User login
Hospital Approval of Human Research
Hospitals have important legal and ethical responsibilities for human participant research conducted within their facilities, such as ensuring that research complies with federal regulations and presents minimal risks to patients. Many hospitals accept as sufficient the federal requirement that human participant research studies have Institutional Review Board (IRB) review and approval. IRBs must review proposed research according to numerous criteria, such as scientific soundness, alignment with accepted ethics principles and weighing of benefit vs. risk to study participants.1, 2 The legally required aspects of IRB review do not, however, include considering practical matters in implementing and operating an interventional clinical trial in the complex environment of the modern acute care hospital.
Our hospital system established a broad policy requiring internal review and formal approval of any human participant research conducted within any of its hospitals, including studies that enroll hospital patients or hospital employees, utilize hospital medical records, or request hospital‐provided services for research tests or procedures. The purpose of this paper is to describe this formal hospital system review and approval process, the reasons for implementing it, and the types of issues considered prior to our hospital system granting a principal investigator permission to conduct a study.
Background
Surprisingly little healthcare or medical literature exists regarding hospital responsibilities toward human subject research conducted on its premises. Much of the literature focuses on ethics issues, the nature of informed consent, and study design. As critical as these discussions are, they seldom address the numerous complex operational issues and challenges that implementation of a clinical trial can create in a hospital setting.
Flanders et al.3 make the case for hospitalists and specialists to work together to support research that includes inpatients as study participants. Moore and Goldberg4 discuss the ingredients of successfully developing a research program in a community hospital and mention the need to involve all affected hospital departments in the initial hospital review of a study, evaluating study impact on hospital workflow, and establishing processes related to budgeting and billing. Jamerson5 also makes the case for hospital departmental review and involvement, assessment of the ability to integrate study activities into the hospital structure, assessment of the resources needed to support the research, determination of whether the hospital will contribute financially to the research, and explicit decision making regarding the assumption of institutional risk.
Despite the recognition that US patients increasingly live with multiple chronic conditions6 and that clinical trial protocols have become more procedure and resource intensive and costly,79 there has not been a corollary recognition of the increasing need for hospitals to understand and manage research activities occurring within their facilities.
Our organization is a hospital system with 9 acute care hospitals, including an academic teaching hospital (affiliated with a university medical school) with a Level 1 Trauma Center, 1 specialty rehabilitation hospital, numerous specialized clinics, and a LifeFlight Program with a 6‐helicopter fleet (Geisinger, Danville, PA). This system of hospitals serves the fourth largest metropolitan community in the US (Houston and Harris County in southeastern TX), with a population of nearly four million and a geographic spread of 1778 square miles.10, 11 The hospital system has approximately 140,700 inpatient admissions per year and 586,000 outpatient visits.
Eight years ago, our hospital system adopted a corporate policy requiring that any activity associated with human participant research receive prior hospital system review and approval. Our organization considers this review process vital to: (1) maintaining our commitment to our Federalwide Assurance with the Office for Human Research Protections, (2) abiding by the Joint Commission requirements related to research,12 (3) protecting the safety and confidentiality of patients and employees who are potential or actual research participants, (4) protecting the confidentiality of participants' medical information, (5) assuring that legal fundamentals and good clinical practice (GCH)13 are a part of study plans, (6) assuring that studies are operationally feasible, and (7) evaluating and minimizing risks to patients and risks to the organization.
Review and Approval Process
Overview
An investigator triggers a formal hospital system review by submitting study documents to 1 of the IRBs listed on the system's Federalwide Assurance through the electronic IRB system and completing the required hospital system's Research Application Form. The hospital system review occurs in parallel with the IRB review, not duplicating it but rather focusing separately on patient safety, operational and financial issues, and hospital risk issues.
Our Clinical Innovation and Research Institute manages the hospital system review. Upon electronic notification of a new study submission, an Institute Clinical Research Associate examines all study‐related documents, including the completed Research Application Form and other submitted documents, such as the study protocol, the investigational product's Investigator's Brochure, consent forms, Food and Drug Administration (FDA) letters, survey questions, and diary and other data collection forms. The Associate may spend considerable time communicating with the investigator's research team, collecting missing information and building a complete study file, including identifying the affected hospitals and hospital departments. The Institute then provides study documents to the individuals responsible for hospital‐level research review, and each affected hospital conducts its own internal review and approval process.
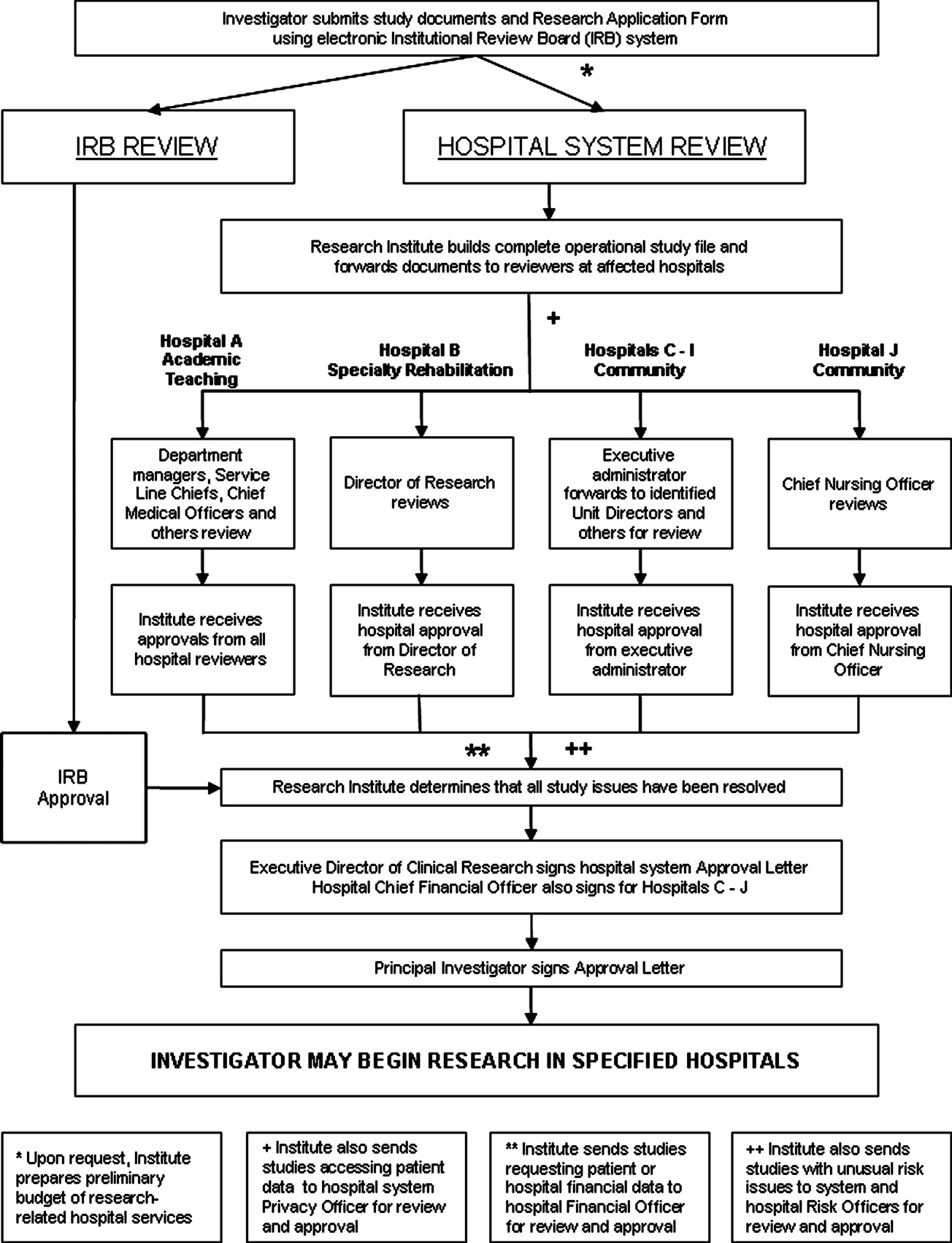
The hospital‐level review process varies depending on the hospitals involved. The academic teaching hospital has the most detailed review process, due to the complexities and risks associated with the full spectrum of human participant research which occurs there (Hospital A in Figure 1). If a study affects this hospital, the Institute provides study documents to each affected Department Manager, the affected Service Line Chief, the Chief Medical Officer or Medical Director of each Intensive Care Unit within which the study will recruit and enroll patients, and the Infection Control Officer and Radiation Safety Officer, as appropriate.
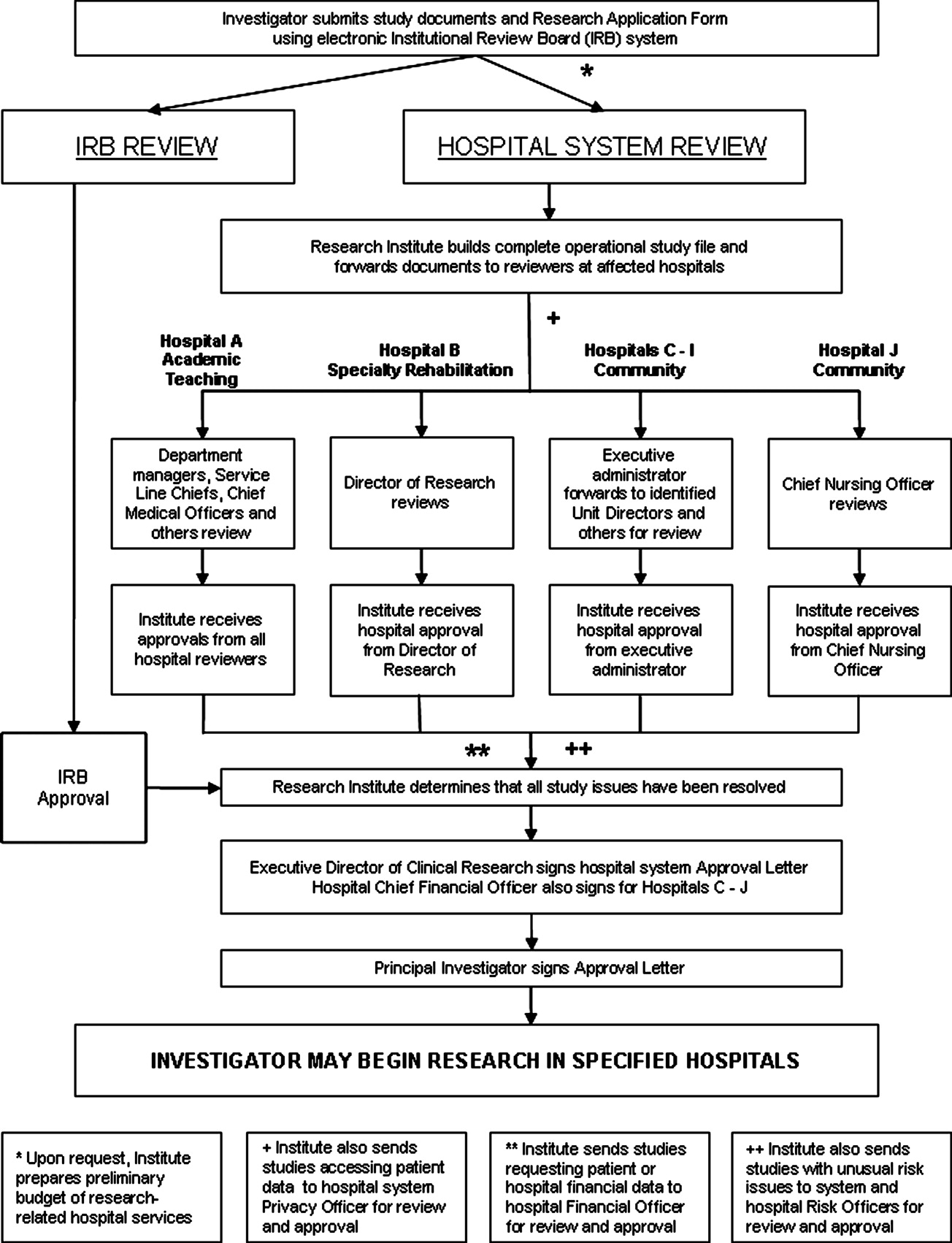
The specialty rehabilitation hospital has a long‐standing national reputation for its research programs; its Director of Research knows each investigator, reviews each study, and provides that hospital's administrative review (Hospital B). Seven of the system's community hospitals have either a Chief Executive Officer, Chief Nursing Officer, or Chief Financial Officer serve as the hospital's executive administrator responsible for research review, and this person distributes the study documents to the Chief Medical Officer, if deemed necessary, and to each affected department (Hospitals C‐I). One of the smaller community hospitals participates in relatively few studies; the Chief Nursing Officer reviews and provides hospital‐level approval (Hospital J). For retrospective studies requesting a clinical data set, the Institute provides the study documents to the Director of the Information Systems Department. All studies accessing patient data are provided to the system Privacy Officer for review and approval.
Studies may involve 1 or more hospitals; some have involved as many as 7 at once.
All reviewers also receive a standardized Research Study Evaluation Form for their written comments and recommendations (Approve, Disapprove, or Defer) regarding whether the hospital system should approve the study.
If a study requests hospital‐provided research services, the Institute's Research Financial Coordinator develops a research budget listing the hospital charges that the researcher will incur for these tests and procedures.
Once reviewers return completed Evaluation Forms, the Institute Clinical Research Associate makes an initial determination whether the review process has satisfactorily answered all questions and resolved all outstanding issues. The Manager of Clinical Research Operations then examines the study file to ensure a satisfactory review. Finally, the system Executive Director for Clinical Research provides a Letter of Approval to the Principal Investigator, which serves as the agreement of terms for conducting the study within the hospital system. The letter contains standard stipulations, such as requiring the Principal Investigator to abide by federal law and International Conference on Harmonization (ICH) GCPs and the budget for the hospital‐provided research tests and procedures. Additionally, it includes any stipulations unique to the studyfor example, that the Principal Investigator will provide training to hospital personnel who will be operating nonhospital equipment. The Institute provides affected hospital departments with copies of the approval letter. Upon signing and returning the letter, the Principal Investigator may begin the study in the designated hospitals.
Some details about the hospital system review are discussed in sections below.
Patient Safety and Human Participant Protections
Participant Recruitment Plans
Sometimes IRB submission documents do not adequately describe how researchers will identify potential study participants and approach them for consent. Key concerns which we address include how researchers will identify potential participants in a Health Insurance Portability and Accountability Act (HIPAA)‐compliant manner, the level and type of illnesses of the patients whom the investigator intends to recruit, whether the researcher must obtain the admitting or attending physician's permission, the qualifications of the person making the initial patient contact, and how and when that person will make contact, with special attention required if subjects may include very ill patients in an Intensive Care Unit (ICU).
In‐service for Unit Personnel and Pharmacists
For most interventional studies, we require that the principal investigator's research team provide a plan for study education in‐service for nurses, technicians, respiratory therapists, and pharmacists who may be involved in the care of a patient enrolled in the study. This is usually an in‐person presentation done at a regular unit meeting, with an additional investigational drug‐specific in‐service provided and available on the hospital system intranet for the pharmacists. We remind researchers that the plan must also include an in‐service for night shift personnel, who are often otherwise overlooked.
Personnel Administering the Investigational Product
Sometimes submitted protocols do not state who will administer the investigational productthe physician investigator, other members of the research team or unit nurses. If the investigator expects unit nurses to administer the product, the hospital needs to determine whether the nurses' experience and training qualifies them to administer it, assess for adverse events, and provide care for patients with these events. If the nurses are not qualified, then the hospital needs to decide whether the nurses should receive training or a member of the research team should administer the product. Some research studies involve investigational agents with novel administration techniques or risks of immediate severe adverse events, requiring the presence of a physician knowledgeable about the investigational product.
Care of Study Participants With Adverse Events
Studies with unusual investigational agents can also raise a Unit Director's or ICU Medical Director's concern as to whether bedside nurses can appropriately and adequately discern and respond to potential adverse events. If the investigational agent might result in an event not normally anticipated in patients in that particular unit, the hospital may need to consider additional preparation or staffing.
Consent Documentation
Federal law and ICH GCPs require that principal investigators have signed consent forms available in the research records, which may be off of the hospital premises in physician clinics or office areas. Our hospital system requires a copy of the consent form in the patient's medical chart if the research team conducted the consent process within the hospital or if the study participant will be an inpatient for a procedure included as part of the study protocol, whether the patient was recruited while an inpatient or prior to inpatient admission. This is important for meeting the Joint Commission's standards related to research. We established an internal monitoring program to verify that researchers were providing copies of consent, assent and parental permission forms to the Health Information Management Office for placement in medical charts.
IRB‐related Issues
Occasionally a hospital system review identifies an IRB‐related concern, such as a known possible adverse event missing from the consent form, unexplained medical terms in the consent form, exclusion criteria not mentioning pregnancy or a consent form not covering a pregnancy test even though the protocol text mentions these, or missing Investigational New Drug Application (IND) or Investigational Device Exemption (IDE) information. Institute staff route such concerns to the IRB for follow‐up with the researcher as necessary. If a hospital system review identifies an ethical concern, the Institute consults with the IRB Chair or ethicist member. Usually the Chair will assess the concern, raise options for addressing it, and recommend a course of action.
Operational Issues
Feasibility and Implementation
We urge researchers to meet with Institute staff to discuss implementation of a protocol as a real‐world, operationalized study and also encourage them to meet with managers of the primary units where the study will take place. Researchers, however, may develop and submit industry‐sponsored clinical trials or investigator‐initiated studies without such prior discussions and may not have adequately considered operational feasibility.
Given the increasing complexity of investigational agents, study designs, study procedures, and patient safety monitoring, hospital reviewers need to consider exactly who will perform study procedures and processes and how those people will do so. If a blood or tissue sample needs to be spun, packaged, and mailed on dry ice within a limited timeframe, for example, who will do this and will the supplies and equipment be available as needed? A study protocol can lead to a change in normal unit processes. Operating suite managers, for instance, may need to adjust schedules or work with research teams if research activities may prolong a procedure beyond average timeframes.
Other potential impacts abound. Research teams sometimes assume, without checking with hospital managers, that hospital staff, usually nurses or respiratory therapists, will perform research procedures of the kind they usually perform as standard clinical procedure. Most commonly, researchers assume that bedside nurses will perform frequent blood draws necessary for a pharmacokinetic substudy. Unit managers, however, may not agree to commit nurses' time to this task, depending on the number and timing of the draws. During surgery, a study may require recording of events or timelines which are not usually recorded. Researchers sometimes assume that operating room personnel will be able to focus on this data collection. Hospital managers and directors are often concerned that researchers assume that unit staff will transport patients within‐hospital for research procedures, which can involve repeatedly moving a patient from their home unit to Radiology or elsewhere and then back to the home unit. For a large hospital, this can involve considerable staff time spent away from the home unit, which may affect unit operations. Occasionally, an investigator requests that a hospital temporarily or permanently store blood or tissue samples. Since hospitals are not necessarily prepared to store large numbers of samples for extended periods, we address each such request on a case‐by‐case basis.
Investigational Product
Hospital reviewers need to consider an assortment of challenges relating to the receipt, storage, dispensing, and accountability recording of investigational products. If a hospital pharmacy will be dispensing an investigational drug, then pharmacists need to anticipate its arrival from the sponsor and know storage and other sponsor, study and FDA requirements. Pharmacists also need to know if they are expected to prepare placebo pills. If a drug is an agent that an external pharmacy needs to prepare and compound, the chain of custody documentation of the drug as it moves from manufacturer to external pharmacy to hospital pharmacy needs to be clear.
While academic hospitals usually have research‐knowledgeable pharmacists, community‐based hospital pharmacists may not be familiar with the special requirements of the FDA or sponsor, such as securing investigational products separately from marketed products, recording batch numbers, maintaining accountability logs, and following procedures for return or destruction of remaining product upon study closure. In our hospital system, 2 Research Pharmacists in the Investigational Drug Pharmacy at the academic teaching hospital serve as expert advisers to pharmacists of the community hospitals.
If a research study involves use of an investigational device, hospital reviewers need to consider what the device is, how it should be secured if stored on the unit, and how to document storage and accountability.
Biologics and radioactive materials can present unique challenges. For instance, our system requires researchers to provide hospitals with chain of custody documentation, similar to that used for organ transplants, when patient biologicals leave the hospital (for instance, for processing at a nearby accredited Cell Processing Facility) and returned to the hospital for infusion back into the patient, to confirm that the right product was returned to the hospital and infused into the right patient.
Unaffiliated Principal Investigators and Other Personnel
Increasingly, researchers who are not affiliated with our hospital system have inquired about conducting studies in 1 or more of our hospitals. We have been quite surprised by the number of inquiries from researchers or sponsors who presume we will grant immediate permission for them to access our patient lists for recruitment purposes, allow unidentified research team members to enter our hospitals, approve team members to conduct active recruitment of our hospital patients, and grant team members access to patient data.
Additionally, as clinical and translational research projects become increasingly multidisciplinary and involve cross‐organizational collaborations, many research teams include unaffiliated personnel from other organizations, such as faculty at local or distant universities, employees of a site management organization, and employees of the city or county health department.
We do not permit clinicians who are not clinically credentialed at our hospitals to engage in interventional research within our hospital system. For studies that include any intervention that qualifies as a clinical procedure, the Principal Investigator must become clinically credentialed by the hospital or an already credentialed clinician must become the local Principal Investigator for the study, and all team members who perform clinical procedures must also become clinically credentialed.
An unusual situation occurred when researchers from a university not formally affiliated with our hospital system sought to transfer a study to 1 of our hospitals following Hurricane Ike. For 15 years, the research team had performed needle muscle biopsies, for which they had received training and credentialing at their home institution. Our hospital's Chief Medical Officer, however, did not feel comfortable accepting the credentialing performed at another institution and required the external researchers to apply for privileges through our hospital's credentialing process. The original credentialing documentation at their home institution was unavailable, in any case, due to flooding and building closures, so the researchers worked through the night to complete our applications. The hospital credentialing committee came to order on short notice and completed the credentialing process in record time, allowing the research team to see the study participants at our hospital with only a few missed visits and few study deviations for the Principal Investigator.
For research team members who will be performing no clinical procedures, our hospital system has a research credentialing process, discussed below.
Financial Issues
Study Finances
Researchers are typically aware of charges for the procedures that their studies most frequently require, such as labs, radiology, and research pharmacy, although they often do not inquire about the hospital's current charges, so their information may be somewhat dated when they negotiate with sponsors or submit grant applications.
When researchers plan on utilizing hospital staff to perform research‐required tasks, such as blood draws or patient transport for tests or procedures, however, they rarely include financial support to the hospital in their study budgets, leaving these tasks unfunded, which can be a problem for hospital reviewers who must approve the research plan.
In our review and approval process, we seek clarification as to payment for research products. Sponsors are not permitted by law to seek payment for investigational drugs, but when a research protocol uses a standard‐of‐care drug for strictly research‐related purposes, determining whether the sponsor, hospital, or study participant incurs financial responsibility becomes an issue requiring careful consideration. We ensure that consent forms explicitly state any special charges to the study participant. An investigational device that falls into an FDA classification that permits charges can be especially problematic. Often the devices are expensive; the hospital must purchase them in batches and pay immediately. If the researcher does not use all the devices, the unused ones sometimes cannot be returned. If investigational devices are more costly than standard‐of‐care devices, then the hospital could incur substantial losses in billings, since hospital charges are diagnosis and procedure code dependent and usually not adjusted for device cost.
Our standardized financial assessment has led to more beneficial arrangements with sponsors for the return and reimbursement of unused products and more informed hospital decisions as to whether to conduct a specific medical device clinical trial.
We also carefully review consent form language to clarify who incurs costs for research‐related adverse events and research injuries. For investigational products with extensive potential side effects or studies enrolling very ill patients, the costs associated with adverse events can be extremely high.
Investigators are affiliated with our hospitals, not employed by them, so our hospital system does not have budget agreements with funding organizations, but relies on these external entities to fund the studies adequately. Some investigator‐initiated studies may be funded by the investigator's organization, such as a private practice or university, but in our experience such funds are sometimes depleted before study completion.
Hospital Financial Information as Study Data
As a part of the study protocol, researchers occasionally request patient‐related cost data pertinent to specific procedures or to the treatment of certain medical conditions, hospital charges and payments received, or other financial data. We forward these research requests to a hospital Finance Chief to determine the appropriateness of releasing the requested data and, if approved, how the hospital will extract and present the data sets to the investigator.
Hospital Risk Issues
Research Equipment
Our academic teaching hospital and the university medical school affiliated with it are distinct organizations, so a particular risk issue arises for the hospital when a researcher wishes to transport and use nonhospital equipment on the hospital premises. Our hospital system has responsibility to ensure appropriate and safe operation of equipment. Consequently, our hospital system review identifies any proposed use of externally‐owned equipment and involves the system and hospital Risk Officers in assessing such use for risk to patients. Specifics addressed include ownership of equipment, whether the research team or hospital staff will operate it; whether the operators have received or will receive training, and the potential risks to patients of equipment malfunctions. Upon determination that individuals will operate the equipment appropriately with minimal risks to study participants, the hospital's Biomedical Department performs their standard safety check prior to the equipment's use in the hospital.
Clinical Data Generated by NonHospital Equipment
Bringing external equipment into a hospital has a rarely anticipated consequence: the generation and storage of patient data. Our hospital system review determines whether these data constitute source data per FDA and ICH GCP definitions, whether the data are clinically pertinent, and whether the data need to go into the patient's hospital medical record. For example, if a university faculty researcher brings a vital signs monitor into the hospital to collect and electronically record data from hospital patients enrolled in a study, we may require that the research team print the data for insertion into the patient's hospital medical record.
Surveys of Hospital Employees
A research study that seeks to survey or interview hospital system employees raises a different type of institutional concern. In such a case, system and hospital Human Resources Department (HR) personnel review the planned study, paying close attention to how the research team will recruit employees and what type of information the team will request from them. HR does not want employees feeling that they must participate in a study simply because it takes place in their facility and wants to protect the identity of employees participating in anonymous surveys. On occasion, Institute staff distribute surveys (and sometimes collect them when completed), providing an identity firewall between the employee who elects to participate and the researcher.
HR may also limit researchers from asking especially personal questions or questions inquiring about the recruitment, hiring, and retention practices of their employer. HR must consider whether the study design and survey questions raise liability concerns. Studies that include employee focus groups or one‐on‐one interviews raise issues pertaining to the purpose and content of the focus groups or interviews, mechanisms to address special issues or complaints that may arise during them, and determination of whether the employees may participate during paid time as opposed to participating while off‐the‐clock.
Nonclinical Research Credentialing Process
It is vital that hospitals know who will be on hospital premises for a research study and what activities they will be engaging in. If a research team member is not already affiliated with our hospital system and will be engaging in noninterventional research activities, such as conducting the research consent process, administering a survey, or providing educational materials, our hospital system review initiates a nonclinical research credentialing process. The Institute's Manager of Clinical Research Operations assesses the team members' qualifications, reviews their resumes, interviews each of them, and discusses exactly what activities they will be engaging in and their training and experience. In addition to evaluating their qualifications, the hospital must determine how such individuals will be identified once inside the hospital, including what type of badge (employee, contractor, or visitor) the hospital will provide them. Upon successful completion of the research credentialing process, the Institute explicitly names the approved individuals in the Letter of Approval to the Principal Investigator, with copies to the affected hospitals' units.
Discussion
Our hospital research review and approval process is critical to ensuring that only safe and regulatory‐compliant research activities occur within our hospital system, but the review and approval process, with its many steps and numerous reviewers, can be cumbersome. There is no substitute for human beings reading and understanding the protocol, consent forms for patient involvement in a study, the proposed use of protected health information, Investigator's Brochure, Research Application Form, and other study documents and then identifying pertinent issues and resolving them, and this process does require significant staff time.
We have improved (continuously) the Research Application Form to help in the crucial initial gathering of information about studies' operational needs. We have also converted from a predominantly paper‐based review process to the widespread use of electronic documents, but we have not automated the distribution process for these electronic documents and a staff person must still do this through email.
Despite our efforts to improve the review process, investigators are sometimes frustrated with it, particularly if someone identifies a new issue late in the process, or if the hospital system's approval for the study lags behind the IRB approval by more than a few days.
Currently, the hospital system provides the majority of study approvals to the Principal Investigator within 2 weeks of IRB approval, with some approvals provided within 1 day of IRB approval and others as long as 3 months afterward. Delays in hospital approval can be due to a study lacking required approval from a Department of Defense IRB, the FDA not providing permission to proceed with a study, the absence of an executed contract with a vendor to pick up and dispose of radioactive waste from the investigational product and many other factors. Of course, when the research team can respond in timely fashion to inquiries or issues that we have raised, that assists all of us in completing the review and approval process as quickly as possible.
The review and approval process benefits hospital patients, hospital personnel who will be supporting studies, and hospitals as institutions. Thinking through, planning, and preparing for study operations, particularly for studies taking place in an ICU, benefits the research team, hospital personnel, and the patients. Overall, the hospital system's research review and approval process affords many protections to our patients and reduces risks to the hospital system while permitting research studies to be conducted within its varied healthcare facilities.
We encourage researchers not to view today's modern hospital as bricks and mortar, but as an institution with deep responsibility for safety on hospital premises. Hospitals must meet over a thousand Joint Commission standards, set goals for patient outcomes, measure and report on quality indicators, protect patient confidentiality, maintain the safety an ever‐expanding array of simple and complex equipment, maintain, check and document contents of adult and pediatric crash carts, have 24‐7 code teams at‐the‐ready, create, maintain and store patient medical records securely, transport patients, and much more.
Conclusion
Our hospital system, in accordance with a system‐wide policy, engages in a comprehensive review and approval process for any human participant research that has been proposed to be conducted within one or more of our facilities. The process focuses primarily on patient safety within the hospital premises, operational study issues, financial issues and hospital risk issues. This process decreases risks to the patients, researchers, and hospital facilities engaging in human participant research.
- Code of Federal Regulations Part 45, Title 46.
- Code of Federal Regulations Part 50, Title 21.
- ,,,.The University of Michigan Specialist‐Hospitalist Allied Research Program: jumpstarting hospital medicine research.J Hosp Med.2008;3:308–313.
- ,.Successfully developing a cardiovascular research program in a community hospital.J Cardiovasc Manag.2004;15:13–19.
- .Developing an infrastructure for research in a free‐standing hospital.J Nurs Adm.2007;37:295–301.
- ,,.Prevalence of multiple chronic conditions in the United States' Medicare population.Health Qual Life Outcomes.2009;7:82.
- ,,,,.Assessing the impact of protocol design changes on clinical trial performance.Am J Ther.2008;15:450–457.
- ,,, et al.Factors affecting workload of cancer clinical trials: results of a multicenter study of the National Cancer Institute of Canada Clinical Trials Group.J Clin Oncol.2002;20:545–556.
- ,,, et al.The changing face of Phase 1 Cancer Clinical Trials: new challenges in study requirements.Cancer.2009;115(8):1592–1597.
- U.S. Census Bureau. Annual Estimates of the Resident Population for Counties of Texas: April 1, 2000 to July 1, 2008. Available at: http://www.census.gov/popest/counties/tables/CO‐EST2008–01‐48.xls. Accessed February 2010.
- Harris County Texas. Available at: http://www.gis.hctx.net. Accessed February 2010.
- Accreditation Program: Hospital. Available by purchase from The Joint Commission on Accreditation of Healthcare Organizations.2008.
- International Conference on Harmonisation. Available at: www.ich.org/cache/compo/276–254‐1.html. Accessed February 2010.
Hospitals have important legal and ethical responsibilities for human participant research conducted within their facilities, such as ensuring that research complies with federal regulations and presents minimal risks to patients. Many hospitals accept as sufficient the federal requirement that human participant research studies have Institutional Review Board (IRB) review and approval. IRBs must review proposed research according to numerous criteria, such as scientific soundness, alignment with accepted ethics principles and weighing of benefit vs. risk to study participants.1, 2 The legally required aspects of IRB review do not, however, include considering practical matters in implementing and operating an interventional clinical trial in the complex environment of the modern acute care hospital.
Our hospital system established a broad policy requiring internal review and formal approval of any human participant research conducted within any of its hospitals, including studies that enroll hospital patients or hospital employees, utilize hospital medical records, or request hospital‐provided services for research tests or procedures. The purpose of this paper is to describe this formal hospital system review and approval process, the reasons for implementing it, and the types of issues considered prior to our hospital system granting a principal investigator permission to conduct a study.
Background
Surprisingly little healthcare or medical literature exists regarding hospital responsibilities toward human subject research conducted on its premises. Much of the literature focuses on ethics issues, the nature of informed consent, and study design. As critical as these discussions are, they seldom address the numerous complex operational issues and challenges that implementation of a clinical trial can create in a hospital setting.
Flanders et al.3 make the case for hospitalists and specialists to work together to support research that includes inpatients as study participants. Moore and Goldberg4 discuss the ingredients of successfully developing a research program in a community hospital and mention the need to involve all affected hospital departments in the initial hospital review of a study, evaluating study impact on hospital workflow, and establishing processes related to budgeting and billing. Jamerson5 also makes the case for hospital departmental review and involvement, assessment of the ability to integrate study activities into the hospital structure, assessment of the resources needed to support the research, determination of whether the hospital will contribute financially to the research, and explicit decision making regarding the assumption of institutional risk.
Despite the recognition that US patients increasingly live with multiple chronic conditions6 and that clinical trial protocols have become more procedure and resource intensive and costly,79 there has not been a corollary recognition of the increasing need for hospitals to understand and manage research activities occurring within their facilities.
Our organization is a hospital system with 9 acute care hospitals, including an academic teaching hospital (affiliated with a university medical school) with a Level 1 Trauma Center, 1 specialty rehabilitation hospital, numerous specialized clinics, and a LifeFlight Program with a 6‐helicopter fleet (Geisinger, Danville, PA). This system of hospitals serves the fourth largest metropolitan community in the US (Houston and Harris County in southeastern TX), with a population of nearly four million and a geographic spread of 1778 square miles.10, 11 The hospital system has approximately 140,700 inpatient admissions per year and 586,000 outpatient visits.
Eight years ago, our hospital system adopted a corporate policy requiring that any activity associated with human participant research receive prior hospital system review and approval. Our organization considers this review process vital to: (1) maintaining our commitment to our Federalwide Assurance with the Office for Human Research Protections, (2) abiding by the Joint Commission requirements related to research,12 (3) protecting the safety and confidentiality of patients and employees who are potential or actual research participants, (4) protecting the confidentiality of participants' medical information, (5) assuring that legal fundamentals and good clinical practice (GCH)13 are a part of study plans, (6) assuring that studies are operationally feasible, and (7) evaluating and minimizing risks to patients and risks to the organization.
Review and Approval Process
Overview
An investigator triggers a formal hospital system review by submitting study documents to 1 of the IRBs listed on the system's Federalwide Assurance through the electronic IRB system and completing the required hospital system's Research Application Form. The hospital system review occurs in parallel with the IRB review, not duplicating it but rather focusing separately on patient safety, operational and financial issues, and hospital risk issues.
Our Clinical Innovation and Research Institute manages the hospital system review. Upon electronic notification of a new study submission, an Institute Clinical Research Associate examines all study‐related documents, including the completed Research Application Form and other submitted documents, such as the study protocol, the investigational product's Investigator's Brochure, consent forms, Food and Drug Administration (FDA) letters, survey questions, and diary and other data collection forms. The Associate may spend considerable time communicating with the investigator's research team, collecting missing information and building a complete study file, including identifying the affected hospitals and hospital departments. The Institute then provides study documents to the individuals responsible for hospital‐level research review, and each affected hospital conducts its own internal review and approval process.
The hospital‐level review process varies depending on the hospitals involved. The academic teaching hospital has the most detailed review process, due to the complexities and risks associated with the full spectrum of human participant research which occurs there (Hospital A in Figure 1). If a study affects this hospital, the Institute provides study documents to each affected Department Manager, the affected Service Line Chief, the Chief Medical Officer or Medical Director of each Intensive Care Unit within which the study will recruit and enroll patients, and the Infection Control Officer and Radiation Safety Officer, as appropriate.

The specialty rehabilitation hospital has a long‐standing national reputation for its research programs; its Director of Research knows each investigator, reviews each study, and provides that hospital's administrative review (Hospital B). Seven of the system's community hospitals have either a Chief Executive Officer, Chief Nursing Officer, or Chief Financial Officer serve as the hospital's executive administrator responsible for research review, and this person distributes the study documents to the Chief Medical Officer, if deemed necessary, and to each affected department (Hospitals C‐I). One of the smaller community hospitals participates in relatively few studies; the Chief Nursing Officer reviews and provides hospital‐level approval (Hospital J). For retrospective studies requesting a clinical data set, the Institute provides the study documents to the Director of the Information Systems Department. All studies accessing patient data are provided to the system Privacy Officer for review and approval.
Studies may involve 1 or more hospitals; some have involved as many as 7 at once.
All reviewers also receive a standardized Research Study Evaluation Form for their written comments and recommendations (Approve, Disapprove, or Defer) regarding whether the hospital system should approve the study.
If a study requests hospital‐provided research services, the Institute's Research Financial Coordinator develops a research budget listing the hospital charges that the researcher will incur for these tests and procedures.
Once reviewers return completed Evaluation Forms, the Institute Clinical Research Associate makes an initial determination whether the review process has satisfactorily answered all questions and resolved all outstanding issues. The Manager of Clinical Research Operations then examines the study file to ensure a satisfactory review. Finally, the system Executive Director for Clinical Research provides a Letter of Approval to the Principal Investigator, which serves as the agreement of terms for conducting the study within the hospital system. The letter contains standard stipulations, such as requiring the Principal Investigator to abide by federal law and International Conference on Harmonization (ICH) GCPs and the budget for the hospital‐provided research tests and procedures. Additionally, it includes any stipulations unique to the studyfor example, that the Principal Investigator will provide training to hospital personnel who will be operating nonhospital equipment. The Institute provides affected hospital departments with copies of the approval letter. Upon signing and returning the letter, the Principal Investigator may begin the study in the designated hospitals.
Some details about the hospital system review are discussed in sections below.
Patient Safety and Human Participant Protections
Participant Recruitment Plans
Sometimes IRB submission documents do not adequately describe how researchers will identify potential study participants and approach them for consent. Key concerns which we address include how researchers will identify potential participants in a Health Insurance Portability and Accountability Act (HIPAA)‐compliant manner, the level and type of illnesses of the patients whom the investigator intends to recruit, whether the researcher must obtain the admitting or attending physician's permission, the qualifications of the person making the initial patient contact, and how and when that person will make contact, with special attention required if subjects may include very ill patients in an Intensive Care Unit (ICU).
In‐service for Unit Personnel and Pharmacists
For most interventional studies, we require that the principal investigator's research team provide a plan for study education in‐service for nurses, technicians, respiratory therapists, and pharmacists who may be involved in the care of a patient enrolled in the study. This is usually an in‐person presentation done at a regular unit meeting, with an additional investigational drug‐specific in‐service provided and available on the hospital system intranet for the pharmacists. We remind researchers that the plan must also include an in‐service for night shift personnel, who are often otherwise overlooked.
Personnel Administering the Investigational Product
Sometimes submitted protocols do not state who will administer the investigational productthe physician investigator, other members of the research team or unit nurses. If the investigator expects unit nurses to administer the product, the hospital needs to determine whether the nurses' experience and training qualifies them to administer it, assess for adverse events, and provide care for patients with these events. If the nurses are not qualified, then the hospital needs to decide whether the nurses should receive training or a member of the research team should administer the product. Some research studies involve investigational agents with novel administration techniques or risks of immediate severe adverse events, requiring the presence of a physician knowledgeable about the investigational product.
Care of Study Participants With Adverse Events
Studies with unusual investigational agents can also raise a Unit Director's or ICU Medical Director's concern as to whether bedside nurses can appropriately and adequately discern and respond to potential adverse events. If the investigational agent might result in an event not normally anticipated in patients in that particular unit, the hospital may need to consider additional preparation or staffing.
Consent Documentation
Federal law and ICH GCPs require that principal investigators have signed consent forms available in the research records, which may be off of the hospital premises in physician clinics or office areas. Our hospital system requires a copy of the consent form in the patient's medical chart if the research team conducted the consent process within the hospital or if the study participant will be an inpatient for a procedure included as part of the study protocol, whether the patient was recruited while an inpatient or prior to inpatient admission. This is important for meeting the Joint Commission's standards related to research. We established an internal monitoring program to verify that researchers were providing copies of consent, assent and parental permission forms to the Health Information Management Office for placement in medical charts.
IRB‐related Issues
Occasionally a hospital system review identifies an IRB‐related concern, such as a known possible adverse event missing from the consent form, unexplained medical terms in the consent form, exclusion criteria not mentioning pregnancy or a consent form not covering a pregnancy test even though the protocol text mentions these, or missing Investigational New Drug Application (IND) or Investigational Device Exemption (IDE) information. Institute staff route such concerns to the IRB for follow‐up with the researcher as necessary. If a hospital system review identifies an ethical concern, the Institute consults with the IRB Chair or ethicist member. Usually the Chair will assess the concern, raise options for addressing it, and recommend a course of action.
Operational Issues
Feasibility and Implementation
We urge researchers to meet with Institute staff to discuss implementation of a protocol as a real‐world, operationalized study and also encourage them to meet with managers of the primary units where the study will take place. Researchers, however, may develop and submit industry‐sponsored clinical trials or investigator‐initiated studies without such prior discussions and may not have adequately considered operational feasibility.
Given the increasing complexity of investigational agents, study designs, study procedures, and patient safety monitoring, hospital reviewers need to consider exactly who will perform study procedures and processes and how those people will do so. If a blood or tissue sample needs to be spun, packaged, and mailed on dry ice within a limited timeframe, for example, who will do this and will the supplies and equipment be available as needed? A study protocol can lead to a change in normal unit processes. Operating suite managers, for instance, may need to adjust schedules or work with research teams if research activities may prolong a procedure beyond average timeframes.
Other potential impacts abound. Research teams sometimes assume, without checking with hospital managers, that hospital staff, usually nurses or respiratory therapists, will perform research procedures of the kind they usually perform as standard clinical procedure. Most commonly, researchers assume that bedside nurses will perform frequent blood draws necessary for a pharmacokinetic substudy. Unit managers, however, may not agree to commit nurses' time to this task, depending on the number and timing of the draws. During surgery, a study may require recording of events or timelines which are not usually recorded. Researchers sometimes assume that operating room personnel will be able to focus on this data collection. Hospital managers and directors are often concerned that researchers assume that unit staff will transport patients within‐hospital for research procedures, which can involve repeatedly moving a patient from their home unit to Radiology or elsewhere and then back to the home unit. For a large hospital, this can involve considerable staff time spent away from the home unit, which may affect unit operations. Occasionally, an investigator requests that a hospital temporarily or permanently store blood or tissue samples. Since hospitals are not necessarily prepared to store large numbers of samples for extended periods, we address each such request on a case‐by‐case basis.
Investigational Product
Hospital reviewers need to consider an assortment of challenges relating to the receipt, storage, dispensing, and accountability recording of investigational products. If a hospital pharmacy will be dispensing an investigational drug, then pharmacists need to anticipate its arrival from the sponsor and know storage and other sponsor, study and FDA requirements. Pharmacists also need to know if they are expected to prepare placebo pills. If a drug is an agent that an external pharmacy needs to prepare and compound, the chain of custody documentation of the drug as it moves from manufacturer to external pharmacy to hospital pharmacy needs to be clear.
While academic hospitals usually have research‐knowledgeable pharmacists, community‐based hospital pharmacists may not be familiar with the special requirements of the FDA or sponsor, such as securing investigational products separately from marketed products, recording batch numbers, maintaining accountability logs, and following procedures for return or destruction of remaining product upon study closure. In our hospital system, 2 Research Pharmacists in the Investigational Drug Pharmacy at the academic teaching hospital serve as expert advisers to pharmacists of the community hospitals.
If a research study involves use of an investigational device, hospital reviewers need to consider what the device is, how it should be secured if stored on the unit, and how to document storage and accountability.
Biologics and radioactive materials can present unique challenges. For instance, our system requires researchers to provide hospitals with chain of custody documentation, similar to that used for organ transplants, when patient biologicals leave the hospital (for instance, for processing at a nearby accredited Cell Processing Facility) and returned to the hospital for infusion back into the patient, to confirm that the right product was returned to the hospital and infused into the right patient.
Unaffiliated Principal Investigators and Other Personnel
Increasingly, researchers who are not affiliated with our hospital system have inquired about conducting studies in 1 or more of our hospitals. We have been quite surprised by the number of inquiries from researchers or sponsors who presume we will grant immediate permission for them to access our patient lists for recruitment purposes, allow unidentified research team members to enter our hospitals, approve team members to conduct active recruitment of our hospital patients, and grant team members access to patient data.
Additionally, as clinical and translational research projects become increasingly multidisciplinary and involve cross‐organizational collaborations, many research teams include unaffiliated personnel from other organizations, such as faculty at local or distant universities, employees of a site management organization, and employees of the city or county health department.
We do not permit clinicians who are not clinically credentialed at our hospitals to engage in interventional research within our hospital system. For studies that include any intervention that qualifies as a clinical procedure, the Principal Investigator must become clinically credentialed by the hospital or an already credentialed clinician must become the local Principal Investigator for the study, and all team members who perform clinical procedures must also become clinically credentialed.
An unusual situation occurred when researchers from a university not formally affiliated with our hospital system sought to transfer a study to 1 of our hospitals following Hurricane Ike. For 15 years, the research team had performed needle muscle biopsies, for which they had received training and credentialing at their home institution. Our hospital's Chief Medical Officer, however, did not feel comfortable accepting the credentialing performed at another institution and required the external researchers to apply for privileges through our hospital's credentialing process. The original credentialing documentation at their home institution was unavailable, in any case, due to flooding and building closures, so the researchers worked through the night to complete our applications. The hospital credentialing committee came to order on short notice and completed the credentialing process in record time, allowing the research team to see the study participants at our hospital with only a few missed visits and few study deviations for the Principal Investigator.
For research team members who will be performing no clinical procedures, our hospital system has a research credentialing process, discussed below.
Financial Issues
Study Finances
Researchers are typically aware of charges for the procedures that their studies most frequently require, such as labs, radiology, and research pharmacy, although they often do not inquire about the hospital's current charges, so their information may be somewhat dated when they negotiate with sponsors or submit grant applications.
When researchers plan on utilizing hospital staff to perform research‐required tasks, such as blood draws or patient transport for tests or procedures, however, they rarely include financial support to the hospital in their study budgets, leaving these tasks unfunded, which can be a problem for hospital reviewers who must approve the research plan.
In our review and approval process, we seek clarification as to payment for research products. Sponsors are not permitted by law to seek payment for investigational drugs, but when a research protocol uses a standard‐of‐care drug for strictly research‐related purposes, determining whether the sponsor, hospital, or study participant incurs financial responsibility becomes an issue requiring careful consideration. We ensure that consent forms explicitly state any special charges to the study participant. An investigational device that falls into an FDA classification that permits charges can be especially problematic. Often the devices are expensive; the hospital must purchase them in batches and pay immediately. If the researcher does not use all the devices, the unused ones sometimes cannot be returned. If investigational devices are more costly than standard‐of‐care devices, then the hospital could incur substantial losses in billings, since hospital charges are diagnosis and procedure code dependent and usually not adjusted for device cost.
Our standardized financial assessment has led to more beneficial arrangements with sponsors for the return and reimbursement of unused products and more informed hospital decisions as to whether to conduct a specific medical device clinical trial.
We also carefully review consent form language to clarify who incurs costs for research‐related adverse events and research injuries. For investigational products with extensive potential side effects or studies enrolling very ill patients, the costs associated with adverse events can be extremely high.
Investigators are affiliated with our hospitals, not employed by them, so our hospital system does not have budget agreements with funding organizations, but relies on these external entities to fund the studies adequately. Some investigator‐initiated studies may be funded by the investigator's organization, such as a private practice or university, but in our experience such funds are sometimes depleted before study completion.
Hospital Financial Information as Study Data
As a part of the study protocol, researchers occasionally request patient‐related cost data pertinent to specific procedures or to the treatment of certain medical conditions, hospital charges and payments received, or other financial data. We forward these research requests to a hospital Finance Chief to determine the appropriateness of releasing the requested data and, if approved, how the hospital will extract and present the data sets to the investigator.
Hospital Risk Issues
Research Equipment
Our academic teaching hospital and the university medical school affiliated with it are distinct organizations, so a particular risk issue arises for the hospital when a researcher wishes to transport and use nonhospital equipment on the hospital premises. Our hospital system has responsibility to ensure appropriate and safe operation of equipment. Consequently, our hospital system review identifies any proposed use of externally‐owned equipment and involves the system and hospital Risk Officers in assessing such use for risk to patients. Specifics addressed include ownership of equipment, whether the research team or hospital staff will operate it; whether the operators have received or will receive training, and the potential risks to patients of equipment malfunctions. Upon determination that individuals will operate the equipment appropriately with minimal risks to study participants, the hospital's Biomedical Department performs their standard safety check prior to the equipment's use in the hospital.
Clinical Data Generated by NonHospital Equipment
Bringing external equipment into a hospital has a rarely anticipated consequence: the generation and storage of patient data. Our hospital system review determines whether these data constitute source data per FDA and ICH GCP definitions, whether the data are clinically pertinent, and whether the data need to go into the patient's hospital medical record. For example, if a university faculty researcher brings a vital signs monitor into the hospital to collect and electronically record data from hospital patients enrolled in a study, we may require that the research team print the data for insertion into the patient's hospital medical record.
Surveys of Hospital Employees
A research study that seeks to survey or interview hospital system employees raises a different type of institutional concern. In such a case, system and hospital Human Resources Department (HR) personnel review the planned study, paying close attention to how the research team will recruit employees and what type of information the team will request from them. HR does not want employees feeling that they must participate in a study simply because it takes place in their facility and wants to protect the identity of employees participating in anonymous surveys. On occasion, Institute staff distribute surveys (and sometimes collect them when completed), providing an identity firewall between the employee who elects to participate and the researcher.
HR may also limit researchers from asking especially personal questions or questions inquiring about the recruitment, hiring, and retention practices of their employer. HR must consider whether the study design and survey questions raise liability concerns. Studies that include employee focus groups or one‐on‐one interviews raise issues pertaining to the purpose and content of the focus groups or interviews, mechanisms to address special issues or complaints that may arise during them, and determination of whether the employees may participate during paid time as opposed to participating while off‐the‐clock.
Nonclinical Research Credentialing Process
It is vital that hospitals know who will be on hospital premises for a research study and what activities they will be engaging in. If a research team member is not already affiliated with our hospital system and will be engaging in noninterventional research activities, such as conducting the research consent process, administering a survey, or providing educational materials, our hospital system review initiates a nonclinical research credentialing process. The Institute's Manager of Clinical Research Operations assesses the team members' qualifications, reviews their resumes, interviews each of them, and discusses exactly what activities they will be engaging in and their training and experience. In addition to evaluating their qualifications, the hospital must determine how such individuals will be identified once inside the hospital, including what type of badge (employee, contractor, or visitor) the hospital will provide them. Upon successful completion of the research credentialing process, the Institute explicitly names the approved individuals in the Letter of Approval to the Principal Investigator, with copies to the affected hospitals' units.
Discussion
Our hospital research review and approval process is critical to ensuring that only safe and regulatory‐compliant research activities occur within our hospital system, but the review and approval process, with its many steps and numerous reviewers, can be cumbersome. There is no substitute for human beings reading and understanding the protocol, consent forms for patient involvement in a study, the proposed use of protected health information, Investigator's Brochure, Research Application Form, and other study documents and then identifying pertinent issues and resolving them, and this process does require significant staff time.
We have improved (continuously) the Research Application Form to help in the crucial initial gathering of information about studies' operational needs. We have also converted from a predominantly paper‐based review process to the widespread use of electronic documents, but we have not automated the distribution process for these electronic documents and a staff person must still do this through email.
Despite our efforts to improve the review process, investigators are sometimes frustrated with it, particularly if someone identifies a new issue late in the process, or if the hospital system's approval for the study lags behind the IRB approval by more than a few days.
Currently, the hospital system provides the majority of study approvals to the Principal Investigator within 2 weeks of IRB approval, with some approvals provided within 1 day of IRB approval and others as long as 3 months afterward. Delays in hospital approval can be due to a study lacking required approval from a Department of Defense IRB, the FDA not providing permission to proceed with a study, the absence of an executed contract with a vendor to pick up and dispose of radioactive waste from the investigational product and many other factors. Of course, when the research team can respond in timely fashion to inquiries or issues that we have raised, that assists all of us in completing the review and approval process as quickly as possible.
The review and approval process benefits hospital patients, hospital personnel who will be supporting studies, and hospitals as institutions. Thinking through, planning, and preparing for study operations, particularly for studies taking place in an ICU, benefits the research team, hospital personnel, and the patients. Overall, the hospital system's research review and approval process affords many protections to our patients and reduces risks to the hospital system while permitting research studies to be conducted within its varied healthcare facilities.
We encourage researchers not to view today's modern hospital as bricks and mortar, but as an institution with deep responsibility for safety on hospital premises. Hospitals must meet over a thousand Joint Commission standards, set goals for patient outcomes, measure and report on quality indicators, protect patient confidentiality, maintain the safety an ever‐expanding array of simple and complex equipment, maintain, check and document contents of adult and pediatric crash carts, have 24‐7 code teams at‐the‐ready, create, maintain and store patient medical records securely, transport patients, and much more.
Conclusion
Our hospital system, in accordance with a system‐wide policy, engages in a comprehensive review and approval process for any human participant research that has been proposed to be conducted within one or more of our facilities. The process focuses primarily on patient safety within the hospital premises, operational study issues, financial issues and hospital risk issues. This process decreases risks to the patients, researchers, and hospital facilities engaging in human participant research.
Hospitals have important legal and ethical responsibilities for human participant research conducted within their facilities, such as ensuring that research complies with federal regulations and presents minimal risks to patients. Many hospitals accept as sufficient the federal requirement that human participant research studies have Institutional Review Board (IRB) review and approval. IRBs must review proposed research according to numerous criteria, such as scientific soundness, alignment with accepted ethics principles and weighing of benefit vs. risk to study participants.1, 2 The legally required aspects of IRB review do not, however, include considering practical matters in implementing and operating an interventional clinical trial in the complex environment of the modern acute care hospital.
Our hospital system established a broad policy requiring internal review and formal approval of any human participant research conducted within any of its hospitals, including studies that enroll hospital patients or hospital employees, utilize hospital medical records, or request hospital‐provided services for research tests or procedures. The purpose of this paper is to describe this formal hospital system review and approval process, the reasons for implementing it, and the types of issues considered prior to our hospital system granting a principal investigator permission to conduct a study.
Background
Surprisingly little healthcare or medical literature exists regarding hospital responsibilities toward human subject research conducted on its premises. Much of the literature focuses on ethics issues, the nature of informed consent, and study design. As critical as these discussions are, they seldom address the numerous complex operational issues and challenges that implementation of a clinical trial can create in a hospital setting.
Flanders et al.3 make the case for hospitalists and specialists to work together to support research that includes inpatients as study participants. Moore and Goldberg4 discuss the ingredients of successfully developing a research program in a community hospital and mention the need to involve all affected hospital departments in the initial hospital review of a study, evaluating study impact on hospital workflow, and establishing processes related to budgeting and billing. Jamerson5 also makes the case for hospital departmental review and involvement, assessment of the ability to integrate study activities into the hospital structure, assessment of the resources needed to support the research, determination of whether the hospital will contribute financially to the research, and explicit decision making regarding the assumption of institutional risk.
Despite the recognition that US patients increasingly live with multiple chronic conditions6 and that clinical trial protocols have become more procedure and resource intensive and costly,79 there has not been a corollary recognition of the increasing need for hospitals to understand and manage research activities occurring within their facilities.
Our organization is a hospital system with 9 acute care hospitals, including an academic teaching hospital (affiliated with a university medical school) with a Level 1 Trauma Center, 1 specialty rehabilitation hospital, numerous specialized clinics, and a LifeFlight Program with a 6‐helicopter fleet (Geisinger, Danville, PA). This system of hospitals serves the fourth largest metropolitan community in the US (Houston and Harris County in southeastern TX), with a population of nearly four million and a geographic spread of 1778 square miles.10, 11 The hospital system has approximately 140,700 inpatient admissions per year and 586,000 outpatient visits.
Eight years ago, our hospital system adopted a corporate policy requiring that any activity associated with human participant research receive prior hospital system review and approval. Our organization considers this review process vital to: (1) maintaining our commitment to our Federalwide Assurance with the Office for Human Research Protections, (2) abiding by the Joint Commission requirements related to research,12 (3) protecting the safety and confidentiality of patients and employees who are potential or actual research participants, (4) protecting the confidentiality of participants' medical information, (5) assuring that legal fundamentals and good clinical practice (GCH)13 are a part of study plans, (6) assuring that studies are operationally feasible, and (7) evaluating and minimizing risks to patients and risks to the organization.
Review and Approval Process
Overview
An investigator triggers a formal hospital system review by submitting study documents to 1 of the IRBs listed on the system's Federalwide Assurance through the electronic IRB system and completing the required hospital system's Research Application Form. The hospital system review occurs in parallel with the IRB review, not duplicating it but rather focusing separately on patient safety, operational and financial issues, and hospital risk issues.
Our Clinical Innovation and Research Institute manages the hospital system review. Upon electronic notification of a new study submission, an Institute Clinical Research Associate examines all study‐related documents, including the completed Research Application Form and other submitted documents, such as the study protocol, the investigational product's Investigator's Brochure, consent forms, Food and Drug Administration (FDA) letters, survey questions, and diary and other data collection forms. The Associate may spend considerable time communicating with the investigator's research team, collecting missing information and building a complete study file, including identifying the affected hospitals and hospital departments. The Institute then provides study documents to the individuals responsible for hospital‐level research review, and each affected hospital conducts its own internal review and approval process.
The hospital‐level review process varies depending on the hospitals involved. The academic teaching hospital has the most detailed review process, due to the complexities and risks associated with the full spectrum of human participant research which occurs there (Hospital A in Figure 1). If a study affects this hospital, the Institute provides study documents to each affected Department Manager, the affected Service Line Chief, the Chief Medical Officer or Medical Director of each Intensive Care Unit within which the study will recruit and enroll patients, and the Infection Control Officer and Radiation Safety Officer, as appropriate.

The specialty rehabilitation hospital has a long‐standing national reputation for its research programs; its Director of Research knows each investigator, reviews each study, and provides that hospital's administrative review (Hospital B). Seven of the system's community hospitals have either a Chief Executive Officer, Chief Nursing Officer, or Chief Financial Officer serve as the hospital's executive administrator responsible for research review, and this person distributes the study documents to the Chief Medical Officer, if deemed necessary, and to each affected department (Hospitals C‐I). One of the smaller community hospitals participates in relatively few studies; the Chief Nursing Officer reviews and provides hospital‐level approval (Hospital J). For retrospective studies requesting a clinical data set, the Institute provides the study documents to the Director of the Information Systems Department. All studies accessing patient data are provided to the system Privacy Officer for review and approval.
Studies may involve 1 or more hospitals; some have involved as many as 7 at once.
All reviewers also receive a standardized Research Study Evaluation Form for their written comments and recommendations (Approve, Disapprove, or Defer) regarding whether the hospital system should approve the study.
If a study requests hospital‐provided research services, the Institute's Research Financial Coordinator develops a research budget listing the hospital charges that the researcher will incur for these tests and procedures.
Once reviewers return completed Evaluation Forms, the Institute Clinical Research Associate makes an initial determination whether the review process has satisfactorily answered all questions and resolved all outstanding issues. The Manager of Clinical Research Operations then examines the study file to ensure a satisfactory review. Finally, the system Executive Director for Clinical Research provides a Letter of Approval to the Principal Investigator, which serves as the agreement of terms for conducting the study within the hospital system. The letter contains standard stipulations, such as requiring the Principal Investigator to abide by federal law and International Conference on Harmonization (ICH) GCPs and the budget for the hospital‐provided research tests and procedures. Additionally, it includes any stipulations unique to the studyfor example, that the Principal Investigator will provide training to hospital personnel who will be operating nonhospital equipment. The Institute provides affected hospital departments with copies of the approval letter. Upon signing and returning the letter, the Principal Investigator may begin the study in the designated hospitals.
Some details about the hospital system review are discussed in sections below.
Patient Safety and Human Participant Protections
Participant Recruitment Plans
Sometimes IRB submission documents do not adequately describe how researchers will identify potential study participants and approach them for consent. Key concerns which we address include how researchers will identify potential participants in a Health Insurance Portability and Accountability Act (HIPAA)‐compliant manner, the level and type of illnesses of the patients whom the investigator intends to recruit, whether the researcher must obtain the admitting or attending physician's permission, the qualifications of the person making the initial patient contact, and how and when that person will make contact, with special attention required if subjects may include very ill patients in an Intensive Care Unit (ICU).
In‐service for Unit Personnel and Pharmacists
For most interventional studies, we require that the principal investigator's research team provide a plan for study education in‐service for nurses, technicians, respiratory therapists, and pharmacists who may be involved in the care of a patient enrolled in the study. This is usually an in‐person presentation done at a regular unit meeting, with an additional investigational drug‐specific in‐service provided and available on the hospital system intranet for the pharmacists. We remind researchers that the plan must also include an in‐service for night shift personnel, who are often otherwise overlooked.
Personnel Administering the Investigational Product
Sometimes submitted protocols do not state who will administer the investigational productthe physician investigator, other members of the research team or unit nurses. If the investigator expects unit nurses to administer the product, the hospital needs to determine whether the nurses' experience and training qualifies them to administer it, assess for adverse events, and provide care for patients with these events. If the nurses are not qualified, then the hospital needs to decide whether the nurses should receive training or a member of the research team should administer the product. Some research studies involve investigational agents with novel administration techniques or risks of immediate severe adverse events, requiring the presence of a physician knowledgeable about the investigational product.
Care of Study Participants With Adverse Events
Studies with unusual investigational agents can also raise a Unit Director's or ICU Medical Director's concern as to whether bedside nurses can appropriately and adequately discern and respond to potential adverse events. If the investigational agent might result in an event not normally anticipated in patients in that particular unit, the hospital may need to consider additional preparation or staffing.
Consent Documentation
Federal law and ICH GCPs require that principal investigators have signed consent forms available in the research records, which may be off of the hospital premises in physician clinics or office areas. Our hospital system requires a copy of the consent form in the patient's medical chart if the research team conducted the consent process within the hospital or if the study participant will be an inpatient for a procedure included as part of the study protocol, whether the patient was recruited while an inpatient or prior to inpatient admission. This is important for meeting the Joint Commission's standards related to research. We established an internal monitoring program to verify that researchers were providing copies of consent, assent and parental permission forms to the Health Information Management Office for placement in medical charts.
IRB‐related Issues
Occasionally a hospital system review identifies an IRB‐related concern, such as a known possible adverse event missing from the consent form, unexplained medical terms in the consent form, exclusion criteria not mentioning pregnancy or a consent form not covering a pregnancy test even though the protocol text mentions these, or missing Investigational New Drug Application (IND) or Investigational Device Exemption (IDE) information. Institute staff route such concerns to the IRB for follow‐up with the researcher as necessary. If a hospital system review identifies an ethical concern, the Institute consults with the IRB Chair or ethicist member. Usually the Chair will assess the concern, raise options for addressing it, and recommend a course of action.
Operational Issues
Feasibility and Implementation
We urge researchers to meet with Institute staff to discuss implementation of a protocol as a real‐world, operationalized study and also encourage them to meet with managers of the primary units where the study will take place. Researchers, however, may develop and submit industry‐sponsored clinical trials or investigator‐initiated studies without such prior discussions and may not have adequately considered operational feasibility.
Given the increasing complexity of investigational agents, study designs, study procedures, and patient safety monitoring, hospital reviewers need to consider exactly who will perform study procedures and processes and how those people will do so. If a blood or tissue sample needs to be spun, packaged, and mailed on dry ice within a limited timeframe, for example, who will do this and will the supplies and equipment be available as needed? A study protocol can lead to a change in normal unit processes. Operating suite managers, for instance, may need to adjust schedules or work with research teams if research activities may prolong a procedure beyond average timeframes.
Other potential impacts abound. Research teams sometimes assume, without checking with hospital managers, that hospital staff, usually nurses or respiratory therapists, will perform research procedures of the kind they usually perform as standard clinical procedure. Most commonly, researchers assume that bedside nurses will perform frequent blood draws necessary for a pharmacokinetic substudy. Unit managers, however, may not agree to commit nurses' time to this task, depending on the number and timing of the draws. During surgery, a study may require recording of events or timelines which are not usually recorded. Researchers sometimes assume that operating room personnel will be able to focus on this data collection. Hospital managers and directors are often concerned that researchers assume that unit staff will transport patients within‐hospital for research procedures, which can involve repeatedly moving a patient from their home unit to Radiology or elsewhere and then back to the home unit. For a large hospital, this can involve considerable staff time spent away from the home unit, which may affect unit operations. Occasionally, an investigator requests that a hospital temporarily or permanently store blood or tissue samples. Since hospitals are not necessarily prepared to store large numbers of samples for extended periods, we address each such request on a case‐by‐case basis.
Investigational Product
Hospital reviewers need to consider an assortment of challenges relating to the receipt, storage, dispensing, and accountability recording of investigational products. If a hospital pharmacy will be dispensing an investigational drug, then pharmacists need to anticipate its arrival from the sponsor and know storage and other sponsor, study and FDA requirements. Pharmacists also need to know if they are expected to prepare placebo pills. If a drug is an agent that an external pharmacy needs to prepare and compound, the chain of custody documentation of the drug as it moves from manufacturer to external pharmacy to hospital pharmacy needs to be clear.
While academic hospitals usually have research‐knowledgeable pharmacists, community‐based hospital pharmacists may not be familiar with the special requirements of the FDA or sponsor, such as securing investigational products separately from marketed products, recording batch numbers, maintaining accountability logs, and following procedures for return or destruction of remaining product upon study closure. In our hospital system, 2 Research Pharmacists in the Investigational Drug Pharmacy at the academic teaching hospital serve as expert advisers to pharmacists of the community hospitals.
If a research study involves use of an investigational device, hospital reviewers need to consider what the device is, how it should be secured if stored on the unit, and how to document storage and accountability.
Biologics and radioactive materials can present unique challenges. For instance, our system requires researchers to provide hospitals with chain of custody documentation, similar to that used for organ transplants, when patient biologicals leave the hospital (for instance, for processing at a nearby accredited Cell Processing Facility) and returned to the hospital for infusion back into the patient, to confirm that the right product was returned to the hospital and infused into the right patient.
Unaffiliated Principal Investigators and Other Personnel
Increasingly, researchers who are not affiliated with our hospital system have inquired about conducting studies in 1 or more of our hospitals. We have been quite surprised by the number of inquiries from researchers or sponsors who presume we will grant immediate permission for them to access our patient lists for recruitment purposes, allow unidentified research team members to enter our hospitals, approve team members to conduct active recruitment of our hospital patients, and grant team members access to patient data.
Additionally, as clinical and translational research projects become increasingly multidisciplinary and involve cross‐organizational collaborations, many research teams include unaffiliated personnel from other organizations, such as faculty at local or distant universities, employees of a site management organization, and employees of the city or county health department.
We do not permit clinicians who are not clinically credentialed at our hospitals to engage in interventional research within our hospital system. For studies that include any intervention that qualifies as a clinical procedure, the Principal Investigator must become clinically credentialed by the hospital or an already credentialed clinician must become the local Principal Investigator for the study, and all team members who perform clinical procedures must also become clinically credentialed.
An unusual situation occurred when researchers from a university not formally affiliated with our hospital system sought to transfer a study to 1 of our hospitals following Hurricane Ike. For 15 years, the research team had performed needle muscle biopsies, for which they had received training and credentialing at their home institution. Our hospital's Chief Medical Officer, however, did not feel comfortable accepting the credentialing performed at another institution and required the external researchers to apply for privileges through our hospital's credentialing process. The original credentialing documentation at their home institution was unavailable, in any case, due to flooding and building closures, so the researchers worked through the night to complete our applications. The hospital credentialing committee came to order on short notice and completed the credentialing process in record time, allowing the research team to see the study participants at our hospital with only a few missed visits and few study deviations for the Principal Investigator.
For research team members who will be performing no clinical procedures, our hospital system has a research credentialing process, discussed below.
Financial Issues
Study Finances
Researchers are typically aware of charges for the procedures that their studies most frequently require, such as labs, radiology, and research pharmacy, although they often do not inquire about the hospital's current charges, so their information may be somewhat dated when they negotiate with sponsors or submit grant applications.
When researchers plan on utilizing hospital staff to perform research‐required tasks, such as blood draws or patient transport for tests or procedures, however, they rarely include financial support to the hospital in their study budgets, leaving these tasks unfunded, which can be a problem for hospital reviewers who must approve the research plan.
In our review and approval process, we seek clarification as to payment for research products. Sponsors are not permitted by law to seek payment for investigational drugs, but when a research protocol uses a standard‐of‐care drug for strictly research‐related purposes, determining whether the sponsor, hospital, or study participant incurs financial responsibility becomes an issue requiring careful consideration. We ensure that consent forms explicitly state any special charges to the study participant. An investigational device that falls into an FDA classification that permits charges can be especially problematic. Often the devices are expensive; the hospital must purchase them in batches and pay immediately. If the researcher does not use all the devices, the unused ones sometimes cannot be returned. If investigational devices are more costly than standard‐of‐care devices, then the hospital could incur substantial losses in billings, since hospital charges are diagnosis and procedure code dependent and usually not adjusted for device cost.
Our standardized financial assessment has led to more beneficial arrangements with sponsors for the return and reimbursement of unused products and more informed hospital decisions as to whether to conduct a specific medical device clinical trial.
We also carefully review consent form language to clarify who incurs costs for research‐related adverse events and research injuries. For investigational products with extensive potential side effects or studies enrolling very ill patients, the costs associated with adverse events can be extremely high.
Investigators are affiliated with our hospitals, not employed by them, so our hospital system does not have budget agreements with funding organizations, but relies on these external entities to fund the studies adequately. Some investigator‐initiated studies may be funded by the investigator's organization, such as a private practice or university, but in our experience such funds are sometimes depleted before study completion.
Hospital Financial Information as Study Data
As a part of the study protocol, researchers occasionally request patient‐related cost data pertinent to specific procedures or to the treatment of certain medical conditions, hospital charges and payments received, or other financial data. We forward these research requests to a hospital Finance Chief to determine the appropriateness of releasing the requested data and, if approved, how the hospital will extract and present the data sets to the investigator.
Hospital Risk Issues
Research Equipment
Our academic teaching hospital and the university medical school affiliated with it are distinct organizations, so a particular risk issue arises for the hospital when a researcher wishes to transport and use nonhospital equipment on the hospital premises. Our hospital system has responsibility to ensure appropriate and safe operation of equipment. Consequently, our hospital system review identifies any proposed use of externally‐owned equipment and involves the system and hospital Risk Officers in assessing such use for risk to patients. Specifics addressed include ownership of equipment, whether the research team or hospital staff will operate it; whether the operators have received or will receive training, and the potential risks to patients of equipment malfunctions. Upon determination that individuals will operate the equipment appropriately with minimal risks to study participants, the hospital's Biomedical Department performs their standard safety check prior to the equipment's use in the hospital.
Clinical Data Generated by NonHospital Equipment
Bringing external equipment into a hospital has a rarely anticipated consequence: the generation and storage of patient data. Our hospital system review determines whether these data constitute source data per FDA and ICH GCP definitions, whether the data are clinically pertinent, and whether the data need to go into the patient's hospital medical record. For example, if a university faculty researcher brings a vital signs monitor into the hospital to collect and electronically record data from hospital patients enrolled in a study, we may require that the research team print the data for insertion into the patient's hospital medical record.
Surveys of Hospital Employees
A research study that seeks to survey or interview hospital system employees raises a different type of institutional concern. In such a case, system and hospital Human Resources Department (HR) personnel review the planned study, paying close attention to how the research team will recruit employees and what type of information the team will request from them. HR does not want employees feeling that they must participate in a study simply because it takes place in their facility and wants to protect the identity of employees participating in anonymous surveys. On occasion, Institute staff distribute surveys (and sometimes collect them when completed), providing an identity firewall between the employee who elects to participate and the researcher.
HR may also limit researchers from asking especially personal questions or questions inquiring about the recruitment, hiring, and retention practices of their employer. HR must consider whether the study design and survey questions raise liability concerns. Studies that include employee focus groups or one‐on‐one interviews raise issues pertaining to the purpose and content of the focus groups or interviews, mechanisms to address special issues or complaints that may arise during them, and determination of whether the employees may participate during paid time as opposed to participating while off‐the‐clock.
Nonclinical Research Credentialing Process
It is vital that hospitals know who will be on hospital premises for a research study and what activities they will be engaging in. If a research team member is not already affiliated with our hospital system and will be engaging in noninterventional research activities, such as conducting the research consent process, administering a survey, or providing educational materials, our hospital system review initiates a nonclinical research credentialing process. The Institute's Manager of Clinical Research Operations assesses the team members' qualifications, reviews their resumes, interviews each of them, and discusses exactly what activities they will be engaging in and their training and experience. In addition to evaluating their qualifications, the hospital must determine how such individuals will be identified once inside the hospital, including what type of badge (employee, contractor, or visitor) the hospital will provide them. Upon successful completion of the research credentialing process, the Institute explicitly names the approved individuals in the Letter of Approval to the Principal Investigator, with copies to the affected hospitals' units.
Discussion
Our hospital research review and approval process is critical to ensuring that only safe and regulatory‐compliant research activities occur within our hospital system, but the review and approval process, with its many steps and numerous reviewers, can be cumbersome. There is no substitute for human beings reading and understanding the protocol, consent forms for patient involvement in a study, the proposed use of protected health information, Investigator's Brochure, Research Application Form, and other study documents and then identifying pertinent issues and resolving them, and this process does require significant staff time.
We have improved (continuously) the Research Application Form to help in the crucial initial gathering of information about studies' operational needs. We have also converted from a predominantly paper‐based review process to the widespread use of electronic documents, but we have not automated the distribution process for these electronic documents and a staff person must still do this through email.
Despite our efforts to improve the review process, investigators are sometimes frustrated with it, particularly if someone identifies a new issue late in the process, or if the hospital system's approval for the study lags behind the IRB approval by more than a few days.
Currently, the hospital system provides the majority of study approvals to the Principal Investigator within 2 weeks of IRB approval, with some approvals provided within 1 day of IRB approval and others as long as 3 months afterward. Delays in hospital approval can be due to a study lacking required approval from a Department of Defense IRB, the FDA not providing permission to proceed with a study, the absence of an executed contract with a vendor to pick up and dispose of radioactive waste from the investigational product and many other factors. Of course, when the research team can respond in timely fashion to inquiries or issues that we have raised, that assists all of us in completing the review and approval process as quickly as possible.
The review and approval process benefits hospital patients, hospital personnel who will be supporting studies, and hospitals as institutions. Thinking through, planning, and preparing for study operations, particularly for studies taking place in an ICU, benefits the research team, hospital personnel, and the patients. Overall, the hospital system's research review and approval process affords many protections to our patients and reduces risks to the hospital system while permitting research studies to be conducted within its varied healthcare facilities.
We encourage researchers not to view today's modern hospital as bricks and mortar, but as an institution with deep responsibility for safety on hospital premises. Hospitals must meet over a thousand Joint Commission standards, set goals for patient outcomes, measure and report on quality indicators, protect patient confidentiality, maintain the safety an ever‐expanding array of simple and complex equipment, maintain, check and document contents of adult and pediatric crash carts, have 24‐7 code teams at‐the‐ready, create, maintain and store patient medical records securely, transport patients, and much more.
Conclusion
Our hospital system, in accordance with a system‐wide policy, engages in a comprehensive review and approval process for any human participant research that has been proposed to be conducted within one or more of our facilities. The process focuses primarily on patient safety within the hospital premises, operational study issues, financial issues and hospital risk issues. This process decreases risks to the patients, researchers, and hospital facilities engaging in human participant research.
- Code of Federal Regulations Part 45, Title 46.
- Code of Federal Regulations Part 50, Title 21.
- ,,,.The University of Michigan Specialist‐Hospitalist Allied Research Program: jumpstarting hospital medicine research.J Hosp Med.2008;3:308–313.
- ,.Successfully developing a cardiovascular research program in a community hospital.J Cardiovasc Manag.2004;15:13–19.
- .Developing an infrastructure for research in a free‐standing hospital.J Nurs Adm.2007;37:295–301.
- ,,.Prevalence of multiple chronic conditions in the United States' Medicare population.Health Qual Life Outcomes.2009;7:82.
- ,,,,.Assessing the impact of protocol design changes on clinical trial performance.Am J Ther.2008;15:450–457.
- ,,, et al.Factors affecting workload of cancer clinical trials: results of a multicenter study of the National Cancer Institute of Canada Clinical Trials Group.J Clin Oncol.2002;20:545–556.
- ,,, et al.The changing face of Phase 1 Cancer Clinical Trials: new challenges in study requirements.Cancer.2009;115(8):1592–1597.
- U.S. Census Bureau. Annual Estimates of the Resident Population for Counties of Texas: April 1, 2000 to July 1, 2008. Available at: http://www.census.gov/popest/counties/tables/CO‐EST2008–01‐48.xls. Accessed February 2010.
- Harris County Texas. Available at: http://www.gis.hctx.net. Accessed February 2010.
- Accreditation Program: Hospital. Available by purchase from The Joint Commission on Accreditation of Healthcare Organizations.2008.
- International Conference on Harmonisation. Available at: www.ich.org/cache/compo/276–254‐1.html. Accessed February 2010.
- Code of Federal Regulations Part 45, Title 46.
- Code of Federal Regulations Part 50, Title 21.
- ,,,.The University of Michigan Specialist‐Hospitalist Allied Research Program: jumpstarting hospital medicine research.J Hosp Med.2008;3:308–313.
- ,.Successfully developing a cardiovascular research program in a community hospital.J Cardiovasc Manag.2004;15:13–19.
- .Developing an infrastructure for research in a free‐standing hospital.J Nurs Adm.2007;37:295–301.
- ,,.Prevalence of multiple chronic conditions in the United States' Medicare population.Health Qual Life Outcomes.2009;7:82.
- ,,,,.Assessing the impact of protocol design changes on clinical trial performance.Am J Ther.2008;15:450–457.
- ,,, et al.Factors affecting workload of cancer clinical trials: results of a multicenter study of the National Cancer Institute of Canada Clinical Trials Group.J Clin Oncol.2002;20:545–556.
- ,,, et al.The changing face of Phase 1 Cancer Clinical Trials: new challenges in study requirements.Cancer.2009;115(8):1592–1597.
- U.S. Census Bureau. Annual Estimates of the Resident Population for Counties of Texas: April 1, 2000 to July 1, 2008. Available at: http://www.census.gov/popest/counties/tables/CO‐EST2008–01‐48.xls. Accessed February 2010.
- Harris County Texas. Available at: http://www.gis.hctx.net. Accessed February 2010.
- Accreditation Program: Hospital. Available by purchase from The Joint Commission on Accreditation of Healthcare Organizations.2008.
- International Conference on Harmonisation. Available at: www.ich.org/cache/compo/276–254‐1.html. Accessed February 2010.
FDA‐warning for IV Haloperidol: A Review
Haloperidol is Food and Drug Administration (FDA)‐approved in the United States for the management of acute and chronic psychotic disorders and widely used in the management of delirium‐associated agitation in hospitalized patients.1 Delirium in the hospital is an acute confusional state that frequently arises from multiple complex factors and may affect up to 30% of hospitalized patients.2 Although the first step in the management of delirium involves identification and treatment of underlying causes and offering supportive behavioral care; medications may be needed to control severe agitation.2 Low dose intravenous (IV) haloperidol (ie, 0.250.5 mg every 4 hours) is a commonly used medication in this setting as recommended by expert‐groups including the Cochrane Collaboration and the American Psychiatric Association.2, 3
Although injectable haloperidol, a butyrophenone‐derived antipsychotic agent pharmacologically related to the piperazine phenothiazines,4 is approved for IV use in many countries (Table 1), parenteral use is approved only for intramuscular (IM) administration in the US. Thus, IV administration of the drug in the US is considered an off‐label use.5
| Indication | Country | |||||
|---|---|---|---|---|---|---|
| Canada24 | France29 | Germany25 | Great Britain37 | Italy30 | Switzerland31 | |
| ||||||
| Mainly delirium (schizophrenia, other psychosis, short‐term management of psychomotor agitation, excitement, violent or dangerously impulsive behavior, vomiting, hiccup) | Short term treatment of agitation and aggressiveness during an acute or chronic psychotic episode, vomiting along with antimitotic post‐radiotherapy treatment | Acute and chronic schizophrenia, psycho‐motorical agitation of psychotic genesis | Schizophrenia, other psychosis, short‐term adjunctive management if psychomotor agitation, violent or dangerous impulsive behavior | Resistant forms of psycho‐motorical excitement, acute delirious and/or hallucinatory psychosis' chronic psychosis High doses restrictions: syndrome of psycho‐motorical excitement, acute delirious and/or hallucinatory psychosis, chronic psychosis | Acute schizophrenic episode, mania, vomiting | |
| IV dosing in adults | 12 mg every 24 hours | The use is limited to adult patients and the drug can be administered IM or IV. The IV route is restricted to the treatment of vomiting. | 510 mg/day, daily max.: 30(100) mg | 210 mg initially, PRN every 48 hours, daily max. 18 mg | 510 mg initially, PRN every hour, daily max. 60 mg | 5 mg PRN every 30 minutes |
| IV dosing in geriatric care | 0.250.5 mg | Single dose of 0.51.5 mg, daily max. 5 mg | Half adult dose | Adjust to appropriate dose | 0.5 mg, than PRN | |
| Risk factors for the development of cardiac adverse events | QT prolonging drugs, diabetes, obesity, hypokalemia, congenital long QT syndrome | Bradycardia 55 beats per minute, hypokalemia, congenital QT prolongation, other medications provoking bradycardia, deceleration of the intra‐cardiac transition or prolonged QT interval | QT syndrome, hypokalemia, other electrolyte imbalance, cardiovascular diseases, QT prolongation in the family history | Cardiovalscular disease, drugs that can prolong the QTc, diabetes, obesity, hypokalemia, congenital long QT syndrome | Contraindications: recent cardiac infarction, uncompensated cardiac insufficiency, cardiac arrhythmias, antiarrhythmic drugs, pre‐existing QT prolongation, cases of arrhythmia or torsades de pointes in the family history, untreated potassium imbalance, QTc prolonging drugs | QT syndrome, hypokalemia, hypomagnesemia, other electrolyte imbalances, cardiovascular diseases, hypothyreosis, QT prolongation in the family history |
| Monitoring recommendations | Electrolytes | ECG monitoring at admission time, electrolytes | ECG monitoring, electrolytes | Metabolic parameters | ECG at baseline and regular ECG monitoring, electrolytes | Close ECG monitoring, electrolytes |
| General recommendations | Regular reevaluation in long‐term use | Apply the lowest effective dose | Apply the lowest effective dose | Application per mouth is the route of choice | Decrease dose if QTc >500 msec | Switch to PO as soon as possible |
Haloperidol is often preferred over other antipsychotics as a result of its effectiveness, low rate of anticholinergic side effects, familiarity with dosing and usage, and minimal respiratory or sedative properties.6 Use of the IV route in patients with acute delirium has several advantages over the IM or oral route,7 including rapid onset, immediate bioavailability, and ease and safety of administration.
Prior to September 2007, the package insert for haloperidol alerted healthcare professionals to the risk of cardiovascular side effects. Based on case reports of potentially fatal cardiac events, the FDA revised the label, warning that the QT prolongation (QTP) and risk of torsades de pointes (TdP) were increased with IV administration of haloperidol or administration of the drug at greater than recommended doses. Unfortunately, neither the typical dosing range nor the minimum dose associated with these cardiac side effects were specified in this recommendation.5
It is well‐established that haloperidol may prolong the QT interval by blocking the repolarizing potassium IKr current.8 Although drugs that block the IKr channel can produce arrhythmia in healthy individuals, additional risk factors, such as underlying heart conditions, electrolyte imbalances (ie, hypokalemia and hypomagnesemia), concomitant proarrhythmic drug use, and mechanical ventilation may increase this risk.9 Prolongation of the QT interval has been associated with subsequent malignant cardiac arrhythmias including ventricular fibrillation and TdP.10 Prolongation of the QT interval is considered the strongest risk factor for TdP, particularly with a baseline QTc > 450 msec.9
Based on the increased risk for QTP and TdP and the case reports of cardiac events, the FDA advisory recommended continuous electrocardiogram (ECG) monitoring in patients receiving IV haloperidol.5 However, such monitoring may be impractical and costly in hospitalized patients who require low doses of IV haloperidol to manage acute delirium and who are not in telemetry or intensive care units.
The aim of this review was to evaluate the case reports leading to the recent FDA warning for IV haloperidol, specifically focusing on the presence of risk factors for arrhythmias. Based upon the evidence, an additional aim was to provide an institutional response to this warning toward the optimal use of this agent.
Method
Two search pathways were used to evaluate reports of haloperidol‐associated TdP and/or QT prolongation:
Literature Review
We searched for published literature in humans indexed in Pubmed (1966April 2009), EMBASE (1972April 2009), and Scopus (1823April 2009) using the search terms haloperidol or Haldol combined with intravenous or infusion and at least one of the following terms: QT prolongation, TdP, torsades de pointes, torsades with a specific focus on case reports.
References from the retrieved articles were also reviewed to search for additional case reports.
In addition to cases reported in English journals, several of our reports originated from Japan11 (translation provided by the FDA), Spain12 and Germany13 (translated by the primary author).
Search of the FDA Database
We reviewed all adverse drug events reported through MedWatch or those submitted by the manufacturer from November 1997 to April 2008 through the Freedom of Information Act (FOIA) request. The FDA provided a full‐text summary of 5944 reports involving oral, intramuscular and IV use of haloperidol. The FDA data were transferred to a Microsoft Access database and screened for the key terms torsade, QT, prolongation, wave. Incident report number, date of report, age, gender, origin of report, medication name, role of drug as categorized by the FDA (suspect, concomitant, primary suspect, secondary suspect), route, dose, units, duration, symptoms and FDA outcome category (death, life‐threatening, hospitalization initial or prolonged, disability, congenital anomaly, required intervention to prevent permanent damage, other) were recorded. Only those reports in which IV haloperidol was considered by the reporter to be the primary causative agent for the adverse event were reviewed. Available information included diagnosis, laboratory parameters, QTc measurement, cardiac symptoms, outcomes and a description of recovery. No peer review was applied to the MedWatch reports and the data reported in this publication reflect the original information from the FDA MedWatch database. Baseline QTc was either the value defined as such in the original report or the lowest QTc reported. Haloperidol doses administered were defined as cumulative dose at event, encompassing all doses administered during the hospital stay until the occurrence of the adverse cardiac event.
The drugs listed in the case reports were assessed for proarrhythmic potential using 2 references: the individual package insert and the website of the Arizona Center for Education and Research on Therapeutics.14
The drugs were only considered proarrhythmic when the 2 resources were in agreement.
Duplicates and/or previously published cases, as well as reports involving adverse cardiac effects not associated with QTP or TdP, were identified and excluded.
In their advisory, the FDA does not state the exact origin of the reports, their specific search strategy to identify haloperidol‐associated adverse events, or the role IV haloperidol played in the individual events included in the extended warning. Consequently, the number of events identified in this review may differ from that published in the FDA extended warning.
Results
A total of 70 reported cases of IV haloperidol associated TdP and/or QTP were identified. Of these 70, 41 were identified through the PubMed/EMBASE/Scopus review, while an additional 29 cases were identified through the FDA database search.
Of the 29 cases in the FDA database, 21 were reported by health care professionals and 8 by manufacturers.
A total of 35 publications described cases originating from the US. Three cases took place in Japan and 1 case each in Canada, Germany and Spain. Several cases in the MedWatch database were reported outside the US: 1 case each originated from Austria, Canada, France, Japan, Spain, Switzerland and the United Kingdom. A summary of the published case reports is displayed in Table 2 and the FDA cases are summarized in Table 3.
| Case | Source (reference#) | Date | Age, Years | Gender | Drugs Pro‐arrhyth. | Venti‐ lated | Max. Daily Dose (mg) | Total Dose at Event (mg) | Time to Event | Prolonged QT | QTc Maximal (baseline), msec | Change in QTc (msec) | TdP | ECG Normalization, Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
| 1 | 35 | 1991 | 56 | m | No | Yes | 1200 | 1540 | NR | Yes | 584 (400) | 184 | NR | NR, uneventful |
| 2 | 13 | 1992 | 36 | m | Yes | No | 11.5 | 11.5 | 20 hours after start | Yes | 714 (428) | 286 | Yes | QTc normalization (440 msec), NR |
| 3 | 38 | 1993 | 39 | f | Yes | Yes | NR | 580 | Max. QTc 72 hours after start | Yes | 650 (420) | 230 | Yes | QTc normalization after 6 days, uneventful |
| 4 | 38 | 1993 | 19 | f | Yes | No | 170 | 170 | Max. QT 12 hours after start | Yes | 600 (480) | 120 | Yes | QTc normalization after 8 days, uneventful |
| 5 | 38 | 1993 | 63 | f | Yes | No | NR | 489 | Max. QT 48 hours after start | Yes | 670 (520) | 150 | Yes | QTc normalization after 8 days, uneventful |
| 6 | 38 | 1993 | 74 | f | Yes | Yes | NR | 10 | NR | No | 430 (410) | 20 | Yes | QTc unchanged after 8 days, uneventful |
| 7 | 17 | 1993 | 39 | m | Yes | Yes | NR | >490 | NR | Yes | 457 (348) | 109 | Yes | QTc normalization within 2 to 3 days, no further TdP, NR |
| 8 | 17 | 1993 | 61 | m | Yes | Yes | 115 | 211 | NR | Yes | 500 (390) | 110 | NR | QTc normalization within 2 days, death |
| 9 | 17 | 1993 | 48 | m | Yes | Yes | 825 | 825 | NR | Yes | 538 (441) | 97 | NR | QTc normalization in 3 days, rehabilitation |
| 10 | 39 | 1994 | 23 | f | Yes | Yes | 120 | 300 | 12 hours after dose increase | Yes | NR (550) | NR | Yes | NR, uneventful, extubation after 5 days, discharge after 10 days |
| 11 | 39 | 1994 | 28 | m | Yes | Yes | 300 | >300 | 24 hours after dose increase | Yes | NR (>520) | NR | Yes | No recurrence of arrhythmia, patient death (multi‐organ failure) |
| 12 | 40 | 1994 | 65 | m | Yes | NR | 230 | 410 | Worsening from day 2 to day 5 | Yes | 594 (490) | 104 | Yes | QTc normalization (406 msec), no cardiac problems at discharge |
| 13 | 40 | 1994 | 65 | f | Yes | NR | 500 | 980 | After the last 60mg | Yes | 628 (403) | 225 | Yes | QTc normalization (400 msec), recurrence with oral haloperidol, rehabilitation |
| 14 | 40 | 1994 | 76 | f | Yes | NR | 21 | 26 | Day 2 after several boluses | Yes | 670 (450) | 220 | Yes | QTc normalization within several days (412 msec), rehabilitation |
| 15 | 41 | 1994 | 59 | m | NR | Yes | 865 | 1013 | NR | Yes | 640 (480) | 160 | NR | QTc normalization in 24 hours, survived |
| 16 | 16 | 1995 | 76 | f | Yes | No | NR | 44.5 plus 1 PO | 15 minutes | Yes | 670 (409) | 261 | Yes | ECG normalized the next morning, no further events |
| 17 | 16 | 1995 | 49 | m | Yes | No | NR | 1150 plus 20 IM | 45 minutes | Yes | 648 (380) | 268 | Yes | QTc normalization in 24 hours, anoxic brain insult/rehabilitation |
| 18 | 16 | 1995 | 65 | f | Yes | No | 600 | 965 | 30 minutes | Yes | 628 (403) | 225 | Yes | 3 more episodes of TdP in 3 hours, QTc normalization in 2 days, no recurrence with further haloperidol, NR |
| 19 | 42 | 1995 | 42 | m | Yes | No | 28 | 28 | 20 minutes | Yes | 610 (533) | 77 | Yes | QTc normalization in 5 days, uneventful, ECG normal |
| 20 | 42 | 1995 | 39 | m | Yes | No | 45 | 45 | 5 minutes | Yes | 654 (NR) | NR | Yes | QTc normalization after 24 hours, uneventful |
| 21 | 11 | 1997 | 56 | f | No | No | 10 | 10 | Shortly after | NR | NR (NR) | NR | Yes | TdP resolved after 8 hours, NR |
| 22 | 11 | 1997 | 82 | f | NR | No | 10 | 10 | Shortly after | Yes | 680 (NR) | NR | Yes | QTc normalization on day 6 after admission (470 msec), NR |
| 23 | 11 | 1997 | 35 | m | NR | No | NR | 90 | After 20 mg | Yes | 520 (NR) | NR | Yes | TdP disappeared 12 hours later, NR |
| 24 | 43,44 | 1998 | 45 | m | NR | Yes* | NR | 9 | 203 minutes | Yes | 638 (560) | 78 | Yes | NR, overall survival 100%, significantly prolonged hospital stay |
| 25 | 43,44 | 1998 | 64 | f | NR | NR | 115 | 220 minutes | Yes | 605 (424) | 181 | Yes | ||
| 26 | 43,44 | 1998 | 75 | f | NR | NR | 85 | 60 minutes | Yes | 567 (508) | 59 | Yes | ||
| 27 | 43,44 | 1998 | 71 | f | NR | NR | 55 | 120 minutes | Paced | Paced | Paced | Yes | ||
| 28 | 43,44 | 1998 | 58 | f | NR | NR | 75 | 38 minutes | Yes | 657 (542) | 115 | Yes | ||
| 29 | 43,44 | 1998 | 40 | m | NR | NR | 35 | 15 minutes | Yes | 679 (475) | 204 | Yes | ||
| 30 | 43,44 | 1998 | 71 | m | NR | NR | 70 | 58 minutes | Yes | 521 (478) | 43 | Yes | ||
| 31 | 43,44 | 1998 | 47 | m | NR | 400 | 400 | 79 minutes | Yes | 574 (444) | 130 | Yes | ||
| 32 | 21 | 1999 | 41 | f | Yes | Yes | 320 | 915 | 55 minutes | Yes | 610 (426) | 184 | Yes | QTc normalization after 5 day, uneventful |
| 33 | 21 | 1999 | 31 | m | Yes | Yes | 480 | 1700 | 40 minutes | Yes | 599 (491) | 108 | Yes | QTc normalized in 4 days, NR |
| 34 | 18 | 2000 | 64 | f | Yes | Yes | 175 | 175 | NR | No | 413 (418) | (‐5) | Yes | QTc remained unchanged, uneventful |
| 35 | 8 | 2000 | 75 | m | No | NR | >2 | >2 | NR | Yes | 615 (435) | 180 | No | QTc normalization in 48 hours, uneventful |
| 36 | 8 | 2000 | 68 | m | Yes | Yes | >2 | >2 | NR | Yes | 650 (407) | 243 | No | QTc normalization after 4 day, uneventful after extubation |
| 37 | 8 | 2000 | 77 | m | NR | NR | (4) | 2 | NR | Yes | 550 (393) | 157 | No | QTc normalization in 24 to 36 hours, NR |
| 38 | 12 | 2004 | 34 | m | Yes | NR | 24.5 | 24.5 | 20 minutes | Yes | 560 (420) | 140 | Yes | QTc normalization (440 msec), ECG normal |
| 39 | 23 | 2004 | 58 | f | Yes | NR | 340 | 1010 | NR | Yes | 533 (460) | 73 | Yes | QTc normalization 7 days later discharge after 27days |
| 40 | 45 | 2008 | 86 | f | Yes | No | 2 mg | 2 mg | 8 hours after last dose | Yes | 524 (NR) | Probably 79 | No | QTc normalization (445 msec), NR |
| 41 | 46 | 2009 | 74 | m | Yes | No | 2 | 2 | Shortly after | Yes | NR (579) | NR | Yes | Pre‐existing heart block and fibrillation resolved, nursing home/rehabilitation |
| Report | MedWatch Identifier | Report Date | Age, Years | Gender | Drugs Pro‐arrh. | Maximum Daily Dose (mg) | Total Dose at Event (mg) | Prolonged QT | QTc Maximal (baseline), msec | Change in QTc (msec) | TdP | Outcome; Recovery |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||
| 1 | 3122988‐1 | 1998 | 61 | m | No | 48 | 48 | Yes | NR | NR | Yes | Intervention; NR |
| 2 | 3157827‐6 | 1998 | 44 | f | No | 160 | 160 | Yes | 550 (440) | 110 | Yes | Intervention; uneventful |
| 3 | 3178715‐5 | 1999 | 60 | m | NR | 415 | 645 | Yes | NR | NR | Yes | Life‐threatening; QTc normalization in 1 day, no recurrence |
| 4 | 3271261‐X | 1999 | 56 | m | NR | NR | 20 | Yes | NR | NR | Yes | Life‐threatening; QTc normalization |
| 5 | 3271080‐4 | 1999 | 35 | m | Yes | 7 | 7 | NR | NR | NR | Yes | NR; event abated after dose stopped/reduced, hospitalization prolonged |
| 6 | 3325391‐4 | 1999 | 55 | f | Yes | 75 | 75 | NR | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 7 | 3381921‐8 | 1999 | 52 | m | No | 320 | 634 | Yes | 458 (430) | 28 | Yes | Death; NA |
| 8 | 3483869‐7 | 2000 | 18 | m | No | >200 | >310 | Yes | NR | NR | Yes | Intervention; no recurrence after haloperidol reinstitution |
| 9 | 3516342‐8 | 2000 | NR | NR | NR | NR | NR | NR | NR | NR | Yes | NR; NR |
| 10 | 3516320‐9 | 2000 | 34 | m | Yes | 5 | 5 | Yes | NR | NR | No | Life‐threatening; event abated after dose stopped |
| 11 | 3552263‐2 | 2000 | 46 | f | Yes | NR | 97.5 | Yes | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 12 | 3574705‐9 | 2000 | 78 | m | Yes | NR | 160 | Yes | 603 (453) | 50 | Yes | Intervention; event abated after dose stopped/reduced |
| 13 | 3703871‐7 | 2001 | 27 | m | NR | 530 | 530 | Yes | NR | NR | Yes | Death, NA |
| 14 | 3724567‐1 | 2001 | 31 | m | Yes | 6 | 6 | Yes | 496 (449) | 47 | No | Life‐threatening; ECG returned to baseline |
| 15 | 3851984‐1 | 2002 | 72 | f | NR | 18 | 18 | NR | NR | NR | Yes | Hospitalization; NR |
| 16 | 3942407‐2 | 2002 | 51 | m | Yes | 14 | 14 | Yes | 461 (444) | 17 | Yes | Life‐threatening; no recurrence |
| 17 | 4066580‐3 | 2003 | >60 | f | NR | 50 | 50 | Yes | >600 (480) | >120 | No | Hospitalization; QTc normalization, patient recovered |
| 18 | 4126280‐8 | 2003 | 47 | f | NR | 60 | 180 | Yes | 550 (450) | 100 | No (bradycardia) | Hospitalization; patient recovered |
| 19 | 4150700‐6 | 2003 | NR | m | NR | 5 | 5 | NR | NR | NR | Yes | NR; event abated after dose stopped/reduced |
| 20 | 4340092‐1 | 2004 | 52 | m | Yes | 5 | 5 | Yes | >500 (490) | >10 | NR (polymorphous VT) | Life‐threatening; NR |
| 21 | 4714692‐0 | 2005 | NR | m | NR | NR | NR | Yes | NR | NR | Yes | Hospitalization; event abated after dose stopped/reduced |
| 22 | 4881813‐9 | 2006 | NR | m | NR | NR | 40 | NR | NR | NR | Yes | Hospitalization; event abated after dose stopped/reduced |
| 23 | 4892225‐6 | 2006 | NR | f | Yes | 10 | >10 | Yes | 493 (300) | 193 | No | Hospitalization; QTc normalization (403 msec) |
| 24 | 4911873‐8 | 2006 | 69 | m | Yes | 6 | 6 | NR | NR | NR | Yes | Cardiac arrest, death; NA |
| 25 | 5366448‐6 | 2007 | 53 | m | Yes | NR | 35 | Yes | NR | NR | NR | Cardiac arrest, life‐threatening; patient recovered |
| 26 | 5563440‐3 | 2007 | 58 | m | Possible | 5 | 5 | Yes | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 27 | 5642929‐2 | 2008 | 42 | m | Yes | 165 | 165 | Yes | 640 (350) | 290 | Yes | Death; NA |
| 28 | 5697758‐0 | 2008 | 38 | m | Yes | NR | 620 | NR | NR | NR | Yes | Hospitalization; patient recovered |
| 29 | 5254840‐X | 2008 | 19 | f | Possible | 15 | 25 | Yes | 461 | NR | NR | Cardiac arrest, hospitalization; patient recovered |
Of the 70 cases, 54 cases of TdP were reported. The remaining 16 of 70 cases involved cases of QTP, 9 of which did not progress to TdP and 7 of which the progression to TdP was unclear. Of note, 42 of 54 of the cases of TdP were reported as preceded by documented QTP. Presence of QTP was unknown in the other 12 original reports. Three out of 70 patients experienced sudden cardiac arrest, 1 of which was fatal. One arrest was preceded by TdP and 2 by QTP (Figure 1).
The patient ages ranged from 18 years to 86 years. Of note, 17 patients experiencing TdP and/or QTP were 40 years old, and 2 of those patients were 30 years old.
Haloperidol‐associated QTP and/or TdP were observed in 27 female and 42 male patients; the gender was not stated in one report. Of the 54 patients experiencing TdP (with or without report of previous QTP), 22 were female and 31 were male (1 gender unknown).
A total of 68 of 70 patients were determined to have associated risk factors15 for QTP/TdP (see Table 4). The circumstances of the remaining 2 patients were not described in sufficient detail to identify associated risk factors.
| Risk Factor | Patients, n (%) |
|---|---|
| |
| Any risk factor | 68/70 (97) |
| Unknown | 2/70 (3) |
| Specific risk factors | |
| Electrolyte imbalance | 27/68 (40) |
| Underlying cardiac disease | 32/68 (47) |
| Concomitant proarrhythmic agents | 39/68 (57) |
| Other drugs influencing cardiac function | 23/68 (34) |
| Baseline QTc >450 msec | 18/68 (26) |
| QTc known: 44 patients | 18/44 (41) |
Overall, 32 patients had underlying heart conditions. Electrolyte imbalances, including hypokalemia, hypomagnesemia, and hypocalcemia, were present in 17 patients. At least 39 patients were receiving potentially proarrhythmic agents (1‐8 proarrhythmic drugs per patient) in addition to IV haloperidol. At least 23 patients were receiving additional drugs with a potential for other cardiac adverse events than QTP and TdP.
A wide range of other disease states previously reported to be associated with QTP15 were identified in these patients: asthma (5 patients), diabetes (5 patients), obesity (3 patients), impaired renal and/or liver function (3 patients each), human immunodeficiency virus (HIV) (2 patients); chronic obstructive pulmonary disease (COPD), pancreatitis and hypothyroidism (1 patient each). A total of 22 patients had a history of substance abuse (alcohol and/or drugs), and 4 patients were smokers.
The administered doses of IV haloperidol varied widely. Considering that information regarding the maximal daily dose was missing in 22 reports and ambiguous in another 20 cases, the results have been presented using cumulative IV haloperidol doses. Patients experiencing TdP without preceding QTP received a cumulative dose (= total dose at event) ranging from 5 mg to 645 mg. Patients with both confirmed QTP and TdP were administered a cumulative dose of 2 mg to 1700 mg. Patients who experienced QTP without TdP received a cumulative dose of 2 mg to 1540 mg of IV haloperidol.
Sudden cardiac arrest following administration of IV haloperidol was observed in cumulative doses ranging from 6 mg to 35 mg. The cardiac arrest leading to a fatal outcome was preceded by an administration of at least 6 mg of IV haloperidol. Overall, 14 out of 70 patients received cumulative doses of 10 mg IV haloperidol.
The time from administration to documentation of QTP and/or TdP ranged from immediately post administration to 8 hours after administration of the last dose of IV haloperidol.
Baseline QTc was known in 44 patients. Baseline QTc was >450 msec in 18 of these 44 patients.
The change from baseline QTc varied widely from 20 msec to 286 msec; 36 patients demonstrated a prolongation of >50 msec.
In those patients with reported haloperidol‐associated QTP, 25 patients demonstrated a QTc >600 msec and 38 patients >520 msec.9 Of the cases with known specific QTc values, the QTc was prolonged >450 msec in 48 out of 50 cases. The lowest reported QTc leading to TdP was 413 msec.
A total of 20 patients were reported as having a normalization of QTc (as defined by the original reports) within several hours to 8 days; minimal QTP was reported as persisting in 2 patients. The specifics of the other patients were unknown, although 25 patients were categorized as recovered, 13 were stated as having an uneventful remainder of hospitalization, and 5 patients were discharged to a rehabilitation facility or a nursing home.
Discussion
The current review was performed in response to the FDA warning recommending the use of continuous ECG monitoring associated with the administration of intravenous haloperiodol.5 This warning has resulted in substantial dilemmas for health care organizations, additional resource allocation, and increased scrutiny from regulatory agencies. The results of our review reveal that intravenous haloperidol‐associated QTP and TdP almost uniformly take place in patients with concomitant risk factors and with cumulative doses 2 mg. In light of these findings, it is possible that hospitals may be able to administer intravenous haloperidol in patients without risk factors without continuous ECG monitoring. In reviewing these published reports, it is important to note that the FDA identified 28 published cases of haloperidol‐associated QTP and TdP, while our review yielded a total of 41 published case reports.
The FDA database included 73 cases of haloperidol‐associated TdP in their database. However, these cases included both oral as well as IV administration; using our methodology, we identified 29 additional case reports associated with intravenous haloperidol from this database. Consequently, our review included 41 published case reports and 29 FDA database cases, resulting in the total of 70 patients.
Our review revealed a number of practical findings. First, our summary demonstrated that neither QTP nor TdP has been documented with a cumulative dose of IV haloperidol of 2 mg. The majority of patients (80%) received cumulative IV doses 10 mg. The lowest dose associated with sudden cardiac arrest was 6 mg and this took place in a 69‐year‐old male patient. Second, the majority (97%) of our patients had additional risk factors for QTP and/or TdP. Pre‐existing heart disease,1619 electrolyte imbalance,17, 1921 concomitant proarrhythmic drugs16, 17, 1922 and mechanical ventilation17, 23 were identified as the most commonly observed risk factors (Table 4). Lastly, in those cases in which the data were reported, baseline QTc was >450 msec in 41% of the patients, and 96% had a QTc at the time of the event >450 msec. Therefore, we conclude that patients: (1) receiving low cumulative doses (2 mg) with (2) no risk factors for prolonged QTc or TdP, and (3) with a normal QTc on baseline EKG can safely be given IV haloperidol in the hospital setting.
This dosage range is consistent with the labelling for IV haloperidol dosing in Canada24 and Germany25 (Table 1), where single doses of 0.25 mg to 1.5 mg are recommended for the treatment of delirium or acute agitation in the geriatric population.24, 25
In a recent Cochrane review, low‐dose IV haloperidol (3 mg per day) was concluded to be as safe and effective as atypical antipsychotics in the treatment of acute delirium with respect to extrapyramidal adverse effects.2
The American Psychiatric Association recommends an initial IV dose of 12 mg every 24 hours as needed (0.250.50 mg every 4 hours as needed for elderly patients), with titration to higher doses for patients who continue to be agitated for the treatment of patients with delirium (issued 1999, updated 2004).3
While several expert‐groups and investigators currently consider IV haloperidol as an important therapeutic option for treating acute delirium and agitation in the dose range presented above, less consensus exists regarding monitoring requirements.2, 3, 26, 27
The American Psychiatric Association recommends IV haloperidol only after a baseline ECG is obtained. These guidelines have not been updated since the release of the FDA extended warning.3 In their recent review, Morandi et al.28 support the dosage recommendation of the 1999 American Psychiatric Association's practice guidelines for treatment of delirium,3 ie, administration of IV haloperidol in single doses of 0.5 mg to 2 mg in elderly patients, however, only after a baseline ECG is obtained.28 While the package insert of IV haloperidol in France29 recommends a baseline ECG, Germany,25 Italy30 and Switzerland's31 package information states the need for regular ECG monitoring. Guidelines for the treatment of delirium in the intensive care unit published by the American College of Critical Care Medicine and the Society of Critical Care Medicine in collaboration with the American Society of Health‐System Pharmacists consider IV haloperidol as the preferred agent for the treatment of delirium in critically ill patients (grade of recommendation = C). These expert groups recommend that patients should be monitored for electrocardiographic changes (QT interval prolongation and arrhythmias) when receiving haloperidol (Grade of recommendation = B).32
Nevertheless, continuous ECG monitoring (ie, telemetry) is expensive, labor‐intensive and, potentially overutilized.33, 34 Requiring clinicians to place all patients receiving intravenous haloperidol on telemetry is impractical and potentially costly. Mandating telemetry could also lead to unintended harm, ie, use of a less effective or less safe drug to avoid compliance with the telemetry mandate.
Based on our findings and the current recommendations in the literature, inpatient providers should be thoughtful and deliberate in the use of haloperidol to treat acute delirium with agitation. Patients requiring pharmacologic management of their delirium should be screened for risk factors for QTP and TdP (Table 4) and a baseline ECG should be obtained prior to haloperidol administration. If significant risk factors exist or the baseline ECG reveals a prolonged QTc, then the patient should receive continuous ECG monitoring. Similarly, if cumulative doses of 2 mg are needed, the patient should be placed on telemetry.
There are some limitations to our study design. Our findings are based upon previously published case reports or data submitted to the FDA MedWatch. While the content of the FDA's MedWatch database is accessible to the public via the Freedom of Information Act (FOIA), the events are neither categorized nor peer‐reviewed upon entry into the database. Consequently, information may be incomplete or inaccurate. In addition, the denominator representing the overall use of IV haloperidol is unknown, thus a rate of event cannot be assigned and the true scope of the problem cannot be determined. Despite these limitations, this summary represents the most comprehensive review of the literature to date, expanding on the analysis performed by the FDA. Of note, in our review of the FDA database, we noted several cases of haloperidol‐associated QTP or TdP associated with other routes of administration. Thus, it is unknown whether this complication is any greater with IV vs. the IM or per os (PO) routes of administration.
Conclusion
Although the proarrhythmic potential of haloperidol and other antipsychotics has been well established in the literature, IV haloperidol has been considered relatively safe with respect to this complication from the time of its approval in 1967.5, 1722, 35, 36 In reviewing all reported cases of cardiac complications associated with IV haloperidol, as well as the current literature, an association with QTP and TdP is likely. However, the case reports reveal that QTP and TdP generally occur in the setting of concomitant risk factors, and no cases have been reported utilizing a cumulative IV dose of 2 mg. It may therefore be safe to administer a cumulative dose of IV haloperidol of 2 mg without ECG monitoring in patients without risk factors for QTP. However, ECG monitoring should take place with IV haloperidol doses 2 mg and/or in those patients with additional risk factors of developing QTP and/or TdP.
Based on the findings of this review complemented by the guidelines of various expert‐groups and the official labelling information of different countries, the Pharmacy & Therapeutics Committee of the UCSF Medical Center revised the IV haloperidol policy: administration of a total dose of 2 mg IV haloperidol without concurrent telemetry is allowed in a noncritical care setting in patients without risk factors for QTP and TdP.
Acknowledgements
The authors acknowledge Gloria Won of the Fishbon Library at UCSF Medical Center at Mount Zion for her support.
- Haldol® injection (for immediate release) Package Insert.Raritan, NJ:Ortho‐McNeil Pharmaceutical Inc.;2005;rev. 05.2007.
- ,,,. Antipsychotics for delirium (review), the Cochrane collaboration2008;2. Available at: www. cochrane.org. Accessed February 2010.
- American Psychiatric Association: practice guideline for the treatment of patients with delirium.Am J Psychiatry.1999;156(5 suppl):1–20, updated 2004.
- Thomson Micromedex.2008. Micromedex healthcare series: “haloperidol” Thomson Micromedex, Greenwood Village.
- FDA alert: haloperidol (marketed as Haldol, Haldol Decanoate and Haldol Lactate).2007. This alert highlights revisions to the labeling for haloperidol. Available at: www.fda.gov. Accessed February 2010.
- ,,.Use of high‐dose intravenous haloperidol in the treatment of agitated cardiac patients,J Clin Psychopharmacol.1985;5(6):344–347.
- ,,, et al.Postoperative delirium,Am J Psychiatry.2008;165:7.
- ,.Corrected QT interval prolongation associated with intravenous haloperidol in acute coronary syndromes,Catheter Cardiovasc Interv.2000;50(3):352–355.
- ,.Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death.Am J Psych.2001;158(11):1774–1782.
- ,,, et al.Accuracy of uncorrected versus corrected QT Interval for Prediction of torsade de pointes associated with intravenous haloperidol.Pharmacotherapy.2007;27(2):175–182.
- ,,.Three cases of ventricular tachycardia and torsades de pointes induced by antipsychotic drugs.Shinzo.1997;29(1):68–74.
- ,,,.Haloperidol por via intravenosa y torsade de pointes.Medicina intensive.2004;28(2):89.
- ,,, et al.QT‐Verlängerung und Kammerflimmern unter Haloperidol‐ und Clonidin‐Therapie des Alkoholentzugssyndroms.Intensivmedizin und Notfallmedizin.1992;29(4):178–183.
- ARIZONA CERT, Arizona Center for Education and Research on Therapeutics. Available at: www.azcert.org. Accessed February 2010.
- ,.Cardiac arrhythmias—a clinical approach.Edinburgh:Mosby;2003.
- ,.The association between intravenous haloperidol and torsades de pointes—three cases and a literature review.Psychosomatics.1995;36:541–549.
- ,.Prolongation of the corrected QT and torsades de pointes cardiac arrhythmia associated with intravenous haloperidol in the medically ill.J Clin Psychopharmacol.1993;13(2):128–132.
- ,,,,.Torsades de pointes secondary to intravenous haloperidol after coronary bypass grafting surgery.Can J Anesth.2000;47(3):251–254.
- ,.Torsade de pointes associated with the administration of intravenous haloperidol: a review of the literature and practical guidelines for use,Expert Opin Drug Saf.2003;2(6):543–547.
- ,.Conduction disturbances associated with administration of butyrophenone antipsychotics in the critically ill: a review of the literature,Pharmacotherapy.1997;17(3):531–537.
- ,,.Haloperidol‐induced torsades de pointes.Ann Pharacother.1999;33(10):1046–1050.
- ,,, et al.Practice parameters for intravenous analgesia and sedation for adult patients in the intensive care unit: an executive summary,Crit Care Med.1995;23(9):1596–1600.
- ,,,,,.Prolonged cardiac repolarization after tacrolimus and haloperidol administration in the critically ill patient.Pharmacotherapy.2004;24(3):404–408.
- CPS Compendium of Pharmaceuticals and Specialties, the Canadian drug reference for health professionals, 2007, Canadian pharmacists association.
- Rote Liste Deutschland2008, Rote Liste Service GmbH Frankfurt am Main. Available at: www.rote‐liste.de. Accessed February 2010.
- ,.Delirium in the hospitalized patient: a primer for the pharmacist clinician.J Pharm Pract.2007;20(5):368–372.
- ,,, et al.Delirium: guidelines for general hospitals.J Psychosom Res.2007;62(3):371–383.
- ,,,.The pharmacological management of delirium in critical illness.Current Drug Therapy.2008,3:148–157.
- VIDAL‐l'information sur les produits de santé2008, Issy les Moulineaux Cedex. Available at: www.vidal.fr. Accessed February 2010.
- Haldol iniettabile—ufficiale monografia italiana. Available at: www. informatorefarmaceutico.it. Accessed February 2010.
- Arzneimittelkompendium der Schweiz2008, documed Verlag Basel. Availabla at: www.kompendium.ch. Accessed February 2010.
- ,,, et al.Clinical practice guidelines for the sustained use of sedatives and analgesics in the critically ill, American College of Critical Care Medicine ACCM, Society of Critical Care Medicine SCCM, American Society of Health‐System Pharmacists ASHP.Crit Care Med.2002;30(1):119–141.
- ,,,,.Is telemetry overused? Is it as helpful as thought?Cleve Clin J Med.2009;76(6):368–372.
- ,,.Telemetry outside critical care units: patterns of utilization and influence on management decisions.Clin Cardiol.1998;21(7):503–505.
- ,,.High‐dose intravenous haloperidol for agitated delirium in a cardiac patient on intra‐aortic balloon pump.J Clin Psychopharmacol.1991;11(2):146–147.
- .Haloperidol, midazolam and intravenous sedation.Aust NZ J Psychiatry.1999;33(6):942–943.
- BNF British National Formulary, compendium of pharmaceuticals and specialties of the UK.2007. Available at: www.bnf.org. Accessed February 2010.
- ,,,.Torsade de pointes associated with the use of intravenous haloperidol.Ann Intern Med.1993;119(5):391–394.
- ,.Torsades de pointes: potential consequence of intravenous haloperidol in the intensive care unit.Intensive Care World.1994;11(3):109–112.
- ,.Torsade de pointes caused by high‐dose intravenous haloperidol in cardiac patients.Clin Cardiol.1995;18:285–290.
- ,,.Continuous infusion of haloperidol controls agitation in critically ill patients.Crit Care Med.1994:22(3):433–440.
- ,,.Torsade de pointes complicating the treatment of bleeding esophageal varices: association with neuroleptics, vasopressin, and electrolyte imbalances.Am J Gastroenterol.1995;90(5):822–824.
- ,,,.Torsades de pointes associated with intravenous haloperidol in critically ill patients.Am J Cardiol.1998;81(2):238–240.
- ,,,,.The effect of intravenous haloperidol on QT interval dispersion in critically ill patients: comparison with QT interval prolongation for assessment of risk of torsades de pointes.J Clin Pharmacol.2001;41:1310–1318.
- ,,.Low‐dose haloperidol associated QTc prolongation.J Am Geriatr Soc.2008;56(10):1963–1964.
- ,,.Torsade de pointes following intravenous haloperidol administration in a patient with complete heart block.WMJ.2009;108(1):48–50.
Haloperidol is Food and Drug Administration (FDA)‐approved in the United States for the management of acute and chronic psychotic disorders and widely used in the management of delirium‐associated agitation in hospitalized patients.1 Delirium in the hospital is an acute confusional state that frequently arises from multiple complex factors and may affect up to 30% of hospitalized patients.2 Although the first step in the management of delirium involves identification and treatment of underlying causes and offering supportive behavioral care; medications may be needed to control severe agitation.2 Low dose intravenous (IV) haloperidol (ie, 0.250.5 mg every 4 hours) is a commonly used medication in this setting as recommended by expert‐groups including the Cochrane Collaboration and the American Psychiatric Association.2, 3
Although injectable haloperidol, a butyrophenone‐derived antipsychotic agent pharmacologically related to the piperazine phenothiazines,4 is approved for IV use in many countries (Table 1), parenteral use is approved only for intramuscular (IM) administration in the US. Thus, IV administration of the drug in the US is considered an off‐label use.5
| Indication | Country | |||||
|---|---|---|---|---|---|---|
| Canada24 | France29 | Germany25 | Great Britain37 | Italy30 | Switzerland31 | |
| ||||||
| Mainly delirium (schizophrenia, other psychosis, short‐term management of psychomotor agitation, excitement, violent or dangerously impulsive behavior, vomiting, hiccup) | Short term treatment of agitation and aggressiveness during an acute or chronic psychotic episode, vomiting along with antimitotic post‐radiotherapy treatment | Acute and chronic schizophrenia, psycho‐motorical agitation of psychotic genesis | Schizophrenia, other psychosis, short‐term adjunctive management if psychomotor agitation, violent or dangerous impulsive behavior | Resistant forms of psycho‐motorical excitement, acute delirious and/or hallucinatory psychosis' chronic psychosis High doses restrictions: syndrome of psycho‐motorical excitement, acute delirious and/or hallucinatory psychosis, chronic psychosis | Acute schizophrenic episode, mania, vomiting | |
| IV dosing in adults | 12 mg every 24 hours | The use is limited to adult patients and the drug can be administered IM or IV. The IV route is restricted to the treatment of vomiting. | 510 mg/day, daily max.: 30(100) mg | 210 mg initially, PRN every 48 hours, daily max. 18 mg | 510 mg initially, PRN every hour, daily max. 60 mg | 5 mg PRN every 30 minutes |
| IV dosing in geriatric care | 0.250.5 mg | Single dose of 0.51.5 mg, daily max. 5 mg | Half adult dose | Adjust to appropriate dose | 0.5 mg, than PRN | |
| Risk factors for the development of cardiac adverse events | QT prolonging drugs, diabetes, obesity, hypokalemia, congenital long QT syndrome | Bradycardia 55 beats per minute, hypokalemia, congenital QT prolongation, other medications provoking bradycardia, deceleration of the intra‐cardiac transition or prolonged QT interval | QT syndrome, hypokalemia, other electrolyte imbalance, cardiovascular diseases, QT prolongation in the family history | Cardiovalscular disease, drugs that can prolong the QTc, diabetes, obesity, hypokalemia, congenital long QT syndrome | Contraindications: recent cardiac infarction, uncompensated cardiac insufficiency, cardiac arrhythmias, antiarrhythmic drugs, pre‐existing QT prolongation, cases of arrhythmia or torsades de pointes in the family history, untreated potassium imbalance, QTc prolonging drugs | QT syndrome, hypokalemia, hypomagnesemia, other electrolyte imbalances, cardiovascular diseases, hypothyreosis, QT prolongation in the family history |
| Monitoring recommendations | Electrolytes | ECG monitoring at admission time, electrolytes | ECG monitoring, electrolytes | Metabolic parameters | ECG at baseline and regular ECG monitoring, electrolytes | Close ECG monitoring, electrolytes |
| General recommendations | Regular reevaluation in long‐term use | Apply the lowest effective dose | Apply the lowest effective dose | Application per mouth is the route of choice | Decrease dose if QTc >500 msec | Switch to PO as soon as possible |
Haloperidol is often preferred over other antipsychotics as a result of its effectiveness, low rate of anticholinergic side effects, familiarity with dosing and usage, and minimal respiratory or sedative properties.6 Use of the IV route in patients with acute delirium has several advantages over the IM or oral route,7 including rapid onset, immediate bioavailability, and ease and safety of administration.
Prior to September 2007, the package insert for haloperidol alerted healthcare professionals to the risk of cardiovascular side effects. Based on case reports of potentially fatal cardiac events, the FDA revised the label, warning that the QT prolongation (QTP) and risk of torsades de pointes (TdP) were increased with IV administration of haloperidol or administration of the drug at greater than recommended doses. Unfortunately, neither the typical dosing range nor the minimum dose associated with these cardiac side effects were specified in this recommendation.5
It is well‐established that haloperidol may prolong the QT interval by blocking the repolarizing potassium IKr current.8 Although drugs that block the IKr channel can produce arrhythmia in healthy individuals, additional risk factors, such as underlying heart conditions, electrolyte imbalances (ie, hypokalemia and hypomagnesemia), concomitant proarrhythmic drug use, and mechanical ventilation may increase this risk.9 Prolongation of the QT interval has been associated with subsequent malignant cardiac arrhythmias including ventricular fibrillation and TdP.10 Prolongation of the QT interval is considered the strongest risk factor for TdP, particularly with a baseline QTc > 450 msec.9
Based on the increased risk for QTP and TdP and the case reports of cardiac events, the FDA advisory recommended continuous electrocardiogram (ECG) monitoring in patients receiving IV haloperidol.5 However, such monitoring may be impractical and costly in hospitalized patients who require low doses of IV haloperidol to manage acute delirium and who are not in telemetry or intensive care units.
The aim of this review was to evaluate the case reports leading to the recent FDA warning for IV haloperidol, specifically focusing on the presence of risk factors for arrhythmias. Based upon the evidence, an additional aim was to provide an institutional response to this warning toward the optimal use of this agent.
Method
Two search pathways were used to evaluate reports of haloperidol‐associated TdP and/or QT prolongation:
Literature Review
We searched for published literature in humans indexed in Pubmed (1966April 2009), EMBASE (1972April 2009), and Scopus (1823April 2009) using the search terms haloperidol or Haldol combined with intravenous or infusion and at least one of the following terms: QT prolongation, TdP, torsades de pointes, torsades with a specific focus on case reports.
References from the retrieved articles were also reviewed to search for additional case reports.
In addition to cases reported in English journals, several of our reports originated from Japan11 (translation provided by the FDA), Spain12 and Germany13 (translated by the primary author).
Search of the FDA Database
We reviewed all adverse drug events reported through MedWatch or those submitted by the manufacturer from November 1997 to April 2008 through the Freedom of Information Act (FOIA) request. The FDA provided a full‐text summary of 5944 reports involving oral, intramuscular and IV use of haloperidol. The FDA data were transferred to a Microsoft Access database and screened for the key terms torsade, QT, prolongation, wave. Incident report number, date of report, age, gender, origin of report, medication name, role of drug as categorized by the FDA (suspect, concomitant, primary suspect, secondary suspect), route, dose, units, duration, symptoms and FDA outcome category (death, life‐threatening, hospitalization initial or prolonged, disability, congenital anomaly, required intervention to prevent permanent damage, other) were recorded. Only those reports in which IV haloperidol was considered by the reporter to be the primary causative agent for the adverse event were reviewed. Available information included diagnosis, laboratory parameters, QTc measurement, cardiac symptoms, outcomes and a description of recovery. No peer review was applied to the MedWatch reports and the data reported in this publication reflect the original information from the FDA MedWatch database. Baseline QTc was either the value defined as such in the original report or the lowest QTc reported. Haloperidol doses administered were defined as cumulative dose at event, encompassing all doses administered during the hospital stay until the occurrence of the adverse cardiac event.
The drugs listed in the case reports were assessed for proarrhythmic potential using 2 references: the individual package insert and the website of the Arizona Center for Education and Research on Therapeutics.14
The drugs were only considered proarrhythmic when the 2 resources were in agreement.
Duplicates and/or previously published cases, as well as reports involving adverse cardiac effects not associated with QTP or TdP, were identified and excluded.
In their advisory, the FDA does not state the exact origin of the reports, their specific search strategy to identify haloperidol‐associated adverse events, or the role IV haloperidol played in the individual events included in the extended warning. Consequently, the number of events identified in this review may differ from that published in the FDA extended warning.
Results
A total of 70 reported cases of IV haloperidol associated TdP and/or QTP were identified. Of these 70, 41 were identified through the PubMed/EMBASE/Scopus review, while an additional 29 cases were identified through the FDA database search.
Of the 29 cases in the FDA database, 21 were reported by health care professionals and 8 by manufacturers.
A total of 35 publications described cases originating from the US. Three cases took place in Japan and 1 case each in Canada, Germany and Spain. Several cases in the MedWatch database were reported outside the US: 1 case each originated from Austria, Canada, France, Japan, Spain, Switzerland and the United Kingdom. A summary of the published case reports is displayed in Table 2 and the FDA cases are summarized in Table 3.
| Case | Source (reference#) | Date | Age, Years | Gender | Drugs Pro‐arrhyth. | Venti‐ lated | Max. Daily Dose (mg) | Total Dose at Event (mg) | Time to Event | Prolonged QT | QTc Maximal (baseline), msec | Change in QTc (msec) | TdP | ECG Normalization, Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
| 1 | 35 | 1991 | 56 | m | No | Yes | 1200 | 1540 | NR | Yes | 584 (400) | 184 | NR | NR, uneventful |
| 2 | 13 | 1992 | 36 | m | Yes | No | 11.5 | 11.5 | 20 hours after start | Yes | 714 (428) | 286 | Yes | QTc normalization (440 msec), NR |
| 3 | 38 | 1993 | 39 | f | Yes | Yes | NR | 580 | Max. QTc 72 hours after start | Yes | 650 (420) | 230 | Yes | QTc normalization after 6 days, uneventful |
| 4 | 38 | 1993 | 19 | f | Yes | No | 170 | 170 | Max. QT 12 hours after start | Yes | 600 (480) | 120 | Yes | QTc normalization after 8 days, uneventful |
| 5 | 38 | 1993 | 63 | f | Yes | No | NR | 489 | Max. QT 48 hours after start | Yes | 670 (520) | 150 | Yes | QTc normalization after 8 days, uneventful |
| 6 | 38 | 1993 | 74 | f | Yes | Yes | NR | 10 | NR | No | 430 (410) | 20 | Yes | QTc unchanged after 8 days, uneventful |
| 7 | 17 | 1993 | 39 | m | Yes | Yes | NR | >490 | NR | Yes | 457 (348) | 109 | Yes | QTc normalization within 2 to 3 days, no further TdP, NR |
| 8 | 17 | 1993 | 61 | m | Yes | Yes | 115 | 211 | NR | Yes | 500 (390) | 110 | NR | QTc normalization within 2 days, death |
| 9 | 17 | 1993 | 48 | m | Yes | Yes | 825 | 825 | NR | Yes | 538 (441) | 97 | NR | QTc normalization in 3 days, rehabilitation |
| 10 | 39 | 1994 | 23 | f | Yes | Yes | 120 | 300 | 12 hours after dose increase | Yes | NR (550) | NR | Yes | NR, uneventful, extubation after 5 days, discharge after 10 days |
| 11 | 39 | 1994 | 28 | m | Yes | Yes | 300 | >300 | 24 hours after dose increase | Yes | NR (>520) | NR | Yes | No recurrence of arrhythmia, patient death (multi‐organ failure) |
| 12 | 40 | 1994 | 65 | m | Yes | NR | 230 | 410 | Worsening from day 2 to day 5 | Yes | 594 (490) | 104 | Yes | QTc normalization (406 msec), no cardiac problems at discharge |
| 13 | 40 | 1994 | 65 | f | Yes | NR | 500 | 980 | After the last 60mg | Yes | 628 (403) | 225 | Yes | QTc normalization (400 msec), recurrence with oral haloperidol, rehabilitation |
| 14 | 40 | 1994 | 76 | f | Yes | NR | 21 | 26 | Day 2 after several boluses | Yes | 670 (450) | 220 | Yes | QTc normalization within several days (412 msec), rehabilitation |
| 15 | 41 | 1994 | 59 | m | NR | Yes | 865 | 1013 | NR | Yes | 640 (480) | 160 | NR | QTc normalization in 24 hours, survived |
| 16 | 16 | 1995 | 76 | f | Yes | No | NR | 44.5 plus 1 PO | 15 minutes | Yes | 670 (409) | 261 | Yes | ECG normalized the next morning, no further events |
| 17 | 16 | 1995 | 49 | m | Yes | No | NR | 1150 plus 20 IM | 45 minutes | Yes | 648 (380) | 268 | Yes | QTc normalization in 24 hours, anoxic brain insult/rehabilitation |
| 18 | 16 | 1995 | 65 | f | Yes | No | 600 | 965 | 30 minutes | Yes | 628 (403) | 225 | Yes | 3 more episodes of TdP in 3 hours, QTc normalization in 2 days, no recurrence with further haloperidol, NR |
| 19 | 42 | 1995 | 42 | m | Yes | No | 28 | 28 | 20 minutes | Yes | 610 (533) | 77 | Yes | QTc normalization in 5 days, uneventful, ECG normal |
| 20 | 42 | 1995 | 39 | m | Yes | No | 45 | 45 | 5 minutes | Yes | 654 (NR) | NR | Yes | QTc normalization after 24 hours, uneventful |
| 21 | 11 | 1997 | 56 | f | No | No | 10 | 10 | Shortly after | NR | NR (NR) | NR | Yes | TdP resolved after 8 hours, NR |
| 22 | 11 | 1997 | 82 | f | NR | No | 10 | 10 | Shortly after | Yes | 680 (NR) | NR | Yes | QTc normalization on day 6 after admission (470 msec), NR |
| 23 | 11 | 1997 | 35 | m | NR | No | NR | 90 | After 20 mg | Yes | 520 (NR) | NR | Yes | TdP disappeared 12 hours later, NR |
| 24 | 43,44 | 1998 | 45 | m | NR | Yes* | NR | 9 | 203 minutes | Yes | 638 (560) | 78 | Yes | NR, overall survival 100%, significantly prolonged hospital stay |
| 25 | 43,44 | 1998 | 64 | f | NR | NR | 115 | 220 minutes | Yes | 605 (424) | 181 | Yes | ||
| 26 | 43,44 | 1998 | 75 | f | NR | NR | 85 | 60 minutes | Yes | 567 (508) | 59 | Yes | ||
| 27 | 43,44 | 1998 | 71 | f | NR | NR | 55 | 120 minutes | Paced | Paced | Paced | Yes | ||
| 28 | 43,44 | 1998 | 58 | f | NR | NR | 75 | 38 minutes | Yes | 657 (542) | 115 | Yes | ||
| 29 | 43,44 | 1998 | 40 | m | NR | NR | 35 | 15 minutes | Yes | 679 (475) | 204 | Yes | ||
| 30 | 43,44 | 1998 | 71 | m | NR | NR | 70 | 58 minutes | Yes | 521 (478) | 43 | Yes | ||
| 31 | 43,44 | 1998 | 47 | m | NR | 400 | 400 | 79 minutes | Yes | 574 (444) | 130 | Yes | ||
| 32 | 21 | 1999 | 41 | f | Yes | Yes | 320 | 915 | 55 minutes | Yes | 610 (426) | 184 | Yes | QTc normalization after 5 day, uneventful |
| 33 | 21 | 1999 | 31 | m | Yes | Yes | 480 | 1700 | 40 minutes | Yes | 599 (491) | 108 | Yes | QTc normalized in 4 days, NR |
| 34 | 18 | 2000 | 64 | f | Yes | Yes | 175 | 175 | NR | No | 413 (418) | (‐5) | Yes | QTc remained unchanged, uneventful |
| 35 | 8 | 2000 | 75 | m | No | NR | >2 | >2 | NR | Yes | 615 (435) | 180 | No | QTc normalization in 48 hours, uneventful |
| 36 | 8 | 2000 | 68 | m | Yes | Yes | >2 | >2 | NR | Yes | 650 (407) | 243 | No | QTc normalization after 4 day, uneventful after extubation |
| 37 | 8 | 2000 | 77 | m | NR | NR | (4) | 2 | NR | Yes | 550 (393) | 157 | No | QTc normalization in 24 to 36 hours, NR |
| 38 | 12 | 2004 | 34 | m | Yes | NR | 24.5 | 24.5 | 20 minutes | Yes | 560 (420) | 140 | Yes | QTc normalization (440 msec), ECG normal |
| 39 | 23 | 2004 | 58 | f | Yes | NR | 340 | 1010 | NR | Yes | 533 (460) | 73 | Yes | QTc normalization 7 days later discharge after 27days |
| 40 | 45 | 2008 | 86 | f | Yes | No | 2 mg | 2 mg | 8 hours after last dose | Yes | 524 (NR) | Probably 79 | No | QTc normalization (445 msec), NR |
| 41 | 46 | 2009 | 74 | m | Yes | No | 2 | 2 | Shortly after | Yes | NR (579) | NR | Yes | Pre‐existing heart block and fibrillation resolved, nursing home/rehabilitation |
| Report | MedWatch Identifier | Report Date | Age, Years | Gender | Drugs Pro‐arrh. | Maximum Daily Dose (mg) | Total Dose at Event (mg) | Prolonged QT | QTc Maximal (baseline), msec | Change in QTc (msec) | TdP | Outcome; Recovery |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||
| 1 | 3122988‐1 | 1998 | 61 | m | No | 48 | 48 | Yes | NR | NR | Yes | Intervention; NR |
| 2 | 3157827‐6 | 1998 | 44 | f | No | 160 | 160 | Yes | 550 (440) | 110 | Yes | Intervention; uneventful |
| 3 | 3178715‐5 | 1999 | 60 | m | NR | 415 | 645 | Yes | NR | NR | Yes | Life‐threatening; QTc normalization in 1 day, no recurrence |
| 4 | 3271261‐X | 1999 | 56 | m | NR | NR | 20 | Yes | NR | NR | Yes | Life‐threatening; QTc normalization |
| 5 | 3271080‐4 | 1999 | 35 | m | Yes | 7 | 7 | NR | NR | NR | Yes | NR; event abated after dose stopped/reduced, hospitalization prolonged |
| 6 | 3325391‐4 | 1999 | 55 | f | Yes | 75 | 75 | NR | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 7 | 3381921‐8 | 1999 | 52 | m | No | 320 | 634 | Yes | 458 (430) | 28 | Yes | Death; NA |
| 8 | 3483869‐7 | 2000 | 18 | m | No | >200 | >310 | Yes | NR | NR | Yes | Intervention; no recurrence after haloperidol reinstitution |
| 9 | 3516342‐8 | 2000 | NR | NR | NR | NR | NR | NR | NR | NR | Yes | NR; NR |
| 10 | 3516320‐9 | 2000 | 34 | m | Yes | 5 | 5 | Yes | NR | NR | No | Life‐threatening; event abated after dose stopped |
| 11 | 3552263‐2 | 2000 | 46 | f | Yes | NR | 97.5 | Yes | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 12 | 3574705‐9 | 2000 | 78 | m | Yes | NR | 160 | Yes | 603 (453) | 50 | Yes | Intervention; event abated after dose stopped/reduced |
| 13 | 3703871‐7 | 2001 | 27 | m | NR | 530 | 530 | Yes | NR | NR | Yes | Death, NA |
| 14 | 3724567‐1 | 2001 | 31 | m | Yes | 6 | 6 | Yes | 496 (449) | 47 | No | Life‐threatening; ECG returned to baseline |
| 15 | 3851984‐1 | 2002 | 72 | f | NR | 18 | 18 | NR | NR | NR | Yes | Hospitalization; NR |
| 16 | 3942407‐2 | 2002 | 51 | m | Yes | 14 | 14 | Yes | 461 (444) | 17 | Yes | Life‐threatening; no recurrence |
| 17 | 4066580‐3 | 2003 | >60 | f | NR | 50 | 50 | Yes | >600 (480) | >120 | No | Hospitalization; QTc normalization, patient recovered |
| 18 | 4126280‐8 | 2003 | 47 | f | NR | 60 | 180 | Yes | 550 (450) | 100 | No (bradycardia) | Hospitalization; patient recovered |
| 19 | 4150700‐6 | 2003 | NR | m | NR | 5 | 5 | NR | NR | NR | Yes | NR; event abated after dose stopped/reduced |
| 20 | 4340092‐1 | 2004 | 52 | m | Yes | 5 | 5 | Yes | >500 (490) | >10 | NR (polymorphous VT) | Life‐threatening; NR |
| 21 | 4714692‐0 | 2005 | NR | m | NR | NR | NR | Yes | NR | NR | Yes | Hospitalization; event abated after dose stopped/reduced |
| 22 | 4881813‐9 | 2006 | NR | m | NR | NR | 40 | NR | NR | NR | Yes | Hospitalization; event abated after dose stopped/reduced |
| 23 | 4892225‐6 | 2006 | NR | f | Yes | 10 | >10 | Yes | 493 (300) | 193 | No | Hospitalization; QTc normalization (403 msec) |
| 24 | 4911873‐8 | 2006 | 69 | m | Yes | 6 | 6 | NR | NR | NR | Yes | Cardiac arrest, death; NA |
| 25 | 5366448‐6 | 2007 | 53 | m | Yes | NR | 35 | Yes | NR | NR | NR | Cardiac arrest, life‐threatening; patient recovered |
| 26 | 5563440‐3 | 2007 | 58 | m | Possible | 5 | 5 | Yes | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 27 | 5642929‐2 | 2008 | 42 | m | Yes | 165 | 165 | Yes | 640 (350) | 290 | Yes | Death; NA |
| 28 | 5697758‐0 | 2008 | 38 | m | Yes | NR | 620 | NR | NR | NR | Yes | Hospitalization; patient recovered |
| 29 | 5254840‐X | 2008 | 19 | f | Possible | 15 | 25 | Yes | 461 | NR | NR | Cardiac arrest, hospitalization; patient recovered |
Of the 70 cases, 54 cases of TdP were reported. The remaining 16 of 70 cases involved cases of QTP, 9 of which did not progress to TdP and 7 of which the progression to TdP was unclear. Of note, 42 of 54 of the cases of TdP were reported as preceded by documented QTP. Presence of QTP was unknown in the other 12 original reports. Three out of 70 patients experienced sudden cardiac arrest, 1 of which was fatal. One arrest was preceded by TdP and 2 by QTP (Figure 1).

The patient ages ranged from 18 years to 86 years. Of note, 17 patients experiencing TdP and/or QTP were 40 years old, and 2 of those patients were 30 years old.
Haloperidol‐associated QTP and/or TdP were observed in 27 female and 42 male patients; the gender was not stated in one report. Of the 54 patients experiencing TdP (with or without report of previous QTP), 22 were female and 31 were male (1 gender unknown).
A total of 68 of 70 patients were determined to have associated risk factors15 for QTP/TdP (see Table 4). The circumstances of the remaining 2 patients were not described in sufficient detail to identify associated risk factors.
| Risk Factor | Patients, n (%) |
|---|---|
| |
| Any risk factor | 68/70 (97) |
| Unknown | 2/70 (3) |
| Specific risk factors | |
| Electrolyte imbalance | 27/68 (40) |
| Underlying cardiac disease | 32/68 (47) |
| Concomitant proarrhythmic agents | 39/68 (57) |
| Other drugs influencing cardiac function | 23/68 (34) |
| Baseline QTc >450 msec | 18/68 (26) |
| QTc known: 44 patients | 18/44 (41) |
Overall, 32 patients had underlying heart conditions. Electrolyte imbalances, including hypokalemia, hypomagnesemia, and hypocalcemia, were present in 17 patients. At least 39 patients were receiving potentially proarrhythmic agents (1‐8 proarrhythmic drugs per patient) in addition to IV haloperidol. At least 23 patients were receiving additional drugs with a potential for other cardiac adverse events than QTP and TdP.
A wide range of other disease states previously reported to be associated with QTP15 were identified in these patients: asthma (5 patients), diabetes (5 patients), obesity (3 patients), impaired renal and/or liver function (3 patients each), human immunodeficiency virus (HIV) (2 patients); chronic obstructive pulmonary disease (COPD), pancreatitis and hypothyroidism (1 patient each). A total of 22 patients had a history of substance abuse (alcohol and/or drugs), and 4 patients were smokers.
The administered doses of IV haloperidol varied widely. Considering that information regarding the maximal daily dose was missing in 22 reports and ambiguous in another 20 cases, the results have been presented using cumulative IV haloperidol doses. Patients experiencing TdP without preceding QTP received a cumulative dose (= total dose at event) ranging from 5 mg to 645 mg. Patients with both confirmed QTP and TdP were administered a cumulative dose of 2 mg to 1700 mg. Patients who experienced QTP without TdP received a cumulative dose of 2 mg to 1540 mg of IV haloperidol.
Sudden cardiac arrest following administration of IV haloperidol was observed in cumulative doses ranging from 6 mg to 35 mg. The cardiac arrest leading to a fatal outcome was preceded by an administration of at least 6 mg of IV haloperidol. Overall, 14 out of 70 patients received cumulative doses of 10 mg IV haloperidol.
The time from administration to documentation of QTP and/or TdP ranged from immediately post administration to 8 hours after administration of the last dose of IV haloperidol.
Baseline QTc was known in 44 patients. Baseline QTc was >450 msec in 18 of these 44 patients.
The change from baseline QTc varied widely from 20 msec to 286 msec; 36 patients demonstrated a prolongation of >50 msec.
In those patients with reported haloperidol‐associated QTP, 25 patients demonstrated a QTc >600 msec and 38 patients >520 msec.9 Of the cases with known specific QTc values, the QTc was prolonged >450 msec in 48 out of 50 cases. The lowest reported QTc leading to TdP was 413 msec.
A total of 20 patients were reported as having a normalization of QTc (as defined by the original reports) within several hours to 8 days; minimal QTP was reported as persisting in 2 patients. The specifics of the other patients were unknown, although 25 patients were categorized as recovered, 13 were stated as having an uneventful remainder of hospitalization, and 5 patients were discharged to a rehabilitation facility or a nursing home.
Discussion
The current review was performed in response to the FDA warning recommending the use of continuous ECG monitoring associated with the administration of intravenous haloperiodol.5 This warning has resulted in substantial dilemmas for health care organizations, additional resource allocation, and increased scrutiny from regulatory agencies. The results of our review reveal that intravenous haloperidol‐associated QTP and TdP almost uniformly take place in patients with concomitant risk factors and with cumulative doses 2 mg. In light of these findings, it is possible that hospitals may be able to administer intravenous haloperidol in patients without risk factors without continuous ECG monitoring. In reviewing these published reports, it is important to note that the FDA identified 28 published cases of haloperidol‐associated QTP and TdP, while our review yielded a total of 41 published case reports.
The FDA database included 73 cases of haloperidol‐associated TdP in their database. However, these cases included both oral as well as IV administration; using our methodology, we identified 29 additional case reports associated with intravenous haloperidol from this database. Consequently, our review included 41 published case reports and 29 FDA database cases, resulting in the total of 70 patients.
Our review revealed a number of practical findings. First, our summary demonstrated that neither QTP nor TdP has been documented with a cumulative dose of IV haloperidol of 2 mg. The majority of patients (80%) received cumulative IV doses 10 mg. The lowest dose associated with sudden cardiac arrest was 6 mg and this took place in a 69‐year‐old male patient. Second, the majority (97%) of our patients had additional risk factors for QTP and/or TdP. Pre‐existing heart disease,1619 electrolyte imbalance,17, 1921 concomitant proarrhythmic drugs16, 17, 1922 and mechanical ventilation17, 23 were identified as the most commonly observed risk factors (Table 4). Lastly, in those cases in which the data were reported, baseline QTc was >450 msec in 41% of the patients, and 96% had a QTc at the time of the event >450 msec. Therefore, we conclude that patients: (1) receiving low cumulative doses (2 mg) with (2) no risk factors for prolonged QTc or TdP, and (3) with a normal QTc on baseline EKG can safely be given IV haloperidol in the hospital setting.
This dosage range is consistent with the labelling for IV haloperidol dosing in Canada24 and Germany25 (Table 1), where single doses of 0.25 mg to 1.5 mg are recommended for the treatment of delirium or acute agitation in the geriatric population.24, 25
In a recent Cochrane review, low‐dose IV haloperidol (3 mg per day) was concluded to be as safe and effective as atypical antipsychotics in the treatment of acute delirium with respect to extrapyramidal adverse effects.2
The American Psychiatric Association recommends an initial IV dose of 12 mg every 24 hours as needed (0.250.50 mg every 4 hours as needed for elderly patients), with titration to higher doses for patients who continue to be agitated for the treatment of patients with delirium (issued 1999, updated 2004).3
While several expert‐groups and investigators currently consider IV haloperidol as an important therapeutic option for treating acute delirium and agitation in the dose range presented above, less consensus exists regarding monitoring requirements.2, 3, 26, 27
The American Psychiatric Association recommends IV haloperidol only after a baseline ECG is obtained. These guidelines have not been updated since the release of the FDA extended warning.3 In their recent review, Morandi et al.28 support the dosage recommendation of the 1999 American Psychiatric Association's practice guidelines for treatment of delirium,3 ie, administration of IV haloperidol in single doses of 0.5 mg to 2 mg in elderly patients, however, only after a baseline ECG is obtained.28 While the package insert of IV haloperidol in France29 recommends a baseline ECG, Germany,25 Italy30 and Switzerland's31 package information states the need for regular ECG monitoring. Guidelines for the treatment of delirium in the intensive care unit published by the American College of Critical Care Medicine and the Society of Critical Care Medicine in collaboration with the American Society of Health‐System Pharmacists consider IV haloperidol as the preferred agent for the treatment of delirium in critically ill patients (grade of recommendation = C). These expert groups recommend that patients should be monitored for electrocardiographic changes (QT interval prolongation and arrhythmias) when receiving haloperidol (Grade of recommendation = B).32
Nevertheless, continuous ECG monitoring (ie, telemetry) is expensive, labor‐intensive and, potentially overutilized.33, 34 Requiring clinicians to place all patients receiving intravenous haloperidol on telemetry is impractical and potentially costly. Mandating telemetry could also lead to unintended harm, ie, use of a less effective or less safe drug to avoid compliance with the telemetry mandate.
Based on our findings and the current recommendations in the literature, inpatient providers should be thoughtful and deliberate in the use of haloperidol to treat acute delirium with agitation. Patients requiring pharmacologic management of their delirium should be screened for risk factors for QTP and TdP (Table 4) and a baseline ECG should be obtained prior to haloperidol administration. If significant risk factors exist or the baseline ECG reveals a prolonged QTc, then the patient should receive continuous ECG monitoring. Similarly, if cumulative doses of 2 mg are needed, the patient should be placed on telemetry.
There are some limitations to our study design. Our findings are based upon previously published case reports or data submitted to the FDA MedWatch. While the content of the FDA's MedWatch database is accessible to the public via the Freedom of Information Act (FOIA), the events are neither categorized nor peer‐reviewed upon entry into the database. Consequently, information may be incomplete or inaccurate. In addition, the denominator representing the overall use of IV haloperidol is unknown, thus a rate of event cannot be assigned and the true scope of the problem cannot be determined. Despite these limitations, this summary represents the most comprehensive review of the literature to date, expanding on the analysis performed by the FDA. Of note, in our review of the FDA database, we noted several cases of haloperidol‐associated QTP or TdP associated with other routes of administration. Thus, it is unknown whether this complication is any greater with IV vs. the IM or per os (PO) routes of administration.
Conclusion
Although the proarrhythmic potential of haloperidol and other antipsychotics has been well established in the literature, IV haloperidol has been considered relatively safe with respect to this complication from the time of its approval in 1967.5, 1722, 35, 36 In reviewing all reported cases of cardiac complications associated with IV haloperidol, as well as the current literature, an association with QTP and TdP is likely. However, the case reports reveal that QTP and TdP generally occur in the setting of concomitant risk factors, and no cases have been reported utilizing a cumulative IV dose of 2 mg. It may therefore be safe to administer a cumulative dose of IV haloperidol of 2 mg without ECG monitoring in patients without risk factors for QTP. However, ECG monitoring should take place with IV haloperidol doses 2 mg and/or in those patients with additional risk factors of developing QTP and/or TdP.
Based on the findings of this review complemented by the guidelines of various expert‐groups and the official labelling information of different countries, the Pharmacy & Therapeutics Committee of the UCSF Medical Center revised the IV haloperidol policy: administration of a total dose of 2 mg IV haloperidol without concurrent telemetry is allowed in a noncritical care setting in patients without risk factors for QTP and TdP.
Acknowledgements
The authors acknowledge Gloria Won of the Fishbon Library at UCSF Medical Center at Mount Zion for her support.
Haloperidol is Food and Drug Administration (FDA)‐approved in the United States for the management of acute and chronic psychotic disorders and widely used in the management of delirium‐associated agitation in hospitalized patients.1 Delirium in the hospital is an acute confusional state that frequently arises from multiple complex factors and may affect up to 30% of hospitalized patients.2 Although the first step in the management of delirium involves identification and treatment of underlying causes and offering supportive behavioral care; medications may be needed to control severe agitation.2 Low dose intravenous (IV) haloperidol (ie, 0.250.5 mg every 4 hours) is a commonly used medication in this setting as recommended by expert‐groups including the Cochrane Collaboration and the American Psychiatric Association.2, 3
Although injectable haloperidol, a butyrophenone‐derived antipsychotic agent pharmacologically related to the piperazine phenothiazines,4 is approved for IV use in many countries (Table 1), parenteral use is approved only for intramuscular (IM) administration in the US. Thus, IV administration of the drug in the US is considered an off‐label use.5
| Indication | Country | |||||
|---|---|---|---|---|---|---|
| Canada24 | France29 | Germany25 | Great Britain37 | Italy30 | Switzerland31 | |
| ||||||
| Mainly delirium (schizophrenia, other psychosis, short‐term management of psychomotor agitation, excitement, violent or dangerously impulsive behavior, vomiting, hiccup) | Short term treatment of agitation and aggressiveness during an acute or chronic psychotic episode, vomiting along with antimitotic post‐radiotherapy treatment | Acute and chronic schizophrenia, psycho‐motorical agitation of psychotic genesis | Schizophrenia, other psychosis, short‐term adjunctive management if psychomotor agitation, violent or dangerous impulsive behavior | Resistant forms of psycho‐motorical excitement, acute delirious and/or hallucinatory psychosis' chronic psychosis High doses restrictions: syndrome of psycho‐motorical excitement, acute delirious and/or hallucinatory psychosis, chronic psychosis | Acute schizophrenic episode, mania, vomiting | |
| IV dosing in adults | 12 mg every 24 hours | The use is limited to adult patients and the drug can be administered IM or IV. The IV route is restricted to the treatment of vomiting. | 510 mg/day, daily max.: 30(100) mg | 210 mg initially, PRN every 48 hours, daily max. 18 mg | 510 mg initially, PRN every hour, daily max. 60 mg | 5 mg PRN every 30 minutes |
| IV dosing in geriatric care | 0.250.5 mg | Single dose of 0.51.5 mg, daily max. 5 mg | Half adult dose | Adjust to appropriate dose | 0.5 mg, than PRN | |
| Risk factors for the development of cardiac adverse events | QT prolonging drugs, diabetes, obesity, hypokalemia, congenital long QT syndrome | Bradycardia 55 beats per minute, hypokalemia, congenital QT prolongation, other medications provoking bradycardia, deceleration of the intra‐cardiac transition or prolonged QT interval | QT syndrome, hypokalemia, other electrolyte imbalance, cardiovascular diseases, QT prolongation in the family history | Cardiovalscular disease, drugs that can prolong the QTc, diabetes, obesity, hypokalemia, congenital long QT syndrome | Contraindications: recent cardiac infarction, uncompensated cardiac insufficiency, cardiac arrhythmias, antiarrhythmic drugs, pre‐existing QT prolongation, cases of arrhythmia or torsades de pointes in the family history, untreated potassium imbalance, QTc prolonging drugs | QT syndrome, hypokalemia, hypomagnesemia, other electrolyte imbalances, cardiovascular diseases, hypothyreosis, QT prolongation in the family history |
| Monitoring recommendations | Electrolytes | ECG monitoring at admission time, electrolytes | ECG monitoring, electrolytes | Metabolic parameters | ECG at baseline and regular ECG monitoring, electrolytes | Close ECG monitoring, electrolytes |
| General recommendations | Regular reevaluation in long‐term use | Apply the lowest effective dose | Apply the lowest effective dose | Application per mouth is the route of choice | Decrease dose if QTc >500 msec | Switch to PO as soon as possible |
Haloperidol is often preferred over other antipsychotics as a result of its effectiveness, low rate of anticholinergic side effects, familiarity with dosing and usage, and minimal respiratory or sedative properties.6 Use of the IV route in patients with acute delirium has several advantages over the IM or oral route,7 including rapid onset, immediate bioavailability, and ease and safety of administration.
Prior to September 2007, the package insert for haloperidol alerted healthcare professionals to the risk of cardiovascular side effects. Based on case reports of potentially fatal cardiac events, the FDA revised the label, warning that the QT prolongation (QTP) and risk of torsades de pointes (TdP) were increased with IV administration of haloperidol or administration of the drug at greater than recommended doses. Unfortunately, neither the typical dosing range nor the minimum dose associated with these cardiac side effects were specified in this recommendation.5
It is well‐established that haloperidol may prolong the QT interval by blocking the repolarizing potassium IKr current.8 Although drugs that block the IKr channel can produce arrhythmia in healthy individuals, additional risk factors, such as underlying heart conditions, electrolyte imbalances (ie, hypokalemia and hypomagnesemia), concomitant proarrhythmic drug use, and mechanical ventilation may increase this risk.9 Prolongation of the QT interval has been associated with subsequent malignant cardiac arrhythmias including ventricular fibrillation and TdP.10 Prolongation of the QT interval is considered the strongest risk factor for TdP, particularly with a baseline QTc > 450 msec.9
Based on the increased risk for QTP and TdP and the case reports of cardiac events, the FDA advisory recommended continuous electrocardiogram (ECG) monitoring in patients receiving IV haloperidol.5 However, such monitoring may be impractical and costly in hospitalized patients who require low doses of IV haloperidol to manage acute delirium and who are not in telemetry or intensive care units.
The aim of this review was to evaluate the case reports leading to the recent FDA warning for IV haloperidol, specifically focusing on the presence of risk factors for arrhythmias. Based upon the evidence, an additional aim was to provide an institutional response to this warning toward the optimal use of this agent.
Method
Two search pathways were used to evaluate reports of haloperidol‐associated TdP and/or QT prolongation:
Literature Review
We searched for published literature in humans indexed in Pubmed (1966April 2009), EMBASE (1972April 2009), and Scopus (1823April 2009) using the search terms haloperidol or Haldol combined with intravenous or infusion and at least one of the following terms: QT prolongation, TdP, torsades de pointes, torsades with a specific focus on case reports.
References from the retrieved articles were also reviewed to search for additional case reports.
In addition to cases reported in English journals, several of our reports originated from Japan11 (translation provided by the FDA), Spain12 and Germany13 (translated by the primary author).
Search of the FDA Database
We reviewed all adverse drug events reported through MedWatch or those submitted by the manufacturer from November 1997 to April 2008 through the Freedom of Information Act (FOIA) request. The FDA provided a full‐text summary of 5944 reports involving oral, intramuscular and IV use of haloperidol. The FDA data were transferred to a Microsoft Access database and screened for the key terms torsade, QT, prolongation, wave. Incident report number, date of report, age, gender, origin of report, medication name, role of drug as categorized by the FDA (suspect, concomitant, primary suspect, secondary suspect), route, dose, units, duration, symptoms and FDA outcome category (death, life‐threatening, hospitalization initial or prolonged, disability, congenital anomaly, required intervention to prevent permanent damage, other) were recorded. Only those reports in which IV haloperidol was considered by the reporter to be the primary causative agent for the adverse event were reviewed. Available information included diagnosis, laboratory parameters, QTc measurement, cardiac symptoms, outcomes and a description of recovery. No peer review was applied to the MedWatch reports and the data reported in this publication reflect the original information from the FDA MedWatch database. Baseline QTc was either the value defined as such in the original report or the lowest QTc reported. Haloperidol doses administered were defined as cumulative dose at event, encompassing all doses administered during the hospital stay until the occurrence of the adverse cardiac event.
The drugs listed in the case reports were assessed for proarrhythmic potential using 2 references: the individual package insert and the website of the Arizona Center for Education and Research on Therapeutics.14
The drugs were only considered proarrhythmic when the 2 resources were in agreement.
Duplicates and/or previously published cases, as well as reports involving adverse cardiac effects not associated with QTP or TdP, were identified and excluded.
In their advisory, the FDA does not state the exact origin of the reports, their specific search strategy to identify haloperidol‐associated adverse events, or the role IV haloperidol played in the individual events included in the extended warning. Consequently, the number of events identified in this review may differ from that published in the FDA extended warning.
Results
A total of 70 reported cases of IV haloperidol associated TdP and/or QTP were identified. Of these 70, 41 were identified through the PubMed/EMBASE/Scopus review, while an additional 29 cases were identified through the FDA database search.
Of the 29 cases in the FDA database, 21 were reported by health care professionals and 8 by manufacturers.
A total of 35 publications described cases originating from the US. Three cases took place in Japan and 1 case each in Canada, Germany and Spain. Several cases in the MedWatch database were reported outside the US: 1 case each originated from Austria, Canada, France, Japan, Spain, Switzerland and the United Kingdom. A summary of the published case reports is displayed in Table 2 and the FDA cases are summarized in Table 3.
| Case | Source (reference#) | Date | Age, Years | Gender | Drugs Pro‐arrhyth. | Venti‐ lated | Max. Daily Dose (mg) | Total Dose at Event (mg) | Time to Event | Prolonged QT | QTc Maximal (baseline), msec | Change in QTc (msec) | TdP | ECG Normalization, Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||
| 1 | 35 | 1991 | 56 | m | No | Yes | 1200 | 1540 | NR | Yes | 584 (400) | 184 | NR | NR, uneventful |
| 2 | 13 | 1992 | 36 | m | Yes | No | 11.5 | 11.5 | 20 hours after start | Yes | 714 (428) | 286 | Yes | QTc normalization (440 msec), NR |
| 3 | 38 | 1993 | 39 | f | Yes | Yes | NR | 580 | Max. QTc 72 hours after start | Yes | 650 (420) | 230 | Yes | QTc normalization after 6 days, uneventful |
| 4 | 38 | 1993 | 19 | f | Yes | No | 170 | 170 | Max. QT 12 hours after start | Yes | 600 (480) | 120 | Yes | QTc normalization after 8 days, uneventful |
| 5 | 38 | 1993 | 63 | f | Yes | No | NR | 489 | Max. QT 48 hours after start | Yes | 670 (520) | 150 | Yes | QTc normalization after 8 days, uneventful |
| 6 | 38 | 1993 | 74 | f | Yes | Yes | NR | 10 | NR | No | 430 (410) | 20 | Yes | QTc unchanged after 8 days, uneventful |
| 7 | 17 | 1993 | 39 | m | Yes | Yes | NR | >490 | NR | Yes | 457 (348) | 109 | Yes | QTc normalization within 2 to 3 days, no further TdP, NR |
| 8 | 17 | 1993 | 61 | m | Yes | Yes | 115 | 211 | NR | Yes | 500 (390) | 110 | NR | QTc normalization within 2 days, death |
| 9 | 17 | 1993 | 48 | m | Yes | Yes | 825 | 825 | NR | Yes | 538 (441) | 97 | NR | QTc normalization in 3 days, rehabilitation |
| 10 | 39 | 1994 | 23 | f | Yes | Yes | 120 | 300 | 12 hours after dose increase | Yes | NR (550) | NR | Yes | NR, uneventful, extubation after 5 days, discharge after 10 days |
| 11 | 39 | 1994 | 28 | m | Yes | Yes | 300 | >300 | 24 hours after dose increase | Yes | NR (>520) | NR | Yes | No recurrence of arrhythmia, patient death (multi‐organ failure) |
| 12 | 40 | 1994 | 65 | m | Yes | NR | 230 | 410 | Worsening from day 2 to day 5 | Yes | 594 (490) | 104 | Yes | QTc normalization (406 msec), no cardiac problems at discharge |
| 13 | 40 | 1994 | 65 | f | Yes | NR | 500 | 980 | After the last 60mg | Yes | 628 (403) | 225 | Yes | QTc normalization (400 msec), recurrence with oral haloperidol, rehabilitation |
| 14 | 40 | 1994 | 76 | f | Yes | NR | 21 | 26 | Day 2 after several boluses | Yes | 670 (450) | 220 | Yes | QTc normalization within several days (412 msec), rehabilitation |
| 15 | 41 | 1994 | 59 | m | NR | Yes | 865 | 1013 | NR | Yes | 640 (480) | 160 | NR | QTc normalization in 24 hours, survived |
| 16 | 16 | 1995 | 76 | f | Yes | No | NR | 44.5 plus 1 PO | 15 minutes | Yes | 670 (409) | 261 | Yes | ECG normalized the next morning, no further events |
| 17 | 16 | 1995 | 49 | m | Yes | No | NR | 1150 plus 20 IM | 45 minutes | Yes | 648 (380) | 268 | Yes | QTc normalization in 24 hours, anoxic brain insult/rehabilitation |
| 18 | 16 | 1995 | 65 | f | Yes | No | 600 | 965 | 30 minutes | Yes | 628 (403) | 225 | Yes | 3 more episodes of TdP in 3 hours, QTc normalization in 2 days, no recurrence with further haloperidol, NR |
| 19 | 42 | 1995 | 42 | m | Yes | No | 28 | 28 | 20 minutes | Yes | 610 (533) | 77 | Yes | QTc normalization in 5 days, uneventful, ECG normal |
| 20 | 42 | 1995 | 39 | m | Yes | No | 45 | 45 | 5 minutes | Yes | 654 (NR) | NR | Yes | QTc normalization after 24 hours, uneventful |
| 21 | 11 | 1997 | 56 | f | No | No | 10 | 10 | Shortly after | NR | NR (NR) | NR | Yes | TdP resolved after 8 hours, NR |
| 22 | 11 | 1997 | 82 | f | NR | No | 10 | 10 | Shortly after | Yes | 680 (NR) | NR | Yes | QTc normalization on day 6 after admission (470 msec), NR |
| 23 | 11 | 1997 | 35 | m | NR | No | NR | 90 | After 20 mg | Yes | 520 (NR) | NR | Yes | TdP disappeared 12 hours later, NR |
| 24 | 43,44 | 1998 | 45 | m | NR | Yes* | NR | 9 | 203 minutes | Yes | 638 (560) | 78 | Yes | NR, overall survival 100%, significantly prolonged hospital stay |
| 25 | 43,44 | 1998 | 64 | f | NR | NR | 115 | 220 minutes | Yes | 605 (424) | 181 | Yes | ||
| 26 | 43,44 | 1998 | 75 | f | NR | NR | 85 | 60 minutes | Yes | 567 (508) | 59 | Yes | ||
| 27 | 43,44 | 1998 | 71 | f | NR | NR | 55 | 120 minutes | Paced | Paced | Paced | Yes | ||
| 28 | 43,44 | 1998 | 58 | f | NR | NR | 75 | 38 minutes | Yes | 657 (542) | 115 | Yes | ||
| 29 | 43,44 | 1998 | 40 | m | NR | NR | 35 | 15 minutes | Yes | 679 (475) | 204 | Yes | ||
| 30 | 43,44 | 1998 | 71 | m | NR | NR | 70 | 58 minutes | Yes | 521 (478) | 43 | Yes | ||
| 31 | 43,44 | 1998 | 47 | m | NR | 400 | 400 | 79 minutes | Yes | 574 (444) | 130 | Yes | ||
| 32 | 21 | 1999 | 41 | f | Yes | Yes | 320 | 915 | 55 minutes | Yes | 610 (426) | 184 | Yes | QTc normalization after 5 day, uneventful |
| 33 | 21 | 1999 | 31 | m | Yes | Yes | 480 | 1700 | 40 minutes | Yes | 599 (491) | 108 | Yes | QTc normalized in 4 days, NR |
| 34 | 18 | 2000 | 64 | f | Yes | Yes | 175 | 175 | NR | No | 413 (418) | (‐5) | Yes | QTc remained unchanged, uneventful |
| 35 | 8 | 2000 | 75 | m | No | NR | >2 | >2 | NR | Yes | 615 (435) | 180 | No | QTc normalization in 48 hours, uneventful |
| 36 | 8 | 2000 | 68 | m | Yes | Yes | >2 | >2 | NR | Yes | 650 (407) | 243 | No | QTc normalization after 4 day, uneventful after extubation |
| 37 | 8 | 2000 | 77 | m | NR | NR | (4) | 2 | NR | Yes | 550 (393) | 157 | No | QTc normalization in 24 to 36 hours, NR |
| 38 | 12 | 2004 | 34 | m | Yes | NR | 24.5 | 24.5 | 20 minutes | Yes | 560 (420) | 140 | Yes | QTc normalization (440 msec), ECG normal |
| 39 | 23 | 2004 | 58 | f | Yes | NR | 340 | 1010 | NR | Yes | 533 (460) | 73 | Yes | QTc normalization 7 days later discharge after 27days |
| 40 | 45 | 2008 | 86 | f | Yes | No | 2 mg | 2 mg | 8 hours after last dose | Yes | 524 (NR) | Probably 79 | No | QTc normalization (445 msec), NR |
| 41 | 46 | 2009 | 74 | m | Yes | No | 2 | 2 | Shortly after | Yes | NR (579) | NR | Yes | Pre‐existing heart block and fibrillation resolved, nursing home/rehabilitation |
| Report | MedWatch Identifier | Report Date | Age, Years | Gender | Drugs Pro‐arrh. | Maximum Daily Dose (mg) | Total Dose at Event (mg) | Prolonged QT | QTc Maximal (baseline), msec | Change in QTc (msec) | TdP | Outcome; Recovery |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||
| 1 | 3122988‐1 | 1998 | 61 | m | No | 48 | 48 | Yes | NR | NR | Yes | Intervention; NR |
| 2 | 3157827‐6 | 1998 | 44 | f | No | 160 | 160 | Yes | 550 (440) | 110 | Yes | Intervention; uneventful |
| 3 | 3178715‐5 | 1999 | 60 | m | NR | 415 | 645 | Yes | NR | NR | Yes | Life‐threatening; QTc normalization in 1 day, no recurrence |
| 4 | 3271261‐X | 1999 | 56 | m | NR | NR | 20 | Yes | NR | NR | Yes | Life‐threatening; QTc normalization |
| 5 | 3271080‐4 | 1999 | 35 | m | Yes | 7 | 7 | NR | NR | NR | Yes | NR; event abated after dose stopped/reduced, hospitalization prolonged |
| 6 | 3325391‐4 | 1999 | 55 | f | Yes | 75 | 75 | NR | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 7 | 3381921‐8 | 1999 | 52 | m | No | 320 | 634 | Yes | 458 (430) | 28 | Yes | Death; NA |
| 8 | 3483869‐7 | 2000 | 18 | m | No | >200 | >310 | Yes | NR | NR | Yes | Intervention; no recurrence after haloperidol reinstitution |
| 9 | 3516342‐8 | 2000 | NR | NR | NR | NR | NR | NR | NR | NR | Yes | NR; NR |
| 10 | 3516320‐9 | 2000 | 34 | m | Yes | 5 | 5 | Yes | NR | NR | No | Life‐threatening; event abated after dose stopped |
| 11 | 3552263‐2 | 2000 | 46 | f | Yes | NR | 97.5 | Yes | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 12 | 3574705‐9 | 2000 | 78 | m | Yes | NR | 160 | Yes | 603 (453) | 50 | Yes | Intervention; event abated after dose stopped/reduced |
| 13 | 3703871‐7 | 2001 | 27 | m | NR | 530 | 530 | Yes | NR | NR | Yes | Death, NA |
| 14 | 3724567‐1 | 2001 | 31 | m | Yes | 6 | 6 | Yes | 496 (449) | 47 | No | Life‐threatening; ECG returned to baseline |
| 15 | 3851984‐1 | 2002 | 72 | f | NR | 18 | 18 | NR | NR | NR | Yes | Hospitalization; NR |
| 16 | 3942407‐2 | 2002 | 51 | m | Yes | 14 | 14 | Yes | 461 (444) | 17 | Yes | Life‐threatening; no recurrence |
| 17 | 4066580‐3 | 2003 | >60 | f | NR | 50 | 50 | Yes | >600 (480) | >120 | No | Hospitalization; QTc normalization, patient recovered |
| 18 | 4126280‐8 | 2003 | 47 | f | NR | 60 | 180 | Yes | 550 (450) | 100 | No (bradycardia) | Hospitalization; patient recovered |
| 19 | 4150700‐6 | 2003 | NR | m | NR | 5 | 5 | NR | NR | NR | Yes | NR; event abated after dose stopped/reduced |
| 20 | 4340092‐1 | 2004 | 52 | m | Yes | 5 | 5 | Yes | >500 (490) | >10 | NR (polymorphous VT) | Life‐threatening; NR |
| 21 | 4714692‐0 | 2005 | NR | m | NR | NR | NR | Yes | NR | NR | Yes | Hospitalization; event abated after dose stopped/reduced |
| 22 | 4881813‐9 | 2006 | NR | m | NR | NR | 40 | NR | NR | NR | Yes | Hospitalization; event abated after dose stopped/reduced |
| 23 | 4892225‐6 | 2006 | NR | f | Yes | 10 | >10 | Yes | 493 (300) | 193 | No | Hospitalization; QTc normalization (403 msec) |
| 24 | 4911873‐8 | 2006 | 69 | m | Yes | 6 | 6 | NR | NR | NR | Yes | Cardiac arrest, death; NA |
| 25 | 5366448‐6 | 2007 | 53 | m | Yes | NR | 35 | Yes | NR | NR | NR | Cardiac arrest, life‐threatening; patient recovered |
| 26 | 5563440‐3 | 2007 | 58 | m | Possible | 5 | 5 | Yes | NR | NR | Yes | Life‐threatening; event abated after dose stopped/reduced |
| 27 | 5642929‐2 | 2008 | 42 | m | Yes | 165 | 165 | Yes | 640 (350) | 290 | Yes | Death; NA |
| 28 | 5697758‐0 | 2008 | 38 | m | Yes | NR | 620 | NR | NR | NR | Yes | Hospitalization; patient recovered |
| 29 | 5254840‐X | 2008 | 19 | f | Possible | 15 | 25 | Yes | 461 | NR | NR | Cardiac arrest, hospitalization; patient recovered |
Of the 70 cases, 54 cases of TdP were reported. The remaining 16 of 70 cases involved cases of QTP, 9 of which did not progress to TdP and 7 of which the progression to TdP was unclear. Of note, 42 of 54 of the cases of TdP were reported as preceded by documented QTP. Presence of QTP was unknown in the other 12 original reports. Three out of 70 patients experienced sudden cardiac arrest, 1 of which was fatal. One arrest was preceded by TdP and 2 by QTP (Figure 1).

The patient ages ranged from 18 years to 86 years. Of note, 17 patients experiencing TdP and/or QTP were 40 years old, and 2 of those patients were 30 years old.
Haloperidol‐associated QTP and/or TdP were observed in 27 female and 42 male patients; the gender was not stated in one report. Of the 54 patients experiencing TdP (with or without report of previous QTP), 22 were female and 31 were male (1 gender unknown).
A total of 68 of 70 patients were determined to have associated risk factors15 for QTP/TdP (see Table 4). The circumstances of the remaining 2 patients were not described in sufficient detail to identify associated risk factors.
| Risk Factor | Patients, n (%) |
|---|---|
| |
| Any risk factor | 68/70 (97) |
| Unknown | 2/70 (3) |
| Specific risk factors | |
| Electrolyte imbalance | 27/68 (40) |
| Underlying cardiac disease | 32/68 (47) |
| Concomitant proarrhythmic agents | 39/68 (57) |
| Other drugs influencing cardiac function | 23/68 (34) |
| Baseline QTc >450 msec | 18/68 (26) |
| QTc known: 44 patients | 18/44 (41) |
Overall, 32 patients had underlying heart conditions. Electrolyte imbalances, including hypokalemia, hypomagnesemia, and hypocalcemia, were present in 17 patients. At least 39 patients were receiving potentially proarrhythmic agents (1‐8 proarrhythmic drugs per patient) in addition to IV haloperidol. At least 23 patients were receiving additional drugs with a potential for other cardiac adverse events than QTP and TdP.
A wide range of other disease states previously reported to be associated with QTP15 were identified in these patients: asthma (5 patients), diabetes (5 patients), obesity (3 patients), impaired renal and/or liver function (3 patients each), human immunodeficiency virus (HIV) (2 patients); chronic obstructive pulmonary disease (COPD), pancreatitis and hypothyroidism (1 patient each). A total of 22 patients had a history of substance abuse (alcohol and/or drugs), and 4 patients were smokers.
The administered doses of IV haloperidol varied widely. Considering that information regarding the maximal daily dose was missing in 22 reports and ambiguous in another 20 cases, the results have been presented using cumulative IV haloperidol doses. Patients experiencing TdP without preceding QTP received a cumulative dose (= total dose at event) ranging from 5 mg to 645 mg. Patients with both confirmed QTP and TdP were administered a cumulative dose of 2 mg to 1700 mg. Patients who experienced QTP without TdP received a cumulative dose of 2 mg to 1540 mg of IV haloperidol.
Sudden cardiac arrest following administration of IV haloperidol was observed in cumulative doses ranging from 6 mg to 35 mg. The cardiac arrest leading to a fatal outcome was preceded by an administration of at least 6 mg of IV haloperidol. Overall, 14 out of 70 patients received cumulative doses of 10 mg IV haloperidol.
The time from administration to documentation of QTP and/or TdP ranged from immediately post administration to 8 hours after administration of the last dose of IV haloperidol.
Baseline QTc was known in 44 patients. Baseline QTc was >450 msec in 18 of these 44 patients.
The change from baseline QTc varied widely from 20 msec to 286 msec; 36 patients demonstrated a prolongation of >50 msec.
In those patients with reported haloperidol‐associated QTP, 25 patients demonstrated a QTc >600 msec and 38 patients >520 msec.9 Of the cases with known specific QTc values, the QTc was prolonged >450 msec in 48 out of 50 cases. The lowest reported QTc leading to TdP was 413 msec.
A total of 20 patients were reported as having a normalization of QTc (as defined by the original reports) within several hours to 8 days; minimal QTP was reported as persisting in 2 patients. The specifics of the other patients were unknown, although 25 patients were categorized as recovered, 13 were stated as having an uneventful remainder of hospitalization, and 5 patients were discharged to a rehabilitation facility or a nursing home.
Discussion
The current review was performed in response to the FDA warning recommending the use of continuous ECG monitoring associated with the administration of intravenous haloperiodol.5 This warning has resulted in substantial dilemmas for health care organizations, additional resource allocation, and increased scrutiny from regulatory agencies. The results of our review reveal that intravenous haloperidol‐associated QTP and TdP almost uniformly take place in patients with concomitant risk factors and with cumulative doses 2 mg. In light of these findings, it is possible that hospitals may be able to administer intravenous haloperidol in patients without risk factors without continuous ECG monitoring. In reviewing these published reports, it is important to note that the FDA identified 28 published cases of haloperidol‐associated QTP and TdP, while our review yielded a total of 41 published case reports.
The FDA database included 73 cases of haloperidol‐associated TdP in their database. However, these cases included both oral as well as IV administration; using our methodology, we identified 29 additional case reports associated with intravenous haloperidol from this database. Consequently, our review included 41 published case reports and 29 FDA database cases, resulting in the total of 70 patients.
Our review revealed a number of practical findings. First, our summary demonstrated that neither QTP nor TdP has been documented with a cumulative dose of IV haloperidol of 2 mg. The majority of patients (80%) received cumulative IV doses 10 mg. The lowest dose associated with sudden cardiac arrest was 6 mg and this took place in a 69‐year‐old male patient. Second, the majority (97%) of our patients had additional risk factors for QTP and/or TdP. Pre‐existing heart disease,1619 electrolyte imbalance,17, 1921 concomitant proarrhythmic drugs16, 17, 1922 and mechanical ventilation17, 23 were identified as the most commonly observed risk factors (Table 4). Lastly, in those cases in which the data were reported, baseline QTc was >450 msec in 41% of the patients, and 96% had a QTc at the time of the event >450 msec. Therefore, we conclude that patients: (1) receiving low cumulative doses (2 mg) with (2) no risk factors for prolonged QTc or TdP, and (3) with a normal QTc on baseline EKG can safely be given IV haloperidol in the hospital setting.
This dosage range is consistent with the labelling for IV haloperidol dosing in Canada24 and Germany25 (Table 1), where single doses of 0.25 mg to 1.5 mg are recommended for the treatment of delirium or acute agitation in the geriatric population.24, 25
In a recent Cochrane review, low‐dose IV haloperidol (3 mg per day) was concluded to be as safe and effective as atypical antipsychotics in the treatment of acute delirium with respect to extrapyramidal adverse effects.2
The American Psychiatric Association recommends an initial IV dose of 12 mg every 24 hours as needed (0.250.50 mg every 4 hours as needed for elderly patients), with titration to higher doses for patients who continue to be agitated for the treatment of patients with delirium (issued 1999, updated 2004).3
While several expert‐groups and investigators currently consider IV haloperidol as an important therapeutic option for treating acute delirium and agitation in the dose range presented above, less consensus exists regarding monitoring requirements.2, 3, 26, 27
The American Psychiatric Association recommends IV haloperidol only after a baseline ECG is obtained. These guidelines have not been updated since the release of the FDA extended warning.3 In their recent review, Morandi et al.28 support the dosage recommendation of the 1999 American Psychiatric Association's practice guidelines for treatment of delirium,3 ie, administration of IV haloperidol in single doses of 0.5 mg to 2 mg in elderly patients, however, only after a baseline ECG is obtained.28 While the package insert of IV haloperidol in France29 recommends a baseline ECG, Germany,25 Italy30 and Switzerland's31 package information states the need for regular ECG monitoring. Guidelines for the treatment of delirium in the intensive care unit published by the American College of Critical Care Medicine and the Society of Critical Care Medicine in collaboration with the American Society of Health‐System Pharmacists consider IV haloperidol as the preferred agent for the treatment of delirium in critically ill patients (grade of recommendation = C). These expert groups recommend that patients should be monitored for electrocardiographic changes (QT interval prolongation and arrhythmias) when receiving haloperidol (Grade of recommendation = B).32
Nevertheless, continuous ECG monitoring (ie, telemetry) is expensive, labor‐intensive and, potentially overutilized.33, 34 Requiring clinicians to place all patients receiving intravenous haloperidol on telemetry is impractical and potentially costly. Mandating telemetry could also lead to unintended harm, ie, use of a less effective or less safe drug to avoid compliance with the telemetry mandate.
Based on our findings and the current recommendations in the literature, inpatient providers should be thoughtful and deliberate in the use of haloperidol to treat acute delirium with agitation. Patients requiring pharmacologic management of their delirium should be screened for risk factors for QTP and TdP (Table 4) and a baseline ECG should be obtained prior to haloperidol administration. If significant risk factors exist or the baseline ECG reveals a prolonged QTc, then the patient should receive continuous ECG monitoring. Similarly, if cumulative doses of 2 mg are needed, the patient should be placed on telemetry.
There are some limitations to our study design. Our findings are based upon previously published case reports or data submitted to the FDA MedWatch. While the content of the FDA's MedWatch database is accessible to the public via the Freedom of Information Act (FOIA), the events are neither categorized nor peer‐reviewed upon entry into the database. Consequently, information may be incomplete or inaccurate. In addition, the denominator representing the overall use of IV haloperidol is unknown, thus a rate of event cannot be assigned and the true scope of the problem cannot be determined. Despite these limitations, this summary represents the most comprehensive review of the literature to date, expanding on the analysis performed by the FDA. Of note, in our review of the FDA database, we noted several cases of haloperidol‐associated QTP or TdP associated with other routes of administration. Thus, it is unknown whether this complication is any greater with IV vs. the IM or per os (PO) routes of administration.
Conclusion
Although the proarrhythmic potential of haloperidol and other antipsychotics has been well established in the literature, IV haloperidol has been considered relatively safe with respect to this complication from the time of its approval in 1967.5, 1722, 35, 36 In reviewing all reported cases of cardiac complications associated with IV haloperidol, as well as the current literature, an association with QTP and TdP is likely. However, the case reports reveal that QTP and TdP generally occur in the setting of concomitant risk factors, and no cases have been reported utilizing a cumulative IV dose of 2 mg. It may therefore be safe to administer a cumulative dose of IV haloperidol of 2 mg without ECG monitoring in patients without risk factors for QTP. However, ECG monitoring should take place with IV haloperidol doses 2 mg and/or in those patients with additional risk factors of developing QTP and/or TdP.
Based on the findings of this review complemented by the guidelines of various expert‐groups and the official labelling information of different countries, the Pharmacy & Therapeutics Committee of the UCSF Medical Center revised the IV haloperidol policy: administration of a total dose of 2 mg IV haloperidol without concurrent telemetry is allowed in a noncritical care setting in patients without risk factors for QTP and TdP.
Acknowledgements
The authors acknowledge Gloria Won of the Fishbon Library at UCSF Medical Center at Mount Zion for her support.
- Haldol® injection (for immediate release) Package Insert.Raritan, NJ:Ortho‐McNeil Pharmaceutical Inc.;2005;rev. 05.2007.
- ,,,. Antipsychotics for delirium (review), the Cochrane collaboration2008;2. Available at: www. cochrane.org. Accessed February 2010.
- American Psychiatric Association: practice guideline for the treatment of patients with delirium.Am J Psychiatry.1999;156(5 suppl):1–20, updated 2004.
- Thomson Micromedex.2008. Micromedex healthcare series: “haloperidol” Thomson Micromedex, Greenwood Village.
- FDA alert: haloperidol (marketed as Haldol, Haldol Decanoate and Haldol Lactate).2007. This alert highlights revisions to the labeling for haloperidol. Available at: www.fda.gov. Accessed February 2010.
- ,,.Use of high‐dose intravenous haloperidol in the treatment of agitated cardiac patients,J Clin Psychopharmacol.1985;5(6):344–347.
- ,,, et al.Postoperative delirium,Am J Psychiatry.2008;165:7.
- ,.Corrected QT interval prolongation associated with intravenous haloperidol in acute coronary syndromes,Catheter Cardiovasc Interv.2000;50(3):352–355.
- ,.Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death.Am J Psych.2001;158(11):1774–1782.
- ,,, et al.Accuracy of uncorrected versus corrected QT Interval for Prediction of torsade de pointes associated with intravenous haloperidol.Pharmacotherapy.2007;27(2):175–182.
- ,,.Three cases of ventricular tachycardia and torsades de pointes induced by antipsychotic drugs.Shinzo.1997;29(1):68–74.
- ,,,.Haloperidol por via intravenosa y torsade de pointes.Medicina intensive.2004;28(2):89.
- ,,, et al.QT‐Verlängerung und Kammerflimmern unter Haloperidol‐ und Clonidin‐Therapie des Alkoholentzugssyndroms.Intensivmedizin und Notfallmedizin.1992;29(4):178–183.
- ARIZONA CERT, Arizona Center for Education and Research on Therapeutics. Available at: www.azcert.org. Accessed February 2010.
- ,.Cardiac arrhythmias—a clinical approach.Edinburgh:Mosby;2003.
- ,.The association between intravenous haloperidol and torsades de pointes—three cases and a literature review.Psychosomatics.1995;36:541–549.
- ,.Prolongation of the corrected QT and torsades de pointes cardiac arrhythmia associated with intravenous haloperidol in the medically ill.J Clin Psychopharmacol.1993;13(2):128–132.
- ,,,,.Torsades de pointes secondary to intravenous haloperidol after coronary bypass grafting surgery.Can J Anesth.2000;47(3):251–254.
- ,.Torsade de pointes associated with the administration of intravenous haloperidol: a review of the literature and practical guidelines for use,Expert Opin Drug Saf.2003;2(6):543–547.
- ,.Conduction disturbances associated with administration of butyrophenone antipsychotics in the critically ill: a review of the literature,Pharmacotherapy.1997;17(3):531–537.
- ,,.Haloperidol‐induced torsades de pointes.Ann Pharacother.1999;33(10):1046–1050.
- ,,, et al.Practice parameters for intravenous analgesia and sedation for adult patients in the intensive care unit: an executive summary,Crit Care Med.1995;23(9):1596–1600.
- ,,,,,.Prolonged cardiac repolarization after tacrolimus and haloperidol administration in the critically ill patient.Pharmacotherapy.2004;24(3):404–408.
- CPS Compendium of Pharmaceuticals and Specialties, the Canadian drug reference for health professionals, 2007, Canadian pharmacists association.
- Rote Liste Deutschland2008, Rote Liste Service GmbH Frankfurt am Main. Available at: www.rote‐liste.de. Accessed February 2010.
- ,.Delirium in the hospitalized patient: a primer for the pharmacist clinician.J Pharm Pract.2007;20(5):368–372.
- ,,, et al.Delirium: guidelines for general hospitals.J Psychosom Res.2007;62(3):371–383.
- ,,,.The pharmacological management of delirium in critical illness.Current Drug Therapy.2008,3:148–157.
- VIDAL‐l'information sur les produits de santé2008, Issy les Moulineaux Cedex. Available at: www.vidal.fr. Accessed February 2010.
- Haldol iniettabile—ufficiale monografia italiana. Available at: www. informatorefarmaceutico.it. Accessed February 2010.
- Arzneimittelkompendium der Schweiz2008, documed Verlag Basel. Availabla at: www.kompendium.ch. Accessed February 2010.
- ,,, et al.Clinical practice guidelines for the sustained use of sedatives and analgesics in the critically ill, American College of Critical Care Medicine ACCM, Society of Critical Care Medicine SCCM, American Society of Health‐System Pharmacists ASHP.Crit Care Med.2002;30(1):119–141.
- ,,,,.Is telemetry overused? Is it as helpful as thought?Cleve Clin J Med.2009;76(6):368–372.
- ,,.Telemetry outside critical care units: patterns of utilization and influence on management decisions.Clin Cardiol.1998;21(7):503–505.
- ,,.High‐dose intravenous haloperidol for agitated delirium in a cardiac patient on intra‐aortic balloon pump.J Clin Psychopharmacol.1991;11(2):146–147.
- .Haloperidol, midazolam and intravenous sedation.Aust NZ J Psychiatry.1999;33(6):942–943.
- BNF British National Formulary, compendium of pharmaceuticals and specialties of the UK.2007. Available at: www.bnf.org. Accessed February 2010.
- ,,,.Torsade de pointes associated with the use of intravenous haloperidol.Ann Intern Med.1993;119(5):391–394.
- ,.Torsades de pointes: potential consequence of intravenous haloperidol in the intensive care unit.Intensive Care World.1994;11(3):109–112.
- ,.Torsade de pointes caused by high‐dose intravenous haloperidol in cardiac patients.Clin Cardiol.1995;18:285–290.
- ,,.Continuous infusion of haloperidol controls agitation in critically ill patients.Crit Care Med.1994:22(3):433–440.
- ,,.Torsade de pointes complicating the treatment of bleeding esophageal varices: association with neuroleptics, vasopressin, and electrolyte imbalances.Am J Gastroenterol.1995;90(5):822–824.
- ,,,.Torsades de pointes associated with intravenous haloperidol in critically ill patients.Am J Cardiol.1998;81(2):238–240.
- ,,,,.The effect of intravenous haloperidol on QT interval dispersion in critically ill patients: comparison with QT interval prolongation for assessment of risk of torsades de pointes.J Clin Pharmacol.2001;41:1310–1318.
- ,,.Low‐dose haloperidol associated QTc prolongation.J Am Geriatr Soc.2008;56(10):1963–1964.
- ,,.Torsade de pointes following intravenous haloperidol administration in a patient with complete heart block.WMJ.2009;108(1):48–50.
- Haldol® injection (for immediate release) Package Insert.Raritan, NJ:Ortho‐McNeil Pharmaceutical Inc.;2005;rev. 05.2007.
- ,,,. Antipsychotics for delirium (review), the Cochrane collaboration2008;2. Available at: www. cochrane.org. Accessed February 2010.
- American Psychiatric Association: practice guideline for the treatment of patients with delirium.Am J Psychiatry.1999;156(5 suppl):1–20, updated 2004.
- Thomson Micromedex.2008. Micromedex healthcare series: “haloperidol” Thomson Micromedex, Greenwood Village.
- FDA alert: haloperidol (marketed as Haldol, Haldol Decanoate and Haldol Lactate).2007. This alert highlights revisions to the labeling for haloperidol. Available at: www.fda.gov. Accessed February 2010.
- ,,.Use of high‐dose intravenous haloperidol in the treatment of agitated cardiac patients,J Clin Psychopharmacol.1985;5(6):344–347.
- ,,, et al.Postoperative delirium,Am J Psychiatry.2008;165:7.
- ,.Corrected QT interval prolongation associated with intravenous haloperidol in acute coronary syndromes,Catheter Cardiovasc Interv.2000;50(3):352–355.
- ,.Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death.Am J Psych.2001;158(11):1774–1782.
- ,,, et al.Accuracy of uncorrected versus corrected QT Interval for Prediction of torsade de pointes associated with intravenous haloperidol.Pharmacotherapy.2007;27(2):175–182.
- ,,.Three cases of ventricular tachycardia and torsades de pointes induced by antipsychotic drugs.Shinzo.1997;29(1):68–74.
- ,,,.Haloperidol por via intravenosa y torsade de pointes.Medicina intensive.2004;28(2):89.
- ,,, et al.QT‐Verlängerung und Kammerflimmern unter Haloperidol‐ und Clonidin‐Therapie des Alkoholentzugssyndroms.Intensivmedizin und Notfallmedizin.1992;29(4):178–183.
- ARIZONA CERT, Arizona Center for Education and Research on Therapeutics. Available at: www.azcert.org. Accessed February 2010.
- ,.Cardiac arrhythmias—a clinical approach.Edinburgh:Mosby;2003.
- ,.The association between intravenous haloperidol and torsades de pointes—three cases and a literature review.Psychosomatics.1995;36:541–549.
- ,.Prolongation of the corrected QT and torsades de pointes cardiac arrhythmia associated with intravenous haloperidol in the medically ill.J Clin Psychopharmacol.1993;13(2):128–132.
- ,,,,.Torsades de pointes secondary to intravenous haloperidol after coronary bypass grafting surgery.Can J Anesth.2000;47(3):251–254.
- ,.Torsade de pointes associated with the administration of intravenous haloperidol: a review of the literature and practical guidelines for use,Expert Opin Drug Saf.2003;2(6):543–547.
- ,.Conduction disturbances associated with administration of butyrophenone antipsychotics in the critically ill: a review of the literature,Pharmacotherapy.1997;17(3):531–537.
- ,,.Haloperidol‐induced torsades de pointes.Ann Pharacother.1999;33(10):1046–1050.
- ,,, et al.Practice parameters for intravenous analgesia and sedation for adult patients in the intensive care unit: an executive summary,Crit Care Med.1995;23(9):1596–1600.
- ,,,,,.Prolonged cardiac repolarization after tacrolimus and haloperidol administration in the critically ill patient.Pharmacotherapy.2004;24(3):404–408.
- CPS Compendium of Pharmaceuticals and Specialties, the Canadian drug reference for health professionals, 2007, Canadian pharmacists association.
- Rote Liste Deutschland2008, Rote Liste Service GmbH Frankfurt am Main. Available at: www.rote‐liste.de. Accessed February 2010.
- ,.Delirium in the hospitalized patient: a primer for the pharmacist clinician.J Pharm Pract.2007;20(5):368–372.
- ,,, et al.Delirium: guidelines for general hospitals.J Psychosom Res.2007;62(3):371–383.
- ,,,.The pharmacological management of delirium in critical illness.Current Drug Therapy.2008,3:148–157.
- VIDAL‐l'information sur les produits de santé2008, Issy les Moulineaux Cedex. Available at: www.vidal.fr. Accessed February 2010.
- Haldol iniettabile—ufficiale monografia italiana. Available at: www. informatorefarmaceutico.it. Accessed February 2010.
- Arzneimittelkompendium der Schweiz2008, documed Verlag Basel. Availabla at: www.kompendium.ch. Accessed February 2010.
- ,,, et al.Clinical practice guidelines for the sustained use of sedatives and analgesics in the critically ill, American College of Critical Care Medicine ACCM, Society of Critical Care Medicine SCCM, American Society of Health‐System Pharmacists ASHP.Crit Care Med.2002;30(1):119–141.
- ,,,,.Is telemetry overused? Is it as helpful as thought?Cleve Clin J Med.2009;76(6):368–372.
- ,,.Telemetry outside critical care units: patterns of utilization and influence on management decisions.Clin Cardiol.1998;21(7):503–505.
- ,,.High‐dose intravenous haloperidol for agitated delirium in a cardiac patient on intra‐aortic balloon pump.J Clin Psychopharmacol.1991;11(2):146–147.
- .Haloperidol, midazolam and intravenous sedation.Aust NZ J Psychiatry.1999;33(6):942–943.
- BNF British National Formulary, compendium of pharmaceuticals and specialties of the UK.2007. Available at: www.bnf.org. Accessed February 2010.
- ,,,.Torsade de pointes associated with the use of intravenous haloperidol.Ann Intern Med.1993;119(5):391–394.
- ,.Torsades de pointes: potential consequence of intravenous haloperidol in the intensive care unit.Intensive Care World.1994;11(3):109–112.
- ,.Torsade de pointes caused by high‐dose intravenous haloperidol in cardiac patients.Clin Cardiol.1995;18:285–290.
- ,,.Continuous infusion of haloperidol controls agitation in critically ill patients.Crit Care Med.1994:22(3):433–440.
- ,,.Torsade de pointes complicating the treatment of bleeding esophageal varices: association with neuroleptics, vasopressin, and electrolyte imbalances.Am J Gastroenterol.1995;90(5):822–824.
- ,,,.Torsades de pointes associated with intravenous haloperidol in critically ill patients.Am J Cardiol.1998;81(2):238–240.
- ,,,,.The effect of intravenous haloperidol on QT interval dispersion in critically ill patients: comparison with QT interval prolongation for assessment of risk of torsades de pointes.J Clin Pharmacol.2001;41:1310–1318.
- ,,.Low‐dose haloperidol associated QTc prolongation.J Am Geriatr Soc.2008;56(10):1963–1964.
- ,,.Torsade de pointes following intravenous haloperidol administration in a patient with complete heart block.WMJ.2009;108(1):48–50.
CVS in an Adult Patient
We present a 26‐year‐old white male with a chief complaint of nausea and vomiting. The patient described prodromal nausea followed by intractable vomiting for 2 days. Over the past 2 years he has experienced similar episodes occurring every 3 to 6 months. He has been hospitalized 5 times for this problem with no diagnosis given. There are no obvious precipitants. The symptoms consistently last 2 to 3 days and resolve with supportive care including intravenous fluids and antiemetics. The patient enjoys good health between the periods of sickness. He has never experienced coffee‐ground emesis or hematemesis. His past medical history is significant for attention deficit disorder and cholecystectomy. He takes no prescription medications. Social history is remarkable for tobacco abuse, binge drinking on weekends, and daily marijuana use. He is unemployed. His family history is unremarkable.
Physical examination at the time of admission was notable for tachycardia, orthostatic hypotension, and hypoactive bowel sounds. Otherwise physical examination was normal.
Diagnostic testing done on admission was notable for white blood cell count of 25,000, hemoglobin of 17.3, blood urea nitrogen 18, creatinine 1.4, aspartate aminotransferase (AST) 64, and alanine aminotransferase (ALT) 55. Pancreatic enzymes and acute abdominal series were normal.
The patient was admitted to the hospital with the presumptive diagnosis of viral gastroenteritis. Initial therapy included intravenous fluids and promethazine. Throughout hospital day 1, he remained nauseated and had multiple bouts of emesis. Records from the patient's hospitalization 5 months ago were obtained and reviewed. During this previous hospitalization, computed tomography (CT) scans of the abdomen and esophagogastroduodenoscopy (EGD) were performed, both of which were negative. Upon review of this recent workup, the diagnosis of cyclic vomiting syndrome (CVS) was entertained and the patient received a therapeutic trial of subcutaneous sumatriptan. His symptoms abated dramatically. Subsequently, he was able to keep oral liquids down and his orthostatic hypotension resolved. On hospital day 2, his white blood cell count normalized without intervention. Blood, urine, and stool cultures remained negative, and workup for acute intermittent porphyria was negative. Upon discharge from the hospital he was counseled to discontinue all marijuana use and was scheduled for follow‐up in the residents' clinic. He failed to keep this appointment. After being lost to follow‐up for 17 months, he presented to the emergency department with nausea and vomiting. As before, his symptoms promptly improved with sumatriptan.
Discussion
CVS, initially described in 1861 as a pediatric illness, is being increasingly recognized in adults.1 It has been estimated that up to 1.6% of children experience symptoms consistent with this disorder, but the prevalence in adults is unknown.2 The essential features of CVS, as noted in our patient, are multiple discrete episodes of nausea and vomiting lasting less than 1 week with absence of nausea and vomiting between episodes. The presentation of adults with CVS often differs from the pediatric form in that adults have longer, less frequent episodes, and the triggers are less evident.3
The etiology and pathogenesis of CVS remain unknown. A variety of physical and psychological stresses, including infection, overexertion, and emotional distress, have been noted to precipitate episodes.4 CVS has variably been associated with autonomic, mitochondrial, and endocrine disorders. The most prevalent theory in the literature, however, is that CVS and migraine headaches are different presentations of the same diathesis.5 Patients with both are noted to have similar patterns of symptoms and positive family history of migraines. The progression from CVS to migraines is noted frequently in individual patients. As many as 82% of the 214 children in a case series of CVS were noted to have a family history of migraines or to have or subsequently develop migraines.6 In addition, electroencephalogram findings and adrenergic autonomic abnormalities are similar in both sets of patients.3 In 1 case series of 17 patients with CVS, patients noted the possible association of episodes with menses (in 57% of women of reproductive age), and the improvement of symptoms with sleep (in 24%), clinical factors common in patients with migraines.3
CVS is one of the functional gastrointestinal disorders for which the diagnosis is clinical, with criteria based upon the consensus of expert opinion in the Rome III Criteria for Functional Gastrointestinal (GI) Disorders.7 At least 3 months, with onset at least 6 months previously of:
-
Stereotypical episodes of vomiting regarding onset (acute) and duration (less than 1 week);
-
3 or more discrete episodes in the prior year; and
-
Absence of nausea and vomiting between episodes.
Supportive criteria: History of migraine headaches or family history of migraine headaches.7
Making the diagnosis of CVS requires the exclusion of other disorders associated with recurrent vomiting. Examples include gastric outlet or small bowel obstruction, gastroparesis, vestibular neuritis, elevated intracranial pressure, inborn errors of metabolism, dysautonomia, porphyria, and alterations in the hypothalamic pituitary adrenal axis. The other functional nausea and vomiting disorders described in Rome III, specifically chronic idiopathic nausea and functional vomiting, also need to be considered.7 Many drugs can cause nausea and vomiting, and chronic marijuana use has been associated with cyclical hyperemesis.8 Our patient meets the diagnostic criteria for CVS, but his frequent marijuana use would preclude a diagnosis of functional vomiting, which by definition requires an absence of chronic cannabinoid use.
Determining which tests and procedures should be performed in the initial evaluation is based on clinical judgment, but commonly includes complete metabolic profile, urinalysis, upper GI series, EGD, neurological imaging, acute abdominal series, and CT of the abdomen and pelvis. In addition, pertinent metabolic screening including serum lactate, cortisol, pyruvate, ammonia, creatinine phosphokinase, carnitine, urinary organic acids, and porphobilinogen may be considered.5
Evidence‐based treatment of CVS is limited by the lack of controlled trials. Acutely, patients often require hospitalization and symptom management with aggressive hydration, antiemetics, and sometimes even sedative agents. Empiric abortive treatment with antimigraine mediations (sumitriptan, prochlorperazine, tricyclic antidepressants, and ketorolac) has been effective in case reports.911 Patients in whom a history of chronic cannabinoid use is elicited should be counseled that cessation may lead to an improvement in symptoms.
Just as with migraines, patients who experience frequent episodes of cyclic vomiting can benefit from prophylactic medications. Tricyclic antidepressants (TCAs) have been reported to be effective as prophylactic agents in children with CVS.12 An open‐label treatment group of 17 adult patients with CVS noted that 17% of patients had a complete remission with TCA therapy and almost 60% had a partial response.3 More recently, a retrospective case series of patients who had failed TCAs as maintenance therapy reported that 15 out of the 20 patients studied had improvement in the frequency of their vomiting episodes with the newer antiepileptic drugs zonisamide and levetiracem. However, moderate or severe side effects were reported in 45%.13
Conclusions
In summary, although CVS is still an uncommon diagnosis, it is being made more frequently in adults. Although recognition is increasing, there remains a significant delay between onset of symptoms and diagnosis in adults.4 CVS is a diagnosis of exclusion and should be considered when initial evaluation for recurrent nausea and vomiting are unrevealing. A wide range of medications show benefit for both abortive and prophylactic therapy. Increasing awareness of this disorder can lead to a reduction in invasive and costly diagnostic workups.
- .Evrose de la digestion, caracteriseo par des crises periodiques de vomissements et une profonde modification de l'assimilation.Gazette Medicale de Paris1861:312. [French]
- ,.Cyclical vomiting syndrome in children: a population‐based study.J Pediatr Gastroenterol Nutr.1995;21(4):454–458.
- ,.Cyclic vomiting syndrome in adults: clinical features and response to tricyclic antidepressants.Am J Gastroenterol.1999;94(10):2855–2860.
- ,,,.Cyclic vomiting syndrome in 41 adults: the illness, the patients, and problems of management.BMC Med.2005;3:20.
- ,,.Consensus statement—2nd International Scientific Symposium on CVS. The Faculty of The 2nd International Scientific Symposium on Cyclic Vomiting Syndrome.Dig Dis Sci.1999;44(8 suppl):9S–11S.
- ,,,,.Is cyclic vomiting syndrome related to migraine?J Pediatr.1999;134(5):567–572.
- ,,, et al.Functional gastroduodenal disorders.Gastroenterology.2006;130:1466–1479.
- ,,,.Cannabinoid hyperemesis: cyclical hyperemesis in association with chronic cannabis use.Gut.2004;53:1566–1570.
- ,,.Sumatriptan in the treatment of cyclic vomiting.Ann Pharmacother.1995;29(10):997–999.
- .Recurrent vomiting in adults. A syndrome?Med J Aust.1983;1(7):329–331.
- ,,,,.Cyclic vomiting: association with multiple homeostatic abnormalities and response to ketorolac.Am J Gastroenterol.1996;91(10):2228–2232.
- .Effective prophylactic therapy for cyclic vomiting syndrome in children using amitriptyline or cyproheptadine.Pediatrics.1997;100(6):977–981.
- ,,,.Zonisamide or levetiracetam for adults with cyclic vomiting syndrome: a case series.Clin Gastroenterol Hepatol.2007;5(1):44–48.
We present a 26‐year‐old white male with a chief complaint of nausea and vomiting. The patient described prodromal nausea followed by intractable vomiting for 2 days. Over the past 2 years he has experienced similar episodes occurring every 3 to 6 months. He has been hospitalized 5 times for this problem with no diagnosis given. There are no obvious precipitants. The symptoms consistently last 2 to 3 days and resolve with supportive care including intravenous fluids and antiemetics. The patient enjoys good health between the periods of sickness. He has never experienced coffee‐ground emesis or hematemesis. His past medical history is significant for attention deficit disorder and cholecystectomy. He takes no prescription medications. Social history is remarkable for tobacco abuse, binge drinking on weekends, and daily marijuana use. He is unemployed. His family history is unremarkable.
Physical examination at the time of admission was notable for tachycardia, orthostatic hypotension, and hypoactive bowel sounds. Otherwise physical examination was normal.
Diagnostic testing done on admission was notable for white blood cell count of 25,000, hemoglobin of 17.3, blood urea nitrogen 18, creatinine 1.4, aspartate aminotransferase (AST) 64, and alanine aminotransferase (ALT) 55. Pancreatic enzymes and acute abdominal series were normal.
The patient was admitted to the hospital with the presumptive diagnosis of viral gastroenteritis. Initial therapy included intravenous fluids and promethazine. Throughout hospital day 1, he remained nauseated and had multiple bouts of emesis. Records from the patient's hospitalization 5 months ago were obtained and reviewed. During this previous hospitalization, computed tomography (CT) scans of the abdomen and esophagogastroduodenoscopy (EGD) were performed, both of which were negative. Upon review of this recent workup, the diagnosis of cyclic vomiting syndrome (CVS) was entertained and the patient received a therapeutic trial of subcutaneous sumatriptan. His symptoms abated dramatically. Subsequently, he was able to keep oral liquids down and his orthostatic hypotension resolved. On hospital day 2, his white blood cell count normalized without intervention. Blood, urine, and stool cultures remained negative, and workup for acute intermittent porphyria was negative. Upon discharge from the hospital he was counseled to discontinue all marijuana use and was scheduled for follow‐up in the residents' clinic. He failed to keep this appointment. After being lost to follow‐up for 17 months, he presented to the emergency department with nausea and vomiting. As before, his symptoms promptly improved with sumatriptan.
Discussion
CVS, initially described in 1861 as a pediatric illness, is being increasingly recognized in adults.1 It has been estimated that up to 1.6% of children experience symptoms consistent with this disorder, but the prevalence in adults is unknown.2 The essential features of CVS, as noted in our patient, are multiple discrete episodes of nausea and vomiting lasting less than 1 week with absence of nausea and vomiting between episodes. The presentation of adults with CVS often differs from the pediatric form in that adults have longer, less frequent episodes, and the triggers are less evident.3
The etiology and pathogenesis of CVS remain unknown. A variety of physical and psychological stresses, including infection, overexertion, and emotional distress, have been noted to precipitate episodes.4 CVS has variably been associated with autonomic, mitochondrial, and endocrine disorders. The most prevalent theory in the literature, however, is that CVS and migraine headaches are different presentations of the same diathesis.5 Patients with both are noted to have similar patterns of symptoms and positive family history of migraines. The progression from CVS to migraines is noted frequently in individual patients. As many as 82% of the 214 children in a case series of CVS were noted to have a family history of migraines or to have or subsequently develop migraines.6 In addition, electroencephalogram findings and adrenergic autonomic abnormalities are similar in both sets of patients.3 In 1 case series of 17 patients with CVS, patients noted the possible association of episodes with menses (in 57% of women of reproductive age), and the improvement of symptoms with sleep (in 24%), clinical factors common in patients with migraines.3
CVS is one of the functional gastrointestinal disorders for which the diagnosis is clinical, with criteria based upon the consensus of expert opinion in the Rome III Criteria for Functional Gastrointestinal (GI) Disorders.7 At least 3 months, with onset at least 6 months previously of:
-
Stereotypical episodes of vomiting regarding onset (acute) and duration (less than 1 week);
-
3 or more discrete episodes in the prior year; and
-
Absence of nausea and vomiting between episodes.
Supportive criteria: History of migraine headaches or family history of migraine headaches.7
Making the diagnosis of CVS requires the exclusion of other disorders associated with recurrent vomiting. Examples include gastric outlet or small bowel obstruction, gastroparesis, vestibular neuritis, elevated intracranial pressure, inborn errors of metabolism, dysautonomia, porphyria, and alterations in the hypothalamic pituitary adrenal axis. The other functional nausea and vomiting disorders described in Rome III, specifically chronic idiopathic nausea and functional vomiting, also need to be considered.7 Many drugs can cause nausea and vomiting, and chronic marijuana use has been associated with cyclical hyperemesis.8 Our patient meets the diagnostic criteria for CVS, but his frequent marijuana use would preclude a diagnosis of functional vomiting, which by definition requires an absence of chronic cannabinoid use.
Determining which tests and procedures should be performed in the initial evaluation is based on clinical judgment, but commonly includes complete metabolic profile, urinalysis, upper GI series, EGD, neurological imaging, acute abdominal series, and CT of the abdomen and pelvis. In addition, pertinent metabolic screening including serum lactate, cortisol, pyruvate, ammonia, creatinine phosphokinase, carnitine, urinary organic acids, and porphobilinogen may be considered.5
Evidence‐based treatment of CVS is limited by the lack of controlled trials. Acutely, patients often require hospitalization and symptom management with aggressive hydration, antiemetics, and sometimes even sedative agents. Empiric abortive treatment with antimigraine mediations (sumitriptan, prochlorperazine, tricyclic antidepressants, and ketorolac) has been effective in case reports.911 Patients in whom a history of chronic cannabinoid use is elicited should be counseled that cessation may lead to an improvement in symptoms.
Just as with migraines, patients who experience frequent episodes of cyclic vomiting can benefit from prophylactic medications. Tricyclic antidepressants (TCAs) have been reported to be effective as prophylactic agents in children with CVS.12 An open‐label treatment group of 17 adult patients with CVS noted that 17% of patients had a complete remission with TCA therapy and almost 60% had a partial response.3 More recently, a retrospective case series of patients who had failed TCAs as maintenance therapy reported that 15 out of the 20 patients studied had improvement in the frequency of their vomiting episodes with the newer antiepileptic drugs zonisamide and levetiracem. However, moderate or severe side effects were reported in 45%.13
Conclusions
In summary, although CVS is still an uncommon diagnosis, it is being made more frequently in adults. Although recognition is increasing, there remains a significant delay between onset of symptoms and diagnosis in adults.4 CVS is a diagnosis of exclusion and should be considered when initial evaluation for recurrent nausea and vomiting are unrevealing. A wide range of medications show benefit for both abortive and prophylactic therapy. Increasing awareness of this disorder can lead to a reduction in invasive and costly diagnostic workups.
We present a 26‐year‐old white male with a chief complaint of nausea and vomiting. The patient described prodromal nausea followed by intractable vomiting for 2 days. Over the past 2 years he has experienced similar episodes occurring every 3 to 6 months. He has been hospitalized 5 times for this problem with no diagnosis given. There are no obvious precipitants. The symptoms consistently last 2 to 3 days and resolve with supportive care including intravenous fluids and antiemetics. The patient enjoys good health between the periods of sickness. He has never experienced coffee‐ground emesis or hematemesis. His past medical history is significant for attention deficit disorder and cholecystectomy. He takes no prescription medications. Social history is remarkable for tobacco abuse, binge drinking on weekends, and daily marijuana use. He is unemployed. His family history is unremarkable.
Physical examination at the time of admission was notable for tachycardia, orthostatic hypotension, and hypoactive bowel sounds. Otherwise physical examination was normal.
Diagnostic testing done on admission was notable for white blood cell count of 25,000, hemoglobin of 17.3, blood urea nitrogen 18, creatinine 1.4, aspartate aminotransferase (AST) 64, and alanine aminotransferase (ALT) 55. Pancreatic enzymes and acute abdominal series were normal.
The patient was admitted to the hospital with the presumptive diagnosis of viral gastroenteritis. Initial therapy included intravenous fluids and promethazine. Throughout hospital day 1, he remained nauseated and had multiple bouts of emesis. Records from the patient's hospitalization 5 months ago were obtained and reviewed. During this previous hospitalization, computed tomography (CT) scans of the abdomen and esophagogastroduodenoscopy (EGD) were performed, both of which were negative. Upon review of this recent workup, the diagnosis of cyclic vomiting syndrome (CVS) was entertained and the patient received a therapeutic trial of subcutaneous sumatriptan. His symptoms abated dramatically. Subsequently, he was able to keep oral liquids down and his orthostatic hypotension resolved. On hospital day 2, his white blood cell count normalized without intervention. Blood, urine, and stool cultures remained negative, and workup for acute intermittent porphyria was negative. Upon discharge from the hospital he was counseled to discontinue all marijuana use and was scheduled for follow‐up in the residents' clinic. He failed to keep this appointment. After being lost to follow‐up for 17 months, he presented to the emergency department with nausea and vomiting. As before, his symptoms promptly improved with sumatriptan.
Discussion
CVS, initially described in 1861 as a pediatric illness, is being increasingly recognized in adults.1 It has been estimated that up to 1.6% of children experience symptoms consistent with this disorder, but the prevalence in adults is unknown.2 The essential features of CVS, as noted in our patient, are multiple discrete episodes of nausea and vomiting lasting less than 1 week with absence of nausea and vomiting between episodes. The presentation of adults with CVS often differs from the pediatric form in that adults have longer, less frequent episodes, and the triggers are less evident.3
The etiology and pathogenesis of CVS remain unknown. A variety of physical and psychological stresses, including infection, overexertion, and emotional distress, have been noted to precipitate episodes.4 CVS has variably been associated with autonomic, mitochondrial, and endocrine disorders. The most prevalent theory in the literature, however, is that CVS and migraine headaches are different presentations of the same diathesis.5 Patients with both are noted to have similar patterns of symptoms and positive family history of migraines. The progression from CVS to migraines is noted frequently in individual patients. As many as 82% of the 214 children in a case series of CVS were noted to have a family history of migraines or to have or subsequently develop migraines.6 In addition, electroencephalogram findings and adrenergic autonomic abnormalities are similar in both sets of patients.3 In 1 case series of 17 patients with CVS, patients noted the possible association of episodes with menses (in 57% of women of reproductive age), and the improvement of symptoms with sleep (in 24%), clinical factors common in patients with migraines.3
CVS is one of the functional gastrointestinal disorders for which the diagnosis is clinical, with criteria based upon the consensus of expert opinion in the Rome III Criteria for Functional Gastrointestinal (GI) Disorders.7 At least 3 months, with onset at least 6 months previously of:
-
Stereotypical episodes of vomiting regarding onset (acute) and duration (less than 1 week);
-
3 or more discrete episodes in the prior year; and
-
Absence of nausea and vomiting between episodes.
Supportive criteria: History of migraine headaches or family history of migraine headaches.7
Making the diagnosis of CVS requires the exclusion of other disorders associated with recurrent vomiting. Examples include gastric outlet or small bowel obstruction, gastroparesis, vestibular neuritis, elevated intracranial pressure, inborn errors of metabolism, dysautonomia, porphyria, and alterations in the hypothalamic pituitary adrenal axis. The other functional nausea and vomiting disorders described in Rome III, specifically chronic idiopathic nausea and functional vomiting, also need to be considered.7 Many drugs can cause nausea and vomiting, and chronic marijuana use has been associated with cyclical hyperemesis.8 Our patient meets the diagnostic criteria for CVS, but his frequent marijuana use would preclude a diagnosis of functional vomiting, which by definition requires an absence of chronic cannabinoid use.
Determining which tests and procedures should be performed in the initial evaluation is based on clinical judgment, but commonly includes complete metabolic profile, urinalysis, upper GI series, EGD, neurological imaging, acute abdominal series, and CT of the abdomen and pelvis. In addition, pertinent metabolic screening including serum lactate, cortisol, pyruvate, ammonia, creatinine phosphokinase, carnitine, urinary organic acids, and porphobilinogen may be considered.5
Evidence‐based treatment of CVS is limited by the lack of controlled trials. Acutely, patients often require hospitalization and symptom management with aggressive hydration, antiemetics, and sometimes even sedative agents. Empiric abortive treatment with antimigraine mediations (sumitriptan, prochlorperazine, tricyclic antidepressants, and ketorolac) has been effective in case reports.911 Patients in whom a history of chronic cannabinoid use is elicited should be counseled that cessation may lead to an improvement in symptoms.
Just as with migraines, patients who experience frequent episodes of cyclic vomiting can benefit from prophylactic medications. Tricyclic antidepressants (TCAs) have been reported to be effective as prophylactic agents in children with CVS.12 An open‐label treatment group of 17 adult patients with CVS noted that 17% of patients had a complete remission with TCA therapy and almost 60% had a partial response.3 More recently, a retrospective case series of patients who had failed TCAs as maintenance therapy reported that 15 out of the 20 patients studied had improvement in the frequency of their vomiting episodes with the newer antiepileptic drugs zonisamide and levetiracem. However, moderate or severe side effects were reported in 45%.13
Conclusions
In summary, although CVS is still an uncommon diagnosis, it is being made more frequently in adults. Although recognition is increasing, there remains a significant delay between onset of symptoms and diagnosis in adults.4 CVS is a diagnosis of exclusion and should be considered when initial evaluation for recurrent nausea and vomiting are unrevealing. A wide range of medications show benefit for both abortive and prophylactic therapy. Increasing awareness of this disorder can lead to a reduction in invasive and costly diagnostic workups.
- .Evrose de la digestion, caracteriseo par des crises periodiques de vomissements et une profonde modification de l'assimilation.Gazette Medicale de Paris1861:312. [French]
- ,.Cyclical vomiting syndrome in children: a population‐based study.J Pediatr Gastroenterol Nutr.1995;21(4):454–458.
- ,.Cyclic vomiting syndrome in adults: clinical features and response to tricyclic antidepressants.Am J Gastroenterol.1999;94(10):2855–2860.
- ,,,.Cyclic vomiting syndrome in 41 adults: the illness, the patients, and problems of management.BMC Med.2005;3:20.
- ,,.Consensus statement—2nd International Scientific Symposium on CVS. The Faculty of The 2nd International Scientific Symposium on Cyclic Vomiting Syndrome.Dig Dis Sci.1999;44(8 suppl):9S–11S.
- ,,,,.Is cyclic vomiting syndrome related to migraine?J Pediatr.1999;134(5):567–572.
- ,,, et al.Functional gastroduodenal disorders.Gastroenterology.2006;130:1466–1479.
- ,,,.Cannabinoid hyperemesis: cyclical hyperemesis in association with chronic cannabis use.Gut.2004;53:1566–1570.
- ,,.Sumatriptan in the treatment of cyclic vomiting.Ann Pharmacother.1995;29(10):997–999.
- .Recurrent vomiting in adults. A syndrome?Med J Aust.1983;1(7):329–331.
- ,,,,.Cyclic vomiting: association with multiple homeostatic abnormalities and response to ketorolac.Am J Gastroenterol.1996;91(10):2228–2232.
- .Effective prophylactic therapy for cyclic vomiting syndrome in children using amitriptyline or cyproheptadine.Pediatrics.1997;100(6):977–981.
- ,,,.Zonisamide or levetiracetam for adults with cyclic vomiting syndrome: a case series.Clin Gastroenterol Hepatol.2007;5(1):44–48.
- .Evrose de la digestion, caracteriseo par des crises periodiques de vomissements et une profonde modification de l'assimilation.Gazette Medicale de Paris1861:312. [French]
- ,.Cyclical vomiting syndrome in children: a population‐based study.J Pediatr Gastroenterol Nutr.1995;21(4):454–458.
- ,.Cyclic vomiting syndrome in adults: clinical features and response to tricyclic antidepressants.Am J Gastroenterol.1999;94(10):2855–2860.
- ,,,.Cyclic vomiting syndrome in 41 adults: the illness, the patients, and problems of management.BMC Med.2005;3:20.
- ,,.Consensus statement—2nd International Scientific Symposium on CVS. The Faculty of The 2nd International Scientific Symposium on Cyclic Vomiting Syndrome.Dig Dis Sci.1999;44(8 suppl):9S–11S.
- ,,,,.Is cyclic vomiting syndrome related to migraine?J Pediatr.1999;134(5):567–572.
- ,,, et al.Functional gastroduodenal disorders.Gastroenterology.2006;130:1466–1479.
- ,,,.Cannabinoid hyperemesis: cyclical hyperemesis in association with chronic cannabis use.Gut.2004;53:1566–1570.
- ,,.Sumatriptan in the treatment of cyclic vomiting.Ann Pharmacother.1995;29(10):997–999.
- .Recurrent vomiting in adults. A syndrome?Med J Aust.1983;1(7):329–331.
- ,,,,.Cyclic vomiting: association with multiple homeostatic abnormalities and response to ketorolac.Am J Gastroenterol.1996;91(10):2228–2232.
- .Effective prophylactic therapy for cyclic vomiting syndrome in children using amitriptyline or cyproheptadine.Pediatrics.1997;100(6):977–981.
- ,,,.Zonisamide or levetiracetam for adults with cyclic vomiting syndrome: a case series.Clin Gastroenterol Hepatol.2007;5(1):44–48.
Short Title
A long‐term tunneled subclavian venous catheter of a 32‐year‐old leukaemia patient blocked. Chest x‐ray (CXR) showed a fracture, with the proximal end underneath the first rib and clavicle (Figure 1, arrow, right panel), and distal fragment at the left hila (broken arrow). A CXR 3 months ago showed catheter kinking and narrowing at the same site, constituting the pinch‐off sign (arrow, left panel).1 The broken fragment was retrieved from the left pulmonary artery by cardiac catheterization. Fractured ends were smooth (central insert).
Spontaneous central venous catheter fracture occurs in 0.1% to 1% of cases.2 The catheter fracture is postulated to be related to compression between the clavicle and first rib due to vigorous movement or heavy object lifting,3 activities that should be avoided. Fractures at other sites are exceptional. The pinch‐off sign may precede fracture; if detected, catheter removal is warranted,4 a fact both clinicians and radiologists should be aware of.
- ,.The “pinch‐off sign”: a warning of impending problems with permanent subclavian catheters.Am J Surg.1984;148:633–636.
- ,,,.Spontaneous leak and transection of permanent subclavian catheters.J Surg Oncol.1998;68:166–168.
- ,,.Pinch‐off syndrome: case report and collective review of the literature.Am Surg.2004;70:635–644.
- ,.Prevention of complications in permanent central venous catheters.Surg Gynecol Obstet.1988;167:6–11.
A long‐term tunneled subclavian venous catheter of a 32‐year‐old leukaemia patient blocked. Chest x‐ray (CXR) showed a fracture, with the proximal end underneath the first rib and clavicle (Figure 1, arrow, right panel), and distal fragment at the left hila (broken arrow). A CXR 3 months ago showed catheter kinking and narrowing at the same site, constituting the pinch‐off sign (arrow, left panel).1 The broken fragment was retrieved from the left pulmonary artery by cardiac catheterization. Fractured ends were smooth (central insert).

Spontaneous central venous catheter fracture occurs in 0.1% to 1% of cases.2 The catheter fracture is postulated to be related to compression between the clavicle and first rib due to vigorous movement or heavy object lifting,3 activities that should be avoided. Fractures at other sites are exceptional. The pinch‐off sign may precede fracture; if detected, catheter removal is warranted,4 a fact both clinicians and radiologists should be aware of.
A long‐term tunneled subclavian venous catheter of a 32‐year‐old leukaemia patient blocked. Chest x‐ray (CXR) showed a fracture, with the proximal end underneath the first rib and clavicle (Figure 1, arrow, right panel), and distal fragment at the left hila (broken arrow). A CXR 3 months ago showed catheter kinking and narrowing at the same site, constituting the pinch‐off sign (arrow, left panel).1 The broken fragment was retrieved from the left pulmonary artery by cardiac catheterization. Fractured ends were smooth (central insert).

Spontaneous central venous catheter fracture occurs in 0.1% to 1% of cases.2 The catheter fracture is postulated to be related to compression between the clavicle and first rib due to vigorous movement or heavy object lifting,3 activities that should be avoided. Fractures at other sites are exceptional. The pinch‐off sign may precede fracture; if detected, catheter removal is warranted,4 a fact both clinicians and radiologists should be aware of.
- ,.The “pinch‐off sign”: a warning of impending problems with permanent subclavian catheters.Am J Surg.1984;148:633–636.
- ,,,.Spontaneous leak and transection of permanent subclavian catheters.J Surg Oncol.1998;68:166–168.
- ,,.Pinch‐off syndrome: case report and collective review of the literature.Am Surg.2004;70:635–644.
- ,.Prevention of complications in permanent central venous catheters.Surg Gynecol Obstet.1988;167:6–11.
- ,.The “pinch‐off sign”: a warning of impending problems with permanent subclavian catheters.Am J Surg.1984;148:633–636.
- ,,,.Spontaneous leak and transection of permanent subclavian catheters.J Surg Oncol.1998;68:166–168.
- ,,.Pinch‐off syndrome: case report and collective review of the literature.Am Surg.2004;70:635–644.
- ,.Prevention of complications in permanent central venous catheters.Surg Gynecol Obstet.1988;167:6–11.
Continuing Medical Education Program in
If you wish to receive credit for this activity, which begins on the next page, please refer to the website: www. blackwellpublishing.com/cme.
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to www.blackwellpublishing.com/cme.
-
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which begins on the next page, please refer to the website: www. blackwellpublishing.com/cme.
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to www.blackwellpublishing.com/cme.
-
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which begins on the next page, please refer to the website: www. blackwellpublishing.com/cme.
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to www.blackwellpublishing.com/cme.
-
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
Myth: LBBB Masks Hyperkalemia
An 80‐year‐old man with end‐stage renal disease requiring maintenance hemodialysis was admitted to the emergency department (ED) with complaints of fever, generalized fatigue, and lethargy. Presenting electrocardiogram (ECG) revealed normal sinus rhythm at 82 beats per minute (bpm), prolonged PR interval, complete left bundle branch block (LBBB) with wide QRS interval and tall T waves (Figure 1). A baseline ECG done 3 months ago also showed LBBB (Figure 2). In view of the underlying LBBB, changes in the presenting ECG were ignored.

Hemodialysis was planned for the patient. A few hours later, repeat ECG revealed a sine wave pattern suggestive of severe hyperkalemia (Figure 3). Laboratory results were available then and his serum potassium was found to be 6.8 mmol/L. He was started on insulin, dextrose, and calcium gluconate, but he developed cardiorespiratory arrest and died.
Retrospectively, looking at the presenting ECG (Figure 1), it was found that the PR interval was longer, the QRS was broader, and the T waves were taller and more peaked than the baseline ECG (Figure 2).
Discussion
Hyperkalemia is a true medical emergency with potential lethal consequences that must be treated accordingly.1, 2 It can be difficult to diagnose due to the paucity of distinctive signs and symptoms. Any ECG change due to hyperkalemia becomes an indication for stabilizing the myocardium with calcium infusion.
Often, the sequence of repolarization due to myocardial infarction is altered on ECG in patients with baseline LBBB, making it difficult to diagnose accurately. Although it is thought that changes due to electrolyte imbalances will also be masked by the presence of LBBB, there is no evidence supporting this in the literature. Hence, it is wrongly believed that LBBB masks changes due to hyperkalemia. It is important that in patients with suspected electrolyte imbalance, baseline ECG showing LBBB is compared to the presenting ECG. In our patient, the presenting ECG (Figure 1) might not look too impressive, but in comparison to the baseline ECG (Figure 2), the PR interval is longer, QRS is wider, and T waves are more peaked and taller. If the admitting physician had closely compared the presenting ECG (Figure 1) to the baseline ECG (Figure 2), the suspicion of hyperkalemia would have been high.
- ,,.Electrolyte disturbances. In:Marx JA,Hockberger RS,Walls RM, et al.,Rosen's Emergency Medicine: Concepts and Clinical Practice.5th ed. Vol2.St. Louis:Mosby;2002:1730–1731.
- ,.Hyperkalemia in hospitalized patients.Int Urol Nephrol.2000;32:177–180.
An 80‐year‐old man with end‐stage renal disease requiring maintenance hemodialysis was admitted to the emergency department (ED) with complaints of fever, generalized fatigue, and lethargy. Presenting electrocardiogram (ECG) revealed normal sinus rhythm at 82 beats per minute (bpm), prolonged PR interval, complete left bundle branch block (LBBB) with wide QRS interval and tall T waves (Figure 1). A baseline ECG done 3 months ago also showed LBBB (Figure 2). In view of the underlying LBBB, changes in the presenting ECG were ignored.


Hemodialysis was planned for the patient. A few hours later, repeat ECG revealed a sine wave pattern suggestive of severe hyperkalemia (Figure 3). Laboratory results were available then and his serum potassium was found to be 6.8 mmol/L. He was started on insulin, dextrose, and calcium gluconate, but he developed cardiorespiratory arrest and died.

Retrospectively, looking at the presenting ECG (Figure 1), it was found that the PR interval was longer, the QRS was broader, and the T waves were taller and more peaked than the baseline ECG (Figure 2).
Discussion
Hyperkalemia is a true medical emergency with potential lethal consequences that must be treated accordingly.1, 2 It can be difficult to diagnose due to the paucity of distinctive signs and symptoms. Any ECG change due to hyperkalemia becomes an indication for stabilizing the myocardium with calcium infusion.
Often, the sequence of repolarization due to myocardial infarction is altered on ECG in patients with baseline LBBB, making it difficult to diagnose accurately. Although it is thought that changes due to electrolyte imbalances will also be masked by the presence of LBBB, there is no evidence supporting this in the literature. Hence, it is wrongly believed that LBBB masks changes due to hyperkalemia. It is important that in patients with suspected electrolyte imbalance, baseline ECG showing LBBB is compared to the presenting ECG. In our patient, the presenting ECG (Figure 1) might not look too impressive, but in comparison to the baseline ECG (Figure 2), the PR interval is longer, QRS is wider, and T waves are more peaked and taller. If the admitting physician had closely compared the presenting ECG (Figure 1) to the baseline ECG (Figure 2), the suspicion of hyperkalemia would have been high.
An 80‐year‐old man with end‐stage renal disease requiring maintenance hemodialysis was admitted to the emergency department (ED) with complaints of fever, generalized fatigue, and lethargy. Presenting electrocardiogram (ECG) revealed normal sinus rhythm at 82 beats per minute (bpm), prolonged PR interval, complete left bundle branch block (LBBB) with wide QRS interval and tall T waves (Figure 1). A baseline ECG done 3 months ago also showed LBBB (Figure 2). In view of the underlying LBBB, changes in the presenting ECG were ignored.


Hemodialysis was planned for the patient. A few hours later, repeat ECG revealed a sine wave pattern suggestive of severe hyperkalemia (Figure 3). Laboratory results were available then and his serum potassium was found to be 6.8 mmol/L. He was started on insulin, dextrose, and calcium gluconate, but he developed cardiorespiratory arrest and died.

Retrospectively, looking at the presenting ECG (Figure 1), it was found that the PR interval was longer, the QRS was broader, and the T waves were taller and more peaked than the baseline ECG (Figure 2).
Discussion
Hyperkalemia is a true medical emergency with potential lethal consequences that must be treated accordingly.1, 2 It can be difficult to diagnose due to the paucity of distinctive signs and symptoms. Any ECG change due to hyperkalemia becomes an indication for stabilizing the myocardium with calcium infusion.
Often, the sequence of repolarization due to myocardial infarction is altered on ECG in patients with baseline LBBB, making it difficult to diagnose accurately. Although it is thought that changes due to electrolyte imbalances will also be masked by the presence of LBBB, there is no evidence supporting this in the literature. Hence, it is wrongly believed that LBBB masks changes due to hyperkalemia. It is important that in patients with suspected electrolyte imbalance, baseline ECG showing LBBB is compared to the presenting ECG. In our patient, the presenting ECG (Figure 1) might not look too impressive, but in comparison to the baseline ECG (Figure 2), the PR interval is longer, QRS is wider, and T waves are more peaked and taller. If the admitting physician had closely compared the presenting ECG (Figure 1) to the baseline ECG (Figure 2), the suspicion of hyperkalemia would have been high.
- ,,.Electrolyte disturbances. In:Marx JA,Hockberger RS,Walls RM, et al.,Rosen's Emergency Medicine: Concepts and Clinical Practice.5th ed. Vol2.St. Louis:Mosby;2002:1730–1731.
- ,.Hyperkalemia in hospitalized patients.Int Urol Nephrol.2000;32:177–180.
- ,,.Electrolyte disturbances. In:Marx JA,Hockberger RS,Walls RM, et al.,Rosen's Emergency Medicine: Concepts and Clinical Practice.5th ed. Vol2.St. Louis:Mosby;2002:1730–1731.
- ,.Hyperkalemia in hospitalized patients.Int Urol Nephrol.2000;32:177–180.
Hospitalists in the AHSC
The successful integration of hospitalists in academic health science centers (AHSCs) has been identified as one of the most challenging areas for the hospitalist movement.1, 2 This has been based on a concern that many hospitalists lack academic and research skills, lack mentorship, and may have little time to develop academic careers because of the significant time they spend in clinical care.
A recent survey highlighted that the pediatric hospitalist workforce is in its infancy and additional perspectives, such as from hospitalists themselves, are essential for a more complete picture of the current state of pediatric hospital medicine.3 Hospitalists have had a long history in Canada.4 The Hospital for Sick Children, Toronto, Canada, has had a Division of Pediatric Medicine since 1981, with hospitalists, as we now know them, from inception. This provided a rich resource to explore pediatric hospital medicine in the academic context and from hospitalists themselves. The objective of this survey was to explore the characteristics, practice, and perceptions of pediatric hospital medicine in an AHSC. Locally, we hoped the results would inform the division on program development, training, and faculty career development. Externally, the findings could contribute to a body of knowledge on the evolving role of pediatric hospitalists and provide insight into opportunities for better integration into AHSCs.
Methods
Study Design
This was a cross‐sectional survey of pediatricians who attend on the pediatric medicine inpatient unit at the Hospital for Sick Children, Toronto. The study protocol was approved by Quality and Risk Management at The Hospital for Sick Children.
Setting
The Hospital for Sick Children is a tertiary care children's hospital affiliated with the University of Toronto, Toronto, Canada. The total hospital bed capacity including intensive care unit beds and subspecialty beds is 320. It is the only free‐standing pediatric hospital for the greater Toronto area with a catchment population of 5 million people.
A formal division for general pediatrics, Pediatric Medicine, has been in existence since 1981 with hospital‐based pediatricians (who now are known as hospitalists) who attend on the inpatient unit. The pediatric medicine inpatient unit (PMIU) has a 60‐bed capacity on 3 units. At all times, the PMIU is staffed by 4 to 6 inpatient attendings from a total of 20 full‐time and major part‐time pediatricians. According to Wachter's staging of hospital care, the PMIU is at stage IV of IV in development (mandatory care by hospitalist).5 All attending pediatricians are members of the Division of Pediatric Medicine. Pediatric subspecialists do not attend on the PMIU. Physicians attend for a minimum of 4 weeks at a time. General pediatric house staff including fellows, residents (postgraduate year [PGY] 1, PGY3) and medical students are supervised by the attending pediatricians. Other inpatient clinical services provided include an inpatient general medical consultation team, a complex care team for inpatients with chronic complex conditions, and a consultative and collaborative role with the team of interventional radiologists. An outpatient pediatric consultation program exists for hospital follow‐up, general pediatric consultation, and specialized generalist care for specific populations of children.
In addition to the clinical program, research and education programs exist within the division. The division has had an academic general pediatric fellowship program since 1992, with the majority of most recent graduates obtaining academic hospitalist faculty positions. A formal research group and infrastructure for clinical, outcomes‐based research within the division, known as the Pediatric Outcomes Research Team (PORT), exists.
Study Population
All pediatricians who attend on the PMIU and in the Division of Pediatric Medicine, Hospital for Sick Children in 2007 were eligible for recruitment including the 2 eligible study authors.
Survey Instrument
We constructed a 43‐item structured questionnaire that asked about and explored training and employment characteristics; clinical roles and nonclinical roles (teaching, quality, research, leadership); and perceptions of hospital medicine and a career in this field. Several items were modeled after questions administered to program directors in a survey reported by Freed et al.3 The questionnaire was designed to be completed by the respondent on paper.
Questionnaire Administration
From September to December 2007, eligible pediatricians were sent a letter explaining the study and the questionnaire through interhospital mail by the research coordinator. Questionnaires were deidentified and assigned a unique identification number. Only the research coordinator had access to the list that linked the unique identification numbers with names. Confidentiality of responses and anonymity of responses was explicitly stated in the letter explaining the study to pediatricians. Nonrespondents were contacted by the research assistant at 3‐week intervals with personal reminders.
Data Analysis
During the analysis phase, responses remained deidentified. Descriptive statistics using means, medians, and proportions were calculated for survey items. For open‐ended questions, answers from respondents were summarized into key concepts or themes by the 3 study authors. Excerpts from responses were abstracted to highlight themes.
Results
Eighteen of 20 (90%) faculty responded to the questionnaire. The results are presented by the domains of the questionnaire, as follows.
Training and Employment Characteristics
Due to the requirements of the Royal College of Physicians and Surgeons of Canada, all faculty had a minimum of 4 years of pediatric specialty training leading to certification (Table 1). Seven of 18 (39%) had fellowship and graduate training. Nine of 18 (50%) had been on faculty for greater than 5 years.
| Training after medical schools, median years (range) | 4.8 (46) |
| Fellowship trained, number (%) | 7 (39) |
| Fellowship duration, median years (range) | 2.7 (15) |
| Graduate studies, number (%) | 7 (39) |
| Clinical epidemiology | 5 |
| Education | 2 |
| Duration since first academic appointment | |
| Median, years (range) | 5.5 (030) |
| Mean, years (standard deviation) | 9.9 (10.4) |
| >5 years, number (%) | 9 (50) |
| Academic rank, number (%) | |
| Lecturer | 2 (11) |
| Assistant professor | 10 (56) |
| Associate professor | 4 (22) |
| Full professor | 1 (6) |
Hospitalists: Impact and Definition
Seventeen of 18 (94%) faculty felt that hospitalists reduce cost, increase patient satisfaction, and increase quality of care. Three of 18 (17%) felt that hospitalism adversely affects the primary care physicianpatient relationship. All felt that hospitalists can contribute to the academic mission of an AHSC.
Most (17/18) felt that the Society of Hospital Medicine definition of a hospitalist is useful but 13 of 18 (72%) felt that it was important to develop an international consensus‐based definition.
Clinical Roles
All faculty attended on the PMIU. Other clinical activities included: 13 of 18 (72%) inpatient general medical consultation, 9 of 18 (50%) interventional radiology clinical team, 8 of 18 (44%) attended in the pediatric consultation clinic.
The median number of weeks attending on the PMIU was 16 (range, 440 weeks).
Nine of 18 (50%) provided leadership in clinical programs or the care of specific populations of children. This included leadership in healthcare systems: director of PMIU, director of inpatient general medical consultation, interventional radiology comanagement team; and leadership in patient populations: children with chronic complex conditions, cyclic vomiting, cancer/genetic syndromes, obesity, child abuse and neglect, failure to thrive, and vascular tumors.
Nonclinical and Academic Roles
Sixteen (89%) considered their job to include activities in addition to their role of providing patient care (Table 2). This included primary activity in clinical research, education, quality improvement and health policy.
| Area of Activity | n = 18 [number (%)] |
|---|---|
| |
| QI | |
| Participates in QI activity | 12 (67) |
| Participants who lead in QI activity | 4 (33) |
| Education | |
| Teaching | |
| Medical students | |
| Inpatient unit | 18 (100) |
| Small group sessions | 10 (56) |
| Lectures | 15 (83) |
| Residents | |
| Inpatient unit | 17 (94) |
| Lectures | 12 (67) |
| EBM critical appraisal course | 5 (28) |
| General pediatric fellows | 7 (39) |
| CME | 11 (61) |
| Curriculum or program development | 9 (50) |
| Research | |
| Holds appointment with research institute | 12 (66) |
| Peer review publication in past 12 months | 13 (72) |
| Currently holds research grant | 12 (66) |
| Presented abstract at 2007 PAS meeting | 10 (56) |
Hospitalist Medicine as a Career: Perceptions
Fifteen (83%) felt that it was important to establish an annual minimum time allocation to practicing hospital medicine. A median of 11 weeks per year (range, 816 weeks) was felt to be the minimum time that should be allocated to practicing hospital medicine. The major themes related to the need to establish a minimum time for clinical practice in hospital medicine were as follows: maintenance of skills, knowledge, and competency; ensuring quality of care; and efficiency of care. One respondent explained that the main reason to have a minimum time for clinical practice was to keep up clinical skills of acute patients, stay aware of and keep up to date of available facilities in the hospital.
Thirteen (72%) felt that it was important to establish an annual maximum time allocation to practicing hospital medicine. A median of 32 weeks per year (range, 2036 weeks) was felt to be the maximum time that should be allocated to practicing hospital medicine. The major themes related to the need to establish a maximum time for clinical practice in hospital medicine were: burnout, balance in career, and desire to develop academic career and to complete nonclinical activities. As this respondent described: I think hospital medicine can be very emotionally difficult and is sustainable if provided time off to pursue other goals such as research, education, creative professional service.
Thirteen (72%) intended to continue their career as a hospitalist and 5 (28%) were unsure. Eleven (61%) felt that a job as a hospitalist was a viable long term career, 6 (33%) were unsure, and 1 (5%) felt that it was not viable. When asked what the barriers to establishing hospital medicine as a long‐term career, the major themes that emerged were as follows: burnout, establishing a nonclinical or academic niche, and the system for career advancement in an academic center. This respondent explained: As long as you have another niche (education for me) that helps provide respite, as well as [you need an] opportunity to have protected time for academic endeavours that are necessary to survive in an academic health science centre.
Another respondent described burnout as a barrier: long hours, emotionally draining with very complex patients, feeling stretchedclinical care and teaching and research and admin.
Perceived advantages to a career as a hospitalist included: working in a team, generalist approach to care, stability relative to community practice, intellectually stimulating and rewarding work, growing area and opportunities for scholarship. One respondent outlined the perceived advantages to a generalist approach: diversity of work, become a generalist specialist, teamwork, develop broad perspective on health.
Others described the perceived advantages over community practice: exciting, interesting, job stability, salary and no worries about administration of the business of community office, stable income, holiday leave.
Perceived disadvantages to a career as a hospitalist included: burnout, recognition and respect, and lack of long‐term relationships with patients. One respondent explained: as an emerging field, we have a lot of growing to do. Although our work (both clinical and academic) seem fundamental to the mission of the hospital and university, we may not feel we receive the respect, support, resources, funding allocated to other areas.
Another response to disadvantages of a career as a hospitalist was, burnout, risk of being seen as a perpetual resident.
Discussion
Freed et al.3 recently conducted a survey of U.S. pediatric hospitalist program directors from a diverse range of settings (ie, teaching vs. nonteaching, free‐standing vs. hospital system, children's hospitals vs. non‐children's hospitals).3 These investigators found that the majority of programs had employed hospitalists for less than 5 years (compared with our program, 30 years); employed 1 to 5 hospitalists (compared with our program, 20); and 25% of programs indicated their hospitalists averaged greater than 5 years on the job (compared with our program, average 10 years on the job). Maniscalco et al.6 conducted a survey in 2007 of hospitalists in a similarly diverse range of settings, found that the mean number of years on the job was 6 and found similar clinical and teaching roles. They also found that the need for advanced training in administration, research, education, and quality improvement was high. Further, we were able to examine academic roles and perceptions of hospital medicine as a career in an AHSC at an individual level. This survey, however, was limited by sampling from a single institution.
Almost all faculty identified an area of focus in addition to clinical care. Educational activities occurred at all levels: undergraduate, residency, fellowship, and continuing medical education. Faculty were engaged in research activities. Hospitalists provide care on all inpatient units as a consultant specialist in general medical care. For example, we have designed a collaborative model of care with the interventional radiology team to comanage children who require image‐guided interventions, such as gastrostomy, chest tube, and central venous line insertions.7 One further area that deserves mention is the leadership of hospitalists in outpatient care of children, especially hospital intense populations, in collaboration with their primary care provider. These groups of children are often medically and socially complex, require repeated and intense hospital resources (including diagnostic testing, subspecialty consultation, and hospitalization), and require generalist care to manage them from a family centered perspective.
A significant proportion of the faculty in this survey acquired advanced academic training. The formal training of hospitalist physicians is in its infancy. A recent work documenting the domains of training for academic general pediatric fellowship in leadership, education, and research seems to be most appropriate for the nonclinical foundation for pediatric academic hospitalists.8
Few studies have examined academic hospitalists' perceptions on the minimum and maximum time per year suitable for clinical service. This undoubtedly will vary depending on the institution, program and financial structure, patient load and complexity, call requirements, academic commitments, and stage of development. Faculty surveyed in this study felt a range of 11 to 32 weeks of clinical inpatient attending per year was ideal. This is consistent with the expert panel recommendations of the Society of Hospital Medicine. What may be equally important to determine is the maximum number of continuous weeks attending on the PMIU.
There have been 3 full‐time faculty who have left the division (all to community hospitalbased generalist practices with academic affiliations) and 1 who has changed from a full‐time to a part‐time role in the division. Most faculty surveyed intended to continue their career as a hospitalist. They identified several positive and satisfying aspects to the career, including relationships with peers, stable salary, numerous opportunities for scholarly work in a young field, and generalist care. Hoff et al.9 described a national US survey of hospitalists in all adult medicine settings that examined personal characteristics, and work‐related attitudes. Similarly, they found that hospital medicine was a source of positive social and professional work experiences related to interactions with peers, patients and families, and coworkers. In the current study, perceived disadvantages to a hospitalist career were burnout, lack of recognition and respect, and lack of long‐term relationships with patients. Hoff et al.9 noted that 37% were burnt out or at risk of burnout, which is less than in the fields of critical care medicine and emergency medicine.
The identified barriers to establishing a career were related to development of an academic focus, balance between clinical and nonclinical time, and the system for career advancement. Few other studies have examined these career issues for hospitalists in the academic setting. Several authors have discussed career issues for clinician‐educators in the US,10, 11 including metrics for promotion and recognition by institutions. Alternate methods have been proposed for promotion, aside from research and education, such as creative professional activity or clinical excellence.12 The developing field of hospital medicine faces similar challenges as individual hospitalists and the specialty itself works to align with the academic mission.1315
The division and hospitalist program have evolved over more than 2 decades to fulfill strategic goals and respond to changing external factors (Table 3). Contextual factors that have supported this evolution and that may be unique to our academic environment merit mention. First, the departments' physicians work in a within a single‐payer universal healthcare system that in some ways is similar to a single‐payer health maintenance organization. The ultimate governance is provided by the provincial Ministry of Health, which is funded through taxation. Second, through an alternative funding plan (AFP) with the provincial government, block funding is providing in lieu of fee for service clinical care that funds physician salaries for clinical work, research, education, and administrative activities.16 Third, the department has a career development compensation program (CDCP) that has an explicit job activity profile which is aligned with the role of hospitaliststhe clinician‐specialist profilewho have a predominate commitment to provide, advance, and promote excellence in clinical care with contributions to education and/or research.16 The compensation and evaluation process for hospitalists is the same as other members in the department. While further refinement of this system is ongoing, this program has demonstrated a support for all roles (ie, clinical, education, and research).17
| Date | Area | Pressure Point | Change |
|---|---|---|---|
| 1981 | Clinical education research | Department priority for academic generalism | Creation of Division of General Pediatrics (now Pediatric Medicine); full‐time hospital‐based pediatricians attending on inpatient unit |
| 1991 | Education | Division priority to foster academic generalism and train future generation of academic generalists | Creation of academic general pediatrics fellowship program |
| 1992 | Research | Division priority to foster clinical, outcomes‐based research | Creation of formal divisional research infrastructure with foundation support for an epidemiologic, outcomes‐based research platform; pediatric outcomes research team (PORT) |
| 1995 | Clinical care | Province‐wide reduction in resident duty hours; division priority to raise the bar for clinical and teaching excellence in hospitalist‐and community‐based pediatrics; need for a financially viable and cost‐effective model for staffing attending pediatricians on inpatient unit | Reorganization of inpatient unit; higher proportion of attending pediatricians who are full‐time, hospital‐based; creation of a staff‐only hospitalist unit19; creation of a section of community pediatrics (2001) |
| 1996 | Clinical care education faculty development | Need to limit attending hospitalists after‐hours clinical care to ensure balance and academic productivity; need for a system of after‐hours physician coverage for inpatient care that is sustainable and financially viable; need for funding sources for academic general pediatrics fellowship | Creation of a clinical departmental fellowship program to fund after hours clinical coverage with qualified pediatricians seeking additional fellowship training20 |
| 1998 | Faculty career development | Need to value and reward all academic contributions, such as the hospitalist role, through an explicit job activity profile within the department16 | Implementation of a career development and compensation program with the clinician specialist role whose primary contributions are to excellence in clinical care |
| 1999 | Clinical care | Expansion of hospital interventional radiology program and need for high quality collaborative care | Comanagement model with hospitalist‐radiologist team7 |
| 20068 | Clinical care research | Division and institution priority to provide high‐quality care for children with complex care health issues and foster a research program21 | Creation of formalized hospitalist complex care program with inpatient and outpatient care22; research support for complex care |
| 2007 | Clinical care | Refinement of inpatient unit organization to improve efficiency of care; increase number of trainees23 | Reorganization of inpatient units to geographic allocation of patients by attending physician; addition of trainees to staff‐only hospitalist unit |
Furthermore, several divisional factors have contributed to the viability of hospitalism within our generalist division. First, hospitalists were integrated into, rather than segregated from the division. Second, hospitalists have the opportunity to engage in diverse clinical activities. Wachter and Goldman18 advocate for hospitalist participation in outpatient care to provide variety and to cement their relationship with their generalist division. Third, a fellowship training program was established in 1992 that integrated principles of academic general pediatrics and hospitalism. Fourth, career development in education, research, and, more recently, quality improvement is fostered.
In summary, the faculty of an established pediatric hospitalist program have diverse and unique clinical, leadership, and scholarly contributions to the academic mission of the department. In order to further promote integration, several issues should be addressed, including optimal training, time allocated to nonclinical activities, systems for career development and promotion of hospitalist faculty, and mentorship. Finally, it is important that leaders in pediatric hospital medicine and general pediatrics engage the larger academic community to strengthen the role and contributions of hospitalists in AHSCs.
Acknowledgements
The authors thank the faculty of the Division of Pediatric Medicine, Hospital for Sick Children for participating in the survey and past and present faculty for their contributions to the development of the division.
- ,.Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- .The hospitalist movement—time to move on.N Engl J Med.2007;25:2627–2629.
- ,,.Characteristics of the pediatric hospitalist workforce: its roles and work environment.Pediatrics.2007;120:33–39.
- .A Canadian perspective on the American hospitalist movement.Arch Int Med.1999;159:1666–1668.
- .An introduction to the hospitalist model.Ann Intern Med.1999;130:338–342.
- ,,,,.Current roles and training needs of pediatric hospitalists: a study from the Pediatric Research in Inpatient Settings (PRIS) network.Paper presented at: Pediatric Academic Society Annual Meeting; May2008;Honolulu, HI. E‐PAS2008:6725.4.
- ,.The pediatric hospitalist and interventional radiologist: a model for clinical care in pediatric interventional radiology.J Vasc Interv Radiol.2006;17:1733–1738.
- ,,, et al.Academic general pediatric fellowships: curriculum design and educational goals and objectives.Ambul Pediatr.2007;7:328–339.
- ,,,,.Characteristics and work experiences of hospitalists in the United States.Arch Intern Med.2001;161:851–858.
- ,.Mission critical—integrating clinician‐educators into academic medical centers.N Engl J Med.1999;341:840–843.
- ,,,.Separate and equitable promotion tracks for clinician‐educators.JAMA.2005;294:1101–1103.
- ,,.Creative professional activity: an additional platform for promotion of faculty.Acad Med.2006;81:568–570.
- .Helping hospitalists achieve academic stature.J Hosp Med.2008;3:285–287.
- ,,,.An innovative approach to supporting hospitalist physicians toward academic success.J Hosp Med.2008;3:314–318.
- ,,,.The University of Michigan Specialist‐Hospitalist Allied Research Program: jumpstarting hospitalist medicine research.J Hosp Med.2008;3:308–313.
- ,,,,,.Evaluation of a peer‐reviewed career development and compensation program for physicians at an academic health science centre.Pediatrics.2003;111:e26–e31.
- ,,,,.Performance of a career development and compensation program at an academic health science center.Pediatrics.2007;119:e791–e797.
- ,.Implications of the hospitalist movement for academic departments of medicine: lessons from the UCSF experience.Am J Med.1999;106:127–133.
- ,,,.Evaluation of a staff‐only hospitalist system in a tertiary care, academic children's hospital.Pediatrics.2004;114:1545–1549.
- ,.Providing after‐hours on‐call clinical coverage in academic health sciences centres: the Hospital for Sick Children experience.CMAJ.2000;163:298–299.
- ,,,,.A home for medically complex children: the role of hospital programs.J Healthc Qual.2008;30:7–15.
- ,,.Comprehensive care for medically complex children: the pediatric nurse practitioner‐hospitalist model of collaborative care.Hosp Pediatr.2009;1:20–23.
- ,,.Pediatric hospitalist medicine: an overview and a perspective from Toronto, Canada.Clin Pediatr (Phila).2008;47:546–548.
The successful integration of hospitalists in academic health science centers (AHSCs) has been identified as one of the most challenging areas for the hospitalist movement.1, 2 This has been based on a concern that many hospitalists lack academic and research skills, lack mentorship, and may have little time to develop academic careers because of the significant time they spend in clinical care.
A recent survey highlighted that the pediatric hospitalist workforce is in its infancy and additional perspectives, such as from hospitalists themselves, are essential for a more complete picture of the current state of pediatric hospital medicine.3 Hospitalists have had a long history in Canada.4 The Hospital for Sick Children, Toronto, Canada, has had a Division of Pediatric Medicine since 1981, with hospitalists, as we now know them, from inception. This provided a rich resource to explore pediatric hospital medicine in the academic context and from hospitalists themselves. The objective of this survey was to explore the characteristics, practice, and perceptions of pediatric hospital medicine in an AHSC. Locally, we hoped the results would inform the division on program development, training, and faculty career development. Externally, the findings could contribute to a body of knowledge on the evolving role of pediatric hospitalists and provide insight into opportunities for better integration into AHSCs.
Methods
Study Design
This was a cross‐sectional survey of pediatricians who attend on the pediatric medicine inpatient unit at the Hospital for Sick Children, Toronto. The study protocol was approved by Quality and Risk Management at The Hospital for Sick Children.
Setting
The Hospital for Sick Children is a tertiary care children's hospital affiliated with the University of Toronto, Toronto, Canada. The total hospital bed capacity including intensive care unit beds and subspecialty beds is 320. It is the only free‐standing pediatric hospital for the greater Toronto area with a catchment population of 5 million people.
A formal division for general pediatrics, Pediatric Medicine, has been in existence since 1981 with hospital‐based pediatricians (who now are known as hospitalists) who attend on the inpatient unit. The pediatric medicine inpatient unit (PMIU) has a 60‐bed capacity on 3 units. At all times, the PMIU is staffed by 4 to 6 inpatient attendings from a total of 20 full‐time and major part‐time pediatricians. According to Wachter's staging of hospital care, the PMIU is at stage IV of IV in development (mandatory care by hospitalist).5 All attending pediatricians are members of the Division of Pediatric Medicine. Pediatric subspecialists do not attend on the PMIU. Physicians attend for a minimum of 4 weeks at a time. General pediatric house staff including fellows, residents (postgraduate year [PGY] 1, PGY3) and medical students are supervised by the attending pediatricians. Other inpatient clinical services provided include an inpatient general medical consultation team, a complex care team for inpatients with chronic complex conditions, and a consultative and collaborative role with the team of interventional radiologists. An outpatient pediatric consultation program exists for hospital follow‐up, general pediatric consultation, and specialized generalist care for specific populations of children.
In addition to the clinical program, research and education programs exist within the division. The division has had an academic general pediatric fellowship program since 1992, with the majority of most recent graduates obtaining academic hospitalist faculty positions. A formal research group and infrastructure for clinical, outcomes‐based research within the division, known as the Pediatric Outcomes Research Team (PORT), exists.
Study Population
All pediatricians who attend on the PMIU and in the Division of Pediatric Medicine, Hospital for Sick Children in 2007 were eligible for recruitment including the 2 eligible study authors.
Survey Instrument
We constructed a 43‐item structured questionnaire that asked about and explored training and employment characteristics; clinical roles and nonclinical roles (teaching, quality, research, leadership); and perceptions of hospital medicine and a career in this field. Several items were modeled after questions administered to program directors in a survey reported by Freed et al.3 The questionnaire was designed to be completed by the respondent on paper.
Questionnaire Administration
From September to December 2007, eligible pediatricians were sent a letter explaining the study and the questionnaire through interhospital mail by the research coordinator. Questionnaires were deidentified and assigned a unique identification number. Only the research coordinator had access to the list that linked the unique identification numbers with names. Confidentiality of responses and anonymity of responses was explicitly stated in the letter explaining the study to pediatricians. Nonrespondents were contacted by the research assistant at 3‐week intervals with personal reminders.
Data Analysis
During the analysis phase, responses remained deidentified. Descriptive statistics using means, medians, and proportions were calculated for survey items. For open‐ended questions, answers from respondents were summarized into key concepts or themes by the 3 study authors. Excerpts from responses were abstracted to highlight themes.
Results
Eighteen of 20 (90%) faculty responded to the questionnaire. The results are presented by the domains of the questionnaire, as follows.
Training and Employment Characteristics
Due to the requirements of the Royal College of Physicians and Surgeons of Canada, all faculty had a minimum of 4 years of pediatric specialty training leading to certification (Table 1). Seven of 18 (39%) had fellowship and graduate training. Nine of 18 (50%) had been on faculty for greater than 5 years.
| Training after medical schools, median years (range) | 4.8 (46) |
| Fellowship trained, number (%) | 7 (39) |
| Fellowship duration, median years (range) | 2.7 (15) |
| Graduate studies, number (%) | 7 (39) |
| Clinical epidemiology | 5 |
| Education | 2 |
| Duration since first academic appointment | |
| Median, years (range) | 5.5 (030) |
| Mean, years (standard deviation) | 9.9 (10.4) |
| >5 years, number (%) | 9 (50) |
| Academic rank, number (%) | |
| Lecturer | 2 (11) |
| Assistant professor | 10 (56) |
| Associate professor | 4 (22) |
| Full professor | 1 (6) |
Hospitalists: Impact and Definition
Seventeen of 18 (94%) faculty felt that hospitalists reduce cost, increase patient satisfaction, and increase quality of care. Three of 18 (17%) felt that hospitalism adversely affects the primary care physicianpatient relationship. All felt that hospitalists can contribute to the academic mission of an AHSC.
Most (17/18) felt that the Society of Hospital Medicine definition of a hospitalist is useful but 13 of 18 (72%) felt that it was important to develop an international consensus‐based definition.
Clinical Roles
All faculty attended on the PMIU. Other clinical activities included: 13 of 18 (72%) inpatient general medical consultation, 9 of 18 (50%) interventional radiology clinical team, 8 of 18 (44%) attended in the pediatric consultation clinic.
The median number of weeks attending on the PMIU was 16 (range, 440 weeks).
Nine of 18 (50%) provided leadership in clinical programs or the care of specific populations of children. This included leadership in healthcare systems: director of PMIU, director of inpatient general medical consultation, interventional radiology comanagement team; and leadership in patient populations: children with chronic complex conditions, cyclic vomiting, cancer/genetic syndromes, obesity, child abuse and neglect, failure to thrive, and vascular tumors.
Nonclinical and Academic Roles
Sixteen (89%) considered their job to include activities in addition to their role of providing patient care (Table 2). This included primary activity in clinical research, education, quality improvement and health policy.
| Area of Activity | n = 18 [number (%)] |
|---|---|
| |
| QI | |
| Participates in QI activity | 12 (67) |
| Participants who lead in QI activity | 4 (33) |
| Education | |
| Teaching | |
| Medical students | |
| Inpatient unit | 18 (100) |
| Small group sessions | 10 (56) |
| Lectures | 15 (83) |
| Residents | |
| Inpatient unit | 17 (94) |
| Lectures | 12 (67) |
| EBM critical appraisal course | 5 (28) |
| General pediatric fellows | 7 (39) |
| CME | 11 (61) |
| Curriculum or program development | 9 (50) |
| Research | |
| Holds appointment with research institute | 12 (66) |
| Peer review publication in past 12 months | 13 (72) |
| Currently holds research grant | 12 (66) |
| Presented abstract at 2007 PAS meeting | 10 (56) |
Hospitalist Medicine as a Career: Perceptions
Fifteen (83%) felt that it was important to establish an annual minimum time allocation to practicing hospital medicine. A median of 11 weeks per year (range, 816 weeks) was felt to be the minimum time that should be allocated to practicing hospital medicine. The major themes related to the need to establish a minimum time for clinical practice in hospital medicine were as follows: maintenance of skills, knowledge, and competency; ensuring quality of care; and efficiency of care. One respondent explained that the main reason to have a minimum time for clinical practice was to keep up clinical skills of acute patients, stay aware of and keep up to date of available facilities in the hospital.
Thirteen (72%) felt that it was important to establish an annual maximum time allocation to practicing hospital medicine. A median of 32 weeks per year (range, 2036 weeks) was felt to be the maximum time that should be allocated to practicing hospital medicine. The major themes related to the need to establish a maximum time for clinical practice in hospital medicine were: burnout, balance in career, and desire to develop academic career and to complete nonclinical activities. As this respondent described: I think hospital medicine can be very emotionally difficult and is sustainable if provided time off to pursue other goals such as research, education, creative professional service.
Thirteen (72%) intended to continue their career as a hospitalist and 5 (28%) were unsure. Eleven (61%) felt that a job as a hospitalist was a viable long term career, 6 (33%) were unsure, and 1 (5%) felt that it was not viable. When asked what the barriers to establishing hospital medicine as a long‐term career, the major themes that emerged were as follows: burnout, establishing a nonclinical or academic niche, and the system for career advancement in an academic center. This respondent explained: As long as you have another niche (education for me) that helps provide respite, as well as [you need an] opportunity to have protected time for academic endeavours that are necessary to survive in an academic health science centre.
Another respondent described burnout as a barrier: long hours, emotionally draining with very complex patients, feeling stretchedclinical care and teaching and research and admin.
Perceived advantages to a career as a hospitalist included: working in a team, generalist approach to care, stability relative to community practice, intellectually stimulating and rewarding work, growing area and opportunities for scholarship. One respondent outlined the perceived advantages to a generalist approach: diversity of work, become a generalist specialist, teamwork, develop broad perspective on health.
Others described the perceived advantages over community practice: exciting, interesting, job stability, salary and no worries about administration of the business of community office, stable income, holiday leave.
Perceived disadvantages to a career as a hospitalist included: burnout, recognition and respect, and lack of long‐term relationships with patients. One respondent explained: as an emerging field, we have a lot of growing to do. Although our work (both clinical and academic) seem fundamental to the mission of the hospital and university, we may not feel we receive the respect, support, resources, funding allocated to other areas.
Another response to disadvantages of a career as a hospitalist was, burnout, risk of being seen as a perpetual resident.
Discussion
Freed et al.3 recently conducted a survey of U.S. pediatric hospitalist program directors from a diverse range of settings (ie, teaching vs. nonteaching, free‐standing vs. hospital system, children's hospitals vs. non‐children's hospitals).3 These investigators found that the majority of programs had employed hospitalists for less than 5 years (compared with our program, 30 years); employed 1 to 5 hospitalists (compared with our program, 20); and 25% of programs indicated their hospitalists averaged greater than 5 years on the job (compared with our program, average 10 years on the job). Maniscalco et al.6 conducted a survey in 2007 of hospitalists in a similarly diverse range of settings, found that the mean number of years on the job was 6 and found similar clinical and teaching roles. They also found that the need for advanced training in administration, research, education, and quality improvement was high. Further, we were able to examine academic roles and perceptions of hospital medicine as a career in an AHSC at an individual level. This survey, however, was limited by sampling from a single institution.
Almost all faculty identified an area of focus in addition to clinical care. Educational activities occurred at all levels: undergraduate, residency, fellowship, and continuing medical education. Faculty were engaged in research activities. Hospitalists provide care on all inpatient units as a consultant specialist in general medical care. For example, we have designed a collaborative model of care with the interventional radiology team to comanage children who require image‐guided interventions, such as gastrostomy, chest tube, and central venous line insertions.7 One further area that deserves mention is the leadership of hospitalists in outpatient care of children, especially hospital intense populations, in collaboration with their primary care provider. These groups of children are often medically and socially complex, require repeated and intense hospital resources (including diagnostic testing, subspecialty consultation, and hospitalization), and require generalist care to manage them from a family centered perspective.
A significant proportion of the faculty in this survey acquired advanced academic training. The formal training of hospitalist physicians is in its infancy. A recent work documenting the domains of training for academic general pediatric fellowship in leadership, education, and research seems to be most appropriate for the nonclinical foundation for pediatric academic hospitalists.8
Few studies have examined academic hospitalists' perceptions on the minimum and maximum time per year suitable for clinical service. This undoubtedly will vary depending on the institution, program and financial structure, patient load and complexity, call requirements, academic commitments, and stage of development. Faculty surveyed in this study felt a range of 11 to 32 weeks of clinical inpatient attending per year was ideal. This is consistent with the expert panel recommendations of the Society of Hospital Medicine. What may be equally important to determine is the maximum number of continuous weeks attending on the PMIU.
There have been 3 full‐time faculty who have left the division (all to community hospitalbased generalist practices with academic affiliations) and 1 who has changed from a full‐time to a part‐time role in the division. Most faculty surveyed intended to continue their career as a hospitalist. They identified several positive and satisfying aspects to the career, including relationships with peers, stable salary, numerous opportunities for scholarly work in a young field, and generalist care. Hoff et al.9 described a national US survey of hospitalists in all adult medicine settings that examined personal characteristics, and work‐related attitudes. Similarly, they found that hospital medicine was a source of positive social and professional work experiences related to interactions with peers, patients and families, and coworkers. In the current study, perceived disadvantages to a hospitalist career were burnout, lack of recognition and respect, and lack of long‐term relationships with patients. Hoff et al.9 noted that 37% were burnt out or at risk of burnout, which is less than in the fields of critical care medicine and emergency medicine.
The identified barriers to establishing a career were related to development of an academic focus, balance between clinical and nonclinical time, and the system for career advancement. Few other studies have examined these career issues for hospitalists in the academic setting. Several authors have discussed career issues for clinician‐educators in the US,10, 11 including metrics for promotion and recognition by institutions. Alternate methods have been proposed for promotion, aside from research and education, such as creative professional activity or clinical excellence.12 The developing field of hospital medicine faces similar challenges as individual hospitalists and the specialty itself works to align with the academic mission.1315
The division and hospitalist program have evolved over more than 2 decades to fulfill strategic goals and respond to changing external factors (Table 3). Contextual factors that have supported this evolution and that may be unique to our academic environment merit mention. First, the departments' physicians work in a within a single‐payer universal healthcare system that in some ways is similar to a single‐payer health maintenance organization. The ultimate governance is provided by the provincial Ministry of Health, which is funded through taxation. Second, through an alternative funding plan (AFP) with the provincial government, block funding is providing in lieu of fee for service clinical care that funds physician salaries for clinical work, research, education, and administrative activities.16 Third, the department has a career development compensation program (CDCP) that has an explicit job activity profile which is aligned with the role of hospitaliststhe clinician‐specialist profilewho have a predominate commitment to provide, advance, and promote excellence in clinical care with contributions to education and/or research.16 The compensation and evaluation process for hospitalists is the same as other members in the department. While further refinement of this system is ongoing, this program has demonstrated a support for all roles (ie, clinical, education, and research).17
| Date | Area | Pressure Point | Change |
|---|---|---|---|
| 1981 | Clinical education research | Department priority for academic generalism | Creation of Division of General Pediatrics (now Pediatric Medicine); full‐time hospital‐based pediatricians attending on inpatient unit |
| 1991 | Education | Division priority to foster academic generalism and train future generation of academic generalists | Creation of academic general pediatrics fellowship program |
| 1992 | Research | Division priority to foster clinical, outcomes‐based research | Creation of formal divisional research infrastructure with foundation support for an epidemiologic, outcomes‐based research platform; pediatric outcomes research team (PORT) |
| 1995 | Clinical care | Province‐wide reduction in resident duty hours; division priority to raise the bar for clinical and teaching excellence in hospitalist‐and community‐based pediatrics; need for a financially viable and cost‐effective model for staffing attending pediatricians on inpatient unit | Reorganization of inpatient unit; higher proportion of attending pediatricians who are full‐time, hospital‐based; creation of a staff‐only hospitalist unit19; creation of a section of community pediatrics (2001) |
| 1996 | Clinical care education faculty development | Need to limit attending hospitalists after‐hours clinical care to ensure balance and academic productivity; need for a system of after‐hours physician coverage for inpatient care that is sustainable and financially viable; need for funding sources for academic general pediatrics fellowship | Creation of a clinical departmental fellowship program to fund after hours clinical coverage with qualified pediatricians seeking additional fellowship training20 |
| 1998 | Faculty career development | Need to value and reward all academic contributions, such as the hospitalist role, through an explicit job activity profile within the department16 | Implementation of a career development and compensation program with the clinician specialist role whose primary contributions are to excellence in clinical care |
| 1999 | Clinical care | Expansion of hospital interventional radiology program and need for high quality collaborative care | Comanagement model with hospitalist‐radiologist team7 |
| 20068 | Clinical care research | Division and institution priority to provide high‐quality care for children with complex care health issues and foster a research program21 | Creation of formalized hospitalist complex care program with inpatient and outpatient care22; research support for complex care |
| 2007 | Clinical care | Refinement of inpatient unit organization to improve efficiency of care; increase number of trainees23 | Reorganization of inpatient units to geographic allocation of patients by attending physician; addition of trainees to staff‐only hospitalist unit |
Furthermore, several divisional factors have contributed to the viability of hospitalism within our generalist division. First, hospitalists were integrated into, rather than segregated from the division. Second, hospitalists have the opportunity to engage in diverse clinical activities. Wachter and Goldman18 advocate for hospitalist participation in outpatient care to provide variety and to cement their relationship with their generalist division. Third, a fellowship training program was established in 1992 that integrated principles of academic general pediatrics and hospitalism. Fourth, career development in education, research, and, more recently, quality improvement is fostered.
In summary, the faculty of an established pediatric hospitalist program have diverse and unique clinical, leadership, and scholarly contributions to the academic mission of the department. In order to further promote integration, several issues should be addressed, including optimal training, time allocated to nonclinical activities, systems for career development and promotion of hospitalist faculty, and mentorship. Finally, it is important that leaders in pediatric hospital medicine and general pediatrics engage the larger academic community to strengthen the role and contributions of hospitalists in AHSCs.
Acknowledgements
The authors thank the faculty of the Division of Pediatric Medicine, Hospital for Sick Children for participating in the survey and past and present faculty for their contributions to the development of the division.
The successful integration of hospitalists in academic health science centers (AHSCs) has been identified as one of the most challenging areas for the hospitalist movement.1, 2 This has been based on a concern that many hospitalists lack academic and research skills, lack mentorship, and may have little time to develop academic careers because of the significant time they spend in clinical care.
A recent survey highlighted that the pediatric hospitalist workforce is in its infancy and additional perspectives, such as from hospitalists themselves, are essential for a more complete picture of the current state of pediatric hospital medicine.3 Hospitalists have had a long history in Canada.4 The Hospital for Sick Children, Toronto, Canada, has had a Division of Pediatric Medicine since 1981, with hospitalists, as we now know them, from inception. This provided a rich resource to explore pediatric hospital medicine in the academic context and from hospitalists themselves. The objective of this survey was to explore the characteristics, practice, and perceptions of pediatric hospital medicine in an AHSC. Locally, we hoped the results would inform the division on program development, training, and faculty career development. Externally, the findings could contribute to a body of knowledge on the evolving role of pediatric hospitalists and provide insight into opportunities for better integration into AHSCs.
Methods
Study Design
This was a cross‐sectional survey of pediatricians who attend on the pediatric medicine inpatient unit at the Hospital for Sick Children, Toronto. The study protocol was approved by Quality and Risk Management at The Hospital for Sick Children.
Setting
The Hospital for Sick Children is a tertiary care children's hospital affiliated with the University of Toronto, Toronto, Canada. The total hospital bed capacity including intensive care unit beds and subspecialty beds is 320. It is the only free‐standing pediatric hospital for the greater Toronto area with a catchment population of 5 million people.
A formal division for general pediatrics, Pediatric Medicine, has been in existence since 1981 with hospital‐based pediatricians (who now are known as hospitalists) who attend on the inpatient unit. The pediatric medicine inpatient unit (PMIU) has a 60‐bed capacity on 3 units. At all times, the PMIU is staffed by 4 to 6 inpatient attendings from a total of 20 full‐time and major part‐time pediatricians. According to Wachter's staging of hospital care, the PMIU is at stage IV of IV in development (mandatory care by hospitalist).5 All attending pediatricians are members of the Division of Pediatric Medicine. Pediatric subspecialists do not attend on the PMIU. Physicians attend for a minimum of 4 weeks at a time. General pediatric house staff including fellows, residents (postgraduate year [PGY] 1, PGY3) and medical students are supervised by the attending pediatricians. Other inpatient clinical services provided include an inpatient general medical consultation team, a complex care team for inpatients with chronic complex conditions, and a consultative and collaborative role with the team of interventional radiologists. An outpatient pediatric consultation program exists for hospital follow‐up, general pediatric consultation, and specialized generalist care for specific populations of children.
In addition to the clinical program, research and education programs exist within the division. The division has had an academic general pediatric fellowship program since 1992, with the majority of most recent graduates obtaining academic hospitalist faculty positions. A formal research group and infrastructure for clinical, outcomes‐based research within the division, known as the Pediatric Outcomes Research Team (PORT), exists.
Study Population
All pediatricians who attend on the PMIU and in the Division of Pediatric Medicine, Hospital for Sick Children in 2007 were eligible for recruitment including the 2 eligible study authors.
Survey Instrument
We constructed a 43‐item structured questionnaire that asked about and explored training and employment characteristics; clinical roles and nonclinical roles (teaching, quality, research, leadership); and perceptions of hospital medicine and a career in this field. Several items were modeled after questions administered to program directors in a survey reported by Freed et al.3 The questionnaire was designed to be completed by the respondent on paper.
Questionnaire Administration
From September to December 2007, eligible pediatricians were sent a letter explaining the study and the questionnaire through interhospital mail by the research coordinator. Questionnaires were deidentified and assigned a unique identification number. Only the research coordinator had access to the list that linked the unique identification numbers with names. Confidentiality of responses and anonymity of responses was explicitly stated in the letter explaining the study to pediatricians. Nonrespondents were contacted by the research assistant at 3‐week intervals with personal reminders.
Data Analysis
During the analysis phase, responses remained deidentified. Descriptive statistics using means, medians, and proportions were calculated for survey items. For open‐ended questions, answers from respondents were summarized into key concepts or themes by the 3 study authors. Excerpts from responses were abstracted to highlight themes.
Results
Eighteen of 20 (90%) faculty responded to the questionnaire. The results are presented by the domains of the questionnaire, as follows.
Training and Employment Characteristics
Due to the requirements of the Royal College of Physicians and Surgeons of Canada, all faculty had a minimum of 4 years of pediatric specialty training leading to certification (Table 1). Seven of 18 (39%) had fellowship and graduate training. Nine of 18 (50%) had been on faculty for greater than 5 years.
| Training after medical schools, median years (range) | 4.8 (46) |
| Fellowship trained, number (%) | 7 (39) |
| Fellowship duration, median years (range) | 2.7 (15) |
| Graduate studies, number (%) | 7 (39) |
| Clinical epidemiology | 5 |
| Education | 2 |
| Duration since first academic appointment | |
| Median, years (range) | 5.5 (030) |
| Mean, years (standard deviation) | 9.9 (10.4) |
| >5 years, number (%) | 9 (50) |
| Academic rank, number (%) | |
| Lecturer | 2 (11) |
| Assistant professor | 10 (56) |
| Associate professor | 4 (22) |
| Full professor | 1 (6) |
Hospitalists: Impact and Definition
Seventeen of 18 (94%) faculty felt that hospitalists reduce cost, increase patient satisfaction, and increase quality of care. Three of 18 (17%) felt that hospitalism adversely affects the primary care physicianpatient relationship. All felt that hospitalists can contribute to the academic mission of an AHSC.
Most (17/18) felt that the Society of Hospital Medicine definition of a hospitalist is useful but 13 of 18 (72%) felt that it was important to develop an international consensus‐based definition.
Clinical Roles
All faculty attended on the PMIU. Other clinical activities included: 13 of 18 (72%) inpatient general medical consultation, 9 of 18 (50%) interventional radiology clinical team, 8 of 18 (44%) attended in the pediatric consultation clinic.
The median number of weeks attending on the PMIU was 16 (range, 440 weeks).
Nine of 18 (50%) provided leadership in clinical programs or the care of specific populations of children. This included leadership in healthcare systems: director of PMIU, director of inpatient general medical consultation, interventional radiology comanagement team; and leadership in patient populations: children with chronic complex conditions, cyclic vomiting, cancer/genetic syndromes, obesity, child abuse and neglect, failure to thrive, and vascular tumors.
Nonclinical and Academic Roles
Sixteen (89%) considered their job to include activities in addition to their role of providing patient care (Table 2). This included primary activity in clinical research, education, quality improvement and health policy.
| Area of Activity | n = 18 [number (%)] |
|---|---|
| |
| QI | |
| Participates in QI activity | 12 (67) |
| Participants who lead in QI activity | 4 (33) |
| Education | |
| Teaching | |
| Medical students | |
| Inpatient unit | 18 (100) |
| Small group sessions | 10 (56) |
| Lectures | 15 (83) |
| Residents | |
| Inpatient unit | 17 (94) |
| Lectures | 12 (67) |
| EBM critical appraisal course | 5 (28) |
| General pediatric fellows | 7 (39) |
| CME | 11 (61) |
| Curriculum or program development | 9 (50) |
| Research | |
| Holds appointment with research institute | 12 (66) |
| Peer review publication in past 12 months | 13 (72) |
| Currently holds research grant | 12 (66) |
| Presented abstract at 2007 PAS meeting | 10 (56) |
Hospitalist Medicine as a Career: Perceptions
Fifteen (83%) felt that it was important to establish an annual minimum time allocation to practicing hospital medicine. A median of 11 weeks per year (range, 816 weeks) was felt to be the minimum time that should be allocated to practicing hospital medicine. The major themes related to the need to establish a minimum time for clinical practice in hospital medicine were as follows: maintenance of skills, knowledge, and competency; ensuring quality of care; and efficiency of care. One respondent explained that the main reason to have a minimum time for clinical practice was to keep up clinical skills of acute patients, stay aware of and keep up to date of available facilities in the hospital.
Thirteen (72%) felt that it was important to establish an annual maximum time allocation to practicing hospital medicine. A median of 32 weeks per year (range, 2036 weeks) was felt to be the maximum time that should be allocated to practicing hospital medicine. The major themes related to the need to establish a maximum time for clinical practice in hospital medicine were: burnout, balance in career, and desire to develop academic career and to complete nonclinical activities. As this respondent described: I think hospital medicine can be very emotionally difficult and is sustainable if provided time off to pursue other goals such as research, education, creative professional service.
Thirteen (72%) intended to continue their career as a hospitalist and 5 (28%) were unsure. Eleven (61%) felt that a job as a hospitalist was a viable long term career, 6 (33%) were unsure, and 1 (5%) felt that it was not viable. When asked what the barriers to establishing hospital medicine as a long‐term career, the major themes that emerged were as follows: burnout, establishing a nonclinical or academic niche, and the system for career advancement in an academic center. This respondent explained: As long as you have another niche (education for me) that helps provide respite, as well as [you need an] opportunity to have protected time for academic endeavours that are necessary to survive in an academic health science centre.
Another respondent described burnout as a barrier: long hours, emotionally draining with very complex patients, feeling stretchedclinical care and teaching and research and admin.
Perceived advantages to a career as a hospitalist included: working in a team, generalist approach to care, stability relative to community practice, intellectually stimulating and rewarding work, growing area and opportunities for scholarship. One respondent outlined the perceived advantages to a generalist approach: diversity of work, become a generalist specialist, teamwork, develop broad perspective on health.
Others described the perceived advantages over community practice: exciting, interesting, job stability, salary and no worries about administration of the business of community office, stable income, holiday leave.
Perceived disadvantages to a career as a hospitalist included: burnout, recognition and respect, and lack of long‐term relationships with patients. One respondent explained: as an emerging field, we have a lot of growing to do. Although our work (both clinical and academic) seem fundamental to the mission of the hospital and university, we may not feel we receive the respect, support, resources, funding allocated to other areas.
Another response to disadvantages of a career as a hospitalist was, burnout, risk of being seen as a perpetual resident.
Discussion
Freed et al.3 recently conducted a survey of U.S. pediatric hospitalist program directors from a diverse range of settings (ie, teaching vs. nonteaching, free‐standing vs. hospital system, children's hospitals vs. non‐children's hospitals).3 These investigators found that the majority of programs had employed hospitalists for less than 5 years (compared with our program, 30 years); employed 1 to 5 hospitalists (compared with our program, 20); and 25% of programs indicated their hospitalists averaged greater than 5 years on the job (compared with our program, average 10 years on the job). Maniscalco et al.6 conducted a survey in 2007 of hospitalists in a similarly diverse range of settings, found that the mean number of years on the job was 6 and found similar clinical and teaching roles. They also found that the need for advanced training in administration, research, education, and quality improvement was high. Further, we were able to examine academic roles and perceptions of hospital medicine as a career in an AHSC at an individual level. This survey, however, was limited by sampling from a single institution.
Almost all faculty identified an area of focus in addition to clinical care. Educational activities occurred at all levels: undergraduate, residency, fellowship, and continuing medical education. Faculty were engaged in research activities. Hospitalists provide care on all inpatient units as a consultant specialist in general medical care. For example, we have designed a collaborative model of care with the interventional radiology team to comanage children who require image‐guided interventions, such as gastrostomy, chest tube, and central venous line insertions.7 One further area that deserves mention is the leadership of hospitalists in outpatient care of children, especially hospital intense populations, in collaboration with their primary care provider. These groups of children are often medically and socially complex, require repeated and intense hospital resources (including diagnostic testing, subspecialty consultation, and hospitalization), and require generalist care to manage them from a family centered perspective.
A significant proportion of the faculty in this survey acquired advanced academic training. The formal training of hospitalist physicians is in its infancy. A recent work documenting the domains of training for academic general pediatric fellowship in leadership, education, and research seems to be most appropriate for the nonclinical foundation for pediatric academic hospitalists.8
Few studies have examined academic hospitalists' perceptions on the minimum and maximum time per year suitable for clinical service. This undoubtedly will vary depending on the institution, program and financial structure, patient load and complexity, call requirements, academic commitments, and stage of development. Faculty surveyed in this study felt a range of 11 to 32 weeks of clinical inpatient attending per year was ideal. This is consistent with the expert panel recommendations of the Society of Hospital Medicine. What may be equally important to determine is the maximum number of continuous weeks attending on the PMIU.
There have been 3 full‐time faculty who have left the division (all to community hospitalbased generalist practices with academic affiliations) and 1 who has changed from a full‐time to a part‐time role in the division. Most faculty surveyed intended to continue their career as a hospitalist. They identified several positive and satisfying aspects to the career, including relationships with peers, stable salary, numerous opportunities for scholarly work in a young field, and generalist care. Hoff et al.9 described a national US survey of hospitalists in all adult medicine settings that examined personal characteristics, and work‐related attitudes. Similarly, they found that hospital medicine was a source of positive social and professional work experiences related to interactions with peers, patients and families, and coworkers. In the current study, perceived disadvantages to a hospitalist career were burnout, lack of recognition and respect, and lack of long‐term relationships with patients. Hoff et al.9 noted that 37% were burnt out or at risk of burnout, which is less than in the fields of critical care medicine and emergency medicine.
The identified barriers to establishing a career were related to development of an academic focus, balance between clinical and nonclinical time, and the system for career advancement. Few other studies have examined these career issues for hospitalists in the academic setting. Several authors have discussed career issues for clinician‐educators in the US,10, 11 including metrics for promotion and recognition by institutions. Alternate methods have been proposed for promotion, aside from research and education, such as creative professional activity or clinical excellence.12 The developing field of hospital medicine faces similar challenges as individual hospitalists and the specialty itself works to align with the academic mission.1315
The division and hospitalist program have evolved over more than 2 decades to fulfill strategic goals and respond to changing external factors (Table 3). Contextual factors that have supported this evolution and that may be unique to our academic environment merit mention. First, the departments' physicians work in a within a single‐payer universal healthcare system that in some ways is similar to a single‐payer health maintenance organization. The ultimate governance is provided by the provincial Ministry of Health, which is funded through taxation. Second, through an alternative funding plan (AFP) with the provincial government, block funding is providing in lieu of fee for service clinical care that funds physician salaries for clinical work, research, education, and administrative activities.16 Third, the department has a career development compensation program (CDCP) that has an explicit job activity profile which is aligned with the role of hospitaliststhe clinician‐specialist profilewho have a predominate commitment to provide, advance, and promote excellence in clinical care with contributions to education and/or research.16 The compensation and evaluation process for hospitalists is the same as other members in the department. While further refinement of this system is ongoing, this program has demonstrated a support for all roles (ie, clinical, education, and research).17
| Date | Area | Pressure Point | Change |
|---|---|---|---|
| 1981 | Clinical education research | Department priority for academic generalism | Creation of Division of General Pediatrics (now Pediatric Medicine); full‐time hospital‐based pediatricians attending on inpatient unit |
| 1991 | Education | Division priority to foster academic generalism and train future generation of academic generalists | Creation of academic general pediatrics fellowship program |
| 1992 | Research | Division priority to foster clinical, outcomes‐based research | Creation of formal divisional research infrastructure with foundation support for an epidemiologic, outcomes‐based research platform; pediatric outcomes research team (PORT) |
| 1995 | Clinical care | Province‐wide reduction in resident duty hours; division priority to raise the bar for clinical and teaching excellence in hospitalist‐and community‐based pediatrics; need for a financially viable and cost‐effective model for staffing attending pediatricians on inpatient unit | Reorganization of inpatient unit; higher proportion of attending pediatricians who are full‐time, hospital‐based; creation of a staff‐only hospitalist unit19; creation of a section of community pediatrics (2001) |
| 1996 | Clinical care education faculty development | Need to limit attending hospitalists after‐hours clinical care to ensure balance and academic productivity; need for a system of after‐hours physician coverage for inpatient care that is sustainable and financially viable; need for funding sources for academic general pediatrics fellowship | Creation of a clinical departmental fellowship program to fund after hours clinical coverage with qualified pediatricians seeking additional fellowship training20 |
| 1998 | Faculty career development | Need to value and reward all academic contributions, such as the hospitalist role, through an explicit job activity profile within the department16 | Implementation of a career development and compensation program with the clinician specialist role whose primary contributions are to excellence in clinical care |
| 1999 | Clinical care | Expansion of hospital interventional radiology program and need for high quality collaborative care | Comanagement model with hospitalist‐radiologist team7 |
| 20068 | Clinical care research | Division and institution priority to provide high‐quality care for children with complex care health issues and foster a research program21 | Creation of formalized hospitalist complex care program with inpatient and outpatient care22; research support for complex care |
| 2007 | Clinical care | Refinement of inpatient unit organization to improve efficiency of care; increase number of trainees23 | Reorganization of inpatient units to geographic allocation of patients by attending physician; addition of trainees to staff‐only hospitalist unit |
Furthermore, several divisional factors have contributed to the viability of hospitalism within our generalist division. First, hospitalists were integrated into, rather than segregated from the division. Second, hospitalists have the opportunity to engage in diverse clinical activities. Wachter and Goldman18 advocate for hospitalist participation in outpatient care to provide variety and to cement their relationship with their generalist division. Third, a fellowship training program was established in 1992 that integrated principles of academic general pediatrics and hospitalism. Fourth, career development in education, research, and, more recently, quality improvement is fostered.
In summary, the faculty of an established pediatric hospitalist program have diverse and unique clinical, leadership, and scholarly contributions to the academic mission of the department. In order to further promote integration, several issues should be addressed, including optimal training, time allocated to nonclinical activities, systems for career development and promotion of hospitalist faculty, and mentorship. Finally, it is important that leaders in pediatric hospital medicine and general pediatrics engage the larger academic community to strengthen the role and contributions of hospitalists in AHSCs.
Acknowledgements
The authors thank the faculty of the Division of Pediatric Medicine, Hospital for Sick Children for participating in the survey and past and present faculty for their contributions to the development of the division.
- ,.Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- .The hospitalist movement—time to move on.N Engl J Med.2007;25:2627–2629.
- ,,.Characteristics of the pediatric hospitalist workforce: its roles and work environment.Pediatrics.2007;120:33–39.
- .A Canadian perspective on the American hospitalist movement.Arch Int Med.1999;159:1666–1668.
- .An introduction to the hospitalist model.Ann Intern Med.1999;130:338–342.
- ,,,,.Current roles and training needs of pediatric hospitalists: a study from the Pediatric Research in Inpatient Settings (PRIS) network.Paper presented at: Pediatric Academic Society Annual Meeting; May2008;Honolulu, HI. E‐PAS2008:6725.4.
- ,.The pediatric hospitalist and interventional radiologist: a model for clinical care in pediatric interventional radiology.J Vasc Interv Radiol.2006;17:1733–1738.
- ,,, et al.Academic general pediatric fellowships: curriculum design and educational goals and objectives.Ambul Pediatr.2007;7:328–339.
- ,,,,.Characteristics and work experiences of hospitalists in the United States.Arch Intern Med.2001;161:851–858.
- ,.Mission critical—integrating clinician‐educators into academic medical centers.N Engl J Med.1999;341:840–843.
- ,,,.Separate and equitable promotion tracks for clinician‐educators.JAMA.2005;294:1101–1103.
- ,,.Creative professional activity: an additional platform for promotion of faculty.Acad Med.2006;81:568–570.
- .Helping hospitalists achieve academic stature.J Hosp Med.2008;3:285–287.
- ,,,.An innovative approach to supporting hospitalist physicians toward academic success.J Hosp Med.2008;3:314–318.
- ,,,.The University of Michigan Specialist‐Hospitalist Allied Research Program: jumpstarting hospitalist medicine research.J Hosp Med.2008;3:308–313.
- ,,,,,.Evaluation of a peer‐reviewed career development and compensation program for physicians at an academic health science centre.Pediatrics.2003;111:e26–e31.
- ,,,,.Performance of a career development and compensation program at an academic health science center.Pediatrics.2007;119:e791–e797.
- ,.Implications of the hospitalist movement for academic departments of medicine: lessons from the UCSF experience.Am J Med.1999;106:127–133.
- ,,,.Evaluation of a staff‐only hospitalist system in a tertiary care, academic children's hospital.Pediatrics.2004;114:1545–1549.
- ,.Providing after‐hours on‐call clinical coverage in academic health sciences centres: the Hospital for Sick Children experience.CMAJ.2000;163:298–299.
- ,,,,.A home for medically complex children: the role of hospital programs.J Healthc Qual.2008;30:7–15.
- ,,.Comprehensive care for medically complex children: the pediatric nurse practitioner‐hospitalist model of collaborative care.Hosp Pediatr.2009;1:20–23.
- ,,.Pediatric hospitalist medicine: an overview and a perspective from Toronto, Canada.Clin Pediatr (Phila).2008;47:546–548.
- ,.Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- .The hospitalist movement—time to move on.N Engl J Med.2007;25:2627–2629.
- ,,.Characteristics of the pediatric hospitalist workforce: its roles and work environment.Pediatrics.2007;120:33–39.
- .A Canadian perspective on the American hospitalist movement.Arch Int Med.1999;159:1666–1668.
- .An introduction to the hospitalist model.Ann Intern Med.1999;130:338–342.
- ,,,,.Current roles and training needs of pediatric hospitalists: a study from the Pediatric Research in Inpatient Settings (PRIS) network.Paper presented at: Pediatric Academic Society Annual Meeting; May2008;Honolulu, HI. E‐PAS2008:6725.4.
- ,.The pediatric hospitalist and interventional radiologist: a model for clinical care in pediatric interventional radiology.J Vasc Interv Radiol.2006;17:1733–1738.
- ,,, et al.Academic general pediatric fellowships: curriculum design and educational goals and objectives.Ambul Pediatr.2007;7:328–339.
- ,,,,.Characteristics and work experiences of hospitalists in the United States.Arch Intern Med.2001;161:851–858.
- ,.Mission critical—integrating clinician‐educators into academic medical centers.N Engl J Med.1999;341:840–843.
- ,,,.Separate and equitable promotion tracks for clinician‐educators.JAMA.2005;294:1101–1103.
- ,,.Creative professional activity: an additional platform for promotion of faculty.Acad Med.2006;81:568–570.
- .Helping hospitalists achieve academic stature.J Hosp Med.2008;3:285–287.
- ,,,.An innovative approach to supporting hospitalist physicians toward academic success.J Hosp Med.2008;3:314–318.
- ,,,.The University of Michigan Specialist‐Hospitalist Allied Research Program: jumpstarting hospitalist medicine research.J Hosp Med.2008;3:308–313.
- ,,,,,.Evaluation of a peer‐reviewed career development and compensation program for physicians at an academic health science centre.Pediatrics.2003;111:e26–e31.
- ,,,,.Performance of a career development and compensation program at an academic health science center.Pediatrics.2007;119:e791–e797.
- ,.Implications of the hospitalist movement for academic departments of medicine: lessons from the UCSF experience.Am J Med.1999;106:127–133.
- ,,,.Evaluation of a staff‐only hospitalist system in a tertiary care, academic children's hospital.Pediatrics.2004;114:1545–1549.
- ,.Providing after‐hours on‐call clinical coverage in academic health sciences centres: the Hospital for Sick Children experience.CMAJ.2000;163:298–299.
- ,,,,.A home for medically complex children: the role of hospital programs.J Healthc Qual.2008;30:7–15.
- ,,.Comprehensive care for medically complex children: the pediatric nurse practitioner‐hospitalist model of collaborative care.Hosp Pediatr.2009;1:20–23.
- ,,.Pediatric hospitalist medicine: an overview and a perspective from Toronto, Canada.Clin Pediatr (Phila).2008;47:546–548.
Copyright © 2010 Society of Hospital Medicine
Treatment of Lactic Acidosis
Lactic acidosis (LA) is common in hospitalized patients and is associated with a high mortality.1, 2 Commonly, it is defined as a lactic acid concentration greater than 5 mmol/L with a pH less than 7.35.3 There are no evidence‐based guidelines for the treatment of LA despite progress in our understanding of its pathophysiology.36 This is not surprising, given the uncertainty regarding the impact of LA itself on clinical outcomes. In this regard, it is interesting to note that, despite its well‐recognized role as a marker of tissue hypoxia, lactate accumulation appears to have beneficial effects and may function as an adaptive mechanism. This raises the possibility that therapy directed at altering this adaptation may be detrimental. Pursuing correction of the pH in LA has been shown to have untoward physiologic effects. These and other ambiguities in the pathophysiology and treatment of LA are the focus of this review.
Lactate Metabolism
The body produces approximately 1400 mmol of lactate daily.7 Lactate is derived from the metabolism of pyruvate through an anaerobic reaction that occurs in all tissues (Figure 1). The liver is the primary site of lactate clearance and can metabolize up to 100 mmol per hour under normal conditions.8 There, lactate is converted to glucose to serve as an energy source during periods of hypoxia (Figure 2).9
Approximately 20% to 30% of the daily lactate load is metabolized by the kidneys.10, 11 Renal clearance is increased in acidosis12 and is maintained even in the presence of low renal perfusion.10, 12, 13 Renal lactate clearance is primarily through metabolism and not excretion.10, 14
LA Subtypes
Generally, lactic acid accumulation results from excess lactic acid production and not from reduced clearance.15 In cases of fulminant liver failure, it is due to a combination of decreased clearance and tissue hypoxia.16 In the setting of tissue hypoxia, an impairment of mitochondrial oxidative capacity results in the accumulation of pyruvate and generation of lactate. Lactic acid accumulation through this mechanism has historically been described as Type A LA.7 Hence, in critically ill patients lactate has traditionally been viewed as a marker of tissue hypoxia.15, 1721 Hyperlactatemia without tissue hypoxia has been referred to as type B LA. This is seen in a variety of circumstances. In sepsis, for example, several studies have shown lactic acid accumulation, despite adequate oxygen delivery.2224
Hyperlactatemia may also occur in cases of pure mitochondrial dysfunction, which can be induced by commonly prescribed medications such as the biguanides, nucleoside analog reverse‐transcriptase inhibitors (NRTIs), and linezolid.2527 Alternatively, lactate generation from metabolism of agents such as propylene glycol is possible. Finally, excessive lactate generation may occur following stress due to altered carbohydrate metabolism, or with respiratory alkalosis.2831
Lactate: A Metabolic Adaptation
Lactate was traditionally considered only as a marker of tissue hypoxia and anaerobic metabolism.17 This is certainly the case in situations of poor perfusion such as cardiogenic,15, 18 vasopressor‐resistant,19 or hypovolemic shock.20, 21
Alternative explanations for lactic acid accumulation, without tissue hypoperfusion, include catecholamine‐induced alterations in glycolysis,32, 33 mitochondrial disturbances,3436 and increased pyruvate production combined with increased glucose entry into cells.24, 37 In addition, the activity of an enzyme regulating lactate metabolism, pyruvate dehydrogenase kinase, increases in sepsis.38 This enzyme inactivates the pyruvate dehydrogenase (PDH) complex, which metabolizes pyruvate. Pyruvate and lactate may accumulate as a result. These changes partly explain the generation of LA in sepsis, independent of any effect of diminished tissue perfusion.
Recognizing the body's tendency toward homeostasis, it is appealing to speculate that lactate accumulation is adaptive.9 A number of findings support this. For example, lactate may act to shuttle energy between organs, or between cell types in the same organ. The astrocyteneuron lactate shuttle and the spermatogenic lactate shuttle are 2 examples of lactate's valuable effects on cellular metabolism.39 In the astrocyteneuron lactate shuttle, astrocytes support the increased metabolic demands of neurons through lactic acid production.40 Specifically, the neurotransmitter glutamate is released by the neurons and taken up by the astrocytes. Astrocytes produce lactate, which then moves back to the neuron to be used as an energy source. Glutamine, also released by the astrocytes, leads to the regeneration of glutamate and the potential to restart the cycle.39
Animal and human studies have suggested that, in periods of stress, lactate is the preferential energy substrate in the brain.4144 The usefulness of increased lactate production routinely seen in sepsis may thus represent multiple adaptive processes aimed primarily at improving the delivery of energy substrates. Thus, therapeutic strategies aimed specifically at lowering lactic acid levels may prove to have deleterious effects on cellular metabolism.
Impact of LA on Morbidity and Mortality
The poor prognosis in patients with LA is well recognized.2, 4548 For example, in a study of 126 patients with various causes of LA, the median survival was 38.5 hours and 30‐day survival was 17%.2
Studies have revealed that LA with low pH is associated with adverse effects on the cardiovascular system, particularly a decrease in cardiac contractility.49, 50 This effect is particularly prominent with a pH below 7.20. In contrast, acidosis in animal models has been shown to limit myocardial infarct size after reperfusion.51, 52 Variable effects of LA on cell death have been found. A worsening of apoptosis in myocytes has been noted;53 alternatively, protection from hypoxic injury in hepatocytes and myocardium has been observed.52, 54 Thus, although LA is associated with poor outcomes in human studies,2, 4547 it is still unclear to what extent lactic acid accumulation is a marker of severe illness, an independent effector of pathology, or a mechanism with the potential to serve a protective role.
Available data indicate that lactate itself is not harmful. Studies on infusion of lactate solutions to postoperative patients was shown to be safe.55 Also, the fact that lactate generation in states of respiratory alkalosis, stress, or altered carbohydrate metabolism without sepsis is not associated with worse outcomes supports the fact that lactic acid alone may not be maladaptive.2831
Similarly, low pH is not necessarily maladaptive. In the postictal state,56 diabetic ketoacidosis,57 spontaneous respiratory acidosis,58 or permissive hypercapnia,59 low blood pH is not deleterious.
In summary, LA is associated with poor outcomes, and indirect evidence suggests that it is the underlying causative condition rather than the low pH or the lactate that is responsible for the dire outcomes.
Treatment of LA with Sodium Bicarbonate
Since excessive lactic acid generation is accompanied by consumption of plasma bicarbonate and a fall in plasma pH, sodium bicarbonate has been long proposed as a treatment for LA. While theoretically appealing, this strategy has not been validated by studies in animals or humans. Indeed, bicarbonate administration in LA often has been shown to be detrimental.60, 61 The adverse effects of bicarbonate administration in LA, while initially paradoxical, have a number of possible explanations.
First, bicarbonate administration can induce a reduction in intracellular pH.60, 62, 63 The mechanism involves bicarbonate's effect to increase carbon dioxide (CO2) generation through mass action effect. Because the cell membrane is more permeable to CO2 than to bicarbonate, intracellular pH falls.64, 65 In sepsis, this intracellular/extracellular pH discrepancy may be more pronounced due to alterations in blood flow.66 Other reports on outcomes of intracellular pH with bicarbonate therapy show variable effects.6772
Second, to the extent that bicarbonate administration raises extracellular pH, it is associated with a reduction in ionized calcium concentration, since the binding of calcium to albumin is pH dependent.73 A sodium bicarbonate load administered to patients with LA was associated with a significant fall in ionized calcium concentration, whereas a sodium chloride load was not.1 This can affect cardiac function, as the latter varies proportionally with calcium levels.74
Third, bicarbonate administration may reduce tissue oxygen delivery since the affinity of hemoglobin for oxygen increases as pH rises (Bohr effect).75 The administration of bicarbonate worsened systemic oxygen consumption in one study76 and decreased oxygen delivery in another.75
Fourth, bicarbonate administration may indirectly increase intracellular calcium concentration. Low intracellular pH (see above) stimulates proton efflux by way of proton transporters and exchangers, increasing intracellular sodium content.77 A high cell sodium content then may increase intracellular calcium, through the Na/Ca exchanger, impairing cellular function.7779 Compounding this, the reduced function of the Na/H ATPase as a regulator of intracellular sodium in sepsis may not be adequate to limit cell swelling.77
Against this background of mechanistic concerns with the use of bicarbonate treatment, it is not surprising that clinical outcomes have been inconsistent at best. In animal models of LA, the use of sodium bicarbonate has either negative effects on cardiac output60, 72 or no significant hemodynamic effect when compared to sodium chloride infusion.67, 80, 81 One animal study did show some benefit with sodium bicarbonate compared to saline, though all animals subsequently died.50
In humans, sodium bicarbonate was studied in 2 randomized trials of sepsis‐induced LA.1, 82 In a study by Cooper et al.,1 14 critically‐ill patients received sequential infusions of sodium bicarbonate or sodium chloride. Neither solution was superior to the other in terms of hemodynamic improvement. No benefit was noted even when analysis was limited to those with very low pH (7.2). Mathieu et al.82 randomized 10 critically‐ill patients to sequential infusion of either sodium bicarbonate or sodium chloride. Similarly, no significant difference in hemodynamic variables was noted.
When taken together, these studies evaluating sodium bicarbonate in LA fail to show convincing benefit and raise serious questions about its detrimental effects. Extracellular pH may be a misleading marker of success in the treatment of LA, given its direct influence by sodium bicarbonate administration.
Treatment of LA and Use of Other Buffers
Other buffers (Carbicarb, dichloroacetate, and tromethamine [THAM]) have been studied for treatment of LA. Human studies have not shown superiority of any of the buffers as far as improving pH,83, 84 hemodynamics, or survival.85
Treatment of LA by Renal Replacement Therapy
Renal replacement therapy (RRT; dialysis and its variants) has been studied for the treatment of severe acidosis. RRT has a number of theoretical advantages over purely medical therapies in the treatment of LA: it can deliver large quantities of base without contributing to volume overload; it can directly remove lactate from the plasma; and it can mitigate the effect of alkalinization on ionized calcium concentration by delivering calcium.
In critically ill patients with intact liver function, continuous venovenous hemofiltration (CVVH) appears to contribute very little (less then 3%) to overall lactate clearance.86 While outcome studies are limited, continuous dialysis modalities consistently show improved resolution of acidosis of various types when compared to intermittent modalities.87, 88 As described above, this is related to base administration and is not a surprising finding. There are no studies comparing RRT and medical therapy with respect to clinical outcomes in patients with LA.
Special Situations
Biguanides
Biguanide‐induced LA can be due to impairment of hepatic neoglucogenesis, in the case of metformin, or increasing hepatic oxidative phosphorylation, in the case of phenformin.89 This infrequent complication90, 91 is associated with a high mortality.92 Proposed therapy has included the use of sodium bicarbonate infusion.93 In this setting, it is unclear if the use of bicarbonate alone improves clinical outcomes.94
Renal replacement therapy in a wide variety of formats has been used to treat this condition.93, 95101 Metformin has a high clearance during dialysis due to its low molecular weight and lack of protein binding.97, 98, 102 Nonetheless, its high volume of distribution suggests a longer dialysis time would be more beneficial if the main goal is reducing metformin levels.97, 103 The limited prospective literature and lack of conclusive evidence about what levels of metformin induce LA makes generalized recommendations about duration of hemodialysis purely speculative.104
NRTIs
The use of NRTIs is associated with LA due to impairment of mitochondrial oxidative phosphorylation.105108 This uncommon complication, if not recognized early, is associated with a high mortality.101, 109 Investigations are ongoing into agents directed at improving mitochondrial function such as riboflavin, thiamine, and L‐carnitine.110112 As with biguanide‐associated LA, RRT decisions should be individualized based on metabolic circumstances.
Lorazepam
Many intravenous medications are formulated in the alcohol solvent, propylene glycol. Injectable lorazepam has the highest proportional amount of propylene glycol compared with other commonly used agents.113, 114 The kidney normally eliminates 12% to 50% of administered propylene glycol via proximal tubule secretion.115 The remainder is metabolized by the liver to form pyruvate and lactate.114, 116, 117
When propylene glycol accumulates, as in cases of reduced renal function, it results in hyperosmolarity, LA, and can even induce additional kidney injury (probably through proximal tubular cell necrosis).118
LA due to propylene glycol has been reported by many authors and its incidence with high dose intravenous (IV) lorazepam has been estimated to be as high as 19%.114, 116, 119, 120 This disorder can frequently go unrecognized, as many other factors that induce LA often coincide in such patients. But when identified and promptly addressed, its prognosis seems to be favorable.114
The best treatment is prevention, by avoiding the use of IV lorazepam in patients with impaired renal function. Once it is recognized, the drug should be promptly withdrawn. In addition, removal by hemodialysis can quickly lower propylene glycol levels since it is a small, highly water soluble, non‐protein‐bound molecule.121 As no rebound in the level is expected, intermittent dialysis should be an acceptable modality.117
Linezolid
Recently, Gram‐positive bacteria in general and methicillin‐resistant Staphylococcus aureus in particular have emerged as major causes of nosocomial and community‐acquired infections. Linezolid, an oxazolidinone, is increasingly used to treat such infections. Several cases of LA have been associated with linezolid.27, 122, 123 and a survey of the Infectious Diseases Society of America (IDSA) Emerging Infections Network members revealed that this complication was commonly encountered.124 Linezolid causes LA by mitochondrial toxicity125, 126 and risk factors include prolonged exposure and older age. Once the disorder is recognized, the clinician should stop the drug immediately. Chemistries should be monitored frequently in patients on long‐term therapy.
Conclusions
Many studies note the association between LA and adverse outcomes.2, 4547 Though metabolic acidosis from elevated lactate levels may negatively affect organ function, the evidence supporting therapy specifically aimed at increasing pH in these settings is consistently poor.3, 127 Limitations have included small numbers of subjects,1, 82 variable outcomes studied, and the inability to assess intracellular metabolic stability.1, 61 When taking these factors into account it is hard to justify aggressive treatment of LA with mechanisms aimed at raising pH. Literature on the treatment of patients with LA and very low pH (below 7.2) is even more limited.
Moreover, lactate elevations may not represent tissue hypoperfusion. Lactate may have an important role in improving energy metabolism. This represents 1 additional reason to be hesitant when attempting to normalize pH in LA; we may be disrupting the body's physiologic response to sepsis. A conflict for clinicians emerges, however, as lactate is often used to define tissue ischemia. Obviously, more specific markers of tissue hypoperfusion would be ideal.
Bicarbonate therapy is an understandably attractive means to improve the acidemia, but there are serious mechanistic concerns with it use. Moreover, neither animal nor human studies, limited as they may be, show a convincing benefit. LA in the setting of acute kidney injury may be best treated with renal replacement therapy with bicarbonate‐based buffers, but controlled trials are lacking.
A number of commonly used drugs can cause LA. A heightened awareness on the part of clinicians will lead to prompt recognition of these cases, and timely treatment.
- ,,,.Bicarbonate does not improve hemodynamics in critically ill patients who have lactic acidosis. A prospective, controlled clinical study.Ann Intern Med.1990;112(7):492–498.
- ,,, et al.Natural history and course of acquired lactic acidosis in adults. DCA‐Lactic Acidosis Study Group.Am J Med.1994;97(1):47–54.
- ,.Sodium bicarbonate for the treatment of lactic acidosis.Chest.2000;117(1):260–267.
- ,.Management of life‐threatening acid‐base disorders. First of two parts.N Engl J Med.1998;338(1):26–34.
- .Indications for use of bicarbonate in patients with metabolic acidosis.Br J Anaesth.1991;67(2):165–177.
- ,.Bench‐to‐bedside review: treating acid‐base abnormalities in the intensive care unit—the role of buffers.Crit Care.2004;8(4):259–265.
- .Lactic acidosis update for critical care clinicians.J Am Soc Nephrol.2001;12(suppl 17):S15–S19.
- ,.Lactic acidosis: diagnosis and treatment.Clin Endocrinol Metab.1980;9(3):513–541.
- .Lactate and shock state: the metabolic view.Curr Opin Crit Care.2006;12(4):315–321.
- .Bench‐to‐bedside review: lactate and the kidney.Crit Care.2002;6(4):322–326.
- ,,,.The influence of renal function on lactate and glucose metabolism.Biochem J.1984;219(1):73–78.
- ,.The effect of acidosis on lactate removal by the perfused rat kidney.Clin Sci Mol Med.1976;50(3):185–194.
- ,,.Transvisceral lactate fluxes during early endotoxemia.Chest.1996;110(1):198–204.
- ,,,,.Metabolism of lactate by the intact functioning kidney of the dog.Am J Physiol.1973;224(6):1463–1467.
- ,,, et al.Lactate and glucose metabolism in severe sepsis and cardiogenic shock.Crit Care Med.2005;33(10):2235–2240.
- ,,,, et al.Lactic acidosis in fulminant hepatic failure. Some aspects of pathogenesis and prognosis.J Hepatol.1985;1(4):405–416.
- ,.Lactic acidosis in critical illness.Crit Care Med.1992;20(1):80–93.
- ,,, et al.Effects of cardiogenic shock on lactate and glucose metabolism after heart surgery.Crit Care Med.2000;28(12):3784–3791.
- ,,,,,.Evolution of lactate/pyruvate and arterial ketone body ratios in the early course of catecholamine‐treated septic shock.Crit Care Med.2000;28(1):114–119.
- ,,, et al.Oxygen debt and metabolic acidemia as quantitative predictors of mortality and the severity of the ischemic insult in hemorrhagic shock.Crit Care Med.1991;19(2):231–243.
- ,,, et al.Early goal‐directed therapy in the treatment of severe sepsis and septic shock.N Engl J Med.2001;345(19):1368–1377.
- ,,,.Skeletal muscle partial pressure of oxygen in patients with sepsis.Crit Care Med.1994;22(4):640–650.
- ,.Reevaluation of the role of cellular hypoxia and bioenergetic failure in sepsis.JAMA.1992;267(11):1503–1510.
- ,,,.Lactic acidosis during sepsis is related to increased pyruvate production, not deficits in tissue oxygen availability.Ann Surg.1996;224(1):97–102.
- ,,,.Biguanide‐associated lactic acidosis. Case report and review of the literature.Arch Intern Med.1992;152(11):2333–2336.
- ,,,.Bench‐to‐bedside review: severe lactic acidosis in HIV patients treated with nucleoside analogue reverse transcriptase inhibitors.Crit Care.2003;7(3):226–232.
- ,,,.Lactic acidosis after treatment with linezolid.Infection.2007;35(4):278–281.
- ,,,.Isotopic evaluation of the metabolism of pyruvate and related substrates in normal adult volunteers and severely burned children: effect of dichloroacetate and glucose infusion.Surgery.1991;110(1):54–67.
- .Alterations in carbohydrate metabolism during stress: a review of the literature.Am J Med.1995;98(1):75–84.
- .,,,.Lactic acid kinetics in respiratory alkalosis.Crit Care Med.1991;19(9):1120–1124.
- .Significance of hyperlactatemia without acidosis during hypermetabolic stress.Crit Care Med.1997;25(11):1780–1781.
- ,,,.Lactate is an unreliable indicator of tissue hypoxia in injury or sepsis.Lancet.1999;354(9177):505–508.
- ,,,,.Relation between muscle Na+K+ ATPase activity and raised lactate concentrations in septic shock: a prospective study.Lancet.2005;365(9462):871–875.
- ,.Sepsis induces diaphragm electron transport chain dysfunction and protein depletion.Am J Respir Crit Care Med.2005;172(7):861–868.
- ,,, et al.Association between mitochondrial dysfunction and severity and outcome of septic shock.Lancet.2002;360(9328):219–223.
- ,.Mitochondrial dysfunction in sepsis.Biochem Soc Symp.1999;66:149–166.
- ,,, et al.Altered glucose transporter mRNA abundance in a rat model of endotoxic shock.Biochem Biophys Res Commun.1991;176(1):535–540.
- .Increased pyruvate dehydrogenase kinase activity in response to sepsis.Am J Physiol.1991;260(5 Pt 1):E669–E674.
- .Lactate metabolism: a new paradigm for the third millennium.J Physiol.2004;558(Pt 1):5–30.
- ,,, et al.Evidence supporting the existence of an activity‐dependent astrocyte‐neuron lactate shuttle.Dev Neurosci.1998;20(4–5):291–299.
- ,,,.Brain lactate is an obligatory aerobic energy substrate for functional recovery after hypoxia: further in vitro validation.J Neurochem.1997;69(1):423–426.
- .Bench‐to‐bedside review: a possible resolution of the glucose paradox of cerebral ischemia.Crit Care.2002;6(4):330–334.
- ,,,,,.Intravenous lactate prevents cerebral dysfunction during hypoglycaemia in insulin‐dependent diabetes mellitus.Clin Sci (Lond).1998;94(2):157–163.
- ,,,.Brain lactate, not glucose, fuels the recovery of synaptic function from hypoxia upon reoxygenation: an in vitro study.Brain Res.1997;744(1):105–111.
- ,,,,,.Clinical prognostic markers in patients with severe sepsis: a prospective analysis of 139 consecutive cases.J Infect.2003;47(4):300–306.
- ,,, et al.Serum lactate as a predictor of mortality in patients with infection.Intensive Care Med.2007;33(6):970–977.
- ,,,.Lactate versus non‐lactate metabolic acidosis: a retrospective outcome evaluation of critically ill patients.Crit Care.2006;10(1):R22.
- .Abnormal resting blood lactate. I. The significance of hyperlactatemia in hospitalized patients.Am J Med.1961;30:840–848.
- ,,,,.Effect of lactic acidosis on canine hemodynamics and left ventricular function.Am J Physiol.1990;258(4 Pt 2):H1193–H1199.
- ,,,.Alkali therapy extends the period of survival during hypoxia: studies in rats.Am J Physiol.1996;271(2 Pt 2):R381–R387.
- ,,.Acidosis during early reperfusion prevents myocardial stunning in perfused ferret hearts.J Clin Invest.1988;82(3):920–927.
- ,,, et al.Effect of acidotic blood reperfusion on reperfusion injury after coronary artery occlusion in the dog heart.J Cardiovasc Pharmacol.1998;31(2):179–186.
- ,,,.Hypoxia and acidosis activate cardiac myocyte death through the Bcl‐2 family protein BNIP3.Proc Natl Acad Sci U S A.2002;99(20):12825–12830.
- ,,,,,.Extracellular acidosis delays onset of cell death in ATP‐depleted hepatocytes.Am J Physiol.1988;255(3 Pt 1):C315–C322.
- ,.Metabolic and hemodynamic effects of hypertonic solutions: sodium‐lactate versus sodium chloride infusion in postoperative patients.Shock.2002;18(4):306–310.
- .Medical complications of status epilepticus.Adv Neurol.1983;34:395–398.
- ,,,,,.Bicarbonate therapy in severe diabetic ketoacidosis. A double blind, randomized, placebo controlled trial.Rev Invest Clin.1991;43(3):234–238.
- ,,.Supercarbia in children: clinical course and outcome.Crit Care Med.1990;18(2):166–168.
- ,,,.Low mortality rate in adult respiratory distress syndrome using low‐volume, pressure‐limited ventilation with permissive hypercapnia: a prospective study.Crit Care Med.1994;22(10):1568–1578.
- ,,,.Systemic effects of NaHCO3 in experimental lactic acidosis in dogs.Am J Physiol.1982;242(6):F586–F591.
- ,,,,.Lactic acidosis: effect of treatment on intracellular pH and energetics in living rat heart.Am J Physiol.1992;262(5 Pt 2):H1572–H1578.
- ,,,,,.Effect of sodium bicarbonate on intracellular pH under different buffering conditions.Kidney Int.1996;49(5):1262–1267.
- ,,.Hemodynamic and hepatic pH responses to sodium bicarbonate and Carbicarb during systemic acidosis.Magn Reson Med.1990;16(3):403–410.
- ,,, et al.The increase in CO2 production induced by NaHCO3 depends on blood albumin and hemoglobin concentrations.Intensive Care Med.2000;26(5):558–564.
- ,,, et al.Initial effect of sodium bicarbonate on intracellular pH depends on the extracellular nonbicarbonate buffering capacity.Crit Care Med.2001;29(5):1033–1039.
- ,,,,,.Dynamic study of the distribution of microcirculatory blood flow in multiple splanchnic organs in septic shock.Crit Care Med.2000;28(9):3233–3241.
- ,,,,,.The effects of sodium bicarbonate and a mixture of sodium bicarbonate and carbonate (“Carbicarb”) on skeletal muscle pH and hemodynamic status in rats with hypovolemic shock.Metabolism.1994;43(4):518–522.
- ,,, et al.Haemodynamic and metabolic effects in diabetic ketoacidosis in rats of treatment with sodium bicarbonate or a mixture of sodium bicarbonate and sodium carbonate.Diabetologia.1995;38(8):889–898.
- ,,,,,.Effects of sodium bicarbonate on striated muscle metabolism and intracellular pH during endotoxic shock.Shock.1994;1(3):196–200.
- ,,,,.Carbicarb, sodium bicarbonate, and sodium chloride in hypoxic lactic acidosis. Effect on arterial blood gases, lactate concentrations, hemodynamic variables, and myocardial intracellular pH.Chest.1993;104(3):913–918.
- ,.Improved hemodynamic function during hypoxia with Carbicarb, a new agent for the management of acidosis.Circulation.1988;77(1):227–233.
- ,,.Metabolic effects of sodium bicarbonate in hypoxic lactic acidosis in dogs.Am J Physiol.1985;249(5 Pt 2):F630–F635.
- .Binding of calcium to serum albumin. II. Effect of pH via competitive hydrogen and calcium ion binding to the imidazole groups of albumin.Scand J Clin Lab Invest.1972;29(1):75–83.
- ,,,,.Left ventricular contractility varies directly with blood ionized calcium.Ann Intern Med.1988;108(4):524–529.
- ,,.Regulatory mechanisms of hemoglobin oxygen affinity in acidosis and alkalosis.J Clin Invest.1971;50(3):700–706.
- ,,.Metabolic and hemodynamic consequences of sodium bicarbonate administration in patients with heart disease.Am J Med.1989;87(1):7–14.
- ,,.Sodium ion/hydrogen ion exchange inhibition: a new pharmacologic approach to myocardial ischemia and reperfusion injury.J Clin Pharmacol.1998;38(10):887–897.
- ,.SEA0400: a novel sodium‐calcium exchange inhibitor with cardioprotective properties.Cardiovasc Drug Rev.2004;22(4):334–347.
- ,,,.Elevation in cytosolic free calcium concentration early in myocardial ischemia in perfused rat heart.Circ Res.1987;60(5):700–707.
- ,.Acute haemodynamic effects of sodium bicarbonate administration in respiratory and metabolic acidosis in anaesthetized dogs.Anaesth Intensive Care.1997;25(6):615–620.
- ,,,.Bicarbonate does not increase left ventricular contractility during L‐lactic acidemia in pigs.Am Rev Respir Dis.1993;148(2):317–322.
- ,,,,.Effects of bicarbonate therapy on hemodynamics and tissue oxygenation in patients with lactic acidosis: a prospective, controlled clinical study.Crit Care Med.1991;19(11):1352–1356.
- ,,, et al.Safety and efficacy of intravenous Carbicarb in patients undergoing surgery: comparison with sodium bicarbonate in the treatment of mild metabolic acidosis. SPI Research Group. Study of Perioperative Ischemia.Crit Care Med.1994;22(10):1540–1549.
- ,,, et al.Sodium bicarbonate versus THAM in ICU patients with mild metabolic acidosis.J Nephrol.2005;18(3):303–307.
- ,,, et al.A controlled clinical trial of dichloroacetate for treatment of lactic acidosis in adults. The Dichloroacetate‐Lactic Acidosis Study Group.N Engl J Med.1992;327(22):1564–1569.
- ,,,,,.Effect of continuous venovenous hemofiltration with dialysis on lactate clearance in critically ill patients.Crit Care Med.1997;25(1):58–62.
- ,,,,.A pilot randomised controlled comparison of continuous veno‐venous haemofiltration and extended daily dialysis with filtration: effect on small solutes and acid‐base balance.Intensive Care Med.2007;33(5):830–835.
- ,,.Intermittent versus continuous renal replacement therapy in the ICU: impact on electrolyte and acid‐base balance.Intensive Care Med.2001;27(6):1037–1043.
- ,,,.Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus: systematic review and meta‐analysis.Arch Intern Med.2003;163(21):2594–2602.
- ,,,,.Severe acidosis in patients taking metformin—rapid reversal and survival despite high APACHE score.Diabet Med.2006;23(4):432–435.
- ,,.Incidence of lactic acidosis in metformin users.Diabetes Care.1999;22(6):925–927.
- ,.Lactic acidosis in metformin‐treated patients. Prognostic value of arterial lactate levels and plasma metformin concentrations.Drug Saf.1999;20(4):377–384.
- ,,,.The management of metformin overdose.Anaesthesia.1998;53(7):698–701.
- ,,.Lactic acidosis in biguanide‐treated diabetics: a review of 330 cases.Diabetologia.1978;14(2):75–87.
- ,,,,.Treatment of metformin‐associated lactic acidosis with closed recirculation bicarbonate‐buffered hemodialysis.Arch Intern Med.1984;144(1):203–205.
- ,,,.High anion gap metabolic acidosis in suicide: don't forget metformin intoxication—two patients' experiences.Ren Fail.2002;24(5):671–675.
- ,,, et al.Hemodialysis in the treatment of lactic acidosis in diabetics treated by metformin: a study of metformin elimination.Int J Clin Pharmacol Ther Toxicol.1989;27(6):285–288.
- ,,.Bicarbonate haemodialysis as a treatment of metformin overdose.Nephrol Dial Transplant.1997;12(5):1046–1047.
- ,,,.Combination of intermittent haemodialysis and high‐volume continuous haemofiltration for the treatment of severe metformin‐induced lactic acidosis.Nephrol Dial Transplant.2004;19(8):2157–2158.
- ,,,.When a friend can become an enemy! Recognition and management of metformin‐associated lactic acidosis.Kidney Int.2007;72(9):1157–1160.
- .,, et al.Severe nucleoside‐associated lactic acidosis in human immunodeficiency virus‐infected patients: report of 12 cases and review of the literature.Clin Infect Dis.2002;34(6):838–846.
- ,,.Clearance of metformin by hemofiltration in overdose.J Toxicol Clin Toxicol.2002;40(2):177–180.
- .Metformin‐associated lactic acidosis.J Emerg Med.2001;20(3):267–272.
- ,.Contraindications to use of metformin. Blanket banning of metformin two days before surgery may not be a good idea.BMJ.2003;326(7392):762; author reply 762.
- ,,, et al.Hepatic failure and lactic acidosis due to fialuridine (FIAU), an investigational nucleoside analogue for chronic hepatitis B.N Engl J Med.1995;333(17):1099–1105.
- ,,,.Zidovudine‐induced fatal lactic acidosis and hepatic failure in patients with acquired immunodeficiency syndrome: report of two patients and review of the literature.Crit Care Med.1997;25(8):1425–1430.
- ,.Mitochondrial toxicity of antiviral drugs.Nat Med.1995;1(5):417–422.
- ,.Mitochondrial toxicity of nucleoside analogue reverse transcriptase inhibitors: a looming obstacle for long‐term antiretroviral therapy?Curr Opin Infect Dis.2000;13(1):5–11.
- ,,, et al.Treatment for adult HIV infection: 2006 recommendations of the International AIDS Society‐USA panel.JAMA.2006;296(7):827–843.
- ,,.Riboflavin to treat nucleoside analogue‐induced lactic acidosis.Lancet.1998;352(9124):291–292.
- ,,,,,.Severe lactic acidosis and thiamine administration in an HIV‐infected patient on HAART.Int J STD AIDS.2001;12(6):407–409.
- ,,, et al.Detecting life‐threatening lactic acidosis related to nucleoside‐analog treatment of human immunodeficiency virus‐infected patients, and treatment with L‐carnitine.Crit Care Med.2003;31(4):1042–1047.
- ,.Hyperosmolar metabolic acidosis and intravenous Lorazepam.N Engl J Med.2002;347(11):857–858; author reply 857–858.
- ,,,.Propylene glycol toxicity: a severe iatrogenic illness in ICU patients receiving IV benzodiazepines: a case series and prospective, observational pilot study.Chest.2005;128(3):1674–1681.
- ,,, et al.Propylene glycol pharmacokinetics and effects after intravenous infusion in humans.Ther Drug Monit.1987;9(3):255–258.
- .Short‐term lorazepam infusion and concern for propylene glycol toxicity: case report and review.Pharmacotherapy.2001;21(9):1140–1144.
- ,,.Recognition, treatment, and prevention of propylene glycol toxicity.Semin Dial.2007;20(3):217–219.
- ,,.Propylene glycol‐mediated cell injury in a primary culture of human proximal tubule cells.Toxicol Sci.1998;46(2):410–417.
- ,.Osmolar gap metabolic acidosis in a 60‐year‐old man treated for hypoxemic respiratory failure.Chest.2000;118(2):545–546.
- ,,,.Relationship of continuous infusion lorazepam to serum propylene glycol concentration in critically ill adults.Crit Care Med.2004;32(8):1709–1714.
- ,,,.Removal of propylene glycol and correction of increased osmolar gap by hemodialysis in a patient on high dose lorazepam infusion therapy.Intensive Care Med.2002;28(1):81–84.
- ,,.Linezolid use associated with lactic acidosis.Scand J Infect Dis.2005;37(2):153–154.
- ,.Linezolid‐induced lactic acidosis.N Engl J Med.2003;348(1):86–87.
- ,,.Toxicity of extended courses of linezolid: results of an Infectious Diseases Society of America Emerging Infections Network survey.Diagn Microbiol Infect Dis.2008;62(4):407–410.
- ,,.Mitochondrial toxicity associated with linezolid.N Engl J Med.2005;353(21):2305–2306.
- ,,, et al.Linezolid‐induced inhibition of mitochondrial protein synthesis.Clin Infect Dis.2006;42(8):1111–1117.
- ,.Use of base in the treatment of severe acidemic states.Am J Kidney Dis.2001;38(4):703–727.
Lactic acidosis (LA) is common in hospitalized patients and is associated with a high mortality.1, 2 Commonly, it is defined as a lactic acid concentration greater than 5 mmol/L with a pH less than 7.35.3 There are no evidence‐based guidelines for the treatment of LA despite progress in our understanding of its pathophysiology.36 This is not surprising, given the uncertainty regarding the impact of LA itself on clinical outcomes. In this regard, it is interesting to note that, despite its well‐recognized role as a marker of tissue hypoxia, lactate accumulation appears to have beneficial effects and may function as an adaptive mechanism. This raises the possibility that therapy directed at altering this adaptation may be detrimental. Pursuing correction of the pH in LA has been shown to have untoward physiologic effects. These and other ambiguities in the pathophysiology and treatment of LA are the focus of this review.
Lactate Metabolism
The body produces approximately 1400 mmol of lactate daily.7 Lactate is derived from the metabolism of pyruvate through an anaerobic reaction that occurs in all tissues (Figure 1). The liver is the primary site of lactate clearance and can metabolize up to 100 mmol per hour under normal conditions.8 There, lactate is converted to glucose to serve as an energy source during periods of hypoxia (Figure 2).9


Approximately 20% to 30% of the daily lactate load is metabolized by the kidneys.10, 11 Renal clearance is increased in acidosis12 and is maintained even in the presence of low renal perfusion.10, 12, 13 Renal lactate clearance is primarily through metabolism and not excretion.10, 14
LA Subtypes
Generally, lactic acid accumulation results from excess lactic acid production and not from reduced clearance.15 In cases of fulminant liver failure, it is due to a combination of decreased clearance and tissue hypoxia.16 In the setting of tissue hypoxia, an impairment of mitochondrial oxidative capacity results in the accumulation of pyruvate and generation of lactate. Lactic acid accumulation through this mechanism has historically been described as Type A LA.7 Hence, in critically ill patients lactate has traditionally been viewed as a marker of tissue hypoxia.15, 1721 Hyperlactatemia without tissue hypoxia has been referred to as type B LA. This is seen in a variety of circumstances. In sepsis, for example, several studies have shown lactic acid accumulation, despite adequate oxygen delivery.2224
Hyperlactatemia may also occur in cases of pure mitochondrial dysfunction, which can be induced by commonly prescribed medications such as the biguanides, nucleoside analog reverse‐transcriptase inhibitors (NRTIs), and linezolid.2527 Alternatively, lactate generation from metabolism of agents such as propylene glycol is possible. Finally, excessive lactate generation may occur following stress due to altered carbohydrate metabolism, or with respiratory alkalosis.2831
Lactate: A Metabolic Adaptation
Lactate was traditionally considered only as a marker of tissue hypoxia and anaerobic metabolism.17 This is certainly the case in situations of poor perfusion such as cardiogenic,15, 18 vasopressor‐resistant,19 or hypovolemic shock.20, 21
Alternative explanations for lactic acid accumulation, without tissue hypoperfusion, include catecholamine‐induced alterations in glycolysis,32, 33 mitochondrial disturbances,3436 and increased pyruvate production combined with increased glucose entry into cells.24, 37 In addition, the activity of an enzyme regulating lactate metabolism, pyruvate dehydrogenase kinase, increases in sepsis.38 This enzyme inactivates the pyruvate dehydrogenase (PDH) complex, which metabolizes pyruvate. Pyruvate and lactate may accumulate as a result. These changes partly explain the generation of LA in sepsis, independent of any effect of diminished tissue perfusion.
Recognizing the body's tendency toward homeostasis, it is appealing to speculate that lactate accumulation is adaptive.9 A number of findings support this. For example, lactate may act to shuttle energy between organs, or between cell types in the same organ. The astrocyteneuron lactate shuttle and the spermatogenic lactate shuttle are 2 examples of lactate's valuable effects on cellular metabolism.39 In the astrocyteneuron lactate shuttle, astrocytes support the increased metabolic demands of neurons through lactic acid production.40 Specifically, the neurotransmitter glutamate is released by the neurons and taken up by the astrocytes. Astrocytes produce lactate, which then moves back to the neuron to be used as an energy source. Glutamine, also released by the astrocytes, leads to the regeneration of glutamate and the potential to restart the cycle.39
Animal and human studies have suggested that, in periods of stress, lactate is the preferential energy substrate in the brain.4144 The usefulness of increased lactate production routinely seen in sepsis may thus represent multiple adaptive processes aimed primarily at improving the delivery of energy substrates. Thus, therapeutic strategies aimed specifically at lowering lactic acid levels may prove to have deleterious effects on cellular metabolism.
Impact of LA on Morbidity and Mortality
The poor prognosis in patients with LA is well recognized.2, 4548 For example, in a study of 126 patients with various causes of LA, the median survival was 38.5 hours and 30‐day survival was 17%.2
Studies have revealed that LA with low pH is associated with adverse effects on the cardiovascular system, particularly a decrease in cardiac contractility.49, 50 This effect is particularly prominent with a pH below 7.20. In contrast, acidosis in animal models has been shown to limit myocardial infarct size after reperfusion.51, 52 Variable effects of LA on cell death have been found. A worsening of apoptosis in myocytes has been noted;53 alternatively, protection from hypoxic injury in hepatocytes and myocardium has been observed.52, 54 Thus, although LA is associated with poor outcomes in human studies,2, 4547 it is still unclear to what extent lactic acid accumulation is a marker of severe illness, an independent effector of pathology, or a mechanism with the potential to serve a protective role.
Available data indicate that lactate itself is not harmful. Studies on infusion of lactate solutions to postoperative patients was shown to be safe.55 Also, the fact that lactate generation in states of respiratory alkalosis, stress, or altered carbohydrate metabolism without sepsis is not associated with worse outcomes supports the fact that lactic acid alone may not be maladaptive.2831
Similarly, low pH is not necessarily maladaptive. In the postictal state,56 diabetic ketoacidosis,57 spontaneous respiratory acidosis,58 or permissive hypercapnia,59 low blood pH is not deleterious.
In summary, LA is associated with poor outcomes, and indirect evidence suggests that it is the underlying causative condition rather than the low pH or the lactate that is responsible for the dire outcomes.
Treatment of LA with Sodium Bicarbonate
Since excessive lactic acid generation is accompanied by consumption of plasma bicarbonate and a fall in plasma pH, sodium bicarbonate has been long proposed as a treatment for LA. While theoretically appealing, this strategy has not been validated by studies in animals or humans. Indeed, bicarbonate administration in LA often has been shown to be detrimental.60, 61 The adverse effects of bicarbonate administration in LA, while initially paradoxical, have a number of possible explanations.
First, bicarbonate administration can induce a reduction in intracellular pH.60, 62, 63 The mechanism involves bicarbonate's effect to increase carbon dioxide (CO2) generation through mass action effect. Because the cell membrane is more permeable to CO2 than to bicarbonate, intracellular pH falls.64, 65 In sepsis, this intracellular/extracellular pH discrepancy may be more pronounced due to alterations in blood flow.66 Other reports on outcomes of intracellular pH with bicarbonate therapy show variable effects.6772
Second, to the extent that bicarbonate administration raises extracellular pH, it is associated with a reduction in ionized calcium concentration, since the binding of calcium to albumin is pH dependent.73 A sodium bicarbonate load administered to patients with LA was associated with a significant fall in ionized calcium concentration, whereas a sodium chloride load was not.1 This can affect cardiac function, as the latter varies proportionally with calcium levels.74
Third, bicarbonate administration may reduce tissue oxygen delivery since the affinity of hemoglobin for oxygen increases as pH rises (Bohr effect).75 The administration of bicarbonate worsened systemic oxygen consumption in one study76 and decreased oxygen delivery in another.75
Fourth, bicarbonate administration may indirectly increase intracellular calcium concentration. Low intracellular pH (see above) stimulates proton efflux by way of proton transporters and exchangers, increasing intracellular sodium content.77 A high cell sodium content then may increase intracellular calcium, through the Na/Ca exchanger, impairing cellular function.7779 Compounding this, the reduced function of the Na/H ATPase as a regulator of intracellular sodium in sepsis may not be adequate to limit cell swelling.77
Against this background of mechanistic concerns with the use of bicarbonate treatment, it is not surprising that clinical outcomes have been inconsistent at best. In animal models of LA, the use of sodium bicarbonate has either negative effects on cardiac output60, 72 or no significant hemodynamic effect when compared to sodium chloride infusion.67, 80, 81 One animal study did show some benefit with sodium bicarbonate compared to saline, though all animals subsequently died.50
In humans, sodium bicarbonate was studied in 2 randomized trials of sepsis‐induced LA.1, 82 In a study by Cooper et al.,1 14 critically‐ill patients received sequential infusions of sodium bicarbonate or sodium chloride. Neither solution was superior to the other in terms of hemodynamic improvement. No benefit was noted even when analysis was limited to those with very low pH (7.2). Mathieu et al.82 randomized 10 critically‐ill patients to sequential infusion of either sodium bicarbonate or sodium chloride. Similarly, no significant difference in hemodynamic variables was noted.
When taken together, these studies evaluating sodium bicarbonate in LA fail to show convincing benefit and raise serious questions about its detrimental effects. Extracellular pH may be a misleading marker of success in the treatment of LA, given its direct influence by sodium bicarbonate administration.
Treatment of LA and Use of Other Buffers
Other buffers (Carbicarb, dichloroacetate, and tromethamine [THAM]) have been studied for treatment of LA. Human studies have not shown superiority of any of the buffers as far as improving pH,83, 84 hemodynamics, or survival.85
Treatment of LA by Renal Replacement Therapy
Renal replacement therapy (RRT; dialysis and its variants) has been studied for the treatment of severe acidosis. RRT has a number of theoretical advantages over purely medical therapies in the treatment of LA: it can deliver large quantities of base without contributing to volume overload; it can directly remove lactate from the plasma; and it can mitigate the effect of alkalinization on ionized calcium concentration by delivering calcium.
In critically ill patients with intact liver function, continuous venovenous hemofiltration (CVVH) appears to contribute very little (less then 3%) to overall lactate clearance.86 While outcome studies are limited, continuous dialysis modalities consistently show improved resolution of acidosis of various types when compared to intermittent modalities.87, 88 As described above, this is related to base administration and is not a surprising finding. There are no studies comparing RRT and medical therapy with respect to clinical outcomes in patients with LA.
Special Situations
Biguanides
Biguanide‐induced LA can be due to impairment of hepatic neoglucogenesis, in the case of metformin, or increasing hepatic oxidative phosphorylation, in the case of phenformin.89 This infrequent complication90, 91 is associated with a high mortality.92 Proposed therapy has included the use of sodium bicarbonate infusion.93 In this setting, it is unclear if the use of bicarbonate alone improves clinical outcomes.94
Renal replacement therapy in a wide variety of formats has been used to treat this condition.93, 95101 Metformin has a high clearance during dialysis due to its low molecular weight and lack of protein binding.97, 98, 102 Nonetheless, its high volume of distribution suggests a longer dialysis time would be more beneficial if the main goal is reducing metformin levels.97, 103 The limited prospective literature and lack of conclusive evidence about what levels of metformin induce LA makes generalized recommendations about duration of hemodialysis purely speculative.104
NRTIs
The use of NRTIs is associated with LA due to impairment of mitochondrial oxidative phosphorylation.105108 This uncommon complication, if not recognized early, is associated with a high mortality.101, 109 Investigations are ongoing into agents directed at improving mitochondrial function such as riboflavin, thiamine, and L‐carnitine.110112 As with biguanide‐associated LA, RRT decisions should be individualized based on metabolic circumstances.
Lorazepam
Many intravenous medications are formulated in the alcohol solvent, propylene glycol. Injectable lorazepam has the highest proportional amount of propylene glycol compared with other commonly used agents.113, 114 The kidney normally eliminates 12% to 50% of administered propylene glycol via proximal tubule secretion.115 The remainder is metabolized by the liver to form pyruvate and lactate.114, 116, 117
When propylene glycol accumulates, as in cases of reduced renal function, it results in hyperosmolarity, LA, and can even induce additional kidney injury (probably through proximal tubular cell necrosis).118
LA due to propylene glycol has been reported by many authors and its incidence with high dose intravenous (IV) lorazepam has been estimated to be as high as 19%.114, 116, 119, 120 This disorder can frequently go unrecognized, as many other factors that induce LA often coincide in such patients. But when identified and promptly addressed, its prognosis seems to be favorable.114
The best treatment is prevention, by avoiding the use of IV lorazepam in patients with impaired renal function. Once it is recognized, the drug should be promptly withdrawn. In addition, removal by hemodialysis can quickly lower propylene glycol levels since it is a small, highly water soluble, non‐protein‐bound molecule.121 As no rebound in the level is expected, intermittent dialysis should be an acceptable modality.117
Linezolid
Recently, Gram‐positive bacteria in general and methicillin‐resistant Staphylococcus aureus in particular have emerged as major causes of nosocomial and community‐acquired infections. Linezolid, an oxazolidinone, is increasingly used to treat such infections. Several cases of LA have been associated with linezolid.27, 122, 123 and a survey of the Infectious Diseases Society of America (IDSA) Emerging Infections Network members revealed that this complication was commonly encountered.124 Linezolid causes LA by mitochondrial toxicity125, 126 and risk factors include prolonged exposure and older age. Once the disorder is recognized, the clinician should stop the drug immediately. Chemistries should be monitored frequently in patients on long‐term therapy.
Conclusions
Many studies note the association between LA and adverse outcomes.2, 4547 Though metabolic acidosis from elevated lactate levels may negatively affect organ function, the evidence supporting therapy specifically aimed at increasing pH in these settings is consistently poor.3, 127 Limitations have included small numbers of subjects,1, 82 variable outcomes studied, and the inability to assess intracellular metabolic stability.1, 61 When taking these factors into account it is hard to justify aggressive treatment of LA with mechanisms aimed at raising pH. Literature on the treatment of patients with LA and very low pH (below 7.2) is even more limited.
Moreover, lactate elevations may not represent tissue hypoperfusion. Lactate may have an important role in improving energy metabolism. This represents 1 additional reason to be hesitant when attempting to normalize pH in LA; we may be disrupting the body's physiologic response to sepsis. A conflict for clinicians emerges, however, as lactate is often used to define tissue ischemia. Obviously, more specific markers of tissue hypoperfusion would be ideal.
Bicarbonate therapy is an understandably attractive means to improve the acidemia, but there are serious mechanistic concerns with it use. Moreover, neither animal nor human studies, limited as they may be, show a convincing benefit. LA in the setting of acute kidney injury may be best treated with renal replacement therapy with bicarbonate‐based buffers, but controlled trials are lacking.
A number of commonly used drugs can cause LA. A heightened awareness on the part of clinicians will lead to prompt recognition of these cases, and timely treatment.
Lactic acidosis (LA) is common in hospitalized patients and is associated with a high mortality.1, 2 Commonly, it is defined as a lactic acid concentration greater than 5 mmol/L with a pH less than 7.35.3 There are no evidence‐based guidelines for the treatment of LA despite progress in our understanding of its pathophysiology.36 This is not surprising, given the uncertainty regarding the impact of LA itself on clinical outcomes. In this regard, it is interesting to note that, despite its well‐recognized role as a marker of tissue hypoxia, lactate accumulation appears to have beneficial effects and may function as an adaptive mechanism. This raises the possibility that therapy directed at altering this adaptation may be detrimental. Pursuing correction of the pH in LA has been shown to have untoward physiologic effects. These and other ambiguities in the pathophysiology and treatment of LA are the focus of this review.
Lactate Metabolism
The body produces approximately 1400 mmol of lactate daily.7 Lactate is derived from the metabolism of pyruvate through an anaerobic reaction that occurs in all tissues (Figure 1). The liver is the primary site of lactate clearance and can metabolize up to 100 mmol per hour under normal conditions.8 There, lactate is converted to glucose to serve as an energy source during periods of hypoxia (Figure 2).9


Approximately 20% to 30% of the daily lactate load is metabolized by the kidneys.10, 11 Renal clearance is increased in acidosis12 and is maintained even in the presence of low renal perfusion.10, 12, 13 Renal lactate clearance is primarily through metabolism and not excretion.10, 14
LA Subtypes
Generally, lactic acid accumulation results from excess lactic acid production and not from reduced clearance.15 In cases of fulminant liver failure, it is due to a combination of decreased clearance and tissue hypoxia.16 In the setting of tissue hypoxia, an impairment of mitochondrial oxidative capacity results in the accumulation of pyruvate and generation of lactate. Lactic acid accumulation through this mechanism has historically been described as Type A LA.7 Hence, in critically ill patients lactate has traditionally been viewed as a marker of tissue hypoxia.15, 1721 Hyperlactatemia without tissue hypoxia has been referred to as type B LA. This is seen in a variety of circumstances. In sepsis, for example, several studies have shown lactic acid accumulation, despite adequate oxygen delivery.2224
Hyperlactatemia may also occur in cases of pure mitochondrial dysfunction, which can be induced by commonly prescribed medications such as the biguanides, nucleoside analog reverse‐transcriptase inhibitors (NRTIs), and linezolid.2527 Alternatively, lactate generation from metabolism of agents such as propylene glycol is possible. Finally, excessive lactate generation may occur following stress due to altered carbohydrate metabolism, or with respiratory alkalosis.2831
Lactate: A Metabolic Adaptation
Lactate was traditionally considered only as a marker of tissue hypoxia and anaerobic metabolism.17 This is certainly the case in situations of poor perfusion such as cardiogenic,15, 18 vasopressor‐resistant,19 or hypovolemic shock.20, 21
Alternative explanations for lactic acid accumulation, without tissue hypoperfusion, include catecholamine‐induced alterations in glycolysis,32, 33 mitochondrial disturbances,3436 and increased pyruvate production combined with increased glucose entry into cells.24, 37 In addition, the activity of an enzyme regulating lactate metabolism, pyruvate dehydrogenase kinase, increases in sepsis.38 This enzyme inactivates the pyruvate dehydrogenase (PDH) complex, which metabolizes pyruvate. Pyruvate and lactate may accumulate as a result. These changes partly explain the generation of LA in sepsis, independent of any effect of diminished tissue perfusion.
Recognizing the body's tendency toward homeostasis, it is appealing to speculate that lactate accumulation is adaptive.9 A number of findings support this. For example, lactate may act to shuttle energy between organs, or between cell types in the same organ. The astrocyteneuron lactate shuttle and the spermatogenic lactate shuttle are 2 examples of lactate's valuable effects on cellular metabolism.39 In the astrocyteneuron lactate shuttle, astrocytes support the increased metabolic demands of neurons through lactic acid production.40 Specifically, the neurotransmitter glutamate is released by the neurons and taken up by the astrocytes. Astrocytes produce lactate, which then moves back to the neuron to be used as an energy source. Glutamine, also released by the astrocytes, leads to the regeneration of glutamate and the potential to restart the cycle.39
Animal and human studies have suggested that, in periods of stress, lactate is the preferential energy substrate in the brain.4144 The usefulness of increased lactate production routinely seen in sepsis may thus represent multiple adaptive processes aimed primarily at improving the delivery of energy substrates. Thus, therapeutic strategies aimed specifically at lowering lactic acid levels may prove to have deleterious effects on cellular metabolism.
Impact of LA on Morbidity and Mortality
The poor prognosis in patients with LA is well recognized.2, 4548 For example, in a study of 126 patients with various causes of LA, the median survival was 38.5 hours and 30‐day survival was 17%.2
Studies have revealed that LA with low pH is associated with adverse effects on the cardiovascular system, particularly a decrease in cardiac contractility.49, 50 This effect is particularly prominent with a pH below 7.20. In contrast, acidosis in animal models has been shown to limit myocardial infarct size after reperfusion.51, 52 Variable effects of LA on cell death have been found. A worsening of apoptosis in myocytes has been noted;53 alternatively, protection from hypoxic injury in hepatocytes and myocardium has been observed.52, 54 Thus, although LA is associated with poor outcomes in human studies,2, 4547 it is still unclear to what extent lactic acid accumulation is a marker of severe illness, an independent effector of pathology, or a mechanism with the potential to serve a protective role.
Available data indicate that lactate itself is not harmful. Studies on infusion of lactate solutions to postoperative patients was shown to be safe.55 Also, the fact that lactate generation in states of respiratory alkalosis, stress, or altered carbohydrate metabolism without sepsis is not associated with worse outcomes supports the fact that lactic acid alone may not be maladaptive.2831
Similarly, low pH is not necessarily maladaptive. In the postictal state,56 diabetic ketoacidosis,57 spontaneous respiratory acidosis,58 or permissive hypercapnia,59 low blood pH is not deleterious.
In summary, LA is associated with poor outcomes, and indirect evidence suggests that it is the underlying causative condition rather than the low pH or the lactate that is responsible for the dire outcomes.
Treatment of LA with Sodium Bicarbonate
Since excessive lactic acid generation is accompanied by consumption of plasma bicarbonate and a fall in plasma pH, sodium bicarbonate has been long proposed as a treatment for LA. While theoretically appealing, this strategy has not been validated by studies in animals or humans. Indeed, bicarbonate administration in LA often has been shown to be detrimental.60, 61 The adverse effects of bicarbonate administration in LA, while initially paradoxical, have a number of possible explanations.
First, bicarbonate administration can induce a reduction in intracellular pH.60, 62, 63 The mechanism involves bicarbonate's effect to increase carbon dioxide (CO2) generation through mass action effect. Because the cell membrane is more permeable to CO2 than to bicarbonate, intracellular pH falls.64, 65 In sepsis, this intracellular/extracellular pH discrepancy may be more pronounced due to alterations in blood flow.66 Other reports on outcomes of intracellular pH with bicarbonate therapy show variable effects.6772
Second, to the extent that bicarbonate administration raises extracellular pH, it is associated with a reduction in ionized calcium concentration, since the binding of calcium to albumin is pH dependent.73 A sodium bicarbonate load administered to patients with LA was associated with a significant fall in ionized calcium concentration, whereas a sodium chloride load was not.1 This can affect cardiac function, as the latter varies proportionally with calcium levels.74
Third, bicarbonate administration may reduce tissue oxygen delivery since the affinity of hemoglobin for oxygen increases as pH rises (Bohr effect).75 The administration of bicarbonate worsened systemic oxygen consumption in one study76 and decreased oxygen delivery in another.75
Fourth, bicarbonate administration may indirectly increase intracellular calcium concentration. Low intracellular pH (see above) stimulates proton efflux by way of proton transporters and exchangers, increasing intracellular sodium content.77 A high cell sodium content then may increase intracellular calcium, through the Na/Ca exchanger, impairing cellular function.7779 Compounding this, the reduced function of the Na/H ATPase as a regulator of intracellular sodium in sepsis may not be adequate to limit cell swelling.77
Against this background of mechanistic concerns with the use of bicarbonate treatment, it is not surprising that clinical outcomes have been inconsistent at best. In animal models of LA, the use of sodium bicarbonate has either negative effects on cardiac output60, 72 or no significant hemodynamic effect when compared to sodium chloride infusion.67, 80, 81 One animal study did show some benefit with sodium bicarbonate compared to saline, though all animals subsequently died.50
In humans, sodium bicarbonate was studied in 2 randomized trials of sepsis‐induced LA.1, 82 In a study by Cooper et al.,1 14 critically‐ill patients received sequential infusions of sodium bicarbonate or sodium chloride. Neither solution was superior to the other in terms of hemodynamic improvement. No benefit was noted even when analysis was limited to those with very low pH (7.2). Mathieu et al.82 randomized 10 critically‐ill patients to sequential infusion of either sodium bicarbonate or sodium chloride. Similarly, no significant difference in hemodynamic variables was noted.
When taken together, these studies evaluating sodium bicarbonate in LA fail to show convincing benefit and raise serious questions about its detrimental effects. Extracellular pH may be a misleading marker of success in the treatment of LA, given its direct influence by sodium bicarbonate administration.
Treatment of LA and Use of Other Buffers
Other buffers (Carbicarb, dichloroacetate, and tromethamine [THAM]) have been studied for treatment of LA. Human studies have not shown superiority of any of the buffers as far as improving pH,83, 84 hemodynamics, or survival.85
Treatment of LA by Renal Replacement Therapy
Renal replacement therapy (RRT; dialysis and its variants) has been studied for the treatment of severe acidosis. RRT has a number of theoretical advantages over purely medical therapies in the treatment of LA: it can deliver large quantities of base without contributing to volume overload; it can directly remove lactate from the plasma; and it can mitigate the effect of alkalinization on ionized calcium concentration by delivering calcium.
In critically ill patients with intact liver function, continuous venovenous hemofiltration (CVVH) appears to contribute very little (less then 3%) to overall lactate clearance.86 While outcome studies are limited, continuous dialysis modalities consistently show improved resolution of acidosis of various types when compared to intermittent modalities.87, 88 As described above, this is related to base administration and is not a surprising finding. There are no studies comparing RRT and medical therapy with respect to clinical outcomes in patients with LA.
Special Situations
Biguanides
Biguanide‐induced LA can be due to impairment of hepatic neoglucogenesis, in the case of metformin, or increasing hepatic oxidative phosphorylation, in the case of phenformin.89 This infrequent complication90, 91 is associated with a high mortality.92 Proposed therapy has included the use of sodium bicarbonate infusion.93 In this setting, it is unclear if the use of bicarbonate alone improves clinical outcomes.94
Renal replacement therapy in a wide variety of formats has been used to treat this condition.93, 95101 Metformin has a high clearance during dialysis due to its low molecular weight and lack of protein binding.97, 98, 102 Nonetheless, its high volume of distribution suggests a longer dialysis time would be more beneficial if the main goal is reducing metformin levels.97, 103 The limited prospective literature and lack of conclusive evidence about what levels of metformin induce LA makes generalized recommendations about duration of hemodialysis purely speculative.104
NRTIs
The use of NRTIs is associated with LA due to impairment of mitochondrial oxidative phosphorylation.105108 This uncommon complication, if not recognized early, is associated with a high mortality.101, 109 Investigations are ongoing into agents directed at improving mitochondrial function such as riboflavin, thiamine, and L‐carnitine.110112 As with biguanide‐associated LA, RRT decisions should be individualized based on metabolic circumstances.
Lorazepam
Many intravenous medications are formulated in the alcohol solvent, propylene glycol. Injectable lorazepam has the highest proportional amount of propylene glycol compared with other commonly used agents.113, 114 The kidney normally eliminates 12% to 50% of administered propylene glycol via proximal tubule secretion.115 The remainder is metabolized by the liver to form pyruvate and lactate.114, 116, 117
When propylene glycol accumulates, as in cases of reduced renal function, it results in hyperosmolarity, LA, and can even induce additional kidney injury (probably through proximal tubular cell necrosis).118
LA due to propylene glycol has been reported by many authors and its incidence with high dose intravenous (IV) lorazepam has been estimated to be as high as 19%.114, 116, 119, 120 This disorder can frequently go unrecognized, as many other factors that induce LA often coincide in such patients. But when identified and promptly addressed, its prognosis seems to be favorable.114
The best treatment is prevention, by avoiding the use of IV lorazepam in patients with impaired renal function. Once it is recognized, the drug should be promptly withdrawn. In addition, removal by hemodialysis can quickly lower propylene glycol levels since it is a small, highly water soluble, non‐protein‐bound molecule.121 As no rebound in the level is expected, intermittent dialysis should be an acceptable modality.117
Linezolid
Recently, Gram‐positive bacteria in general and methicillin‐resistant Staphylococcus aureus in particular have emerged as major causes of nosocomial and community‐acquired infections. Linezolid, an oxazolidinone, is increasingly used to treat such infections. Several cases of LA have been associated with linezolid.27, 122, 123 and a survey of the Infectious Diseases Society of America (IDSA) Emerging Infections Network members revealed that this complication was commonly encountered.124 Linezolid causes LA by mitochondrial toxicity125, 126 and risk factors include prolonged exposure and older age. Once the disorder is recognized, the clinician should stop the drug immediately. Chemistries should be monitored frequently in patients on long‐term therapy.
Conclusions
Many studies note the association between LA and adverse outcomes.2, 4547 Though metabolic acidosis from elevated lactate levels may negatively affect organ function, the evidence supporting therapy specifically aimed at increasing pH in these settings is consistently poor.3, 127 Limitations have included small numbers of subjects,1, 82 variable outcomes studied, and the inability to assess intracellular metabolic stability.1, 61 When taking these factors into account it is hard to justify aggressive treatment of LA with mechanisms aimed at raising pH. Literature on the treatment of patients with LA and very low pH (below 7.2) is even more limited.
Moreover, lactate elevations may not represent tissue hypoperfusion. Lactate may have an important role in improving energy metabolism. This represents 1 additional reason to be hesitant when attempting to normalize pH in LA; we may be disrupting the body's physiologic response to sepsis. A conflict for clinicians emerges, however, as lactate is often used to define tissue ischemia. Obviously, more specific markers of tissue hypoperfusion would be ideal.
Bicarbonate therapy is an understandably attractive means to improve the acidemia, but there are serious mechanistic concerns with it use. Moreover, neither animal nor human studies, limited as they may be, show a convincing benefit. LA in the setting of acute kidney injury may be best treated with renal replacement therapy with bicarbonate‐based buffers, but controlled trials are lacking.
A number of commonly used drugs can cause LA. A heightened awareness on the part of clinicians will lead to prompt recognition of these cases, and timely treatment.
- ,,,.Bicarbonate does not improve hemodynamics in critically ill patients who have lactic acidosis. A prospective, controlled clinical study.Ann Intern Med.1990;112(7):492–498.
- ,,, et al.Natural history and course of acquired lactic acidosis in adults. DCA‐Lactic Acidosis Study Group.Am J Med.1994;97(1):47–54.
- ,.Sodium bicarbonate for the treatment of lactic acidosis.Chest.2000;117(1):260–267.
- ,.Management of life‐threatening acid‐base disorders. First of two parts.N Engl J Med.1998;338(1):26–34.
- .Indications for use of bicarbonate in patients with metabolic acidosis.Br J Anaesth.1991;67(2):165–177.
- ,.Bench‐to‐bedside review: treating acid‐base abnormalities in the intensive care unit—the role of buffers.Crit Care.2004;8(4):259–265.
- .Lactic acidosis update for critical care clinicians.J Am Soc Nephrol.2001;12(suppl 17):S15–S19.
- ,.Lactic acidosis: diagnosis and treatment.Clin Endocrinol Metab.1980;9(3):513–541.
- .Lactate and shock state: the metabolic view.Curr Opin Crit Care.2006;12(4):315–321.
- .Bench‐to‐bedside review: lactate and the kidney.Crit Care.2002;6(4):322–326.
- ,,,.The influence of renal function on lactate and glucose metabolism.Biochem J.1984;219(1):73–78.
- ,.The effect of acidosis on lactate removal by the perfused rat kidney.Clin Sci Mol Med.1976;50(3):185–194.
- ,,.Transvisceral lactate fluxes during early endotoxemia.Chest.1996;110(1):198–204.
- ,,,,.Metabolism of lactate by the intact functioning kidney of the dog.Am J Physiol.1973;224(6):1463–1467.
- ,,, et al.Lactate and glucose metabolism in severe sepsis and cardiogenic shock.Crit Care Med.2005;33(10):2235–2240.
- ,,,, et al.Lactic acidosis in fulminant hepatic failure. Some aspects of pathogenesis and prognosis.J Hepatol.1985;1(4):405–416.
- ,.Lactic acidosis in critical illness.Crit Care Med.1992;20(1):80–93.
- ,,, et al.Effects of cardiogenic shock on lactate and glucose metabolism after heart surgery.Crit Care Med.2000;28(12):3784–3791.
- ,,,,,.Evolution of lactate/pyruvate and arterial ketone body ratios in the early course of catecholamine‐treated septic shock.Crit Care Med.2000;28(1):114–119.
- ,,, et al.Oxygen debt and metabolic acidemia as quantitative predictors of mortality and the severity of the ischemic insult in hemorrhagic shock.Crit Care Med.1991;19(2):231–243.
- ,,, et al.Early goal‐directed therapy in the treatment of severe sepsis and septic shock.N Engl J Med.2001;345(19):1368–1377.
- ,,,.Skeletal muscle partial pressure of oxygen in patients with sepsis.Crit Care Med.1994;22(4):640–650.
- ,.Reevaluation of the role of cellular hypoxia and bioenergetic failure in sepsis.JAMA.1992;267(11):1503–1510.
- ,,,.Lactic acidosis during sepsis is related to increased pyruvate production, not deficits in tissue oxygen availability.Ann Surg.1996;224(1):97–102.
- ,,,.Biguanide‐associated lactic acidosis. Case report and review of the literature.Arch Intern Med.1992;152(11):2333–2336.
- ,,,.Bench‐to‐bedside review: severe lactic acidosis in HIV patients treated with nucleoside analogue reverse transcriptase inhibitors.Crit Care.2003;7(3):226–232.
- ,,,.Lactic acidosis after treatment with linezolid.Infection.2007;35(4):278–281.
- ,,,.Isotopic evaluation of the metabolism of pyruvate and related substrates in normal adult volunteers and severely burned children: effect of dichloroacetate and glucose infusion.Surgery.1991;110(1):54–67.
- .Alterations in carbohydrate metabolism during stress: a review of the literature.Am J Med.1995;98(1):75–84.
- .,,,.Lactic acid kinetics in respiratory alkalosis.Crit Care Med.1991;19(9):1120–1124.
- .Significance of hyperlactatemia without acidosis during hypermetabolic stress.Crit Care Med.1997;25(11):1780–1781.
- ,,,.Lactate is an unreliable indicator of tissue hypoxia in injury or sepsis.Lancet.1999;354(9177):505–508.
- ,,,,.Relation between muscle Na+K+ ATPase activity and raised lactate concentrations in septic shock: a prospective study.Lancet.2005;365(9462):871–875.
- ,.Sepsis induces diaphragm electron transport chain dysfunction and protein depletion.Am J Respir Crit Care Med.2005;172(7):861–868.
- ,,, et al.Association between mitochondrial dysfunction and severity and outcome of septic shock.Lancet.2002;360(9328):219–223.
- ,.Mitochondrial dysfunction in sepsis.Biochem Soc Symp.1999;66:149–166.
- ,,, et al.Altered glucose transporter mRNA abundance in a rat model of endotoxic shock.Biochem Biophys Res Commun.1991;176(1):535–540.
- .Increased pyruvate dehydrogenase kinase activity in response to sepsis.Am J Physiol.1991;260(5 Pt 1):E669–E674.
- .Lactate metabolism: a new paradigm for the third millennium.J Physiol.2004;558(Pt 1):5–30.
- ,,, et al.Evidence supporting the existence of an activity‐dependent astrocyte‐neuron lactate shuttle.Dev Neurosci.1998;20(4–5):291–299.
- ,,,.Brain lactate is an obligatory aerobic energy substrate for functional recovery after hypoxia: further in vitro validation.J Neurochem.1997;69(1):423–426.
- .Bench‐to‐bedside review: a possible resolution of the glucose paradox of cerebral ischemia.Crit Care.2002;6(4):330–334.
- ,,,,,.Intravenous lactate prevents cerebral dysfunction during hypoglycaemia in insulin‐dependent diabetes mellitus.Clin Sci (Lond).1998;94(2):157–163.
- ,,,.Brain lactate, not glucose, fuels the recovery of synaptic function from hypoxia upon reoxygenation: an in vitro study.Brain Res.1997;744(1):105–111.
- ,,,,,.Clinical prognostic markers in patients with severe sepsis: a prospective analysis of 139 consecutive cases.J Infect.2003;47(4):300–306.
- ,,, et al.Serum lactate as a predictor of mortality in patients with infection.Intensive Care Med.2007;33(6):970–977.
- ,,,.Lactate versus non‐lactate metabolic acidosis: a retrospective outcome evaluation of critically ill patients.Crit Care.2006;10(1):R22.
- .Abnormal resting blood lactate. I. The significance of hyperlactatemia in hospitalized patients.Am J Med.1961;30:840–848.
- ,,,,.Effect of lactic acidosis on canine hemodynamics and left ventricular function.Am J Physiol.1990;258(4 Pt 2):H1193–H1199.
- ,,,.Alkali therapy extends the period of survival during hypoxia: studies in rats.Am J Physiol.1996;271(2 Pt 2):R381–R387.
- ,,.Acidosis during early reperfusion prevents myocardial stunning in perfused ferret hearts.J Clin Invest.1988;82(3):920–927.
- ,,, et al.Effect of acidotic blood reperfusion on reperfusion injury after coronary artery occlusion in the dog heart.J Cardiovasc Pharmacol.1998;31(2):179–186.
- ,,,.Hypoxia and acidosis activate cardiac myocyte death through the Bcl‐2 family protein BNIP3.Proc Natl Acad Sci U S A.2002;99(20):12825–12830.
- ,,,,,.Extracellular acidosis delays onset of cell death in ATP‐depleted hepatocytes.Am J Physiol.1988;255(3 Pt 1):C315–C322.
- ,.Metabolic and hemodynamic effects of hypertonic solutions: sodium‐lactate versus sodium chloride infusion in postoperative patients.Shock.2002;18(4):306–310.
- .Medical complications of status epilepticus.Adv Neurol.1983;34:395–398.
- ,,,,,.Bicarbonate therapy in severe diabetic ketoacidosis. A double blind, randomized, placebo controlled trial.Rev Invest Clin.1991;43(3):234–238.
- ,,.Supercarbia in children: clinical course and outcome.Crit Care Med.1990;18(2):166–168.
- ,,,.Low mortality rate in adult respiratory distress syndrome using low‐volume, pressure‐limited ventilation with permissive hypercapnia: a prospective study.Crit Care Med.1994;22(10):1568–1578.
- ,,,.Systemic effects of NaHCO3 in experimental lactic acidosis in dogs.Am J Physiol.1982;242(6):F586–F591.
- ,,,,.Lactic acidosis: effect of treatment on intracellular pH and energetics in living rat heart.Am J Physiol.1992;262(5 Pt 2):H1572–H1578.
- ,,,,,.Effect of sodium bicarbonate on intracellular pH under different buffering conditions.Kidney Int.1996;49(5):1262–1267.
- ,,.Hemodynamic and hepatic pH responses to sodium bicarbonate and Carbicarb during systemic acidosis.Magn Reson Med.1990;16(3):403–410.
- ,,, et al.The increase in CO2 production induced by NaHCO3 depends on blood albumin and hemoglobin concentrations.Intensive Care Med.2000;26(5):558–564.
- ,,, et al.Initial effect of sodium bicarbonate on intracellular pH depends on the extracellular nonbicarbonate buffering capacity.Crit Care Med.2001;29(5):1033–1039.
- ,,,,,.Dynamic study of the distribution of microcirculatory blood flow in multiple splanchnic organs in septic shock.Crit Care Med.2000;28(9):3233–3241.
- ,,,,,.The effects of sodium bicarbonate and a mixture of sodium bicarbonate and carbonate (“Carbicarb”) on skeletal muscle pH and hemodynamic status in rats with hypovolemic shock.Metabolism.1994;43(4):518–522.
- ,,, et al.Haemodynamic and metabolic effects in diabetic ketoacidosis in rats of treatment with sodium bicarbonate or a mixture of sodium bicarbonate and sodium carbonate.Diabetologia.1995;38(8):889–898.
- ,,,,,.Effects of sodium bicarbonate on striated muscle metabolism and intracellular pH during endotoxic shock.Shock.1994;1(3):196–200.
- ,,,,.Carbicarb, sodium bicarbonate, and sodium chloride in hypoxic lactic acidosis. Effect on arterial blood gases, lactate concentrations, hemodynamic variables, and myocardial intracellular pH.Chest.1993;104(3):913–918.
- ,.Improved hemodynamic function during hypoxia with Carbicarb, a new agent for the management of acidosis.Circulation.1988;77(1):227–233.
- ,,.Metabolic effects of sodium bicarbonate in hypoxic lactic acidosis in dogs.Am J Physiol.1985;249(5 Pt 2):F630–F635.
- .Binding of calcium to serum albumin. II. Effect of pH via competitive hydrogen and calcium ion binding to the imidazole groups of albumin.Scand J Clin Lab Invest.1972;29(1):75–83.
- ,,,,.Left ventricular contractility varies directly with blood ionized calcium.Ann Intern Med.1988;108(4):524–529.
- ,,.Regulatory mechanisms of hemoglobin oxygen affinity in acidosis and alkalosis.J Clin Invest.1971;50(3):700–706.
- ,,.Metabolic and hemodynamic consequences of sodium bicarbonate administration in patients with heart disease.Am J Med.1989;87(1):7–14.
- ,,.Sodium ion/hydrogen ion exchange inhibition: a new pharmacologic approach to myocardial ischemia and reperfusion injury.J Clin Pharmacol.1998;38(10):887–897.
- ,.SEA0400: a novel sodium‐calcium exchange inhibitor with cardioprotective properties.Cardiovasc Drug Rev.2004;22(4):334–347.
- ,,,.Elevation in cytosolic free calcium concentration early in myocardial ischemia in perfused rat heart.Circ Res.1987;60(5):700–707.
- ,.Acute haemodynamic effects of sodium bicarbonate administration in respiratory and metabolic acidosis in anaesthetized dogs.Anaesth Intensive Care.1997;25(6):615–620.
- ,,,.Bicarbonate does not increase left ventricular contractility during L‐lactic acidemia in pigs.Am Rev Respir Dis.1993;148(2):317–322.
- ,,,,.Effects of bicarbonate therapy on hemodynamics and tissue oxygenation in patients with lactic acidosis: a prospective, controlled clinical study.Crit Care Med.1991;19(11):1352–1356.
- ,,, et al.Safety and efficacy of intravenous Carbicarb in patients undergoing surgery: comparison with sodium bicarbonate in the treatment of mild metabolic acidosis. SPI Research Group. Study of Perioperative Ischemia.Crit Care Med.1994;22(10):1540–1549.
- ,,, et al.Sodium bicarbonate versus THAM in ICU patients with mild metabolic acidosis.J Nephrol.2005;18(3):303–307.
- ,,, et al.A controlled clinical trial of dichloroacetate for treatment of lactic acidosis in adults. The Dichloroacetate‐Lactic Acidosis Study Group.N Engl J Med.1992;327(22):1564–1569.
- ,,,,,.Effect of continuous venovenous hemofiltration with dialysis on lactate clearance in critically ill patients.Crit Care Med.1997;25(1):58–62.
- ,,,,.A pilot randomised controlled comparison of continuous veno‐venous haemofiltration and extended daily dialysis with filtration: effect on small solutes and acid‐base balance.Intensive Care Med.2007;33(5):830–835.
- ,,.Intermittent versus continuous renal replacement therapy in the ICU: impact on electrolyte and acid‐base balance.Intensive Care Med.2001;27(6):1037–1043.
- ,,,.Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus: systematic review and meta‐analysis.Arch Intern Med.2003;163(21):2594–2602.
- ,,,,.Severe acidosis in patients taking metformin—rapid reversal and survival despite high APACHE score.Diabet Med.2006;23(4):432–435.
- ,,.Incidence of lactic acidosis in metformin users.Diabetes Care.1999;22(6):925–927.
- ,.Lactic acidosis in metformin‐treated patients. Prognostic value of arterial lactate levels and plasma metformin concentrations.Drug Saf.1999;20(4):377–384.
- ,,,.The management of metformin overdose.Anaesthesia.1998;53(7):698–701.
- ,,.Lactic acidosis in biguanide‐treated diabetics: a review of 330 cases.Diabetologia.1978;14(2):75–87.
- ,,,,.Treatment of metformin‐associated lactic acidosis with closed recirculation bicarbonate‐buffered hemodialysis.Arch Intern Med.1984;144(1):203–205.
- ,,,.High anion gap metabolic acidosis in suicide: don't forget metformin intoxication—two patients' experiences.Ren Fail.2002;24(5):671–675.
- ,,, et al.Hemodialysis in the treatment of lactic acidosis in diabetics treated by metformin: a study of metformin elimination.Int J Clin Pharmacol Ther Toxicol.1989;27(6):285–288.
- ,,.Bicarbonate haemodialysis as a treatment of metformin overdose.Nephrol Dial Transplant.1997;12(5):1046–1047.
- ,,,.Combination of intermittent haemodialysis and high‐volume continuous haemofiltration for the treatment of severe metformin‐induced lactic acidosis.Nephrol Dial Transplant.2004;19(8):2157–2158.
- ,,,.When a friend can become an enemy! Recognition and management of metformin‐associated lactic acidosis.Kidney Int.2007;72(9):1157–1160.
- .,, et al.Severe nucleoside‐associated lactic acidosis in human immunodeficiency virus‐infected patients: report of 12 cases and review of the literature.Clin Infect Dis.2002;34(6):838–846.
- ,,.Clearance of metformin by hemofiltration in overdose.J Toxicol Clin Toxicol.2002;40(2):177–180.
- .Metformin‐associated lactic acidosis.J Emerg Med.2001;20(3):267–272.
- ,.Contraindications to use of metformin. Blanket banning of metformin two days before surgery may not be a good idea.BMJ.2003;326(7392):762; author reply 762.
- ,,, et al.Hepatic failure and lactic acidosis due to fialuridine (FIAU), an investigational nucleoside analogue for chronic hepatitis B.N Engl J Med.1995;333(17):1099–1105.
- ,,,.Zidovudine‐induced fatal lactic acidosis and hepatic failure in patients with acquired immunodeficiency syndrome: report of two patients and review of the literature.Crit Care Med.1997;25(8):1425–1430.
- ,.Mitochondrial toxicity of antiviral drugs.Nat Med.1995;1(5):417–422.
- ,.Mitochondrial toxicity of nucleoside analogue reverse transcriptase inhibitors: a looming obstacle for long‐term antiretroviral therapy?Curr Opin Infect Dis.2000;13(1):5–11.
- ,,, et al.Treatment for adult HIV infection: 2006 recommendations of the International AIDS Society‐USA panel.JAMA.2006;296(7):827–843.
- ,,.Riboflavin to treat nucleoside analogue‐induced lactic acidosis.Lancet.1998;352(9124):291–292.
- ,,,,,.Severe lactic acidosis and thiamine administration in an HIV‐infected patient on HAART.Int J STD AIDS.2001;12(6):407–409.
- ,,, et al.Detecting life‐threatening lactic acidosis related to nucleoside‐analog treatment of human immunodeficiency virus‐infected patients, and treatment with L‐carnitine.Crit Care Med.2003;31(4):1042–1047.
- ,.Hyperosmolar metabolic acidosis and intravenous Lorazepam.N Engl J Med.2002;347(11):857–858; author reply 857–858.
- ,,,.Propylene glycol toxicity: a severe iatrogenic illness in ICU patients receiving IV benzodiazepines: a case series and prospective, observational pilot study.Chest.2005;128(3):1674–1681.
- ,,, et al.Propylene glycol pharmacokinetics and effects after intravenous infusion in humans.Ther Drug Monit.1987;9(3):255–258.
- .Short‐term lorazepam infusion and concern for propylene glycol toxicity: case report and review.Pharmacotherapy.2001;21(9):1140–1144.
- ,,.Recognition, treatment, and prevention of propylene glycol toxicity.Semin Dial.2007;20(3):217–219.
- ,,.Propylene glycol‐mediated cell injury in a primary culture of human proximal tubule cells.Toxicol Sci.1998;46(2):410–417.
- ,.Osmolar gap metabolic acidosis in a 60‐year‐old man treated for hypoxemic respiratory failure.Chest.2000;118(2):545–546.
- ,,,.Relationship of continuous infusion lorazepam to serum propylene glycol concentration in critically ill adults.Crit Care Med.2004;32(8):1709–1714.
- ,,,.Removal of propylene glycol and correction of increased osmolar gap by hemodialysis in a patient on high dose lorazepam infusion therapy.Intensive Care Med.2002;28(1):81–84.
- ,,.Linezolid use associated with lactic acidosis.Scand J Infect Dis.2005;37(2):153–154.
- ,.Linezolid‐induced lactic acidosis.N Engl J Med.2003;348(1):86–87.
- ,,.Toxicity of extended courses of linezolid: results of an Infectious Diseases Society of America Emerging Infections Network survey.Diagn Microbiol Infect Dis.2008;62(4):407–410.
- ,,.Mitochondrial toxicity associated with linezolid.N Engl J Med.2005;353(21):2305–2306.
- ,,, et al.Linezolid‐induced inhibition of mitochondrial protein synthesis.Clin Infect Dis.2006;42(8):1111–1117.
- ,.Use of base in the treatment of severe acidemic states.Am J Kidney Dis.2001;38(4):703–727.
- ,,,.Bicarbonate does not improve hemodynamics in critically ill patients who have lactic acidosis. A prospective, controlled clinical study.Ann Intern Med.1990;112(7):492–498.
- ,,, et al.Natural history and course of acquired lactic acidosis in adults. DCA‐Lactic Acidosis Study Group.Am J Med.1994;97(1):47–54.
- ,.Sodium bicarbonate for the treatment of lactic acidosis.Chest.2000;117(1):260–267.
- ,.Management of life‐threatening acid‐base disorders. First of two parts.N Engl J Med.1998;338(1):26–34.
- .Indications for use of bicarbonate in patients with metabolic acidosis.Br J Anaesth.1991;67(2):165–177.
- ,.Bench‐to‐bedside review: treating acid‐base abnormalities in the intensive care unit—the role of buffers.Crit Care.2004;8(4):259–265.
- .Lactic acidosis update for critical care clinicians.J Am Soc Nephrol.2001;12(suppl 17):S15–S19.
- ,.Lactic acidosis: diagnosis and treatment.Clin Endocrinol Metab.1980;9(3):513–541.
- .Lactate and shock state: the metabolic view.Curr Opin Crit Care.2006;12(4):315–321.
- .Bench‐to‐bedside review: lactate and the kidney.Crit Care.2002;6(4):322–326.
- ,,,.The influence of renal function on lactate and glucose metabolism.Biochem J.1984;219(1):73–78.
- ,.The effect of acidosis on lactate removal by the perfused rat kidney.Clin Sci Mol Med.1976;50(3):185–194.
- ,,.Transvisceral lactate fluxes during early endotoxemia.Chest.1996;110(1):198–204.
- ,,,,.Metabolism of lactate by the intact functioning kidney of the dog.Am J Physiol.1973;224(6):1463–1467.
- ,,, et al.Lactate and glucose metabolism in severe sepsis and cardiogenic shock.Crit Care Med.2005;33(10):2235–2240.
- ,,,, et al.Lactic acidosis in fulminant hepatic failure. Some aspects of pathogenesis and prognosis.J Hepatol.1985;1(4):405–416.
- ,.Lactic acidosis in critical illness.Crit Care Med.1992;20(1):80–93.
- ,,, et al.Effects of cardiogenic shock on lactate and glucose metabolism after heart surgery.Crit Care Med.2000;28(12):3784–3791.
- ,,,,,.Evolution of lactate/pyruvate and arterial ketone body ratios in the early course of catecholamine‐treated septic shock.Crit Care Med.2000;28(1):114–119.
- ,,, et al.Oxygen debt and metabolic acidemia as quantitative predictors of mortality and the severity of the ischemic insult in hemorrhagic shock.Crit Care Med.1991;19(2):231–243.
- ,,, et al.Early goal‐directed therapy in the treatment of severe sepsis and septic shock.N Engl J Med.2001;345(19):1368–1377.
- ,,,.Skeletal muscle partial pressure of oxygen in patients with sepsis.Crit Care Med.1994;22(4):640–650.
- ,.Reevaluation of the role of cellular hypoxia and bioenergetic failure in sepsis.JAMA.1992;267(11):1503–1510.
- ,,,.Lactic acidosis during sepsis is related to increased pyruvate production, not deficits in tissue oxygen availability.Ann Surg.1996;224(1):97–102.
- ,,,.Biguanide‐associated lactic acidosis. Case report and review of the literature.Arch Intern Med.1992;152(11):2333–2336.
- ,,,.Bench‐to‐bedside review: severe lactic acidosis in HIV patients treated with nucleoside analogue reverse transcriptase inhibitors.Crit Care.2003;7(3):226–232.
- ,,,.Lactic acidosis after treatment with linezolid.Infection.2007;35(4):278–281.
- ,,,.Isotopic evaluation of the metabolism of pyruvate and related substrates in normal adult volunteers and severely burned children: effect of dichloroacetate and glucose infusion.Surgery.1991;110(1):54–67.
- .Alterations in carbohydrate metabolism during stress: a review of the literature.Am J Med.1995;98(1):75–84.
- .,,,.Lactic acid kinetics in respiratory alkalosis.Crit Care Med.1991;19(9):1120–1124.
- .Significance of hyperlactatemia without acidosis during hypermetabolic stress.Crit Care Med.1997;25(11):1780–1781.
- ,,,.Lactate is an unreliable indicator of tissue hypoxia in injury or sepsis.Lancet.1999;354(9177):505–508.
- ,,,,.Relation between muscle Na+K+ ATPase activity and raised lactate concentrations in septic shock: a prospective study.Lancet.2005;365(9462):871–875.
- ,.Sepsis induces diaphragm electron transport chain dysfunction and protein depletion.Am J Respir Crit Care Med.2005;172(7):861–868.
- ,,, et al.Association between mitochondrial dysfunction and severity and outcome of septic shock.Lancet.2002;360(9328):219–223.
- ,.Mitochondrial dysfunction in sepsis.Biochem Soc Symp.1999;66:149–166.
- ,,, et al.Altered glucose transporter mRNA abundance in a rat model of endotoxic shock.Biochem Biophys Res Commun.1991;176(1):535–540.
- .Increased pyruvate dehydrogenase kinase activity in response to sepsis.Am J Physiol.1991;260(5 Pt 1):E669–E674.
- .Lactate metabolism: a new paradigm for the third millennium.J Physiol.2004;558(Pt 1):5–30.
- ,,, et al.Evidence supporting the existence of an activity‐dependent astrocyte‐neuron lactate shuttle.Dev Neurosci.1998;20(4–5):291–299.
- ,,,.Brain lactate is an obligatory aerobic energy substrate for functional recovery after hypoxia: further in vitro validation.J Neurochem.1997;69(1):423–426.
- .Bench‐to‐bedside review: a possible resolution of the glucose paradox of cerebral ischemia.Crit Care.2002;6(4):330–334.
- ,,,,,.Intravenous lactate prevents cerebral dysfunction during hypoglycaemia in insulin‐dependent diabetes mellitus.Clin Sci (Lond).1998;94(2):157–163.
- ,,,.Brain lactate, not glucose, fuels the recovery of synaptic function from hypoxia upon reoxygenation: an in vitro study.Brain Res.1997;744(1):105–111.
- ,,,,,.Clinical prognostic markers in patients with severe sepsis: a prospective analysis of 139 consecutive cases.J Infect.2003;47(4):300–306.
- ,,, et al.Serum lactate as a predictor of mortality in patients with infection.Intensive Care Med.2007;33(6):970–977.
- ,,,.Lactate versus non‐lactate metabolic acidosis: a retrospective outcome evaluation of critically ill patients.Crit Care.2006;10(1):R22.
- .Abnormal resting blood lactate. I. The significance of hyperlactatemia in hospitalized patients.Am J Med.1961;30:840–848.
- ,,,,.Effect of lactic acidosis on canine hemodynamics and left ventricular function.Am J Physiol.1990;258(4 Pt 2):H1193–H1199.
- ,,,.Alkali therapy extends the period of survival during hypoxia: studies in rats.Am J Physiol.1996;271(2 Pt 2):R381–R387.
- ,,.Acidosis during early reperfusion prevents myocardial stunning in perfused ferret hearts.J Clin Invest.1988;82(3):920–927.
- ,,, et al.Effect of acidotic blood reperfusion on reperfusion injury after coronary artery occlusion in the dog heart.J Cardiovasc Pharmacol.1998;31(2):179–186.
- ,,,.Hypoxia and acidosis activate cardiac myocyte death through the Bcl‐2 family protein BNIP3.Proc Natl Acad Sci U S A.2002;99(20):12825–12830.
- ,,,,,.Extracellular acidosis delays onset of cell death in ATP‐depleted hepatocytes.Am J Physiol.1988;255(3 Pt 1):C315–C322.
- ,.Metabolic and hemodynamic effects of hypertonic solutions: sodium‐lactate versus sodium chloride infusion in postoperative patients.Shock.2002;18(4):306–310.
- .Medical complications of status epilepticus.Adv Neurol.1983;34:395–398.
- ,,,,,.Bicarbonate therapy in severe diabetic ketoacidosis. A double blind, randomized, placebo controlled trial.Rev Invest Clin.1991;43(3):234–238.
- ,,.Supercarbia in children: clinical course and outcome.Crit Care Med.1990;18(2):166–168.
- ,,,.Low mortality rate in adult respiratory distress syndrome using low‐volume, pressure‐limited ventilation with permissive hypercapnia: a prospective study.Crit Care Med.1994;22(10):1568–1578.
- ,,,.Systemic effects of NaHCO3 in experimental lactic acidosis in dogs.Am J Physiol.1982;242(6):F586–F591.
- ,,,,.Lactic acidosis: effect of treatment on intracellular pH and energetics in living rat heart.Am J Physiol.1992;262(5 Pt 2):H1572–H1578.
- ,,,,,.Effect of sodium bicarbonate on intracellular pH under different buffering conditions.Kidney Int.1996;49(5):1262–1267.
- ,,.Hemodynamic and hepatic pH responses to sodium bicarbonate and Carbicarb during systemic acidosis.Magn Reson Med.1990;16(3):403–410.
- ,,, et al.The increase in CO2 production induced by NaHCO3 depends on blood albumin and hemoglobin concentrations.Intensive Care Med.2000;26(5):558–564.
- ,,, et al.Initial effect of sodium bicarbonate on intracellular pH depends on the extracellular nonbicarbonate buffering capacity.Crit Care Med.2001;29(5):1033–1039.
- ,,,,,.Dynamic study of the distribution of microcirculatory blood flow in multiple splanchnic organs in septic shock.Crit Care Med.2000;28(9):3233–3241.
- ,,,,,.The effects of sodium bicarbonate and a mixture of sodium bicarbonate and carbonate (“Carbicarb”) on skeletal muscle pH and hemodynamic status in rats with hypovolemic shock.Metabolism.1994;43(4):518–522.
- ,,, et al.Haemodynamic and metabolic effects in diabetic ketoacidosis in rats of treatment with sodium bicarbonate or a mixture of sodium bicarbonate and sodium carbonate.Diabetologia.1995;38(8):889–898.
- ,,,,,.Effects of sodium bicarbonate on striated muscle metabolism and intracellular pH during endotoxic shock.Shock.1994;1(3):196–200.
- ,,,,.Carbicarb, sodium bicarbonate, and sodium chloride in hypoxic lactic acidosis. Effect on arterial blood gases, lactate concentrations, hemodynamic variables, and myocardial intracellular pH.Chest.1993;104(3):913–918.
- ,.Improved hemodynamic function during hypoxia with Carbicarb, a new agent for the management of acidosis.Circulation.1988;77(1):227–233.
- ,,.Metabolic effects of sodium bicarbonate in hypoxic lactic acidosis in dogs.Am J Physiol.1985;249(5 Pt 2):F630–F635.
- .Binding of calcium to serum albumin. II. Effect of pH via competitive hydrogen and calcium ion binding to the imidazole groups of albumin.Scand J Clin Lab Invest.1972;29(1):75–83.
- ,,,,.Left ventricular contractility varies directly with blood ionized calcium.Ann Intern Med.1988;108(4):524–529.
- ,,.Regulatory mechanisms of hemoglobin oxygen affinity in acidosis and alkalosis.J Clin Invest.1971;50(3):700–706.
- ,,.Metabolic and hemodynamic consequences of sodium bicarbonate administration in patients with heart disease.Am J Med.1989;87(1):7–14.
- ,,.Sodium ion/hydrogen ion exchange inhibition: a new pharmacologic approach to myocardial ischemia and reperfusion injury.J Clin Pharmacol.1998;38(10):887–897.
- ,.SEA0400: a novel sodium‐calcium exchange inhibitor with cardioprotective properties.Cardiovasc Drug Rev.2004;22(4):334–347.
- ,,,.Elevation in cytosolic free calcium concentration early in myocardial ischemia in perfused rat heart.Circ Res.1987;60(5):700–707.
- ,.Acute haemodynamic effects of sodium bicarbonate administration in respiratory and metabolic acidosis in anaesthetized dogs.Anaesth Intensive Care.1997;25(6):615–620.
- ,,,.Bicarbonate does not increase left ventricular contractility during L‐lactic acidemia in pigs.Am Rev Respir Dis.1993;148(2):317–322.
- ,,,,.Effects of bicarbonate therapy on hemodynamics and tissue oxygenation in patients with lactic acidosis: a prospective, controlled clinical study.Crit Care Med.1991;19(11):1352–1356.
- ,,, et al.Safety and efficacy of intravenous Carbicarb in patients undergoing surgery: comparison with sodium bicarbonate in the treatment of mild metabolic acidosis. SPI Research Group. Study of Perioperative Ischemia.Crit Care Med.1994;22(10):1540–1549.
- ,,, et al.Sodium bicarbonate versus THAM in ICU patients with mild metabolic acidosis.J Nephrol.2005;18(3):303–307.
- ,,, et al.A controlled clinical trial of dichloroacetate for treatment of lactic acidosis in adults. The Dichloroacetate‐Lactic Acidosis Study Group.N Engl J Med.1992;327(22):1564–1569.
- ,,,,,.Effect of continuous venovenous hemofiltration with dialysis on lactate clearance in critically ill patients.Crit Care Med.1997;25(1):58–62.
- ,,,,.A pilot randomised controlled comparison of continuous veno‐venous haemofiltration and extended daily dialysis with filtration: effect on small solutes and acid‐base balance.Intensive Care Med.2007;33(5):830–835.
- ,,.Intermittent versus continuous renal replacement therapy in the ICU: impact on electrolyte and acid‐base balance.Intensive Care Med.2001;27(6):1037–1043.
- ,,,.Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus: systematic review and meta‐analysis.Arch Intern Med.2003;163(21):2594–2602.
- ,,,,.Severe acidosis in patients taking metformin—rapid reversal and survival despite high APACHE score.Diabet Med.2006;23(4):432–435.
- ,,.Incidence of lactic acidosis in metformin users.Diabetes Care.1999;22(6):925–927.
- ,.Lactic acidosis in metformin‐treated patients. Prognostic value of arterial lactate levels and plasma metformin concentrations.Drug Saf.1999;20(4):377–384.
- ,,,.The management of metformin overdose.Anaesthesia.1998;53(7):698–701.
- ,,.Lactic acidosis in biguanide‐treated diabetics: a review of 330 cases.Diabetologia.1978;14(2):75–87.
- ,,,,.Treatment of metformin‐associated lactic acidosis with closed recirculation bicarbonate‐buffered hemodialysis.Arch Intern Med.1984;144(1):203–205.
- ,,,.High anion gap metabolic acidosis in suicide: don't forget metformin intoxication—two patients' experiences.Ren Fail.2002;24(5):671–675.
- ,,, et al.Hemodialysis in the treatment of lactic acidosis in diabetics treated by metformin: a study of metformin elimination.Int J Clin Pharmacol Ther Toxicol.1989;27(6):285–288.
- ,,.Bicarbonate haemodialysis as a treatment of metformin overdose.Nephrol Dial Transplant.1997;12(5):1046–1047.
- ,,,.Combination of intermittent haemodialysis and high‐volume continuous haemofiltration for the treatment of severe metformin‐induced lactic acidosis.Nephrol Dial Transplant.2004;19(8):2157–2158.
- ,,,.When a friend can become an enemy! Recognition and management of metformin‐associated lactic acidosis.Kidney Int.2007;72(9):1157–1160.
- .,, et al.Severe nucleoside‐associated lactic acidosis in human immunodeficiency virus‐infected patients: report of 12 cases and review of the literature.Clin Infect Dis.2002;34(6):838–846.
- ,,.Clearance of metformin by hemofiltration in overdose.J Toxicol Clin Toxicol.2002;40(2):177–180.
- .Metformin‐associated lactic acidosis.J Emerg Med.2001;20(3):267–272.
- ,.Contraindications to use of metformin. Blanket banning of metformin two days before surgery may not be a good idea.BMJ.2003;326(7392):762; author reply 762.
- ,,, et al.Hepatic failure and lactic acidosis due to fialuridine (FIAU), an investigational nucleoside analogue for chronic hepatitis B.N Engl J Med.1995;333(17):1099–1105.
- ,,,.Zidovudine‐induced fatal lactic acidosis and hepatic failure in patients with acquired immunodeficiency syndrome: report of two patients and review of the literature.Crit Care Med.1997;25(8):1425–1430.
- ,.Mitochondrial toxicity of antiviral drugs.Nat Med.1995;1(5):417–422.
- ,.Mitochondrial toxicity of nucleoside analogue reverse transcriptase inhibitors: a looming obstacle for long‐term antiretroviral therapy?Curr Opin Infect Dis.2000;13(1):5–11.
- ,,, et al.Treatment for adult HIV infection: 2006 recommendations of the International AIDS Society‐USA panel.JAMA.2006;296(7):827–843.
- ,,.Riboflavin to treat nucleoside analogue‐induced lactic acidosis.Lancet.1998;352(9124):291–292.
- ,,,,,.Severe lactic acidosis and thiamine administration in an HIV‐infected patient on HAART.Int J STD AIDS.2001;12(6):407–409.
- ,,, et al.Detecting life‐threatening lactic acidosis related to nucleoside‐analog treatment of human immunodeficiency virus‐infected patients, and treatment with L‐carnitine.Crit Care Med.2003;31(4):1042–1047.
- ,.Hyperosmolar metabolic acidosis and intravenous Lorazepam.N Engl J Med.2002;347(11):857–858; author reply 857–858.
- ,,,.Propylene glycol toxicity: a severe iatrogenic illness in ICU patients receiving IV benzodiazepines: a case series and prospective, observational pilot study.Chest.2005;128(3):1674–1681.
- ,,, et al.Propylene glycol pharmacokinetics and effects after intravenous infusion in humans.Ther Drug Monit.1987;9(3):255–258.
- .Short‐term lorazepam infusion and concern for propylene glycol toxicity: case report and review.Pharmacotherapy.2001;21(9):1140–1144.
- ,,.Recognition, treatment, and prevention of propylene glycol toxicity.Semin Dial.2007;20(3):217–219.
- ,,.Propylene glycol‐mediated cell injury in a primary culture of human proximal tubule cells.Toxicol Sci.1998;46(2):410–417.
- ,.Osmolar gap metabolic acidosis in a 60‐year‐old man treated for hypoxemic respiratory failure.Chest.2000;118(2):545–546.
- ,,,.Relationship of continuous infusion lorazepam to serum propylene glycol concentration in critically ill adults.Crit Care Med.2004;32(8):1709–1714.
- ,,,.Removal of propylene glycol and correction of increased osmolar gap by hemodialysis in a patient on high dose lorazepam infusion therapy.Intensive Care Med.2002;28(1):81–84.
- ,,.Linezolid use associated with lactic acidosis.Scand J Infect Dis.2005;37(2):153–154.
- ,.Linezolid‐induced lactic acidosis.N Engl J Med.2003;348(1):86–87.
- ,,.Toxicity of extended courses of linezolid: results of an Infectious Diseases Society of America Emerging Infections Network survey.Diagn Microbiol Infect Dis.2008;62(4):407–410.
- ,,.Mitochondrial toxicity associated with linezolid.N Engl J Med.2005;353(21):2305–2306.
- ,,, et al.Linezolid‐induced inhibition of mitochondrial protein synthesis.Clin Infect Dis.2006;42(8):1111–1117.
- ,.Use of base in the treatment of severe acidemic states.Am J Kidney Dis.2001;38(4):703–727.
The New Vocabulary of Healthcare Reform
On March 21, 2010, the United States Congress passed the most comprehensive healthcare reform bill since the formation of Medicare. The legislation's greatest impact will be to improve access for nearly 50 million Americans who are presently uninsured. Yet the bill does little to tackle the fundamental problems of the payment and delivery systemsproblems that have resulted in major quality gaps, large numbers of medical errors, fragmented care, and backbreaking costs.
While these tough questions were mostly kicked down the road, the debate did bring many of the key questions and potential solutions into high relief. Our political leaders, pundits, and health policy scholars introduced or popularized a number of terms during the healthcare debates of 2009‐2010 (Table 1). I will attempt to place them in context and discuss their implications for future healthcare reform efforts.
|
| Value‐based purchasing |
| Bending the cost curve |
| Comparative effectiveness research (see also NICE) |
| Dartmouth atlas (see also McAllen, Texas) |
| Death panels (see also rationing) |
| Bundled payments |
| Accountable care organizations (see also Mayo Clinic, Cleveland Clinic, Geisinger; replaces HMOs) |
Some Context for the Healthcare Reform Debate
In our capitalistic economy, we make most purchases based on considerations of value: quality divided by cost. There are few among us wealthy enough to always buy the best product, or cheap enough to always buy the least expensive. Instead, we try to determine value when we purchase a restaurant meal, a house, or a vacation.
Healthcare has traditionally been the major exception to this rule, both because healthcare insurance has partly insulated consumers (patients or their proxies) from the cost consequences of their decisions, and because it is so difficult to determine the quality of healthcare. But, over the past 10 to 15 years, problems with both the numerator and denominator of this equation have created widespread recognition of the need for change.
In the numerator, we now appreciate that there are nearly 100,000 deaths per year from medical mistakes;1 that we deliver evidence‐based care only about half the time,2 and that our healthcare system is extraordinarily fragmented and chaotic. We also know that there are more than 40 million people without healthcare insurance, a uniquely American problem, since other industrialized countries manage to guarantee coverage.
This is the fundamental conundrum that needs to be addressed by healthcare reform: we have a system that produces surprisingly low‐quality, unreliable care at an exorbitant and ever‐increasing cost, and does so while leaving more than 1 out of 8 citizens without coverage. Although government is a large payer (through Medicare, Medicaid, the Veterans Affairs [VA] programs and others), most Americans receive healthcare coverage as an employee benefit; a smaller number pay for health insurance themselves. The government has a key role even in these nongovernment‐sponsored payment systems, by providing tax breaks for healthcare coverage, creating a regulatory framework, and often defining the market through its actions in its public programs.
The end result is that all the involved partiesgovernments, businesses, providers, and patientsare crying out for change. An observer of this situation feels compelled to invoke the popular version of Stein's Law: if a trend can't continue, it won't.
Bending the Cost Curve
Everyone is now familiar with the scary trends (such as in Figure 1) demonstrating the unsustainable rate of healthcare inflation in the US, trends that are projected to lead to the insolvency of the Medicare Trust Fund within a decade. The term bending the cost curve implies that our solvency depends not on lowering total costs (a political impossibility), but rather on simply decreasing the rate of rise. There are only so many ways to do this.
The most attractive, of course, is to stop providing expensive care that adds no or little value in terms of patient outcomes. The term comparative effectiveness research (CER) emerged over the past few years to describe research that pits one approach against another (or, presumably, against no treatment) on both outcomes and cost.3 Obviously, one would favor the less expensive treatment if the efficacy were equal. However, the more common (and politically fraught) question is whether a more expensive but slightly better approach is worth its additional cost.
This, of course, makes complete sense in a world of limited resources, and some countries, mostly notably the United Kingdom, are using CER to inform healthcare coverage decisions. In the United Kingdom, the research is analyzed, and coverage recommendations made, by an organization called the National Institute for Health and Clinical Excellence (NICE).4 While NICE appears to be working well, all signs indicate that the US political system is not ready for such an approach. In fact, although Medicare generally supports CER, most of the healthcare reform proposals considered by Congress explicitly prohibited Medicare from using CER results to influence payment decisions.
If an overall CER approach is too politically difficult for the US, how about focusing on 1 small segment of healthcare: expensive care at the end of life? Over the past 30 years, a group of Dartmouth researchers has examined the costs and quality of care across the entire country, demonstrating a ubiquitous pattern of highly variable costs (varying up to 2‐fold) that is unassociated with quality and outcomes (and sometimes even inversely associated).5 The findings, well known among healthcare researchers but relatively unknown by the public until recently, were brought to public attention by a 2009 New Yorker article that made the border town of McAllen, Texas the poster child for a medical culture that produces high costs without comparable benefits.6 The Dartmouth researchers, who publish their data in a document known as The Dartmouth Atlas, have found striking variations in care at the end of life. For example, even among academic medical centers (which presumably have similarly sick patient populations), the number of hospital days in the last 6 months of life varies strikingly: patients at New York University average 27.1 days, whereas those in my hospital average 11.5 days.7
So promoting better end‐of‐life carebeing sure that patients are aware of their options and that high‐quality palliative care is availableseemed like an obvious solution to part of our cost‐quality conundrum. Some early drafts of reform bills in Congress contained provisions to pay for physicians' time to discuss end‐of‐life options. This, of course, was caricaturized into the now famous Death Panelsproving that American political discourse is not yet mature enough to support realistic discussions about difficult subjects.8
It seems like having payers (government, insurance companies) make formal decisions about which services to cover (ie, rationing) is too hard. Is there another way to force these tough choices but do so without creating a political piata?
Encircling a Population
Rather than explicitly rationing care (using CER results, for example), another way of constraining costs is to place a population of patients on a fixed budget. There is evidence that provider organizations, working within such a budget (structured in a way that permits the providers to pocket any savings), are able to reorganize and change their practice style in a way that can cut costs.9 In the 1990s, we conducted a national experiment by promoting managed care, working through integrated delivery systems called Health Maintenance Organizations (HMOs) that received fixed, capitated payments for every patient. And, in fact, these organizations did cut overall costs.
The problem was that patients neither liked HMOs nor trusted that they were acting in their best interests. Ultimately, managed care became a less important delivery mechanism, and even patients who remained in HMOs had fewer constraints on their choices. Of course, the softening of the managed care market resulted in an uptick in healthcare inflation, contributing to our present predicament.
The concept of fixed payments has resurfaced, but with some modern twists. It appears that organizations that perform best on the Dartmouth measures (namely, they provide high quality care at lower costs) are generally large delivery systems with advanced information technology, strong primary care infrastructures, andprobably most importantlytight integration between physicians and the rest of the organization. During the healthcare debate, the organizations that received the most attention were the Mayo and Cleveland Clinics and the Geisinger system in central Pennsylvania. The problem is that the defining characteristic of these organizations (and others like them) is that they have been at this business of integrated care for more than 50 years! Can the model be emulated?
Two main policy changes have been promoted to try to achieve this integration: one is a change in payment structure, the other a change in organization. The first is known as bundlingin which multiple providers are reimbursed a single sum for all the care related to an episode of illness (such as a hospitalization and a 60‐ or 90‐day period afterwards). You will recognize this as a new form of capitation, but, rather than covering all of a patient's care, a more circumscribed version, focusing on a single illness or procedure. There is some evidence that bundling does reduce costs and may improve quality, by forcing hospitals, post‐acute care facilities, and doctors into collaborative arrangements (both to deliver care and, just as complex, to split the single payment without undue acrimony).10 Fisher et al.11 have promoted a new structure to deliver this kind of bundled care more effectively: The Accountable Care Organization (ACO), which is best thought of as a less ambitious, and potentially more virtual, incarnation of the HMO.
Interestingly, while many healthcare organizations have struggled to remake themselves in Mayo's image in preparation for upcoming pressures to form ACOs, some organizations with hospitalist programs need look no further than these programs to chart a course toward more effective physician‐hospital integration.12 Why? The majority of US hospitals now have hospitalists, and virtually all hospitalist programs receive support payments from their hospital (a sizable minority are on salary from the hospital). Hospitalists recognize that part of their value equation (which justifies the hospital support dollars) is that they help the hospital deliver higher quality care more efficiently. Because of this relationship, a well‐functioning hospitalist program can assume many of the attributes of an ACO, even in organizations with otherwise challenging physician‐hospital relations. It may be that hospitals and doctors need not look to Rochester, Minnesota or Danville, Pennsylvania for positive examples of physician‐hospital integration, but simply to their own local hospitalist groups.
The Bottom Line
While proponents of the Obama reform plan celebrate its passage, virtually all experts agree that it left fundamental problems with the healthcare system unaddressed. Although the 20092010 debate did not solve these problems, the new vocabulary introduced during the debateboth reasonable policy ideas like bundling and ACOs and cynical caricatures like death panelsare here to stay. Understanding these terms and the context that shaped them will be critical for hospitalists and other stakeholders interested in the future of the American healthcare system.
- ,,, eds.To Err is Human: Building a Safer Health System.Washington DC:Committee on Quality of Health Care in America, Institute of Medicine. National Academy Press,2000.
- ,,, et al.The quality of health care delivered to adults in the United States.N Engl J Med.2003;348:2635–2645.
- ,.Health care reform and the need for comparative‐effectiveness research.N Engl J Med.2010;362:e6.
- .Saying no isn't NICE—the travails of Britain's National Institute for health and clinical excellence.N Engl J Med.2008;359:1977–1981.
- .Wrestling with variation: an interview with Jack Wennberg [interviewed by Fitzhugh Mullan].Health Aff (Millwood).2004; Suppl Web Exclsives:VAR73–80.
- .The cost conundrum. What a Texas town can teach us about health care.The New Yorker2009. Available at: http://www.newyorker. com/reporting/2009/06/01/090601fa_fact_gawande. Accessed February 2010.
- .Researchers find huge variations in end‐of‐life treatment.New York Times.2008. Available at: http://www.nytimes.com/2008/04/07/health/policy/07care.html?_r=1. Accessed February 2010.
- .Ending end‐of‐life phobia—a prescription for enlightened health care reform.N Engl J Med.2009. Available at: http://healthcarere form.nejm.org/?p=2580. Accessed February 2010.
- ,.The RAND Health Insurance Experiment and HMOs.Med Care.1990;28:191–200.
- ,,.Cost savings and physician responses to global bundled payments for Medicare heart bypass surgery.Health Care Fin Rev.1997;19:41–57.
- ,,,.Creating accountable care organizations: the extended hospital medical staff.Health Aff (Millwood).2007;26(1):w44–w57.
- . Hospitalists: a little slice of Mayo. Available at: http://community.the‐hospitalist.org/blogs/wachters_world/archive/2009/08/30/hospitalists‐a‐little‐slice‐of‐mayo.aspx. Accessed February 2010.
On March 21, 2010, the United States Congress passed the most comprehensive healthcare reform bill since the formation of Medicare. The legislation's greatest impact will be to improve access for nearly 50 million Americans who are presently uninsured. Yet the bill does little to tackle the fundamental problems of the payment and delivery systemsproblems that have resulted in major quality gaps, large numbers of medical errors, fragmented care, and backbreaking costs.
While these tough questions were mostly kicked down the road, the debate did bring many of the key questions and potential solutions into high relief. Our political leaders, pundits, and health policy scholars introduced or popularized a number of terms during the healthcare debates of 2009‐2010 (Table 1). I will attempt to place them in context and discuss their implications for future healthcare reform efforts.
|
| Value‐based purchasing |
| Bending the cost curve |
| Comparative effectiveness research (see also NICE) |
| Dartmouth atlas (see also McAllen, Texas) |
| Death panels (see also rationing) |
| Bundled payments |
| Accountable care organizations (see also Mayo Clinic, Cleveland Clinic, Geisinger; replaces HMOs) |
Some Context for the Healthcare Reform Debate
In our capitalistic economy, we make most purchases based on considerations of value: quality divided by cost. There are few among us wealthy enough to always buy the best product, or cheap enough to always buy the least expensive. Instead, we try to determine value when we purchase a restaurant meal, a house, or a vacation.
Healthcare has traditionally been the major exception to this rule, both because healthcare insurance has partly insulated consumers (patients or their proxies) from the cost consequences of their decisions, and because it is so difficult to determine the quality of healthcare. But, over the past 10 to 15 years, problems with both the numerator and denominator of this equation have created widespread recognition of the need for change.
In the numerator, we now appreciate that there are nearly 100,000 deaths per year from medical mistakes;1 that we deliver evidence‐based care only about half the time,2 and that our healthcare system is extraordinarily fragmented and chaotic. We also know that there are more than 40 million people without healthcare insurance, a uniquely American problem, since other industrialized countries manage to guarantee coverage.
This is the fundamental conundrum that needs to be addressed by healthcare reform: we have a system that produces surprisingly low‐quality, unreliable care at an exorbitant and ever‐increasing cost, and does so while leaving more than 1 out of 8 citizens without coverage. Although government is a large payer (through Medicare, Medicaid, the Veterans Affairs [VA] programs and others), most Americans receive healthcare coverage as an employee benefit; a smaller number pay for health insurance themselves. The government has a key role even in these nongovernment‐sponsored payment systems, by providing tax breaks for healthcare coverage, creating a regulatory framework, and often defining the market through its actions in its public programs.
The end result is that all the involved partiesgovernments, businesses, providers, and patientsare crying out for change. An observer of this situation feels compelled to invoke the popular version of Stein's Law: if a trend can't continue, it won't.
Bending the Cost Curve
Everyone is now familiar with the scary trends (such as in Figure 1) demonstrating the unsustainable rate of healthcare inflation in the US, trends that are projected to lead to the insolvency of the Medicare Trust Fund within a decade. The term bending the cost curve implies that our solvency depends not on lowering total costs (a political impossibility), but rather on simply decreasing the rate of rise. There are only so many ways to do this.

The most attractive, of course, is to stop providing expensive care that adds no or little value in terms of patient outcomes. The term comparative effectiveness research (CER) emerged over the past few years to describe research that pits one approach against another (or, presumably, against no treatment) on both outcomes and cost.3 Obviously, one would favor the less expensive treatment if the efficacy were equal. However, the more common (and politically fraught) question is whether a more expensive but slightly better approach is worth its additional cost.
This, of course, makes complete sense in a world of limited resources, and some countries, mostly notably the United Kingdom, are using CER to inform healthcare coverage decisions. In the United Kingdom, the research is analyzed, and coverage recommendations made, by an organization called the National Institute for Health and Clinical Excellence (NICE).4 While NICE appears to be working well, all signs indicate that the US political system is not ready for such an approach. In fact, although Medicare generally supports CER, most of the healthcare reform proposals considered by Congress explicitly prohibited Medicare from using CER results to influence payment decisions.
If an overall CER approach is too politically difficult for the US, how about focusing on 1 small segment of healthcare: expensive care at the end of life? Over the past 30 years, a group of Dartmouth researchers has examined the costs and quality of care across the entire country, demonstrating a ubiquitous pattern of highly variable costs (varying up to 2‐fold) that is unassociated with quality and outcomes (and sometimes even inversely associated).5 The findings, well known among healthcare researchers but relatively unknown by the public until recently, were brought to public attention by a 2009 New Yorker article that made the border town of McAllen, Texas the poster child for a medical culture that produces high costs without comparable benefits.6 The Dartmouth researchers, who publish their data in a document known as The Dartmouth Atlas, have found striking variations in care at the end of life. For example, even among academic medical centers (which presumably have similarly sick patient populations), the number of hospital days in the last 6 months of life varies strikingly: patients at New York University average 27.1 days, whereas those in my hospital average 11.5 days.7
So promoting better end‐of‐life carebeing sure that patients are aware of their options and that high‐quality palliative care is availableseemed like an obvious solution to part of our cost‐quality conundrum. Some early drafts of reform bills in Congress contained provisions to pay for physicians' time to discuss end‐of‐life options. This, of course, was caricaturized into the now famous Death Panelsproving that American political discourse is not yet mature enough to support realistic discussions about difficult subjects.8
It seems like having payers (government, insurance companies) make formal decisions about which services to cover (ie, rationing) is too hard. Is there another way to force these tough choices but do so without creating a political piata?
Encircling a Population
Rather than explicitly rationing care (using CER results, for example), another way of constraining costs is to place a population of patients on a fixed budget. There is evidence that provider organizations, working within such a budget (structured in a way that permits the providers to pocket any savings), are able to reorganize and change their practice style in a way that can cut costs.9 In the 1990s, we conducted a national experiment by promoting managed care, working through integrated delivery systems called Health Maintenance Organizations (HMOs) that received fixed, capitated payments for every patient. And, in fact, these organizations did cut overall costs.
The problem was that patients neither liked HMOs nor trusted that they were acting in their best interests. Ultimately, managed care became a less important delivery mechanism, and even patients who remained in HMOs had fewer constraints on their choices. Of course, the softening of the managed care market resulted in an uptick in healthcare inflation, contributing to our present predicament.
The concept of fixed payments has resurfaced, but with some modern twists. It appears that organizations that perform best on the Dartmouth measures (namely, they provide high quality care at lower costs) are generally large delivery systems with advanced information technology, strong primary care infrastructures, andprobably most importantlytight integration between physicians and the rest of the organization. During the healthcare debate, the organizations that received the most attention were the Mayo and Cleveland Clinics and the Geisinger system in central Pennsylvania. The problem is that the defining characteristic of these organizations (and others like them) is that they have been at this business of integrated care for more than 50 years! Can the model be emulated?
Two main policy changes have been promoted to try to achieve this integration: one is a change in payment structure, the other a change in organization. The first is known as bundlingin which multiple providers are reimbursed a single sum for all the care related to an episode of illness (such as a hospitalization and a 60‐ or 90‐day period afterwards). You will recognize this as a new form of capitation, but, rather than covering all of a patient's care, a more circumscribed version, focusing on a single illness or procedure. There is some evidence that bundling does reduce costs and may improve quality, by forcing hospitals, post‐acute care facilities, and doctors into collaborative arrangements (both to deliver care and, just as complex, to split the single payment without undue acrimony).10 Fisher et al.11 have promoted a new structure to deliver this kind of bundled care more effectively: The Accountable Care Organization (ACO), which is best thought of as a less ambitious, and potentially more virtual, incarnation of the HMO.
Interestingly, while many healthcare organizations have struggled to remake themselves in Mayo's image in preparation for upcoming pressures to form ACOs, some organizations with hospitalist programs need look no further than these programs to chart a course toward more effective physician‐hospital integration.12 Why? The majority of US hospitals now have hospitalists, and virtually all hospitalist programs receive support payments from their hospital (a sizable minority are on salary from the hospital). Hospitalists recognize that part of their value equation (which justifies the hospital support dollars) is that they help the hospital deliver higher quality care more efficiently. Because of this relationship, a well‐functioning hospitalist program can assume many of the attributes of an ACO, even in organizations with otherwise challenging physician‐hospital relations. It may be that hospitals and doctors need not look to Rochester, Minnesota or Danville, Pennsylvania for positive examples of physician‐hospital integration, but simply to their own local hospitalist groups.
The Bottom Line
While proponents of the Obama reform plan celebrate its passage, virtually all experts agree that it left fundamental problems with the healthcare system unaddressed. Although the 20092010 debate did not solve these problems, the new vocabulary introduced during the debateboth reasonable policy ideas like bundling and ACOs and cynical caricatures like death panelsare here to stay. Understanding these terms and the context that shaped them will be critical for hospitalists and other stakeholders interested in the future of the American healthcare system.
On March 21, 2010, the United States Congress passed the most comprehensive healthcare reform bill since the formation of Medicare. The legislation's greatest impact will be to improve access for nearly 50 million Americans who are presently uninsured. Yet the bill does little to tackle the fundamental problems of the payment and delivery systemsproblems that have resulted in major quality gaps, large numbers of medical errors, fragmented care, and backbreaking costs.
While these tough questions were mostly kicked down the road, the debate did bring many of the key questions and potential solutions into high relief. Our political leaders, pundits, and health policy scholars introduced or popularized a number of terms during the healthcare debates of 2009‐2010 (Table 1). I will attempt to place them in context and discuss their implications for future healthcare reform efforts.
|
| Value‐based purchasing |
| Bending the cost curve |
| Comparative effectiveness research (see also NICE) |
| Dartmouth atlas (see also McAllen, Texas) |
| Death panels (see also rationing) |
| Bundled payments |
| Accountable care organizations (see also Mayo Clinic, Cleveland Clinic, Geisinger; replaces HMOs) |
Some Context for the Healthcare Reform Debate
In our capitalistic economy, we make most purchases based on considerations of value: quality divided by cost. There are few among us wealthy enough to always buy the best product, or cheap enough to always buy the least expensive. Instead, we try to determine value when we purchase a restaurant meal, a house, or a vacation.
Healthcare has traditionally been the major exception to this rule, both because healthcare insurance has partly insulated consumers (patients or their proxies) from the cost consequences of their decisions, and because it is so difficult to determine the quality of healthcare. But, over the past 10 to 15 years, problems with both the numerator and denominator of this equation have created widespread recognition of the need for change.
In the numerator, we now appreciate that there are nearly 100,000 deaths per year from medical mistakes;1 that we deliver evidence‐based care only about half the time,2 and that our healthcare system is extraordinarily fragmented and chaotic. We also know that there are more than 40 million people without healthcare insurance, a uniquely American problem, since other industrialized countries manage to guarantee coverage.
This is the fundamental conundrum that needs to be addressed by healthcare reform: we have a system that produces surprisingly low‐quality, unreliable care at an exorbitant and ever‐increasing cost, and does so while leaving more than 1 out of 8 citizens without coverage. Although government is a large payer (through Medicare, Medicaid, the Veterans Affairs [VA] programs and others), most Americans receive healthcare coverage as an employee benefit; a smaller number pay for health insurance themselves. The government has a key role even in these nongovernment‐sponsored payment systems, by providing tax breaks for healthcare coverage, creating a regulatory framework, and often defining the market through its actions in its public programs.
The end result is that all the involved partiesgovernments, businesses, providers, and patientsare crying out for change. An observer of this situation feels compelled to invoke the popular version of Stein's Law: if a trend can't continue, it won't.
Bending the Cost Curve
Everyone is now familiar with the scary trends (such as in Figure 1) demonstrating the unsustainable rate of healthcare inflation in the US, trends that are projected to lead to the insolvency of the Medicare Trust Fund within a decade. The term bending the cost curve implies that our solvency depends not on lowering total costs (a political impossibility), but rather on simply decreasing the rate of rise. There are only so many ways to do this.

The most attractive, of course, is to stop providing expensive care that adds no or little value in terms of patient outcomes. The term comparative effectiveness research (CER) emerged over the past few years to describe research that pits one approach against another (or, presumably, against no treatment) on both outcomes and cost.3 Obviously, one would favor the less expensive treatment if the efficacy were equal. However, the more common (and politically fraught) question is whether a more expensive but slightly better approach is worth its additional cost.
This, of course, makes complete sense in a world of limited resources, and some countries, mostly notably the United Kingdom, are using CER to inform healthcare coverage decisions. In the United Kingdom, the research is analyzed, and coverage recommendations made, by an organization called the National Institute for Health and Clinical Excellence (NICE).4 While NICE appears to be working well, all signs indicate that the US political system is not ready for such an approach. In fact, although Medicare generally supports CER, most of the healthcare reform proposals considered by Congress explicitly prohibited Medicare from using CER results to influence payment decisions.
If an overall CER approach is too politically difficult for the US, how about focusing on 1 small segment of healthcare: expensive care at the end of life? Over the past 30 years, a group of Dartmouth researchers has examined the costs and quality of care across the entire country, demonstrating a ubiquitous pattern of highly variable costs (varying up to 2‐fold) that is unassociated with quality and outcomes (and sometimes even inversely associated).5 The findings, well known among healthcare researchers but relatively unknown by the public until recently, were brought to public attention by a 2009 New Yorker article that made the border town of McAllen, Texas the poster child for a medical culture that produces high costs without comparable benefits.6 The Dartmouth researchers, who publish their data in a document known as The Dartmouth Atlas, have found striking variations in care at the end of life. For example, even among academic medical centers (which presumably have similarly sick patient populations), the number of hospital days in the last 6 months of life varies strikingly: patients at New York University average 27.1 days, whereas those in my hospital average 11.5 days.7
So promoting better end‐of‐life carebeing sure that patients are aware of their options and that high‐quality palliative care is availableseemed like an obvious solution to part of our cost‐quality conundrum. Some early drafts of reform bills in Congress contained provisions to pay for physicians' time to discuss end‐of‐life options. This, of course, was caricaturized into the now famous Death Panelsproving that American political discourse is not yet mature enough to support realistic discussions about difficult subjects.8
It seems like having payers (government, insurance companies) make formal decisions about which services to cover (ie, rationing) is too hard. Is there another way to force these tough choices but do so without creating a political piata?
Encircling a Population
Rather than explicitly rationing care (using CER results, for example), another way of constraining costs is to place a population of patients on a fixed budget. There is evidence that provider organizations, working within such a budget (structured in a way that permits the providers to pocket any savings), are able to reorganize and change their practice style in a way that can cut costs.9 In the 1990s, we conducted a national experiment by promoting managed care, working through integrated delivery systems called Health Maintenance Organizations (HMOs) that received fixed, capitated payments for every patient. And, in fact, these organizations did cut overall costs.
The problem was that patients neither liked HMOs nor trusted that they were acting in their best interests. Ultimately, managed care became a less important delivery mechanism, and even patients who remained in HMOs had fewer constraints on their choices. Of course, the softening of the managed care market resulted in an uptick in healthcare inflation, contributing to our present predicament.
The concept of fixed payments has resurfaced, but with some modern twists. It appears that organizations that perform best on the Dartmouth measures (namely, they provide high quality care at lower costs) are generally large delivery systems with advanced information technology, strong primary care infrastructures, andprobably most importantlytight integration between physicians and the rest of the organization. During the healthcare debate, the organizations that received the most attention were the Mayo and Cleveland Clinics and the Geisinger system in central Pennsylvania. The problem is that the defining characteristic of these organizations (and others like them) is that they have been at this business of integrated care for more than 50 years! Can the model be emulated?
Two main policy changes have been promoted to try to achieve this integration: one is a change in payment structure, the other a change in organization. The first is known as bundlingin which multiple providers are reimbursed a single sum for all the care related to an episode of illness (such as a hospitalization and a 60‐ or 90‐day period afterwards). You will recognize this as a new form of capitation, but, rather than covering all of a patient's care, a more circumscribed version, focusing on a single illness or procedure. There is some evidence that bundling does reduce costs and may improve quality, by forcing hospitals, post‐acute care facilities, and doctors into collaborative arrangements (both to deliver care and, just as complex, to split the single payment without undue acrimony).10 Fisher et al.11 have promoted a new structure to deliver this kind of bundled care more effectively: The Accountable Care Organization (ACO), which is best thought of as a less ambitious, and potentially more virtual, incarnation of the HMO.
Interestingly, while many healthcare organizations have struggled to remake themselves in Mayo's image in preparation for upcoming pressures to form ACOs, some organizations with hospitalist programs need look no further than these programs to chart a course toward more effective physician‐hospital integration.12 Why? The majority of US hospitals now have hospitalists, and virtually all hospitalist programs receive support payments from their hospital (a sizable minority are on salary from the hospital). Hospitalists recognize that part of their value equation (which justifies the hospital support dollars) is that they help the hospital deliver higher quality care more efficiently. Because of this relationship, a well‐functioning hospitalist program can assume many of the attributes of an ACO, even in organizations with otherwise challenging physician‐hospital relations. It may be that hospitals and doctors need not look to Rochester, Minnesota or Danville, Pennsylvania for positive examples of physician‐hospital integration, but simply to their own local hospitalist groups.
The Bottom Line
While proponents of the Obama reform plan celebrate its passage, virtually all experts agree that it left fundamental problems with the healthcare system unaddressed. Although the 20092010 debate did not solve these problems, the new vocabulary introduced during the debateboth reasonable policy ideas like bundling and ACOs and cynical caricatures like death panelsare here to stay. Understanding these terms and the context that shaped them will be critical for hospitalists and other stakeholders interested in the future of the American healthcare system.
- ,,, eds.To Err is Human: Building a Safer Health System.Washington DC:Committee on Quality of Health Care in America, Institute of Medicine. National Academy Press,2000.
- ,,, et al.The quality of health care delivered to adults in the United States.N Engl J Med.2003;348:2635–2645.
- ,.Health care reform and the need for comparative‐effectiveness research.N Engl J Med.2010;362:e6.
- .Saying no isn't NICE—the travails of Britain's National Institute for health and clinical excellence.N Engl J Med.2008;359:1977–1981.
- .Wrestling with variation: an interview with Jack Wennberg [interviewed by Fitzhugh Mullan].Health Aff (Millwood).2004; Suppl Web Exclsives:VAR73–80.
- .The cost conundrum. What a Texas town can teach us about health care.The New Yorker2009. Available at: http://www.newyorker. com/reporting/2009/06/01/090601fa_fact_gawande. Accessed February 2010.
- .Researchers find huge variations in end‐of‐life treatment.New York Times.2008. Available at: http://www.nytimes.com/2008/04/07/health/policy/07care.html?_r=1. Accessed February 2010.
- .Ending end‐of‐life phobia—a prescription for enlightened health care reform.N Engl J Med.2009. Available at: http://healthcarere form.nejm.org/?p=2580. Accessed February 2010.
- ,.The RAND Health Insurance Experiment and HMOs.Med Care.1990;28:191–200.
- ,,.Cost savings and physician responses to global bundled payments for Medicare heart bypass surgery.Health Care Fin Rev.1997;19:41–57.
- ,,,.Creating accountable care organizations: the extended hospital medical staff.Health Aff (Millwood).2007;26(1):w44–w57.
- . Hospitalists: a little slice of Mayo. Available at: http://community.the‐hospitalist.org/blogs/wachters_world/archive/2009/08/30/hospitalists‐a‐little‐slice‐of‐mayo.aspx. Accessed February 2010.
- ,,, eds.To Err is Human: Building a Safer Health System.Washington DC:Committee on Quality of Health Care in America, Institute of Medicine. National Academy Press,2000.
- ,,, et al.The quality of health care delivered to adults in the United States.N Engl J Med.2003;348:2635–2645.
- ,.Health care reform and the need for comparative‐effectiveness research.N Engl J Med.2010;362:e6.
- .Saying no isn't NICE—the travails of Britain's National Institute for health and clinical excellence.N Engl J Med.2008;359:1977–1981.
- .Wrestling with variation: an interview with Jack Wennberg [interviewed by Fitzhugh Mullan].Health Aff (Millwood).2004; Suppl Web Exclsives:VAR73–80.
- .The cost conundrum. What a Texas town can teach us about health care.The New Yorker2009. Available at: http://www.newyorker. com/reporting/2009/06/01/090601fa_fact_gawande. Accessed February 2010.
- .Researchers find huge variations in end‐of‐life treatment.New York Times.2008. Available at: http://www.nytimes.com/2008/04/07/health/policy/07care.html?_r=1. Accessed February 2010.
- .Ending end‐of‐life phobia—a prescription for enlightened health care reform.N Engl J Med.2009. Available at: http://healthcarere form.nejm.org/?p=2580. Accessed February 2010.
- ,.The RAND Health Insurance Experiment and HMOs.Med Care.1990;28:191–200.
- ,,.Cost savings and physician responses to global bundled payments for Medicare heart bypass surgery.Health Care Fin Rev.1997;19:41–57.
- ,,,.Creating accountable care organizations: the extended hospital medical staff.Health Aff (Millwood).2007;26(1):w44–w57.
- . Hospitalists: a little slice of Mayo. Available at: http://community.the‐hospitalist.org/blogs/wachters_world/archive/2009/08/30/hospitalists‐a‐little‐slice‐of‐mayo.aspx. Accessed February 2010.
Acute Renal Infarction
A 58‐year‐old man presented with a 1‐day history of left lower quadrant abdominal pain. He had a history of remote tobacco use, hypertension, hypercholesterolemia, deep venous thrombosis, and pulmonary embolism. His white cell count was 18,500/mm,3 and urinalysis revealed hematuria. Contrast‐enhanced abdominal computed tomography (CT) scan showed decreased perfusion of a large section of the lower pole of the left kidney (Figure 1). On day 3, his flank pain persisted, despite hydration, analgesics, and intravenous (IV) antibiotics. His serum lactate dehydrogenase (LDH) was elevated and renal infarction was suspected. Heparin was started and the patient was later discharged on warfarin. One month later, repeat contrast‐enhanced abdominal CT showed a less extensive wedge defect with scarring of the left kidney.
The diagnosis of acute renal infarction is often missed due to its nonspecific symptoms and the fact that it is an uncommon disease. It should be suspected in patients with acute flank pain and risk factors for thromboembolism including: valvular or ischemic heart disease, atrial fibrillation, and previous thromboembolic events.1, 2 Hypertension, which our patient had, is also a risk factor. In the appropriate setting hematuria and elevated LDH strongly suggest the diagnosis.3 Angiography remains the gold‐standard for diagnosis, but contrast‐enhanced CT is an acceptable alternative.3 In the setting of gross hematuria, we recommend that acute renal infarction should be suspected in all patients with the triad of persistent flank pain, high thromboembolic risk, and an increased LDH.
- ,,, et al.ED presentations of acute renal infarction.Am J Emerg Med.2007;25:164–169.
- ,,,.Renal artery embolism: clinical features and long‐term follow‐up of 17 cases.Ann Intern Med.1978;89:477–482.
- ,,, et al.Acute renal infarction. Clinical characteristics of 17 patients.Medicine (Baltimore).1999;78:386–394.
A 58‐year‐old man presented with a 1‐day history of left lower quadrant abdominal pain. He had a history of remote tobacco use, hypertension, hypercholesterolemia, deep venous thrombosis, and pulmonary embolism. His white cell count was 18,500/mm,3 and urinalysis revealed hematuria. Contrast‐enhanced abdominal computed tomography (CT) scan showed decreased perfusion of a large section of the lower pole of the left kidney (Figure 1). On day 3, his flank pain persisted, despite hydration, analgesics, and intravenous (IV) antibiotics. His serum lactate dehydrogenase (LDH) was elevated and renal infarction was suspected. Heparin was started and the patient was later discharged on warfarin. One month later, repeat contrast‐enhanced abdominal CT showed a less extensive wedge defect with scarring of the left kidney.

The diagnosis of acute renal infarction is often missed due to its nonspecific symptoms and the fact that it is an uncommon disease. It should be suspected in patients with acute flank pain and risk factors for thromboembolism including: valvular or ischemic heart disease, atrial fibrillation, and previous thromboembolic events.1, 2 Hypertension, which our patient had, is also a risk factor. In the appropriate setting hematuria and elevated LDH strongly suggest the diagnosis.3 Angiography remains the gold‐standard for diagnosis, but contrast‐enhanced CT is an acceptable alternative.3 In the setting of gross hematuria, we recommend that acute renal infarction should be suspected in all patients with the triad of persistent flank pain, high thromboembolic risk, and an increased LDH.
A 58‐year‐old man presented with a 1‐day history of left lower quadrant abdominal pain. He had a history of remote tobacco use, hypertension, hypercholesterolemia, deep venous thrombosis, and pulmonary embolism. His white cell count was 18,500/mm,3 and urinalysis revealed hematuria. Contrast‐enhanced abdominal computed tomography (CT) scan showed decreased perfusion of a large section of the lower pole of the left kidney (Figure 1). On day 3, his flank pain persisted, despite hydration, analgesics, and intravenous (IV) antibiotics. His serum lactate dehydrogenase (LDH) was elevated and renal infarction was suspected. Heparin was started and the patient was later discharged on warfarin. One month later, repeat contrast‐enhanced abdominal CT showed a less extensive wedge defect with scarring of the left kidney.

The diagnosis of acute renal infarction is often missed due to its nonspecific symptoms and the fact that it is an uncommon disease. It should be suspected in patients with acute flank pain and risk factors for thromboembolism including: valvular or ischemic heart disease, atrial fibrillation, and previous thromboembolic events.1, 2 Hypertension, which our patient had, is also a risk factor. In the appropriate setting hematuria and elevated LDH strongly suggest the diagnosis.3 Angiography remains the gold‐standard for diagnosis, but contrast‐enhanced CT is an acceptable alternative.3 In the setting of gross hematuria, we recommend that acute renal infarction should be suspected in all patients with the triad of persistent flank pain, high thromboembolic risk, and an increased LDH.
- ,,, et al.ED presentations of acute renal infarction.Am J Emerg Med.2007;25:164–169.
- ,,,.Renal artery embolism: clinical features and long‐term follow‐up of 17 cases.Ann Intern Med.1978;89:477–482.
- ,,, et al.Acute renal infarction. Clinical characteristics of 17 patients.Medicine (Baltimore).1999;78:386–394.
- ,,, et al.ED presentations of acute renal infarction.Am J Emerg Med.2007;25:164–169.
- ,,,.Renal artery embolism: clinical features and long‐term follow‐up of 17 cases.Ann Intern Med.1978;89:477–482.
- ,,, et al.Acute renal infarction. Clinical characteristics of 17 patients.Medicine (Baltimore).1999;78:386–394.