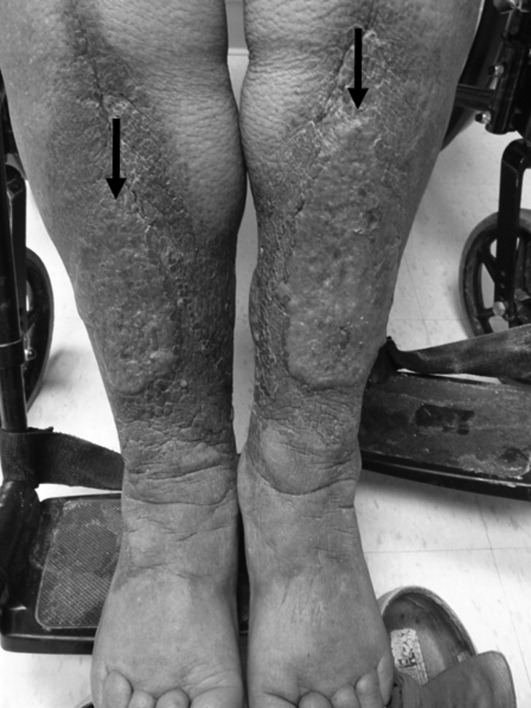
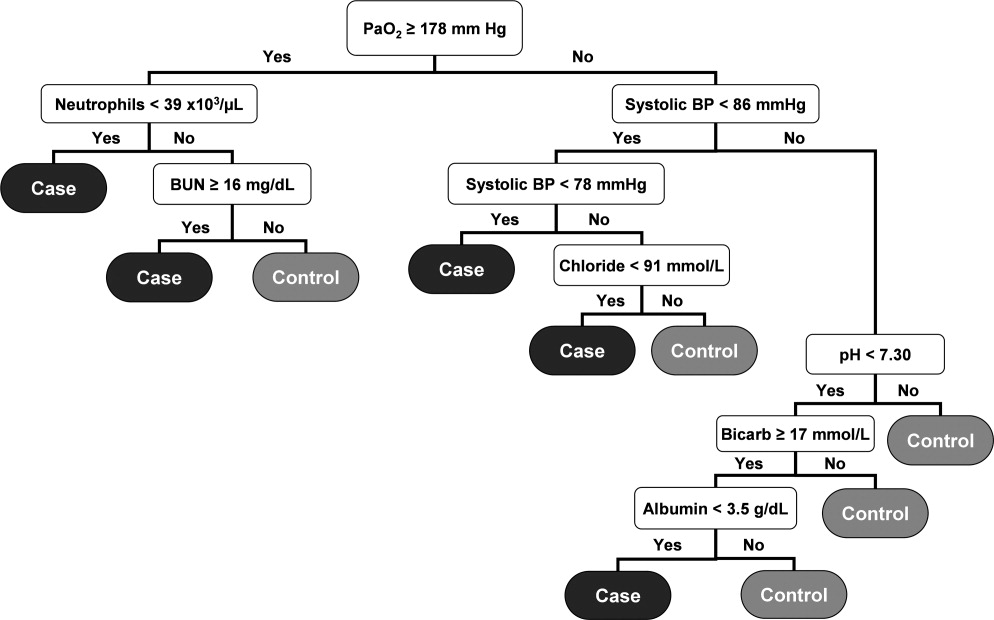
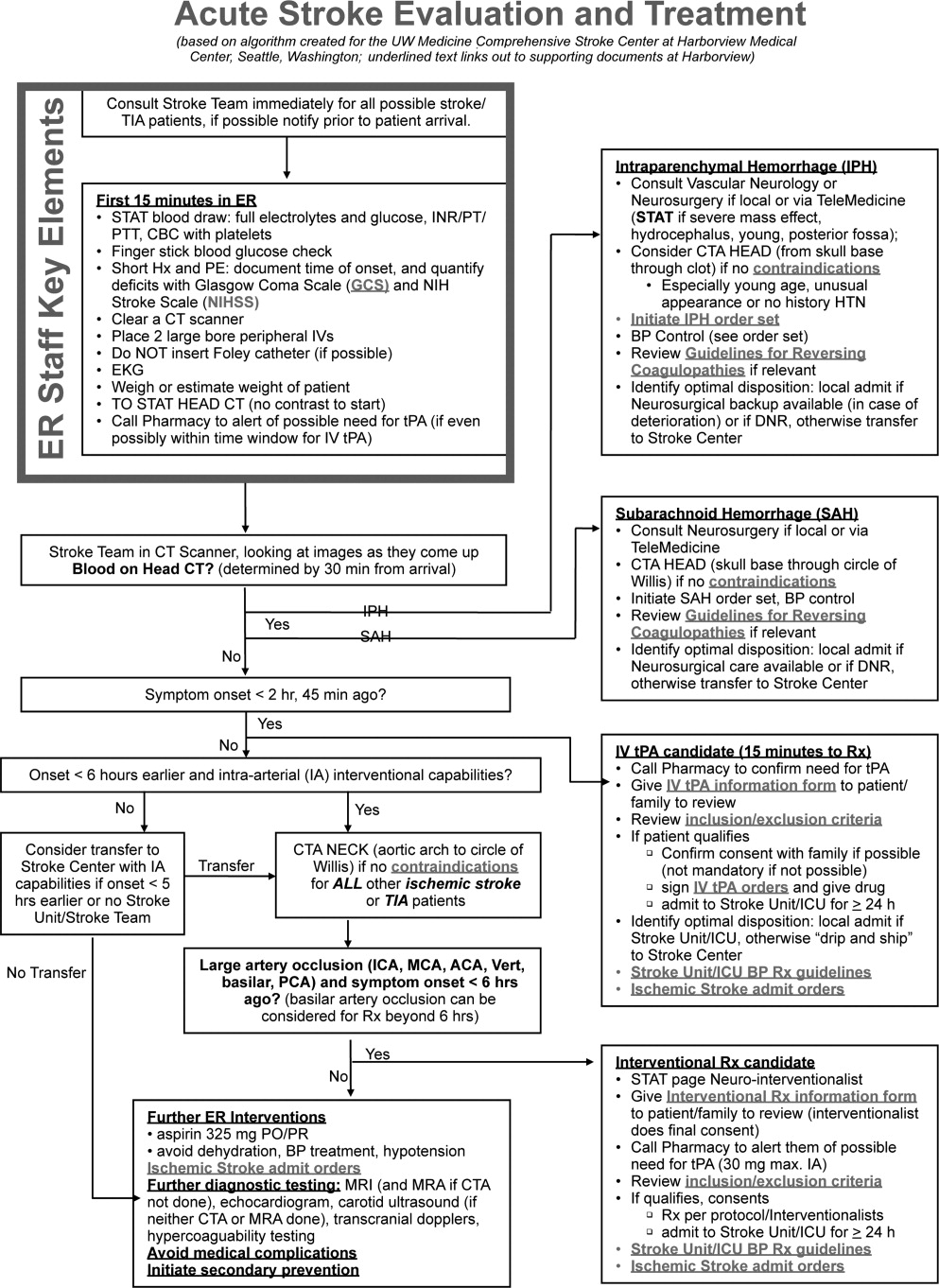
User login
Top 10 Infectious Disease Pitfalls
Hospitalists commonly encounter the challenges of infectious diseases in their hospitalized patients. Choosing the correct antibiotic, interpreting blood cultures, working up causes of fever, treating patients with an allergy to penicillin, and caring for patients with human immunodeficiency virus (HIV) commonly confront the hospitalist. This article presents evidence‐based pearls which will help hospitalists avoid common infectious disease pitfalls and guide their decision about when to consult an infectious diseases specialist.
1. Avoid Spiraling Empiricism and Understand Common Fallacies in Prescribing Empiric Antimicrobial Therapy
The term spiraling empiricism describes the inappropriate treatment, or the unjustifiable escalation of treatment, of suspected but undocumented infectious diseases.1 Initiation of carefully considered empiric broad‐spectrum antibiotic therapy for an acutely ill patient is an entirely appropriate and reasonable strategy. But all too often, practitioners are confronted with clinical dilemmas such as persistent fever or lack of response to therapy. In these circumstances, clinicians are faced with deciding whether to add or change antibiotics to broaden coverage. Changes in empiric therapy should be made sparingly, and only when there is new information or symptoms to justify an addition or change. In order to make an accurate assessment of response, steady‐state levels should be achieved and usually 3 to 5 days should be allowed to pass. Lack of response to broad‐spectrum therapy should trigger further investigation for occult infection or consideration of noninfectious etiologies and not simply the addition of a new antimicrobial agent. If a microbial pathogen is isolated from a blood culture(s) or other relevant source, antimicrobials should be tailored to the narrowest spectrum and least toxic therapy based on the sensitivities of that organism. For critically ill patients or patients who do not appear to be improving, an infectious diseases consultation may be warranted.
2. Know the Important Drug‐Drug Interactions Between Antimicrobials and Commonly‐used Inpatient Medications, Particularly With Those Involving Warfarin
Most antimicrobials (especially antifungals, quinolones, metronidazole, and sulfonamides) can cause unpredictable elevations in the international normalized ratio (INR) concurrent with warfarin administration, either through inhibition of warfarin metabolism or alterations in vitamin Kproducing gut flora. When using antimicrobials in patients on warfarin, the patient's INR should be carefully monitored and adjustment of the warfarin dose may be necessary. Antimicrobials that are inhibitors of cytochrome P‐450 enzymes include ciprofloxacin, levofloxacin, isoniazid, fluconazole, and clarithromycin. In contrast, rifampin is a potent inducer of most known cytochrome P‐450 enzymes and increases the metabolism of many drugs used in patients in the hospital setting, including anticonvulsants, beta‐blockers, calcium channel blockers, and other antibiotics like fluoroquinolones, and sulfonylureas. Moreover, the concurrent oral intake of tablets or solutions (including tube feeds) with a high concentration of trivalent and divalent cations (such as aluminum, magnesium, and, to a lesser extent, calcium, iron, and zinc) impairs gastrointestinal absorption of fluoroquinolones and should be avoided or spaced apart in time. Since fluoroquinolones can potentially prolong the QT interval, careful monitoring is necessary when a patient is prescribed other QT prolonging agents. Finally, many antimicrobials reduce the effectiveness of oral or other systemic hormonal contraceptives and patients should be routinely advised to use nonhormonal methods of birth control during therapy.
3. Positive Blood Cultures for Bacteria or Fungus Should be Repeated Serially Every 24 to 48 Hours Until the Cultures Are Negative
An important step in the management of a positive blood culture for bacteria or yeast is to check follow‐up blood cultures every 24 to 48 hours until the bacteremia or fungemia has cleared. This is particularly true of bacteremia caused by Staphylococcus aureus (S. aureus), Enterococcus species, and fungemia caused by Candida species. The duration of bacteremia or fungemia has a significant impact on the predictive values of further testing for endovascular or deep‐seated sources of infection as well as treatment duration. This is particularly true for the treatment of candidemia in nonneutropenic adults and for bacterial endocarditis, in which the recommended duration of treatment starts from the day of the last positive blood culture.2, 3 In addition to repeat blood cultures, a blood culture positive for S. aureus should always prompt an aggressive workup for a source (including strong consideration of a transesophageal echocardiogram to evaluate for endocarditis). S. aureus bacteremia should never be disregarded as a contaminant, and should prompt strong consideration of removal of all indwelling intravenous lines.4
4. Removal of Indwelling Intravascular Catheters Is Essential in the Management of Patients with Candidemia. In These Patients, Retention of Central Lines Is Significantly Related to Poor Outcomes
In patients with culture‐proven Candida fungemia, all intravascular catheters must be removed if at all possible. In a study by Nguyen et al.,5 the mortality rate for patients with a catheter‐related candidemia in whom catheters were retained was significantly higher than that of patients in whom the catheters were removed (41% vs. 21%, P 0.001). Likewise, in a separate study, Luzzati et al.6 noted that central line removal independently reduced the high mortality of the disease. This recommendation applies to all Candida species.
5. Although Candida Species Are Frequently Noted to Colonize Sputum and Urine Cultures, Their Recovery From Multiple Sites May Be an Indicator of Occult Candidemia in an Acutely Ill Patient
Candida species uncommonly cause pneumonia or urinary tract infection, so their isolation from cultures of the respiratory and genitourinary tract often represents colonization. However, the presence of Candida species at multiple sites may be an indicator of occult candidemia in a patient with multiple risk factors for candidemia, including intensive care unit (ICU) admission, immunosuppression (particularly neutropenia and recent receipt of corticosteroids), central venous catheterization, total parenteral nutrition, recent broad‐spectrum antibiotics, and recent abdominal or gastrointestinal surgery.7
6. Patients with Asymptomatic Bacteriuria, With or Without Pyuria, Should Not Be Treated with Antibiotics. Pregnant Women and Patients Undergoing a Genitourinary Procedure Are the Exception and Should Be Treated With Antibiotics
Asymptomatic bacteriuria is commonly encountered in the hospital setting, but is usually benign. Bacteriuria is defined as a voided urine specimen with 1 bacterial species isolated in a quantitative count of 105 cfu/mL. Treatment of asymptomatic bacteriuria is only recommended for pregnant women or prior to invasive genitourinary procedures, including transurethral resection of the prostate. Patients with structural or functional abnormalities of the urinary tract may have a high prevalence of bacteriuria. Despite its prevalence, asymptomatic bacteriuria is seldom associated with adverse outcomes. Studies have noted that antimicrobial treatment of asymptomatic bacteriuria does not decrease recurrence. Negative outcomes with antimicrobial treatment do occur, including adverse drug reactions and reinfection with organisms of increasing resistance. Clinical trials in spinal‐cord injury patients, diabetic women, elderly patients living in the community or nursing home, and patients with indwelling urethral catheters have consistently found no benefit with treatment of asymptomatic bacteriuria.8, 9 The presence or absence of pyuria does not differentiate symptomatic from asymptomatic urinary infection. Patients with symptomatic urinary tract infection (fever and/or dysuria) should be treated after urine cultures are obtained. Other causes of pyuria in the absence of an acute urinary tract infection include urethritis, tuberculosis, prostatitis, nephrolithiasis, and malignancy.
7. Evaluate All Patients Who Have a History of Penicillin Allergy and Consider Desensitization for Patients With a History Consistent With Immunoglobulin Emediated Allergy Who Require Treatment With a Beta‐Lactam Antibiotic
Patients commonly claim to have an allergy to penicillin. True penicillin allergy is very serious and can be life‐threatening. Because of this, patients labeled as penicillin allergic are typically not treated with beta‐lactam antibiotics. Instead, they may be prescribed medications which are typically less effective, more toxic, have a broader spectrum, or are more expensive.10, 11 Many patients are inappropriately labeled as having a penicillin allergy. A history of penicillin allergy is reported in approximately 10% of hospitalized patients, but only approximately 10% of those who report a history of penicillin allergy actually have an allergic reaction when treated with penicillin. Exanthems are frequently associated with beta‐lactam use during an episode of infectious mononucleosis but these are not considered an allergic reaction. Such patients are generally able to tolerate beta‐lactams subsequent to this episode. Nonpruritic maculopapular rashes are also reported in 3% to 7% of children taking amoxicillin and are not a contraindication for future beta‐lactam or cephalosporin use.12 All patients who describe an allergy should be questioned in detail about the type of penicillin received, as well as the type, severity, and timing of the reaction. Typical immunoglobulin E (IgE)‐mediated severe reactions to penicillin include urticaria, pruritus, angioedema, bronchospasm, and hypotension. These patients should not be given other agents that share the same beta‐lactam ring, including cephalosporins (risk of cross‐reactivity is greatest with first‐generation and second‐generation cephalosporins). Carbapenems have minimal cross‐reactivity, particularly meropenem.13 Monobactams (eg, aztreonam) do not cross‐react. While skin testing to penicillin can be considered in patients with a history of a severe reaction to penicillin, neither the major nor minor determinants are commercially available at this time. In patients with a history of a possible IgE‐mediated reaction and when there is no suitable alternative antibiotic (usually determined from infectious diseases consultation), desensitization to beta‐lactams or carbapenems can be considered. Desensitization should be reserved only for clinicians experienced with these techniques, preferably in consultation with a specialist in allergy and immunology. Patients who report a non‐IgE‐mediated reaction may be prescribed a cephalosporin if necessary (preferably a third‐generation or fourth‐generation).14
8. An Abrupt Increase in Leukocytosis In a Hospitalized Patient Should Prompt Consideration of Clostridium difficile Infection
In recent years, there has been a marked increase in the incidence and severity of Clostridium difficile (C. difficile) infection (CDI). A new hypervirulent strain, NAP1/BI/027, has emerged and is becoming endemic in the United States, Canada, and Europe. Typically C. difficile causes diarrhea, abdominal pain, and fever. Often patients have received antibiotics in the recent past, placing them at higher risk, but cases can occur sporadically (even in the community setting) or be transmitted nosocomially. Early detection appears to be essential in reducing the serious morbidity and mortality associated with this disease. Observational studies suggested that C. difficile infection is a common cause of unexplained leukocytosis or a sudden worsening of preexisting leukocytosis.15, 16 In a prospective study evaluating 60 patients with unexplained leukocytosis (white blood cell count 15,000/mm3), 58% of patients with leukocytosis in the absence of localizing symptoms and signs of infection were subsequently diagnosed with CDI. The authors believe that the percent may have been as high as 73% when they included patients with a negative toxin assay who rapidly responded to metronidazole therapy.17 White blood cell counts can range from 10,000 to 20,000/mm3 in moderate disease. Counts as high as 40,000/mm3 can occur, especially in patients with severe disease. Although the use of clindamycin and cephalosporins have been classically associated with the subsequent development of CDI, the current widespread use of fluoroquinolones has led to significant fluoroquinolone resistance among strains of C. difficile, especially the hypervirulent NAP1/BI/027 strain.18 The judicious use of antibiotics, especially fluoroquinolones, remains the cornerstone in preventing CDI. Remember that hand washing with soap and water is essential as alcohol‐based hand sanitizers do not eradicate the C. difficile spores. The drug of choice for initial treatment of mild to moderate CDI remains oral metronidazole, and it may be used for a first recurrence of CDI. Increasing data support the use of oral vancomycin for moderately severe to severe CDI or for multiple recurrences.19 Intravenous metronidazole is often added to oral vancomycin in patients with ileus, but it is not reliably effective alone for CDI.
9. Fever Is Common in the First 48 Hours After a Major Surgical Procedure, and Is a Poor Indicator of Infection. The use of Antibiotics in Response to Fever in the Absence of Other Localizing Signs and Symptoms of Infection Should Be Avoided
Early postoperative fever is relatively common but most fevers that develop within the first 48 hours after surgery do not have an infectious etiology.2023 However, fever that begins or persists beyond the fifth postoperative day is much more likely to represent a clinically significant infection. The continued use of antibiotics outside the window for wound prophylaxis (>24 hours) does not decrease the risk of postoperative infection but it does increase the risk of acquiring resistant bacteria and adverse drug reactions, including CDI.
10. Facts All Clinicians Should Know About Patients with HIV Infection
The 2 most common laboratory abnormalities routinely associated with antiretroviral therapy for HIV infection are unconjugated hyperbilirubinemia associated with atazanavir and an elevated mean corpuscular volume (MCV) associated with zidovudine (and, to a lesser extent, stavudine). Immune reconstitution inflammatory syndrome (IRIS) is a condition seen in patients with advanced acquired immune deficiency syndrome (AIDS) who have recently started antiretroviral therapy. As the immune system begins to recover, it may respond to a previously acquired opportunistic infection with an overwhelming inflammatory response that paradoxically makes the symptoms of infection worse. IRIS is associated with a pathological inflammatory response that can have substantial morbidity and mortality.24 For this reason, when considering whether to start or stop continuous or highly active antiretroviral therapy (also known as HAART), an infectious diseases consult is recommended. Pneumocystis jiroveci (PCP) remains a cause of pneumonia in patients with advanced AIDS' though in the era of HAART, its presentation may be more subtle. Finally, the principle of parsimony (Occam's razor) often does not hold in the diagnosis of opportunistic infections in patients with advanced AIDS, as these patients can often present with multiple infections simultaneously.25, 26
Conclusion
Infectious diseases are commonly encountered by physicians who care for hospitalized patients. Early recognition, evaluation, and appropriate treatment and/or referral to an infectious diseases specialist are necessary to moderate the significant morbidity and mortality that are often associated with infectious diseases.
- ,.Observations on spiraling empiricism: its causes, allure, and perils, with particular reference to antibiotic therapy.Am J Med.1989;87(2):201–206.
- ,,, et al.Guidelines for the treatment of candidiasis.Clin Infect Dis.2004;38:161–189.
- ,,, et al.Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America.Circulation.2005;111:e394–e434.
- ,.Management of methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46:S386–S393.
- ,,, et al.Therapeutic approaches in patients with candidemia. Evaluation in a multicenter, prospective, observational study.Arch Intern Med.1995;155(22):2429–2435.
- ,,, et al.Nosocomial candidemia in non‐neutropenic patients at an Italian tertiary care hospital.Eur J Clin Microbiol Infect Dis.2000;19(8):602–607.
- .Candidemia in adults. In: Marr KA, ed.UpToDate.Waltham, MA:UpToDate, Inc.;2008.
- ,,, et al.Infectious Diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults.Clin Infect Dis.2005;40(5):643–654.
- .Asymptomatic bacteriuria: when to screen and when to treat.Infect Dis Clin North Am.2003;17(2):367–394.
- .Management of patients with a history of allergy to beta‐lactam antibiotics.Am J Med.2008;121(7):572–576.
- ,,.Practical aspects of choosing an antibiotic for patients with a reported allergy to an antibiotic.Clin Infect Dis.2002;35(1):26–31.
- ,,, et al.Adverse effects of orally administered ampicillin.J Pediatr.1973;83:106–108.
- ,,, et al.Brief communication: tolerability of meropenem in patients with IgE‐mediated hypersensitivity to penicillins.Ann Intern Med.2007;146(4):266–269.
- ,,.The rational clinical examination. Is this patient allergic to penicillin? An evidence‐based analysis of the likelihood of penicillin allergy.JAMA.2001;285(19):2498–2505.
- ,,.Leukocytosis in a tertiary care hospital with particular attention to the role of infection caused by Clostridium difficile.Clin Infect Dis.2002;34:1585–1592.
- ,,,.Leukocytosis as a harbinger and surrogate marker of Clostridium difficile infection in hospitalized patients with diarrhea.Am J Gastroenterol.2000;95:3137–3141.
- ,,,.Clostridium difficile infection in patients with unexplained leukocytosis.Am J Med.2003;115:543–546.
- ,.The challenges posed by reemerging Clostridium difficile infection.Clin Infect Dis.2007;45(2):222–227.
- ,,.Treatment of Clostridium difficile infection.Clin Infect Dis.2008;46(suppl 1):S32–S42.
- ,,,,.Diagnostic accuracy of routine postoperative body temperature measurements.Clin Infect Dis.2005;40:1404–1410.
- .Should we measure body temperature for patients who have recently undergone surgery?Clin Infect Dis.2005;40(10):1411–1412.
- ,,,.Evidence for the noninfectious etiology of early postoperative fever.Infect Control.1985;6:273–277.
- .Evaluating postoperative fever: a focused approach.Cleve Clin J Med.2006;73(suppl 1):S62–S66.
- ,,.Immune reconstitution inflammatory syndrome: more answers, more questions.J Antimicrob Chemother.2006;57(2):167–170.
- ,,,,.Clinical problem‐solving. Occam's razor versus Saint's Triad.N Engl J Med.2004;350(6):599–603.
- ,.William of Occam and Occam's razor.Ann Intern Med.2002;136(8):634–635.
Hospitalists commonly encounter the challenges of infectious diseases in their hospitalized patients. Choosing the correct antibiotic, interpreting blood cultures, working up causes of fever, treating patients with an allergy to penicillin, and caring for patients with human immunodeficiency virus (HIV) commonly confront the hospitalist. This article presents evidence‐based pearls which will help hospitalists avoid common infectious disease pitfalls and guide their decision about when to consult an infectious diseases specialist.
1. Avoid Spiraling Empiricism and Understand Common Fallacies in Prescribing Empiric Antimicrobial Therapy
The term spiraling empiricism describes the inappropriate treatment, or the unjustifiable escalation of treatment, of suspected but undocumented infectious diseases.1 Initiation of carefully considered empiric broad‐spectrum antibiotic therapy for an acutely ill patient is an entirely appropriate and reasonable strategy. But all too often, practitioners are confronted with clinical dilemmas such as persistent fever or lack of response to therapy. In these circumstances, clinicians are faced with deciding whether to add or change antibiotics to broaden coverage. Changes in empiric therapy should be made sparingly, and only when there is new information or symptoms to justify an addition or change. In order to make an accurate assessment of response, steady‐state levels should be achieved and usually 3 to 5 days should be allowed to pass. Lack of response to broad‐spectrum therapy should trigger further investigation for occult infection or consideration of noninfectious etiologies and not simply the addition of a new antimicrobial agent. If a microbial pathogen is isolated from a blood culture(s) or other relevant source, antimicrobials should be tailored to the narrowest spectrum and least toxic therapy based on the sensitivities of that organism. For critically ill patients or patients who do not appear to be improving, an infectious diseases consultation may be warranted.
2. Know the Important Drug‐Drug Interactions Between Antimicrobials and Commonly‐used Inpatient Medications, Particularly With Those Involving Warfarin
Most antimicrobials (especially antifungals, quinolones, metronidazole, and sulfonamides) can cause unpredictable elevations in the international normalized ratio (INR) concurrent with warfarin administration, either through inhibition of warfarin metabolism or alterations in vitamin Kproducing gut flora. When using antimicrobials in patients on warfarin, the patient's INR should be carefully monitored and adjustment of the warfarin dose may be necessary. Antimicrobials that are inhibitors of cytochrome P‐450 enzymes include ciprofloxacin, levofloxacin, isoniazid, fluconazole, and clarithromycin. In contrast, rifampin is a potent inducer of most known cytochrome P‐450 enzymes and increases the metabolism of many drugs used in patients in the hospital setting, including anticonvulsants, beta‐blockers, calcium channel blockers, and other antibiotics like fluoroquinolones, and sulfonylureas. Moreover, the concurrent oral intake of tablets or solutions (including tube feeds) with a high concentration of trivalent and divalent cations (such as aluminum, magnesium, and, to a lesser extent, calcium, iron, and zinc) impairs gastrointestinal absorption of fluoroquinolones and should be avoided or spaced apart in time. Since fluoroquinolones can potentially prolong the QT interval, careful monitoring is necessary when a patient is prescribed other QT prolonging agents. Finally, many antimicrobials reduce the effectiveness of oral or other systemic hormonal contraceptives and patients should be routinely advised to use nonhormonal methods of birth control during therapy.
3. Positive Blood Cultures for Bacteria or Fungus Should be Repeated Serially Every 24 to 48 Hours Until the Cultures Are Negative
An important step in the management of a positive blood culture for bacteria or yeast is to check follow‐up blood cultures every 24 to 48 hours until the bacteremia or fungemia has cleared. This is particularly true of bacteremia caused by Staphylococcus aureus (S. aureus), Enterococcus species, and fungemia caused by Candida species. The duration of bacteremia or fungemia has a significant impact on the predictive values of further testing for endovascular or deep‐seated sources of infection as well as treatment duration. This is particularly true for the treatment of candidemia in nonneutropenic adults and for bacterial endocarditis, in which the recommended duration of treatment starts from the day of the last positive blood culture.2, 3 In addition to repeat blood cultures, a blood culture positive for S. aureus should always prompt an aggressive workup for a source (including strong consideration of a transesophageal echocardiogram to evaluate for endocarditis). S. aureus bacteremia should never be disregarded as a contaminant, and should prompt strong consideration of removal of all indwelling intravenous lines.4
4. Removal of Indwelling Intravascular Catheters Is Essential in the Management of Patients with Candidemia. In These Patients, Retention of Central Lines Is Significantly Related to Poor Outcomes
In patients with culture‐proven Candida fungemia, all intravascular catheters must be removed if at all possible. In a study by Nguyen et al.,5 the mortality rate for patients with a catheter‐related candidemia in whom catheters were retained was significantly higher than that of patients in whom the catheters were removed (41% vs. 21%, P 0.001). Likewise, in a separate study, Luzzati et al.6 noted that central line removal independently reduced the high mortality of the disease. This recommendation applies to all Candida species.
5. Although Candida Species Are Frequently Noted to Colonize Sputum and Urine Cultures, Their Recovery From Multiple Sites May Be an Indicator of Occult Candidemia in an Acutely Ill Patient
Candida species uncommonly cause pneumonia or urinary tract infection, so their isolation from cultures of the respiratory and genitourinary tract often represents colonization. However, the presence of Candida species at multiple sites may be an indicator of occult candidemia in a patient with multiple risk factors for candidemia, including intensive care unit (ICU) admission, immunosuppression (particularly neutropenia and recent receipt of corticosteroids), central venous catheterization, total parenteral nutrition, recent broad‐spectrum antibiotics, and recent abdominal or gastrointestinal surgery.7
6. Patients with Asymptomatic Bacteriuria, With or Without Pyuria, Should Not Be Treated with Antibiotics. Pregnant Women and Patients Undergoing a Genitourinary Procedure Are the Exception and Should Be Treated With Antibiotics
Asymptomatic bacteriuria is commonly encountered in the hospital setting, but is usually benign. Bacteriuria is defined as a voided urine specimen with 1 bacterial species isolated in a quantitative count of 105 cfu/mL. Treatment of asymptomatic bacteriuria is only recommended for pregnant women or prior to invasive genitourinary procedures, including transurethral resection of the prostate. Patients with structural or functional abnormalities of the urinary tract may have a high prevalence of bacteriuria. Despite its prevalence, asymptomatic bacteriuria is seldom associated with adverse outcomes. Studies have noted that antimicrobial treatment of asymptomatic bacteriuria does not decrease recurrence. Negative outcomes with antimicrobial treatment do occur, including adverse drug reactions and reinfection with organisms of increasing resistance. Clinical trials in spinal‐cord injury patients, diabetic women, elderly patients living in the community or nursing home, and patients with indwelling urethral catheters have consistently found no benefit with treatment of asymptomatic bacteriuria.8, 9 The presence or absence of pyuria does not differentiate symptomatic from asymptomatic urinary infection. Patients with symptomatic urinary tract infection (fever and/or dysuria) should be treated after urine cultures are obtained. Other causes of pyuria in the absence of an acute urinary tract infection include urethritis, tuberculosis, prostatitis, nephrolithiasis, and malignancy.
7. Evaluate All Patients Who Have a History of Penicillin Allergy and Consider Desensitization for Patients With a History Consistent With Immunoglobulin Emediated Allergy Who Require Treatment With a Beta‐Lactam Antibiotic
Patients commonly claim to have an allergy to penicillin. True penicillin allergy is very serious and can be life‐threatening. Because of this, patients labeled as penicillin allergic are typically not treated with beta‐lactam antibiotics. Instead, they may be prescribed medications which are typically less effective, more toxic, have a broader spectrum, or are more expensive.10, 11 Many patients are inappropriately labeled as having a penicillin allergy. A history of penicillin allergy is reported in approximately 10% of hospitalized patients, but only approximately 10% of those who report a history of penicillin allergy actually have an allergic reaction when treated with penicillin. Exanthems are frequently associated with beta‐lactam use during an episode of infectious mononucleosis but these are not considered an allergic reaction. Such patients are generally able to tolerate beta‐lactams subsequent to this episode. Nonpruritic maculopapular rashes are also reported in 3% to 7% of children taking amoxicillin and are not a contraindication for future beta‐lactam or cephalosporin use.12 All patients who describe an allergy should be questioned in detail about the type of penicillin received, as well as the type, severity, and timing of the reaction. Typical immunoglobulin E (IgE)‐mediated severe reactions to penicillin include urticaria, pruritus, angioedema, bronchospasm, and hypotension. These patients should not be given other agents that share the same beta‐lactam ring, including cephalosporins (risk of cross‐reactivity is greatest with first‐generation and second‐generation cephalosporins). Carbapenems have minimal cross‐reactivity, particularly meropenem.13 Monobactams (eg, aztreonam) do not cross‐react. While skin testing to penicillin can be considered in patients with a history of a severe reaction to penicillin, neither the major nor minor determinants are commercially available at this time. In patients with a history of a possible IgE‐mediated reaction and when there is no suitable alternative antibiotic (usually determined from infectious diseases consultation), desensitization to beta‐lactams or carbapenems can be considered. Desensitization should be reserved only for clinicians experienced with these techniques, preferably in consultation with a specialist in allergy and immunology. Patients who report a non‐IgE‐mediated reaction may be prescribed a cephalosporin if necessary (preferably a third‐generation or fourth‐generation).14
8. An Abrupt Increase in Leukocytosis In a Hospitalized Patient Should Prompt Consideration of Clostridium difficile Infection
In recent years, there has been a marked increase in the incidence and severity of Clostridium difficile (C. difficile) infection (CDI). A new hypervirulent strain, NAP1/BI/027, has emerged and is becoming endemic in the United States, Canada, and Europe. Typically C. difficile causes diarrhea, abdominal pain, and fever. Often patients have received antibiotics in the recent past, placing them at higher risk, but cases can occur sporadically (even in the community setting) or be transmitted nosocomially. Early detection appears to be essential in reducing the serious morbidity and mortality associated with this disease. Observational studies suggested that C. difficile infection is a common cause of unexplained leukocytosis or a sudden worsening of preexisting leukocytosis.15, 16 In a prospective study evaluating 60 patients with unexplained leukocytosis (white blood cell count 15,000/mm3), 58% of patients with leukocytosis in the absence of localizing symptoms and signs of infection were subsequently diagnosed with CDI. The authors believe that the percent may have been as high as 73% when they included patients with a negative toxin assay who rapidly responded to metronidazole therapy.17 White blood cell counts can range from 10,000 to 20,000/mm3 in moderate disease. Counts as high as 40,000/mm3 can occur, especially in patients with severe disease. Although the use of clindamycin and cephalosporins have been classically associated with the subsequent development of CDI, the current widespread use of fluoroquinolones has led to significant fluoroquinolone resistance among strains of C. difficile, especially the hypervirulent NAP1/BI/027 strain.18 The judicious use of antibiotics, especially fluoroquinolones, remains the cornerstone in preventing CDI. Remember that hand washing with soap and water is essential as alcohol‐based hand sanitizers do not eradicate the C. difficile spores. The drug of choice for initial treatment of mild to moderate CDI remains oral metronidazole, and it may be used for a first recurrence of CDI. Increasing data support the use of oral vancomycin for moderately severe to severe CDI or for multiple recurrences.19 Intravenous metronidazole is often added to oral vancomycin in patients with ileus, but it is not reliably effective alone for CDI.
9. Fever Is Common in the First 48 Hours After a Major Surgical Procedure, and Is a Poor Indicator of Infection. The use of Antibiotics in Response to Fever in the Absence of Other Localizing Signs and Symptoms of Infection Should Be Avoided
Early postoperative fever is relatively common but most fevers that develop within the first 48 hours after surgery do not have an infectious etiology.2023 However, fever that begins or persists beyond the fifth postoperative day is much more likely to represent a clinically significant infection. The continued use of antibiotics outside the window for wound prophylaxis (>24 hours) does not decrease the risk of postoperative infection but it does increase the risk of acquiring resistant bacteria and adverse drug reactions, including CDI.
10. Facts All Clinicians Should Know About Patients with HIV Infection
The 2 most common laboratory abnormalities routinely associated with antiretroviral therapy for HIV infection are unconjugated hyperbilirubinemia associated with atazanavir and an elevated mean corpuscular volume (MCV) associated with zidovudine (and, to a lesser extent, stavudine). Immune reconstitution inflammatory syndrome (IRIS) is a condition seen in patients with advanced acquired immune deficiency syndrome (AIDS) who have recently started antiretroviral therapy. As the immune system begins to recover, it may respond to a previously acquired opportunistic infection with an overwhelming inflammatory response that paradoxically makes the symptoms of infection worse. IRIS is associated with a pathological inflammatory response that can have substantial morbidity and mortality.24 For this reason, when considering whether to start or stop continuous or highly active antiretroviral therapy (also known as HAART), an infectious diseases consult is recommended. Pneumocystis jiroveci (PCP) remains a cause of pneumonia in patients with advanced AIDS' though in the era of HAART, its presentation may be more subtle. Finally, the principle of parsimony (Occam's razor) often does not hold in the diagnosis of opportunistic infections in patients with advanced AIDS, as these patients can often present with multiple infections simultaneously.25, 26
Conclusion
Infectious diseases are commonly encountered by physicians who care for hospitalized patients. Early recognition, evaluation, and appropriate treatment and/or referral to an infectious diseases specialist are necessary to moderate the significant morbidity and mortality that are often associated with infectious diseases.
Hospitalists commonly encounter the challenges of infectious diseases in their hospitalized patients. Choosing the correct antibiotic, interpreting blood cultures, working up causes of fever, treating patients with an allergy to penicillin, and caring for patients with human immunodeficiency virus (HIV) commonly confront the hospitalist. This article presents evidence‐based pearls which will help hospitalists avoid common infectious disease pitfalls and guide their decision about when to consult an infectious diseases specialist.
1. Avoid Spiraling Empiricism and Understand Common Fallacies in Prescribing Empiric Antimicrobial Therapy
The term spiraling empiricism describes the inappropriate treatment, or the unjustifiable escalation of treatment, of suspected but undocumented infectious diseases.1 Initiation of carefully considered empiric broad‐spectrum antibiotic therapy for an acutely ill patient is an entirely appropriate and reasonable strategy. But all too often, practitioners are confronted with clinical dilemmas such as persistent fever or lack of response to therapy. In these circumstances, clinicians are faced with deciding whether to add or change antibiotics to broaden coverage. Changes in empiric therapy should be made sparingly, and only when there is new information or symptoms to justify an addition or change. In order to make an accurate assessment of response, steady‐state levels should be achieved and usually 3 to 5 days should be allowed to pass. Lack of response to broad‐spectrum therapy should trigger further investigation for occult infection or consideration of noninfectious etiologies and not simply the addition of a new antimicrobial agent. If a microbial pathogen is isolated from a blood culture(s) or other relevant source, antimicrobials should be tailored to the narrowest spectrum and least toxic therapy based on the sensitivities of that organism. For critically ill patients or patients who do not appear to be improving, an infectious diseases consultation may be warranted.
2. Know the Important Drug‐Drug Interactions Between Antimicrobials and Commonly‐used Inpatient Medications, Particularly With Those Involving Warfarin
Most antimicrobials (especially antifungals, quinolones, metronidazole, and sulfonamides) can cause unpredictable elevations in the international normalized ratio (INR) concurrent with warfarin administration, either through inhibition of warfarin metabolism or alterations in vitamin Kproducing gut flora. When using antimicrobials in patients on warfarin, the patient's INR should be carefully monitored and adjustment of the warfarin dose may be necessary. Antimicrobials that are inhibitors of cytochrome P‐450 enzymes include ciprofloxacin, levofloxacin, isoniazid, fluconazole, and clarithromycin. In contrast, rifampin is a potent inducer of most known cytochrome P‐450 enzymes and increases the metabolism of many drugs used in patients in the hospital setting, including anticonvulsants, beta‐blockers, calcium channel blockers, and other antibiotics like fluoroquinolones, and sulfonylureas. Moreover, the concurrent oral intake of tablets or solutions (including tube feeds) with a high concentration of trivalent and divalent cations (such as aluminum, magnesium, and, to a lesser extent, calcium, iron, and zinc) impairs gastrointestinal absorption of fluoroquinolones and should be avoided or spaced apart in time. Since fluoroquinolones can potentially prolong the QT interval, careful monitoring is necessary when a patient is prescribed other QT prolonging agents. Finally, many antimicrobials reduce the effectiveness of oral or other systemic hormonal contraceptives and patients should be routinely advised to use nonhormonal methods of birth control during therapy.
3. Positive Blood Cultures for Bacteria or Fungus Should be Repeated Serially Every 24 to 48 Hours Until the Cultures Are Negative
An important step in the management of a positive blood culture for bacteria or yeast is to check follow‐up blood cultures every 24 to 48 hours until the bacteremia or fungemia has cleared. This is particularly true of bacteremia caused by Staphylococcus aureus (S. aureus), Enterococcus species, and fungemia caused by Candida species. The duration of bacteremia or fungemia has a significant impact on the predictive values of further testing for endovascular or deep‐seated sources of infection as well as treatment duration. This is particularly true for the treatment of candidemia in nonneutropenic adults and for bacterial endocarditis, in which the recommended duration of treatment starts from the day of the last positive blood culture.2, 3 In addition to repeat blood cultures, a blood culture positive for S. aureus should always prompt an aggressive workup for a source (including strong consideration of a transesophageal echocardiogram to evaluate for endocarditis). S. aureus bacteremia should never be disregarded as a contaminant, and should prompt strong consideration of removal of all indwelling intravenous lines.4
4. Removal of Indwelling Intravascular Catheters Is Essential in the Management of Patients with Candidemia. In These Patients, Retention of Central Lines Is Significantly Related to Poor Outcomes
In patients with culture‐proven Candida fungemia, all intravascular catheters must be removed if at all possible. In a study by Nguyen et al.,5 the mortality rate for patients with a catheter‐related candidemia in whom catheters were retained was significantly higher than that of patients in whom the catheters were removed (41% vs. 21%, P 0.001). Likewise, in a separate study, Luzzati et al.6 noted that central line removal independently reduced the high mortality of the disease. This recommendation applies to all Candida species.
5. Although Candida Species Are Frequently Noted to Colonize Sputum and Urine Cultures, Their Recovery From Multiple Sites May Be an Indicator of Occult Candidemia in an Acutely Ill Patient
Candida species uncommonly cause pneumonia or urinary tract infection, so their isolation from cultures of the respiratory and genitourinary tract often represents colonization. However, the presence of Candida species at multiple sites may be an indicator of occult candidemia in a patient with multiple risk factors for candidemia, including intensive care unit (ICU) admission, immunosuppression (particularly neutropenia and recent receipt of corticosteroids), central venous catheterization, total parenteral nutrition, recent broad‐spectrum antibiotics, and recent abdominal or gastrointestinal surgery.7
6. Patients with Asymptomatic Bacteriuria, With or Without Pyuria, Should Not Be Treated with Antibiotics. Pregnant Women and Patients Undergoing a Genitourinary Procedure Are the Exception and Should Be Treated With Antibiotics
Asymptomatic bacteriuria is commonly encountered in the hospital setting, but is usually benign. Bacteriuria is defined as a voided urine specimen with 1 bacterial species isolated in a quantitative count of 105 cfu/mL. Treatment of asymptomatic bacteriuria is only recommended for pregnant women or prior to invasive genitourinary procedures, including transurethral resection of the prostate. Patients with structural or functional abnormalities of the urinary tract may have a high prevalence of bacteriuria. Despite its prevalence, asymptomatic bacteriuria is seldom associated with adverse outcomes. Studies have noted that antimicrobial treatment of asymptomatic bacteriuria does not decrease recurrence. Negative outcomes with antimicrobial treatment do occur, including adverse drug reactions and reinfection with organisms of increasing resistance. Clinical trials in spinal‐cord injury patients, diabetic women, elderly patients living in the community or nursing home, and patients with indwelling urethral catheters have consistently found no benefit with treatment of asymptomatic bacteriuria.8, 9 The presence or absence of pyuria does not differentiate symptomatic from asymptomatic urinary infection. Patients with symptomatic urinary tract infection (fever and/or dysuria) should be treated after urine cultures are obtained. Other causes of pyuria in the absence of an acute urinary tract infection include urethritis, tuberculosis, prostatitis, nephrolithiasis, and malignancy.
7. Evaluate All Patients Who Have a History of Penicillin Allergy and Consider Desensitization for Patients With a History Consistent With Immunoglobulin Emediated Allergy Who Require Treatment With a Beta‐Lactam Antibiotic
Patients commonly claim to have an allergy to penicillin. True penicillin allergy is very serious and can be life‐threatening. Because of this, patients labeled as penicillin allergic are typically not treated with beta‐lactam antibiotics. Instead, they may be prescribed medications which are typically less effective, more toxic, have a broader spectrum, or are more expensive.10, 11 Many patients are inappropriately labeled as having a penicillin allergy. A history of penicillin allergy is reported in approximately 10% of hospitalized patients, but only approximately 10% of those who report a history of penicillin allergy actually have an allergic reaction when treated with penicillin. Exanthems are frequently associated with beta‐lactam use during an episode of infectious mononucleosis but these are not considered an allergic reaction. Such patients are generally able to tolerate beta‐lactams subsequent to this episode. Nonpruritic maculopapular rashes are also reported in 3% to 7% of children taking amoxicillin and are not a contraindication for future beta‐lactam or cephalosporin use.12 All patients who describe an allergy should be questioned in detail about the type of penicillin received, as well as the type, severity, and timing of the reaction. Typical immunoglobulin E (IgE)‐mediated severe reactions to penicillin include urticaria, pruritus, angioedema, bronchospasm, and hypotension. These patients should not be given other agents that share the same beta‐lactam ring, including cephalosporins (risk of cross‐reactivity is greatest with first‐generation and second‐generation cephalosporins). Carbapenems have minimal cross‐reactivity, particularly meropenem.13 Monobactams (eg, aztreonam) do not cross‐react. While skin testing to penicillin can be considered in patients with a history of a severe reaction to penicillin, neither the major nor minor determinants are commercially available at this time. In patients with a history of a possible IgE‐mediated reaction and when there is no suitable alternative antibiotic (usually determined from infectious diseases consultation), desensitization to beta‐lactams or carbapenems can be considered. Desensitization should be reserved only for clinicians experienced with these techniques, preferably in consultation with a specialist in allergy and immunology. Patients who report a non‐IgE‐mediated reaction may be prescribed a cephalosporin if necessary (preferably a third‐generation or fourth‐generation).14
8. An Abrupt Increase in Leukocytosis In a Hospitalized Patient Should Prompt Consideration of Clostridium difficile Infection
In recent years, there has been a marked increase in the incidence and severity of Clostridium difficile (C. difficile) infection (CDI). A new hypervirulent strain, NAP1/BI/027, has emerged and is becoming endemic in the United States, Canada, and Europe. Typically C. difficile causes diarrhea, abdominal pain, and fever. Often patients have received antibiotics in the recent past, placing them at higher risk, but cases can occur sporadically (even in the community setting) or be transmitted nosocomially. Early detection appears to be essential in reducing the serious morbidity and mortality associated with this disease. Observational studies suggested that C. difficile infection is a common cause of unexplained leukocytosis or a sudden worsening of preexisting leukocytosis.15, 16 In a prospective study evaluating 60 patients with unexplained leukocytosis (white blood cell count 15,000/mm3), 58% of patients with leukocytosis in the absence of localizing symptoms and signs of infection were subsequently diagnosed with CDI. The authors believe that the percent may have been as high as 73% when they included patients with a negative toxin assay who rapidly responded to metronidazole therapy.17 White blood cell counts can range from 10,000 to 20,000/mm3 in moderate disease. Counts as high as 40,000/mm3 can occur, especially in patients with severe disease. Although the use of clindamycin and cephalosporins have been classically associated with the subsequent development of CDI, the current widespread use of fluoroquinolones has led to significant fluoroquinolone resistance among strains of C. difficile, especially the hypervirulent NAP1/BI/027 strain.18 The judicious use of antibiotics, especially fluoroquinolones, remains the cornerstone in preventing CDI. Remember that hand washing with soap and water is essential as alcohol‐based hand sanitizers do not eradicate the C. difficile spores. The drug of choice for initial treatment of mild to moderate CDI remains oral metronidazole, and it may be used for a first recurrence of CDI. Increasing data support the use of oral vancomycin for moderately severe to severe CDI or for multiple recurrences.19 Intravenous metronidazole is often added to oral vancomycin in patients with ileus, but it is not reliably effective alone for CDI.
9. Fever Is Common in the First 48 Hours After a Major Surgical Procedure, and Is a Poor Indicator of Infection. The use of Antibiotics in Response to Fever in the Absence of Other Localizing Signs and Symptoms of Infection Should Be Avoided
Early postoperative fever is relatively common but most fevers that develop within the first 48 hours after surgery do not have an infectious etiology.2023 However, fever that begins or persists beyond the fifth postoperative day is much more likely to represent a clinically significant infection. The continued use of antibiotics outside the window for wound prophylaxis (>24 hours) does not decrease the risk of postoperative infection but it does increase the risk of acquiring resistant bacteria and adverse drug reactions, including CDI.
10. Facts All Clinicians Should Know About Patients with HIV Infection
The 2 most common laboratory abnormalities routinely associated with antiretroviral therapy for HIV infection are unconjugated hyperbilirubinemia associated with atazanavir and an elevated mean corpuscular volume (MCV) associated with zidovudine (and, to a lesser extent, stavudine). Immune reconstitution inflammatory syndrome (IRIS) is a condition seen in patients with advanced acquired immune deficiency syndrome (AIDS) who have recently started antiretroviral therapy. As the immune system begins to recover, it may respond to a previously acquired opportunistic infection with an overwhelming inflammatory response that paradoxically makes the symptoms of infection worse. IRIS is associated with a pathological inflammatory response that can have substantial morbidity and mortality.24 For this reason, when considering whether to start or stop continuous or highly active antiretroviral therapy (also known as HAART), an infectious diseases consult is recommended. Pneumocystis jiroveci (PCP) remains a cause of pneumonia in patients with advanced AIDS' though in the era of HAART, its presentation may be more subtle. Finally, the principle of parsimony (Occam's razor) often does not hold in the diagnosis of opportunistic infections in patients with advanced AIDS, as these patients can often present with multiple infections simultaneously.25, 26
Conclusion
Infectious diseases are commonly encountered by physicians who care for hospitalized patients. Early recognition, evaluation, and appropriate treatment and/or referral to an infectious diseases specialist are necessary to moderate the significant morbidity and mortality that are often associated with infectious diseases.
- ,.Observations on spiraling empiricism: its causes, allure, and perils, with particular reference to antibiotic therapy.Am J Med.1989;87(2):201–206.
- ,,, et al.Guidelines for the treatment of candidiasis.Clin Infect Dis.2004;38:161–189.
- ,,, et al.Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America.Circulation.2005;111:e394–e434.
- ,.Management of methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46:S386–S393.
- ,,, et al.Therapeutic approaches in patients with candidemia. Evaluation in a multicenter, prospective, observational study.Arch Intern Med.1995;155(22):2429–2435.
- ,,, et al.Nosocomial candidemia in non‐neutropenic patients at an Italian tertiary care hospital.Eur J Clin Microbiol Infect Dis.2000;19(8):602–607.
- .Candidemia in adults. In: Marr KA, ed.UpToDate.Waltham, MA:UpToDate, Inc.;2008.
- ,,, et al.Infectious Diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults.Clin Infect Dis.2005;40(5):643–654.
- .Asymptomatic bacteriuria: when to screen and when to treat.Infect Dis Clin North Am.2003;17(2):367–394.
- .Management of patients with a history of allergy to beta‐lactam antibiotics.Am J Med.2008;121(7):572–576.
- ,,.Practical aspects of choosing an antibiotic for patients with a reported allergy to an antibiotic.Clin Infect Dis.2002;35(1):26–31.
- ,,, et al.Adverse effects of orally administered ampicillin.J Pediatr.1973;83:106–108.
- ,,, et al.Brief communication: tolerability of meropenem in patients with IgE‐mediated hypersensitivity to penicillins.Ann Intern Med.2007;146(4):266–269.
- ,,.The rational clinical examination. Is this patient allergic to penicillin? An evidence‐based analysis of the likelihood of penicillin allergy.JAMA.2001;285(19):2498–2505.
- ,,.Leukocytosis in a tertiary care hospital with particular attention to the role of infection caused by Clostridium difficile.Clin Infect Dis.2002;34:1585–1592.
- ,,,.Leukocytosis as a harbinger and surrogate marker of Clostridium difficile infection in hospitalized patients with diarrhea.Am J Gastroenterol.2000;95:3137–3141.
- ,,,.Clostridium difficile infection in patients with unexplained leukocytosis.Am J Med.2003;115:543–546.
- ,.The challenges posed by reemerging Clostridium difficile infection.Clin Infect Dis.2007;45(2):222–227.
- ,,.Treatment of Clostridium difficile infection.Clin Infect Dis.2008;46(suppl 1):S32–S42.
- ,,,,.Diagnostic accuracy of routine postoperative body temperature measurements.Clin Infect Dis.2005;40:1404–1410.
- .Should we measure body temperature for patients who have recently undergone surgery?Clin Infect Dis.2005;40(10):1411–1412.
- ,,,.Evidence for the noninfectious etiology of early postoperative fever.Infect Control.1985;6:273–277.
- .Evaluating postoperative fever: a focused approach.Cleve Clin J Med.2006;73(suppl 1):S62–S66.
- ,,.Immune reconstitution inflammatory syndrome: more answers, more questions.J Antimicrob Chemother.2006;57(2):167–170.
- ,,,,.Clinical problem‐solving. Occam's razor versus Saint's Triad.N Engl J Med.2004;350(6):599–603.
- ,.William of Occam and Occam's razor.Ann Intern Med.2002;136(8):634–635.
- ,.Observations on spiraling empiricism: its causes, allure, and perils, with particular reference to antibiotic therapy.Am J Med.1989;87(2):201–206.
- ,,, et al.Guidelines for the treatment of candidiasis.Clin Infect Dis.2004;38:161–189.
- ,,, et al.Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America.Circulation.2005;111:e394–e434.
- ,.Management of methicillin‐resistant Staphylococcus aureus bacteremia.Clin Infect Dis.2008;46:S386–S393.
- ,,, et al.Therapeutic approaches in patients with candidemia. Evaluation in a multicenter, prospective, observational study.Arch Intern Med.1995;155(22):2429–2435.
- ,,, et al.Nosocomial candidemia in non‐neutropenic patients at an Italian tertiary care hospital.Eur J Clin Microbiol Infect Dis.2000;19(8):602–607.
- .Candidemia in adults. In: Marr KA, ed.UpToDate.Waltham, MA:UpToDate, Inc.;2008.
- ,,, et al.Infectious Diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults.Clin Infect Dis.2005;40(5):643–654.
- .Asymptomatic bacteriuria: when to screen and when to treat.Infect Dis Clin North Am.2003;17(2):367–394.
- .Management of patients with a history of allergy to beta‐lactam antibiotics.Am J Med.2008;121(7):572–576.
- ,,.Practical aspects of choosing an antibiotic for patients with a reported allergy to an antibiotic.Clin Infect Dis.2002;35(1):26–31.
- ,,, et al.Adverse effects of orally administered ampicillin.J Pediatr.1973;83:106–108.
- ,,, et al.Brief communication: tolerability of meropenem in patients with IgE‐mediated hypersensitivity to penicillins.Ann Intern Med.2007;146(4):266–269.
- ,,.The rational clinical examination. Is this patient allergic to penicillin? An evidence‐based analysis of the likelihood of penicillin allergy.JAMA.2001;285(19):2498–2505.
- ,,.Leukocytosis in a tertiary care hospital with particular attention to the role of infection caused by Clostridium difficile.Clin Infect Dis.2002;34:1585–1592.
- ,,,.Leukocytosis as a harbinger and surrogate marker of Clostridium difficile infection in hospitalized patients with diarrhea.Am J Gastroenterol.2000;95:3137–3141.
- ,,,.Clostridium difficile infection in patients with unexplained leukocytosis.Am J Med.2003;115:543–546.
- ,.The challenges posed by reemerging Clostridium difficile infection.Clin Infect Dis.2007;45(2):222–227.
- ,,.Treatment of Clostridium difficile infection.Clin Infect Dis.2008;46(suppl 1):S32–S42.
- ,,,,.Diagnostic accuracy of routine postoperative body temperature measurements.Clin Infect Dis.2005;40:1404–1410.
- .Should we measure body temperature for patients who have recently undergone surgery?Clin Infect Dis.2005;40(10):1411–1412.
- ,,,.Evidence for the noninfectious etiology of early postoperative fever.Infect Control.1985;6:273–277.
- .Evaluating postoperative fever: a focused approach.Cleve Clin J Med.2006;73(suppl 1):S62–S66.
- ,,.Immune reconstitution inflammatory syndrome: more answers, more questions.J Antimicrob Chemother.2006;57(2):167–170.
- ,,,,.Clinical problem‐solving. Occam's razor versus Saint's Triad.N Engl J Med.2004;350(6):599–603.
- ,.William of Occam and Occam's razor.Ann Intern Med.2002;136(8):634–635.
Hospitalists and Intensivists
A looming gap in the supply of intensivists prompted the American College of Chest Physicians (ACCP), the American Thoracic Society (ATS), and the Society of Critical Care Medicine (SCCM) to publish a report in 2000 by the Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). This study predicted that beginning in 2007 a shortfall would become apparent and steadily increase to 22% by 2020 and to 35% by 2030. Subsequent reports have reiterated those projections, including a report to Congress in 2006 by the U.S. Department of Health and Human Services/Health Resources and Services Administration.14
The concern regarding the shortage of intensivists has been increased by the growing evidence that supports improved critical care outcomesespecially decreased intensive care unit (ICU) and hospital mortalitywith intensivist staffing of ICUs.5, 6 Based on this data and on recommendations from the Society of Critical Care Medicine, the Leapfrog Group made onsite, high‐intensity ICU staffing with intensivists 1 of their 4 leaps.7 A paper by Pronovost et al.8 published in 2001, however, noted that in order for all ICUs in the United States to meet the Leapfrog ICU Physician Staffing (IPS) standard, the number of intensivists would need to increase by a factor of 2.6. Interestingly, a retrospective study published in the Annals of Internal Medicine in June of 2008 by Levy et al.9 suggested that mortality rates may actually be higher in intensivist‐staffed ICUs. An accompanying editorial raised concerns about limitations of the study design, but endorsed Levy's recommendation that more carefully designed, prospective studies were needed; (ie, we still are not certain as to optimal physician staffing for the care of patients requiring the sophisticated treatment available only in an ICU.)10
The health policy challenge, however, remains clear: while there is basic consensus that care of critically ill patients by intensivists improves outcomes, the reality is that the shortage of intensivists in the United States as predicted by the COMPACCS report will only increase, leading some to refer to this as a healthcare crisis. Two major task forces attempted to address this situation, resulting in the publication of the 2004 Framing Options for Critical Care in the United States (FOCCUS) report, The Critical Care Medicine Crisis: A Call for Federal ActionA White Paper from the Critical Care Professional Societies; and the 2007 Prioritizing the Organization and Management of Intensive Care Services in the Unites States (PrOMIS) Conference Report.11, 12 Both reports made specific recommendations including, for example, development of uniform standards for accreditation of institutional critical care capacity, identification and endorsement of core competencies in critical care, investment in health services research, the use of uniform protocols for ICU care, leverage of information technology to promote standardization and improve efficiency, and the development of incentives to attract healthcare professionals to critical care medicine.
A Possible Solution: The Role of Hospitalists
Multiple important efforts are already underway to increase the competency of professionals providing critical care services including the Society of Critical Care's Fundamentals in Critical Care Support (FCCS) program. Additionally, physician assistants and nurse practitioners are playing an increasingly important role as members of critical care services. As another component of this collaborative effort, the PrOMIS Report noted the potential impact of hospitalists in addressing this crisis.
As early as 1999, surveys revealed that as many as 35% of hospitalists were providing critical care services.13 According to the 2005/2006 Society of Hospital Medicine (SHM) National Survey, that number has increased to 75% with a low of 66% in the eastern United States and a high of 84% in the western United States. In community hospitals, 87% of hospitalists care for patients in the ICU, and 30% provide critical care services in academic medical centers.13 While there is some research14, 15 and many anecdotal reports that suggest hospitalists perform well in the ICU, there is, unfortunately, little data addressing outcomes for patients cared for by hospitalists. The results from a prospective, severity‐adjusted study from the Emory University Section of Hospital Medicine and the Division of Pulmonary/Critical Care Medicine examining outcomes for critical care patients cared for by hospitalists with criteria for Pulmonary/Intensivist consults vs. patients cared for by the Pulmonary/Critical Care Medical ICU Service await peer‐review publication.
Despite the lack of outcome data regarding adult hospitalists, it is clear that by default they are already providing a significant proportion of critical care services across the healthcare system, including in tertiary care centers. The two primary models of care include: (1) hospitalists serving as the primary provider without critical care consultant services and (2) comanagement of patients where intensivists and hospitalists collaborate. These collaborative models involve hospitalists actively co‐managing critical care patients along with intensivists or hospitalists managing less critically ill patients with intensivist consultation when indicated. In hospitals lacking intensivists, hospitalists often manage critically ill patients either with intensivist phone consultation, or with the intent to stabilize and transfer. Electronic ICUs are another expanding model of care that provide intensivist support to hospitalists and other primary care providersdecreasing ICU length of stay and severity‐adjusted ICU mortality.16 There are now 40 electronic ICU programs in the United States, and that number continues to grow.
In 2003, there were approximately 10,000 hospitalists in the United States,17 and recent data from an American Hospital Association survey indicates that the number has grown to about 28,000 in 2009. Recent research also documents that hospitalists are soon likely to care for the majority of elderly hospitalized patients in America.18 Aware that the number of intensivists is unlikely to change significantly over the next 25 years the question is no longer if hospitalists should be in the ICU; rather, the question is how to assure quality and improved clinical outcomes through enhanced collaboration between Hospital Medicine and Critical Care Medicine.
Recommendations
There are 3 steps that should be taken urgently to meet this challenge:
-
Per the recommendation of the FOCCUS Report and the PrOMIS Conference Report, uniform protocols for intensive care treatmentmany of which already exist but are not used consistentlyshould be identified and implemented across all ICUs regardless of the level or certification of the provider.
-
Also per the PrOMIS Report, a process for certification of physicians providing critical care services should be established by the appropriate governing bodies, including the Society for Critical Care Medicine, the Society of Hospital Medicine, and the American Thoracic Society, among others. While the PrOMIS Report called for cross‐training of hospital‐based providers to provide intensive care services in lower tier hospitals, a more realistic recommendation given current involvement of hospitalists in the provision of critical care services in secondary and tertiary centers is a competency‐assurance process that includes hospitalists practicing at all levels. This would not be equivalent to board certification, but would be based on a rigorous, comprehensive education and skills training process leading to recognition that would distinguish the recipient as having competencies beyond those obtained in internal medicine residency training. Models for certification could include 4‐month onsite training or a distance learning curriculum with regular blocks of onsite training. Another strategy might be for appropriate governing bodies to establish basic criteria for competency that would then be provided by individual institutions. Emory University, for example, has developed a pilot program incorporating significant components from the European Society for Critical Care Medicine's Syllabus for Competency Based Training in Intensive Care Medicine in Europe.19 Other institutions are also exploring the creation of certification/competency programs. Minimally, and prior to any decision about establishing formal criteria, institutions could identify designated hospitalists within groups who have particular interest and ability in the critical care setting. These providers, based on models already in place at sites across the United States, could, as an example, be required to spend a minimum of 50% of their clinical time in the ICU and to complete 10 to 20 hours of critical care continuing medical education (CME) per year. One strategy to address this issue and develop clear consensus and guidelines would be to convene the often discussed PrOMIS II working group.
-
Per both the FOCCUS Report and the PrOMIS Report as well as a number of other publications,19 health services research in ICU care should be identified, funded, and implemented. A major focus of this effort should be the evaluation of clinical outcomes for ICU patients cared for by hospitalists. This research is needed for at least 2 reasons:
-
As noted, there is little research that has assessed hospitalists' impact on outcomes of ICU patients. Hospitalists are already caring for patients in ICUs across the United States and given the research that has identified the outcomes benefit provided by intensivists, it is important to know objectively if hospitalists have similar levels of performance.
-
An increasing number of hospitals and healthcare systems are now committed to achieving the Leapfrog IPS standard‐a challenge for many because of the difficulty with recruiting intensivists. If new research reveals that hospitalists with board certification in Internal Medicine, and more specifically with additional competency training in critical care, also improve outcomes in the ICU then it may be possible for Leapfrog to revise the criteria for meeting the IPS standard.
Summary
As discussed in a number of publications,20 including an article from the Mayo Clinic in the April 2009 edition of Chest entitled, Physicians Staffing Models and Patient Safety in the ICU,21 along with an accompanying editorial, Should Intensive Care Medicine Itself Be on the Critical List,22 creative and realistic solutions are urgently needed to address the crisis in critical care in the U.S. Collaborative efforts between Critical Care Medicine and Hospital Medicine to meet this challenge benefit all involved:
-
Intensivists will continue to direct tertiary care units and/or co‐manage patients in tertiary and secondary care centers with Hospitalists.
-
Hospitalists will benefit by having the opportunity to secure critical care competency training and by having their appropriate role in the ICU defined.
-
All secondary and tertiary care institutions will have a realistic opportunity to meet Leapfrog IPS criteria and therefore benefit from the potential decreased length of stay (LOS), decreased mortality, and improved quality.
-
Patients benefit by receiving uniform, evidence‐based, protocol‐driven care.
There is now a need and an opportunity for ACCP, SCCM, ATS, and the American Association of Critical Care Nurses (ACCN), to expand the important work they have already begun through the Critical Care Workforce Partnership. The Partnership should join with the SHM to take the lead in supporting and promoting this collaborative relationship between intensivists and hospitalists: aware that in the final analysis, it is the patients we serve who will benefit the most.
- ,,, et al.Caring for the critically ill patient. Current and projected workforce requirements for care of the critically ill and patients with pulmonary disease: can we meet the requirements of an aging population?JAMA.2000;284:2762–2770.
- ,,.The critical care professional societies address the critical care crisis in the united states.Chest.2004;125:1512–1513.
- ,,, et al.The critical care crisis in the United States; a report from the profession.Chest.2004;125:1514–1517.
- U.S. Department of Health and Human Services, Health Resources and Services Administration. Report to Congress: the critical care workforce; a study of the supply and demand for critical care physicians. Senate Report 108–181. May2006.
- ,,, et al.Physician staffing patterns and clinical outcomes in critically ill patients.JAMA.2002:288:2151–2162.
- ,,.Do intensivists in ICU improve outcome? Best practice and research.Best Pract Res Clin Anaesthesiol.2005;19:125–135.
- The Leap Frog Group website. Available at: http://www.leapfroggroup.org. Accessed July2009.
- ,,.Impact of critical care physician workforce for intensive care unit physician staffing.Curr Opin Crit Care.2001;7:456–459.
- ,,,,,.Association between critical care physician management and patient mortality in the intensive care unit.Ann Intern Med.2008;148:801–810.
- ,.Are intensivists safe?Ann Intern Med.2008;148:877–878.
- ,,,,,.The critical care medicine crisis: a call for federal action; a white paper from the critical care professional societies.Chest.2004;125:1518–1521.
- ,,, et al.Prioritizing the organization and management of intensive care services in the Unites States: the PrOMIS conference.Crit Care Med.2007;35:1103–1111.
- ,,,.Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians.Ann Intern Med.1999;130:343–349.
- ,,.Improved survival with hospitalists in a pediatric intensive care unit.Crit Care Med.2003;31:847–852.
- .Integrating hospitalists into the pediatric intensive care unit.Crit Care Med.2003;32:813–816.
- ,,, et al.Prognostic outcomes after the initiation of an electronic telemedicine intensive care unit (eICU) in a rural health system.SD Med2006;59(9):391–393.
- ,,,.The status of hospital medicine groups in the United States.J Hosp Med.2006;1:75–80.
- ,,,.Growth in the care of older patients by hospitalists in the United States.N Engl J Med.2009;360:1102–1112.
- CoBaTrICE Syllabus (Competency‐Based Training in Intensive Care Medicine in Europe), Version 1.0.Brussels:European Society of Intensive Care Medicine;2006.
- .Caring for the critically ill. patient challenges and opportunities.JAMA.2007;298(4):456–458.
- ,.Physician staffing models and patient safety in the ICU.Chest.2009;135:1038–1044.
- ,.Should intensive care medicine itself be on the critical list.Chest.2009;135:892–894.
A looming gap in the supply of intensivists prompted the American College of Chest Physicians (ACCP), the American Thoracic Society (ATS), and the Society of Critical Care Medicine (SCCM) to publish a report in 2000 by the Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). This study predicted that beginning in 2007 a shortfall would become apparent and steadily increase to 22% by 2020 and to 35% by 2030. Subsequent reports have reiterated those projections, including a report to Congress in 2006 by the U.S. Department of Health and Human Services/Health Resources and Services Administration.14
The concern regarding the shortage of intensivists has been increased by the growing evidence that supports improved critical care outcomesespecially decreased intensive care unit (ICU) and hospital mortalitywith intensivist staffing of ICUs.5, 6 Based on this data and on recommendations from the Society of Critical Care Medicine, the Leapfrog Group made onsite, high‐intensity ICU staffing with intensivists 1 of their 4 leaps.7 A paper by Pronovost et al.8 published in 2001, however, noted that in order for all ICUs in the United States to meet the Leapfrog ICU Physician Staffing (IPS) standard, the number of intensivists would need to increase by a factor of 2.6. Interestingly, a retrospective study published in the Annals of Internal Medicine in June of 2008 by Levy et al.9 suggested that mortality rates may actually be higher in intensivist‐staffed ICUs. An accompanying editorial raised concerns about limitations of the study design, but endorsed Levy's recommendation that more carefully designed, prospective studies were needed; (ie, we still are not certain as to optimal physician staffing for the care of patients requiring the sophisticated treatment available only in an ICU.)10
The health policy challenge, however, remains clear: while there is basic consensus that care of critically ill patients by intensivists improves outcomes, the reality is that the shortage of intensivists in the United States as predicted by the COMPACCS report will only increase, leading some to refer to this as a healthcare crisis. Two major task forces attempted to address this situation, resulting in the publication of the 2004 Framing Options for Critical Care in the United States (FOCCUS) report, The Critical Care Medicine Crisis: A Call for Federal ActionA White Paper from the Critical Care Professional Societies; and the 2007 Prioritizing the Organization and Management of Intensive Care Services in the Unites States (PrOMIS) Conference Report.11, 12 Both reports made specific recommendations including, for example, development of uniform standards for accreditation of institutional critical care capacity, identification and endorsement of core competencies in critical care, investment in health services research, the use of uniform protocols for ICU care, leverage of information technology to promote standardization and improve efficiency, and the development of incentives to attract healthcare professionals to critical care medicine.
A Possible Solution: The Role of Hospitalists
Multiple important efforts are already underway to increase the competency of professionals providing critical care services including the Society of Critical Care's Fundamentals in Critical Care Support (FCCS) program. Additionally, physician assistants and nurse practitioners are playing an increasingly important role as members of critical care services. As another component of this collaborative effort, the PrOMIS Report noted the potential impact of hospitalists in addressing this crisis.
As early as 1999, surveys revealed that as many as 35% of hospitalists were providing critical care services.13 According to the 2005/2006 Society of Hospital Medicine (SHM) National Survey, that number has increased to 75% with a low of 66% in the eastern United States and a high of 84% in the western United States. In community hospitals, 87% of hospitalists care for patients in the ICU, and 30% provide critical care services in academic medical centers.13 While there is some research14, 15 and many anecdotal reports that suggest hospitalists perform well in the ICU, there is, unfortunately, little data addressing outcomes for patients cared for by hospitalists. The results from a prospective, severity‐adjusted study from the Emory University Section of Hospital Medicine and the Division of Pulmonary/Critical Care Medicine examining outcomes for critical care patients cared for by hospitalists with criteria for Pulmonary/Intensivist consults vs. patients cared for by the Pulmonary/Critical Care Medical ICU Service await peer‐review publication.
Despite the lack of outcome data regarding adult hospitalists, it is clear that by default they are already providing a significant proportion of critical care services across the healthcare system, including in tertiary care centers. The two primary models of care include: (1) hospitalists serving as the primary provider without critical care consultant services and (2) comanagement of patients where intensivists and hospitalists collaborate. These collaborative models involve hospitalists actively co‐managing critical care patients along with intensivists or hospitalists managing less critically ill patients with intensivist consultation when indicated. In hospitals lacking intensivists, hospitalists often manage critically ill patients either with intensivist phone consultation, or with the intent to stabilize and transfer. Electronic ICUs are another expanding model of care that provide intensivist support to hospitalists and other primary care providersdecreasing ICU length of stay and severity‐adjusted ICU mortality.16 There are now 40 electronic ICU programs in the United States, and that number continues to grow.
In 2003, there were approximately 10,000 hospitalists in the United States,17 and recent data from an American Hospital Association survey indicates that the number has grown to about 28,000 in 2009. Recent research also documents that hospitalists are soon likely to care for the majority of elderly hospitalized patients in America.18 Aware that the number of intensivists is unlikely to change significantly over the next 25 years the question is no longer if hospitalists should be in the ICU; rather, the question is how to assure quality and improved clinical outcomes through enhanced collaboration between Hospital Medicine and Critical Care Medicine.
Recommendations
There are 3 steps that should be taken urgently to meet this challenge:
-
Per the recommendation of the FOCCUS Report and the PrOMIS Conference Report, uniform protocols for intensive care treatmentmany of which already exist but are not used consistentlyshould be identified and implemented across all ICUs regardless of the level or certification of the provider.
-
Also per the PrOMIS Report, a process for certification of physicians providing critical care services should be established by the appropriate governing bodies, including the Society for Critical Care Medicine, the Society of Hospital Medicine, and the American Thoracic Society, among others. While the PrOMIS Report called for cross‐training of hospital‐based providers to provide intensive care services in lower tier hospitals, a more realistic recommendation given current involvement of hospitalists in the provision of critical care services in secondary and tertiary centers is a competency‐assurance process that includes hospitalists practicing at all levels. This would not be equivalent to board certification, but would be based on a rigorous, comprehensive education and skills training process leading to recognition that would distinguish the recipient as having competencies beyond those obtained in internal medicine residency training. Models for certification could include 4‐month onsite training or a distance learning curriculum with regular blocks of onsite training. Another strategy might be for appropriate governing bodies to establish basic criteria for competency that would then be provided by individual institutions. Emory University, for example, has developed a pilot program incorporating significant components from the European Society for Critical Care Medicine's Syllabus for Competency Based Training in Intensive Care Medicine in Europe.19 Other institutions are also exploring the creation of certification/competency programs. Minimally, and prior to any decision about establishing formal criteria, institutions could identify designated hospitalists within groups who have particular interest and ability in the critical care setting. These providers, based on models already in place at sites across the United States, could, as an example, be required to spend a minimum of 50% of their clinical time in the ICU and to complete 10 to 20 hours of critical care continuing medical education (CME) per year. One strategy to address this issue and develop clear consensus and guidelines would be to convene the often discussed PrOMIS II working group.
-
Per both the FOCCUS Report and the PrOMIS Report as well as a number of other publications,19 health services research in ICU care should be identified, funded, and implemented. A major focus of this effort should be the evaluation of clinical outcomes for ICU patients cared for by hospitalists. This research is needed for at least 2 reasons:
-
As noted, there is little research that has assessed hospitalists' impact on outcomes of ICU patients. Hospitalists are already caring for patients in ICUs across the United States and given the research that has identified the outcomes benefit provided by intensivists, it is important to know objectively if hospitalists have similar levels of performance.
-
An increasing number of hospitals and healthcare systems are now committed to achieving the Leapfrog IPS standard‐a challenge for many because of the difficulty with recruiting intensivists. If new research reveals that hospitalists with board certification in Internal Medicine, and more specifically with additional competency training in critical care, also improve outcomes in the ICU then it may be possible for Leapfrog to revise the criteria for meeting the IPS standard.
Summary
As discussed in a number of publications,20 including an article from the Mayo Clinic in the April 2009 edition of Chest entitled, Physicians Staffing Models and Patient Safety in the ICU,21 along with an accompanying editorial, Should Intensive Care Medicine Itself Be on the Critical List,22 creative and realistic solutions are urgently needed to address the crisis in critical care in the U.S. Collaborative efforts between Critical Care Medicine and Hospital Medicine to meet this challenge benefit all involved:
-
Intensivists will continue to direct tertiary care units and/or co‐manage patients in tertiary and secondary care centers with Hospitalists.
-
Hospitalists will benefit by having the opportunity to secure critical care competency training and by having their appropriate role in the ICU defined.
-
All secondary and tertiary care institutions will have a realistic opportunity to meet Leapfrog IPS criteria and therefore benefit from the potential decreased length of stay (LOS), decreased mortality, and improved quality.
-
Patients benefit by receiving uniform, evidence‐based, protocol‐driven care.
There is now a need and an opportunity for ACCP, SCCM, ATS, and the American Association of Critical Care Nurses (ACCN), to expand the important work they have already begun through the Critical Care Workforce Partnership. The Partnership should join with the SHM to take the lead in supporting and promoting this collaborative relationship between intensivists and hospitalists: aware that in the final analysis, it is the patients we serve who will benefit the most.
A looming gap in the supply of intensivists prompted the American College of Chest Physicians (ACCP), the American Thoracic Society (ATS), and the Society of Critical Care Medicine (SCCM) to publish a report in 2000 by the Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). This study predicted that beginning in 2007 a shortfall would become apparent and steadily increase to 22% by 2020 and to 35% by 2030. Subsequent reports have reiterated those projections, including a report to Congress in 2006 by the U.S. Department of Health and Human Services/Health Resources and Services Administration.14
The concern regarding the shortage of intensivists has been increased by the growing evidence that supports improved critical care outcomesespecially decreased intensive care unit (ICU) and hospital mortalitywith intensivist staffing of ICUs.5, 6 Based on this data and on recommendations from the Society of Critical Care Medicine, the Leapfrog Group made onsite, high‐intensity ICU staffing with intensivists 1 of their 4 leaps.7 A paper by Pronovost et al.8 published in 2001, however, noted that in order for all ICUs in the United States to meet the Leapfrog ICU Physician Staffing (IPS) standard, the number of intensivists would need to increase by a factor of 2.6. Interestingly, a retrospective study published in the Annals of Internal Medicine in June of 2008 by Levy et al.9 suggested that mortality rates may actually be higher in intensivist‐staffed ICUs. An accompanying editorial raised concerns about limitations of the study design, but endorsed Levy's recommendation that more carefully designed, prospective studies were needed; (ie, we still are not certain as to optimal physician staffing for the care of patients requiring the sophisticated treatment available only in an ICU.)10
The health policy challenge, however, remains clear: while there is basic consensus that care of critically ill patients by intensivists improves outcomes, the reality is that the shortage of intensivists in the United States as predicted by the COMPACCS report will only increase, leading some to refer to this as a healthcare crisis. Two major task forces attempted to address this situation, resulting in the publication of the 2004 Framing Options for Critical Care in the United States (FOCCUS) report, The Critical Care Medicine Crisis: A Call for Federal ActionA White Paper from the Critical Care Professional Societies; and the 2007 Prioritizing the Organization and Management of Intensive Care Services in the Unites States (PrOMIS) Conference Report.11, 12 Both reports made specific recommendations including, for example, development of uniform standards for accreditation of institutional critical care capacity, identification and endorsement of core competencies in critical care, investment in health services research, the use of uniform protocols for ICU care, leverage of information technology to promote standardization and improve efficiency, and the development of incentives to attract healthcare professionals to critical care medicine.
A Possible Solution: The Role of Hospitalists
Multiple important efforts are already underway to increase the competency of professionals providing critical care services including the Society of Critical Care's Fundamentals in Critical Care Support (FCCS) program. Additionally, physician assistants and nurse practitioners are playing an increasingly important role as members of critical care services. As another component of this collaborative effort, the PrOMIS Report noted the potential impact of hospitalists in addressing this crisis.
As early as 1999, surveys revealed that as many as 35% of hospitalists were providing critical care services.13 According to the 2005/2006 Society of Hospital Medicine (SHM) National Survey, that number has increased to 75% with a low of 66% in the eastern United States and a high of 84% in the western United States. In community hospitals, 87% of hospitalists care for patients in the ICU, and 30% provide critical care services in academic medical centers.13 While there is some research14, 15 and many anecdotal reports that suggest hospitalists perform well in the ICU, there is, unfortunately, little data addressing outcomes for patients cared for by hospitalists. The results from a prospective, severity‐adjusted study from the Emory University Section of Hospital Medicine and the Division of Pulmonary/Critical Care Medicine examining outcomes for critical care patients cared for by hospitalists with criteria for Pulmonary/Intensivist consults vs. patients cared for by the Pulmonary/Critical Care Medical ICU Service await peer‐review publication.
Despite the lack of outcome data regarding adult hospitalists, it is clear that by default they are already providing a significant proportion of critical care services across the healthcare system, including in tertiary care centers. The two primary models of care include: (1) hospitalists serving as the primary provider without critical care consultant services and (2) comanagement of patients where intensivists and hospitalists collaborate. These collaborative models involve hospitalists actively co‐managing critical care patients along with intensivists or hospitalists managing less critically ill patients with intensivist consultation when indicated. In hospitals lacking intensivists, hospitalists often manage critically ill patients either with intensivist phone consultation, or with the intent to stabilize and transfer. Electronic ICUs are another expanding model of care that provide intensivist support to hospitalists and other primary care providersdecreasing ICU length of stay and severity‐adjusted ICU mortality.16 There are now 40 electronic ICU programs in the United States, and that number continues to grow.
In 2003, there were approximately 10,000 hospitalists in the United States,17 and recent data from an American Hospital Association survey indicates that the number has grown to about 28,000 in 2009. Recent research also documents that hospitalists are soon likely to care for the majority of elderly hospitalized patients in America.18 Aware that the number of intensivists is unlikely to change significantly over the next 25 years the question is no longer if hospitalists should be in the ICU; rather, the question is how to assure quality and improved clinical outcomes through enhanced collaboration between Hospital Medicine and Critical Care Medicine.
Recommendations
There are 3 steps that should be taken urgently to meet this challenge:
-
Per the recommendation of the FOCCUS Report and the PrOMIS Conference Report, uniform protocols for intensive care treatmentmany of which already exist but are not used consistentlyshould be identified and implemented across all ICUs regardless of the level or certification of the provider.
-
Also per the PrOMIS Report, a process for certification of physicians providing critical care services should be established by the appropriate governing bodies, including the Society for Critical Care Medicine, the Society of Hospital Medicine, and the American Thoracic Society, among others. While the PrOMIS Report called for cross‐training of hospital‐based providers to provide intensive care services in lower tier hospitals, a more realistic recommendation given current involvement of hospitalists in the provision of critical care services in secondary and tertiary centers is a competency‐assurance process that includes hospitalists practicing at all levels. This would not be equivalent to board certification, but would be based on a rigorous, comprehensive education and skills training process leading to recognition that would distinguish the recipient as having competencies beyond those obtained in internal medicine residency training. Models for certification could include 4‐month onsite training or a distance learning curriculum with regular blocks of onsite training. Another strategy might be for appropriate governing bodies to establish basic criteria for competency that would then be provided by individual institutions. Emory University, for example, has developed a pilot program incorporating significant components from the European Society for Critical Care Medicine's Syllabus for Competency Based Training in Intensive Care Medicine in Europe.19 Other institutions are also exploring the creation of certification/competency programs. Minimally, and prior to any decision about establishing formal criteria, institutions could identify designated hospitalists within groups who have particular interest and ability in the critical care setting. These providers, based on models already in place at sites across the United States, could, as an example, be required to spend a minimum of 50% of their clinical time in the ICU and to complete 10 to 20 hours of critical care continuing medical education (CME) per year. One strategy to address this issue and develop clear consensus and guidelines would be to convene the often discussed PrOMIS II working group.
-
Per both the FOCCUS Report and the PrOMIS Report as well as a number of other publications,19 health services research in ICU care should be identified, funded, and implemented. A major focus of this effort should be the evaluation of clinical outcomes for ICU patients cared for by hospitalists. This research is needed for at least 2 reasons:
-
As noted, there is little research that has assessed hospitalists' impact on outcomes of ICU patients. Hospitalists are already caring for patients in ICUs across the United States and given the research that has identified the outcomes benefit provided by intensivists, it is important to know objectively if hospitalists have similar levels of performance.
-
An increasing number of hospitals and healthcare systems are now committed to achieving the Leapfrog IPS standard‐a challenge for many because of the difficulty with recruiting intensivists. If new research reveals that hospitalists with board certification in Internal Medicine, and more specifically with additional competency training in critical care, also improve outcomes in the ICU then it may be possible for Leapfrog to revise the criteria for meeting the IPS standard.
Summary
As discussed in a number of publications,20 including an article from the Mayo Clinic in the April 2009 edition of Chest entitled, Physicians Staffing Models and Patient Safety in the ICU,21 along with an accompanying editorial, Should Intensive Care Medicine Itself Be on the Critical List,22 creative and realistic solutions are urgently needed to address the crisis in critical care in the U.S. Collaborative efforts between Critical Care Medicine and Hospital Medicine to meet this challenge benefit all involved:
-
Intensivists will continue to direct tertiary care units and/or co‐manage patients in tertiary and secondary care centers with Hospitalists.
-
Hospitalists will benefit by having the opportunity to secure critical care competency training and by having their appropriate role in the ICU defined.
-
All secondary and tertiary care institutions will have a realistic opportunity to meet Leapfrog IPS criteria and therefore benefit from the potential decreased length of stay (LOS), decreased mortality, and improved quality.
-
Patients benefit by receiving uniform, evidence‐based, protocol‐driven care.
There is now a need and an opportunity for ACCP, SCCM, ATS, and the American Association of Critical Care Nurses (ACCN), to expand the important work they have already begun through the Critical Care Workforce Partnership. The Partnership should join with the SHM to take the lead in supporting and promoting this collaborative relationship between intensivists and hospitalists: aware that in the final analysis, it is the patients we serve who will benefit the most.
- ,,, et al.Caring for the critically ill patient. Current and projected workforce requirements for care of the critically ill and patients with pulmonary disease: can we meet the requirements of an aging population?JAMA.2000;284:2762–2770.
- ,,.The critical care professional societies address the critical care crisis in the united states.Chest.2004;125:1512–1513.
- ,,, et al.The critical care crisis in the United States; a report from the profession.Chest.2004;125:1514–1517.
- U.S. Department of Health and Human Services, Health Resources and Services Administration. Report to Congress: the critical care workforce; a study of the supply and demand for critical care physicians. Senate Report 108–181. May2006.
- ,,, et al.Physician staffing patterns and clinical outcomes in critically ill patients.JAMA.2002:288:2151–2162.
- ,,.Do intensivists in ICU improve outcome? Best practice and research.Best Pract Res Clin Anaesthesiol.2005;19:125–135.
- The Leap Frog Group website. Available at: http://www.leapfroggroup.org. Accessed July2009.
- ,,.Impact of critical care physician workforce for intensive care unit physician staffing.Curr Opin Crit Care.2001;7:456–459.
- ,,,,,.Association between critical care physician management and patient mortality in the intensive care unit.Ann Intern Med.2008;148:801–810.
- ,.Are intensivists safe?Ann Intern Med.2008;148:877–878.
- ,,,,,.The critical care medicine crisis: a call for federal action; a white paper from the critical care professional societies.Chest.2004;125:1518–1521.
- ,,, et al.Prioritizing the organization and management of intensive care services in the Unites States: the PrOMIS conference.Crit Care Med.2007;35:1103–1111.
- ,,,.Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians.Ann Intern Med.1999;130:343–349.
- ,,.Improved survival with hospitalists in a pediatric intensive care unit.Crit Care Med.2003;31:847–852.
- .Integrating hospitalists into the pediatric intensive care unit.Crit Care Med.2003;32:813–816.
- ,,, et al.Prognostic outcomes after the initiation of an electronic telemedicine intensive care unit (eICU) in a rural health system.SD Med2006;59(9):391–393.
- ,,,.The status of hospital medicine groups in the United States.J Hosp Med.2006;1:75–80.
- ,,,.Growth in the care of older patients by hospitalists in the United States.N Engl J Med.2009;360:1102–1112.
- CoBaTrICE Syllabus (Competency‐Based Training in Intensive Care Medicine in Europe), Version 1.0.Brussels:European Society of Intensive Care Medicine;2006.
- .Caring for the critically ill. patient challenges and opportunities.JAMA.2007;298(4):456–458.
- ,.Physician staffing models and patient safety in the ICU.Chest.2009;135:1038–1044.
- ,.Should intensive care medicine itself be on the critical list.Chest.2009;135:892–894.
- ,,, et al.Caring for the critically ill patient. Current and projected workforce requirements for care of the critically ill and patients with pulmonary disease: can we meet the requirements of an aging population?JAMA.2000;284:2762–2770.
- ,,.The critical care professional societies address the critical care crisis in the united states.Chest.2004;125:1512–1513.
- ,,, et al.The critical care crisis in the United States; a report from the profession.Chest.2004;125:1514–1517.
- U.S. Department of Health and Human Services, Health Resources and Services Administration. Report to Congress: the critical care workforce; a study of the supply and demand for critical care physicians. Senate Report 108–181. May2006.
- ,,, et al.Physician staffing patterns and clinical outcomes in critically ill patients.JAMA.2002:288:2151–2162.
- ,,.Do intensivists in ICU improve outcome? Best practice and research.Best Pract Res Clin Anaesthesiol.2005;19:125–135.
- The Leap Frog Group website. Available at: http://www.leapfroggroup.org. Accessed July2009.
- ,,.Impact of critical care physician workforce for intensive care unit physician staffing.Curr Opin Crit Care.2001;7:456–459.
- ,,,,,.Association between critical care physician management and patient mortality in the intensive care unit.Ann Intern Med.2008;148:801–810.
- ,.Are intensivists safe?Ann Intern Med.2008;148:877–878.
- ,,,,,.The critical care medicine crisis: a call for federal action; a white paper from the critical care professional societies.Chest.2004;125:1518–1521.
- ,,, et al.Prioritizing the organization and management of intensive care services in the Unites States: the PrOMIS conference.Crit Care Med.2007;35:1103–1111.
- ,,,.Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians.Ann Intern Med.1999;130:343–349.
- ,,.Improved survival with hospitalists in a pediatric intensive care unit.Crit Care Med.2003;31:847–852.
- .Integrating hospitalists into the pediatric intensive care unit.Crit Care Med.2003;32:813–816.
- ,,, et al.Prognostic outcomes after the initiation of an electronic telemedicine intensive care unit (eICU) in a rural health system.SD Med2006;59(9):391–393.
- ,,,.The status of hospital medicine groups in the United States.J Hosp Med.2006;1:75–80.
- ,,,.Growth in the care of older patients by hospitalists in the United States.N Engl J Med.2009;360:1102–1112.
- CoBaTrICE Syllabus (Competency‐Based Training in Intensive Care Medicine in Europe), Version 1.0.Brussels:European Society of Intensive Care Medicine;2006.
- .Caring for the critically ill. patient challenges and opportunities.JAMA.2007;298(4):456–458.
- ,.Physician staffing models and patient safety in the ICU.Chest.2009;135:1038–1044.
- ,.Should intensive care medicine itself be on the critical list.Chest.2009;135:892–894.
Pretibial Myxedema
An 83‐year‐old female reported increased swelling of her legs over the past 2 years. She noted temperature intolerance, low energy, and constipation, but denied any hair loss or nail changes. On exam, she had marked bilateral lower extremity edema that was predominately nonpitting. Overlying the edema there were thickened, well‐defined plaques with a peau d'orange appearance surrounded by brown, thin plaques on the pretibial areas sparing the dorsum of the feet (Figure 1). A punch biopsy was obtained and demonstrated increased deposition of mucin throughout the dermis along with fragmentation and increased numbers of elastic fibers consistent with a diagnosis of pretibial myxedema. Measured thyrotropin level was elevated at 3.9 U/mL (normal, 0.3‐3.8 U/mL), consistent with hypothyroidism. This is a severe example of pretibial myxedema, or infiltrative dermopathy, which can occur, more commonly, in the setting of Graves' disease or, in rare circumstances, hypothyroidism.1 Myxedema results from the accumulation of hyaluronic acid and chondroitin sulfate in the dermis.2 Treatment is difficult and includes topical glucocorticoids under occlusion and, if indicated, thyroid corrective therapy.3

- .Pretibial myxedema.Dermatol Online J.7(1):18.
- ,,.Pretibial myxoedema: a manifestation of lymphoedema?Lancet.1993;341(8842):403–404.
- .Successful treatment of chronic skin diseases with clobetasol propionate and a hydrocolloid occlusive dressing.Acta Derm Venereol.1992;72(1):69–71.
An 83‐year‐old female reported increased swelling of her legs over the past 2 years. She noted temperature intolerance, low energy, and constipation, but denied any hair loss or nail changes. On exam, she had marked bilateral lower extremity edema that was predominately nonpitting. Overlying the edema there were thickened, well‐defined plaques with a peau d'orange appearance surrounded by brown, thin plaques on the pretibial areas sparing the dorsum of the feet (Figure 1). A punch biopsy was obtained and demonstrated increased deposition of mucin throughout the dermis along with fragmentation and increased numbers of elastic fibers consistent with a diagnosis of pretibial myxedema. Measured thyrotropin level was elevated at 3.9 U/mL (normal, 0.3‐3.8 U/mL), consistent with hypothyroidism. This is a severe example of pretibial myxedema, or infiltrative dermopathy, which can occur, more commonly, in the setting of Graves' disease or, in rare circumstances, hypothyroidism.1 Myxedema results from the accumulation of hyaluronic acid and chondroitin sulfate in the dermis.2 Treatment is difficult and includes topical glucocorticoids under occlusion and, if indicated, thyroid corrective therapy.3

An 83‐year‐old female reported increased swelling of her legs over the past 2 years. She noted temperature intolerance, low energy, and constipation, but denied any hair loss or nail changes. On exam, she had marked bilateral lower extremity edema that was predominately nonpitting. Overlying the edema there were thickened, well‐defined plaques with a peau d'orange appearance surrounded by brown, thin plaques on the pretibial areas sparing the dorsum of the feet (Figure 1). A punch biopsy was obtained and demonstrated increased deposition of mucin throughout the dermis along with fragmentation and increased numbers of elastic fibers consistent with a diagnosis of pretibial myxedema. Measured thyrotropin level was elevated at 3.9 U/mL (normal, 0.3‐3.8 U/mL), consistent with hypothyroidism. This is a severe example of pretibial myxedema, or infiltrative dermopathy, which can occur, more commonly, in the setting of Graves' disease or, in rare circumstances, hypothyroidism.1 Myxedema results from the accumulation of hyaluronic acid and chondroitin sulfate in the dermis.2 Treatment is difficult and includes topical glucocorticoids under occlusion and, if indicated, thyroid corrective therapy.3

- .Pretibial myxedema.Dermatol Online J.7(1):18.
- ,,.Pretibial myxoedema: a manifestation of lymphoedema?Lancet.1993;341(8842):403–404.
- .Successful treatment of chronic skin diseases with clobetasol propionate and a hydrocolloid occlusive dressing.Acta Derm Venereol.1992;72(1):69–71.
- .Pretibial myxedema.Dermatol Online J.7(1):18.
- ,,.Pretibial myxoedema: a manifestation of lymphoedema?Lancet.1993;341(8842):403–404.
- .Successful treatment of chronic skin diseases with clobetasol propionate and a hydrocolloid occlusive dressing.Acta Derm Venereol.1992;72(1):69–71.
Early Prediction of Septic Shock
Severe sepsis is responsible for significant morbidity and mortality. In the United States, approximately 750,000 cases occur each year with an estimated mortality of 30% to 50%.1 Early goal‐directed therapy has been shown to decrease mortality in patients with severe sepsis and septic shock.2, 3 As a result, efforts have been focused toward providing early and aggressive intervention once sepsis has been established. In many cases this has been accomplished through the implementation of a protocol with guidelines for fluid management, antibiotic and vasopressor administration, and other interventions.410 Prior studies have demonstrated that care of hospitalized patients before intensive care unit (ICU) admission is often suboptimal,1113 and have suggested that patients with clear indicators of acute deterioration may go unrecognized on the ward. We previously reported the effects of implementing a hospital‐wide protocol for the management of severe sepsis,14 finding that although there was a significant reduction in overall mortality there was no difference for patients who developed severe sepsis on the hospital ward. This finding also suggests that the initial care of patients with severe sepsis on hospital wards may differ in intensity compared to emergency departments and ICUs. Failure on the part of the clinician to recognize the harbingers of impending sepsis before the onset of organ dysfunction or hypotension may contribute to a delay in aggressive therapy.
Previous efforts at early recognition of sepsis have relied on diagnostic studies or specific biomarkers to screen at‐risk patients. These have included such studies as messenger RNA (mRNA) expression,15 C‐reactive protein,16 procalcitonin in newborns,17 immunocompetence measures in burn patients,18 protein C concentration in neutropenic patients,19 and several immune markers (eg, tumor necrosis factor‐alpha, interleukin [IL]‐1 beta, IL‐6, IL‐8, and IL‐10).20 However, these biomarkers have been studied only in specific patient populations, require suspicion on the part of the clinician and the measurement of diagnostic or laboratory values that would otherwise not have been obtained. The ideal tool for predicting the onset of sepsis would be applicable to a broad patient population, not require specific suspicion on the part of the clinician, and use only routinely obtained clinical measurements and laboratory values.
Prediction models and scoring systems that use routine hemodynamic and laboratory values for several endpoints related to sepsis and septic shock have been developed. Many such tools are used to define severity of illness and predict outcome, while others have been developed to predict such events as bacteremia in patients presenting with fever,21 the probability of infection in the critically ill,22 and end‐organ dysfunction in severe sepsis.23 Little work has been done to develop such a model capable of predicting the onset of sepsis,24 and there have been no attempts to deploy a model as a large‐scale screening tool.
Our objective was to develop a simple algorithm that can be used in an automated fashion to screen hospitalized patients for impending septic shock. Such a model would be derived from routine hemodynamic and laboratory values, and take advantage of a computerized medical record system for data collection.
Patients and Methods
Patient Enrollment and Data Collection
This study was conducted at Barnes‐Jewish Hospital, St. Louis, MO, a university‐affiliated, urban teaching hospital. The study was approved by the Washington University (St. Louis, MO) School of Medicine Human Studies Committee. Patients included in the study where those hospitalized during 2005, 2006, and 2007, and who had at least 1 International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD9) discharge diagnosis code for the medical/nonsurgical diagnoses listed in Appendix 1. From this pool of patients, septic shock patients were identified as those who were admitted to the hospital ward and later developed septic shock requiring transfer to an ICU for vasopressor support and hemodynamic monitoring. This was accomplished by using discharge ICD9 codes for acute infection matched to codes for acute organ dysfunction and the need for vasopressors within 24 hours of ICU transfer (Appendix 1). The patients used as controls were then all those remaining in the pool once the septic shock patients were identified and separated.
Case patients were excluded from the analysis if they were transferred to the ICU within 2 hours of hospital admission, as these patients are unlikely to have an adequate amount of pretransfer clinical data available for analysis. Both case and control patients were excluded if they lacked any value for basic, routine laboratory data (serum sodium, chloride, total bicarbonate, urea nitrogen, creatinine, glucose, white blood cell count, neutrophil count, hemoglobin, hematocrit, and platelet count) and certain vital signs (blood pressure, heart rate, temperature). Patient data from 2005 were used in the derivation of the prediction model, and 2006 and 2007 patient data were used to prospectively validate the model. Clinical variables used in the analysis were selected based on both ease of access from the electronic medical record and clinical relevance, and are shown in Table 1.
|
| Age (years) |
| Albumin (g/dL) |
| Arterial blood gas (pH, PaCO2, PaO2) |
| Anion gap |
| Bilirubin (mg/dL) |
| BP, systolic and diastolic (mm of Hg) |
| Blood urea nitrogen (mg/dL) |
| Chloride (mmol/L) |
| Creatinine (mg/dL) |
| Glucose (mg/dL) |
| Hemoglobin (g/dL) |
| International normalized ratio |
| Neutrophil count, absolute (1 103/L) |
| Platelet count (1 103/L) |
| Pulse (beats/minute) |
| Pulse pressure (mm of Hg) |
| Shock index (pulse divided by systolic BP) |
| Sodium (mmol/L) |
| Total bicarbonate (mmol/L) |
| Temperature (degrees Celsius) |
| White blood cell count (1 103/L) |
In performing the Recursive Partitioning And Regression Tree (RPART) analysis to generate a prediction model, data for case patients were extracted in a window from 24 hours to 2 hours before ICU admission. The data collection window excluded the 2 hours prior to ICU transfer in order to minimize the effect of acute hemodynamic or laboratory changes that may have prompted the transfer; the purpose of the model is to identify hemodynamic and laboratory patterns in the several hours before the onset of clinically evident shock, so data from a time during which impending shock was clinically apparent were excluded. For the control patients, data from the first 48 hours of their hospitalization were included in the analysis.
Statistical Analysis
RPART analysis was performed on the 2005 patient data set to generate a prediction algorithm. This method of analysis results in a classification tree that contains a series of binary splits designed to separate patients into mutually exclusive subgroups.25 Each split in the tree is selected based on its ability to produce a partition with the greatest purity. Initially, a large tree that contains splits for all input variables is generated. This initial tree is generally too large to be useful as the final subgroups are too small to make sensible statistical inference.25 A pruning process is then applied to the initial tree with the goal of finding the subtree that is most predictive of the outcome of interest. The analysis was done using the RPART package of the R statistical analysis program, version 2.7.0 (R: A Language and Environment for Statistical Computing, R Development Core Team, Foundation for Statistical Computing, Vienna, Austria). The resulting classification tree was then used as a prediction algorithm and applied in a prospective fashion to the 2006 and 2007 patient data sets.
For the purpose of performing the RPART analysis, each set of case data entered into the analysis consisted of a random extraction of the desired clinical data within the specified extraction window from a single case patient. Thus, if a case patient had more than 1 value available for any variable of interest, 1 value was randomly selected to be entered in combination with the other available clinical data. Furthermore, in order to ensure that the majority of case patient data were included in the analysis, this process was iterated 10 times for each case patient. This resulted in 10 sets of case patient data being entered into the analysis for each case patient in the database, with each set containing a value for all variables of interest randomly extracted from those available for that patient. In addition to ensuring that the majority of case patient data were included, this technique also functionally expands the number of case patients present in the analysis. As there were far more control patients than case patients in the database, this in turn results in a classification tree that does not simply identify controls without regard to the relatively small number of case patients.
Data for the control patients entered into the analysis were extracted in a similar fashion, though only 1 set of data were included in the analysis for each control patient present in the database. As a result, only 1 randomly selected value per variable was included in the analysis.
Results
Patients
During 2005, 562 septic patients and 13,223 control patients were identified. For 2006 and 2007 there were 635 and 667 case patients, and 13,102 and 13,270 control patients, respectively.
Predictors of Sepsis
RPART analysis of the 2005 patient data set demonstrated that the most significant predictors of sepsis in the 24 hours preceding transfer to the medical ICU were the partial pressure of arterial oxygen (PaO2), systolic blood pressure, absolute neutrophil count, blood urea nitrogen (BUN), pH, bicarbonate, chloride, and albumin. This resulted in a simple algorithm with nine classification splits (Figure 1), which was then prospectively applied to the 2006 and 2007 patient data sets. These results are summarized in Table 2.
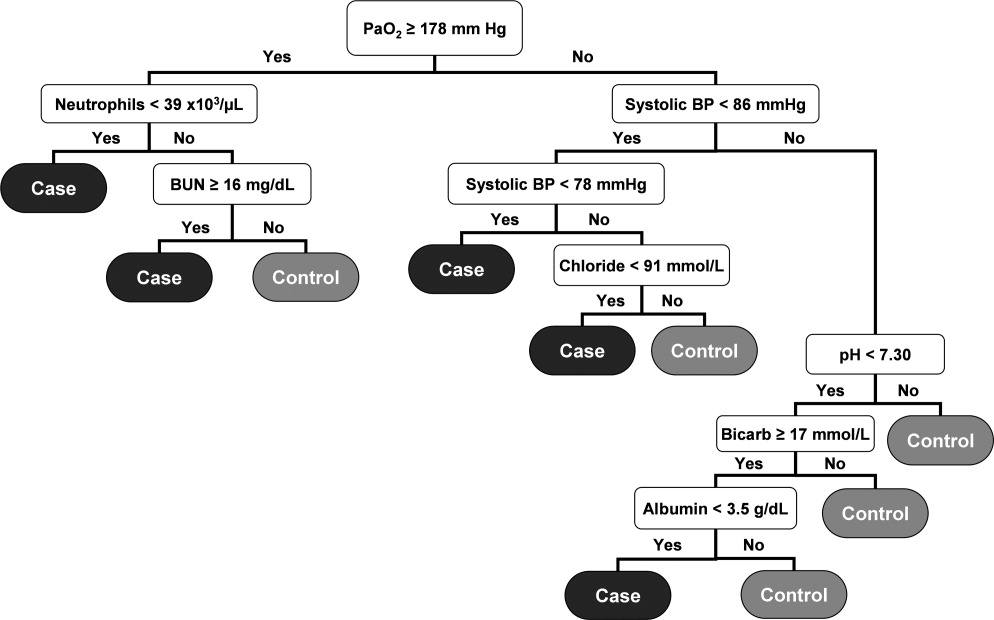
| Total Number | Number Correctly Classified (%) | Case Identification Time Before ICU Admission (minutes) | PPV (%) | NPV (%) | MCR (%) | |
|---|---|---|---|---|---|---|
| ||||||
| 2005 | 27.9 | 98.1 | 7.8 | |||
| Cases | 562 | 320 (56.9) | ||||
| Controls | 13,223 | 12,394 (93.7) | ||||
| 2006 | 179 230 | 28.7 | 97.7 | 8.4 | ||
| Cases | 635 | 347 (54.7) | ||||
| Controls | 13,102 | 12,241 (93.4) | ||||
| 2007 | 192 210 | 28.3 | 97.6 | 8.8 | ||
| Cases | 667 | 367 (55.0) | ||||
| Controls | 13,270 | 12,341 (93.0) | ||||
The resulting classification model had a low total misclassification rate for the 2005 data. Of the 562 septic patients, 320 (56.9%) were correctly classified, and 12,394 (93.7%) of the control patients were appropriately identified. The number of septic and control patients misclassified was 242 and 829, respectively, yielding a total misclassification rate of 7.8%. When applied to the 2006 patient data set, 347 (54.7%) of the 635 septic shock patients were correctly identified, while 12,241 (93.4%) of the 13,102 control patients were correctly classified. The total misclassification rate for the 2006 patient set was 8.4%. For the 2007 patient data, 367 (55.0%) of the 667 case patients were correctly identified, and 12,341 (93.0%) of the 13,270 control patients were correctly identified. This resulted in a total misclassification rate of 8.8%.
The 2006 and 2007 case patients were identified 179 230 minutes and 192 210 minutes before ICU transfer, respectively (Figure 2). The algorithm demonstrated positive and negative predictive values of 28.7% and 97.7% for the 2006 patient set, respectively, and 28.3% and 97.6% for the 2007 patient set, respectively.
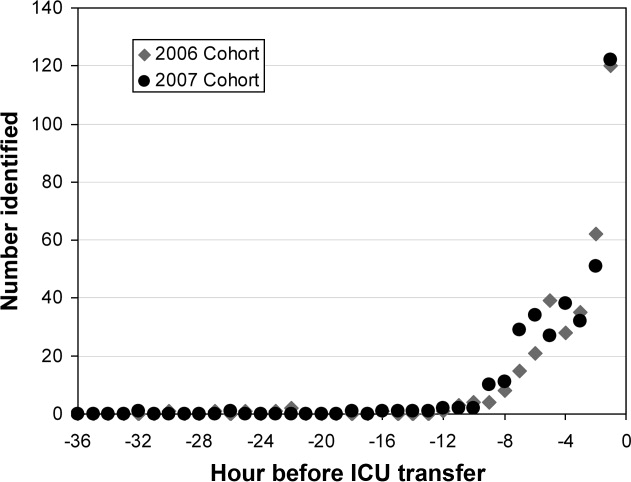
Although the prediction algorithm shown in Figure 1 identified the majority of the case patients with ample time for clinical intervention prior to ICU transfer, the analysis used to derive this model included values for the arterial blood gas (ABG). As this is not a routinely obtained study for hospitalized patients outside of an ICU, it is possible that the performance of this model can in part be attributed to clinical acumen rather than changes in patient physiology. The ABG would likely only be obtained in patients with a more concerning or deteriorating clinical course, and thus more likely to develop shock. To address this possibility, a second analysis was performed that did not include the values for the ABG. The result was an algorithm with 13 classification splits, as shown in Figure 3.
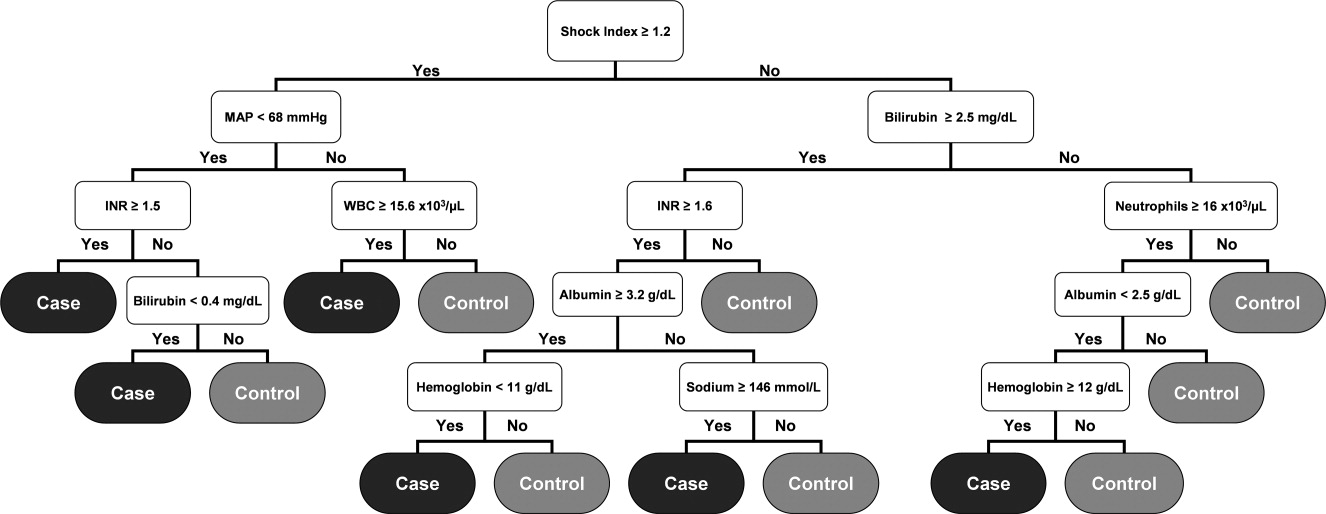
The most predictive clinical variables in this analysis included the shock index (heart rate divided by systolic blood pressure), mean arterial pressure, total bilirubin, international normalized ratio (INR), total white blood cell count, absolute neutrophil count, albumin, hemoglobin, and sodium. This model was again applied to the 2006 and 2007 patient data sets (Table 3).
| Total Number | Number Correctly Classified (%) | Case Identification Time Before ICU Admission (minutes) | PPV (%) | NPV (%) | MCR (%) | |
|---|---|---|---|---|---|---|
| ||||||
| 2005 | 20.5 | 96.7 | 6.7 | |||
| Cases | 562 | 126 (22.4) | ||||
| Controls | 13,223 | 12,735 (96.3) | ||||
| 2006 | 508 536 | 21.4 | 96.1 | 7.0 | ||
| Cases | 635 | 121 (19.1) | ||||
| Controls | 13,102 | 12,657 (96.6) | ||||
| 2007 | 496 512 | 19.5 | 95.8 | 7.1 | ||
| Cases | 667 | 102 (15.3) | ||||
| Controls | 13,270 | 12,850 (96.8) | ||||
The overall misclassification rates for 2006 and 2007 were 7.0% and 7.1%, respectively. The model correctly identified 121 (19.1%) of the 635 cases and 12,657 (96.6%) of the 13,102 control patients from 2006, and 102 (15.3%) of the 667 cases and 12,850 (96.8%) of the 13,270 control patients from 2007. The respective positive and negative predictive values were 21.4% and 96.1% for 2006, respectively, and 19.5% and 95.8% for 2007, respectively.
Although the overall performance of the model derived without the ABG data was not as good, the identification times prior to ICU transfer were significantly improved. For the 2006 data, patients were identified 508 536 minutes before transfer (Figure 4), compared to 179 230 minutes for the model that included the ABG data (P < 0.01). For the 2007 data, patients were identified 496 512 minutes prior to ICU admission (Figure 4), compared to 192 210 minutes for the previous model (P < 0.01).
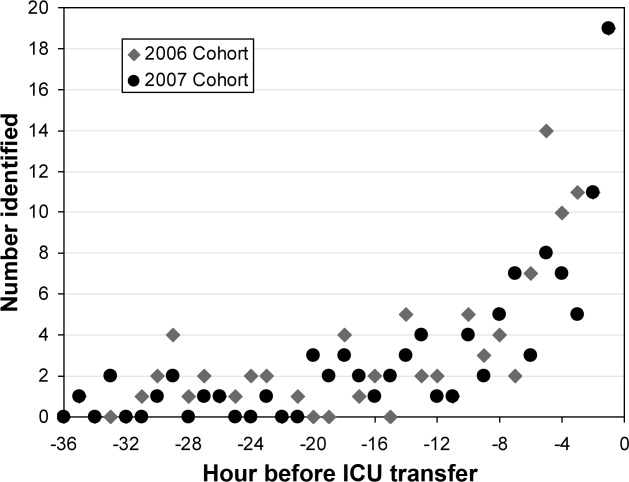
Discussion
We have demonstrated a simple method for generating an algorithm derived from routine laboratory and hemodynamic values that is capable of predicting the onset of sepsis in a significant proportion of non‐ICU patients. Two prediction models were generated, 1 with and 1 without ABG data included in the analysis. In the 2006 and 2007 validation cohorts, the model including these data correctly classified 54.7% and 55.0% of the patients who developed septic shock and 93.4% and 93.0% of control patients, respectively. The second model identified 19.1% and 15.3% of the septic shock patients and 96.6% and 96.8% of the control patients for 2006 and 2007, respectively. The methods used in generating this model are relatively simple and can be executed with the use of an electronic medical record system.
Early, goal‐directed cardiovascular resuscitation and adequate initial antibiotic therapy have been shown to decrease mortality in patients with severe sepsis and septic shock.2, 26 Prior studies employing early, targeted resuscitation strategies have demonstrated decreased use of vasopressors10 and decreased mortality.510 In addition, we previously demonstrated that a standardized order set for the management of severe sepsis in the emergency department that focused on early and aggressive intervention was associated with decreased 28‐day mortality.1 These studies suggest that early, aggressive management of septic shock can improve outcomes. Identification of patients prior to overt clinical deterioration may allow for early intervention aimed at preventing shock or improving its outcome.
The purpose of this method is to develop a model capable of recognizing patterns in clinical data that herald a patient's otherwise unidentified clinical deterioration. It is not intended to replace existing outcome prediction tools or severity of illness scoring systems, where a high degree of accuracy would be required. Rather, it would be best implemented as an automated screening tool incorporated into an electronic medical record system. When a hospitalized patient is identified as a possible septic shock patient by the classification tree, a notification is then issued to the clinicians caring for the patient. The primary goal of this method is to notify clinicians of potential clinical deterioration. Any action taken as a result of this notification is at the discretion of the clinician. This method could be employed for any population of hospitalized patients, though because of variations in clinical practice and patient physiology, different models would need to be generated for differing patient populations.
This method has limitations, the foremost of which is the possible instability of the resulting classification model. This type of analysis results in an algorithm that depends on binary splits to classify patients. In generating the algorithm, the recursive partitioning analysis selects the variables and cutoff values that result in the strongest decision tree with the most pure classifications at the end nodes. These variables and cutoff values may not immediately seem logical from a clinical standpoint, and may vary with changes in practice and even possibly between divisions within a hospital. As a result, the algorithm would likely require intermittent updating to remain effective and a model derived from 1 hospital or patient population would not necessarily be applicable to patients at another institution or from a different population. However, once the method has been developed at an institution, the process of revising the algorithm could be essentially automated and uses few resources.
Another shortcoming of this method is the relatively low sensitivity of the resulting algorithm. In a role as an automated alert system, a low false‐positive rate is particularly desirable to avoid unnecessary frequent distraction of clinicians. The sensitivity of the model can be improved through manipulation of how the analysis is performed, but this would be at the expense of a higher false‐positive rate, which is not acceptable. Finally, prior studies examining treatment for sepsis have demonstrated an advantage to early and aggressive therapy. It is not clear, however, if identifying these patients prior to the onset of clinically evident sepsis would result in improved outcomes. Further work is required to determine if this is the case. We are currently conducting a prospective study that employs the method described here in conjunction with an automated alert system to ascertain if it impacts outcomes on patients admitted to the medicine wards of Barnes‐Jewish Hospital.
In conclusion, the method presented here represents a technique that consumes few resources and is capable of identifying some patients before septic shock becomes clinically evident. When applied in an automated fashion with the capability to alert clinicians caring for a patient, the method demonstrated here may allow for earlier diagnosis and possibly intervention for septic shock patients.
- ,,, et al.Before–after study of a standardized hospital order set for the management of septic shock.Crit Care Med.2006;34(11):2707–2713.
- ,,, et al.Early goal‐directed therapy in the treatment of severe sepsis and septic shock.N Engl J Med.2001;345:1368–1377.
- ,,, et al.Early goal‐directed therapy in severe sepsis and septic shock revisited; concepts, controversies, and contemporary findings.Chest.2006;130(5):1579–1595.
- .Implementing the severe sepsis care bundles outside the ICU by outreach.Nurs Crit Care.2007;12:225–229.
- ,,, et al.The impact of compliance with 6‐hour and 24‐hour sepsis bundles on hospital mortality in patients with severe sepsis: a prospective observational study.Crit Care.2005;9:R764–R770.
- ,,, et al.Translating research to clinical practice, a 1‐year experience with implementing early goal‐directed therapy for septic shock in the emergency department.Chest.2006;129:225–232.
- ,,, et al.Prospective external validation of the clinical effectiveness of an emergency department‐based early goal‐directed therapy protocol for severe sepsis and septic shock.Chest.2007;132:425–432.
- ,,, et al.Implementation of a bundle of quality indicators for the early management of severe sepsis and septic shock is associated with decreased mortality.Crit Care Med.2007;35:1105–1112.
- ,,, et al.Implementation of an evidence‐based “standard operating procedure” and outcome in septic shock.Crit Care Med.2006;34:943–949.
- ,,, et al.Outcome of septic shock in older adults after implementation of the sepsis “bundle”.J Am Geriatr Soc.2008;56:272–278.
- ,,, et al.Confidential inquiry into quality of care before admission to intensive care.BMJ.1998;316:1853–1858.
- ,,.Unexpected deaths and referrals to intensive care of patients on general wards. Are some cases potentially avoidable?J R Coll Physicians Lond.1999;33(3):255–259.
- ,,, et al.Septic shock: an analysis of outcomes for patients with onset on hospital wards versus intensive care units.Crit Care Med.1998;26(6):1020–1024.
- ,,,,,.Hospital‐wide impact of a standardized order set for the management of bacteremic severe sepsis.Crit Care Med.2009;37(3):819–824.
- ,,,,,.The mRNA expression of fatty acid amide hydrolase in human whole blood correlates with sepsis.J Endotoxin Res.2007;13(1):35–38.
- ,,, et al.C‐reactive protein use as an early indicator of infection in patients with systemic inflammatory response syndrome.Intensive Care Med.2004;30(11):2038–2045.
- ,,,,.Procalcitonin as a screening test of late‐onset sepsis in preterm very low birth weight infants.J Perinatol.2005;25(6):397–402.
- ,,.A simple method for predicting severe sepsis in burn patients.Am J Surg.1980;139(4):513–517.
- ,,, et al.Prognostic value of protein C concentrations in neutropenic patients at high risk of severe septic complications.Crit Care Med.2000;28(7):2209–2216.
- ,,,,.Circulating immune parameters predicting the progression from hospital‐acquired pneumonia to septic shock in surgical patients.Crit Care Med.2005;9(6):R662–R669.
- ,,.A simple prediction algorithm for bacteremia in patients with acute febrile illness.Q J Med.2005;98:813–820.
- ,,,.Infection probability score (IPS): a method to help assess the probability of infection in critically ill patients.Crit Care Med.2003;31(11):2579–2584.
- ,.Multivariate regression modeling for the prediction of inflammation, systemic pressure, and end‐organ function in severe sepsis.Shock.1997;8(3):225–231.
- ,,,,,.Abnormal heart rate characteristics preceding neonatal sepsis and sepsis‐like illness.Pediatr Res.2003;53:920–926.
- ,.Statistics for Biology and Health.New York:Springer‐Verlag;1999.
- ,,, et al.Impact of adequate empiric antibiotic therapy on the outcome of patients admitted to the intensive care unit with sepsis.Crit Care Med.2003;31:2742–2751.
Severe sepsis is responsible for significant morbidity and mortality. In the United States, approximately 750,000 cases occur each year with an estimated mortality of 30% to 50%.1 Early goal‐directed therapy has been shown to decrease mortality in patients with severe sepsis and septic shock.2, 3 As a result, efforts have been focused toward providing early and aggressive intervention once sepsis has been established. In many cases this has been accomplished through the implementation of a protocol with guidelines for fluid management, antibiotic and vasopressor administration, and other interventions.410 Prior studies have demonstrated that care of hospitalized patients before intensive care unit (ICU) admission is often suboptimal,1113 and have suggested that patients with clear indicators of acute deterioration may go unrecognized on the ward. We previously reported the effects of implementing a hospital‐wide protocol for the management of severe sepsis,14 finding that although there was a significant reduction in overall mortality there was no difference for patients who developed severe sepsis on the hospital ward. This finding also suggests that the initial care of patients with severe sepsis on hospital wards may differ in intensity compared to emergency departments and ICUs. Failure on the part of the clinician to recognize the harbingers of impending sepsis before the onset of organ dysfunction or hypotension may contribute to a delay in aggressive therapy.
Previous efforts at early recognition of sepsis have relied on diagnostic studies or specific biomarkers to screen at‐risk patients. These have included such studies as messenger RNA (mRNA) expression,15 C‐reactive protein,16 procalcitonin in newborns,17 immunocompetence measures in burn patients,18 protein C concentration in neutropenic patients,19 and several immune markers (eg, tumor necrosis factor‐alpha, interleukin [IL]‐1 beta, IL‐6, IL‐8, and IL‐10).20 However, these biomarkers have been studied only in specific patient populations, require suspicion on the part of the clinician and the measurement of diagnostic or laboratory values that would otherwise not have been obtained. The ideal tool for predicting the onset of sepsis would be applicable to a broad patient population, not require specific suspicion on the part of the clinician, and use only routinely obtained clinical measurements and laboratory values.
Prediction models and scoring systems that use routine hemodynamic and laboratory values for several endpoints related to sepsis and septic shock have been developed. Many such tools are used to define severity of illness and predict outcome, while others have been developed to predict such events as bacteremia in patients presenting with fever,21 the probability of infection in the critically ill,22 and end‐organ dysfunction in severe sepsis.23 Little work has been done to develop such a model capable of predicting the onset of sepsis,24 and there have been no attempts to deploy a model as a large‐scale screening tool.
Our objective was to develop a simple algorithm that can be used in an automated fashion to screen hospitalized patients for impending septic shock. Such a model would be derived from routine hemodynamic and laboratory values, and take advantage of a computerized medical record system for data collection.
Patients and Methods
Patient Enrollment and Data Collection
This study was conducted at Barnes‐Jewish Hospital, St. Louis, MO, a university‐affiliated, urban teaching hospital. The study was approved by the Washington University (St. Louis, MO) School of Medicine Human Studies Committee. Patients included in the study where those hospitalized during 2005, 2006, and 2007, and who had at least 1 International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD9) discharge diagnosis code for the medical/nonsurgical diagnoses listed in Appendix 1. From this pool of patients, septic shock patients were identified as those who were admitted to the hospital ward and later developed septic shock requiring transfer to an ICU for vasopressor support and hemodynamic monitoring. This was accomplished by using discharge ICD9 codes for acute infection matched to codes for acute organ dysfunction and the need for vasopressors within 24 hours of ICU transfer (Appendix 1). The patients used as controls were then all those remaining in the pool once the septic shock patients were identified and separated.
Case patients were excluded from the analysis if they were transferred to the ICU within 2 hours of hospital admission, as these patients are unlikely to have an adequate amount of pretransfer clinical data available for analysis. Both case and control patients were excluded if they lacked any value for basic, routine laboratory data (serum sodium, chloride, total bicarbonate, urea nitrogen, creatinine, glucose, white blood cell count, neutrophil count, hemoglobin, hematocrit, and platelet count) and certain vital signs (blood pressure, heart rate, temperature). Patient data from 2005 were used in the derivation of the prediction model, and 2006 and 2007 patient data were used to prospectively validate the model. Clinical variables used in the analysis were selected based on both ease of access from the electronic medical record and clinical relevance, and are shown in Table 1.
|
| Age (years) |
| Albumin (g/dL) |
| Arterial blood gas (pH, PaCO2, PaO2) |
| Anion gap |
| Bilirubin (mg/dL) |
| BP, systolic and diastolic (mm of Hg) |
| Blood urea nitrogen (mg/dL) |
| Chloride (mmol/L) |
| Creatinine (mg/dL) |
| Glucose (mg/dL) |
| Hemoglobin (g/dL) |
| International normalized ratio |
| Neutrophil count, absolute (1 103/L) |
| Platelet count (1 103/L) |
| Pulse (beats/minute) |
| Pulse pressure (mm of Hg) |
| Shock index (pulse divided by systolic BP) |
| Sodium (mmol/L) |
| Total bicarbonate (mmol/L) |
| Temperature (degrees Celsius) |
| White blood cell count (1 103/L) |
In performing the Recursive Partitioning And Regression Tree (RPART) analysis to generate a prediction model, data for case patients were extracted in a window from 24 hours to 2 hours before ICU admission. The data collection window excluded the 2 hours prior to ICU transfer in order to minimize the effect of acute hemodynamic or laboratory changes that may have prompted the transfer; the purpose of the model is to identify hemodynamic and laboratory patterns in the several hours before the onset of clinically evident shock, so data from a time during which impending shock was clinically apparent were excluded. For the control patients, data from the first 48 hours of their hospitalization were included in the analysis.
Statistical Analysis
RPART analysis was performed on the 2005 patient data set to generate a prediction algorithm. This method of analysis results in a classification tree that contains a series of binary splits designed to separate patients into mutually exclusive subgroups.25 Each split in the tree is selected based on its ability to produce a partition with the greatest purity. Initially, a large tree that contains splits for all input variables is generated. This initial tree is generally too large to be useful as the final subgroups are too small to make sensible statistical inference.25 A pruning process is then applied to the initial tree with the goal of finding the subtree that is most predictive of the outcome of interest. The analysis was done using the RPART package of the R statistical analysis program, version 2.7.0 (R: A Language and Environment for Statistical Computing, R Development Core Team, Foundation for Statistical Computing, Vienna, Austria). The resulting classification tree was then used as a prediction algorithm and applied in a prospective fashion to the 2006 and 2007 patient data sets.
For the purpose of performing the RPART analysis, each set of case data entered into the analysis consisted of a random extraction of the desired clinical data within the specified extraction window from a single case patient. Thus, if a case patient had more than 1 value available for any variable of interest, 1 value was randomly selected to be entered in combination with the other available clinical data. Furthermore, in order to ensure that the majority of case patient data were included in the analysis, this process was iterated 10 times for each case patient. This resulted in 10 sets of case patient data being entered into the analysis for each case patient in the database, with each set containing a value for all variables of interest randomly extracted from those available for that patient. In addition to ensuring that the majority of case patient data were included, this technique also functionally expands the number of case patients present in the analysis. As there were far more control patients than case patients in the database, this in turn results in a classification tree that does not simply identify controls without regard to the relatively small number of case patients.
Data for the control patients entered into the analysis were extracted in a similar fashion, though only 1 set of data were included in the analysis for each control patient present in the database. As a result, only 1 randomly selected value per variable was included in the analysis.
Results
Patients
During 2005, 562 septic patients and 13,223 control patients were identified. For 2006 and 2007 there were 635 and 667 case patients, and 13,102 and 13,270 control patients, respectively.
Predictors of Sepsis
RPART analysis of the 2005 patient data set demonstrated that the most significant predictors of sepsis in the 24 hours preceding transfer to the medical ICU were the partial pressure of arterial oxygen (PaO2), systolic blood pressure, absolute neutrophil count, blood urea nitrogen (BUN), pH, bicarbonate, chloride, and albumin. This resulted in a simple algorithm with nine classification splits (Figure 1), which was then prospectively applied to the 2006 and 2007 patient data sets. These results are summarized in Table 2.

| Total Number | Number Correctly Classified (%) | Case Identification Time Before ICU Admission (minutes) | PPV (%) | NPV (%) | MCR (%) | |
|---|---|---|---|---|---|---|
| ||||||
| 2005 | 27.9 | 98.1 | 7.8 | |||
| Cases | 562 | 320 (56.9) | ||||
| Controls | 13,223 | 12,394 (93.7) | ||||
| 2006 | 179 230 | 28.7 | 97.7 | 8.4 | ||
| Cases | 635 | 347 (54.7) | ||||
| Controls | 13,102 | 12,241 (93.4) | ||||
| 2007 | 192 210 | 28.3 | 97.6 | 8.8 | ||
| Cases | 667 | 367 (55.0) | ||||
| Controls | 13,270 | 12,341 (93.0) | ||||
The resulting classification model had a low total misclassification rate for the 2005 data. Of the 562 septic patients, 320 (56.9%) were correctly classified, and 12,394 (93.7%) of the control patients were appropriately identified. The number of septic and control patients misclassified was 242 and 829, respectively, yielding a total misclassification rate of 7.8%. When applied to the 2006 patient data set, 347 (54.7%) of the 635 septic shock patients were correctly identified, while 12,241 (93.4%) of the 13,102 control patients were correctly classified. The total misclassification rate for the 2006 patient set was 8.4%. For the 2007 patient data, 367 (55.0%) of the 667 case patients were correctly identified, and 12,341 (93.0%) of the 13,270 control patients were correctly identified. This resulted in a total misclassification rate of 8.8%.
The 2006 and 2007 case patients were identified 179 230 minutes and 192 210 minutes before ICU transfer, respectively (Figure 2). The algorithm demonstrated positive and negative predictive values of 28.7% and 97.7% for the 2006 patient set, respectively, and 28.3% and 97.6% for the 2007 patient set, respectively.

Although the prediction algorithm shown in Figure 1 identified the majority of the case patients with ample time for clinical intervention prior to ICU transfer, the analysis used to derive this model included values for the arterial blood gas (ABG). As this is not a routinely obtained study for hospitalized patients outside of an ICU, it is possible that the performance of this model can in part be attributed to clinical acumen rather than changes in patient physiology. The ABG would likely only be obtained in patients with a more concerning or deteriorating clinical course, and thus more likely to develop shock. To address this possibility, a second analysis was performed that did not include the values for the ABG. The result was an algorithm with 13 classification splits, as shown in Figure 3.

The most predictive clinical variables in this analysis included the shock index (heart rate divided by systolic blood pressure), mean arterial pressure, total bilirubin, international normalized ratio (INR), total white blood cell count, absolute neutrophil count, albumin, hemoglobin, and sodium. This model was again applied to the 2006 and 2007 patient data sets (Table 3).
| Total Number | Number Correctly Classified (%) | Case Identification Time Before ICU Admission (minutes) | PPV (%) | NPV (%) | MCR (%) | |
|---|---|---|---|---|---|---|
| ||||||
| 2005 | 20.5 | 96.7 | 6.7 | |||
| Cases | 562 | 126 (22.4) | ||||
| Controls | 13,223 | 12,735 (96.3) | ||||
| 2006 | 508 536 | 21.4 | 96.1 | 7.0 | ||
| Cases | 635 | 121 (19.1) | ||||
| Controls | 13,102 | 12,657 (96.6) | ||||
| 2007 | 496 512 | 19.5 | 95.8 | 7.1 | ||
| Cases | 667 | 102 (15.3) | ||||
| Controls | 13,270 | 12,850 (96.8) | ||||
The overall misclassification rates for 2006 and 2007 were 7.0% and 7.1%, respectively. The model correctly identified 121 (19.1%) of the 635 cases and 12,657 (96.6%) of the 13,102 control patients from 2006, and 102 (15.3%) of the 667 cases and 12,850 (96.8%) of the 13,270 control patients from 2007. The respective positive and negative predictive values were 21.4% and 96.1% for 2006, respectively, and 19.5% and 95.8% for 2007, respectively.
Although the overall performance of the model derived without the ABG data was not as good, the identification times prior to ICU transfer were significantly improved. For the 2006 data, patients were identified 508 536 minutes before transfer (Figure 4), compared to 179 230 minutes for the model that included the ABG data (P < 0.01). For the 2007 data, patients were identified 496 512 minutes prior to ICU admission (Figure 4), compared to 192 210 minutes for the previous model (P < 0.01).

Discussion
We have demonstrated a simple method for generating an algorithm derived from routine laboratory and hemodynamic values that is capable of predicting the onset of sepsis in a significant proportion of non‐ICU patients. Two prediction models were generated, 1 with and 1 without ABG data included in the analysis. In the 2006 and 2007 validation cohorts, the model including these data correctly classified 54.7% and 55.0% of the patients who developed septic shock and 93.4% and 93.0% of control patients, respectively. The second model identified 19.1% and 15.3% of the septic shock patients and 96.6% and 96.8% of the control patients for 2006 and 2007, respectively. The methods used in generating this model are relatively simple and can be executed with the use of an electronic medical record system.
Early, goal‐directed cardiovascular resuscitation and adequate initial antibiotic therapy have been shown to decrease mortality in patients with severe sepsis and septic shock.2, 26 Prior studies employing early, targeted resuscitation strategies have demonstrated decreased use of vasopressors10 and decreased mortality.510 In addition, we previously demonstrated that a standardized order set for the management of severe sepsis in the emergency department that focused on early and aggressive intervention was associated with decreased 28‐day mortality.1 These studies suggest that early, aggressive management of septic shock can improve outcomes. Identification of patients prior to overt clinical deterioration may allow for early intervention aimed at preventing shock or improving its outcome.
The purpose of this method is to develop a model capable of recognizing patterns in clinical data that herald a patient's otherwise unidentified clinical deterioration. It is not intended to replace existing outcome prediction tools or severity of illness scoring systems, where a high degree of accuracy would be required. Rather, it would be best implemented as an automated screening tool incorporated into an electronic medical record system. When a hospitalized patient is identified as a possible septic shock patient by the classification tree, a notification is then issued to the clinicians caring for the patient. The primary goal of this method is to notify clinicians of potential clinical deterioration. Any action taken as a result of this notification is at the discretion of the clinician. This method could be employed for any population of hospitalized patients, though because of variations in clinical practice and patient physiology, different models would need to be generated for differing patient populations.
This method has limitations, the foremost of which is the possible instability of the resulting classification model. This type of analysis results in an algorithm that depends on binary splits to classify patients. In generating the algorithm, the recursive partitioning analysis selects the variables and cutoff values that result in the strongest decision tree with the most pure classifications at the end nodes. These variables and cutoff values may not immediately seem logical from a clinical standpoint, and may vary with changes in practice and even possibly between divisions within a hospital. As a result, the algorithm would likely require intermittent updating to remain effective and a model derived from 1 hospital or patient population would not necessarily be applicable to patients at another institution or from a different population. However, once the method has been developed at an institution, the process of revising the algorithm could be essentially automated and uses few resources.
Another shortcoming of this method is the relatively low sensitivity of the resulting algorithm. In a role as an automated alert system, a low false‐positive rate is particularly desirable to avoid unnecessary frequent distraction of clinicians. The sensitivity of the model can be improved through manipulation of how the analysis is performed, but this would be at the expense of a higher false‐positive rate, which is not acceptable. Finally, prior studies examining treatment for sepsis have demonstrated an advantage to early and aggressive therapy. It is not clear, however, if identifying these patients prior to the onset of clinically evident sepsis would result in improved outcomes. Further work is required to determine if this is the case. We are currently conducting a prospective study that employs the method described here in conjunction with an automated alert system to ascertain if it impacts outcomes on patients admitted to the medicine wards of Barnes‐Jewish Hospital.
In conclusion, the method presented here represents a technique that consumes few resources and is capable of identifying some patients before septic shock becomes clinically evident. When applied in an automated fashion with the capability to alert clinicians caring for a patient, the method demonstrated here may allow for earlier diagnosis and possibly intervention for septic shock patients.
Severe sepsis is responsible for significant morbidity and mortality. In the United States, approximately 750,000 cases occur each year with an estimated mortality of 30% to 50%.1 Early goal‐directed therapy has been shown to decrease mortality in patients with severe sepsis and septic shock.2, 3 As a result, efforts have been focused toward providing early and aggressive intervention once sepsis has been established. In many cases this has been accomplished through the implementation of a protocol with guidelines for fluid management, antibiotic and vasopressor administration, and other interventions.410 Prior studies have demonstrated that care of hospitalized patients before intensive care unit (ICU) admission is often suboptimal,1113 and have suggested that patients with clear indicators of acute deterioration may go unrecognized on the ward. We previously reported the effects of implementing a hospital‐wide protocol for the management of severe sepsis,14 finding that although there was a significant reduction in overall mortality there was no difference for patients who developed severe sepsis on the hospital ward. This finding also suggests that the initial care of patients with severe sepsis on hospital wards may differ in intensity compared to emergency departments and ICUs. Failure on the part of the clinician to recognize the harbingers of impending sepsis before the onset of organ dysfunction or hypotension may contribute to a delay in aggressive therapy.
Previous efforts at early recognition of sepsis have relied on diagnostic studies or specific biomarkers to screen at‐risk patients. These have included such studies as messenger RNA (mRNA) expression,15 C‐reactive protein,16 procalcitonin in newborns,17 immunocompetence measures in burn patients,18 protein C concentration in neutropenic patients,19 and several immune markers (eg, tumor necrosis factor‐alpha, interleukin [IL]‐1 beta, IL‐6, IL‐8, and IL‐10).20 However, these biomarkers have been studied only in specific patient populations, require suspicion on the part of the clinician and the measurement of diagnostic or laboratory values that would otherwise not have been obtained. The ideal tool for predicting the onset of sepsis would be applicable to a broad patient population, not require specific suspicion on the part of the clinician, and use only routinely obtained clinical measurements and laboratory values.
Prediction models and scoring systems that use routine hemodynamic and laboratory values for several endpoints related to sepsis and septic shock have been developed. Many such tools are used to define severity of illness and predict outcome, while others have been developed to predict such events as bacteremia in patients presenting with fever,21 the probability of infection in the critically ill,22 and end‐organ dysfunction in severe sepsis.23 Little work has been done to develop such a model capable of predicting the onset of sepsis,24 and there have been no attempts to deploy a model as a large‐scale screening tool.
Our objective was to develop a simple algorithm that can be used in an automated fashion to screen hospitalized patients for impending septic shock. Such a model would be derived from routine hemodynamic and laboratory values, and take advantage of a computerized medical record system for data collection.
Patients and Methods
Patient Enrollment and Data Collection
This study was conducted at Barnes‐Jewish Hospital, St. Louis, MO, a university‐affiliated, urban teaching hospital. The study was approved by the Washington University (St. Louis, MO) School of Medicine Human Studies Committee. Patients included in the study where those hospitalized during 2005, 2006, and 2007, and who had at least 1 International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD9) discharge diagnosis code for the medical/nonsurgical diagnoses listed in Appendix 1. From this pool of patients, septic shock patients were identified as those who were admitted to the hospital ward and later developed septic shock requiring transfer to an ICU for vasopressor support and hemodynamic monitoring. This was accomplished by using discharge ICD9 codes for acute infection matched to codes for acute organ dysfunction and the need for vasopressors within 24 hours of ICU transfer (Appendix 1). The patients used as controls were then all those remaining in the pool once the septic shock patients were identified and separated.
Case patients were excluded from the analysis if they were transferred to the ICU within 2 hours of hospital admission, as these patients are unlikely to have an adequate amount of pretransfer clinical data available for analysis. Both case and control patients were excluded if they lacked any value for basic, routine laboratory data (serum sodium, chloride, total bicarbonate, urea nitrogen, creatinine, glucose, white blood cell count, neutrophil count, hemoglobin, hematocrit, and platelet count) and certain vital signs (blood pressure, heart rate, temperature). Patient data from 2005 were used in the derivation of the prediction model, and 2006 and 2007 patient data were used to prospectively validate the model. Clinical variables used in the analysis were selected based on both ease of access from the electronic medical record and clinical relevance, and are shown in Table 1.
|
| Age (years) |
| Albumin (g/dL) |
| Arterial blood gas (pH, PaCO2, PaO2) |
| Anion gap |
| Bilirubin (mg/dL) |
| BP, systolic and diastolic (mm of Hg) |
| Blood urea nitrogen (mg/dL) |
| Chloride (mmol/L) |
| Creatinine (mg/dL) |
| Glucose (mg/dL) |
| Hemoglobin (g/dL) |
| International normalized ratio |
| Neutrophil count, absolute (1 103/L) |
| Platelet count (1 103/L) |
| Pulse (beats/minute) |
| Pulse pressure (mm of Hg) |
| Shock index (pulse divided by systolic BP) |
| Sodium (mmol/L) |
| Total bicarbonate (mmol/L) |
| Temperature (degrees Celsius) |
| White blood cell count (1 103/L) |
In performing the Recursive Partitioning And Regression Tree (RPART) analysis to generate a prediction model, data for case patients were extracted in a window from 24 hours to 2 hours before ICU admission. The data collection window excluded the 2 hours prior to ICU transfer in order to minimize the effect of acute hemodynamic or laboratory changes that may have prompted the transfer; the purpose of the model is to identify hemodynamic and laboratory patterns in the several hours before the onset of clinically evident shock, so data from a time during which impending shock was clinically apparent were excluded. For the control patients, data from the first 48 hours of their hospitalization were included in the analysis.
Statistical Analysis
RPART analysis was performed on the 2005 patient data set to generate a prediction algorithm. This method of analysis results in a classification tree that contains a series of binary splits designed to separate patients into mutually exclusive subgroups.25 Each split in the tree is selected based on its ability to produce a partition with the greatest purity. Initially, a large tree that contains splits for all input variables is generated. This initial tree is generally too large to be useful as the final subgroups are too small to make sensible statistical inference.25 A pruning process is then applied to the initial tree with the goal of finding the subtree that is most predictive of the outcome of interest. The analysis was done using the RPART package of the R statistical analysis program, version 2.7.0 (R: A Language and Environment for Statistical Computing, R Development Core Team, Foundation for Statistical Computing, Vienna, Austria). The resulting classification tree was then used as a prediction algorithm and applied in a prospective fashion to the 2006 and 2007 patient data sets.
For the purpose of performing the RPART analysis, each set of case data entered into the analysis consisted of a random extraction of the desired clinical data within the specified extraction window from a single case patient. Thus, if a case patient had more than 1 value available for any variable of interest, 1 value was randomly selected to be entered in combination with the other available clinical data. Furthermore, in order to ensure that the majority of case patient data were included in the analysis, this process was iterated 10 times for each case patient. This resulted in 10 sets of case patient data being entered into the analysis for each case patient in the database, with each set containing a value for all variables of interest randomly extracted from those available for that patient. In addition to ensuring that the majority of case patient data were included, this technique also functionally expands the number of case patients present in the analysis. As there were far more control patients than case patients in the database, this in turn results in a classification tree that does not simply identify controls without regard to the relatively small number of case patients.
Data for the control patients entered into the analysis were extracted in a similar fashion, though only 1 set of data were included in the analysis for each control patient present in the database. As a result, only 1 randomly selected value per variable was included in the analysis.
Results
Patients
During 2005, 562 septic patients and 13,223 control patients were identified. For 2006 and 2007 there were 635 and 667 case patients, and 13,102 and 13,270 control patients, respectively.
Predictors of Sepsis
RPART analysis of the 2005 patient data set demonstrated that the most significant predictors of sepsis in the 24 hours preceding transfer to the medical ICU were the partial pressure of arterial oxygen (PaO2), systolic blood pressure, absolute neutrophil count, blood urea nitrogen (BUN), pH, bicarbonate, chloride, and albumin. This resulted in a simple algorithm with nine classification splits (Figure 1), which was then prospectively applied to the 2006 and 2007 patient data sets. These results are summarized in Table 2.

| Total Number | Number Correctly Classified (%) | Case Identification Time Before ICU Admission (minutes) | PPV (%) | NPV (%) | MCR (%) | |
|---|---|---|---|---|---|---|
| ||||||
| 2005 | 27.9 | 98.1 | 7.8 | |||
| Cases | 562 | 320 (56.9) | ||||
| Controls | 13,223 | 12,394 (93.7) | ||||
| 2006 | 179 230 | 28.7 | 97.7 | 8.4 | ||
| Cases | 635 | 347 (54.7) | ||||
| Controls | 13,102 | 12,241 (93.4) | ||||
| 2007 | 192 210 | 28.3 | 97.6 | 8.8 | ||
| Cases | 667 | 367 (55.0) | ||||
| Controls | 13,270 | 12,341 (93.0) | ||||
The resulting classification model had a low total misclassification rate for the 2005 data. Of the 562 septic patients, 320 (56.9%) were correctly classified, and 12,394 (93.7%) of the control patients were appropriately identified. The number of septic and control patients misclassified was 242 and 829, respectively, yielding a total misclassification rate of 7.8%. When applied to the 2006 patient data set, 347 (54.7%) of the 635 septic shock patients were correctly identified, while 12,241 (93.4%) of the 13,102 control patients were correctly classified. The total misclassification rate for the 2006 patient set was 8.4%. For the 2007 patient data, 367 (55.0%) of the 667 case patients were correctly identified, and 12,341 (93.0%) of the 13,270 control patients were correctly identified. This resulted in a total misclassification rate of 8.8%.
The 2006 and 2007 case patients were identified 179 230 minutes and 192 210 minutes before ICU transfer, respectively (Figure 2). The algorithm demonstrated positive and negative predictive values of 28.7% and 97.7% for the 2006 patient set, respectively, and 28.3% and 97.6% for the 2007 patient set, respectively.

Although the prediction algorithm shown in Figure 1 identified the majority of the case patients with ample time for clinical intervention prior to ICU transfer, the analysis used to derive this model included values for the arterial blood gas (ABG). As this is not a routinely obtained study for hospitalized patients outside of an ICU, it is possible that the performance of this model can in part be attributed to clinical acumen rather than changes in patient physiology. The ABG would likely only be obtained in patients with a more concerning or deteriorating clinical course, and thus more likely to develop shock. To address this possibility, a second analysis was performed that did not include the values for the ABG. The result was an algorithm with 13 classification splits, as shown in Figure 3.

The most predictive clinical variables in this analysis included the shock index (heart rate divided by systolic blood pressure), mean arterial pressure, total bilirubin, international normalized ratio (INR), total white blood cell count, absolute neutrophil count, albumin, hemoglobin, and sodium. This model was again applied to the 2006 and 2007 patient data sets (Table 3).
| Total Number | Number Correctly Classified (%) | Case Identification Time Before ICU Admission (minutes) | PPV (%) | NPV (%) | MCR (%) | |
|---|---|---|---|---|---|---|
| ||||||
| 2005 | 20.5 | 96.7 | 6.7 | |||
| Cases | 562 | 126 (22.4) | ||||
| Controls | 13,223 | 12,735 (96.3) | ||||
| 2006 | 508 536 | 21.4 | 96.1 | 7.0 | ||
| Cases | 635 | 121 (19.1) | ||||
| Controls | 13,102 | 12,657 (96.6) | ||||
| 2007 | 496 512 | 19.5 | 95.8 | 7.1 | ||
| Cases | 667 | 102 (15.3) | ||||
| Controls | 13,270 | 12,850 (96.8) | ||||
The overall misclassification rates for 2006 and 2007 were 7.0% and 7.1%, respectively. The model correctly identified 121 (19.1%) of the 635 cases and 12,657 (96.6%) of the 13,102 control patients from 2006, and 102 (15.3%) of the 667 cases and 12,850 (96.8%) of the 13,270 control patients from 2007. The respective positive and negative predictive values were 21.4% and 96.1% for 2006, respectively, and 19.5% and 95.8% for 2007, respectively.
Although the overall performance of the model derived without the ABG data was not as good, the identification times prior to ICU transfer were significantly improved. For the 2006 data, patients were identified 508 536 minutes before transfer (Figure 4), compared to 179 230 minutes for the model that included the ABG data (P < 0.01). For the 2007 data, patients were identified 496 512 minutes prior to ICU admission (Figure 4), compared to 192 210 minutes for the previous model (P < 0.01).

Discussion
We have demonstrated a simple method for generating an algorithm derived from routine laboratory and hemodynamic values that is capable of predicting the onset of sepsis in a significant proportion of non‐ICU patients. Two prediction models were generated, 1 with and 1 without ABG data included in the analysis. In the 2006 and 2007 validation cohorts, the model including these data correctly classified 54.7% and 55.0% of the patients who developed septic shock and 93.4% and 93.0% of control patients, respectively. The second model identified 19.1% and 15.3% of the septic shock patients and 96.6% and 96.8% of the control patients for 2006 and 2007, respectively. The methods used in generating this model are relatively simple and can be executed with the use of an electronic medical record system.
Early, goal‐directed cardiovascular resuscitation and adequate initial antibiotic therapy have been shown to decrease mortality in patients with severe sepsis and septic shock.2, 26 Prior studies employing early, targeted resuscitation strategies have demonstrated decreased use of vasopressors10 and decreased mortality.510 In addition, we previously demonstrated that a standardized order set for the management of severe sepsis in the emergency department that focused on early and aggressive intervention was associated with decreased 28‐day mortality.1 These studies suggest that early, aggressive management of septic shock can improve outcomes. Identification of patients prior to overt clinical deterioration may allow for early intervention aimed at preventing shock or improving its outcome.
The purpose of this method is to develop a model capable of recognizing patterns in clinical data that herald a patient's otherwise unidentified clinical deterioration. It is not intended to replace existing outcome prediction tools or severity of illness scoring systems, where a high degree of accuracy would be required. Rather, it would be best implemented as an automated screening tool incorporated into an electronic medical record system. When a hospitalized patient is identified as a possible septic shock patient by the classification tree, a notification is then issued to the clinicians caring for the patient. The primary goal of this method is to notify clinicians of potential clinical deterioration. Any action taken as a result of this notification is at the discretion of the clinician. This method could be employed for any population of hospitalized patients, though because of variations in clinical practice and patient physiology, different models would need to be generated for differing patient populations.
This method has limitations, the foremost of which is the possible instability of the resulting classification model. This type of analysis results in an algorithm that depends on binary splits to classify patients. In generating the algorithm, the recursive partitioning analysis selects the variables and cutoff values that result in the strongest decision tree with the most pure classifications at the end nodes. These variables and cutoff values may not immediately seem logical from a clinical standpoint, and may vary with changes in practice and even possibly between divisions within a hospital. As a result, the algorithm would likely require intermittent updating to remain effective and a model derived from 1 hospital or patient population would not necessarily be applicable to patients at another institution or from a different population. However, once the method has been developed at an institution, the process of revising the algorithm could be essentially automated and uses few resources.
Another shortcoming of this method is the relatively low sensitivity of the resulting algorithm. In a role as an automated alert system, a low false‐positive rate is particularly desirable to avoid unnecessary frequent distraction of clinicians. The sensitivity of the model can be improved through manipulation of how the analysis is performed, but this would be at the expense of a higher false‐positive rate, which is not acceptable. Finally, prior studies examining treatment for sepsis have demonstrated an advantage to early and aggressive therapy. It is not clear, however, if identifying these patients prior to the onset of clinically evident sepsis would result in improved outcomes. Further work is required to determine if this is the case. We are currently conducting a prospective study that employs the method described here in conjunction with an automated alert system to ascertain if it impacts outcomes on patients admitted to the medicine wards of Barnes‐Jewish Hospital.
In conclusion, the method presented here represents a technique that consumes few resources and is capable of identifying some patients before septic shock becomes clinically evident. When applied in an automated fashion with the capability to alert clinicians caring for a patient, the method demonstrated here may allow for earlier diagnosis and possibly intervention for septic shock patients.
- ,,, et al.Before–after study of a standardized hospital order set for the management of septic shock.Crit Care Med.2006;34(11):2707–2713.
- ,,, et al.Early goal‐directed therapy in the treatment of severe sepsis and septic shock.N Engl J Med.2001;345:1368–1377.
- ,,, et al.Early goal‐directed therapy in severe sepsis and septic shock revisited; concepts, controversies, and contemporary findings.Chest.2006;130(5):1579–1595.
- .Implementing the severe sepsis care bundles outside the ICU by outreach.Nurs Crit Care.2007;12:225–229.
- ,,, et al.The impact of compliance with 6‐hour and 24‐hour sepsis bundles on hospital mortality in patients with severe sepsis: a prospective observational study.Crit Care.2005;9:R764–R770.
- ,,, et al.Translating research to clinical practice, a 1‐year experience with implementing early goal‐directed therapy for septic shock in the emergency department.Chest.2006;129:225–232.
- ,,, et al.Prospective external validation of the clinical effectiveness of an emergency department‐based early goal‐directed therapy protocol for severe sepsis and septic shock.Chest.2007;132:425–432.
- ,,, et al.Implementation of a bundle of quality indicators for the early management of severe sepsis and septic shock is associated with decreased mortality.Crit Care Med.2007;35:1105–1112.
- ,,, et al.Implementation of an evidence‐based “standard operating procedure” and outcome in septic shock.Crit Care Med.2006;34:943–949.
- ,,, et al.Outcome of septic shock in older adults after implementation of the sepsis “bundle”.J Am Geriatr Soc.2008;56:272–278.
- ,,, et al.Confidential inquiry into quality of care before admission to intensive care.BMJ.1998;316:1853–1858.
- ,,.Unexpected deaths and referrals to intensive care of patients on general wards. Are some cases potentially avoidable?J R Coll Physicians Lond.1999;33(3):255–259.
- ,,, et al.Septic shock: an analysis of outcomes for patients with onset on hospital wards versus intensive care units.Crit Care Med.1998;26(6):1020–1024.
- ,,,,,.Hospital‐wide impact of a standardized order set for the management of bacteremic severe sepsis.Crit Care Med.2009;37(3):819–824.
- ,,,,,.The mRNA expression of fatty acid amide hydrolase in human whole blood correlates with sepsis.J Endotoxin Res.2007;13(1):35–38.
- ,,, et al.C‐reactive protein use as an early indicator of infection in patients with systemic inflammatory response syndrome.Intensive Care Med.2004;30(11):2038–2045.
- ,,,,.Procalcitonin as a screening test of late‐onset sepsis in preterm very low birth weight infants.J Perinatol.2005;25(6):397–402.
- ,,.A simple method for predicting severe sepsis in burn patients.Am J Surg.1980;139(4):513–517.
- ,,, et al.Prognostic value of protein C concentrations in neutropenic patients at high risk of severe septic complications.Crit Care Med.2000;28(7):2209–2216.
- ,,,,.Circulating immune parameters predicting the progression from hospital‐acquired pneumonia to septic shock in surgical patients.Crit Care Med.2005;9(6):R662–R669.
- ,,.A simple prediction algorithm for bacteremia in patients with acute febrile illness.Q J Med.2005;98:813–820.
- ,,,.Infection probability score (IPS): a method to help assess the probability of infection in critically ill patients.Crit Care Med.2003;31(11):2579–2584.
- ,.Multivariate regression modeling for the prediction of inflammation, systemic pressure, and end‐organ function in severe sepsis.Shock.1997;8(3):225–231.
- ,,,,,.Abnormal heart rate characteristics preceding neonatal sepsis and sepsis‐like illness.Pediatr Res.2003;53:920–926.
- ,.Statistics for Biology and Health.New York:Springer‐Verlag;1999.
- ,,, et al.Impact of adequate empiric antibiotic therapy on the outcome of patients admitted to the intensive care unit with sepsis.Crit Care Med.2003;31:2742–2751.
- ,,, et al.Before–after study of a standardized hospital order set for the management of septic shock.Crit Care Med.2006;34(11):2707–2713.
- ,,, et al.Early goal‐directed therapy in the treatment of severe sepsis and septic shock.N Engl J Med.2001;345:1368–1377.
- ,,, et al.Early goal‐directed therapy in severe sepsis and septic shock revisited; concepts, controversies, and contemporary findings.Chest.2006;130(5):1579–1595.
- .Implementing the severe sepsis care bundles outside the ICU by outreach.Nurs Crit Care.2007;12:225–229.
- ,,, et al.The impact of compliance with 6‐hour and 24‐hour sepsis bundles on hospital mortality in patients with severe sepsis: a prospective observational study.Crit Care.2005;9:R764–R770.
- ,,, et al.Translating research to clinical practice, a 1‐year experience with implementing early goal‐directed therapy for septic shock in the emergency department.Chest.2006;129:225–232.
- ,,, et al.Prospective external validation of the clinical effectiveness of an emergency department‐based early goal‐directed therapy protocol for severe sepsis and septic shock.Chest.2007;132:425–432.
- ,,, et al.Implementation of a bundle of quality indicators for the early management of severe sepsis and septic shock is associated with decreased mortality.Crit Care Med.2007;35:1105–1112.
- ,,, et al.Implementation of an evidence‐based “standard operating procedure” and outcome in septic shock.Crit Care Med.2006;34:943–949.
- ,,, et al.Outcome of septic shock in older adults after implementation of the sepsis “bundle”.J Am Geriatr Soc.2008;56:272–278.
- ,,, et al.Confidential inquiry into quality of care before admission to intensive care.BMJ.1998;316:1853–1858.
- ,,.Unexpected deaths and referrals to intensive care of patients on general wards. Are some cases potentially avoidable?J R Coll Physicians Lond.1999;33(3):255–259.
- ,,, et al.Septic shock: an analysis of outcomes for patients with onset on hospital wards versus intensive care units.Crit Care Med.1998;26(6):1020–1024.
- ,,,,,.Hospital‐wide impact of a standardized order set for the management of bacteremic severe sepsis.Crit Care Med.2009;37(3):819–824.
- ,,,,,.The mRNA expression of fatty acid amide hydrolase in human whole blood correlates with sepsis.J Endotoxin Res.2007;13(1):35–38.
- ,,, et al.C‐reactive protein use as an early indicator of infection in patients with systemic inflammatory response syndrome.Intensive Care Med.2004;30(11):2038–2045.
- ,,,,.Procalcitonin as a screening test of late‐onset sepsis in preterm very low birth weight infants.J Perinatol.2005;25(6):397–402.
- ,,.A simple method for predicting severe sepsis in burn patients.Am J Surg.1980;139(4):513–517.
- ,,, et al.Prognostic value of protein C concentrations in neutropenic patients at high risk of severe septic complications.Crit Care Med.2000;28(7):2209–2216.
- ,,,,.Circulating immune parameters predicting the progression from hospital‐acquired pneumonia to septic shock in surgical patients.Crit Care Med.2005;9(6):R662–R669.
- ,,.A simple prediction algorithm for bacteremia in patients with acute febrile illness.Q J Med.2005;98:813–820.
- ,,,.Infection probability score (IPS): a method to help assess the probability of infection in critically ill patients.Crit Care Med.2003;31(11):2579–2584.
- ,.Multivariate regression modeling for the prediction of inflammation, systemic pressure, and end‐organ function in severe sepsis.Shock.1997;8(3):225–231.
- ,,,,,.Abnormal heart rate characteristics preceding neonatal sepsis and sepsis‐like illness.Pediatr Res.2003;53:920–926.
- ,.Statistics for Biology and Health.New York:Springer‐Verlag;1999.
- ,,, et al.Impact of adequate empiric antibiotic therapy on the outcome of patients admitted to the intensive care unit with sepsis.Crit Care Med.2003;31:2742–2751.
Copyright © 2010 Society of Hospital Medicine
In response to: A quality conundrum: Well done but not enough—Quality improvement conundrums: Looking back before moving forward
If clinician‐quality improvers are to gain traction as academicians,1 their first objective should be to bring quality improvement (QI) sandly into the world of scientific method. We believe that Dr. Chakraborti's 2 pointsthat the reasons for afferent limb failure need to be more closely investigated, and that lessons learned from 1 hospital's rapid response system (RRS) may not generalize to other hospitalsreflect the immaturity of QI as a science. In clinical science, 3 well‐defined testing phases bring 1 homogeneous, rigorously tested product to market that is monitored in a fourth phase. While Dr. Chakraborti urges us to examine our afferent limb failures more closely, the monitoring and reporting strategies used in the Josie King Patient Safety Program2 resonate with the postmarketing surveillance of Phase IV trials.
Although necessary and valid, we believe that the majority of the QI conundrum of RRS lies in the lack of premarket, stepwise testing of QI products. QI initiatives are often promulgated before an appropriate evidence base has been established. This lack of scientific rigor has resulted in RRS with calling criteria that have poor operating characteristics,3 undetermined methods for achieving afferent success,4 and efferent response arms of varying sizes and compositions.5 Consequently, a heterogeneous group of RRS have produced equivocal outcomes6 and diminished the applicability of lessons learned across institutions.
Indeed, while it is important to ask, What do we do now?, it may be more informative to answer the question, How did we get here?
- ,.Clinicians in quality improvement. A new career pathway in academic medicine.JAMA.2009;301(7):766–768.
- Josie King Foundation. Josie King Patient Safety Program. Available at: http://www.josieking.org/page.cfm?pageID=27. Accessed September2009.
- ,,,.Rapid response: a quality improvement conundrum.J Hosp Med.2009;4(4):255–257.
- .The rapid response team paradox: why doesn't anyone call for help?Crit Care Med.2008;36(2):634–636.
- ,,,.Dress for the occasion.Jt Comm J Qual Patient Saf.2009;35(6):295.
- ,,,,.Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis.J Hosp Med.2007;2(6):422–432.
If clinician‐quality improvers are to gain traction as academicians,1 their first objective should be to bring quality improvement (QI) sandly into the world of scientific method. We believe that Dr. Chakraborti's 2 pointsthat the reasons for afferent limb failure need to be more closely investigated, and that lessons learned from 1 hospital's rapid response system (RRS) may not generalize to other hospitalsreflect the immaturity of QI as a science. In clinical science, 3 well‐defined testing phases bring 1 homogeneous, rigorously tested product to market that is monitored in a fourth phase. While Dr. Chakraborti urges us to examine our afferent limb failures more closely, the monitoring and reporting strategies used in the Josie King Patient Safety Program2 resonate with the postmarketing surveillance of Phase IV trials.
Although necessary and valid, we believe that the majority of the QI conundrum of RRS lies in the lack of premarket, stepwise testing of QI products. QI initiatives are often promulgated before an appropriate evidence base has been established. This lack of scientific rigor has resulted in RRS with calling criteria that have poor operating characteristics,3 undetermined methods for achieving afferent success,4 and efferent response arms of varying sizes and compositions.5 Consequently, a heterogeneous group of RRS have produced equivocal outcomes6 and diminished the applicability of lessons learned across institutions.
Indeed, while it is important to ask, What do we do now?, it may be more informative to answer the question, How did we get here?
If clinician‐quality improvers are to gain traction as academicians,1 their first objective should be to bring quality improvement (QI) sandly into the world of scientific method. We believe that Dr. Chakraborti's 2 pointsthat the reasons for afferent limb failure need to be more closely investigated, and that lessons learned from 1 hospital's rapid response system (RRS) may not generalize to other hospitalsreflect the immaturity of QI as a science. In clinical science, 3 well‐defined testing phases bring 1 homogeneous, rigorously tested product to market that is monitored in a fourth phase. While Dr. Chakraborti urges us to examine our afferent limb failures more closely, the monitoring and reporting strategies used in the Josie King Patient Safety Program2 resonate with the postmarketing surveillance of Phase IV trials.
Although necessary and valid, we believe that the majority of the QI conundrum of RRS lies in the lack of premarket, stepwise testing of QI products. QI initiatives are often promulgated before an appropriate evidence base has been established. This lack of scientific rigor has resulted in RRS with calling criteria that have poor operating characteristics,3 undetermined methods for achieving afferent success,4 and efferent response arms of varying sizes and compositions.5 Consequently, a heterogeneous group of RRS have produced equivocal outcomes6 and diminished the applicability of lessons learned across institutions.
Indeed, while it is important to ask, What do we do now?, it may be more informative to answer the question, How did we get here?
- ,.Clinicians in quality improvement. A new career pathway in academic medicine.JAMA.2009;301(7):766–768.
- Josie King Foundation. Josie King Patient Safety Program. Available at: http://www.josieking.org/page.cfm?pageID=27. Accessed September2009.
- ,,,.Rapid response: a quality improvement conundrum.J Hosp Med.2009;4(4):255–257.
- .The rapid response team paradox: why doesn't anyone call for help?Crit Care Med.2008;36(2):634–636.
- ,,,.Dress for the occasion.Jt Comm J Qual Patient Saf.2009;35(6):295.
- ,,,,.Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis.J Hosp Med.2007;2(6):422–432.
- ,.Clinicians in quality improvement. A new career pathway in academic medicine.JAMA.2009;301(7):766–768.
- Josie King Foundation. Josie King Patient Safety Program. Available at: http://www.josieking.org/page.cfm?pageID=27. Accessed September2009.
- ,,,.Rapid response: a quality improvement conundrum.J Hosp Med.2009;4(4):255–257.
- .The rapid response team paradox: why doesn't anyone call for help?Crit Care Med.2008;36(2):634–636.
- ,,,.Dress for the occasion.Jt Comm J Qual Patient Saf.2009;35(6):295.
- ,,,,.Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis.J Hosp Med.2007;2(6):422–432.
Activities of Daily Living
Tears well up in my eyes. Not from pain, but frustration.
I pleaded with my 3‐year‐olds to come to me, so I could help them wash and dress for the day. Ordinarily, I would have come into their room, leaned over their beds, and whispered good morning into their ears.
Four days after surgery, I wasn't able to do that. Not without the crutches. Even with the crutches, I was moving slowly. I scooted up, put a few pillows behind my back, and carefully lifted up my leg. Putting on the knee immobilizer would take too long; I would only be crossing the hall.
Weighing less than 800 g, the immobilizer is an adjustable aluminum frame attached to foam rubber and 4 wide Velcro straps. It is a device of torment. I can never seem to find the proper fit. If it is too tight, my leg hurts and starts turning blue. When it is too loose, it pulls on my incision, multiplying the pain. You put it on while sitting, but you only discover whether it is too tight or too loose when you stand up.
With one foot on the floor at the edge of the bed, I took one crutch in hand, shifted to the left, and grabbed for the other crutch. I squeezed the handgrips of the crutches tightly and pulled myself up.
Put your weight on your hands, NOT your armpits, the instructions said. Easier said than done, because my armpits were now sore too.
Keep your crutches even with each other. I tried to remember to make an equilateral triangle with my good foot and 2 crutches, but the instructions only seemed to account for movement in a straight line.
Keep your elbows slightly bent and close to your sides to help keep the crutches under your arm. I am trying that too.
Lock your elbows. This instruction contradicts the previous one. Which should I follow?
Place your crutches 2 to 3 inches outside of each foot. How do I do this and keep my elbows close to my sides?
Swing your injured leg through first. But not too much, or I'll pole vault across the room.
After 3 steps, my leg started to throb, and my quadriceps went into a spasm. I needed to sit down, immediately.
Non‐weight‐bearing for next 4 weeks, was written on the doctor's note. Four weeks feels like an eternity. If my heel or toe even touched the ground, I felt an immediate throb of pain in my knee.
How do my orthopedic patients do it: those with broken hips and bad or broken knees, with hip replacements, or knee replacements? I was sensitive to their pain and could optimize control. Postoperatively, I could support gastrointestinal and other organ systems. I made sure that the basic weight‐bearing order was correct. I carefully followed the physical and/or occupational therapy recommendation for home, rehabilitation, or a nursing home. I spoke with families. Yet I did not dwell on activities of daily living nor how my patients felt to be dependent on others for simple things they needed. Some patients were non‐weight‐bearing for weeks, some for months, others never walked again. How can one deeply understand it if one has never experienced it?
According to Centers for Disease Control and Prevention (CDC) statistics,1 unintentional injuries are the leading cause of death between the ages of 1 and 44 years. Accidents are again the leading cause of death after age 75 years. More startling is the fact that these numbers have not been significantly reduced since these data were first collected. Nor do these numbers reveal the number of those debilitated, but still living. In my case, I was not playing basketball, water skiing, or rock climbing: I slipped and fell while stepping into a wading pool with my children, severing ligaments, tearing meniscus, and creating a hairline fracture of my tibia.
Expect to move slowly with the crutches. Yes, I am moving very slowly, only 1 to 2 feet with each stride. If I take a longer stride, I lose my balance.
Learn to sit down with the crutches. Learn to stand up. Learn to get into a car.
Learn to go up the stairs. Learn to go down the stairs.
Avoid wet surfaces. Otherwise you'll start skating and reinjure that knee.
It was effortless to think of patients and colleagues of mine. Mr. S., how do you do it? At age 30 years, he was quadriplegic from falling from a tree as a teenager. Mr. D, how about you, in a wheelchair, after being hit by a car and multiple postoperative leg infections and amputations: living in hotels, with estranged family and no social support. How did you do it, while we nagged you about controlling your glucose. Dr. J? The sadness fills my heart. At only 60 years old, my wonderful professor now made rounds in an electric wheelchair, the victim of amyotrophic lateral sclerosis (ALS). My fate, in comparison, is fortunate; my immobility only temporary. I can still think. And talk. And use a phone. And eat, as well as write this essay. And, best of all, read stories to my children. Be careful about this leg, I remind them. Don't come too close!
Maybe what life is trying to tell you is, slow down. In fact, it's forcing you to do that, a colleague and friend said. I had prided myself in thorough and compassionate doctoring. Now, having been a patient on crutches, I have a greater understanding of how limits to mobility can impact daily living on my patients after I discharge them. And the mundane subject of accident prevention has gained a new urgency.
- ,,,.Deaths: final data for 2005.Natl Vital Stat Rep.2008;56(10):1–120.
Tears well up in my eyes. Not from pain, but frustration.
I pleaded with my 3‐year‐olds to come to me, so I could help them wash and dress for the day. Ordinarily, I would have come into their room, leaned over their beds, and whispered good morning into their ears.
Four days after surgery, I wasn't able to do that. Not without the crutches. Even with the crutches, I was moving slowly. I scooted up, put a few pillows behind my back, and carefully lifted up my leg. Putting on the knee immobilizer would take too long; I would only be crossing the hall.
Weighing less than 800 g, the immobilizer is an adjustable aluminum frame attached to foam rubber and 4 wide Velcro straps. It is a device of torment. I can never seem to find the proper fit. If it is too tight, my leg hurts and starts turning blue. When it is too loose, it pulls on my incision, multiplying the pain. You put it on while sitting, but you only discover whether it is too tight or too loose when you stand up.
With one foot on the floor at the edge of the bed, I took one crutch in hand, shifted to the left, and grabbed for the other crutch. I squeezed the handgrips of the crutches tightly and pulled myself up.
Put your weight on your hands, NOT your armpits, the instructions said. Easier said than done, because my armpits were now sore too.
Keep your crutches even with each other. I tried to remember to make an equilateral triangle with my good foot and 2 crutches, but the instructions only seemed to account for movement in a straight line.
Keep your elbows slightly bent and close to your sides to help keep the crutches under your arm. I am trying that too.
Lock your elbows. This instruction contradicts the previous one. Which should I follow?
Place your crutches 2 to 3 inches outside of each foot. How do I do this and keep my elbows close to my sides?
Swing your injured leg through first. But not too much, or I'll pole vault across the room.
After 3 steps, my leg started to throb, and my quadriceps went into a spasm. I needed to sit down, immediately.
Non‐weight‐bearing for next 4 weeks, was written on the doctor's note. Four weeks feels like an eternity. If my heel or toe even touched the ground, I felt an immediate throb of pain in my knee.
How do my orthopedic patients do it: those with broken hips and bad or broken knees, with hip replacements, or knee replacements? I was sensitive to their pain and could optimize control. Postoperatively, I could support gastrointestinal and other organ systems. I made sure that the basic weight‐bearing order was correct. I carefully followed the physical and/or occupational therapy recommendation for home, rehabilitation, or a nursing home. I spoke with families. Yet I did not dwell on activities of daily living nor how my patients felt to be dependent on others for simple things they needed. Some patients were non‐weight‐bearing for weeks, some for months, others never walked again. How can one deeply understand it if one has never experienced it?
According to Centers for Disease Control and Prevention (CDC) statistics,1 unintentional injuries are the leading cause of death between the ages of 1 and 44 years. Accidents are again the leading cause of death after age 75 years. More startling is the fact that these numbers have not been significantly reduced since these data were first collected. Nor do these numbers reveal the number of those debilitated, but still living. In my case, I was not playing basketball, water skiing, or rock climbing: I slipped and fell while stepping into a wading pool with my children, severing ligaments, tearing meniscus, and creating a hairline fracture of my tibia.
Expect to move slowly with the crutches. Yes, I am moving very slowly, only 1 to 2 feet with each stride. If I take a longer stride, I lose my balance.
Learn to sit down with the crutches. Learn to stand up. Learn to get into a car.
Learn to go up the stairs. Learn to go down the stairs.
Avoid wet surfaces. Otherwise you'll start skating and reinjure that knee.
It was effortless to think of patients and colleagues of mine. Mr. S., how do you do it? At age 30 years, he was quadriplegic from falling from a tree as a teenager. Mr. D, how about you, in a wheelchair, after being hit by a car and multiple postoperative leg infections and amputations: living in hotels, with estranged family and no social support. How did you do it, while we nagged you about controlling your glucose. Dr. J? The sadness fills my heart. At only 60 years old, my wonderful professor now made rounds in an electric wheelchair, the victim of amyotrophic lateral sclerosis (ALS). My fate, in comparison, is fortunate; my immobility only temporary. I can still think. And talk. And use a phone. And eat, as well as write this essay. And, best of all, read stories to my children. Be careful about this leg, I remind them. Don't come too close!
Maybe what life is trying to tell you is, slow down. In fact, it's forcing you to do that, a colleague and friend said. I had prided myself in thorough and compassionate doctoring. Now, having been a patient on crutches, I have a greater understanding of how limits to mobility can impact daily living on my patients after I discharge them. And the mundane subject of accident prevention has gained a new urgency.
Tears well up in my eyes. Not from pain, but frustration.
I pleaded with my 3‐year‐olds to come to me, so I could help them wash and dress for the day. Ordinarily, I would have come into their room, leaned over their beds, and whispered good morning into their ears.
Four days after surgery, I wasn't able to do that. Not without the crutches. Even with the crutches, I was moving slowly. I scooted up, put a few pillows behind my back, and carefully lifted up my leg. Putting on the knee immobilizer would take too long; I would only be crossing the hall.
Weighing less than 800 g, the immobilizer is an adjustable aluminum frame attached to foam rubber and 4 wide Velcro straps. It is a device of torment. I can never seem to find the proper fit. If it is too tight, my leg hurts and starts turning blue. When it is too loose, it pulls on my incision, multiplying the pain. You put it on while sitting, but you only discover whether it is too tight or too loose when you stand up.
With one foot on the floor at the edge of the bed, I took one crutch in hand, shifted to the left, and grabbed for the other crutch. I squeezed the handgrips of the crutches tightly and pulled myself up.
Put your weight on your hands, NOT your armpits, the instructions said. Easier said than done, because my armpits were now sore too.
Keep your crutches even with each other. I tried to remember to make an equilateral triangle with my good foot and 2 crutches, but the instructions only seemed to account for movement in a straight line.
Keep your elbows slightly bent and close to your sides to help keep the crutches under your arm. I am trying that too.
Lock your elbows. This instruction contradicts the previous one. Which should I follow?
Place your crutches 2 to 3 inches outside of each foot. How do I do this and keep my elbows close to my sides?
Swing your injured leg through first. But not too much, or I'll pole vault across the room.
After 3 steps, my leg started to throb, and my quadriceps went into a spasm. I needed to sit down, immediately.
Non‐weight‐bearing for next 4 weeks, was written on the doctor's note. Four weeks feels like an eternity. If my heel or toe even touched the ground, I felt an immediate throb of pain in my knee.
How do my orthopedic patients do it: those with broken hips and bad or broken knees, with hip replacements, or knee replacements? I was sensitive to their pain and could optimize control. Postoperatively, I could support gastrointestinal and other organ systems. I made sure that the basic weight‐bearing order was correct. I carefully followed the physical and/or occupational therapy recommendation for home, rehabilitation, or a nursing home. I spoke with families. Yet I did not dwell on activities of daily living nor how my patients felt to be dependent on others for simple things they needed. Some patients were non‐weight‐bearing for weeks, some for months, others never walked again. How can one deeply understand it if one has never experienced it?
According to Centers for Disease Control and Prevention (CDC) statistics,1 unintentional injuries are the leading cause of death between the ages of 1 and 44 years. Accidents are again the leading cause of death after age 75 years. More startling is the fact that these numbers have not been significantly reduced since these data were first collected. Nor do these numbers reveal the number of those debilitated, but still living. In my case, I was not playing basketball, water skiing, or rock climbing: I slipped and fell while stepping into a wading pool with my children, severing ligaments, tearing meniscus, and creating a hairline fracture of my tibia.
Expect to move slowly with the crutches. Yes, I am moving very slowly, only 1 to 2 feet with each stride. If I take a longer stride, I lose my balance.
Learn to sit down with the crutches. Learn to stand up. Learn to get into a car.
Learn to go up the stairs. Learn to go down the stairs.
Avoid wet surfaces. Otherwise you'll start skating and reinjure that knee.
It was effortless to think of patients and colleagues of mine. Mr. S., how do you do it? At age 30 years, he was quadriplegic from falling from a tree as a teenager. Mr. D, how about you, in a wheelchair, after being hit by a car and multiple postoperative leg infections and amputations: living in hotels, with estranged family and no social support. How did you do it, while we nagged you about controlling your glucose. Dr. J? The sadness fills my heart. At only 60 years old, my wonderful professor now made rounds in an electric wheelchair, the victim of amyotrophic lateral sclerosis (ALS). My fate, in comparison, is fortunate; my immobility only temporary. I can still think. And talk. And use a phone. And eat, as well as write this essay. And, best of all, read stories to my children. Be careful about this leg, I remind them. Don't come too close!
Maybe what life is trying to tell you is, slow down. In fact, it's forcing you to do that, a colleague and friend said. I had prided myself in thorough and compassionate doctoring. Now, having been a patient on crutches, I have a greater understanding of how limits to mobility can impact daily living on my patients after I discharge them. And the mundane subject of accident prevention has gained a new urgency.
- ,,,.Deaths: final data for 2005.Natl Vital Stat Rep.2008;56(10):1–120.
- ,,,.Deaths: final data for 2005.Natl Vital Stat Rep.2008;56(10):1–120.
IVIG for Severe Colitis
Clostridium difficile colitis (CDC) is the most common cause of hospital‐acquired diarrhea.1 The incidence of CDC has sharply increased over the past decade despite increasing awareness among health care professionals.24 C. difficile pathogenic strains induce diarrhea through the elaboration and secretion of 2 exotoxins: toxin A and toxin B. Toxin A is an inflammatory toxin, leading to fluid secretion, increased mucosal permeability, and marked enteritis and colitis.5 Toxin B is cytotoxic, leading to cell injury and apoptosis.5 Combined, toxin A and toxin B can cause a wide spectrum of clinical presentations, ranging from mild diarrhea that resolves with the discontinuation of antibiotics to a fulminant colitis requiring surgical intervention.
The severity of clinical manifestations has been shown to be inversely proportional to the host anti‐toxin A antibody level in response to toxin exposure. Kyne et al.6 demonstrated that asymptomatic C. difficile carriers produced significantly higher anti‐toxin A immunoglobulin G (IgG) levels compared to symptomatic patients. Among the latter group, patients with mild disease had a higher antibody level compared to those with severe colitis.6, 7 Additional risk factors predisposing to severe colitis are advanced age, severe underlying illness,8 and immunocompromised state.9
Recently, a new C. difficile strain (BI/NAP1) with a mutated toxin A and toxin B promoter silencer, a binary toxin gene, and fluoroquinolone resistance has been described in Canada and the United States.3, 10 This strain has been associated with an increased incidence of CDC among hospitalized patients, especially the incidence of severe disease requiring colectomy. At the same time, several reports describing metronidazole treatment failure have been published.1114 These recent findings emphasize the importance of finding alternative treatments for CDC.
Intravenous immunoglobulin (IVIG) was used to treat CDC for the first time in 1991.15 Since then, 12 case reports and small case series, along with 1 case‐control study have been published documenting IVIG treatment outcomes.1527 However, only 5 reports to date examined patients with severe CDC.2125 In the present study, we report the largest series of patients with severe CDC treated with IVIG in the literature to our knowledge.
Patients and Methods
Case Series
We used CareScience software (CareScience, Inc., Philadelphia, PA) to retrospectively identify all patients admitted to our institution with a primary or secondary diagnosis of CDC (code 00845) between July 1, 2002 and May 1, 2006. CareScience is commercially available software that tracks all admissions to our institution and allows the performance of patient searches with a wide spectrum of user‐defined search criteria. Using the same software, we further identified those patients who received IVIG during their hospital stay. We then obtained the hospital chart for each patient, from the medical records department and established the study database.
A case was defined as a patient with diarrhea (at least 3 loose stools daily) for at least 2 days who had C. difficile cytotoxin‐positive feces and at least 1 of the following criteria: clinical symptoms (abdominal pain and/or distension and fever); leukemoid reaction (defined as white blood cell count of 20,000 cells/mm3 or above. This cutoff value was chosen as it has been used previously as a prognostic factor)4; radiographic evidence of colitis by computed tomography (CT) of the abdomen; and/or the presence of pseudomembranes on flexible sigmoidoscopy or colonoscopy. We excluded all patients who received IVIG for an indication other than CDC treatment (n = 3). There were no other exclusion criteria.
We used a standardized data collection tool and recorded demographics (age, gender, principal diagnosis), past medical and surgical history, other risk factors for C. difficile infection (previous CDC, antibiotics received during hospital stay, immunosuppressive medications or organ transplantation within the previous 6 weeks, history of malignancy or diabetes mellitus); clinical presentation (abdominal distention, abdominal pain, diarrhea, fever, leukemoid reaction, and hypotension defined as systolic blood pressure <85 mm Hg despite at least 1 L of intravenous normal saline administration and the need for vasopressor use); colonoscopy findings; CT scan and x‐ray findings; laboratory values; date and dose of IVIG infused and other C. difficile pharmacological treatments; and Acute Physiological Assessment and Chronic Health Evaluation (APACHE II) score28 at the first day of IVIG infusion. The primary outcomes were survival at the end of the hospital stay and clinical disease resolution, defined as 2 formed bowel movements or less per day without abdominal pain or distention.
The decision to initiate IVIG therapy and the dose to be used was made by the individual attending physician.
Statistical Analysis
Single (univariate) and multiple (multivariate) logistic regression analysis were applied to identify variables among the ones collected that are independent predictors of CDC mortality. All statistical analyses were completed with the STATA 10 software package (StatCorp LP, College Station, TX).
Review of the Literature
We used PubMed, Web of Science, Scopus, and Excerpta Medica databases to search for any publication in a peer‐reviewed journal on the use of IVIG for the treatment of severe CDC. We used the search words: IVIG or intravenous immunoglobulin and clostridium difficile. Only publications published in English were selected. The date range used was January 1, 1950 to January 7, 2009. We were able to find 5 publications using this search criteria.
Results
Study Population
Of the 1230 patients diagnosed with CDC over the 4‐year study period, 21 patients were treated with IVIG. Table 1 summarizes the patients' characteristics. There were 13 women and 8 men. The mean age was 68 years, with a standard deviation (SD) of 13 years. Sepsis was the primary diagnosis in all patients. Sixteen patients had predisposing risk factors for CDC, including immunosuppression (immunosuppressive medication [n = 2], human immunodeficiency virus [HIV] infection [n = 2]); cancer (n = 3); recent surgery (n = 3); and diabetes mellitus (n = 11). Nine patients had documented previous CDC episodes. The indications for IVIG administration were evidence of pancolitis on abdominal CT scan (n = 12) or severe ileus with cessation of diarrhea, abdominal distention, and requirement for total parenteral nutrition (n = 5), or severe hypotension (n = 4) (defined as systolic blood pressure <85 mm Hg despite at least 1 L of intravenous normal saline administration and the subsequent need for vasopressor use).
| Patient | Age (gender) | Diagnosis | Medical and Surgical History | CDC History | Colonoscopy Findings | Radiographic Findings |
|---|---|---|---|---|---|---|
| ||||||
| A | 40 (female) | CDC with sec. sepsis | Gastric stapling | Yes | * | Diffuse colitis |
| B | 86 (female) | Fulminant CDC | Metastatic ovarian carcinoma | PC | No colonic thickening | |
| C | 72 (male) | Sepsis | Acute pancreatitis with sec. pseudocyst, DM | Yes | PC | Diffuse colitis |
| D | 78 (male) | Discitis | Delayed: normal mucosa | Dilation of small and large bowel | ||
| E | 98 (female) | Urosepsis | Yes | * | No bowel distention | |
| F | 90 (female) | Right lower extremity cellulitis | DM | * | Concentric thickening of rectal wall | |
| G | 64 (male) | Ischemic colitis | DM, recent Hartman pouch closure | Marked inflammation | Diffuse colitis | |
| H | 78 (female) | Toe osteomyelitis and CDC with sec. sepsis | DM | * | Diffuse colitis | |
| I | 35 (female) | Sepsis | Yes | * | Diffuse nonspecific colitis, minimal ascites | |
| J | 47 (female) | Pneumonia and sec. sepsis | * | Diffuse colitis | ||
| K | 56 (female) | Urosepsis | HIV | Yes | * | Colitis involving the right colon |
| L | 76 (male) | CDC with sec. sepsis | Sigmoid bladder fistula repair, DM | * | Diffuse colitis | |
| M | 71 (female) | Pneumonia with sec. sepsis | * | Diffuse colitis | ||
| N | 63 (male) | Urosepsis | DM, lymphoma resection from small intestine | PC | Marked small and large bowel distention | |
| O | 86 (male) | Enterococcus‐induced sepsis | Rheumatoid arthritis on methotrexate | * | Fat stranding suggesting peritonitis | |
| P | 60 (female) | Gastrointestinal bleed and CDC | DM with neuropathy and retinopathies | Yes | * | Thickening of wall of colon in most of the colon |
| Q | 57 (female) | Sepsis | DM | Yes | * | Normal |
| R | 67 (female) | CDC with sec. sepsis | Candidal esophagitis | Yes | * | Large amount of peritoneal fluid, mild small bowel thickening |
| S | 60 (female) | Sepsis sec. to S. aureus and P. aerogenosa | DM, renal transplant (myco, prednisone) | * | Ileus with air fluid level in the small intestine | |
| T | 80 (female) | Sepsis | DM | Yes | * | Thickening of descending colon consistent with colitis |
| U | 72 (male) | CDC, widespread metastatic cancer | DM, metastatic cancer (unknown primary site) | * | Severe colitis up to the splenic flexure | |
Table 2 describes disease severity in these patients. Since CDC starts locally in the colon then secondarily involves multiple organs as part of the systemic inflammatory response syndrome (SIRS), 2 scales were used to characterize the disease in each patient: (1) extent of local colonic inflammation, and (2) severity of systemic involvement. Extensive colonic involvement was evidenced in all patients by pancolitis on abdominal imaging modalities (12 patients), severe ileus requiring total parenteral nutrition (13 patients), or referral for surgical consultation for possible colectomy (12 patients).
| Patient | Complications During the Hospital Stay | APACHE II Score | Monitored Unit | WBC* | K | Alb | Lactate | Cr | Hospital Stay (days) | Surgical Consult/Surgery | TPN for Colitis |
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| A | Sepsis, DIC, ARDS, intubation | 15 | Yes | 48 | 4 | 1.1 | 2.9 | 1.3 | 26 | No/No | Yes |
| B | Dehydration, weakness | 12 | No | 19 | 3.4 | 3.1 | 1.1 | 16 | No/No | Yes | |
| C | HTP, GI bleed, ischemic colitis, F | 22 | Yes | 21 | 2.7 | 2.5 | 1.3 | ESRD | 34 | Yes/No | Yes |
| D | AD, megacolon, HT, intubation, ARF | 18 | Yes | 25 | 3.4 | 1.4 | 1.1 | 2.6 | 52 | Yes/No | Yes |
| E | Exacerbation of CHF with respiratory distress, Bipap support, confusion | 21 | No | 10 | 2.6 | 2.3 | 1 | 11 | No/No | No | |
| F | Confusion, gout acute attack | 21 | No | 33 | 3.1 | 2.6 | ESRD | 15 | No/No | No | |
| G | Intubation, cardiac arrest, AF with RVR, PNA, ARF, DVT, dysphagia, PEG | 6 | Yes | 15 | 3.1 | 1.7 | 1.9 | 2.2 | 32 | Yes/No | Yes |
| H | Sepsis, intubation, ARF, PEG, toe amputation, PNA, pulmonary edema, TPN, vitamin D deficiency | 34 | Yes | 59 | 2.8 | 1.5 | 4 | 3.8 | 17 | No/No | Yes |
| I | CHF, transient third cranial nerve palsy, DIC | 20 | Yes | 52 | 3.2 | 3.3 | 0.7 | ESRD | 11 | No/No | No |
| J | Aspiration PNA, sepsis, DIC, intubation, MI, F, HT | 32 | Yes | 17 | 2.7 | 1.8 | 14 | ESRD | 21 | Yes/no | No |
| K | Intubation, cardiac arrest, ARF, DIC, AP, F | 30 | Yes | 25 | 2.1 | 2.1 | 13 | 3.6 | 10 | Yes/no | No |
| L | Intubation, ARF, HTP, MI, AD, F | 23 | Yes | 69 | 3.2 | 1.8 | 1.4 | 3.6 | 23 | Yes/no | No |
| M | Intubation, DIC, ARF, GI bleeding, hypothermia, AD | 23 | Yes | 47 | 2.8 | 1.4 | 1.4 | 4.3 | 23 | Yes/no | Yes |
| N | Intubation, HTP, AD, F, ARF with HD, AF, osteomyelitis | 23 | Yes | 46 | 2.9 | 1.5 | 1.5 | 2.7 | 27 | Yes/no | Yes |
| O | Intubation, ARF, 2 cardiac arrests, GI bleed, UTI, rhabdomyolysis, liver shock, AF | 31 | Yes | 49 | 2.5 | 1.1 | 2.3 | 2.9 | 25 | Yes/no | Yes |
| P | Bowel ischemia with bowel resection, ARF, MI, ischemic bowel, fluid overload, respiratory failure | 23 | Yes | 26 | 2.6 | 0.8 | 2.9 | 3.1 | 9 | Yes/Yes | Yes |
| Q | Pulmonary embolism | 39 | Yes | 23 | 2.8 | 1.1 | 8.1 | ESRD | 9 | No/No | Yes |
| R | Fungal peritonitis, aspiration pneumonia, cardiac arrest | 26 | Yes | 30 | 4.1 | 1.1 | 0.9 | ESRD | 36 | No/No | Yes |
| S | Intubation, pneumothorax, CRT, pressor‐dependent shock, ARF | 36 | Yes | 46 | 3.1 | 1.8 | 2.1 | 5.4 | 64 | Yes/Yes | No |
| T | Pressor‐dependent sepsis, pulmonary edema, ARF with HD | 36 | Yes | 35 | 3.3 | 1.3 | 1.8 | 5.5 | 11 | Yes/No | Yes |
| U | Sepsis | 34 | Yes | 58 | 3.1 | 2.2 | 3.3 | 1.6 | 9 | No/No | No |
Of the 21 patients treated with IVIG, 9 did not receive a surgical consultation either because they responded to medical treatment promptly (6 patients), were too unstable for surgery (2 patients), or because the patient/family refused surgery (1 patient). Of the 12 patients who received surgical consultation, 2 underwent surgery. The remainder did not proceed to surgery for the following reasons: they were deemed medically unstable for surgery (6 patients), declined surgery (2 patients), were diagnosed with cancer on colonoscopy (1 patient), or improved with medical treatment (1 patient).
The severity of systemic involvement was measured using the APACHE II score on day 1 of IVIG infusion. The mean APACHE II score was 25. Eighteen patients were in a monitored unit when IVIG was administered. The study group had laboratory results in keeping with those previously used to define severe colitis:4, 9, 19 leukocytosis (defined as white blood cell count higher than 12,000 cells/mL [mean = 36,000 cells/mL]), hypoalbuminemia (mean = 1.78 g/dL, SD = 0.68 g/dL), hypokalemia (mean = 3.02 mg/dL, SD = 0.47 g/dL), and acute renal failure (defined as serum creatinine level >1.5 mg/dL [mean = 2.98 mg/dL, SD = 1.42 g/dL]).
IVIG Use
Table 3 describes the treatment patients received for CDC as well as the total number of antibiotics used throughout the hospital stay. IVIG was used as adjuvant treatment (defined as IVIG administration within 4 days or less after CDC diagnosis) in 8 patients and as second‐line treatment (defined as IVIG administration more than 4 days after CDC diagnosis) in 13 patients. Metronidazole, vancomycin, cholestyramine, and probiotic treatment alone or in different combinations were used for an average of 8 days (SD = 8 days; range, 025 days) before IVIG infusion. The total IVIG dose administered varied depending on the prescribing attending, with a range of 200 mg/kg to 1250 mg/kg and a mode of 250 mg/kg for 1 to 3 days. An average of 5 (SD = 2) different antibiotics that were not active against C. difficile were used per patient without being discontinued after a CDC diagnosis was made. The 3 most common were: cephalosporins (cefazolin, ceftriaxone, cefepime), fluoroquinolones (levofloxacin), and combination antibiotics (piperacillin and tazobactam or ampicillin and sulbactam).
| Patient | Number of Antibiotics | Duration of Treatment Before IVIG (days) | Treatment Before IVIG (days) | Total CDC Treatment (days) | IgG Level | IVIG Dose |
|---|---|---|---|---|---|---|
| ||||||
| A | 5 | 7 | Metro (7), Vanc (7), Choles (2) | Oral and rectal Vanc (26,19), IV Metro (19), Choles (2), Lacto (6) | Low | 300 mg/kg for 1 day |
| B | 1 | 13 | Metro (13), Vanc (13) | Oral Vanc (17) and IV Metro (12) | 300 mg/kg for 1 day | |
| C | 3 | 7 | Metro (7), Vanc (3) | IV Metro (28),Vanc oral and enema (18,3), Lacto (10) | Low | 125 mg/kg for 5 days |
| D | 5 | 25 | Metro (25), Vanc (15) | Oral then IV Metro (10,15), oral Vanc (25), Choles (7) and Lacto (13) | Low | 200 mg/kg for 1 day |
| E | 1 | 4 | Metro (1), Vanc (4), Choles(4) | IV Metro (8), oral Vanc (10) and Choles (4) | 75 mg/kg for 5 days | |
| F | 4 | 2 | Metro (2), Vanc (1) | IV Metro (8) and oral Vanc (9) | 250 mg/kg for 5 days | |
| G | 3 | 17 | Metro (17), Vanc (14) | IV Metro (49) and oral Vanc (61) | 250 mg/kg for 2 days | |
| H | 5 | 1 | Metro (1), Vanc (1) | Oral Metro (18), oral Vanc (22 ), IV Metro (3) | 250 mg/kg for 3 days | |
| I | 6 | 1 | Metro (1) | Oral and IV Metro (7,9), Vanc oral and enema (8,3) | 250 mg/kg for 2 days | |
| J | 8 | 16 | Metro (14), Vanc (2), Lacto (6) | Oral then IV Metro (10,5), oral and rectal Vanc (3,1), Lacto (6) | 300 mg/ kg for 1 day | |
| K | 6 | 7 | Metro (7), Vanc (3) | Oral then IV Metro (10) and oral Vanc (9) | Normal | 400 mg/kg for 2 days |
| L | 4 | 0 | None | IV then oral Metro (7,10), oral Vanc (23), Choles (7), and Lacto (5) | 150 mg/kg for 5 days | |
| M | 6 | 1 | Metro (1) | IV Metro (23) and oral Vanc (9) | 250 mg/kg for 2 days | |
| N | 7 | 1 | Metro (1), Vanc (1) | IV Metro (16), oral Vanc (14), oral Metro (7) | 250 mg /kg for 2 days | |
| O | 6 | 7 | Metro (6), Vanc (4) | IV Metro (6) and oral Vanc (22) | Low | 250 mg/kg for 2 days |
| P | 3 | 6 | Metro (6), Vanc (6) | IV Metro (25) and oral Vanc (17) | 150 mg/kg fro 3 days | |
| Q | 3 | 4 | Metro (4), Vanc (3) | Oral Metro (8),Vanc oral and enema (6,3) | 250 mg/kg for 3 days | |
| R | 5 | 9 | Vanc (9) | Vanc oral and enema (5,4), IV Metro (2) | 250 mg/kg for 1 day | |
| S | 8 | 23 | Metro (23), Vanc (23) | Oral then IV Metro (12,22), oral Vanc (39) | 250 mg/kg for 3 days | |
| T | 4 | 6 | Metro (6), Vanc (1) | Oral Vanc and IV Metro (11) | 250 mg/kg for 1 day | |
| U | 4 | 2 | Metro (2), Vanc (2) | Oral Vanc and IV Metro (6) | 250 mg/kg for 3 days | |
Survival with IVIG Use
Nine patients (43%) survived their illness and were discharged from the hospital. They experienced complete clinical resolution after an average of 10 days from IVIG administration (range, 220 days) (Table 4). The other 12 patients (57%) died during the index hospitalization. The average length of stay was 23 (range, 964) days.0
| Patient | Disposition | Clearance of Clostridium difficile Colitis? | Days to Resolution |
|---|---|---|---|
| |||
| A | Alive | Loose BM persisted but diarrhea resolved 9 days after IVIG. | 9 |
| B | Alive | BM became formed and diarrhea resolved 48 hours after IVIG. | 2 |
| C | Alive | Diarrhea resolved 20 days post‐IVIG infusion. CAT scan: colonic thickening improved. | 20 |
| D | Alive | Diarrhea resolved on discharge. Response to IVIG started next day after administration. | 18 |
| E | Alive | Diarrhea improved next day after IVIG administration and resolved on discharge. | 5 |
| F | Alive | Diarrhea resolved 5 days after IVIG administration. | 5 |
| G | Alive | Diarrhea resolved. C. difficile test became negative. | 13 |
| H | Alive | Diarrhea resolved 2 days before discharge. | 15 |
| I | Alive | Diarrhea slowly improved and resolved 4 days before discharge. | 7 |
| J | Deceased | ||
| K | Deceased | ||
| L | Deceased | ||
| M | Deceased | ||
| N | Deceased | ||
| O | Deceased | ||
| P | Deceased | ||
| Q | Deceased | ||
| R | Deceased | ||
| S | Deceased | ||
| T | Deceased | ||
| U | Deceased | ||
| Study | Number of Patients | Age (SD) (years) | Male | Female | Severity Definition | IVIG Dose | Days to Resolution | Days IVIG Infused | Alive? (%) | Recurrence |
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
| Salcedo et al.22 | 2 | 63, 64 | 1 | 1 | Pancolitis or thumbprinting on CAT scan | 200300 mg/kg once | 12 | 59 | 100 | 1 out of 2 |
| McPherson et al.21 | 8 | 72 (12) | ? | ? | Pancolitis | 200400 mg/kg twice | 226 | 1165 | 75 | 2 out of 6 |
| Juang et al.25 | 18 | 67 (17.4) | 5 | 13 | Modified Rubin et al.9 criteria | 200300 mg/kg once | ? | ? | 83 | ? |
| Hassoun et al.24 | 1 | 72 | 1 | 0 | Pancolitis | 400 mg/kg once | 6 | 15 | 100 | None |
| Chandrasekar et al.23 | 1 | 67 | 0 | 1 | Shock requiring inotropic support and pseudomembranes on colonoscopy | 400 mg/kg for 5 doses | 16 | 35 | 100 | ? |
| This paper | 21 | 68 (16) | 7 | 14 | Pancolitis and APACHE II score | 300 mg/kg once; 250 mg/kg for 5 doses | 220 | 025 | 43 | ? |
To further assess the impact of IVIG on colitis resolution, we investigated all variables in the data set that may have been associated with mortality using univariate Cox regression analysis. Those variables were as follows: APACHE II score on the first day of IVIG infusion, age, sex, previous history of CDC, number of days before IVIG use, peak white blood cell count, serum potassium level and creatinine level, lactate level on first day of IVIG infusion, and number of antibiotics administered that are not active against CDC. Only the APACHE II score (P = 0.006) and lactate level on the day of IVIG infusion (P = 0.004) were (positively) associated with CDC mortality. The positive association between CDC mortality and APACHE II score remained significant (P = 0.04) after adjusting for sex, previous history of CDC, number of days before IVIG use, lactate level on first day of IVIG infusion, and number of antibiotics administered that are not active against CDC using a multivariate Cox regression analysis model. No adjustments were made for age, white blood cell count, potassium level, or creatinine level as those are included within the APACHE II score. The positive association between lactate level on the first day of IVIG infusion and CDC mortality was not statistically significant after adjusting for the factors listed above in the same model (P = 0.13).
Discussion
To our knowledge, the present study is the largest series published in the literature to date on the use of IVIG for severe CDC. It is also the first study to report a high mortality rate compared to the 5 previous smaller studies on this topic. In the first report on IVIG use for CDC, Leung et al.15 used IVIG to treat 5 pediatric patients suffering from chronic relapsing CDC. It was not until 7 years later, in 1997, that the first IVIG use for severe CDC was reported.22 Since then, a total of 13 works have been published on IVIG for CDC treatment, and only in 5 of these was IVIG administered for severe CDC treatment:2125 3 case reports, 1 case series, and 1 case‐control study. Although the 4 uncontrolled reports concluded that IVIG is beneficial for severe CDC, the only controlled study reported no significant difference between cases and controls for all‐cause mortality, length of stay, and colectomy rate.25
The definition of severe CDC varied between reports, making comparison difficult. McPherson et al.21 and Hassoun and Ibrahim24 defined severe disease as one causing pancolitis on CT scan either with or without megacolon. In the study by Juang et al.,25 disease severity was assessed using the modified criteria of Rubin et al.9 Salcedo et al.22 defined severe CDC as one causing pancolitis in one patient and thumbprinting on CT scan in another, whereas Chandrasekar et al.23 defined it as one causing shock requiring inotropic support and presence of pseudomembranes on colonoscopy.
The present report is unique in that it provided 2 scales to characterize disease in each patient. The first scale measured colonic involvement anatomically and physiologically using a combination of computerized axial tomography (CAT) scan findings, presence or absence of ileus or referral for possible colectomy. This is not a prognostic scale, however, since CAT scan findings have been previously shown to be poor predictors of treatment outcome.29 The second scale measured the severity of systemic involvement using a well‐validated and standardized scale, the APACHE II score. We have shown in this report that it is associated with prognosis in the context of IVIG use.
Our study reports a higher mortality rate than previously described, and suggests that risk stratification and patient selection are important before IVIG administration, since not all patients seem to benefit from this treatment as previously suggested by smaller case series. Previously, several physical findings and laboratory values were found to be associated with worse outcome in CDC. These were increasing age, immunosuppression, shock requiring vasopressors, peak white blood cell count, peak serum lactate level, hypoalbuminemia, a fall in serum albumin level of >1.1 g/dL at the onset of CDC symptoms, use of 3 or more antibiotics, comorbid disease, previous history of CDC, acute renal failure and hypotension, underlying altered or depressed mental status, abdominal pain or distention, white blood cell count over 20,000/mm3 or <1500/mm3 and/or a >10% band forms on the white blood cell differential count, and ascites or pneumatosis coli by abdominal imaging.9, 3033 Using the APACHE II scale for the same purpose has the advantage of utilizing a well‐validated and objective scale that is expected to measure the degree of systemic involvement more reliably compared to the clinical and laboratory values above.
Timing of IVIG infusion remains controversial. Due to the lack of randomized controlled trials, the current practice is guided by expert opinion, leading to wide variations between reports. Since the APACHE II score was positively associated with mortality in the setting of IVIG treatment, the same scale could be used to guide decisions regarding timing of IVIG infusion. Our results suggest that IVIG should be preferentially used while the APACHE II score is still relatively low. This association and the specific APACHE II score at which to initiate or hold treatment need to be validated in the setting of a randomized controlled study before being used in clinical practice.
Although the current study was not designed to test this theory, IVIG could be associated conceptually with treatment success for patients with severe disease that is still restricted to the colon (without other organ dysfunction or at least at an early stage of extracolonic organ failure and thus associated with a low APACHE II score) but not for severe colonic disease with secondary multiple organ failure (high APACHE II score). This may be because colonic disease is toxin‐mediated whereas secondary systemic involvement is mediated through toxin‐induced inflammatory mediators (interleukin‐8, macrophage‐inflammatory protein‐2, substance P, tumor necrosis factor‐alpha) released locally in the colon,3436 triggering a SIRS and hematogenous translocation of colonic bacteria,37 both of which are poorly responsive to immunoglobulin infusion. Along the same lines, waiting for failure of conventional therapy before IVIG use might result in IVIG treatment failure because of disease progression and secondary sepsis, at which point no treatment may be effective. No study thus far has addressed this issue specifically.
Overall, the combined cohort of patients with severe CDC treated with IVIG in the literature includes 51 patients (Table 4). The current report contributes 41% of these patients. The patients' average age was 68 years, with a 2 to 1 female‐male ratio. The dose of IVIG used varied largely also, with 400 mg/kg being the mode (range, 75400 mg/kg from 1 to 5 doses). This dose is significantly below the doses used in the treatment of other diseases, like Guillain‐Barr syndrome, myasthenia gravis, Kawasaki disease, autoimmune hemolytic anemia, agammaglobulinemia, and hypogammaglobulinemia, where the usual dose is 400 mg/kg for 5 days. The resolution of diarrhea in these cases occurred after an average of 9 (range, 142) days. The index hospitalization survival rate varied from 43% to 100%. Patients received standard treatment for an average of 13 (range, 065) days before IVIG infusion. Thirty‐two of 51 patients survived their illness (63%). Neither total IgG nor anti‐toxin A IgG levels were measured in any of the reports. Of the 32 patients who had clinical resolution, 3 (10%) experienced symptoms recurrence in a follow‐up period of 1 to 13 months. This number is most probably an underestimation of the true recurrence rate resulting from an incomplete reporting because there was no uniform or active recurrence ascertainment mechanism in any of the studies. The recurrences were at 10, 14, and 30 days posttreatment. Since standard treatment was not discontinued in any of the reports once IVIG was given, the relative contribution and the ideal timing for IVIG infusion are still unclear.
The mechanism of action of IVIG is passive immunization (with anti‐toxin A and anti‐toxin B antibodies present in the pooled immunoglobulin) of a host who is usually unable to mount an adequate protective immune response.15, 22 IVIG is formed from pooling immunoglobulin from several random donors. It has been shown that many such donors express high anti‐toxin A and anti‐toxin B antibody serum titers.38, 39 In addition, high levels of anti‐toxin A and anti‐toxin B antibodies were present in the IVIG preparations and the recipients after infusion.1517, 22 Although constituting only a small fraction of the total IVIG administered, these antitoxin antibodies are believed to neutralize toxin A and B and help the host recover from the disease. In fact, Babcock et al.40 used an experimental hamster model of CDC to demonstrate a mortality reduction from 100% to 55% postinfusion of combined anti‐toxin A and anti‐toxin B antibodies. While some early reports indicated that anti‐toxin B antibodies were the major determinants of protection against colitis,41 later reports correlated disease severity pathologically42 and clinically43, 44 with anti‐toxin A levels. Anti‐toxin B antibodies were later shown to play an adjunctive role in conferring immunity against CDC40, 45, 46 when added to anti‐toxin A antibodies, but not to have any significant role on their own.
However, IVIG has been shown to contain IgG, but not IgA, anti‐toxin A and anti‐toxin B antibodies while it is only the IgA class of anti‐toxin A antibodies, and not the IgG class, that could neutralize toxin A in vitro and in vivo.47, 48 Babcock et al.40 solved this apparent dilemma by showing that a combination of 3 different monoclonal IgG anti‐toxin A antibodies could neutralize toxin A activity in vitro and prevent disease in the hamster model in vivo. Each of the 3 antibodies recognized a different toxin A domain: the first neutralized toxin A enzymatic activity, while the second prevented toxin A binding to its receptor on enterocytes, and the third prevented toxin internalization after binding to the receptor.
Thus, the mechanism of action of IVIG is most likely through the transfer of IgG anti‐toxin A antibodies that gain access to the intestinal lumen presumably secondary to inflammation‐induced mucosal damage and neutralize toxin A. Transfer of yet undetected IgA anti‐toxin A antibodies that prevent toxin A from binding to its receptor is much less likely, although possible.
The present study has limitations. As in all retrospective studies, selection bias was unavoidable. In addition, the decision to initiate IVIG administration was dependent on the attending physician, who also decided the dose, leading to heterogeneity in the total dose of IVIG infused. Such heterogeneity, however, is primarily the result of a lack of a standard dose for IVIG infusion for CDC in the published literature, as reported above. In addition, since IVIG is not yet approved by the U.S. Food and Drug Administration (FDA) for the treatment of severe CDC, standard treatment was not discontinued in any of the reports to date, including ours.
Choosing appropriate controls for patients suffering from severe CDC is challenging. This patient population is usually frail, with severe and multiple underlying diseases. The deteriorating clinical condition (and subsequently the need for multiple antibiotics) may be either the result of or the cause of CDC. Furthermore, IVIG has been in short supply for several years and therefore it has been expensive, making its administration to the number of patients needed to design adequately‐powered controlled studies difficult. These are mainly the reasons no randomized, multicenter, placebo‐controlled trial on IVIG use in severe CDC has been conducted to date.
Conclusions
In the present study, we report the results of IVIG use for the treatment of 21 patients with severe CDC. This is the largest cohort to our knowledge in the literature. Unlike previous studies on the subject, the present report provided 2 scales for disease assessment: the first based on the extent of colonic involvement and the second measuring the severity of systemic involvement using the APACHE II score. The latter was positively associated with mortality in this context. Of the 21 study patients treated with IVIG, only 9 patients (43%) survived their illness. This is the highest reported mortality rate among all studies on this subject so far. Further studies on the ideal timing of IVIG infusion, dose, and patient selection are needed before accepting IVIG as a standard of care for severe CDC treatment. The role of APACHE II score in the decision to use IVIG is promising and should be validated in randomized controlled trials.
Acknowledgements
The authors thank Indrani Mukherjee, MD, for her help and expertise in proofreading the manuscript.
- ,,.Secular trends in hospital‐acquired Clostridium difficile disease in the United States, 1987–2001.J Infect Dis.2004;189:1585–1589.
- ,,, et al.Fulminant Clostridium difficile: an underappreciated and increasing cause of death and complications.Ann Surg.2002;235:363–372.
- ,,, et al.An epidemic, toxin gene‐variant strain of Clostridium difficile.N Engl J Med.2005;353:2433–2441.
- ,,, et al.Clostridium difficile‐associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity.CMAJ.2004;171:466–472.
- ,,.Comparison of two toxins produced by Clostridium difficile.Infect Immun.1981;34:1036–1043.
- ,,,.Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A.N Engl J Med.2000;342:390–397.
- ,,,.Human antibody response to Clostridium difficile toxin A in relation to clinical course of infection.Infect Immun.1994;62:384–389.
- ,,.Risk factors for Clostridium difficile carriage and C. difficile‐associated diarrhea in a cohort of hospitalized patients.J Infect Dis.1990;162:678–684.
- ,,.Severe Clostridium difficile colitis.Dis Colon Rectum.1995;38:350–354.
- ,, et al.A predominantly clonal multi‐institutional outbreak of Clostridium difficile‐associated diarrhea with high morbidity and mortality.N Engl J Med.2005;353:2442–2449.
- ,,.Factors associated with failure of metronidazole in Clostridium difficile‐associated disease.J Clin Gastroenterol.2004;38:414–418.
- ,,.Treatment of Clostridium difficile‐associated disease: old therapies and new strategies.Lancet Infect Dis.2005;5:549–557.
- ,,, et al.Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole.Clin Infect Dis.2005;40:1586–1590.
- ,,, et al.Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada.Clin Infect Dis.2005;40:1591–1597.
- ,,,,,.Treatment with intravenously administered gamma globulin of chronic relapsing colitis induced by Clostridium difficile toxin.J Pediatr.1991;118:633–637.
- ,,,.Gamma globulin administration in relapsing Clostridium difficile‐induced pseudomembranous colitis with a defective antibody response to toxin A.Acta Clin Belg.1995;50:36–39.
- ,,,.Recurrent Clostridium difficile infection in a patient with selective IgG1 deficiency treated with intravenous immune globulin and Saccharomyces boulardii.Clin Infect Dis.1995;20(suppl 2):S266–S268.
- .Intravenous immunoglobulin for recurrent Clostridium difficile diarrhoea.Gut.2002;51:456.
- .Descriptive study of intravenous immunoglobulin for the treatment of recurrent Clostridium difficile diarrhoea.J Antimicrob Chemother.2004;53:882–884.
- ,,.Intravenous immunoglobulin for resistant Clostridium difficile infection.Age Ageing.2006;35:85–86.
- ,,,,.Intravenous immunoglobulin for the treatment of severe, refractory, and recurrent Clostridium difficile diarrhea.Dis Colon Rectum.2006;49:640–645.
- ,,, et al.Intravenous immunoglobulin therapy for severe Clostridium difficile colitis.Gut.1997;41:366–370.
- ,,, et. al.Intravenous immunoglobulin therapy for refractory Clostridium difficile toxin colitis in chronic kidney disease: case reports and literature review.NDT Plus.2008;1:20–22.
- ,.Use of intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis.Am J Geriatr Pharmacother.2007;5:48–51.
- ,,, et al.Clinical outcomes of intravenous immune globulin in severe clostridium difficile‐associated diarrhea.Am J Infect Control.2007;35:131–137.
- ,,,.Successful treatment of Clostridium difficile colitis with intravenous immunoglobulin.J Gastrointestin Liver Dis.2008;17:353–359.
- ,,, et al.A durable response to relapsing Clostridium difficile colitis may require combined therapy with high‐dose oral Vancomycin and intravenous immune globulin.Infect Dis Clin Pract.2006;14:217–220.
- ,,,.APACHE II: a severity of disease classification system.Crit Care Med.1985;13:818–829.
- ,,,,,.Colonic abnormalities on CT in adult hospitalized patients with Clostridium difficile colitis: prevalence and significance of findings.AJR Am J Roentgenol.2006;186:1393–1400.
- ,,,.Clostridium difficile‐associated diarrhea: predictors of severity in patients presenting to the emergency department.Can J Gastroenterol.2003;17:369–373.
- ,,, et al.Impact of emergency colectomy on survival of patients with fulminant Clostridium difficile colitis during an epidemic caused by a hypervirulent strain.Ann Surg.2007;245:267–272.
- ,, et al.Risk factors for severity and relapse of pseudomembranous colitis in an elderly population.Colorectal Dis.2007;9:173–177.
- ,,,,.Prognostic criteria in Clostridium difficile colitis.Am J Gastroenterol.1996;91:460–464.
- ,,, et al.Clostridium difficile toxin a stimulates macrophage‐inflammatory protein‐2 production in rat intestinal epithelial cells.J Immunol.1998;160:6039–6045.
- ,,, et al.Increased substance P responses in dorsal root ganglia, intestinal macrophages during Clostridium difficile toxin a enteritis in rats.Proc Natl Acad Sci U S A.1997;94:4788–4793.
- ,,,,,.Cytokine response by human monocytes to Clostridium difficile toxin a and toxin B.Infect Immun.1991;59:3659–3666.
- ,,,.Bacterial translocation, intestinal microflora and morphological changes of intestinal mucosa in experimental models of Clostridium difficile infection.J Med Microbiol.1998;47:591–598.
- ,.Immunoglobulin G directed against toxins A and B of Clostridium difficile in the general population and patients with antibiotic‐associated diarrhea.Diagn Microbiol Infect Dis.1994;18:205–209.
- ,,, et al.Serum antibody response to toxins A and B of Clostridium difficile.J Infect Dis.1983;148:93–100.
- ,,, et al.Human monoclonal antibodies directed against toxins A and B prevent Clostridium difficile‐induced mortality in hamsters.Infect Immun.2006;74:6339–6347.
- ,,,.Serum antibody response to Clostridium difficile toxins in patients with Clostridium difficile diarrhoea.Infection.1985;13:97–101.
- ,,,.Relationship between levels of Clostridium difficile toxin A and toxin B and cecal lesions in gnotobiotic mice.Infect Immun.1989;57:2123–2127.
- ,,,,.Protection against experimental pseudomembranous colitis in gnotobiotic mice by use of monoclonal antibodies against Clostridium difficile toxin A.Infect Immun.1991;59:1192–1195.
- ,,.Systemic and mucosal antibody responses to toxin A in patients infected with Clostridium difficile.J Infect Dis.1992;166:1287–1294.
- ,,, et al.Transcutaneous immunization with Clostridium difficile toxoid A induces systemic and mucosal immune responses and toxin A‐neutralizing antibodies in mice.Infect Immun.2007;75:2826–2832.
- ,,, et al.Serum antitoxin antibodies mediate systemic and mucosal protection from Clostridium difficile disease in hamsters.Infect Immun.1999;67:527–538.
- ,,,.Human colonic aspirates containing immunoglobulin A antibody to Clostridium difficile toxin A inhibit toxin A‐receptor binding.Gastroenterology.1992;102:35–40.
- ,,,,.Selective neutralization of a bacterial enterotoxin by serum immunoglobulin A in response to mucosal disease.Infect Immun.1995;63:3166–3173.
Clostridium difficile colitis (CDC) is the most common cause of hospital‐acquired diarrhea.1 The incidence of CDC has sharply increased over the past decade despite increasing awareness among health care professionals.24 C. difficile pathogenic strains induce diarrhea through the elaboration and secretion of 2 exotoxins: toxin A and toxin B. Toxin A is an inflammatory toxin, leading to fluid secretion, increased mucosal permeability, and marked enteritis and colitis.5 Toxin B is cytotoxic, leading to cell injury and apoptosis.5 Combined, toxin A and toxin B can cause a wide spectrum of clinical presentations, ranging from mild diarrhea that resolves with the discontinuation of antibiotics to a fulminant colitis requiring surgical intervention.
The severity of clinical manifestations has been shown to be inversely proportional to the host anti‐toxin A antibody level in response to toxin exposure. Kyne et al.6 demonstrated that asymptomatic C. difficile carriers produced significantly higher anti‐toxin A immunoglobulin G (IgG) levels compared to symptomatic patients. Among the latter group, patients with mild disease had a higher antibody level compared to those with severe colitis.6, 7 Additional risk factors predisposing to severe colitis are advanced age, severe underlying illness,8 and immunocompromised state.9
Recently, a new C. difficile strain (BI/NAP1) with a mutated toxin A and toxin B promoter silencer, a binary toxin gene, and fluoroquinolone resistance has been described in Canada and the United States.3, 10 This strain has been associated with an increased incidence of CDC among hospitalized patients, especially the incidence of severe disease requiring colectomy. At the same time, several reports describing metronidazole treatment failure have been published.1114 These recent findings emphasize the importance of finding alternative treatments for CDC.
Intravenous immunoglobulin (IVIG) was used to treat CDC for the first time in 1991.15 Since then, 12 case reports and small case series, along with 1 case‐control study have been published documenting IVIG treatment outcomes.1527 However, only 5 reports to date examined patients with severe CDC.2125 In the present study, we report the largest series of patients with severe CDC treated with IVIG in the literature to our knowledge.
Patients and Methods
Case Series
We used CareScience software (CareScience, Inc., Philadelphia, PA) to retrospectively identify all patients admitted to our institution with a primary or secondary diagnosis of CDC (code 00845) between July 1, 2002 and May 1, 2006. CareScience is commercially available software that tracks all admissions to our institution and allows the performance of patient searches with a wide spectrum of user‐defined search criteria. Using the same software, we further identified those patients who received IVIG during their hospital stay. We then obtained the hospital chart for each patient, from the medical records department and established the study database.
A case was defined as a patient with diarrhea (at least 3 loose stools daily) for at least 2 days who had C. difficile cytotoxin‐positive feces and at least 1 of the following criteria: clinical symptoms (abdominal pain and/or distension and fever); leukemoid reaction (defined as white blood cell count of 20,000 cells/mm3 or above. This cutoff value was chosen as it has been used previously as a prognostic factor)4; radiographic evidence of colitis by computed tomography (CT) of the abdomen; and/or the presence of pseudomembranes on flexible sigmoidoscopy or colonoscopy. We excluded all patients who received IVIG for an indication other than CDC treatment (n = 3). There were no other exclusion criteria.
We used a standardized data collection tool and recorded demographics (age, gender, principal diagnosis), past medical and surgical history, other risk factors for C. difficile infection (previous CDC, antibiotics received during hospital stay, immunosuppressive medications or organ transplantation within the previous 6 weeks, history of malignancy or diabetes mellitus); clinical presentation (abdominal distention, abdominal pain, diarrhea, fever, leukemoid reaction, and hypotension defined as systolic blood pressure <85 mm Hg despite at least 1 L of intravenous normal saline administration and the need for vasopressor use); colonoscopy findings; CT scan and x‐ray findings; laboratory values; date and dose of IVIG infused and other C. difficile pharmacological treatments; and Acute Physiological Assessment and Chronic Health Evaluation (APACHE II) score28 at the first day of IVIG infusion. The primary outcomes were survival at the end of the hospital stay and clinical disease resolution, defined as 2 formed bowel movements or less per day without abdominal pain or distention.
The decision to initiate IVIG therapy and the dose to be used was made by the individual attending physician.
Statistical Analysis
Single (univariate) and multiple (multivariate) logistic regression analysis were applied to identify variables among the ones collected that are independent predictors of CDC mortality. All statistical analyses were completed with the STATA 10 software package (StatCorp LP, College Station, TX).
Review of the Literature
We used PubMed, Web of Science, Scopus, and Excerpta Medica databases to search for any publication in a peer‐reviewed journal on the use of IVIG for the treatment of severe CDC. We used the search words: IVIG or intravenous immunoglobulin and clostridium difficile. Only publications published in English were selected. The date range used was January 1, 1950 to January 7, 2009. We were able to find 5 publications using this search criteria.
Results
Study Population
Of the 1230 patients diagnosed with CDC over the 4‐year study period, 21 patients were treated with IVIG. Table 1 summarizes the patients' characteristics. There were 13 women and 8 men. The mean age was 68 years, with a standard deviation (SD) of 13 years. Sepsis was the primary diagnosis in all patients. Sixteen patients had predisposing risk factors for CDC, including immunosuppression (immunosuppressive medication [n = 2], human immunodeficiency virus [HIV] infection [n = 2]); cancer (n = 3); recent surgery (n = 3); and diabetes mellitus (n = 11). Nine patients had documented previous CDC episodes. The indications for IVIG administration were evidence of pancolitis on abdominal CT scan (n = 12) or severe ileus with cessation of diarrhea, abdominal distention, and requirement for total parenteral nutrition (n = 5), or severe hypotension (n = 4) (defined as systolic blood pressure <85 mm Hg despite at least 1 L of intravenous normal saline administration and the subsequent need for vasopressor use).
| Patient | Age (gender) | Diagnosis | Medical and Surgical History | CDC History | Colonoscopy Findings | Radiographic Findings |
|---|---|---|---|---|---|---|
| ||||||
| A | 40 (female) | CDC with sec. sepsis | Gastric stapling | Yes | * | Diffuse colitis |
| B | 86 (female) | Fulminant CDC | Metastatic ovarian carcinoma | PC | No colonic thickening | |
| C | 72 (male) | Sepsis | Acute pancreatitis with sec. pseudocyst, DM | Yes | PC | Diffuse colitis |
| D | 78 (male) | Discitis | Delayed: normal mucosa | Dilation of small and large bowel | ||
| E | 98 (female) | Urosepsis | Yes | * | No bowel distention | |
| F | 90 (female) | Right lower extremity cellulitis | DM | * | Concentric thickening of rectal wall | |
| G | 64 (male) | Ischemic colitis | DM, recent Hartman pouch closure | Marked inflammation | Diffuse colitis | |
| H | 78 (female) | Toe osteomyelitis and CDC with sec. sepsis | DM | * | Diffuse colitis | |
| I | 35 (female) | Sepsis | Yes | * | Diffuse nonspecific colitis, minimal ascites | |
| J | 47 (female) | Pneumonia and sec. sepsis | * | Diffuse colitis | ||
| K | 56 (female) | Urosepsis | HIV | Yes | * | Colitis involving the right colon |
| L | 76 (male) | CDC with sec. sepsis | Sigmoid bladder fistula repair, DM | * | Diffuse colitis | |
| M | 71 (female) | Pneumonia with sec. sepsis | * | Diffuse colitis | ||
| N | 63 (male) | Urosepsis | DM, lymphoma resection from small intestine | PC | Marked small and large bowel distention | |
| O | 86 (male) | Enterococcus‐induced sepsis | Rheumatoid arthritis on methotrexate | * | Fat stranding suggesting peritonitis | |
| P | 60 (female) | Gastrointestinal bleed and CDC | DM with neuropathy and retinopathies | Yes | * | Thickening of wall of colon in most of the colon |
| Q | 57 (female) | Sepsis | DM | Yes | * | Normal |
| R | 67 (female) | CDC with sec. sepsis | Candidal esophagitis | Yes | * | Large amount of peritoneal fluid, mild small bowel thickening |
| S | 60 (female) | Sepsis sec. to S. aureus and P. aerogenosa | DM, renal transplant (myco, prednisone) | * | Ileus with air fluid level in the small intestine | |
| T | 80 (female) | Sepsis | DM | Yes | * | Thickening of descending colon consistent with colitis |
| U | 72 (male) | CDC, widespread metastatic cancer | DM, metastatic cancer (unknown primary site) | * | Severe colitis up to the splenic flexure | |
Table 2 describes disease severity in these patients. Since CDC starts locally in the colon then secondarily involves multiple organs as part of the systemic inflammatory response syndrome (SIRS), 2 scales were used to characterize the disease in each patient: (1) extent of local colonic inflammation, and (2) severity of systemic involvement. Extensive colonic involvement was evidenced in all patients by pancolitis on abdominal imaging modalities (12 patients), severe ileus requiring total parenteral nutrition (13 patients), or referral for surgical consultation for possible colectomy (12 patients).
| Patient | Complications During the Hospital Stay | APACHE II Score | Monitored Unit | WBC* | K | Alb | Lactate | Cr | Hospital Stay (days) | Surgical Consult/Surgery | TPN for Colitis |
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| A | Sepsis, DIC, ARDS, intubation | 15 | Yes | 48 | 4 | 1.1 | 2.9 | 1.3 | 26 | No/No | Yes |
| B | Dehydration, weakness | 12 | No | 19 | 3.4 | 3.1 | 1.1 | 16 | No/No | Yes | |
| C | HTP, GI bleed, ischemic colitis, F | 22 | Yes | 21 | 2.7 | 2.5 | 1.3 | ESRD | 34 | Yes/No | Yes |
| D | AD, megacolon, HT, intubation, ARF | 18 | Yes | 25 | 3.4 | 1.4 | 1.1 | 2.6 | 52 | Yes/No | Yes |
| E | Exacerbation of CHF with respiratory distress, Bipap support, confusion | 21 | No | 10 | 2.6 | 2.3 | 1 | 11 | No/No | No | |
| F | Confusion, gout acute attack | 21 | No | 33 | 3.1 | 2.6 | ESRD | 15 | No/No | No | |
| G | Intubation, cardiac arrest, AF with RVR, PNA, ARF, DVT, dysphagia, PEG | 6 | Yes | 15 | 3.1 | 1.7 | 1.9 | 2.2 | 32 | Yes/No | Yes |
| H | Sepsis, intubation, ARF, PEG, toe amputation, PNA, pulmonary edema, TPN, vitamin D deficiency | 34 | Yes | 59 | 2.8 | 1.5 | 4 | 3.8 | 17 | No/No | Yes |
| I | CHF, transient third cranial nerve palsy, DIC | 20 | Yes | 52 | 3.2 | 3.3 | 0.7 | ESRD | 11 | No/No | No |
| J | Aspiration PNA, sepsis, DIC, intubation, MI, F, HT | 32 | Yes | 17 | 2.7 | 1.8 | 14 | ESRD | 21 | Yes/no | No |
| K | Intubation, cardiac arrest, ARF, DIC, AP, F | 30 | Yes | 25 | 2.1 | 2.1 | 13 | 3.6 | 10 | Yes/no | No |
| L | Intubation, ARF, HTP, MI, AD, F | 23 | Yes | 69 | 3.2 | 1.8 | 1.4 | 3.6 | 23 | Yes/no | No |
| M | Intubation, DIC, ARF, GI bleeding, hypothermia, AD | 23 | Yes | 47 | 2.8 | 1.4 | 1.4 | 4.3 | 23 | Yes/no | Yes |
| N | Intubation, HTP, AD, F, ARF with HD, AF, osteomyelitis | 23 | Yes | 46 | 2.9 | 1.5 | 1.5 | 2.7 | 27 | Yes/no | Yes |
| O | Intubation, ARF, 2 cardiac arrests, GI bleed, UTI, rhabdomyolysis, liver shock, AF | 31 | Yes | 49 | 2.5 | 1.1 | 2.3 | 2.9 | 25 | Yes/no | Yes |
| P | Bowel ischemia with bowel resection, ARF, MI, ischemic bowel, fluid overload, respiratory failure | 23 | Yes | 26 | 2.6 | 0.8 | 2.9 | 3.1 | 9 | Yes/Yes | Yes |
| Q | Pulmonary embolism | 39 | Yes | 23 | 2.8 | 1.1 | 8.1 | ESRD | 9 | No/No | Yes |
| R | Fungal peritonitis, aspiration pneumonia, cardiac arrest | 26 | Yes | 30 | 4.1 | 1.1 | 0.9 | ESRD | 36 | No/No | Yes |
| S | Intubation, pneumothorax, CRT, pressor‐dependent shock, ARF | 36 | Yes | 46 | 3.1 | 1.8 | 2.1 | 5.4 | 64 | Yes/Yes | No |
| T | Pressor‐dependent sepsis, pulmonary edema, ARF with HD | 36 | Yes | 35 | 3.3 | 1.3 | 1.8 | 5.5 | 11 | Yes/No | Yes |
| U | Sepsis | 34 | Yes | 58 | 3.1 | 2.2 | 3.3 | 1.6 | 9 | No/No | No |
Of the 21 patients treated with IVIG, 9 did not receive a surgical consultation either because they responded to medical treatment promptly (6 patients), were too unstable for surgery (2 patients), or because the patient/family refused surgery (1 patient). Of the 12 patients who received surgical consultation, 2 underwent surgery. The remainder did not proceed to surgery for the following reasons: they were deemed medically unstable for surgery (6 patients), declined surgery (2 patients), were diagnosed with cancer on colonoscopy (1 patient), or improved with medical treatment (1 patient).
The severity of systemic involvement was measured using the APACHE II score on day 1 of IVIG infusion. The mean APACHE II score was 25. Eighteen patients were in a monitored unit when IVIG was administered. The study group had laboratory results in keeping with those previously used to define severe colitis:4, 9, 19 leukocytosis (defined as white blood cell count higher than 12,000 cells/mL [mean = 36,000 cells/mL]), hypoalbuminemia (mean = 1.78 g/dL, SD = 0.68 g/dL), hypokalemia (mean = 3.02 mg/dL, SD = 0.47 g/dL), and acute renal failure (defined as serum creatinine level >1.5 mg/dL [mean = 2.98 mg/dL, SD = 1.42 g/dL]).
IVIG Use
Table 3 describes the treatment patients received for CDC as well as the total number of antibiotics used throughout the hospital stay. IVIG was used as adjuvant treatment (defined as IVIG administration within 4 days or less after CDC diagnosis) in 8 patients and as second‐line treatment (defined as IVIG administration more than 4 days after CDC diagnosis) in 13 patients. Metronidazole, vancomycin, cholestyramine, and probiotic treatment alone or in different combinations were used for an average of 8 days (SD = 8 days; range, 025 days) before IVIG infusion. The total IVIG dose administered varied depending on the prescribing attending, with a range of 200 mg/kg to 1250 mg/kg and a mode of 250 mg/kg for 1 to 3 days. An average of 5 (SD = 2) different antibiotics that were not active against C. difficile were used per patient without being discontinued after a CDC diagnosis was made. The 3 most common were: cephalosporins (cefazolin, ceftriaxone, cefepime), fluoroquinolones (levofloxacin), and combination antibiotics (piperacillin and tazobactam or ampicillin and sulbactam).
| Patient | Number of Antibiotics | Duration of Treatment Before IVIG (days) | Treatment Before IVIG (days) | Total CDC Treatment (days) | IgG Level | IVIG Dose |
|---|---|---|---|---|---|---|
| ||||||
| A | 5 | 7 | Metro (7), Vanc (7), Choles (2) | Oral and rectal Vanc (26,19), IV Metro (19), Choles (2), Lacto (6) | Low | 300 mg/kg for 1 day |
| B | 1 | 13 | Metro (13), Vanc (13) | Oral Vanc (17) and IV Metro (12) | 300 mg/kg for 1 day | |
| C | 3 | 7 | Metro (7), Vanc (3) | IV Metro (28),Vanc oral and enema (18,3), Lacto (10) | Low | 125 mg/kg for 5 days |
| D | 5 | 25 | Metro (25), Vanc (15) | Oral then IV Metro (10,15), oral Vanc (25), Choles (7) and Lacto (13) | Low | 200 mg/kg for 1 day |
| E | 1 | 4 | Metro (1), Vanc (4), Choles(4) | IV Metro (8), oral Vanc (10) and Choles (4) | 75 mg/kg for 5 days | |
| F | 4 | 2 | Metro (2), Vanc (1) | IV Metro (8) and oral Vanc (9) | 250 mg/kg for 5 days | |
| G | 3 | 17 | Metro (17), Vanc (14) | IV Metro (49) and oral Vanc (61) | 250 mg/kg for 2 days | |
| H | 5 | 1 | Metro (1), Vanc (1) | Oral Metro (18), oral Vanc (22 ), IV Metro (3) | 250 mg/kg for 3 days | |
| I | 6 | 1 | Metro (1) | Oral and IV Metro (7,9), Vanc oral and enema (8,3) | 250 mg/kg for 2 days | |
| J | 8 | 16 | Metro (14), Vanc (2), Lacto (6) | Oral then IV Metro (10,5), oral and rectal Vanc (3,1), Lacto (6) | 300 mg/ kg for 1 day | |
| K | 6 | 7 | Metro (7), Vanc (3) | Oral then IV Metro (10) and oral Vanc (9) | Normal | 400 mg/kg for 2 days |
| L | 4 | 0 | None | IV then oral Metro (7,10), oral Vanc (23), Choles (7), and Lacto (5) | 150 mg/kg for 5 days | |
| M | 6 | 1 | Metro (1) | IV Metro (23) and oral Vanc (9) | 250 mg/kg for 2 days | |
| N | 7 | 1 | Metro (1), Vanc (1) | IV Metro (16), oral Vanc (14), oral Metro (7) | 250 mg /kg for 2 days | |
| O | 6 | 7 | Metro (6), Vanc (4) | IV Metro (6) and oral Vanc (22) | Low | 250 mg/kg for 2 days |
| P | 3 | 6 | Metro (6), Vanc (6) | IV Metro (25) and oral Vanc (17) | 150 mg/kg fro 3 days | |
| Q | 3 | 4 | Metro (4), Vanc (3) | Oral Metro (8),Vanc oral and enema (6,3) | 250 mg/kg for 3 days | |
| R | 5 | 9 | Vanc (9) | Vanc oral and enema (5,4), IV Metro (2) | 250 mg/kg for 1 day | |
| S | 8 | 23 | Metro (23), Vanc (23) | Oral then IV Metro (12,22), oral Vanc (39) | 250 mg/kg for 3 days | |
| T | 4 | 6 | Metro (6), Vanc (1) | Oral Vanc and IV Metro (11) | 250 mg/kg for 1 day | |
| U | 4 | 2 | Metro (2), Vanc (2) | Oral Vanc and IV Metro (6) | 250 mg/kg for 3 days | |
Survival with IVIG Use
Nine patients (43%) survived their illness and were discharged from the hospital. They experienced complete clinical resolution after an average of 10 days from IVIG administration (range, 220 days) (Table 4). The other 12 patients (57%) died during the index hospitalization. The average length of stay was 23 (range, 964) days.0
| Patient | Disposition | Clearance of Clostridium difficile Colitis? | Days to Resolution |
|---|---|---|---|
| |||
| A | Alive | Loose BM persisted but diarrhea resolved 9 days after IVIG. | 9 |
| B | Alive | BM became formed and diarrhea resolved 48 hours after IVIG. | 2 |
| C | Alive | Diarrhea resolved 20 days post‐IVIG infusion. CAT scan: colonic thickening improved. | 20 |
| D | Alive | Diarrhea resolved on discharge. Response to IVIG started next day after administration. | 18 |
| E | Alive | Diarrhea improved next day after IVIG administration and resolved on discharge. | 5 |
| F | Alive | Diarrhea resolved 5 days after IVIG administration. | 5 |
| G | Alive | Diarrhea resolved. C. difficile test became negative. | 13 |
| H | Alive | Diarrhea resolved 2 days before discharge. | 15 |
| I | Alive | Diarrhea slowly improved and resolved 4 days before discharge. | 7 |
| J | Deceased | ||
| K | Deceased | ||
| L | Deceased | ||
| M | Deceased | ||
| N | Deceased | ||
| O | Deceased | ||
| P | Deceased | ||
| Q | Deceased | ||
| R | Deceased | ||
| S | Deceased | ||
| T | Deceased | ||
| U | Deceased | ||
| Study | Number of Patients | Age (SD) (years) | Male | Female | Severity Definition | IVIG Dose | Days to Resolution | Days IVIG Infused | Alive? (%) | Recurrence |
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
| Salcedo et al.22 | 2 | 63, 64 | 1 | 1 | Pancolitis or thumbprinting on CAT scan | 200300 mg/kg once | 12 | 59 | 100 | 1 out of 2 |
| McPherson et al.21 | 8 | 72 (12) | ? | ? | Pancolitis | 200400 mg/kg twice | 226 | 1165 | 75 | 2 out of 6 |
| Juang et al.25 | 18 | 67 (17.4) | 5 | 13 | Modified Rubin et al.9 criteria | 200300 mg/kg once | ? | ? | 83 | ? |
| Hassoun et al.24 | 1 | 72 | 1 | 0 | Pancolitis | 400 mg/kg once | 6 | 15 | 100 | None |
| Chandrasekar et al.23 | 1 | 67 | 0 | 1 | Shock requiring inotropic support and pseudomembranes on colonoscopy | 400 mg/kg for 5 doses | 16 | 35 | 100 | ? |
| This paper | 21 | 68 (16) | 7 | 14 | Pancolitis and APACHE II score | 300 mg/kg once; 250 mg/kg for 5 doses | 220 | 025 | 43 | ? |
To further assess the impact of IVIG on colitis resolution, we investigated all variables in the data set that may have been associated with mortality using univariate Cox regression analysis. Those variables were as follows: APACHE II score on the first day of IVIG infusion, age, sex, previous history of CDC, number of days before IVIG use, peak white blood cell count, serum potassium level and creatinine level, lactate level on first day of IVIG infusion, and number of antibiotics administered that are not active against CDC. Only the APACHE II score (P = 0.006) and lactate level on the day of IVIG infusion (P = 0.004) were (positively) associated with CDC mortality. The positive association between CDC mortality and APACHE II score remained significant (P = 0.04) after adjusting for sex, previous history of CDC, number of days before IVIG use, lactate level on first day of IVIG infusion, and number of antibiotics administered that are not active against CDC using a multivariate Cox regression analysis model. No adjustments were made for age, white blood cell count, potassium level, or creatinine level as those are included within the APACHE II score. The positive association between lactate level on the first day of IVIG infusion and CDC mortality was not statistically significant after adjusting for the factors listed above in the same model (P = 0.13).
Discussion
To our knowledge, the present study is the largest series published in the literature to date on the use of IVIG for severe CDC. It is also the first study to report a high mortality rate compared to the 5 previous smaller studies on this topic. In the first report on IVIG use for CDC, Leung et al.15 used IVIG to treat 5 pediatric patients suffering from chronic relapsing CDC. It was not until 7 years later, in 1997, that the first IVIG use for severe CDC was reported.22 Since then, a total of 13 works have been published on IVIG for CDC treatment, and only in 5 of these was IVIG administered for severe CDC treatment:2125 3 case reports, 1 case series, and 1 case‐control study. Although the 4 uncontrolled reports concluded that IVIG is beneficial for severe CDC, the only controlled study reported no significant difference between cases and controls for all‐cause mortality, length of stay, and colectomy rate.25
The definition of severe CDC varied between reports, making comparison difficult. McPherson et al.21 and Hassoun and Ibrahim24 defined severe disease as one causing pancolitis on CT scan either with or without megacolon. In the study by Juang et al.,25 disease severity was assessed using the modified criteria of Rubin et al.9 Salcedo et al.22 defined severe CDC as one causing pancolitis in one patient and thumbprinting on CT scan in another, whereas Chandrasekar et al.23 defined it as one causing shock requiring inotropic support and presence of pseudomembranes on colonoscopy.
The present report is unique in that it provided 2 scales to characterize disease in each patient. The first scale measured colonic involvement anatomically and physiologically using a combination of computerized axial tomography (CAT) scan findings, presence or absence of ileus or referral for possible colectomy. This is not a prognostic scale, however, since CAT scan findings have been previously shown to be poor predictors of treatment outcome.29 The second scale measured the severity of systemic involvement using a well‐validated and standardized scale, the APACHE II score. We have shown in this report that it is associated with prognosis in the context of IVIG use.
Our study reports a higher mortality rate than previously described, and suggests that risk stratification and patient selection are important before IVIG administration, since not all patients seem to benefit from this treatment as previously suggested by smaller case series. Previously, several physical findings and laboratory values were found to be associated with worse outcome in CDC. These were increasing age, immunosuppression, shock requiring vasopressors, peak white blood cell count, peak serum lactate level, hypoalbuminemia, a fall in serum albumin level of >1.1 g/dL at the onset of CDC symptoms, use of 3 or more antibiotics, comorbid disease, previous history of CDC, acute renal failure and hypotension, underlying altered or depressed mental status, abdominal pain or distention, white blood cell count over 20,000/mm3 or <1500/mm3 and/or a >10% band forms on the white blood cell differential count, and ascites or pneumatosis coli by abdominal imaging.9, 3033 Using the APACHE II scale for the same purpose has the advantage of utilizing a well‐validated and objective scale that is expected to measure the degree of systemic involvement more reliably compared to the clinical and laboratory values above.
Timing of IVIG infusion remains controversial. Due to the lack of randomized controlled trials, the current practice is guided by expert opinion, leading to wide variations between reports. Since the APACHE II score was positively associated with mortality in the setting of IVIG treatment, the same scale could be used to guide decisions regarding timing of IVIG infusion. Our results suggest that IVIG should be preferentially used while the APACHE II score is still relatively low. This association and the specific APACHE II score at which to initiate or hold treatment need to be validated in the setting of a randomized controlled study before being used in clinical practice.
Although the current study was not designed to test this theory, IVIG could be associated conceptually with treatment success for patients with severe disease that is still restricted to the colon (without other organ dysfunction or at least at an early stage of extracolonic organ failure and thus associated with a low APACHE II score) but not for severe colonic disease with secondary multiple organ failure (high APACHE II score). This may be because colonic disease is toxin‐mediated whereas secondary systemic involvement is mediated through toxin‐induced inflammatory mediators (interleukin‐8, macrophage‐inflammatory protein‐2, substance P, tumor necrosis factor‐alpha) released locally in the colon,3436 triggering a SIRS and hematogenous translocation of colonic bacteria,37 both of which are poorly responsive to immunoglobulin infusion. Along the same lines, waiting for failure of conventional therapy before IVIG use might result in IVIG treatment failure because of disease progression and secondary sepsis, at which point no treatment may be effective. No study thus far has addressed this issue specifically.
Overall, the combined cohort of patients with severe CDC treated with IVIG in the literature includes 51 patients (Table 4). The current report contributes 41% of these patients. The patients' average age was 68 years, with a 2 to 1 female‐male ratio. The dose of IVIG used varied largely also, with 400 mg/kg being the mode (range, 75400 mg/kg from 1 to 5 doses). This dose is significantly below the doses used in the treatment of other diseases, like Guillain‐Barr syndrome, myasthenia gravis, Kawasaki disease, autoimmune hemolytic anemia, agammaglobulinemia, and hypogammaglobulinemia, where the usual dose is 400 mg/kg for 5 days. The resolution of diarrhea in these cases occurred after an average of 9 (range, 142) days. The index hospitalization survival rate varied from 43% to 100%. Patients received standard treatment for an average of 13 (range, 065) days before IVIG infusion. Thirty‐two of 51 patients survived their illness (63%). Neither total IgG nor anti‐toxin A IgG levels were measured in any of the reports. Of the 32 patients who had clinical resolution, 3 (10%) experienced symptoms recurrence in a follow‐up period of 1 to 13 months. This number is most probably an underestimation of the true recurrence rate resulting from an incomplete reporting because there was no uniform or active recurrence ascertainment mechanism in any of the studies. The recurrences were at 10, 14, and 30 days posttreatment. Since standard treatment was not discontinued in any of the reports once IVIG was given, the relative contribution and the ideal timing for IVIG infusion are still unclear.
The mechanism of action of IVIG is passive immunization (with anti‐toxin A and anti‐toxin B antibodies present in the pooled immunoglobulin) of a host who is usually unable to mount an adequate protective immune response.15, 22 IVIG is formed from pooling immunoglobulin from several random donors. It has been shown that many such donors express high anti‐toxin A and anti‐toxin B antibody serum titers.38, 39 In addition, high levels of anti‐toxin A and anti‐toxin B antibodies were present in the IVIG preparations and the recipients after infusion.1517, 22 Although constituting only a small fraction of the total IVIG administered, these antitoxin antibodies are believed to neutralize toxin A and B and help the host recover from the disease. In fact, Babcock et al.40 used an experimental hamster model of CDC to demonstrate a mortality reduction from 100% to 55% postinfusion of combined anti‐toxin A and anti‐toxin B antibodies. While some early reports indicated that anti‐toxin B antibodies were the major determinants of protection against colitis,41 later reports correlated disease severity pathologically42 and clinically43, 44 with anti‐toxin A levels. Anti‐toxin B antibodies were later shown to play an adjunctive role in conferring immunity against CDC40, 45, 46 when added to anti‐toxin A antibodies, but not to have any significant role on their own.
However, IVIG has been shown to contain IgG, but not IgA, anti‐toxin A and anti‐toxin B antibodies while it is only the IgA class of anti‐toxin A antibodies, and not the IgG class, that could neutralize toxin A in vitro and in vivo.47, 48 Babcock et al.40 solved this apparent dilemma by showing that a combination of 3 different monoclonal IgG anti‐toxin A antibodies could neutralize toxin A activity in vitro and prevent disease in the hamster model in vivo. Each of the 3 antibodies recognized a different toxin A domain: the first neutralized toxin A enzymatic activity, while the second prevented toxin A binding to its receptor on enterocytes, and the third prevented toxin internalization after binding to the receptor.
Thus, the mechanism of action of IVIG is most likely through the transfer of IgG anti‐toxin A antibodies that gain access to the intestinal lumen presumably secondary to inflammation‐induced mucosal damage and neutralize toxin A. Transfer of yet undetected IgA anti‐toxin A antibodies that prevent toxin A from binding to its receptor is much less likely, although possible.
The present study has limitations. As in all retrospective studies, selection bias was unavoidable. In addition, the decision to initiate IVIG administration was dependent on the attending physician, who also decided the dose, leading to heterogeneity in the total dose of IVIG infused. Such heterogeneity, however, is primarily the result of a lack of a standard dose for IVIG infusion for CDC in the published literature, as reported above. In addition, since IVIG is not yet approved by the U.S. Food and Drug Administration (FDA) for the treatment of severe CDC, standard treatment was not discontinued in any of the reports to date, including ours.
Choosing appropriate controls for patients suffering from severe CDC is challenging. This patient population is usually frail, with severe and multiple underlying diseases. The deteriorating clinical condition (and subsequently the need for multiple antibiotics) may be either the result of or the cause of CDC. Furthermore, IVIG has been in short supply for several years and therefore it has been expensive, making its administration to the number of patients needed to design adequately‐powered controlled studies difficult. These are mainly the reasons no randomized, multicenter, placebo‐controlled trial on IVIG use in severe CDC has been conducted to date.
Conclusions
In the present study, we report the results of IVIG use for the treatment of 21 patients with severe CDC. This is the largest cohort to our knowledge in the literature. Unlike previous studies on the subject, the present report provided 2 scales for disease assessment: the first based on the extent of colonic involvement and the second measuring the severity of systemic involvement using the APACHE II score. The latter was positively associated with mortality in this context. Of the 21 study patients treated with IVIG, only 9 patients (43%) survived their illness. This is the highest reported mortality rate among all studies on this subject so far. Further studies on the ideal timing of IVIG infusion, dose, and patient selection are needed before accepting IVIG as a standard of care for severe CDC treatment. The role of APACHE II score in the decision to use IVIG is promising and should be validated in randomized controlled trials.
Acknowledgements
The authors thank Indrani Mukherjee, MD, for her help and expertise in proofreading the manuscript.
Clostridium difficile colitis (CDC) is the most common cause of hospital‐acquired diarrhea.1 The incidence of CDC has sharply increased over the past decade despite increasing awareness among health care professionals.24 C. difficile pathogenic strains induce diarrhea through the elaboration and secretion of 2 exotoxins: toxin A and toxin B. Toxin A is an inflammatory toxin, leading to fluid secretion, increased mucosal permeability, and marked enteritis and colitis.5 Toxin B is cytotoxic, leading to cell injury and apoptosis.5 Combined, toxin A and toxin B can cause a wide spectrum of clinical presentations, ranging from mild diarrhea that resolves with the discontinuation of antibiotics to a fulminant colitis requiring surgical intervention.
The severity of clinical manifestations has been shown to be inversely proportional to the host anti‐toxin A antibody level in response to toxin exposure. Kyne et al.6 demonstrated that asymptomatic C. difficile carriers produced significantly higher anti‐toxin A immunoglobulin G (IgG) levels compared to symptomatic patients. Among the latter group, patients with mild disease had a higher antibody level compared to those with severe colitis.6, 7 Additional risk factors predisposing to severe colitis are advanced age, severe underlying illness,8 and immunocompromised state.9
Recently, a new C. difficile strain (BI/NAP1) with a mutated toxin A and toxin B promoter silencer, a binary toxin gene, and fluoroquinolone resistance has been described in Canada and the United States.3, 10 This strain has been associated with an increased incidence of CDC among hospitalized patients, especially the incidence of severe disease requiring colectomy. At the same time, several reports describing metronidazole treatment failure have been published.1114 These recent findings emphasize the importance of finding alternative treatments for CDC.
Intravenous immunoglobulin (IVIG) was used to treat CDC for the first time in 1991.15 Since then, 12 case reports and small case series, along with 1 case‐control study have been published documenting IVIG treatment outcomes.1527 However, only 5 reports to date examined patients with severe CDC.2125 In the present study, we report the largest series of patients with severe CDC treated with IVIG in the literature to our knowledge.
Patients and Methods
Case Series
We used CareScience software (CareScience, Inc., Philadelphia, PA) to retrospectively identify all patients admitted to our institution with a primary or secondary diagnosis of CDC (code 00845) between July 1, 2002 and May 1, 2006. CareScience is commercially available software that tracks all admissions to our institution and allows the performance of patient searches with a wide spectrum of user‐defined search criteria. Using the same software, we further identified those patients who received IVIG during their hospital stay. We then obtained the hospital chart for each patient, from the medical records department and established the study database.
A case was defined as a patient with diarrhea (at least 3 loose stools daily) for at least 2 days who had C. difficile cytotoxin‐positive feces and at least 1 of the following criteria: clinical symptoms (abdominal pain and/or distension and fever); leukemoid reaction (defined as white blood cell count of 20,000 cells/mm3 or above. This cutoff value was chosen as it has been used previously as a prognostic factor)4; radiographic evidence of colitis by computed tomography (CT) of the abdomen; and/or the presence of pseudomembranes on flexible sigmoidoscopy or colonoscopy. We excluded all patients who received IVIG for an indication other than CDC treatment (n = 3). There were no other exclusion criteria.
We used a standardized data collection tool and recorded demographics (age, gender, principal diagnosis), past medical and surgical history, other risk factors for C. difficile infection (previous CDC, antibiotics received during hospital stay, immunosuppressive medications or organ transplantation within the previous 6 weeks, history of malignancy or diabetes mellitus); clinical presentation (abdominal distention, abdominal pain, diarrhea, fever, leukemoid reaction, and hypotension defined as systolic blood pressure <85 mm Hg despite at least 1 L of intravenous normal saline administration and the need for vasopressor use); colonoscopy findings; CT scan and x‐ray findings; laboratory values; date and dose of IVIG infused and other C. difficile pharmacological treatments; and Acute Physiological Assessment and Chronic Health Evaluation (APACHE II) score28 at the first day of IVIG infusion. The primary outcomes were survival at the end of the hospital stay and clinical disease resolution, defined as 2 formed bowel movements or less per day without abdominal pain or distention.
The decision to initiate IVIG therapy and the dose to be used was made by the individual attending physician.
Statistical Analysis
Single (univariate) and multiple (multivariate) logistic regression analysis were applied to identify variables among the ones collected that are independent predictors of CDC mortality. All statistical analyses were completed with the STATA 10 software package (StatCorp LP, College Station, TX).
Review of the Literature
We used PubMed, Web of Science, Scopus, and Excerpta Medica databases to search for any publication in a peer‐reviewed journal on the use of IVIG for the treatment of severe CDC. We used the search words: IVIG or intravenous immunoglobulin and clostridium difficile. Only publications published in English were selected. The date range used was January 1, 1950 to January 7, 2009. We were able to find 5 publications using this search criteria.
Results
Study Population
Of the 1230 patients diagnosed with CDC over the 4‐year study period, 21 patients were treated with IVIG. Table 1 summarizes the patients' characteristics. There were 13 women and 8 men. The mean age was 68 years, with a standard deviation (SD) of 13 years. Sepsis was the primary diagnosis in all patients. Sixteen patients had predisposing risk factors for CDC, including immunosuppression (immunosuppressive medication [n = 2], human immunodeficiency virus [HIV] infection [n = 2]); cancer (n = 3); recent surgery (n = 3); and diabetes mellitus (n = 11). Nine patients had documented previous CDC episodes. The indications for IVIG administration were evidence of pancolitis on abdominal CT scan (n = 12) or severe ileus with cessation of diarrhea, abdominal distention, and requirement for total parenteral nutrition (n = 5), or severe hypotension (n = 4) (defined as systolic blood pressure <85 mm Hg despite at least 1 L of intravenous normal saline administration and the subsequent need for vasopressor use).
| Patient | Age (gender) | Diagnosis | Medical and Surgical History | CDC History | Colonoscopy Findings | Radiographic Findings |
|---|---|---|---|---|---|---|
| ||||||
| A | 40 (female) | CDC with sec. sepsis | Gastric stapling | Yes | * | Diffuse colitis |
| B | 86 (female) | Fulminant CDC | Metastatic ovarian carcinoma | PC | No colonic thickening | |
| C | 72 (male) | Sepsis | Acute pancreatitis with sec. pseudocyst, DM | Yes | PC | Diffuse colitis |
| D | 78 (male) | Discitis | Delayed: normal mucosa | Dilation of small and large bowel | ||
| E | 98 (female) | Urosepsis | Yes | * | No bowel distention | |
| F | 90 (female) | Right lower extremity cellulitis | DM | * | Concentric thickening of rectal wall | |
| G | 64 (male) | Ischemic colitis | DM, recent Hartman pouch closure | Marked inflammation | Diffuse colitis | |
| H | 78 (female) | Toe osteomyelitis and CDC with sec. sepsis | DM | * | Diffuse colitis | |
| I | 35 (female) | Sepsis | Yes | * | Diffuse nonspecific colitis, minimal ascites | |
| J | 47 (female) | Pneumonia and sec. sepsis | * | Diffuse colitis | ||
| K | 56 (female) | Urosepsis | HIV | Yes | * | Colitis involving the right colon |
| L | 76 (male) | CDC with sec. sepsis | Sigmoid bladder fistula repair, DM | * | Diffuse colitis | |
| M | 71 (female) | Pneumonia with sec. sepsis | * | Diffuse colitis | ||
| N | 63 (male) | Urosepsis | DM, lymphoma resection from small intestine | PC | Marked small and large bowel distention | |
| O | 86 (male) | Enterococcus‐induced sepsis | Rheumatoid arthritis on methotrexate | * | Fat stranding suggesting peritonitis | |
| P | 60 (female) | Gastrointestinal bleed and CDC | DM with neuropathy and retinopathies | Yes | * | Thickening of wall of colon in most of the colon |
| Q | 57 (female) | Sepsis | DM | Yes | * | Normal |
| R | 67 (female) | CDC with sec. sepsis | Candidal esophagitis | Yes | * | Large amount of peritoneal fluid, mild small bowel thickening |
| S | 60 (female) | Sepsis sec. to S. aureus and P. aerogenosa | DM, renal transplant (myco, prednisone) | * | Ileus with air fluid level in the small intestine | |
| T | 80 (female) | Sepsis | DM | Yes | * | Thickening of descending colon consistent with colitis |
| U | 72 (male) | CDC, widespread metastatic cancer | DM, metastatic cancer (unknown primary site) | * | Severe colitis up to the splenic flexure | |
Table 2 describes disease severity in these patients. Since CDC starts locally in the colon then secondarily involves multiple organs as part of the systemic inflammatory response syndrome (SIRS), 2 scales were used to characterize the disease in each patient: (1) extent of local colonic inflammation, and (2) severity of systemic involvement. Extensive colonic involvement was evidenced in all patients by pancolitis on abdominal imaging modalities (12 patients), severe ileus requiring total parenteral nutrition (13 patients), or referral for surgical consultation for possible colectomy (12 patients).
| Patient | Complications During the Hospital Stay | APACHE II Score | Monitored Unit | WBC* | K | Alb | Lactate | Cr | Hospital Stay (days) | Surgical Consult/Surgery | TPN for Colitis |
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| A | Sepsis, DIC, ARDS, intubation | 15 | Yes | 48 | 4 | 1.1 | 2.9 | 1.3 | 26 | No/No | Yes |
| B | Dehydration, weakness | 12 | No | 19 | 3.4 | 3.1 | 1.1 | 16 | No/No | Yes | |
| C | HTP, GI bleed, ischemic colitis, F | 22 | Yes | 21 | 2.7 | 2.5 | 1.3 | ESRD | 34 | Yes/No | Yes |
| D | AD, megacolon, HT, intubation, ARF | 18 | Yes | 25 | 3.4 | 1.4 | 1.1 | 2.6 | 52 | Yes/No | Yes |
| E | Exacerbation of CHF with respiratory distress, Bipap support, confusion | 21 | No | 10 | 2.6 | 2.3 | 1 | 11 | No/No | No | |
| F | Confusion, gout acute attack | 21 | No | 33 | 3.1 | 2.6 | ESRD | 15 | No/No | No | |
| G | Intubation, cardiac arrest, AF with RVR, PNA, ARF, DVT, dysphagia, PEG | 6 | Yes | 15 | 3.1 | 1.7 | 1.9 | 2.2 | 32 | Yes/No | Yes |
| H | Sepsis, intubation, ARF, PEG, toe amputation, PNA, pulmonary edema, TPN, vitamin D deficiency | 34 | Yes | 59 | 2.8 | 1.5 | 4 | 3.8 | 17 | No/No | Yes |
| I | CHF, transient third cranial nerve palsy, DIC | 20 | Yes | 52 | 3.2 | 3.3 | 0.7 | ESRD | 11 | No/No | No |
| J | Aspiration PNA, sepsis, DIC, intubation, MI, F, HT | 32 | Yes | 17 | 2.7 | 1.8 | 14 | ESRD | 21 | Yes/no | No |
| K | Intubation, cardiac arrest, ARF, DIC, AP, F | 30 | Yes | 25 | 2.1 | 2.1 | 13 | 3.6 | 10 | Yes/no | No |
| L | Intubation, ARF, HTP, MI, AD, F | 23 | Yes | 69 | 3.2 | 1.8 | 1.4 | 3.6 | 23 | Yes/no | No |
| M | Intubation, DIC, ARF, GI bleeding, hypothermia, AD | 23 | Yes | 47 | 2.8 | 1.4 | 1.4 | 4.3 | 23 | Yes/no | Yes |
| N | Intubation, HTP, AD, F, ARF with HD, AF, osteomyelitis | 23 | Yes | 46 | 2.9 | 1.5 | 1.5 | 2.7 | 27 | Yes/no | Yes |
| O | Intubation, ARF, 2 cardiac arrests, GI bleed, UTI, rhabdomyolysis, liver shock, AF | 31 | Yes | 49 | 2.5 | 1.1 | 2.3 | 2.9 | 25 | Yes/no | Yes |
| P | Bowel ischemia with bowel resection, ARF, MI, ischemic bowel, fluid overload, respiratory failure | 23 | Yes | 26 | 2.6 | 0.8 | 2.9 | 3.1 | 9 | Yes/Yes | Yes |
| Q | Pulmonary embolism | 39 | Yes | 23 | 2.8 | 1.1 | 8.1 | ESRD | 9 | No/No | Yes |
| R | Fungal peritonitis, aspiration pneumonia, cardiac arrest | 26 | Yes | 30 | 4.1 | 1.1 | 0.9 | ESRD | 36 | No/No | Yes |
| S | Intubation, pneumothorax, CRT, pressor‐dependent shock, ARF | 36 | Yes | 46 | 3.1 | 1.8 | 2.1 | 5.4 | 64 | Yes/Yes | No |
| T | Pressor‐dependent sepsis, pulmonary edema, ARF with HD | 36 | Yes | 35 | 3.3 | 1.3 | 1.8 | 5.5 | 11 | Yes/No | Yes |
| U | Sepsis | 34 | Yes | 58 | 3.1 | 2.2 | 3.3 | 1.6 | 9 | No/No | No |
Of the 21 patients treated with IVIG, 9 did not receive a surgical consultation either because they responded to medical treatment promptly (6 patients), were too unstable for surgery (2 patients), or because the patient/family refused surgery (1 patient). Of the 12 patients who received surgical consultation, 2 underwent surgery. The remainder did not proceed to surgery for the following reasons: they were deemed medically unstable for surgery (6 patients), declined surgery (2 patients), were diagnosed with cancer on colonoscopy (1 patient), or improved with medical treatment (1 patient).
The severity of systemic involvement was measured using the APACHE II score on day 1 of IVIG infusion. The mean APACHE II score was 25. Eighteen patients were in a monitored unit when IVIG was administered. The study group had laboratory results in keeping with those previously used to define severe colitis:4, 9, 19 leukocytosis (defined as white blood cell count higher than 12,000 cells/mL [mean = 36,000 cells/mL]), hypoalbuminemia (mean = 1.78 g/dL, SD = 0.68 g/dL), hypokalemia (mean = 3.02 mg/dL, SD = 0.47 g/dL), and acute renal failure (defined as serum creatinine level >1.5 mg/dL [mean = 2.98 mg/dL, SD = 1.42 g/dL]).
IVIG Use
Table 3 describes the treatment patients received for CDC as well as the total number of antibiotics used throughout the hospital stay. IVIG was used as adjuvant treatment (defined as IVIG administration within 4 days or less after CDC diagnosis) in 8 patients and as second‐line treatment (defined as IVIG administration more than 4 days after CDC diagnosis) in 13 patients. Metronidazole, vancomycin, cholestyramine, and probiotic treatment alone or in different combinations were used for an average of 8 days (SD = 8 days; range, 025 days) before IVIG infusion. The total IVIG dose administered varied depending on the prescribing attending, with a range of 200 mg/kg to 1250 mg/kg and a mode of 250 mg/kg for 1 to 3 days. An average of 5 (SD = 2) different antibiotics that were not active against C. difficile were used per patient without being discontinued after a CDC diagnosis was made. The 3 most common were: cephalosporins (cefazolin, ceftriaxone, cefepime), fluoroquinolones (levofloxacin), and combination antibiotics (piperacillin and tazobactam or ampicillin and sulbactam).
| Patient | Number of Antibiotics | Duration of Treatment Before IVIG (days) | Treatment Before IVIG (days) | Total CDC Treatment (days) | IgG Level | IVIG Dose |
|---|---|---|---|---|---|---|
| ||||||
| A | 5 | 7 | Metro (7), Vanc (7), Choles (2) | Oral and rectal Vanc (26,19), IV Metro (19), Choles (2), Lacto (6) | Low | 300 mg/kg for 1 day |
| B | 1 | 13 | Metro (13), Vanc (13) | Oral Vanc (17) and IV Metro (12) | 300 mg/kg for 1 day | |
| C | 3 | 7 | Metro (7), Vanc (3) | IV Metro (28),Vanc oral and enema (18,3), Lacto (10) | Low | 125 mg/kg for 5 days |
| D | 5 | 25 | Metro (25), Vanc (15) | Oral then IV Metro (10,15), oral Vanc (25), Choles (7) and Lacto (13) | Low | 200 mg/kg for 1 day |
| E | 1 | 4 | Metro (1), Vanc (4), Choles(4) | IV Metro (8), oral Vanc (10) and Choles (4) | 75 mg/kg for 5 days | |
| F | 4 | 2 | Metro (2), Vanc (1) | IV Metro (8) and oral Vanc (9) | 250 mg/kg for 5 days | |
| G | 3 | 17 | Metro (17), Vanc (14) | IV Metro (49) and oral Vanc (61) | 250 mg/kg for 2 days | |
| H | 5 | 1 | Metro (1), Vanc (1) | Oral Metro (18), oral Vanc (22 ), IV Metro (3) | 250 mg/kg for 3 days | |
| I | 6 | 1 | Metro (1) | Oral and IV Metro (7,9), Vanc oral and enema (8,3) | 250 mg/kg for 2 days | |
| J | 8 | 16 | Metro (14), Vanc (2), Lacto (6) | Oral then IV Metro (10,5), oral and rectal Vanc (3,1), Lacto (6) | 300 mg/ kg for 1 day | |
| K | 6 | 7 | Metro (7), Vanc (3) | Oral then IV Metro (10) and oral Vanc (9) | Normal | 400 mg/kg for 2 days |
| L | 4 | 0 | None | IV then oral Metro (7,10), oral Vanc (23), Choles (7), and Lacto (5) | 150 mg/kg for 5 days | |
| M | 6 | 1 | Metro (1) | IV Metro (23) and oral Vanc (9) | 250 mg/kg for 2 days | |
| N | 7 | 1 | Metro (1), Vanc (1) | IV Metro (16), oral Vanc (14), oral Metro (7) | 250 mg /kg for 2 days | |
| O | 6 | 7 | Metro (6), Vanc (4) | IV Metro (6) and oral Vanc (22) | Low | 250 mg/kg for 2 days |
| P | 3 | 6 | Metro (6), Vanc (6) | IV Metro (25) and oral Vanc (17) | 150 mg/kg fro 3 days | |
| Q | 3 | 4 | Metro (4), Vanc (3) | Oral Metro (8),Vanc oral and enema (6,3) | 250 mg/kg for 3 days | |
| R | 5 | 9 | Vanc (9) | Vanc oral and enema (5,4), IV Metro (2) | 250 mg/kg for 1 day | |
| S | 8 | 23 | Metro (23), Vanc (23) | Oral then IV Metro (12,22), oral Vanc (39) | 250 mg/kg for 3 days | |
| T | 4 | 6 | Metro (6), Vanc (1) | Oral Vanc and IV Metro (11) | 250 mg/kg for 1 day | |
| U | 4 | 2 | Metro (2), Vanc (2) | Oral Vanc and IV Metro (6) | 250 mg/kg for 3 days | |
Survival with IVIG Use
Nine patients (43%) survived their illness and were discharged from the hospital. They experienced complete clinical resolution after an average of 10 days from IVIG administration (range, 220 days) (Table 4). The other 12 patients (57%) died during the index hospitalization. The average length of stay was 23 (range, 964) days.0
| Patient | Disposition | Clearance of Clostridium difficile Colitis? | Days to Resolution |
|---|---|---|---|
| |||
| A | Alive | Loose BM persisted but diarrhea resolved 9 days after IVIG. | 9 |
| B | Alive | BM became formed and diarrhea resolved 48 hours after IVIG. | 2 |
| C | Alive | Diarrhea resolved 20 days post‐IVIG infusion. CAT scan: colonic thickening improved. | 20 |
| D | Alive | Diarrhea resolved on discharge. Response to IVIG started next day after administration. | 18 |
| E | Alive | Diarrhea improved next day after IVIG administration and resolved on discharge. | 5 |
| F | Alive | Diarrhea resolved 5 days after IVIG administration. | 5 |
| G | Alive | Diarrhea resolved. C. difficile test became negative. | 13 |
| H | Alive | Diarrhea resolved 2 days before discharge. | 15 |
| I | Alive | Diarrhea slowly improved and resolved 4 days before discharge. | 7 |
| J | Deceased | ||
| K | Deceased | ||
| L | Deceased | ||
| M | Deceased | ||
| N | Deceased | ||
| O | Deceased | ||
| P | Deceased | ||
| Q | Deceased | ||
| R | Deceased | ||
| S | Deceased | ||
| T | Deceased | ||
| U | Deceased | ||
| Study | Number of Patients | Age (SD) (years) | Male | Female | Severity Definition | IVIG Dose | Days to Resolution | Days IVIG Infused | Alive? (%) | Recurrence |
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
| Salcedo et al.22 | 2 | 63, 64 | 1 | 1 | Pancolitis or thumbprinting on CAT scan | 200300 mg/kg once | 12 | 59 | 100 | 1 out of 2 |
| McPherson et al.21 | 8 | 72 (12) | ? | ? | Pancolitis | 200400 mg/kg twice | 226 | 1165 | 75 | 2 out of 6 |
| Juang et al.25 | 18 | 67 (17.4) | 5 | 13 | Modified Rubin et al.9 criteria | 200300 mg/kg once | ? | ? | 83 | ? |
| Hassoun et al.24 | 1 | 72 | 1 | 0 | Pancolitis | 400 mg/kg once | 6 | 15 | 100 | None |
| Chandrasekar et al.23 | 1 | 67 | 0 | 1 | Shock requiring inotropic support and pseudomembranes on colonoscopy | 400 mg/kg for 5 doses | 16 | 35 | 100 | ? |
| This paper | 21 | 68 (16) | 7 | 14 | Pancolitis and APACHE II score | 300 mg/kg once; 250 mg/kg for 5 doses | 220 | 025 | 43 | ? |
To further assess the impact of IVIG on colitis resolution, we investigated all variables in the data set that may have been associated with mortality using univariate Cox regression analysis. Those variables were as follows: APACHE II score on the first day of IVIG infusion, age, sex, previous history of CDC, number of days before IVIG use, peak white blood cell count, serum potassium level and creatinine level, lactate level on first day of IVIG infusion, and number of antibiotics administered that are not active against CDC. Only the APACHE II score (P = 0.006) and lactate level on the day of IVIG infusion (P = 0.004) were (positively) associated with CDC mortality. The positive association between CDC mortality and APACHE II score remained significant (P = 0.04) after adjusting for sex, previous history of CDC, number of days before IVIG use, lactate level on first day of IVIG infusion, and number of antibiotics administered that are not active against CDC using a multivariate Cox regression analysis model. No adjustments were made for age, white blood cell count, potassium level, or creatinine level as those are included within the APACHE II score. The positive association between lactate level on the first day of IVIG infusion and CDC mortality was not statistically significant after adjusting for the factors listed above in the same model (P = 0.13).
Discussion
To our knowledge, the present study is the largest series published in the literature to date on the use of IVIG for severe CDC. It is also the first study to report a high mortality rate compared to the 5 previous smaller studies on this topic. In the first report on IVIG use for CDC, Leung et al.15 used IVIG to treat 5 pediatric patients suffering from chronic relapsing CDC. It was not until 7 years later, in 1997, that the first IVIG use for severe CDC was reported.22 Since then, a total of 13 works have been published on IVIG for CDC treatment, and only in 5 of these was IVIG administered for severe CDC treatment:2125 3 case reports, 1 case series, and 1 case‐control study. Although the 4 uncontrolled reports concluded that IVIG is beneficial for severe CDC, the only controlled study reported no significant difference between cases and controls for all‐cause mortality, length of stay, and colectomy rate.25
The definition of severe CDC varied between reports, making comparison difficult. McPherson et al.21 and Hassoun and Ibrahim24 defined severe disease as one causing pancolitis on CT scan either with or without megacolon. In the study by Juang et al.,25 disease severity was assessed using the modified criteria of Rubin et al.9 Salcedo et al.22 defined severe CDC as one causing pancolitis in one patient and thumbprinting on CT scan in another, whereas Chandrasekar et al.23 defined it as one causing shock requiring inotropic support and presence of pseudomembranes on colonoscopy.
The present report is unique in that it provided 2 scales to characterize disease in each patient. The first scale measured colonic involvement anatomically and physiologically using a combination of computerized axial tomography (CAT) scan findings, presence or absence of ileus or referral for possible colectomy. This is not a prognostic scale, however, since CAT scan findings have been previously shown to be poor predictors of treatment outcome.29 The second scale measured the severity of systemic involvement using a well‐validated and standardized scale, the APACHE II score. We have shown in this report that it is associated with prognosis in the context of IVIG use.
Our study reports a higher mortality rate than previously described, and suggests that risk stratification and patient selection are important before IVIG administration, since not all patients seem to benefit from this treatment as previously suggested by smaller case series. Previously, several physical findings and laboratory values were found to be associated with worse outcome in CDC. These were increasing age, immunosuppression, shock requiring vasopressors, peak white blood cell count, peak serum lactate level, hypoalbuminemia, a fall in serum albumin level of >1.1 g/dL at the onset of CDC symptoms, use of 3 or more antibiotics, comorbid disease, previous history of CDC, acute renal failure and hypotension, underlying altered or depressed mental status, abdominal pain or distention, white blood cell count over 20,000/mm3 or <1500/mm3 and/or a >10% band forms on the white blood cell differential count, and ascites or pneumatosis coli by abdominal imaging.9, 3033 Using the APACHE II scale for the same purpose has the advantage of utilizing a well‐validated and objective scale that is expected to measure the degree of systemic involvement more reliably compared to the clinical and laboratory values above.
Timing of IVIG infusion remains controversial. Due to the lack of randomized controlled trials, the current practice is guided by expert opinion, leading to wide variations between reports. Since the APACHE II score was positively associated with mortality in the setting of IVIG treatment, the same scale could be used to guide decisions regarding timing of IVIG infusion. Our results suggest that IVIG should be preferentially used while the APACHE II score is still relatively low. This association and the specific APACHE II score at which to initiate or hold treatment need to be validated in the setting of a randomized controlled study before being used in clinical practice.
Although the current study was not designed to test this theory, IVIG could be associated conceptually with treatment success for patients with severe disease that is still restricted to the colon (without other organ dysfunction or at least at an early stage of extracolonic organ failure and thus associated with a low APACHE II score) but not for severe colonic disease with secondary multiple organ failure (high APACHE II score). This may be because colonic disease is toxin‐mediated whereas secondary systemic involvement is mediated through toxin‐induced inflammatory mediators (interleukin‐8, macrophage‐inflammatory protein‐2, substance P, tumor necrosis factor‐alpha) released locally in the colon,3436 triggering a SIRS and hematogenous translocation of colonic bacteria,37 both of which are poorly responsive to immunoglobulin infusion. Along the same lines, waiting for failure of conventional therapy before IVIG use might result in IVIG treatment failure because of disease progression and secondary sepsis, at which point no treatment may be effective. No study thus far has addressed this issue specifically.
Overall, the combined cohort of patients with severe CDC treated with IVIG in the literature includes 51 patients (Table 4). The current report contributes 41% of these patients. The patients' average age was 68 years, with a 2 to 1 female‐male ratio. The dose of IVIG used varied largely also, with 400 mg/kg being the mode (range, 75400 mg/kg from 1 to 5 doses). This dose is significantly below the doses used in the treatment of other diseases, like Guillain‐Barr syndrome, myasthenia gravis, Kawasaki disease, autoimmune hemolytic anemia, agammaglobulinemia, and hypogammaglobulinemia, where the usual dose is 400 mg/kg for 5 days. The resolution of diarrhea in these cases occurred after an average of 9 (range, 142) days. The index hospitalization survival rate varied from 43% to 100%. Patients received standard treatment for an average of 13 (range, 065) days before IVIG infusion. Thirty‐two of 51 patients survived their illness (63%). Neither total IgG nor anti‐toxin A IgG levels were measured in any of the reports. Of the 32 patients who had clinical resolution, 3 (10%) experienced symptoms recurrence in a follow‐up period of 1 to 13 months. This number is most probably an underestimation of the true recurrence rate resulting from an incomplete reporting because there was no uniform or active recurrence ascertainment mechanism in any of the studies. The recurrences were at 10, 14, and 30 days posttreatment. Since standard treatment was not discontinued in any of the reports once IVIG was given, the relative contribution and the ideal timing for IVIG infusion are still unclear.
The mechanism of action of IVIG is passive immunization (with anti‐toxin A and anti‐toxin B antibodies present in the pooled immunoglobulin) of a host who is usually unable to mount an adequate protective immune response.15, 22 IVIG is formed from pooling immunoglobulin from several random donors. It has been shown that many such donors express high anti‐toxin A and anti‐toxin B antibody serum titers.38, 39 In addition, high levels of anti‐toxin A and anti‐toxin B antibodies were present in the IVIG preparations and the recipients after infusion.1517, 22 Although constituting only a small fraction of the total IVIG administered, these antitoxin antibodies are believed to neutralize toxin A and B and help the host recover from the disease. In fact, Babcock et al.40 used an experimental hamster model of CDC to demonstrate a mortality reduction from 100% to 55% postinfusion of combined anti‐toxin A and anti‐toxin B antibodies. While some early reports indicated that anti‐toxin B antibodies were the major determinants of protection against colitis,41 later reports correlated disease severity pathologically42 and clinically43, 44 with anti‐toxin A levels. Anti‐toxin B antibodies were later shown to play an adjunctive role in conferring immunity against CDC40, 45, 46 when added to anti‐toxin A antibodies, but not to have any significant role on their own.
However, IVIG has been shown to contain IgG, but not IgA, anti‐toxin A and anti‐toxin B antibodies while it is only the IgA class of anti‐toxin A antibodies, and not the IgG class, that could neutralize toxin A in vitro and in vivo.47, 48 Babcock et al.40 solved this apparent dilemma by showing that a combination of 3 different monoclonal IgG anti‐toxin A antibodies could neutralize toxin A activity in vitro and prevent disease in the hamster model in vivo. Each of the 3 antibodies recognized a different toxin A domain: the first neutralized toxin A enzymatic activity, while the second prevented toxin A binding to its receptor on enterocytes, and the third prevented toxin internalization after binding to the receptor.
Thus, the mechanism of action of IVIG is most likely through the transfer of IgG anti‐toxin A antibodies that gain access to the intestinal lumen presumably secondary to inflammation‐induced mucosal damage and neutralize toxin A. Transfer of yet undetected IgA anti‐toxin A antibodies that prevent toxin A from binding to its receptor is much less likely, although possible.
The present study has limitations. As in all retrospective studies, selection bias was unavoidable. In addition, the decision to initiate IVIG administration was dependent on the attending physician, who also decided the dose, leading to heterogeneity in the total dose of IVIG infused. Such heterogeneity, however, is primarily the result of a lack of a standard dose for IVIG infusion for CDC in the published literature, as reported above. In addition, since IVIG is not yet approved by the U.S. Food and Drug Administration (FDA) for the treatment of severe CDC, standard treatment was not discontinued in any of the reports to date, including ours.
Choosing appropriate controls for patients suffering from severe CDC is challenging. This patient population is usually frail, with severe and multiple underlying diseases. The deteriorating clinical condition (and subsequently the need for multiple antibiotics) may be either the result of or the cause of CDC. Furthermore, IVIG has been in short supply for several years and therefore it has been expensive, making its administration to the number of patients needed to design adequately‐powered controlled studies difficult. These are mainly the reasons no randomized, multicenter, placebo‐controlled trial on IVIG use in severe CDC has been conducted to date.
Conclusions
In the present study, we report the results of IVIG use for the treatment of 21 patients with severe CDC. This is the largest cohort to our knowledge in the literature. Unlike previous studies on the subject, the present report provided 2 scales for disease assessment: the first based on the extent of colonic involvement and the second measuring the severity of systemic involvement using the APACHE II score. The latter was positively associated with mortality in this context. Of the 21 study patients treated with IVIG, only 9 patients (43%) survived their illness. This is the highest reported mortality rate among all studies on this subject so far. Further studies on the ideal timing of IVIG infusion, dose, and patient selection are needed before accepting IVIG as a standard of care for severe CDC treatment. The role of APACHE II score in the decision to use IVIG is promising and should be validated in randomized controlled trials.
Acknowledgements
The authors thank Indrani Mukherjee, MD, for her help and expertise in proofreading the manuscript.
- ,,.Secular trends in hospital‐acquired Clostridium difficile disease in the United States, 1987–2001.J Infect Dis.2004;189:1585–1589.
- ,,, et al.Fulminant Clostridium difficile: an underappreciated and increasing cause of death and complications.Ann Surg.2002;235:363–372.
- ,,, et al.An epidemic, toxin gene‐variant strain of Clostridium difficile.N Engl J Med.2005;353:2433–2441.
- ,,, et al.Clostridium difficile‐associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity.CMAJ.2004;171:466–472.
- ,,.Comparison of two toxins produced by Clostridium difficile.Infect Immun.1981;34:1036–1043.
- ,,,.Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A.N Engl J Med.2000;342:390–397.
- ,,,.Human antibody response to Clostridium difficile toxin A in relation to clinical course of infection.Infect Immun.1994;62:384–389.
- ,,.Risk factors for Clostridium difficile carriage and C. difficile‐associated diarrhea in a cohort of hospitalized patients.J Infect Dis.1990;162:678–684.
- ,,.Severe Clostridium difficile colitis.Dis Colon Rectum.1995;38:350–354.
- ,, et al.A predominantly clonal multi‐institutional outbreak of Clostridium difficile‐associated diarrhea with high morbidity and mortality.N Engl J Med.2005;353:2442–2449.
- ,,.Factors associated with failure of metronidazole in Clostridium difficile‐associated disease.J Clin Gastroenterol.2004;38:414–418.
- ,,.Treatment of Clostridium difficile‐associated disease: old therapies and new strategies.Lancet Infect Dis.2005;5:549–557.
- ,,, et al.Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole.Clin Infect Dis.2005;40:1586–1590.
- ,,, et al.Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada.Clin Infect Dis.2005;40:1591–1597.
- ,,,,,.Treatment with intravenously administered gamma globulin of chronic relapsing colitis induced by Clostridium difficile toxin.J Pediatr.1991;118:633–637.
- ,,,.Gamma globulin administration in relapsing Clostridium difficile‐induced pseudomembranous colitis with a defective antibody response to toxin A.Acta Clin Belg.1995;50:36–39.
- ,,,.Recurrent Clostridium difficile infection in a patient with selective IgG1 deficiency treated with intravenous immune globulin and Saccharomyces boulardii.Clin Infect Dis.1995;20(suppl 2):S266–S268.
- .Intravenous immunoglobulin for recurrent Clostridium difficile diarrhoea.Gut.2002;51:456.
- .Descriptive study of intravenous immunoglobulin for the treatment of recurrent Clostridium difficile diarrhoea.J Antimicrob Chemother.2004;53:882–884.
- ,,.Intravenous immunoglobulin for resistant Clostridium difficile infection.Age Ageing.2006;35:85–86.
- ,,,,.Intravenous immunoglobulin for the treatment of severe, refractory, and recurrent Clostridium difficile diarrhea.Dis Colon Rectum.2006;49:640–645.
- ,,, et al.Intravenous immunoglobulin therapy for severe Clostridium difficile colitis.Gut.1997;41:366–370.
- ,,, et. al.Intravenous immunoglobulin therapy for refractory Clostridium difficile toxin colitis in chronic kidney disease: case reports and literature review.NDT Plus.2008;1:20–22.
- ,.Use of intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis.Am J Geriatr Pharmacother.2007;5:48–51.
- ,,, et al.Clinical outcomes of intravenous immune globulin in severe clostridium difficile‐associated diarrhea.Am J Infect Control.2007;35:131–137.
- ,,,.Successful treatment of Clostridium difficile colitis with intravenous immunoglobulin.J Gastrointestin Liver Dis.2008;17:353–359.
- ,,, et al.A durable response to relapsing Clostridium difficile colitis may require combined therapy with high‐dose oral Vancomycin and intravenous immune globulin.Infect Dis Clin Pract.2006;14:217–220.
- ,,,.APACHE II: a severity of disease classification system.Crit Care Med.1985;13:818–829.
- ,,,,,.Colonic abnormalities on CT in adult hospitalized patients with Clostridium difficile colitis: prevalence and significance of findings.AJR Am J Roentgenol.2006;186:1393–1400.
- ,,,.Clostridium difficile‐associated diarrhea: predictors of severity in patients presenting to the emergency department.Can J Gastroenterol.2003;17:369–373.
- ,,, et al.Impact of emergency colectomy on survival of patients with fulminant Clostridium difficile colitis during an epidemic caused by a hypervirulent strain.Ann Surg.2007;245:267–272.
- ,, et al.Risk factors for severity and relapse of pseudomembranous colitis in an elderly population.Colorectal Dis.2007;9:173–177.
- ,,,,.Prognostic criteria in Clostridium difficile colitis.Am J Gastroenterol.1996;91:460–464.
- ,,, et al.Clostridium difficile toxin a stimulates macrophage‐inflammatory protein‐2 production in rat intestinal epithelial cells.J Immunol.1998;160:6039–6045.
- ,,, et al.Increased substance P responses in dorsal root ganglia, intestinal macrophages during Clostridium difficile toxin a enteritis in rats.Proc Natl Acad Sci U S A.1997;94:4788–4793.
- ,,,,,.Cytokine response by human monocytes to Clostridium difficile toxin a and toxin B.Infect Immun.1991;59:3659–3666.
- ,,,.Bacterial translocation, intestinal microflora and morphological changes of intestinal mucosa in experimental models of Clostridium difficile infection.J Med Microbiol.1998;47:591–598.
- ,.Immunoglobulin G directed against toxins A and B of Clostridium difficile in the general population and patients with antibiotic‐associated diarrhea.Diagn Microbiol Infect Dis.1994;18:205–209.
- ,,, et al.Serum antibody response to toxins A and B of Clostridium difficile.J Infect Dis.1983;148:93–100.
- ,,, et al.Human monoclonal antibodies directed against toxins A and B prevent Clostridium difficile‐induced mortality in hamsters.Infect Immun.2006;74:6339–6347.
- ,,,.Serum antibody response to Clostridium difficile toxins in patients with Clostridium difficile diarrhoea.Infection.1985;13:97–101.
- ,,,.Relationship between levels of Clostridium difficile toxin A and toxin B and cecal lesions in gnotobiotic mice.Infect Immun.1989;57:2123–2127.
- ,,,,.Protection against experimental pseudomembranous colitis in gnotobiotic mice by use of monoclonal antibodies against Clostridium difficile toxin A.Infect Immun.1991;59:1192–1195.
- ,,.Systemic and mucosal antibody responses to toxin A in patients infected with Clostridium difficile.J Infect Dis.1992;166:1287–1294.
- ,,, et al.Transcutaneous immunization with Clostridium difficile toxoid A induces systemic and mucosal immune responses and toxin A‐neutralizing antibodies in mice.Infect Immun.2007;75:2826–2832.
- ,,, et al.Serum antitoxin antibodies mediate systemic and mucosal protection from Clostridium difficile disease in hamsters.Infect Immun.1999;67:527–538.
- ,,,.Human colonic aspirates containing immunoglobulin A antibody to Clostridium difficile toxin A inhibit toxin A‐receptor binding.Gastroenterology.1992;102:35–40.
- ,,,,.Selective neutralization of a bacterial enterotoxin by serum immunoglobulin A in response to mucosal disease.Infect Immun.1995;63:3166–3173.
- ,,.Secular trends in hospital‐acquired Clostridium difficile disease in the United States, 1987–2001.J Infect Dis.2004;189:1585–1589.
- ,,, et al.Fulminant Clostridium difficile: an underappreciated and increasing cause of death and complications.Ann Surg.2002;235:363–372.
- ,,, et al.An epidemic, toxin gene‐variant strain of Clostridium difficile.N Engl J Med.2005;353:2433–2441.
- ,,, et al.Clostridium difficile‐associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity.CMAJ.2004;171:466–472.
- ,,.Comparison of two toxins produced by Clostridium difficile.Infect Immun.1981;34:1036–1043.
- ,,,.Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A.N Engl J Med.2000;342:390–397.
- ,,,.Human antibody response to Clostridium difficile toxin A in relation to clinical course of infection.Infect Immun.1994;62:384–389.
- ,,.Risk factors for Clostridium difficile carriage and C. difficile‐associated diarrhea in a cohort of hospitalized patients.J Infect Dis.1990;162:678–684.
- ,,.Severe Clostridium difficile colitis.Dis Colon Rectum.1995;38:350–354.
- ,, et al.A predominantly clonal multi‐institutional outbreak of Clostridium difficile‐associated diarrhea with high morbidity and mortality.N Engl J Med.2005;353:2442–2449.
- ,,.Factors associated with failure of metronidazole in Clostridium difficile‐associated disease.J Clin Gastroenterol.2004;38:414–418.
- ,,.Treatment of Clostridium difficile‐associated disease: old therapies and new strategies.Lancet Infect Dis.2005;5:549–557.
- ,,, et al.Relatively poor outcome after treatment of Clostridium difficile colitis with metronidazole.Clin Infect Dis.2005;40:1586–1590.
- ,,, et al.Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada.Clin Infect Dis.2005;40:1591–1597.
- ,,,,,.Treatment with intravenously administered gamma globulin of chronic relapsing colitis induced by Clostridium difficile toxin.J Pediatr.1991;118:633–637.
- ,,,.Gamma globulin administration in relapsing Clostridium difficile‐induced pseudomembranous colitis with a defective antibody response to toxin A.Acta Clin Belg.1995;50:36–39.
- ,,,.Recurrent Clostridium difficile infection in a patient with selective IgG1 deficiency treated with intravenous immune globulin and Saccharomyces boulardii.Clin Infect Dis.1995;20(suppl 2):S266–S268.
- .Intravenous immunoglobulin for recurrent Clostridium difficile diarrhoea.Gut.2002;51:456.
- .Descriptive study of intravenous immunoglobulin for the treatment of recurrent Clostridium difficile diarrhoea.J Antimicrob Chemother.2004;53:882–884.
- ,,.Intravenous immunoglobulin for resistant Clostridium difficile infection.Age Ageing.2006;35:85–86.
- ,,,,.Intravenous immunoglobulin for the treatment of severe, refractory, and recurrent Clostridium difficile diarrhea.Dis Colon Rectum.2006;49:640–645.
- ,,, et al.Intravenous immunoglobulin therapy for severe Clostridium difficile colitis.Gut.1997;41:366–370.
- ,,, et. al.Intravenous immunoglobulin therapy for refractory Clostridium difficile toxin colitis in chronic kidney disease: case reports and literature review.NDT Plus.2008;1:20–22.
- ,.Use of intravenous immunoglobulin for the treatment of severe Clostridium difficile colitis.Am J Geriatr Pharmacother.2007;5:48–51.
- ,,, et al.Clinical outcomes of intravenous immune globulin in severe clostridium difficile‐associated diarrhea.Am J Infect Control.2007;35:131–137.
- ,,,.Successful treatment of Clostridium difficile colitis with intravenous immunoglobulin.J Gastrointestin Liver Dis.2008;17:353–359.
- ,,, et al.A durable response to relapsing Clostridium difficile colitis may require combined therapy with high‐dose oral Vancomycin and intravenous immune globulin.Infect Dis Clin Pract.2006;14:217–220.
- ,,,.APACHE II: a severity of disease classification system.Crit Care Med.1985;13:818–829.
- ,,,,,.Colonic abnormalities on CT in adult hospitalized patients with Clostridium difficile colitis: prevalence and significance of findings.AJR Am J Roentgenol.2006;186:1393–1400.
- ,,,.Clostridium difficile‐associated diarrhea: predictors of severity in patients presenting to the emergency department.Can J Gastroenterol.2003;17:369–373.
- ,,, et al.Impact of emergency colectomy on survival of patients with fulminant Clostridium difficile colitis during an epidemic caused by a hypervirulent strain.Ann Surg.2007;245:267–272.
- ,, et al.Risk factors for severity and relapse of pseudomembranous colitis in an elderly population.Colorectal Dis.2007;9:173–177.
- ,,,,.Prognostic criteria in Clostridium difficile colitis.Am J Gastroenterol.1996;91:460–464.
- ,,, et al.Clostridium difficile toxin a stimulates macrophage‐inflammatory protein‐2 production in rat intestinal epithelial cells.J Immunol.1998;160:6039–6045.
- ,,, et al.Increased substance P responses in dorsal root ganglia, intestinal macrophages during Clostridium difficile toxin a enteritis in rats.Proc Natl Acad Sci U S A.1997;94:4788–4793.
- ,,,,,.Cytokine response by human monocytes to Clostridium difficile toxin a and toxin B.Infect Immun.1991;59:3659–3666.
- ,,,.Bacterial translocation, intestinal microflora and morphological changes of intestinal mucosa in experimental models of Clostridium difficile infection.J Med Microbiol.1998;47:591–598.
- ,.Immunoglobulin G directed against toxins A and B of Clostridium difficile in the general population and patients with antibiotic‐associated diarrhea.Diagn Microbiol Infect Dis.1994;18:205–209.
- ,,, et al.Serum antibody response to toxins A and B of Clostridium difficile.J Infect Dis.1983;148:93–100.
- ,,, et al.Human monoclonal antibodies directed against toxins A and B prevent Clostridium difficile‐induced mortality in hamsters.Infect Immun.2006;74:6339–6347.
- ,,,.Serum antibody response to Clostridium difficile toxins in patients with Clostridium difficile diarrhoea.Infection.1985;13:97–101.
- ,,,.Relationship between levels of Clostridium difficile toxin A and toxin B and cecal lesions in gnotobiotic mice.Infect Immun.1989;57:2123–2127.
- ,,,,.Protection against experimental pseudomembranous colitis in gnotobiotic mice by use of monoclonal antibodies against Clostridium difficile toxin A.Infect Immun.1991;59:1192–1195.
- ,,.Systemic and mucosal antibody responses to toxin A in patients infected with Clostridium difficile.J Infect Dis.1992;166:1287–1294.
- ,,, et al.Transcutaneous immunization with Clostridium difficile toxoid A induces systemic and mucosal immune responses and toxin A‐neutralizing antibodies in mice.Infect Immun.2007;75:2826–2832.
- ,,, et al.Serum antitoxin antibodies mediate systemic and mucosal protection from Clostridium difficile disease in hamsters.Infect Immun.1999;67:527–538.
- ,,,.Human colonic aspirates containing immunoglobulin A antibody to Clostridium difficile toxin A inhibit toxin A‐receptor binding.Gastroenterology.1992;102:35–40.
- ,,,,.Selective neutralization of a bacterial enterotoxin by serum immunoglobulin A in response to mucosal disease.Infect Immun.1995;63:3166–3173.
Copyright © 2010 Society of Hospital Medicine
Evidence‐Based Strategies for VTE
Pretest probability assessment is an important first step in the diagnosis of venous thromboembolism (VTE) and models incorporating Wells criteria1 can be used accurately in emergency department (ED) and inpatient settings.2 Gestalt has the disadvantage of poor interobserver reliability,3 and use of clinical prediction rules has been advocated instead.4 In academic institutions, trainees frequently first evaluate patients with suspected VTE, and although gestalt improves with degree of experience, the performance of gestalt in 1 study5 was better for attendings than interns or residents (for whom it was equivalent), suggesting that structured pretest probability assessment may be more important for trainees.
From an imaging perspective, multidetector computed tomography (CT),6 is more accurate than ventilation perfusion (VP) scanning7, 8 in diagnosing VTE in any setting, including the critically ill.9, 10 Lower extremity CT venography (LECTV) has comparable sensitivity to contrast venography and sonography,11 and in combination with computed tomographic pulmonary angiography (CTPA) is important when imaging results are discordant with pretest probability.12 Guidelines for diagnostic pathways in VTE based on published literature incorporating D‐dimer testing have been updated recently,13 the degree of adoption and use of diagnostic algorithms among trainees has been understudied.
Clinical trials14, 15 have confirmed the safety and efficacy of low molecular weight heparin (LMWH) in the treatment of pulmonary embolism (PE) in inpatients, but the degree of adoption of this therapy is unclear. The primary objective of our survey of was to assess the knowledge, attitudes, practices, and preferences of trainees and attendings who order and evaluate the results of diagnostic studies in the management of VTE. A secondary objective was to assess willingness to use LMWH to treat VTE in the inpatient setting among non‐ED respondents.
Methods
Survey Design and Administration
The study was cross‐sectional and was approved by the institutional review board. The survey was paper‐based and anonymous, and the requirement for written informed consent was waived. The survey instrument was reviewed for clarity, lack of bias, and accuracy by a panel of hospitalists at the State University of New York (SUNY) Downstate Medical Center. Closed‐ended questions were used, including a 5‐point Likert scale (1 = strongly agree, 5 = strongly disagree) and multiple‐choice queries. Between October 2006 and March 2008, paper‐based survey questionnaires were distributed to internal medicine (IM) attendings, residents, and students from institutions in the New York, New Jersey, and Connecticut tri‐state area taking medicine review courses in New York City and attending grand rounds at SUNY Downstate. Of 319 non‐ED respondents, 116 (30%) were from the SUNY Downstate system. All third‐year medical students (58/116) were from the SUNY Downstate training program. representing 5 different training institutions for medical students and 4 institutions for residents. ED physicians (n = 46) were selected randomly and telephoned at work and questioned about their practices with an abbreviated version of the survey. Response rates were 80% for the paper‐based surveys and 20% for the ED physicians. Data was recorded into an electronic database (Microsoft Access; Microsoft Corp., Redmond, WA). Simple clinical vignettes were used to assess diagnostic and therapeutic strategies in the setting of VTE for non‐ED respondents only.
Data Analysis
Descriptive statistics were used to report respondents' demographic information and work environments. Data are expressed as proportions, means SD, or medians with interquartile range. Differences in response levels between groups were compared by Fisher's exact test, chi square test, or the Kruskal‐Wallis test, where appropriate. Two‐sided P values of less than 0.05 were considered significant. Since no difference in the ability of residents and interns to predict PE has been noted,5 both groups were analyzed together, as were third‐year and fourth‐year medical students. JMP version 7.0 software (SAS Institute, Cary, NC) was used to perform all analyses.
Results
Table 1 lists the characteristics of respondents. Medical attendings reported practicing in up to 5 different institutions, and residents reported rotating through up to 10 different institutions during their residency. Students reported rotating through up to 11 different institutions.
| n (%) | Institutions Rotated Through [median (IQR)] | |
|---|---|---|
| ||
| Emergency department attendings | 46 | |
| Medicine attendings | 46 | 1 (12) |
| Residents | 139 | 3 (23) |
| PGY1 | 39 (28) | |
| PGY2 | 27 (19) | |
| PGY3 | 34 (24) | |
| PGY4 | 3 (2) | |
| PGY5 | 19 (14) | |
| Year not checked | 17 (13) | |
| Medical students | 134 | 3 (15) |
| Third year | 58 (43) | |
| Fourth year | 76 (57) | |
Pretest Probability Assessment
Table 2 depicts differences between ED and IM attending responses. More than 60% of all attendings used no structured pretest probability assessment; the rest reported using the Wells criteria. An equivalent proportion of ED and IM attendings thought prediction rules were too complex to use (P = 0.2). Years of attending experience did not predict responses regarding perceptions of the complexity of prediction rules (P = 0.5). More IM attendings than residents or students felt that prediction rules were too complex for routine use (P = 0.02). Among trainees, significantly more residents than students reported using the Wells model (P < 0.001); 40% of residents did not use any model. Advanced years in training among residents did not predict an increased likelihood of using prediction rules.
| ED Attendings (n = 46) | IM Attendings (n = 43) | P Value | |
|---|---|---|---|
| |||
| Years of experience [median (interquartile range)] | 12.5 (7.521) | 6 (214) | <0.001 |
| Academic practice [n (%)] | 23 (50) | 27 (63) | 0.7 |
| Do not use prediction rules [n (%)] | 28 (61) | 28 (65) | 0.8 |
| Prediction rules too complex to use [n (% agree)] | 22 (48) | 13 (30) | 0.5 |
| Aware of a written algorithm for diagnosis of VTE [n (%)] | 2 (4) | 21 (50) | <0.001 |
D‐Dimer Testing
Among trainees, 25% of residents and students and 20% of IM attendings were unaware of the sensitivity or specificity of D‐dimer assays in use in their institution (P = 0.8), and 70% of ED attendings were unaware. Almost all residents, students, and IM attendings were unable to identify the name of the D‐dimer test used in their institutions (>95% in each category); while 54% of ED attendings were also unable to do so (P < 0.0001).
Imaging Strategies
Table 3 depicts responses regarding knowledge about various VTE imaging strategies. The majority of students responded that they would use VP scanning as the initial modality and a substantial number of attendings and residents would too. All ED attendings reported using CTPA as the initial modality of choice. A substantial number of students, residents, and IM attendings did not know whether LECTV had to be ordered separately or was done by default and a large proportion incorrectly surmised that the sensitivity of LECTV was not equivalent to lower extremity Doppler.
| ED Attending (n = 46) | IM Attending (n = 43) | Residents (n = 139) | Medical Students (n = 134) | P value | |
|---|---|---|---|---|---|
| |||||
| VP scanning test of choice in suspected PE [n (%)] | 0 (0) | 9 (22) | 24 (17) | 78 (58) | <0.001 |
| CTV ordered separate from or with CTPA by default [n (% unaware)] | 12 (27) | 53 (38) | 96 (72) | <0.001 | |
| Sensitivity of CTV = LE US [n (% agree)] | 22 (51) | 69 (50) | 42 (31) | 0.01 | |
Clinical Vignettes
Table 4 depicts responses by non‐ED respondents to various clinical scenarios presented. Faced with a dyspneic patient 2 days after a hip fracture and a negative CTPA alone, almost 25% of all respondents would incorrectly withhold anticoagulation. In outpatients with low probability Wells score for DVT and a negative D‐dimer, substantial proportions of all respondents would incorrectly order further imaging. For treatment of inpatients with DVT and non‐massive PE, 17% of students disagreed that LMWH was appropriate, and similar proportions of residents (12%) and IM attendings (13%) disagreed.
| IM Attending (n = 46) | Residents (n = 139) | Medical Students (n = 134) | P Value | |
|---|---|---|---|---|
| ||||
| Anticoagulate high risk patient with negative CTPA alone [n (% agree)] | 35 (76) | 104 (75) | 99 (74) | 0.9 |
| Order further imaging in outpatient with negative D‐dimer and low probability for DVT [n (% agree)] | 25 (54) | 68 (49) | 59 (44) | 0.6 |
| LMWH appropriate for DVT and non‐massive PE among inpatients [n (% agree)] | 40 (88) | 119 (86) | 88 (66) | 0.02 |
Discussion
Pretest Probability Assessment
Our findings that only a minority of trainees and practicing physicians calculate pretest probability using a prediction score translate into potentially inferior (and more costly) care for patients with suspected VTE. This is especially true for academic institutions, where trainees are ordinarily first responders. Among practitioners in the United States,16 72.5% prefer an unstructured approach to pretest assessment, whereas 22.9% use published prediction rules. In this survey, more residents than students or attendings used the Wells criteria for pretest probability testing. The majority of ED attendings surveyed (61%) used no structured pretest probability assessment, consistent with a retrospective study published recently17; however, this may have been because of the relatively experienced group sampled (median number of years in practice was 12.5 compared to 6 years among IM attendings). Students may not be receiving training to use prediction rules because attendings may feel they are too complex to use and/or may not use these rules themselves. A substantial proportion of residents (40% in our study) do not use them. Awareness of written algorithms was reported by a minority of all respondents, but did not translate into greater use of prediction rules.
D‐Dimer Testing
Only a few highly sensitive quantitative assays (VIDAS, Tinaquant, Liatest, and Simplired)1821 have been validated in large clinical trials incorporating structured pretest probability assessment and CTPA. Guidelines for diagnosis of VTE recommend that physicians be informed about the type of D‐dimer being used in their practice setting given the substantial variation in D‐dimer sensitivity.22 The sensitivities of quantitative enzyme‐linked immunosorbent assays (ELISAs) are clinically and statistically superior to other types of D‐dimer tests among patients with VTE.23 Over 20% of all non‐ED respondents did not know the sensitivity and specificity of the D‐dimer assay in use in their respective institutions and most (>70% in each category) could not name the assay, resulting in potentially inappropriate decision making if nonquantitative ELISA D‐dimers were used alone or gestalt were used, especially by trainees.
Imaging Strategies
Weiss et al.24 surveyed U.S. clinicians and found a clear preference for CTPA as the initial imaging modality in patients with suspected PE but did not include the trainee perspective. As level of training progressed, we found a decrease in the percentage of respondents that preferred VP scanning over CTPA as the first test of choice; however, 25% of residents and 17% of attendings still designated VP scanning as their first choice. The perception of the majority of students in our survey is that VP scanning is the preferred initial test. We conjecture that students do not receive the pertinent training from supervising clinicians in this regard. All ED attendings surveyed used CTPA as their first choice of imaging. Knowledge about whether LECTV was ordered separately from CTPA or done by default was lacking in over 25% of all non‐ED respondents. The lower the level of experience, the more incorrect answers were given. Apropos of the PIOPED II study,12 lack of awareness about lower extremity imaging in association with CTPA may therefore contribute to inappropriate decision making, especially in patients with high pretest probability of PE and a negative CTPA alone.
Clinical Vignettes
Two studies25, 26 analyzed outcomes in patients with low to intermediate pretest probability PE and negative CTPA alone who did not receive anticoagulation. Both suggest that withholding anticoagulation in these patients is safe. The 25% of non‐ED respondents who would consider withholding anticoagulation in high‐risk settings translates into a large number of potentially inappropriate decisions, especially if gestalt is used in pretest probability assessment. This is in line with recommendations from the PIOPED II study that lower extremity imaging and, if necessary, serial lower extremity ultrasonography be performed in high‐risk groups.11, 12 A negative validated D‐dimer study and a low pretest probability exclude the need for further testing in outpatients with suspected DVT27; however, 50% of all respondents would order further testing. Thus, regardless of experience, a disparity exists between practice and published literature among both trainees and attendings, especially since further imaging in this setting is not cost effective.28
Use of LMWH
In a cohort of 946 inpatients in one study,29 only 56.1% of inpatients with DVT or PE were treated with LMWH. In our survey a substantial minority of IM attendings, residents, and students (12%, 13%, and 17%, respectively) would not consider LMWH one of the prefered therapies for VTE in the right clinical setting.
Limitations
The cross‐sectional nature of the survey and localization of non‐ED respondents to the New York, New Jersey, and Connecticut tri‐state region, limits generalizability to other geographic regions of the country. Responses of ED attendings were sampled nationally. The attendings (ED and IM) sampled were a relatively experienced group (6‐12 years of practice) and this may explain the relatively low adoption of prediction rules reflecting the use of gestalt in this group. Additionally, over time, knowledge (and use) of validated D‐dimer assays may have increased in the practices evaluated. Among non‐ED respondents, 30% (116/319) were from a single training program (SUNY Downstate) and the responses of these respondents may reflect practice in the institutions sampled, limiting nationwide generalization with the potential for selection bias. The low rate of response from ED physicians (20%) was presumably a result of being called at work. We believe the responses are still a valuable insight into the real‐time practices of the clinicians surveyed and do not preclude a meaningful comparison to the rest of the respondents especially given the significant differences between ED and IM attending knowledge and awareness (Tables 3 and 4).
Conclusions
Our survey identifies the use of evidence‐based strategies in the management of VTE among trainees, a perspective that has been lacking in other studies of physicians in practice.16, 24, 27 Substantial variability in attending practice identified in this survey may impede the adoption of a structured approach to the diagnosis of VTE among trainees, and this survey raises major concerns about mechanisms of diagnosis of VTE. Caprini et al.29 believe that physician knowledge, attitudes, and beliefs are partially responsible for the gap between actual practice and international guidelines.27 The results of our survey extend this suggestion to trainees and imply that supervisor attitudes may negatively influence trainee practices. Development of written protocols or standardized pathway order sets based on published evidence‐based guidelines13 in the management of VTE could improve the use of structured pretest probability determination and use of evidence‐based strategies among trainees. Finally, comparisons of outcomes using algorithms and usual practice could provide valuable, clinically important data that could inform clinical practice.
- ,,, et al.Assessment of pretest probability of pulmonary embolism in the emergency department by physicians in training using the Wells model.Thromb Res.2007;120(2):173–179.
- ,,,.A prospective reassessment of the utility of the Wells score in identifying pulmonary embolism.Med J Aust.2007;187(6):333–336.
- ,,,,.The interobserver reliability of pretest probability assessment in patients with suspected pulmonary embolism.Thromb Res.2005;116(2):101–107.
- ,,, et al.Does this patient have pulmonary embolism?JAMA.2003;290(21):2849–2858.
- ,,,,.Does a physician's ability to accurately assess the likelihood of pulmonary embolism increase with training?Acad Med.2000;75(12):1199–1205.
- ,,, et al.Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and D‐dimer.Ann Intern Med.2001;135(2):98–107.
- ,,, et al.Pulmonary embolism revealed on helical CT angiography: comparison with ventilation perfusion radionuclide lung scanning.AJR Am J Roentgenol.2000;174:1041–1047.
- ,,, et al.Clinical validity of a negative computed tomography scan in patients with suspected pulmonary embolism: a systematic review.JAMA.2005;293(16):2012–2017.
- ,,,,.Multidetector CT: a new gold standard in the diagnosis of pulmonary embolism? State of the art and diagnostic algorithms.Radiol Med.2005;109(1–2):49–61.
- ,,,,.Multidetector row CT pulmonary angiography and indirect venography for the diagnosis of venous thromboembolic disease in intensive care unit patients.Acad Radiol.2006;13(4):486–495.
- ,,.The role of multidetector computed tomography angiography for the diagnosis of pulmonary embolism.Semin Nucl Med.2008;38:418–431.
- ,,, et al.Multidetector computed tomography for acute pulmonary embolism.N Engl J Med.2006;354:2317–2327.
- Institute for Clinical Systems Improvement. Venous Thromboembolism Diagnosis and Treatment. Available at: http://www.icsi.org. Accessed October2009.
- The Columbus Investigators.Low‐molecular‐weight heparin in the treatment of patients with venous thromboembolism.N Engl J Med.1997;337:657–662.
- ,,, et al.A comparison of low‐molecular‐weight heparin with unfractionated heparin for acute pulmonary embolism. The THESEE Study Group.N Engl J Med.1997;337(10):663–669.
- ,,,,,.Pretest risk assessment in suspected acute pulmonary embolism.Acad Radiol.2008;15(1):3–14.
- ,,,.Is pretest probability assessment on emergency department patients with suspected venous thromboembolism documented before SimpliRED D‐dimer testing?CJEM.2008;10(6):519–523.
- ,,,,,.Exclusion of DVT with D‐dimer testing: comparison of 13 D‐dimer methods in 99 outpatients suspected of DVT using venography as a standard.Thromb Haemost.2000;83:191–198.
- ,,, et al.Multidetector‐row computed tomography in suspected pulmonary embolism.N Engl J Med.2005;352(17):1760–1768.
- ,,, et al.Simple and safe exclusion of pulmonary embolism in outpatients using quantitative D‐dimer and Wells' simplified decision rule.Thromb Haemost.2007;97(1):146–150.
- ,,,,;ANTELOPE Study Group. The performance of two rapid quantitative D‐dimer assays in 287 patients with clinically suspected pulmonary embolism.Thromb Res.2002;107(6):283–286.
- ,,, et al.Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians.Ann Fam Med.2007;5:57–62.
- ,,, et al.D‐dimer for the exclusion of acute venous thrombosis and pulmonary embolism. A systematic review.Ann Intern Med.2004;140:589–602.
- ,,, et al.CT pulmonary angiography is the first‐line imaging test for acute pulmonary embolism: a survey of US clinicians.Acad Radiol.2006;13:434–446.
- ,,, et al.Outcomes after withholding anticoagulation from patients with suspected pulmonary embolism and negative computed tomographic findings: a cohort study.Mayo Clin Proc.2002;77:130–138.
- ,,,.Meta‐analysis: outcomes in patients with suspected pulmonary embolism managed with computed tomographic pulmonary angiography.Ann Intern Med.2004;141:866–874.
- ,,, et al.Does this patient have deep vein thrombosis?JAMA.2006;295(2):199–207.
- ,,, et al.Measurement of the clinical and cost‐effectiveness of non‐invasive diagnostic testing strategies for deep vein thrombosis.Health Technol Assess.2006;10(15):1–168.
- ,,, et al.NABOR Steering Committee. Treatment of venous thromboembolism: adherence to guidelines and impact of physician knowledge, attitudes, and beliefs.J Vasc Surg.2005;42(4):726–733.
Pretest probability assessment is an important first step in the diagnosis of venous thromboembolism (VTE) and models incorporating Wells criteria1 can be used accurately in emergency department (ED) and inpatient settings.2 Gestalt has the disadvantage of poor interobserver reliability,3 and use of clinical prediction rules has been advocated instead.4 In academic institutions, trainees frequently first evaluate patients with suspected VTE, and although gestalt improves with degree of experience, the performance of gestalt in 1 study5 was better for attendings than interns or residents (for whom it was equivalent), suggesting that structured pretest probability assessment may be more important for trainees.
From an imaging perspective, multidetector computed tomography (CT),6 is more accurate than ventilation perfusion (VP) scanning7, 8 in diagnosing VTE in any setting, including the critically ill.9, 10 Lower extremity CT venography (LECTV) has comparable sensitivity to contrast venography and sonography,11 and in combination with computed tomographic pulmonary angiography (CTPA) is important when imaging results are discordant with pretest probability.12 Guidelines for diagnostic pathways in VTE based on published literature incorporating D‐dimer testing have been updated recently,13 the degree of adoption and use of diagnostic algorithms among trainees has been understudied.
Clinical trials14, 15 have confirmed the safety and efficacy of low molecular weight heparin (LMWH) in the treatment of pulmonary embolism (PE) in inpatients, but the degree of adoption of this therapy is unclear. The primary objective of our survey of was to assess the knowledge, attitudes, practices, and preferences of trainees and attendings who order and evaluate the results of diagnostic studies in the management of VTE. A secondary objective was to assess willingness to use LMWH to treat VTE in the inpatient setting among non‐ED respondents.
Methods
Survey Design and Administration
The study was cross‐sectional and was approved by the institutional review board. The survey was paper‐based and anonymous, and the requirement for written informed consent was waived. The survey instrument was reviewed for clarity, lack of bias, and accuracy by a panel of hospitalists at the State University of New York (SUNY) Downstate Medical Center. Closed‐ended questions were used, including a 5‐point Likert scale (1 = strongly agree, 5 = strongly disagree) and multiple‐choice queries. Between October 2006 and March 2008, paper‐based survey questionnaires were distributed to internal medicine (IM) attendings, residents, and students from institutions in the New York, New Jersey, and Connecticut tri‐state area taking medicine review courses in New York City and attending grand rounds at SUNY Downstate. Of 319 non‐ED respondents, 116 (30%) were from the SUNY Downstate system. All third‐year medical students (58/116) were from the SUNY Downstate training program. representing 5 different training institutions for medical students and 4 institutions for residents. ED physicians (n = 46) were selected randomly and telephoned at work and questioned about their practices with an abbreviated version of the survey. Response rates were 80% for the paper‐based surveys and 20% for the ED physicians. Data was recorded into an electronic database (Microsoft Access; Microsoft Corp., Redmond, WA). Simple clinical vignettes were used to assess diagnostic and therapeutic strategies in the setting of VTE for non‐ED respondents only.
Data Analysis
Descriptive statistics were used to report respondents' demographic information and work environments. Data are expressed as proportions, means SD, or medians with interquartile range. Differences in response levels between groups were compared by Fisher's exact test, chi square test, or the Kruskal‐Wallis test, where appropriate. Two‐sided P values of less than 0.05 were considered significant. Since no difference in the ability of residents and interns to predict PE has been noted,5 both groups were analyzed together, as were third‐year and fourth‐year medical students. JMP version 7.0 software (SAS Institute, Cary, NC) was used to perform all analyses.
Results
Table 1 lists the characteristics of respondents. Medical attendings reported practicing in up to 5 different institutions, and residents reported rotating through up to 10 different institutions during their residency. Students reported rotating through up to 11 different institutions.
| n (%) | Institutions Rotated Through [median (IQR)] | |
|---|---|---|
| ||
| Emergency department attendings | 46 | |
| Medicine attendings | 46 | 1 (12) |
| Residents | 139 | 3 (23) |
| PGY1 | 39 (28) | |
| PGY2 | 27 (19) | |
| PGY3 | 34 (24) | |
| PGY4 | 3 (2) | |
| PGY5 | 19 (14) | |
| Year not checked | 17 (13) | |
| Medical students | 134 | 3 (15) |
| Third year | 58 (43) | |
| Fourth year | 76 (57) | |
Pretest Probability Assessment
Table 2 depicts differences between ED and IM attending responses. More than 60% of all attendings used no structured pretest probability assessment; the rest reported using the Wells criteria. An equivalent proportion of ED and IM attendings thought prediction rules were too complex to use (P = 0.2). Years of attending experience did not predict responses regarding perceptions of the complexity of prediction rules (P = 0.5). More IM attendings than residents or students felt that prediction rules were too complex for routine use (P = 0.02). Among trainees, significantly more residents than students reported using the Wells model (P < 0.001); 40% of residents did not use any model. Advanced years in training among residents did not predict an increased likelihood of using prediction rules.
| ED Attendings (n = 46) | IM Attendings (n = 43) | P Value | |
|---|---|---|---|
| |||
| Years of experience [median (interquartile range)] | 12.5 (7.521) | 6 (214) | <0.001 |
| Academic practice [n (%)] | 23 (50) | 27 (63) | 0.7 |
| Do not use prediction rules [n (%)] | 28 (61) | 28 (65) | 0.8 |
| Prediction rules too complex to use [n (% agree)] | 22 (48) | 13 (30) | 0.5 |
| Aware of a written algorithm for diagnosis of VTE [n (%)] | 2 (4) | 21 (50) | <0.001 |
D‐Dimer Testing
Among trainees, 25% of residents and students and 20% of IM attendings were unaware of the sensitivity or specificity of D‐dimer assays in use in their institution (P = 0.8), and 70% of ED attendings were unaware. Almost all residents, students, and IM attendings were unable to identify the name of the D‐dimer test used in their institutions (>95% in each category); while 54% of ED attendings were also unable to do so (P < 0.0001).
Imaging Strategies
Table 3 depicts responses regarding knowledge about various VTE imaging strategies. The majority of students responded that they would use VP scanning as the initial modality and a substantial number of attendings and residents would too. All ED attendings reported using CTPA as the initial modality of choice. A substantial number of students, residents, and IM attendings did not know whether LECTV had to be ordered separately or was done by default and a large proportion incorrectly surmised that the sensitivity of LECTV was not equivalent to lower extremity Doppler.
| ED Attending (n = 46) | IM Attending (n = 43) | Residents (n = 139) | Medical Students (n = 134) | P value | |
|---|---|---|---|---|---|
| |||||
| VP scanning test of choice in suspected PE [n (%)] | 0 (0) | 9 (22) | 24 (17) | 78 (58) | <0.001 |
| CTV ordered separate from or with CTPA by default [n (% unaware)] | 12 (27) | 53 (38) | 96 (72) | <0.001 | |
| Sensitivity of CTV = LE US [n (% agree)] | 22 (51) | 69 (50) | 42 (31) | 0.01 | |
Clinical Vignettes
Table 4 depicts responses by non‐ED respondents to various clinical scenarios presented. Faced with a dyspneic patient 2 days after a hip fracture and a negative CTPA alone, almost 25% of all respondents would incorrectly withhold anticoagulation. In outpatients with low probability Wells score for DVT and a negative D‐dimer, substantial proportions of all respondents would incorrectly order further imaging. For treatment of inpatients with DVT and non‐massive PE, 17% of students disagreed that LMWH was appropriate, and similar proportions of residents (12%) and IM attendings (13%) disagreed.
| IM Attending (n = 46) | Residents (n = 139) | Medical Students (n = 134) | P Value | |
|---|---|---|---|---|
| ||||
| Anticoagulate high risk patient with negative CTPA alone [n (% agree)] | 35 (76) | 104 (75) | 99 (74) | 0.9 |
| Order further imaging in outpatient with negative D‐dimer and low probability for DVT [n (% agree)] | 25 (54) | 68 (49) | 59 (44) | 0.6 |
| LMWH appropriate for DVT and non‐massive PE among inpatients [n (% agree)] | 40 (88) | 119 (86) | 88 (66) | 0.02 |
Discussion
Pretest Probability Assessment
Our findings that only a minority of trainees and practicing physicians calculate pretest probability using a prediction score translate into potentially inferior (and more costly) care for patients with suspected VTE. This is especially true for academic institutions, where trainees are ordinarily first responders. Among practitioners in the United States,16 72.5% prefer an unstructured approach to pretest assessment, whereas 22.9% use published prediction rules. In this survey, more residents than students or attendings used the Wells criteria for pretest probability testing. The majority of ED attendings surveyed (61%) used no structured pretest probability assessment, consistent with a retrospective study published recently17; however, this may have been because of the relatively experienced group sampled (median number of years in practice was 12.5 compared to 6 years among IM attendings). Students may not be receiving training to use prediction rules because attendings may feel they are too complex to use and/or may not use these rules themselves. A substantial proportion of residents (40% in our study) do not use them. Awareness of written algorithms was reported by a minority of all respondents, but did not translate into greater use of prediction rules.
D‐Dimer Testing
Only a few highly sensitive quantitative assays (VIDAS, Tinaquant, Liatest, and Simplired)1821 have been validated in large clinical trials incorporating structured pretest probability assessment and CTPA. Guidelines for diagnosis of VTE recommend that physicians be informed about the type of D‐dimer being used in their practice setting given the substantial variation in D‐dimer sensitivity.22 The sensitivities of quantitative enzyme‐linked immunosorbent assays (ELISAs) are clinically and statistically superior to other types of D‐dimer tests among patients with VTE.23 Over 20% of all non‐ED respondents did not know the sensitivity and specificity of the D‐dimer assay in use in their respective institutions and most (>70% in each category) could not name the assay, resulting in potentially inappropriate decision making if nonquantitative ELISA D‐dimers were used alone or gestalt were used, especially by trainees.
Imaging Strategies
Weiss et al.24 surveyed U.S. clinicians and found a clear preference for CTPA as the initial imaging modality in patients with suspected PE but did not include the trainee perspective. As level of training progressed, we found a decrease in the percentage of respondents that preferred VP scanning over CTPA as the first test of choice; however, 25% of residents and 17% of attendings still designated VP scanning as their first choice. The perception of the majority of students in our survey is that VP scanning is the preferred initial test. We conjecture that students do not receive the pertinent training from supervising clinicians in this regard. All ED attendings surveyed used CTPA as their first choice of imaging. Knowledge about whether LECTV was ordered separately from CTPA or done by default was lacking in over 25% of all non‐ED respondents. The lower the level of experience, the more incorrect answers were given. Apropos of the PIOPED II study,12 lack of awareness about lower extremity imaging in association with CTPA may therefore contribute to inappropriate decision making, especially in patients with high pretest probability of PE and a negative CTPA alone.
Clinical Vignettes
Two studies25, 26 analyzed outcomes in patients with low to intermediate pretest probability PE and negative CTPA alone who did not receive anticoagulation. Both suggest that withholding anticoagulation in these patients is safe. The 25% of non‐ED respondents who would consider withholding anticoagulation in high‐risk settings translates into a large number of potentially inappropriate decisions, especially if gestalt is used in pretest probability assessment. This is in line with recommendations from the PIOPED II study that lower extremity imaging and, if necessary, serial lower extremity ultrasonography be performed in high‐risk groups.11, 12 A negative validated D‐dimer study and a low pretest probability exclude the need for further testing in outpatients with suspected DVT27; however, 50% of all respondents would order further testing. Thus, regardless of experience, a disparity exists between practice and published literature among both trainees and attendings, especially since further imaging in this setting is not cost effective.28
Use of LMWH
In a cohort of 946 inpatients in one study,29 only 56.1% of inpatients with DVT or PE were treated with LMWH. In our survey a substantial minority of IM attendings, residents, and students (12%, 13%, and 17%, respectively) would not consider LMWH one of the prefered therapies for VTE in the right clinical setting.
Limitations
The cross‐sectional nature of the survey and localization of non‐ED respondents to the New York, New Jersey, and Connecticut tri‐state region, limits generalizability to other geographic regions of the country. Responses of ED attendings were sampled nationally. The attendings (ED and IM) sampled were a relatively experienced group (6‐12 years of practice) and this may explain the relatively low adoption of prediction rules reflecting the use of gestalt in this group. Additionally, over time, knowledge (and use) of validated D‐dimer assays may have increased in the practices evaluated. Among non‐ED respondents, 30% (116/319) were from a single training program (SUNY Downstate) and the responses of these respondents may reflect practice in the institutions sampled, limiting nationwide generalization with the potential for selection bias. The low rate of response from ED physicians (20%) was presumably a result of being called at work. We believe the responses are still a valuable insight into the real‐time practices of the clinicians surveyed and do not preclude a meaningful comparison to the rest of the respondents especially given the significant differences between ED and IM attending knowledge and awareness (Tables 3 and 4).
Conclusions
Our survey identifies the use of evidence‐based strategies in the management of VTE among trainees, a perspective that has been lacking in other studies of physicians in practice.16, 24, 27 Substantial variability in attending practice identified in this survey may impede the adoption of a structured approach to the diagnosis of VTE among trainees, and this survey raises major concerns about mechanisms of diagnosis of VTE. Caprini et al.29 believe that physician knowledge, attitudes, and beliefs are partially responsible for the gap between actual practice and international guidelines.27 The results of our survey extend this suggestion to trainees and imply that supervisor attitudes may negatively influence trainee practices. Development of written protocols or standardized pathway order sets based on published evidence‐based guidelines13 in the management of VTE could improve the use of structured pretest probability determination and use of evidence‐based strategies among trainees. Finally, comparisons of outcomes using algorithms and usual practice could provide valuable, clinically important data that could inform clinical practice.
Pretest probability assessment is an important first step in the diagnosis of venous thromboembolism (VTE) and models incorporating Wells criteria1 can be used accurately in emergency department (ED) and inpatient settings.2 Gestalt has the disadvantage of poor interobserver reliability,3 and use of clinical prediction rules has been advocated instead.4 In academic institutions, trainees frequently first evaluate patients with suspected VTE, and although gestalt improves with degree of experience, the performance of gestalt in 1 study5 was better for attendings than interns or residents (for whom it was equivalent), suggesting that structured pretest probability assessment may be more important for trainees.
From an imaging perspective, multidetector computed tomography (CT),6 is more accurate than ventilation perfusion (VP) scanning7, 8 in diagnosing VTE in any setting, including the critically ill.9, 10 Lower extremity CT venography (LECTV) has comparable sensitivity to contrast venography and sonography,11 and in combination with computed tomographic pulmonary angiography (CTPA) is important when imaging results are discordant with pretest probability.12 Guidelines for diagnostic pathways in VTE based on published literature incorporating D‐dimer testing have been updated recently,13 the degree of adoption and use of diagnostic algorithms among trainees has been understudied.
Clinical trials14, 15 have confirmed the safety and efficacy of low molecular weight heparin (LMWH) in the treatment of pulmonary embolism (PE) in inpatients, but the degree of adoption of this therapy is unclear. The primary objective of our survey of was to assess the knowledge, attitudes, practices, and preferences of trainees and attendings who order and evaluate the results of diagnostic studies in the management of VTE. A secondary objective was to assess willingness to use LMWH to treat VTE in the inpatient setting among non‐ED respondents.
Methods
Survey Design and Administration
The study was cross‐sectional and was approved by the institutional review board. The survey was paper‐based and anonymous, and the requirement for written informed consent was waived. The survey instrument was reviewed for clarity, lack of bias, and accuracy by a panel of hospitalists at the State University of New York (SUNY) Downstate Medical Center. Closed‐ended questions were used, including a 5‐point Likert scale (1 = strongly agree, 5 = strongly disagree) and multiple‐choice queries. Between October 2006 and March 2008, paper‐based survey questionnaires were distributed to internal medicine (IM) attendings, residents, and students from institutions in the New York, New Jersey, and Connecticut tri‐state area taking medicine review courses in New York City and attending grand rounds at SUNY Downstate. Of 319 non‐ED respondents, 116 (30%) were from the SUNY Downstate system. All third‐year medical students (58/116) were from the SUNY Downstate training program. representing 5 different training institutions for medical students and 4 institutions for residents. ED physicians (n = 46) were selected randomly and telephoned at work and questioned about their practices with an abbreviated version of the survey. Response rates were 80% for the paper‐based surveys and 20% for the ED physicians. Data was recorded into an electronic database (Microsoft Access; Microsoft Corp., Redmond, WA). Simple clinical vignettes were used to assess diagnostic and therapeutic strategies in the setting of VTE for non‐ED respondents only.
Data Analysis
Descriptive statistics were used to report respondents' demographic information and work environments. Data are expressed as proportions, means SD, or medians with interquartile range. Differences in response levels between groups were compared by Fisher's exact test, chi square test, or the Kruskal‐Wallis test, where appropriate. Two‐sided P values of less than 0.05 were considered significant. Since no difference in the ability of residents and interns to predict PE has been noted,5 both groups were analyzed together, as were third‐year and fourth‐year medical students. JMP version 7.0 software (SAS Institute, Cary, NC) was used to perform all analyses.
Results
Table 1 lists the characteristics of respondents. Medical attendings reported practicing in up to 5 different institutions, and residents reported rotating through up to 10 different institutions during their residency. Students reported rotating through up to 11 different institutions.
| n (%) | Institutions Rotated Through [median (IQR)] | |
|---|---|---|
| ||
| Emergency department attendings | 46 | |
| Medicine attendings | 46 | 1 (12) |
| Residents | 139 | 3 (23) |
| PGY1 | 39 (28) | |
| PGY2 | 27 (19) | |
| PGY3 | 34 (24) | |
| PGY4 | 3 (2) | |
| PGY5 | 19 (14) | |
| Year not checked | 17 (13) | |
| Medical students | 134 | 3 (15) |
| Third year | 58 (43) | |
| Fourth year | 76 (57) | |
Pretest Probability Assessment
Table 2 depicts differences between ED and IM attending responses. More than 60% of all attendings used no structured pretest probability assessment; the rest reported using the Wells criteria. An equivalent proportion of ED and IM attendings thought prediction rules were too complex to use (P = 0.2). Years of attending experience did not predict responses regarding perceptions of the complexity of prediction rules (P = 0.5). More IM attendings than residents or students felt that prediction rules were too complex for routine use (P = 0.02). Among trainees, significantly more residents than students reported using the Wells model (P < 0.001); 40% of residents did not use any model. Advanced years in training among residents did not predict an increased likelihood of using prediction rules.
| ED Attendings (n = 46) | IM Attendings (n = 43) | P Value | |
|---|---|---|---|
| |||
| Years of experience [median (interquartile range)] | 12.5 (7.521) | 6 (214) | <0.001 |
| Academic practice [n (%)] | 23 (50) | 27 (63) | 0.7 |
| Do not use prediction rules [n (%)] | 28 (61) | 28 (65) | 0.8 |
| Prediction rules too complex to use [n (% agree)] | 22 (48) | 13 (30) | 0.5 |
| Aware of a written algorithm for diagnosis of VTE [n (%)] | 2 (4) | 21 (50) | <0.001 |
D‐Dimer Testing
Among trainees, 25% of residents and students and 20% of IM attendings were unaware of the sensitivity or specificity of D‐dimer assays in use in their institution (P = 0.8), and 70% of ED attendings were unaware. Almost all residents, students, and IM attendings were unable to identify the name of the D‐dimer test used in their institutions (>95% in each category); while 54% of ED attendings were also unable to do so (P < 0.0001).
Imaging Strategies
Table 3 depicts responses regarding knowledge about various VTE imaging strategies. The majority of students responded that they would use VP scanning as the initial modality and a substantial number of attendings and residents would too. All ED attendings reported using CTPA as the initial modality of choice. A substantial number of students, residents, and IM attendings did not know whether LECTV had to be ordered separately or was done by default and a large proportion incorrectly surmised that the sensitivity of LECTV was not equivalent to lower extremity Doppler.
| ED Attending (n = 46) | IM Attending (n = 43) | Residents (n = 139) | Medical Students (n = 134) | P value | |
|---|---|---|---|---|---|
| |||||
| VP scanning test of choice in suspected PE [n (%)] | 0 (0) | 9 (22) | 24 (17) | 78 (58) | <0.001 |
| CTV ordered separate from or with CTPA by default [n (% unaware)] | 12 (27) | 53 (38) | 96 (72) | <0.001 | |
| Sensitivity of CTV = LE US [n (% agree)] | 22 (51) | 69 (50) | 42 (31) | 0.01 | |
Clinical Vignettes
Table 4 depicts responses by non‐ED respondents to various clinical scenarios presented. Faced with a dyspneic patient 2 days after a hip fracture and a negative CTPA alone, almost 25% of all respondents would incorrectly withhold anticoagulation. In outpatients with low probability Wells score for DVT and a negative D‐dimer, substantial proportions of all respondents would incorrectly order further imaging. For treatment of inpatients with DVT and non‐massive PE, 17% of students disagreed that LMWH was appropriate, and similar proportions of residents (12%) and IM attendings (13%) disagreed.
| IM Attending (n = 46) | Residents (n = 139) | Medical Students (n = 134) | P Value | |
|---|---|---|---|---|
| ||||
| Anticoagulate high risk patient with negative CTPA alone [n (% agree)] | 35 (76) | 104 (75) | 99 (74) | 0.9 |
| Order further imaging in outpatient with negative D‐dimer and low probability for DVT [n (% agree)] | 25 (54) | 68 (49) | 59 (44) | 0.6 |
| LMWH appropriate for DVT and non‐massive PE among inpatients [n (% agree)] | 40 (88) | 119 (86) | 88 (66) | 0.02 |
Discussion
Pretest Probability Assessment
Our findings that only a minority of trainees and practicing physicians calculate pretest probability using a prediction score translate into potentially inferior (and more costly) care for patients with suspected VTE. This is especially true for academic institutions, where trainees are ordinarily first responders. Among practitioners in the United States,16 72.5% prefer an unstructured approach to pretest assessment, whereas 22.9% use published prediction rules. In this survey, more residents than students or attendings used the Wells criteria for pretest probability testing. The majority of ED attendings surveyed (61%) used no structured pretest probability assessment, consistent with a retrospective study published recently17; however, this may have been because of the relatively experienced group sampled (median number of years in practice was 12.5 compared to 6 years among IM attendings). Students may not be receiving training to use prediction rules because attendings may feel they are too complex to use and/or may not use these rules themselves. A substantial proportion of residents (40% in our study) do not use them. Awareness of written algorithms was reported by a minority of all respondents, but did not translate into greater use of prediction rules.
D‐Dimer Testing
Only a few highly sensitive quantitative assays (VIDAS, Tinaquant, Liatest, and Simplired)1821 have been validated in large clinical trials incorporating structured pretest probability assessment and CTPA. Guidelines for diagnosis of VTE recommend that physicians be informed about the type of D‐dimer being used in their practice setting given the substantial variation in D‐dimer sensitivity.22 The sensitivities of quantitative enzyme‐linked immunosorbent assays (ELISAs) are clinically and statistically superior to other types of D‐dimer tests among patients with VTE.23 Over 20% of all non‐ED respondents did not know the sensitivity and specificity of the D‐dimer assay in use in their respective institutions and most (>70% in each category) could not name the assay, resulting in potentially inappropriate decision making if nonquantitative ELISA D‐dimers were used alone or gestalt were used, especially by trainees.
Imaging Strategies
Weiss et al.24 surveyed U.S. clinicians and found a clear preference for CTPA as the initial imaging modality in patients with suspected PE but did not include the trainee perspective. As level of training progressed, we found a decrease in the percentage of respondents that preferred VP scanning over CTPA as the first test of choice; however, 25% of residents and 17% of attendings still designated VP scanning as their first choice. The perception of the majority of students in our survey is that VP scanning is the preferred initial test. We conjecture that students do not receive the pertinent training from supervising clinicians in this regard. All ED attendings surveyed used CTPA as their first choice of imaging. Knowledge about whether LECTV was ordered separately from CTPA or done by default was lacking in over 25% of all non‐ED respondents. The lower the level of experience, the more incorrect answers were given. Apropos of the PIOPED II study,12 lack of awareness about lower extremity imaging in association with CTPA may therefore contribute to inappropriate decision making, especially in patients with high pretest probability of PE and a negative CTPA alone.
Clinical Vignettes
Two studies25, 26 analyzed outcomes in patients with low to intermediate pretest probability PE and negative CTPA alone who did not receive anticoagulation. Both suggest that withholding anticoagulation in these patients is safe. The 25% of non‐ED respondents who would consider withholding anticoagulation in high‐risk settings translates into a large number of potentially inappropriate decisions, especially if gestalt is used in pretest probability assessment. This is in line with recommendations from the PIOPED II study that lower extremity imaging and, if necessary, serial lower extremity ultrasonography be performed in high‐risk groups.11, 12 A negative validated D‐dimer study and a low pretest probability exclude the need for further testing in outpatients with suspected DVT27; however, 50% of all respondents would order further testing. Thus, regardless of experience, a disparity exists between practice and published literature among both trainees and attendings, especially since further imaging in this setting is not cost effective.28
Use of LMWH
In a cohort of 946 inpatients in one study,29 only 56.1% of inpatients with DVT or PE were treated with LMWH. In our survey a substantial minority of IM attendings, residents, and students (12%, 13%, and 17%, respectively) would not consider LMWH one of the prefered therapies for VTE in the right clinical setting.
Limitations
The cross‐sectional nature of the survey and localization of non‐ED respondents to the New York, New Jersey, and Connecticut tri‐state region, limits generalizability to other geographic regions of the country. Responses of ED attendings were sampled nationally. The attendings (ED and IM) sampled were a relatively experienced group (6‐12 years of practice) and this may explain the relatively low adoption of prediction rules reflecting the use of gestalt in this group. Additionally, over time, knowledge (and use) of validated D‐dimer assays may have increased in the practices evaluated. Among non‐ED respondents, 30% (116/319) were from a single training program (SUNY Downstate) and the responses of these respondents may reflect practice in the institutions sampled, limiting nationwide generalization with the potential for selection bias. The low rate of response from ED physicians (20%) was presumably a result of being called at work. We believe the responses are still a valuable insight into the real‐time practices of the clinicians surveyed and do not preclude a meaningful comparison to the rest of the respondents especially given the significant differences between ED and IM attending knowledge and awareness (Tables 3 and 4).
Conclusions
Our survey identifies the use of evidence‐based strategies in the management of VTE among trainees, a perspective that has been lacking in other studies of physicians in practice.16, 24, 27 Substantial variability in attending practice identified in this survey may impede the adoption of a structured approach to the diagnosis of VTE among trainees, and this survey raises major concerns about mechanisms of diagnosis of VTE. Caprini et al.29 believe that physician knowledge, attitudes, and beliefs are partially responsible for the gap between actual practice and international guidelines.27 The results of our survey extend this suggestion to trainees and imply that supervisor attitudes may negatively influence trainee practices. Development of written protocols or standardized pathway order sets based on published evidence‐based guidelines13 in the management of VTE could improve the use of structured pretest probability determination and use of evidence‐based strategies among trainees. Finally, comparisons of outcomes using algorithms and usual practice could provide valuable, clinically important data that could inform clinical practice.
- ,,, et al.Assessment of pretest probability of pulmonary embolism in the emergency department by physicians in training using the Wells model.Thromb Res.2007;120(2):173–179.
- ,,,.A prospective reassessment of the utility of the Wells score in identifying pulmonary embolism.Med J Aust.2007;187(6):333–336.
- ,,,,.The interobserver reliability of pretest probability assessment in patients with suspected pulmonary embolism.Thromb Res.2005;116(2):101–107.
- ,,, et al.Does this patient have pulmonary embolism?JAMA.2003;290(21):2849–2858.
- ,,,,.Does a physician's ability to accurately assess the likelihood of pulmonary embolism increase with training?Acad Med.2000;75(12):1199–1205.
- ,,, et al.Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and D‐dimer.Ann Intern Med.2001;135(2):98–107.
- ,,, et al.Pulmonary embolism revealed on helical CT angiography: comparison with ventilation perfusion radionuclide lung scanning.AJR Am J Roentgenol.2000;174:1041–1047.
- ,,, et al.Clinical validity of a negative computed tomography scan in patients with suspected pulmonary embolism: a systematic review.JAMA.2005;293(16):2012–2017.
- ,,,,.Multidetector CT: a new gold standard in the diagnosis of pulmonary embolism? State of the art and diagnostic algorithms.Radiol Med.2005;109(1–2):49–61.
- ,,,,.Multidetector row CT pulmonary angiography and indirect venography for the diagnosis of venous thromboembolic disease in intensive care unit patients.Acad Radiol.2006;13(4):486–495.
- ,,.The role of multidetector computed tomography angiography for the diagnosis of pulmonary embolism.Semin Nucl Med.2008;38:418–431.
- ,,, et al.Multidetector computed tomography for acute pulmonary embolism.N Engl J Med.2006;354:2317–2327.
- Institute for Clinical Systems Improvement. Venous Thromboembolism Diagnosis and Treatment. Available at: http://www.icsi.org. Accessed October2009.
- The Columbus Investigators.Low‐molecular‐weight heparin in the treatment of patients with venous thromboembolism.N Engl J Med.1997;337:657–662.
- ,,, et al.A comparison of low‐molecular‐weight heparin with unfractionated heparin for acute pulmonary embolism. The THESEE Study Group.N Engl J Med.1997;337(10):663–669.
- ,,,,,.Pretest risk assessment in suspected acute pulmonary embolism.Acad Radiol.2008;15(1):3–14.
- ,,,.Is pretest probability assessment on emergency department patients with suspected venous thromboembolism documented before SimpliRED D‐dimer testing?CJEM.2008;10(6):519–523.
- ,,,,,.Exclusion of DVT with D‐dimer testing: comparison of 13 D‐dimer methods in 99 outpatients suspected of DVT using venography as a standard.Thromb Haemost.2000;83:191–198.
- ,,, et al.Multidetector‐row computed tomography in suspected pulmonary embolism.N Engl J Med.2005;352(17):1760–1768.
- ,,, et al.Simple and safe exclusion of pulmonary embolism in outpatients using quantitative D‐dimer and Wells' simplified decision rule.Thromb Haemost.2007;97(1):146–150.
- ,,,,;ANTELOPE Study Group. The performance of two rapid quantitative D‐dimer assays in 287 patients with clinically suspected pulmonary embolism.Thromb Res.2002;107(6):283–286.
- ,,, et al.Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians.Ann Fam Med.2007;5:57–62.
- ,,, et al.D‐dimer for the exclusion of acute venous thrombosis and pulmonary embolism. A systematic review.Ann Intern Med.2004;140:589–602.
- ,,, et al.CT pulmonary angiography is the first‐line imaging test for acute pulmonary embolism: a survey of US clinicians.Acad Radiol.2006;13:434–446.
- ,,, et al.Outcomes after withholding anticoagulation from patients with suspected pulmonary embolism and negative computed tomographic findings: a cohort study.Mayo Clin Proc.2002;77:130–138.
- ,,,.Meta‐analysis: outcomes in patients with suspected pulmonary embolism managed with computed tomographic pulmonary angiography.Ann Intern Med.2004;141:866–874.
- ,,, et al.Does this patient have deep vein thrombosis?JAMA.2006;295(2):199–207.
- ,,, et al.Measurement of the clinical and cost‐effectiveness of non‐invasive diagnostic testing strategies for deep vein thrombosis.Health Technol Assess.2006;10(15):1–168.
- ,,, et al.NABOR Steering Committee. Treatment of venous thromboembolism: adherence to guidelines and impact of physician knowledge, attitudes, and beliefs.J Vasc Surg.2005;42(4):726–733.
- ,,, et al.Assessment of pretest probability of pulmonary embolism in the emergency department by physicians in training using the Wells model.Thromb Res.2007;120(2):173–179.
- ,,,.A prospective reassessment of the utility of the Wells score in identifying pulmonary embolism.Med J Aust.2007;187(6):333–336.
- ,,,,.The interobserver reliability of pretest probability assessment in patients with suspected pulmonary embolism.Thromb Res.2005;116(2):101–107.
- ,,, et al.Does this patient have pulmonary embolism?JAMA.2003;290(21):2849–2858.
- ,,,,.Does a physician's ability to accurately assess the likelihood of pulmonary embolism increase with training?Acad Med.2000;75(12):1199–1205.
- ,,, et al.Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and D‐dimer.Ann Intern Med.2001;135(2):98–107.
- ,,, et al.Pulmonary embolism revealed on helical CT angiography: comparison with ventilation perfusion radionuclide lung scanning.AJR Am J Roentgenol.2000;174:1041–1047.
- ,,, et al.Clinical validity of a negative computed tomography scan in patients with suspected pulmonary embolism: a systematic review.JAMA.2005;293(16):2012–2017.
- ,,,,.Multidetector CT: a new gold standard in the diagnosis of pulmonary embolism? State of the art and diagnostic algorithms.Radiol Med.2005;109(1–2):49–61.
- ,,,,.Multidetector row CT pulmonary angiography and indirect venography for the diagnosis of venous thromboembolic disease in intensive care unit patients.Acad Radiol.2006;13(4):486–495.
- ,,.The role of multidetector computed tomography angiography for the diagnosis of pulmonary embolism.Semin Nucl Med.2008;38:418–431.
- ,,, et al.Multidetector computed tomography for acute pulmonary embolism.N Engl J Med.2006;354:2317–2327.
- Institute for Clinical Systems Improvement. Venous Thromboembolism Diagnosis and Treatment. Available at: http://www.icsi.org. Accessed October2009.
- The Columbus Investigators.Low‐molecular‐weight heparin in the treatment of patients with venous thromboembolism.N Engl J Med.1997;337:657–662.
- ,,, et al.A comparison of low‐molecular‐weight heparin with unfractionated heparin for acute pulmonary embolism. The THESEE Study Group.N Engl J Med.1997;337(10):663–669.
- ,,,,,.Pretest risk assessment in suspected acute pulmonary embolism.Acad Radiol.2008;15(1):3–14.
- ,,,.Is pretest probability assessment on emergency department patients with suspected venous thromboembolism documented before SimpliRED D‐dimer testing?CJEM.2008;10(6):519–523.
- ,,,,,.Exclusion of DVT with D‐dimer testing: comparison of 13 D‐dimer methods in 99 outpatients suspected of DVT using venography as a standard.Thromb Haemost.2000;83:191–198.
- ,,, et al.Multidetector‐row computed tomography in suspected pulmonary embolism.N Engl J Med.2005;352(17):1760–1768.
- ,,, et al.Simple and safe exclusion of pulmonary embolism in outpatients using quantitative D‐dimer and Wells' simplified decision rule.Thromb Haemost.2007;97(1):146–150.
- ,,,,;ANTELOPE Study Group. The performance of two rapid quantitative D‐dimer assays in 287 patients with clinically suspected pulmonary embolism.Thromb Res.2002;107(6):283–286.
- ,,, et al.Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians.Ann Fam Med.2007;5:57–62.
- ,,, et al.D‐dimer for the exclusion of acute venous thrombosis and pulmonary embolism. A systematic review.Ann Intern Med.2004;140:589–602.
- ,,, et al.CT pulmonary angiography is the first‐line imaging test for acute pulmonary embolism: a survey of US clinicians.Acad Radiol.2006;13:434–446.
- ,,, et al.Outcomes after withholding anticoagulation from patients with suspected pulmonary embolism and negative computed tomographic findings: a cohort study.Mayo Clin Proc.2002;77:130–138.
- ,,,.Meta‐analysis: outcomes in patients with suspected pulmonary embolism managed with computed tomographic pulmonary angiography.Ann Intern Med.2004;141:866–874.
- ,,, et al.Does this patient have deep vein thrombosis?JAMA.2006;295(2):199–207.
- ,,, et al.Measurement of the clinical and cost‐effectiveness of non‐invasive diagnostic testing strategies for deep vein thrombosis.Health Technol Assess.2006;10(15):1–168.
- ,,, et al.NABOR Steering Committee. Treatment of venous thromboembolism: adherence to guidelines and impact of physician knowledge, attitudes, and beliefs.J Vasc Surg.2005;42(4):726–733.
Copyright © 2010 Society of Hospital Medicine
Management of Ischemic Stroke: Part 1
The term stroke is defined by the World Health Organization as rapidly developed clinical signs of focal (or global) disturbance of cerebral function lasting more than 24 hours (unless interrupted by surgery or death), with no apparent cause other than a vascular origin; it includes patients presenting clinical signs and symptoms suggestive of subarachnoid hemorrhage (SAH), intracerebral hemorrhage, or cerebral ischemic necrosis.1 Stroke is 1 of the leading causes of death and the number 1 cause of long‐term disability in the United States, with over 700,000 strokes and over 150,000 stroke deaths each year.2
Given the projections of 30,000 hospitalists nationally by 2010 (
Case Presentation
A 76‐year‐old right‐handed male with a history of hyperlipidemia and myocardial infarction was found at 7 AM with right‐sided paralysis and poor responsiveness on the morning of admission. He seemed to prefer looking to the left and to understand what was being said to him, but had great difficulty speaking. When he went to bed at 9 PM, he was at his neurological baseline. Upon finding him that morning, his wife called 911.
With increased knowledge regarding the pathophysiology of stroke, it has become clear that timeliness is of utmost importance (time is brain) and that acute stroke should be regarded as an acute medical/neurological emergency.
This article reviews the approach in evaluating an acute stroke patient, management strategies, and treatment options. Where not otherwise referenced, data to support our comments come from the recently updated and exhaustive American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for the Early Management of Adults With Ischemic Stroke and will be referred to herein as the Guidelines.4 Harborview Medical Center in Seattle is a Joint Commissioncertified Primary Stroke Center and the home hospital of 2 of the authors (C.L.E., D.L.T.); it is referred to herein as Harborview.
Emergency Room Care (see Acute Stroke Algorithm, Figure 1)
The First 15 Minutes
After assuring stable airway, breathing, and circulation, immediate (STAT) blood draws should be performed, including full complete blood count (CBC) with platelets, international normalized ratio/prothrombin time/partial thromboplastin time (INR/PT/PTT), full electrolytes, and glucose (finger‐stick blood glucose also recommended). Glasgow Coma Scale (GCS) score and NIH Stroke Scale (NIHSS) score should be established via a focused history and physical exam. The GCS is most appropriate for patients with a significantly depressed level of consciousness, while the NIHSS can be scored for any stroke patient (1‐page version of NIHSS used at Harborview is shown in Figure 2). By quantifying stroke severity, the NIHSS score helps both to facilitate communication about neurologic deficit as well as serve as a documented baseline in case of subsequent clinical change. Emergency department (ED) physicians, hospitalists, neurologists, and nursing staff regularly caring for acute stroke patients would be well‐served by obtaining certification in the NIHSS (available free online at
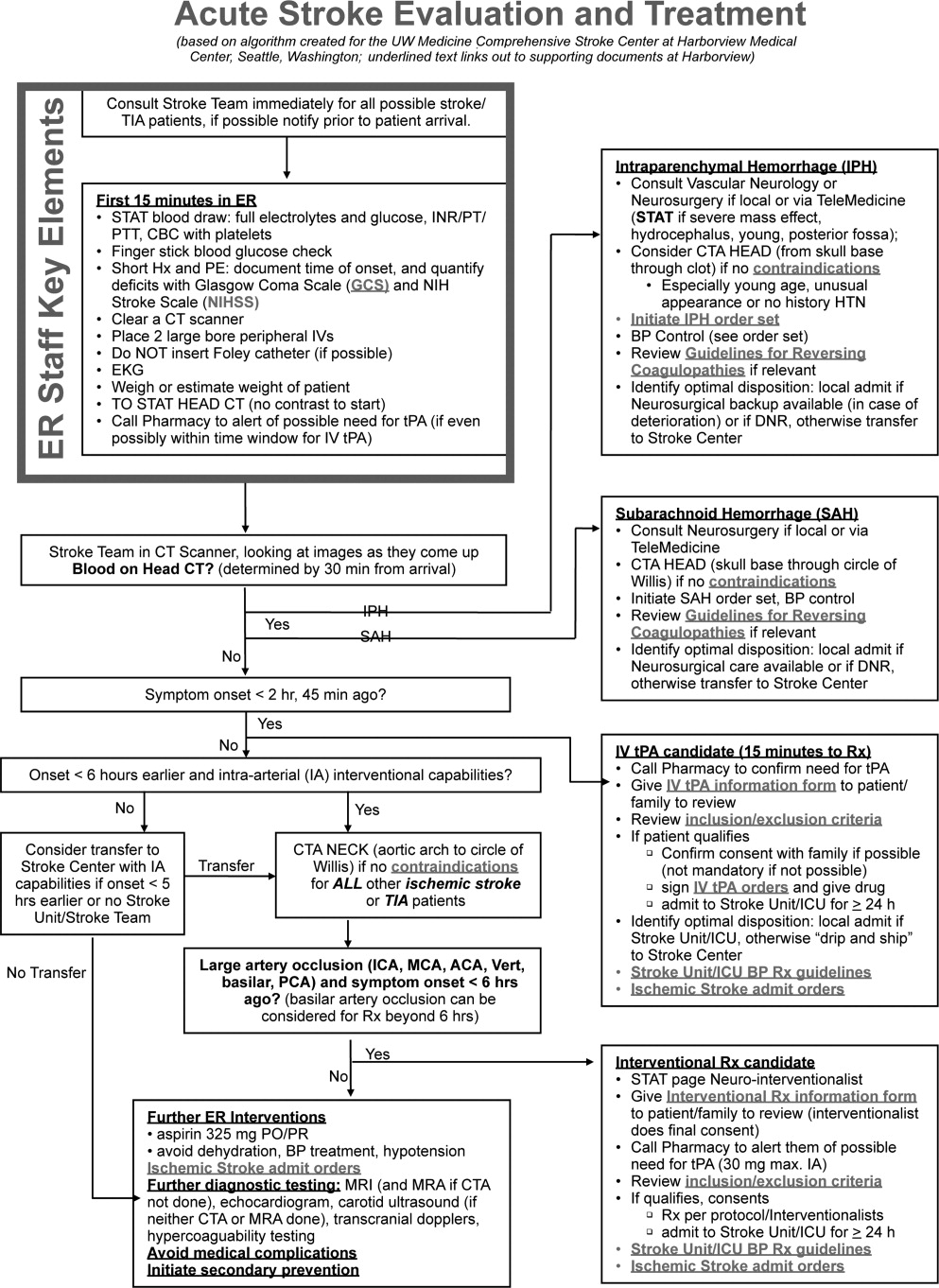
Our case patient's initial NIHSS score was 15, with points given for drowsiness, inability to answer questions, partial facial palsy, no movement in right arm or leg, mild‐moderate aphasia, and mild‐moderate dysarthria (Figure 2).
Differential Diagnosis
Many acute conditions can mimic stroke, and 1 of the goals of the initial emergency room (ER) evaluation is to rule out such stroke mimics. A report of 411 initial ER stroke diagnoses identified 19% as stroke mimics; the most common mimic diagnoses were seizure, systemic infection, brain tumor, and toxic‐metabolic.5 The same study identified decreased level of alertness as associated with a final mimic diagnosis and history of angina as associated with a final diagnosis of stroke. Another study looked at 350 presentations with an initial stroke diagnosis and found 31% stroke mimics; similarly, the main alternative diagnoses were seizure, sepsis, toxic‐metabolic, space‐occupying lesion, and syncope/presyncope.6 Findings associated with a mimic diagnosis included no cognitive impairment and abnormal findings in any other system, while findings associated with a stroke diagnosis were a definite history of focal neurological symptoms, NIHSS score, stroke type classification possible, an exact onset that could be determined, and abnormal vascular findings on imaging.6
Initial Imaging
The patient should receive a STAT noncontrast head CT to evaluate for the presence or absence of blood. At this time, magnetic resonance imaging (MRI) is not essential to confirm the diagnosis of ischemic stroke, as diagnosis is based on clinical suspicion. MRI is more sensitive at imaging acute ischemia (on diffusion‐weighted sequences) and recently has been shown to be equally sensitive in identifying acute blood (previously thought to be a relative advantage of CT).7, 8 Practical and pervasive barriers to emergent MRI include study duration, significant patient cooperation, and that few hospitals are currently set up to perform such rapid MRIS. The Guidelines specifically state that In most instances, CT will provide the information to make decisions about emergency management (p. 1668),4 that vascular imaging should not delay treatment of patients whose symptoms started 3 hours ago and who have acute ischemic stroke, and that emergency treatment of stroke should not be delayed in order to obtain multimodal imaging studies (p. 1669).4
Our case patient's initial imaging, a noncontrast head CT (Supporting Figures 1 and 2), showed subtle clues consistent with the diagnosis of acute ischemic stroke. These include a hyperdense middle cerebral artery (MCA) sign (presumably representing thrombus), possible obscuration of the basal ganglia, and, importantly, no acute intraparenchymal (IPH), SAH, or subdural hemorrhage.
Acute Treatments
After the patient's head CT is completed, the next steps are dependent upon what was seen on the scan and the time from symptom onset.
Blood on the CT Scan
If the initial brain imaging reveals IPH or SAH, further diagnostic testing and early treatments are quite different than for ischemic stroke. New guidelines are available for IPH management,9 and there have been recent review articles of care for SAH.1012 At the authors' institutions, early care of such patients always involves aggressive reversal of any antithrombotic medications the patient was taking prior to presentation. Our approach to warfarin reversal includes vitamin K and fresh frozen plasma (FFP) to achieve an INR 1.4; others have used prothrombin complex concentrate (PCC).13 Blood pressure (BP) treatment goals are generally more aggressive than for ischemic stroke, while supportive care to avoid aspiration, hyperglycemia, fever, and venous thrombosis (here initially with sequential compression devices alone) are similar. Early estimation of prognosis for these patients with IPH and SAH and discussions with families about continued aggressive care are of utmost importance, and should involve providers with sufficient expertise. Care should be taken to avoid overly pessimistic early prognostication, as early do not resuscitate (DNR) decisions in intercranial hemorrhage (ICH) can become a self‐fulfilling prophecy.1416 If the decision is to continue aggressive and supportive care, or if an appropriately expert consultation is not available at the presentation hospital, IPH and SAH patients should be considered for transfer to a hospital with the appropriate resources (including emergency access to neurosurgeons) or be evaluated by such an expert by telemedicine if available.
No Blood on the CT Scan, Results Back in 3 Hours From Symptom Onset
If such a patient is not rapidly resolving their symptoms, and the diagnosis continues to remain clear, inclusion/exclusion criteria for IV tPA should be reviewed (Table 1). Consent should be obtained much like any other procedure with significant risk. As many consider tPA to be standard of care, it is reasonable to proceed in cases of unobtainable consent as one would with any other emergent therapy. This situation is a topic of ongoing debate.17, 18 The Guidelines state that although written consent is not necessary before administration of recombinant tPA (rtPA) for treatment of stroke, a full discussion of the potential risks and benefits of treatment with rtPA with the family and the patient if possible is recommended (p. 1676).4 After tPA is given in the ER, the patient should be admitted to an intensive care unit (ICU) setting for 24 hours for careful monitoring of BP, avoidance of invasive procedures, and no use of antithrombotic medications during that period of time.
| Comments (from the authors) | |
|---|---|
| |
| Inclusion criteria | |
| Diagnosis of ischemic stroke causing measurable neurological deficit | Usually NIHSS > 4 |
| Neurological signs should not be clearing spontaneously | Such a patient may do well without tPA, but there is debate82 |
| Neurological signs should not be minor and isolated. | |
| Onset of symptoms >3 hours before beginning treatment | |
| Patient or family members understand the potential risks and benefits from treatment | Debated, as tPA considered standard of care by many |
| Cautionary criteria | |
| Caution should be exercised in treating a patient with major deficits | Higher risk of hemorrhage, but still may benefit from treatment |
| Exclusion criteria | |
| Symptoms of stroke should not be suggestive of subarachnoid hemorrhage | |
| No head trauma or prior stroke in previous 3 months | |
| No myocardial infarction in the previous 3 months | |
| No gastrointestinal or urinary tract hemorrhage in previous 21 days | |
| No major surgery in the previous 14 days | |
| No arterial puncture at a noncompressible site in the previous 7 days | |
| No history of previous intracranial hemorrhage | |
| Blood pressure not elevated (systolic >185 mm Hg or diastolic 110 mm Hg) | Okay to bring down with labetolol, nitropaste, or nicardipine* |
| No evidence of active bleeding or acute trauma (fracture) on examination | |
| Not taking an oral anticoagulant or, if anticoagulant being taken, INR 1.7 | |
| If receiving heparin in previous 48 hours, aPTT must be in normal range | |
| Platelet count 100,000 mm3 | |
| Blood glucose concentration 50 mg/dL (2.7 mmol/L) | |
| Seizure with postictal residual neurological impairments | Not absolute if treating physician feels stroke also present, or if confirmed by imaging |
| CT does not show a multilobar infarction (hypodensity >1/3 cerebral hemisphere) | Not strictly evidence based, in NINDS trial this finding did not preclude benefit of tPA |
Based mainly on the results of the National Institute of Neurological Disorders and Stroke (NINDS) tPA trial,19 and recently supported by a large Phase IV observational study from the European Union,20 IV tPA for acute ischemic stroke is approved for use in many countries and is endorsed for the treatment of carefully selected ischemic stroke patients in a number of practice guidelines.4 Despite this, the emergency medicine community has been less enthusiastic about the use of IV tPA.21, 22 Although the risk of hemorrhagic complications is greater in certain subgroups of patients (ie, the most severe strokes, significant early CT changes, older age), there is no definitive evidence to suggest that these groups do not still benefit from the treatment.23 It is also clear that if patients are not carefully selected, meeting strict inclusion and exclusion criteria, the rate of complications is increased.24 Thus, as summarized in a practice statement of the American College of Emergency Physicians, There is insufficient evidence at this time to endorse the use of intravenous tPA in clinical practice when systems are not in place to ensure that the inclusion/exclusion criteria established by the NINDS guidelines for tPA use in acute stroke are followed.21 When counseling patients and their families about the benefits and risks of IV tPA, one should keep in mind that the NINDS trial demonstrated increased odds of excellent outcomes despite a significant 10‐fold increase in the risk of symptomatic intracranial hemorrhage (6.4% vs. 0.6%), and did not alter 30‐day mortality. The largest Phase IV cohort study of IV tPA treatment, Safe Implementation of Thrombolysis in Stroke Monitoring Study (SITS‐MOST) was mandated by the European Union upon approval of the medication for use in acute ischemic stroke.20 The results in 6483 patients showed that tPA, when used in strict accordance with published inclusion and exclusion criteria, could perform as well as it did in randomized trials.
The recently published European Cooperative Acute Stroke Study3 (ECASS‐3) trial demonstrated that IV tPA has efficacy with adequate safety up to 4.5 hours after the onset of symptoms. A total of 821 patients were enrolled and 375 received tPA. Exclusion criteria included diabetes being treated with medication with a history of prior stroke, an NIHSS score >25, or treatment with warfarin. The rates of hemorrhage (27.0% vs. 17.6%, P = 0.001) were in line with those of the SITS‐MOST study patients who were treated within the 3‐hour time window. There was no significant difference in mortality (7.7% tPA vs. 8.4% placebo). This study is relatively new; therefore, the data have not been reviewed by guideline committees.25
No Blood on the CT Scan, Results Back in >3 Hours, but 8 Hours, From Symptom Onset
Unfortunately as with our patient, most people do not present to an ER in a timely fashion. Nonetheless, there may be other treatments and interventions possible. If the patient arrives 8 hours from onset of symptoms, intraarterial (IA) interventions are a possibility. In such a case, a CT angiogram (CTA) of the neck from the arch of the aorta to the circle of Willis is recommended (barring any contraindications such as renal failure or iodine allergy). The rationale behind this study is that other treatment options, such as IA tPA or mechanical thrombectomy may be considered if a large arterial occlusion is identified. CTA is preferred over magnetic resonance angiography (MRA) due to the same time and patient cooperation issues mentioned above, though some expert centers may be set up to perform MRI and MRA rapidly in the acute setting. CTA or MRA is of great value early on in the emergent assessment of ischemic stroke patients, as it allows detailed evaluation of the cerebral vasculature; this knowledge helps define the pathophysiology of the ongoing stroke (eg, is there a larger artery occlusion?) and can help inform the approach to subsequent therapies.
The Guidelines (p. 1678)4 recommend IA thrombolysis as a treatment option if it can be started within 6 hours, based on results from the Prolyse in Acute Cerebral Thromboembolism (PROACT) II trial. This study involved angiography with identification of the occluded vessel (the proximal MCA‐M1 in this study) and administration of recombinant pro‐urokinase to the clot with functional outcome as the primary endpoint.26 At 3 months, patients who received the IA thrombolytic had a 40% chance of slight disability; unable to carry out all previous activities, but able to look after own affairs without assistance or better (ie, a modified Rankin Scale score of 2) vs. 25% of those not receiving the IA thrombolytic. Pro‐urokinase is not available in the United States; therefore, many institutions substitute IA tPA. The Guidelines further state that IA thrombolysis can be considered for use in some patients with contraindications to IV tPA (eg, recent surgery), but should not be used instead of IV tPA in patients otherwise eligible (p. 1678).4
There are now two U.S. Food and Drug Administration (FDA)‐approved devices for mechanical cerebral vasculature thrombectomy for use up to 8 hours from symptom onset. The mechanical embolus removal in cerebral ischemia (MERCI) clot retrieval device was originally approved by the FDA in August 2004 for restoring blood flow in the neurovasculature by removing thrombus in patients experiencing ischemic stroke. Modified devices have been approved as recently as January 2007.27 The Penumbra System was FDA‐approved in December 2007 for revascularization of patients with acute ischemic stroke secondary to intracranial large vessel occlusive disease.28 In both cases, the FDA approval was based on demonstration of safety in case series of patients treated with the devices.2931 No randomized trials have shown the use of these devices improves outcomes for stroke patients. The Guidelines state that Although the MERCI device is a reasonable intervention for extraction of IA thrombi in carefully selected patients, the panel also recognizes that the utility of the device in improving outcomes after stroke is unclear (p. 1684);4 this statement applies similarly to the Penumbra device.
More complex imaging techniques, including multimodal CT (CT, CTA, and CT perfusion) and MR (MRI with diffusion, MRA, and MR perfusion) are being used in some stroke centers to make decisions about acute ischemic stroke treatments.32, 33 The theory is that by using these techniques, one can determine the presence or absence of a mismatch, whereby the perfusion imaging suggests more tissue at risk of infarction than is seen as already abnormal on MR diffusion‐weighted images or compared to a clinical assessment. These mismatch patients are then seen as appropriate candidates for the more aggressive interventions (ie, late IV tPA or IA interventions).34 Unfortunately, the 2 largest randomized trials to look at this issue with respect to >3‐hour IV tPA both failed to show a benefit for patients selected in this manner.35, 36 Standardized definitions of mismatch are still needed, and larger randomized trials are needed before this approach can be suggested for routine care.3739
More complex interventions, available only at tertiary or comprehensive stroke centers, include a bridging approach in which IV tPA (at 2/3 standard dose) is followed by IA tPA, IV tPA with transcranial Doppler (TCD)‐enhanced thrombolysis or IA rescue thrombectomy when vascular imaging after IV tPA shows a persistent large artery occlusion. The Guidelines suggests that these more complex combinations of interventions to restore perfusion cannot be recommended outside the setting of clinical trials (p. 1685).4
No Blood on the CT Scan, Results Back in >8 Hours From Symptom Onset (or if Contraindications to Above Interventions)
This time frame takes the more aggressive interventions off the table. Per the Guidelines, 325 mg of aspirin is the default antiplatelet agent for use, and has been shown in 2 very large randomized trials to reduce early death and longer‐term disability vs. placebo after acute ischemic stroke.40, 41 Importantly, all patients who do not qualify for thrombolysis in the 0‐hour to 8‐hour time window should receive aspirin.
Although a number of small or pilot studies suggest a benefit of the addition of clopidogrel to aspirin for a period (13 months) immediately after ischemic stroke,4244 this more aggressive antiplatelet intervention is not an endorsed standard of care. As described below, the long‐term use of this antiplatelet combination has been consistently associated with a higher risk of hemorrhagic complications. There are no published data regarding the use of aspirin plus dipyridamole in the acute stroke setting. A number of randomized trials have now been performed that have consistently failed to show a benefit of heparin, or heparin‐like medications, for the routine treatment of acute ischemic stroke. Despite this, a number of exceptions exist, based more on tradition and theory than on evidence. These exceptions, for which an IV heparin drip will at times still be considered, include acute ischemic stroke due to dissection of the carotid or vertebral arteries, cardioembolic stroke with fresh clot seen on echocardiogram (ECHO), and a clinically progressive syndrome suggestive of basilar artery occlusion (see below).45, 46 Good evidence exists to specifically recommend the use of full‐dose heparin in the setting of cerebral venous sinus thrombosis.47
Basilar Artery Occlusion Syndromes
Basilar artery occlusion syndromes warrant special mention. These may involve patients who present with quadriparesis, altered mental status, vertigo, diplopia, and other brainstem signs. Conventional treatment of basilar artery occlusion has been associated with 40% mortality with 65% of survivors having severe disability.48 If suspected, an urgent CTA can usually confirm the diagnosis, and urge the clinician to expeditiously consider aggressive intervention. Only case series have been reported regarding basilar artery thrombosis and acute treatments. Based on these studies, it is generally agreed upon that patients who appear comatose or quadriplegic for more than 3 hours will likely have a very poor functional outcome regardless of treatment, and interventional treatment is withheld. If a basilar occlusion patient presents within the 3‐hour time window for IV tPA, they are thus treated, with follow‐up vascular imaging, and possible rescue IA mechanical thrombectomy if recanalization from the IV tPA does not occur. However, if the patient still has preserved neurologic function, or is waxing and waning, there is no clear time limit for IA interventions and they may be useful a day or more after presentation. For basilar occlusion patients with severe stenoses not responsive to lysis, or continuing to be symptomatic, angioplasty and stenting has also been used.46 Despite a lack of evidence, many stroke clinicians will use an IV heparin drip for treatment of acute basilar occlusive disease.
Malignant Middle Cerebral Artery (MCA) Infarction
Malignant MCA infarction is another specific clinical syndrome worthy of special consideration. It is most generally defined as a large infarction (1/2 or 2/3) of the MCA territory, somewhat depressed level of consciousness, and high stroke scale scores (ie, severe deficits) that goes on to severe cerebral edema, mass effect, and often herniation with death.49, 50 Associated patient characteristics include younger age, abnormal (incomplete) ipsilateral collateral circulation, and internal carotid artery occlusion.51 Maximal edema occurs 2 to 5 days from stroke onset and, despite best intensive therapy, has been associated with mortality rates of 70% to 80%.49, 50 A recent pooling of 3 small randomized trials of early decompressive hemicraniectomy and durotomy showed a 50% absolute risk reduction for mortality and a 23% absolute benefit in long‐term independence (modified Rankin scale 3).49 This treatment option should be strongly considered in carefully selected patients., Transfer to an appropriately equipped facility should be offered if not available at your hospital.
Returning to our case patient, upon arrival to the ED with symptoms of partial aphasia, right hemiplegia, and left gaze preference, there was a high suspicion for a left MCA stroke. Unfortunately, he was excluded from receiving IV tPA or any other interventions, as the last time he was known to be neurologically intact was the prior evening, which is taken to be the time of onset. Antiplatelet therapy was continued, and the patient was admitted for further workup.
The initial care of the patient with a cerebrovascular event is often quite complicated. Assimilation of a great deal of data must occur and decisions around therapy must be made in a timely fashion. In prior years there was little to offer in the way of therapy, which also meant there was little initial potential for iatrogenic complication. Both diagnostic and therapeutic options are evolving rapidly. We now have much to offer these patients both emergently and in areas of secondary prevention. In part 2 of this article, the patient's inpatient course and therapy will be reviewed.
- Organization WH. MONICA Manual, Part IV: Event Registration. Available at: http://www.ktl.fi/publications/monica/manual/part4/iv‐2.htm#s2. Accessed May2009.
- ,,, et al.Heart disease and stroke statistics 2008 update. A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee.Circulation.200829;117(4):e25–e146.
- .Neurology in the next two decades: report of the Workforce Task Force of the American Academy of Neurology.Neurology.2000;54(4):787–789.
- ,,, et al.Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists.Stroke.2007;38(5):1655–1711.
- ,,,.Conditions that mimic stroke in the emergency department. Implications for acute stroke trials.Arch Neurol.1995;52(11):1119–1122.
- ,,,,.Distinguishing between stroke and mimic at the bedside: the brain attack study.Stroke.2006;37(3):769–775.
- ,,, et al.Stroke magnetic resonance imaging is accurate in hyperacute intracerebral hemorrhage: a multicenter study on the validity of stroke imaging.Stroke.2004;35(2):502–506.
- ,,, et al.Comparison of MRI and CT for detection of acute intracerebral hemorrhage.JAMA.2004;292(15):1823–1830.
- ,,, et al.Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group.Stroke.2007;38(6):2001–2023.
- ,,.Aneurysmal subarachnoid hemorrhage.N Engl J Med.2006;354(4):387–396.
- ,,,.Subarachnoid haemorrhage.BMJ.2006;333(7561):235–240.
- ,,.Subarachnoid haemorrhage.Lancet.2007;369(9558):306–318.
- ,,.Intracerebral hemorrhage associated with oral anticoagulant therapy: current practices and unresolved questions.Stroke.2006;37(1):256–262.
- ,,, et al.Withdrawal of support in intracerebral hemorrhage may lead to self‐fulfilling prophecies.Neurology.2001;56(6):766–772.
- ,,,.Hospital usage of early do‐not‐resuscitate orders and outcome after intracerebral hemorrhage.Stroke.2004;35(5):1130–1134.
- ,,, et al.Early care limitations independently predict mortality after intracerebral hemorrhage.Neurology.2007;68(20):1651–1657.
- ,,,.Consent for intravenous thrombolysis in acute stroke: review and future directions.Arch Neurol.2007;64(6):785–792.
- .Thrombolysis (tissue plasminogen activator) in stroke: a medicolegal quagmire.Stroke.2006;37(7):1917–1922.
- Tissue plasminogen activator for acute ischemic stroke.The National Institute of Neurological Disorders and Stroke rt‐PA Stroke Study Group.N Engl J Med.1995;333(24):1581–1587.
- ,,, et al.Thrombolysis with alteplase for acute ischaemic stroke in the Safe Implementation of Thrombolysis in Stroke‐Monitoring Study (SITS‐MOST): an observational study.Lancet.2007;369(9558):275–282.
- American College of Emergency Physicians (ACEP). Use of Intravenous tPA for the Management of Acute Stroke in the Emergency Department. ACEP Policy Statement. February 2002. Available at: http://www.acep.org/practres.aspx?id=29834. Accessed May2009.
- American Academy of Emergency Medicine (AAEM). Position statement on the use of intravenous thrombolytic therapy in the treatment of stroke. January 2002. Available at: http://aaem.org/positionstatements/thrombolytictherapy.php. Accessed May2009.
- ,,, et al.Lack of clinical significance of early ischemic changes on computed tomography in acute stroke.JAMA.2001;286(22):2830–2838.
- ,,,,.Thrombolysis for acute stroke in routine clinical practice.Arch Intern Med.2002;162(17):1994–2001.
- ,,, et al.Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke.N Engl J Med.2008;359:1317–1329,1393–1395.
- ,,, et al.Intra‐arterial prourokinase for acute ischemic stroke. The PROACT II study: a randomized controlled trial. Prolyse in Acute Cerebral Thromboembolism.JAMA.1999;282(21):2003–2011.
- Modified MERCI Retriever FDA marketing approval letter. Available at: www.fda.gov/cdrh/pdf6/K062046.pdf. Accessed May2009.
- Penumbra System FDA marketing approval letter. Available at: www.fda.gov/cdrh/pdf7/K072718.pdf. Accessed May2009.
- .Safety of mechanical thrombectomy and intravenous tissue plasminogen activator in acute ischemic stroke. Results of the multi mechanical embolus removal in cerebral ischemia (MERCI) trial, part I.AJNR Am J Neuroradiol.2006;27(6):1177–1182.
- ,,, et al.Safety and efficacy of mechanical embolectomy in acute ischemic stroke: results of the MERCI trial.Stroke.2005;36(7):1432–1438.
- ,,, et al.The Penumbra System: a mechanical device for the treatment of acute stroke due to thromboembolism.AJNR Am J Neuroradiol.2008;29(7):1409–1413.
- ,,, et al.Comparison of CT perfusion and angiography and MRI in selecting stroke patients for acute treatment.Neurology.2007;68(9):694–697.
- ,,, et al.Combined intravenous and intraarterial revascularization therapy using MRI perfusion/diffusion mismatch selection for acute ischemic stroke at 3–6 h after symptom onset.Neurocrit Care.2008;8(3):353–359.
- ,,, et al.Refining the perfusion‐diffusion mismatch hypothesis.Stroke.2005;36(6):1153–1159.
- . DIAS‐2: no benefit of desmoteplase in acute ischemic stroke. Available at: www.medscape.com/viewarticle/557663. Accessed May2009.
- ,,, et al.Effects of alteplase beyond 3 h after stroke in the Echoplanar Imaging Thrombolytic Evaluation Trial (EPITHET): a placebo‐controlled randomised trial.Lancet Neurol.2008;7(4):299–309.
- ,,.Magnetic resonance perfusion diffusion mismatch and thrombolysis in acute ischaemic stroke: a systematic review of the evidence to date.J Neurol Neurosurg Psychiatry.2007;78(5):485–491.
- ,,, et al.Optimal definition for PWI/DWI mismatch in acute ischemic stroke patients.J Cereb Blood Flow Metab.2008;28(5):887–891.
- ,,, et al.Rapid assessment of perfusion‐diffusion mismatch.Stroke.2008;39(1):75–81.
- CAST: randomised placebo‐controlled trial of early aspirin use in 20,000 patients with acute ischaemic stroke.CAST (Chinese Acute Stroke Trial) Collaborative Group.Lancet.1997;349(9066):1641–1649.
- The International Stroke Trial (IST): a randomised trial of aspirin, subcutaneous heparin, both, or neither among 19435 patients with acute ischaemic stroke.International Stroke Trial Collaborative Group.Lancet.1997;349(9065):1569–1581.
- ,,, et al.Dual antiplatelet therapy with clopidogrel and aspirin in symptomatic carotid stenosis evaluated using doppler embolic signal detection: the clopidogrel and aspirin for reduction of emboli in symptomatic carotid stenosis (CARESS) trial.Circulation.2005;111(17):2233–2240.
- ,,, et al.Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population‐based sequential comparison.Lancet.2007;370(9596):1432–1442.
- ,,,,,.Fast assessment of stroke and transient ischaemic attack to prevent early recurrence (FASTER): a randomised controlled pilot trial.Lancet Neurol.2007;6(11):961–969.
- ,,, et al.Antiplatelets versus anticoagulation in cervical artery dissection.Stroke.2007;38(9):2605–2611.
- ,,.Basilar artery occlusion.Neurocrit Care.2004;1(3):319–329.
- ,.Cerebral venous thrombosis: an update.Lancet Neurol.2007;6(2):162–170.
- ,,,,.Outcome in patients with basilar artery occlusion treated conventionally.J Neurol Neurosurg Psychiatry.2005;76(9):1238–1241.
- ,,, et al.Early decompressive surgery in malignant infarction of the middle cerebral artery: a pooled analysis of three randomised controlled trials.Lancet Neurol.2007;6(3):215–222.
- ,,,,,.‘Malignant’ middle cerebral artery territory infarction: clinical course and prognostic signs.Arch Neurol.1996;53(4):309–315.
- ,,,,.Predictors for malignant middle cerebral artery infarctions: a postmortem analysis.Neurology. 282006;66(6):815–820.
- ,,,,,.Poor outcomes in patients who do not receive intravenous tissue plasminogen activator because of mild or improving ischemic stroke.Stroke.2005;36(11):2497–2499.
The term stroke is defined by the World Health Organization as rapidly developed clinical signs of focal (or global) disturbance of cerebral function lasting more than 24 hours (unless interrupted by surgery or death), with no apparent cause other than a vascular origin; it includes patients presenting clinical signs and symptoms suggestive of subarachnoid hemorrhage (SAH), intracerebral hemorrhage, or cerebral ischemic necrosis.1 Stroke is 1 of the leading causes of death and the number 1 cause of long‐term disability in the United States, with over 700,000 strokes and over 150,000 stroke deaths each year.2
Given the projections of 30,000 hospitalists nationally by 2010 (
Case Presentation
A 76‐year‐old right‐handed male with a history of hyperlipidemia and myocardial infarction was found at 7 AM with right‐sided paralysis and poor responsiveness on the morning of admission. He seemed to prefer looking to the left and to understand what was being said to him, but had great difficulty speaking. When he went to bed at 9 PM, he was at his neurological baseline. Upon finding him that morning, his wife called 911.
With increased knowledge regarding the pathophysiology of stroke, it has become clear that timeliness is of utmost importance (time is brain) and that acute stroke should be regarded as an acute medical/neurological emergency.
This article reviews the approach in evaluating an acute stroke patient, management strategies, and treatment options. Where not otherwise referenced, data to support our comments come from the recently updated and exhaustive American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for the Early Management of Adults With Ischemic Stroke and will be referred to herein as the Guidelines.4 Harborview Medical Center in Seattle is a Joint Commissioncertified Primary Stroke Center and the home hospital of 2 of the authors (C.L.E., D.L.T.); it is referred to herein as Harborview.
Emergency Room Care (see Acute Stroke Algorithm, Figure 1)
The First 15 Minutes
After assuring stable airway, breathing, and circulation, immediate (STAT) blood draws should be performed, including full complete blood count (CBC) with platelets, international normalized ratio/prothrombin time/partial thromboplastin time (INR/PT/PTT), full electrolytes, and glucose (finger‐stick blood glucose also recommended). Glasgow Coma Scale (GCS) score and NIH Stroke Scale (NIHSS) score should be established via a focused history and physical exam. The GCS is most appropriate for patients with a significantly depressed level of consciousness, while the NIHSS can be scored for any stroke patient (1‐page version of NIHSS used at Harborview is shown in Figure 2). By quantifying stroke severity, the NIHSS score helps both to facilitate communication about neurologic deficit as well as serve as a documented baseline in case of subsequent clinical change. Emergency department (ED) physicians, hospitalists, neurologists, and nursing staff regularly caring for acute stroke patients would be well‐served by obtaining certification in the NIHSS (available free online at


Our case patient's initial NIHSS score was 15, with points given for drowsiness, inability to answer questions, partial facial palsy, no movement in right arm or leg, mild‐moderate aphasia, and mild‐moderate dysarthria (Figure 2).
Differential Diagnosis
Many acute conditions can mimic stroke, and 1 of the goals of the initial emergency room (ER) evaluation is to rule out such stroke mimics. A report of 411 initial ER stroke diagnoses identified 19% as stroke mimics; the most common mimic diagnoses were seizure, systemic infection, brain tumor, and toxic‐metabolic.5 The same study identified decreased level of alertness as associated with a final mimic diagnosis and history of angina as associated with a final diagnosis of stroke. Another study looked at 350 presentations with an initial stroke diagnosis and found 31% stroke mimics; similarly, the main alternative diagnoses were seizure, sepsis, toxic‐metabolic, space‐occupying lesion, and syncope/presyncope.6 Findings associated with a mimic diagnosis included no cognitive impairment and abnormal findings in any other system, while findings associated with a stroke diagnosis were a definite history of focal neurological symptoms, NIHSS score, stroke type classification possible, an exact onset that could be determined, and abnormal vascular findings on imaging.6
Initial Imaging
The patient should receive a STAT noncontrast head CT to evaluate for the presence or absence of blood. At this time, magnetic resonance imaging (MRI) is not essential to confirm the diagnosis of ischemic stroke, as diagnosis is based on clinical suspicion. MRI is more sensitive at imaging acute ischemia (on diffusion‐weighted sequences) and recently has been shown to be equally sensitive in identifying acute blood (previously thought to be a relative advantage of CT).7, 8 Practical and pervasive barriers to emergent MRI include study duration, significant patient cooperation, and that few hospitals are currently set up to perform such rapid MRIS. The Guidelines specifically state that In most instances, CT will provide the information to make decisions about emergency management (p. 1668),4 that vascular imaging should not delay treatment of patients whose symptoms started 3 hours ago and who have acute ischemic stroke, and that emergency treatment of stroke should not be delayed in order to obtain multimodal imaging studies (p. 1669).4
Our case patient's initial imaging, a noncontrast head CT (Supporting Figures 1 and 2), showed subtle clues consistent with the diagnosis of acute ischemic stroke. These include a hyperdense middle cerebral artery (MCA) sign (presumably representing thrombus), possible obscuration of the basal ganglia, and, importantly, no acute intraparenchymal (IPH), SAH, or subdural hemorrhage.
Acute Treatments
After the patient's head CT is completed, the next steps are dependent upon what was seen on the scan and the time from symptom onset.
Blood on the CT Scan
If the initial brain imaging reveals IPH or SAH, further diagnostic testing and early treatments are quite different than for ischemic stroke. New guidelines are available for IPH management,9 and there have been recent review articles of care for SAH.1012 At the authors' institutions, early care of such patients always involves aggressive reversal of any antithrombotic medications the patient was taking prior to presentation. Our approach to warfarin reversal includes vitamin K and fresh frozen plasma (FFP) to achieve an INR 1.4; others have used prothrombin complex concentrate (PCC).13 Blood pressure (BP) treatment goals are generally more aggressive than for ischemic stroke, while supportive care to avoid aspiration, hyperglycemia, fever, and venous thrombosis (here initially with sequential compression devices alone) are similar. Early estimation of prognosis for these patients with IPH and SAH and discussions with families about continued aggressive care are of utmost importance, and should involve providers with sufficient expertise. Care should be taken to avoid overly pessimistic early prognostication, as early do not resuscitate (DNR) decisions in intercranial hemorrhage (ICH) can become a self‐fulfilling prophecy.1416 If the decision is to continue aggressive and supportive care, or if an appropriately expert consultation is not available at the presentation hospital, IPH and SAH patients should be considered for transfer to a hospital with the appropriate resources (including emergency access to neurosurgeons) or be evaluated by such an expert by telemedicine if available.
No Blood on the CT Scan, Results Back in 3 Hours From Symptom Onset
If such a patient is not rapidly resolving their symptoms, and the diagnosis continues to remain clear, inclusion/exclusion criteria for IV tPA should be reviewed (Table 1). Consent should be obtained much like any other procedure with significant risk. As many consider tPA to be standard of care, it is reasonable to proceed in cases of unobtainable consent as one would with any other emergent therapy. This situation is a topic of ongoing debate.17, 18 The Guidelines state that although written consent is not necessary before administration of recombinant tPA (rtPA) for treatment of stroke, a full discussion of the potential risks and benefits of treatment with rtPA with the family and the patient if possible is recommended (p. 1676).4 After tPA is given in the ER, the patient should be admitted to an intensive care unit (ICU) setting for 24 hours for careful monitoring of BP, avoidance of invasive procedures, and no use of antithrombotic medications during that period of time.
| Comments (from the authors) | |
|---|---|
| |
| Inclusion criteria | |
| Diagnosis of ischemic stroke causing measurable neurological deficit | Usually NIHSS > 4 |
| Neurological signs should not be clearing spontaneously | Such a patient may do well without tPA, but there is debate82 |
| Neurological signs should not be minor and isolated. | |
| Onset of symptoms >3 hours before beginning treatment | |
| Patient or family members understand the potential risks and benefits from treatment | Debated, as tPA considered standard of care by many |
| Cautionary criteria | |
| Caution should be exercised in treating a patient with major deficits | Higher risk of hemorrhage, but still may benefit from treatment |
| Exclusion criteria | |
| Symptoms of stroke should not be suggestive of subarachnoid hemorrhage | |
| No head trauma or prior stroke in previous 3 months | |
| No myocardial infarction in the previous 3 months | |
| No gastrointestinal or urinary tract hemorrhage in previous 21 days | |
| No major surgery in the previous 14 days | |
| No arterial puncture at a noncompressible site in the previous 7 days | |
| No history of previous intracranial hemorrhage | |
| Blood pressure not elevated (systolic >185 mm Hg or diastolic 110 mm Hg) | Okay to bring down with labetolol, nitropaste, or nicardipine* |
| No evidence of active bleeding or acute trauma (fracture) on examination | |
| Not taking an oral anticoagulant or, if anticoagulant being taken, INR 1.7 | |
| If receiving heparin in previous 48 hours, aPTT must be in normal range | |
| Platelet count 100,000 mm3 | |
| Blood glucose concentration 50 mg/dL (2.7 mmol/L) | |
| Seizure with postictal residual neurological impairments | Not absolute if treating physician feels stroke also present, or if confirmed by imaging |
| CT does not show a multilobar infarction (hypodensity >1/3 cerebral hemisphere) | Not strictly evidence based, in NINDS trial this finding did not preclude benefit of tPA |
Based mainly on the results of the National Institute of Neurological Disorders and Stroke (NINDS) tPA trial,19 and recently supported by a large Phase IV observational study from the European Union,20 IV tPA for acute ischemic stroke is approved for use in many countries and is endorsed for the treatment of carefully selected ischemic stroke patients in a number of practice guidelines.4 Despite this, the emergency medicine community has been less enthusiastic about the use of IV tPA.21, 22 Although the risk of hemorrhagic complications is greater in certain subgroups of patients (ie, the most severe strokes, significant early CT changes, older age), there is no definitive evidence to suggest that these groups do not still benefit from the treatment.23 It is also clear that if patients are not carefully selected, meeting strict inclusion and exclusion criteria, the rate of complications is increased.24 Thus, as summarized in a practice statement of the American College of Emergency Physicians, There is insufficient evidence at this time to endorse the use of intravenous tPA in clinical practice when systems are not in place to ensure that the inclusion/exclusion criteria established by the NINDS guidelines for tPA use in acute stroke are followed.21 When counseling patients and their families about the benefits and risks of IV tPA, one should keep in mind that the NINDS trial demonstrated increased odds of excellent outcomes despite a significant 10‐fold increase in the risk of symptomatic intracranial hemorrhage (6.4% vs. 0.6%), and did not alter 30‐day mortality. The largest Phase IV cohort study of IV tPA treatment, Safe Implementation of Thrombolysis in Stroke Monitoring Study (SITS‐MOST) was mandated by the European Union upon approval of the medication for use in acute ischemic stroke.20 The results in 6483 patients showed that tPA, when used in strict accordance with published inclusion and exclusion criteria, could perform as well as it did in randomized trials.
The recently published European Cooperative Acute Stroke Study3 (ECASS‐3) trial demonstrated that IV tPA has efficacy with adequate safety up to 4.5 hours after the onset of symptoms. A total of 821 patients were enrolled and 375 received tPA. Exclusion criteria included diabetes being treated with medication with a history of prior stroke, an NIHSS score >25, or treatment with warfarin. The rates of hemorrhage (27.0% vs. 17.6%, P = 0.001) were in line with those of the SITS‐MOST study patients who were treated within the 3‐hour time window. There was no significant difference in mortality (7.7% tPA vs. 8.4% placebo). This study is relatively new; therefore, the data have not been reviewed by guideline committees.25
No Blood on the CT Scan, Results Back in >3 Hours, but 8 Hours, From Symptom Onset
Unfortunately as with our patient, most people do not present to an ER in a timely fashion. Nonetheless, there may be other treatments and interventions possible. If the patient arrives 8 hours from onset of symptoms, intraarterial (IA) interventions are a possibility. In such a case, a CT angiogram (CTA) of the neck from the arch of the aorta to the circle of Willis is recommended (barring any contraindications such as renal failure or iodine allergy). The rationale behind this study is that other treatment options, such as IA tPA or mechanical thrombectomy may be considered if a large arterial occlusion is identified. CTA is preferred over magnetic resonance angiography (MRA) due to the same time and patient cooperation issues mentioned above, though some expert centers may be set up to perform MRI and MRA rapidly in the acute setting. CTA or MRA is of great value early on in the emergent assessment of ischemic stroke patients, as it allows detailed evaluation of the cerebral vasculature; this knowledge helps define the pathophysiology of the ongoing stroke (eg, is there a larger artery occlusion?) and can help inform the approach to subsequent therapies.
The Guidelines (p. 1678)4 recommend IA thrombolysis as a treatment option if it can be started within 6 hours, based on results from the Prolyse in Acute Cerebral Thromboembolism (PROACT) II trial. This study involved angiography with identification of the occluded vessel (the proximal MCA‐M1 in this study) and administration of recombinant pro‐urokinase to the clot with functional outcome as the primary endpoint.26 At 3 months, patients who received the IA thrombolytic had a 40% chance of slight disability; unable to carry out all previous activities, but able to look after own affairs without assistance or better (ie, a modified Rankin Scale score of 2) vs. 25% of those not receiving the IA thrombolytic. Pro‐urokinase is not available in the United States; therefore, many institutions substitute IA tPA. The Guidelines further state that IA thrombolysis can be considered for use in some patients with contraindications to IV tPA (eg, recent surgery), but should not be used instead of IV tPA in patients otherwise eligible (p. 1678).4
There are now two U.S. Food and Drug Administration (FDA)‐approved devices for mechanical cerebral vasculature thrombectomy for use up to 8 hours from symptom onset. The mechanical embolus removal in cerebral ischemia (MERCI) clot retrieval device was originally approved by the FDA in August 2004 for restoring blood flow in the neurovasculature by removing thrombus in patients experiencing ischemic stroke. Modified devices have been approved as recently as January 2007.27 The Penumbra System was FDA‐approved in December 2007 for revascularization of patients with acute ischemic stroke secondary to intracranial large vessel occlusive disease.28 In both cases, the FDA approval was based on demonstration of safety in case series of patients treated with the devices.2931 No randomized trials have shown the use of these devices improves outcomes for stroke patients. The Guidelines state that Although the MERCI device is a reasonable intervention for extraction of IA thrombi in carefully selected patients, the panel also recognizes that the utility of the device in improving outcomes after stroke is unclear (p. 1684);4 this statement applies similarly to the Penumbra device.
More complex imaging techniques, including multimodal CT (CT, CTA, and CT perfusion) and MR (MRI with diffusion, MRA, and MR perfusion) are being used in some stroke centers to make decisions about acute ischemic stroke treatments.32, 33 The theory is that by using these techniques, one can determine the presence or absence of a mismatch, whereby the perfusion imaging suggests more tissue at risk of infarction than is seen as already abnormal on MR diffusion‐weighted images or compared to a clinical assessment. These mismatch patients are then seen as appropriate candidates for the more aggressive interventions (ie, late IV tPA or IA interventions).34 Unfortunately, the 2 largest randomized trials to look at this issue with respect to >3‐hour IV tPA both failed to show a benefit for patients selected in this manner.35, 36 Standardized definitions of mismatch are still needed, and larger randomized trials are needed before this approach can be suggested for routine care.3739
More complex interventions, available only at tertiary or comprehensive stroke centers, include a bridging approach in which IV tPA (at 2/3 standard dose) is followed by IA tPA, IV tPA with transcranial Doppler (TCD)‐enhanced thrombolysis or IA rescue thrombectomy when vascular imaging after IV tPA shows a persistent large artery occlusion. The Guidelines suggests that these more complex combinations of interventions to restore perfusion cannot be recommended outside the setting of clinical trials (p. 1685).4
No Blood on the CT Scan, Results Back in >8 Hours From Symptom Onset (or if Contraindications to Above Interventions)
This time frame takes the more aggressive interventions off the table. Per the Guidelines, 325 mg of aspirin is the default antiplatelet agent for use, and has been shown in 2 very large randomized trials to reduce early death and longer‐term disability vs. placebo after acute ischemic stroke.40, 41 Importantly, all patients who do not qualify for thrombolysis in the 0‐hour to 8‐hour time window should receive aspirin.
Although a number of small or pilot studies suggest a benefit of the addition of clopidogrel to aspirin for a period (13 months) immediately after ischemic stroke,4244 this more aggressive antiplatelet intervention is not an endorsed standard of care. As described below, the long‐term use of this antiplatelet combination has been consistently associated with a higher risk of hemorrhagic complications. There are no published data regarding the use of aspirin plus dipyridamole in the acute stroke setting. A number of randomized trials have now been performed that have consistently failed to show a benefit of heparin, or heparin‐like medications, for the routine treatment of acute ischemic stroke. Despite this, a number of exceptions exist, based more on tradition and theory than on evidence. These exceptions, for which an IV heparin drip will at times still be considered, include acute ischemic stroke due to dissection of the carotid or vertebral arteries, cardioembolic stroke with fresh clot seen on echocardiogram (ECHO), and a clinically progressive syndrome suggestive of basilar artery occlusion (see below).45, 46 Good evidence exists to specifically recommend the use of full‐dose heparin in the setting of cerebral venous sinus thrombosis.47
Basilar Artery Occlusion Syndromes
Basilar artery occlusion syndromes warrant special mention. These may involve patients who present with quadriparesis, altered mental status, vertigo, diplopia, and other brainstem signs. Conventional treatment of basilar artery occlusion has been associated with 40% mortality with 65% of survivors having severe disability.48 If suspected, an urgent CTA can usually confirm the diagnosis, and urge the clinician to expeditiously consider aggressive intervention. Only case series have been reported regarding basilar artery thrombosis and acute treatments. Based on these studies, it is generally agreed upon that patients who appear comatose or quadriplegic for more than 3 hours will likely have a very poor functional outcome regardless of treatment, and interventional treatment is withheld. If a basilar occlusion patient presents within the 3‐hour time window for IV tPA, they are thus treated, with follow‐up vascular imaging, and possible rescue IA mechanical thrombectomy if recanalization from the IV tPA does not occur. However, if the patient still has preserved neurologic function, or is waxing and waning, there is no clear time limit for IA interventions and they may be useful a day or more after presentation. For basilar occlusion patients with severe stenoses not responsive to lysis, or continuing to be symptomatic, angioplasty and stenting has also been used.46 Despite a lack of evidence, many stroke clinicians will use an IV heparin drip for treatment of acute basilar occlusive disease.
Malignant Middle Cerebral Artery (MCA) Infarction
Malignant MCA infarction is another specific clinical syndrome worthy of special consideration. It is most generally defined as a large infarction (1/2 or 2/3) of the MCA territory, somewhat depressed level of consciousness, and high stroke scale scores (ie, severe deficits) that goes on to severe cerebral edema, mass effect, and often herniation with death.49, 50 Associated patient characteristics include younger age, abnormal (incomplete) ipsilateral collateral circulation, and internal carotid artery occlusion.51 Maximal edema occurs 2 to 5 days from stroke onset and, despite best intensive therapy, has been associated with mortality rates of 70% to 80%.49, 50 A recent pooling of 3 small randomized trials of early decompressive hemicraniectomy and durotomy showed a 50% absolute risk reduction for mortality and a 23% absolute benefit in long‐term independence (modified Rankin scale 3).49 This treatment option should be strongly considered in carefully selected patients., Transfer to an appropriately equipped facility should be offered if not available at your hospital.
Returning to our case patient, upon arrival to the ED with symptoms of partial aphasia, right hemiplegia, and left gaze preference, there was a high suspicion for a left MCA stroke. Unfortunately, he was excluded from receiving IV tPA or any other interventions, as the last time he was known to be neurologically intact was the prior evening, which is taken to be the time of onset. Antiplatelet therapy was continued, and the patient was admitted for further workup.
The initial care of the patient with a cerebrovascular event is often quite complicated. Assimilation of a great deal of data must occur and decisions around therapy must be made in a timely fashion. In prior years there was little to offer in the way of therapy, which also meant there was little initial potential for iatrogenic complication. Both diagnostic and therapeutic options are evolving rapidly. We now have much to offer these patients both emergently and in areas of secondary prevention. In part 2 of this article, the patient's inpatient course and therapy will be reviewed.
The term stroke is defined by the World Health Organization as rapidly developed clinical signs of focal (or global) disturbance of cerebral function lasting more than 24 hours (unless interrupted by surgery or death), with no apparent cause other than a vascular origin; it includes patients presenting clinical signs and symptoms suggestive of subarachnoid hemorrhage (SAH), intracerebral hemorrhage, or cerebral ischemic necrosis.1 Stroke is 1 of the leading causes of death and the number 1 cause of long‐term disability in the United States, with over 700,000 strokes and over 150,000 stroke deaths each year.2
Given the projections of 30,000 hospitalists nationally by 2010 (
Case Presentation
A 76‐year‐old right‐handed male with a history of hyperlipidemia and myocardial infarction was found at 7 AM with right‐sided paralysis and poor responsiveness on the morning of admission. He seemed to prefer looking to the left and to understand what was being said to him, but had great difficulty speaking. When he went to bed at 9 PM, he was at his neurological baseline. Upon finding him that morning, his wife called 911.
With increased knowledge regarding the pathophysiology of stroke, it has become clear that timeliness is of utmost importance (time is brain) and that acute stroke should be regarded as an acute medical/neurological emergency.
This article reviews the approach in evaluating an acute stroke patient, management strategies, and treatment options. Where not otherwise referenced, data to support our comments come from the recently updated and exhaustive American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for the Early Management of Adults With Ischemic Stroke and will be referred to herein as the Guidelines.4 Harborview Medical Center in Seattle is a Joint Commissioncertified Primary Stroke Center and the home hospital of 2 of the authors (C.L.E., D.L.T.); it is referred to herein as Harborview.
Emergency Room Care (see Acute Stroke Algorithm, Figure 1)
The First 15 Minutes
After assuring stable airway, breathing, and circulation, immediate (STAT) blood draws should be performed, including full complete blood count (CBC) with platelets, international normalized ratio/prothrombin time/partial thromboplastin time (INR/PT/PTT), full electrolytes, and glucose (finger‐stick blood glucose also recommended). Glasgow Coma Scale (GCS) score and NIH Stroke Scale (NIHSS) score should be established via a focused history and physical exam. The GCS is most appropriate for patients with a significantly depressed level of consciousness, while the NIHSS can be scored for any stroke patient (1‐page version of NIHSS used at Harborview is shown in Figure 2). By quantifying stroke severity, the NIHSS score helps both to facilitate communication about neurologic deficit as well as serve as a documented baseline in case of subsequent clinical change. Emergency department (ED) physicians, hospitalists, neurologists, and nursing staff regularly caring for acute stroke patients would be well‐served by obtaining certification in the NIHSS (available free online at


Our case patient's initial NIHSS score was 15, with points given for drowsiness, inability to answer questions, partial facial palsy, no movement in right arm or leg, mild‐moderate aphasia, and mild‐moderate dysarthria (Figure 2).
Differential Diagnosis
Many acute conditions can mimic stroke, and 1 of the goals of the initial emergency room (ER) evaluation is to rule out such stroke mimics. A report of 411 initial ER stroke diagnoses identified 19% as stroke mimics; the most common mimic diagnoses were seizure, systemic infection, brain tumor, and toxic‐metabolic.5 The same study identified decreased level of alertness as associated with a final mimic diagnosis and history of angina as associated with a final diagnosis of stroke. Another study looked at 350 presentations with an initial stroke diagnosis and found 31% stroke mimics; similarly, the main alternative diagnoses were seizure, sepsis, toxic‐metabolic, space‐occupying lesion, and syncope/presyncope.6 Findings associated with a mimic diagnosis included no cognitive impairment and abnormal findings in any other system, while findings associated with a stroke diagnosis were a definite history of focal neurological symptoms, NIHSS score, stroke type classification possible, an exact onset that could be determined, and abnormal vascular findings on imaging.6
Initial Imaging
The patient should receive a STAT noncontrast head CT to evaluate for the presence or absence of blood. At this time, magnetic resonance imaging (MRI) is not essential to confirm the diagnosis of ischemic stroke, as diagnosis is based on clinical suspicion. MRI is more sensitive at imaging acute ischemia (on diffusion‐weighted sequences) and recently has been shown to be equally sensitive in identifying acute blood (previously thought to be a relative advantage of CT).7, 8 Practical and pervasive barriers to emergent MRI include study duration, significant patient cooperation, and that few hospitals are currently set up to perform such rapid MRIS. The Guidelines specifically state that In most instances, CT will provide the information to make decisions about emergency management (p. 1668),4 that vascular imaging should not delay treatment of patients whose symptoms started 3 hours ago and who have acute ischemic stroke, and that emergency treatment of stroke should not be delayed in order to obtain multimodal imaging studies (p. 1669).4
Our case patient's initial imaging, a noncontrast head CT (Supporting Figures 1 and 2), showed subtle clues consistent with the diagnosis of acute ischemic stroke. These include a hyperdense middle cerebral artery (MCA) sign (presumably representing thrombus), possible obscuration of the basal ganglia, and, importantly, no acute intraparenchymal (IPH), SAH, or subdural hemorrhage.
Acute Treatments
After the patient's head CT is completed, the next steps are dependent upon what was seen on the scan and the time from symptom onset.
Blood on the CT Scan
If the initial brain imaging reveals IPH or SAH, further diagnostic testing and early treatments are quite different than for ischemic stroke. New guidelines are available for IPH management,9 and there have been recent review articles of care for SAH.1012 At the authors' institutions, early care of such patients always involves aggressive reversal of any antithrombotic medications the patient was taking prior to presentation. Our approach to warfarin reversal includes vitamin K and fresh frozen plasma (FFP) to achieve an INR 1.4; others have used prothrombin complex concentrate (PCC).13 Blood pressure (BP) treatment goals are generally more aggressive than for ischemic stroke, while supportive care to avoid aspiration, hyperglycemia, fever, and venous thrombosis (here initially with sequential compression devices alone) are similar. Early estimation of prognosis for these patients with IPH and SAH and discussions with families about continued aggressive care are of utmost importance, and should involve providers with sufficient expertise. Care should be taken to avoid overly pessimistic early prognostication, as early do not resuscitate (DNR) decisions in intercranial hemorrhage (ICH) can become a self‐fulfilling prophecy.1416 If the decision is to continue aggressive and supportive care, or if an appropriately expert consultation is not available at the presentation hospital, IPH and SAH patients should be considered for transfer to a hospital with the appropriate resources (including emergency access to neurosurgeons) or be evaluated by such an expert by telemedicine if available.
No Blood on the CT Scan, Results Back in 3 Hours From Symptom Onset
If such a patient is not rapidly resolving their symptoms, and the diagnosis continues to remain clear, inclusion/exclusion criteria for IV tPA should be reviewed (Table 1). Consent should be obtained much like any other procedure with significant risk. As many consider tPA to be standard of care, it is reasonable to proceed in cases of unobtainable consent as one would with any other emergent therapy. This situation is a topic of ongoing debate.17, 18 The Guidelines state that although written consent is not necessary before administration of recombinant tPA (rtPA) for treatment of stroke, a full discussion of the potential risks and benefits of treatment with rtPA with the family and the patient if possible is recommended (p. 1676).4 After tPA is given in the ER, the patient should be admitted to an intensive care unit (ICU) setting for 24 hours for careful monitoring of BP, avoidance of invasive procedures, and no use of antithrombotic medications during that period of time.
| Comments (from the authors) | |
|---|---|
| |
| Inclusion criteria | |
| Diagnosis of ischemic stroke causing measurable neurological deficit | Usually NIHSS > 4 |
| Neurological signs should not be clearing spontaneously | Such a patient may do well without tPA, but there is debate82 |
| Neurological signs should not be minor and isolated. | |
| Onset of symptoms >3 hours before beginning treatment | |
| Patient or family members understand the potential risks and benefits from treatment | Debated, as tPA considered standard of care by many |
| Cautionary criteria | |
| Caution should be exercised in treating a patient with major deficits | Higher risk of hemorrhage, but still may benefit from treatment |
| Exclusion criteria | |
| Symptoms of stroke should not be suggestive of subarachnoid hemorrhage | |
| No head trauma or prior stroke in previous 3 months | |
| No myocardial infarction in the previous 3 months | |
| No gastrointestinal or urinary tract hemorrhage in previous 21 days | |
| No major surgery in the previous 14 days | |
| No arterial puncture at a noncompressible site in the previous 7 days | |
| No history of previous intracranial hemorrhage | |
| Blood pressure not elevated (systolic >185 mm Hg or diastolic 110 mm Hg) | Okay to bring down with labetolol, nitropaste, or nicardipine* |
| No evidence of active bleeding or acute trauma (fracture) on examination | |
| Not taking an oral anticoagulant or, if anticoagulant being taken, INR 1.7 | |
| If receiving heparin in previous 48 hours, aPTT must be in normal range | |
| Platelet count 100,000 mm3 | |
| Blood glucose concentration 50 mg/dL (2.7 mmol/L) | |
| Seizure with postictal residual neurological impairments | Not absolute if treating physician feels stroke also present, or if confirmed by imaging |
| CT does not show a multilobar infarction (hypodensity >1/3 cerebral hemisphere) | Not strictly evidence based, in NINDS trial this finding did not preclude benefit of tPA |
Based mainly on the results of the National Institute of Neurological Disorders and Stroke (NINDS) tPA trial,19 and recently supported by a large Phase IV observational study from the European Union,20 IV tPA for acute ischemic stroke is approved for use in many countries and is endorsed for the treatment of carefully selected ischemic stroke patients in a number of practice guidelines.4 Despite this, the emergency medicine community has been less enthusiastic about the use of IV tPA.21, 22 Although the risk of hemorrhagic complications is greater in certain subgroups of patients (ie, the most severe strokes, significant early CT changes, older age), there is no definitive evidence to suggest that these groups do not still benefit from the treatment.23 It is also clear that if patients are not carefully selected, meeting strict inclusion and exclusion criteria, the rate of complications is increased.24 Thus, as summarized in a practice statement of the American College of Emergency Physicians, There is insufficient evidence at this time to endorse the use of intravenous tPA in clinical practice when systems are not in place to ensure that the inclusion/exclusion criteria established by the NINDS guidelines for tPA use in acute stroke are followed.21 When counseling patients and their families about the benefits and risks of IV tPA, one should keep in mind that the NINDS trial demonstrated increased odds of excellent outcomes despite a significant 10‐fold increase in the risk of symptomatic intracranial hemorrhage (6.4% vs. 0.6%), and did not alter 30‐day mortality. The largest Phase IV cohort study of IV tPA treatment, Safe Implementation of Thrombolysis in Stroke Monitoring Study (SITS‐MOST) was mandated by the European Union upon approval of the medication for use in acute ischemic stroke.20 The results in 6483 patients showed that tPA, when used in strict accordance with published inclusion and exclusion criteria, could perform as well as it did in randomized trials.
The recently published European Cooperative Acute Stroke Study3 (ECASS‐3) trial demonstrated that IV tPA has efficacy with adequate safety up to 4.5 hours after the onset of symptoms. A total of 821 patients were enrolled and 375 received tPA. Exclusion criteria included diabetes being treated with medication with a history of prior stroke, an NIHSS score >25, or treatment with warfarin. The rates of hemorrhage (27.0% vs. 17.6%, P = 0.001) were in line with those of the SITS‐MOST study patients who were treated within the 3‐hour time window. There was no significant difference in mortality (7.7% tPA vs. 8.4% placebo). This study is relatively new; therefore, the data have not been reviewed by guideline committees.25
No Blood on the CT Scan, Results Back in >3 Hours, but 8 Hours, From Symptom Onset
Unfortunately as with our patient, most people do not present to an ER in a timely fashion. Nonetheless, there may be other treatments and interventions possible. If the patient arrives 8 hours from onset of symptoms, intraarterial (IA) interventions are a possibility. In such a case, a CT angiogram (CTA) of the neck from the arch of the aorta to the circle of Willis is recommended (barring any contraindications such as renal failure or iodine allergy). The rationale behind this study is that other treatment options, such as IA tPA or mechanical thrombectomy may be considered if a large arterial occlusion is identified. CTA is preferred over magnetic resonance angiography (MRA) due to the same time and patient cooperation issues mentioned above, though some expert centers may be set up to perform MRI and MRA rapidly in the acute setting. CTA or MRA is of great value early on in the emergent assessment of ischemic stroke patients, as it allows detailed evaluation of the cerebral vasculature; this knowledge helps define the pathophysiology of the ongoing stroke (eg, is there a larger artery occlusion?) and can help inform the approach to subsequent therapies.
The Guidelines (p. 1678)4 recommend IA thrombolysis as a treatment option if it can be started within 6 hours, based on results from the Prolyse in Acute Cerebral Thromboembolism (PROACT) II trial. This study involved angiography with identification of the occluded vessel (the proximal MCA‐M1 in this study) and administration of recombinant pro‐urokinase to the clot with functional outcome as the primary endpoint.26 At 3 months, patients who received the IA thrombolytic had a 40% chance of slight disability; unable to carry out all previous activities, but able to look after own affairs without assistance or better (ie, a modified Rankin Scale score of 2) vs. 25% of those not receiving the IA thrombolytic. Pro‐urokinase is not available in the United States; therefore, many institutions substitute IA tPA. The Guidelines further state that IA thrombolysis can be considered for use in some patients with contraindications to IV tPA (eg, recent surgery), but should not be used instead of IV tPA in patients otherwise eligible (p. 1678).4
There are now two U.S. Food and Drug Administration (FDA)‐approved devices for mechanical cerebral vasculature thrombectomy for use up to 8 hours from symptom onset. The mechanical embolus removal in cerebral ischemia (MERCI) clot retrieval device was originally approved by the FDA in August 2004 for restoring blood flow in the neurovasculature by removing thrombus in patients experiencing ischemic stroke. Modified devices have been approved as recently as January 2007.27 The Penumbra System was FDA‐approved in December 2007 for revascularization of patients with acute ischemic stroke secondary to intracranial large vessel occlusive disease.28 In both cases, the FDA approval was based on demonstration of safety in case series of patients treated with the devices.2931 No randomized trials have shown the use of these devices improves outcomes for stroke patients. The Guidelines state that Although the MERCI device is a reasonable intervention for extraction of IA thrombi in carefully selected patients, the panel also recognizes that the utility of the device in improving outcomes after stroke is unclear (p. 1684);4 this statement applies similarly to the Penumbra device.
More complex imaging techniques, including multimodal CT (CT, CTA, and CT perfusion) and MR (MRI with diffusion, MRA, and MR perfusion) are being used in some stroke centers to make decisions about acute ischemic stroke treatments.32, 33 The theory is that by using these techniques, one can determine the presence or absence of a mismatch, whereby the perfusion imaging suggests more tissue at risk of infarction than is seen as already abnormal on MR diffusion‐weighted images or compared to a clinical assessment. These mismatch patients are then seen as appropriate candidates for the more aggressive interventions (ie, late IV tPA or IA interventions).34 Unfortunately, the 2 largest randomized trials to look at this issue with respect to >3‐hour IV tPA both failed to show a benefit for patients selected in this manner.35, 36 Standardized definitions of mismatch are still needed, and larger randomized trials are needed before this approach can be suggested for routine care.3739
More complex interventions, available only at tertiary or comprehensive stroke centers, include a bridging approach in which IV tPA (at 2/3 standard dose) is followed by IA tPA, IV tPA with transcranial Doppler (TCD)‐enhanced thrombolysis or IA rescue thrombectomy when vascular imaging after IV tPA shows a persistent large artery occlusion. The Guidelines suggests that these more complex combinations of interventions to restore perfusion cannot be recommended outside the setting of clinical trials (p. 1685).4
No Blood on the CT Scan, Results Back in >8 Hours From Symptom Onset (or if Contraindications to Above Interventions)
This time frame takes the more aggressive interventions off the table. Per the Guidelines, 325 mg of aspirin is the default antiplatelet agent for use, and has been shown in 2 very large randomized trials to reduce early death and longer‐term disability vs. placebo after acute ischemic stroke.40, 41 Importantly, all patients who do not qualify for thrombolysis in the 0‐hour to 8‐hour time window should receive aspirin.
Although a number of small or pilot studies suggest a benefit of the addition of clopidogrel to aspirin for a period (13 months) immediately after ischemic stroke,4244 this more aggressive antiplatelet intervention is not an endorsed standard of care. As described below, the long‐term use of this antiplatelet combination has been consistently associated with a higher risk of hemorrhagic complications. There are no published data regarding the use of aspirin plus dipyridamole in the acute stroke setting. A number of randomized trials have now been performed that have consistently failed to show a benefit of heparin, or heparin‐like medications, for the routine treatment of acute ischemic stroke. Despite this, a number of exceptions exist, based more on tradition and theory than on evidence. These exceptions, for which an IV heparin drip will at times still be considered, include acute ischemic stroke due to dissection of the carotid or vertebral arteries, cardioembolic stroke with fresh clot seen on echocardiogram (ECHO), and a clinically progressive syndrome suggestive of basilar artery occlusion (see below).45, 46 Good evidence exists to specifically recommend the use of full‐dose heparin in the setting of cerebral venous sinus thrombosis.47
Basilar Artery Occlusion Syndromes
Basilar artery occlusion syndromes warrant special mention. These may involve patients who present with quadriparesis, altered mental status, vertigo, diplopia, and other brainstem signs. Conventional treatment of basilar artery occlusion has been associated with 40% mortality with 65% of survivors having severe disability.48 If suspected, an urgent CTA can usually confirm the diagnosis, and urge the clinician to expeditiously consider aggressive intervention. Only case series have been reported regarding basilar artery thrombosis and acute treatments. Based on these studies, it is generally agreed upon that patients who appear comatose or quadriplegic for more than 3 hours will likely have a very poor functional outcome regardless of treatment, and interventional treatment is withheld. If a basilar occlusion patient presents within the 3‐hour time window for IV tPA, they are thus treated, with follow‐up vascular imaging, and possible rescue IA mechanical thrombectomy if recanalization from the IV tPA does not occur. However, if the patient still has preserved neurologic function, or is waxing and waning, there is no clear time limit for IA interventions and they may be useful a day or more after presentation. For basilar occlusion patients with severe stenoses not responsive to lysis, or continuing to be symptomatic, angioplasty and stenting has also been used.46 Despite a lack of evidence, many stroke clinicians will use an IV heparin drip for treatment of acute basilar occlusive disease.
Malignant Middle Cerebral Artery (MCA) Infarction
Malignant MCA infarction is another specific clinical syndrome worthy of special consideration. It is most generally defined as a large infarction (1/2 or 2/3) of the MCA territory, somewhat depressed level of consciousness, and high stroke scale scores (ie, severe deficits) that goes on to severe cerebral edema, mass effect, and often herniation with death.49, 50 Associated patient characteristics include younger age, abnormal (incomplete) ipsilateral collateral circulation, and internal carotid artery occlusion.51 Maximal edema occurs 2 to 5 days from stroke onset and, despite best intensive therapy, has been associated with mortality rates of 70% to 80%.49, 50 A recent pooling of 3 small randomized trials of early decompressive hemicraniectomy and durotomy showed a 50% absolute risk reduction for mortality and a 23% absolute benefit in long‐term independence (modified Rankin scale 3).49 This treatment option should be strongly considered in carefully selected patients., Transfer to an appropriately equipped facility should be offered if not available at your hospital.
Returning to our case patient, upon arrival to the ED with symptoms of partial aphasia, right hemiplegia, and left gaze preference, there was a high suspicion for a left MCA stroke. Unfortunately, he was excluded from receiving IV tPA or any other interventions, as the last time he was known to be neurologically intact was the prior evening, which is taken to be the time of onset. Antiplatelet therapy was continued, and the patient was admitted for further workup.
The initial care of the patient with a cerebrovascular event is often quite complicated. Assimilation of a great deal of data must occur and decisions around therapy must be made in a timely fashion. In prior years there was little to offer in the way of therapy, which also meant there was little initial potential for iatrogenic complication. Both diagnostic and therapeutic options are evolving rapidly. We now have much to offer these patients both emergently and in areas of secondary prevention. In part 2 of this article, the patient's inpatient course and therapy will be reviewed.
- Organization WH. MONICA Manual, Part IV: Event Registration. Available at: http://www.ktl.fi/publications/monica/manual/part4/iv‐2.htm#s2. Accessed May2009.
- ,,, et al.Heart disease and stroke statistics 2008 update. A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee.Circulation.200829;117(4):e25–e146.
- .Neurology in the next two decades: report of the Workforce Task Force of the American Academy of Neurology.Neurology.2000;54(4):787–789.
- ,,, et al.Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists.Stroke.2007;38(5):1655–1711.
- ,,,.Conditions that mimic stroke in the emergency department. Implications for acute stroke trials.Arch Neurol.1995;52(11):1119–1122.
- ,,,,.Distinguishing between stroke and mimic at the bedside: the brain attack study.Stroke.2006;37(3):769–775.
- ,,, et al.Stroke magnetic resonance imaging is accurate in hyperacute intracerebral hemorrhage: a multicenter study on the validity of stroke imaging.Stroke.2004;35(2):502–506.
- ,,, et al.Comparison of MRI and CT for detection of acute intracerebral hemorrhage.JAMA.2004;292(15):1823–1830.
- ,,, et al.Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group.Stroke.2007;38(6):2001–2023.
- ,,.Aneurysmal subarachnoid hemorrhage.N Engl J Med.2006;354(4):387–396.
- ,,,.Subarachnoid haemorrhage.BMJ.2006;333(7561):235–240.
- ,,.Subarachnoid haemorrhage.Lancet.2007;369(9558):306–318.
- ,,.Intracerebral hemorrhage associated with oral anticoagulant therapy: current practices and unresolved questions.Stroke.2006;37(1):256–262.
- ,,, et al.Withdrawal of support in intracerebral hemorrhage may lead to self‐fulfilling prophecies.Neurology.2001;56(6):766–772.
- ,,,.Hospital usage of early do‐not‐resuscitate orders and outcome after intracerebral hemorrhage.Stroke.2004;35(5):1130–1134.
- ,,, et al.Early care limitations independently predict mortality after intracerebral hemorrhage.Neurology.2007;68(20):1651–1657.
- ,,,.Consent for intravenous thrombolysis in acute stroke: review and future directions.Arch Neurol.2007;64(6):785–792.
- .Thrombolysis (tissue plasminogen activator) in stroke: a medicolegal quagmire.Stroke.2006;37(7):1917–1922.
- Tissue plasminogen activator for acute ischemic stroke.The National Institute of Neurological Disorders and Stroke rt‐PA Stroke Study Group.N Engl J Med.1995;333(24):1581–1587.
- ,,, et al.Thrombolysis with alteplase for acute ischaemic stroke in the Safe Implementation of Thrombolysis in Stroke‐Monitoring Study (SITS‐MOST): an observational study.Lancet.2007;369(9558):275–282.
- American College of Emergency Physicians (ACEP). Use of Intravenous tPA for the Management of Acute Stroke in the Emergency Department. ACEP Policy Statement. February 2002. Available at: http://www.acep.org/practres.aspx?id=29834. Accessed May2009.
- American Academy of Emergency Medicine (AAEM). Position statement on the use of intravenous thrombolytic therapy in the treatment of stroke. January 2002. Available at: http://aaem.org/positionstatements/thrombolytictherapy.php. Accessed May2009.
- ,,, et al.Lack of clinical significance of early ischemic changes on computed tomography in acute stroke.JAMA.2001;286(22):2830–2838.
- ,,,,.Thrombolysis for acute stroke in routine clinical practice.Arch Intern Med.2002;162(17):1994–2001.
- ,,, et al.Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke.N Engl J Med.2008;359:1317–1329,1393–1395.
- ,,, et al.Intra‐arterial prourokinase for acute ischemic stroke. The PROACT II study: a randomized controlled trial. Prolyse in Acute Cerebral Thromboembolism.JAMA.1999;282(21):2003–2011.
- Modified MERCI Retriever FDA marketing approval letter. Available at: www.fda.gov/cdrh/pdf6/K062046.pdf. Accessed May2009.
- Penumbra System FDA marketing approval letter. Available at: www.fda.gov/cdrh/pdf7/K072718.pdf. Accessed May2009.
- .Safety of mechanical thrombectomy and intravenous tissue plasminogen activator in acute ischemic stroke. Results of the multi mechanical embolus removal in cerebral ischemia (MERCI) trial, part I.AJNR Am J Neuroradiol.2006;27(6):1177–1182.
- ,,, et al.Safety and efficacy of mechanical embolectomy in acute ischemic stroke: results of the MERCI trial.Stroke.2005;36(7):1432–1438.
- ,,, et al.The Penumbra System: a mechanical device for the treatment of acute stroke due to thromboembolism.AJNR Am J Neuroradiol.2008;29(7):1409–1413.
- ,,, et al.Comparison of CT perfusion and angiography and MRI in selecting stroke patients for acute treatment.Neurology.2007;68(9):694–697.
- ,,, et al.Combined intravenous and intraarterial revascularization therapy using MRI perfusion/diffusion mismatch selection for acute ischemic stroke at 3–6 h after symptom onset.Neurocrit Care.2008;8(3):353–359.
- ,,, et al.Refining the perfusion‐diffusion mismatch hypothesis.Stroke.2005;36(6):1153–1159.
- . DIAS‐2: no benefit of desmoteplase in acute ischemic stroke. Available at: www.medscape.com/viewarticle/557663. Accessed May2009.
- ,,, et al.Effects of alteplase beyond 3 h after stroke in the Echoplanar Imaging Thrombolytic Evaluation Trial (EPITHET): a placebo‐controlled randomised trial.Lancet Neurol.2008;7(4):299–309.
- ,,.Magnetic resonance perfusion diffusion mismatch and thrombolysis in acute ischaemic stroke: a systematic review of the evidence to date.J Neurol Neurosurg Psychiatry.2007;78(5):485–491.
- ,,, et al.Optimal definition for PWI/DWI mismatch in acute ischemic stroke patients.J Cereb Blood Flow Metab.2008;28(5):887–891.
- ,,, et al.Rapid assessment of perfusion‐diffusion mismatch.Stroke.2008;39(1):75–81.
- CAST: randomised placebo‐controlled trial of early aspirin use in 20,000 patients with acute ischaemic stroke.CAST (Chinese Acute Stroke Trial) Collaborative Group.Lancet.1997;349(9066):1641–1649.
- The International Stroke Trial (IST): a randomised trial of aspirin, subcutaneous heparin, both, or neither among 19435 patients with acute ischaemic stroke.International Stroke Trial Collaborative Group.Lancet.1997;349(9065):1569–1581.
- ,,, et al.Dual antiplatelet therapy with clopidogrel and aspirin in symptomatic carotid stenosis evaluated using doppler embolic signal detection: the clopidogrel and aspirin for reduction of emboli in symptomatic carotid stenosis (CARESS) trial.Circulation.2005;111(17):2233–2240.
- ,,, et al.Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population‐based sequential comparison.Lancet.2007;370(9596):1432–1442.
- ,,,,,.Fast assessment of stroke and transient ischaemic attack to prevent early recurrence (FASTER): a randomised controlled pilot trial.Lancet Neurol.2007;6(11):961–969.
- ,,, et al.Antiplatelets versus anticoagulation in cervical artery dissection.Stroke.2007;38(9):2605–2611.
- ,,.Basilar artery occlusion.Neurocrit Care.2004;1(3):319–329.
- ,.Cerebral venous thrombosis: an update.Lancet Neurol.2007;6(2):162–170.
- ,,,,.Outcome in patients with basilar artery occlusion treated conventionally.J Neurol Neurosurg Psychiatry.2005;76(9):1238–1241.
- ,,, et al.Early decompressive surgery in malignant infarction of the middle cerebral artery: a pooled analysis of three randomised controlled trials.Lancet Neurol.2007;6(3):215–222.
- ,,,,,.‘Malignant’ middle cerebral artery territory infarction: clinical course and prognostic signs.Arch Neurol.1996;53(4):309–315.
- ,,,,.Predictors for malignant middle cerebral artery infarctions: a postmortem analysis.Neurology. 282006;66(6):815–820.
- ,,,,,.Poor outcomes in patients who do not receive intravenous tissue plasminogen activator because of mild or improving ischemic stroke.Stroke.2005;36(11):2497–2499.
- Organization WH. MONICA Manual, Part IV: Event Registration. Available at: http://www.ktl.fi/publications/monica/manual/part4/iv‐2.htm#s2. Accessed May2009.
- ,,, et al.Heart disease and stroke statistics 2008 update. A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee.Circulation.200829;117(4):e25–e146.
- .Neurology in the next two decades: report of the Workforce Task Force of the American Academy of Neurology.Neurology.2000;54(4):787–789.
- ,,, et al.Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists.Stroke.2007;38(5):1655–1711.
- ,,,.Conditions that mimic stroke in the emergency department. Implications for acute stroke trials.Arch Neurol.1995;52(11):1119–1122.
- ,,,,.Distinguishing between stroke and mimic at the bedside: the brain attack study.Stroke.2006;37(3):769–775.
- ,,, et al.Stroke magnetic resonance imaging is accurate in hyperacute intracerebral hemorrhage: a multicenter study on the validity of stroke imaging.Stroke.2004;35(2):502–506.
- ,,, et al.Comparison of MRI and CT for detection of acute intracerebral hemorrhage.JAMA.2004;292(15):1823–1830.
- ,,, et al.Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group.Stroke.2007;38(6):2001–2023.
- ,,.Aneurysmal subarachnoid hemorrhage.N Engl J Med.2006;354(4):387–396.
- ,,,.Subarachnoid haemorrhage.BMJ.2006;333(7561):235–240.
- ,,.Subarachnoid haemorrhage.Lancet.2007;369(9558):306–318.
- ,,.Intracerebral hemorrhage associated with oral anticoagulant therapy: current practices and unresolved questions.Stroke.2006;37(1):256–262.
- ,,, et al.Withdrawal of support in intracerebral hemorrhage may lead to self‐fulfilling prophecies.Neurology.2001;56(6):766–772.
- ,,,.Hospital usage of early do‐not‐resuscitate orders and outcome after intracerebral hemorrhage.Stroke.2004;35(5):1130–1134.
- ,,, et al.Early care limitations independently predict mortality after intracerebral hemorrhage.Neurology.2007;68(20):1651–1657.
- ,,,.Consent for intravenous thrombolysis in acute stroke: review and future directions.Arch Neurol.2007;64(6):785–792.
- .Thrombolysis (tissue plasminogen activator) in stroke: a medicolegal quagmire.Stroke.2006;37(7):1917–1922.
- Tissue plasminogen activator for acute ischemic stroke.The National Institute of Neurological Disorders and Stroke rt‐PA Stroke Study Group.N Engl J Med.1995;333(24):1581–1587.
- ,,, et al.Thrombolysis with alteplase for acute ischaemic stroke in the Safe Implementation of Thrombolysis in Stroke‐Monitoring Study (SITS‐MOST): an observational study.Lancet.2007;369(9558):275–282.
- American College of Emergency Physicians (ACEP). Use of Intravenous tPA for the Management of Acute Stroke in the Emergency Department. ACEP Policy Statement. February 2002. Available at: http://www.acep.org/practres.aspx?id=29834. Accessed May2009.
- American Academy of Emergency Medicine (AAEM). Position statement on the use of intravenous thrombolytic therapy in the treatment of stroke. January 2002. Available at: http://aaem.org/positionstatements/thrombolytictherapy.php. Accessed May2009.
- ,,, et al.Lack of clinical significance of early ischemic changes on computed tomography in acute stroke.JAMA.2001;286(22):2830–2838.
- ,,,,.Thrombolysis for acute stroke in routine clinical practice.Arch Intern Med.2002;162(17):1994–2001.
- ,,, et al.Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke.N Engl J Med.2008;359:1317–1329,1393–1395.
- ,,, et al.Intra‐arterial prourokinase for acute ischemic stroke. The PROACT II study: a randomized controlled trial. Prolyse in Acute Cerebral Thromboembolism.JAMA.1999;282(21):2003–2011.
- Modified MERCI Retriever FDA marketing approval letter. Available at: www.fda.gov/cdrh/pdf6/K062046.pdf. Accessed May2009.
- Penumbra System FDA marketing approval letter. Available at: www.fda.gov/cdrh/pdf7/K072718.pdf. Accessed May2009.
- .Safety of mechanical thrombectomy and intravenous tissue plasminogen activator in acute ischemic stroke. Results of the multi mechanical embolus removal in cerebral ischemia (MERCI) trial, part I.AJNR Am J Neuroradiol.2006;27(6):1177–1182.
- ,,, et al.Safety and efficacy of mechanical embolectomy in acute ischemic stroke: results of the MERCI trial.Stroke.2005;36(7):1432–1438.
- ,,, et al.The Penumbra System: a mechanical device for the treatment of acute stroke due to thromboembolism.AJNR Am J Neuroradiol.2008;29(7):1409–1413.
- ,,, et al.Comparison of CT perfusion and angiography and MRI in selecting stroke patients for acute treatment.Neurology.2007;68(9):694–697.
- ,,, et al.Combined intravenous and intraarterial revascularization therapy using MRI perfusion/diffusion mismatch selection for acute ischemic stroke at 3–6 h after symptom onset.Neurocrit Care.2008;8(3):353–359.
- ,,, et al.Refining the perfusion‐diffusion mismatch hypothesis.Stroke.2005;36(6):1153–1159.
- . DIAS‐2: no benefit of desmoteplase in acute ischemic stroke. Available at: www.medscape.com/viewarticle/557663. Accessed May2009.
- ,,, et al.Effects of alteplase beyond 3 h after stroke in the Echoplanar Imaging Thrombolytic Evaluation Trial (EPITHET): a placebo‐controlled randomised trial.Lancet Neurol.2008;7(4):299–309.
- ,,.Magnetic resonance perfusion diffusion mismatch and thrombolysis in acute ischaemic stroke: a systematic review of the evidence to date.J Neurol Neurosurg Psychiatry.2007;78(5):485–491.
- ,,, et al.Optimal definition for PWI/DWI mismatch in acute ischemic stroke patients.J Cereb Blood Flow Metab.2008;28(5):887–891.
- ,,, et al.Rapid assessment of perfusion‐diffusion mismatch.Stroke.2008;39(1):75–81.
- CAST: randomised placebo‐controlled trial of early aspirin use in 20,000 patients with acute ischaemic stroke.CAST (Chinese Acute Stroke Trial) Collaborative Group.Lancet.1997;349(9066):1641–1649.
- The International Stroke Trial (IST): a randomised trial of aspirin, subcutaneous heparin, both, or neither among 19435 patients with acute ischaemic stroke.International Stroke Trial Collaborative Group.Lancet.1997;349(9065):1569–1581.
- ,,, et al.Dual antiplatelet therapy with clopidogrel and aspirin in symptomatic carotid stenosis evaluated using doppler embolic signal detection: the clopidogrel and aspirin for reduction of emboli in symptomatic carotid stenosis (CARESS) trial.Circulation.2005;111(17):2233–2240.
- ,,, et al.Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population‐based sequential comparison.Lancet.2007;370(9596):1432–1442.
- ,,,,,.Fast assessment of stroke and transient ischaemic attack to prevent early recurrence (FASTER): a randomised controlled pilot trial.Lancet Neurol.2007;6(11):961–969.
- ,,, et al.Antiplatelets versus anticoagulation in cervical artery dissection.Stroke.2007;38(9):2605–2611.
- ,,.Basilar artery occlusion.Neurocrit Care.2004;1(3):319–329.
- ,.Cerebral venous thrombosis: an update.Lancet Neurol.2007;6(2):162–170.
- ,,,,.Outcome in patients with basilar artery occlusion treated conventionally.J Neurol Neurosurg Psychiatry.2005;76(9):1238–1241.
- ,,, et al.Early decompressive surgery in malignant infarction of the middle cerebral artery: a pooled analysis of three randomised controlled trials.Lancet Neurol.2007;6(3):215–222.
- ,,,,,.‘Malignant’ middle cerebral artery territory infarction: clinical course and prognostic signs.Arch Neurol.1996;53(4):309–315.
- ,,,,.Predictors for malignant middle cerebral artery infarctions: a postmortem analysis.Neurology. 282006;66(6):815–820.
- ,,,,,.Poor outcomes in patients who do not receive intravenous tissue plasminogen activator because of mild or improving ischemic stroke.Stroke.2005;36(11):2497–2499.
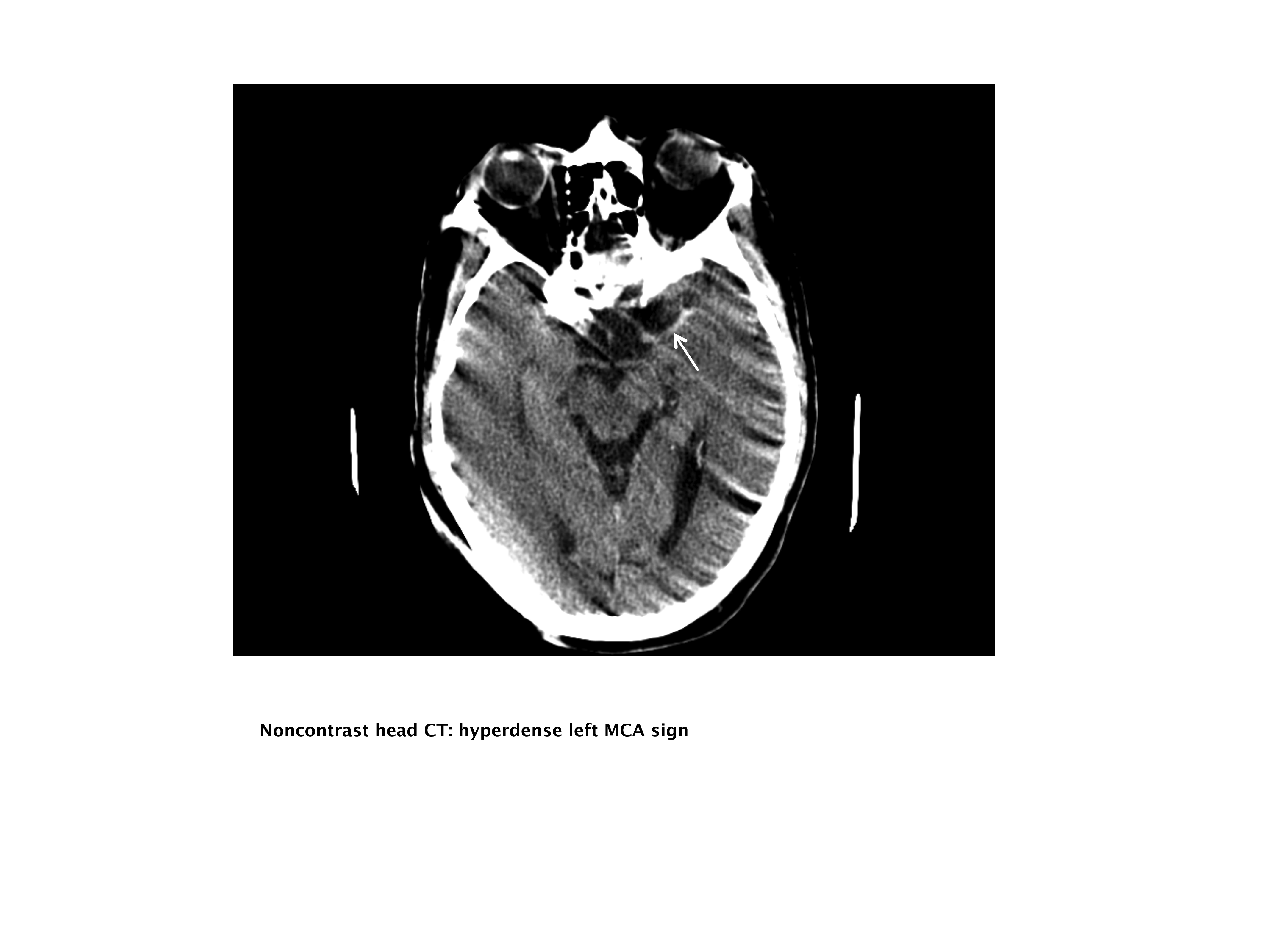
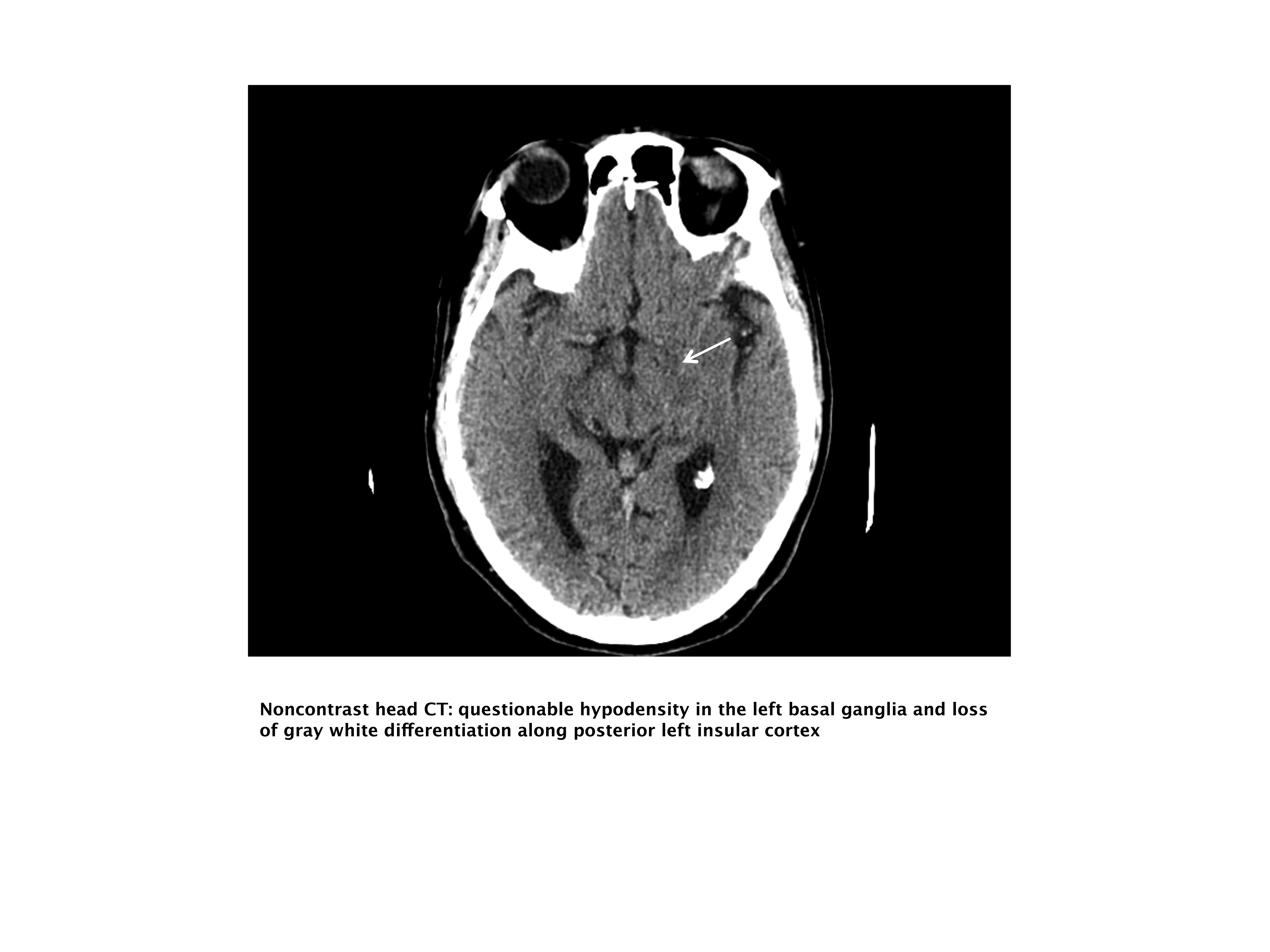
The Hospitalist and NSF
What Is Nephrogenic Systemic Fibrosis?
Nephrogenic systemic fibrosis (NSF) is a systemic fibrosing disease that occurs after exposure to gadolinium‐based contrast (GBC) in the presence of severe renal failure of acute or chronic nature.1, 27 As suggested by its former name, nephrogenic fibrosing dermopathy, the cardinal feature of this disorder is skin involvement. Symptoms begin anywhere from 2 to 75 days after exposure to GBC, though usually within 2 months.27 Initial signs and symptoms may include sharp and sometimes excruciating pain, tightening and burning of the skin associated with redness and swelling, symmetrical involvement, distribution with predilection for the extremities more than the trunk, and sparing of the face. The dependent lower extremities are more severely involved than the upper extremities. Dermal induration may occur in the form of plaques, nodules, and papules resulting in a woody texture on palpation. These findings usually progress over weeks to months with extensive dermal fibrosis involving entire limbs. Ultimately the patient may develop severe joint contractures and marked limitations in mobility.8 A fulminant presentation is seen in approximately 5% of patients who develop a rapidly progressive course over as short a time period as 2 weeks.
Systemic organ involvement including fibrosis of the heart, lung, diaphragm, skeletal muscles, and other organs has been described and has been associated with fatal outcomes.79
Though more frequent in those with end‐stage renal disease (ESRD), NSF has been seen in those with stage 4 and 5 chronic kidney disease (CKD) and acute kidney injury (AKI). Incidence rates have been difficult to calculate due to lack of exposure data in most studies, though 1 small case‐control study found 4.3 cases per 1000 patient years among hemodialysis patients with an absolute risk of 3.4% in the exposed patient.4 Interestingly, incident NSF rates published in a Centers for Disease Control case‐control study of 19 NSF sufferers were much higher for peritoneal dialysis (4.6 cases/100 patients) than for hemodialysis (0.61/100 patients).2 This is likely related to the different GBC clearance achieved with these modalities.
NSF has no predilection for gender, race, nationality or age group. Those with liver disease and lower body weight or lower muscle mass appear to be at greater risk, which may be related to overestimation of glomerular filtration rate (GFR) with falsely low creatinines seen in such patients. Risk is likely increased as well by multiple exposures to GBC in close proximity. Related host cofactors have not been identified, though elevated serum calcium and phosphate concentrations, exposure to high dose erythropoietin, and iron overload have been considered.10, 11
The diagnosis of NSF requires compatible clinical findings along with consistent histopathology. Suspicious clinical findings in a patient with underlying kidney disease (AKI, CKD stages 4 and 5) who has been exposed to a GBC agent, should prompt skin biopsy. An incisional or deep punch biopsy to allow examination of dermis, epidermis and subcutaneous fat is required. The primary feature is the presence of collagen bundles with increased dermal spindle cells that stain for CD34 and procollagen I. Importantly, an inflammatory infiltrate is absent.12, 13
The major differential diagnosis includes scleroderma, eosinophilic fasciitis, morphea, scleromyxedema, and calcific uremic arteriolopathy. Scleroderma is distinguished by clinical findings such as facial involvement, Raynaud's phenomenon, and sclerodactyly with histology demonstrating normal or decreased numbers of fibroblasts on skin biopsy. Scleromyxedema is marked clinically by facial involvement, paraproteinemia on laboratory testing, and presence of inflammation sometimes seen on biopsy. Calcific uremic arteriolopathy (called calciphylaxis by some), which also occurs in those with kidney failure, is distinguished clinically by usually focal skin changes with cutaneous necrosis and ulceration and livedo reticularis; skin biopsy often reveals medial calcification of the vasculature with intimal fibrosis and luminal thrombosis.
What Is the Role of GBC in NSF?
The cause of NSF remained elusive for several years. Initially described in 2006 with several case series confirming the association, the role GBC agents in the pathogenesis of NSF gained widespread acceptance.1, 27 It should be noted that there are 5 cases of NSF described in kidney transplant patients where no exposure to Gadolinium was found.14, 15 Therefore, the possibility of other triggers remains.
The currently proposed pathogenesis needs to be understood in the context of gadolinium's pharmacologic properties. Gadolinium in its free ionic form (Gd3+) is highly toxic and therefore is sequestered by a non‐toxic organic molecule called a chelate.16, 17 Dissociation of the Gd3+ from a chelate may occur through a process called transmetallation when the chelate binds with another endogenous metal such as zinc or copper, allowing the release of free Gd3+. It is this free gadolinium that appears to be culpable in development of NSF.18 GBC chelates can be categorized based on their biochemical structure (linear vs. macrocyclic) and their charge (ionic vs. non‐ionic). Macrocyclic chelates bind Gd3+ more tightly than linear chelates and possess lower dissociation rates,19 which may have implications for possible toxicity.
The prolonged half‐life of GBC in the context of renal failure appears to predispose GBC to transmetallation and dissociation of Gd3+ from its chelate. Following intravenous injection, GBC is excreted unchanged by the kidneys via glomerular filtration. As a result, elimination half‐life, which is approximately 1.6 hours in normal individuals, is increased approximately 4‐ to 33‐fold in renal failure, depending on the level of GFR.16, 17, 20, 21 This increases the potential for Gd3+ dissociation through prolonged circulation times.
It has been postulated that once dissociated, deposition of the Gd3+ ion into skin and other organs sets off a cascade of poorly understood events that result in edema and fibrosis.18 Recent findings of gadolinium deposition in the skin of patients with NSF as well as an animal model of NSF following GBC exposure support this hypothesis.2225 It appears that vascular trauma, endothelial dysfunction or transudation (edema) allows the Gd3+ metal to enter the tissues. This may explain the preponderance of initial symptoms in dependent areas of the limbs.
What Can Be Done to Prevent NSF?
Avoid GBC Exposure in at Risk Patients
GBC agents are contraindicated in those with ESRD, CKD with estimated GFR 30 mL/minute/1.73 m2 (stages 4 and 5) and AKI. It has become common practice to use the 4‐variable Modification of Diet in Renal Disease (MDRD) formula in estimating GFR.26 Importantly, no estimating formula can be used in the context of a rising serum creatinine concentration as occurs with AKI. If a patient has AKI, one must assume a GFR 15 mL/minute until proven otherwise.
In those with low muscle mass the MDRD estimated GFR may overestimate the true GFR.27 Therefore, the Cockcroft‐Gault estimated creatinine clearance or a 24 hour urine‐based creatinine clearance may be useful in identifying at risk patients with underlying CKD.
Choose the Lowest Risk GBC Agent
When GBC use is deemed necessary in the high risk individual, an agent with a macrocyclic chelate (gadoteridol in the United States) is recommended.28 No published cases of NSF have been described with singular use of such agents. In addition, a retrospective study demonstrated no cases of NSF in ESRD patients on hemodialysis exposed to gadoteridol over a 7‐year period.29 This is not unexpected given the pharmacologic properties of this GBC agent.
Gadodiamide, a linear, non‐ionic agent, appears to produce the greatest risk of NSF as the largest number of NSF cases has been reported with this agent. By October 2007, 283 of 447 cases reported to the Food and Drug Administration (FDA) were exposed to gadodiamide.28 The significant preponderance with this agent is unlikely related to market share, reporting bias or publication bias. Gadopentetate, a linear, ionic agent, which had the greatest market share during this time, was responsible for approximately a quarter of cases reported to the FDA.28 Based on these data, gadodiamide and gadopentetate (and probably all linear agents) should be avoided in high risk patients.
Use Lower Doses of GBC
The FDA approved dose of all GBC agents, except the macrocyclic agent gadoteridol, is 0.1 mmol/kg.30 It appears that higher off‐label doses of GBC agents (0.3‐0.4 mmol/kg) which have been utilized for vascular studies (magnetic resonance angiography [MRA]), may have contributed to the emergence of NSF several years after these agents became available.
Develop a Protocol With Radiology and Nephrology Departments
Assessment of Renal Function Prior to Contrast Administration Is Required
Radiology departments should identify those with ESRD, CKD with estimated GFR 30 mL/minute/1.73 m2 (stages 4 and 5) and AKI. Using the 4‐variable MDRD formula in estimating GFR with the caveats previously noted, radiology departments will identify most at‐risk patients. Since the MDRD formula will be inaccurate in the setting of ESRD and AKI, these diagnoses should be determined through other means (for example, the patient's medical history) as part of the consent process.
Alternative Radiologic Imaging Modalities to GBC Enhanced Magnetic Resonance Imaging Should Be Utilized When Suitable in Those at High Risk
Newer techniques should be investigated as alternatives to GBC exposure. These include Magnetic Resonance Imaging (MRI) without GBC‐enhancement, where options such as 3D time‐of‐flight MRA, phase‐contrast angiography, and arterial spin labeling‐MR provide excellent information about blood vessels and blood flow.31 MRI with ultra‐small paramagnetic iron oxide particles may offer a future alternative in those that need a contrast‐based scan for diagnosis.32
However, since contrast enhanced MRI/MRA studies remain extremely important imaging modalities, their use may be required in some high risk individuals. In this circumstance, a macrocyclic chelate employed at the lowest dose possible, is recommended. The radiologist and nephrologist should be consulted in these instances.
Hemodialysis
Although hemodialysis efficiently clears GBC, its removal is not complete. Furthermore, it is not clear whether the damage has already occurred by the time a hemodialysis treatment can be instituted.33 It should be recognized that GBC removal after one treatment averages 65% to 73.8%; 3 to 4 sessions are required to remove 99% of the contrast agent.21, 34 Peritoneal dialysis on the other hand is an ineffective method of GBC removal (T1/2 of 52.7 hours).21 Because not all of the circulating Gd3+ is removed with a single hemodialysis treatment, prolonged tissue exposure occurs in these patients. This is reflected by the development of NSF in patients despite undergoing consecutive hemodialysis treatments following GBC exposure.3 Therefore, based on incomplete GBC removal with hemodialysis and the lack of evidence supporting prevention of NSF with this modality, we and others33, 35 strongly recommend avoidance of GBC in all patients with advanced kidney disease (GFR 30), regardless of the availability of hemodialysis. As such, the ability to perform hemodialysis after GBC in and of itself does not justify such exposure. However, if GBC use is deemed essential, then immediate hemodialysis should be strongly considered after exposure with further treatment on consecutive days.
Once NSF Develops, What Treatments Options are Available?
Unfortunately there is lack of a universally effective therapy for NSF. Several interventions have been described mainly in anecdotal case reports and very small case series. They have been recently reviewed (Table 2).360
| GBC Formulation | Year of Approval | Charge | Molecular Structure | Probable Risk of NSF* |
|---|---|---|---|---|
| Gadopentetate (Magnevist) | 1988 | Ionic | Linear | Medium |
| Gadoteridol (Prohance) | 1992 | Non‐ionic | Cyclic | Very low |
| Gadodiamide (Omniscan) | 1993 | Non‐ionic | Linear | High |
| Gadoversetamide (OptiMARK) | 1999 | Non‐ionic | Linear | Medium |
| Gadobenate (MultiHance) | 2004 | Ionic | Linear | Low |
|
| Therapies most likely to benefit |
| Kidney transplant (in ESRD) |
| Physical therapy |
| Pain control |
| Therapies with anecdotal success |
| Extracorporeal photopharesis |
| Sodium thiosulfate |
| Therapies with limited success |
| Drugs: Glucocorticoids, Pentoxifylline, Cyclophosphamide, Thalidomide |
| Immunomodulatory: Plasmapharesis, Intravenous immunoglobulin |
| Local: Intralesional IFN‐alpha, topical calcipotriene, other phototherapy |
Physical therapy is the mainstay of treatment for NSF. Physical therapy (and occupational therapy if needed) is essential to help prevent or slow the progression of joint contractures. Adequate pain relief, often with narcotics, is essential for patient comfort and to allow tolerance of physical therapy. Therapies with anecdotal benefit include extracorporeal photopheresis and infusions of sodium thiosulfate, a substance with chelating properties. Other interventions, such as immunosuppressive agents, topical agents and other phototherapies have shown limited success.
AKI resolution has been observed to result in regression of lesions.1, 3740 Presumably, resolution of the AKI allows for clearance of gadolinium and other profibrotic mediators, though definitive evidence of this is not available. Based on the observed response to AKI recovery, it is not surprising that improvement after kidney transplantation has also been described.1, 41 However, responses have not been consistent.39, 42
Consensus Guidelines and Recommendations
Nephrology societies have not yet developed consensus guidelines. Only the European Society of Urogenital Radiology has issued guidelines to date.43 These guidelines are consistent with expert opinions published elsewhere and are reflected in our approach regarding prevention of NSF (Table 3).
|
| 1. GBC agents are contraindicated in patients on dialysis regardless of availability of rapid treatment after exposure |
| 2. Avoid MRI with GBC in those with GFR 30 ml/min (estimated by MDRD formula) |
| MDRD formula may overestimate GFR in those of low weightconsider Cockcroft‐Gault calculation or 24 hour urine collection for creatinine clearance |
| MDRD is invalid in patient with a rising serum creatinine concentration. Assume GFR 30 in those with acutely rising serum creatinine concentration |
| 3. Consider alternative imaging studies or MRI studies without Gadolinium consult radiologist |
| 4. If GBC study is a necessity, then as low a dose as possible of a macrocyclic chelate would be recommended |
| 5. If an exposure to gadolinium occurs in ESRD, hemodialysis should be performed as soon as possible and repeated on consecutive days |
| 6. If an exposure to gadolinium occurs in CKD 4 or 5 or AKI patient (not on dialysis), an individualized approach should be undertaken when considering temporary catheter placement and initiation of hemodialysis |
The FDA has sent out several alerts since June 2006, the most recent in May 2007.30, 4446 In its Information for Healthcare Professionals alert, the FDA outlines recommendations. These are included in our final recommendations shown in Table 3.30 Those with a recent liver transplant, or those with chronic liver disease, who have associated kidney insufficiency of any severity, have also been identified by the FDA as an at risk group. This is based on reports of NSF occurring more commonly in patients with AKI who have these underlying conditions.47
Conclusions
With the high and increasing rates of AKI, CKD and ESRD seen among hospitalized patients,48 the need for vigilance when obtaining imaging with GBC agents becomes particularly important in the inpatient setting. As a preventable disease, it is incumbent upon us to fully understand the risk factors and potential pitfalls that may result in a patient exposed to these agents. The hospitalist has the unique role of acting as a firewall between the patient and the imaging study that may put him or her at risk for this devastating disorder.
Identification of GBC as a major culprit in the development of NSF and hence avoidance of this agent in those at the highest risk is expected to reduce the incidence of NSF. It is likely that the future will bring further understanding of the underlying mechanisms of gadolinium‐induced NSF and with this understanding, even safer strategies for GBC usage. However, until safer contrast agents become available, avoidance of GBC exposure in those with advanced acute or CKD remains our most important defense.
- .Gadolinium–a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?Nephrol Dial Transplant.2006;21(4):1104–1108.
- Nephrogenic fibrosing dermopathy associated with exposure to gadolinium‐containing contrast agents–St. Louis, Missouri, 2002–2006.MMWR Morb Mortal Wkly Rep.2007;56(7):137–141.
- ,,,,,.Gadodiamide‐associated nephrogenic systemic fibrosis: why radiologists should be concerned.AJR Am J Roentgenol.2007;188(2):586–592.
- ,,.Nephrogenic systemic fibrosis: a population study examining the relationship of disease development to gadolinium exposure.Clin J Am Soc Nephrol.2007;2(2):264–267.
- ,,,,.Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (omniscan).Invest Radiol.2007;42(2):139–145.
- ,,, et al.Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast‐enhanced magnetic resonance imaging.J Am Soc Nephrol.2006;17(9):2359–2362.
- ,,, et al.Nephrogenic systemic fibrosis: risk factors and incidence estimation.Radiology.2007;243(1):148–157.
- ,,.Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy).Curr Opin Rheumatol.2006;18(6):614–617.
- . Nephrogenic Fibrosing Dermopathy [NFD/NSF Website]. 2001–2007. Available at http://www.icnfdr.org. Accessed December 2009.
- ,,,,.Case‐control study of gadodiamide‐related nephrogenic systemic fibrosis.Nephrol Dial Transplant.2007;22(11):3174–3178.
- ,,, et al.Nephrogenic fibrosing dermopathy and high‐dose erythropoietin therapy.Ann Intern Med.2006;145(3):234–235.
- ,.Nephrogenic systemic fibrosis: an update.Curr Rheumatol Rep.2006;8(2):151–157.
- ,.Nephrogenic systemic fibrosis: early recognition and treatment.Semin Dial.2008;21(2):123–128.
- ,,.Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature.Am J Transplant.2007;7(10):2425–2432.
- .Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: a summary of the medical literature reporting.Eur J Radiol.2008;66(2):230–234.
- .MR contrast agents, the old and the new.Eur J Radiol.2006;60(3):314–323.
- ,,,,,.Magnetic resonance contrast agents: from the bench to the patient.Curr Pharm Des.2005;11(31):4079–4098.
- .Tissue deposition of gadolinium and development of NSF: a convergence of factors.Semin Dial.232008.
- .Safety of magnetic resonance contrast media.Top Magn Reson Imaging.2001;12(4):309–314.
- ,,, et al.Safety and pharmacokinetic profile of gadobenate dimeglumine in subjects with renal impairment.Invest Radiol.1999;34(7):443–448.
- ,,.Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis.Acad Radiol.1998;5(7):491–502.
- ,,.Gadolinium deposition in nephrogenic fibrosing dermopathy.J Am Acad Dermatol.2007;56(1):27–30.
- ,,,,.Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis.J Am Acad Dermatol.2007;56(1):21–26.
- ,,.Gadolinium is quantifiable within the tissue of patients with nephrogenic systemic fibrosis.J Am Acad Dermatol.2007;56(4):710–712.
- ,,,,,.A preclinical study to investigate the development of nephrogenic systemic fibrosis: a possible role for gadolinium‐based contrast media.Invest Radiol.2008;43(1):65–75.
- ,,,.A simplified equation to predict GFR from S‐creatinine [abstract].J Am Soc Nephrol.2000;11:155A.
- ,,,.Assessing kidney function–measured and estimated glomerular filtration rate.N Engl J Med.2006;354(23):2473–2483.
- ,.Nephrogenic systemic fibrosis risk: is there a difference between gadolinium‐based contrast agents?Semin Dial.2008;21(2):129–134.
- .Risk for nephrogenic systemic fibrosis with gadoteridol (ProHance) in patients who are on long‐term hemodialysis.Clin J Am Soc Nephrol.2008;3(3):747–751.
- US Food and Drug Administration: Information for Healthcare Professionals: Gadolinium‐Containing Contrast Agents for Magnetic Resonance Imaging (MRI) ProHance, and MultiHance). Available at: http://www.fda.gov/cder/drug/InfoSheets/HCP/gcca_200705HCP.pdf. Accessed December 2009.
- ,.Nephrogenic systemic fibrosis: non‐gadolinium options for the imaging of CKD/ESRD patients.Semin Dial.2008;21(2):160–165.
- ,,, et al.Ultrasmall superparamagnetic iron oxides (USPIOs): a future alternative magnetic resonance (MR) contrast agent for patients at risk for nephrogenic systemic fibrosis (NSF)?Kidney Int.2008;75(5):465–474.
- .Dialytic therapies to prevent NSF following gadolinium exposure in high‐risk patients.Semin Dial.2008;21(2):145–149.
- ,,,,.Dialyzability of gadodiamide in hemodialysis patients.Radiat Med.2006;24(6):445–451.
- ,,,,,.Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI.Cleve Clin J Med.2008;75(2):95–97, 103–104, 106 passim.
- ,,.Treatment of nephrogenic systemic fibrosis: limited options but hope for the future.Semin Dial.2008;21(2):155–159.
- ,,,,.Nephrogenic fibrosing dermopathy.Am J Dermatopathol.2001;23(5):383–393.
- ,,,,.Nephrogenic fibrosing dermopathy: a novel cutaneous fibrosing disorder in patients with renal failure.Am J Med.2003;114(7):563–572.
- ,,,.Nephrogenic systemic fibrosis: relationship to gadolinium and response to photopheresis.Arch Dermatol.2007;143(8):1025–1030.
- ,,,.A case of nephrogenic fibrosing dermopathy.Ann Acad Med Singapore.2004;33(4):527–529.
- ,,,,,.Nephrogenic fibrosing dermopathy: two pediatric cases.J Pediatr.2003;143(5):678–681.
- ,,.Nephrogenic fibrosing dermopathy in children.Pediatr Nephrol.2006;21(9):1307–1311.
- .ESUR guideline: gadolinium‐based contrast media and nephrogenic systemic fibrosis.Eur Radiol.2007;17(10):2692–2696.
- US Food and Drug Administration: FDA News: FDA Requests Boxed Warning for Contrast Agents Used to Improve MRI Images. Available at: http://www.fda.gov/bbs/topics/NEWS/2007/NEW01638.html. Accessed December 2009.
- US Food and Drug Administration: Public Health Advisory: Gadolinium‐containing Contrast Agents for Magnetic Resonance Imaging (MRI): Omniscan, OptiMARK, Magnevist, ProHance, and MultiHance. Available at: http://www.fda.gov/cder/drug/advisory/gadolinium_agents.htm. Accessed December 2009.
- US Food and Drug Administration: Public Health Advisory: Update on Magnetic Resonance Imaging (MRI) Contrast Agents Containing Gadolinium and Nephrogenic Fibrosing Dermopathy. Available at: http://www.fda.gov/cder/drug/advisory/gadolinium_agents_20061222.htm. Accessed December 2009.
- ,,, et al.Nephrogenic systemic fibrosis among liver transplant recipients: a single institution experience and topic update.Am J Transplant.2006;6(9):2212–2217.
- Hospitalization discharge diagnoses for kidney disease–United States, 1980–2005.MMWR Morb Mortal Wkly Rep. 282008;57(12):309–312.
- ,,,.Response to the FDA's May 23, 2007, nephrogenic systemic fibrosis update.Radiology.2008;246(1):11–14.
- .How should nephrologists approach gadolinium‐based contrast imaging in patients with kidney disease?Clin J Am Soc Nephrol.2008;3(3):649–651.
What Is Nephrogenic Systemic Fibrosis?
Nephrogenic systemic fibrosis (NSF) is a systemic fibrosing disease that occurs after exposure to gadolinium‐based contrast (GBC) in the presence of severe renal failure of acute or chronic nature.1, 27 As suggested by its former name, nephrogenic fibrosing dermopathy, the cardinal feature of this disorder is skin involvement. Symptoms begin anywhere from 2 to 75 days after exposure to GBC, though usually within 2 months.27 Initial signs and symptoms may include sharp and sometimes excruciating pain, tightening and burning of the skin associated with redness and swelling, symmetrical involvement, distribution with predilection for the extremities more than the trunk, and sparing of the face. The dependent lower extremities are more severely involved than the upper extremities. Dermal induration may occur in the form of plaques, nodules, and papules resulting in a woody texture on palpation. These findings usually progress over weeks to months with extensive dermal fibrosis involving entire limbs. Ultimately the patient may develop severe joint contractures and marked limitations in mobility.8 A fulminant presentation is seen in approximately 5% of patients who develop a rapidly progressive course over as short a time period as 2 weeks.
Systemic organ involvement including fibrosis of the heart, lung, diaphragm, skeletal muscles, and other organs has been described and has been associated with fatal outcomes.79
Though more frequent in those with end‐stage renal disease (ESRD), NSF has been seen in those with stage 4 and 5 chronic kidney disease (CKD) and acute kidney injury (AKI). Incidence rates have been difficult to calculate due to lack of exposure data in most studies, though 1 small case‐control study found 4.3 cases per 1000 patient years among hemodialysis patients with an absolute risk of 3.4% in the exposed patient.4 Interestingly, incident NSF rates published in a Centers for Disease Control case‐control study of 19 NSF sufferers were much higher for peritoneal dialysis (4.6 cases/100 patients) than for hemodialysis (0.61/100 patients).2 This is likely related to the different GBC clearance achieved with these modalities.
NSF has no predilection for gender, race, nationality or age group. Those with liver disease and lower body weight or lower muscle mass appear to be at greater risk, which may be related to overestimation of glomerular filtration rate (GFR) with falsely low creatinines seen in such patients. Risk is likely increased as well by multiple exposures to GBC in close proximity. Related host cofactors have not been identified, though elevated serum calcium and phosphate concentrations, exposure to high dose erythropoietin, and iron overload have been considered.10, 11
The diagnosis of NSF requires compatible clinical findings along with consistent histopathology. Suspicious clinical findings in a patient with underlying kidney disease (AKI, CKD stages 4 and 5) who has been exposed to a GBC agent, should prompt skin biopsy. An incisional or deep punch biopsy to allow examination of dermis, epidermis and subcutaneous fat is required. The primary feature is the presence of collagen bundles with increased dermal spindle cells that stain for CD34 and procollagen I. Importantly, an inflammatory infiltrate is absent.12, 13
The major differential diagnosis includes scleroderma, eosinophilic fasciitis, morphea, scleromyxedema, and calcific uremic arteriolopathy. Scleroderma is distinguished by clinical findings such as facial involvement, Raynaud's phenomenon, and sclerodactyly with histology demonstrating normal or decreased numbers of fibroblasts on skin biopsy. Scleromyxedema is marked clinically by facial involvement, paraproteinemia on laboratory testing, and presence of inflammation sometimes seen on biopsy. Calcific uremic arteriolopathy (called calciphylaxis by some), which also occurs in those with kidney failure, is distinguished clinically by usually focal skin changes with cutaneous necrosis and ulceration and livedo reticularis; skin biopsy often reveals medial calcification of the vasculature with intimal fibrosis and luminal thrombosis.
What Is the Role of GBC in NSF?
The cause of NSF remained elusive for several years. Initially described in 2006 with several case series confirming the association, the role GBC agents in the pathogenesis of NSF gained widespread acceptance.1, 27 It should be noted that there are 5 cases of NSF described in kidney transplant patients where no exposure to Gadolinium was found.14, 15 Therefore, the possibility of other triggers remains.
The currently proposed pathogenesis needs to be understood in the context of gadolinium's pharmacologic properties. Gadolinium in its free ionic form (Gd3+) is highly toxic and therefore is sequestered by a non‐toxic organic molecule called a chelate.16, 17 Dissociation of the Gd3+ from a chelate may occur through a process called transmetallation when the chelate binds with another endogenous metal such as zinc or copper, allowing the release of free Gd3+. It is this free gadolinium that appears to be culpable in development of NSF.18 GBC chelates can be categorized based on their biochemical structure (linear vs. macrocyclic) and their charge (ionic vs. non‐ionic). Macrocyclic chelates bind Gd3+ more tightly than linear chelates and possess lower dissociation rates,19 which may have implications for possible toxicity.
The prolonged half‐life of GBC in the context of renal failure appears to predispose GBC to transmetallation and dissociation of Gd3+ from its chelate. Following intravenous injection, GBC is excreted unchanged by the kidneys via glomerular filtration. As a result, elimination half‐life, which is approximately 1.6 hours in normal individuals, is increased approximately 4‐ to 33‐fold in renal failure, depending on the level of GFR.16, 17, 20, 21 This increases the potential for Gd3+ dissociation through prolonged circulation times.
It has been postulated that once dissociated, deposition of the Gd3+ ion into skin and other organs sets off a cascade of poorly understood events that result in edema and fibrosis.18 Recent findings of gadolinium deposition in the skin of patients with NSF as well as an animal model of NSF following GBC exposure support this hypothesis.2225 It appears that vascular trauma, endothelial dysfunction or transudation (edema) allows the Gd3+ metal to enter the tissues. This may explain the preponderance of initial symptoms in dependent areas of the limbs.
What Can Be Done to Prevent NSF?
Avoid GBC Exposure in at Risk Patients
GBC agents are contraindicated in those with ESRD, CKD with estimated GFR 30 mL/minute/1.73 m2 (stages 4 and 5) and AKI. It has become common practice to use the 4‐variable Modification of Diet in Renal Disease (MDRD) formula in estimating GFR.26 Importantly, no estimating formula can be used in the context of a rising serum creatinine concentration as occurs with AKI. If a patient has AKI, one must assume a GFR 15 mL/minute until proven otherwise.
In those with low muscle mass the MDRD estimated GFR may overestimate the true GFR.27 Therefore, the Cockcroft‐Gault estimated creatinine clearance or a 24 hour urine‐based creatinine clearance may be useful in identifying at risk patients with underlying CKD.
Choose the Lowest Risk GBC Agent
When GBC use is deemed necessary in the high risk individual, an agent with a macrocyclic chelate (gadoteridol in the United States) is recommended.28 No published cases of NSF have been described with singular use of such agents. In addition, a retrospective study demonstrated no cases of NSF in ESRD patients on hemodialysis exposed to gadoteridol over a 7‐year period.29 This is not unexpected given the pharmacologic properties of this GBC agent.
Gadodiamide, a linear, non‐ionic agent, appears to produce the greatest risk of NSF as the largest number of NSF cases has been reported with this agent. By October 2007, 283 of 447 cases reported to the Food and Drug Administration (FDA) were exposed to gadodiamide.28 The significant preponderance with this agent is unlikely related to market share, reporting bias or publication bias. Gadopentetate, a linear, ionic agent, which had the greatest market share during this time, was responsible for approximately a quarter of cases reported to the FDA.28 Based on these data, gadodiamide and gadopentetate (and probably all linear agents) should be avoided in high risk patients.
Use Lower Doses of GBC
The FDA approved dose of all GBC agents, except the macrocyclic agent gadoteridol, is 0.1 mmol/kg.30 It appears that higher off‐label doses of GBC agents (0.3‐0.4 mmol/kg) which have been utilized for vascular studies (magnetic resonance angiography [MRA]), may have contributed to the emergence of NSF several years after these agents became available.
Develop a Protocol With Radiology and Nephrology Departments
Assessment of Renal Function Prior to Contrast Administration Is Required
Radiology departments should identify those with ESRD, CKD with estimated GFR 30 mL/minute/1.73 m2 (stages 4 and 5) and AKI. Using the 4‐variable MDRD formula in estimating GFR with the caveats previously noted, radiology departments will identify most at‐risk patients. Since the MDRD formula will be inaccurate in the setting of ESRD and AKI, these diagnoses should be determined through other means (for example, the patient's medical history) as part of the consent process.
Alternative Radiologic Imaging Modalities to GBC Enhanced Magnetic Resonance Imaging Should Be Utilized When Suitable in Those at High Risk
Newer techniques should be investigated as alternatives to GBC exposure. These include Magnetic Resonance Imaging (MRI) without GBC‐enhancement, where options such as 3D time‐of‐flight MRA, phase‐contrast angiography, and arterial spin labeling‐MR provide excellent information about blood vessels and blood flow.31 MRI with ultra‐small paramagnetic iron oxide particles may offer a future alternative in those that need a contrast‐based scan for diagnosis.32
However, since contrast enhanced MRI/MRA studies remain extremely important imaging modalities, their use may be required in some high risk individuals. In this circumstance, a macrocyclic chelate employed at the lowest dose possible, is recommended. The radiologist and nephrologist should be consulted in these instances.
Hemodialysis
Although hemodialysis efficiently clears GBC, its removal is not complete. Furthermore, it is not clear whether the damage has already occurred by the time a hemodialysis treatment can be instituted.33 It should be recognized that GBC removal after one treatment averages 65% to 73.8%; 3 to 4 sessions are required to remove 99% of the contrast agent.21, 34 Peritoneal dialysis on the other hand is an ineffective method of GBC removal (T1/2 of 52.7 hours).21 Because not all of the circulating Gd3+ is removed with a single hemodialysis treatment, prolonged tissue exposure occurs in these patients. This is reflected by the development of NSF in patients despite undergoing consecutive hemodialysis treatments following GBC exposure.3 Therefore, based on incomplete GBC removal with hemodialysis and the lack of evidence supporting prevention of NSF with this modality, we and others33, 35 strongly recommend avoidance of GBC in all patients with advanced kidney disease (GFR 30), regardless of the availability of hemodialysis. As such, the ability to perform hemodialysis after GBC in and of itself does not justify such exposure. However, if GBC use is deemed essential, then immediate hemodialysis should be strongly considered after exposure with further treatment on consecutive days.
Once NSF Develops, What Treatments Options are Available?
Unfortunately there is lack of a universally effective therapy for NSF. Several interventions have been described mainly in anecdotal case reports and very small case series. They have been recently reviewed (Table 2).360
| GBC Formulation | Year of Approval | Charge | Molecular Structure | Probable Risk of NSF* |
|---|---|---|---|---|
| Gadopentetate (Magnevist) | 1988 | Ionic | Linear | Medium |
| Gadoteridol (Prohance) | 1992 | Non‐ionic | Cyclic | Very low |
| Gadodiamide (Omniscan) | 1993 | Non‐ionic | Linear | High |
| Gadoversetamide (OptiMARK) | 1999 | Non‐ionic | Linear | Medium |
| Gadobenate (MultiHance) | 2004 | Ionic | Linear | Low |
|
| Therapies most likely to benefit |
| Kidney transplant (in ESRD) |
| Physical therapy |
| Pain control |
| Therapies with anecdotal success |
| Extracorporeal photopharesis |
| Sodium thiosulfate |
| Therapies with limited success |
| Drugs: Glucocorticoids, Pentoxifylline, Cyclophosphamide, Thalidomide |
| Immunomodulatory: Plasmapharesis, Intravenous immunoglobulin |
| Local: Intralesional IFN‐alpha, topical calcipotriene, other phototherapy |
Physical therapy is the mainstay of treatment for NSF. Physical therapy (and occupational therapy if needed) is essential to help prevent or slow the progression of joint contractures. Adequate pain relief, often with narcotics, is essential for patient comfort and to allow tolerance of physical therapy. Therapies with anecdotal benefit include extracorporeal photopheresis and infusions of sodium thiosulfate, a substance with chelating properties. Other interventions, such as immunosuppressive agents, topical agents and other phototherapies have shown limited success.
AKI resolution has been observed to result in regression of lesions.1, 3740 Presumably, resolution of the AKI allows for clearance of gadolinium and other profibrotic mediators, though definitive evidence of this is not available. Based on the observed response to AKI recovery, it is not surprising that improvement after kidney transplantation has also been described.1, 41 However, responses have not been consistent.39, 42
Consensus Guidelines and Recommendations
Nephrology societies have not yet developed consensus guidelines. Only the European Society of Urogenital Radiology has issued guidelines to date.43 These guidelines are consistent with expert opinions published elsewhere and are reflected in our approach regarding prevention of NSF (Table 3).
|
| 1. GBC agents are contraindicated in patients on dialysis regardless of availability of rapid treatment after exposure |
| 2. Avoid MRI with GBC in those with GFR 30 ml/min (estimated by MDRD formula) |
| MDRD formula may overestimate GFR in those of low weightconsider Cockcroft‐Gault calculation or 24 hour urine collection for creatinine clearance |
| MDRD is invalid in patient with a rising serum creatinine concentration. Assume GFR 30 in those with acutely rising serum creatinine concentration |
| 3. Consider alternative imaging studies or MRI studies without Gadolinium consult radiologist |
| 4. If GBC study is a necessity, then as low a dose as possible of a macrocyclic chelate would be recommended |
| 5. If an exposure to gadolinium occurs in ESRD, hemodialysis should be performed as soon as possible and repeated on consecutive days |
| 6. If an exposure to gadolinium occurs in CKD 4 or 5 or AKI patient (not on dialysis), an individualized approach should be undertaken when considering temporary catheter placement and initiation of hemodialysis |
The FDA has sent out several alerts since June 2006, the most recent in May 2007.30, 4446 In its Information for Healthcare Professionals alert, the FDA outlines recommendations. These are included in our final recommendations shown in Table 3.30 Those with a recent liver transplant, or those with chronic liver disease, who have associated kidney insufficiency of any severity, have also been identified by the FDA as an at risk group. This is based on reports of NSF occurring more commonly in patients with AKI who have these underlying conditions.47
Conclusions
With the high and increasing rates of AKI, CKD and ESRD seen among hospitalized patients,48 the need for vigilance when obtaining imaging with GBC agents becomes particularly important in the inpatient setting. As a preventable disease, it is incumbent upon us to fully understand the risk factors and potential pitfalls that may result in a patient exposed to these agents. The hospitalist has the unique role of acting as a firewall between the patient and the imaging study that may put him or her at risk for this devastating disorder.
Identification of GBC as a major culprit in the development of NSF and hence avoidance of this agent in those at the highest risk is expected to reduce the incidence of NSF. It is likely that the future will bring further understanding of the underlying mechanisms of gadolinium‐induced NSF and with this understanding, even safer strategies for GBC usage. However, until safer contrast agents become available, avoidance of GBC exposure in those with advanced acute or CKD remains our most important defense.
What Is Nephrogenic Systemic Fibrosis?
Nephrogenic systemic fibrosis (NSF) is a systemic fibrosing disease that occurs after exposure to gadolinium‐based contrast (GBC) in the presence of severe renal failure of acute or chronic nature.1, 27 As suggested by its former name, nephrogenic fibrosing dermopathy, the cardinal feature of this disorder is skin involvement. Symptoms begin anywhere from 2 to 75 days after exposure to GBC, though usually within 2 months.27 Initial signs and symptoms may include sharp and sometimes excruciating pain, tightening and burning of the skin associated with redness and swelling, symmetrical involvement, distribution with predilection for the extremities more than the trunk, and sparing of the face. The dependent lower extremities are more severely involved than the upper extremities. Dermal induration may occur in the form of plaques, nodules, and papules resulting in a woody texture on palpation. These findings usually progress over weeks to months with extensive dermal fibrosis involving entire limbs. Ultimately the patient may develop severe joint contractures and marked limitations in mobility.8 A fulminant presentation is seen in approximately 5% of patients who develop a rapidly progressive course over as short a time period as 2 weeks.
Systemic organ involvement including fibrosis of the heart, lung, diaphragm, skeletal muscles, and other organs has been described and has been associated with fatal outcomes.79
Though more frequent in those with end‐stage renal disease (ESRD), NSF has been seen in those with stage 4 and 5 chronic kidney disease (CKD) and acute kidney injury (AKI). Incidence rates have been difficult to calculate due to lack of exposure data in most studies, though 1 small case‐control study found 4.3 cases per 1000 patient years among hemodialysis patients with an absolute risk of 3.4% in the exposed patient.4 Interestingly, incident NSF rates published in a Centers for Disease Control case‐control study of 19 NSF sufferers were much higher for peritoneal dialysis (4.6 cases/100 patients) than for hemodialysis (0.61/100 patients).2 This is likely related to the different GBC clearance achieved with these modalities.
NSF has no predilection for gender, race, nationality or age group. Those with liver disease and lower body weight or lower muscle mass appear to be at greater risk, which may be related to overestimation of glomerular filtration rate (GFR) with falsely low creatinines seen in such patients. Risk is likely increased as well by multiple exposures to GBC in close proximity. Related host cofactors have not been identified, though elevated serum calcium and phosphate concentrations, exposure to high dose erythropoietin, and iron overload have been considered.10, 11
The diagnosis of NSF requires compatible clinical findings along with consistent histopathology. Suspicious clinical findings in a patient with underlying kidney disease (AKI, CKD stages 4 and 5) who has been exposed to a GBC agent, should prompt skin biopsy. An incisional or deep punch biopsy to allow examination of dermis, epidermis and subcutaneous fat is required. The primary feature is the presence of collagen bundles with increased dermal spindle cells that stain for CD34 and procollagen I. Importantly, an inflammatory infiltrate is absent.12, 13
The major differential diagnosis includes scleroderma, eosinophilic fasciitis, morphea, scleromyxedema, and calcific uremic arteriolopathy. Scleroderma is distinguished by clinical findings such as facial involvement, Raynaud's phenomenon, and sclerodactyly with histology demonstrating normal or decreased numbers of fibroblasts on skin biopsy. Scleromyxedema is marked clinically by facial involvement, paraproteinemia on laboratory testing, and presence of inflammation sometimes seen on biopsy. Calcific uremic arteriolopathy (called calciphylaxis by some), which also occurs in those with kidney failure, is distinguished clinically by usually focal skin changes with cutaneous necrosis and ulceration and livedo reticularis; skin biopsy often reveals medial calcification of the vasculature with intimal fibrosis and luminal thrombosis.
What Is the Role of GBC in NSF?
The cause of NSF remained elusive for several years. Initially described in 2006 with several case series confirming the association, the role GBC agents in the pathogenesis of NSF gained widespread acceptance.1, 27 It should be noted that there are 5 cases of NSF described in kidney transplant patients where no exposure to Gadolinium was found.14, 15 Therefore, the possibility of other triggers remains.
The currently proposed pathogenesis needs to be understood in the context of gadolinium's pharmacologic properties. Gadolinium in its free ionic form (Gd3+) is highly toxic and therefore is sequestered by a non‐toxic organic molecule called a chelate.16, 17 Dissociation of the Gd3+ from a chelate may occur through a process called transmetallation when the chelate binds with another endogenous metal such as zinc or copper, allowing the release of free Gd3+. It is this free gadolinium that appears to be culpable in development of NSF.18 GBC chelates can be categorized based on their biochemical structure (linear vs. macrocyclic) and their charge (ionic vs. non‐ionic). Macrocyclic chelates bind Gd3+ more tightly than linear chelates and possess lower dissociation rates,19 which may have implications for possible toxicity.
The prolonged half‐life of GBC in the context of renal failure appears to predispose GBC to transmetallation and dissociation of Gd3+ from its chelate. Following intravenous injection, GBC is excreted unchanged by the kidneys via glomerular filtration. As a result, elimination half‐life, which is approximately 1.6 hours in normal individuals, is increased approximately 4‐ to 33‐fold in renal failure, depending on the level of GFR.16, 17, 20, 21 This increases the potential for Gd3+ dissociation through prolonged circulation times.
It has been postulated that once dissociated, deposition of the Gd3+ ion into skin and other organs sets off a cascade of poorly understood events that result in edema and fibrosis.18 Recent findings of gadolinium deposition in the skin of patients with NSF as well as an animal model of NSF following GBC exposure support this hypothesis.2225 It appears that vascular trauma, endothelial dysfunction or transudation (edema) allows the Gd3+ metal to enter the tissues. This may explain the preponderance of initial symptoms in dependent areas of the limbs.
What Can Be Done to Prevent NSF?
Avoid GBC Exposure in at Risk Patients
GBC agents are contraindicated in those with ESRD, CKD with estimated GFR 30 mL/minute/1.73 m2 (stages 4 and 5) and AKI. It has become common practice to use the 4‐variable Modification of Diet in Renal Disease (MDRD) formula in estimating GFR.26 Importantly, no estimating formula can be used in the context of a rising serum creatinine concentration as occurs with AKI. If a patient has AKI, one must assume a GFR 15 mL/minute until proven otherwise.
In those with low muscle mass the MDRD estimated GFR may overestimate the true GFR.27 Therefore, the Cockcroft‐Gault estimated creatinine clearance or a 24 hour urine‐based creatinine clearance may be useful in identifying at risk patients with underlying CKD.
Choose the Lowest Risk GBC Agent
When GBC use is deemed necessary in the high risk individual, an agent with a macrocyclic chelate (gadoteridol in the United States) is recommended.28 No published cases of NSF have been described with singular use of such agents. In addition, a retrospective study demonstrated no cases of NSF in ESRD patients on hemodialysis exposed to gadoteridol over a 7‐year period.29 This is not unexpected given the pharmacologic properties of this GBC agent.
Gadodiamide, a linear, non‐ionic agent, appears to produce the greatest risk of NSF as the largest number of NSF cases has been reported with this agent. By October 2007, 283 of 447 cases reported to the Food and Drug Administration (FDA) were exposed to gadodiamide.28 The significant preponderance with this agent is unlikely related to market share, reporting bias or publication bias. Gadopentetate, a linear, ionic agent, which had the greatest market share during this time, was responsible for approximately a quarter of cases reported to the FDA.28 Based on these data, gadodiamide and gadopentetate (and probably all linear agents) should be avoided in high risk patients.
Use Lower Doses of GBC
The FDA approved dose of all GBC agents, except the macrocyclic agent gadoteridol, is 0.1 mmol/kg.30 It appears that higher off‐label doses of GBC agents (0.3‐0.4 mmol/kg) which have been utilized for vascular studies (magnetic resonance angiography [MRA]), may have contributed to the emergence of NSF several years after these agents became available.
Develop a Protocol With Radiology and Nephrology Departments
Assessment of Renal Function Prior to Contrast Administration Is Required
Radiology departments should identify those with ESRD, CKD with estimated GFR 30 mL/minute/1.73 m2 (stages 4 and 5) and AKI. Using the 4‐variable MDRD formula in estimating GFR with the caveats previously noted, radiology departments will identify most at‐risk patients. Since the MDRD formula will be inaccurate in the setting of ESRD and AKI, these diagnoses should be determined through other means (for example, the patient's medical history) as part of the consent process.
Alternative Radiologic Imaging Modalities to GBC Enhanced Magnetic Resonance Imaging Should Be Utilized When Suitable in Those at High Risk
Newer techniques should be investigated as alternatives to GBC exposure. These include Magnetic Resonance Imaging (MRI) without GBC‐enhancement, where options such as 3D time‐of‐flight MRA, phase‐contrast angiography, and arterial spin labeling‐MR provide excellent information about blood vessels and blood flow.31 MRI with ultra‐small paramagnetic iron oxide particles may offer a future alternative in those that need a contrast‐based scan for diagnosis.32
However, since contrast enhanced MRI/MRA studies remain extremely important imaging modalities, their use may be required in some high risk individuals. In this circumstance, a macrocyclic chelate employed at the lowest dose possible, is recommended. The radiologist and nephrologist should be consulted in these instances.
Hemodialysis
Although hemodialysis efficiently clears GBC, its removal is not complete. Furthermore, it is not clear whether the damage has already occurred by the time a hemodialysis treatment can be instituted.33 It should be recognized that GBC removal after one treatment averages 65% to 73.8%; 3 to 4 sessions are required to remove 99% of the contrast agent.21, 34 Peritoneal dialysis on the other hand is an ineffective method of GBC removal (T1/2 of 52.7 hours).21 Because not all of the circulating Gd3+ is removed with a single hemodialysis treatment, prolonged tissue exposure occurs in these patients. This is reflected by the development of NSF in patients despite undergoing consecutive hemodialysis treatments following GBC exposure.3 Therefore, based on incomplete GBC removal with hemodialysis and the lack of evidence supporting prevention of NSF with this modality, we and others33, 35 strongly recommend avoidance of GBC in all patients with advanced kidney disease (GFR 30), regardless of the availability of hemodialysis. As such, the ability to perform hemodialysis after GBC in and of itself does not justify such exposure. However, if GBC use is deemed essential, then immediate hemodialysis should be strongly considered after exposure with further treatment on consecutive days.
Once NSF Develops, What Treatments Options are Available?
Unfortunately there is lack of a universally effective therapy for NSF. Several interventions have been described mainly in anecdotal case reports and very small case series. They have been recently reviewed (Table 2).360
| GBC Formulation | Year of Approval | Charge | Molecular Structure | Probable Risk of NSF* |
|---|---|---|---|---|
| Gadopentetate (Magnevist) | 1988 | Ionic | Linear | Medium |
| Gadoteridol (Prohance) | 1992 | Non‐ionic | Cyclic | Very low |
| Gadodiamide (Omniscan) | 1993 | Non‐ionic | Linear | High |
| Gadoversetamide (OptiMARK) | 1999 | Non‐ionic | Linear | Medium |
| Gadobenate (MultiHance) | 2004 | Ionic | Linear | Low |
|
| Therapies most likely to benefit |
| Kidney transplant (in ESRD) |
| Physical therapy |
| Pain control |
| Therapies with anecdotal success |
| Extracorporeal photopharesis |
| Sodium thiosulfate |
| Therapies with limited success |
| Drugs: Glucocorticoids, Pentoxifylline, Cyclophosphamide, Thalidomide |
| Immunomodulatory: Plasmapharesis, Intravenous immunoglobulin |
| Local: Intralesional IFN‐alpha, topical calcipotriene, other phototherapy |
Physical therapy is the mainstay of treatment for NSF. Physical therapy (and occupational therapy if needed) is essential to help prevent or slow the progression of joint contractures. Adequate pain relief, often with narcotics, is essential for patient comfort and to allow tolerance of physical therapy. Therapies with anecdotal benefit include extracorporeal photopheresis and infusions of sodium thiosulfate, a substance with chelating properties. Other interventions, such as immunosuppressive agents, topical agents and other phototherapies have shown limited success.
AKI resolution has been observed to result in regression of lesions.1, 3740 Presumably, resolution of the AKI allows for clearance of gadolinium and other profibrotic mediators, though definitive evidence of this is not available. Based on the observed response to AKI recovery, it is not surprising that improvement after kidney transplantation has also been described.1, 41 However, responses have not been consistent.39, 42
Consensus Guidelines and Recommendations
Nephrology societies have not yet developed consensus guidelines. Only the European Society of Urogenital Radiology has issued guidelines to date.43 These guidelines are consistent with expert opinions published elsewhere and are reflected in our approach regarding prevention of NSF (Table 3).
|
| 1. GBC agents are contraindicated in patients on dialysis regardless of availability of rapid treatment after exposure |
| 2. Avoid MRI with GBC in those with GFR 30 ml/min (estimated by MDRD formula) |
| MDRD formula may overestimate GFR in those of low weightconsider Cockcroft‐Gault calculation or 24 hour urine collection for creatinine clearance |
| MDRD is invalid in patient with a rising serum creatinine concentration. Assume GFR 30 in those with acutely rising serum creatinine concentration |
| 3. Consider alternative imaging studies or MRI studies without Gadolinium consult radiologist |
| 4. If GBC study is a necessity, then as low a dose as possible of a macrocyclic chelate would be recommended |
| 5. If an exposure to gadolinium occurs in ESRD, hemodialysis should be performed as soon as possible and repeated on consecutive days |
| 6. If an exposure to gadolinium occurs in CKD 4 or 5 or AKI patient (not on dialysis), an individualized approach should be undertaken when considering temporary catheter placement and initiation of hemodialysis |
The FDA has sent out several alerts since June 2006, the most recent in May 2007.30, 4446 In its Information for Healthcare Professionals alert, the FDA outlines recommendations. These are included in our final recommendations shown in Table 3.30 Those with a recent liver transplant, or those with chronic liver disease, who have associated kidney insufficiency of any severity, have also been identified by the FDA as an at risk group. This is based on reports of NSF occurring more commonly in patients with AKI who have these underlying conditions.47
Conclusions
With the high and increasing rates of AKI, CKD and ESRD seen among hospitalized patients,48 the need for vigilance when obtaining imaging with GBC agents becomes particularly important in the inpatient setting. As a preventable disease, it is incumbent upon us to fully understand the risk factors and potential pitfalls that may result in a patient exposed to these agents. The hospitalist has the unique role of acting as a firewall between the patient and the imaging study that may put him or her at risk for this devastating disorder.
Identification of GBC as a major culprit in the development of NSF and hence avoidance of this agent in those at the highest risk is expected to reduce the incidence of NSF. It is likely that the future will bring further understanding of the underlying mechanisms of gadolinium‐induced NSF and with this understanding, even safer strategies for GBC usage. However, until safer contrast agents become available, avoidance of GBC exposure in those with advanced acute or CKD remains our most important defense.
- .Gadolinium–a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?Nephrol Dial Transplant.2006;21(4):1104–1108.
- Nephrogenic fibrosing dermopathy associated with exposure to gadolinium‐containing contrast agents–St. Louis, Missouri, 2002–2006.MMWR Morb Mortal Wkly Rep.2007;56(7):137–141.
- ,,,,,.Gadodiamide‐associated nephrogenic systemic fibrosis: why radiologists should be concerned.AJR Am J Roentgenol.2007;188(2):586–592.
- ,,.Nephrogenic systemic fibrosis: a population study examining the relationship of disease development to gadolinium exposure.Clin J Am Soc Nephrol.2007;2(2):264–267.
- ,,,,.Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (omniscan).Invest Radiol.2007;42(2):139–145.
- ,,, et al.Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast‐enhanced magnetic resonance imaging.J Am Soc Nephrol.2006;17(9):2359–2362.
- ,,, et al.Nephrogenic systemic fibrosis: risk factors and incidence estimation.Radiology.2007;243(1):148–157.
- ,,.Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy).Curr Opin Rheumatol.2006;18(6):614–617.
- . Nephrogenic Fibrosing Dermopathy [NFD/NSF Website]. 2001–2007. Available at http://www.icnfdr.org. Accessed December 2009.
- ,,,,.Case‐control study of gadodiamide‐related nephrogenic systemic fibrosis.Nephrol Dial Transplant.2007;22(11):3174–3178.
- ,,, et al.Nephrogenic fibrosing dermopathy and high‐dose erythropoietin therapy.Ann Intern Med.2006;145(3):234–235.
- ,.Nephrogenic systemic fibrosis: an update.Curr Rheumatol Rep.2006;8(2):151–157.
- ,.Nephrogenic systemic fibrosis: early recognition and treatment.Semin Dial.2008;21(2):123–128.
- ,,.Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature.Am J Transplant.2007;7(10):2425–2432.
- .Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: a summary of the medical literature reporting.Eur J Radiol.2008;66(2):230–234.
- .MR contrast agents, the old and the new.Eur J Radiol.2006;60(3):314–323.
- ,,,,,.Magnetic resonance contrast agents: from the bench to the patient.Curr Pharm Des.2005;11(31):4079–4098.
- .Tissue deposition of gadolinium and development of NSF: a convergence of factors.Semin Dial.232008.
- .Safety of magnetic resonance contrast media.Top Magn Reson Imaging.2001;12(4):309–314.
- ,,, et al.Safety and pharmacokinetic profile of gadobenate dimeglumine in subjects with renal impairment.Invest Radiol.1999;34(7):443–448.
- ,,.Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis.Acad Radiol.1998;5(7):491–502.
- ,,.Gadolinium deposition in nephrogenic fibrosing dermopathy.J Am Acad Dermatol.2007;56(1):27–30.
- ,,,,.Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis.J Am Acad Dermatol.2007;56(1):21–26.
- ,,.Gadolinium is quantifiable within the tissue of patients with nephrogenic systemic fibrosis.J Am Acad Dermatol.2007;56(4):710–712.
- ,,,,,.A preclinical study to investigate the development of nephrogenic systemic fibrosis: a possible role for gadolinium‐based contrast media.Invest Radiol.2008;43(1):65–75.
- ,,,.A simplified equation to predict GFR from S‐creatinine [abstract].J Am Soc Nephrol.2000;11:155A.
- ,,,.Assessing kidney function–measured and estimated glomerular filtration rate.N Engl J Med.2006;354(23):2473–2483.
- ,.Nephrogenic systemic fibrosis risk: is there a difference between gadolinium‐based contrast agents?Semin Dial.2008;21(2):129–134.
- .Risk for nephrogenic systemic fibrosis with gadoteridol (ProHance) in patients who are on long‐term hemodialysis.Clin J Am Soc Nephrol.2008;3(3):747–751.
- US Food and Drug Administration: Information for Healthcare Professionals: Gadolinium‐Containing Contrast Agents for Magnetic Resonance Imaging (MRI) ProHance, and MultiHance). Available at: http://www.fda.gov/cder/drug/InfoSheets/HCP/gcca_200705HCP.pdf. Accessed December 2009.
- ,.Nephrogenic systemic fibrosis: non‐gadolinium options for the imaging of CKD/ESRD patients.Semin Dial.2008;21(2):160–165.
- ,,, et al.Ultrasmall superparamagnetic iron oxides (USPIOs): a future alternative magnetic resonance (MR) contrast agent for patients at risk for nephrogenic systemic fibrosis (NSF)?Kidney Int.2008;75(5):465–474.
- .Dialytic therapies to prevent NSF following gadolinium exposure in high‐risk patients.Semin Dial.2008;21(2):145–149.
- ,,,,.Dialyzability of gadodiamide in hemodialysis patients.Radiat Med.2006;24(6):445–451.
- ,,,,,.Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI.Cleve Clin J Med.2008;75(2):95–97, 103–104, 106 passim.
- ,,.Treatment of nephrogenic systemic fibrosis: limited options but hope for the future.Semin Dial.2008;21(2):155–159.
- ,,,,.Nephrogenic fibrosing dermopathy.Am J Dermatopathol.2001;23(5):383–393.
- ,,,,.Nephrogenic fibrosing dermopathy: a novel cutaneous fibrosing disorder in patients with renal failure.Am J Med.2003;114(7):563–572.
- ,,,.Nephrogenic systemic fibrosis: relationship to gadolinium and response to photopheresis.Arch Dermatol.2007;143(8):1025–1030.
- ,,,.A case of nephrogenic fibrosing dermopathy.Ann Acad Med Singapore.2004;33(4):527–529.
- ,,,,,.Nephrogenic fibrosing dermopathy: two pediatric cases.J Pediatr.2003;143(5):678–681.
- ,,.Nephrogenic fibrosing dermopathy in children.Pediatr Nephrol.2006;21(9):1307–1311.
- .ESUR guideline: gadolinium‐based contrast media and nephrogenic systemic fibrosis.Eur Radiol.2007;17(10):2692–2696.
- US Food and Drug Administration: FDA News: FDA Requests Boxed Warning for Contrast Agents Used to Improve MRI Images. Available at: http://www.fda.gov/bbs/topics/NEWS/2007/NEW01638.html. Accessed December 2009.
- US Food and Drug Administration: Public Health Advisory: Gadolinium‐containing Contrast Agents for Magnetic Resonance Imaging (MRI): Omniscan, OptiMARK, Magnevist, ProHance, and MultiHance. Available at: http://www.fda.gov/cder/drug/advisory/gadolinium_agents.htm. Accessed December 2009.
- US Food and Drug Administration: Public Health Advisory: Update on Magnetic Resonance Imaging (MRI) Contrast Agents Containing Gadolinium and Nephrogenic Fibrosing Dermopathy. Available at: http://www.fda.gov/cder/drug/advisory/gadolinium_agents_20061222.htm. Accessed December 2009.
- ,,, et al.Nephrogenic systemic fibrosis among liver transplant recipients: a single institution experience and topic update.Am J Transplant.2006;6(9):2212–2217.
- Hospitalization discharge diagnoses for kidney disease–United States, 1980–2005.MMWR Morb Mortal Wkly Rep. 282008;57(12):309–312.
- ,,,.Response to the FDA's May 23, 2007, nephrogenic systemic fibrosis update.Radiology.2008;246(1):11–14.
- .How should nephrologists approach gadolinium‐based contrast imaging in patients with kidney disease?Clin J Am Soc Nephrol.2008;3(3):649–651.
- .Gadolinium–a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?Nephrol Dial Transplant.2006;21(4):1104–1108.
- Nephrogenic fibrosing dermopathy associated with exposure to gadolinium‐containing contrast agents–St. Louis, Missouri, 2002–2006.MMWR Morb Mortal Wkly Rep.2007;56(7):137–141.
- ,,,,,.Gadodiamide‐associated nephrogenic systemic fibrosis: why radiologists should be concerned.AJR Am J Roentgenol.2007;188(2):586–592.
- ,,.Nephrogenic systemic fibrosis: a population study examining the relationship of disease development to gadolinium exposure.Clin J Am Soc Nephrol.2007;2(2):264–267.
- ,,,,.Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (omniscan).Invest Radiol.2007;42(2):139–145.
- ,,, et al.Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast‐enhanced magnetic resonance imaging.J Am Soc Nephrol.2006;17(9):2359–2362.
- ,,, et al.Nephrogenic systemic fibrosis: risk factors and incidence estimation.Radiology.2007;243(1):148–157.
- ,,.Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy).Curr Opin Rheumatol.2006;18(6):614–617.
- . Nephrogenic Fibrosing Dermopathy [NFD/NSF Website]. 2001–2007. Available at http://www.icnfdr.org. Accessed December 2009.
- ,,,,.Case‐control study of gadodiamide‐related nephrogenic systemic fibrosis.Nephrol Dial Transplant.2007;22(11):3174–3178.
- ,,, et al.Nephrogenic fibrosing dermopathy and high‐dose erythropoietin therapy.Ann Intern Med.2006;145(3):234–235.
- ,.Nephrogenic systemic fibrosis: an update.Curr Rheumatol Rep.2006;8(2):151–157.
- ,.Nephrogenic systemic fibrosis: early recognition and treatment.Semin Dial.2008;21(2):123–128.
- ,,.Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature.Am J Transplant.2007;7(10):2425–2432.
- .Nephrogenic systemic fibrosis associated with gadolinium based contrast agents: a summary of the medical literature reporting.Eur J Radiol.2008;66(2):230–234.
- .MR contrast agents, the old and the new.Eur J Radiol.2006;60(3):314–323.
- ,,,,,.Magnetic resonance contrast agents: from the bench to the patient.Curr Pharm Des.2005;11(31):4079–4098.
- .Tissue deposition of gadolinium and development of NSF: a convergence of factors.Semin Dial.232008.
- .Safety of magnetic resonance contrast media.Top Magn Reson Imaging.2001;12(4):309–314.
- ,,, et al.Safety and pharmacokinetic profile of gadobenate dimeglumine in subjects with renal impairment.Invest Radiol.1999;34(7):443–448.
- ,,.Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis.Acad Radiol.1998;5(7):491–502.
- ,,.Gadolinium deposition in nephrogenic fibrosing dermopathy.J Am Acad Dermatol.2007;56(1):27–30.
- ,,,,.Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis.J Am Acad Dermatol.2007;56(1):21–26.
- ,,.Gadolinium is quantifiable within the tissue of patients with nephrogenic systemic fibrosis.J Am Acad Dermatol.2007;56(4):710–712.
- ,,,,,.A preclinical study to investigate the development of nephrogenic systemic fibrosis: a possible role for gadolinium‐based contrast media.Invest Radiol.2008;43(1):65–75.
- ,,,.A simplified equation to predict GFR from S‐creatinine [abstract].J Am Soc Nephrol.2000;11:155A.
- ,,,.Assessing kidney function–measured and estimated glomerular filtration rate.N Engl J Med.2006;354(23):2473–2483.
- ,.Nephrogenic systemic fibrosis risk: is there a difference between gadolinium‐based contrast agents?Semin Dial.2008;21(2):129–134.
- .Risk for nephrogenic systemic fibrosis with gadoteridol (ProHance) in patients who are on long‐term hemodialysis.Clin J Am Soc Nephrol.2008;3(3):747–751.
- US Food and Drug Administration: Information for Healthcare Professionals: Gadolinium‐Containing Contrast Agents for Magnetic Resonance Imaging (MRI) ProHance, and MultiHance). Available at: http://www.fda.gov/cder/drug/InfoSheets/HCP/gcca_200705HCP.pdf. Accessed December 2009.
- ,.Nephrogenic systemic fibrosis: non‐gadolinium options for the imaging of CKD/ESRD patients.Semin Dial.2008;21(2):160–165.
- ,,, et al.Ultrasmall superparamagnetic iron oxides (USPIOs): a future alternative magnetic resonance (MR) contrast agent for patients at risk for nephrogenic systemic fibrosis (NSF)?Kidney Int.2008;75(5):465–474.
- .Dialytic therapies to prevent NSF following gadolinium exposure in high‐risk patients.Semin Dial.2008;21(2):145–149.
- ,,,,.Dialyzability of gadodiamide in hemodialysis patients.Radiat Med.2006;24(6):445–451.
- ,,,,,.Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI.Cleve Clin J Med.2008;75(2):95–97, 103–104, 106 passim.
- ,,.Treatment of nephrogenic systemic fibrosis: limited options but hope for the future.Semin Dial.2008;21(2):155–159.
- ,,,,.Nephrogenic fibrosing dermopathy.Am J Dermatopathol.2001;23(5):383–393.
- ,,,,.Nephrogenic fibrosing dermopathy: a novel cutaneous fibrosing disorder in patients with renal failure.Am J Med.2003;114(7):563–572.
- ,,,.Nephrogenic systemic fibrosis: relationship to gadolinium and response to photopheresis.Arch Dermatol.2007;143(8):1025–1030.
- ,,,.A case of nephrogenic fibrosing dermopathy.Ann Acad Med Singapore.2004;33(4):527–529.
- ,,,,,.Nephrogenic fibrosing dermopathy: two pediatric cases.J Pediatr.2003;143(5):678–681.
- ,,.Nephrogenic fibrosing dermopathy in children.Pediatr Nephrol.2006;21(9):1307–1311.
- .ESUR guideline: gadolinium‐based contrast media and nephrogenic systemic fibrosis.Eur Radiol.2007;17(10):2692–2696.
- US Food and Drug Administration: FDA News: FDA Requests Boxed Warning for Contrast Agents Used to Improve MRI Images. Available at: http://www.fda.gov/bbs/topics/NEWS/2007/NEW01638.html. Accessed December 2009.
- US Food and Drug Administration: Public Health Advisory: Gadolinium‐containing Contrast Agents for Magnetic Resonance Imaging (MRI): Omniscan, OptiMARK, Magnevist, ProHance, and MultiHance. Available at: http://www.fda.gov/cder/drug/advisory/gadolinium_agents.htm. Accessed December 2009.
- US Food and Drug Administration: Public Health Advisory: Update on Magnetic Resonance Imaging (MRI) Contrast Agents Containing Gadolinium and Nephrogenic Fibrosing Dermopathy. Available at: http://www.fda.gov/cder/drug/advisory/gadolinium_agents_20061222.htm. Accessed December 2009.
- ,,, et al.Nephrogenic systemic fibrosis among liver transplant recipients: a single institution experience and topic update.Am J Transplant.2006;6(9):2212–2217.
- Hospitalization discharge diagnoses for kidney disease–United States, 1980–2005.MMWR Morb Mortal Wkly Rep. 282008;57(12):309–312.
- ,,,.Response to the FDA's May 23, 2007, nephrogenic systemic fibrosis update.Radiology.2008;246(1):11–14.
- .How should nephrologists approach gadolinium‐based contrast imaging in patients with kidney disease?Clin J Am Soc Nephrol.2008;3(3):649–651.