarding the bile acid level cutoff that should be used to make a diagnosis of ICP.8 It has been noted that nonfasting serum bile acid levels in pregnancy are considerably higher than those in in the nonpregnant state, and an upper limit of 18 µmol/L has been proposed as a cutoff in pregnancy.9 However, nonfasting total serum bile acids also have been shown to vary considerably by race, with levels 25.8% higher in Black women compared with those in White women and 24.3% higher in Black women compared with those in south Asian women.9 This raises the question of whether we should be using race-specific bile acid values to make a diagnosis of ICP.
Bile acid levels also vary based on whether a patient is in a fasting or postprandial state.10 Despite this variation, most guidelines do not recommend testing fasting bile acid levels as the postprandial state effect overall is small.7,9,11 The Society for Maternal-Fetal Medicine (SMFM) recommends that if a pregnancy-specific bile acid range is available from the laboratory, then the upper limit of normal for pregnancy should be used when making a diagnosis of ICP.7 The SMFM guidelines also acknowledge, however, that pregnancy-specific values rarely are available, and in this case, levels above the upper limit of normal—often 10 µmol/L should be considered diagnostic for ICP until further evidence regarding optimal bile acid cutoff levels in pregnancy becomes available.7
For patients with suspected ICP, liver transaminase levels should be measured in addition to nonfasting serum bile acid levels.7 A thorough history should include assessment for additional symptoms of liver disease, such as changes in weight, appetite, jaundice, excessive fatigue, malaise, and abdominal pain.7 Elevated transaminases levels may be associated with ICP, but they are not necessary for diagnosis. In the absence of additional clinical symptoms that suggest underlying liver disease or severe early onset ICP, additional evaluation beyond nonfasting serum bile acids and liver transaminase levels, such as liver ultrasonography or evaluation for viral or autoimmune hepatitis, is not recommended.7 Obstetric care clinicians should be aware that there is an increased incidence of preeclampsia among patients with ICP, although no specific guidance regarding further recommendations for screening is provided.7

Continue to: Risks associated with ICP...
Risks associated with ICP
For both patients and clinicians, the greatest concern among patients with ICP is the increased risk of stillbirth. Stillbirth risk in ICP appears to be related to serum bile acid levels and has been reported to be highest in patients with bile acid levels greater than 100 µmol/L. A systematic review and meta-analysis of ICP studies demonstrated no increased risk of stillbirth among patients with bile acid levels less than 100 µmol/L.12 These results, however, must be interpreted with extreme caution as the majority of studies included patients with ICP who were actively managed with attempts to mitigate the risk of stillbirth.7
In the absence of additional pregnancy risk factors, the risk of stillbirth among patients with ICP and serum bile acid levels between 19 and 39 µmol/L does not appear to be elevated above their baseline risk.11 The same is true for pregnant individuals with ICP and no additional pregnancy risk factors with serum bile acid levels between 40 and 99 µmol/L until approximately 38 weeks’ gestation, when the risk of stillbirth is elevated.11 The risk of stillbirth is elevated in ICP with peak bile acid levels greater than 100 µmol/L, with an absolute risk of 3.44%.11
Management of patients with ICP
Laboratory evaluation
There is no consensus on the need for repeat testing of bile acid levels in patients with ICP. SMFM advises that follow-up testing of bile acid levels may help to guide delivery timing, especially in cases of severe ICP, but the society recommends against serial testing.7 By contrast, the Royal College of Obstetricians and Gynaecologists (RCOG) provides a detailed algorithm regarding time intervals between serum bile acid level testing to guide delivery timing.11 The TABLE lists the strategy for reassessment of serum bile acid levels in ICP as recommended by the RCOG.11

In the United States, bile acid testing traditionally takes several days as the testing is commonly performed at reference laboratories. We therefore suggest that clinicians consider repeating bile acid level testing in situations in which the timing of delivery may be altered if further elevations of bile acid levels were noted. This is particularly relevant for patients diagnosed with ICP early in the third trimester when repeat bile acid levels would still allow for an adjustment in delivery timing.
Antepartum fetal surveillance
Unfortunately, antepartum fetal testing for pregnant patients with ICP does not appear to reliably predict or prevent stillbirth as several studies have reported stillbirths within days of normal fetal testing.13-16 It is therefore important to counsel pregnant patients regarding monitoring of fetal movements and advise them to present for evaluation if concerns arise.
Currently, SMFM recommends that patients with ICP should begin antenatal fetal surveillance at a gestational age when abnormal fetal testing would result in delivery.7 Patients should be counseled, however, regarding the unpredictability of stillbirth with ICP in the setting of a low absolute risk of such.
Medications
While SMFM recommends a starting dose of ursodeoxycholic acid 10 to 15 mg/kg per day divided into 2 or 3 daily doses as first-line therapy for the treatment of maternal symptoms of ICP, it is important to acknowledge that the goal of treatment is to alleviate maternal symptoms as there is no evidence that ursodeoxycholic acid improves either maternal serum bile acid levels or perinatal outcomes.7,17,18 Since publication of the guidelines, ursodeoxycholic acid’s lack of benefit has been further confirmed in a meta-analysis, and thus discontinuation is not unreasonable in the absence of any improvement in maternal symptoms.18
Timing of delivery
The optimal management of ICP remains unknown. SMFM recommends delivery based on peak serum bile acid levels. Delivery is recommended at 36 weeks’ gestation with ICP and total bile acid levels greater than 100 µmol/L as these patients have the greatest risk of stillbirth.7 For patients with ICP and bile acid levels less than 100 µmol/L, delivery is recommended between 36 0/7 and 39 0/7 weeks’ gestation.7 This is a wide gestational age window for clinicians to consider timing of delivery, and certainly the risks of stillbirth should be carefully balanced with the morbidity associated with a preterm or early term delivery.
For patients with ICP who have bile acid levels greater than 40 µmol/L, it is reasonable to consider delivery earlier in the gestational age window, given an evidence of increased risk of stillbirth after 38 weeks.7,12 For patients with ICP who have bile acid levels less than 40 µmol/L, delivery closer to 39 weeks’ gestation is recommended, as the risk of stillbirth does not appear to be increased above the baseline risk.7,12 Clinicians should be aware that the presence of concomitant morbidities, such as preeclampsia and gestational diabetes, are associated with an increased risk of stillbirth and should be considered for delivery planning.19
Postpartum follow-up
Routine laboratory evaluation following delivery is not recommended.7 However, in the presence of persistent pruritus or other signs and symptoms of hepatobiliary disease, liver function tests should be repeated with referral to hepatology if results are persistently abnormal 4 to 6 weeks postpartum.7
CASE Patient follow-up and outcomes
- Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37.
- Kenyon AP, Tribe RM, Nelson-Piercy C, et al. Pruritus in pregnancy: a study of anatomical distribution and prevalence in relation to the development of obstetric cholestasis. Obstet Med. 2010;3:25-29.
- Wikstrom Shemer E, Marschall HU, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated adverse pregnancy and fetal outcomes: a 12-year population-based cohort study. BJOG. 2013;120:717-723.
- Ambros-Rudolph CM, Glatz M, Trauner M, et al. The importance of serum bile acid level analysis and treatment with ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a case series from central Europe. Arch Dermatol. 2007;143:757-762.
- Szczech J, Wiatrowski A, Hirnle L, et al. Prevalence and relevance of pruritus in pregnancy. Biomed Res Int. 2017;2017:4238139.
- Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049-2066.
- Society for Maternal-Fetal Medicine; Lee RH, Greenberg M, Metz TD, et al. Society for Maternal-Fetal Medicine Consult Series #53: intrahepatic cholestasis of pregnancy: replaces Consult #13, April 2011. Am J Obstet Gynecol. 2021;224:B2-B9.
- Horgan R, Bitas C, Abuhamad A. Intrahepatic cholestasis of pregnancy: a comparison of Society for Maternal-Fetal Medicine and the Royal College of Obstetricians and Gynaecologists’ guidelines. Am J Obstet Gynecol MFM. 2023;5:100838.
- Mitchell AL, Ovadia C, Syngelaki A, et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: case-control and cohort study. BJOG. 2021;128:1635-1644.
- Adams A, Jacobs K, Vogel RI, et al. Bile acid determination after standardized glucose load in pregnant women. AJP Rep. 2015;5:e168-e171.
- Girling J, Knight CL, Chappell L; Royal College of Obstetricians and Gynaecologists. Intrahepatic cholestasis of pregnancy: Green-top guideline no. 43, June 2022. BJOG. 2022;129:e95-e114.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393:899-909.
- Alsulyman OM, Ouzounian JG, Ames-Castro M, et al. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960.
- Herrera CA, Manuck TA, Stoddard GJ, et al. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2018;31:1913-1920.
- Lee RH, Incerpi MH, Miller DA, et al. Sudden fetal death in intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2009;113:528-531.
- Sentilhes L, Verspyck E, Pia P, et al. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460.
- Chappell LC, Bell JL, Smith A, et al; PITCHES Study Group. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial. Lancet. 2019;394:849-860.
- Ovadia C, Sajous J, Seed PT, et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a systematic review and individual participant data meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:547-558.
- Geenes V, Chappell LC, Seed PT, et al. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology. 2014;59:1482-1491.
- Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37.
- Kenyon AP, Tribe RM, Nelson-Piercy C, et al. Pruritus in pregnancy: a study of anatomical distribution and prevalence in relation to the development of obstetric cholestasis. Obstet Med. 2010;3:25-29.
- Wikstrom Shemer E, Marschall HU, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated adverse pregnancy and fetal outcomes: a 12-year population-based cohort study. BJOG. 2013;120:717-723.
- Ambros-Rudolph CM, Glatz M, Trauner M, et al. The importance of serum bile acid level analysis and treatment with ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a case series from central Europe. Arch Dermatol. 2007;143:757-762.
- Szczech J, Wiatrowski A, Hirnle L, et al. Prevalence and relevance of pruritus in pregnancy. Biomed Res Int. 2017;2017:4238139.
- Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049-2066.
- Society for Maternal-Fetal Medicine; Lee RH, Greenberg M, Metz TD, et al. Society for Maternal-Fetal Medicine Consult Series #53: intrahepatic cholestasis of pregnancy: replaces Consult #13, April 2011. Am J Obstet Gynecol. 2021;224:B2-B9.
- Horgan R, Bitas C, Abuhamad A. Intrahepatic cholestasis of pregnancy: a comparison of Society for Maternal-Fetal Medicine and the Royal College of Obstetricians and Gynaecologists’ guidelines. Am J Obstet Gynecol MFM. 2023;5:100838.
- Mitchell AL, Ovadia C, Syngelaki A, et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: case-control and cohort study. BJOG. 2021;128:1635-1644.
- Adams A, Jacobs K, Vogel RI, et al. Bile acid determination after standardized glucose load in pregnant women. AJP Rep. 2015;5:e168-e171.
- Girling J, Knight CL, Chappell L; Royal College of Obstetricians and Gynaecologists. Intrahepatic cholestasis of pregnancy: Green-top guideline no. 43, June 2022. BJOG. 2022;129:e95-e114.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393:899-909.
- Alsulyman OM, Ouzounian JG, Ames-Castro M, et al. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960.
- Herrera CA, Manuck TA, Stoddard GJ, et al. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2018;31:1913-1920.
- Lee RH, Incerpi MH, Miller DA, et al. Sudden fetal death in intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2009;113:528-531.
- Sentilhes L, Verspyck E, Pia P, et al. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460.
- Chappell LC, Bell JL, Smith A, et al; PITCHES Study Group. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy (PITCHES): a randomised controlled trial. Lancet. 2019;394:849-860.
- Ovadia C, Sajous J, Seed PT, et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: a systematic review and individual participant data meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:547-558.
- Geenes V, Chappell LC, Seed PT, et al. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: a prospective population-based case-control study. Hepatology. 2014;59:1482-1491.
Managing clinician burnout: Challenges and opportunities
Physicians have some of the highest rates of burnout among all professions.1 Complicating matters is that clinicians (including residents)2 may avoid seeking treatment out of fear it will affect their license or privileges.3 In this article, we consider burnout in greater detail, as well as ways of successfully addressing the level of burnout in the profession (FIGURE 1), including steps individual practitioners, health care entities, and regulators should consider to reduce burnout and its harmful effects.
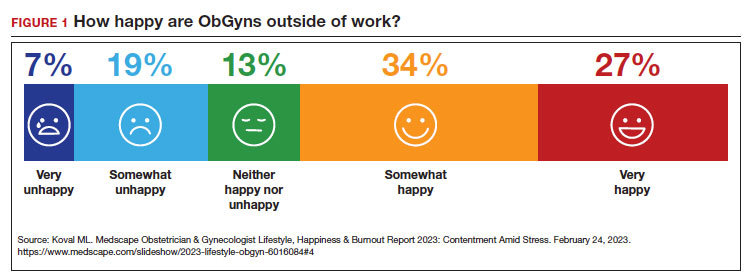
How burnout becomes a problem
Six general factors are commonly identified as leading to clinician career dissatisfaction and burnout:4
1. work overload
2. lack of autonomy and control
3. inadequate rewards, financial and otherwise
4. work-home schedules
5. perception of lack of fairness
6. values conflict between the clinician and employer (including a breakdown of professional community).
At the top of the list of causes of burnout is often “administrative and bureaucratic headaches.”5 More specifically, electronic health records (EHRs), including computerized order entry, is commonly cited as a major cause of burnout.6,7 According to some studies, clinicians spend as much as 49% of working time doing clerical work,8 and studies found the extension of work into home life.9
Increased measurement of performance metrics in health care services are a significant contributor to physician burnout.10 These include pressure to see more patients, perform more procedures, and respond quickly to patient requests (eg, through email).7 As we will see, medical malpractice cases, or the risk of such cases, have also played a role in burnout in some medical specialties.11 The pandemic also contributed, at least temporarily, to burnout.12,13
Rates of burnout among physicians are notably higher than among the general population14 or other professions.6 Although physicians have generally entered clinical practice with lower rates of burnout than the general population,15 The American College of Obstetricians and Gynecologists (ACOG) reports that 40% to 75% of ObGyns “experience some form of professional burnout.”16,17 Other source(s) cite that 53% of ObGyns report burnout (TABLE 1).
Code QD85
Burnout is a syndrome conceptualized as resulting from chronic workplace stress that has not been successfully managed. It is characterized by 3 dimensions:
- feelings of energy depletion or exhaustion
- increased mental distance from one’s job, or feelings of negativism or cynicism related to one’s job
- a sense of ineffectiveness and lack of accomplishment. Burn-out refers specifically to phenomena in the occupational context and should not be applied to describe experiences in other areas of life. Exclusions to burnout diagnosis include adjustment disorder, disorders specifically associated with stress, anxiety or fear-related disorders, and mood disorders.
Reference
1. International Classification of Diseases Eleventh Revision (ICD-11). Geneva, Switzerland: World Health Organization; 2022.
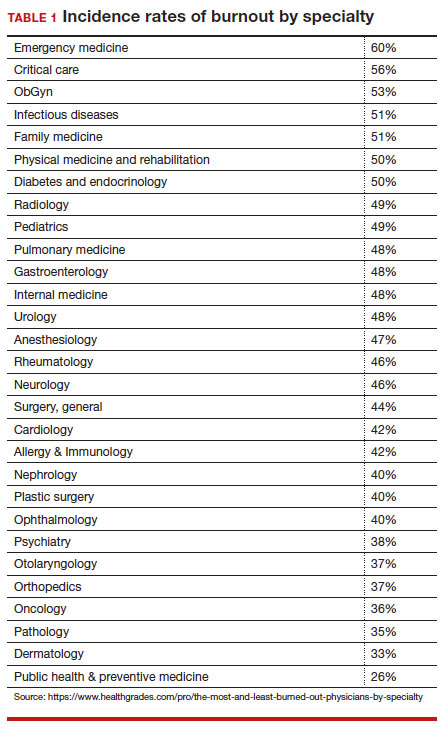
Burnout undoubtedly contributes to professionals leaving practice, leading to a significant shortage of ObGyns.18 It also raises several significant legal concerns. Despite the enormity and seriousness of the problem, there is considerable optimism and assurance that the epidemic of burnout is solvable on the individual, specialty, and profession-wide levels. ACOG and other organizations have made suggestions for physicians, the profession, and to health care institutions for reducing burnout.19 This is not to say that solutions are simple or easy for individual professionals or institutions, but they are within the reach of the profession (FIGURE 2).
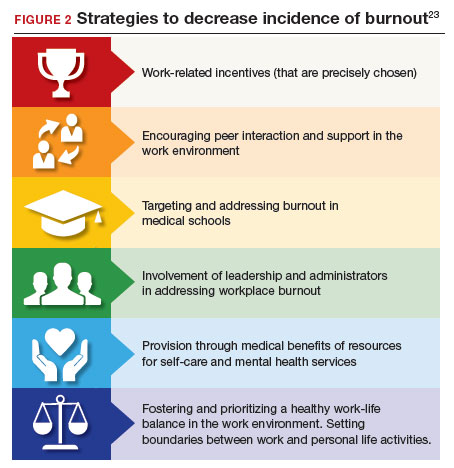
Suicide among health care professionals is one other concern (TABLE 2)20 and theoretically can stem from burnout, depression, and other psychosocial concerns.

Costs of clinician burnout
Burnout is endemic among health care providers, with numerous studies detailing the professional, emotional, and financial costs. Prior to the pandemic, one analysis of nationwide fiscal costs associated with burnout estimated an annual cost of $4.6B due to physician turnover and reduced clinical hours.21 The COVID-19 epidemic has by all accounts worsened rates of health care worker burnout, particularly for those in high patient-contact positions.22
Female clinicians appear to be differentially affected; in one recent study women reported symptoms of burnout at twice the rate of their male counterparts.23 Whether burnout rates will return to pre-pandemic levels remains an open question, but since burnout is frequently related to one’s own assessment of work-life balance, it is possible that a longer term shift in burnout rates associated with post-pandemic occupational attitudes will be observed.
Combining factors contribute to burnout
Burnout is a universal occupational hazard, but extant data suggest that physicians and other health care providers may be at higher risk. Among physicians, younger age, female gender, and front-line specialty status appear associated with higher burnout rates.24 Given that ObGyn physicians are overwhelmingly female (60% of physicians and 86% of residents),25,26 gender-related burnout factors exist alongside other specific occupational burnout risks. While gender parity has been achieved among health care providers, gender disparities persist in terms of those in leadership positions, compensation, and other factors.22
The smattering of evidence suggesting that ObGyns have higher rates of burnout than many other specialties is understandable given the unique legal challenges confronting ObGyn practice. This may be of special significance because ObGyn malpractice insurance rates are among the highest of all specialties.27 The overall shortage of ObGyns has been exacerbated by the demonstrated negative effects on training and workforce representation stemming from recent legislation that has the effect of criminalizing certain aspects of ObGyn practice;28 for instance, uncertainty regarding abortion regulations.
These negative effects are particularly heightened in states in which the law is in flux or where there are continuing efforts to substantially limit access to abortion. The efforts to increase civil and even criminal penalties related to abortion care challenge ObGyns’ professional practices, as legal rules are frequently changing. In some states, ObGyns may face additional workloads secondary to a flight of ObGyns from restrictive jurisdictions in addition to legal and professional repercussions. In a small study of 19 genetic counselors dealing with restrictive legislation in the state of Ohio,29 increased stress and burnout rates were identified as a consequence of practice uncertainties under this legislation. It is certain that other professionals working in reproductive health care are similarly affected.30
The programs provide individual resources to providers in distress, periodically survey initiatives at Stanford to assess burnout at the organizational level, and provide input designed to spur organizational change to reduce the burden of burnout. Ways that they build community and connections include:
- Live Story Rounds events (as told by Stanford Medicine physicians)
- Commensality Groups (facilitated small discussion groups built around tested evidence)
- Aim to increase sense of connection and collegiality among physicians and build comradery at work
- CME-accredited physician wellness forum, including annual doctor’s day events
Continue to: Assessment of burnout...
Assessment of burnout
Numerous scales for the assessment of burnout exist. Of these, the 22-item Maslach Burnout Inventory (MBI) is the best studied. The MBI is a well-investigated tool for assessing burnout. The MBI consists of 3 major subscales measuring overall burnout, emotional exhaustion, depersonalization, and low personal accomplishment. It exists in numerous forms. For instance, the MBI-HSS (MP), adapted for medical personnel, is available. However, the most commonly used form for assessing burnout in clinicians is the MBI-HHS (Human Services Survey); approximately 85% of all burnout studies examined in a recent meta-analysis used this survey version.31 As those authors commented, while burnout is a recognized phenomenon, a great deal of variability in study design, interpretation of subscale scores, and sample selection makes generalizations regarding burnout difficult to assess.
The MBI in various forms has been extensively used over the past 40 years to assess burnout amongst physicians and physicians in training. While not the only instrument designed to measure such factors, it is by far the most prevalent. Williamson and colleagues32 compared the MBI with several other measures of quality of life and found good correlation between the various instruments used, a finding replicated by other studies.33 Brady and colleagues compared item responses to the Stanford Professional Fulfillment Index and the Min-Z Single-item Burnout scale (a 1-item screening measure) to MBI’s Emotional Exhaustion and Depersonalization subscales. Basing their findings on a survey of more than 1,300 physicians, they found that all analyzed scales were significantly correlated with such adverse outcomes as depression, distress, or intent to leave the profession.
It is important to note that most surveys of clinician burnout were conducted prior to the pandemic. While the psychometric analyses of the MBI and other scales are likely still germane, observed rates of clinician burnout have likely increased. Thus, comparisons of pre- and post-pandemic studies should factor in an increase in the incidence and prevalence of burnout.
Management strategies
In general, there are several interventions for managing burnout34:
- individual-focused (including self-care and communications-skills workshops)
- mindfulness training
- yoga
- meditation
- organizational/structural (workload reduction, schedule realignment, teamwork training, and group-delivered stress management interventions)
- combination(s) of the above.
There is little evidence to suggest that any particular individual intervention (whether delivered in individual or group-based formats) is superior to any other in treating clinician burnout. A recent analysis of 24 studies employing mindfulness-based interventions demonstrated generally positive results for such interventions.35 Other studies have also found general support for mindfulness-based interventions, although mindfulness is often integrated with other stress-reduction techniques, such as meditation, yoga, and communication skills. Such interventions are nonspecific but generally effective.
An accumulation of evidence to date suggests that a combination of individual and organizational interventions is most effective in combatting clinician burnout. No individual intervention can be successful without addressing root causes, such as overscheduling, lack of organizational support, and the effect of restrictive legislation on practice.
Several large teaching hospitals have established programs to address physician and health care provider burnout. Notable among these is the Stanford University School of Medicine’s WellMD and WellPhD programs (https://wellmd.stanford.edu/about.html). These programs were described by Olson and colleagues36 as using a model focused on practice efficiency, organizational culture, and personal resilience to enhance physicians’ well-being. (See “Aspects of the WellMD and WellPhD programs from Stanford University.”)
A growing number of institutions have established burnout programs to support physicians experiencing work/life imbalances and other aspects of burnout.37 In general, these share common features of assessment, individual and/or group intervention, and organizational change. Fear of repercussion may be one factor preventing physicians from seeking individual treatment for burnout.38 Importantly, they emphasize the need for professional confidentiality when offering treatment to patients within organizational settings. Those authors also reported that a focus on organizational engagement may be an important factor in addressing burnout in female physicians, as they tend to report lower levels of organizational engagement.
Continue to: Legal considerations...
Legal considerations
Until recently, physician burnout “received little notice in the legal literature.”39 Although there have been burnout legal consequences in the past, the legal issues are now becoming more visible.40
Medical malpractice
A well-documented consequence of burnout is an increase in errors.14 Medical errors, of course, are at the heart of malpractice claims. Technically, malpractice is medical or professional negligence. It is the breach of a duty owed by the physician, or other provider, or organization (defendant) to the patient, which causes injury to the plaintiff/patient.41
“Medical error” is generally a meaningful deviation from the “standard of care” or accepted medical practice.42 Many medical errors do not cause injury to the patient; in those cases, the negligence does not result in liability. In instances in which the negligence causes harm, the clinician and health care facility may be subject to liability for that injury. Fortunately, however, for a variety of reasons, most harmful medical errors do not result in a medical malpractice claim or lawsuit. The absence of a good clinician-patient relationship is likely associated with an increased inclination of a patient to file a malpractice action.43Clinician burnout may, therefore, contribute to increased malpractice claims in two ways. First, burnout likely leads to increased medical errors, perhaps because burnout is associated with lower concentration, inattention, reduced cognitive vigilance, and fatigue.8,44 It may also lead to less time with patients, reduced patient empathy, and lower patient rapport, which may make injured patients more likely to file a claim or lawsuit.45 Because the relationshipbetween burnout and medical error is bidirectional, malpractice claims tend to increase burnout, which increases error. Given the time it takes to resolve most malpractice claims, the uncertainty of medical malpractice may be especially stressful for health care providers.46,47
Burnout is not a mitigating factor in malpractice. Our sympathies may go out to a professional suffering from burnout, but it does not excuse or reduce liability—it may, indeed, be an aggravating factor. Clinicians who can diagnose burnout and know its negative consequences but fail to deal with their own burnout may be demonstrating negligence if there has been harm to a patient related to the burnout.48
Institutional or corporate liability to patients
Health care institutions have obligations to avoid injury to patients. Just as poorly maintained medical equipment may harm patients, so may burned-out professionals. Therefore, institutions have some obligation to supervise and avoid the increased risks to patients posed by professionals suffering from burnout.
Respondeat superior and institutional negligence. Institutional liability may arise in two ways, the first through agency, or respondeat superior. That is, if the physician or other professional is an employee (or similar agent) of the health care institution, that institution is generally responsible for the physician’s negligence during the employment.49 Even if the physician is not an employee (for example, an independent contractor providing care or using the hospital facilities), the health care facility may be liable for the physician’s negligence.50 Liability may occur, for example, if the health care facility was aware that the physician was engaged in careless practice or was otherwise a risk to patients but the facility did not take steps to avoid those risks.51 The basis for liability is that the health care organization owes a duty to patients to take reasonable care to ensure that its facilities are not used to injure patients negligently.52 Just as it must take care that unqualified physicians are not granted privileges to practice, it also must take reasonable steps to protect patients when it is aware (through nurses or other agents) of a physician’s negligent practice.
In one case, for example, the court found liability where a staff member had “severe” burnout in a physician’s office and failed to read fetal monitoring strips. The physician was found negligent for relying on the staff member who was obviously making errors in interpretation of fetal distress.53
Continue to: Legal obligations of health care organizations to physicians and others...
Legal obligations of health care organizations to physicians and others
In addition to obligations to patients, health care organizations may have obligations to employees (and others) at risk for injury. For example, assume a patient is diagnosed with a highly contagious disease. The health care organization would be obligated to warn, and take reasonable steps to protect, the staff (employees and independent contractors) from being harmed from exposure to the disease. This principle may apply to coworkers of employees with significant burnout, thereby presenting a danger in the workplace. The liability issue is more difficult for employees experiencing job-related burnout themselves. Organizations generally compensate injured employees through no-fault workers’ compensation (an insurance-like system); for independent contractors, the liability is usually through a tort claim (negligence).54
In modern times, a focus has been on preventing those injuries, not just providing compensation after injuries have occurred. Notably, federal and state occupational health and safety laws (particularly the Occupational Safety and Health Administration [OSHA]) require most organizations (including those employing health care providers) to take steps to mitigate various kinds of worker injuries.55
Although these worker protections have commonly been applied to hospitals and other health care providers, burnout has not traditionally been a significant concern in federal or state OSHA enforcement. For example, no formal federal OSHA regulations govern work-related burnout. Regulators, including OSHA, are increasingly interested in burnout that may affect many employees. OSHA has several recommendations for reducing health care work burnout.56 The Surgeon General has expressed similar concerns.57 The federal government recently allocated $103 million from the American Rescue Plan to address burnout among health care workers.58 Also, OSHA appears to be increasing its oversight of healthcare-institution-worker injuries.55
Is burnout a “disability”?
The federal Americans with Disabilities Act (ADA) and similar state laws prohibit discrimination based on disability.59 A disability is defined as a “physical or mental impairment that substantially limits one or more major life activities” or “perceived as having such an impairment.”60 The initial issue is whether burnout is a “mental impairment.” As noted earlier, it is not officially a “medical condition.”61 To date, the United Nations has classified it as an “occupational phenomenon.”62 It may, therefore, not qualify under the ADA, even if it “interferes with a major life activity.” There is, however, some movement toward defining burnout as a mental condition. Even if defined as a disability, there would still be legal issues of how severe it must be to qualify as a disability and the proper accommodation. Apart from the legal definition of an ADA disability, as a practical matter it likely is in the best interest of health care facilities to provide accommodations that reduce burnout. A number of strategies to decrease the incidence of burnout include the role of health care systems (FIGURE 2).
In conclusion we look at several things that can be done to “treat” or reduce burnout. That effort requires the cooperation of physicians and other providers, health care facilities, training programs, licensing authorities, and professional organizations. See suggestions below.
Conclusion
There are many excellent suggestions for reducing burnout and improving patient care and practitioner satisfaction.63-65 We conclude with a summary of some of these suggestions for individual practitioners, health care organizations, the profession, and licensing. It is worth remembering, however, that it will require the efforts of each area to reduce burnout substantially.
For practitioners:
- Engage in quality coaching/therapy on mindfulness and stress management.
- Practice self-care, including exercise and relaxation techniques.
- Make work-life balance a priority.
- Take opportunities for collegial social and professional discussions.
- Prioritize (and periodically assess) your own professional satisfaction and burnout risk.
- Smile—enjoy a sense of humor (endorphins and cortisol).
For health care organizations:
- Urgently work with vendors and regulators to revise electronic health records to reduce their substantial impact on burnout.
- Reduce physicians’ time on clerical and administrative tasks (eg, by enhancing the use of quality AI, scribes, and automated notes from appointments. (This may increase the time they spend with patients.) Eliminate “pajama-time” charting.
- Provide various kinds of confidential professional counseling, therapy, and support related to burnout prevention and treatment, and avoid any penalty or stigma related to their use.
- Provide reasonable flexibility in scheduling.
- Routinely provide employees with information about burnout prevention and services.
- Appoint a wellness officer with authority to ensure the organization maximizes its prevention and treatment services.
- Constantly seek input from practitioners on how to improve the atmosphere for practice to maximize patient care and practitioner satisfaction.
- Provide ample professional and social opportunities for discussing and learning about work-life balance, resilience, intellectual stimulation, and career development.
For regulators, licensors, and professional organizations:
- Work with health care organizations and EHR vendors to substantially reduce the complexity, physician effort, and stress associated with those record systems. Streamlining should, in the future, be part of formally certifying EHR systems.
- Reduce the administrative burden on physicians by modifying complex regulations and using AI and other technology to the extent possible to obtain necessary reimbursement information.
- Eliminate unnecessary data gathering that requires practitioner time or attention.
- Licensing, educational, and certifying bodies should eliminate any questions regarding the diagnosis or treatment of mental health and focus on current (or very recent) impairments.
- Seek funding for research on burnout prevention and treatment.
Dr. H is a 58-year-old ObGyn who, after completing residency, went into solo practice. The practice grew, and Dr. H found it increasingly more challenging to cover, especially the obstetrics sector. Dr. H then merged the practice with a group of 3 other ObGyns. Their practice expanded, and began recruiting recent residency graduates. In time, the practice was bought out by the local hospital health care system. Dr. H was faced with complying with the rules and regulations of that health care system. The electronic health record (EHR) component proved challenging, as did the restrictions on staff hiring (and firing), but Dr. H did receive a paycheck each month and complied with it all. The health care system administrators had clear financial targets Dr. H was to meet each quarter, which created additional pressure. Dr. H used to love being an OB and providing excellent care for every patient, but that sense of accomplishment was being lost.
Dr. H increasingly found it difficult to focus because of mind wandering, especially in the operating room (OR). Thoughts occurred about retirement, the current challenges imposed by “the new way of practicing medicine” (more focused on financial productivity restraints and reimbursement), and EHR challenges. Then Dr. H’s attention would return to the OR case at hand. All of this resulted in considerable stress and emotional exhaustion, and sometimes a sense of being disconnected. A few times, colleagues or nurses had asked Dr. H if everything was “okay,” or if a break would help. Dr. H made more small errors than usual, but Dr. H’s self-assessment was “doing an adequate job.” Patient satisfaction scores (collected routinely by the health care system) declined over the last 9 months.
Six months ago, Dr. H finished doing a laparoscopic total hysterectomy and bilateral salpingo-oophorectomy and got into the right uterine artery. The estimated blood loss was 3,500 mL. Using minimally invasive techniques, Dr. H identified the bleeder and, with monopolar current, got everything under control. The patient went to the post-anesthesia care unit, and all appeared to be in order. Her vital signs were stable, and she was discharged home the same day.
The patient presented 1 week later with lower abdominal and right flank pain. Dr. H addressed the problem in the emergency department and admitted the patient for further evaluation and urology consultation. The right ureter was damaged and obstructed; ultimately, the urologist performed a psoas bladder hitch. The patient recovered slowly, lost several weeks of work, experienced significant pain, and had other disruptions and costs. Additional medical care related to the surgery is ongoing. A health care system committee asked Dr. H to explain the problem. Over the last 6 months, Dr. H’s frustration with practice and being tired and disconnected have increased.
Dr. H has received a letter from a law firm saying that he and the health care system are being sued for malpractice focused on an iatrogenic ureter injury. The letter names two very reputable experts who are prepared to testify that the patient’s injury resulted from clear negligence. Dr. H has told the malpractice carrier absolutely not to settle this case—it is “a sham— without merit.” The health care system has asked Dr. H to take a “burnout test.”
Legal considerations
Dr. H exhibits relatively clear signs of professional burnout. The fact that there was a bad outcome while Dr. H was experiencing burnout is not proof of negligence (or, breach of duty of care to the patient). Nor is it a defense or mitigation to any malpractice that occurred.
In the malpractice case, the plaintiff will have the burden of proving that Dr. H’s treatment was negligent in that it fell below the standard of care. Even if it was a medical error, the question is whether it was negligence. If the patient/plaintiff, using expert witnesses, can prove that Dr. H fell below the standard of care that caused injury, Dr. H may be liable for the resulting extra costs, loss of income, and pain and suffering resulting from the negligent care.
The health care system likely will also be responsible for Dr. H’s negligence, either through respondeat superior (for example, if Dr. H is an employee) or for its own negligence. The case for its negligence is that the nurses and assistants had repeatedly seen him making errors and becoming disengaged (to the extent that they asked Dr. H if “everything is okay” or if a break would help). Furthermore, Dr. H’s patient satisfaction scores have been declining for several months. The plaintiff will argue that Dr. H exhibited classic burnout symptoms with the attendant risks of medical errors. However, the health care system did not take action to protect patients or to assist Dr. H. In short, one way or another, there is some likelihood that the health care system may also be liable if patient injuries are found to have been caused by negligence.
At this point, the health care system also faces the question of how to work with Dr. H in the future. The most pressing question is whether or not to allow Dr. H to continue practicing. If, as it appears, Dr. H is dealing with burnout, the pressure of the malpractice claim could well increase the probability of other medical mistakes. The institution has asked Dr. H to take a burnout test, but it is unclear where things go if the test (as likely) demonstrates significant burnout. This is a counseling and human relations question, at least as much as a legal issue, and the institution should probably proceed in that way—which is, trying to understand and support Dr. H and determining what can be done to address the burnout. At the same time, the system must reasonably assess Dr. H’s fitness to continue practicing as the matters are resolved. Almost everyone shares the goal to provide every individual and corporate opportunity for Dr. H to deal with burnout issues and return to successful practice.
Dr. H will be represented in the malpractice case by counsel provided through the insurance carrier. However, Dr. H would be well advised to retain a trusted and knowledgeable personal attorney. For example, the instruction not to consider settlement is likely misguided, but Dr. H needs to talk with an attorney that Dr. H has chosen and trusts. In addition, the attorney can help guide Dr. H through a rational process of dealing with the health care system, putting the practice in order, and considering the options for the future.
The health care system should reconsider its processes to deal with burnout to ensure the quality of care, patient satisfaction, professional retention, and economic stability. Several burnoutresponse programs have had success in achieving these goals.
What’s the Verdict?
Dr. H received good mental health, legal, and professional advice. As a result, an out of court settlement was reached following pretrial discovery. Dr. H has continued consultation regarding burnout and has returned to productive practice.
- Shanafelt TD, West CP, Sinsky C, et al. Changes in burnout and satisfaction with work-life integration in physicians and the general US working population between 2011 and 2017. Mayo Clinic Proceed. 2019;94:1681-1694.
- Smith R, Rayburn W. Burnout in obstetrician-gynecologists. Its prevalence, identification, prevention, and reversal. Obstet Gynecol Clin North Am. 2021;48:231-245. https://doi. org/10.1016/j.ogc.2021.06.003
- Patti MG, Schlottmann F, Sarr MG. The problem of burnout among surgeons. JAMA Surg. 2018;153:403-404. doi:10.1001 /jamasurg.2018.0047
- Carrau D, Janis JE. Physician burnout: solutions for individuals and organizations. Plastic and Reconstructive Surgery Global Open. 2021;91-97.
- Southwick R. The key to fixing physician burnout is the workplace not the worker. Contemporary Ob/Gyn. March 13, 2023.
- Patel RS, Bachu R, Adikey A, et al. Factors related to physician burnout and its consequences: a review. Behav Sciences. 2018;8:98.
- Melnick ER, Dyrbye LN, Sinsky CA, et al. The association between perceived electronic health record usability and professional burnout among US physicians. Mayo Clinic Proceed. 2020;95:476-487.
- Shanafelt TD, Dyrbye LN, West CP. Addressing physician burnout: the way forward. JAMA. 2017;317:901-902. doi:10.1001/jama.2017.0076
- Ommaya AK, Cipriano PF, Hoyt DB, et al. Care-centered clinical documentation in the digital environment: Solutions to alleviate burnout. National Academy of Medicine Perspectives. 2018.
- Hartzband P, Groopman J. Physician burnout, interrupted. N Engl J Med. 2020;382:2485-2487. Discussion Paper, National Academy of Medicine. Accessed July 21, 2023. https://nam .edu/care
- Ji YD, Robertson FC, Patel NA, et al. Assessment of risk factors for suicide among US health care professionals. JAMA Surg. 2020;155:713-721. centered-clinical-documentation-digital -environment-solutions-alleviate-burnout/
- Shanafelt TD, West CP, Dyrbye LN, et al. Changes in burnout and satisfaction with work-life integration in physicians during the first 2 years of the COVID-19 pandemic. Mayo Clinic Proceed. 2022;97:2248-2258.
- Herber-Valdez C, Kupesic-Plavsic S. Satisfaction and shortfall of OB-GYN physicians and radiologists. J. Ultrasound Obstet Gynecol. 2021;15:387-392.
- Dyrbye LN, Shanafelt TD, Sinsky CA, et al. Burnout among health care professionals: a call to explore and address this underrecognized threat to safe, high-quality care. National Academy of Medicine Perspectives. Accessed July 5, 2017. https://iuhcpe.org/file_manager/1501524077-Burnout -Among-Health-Care-Professionals-A-Call-to-Explore-and -Address-This-Underrecognized-Threat.pdf
- Olson KD. Physician burnout—a leading indicator of health system performance? Mayo Clinic Proceed. 2017;92: 1608-1611.
- American College of Obstetricians and Gynecologists. Why obgyns are burning out. October 28, 2019. Accessed July 21, 2023. https://www.acog.org/news/news-articles/2019/10/why-ob -gyns-are-burning-out#:~:text=A%202017%20report%20 by%20the,exhaustion%20or%20lack%20of%20motivation
- Peckham C. National physician burnout & depression report 2018. Medscape. January 17, 2018. https://nap. nationalacademies.org/catalog/25521/taking-action -against-clinician-burnout-a-systems-approach-to -professional
- Marsa L. Labor pains: The OB-GYN shortage. AAMC News. Nov. 15, 2018. Accessed July 21, 2023. https://www.aamc.org /news-insights/labor-pains-ob-gyn-shortage
- American College of Obstetricians and Gynecologists. Coping with the stress of medical professional liability litigation. ACOG Committee Opinion. February 2005;309:453454. Accessed July 21, 2023. https://www.acog.org/clinical /clinical-guidance/committee-opinion/articles/2013/01 /coping-with-the-stress-of-medical-professional-liability -litigation
- Reith TP. Burnout in United States healthcare professionals: a narrative review. Cureus. 2018;10:e3681. doi: 10.7759 /cureus.3681
- Han S, Shanafelt TD, Sinsky CA, et al. Estimating the attributable cost of physician burnout in the United States. Ann Intern Med. 2019;4:784-790.
- Sullivan D, Sullivan V, Weatherspoon D, et al. Comparison of nurse burnout, before and during the COVID-19 pandemic. Nurs Clin North Am. 2022;57:79-99. doi: 10.1016 /j.cnur.2021.11.006
- Chandawarkar A, Chaparro JD. Burnout in clinicians. Curr Prob Pediatr Adolesc Health Care. 2021;51:101-104. https ://doi.org/10.1016/j.cppeds.2021.101104
- Brady KJS, Sheldrick RC, Ni P, et al. Examining the measurement equivalence of the Maslach Burnout Inventory across age, gender, and specialty groups in US physicians. J Patient-Reported Outcomes. 2021;5.
- Association of American Medical Colleges. Physician Specialty Data Report—Active Physicians by Sex and Specialty, 2021. Accessed June 19, 2023. https://www.aamc .org/data-reports/workforce/data/active-physicians-sex -specialty-2021
- Association of American Medical Colleges. Physician Specialty Data Report—ACGME Residents and Fellows by Sex and Specialty, 2021. Accessed June 19, 2023. https://www .aamc.org/data-reports/workforce/data/acgme-residents -fellows-sex-and-specialty-2021
- Painter LM, Biggans KA, Turner CT. Risk managementobstetrics and gynecology perspective. Clin Obstet Gynecol. 2023;66:331-341. DOI:10.1097/GRF.0000000000000775
- Darney BG, Boniface E, Liberty A. Assessing the effect of abortion restrictions. Obstetr Gynecol. 2023;141:233-235.
- Heuerman AC, Bessett D, Antommaria AHM, et al. Experiences of reproductive genetic counselors with abortion regulations in Ohio. J Genet Counseling. 2022;31:641-652.
- Brandi K, Gill P. Abortion restrictions threaten all reproductive health care clinicians. Am J Public Health. 2023;113:384-385.
- Rotenstein LS, Torre M, Ramos MA, et al. Prevalence of burnout among physicians: a systematic review. JAMA. 2018;320:1131-1150. doi: 10.1001/jama.2018.1277
- Williamson K, Lank PM, Cheema N, et al. Comparing the Maslach Burnout Inventory to other well-being instruments in emergency medicine residents. J Graduate Med Education. 2018;532-536. DOI: http://dx.doi.org/10.4300 /JGME-D-18-00155.1
- Brady KJS, Sheldrick RC, Ni P, et al. Establishing crosswalks between common measures of burnout in US physicians. J Gen Intern Med. 2022;37:777-784.
- Zhang X, Song Y, Jiang T, et al. Interventions to reduce burnout of physicians and nurses: an overview of systematic reviews and meta-analyses. Medicine (Baltimore). 2020;26:e20992. DOI: 10.1097/MD.0000000000020992
- Scheepers RA, Emke H, Ronald M, et al. The impact of mindfulness-based interventions on doctors’ well-being and performance: a systematic review. Med Education. 2020;54:138-149. https://doi.org/10.1111/medu.14020
- Olson K, Marchalik D, Farley H, et al. Organizational strategies to reduce physician burnout and improve professional fulfillment. Curr Prob Pediatr Adolesc Health Care. 2019;49:12. https://doi.org/10.1016/j.cppeds.2019.100664
- Berry LL, Awdish RLA, Swensen SJ. 5 ways to restore depleted health care workers. Harvard Business Rev. February 11, 2022.
- Sullivan AB, Hersh CM, Rensel M, et al. Leadership inequity, burnout, and lower engagement of women in medicine. J Health Serv Psychol. 2023;49:33-39.
- Hoffman S. Healing the healers: legal remedies for physician burnout. Yale J Health Policy Law Ethics. 2018;18:56-113.
- Federation of State Medical Boards. Physician wellness and burnout: report and recommendations of the workgroup on physician wellness and burnout. (Policy adopted by FSMB). April 2018. Accessed July 21, 2023. https://www.fsmb.org /siteassets/advocacy/policies/policy-on-wellness-and -burnout.pdf
- Robinson C, Kettering C, Sanfilippo JS. Medical malpractice lawsuits. Clin Obstet Gynecol. 2023;66:256-260. DOI: https ://doi.org/10.1097/GRF.0000000000000777
- Gittler GJ, Goldstein EJ. The elements of medical malpractice: an overview. Clin Infect Dis. 1996;23:1152-1155.
- Bal BS. An introduction to medical malpractice in the United States. Clin Orthop Relat Res. 2009;467:339-347.
- Tawfik DS, Profit J, Morgenthaler TI, et al. Physician burnout, well-being, and work unit safety grades in relationship to reported medical errors. Mayo Clinic Proceed. 2018;93: 1571-1580.
- Sundholm B. Elevating physician-patient relationships in the shadow of metric mania. Drexel L Rev. 2020;12:287-330.
- Ghaith S, Campbell RL, Pollock JR, et al. Medical malpractice lawsuits involving trainees in obstetrics and gynecology in the USA. Healthcare. 2022;10:1328.
- Muller TM, Warsi S. Litigation culture causing burnout in American physicians. Trauma Mental Health Report. April 9, 2021.
- Levine AS. Legal 101: Tort law and medical malpractice for physicians. Contemp OBGYN. 2015:60;26-28, 30.
- Regan JJ, Regan WM. Medical malpractice and respondeat superior. Southern Med J. 2002;95.5:545-549. DOI 10.1097/00007611-200295050-00018
- Levin H. Hospital vicarious liability for negligence by independent contractor physicians: new rule for new times. Univ Illinois Law Rev. 2005:1291-1332.
- Darling v Charleston Hospital, 33 Ill. 2d 326, 211 N.E.2d 253 (Ill. 1965).
- Dangel R. Hospital liability for physician malpractice. Ohio State Law J. 1986;47:1077-1098.
- Reffitt v Hajjar, 892 S.W.2d 599, 605 (Ky. Ct. App. 1994).
- McMichael BJ. Malpractice. In Laws of Medicine: Core Legal Aspects for the Healthcare Professional. New York, NY: Springer International; 2022:129-150.
- Occupational Safety and Health Administration. Worker safety in hospitals: caring for our caregivers. Accessed June 8, 2023. https://www.osha.gov/hospitals
- Occupational Safety and Health Administration. Workplace stress. Accessed June 8, 2023. https://www.osha.gov /workplace-stress/understanding-the-problem
- U.S. Surgeon General’s Advisory on Building a Thriving Health Workforce. Addressing health worker burnout. Accessed July 21, 2023. https://www.hhs.gov/sites/default/files/health -worker-wellbeing-advisory.pdf
- Department of Health & Human Services. Biden-Harris administration awards $103 Million in American Rescue Plan funds to reduce burnout and promote mental health and wellness among health care workforce. January 20, 2022. Accessed July 24, 2023. https://www.hhs.gov/about /news/2022/01/20/biden-harris-administration-awards -103-million-american-rescue-plan-funds-reduce-burnout -promote-mental-health-wellness-among-health-care -workforce.html
- Rothstein LF, Irzyk J. Disabilities and the Law. 4th ed. Toronto, Canada: Thompson Reuters; 2023.
- Department of Labor. Guide to disability rights laws. February 28, 2020. Accessed July 24, 2023. https://www .ada.gov/resources/disability-rights-guide/#:~:text=An%20 individual%20with%20a%20disability%20is%20defined%20 by%20the%20ADA,as%20having%20such%20an%20 impairment
- Nadon L, De Beer LT, Morin AJS. Should burnout be conceptualized as a mental disorder? Behavioral Sci. 2022;12:82.
- World Health Organization. Burn-out an “occupational phenomenon”: International Classification of Diseases. May 28, 2019. Accessed July 21, 2023. https://www.who.int/news /item/28-05-2019-burn-out-an-occupational-phenomenon -international-classification-of-diseases
- Hoffman S. Physician burnout: why legal and regulatory systems may need to step in. The Conversation. July 9, 2019. https://theconversation.com/physician-burnout-why-legal -and-regulatory-systems-may-need-to-step-in-119705
- Jha A, Iliff A, Chaoi A, et al. A crisis in healthcare: a call to action on physician burnout. Harvard Global Health Institute. 2019. Accessed July 21, 2023. https://www.massmed.org /Publications/Research,-Studies,-and-Reports/Physician -Burnout-Report-2018/
- Arnsten AF, Shanafelt T. Physician distress and burnout: the neurobiological perspective. Mayo Clin Proceed. 2021;96:763-769.
Physicians have some of the highest rates of burnout among all professions.1 Complicating matters is that clinicians (including residents)2 may avoid seeking treatment out of fear it will affect their license or privileges.3 In this article, we consider burnout in greater detail, as well as ways of successfully addressing the level of burnout in the profession (FIGURE 1), including steps individual practitioners, health care entities, and regulators should consider to reduce burnout and its harmful effects.

How burnout becomes a problem
Six general factors are commonly identified as leading to clinician career dissatisfaction and burnout:4
1. work overload
2. lack of autonomy and control
3. inadequate rewards, financial and otherwise
4. work-home schedules
5. perception of lack of fairness
6. values conflict between the clinician and employer (including a breakdown of professional community).
At the top of the list of causes of burnout is often “administrative and bureaucratic headaches.”5 More specifically, electronic health records (EHRs), including computerized order entry, is commonly cited as a major cause of burnout.6,7 According to some studies, clinicians spend as much as 49% of working time doing clerical work,8 and studies found the extension of work into home life.9
Increased measurement of performance metrics in health care services are a significant contributor to physician burnout.10 These include pressure to see more patients, perform more procedures, and respond quickly to patient requests (eg, through email).7 As we will see, medical malpractice cases, or the risk of such cases, have also played a role in burnout in some medical specialties.11 The pandemic also contributed, at least temporarily, to burnout.12,13
Rates of burnout among physicians are notably higher than among the general population14 or other professions.6 Although physicians have generally entered clinical practice with lower rates of burnout than the general population,15 The American College of Obstetricians and Gynecologists (ACOG) reports that 40% to 75% of ObGyns “experience some form of professional burnout.”16,17 Other source(s) cite that 53% of ObGyns report burnout (TABLE 1).
Code QD85
Burnout is a syndrome conceptualized as resulting from chronic workplace stress that has not been successfully managed. It is characterized by 3 dimensions:
- feelings of energy depletion or exhaustion
- increased mental distance from one’s job, or feelings of negativism or cynicism related to one’s job
- a sense of ineffectiveness and lack of accomplishment. Burn-out refers specifically to phenomena in the occupational context and should not be applied to describe experiences in other areas of life. Exclusions to burnout diagnosis include adjustment disorder, disorders specifically associated with stress, anxiety or fear-related disorders, and mood disorders.
Reference
1. International Classification of Diseases Eleventh Revision (ICD-11). Geneva, Switzerland: World Health Organization; 2022.

Burnout undoubtedly contributes to professionals leaving practice, leading to a significant shortage of ObGyns.18 It also raises several significant legal concerns. Despite the enormity and seriousness of the problem, there is considerable optimism and assurance that the epidemic of burnout is solvable on the individual, specialty, and profession-wide levels. ACOG and other organizations have made suggestions for physicians, the profession, and to health care institutions for reducing burnout.19 This is not to say that solutions are simple or easy for individual professionals or institutions, but they are within the reach of the profession (FIGURE 2).

Suicide among health care professionals is one other concern (TABLE 2)20 and theoretically can stem from burnout, depression, and other psychosocial concerns.

Costs of clinician burnout
Burnout is endemic among health care providers, with numerous studies detailing the professional, emotional, and financial costs. Prior to the pandemic, one analysis of nationwide fiscal costs associated with burnout estimated an annual cost of $4.6B due to physician turnover and reduced clinical hours.21 The COVID-19 epidemic has by all accounts worsened rates of health care worker burnout, particularly for those in high patient-contact positions.22
Female clinicians appear to be differentially affected; in one recent study women reported symptoms of burnout at twice the rate of their male counterparts.23 Whether burnout rates will return to pre-pandemic levels remains an open question, but since burnout is frequently related to one’s own assessment of work-life balance, it is possible that a longer term shift in burnout rates associated with post-pandemic occupational attitudes will be observed.
Combining factors contribute to burnout
Burnout is a universal occupational hazard, but extant data suggest that physicians and other health care providers may be at higher risk. Among physicians, younger age, female gender, and front-line specialty status appear associated with higher burnout rates.24 Given that ObGyn physicians are overwhelmingly female (60% of physicians and 86% of residents),25,26 gender-related burnout factors exist alongside other specific occupational burnout risks. While gender parity has been achieved among health care providers, gender disparities persist in terms of those in leadership positions, compensation, and other factors.22
The smattering of evidence suggesting that ObGyns have higher rates of burnout than many other specialties is understandable given the unique legal challenges confronting ObGyn practice. This may be of special significance because ObGyn malpractice insurance rates are among the highest of all specialties.27 The overall shortage of ObGyns has been exacerbated by the demonstrated negative effects on training and workforce representation stemming from recent legislation that has the effect of criminalizing certain aspects of ObGyn practice;28 for instance, uncertainty regarding abortion regulations.
These negative effects are particularly heightened in states in which the law is in flux or where there are continuing efforts to substantially limit access to abortion. The efforts to increase civil and even criminal penalties related to abortion care challenge ObGyns’ professional practices, as legal rules are frequently changing. In some states, ObGyns may face additional workloads secondary to a flight of ObGyns from restrictive jurisdictions in addition to legal and professional repercussions. In a small study of 19 genetic counselors dealing with restrictive legislation in the state of Ohio,29 increased stress and burnout rates were identified as a consequence of practice uncertainties under this legislation. It is certain that other professionals working in reproductive health care are similarly affected.30
The programs provide individual resources to providers in distress, periodically survey initiatives at Stanford to assess burnout at the organizational level, and provide input designed to spur organizational change to reduce the burden of burnout. Ways that they build community and connections include:
- Live Story Rounds events (as told by Stanford Medicine physicians)
- Commensality Groups (facilitated small discussion groups built around tested evidence)
- Aim to increase sense of connection and collegiality among physicians and build comradery at work
- CME-accredited physician wellness forum, including annual doctor’s day events
Continue to: Assessment of burnout...
Assessment of burnout
Numerous scales for the assessment of burnout exist. Of these, the 22-item Maslach Burnout Inventory (MBI) is the best studied. The MBI is a well-investigated tool for assessing burnout. The MBI consists of 3 major subscales measuring overall burnout, emotional exhaustion, depersonalization, and low personal accomplishment. It exists in numerous forms. For instance, the MBI-HSS (MP), adapted for medical personnel, is available. However, the most commonly used form for assessing burnout in clinicians is the MBI-HHS (Human Services Survey); approximately 85% of all burnout studies examined in a recent meta-analysis used this survey version.31 As those authors commented, while burnout is a recognized phenomenon, a great deal of variability in study design, interpretation of subscale scores, and sample selection makes generalizations regarding burnout difficult to assess.
The MBI in various forms has been extensively used over the past 40 years to assess burnout amongst physicians and physicians in training. While not the only instrument designed to measure such factors, it is by far the most prevalent. Williamson and colleagues32 compared the MBI with several other measures of quality of life and found good correlation between the various instruments used, a finding replicated by other studies.33 Brady and colleagues compared item responses to the Stanford Professional Fulfillment Index and the Min-Z Single-item Burnout scale (a 1-item screening measure) to MBI’s Emotional Exhaustion and Depersonalization subscales. Basing their findings on a survey of more than 1,300 physicians, they found that all analyzed scales were significantly correlated with such adverse outcomes as depression, distress, or intent to leave the profession.
It is important to note that most surveys of clinician burnout were conducted prior to the pandemic. While the psychometric analyses of the MBI and other scales are likely still germane, observed rates of clinician burnout have likely increased. Thus, comparisons of pre- and post-pandemic studies should factor in an increase in the incidence and prevalence of burnout.
Management strategies
In general, there are several interventions for managing burnout34:
- individual-focused (including self-care and communications-skills workshops)
- mindfulness training
- yoga
- meditation
- organizational/structural (workload reduction, schedule realignment, teamwork training, and group-delivered stress management interventions)
- combination(s) of the above.
There is little evidence to suggest that any particular individual intervention (whether delivered in individual or group-based formats) is superior to any other in treating clinician burnout. A recent analysis of 24 studies employing mindfulness-based interventions demonstrated generally positive results for such interventions.35 Other studies have also found general support for mindfulness-based interventions, although mindfulness is often integrated with other stress-reduction techniques, such as meditation, yoga, and communication skills. Such interventions are nonspecific but generally effective.
An accumulation of evidence to date suggests that a combination of individual and organizational interventions is most effective in combatting clinician burnout. No individual intervention can be successful without addressing root causes, such as overscheduling, lack of organizational support, and the effect of restrictive legislation on practice.
Several large teaching hospitals have established programs to address physician and health care provider burnout. Notable among these is the Stanford University School of Medicine’s WellMD and WellPhD programs (https://wellmd.stanford.edu/about.html). These programs were described by Olson and colleagues36 as using a model focused on practice efficiency, organizational culture, and personal resilience to enhance physicians’ well-being. (See “Aspects of the WellMD and WellPhD programs from Stanford University.”)
A growing number of institutions have established burnout programs to support physicians experiencing work/life imbalances and other aspects of burnout.37 In general, these share common features of assessment, individual and/or group intervention, and organizational change. Fear of repercussion may be one factor preventing physicians from seeking individual treatment for burnout.38 Importantly, they emphasize the need for professional confidentiality when offering treatment to patients within organizational settings. Those authors also reported that a focus on organizational engagement may be an important factor in addressing burnout in female physicians, as they tend to report lower levels of organizational engagement.
Continue to: Legal considerations...
Legal considerations
Until recently, physician burnout “received little notice in the legal literature.”39 Although there have been burnout legal consequences in the past, the legal issues are now becoming more visible.40
Medical malpractice
A well-documented consequence of burnout is an increase in errors.14 Medical errors, of course, are at the heart of malpractice claims. Technically, malpractice is medical or professional negligence. It is the breach of a duty owed by the physician, or other provider, or organization (defendant) to the patient, which causes injury to the plaintiff/patient.41
“Medical error” is generally a meaningful deviation from the “standard of care” or accepted medical practice.42 Many medical errors do not cause injury to the patient; in those cases, the negligence does not result in liability. In instances in which the negligence causes harm, the clinician and health care facility may be subject to liability for that injury. Fortunately, however, for a variety of reasons, most harmful medical errors do not result in a medical malpractice claim or lawsuit. The absence of a good clinician-patient relationship is likely associated with an increased inclination of a patient to file a malpractice action.43Clinician burnout may, therefore, contribute to increased malpractice claims in two ways. First, burnout likely leads to increased medical errors, perhaps because burnout is associated with lower concentration, inattention, reduced cognitive vigilance, and fatigue.8,44 It may also lead to less time with patients, reduced patient empathy, and lower patient rapport, which may make injured patients more likely to file a claim or lawsuit.45 Because the relationshipbetween burnout and medical error is bidirectional, malpractice claims tend to increase burnout, which increases error. Given the time it takes to resolve most malpractice claims, the uncertainty of medical malpractice may be especially stressful for health care providers.46,47
Burnout is not a mitigating factor in malpractice. Our sympathies may go out to a professional suffering from burnout, but it does not excuse or reduce liability—it may, indeed, be an aggravating factor. Clinicians who can diagnose burnout and know its negative consequences but fail to deal with their own burnout may be demonstrating negligence if there has been harm to a patient related to the burnout.48
Institutional or corporate liability to patients
Health care institutions have obligations to avoid injury to patients. Just as poorly maintained medical equipment may harm patients, so may burned-out professionals. Therefore, institutions have some obligation to supervise and avoid the increased risks to patients posed by professionals suffering from burnout.
Respondeat superior and institutional negligence. Institutional liability may arise in two ways, the first through agency, or respondeat superior. That is, if the physician or other professional is an employee (or similar agent) of the health care institution, that institution is generally responsible for the physician’s negligence during the employment.49 Even if the physician is not an employee (for example, an independent contractor providing care or using the hospital facilities), the health care facility may be liable for the physician’s negligence.50 Liability may occur, for example, if the health care facility was aware that the physician was engaged in careless practice or was otherwise a risk to patients but the facility did not take steps to avoid those risks.51 The basis for liability is that the health care organization owes a duty to patients to take reasonable care to ensure that its facilities are not used to injure patients negligently.52 Just as it must take care that unqualified physicians are not granted privileges to practice, it also must take reasonable steps to protect patients when it is aware (through nurses or other agents) of a physician’s negligent practice.
In one case, for example, the court found liability where a staff member had “severe” burnout in a physician’s office and failed to read fetal monitoring strips. The physician was found negligent for relying on the staff member who was obviously making errors in interpretation of fetal distress.53
Continue to: Legal obligations of health care organizations to physicians and others...
Legal obligations of health care organizations to physicians and others
In addition to obligations to patients, health care organizations may have obligations to employees (and others) at risk for injury. For example, assume a patient is diagnosed with a highly contagious disease. The health care organization would be obligated to warn, and take reasonable steps to protect, the staff (employees and independent contractors) from being harmed from exposure to the disease. This principle may apply to coworkers of employees with significant burnout, thereby presenting a danger in the workplace. The liability issue is more difficult for employees experiencing job-related burnout themselves. Organizations generally compensate injured employees through no-fault workers’ compensation (an insurance-like system); for independent contractors, the liability is usually through a tort claim (negligence).54
In modern times, a focus has been on preventing those injuries, not just providing compensation after injuries have occurred. Notably, federal and state occupational health and safety laws (particularly the Occupational Safety and Health Administration [OSHA]) require most organizations (including those employing health care providers) to take steps to mitigate various kinds of worker injuries.55
Although these worker protections have commonly been applied to hospitals and other health care providers, burnout has not traditionally been a significant concern in federal or state OSHA enforcement. For example, no formal federal OSHA regulations govern work-related burnout. Regulators, including OSHA, are increasingly interested in burnout that may affect many employees. OSHA has several recommendations for reducing health care work burnout.56 The Surgeon General has expressed similar concerns.57 The federal government recently allocated $103 million from the American Rescue Plan to address burnout among health care workers.58 Also, OSHA appears to be increasing its oversight of healthcare-institution-worker injuries.55
Is burnout a “disability”?
The federal Americans with Disabilities Act (ADA) and similar state laws prohibit discrimination based on disability.59 A disability is defined as a “physical or mental impairment that substantially limits one or more major life activities” or “perceived as having such an impairment.”60 The initial issue is whether burnout is a “mental impairment.” As noted earlier, it is not officially a “medical condition.”61 To date, the United Nations has classified it as an “occupational phenomenon.”62 It may, therefore, not qualify under the ADA, even if it “interferes with a major life activity.” There is, however, some movement toward defining burnout as a mental condition. Even if defined as a disability, there would still be legal issues of how severe it must be to qualify as a disability and the proper accommodation. Apart from the legal definition of an ADA disability, as a practical matter it likely is in the best interest of health care facilities to provide accommodations that reduce burnout. A number of strategies to decrease the incidence of burnout include the role of health care systems (FIGURE 2).
In conclusion we look at several things that can be done to “treat” or reduce burnout. That effort requires the cooperation of physicians and other providers, health care facilities, training programs, licensing authorities, and professional organizations. See suggestions below.
Conclusion
There are many excellent suggestions for reducing burnout and improving patient care and practitioner satisfaction.63-65 We conclude with a summary of some of these suggestions for individual practitioners, health care organizations, the profession, and licensing. It is worth remembering, however, that it will require the efforts of each area to reduce burnout substantially.
For practitioners:
- Engage in quality coaching/therapy on mindfulness and stress management.
- Practice self-care, including exercise and relaxation techniques.
- Make work-life balance a priority.
- Take opportunities for collegial social and professional discussions.
- Prioritize (and periodically assess) your own professional satisfaction and burnout risk.
- Smile—enjoy a sense of humor (endorphins and cortisol).
For health care organizations:
- Urgently work with vendors and regulators to revise electronic health records to reduce their substantial impact on burnout.
- Reduce physicians’ time on clerical and administrative tasks (eg, by enhancing the use of quality AI, scribes, and automated notes from appointments. (This may increase the time they spend with patients.) Eliminate “pajama-time” charting.
- Provide various kinds of confidential professional counseling, therapy, and support related to burnout prevention and treatment, and avoid any penalty or stigma related to their use.
- Provide reasonable flexibility in scheduling.
- Routinely provide employees with information about burnout prevention and services.
- Appoint a wellness officer with authority to ensure the organization maximizes its prevention and treatment services.
- Constantly seek input from practitioners on how to improve the atmosphere for practice to maximize patient care and practitioner satisfaction.
- Provide ample professional and social opportunities for discussing and learning about work-life balance, resilience, intellectual stimulation, and career development.
For regulators, licensors, and professional organizations:
- Work with health care organizations and EHR vendors to substantially reduce the complexity, physician effort, and stress associated with those record systems. Streamlining should, in the future, be part of formally certifying EHR systems.
- Reduce the administrative burden on physicians by modifying complex regulations and using AI and other technology to the extent possible to obtain necessary reimbursement information.
- Eliminate unnecessary data gathering that requires practitioner time or attention.
- Licensing, educational, and certifying bodies should eliminate any questions regarding the diagnosis or treatment of mental health and focus on current (or very recent) impairments.
- Seek funding for research on burnout prevention and treatment.
Dr. H is a 58-year-old ObGyn who, after completing residency, went into solo practice. The practice grew, and Dr. H found it increasingly more challenging to cover, especially the obstetrics sector. Dr. H then merged the practice with a group of 3 other ObGyns. Their practice expanded, and began recruiting recent residency graduates. In time, the practice was bought out by the local hospital health care system. Dr. H was faced with complying with the rules and regulations of that health care system. The electronic health record (EHR) component proved challenging, as did the restrictions on staff hiring (and firing), but Dr. H did receive a paycheck each month and complied with it all. The health care system administrators had clear financial targets Dr. H was to meet each quarter, which created additional pressure. Dr. H used to love being an OB and providing excellent care for every patient, but that sense of accomplishment was being lost.
Dr. H increasingly found it difficult to focus because of mind wandering, especially in the operating room (OR). Thoughts occurred about retirement, the current challenges imposed by “the new way of practicing medicine” (more focused on financial productivity restraints and reimbursement), and EHR challenges. Then Dr. H’s attention would return to the OR case at hand. All of this resulted in considerable stress and emotional exhaustion, and sometimes a sense of being disconnected. A few times, colleagues or nurses had asked Dr. H if everything was “okay,” or if a break would help. Dr. H made more small errors than usual, but Dr. H’s self-assessment was “doing an adequate job.” Patient satisfaction scores (collected routinely by the health care system) declined over the last 9 months.
Six months ago, Dr. H finished doing a laparoscopic total hysterectomy and bilateral salpingo-oophorectomy and got into the right uterine artery. The estimated blood loss was 3,500 mL. Using minimally invasive techniques, Dr. H identified the bleeder and, with monopolar current, got everything under control. The patient went to the post-anesthesia care unit, and all appeared to be in order. Her vital signs were stable, and she was discharged home the same day.
The patient presented 1 week later with lower abdominal and right flank pain. Dr. H addressed the problem in the emergency department and admitted the patient for further evaluation and urology consultation. The right ureter was damaged and obstructed; ultimately, the urologist performed a psoas bladder hitch. The patient recovered slowly, lost several weeks of work, experienced significant pain, and had other disruptions and costs. Additional medical care related to the surgery is ongoing. A health care system committee asked Dr. H to explain the problem. Over the last 6 months, Dr. H’s frustration with practice and being tired and disconnected have increased.
Dr. H has received a letter from a law firm saying that he and the health care system are being sued for malpractice focused on an iatrogenic ureter injury. The letter names two very reputable experts who are prepared to testify that the patient’s injury resulted from clear negligence. Dr. H has told the malpractice carrier absolutely not to settle this case—it is “a sham— without merit.” The health care system has asked Dr. H to take a “burnout test.”
Legal considerations
Dr. H exhibits relatively clear signs of professional burnout. The fact that there was a bad outcome while Dr. H was experiencing burnout is not proof of negligence (or, breach of duty of care to the patient). Nor is it a defense or mitigation to any malpractice that occurred.
In the malpractice case, the plaintiff will have the burden of proving that Dr. H’s treatment was negligent in that it fell below the standard of care. Even if it was a medical error, the question is whether it was negligence. If the patient/plaintiff, using expert witnesses, can prove that Dr. H fell below the standard of care that caused injury, Dr. H may be liable for the resulting extra costs, loss of income, and pain and suffering resulting from the negligent care.
The health care system likely will also be responsible for Dr. H’s negligence, either through respondeat superior (for example, if Dr. H is an employee) or for its own negligence. The case for its negligence is that the nurses and assistants had repeatedly seen him making errors and becoming disengaged (to the extent that they asked Dr. H if “everything is okay” or if a break would help). Furthermore, Dr. H’s patient satisfaction scores have been declining for several months. The plaintiff will argue that Dr. H exhibited classic burnout symptoms with the attendant risks of medical errors. However, the health care system did not take action to protect patients or to assist Dr. H. In short, one way or another, there is some likelihood that the health care system may also be liable if patient injuries are found to have been caused by negligence.
At this point, the health care system also faces the question of how to work with Dr. H in the future. The most pressing question is whether or not to allow Dr. H to continue practicing. If, as it appears, Dr. H is dealing with burnout, the pressure of the malpractice claim could well increase the probability of other medical mistakes. The institution has asked Dr. H to take a burnout test, but it is unclear where things go if the test (as likely) demonstrates significant burnout. This is a counseling and human relations question, at least as much as a legal issue, and the institution should probably proceed in that way—which is, trying to understand and support Dr. H and determining what can be done to address the burnout. At the same time, the system must reasonably assess Dr. H’s fitness to continue practicing as the matters are resolved. Almost everyone shares the goal to provide every individual and corporate opportunity for Dr. H to deal with burnout issues and return to successful practice.
Dr. H will be represented in the malpractice case by counsel provided through the insurance carrier. However, Dr. H would be well advised to retain a trusted and knowledgeable personal attorney. For example, the instruction not to consider settlement is likely misguided, but Dr. H needs to talk with an attorney that Dr. H has chosen and trusts. In addition, the attorney can help guide Dr. H through a rational process of dealing with the health care system, putting the practice in order, and considering the options for the future.
The health care system should reconsider its processes to deal with burnout to ensure the quality of care, patient satisfaction, professional retention, and economic stability. Several burnoutresponse programs have had success in achieving these goals.
What’s the Verdict?
Dr. H received good mental health, legal, and professional advice. As a result, an out of court settlement was reached following pretrial discovery. Dr. H has continued consultation regarding burnout and has returned to productive practice.
Physicians have some of the highest rates of burnout among all professions.1 Complicating matters is that clinicians (including residents)2 may avoid seeking treatment out of fear it will affect their license or privileges.3 In this article, we consider burnout in greater detail, as well as ways of successfully addressing the level of burnout in the profession (FIGURE 1), including steps individual practitioners, health care entities, and regulators should consider to reduce burnout and its harmful effects.

How burnout becomes a problem
Six general factors are commonly identified as leading to clinician career dissatisfaction and burnout:4
1. work overload
2. lack of autonomy and control
3. inadequate rewards, financial and otherwise
4. work-home schedules
5. perception of lack of fairness
6. values conflict between the clinician and employer (including a breakdown of professional community).
At the top of the list of causes of burnout is often “administrative and bureaucratic headaches.”5 More specifically, electronic health records (EHRs), including computerized order entry, is commonly cited as a major cause of burnout.6,7 According to some studies, clinicians spend as much as 49% of working time doing clerical work,8 and studies found the extension of work into home life.9
Increased measurement of performance metrics in health care services are a significant contributor to physician burnout.10 These include pressure to see more patients, perform more procedures, and respond quickly to patient requests (eg, through email).7 As we will see, medical malpractice cases, or the risk of such cases, have also played a role in burnout in some medical specialties.11 The pandemic also contributed, at least temporarily, to burnout.12,13
Rates of burnout among physicians are notably higher than among the general population14 or other professions.6 Although physicians have generally entered clinical practice with lower rates of burnout than the general population,15 The American College of Obstetricians and Gynecologists (ACOG) reports that 40% to 75% of ObGyns “experience some form of professional burnout.”16,17 Other source(s) cite that 53% of ObGyns report burnout (TABLE 1).
Code QD85
Burnout is a syndrome conceptualized as resulting from chronic workplace stress that has not been successfully managed. It is characterized by 3 dimensions:
- feelings of energy depletion or exhaustion
- increased mental distance from one’s job, or feelings of negativism or cynicism related to one’s job
- a sense of ineffectiveness and lack of accomplishment. Burn-out refers specifically to phenomena in the occupational context and should not be applied to describe experiences in other areas of life. Exclusions to burnout diagnosis include adjustment disorder, disorders specifically associated with stress, anxiety or fear-related disorders, and mood disorders.
Reference
1. International Classification of Diseases Eleventh Revision (ICD-11). Geneva, Switzerland: World Health Organization; 2022.

Burnout undoubtedly contributes to professionals leaving practice, leading to a significant shortage of ObGyns.18 It also raises several significant legal concerns. Despite the enormity and seriousness of the problem, there is considerable optimism and assurance that the epidemic of burnout is solvable on the individual, specialty, and profession-wide levels. ACOG and other organizations have made suggestions for physicians, the profession, and to health care institutions for reducing burnout.19 This is not to say that solutions are simple or easy for individual professionals or institutions, but they are within the reach of the profession (FIGURE 2).

Suicide among health care professionals is one other concern (TABLE 2)20 and theoretically can stem from burnout, depression, and other psychosocial concerns.

Costs of clinician burnout
Burnout is endemic among health care providers, with numerous studies detailing the professional, emotional, and financial costs. Prior to the pandemic, one analysis of nationwide fiscal costs associated with burnout estimated an annual cost of $4.6B due to physician turnover and reduced clinical hours.21 The COVID-19 epidemic has by all accounts worsened rates of health care worker burnout, particularly for those in high patient-contact positions.22
Female clinicians appear to be differentially affected; in one recent study women reported symptoms of burnout at twice the rate of their male counterparts.23 Whether burnout rates will return to pre-pandemic levels remains an open question, but since burnout is frequently related to one’s own assessment of work-life balance, it is possible that a longer term shift in burnout rates associated with post-pandemic occupational attitudes will be observed.
Combining factors contribute to burnout
Burnout is a universal occupational hazard, but extant data suggest that physicians and other health care providers may be at higher risk. Among physicians, younger age, female gender, and front-line specialty status appear associated with higher burnout rates.24 Given that ObGyn physicians are overwhelmingly female (60% of physicians and 86% of residents),25,26 gender-related burnout factors exist alongside other specific occupational burnout risks. While gender parity has been achieved among health care providers, gender disparities persist in terms of those in leadership positions, compensation, and other factors.22
The smattering of evidence suggesting that ObGyns have higher rates of burnout than many other specialties is understandable given the unique legal challenges confronting ObGyn practice. This may be of special significance because ObGyn malpractice insurance rates are among the highest of all specialties.27 The overall shortage of ObGyns has been exacerbated by the demonstrated negative effects on training and workforce representation stemming from recent legislation that has the effect of criminalizing certain aspects of ObGyn practice;28 for instance, uncertainty regarding abortion regulations.
These negative effects are particularly heightened in states in which the law is in flux or where there are continuing efforts to substantially limit access to abortion. The efforts to increase civil and even criminal penalties related to abortion care challenge ObGyns’ professional practices, as legal rules are frequently changing. In some states, ObGyns may face additional workloads secondary to a flight of ObGyns from restrictive jurisdictions in addition to legal and professional repercussions. In a small study of 19 genetic counselors dealing with restrictive legislation in the state of Ohio,29 increased stress and burnout rates were identified as a consequence of practice uncertainties under this legislation. It is certain that other professionals working in reproductive health care are similarly affected.30
The programs provide individual resources to providers in distress, periodically survey initiatives at Stanford to assess burnout at the organizational level, and provide input designed to spur organizational change to reduce the burden of burnout. Ways that they build community and connections include:
- Live Story Rounds events (as told by Stanford Medicine physicians)
- Commensality Groups (facilitated small discussion groups built around tested evidence)
- Aim to increase sense of connection and collegiality among physicians and build comradery at work
- CME-accredited physician wellness forum, including annual doctor’s day events
Continue to: Assessment of burnout...
Assessment of burnout
Numerous scales for the assessment of burnout exist. Of these, the 22-item Maslach Burnout Inventory (MBI) is the best studied. The MBI is a well-investigated tool for assessing burnout. The MBI consists of 3 major subscales measuring overall burnout, emotional exhaustion, depersonalization, and low personal accomplishment. It exists in numerous forms. For instance, the MBI-HSS (MP), adapted for medical personnel, is available. However, the most commonly used form for assessing burnout in clinicians is the MBI-HHS (Human Services Survey); approximately 85% of all burnout studies examined in a recent meta-analysis used this survey version.31 As those authors commented, while burnout is a recognized phenomenon, a great deal of variability in study design, interpretation of subscale scores, and sample selection makes generalizations regarding burnout difficult to assess.
The MBI in various forms has been extensively used over the past 40 years to assess burnout amongst physicians and physicians in training. While not the only instrument designed to measure such factors, it is by far the most prevalent. Williamson and colleagues32 compared the MBI with several other measures of quality of life and found good correlation between the various instruments used, a finding replicated by other studies.33 Brady and colleagues compared item responses to the Stanford Professional Fulfillment Index and the Min-Z Single-item Burnout scale (a 1-item screening measure) to MBI’s Emotional Exhaustion and Depersonalization subscales. Basing their findings on a survey of more than 1,300 physicians, they found that all analyzed scales were significantly correlated with such adverse outcomes as depression, distress, or intent to leave the profession.
It is important to note that most surveys of clinician burnout were conducted prior to the pandemic. While the psychometric analyses of the MBI and other scales are likely still germane, observed rates of clinician burnout have likely increased. Thus, comparisons of pre- and post-pandemic studies should factor in an increase in the incidence and prevalence of burnout.
Management strategies
In general, there are several interventions for managing burnout34:
- individual-focused (including self-care and communications-skills workshops)
- mindfulness training
- yoga
- meditation
- organizational/structural (workload reduction, schedule realignment, teamwork training, and group-delivered stress management interventions)
- combination(s) of the above.
There is little evidence to suggest that any particular individual intervention (whether delivered in individual or group-based formats) is superior to any other in treating clinician burnout. A recent analysis of 24 studies employing mindfulness-based interventions demonstrated generally positive results for such interventions.35 Other studies have also found general support for mindfulness-based interventions, although mindfulness is often integrated with other stress-reduction techniques, such as meditation, yoga, and communication skills. Such interventions are nonspecific but generally effective.
An accumulation of evidence to date suggests that a combination of individual and organizational interventions is most effective in combatting clinician burnout. No individual intervention can be successful without addressing root causes, such as overscheduling, lack of organizational support, and the effect of restrictive legislation on practice.
Several large teaching hospitals have established programs to address physician and health care provider burnout. Notable among these is the Stanford University School of Medicine’s WellMD and WellPhD programs (https://wellmd.stanford.edu/about.html). These programs were described by Olson and colleagues36 as using a model focused on practice efficiency, organizational culture, and personal resilience to enhance physicians’ well-being. (See “Aspects of the WellMD and WellPhD programs from Stanford University.”)
A growing number of institutions have established burnout programs to support physicians experiencing work/life imbalances and other aspects of burnout.37 In general, these share common features of assessment, individual and/or group intervention, and organizational change. Fear of repercussion may be one factor preventing physicians from seeking individual treatment for burnout.38 Importantly, they emphasize the need for professional confidentiality when offering treatment to patients within organizational settings. Those authors also reported that a focus on organizational engagement may be an important factor in addressing burnout in female physicians, as they tend to report lower levels of organizational engagement.
Continue to: Legal considerations...
Legal considerations
Until recently, physician burnout “received little notice in the legal literature.”39 Although there have been burnout legal consequences in the past, the legal issues are now becoming more visible.40
Medical malpractice
A well-documented consequence of burnout is an increase in errors.14 Medical errors, of course, are at the heart of malpractice claims. Technically, malpractice is medical or professional negligence. It is the breach of a duty owed by the physician, or other provider, or organization (defendant) to the patient, which causes injury to the plaintiff/patient.41
“Medical error” is generally a meaningful deviation from the “standard of care” or accepted medical practice.42 Many medical errors do not cause injury to the patient; in those cases, the negligence does not result in liability. In instances in which the negligence causes harm, the clinician and health care facility may be subject to liability for that injury. Fortunately, however, for a variety of reasons, most harmful medical errors do not result in a medical malpractice claim or lawsuit. The absence of a good clinician-patient relationship is likely associated with an increased inclination of a patient to file a malpractice action.43Clinician burnout may, therefore, contribute to increased malpractice claims in two ways. First, burnout likely leads to increased medical errors, perhaps because burnout is associated with lower concentration, inattention, reduced cognitive vigilance, and fatigue.8,44 It may also lead to less time with patients, reduced patient empathy, and lower patient rapport, which may make injured patients more likely to file a claim or lawsuit.45 Because the relationshipbetween burnout and medical error is bidirectional, malpractice claims tend to increase burnout, which increases error. Given the time it takes to resolve most malpractice claims, the uncertainty of medical malpractice may be especially stressful for health care providers.46,47
Burnout is not a mitigating factor in malpractice. Our sympathies may go out to a professional suffering from burnout, but it does not excuse or reduce liability—it may, indeed, be an aggravating factor. Clinicians who can diagnose burnout and know its negative consequences but fail to deal with their own burnout may be demonstrating negligence if there has been harm to a patient related to the burnout.48
Institutional or corporate liability to patients
Health care institutions have obligations to avoid injury to patients. Just as poorly maintained medical equipment may harm patients, so may burned-out professionals. Therefore, institutions have some obligation to supervise and avoid the increased risks to patients posed by professionals suffering from burnout.
Respondeat superior and institutional negligence. Institutional liability may arise in two ways, the first through agency, or respondeat superior. That is, if the physician or other professional is an employee (or similar agent) of the health care institution, that institution is generally responsible for the physician’s negligence during the employment.49 Even if the physician is not an employee (for example, an independent contractor providing care or using the hospital facilities), the health care facility may be liable for the physician’s negligence.50 Liability may occur, for example, if the health care facility was aware that the physician was engaged in careless practice or was otherwise a risk to patients but the facility did not take steps to avoid those risks.51 The basis for liability is that the health care organization owes a duty to patients to take reasonable care to ensure that its facilities are not used to injure patients negligently.52 Just as it must take care that unqualified physicians are not granted privileges to practice, it also must take reasonable steps to protect patients when it is aware (through nurses or other agents) of a physician’s negligent practice.
In one case, for example, the court found liability where a staff member had “severe” burnout in a physician’s office and failed to read fetal monitoring strips. The physician was found negligent for relying on the staff member who was obviously making errors in interpretation of fetal distress.53
Continue to: Legal obligations of health care organizations to physicians and others...
Legal obligations of health care organizations to physicians and others
In addition to obligations to patients, health care organizations may have obligations to employees (and others) at risk for injury. For example, assume a patient is diagnosed with a highly contagious disease. The health care organization would be obligated to warn, and take reasonable steps to protect, the staff (employees and independent contractors) from being harmed from exposure to the disease. This principle may apply to coworkers of employees with significant burnout, thereby presenting a danger in the workplace. The liability issue is more difficult for employees experiencing job-related burnout themselves. Organizations generally compensate injured employees through no-fault workers’ compensation (an insurance-like system); for independent contractors, the liability is usually through a tort claim (negligence).54
In modern times, a focus has been on preventing those injuries, not just providing compensation after injuries have occurred. Notably, federal and state occupational health and safety laws (particularly the Occupational Safety and Health Administration [OSHA]) require most organizations (including those employing health care providers) to take steps to mitigate various kinds of worker injuries.55
Although these worker protections have commonly been applied to hospitals and other health care providers, burnout has not traditionally been a significant concern in federal or state OSHA enforcement. For example, no formal federal OSHA regulations govern work-related burnout. Regulators, including OSHA, are increasingly interested in burnout that may affect many employees. OSHA has several recommendations for reducing health care work burnout.56 The Surgeon General has expressed similar concerns.57 The federal government recently allocated $103 million from the American Rescue Plan to address burnout among health care workers.58 Also, OSHA appears to be increasing its oversight of healthcare-institution-worker injuries.55
Is burnout a “disability”?
The federal Americans with Disabilities Act (ADA) and similar state laws prohibit discrimination based on disability.59 A disability is defined as a “physical or mental impairment that substantially limits one or more major life activities” or “perceived as having such an impairment.”60 The initial issue is whether burnout is a “mental impairment.” As noted earlier, it is not officially a “medical condition.”61 To date, the United Nations has classified it as an “occupational phenomenon.”62 It may, therefore, not qualify under the ADA, even if it “interferes with a major life activity.” There is, however, some movement toward defining burnout as a mental condition. Even if defined as a disability, there would still be legal issues of how severe it must be to qualify as a disability and the proper accommodation. Apart from the legal definition of an ADA disability, as a practical matter it likely is in the best interest of health care facilities to provide accommodations that reduce burnout. A number of strategies to decrease the incidence of burnout include the role of health care systems (FIGURE 2).
In conclusion we look at several things that can be done to “treat” or reduce burnout. That effort requires the cooperation of physicians and other providers, health care facilities, training programs, licensing authorities, and professional organizations. See suggestions below.
Conclusion
There are many excellent suggestions for reducing burnout and improving patient care and practitioner satisfaction.63-65 We conclude with a summary of some of these suggestions for individual practitioners, health care organizations, the profession, and licensing. It is worth remembering, however, that it will require the efforts of each area to reduce burnout substantially.
For practitioners:
- Engage in quality coaching/therapy on mindfulness and stress management.
- Practice self-care, including exercise and relaxation techniques.
- Make work-life balance a priority.
- Take opportunities for collegial social and professional discussions.
- Prioritize (and periodically assess) your own professional satisfaction and burnout risk.
- Smile—enjoy a sense of humor (endorphins and cortisol).
For health care organizations:
- Urgently work with vendors and regulators to revise electronic health records to reduce their substantial impact on burnout.
- Reduce physicians’ time on clerical and administrative tasks (eg, by enhancing the use of quality AI, scribes, and automated notes from appointments. (This may increase the time they spend with patients.) Eliminate “pajama-time” charting.
- Provide various kinds of confidential professional counseling, therapy, and support related to burnout prevention and treatment, and avoid any penalty or stigma related to their use.
- Provide reasonable flexibility in scheduling.
- Routinely provide employees with information about burnout prevention and services.
- Appoint a wellness officer with authority to ensure the organization maximizes its prevention and treatment services.
- Constantly seek input from practitioners on how to improve the atmosphere for practice to maximize patient care and practitioner satisfaction.
- Provide ample professional and social opportunities for discussing and learning about work-life balance, resilience, intellectual stimulation, and career development.
For regulators, licensors, and professional organizations:
- Work with health care organizations and EHR vendors to substantially reduce the complexity, physician effort, and stress associated with those record systems. Streamlining should, in the future, be part of formally certifying EHR systems.
- Reduce the administrative burden on physicians by modifying complex regulations and using AI and other technology to the extent possible to obtain necessary reimbursement information.
- Eliminate unnecessary data gathering that requires practitioner time or attention.
- Licensing, educational, and certifying bodies should eliminate any questions regarding the diagnosis or treatment of mental health and focus on current (or very recent) impairments.
- Seek funding for research on burnout prevention and treatment.
Dr. H is a 58-year-old ObGyn who, after completing residency, went into solo practice. The practice grew, and Dr. H found it increasingly more challenging to cover, especially the obstetrics sector. Dr. H then merged the practice with a group of 3 other ObGyns. Their practice expanded, and began recruiting recent residency graduates. In time, the practice was bought out by the local hospital health care system. Dr. H was faced with complying with the rules and regulations of that health care system. The electronic health record (EHR) component proved challenging, as did the restrictions on staff hiring (and firing), but Dr. H did receive a paycheck each month and complied with it all. The health care system administrators had clear financial targets Dr. H was to meet each quarter, which created additional pressure. Dr. H used to love being an OB and providing excellent care for every patient, but that sense of accomplishment was being lost.
Dr. H increasingly found it difficult to focus because of mind wandering, especially in the operating room (OR). Thoughts occurred about retirement, the current challenges imposed by “the new way of practicing medicine” (more focused on financial productivity restraints and reimbursement), and EHR challenges. Then Dr. H’s attention would return to the OR case at hand. All of this resulted in considerable stress and emotional exhaustion, and sometimes a sense of being disconnected. A few times, colleagues or nurses had asked Dr. H if everything was “okay,” or if a break would help. Dr. H made more small errors than usual, but Dr. H’s self-assessment was “doing an adequate job.” Patient satisfaction scores (collected routinely by the health care system) declined over the last 9 months.
Six months ago, Dr. H finished doing a laparoscopic total hysterectomy and bilateral salpingo-oophorectomy and got into the right uterine artery. The estimated blood loss was 3,500 mL. Using minimally invasive techniques, Dr. H identified the bleeder and, with monopolar current, got everything under control. The patient went to the post-anesthesia care unit, and all appeared to be in order. Her vital signs were stable, and she was discharged home the same day.
The patient presented 1 week later with lower abdominal and right flank pain. Dr. H addressed the problem in the emergency department and admitted the patient for further evaluation and urology consultation. The right ureter was damaged and obstructed; ultimately, the urologist performed a psoas bladder hitch. The patient recovered slowly, lost several weeks of work, experienced significant pain, and had other disruptions and costs. Additional medical care related to the surgery is ongoing. A health care system committee asked Dr. H to explain the problem. Over the last 6 months, Dr. H’s frustration with practice and being tired and disconnected have increased.
Dr. H has received a letter from a law firm saying that he and the health care system are being sued for malpractice focused on an iatrogenic ureter injury. The letter names two very reputable experts who are prepared to testify that the patient’s injury resulted from clear negligence. Dr. H has told the malpractice carrier absolutely not to settle this case—it is “a sham— without merit.” The health care system has asked Dr. H to take a “burnout test.”
Legal considerations
Dr. H exhibits relatively clear signs of professional burnout. The fact that there was a bad outcome while Dr. H was experiencing burnout is not proof of negligence (or, breach of duty of care to the patient). Nor is it a defense or mitigation to any malpractice that occurred.
In the malpractice case, the plaintiff will have the burden of proving that Dr. H’s treatment was negligent in that it fell below the standard of care. Even if it was a medical error, the question is whether it was negligence. If the patient/plaintiff, using expert witnesses, can prove that Dr. H fell below the standard of care that caused injury, Dr. H may be liable for the resulting extra costs, loss of income, and pain and suffering resulting from the negligent care.
The health care system likely will also be responsible for Dr. H’s negligence, either through respondeat superior (for example, if Dr. H is an employee) or for its own negligence. The case for its negligence is that the nurses and assistants had repeatedly seen him making errors and becoming disengaged (to the extent that they asked Dr. H if “everything is okay” or if a break would help). Furthermore, Dr. H’s patient satisfaction scores have been declining for several months. The plaintiff will argue that Dr. H exhibited classic burnout symptoms with the attendant risks of medical errors. However, the health care system did not take action to protect patients or to assist Dr. H. In short, one way or another, there is some likelihood that the health care system may also be liable if patient injuries are found to have been caused by negligence.
At this point, the health care system also faces the question of how to work with Dr. H in the future. The most pressing question is whether or not to allow Dr. H to continue practicing. If, as it appears, Dr. H is dealing with burnout, the pressure of the malpractice claim could well increase the probability of other medical mistakes. The institution has asked Dr. H to take a burnout test, but it is unclear where things go if the test (as likely) demonstrates significant burnout. This is a counseling and human relations question, at least as much as a legal issue, and the institution should probably proceed in that way—which is, trying to understand and support Dr. H and determining what can be done to address the burnout. At the same time, the system must reasonably assess Dr. H’s fitness to continue practicing as the matters are resolved. Almost everyone shares the goal to provide every individual and corporate opportunity for Dr. H to deal with burnout issues and return to successful practice.
Dr. H will be represented in the malpractice case by counsel provided through the insurance carrier. However, Dr. H would be well advised to retain a trusted and knowledgeable personal attorney. For example, the instruction not to consider settlement is likely misguided, but Dr. H needs to talk with an attorney that Dr. H has chosen and trusts. In addition, the attorney can help guide Dr. H through a rational process of dealing with the health care system, putting the practice in order, and considering the options for the future.
The health care system should reconsider its processes to deal with burnout to ensure the quality of care, patient satisfaction, professional retention, and economic stability. Several burnoutresponse programs have had success in achieving these goals.
What’s the Verdict?
Dr. H received good mental health, legal, and professional advice. As a result, an out of court settlement was reached following pretrial discovery. Dr. H has continued consultation regarding burnout and has returned to productive practice.
- Shanafelt TD, West CP, Sinsky C, et al. Changes in burnout and satisfaction with work-life integration in physicians and the general US working population between 2011 and 2017. Mayo Clinic Proceed. 2019;94:1681-1694.
- Smith R, Rayburn W. Burnout in obstetrician-gynecologists. Its prevalence, identification, prevention, and reversal. Obstet Gynecol Clin North Am. 2021;48:231-245. https://doi. org/10.1016/j.ogc.2021.06.003
- Patti MG, Schlottmann F, Sarr MG. The problem of burnout among surgeons. JAMA Surg. 2018;153:403-404. doi:10.1001 /jamasurg.2018.0047
- Carrau D, Janis JE. Physician burnout: solutions for individuals and organizations. Plastic and Reconstructive Surgery Global Open. 2021;91-97.
- Southwick R. The key to fixing physician burnout is the workplace not the worker. Contemporary Ob/Gyn. March 13, 2023.
- Patel RS, Bachu R, Adikey A, et al. Factors related to physician burnout and its consequences: a review. Behav Sciences. 2018;8:98.
- Melnick ER, Dyrbye LN, Sinsky CA, et al. The association between perceived electronic health record usability and professional burnout among US physicians. Mayo Clinic Proceed. 2020;95:476-487.
- Shanafelt TD, Dyrbye LN, West CP. Addressing physician burnout: the way forward. JAMA. 2017;317:901-902. doi:10.1001/jama.2017.0076
- Ommaya AK, Cipriano PF, Hoyt DB, et al. Care-centered clinical documentation in the digital environment: Solutions to alleviate burnout. National Academy of Medicine Perspectives. 2018.
- Hartzband P, Groopman J. Physician burnout, interrupted. N Engl J Med. 2020;382:2485-2487. Discussion Paper, National Academy of Medicine. Accessed July 21, 2023. https://nam .edu/care
- Ji YD, Robertson FC, Patel NA, et al. Assessment of risk factors for suicide among US health care professionals. JAMA Surg. 2020;155:713-721. centered-clinical-documentation-digital -environment-solutions-alleviate-burnout/
- Shanafelt TD, West CP, Dyrbye LN, et al. Changes in burnout and satisfaction with work-life integration in physicians during the first 2 years of the COVID-19 pandemic. Mayo Clinic Proceed. 2022;97:2248-2258.
- Herber-Valdez C, Kupesic-Plavsic S. Satisfaction and shortfall of OB-GYN physicians and radiologists. J. Ultrasound Obstet Gynecol. 2021;15:387-392.
- Dyrbye LN, Shanafelt TD, Sinsky CA, et al. Burnout among health care professionals: a call to explore and address this underrecognized threat to safe, high-quality care. National Academy of Medicine Perspectives. Accessed July 5, 2017. https://iuhcpe.org/file_manager/1501524077-Burnout -Among-Health-Care-Professionals-A-Call-to-Explore-and -Address-This-Underrecognized-Threat.pdf
- Olson KD. Physician burnout—a leading indicator of health system performance? Mayo Clinic Proceed. 2017;92: 1608-1611.
- American College of Obstetricians and Gynecologists. Why obgyns are burning out. October 28, 2019. Accessed July 21, 2023. https://www.acog.org/news/news-articles/2019/10/why-ob -gyns-are-burning-out#:~:text=A%202017%20report%20 by%20the,exhaustion%20or%20lack%20of%20motivation
- Peckham C. National physician burnout & depression report 2018. Medscape. January 17, 2018. https://nap. nationalacademies.org/catalog/25521/taking-action -against-clinician-burnout-a-systems-approach-to -professional
- Marsa L. Labor pains: The OB-GYN shortage. AAMC News. Nov. 15, 2018. Accessed July 21, 2023. https://www.aamc.org /news-insights/labor-pains-ob-gyn-shortage
- American College of Obstetricians and Gynecologists. Coping with the stress of medical professional liability litigation. ACOG Committee Opinion. February 2005;309:453454. Accessed July 21, 2023. https://www.acog.org/clinical /clinical-guidance/committee-opinion/articles/2013/01 /coping-with-the-stress-of-medical-professional-liability -litigation
- Reith TP. Burnout in United States healthcare professionals: a narrative review. Cureus. 2018;10:e3681. doi: 10.7759 /cureus.3681
- Han S, Shanafelt TD, Sinsky CA, et al. Estimating the attributable cost of physician burnout in the United States. Ann Intern Med. 2019;4:784-790.
- Sullivan D, Sullivan V, Weatherspoon D, et al. Comparison of nurse burnout, before and during the COVID-19 pandemic. Nurs Clin North Am. 2022;57:79-99. doi: 10.1016 /j.cnur.2021.11.006
- Chandawarkar A, Chaparro JD. Burnout in clinicians. Curr Prob Pediatr Adolesc Health Care. 2021;51:101-104. https ://doi.org/10.1016/j.cppeds.2021.101104
- Brady KJS, Sheldrick RC, Ni P, et al. Examining the measurement equivalence of the Maslach Burnout Inventory across age, gender, and specialty groups in US physicians. J Patient-Reported Outcomes. 2021;5.
- Association of American Medical Colleges. Physician Specialty Data Report—Active Physicians by Sex and Specialty, 2021. Accessed June 19, 2023. https://www.aamc .org/data-reports/workforce/data/active-physicians-sex -specialty-2021
- Association of American Medical Colleges. Physician Specialty Data Report—ACGME Residents and Fellows by Sex and Specialty, 2021. Accessed June 19, 2023. https://www .aamc.org/data-reports/workforce/data/acgme-residents -fellows-sex-and-specialty-2021
- Painter LM, Biggans KA, Turner CT. Risk managementobstetrics and gynecology perspective. Clin Obstet Gynecol. 2023;66:331-341. DOI:10.1097/GRF.0000000000000775
- Darney BG, Boniface E, Liberty A. Assessing the effect of abortion restrictions. Obstetr Gynecol. 2023;141:233-235.
- Heuerman AC, Bessett D, Antommaria AHM, et al. Experiences of reproductive genetic counselors with abortion regulations in Ohio. J Genet Counseling. 2022;31:641-652.
- Brandi K, Gill P. Abortion restrictions threaten all reproductive health care clinicians. Am J Public Health. 2023;113:384-385.
- Rotenstein LS, Torre M, Ramos MA, et al. Prevalence of burnout among physicians: a systematic review. JAMA. 2018;320:1131-1150. doi: 10.1001/jama.2018.1277
- Williamson K, Lank PM, Cheema N, et al. Comparing the Maslach Burnout Inventory to other well-being instruments in emergency medicine residents. J Graduate Med Education. 2018;532-536. DOI: http://dx.doi.org/10.4300 /JGME-D-18-00155.1
- Brady KJS, Sheldrick RC, Ni P, et al. Establishing crosswalks between common measures of burnout in US physicians. J Gen Intern Med. 2022;37:777-784.
- Zhang X, Song Y, Jiang T, et al. Interventions to reduce burnout of physicians and nurses: an overview of systematic reviews and meta-analyses. Medicine (Baltimore). 2020;26:e20992. DOI: 10.1097/MD.0000000000020992
- Scheepers RA, Emke H, Ronald M, et al. The impact of mindfulness-based interventions on doctors’ well-being and performance: a systematic review. Med Education. 2020;54:138-149. https://doi.org/10.1111/medu.14020
- Olson K, Marchalik D, Farley H, et al. Organizational strategies to reduce physician burnout and improve professional fulfillment. Curr Prob Pediatr Adolesc Health Care. 2019;49:12. https://doi.org/10.1016/j.cppeds.2019.100664
- Berry LL, Awdish RLA, Swensen SJ. 5 ways to restore depleted health care workers. Harvard Business Rev. February 11, 2022.
- Sullivan AB, Hersh CM, Rensel M, et al. Leadership inequity, burnout, and lower engagement of women in medicine. J Health Serv Psychol. 2023;49:33-39.
- Hoffman S. Healing the healers: legal remedies for physician burnout. Yale J Health Policy Law Ethics. 2018;18:56-113.
- Federation of State Medical Boards. Physician wellness and burnout: report and recommendations of the workgroup on physician wellness and burnout. (Policy adopted by FSMB). April 2018. Accessed July 21, 2023. https://www.fsmb.org /siteassets/advocacy/policies/policy-on-wellness-and -burnout.pdf
- Robinson C, Kettering C, Sanfilippo JS. Medical malpractice lawsuits. Clin Obstet Gynecol. 2023;66:256-260. DOI: https ://doi.org/10.1097/GRF.0000000000000777
- Gittler GJ, Goldstein EJ. The elements of medical malpractice: an overview. Clin Infect Dis. 1996;23:1152-1155.
- Bal BS. An introduction to medical malpractice in the United States. Clin Orthop Relat Res. 2009;467:339-347.
- Tawfik DS, Profit J, Morgenthaler TI, et al. Physician burnout, well-being, and work unit safety grades in relationship to reported medical errors. Mayo Clinic Proceed. 2018;93: 1571-1580.
- Sundholm B. Elevating physician-patient relationships in the shadow of metric mania. Drexel L Rev. 2020;12:287-330.
- Ghaith S, Campbell RL, Pollock JR, et al. Medical malpractice lawsuits involving trainees in obstetrics and gynecology in the USA. Healthcare. 2022;10:1328.
- Muller TM, Warsi S. Litigation culture causing burnout in American physicians. Trauma Mental Health Report. April 9, 2021.
- Levine AS. Legal 101: Tort law and medical malpractice for physicians. Contemp OBGYN. 2015:60;26-28, 30.
- Regan JJ, Regan WM. Medical malpractice and respondeat superior. Southern Med J. 2002;95.5:545-549. DOI 10.1097/00007611-200295050-00018
- Levin H. Hospital vicarious liability for negligence by independent contractor physicians: new rule for new times. Univ Illinois Law Rev. 2005:1291-1332.
- Darling v Charleston Hospital, 33 Ill. 2d 326, 211 N.E.2d 253 (Ill. 1965).
- Dangel R. Hospital liability for physician malpractice. Ohio State Law J. 1986;47:1077-1098.
- Reffitt v Hajjar, 892 S.W.2d 599, 605 (Ky. Ct. App. 1994).
- McMichael BJ. Malpractice. In Laws of Medicine: Core Legal Aspects for the Healthcare Professional. New York, NY: Springer International; 2022:129-150.
- Occupational Safety and Health Administration. Worker safety in hospitals: caring for our caregivers. Accessed June 8, 2023. https://www.osha.gov/hospitals
- Occupational Safety and Health Administration. Workplace stress. Accessed June 8, 2023. https://www.osha.gov /workplace-stress/understanding-the-problem
- U.S. Surgeon General’s Advisory on Building a Thriving Health Workforce. Addressing health worker burnout. Accessed July 21, 2023. https://www.hhs.gov/sites/default/files/health -worker-wellbeing-advisory.pdf
- Department of Health & Human Services. Biden-Harris administration awards $103 Million in American Rescue Plan funds to reduce burnout and promote mental health and wellness among health care workforce. January 20, 2022. Accessed July 24, 2023. https://www.hhs.gov/about /news/2022/01/20/biden-harris-administration-awards -103-million-american-rescue-plan-funds-reduce-burnout -promote-mental-health-wellness-among-health-care -workforce.html
- Rothstein LF, Irzyk J. Disabilities and the Law. 4th ed. Toronto, Canada: Thompson Reuters; 2023.
- Department of Labor. Guide to disability rights laws. February 28, 2020. Accessed July 24, 2023. https://www .ada.gov/resources/disability-rights-guide/#:~:text=An%20 individual%20with%20a%20disability%20is%20defined%20 by%20the%20ADA,as%20having%20such%20an%20 impairment
- Nadon L, De Beer LT, Morin AJS. Should burnout be conceptualized as a mental disorder? Behavioral Sci. 2022;12:82.
- World Health Organization. Burn-out an “occupational phenomenon”: International Classification of Diseases. May 28, 2019. Accessed July 21, 2023. https://www.who.int/news /item/28-05-2019-burn-out-an-occupational-phenomenon -international-classification-of-diseases
- Hoffman S. Physician burnout: why legal and regulatory systems may need to step in. The Conversation. July 9, 2019. https://theconversation.com/physician-burnout-why-legal -and-regulatory-systems-may-need-to-step-in-119705
- Jha A, Iliff A, Chaoi A, et al. A crisis in healthcare: a call to action on physician burnout. Harvard Global Health Institute. 2019. Accessed July 21, 2023. https://www.massmed.org /Publications/Research,-Studies,-and-Reports/Physician -Burnout-Report-2018/
- Arnsten AF, Shanafelt T. Physician distress and burnout: the neurobiological perspective. Mayo Clin Proceed. 2021;96:763-769.
- Shanafelt TD, West CP, Sinsky C, et al. Changes in burnout and satisfaction with work-life integration in physicians and the general US working population between 2011 and 2017. Mayo Clinic Proceed. 2019;94:1681-1694.
- Smith R, Rayburn W. Burnout in obstetrician-gynecologists. Its prevalence, identification, prevention, and reversal. Obstet Gynecol Clin North Am. 2021;48:231-245. https://doi. org/10.1016/j.ogc.2021.06.003
- Patti MG, Schlottmann F, Sarr MG. The problem of burnout among surgeons. JAMA Surg. 2018;153:403-404. doi:10.1001 /jamasurg.2018.0047
- Carrau D, Janis JE. Physician burnout: solutions for individuals and organizations. Plastic and Reconstructive Surgery Global Open. 2021;91-97.
- Southwick R. The key to fixing physician burnout is the workplace not the worker. Contemporary Ob/Gyn. March 13, 2023.
- Patel RS, Bachu R, Adikey A, et al. Factors related to physician burnout and its consequences: a review. Behav Sciences. 2018;8:98.
- Melnick ER, Dyrbye LN, Sinsky CA, et al. The association between perceived electronic health record usability and professional burnout among US physicians. Mayo Clinic Proceed. 2020;95:476-487.
- Shanafelt TD, Dyrbye LN, West CP. Addressing physician burnout: the way forward. JAMA. 2017;317:901-902. doi:10.1001/jama.2017.0076
- Ommaya AK, Cipriano PF, Hoyt DB, et al. Care-centered clinical documentation in the digital environment: Solutions to alleviate burnout. National Academy of Medicine Perspectives. 2018.
- Hartzband P, Groopman J. Physician burnout, interrupted. N Engl J Med. 2020;382:2485-2487. Discussion Paper, National Academy of Medicine. Accessed July 21, 2023. https://nam .edu/care
- Ji YD, Robertson FC, Patel NA, et al. Assessment of risk factors for suicide among US health care professionals. JAMA Surg. 2020;155:713-721. centered-clinical-documentation-digital -environment-solutions-alleviate-burnout/
- Shanafelt TD, West CP, Dyrbye LN, et al. Changes in burnout and satisfaction with work-life integration in physicians during the first 2 years of the COVID-19 pandemic. Mayo Clinic Proceed. 2022;97:2248-2258.
- Herber-Valdez C, Kupesic-Plavsic S. Satisfaction and shortfall of OB-GYN physicians and radiologists. J. Ultrasound Obstet Gynecol. 2021;15:387-392.
- Dyrbye LN, Shanafelt TD, Sinsky CA, et al. Burnout among health care professionals: a call to explore and address this underrecognized threat to safe, high-quality care. National Academy of Medicine Perspectives. Accessed July 5, 2017. https://iuhcpe.org/file_manager/1501524077-Burnout -Among-Health-Care-Professionals-A-Call-to-Explore-and -Address-This-Underrecognized-Threat.pdf
- Olson KD. Physician burnout—a leading indicator of health system performance? Mayo Clinic Proceed. 2017;92: 1608-1611.
- American College of Obstetricians and Gynecologists. Why obgyns are burning out. October 28, 2019. Accessed July 21, 2023. https://www.acog.org/news/news-articles/2019/10/why-ob -gyns-are-burning-out#:~:text=A%202017%20report%20 by%20the,exhaustion%20or%20lack%20of%20motivation
- Peckham C. National physician burnout & depression report 2018. Medscape. January 17, 2018. https://nap. nationalacademies.org/catalog/25521/taking-action -against-clinician-burnout-a-systems-approach-to -professional
- Marsa L. Labor pains: The OB-GYN shortage. AAMC News. Nov. 15, 2018. Accessed July 21, 2023. https://www.aamc.org /news-insights/labor-pains-ob-gyn-shortage
- American College of Obstetricians and Gynecologists. Coping with the stress of medical professional liability litigation. ACOG Committee Opinion. February 2005;309:453454. Accessed July 21, 2023. https://www.acog.org/clinical /clinical-guidance/committee-opinion/articles/2013/01 /coping-with-the-stress-of-medical-professional-liability -litigation
- Reith TP. Burnout in United States healthcare professionals: a narrative review. Cureus. 2018;10:e3681. doi: 10.7759 /cureus.3681
- Han S, Shanafelt TD, Sinsky CA, et al. Estimating the attributable cost of physician burnout in the United States. Ann Intern Med. 2019;4:784-790.
- Sullivan D, Sullivan V, Weatherspoon D, et al. Comparison of nurse burnout, before and during the COVID-19 pandemic. Nurs Clin North Am. 2022;57:79-99. doi: 10.1016 /j.cnur.2021.11.006
- Chandawarkar A, Chaparro JD. Burnout in clinicians. Curr Prob Pediatr Adolesc Health Care. 2021;51:101-104. https ://doi.org/10.1016/j.cppeds.2021.101104
- Brady KJS, Sheldrick RC, Ni P, et al. Examining the measurement equivalence of the Maslach Burnout Inventory across age, gender, and specialty groups in US physicians. J Patient-Reported Outcomes. 2021;5.
- Association of American Medical Colleges. Physician Specialty Data Report—Active Physicians by Sex and Specialty, 2021. Accessed June 19, 2023. https://www.aamc .org/data-reports/workforce/data/active-physicians-sex -specialty-2021
- Association of American Medical Colleges. Physician Specialty Data Report—ACGME Residents and Fellows by Sex and Specialty, 2021. Accessed June 19, 2023. https://www .aamc.org/data-reports/workforce/data/acgme-residents -fellows-sex-and-specialty-2021
- Painter LM, Biggans KA, Turner CT. Risk managementobstetrics and gynecology perspective. Clin Obstet Gynecol. 2023;66:331-341. DOI:10.1097/GRF.0000000000000775
- Darney BG, Boniface E, Liberty A. Assessing the effect of abortion restrictions. Obstetr Gynecol. 2023;141:233-235.
- Heuerman AC, Bessett D, Antommaria AHM, et al. Experiences of reproductive genetic counselors with abortion regulations in Ohio. J Genet Counseling. 2022;31:641-652.
- Brandi K, Gill P. Abortion restrictions threaten all reproductive health care clinicians. Am J Public Health. 2023;113:384-385.
- Rotenstein LS, Torre M, Ramos MA, et al. Prevalence of burnout among physicians: a systematic review. JAMA. 2018;320:1131-1150. doi: 10.1001/jama.2018.1277
- Williamson K, Lank PM, Cheema N, et al. Comparing the Maslach Burnout Inventory to other well-being instruments in emergency medicine residents. J Graduate Med Education. 2018;532-536. DOI: http://dx.doi.org/10.4300 /JGME-D-18-00155.1
- Brady KJS, Sheldrick RC, Ni P, et al. Establishing crosswalks between common measures of burnout in US physicians. J Gen Intern Med. 2022;37:777-784.
- Zhang X, Song Y, Jiang T, et al. Interventions to reduce burnout of physicians and nurses: an overview of systematic reviews and meta-analyses. Medicine (Baltimore). 2020;26:e20992. DOI: 10.1097/MD.0000000000020992
- Scheepers RA, Emke H, Ronald M, et al. The impact of mindfulness-based interventions on doctors’ well-being and performance: a systematic review. Med Education. 2020;54:138-149. https://doi.org/10.1111/medu.14020
- Olson K, Marchalik D, Farley H, et al. Organizational strategies to reduce physician burnout and improve professional fulfillment. Curr Prob Pediatr Adolesc Health Care. 2019;49:12. https://doi.org/10.1016/j.cppeds.2019.100664
- Berry LL, Awdish RLA, Swensen SJ. 5 ways to restore depleted health care workers. Harvard Business Rev. February 11, 2022.
- Sullivan AB, Hersh CM, Rensel M, et al. Leadership inequity, burnout, and lower engagement of women in medicine. J Health Serv Psychol. 2023;49:33-39.
- Hoffman S. Healing the healers: legal remedies for physician burnout. Yale J Health Policy Law Ethics. 2018;18:56-113.
- Federation of State Medical Boards. Physician wellness and burnout: report and recommendations of the workgroup on physician wellness and burnout. (Policy adopted by FSMB). April 2018. Accessed July 21, 2023. https://www.fsmb.org /siteassets/advocacy/policies/policy-on-wellness-and -burnout.pdf
- Robinson C, Kettering C, Sanfilippo JS. Medical malpractice lawsuits. Clin Obstet Gynecol. 2023;66:256-260. DOI: https ://doi.org/10.1097/GRF.0000000000000777
- Gittler GJ, Goldstein EJ. The elements of medical malpractice: an overview. Clin Infect Dis. 1996;23:1152-1155.
- Bal BS. An introduction to medical malpractice in the United States. Clin Orthop Relat Res. 2009;467:339-347.
- Tawfik DS, Profit J, Morgenthaler TI, et al. Physician burnout, well-being, and work unit safety grades in relationship to reported medical errors. Mayo Clinic Proceed. 2018;93: 1571-1580.
- Sundholm B. Elevating physician-patient relationships in the shadow of metric mania. Drexel L Rev. 2020;12:287-330.
- Ghaith S, Campbell RL, Pollock JR, et al. Medical malpractice lawsuits involving trainees in obstetrics and gynecology in the USA. Healthcare. 2022;10:1328.
- Muller TM, Warsi S. Litigation culture causing burnout in American physicians. Trauma Mental Health Report. April 9, 2021.
- Levine AS. Legal 101: Tort law and medical malpractice for physicians. Contemp OBGYN. 2015:60;26-28, 30.
- Regan JJ, Regan WM. Medical malpractice and respondeat superior. Southern Med J. 2002;95.5:545-549. DOI 10.1097/00007611-200295050-00018
- Levin H. Hospital vicarious liability for negligence by independent contractor physicians: new rule for new times. Univ Illinois Law Rev. 2005:1291-1332.
- Darling v Charleston Hospital, 33 Ill. 2d 326, 211 N.E.2d 253 (Ill. 1965).
- Dangel R. Hospital liability for physician malpractice. Ohio State Law J. 1986;47:1077-1098.
- Reffitt v Hajjar, 892 S.W.2d 599, 605 (Ky. Ct. App. 1994).
- McMichael BJ. Malpractice. In Laws of Medicine: Core Legal Aspects for the Healthcare Professional. New York, NY: Springer International; 2022:129-150.
- Occupational Safety and Health Administration. Worker safety in hospitals: caring for our caregivers. Accessed June 8, 2023. https://www.osha.gov/hospitals
- Occupational Safety and Health Administration. Workplace stress. Accessed June 8, 2023. https://www.osha.gov /workplace-stress/understanding-the-problem
- U.S. Surgeon General’s Advisory on Building a Thriving Health Workforce. Addressing health worker burnout. Accessed July 21, 2023. https://www.hhs.gov/sites/default/files/health -worker-wellbeing-advisory.pdf
- Department of Health & Human Services. Biden-Harris administration awards $103 Million in American Rescue Plan funds to reduce burnout and promote mental health and wellness among health care workforce. January 20, 2022. Accessed July 24, 2023. https://www.hhs.gov/about /news/2022/01/20/biden-harris-administration-awards -103-million-american-rescue-plan-funds-reduce-burnout -promote-mental-health-wellness-among-health-care -workforce.html
- Rothstein LF, Irzyk J. Disabilities and the Law. 4th ed. Toronto, Canada: Thompson Reuters; 2023.
- Department of Labor. Guide to disability rights laws. February 28, 2020. Accessed July 24, 2023. https://www .ada.gov/resources/disability-rights-guide/#:~:text=An%20 individual%20with%20a%20disability%20is%20defined%20 by%20the%20ADA,as%20having%20such%20an%20 impairment
- Nadon L, De Beer LT, Morin AJS. Should burnout be conceptualized as a mental disorder? Behavioral Sci. 2022;12:82.
- World Health Organization. Burn-out an “occupational phenomenon”: International Classification of Diseases. May 28, 2019. Accessed July 21, 2023. https://www.who.int/news /item/28-05-2019-burn-out-an-occupational-phenomenon -international-classification-of-diseases
- Hoffman S. Physician burnout: why legal and regulatory systems may need to step in. The Conversation. July 9, 2019. https://theconversation.com/physician-burnout-why-legal -and-regulatory-systems-may-need-to-step-in-119705
- Jha A, Iliff A, Chaoi A, et al. A crisis in healthcare: a call to action on physician burnout. Harvard Global Health Institute. 2019. Accessed July 21, 2023. https://www.massmed.org /Publications/Research,-Studies,-and-Reports/Physician -Burnout-Report-2018/
- Arnsten AF, Shanafelt T. Physician distress and burnout: the neurobiological perspective. Mayo Clin Proceed. 2021;96:763-769.
Imaging Tools for Noninvasive Hair Assessment
New imaging tools along with adaptations to existing technologies have been emerging in recent years, with the potential to improve hair diagnostics and treatment monitoring. We provide an overview of 4 noninvasive hair imaging technologies: global photography, trichoscopy, reflectance confocal microscopy (RCM), and optical coherence tomography (OCT). For each instrument, we discuss current and future applications in clinical practice and research along with advantages and disadvantages.
Global Photography
Global photography allows for the analysis of hair growth, volume, distribution, and density through serial standardized photographs.1 Global photography was first introduced for hair growth studies in 1987 and soon after was used for hair and scalp assessments in finasteride clinical trials.2
Hair Assessment—Washed, dried, and combed hair, without hair product, are required for accurate imaging; wet conditions increase reflection and promote hair clumping, thus revealing more scalp and depicting the patient as having less hair.1 Headshots are taken from short distances and use stereotactic positioning devices to create 4 global views: vertex, midline, frontal, and temporal.3 Stereotactic positioning involves fixing the patient’s chin and forehead as well as mounting the camera and flash device to ensure proper magnification. These adjustments ensure lighting remains consistent throughout consecutive study visits.4 Various grading scales are available for use in hair growth clinical studies to increase objectivity in the analysis of serial global photographs. A blinded evaluator should assess the before and after photographs to limit experimenter bias. Global photography often is combined with quantitative software analysis for improved detection of hair changes.1
Advancements—Growing interest in improving global photography has resulted in various application-based, artificial intelligence (AI)–mediated tools to simplify photograph collection and analysis. For instance, new hair analysis software utilizes AI algorithms to account for facial features in determining the optimal angle for capturing global photographs (Figure 1), which simplifies the generation of global photography images through smartphone applications and obviates the need for additional stereotactic positioning equipment.5,6

Limitations—Clinicians should be aware of global photography’s requirements for consistency in lighting, camera settings, film, and image processing, which can limit the accuracy of hair assessment over time if not replicated correctly.7,8 Emerging global photography software has helped to overcome some of these limitations.
Global photography is less precise when a patient’s hair loss is less than 50%, as it is difficult to discern subtle hair changes. Thus, global photography provides limited utility in assessing minimal to moderate hair loss.9 Currently, global photography largely functions as an adjunct tool for other hair analysis methods rather than as a stand-alone tool.
Trichoscopy
Trichoscopy (also known as dermoscopy of the hair and scalp) may be performed with a manual dermoscope (with 10× magnification) or a digital videodermatoscope (up to 1000× magnification).10-12 Unlike global photography, trichoscopy provides a detailed structural analysis of hair shafts, follicular openings, and perifollicular and interfollicular areas.13 Kinoshita-Ise and Sachdeva13 provided an in-depth, updated review of trichoscopy terminology with their definitions and associated conditions (with prevalence), which should be referenced when performing trichoscopic examination.
Hair Assessment—Trichoscopic assessment begins with inspection of follicular openings (also referred to as “dots”), which vary in color depending on the material filling them—degrading keratinocytes, keratin, sebaceous debris, melanin, or fractured hairs.13 The structure of hair shafts also is examined, showing broken hairs, short vellus hairs, and comma hairs, among others. Perifollicular areas are examined for scale, erythema, blue-gray dots, and whitish halos. Interfollicular areas are examined for pigment pattern as well as vascularization, which often presents in a looping configuration under dermoscopy. A combination of dot colorization, hair shaft structure, and perifollicular and interfollicular findings inform diagnostic algorithms of hair and scalp conditions. For example, central centrifugal cicatricial alopecia, the most common alopecia seen in Black women, has been associated with a combination of honeycomb pigment pattern, perifollicular whitish halo, pinpoint white dots, white patches, and perifollicular erythema.13
Advantages—Perhaps the most useful feature of trichoscopy is its ability to translate visualized features into simple diagnostic algorithms. For instance, if the clinician has diagnosed the patient with noncicatricial alopecia, they would next focus on dot colors. With black dots, the next step would be to determine whether the hairs are tapered or coiled, and so on. This systematic approach enables the clinician to narrow possible diagnoses.2 An additional advantage of trichoscopy is that it examines large surface areas noninvasively as compared to hair-pull tests and scalp biopsy.14,15 Trichoscopy allows temporal comparisons of the same area for disease and treatment monitoring with more diagnostic detail than global photography.16 Trichoscopy also is useful in selecting biopsy locations by discerning and avoiding areas of scar tissue.17
Limitations—Diagnosis via the trichoscopy algorithm is limiting because it is not comprehensive of all hair and scalp disease.18 Additionally, many pathologies exhibit overlapping follicular and interfollicular patterning. For example, almost all subtypes of scarring alopecia present with hair loss and scarred follicles once they have progressed to advanced stages. Further studies should identify more specific patterns of hair and scalp pathologies, which could then be incorporated into a diagnostic algorithm.13
Advancements—The advent of hair analysis software has expanded the role of videodermoscopy by rapidly quantifying hair growth parameters such as hair count, follicular density, and follicular diameter, as well as interfollicular distances (Figure 2).14,17 Vellus and terminal hairs are differentiated according to their thickness and length.17 Moreover, the software can analyze the same area of the scalp over time by either virtual tattoos, semipermanent markings, or precise location measurements, increasing intra- and interclass correlation. The rate of hair growth, hair shedding, and parameters of anagen and telogen hairs can be studied by a method termed phototrichogram whereby a transitional area of hair loss and normal hair growth is identified and trimmed to less than 1 mm from the skin surface.19 A baseline photograph is taken using videodermoscopy. After approximately 3 days, the identical region is photographed and compared with the initial image to observe changes in the hair. Software programs can distinguish the growing hair as anagen and nongrowing hair as telogen, calculating the anagen-to-telogen ratio as well as hair growth rate, which are essential measurements in hair research and clinical studies. Software programs have replaced laborious and time-consuming manual hair counts and have rapidly grown in popularity in evaluating patterned hair loss.
Reflectance Confocal Microscopy
Reflectance confocal microscopy is a noninvasive imaging tool that visualizes skin and its appendages at near-histologic resolution (lateral resolution of 0.5–1 μm). It produces grayscale horizontal images that can be taken at levels ranging from the stratum corneum to the superficial papillary dermis, corresponding to a depth of approximately 100 to 150 µm. Thus, a hair follicle can be imaged starting from the follicular ostia down to the reachable papillary dermis (Figure 3).20 Image contrast is provided by differences in the size and refractive indices of cellular organelles.21,22 There are 2 commercially available RCM devices: VivaScope 1500 and VivaScope 3000 (Caliber Imaging & Diagnostics, Inc).
VivaScope 1500, a wide-probe microscope, requires the attachment of a plastic window to the desired imaging area. The plastic window is lined with medical adhesive tape to prevent movement during imaging. The adhesive tape can pull on hair upon removal, which is not ideal for patients with existing hair loss. Additionally, the image quality of VivaSope 1500 is best in flat areas and areas where hair is shaved.20,23,24 Despite these disadvantages, VivaScope 1500 has successfully shown utility in research studies, which suggests that these obstacles can be overcome by experienced users. The handheld VivaScope 3000 is ergonomically designed and suitable for curved surfaces such as the scalp, with the advantage of not requiring any adhesive. However, the images acquired from the VivaScope 3000 cover a smaller surface area.
Structures Visualized—Structures distinguished with RCM include keratinocytes, melanocytes, inflammatory cells, hair follicles, hair shafts, adnexal infundibular epithelium, blood vessels, fibroblasts, and collagen.23 Real-time visualization of blood flow also can be seen.
Applications of RCM—Reflectance confocal microscopy has been used to study scalp discoid lupus, lichen planopilaris, frontal fibrosing alopecia, folliculitis decalvans, chemotherapy-induced alopecia (CIA), alopecia areata, and androgenetic alopecia. Diagnostic RCM criteria for such alopecias have been developed based on their correspondence to histopathology. An RCM study of classic lichen planopilaris and frontal fibrosing alopecia identified features of epidermal disarray, infundibular hyperkeratosis, inflammatory cells, pigment incontinence, perifollicular fibrosis, bandlike scarring, melanophages in the dermis, dilated blood vessels, basal layer vacuolar degeneration, and necrotic keratinocytes.25 Pigment incontinence in the superficial epidermis, perifollicular lichenoid inflammation, and hyperkeratosis were characteristic RCM features of early-stage lichen planopilaris, while perifollicular fibrosis and dilated blood vessels were characteristic RCM features of late-stage disease. The ability of RCM features to distinguish different stages of lichen planopilaris shows its potential in treating early disease and preventing irreversible hair loss.
Differentiating between scarring and nonscarring alopecia also is possible through RCM. The presence of periadnexal, epidermal, and dermal inflammatory cells, in addition to periadnexal sclerosis, are defining RCM features of scarring alopecia.26 These features are absent in nonscarring alopecias. Reflectance confocal microscopy additionally has been shown to be useful in the treatment monitoring of lichen planopilaris and discoid lupus erythematosus.20 Independent reviewers, blinded to the patients’ identities, were able to characterize and follow features of these scarring alopecias by RCM. The assessed RCM features were comparable to those observed by histopathologic evaluation: epidermal disarray, spongiosis, exocytosis of inflammatory cells in the epidermis, interface dermatitis, peri- and intra-adnexal infiltration of inflammatory cells, dilated vessels in the dermis, dermal infiltration of inflammatory cells and melanophages, and dermal sclerosis. A reduction in inflammatory cells across multiple skin layers and at the level of the adnexal epithelium correlated with clinical response to treatment. Reflectance confocal microscopy also was able to detect recurrence of inflammation in cases where treatment had been interrupted before clinical signs of disease recurrence were evident. The authors thus concluded that RCM’s sensitivity can guide timing of treatment and avoid delays in starting or restarting treatment.20
Reflectance confocal microscopy also has served as a learning tool for new subclinical understandings of alopecia. In a study of CIA, the disease was found to be a dynamic process that could be categorized into 4 distinct phases distinguishable by combined confocal and dermoscopic features. This study also identified a new feature observable on RCM images—a CIA dot—defined as a dilated follicular infundibulum containing mashed, malted, nonhomogeneous material and normal or fragmented hair. This dot is thought to represent the initial microscopic sign of direct toxicity of chemotherapy on the hair follicle. Chemotherapy-induced alopecia dots persist throughout chemotherapy and subsequently disappear after chemotherapy ends.27
Limitations and Advantages—Currently, subtypes of cicatricial alopecias cannot be characterized on RCM because inflammatory cell types are not distinguished from each other (eg, eosinophils vs neutrophils). Another limitation of RCM is the loss of resolution below the superficial papillary dermis (a depth of approximately 150 µm); thus, deeper structures, such as the hair bulb, cannot be visualized.
Unlike global photography and trichoscopy, which are low-cost methods, RCM is much more costly, ranging upwards of several thousand dollars, and it may require additional technical support fees, making it less accessible for clinical practice. However, RCM imaging continues to be recommended as an intermediate step between trichoscopy and histology for the diagnosis and management of hair disease.26 If a biopsy is required, RCM can aid in the selection of a biopsy site, as areas with active inflammation are more informative than atrophic and fibrosed areas.23 The role of RCM in trichoscopy can be expanded by designing a more cost-effective and ergonomically suited scope for hair and scalp assessment.
Optical Coherence Tomography
Optical coherence tomography is a noninvasive handheld device that emits low-power infrared light to visualize the skin and adnexal structures. Optical coherence tomography relies on the principle of interferometry to detect phase differences in optical backscattering at varying tissue depths.28,29 It allows visualization up to 2 mm, which is 2 to 5 times deeper than RCM.36 Unlike RCM, which has cellular resolution, OCT has an axial resolution of 3 to 15 μm, which allows only for the detection of structural boundaries.30 There are various OCT modalities that differ in lateral and axial resolutions and maximum depth. Commercial software is available that measures changes in vascular density by depth, epidermal thickness, skin surface texture, and optical attenuation—the latter being an indirect measurement of collagen density and skin hydration.
Structures Visualized—Hair follicles can be well distinguished on OCT images, and as such, OCT is recognized as a diagnostic tool in trichology (Figure 4).31 Follicular openings, interfollicular collagen, and outlines of the hair shafts are visible; however, detailed components of the follicular unit cannot be visualized by OCT. Keratin hyperrefractivity identifies the hair shaft. Additionally, the hair matrix is denoted by a slightly granular texture in the dermis. Dynamic OCT produces colorized images that visualize blood flow within vessels.
Applications of OCT—Optical coherence tomography is utilized in investigative trichology because it provides highly reproducible measurements of hair shaft diameters, cross-sectional surface areas, and form factor, which is a surrogate parameter for hair shape. The cross-section of hair shafts provides insight into local metabolism and perifollicular inflammation. Cross-sections of hair shafts in areas of alopecia areata were found to be smaller than cross-sections in the unaffected scalp within the same individual.32 Follicular density can be manually quantified on OCT images, but there also is promise for automated quantification. A recent study by Urban et al33 described training a convolutional neural network to automatically count hair as well as hair-bearing and non–hair-bearing follicles in OCT scans. These investigators also were able to color-code hair according to height, resulting in the creation of a “height” map.
Optical coherence tomography has furthered our understanding of the pathophysiology of cicatricial and nonscarring alopecias. Vazquez-Herrera et al34 assessed the inflammatory and cicatricial stages of frontal fibrosing alopecia by OCT imaging. Inflammatory hairlines, which are seen in the early stages of frontal fibrosing alopecia, exhibited a thickened dermis, irregular distribution of collagen, and increased vascularity in both the superficial and deep dermal layers compared to cicatricial and healthy scalp. Conversely, late-stage cicatricial areas exhibited a thin dermis and collagen that appeared in a hyperreflective, concentric, onion-shaped pattern around remnant follicular openings. Vascular flow was reduced in the superficial dermis of a cicatricial scalp but increased in the deep dermal layers compared with a healthy scalp. The attenuation coefficients of these disease stages also were assessed. The attenuation coefficient of the inflammatory hairline was higher compared with normal skin, likely as a reflection of inflammatory infiltrate and edema, whereas the attenuation coefficient of cicatricial scalp was lower compared with normal skin, likely reflecting the reduced water content of atrophic skin.34 This differentiation of early- and late-stage cicatricial alopecias has implications for early treatment and improved prognosis. Additionally, there is potential for OCT to assist in the differentiation of alopecia subtypes, as it can measure the epidermal thickness and follicular density and was previously used to compare scarring and nonscarring alopecia.35
Advantages and Limitations—Similar to RCM, OCT may be cost prohibitive for some clinicians. In addition, OCT cannot visualize the follicular unit in cellular detail. However, the extent of OCT’s capabilities may not be fully realized. Dynamic OCT is a new angiographic type of OCT that shows potential in monitoring early subclinical responses to novel alopecia therapies, such as platelet-rich plasminogen, which is hypothesized to stimulate hair growth through angiogenesis. Additionally, OCT may improve outcomes of hair transplantation procedures by allowing for visualization of the subcutaneous angle of hair follicles. Blind extraction of hair follicles in follicular unit extraction procedures can result in inadvertent transection and damage to the hair follicle; OCT could help identify good candidates for follicular unit extraction, such as patients with hair follicles in parallel arrangement, who are predicted to have better results.36
Conclusion
The field of trichology will continue to evolve with the emergence of noninvasive imaging technologies that diagnose hair disease in early stages and enable treatment monitoring with quantification of hair parameters. As discussed in this review, global photography, trichoscopy, RCM, and OCT have furthered our understanding of alopecia pathophysiology and provided objective methods of treatment evaluation. The capabilities of these tools will continue to expand with advancements in add-on software and AI algorithms.
- Canfield D. Photographic documentation of hair growth in androgenetic alopecia. Dermatol Clin. 1996;14:713-721.
- Peytavi U, Hillmann K, Guarrera M. Hair growth assessment techniques. In: Peytavi U, Hillmann K, Guarrera M, eds. Hair Growth and Disorders. 4th ed. Springer; 2008:140-144.
- Chamberlain AJ, Dawber RP. Methods of evaluating hair growth. Australas J Dermatol. 2003;44:10-18.
- Dhurat R, Saraogi P. Hair evaluation methods: merits and demerits. Int J Trichology. 2009;1:108-119.
- Kaufman KD, Olsen EA, Whiting D, et al. Finasteride in the treatment of men with androgenetic alopecia. J Am Acad Dermatol. 1998;39:578-579.
- Capily Institute. Artificial intelligence (A.I.) powered hair growth tracking. Accessed July 31, 2023. https://tss-aesthetics.com/capily-hair-tracking-syst
- Dinh Q, Sinclair R. Female pattern hair loss: current treatment concepts. Clin Interv Aging. 2007;2:189-199.
- Dhurat R, Saraogi P. Hair evaluation methods: merits and demerits. Int J Trichology. 2009;1:108-119.
- Wikramanayake TC, Mauro LM, Tabas IA, et al. Cross-section trichometry: a clinical tool for assessing the progression and treatment response of alopecia. Int J Trichology. 2012;4:259-264.
- Alessandrini A, Bruni F, Piraccini BM, et al. Common causes of hair loss—clinical manifestations, trichoscopy and therapy. J Eur Acad Dermatol Venereol. 2021;35:629-640.
- Ashique K, Kaliyadan F. Clinical photography for trichology practice: tips and tricks. Int J Trichology. 2011;3:7-13.
- Rudnicka L, Olszewska M, Rakowska A, et al. Trichoscopy: a new method for diagnosing hair loss. J Drugs Dermatol. 2008;7:651-654.
- Kinoshita-Ise M, Sachdeva M. Update on trichoscopy: integration of the terminology by systematic approach and a proposal of a diagnostic flowchart. J Dermatol. 2022;49:4-18. doi:10.1111/1346-8138.16233
- Van Neste D, Trüeb RM. Critical study of hair growth analysis with computer-assisted methods. J Eur Acad Dermatol Venereol. 2006;20:578-583.
- Romero J, Grimalt R. Trichoscopy: essentials for the dermatologist. World J Dermatol. 2015;4:63-68.
- Trichoscopy: a new frontier for the diagnosis of hair diseases. Exp Rev Dermatol. 2012;7:429-437.
- Lee B, Chan J, Monselise A, et al. Assessment of hair density and caliber in Caucasian and Asian female subjects with female pattern hair loss by using the Folliscope. J Am Acad Dermatol. 2012;66:166-167.
- Inui S. Trichoscopy for common hair loss diseases: algorithmic method for diagnosis. J Dermatol. 2010;38:71-75.
- Dhurat R. Phototrichogram. Indian J Dermatol Venereol Leprol. 2006;72:242-244.
- Agozzino M, Tosti A, Barbieri L, et al. Confocal microscopic features of scarring alopecia: preliminary report. Br J Dermatol. 2011;165:534-540.
- Kuck M, Schanzer S, Ulrich M, et al. Analysis of the efficiency of hair removal by different optical methods: comparison of Trichoscan, reflectance confocal microscopy, and optical coherence tomography. J Biomed Opt. 2012;17:101504.
- Levine A, Markowitz O. Introduction to reflectance confocal microscopy and its use in clinical practice. JAAD Case Rep. 2018;4:1014-1023.
- Agozzino M, Ardigò M. Scalp confocal microscopy. In: Humbert P, Maibach H, Fanian F, et al, eds. Agache’s Measuring the Skin: Non-invasive Investigations, Physiology, Normal Constants. 2nd ed. Springer International Publishing; 2016:311-326.
- Rudnicka L, Olszewska M, Rakowska A. In vivo reflectance confocal microscopy: usefulness for diagnosing hair diseases. J Dermatol Case Rep. 2008;2:55-59.
- Kurzeja M, Czuwara J, Walecka I, et al. Features of classic lichen planopilaris and frontal fibrosing alopecia in reflectance confocal microscopy: a preliminary study. Skin Res Technol. 2021;27:266-271.
- Ardigò M, Agozzino M, Franceschini C, et al. Reflectance confocal microscopy for scarring and non-scarring alopecia real-time assessment. Arch Dermatol Res. 2016;308:309-318.
- Franceschini C, Garelli V, Persechino F, et al. Dermoscopy and confocal microscopy for different chemotherapy-induced alopecia (CIA) phases characterization: preliminary study. Skin Res Technol. 2020;26:269-276.
- Martinez-Velasco MA, Perper M, Maddy AJ, et al. In vitro determination of Mexican Mestizo hair shaft diameter using optical coherence tomography. Skin Res Technol. 2018;24;274-277.
- Srivastava R, Manfredini M, Rao BK. Noninvasive imaging tools in dermatology. Cutis. 2019;104:108-113.
- Wan B, Ganier C, Du-Harpur X, et al. Applications and future directions for optical coherence tomography in dermatology. Br J Dermatol. 2021;184:1014-1022.
- Blume-Peytavi U, Vieten J, Knuttel A et al. Optical coherent tomography (OCT): a new method for online-measurement of hair shaft thickness. J Dtsch Dermatol Ges. 2004;2:546.
- Garcia Bartels N, Jahnke I, Patzelt A, et al. Hair shaft abnormalities in alopecia areata evaluated by optical coherence tomography. Skin Res Technol. 2011;17:201-205.
- Urban G, Feil N, Csuka E, et al. Combining deep learning with optical coherence tomography imaging to determine scalp hair and follicle counts. Lasers Surg Med. 2021;53:171-178.
- Vazquez-Herrera NE, Eber AE, Martinez-Velasco MA, et al. Optical coherence tomography for the investigation of frontal fibrosing alopecia. J Eur Acad Dermatol Venereol. 2018;32:318-322.
- Ekelem C, Feil N, Csuka E, et al. Optical coherence tomography in the evaluation of the scalp and hair: common features and clinical utility. Lasers Surg Med. 2021;53:129-140.
- Schicho K, Seemann R, Binder M, et al. Optical coherence tomography for planning of follicular unit extraction. Dermatol Surg. 2015;41:358-363.
New imaging tools along with adaptations to existing technologies have been emerging in recent years, with the potential to improve hair diagnostics and treatment monitoring. We provide an overview of 4 noninvasive hair imaging technologies: global photography, trichoscopy, reflectance confocal microscopy (RCM), and optical coherence tomography (OCT). For each instrument, we discuss current and future applications in clinical practice and research along with advantages and disadvantages.
Global Photography
Global photography allows for the analysis of hair growth, volume, distribution, and density through serial standardized photographs.1 Global photography was first introduced for hair growth studies in 1987 and soon after was used for hair and scalp assessments in finasteride clinical trials.2
Hair Assessment—Washed, dried, and combed hair, without hair product, are required for accurate imaging; wet conditions increase reflection and promote hair clumping, thus revealing more scalp and depicting the patient as having less hair.1 Headshots are taken from short distances and use stereotactic positioning devices to create 4 global views: vertex, midline, frontal, and temporal.3 Stereotactic positioning involves fixing the patient’s chin and forehead as well as mounting the camera and flash device to ensure proper magnification. These adjustments ensure lighting remains consistent throughout consecutive study visits.4 Various grading scales are available for use in hair growth clinical studies to increase objectivity in the analysis of serial global photographs. A blinded evaluator should assess the before and after photographs to limit experimenter bias. Global photography often is combined with quantitative software analysis for improved detection of hair changes.1
Advancements—Growing interest in improving global photography has resulted in various application-based, artificial intelligence (AI)–mediated tools to simplify photograph collection and analysis. For instance, new hair analysis software utilizes AI algorithms to account for facial features in determining the optimal angle for capturing global photographs (Figure 1), which simplifies the generation of global photography images through smartphone applications and obviates the need for additional stereotactic positioning equipment.5,6

Limitations—Clinicians should be aware of global photography’s requirements for consistency in lighting, camera settings, film, and image processing, which can limit the accuracy of hair assessment over time if not replicated correctly.7,8 Emerging global photography software has helped to overcome some of these limitations.
Global photography is less precise when a patient’s hair loss is less than 50%, as it is difficult to discern subtle hair changes. Thus, global photography provides limited utility in assessing minimal to moderate hair loss.9 Currently, global photography largely functions as an adjunct tool for other hair analysis methods rather than as a stand-alone tool.
Trichoscopy
Trichoscopy (also known as dermoscopy of the hair and scalp) may be performed with a manual dermoscope (with 10× magnification) or a digital videodermatoscope (up to 1000× magnification).10-12 Unlike global photography, trichoscopy provides a detailed structural analysis of hair shafts, follicular openings, and perifollicular and interfollicular areas.13 Kinoshita-Ise and Sachdeva13 provided an in-depth, updated review of trichoscopy terminology with their definitions and associated conditions (with prevalence), which should be referenced when performing trichoscopic examination.
Hair Assessment—Trichoscopic assessment begins with inspection of follicular openings (also referred to as “dots”), which vary in color depending on the material filling them—degrading keratinocytes, keratin, sebaceous debris, melanin, or fractured hairs.13 The structure of hair shafts also is examined, showing broken hairs, short vellus hairs, and comma hairs, among others. Perifollicular areas are examined for scale, erythema, blue-gray dots, and whitish halos. Interfollicular areas are examined for pigment pattern as well as vascularization, which often presents in a looping configuration under dermoscopy. A combination of dot colorization, hair shaft structure, and perifollicular and interfollicular findings inform diagnostic algorithms of hair and scalp conditions. For example, central centrifugal cicatricial alopecia, the most common alopecia seen in Black women, has been associated with a combination of honeycomb pigment pattern, perifollicular whitish halo, pinpoint white dots, white patches, and perifollicular erythema.13
Advantages—Perhaps the most useful feature of trichoscopy is its ability to translate visualized features into simple diagnostic algorithms. For instance, if the clinician has diagnosed the patient with noncicatricial alopecia, they would next focus on dot colors. With black dots, the next step would be to determine whether the hairs are tapered or coiled, and so on. This systematic approach enables the clinician to narrow possible diagnoses.2 An additional advantage of trichoscopy is that it examines large surface areas noninvasively as compared to hair-pull tests and scalp biopsy.14,15 Trichoscopy allows temporal comparisons of the same area for disease and treatment monitoring with more diagnostic detail than global photography.16 Trichoscopy also is useful in selecting biopsy locations by discerning and avoiding areas of scar tissue.17
Limitations—Diagnosis via the trichoscopy algorithm is limiting because it is not comprehensive of all hair and scalp disease.18 Additionally, many pathologies exhibit overlapping follicular and interfollicular patterning. For example, almost all subtypes of scarring alopecia present with hair loss and scarred follicles once they have progressed to advanced stages. Further studies should identify more specific patterns of hair and scalp pathologies, which could then be incorporated into a diagnostic algorithm.13
Advancements—The advent of hair analysis software has expanded the role of videodermoscopy by rapidly quantifying hair growth parameters such as hair count, follicular density, and follicular diameter, as well as interfollicular distances (Figure 2).14,17 Vellus and terminal hairs are differentiated according to their thickness and length.17 Moreover, the software can analyze the same area of the scalp over time by either virtual tattoos, semipermanent markings, or precise location measurements, increasing intra- and interclass correlation. The rate of hair growth, hair shedding, and parameters of anagen and telogen hairs can be studied by a method termed phototrichogram whereby a transitional area of hair loss and normal hair growth is identified and trimmed to less than 1 mm from the skin surface.19 A baseline photograph is taken using videodermoscopy. After approximately 3 days, the identical region is photographed and compared with the initial image to observe changes in the hair. Software programs can distinguish the growing hair as anagen and nongrowing hair as telogen, calculating the anagen-to-telogen ratio as well as hair growth rate, which are essential measurements in hair research and clinical studies. Software programs have replaced laborious and time-consuming manual hair counts and have rapidly grown in popularity in evaluating patterned hair loss.

Reflectance Confocal Microscopy
Reflectance confocal microscopy is a noninvasive imaging tool that visualizes skin and its appendages at near-histologic resolution (lateral resolution of 0.5–1 μm). It produces grayscale horizontal images that can be taken at levels ranging from the stratum corneum to the superficial papillary dermis, corresponding to a depth of approximately 100 to 150 µm. Thus, a hair follicle can be imaged starting from the follicular ostia down to the reachable papillary dermis (Figure 3).20 Image contrast is provided by differences in the size and refractive indices of cellular organelles.21,22 There are 2 commercially available RCM devices: VivaScope 1500 and VivaScope 3000 (Caliber Imaging & Diagnostics, Inc).

VivaScope 1500, a wide-probe microscope, requires the attachment of a plastic window to the desired imaging area. The plastic window is lined with medical adhesive tape to prevent movement during imaging. The adhesive tape can pull on hair upon removal, which is not ideal for patients with existing hair loss. Additionally, the image quality of VivaSope 1500 is best in flat areas and areas where hair is shaved.20,23,24 Despite these disadvantages, VivaScope 1500 has successfully shown utility in research studies, which suggests that these obstacles can be overcome by experienced users. The handheld VivaScope 3000 is ergonomically designed and suitable for curved surfaces such as the scalp, with the advantage of not requiring any adhesive. However, the images acquired from the VivaScope 3000 cover a smaller surface area.
Structures Visualized—Structures distinguished with RCM include keratinocytes, melanocytes, inflammatory cells, hair follicles, hair shafts, adnexal infundibular epithelium, blood vessels, fibroblasts, and collagen.23 Real-time visualization of blood flow also can be seen.
Applications of RCM—Reflectance confocal microscopy has been used to study scalp discoid lupus, lichen planopilaris, frontal fibrosing alopecia, folliculitis decalvans, chemotherapy-induced alopecia (CIA), alopecia areata, and androgenetic alopecia. Diagnostic RCM criteria for such alopecias have been developed based on their correspondence to histopathology. An RCM study of classic lichen planopilaris and frontal fibrosing alopecia identified features of epidermal disarray, infundibular hyperkeratosis, inflammatory cells, pigment incontinence, perifollicular fibrosis, bandlike scarring, melanophages in the dermis, dilated blood vessels, basal layer vacuolar degeneration, and necrotic keratinocytes.25 Pigment incontinence in the superficial epidermis, perifollicular lichenoid inflammation, and hyperkeratosis were characteristic RCM features of early-stage lichen planopilaris, while perifollicular fibrosis and dilated blood vessels were characteristic RCM features of late-stage disease. The ability of RCM features to distinguish different stages of lichen planopilaris shows its potential in treating early disease and preventing irreversible hair loss.
Differentiating between scarring and nonscarring alopecia also is possible through RCM. The presence of periadnexal, epidermal, and dermal inflammatory cells, in addition to periadnexal sclerosis, are defining RCM features of scarring alopecia.26 These features are absent in nonscarring alopecias. Reflectance confocal microscopy additionally has been shown to be useful in the treatment monitoring of lichen planopilaris and discoid lupus erythematosus.20 Independent reviewers, blinded to the patients’ identities, were able to characterize and follow features of these scarring alopecias by RCM. The assessed RCM features were comparable to those observed by histopathologic evaluation: epidermal disarray, spongiosis, exocytosis of inflammatory cells in the epidermis, interface dermatitis, peri- and intra-adnexal infiltration of inflammatory cells, dilated vessels in the dermis, dermal infiltration of inflammatory cells and melanophages, and dermal sclerosis. A reduction in inflammatory cells across multiple skin layers and at the level of the adnexal epithelium correlated with clinical response to treatment. Reflectance confocal microscopy also was able to detect recurrence of inflammation in cases where treatment had been interrupted before clinical signs of disease recurrence were evident. The authors thus concluded that RCM’s sensitivity can guide timing of treatment and avoid delays in starting or restarting treatment.20
Reflectance confocal microscopy also has served as a learning tool for new subclinical understandings of alopecia. In a study of CIA, the disease was found to be a dynamic process that could be categorized into 4 distinct phases distinguishable by combined confocal and dermoscopic features. This study also identified a new feature observable on RCM images—a CIA dot—defined as a dilated follicular infundibulum containing mashed, malted, nonhomogeneous material and normal or fragmented hair. This dot is thought to represent the initial microscopic sign of direct toxicity of chemotherapy on the hair follicle. Chemotherapy-induced alopecia dots persist throughout chemotherapy and subsequently disappear after chemotherapy ends.27
Limitations and Advantages—Currently, subtypes of cicatricial alopecias cannot be characterized on RCM because inflammatory cell types are not distinguished from each other (eg, eosinophils vs neutrophils). Another limitation of RCM is the loss of resolution below the superficial papillary dermis (a depth of approximately 150 µm); thus, deeper structures, such as the hair bulb, cannot be visualized.
Unlike global photography and trichoscopy, which are low-cost methods, RCM is much more costly, ranging upwards of several thousand dollars, and it may require additional technical support fees, making it less accessible for clinical practice. However, RCM imaging continues to be recommended as an intermediate step between trichoscopy and histology for the diagnosis and management of hair disease.26 If a biopsy is required, RCM can aid in the selection of a biopsy site, as areas with active inflammation are more informative than atrophic and fibrosed areas.23 The role of RCM in trichoscopy can be expanded by designing a more cost-effective and ergonomically suited scope for hair and scalp assessment.
Optical Coherence Tomography
Optical coherence tomography is a noninvasive handheld device that emits low-power infrared light to visualize the skin and adnexal structures. Optical coherence tomography relies on the principle of interferometry to detect phase differences in optical backscattering at varying tissue depths.28,29 It allows visualization up to 2 mm, which is 2 to 5 times deeper than RCM.36 Unlike RCM, which has cellular resolution, OCT has an axial resolution of 3 to 15 μm, which allows only for the detection of structural boundaries.30 There are various OCT modalities that differ in lateral and axial resolutions and maximum depth. Commercial software is available that measures changes in vascular density by depth, epidermal thickness, skin surface texture, and optical attenuation—the latter being an indirect measurement of collagen density and skin hydration.
Structures Visualized—Hair follicles can be well distinguished on OCT images, and as such, OCT is recognized as a diagnostic tool in trichology (Figure 4).31 Follicular openings, interfollicular collagen, and outlines of the hair shafts are visible; however, detailed components of the follicular unit cannot be visualized by OCT. Keratin hyperrefractivity identifies the hair shaft. Additionally, the hair matrix is denoted by a slightly granular texture in the dermis. Dynamic OCT produces colorized images that visualize blood flow within vessels.

Applications of OCT—Optical coherence tomography is utilized in investigative trichology because it provides highly reproducible measurements of hair shaft diameters, cross-sectional surface areas, and form factor, which is a surrogate parameter for hair shape. The cross-section of hair shafts provides insight into local metabolism and perifollicular inflammation. Cross-sections of hair shafts in areas of alopecia areata were found to be smaller than cross-sections in the unaffected scalp within the same individual.32 Follicular density can be manually quantified on OCT images, but there also is promise for automated quantification. A recent study by Urban et al33 described training a convolutional neural network to automatically count hair as well as hair-bearing and non–hair-bearing follicles in OCT scans. These investigators also were able to color-code hair according to height, resulting in the creation of a “height” map.
Optical coherence tomography has furthered our understanding of the pathophysiology of cicatricial and nonscarring alopecias. Vazquez-Herrera et al34 assessed the inflammatory and cicatricial stages of frontal fibrosing alopecia by OCT imaging. Inflammatory hairlines, which are seen in the early stages of frontal fibrosing alopecia, exhibited a thickened dermis, irregular distribution of collagen, and increased vascularity in both the superficial and deep dermal layers compared to cicatricial and healthy scalp. Conversely, late-stage cicatricial areas exhibited a thin dermis and collagen that appeared in a hyperreflective, concentric, onion-shaped pattern around remnant follicular openings. Vascular flow was reduced in the superficial dermis of a cicatricial scalp but increased in the deep dermal layers compared with a healthy scalp. The attenuation coefficients of these disease stages also were assessed. The attenuation coefficient of the inflammatory hairline was higher compared with normal skin, likely as a reflection of inflammatory infiltrate and edema, whereas the attenuation coefficient of cicatricial scalp was lower compared with normal skin, likely reflecting the reduced water content of atrophic skin.34 This differentiation of early- and late-stage cicatricial alopecias has implications for early treatment and improved prognosis. Additionally, there is potential for OCT to assist in the differentiation of alopecia subtypes, as it can measure the epidermal thickness and follicular density and was previously used to compare scarring and nonscarring alopecia.35
Advantages and Limitations—Similar to RCM, OCT may be cost prohibitive for some clinicians. In addition, OCT cannot visualize the follicular unit in cellular detail. However, the extent of OCT’s capabilities may not be fully realized. Dynamic OCT is a new angiographic type of OCT that shows potential in monitoring early subclinical responses to novel alopecia therapies, such as platelet-rich plasminogen, which is hypothesized to stimulate hair growth through angiogenesis. Additionally, OCT may improve outcomes of hair transplantation procedures by allowing for visualization of the subcutaneous angle of hair follicles. Blind extraction of hair follicles in follicular unit extraction procedures can result in inadvertent transection and damage to the hair follicle; OCT could help identify good candidates for follicular unit extraction, such as patients with hair follicles in parallel arrangement, who are predicted to have better results.36
Conclusion
The field of trichology will continue to evolve with the emergence of noninvasive imaging technologies that diagnose hair disease in early stages and enable treatment monitoring with quantification of hair parameters. As discussed in this review, global photography, trichoscopy, RCM, and OCT have furthered our understanding of alopecia pathophysiology and provided objective methods of treatment evaluation. The capabilities of these tools will continue to expand with advancements in add-on software and AI algorithms.
New imaging tools along with adaptations to existing technologies have been emerging in recent years, with the potential to improve hair diagnostics and treatment monitoring. We provide an overview of 4 noninvasive hair imaging technologies: global photography, trichoscopy, reflectance confocal microscopy (RCM), and optical coherence tomography (OCT). For each instrument, we discuss current and future applications in clinical practice and research along with advantages and disadvantages.
Global Photography
Global photography allows for the analysis of hair growth, volume, distribution, and density through serial standardized photographs.1 Global photography was first introduced for hair growth studies in 1987 and soon after was used for hair and scalp assessments in finasteride clinical trials.2
Hair Assessment—Washed, dried, and combed hair, without hair product, are required for accurate imaging; wet conditions increase reflection and promote hair clumping, thus revealing more scalp and depicting the patient as having less hair.1 Headshots are taken from short distances and use stereotactic positioning devices to create 4 global views: vertex, midline, frontal, and temporal.3 Stereotactic positioning involves fixing the patient’s chin and forehead as well as mounting the camera and flash device to ensure proper magnification. These adjustments ensure lighting remains consistent throughout consecutive study visits.4 Various grading scales are available for use in hair growth clinical studies to increase objectivity in the analysis of serial global photographs. A blinded evaluator should assess the before and after photographs to limit experimenter bias. Global photography often is combined with quantitative software analysis for improved detection of hair changes.1
Advancements—Growing interest in improving global photography has resulted in various application-based, artificial intelligence (AI)–mediated tools to simplify photograph collection and analysis. For instance, new hair analysis software utilizes AI algorithms to account for facial features in determining the optimal angle for capturing global photographs (Figure 1), which simplifies the generation of global photography images through smartphone applications and obviates the need for additional stereotactic positioning equipment.5,6

Limitations—Clinicians should be aware of global photography’s requirements for consistency in lighting, camera settings, film, and image processing, which can limit the accuracy of hair assessment over time if not replicated correctly.7,8 Emerging global photography software has helped to overcome some of these limitations.
Global photography is less precise when a patient’s hair loss is less than 50%, as it is difficult to discern subtle hair changes. Thus, global photography provides limited utility in assessing minimal to moderate hair loss.9 Currently, global photography largely functions as an adjunct tool for other hair analysis methods rather than as a stand-alone tool.
Trichoscopy
Trichoscopy (also known as dermoscopy of the hair and scalp) may be performed with a manual dermoscope (with 10× magnification) or a digital videodermatoscope (up to 1000× magnification).10-12 Unlike global photography, trichoscopy provides a detailed structural analysis of hair shafts, follicular openings, and perifollicular and interfollicular areas.13 Kinoshita-Ise and Sachdeva13 provided an in-depth, updated review of trichoscopy terminology with their definitions and associated conditions (with prevalence), which should be referenced when performing trichoscopic examination.
Hair Assessment—Trichoscopic assessment begins with inspection of follicular openings (also referred to as “dots”), which vary in color depending on the material filling them—degrading keratinocytes, keratin, sebaceous debris, melanin, or fractured hairs.13 The structure of hair shafts also is examined, showing broken hairs, short vellus hairs, and comma hairs, among others. Perifollicular areas are examined for scale, erythema, blue-gray dots, and whitish halos. Interfollicular areas are examined for pigment pattern as well as vascularization, which often presents in a looping configuration under dermoscopy. A combination of dot colorization, hair shaft structure, and perifollicular and interfollicular findings inform diagnostic algorithms of hair and scalp conditions. For example, central centrifugal cicatricial alopecia, the most common alopecia seen in Black women, has been associated with a combination of honeycomb pigment pattern, perifollicular whitish halo, pinpoint white dots, white patches, and perifollicular erythema.13
Advantages—Perhaps the most useful feature of trichoscopy is its ability to translate visualized features into simple diagnostic algorithms. For instance, if the clinician has diagnosed the patient with noncicatricial alopecia, they would next focus on dot colors. With black dots, the next step would be to determine whether the hairs are tapered or coiled, and so on. This systematic approach enables the clinician to narrow possible diagnoses.2 An additional advantage of trichoscopy is that it examines large surface areas noninvasively as compared to hair-pull tests and scalp biopsy.14,15 Trichoscopy allows temporal comparisons of the same area for disease and treatment monitoring with more diagnostic detail than global photography.16 Trichoscopy also is useful in selecting biopsy locations by discerning and avoiding areas of scar tissue.17
Limitations—Diagnosis via the trichoscopy algorithm is limiting because it is not comprehensive of all hair and scalp disease.18 Additionally, many pathologies exhibit overlapping follicular and interfollicular patterning. For example, almost all subtypes of scarring alopecia present with hair loss and scarred follicles once they have progressed to advanced stages. Further studies should identify more specific patterns of hair and scalp pathologies, which could then be incorporated into a diagnostic algorithm.13
Advancements—The advent of hair analysis software has expanded the role of videodermoscopy by rapidly quantifying hair growth parameters such as hair count, follicular density, and follicular diameter, as well as interfollicular distances (Figure 2).14,17 Vellus and terminal hairs are differentiated according to their thickness and length.17 Moreover, the software can analyze the same area of the scalp over time by either virtual tattoos, semipermanent markings, or precise location measurements, increasing intra- and interclass correlation. The rate of hair growth, hair shedding, and parameters of anagen and telogen hairs can be studied by a method termed phototrichogram whereby a transitional area of hair loss and normal hair growth is identified and trimmed to less than 1 mm from the skin surface.19 A baseline photograph is taken using videodermoscopy. After approximately 3 days, the identical region is photographed and compared with the initial image to observe changes in the hair. Software programs can distinguish the growing hair as anagen and nongrowing hair as telogen, calculating the anagen-to-telogen ratio as well as hair growth rate, which are essential measurements in hair research and clinical studies. Software programs have replaced laborious and time-consuming manual hair counts and have rapidly grown in popularity in evaluating patterned hair loss.

Reflectance Confocal Microscopy
Reflectance confocal microscopy is a noninvasive imaging tool that visualizes skin and its appendages at near-histologic resolution (lateral resolution of 0.5–1 μm). It produces grayscale horizontal images that can be taken at levels ranging from the stratum corneum to the superficial papillary dermis, corresponding to a depth of approximately 100 to 150 µm. Thus, a hair follicle can be imaged starting from the follicular ostia down to the reachable papillary dermis (Figure 3).20 Image contrast is provided by differences in the size and refractive indices of cellular organelles.21,22 There are 2 commercially available RCM devices: VivaScope 1500 and VivaScope 3000 (Caliber Imaging & Diagnostics, Inc).

VivaScope 1500, a wide-probe microscope, requires the attachment of a plastic window to the desired imaging area. The plastic window is lined with medical adhesive tape to prevent movement during imaging. The adhesive tape can pull on hair upon removal, which is not ideal for patients with existing hair loss. Additionally, the image quality of VivaSope 1500 is best in flat areas and areas where hair is shaved.20,23,24 Despite these disadvantages, VivaScope 1500 has successfully shown utility in research studies, which suggests that these obstacles can be overcome by experienced users. The handheld VivaScope 3000 is ergonomically designed and suitable for curved surfaces such as the scalp, with the advantage of not requiring any adhesive. However, the images acquired from the VivaScope 3000 cover a smaller surface area.
Structures Visualized—Structures distinguished with RCM include keratinocytes, melanocytes, inflammatory cells, hair follicles, hair shafts, adnexal infundibular epithelium, blood vessels, fibroblasts, and collagen.23 Real-time visualization of blood flow also can be seen.
Applications of RCM—Reflectance confocal microscopy has been used to study scalp discoid lupus, lichen planopilaris, frontal fibrosing alopecia, folliculitis decalvans, chemotherapy-induced alopecia (CIA), alopecia areata, and androgenetic alopecia. Diagnostic RCM criteria for such alopecias have been developed based on their correspondence to histopathology. An RCM study of classic lichen planopilaris and frontal fibrosing alopecia identified features of epidermal disarray, infundibular hyperkeratosis, inflammatory cells, pigment incontinence, perifollicular fibrosis, bandlike scarring, melanophages in the dermis, dilated blood vessels, basal layer vacuolar degeneration, and necrotic keratinocytes.25 Pigment incontinence in the superficial epidermis, perifollicular lichenoid inflammation, and hyperkeratosis were characteristic RCM features of early-stage lichen planopilaris, while perifollicular fibrosis and dilated blood vessels were characteristic RCM features of late-stage disease. The ability of RCM features to distinguish different stages of lichen planopilaris shows its potential in treating early disease and preventing irreversible hair loss.
Differentiating between scarring and nonscarring alopecia also is possible through RCM. The presence of periadnexal, epidermal, and dermal inflammatory cells, in addition to periadnexal sclerosis, are defining RCM features of scarring alopecia.26 These features are absent in nonscarring alopecias. Reflectance confocal microscopy additionally has been shown to be useful in the treatment monitoring of lichen planopilaris and discoid lupus erythematosus.20 Independent reviewers, blinded to the patients’ identities, were able to characterize and follow features of these scarring alopecias by RCM. The assessed RCM features were comparable to those observed by histopathologic evaluation: epidermal disarray, spongiosis, exocytosis of inflammatory cells in the epidermis, interface dermatitis, peri- and intra-adnexal infiltration of inflammatory cells, dilated vessels in the dermis, dermal infiltration of inflammatory cells and melanophages, and dermal sclerosis. A reduction in inflammatory cells across multiple skin layers and at the level of the adnexal epithelium correlated with clinical response to treatment. Reflectance confocal microscopy also was able to detect recurrence of inflammation in cases where treatment had been interrupted before clinical signs of disease recurrence were evident. The authors thus concluded that RCM’s sensitivity can guide timing of treatment and avoid delays in starting or restarting treatment.20
Reflectance confocal microscopy also has served as a learning tool for new subclinical understandings of alopecia. In a study of CIA, the disease was found to be a dynamic process that could be categorized into 4 distinct phases distinguishable by combined confocal and dermoscopic features. This study also identified a new feature observable on RCM images—a CIA dot—defined as a dilated follicular infundibulum containing mashed, malted, nonhomogeneous material and normal or fragmented hair. This dot is thought to represent the initial microscopic sign of direct toxicity of chemotherapy on the hair follicle. Chemotherapy-induced alopecia dots persist throughout chemotherapy and subsequently disappear after chemotherapy ends.27
Limitations and Advantages—Currently, subtypes of cicatricial alopecias cannot be characterized on RCM because inflammatory cell types are not distinguished from each other (eg, eosinophils vs neutrophils). Another limitation of RCM is the loss of resolution below the superficial papillary dermis (a depth of approximately 150 µm); thus, deeper structures, such as the hair bulb, cannot be visualized.
Unlike global photography and trichoscopy, which are low-cost methods, RCM is much more costly, ranging upwards of several thousand dollars, and it may require additional technical support fees, making it less accessible for clinical practice. However, RCM imaging continues to be recommended as an intermediate step between trichoscopy and histology for the diagnosis and management of hair disease.26 If a biopsy is required, RCM can aid in the selection of a biopsy site, as areas with active inflammation are more informative than atrophic and fibrosed areas.23 The role of RCM in trichoscopy can be expanded by designing a more cost-effective and ergonomically suited scope for hair and scalp assessment.
Optical Coherence Tomography
Optical coherence tomography is a noninvasive handheld device that emits low-power infrared light to visualize the skin and adnexal structures. Optical coherence tomography relies on the principle of interferometry to detect phase differences in optical backscattering at varying tissue depths.28,29 It allows visualization up to 2 mm, which is 2 to 5 times deeper than RCM.36 Unlike RCM, which has cellular resolution, OCT has an axial resolution of 3 to 15 μm, which allows only for the detection of structural boundaries.30 There are various OCT modalities that differ in lateral and axial resolutions and maximum depth. Commercial software is available that measures changes in vascular density by depth, epidermal thickness, skin surface texture, and optical attenuation—the latter being an indirect measurement of collagen density and skin hydration.
Structures Visualized—Hair follicles can be well distinguished on OCT images, and as such, OCT is recognized as a diagnostic tool in trichology (Figure 4).31 Follicular openings, interfollicular collagen, and outlines of the hair shafts are visible; however, detailed components of the follicular unit cannot be visualized by OCT. Keratin hyperrefractivity identifies the hair shaft. Additionally, the hair matrix is denoted by a slightly granular texture in the dermis. Dynamic OCT produces colorized images that visualize blood flow within vessels.

Applications of OCT—Optical coherence tomography is utilized in investigative trichology because it provides highly reproducible measurements of hair shaft diameters, cross-sectional surface areas, and form factor, which is a surrogate parameter for hair shape. The cross-section of hair shafts provides insight into local metabolism and perifollicular inflammation. Cross-sections of hair shafts in areas of alopecia areata were found to be smaller than cross-sections in the unaffected scalp within the same individual.32 Follicular density can be manually quantified on OCT images, but there also is promise for automated quantification. A recent study by Urban et al33 described training a convolutional neural network to automatically count hair as well as hair-bearing and non–hair-bearing follicles in OCT scans. These investigators also were able to color-code hair according to height, resulting in the creation of a “height” map.
Optical coherence tomography has furthered our understanding of the pathophysiology of cicatricial and nonscarring alopecias. Vazquez-Herrera et al34 assessed the inflammatory and cicatricial stages of frontal fibrosing alopecia by OCT imaging. Inflammatory hairlines, which are seen in the early stages of frontal fibrosing alopecia, exhibited a thickened dermis, irregular distribution of collagen, and increased vascularity in both the superficial and deep dermal layers compared to cicatricial and healthy scalp. Conversely, late-stage cicatricial areas exhibited a thin dermis and collagen that appeared in a hyperreflective, concentric, onion-shaped pattern around remnant follicular openings. Vascular flow was reduced in the superficial dermis of a cicatricial scalp but increased in the deep dermal layers compared with a healthy scalp. The attenuation coefficients of these disease stages also were assessed. The attenuation coefficient of the inflammatory hairline was higher compared with normal skin, likely as a reflection of inflammatory infiltrate and edema, whereas the attenuation coefficient of cicatricial scalp was lower compared with normal skin, likely reflecting the reduced water content of atrophic skin.34 This differentiation of early- and late-stage cicatricial alopecias has implications for early treatment and improved prognosis. Additionally, there is potential for OCT to assist in the differentiation of alopecia subtypes, as it can measure the epidermal thickness and follicular density and was previously used to compare scarring and nonscarring alopecia.35
Advantages and Limitations—Similar to RCM, OCT may be cost prohibitive for some clinicians. In addition, OCT cannot visualize the follicular unit in cellular detail. However, the extent of OCT’s capabilities may not be fully realized. Dynamic OCT is a new angiographic type of OCT that shows potential in monitoring early subclinical responses to novel alopecia therapies, such as platelet-rich plasminogen, which is hypothesized to stimulate hair growth through angiogenesis. Additionally, OCT may improve outcomes of hair transplantation procedures by allowing for visualization of the subcutaneous angle of hair follicles. Blind extraction of hair follicles in follicular unit extraction procedures can result in inadvertent transection and damage to the hair follicle; OCT could help identify good candidates for follicular unit extraction, such as patients with hair follicles in parallel arrangement, who are predicted to have better results.36
Conclusion
The field of trichology will continue to evolve with the emergence of noninvasive imaging technologies that diagnose hair disease in early stages and enable treatment monitoring with quantification of hair parameters. As discussed in this review, global photography, trichoscopy, RCM, and OCT have furthered our understanding of alopecia pathophysiology and provided objective methods of treatment evaluation. The capabilities of these tools will continue to expand with advancements in add-on software and AI algorithms.
- Canfield D. Photographic documentation of hair growth in androgenetic alopecia. Dermatol Clin. 1996;14:713-721.
- Peytavi U, Hillmann K, Guarrera M. Hair growth assessment techniques. In: Peytavi U, Hillmann K, Guarrera M, eds. Hair Growth and Disorders. 4th ed. Springer; 2008:140-144.
- Chamberlain AJ, Dawber RP. Methods of evaluating hair growth. Australas J Dermatol. 2003;44:10-18.
- Dhurat R, Saraogi P. Hair evaluation methods: merits and demerits. Int J Trichology. 2009;1:108-119.
- Kaufman KD, Olsen EA, Whiting D, et al. Finasteride in the treatment of men with androgenetic alopecia. J Am Acad Dermatol. 1998;39:578-579.
- Capily Institute. Artificial intelligence (A.I.) powered hair growth tracking. Accessed July 31, 2023. https://tss-aesthetics.com/capily-hair-tracking-syst
- Dinh Q, Sinclair R. Female pattern hair loss: current treatment concepts. Clin Interv Aging. 2007;2:189-199.
- Dhurat R, Saraogi P. Hair evaluation methods: merits and demerits. Int J Trichology. 2009;1:108-119.
- Wikramanayake TC, Mauro LM, Tabas IA, et al. Cross-section trichometry: a clinical tool for assessing the progression and treatment response of alopecia. Int J Trichology. 2012;4:259-264.
- Alessandrini A, Bruni F, Piraccini BM, et al. Common causes of hair loss—clinical manifestations, trichoscopy and therapy. J Eur Acad Dermatol Venereol. 2021;35:629-640.
- Ashique K, Kaliyadan F. Clinical photography for trichology practice: tips and tricks. Int J Trichology. 2011;3:7-13.
- Rudnicka L, Olszewska M, Rakowska A, et al. Trichoscopy: a new method for diagnosing hair loss. J Drugs Dermatol. 2008;7:651-654.
- Kinoshita-Ise M, Sachdeva M. Update on trichoscopy: integration of the terminology by systematic approach and a proposal of a diagnostic flowchart. J Dermatol. 2022;49:4-18. doi:10.1111/1346-8138.16233
- Van Neste D, Trüeb RM. Critical study of hair growth analysis with computer-assisted methods. J Eur Acad Dermatol Venereol. 2006;20:578-583.
- Romero J, Grimalt R. Trichoscopy: essentials for the dermatologist. World J Dermatol. 2015;4:63-68.
- Trichoscopy: a new frontier for the diagnosis of hair diseases. Exp Rev Dermatol. 2012;7:429-437.
- Lee B, Chan J, Monselise A, et al. Assessment of hair density and caliber in Caucasian and Asian female subjects with female pattern hair loss by using the Folliscope. J Am Acad Dermatol. 2012;66:166-167.
- Inui S. Trichoscopy for common hair loss diseases: algorithmic method for diagnosis. J Dermatol. 2010;38:71-75.
- Dhurat R. Phototrichogram. Indian J Dermatol Venereol Leprol. 2006;72:242-244.
- Agozzino M, Tosti A, Barbieri L, et al. Confocal microscopic features of scarring alopecia: preliminary report. Br J Dermatol. 2011;165:534-540.
- Kuck M, Schanzer S, Ulrich M, et al. Analysis of the efficiency of hair removal by different optical methods: comparison of Trichoscan, reflectance confocal microscopy, and optical coherence tomography. J Biomed Opt. 2012;17:101504.
- Levine A, Markowitz O. Introduction to reflectance confocal microscopy and its use in clinical practice. JAAD Case Rep. 2018;4:1014-1023.
- Agozzino M, Ardigò M. Scalp confocal microscopy. In: Humbert P, Maibach H, Fanian F, et al, eds. Agache’s Measuring the Skin: Non-invasive Investigations, Physiology, Normal Constants. 2nd ed. Springer International Publishing; 2016:311-326.
- Rudnicka L, Olszewska M, Rakowska A. In vivo reflectance confocal microscopy: usefulness for diagnosing hair diseases. J Dermatol Case Rep. 2008;2:55-59.
- Kurzeja M, Czuwara J, Walecka I, et al. Features of classic lichen planopilaris and frontal fibrosing alopecia in reflectance confocal microscopy: a preliminary study. Skin Res Technol. 2021;27:266-271.
- Ardigò M, Agozzino M, Franceschini C, et al. Reflectance confocal microscopy for scarring and non-scarring alopecia real-time assessment. Arch Dermatol Res. 2016;308:309-318.
- Franceschini C, Garelli V, Persechino F, et al. Dermoscopy and confocal microscopy for different chemotherapy-induced alopecia (CIA) phases characterization: preliminary study. Skin Res Technol. 2020;26:269-276.
- Martinez-Velasco MA, Perper M, Maddy AJ, et al. In vitro determination of Mexican Mestizo hair shaft diameter using optical coherence tomography. Skin Res Technol. 2018;24;274-277.
- Srivastava R, Manfredini M, Rao BK. Noninvasive imaging tools in dermatology. Cutis. 2019;104:108-113.
- Wan B, Ganier C, Du-Harpur X, et al. Applications and future directions for optical coherence tomography in dermatology. Br J Dermatol. 2021;184:1014-1022.
- Blume-Peytavi U, Vieten J, Knuttel A et al. Optical coherent tomography (OCT): a new method for online-measurement of hair shaft thickness. J Dtsch Dermatol Ges. 2004;2:546.
- Garcia Bartels N, Jahnke I, Patzelt A, et al. Hair shaft abnormalities in alopecia areata evaluated by optical coherence tomography. Skin Res Technol. 2011;17:201-205.
- Urban G, Feil N, Csuka E, et al. Combining deep learning with optical coherence tomography imaging to determine scalp hair and follicle counts. Lasers Surg Med. 2021;53:171-178.
- Vazquez-Herrera NE, Eber AE, Martinez-Velasco MA, et al. Optical coherence tomography for the investigation of frontal fibrosing alopecia. J Eur Acad Dermatol Venereol. 2018;32:318-322.
- Ekelem C, Feil N, Csuka E, et al. Optical coherence tomography in the evaluation of the scalp and hair: common features and clinical utility. Lasers Surg Med. 2021;53:129-140.
- Schicho K, Seemann R, Binder M, et al. Optical coherence tomography for planning of follicular unit extraction. Dermatol Surg. 2015;41:358-363.
- Canfield D. Photographic documentation of hair growth in androgenetic alopecia. Dermatol Clin. 1996;14:713-721.
- Peytavi U, Hillmann K, Guarrera M. Hair growth assessment techniques. In: Peytavi U, Hillmann K, Guarrera M, eds. Hair Growth and Disorders. 4th ed. Springer; 2008:140-144.
- Chamberlain AJ, Dawber RP. Methods of evaluating hair growth. Australas J Dermatol. 2003;44:10-18.
- Dhurat R, Saraogi P. Hair evaluation methods: merits and demerits. Int J Trichology. 2009;1:108-119.
- Kaufman KD, Olsen EA, Whiting D, et al. Finasteride in the treatment of men with androgenetic alopecia. J Am Acad Dermatol. 1998;39:578-579.
- Capily Institute. Artificial intelligence (A.I.) powered hair growth tracking. Accessed July 31, 2023. https://tss-aesthetics.com/capily-hair-tracking-syst
- Dinh Q, Sinclair R. Female pattern hair loss: current treatment concepts. Clin Interv Aging. 2007;2:189-199.
- Dhurat R, Saraogi P. Hair evaluation methods: merits and demerits. Int J Trichology. 2009;1:108-119.
- Wikramanayake TC, Mauro LM, Tabas IA, et al. Cross-section trichometry: a clinical tool for assessing the progression and treatment response of alopecia. Int J Trichology. 2012;4:259-264.
- Alessandrini A, Bruni F, Piraccini BM, et al. Common causes of hair loss—clinical manifestations, trichoscopy and therapy. J Eur Acad Dermatol Venereol. 2021;35:629-640.
- Ashique K, Kaliyadan F. Clinical photography for trichology practice: tips and tricks. Int J Trichology. 2011;3:7-13.
- Rudnicka L, Olszewska M, Rakowska A, et al. Trichoscopy: a new method for diagnosing hair loss. J Drugs Dermatol. 2008;7:651-654.
- Kinoshita-Ise M, Sachdeva M. Update on trichoscopy: integration of the terminology by systematic approach and a proposal of a diagnostic flowchart. J Dermatol. 2022;49:4-18. doi:10.1111/1346-8138.16233
- Van Neste D, Trüeb RM. Critical study of hair growth analysis with computer-assisted methods. J Eur Acad Dermatol Venereol. 2006;20:578-583.
- Romero J, Grimalt R. Trichoscopy: essentials for the dermatologist. World J Dermatol. 2015;4:63-68.
- Trichoscopy: a new frontier for the diagnosis of hair diseases. Exp Rev Dermatol. 2012;7:429-437.
- Lee B, Chan J, Monselise A, et al. Assessment of hair density and caliber in Caucasian and Asian female subjects with female pattern hair loss by using the Folliscope. J Am Acad Dermatol. 2012;66:166-167.
- Inui S. Trichoscopy for common hair loss diseases: algorithmic method for diagnosis. J Dermatol. 2010;38:71-75.
- Dhurat R. Phototrichogram. Indian J Dermatol Venereol Leprol. 2006;72:242-244.
- Agozzino M, Tosti A, Barbieri L, et al. Confocal microscopic features of scarring alopecia: preliminary report. Br J Dermatol. 2011;165:534-540.
- Kuck M, Schanzer S, Ulrich M, et al. Analysis of the efficiency of hair removal by different optical methods: comparison of Trichoscan, reflectance confocal microscopy, and optical coherence tomography. J Biomed Opt. 2012;17:101504.
- Levine A, Markowitz O. Introduction to reflectance confocal microscopy and its use in clinical practice. JAAD Case Rep. 2018;4:1014-1023.
- Agozzino M, Ardigò M. Scalp confocal microscopy. In: Humbert P, Maibach H, Fanian F, et al, eds. Agache’s Measuring the Skin: Non-invasive Investigations, Physiology, Normal Constants. 2nd ed. Springer International Publishing; 2016:311-326.
- Rudnicka L, Olszewska M, Rakowska A. In vivo reflectance confocal microscopy: usefulness for diagnosing hair diseases. J Dermatol Case Rep. 2008;2:55-59.
- Kurzeja M, Czuwara J, Walecka I, et al. Features of classic lichen planopilaris and frontal fibrosing alopecia in reflectance confocal microscopy: a preliminary study. Skin Res Technol. 2021;27:266-271.
- Ardigò M, Agozzino M, Franceschini C, et al. Reflectance confocal microscopy for scarring and non-scarring alopecia real-time assessment. Arch Dermatol Res. 2016;308:309-318.
- Franceschini C, Garelli V, Persechino F, et al. Dermoscopy and confocal microscopy for different chemotherapy-induced alopecia (CIA) phases characterization: preliminary study. Skin Res Technol. 2020;26:269-276.
- Martinez-Velasco MA, Perper M, Maddy AJ, et al. In vitro determination of Mexican Mestizo hair shaft diameter using optical coherence tomography. Skin Res Technol. 2018;24;274-277.
- Srivastava R, Manfredini M, Rao BK. Noninvasive imaging tools in dermatology. Cutis. 2019;104:108-113.
- Wan B, Ganier C, Du-Harpur X, et al. Applications and future directions for optical coherence tomography in dermatology. Br J Dermatol. 2021;184:1014-1022.
- Blume-Peytavi U, Vieten J, Knuttel A et al. Optical coherent tomography (OCT): a new method for online-measurement of hair shaft thickness. J Dtsch Dermatol Ges. 2004;2:546.
- Garcia Bartels N, Jahnke I, Patzelt A, et al. Hair shaft abnormalities in alopecia areata evaluated by optical coherence tomography. Skin Res Technol. 2011;17:201-205.
- Urban G, Feil N, Csuka E, et al. Combining deep learning with optical coherence tomography imaging to determine scalp hair and follicle counts. Lasers Surg Med. 2021;53:171-178.
- Vazquez-Herrera NE, Eber AE, Martinez-Velasco MA, et al. Optical coherence tomography for the investigation of frontal fibrosing alopecia. J Eur Acad Dermatol Venereol. 2018;32:318-322.
- Ekelem C, Feil N, Csuka E, et al. Optical coherence tomography in the evaluation of the scalp and hair: common features and clinical utility. Lasers Surg Med. 2021;53:129-140.
- Schicho K, Seemann R, Binder M, et al. Optical coherence tomography for planning of follicular unit extraction. Dermatol Surg. 2015;41:358-363.
Practice Points
- Reflectance confocal microscopy (RCM) imaging can be taken at levels from the stratum corneum to the papillary dermis and can be used to study scalp discoid lupus, lichen planopilaris, frontal fibrosing alopecia, alopecia areata, and androgenetic alopecia.
- Because of its ability to distinguish different stages of disease, RCM can be recommended as an intermediate step between trichoscopy and histology for the diagnosis and management of hair disease.
- Optical coherence tomography has the potential to monitor early subclinical responses to alopecia therapies while also improving hair transplantation outcomes by allowing for visualization of the subcutaneous angle of hair follicles.
- Software development paired with trichoscopy has the ability to quantify hair growth parameters such as hair count, density, and diameter.
Brachioradial Pruritus: An Etiologic Review and Treatment Summary
Brachioradial pruritus (BRP) is a neuropathic condition typically characterized by localized dysesthesia of the dorsolateral arms.1 This dysesthesia has been described as a persistent painful itching, burning, tingling, or stinging sensation2-4 and has a median duration of expression of 24 months.5,6 The condition may be unilateral or bilateral in nature but tends to have a predilection for a bilateral distribution along the C5 to C6 dermatomes.1,7,8 There are no primary skin lesions associated with BRP; however, excoriations, prurigo nodules, and lichenification may arise secondary to scratching of the irritated skin.1,4,5,9 Brachioradial pruritus tends to have a predilection for adult females (3:1 ratio) with lighter skin. The mean age at diagnosis is 59 years, but cases have been reported in patients aged 12 to 84 years.1,5 The diagnosis of BRP is based on clinical signs and symptoms, though the ice-pack sign tends to be pathognomonic for the diagnosis.10,11 Although there is no clear evidence on the exact cause of BRP, there are 2 prevalent theories: cervical radiculopathy secondary to cervical spine pathology and/or excessive exposure to UV radiation (UVR) in the summer months.3-5,12 Brachioradial pruritus remains poorly described in the literature, and even its origin is under debate. As such, the clinician may have difficulty deciding on the best course of management. The goal of this article is to identify and discuss known treatment options for BRP (Table).

Etiology
Cervical Spine Pathology—A correlation appears to exist between BRP and cervical spine changes seen on plain film radiographs at the levels of C3 to C7, with increased incidence at the C5 to C6 levels. These plain film radiographs typically show degenerative joint disease and neural foraminal stenosis at levels that correlate to the dermatomal distribution of BRP.1,7,10,12-14 In addition to plain film radiography, some studies have utilized magnetic resonance imaging to view the cervical spine and have documented evidence of intervertebral disc protrusion/bulging, central canal stenosis, neuroforaminal stenosis, and spondylosis at the affected regions.5,15-17 Moreover, supporting the theory that the cervical spine is responsible for the emergence of BRP, Marziniak et al17 investigated 41 patients with BRP utilizing magnetic resonance tomography to find that 33 patients (80.5%) had changes in nerve compression, and 8 patients (19.5%) had degenerative changes. In addition to these findings, they found that there was a significant correlation (P<.01) between the dermatomal expression of BRP and the location of cervical anatomical changes.17 Further validating the relationship between cervical spine pathology and BRP is a case study of a patient who saw rapid and complete resolution of the pruritus following spinal decompression surgery.10 Another case study described an intramedullary tumor found in a patient with BRP that was diagnosed as an ependymoma after magnetic resonance imaging revealed an intramedullary lesion within the spinal cord between C4 and C7. The location of the tumor and dermatomal pattern of the neuropathic itch pointed to a possible association between nerve compression and BRP.14 Electromyography studies performed on individuals with BRP have shown an increase in polyphasic units, decreased motor units, and/or denervation changes along the C5/C6 or C6 nerve roots, which provides additional support for the theory of cervical spine pathology as a causative factor for BRP.16
UVR Exposure—Another etiologic theory for BRP is that UVR exposure may be responsible for the genesis of pruritus. Previously known as solar pruritus, BRP was deemed a clinical condition, as there was increased prevalence in patients living in warmer climates, such as Florida.9 Wallengren and Dahlbäck18 reported that sun exposure is a notable factor in the onset of BRP, as they saw an increase in symptoms during the late summer and a decrease in symptoms over the winter months. To further support the theory that UVR is linked to BRP, several studies have shown that the utilization of sun protection is linked to a reduction of symptoms, specifically in patients who showed seasonal variations of their symptoms.9,12,19 Additionally, a study by Mirzoyev and Davis5 retrospectively reviewed 111 patients diagnosed with BRP. Of these patients, 84 (75.7%) presented with bilateral symptoms, and 54 (48.6%) reported prolonged sun exposure. Both of these findings demonstrate correlation between UVR and BRP.5 Interestingly, UV light exposure is known to release β-endorphin in the skin and may theoretically provide an area of exploration between UVR and cervical spine theories.
Conservative Treatment
Chiropractic Manipulation—Because one etiologic theory includes disease of the cervical spine, there is evidence that targeting this region with treatment is beneficial.7 Two case reports found in the literature noted that cervical spine manipulation and cervical traction yielded positive results.20,21 It has been established that pain generated by disc lesions can be the result of local nociceptive fiber activation, direct mechanical compression of the nerve roots, or inflammatory mediators.22 There are several postulated models describing the hypoalgesic effects of spinal manipulation, which contains both biomechanical and neurophysiological mechanisms. Biomechanical changes theorized to elicit analgesia include restoration of faulty biomechanical movement patterns, breaking up of periarticular adhesions, and reflexogenic muscle inhibition of hypertonic musculature. Hypothesized neurophysiological effects of joint manipulation include an increase in afferent information overwhelming the nociceptive input, reduction of temporal summation, and autonomic activation leading to non–opioid-induced hypoalgesia.23 Cervical traction is another plausible treatment for BRP, wherein the physiological effects of traction allow for a separation of vertebral bodies and expansion of the intervertebral foramen circumference, thus decreasing compression of the nerve roots.24
Acupuncture—Neurogenic pruritus, including BRP, is a group of conditions that have been treated using acupuncture. Acupuncture treatment consists of intramuscular needle stimulation and has been found to alleviate itching in patients with neurogenic pruritus. In 1 retrospective case series, acupuncture was used to treat 16 patients who were identified as having segmental pruritus. Acupuncture targeted the spasmed paravertebral muscles of the affected dermatomal levels as well as other regions of the body, and it was found that 12 patients (75%) experienced full resolution of symptoms. However, relapse did occur in 6 patients (37%) within 1 to 12 months following treatment.25 Multiple theories exist as to why acupuncture may help. One is that it relieves muscle spasms, which in turn relieves neural irritation of the spinal nerves as they traverse the respective paraspinal musculature. Another is that acupuncture decreases nociception by stimulating release of opioid peptides in the dorsal horn.26 A third proposed theory is that acupuncture acts on the afferent nerve fibers responsible for transmitting pain—Aδ and C fibers—activating these afferent nerves to produce an analgesic effect.27
Physiotherapy—The literature suggests that possible first-line therapies for neurogenic pruritus, including BRP and notalgia paresthetica, consist of noninvasive nondermatologic treatments that target cervical spine disease. Notalgia paresthetica and BRP have similar proposed mechanisms of nerve impingement; therefore, they often are grouped together when discussing proposed manual treatment options. Physiotherapy treatment includes cervical muscle strengthening, increased range of motion, application of cervical soft collars, massage, transcutaneous electronic nerve stimulation, and cervical traction.7 A study of 12 patients by Raison-Peyron et al28 in 1999 discussed the use of spinal and paraspinal ultrasound or radiation physiotherapy. Six patients underwent this treatment, and the symptoms subsided in 4 cases.28 Another study by Fleischer et al29 in 2011 discussed improvement in 2 patients with notalgia paresthetica by exercise involving active range of motion and strengthening.
Photoprotection—Avoidance of UVR exposure has been beneficial to some patients to reduce symptoms. Use of sunscreen and long-sleeved UV-protective clothing during outdoor activities or the warmer summer months may be beneficial.1
Medical Treatment
Medication—Because of the nonspecific clinical presentation of BRP, initial treatment often involves prescription of first-line antipruritic agents, including steroid creams and systemic antihistamines, both of which generally fail to provide symptom relief.1,30 Medications with neurologic mechanisms of action appear to provide potentially superior outcomes.
Topical interventions for BRP and related neurogenic pruritus have shown limited success. A case series evaluating capsaicin for pruritus offered only transient relief, likely because of its temporary hyperstimulatory and desensitizing effect on neuropeptides.7,33 In small populations, the use of topical antidepressants has yielded cutaneous and pathological relief for BRP. A case study of a 70-year-old woman evaluated the efficacy of a combination cream of ketamine and amitriptyline (a tricyclic antidepressant) yielding moderate pruritus improvement and notable improvement of secondary brachial skin lesions.34 Oral steroids also have shown success in the treatment of chronic pruritus; however, limited research is available on the efficacy of such medications for BRP, and the long-term use of oral steroids is limited by many side effects.30
Interventional Pain Procedure—A 2018 case series investigated 3 patients with a clinical diagnosis of BRP who were treated between 2010 and 2016 with
Surgery—There are multiple case studies in the literature that discuss
Conclusion
The pathogenesis of BRP continues to be an area of debate—it may be secondary to cervical spine disease or UVR. This review found there is more research pointing to cervical spine disease. There is an abundance of literature discussing both conservative and invasive treatment strategies, both of which carry benefits. Further research is needed to better establish the etiology of BRP so that formal treatment guidelines may be established.
Neuropathic itch can be a frustrating condition for providers and patients, and many treatment modalities often are tried before arriving at a helpful treatment for a particular patient. Clinicians who may encounter BRP in practice benefit from up-to-date literature reviews that provide a summary of management strategies.
- Robbins BA, Schmieder GJ. Brachioradial pruritus. StatPearls Publishing; 2020. Updated September 12, 2022. Accessed July 25, 2023. https://www.ncbi.nlm.nih.gov/books/NBK459321/
- Crevits L. Brachioradial pruritus—a peculiar neuropathic disorder. Clin Neurol Neurosurg. 2006;108:803-805.
- Lane J, McKenzie J, Spiegel J. Brachioradial pruritus: a case report and review of the literature. Cutis. 2008;81:37-40.
- Wallengren J. Brachioradial pruritus: a recurrent solar dermopathy. J Am Acad Dermatol. 1998;39:803-806.
- Mirzoyev S, Davis M. Brachioradial pruritus: Mayo Clinic experience over the past decade. Br J Dermatol. 2013;169:1007-1015.
- Pinto AC, Wachholz PA, Masuda PY, et al. Clinical, epidemiological and therapeutic profile of patients with brachioradial pruritus in a reference service in dermatology. An Bras Dermatol. 2016;91:549-551. doi:10.1590/abd1806-4841.201644767
- Alai NN, Skinner HB. Concurrent notalgia paresthetica and brachioradial pruritus associated with cervical degenerative disc disease. Cutis. 2018;102:185, 186, 189, 190.
- Atis¸ G, Bilir Kaya B. Pregabalin treatment of three cases with brachioradial pruritus. Dermatol Ther. 2017;30:e12459.
- Waisman M. Solar pruritus of the elbows (brachioradial summer pruritus). Arch Dermatol. 1968;98:481-485.
- Binder A, Fölster-Holst R, Sahan G, et al. A case of neuropathic brachioradial pruritus caused by cervical disc herniation. Nat Clin Pract Neurol. 2008;4:338-342.
- Bernhard JD, Bordeaux JS. Medical pearl: the ice-pack sign in brachioradial pruritus. J Am Acad Dermatol. 2005;52:1073.
- Veien N, Laurberg G. Brachioradial pruritus: a follow-up of 76 patients. Acta Derm Venereol. 2011;91:183-185.
- Mataix J, Silvestre JF, Climent JM, et al. Brachioradial pruritus as a symptom of cervical radiculopathy. Article in Spanish. Actas Dermosifiliogr. 2008;99:719-722.
- Kavak A, Dosoglu M. Can a spinal cord tumor cause brachioradial pruritus? J Am Acad Dermatol. 2002;46:437-440.
- Zeidler C, Pereira MP, Ständer S. Brachioradial pruritus successfully treated with intravenous naloxone. J Eur Acad Dermatol Venereol. 2023;37:e87-e89. doi:10.1111/jdv.18553
- Shields LB, Iyer VG, Zhang Y, et al. Brachioradial pruritus: clinical, electromyographic, and cervical MRI features in nine patients. Cureus. 2022;14:e21811. doi:10.7759/cureus.21811
- Marziniak M, Phan NQ, Raap U, et al. Brachioradial pruritus as a result of cervical spine pathology: the results of a magneticresonance tomography study. J Am Acad Dermatol. 2011;65:756-762. doi:10.1016/j.jaad.2010.07.036
- Wallengren J, Dahlbäck K. Familial brachioradial pruritus. Br J Dermatol. 2005;153:1016-1018.
- Salzmann SN, Okano I, Shue J, et al. Disabling pruritus in a patient with cervical stenosis. J Am Acad Orthop Surg Glob Res Rev. 2020;4:e19.00178. doi:10.5435/JAAOSGlobal-D-19-00178
- Golden KJ, Diana RM. A case of brachioradial pruritus treated with chiropractic and acupuncture. Case Rep Dermatol. 2022;14:93-97. doi:10.1159/000524054
- Tait CP, Grigg E, Quirk CJ. Brachioradial pruritus and cervical spine manipulation. Australas J Dermatol. 1998;39:168-170. doi:10.1111/j.1440-0960.1998.tb01274.x
- Freynhagen R, Baron R. The evaluation of neuropathic components in low back pain. Curr Pain Headache Rep. 2009;13:185-190. doi:10.1007/s11916-009-0032-y
- Gyer G, Michael J, Inklebarger J, et al. Spinal manipulation therapy: is it all about the brain? A current review of the neurophysiological effects of manipulation. J Integr Med. 2019;17:328-337. doi:10.1016/j.joim.2019.05.004
- Graham N, Gross A, Goldsmith CH, et al. Mechanical traction for neck pain with or without radiculopathy. Cochrane Database Syst Rev. 2008:CD006408. doi:10.1002/14651858.CD006408.pub2
- Stellon A. Neurogenic pruritus: an unrecognised problem? A retrospective case series of treatment by acupuncture. Acupunct Med. 2002;20:186-190. doi:10.1136/aim.20.4.186
- Bowsher D. Mechanisms of acupuncture. In: Filshie J, White A, eds. Medical Acupuncture: A Western Scientific Approach. Churchill Livingstone; 1998:69-82.
- Lim TK, Ma Y, Berger F, et al. Acupuncture and neural mechanism in the management of low back pain-an update. Medicines (Basel). 2018;5:63.
- Raison-Peyron N, Meunier L, Acevedo M, et al. Notalgia paresthetica: clinical, physiopathological and therapeutic aspects. a study of 12 cases. J Eur Acad Dermatol Venereol. 1999;12:215-221.
- Fleischer AB, Meade TJ, Fleischer AB. Notalgia paresthetica: successful treatment with exercises. Acta Derm Venereol. 2011;91:356-357. doi:10.2340/00015555-1039
- Kouwenhoven TA, van de Kerkhof PCM, Kamsteeg M. Use of oral antidepressants in patients with chronic pruritus: a systematic review. J Am Acad Dermatol. 2017;77:1068-1073.e7. doi:10.1016/j.jaad.2017.08.025
- Matsuda KM, Sharma D, Schonfeld AR, et al. Gabapentin and pregabalin for the treatment of chronic pruritus. J Am Acad Dermatol. 2016;75:619-625.e6. doi:10.1016/j.jaad.2016.02.1237
- Okuno S, Hashimoto T, Satoh T. Case of neuropathic itch-associated prurigo nodules on the bilateral upper arms after unilateral herpes zoster in a patient with cervical herniated discs: successful treatment with mirogabalin. J Dermatol. 2021;48:e585-e586.
- Papoiu AD, Yosipovitch G. Topical capsaicin. The fire of a ‘hot’ medicine is reignited. Expert Opin Pharmacother. 2010;11:1359-1371. doi:10.1517/14656566.2010.481670
- Magazin M, Daze RP, Okeson N. Treatment refractory brachioradial pruritus treated with topical amitriptyline and ketamine. Cureus. 2019;11:e5117. doi:10.7759/cureus.5117
- Weinberg BD, Amans M, Deviren S, et al. Brachioradial pruritus treated with computed tomography-guided cervical nerve root block: a case series. JAAD Case Rep. 2018;4:640-644. doi:10.1016/j.jdcr.2018.03.025
- De Ridder D, Hans G, Pals P, et al. A C-fiber-mediated neuropathic brachioradial pruritus. J Neurosurg. 2010;113:118-121. doi:10.3171/2009.9.JNS09620
- Morosanu CO, Etim G, Alalade AF. Brachioradial pruritus secondary to cervical disc protrusion—a case report. J Surg Case Rep. 2022:rjac277. doi:10.1093/jscr/rjac277
Brachioradial pruritus (BRP) is a neuropathic condition typically characterized by localized dysesthesia of the dorsolateral arms.1 This dysesthesia has been described as a persistent painful itching, burning, tingling, or stinging sensation2-4 and has a median duration of expression of 24 months.5,6 The condition may be unilateral or bilateral in nature but tends to have a predilection for a bilateral distribution along the C5 to C6 dermatomes.1,7,8 There are no primary skin lesions associated with BRP; however, excoriations, prurigo nodules, and lichenification may arise secondary to scratching of the irritated skin.1,4,5,9 Brachioradial pruritus tends to have a predilection for adult females (3:1 ratio) with lighter skin. The mean age at diagnosis is 59 years, but cases have been reported in patients aged 12 to 84 years.1,5 The diagnosis of BRP is based on clinical signs and symptoms, though the ice-pack sign tends to be pathognomonic for the diagnosis.10,11 Although there is no clear evidence on the exact cause of BRP, there are 2 prevalent theories: cervical radiculopathy secondary to cervical spine pathology and/or excessive exposure to UV radiation (UVR) in the summer months.3-5,12 Brachioradial pruritus remains poorly described in the literature, and even its origin is under debate. As such, the clinician may have difficulty deciding on the best course of management. The goal of this article is to identify and discuss known treatment options for BRP (Table).

Etiology
Cervical Spine Pathology—A correlation appears to exist between BRP and cervical spine changes seen on plain film radiographs at the levels of C3 to C7, with increased incidence at the C5 to C6 levels. These plain film radiographs typically show degenerative joint disease and neural foraminal stenosis at levels that correlate to the dermatomal distribution of BRP.1,7,10,12-14 In addition to plain film radiography, some studies have utilized magnetic resonance imaging to view the cervical spine and have documented evidence of intervertebral disc protrusion/bulging, central canal stenosis, neuroforaminal stenosis, and spondylosis at the affected regions.5,15-17 Moreover, supporting the theory that the cervical spine is responsible for the emergence of BRP, Marziniak et al17 investigated 41 patients with BRP utilizing magnetic resonance tomography to find that 33 patients (80.5%) had changes in nerve compression, and 8 patients (19.5%) had degenerative changes. In addition to these findings, they found that there was a significant correlation (P<.01) between the dermatomal expression of BRP and the location of cervical anatomical changes.17 Further validating the relationship between cervical spine pathology and BRP is a case study of a patient who saw rapid and complete resolution of the pruritus following spinal decompression surgery.10 Another case study described an intramedullary tumor found in a patient with BRP that was diagnosed as an ependymoma after magnetic resonance imaging revealed an intramedullary lesion within the spinal cord between C4 and C7. The location of the tumor and dermatomal pattern of the neuropathic itch pointed to a possible association between nerve compression and BRP.14 Electromyography studies performed on individuals with BRP have shown an increase in polyphasic units, decreased motor units, and/or denervation changes along the C5/C6 or C6 nerve roots, which provides additional support for the theory of cervical spine pathology as a causative factor for BRP.16
UVR Exposure—Another etiologic theory for BRP is that UVR exposure may be responsible for the genesis of pruritus. Previously known as solar pruritus, BRP was deemed a clinical condition, as there was increased prevalence in patients living in warmer climates, such as Florida.9 Wallengren and Dahlbäck18 reported that sun exposure is a notable factor in the onset of BRP, as they saw an increase in symptoms during the late summer and a decrease in symptoms over the winter months. To further support the theory that UVR is linked to BRP, several studies have shown that the utilization of sun protection is linked to a reduction of symptoms, specifically in patients who showed seasonal variations of their symptoms.9,12,19 Additionally, a study by Mirzoyev and Davis5 retrospectively reviewed 111 patients diagnosed with BRP. Of these patients, 84 (75.7%) presented with bilateral symptoms, and 54 (48.6%) reported prolonged sun exposure. Both of these findings demonstrate correlation between UVR and BRP.5 Interestingly, UV light exposure is known to release β-endorphin in the skin and may theoretically provide an area of exploration between UVR and cervical spine theories.
Conservative Treatment
Chiropractic Manipulation—Because one etiologic theory includes disease of the cervical spine, there is evidence that targeting this region with treatment is beneficial.7 Two case reports found in the literature noted that cervical spine manipulation and cervical traction yielded positive results.20,21 It has been established that pain generated by disc lesions can be the result of local nociceptive fiber activation, direct mechanical compression of the nerve roots, or inflammatory mediators.22 There are several postulated models describing the hypoalgesic effects of spinal manipulation, which contains both biomechanical and neurophysiological mechanisms. Biomechanical changes theorized to elicit analgesia include restoration of faulty biomechanical movement patterns, breaking up of periarticular adhesions, and reflexogenic muscle inhibition of hypertonic musculature. Hypothesized neurophysiological effects of joint manipulation include an increase in afferent information overwhelming the nociceptive input, reduction of temporal summation, and autonomic activation leading to non–opioid-induced hypoalgesia.23 Cervical traction is another plausible treatment for BRP, wherein the physiological effects of traction allow for a separation of vertebral bodies and expansion of the intervertebral foramen circumference, thus decreasing compression of the nerve roots.24
Acupuncture—Neurogenic pruritus, including BRP, is a group of conditions that have been treated using acupuncture. Acupuncture treatment consists of intramuscular needle stimulation and has been found to alleviate itching in patients with neurogenic pruritus. In 1 retrospective case series, acupuncture was used to treat 16 patients who were identified as having segmental pruritus. Acupuncture targeted the spasmed paravertebral muscles of the affected dermatomal levels as well as other regions of the body, and it was found that 12 patients (75%) experienced full resolution of symptoms. However, relapse did occur in 6 patients (37%) within 1 to 12 months following treatment.25 Multiple theories exist as to why acupuncture may help. One is that it relieves muscle spasms, which in turn relieves neural irritation of the spinal nerves as they traverse the respective paraspinal musculature. Another is that acupuncture decreases nociception by stimulating release of opioid peptides in the dorsal horn.26 A third proposed theory is that acupuncture acts on the afferent nerve fibers responsible for transmitting pain—Aδ and C fibers—activating these afferent nerves to produce an analgesic effect.27
Physiotherapy—The literature suggests that possible first-line therapies for neurogenic pruritus, including BRP and notalgia paresthetica, consist of noninvasive nondermatologic treatments that target cervical spine disease. Notalgia paresthetica and BRP have similar proposed mechanisms of nerve impingement; therefore, they often are grouped together when discussing proposed manual treatment options. Physiotherapy treatment includes cervical muscle strengthening, increased range of motion, application of cervical soft collars, massage, transcutaneous electronic nerve stimulation, and cervical traction.7 A study of 12 patients by Raison-Peyron et al28 in 1999 discussed the use of spinal and paraspinal ultrasound or radiation physiotherapy. Six patients underwent this treatment, and the symptoms subsided in 4 cases.28 Another study by Fleischer et al29 in 2011 discussed improvement in 2 patients with notalgia paresthetica by exercise involving active range of motion and strengthening.
Photoprotection—Avoidance of UVR exposure has been beneficial to some patients to reduce symptoms. Use of sunscreen and long-sleeved UV-protective clothing during outdoor activities or the warmer summer months may be beneficial.1
Medical Treatment
Medication—Because of the nonspecific clinical presentation of BRP, initial treatment often involves prescription of first-line antipruritic agents, including steroid creams and systemic antihistamines, both of which generally fail to provide symptom relief.1,30 Medications with neurologic mechanisms of action appear to provide potentially superior outcomes.
Topical interventions for BRP and related neurogenic pruritus have shown limited success. A case series evaluating capsaicin for pruritus offered only transient relief, likely because of its temporary hyperstimulatory and desensitizing effect on neuropeptides.7,33 In small populations, the use of topical antidepressants has yielded cutaneous and pathological relief for BRP. A case study of a 70-year-old woman evaluated the efficacy of a combination cream of ketamine and amitriptyline (a tricyclic antidepressant) yielding moderate pruritus improvement and notable improvement of secondary brachial skin lesions.34 Oral steroids also have shown success in the treatment of chronic pruritus; however, limited research is available on the efficacy of such medications for BRP, and the long-term use of oral steroids is limited by many side effects.30
Interventional Pain Procedure—A 2018 case series investigated 3 patients with a clinical diagnosis of BRP who were treated between 2010 and 2016 with
Surgery—There are multiple case studies in the literature that discuss
Conclusion
The pathogenesis of BRP continues to be an area of debate—it may be secondary to cervical spine disease or UVR. This review found there is more research pointing to cervical spine disease. There is an abundance of literature discussing both conservative and invasive treatment strategies, both of which carry benefits. Further research is needed to better establish the etiology of BRP so that formal treatment guidelines may be established.
Neuropathic itch can be a frustrating condition for providers and patients, and many treatment modalities often are tried before arriving at a helpful treatment for a particular patient. Clinicians who may encounter BRP in practice benefit from up-to-date literature reviews that provide a summary of management strategies.
Brachioradial pruritus (BRP) is a neuropathic condition typically characterized by localized dysesthesia of the dorsolateral arms.1 This dysesthesia has been described as a persistent painful itching, burning, tingling, or stinging sensation2-4 and has a median duration of expression of 24 months.5,6 The condition may be unilateral or bilateral in nature but tends to have a predilection for a bilateral distribution along the C5 to C6 dermatomes.1,7,8 There are no primary skin lesions associated with BRP; however, excoriations, prurigo nodules, and lichenification may arise secondary to scratching of the irritated skin.1,4,5,9 Brachioradial pruritus tends to have a predilection for adult females (3:1 ratio) with lighter skin. The mean age at diagnosis is 59 years, but cases have been reported in patients aged 12 to 84 years.1,5 The diagnosis of BRP is based on clinical signs and symptoms, though the ice-pack sign tends to be pathognomonic for the diagnosis.10,11 Although there is no clear evidence on the exact cause of BRP, there are 2 prevalent theories: cervical radiculopathy secondary to cervical spine pathology and/or excessive exposure to UV radiation (UVR) in the summer months.3-5,12 Brachioradial pruritus remains poorly described in the literature, and even its origin is under debate. As such, the clinician may have difficulty deciding on the best course of management. The goal of this article is to identify and discuss known treatment options for BRP (Table).

Etiology
Cervical Spine Pathology—A correlation appears to exist between BRP and cervical spine changes seen on plain film radiographs at the levels of C3 to C7, with increased incidence at the C5 to C6 levels. These plain film radiographs typically show degenerative joint disease and neural foraminal stenosis at levels that correlate to the dermatomal distribution of BRP.1,7,10,12-14 In addition to plain film radiography, some studies have utilized magnetic resonance imaging to view the cervical spine and have documented evidence of intervertebral disc protrusion/bulging, central canal stenosis, neuroforaminal stenosis, and spondylosis at the affected regions.5,15-17 Moreover, supporting the theory that the cervical spine is responsible for the emergence of BRP, Marziniak et al17 investigated 41 patients with BRP utilizing magnetic resonance tomography to find that 33 patients (80.5%) had changes in nerve compression, and 8 patients (19.5%) had degenerative changes. In addition to these findings, they found that there was a significant correlation (P<.01) between the dermatomal expression of BRP and the location of cervical anatomical changes.17 Further validating the relationship between cervical spine pathology and BRP is a case study of a patient who saw rapid and complete resolution of the pruritus following spinal decompression surgery.10 Another case study described an intramedullary tumor found in a patient with BRP that was diagnosed as an ependymoma after magnetic resonance imaging revealed an intramedullary lesion within the spinal cord between C4 and C7. The location of the tumor and dermatomal pattern of the neuropathic itch pointed to a possible association between nerve compression and BRP.14 Electromyography studies performed on individuals with BRP have shown an increase in polyphasic units, decreased motor units, and/or denervation changes along the C5/C6 or C6 nerve roots, which provides additional support for the theory of cervical spine pathology as a causative factor for BRP.16
UVR Exposure—Another etiologic theory for BRP is that UVR exposure may be responsible for the genesis of pruritus. Previously known as solar pruritus, BRP was deemed a clinical condition, as there was increased prevalence in patients living in warmer climates, such as Florida.9 Wallengren and Dahlbäck18 reported that sun exposure is a notable factor in the onset of BRP, as they saw an increase in symptoms during the late summer and a decrease in symptoms over the winter months. To further support the theory that UVR is linked to BRP, several studies have shown that the utilization of sun protection is linked to a reduction of symptoms, specifically in patients who showed seasonal variations of their symptoms.9,12,19 Additionally, a study by Mirzoyev and Davis5 retrospectively reviewed 111 patients diagnosed with BRP. Of these patients, 84 (75.7%) presented with bilateral symptoms, and 54 (48.6%) reported prolonged sun exposure. Both of these findings demonstrate correlation between UVR and BRP.5 Interestingly, UV light exposure is known to release β-endorphin in the skin and may theoretically provide an area of exploration between UVR and cervical spine theories.
Conservative Treatment
Chiropractic Manipulation—Because one etiologic theory includes disease of the cervical spine, there is evidence that targeting this region with treatment is beneficial.7 Two case reports found in the literature noted that cervical spine manipulation and cervical traction yielded positive results.20,21 It has been established that pain generated by disc lesions can be the result of local nociceptive fiber activation, direct mechanical compression of the nerve roots, or inflammatory mediators.22 There are several postulated models describing the hypoalgesic effects of spinal manipulation, which contains both biomechanical and neurophysiological mechanisms. Biomechanical changes theorized to elicit analgesia include restoration of faulty biomechanical movement patterns, breaking up of periarticular adhesions, and reflexogenic muscle inhibition of hypertonic musculature. Hypothesized neurophysiological effects of joint manipulation include an increase in afferent information overwhelming the nociceptive input, reduction of temporal summation, and autonomic activation leading to non–opioid-induced hypoalgesia.23 Cervical traction is another plausible treatment for BRP, wherein the physiological effects of traction allow for a separation of vertebral bodies and expansion of the intervertebral foramen circumference, thus decreasing compression of the nerve roots.24
Acupuncture—Neurogenic pruritus, including BRP, is a group of conditions that have been treated using acupuncture. Acupuncture treatment consists of intramuscular needle stimulation and has been found to alleviate itching in patients with neurogenic pruritus. In 1 retrospective case series, acupuncture was used to treat 16 patients who were identified as having segmental pruritus. Acupuncture targeted the spasmed paravertebral muscles of the affected dermatomal levels as well as other regions of the body, and it was found that 12 patients (75%) experienced full resolution of symptoms. However, relapse did occur in 6 patients (37%) within 1 to 12 months following treatment.25 Multiple theories exist as to why acupuncture may help. One is that it relieves muscle spasms, which in turn relieves neural irritation of the spinal nerves as they traverse the respective paraspinal musculature. Another is that acupuncture decreases nociception by stimulating release of opioid peptides in the dorsal horn.26 A third proposed theory is that acupuncture acts on the afferent nerve fibers responsible for transmitting pain—Aδ and C fibers—activating these afferent nerves to produce an analgesic effect.27
Physiotherapy—The literature suggests that possible first-line therapies for neurogenic pruritus, including BRP and notalgia paresthetica, consist of noninvasive nondermatologic treatments that target cervical spine disease. Notalgia paresthetica and BRP have similar proposed mechanisms of nerve impingement; therefore, they often are grouped together when discussing proposed manual treatment options. Physiotherapy treatment includes cervical muscle strengthening, increased range of motion, application of cervical soft collars, massage, transcutaneous electronic nerve stimulation, and cervical traction.7 A study of 12 patients by Raison-Peyron et al28 in 1999 discussed the use of spinal and paraspinal ultrasound or radiation physiotherapy. Six patients underwent this treatment, and the symptoms subsided in 4 cases.28 Another study by Fleischer et al29 in 2011 discussed improvement in 2 patients with notalgia paresthetica by exercise involving active range of motion and strengthening.
Photoprotection—Avoidance of UVR exposure has been beneficial to some patients to reduce symptoms. Use of sunscreen and long-sleeved UV-protective clothing during outdoor activities or the warmer summer months may be beneficial.1
Medical Treatment
Medication—Because of the nonspecific clinical presentation of BRP, initial treatment often involves prescription of first-line antipruritic agents, including steroid creams and systemic antihistamines, both of which generally fail to provide symptom relief.1,30 Medications with neurologic mechanisms of action appear to provide potentially superior outcomes.
Topical interventions for BRP and related neurogenic pruritus have shown limited success. A case series evaluating capsaicin for pruritus offered only transient relief, likely because of its temporary hyperstimulatory and desensitizing effect on neuropeptides.7,33 In small populations, the use of topical antidepressants has yielded cutaneous and pathological relief for BRP. A case study of a 70-year-old woman evaluated the efficacy of a combination cream of ketamine and amitriptyline (a tricyclic antidepressant) yielding moderate pruritus improvement and notable improvement of secondary brachial skin lesions.34 Oral steroids also have shown success in the treatment of chronic pruritus; however, limited research is available on the efficacy of such medications for BRP, and the long-term use of oral steroids is limited by many side effects.30
Interventional Pain Procedure—A 2018 case series investigated 3 patients with a clinical diagnosis of BRP who were treated between 2010 and 2016 with
Surgery—There are multiple case studies in the literature that discuss
Conclusion
The pathogenesis of BRP continues to be an area of debate—it may be secondary to cervical spine disease or UVR. This review found there is more research pointing to cervical spine disease. There is an abundance of literature discussing both conservative and invasive treatment strategies, both of which carry benefits. Further research is needed to better establish the etiology of BRP so that formal treatment guidelines may be established.
Neuropathic itch can be a frustrating condition for providers and patients, and many treatment modalities often are tried before arriving at a helpful treatment for a particular patient. Clinicians who may encounter BRP in practice benefit from up-to-date literature reviews that provide a summary of management strategies.
- Robbins BA, Schmieder GJ. Brachioradial pruritus. StatPearls Publishing; 2020. Updated September 12, 2022. Accessed July 25, 2023. https://www.ncbi.nlm.nih.gov/books/NBK459321/
- Crevits L. Brachioradial pruritus—a peculiar neuropathic disorder. Clin Neurol Neurosurg. 2006;108:803-805.
- Lane J, McKenzie J, Spiegel J. Brachioradial pruritus: a case report and review of the literature. Cutis. 2008;81:37-40.
- Wallengren J. Brachioradial pruritus: a recurrent solar dermopathy. J Am Acad Dermatol. 1998;39:803-806.
- Mirzoyev S, Davis M. Brachioradial pruritus: Mayo Clinic experience over the past decade. Br J Dermatol. 2013;169:1007-1015.
- Pinto AC, Wachholz PA, Masuda PY, et al. Clinical, epidemiological and therapeutic profile of patients with brachioradial pruritus in a reference service in dermatology. An Bras Dermatol. 2016;91:549-551. doi:10.1590/abd1806-4841.201644767
- Alai NN, Skinner HB. Concurrent notalgia paresthetica and brachioradial pruritus associated with cervical degenerative disc disease. Cutis. 2018;102:185, 186, 189, 190.
- Atis¸ G, Bilir Kaya B. Pregabalin treatment of three cases with brachioradial pruritus. Dermatol Ther. 2017;30:e12459.
- Waisman M. Solar pruritus of the elbows (brachioradial summer pruritus). Arch Dermatol. 1968;98:481-485.
- Binder A, Fölster-Holst R, Sahan G, et al. A case of neuropathic brachioradial pruritus caused by cervical disc herniation. Nat Clin Pract Neurol. 2008;4:338-342.
- Bernhard JD, Bordeaux JS. Medical pearl: the ice-pack sign in brachioradial pruritus. J Am Acad Dermatol. 2005;52:1073.
- Veien N, Laurberg G. Brachioradial pruritus: a follow-up of 76 patients. Acta Derm Venereol. 2011;91:183-185.
- Mataix J, Silvestre JF, Climent JM, et al. Brachioradial pruritus as a symptom of cervical radiculopathy. Article in Spanish. Actas Dermosifiliogr. 2008;99:719-722.
- Kavak A, Dosoglu M. Can a spinal cord tumor cause brachioradial pruritus? J Am Acad Dermatol. 2002;46:437-440.
- Zeidler C, Pereira MP, Ständer S. Brachioradial pruritus successfully treated with intravenous naloxone. J Eur Acad Dermatol Venereol. 2023;37:e87-e89. doi:10.1111/jdv.18553
- Shields LB, Iyer VG, Zhang Y, et al. Brachioradial pruritus: clinical, electromyographic, and cervical MRI features in nine patients. Cureus. 2022;14:e21811. doi:10.7759/cureus.21811
- Marziniak M, Phan NQ, Raap U, et al. Brachioradial pruritus as a result of cervical spine pathology: the results of a magneticresonance tomography study. J Am Acad Dermatol. 2011;65:756-762. doi:10.1016/j.jaad.2010.07.036
- Wallengren J, Dahlbäck K. Familial brachioradial pruritus. Br J Dermatol. 2005;153:1016-1018.
- Salzmann SN, Okano I, Shue J, et al. Disabling pruritus in a patient with cervical stenosis. J Am Acad Orthop Surg Glob Res Rev. 2020;4:e19.00178. doi:10.5435/JAAOSGlobal-D-19-00178
- Golden KJ, Diana RM. A case of brachioradial pruritus treated with chiropractic and acupuncture. Case Rep Dermatol. 2022;14:93-97. doi:10.1159/000524054
- Tait CP, Grigg E, Quirk CJ. Brachioradial pruritus and cervical spine manipulation. Australas J Dermatol. 1998;39:168-170. doi:10.1111/j.1440-0960.1998.tb01274.x
- Freynhagen R, Baron R. The evaluation of neuropathic components in low back pain. Curr Pain Headache Rep. 2009;13:185-190. doi:10.1007/s11916-009-0032-y
- Gyer G, Michael J, Inklebarger J, et al. Spinal manipulation therapy: is it all about the brain? A current review of the neurophysiological effects of manipulation. J Integr Med. 2019;17:328-337. doi:10.1016/j.joim.2019.05.004
- Graham N, Gross A, Goldsmith CH, et al. Mechanical traction for neck pain with or without radiculopathy. Cochrane Database Syst Rev. 2008:CD006408. doi:10.1002/14651858.CD006408.pub2
- Stellon A. Neurogenic pruritus: an unrecognised problem? A retrospective case series of treatment by acupuncture. Acupunct Med. 2002;20:186-190. doi:10.1136/aim.20.4.186
- Bowsher D. Mechanisms of acupuncture. In: Filshie J, White A, eds. Medical Acupuncture: A Western Scientific Approach. Churchill Livingstone; 1998:69-82.
- Lim TK, Ma Y, Berger F, et al. Acupuncture and neural mechanism in the management of low back pain-an update. Medicines (Basel). 2018;5:63.
- Raison-Peyron N, Meunier L, Acevedo M, et al. Notalgia paresthetica: clinical, physiopathological and therapeutic aspects. a study of 12 cases. J Eur Acad Dermatol Venereol. 1999;12:215-221.
- Fleischer AB, Meade TJ, Fleischer AB. Notalgia paresthetica: successful treatment with exercises. Acta Derm Venereol. 2011;91:356-357. doi:10.2340/00015555-1039
- Kouwenhoven TA, van de Kerkhof PCM, Kamsteeg M. Use of oral antidepressants in patients with chronic pruritus: a systematic review. J Am Acad Dermatol. 2017;77:1068-1073.e7. doi:10.1016/j.jaad.2017.08.025
- Matsuda KM, Sharma D, Schonfeld AR, et al. Gabapentin and pregabalin for the treatment of chronic pruritus. J Am Acad Dermatol. 2016;75:619-625.e6. doi:10.1016/j.jaad.2016.02.1237
- Okuno S, Hashimoto T, Satoh T. Case of neuropathic itch-associated prurigo nodules on the bilateral upper arms after unilateral herpes zoster in a patient with cervical herniated discs: successful treatment with mirogabalin. J Dermatol. 2021;48:e585-e586.
- Papoiu AD, Yosipovitch G. Topical capsaicin. The fire of a ‘hot’ medicine is reignited. Expert Opin Pharmacother. 2010;11:1359-1371. doi:10.1517/14656566.2010.481670
- Magazin M, Daze RP, Okeson N. Treatment refractory brachioradial pruritus treated with topical amitriptyline and ketamine. Cureus. 2019;11:e5117. doi:10.7759/cureus.5117
- Weinberg BD, Amans M, Deviren S, et al. Brachioradial pruritus treated with computed tomography-guided cervical nerve root block: a case series. JAAD Case Rep. 2018;4:640-644. doi:10.1016/j.jdcr.2018.03.025
- De Ridder D, Hans G, Pals P, et al. A C-fiber-mediated neuropathic brachioradial pruritus. J Neurosurg. 2010;113:118-121. doi:10.3171/2009.9.JNS09620
- Morosanu CO, Etim G, Alalade AF. Brachioradial pruritus secondary to cervical disc protrusion—a case report. J Surg Case Rep. 2022:rjac277. doi:10.1093/jscr/rjac277
- Robbins BA, Schmieder GJ. Brachioradial pruritus. StatPearls Publishing; 2020. Updated September 12, 2022. Accessed July 25, 2023. https://www.ncbi.nlm.nih.gov/books/NBK459321/
- Crevits L. Brachioradial pruritus—a peculiar neuropathic disorder. Clin Neurol Neurosurg. 2006;108:803-805.
- Lane J, McKenzie J, Spiegel J. Brachioradial pruritus: a case report and review of the literature. Cutis. 2008;81:37-40.
- Wallengren J. Brachioradial pruritus: a recurrent solar dermopathy. J Am Acad Dermatol. 1998;39:803-806.
- Mirzoyev S, Davis M. Brachioradial pruritus: Mayo Clinic experience over the past decade. Br J Dermatol. 2013;169:1007-1015.
- Pinto AC, Wachholz PA, Masuda PY, et al. Clinical, epidemiological and therapeutic profile of patients with brachioradial pruritus in a reference service in dermatology. An Bras Dermatol. 2016;91:549-551. doi:10.1590/abd1806-4841.201644767
- Alai NN, Skinner HB. Concurrent notalgia paresthetica and brachioradial pruritus associated with cervical degenerative disc disease. Cutis. 2018;102:185, 186, 189, 190.
- Atis¸ G, Bilir Kaya B. Pregabalin treatment of three cases with brachioradial pruritus. Dermatol Ther. 2017;30:e12459.
- Waisman M. Solar pruritus of the elbows (brachioradial summer pruritus). Arch Dermatol. 1968;98:481-485.
- Binder A, Fölster-Holst R, Sahan G, et al. A case of neuropathic brachioradial pruritus caused by cervical disc herniation. Nat Clin Pract Neurol. 2008;4:338-342.
- Bernhard JD, Bordeaux JS. Medical pearl: the ice-pack sign in brachioradial pruritus. J Am Acad Dermatol. 2005;52:1073.
- Veien N, Laurberg G. Brachioradial pruritus: a follow-up of 76 patients. Acta Derm Venereol. 2011;91:183-185.
- Mataix J, Silvestre JF, Climent JM, et al. Brachioradial pruritus as a symptom of cervical radiculopathy. Article in Spanish. Actas Dermosifiliogr. 2008;99:719-722.
- Kavak A, Dosoglu M. Can a spinal cord tumor cause brachioradial pruritus? J Am Acad Dermatol. 2002;46:437-440.
- Zeidler C, Pereira MP, Ständer S. Brachioradial pruritus successfully treated with intravenous naloxone. J Eur Acad Dermatol Venereol. 2023;37:e87-e89. doi:10.1111/jdv.18553
- Shields LB, Iyer VG, Zhang Y, et al. Brachioradial pruritus: clinical, electromyographic, and cervical MRI features in nine patients. Cureus. 2022;14:e21811. doi:10.7759/cureus.21811
- Marziniak M, Phan NQ, Raap U, et al. Brachioradial pruritus as a result of cervical spine pathology: the results of a magneticresonance tomography study. J Am Acad Dermatol. 2011;65:756-762. doi:10.1016/j.jaad.2010.07.036
- Wallengren J, Dahlbäck K. Familial brachioradial pruritus. Br J Dermatol. 2005;153:1016-1018.
- Salzmann SN, Okano I, Shue J, et al. Disabling pruritus in a patient with cervical stenosis. J Am Acad Orthop Surg Glob Res Rev. 2020;4:e19.00178. doi:10.5435/JAAOSGlobal-D-19-00178
- Golden KJ, Diana RM. A case of brachioradial pruritus treated with chiropractic and acupuncture. Case Rep Dermatol. 2022;14:93-97. doi:10.1159/000524054
- Tait CP, Grigg E, Quirk CJ. Brachioradial pruritus and cervical spine manipulation. Australas J Dermatol. 1998;39:168-170. doi:10.1111/j.1440-0960.1998.tb01274.x
- Freynhagen R, Baron R. The evaluation of neuropathic components in low back pain. Curr Pain Headache Rep. 2009;13:185-190. doi:10.1007/s11916-009-0032-y
- Gyer G, Michael J, Inklebarger J, et al. Spinal manipulation therapy: is it all about the brain? A current review of the neurophysiological effects of manipulation. J Integr Med. 2019;17:328-337. doi:10.1016/j.joim.2019.05.004
- Graham N, Gross A, Goldsmith CH, et al. Mechanical traction for neck pain with or without radiculopathy. Cochrane Database Syst Rev. 2008:CD006408. doi:10.1002/14651858.CD006408.pub2
- Stellon A. Neurogenic pruritus: an unrecognised problem? A retrospective case series of treatment by acupuncture. Acupunct Med. 2002;20:186-190. doi:10.1136/aim.20.4.186
- Bowsher D. Mechanisms of acupuncture. In: Filshie J, White A, eds. Medical Acupuncture: A Western Scientific Approach. Churchill Livingstone; 1998:69-82.
- Lim TK, Ma Y, Berger F, et al. Acupuncture and neural mechanism in the management of low back pain-an update. Medicines (Basel). 2018;5:63.
- Raison-Peyron N, Meunier L, Acevedo M, et al. Notalgia paresthetica: clinical, physiopathological and therapeutic aspects. a study of 12 cases. J Eur Acad Dermatol Venereol. 1999;12:215-221.
- Fleischer AB, Meade TJ, Fleischer AB. Notalgia paresthetica: successful treatment with exercises. Acta Derm Venereol. 2011;91:356-357. doi:10.2340/00015555-1039
- Kouwenhoven TA, van de Kerkhof PCM, Kamsteeg M. Use of oral antidepressants in patients with chronic pruritus: a systematic review. J Am Acad Dermatol. 2017;77:1068-1073.e7. doi:10.1016/j.jaad.2017.08.025
- Matsuda KM, Sharma D, Schonfeld AR, et al. Gabapentin and pregabalin for the treatment of chronic pruritus. J Am Acad Dermatol. 2016;75:619-625.e6. doi:10.1016/j.jaad.2016.02.1237
- Okuno S, Hashimoto T, Satoh T. Case of neuropathic itch-associated prurigo nodules on the bilateral upper arms after unilateral herpes zoster in a patient with cervical herniated discs: successful treatment with mirogabalin. J Dermatol. 2021;48:e585-e586.
- Papoiu AD, Yosipovitch G. Topical capsaicin. The fire of a ‘hot’ medicine is reignited. Expert Opin Pharmacother. 2010;11:1359-1371. doi:10.1517/14656566.2010.481670
- Magazin M, Daze RP, Okeson N. Treatment refractory brachioradial pruritus treated with topical amitriptyline and ketamine. Cureus. 2019;11:e5117. doi:10.7759/cureus.5117
- Weinberg BD, Amans M, Deviren S, et al. Brachioradial pruritus treated with computed tomography-guided cervical nerve root block: a case series. JAAD Case Rep. 2018;4:640-644. doi:10.1016/j.jdcr.2018.03.025
- De Ridder D, Hans G, Pals P, et al. A C-fiber-mediated neuropathic brachioradial pruritus. J Neurosurg. 2010;113:118-121. doi:10.3171/2009.9.JNS09620
- Morosanu CO, Etim G, Alalade AF. Brachioradial pruritus secondary to cervical disc protrusion—a case report. J Surg Case Rep. 2022:rjac277. doi:10.1093/jscr/rjac277
Practice Points
- The etiology of brachioradial pruritus (BRP) has been associated with cervical spine pathology and/or UV radiation exposure.
- Treatment options for BRP range from conservative to invasive, and clinicians should consider the evidence for all options to decide what is best for each patient.
Pigmenting Purpuric Dermatoses: Striking But Not a Manifestation of COVID-19 Infection
Pigmented purpuric dermatoses (PPDs) are characterized by petechiae, dusky macules representative of postinflammatory hyperpigmentation and dermal hemosiderin, and purpura generally localized to the lower extremities. They typically represent a spectrum of lymphocytic capillaritis, variable erythrocyte extravasation from papillary dermal blood vessels, and deposition of hemosiderin, yielding the classic red to orange to golden-brown findings on gross examination. Clinical overlap exists, but variants include Schamberg disease (SD), Majocchi purpura, Gougerot-Blum purpura, eczematoid purpura of Doucas and Kapetanakis (DK), and lichen aureus.1 Other forms are rarer, including linear, granulomatous, quadrantic, transitory, and familial variants. It remains controversial whether PPD may precede or have an association with cutaneous T-cell lymphoma.2 Dermoscopy usually shows copper-red pigmentation in the background, oval red dots, linear vessels, brown globules, and follicular openings. Although these findings may be useful in PPD diagnosis, they are not applicable in differentiating among the variants.
Pigmented purpuric dermatoses can easily be mistaken for stasis dermatitis or cellulitis, as these may occur concomitantly or in populations at risk for all 3 conditions, such as women older than 50 years with recent trauma or infection in the affected area. Tissue biopsy and clinical laboratory evaluation may be required to differentiate between PPD from leukocytoclastic vasculitis or the myriad causes of retiform purpura. Importantly, clinicians also should differentiate PPD from the purpuric eruptions of the lower extremities associated with COVID-19 infection.
Pigmented Purpuric Dermatoses
Schamberg Disease—In 1901, Jay Frank Schamberg, a distinguished professor of dermatology in Philadelphia, Pennsylvania, described “a peculiar progressive pigmentary disease of the skin” in a 15-year-old adolescent boy.3 Schamberg disease is the most common PPD, characterized by pruritic spots resembling cayenne pepper (Figure 1) with orange-brown pigmented macules on the legs and feet.4 Although platelet dysfunction, coagulation deficiencies, or dermal atrophy may contribute to hemorrhaging that manifests as petechiae or ecchymoses, SD typically is not associated with any laboratory abnormalities, and petechial eruption is not widespread.5 Capillary fragility can be assessed by the tourniquet test, in which pressure is applied to the forearm with a blood pressure cuff inflated between systolic and diastolic blood pressure for 5 to 10 minutes. Upon removing the cuff, a positive test is indicated by 15 or more petechiae in an area 5 cm in diameter due to poor platelet function. A positive result may be seen in SD.6
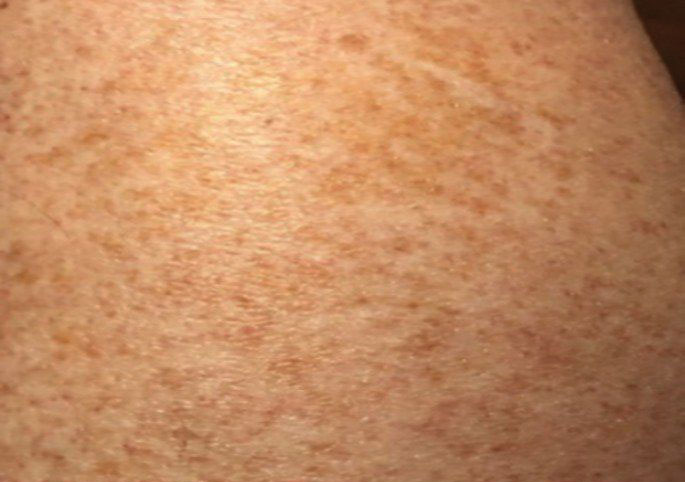
Histologically, SD is characterized by patchy parakeratosis, mild spongiosis of the stratum Malpighi, and lymphoid capillaritis (Figure 2).7 In addition to CD3+, CD4+, CD8+, CD1a+, and CD36+ lymphocytes, histology also may contain dendritic cells and cellular adhesion molecules (intercellular adhesion molecule 1, epithelial cell adhesion molecule 1) within the superficial perivascular infiltrate.8 There is no definitive therapy, but first-line interventions include emollients, topical steroids, and oral antihistamines. Nonpharmacologic management includes compression or support stockings, elevation of the lower extremities, and avoidance of offending medications (if identifiable).1
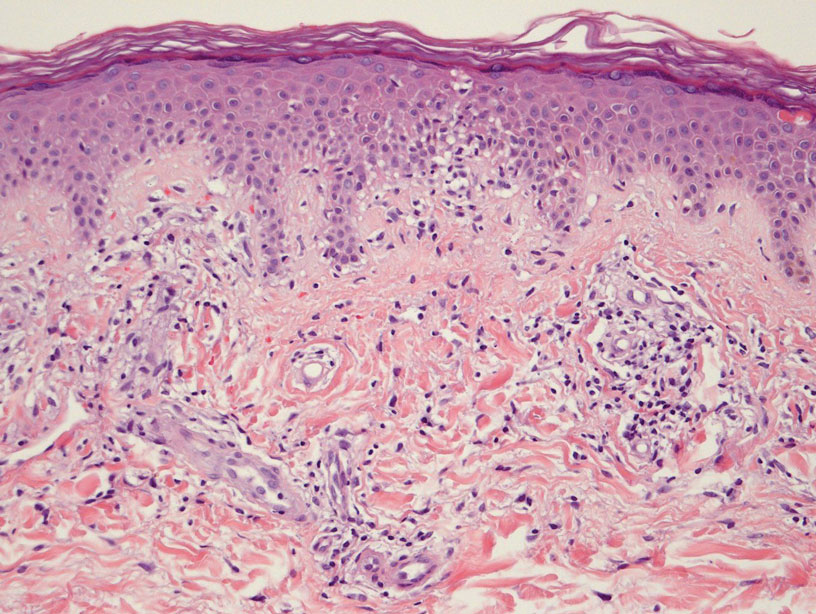
Majocchi Purpura—Domenico Majocchi was a renowned Italian dermatologist who described an entity in 1898 that he called purpura annularis telangiectodes, now also known as Majocchi purpura.9 It is more common in females, young adults, and children. Majocchi purpura has rarely been reported in families with a possible autosomal-dominant inheritance.10 Typically, bluish-red annular macules with central atrophy surrounded by hyperpigmentation may be seen on the lower extremities, potentially extending to the upper extremities.1 Treatment of Majocchi purpura remains a challenge but may respond to narrowband UVB phototherapy. Emollients and topical steroids also are used as first-line treatments. Biopsy demonstrates telangiectasia, pericapillary infiltration of mononuclear lymphocytes, and papillary dermal hemosiderin.11
Gougerot-Blum Purpura—In 1925, French dermatologists Henri Gougerot and Paul Blum described a pigmented purpuric lichenoid dermatitis known as Gougerot-Blum purpura,12 a rare PPD characterized by lichenoid papules that eventually coalesce into plaques of various colors, along with red-brown hyperpigmentation.4 As with other PPD variants, the legs are most involved, with rare extension to the trunk or thighs. The plaques may resemble and be mistaken for Kaposi sarcoma, cutaneous vasculitis, traumatic purpura, or mycosis fungoides. Dermoscopic examination reveals small, polygonal or round, red dots underlying brown scaly patches.13 Gougerot-Blum purpura is found more commonly in adult men and rarely affects children.4 Histologically, a lichenoid and superficial perivascular infiltrate composed of lymphocytes and macrophages is seen. Various therapies have been described, including topical steroids, antihistamines, psoralen plus UVA phototherapy, and cyclosporin A.14
Eczematoid Purpura of Doucas and Kapetanakis—In 1949, Greek dermatologists Christopher Doucas and John Kapetanakis observed several cases of purpuric dermatosis similar in form to the “pigmented purpuric lichenoid dermatitis” of Gougerot-Blum purpura12 and to the “progressive pigmentary dermatitis” of Schamberg disease.3 After observing a gradual disappearance of the classic yellow color from hemosiderin deposition, Doucas and Kapetanakis described a new bright red eruption with lichenification.15 Eczematoid purpura of Doucas and Kapetanakis is rare and predominantly seen in middle-aged males. Hyperpigmented or dark brown macules may develop bilaterally on the legs, progressing to the thighs and upper extremities. Unlike the other types of PPD, DK is extensive and severely pruritic.4
Although most PPD can be drug induced, DK has shown the greatest tendency for pruritic erythematous plaques following drug usage including but not limited to amlodipine, aspirin, acetaminophen, thiamine, interferon alfa, chlordiazepoxide, and isotretinoin. Additionally, DK has been associated with a contact allergy to clothing dyes and rubber.4 On histology, epidermal spongiosis may be seen, correlating with the eczematoid clinical findings. Spontaneous remission also is more common compared to the other PPDs. Treatment consists of topical corticosteroids and antihistamines.16
Lichen Aureus—Lichen aureus was first observed by the dermatologist R.H. Martin in 1958.17 It is clinically characterized by closely aggregated purpuric papules with a distinctive golden-brown color more often localized to the lower extremities and sometimes in a dermatomal distribution. Lichen aureus affects males and females equally, and similar to Majocchi purpura can be seen in children.4 Histopathologic examination reveals a prominent lichenoid plus superficial and deep perivascular lymphocytic infiltrate, extravasated erythrocytes, papillary dermal edema, hemosiderophages, and an unaffected epidermis. In rare cases, perineural infiltrates may be seen. Topical steroids usually are ineffective in lichen aureus treatment, but responses to psoralen plus UVA therapy also have been noted.17
Differential Diagnosis
COVID-19–Related Cutaneous Changes—Because COVID-19–related pathology is now a common differential diagnosis for many cutaneous eruptions, one must be mindful of the possibility for patients to have PPD, cutaneous changes from underlying COVID-19, or both.18 The microvascular changes from COVID-19 infection can be variable.19 Besides the presence of erythema along a distal digit, manifestations can include reticulated dusky erythema mimicking livedoid vasculopathy or inflammatory purpura.19
Retiform Purpura—Retiform purpura may occur in the setting of microvascular occlusion and can represent the pattern of underlying dermal vasculature. It is nonblanching and typically stellate or branching.20 The microvascular occlusion may be a result of hypercoagulability or may be secondary to cutaneous vasculitis, resulting in thrombosis and subsequent vascular occlusion.21 There are many reasons for hypercoagulability in retiform purpura, including disseminated intravascular coagulation in the setting of COVID-19 infection.22 The treatment of retiform purpura is aimed at alleviating the underlying cause and providing symptomatic relief. Conversely, the PPDs generally are benign and require minimal workup.
Leukocytoclastic Vasculitis—The hallmark of leukocytoclastic vasculitis is palpable purpura, often appearing as nonblanchable papules, typically in a dependent distribution such as the lower extremities (Figure 3). Although it primarily affects children, Henoch-Schönlein purpura is a type of leukocytoclastic vasculitis with lesions potentially similar in appearance to those of PPD.23 Palpable purpura may be painful and may ulcerate but rarely is pruritic. Leukocytoclastic vasculitis represents perivascular infiltrates composed of neutrophils, lymphocytes, and occasionally eosinophils, along with karyorrhexis, luminal fibrin, and fibrinoid degeneration of blood vessel walls, often resulting from immune complex deposition. Leukocytoclastic vasculitis may affect blood vessels of any size and requires further clinical and laboratory evaluation for infection (including COVID-19), hypercoagulability, autoimmune disease, or medication-related reactions.24
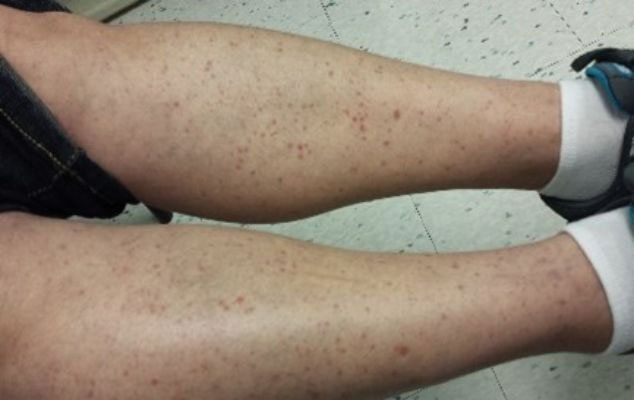
Stasis Dermatitis—Stasis dermatitis, a chronic inflammatory condition stemming from retrograde venous flow due to incompetent venous valves, mimics PPD. Stasis dermatitis initially appears as demarcated erythematous plaques, fissures, and scaling of the lower legs bilaterally, usually involving the medial malleolus.25 With time, the affected region develops overlying brawny hyperpigmentation and fibrosis (Figure 4). Pruritus or pain are common features, while fissures and superficial erosions may heal and recur, leading to lichenification.
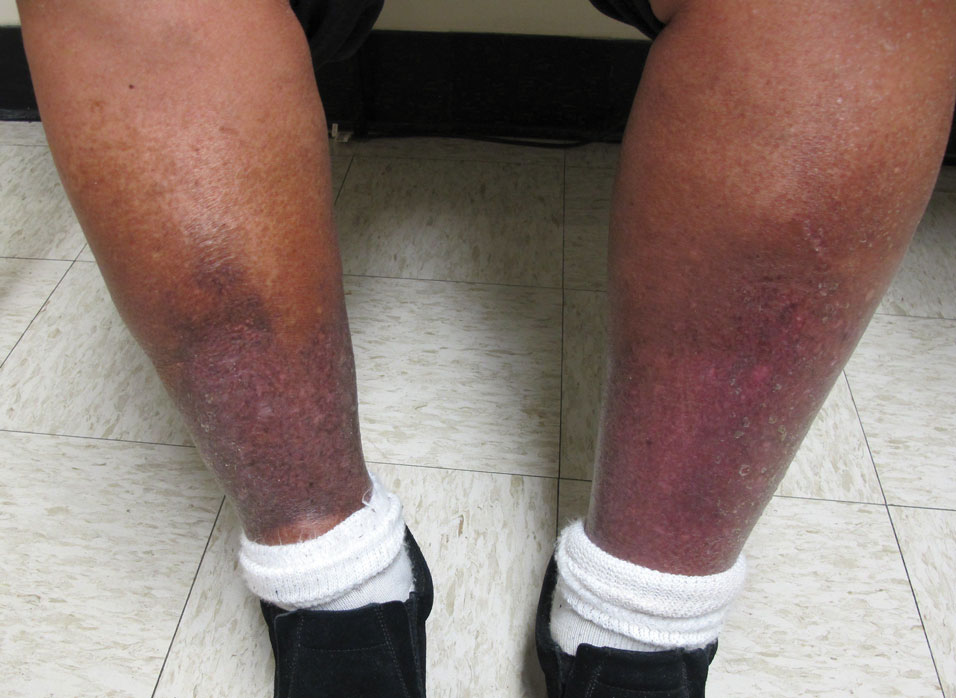
Although both commonly appear on the lower extremities, duplex ultrasonography may be helpful to distinguish PPDs from stasis dermatitis since the latter occurs in the context of chronic venous insufficiency, varicose veins, soft tissue edema, and lymphedema.25 Additionally, pruritus, lichenification, and edema often are not seen in most PPD variants, although stasis dermatitis and PPD may occur in tandem. Conservative treatment involves elevation of the extremities, compression, and topical steroids for symptomatic relief.
Cellulitis—The key characteristics of cellulitis are redness, swelling, warmth, tenderness, fever, and leukocytosis. A history of trauma, such as a prior break in the skin, and pain in the affected area suggest cellulitis. Several skin conditions present similarly to cellulitis, including PPD, and thus approximately 30% of cases are misdiagnosed.26 Cellulitis rarely presents in a bilateral or diffusely scattered pattern as seen in PPDs. Rather, it is unilateral with smooth indistinct borders. Variables suggestive of cellulitis include immunosuppression, rapid progression, and previous occurrences. Hyperpigmented plaques or thickening of the skin are more indicative of a chronic process such as stasis dermatitis or lipodermatosclerosis rather than acute cellulitis. Purpura is not a typical finding in most cases of soft tissue cellulitis. Treatment may be case specific depending on severity, presence or absence of sepsis, findings on blood cultures, or other pathologic evaluation. Antibiotics are directed to the causative organism, typically Streptococcus and Staphylococcus species, although coverage against various gram-negative organisms may be indicated.27
Caution With Teledermatology
COVID-19 has established the value of telemedicine in providing access to health care services for at-risk or underserved individuals. The PPDs are benign, often asymptomatic, and potentially identifiable with teledermatology alone; however, they also can easily be mistaken for COVID-19–related eruptions, vasculitis, other types of purpura, stasis dermatitis, or other complications of lower extremity stasis and lymphedema, especially in an aging population. If tissue biopsy is required, as in the workup of vasculitis, the efficacy of telemedicine becomes more questionable. It is important to delineate the potentially confusing PPDs from other potentially dangerous or life-threatening inflammatory dermatoses.28
- Sardana K, Sarkar R , Sehgal VN. Pigmented purpuric dermatoses: an overview. Int J Dermatol. 2004;43:482-488.
- Çaytemel C, Baykut B, Ag˘ırgöl S¸, et al. Pigmented purpuric dermatosis: ten years of experience in a tertiary hospital and awareness of mycosis fungoides in differential diagnosis. J Cutan Pathol. 2021;48:611-616.
- Schamberg JF. A peculiar progressive pigmentary disease of the skin. Br J Dermatol. 1901;13:1-5.
- Martínez Pallás I, Conejero Del Mazo R, Lezcano Biosca V. Pigmented purpuric dermatosis: a review of the literature. Actas Dermosifiliogr (Engl Ed). 2020;111:196-204.
- Ozkaya DB, Emiroglu N, Su O, et al. Dermatoscopic findings of pigmented purpuric dermatosis. An Bras Dermatol. 2016;91:584-587.
- Lava SAG, Milani GP, Fossali EF, et al. Cutaneous manifestations of small-vessel leukocytoclastic vasculitides in childhood. Clin Rev Allergy Immunol. 2017;53:439-451.
- Bonnet U, Selle C, Isbruch K, et al. Recurrent purpura due to alcohol-related Schamberg’s disease and its association with serum immunoglobulins: a longitudinal observation of a heavy drinker. J Med Case Rep. 2016;10:301.
- Zaldivar Fujigaki JL, Anjum F. Schamberg Disease. StatPearls Publishing; 2021.
- Majocchi J. Purpura annularis telangiectodes. Arch Dermatol Syph. 1898;43:447.
- Sethuraman G, Sugandhan S, Bansal A, et al. Familial pigmented purpuric dermatoses. J Dermatol. 2006;33:639-641.
- Miller K, Fischer M, Kamino H, et al. Purpura annularis telangiectoides. Dermatol Online J. 2012;18:5.
- Coulombe J, Jean SE, Hatami A, et al. Pigmented purpuric dermatosis: clinicopathologic characterization in a pediatric series. Pediatr Dermatol. 2015;32:358-362.
- Park MY, Shim WH, Kim JM, et al. Dermoscopic finding in pigmented purpuric lichenoid dermatosis of Gougerot-Blum: a useful tool for clinical diagnosis. Ann Dermatol. 2018;30:245-247.
- Risikesan J, Sommerlund M, Ramsing M, et al. Successful topical treatment of pigmented purpuric lichenoid dermatitis of Gougerot-Blum in a young patient: a case report and summary of the most common pigmented purpuric dermatoses. Case Rep Dermatol. 2017;9:169-176.
- Doucas C, Kapetanakis J. Eczematid-like purpura. Dermatologica. 1953;106:86-95.
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410.
- Aung PP, Burns SJ, Bhawan J. Lichen aureus: an unusual histopathological presentation: a case report and a review of literature. Am J Dermatopathol. 2014;36:E1-E4.
- Singh P, Schwartz RA. Disseminated intravascular coagulation: a devastating systemic disorder of special concern with COVID-19. Dermatol Ther. 2020;33:E14053.
- Almutairi N, Schwartz RA. COVID-19 with dermatologic manifestations and implications: an unfolding conundrum. Dermatol Ther. 2020;33:E13544.
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796.
- Torregrosa Calatayud JL, Garcías Ladaria J, De Unamuno Bustos B, et al. Retiform purpura caused by the use of cocaine, that was probably adulterated with levamisole. Ann Dermatol. 2015;27:117-119.
- Keim CK, Schwartz RA, Kapila R. Levamisole-induced and COVID-19-induced retiform purpura: two overlapping, emerging clinical syndromes. Arch Dermatol Res. 2021;22:1-9.
- González LM, Janniger CK, Schwartz RA. Pediatric Henoch-Schönlein purpura. Int J Dermatol. 2009;48:1157-1165.
- Yıldırım Bay E, Moustafa E, Semiz Y, et al. Leukocytoclastic vasculitis secondary to COVID-19 infection presenting with inclusion bodies: a histopathological correlation. J Cosmet Dermatol. 2022;21:27-29.
- Sundaresan S, Migden MR, Silapunt S. Stasis dermatitis: pathophysiology, evaluation, and management. Am J Clin Dermatol. 2017;18:383-390.
- Hirschmann JV, Raugi GJ. Lower limb cellulitis and its mimics: part I. lower limb cellulitis. J Am Acad Dermatol. 2012;67:163.E1-E12; quiz 75-76.
- Keller EC, Tomecki KJ, Alraies MC. Distinguishing cellulitis from its mimics. Cleveland Clin J Med. 2012;79:547-552.
- Georgesen C, Fox LP, Harp J. Retiform purpura: workup and therapeutic considerations in select conditions. J Am Acad Dermatol. 2020;82:799-816.
Pigmented purpuric dermatoses (PPDs) are characterized by petechiae, dusky macules representative of postinflammatory hyperpigmentation and dermal hemosiderin, and purpura generally localized to the lower extremities. They typically represent a spectrum of lymphocytic capillaritis, variable erythrocyte extravasation from papillary dermal blood vessels, and deposition of hemosiderin, yielding the classic red to orange to golden-brown findings on gross examination. Clinical overlap exists, but variants include Schamberg disease (SD), Majocchi purpura, Gougerot-Blum purpura, eczematoid purpura of Doucas and Kapetanakis (DK), and lichen aureus.1 Other forms are rarer, including linear, granulomatous, quadrantic, transitory, and familial variants. It remains controversial whether PPD may precede or have an association with cutaneous T-cell lymphoma.2 Dermoscopy usually shows copper-red pigmentation in the background, oval red dots, linear vessels, brown globules, and follicular openings. Although these findings may be useful in PPD diagnosis, they are not applicable in differentiating among the variants.
Pigmented purpuric dermatoses can easily be mistaken for stasis dermatitis or cellulitis, as these may occur concomitantly or in populations at risk for all 3 conditions, such as women older than 50 years with recent trauma or infection in the affected area. Tissue biopsy and clinical laboratory evaluation may be required to differentiate between PPD from leukocytoclastic vasculitis or the myriad causes of retiform purpura. Importantly, clinicians also should differentiate PPD from the purpuric eruptions of the lower extremities associated with COVID-19 infection.
Pigmented Purpuric Dermatoses
Schamberg Disease—In 1901, Jay Frank Schamberg, a distinguished professor of dermatology in Philadelphia, Pennsylvania, described “a peculiar progressive pigmentary disease of the skin” in a 15-year-old adolescent boy.3 Schamberg disease is the most common PPD, characterized by pruritic spots resembling cayenne pepper (Figure 1) with orange-brown pigmented macules on the legs and feet.4 Although platelet dysfunction, coagulation deficiencies, or dermal atrophy may contribute to hemorrhaging that manifests as petechiae or ecchymoses, SD typically is not associated with any laboratory abnormalities, and petechial eruption is not widespread.5 Capillary fragility can be assessed by the tourniquet test, in which pressure is applied to the forearm with a blood pressure cuff inflated between systolic and diastolic blood pressure for 5 to 10 minutes. Upon removing the cuff, a positive test is indicated by 15 or more petechiae in an area 5 cm in diameter due to poor platelet function. A positive result may be seen in SD.6

Histologically, SD is characterized by patchy parakeratosis, mild spongiosis of the stratum Malpighi, and lymphoid capillaritis (Figure 2).7 In addition to CD3+, CD4+, CD8+, CD1a+, and CD36+ lymphocytes, histology also may contain dendritic cells and cellular adhesion molecules (intercellular adhesion molecule 1, epithelial cell adhesion molecule 1) within the superficial perivascular infiltrate.8 There is no definitive therapy, but first-line interventions include emollients, topical steroids, and oral antihistamines. Nonpharmacologic management includes compression or support stockings, elevation of the lower extremities, and avoidance of offending medications (if identifiable).1

Majocchi Purpura—Domenico Majocchi was a renowned Italian dermatologist who described an entity in 1898 that he called purpura annularis telangiectodes, now also known as Majocchi purpura.9 It is more common in females, young adults, and children. Majocchi purpura has rarely been reported in families with a possible autosomal-dominant inheritance.10 Typically, bluish-red annular macules with central atrophy surrounded by hyperpigmentation may be seen on the lower extremities, potentially extending to the upper extremities.1 Treatment of Majocchi purpura remains a challenge but may respond to narrowband UVB phototherapy. Emollients and topical steroids also are used as first-line treatments. Biopsy demonstrates telangiectasia, pericapillary infiltration of mononuclear lymphocytes, and papillary dermal hemosiderin.11
Gougerot-Blum Purpura—In 1925, French dermatologists Henri Gougerot and Paul Blum described a pigmented purpuric lichenoid dermatitis known as Gougerot-Blum purpura,12 a rare PPD characterized by lichenoid papules that eventually coalesce into plaques of various colors, along with red-brown hyperpigmentation.4 As with other PPD variants, the legs are most involved, with rare extension to the trunk or thighs. The plaques may resemble and be mistaken for Kaposi sarcoma, cutaneous vasculitis, traumatic purpura, or mycosis fungoides. Dermoscopic examination reveals small, polygonal or round, red dots underlying brown scaly patches.13 Gougerot-Blum purpura is found more commonly in adult men and rarely affects children.4 Histologically, a lichenoid and superficial perivascular infiltrate composed of lymphocytes and macrophages is seen. Various therapies have been described, including topical steroids, antihistamines, psoralen plus UVA phototherapy, and cyclosporin A.14
Eczematoid Purpura of Doucas and Kapetanakis—In 1949, Greek dermatologists Christopher Doucas and John Kapetanakis observed several cases of purpuric dermatosis similar in form to the “pigmented purpuric lichenoid dermatitis” of Gougerot-Blum purpura12 and to the “progressive pigmentary dermatitis” of Schamberg disease.3 After observing a gradual disappearance of the classic yellow color from hemosiderin deposition, Doucas and Kapetanakis described a new bright red eruption with lichenification.15 Eczematoid purpura of Doucas and Kapetanakis is rare and predominantly seen in middle-aged males. Hyperpigmented or dark brown macules may develop bilaterally on the legs, progressing to the thighs and upper extremities. Unlike the other types of PPD, DK is extensive and severely pruritic.4
Although most PPD can be drug induced, DK has shown the greatest tendency for pruritic erythematous plaques following drug usage including but not limited to amlodipine, aspirin, acetaminophen, thiamine, interferon alfa, chlordiazepoxide, and isotretinoin. Additionally, DK has been associated with a contact allergy to clothing dyes and rubber.4 On histology, epidermal spongiosis may be seen, correlating with the eczematoid clinical findings. Spontaneous remission also is more common compared to the other PPDs. Treatment consists of topical corticosteroids and antihistamines.16
Lichen Aureus—Lichen aureus was first observed by the dermatologist R.H. Martin in 1958.17 It is clinically characterized by closely aggregated purpuric papules with a distinctive golden-brown color more often localized to the lower extremities and sometimes in a dermatomal distribution. Lichen aureus affects males and females equally, and similar to Majocchi purpura can be seen in children.4 Histopathologic examination reveals a prominent lichenoid plus superficial and deep perivascular lymphocytic infiltrate, extravasated erythrocytes, papillary dermal edema, hemosiderophages, and an unaffected epidermis. In rare cases, perineural infiltrates may be seen. Topical steroids usually are ineffective in lichen aureus treatment, but responses to psoralen plus UVA therapy also have been noted.17
Differential Diagnosis
COVID-19–Related Cutaneous Changes—Because COVID-19–related pathology is now a common differential diagnosis for many cutaneous eruptions, one must be mindful of the possibility for patients to have PPD, cutaneous changes from underlying COVID-19, or both.18 The microvascular changes from COVID-19 infection can be variable.19 Besides the presence of erythema along a distal digit, manifestations can include reticulated dusky erythema mimicking livedoid vasculopathy or inflammatory purpura.19
Retiform Purpura—Retiform purpura may occur in the setting of microvascular occlusion and can represent the pattern of underlying dermal vasculature. It is nonblanching and typically stellate or branching.20 The microvascular occlusion may be a result of hypercoagulability or may be secondary to cutaneous vasculitis, resulting in thrombosis and subsequent vascular occlusion.21 There are many reasons for hypercoagulability in retiform purpura, including disseminated intravascular coagulation in the setting of COVID-19 infection.22 The treatment of retiform purpura is aimed at alleviating the underlying cause and providing symptomatic relief. Conversely, the PPDs generally are benign and require minimal workup.
Leukocytoclastic Vasculitis—The hallmark of leukocytoclastic vasculitis is palpable purpura, often appearing as nonblanchable papules, typically in a dependent distribution such as the lower extremities (Figure 3). Although it primarily affects children, Henoch-Schönlein purpura is a type of leukocytoclastic vasculitis with lesions potentially similar in appearance to those of PPD.23 Palpable purpura may be painful and may ulcerate but rarely is pruritic. Leukocytoclastic vasculitis represents perivascular infiltrates composed of neutrophils, lymphocytes, and occasionally eosinophils, along with karyorrhexis, luminal fibrin, and fibrinoid degeneration of blood vessel walls, often resulting from immune complex deposition. Leukocytoclastic vasculitis may affect blood vessels of any size and requires further clinical and laboratory evaluation for infection (including COVID-19), hypercoagulability, autoimmune disease, or medication-related reactions.24

Stasis Dermatitis—Stasis dermatitis, a chronic inflammatory condition stemming from retrograde venous flow due to incompetent venous valves, mimics PPD. Stasis dermatitis initially appears as demarcated erythematous plaques, fissures, and scaling of the lower legs bilaterally, usually involving the medial malleolus.25 With time, the affected region develops overlying brawny hyperpigmentation and fibrosis (Figure 4). Pruritus or pain are common features, while fissures and superficial erosions may heal and recur, leading to lichenification.

Although both commonly appear on the lower extremities, duplex ultrasonography may be helpful to distinguish PPDs from stasis dermatitis since the latter occurs in the context of chronic venous insufficiency, varicose veins, soft tissue edema, and lymphedema.25 Additionally, pruritus, lichenification, and edema often are not seen in most PPD variants, although stasis dermatitis and PPD may occur in tandem. Conservative treatment involves elevation of the extremities, compression, and topical steroids for symptomatic relief.
Cellulitis—The key characteristics of cellulitis are redness, swelling, warmth, tenderness, fever, and leukocytosis. A history of trauma, such as a prior break in the skin, and pain in the affected area suggest cellulitis. Several skin conditions present similarly to cellulitis, including PPD, and thus approximately 30% of cases are misdiagnosed.26 Cellulitis rarely presents in a bilateral or diffusely scattered pattern as seen in PPDs. Rather, it is unilateral with smooth indistinct borders. Variables suggestive of cellulitis include immunosuppression, rapid progression, and previous occurrences. Hyperpigmented plaques or thickening of the skin are more indicative of a chronic process such as stasis dermatitis or lipodermatosclerosis rather than acute cellulitis. Purpura is not a typical finding in most cases of soft tissue cellulitis. Treatment may be case specific depending on severity, presence or absence of sepsis, findings on blood cultures, or other pathologic evaluation. Antibiotics are directed to the causative organism, typically Streptococcus and Staphylococcus species, although coverage against various gram-negative organisms may be indicated.27
Caution With Teledermatology
COVID-19 has established the value of telemedicine in providing access to health care services for at-risk or underserved individuals. The PPDs are benign, often asymptomatic, and potentially identifiable with teledermatology alone; however, they also can easily be mistaken for COVID-19–related eruptions, vasculitis, other types of purpura, stasis dermatitis, or other complications of lower extremity stasis and lymphedema, especially in an aging population. If tissue biopsy is required, as in the workup of vasculitis, the efficacy of telemedicine becomes more questionable. It is important to delineate the potentially confusing PPDs from other potentially dangerous or life-threatening inflammatory dermatoses.28
Pigmented purpuric dermatoses (PPDs) are characterized by petechiae, dusky macules representative of postinflammatory hyperpigmentation and dermal hemosiderin, and purpura generally localized to the lower extremities. They typically represent a spectrum of lymphocytic capillaritis, variable erythrocyte extravasation from papillary dermal blood vessels, and deposition of hemosiderin, yielding the classic red to orange to golden-brown findings on gross examination. Clinical overlap exists, but variants include Schamberg disease (SD), Majocchi purpura, Gougerot-Blum purpura, eczematoid purpura of Doucas and Kapetanakis (DK), and lichen aureus.1 Other forms are rarer, including linear, granulomatous, quadrantic, transitory, and familial variants. It remains controversial whether PPD may precede or have an association with cutaneous T-cell lymphoma.2 Dermoscopy usually shows copper-red pigmentation in the background, oval red dots, linear vessels, brown globules, and follicular openings. Although these findings may be useful in PPD diagnosis, they are not applicable in differentiating among the variants.
Pigmented purpuric dermatoses can easily be mistaken for stasis dermatitis or cellulitis, as these may occur concomitantly or in populations at risk for all 3 conditions, such as women older than 50 years with recent trauma or infection in the affected area. Tissue biopsy and clinical laboratory evaluation may be required to differentiate between PPD from leukocytoclastic vasculitis or the myriad causes of retiform purpura. Importantly, clinicians also should differentiate PPD from the purpuric eruptions of the lower extremities associated with COVID-19 infection.
Pigmented Purpuric Dermatoses
Schamberg Disease—In 1901, Jay Frank Schamberg, a distinguished professor of dermatology in Philadelphia, Pennsylvania, described “a peculiar progressive pigmentary disease of the skin” in a 15-year-old adolescent boy.3 Schamberg disease is the most common PPD, characterized by pruritic spots resembling cayenne pepper (Figure 1) with orange-brown pigmented macules on the legs and feet.4 Although platelet dysfunction, coagulation deficiencies, or dermal atrophy may contribute to hemorrhaging that manifests as petechiae or ecchymoses, SD typically is not associated with any laboratory abnormalities, and petechial eruption is not widespread.5 Capillary fragility can be assessed by the tourniquet test, in which pressure is applied to the forearm with a blood pressure cuff inflated between systolic and diastolic blood pressure for 5 to 10 minutes. Upon removing the cuff, a positive test is indicated by 15 or more petechiae in an area 5 cm in diameter due to poor platelet function. A positive result may be seen in SD.6

Histologically, SD is characterized by patchy parakeratosis, mild spongiosis of the stratum Malpighi, and lymphoid capillaritis (Figure 2).7 In addition to CD3+, CD4+, CD8+, CD1a+, and CD36+ lymphocytes, histology also may contain dendritic cells and cellular adhesion molecules (intercellular adhesion molecule 1, epithelial cell adhesion molecule 1) within the superficial perivascular infiltrate.8 There is no definitive therapy, but first-line interventions include emollients, topical steroids, and oral antihistamines. Nonpharmacologic management includes compression or support stockings, elevation of the lower extremities, and avoidance of offending medications (if identifiable).1

Majocchi Purpura—Domenico Majocchi was a renowned Italian dermatologist who described an entity in 1898 that he called purpura annularis telangiectodes, now also known as Majocchi purpura.9 It is more common in females, young adults, and children. Majocchi purpura has rarely been reported in families with a possible autosomal-dominant inheritance.10 Typically, bluish-red annular macules with central atrophy surrounded by hyperpigmentation may be seen on the lower extremities, potentially extending to the upper extremities.1 Treatment of Majocchi purpura remains a challenge but may respond to narrowband UVB phototherapy. Emollients and topical steroids also are used as first-line treatments. Biopsy demonstrates telangiectasia, pericapillary infiltration of mononuclear lymphocytes, and papillary dermal hemosiderin.11
Gougerot-Blum Purpura—In 1925, French dermatologists Henri Gougerot and Paul Blum described a pigmented purpuric lichenoid dermatitis known as Gougerot-Blum purpura,12 a rare PPD characterized by lichenoid papules that eventually coalesce into plaques of various colors, along with red-brown hyperpigmentation.4 As with other PPD variants, the legs are most involved, with rare extension to the trunk or thighs. The plaques may resemble and be mistaken for Kaposi sarcoma, cutaneous vasculitis, traumatic purpura, or mycosis fungoides. Dermoscopic examination reveals small, polygonal or round, red dots underlying brown scaly patches.13 Gougerot-Blum purpura is found more commonly in adult men and rarely affects children.4 Histologically, a lichenoid and superficial perivascular infiltrate composed of lymphocytes and macrophages is seen. Various therapies have been described, including topical steroids, antihistamines, psoralen plus UVA phototherapy, and cyclosporin A.14
Eczematoid Purpura of Doucas and Kapetanakis—In 1949, Greek dermatologists Christopher Doucas and John Kapetanakis observed several cases of purpuric dermatosis similar in form to the “pigmented purpuric lichenoid dermatitis” of Gougerot-Blum purpura12 and to the “progressive pigmentary dermatitis” of Schamberg disease.3 After observing a gradual disappearance of the classic yellow color from hemosiderin deposition, Doucas and Kapetanakis described a new bright red eruption with lichenification.15 Eczematoid purpura of Doucas and Kapetanakis is rare and predominantly seen in middle-aged males. Hyperpigmented or dark brown macules may develop bilaterally on the legs, progressing to the thighs and upper extremities. Unlike the other types of PPD, DK is extensive and severely pruritic.4
Although most PPD can be drug induced, DK has shown the greatest tendency for pruritic erythematous plaques following drug usage including but not limited to amlodipine, aspirin, acetaminophen, thiamine, interferon alfa, chlordiazepoxide, and isotretinoin. Additionally, DK has been associated with a contact allergy to clothing dyes and rubber.4 On histology, epidermal spongiosis may be seen, correlating with the eczematoid clinical findings. Spontaneous remission also is more common compared to the other PPDs. Treatment consists of topical corticosteroids and antihistamines.16
Lichen Aureus—Lichen aureus was first observed by the dermatologist R.H. Martin in 1958.17 It is clinically characterized by closely aggregated purpuric papules with a distinctive golden-brown color more often localized to the lower extremities and sometimes in a dermatomal distribution. Lichen aureus affects males and females equally, and similar to Majocchi purpura can be seen in children.4 Histopathologic examination reveals a prominent lichenoid plus superficial and deep perivascular lymphocytic infiltrate, extravasated erythrocytes, papillary dermal edema, hemosiderophages, and an unaffected epidermis. In rare cases, perineural infiltrates may be seen. Topical steroids usually are ineffective in lichen aureus treatment, but responses to psoralen plus UVA therapy also have been noted.17
Differential Diagnosis
COVID-19–Related Cutaneous Changes—Because COVID-19–related pathology is now a common differential diagnosis for many cutaneous eruptions, one must be mindful of the possibility for patients to have PPD, cutaneous changes from underlying COVID-19, or both.18 The microvascular changes from COVID-19 infection can be variable.19 Besides the presence of erythema along a distal digit, manifestations can include reticulated dusky erythema mimicking livedoid vasculopathy or inflammatory purpura.19
Retiform Purpura—Retiform purpura may occur in the setting of microvascular occlusion and can represent the pattern of underlying dermal vasculature. It is nonblanching and typically stellate or branching.20 The microvascular occlusion may be a result of hypercoagulability or may be secondary to cutaneous vasculitis, resulting in thrombosis and subsequent vascular occlusion.21 There are many reasons for hypercoagulability in retiform purpura, including disseminated intravascular coagulation in the setting of COVID-19 infection.22 The treatment of retiform purpura is aimed at alleviating the underlying cause and providing symptomatic relief. Conversely, the PPDs generally are benign and require minimal workup.
Leukocytoclastic Vasculitis—The hallmark of leukocytoclastic vasculitis is palpable purpura, often appearing as nonblanchable papules, typically in a dependent distribution such as the lower extremities (Figure 3). Although it primarily affects children, Henoch-Schönlein purpura is a type of leukocytoclastic vasculitis with lesions potentially similar in appearance to those of PPD.23 Palpable purpura may be painful and may ulcerate but rarely is pruritic. Leukocytoclastic vasculitis represents perivascular infiltrates composed of neutrophils, lymphocytes, and occasionally eosinophils, along with karyorrhexis, luminal fibrin, and fibrinoid degeneration of blood vessel walls, often resulting from immune complex deposition. Leukocytoclastic vasculitis may affect blood vessels of any size and requires further clinical and laboratory evaluation for infection (including COVID-19), hypercoagulability, autoimmune disease, or medication-related reactions.24

Stasis Dermatitis—Stasis dermatitis, a chronic inflammatory condition stemming from retrograde venous flow due to incompetent venous valves, mimics PPD. Stasis dermatitis initially appears as demarcated erythematous plaques, fissures, and scaling of the lower legs bilaterally, usually involving the medial malleolus.25 With time, the affected region develops overlying brawny hyperpigmentation and fibrosis (Figure 4). Pruritus or pain are common features, while fissures and superficial erosions may heal and recur, leading to lichenification.

Although both commonly appear on the lower extremities, duplex ultrasonography may be helpful to distinguish PPDs from stasis dermatitis since the latter occurs in the context of chronic venous insufficiency, varicose veins, soft tissue edema, and lymphedema.25 Additionally, pruritus, lichenification, and edema often are not seen in most PPD variants, although stasis dermatitis and PPD may occur in tandem. Conservative treatment involves elevation of the extremities, compression, and topical steroids for symptomatic relief.
Cellulitis—The key characteristics of cellulitis are redness, swelling, warmth, tenderness, fever, and leukocytosis. A history of trauma, such as a prior break in the skin, and pain in the affected area suggest cellulitis. Several skin conditions present similarly to cellulitis, including PPD, and thus approximately 30% of cases are misdiagnosed.26 Cellulitis rarely presents in a bilateral or diffusely scattered pattern as seen in PPDs. Rather, it is unilateral with smooth indistinct borders. Variables suggestive of cellulitis include immunosuppression, rapid progression, and previous occurrences. Hyperpigmented plaques or thickening of the skin are more indicative of a chronic process such as stasis dermatitis or lipodermatosclerosis rather than acute cellulitis. Purpura is not a typical finding in most cases of soft tissue cellulitis. Treatment may be case specific depending on severity, presence or absence of sepsis, findings on blood cultures, or other pathologic evaluation. Antibiotics are directed to the causative organism, typically Streptococcus and Staphylococcus species, although coverage against various gram-negative organisms may be indicated.27
Caution With Teledermatology
COVID-19 has established the value of telemedicine in providing access to health care services for at-risk or underserved individuals. The PPDs are benign, often asymptomatic, and potentially identifiable with teledermatology alone; however, they also can easily be mistaken for COVID-19–related eruptions, vasculitis, other types of purpura, stasis dermatitis, or other complications of lower extremity stasis and lymphedema, especially in an aging population. If tissue biopsy is required, as in the workup of vasculitis, the efficacy of telemedicine becomes more questionable. It is important to delineate the potentially confusing PPDs from other potentially dangerous or life-threatening inflammatory dermatoses.28
- Sardana K, Sarkar R , Sehgal VN. Pigmented purpuric dermatoses: an overview. Int J Dermatol. 2004;43:482-488.
- Çaytemel C, Baykut B, Ag˘ırgöl S¸, et al. Pigmented purpuric dermatosis: ten years of experience in a tertiary hospital and awareness of mycosis fungoides in differential diagnosis. J Cutan Pathol. 2021;48:611-616.
- Schamberg JF. A peculiar progressive pigmentary disease of the skin. Br J Dermatol. 1901;13:1-5.
- Martínez Pallás I, Conejero Del Mazo R, Lezcano Biosca V. Pigmented purpuric dermatosis: a review of the literature. Actas Dermosifiliogr (Engl Ed). 2020;111:196-204.
- Ozkaya DB, Emiroglu N, Su O, et al. Dermatoscopic findings of pigmented purpuric dermatosis. An Bras Dermatol. 2016;91:584-587.
- Lava SAG, Milani GP, Fossali EF, et al. Cutaneous manifestations of small-vessel leukocytoclastic vasculitides in childhood. Clin Rev Allergy Immunol. 2017;53:439-451.
- Bonnet U, Selle C, Isbruch K, et al. Recurrent purpura due to alcohol-related Schamberg’s disease and its association with serum immunoglobulins: a longitudinal observation of a heavy drinker. J Med Case Rep. 2016;10:301.
- Zaldivar Fujigaki JL, Anjum F. Schamberg Disease. StatPearls Publishing; 2021.
- Majocchi J. Purpura annularis telangiectodes. Arch Dermatol Syph. 1898;43:447.
- Sethuraman G, Sugandhan S, Bansal A, et al. Familial pigmented purpuric dermatoses. J Dermatol. 2006;33:639-641.
- Miller K, Fischer M, Kamino H, et al. Purpura annularis telangiectoides. Dermatol Online J. 2012;18:5.
- Coulombe J, Jean SE, Hatami A, et al. Pigmented purpuric dermatosis: clinicopathologic characterization in a pediatric series. Pediatr Dermatol. 2015;32:358-362.
- Park MY, Shim WH, Kim JM, et al. Dermoscopic finding in pigmented purpuric lichenoid dermatosis of Gougerot-Blum: a useful tool for clinical diagnosis. Ann Dermatol. 2018;30:245-247.
- Risikesan J, Sommerlund M, Ramsing M, et al. Successful topical treatment of pigmented purpuric lichenoid dermatitis of Gougerot-Blum in a young patient: a case report and summary of the most common pigmented purpuric dermatoses. Case Rep Dermatol. 2017;9:169-176.
- Doucas C, Kapetanakis J. Eczematid-like purpura. Dermatologica. 1953;106:86-95.
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410.
- Aung PP, Burns SJ, Bhawan J. Lichen aureus: an unusual histopathological presentation: a case report and a review of literature. Am J Dermatopathol. 2014;36:E1-E4.
- Singh P, Schwartz RA. Disseminated intravascular coagulation: a devastating systemic disorder of special concern with COVID-19. Dermatol Ther. 2020;33:E14053.
- Almutairi N, Schwartz RA. COVID-19 with dermatologic manifestations and implications: an unfolding conundrum. Dermatol Ther. 2020;33:E13544.
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796.
- Torregrosa Calatayud JL, Garcías Ladaria J, De Unamuno Bustos B, et al. Retiform purpura caused by the use of cocaine, that was probably adulterated with levamisole. Ann Dermatol. 2015;27:117-119.
- Keim CK, Schwartz RA, Kapila R. Levamisole-induced and COVID-19-induced retiform purpura: two overlapping, emerging clinical syndromes. Arch Dermatol Res. 2021;22:1-9.
- González LM, Janniger CK, Schwartz RA. Pediatric Henoch-Schönlein purpura. Int J Dermatol. 2009;48:1157-1165.
- Yıldırım Bay E, Moustafa E, Semiz Y, et al. Leukocytoclastic vasculitis secondary to COVID-19 infection presenting with inclusion bodies: a histopathological correlation. J Cosmet Dermatol. 2022;21:27-29.
- Sundaresan S, Migden MR, Silapunt S. Stasis dermatitis: pathophysiology, evaluation, and management. Am J Clin Dermatol. 2017;18:383-390.
- Hirschmann JV, Raugi GJ. Lower limb cellulitis and its mimics: part I. lower limb cellulitis. J Am Acad Dermatol. 2012;67:163.E1-E12; quiz 75-76.
- Keller EC, Tomecki KJ, Alraies MC. Distinguishing cellulitis from its mimics. Cleveland Clin J Med. 2012;79:547-552.
- Georgesen C, Fox LP, Harp J. Retiform purpura: workup and therapeutic considerations in select conditions. J Am Acad Dermatol. 2020;82:799-816.
- Sardana K, Sarkar R , Sehgal VN. Pigmented purpuric dermatoses: an overview. Int J Dermatol. 2004;43:482-488.
- Çaytemel C, Baykut B, Ag˘ırgöl S¸, et al. Pigmented purpuric dermatosis: ten years of experience in a tertiary hospital and awareness of mycosis fungoides in differential diagnosis. J Cutan Pathol. 2021;48:611-616.
- Schamberg JF. A peculiar progressive pigmentary disease of the skin. Br J Dermatol. 1901;13:1-5.
- Martínez Pallás I, Conejero Del Mazo R, Lezcano Biosca V. Pigmented purpuric dermatosis: a review of the literature. Actas Dermosifiliogr (Engl Ed). 2020;111:196-204.
- Ozkaya DB, Emiroglu N, Su O, et al. Dermatoscopic findings of pigmented purpuric dermatosis. An Bras Dermatol. 2016;91:584-587.
- Lava SAG, Milani GP, Fossali EF, et al. Cutaneous manifestations of small-vessel leukocytoclastic vasculitides in childhood. Clin Rev Allergy Immunol. 2017;53:439-451.
- Bonnet U, Selle C, Isbruch K, et al. Recurrent purpura due to alcohol-related Schamberg’s disease and its association with serum immunoglobulins: a longitudinal observation of a heavy drinker. J Med Case Rep. 2016;10:301.
- Zaldivar Fujigaki JL, Anjum F. Schamberg Disease. StatPearls Publishing; 2021.
- Majocchi J. Purpura annularis telangiectodes. Arch Dermatol Syph. 1898;43:447.
- Sethuraman G, Sugandhan S, Bansal A, et al. Familial pigmented purpuric dermatoses. J Dermatol. 2006;33:639-641.
- Miller K, Fischer M, Kamino H, et al. Purpura annularis telangiectoides. Dermatol Online J. 2012;18:5.
- Coulombe J, Jean SE, Hatami A, et al. Pigmented purpuric dermatosis: clinicopathologic characterization in a pediatric series. Pediatr Dermatol. 2015;32:358-362.
- Park MY, Shim WH, Kim JM, et al. Dermoscopic finding in pigmented purpuric lichenoid dermatosis of Gougerot-Blum: a useful tool for clinical diagnosis. Ann Dermatol. 2018;30:245-247.
- Risikesan J, Sommerlund M, Ramsing M, et al. Successful topical treatment of pigmented purpuric lichenoid dermatitis of Gougerot-Blum in a young patient: a case report and summary of the most common pigmented purpuric dermatoses. Case Rep Dermatol. 2017;9:169-176.
- Doucas C, Kapetanakis J. Eczematid-like purpura. Dermatologica. 1953;106:86-95.
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410.
- Aung PP, Burns SJ, Bhawan J. Lichen aureus: an unusual histopathological presentation: a case report and a review of literature. Am J Dermatopathol. 2014;36:E1-E4.
- Singh P, Schwartz RA. Disseminated intravascular coagulation: a devastating systemic disorder of special concern with COVID-19. Dermatol Ther. 2020;33:E14053.
- Almutairi N, Schwartz RA. COVID-19 with dermatologic manifestations and implications: an unfolding conundrum. Dermatol Ther. 2020;33:E13544.
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796.
- Torregrosa Calatayud JL, Garcías Ladaria J, De Unamuno Bustos B, et al. Retiform purpura caused by the use of cocaine, that was probably adulterated with levamisole. Ann Dermatol. 2015;27:117-119.
- Keim CK, Schwartz RA, Kapila R. Levamisole-induced and COVID-19-induced retiform purpura: two overlapping, emerging clinical syndromes. Arch Dermatol Res. 2021;22:1-9.
- González LM, Janniger CK, Schwartz RA. Pediatric Henoch-Schönlein purpura. Int J Dermatol. 2009;48:1157-1165.
- Yıldırım Bay E, Moustafa E, Semiz Y, et al. Leukocytoclastic vasculitis secondary to COVID-19 infection presenting with inclusion bodies: a histopathological correlation. J Cosmet Dermatol. 2022;21:27-29.
- Sundaresan S, Migden MR, Silapunt S. Stasis dermatitis: pathophysiology, evaluation, and management. Am J Clin Dermatol. 2017;18:383-390.
- Hirschmann JV, Raugi GJ. Lower limb cellulitis and its mimics: part I. lower limb cellulitis. J Am Acad Dermatol. 2012;67:163.E1-E12; quiz 75-76.
- Keller EC, Tomecki KJ, Alraies MC. Distinguishing cellulitis from its mimics. Cleveland Clin J Med. 2012;79:547-552.
- Georgesen C, Fox LP, Harp J. Retiform purpura: workup and therapeutic considerations in select conditions. J Am Acad Dermatol. 2020;82:799-816.
Practice Points
- Dermatologists should be aware of the clinical presentations of pigmenting purpuric dermatoses (PPDs).
- Certain PPDs may resemble the thromboembolic events seen in COVID-19. Clinicians should especially be aware of how to differentiate these benign pigmentary disorders from other serious conditions.
- Teledermatology is widely utilized, but caution may be prudent when evaluating erythematous or purpuric dermatoses, especially those of the lower extremities.
- Pigmenting purpuric dermatoses generally are benign and do not require immediate treatment.