User login
Antiplatelet Therapy: Role of Effient® (prasugrel)
A Best Practices supplement to Internal Medicine News. This promotional supplement was supported by Daiichi-Sankyo, Inc. and Eli Lilly and Company.
Antiplatelet Therapy: Role of Effient® (prasugrel)
•Topics
•Faculty/Faculty Disclosures
To view the supplement, click the image above.
- Introduction
- Clinical Pharmacology
- TRITON-TIMI 38
- Bleeding
- Application of TRITON-TIMI 38 to Clinical Practice
- Conclusion
- Effient Important Safety Information
Roxana Mehran, MD
Director of Interventional Cardiovascular Research and Clinical Trials
Zena and Michael A. Wiener Cardiovascular Institute
Mount Sinai School of Medicine
Professor of Medicine
Mount Sinai School of Medicine
Chief Scientific Officer
Clinical Trials Center
Cardiovascular Research Foundation
New York, NY
Dr Mehran receives grant support from sanofi-aventis/Bristol-Myers Squibb and is a consultant for Abbott Vascular, Accumetrics, Cardiva, and The Medicines Company and is a paid consultant to Daiichi Sankyo, Inc. and Lilly USA, LLC.
Benjamin Z. Galper, MD, MPH
Medical Resident
Department of Medicine
New York Presbyterian Hospital/Columbia University Medical Center
New York, NY
Dr Galper is a paid consultant of Daiichi Sankyo, Inc. and Lilly USA, LLC.
George D. Dangas, MD, PhD
Director of Cardiovascular Innovation
Zena and Michael A. Wiener Cardiovascular Institute
Mount Sinai School of Medicine
Professor of Medicine
Mount Sinai School of Medicine
Director, Academic Affairs
Cardiovascular Research Foundation
New York, NY
Dr Dangas is a consultant for AstraZeneca and Cordis Corp. (a Johnson & Johnson Company) and a paid consultant to
Daiichi Sankyo, Inc. and Lilly USA, LLC.
Copyright © 2010 Elsevier Inc.
A Best Practices supplement to Internal Medicine News. This promotional supplement was supported by Daiichi-Sankyo, Inc. and Eli Lilly and Company.
Antiplatelet Therapy: Role of Effient® (prasugrel)
•Topics
•Faculty/Faculty Disclosures
To view the supplement, click the image above.
- Introduction
- Clinical Pharmacology
- TRITON-TIMI 38
- Bleeding
- Application of TRITON-TIMI 38 to Clinical Practice
- Conclusion
- Effient Important Safety Information
Roxana Mehran, MD
Director of Interventional Cardiovascular Research and Clinical Trials
Zena and Michael A. Wiener Cardiovascular Institute
Mount Sinai School of Medicine
Professor of Medicine
Mount Sinai School of Medicine
Chief Scientific Officer
Clinical Trials Center
Cardiovascular Research Foundation
New York, NY
Dr Mehran receives grant support from sanofi-aventis/Bristol-Myers Squibb and is a consultant for Abbott Vascular, Accumetrics, Cardiva, and The Medicines Company and is a paid consultant to Daiichi Sankyo, Inc. and Lilly USA, LLC.
Benjamin Z. Galper, MD, MPH
Medical Resident
Department of Medicine
New York Presbyterian Hospital/Columbia University Medical Center
New York, NY
Dr Galper is a paid consultant of Daiichi Sankyo, Inc. and Lilly USA, LLC.
George D. Dangas, MD, PhD
Director of Cardiovascular Innovation
Zena and Michael A. Wiener Cardiovascular Institute
Mount Sinai School of Medicine
Professor of Medicine
Mount Sinai School of Medicine
Director, Academic Affairs
Cardiovascular Research Foundation
New York, NY
Dr Dangas is a consultant for AstraZeneca and Cordis Corp. (a Johnson & Johnson Company) and a paid consultant to
Daiichi Sankyo, Inc. and Lilly USA, LLC.
Copyright © 2010 Elsevier Inc.
A Best Practices supplement to Internal Medicine News. This promotional supplement was supported by Daiichi-Sankyo, Inc. and Eli Lilly and Company.
Antiplatelet Therapy: Role of Effient® (prasugrel)
•Topics
•Faculty/Faculty Disclosures
To view the supplement, click the image above.
- Introduction
- Clinical Pharmacology
- TRITON-TIMI 38
- Bleeding
- Application of TRITON-TIMI 38 to Clinical Practice
- Conclusion
- Effient Important Safety Information
Roxana Mehran, MD
Director of Interventional Cardiovascular Research and Clinical Trials
Zena and Michael A. Wiener Cardiovascular Institute
Mount Sinai School of Medicine
Professor of Medicine
Mount Sinai School of Medicine
Chief Scientific Officer
Clinical Trials Center
Cardiovascular Research Foundation
New York, NY
Dr Mehran receives grant support from sanofi-aventis/Bristol-Myers Squibb and is a consultant for Abbott Vascular, Accumetrics, Cardiva, and The Medicines Company and is a paid consultant to Daiichi Sankyo, Inc. and Lilly USA, LLC.
Benjamin Z. Galper, MD, MPH
Medical Resident
Department of Medicine
New York Presbyterian Hospital/Columbia University Medical Center
New York, NY
Dr Galper is a paid consultant of Daiichi Sankyo, Inc. and Lilly USA, LLC.
George D. Dangas, MD, PhD
Director of Cardiovascular Innovation
Zena and Michael A. Wiener Cardiovascular Institute
Mount Sinai School of Medicine
Professor of Medicine
Mount Sinai School of Medicine
Director, Academic Affairs
Cardiovascular Research Foundation
New York, NY
Dr Dangas is a consultant for AstraZeneca and Cordis Corp. (a Johnson & Johnson Company) and a paid consultant to
Daiichi Sankyo, Inc. and Lilly USA, LLC.
Copyright © 2010 Elsevier Inc.
Acute Coronary Syndrome Update
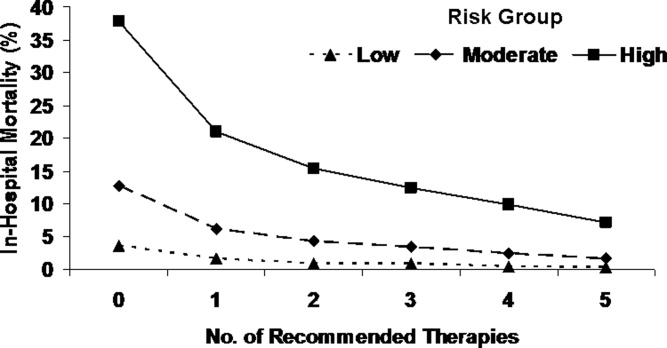
Acute coronary syndrome (ACS) remains a major healthcare challenge. Currently, the majority of patients with ACS have nonST‐segment (ST, part of an electrocardiogram between the QRS complex and the T wave) elevation myocardial infarction (MI) and unstable angina.1 Nevertheless, ST‐segment elevation MI is also an important cause of morbidity and mortality. In recent years, our understanding of ACS has improved as a result of several major advances based on results from multiple randomized clinical trials and registry analyses. The results of these analyses have influenced guidelines issued by professional societies and in some cases have become performance metrics. Therefore, it is particularly important for physicians involved in the care of patients with ACS to be aware of evolving treatment patterns (Table 1).
|
| Hospitalists should demonstrate a knowledge of: |
| ACS without enzyme leak, NSTEMI, and STEMI |
| Variable presentations of unstable angina, acute MI |
| Conditions that mimic ACS |
| Cardiac biomarkers |
| Role of noninvasive cardiac testing |
| Risks; indications for cardiac catheterization |
| Risk factors for CAD |
| Validated risk stratification tools |
| Indications for hospitalization of patients with chest pain |
| Indications, contraindications for thrombolytic therapy |
| Indications, contraindications, and pharmacology of drugs for ACS |
| Indications for early invasive interventions |
| Angiography, stenting and/or CABG |
| Laboratory studies or imaging indicative of disease severity |
| Safe hospital discharge |
| Hospitalists should demonstrate skill in: |
| History and physical exam relative to cardiac disease |
| Recognizing signs and severity of ACS |
| Diagnosing ACS through appropriate testing |
| History and physical, ECG, x‐rays, biomarkers |
| Risk stratification using validated tools |
| Formulating an evidence‐based treatment plan |
| Identifying patients for thrombolytics and/or early revascularization |
| Recognizing and treating patient discomfort |
| Recognizing decompensation, initiating immediate therapy |
| Managing complicating factors |
| Bleeding, inadequate response, cardiopulmonary compromise |
| Timely patient assessment, co‐management with other providers |
| Hospitalists should demonstrate attitudes that facilitate: |
| Communication with patients and families relative to cardiac disease and all aspects of care plan |
| Obtain informed consent |
| Early specialty consultation |
| Initiation of secondary prevention measures before discharge |
| Multidisciplinary care throughout the hospital stay |
| Safe discharge and transition back into primary care |
Case Study
A 64‐year‐old man presents to the emergency department with the chief complaint of chest pressure for the past 2 hours. His chest pressure began after he moved furniture in his home. He initially believed that a pulled muscle was the cause of the pain, but when the discomfort did not improve with rest and continued to worsen, he thought it best that his wife drive him to the emergency department, where he continues to have chest pressure. He has never had this symptom before. His past medical history is notable only for mild hypertension for which he takes hydrochlorothiazide 25 mg daily. Otherwise, he has been healthy.
Clinical Presentation and Risk Assessment
The clinical presentation of ACS is not always straightforward. Although physicians frequently inquire about chest pain, the pain often manifests as chest heaviness or chest pressure. Additionally, some patients have a more atypical presentation, where the predominant symptom of acute coronary ischemia is dyspnea or extreme fatigue. These atypical presentations are believed to be somewhat more common in women and in the elderly, but it is important to realize that they can occur in any patient. Nausea, vomiting, or diaphoresis may accompany these symptoms or occur in isolation. Chest discomfort radiating to the jaw, neck, or left arm may be present, but is not necessary to the diagnosis. Thus, we see a variety of symptoms presenting in a patient with ACS.
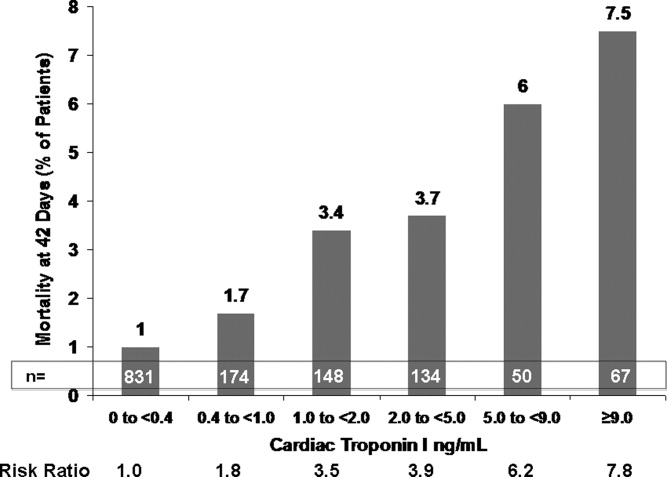
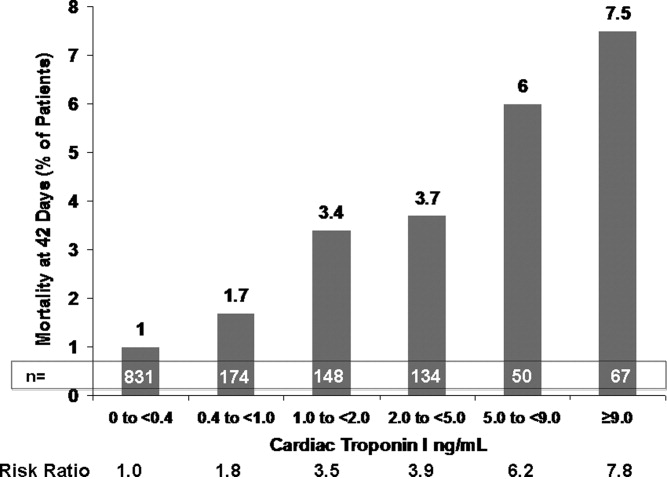
This varied presentation makes objective assessment of ACS particularly important. To inform assessment, biomarkers have emerged as a quick and effective tool to help with the diagnosis of ACS. In particular, troponin measurement is important and serial troponin measurement is useful to exclude myonecrosis. It should be noted that the initial troponin level may be normal during the early stages of ACS. A bedside troponin measurement can be useful for rapid identification of myocardial damage. Quantitative troponin measurement also adds value, as higher levels of troponin are associated with progressively worse outcomes, including mortality (Figure 1). Although a number of biomarkers are available, the most important commonly used at present is troponin.0, 0, 0, 0
In addition to biomarkers, the electrocardiogram (ECG) remains extremely important in diagnosis and risk stratification. In a moderate‐level emergency department, the triage nurse should obtain a 12‐lead ECG for a patient with a history that is suspicious for coronary ischemia, and ask the attending physician to review the ECG immediately. If there is ST‐segment elevation or any new or presumed new left bundle‐branch block, the patient should be triaged to the ST‐segment elevation MI pathway of care. If there is ST‐segment depression or marked T‐wave inversion, this greatly raises the suspicion for nonST‐segment elevation MI or unstable angina. The presence of any of these features on the ECG places the patient at markedly elevated risk of short‐term ischemic complications.
A protocol should be in place for rapid treatment of patients with ST‐segment elevation MI.2 If the hospital has 24/7 percutaneous coronary intervention (PCI) capability, the catheterization lab should be immediately activated and the patient should proceed to primary PCI. The goal door‐to‐balloon time is 90 minutes or less. A patient who presents to a hospital without primary PCI capability should receive either fibrinolysis or be transferred to a center that can perform primary PCI. If fibrinolytic therapy is planned, it is essential that the patient not have any absolute contraindications to fibrinolytic therapy. Fibrinolysis should be administered within 30 minutes of patient contact. If transfer for primary PCI is planned, it is important that systems to support the transfer are in place so that the time from first medical contact to PCI does not exceed 90 minutes. As a practical point, it can be difficult to achieve these short transfer times in many geographic regions of the United States. However, with organized systems of care, it is certainly possible to have effective transfer systems and to achieve a short door‐to‐balloon time.3
If the patient does not have ST‐segment elevation MI, the next step depends on the patient's level of risk, where risk stratification is particularly important. As mentioned above, troponin measurement and the ECG are both essential aspects of risk stratification, but they alone are not sufficient to establish risk. It is recommended that an objective risk tool also be used. This is especially important because the patient can be initially troponin‐negative and have a normal ECG but still be at high risk for ischemic complications. The TIMI risk score (Table 2) is 1 of a number of resources that can help determine whether patients are at high risk for short‐term ischemic complications using means more objective than the eyeball test (Table 3).
|
| Historical |
| Age 65 year |
| 3 CAD risk factors |
| Family history, hypertension, hypercholesterolemia, diabetes, active smoker |
| Known CAD (stenosis 50%) |
| ASA use in past 7 days |
| At presentation |
| Recent (24 hours) severe angina |
| Elevated cardiac markers |
| ST deviation 0.5 mm |
| Risk score = total points, range: 07 |
| Risk Score | Death or MI (%) | Death, MI, or Urgent Revascularization (%) |
|---|---|---|
| ||
| 0/1 | 3 | 5 |
| 2 | 3 | 8 |
| 3 | 5 | 13 |
| 4 | 7 | 20 |
| 5 | 12 | 26 |
| 6/7 | 19 | 41 |
Delineation of the coronary anatomy in the catheterization lab is warranted for patients judged to be at high risk on the basis of the TIMI risk score, which would include most patients with elevated troponin or ST‐segment deviation. Many patients will undergo PCI on the basis of those test results and a smaller percentage might undergo coronary artery bypass grafting (CABG). Additionally, a sizeable minority of patients will be managed medically. This latter group is challenging because it consists of patients who have either trivial coronary artery disease or extensive coronary artery disease not amenable to revascularization and who have either a very low or very high risk of ischemic complications.
Even though catheterization may not be necessary, further evaluation is warranted in patients with ACS deemed to be at low risk. Typically, some form of functional assessment is indicated. In patients who are able to exercise, this would consist of exercise stress testing, often with an imaging modality. If the stress test is abnormal, cardiac catheterization is often the next step.
Case Study (cont)
An ECG is rapidly obtained on this patient and there is ST‐segment depression in leads II, III, and aVF. A bedside troponin is positive. The patient is at high risk of ischemic complications. He is diagnosed with nonST‐segment elevation MI. The next step is to initiate medical therapy. Presumably, the patient would have already (or at least should have already) received aspirin. Chewing or swallowing a dose of 325 mg nonenteric coated aspirin should provide a prompt aspirin effect. It would be reasonable to initiate anticoagulation as well, and the guidelines support a number of choices such as unfractionated heparin or low molecular weight heparin. Consideration should also be given to starting additional antiplatelet therapy, such as a loading dose of clopidogrel. Although aspirin provides some degree of antiplatelet effect, in a patient with activated platelets who presents with an ACS, additional antiplatelet therapy is necessary, although the exact timing of it is a matter of debate. Finally, consideration needs to be given to the need for catheterization. This patient, on the basis of his high ischemic risk and lack of obvious contraindications, should go to the catheterization laboratory, and the timing of catheterization requires further thought.
Guideline Update
The American College of Cardiology/American Heart Association 2009 Focused Guideline Update provides new information and recommendations pertinent to the care of patients with ACS2 and incorporates new data relevant to the initial emergency care and subsequent inpatient care of patients with ACS. Guideline highlights are presented in Table 4.
| Intervention | Recommendation |
|---|---|
| |
| GP IIb/IIIa receptor antagonists | |
| Class IIa | Start abciximab, tirofiban, or eptifibatide at primary PCI (with/without stenting) in selected patients with STEMI. |
| Class IIb | Uncertain value in STEMI when given before arrival at catheterization lab. |
| Thienopyridines | |
| Class I | Use loading dose for planned PCI in STEMI. Regimens for primary and nonprimary PCI are detailed within the guideline. |
| Duration of therapy after stent placement of at least 12 months. Stop early if bleeding risk outweighs benefit. | |
| Discontinue before planned, delayed CABG (5 d clopidogrel; 7 d prasugrel) unless the need for CABG outweighs bleeding risk. | |
| Class IIb | After DES placement, consider continuing clopidogrel or prasugrel beyond the first 15 months of therapy. |
| Class III | Prasugrel is not recommended for primary PCI in patients with STEMI who have a history of stroke or TIA. |
| Parenteral anticoagulants | |
| Class I | In primary PCI, supportive anticoagulants include unfractionated heparin, enoxaparin, fondaparinux, or bivalirudin, following ASA and a thienopyridine. |
| Class IIa | Bivalirudin is reasonable in patients at high risk of bleeding undergoing PCI for STEMI. |
| Triage and transfer for PCI | |
| Class I | STEMI system of care is supported by dedicated teams and protocols required for all communities. |
| Class IIa | Transfer patients who received fibrinolytic therapy at a nonPCI‐capable facility to a PCI‐capable facility. Consider preparatory antithrombotic regimen before or during transfer. |
| Class IIb | Consider expeditious transfer of nonhigh‐risk patients from a nonPCI‐capable facility to a PCI‐capable facility after administration of fibrinolytic. Consider preparatory antithrombotic regimen before or during transfer. |
| Intensive glucose control in STEMI | |
| Class IIa | Insulin is reasonable to maintain glucose 180 mg/dL (avoid hypoglycemia) for any patient with STEMI. |
| Thrombus aspiration during PCI of STEMI | |
| Class IIa | Aspiration thombectomy is reasonable. |
| Use of stents in STEMI | |
| Class IIa | DES is a reasonable alternative to BMS for primary PCI in STEMI. |
| Class IIb | Consider DES when clinical or anatomical factors suggest favorable safety and efficacy for DES. |
| Angiography in CKD | |
| Class I | Isomolar contrast or low molecular weight contrast (not ioxaglate or iohexol) is indicated for CKD patients not on dialysis. |
| Fractional flow reserve | |
| Class IIa | FFR is useful to assess a specific coronary lesion or as an alternative to noninvasive functional testing to justify PCI. Reasonable for intermediate coronary stenosis in patients with angina. |
| Class III | Routine use of FFR is not recommended to assess severity of CAD in patients with angina who have had a positive, unequivocal, noninvasive functional study. |
| PCI for unprotected left main CAD | |
| Class IIb | PCI of left main coronary artery with stents is an alternative to CABG for anatomy associated with low risk of PCI complications and a clinical scenario with higher risk of adverse surgical outcomes. |
| Timing of angiography and antiplatelet therapy in UA/NSTEMI | |
| Class I | Initiate dual‐antiplatelet therapy for UA/NSTEMI and an invasive approach. Start ASA on presentation. Clopidogrel (before or at PCI) or prasugrel (at PCI) as a second antiplatelet agent. |
| Class IIa | Early invasive strategy within 12 to 24 hours of admission is reasonable for stabilized high‐risk UA/NSTEMI; an early approach is also reasonable for UA/STEMI not at high‐risk. |
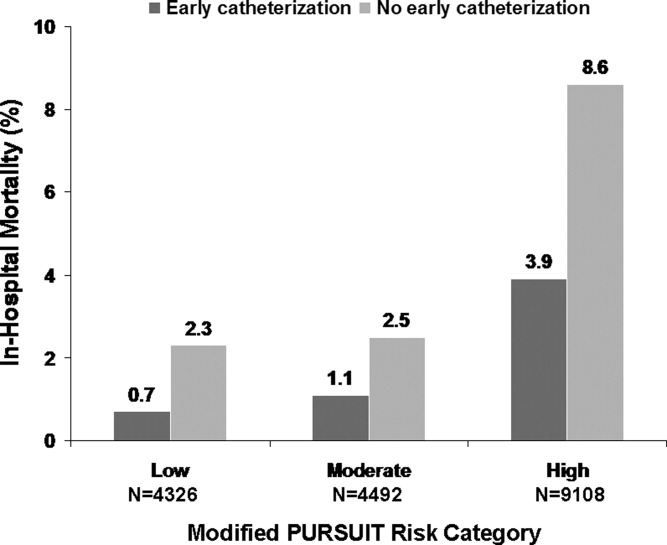
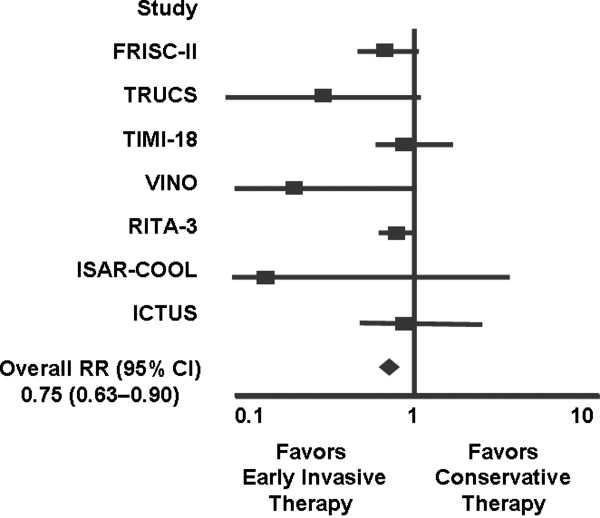
Several studies support an invasive strategy to assess high‐risk ACS patients. Randomized clinical trials and meta‐analyses of these trials have confirmed a significant reduction in subsequent ischemic events, including mortality, in patients who undergo an invasive vs. a more conservative strategy.4 Registry data have confirmed that these randomized clinical trial data reflect patients in the real‐world setting of clinical practice.5
The timing of angiography has recently been examined in detail.6, 7 It appears that for patients with nonST‐segment elevation ACS, unlike those with ST‐segment elevation MI, there is no need for emergent transfer to the catheterization laboratory, assuming patients are electrically and hemodynamically stable. Emergency transfer is warranted for unstable patients and those with ongoing chest discomfort. Otherwise, it appears sufficient to send the patient with nonST‐segment elevation ACS for catheterization within the subsequent 48 hours, or, alternatively, to adopt a more expectant approach in which catheterization is deferred until either recurrent symptoms develop or risk stratification suggests that there is substantial myocardium in jeopardy.
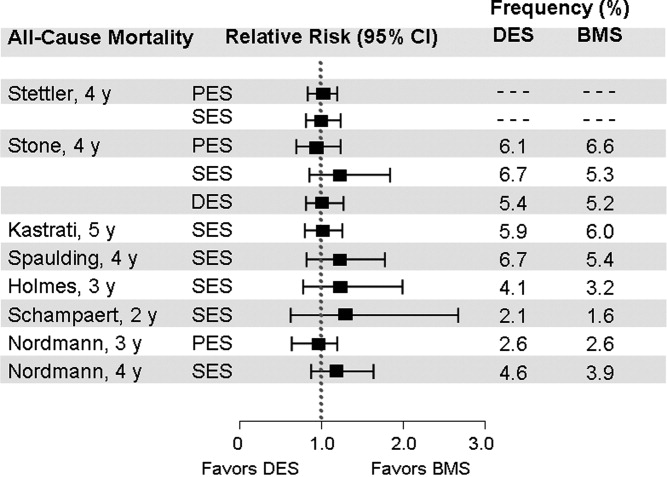
PCI is performed in the catheterization laboratory most often in the setting of ACS.5 When PCI is performed, an important consideration is whether to use a bare metal stent or a drug‐eluting stent.8 Drug‐eluting stents have been shown to have a significant benefit in reducing restenosis and the need for repeat revascularization. However, in aggregate, they have not been shown to either increase or decrease mortality.9 A key issue for the referring physician is to ascertain whether patients who go to the catheterization laboratory are likely to tolerate and be compliant with prolonged dual antiplatelet therapy. If it appears that the patient can or will not be compliant, a bare metal stent is preferable to a drug‐eluting stent; a bare metal stent requires dual antiplatelet therapy of shorter duration.
Additional considerations when sending patients to the catheterization laboratory are related to renal function. In patients with renal dysfunction, the most important way to prevent contrast nephropathy is adequate hydration prior to the procedure. In patients with left ventricular dysfunction, hydration must be done judiciously. Other strategies for preventing contrast nephropathy are being studied, although it is not entirely clear which strategies beyond hydration are truly effective.
Use of upstream glycoprotein IIb/IIIa inhibitors has become more common in patients with nonST‐segment elevation ACS. However, the most recent trial to examine this issue, the Early Glycoprotein IIb/IIIa Inhibition in NonST‐Segment Elevation Acute Coronary Syndrome (EARLY ACS) trial, did not find a clear benefit for routine administration of upstream glycoprotein IIb/IIIa inhibitors when studying all patients with ACS.10 There did appear to be a signal of benefit in troponin‐positive patients, but as an overall strategy no significant benefit and even some detriment associated with an increase in bleeding were shown. Similarly, the results of the Acute Catheterization and Urgent Intervention Triage strategY (ACUITY) trial did not support the benefit of upstream glycoprotein IIb/IIIa inhibitors in patients with ACS.11
New data have also been released with respect to the thienopyridines. The Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with PrasugrelThrombolysis in Myocardial Infarction (TRITONTIMI) 38 study found that the more potent thienopyridine prasugrel significantly reduced ischemic events when compared with clopidogrel in patients with ACS undergoing PCI.1214 A significant reduction in stent thrombosis was reported regardless of the type of stent.15 However, the study reported a significant increase in major bleeding and a small, but statistically significant, excess of fatal bleeding. A subgroup analysis of patients with diabetes or with ST‐segment elevation MI from the TRITON‐TIMI 38 study showed a particularly large benefit associated with the use of prasugrel vs. clopidogrel and, interestingly, bleeding hazards were attenuated in these subgroups.16, 17 In the small subgroup of patients with prior stroke or transient ischemic attack (TIA), there was an excessive rate of intracranial hemorrhage with prasugrel vs. clopidogrel, indicating that prasugrel should not be used in these patients. Patients age 75 years or older or who weighed less than 60 kg also appeared to have a higher bleeding risk with prasugrel compared to clopidogrel. Careful thought is needed before using prasugrel in those patients identified as having a higher risk of bleeding.
Recently, a higher clopidogrel loading dose of 600 mg vs. the standard 300 mg dose was tested in patients who presented with ACS in the Clopidogrel optimal loading dose Usage to Reduce Recurrent EveNTs‐Organization to Assess Strategies in Ischemic Syndromes (CURRENT‐OASIS) 7 trial.18 Patients also received 150 mg of clopidogrel daily for the ensuing 6 days vs. the standard 75 mg daily dose. All patients then received 75 mg clopidogrel for 1 month of follow‐up. In the overall population, there was no benefit to using the higher clopidogrel loading dose. In contrast, there was a significant reduction in stent thrombosis in patients who received stents. The higher loading dose of clopidogrel was associated with a higher rate of bleeding.
Ticagrelor is a novel adenosine diphosphate receptor antagonist that was compared with clopidogrel in patients with ACS.19, 20 Compared to clopidogrel, ticagrelor significantly reduced ischemic events and there was also a significant reduction in cardiovascular mortality and in all‐cause mortality. Surprisingly, overall major bleeding did not increase with ticagrelor, but nonCABG‐related major bleeding increased.
The use of proton pump inhibitors (PPIs) in patients receiving dual antiplatelet therapy has also been a matter of vigorous recent debate.21 Evidence to date suggests there is no significant clinical interaction between PPIs and prasugrel. The data with clopidogrel and PPIs are mixed, although data are limited because much were derived from observational studies. Randomized clinical trial data are needed to assess whether there is an interaction between clopidogrel and PPIs that warrants clinical action, although preliminary data suggest there is no adverse cardiovascular interaction.22
New data regarding the intravenous anticoagulant bivalirudin have become available and have been incorporated into the Focused Guideline Update. Although bivalirudin is to be used primarily in the catheterization laboratory during PCI, it does appear to be associated with significantly less bleeding than heparin plus glycoprotein IIb/IIIa inhibitors.11, 2326
Case Study (cont)
The patient undergoes cardiac catheterization. An occluded dominant left circumflex artery is noted and is opened up with balloon angioplasty after aspiration thrombectomy. The patient receives 60 mg of prasugrel as a loading dose and bivalirudin as the anticoagulant during the procedure. A drug‐eluting stent is implanted with excellent results. The patient is transferred to the cardiac care unit for further care. It appears that this is a patient who functionally has an ST‐segment elevation MI with an occluded artery, although it manifested on the ECG as ST depression. Because of the patient's ongoing chest discomfort, it was fortunate that prompt angiography was performed.
Discussion
Patients with ACS present several challenges in management. Risk stratification is particularly important for nonST‐segment elevation ACS and requires thoughtful evaluation by the physician. Additionally, the large amount of new data and guideline updates create a rapidly evolving field, making it difficult to keep abreast of new developments. Physicians of patients with ACS need to be aware of these key developments so that they can provide optimal care to their patients with potentially life‐threatening ACS.
Acknowledgements
Denise M. Erkkila, RPh of DIME, provided editorial assistance consisting of help with tables, figures, and reference formatting for this manuscript.
- ,,, et al.Heart disease and stroke statistics‐‐2010 update: a report from the American Heart Association.Circulation.2010;121:e46–e215.
- ,,, et al.2009 focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.2009;54:2205–2241.
- ,,, et al.Association of hospital primary angioplasty volume in ST‐segment elevation myocardial infarction with quality and outcomes.JAMA.2009;302:2207–2213.
- ,,,,.Benefit of early invasive therapy in acute coronary syndromes: a meta‐analysis of contemporary randomized clinical trials.J Am Coll Cardiol.2006;48:1319–1325.
- ,,, et al.Utilization of early invasive management strategies for high‐risk patients with non‐ST‐segment elevation acute coronary syndromes: results from the CRUSADE Quality Improvement Initiative.JAMA.2004;292:2096–2104.
- ,,, et al.Early versus delayed invasive intervention in acute coronary syndromes.N Engl J Med.2009;360:2165–2175.
- ,,, et al.Immediate vs delayed intervention for acute coronary syndromes: a randomized clinical trial.JAMA.2009;302:947–954.
- ,.Drug‐eluting stents: dual antiplatelet therapy for every survivor?Circulation.2007;116:696–699.
- ,.Appropriate use of drug‐eluting stents: balancing the reduction in restenosis with the concern of late thrombosis.Lancet.2008;371:2134–2143.
- ,,, et al.Early versus delayed, provisional eptifibatide in acute coronary syndromes.N Engl J Med.2009;360:2176–2190.
- ,,, et al.Antithrombotic strategies in patients with acute coronary syndromes undergoing early invasive management: one‐year results from the ACUITY trial.JAMA.2007;298:2497–2506.
- .Intensifying platelet inhibition‐‐navigating between Scylla and Charybdis.N Engl J Med.2007;357:2078–2081.
- .Prasugrel in clinical practice.N Engl J Med.2009;361:940–942.
- ,,, et al.Prasugrel versus clopidogrel in patients with acute coronary syndromes.N Engl J Med.2007;357:2001–2015.
- ,,, et al.Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON‐TIMI 38 trial: a subanalysis of a randomised trial.Lancet.2008;371:1353–1363.
- ,,, et al.Prasugrel compared with clopidogrel in patients undergoing percutaneous coronary intervention for ST‐elevation myocardial infarction (TRITON‐TIMI 38): double‐blind, randomised controlled trial.Lancet.2009;373:723–731.
- ,,, et al.Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the trial to assess improvement in therapeutic outcomes by optimizing platelet inhibition with prasugrel—Thrombolysis in Myocardial Infarction 38.Circulation.2008;118:1626–1636.
- , CURRENT Investigators. A randomized comparison of a clopidogrel high loading and maintenance dose regimen versus standard dose and high versus low dose aspirin in 25,000 patients with acute coronary syndromes: results of the CURRENT OASIS 7 trial. Available at: http://www.escardio.org/congresses/esc‐2009/congress‐reports/Pages/706003‐706004‐mehta‐vandewerf.aspx#discussant.2009. Accessed July 2010.
- .Antiplatelet therapy: ticagrelor in ACS‐what does PLATO teach us?Nat Rev Cardiol.2009;6:737–738.
- ,,, et al.Ticagrelor versus clopidogrel in patients with acute coronary syndromes.N Engl J Med.2009;361:1045–1057.
- ,.Omeprazole and clopidogrel: Should clinicians be worried?Cleve Clin J Med.2010;77:113–116.
- .COGENT: a prospective, randomized, placebo‐controlled trial of omeprazole in patients receiving aspirin and clopidogrel. Transcatheter Cardiovascular Therapeutics (TCT) 2009; September 24,2009;San Francisco, CA.
- ,,, et al.The Harmonizing Outcomes with RevasculariZatiON and Stents in Acute Myocardial Infarction (HORIZONS‐AMI) Trial: study design and rationale.Am Heart J.2008;156:44–56.
- ,,, et al.Bivalirudin in patients undergoing primary angioplasty for acute myocardial infarction (HORIZONS‐AMI): 1‐year results of a randomised controlled trial.Lancet.2009;374:1149–1159.
- ,,, et al.Acute Catheterization and Urgent Intervention Triage strategY (ACUITY) trial: study design and rationale.Am Heart J.2004;148:764–775.
- ,,, et al.Bivalirudin in patients with acute coronary syndromes undergoing percutaneous coronary intervention: a subgroup analysis from the Acute Catheterization and Urgent Intervention Triage strategy (ACUITY) trial.Lancet.2007;369:907–919.
- Society of Hospital Medicine.Acute coronary syndrome.J Hosp Med.2006;1 (suppl 1):2–3.
- ,,, et al.The TIMI risk score for unstable angina/non‐ST elevation MI: A method for prognostication and therapeutic decision making.JAMA.2000;284:835–842.
Acute coronary syndrome (ACS) remains a major healthcare challenge. Currently, the majority of patients with ACS have nonST‐segment (ST, part of an electrocardiogram between the QRS complex and the T wave) elevation myocardial infarction (MI) and unstable angina.1 Nevertheless, ST‐segment elevation MI is also an important cause of morbidity and mortality. In recent years, our understanding of ACS has improved as a result of several major advances based on results from multiple randomized clinical trials and registry analyses. The results of these analyses have influenced guidelines issued by professional societies and in some cases have become performance metrics. Therefore, it is particularly important for physicians involved in the care of patients with ACS to be aware of evolving treatment patterns (Table 1).
|
| Hospitalists should demonstrate a knowledge of: |
| ACS without enzyme leak, NSTEMI, and STEMI |
| Variable presentations of unstable angina, acute MI |
| Conditions that mimic ACS |
| Cardiac biomarkers |
| Role of noninvasive cardiac testing |
| Risks; indications for cardiac catheterization |
| Risk factors for CAD |
| Validated risk stratification tools |
| Indications for hospitalization of patients with chest pain |
| Indications, contraindications for thrombolytic therapy |
| Indications, contraindications, and pharmacology of drugs for ACS |
| Indications for early invasive interventions |
| Angiography, stenting and/or CABG |
| Laboratory studies or imaging indicative of disease severity |
| Safe hospital discharge |
| Hospitalists should demonstrate skill in: |
| History and physical exam relative to cardiac disease |
| Recognizing signs and severity of ACS |
| Diagnosing ACS through appropriate testing |
| History and physical, ECG, x‐rays, biomarkers |
| Risk stratification using validated tools |
| Formulating an evidence‐based treatment plan |
| Identifying patients for thrombolytics and/or early revascularization |
| Recognizing and treating patient discomfort |
| Recognizing decompensation, initiating immediate therapy |
| Managing complicating factors |
| Bleeding, inadequate response, cardiopulmonary compromise |
| Timely patient assessment, co‐management with other providers |
| Hospitalists should demonstrate attitudes that facilitate: |
| Communication with patients and families relative to cardiac disease and all aspects of care plan |
| Obtain informed consent |
| Early specialty consultation |
| Initiation of secondary prevention measures before discharge |
| Multidisciplinary care throughout the hospital stay |
| Safe discharge and transition back into primary care |
Case Study
A 64‐year‐old man presents to the emergency department with the chief complaint of chest pressure for the past 2 hours. His chest pressure began after he moved furniture in his home. He initially believed that a pulled muscle was the cause of the pain, but when the discomfort did not improve with rest and continued to worsen, he thought it best that his wife drive him to the emergency department, where he continues to have chest pressure. He has never had this symptom before. His past medical history is notable only for mild hypertension for which he takes hydrochlorothiazide 25 mg daily. Otherwise, he has been healthy.
Clinical Presentation and Risk Assessment
The clinical presentation of ACS is not always straightforward. Although physicians frequently inquire about chest pain, the pain often manifests as chest heaviness or chest pressure. Additionally, some patients have a more atypical presentation, where the predominant symptom of acute coronary ischemia is dyspnea or extreme fatigue. These atypical presentations are believed to be somewhat more common in women and in the elderly, but it is important to realize that they can occur in any patient. Nausea, vomiting, or diaphoresis may accompany these symptoms or occur in isolation. Chest discomfort radiating to the jaw, neck, or left arm may be present, but is not necessary to the diagnosis. Thus, we see a variety of symptoms presenting in a patient with ACS.
This varied presentation makes objective assessment of ACS particularly important. To inform assessment, biomarkers have emerged as a quick and effective tool to help with the diagnosis of ACS. In particular, troponin measurement is important and serial troponin measurement is useful to exclude myonecrosis. It should be noted that the initial troponin level may be normal during the early stages of ACS. A bedside troponin measurement can be useful for rapid identification of myocardial damage. Quantitative troponin measurement also adds value, as higher levels of troponin are associated with progressively worse outcomes, including mortality (Figure 1). Although a number of biomarkers are available, the most important commonly used at present is troponin.0, 0, 0, 0





In addition to biomarkers, the electrocardiogram (ECG) remains extremely important in diagnosis and risk stratification. In a moderate‐level emergency department, the triage nurse should obtain a 12‐lead ECG for a patient with a history that is suspicious for coronary ischemia, and ask the attending physician to review the ECG immediately. If there is ST‐segment elevation or any new or presumed new left bundle‐branch block, the patient should be triaged to the ST‐segment elevation MI pathway of care. If there is ST‐segment depression or marked T‐wave inversion, this greatly raises the suspicion for nonST‐segment elevation MI or unstable angina. The presence of any of these features on the ECG places the patient at markedly elevated risk of short‐term ischemic complications.
A protocol should be in place for rapid treatment of patients with ST‐segment elevation MI.2 If the hospital has 24/7 percutaneous coronary intervention (PCI) capability, the catheterization lab should be immediately activated and the patient should proceed to primary PCI. The goal door‐to‐balloon time is 90 minutes or less. A patient who presents to a hospital without primary PCI capability should receive either fibrinolysis or be transferred to a center that can perform primary PCI. If fibrinolytic therapy is planned, it is essential that the patient not have any absolute contraindications to fibrinolytic therapy. Fibrinolysis should be administered within 30 minutes of patient contact. If transfer for primary PCI is planned, it is important that systems to support the transfer are in place so that the time from first medical contact to PCI does not exceed 90 minutes. As a practical point, it can be difficult to achieve these short transfer times in many geographic regions of the United States. However, with organized systems of care, it is certainly possible to have effective transfer systems and to achieve a short door‐to‐balloon time.3
If the patient does not have ST‐segment elevation MI, the next step depends on the patient's level of risk, where risk stratification is particularly important. As mentioned above, troponin measurement and the ECG are both essential aspects of risk stratification, but they alone are not sufficient to establish risk. It is recommended that an objective risk tool also be used. This is especially important because the patient can be initially troponin‐negative and have a normal ECG but still be at high risk for ischemic complications. The TIMI risk score (Table 2) is 1 of a number of resources that can help determine whether patients are at high risk for short‐term ischemic complications using means more objective than the eyeball test (Table 3).
|
| Historical |
| Age 65 year |
| 3 CAD risk factors |
| Family history, hypertension, hypercholesterolemia, diabetes, active smoker |
| Known CAD (stenosis 50%) |
| ASA use in past 7 days |
| At presentation |
| Recent (24 hours) severe angina |
| Elevated cardiac markers |
| ST deviation 0.5 mm |
| Risk score = total points, range: 07 |
| Risk Score | Death or MI (%) | Death, MI, or Urgent Revascularization (%) |
|---|---|---|
| ||
| 0/1 | 3 | 5 |
| 2 | 3 | 8 |
| 3 | 5 | 13 |
| 4 | 7 | 20 |
| 5 | 12 | 26 |
| 6/7 | 19 | 41 |
Delineation of the coronary anatomy in the catheterization lab is warranted for patients judged to be at high risk on the basis of the TIMI risk score, which would include most patients with elevated troponin or ST‐segment deviation. Many patients will undergo PCI on the basis of those test results and a smaller percentage might undergo coronary artery bypass grafting (CABG). Additionally, a sizeable minority of patients will be managed medically. This latter group is challenging because it consists of patients who have either trivial coronary artery disease or extensive coronary artery disease not amenable to revascularization and who have either a very low or very high risk of ischemic complications.
Even though catheterization may not be necessary, further evaluation is warranted in patients with ACS deemed to be at low risk. Typically, some form of functional assessment is indicated. In patients who are able to exercise, this would consist of exercise stress testing, often with an imaging modality. If the stress test is abnormal, cardiac catheterization is often the next step.
Case Study (cont)
An ECG is rapidly obtained on this patient and there is ST‐segment depression in leads II, III, and aVF. A bedside troponin is positive. The patient is at high risk of ischemic complications. He is diagnosed with nonST‐segment elevation MI. The next step is to initiate medical therapy. Presumably, the patient would have already (or at least should have already) received aspirin. Chewing or swallowing a dose of 325 mg nonenteric coated aspirin should provide a prompt aspirin effect. It would be reasonable to initiate anticoagulation as well, and the guidelines support a number of choices such as unfractionated heparin or low molecular weight heparin. Consideration should also be given to starting additional antiplatelet therapy, such as a loading dose of clopidogrel. Although aspirin provides some degree of antiplatelet effect, in a patient with activated platelets who presents with an ACS, additional antiplatelet therapy is necessary, although the exact timing of it is a matter of debate. Finally, consideration needs to be given to the need for catheterization. This patient, on the basis of his high ischemic risk and lack of obvious contraindications, should go to the catheterization laboratory, and the timing of catheterization requires further thought.
Guideline Update
The American College of Cardiology/American Heart Association 2009 Focused Guideline Update provides new information and recommendations pertinent to the care of patients with ACS2 and incorporates new data relevant to the initial emergency care and subsequent inpatient care of patients with ACS. Guideline highlights are presented in Table 4.
| Intervention | Recommendation |
|---|---|
| |
| GP IIb/IIIa receptor antagonists | |
| Class IIa | Start abciximab, tirofiban, or eptifibatide at primary PCI (with/without stenting) in selected patients with STEMI. |
| Class IIb | Uncertain value in STEMI when given before arrival at catheterization lab. |
| Thienopyridines | |
| Class I | Use loading dose for planned PCI in STEMI. Regimens for primary and nonprimary PCI are detailed within the guideline. |
| Duration of therapy after stent placement of at least 12 months. Stop early if bleeding risk outweighs benefit. | |
| Discontinue before planned, delayed CABG (5 d clopidogrel; 7 d prasugrel) unless the need for CABG outweighs bleeding risk. | |
| Class IIb | After DES placement, consider continuing clopidogrel or prasugrel beyond the first 15 months of therapy. |
| Class III | Prasugrel is not recommended for primary PCI in patients with STEMI who have a history of stroke or TIA. |
| Parenteral anticoagulants | |
| Class I | In primary PCI, supportive anticoagulants include unfractionated heparin, enoxaparin, fondaparinux, or bivalirudin, following ASA and a thienopyridine. |
| Class IIa | Bivalirudin is reasonable in patients at high risk of bleeding undergoing PCI for STEMI. |
| Triage and transfer for PCI | |
| Class I | STEMI system of care is supported by dedicated teams and protocols required for all communities. |
| Class IIa | Transfer patients who received fibrinolytic therapy at a nonPCI‐capable facility to a PCI‐capable facility. Consider preparatory antithrombotic regimen before or during transfer. |
| Class IIb | Consider expeditious transfer of nonhigh‐risk patients from a nonPCI‐capable facility to a PCI‐capable facility after administration of fibrinolytic. Consider preparatory antithrombotic regimen before or during transfer. |
| Intensive glucose control in STEMI | |
| Class IIa | Insulin is reasonable to maintain glucose 180 mg/dL (avoid hypoglycemia) for any patient with STEMI. |
| Thrombus aspiration during PCI of STEMI | |
| Class IIa | Aspiration thombectomy is reasonable. |
| Use of stents in STEMI | |
| Class IIa | DES is a reasonable alternative to BMS for primary PCI in STEMI. |
| Class IIb | Consider DES when clinical or anatomical factors suggest favorable safety and efficacy for DES. |
| Angiography in CKD | |
| Class I | Isomolar contrast or low molecular weight contrast (not ioxaglate or iohexol) is indicated for CKD patients not on dialysis. |
| Fractional flow reserve | |
| Class IIa | FFR is useful to assess a specific coronary lesion or as an alternative to noninvasive functional testing to justify PCI. Reasonable for intermediate coronary stenosis in patients with angina. |
| Class III | Routine use of FFR is not recommended to assess severity of CAD in patients with angina who have had a positive, unequivocal, noninvasive functional study. |
| PCI for unprotected left main CAD | |
| Class IIb | PCI of left main coronary artery with stents is an alternative to CABG for anatomy associated with low risk of PCI complications and a clinical scenario with higher risk of adverse surgical outcomes. |
| Timing of angiography and antiplatelet therapy in UA/NSTEMI | |
| Class I | Initiate dual‐antiplatelet therapy for UA/NSTEMI and an invasive approach. Start ASA on presentation. Clopidogrel (before or at PCI) or prasugrel (at PCI) as a second antiplatelet agent. |
| Class IIa | Early invasive strategy within 12 to 24 hours of admission is reasonable for stabilized high‐risk UA/NSTEMI; an early approach is also reasonable for UA/STEMI not at high‐risk. |
Several studies support an invasive strategy to assess high‐risk ACS patients. Randomized clinical trials and meta‐analyses of these trials have confirmed a significant reduction in subsequent ischemic events, including mortality, in patients who undergo an invasive vs. a more conservative strategy.4 Registry data have confirmed that these randomized clinical trial data reflect patients in the real‐world setting of clinical practice.5
The timing of angiography has recently been examined in detail.6, 7 It appears that for patients with nonST‐segment elevation ACS, unlike those with ST‐segment elevation MI, there is no need for emergent transfer to the catheterization laboratory, assuming patients are electrically and hemodynamically stable. Emergency transfer is warranted for unstable patients and those with ongoing chest discomfort. Otherwise, it appears sufficient to send the patient with nonST‐segment elevation ACS for catheterization within the subsequent 48 hours, or, alternatively, to adopt a more expectant approach in which catheterization is deferred until either recurrent symptoms develop or risk stratification suggests that there is substantial myocardium in jeopardy.
PCI is performed in the catheterization laboratory most often in the setting of ACS.5 When PCI is performed, an important consideration is whether to use a bare metal stent or a drug‐eluting stent.8 Drug‐eluting stents have been shown to have a significant benefit in reducing restenosis and the need for repeat revascularization. However, in aggregate, they have not been shown to either increase or decrease mortality.9 A key issue for the referring physician is to ascertain whether patients who go to the catheterization laboratory are likely to tolerate and be compliant with prolonged dual antiplatelet therapy. If it appears that the patient can or will not be compliant, a bare metal stent is preferable to a drug‐eluting stent; a bare metal stent requires dual antiplatelet therapy of shorter duration.
Additional considerations when sending patients to the catheterization laboratory are related to renal function. In patients with renal dysfunction, the most important way to prevent contrast nephropathy is adequate hydration prior to the procedure. In patients with left ventricular dysfunction, hydration must be done judiciously. Other strategies for preventing contrast nephropathy are being studied, although it is not entirely clear which strategies beyond hydration are truly effective.
Use of upstream glycoprotein IIb/IIIa inhibitors has become more common in patients with nonST‐segment elevation ACS. However, the most recent trial to examine this issue, the Early Glycoprotein IIb/IIIa Inhibition in NonST‐Segment Elevation Acute Coronary Syndrome (EARLY ACS) trial, did not find a clear benefit for routine administration of upstream glycoprotein IIb/IIIa inhibitors when studying all patients with ACS.10 There did appear to be a signal of benefit in troponin‐positive patients, but as an overall strategy no significant benefit and even some detriment associated with an increase in bleeding were shown. Similarly, the results of the Acute Catheterization and Urgent Intervention Triage strategY (ACUITY) trial did not support the benefit of upstream glycoprotein IIb/IIIa inhibitors in patients with ACS.11
New data have also been released with respect to the thienopyridines. The Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with PrasugrelThrombolysis in Myocardial Infarction (TRITONTIMI) 38 study found that the more potent thienopyridine prasugrel significantly reduced ischemic events when compared with clopidogrel in patients with ACS undergoing PCI.1214 A significant reduction in stent thrombosis was reported regardless of the type of stent.15 However, the study reported a significant increase in major bleeding and a small, but statistically significant, excess of fatal bleeding. A subgroup analysis of patients with diabetes or with ST‐segment elevation MI from the TRITON‐TIMI 38 study showed a particularly large benefit associated with the use of prasugrel vs. clopidogrel and, interestingly, bleeding hazards were attenuated in these subgroups.16, 17 In the small subgroup of patients with prior stroke or transient ischemic attack (TIA), there was an excessive rate of intracranial hemorrhage with prasugrel vs. clopidogrel, indicating that prasugrel should not be used in these patients. Patients age 75 years or older or who weighed less than 60 kg also appeared to have a higher bleeding risk with prasugrel compared to clopidogrel. Careful thought is needed before using prasugrel in those patients identified as having a higher risk of bleeding.
Recently, a higher clopidogrel loading dose of 600 mg vs. the standard 300 mg dose was tested in patients who presented with ACS in the Clopidogrel optimal loading dose Usage to Reduce Recurrent EveNTs‐Organization to Assess Strategies in Ischemic Syndromes (CURRENT‐OASIS) 7 trial.18 Patients also received 150 mg of clopidogrel daily for the ensuing 6 days vs. the standard 75 mg daily dose. All patients then received 75 mg clopidogrel for 1 month of follow‐up. In the overall population, there was no benefit to using the higher clopidogrel loading dose. In contrast, there was a significant reduction in stent thrombosis in patients who received stents. The higher loading dose of clopidogrel was associated with a higher rate of bleeding.
Ticagrelor is a novel adenosine diphosphate receptor antagonist that was compared with clopidogrel in patients with ACS.19, 20 Compared to clopidogrel, ticagrelor significantly reduced ischemic events and there was also a significant reduction in cardiovascular mortality and in all‐cause mortality. Surprisingly, overall major bleeding did not increase with ticagrelor, but nonCABG‐related major bleeding increased.
The use of proton pump inhibitors (PPIs) in patients receiving dual antiplatelet therapy has also been a matter of vigorous recent debate.21 Evidence to date suggests there is no significant clinical interaction between PPIs and prasugrel. The data with clopidogrel and PPIs are mixed, although data are limited because much were derived from observational studies. Randomized clinical trial data are needed to assess whether there is an interaction between clopidogrel and PPIs that warrants clinical action, although preliminary data suggest there is no adverse cardiovascular interaction.22
New data regarding the intravenous anticoagulant bivalirudin have become available and have been incorporated into the Focused Guideline Update. Although bivalirudin is to be used primarily in the catheterization laboratory during PCI, it does appear to be associated with significantly less bleeding than heparin plus glycoprotein IIb/IIIa inhibitors.11, 2326
Case Study (cont)
The patient undergoes cardiac catheterization. An occluded dominant left circumflex artery is noted and is opened up with balloon angioplasty after aspiration thrombectomy. The patient receives 60 mg of prasugrel as a loading dose and bivalirudin as the anticoagulant during the procedure. A drug‐eluting stent is implanted with excellent results. The patient is transferred to the cardiac care unit for further care. It appears that this is a patient who functionally has an ST‐segment elevation MI with an occluded artery, although it manifested on the ECG as ST depression. Because of the patient's ongoing chest discomfort, it was fortunate that prompt angiography was performed.
Discussion
Patients with ACS present several challenges in management. Risk stratification is particularly important for nonST‐segment elevation ACS and requires thoughtful evaluation by the physician. Additionally, the large amount of new data and guideline updates create a rapidly evolving field, making it difficult to keep abreast of new developments. Physicians of patients with ACS need to be aware of these key developments so that they can provide optimal care to their patients with potentially life‐threatening ACS.
Acknowledgements
Denise M. Erkkila, RPh of DIME, provided editorial assistance consisting of help with tables, figures, and reference formatting for this manuscript.
Acute coronary syndrome (ACS) remains a major healthcare challenge. Currently, the majority of patients with ACS have nonST‐segment (ST, part of an electrocardiogram between the QRS complex and the T wave) elevation myocardial infarction (MI) and unstable angina.1 Nevertheless, ST‐segment elevation MI is also an important cause of morbidity and mortality. In recent years, our understanding of ACS has improved as a result of several major advances based on results from multiple randomized clinical trials and registry analyses. The results of these analyses have influenced guidelines issued by professional societies and in some cases have become performance metrics. Therefore, it is particularly important for physicians involved in the care of patients with ACS to be aware of evolving treatment patterns (Table 1).
|
| Hospitalists should demonstrate a knowledge of: |
| ACS without enzyme leak, NSTEMI, and STEMI |
| Variable presentations of unstable angina, acute MI |
| Conditions that mimic ACS |
| Cardiac biomarkers |
| Role of noninvasive cardiac testing |
| Risks; indications for cardiac catheterization |
| Risk factors for CAD |
| Validated risk stratification tools |
| Indications for hospitalization of patients with chest pain |
| Indications, contraindications for thrombolytic therapy |
| Indications, contraindications, and pharmacology of drugs for ACS |
| Indications for early invasive interventions |
| Angiography, stenting and/or CABG |
| Laboratory studies or imaging indicative of disease severity |
| Safe hospital discharge |
| Hospitalists should demonstrate skill in: |
| History and physical exam relative to cardiac disease |
| Recognizing signs and severity of ACS |
| Diagnosing ACS through appropriate testing |
| History and physical, ECG, x‐rays, biomarkers |
| Risk stratification using validated tools |
| Formulating an evidence‐based treatment plan |
| Identifying patients for thrombolytics and/or early revascularization |
| Recognizing and treating patient discomfort |
| Recognizing decompensation, initiating immediate therapy |
| Managing complicating factors |
| Bleeding, inadequate response, cardiopulmonary compromise |
| Timely patient assessment, co‐management with other providers |
| Hospitalists should demonstrate attitudes that facilitate: |
| Communication with patients and families relative to cardiac disease and all aspects of care plan |
| Obtain informed consent |
| Early specialty consultation |
| Initiation of secondary prevention measures before discharge |
| Multidisciplinary care throughout the hospital stay |
| Safe discharge and transition back into primary care |
Case Study
A 64‐year‐old man presents to the emergency department with the chief complaint of chest pressure for the past 2 hours. His chest pressure began after he moved furniture in his home. He initially believed that a pulled muscle was the cause of the pain, but when the discomfort did not improve with rest and continued to worsen, he thought it best that his wife drive him to the emergency department, where he continues to have chest pressure. He has never had this symptom before. His past medical history is notable only for mild hypertension for which he takes hydrochlorothiazide 25 mg daily. Otherwise, he has been healthy.
Clinical Presentation and Risk Assessment
The clinical presentation of ACS is not always straightforward. Although physicians frequently inquire about chest pain, the pain often manifests as chest heaviness or chest pressure. Additionally, some patients have a more atypical presentation, where the predominant symptom of acute coronary ischemia is dyspnea or extreme fatigue. These atypical presentations are believed to be somewhat more common in women and in the elderly, but it is important to realize that they can occur in any patient. Nausea, vomiting, or diaphoresis may accompany these symptoms or occur in isolation. Chest discomfort radiating to the jaw, neck, or left arm may be present, but is not necessary to the diagnosis. Thus, we see a variety of symptoms presenting in a patient with ACS.
This varied presentation makes objective assessment of ACS particularly important. To inform assessment, biomarkers have emerged as a quick and effective tool to help with the diagnosis of ACS. In particular, troponin measurement is important and serial troponin measurement is useful to exclude myonecrosis. It should be noted that the initial troponin level may be normal during the early stages of ACS. A bedside troponin measurement can be useful for rapid identification of myocardial damage. Quantitative troponin measurement also adds value, as higher levels of troponin are associated with progressively worse outcomes, including mortality (Figure 1). Although a number of biomarkers are available, the most important commonly used at present is troponin.0, 0, 0, 0





In addition to biomarkers, the electrocardiogram (ECG) remains extremely important in diagnosis and risk stratification. In a moderate‐level emergency department, the triage nurse should obtain a 12‐lead ECG for a patient with a history that is suspicious for coronary ischemia, and ask the attending physician to review the ECG immediately. If there is ST‐segment elevation or any new or presumed new left bundle‐branch block, the patient should be triaged to the ST‐segment elevation MI pathway of care. If there is ST‐segment depression or marked T‐wave inversion, this greatly raises the suspicion for nonST‐segment elevation MI or unstable angina. The presence of any of these features on the ECG places the patient at markedly elevated risk of short‐term ischemic complications.
A protocol should be in place for rapid treatment of patients with ST‐segment elevation MI.2 If the hospital has 24/7 percutaneous coronary intervention (PCI) capability, the catheterization lab should be immediately activated and the patient should proceed to primary PCI. The goal door‐to‐balloon time is 90 minutes or less. A patient who presents to a hospital without primary PCI capability should receive either fibrinolysis or be transferred to a center that can perform primary PCI. If fibrinolytic therapy is planned, it is essential that the patient not have any absolute contraindications to fibrinolytic therapy. Fibrinolysis should be administered within 30 minutes of patient contact. If transfer for primary PCI is planned, it is important that systems to support the transfer are in place so that the time from first medical contact to PCI does not exceed 90 minutes. As a practical point, it can be difficult to achieve these short transfer times in many geographic regions of the United States. However, with organized systems of care, it is certainly possible to have effective transfer systems and to achieve a short door‐to‐balloon time.3
If the patient does not have ST‐segment elevation MI, the next step depends on the patient's level of risk, where risk stratification is particularly important. As mentioned above, troponin measurement and the ECG are both essential aspects of risk stratification, but they alone are not sufficient to establish risk. It is recommended that an objective risk tool also be used. This is especially important because the patient can be initially troponin‐negative and have a normal ECG but still be at high risk for ischemic complications. The TIMI risk score (Table 2) is 1 of a number of resources that can help determine whether patients are at high risk for short‐term ischemic complications using means more objective than the eyeball test (Table 3).
|
| Historical |
| Age 65 year |
| 3 CAD risk factors |
| Family history, hypertension, hypercholesterolemia, diabetes, active smoker |
| Known CAD (stenosis 50%) |
| ASA use in past 7 days |
| At presentation |
| Recent (24 hours) severe angina |
| Elevated cardiac markers |
| ST deviation 0.5 mm |
| Risk score = total points, range: 07 |
| Risk Score | Death or MI (%) | Death, MI, or Urgent Revascularization (%) |
|---|---|---|
| ||
| 0/1 | 3 | 5 |
| 2 | 3 | 8 |
| 3 | 5 | 13 |
| 4 | 7 | 20 |
| 5 | 12 | 26 |
| 6/7 | 19 | 41 |
Delineation of the coronary anatomy in the catheterization lab is warranted for patients judged to be at high risk on the basis of the TIMI risk score, which would include most patients with elevated troponin or ST‐segment deviation. Many patients will undergo PCI on the basis of those test results and a smaller percentage might undergo coronary artery bypass grafting (CABG). Additionally, a sizeable minority of patients will be managed medically. This latter group is challenging because it consists of patients who have either trivial coronary artery disease or extensive coronary artery disease not amenable to revascularization and who have either a very low or very high risk of ischemic complications.
Even though catheterization may not be necessary, further evaluation is warranted in patients with ACS deemed to be at low risk. Typically, some form of functional assessment is indicated. In patients who are able to exercise, this would consist of exercise stress testing, often with an imaging modality. If the stress test is abnormal, cardiac catheterization is often the next step.
Case Study (cont)
An ECG is rapidly obtained on this patient and there is ST‐segment depression in leads II, III, and aVF. A bedside troponin is positive. The patient is at high risk of ischemic complications. He is diagnosed with nonST‐segment elevation MI. The next step is to initiate medical therapy. Presumably, the patient would have already (or at least should have already) received aspirin. Chewing or swallowing a dose of 325 mg nonenteric coated aspirin should provide a prompt aspirin effect. It would be reasonable to initiate anticoagulation as well, and the guidelines support a number of choices such as unfractionated heparin or low molecular weight heparin. Consideration should also be given to starting additional antiplatelet therapy, such as a loading dose of clopidogrel. Although aspirin provides some degree of antiplatelet effect, in a patient with activated platelets who presents with an ACS, additional antiplatelet therapy is necessary, although the exact timing of it is a matter of debate. Finally, consideration needs to be given to the need for catheterization. This patient, on the basis of his high ischemic risk and lack of obvious contraindications, should go to the catheterization laboratory, and the timing of catheterization requires further thought.
Guideline Update
The American College of Cardiology/American Heart Association 2009 Focused Guideline Update provides new information and recommendations pertinent to the care of patients with ACS2 and incorporates new data relevant to the initial emergency care and subsequent inpatient care of patients with ACS. Guideline highlights are presented in Table 4.
| Intervention | Recommendation |
|---|---|
| |
| GP IIb/IIIa receptor antagonists | |
| Class IIa | Start abciximab, tirofiban, or eptifibatide at primary PCI (with/without stenting) in selected patients with STEMI. |
| Class IIb | Uncertain value in STEMI when given before arrival at catheterization lab. |
| Thienopyridines | |
| Class I | Use loading dose for planned PCI in STEMI. Regimens for primary and nonprimary PCI are detailed within the guideline. |
| Duration of therapy after stent placement of at least 12 months. Stop early if bleeding risk outweighs benefit. | |
| Discontinue before planned, delayed CABG (5 d clopidogrel; 7 d prasugrel) unless the need for CABG outweighs bleeding risk. | |
| Class IIb | After DES placement, consider continuing clopidogrel or prasugrel beyond the first 15 months of therapy. |
| Class III | Prasugrel is not recommended for primary PCI in patients with STEMI who have a history of stroke or TIA. |
| Parenteral anticoagulants | |
| Class I | In primary PCI, supportive anticoagulants include unfractionated heparin, enoxaparin, fondaparinux, or bivalirudin, following ASA and a thienopyridine. |
| Class IIa | Bivalirudin is reasonable in patients at high risk of bleeding undergoing PCI for STEMI. |
| Triage and transfer for PCI | |
| Class I | STEMI system of care is supported by dedicated teams and protocols required for all communities. |
| Class IIa | Transfer patients who received fibrinolytic therapy at a nonPCI‐capable facility to a PCI‐capable facility. Consider preparatory antithrombotic regimen before or during transfer. |
| Class IIb | Consider expeditious transfer of nonhigh‐risk patients from a nonPCI‐capable facility to a PCI‐capable facility after administration of fibrinolytic. Consider preparatory antithrombotic regimen before or during transfer. |
| Intensive glucose control in STEMI | |
| Class IIa | Insulin is reasonable to maintain glucose 180 mg/dL (avoid hypoglycemia) for any patient with STEMI. |
| Thrombus aspiration during PCI of STEMI | |
| Class IIa | Aspiration thombectomy is reasonable. |
| Use of stents in STEMI | |
| Class IIa | DES is a reasonable alternative to BMS for primary PCI in STEMI. |
| Class IIb | Consider DES when clinical or anatomical factors suggest favorable safety and efficacy for DES. |
| Angiography in CKD | |
| Class I | Isomolar contrast or low molecular weight contrast (not ioxaglate or iohexol) is indicated for CKD patients not on dialysis. |
| Fractional flow reserve | |
| Class IIa | FFR is useful to assess a specific coronary lesion or as an alternative to noninvasive functional testing to justify PCI. Reasonable for intermediate coronary stenosis in patients with angina. |
| Class III | Routine use of FFR is not recommended to assess severity of CAD in patients with angina who have had a positive, unequivocal, noninvasive functional study. |
| PCI for unprotected left main CAD | |
| Class IIb | PCI of left main coronary artery with stents is an alternative to CABG for anatomy associated with low risk of PCI complications and a clinical scenario with higher risk of adverse surgical outcomes. |
| Timing of angiography and antiplatelet therapy in UA/NSTEMI | |
| Class I | Initiate dual‐antiplatelet therapy for UA/NSTEMI and an invasive approach. Start ASA on presentation. Clopidogrel (before or at PCI) or prasugrel (at PCI) as a second antiplatelet agent. |
| Class IIa | Early invasive strategy within 12 to 24 hours of admission is reasonable for stabilized high‐risk UA/NSTEMI; an early approach is also reasonable for UA/STEMI not at high‐risk. |
Several studies support an invasive strategy to assess high‐risk ACS patients. Randomized clinical trials and meta‐analyses of these trials have confirmed a significant reduction in subsequent ischemic events, including mortality, in patients who undergo an invasive vs. a more conservative strategy.4 Registry data have confirmed that these randomized clinical trial data reflect patients in the real‐world setting of clinical practice.5
The timing of angiography has recently been examined in detail.6, 7 It appears that for patients with nonST‐segment elevation ACS, unlike those with ST‐segment elevation MI, there is no need for emergent transfer to the catheterization laboratory, assuming patients are electrically and hemodynamically stable. Emergency transfer is warranted for unstable patients and those with ongoing chest discomfort. Otherwise, it appears sufficient to send the patient with nonST‐segment elevation ACS for catheterization within the subsequent 48 hours, or, alternatively, to adopt a more expectant approach in which catheterization is deferred until either recurrent symptoms develop or risk stratification suggests that there is substantial myocardium in jeopardy.
PCI is performed in the catheterization laboratory most often in the setting of ACS.5 When PCI is performed, an important consideration is whether to use a bare metal stent or a drug‐eluting stent.8 Drug‐eluting stents have been shown to have a significant benefit in reducing restenosis and the need for repeat revascularization. However, in aggregate, they have not been shown to either increase or decrease mortality.9 A key issue for the referring physician is to ascertain whether patients who go to the catheterization laboratory are likely to tolerate and be compliant with prolonged dual antiplatelet therapy. If it appears that the patient can or will not be compliant, a bare metal stent is preferable to a drug‐eluting stent; a bare metal stent requires dual antiplatelet therapy of shorter duration.
Additional considerations when sending patients to the catheterization laboratory are related to renal function. In patients with renal dysfunction, the most important way to prevent contrast nephropathy is adequate hydration prior to the procedure. In patients with left ventricular dysfunction, hydration must be done judiciously. Other strategies for preventing contrast nephropathy are being studied, although it is not entirely clear which strategies beyond hydration are truly effective.
Use of upstream glycoprotein IIb/IIIa inhibitors has become more common in patients with nonST‐segment elevation ACS. However, the most recent trial to examine this issue, the Early Glycoprotein IIb/IIIa Inhibition in NonST‐Segment Elevation Acute Coronary Syndrome (EARLY ACS) trial, did not find a clear benefit for routine administration of upstream glycoprotein IIb/IIIa inhibitors when studying all patients with ACS.10 There did appear to be a signal of benefit in troponin‐positive patients, but as an overall strategy no significant benefit and even some detriment associated with an increase in bleeding were shown. Similarly, the results of the Acute Catheterization and Urgent Intervention Triage strategY (ACUITY) trial did not support the benefit of upstream glycoprotein IIb/IIIa inhibitors in patients with ACS.11
New data have also been released with respect to the thienopyridines. The Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with PrasugrelThrombolysis in Myocardial Infarction (TRITONTIMI) 38 study found that the more potent thienopyridine prasugrel significantly reduced ischemic events when compared with clopidogrel in patients with ACS undergoing PCI.1214 A significant reduction in stent thrombosis was reported regardless of the type of stent.15 However, the study reported a significant increase in major bleeding and a small, but statistically significant, excess of fatal bleeding. A subgroup analysis of patients with diabetes or with ST‐segment elevation MI from the TRITON‐TIMI 38 study showed a particularly large benefit associated with the use of prasugrel vs. clopidogrel and, interestingly, bleeding hazards were attenuated in these subgroups.16, 17 In the small subgroup of patients with prior stroke or transient ischemic attack (TIA), there was an excessive rate of intracranial hemorrhage with prasugrel vs. clopidogrel, indicating that prasugrel should not be used in these patients. Patients age 75 years or older or who weighed less than 60 kg also appeared to have a higher bleeding risk with prasugrel compared to clopidogrel. Careful thought is needed before using prasugrel in those patients identified as having a higher risk of bleeding.
Recently, a higher clopidogrel loading dose of 600 mg vs. the standard 300 mg dose was tested in patients who presented with ACS in the Clopidogrel optimal loading dose Usage to Reduce Recurrent EveNTs‐Organization to Assess Strategies in Ischemic Syndromes (CURRENT‐OASIS) 7 trial.18 Patients also received 150 mg of clopidogrel daily for the ensuing 6 days vs. the standard 75 mg daily dose. All patients then received 75 mg clopidogrel for 1 month of follow‐up. In the overall population, there was no benefit to using the higher clopidogrel loading dose. In contrast, there was a significant reduction in stent thrombosis in patients who received stents. The higher loading dose of clopidogrel was associated with a higher rate of bleeding.
Ticagrelor is a novel adenosine diphosphate receptor antagonist that was compared with clopidogrel in patients with ACS.19, 20 Compared to clopidogrel, ticagrelor significantly reduced ischemic events and there was also a significant reduction in cardiovascular mortality and in all‐cause mortality. Surprisingly, overall major bleeding did not increase with ticagrelor, but nonCABG‐related major bleeding increased.
The use of proton pump inhibitors (PPIs) in patients receiving dual antiplatelet therapy has also been a matter of vigorous recent debate.21 Evidence to date suggests there is no significant clinical interaction between PPIs and prasugrel. The data with clopidogrel and PPIs are mixed, although data are limited because much were derived from observational studies. Randomized clinical trial data are needed to assess whether there is an interaction between clopidogrel and PPIs that warrants clinical action, although preliminary data suggest there is no adverse cardiovascular interaction.22
New data regarding the intravenous anticoagulant bivalirudin have become available and have been incorporated into the Focused Guideline Update. Although bivalirudin is to be used primarily in the catheterization laboratory during PCI, it does appear to be associated with significantly less bleeding than heparin plus glycoprotein IIb/IIIa inhibitors.11, 2326
Case Study (cont)
The patient undergoes cardiac catheterization. An occluded dominant left circumflex artery is noted and is opened up with balloon angioplasty after aspiration thrombectomy. The patient receives 60 mg of prasugrel as a loading dose and bivalirudin as the anticoagulant during the procedure. A drug‐eluting stent is implanted with excellent results. The patient is transferred to the cardiac care unit for further care. It appears that this is a patient who functionally has an ST‐segment elevation MI with an occluded artery, although it manifested on the ECG as ST depression. Because of the patient's ongoing chest discomfort, it was fortunate that prompt angiography was performed.
Discussion
Patients with ACS present several challenges in management. Risk stratification is particularly important for nonST‐segment elevation ACS and requires thoughtful evaluation by the physician. Additionally, the large amount of new data and guideline updates create a rapidly evolving field, making it difficult to keep abreast of new developments. Physicians of patients with ACS need to be aware of these key developments so that they can provide optimal care to their patients with potentially life‐threatening ACS.
Acknowledgements
Denise M. Erkkila, RPh of DIME, provided editorial assistance consisting of help with tables, figures, and reference formatting for this manuscript.
- ,,, et al.Heart disease and stroke statistics‐‐2010 update: a report from the American Heart Association.Circulation.2010;121:e46–e215.
- ,,, et al.2009 focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.2009;54:2205–2241.
- ,,, et al.Association of hospital primary angioplasty volume in ST‐segment elevation myocardial infarction with quality and outcomes.JAMA.2009;302:2207–2213.
- ,,,,.Benefit of early invasive therapy in acute coronary syndromes: a meta‐analysis of contemporary randomized clinical trials.J Am Coll Cardiol.2006;48:1319–1325.
- ,,, et al.Utilization of early invasive management strategies for high‐risk patients with non‐ST‐segment elevation acute coronary syndromes: results from the CRUSADE Quality Improvement Initiative.JAMA.2004;292:2096–2104.
- ,,, et al.Early versus delayed invasive intervention in acute coronary syndromes.N Engl J Med.2009;360:2165–2175.
- ,,, et al.Immediate vs delayed intervention for acute coronary syndromes: a randomized clinical trial.JAMA.2009;302:947–954.
- ,.Drug‐eluting stents: dual antiplatelet therapy for every survivor?Circulation.2007;116:696–699.
- ,.Appropriate use of drug‐eluting stents: balancing the reduction in restenosis with the concern of late thrombosis.Lancet.2008;371:2134–2143.
- ,,, et al.Early versus delayed, provisional eptifibatide in acute coronary syndromes.N Engl J Med.2009;360:2176–2190.
- ,,, et al.Antithrombotic strategies in patients with acute coronary syndromes undergoing early invasive management: one‐year results from the ACUITY trial.JAMA.2007;298:2497–2506.
- .Intensifying platelet inhibition‐‐navigating between Scylla and Charybdis.N Engl J Med.2007;357:2078–2081.
- .Prasugrel in clinical practice.N Engl J Med.2009;361:940–942.
- ,,, et al.Prasugrel versus clopidogrel in patients with acute coronary syndromes.N Engl J Med.2007;357:2001–2015.
- ,,, et al.Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON‐TIMI 38 trial: a subanalysis of a randomised trial.Lancet.2008;371:1353–1363.
- ,,, et al.Prasugrel compared with clopidogrel in patients undergoing percutaneous coronary intervention for ST‐elevation myocardial infarction (TRITON‐TIMI 38): double‐blind, randomised controlled trial.Lancet.2009;373:723–731.
- ,,, et al.Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the trial to assess improvement in therapeutic outcomes by optimizing platelet inhibition with prasugrel—Thrombolysis in Myocardial Infarction 38.Circulation.2008;118:1626–1636.
- , CURRENT Investigators. A randomized comparison of a clopidogrel high loading and maintenance dose regimen versus standard dose and high versus low dose aspirin in 25,000 patients with acute coronary syndromes: results of the CURRENT OASIS 7 trial. Available at: http://www.escardio.org/congresses/esc‐2009/congress‐reports/Pages/706003‐706004‐mehta‐vandewerf.aspx#discussant.2009. Accessed July 2010.
- .Antiplatelet therapy: ticagrelor in ACS‐what does PLATO teach us?Nat Rev Cardiol.2009;6:737–738.
- ,,, et al.Ticagrelor versus clopidogrel in patients with acute coronary syndromes.N Engl J Med.2009;361:1045–1057.
- ,.Omeprazole and clopidogrel: Should clinicians be worried?Cleve Clin J Med.2010;77:113–116.
- .COGENT: a prospective, randomized, placebo‐controlled trial of omeprazole in patients receiving aspirin and clopidogrel. Transcatheter Cardiovascular Therapeutics (TCT) 2009; September 24,2009;San Francisco, CA.
- ,,, et al.The Harmonizing Outcomes with RevasculariZatiON and Stents in Acute Myocardial Infarction (HORIZONS‐AMI) Trial: study design and rationale.Am Heart J.2008;156:44–56.
- ,,, et al.Bivalirudin in patients undergoing primary angioplasty for acute myocardial infarction (HORIZONS‐AMI): 1‐year results of a randomised controlled trial.Lancet.2009;374:1149–1159.
- ,,, et al.Acute Catheterization and Urgent Intervention Triage strategY (ACUITY) trial: study design and rationale.Am Heart J.2004;148:764–775.
- ,,, et al.Bivalirudin in patients with acute coronary syndromes undergoing percutaneous coronary intervention: a subgroup analysis from the Acute Catheterization and Urgent Intervention Triage strategy (ACUITY) trial.Lancet.2007;369:907–919.
- Society of Hospital Medicine.Acute coronary syndrome.J Hosp Med.2006;1 (suppl 1):2–3.
- ,,, et al.The TIMI risk score for unstable angina/non‐ST elevation MI: A method for prognostication and therapeutic decision making.JAMA.2000;284:835–842.
- ,,, et al.Heart disease and stroke statistics‐‐2010 update: a report from the American Heart Association.Circulation.2010;121:e46–e215.
- ,,, et al.2009 focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.2009;54:2205–2241.
- ,,, et al.Association of hospital primary angioplasty volume in ST‐segment elevation myocardial infarction with quality and outcomes.JAMA.2009;302:2207–2213.
- ,,,,.Benefit of early invasive therapy in acute coronary syndromes: a meta‐analysis of contemporary randomized clinical trials.J Am Coll Cardiol.2006;48:1319–1325.
- ,,, et al.Utilization of early invasive management strategies for high‐risk patients with non‐ST‐segment elevation acute coronary syndromes: results from the CRUSADE Quality Improvement Initiative.JAMA.2004;292:2096–2104.
- ,,, et al.Early versus delayed invasive intervention in acute coronary syndromes.N Engl J Med.2009;360:2165–2175.
- ,,, et al.Immediate vs delayed intervention for acute coronary syndromes: a randomized clinical trial.JAMA.2009;302:947–954.
- ,.Drug‐eluting stents: dual antiplatelet therapy for every survivor?Circulation.2007;116:696–699.
- ,.Appropriate use of drug‐eluting stents: balancing the reduction in restenosis with the concern of late thrombosis.Lancet.2008;371:2134–2143.
- ,,, et al.Early versus delayed, provisional eptifibatide in acute coronary syndromes.N Engl J Med.2009;360:2176–2190.
- ,,, et al.Antithrombotic strategies in patients with acute coronary syndromes undergoing early invasive management: one‐year results from the ACUITY trial.JAMA.2007;298:2497–2506.
- .Intensifying platelet inhibition‐‐navigating between Scylla and Charybdis.N Engl J Med.2007;357:2078–2081.
- .Prasugrel in clinical practice.N Engl J Med.2009;361:940–942.
- ,,, et al.Prasugrel versus clopidogrel in patients with acute coronary syndromes.N Engl J Med.2007;357:2001–2015.
- ,,, et al.Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON‐TIMI 38 trial: a subanalysis of a randomised trial.Lancet.2008;371:1353–1363.
- ,,, et al.Prasugrel compared with clopidogrel in patients undergoing percutaneous coronary intervention for ST‐elevation myocardial infarction (TRITON‐TIMI 38): double‐blind, randomised controlled trial.Lancet.2009;373:723–731.
- ,,, et al.Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the trial to assess improvement in therapeutic outcomes by optimizing platelet inhibition with prasugrel—Thrombolysis in Myocardial Infarction 38.Circulation.2008;118:1626–1636.
- , CURRENT Investigators. A randomized comparison of a clopidogrel high loading and maintenance dose regimen versus standard dose and high versus low dose aspirin in 25,000 patients with acute coronary syndromes: results of the CURRENT OASIS 7 trial. Available at: http://www.escardio.org/congresses/esc‐2009/congress‐reports/Pages/706003‐706004‐mehta‐vandewerf.aspx#discussant.2009. Accessed July 2010.
- .Antiplatelet therapy: ticagrelor in ACS‐what does PLATO teach us?Nat Rev Cardiol.2009;6:737–738.
- ,,, et al.Ticagrelor versus clopidogrel in patients with acute coronary syndromes.N Engl J Med.2009;361:1045–1057.
- ,.Omeprazole and clopidogrel: Should clinicians be worried?Cleve Clin J Med.2010;77:113–116.
- .COGENT: a prospective, randomized, placebo‐controlled trial of omeprazole in patients receiving aspirin and clopidogrel. Transcatheter Cardiovascular Therapeutics (TCT) 2009; September 24,2009;San Francisco, CA.
- ,,, et al.The Harmonizing Outcomes with RevasculariZatiON and Stents in Acute Myocardial Infarction (HORIZONS‐AMI) Trial: study design and rationale.Am Heart J.2008;156:44–56.
- ,,, et al.Bivalirudin in patients undergoing primary angioplasty for acute myocardial infarction (HORIZONS‐AMI): 1‐year results of a randomised controlled trial.Lancet.2009;374:1149–1159.
- ,,, et al.Acute Catheterization and Urgent Intervention Triage strategY (ACUITY) trial: study design and rationale.Am Heart J.2004;148:764–775.
- ,,, et al.Bivalirudin in patients with acute coronary syndromes undergoing percutaneous coronary intervention: a subgroup analysis from the Acute Catheterization and Urgent Intervention Triage strategy (ACUITY) trial.Lancet.2007;369:907–919.
- Society of Hospital Medicine.Acute coronary syndrome.J Hosp Med.2006;1 (suppl 1):2–3.
- ,,, et al.The TIMI risk score for unstable angina/non‐ST elevation MI: A method for prognostication and therapeutic decision making.JAMA.2000;284:835–842.
QI Systems for Managing ACS
Addressing quality improvement (QI) for the management of acute coronary syndrome (ACS) at the institutional level is essential for supporting hospitalists and other clinicians as they manage patients with ACS and achieve desired institutional outcomes. This systems approach may identify institution‐specific barriers to quality care, including those that affect the complex management of ACS. Having a firsthand view of patient care puts the hospitalist in a good position to assess the viability of existing processes and protocols that support care. Indeed, the hospitalist has a vested interest in improving systems of care as these systems directly affect the hospitalist's practice. This unique perspective gives the hospitalist an opportunity to facilitate systems change within the institution and to become an integral participant or leader in QI initiatives.
An increasing number of hospitalists are providing critical care at secondary and tertiary care facilities, driven by a shortage of intensivists in the United States. In a 20052006 survey about 75% of hospitalists reported they provide critical care services as part of their practice,1 and this would include care of patients with ACS. The Society of Hospital Medicine (SHM) has developed core competencies that recognize the important role of hospitalists in leading or participating in QI teams for ACS.2 Hospitalists must also be able to apply evidenced‐based outcomes data to support these initiatives.3 Hospitalist competencies for ACS include protocol development that supports the timely diagnosis and treatment of ACS, evaluation of resource utilization, staff education of secondary prevention measures, and implementation of measures to ensure institutional compliance with national quality standards (Table 1).2 Most hospital medicine groups will be expected to contribute to systems improvement, an area where hospitalists have already shown leadership for QI protocols related to glycemic control and venous thromboembolism prophylaxis. Hospitalists were prominent in targeting QI in these areas even though these QI initiatives could easily have been spearheaded by specialists.46
|
| Lead, coordinate, or participate in: |
| Protocol development to rapidly identify ACS, minimizing time to intervention |
| Protocol development for rapid identification and transfer of patients with ACS to a facility with an appropriate level of care |
| Multidisciplinary initiatives, including order sets for ACS and chest pain, that promote patient safety and optimize the use of resources |
| Staff education initiatives on the value of smoking cessation counseling and other prevention measures |
| Implement or integrate: |
| Systems to ensure hospitalwide adherence to national standards, documenting adherence as required by certifying organizations |
| Outcomes research, institution‐specific laboratory policies, and hospital formulary to create indicated and cost‐effective diagnostic and management strategies for patients with ACS |
QI Basics
QI at the institutional level addresses systems of care rather than individual performance, targeting both institutional performance and use of resources.3 QI is a continuous process in which practices and procedures related to patient management are regularly assessed to ascertain whether a quality gap exists. This in turn may lead to new processes, protocols, and algorithms that help the institution and clinicians meet benchmarks of quality care.
QI starts when an existing gap is recognizedthe gap between the scientific understanding of optimal care and actual patient care. The goal is to narrow or close this gap so that each patient receives optimal care. Underlying any QI initiative are 2 essential concepts. First, improvement requires systems changeany system will produce exactly what it is designed to produce. For example, if procedures are not in place to educate patients about smoking cessation while they are hospitalized, it is unlikely that the majority will routinely receive this counseling before they are discharged. Second, less is moreproductivity is not destroyed but often is enhanced by initiating simple and practical change. If a patient arrives in the emergency department (ED) with chest pain, instituting an order set that reminds clinicians to start antiplatelet therapy or a beta blocker can lessen the chance that these medications will be overlooked, especially when the patient transitions between providers or services within the hospital.
SHM has identified 7 essential elements of any QI initiative, and these are applicable to the care of the ACS patient (Table 2).7 These elements highlight the need for institutional support and teamwork that support standardized measures and tools specific to issues in ACS management. These issues include: (1) rapidly identifying a patient with ACS and initiating a care plan when the patient is admitted; (2) encouraging good communication between providers; (3) symptom management; (4) medication safety, polypharmacy, and medication reconciliation; (5) patient and caregiver education; (6) safe discharge and transitions in care; and (7) meeting Centers for Medicare and Medicaid Services (CMS) core measures.
| |
| Institutional support | Commitment of time, personnel, and tools to support the initiative |
| Multidisciplinary team | Team that focuses on quality of care for patients with ACS |
| Reliable metrics | Reflect CMS core and applicable PQRI measures; also reportable to inform team decision making |
| Identify the goal | Establish a measurable, achievable goal with an established timeline |
| Standard order sets | Defined clinical pathways that support evidence‐based treatment strategies, risk stratification, and safe transitions in care |
| Policies that support algorithms and protocols | Institution‐specific to support order sets |
| Education programs | Targeted to clinicians and patients; should cover items addressed in order sets, algorithms, and protocols |
Tools such as process flow mapping and run charts can reveal quality gaps and indicate if process improvements are leading to stated objectives. Process flow mapping makes it possible to identify and visualize quality gaps that might otherwise be hidden and to identify their source or cause. Process mapping documents discrete steps within the flow and usually requires input from multiple disciplines; this information can guide a multidisciplinary QI team when formulating interventions for process improvement.7 Figure 1 shows process flow mapping for the early identification of ST‐segment elevation myocardial infarction (STEMI) when hospitalists have open access to activate the catheterization laboratory (cath lab). The time from door‐to‐balloon is a critical factor in reducing STEMI‐related morbidity8; processes that overcome delays to percutaneous coronary intervention (PCI) and improve communication can have a direct benefit on patient outcomes.
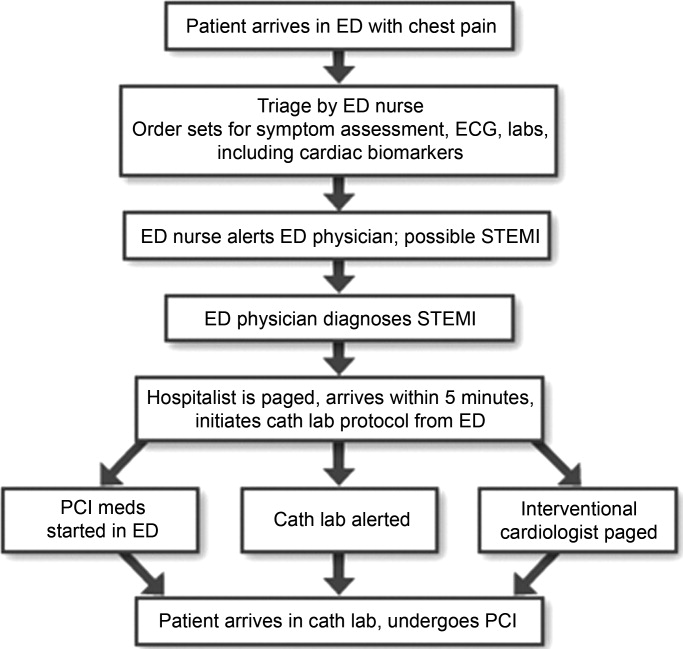
A run chart is a commonly used tool which graphically depicts progress in attaining a goal over time, before and after an intervention. Figure 2a is a run chart that shows the average time to PCI following implementation of an order set designed to support the use of a risk stratification tool for the early diagnosis of STEMI. In this case, the average time to PCI is observed to decrease over time, but still lags behind the desired goal of less than 90 minutes. This run chart indicates that further work is needed to improve the process of identifying patients with STEMI. Figure 2b shows the number of eligible STEMI patients who received aspirin at discharge following implementation of a discharge order set; here, the hospital has clearly made progress toward meeting this CMS core measure.
The following is a case study that illustrates how the implementation of standardized measures and tools can help hospitalists and other clinicians achieve quality measures in the care of a patient with ACS.
Case Study
Mary, a 68‐year‐old woman, presents to the ED with fatigue and some heartburn. She has a history of hypertension and may have had a mini stroke a few years ago. Her symptoms, which she has had on and off for the past few days, worsened considerably right before her arrival. She has been taking chewable antacids but is unsure if they have helped. Her physical exam is unremarkable. A stat electrocardiogram (ECG) shows inferior ST (part of an electrocardiogram between the QRS complex and the T wave) elevation in leads II and III, and augmented vector foot (aVF). Troponins are positive. This case can be approached from 2 perspectives.
Scenario 1
Mary arrives at a hospital that has few standardized systems or protocols for triage and transitions in care. The triage nurse recognizes the need for and obtains an ECG, but fails to alert the ED physician. When the ED physician eventually sees the patient and reviews the ECG, she immediately administers nitroglycerin and pages the cardiologist on call and the hospitalist. The hospitalist arrives first and recognizes the STEMI, but hesitates to start unfractionated or low molecular weight heparin until the cardiologist determines whether the patient will undergo PCI. The cardiologist orders PCI, the cath lab is alerted, and the patient is started on the appropriate medications; the patient does not start an aspirin until after PCI. In this scenario, several short delays amount to a significant delay of about 2 hours before the patient reaches the cath lab. In addition, although aspirin is given on day 1, it is not done on arrival. Following PCI, the patient is transferred to the coronary care unit (CCU).
Scenario 2
Mary arrives at an institution with well‐defined, institution‐specific protocols for triage and transitions in care. The triage nurse recognizes the need for and obtains an ECG; the results are immediately reviewed with the ED physician. STEMI is diagnosed and the cath lab protocol is activated; the patient receives aspirin as part of a standing order. The hospitalist meets the patient in the ED within 5 minutes and begins the protocol for unfractionated heparin and preparation for immediate PCI, allowing the ED physician to return to ED care. Simultaneously, the interventional cardiologist and cath lab are mobilized and the patient is transferred within 15 minutes. Additional appropriate medications are begun. The door‐to‐balloon time is 60 minutes, well within recommended timeframes. Following PCI, the patient is transferred to the CCU.
Measuring Performance Relative to ACS
Two agencies promulgating quality measures for hospital inpatient care are The Joint Commission (TJC) and the CMS. TJC and CMS jointly established core measures for patients with acute myocardial infarction (AMI) and these are considered key indicators of quality, evidence‐based care. As outlined in Table 3, the TJC/CMS recommendations promote, unless contraindicated, a minimum standard of care for patients with AMI.9, 10 Public reporting of institutional core measure performance has led to an intense focus on improving these metrics.
| Core Measure | Sample Metric |
|---|---|
| |
| Aspirin at arrival | Percentage of patients receiving aspirin within 24 hours before or after hospital arrival |
| Aspirin at discharge | Percentage of patients prescribed aspirin at hospital discharge |
| ACE inhibitor or ARB for LVSD | Percentage of patients with LVSD prescribed an ACEI or ARB at hospital discharge |
| Beta blocker | Percentage of patients prescribed a beta blocker at hospital discharge |
| Fibrinolytic medication given within 30 minutes of hospital arrival | Median time to fibrinolysis |
| Percentage of patients given fibrinolytic medication within 30 minutes of hospital arrival | |
| PCI received within 90 minutes of hospital arrival | Median time to PCI |
| Percentage of patients receiving primary PCI within 90 minutes of hospital arrival | |
| Smoking cessation counseling | Percentage of patients with a history of smoking cigarettes who are counseled about smoking cessation during hospitalization |
On a national level, TJC documented performance improvement from 2002 to 2008 for each of the core measures. Compliance with smoking cessation counseling improved the most, rising from 67% to almost 99%. In 2008, a combined measure of all individual core measures indicated that, overall, care of heart attack patients is of high quality. Hospitals delivered evidenced‐based care in 96.7% of the opportunities they had to provide this care. Compliance related to oral medications was also good (95%); however, a closer look at other individual measures shows that improvement is needed to reduce the time to fibrinolysis (52.4% compliance rate for 30 minutes or less) and time to primary PCI (81.6% compliance rate for 90 minutes or less).11
Financial incentives are tied to QI measures including those from CMS. At present, this system is in the pay‐for‐reporting phase, in which institutions are penalized for not reporting quality metrics for the CMS core measures. It is likely that, to further incentivize institutions to meet quality benchmarks, this will be expanded to a pay‐for‐performance system (eg, differential payments for readmissions or different payment scales based on prior performance). Public disclosure of institutional performance relative to ACS and other medical conditions is available through the HHS.gov website (www.hospitalcompare.hhs.gov), which compares performance between hospitals and provides a clear business motivation for institutions to improve and provide high quality care.
Two other reporting systems should be noted. The CMS Physician Quality Reporting Initiative (PQRI) includes physician‐related quality measures specific to ACS12 that overlap with institution‐level CMS core measures with regard to prescribing ACS medications. Payments associated with the PQRI are currently a small financial factor for hospitalists, but will likely grow as quality initiatives develop overall. The Hospital Care Quality Information From the Consumer Perspective (HCAHPS) initiative, also from CMS, strives to capture patients' perspectives on hospital care through a standardized survey. The goals are to collect data that can be used to compare hospital performance, create an incentive for QI through public disclosure of results, and increase transparency regarding the quality of hospital care.13 However, the focus is not ACS‐specific. Patients are queried about communication with hospital staff, communication about medications, and information about discharge, all areas of concern for the hospitalist in general that have been identified as areas for improvement relative to ACS.
Case Study (cont)
Scenario 1 (cont)
Mary recuperates in the CCU and is transferred to the medical floor. The hospitalist and cardiologist see her separately on rounds and each assumes the other has a reason for not starting a beta blocker; therefore, a beta blocker is not prescribed. The nurse cannot tell who is in charge and does not anticipate the day of dischargeshe is just implementing the orders as they are written. The day of discharge arrives; the nurse learns from Mary that the hospitalist will discharge her that day. The hospitalist reviews the list of ACS medications and realizes that Mary still has not started a beta blocker. He cannot reach the cardiologist before Mary's ride home arrives, so he writes a note in the discharge summary alerting the primary care physician (PCP) to consider a beta blocker at follow‐up. Because of this lack of communication and systems for tracking the implementation of guideline‐recommended therapies, Mary is discharged without a CMS core measure medication, with no assurance that this will be addressed by the PCP.
Scenario 2 (cont)
Mary recuperates in the CCU and is transferred to the medical floor. Standard post‐PCI/STEMI orders are in place according to institutional protocol. The hospitalist is able to confirm that all interventions required at admission (aspirin, beta blocker, assessment of smoking status) have occurred by reviewing a well‐structured checklist that includes easy‐to‐read visual cues. The checklist indicates that Mary was counseled about smoking cessation on day 1 of her stay in the CCU. Mary and her physicians and nurses are all aware of the target discharge date and the milestones that must be met prior to discharge (eg, echocardiogram, medication review, education, assessment of mobility, etc). Mary is instructed about each new medication and given educational materials.14 Follow‐up appointments postdischarge are made, and the discharge summary is sent electronically to the PCP. These institution‐specific protocols enhance communication overall and help the hospital meet high standards of patient care.
Special QI Issues in ACS Management for the Hospitalist
Coordination is especially important for patients with ACS because their care is so complex. Seamless transitions promote safe care as the patient moves from the ED, cath lab, recovery unit, medical floor, and discharge through the transition to primary care. Poor communication between clinicians during these transitions may result in delayed or overlooked treatment and other medical errors. Implementing an institutional system of care may overcome communication barriers and help ensure the institution meets its quality benchmarks, such as the CMS core measures. Standard order sets and protocols identify the steps and components needed to manage ACS. At admission, these measures promote early assessment of patient risk, triage to early intervention, medication reconciliation, and communication between stakeholders such as the hospitalist, cardiologist, and the cath lab.15, 16 During hospitalization, they help assure ongoing risk assessment and early consideration of discharge planning, culminating in discharge and the transition back to primary care.
A recent guideline update8 focused attention on the coordination of care between institutions and the critical importance of rapid triage for patients who need primary PCI and transfer from a non‐PCI to a PCI‐capable institution. The decision to transfer rests on multiple factors and requires rapid decision making on the part of clinicians. Time to reperfusion is shown to have a direct effect on patient outcomes. Established protocols within a non‐PCI facility can support timely transfer to a PCI‐capable facility if indicated. Factors such as the patient's mortality risk, the risk of bleeding from fibrinolytic therapy, duration of symptoms, and the time for transport to a PCI facility all must be considered. It is recognized that a regional system of STEMI care best supports collaborative efforts between institutions and community resources that support QI efforts.
Transitions in care, and particularly discharge, are areas with identified gaps in quality care17, 18 for which improvement has been pursued on an institutional level. Project BOOST (Better Outcomes for Older Adults Through Safe Transitions) seeks to improve the care of patients during the transition from inpatient to outpatient care, and focuses on elderly patients identified to be at high‐risk for adverse events during this transition.19 The goal is to improve outcomes related to 30‐day readmission rates, patient satisfaction, communication between inpatient and outpatient providers, identification of high‐risk patients who need intervention to reduce risk, and patient education about their risk for an adverse event. BOOST addresses these outcomes at an institutional level by offering resources related to project management, data collection, tools for clinicians and patients, and participating mentor institutions. These resources help an institution assess its readiness for change, identify quality gaps, promote teamwork, and guide the implementation and subsequent evaluation of process improvements. Specific tools for clinicians also support institutional goals for teamwork and communication, creating an environment for safe transitions. Both BOOST19 and the adaptable Transitions Tool from the SHM20 provide a framework for understanding processes that involve multiple departments and stakeholders, breaking complex processes into discrete parts for which quality gaps can be identified and change instituted to improve care. A checklist can also be a useful tool for ensuring specific issues are addressed during transitions in care. SHM developed a checklist for hospitalist use that lists elements of a discharge summary for patients with ACS (Table 4).21
| |
| Diagnoses | Elaborate on details of MI such as location, complications |
| Comorbidities | List, including diabetes, lipids, hypertension, renal disease |
| Medications | Note medication reconciliation, reason for not prescribing core measure medications, titration of any medications |
| Specific medications to address include ACE/ARB inhibitors, aspirin, beta blockers, statin, sublingual NTG, clopidogrel (include duration of therapy) | |
| Procedures | Type of stent (bare‐metal stent, drug‐eluting stent) and stent location |
| Complications (hematoma, transfusion) | |
| If ECHO, note type, ejection fraction; provide copy of ECG | |
| Follow‐up appointment(s) | Primary care, cardiology, others such as cardiac rehabilitation |
| Follow‐up testing | ETT (type, timeframe); ECHO if indicated; laboratory assessments |
| Code status | |
| Activity | |
| Diet | |
| Wound care (eg, groin) | |
| Treatment course | Address cognitive level, discharge LDL, discharge creatinine, INR if on warfarin, LFTs if on statin |
| Copy all providers | |
Staff and patient education is also an area that can benefit from evaluation. Clinicians from multiple disciplines are stakeholders in QI and they should receive education about its purpose and goals. Usually institutions will have a QI methodology in place, and this should be conveyed to the members of the ACS team. Staff education about ACS should be tailored to each specialty, be institution‐specific, current, and comprehensive, and include methods for assessing the learner. Education should be easily available (eg, on the Internet or via an electronic format), interactive, case‐based, and mandatory. For patients, education should be available in languages other than English and identify community resources and opportunities for additional outpatient education. The value of any educational program should be assessed to measure learner participation, satisfaction, and comprehension.
Gathering metrics and generating objective evidence of change is critical to QI; quantifying improvement (or lack thereof) must be done to determine whether the changes implemented improve care and ultimately whether desired outcomes are met. Metrics should be meaningful, associated with standards of care (eg, CMS core measures), and carefully chosen to reflect current practice. CMS core measures are a good target for collecting metrics to assess an institution's performance relative to ACS (Table 3). The Specifications Manual for National and Hospital Inpatient Quality Measures10 from TJC and CMS identifies the data elements needed for reporting. Other metrics not specified as core measures are worthy of measurement because they are considered the standard of care for patients with ACS (Table 5). The metrics shown in Tables 3 and 5 assume that all patients are eligible for core‐measure or standard‐of‐care medications and procedures. Because some measures are contraindicated in some patients, it is more meaningful to measure the percentage of patients without a contraindication who receive the measure. If a measure is contraindicated, the patient's medical record must include supporting documentation indicating why a core measure was not met. To be objective and reduce bias regarding the effectiveness of QI measures, data collection is best done prospectively. If necessary, periodic assessments against performance measures (institutional, government, professional association) should be made to support timely intervention. Run charts can be particularly useful here, measuring change over time to identify trends or an intervention that supported, did not affect, or was a barrier to the desired change.
Case Study (cont)
Scenario 1 (cont)
Mary tells the hospitalist she felt the discharge process was rushed and confusing and that she was dissatisfied with her care overall. Recalling a recent review article on the process for public disclosure of patient ratings related to hospital stay, the hospitalist locates HospitalCompare.com on the Internet. He reviews results from patient satisfaction surveys that compare his institution with others in the geographic area and is surprised to discover that patients generally give his institution a poor rating as well as low marks for the quality of nursing and physicianpatient communication. He is interested in this information but is not aware of resources for further exploration.
Scenario 2 (cont)
At discharge, the hospitalist confirms with Mary that she has received smoking cessation counseling during her hospital stay; he documents this in her chart and discharge summary. The hospitalist was aware of this particular quality measure because the recent weekly score card of hospital performance, posted in the unit, reported that the hospital was not meeting its goal of 100% compliance for this CMS core measure. Among heart attack patients who were identified as smokers, only 80% of charts documented that the patient had been counseled about smoking cessation during the hospital stay. Mary says that she understands the importance of not smoking and says she will make an effort to stop.
| Standard of Care Measure | Sample Metric |
|---|---|
| |
| LDL‐cholesterol assessment | Percentage of patients who have LDL cholesterol measured during hospitalization |
| Lipid‐lowering therapy at discharge | Percentage of patients prescribed a statin at hospital discharge |
| Dietary consultation | Percentage of patients who receive a dietary consult during hospitalization |
| Time to receipt of high‐risk abnormal laboratory assessments | Median time to receipt of high‐risk laboratory results, eg, troponins |
| Cardiac risk assessment | Percentage of patients who receive a cardiac risk assessment during admission |
| Measurement of LVEF | Percentage of patients who receive a cardiac echocardiogram to measure of LVEF before discharge |
| Document communication with PCP | Percentage of patients whose communication with the PCP was documented at discharge |
| Completed medication reconciliation | Percentage of patients for whom medication reconciliation was documented by the time of discharge |
| Make 1‐week follow‐up appointment with PCP | Percentage of patients for whom a 1‐week follow‐up appointment with the PCP was documented at the time of discharge |
| Additional Measure | |
| Inpatient mortality | |
Conclusion
Each hospitalist can have an impact on ACS care systemwide. Hospitalists are on the front line of care and have a unique perspective on patients as they are transitioned through the hospital stay and on how an institution handles patient care overall. They experience firsthand the challenges presented by poor communication between providers, patients, and their families. They can offer breadth of experience and perspective when assessing processes linked to patient care and can be instrumental in ensuring each patient experiences safe transitions during the hospital stay. Hospitalists should participate in QI initiatives for ACS and should consider opportunities to take the lead on these initiatives within their institutions.
Acknowledgements
The author thanks Denise Erkkila, RPh for her editorial assistance in the preparation of this manuscript.
- Hospitalists and intensivists: partners in caring for the critically ill—the time has come.J Hosp Med.2010;5:1–3.
- Society of Hospital Medicine. Acute coronary syndrome.J Hosp Med.2006;1(suppl 1):2–3.
- Society of Hospital Medicine. Quality improvement.J Hosp Med.2006;1 (suppl):92.
- ,,, et al.Curriculum development: the venous thromboembolism quality improvement resource room.J Hosp Med.2006;1:124–132.
- ,,,,Society of Hospital Medicine Glycemic Control Task Force summary: practical recommendations for assessing the impact of glycemic control efforts.J Hosp Med.2008;3:66–75.
- ,,,Effects of a subcutaneous insulin protocol, clinical education, and computerized order set on the quality of inpatient management of hyperglycemia: results of a clinical trial.J Hosp Med.2009;4:16–27.
- SHM Acute Coronary Syndrome Advisory Board. A guide for effective quality improvement: improving acute coronary syndrome care for hospitalized patients. Available at: http://www.hospitalmedicine.org. 2010. Accessed July 2010.
- ,,, et al.2009focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.year="2009"2009;54:2205–2241.
- The Joint Commission. Performance measure intiatives. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/Acute+Myocardial+Infarction+Core+Measure+Set.htm. 2010. Accessed July 2010.
- The Joint Commission, Centers for Medicare and Medicare Services. Specifications manual for national hospital inpatient quality measures, version 2.5. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/Current+NHQM+Manual.htm. 2009 November 6. Accessed July 2010.
- The Joint Commission. Improving America's hospitals: The Joint Commission's annual report on quality and safety 2009. Available at: http://www.jointcommission.org/Library/annual_report.2009. Accessed July 2010.
- Centers for Medicare and Medicare Services. Physician quality reporting initiative (PQRI). Available at: http://www.cms.hhs.gov/pqri. 2010. Accessed July 2010.
- Hospital Care Quality Information from the Consumer Perspective. CAHPS hospital survey. Available at: http://www.hcahpsonline.org. 2010. Accessed July 2010.
- ,,,.Discharge education improves clinical outcomes in patients with chronic heart failure.Circulation.2005;111:179–185.
- ,,, et al.National efforts to improve door‐to‐balloon time results from the Door‐to‐Balloon Alliance.J Am Coll Cardiol.2009;54:2423–2429.
- ,,, et al.Sustaining improvement in door‐to‐balloon time over 4 years: the Mayo clinic ST‐elevation myocardial infarction protocol.Circ Cardiovasc Qual Outcomes.2009;2:508–513.
- ,,,Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists.J Hosp Med.2007;2:314–323.
- ,.Lost in transition: challenges and opportunities for improving the quality of transitional care.Ann Intern Med.2004;141:533–536.
- Society of Hospital Medicine. Boosting Care Transitions Resource Room. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_CareTransitions/CT_Home.cfm. 2010. Accessed July 2010.
- SHM ACS Transitions Workgroup. SHM ACS Transitions Tool. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/05_Transitions.cfm. 2010. Accessed July 2010.
- ,,, et al.Transition of care for hospitalized elderly patients‐‐development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
Addressing quality improvement (QI) for the management of acute coronary syndrome (ACS) at the institutional level is essential for supporting hospitalists and other clinicians as they manage patients with ACS and achieve desired institutional outcomes. This systems approach may identify institution‐specific barriers to quality care, including those that affect the complex management of ACS. Having a firsthand view of patient care puts the hospitalist in a good position to assess the viability of existing processes and protocols that support care. Indeed, the hospitalist has a vested interest in improving systems of care as these systems directly affect the hospitalist's practice. This unique perspective gives the hospitalist an opportunity to facilitate systems change within the institution and to become an integral participant or leader in QI initiatives.
An increasing number of hospitalists are providing critical care at secondary and tertiary care facilities, driven by a shortage of intensivists in the United States. In a 20052006 survey about 75% of hospitalists reported they provide critical care services as part of their practice,1 and this would include care of patients with ACS. The Society of Hospital Medicine (SHM) has developed core competencies that recognize the important role of hospitalists in leading or participating in QI teams for ACS.2 Hospitalists must also be able to apply evidenced‐based outcomes data to support these initiatives.3 Hospitalist competencies for ACS include protocol development that supports the timely diagnosis and treatment of ACS, evaluation of resource utilization, staff education of secondary prevention measures, and implementation of measures to ensure institutional compliance with national quality standards (Table 1).2 Most hospital medicine groups will be expected to contribute to systems improvement, an area where hospitalists have already shown leadership for QI protocols related to glycemic control and venous thromboembolism prophylaxis. Hospitalists were prominent in targeting QI in these areas even though these QI initiatives could easily have been spearheaded by specialists.46
|
| Lead, coordinate, or participate in: |
| Protocol development to rapidly identify ACS, minimizing time to intervention |
| Protocol development for rapid identification and transfer of patients with ACS to a facility with an appropriate level of care |
| Multidisciplinary initiatives, including order sets for ACS and chest pain, that promote patient safety and optimize the use of resources |
| Staff education initiatives on the value of smoking cessation counseling and other prevention measures |
| Implement or integrate: |
| Systems to ensure hospitalwide adherence to national standards, documenting adherence as required by certifying organizations |
| Outcomes research, institution‐specific laboratory policies, and hospital formulary to create indicated and cost‐effective diagnostic and management strategies for patients with ACS |
QI Basics
QI at the institutional level addresses systems of care rather than individual performance, targeting both institutional performance and use of resources.3 QI is a continuous process in which practices and procedures related to patient management are regularly assessed to ascertain whether a quality gap exists. This in turn may lead to new processes, protocols, and algorithms that help the institution and clinicians meet benchmarks of quality care.
QI starts when an existing gap is recognizedthe gap between the scientific understanding of optimal care and actual patient care. The goal is to narrow or close this gap so that each patient receives optimal care. Underlying any QI initiative are 2 essential concepts. First, improvement requires systems changeany system will produce exactly what it is designed to produce. For example, if procedures are not in place to educate patients about smoking cessation while they are hospitalized, it is unlikely that the majority will routinely receive this counseling before they are discharged. Second, less is moreproductivity is not destroyed but often is enhanced by initiating simple and practical change. If a patient arrives in the emergency department (ED) with chest pain, instituting an order set that reminds clinicians to start antiplatelet therapy or a beta blocker can lessen the chance that these medications will be overlooked, especially when the patient transitions between providers or services within the hospital.
SHM has identified 7 essential elements of any QI initiative, and these are applicable to the care of the ACS patient (Table 2).7 These elements highlight the need for institutional support and teamwork that support standardized measures and tools specific to issues in ACS management. These issues include: (1) rapidly identifying a patient with ACS and initiating a care plan when the patient is admitted; (2) encouraging good communication between providers; (3) symptom management; (4) medication safety, polypharmacy, and medication reconciliation; (5) patient and caregiver education; (6) safe discharge and transitions in care; and (7) meeting Centers for Medicare and Medicaid Services (CMS) core measures.
| |
| Institutional support | Commitment of time, personnel, and tools to support the initiative |
| Multidisciplinary team | Team that focuses on quality of care for patients with ACS |
| Reliable metrics | Reflect CMS core and applicable PQRI measures; also reportable to inform team decision making |
| Identify the goal | Establish a measurable, achievable goal with an established timeline |
| Standard order sets | Defined clinical pathways that support evidence‐based treatment strategies, risk stratification, and safe transitions in care |
| Policies that support algorithms and protocols | Institution‐specific to support order sets |
| Education programs | Targeted to clinicians and patients; should cover items addressed in order sets, algorithms, and protocols |
Tools such as process flow mapping and run charts can reveal quality gaps and indicate if process improvements are leading to stated objectives. Process flow mapping makes it possible to identify and visualize quality gaps that might otherwise be hidden and to identify their source or cause. Process mapping documents discrete steps within the flow and usually requires input from multiple disciplines; this information can guide a multidisciplinary QI team when formulating interventions for process improvement.7 Figure 1 shows process flow mapping for the early identification of ST‐segment elevation myocardial infarction (STEMI) when hospitalists have open access to activate the catheterization laboratory (cath lab). The time from door‐to‐balloon is a critical factor in reducing STEMI‐related morbidity8; processes that overcome delays to percutaneous coronary intervention (PCI) and improve communication can have a direct benefit on patient outcomes.

A run chart is a commonly used tool which graphically depicts progress in attaining a goal over time, before and after an intervention. Figure 2a is a run chart that shows the average time to PCI following implementation of an order set designed to support the use of a risk stratification tool for the early diagnosis of STEMI. In this case, the average time to PCI is observed to decrease over time, but still lags behind the desired goal of less than 90 minutes. This run chart indicates that further work is needed to improve the process of identifying patients with STEMI. Figure 2b shows the number of eligible STEMI patients who received aspirin at discharge following implementation of a discharge order set; here, the hospital has clearly made progress toward meeting this CMS core measure.

The following is a case study that illustrates how the implementation of standardized measures and tools can help hospitalists and other clinicians achieve quality measures in the care of a patient with ACS.
Case Study
Mary, a 68‐year‐old woman, presents to the ED with fatigue and some heartburn. She has a history of hypertension and may have had a mini stroke a few years ago. Her symptoms, which she has had on and off for the past few days, worsened considerably right before her arrival. She has been taking chewable antacids but is unsure if they have helped. Her physical exam is unremarkable. A stat electrocardiogram (ECG) shows inferior ST (part of an electrocardiogram between the QRS complex and the T wave) elevation in leads II and III, and augmented vector foot (aVF). Troponins are positive. This case can be approached from 2 perspectives.
Scenario 1
Mary arrives at a hospital that has few standardized systems or protocols for triage and transitions in care. The triage nurse recognizes the need for and obtains an ECG, but fails to alert the ED physician. When the ED physician eventually sees the patient and reviews the ECG, she immediately administers nitroglycerin and pages the cardiologist on call and the hospitalist. The hospitalist arrives first and recognizes the STEMI, but hesitates to start unfractionated or low molecular weight heparin until the cardiologist determines whether the patient will undergo PCI. The cardiologist orders PCI, the cath lab is alerted, and the patient is started on the appropriate medications; the patient does not start an aspirin until after PCI. In this scenario, several short delays amount to a significant delay of about 2 hours before the patient reaches the cath lab. In addition, although aspirin is given on day 1, it is not done on arrival. Following PCI, the patient is transferred to the coronary care unit (CCU).
Scenario 2
Mary arrives at an institution with well‐defined, institution‐specific protocols for triage and transitions in care. The triage nurse recognizes the need for and obtains an ECG; the results are immediately reviewed with the ED physician. STEMI is diagnosed and the cath lab protocol is activated; the patient receives aspirin as part of a standing order. The hospitalist meets the patient in the ED within 5 minutes and begins the protocol for unfractionated heparin and preparation for immediate PCI, allowing the ED physician to return to ED care. Simultaneously, the interventional cardiologist and cath lab are mobilized and the patient is transferred within 15 minutes. Additional appropriate medications are begun. The door‐to‐balloon time is 60 minutes, well within recommended timeframes. Following PCI, the patient is transferred to the CCU.
Measuring Performance Relative to ACS
Two agencies promulgating quality measures for hospital inpatient care are The Joint Commission (TJC) and the CMS. TJC and CMS jointly established core measures for patients with acute myocardial infarction (AMI) and these are considered key indicators of quality, evidence‐based care. As outlined in Table 3, the TJC/CMS recommendations promote, unless contraindicated, a minimum standard of care for patients with AMI.9, 10 Public reporting of institutional core measure performance has led to an intense focus on improving these metrics.
| Core Measure | Sample Metric |
|---|---|
| |
| Aspirin at arrival | Percentage of patients receiving aspirin within 24 hours before or after hospital arrival |
| Aspirin at discharge | Percentage of patients prescribed aspirin at hospital discharge |
| ACE inhibitor or ARB for LVSD | Percentage of patients with LVSD prescribed an ACEI or ARB at hospital discharge |
| Beta blocker | Percentage of patients prescribed a beta blocker at hospital discharge |
| Fibrinolytic medication given within 30 minutes of hospital arrival | Median time to fibrinolysis |
| Percentage of patients given fibrinolytic medication within 30 minutes of hospital arrival | |
| PCI received within 90 minutes of hospital arrival | Median time to PCI |
| Percentage of patients receiving primary PCI within 90 minutes of hospital arrival | |
| Smoking cessation counseling | Percentage of patients with a history of smoking cigarettes who are counseled about smoking cessation during hospitalization |
On a national level, TJC documented performance improvement from 2002 to 2008 for each of the core measures. Compliance with smoking cessation counseling improved the most, rising from 67% to almost 99%. In 2008, a combined measure of all individual core measures indicated that, overall, care of heart attack patients is of high quality. Hospitals delivered evidenced‐based care in 96.7% of the opportunities they had to provide this care. Compliance related to oral medications was also good (95%); however, a closer look at other individual measures shows that improvement is needed to reduce the time to fibrinolysis (52.4% compliance rate for 30 minutes or less) and time to primary PCI (81.6% compliance rate for 90 minutes or less).11
Financial incentives are tied to QI measures including those from CMS. At present, this system is in the pay‐for‐reporting phase, in which institutions are penalized for not reporting quality metrics for the CMS core measures. It is likely that, to further incentivize institutions to meet quality benchmarks, this will be expanded to a pay‐for‐performance system (eg, differential payments for readmissions or different payment scales based on prior performance). Public disclosure of institutional performance relative to ACS and other medical conditions is available through the HHS.gov website (www.hospitalcompare.hhs.gov), which compares performance between hospitals and provides a clear business motivation for institutions to improve and provide high quality care.
Two other reporting systems should be noted. The CMS Physician Quality Reporting Initiative (PQRI) includes physician‐related quality measures specific to ACS12 that overlap with institution‐level CMS core measures with regard to prescribing ACS medications. Payments associated with the PQRI are currently a small financial factor for hospitalists, but will likely grow as quality initiatives develop overall. The Hospital Care Quality Information From the Consumer Perspective (HCAHPS) initiative, also from CMS, strives to capture patients' perspectives on hospital care through a standardized survey. The goals are to collect data that can be used to compare hospital performance, create an incentive for QI through public disclosure of results, and increase transparency regarding the quality of hospital care.13 However, the focus is not ACS‐specific. Patients are queried about communication with hospital staff, communication about medications, and information about discharge, all areas of concern for the hospitalist in general that have been identified as areas for improvement relative to ACS.
Case Study (cont)
Scenario 1 (cont)
Mary recuperates in the CCU and is transferred to the medical floor. The hospitalist and cardiologist see her separately on rounds and each assumes the other has a reason for not starting a beta blocker; therefore, a beta blocker is not prescribed. The nurse cannot tell who is in charge and does not anticipate the day of dischargeshe is just implementing the orders as they are written. The day of discharge arrives; the nurse learns from Mary that the hospitalist will discharge her that day. The hospitalist reviews the list of ACS medications and realizes that Mary still has not started a beta blocker. He cannot reach the cardiologist before Mary's ride home arrives, so he writes a note in the discharge summary alerting the primary care physician (PCP) to consider a beta blocker at follow‐up. Because of this lack of communication and systems for tracking the implementation of guideline‐recommended therapies, Mary is discharged without a CMS core measure medication, with no assurance that this will be addressed by the PCP.
Scenario 2 (cont)
Mary recuperates in the CCU and is transferred to the medical floor. Standard post‐PCI/STEMI orders are in place according to institutional protocol. The hospitalist is able to confirm that all interventions required at admission (aspirin, beta blocker, assessment of smoking status) have occurred by reviewing a well‐structured checklist that includes easy‐to‐read visual cues. The checklist indicates that Mary was counseled about smoking cessation on day 1 of her stay in the CCU. Mary and her physicians and nurses are all aware of the target discharge date and the milestones that must be met prior to discharge (eg, echocardiogram, medication review, education, assessment of mobility, etc). Mary is instructed about each new medication and given educational materials.14 Follow‐up appointments postdischarge are made, and the discharge summary is sent electronically to the PCP. These institution‐specific protocols enhance communication overall and help the hospital meet high standards of patient care.
Special QI Issues in ACS Management for the Hospitalist
Coordination is especially important for patients with ACS because their care is so complex. Seamless transitions promote safe care as the patient moves from the ED, cath lab, recovery unit, medical floor, and discharge through the transition to primary care. Poor communication between clinicians during these transitions may result in delayed or overlooked treatment and other medical errors. Implementing an institutional system of care may overcome communication barriers and help ensure the institution meets its quality benchmarks, such as the CMS core measures. Standard order sets and protocols identify the steps and components needed to manage ACS. At admission, these measures promote early assessment of patient risk, triage to early intervention, medication reconciliation, and communication between stakeholders such as the hospitalist, cardiologist, and the cath lab.15, 16 During hospitalization, they help assure ongoing risk assessment and early consideration of discharge planning, culminating in discharge and the transition back to primary care.
A recent guideline update8 focused attention on the coordination of care between institutions and the critical importance of rapid triage for patients who need primary PCI and transfer from a non‐PCI to a PCI‐capable institution. The decision to transfer rests on multiple factors and requires rapid decision making on the part of clinicians. Time to reperfusion is shown to have a direct effect on patient outcomes. Established protocols within a non‐PCI facility can support timely transfer to a PCI‐capable facility if indicated. Factors such as the patient's mortality risk, the risk of bleeding from fibrinolytic therapy, duration of symptoms, and the time for transport to a PCI facility all must be considered. It is recognized that a regional system of STEMI care best supports collaborative efforts between institutions and community resources that support QI efforts.
Transitions in care, and particularly discharge, are areas with identified gaps in quality care17, 18 for which improvement has been pursued on an institutional level. Project BOOST (Better Outcomes for Older Adults Through Safe Transitions) seeks to improve the care of patients during the transition from inpatient to outpatient care, and focuses on elderly patients identified to be at high‐risk for adverse events during this transition.19 The goal is to improve outcomes related to 30‐day readmission rates, patient satisfaction, communication between inpatient and outpatient providers, identification of high‐risk patients who need intervention to reduce risk, and patient education about their risk for an adverse event. BOOST addresses these outcomes at an institutional level by offering resources related to project management, data collection, tools for clinicians and patients, and participating mentor institutions. These resources help an institution assess its readiness for change, identify quality gaps, promote teamwork, and guide the implementation and subsequent evaluation of process improvements. Specific tools for clinicians also support institutional goals for teamwork and communication, creating an environment for safe transitions. Both BOOST19 and the adaptable Transitions Tool from the SHM20 provide a framework for understanding processes that involve multiple departments and stakeholders, breaking complex processes into discrete parts for which quality gaps can be identified and change instituted to improve care. A checklist can also be a useful tool for ensuring specific issues are addressed during transitions in care. SHM developed a checklist for hospitalist use that lists elements of a discharge summary for patients with ACS (Table 4).21
| |
| Diagnoses | Elaborate on details of MI such as location, complications |
| Comorbidities | List, including diabetes, lipids, hypertension, renal disease |
| Medications | Note medication reconciliation, reason for not prescribing core measure medications, titration of any medications |
| Specific medications to address include ACE/ARB inhibitors, aspirin, beta blockers, statin, sublingual NTG, clopidogrel (include duration of therapy) | |
| Procedures | Type of stent (bare‐metal stent, drug‐eluting stent) and stent location |
| Complications (hematoma, transfusion) | |
| If ECHO, note type, ejection fraction; provide copy of ECG | |
| Follow‐up appointment(s) | Primary care, cardiology, others such as cardiac rehabilitation |
| Follow‐up testing | ETT (type, timeframe); ECHO if indicated; laboratory assessments |
| Code status | |
| Activity | |
| Diet | |
| Wound care (eg, groin) | |
| Treatment course | Address cognitive level, discharge LDL, discharge creatinine, INR if on warfarin, LFTs if on statin |
| Copy all providers | |
Staff and patient education is also an area that can benefit from evaluation. Clinicians from multiple disciplines are stakeholders in QI and they should receive education about its purpose and goals. Usually institutions will have a QI methodology in place, and this should be conveyed to the members of the ACS team. Staff education about ACS should be tailored to each specialty, be institution‐specific, current, and comprehensive, and include methods for assessing the learner. Education should be easily available (eg, on the Internet or via an electronic format), interactive, case‐based, and mandatory. For patients, education should be available in languages other than English and identify community resources and opportunities for additional outpatient education. The value of any educational program should be assessed to measure learner participation, satisfaction, and comprehension.
Gathering metrics and generating objective evidence of change is critical to QI; quantifying improvement (or lack thereof) must be done to determine whether the changes implemented improve care and ultimately whether desired outcomes are met. Metrics should be meaningful, associated with standards of care (eg, CMS core measures), and carefully chosen to reflect current practice. CMS core measures are a good target for collecting metrics to assess an institution's performance relative to ACS (Table 3). The Specifications Manual for National and Hospital Inpatient Quality Measures10 from TJC and CMS identifies the data elements needed for reporting. Other metrics not specified as core measures are worthy of measurement because they are considered the standard of care for patients with ACS (Table 5). The metrics shown in Tables 3 and 5 assume that all patients are eligible for core‐measure or standard‐of‐care medications and procedures. Because some measures are contraindicated in some patients, it is more meaningful to measure the percentage of patients without a contraindication who receive the measure. If a measure is contraindicated, the patient's medical record must include supporting documentation indicating why a core measure was not met. To be objective and reduce bias regarding the effectiveness of QI measures, data collection is best done prospectively. If necessary, periodic assessments against performance measures (institutional, government, professional association) should be made to support timely intervention. Run charts can be particularly useful here, measuring change over time to identify trends or an intervention that supported, did not affect, or was a barrier to the desired change.
Case Study (cont)
Scenario 1 (cont)
Mary tells the hospitalist she felt the discharge process was rushed and confusing and that she was dissatisfied with her care overall. Recalling a recent review article on the process for public disclosure of patient ratings related to hospital stay, the hospitalist locates HospitalCompare.com on the Internet. He reviews results from patient satisfaction surveys that compare his institution with others in the geographic area and is surprised to discover that patients generally give his institution a poor rating as well as low marks for the quality of nursing and physicianpatient communication. He is interested in this information but is not aware of resources for further exploration.
Scenario 2 (cont)
At discharge, the hospitalist confirms with Mary that she has received smoking cessation counseling during her hospital stay; he documents this in her chart and discharge summary. The hospitalist was aware of this particular quality measure because the recent weekly score card of hospital performance, posted in the unit, reported that the hospital was not meeting its goal of 100% compliance for this CMS core measure. Among heart attack patients who were identified as smokers, only 80% of charts documented that the patient had been counseled about smoking cessation during the hospital stay. Mary says that she understands the importance of not smoking and says she will make an effort to stop.
| Standard of Care Measure | Sample Metric |
|---|---|
| |
| LDL‐cholesterol assessment | Percentage of patients who have LDL cholesterol measured during hospitalization |
| Lipid‐lowering therapy at discharge | Percentage of patients prescribed a statin at hospital discharge |
| Dietary consultation | Percentage of patients who receive a dietary consult during hospitalization |
| Time to receipt of high‐risk abnormal laboratory assessments | Median time to receipt of high‐risk laboratory results, eg, troponins |
| Cardiac risk assessment | Percentage of patients who receive a cardiac risk assessment during admission |
| Measurement of LVEF | Percentage of patients who receive a cardiac echocardiogram to measure of LVEF before discharge |
| Document communication with PCP | Percentage of patients whose communication with the PCP was documented at discharge |
| Completed medication reconciliation | Percentage of patients for whom medication reconciliation was documented by the time of discharge |
| Make 1‐week follow‐up appointment with PCP | Percentage of patients for whom a 1‐week follow‐up appointment with the PCP was documented at the time of discharge |
| Additional Measure | |
| Inpatient mortality | |
Conclusion
Each hospitalist can have an impact on ACS care systemwide. Hospitalists are on the front line of care and have a unique perspective on patients as they are transitioned through the hospital stay and on how an institution handles patient care overall. They experience firsthand the challenges presented by poor communication between providers, patients, and their families. They can offer breadth of experience and perspective when assessing processes linked to patient care and can be instrumental in ensuring each patient experiences safe transitions during the hospital stay. Hospitalists should participate in QI initiatives for ACS and should consider opportunities to take the lead on these initiatives within their institutions.
Acknowledgements
The author thanks Denise Erkkila, RPh for her editorial assistance in the preparation of this manuscript.
Addressing quality improvement (QI) for the management of acute coronary syndrome (ACS) at the institutional level is essential for supporting hospitalists and other clinicians as they manage patients with ACS and achieve desired institutional outcomes. This systems approach may identify institution‐specific barriers to quality care, including those that affect the complex management of ACS. Having a firsthand view of patient care puts the hospitalist in a good position to assess the viability of existing processes and protocols that support care. Indeed, the hospitalist has a vested interest in improving systems of care as these systems directly affect the hospitalist's practice. This unique perspective gives the hospitalist an opportunity to facilitate systems change within the institution and to become an integral participant or leader in QI initiatives.
An increasing number of hospitalists are providing critical care at secondary and tertiary care facilities, driven by a shortage of intensivists in the United States. In a 20052006 survey about 75% of hospitalists reported they provide critical care services as part of their practice,1 and this would include care of patients with ACS. The Society of Hospital Medicine (SHM) has developed core competencies that recognize the important role of hospitalists in leading or participating in QI teams for ACS.2 Hospitalists must also be able to apply evidenced‐based outcomes data to support these initiatives.3 Hospitalist competencies for ACS include protocol development that supports the timely diagnosis and treatment of ACS, evaluation of resource utilization, staff education of secondary prevention measures, and implementation of measures to ensure institutional compliance with national quality standards (Table 1).2 Most hospital medicine groups will be expected to contribute to systems improvement, an area where hospitalists have already shown leadership for QI protocols related to glycemic control and venous thromboembolism prophylaxis. Hospitalists were prominent in targeting QI in these areas even though these QI initiatives could easily have been spearheaded by specialists.46
|
| Lead, coordinate, or participate in: |
| Protocol development to rapidly identify ACS, minimizing time to intervention |
| Protocol development for rapid identification and transfer of patients with ACS to a facility with an appropriate level of care |
| Multidisciplinary initiatives, including order sets for ACS and chest pain, that promote patient safety and optimize the use of resources |
| Staff education initiatives on the value of smoking cessation counseling and other prevention measures |
| Implement or integrate: |
| Systems to ensure hospitalwide adherence to national standards, documenting adherence as required by certifying organizations |
| Outcomes research, institution‐specific laboratory policies, and hospital formulary to create indicated and cost‐effective diagnostic and management strategies for patients with ACS |
QI Basics
QI at the institutional level addresses systems of care rather than individual performance, targeting both institutional performance and use of resources.3 QI is a continuous process in which practices and procedures related to patient management are regularly assessed to ascertain whether a quality gap exists. This in turn may lead to new processes, protocols, and algorithms that help the institution and clinicians meet benchmarks of quality care.
QI starts when an existing gap is recognizedthe gap between the scientific understanding of optimal care and actual patient care. The goal is to narrow or close this gap so that each patient receives optimal care. Underlying any QI initiative are 2 essential concepts. First, improvement requires systems changeany system will produce exactly what it is designed to produce. For example, if procedures are not in place to educate patients about smoking cessation while they are hospitalized, it is unlikely that the majority will routinely receive this counseling before they are discharged. Second, less is moreproductivity is not destroyed but often is enhanced by initiating simple and practical change. If a patient arrives in the emergency department (ED) with chest pain, instituting an order set that reminds clinicians to start antiplatelet therapy or a beta blocker can lessen the chance that these medications will be overlooked, especially when the patient transitions between providers or services within the hospital.
SHM has identified 7 essential elements of any QI initiative, and these are applicable to the care of the ACS patient (Table 2).7 These elements highlight the need for institutional support and teamwork that support standardized measures and tools specific to issues in ACS management. These issues include: (1) rapidly identifying a patient with ACS and initiating a care plan when the patient is admitted; (2) encouraging good communication between providers; (3) symptom management; (4) medication safety, polypharmacy, and medication reconciliation; (5) patient and caregiver education; (6) safe discharge and transitions in care; and (7) meeting Centers for Medicare and Medicaid Services (CMS) core measures.
| |
| Institutional support | Commitment of time, personnel, and tools to support the initiative |
| Multidisciplinary team | Team that focuses on quality of care for patients with ACS |
| Reliable metrics | Reflect CMS core and applicable PQRI measures; also reportable to inform team decision making |
| Identify the goal | Establish a measurable, achievable goal with an established timeline |
| Standard order sets | Defined clinical pathways that support evidence‐based treatment strategies, risk stratification, and safe transitions in care |
| Policies that support algorithms and protocols | Institution‐specific to support order sets |
| Education programs | Targeted to clinicians and patients; should cover items addressed in order sets, algorithms, and protocols |
Tools such as process flow mapping and run charts can reveal quality gaps and indicate if process improvements are leading to stated objectives. Process flow mapping makes it possible to identify and visualize quality gaps that might otherwise be hidden and to identify their source or cause. Process mapping documents discrete steps within the flow and usually requires input from multiple disciplines; this information can guide a multidisciplinary QI team when formulating interventions for process improvement.7 Figure 1 shows process flow mapping for the early identification of ST‐segment elevation myocardial infarction (STEMI) when hospitalists have open access to activate the catheterization laboratory (cath lab). The time from door‐to‐balloon is a critical factor in reducing STEMI‐related morbidity8; processes that overcome delays to percutaneous coronary intervention (PCI) and improve communication can have a direct benefit on patient outcomes.

A run chart is a commonly used tool which graphically depicts progress in attaining a goal over time, before and after an intervention. Figure 2a is a run chart that shows the average time to PCI following implementation of an order set designed to support the use of a risk stratification tool for the early diagnosis of STEMI. In this case, the average time to PCI is observed to decrease over time, but still lags behind the desired goal of less than 90 minutes. This run chart indicates that further work is needed to improve the process of identifying patients with STEMI. Figure 2b shows the number of eligible STEMI patients who received aspirin at discharge following implementation of a discharge order set; here, the hospital has clearly made progress toward meeting this CMS core measure.

The following is a case study that illustrates how the implementation of standardized measures and tools can help hospitalists and other clinicians achieve quality measures in the care of a patient with ACS.
Case Study
Mary, a 68‐year‐old woman, presents to the ED with fatigue and some heartburn. She has a history of hypertension and may have had a mini stroke a few years ago. Her symptoms, which she has had on and off for the past few days, worsened considerably right before her arrival. She has been taking chewable antacids but is unsure if they have helped. Her physical exam is unremarkable. A stat electrocardiogram (ECG) shows inferior ST (part of an electrocardiogram between the QRS complex and the T wave) elevation in leads II and III, and augmented vector foot (aVF). Troponins are positive. This case can be approached from 2 perspectives.
Scenario 1
Mary arrives at a hospital that has few standardized systems or protocols for triage and transitions in care. The triage nurse recognizes the need for and obtains an ECG, but fails to alert the ED physician. When the ED physician eventually sees the patient and reviews the ECG, she immediately administers nitroglycerin and pages the cardiologist on call and the hospitalist. The hospitalist arrives first and recognizes the STEMI, but hesitates to start unfractionated or low molecular weight heparin until the cardiologist determines whether the patient will undergo PCI. The cardiologist orders PCI, the cath lab is alerted, and the patient is started on the appropriate medications; the patient does not start an aspirin until after PCI. In this scenario, several short delays amount to a significant delay of about 2 hours before the patient reaches the cath lab. In addition, although aspirin is given on day 1, it is not done on arrival. Following PCI, the patient is transferred to the coronary care unit (CCU).
Scenario 2
Mary arrives at an institution with well‐defined, institution‐specific protocols for triage and transitions in care. The triage nurse recognizes the need for and obtains an ECG; the results are immediately reviewed with the ED physician. STEMI is diagnosed and the cath lab protocol is activated; the patient receives aspirin as part of a standing order. The hospitalist meets the patient in the ED within 5 minutes and begins the protocol for unfractionated heparin and preparation for immediate PCI, allowing the ED physician to return to ED care. Simultaneously, the interventional cardiologist and cath lab are mobilized and the patient is transferred within 15 minutes. Additional appropriate medications are begun. The door‐to‐balloon time is 60 minutes, well within recommended timeframes. Following PCI, the patient is transferred to the CCU.
Measuring Performance Relative to ACS
Two agencies promulgating quality measures for hospital inpatient care are The Joint Commission (TJC) and the CMS. TJC and CMS jointly established core measures for patients with acute myocardial infarction (AMI) and these are considered key indicators of quality, evidence‐based care. As outlined in Table 3, the TJC/CMS recommendations promote, unless contraindicated, a minimum standard of care for patients with AMI.9, 10 Public reporting of institutional core measure performance has led to an intense focus on improving these metrics.
| Core Measure | Sample Metric |
|---|---|
| |
| Aspirin at arrival | Percentage of patients receiving aspirin within 24 hours before or after hospital arrival |
| Aspirin at discharge | Percentage of patients prescribed aspirin at hospital discharge |
| ACE inhibitor or ARB for LVSD | Percentage of patients with LVSD prescribed an ACEI or ARB at hospital discharge |
| Beta blocker | Percentage of patients prescribed a beta blocker at hospital discharge |
| Fibrinolytic medication given within 30 minutes of hospital arrival | Median time to fibrinolysis |
| Percentage of patients given fibrinolytic medication within 30 minutes of hospital arrival | |
| PCI received within 90 minutes of hospital arrival | Median time to PCI |
| Percentage of patients receiving primary PCI within 90 minutes of hospital arrival | |
| Smoking cessation counseling | Percentage of patients with a history of smoking cigarettes who are counseled about smoking cessation during hospitalization |
On a national level, TJC documented performance improvement from 2002 to 2008 for each of the core measures. Compliance with smoking cessation counseling improved the most, rising from 67% to almost 99%. In 2008, a combined measure of all individual core measures indicated that, overall, care of heart attack patients is of high quality. Hospitals delivered evidenced‐based care in 96.7% of the opportunities they had to provide this care. Compliance related to oral medications was also good (95%); however, a closer look at other individual measures shows that improvement is needed to reduce the time to fibrinolysis (52.4% compliance rate for 30 minutes or less) and time to primary PCI (81.6% compliance rate for 90 minutes or less).11
Financial incentives are tied to QI measures including those from CMS. At present, this system is in the pay‐for‐reporting phase, in which institutions are penalized for not reporting quality metrics for the CMS core measures. It is likely that, to further incentivize institutions to meet quality benchmarks, this will be expanded to a pay‐for‐performance system (eg, differential payments for readmissions or different payment scales based on prior performance). Public disclosure of institutional performance relative to ACS and other medical conditions is available through the HHS.gov website (www.hospitalcompare.hhs.gov), which compares performance between hospitals and provides a clear business motivation for institutions to improve and provide high quality care.
Two other reporting systems should be noted. The CMS Physician Quality Reporting Initiative (PQRI) includes physician‐related quality measures specific to ACS12 that overlap with institution‐level CMS core measures with regard to prescribing ACS medications. Payments associated with the PQRI are currently a small financial factor for hospitalists, but will likely grow as quality initiatives develop overall. The Hospital Care Quality Information From the Consumer Perspective (HCAHPS) initiative, also from CMS, strives to capture patients' perspectives on hospital care through a standardized survey. The goals are to collect data that can be used to compare hospital performance, create an incentive for QI through public disclosure of results, and increase transparency regarding the quality of hospital care.13 However, the focus is not ACS‐specific. Patients are queried about communication with hospital staff, communication about medications, and information about discharge, all areas of concern for the hospitalist in general that have been identified as areas for improvement relative to ACS.
Case Study (cont)
Scenario 1 (cont)
Mary recuperates in the CCU and is transferred to the medical floor. The hospitalist and cardiologist see her separately on rounds and each assumes the other has a reason for not starting a beta blocker; therefore, a beta blocker is not prescribed. The nurse cannot tell who is in charge and does not anticipate the day of dischargeshe is just implementing the orders as they are written. The day of discharge arrives; the nurse learns from Mary that the hospitalist will discharge her that day. The hospitalist reviews the list of ACS medications and realizes that Mary still has not started a beta blocker. He cannot reach the cardiologist before Mary's ride home arrives, so he writes a note in the discharge summary alerting the primary care physician (PCP) to consider a beta blocker at follow‐up. Because of this lack of communication and systems for tracking the implementation of guideline‐recommended therapies, Mary is discharged without a CMS core measure medication, with no assurance that this will be addressed by the PCP.
Scenario 2 (cont)
Mary recuperates in the CCU and is transferred to the medical floor. Standard post‐PCI/STEMI orders are in place according to institutional protocol. The hospitalist is able to confirm that all interventions required at admission (aspirin, beta blocker, assessment of smoking status) have occurred by reviewing a well‐structured checklist that includes easy‐to‐read visual cues. The checklist indicates that Mary was counseled about smoking cessation on day 1 of her stay in the CCU. Mary and her physicians and nurses are all aware of the target discharge date and the milestones that must be met prior to discharge (eg, echocardiogram, medication review, education, assessment of mobility, etc). Mary is instructed about each new medication and given educational materials.14 Follow‐up appointments postdischarge are made, and the discharge summary is sent electronically to the PCP. These institution‐specific protocols enhance communication overall and help the hospital meet high standards of patient care.
Special QI Issues in ACS Management for the Hospitalist
Coordination is especially important for patients with ACS because their care is so complex. Seamless transitions promote safe care as the patient moves from the ED, cath lab, recovery unit, medical floor, and discharge through the transition to primary care. Poor communication between clinicians during these transitions may result in delayed or overlooked treatment and other medical errors. Implementing an institutional system of care may overcome communication barriers and help ensure the institution meets its quality benchmarks, such as the CMS core measures. Standard order sets and protocols identify the steps and components needed to manage ACS. At admission, these measures promote early assessment of patient risk, triage to early intervention, medication reconciliation, and communication between stakeholders such as the hospitalist, cardiologist, and the cath lab.15, 16 During hospitalization, they help assure ongoing risk assessment and early consideration of discharge planning, culminating in discharge and the transition back to primary care.
A recent guideline update8 focused attention on the coordination of care between institutions and the critical importance of rapid triage for patients who need primary PCI and transfer from a non‐PCI to a PCI‐capable institution. The decision to transfer rests on multiple factors and requires rapid decision making on the part of clinicians. Time to reperfusion is shown to have a direct effect on patient outcomes. Established protocols within a non‐PCI facility can support timely transfer to a PCI‐capable facility if indicated. Factors such as the patient's mortality risk, the risk of bleeding from fibrinolytic therapy, duration of symptoms, and the time for transport to a PCI facility all must be considered. It is recognized that a regional system of STEMI care best supports collaborative efforts between institutions and community resources that support QI efforts.
Transitions in care, and particularly discharge, are areas with identified gaps in quality care17, 18 for which improvement has been pursued on an institutional level. Project BOOST (Better Outcomes for Older Adults Through Safe Transitions) seeks to improve the care of patients during the transition from inpatient to outpatient care, and focuses on elderly patients identified to be at high‐risk for adverse events during this transition.19 The goal is to improve outcomes related to 30‐day readmission rates, patient satisfaction, communication between inpatient and outpatient providers, identification of high‐risk patients who need intervention to reduce risk, and patient education about their risk for an adverse event. BOOST addresses these outcomes at an institutional level by offering resources related to project management, data collection, tools for clinicians and patients, and participating mentor institutions. These resources help an institution assess its readiness for change, identify quality gaps, promote teamwork, and guide the implementation and subsequent evaluation of process improvements. Specific tools for clinicians also support institutional goals for teamwork and communication, creating an environment for safe transitions. Both BOOST19 and the adaptable Transitions Tool from the SHM20 provide a framework for understanding processes that involve multiple departments and stakeholders, breaking complex processes into discrete parts for which quality gaps can be identified and change instituted to improve care. A checklist can also be a useful tool for ensuring specific issues are addressed during transitions in care. SHM developed a checklist for hospitalist use that lists elements of a discharge summary for patients with ACS (Table 4).21
| |
| Diagnoses | Elaborate on details of MI such as location, complications |
| Comorbidities | List, including diabetes, lipids, hypertension, renal disease |
| Medications | Note medication reconciliation, reason for not prescribing core measure medications, titration of any medications |
| Specific medications to address include ACE/ARB inhibitors, aspirin, beta blockers, statin, sublingual NTG, clopidogrel (include duration of therapy) | |
| Procedures | Type of stent (bare‐metal stent, drug‐eluting stent) and stent location |
| Complications (hematoma, transfusion) | |
| If ECHO, note type, ejection fraction; provide copy of ECG | |
| Follow‐up appointment(s) | Primary care, cardiology, others such as cardiac rehabilitation |
| Follow‐up testing | ETT (type, timeframe); ECHO if indicated; laboratory assessments |
| Code status | |
| Activity | |
| Diet | |
| Wound care (eg, groin) | |
| Treatment course | Address cognitive level, discharge LDL, discharge creatinine, INR if on warfarin, LFTs if on statin |
| Copy all providers | |
Staff and patient education is also an area that can benefit from evaluation. Clinicians from multiple disciplines are stakeholders in QI and they should receive education about its purpose and goals. Usually institutions will have a QI methodology in place, and this should be conveyed to the members of the ACS team. Staff education about ACS should be tailored to each specialty, be institution‐specific, current, and comprehensive, and include methods for assessing the learner. Education should be easily available (eg, on the Internet or via an electronic format), interactive, case‐based, and mandatory. For patients, education should be available in languages other than English and identify community resources and opportunities for additional outpatient education. The value of any educational program should be assessed to measure learner participation, satisfaction, and comprehension.
Gathering metrics and generating objective evidence of change is critical to QI; quantifying improvement (or lack thereof) must be done to determine whether the changes implemented improve care and ultimately whether desired outcomes are met. Metrics should be meaningful, associated with standards of care (eg, CMS core measures), and carefully chosen to reflect current practice. CMS core measures are a good target for collecting metrics to assess an institution's performance relative to ACS (Table 3). The Specifications Manual for National and Hospital Inpatient Quality Measures10 from TJC and CMS identifies the data elements needed for reporting. Other metrics not specified as core measures are worthy of measurement because they are considered the standard of care for patients with ACS (Table 5). The metrics shown in Tables 3 and 5 assume that all patients are eligible for core‐measure or standard‐of‐care medications and procedures. Because some measures are contraindicated in some patients, it is more meaningful to measure the percentage of patients without a contraindication who receive the measure. If a measure is contraindicated, the patient's medical record must include supporting documentation indicating why a core measure was not met. To be objective and reduce bias regarding the effectiveness of QI measures, data collection is best done prospectively. If necessary, periodic assessments against performance measures (institutional, government, professional association) should be made to support timely intervention. Run charts can be particularly useful here, measuring change over time to identify trends or an intervention that supported, did not affect, or was a barrier to the desired change.
Case Study (cont)
Scenario 1 (cont)
Mary tells the hospitalist she felt the discharge process was rushed and confusing and that she was dissatisfied with her care overall. Recalling a recent review article on the process for public disclosure of patient ratings related to hospital stay, the hospitalist locates HospitalCompare.com on the Internet. He reviews results from patient satisfaction surveys that compare his institution with others in the geographic area and is surprised to discover that patients generally give his institution a poor rating as well as low marks for the quality of nursing and physicianpatient communication. He is interested in this information but is not aware of resources for further exploration.
Scenario 2 (cont)
At discharge, the hospitalist confirms with Mary that she has received smoking cessation counseling during her hospital stay; he documents this in her chart and discharge summary. The hospitalist was aware of this particular quality measure because the recent weekly score card of hospital performance, posted in the unit, reported that the hospital was not meeting its goal of 100% compliance for this CMS core measure. Among heart attack patients who were identified as smokers, only 80% of charts documented that the patient had been counseled about smoking cessation during the hospital stay. Mary says that she understands the importance of not smoking and says she will make an effort to stop.
| Standard of Care Measure | Sample Metric |
|---|---|
| |
| LDL‐cholesterol assessment | Percentage of patients who have LDL cholesterol measured during hospitalization |
| Lipid‐lowering therapy at discharge | Percentage of patients prescribed a statin at hospital discharge |
| Dietary consultation | Percentage of patients who receive a dietary consult during hospitalization |
| Time to receipt of high‐risk abnormal laboratory assessments | Median time to receipt of high‐risk laboratory results, eg, troponins |
| Cardiac risk assessment | Percentage of patients who receive a cardiac risk assessment during admission |
| Measurement of LVEF | Percentage of patients who receive a cardiac echocardiogram to measure of LVEF before discharge |
| Document communication with PCP | Percentage of patients whose communication with the PCP was documented at discharge |
| Completed medication reconciliation | Percentage of patients for whom medication reconciliation was documented by the time of discharge |
| Make 1‐week follow‐up appointment with PCP | Percentage of patients for whom a 1‐week follow‐up appointment with the PCP was documented at the time of discharge |
| Additional Measure | |
| Inpatient mortality | |
Conclusion
Each hospitalist can have an impact on ACS care systemwide. Hospitalists are on the front line of care and have a unique perspective on patients as they are transitioned through the hospital stay and on how an institution handles patient care overall. They experience firsthand the challenges presented by poor communication between providers, patients, and their families. They can offer breadth of experience and perspective when assessing processes linked to patient care and can be instrumental in ensuring each patient experiences safe transitions during the hospital stay. Hospitalists should participate in QI initiatives for ACS and should consider opportunities to take the lead on these initiatives within their institutions.
Acknowledgements
The author thanks Denise Erkkila, RPh for her editorial assistance in the preparation of this manuscript.
- Hospitalists and intensivists: partners in caring for the critically ill—the time has come.J Hosp Med.2010;5:1–3.
- Society of Hospital Medicine. Acute coronary syndrome.J Hosp Med.2006;1(suppl 1):2–3.
- Society of Hospital Medicine. Quality improvement.J Hosp Med.2006;1 (suppl):92.
- ,,, et al.Curriculum development: the venous thromboembolism quality improvement resource room.J Hosp Med.2006;1:124–132.
- ,,,,Society of Hospital Medicine Glycemic Control Task Force summary: practical recommendations for assessing the impact of glycemic control efforts.J Hosp Med.2008;3:66–75.
- ,,,Effects of a subcutaneous insulin protocol, clinical education, and computerized order set on the quality of inpatient management of hyperglycemia: results of a clinical trial.J Hosp Med.2009;4:16–27.
- SHM Acute Coronary Syndrome Advisory Board. A guide for effective quality improvement: improving acute coronary syndrome care for hospitalized patients. Available at: http://www.hospitalmedicine.org. 2010. Accessed July 2010.
- ,,, et al.2009focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.year="2009"2009;54:2205–2241.
- The Joint Commission. Performance measure intiatives. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/Acute+Myocardial+Infarction+Core+Measure+Set.htm. 2010. Accessed July 2010.
- The Joint Commission, Centers for Medicare and Medicare Services. Specifications manual for national hospital inpatient quality measures, version 2.5. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/Current+NHQM+Manual.htm. 2009 November 6. Accessed July 2010.
- The Joint Commission. Improving America's hospitals: The Joint Commission's annual report on quality and safety 2009. Available at: http://www.jointcommission.org/Library/annual_report.2009. Accessed July 2010.
- Centers for Medicare and Medicare Services. Physician quality reporting initiative (PQRI). Available at: http://www.cms.hhs.gov/pqri. 2010. Accessed July 2010.
- Hospital Care Quality Information from the Consumer Perspective. CAHPS hospital survey. Available at: http://www.hcahpsonline.org. 2010. Accessed July 2010.
- ,,,.Discharge education improves clinical outcomes in patients with chronic heart failure.Circulation.2005;111:179–185.
- ,,, et al.National efforts to improve door‐to‐balloon time results from the Door‐to‐Balloon Alliance.J Am Coll Cardiol.2009;54:2423–2429.
- ,,, et al.Sustaining improvement in door‐to‐balloon time over 4 years: the Mayo clinic ST‐elevation myocardial infarction protocol.Circ Cardiovasc Qual Outcomes.2009;2:508–513.
- ,,,Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists.J Hosp Med.2007;2:314–323.
- ,.Lost in transition: challenges and opportunities for improving the quality of transitional care.Ann Intern Med.2004;141:533–536.
- Society of Hospital Medicine. Boosting Care Transitions Resource Room. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_CareTransitions/CT_Home.cfm. 2010. Accessed July 2010.
- SHM ACS Transitions Workgroup. SHM ACS Transitions Tool. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/05_Transitions.cfm. 2010. Accessed July 2010.
- ,,, et al.Transition of care for hospitalized elderly patients‐‐development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- Hospitalists and intensivists: partners in caring for the critically ill—the time has come.J Hosp Med.2010;5:1–3.
- Society of Hospital Medicine. Acute coronary syndrome.J Hosp Med.2006;1(suppl 1):2–3.
- Society of Hospital Medicine. Quality improvement.J Hosp Med.2006;1 (suppl):92.
- ,,, et al.Curriculum development: the venous thromboembolism quality improvement resource room.J Hosp Med.2006;1:124–132.
- ,,,,Society of Hospital Medicine Glycemic Control Task Force summary: practical recommendations for assessing the impact of glycemic control efforts.J Hosp Med.2008;3:66–75.
- ,,,Effects of a subcutaneous insulin protocol, clinical education, and computerized order set on the quality of inpatient management of hyperglycemia: results of a clinical trial.J Hosp Med.2009;4:16–27.
- SHM Acute Coronary Syndrome Advisory Board. A guide for effective quality improvement: improving acute coronary syndrome care for hospitalized patients. Available at: http://www.hospitalmedicine.org. 2010. Accessed July 2010.
- ,,, et al.2009focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.year="2009"2009;54:2205–2241.
- The Joint Commission. Performance measure intiatives. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/Acute+Myocardial+Infarction+Core+Measure+Set.htm. 2010. Accessed July 2010.
- The Joint Commission, Centers for Medicare and Medicare Services. Specifications manual for national hospital inpatient quality measures, version 2.5. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/Current+NHQM+Manual.htm. 2009 November 6. Accessed July 2010.
- The Joint Commission. Improving America's hospitals: The Joint Commission's annual report on quality and safety 2009. Available at: http://www.jointcommission.org/Library/annual_report.2009. Accessed July 2010.
- Centers for Medicare and Medicare Services. Physician quality reporting initiative (PQRI). Available at: http://www.cms.hhs.gov/pqri. 2010. Accessed July 2010.
- Hospital Care Quality Information from the Consumer Perspective. CAHPS hospital survey. Available at: http://www.hcahpsonline.org. 2010. Accessed July 2010.
- ,,,.Discharge education improves clinical outcomes in patients with chronic heart failure.Circulation.2005;111:179–185.
- ,,, et al.National efforts to improve door‐to‐balloon time results from the Door‐to‐Balloon Alliance.J Am Coll Cardiol.2009;54:2423–2429.
- ,,, et al.Sustaining improvement in door‐to‐balloon time over 4 years: the Mayo clinic ST‐elevation myocardial infarction protocol.Circ Cardiovasc Qual Outcomes.2009;2:508–513.
- ,,,Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists.J Hosp Med.2007;2:314–323.
- ,.Lost in transition: challenges and opportunities for improving the quality of transitional care.Ann Intern Med.2004;141:533–536.
- Society of Hospital Medicine. Boosting Care Transitions Resource Room. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_CareTransitions/CT_Home.cfm. 2010. Accessed July 2010.
- SHM ACS Transitions Workgroup. SHM ACS Transitions Tool. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/05_Transitions.cfm. 2010. Accessed July 2010.
- ,,, et al.Transition of care for hospitalized elderly patients‐‐development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
Transfer of ACS Patients to Primary Care
Patients with acute coronary syndrome (ACS) are a challenge for the hospitalist, as they require substantial coordination of care and support during hospitalization and in the transition back into primary care. ACS accounted for 733,000 discharge diagnoses in 2006 (inclusive of unstable angina or acute myocardial infarction [MI]), and the American Heart Association considers this a conservative estimate.1 Readmission of these patients occurs with some frequency and expends healthcare resources. A multiemployer claims database showed a 20% rehospitalization rate for patients with ischemic heart disease within 1 year of discharge after ACS.2
Implementation of evidenced‐based care for patients with ACS and an emphasis on preventive measures for coronary heart disease (CHD) have improved CHD‐related outcomes. A 2007 study revealed that almost one‐half of the 40% decrease in CHD‐related mortality between 1988 and 2000 was directly attributable to therapeutic interventions and prevention and treatment of recognized risk factors for CHD. However, this news was tempered by the effect of 2 risk factors, increased body mass index and diabetes, which accounted for additional CHD‐related deaths in 2000.3
The Society of Hospital Medicine (SHM) has established core competencies for hospitalists who manage patients with ACS, defining the scope of interactions between the hospitalist, patient, and other clinicians such as specialists and primary care providers (PCP).4, 5 Several competencies focus on ACS and transitions in care, requiring the hospitalist to demonstrate the skills and attitudes outlined in Table 1. Of concern is the risk for adverse events associated with medication errors and lack of follow‐up related to diagnostic tests during the postdischarge period, which in turn raises the risk of readmission.6 The hospitalist is in a unique position to have a positive impact on the transition from inpatient to primary care by proactively addressing patient‐specific issues through careful planning and coordination with the patient, the PCP, and other stakeholders. The hospitalist cannot be accountable for all elements of the transition process, but can be proactive, working with hospital leadership and allied health professionals to promote systems that support safe transitions in care.
| |
| Knowledge | Explain goals for hospital discharge, including specific measures of clinical stability for safe care transition |
| Skills | Synthesize patient history and the results of physical examination, ECG, laboratory, and imaging studies, using risk stratification tools to determine therapeutic options, formulate an evidence‐based treatment plan, and determine the level of care required |
| Assess patients with suspected ACS in a timely manner, identify the level of care required, and manage or co‐manage the patient with the primary requesting service | |
| Attitudes | Communicate with patients and their families to explain the history and prognosis of the patient's cardiac disease |
| Communicate patient risk factors and educate patients in ways to reduce risk | |
| Communicate with patients and their families to explain the goals of the care plan, discharge instructions, and management after hospital discharge; include information about medications, diet, and physical activity | |
| Recognize indications for early specialty consultation, including cardiology and cardiothoracic surgery | |
| Initiate secondary prevention measures prior to discharge including, as appropriate, smoking cessation, dietary modification, and evidencebased medical therapies | |
| Use a multidisciplinary approach in the care of patients with ACS that begins at admission and continues through all care transitions; members of the multidisciplinary team may include nurses, nutritionists, and rehabilitation and social services | |
| Communicate to outpatient providers the notable events of the patient's hospitalization and postdischarge needs, including new therapies, duration of treatment, and outpatient cardiac rehabilitation | |
| Provide and coordinate resources that will help patients make a safe transition from the hospital to arranged follow‐up care and tests | |
Case Study
Jose is a 66‐year‐old retiree recovering on the medical floor after diagnosis of an ST‐segment elevation myocardial infarction (STEMI) and implantation of a drug‐eluting stent (DES). Jose and his family were poor historians on admission and it is unclear whether his medical and medication history are accurate. Jose is anxious to get out of here and thinks this is no big deal. Upon admission Jose was hypertensive and mildly obese. He denies smoking and exercises once in awhile by working in his yard. His lipid profile on admission indicated elevated low‐density lipoprotein (LDL) and total cholesterol.
You decide to address several issues during Jose's hospital stay. These include contacting Jose's PCP to obtain a complete medical and medication history, and educating Jose about his cardiac disease, his risk of future events, and strategies for risk reduction.
Continuity of Care: Key Information Exchange Between the Hospitalist and the Primary Care Physician
Prehospitalization and Hospitalization
Prehospitalization and hospitalization are critical times for the patient with ACS: decisions not only affect the inpatient course but lay the groundwork for care after discharge. For ACS in particular, early identification and understanding of a patient's risk for further ischemic events is critical to determining the therapeutic course, and evidence shows that timely intervention decreases morbidity and mortality.7, 8 Notably, the evaluation and risk stratification of patients with ACS are hospitalist core competencies (Table 1); the initiation of a beta blocker and antiplatelet therapy are considered by the Center for Medicare and Medicaid Services (CMS) to be core measures by which to measure the quality of hospital care. The hospitalist may provide oversight and assure that care is coordinated and patients are assessed and triaged in a timely manner according to recommended guidelines.
Information‐gathering and evaluation begins in the emergency department (ED)9 (Table 2) and continues after admission9 (Table 3), when the hospitalist may need to seek additional medical and medication history to inform risk assessment. In fact, risk assessment should continue throughout the hospital stay as additional diagnostic information is acquired and consultations are provided. Medication reconciliation started during prehospitalization may be complicated by the lack of a reliable source of medication history and should be reevaluated 24 hours after the patient is admitted. Contact with the PCP is appropriate during the hospital stay, with the hospitalist apprising the PCP of diagnoses, interventions, and major clinical events during hospitalization. The PCP may offer valuable insight about issues related to discharge planning.
|
| History |
| Signs and symptoms of current event |
| When available: |
| Prior ECGs |
| Prior related hospitalizations |
| Relevant labs and diagnostics |
| Diagnostics, Laboratory |
| Stat ECG |
| CBC, CMP, INR, Mg, CK‐MB, troponins |
| Fasting lipids, stool guaiac |
| Medication Reconciliation |
| Start beta blocker* |
| Start antiplatelet therapy* |
| Start ACE inhibitor when indicated |
| Statin |
| Evaluation |
| Risk assessment, TIMI score |
| Request cardiology consult if indicated |
|
| History (if incomplete from prehospitalization) |
| Baseline ECG |
| Relevant medical history |
| PCP |
| Patient, family |
| Diagnostics, Laboratory |
| Serial ECG, if indicated |
| CBC, if LMWH or UFH; or PTT if heparin |
| CK‐MB, troponins, serial if indicated |
| VTE prophylaxis |
| Renal function for contrast studies |
| Medication Reconciliation |
| Start beta blocker* |
| Start antiplatelet therapy* |
| Start ACE inhibitor when indicated |
| Statin |
| Evaluation |
| Continuing risk assessment |
| ECHO |
| Stress test |
| Outpatient exercise prescription from physical therapist |
| Cardiology consults |
| Other consults as needed |
| Begin Discharge Planning |
Discharge planning ideally begins soon after admission in order to adequately address issues which may complicate recovery. Medication reconciliation continues during discharge planning, and CMS core measures still apply relative to the use of beta blockers and antiplatelet therapies. Confirming the accuracy of the patient's medication history during hospitalization is an important step in medication reconciliation to ensure that therapies are appropriate and to avoid discrepancies in discharge medications.
A particular challenge of ACS care is the extensive amount of complex information which must be shared quickly and accurately with all stakeholders. The risk of miscommunication is real, but systems and tools are available to lower this risk. At this juncture, technology‐based resources can be especially useful for obtaining and organizing information. Standardized order entry programs or order sets are a reliable method that clinicians can use to meet quality standards during the patient's hospitalization and are highly recommended for patient safety. The SHM ACS Transitions Workgroup has also developed an adaptable multidisciplinary tracking tool that can be used to monitor a patient with ACS through the anticipated transitions in care. This tool tracks key pieces of clinical information throughout the hospital stay and facilitates communication between clinicians. The tool is detailed to include quality measures such as the CMS core measures and can serve as documentation to measure compliance.10 The tracking tool and examples of order sets for patients with ACS are available from the SHM in the Clinical Tools section of the ACS Quality Improvement (QI) Resource Room available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/ACS_Home.cfm.
Discharge and Postdischarge
Discharge is one of the most crucial transitions in care, with potential impact on patient outcomes postdischarge, including readmission. In the past decade, initiatives to improve the discharge process, particularly discharge summaries,11 have yielded standards and tools to improve the process. Current standards for a safe discharge12, 13 are summarized in Table 4. Generally, standards address the need for delivery of a written discharge summary to the clinician who is assuming care after discharge; educating patients about their diagnosis, hospital course, and future medical needs; medication reconciliation; and, arrangement of postdischarge services such as follow‐up appointments. Additionally, the SHM has developed a discharge checklist14 (Table 5) and a template15 for the discharge summary, each to facilitate communication between stakeholders. The discharge summary is an obvious target for QI, as it is the most common vehicle for sharing patient information with the PCP and other healthcare providers. Essential elements of a discharge summary are content (Is key information captured?); format (Is content clear, concise, and accurate?); and delivery (Does the discharge summary reach the right people in a timely manner?).
| Project RED (Re‐Engineered Discharge)* | 2009 National Quality Forum (NQF) |
|---|---|
| Educate the patient about diagnosis during hospitalization | Prepare a written discharge plan |
| Make appointments for clinician follow‐up and postdischarge testing; identify and resolve barriers to follow‐up care | Prepare a written discharge summary |
| Talk to the patient about testing done in the hospital and who will follow up on results | Provide a discharge summary to a licensed clinician who will provide care after discharge |
| Organize postdischarge services; identify and resolve barriers to receiving services | Develop an institutional system to confirm receipt of the discharge summary by a licensed clinician |
| Medication reconciliation: counsel the patient about medications and identify barriers to adherence and compliance | |
| Reconcile the discharge plan with evidence‐based guidelines | |
| Educate the patient on problem‐solving strategies, including contacting the PCP | |
| Expedite transmission of the discharge summary to a licensed clinician and services that will be involved with the patient's care postdischarge | |
| Assess the patient's understanding of the discharge plan; ask patients to explain in their own words; identify and resolve barriers to understanding | |
| Provide the patient with a written summary detailing clinical course, follow‐up, and medication instructions | |
| Telephone the patient 2 to 3 days after discharge to review the plan and address problems | |
|
| Diagnoses |
| Detail location of MI and complications |
| Comorbidities |
| Note diabetes, results of lipid panel, hypertension, renal disease |
| Medications |
| Medication reconciliation |
| Note reason if core measure medications are not prescribed (beta blockers, antiplatelet therapies) |
| ACE/ARB, aspirin, beta‐blockers, statin, sublingual NTG, clopidogrel (include duration of therapy) |
| Titration of any medications |
| Procedures |
| Type, location of stent |
| Complications |
| If ECHO, include type, ejection fraction; provide copy of ECG if available |
| Follow‐up appointment |
| PCP, cardiologist, others such as cardiac rehab |
| Follow‐up testing |
| ETT (type, timeframe); ECHO; required lab work |
| Code status |
| Activity |
| Diet |
| Wound care (eg, groin) |
| Treatment course |
| Cognitive level |
| Discharge LDL |
| Discharge creatinine |
| INR if on warfarin |
| LFTs if on statin |
| Copy all providers |
Evidence indicates that the danger for patients at discharge is often related to medication reconciliation, adverse drug events (ADEs), and pending test results or testing needed after hospitalization.6 Errors affect a sizable proportion of patients, with 49% of patients in 1 study subject to at least 1 medical error within 2 months of discharge. This error was directly attributed to discontinuity during the transition from hospitalist care to the affiliated PCP practice. Errors were related to discharge medications, test results, or lack of PCP follow‐up on testing recommended by the inpatient provider (a work‐up error). Patients with a work‐up error were 6 times more likely to be rehospitalized in the 3 months following the first outpatient visit.16 Another study found that 41% of discharged patients had inhospital test results return after they were discharged and that PCPs were often unaware of these results. In some of these cases, test results required action, sometimes urgently.17
Most adverse events after discharge appear to be ADEs, with up to two‐thirds identified as preventable or ameliorable.1820 In a general medical population, Forster et al.20 found that ADEs resulted in significant injury in 71% of patients, serious injury in 13%, and life‐threatening injury in 16%. ADEs also resulted in 27% of patients requiring emergency care or readmission. Anticoagulants and cardiovascular medications were ranked high among medications associated with an ADE; failure to monitor medications was the most common cause of a preventable or ameliorable ADE, suggesting that ACS patients may be particularly at risk for an ADE following discharge. Patients had good recall of general drug information provided at discharge; patients who could not recall receiving specific information about ADEs were more likely to have an ADE. This is an area of opportunity for the hospitalist to improve patient safety during the transition to primary care. It is also of special concern for patients with ACS, given the complexity of medication regimens and the potential for serious drug reactions.
The risk of adverse events after discharge is higher in certain populations such as the elderly. Project BOOST (Better Outcomes for Older adults through Safe Transitions) is a QI initiative to improve patient care during discharge.21 BOOST aims to reduce 30‐day readmission rates for general medicine patients (especially older adults), improve patient satisfaction, improve communication between the institution and the PCP, identify high‐risk patients and mitigate risk, and improve patient and family education with a focus on patient‐specific risk factors.
BOOST uses a multidisciplinary 7P Screening tool to identify high‐risk patients, specifically through discharge planning and risk stratification.21 Seven areas are assessed: Problem medications, Punk (depression), Principal diagnosis, Polypharmacy, Poor health literacy, Patient support, and Prior hospitalization. For each, specific interventions are recommended to lower the risk of untoward events. 7P Screening applies to patients with ACS and can be an important step in identifying concerns at discharge, with the goals of promoting recovery and reducing the risk of readmission. The BOOST tool also includes a Universal Patient Discharge List reflective of those proposed by Project RED (Re‐Engineered Discharge) and the SHM. However, BOOST suggests additional steps to enhance communication: multidisciplinary rounds at discharge, direct communication with the PCP before discharge, phone contact with the patient or caregiver within 3 days of discharge, and phone numbers for hospital personnel familiar with the patient if the patient is unable to reach the PCP about an issue before the patient's first scheduled follow‐up visit. These steps recognize the value of direct communication between the hospitalist, the patient, and the PCP.
Overcoming Barriers to Communication Between the Hospitalist and the PCP
A successful transition from the hospital to primary care rests largely on the quality of communication between the hospitalist and the PCP. However, only 56% of PCPs expressed satisfaction with the communication they have with hospitalists22 and direct communication is infrequent.11 The PCPs surveyed desired direct and frequent communication, with three‐quarters preferring to speak with the hospitalist by phone at both the patient's admission and discharge.22
PCPs deemed discharge medications and discussion of the reasoning for medication changes and duration of treatment, diagnoses, physical findings, test results, follow‐up needs and plan details, and pending test results to be the most important shared information,11, 22 but this information is frequently unavailable. Details about pending test results at discharge were missed in 65% of summaries, followed by inhospital test results in 33% to 63%, discharge medications in 2% to 40%, and a description of the follow‐up plan in 2% to 43%. Late discharge summaries, some arriving after the first follow‐up visit, are also a barrier to quality care.11, 22, 23 Structured discharge summaries can yield organized and easily retrievable information, with the structure providing cues to include all necessary details.11 These may be computer‐generated vs. traditional dictated or handwritten summaries.6
Use of standardized instruments such as the SHM transitions tool can also help facilitate communication between providers. This tool provides a detailed checklist of recommended diagnostics and therapeutics for patients with ACS that should be considered when the patient is transferred and during discharge. This type of tool has been shown to enhance communication and alert multidisciplinary providers to address issues prior to discharge. A standardized toolkit consisting of a standard admission form, a facsimile to the PCP at admission, a worksheet to identify barriers at discharge, pharmacistphysician medication reconciliation, and predischarge planning appointments reportedly reduced the number of return visits to the ED within 3 days of discharge in an elderly general medicine population; at 30 days there were fewer ED visits and readmissions.24
It is worth noting that patients and their caregivers also have barriers to communication that hospitalists may be able to help address by discharge. These barriers include poor literacy, poor English proficiency, poor understanding of medical jargon, inadequate time with the clinician for questions and answers, poor cognition, highly complex information, and a diagnosis the patient may consider overwhelming.6 Specifically, patients with ACS demonstrated a poor comprehension of their medication regimen after discharge, manifested as either a delay or not filling of prescriptions, followed by poor adherence to the regimen.25 It is also helpful if financial barriers to medication use, either because of direct cost or restriction in outpatient formularies, are identified prior to discharge. Patients report that a follow‐up call from the clinician after discharge or pharmacist counseling before discharge, and use of a pillbox would remove some barriers to adherence. In teach‐back, patients are asked to repeat instructions in their own words, avoiding yes or no answers, thereby revealing gaps in understanding. This strategy checks patient comprehension and provides an opportunity for dialog if it is apparent that patients do not understand information related to their disease and recovery.
Case Study (cont)
Jose's PCP provided more information about his medical and medication history. His father died of a heart attack at 62 years old. Jose has smoked on and off for several years and has been poorly compliant with measures to reduce his risk for CHD such as diet, exercise, and taking statins. Jose may not comprehend the seriousness of his heart disease and how secondary preventive measures may reduce his risk of further events. His history of poor compliance raises concern that he will not persist with recommended ACS medications or antiplatelet regimens after discharge.
Impact of the Hospitalist on Long‐Term Outcomes
Evidenced‐based guidelines stress the need for aggressive modification of risk factors and treatment with antiplatelet, antihypertensive, and lipid‐lowering agents started during hospitalization and continued long‐term as part of secondary prevention strategies.7, 8 There is a missed opportunity for improving patient outcomes after ACS,26, 27 shown by the underuse of guideline‐recommended therapies (antiplatelet therapies such as clopidogrel and aspirin, beta blockers, angiotensin‐converting enzyme [ACE] inhibitors, and statins).28 More than one‐half of patients stopped evidence‐based medications without input from their providers, partly attributed to patients' perception that medication was not needed.26, 27, 29 In another study, 1 in 6 patients who received a DES delayed filling their antiplatelet prescription following discharge (median, 3 days; range, 1‐23 days). Patients who delayed filling the prescription were at increased risk of death or MI compared with patients who filled the prescription on the day of discharge. These findings underscore the importance of discharge planning and patient counseling to improve adherence to medications given at discharge.30 Through education, the hospitalist can directly influence patient and caregiver understanding of the benefit of ACS medications and their effect on long‐term outcomes.
Educating the patient and family about the nature, prognosis, and treatment of cardiac disease is equally important, and secondary prevention measures should be addressed prior to discharge. Prevention includes urging the patient to make therapeutic lifestyle choices such as smoking cessation (a core measure), maintaining a healthy diet, and regular exercise. Addressing these issues is important for the hospitalist and reinforced by the PCP, because adherence to behavioral changes after ACS has been shown to be poor and to directly impact outcomes. In 1 study, one‐third of smokers continued to smoke after 1 month and about one‐third of patients did not adhere to a recommended diet or exercise regimen. At 6 months, those patients who did quit smoking had a 43% lower risk of MI; compliance with the diet and exercise regimen lowered their risk of MI by 48%. Persistent smoking and nonadherence to diet and exercise resulted in an almost 4‐fold increased risk of MI, stroke, or death vs. never‐smokers who adhered to diet and exercise recommendations.31 This presents a clear opportunity for the hospitalist to intervene and affect change. Assessment of needed secondary prevention measures should occur in preparation for discharge, and an emphasis on patient teaching and communicating the plan to the PCP may overcome patient barriers to adhering to recommended lifestyle changes.
Case Study (cont)
After verbally describing his discharge medications to Jose and his family, and providing written patient materials, you ask Jose to explain why his prescribed dual antiplatelet therapy is important. He states it will help his occasional arthritis, because aspirin is one of the drugs, revealing that Jose lacks understanding of why he is taking the antiplatelet therapies or why they are important. A consult is requested from pharmacy for additional counseling. Because you have been in direct contact with the PCP, you call now to express your concerns, in addition to noting Jose's poor comprehension in the discharge summary.
Conclusion
Timely and accurate communication between the hospitalist and the PCP is a vital component of a safe transition from inpatient to primary care. This communication directly impacts the continuity of care, patient outcomes, patient and caregiver satisfaction, and use of healthcare resources. The role of the hospitalist is still evolving. Hospitalists will continue to have a pivotal role in transitions of care, and have a direct impact on the quality of the transition at discharge and patient outcomes after ACS. Hospitalists should be cognizant of gaps in care related to how information is generated, recorded, and shared between the inpatient setting and primary care, and should be proactive in identifying barriers and facilitating solutions. The hospitalist's responsibility for the patient does not end at the time of discharge but extends until the PCP assumes responsibility for patient care following hospitalization. We must make every reasonable effort to assure that our patients and their outpatient providers and caregivers are given all the tools necessary to complete and maintain the patient's therapy.
Acknowledgements
The author thanks Denise Erkkila, RPh for her editorial assistance in preparation of this manuscript.
- American Heart Association. Heart disease and stroke statistics‐2010 update. Dallas, Texas: American Heart Association;2010.
- ,,,,.One‐year costs of ischemic heart disease among patients with acute coronary syndromes: findings from a multi‐employer claims database.Curr Med Res Opin.2008;24:461–468.
- ,,, et al.Explaining the decrease in U.S. deaths from coronary disease, 1980‐2000.N Engl J Med.2007;356:2388–2398.
- ,,,,.Core competencies in hospital medicine: development and methodology.J Hosp Med.2006;1:48–56.
- Society of Hospital Medicine.Acute coronary syndrome.J Hosp Med.2006;1(suppl 1):2–3.
- ,,,.Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists.J Hosp Med.2007;2:314–323.
- ,,, et al.ACC/AHA 2007 guidelines for the management of patients with unstable angina/non ST‐elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non ST‐Elevation Myocardial Infarction): developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons: endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine.Circulation.2007;116:e148–e304.
- ,,, et al.2009 focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.2009;54:2205–2241.
- SHM Acute Coronary Syndrome Advisory Board.A guide for effective quality improvement: improving acute coronary syndrome care for hospitalized patients. Available at: http://www.hospitalmedicine.org.2010. Accessed July 2010.
- SHM ACS Transitions Workgroup.SHM ACS Transitions Tool. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/05_Transitions.cfm.2010. Accessed July 2010.
- ,,, et al.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- .Reengineering hospital discharge: a protocol to improve patient safety, reduce costs, and boost patient satisfaction.Am J Med Qual.2009;24:344–346.
- National Quality Forum.National Quality Forum (NQF) endorsed set of 34 safe practices. Available at: http://www.hfap.org/pdf/patient_safety.pdf.2009. Accessed July 2010.
- ,,, et al.Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- Society of Hospital Medicine.SHM Acute Coronary Syndrome (ACS) Discharge Planning Checklist. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/04_Discharge. cfm.2010. Accessed July 2010.
- ,,,.Medical errors related to discontinuity of care from an inpatient to an outpatient setting.J Gen Intern Med.2003;18:646–651.
- ,,, et al.Patient safety concerns arising from test results that return after hospital discharge.Ann Intern Med.2005;143:121–128.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- ,,, et al.Adverse events among medical patients after discharge from hospital.CMAJ.2004;170:345–349.
- ,,,,.Adverse drug events occurring following hospital discharge.J Gen Intern Med.2005;20:317–323.
- Society of Hospital Medicine.Boosting Care Transitions Resource Room. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_CareTransitions/CT_Home.cfm.2010. Accessed July 2010.
- ,,,.Primary care physician attitudes regarding communication with hospitalists.Dis Mon.2002;48:218–229.
- ,,,.Effect of discharge summary availability during post‐discharge visits on hospital readmission.J Gen Intern Med.2002;17:186–192.
- ,,, et al.A quality improvement intervention to facilitate the transition of older adults from three hospitals back to their homes.J Am Geriatr Soc.2009;57:1540–1546.
- ,,,.Medication use among inner‐city patients after hospital discharge: patient‐reported barriers and solutions.Mayo Clin Proc.2008;83:529–535.
- ,,, et al.Predictors of early discontinuation of evidence‐based medicine after acute coronary syndrome.Am J Cardiol.2009;104:175–181.
- ,,, et al.Optimal medical therapy at discharge in patients with acute coronary syndromes: temporal changes, characteristics, and 1‐year outcome.Am Heart J.2007;154:1108–1115.
- ,,.Reinforcing a continuum of care: in‐hospital initiation of long‐term secondary prevention following acute coronary syndromes.Cardiovasc Drugs Ther.2007;21:375–388.
- ,,, et al.Age and persistent use of cardiovascular medication after acute coronary syndrome: results from medication applied and sustained over time.J Am Geriatr Soc.2009;57:1990–1996.
- ,,, et al.Delays in filling clopidogrel prescription after hospital discharge and adverse outcomes after drug‐eluting stent implantation.Circ Cardiovasc Qual Outcomes.2010;3:261–266.
- ,,, et al.Association of diet, exercise, and smoking modification with risk of early cardiovascular events after acute coronary syndromes.Circulation.2010;121:750–758.
Patients with acute coronary syndrome (ACS) are a challenge for the hospitalist, as they require substantial coordination of care and support during hospitalization and in the transition back into primary care. ACS accounted for 733,000 discharge diagnoses in 2006 (inclusive of unstable angina or acute myocardial infarction [MI]), and the American Heart Association considers this a conservative estimate.1 Readmission of these patients occurs with some frequency and expends healthcare resources. A multiemployer claims database showed a 20% rehospitalization rate for patients with ischemic heart disease within 1 year of discharge after ACS.2
Implementation of evidenced‐based care for patients with ACS and an emphasis on preventive measures for coronary heart disease (CHD) have improved CHD‐related outcomes. A 2007 study revealed that almost one‐half of the 40% decrease in CHD‐related mortality between 1988 and 2000 was directly attributable to therapeutic interventions and prevention and treatment of recognized risk factors for CHD. However, this news was tempered by the effect of 2 risk factors, increased body mass index and diabetes, which accounted for additional CHD‐related deaths in 2000.3
The Society of Hospital Medicine (SHM) has established core competencies for hospitalists who manage patients with ACS, defining the scope of interactions between the hospitalist, patient, and other clinicians such as specialists and primary care providers (PCP).4, 5 Several competencies focus on ACS and transitions in care, requiring the hospitalist to demonstrate the skills and attitudes outlined in Table 1. Of concern is the risk for adverse events associated with medication errors and lack of follow‐up related to diagnostic tests during the postdischarge period, which in turn raises the risk of readmission.6 The hospitalist is in a unique position to have a positive impact on the transition from inpatient to primary care by proactively addressing patient‐specific issues through careful planning and coordination with the patient, the PCP, and other stakeholders. The hospitalist cannot be accountable for all elements of the transition process, but can be proactive, working with hospital leadership and allied health professionals to promote systems that support safe transitions in care.
| |
| Knowledge | Explain goals for hospital discharge, including specific measures of clinical stability for safe care transition |
| Skills | Synthesize patient history and the results of physical examination, ECG, laboratory, and imaging studies, using risk stratification tools to determine therapeutic options, formulate an evidence‐based treatment plan, and determine the level of care required |
| Assess patients with suspected ACS in a timely manner, identify the level of care required, and manage or co‐manage the patient with the primary requesting service | |
| Attitudes | Communicate with patients and their families to explain the history and prognosis of the patient's cardiac disease |
| Communicate patient risk factors and educate patients in ways to reduce risk | |
| Communicate with patients and their families to explain the goals of the care plan, discharge instructions, and management after hospital discharge; include information about medications, diet, and physical activity | |
| Recognize indications for early specialty consultation, including cardiology and cardiothoracic surgery | |
| Initiate secondary prevention measures prior to discharge including, as appropriate, smoking cessation, dietary modification, and evidencebased medical therapies | |
| Use a multidisciplinary approach in the care of patients with ACS that begins at admission and continues through all care transitions; members of the multidisciplinary team may include nurses, nutritionists, and rehabilitation and social services | |
| Communicate to outpatient providers the notable events of the patient's hospitalization and postdischarge needs, including new therapies, duration of treatment, and outpatient cardiac rehabilitation | |
| Provide and coordinate resources that will help patients make a safe transition from the hospital to arranged follow‐up care and tests | |
Case Study
Jose is a 66‐year‐old retiree recovering on the medical floor after diagnosis of an ST‐segment elevation myocardial infarction (STEMI) and implantation of a drug‐eluting stent (DES). Jose and his family were poor historians on admission and it is unclear whether his medical and medication history are accurate. Jose is anxious to get out of here and thinks this is no big deal. Upon admission Jose was hypertensive and mildly obese. He denies smoking and exercises once in awhile by working in his yard. His lipid profile on admission indicated elevated low‐density lipoprotein (LDL) and total cholesterol.
You decide to address several issues during Jose's hospital stay. These include contacting Jose's PCP to obtain a complete medical and medication history, and educating Jose about his cardiac disease, his risk of future events, and strategies for risk reduction.
Continuity of Care: Key Information Exchange Between the Hospitalist and the Primary Care Physician
Prehospitalization and Hospitalization
Prehospitalization and hospitalization are critical times for the patient with ACS: decisions not only affect the inpatient course but lay the groundwork for care after discharge. For ACS in particular, early identification and understanding of a patient's risk for further ischemic events is critical to determining the therapeutic course, and evidence shows that timely intervention decreases morbidity and mortality.7, 8 Notably, the evaluation and risk stratification of patients with ACS are hospitalist core competencies (Table 1); the initiation of a beta blocker and antiplatelet therapy are considered by the Center for Medicare and Medicaid Services (CMS) to be core measures by which to measure the quality of hospital care. The hospitalist may provide oversight and assure that care is coordinated and patients are assessed and triaged in a timely manner according to recommended guidelines.
Information‐gathering and evaluation begins in the emergency department (ED)9 (Table 2) and continues after admission9 (Table 3), when the hospitalist may need to seek additional medical and medication history to inform risk assessment. In fact, risk assessment should continue throughout the hospital stay as additional diagnostic information is acquired and consultations are provided. Medication reconciliation started during prehospitalization may be complicated by the lack of a reliable source of medication history and should be reevaluated 24 hours after the patient is admitted. Contact with the PCP is appropriate during the hospital stay, with the hospitalist apprising the PCP of diagnoses, interventions, and major clinical events during hospitalization. The PCP may offer valuable insight about issues related to discharge planning.
|
| History |
| Signs and symptoms of current event |
| When available: |
| Prior ECGs |
| Prior related hospitalizations |
| Relevant labs and diagnostics |
| Diagnostics, Laboratory |
| Stat ECG |
| CBC, CMP, INR, Mg, CK‐MB, troponins |
| Fasting lipids, stool guaiac |
| Medication Reconciliation |
| Start beta blocker* |
| Start antiplatelet therapy* |
| Start ACE inhibitor when indicated |
| Statin |
| Evaluation |
| Risk assessment, TIMI score |
| Request cardiology consult if indicated |
|
| History (if incomplete from prehospitalization) |
| Baseline ECG |
| Relevant medical history |
| PCP |
| Patient, family |
| Diagnostics, Laboratory |
| Serial ECG, if indicated |
| CBC, if LMWH or UFH; or PTT if heparin |
| CK‐MB, troponins, serial if indicated |
| VTE prophylaxis |
| Renal function for contrast studies |
| Medication Reconciliation |
| Start beta blocker* |
| Start antiplatelet therapy* |
| Start ACE inhibitor when indicated |
| Statin |
| Evaluation |
| Continuing risk assessment |
| ECHO |
| Stress test |
| Outpatient exercise prescription from physical therapist |
| Cardiology consults |
| Other consults as needed |
| Begin Discharge Planning |
Discharge planning ideally begins soon after admission in order to adequately address issues which may complicate recovery. Medication reconciliation continues during discharge planning, and CMS core measures still apply relative to the use of beta blockers and antiplatelet therapies. Confirming the accuracy of the patient's medication history during hospitalization is an important step in medication reconciliation to ensure that therapies are appropriate and to avoid discrepancies in discharge medications.
A particular challenge of ACS care is the extensive amount of complex information which must be shared quickly and accurately with all stakeholders. The risk of miscommunication is real, but systems and tools are available to lower this risk. At this juncture, technology‐based resources can be especially useful for obtaining and organizing information. Standardized order entry programs or order sets are a reliable method that clinicians can use to meet quality standards during the patient's hospitalization and are highly recommended for patient safety. The SHM ACS Transitions Workgroup has also developed an adaptable multidisciplinary tracking tool that can be used to monitor a patient with ACS through the anticipated transitions in care. This tool tracks key pieces of clinical information throughout the hospital stay and facilitates communication between clinicians. The tool is detailed to include quality measures such as the CMS core measures and can serve as documentation to measure compliance.10 The tracking tool and examples of order sets for patients with ACS are available from the SHM in the Clinical Tools section of the ACS Quality Improvement (QI) Resource Room available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/ACS_Home.cfm.
Discharge and Postdischarge
Discharge is one of the most crucial transitions in care, with potential impact on patient outcomes postdischarge, including readmission. In the past decade, initiatives to improve the discharge process, particularly discharge summaries,11 have yielded standards and tools to improve the process. Current standards for a safe discharge12, 13 are summarized in Table 4. Generally, standards address the need for delivery of a written discharge summary to the clinician who is assuming care after discharge; educating patients about their diagnosis, hospital course, and future medical needs; medication reconciliation; and, arrangement of postdischarge services such as follow‐up appointments. Additionally, the SHM has developed a discharge checklist14 (Table 5) and a template15 for the discharge summary, each to facilitate communication between stakeholders. The discharge summary is an obvious target for QI, as it is the most common vehicle for sharing patient information with the PCP and other healthcare providers. Essential elements of a discharge summary are content (Is key information captured?); format (Is content clear, concise, and accurate?); and delivery (Does the discharge summary reach the right people in a timely manner?).
| Project RED (Re‐Engineered Discharge)* | 2009 National Quality Forum (NQF) |
|---|---|
| Educate the patient about diagnosis during hospitalization | Prepare a written discharge plan |
| Make appointments for clinician follow‐up and postdischarge testing; identify and resolve barriers to follow‐up care | Prepare a written discharge summary |
| Talk to the patient about testing done in the hospital and who will follow up on results | Provide a discharge summary to a licensed clinician who will provide care after discharge |
| Organize postdischarge services; identify and resolve barriers to receiving services | Develop an institutional system to confirm receipt of the discharge summary by a licensed clinician |
| Medication reconciliation: counsel the patient about medications and identify barriers to adherence and compliance | |
| Reconcile the discharge plan with evidence‐based guidelines | |
| Educate the patient on problem‐solving strategies, including contacting the PCP | |
| Expedite transmission of the discharge summary to a licensed clinician and services that will be involved with the patient's care postdischarge | |
| Assess the patient's understanding of the discharge plan; ask patients to explain in their own words; identify and resolve barriers to understanding | |
| Provide the patient with a written summary detailing clinical course, follow‐up, and medication instructions | |
| Telephone the patient 2 to 3 days after discharge to review the plan and address problems | |
|
| Diagnoses |
| Detail location of MI and complications |
| Comorbidities |
| Note diabetes, results of lipid panel, hypertension, renal disease |
| Medications |
| Medication reconciliation |
| Note reason if core measure medications are not prescribed (beta blockers, antiplatelet therapies) |
| ACE/ARB, aspirin, beta‐blockers, statin, sublingual NTG, clopidogrel (include duration of therapy) |
| Titration of any medications |
| Procedures |
| Type, location of stent |
| Complications |
| If ECHO, include type, ejection fraction; provide copy of ECG if available |
| Follow‐up appointment |
| PCP, cardiologist, others such as cardiac rehab |
| Follow‐up testing |
| ETT (type, timeframe); ECHO; required lab work |
| Code status |
| Activity |
| Diet |
| Wound care (eg, groin) |
| Treatment course |
| Cognitive level |
| Discharge LDL |
| Discharge creatinine |
| INR if on warfarin |
| LFTs if on statin |
| Copy all providers |
Evidence indicates that the danger for patients at discharge is often related to medication reconciliation, adverse drug events (ADEs), and pending test results or testing needed after hospitalization.6 Errors affect a sizable proportion of patients, with 49% of patients in 1 study subject to at least 1 medical error within 2 months of discharge. This error was directly attributed to discontinuity during the transition from hospitalist care to the affiliated PCP practice. Errors were related to discharge medications, test results, or lack of PCP follow‐up on testing recommended by the inpatient provider (a work‐up error). Patients with a work‐up error were 6 times more likely to be rehospitalized in the 3 months following the first outpatient visit.16 Another study found that 41% of discharged patients had inhospital test results return after they were discharged and that PCPs were often unaware of these results. In some of these cases, test results required action, sometimes urgently.17
Most adverse events after discharge appear to be ADEs, with up to two‐thirds identified as preventable or ameliorable.1820 In a general medical population, Forster et al.20 found that ADEs resulted in significant injury in 71% of patients, serious injury in 13%, and life‐threatening injury in 16%. ADEs also resulted in 27% of patients requiring emergency care or readmission. Anticoagulants and cardiovascular medications were ranked high among medications associated with an ADE; failure to monitor medications was the most common cause of a preventable or ameliorable ADE, suggesting that ACS patients may be particularly at risk for an ADE following discharge. Patients had good recall of general drug information provided at discharge; patients who could not recall receiving specific information about ADEs were more likely to have an ADE. This is an area of opportunity for the hospitalist to improve patient safety during the transition to primary care. It is also of special concern for patients with ACS, given the complexity of medication regimens and the potential for serious drug reactions.
The risk of adverse events after discharge is higher in certain populations such as the elderly. Project BOOST (Better Outcomes for Older adults through Safe Transitions) is a QI initiative to improve patient care during discharge.21 BOOST aims to reduce 30‐day readmission rates for general medicine patients (especially older adults), improve patient satisfaction, improve communication between the institution and the PCP, identify high‐risk patients and mitigate risk, and improve patient and family education with a focus on patient‐specific risk factors.
BOOST uses a multidisciplinary 7P Screening tool to identify high‐risk patients, specifically through discharge planning and risk stratification.21 Seven areas are assessed: Problem medications, Punk (depression), Principal diagnosis, Polypharmacy, Poor health literacy, Patient support, and Prior hospitalization. For each, specific interventions are recommended to lower the risk of untoward events. 7P Screening applies to patients with ACS and can be an important step in identifying concerns at discharge, with the goals of promoting recovery and reducing the risk of readmission. The BOOST tool also includes a Universal Patient Discharge List reflective of those proposed by Project RED (Re‐Engineered Discharge) and the SHM. However, BOOST suggests additional steps to enhance communication: multidisciplinary rounds at discharge, direct communication with the PCP before discharge, phone contact with the patient or caregiver within 3 days of discharge, and phone numbers for hospital personnel familiar with the patient if the patient is unable to reach the PCP about an issue before the patient's first scheduled follow‐up visit. These steps recognize the value of direct communication between the hospitalist, the patient, and the PCP.
Overcoming Barriers to Communication Between the Hospitalist and the PCP
A successful transition from the hospital to primary care rests largely on the quality of communication between the hospitalist and the PCP. However, only 56% of PCPs expressed satisfaction with the communication they have with hospitalists22 and direct communication is infrequent.11 The PCPs surveyed desired direct and frequent communication, with three‐quarters preferring to speak with the hospitalist by phone at both the patient's admission and discharge.22
PCPs deemed discharge medications and discussion of the reasoning for medication changes and duration of treatment, diagnoses, physical findings, test results, follow‐up needs and plan details, and pending test results to be the most important shared information,11, 22 but this information is frequently unavailable. Details about pending test results at discharge were missed in 65% of summaries, followed by inhospital test results in 33% to 63%, discharge medications in 2% to 40%, and a description of the follow‐up plan in 2% to 43%. Late discharge summaries, some arriving after the first follow‐up visit, are also a barrier to quality care.11, 22, 23 Structured discharge summaries can yield organized and easily retrievable information, with the structure providing cues to include all necessary details.11 These may be computer‐generated vs. traditional dictated or handwritten summaries.6
Use of standardized instruments such as the SHM transitions tool can also help facilitate communication between providers. This tool provides a detailed checklist of recommended diagnostics and therapeutics for patients with ACS that should be considered when the patient is transferred and during discharge. This type of tool has been shown to enhance communication and alert multidisciplinary providers to address issues prior to discharge. A standardized toolkit consisting of a standard admission form, a facsimile to the PCP at admission, a worksheet to identify barriers at discharge, pharmacistphysician medication reconciliation, and predischarge planning appointments reportedly reduced the number of return visits to the ED within 3 days of discharge in an elderly general medicine population; at 30 days there were fewer ED visits and readmissions.24
It is worth noting that patients and their caregivers also have barriers to communication that hospitalists may be able to help address by discharge. These barriers include poor literacy, poor English proficiency, poor understanding of medical jargon, inadequate time with the clinician for questions and answers, poor cognition, highly complex information, and a diagnosis the patient may consider overwhelming.6 Specifically, patients with ACS demonstrated a poor comprehension of their medication regimen after discharge, manifested as either a delay or not filling of prescriptions, followed by poor adherence to the regimen.25 It is also helpful if financial barriers to medication use, either because of direct cost or restriction in outpatient formularies, are identified prior to discharge. Patients report that a follow‐up call from the clinician after discharge or pharmacist counseling before discharge, and use of a pillbox would remove some barriers to adherence. In teach‐back, patients are asked to repeat instructions in their own words, avoiding yes or no answers, thereby revealing gaps in understanding. This strategy checks patient comprehension and provides an opportunity for dialog if it is apparent that patients do not understand information related to their disease and recovery.
Case Study (cont)
Jose's PCP provided more information about his medical and medication history. His father died of a heart attack at 62 years old. Jose has smoked on and off for several years and has been poorly compliant with measures to reduce his risk for CHD such as diet, exercise, and taking statins. Jose may not comprehend the seriousness of his heart disease and how secondary preventive measures may reduce his risk of further events. His history of poor compliance raises concern that he will not persist with recommended ACS medications or antiplatelet regimens after discharge.
Impact of the Hospitalist on Long‐Term Outcomes
Evidenced‐based guidelines stress the need for aggressive modification of risk factors and treatment with antiplatelet, antihypertensive, and lipid‐lowering agents started during hospitalization and continued long‐term as part of secondary prevention strategies.7, 8 There is a missed opportunity for improving patient outcomes after ACS,26, 27 shown by the underuse of guideline‐recommended therapies (antiplatelet therapies such as clopidogrel and aspirin, beta blockers, angiotensin‐converting enzyme [ACE] inhibitors, and statins).28 More than one‐half of patients stopped evidence‐based medications without input from their providers, partly attributed to patients' perception that medication was not needed.26, 27, 29 In another study, 1 in 6 patients who received a DES delayed filling their antiplatelet prescription following discharge (median, 3 days; range, 1‐23 days). Patients who delayed filling the prescription were at increased risk of death or MI compared with patients who filled the prescription on the day of discharge. These findings underscore the importance of discharge planning and patient counseling to improve adherence to medications given at discharge.30 Through education, the hospitalist can directly influence patient and caregiver understanding of the benefit of ACS medications and their effect on long‐term outcomes.
Educating the patient and family about the nature, prognosis, and treatment of cardiac disease is equally important, and secondary prevention measures should be addressed prior to discharge. Prevention includes urging the patient to make therapeutic lifestyle choices such as smoking cessation (a core measure), maintaining a healthy diet, and regular exercise. Addressing these issues is important for the hospitalist and reinforced by the PCP, because adherence to behavioral changes after ACS has been shown to be poor and to directly impact outcomes. In 1 study, one‐third of smokers continued to smoke after 1 month and about one‐third of patients did not adhere to a recommended diet or exercise regimen. At 6 months, those patients who did quit smoking had a 43% lower risk of MI; compliance with the diet and exercise regimen lowered their risk of MI by 48%. Persistent smoking and nonadherence to diet and exercise resulted in an almost 4‐fold increased risk of MI, stroke, or death vs. never‐smokers who adhered to diet and exercise recommendations.31 This presents a clear opportunity for the hospitalist to intervene and affect change. Assessment of needed secondary prevention measures should occur in preparation for discharge, and an emphasis on patient teaching and communicating the plan to the PCP may overcome patient barriers to adhering to recommended lifestyle changes.
Case Study (cont)
After verbally describing his discharge medications to Jose and his family, and providing written patient materials, you ask Jose to explain why his prescribed dual antiplatelet therapy is important. He states it will help his occasional arthritis, because aspirin is one of the drugs, revealing that Jose lacks understanding of why he is taking the antiplatelet therapies or why they are important. A consult is requested from pharmacy for additional counseling. Because you have been in direct contact with the PCP, you call now to express your concerns, in addition to noting Jose's poor comprehension in the discharge summary.
Conclusion
Timely and accurate communication between the hospitalist and the PCP is a vital component of a safe transition from inpatient to primary care. This communication directly impacts the continuity of care, patient outcomes, patient and caregiver satisfaction, and use of healthcare resources. The role of the hospitalist is still evolving. Hospitalists will continue to have a pivotal role in transitions of care, and have a direct impact on the quality of the transition at discharge and patient outcomes after ACS. Hospitalists should be cognizant of gaps in care related to how information is generated, recorded, and shared between the inpatient setting and primary care, and should be proactive in identifying barriers and facilitating solutions. The hospitalist's responsibility for the patient does not end at the time of discharge but extends until the PCP assumes responsibility for patient care following hospitalization. We must make every reasonable effort to assure that our patients and their outpatient providers and caregivers are given all the tools necessary to complete and maintain the patient's therapy.
Acknowledgements
The author thanks Denise Erkkila, RPh for her editorial assistance in preparation of this manuscript.
Patients with acute coronary syndrome (ACS) are a challenge for the hospitalist, as they require substantial coordination of care and support during hospitalization and in the transition back into primary care. ACS accounted for 733,000 discharge diagnoses in 2006 (inclusive of unstable angina or acute myocardial infarction [MI]), and the American Heart Association considers this a conservative estimate.1 Readmission of these patients occurs with some frequency and expends healthcare resources. A multiemployer claims database showed a 20% rehospitalization rate for patients with ischemic heart disease within 1 year of discharge after ACS.2
Implementation of evidenced‐based care for patients with ACS and an emphasis on preventive measures for coronary heart disease (CHD) have improved CHD‐related outcomes. A 2007 study revealed that almost one‐half of the 40% decrease in CHD‐related mortality between 1988 and 2000 was directly attributable to therapeutic interventions and prevention and treatment of recognized risk factors for CHD. However, this news was tempered by the effect of 2 risk factors, increased body mass index and diabetes, which accounted for additional CHD‐related deaths in 2000.3
The Society of Hospital Medicine (SHM) has established core competencies for hospitalists who manage patients with ACS, defining the scope of interactions between the hospitalist, patient, and other clinicians such as specialists and primary care providers (PCP).4, 5 Several competencies focus on ACS and transitions in care, requiring the hospitalist to demonstrate the skills and attitudes outlined in Table 1. Of concern is the risk for adverse events associated with medication errors and lack of follow‐up related to diagnostic tests during the postdischarge period, which in turn raises the risk of readmission.6 The hospitalist is in a unique position to have a positive impact on the transition from inpatient to primary care by proactively addressing patient‐specific issues through careful planning and coordination with the patient, the PCP, and other stakeholders. The hospitalist cannot be accountable for all elements of the transition process, but can be proactive, working with hospital leadership and allied health professionals to promote systems that support safe transitions in care.
| |
| Knowledge | Explain goals for hospital discharge, including specific measures of clinical stability for safe care transition |
| Skills | Synthesize patient history and the results of physical examination, ECG, laboratory, and imaging studies, using risk stratification tools to determine therapeutic options, formulate an evidence‐based treatment plan, and determine the level of care required |
| Assess patients with suspected ACS in a timely manner, identify the level of care required, and manage or co‐manage the patient with the primary requesting service | |
| Attitudes | Communicate with patients and their families to explain the history and prognosis of the patient's cardiac disease |
| Communicate patient risk factors and educate patients in ways to reduce risk | |
| Communicate with patients and their families to explain the goals of the care plan, discharge instructions, and management after hospital discharge; include information about medications, diet, and physical activity | |
| Recognize indications for early specialty consultation, including cardiology and cardiothoracic surgery | |
| Initiate secondary prevention measures prior to discharge including, as appropriate, smoking cessation, dietary modification, and evidencebased medical therapies | |
| Use a multidisciplinary approach in the care of patients with ACS that begins at admission and continues through all care transitions; members of the multidisciplinary team may include nurses, nutritionists, and rehabilitation and social services | |
| Communicate to outpatient providers the notable events of the patient's hospitalization and postdischarge needs, including new therapies, duration of treatment, and outpatient cardiac rehabilitation | |
| Provide and coordinate resources that will help patients make a safe transition from the hospital to arranged follow‐up care and tests | |
Case Study
Jose is a 66‐year‐old retiree recovering on the medical floor after diagnosis of an ST‐segment elevation myocardial infarction (STEMI) and implantation of a drug‐eluting stent (DES). Jose and his family were poor historians on admission and it is unclear whether his medical and medication history are accurate. Jose is anxious to get out of here and thinks this is no big deal. Upon admission Jose was hypertensive and mildly obese. He denies smoking and exercises once in awhile by working in his yard. His lipid profile on admission indicated elevated low‐density lipoprotein (LDL) and total cholesterol.
You decide to address several issues during Jose's hospital stay. These include contacting Jose's PCP to obtain a complete medical and medication history, and educating Jose about his cardiac disease, his risk of future events, and strategies for risk reduction.
Continuity of Care: Key Information Exchange Between the Hospitalist and the Primary Care Physician
Prehospitalization and Hospitalization
Prehospitalization and hospitalization are critical times for the patient with ACS: decisions not only affect the inpatient course but lay the groundwork for care after discharge. For ACS in particular, early identification and understanding of a patient's risk for further ischemic events is critical to determining the therapeutic course, and evidence shows that timely intervention decreases morbidity and mortality.7, 8 Notably, the evaluation and risk stratification of patients with ACS are hospitalist core competencies (Table 1); the initiation of a beta blocker and antiplatelet therapy are considered by the Center for Medicare and Medicaid Services (CMS) to be core measures by which to measure the quality of hospital care. The hospitalist may provide oversight and assure that care is coordinated and patients are assessed and triaged in a timely manner according to recommended guidelines.
Information‐gathering and evaluation begins in the emergency department (ED)9 (Table 2) and continues after admission9 (Table 3), when the hospitalist may need to seek additional medical and medication history to inform risk assessment. In fact, risk assessment should continue throughout the hospital stay as additional diagnostic information is acquired and consultations are provided. Medication reconciliation started during prehospitalization may be complicated by the lack of a reliable source of medication history and should be reevaluated 24 hours after the patient is admitted. Contact with the PCP is appropriate during the hospital stay, with the hospitalist apprising the PCP of diagnoses, interventions, and major clinical events during hospitalization. The PCP may offer valuable insight about issues related to discharge planning.
|
| History |
| Signs and symptoms of current event |
| When available: |
| Prior ECGs |
| Prior related hospitalizations |
| Relevant labs and diagnostics |
| Diagnostics, Laboratory |
| Stat ECG |
| CBC, CMP, INR, Mg, CK‐MB, troponins |
| Fasting lipids, stool guaiac |
| Medication Reconciliation |
| Start beta blocker* |
| Start antiplatelet therapy* |
| Start ACE inhibitor when indicated |
| Statin |
| Evaluation |
| Risk assessment, TIMI score |
| Request cardiology consult if indicated |
|
| History (if incomplete from prehospitalization) |
| Baseline ECG |
| Relevant medical history |
| PCP |
| Patient, family |
| Diagnostics, Laboratory |
| Serial ECG, if indicated |
| CBC, if LMWH or UFH; or PTT if heparin |
| CK‐MB, troponins, serial if indicated |
| VTE prophylaxis |
| Renal function for contrast studies |
| Medication Reconciliation |
| Start beta blocker* |
| Start antiplatelet therapy* |
| Start ACE inhibitor when indicated |
| Statin |
| Evaluation |
| Continuing risk assessment |
| ECHO |
| Stress test |
| Outpatient exercise prescription from physical therapist |
| Cardiology consults |
| Other consults as needed |
| Begin Discharge Planning |
Discharge planning ideally begins soon after admission in order to adequately address issues which may complicate recovery. Medication reconciliation continues during discharge planning, and CMS core measures still apply relative to the use of beta blockers and antiplatelet therapies. Confirming the accuracy of the patient's medication history during hospitalization is an important step in medication reconciliation to ensure that therapies are appropriate and to avoid discrepancies in discharge medications.
A particular challenge of ACS care is the extensive amount of complex information which must be shared quickly and accurately with all stakeholders. The risk of miscommunication is real, but systems and tools are available to lower this risk. At this juncture, technology‐based resources can be especially useful for obtaining and organizing information. Standardized order entry programs or order sets are a reliable method that clinicians can use to meet quality standards during the patient's hospitalization and are highly recommended for patient safety. The SHM ACS Transitions Workgroup has also developed an adaptable multidisciplinary tracking tool that can be used to monitor a patient with ACS through the anticipated transitions in care. This tool tracks key pieces of clinical information throughout the hospital stay and facilitates communication between clinicians. The tool is detailed to include quality measures such as the CMS core measures and can serve as documentation to measure compliance.10 The tracking tool and examples of order sets for patients with ACS are available from the SHM in the Clinical Tools section of the ACS Quality Improvement (QI) Resource Room available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/ACS_Home.cfm.
Discharge and Postdischarge
Discharge is one of the most crucial transitions in care, with potential impact on patient outcomes postdischarge, including readmission. In the past decade, initiatives to improve the discharge process, particularly discharge summaries,11 have yielded standards and tools to improve the process. Current standards for a safe discharge12, 13 are summarized in Table 4. Generally, standards address the need for delivery of a written discharge summary to the clinician who is assuming care after discharge; educating patients about their diagnosis, hospital course, and future medical needs; medication reconciliation; and, arrangement of postdischarge services such as follow‐up appointments. Additionally, the SHM has developed a discharge checklist14 (Table 5) and a template15 for the discharge summary, each to facilitate communication between stakeholders. The discharge summary is an obvious target for QI, as it is the most common vehicle for sharing patient information with the PCP and other healthcare providers. Essential elements of a discharge summary are content (Is key information captured?); format (Is content clear, concise, and accurate?); and delivery (Does the discharge summary reach the right people in a timely manner?).
| Project RED (Re‐Engineered Discharge)* | 2009 National Quality Forum (NQF) |
|---|---|
| Educate the patient about diagnosis during hospitalization | Prepare a written discharge plan |
| Make appointments for clinician follow‐up and postdischarge testing; identify and resolve barriers to follow‐up care | Prepare a written discharge summary |
| Talk to the patient about testing done in the hospital and who will follow up on results | Provide a discharge summary to a licensed clinician who will provide care after discharge |
| Organize postdischarge services; identify and resolve barriers to receiving services | Develop an institutional system to confirm receipt of the discharge summary by a licensed clinician |
| Medication reconciliation: counsel the patient about medications and identify barriers to adherence and compliance | |
| Reconcile the discharge plan with evidence‐based guidelines | |
| Educate the patient on problem‐solving strategies, including contacting the PCP | |
| Expedite transmission of the discharge summary to a licensed clinician and services that will be involved with the patient's care postdischarge | |
| Assess the patient's understanding of the discharge plan; ask patients to explain in their own words; identify and resolve barriers to understanding | |
| Provide the patient with a written summary detailing clinical course, follow‐up, and medication instructions | |
| Telephone the patient 2 to 3 days after discharge to review the plan and address problems | |
|
| Diagnoses |
| Detail location of MI and complications |
| Comorbidities |
| Note diabetes, results of lipid panel, hypertension, renal disease |
| Medications |
| Medication reconciliation |
| Note reason if core measure medications are not prescribed (beta blockers, antiplatelet therapies) |
| ACE/ARB, aspirin, beta‐blockers, statin, sublingual NTG, clopidogrel (include duration of therapy) |
| Titration of any medications |
| Procedures |
| Type, location of stent |
| Complications |
| If ECHO, include type, ejection fraction; provide copy of ECG if available |
| Follow‐up appointment |
| PCP, cardiologist, others such as cardiac rehab |
| Follow‐up testing |
| ETT (type, timeframe); ECHO; required lab work |
| Code status |
| Activity |
| Diet |
| Wound care (eg, groin) |
| Treatment course |
| Cognitive level |
| Discharge LDL |
| Discharge creatinine |
| INR if on warfarin |
| LFTs if on statin |
| Copy all providers |
Evidence indicates that the danger for patients at discharge is often related to medication reconciliation, adverse drug events (ADEs), and pending test results or testing needed after hospitalization.6 Errors affect a sizable proportion of patients, with 49% of patients in 1 study subject to at least 1 medical error within 2 months of discharge. This error was directly attributed to discontinuity during the transition from hospitalist care to the affiliated PCP practice. Errors were related to discharge medications, test results, or lack of PCP follow‐up on testing recommended by the inpatient provider (a work‐up error). Patients with a work‐up error were 6 times more likely to be rehospitalized in the 3 months following the first outpatient visit.16 Another study found that 41% of discharged patients had inhospital test results return after they were discharged and that PCPs were often unaware of these results. In some of these cases, test results required action, sometimes urgently.17
Most adverse events after discharge appear to be ADEs, with up to two‐thirds identified as preventable or ameliorable.1820 In a general medical population, Forster et al.20 found that ADEs resulted in significant injury in 71% of patients, serious injury in 13%, and life‐threatening injury in 16%. ADEs also resulted in 27% of patients requiring emergency care or readmission. Anticoagulants and cardiovascular medications were ranked high among medications associated with an ADE; failure to monitor medications was the most common cause of a preventable or ameliorable ADE, suggesting that ACS patients may be particularly at risk for an ADE following discharge. Patients had good recall of general drug information provided at discharge; patients who could not recall receiving specific information about ADEs were more likely to have an ADE. This is an area of opportunity for the hospitalist to improve patient safety during the transition to primary care. It is also of special concern for patients with ACS, given the complexity of medication regimens and the potential for serious drug reactions.
The risk of adverse events after discharge is higher in certain populations such as the elderly. Project BOOST (Better Outcomes for Older adults through Safe Transitions) is a QI initiative to improve patient care during discharge.21 BOOST aims to reduce 30‐day readmission rates for general medicine patients (especially older adults), improve patient satisfaction, improve communication between the institution and the PCP, identify high‐risk patients and mitigate risk, and improve patient and family education with a focus on patient‐specific risk factors.
BOOST uses a multidisciplinary 7P Screening tool to identify high‐risk patients, specifically through discharge planning and risk stratification.21 Seven areas are assessed: Problem medications, Punk (depression), Principal diagnosis, Polypharmacy, Poor health literacy, Patient support, and Prior hospitalization. For each, specific interventions are recommended to lower the risk of untoward events. 7P Screening applies to patients with ACS and can be an important step in identifying concerns at discharge, with the goals of promoting recovery and reducing the risk of readmission. The BOOST tool also includes a Universal Patient Discharge List reflective of those proposed by Project RED (Re‐Engineered Discharge) and the SHM. However, BOOST suggests additional steps to enhance communication: multidisciplinary rounds at discharge, direct communication with the PCP before discharge, phone contact with the patient or caregiver within 3 days of discharge, and phone numbers for hospital personnel familiar with the patient if the patient is unable to reach the PCP about an issue before the patient's first scheduled follow‐up visit. These steps recognize the value of direct communication between the hospitalist, the patient, and the PCP.
Overcoming Barriers to Communication Between the Hospitalist and the PCP
A successful transition from the hospital to primary care rests largely on the quality of communication between the hospitalist and the PCP. However, only 56% of PCPs expressed satisfaction with the communication they have with hospitalists22 and direct communication is infrequent.11 The PCPs surveyed desired direct and frequent communication, with three‐quarters preferring to speak with the hospitalist by phone at both the patient's admission and discharge.22
PCPs deemed discharge medications and discussion of the reasoning for medication changes and duration of treatment, diagnoses, physical findings, test results, follow‐up needs and plan details, and pending test results to be the most important shared information,11, 22 but this information is frequently unavailable. Details about pending test results at discharge were missed in 65% of summaries, followed by inhospital test results in 33% to 63%, discharge medications in 2% to 40%, and a description of the follow‐up plan in 2% to 43%. Late discharge summaries, some arriving after the first follow‐up visit, are also a barrier to quality care.11, 22, 23 Structured discharge summaries can yield organized and easily retrievable information, with the structure providing cues to include all necessary details.11 These may be computer‐generated vs. traditional dictated or handwritten summaries.6
Use of standardized instruments such as the SHM transitions tool can also help facilitate communication between providers. This tool provides a detailed checklist of recommended diagnostics and therapeutics for patients with ACS that should be considered when the patient is transferred and during discharge. This type of tool has been shown to enhance communication and alert multidisciplinary providers to address issues prior to discharge. A standardized toolkit consisting of a standard admission form, a facsimile to the PCP at admission, a worksheet to identify barriers at discharge, pharmacistphysician medication reconciliation, and predischarge planning appointments reportedly reduced the number of return visits to the ED within 3 days of discharge in an elderly general medicine population; at 30 days there were fewer ED visits and readmissions.24
It is worth noting that patients and their caregivers also have barriers to communication that hospitalists may be able to help address by discharge. These barriers include poor literacy, poor English proficiency, poor understanding of medical jargon, inadequate time with the clinician for questions and answers, poor cognition, highly complex information, and a diagnosis the patient may consider overwhelming.6 Specifically, patients with ACS demonstrated a poor comprehension of their medication regimen after discharge, manifested as either a delay or not filling of prescriptions, followed by poor adherence to the regimen.25 It is also helpful if financial barriers to medication use, either because of direct cost or restriction in outpatient formularies, are identified prior to discharge. Patients report that a follow‐up call from the clinician after discharge or pharmacist counseling before discharge, and use of a pillbox would remove some barriers to adherence. In teach‐back, patients are asked to repeat instructions in their own words, avoiding yes or no answers, thereby revealing gaps in understanding. This strategy checks patient comprehension and provides an opportunity for dialog if it is apparent that patients do not understand information related to their disease and recovery.
Case Study (cont)
Jose's PCP provided more information about his medical and medication history. His father died of a heart attack at 62 years old. Jose has smoked on and off for several years and has been poorly compliant with measures to reduce his risk for CHD such as diet, exercise, and taking statins. Jose may not comprehend the seriousness of his heart disease and how secondary preventive measures may reduce his risk of further events. His history of poor compliance raises concern that he will not persist with recommended ACS medications or antiplatelet regimens after discharge.
Impact of the Hospitalist on Long‐Term Outcomes
Evidenced‐based guidelines stress the need for aggressive modification of risk factors and treatment with antiplatelet, antihypertensive, and lipid‐lowering agents started during hospitalization and continued long‐term as part of secondary prevention strategies.7, 8 There is a missed opportunity for improving patient outcomes after ACS,26, 27 shown by the underuse of guideline‐recommended therapies (antiplatelet therapies such as clopidogrel and aspirin, beta blockers, angiotensin‐converting enzyme [ACE] inhibitors, and statins).28 More than one‐half of patients stopped evidence‐based medications without input from their providers, partly attributed to patients' perception that medication was not needed.26, 27, 29 In another study, 1 in 6 patients who received a DES delayed filling their antiplatelet prescription following discharge (median, 3 days; range, 1‐23 days). Patients who delayed filling the prescription were at increased risk of death or MI compared with patients who filled the prescription on the day of discharge. These findings underscore the importance of discharge planning and patient counseling to improve adherence to medications given at discharge.30 Through education, the hospitalist can directly influence patient and caregiver understanding of the benefit of ACS medications and their effect on long‐term outcomes.
Educating the patient and family about the nature, prognosis, and treatment of cardiac disease is equally important, and secondary prevention measures should be addressed prior to discharge. Prevention includes urging the patient to make therapeutic lifestyle choices such as smoking cessation (a core measure), maintaining a healthy diet, and regular exercise. Addressing these issues is important for the hospitalist and reinforced by the PCP, because adherence to behavioral changes after ACS has been shown to be poor and to directly impact outcomes. In 1 study, one‐third of smokers continued to smoke after 1 month and about one‐third of patients did not adhere to a recommended diet or exercise regimen. At 6 months, those patients who did quit smoking had a 43% lower risk of MI; compliance with the diet and exercise regimen lowered their risk of MI by 48%. Persistent smoking and nonadherence to diet and exercise resulted in an almost 4‐fold increased risk of MI, stroke, or death vs. never‐smokers who adhered to diet and exercise recommendations.31 This presents a clear opportunity for the hospitalist to intervene and affect change. Assessment of needed secondary prevention measures should occur in preparation for discharge, and an emphasis on patient teaching and communicating the plan to the PCP may overcome patient barriers to adhering to recommended lifestyle changes.
Case Study (cont)
After verbally describing his discharge medications to Jose and his family, and providing written patient materials, you ask Jose to explain why his prescribed dual antiplatelet therapy is important. He states it will help his occasional arthritis, because aspirin is one of the drugs, revealing that Jose lacks understanding of why he is taking the antiplatelet therapies or why they are important. A consult is requested from pharmacy for additional counseling. Because you have been in direct contact with the PCP, you call now to express your concerns, in addition to noting Jose's poor comprehension in the discharge summary.
Conclusion
Timely and accurate communication between the hospitalist and the PCP is a vital component of a safe transition from inpatient to primary care. This communication directly impacts the continuity of care, patient outcomes, patient and caregiver satisfaction, and use of healthcare resources. The role of the hospitalist is still evolving. Hospitalists will continue to have a pivotal role in transitions of care, and have a direct impact on the quality of the transition at discharge and patient outcomes after ACS. Hospitalists should be cognizant of gaps in care related to how information is generated, recorded, and shared between the inpatient setting and primary care, and should be proactive in identifying barriers and facilitating solutions. The hospitalist's responsibility for the patient does not end at the time of discharge but extends until the PCP assumes responsibility for patient care following hospitalization. We must make every reasonable effort to assure that our patients and their outpatient providers and caregivers are given all the tools necessary to complete and maintain the patient's therapy.
Acknowledgements
The author thanks Denise Erkkila, RPh for her editorial assistance in preparation of this manuscript.
- American Heart Association. Heart disease and stroke statistics‐2010 update. Dallas, Texas: American Heart Association;2010.
- ,,,,.One‐year costs of ischemic heart disease among patients with acute coronary syndromes: findings from a multi‐employer claims database.Curr Med Res Opin.2008;24:461–468.
- ,,, et al.Explaining the decrease in U.S. deaths from coronary disease, 1980‐2000.N Engl J Med.2007;356:2388–2398.
- ,,,,.Core competencies in hospital medicine: development and methodology.J Hosp Med.2006;1:48–56.
- Society of Hospital Medicine.Acute coronary syndrome.J Hosp Med.2006;1(suppl 1):2–3.
- ,,,.Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists.J Hosp Med.2007;2:314–323.
- ,,, et al.ACC/AHA 2007 guidelines for the management of patients with unstable angina/non ST‐elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non ST‐Elevation Myocardial Infarction): developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons: endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine.Circulation.2007;116:e148–e304.
- ,,, et al.2009 focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.2009;54:2205–2241.
- SHM Acute Coronary Syndrome Advisory Board.A guide for effective quality improvement: improving acute coronary syndrome care for hospitalized patients. Available at: http://www.hospitalmedicine.org.2010. Accessed July 2010.
- SHM ACS Transitions Workgroup.SHM ACS Transitions Tool. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/05_Transitions.cfm.2010. Accessed July 2010.
- ,,, et al.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- .Reengineering hospital discharge: a protocol to improve patient safety, reduce costs, and boost patient satisfaction.Am J Med Qual.2009;24:344–346.
- National Quality Forum.National Quality Forum (NQF) endorsed set of 34 safe practices. Available at: http://www.hfap.org/pdf/patient_safety.pdf.2009. Accessed July 2010.
- ,,, et al.Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- Society of Hospital Medicine.SHM Acute Coronary Syndrome (ACS) Discharge Planning Checklist. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/04_Discharge. cfm.2010. Accessed July 2010.
- ,,,.Medical errors related to discontinuity of care from an inpatient to an outpatient setting.J Gen Intern Med.2003;18:646–651.
- ,,, et al.Patient safety concerns arising from test results that return after hospital discharge.Ann Intern Med.2005;143:121–128.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- ,,, et al.Adverse events among medical patients after discharge from hospital.CMAJ.2004;170:345–349.
- ,,,,.Adverse drug events occurring following hospital discharge.J Gen Intern Med.2005;20:317–323.
- Society of Hospital Medicine.Boosting Care Transitions Resource Room. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_CareTransitions/CT_Home.cfm.2010. Accessed July 2010.
- ,,,.Primary care physician attitudes regarding communication with hospitalists.Dis Mon.2002;48:218–229.
- ,,,.Effect of discharge summary availability during post‐discharge visits on hospital readmission.J Gen Intern Med.2002;17:186–192.
- ,,, et al.A quality improvement intervention to facilitate the transition of older adults from three hospitals back to their homes.J Am Geriatr Soc.2009;57:1540–1546.
- ,,,.Medication use among inner‐city patients after hospital discharge: patient‐reported barriers and solutions.Mayo Clin Proc.2008;83:529–535.
- ,,, et al.Predictors of early discontinuation of evidence‐based medicine after acute coronary syndrome.Am J Cardiol.2009;104:175–181.
- ,,, et al.Optimal medical therapy at discharge in patients with acute coronary syndromes: temporal changes, characteristics, and 1‐year outcome.Am Heart J.2007;154:1108–1115.
- ,,.Reinforcing a continuum of care: in‐hospital initiation of long‐term secondary prevention following acute coronary syndromes.Cardiovasc Drugs Ther.2007;21:375–388.
- ,,, et al.Age and persistent use of cardiovascular medication after acute coronary syndrome: results from medication applied and sustained over time.J Am Geriatr Soc.2009;57:1990–1996.
- ,,, et al.Delays in filling clopidogrel prescription after hospital discharge and adverse outcomes after drug‐eluting stent implantation.Circ Cardiovasc Qual Outcomes.2010;3:261–266.
- ,,, et al.Association of diet, exercise, and smoking modification with risk of early cardiovascular events after acute coronary syndromes.Circulation.2010;121:750–758.
- American Heart Association. Heart disease and stroke statistics‐2010 update. Dallas, Texas: American Heart Association;2010.
- ,,,,.One‐year costs of ischemic heart disease among patients with acute coronary syndromes: findings from a multi‐employer claims database.Curr Med Res Opin.2008;24:461–468.
- ,,, et al.Explaining the decrease in U.S. deaths from coronary disease, 1980‐2000.N Engl J Med.2007;356:2388–2398.
- ,,,,.Core competencies in hospital medicine: development and methodology.J Hosp Med.2006;1:48–56.
- Society of Hospital Medicine.Acute coronary syndrome.J Hosp Med.2006;1(suppl 1):2–3.
- ,,,.Promoting effective transitions of care at hospital discharge: a review of key issues for hospitalists.J Hosp Med.2007;2:314–323.
- ,,, et al.ACC/AHA 2007 guidelines for the management of patients with unstable angina/non ST‐elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non ST‐Elevation Myocardial Infarction): developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons: endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine.Circulation.2007;116:e148–e304.
- ,,, et al.2009 focused updates: ACC/AHA guidelines for the management of patients with ST‐elevation myocardial infarction (updating the 2004 guideline and 2007 focused update) and ACC/AHA/SCAI guidelines on percutaneous coronary intervention (updating the 2005 guideline and 2007 focused update) a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.J Am Coll Cardiol.2009;54:2205–2241.
- SHM Acute Coronary Syndrome Advisory Board.A guide for effective quality improvement: improving acute coronary syndrome care for hospitalized patients. Available at: http://www.hospitalmedicine.org.2010. Accessed July 2010.
- SHM ACS Transitions Workgroup.SHM ACS Transitions Tool. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/05_Transitions.cfm.2010. Accessed July 2010.
- ,,, et al.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- .Reengineering hospital discharge: a protocol to improve patient safety, reduce costs, and boost patient satisfaction.Am J Med Qual.2009;24:344–346.
- National Quality Forum.National Quality Forum (NQF) endorsed set of 34 safe practices. Available at: http://www.hfap.org/pdf/patient_safety.pdf.2009. Accessed July 2010.
- ,,, et al.Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- Society of Hospital Medicine.SHM Acute Coronary Syndrome (ACS) Discharge Planning Checklist. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_ACS/html_ACS/12ClinicalTools/04_Discharge. cfm.2010. Accessed July 2010.
- ,,,.Medical errors related to discontinuity of care from an inpatient to an outpatient setting.J Gen Intern Med.2003;18:646–651.
- ,,, et al.Patient safety concerns arising from test results that return after hospital discharge.Ann Intern Med.2005;143:121–128.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- ,,, et al.Adverse events among medical patients after discharge from hospital.CMAJ.2004;170:345–349.
- ,,,,.Adverse drug events occurring following hospital discharge.J Gen Intern Med.2005;20:317–323.
- Society of Hospital Medicine.Boosting Care Transitions Resource Room. Available at: http://www.hospitalmedicine.org/ResourceRoomRedesign/RR_CareTransitions/CT_Home.cfm.2010. Accessed July 2010.
- ,,,.Primary care physician attitudes regarding communication with hospitalists.Dis Mon.2002;48:218–229.
- ,,,.Effect of discharge summary availability during post‐discharge visits on hospital readmission.J Gen Intern Med.2002;17:186–192.
- ,,, et al.A quality improvement intervention to facilitate the transition of older adults from three hospitals back to their homes.J Am Geriatr Soc.2009;57:1540–1546.
- ,,,.Medication use among inner‐city patients after hospital discharge: patient‐reported barriers and solutions.Mayo Clin Proc.2008;83:529–535.
- ,,, et al.Predictors of early discontinuation of evidence‐based medicine after acute coronary syndrome.Am J Cardiol.2009;104:175–181.
- ,,, et al.Optimal medical therapy at discharge in patients with acute coronary syndromes: temporal changes, characteristics, and 1‐year outcome.Am Heart J.2007;154:1108–1115.
- ,,.Reinforcing a continuum of care: in‐hospital initiation of long‐term secondary prevention following acute coronary syndromes.Cardiovasc Drugs Ther.2007;21:375–388.
- ,,, et al.Age and persistent use of cardiovascular medication after acute coronary syndrome: results from medication applied and sustained over time.J Am Geriatr Soc.2009;57:1990–1996.
- ,,, et al.Delays in filling clopidogrel prescription after hospital discharge and adverse outcomes after drug‐eluting stent implantation.Circ Cardiovasc Qual Outcomes.2010;3:261–266.
- ,,, et al.Association of diet, exercise, and smoking modification with risk of early cardiovascular events after acute coronary syndromes.Circulation.2010;121:750–758.
Causes of Unplanned ICU Transfers
Two national surveys indicate that 14% to 28% of patients admitted to intensive care units (ICU's) are unplanned transfers (i.e., moving a patient to the ICU from other areas in the hospital providing lower intensity care due to an unanticipated change in the patient's clinical status), and that the most common reason for unplanned transfers is respiratory insufficiency/failure.1, 2 Patients suffering adverse events during a hospitalization are more likely to have an unplanned ICU transfer and patients requiring unplanned transfers have a higher mortality.35 Accordingly, the Joint Commission has identified improved recognition and response to changes in a patient's condition as a national patient safety goal,6 and Rapid Response Teams (RRTs) have been advocated to deal with these changes,7 although recent studies question the effectiveness of RRTs.811
We sought to classify the causes of unplanned, in‐hospital transfers to a medical ICU (MICU) with the idea of identifying common problems in care that might be addressed by process improvement activities. We also sought to determine the fraction of patients requiring an unplanned MICU transfer that had evidence of clinical deterioration prior to the time of transfer and whether, in retrospect, different or earlier interventions might have prevented the transfer. Our hypotheses were that (1) most unplanned MICU transfers occurred as a result of errors in care, (2) most were preceded by clinical deterioration within 12 hours prior to the transfer, and (3) most were preventable.
Methods
We conducted a retrospective cohort study of patients transferring to the MICU from non‐ICU Medicine units at Denver Health, a university‐affiliated, public safety net hospital. All adult patients between 18 to 89 years of age, who were admitted to the Medicine service between June, 2005 and May, 2006 were included in the study. Exclusion criteria included patients who (1) transferred from outside hospitals, (2) transferred from nonMedicine units within Denver Health, (3) were admitted directly to the MICU from the emergency department (ED), (4) were prisoners, (5) were readmitted to the MICU during the same hospitalization, (6) were known to be pregnant, or (7) were planned MICU transfers following invasive procedures (eg, elective cardiac catheterization, defibrillator placement, ablations). Patients readmitted to the MICU were excluded because of the difficulty distinguishing between premature transfer from the MICU or potential problems in care that might have occurred prior to the time of transfer from those occurring during follow‐up care on the Medicine floor services.
Computerized medical records of eligible patients were searched for demographic information and for admitting and transfer diagnoses (with the latter being categorized using a taxonomy we developed for classifying unplanned transfers, Table 1). Three independent observers (all of whom were board certified in Internal Medicine and had been practicing as Hospitalists at our institution for a minimum of three years) retrospectively reviewed each patient's hospital record to determine the cause of the unplanned transfer using this taxonomy. All three also made a judgment as to whether deterioration was evident at any time within the 12 hours preceding the unplanned transfer on the basis of clinical criteria used as our hospital's rapid response triggers (Table 2). When clinical triggers were found, each of the reviewers independently judged whether the unplanned transfer might have been prevented had different or earlier interventions been instituted. Each reviewer was blinded to the results of the other two.
|
| 1. Errors in triage from the Emergency Department |
| A. Diagnostic errors (conditions that were overlooked at the time of admission but explained the chief complaint). |
| B. Inadequate assessment (new diagnosis established after more extensive evaluation that could have been performed at the time of admission). |
| C. Overlooked severity (patients meeting MICU admission criteria at the time of admission from the ED). |
| 2. Worsening of condition for which the patient was admitted |
| A. Errors with assessment or treatment (evaluation or treatment that was not thought to be standard of care for the admitting diagnosis). |
| 1. Delayed (could reasonably have been instituted earlier) |
| 2. Incorrect (not thought to represent standard of care) |
| 3. Inadequate (correct, but insufficient for the admitting diagnosis) |
| B. Spontaneous worsening (worsening of the problem for which the patients were admitted to the point of requiring MICU transfer for which no specific cause could be identified) |
| 3. Development of a new problem |
| A. Iatrogenic (thought to be caused by a diagnostic or therapeutic intervention) |
| B. Spontaneous (no specific cause could be identified) |
| 4. Critical laboratory values (laboratory values needing frequent monitoring of patient and/or blood draws) |
| A. Respiratory |
| Respiratory rate <8 or >28/minute |
| Acute change in oxygen saturation to <90% despite oxygen administration |
| Threatened airway |
| B. Cardiovascular |
| Acute change in systolic blood pressure to <90 mmHg |
| Acute, sustained increase in diastolic blood pressure to >110 mmHg |
| Acute change in heart rate to <50 or >120 beats/minute |
| New onset chest pain or chest pain different than on admission assessment |
| Acutely cold and pulseless extremity. |
| C. Neurological |
| Confusion, agitation or delirium |
| Unexplained lethargy/difficult to arouse |
| Difficulty speaking or swallowing |
| Acute change in pupillary response |
| New seizure |
| D. Other |
| Temperature >39.0 Celsius |
| Uncontrolled pain (if different than admission pain assessment) |
| Acute change in urine output <50 mL/4 hours |
| Acute bleeding (bleeding with a change in vitals, urine output or mental status) |
All analyses were done using SAS Enterprise Guide 4.1, SAS Institute, Cary, NC. Data are presented as mean (standard deviation [SD]). Interobserver agreement was measured by calculating a statistic. values were interpreted by using the guidelines suggested by Landis and colleagues.12 A chi‐square test was used to seek associations between baseline characteristics, reasons for MICU transfer and mortality. P < 0.05 was considered to be statistically significant. The Colorado Multiple Institutional Review Board approved the research protocol.
Results
Over the period of the study the Medicine floor services had 4468 admissions of which 152 met the inclusion criteria for having an unplanned MICU transfer (Table 3). The most common admitting diagnoses were heart failure (12%) and community acquired pneumonia (9%). The most common diagnoses to which the unplanned MICU transfers were attributed were respiratory failure (27%) and sepsis (9%) (Table 4). Seven cardiopulmonary arrests were successfully resuscitated and transferred to the MICU. Throughout the period of the study, no patients were admitted to non‐MICU units because the MICU was at full capacity. Additionally the investigators did not find any inordinate delays in transfer to the ICU while waiting for a bed.
| |
| Age (years) mean (SD) | 52 14 |
| Gender (male:female) | |
| Number | 95:57 |
| % | 63:37 |
| Race, n (%) | |
| White, non‐Hispanic | 54 (35) |
| White, Hispanic | 59 (39) |
| Black | 30 (20) |
| Other | 9 (6) |
| Primary language, n (%) | |
| English | 131 (86) |
| Spanish | 17 (11) |
| Other | 4 (3) |
| Length of stay prior to transfer (hours) (median, IQR) | 46, 89 |
| Admitting diagnosis, n (%) | |
| Acute decompensated heart failure (systolic/diastolic) | 18 (12) |
| Community acquired pneumonia | 13 (9) |
| Suspected acute coronary syndrome | 9 (6) |
| Delirium | 8 (5) |
| Acute kidney injury | 8 (5) |
| Abdominal pain | 8 (5) |
| Respiratory failure | 6 (4) |
| |
| Respiratory failure (cardiogenic/non‐cardiogenic) | 41 (27) |
| Sepsis | 14 (9) |
| Hypotension | 13 (9) |
| Gastrointestinal bleeding | 12 (8) |
| Tachyarrhythmia | 9 (6) |
| Cardiac arrest | 7 (5) |
| Hypertensive emergency | 7 (5) |
| Acute coronary syndrome | 7 (5) |
A total of 51 patients (34%) were transferred within the first 24 hours of admission. The most common diagnoses resulting in transfer in this group were respiratory failure, hypertensive emergency, hypotension, gastrointestinal bleed, and acute coronary syndrome. The remaining 101 patients (66%) were transferred from two to 15 days following admission for a variety of problems but respiratory failure was most common (34 patients, 22%).
Worsening of the problem for which the patients were initially admitted accounted for the unplanned transfers of 73 patients (48%) (Table 5). Development of a new problem unrelated to the admitting diagnosis accounted for the transfer in 59 patients (39%). Five patients were transferred to the ICU for a critical laboratory value that required a closer monitoring of the patient or needed more frequent lab draws that could not be achieved on the floor.
| Causes | n (%) |
|---|---|
| |
| 1. Errors in triage from the emergency department: | 15 (10) |
| A. Diagnostic errors: | 1 (0.7) |
| B. Inadequate assessment: | 0 (0) |
| C. Overlooked severity: | 14 (9) |
| 2. Worsening of condition for which the patient was admitted: | 73 (48) |
| A. Problems with assessment or treatment: | 5 (3) |
| 1. Delayed | 1 (0.7) |
| 2. Incorrect | 1 (0.7) |
| 3. Inadequate | 3 (2) |
| B. Spontaneous worsening | 68 (45) |
| 3. Development of a new problem | 59 (39) |
| A. Iatrogenic | 9 (6) |
| B. Spontaneous | 50 (33) |
| 4. Critical laboratory values | 5 (3) |
Errors in care were thought to be present in 29 patients (19% of the unplanned transfers). For 15 of these (52%) the error involved incorrect triage from the ED as 14 of the 15 patients met MICU admission criteria at the time they were triaged to non‐MICU units (Table 6). The remaining patient had a dissecting aortic aneurysm that was not considered while he was being evaluated for acute chest pain. All these patients were transferred to the ICU within 24 hours of their admission and the reviewers agreed that all could have been prevented if existing diagnostic and admission algorithms were followed.
|
| Hemodynamic instability requiring vasopressor agents, continued aggressive fluid resuscitation, or central venous/pulmonary artery catheter monitoring or balloon pump |
| Acute respiratory failure with ongoing or impending need for ventilatory support (either invasively or non‐ invasively). |
| Gastrointestinal bleeding meeting ICU admission criteria (>2 clinical risk factors and Rockall score >3 per Gastrointestinal Bleeding Protocol) |
| Cardiac chest pains associated with two of the three criteria |
| Ongoing ischemic chest pain |
| Enzyme elevation |
| ST segment depression <0.5 mm in 2 consecutives leads or transient ST‐segment elevation |
| Chest pain requiring IV nitroglycerin infusion. |
| Complex cardiac arrhythmia requiring close monitoring and/or intravenous infusion therapy |
| Temporary pacemaker. |
| Hypertensive crisis with end‐organ dysfunction or aortic dissection requiring intravenous treatment. |
| Massive hemoptysis (>500 cc/24 hours) |
| Acute neurological dysfunction requiring one of |
| ICP monitoring, |
| Acute respiratory failure with impending need for ventilatory support |
| Hourly neurological checks. |
| Status epilepticus |
| Post‐operative patients requiring hemodynamic monitoring/ventilator support of extensive nursing care. |
| Severe metabolic disorder or intoxication requiring frequent monitoring and/or intravenous infusion therapy that cannot be administered on a floor. |
| Multiple trauma, including severe head and spine trauma |
| Other indication (please specify) |
Of the remaining 14 patients thought to have errors in care, nine were classified as the development of a new, iatrogenic problem (ie, opiate or benzodiazepine overdose occurring during treatment for pain and/or anxiety in 3, volume overload in 2, insulin‐induced hypoglycemia, antibiotic associated reaction, ‐blocker overdose and acute renal failure from over‐diuresis in one each) and five occurred because the patient's admitting problem worsened because treatment was thought to be either delayed, incorrect, or inadequate (Table 5). The reviewers all agreed that the unplanned transfers could have been prevented in eight of the 14 patients who developed iatrogenic problems if existing algorithms were followed or if an earlier or different intervention had occurred. The reviewers did not agree about whether the unplanned transfer could have been prevented in one patient who developed an iatrogenic problem and in all five patients whose underlying condition worsened. Accordingly, in sum, the reviewers felt that 23 of the 152 unplanned transfers (15%) could have been prevented.
In addition to trying to determine how many of the unplanned MICU transfers could have been prevented, we also investigated the utility of rapid response triggers in alerting the physicians and nurses of impending deteriorations in status and whether earlier recognition of this deterioration might have prevented the transfers. Of the 152 unplanned transfers, 106 (70%) had one or more rapid response triggers within the preceding 12 hours. All three reviewers agreed and concluded that in 94 (89%) of these, the unplanned transfer could not have been prevented, even with different or earlier interventions. For five patients (5% of the 106) all reviewers agreed and concluded that earlier intervention might have averted the subsequent transfer. For the other seven patients (6%), no consensus was reached. If we assume that, for all of these latter seven, earlier or different intervention might have averted the unplanned transfer, a maximum of 12 unplanned transfers (11% of the 106) might have been prevented by having a system of care that employed regularly assessing rapid response triggers and acting on them when recognized.
The interobserver reliability for the three reviewers was moderate to almost perfect with = 0.60, 95% confidence interval (CI) (0.31, 0.88); = 0.90, 95% CI (0.71, 1); = 0.55, 95% CI (0.26, 0.84).
A total of 27 (18%) of the patients with unplanned transfers died in the MICU. During this same time period 91 of 1511 patients (6%) admitted directly from the ED to the MICU died (P < 0.05). Mortality was lower for patients transferred within 24 hours of admission compared to those transferred > 24 hours after admission (4% vs. 22% mortality, respectively, P < 0.05; 95% CI, 0.09‐0.89). We found no difference in mortality as a function of time of admission or time of transfer implying that differences in staffing, or the availability of various services, did not contribute to the unplanned transfers.
Discussion
The important findings of this study were that (1) 19% of unplanned, in‐hospital transfers from Medicine floor services to the MICU seemed to result from apparent errors in care, (2) 15% of the unplanned transfers were potentially preventable, (3) the majority of the errors in care involved inappropriate triage of patients from the ED to the non‐MICU units, (4) 106 (70%) of the patients requiring unplanned transfers developed rapid response criteria within 12 hours prior to the transfer, but on review of these (5) the transfer was thought to be preventable in only a maximum of 12 (11%).
We designed our study in part to find specific errors that commonly resulted in unplanned MICU transfers with the idea that, if these could be identified, they might be corrected, thereby improving care. Contrary to our hypothesis we found that only 29 (19%) of the unplanned transfers seemed to result from errors in care. Of these, however, half were attributable to overlooking that patients met our own institution's MICU admission criteria at the time they were triaged to non‐MICU units. This result is consistent with Walter et al.13 finding that while 88% of MICUs in academic health centers had written MICU admission criteria, only 25% used these criteria on a regular basis. Hospital mortality is likely lower for patients meeting MICU admission criteria when they are appropriately and expeditiously triaged.1418 Accordingly, developing mechanisms by which patients are routinely screened for meeting MICU admission criteria could and should reduce this source of error and improve patient outcomes.
Nine of the remaining 14 errors in care resulted from what the chart reviewers concluded was overly aggressive treatment; either excess fluid resuscitation or excess treatment of pain or anxiety. It is not clear that these represent correctable errors in care, however, as hypotensive patients require fluid resuscitation, and patients with pain or anxiety should receive analgesics or anxiolytics and it is not reasonable to expect that these interventions will be appropriately titrated in every instance. Nonetheless, our reviewers all agreed that, in eight of these patients, different interventions could have prevented the unplanned transfer.
Since 41 (27%) of the unplanned transfers were for respiratory failure, we reviewed each of these patients' records seeking evidence suggesting that the problem might have resulted from excessive use of fluids, narcotics, or anxiolytics. By retrospective analysis only six such cases could be identified. Most were due to worsening of the problem for which the patient was admitted.
Consistent with our hypothesis the majority of patients requiring unplanned MICU transfers (106/152, 70%) developed rapid response clinical triggers within the 12 hours preceding transfer, as has been previously demonstrated by Hillman et al.7 and others.8‐10, 19 Our reviewers tried to determine whether earlier or different interventions might have prevented the deterioration and the resulting unplanned transfer. Interestingly, in the large majority (94/106, 89%) they concluded that nothing different could have been done and that the transfer could not have been avoided. While this observation contrasts with our hypothesis, it is consistent with two studies questioning the utility of RRTs in preventing unplanned ICU transfers.9, 10 In addition some patients may ultimately need an ICU transfer despite receiving appropriate interventions as it is impossible to prevent an ICU transfer in every patient. Conversely, just because a patient meets a rapid response criteria does not mean that the patient needs a higher level of care or an ICU transfer as some can be safely managed on the floor.
Our study has a number of potential limitations. The data came from a single teaching hospital and we only assessed patients admitted to General Internal Medicine units and transferred to a MICU. Accordingly, our results might not generalize to other hospitals (teaching or nonteaching), to other services or to other types of ICUs. We found, however, that (1) unplanned transfers accounted for 10% of the total admissions to our MICU, a similar fraction as reported by Angus et al.1 in 2006; (2) respiratory failure/emnsufficiency and sepsis were the most common diagnoses leading to unplanned transfers as previously reported by Groeger et al.2 and Hillman et al.5; (3) mortality was increased in patients requiring unplanned transfer, as noted by Escarce and Kelley3 and Hillman et al.5; and (4) patients who were transferred to the MICU within 24 hours of admission had better outcomes than those who were transferred later, as reported by Goldhill et al.4 Accordingly, our patient population seems quite similar to others in the literature.
Since we did not use objective criteria to assign patients to each of the categories itemized in Table 5 we could have misclassified patients with respect to the cause for their unplanned MICU transfer. Despite this shortcoming, however, the scores among our independent reviewers were moderate to almost perfect suggesting misclassification did not occur commonly.
Our retrospective study design may have underestimated the utility of RRTs as we had no way of knowing the outcomes of patients who met rapid response criteria and had interventions that prevented unplanned MICU transfers.
In summary, approximately 15% of unplanned MICU transfers seem to be preventable and approximately one‐fifth seem to result from errors in care, the majority of which are errors in triage from the ED. While the large majority of unplanned transfers were preceded by clinical deterioration within the preceding 12 hours, manifested by the presence of rapid response triggers, the large majority of these do not seem to be preventable. From these findings we suggest that unplanned transfers could be reduced by more closely screening patients for the presence of defined MICU admission criteria at the time of admission from the ED, by recognizing that fluid resuscitation and control of pain and/or anxiety can have adverse effects and by monitoring patients receiving these interventions more closely.
- ,,,,,.Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). Critical care delivery in the United States: distribution of services and compliance with Leapfrog recommendations.Crit Care Med.2006;34(4):1016–1024.
- ,,, et al.Descriptive analysis of critical care units in the United States: patient characteristics and intensive care unit utilization.Crit Care Med.1993;21(2):279–291.
- ,.Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score.JAMA.1990;264(18):2389–2394.
- ,,,.The longer patients are in hospital before Intensive Care admission the higher their mortality.Intensive Care Med.2004;30(10):1908–1913.
- ,,,,, et al.Duration of life‐threatening antecedents prior to intensive care admission.Intensive Care Med.2002;28(11):1629–1634.
- Joint Commission on Accreditation of Healthcare Organizations. The Joint Commission Hospital Accreditation Program, National Patient Safety Goals, Goal 16; 2008. Available at: http://www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/08_hap_npsgs.htm. Accessed May2010.
- ,,, et al.MERIT study investigators. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial.Lancet.2005;365(9477):2091–2097.
- ,,,,,.Rapid response systems: a systematic review.Crit Care Med.2007;35(5):1238–1243.
- ,,,,,.Hospital‐wide code rates and mortality before and after implementation of a rapid response team.JAMA.2008;300(21):2506–2513.
- ,,,,.Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis.J Hosp Med.2007;2(6):422–432.
- ,,,,.Rapid response teams: a systematic review and meta‐analysis.Arch Intern Med.2010;170(1):18–26.
- ,.The measurement of observer agreement for categorical data.Biometrics.1977;33(1):159–174.
- ,,.How decisions are made to admit patients to medical intensive care units (MICUs): A survey of MICU directors at academic medical centers across the United States.Crit Care Med.2008;36:414–420.
- ,,.Mortality among appropriately referred patients refused admission to intensive‐care units.Lancet.1997;350:7–12.
- ,,,,,.Prospective evaluation of patients refused admission to an intensive care unit: triage, futility and outcome.Intensive Care Med.2001;27:1459–1465.
- ,,,,,for the Values, Ethics and Rationing in Critical Care (VERICC) Task Force. Rationing critical care beds: A systematic review.Crit Care Med.2004;32:1588–1597.
- ,,, et al.Survival of critically ill patients hospitalized in and out of intensive care.Crit Care Med.2007;35:449–457.
- ,,,,,for the DELAY‐ED study group. Impact of delayed transfer of critically ill patients form the emergency department to the intensive care unit.Crit Care Med.2007;35:1477–1483.
- ,,, et al.Antecedents to hospital deaths.Intern Med J.2001;31(6):343–348.
Two national surveys indicate that 14% to 28% of patients admitted to intensive care units (ICU's) are unplanned transfers (i.e., moving a patient to the ICU from other areas in the hospital providing lower intensity care due to an unanticipated change in the patient's clinical status), and that the most common reason for unplanned transfers is respiratory insufficiency/failure.1, 2 Patients suffering adverse events during a hospitalization are more likely to have an unplanned ICU transfer and patients requiring unplanned transfers have a higher mortality.35 Accordingly, the Joint Commission has identified improved recognition and response to changes in a patient's condition as a national patient safety goal,6 and Rapid Response Teams (RRTs) have been advocated to deal with these changes,7 although recent studies question the effectiveness of RRTs.811
We sought to classify the causes of unplanned, in‐hospital transfers to a medical ICU (MICU) with the idea of identifying common problems in care that might be addressed by process improvement activities. We also sought to determine the fraction of patients requiring an unplanned MICU transfer that had evidence of clinical deterioration prior to the time of transfer and whether, in retrospect, different or earlier interventions might have prevented the transfer. Our hypotheses were that (1) most unplanned MICU transfers occurred as a result of errors in care, (2) most were preceded by clinical deterioration within 12 hours prior to the transfer, and (3) most were preventable.
Methods
We conducted a retrospective cohort study of patients transferring to the MICU from non‐ICU Medicine units at Denver Health, a university‐affiliated, public safety net hospital. All adult patients between 18 to 89 years of age, who were admitted to the Medicine service between June, 2005 and May, 2006 were included in the study. Exclusion criteria included patients who (1) transferred from outside hospitals, (2) transferred from nonMedicine units within Denver Health, (3) were admitted directly to the MICU from the emergency department (ED), (4) were prisoners, (5) were readmitted to the MICU during the same hospitalization, (6) were known to be pregnant, or (7) were planned MICU transfers following invasive procedures (eg, elective cardiac catheterization, defibrillator placement, ablations). Patients readmitted to the MICU were excluded because of the difficulty distinguishing between premature transfer from the MICU or potential problems in care that might have occurred prior to the time of transfer from those occurring during follow‐up care on the Medicine floor services.
Computerized medical records of eligible patients were searched for demographic information and for admitting and transfer diagnoses (with the latter being categorized using a taxonomy we developed for classifying unplanned transfers, Table 1). Three independent observers (all of whom were board certified in Internal Medicine and had been practicing as Hospitalists at our institution for a minimum of three years) retrospectively reviewed each patient's hospital record to determine the cause of the unplanned transfer using this taxonomy. All three also made a judgment as to whether deterioration was evident at any time within the 12 hours preceding the unplanned transfer on the basis of clinical criteria used as our hospital's rapid response triggers (Table 2). When clinical triggers were found, each of the reviewers independently judged whether the unplanned transfer might have been prevented had different or earlier interventions been instituted. Each reviewer was blinded to the results of the other two.
|
| 1. Errors in triage from the Emergency Department |
| A. Diagnostic errors (conditions that were overlooked at the time of admission but explained the chief complaint). |
| B. Inadequate assessment (new diagnosis established after more extensive evaluation that could have been performed at the time of admission). |
| C. Overlooked severity (patients meeting MICU admission criteria at the time of admission from the ED). |
| 2. Worsening of condition for which the patient was admitted |
| A. Errors with assessment or treatment (evaluation or treatment that was not thought to be standard of care for the admitting diagnosis). |
| 1. Delayed (could reasonably have been instituted earlier) |
| 2. Incorrect (not thought to represent standard of care) |
| 3. Inadequate (correct, but insufficient for the admitting diagnosis) |
| B. Spontaneous worsening (worsening of the problem for which the patients were admitted to the point of requiring MICU transfer for which no specific cause could be identified) |
| 3. Development of a new problem |
| A. Iatrogenic (thought to be caused by a diagnostic or therapeutic intervention) |
| B. Spontaneous (no specific cause could be identified) |
| 4. Critical laboratory values (laboratory values needing frequent monitoring of patient and/or blood draws) |
| A. Respiratory |
| Respiratory rate <8 or >28/minute |
| Acute change in oxygen saturation to <90% despite oxygen administration |
| Threatened airway |
| B. Cardiovascular |
| Acute change in systolic blood pressure to <90 mmHg |
| Acute, sustained increase in diastolic blood pressure to >110 mmHg |
| Acute change in heart rate to <50 or >120 beats/minute |
| New onset chest pain or chest pain different than on admission assessment |
| Acutely cold and pulseless extremity. |
| C. Neurological |
| Confusion, agitation or delirium |
| Unexplained lethargy/difficult to arouse |
| Difficulty speaking or swallowing |
| Acute change in pupillary response |
| New seizure |
| D. Other |
| Temperature >39.0 Celsius |
| Uncontrolled pain (if different than admission pain assessment) |
| Acute change in urine output <50 mL/4 hours |
| Acute bleeding (bleeding with a change in vitals, urine output or mental status) |
All analyses were done using SAS Enterprise Guide 4.1, SAS Institute, Cary, NC. Data are presented as mean (standard deviation [SD]). Interobserver agreement was measured by calculating a statistic. values were interpreted by using the guidelines suggested by Landis and colleagues.12 A chi‐square test was used to seek associations between baseline characteristics, reasons for MICU transfer and mortality. P < 0.05 was considered to be statistically significant. The Colorado Multiple Institutional Review Board approved the research protocol.
Results
Over the period of the study the Medicine floor services had 4468 admissions of which 152 met the inclusion criteria for having an unplanned MICU transfer (Table 3). The most common admitting diagnoses were heart failure (12%) and community acquired pneumonia (9%). The most common diagnoses to which the unplanned MICU transfers were attributed were respiratory failure (27%) and sepsis (9%) (Table 4). Seven cardiopulmonary arrests were successfully resuscitated and transferred to the MICU. Throughout the period of the study, no patients were admitted to non‐MICU units because the MICU was at full capacity. Additionally the investigators did not find any inordinate delays in transfer to the ICU while waiting for a bed.
| |
| Age (years) mean (SD) | 52 14 |
| Gender (male:female) | |
| Number | 95:57 |
| % | 63:37 |
| Race, n (%) | |
| White, non‐Hispanic | 54 (35) |
| White, Hispanic | 59 (39) |
| Black | 30 (20) |
| Other | 9 (6) |
| Primary language, n (%) | |
| English | 131 (86) |
| Spanish | 17 (11) |
| Other | 4 (3) |
| Length of stay prior to transfer (hours) (median, IQR) | 46, 89 |
| Admitting diagnosis, n (%) | |
| Acute decompensated heart failure (systolic/diastolic) | 18 (12) |
| Community acquired pneumonia | 13 (9) |
| Suspected acute coronary syndrome | 9 (6) |
| Delirium | 8 (5) |
| Acute kidney injury | 8 (5) |
| Abdominal pain | 8 (5) |
| Respiratory failure | 6 (4) |
| |
| Respiratory failure (cardiogenic/non‐cardiogenic) | 41 (27) |
| Sepsis | 14 (9) |
| Hypotension | 13 (9) |
| Gastrointestinal bleeding | 12 (8) |
| Tachyarrhythmia | 9 (6) |
| Cardiac arrest | 7 (5) |
| Hypertensive emergency | 7 (5) |
| Acute coronary syndrome | 7 (5) |
A total of 51 patients (34%) were transferred within the first 24 hours of admission. The most common diagnoses resulting in transfer in this group were respiratory failure, hypertensive emergency, hypotension, gastrointestinal bleed, and acute coronary syndrome. The remaining 101 patients (66%) were transferred from two to 15 days following admission for a variety of problems but respiratory failure was most common (34 patients, 22%).
Worsening of the problem for which the patients were initially admitted accounted for the unplanned transfers of 73 patients (48%) (Table 5). Development of a new problem unrelated to the admitting diagnosis accounted for the transfer in 59 patients (39%). Five patients were transferred to the ICU for a critical laboratory value that required a closer monitoring of the patient or needed more frequent lab draws that could not be achieved on the floor.
| Causes | n (%) |
|---|---|
| |
| 1. Errors in triage from the emergency department: | 15 (10) |
| A. Diagnostic errors: | 1 (0.7) |
| B. Inadequate assessment: | 0 (0) |
| C. Overlooked severity: | 14 (9) |
| 2. Worsening of condition for which the patient was admitted: | 73 (48) |
| A. Problems with assessment or treatment: | 5 (3) |
| 1. Delayed | 1 (0.7) |
| 2. Incorrect | 1 (0.7) |
| 3. Inadequate | 3 (2) |
| B. Spontaneous worsening | 68 (45) |
| 3. Development of a new problem | 59 (39) |
| A. Iatrogenic | 9 (6) |
| B. Spontaneous | 50 (33) |
| 4. Critical laboratory values | 5 (3) |
Errors in care were thought to be present in 29 patients (19% of the unplanned transfers). For 15 of these (52%) the error involved incorrect triage from the ED as 14 of the 15 patients met MICU admission criteria at the time they were triaged to non‐MICU units (Table 6). The remaining patient had a dissecting aortic aneurysm that was not considered while he was being evaluated for acute chest pain. All these patients were transferred to the ICU within 24 hours of their admission and the reviewers agreed that all could have been prevented if existing diagnostic and admission algorithms were followed.
|
| Hemodynamic instability requiring vasopressor agents, continued aggressive fluid resuscitation, or central venous/pulmonary artery catheter monitoring or balloon pump |
| Acute respiratory failure with ongoing or impending need for ventilatory support (either invasively or non‐ invasively). |
| Gastrointestinal bleeding meeting ICU admission criteria (>2 clinical risk factors and Rockall score >3 per Gastrointestinal Bleeding Protocol) |
| Cardiac chest pains associated with two of the three criteria |
| Ongoing ischemic chest pain |
| Enzyme elevation |
| ST segment depression <0.5 mm in 2 consecutives leads or transient ST‐segment elevation |
| Chest pain requiring IV nitroglycerin infusion. |
| Complex cardiac arrhythmia requiring close monitoring and/or intravenous infusion therapy |
| Temporary pacemaker. |
| Hypertensive crisis with end‐organ dysfunction or aortic dissection requiring intravenous treatment. |
| Massive hemoptysis (>500 cc/24 hours) |
| Acute neurological dysfunction requiring one of |
| ICP monitoring, |
| Acute respiratory failure with impending need for ventilatory support |
| Hourly neurological checks. |
| Status epilepticus |
| Post‐operative patients requiring hemodynamic monitoring/ventilator support of extensive nursing care. |
| Severe metabolic disorder or intoxication requiring frequent monitoring and/or intravenous infusion therapy that cannot be administered on a floor. |
| Multiple trauma, including severe head and spine trauma |
| Other indication (please specify) |
Of the remaining 14 patients thought to have errors in care, nine were classified as the development of a new, iatrogenic problem (ie, opiate or benzodiazepine overdose occurring during treatment for pain and/or anxiety in 3, volume overload in 2, insulin‐induced hypoglycemia, antibiotic associated reaction, ‐blocker overdose and acute renal failure from over‐diuresis in one each) and five occurred because the patient's admitting problem worsened because treatment was thought to be either delayed, incorrect, or inadequate (Table 5). The reviewers all agreed that the unplanned transfers could have been prevented in eight of the 14 patients who developed iatrogenic problems if existing algorithms were followed or if an earlier or different intervention had occurred. The reviewers did not agree about whether the unplanned transfer could have been prevented in one patient who developed an iatrogenic problem and in all five patients whose underlying condition worsened. Accordingly, in sum, the reviewers felt that 23 of the 152 unplanned transfers (15%) could have been prevented.
In addition to trying to determine how many of the unplanned MICU transfers could have been prevented, we also investigated the utility of rapid response triggers in alerting the physicians and nurses of impending deteriorations in status and whether earlier recognition of this deterioration might have prevented the transfers. Of the 152 unplanned transfers, 106 (70%) had one or more rapid response triggers within the preceding 12 hours. All three reviewers agreed and concluded that in 94 (89%) of these, the unplanned transfer could not have been prevented, even with different or earlier interventions. For five patients (5% of the 106) all reviewers agreed and concluded that earlier intervention might have averted the subsequent transfer. For the other seven patients (6%), no consensus was reached. If we assume that, for all of these latter seven, earlier or different intervention might have averted the unplanned transfer, a maximum of 12 unplanned transfers (11% of the 106) might have been prevented by having a system of care that employed regularly assessing rapid response triggers and acting on them when recognized.
The interobserver reliability for the three reviewers was moderate to almost perfect with = 0.60, 95% confidence interval (CI) (0.31, 0.88); = 0.90, 95% CI (0.71, 1); = 0.55, 95% CI (0.26, 0.84).
A total of 27 (18%) of the patients with unplanned transfers died in the MICU. During this same time period 91 of 1511 patients (6%) admitted directly from the ED to the MICU died (P < 0.05). Mortality was lower for patients transferred within 24 hours of admission compared to those transferred > 24 hours after admission (4% vs. 22% mortality, respectively, P < 0.05; 95% CI, 0.09‐0.89). We found no difference in mortality as a function of time of admission or time of transfer implying that differences in staffing, or the availability of various services, did not contribute to the unplanned transfers.
Discussion
The important findings of this study were that (1) 19% of unplanned, in‐hospital transfers from Medicine floor services to the MICU seemed to result from apparent errors in care, (2) 15% of the unplanned transfers were potentially preventable, (3) the majority of the errors in care involved inappropriate triage of patients from the ED to the non‐MICU units, (4) 106 (70%) of the patients requiring unplanned transfers developed rapid response criteria within 12 hours prior to the transfer, but on review of these (5) the transfer was thought to be preventable in only a maximum of 12 (11%).
We designed our study in part to find specific errors that commonly resulted in unplanned MICU transfers with the idea that, if these could be identified, they might be corrected, thereby improving care. Contrary to our hypothesis we found that only 29 (19%) of the unplanned transfers seemed to result from errors in care. Of these, however, half were attributable to overlooking that patients met our own institution's MICU admission criteria at the time they were triaged to non‐MICU units. This result is consistent with Walter et al.13 finding that while 88% of MICUs in academic health centers had written MICU admission criteria, only 25% used these criteria on a regular basis. Hospital mortality is likely lower for patients meeting MICU admission criteria when they are appropriately and expeditiously triaged.1418 Accordingly, developing mechanisms by which patients are routinely screened for meeting MICU admission criteria could and should reduce this source of error and improve patient outcomes.
Nine of the remaining 14 errors in care resulted from what the chart reviewers concluded was overly aggressive treatment; either excess fluid resuscitation or excess treatment of pain or anxiety. It is not clear that these represent correctable errors in care, however, as hypotensive patients require fluid resuscitation, and patients with pain or anxiety should receive analgesics or anxiolytics and it is not reasonable to expect that these interventions will be appropriately titrated in every instance. Nonetheless, our reviewers all agreed that, in eight of these patients, different interventions could have prevented the unplanned transfer.
Since 41 (27%) of the unplanned transfers were for respiratory failure, we reviewed each of these patients' records seeking evidence suggesting that the problem might have resulted from excessive use of fluids, narcotics, or anxiolytics. By retrospective analysis only six such cases could be identified. Most were due to worsening of the problem for which the patient was admitted.
Consistent with our hypothesis the majority of patients requiring unplanned MICU transfers (106/152, 70%) developed rapid response clinical triggers within the 12 hours preceding transfer, as has been previously demonstrated by Hillman et al.7 and others.8‐10, 19 Our reviewers tried to determine whether earlier or different interventions might have prevented the deterioration and the resulting unplanned transfer. Interestingly, in the large majority (94/106, 89%) they concluded that nothing different could have been done and that the transfer could not have been avoided. While this observation contrasts with our hypothesis, it is consistent with two studies questioning the utility of RRTs in preventing unplanned ICU transfers.9, 10 In addition some patients may ultimately need an ICU transfer despite receiving appropriate interventions as it is impossible to prevent an ICU transfer in every patient. Conversely, just because a patient meets a rapid response criteria does not mean that the patient needs a higher level of care or an ICU transfer as some can be safely managed on the floor.
Our study has a number of potential limitations. The data came from a single teaching hospital and we only assessed patients admitted to General Internal Medicine units and transferred to a MICU. Accordingly, our results might not generalize to other hospitals (teaching or nonteaching), to other services or to other types of ICUs. We found, however, that (1) unplanned transfers accounted for 10% of the total admissions to our MICU, a similar fraction as reported by Angus et al.1 in 2006; (2) respiratory failure/emnsufficiency and sepsis were the most common diagnoses leading to unplanned transfers as previously reported by Groeger et al.2 and Hillman et al.5; (3) mortality was increased in patients requiring unplanned transfer, as noted by Escarce and Kelley3 and Hillman et al.5; and (4) patients who were transferred to the MICU within 24 hours of admission had better outcomes than those who were transferred later, as reported by Goldhill et al.4 Accordingly, our patient population seems quite similar to others in the literature.
Since we did not use objective criteria to assign patients to each of the categories itemized in Table 5 we could have misclassified patients with respect to the cause for their unplanned MICU transfer. Despite this shortcoming, however, the scores among our independent reviewers were moderate to almost perfect suggesting misclassification did not occur commonly.
Our retrospective study design may have underestimated the utility of RRTs as we had no way of knowing the outcomes of patients who met rapid response criteria and had interventions that prevented unplanned MICU transfers.
In summary, approximately 15% of unplanned MICU transfers seem to be preventable and approximately one‐fifth seem to result from errors in care, the majority of which are errors in triage from the ED. While the large majority of unplanned transfers were preceded by clinical deterioration within the preceding 12 hours, manifested by the presence of rapid response triggers, the large majority of these do not seem to be preventable. From these findings we suggest that unplanned transfers could be reduced by more closely screening patients for the presence of defined MICU admission criteria at the time of admission from the ED, by recognizing that fluid resuscitation and control of pain and/or anxiety can have adverse effects and by monitoring patients receiving these interventions more closely.
Two national surveys indicate that 14% to 28% of patients admitted to intensive care units (ICU's) are unplanned transfers (i.e., moving a patient to the ICU from other areas in the hospital providing lower intensity care due to an unanticipated change in the patient's clinical status), and that the most common reason for unplanned transfers is respiratory insufficiency/failure.1, 2 Patients suffering adverse events during a hospitalization are more likely to have an unplanned ICU transfer and patients requiring unplanned transfers have a higher mortality.35 Accordingly, the Joint Commission has identified improved recognition and response to changes in a patient's condition as a national patient safety goal,6 and Rapid Response Teams (RRTs) have been advocated to deal with these changes,7 although recent studies question the effectiveness of RRTs.811
We sought to classify the causes of unplanned, in‐hospital transfers to a medical ICU (MICU) with the idea of identifying common problems in care that might be addressed by process improvement activities. We also sought to determine the fraction of patients requiring an unplanned MICU transfer that had evidence of clinical deterioration prior to the time of transfer and whether, in retrospect, different or earlier interventions might have prevented the transfer. Our hypotheses were that (1) most unplanned MICU transfers occurred as a result of errors in care, (2) most were preceded by clinical deterioration within 12 hours prior to the transfer, and (3) most were preventable.
Methods
We conducted a retrospective cohort study of patients transferring to the MICU from non‐ICU Medicine units at Denver Health, a university‐affiliated, public safety net hospital. All adult patients between 18 to 89 years of age, who were admitted to the Medicine service between June, 2005 and May, 2006 were included in the study. Exclusion criteria included patients who (1) transferred from outside hospitals, (2) transferred from nonMedicine units within Denver Health, (3) were admitted directly to the MICU from the emergency department (ED), (4) were prisoners, (5) were readmitted to the MICU during the same hospitalization, (6) were known to be pregnant, or (7) were planned MICU transfers following invasive procedures (eg, elective cardiac catheterization, defibrillator placement, ablations). Patients readmitted to the MICU were excluded because of the difficulty distinguishing between premature transfer from the MICU or potential problems in care that might have occurred prior to the time of transfer from those occurring during follow‐up care on the Medicine floor services.
Computerized medical records of eligible patients were searched for demographic information and for admitting and transfer diagnoses (with the latter being categorized using a taxonomy we developed for classifying unplanned transfers, Table 1). Three independent observers (all of whom were board certified in Internal Medicine and had been practicing as Hospitalists at our institution for a minimum of three years) retrospectively reviewed each patient's hospital record to determine the cause of the unplanned transfer using this taxonomy. All three also made a judgment as to whether deterioration was evident at any time within the 12 hours preceding the unplanned transfer on the basis of clinical criteria used as our hospital's rapid response triggers (Table 2). When clinical triggers were found, each of the reviewers independently judged whether the unplanned transfer might have been prevented had different or earlier interventions been instituted. Each reviewer was blinded to the results of the other two.
|
| 1. Errors in triage from the Emergency Department |
| A. Diagnostic errors (conditions that were overlooked at the time of admission but explained the chief complaint). |
| B. Inadequate assessment (new diagnosis established after more extensive evaluation that could have been performed at the time of admission). |
| C. Overlooked severity (patients meeting MICU admission criteria at the time of admission from the ED). |
| 2. Worsening of condition for which the patient was admitted |
| A. Errors with assessment or treatment (evaluation or treatment that was not thought to be standard of care for the admitting diagnosis). |
| 1. Delayed (could reasonably have been instituted earlier) |
| 2. Incorrect (not thought to represent standard of care) |
| 3. Inadequate (correct, but insufficient for the admitting diagnosis) |
| B. Spontaneous worsening (worsening of the problem for which the patients were admitted to the point of requiring MICU transfer for which no specific cause could be identified) |
| 3. Development of a new problem |
| A. Iatrogenic (thought to be caused by a diagnostic or therapeutic intervention) |
| B. Spontaneous (no specific cause could be identified) |
| 4. Critical laboratory values (laboratory values needing frequent monitoring of patient and/or blood draws) |
| A. Respiratory |
| Respiratory rate <8 or >28/minute |
| Acute change in oxygen saturation to <90% despite oxygen administration |
| Threatened airway |
| B. Cardiovascular |
| Acute change in systolic blood pressure to <90 mmHg |
| Acute, sustained increase in diastolic blood pressure to >110 mmHg |
| Acute change in heart rate to <50 or >120 beats/minute |
| New onset chest pain or chest pain different than on admission assessment |
| Acutely cold and pulseless extremity. |
| C. Neurological |
| Confusion, agitation or delirium |
| Unexplained lethargy/difficult to arouse |
| Difficulty speaking or swallowing |
| Acute change in pupillary response |
| New seizure |
| D. Other |
| Temperature >39.0 Celsius |
| Uncontrolled pain (if different than admission pain assessment) |
| Acute change in urine output <50 mL/4 hours |
| Acute bleeding (bleeding with a change in vitals, urine output or mental status) |
All analyses were done using SAS Enterprise Guide 4.1, SAS Institute, Cary, NC. Data are presented as mean (standard deviation [SD]). Interobserver agreement was measured by calculating a statistic. values were interpreted by using the guidelines suggested by Landis and colleagues.12 A chi‐square test was used to seek associations between baseline characteristics, reasons for MICU transfer and mortality. P < 0.05 was considered to be statistically significant. The Colorado Multiple Institutional Review Board approved the research protocol.
Results
Over the period of the study the Medicine floor services had 4468 admissions of which 152 met the inclusion criteria for having an unplanned MICU transfer (Table 3). The most common admitting diagnoses were heart failure (12%) and community acquired pneumonia (9%). The most common diagnoses to which the unplanned MICU transfers were attributed were respiratory failure (27%) and sepsis (9%) (Table 4). Seven cardiopulmonary arrests were successfully resuscitated and transferred to the MICU. Throughout the period of the study, no patients were admitted to non‐MICU units because the MICU was at full capacity. Additionally the investigators did not find any inordinate delays in transfer to the ICU while waiting for a bed.
| |
| Age (years) mean (SD) | 52 14 |
| Gender (male:female) | |
| Number | 95:57 |
| % | 63:37 |
| Race, n (%) | |
| White, non‐Hispanic | 54 (35) |
| White, Hispanic | 59 (39) |
| Black | 30 (20) |
| Other | 9 (6) |
| Primary language, n (%) | |
| English | 131 (86) |
| Spanish | 17 (11) |
| Other | 4 (3) |
| Length of stay prior to transfer (hours) (median, IQR) | 46, 89 |
| Admitting diagnosis, n (%) | |
| Acute decompensated heart failure (systolic/diastolic) | 18 (12) |
| Community acquired pneumonia | 13 (9) |
| Suspected acute coronary syndrome | 9 (6) |
| Delirium | 8 (5) |
| Acute kidney injury | 8 (5) |
| Abdominal pain | 8 (5) |
| Respiratory failure | 6 (4) |
| |
| Respiratory failure (cardiogenic/non‐cardiogenic) | 41 (27) |
| Sepsis | 14 (9) |
| Hypotension | 13 (9) |
| Gastrointestinal bleeding | 12 (8) |
| Tachyarrhythmia | 9 (6) |
| Cardiac arrest | 7 (5) |
| Hypertensive emergency | 7 (5) |
| Acute coronary syndrome | 7 (5) |
A total of 51 patients (34%) were transferred within the first 24 hours of admission. The most common diagnoses resulting in transfer in this group were respiratory failure, hypertensive emergency, hypotension, gastrointestinal bleed, and acute coronary syndrome. The remaining 101 patients (66%) were transferred from two to 15 days following admission for a variety of problems but respiratory failure was most common (34 patients, 22%).
Worsening of the problem for which the patients were initially admitted accounted for the unplanned transfers of 73 patients (48%) (Table 5). Development of a new problem unrelated to the admitting diagnosis accounted for the transfer in 59 patients (39%). Five patients were transferred to the ICU for a critical laboratory value that required a closer monitoring of the patient or needed more frequent lab draws that could not be achieved on the floor.
| Causes | n (%) |
|---|---|
| |
| 1. Errors in triage from the emergency department: | 15 (10) |
| A. Diagnostic errors: | 1 (0.7) |
| B. Inadequate assessment: | 0 (0) |
| C. Overlooked severity: | 14 (9) |
| 2. Worsening of condition for which the patient was admitted: | 73 (48) |
| A. Problems with assessment or treatment: | 5 (3) |
| 1. Delayed | 1 (0.7) |
| 2. Incorrect | 1 (0.7) |
| 3. Inadequate | 3 (2) |
| B. Spontaneous worsening | 68 (45) |
| 3. Development of a new problem | 59 (39) |
| A. Iatrogenic | 9 (6) |
| B. Spontaneous | 50 (33) |
| 4. Critical laboratory values | 5 (3) |
Errors in care were thought to be present in 29 patients (19% of the unplanned transfers). For 15 of these (52%) the error involved incorrect triage from the ED as 14 of the 15 patients met MICU admission criteria at the time they were triaged to non‐MICU units (Table 6). The remaining patient had a dissecting aortic aneurysm that was not considered while he was being evaluated for acute chest pain. All these patients were transferred to the ICU within 24 hours of their admission and the reviewers agreed that all could have been prevented if existing diagnostic and admission algorithms were followed.
|
| Hemodynamic instability requiring vasopressor agents, continued aggressive fluid resuscitation, or central venous/pulmonary artery catheter monitoring or balloon pump |
| Acute respiratory failure with ongoing or impending need for ventilatory support (either invasively or non‐ invasively). |
| Gastrointestinal bleeding meeting ICU admission criteria (>2 clinical risk factors and Rockall score >3 per Gastrointestinal Bleeding Protocol) |
| Cardiac chest pains associated with two of the three criteria |
| Ongoing ischemic chest pain |
| Enzyme elevation |
| ST segment depression <0.5 mm in 2 consecutives leads or transient ST‐segment elevation |
| Chest pain requiring IV nitroglycerin infusion. |
| Complex cardiac arrhythmia requiring close monitoring and/or intravenous infusion therapy |
| Temporary pacemaker. |
| Hypertensive crisis with end‐organ dysfunction or aortic dissection requiring intravenous treatment. |
| Massive hemoptysis (>500 cc/24 hours) |
| Acute neurological dysfunction requiring one of |
| ICP monitoring, |
| Acute respiratory failure with impending need for ventilatory support |
| Hourly neurological checks. |
| Status epilepticus |
| Post‐operative patients requiring hemodynamic monitoring/ventilator support of extensive nursing care. |
| Severe metabolic disorder or intoxication requiring frequent monitoring and/or intravenous infusion therapy that cannot be administered on a floor. |
| Multiple trauma, including severe head and spine trauma |
| Other indication (please specify) |
Of the remaining 14 patients thought to have errors in care, nine were classified as the development of a new, iatrogenic problem (ie, opiate or benzodiazepine overdose occurring during treatment for pain and/or anxiety in 3, volume overload in 2, insulin‐induced hypoglycemia, antibiotic associated reaction, ‐blocker overdose and acute renal failure from over‐diuresis in one each) and five occurred because the patient's admitting problem worsened because treatment was thought to be either delayed, incorrect, or inadequate (Table 5). The reviewers all agreed that the unplanned transfers could have been prevented in eight of the 14 patients who developed iatrogenic problems if existing algorithms were followed or if an earlier or different intervention had occurred. The reviewers did not agree about whether the unplanned transfer could have been prevented in one patient who developed an iatrogenic problem and in all five patients whose underlying condition worsened. Accordingly, in sum, the reviewers felt that 23 of the 152 unplanned transfers (15%) could have been prevented.
In addition to trying to determine how many of the unplanned MICU transfers could have been prevented, we also investigated the utility of rapid response triggers in alerting the physicians and nurses of impending deteriorations in status and whether earlier recognition of this deterioration might have prevented the transfers. Of the 152 unplanned transfers, 106 (70%) had one or more rapid response triggers within the preceding 12 hours. All three reviewers agreed and concluded that in 94 (89%) of these, the unplanned transfer could not have been prevented, even with different or earlier interventions. For five patients (5% of the 106) all reviewers agreed and concluded that earlier intervention might have averted the subsequent transfer. For the other seven patients (6%), no consensus was reached. If we assume that, for all of these latter seven, earlier or different intervention might have averted the unplanned transfer, a maximum of 12 unplanned transfers (11% of the 106) might have been prevented by having a system of care that employed regularly assessing rapid response triggers and acting on them when recognized.
The interobserver reliability for the three reviewers was moderate to almost perfect with = 0.60, 95% confidence interval (CI) (0.31, 0.88); = 0.90, 95% CI (0.71, 1); = 0.55, 95% CI (0.26, 0.84).
A total of 27 (18%) of the patients with unplanned transfers died in the MICU. During this same time period 91 of 1511 patients (6%) admitted directly from the ED to the MICU died (P < 0.05). Mortality was lower for patients transferred within 24 hours of admission compared to those transferred > 24 hours after admission (4% vs. 22% mortality, respectively, P < 0.05; 95% CI, 0.09‐0.89). We found no difference in mortality as a function of time of admission or time of transfer implying that differences in staffing, or the availability of various services, did not contribute to the unplanned transfers.
Discussion
The important findings of this study were that (1) 19% of unplanned, in‐hospital transfers from Medicine floor services to the MICU seemed to result from apparent errors in care, (2) 15% of the unplanned transfers were potentially preventable, (3) the majority of the errors in care involved inappropriate triage of patients from the ED to the non‐MICU units, (4) 106 (70%) of the patients requiring unplanned transfers developed rapid response criteria within 12 hours prior to the transfer, but on review of these (5) the transfer was thought to be preventable in only a maximum of 12 (11%).
We designed our study in part to find specific errors that commonly resulted in unplanned MICU transfers with the idea that, if these could be identified, they might be corrected, thereby improving care. Contrary to our hypothesis we found that only 29 (19%) of the unplanned transfers seemed to result from errors in care. Of these, however, half were attributable to overlooking that patients met our own institution's MICU admission criteria at the time they were triaged to non‐MICU units. This result is consistent with Walter et al.13 finding that while 88% of MICUs in academic health centers had written MICU admission criteria, only 25% used these criteria on a regular basis. Hospital mortality is likely lower for patients meeting MICU admission criteria when they are appropriately and expeditiously triaged.1418 Accordingly, developing mechanisms by which patients are routinely screened for meeting MICU admission criteria could and should reduce this source of error and improve patient outcomes.
Nine of the remaining 14 errors in care resulted from what the chart reviewers concluded was overly aggressive treatment; either excess fluid resuscitation or excess treatment of pain or anxiety. It is not clear that these represent correctable errors in care, however, as hypotensive patients require fluid resuscitation, and patients with pain or anxiety should receive analgesics or anxiolytics and it is not reasonable to expect that these interventions will be appropriately titrated in every instance. Nonetheless, our reviewers all agreed that, in eight of these patients, different interventions could have prevented the unplanned transfer.
Since 41 (27%) of the unplanned transfers were for respiratory failure, we reviewed each of these patients' records seeking evidence suggesting that the problem might have resulted from excessive use of fluids, narcotics, or anxiolytics. By retrospective analysis only six such cases could be identified. Most were due to worsening of the problem for which the patient was admitted.
Consistent with our hypothesis the majority of patients requiring unplanned MICU transfers (106/152, 70%) developed rapid response clinical triggers within the 12 hours preceding transfer, as has been previously demonstrated by Hillman et al.7 and others.8‐10, 19 Our reviewers tried to determine whether earlier or different interventions might have prevented the deterioration and the resulting unplanned transfer. Interestingly, in the large majority (94/106, 89%) they concluded that nothing different could have been done and that the transfer could not have been avoided. While this observation contrasts with our hypothesis, it is consistent with two studies questioning the utility of RRTs in preventing unplanned ICU transfers.9, 10 In addition some patients may ultimately need an ICU transfer despite receiving appropriate interventions as it is impossible to prevent an ICU transfer in every patient. Conversely, just because a patient meets a rapid response criteria does not mean that the patient needs a higher level of care or an ICU transfer as some can be safely managed on the floor.
Our study has a number of potential limitations. The data came from a single teaching hospital and we only assessed patients admitted to General Internal Medicine units and transferred to a MICU. Accordingly, our results might not generalize to other hospitals (teaching or nonteaching), to other services or to other types of ICUs. We found, however, that (1) unplanned transfers accounted for 10% of the total admissions to our MICU, a similar fraction as reported by Angus et al.1 in 2006; (2) respiratory failure/emnsufficiency and sepsis were the most common diagnoses leading to unplanned transfers as previously reported by Groeger et al.2 and Hillman et al.5; (3) mortality was increased in patients requiring unplanned transfer, as noted by Escarce and Kelley3 and Hillman et al.5; and (4) patients who were transferred to the MICU within 24 hours of admission had better outcomes than those who were transferred later, as reported by Goldhill et al.4 Accordingly, our patient population seems quite similar to others in the literature.
Since we did not use objective criteria to assign patients to each of the categories itemized in Table 5 we could have misclassified patients with respect to the cause for their unplanned MICU transfer. Despite this shortcoming, however, the scores among our independent reviewers were moderate to almost perfect suggesting misclassification did not occur commonly.
Our retrospective study design may have underestimated the utility of RRTs as we had no way of knowing the outcomes of patients who met rapid response criteria and had interventions that prevented unplanned MICU transfers.
In summary, approximately 15% of unplanned MICU transfers seem to be preventable and approximately one‐fifth seem to result from errors in care, the majority of which are errors in triage from the ED. While the large majority of unplanned transfers were preceded by clinical deterioration within the preceding 12 hours, manifested by the presence of rapid response triggers, the large majority of these do not seem to be preventable. From these findings we suggest that unplanned transfers could be reduced by more closely screening patients for the presence of defined MICU admission criteria at the time of admission from the ED, by recognizing that fluid resuscitation and control of pain and/or anxiety can have adverse effects and by monitoring patients receiving these interventions more closely.
- ,,,,,.Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). Critical care delivery in the United States: distribution of services and compliance with Leapfrog recommendations.Crit Care Med.2006;34(4):1016–1024.
- ,,, et al.Descriptive analysis of critical care units in the United States: patient characteristics and intensive care unit utilization.Crit Care Med.1993;21(2):279–291.
- ,.Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score.JAMA.1990;264(18):2389–2394.
- ,,,.The longer patients are in hospital before Intensive Care admission the higher their mortality.Intensive Care Med.2004;30(10):1908–1913.
- ,,,,, et al.Duration of life‐threatening antecedents prior to intensive care admission.Intensive Care Med.2002;28(11):1629–1634.
- Joint Commission on Accreditation of Healthcare Organizations. The Joint Commission Hospital Accreditation Program, National Patient Safety Goals, Goal 16; 2008. Available at: http://www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/08_hap_npsgs.htm. Accessed May2010.
- ,,, et al.MERIT study investigators. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial.Lancet.2005;365(9477):2091–2097.
- ,,,,,.Rapid response systems: a systematic review.Crit Care Med.2007;35(5):1238–1243.
- ,,,,,.Hospital‐wide code rates and mortality before and after implementation of a rapid response team.JAMA.2008;300(21):2506–2513.
- ,,,,.Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis.J Hosp Med.2007;2(6):422–432.
- ,,,,.Rapid response teams: a systematic review and meta‐analysis.Arch Intern Med.2010;170(1):18–26.
- ,.The measurement of observer agreement for categorical data.Biometrics.1977;33(1):159–174.
- ,,.How decisions are made to admit patients to medical intensive care units (MICUs): A survey of MICU directors at academic medical centers across the United States.Crit Care Med.2008;36:414–420.
- ,,.Mortality among appropriately referred patients refused admission to intensive‐care units.Lancet.1997;350:7–12.
- ,,,,,.Prospective evaluation of patients refused admission to an intensive care unit: triage, futility and outcome.Intensive Care Med.2001;27:1459–1465.
- ,,,,,for the Values, Ethics and Rationing in Critical Care (VERICC) Task Force. Rationing critical care beds: A systematic review.Crit Care Med.2004;32:1588–1597.
- ,,, et al.Survival of critically ill patients hospitalized in and out of intensive care.Crit Care Med.2007;35:449–457.
- ,,,,,for the DELAY‐ED study group. Impact of delayed transfer of critically ill patients form the emergency department to the intensive care unit.Crit Care Med.2007;35:1477–1483.
- ,,, et al.Antecedents to hospital deaths.Intern Med J.2001;31(6):343–348.
- ,,,,,.Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). Critical care delivery in the United States: distribution of services and compliance with Leapfrog recommendations.Crit Care Med.2006;34(4):1016–1024.
- ,,, et al.Descriptive analysis of critical care units in the United States: patient characteristics and intensive care unit utilization.Crit Care Med.1993;21(2):279–291.
- ,.Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score.JAMA.1990;264(18):2389–2394.
- ,,,.The longer patients are in hospital before Intensive Care admission the higher their mortality.Intensive Care Med.2004;30(10):1908–1913.
- ,,,,, et al.Duration of life‐threatening antecedents prior to intensive care admission.Intensive Care Med.2002;28(11):1629–1634.
- Joint Commission on Accreditation of Healthcare Organizations. The Joint Commission Hospital Accreditation Program, National Patient Safety Goals, Goal 16; 2008. Available at: http://www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/08_hap_npsgs.htm. Accessed May2010.
- ,,, et al.MERIT study investigators. Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial.Lancet.2005;365(9477):2091–2097.
- ,,,,,.Rapid response systems: a systematic review.Crit Care Med.2007;35(5):1238–1243.
- ,,,,,.Hospital‐wide code rates and mortality before and after implementation of a rapid response team.JAMA.2008;300(21):2506–2513.
- ,,,,.Effects of rapid response systems on clinical outcomes: systematic review and meta‐analysis.J Hosp Med.2007;2(6):422–432.
- ,,,,.Rapid response teams: a systematic review and meta‐analysis.Arch Intern Med.2010;170(1):18–26.
- ,.The measurement of observer agreement for categorical data.Biometrics.1977;33(1):159–174.
- ,,.How decisions are made to admit patients to medical intensive care units (MICUs): A survey of MICU directors at academic medical centers across the United States.Crit Care Med.2008;36:414–420.
- ,,.Mortality among appropriately referred patients refused admission to intensive‐care units.Lancet.1997;350:7–12.
- ,,,,,.Prospective evaluation of patients refused admission to an intensive care unit: triage, futility and outcome.Intensive Care Med.2001;27:1459–1465.
- ,,,,,for the Values, Ethics and Rationing in Critical Care (VERICC) Task Force. Rationing critical care beds: A systematic review.Crit Care Med.2004;32:1588–1597.
- ,,, et al.Survival of critically ill patients hospitalized in and out of intensive care.Crit Care Med.2007;35:449–457.
- ,,,,,for the DELAY‐ED study group. Impact of delayed transfer of critically ill patients form the emergency department to the intensive care unit.Crit Care Med.2007;35:1477–1483.
- ,,, et al.Antecedents to hospital deaths.Intern Med J.2001;31(6):343–348.
Copyright © 2010 Society of Hospital Medicine
Scheduled Admissions and Occupancy
Patient flow in a hospital refers to the management and movement of patients through the facility. Optimizing patient flow is considered of great importance to improvement of quality (including safety, efficiency, timeliness, equity, effectiveness, and patient‐centeredness), as well as finance, staff satisfaction, education and overall healthcare value.18 Central to concerns about patient flow at hospitals is occupancy, which is the census (number of patients at a point in time) divided by the bed capacity. Occupancy that is too high is associated with challenges to quality and access,913 while occupancy that is too low may underutilize resources and be costly.14, 15 Occupancy is determined by the pattern of admission and discharge, thus including length of stay (LOS) as a factor. While all related, admissions, census, occupancy, and LOS convey different aspects of hospital operations and may point to different opportunities to improve patient flow.
Variability in patient flow over time has been noted as a common occurrence in adult hospitals, due to uneven patterns of scheduled (elective) admissions, as well as uncontrollable variability of emergent admissions.2, 45, 16 Typically very few patients are scheduled to enter hospitals over weekends. In addition, when the admission is expected to be 5 days or less, clinical and operational staff may schedule those admissions early in the week to avoid patients staying the weekend. This artificial variability has been shown to lead to uneven levels of occupancy, with crowding on some days of the week more than others.2, 45, 16 As hospital crowding adversely affects access to emergent and elective care, quality and safety of care, and patient and staff satisfaction, many groups are focusing attention on patient flow and strategies to avoid high occupancy.19, 17 This is true for children's hospitals, as well, particularly as these scarce resources have ever increasing demand placed on them.1820
Patient flow improvements can be made by increasing efficiency of throughput, primarily measured by decreased LOS, or by addressing artificial variability in how hospital beds are used. As children's hospitals have short LOSs and are relatively efficient (as measured by standardized LOS ratios), we sought to evaluate how much artificial variability was active at 1 large children's hospital. We did this to both evaluate flow at 1 institution and to create methodology for other hospitals to use in order to better understand and improve their flow.
Our specific aims were to describe daily and monthly variability in admission, discharge, LOS, and occupancy patterns at a large children's hospital and assess the relationship between scheduled admissions and occupancy.
Methods
This retrospective administrative data analysis was performed with admission‐discharge‐transfer (ADT) data for inpatient admissions from one urban, tertiary‐care children's hospital for the period July 1, 2007 to June 30, 2008. The dataset included the date and time of all arrivals and departures from all inpatient units (including observation‐status patients), as entered by the unit clerks into the electronic ADT system. The dataset also included categorization of the admission as emergent, urgent, or elective (hereafter referred to as scheduled.) Registration staff entered these codes at or prior to admission. Using the timestamps, LOS was calculated by subtracting admission date and time from discharge date and time. An SAS macro was applied to the timestamps to calculate a hospital census for every hour of each calendar day. Peak census figures were extracted for each day. Occupancy was calculated as census over number of beds in use (monthly average). Data for the hospital's peak daily census and occupancy were utilized to analyze patterns of occupancy by day of week and month of year. To express variability, coefficient of variation (CV) (standard deviation [SD] divided by its mean) was used, as it is helpful when samples sizes are different.21
Analysis of number of admissions per day of week and month by type was performed with descriptive statistics and t‐tests for significant differences across seasons. We calculated a measure of patient hours generated by day of admission based on the LOS generated by each admission, in which the average number of admissions for each day of the week was multiplied by the average LOS (in hours) for those admissions. In order to remove outliers and focus on patients whose occupancy would affect weekly variation, we analyzed in detail the admissions with LOS 30 days and 7 days, respectively.
Statistical analyses were performed with SAS 9.2 (SAS Institute, Cary, NC), Stata 10.0 (StataCorp, College Station, TX) and Microsoft Excel (Microsoft, Redmond, WA). The study was approved by the Human Subjects Committee of the hospital's Institutional Review Board.
Results
A total of 22,310 patients were admitted over the period July 1, 2007 to June 30, 2008, including 4957 (22%) coded as scheduled and 17,353 (78%) coded as emergent. (Only 200 patients were registered as urgent and these were recoded as emergent for this analysis). Details on admission types and discharging departments are provided in Table 1. Overall, mean LOS was 5.6 days (median 2.29 days). For patients with LOS 30 days, mean LOS was 3.88 days (median 2.22 days). For patients staying 7 days, mean LOS was 2.4 days (median 1.98 days). Among patients with LOS 7 days, mean LOS for scheduled patients was longer for those admitted on Monday than on any other weekday (2.49 vs. 2.08 days, P < 0.0001). In contrast, mean LOS for emergent patients was longer for patients admitted on Friday and Saturday than the rest of the week (2.57 vs. 2.44 days, P < 0.0001).
| All | Scheduled | Emergent | |
|---|---|---|---|
| |||
| Total Admissions, n (%)* | 22,310 | 4957 (22) | 17,353 (78) |
| Median LOS (days) | 2.29 | 1.93 | 2.50 |
| Mean LOS (days) (95% CI) | 5.60 (5.41, 5.79) | 4.20 (3.95, 4.45) | 5.78 (5.596.0) |
| % Patients with LOS 30 days (%) | 97 | 98 | 96 |
| % Patients with LOS 7 days (%) | 84 | 89 | 83 |
| Medical patients at discharge, n (%) | 16,586 (74) | 2363 (48) | 14,403 (83) |
| Surgical patients at discharge, n (%) | 4276 (19) | 2450 (49) | 1826 (10.5) |
| Critical care patients at discharge (NICU, PICU, CICU), n (%) | 1433 (6) | 140 (3) | 1293 (7.5) |
Total admissions per month (Figure 1) averaged 1937 in October through April and 1751 in May through September (P = 0.03). Variation in the number of emergent and scheduled patients over months of the year were similar (CV 10% for each), but emergent admissions did decrease in summer (mean 1299 for June‐September vs. 1520 for the rest of the year, P = 0.003). Conversely, scheduled admissions remained relatively stable all year‐long: mean 423 per month for May through September vs. mean 413 per month for October through April (P = 0.48). Even just the summer months of June‐August, when school‐age children are on vacation, were not significantly different from other months (440 vs. 404, P = 0.2).
Variation in volume of admissions was large over days of the week, driven primarily by the pattern of scheduled admissions (CV 65.3%), which dropped off completely on weekends (Table 2, Figure 2). In contrast, there was much less variation in the number of emergent admissions across days of the week (CV 12%). For both emergent and scheduled admissions, more patients came in on Mondays than any other day of the week, but even more so for scheduled patients. While emergent admissions did decline on weekends, it was driven primarily by a decrease in physician referrals (ie, direct admission) from clinics (mean 7.48 per weekday vs. 0.73 per weekend day for the entire year, P < 0.001), while emergency department (ED) admissions remained relatively stable (mean 35.8 per weekday vs. 32.7 per weekend day, P = 0.08). Emergency transports were also stable (mean 7.15 per weekday vs. 6.49 per weekend day, P = 0.10).
| All (%) | Scheduled (%) | Emergent (%) | |
|---|---|---|---|
| |||
| CV on admissions by month | 8 | 10 | 10 |
| CV on admissions over days of week (including weekends) | 24 | 65 | 12 |
| CV on admissions over days of week (excluding weekends) | 6 | 10 | 5 |
| CV on monthly occupancy over 12 months | 4 | 14 | 2 |
Although scheduled patients contributed less to the hospital's overall occupancy, they conferred most of the variability by day of week. Over the days of the week, variation for scheduled occupancy was nearly twice that for emergent occupancy (CV 19% vs. 10%). Within the higher‐volume period of October to April, the differential was even more evident (CV 19% for scheduled occupancy versus 6% for emergent).
For scheduled patients with LOS 30 days (98% of scheduled patients), Mondays and Tuesdays together accounted for 42.5% of admission volume and 44.7% of the patient‐hours generated. For scheduled patients with LOS 7 days (89% of scheduled patients), Mondays and Tuesdays together accounted for 42% of admission volume and 45.2% of the patient‐hours generated. This combined impact of volume and LOS from admissions earlier in the week (restricted to patients with LOS 7days) is displayed graphically in Figure 3, which depicts the unevenness of scheduled admissions and their time in the hospital, with many patients overlapping in the middle of the week. Together with the more steady flow of emergent patients, this variability in scheduled occupancy contributed to mid‐week crowding, with higher risk of the hospital being >90% and >95% occupied on Wednesday through Friday (Figure 4). Detailed hourly analysis (not displayed) showed this risk to be highest from Wednesday afternoon to Friday afternoon. Due to higher emergent census, certain months also had a higher risk of high occupancy at daily peak. For example, while the entire year had 50% to 60% of Wednesdays and Thursdays with occupancy >90%, during the months of November through February, 70% to 85% of those days had occupancy at that level or higher (all these patterns were seen for both stays with LOS 30 days and 7days).
Discussion
In this study, we found that a large children's hospital was frequently at high occupancy in certain months and on certain days more than others, driven largely by predictable seasonal increases in emergent admissions and variability in scheduled admissions by day of week, respectively. Patient‐hours generated by day of admission varied as a result of both volume and LOS, both of which were larger in the early part of the week and diminished as the week progressed for scheduled admissions. But, the cumulative effect of many admissions with relatively‐longer LOS on Monday through Wednesday contributed to high occupancy on Wednesday afternoon to Friday morning, underscoring the importance of admission patterns on census later in the week. Our finding that the occupancy of scheduled patientsthe result of both the admission pattern and their LOSis also highly variable suggests that managing the inflow of scheduled patients could decrease crowding on weekdays, assure a consistent supply of capacity for regular admissions and surges, and improve the value of the delivery system.18 This inflow management would ideally consider both admissions and associated LOS, since rescheduling patients with a longer LOS (eg, 34 days) would have a greater impact on occupancy than rescheduling patients with a shorter LOS (eg, 12 days).
Not surprisingly, total admissions decreased in summer months, especially in July and August, due primarily to fewer emergent admissions. In fact, scheduled admissions per month remained relatively stable over the entire year. The decrease in summer emergent admissions may present an opportunity to stepwise shift a proportion of scheduled admissions from the spring and fall into the summer months, and winter into spring and fall, to alleviate crowding in the winter (Figure 1). Assuming clinical conditions, families and staff members were amenable to this change, hospitals with similar patterns could use this method to reduce the crowding (eg, days over 90% or 95% occupancy) that occurs in the winter.
Using patient‐hours (or days) generated by day of admission, it is evident that admission of more and longer‐stay patients at the start of the week contributes to higher occupancy later in the week (Figure 4). Mid‐week crowding could potentially contribute to a number of operational issues, including delays of new admissions, decreases in physician referrals and patient satisfaction, and an increased use of nontraditional beds (eg, treatment rooms, playrooms, doubling up single rooms) that lead to excessive patient to staff ratios and burnout for clinical staff.
The reasons for these patterns of admissions may include clinician or patient preference to avoid weekend admissions, lack of availability of particular services or resources on weekends, or concerns about safety and efficiency (due to relatively lower staffing on weekends in many hospitals).2230 While clinicians may prefer to avoid additional work on weekends, there are benefits to smoothing occupancy, including less risk of excessive work mid‐week and potential revenue opportunities. In addition, when contrasted with the estimated $1 million to $2 million cost per bed of construction, the marginal cost of staffing to provide safe, high‐quality care on weekends is dramatically lower than that of adding more beds (when faced with mid‐week crowding and unused weekend capacity). In addition, empty beds also do not generate revenue to cover fixed or variable costs, meaning that hospitals are not matching revenue to cost when there is unused capacity due to artificial variability.15, 31 Hospitals looking to make greater use of weekends, however, must be sensitive to staff concerns and the organizational dynamics of 7‐day operations, including the risk for burn‐out and attrition. Such factors should not be perceived as insurmountable barriers, particularly in light of opportunities for flexible scheduling and gain‐sharing.
Patients' and parents' preferences may partially drive admitting patterns, but a reasonable proportion of them may prefer to minimize the number of work and school days missed by being admitted near or on weekends. For example, an expected 3‐day admission could start on Friday and end on Sunday or Monday, rather than the current practice which appears to be to admit on Monday and discharge before the weekend. This may not only meet preferences among some parents to avoid missing work or school, but also by consideration of educational outcomes for hospitalized children.32
In addition, higher mean LOS for emergent patients on the weekends suggests that some services are currently unavailable on weekends to treat patients who are admitted on Fridays through Sundays.2, 25, 29, 33 More even staffing and provision of diagnostic and therapeutic services on weekends (eg, advanced radiology, consult, and laboratory services) would not only remove the barrier to weekend occupancy, it would also improve efficiency, timeliness, patient‐centeredness, and potentially effectiveness and safety for emergent patients. Running hospitals at full functionality on only 5 days of the week means that 2 out of 7 days puts patients at risk for disparate care, which may be appearing in this analysis as prolonged LOS due to lack of services over the weekenda pattern reported in the literature for adult hospitals.
Operations management and queuing theory suggest that systems function well up to 85% to 90% of capacity.34 Hospitals that plan ahead and ensure a buffer for unscheduled admissions during months or days when that demand is known to rise are less likely to cross into high occupancy. On the other hand, hospitals that do not anticipate increases in unscheduled admissions are more likely to encounter excess capacity problems.35 Aligning incentives with all staff can assist in this planning and maximize control of capacity.
Adopting the use of CV in health care operations would also be of value as a way to better express and track variation in admissions, occupancy, and discharges. Since different patient populations, different units, different hospitals, and different months have different scales, SD is not easily comparable across these settings. CV allows for comparison of variation by normalizing on the mean. In this study, it clearly differentiated the variation in elective admissions (CV 65%) over days of the week from the relative stability of emergent admissions (CV 12%). As variability and its management are important to operations, quality control, and quality improvement, use of CV can play an important role in hospital management and health services research. As days with high levels of activity may put more stress on the system, tracking this variation could lead to improvements in quality and value.
This study has several limitations. Data were analyzed for 1 children's hospital, so the analysis may or may not generally apply to other hospitals. However, in a separate study, we analyzed data from the Pediatric Health Information System database, and observed similar patterns.18 In addition, the proportion of elective patients shown in this study was similar to the national data in Kids Inpatient Database (KID, about 15% of all admissions elective).36 Moreover, the methods are reproducible for other settings, which would be useful to clinical and hospital leadership. Second, the trends depicted in the data only reflected data for one year. Third, coding of the admission as emergent or elective was done by registrars at or before arrival and may not reflect actual clinical need. In addition, those admissions coded as elective included a heterogeneous population (eg, chemotherapy to research studies).
Further studies should analyze trends for other hospitals and evaluate the effect of high peak census and high levels of variation with quality, safety, efficiency, patient satisfaction, financial, and educational outcomes for those receiving care, working, or learning at hospitals. In addition, a qualitative study that develops insights into clinician and patient/parent preferences would help answer questions in regard to usage of weekends for scheduled patients.
Conclusions
Scheduled admissions drive most variability in day‐to‐day occupancy despite the fact that they are a smaller proportion of the inpatient population. Variation in scheduled admissions by day of week provides hospitals with an opportunity to address crowding without adding beds or delaying admissions. Rather, fully utilizing capacity by smoothing occupancy over all days of the week can reduce the risk of high occupancy and thereby improve accessibility and patient/parent satisfaction. While family and staff preferences need to be considered, some combination of within‐week smoothing and shifting admissions towards summer are likely to achieve dramatic improvements in patient flow without large expenditures of capital. The key, then, is to ensure that organizational dynamic factors support these changes, so that staff members do not become stressed working at a 7‐day facility. Taken together, these strategies would better match revenue to capacity, and ultimately increase the quality and value of healthcare operations.
Acknowledgements
Authors' contributions: Study concept and design: Fieldston, Ragavan. Analysis and interpretation of data: Ragavan, Fieldston, Jayaraman, Pati. Drafting of the manuscript: Ragavan, Fieldston. Critical Revision of the manuscript for important intellectual content: Fieldston, Ragavan, Pati, Metlay. Statistical analysis: Fieldston, Jayaraman, Ragavan, Allebach. Study supervision: Fieldston, Pati, Metlay.
Additional contributions: The authors the fellows and faculty of the Robert Wood Johnson Foundation Clinical Scholars Program at the University of Pennsylvania and members of its Community Advisory Board for their suggestions to this work. They also wish to thank Tracy Kish, Jennifer Massenburg, and Brian Smith for assistance with access to and interpretation of hospital census and bed capacity data.
- ,,,,,.Relationship between resident workload and self‐perceived learning on inpatient medicine wards: a longitudinal study.BMC Med Educ.2006;6(1):35.
- AHA Solutions, Patient Flow Challenges Assessment 2009. Chicago, IL.2009.
- ,.Patient flow in hospitals: understanding and controlling it better.Front Health Serv Manage.2004;20:3–15.
- . Managing Variability in Patient Flow is the Key to Improving Access to Care, Nursing Staffing, Quality of Care, and Reducing Its Cost. Paper presented at: Institute of Medicine; June 24,2004.
- ,,,,,.Managing unnecessary variability in patient demand to reduce nursing stress and improve patient safety.Jt Comm J Qual Patient Saf.2005;31(6):330–338.
- ,,.Developing models for patient flow and daily surge capacity research.Acad Emerg Med.2006;13(11):1109–1113.
- Institute for Healthcare Improvement, Flow initiatives. 2008. Available at: http://www.ihi.org/IHI/Topics/Flow. Accessed June2010.
- ,,, et al.Hospital workload and adverse events.Med Care.2007;45(5):448–455.
- ,,,,.Impact of admission‐day crowding on the length of stay of pediatric hospitalizations.Pediatrics.2008;121(4):e718–730.
- ,,,,.Emergency department crowding, Part 1: concept, causes, and moral consequences.Ann Emerg Med.2009;53(5):605–611.
- ,.Emergency department overcrowding and ambulance diversion: the impact and potential solutions of extended boarding of admitted patients in the emergency department.J Emerg Med.2006;30(3):351–356.
- ,,,.A comparison of in‐hospital mortality risk conferred by high hospital occupancy, differences in nurse staffing levels, weekend admission, and seasonal influenza.Medical Care.2010;48(3):224–232.
- .,,.The effect of hospital bed occupancy on throughput in the pediatric emergency department.Ann Emerg Med.2009;53(6):767–776.e763.
- .Interpreting the Volume‐Outcome Relationship in the Context of Health Care Quality: Workshop Summary.Washington, DC:National Academies Press;2000.
- ,.Uncertain demand, the structure of hospital costs, and the cost of empty hospital beds.J Health Econ.1995;14(3):291–317.
- ,,, et al.Variability in surgical caseload and access to intensive care services.Anesthesiology.2003;98(6):1491–1496.
- ,,,,.Effects of hospital care environment on patient mortality and nurse outcomes.J Nurs Adm.2009;39(7/8):S45–S51.
- ,,, et al.Children's hospitals do not acutely respond to high occupancy.Pediatrics.2010;125:974–981.
- ,.Easing the strain on a pediatric tertiary care center: use of a redistribution system.Arch Pediatr Adolesc Med.2007;161(9):870–876.
- ,,.Lengths of stay and costs associated with Children's Hospitals.Pediatrics.2005;115:839–844.
- ,.Matching Supply with Demand: An introduction to operations management.New York:McGraw‐Hill;2006.
- .Do hospitals provide lower quality care on weekends?Health Serv Res.2007;42:1589–1612.
- ,.Waiting for urgent procedures on the weekend among emergently hospitalized patients.Am J Med.2004;117:175–181.
- ,.Mortality among patients admitted to hospitals on weekends as compared with weekdays.N Engl J Med.2001;345:663–668.
- ,.Enhanced weekend service: an affordable means to increased hospital procedure volume.CMAJ.2005;172(4):503–504.
- .Hospital deaths and weekend admissions‐how do we leap across a chasm?Clin Nurse Spec.2002;16:91–92.
- ,.Weekend birth and higher neonatal mortality: a problem of patient acuity or quality of care?J Obstet Gynecol Neonatal Nurs.2003;32(6):724–733.
- ,,, et al.Weekend versus weekday admission and mortality from myocardial infarction.N Engl J Med.2007;356:1099–1109.
- .Weekend admission and treatment of patients with renal colic: a case of avoidable variation?Urology.2009;73(4):720–724.
- ,,.Characteristics of weekday and weekend hospital admissions.HCUP Statistical Brief.2010;87. http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb87.pdf.
- ,. Hospital Costs and the Cost of Empty Hospital Beds (NBER Working Paper No. W3872).1991.
- ,,.The effects of timing of pediatric knee ligament surgery on short‐term academic performance in school‐aged athletes.Am J Sports Med.2009;37(9):1684–1691.
- Institute for Healthcare Improvement, Smoothing Elective Surgical Admissions. Available at: http://www.ihi.org/IHI/Topics/Flow/PatientFlow/EmergingContent/SmoothingElectiveSurgicalAdmissions.htm. Accessed June2010.
- . Institute for Healthcare Improvement, Patient Flow Comments. Available at: http://www.ihi.org/IHI/Topics/Flow. Accessed June2010.
- .OPIM 631: Operations Management.Wharton School, University of Pennsylvania.Philadelphia, PA.2008.
- Agency for Healthcare Research and Quality. HCUP Databases, Healthcare Cost and Utilization Project (HCUP). 2008. Available at: www.hcup‐us.ahrq.gov/kidoverview.jsp. Accessed June2010.
Patient flow in a hospital refers to the management and movement of patients through the facility. Optimizing patient flow is considered of great importance to improvement of quality (including safety, efficiency, timeliness, equity, effectiveness, and patient‐centeredness), as well as finance, staff satisfaction, education and overall healthcare value.18 Central to concerns about patient flow at hospitals is occupancy, which is the census (number of patients at a point in time) divided by the bed capacity. Occupancy that is too high is associated with challenges to quality and access,913 while occupancy that is too low may underutilize resources and be costly.14, 15 Occupancy is determined by the pattern of admission and discharge, thus including length of stay (LOS) as a factor. While all related, admissions, census, occupancy, and LOS convey different aspects of hospital operations and may point to different opportunities to improve patient flow.
Variability in patient flow over time has been noted as a common occurrence in adult hospitals, due to uneven patterns of scheduled (elective) admissions, as well as uncontrollable variability of emergent admissions.2, 45, 16 Typically very few patients are scheduled to enter hospitals over weekends. In addition, when the admission is expected to be 5 days or less, clinical and operational staff may schedule those admissions early in the week to avoid patients staying the weekend. This artificial variability has been shown to lead to uneven levels of occupancy, with crowding on some days of the week more than others.2, 45, 16 As hospital crowding adversely affects access to emergent and elective care, quality and safety of care, and patient and staff satisfaction, many groups are focusing attention on patient flow and strategies to avoid high occupancy.19, 17 This is true for children's hospitals, as well, particularly as these scarce resources have ever increasing demand placed on them.1820
Patient flow improvements can be made by increasing efficiency of throughput, primarily measured by decreased LOS, or by addressing artificial variability in how hospital beds are used. As children's hospitals have short LOSs and are relatively efficient (as measured by standardized LOS ratios), we sought to evaluate how much artificial variability was active at 1 large children's hospital. We did this to both evaluate flow at 1 institution and to create methodology for other hospitals to use in order to better understand and improve their flow.
Our specific aims were to describe daily and monthly variability in admission, discharge, LOS, and occupancy patterns at a large children's hospital and assess the relationship between scheduled admissions and occupancy.
Methods
This retrospective administrative data analysis was performed with admission‐discharge‐transfer (ADT) data for inpatient admissions from one urban, tertiary‐care children's hospital for the period July 1, 2007 to June 30, 2008. The dataset included the date and time of all arrivals and departures from all inpatient units (including observation‐status patients), as entered by the unit clerks into the electronic ADT system. The dataset also included categorization of the admission as emergent, urgent, or elective (hereafter referred to as scheduled.) Registration staff entered these codes at or prior to admission. Using the timestamps, LOS was calculated by subtracting admission date and time from discharge date and time. An SAS macro was applied to the timestamps to calculate a hospital census for every hour of each calendar day. Peak census figures were extracted for each day. Occupancy was calculated as census over number of beds in use (monthly average). Data for the hospital's peak daily census and occupancy were utilized to analyze patterns of occupancy by day of week and month of year. To express variability, coefficient of variation (CV) (standard deviation [SD] divided by its mean) was used, as it is helpful when samples sizes are different.21
Analysis of number of admissions per day of week and month by type was performed with descriptive statistics and t‐tests for significant differences across seasons. We calculated a measure of patient hours generated by day of admission based on the LOS generated by each admission, in which the average number of admissions for each day of the week was multiplied by the average LOS (in hours) for those admissions. In order to remove outliers and focus on patients whose occupancy would affect weekly variation, we analyzed in detail the admissions with LOS 30 days and 7 days, respectively.
Statistical analyses were performed with SAS 9.2 (SAS Institute, Cary, NC), Stata 10.0 (StataCorp, College Station, TX) and Microsoft Excel (Microsoft, Redmond, WA). The study was approved by the Human Subjects Committee of the hospital's Institutional Review Board.
Results
A total of 22,310 patients were admitted over the period July 1, 2007 to June 30, 2008, including 4957 (22%) coded as scheduled and 17,353 (78%) coded as emergent. (Only 200 patients were registered as urgent and these were recoded as emergent for this analysis). Details on admission types and discharging departments are provided in Table 1. Overall, mean LOS was 5.6 days (median 2.29 days). For patients with LOS 30 days, mean LOS was 3.88 days (median 2.22 days). For patients staying 7 days, mean LOS was 2.4 days (median 1.98 days). Among patients with LOS 7 days, mean LOS for scheduled patients was longer for those admitted on Monday than on any other weekday (2.49 vs. 2.08 days, P < 0.0001). In contrast, mean LOS for emergent patients was longer for patients admitted on Friday and Saturday than the rest of the week (2.57 vs. 2.44 days, P < 0.0001).
| All | Scheduled | Emergent | |
|---|---|---|---|
| |||
| Total Admissions, n (%)* | 22,310 | 4957 (22) | 17,353 (78) |
| Median LOS (days) | 2.29 | 1.93 | 2.50 |
| Mean LOS (days) (95% CI) | 5.60 (5.41, 5.79) | 4.20 (3.95, 4.45) | 5.78 (5.596.0) |
| % Patients with LOS 30 days (%) | 97 | 98 | 96 |
| % Patients with LOS 7 days (%) | 84 | 89 | 83 |
| Medical patients at discharge, n (%) | 16,586 (74) | 2363 (48) | 14,403 (83) |
| Surgical patients at discharge, n (%) | 4276 (19) | 2450 (49) | 1826 (10.5) |
| Critical care patients at discharge (NICU, PICU, CICU), n (%) | 1433 (6) | 140 (3) | 1293 (7.5) |
Total admissions per month (Figure 1) averaged 1937 in October through April and 1751 in May through September (P = 0.03). Variation in the number of emergent and scheduled patients over months of the year were similar (CV 10% for each), but emergent admissions did decrease in summer (mean 1299 for June‐September vs. 1520 for the rest of the year, P = 0.003). Conversely, scheduled admissions remained relatively stable all year‐long: mean 423 per month for May through September vs. mean 413 per month for October through April (P = 0.48). Even just the summer months of June‐August, when school‐age children are on vacation, were not significantly different from other months (440 vs. 404, P = 0.2).

Variation in volume of admissions was large over days of the week, driven primarily by the pattern of scheduled admissions (CV 65.3%), which dropped off completely on weekends (Table 2, Figure 2). In contrast, there was much less variation in the number of emergent admissions across days of the week (CV 12%). For both emergent and scheduled admissions, more patients came in on Mondays than any other day of the week, but even more so for scheduled patients. While emergent admissions did decline on weekends, it was driven primarily by a decrease in physician referrals (ie, direct admission) from clinics (mean 7.48 per weekday vs. 0.73 per weekend day for the entire year, P < 0.001), while emergency department (ED) admissions remained relatively stable (mean 35.8 per weekday vs. 32.7 per weekend day, P = 0.08). Emergency transports were also stable (mean 7.15 per weekday vs. 6.49 per weekend day, P = 0.10).

| All (%) | Scheduled (%) | Emergent (%) | |
|---|---|---|---|
| |||
| CV on admissions by month | 8 | 10 | 10 |
| CV on admissions over days of week (including weekends) | 24 | 65 | 12 |
| CV on admissions over days of week (excluding weekends) | 6 | 10 | 5 |
| CV on monthly occupancy over 12 months | 4 | 14 | 2 |
Although scheduled patients contributed less to the hospital's overall occupancy, they conferred most of the variability by day of week. Over the days of the week, variation for scheduled occupancy was nearly twice that for emergent occupancy (CV 19% vs. 10%). Within the higher‐volume period of October to April, the differential was even more evident (CV 19% for scheduled occupancy versus 6% for emergent).
For scheduled patients with LOS 30 days (98% of scheduled patients), Mondays and Tuesdays together accounted for 42.5% of admission volume and 44.7% of the patient‐hours generated. For scheduled patients with LOS 7 days (89% of scheduled patients), Mondays and Tuesdays together accounted for 42% of admission volume and 45.2% of the patient‐hours generated. This combined impact of volume and LOS from admissions earlier in the week (restricted to patients with LOS 7days) is displayed graphically in Figure 3, which depicts the unevenness of scheduled admissions and their time in the hospital, with many patients overlapping in the middle of the week. Together with the more steady flow of emergent patients, this variability in scheduled occupancy contributed to mid‐week crowding, with higher risk of the hospital being >90% and >95% occupied on Wednesday through Friday (Figure 4). Detailed hourly analysis (not displayed) showed this risk to be highest from Wednesday afternoon to Friday afternoon. Due to higher emergent census, certain months also had a higher risk of high occupancy at daily peak. For example, while the entire year had 50% to 60% of Wednesdays and Thursdays with occupancy >90%, during the months of November through February, 70% to 85% of those days had occupancy at that level or higher (all these patterns were seen for both stays with LOS 30 days and 7days).


Discussion
In this study, we found that a large children's hospital was frequently at high occupancy in certain months and on certain days more than others, driven largely by predictable seasonal increases in emergent admissions and variability in scheduled admissions by day of week, respectively. Patient‐hours generated by day of admission varied as a result of both volume and LOS, both of which were larger in the early part of the week and diminished as the week progressed for scheduled admissions. But, the cumulative effect of many admissions with relatively‐longer LOS on Monday through Wednesday contributed to high occupancy on Wednesday afternoon to Friday morning, underscoring the importance of admission patterns on census later in the week. Our finding that the occupancy of scheduled patientsthe result of both the admission pattern and their LOSis also highly variable suggests that managing the inflow of scheduled patients could decrease crowding on weekdays, assure a consistent supply of capacity for regular admissions and surges, and improve the value of the delivery system.18 This inflow management would ideally consider both admissions and associated LOS, since rescheduling patients with a longer LOS (eg, 34 days) would have a greater impact on occupancy than rescheduling patients with a shorter LOS (eg, 12 days).
Not surprisingly, total admissions decreased in summer months, especially in July and August, due primarily to fewer emergent admissions. In fact, scheduled admissions per month remained relatively stable over the entire year. The decrease in summer emergent admissions may present an opportunity to stepwise shift a proportion of scheduled admissions from the spring and fall into the summer months, and winter into spring and fall, to alleviate crowding in the winter (Figure 1). Assuming clinical conditions, families and staff members were amenable to this change, hospitals with similar patterns could use this method to reduce the crowding (eg, days over 90% or 95% occupancy) that occurs in the winter.
Using patient‐hours (or days) generated by day of admission, it is evident that admission of more and longer‐stay patients at the start of the week contributes to higher occupancy later in the week (Figure 4). Mid‐week crowding could potentially contribute to a number of operational issues, including delays of new admissions, decreases in physician referrals and patient satisfaction, and an increased use of nontraditional beds (eg, treatment rooms, playrooms, doubling up single rooms) that lead to excessive patient to staff ratios and burnout for clinical staff.
The reasons for these patterns of admissions may include clinician or patient preference to avoid weekend admissions, lack of availability of particular services or resources on weekends, or concerns about safety and efficiency (due to relatively lower staffing on weekends in many hospitals).2230 While clinicians may prefer to avoid additional work on weekends, there are benefits to smoothing occupancy, including less risk of excessive work mid‐week and potential revenue opportunities. In addition, when contrasted with the estimated $1 million to $2 million cost per bed of construction, the marginal cost of staffing to provide safe, high‐quality care on weekends is dramatically lower than that of adding more beds (when faced with mid‐week crowding and unused weekend capacity). In addition, empty beds also do not generate revenue to cover fixed or variable costs, meaning that hospitals are not matching revenue to cost when there is unused capacity due to artificial variability.15, 31 Hospitals looking to make greater use of weekends, however, must be sensitive to staff concerns and the organizational dynamics of 7‐day operations, including the risk for burn‐out and attrition. Such factors should not be perceived as insurmountable barriers, particularly in light of opportunities for flexible scheduling and gain‐sharing.
Patients' and parents' preferences may partially drive admitting patterns, but a reasonable proportion of them may prefer to minimize the number of work and school days missed by being admitted near or on weekends. For example, an expected 3‐day admission could start on Friday and end on Sunday or Monday, rather than the current practice which appears to be to admit on Monday and discharge before the weekend. This may not only meet preferences among some parents to avoid missing work or school, but also by consideration of educational outcomes for hospitalized children.32
In addition, higher mean LOS for emergent patients on the weekends suggests that some services are currently unavailable on weekends to treat patients who are admitted on Fridays through Sundays.2, 25, 29, 33 More even staffing and provision of diagnostic and therapeutic services on weekends (eg, advanced radiology, consult, and laboratory services) would not only remove the barrier to weekend occupancy, it would also improve efficiency, timeliness, patient‐centeredness, and potentially effectiveness and safety for emergent patients. Running hospitals at full functionality on only 5 days of the week means that 2 out of 7 days puts patients at risk for disparate care, which may be appearing in this analysis as prolonged LOS due to lack of services over the weekenda pattern reported in the literature for adult hospitals.
Operations management and queuing theory suggest that systems function well up to 85% to 90% of capacity.34 Hospitals that plan ahead and ensure a buffer for unscheduled admissions during months or days when that demand is known to rise are less likely to cross into high occupancy. On the other hand, hospitals that do not anticipate increases in unscheduled admissions are more likely to encounter excess capacity problems.35 Aligning incentives with all staff can assist in this planning and maximize control of capacity.
Adopting the use of CV in health care operations would also be of value as a way to better express and track variation in admissions, occupancy, and discharges. Since different patient populations, different units, different hospitals, and different months have different scales, SD is not easily comparable across these settings. CV allows for comparison of variation by normalizing on the mean. In this study, it clearly differentiated the variation in elective admissions (CV 65%) over days of the week from the relative stability of emergent admissions (CV 12%). As variability and its management are important to operations, quality control, and quality improvement, use of CV can play an important role in hospital management and health services research. As days with high levels of activity may put more stress on the system, tracking this variation could lead to improvements in quality and value.
This study has several limitations. Data were analyzed for 1 children's hospital, so the analysis may or may not generally apply to other hospitals. However, in a separate study, we analyzed data from the Pediatric Health Information System database, and observed similar patterns.18 In addition, the proportion of elective patients shown in this study was similar to the national data in Kids Inpatient Database (KID, about 15% of all admissions elective).36 Moreover, the methods are reproducible for other settings, which would be useful to clinical and hospital leadership. Second, the trends depicted in the data only reflected data for one year. Third, coding of the admission as emergent or elective was done by registrars at or before arrival and may not reflect actual clinical need. In addition, those admissions coded as elective included a heterogeneous population (eg, chemotherapy to research studies).
Further studies should analyze trends for other hospitals and evaluate the effect of high peak census and high levels of variation with quality, safety, efficiency, patient satisfaction, financial, and educational outcomes for those receiving care, working, or learning at hospitals. In addition, a qualitative study that develops insights into clinician and patient/parent preferences would help answer questions in regard to usage of weekends for scheduled patients.
Conclusions
Scheduled admissions drive most variability in day‐to‐day occupancy despite the fact that they are a smaller proportion of the inpatient population. Variation in scheduled admissions by day of week provides hospitals with an opportunity to address crowding without adding beds or delaying admissions. Rather, fully utilizing capacity by smoothing occupancy over all days of the week can reduce the risk of high occupancy and thereby improve accessibility and patient/parent satisfaction. While family and staff preferences need to be considered, some combination of within‐week smoothing and shifting admissions towards summer are likely to achieve dramatic improvements in patient flow without large expenditures of capital. The key, then, is to ensure that organizational dynamic factors support these changes, so that staff members do not become stressed working at a 7‐day facility. Taken together, these strategies would better match revenue to capacity, and ultimately increase the quality and value of healthcare operations.
Acknowledgements
Authors' contributions: Study concept and design: Fieldston, Ragavan. Analysis and interpretation of data: Ragavan, Fieldston, Jayaraman, Pati. Drafting of the manuscript: Ragavan, Fieldston. Critical Revision of the manuscript for important intellectual content: Fieldston, Ragavan, Pati, Metlay. Statistical analysis: Fieldston, Jayaraman, Ragavan, Allebach. Study supervision: Fieldston, Pati, Metlay.
Additional contributions: The authors the fellows and faculty of the Robert Wood Johnson Foundation Clinical Scholars Program at the University of Pennsylvania and members of its Community Advisory Board for their suggestions to this work. They also wish to thank Tracy Kish, Jennifer Massenburg, and Brian Smith for assistance with access to and interpretation of hospital census and bed capacity data.
Patient flow in a hospital refers to the management and movement of patients through the facility. Optimizing patient flow is considered of great importance to improvement of quality (including safety, efficiency, timeliness, equity, effectiveness, and patient‐centeredness), as well as finance, staff satisfaction, education and overall healthcare value.18 Central to concerns about patient flow at hospitals is occupancy, which is the census (number of patients at a point in time) divided by the bed capacity. Occupancy that is too high is associated with challenges to quality and access,913 while occupancy that is too low may underutilize resources and be costly.14, 15 Occupancy is determined by the pattern of admission and discharge, thus including length of stay (LOS) as a factor. While all related, admissions, census, occupancy, and LOS convey different aspects of hospital operations and may point to different opportunities to improve patient flow.
Variability in patient flow over time has been noted as a common occurrence in adult hospitals, due to uneven patterns of scheduled (elective) admissions, as well as uncontrollable variability of emergent admissions.2, 45, 16 Typically very few patients are scheduled to enter hospitals over weekends. In addition, when the admission is expected to be 5 days or less, clinical and operational staff may schedule those admissions early in the week to avoid patients staying the weekend. This artificial variability has been shown to lead to uneven levels of occupancy, with crowding on some days of the week more than others.2, 45, 16 As hospital crowding adversely affects access to emergent and elective care, quality and safety of care, and patient and staff satisfaction, many groups are focusing attention on patient flow and strategies to avoid high occupancy.19, 17 This is true for children's hospitals, as well, particularly as these scarce resources have ever increasing demand placed on them.1820
Patient flow improvements can be made by increasing efficiency of throughput, primarily measured by decreased LOS, or by addressing artificial variability in how hospital beds are used. As children's hospitals have short LOSs and are relatively efficient (as measured by standardized LOS ratios), we sought to evaluate how much artificial variability was active at 1 large children's hospital. We did this to both evaluate flow at 1 institution and to create methodology for other hospitals to use in order to better understand and improve their flow.
Our specific aims were to describe daily and monthly variability in admission, discharge, LOS, and occupancy patterns at a large children's hospital and assess the relationship between scheduled admissions and occupancy.
Methods
This retrospective administrative data analysis was performed with admission‐discharge‐transfer (ADT) data for inpatient admissions from one urban, tertiary‐care children's hospital for the period July 1, 2007 to June 30, 2008. The dataset included the date and time of all arrivals and departures from all inpatient units (including observation‐status patients), as entered by the unit clerks into the electronic ADT system. The dataset also included categorization of the admission as emergent, urgent, or elective (hereafter referred to as scheduled.) Registration staff entered these codes at or prior to admission. Using the timestamps, LOS was calculated by subtracting admission date and time from discharge date and time. An SAS macro was applied to the timestamps to calculate a hospital census for every hour of each calendar day. Peak census figures were extracted for each day. Occupancy was calculated as census over number of beds in use (monthly average). Data for the hospital's peak daily census and occupancy were utilized to analyze patterns of occupancy by day of week and month of year. To express variability, coefficient of variation (CV) (standard deviation [SD] divided by its mean) was used, as it is helpful when samples sizes are different.21
Analysis of number of admissions per day of week and month by type was performed with descriptive statistics and t‐tests for significant differences across seasons. We calculated a measure of patient hours generated by day of admission based on the LOS generated by each admission, in which the average number of admissions for each day of the week was multiplied by the average LOS (in hours) for those admissions. In order to remove outliers and focus on patients whose occupancy would affect weekly variation, we analyzed in detail the admissions with LOS 30 days and 7 days, respectively.
Statistical analyses were performed with SAS 9.2 (SAS Institute, Cary, NC), Stata 10.0 (StataCorp, College Station, TX) and Microsoft Excel (Microsoft, Redmond, WA). The study was approved by the Human Subjects Committee of the hospital's Institutional Review Board.
Results
A total of 22,310 patients were admitted over the period July 1, 2007 to June 30, 2008, including 4957 (22%) coded as scheduled and 17,353 (78%) coded as emergent. (Only 200 patients were registered as urgent and these were recoded as emergent for this analysis). Details on admission types and discharging departments are provided in Table 1. Overall, mean LOS was 5.6 days (median 2.29 days). For patients with LOS 30 days, mean LOS was 3.88 days (median 2.22 days). For patients staying 7 days, mean LOS was 2.4 days (median 1.98 days). Among patients with LOS 7 days, mean LOS for scheduled patients was longer for those admitted on Monday than on any other weekday (2.49 vs. 2.08 days, P < 0.0001). In contrast, mean LOS for emergent patients was longer for patients admitted on Friday and Saturday than the rest of the week (2.57 vs. 2.44 days, P < 0.0001).
| All | Scheduled | Emergent | |
|---|---|---|---|
| |||
| Total Admissions, n (%)* | 22,310 | 4957 (22) | 17,353 (78) |
| Median LOS (days) | 2.29 | 1.93 | 2.50 |
| Mean LOS (days) (95% CI) | 5.60 (5.41, 5.79) | 4.20 (3.95, 4.45) | 5.78 (5.596.0) |
| % Patients with LOS 30 days (%) | 97 | 98 | 96 |
| % Patients with LOS 7 days (%) | 84 | 89 | 83 |
| Medical patients at discharge, n (%) | 16,586 (74) | 2363 (48) | 14,403 (83) |
| Surgical patients at discharge, n (%) | 4276 (19) | 2450 (49) | 1826 (10.5) |
| Critical care patients at discharge (NICU, PICU, CICU), n (%) | 1433 (6) | 140 (3) | 1293 (7.5) |
Total admissions per month (Figure 1) averaged 1937 in October through April and 1751 in May through September (P = 0.03). Variation in the number of emergent and scheduled patients over months of the year were similar (CV 10% for each), but emergent admissions did decrease in summer (mean 1299 for June‐September vs. 1520 for the rest of the year, P = 0.003). Conversely, scheduled admissions remained relatively stable all year‐long: mean 423 per month for May through September vs. mean 413 per month for October through April (P = 0.48). Even just the summer months of June‐August, when school‐age children are on vacation, were not significantly different from other months (440 vs. 404, P = 0.2).

Variation in volume of admissions was large over days of the week, driven primarily by the pattern of scheduled admissions (CV 65.3%), which dropped off completely on weekends (Table 2, Figure 2). In contrast, there was much less variation in the number of emergent admissions across days of the week (CV 12%). For both emergent and scheduled admissions, more patients came in on Mondays than any other day of the week, but even more so for scheduled patients. While emergent admissions did decline on weekends, it was driven primarily by a decrease in physician referrals (ie, direct admission) from clinics (mean 7.48 per weekday vs. 0.73 per weekend day for the entire year, P < 0.001), while emergency department (ED) admissions remained relatively stable (mean 35.8 per weekday vs. 32.7 per weekend day, P = 0.08). Emergency transports were also stable (mean 7.15 per weekday vs. 6.49 per weekend day, P = 0.10).

| All (%) | Scheduled (%) | Emergent (%) | |
|---|---|---|---|
| |||
| CV on admissions by month | 8 | 10 | 10 |
| CV on admissions over days of week (including weekends) | 24 | 65 | 12 |
| CV on admissions over days of week (excluding weekends) | 6 | 10 | 5 |
| CV on monthly occupancy over 12 months | 4 | 14 | 2 |
Although scheduled patients contributed less to the hospital's overall occupancy, they conferred most of the variability by day of week. Over the days of the week, variation for scheduled occupancy was nearly twice that for emergent occupancy (CV 19% vs. 10%). Within the higher‐volume period of October to April, the differential was even more evident (CV 19% for scheduled occupancy versus 6% for emergent).
For scheduled patients with LOS 30 days (98% of scheduled patients), Mondays and Tuesdays together accounted for 42.5% of admission volume and 44.7% of the patient‐hours generated. For scheduled patients with LOS 7 days (89% of scheduled patients), Mondays and Tuesdays together accounted for 42% of admission volume and 45.2% of the patient‐hours generated. This combined impact of volume and LOS from admissions earlier in the week (restricted to patients with LOS 7days) is displayed graphically in Figure 3, which depicts the unevenness of scheduled admissions and their time in the hospital, with many patients overlapping in the middle of the week. Together with the more steady flow of emergent patients, this variability in scheduled occupancy contributed to mid‐week crowding, with higher risk of the hospital being >90% and >95% occupied on Wednesday through Friday (Figure 4). Detailed hourly analysis (not displayed) showed this risk to be highest from Wednesday afternoon to Friday afternoon. Due to higher emergent census, certain months also had a higher risk of high occupancy at daily peak. For example, while the entire year had 50% to 60% of Wednesdays and Thursdays with occupancy >90%, during the months of November through February, 70% to 85% of those days had occupancy at that level or higher (all these patterns were seen for both stays with LOS 30 days and 7days).


Discussion
In this study, we found that a large children's hospital was frequently at high occupancy in certain months and on certain days more than others, driven largely by predictable seasonal increases in emergent admissions and variability in scheduled admissions by day of week, respectively. Patient‐hours generated by day of admission varied as a result of both volume and LOS, both of which were larger in the early part of the week and diminished as the week progressed for scheduled admissions. But, the cumulative effect of many admissions with relatively‐longer LOS on Monday through Wednesday contributed to high occupancy on Wednesday afternoon to Friday morning, underscoring the importance of admission patterns on census later in the week. Our finding that the occupancy of scheduled patientsthe result of both the admission pattern and their LOSis also highly variable suggests that managing the inflow of scheduled patients could decrease crowding on weekdays, assure a consistent supply of capacity for regular admissions and surges, and improve the value of the delivery system.18 This inflow management would ideally consider both admissions and associated LOS, since rescheduling patients with a longer LOS (eg, 34 days) would have a greater impact on occupancy than rescheduling patients with a shorter LOS (eg, 12 days).
Not surprisingly, total admissions decreased in summer months, especially in July and August, due primarily to fewer emergent admissions. In fact, scheduled admissions per month remained relatively stable over the entire year. The decrease in summer emergent admissions may present an opportunity to stepwise shift a proportion of scheduled admissions from the spring and fall into the summer months, and winter into spring and fall, to alleviate crowding in the winter (Figure 1). Assuming clinical conditions, families and staff members were amenable to this change, hospitals with similar patterns could use this method to reduce the crowding (eg, days over 90% or 95% occupancy) that occurs in the winter.
Using patient‐hours (or days) generated by day of admission, it is evident that admission of more and longer‐stay patients at the start of the week contributes to higher occupancy later in the week (Figure 4). Mid‐week crowding could potentially contribute to a number of operational issues, including delays of new admissions, decreases in physician referrals and patient satisfaction, and an increased use of nontraditional beds (eg, treatment rooms, playrooms, doubling up single rooms) that lead to excessive patient to staff ratios and burnout for clinical staff.
The reasons for these patterns of admissions may include clinician or patient preference to avoid weekend admissions, lack of availability of particular services or resources on weekends, or concerns about safety and efficiency (due to relatively lower staffing on weekends in many hospitals).2230 While clinicians may prefer to avoid additional work on weekends, there are benefits to smoothing occupancy, including less risk of excessive work mid‐week and potential revenue opportunities. In addition, when contrasted with the estimated $1 million to $2 million cost per bed of construction, the marginal cost of staffing to provide safe, high‐quality care on weekends is dramatically lower than that of adding more beds (when faced with mid‐week crowding and unused weekend capacity). In addition, empty beds also do not generate revenue to cover fixed or variable costs, meaning that hospitals are not matching revenue to cost when there is unused capacity due to artificial variability.15, 31 Hospitals looking to make greater use of weekends, however, must be sensitive to staff concerns and the organizational dynamics of 7‐day operations, including the risk for burn‐out and attrition. Such factors should not be perceived as insurmountable barriers, particularly in light of opportunities for flexible scheduling and gain‐sharing.
Patients' and parents' preferences may partially drive admitting patterns, but a reasonable proportion of them may prefer to minimize the number of work and school days missed by being admitted near or on weekends. For example, an expected 3‐day admission could start on Friday and end on Sunday or Monday, rather than the current practice which appears to be to admit on Monday and discharge before the weekend. This may not only meet preferences among some parents to avoid missing work or school, but also by consideration of educational outcomes for hospitalized children.32
In addition, higher mean LOS for emergent patients on the weekends suggests that some services are currently unavailable on weekends to treat patients who are admitted on Fridays through Sundays.2, 25, 29, 33 More even staffing and provision of diagnostic and therapeutic services on weekends (eg, advanced radiology, consult, and laboratory services) would not only remove the barrier to weekend occupancy, it would also improve efficiency, timeliness, patient‐centeredness, and potentially effectiveness and safety for emergent patients. Running hospitals at full functionality on only 5 days of the week means that 2 out of 7 days puts patients at risk for disparate care, which may be appearing in this analysis as prolonged LOS due to lack of services over the weekenda pattern reported in the literature for adult hospitals.
Operations management and queuing theory suggest that systems function well up to 85% to 90% of capacity.34 Hospitals that plan ahead and ensure a buffer for unscheduled admissions during months or days when that demand is known to rise are less likely to cross into high occupancy. On the other hand, hospitals that do not anticipate increases in unscheduled admissions are more likely to encounter excess capacity problems.35 Aligning incentives with all staff can assist in this planning and maximize control of capacity.
Adopting the use of CV in health care operations would also be of value as a way to better express and track variation in admissions, occupancy, and discharges. Since different patient populations, different units, different hospitals, and different months have different scales, SD is not easily comparable across these settings. CV allows for comparison of variation by normalizing on the mean. In this study, it clearly differentiated the variation in elective admissions (CV 65%) over days of the week from the relative stability of emergent admissions (CV 12%). As variability and its management are important to operations, quality control, and quality improvement, use of CV can play an important role in hospital management and health services research. As days with high levels of activity may put more stress on the system, tracking this variation could lead to improvements in quality and value.
This study has several limitations. Data were analyzed for 1 children's hospital, so the analysis may or may not generally apply to other hospitals. However, in a separate study, we analyzed data from the Pediatric Health Information System database, and observed similar patterns.18 In addition, the proportion of elective patients shown in this study was similar to the national data in Kids Inpatient Database (KID, about 15% of all admissions elective).36 Moreover, the methods are reproducible for other settings, which would be useful to clinical and hospital leadership. Second, the trends depicted in the data only reflected data for one year. Third, coding of the admission as emergent or elective was done by registrars at or before arrival and may not reflect actual clinical need. In addition, those admissions coded as elective included a heterogeneous population (eg, chemotherapy to research studies).
Further studies should analyze trends for other hospitals and evaluate the effect of high peak census and high levels of variation with quality, safety, efficiency, patient satisfaction, financial, and educational outcomes for those receiving care, working, or learning at hospitals. In addition, a qualitative study that develops insights into clinician and patient/parent preferences would help answer questions in regard to usage of weekends for scheduled patients.
Conclusions
Scheduled admissions drive most variability in day‐to‐day occupancy despite the fact that they are a smaller proportion of the inpatient population. Variation in scheduled admissions by day of week provides hospitals with an opportunity to address crowding without adding beds or delaying admissions. Rather, fully utilizing capacity by smoothing occupancy over all days of the week can reduce the risk of high occupancy and thereby improve accessibility and patient/parent satisfaction. While family and staff preferences need to be considered, some combination of within‐week smoothing and shifting admissions towards summer are likely to achieve dramatic improvements in patient flow without large expenditures of capital. The key, then, is to ensure that organizational dynamic factors support these changes, so that staff members do not become stressed working at a 7‐day facility. Taken together, these strategies would better match revenue to capacity, and ultimately increase the quality and value of healthcare operations.
Acknowledgements
Authors' contributions: Study concept and design: Fieldston, Ragavan. Analysis and interpretation of data: Ragavan, Fieldston, Jayaraman, Pati. Drafting of the manuscript: Ragavan, Fieldston. Critical Revision of the manuscript for important intellectual content: Fieldston, Ragavan, Pati, Metlay. Statistical analysis: Fieldston, Jayaraman, Ragavan, Allebach. Study supervision: Fieldston, Pati, Metlay.
Additional contributions: The authors the fellows and faculty of the Robert Wood Johnson Foundation Clinical Scholars Program at the University of Pennsylvania and members of its Community Advisory Board for their suggestions to this work. They also wish to thank Tracy Kish, Jennifer Massenburg, and Brian Smith for assistance with access to and interpretation of hospital census and bed capacity data.
- ,,,,,.Relationship between resident workload and self‐perceived learning on inpatient medicine wards: a longitudinal study.BMC Med Educ.2006;6(1):35.
- AHA Solutions, Patient Flow Challenges Assessment 2009. Chicago, IL.2009.
- ,.Patient flow in hospitals: understanding and controlling it better.Front Health Serv Manage.2004;20:3–15.
- . Managing Variability in Patient Flow is the Key to Improving Access to Care, Nursing Staffing, Quality of Care, and Reducing Its Cost. Paper presented at: Institute of Medicine; June 24,2004.
- ,,,,,.Managing unnecessary variability in patient demand to reduce nursing stress and improve patient safety.Jt Comm J Qual Patient Saf.2005;31(6):330–338.
- ,,.Developing models for patient flow and daily surge capacity research.Acad Emerg Med.2006;13(11):1109–1113.
- Institute for Healthcare Improvement, Flow initiatives. 2008. Available at: http://www.ihi.org/IHI/Topics/Flow. Accessed June2010.
- ,,, et al.Hospital workload and adverse events.Med Care.2007;45(5):448–455.
- ,,,,.Impact of admission‐day crowding on the length of stay of pediatric hospitalizations.Pediatrics.2008;121(4):e718–730.
- ,,,,.Emergency department crowding, Part 1: concept, causes, and moral consequences.Ann Emerg Med.2009;53(5):605–611.
- ,.Emergency department overcrowding and ambulance diversion: the impact and potential solutions of extended boarding of admitted patients in the emergency department.J Emerg Med.2006;30(3):351–356.
- ,,,.A comparison of in‐hospital mortality risk conferred by high hospital occupancy, differences in nurse staffing levels, weekend admission, and seasonal influenza.Medical Care.2010;48(3):224–232.
- .,,.The effect of hospital bed occupancy on throughput in the pediatric emergency department.Ann Emerg Med.2009;53(6):767–776.e763.
- .Interpreting the Volume‐Outcome Relationship in the Context of Health Care Quality: Workshop Summary.Washington, DC:National Academies Press;2000.
- ,.Uncertain demand, the structure of hospital costs, and the cost of empty hospital beds.J Health Econ.1995;14(3):291–317.
- ,,, et al.Variability in surgical caseload and access to intensive care services.Anesthesiology.2003;98(6):1491–1496.
- ,,,,.Effects of hospital care environment on patient mortality and nurse outcomes.J Nurs Adm.2009;39(7/8):S45–S51.
- ,,, et al.Children's hospitals do not acutely respond to high occupancy.Pediatrics.2010;125:974–981.
- ,.Easing the strain on a pediatric tertiary care center: use of a redistribution system.Arch Pediatr Adolesc Med.2007;161(9):870–876.
- ,,.Lengths of stay and costs associated with Children's Hospitals.Pediatrics.2005;115:839–844.
- ,.Matching Supply with Demand: An introduction to operations management.New York:McGraw‐Hill;2006.
- .Do hospitals provide lower quality care on weekends?Health Serv Res.2007;42:1589–1612.
- ,.Waiting for urgent procedures on the weekend among emergently hospitalized patients.Am J Med.2004;117:175–181.
- ,.Mortality among patients admitted to hospitals on weekends as compared with weekdays.N Engl J Med.2001;345:663–668.
- ,.Enhanced weekend service: an affordable means to increased hospital procedure volume.CMAJ.2005;172(4):503–504.
- .Hospital deaths and weekend admissions‐how do we leap across a chasm?Clin Nurse Spec.2002;16:91–92.
- ,.Weekend birth and higher neonatal mortality: a problem of patient acuity or quality of care?J Obstet Gynecol Neonatal Nurs.2003;32(6):724–733.
- ,,, et al.Weekend versus weekday admission and mortality from myocardial infarction.N Engl J Med.2007;356:1099–1109.
- .Weekend admission and treatment of patients with renal colic: a case of avoidable variation?Urology.2009;73(4):720–724.
- ,,.Characteristics of weekday and weekend hospital admissions.HCUP Statistical Brief.2010;87. http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb87.pdf.
- ,. Hospital Costs and the Cost of Empty Hospital Beds (NBER Working Paper No. W3872).1991.
- ,,.The effects of timing of pediatric knee ligament surgery on short‐term academic performance in school‐aged athletes.Am J Sports Med.2009;37(9):1684–1691.
- Institute for Healthcare Improvement, Smoothing Elective Surgical Admissions. Available at: http://www.ihi.org/IHI/Topics/Flow/PatientFlow/EmergingContent/SmoothingElectiveSurgicalAdmissions.htm. Accessed June2010.
- . Institute for Healthcare Improvement, Patient Flow Comments. Available at: http://www.ihi.org/IHI/Topics/Flow. Accessed June2010.
- .OPIM 631: Operations Management.Wharton School, University of Pennsylvania.Philadelphia, PA.2008.
- Agency for Healthcare Research and Quality. HCUP Databases, Healthcare Cost and Utilization Project (HCUP). 2008. Available at: www.hcup‐us.ahrq.gov/kidoverview.jsp. Accessed June2010.
- ,,,,,.Relationship between resident workload and self‐perceived learning on inpatient medicine wards: a longitudinal study.BMC Med Educ.2006;6(1):35.
- AHA Solutions, Patient Flow Challenges Assessment 2009. Chicago, IL.2009.
- ,.Patient flow in hospitals: understanding and controlling it better.Front Health Serv Manage.2004;20:3–15.
- . Managing Variability in Patient Flow is the Key to Improving Access to Care, Nursing Staffing, Quality of Care, and Reducing Its Cost. Paper presented at: Institute of Medicine; June 24,2004.
- ,,,,,.Managing unnecessary variability in patient demand to reduce nursing stress and improve patient safety.Jt Comm J Qual Patient Saf.2005;31(6):330–338.
- ,,.Developing models for patient flow and daily surge capacity research.Acad Emerg Med.2006;13(11):1109–1113.
- Institute for Healthcare Improvement, Flow initiatives. 2008. Available at: http://www.ihi.org/IHI/Topics/Flow. Accessed June2010.
- ,,, et al.Hospital workload and adverse events.Med Care.2007;45(5):448–455.
- ,,,,.Impact of admission‐day crowding on the length of stay of pediatric hospitalizations.Pediatrics.2008;121(4):e718–730.
- ,,,,.Emergency department crowding, Part 1: concept, causes, and moral consequences.Ann Emerg Med.2009;53(5):605–611.
- ,.Emergency department overcrowding and ambulance diversion: the impact and potential solutions of extended boarding of admitted patients in the emergency department.J Emerg Med.2006;30(3):351–356.
- ,,,.A comparison of in‐hospital mortality risk conferred by high hospital occupancy, differences in nurse staffing levels, weekend admission, and seasonal influenza.Medical Care.2010;48(3):224–232.
- .,,.The effect of hospital bed occupancy on throughput in the pediatric emergency department.Ann Emerg Med.2009;53(6):767–776.e763.
- .Interpreting the Volume‐Outcome Relationship in the Context of Health Care Quality: Workshop Summary.Washington, DC:National Academies Press;2000.
- ,.Uncertain demand, the structure of hospital costs, and the cost of empty hospital beds.J Health Econ.1995;14(3):291–317.
- ,,, et al.Variability in surgical caseload and access to intensive care services.Anesthesiology.2003;98(6):1491–1496.
- ,,,,.Effects of hospital care environment on patient mortality and nurse outcomes.J Nurs Adm.2009;39(7/8):S45–S51.
- ,,, et al.Children's hospitals do not acutely respond to high occupancy.Pediatrics.2010;125:974–981.
- ,.Easing the strain on a pediatric tertiary care center: use of a redistribution system.Arch Pediatr Adolesc Med.2007;161(9):870–876.
- ,,.Lengths of stay and costs associated with Children's Hospitals.Pediatrics.2005;115:839–844.
- ,.Matching Supply with Demand: An introduction to operations management.New York:McGraw‐Hill;2006.
- .Do hospitals provide lower quality care on weekends?Health Serv Res.2007;42:1589–1612.
- ,.Waiting for urgent procedures on the weekend among emergently hospitalized patients.Am J Med.2004;117:175–181.
- ,.Mortality among patients admitted to hospitals on weekends as compared with weekdays.N Engl J Med.2001;345:663–668.
- ,.Enhanced weekend service: an affordable means to increased hospital procedure volume.CMAJ.2005;172(4):503–504.
- .Hospital deaths and weekend admissions‐how do we leap across a chasm?Clin Nurse Spec.2002;16:91–92.
- ,.Weekend birth and higher neonatal mortality: a problem of patient acuity or quality of care?J Obstet Gynecol Neonatal Nurs.2003;32(6):724–733.
- ,,, et al.Weekend versus weekday admission and mortality from myocardial infarction.N Engl J Med.2007;356:1099–1109.
- .Weekend admission and treatment of patients with renal colic: a case of avoidable variation?Urology.2009;73(4):720–724.
- ,,.Characteristics of weekday and weekend hospital admissions.HCUP Statistical Brief.2010;87. http://www.hcup‐us.ahrq.gov/reports/statbriefs/sb87.pdf.
- ,. Hospital Costs and the Cost of Empty Hospital Beds (NBER Working Paper No. W3872).1991.
- ,,.The effects of timing of pediatric knee ligament surgery on short‐term academic performance in school‐aged athletes.Am J Sports Med.2009;37(9):1684–1691.
- Institute for Healthcare Improvement, Smoothing Elective Surgical Admissions. Available at: http://www.ihi.org/IHI/Topics/Flow/PatientFlow/EmergingContent/SmoothingElectiveSurgicalAdmissions.htm. Accessed June2010.
- . Institute for Healthcare Improvement, Patient Flow Comments. Available at: http://www.ihi.org/IHI/Topics/Flow. Accessed June2010.
- .OPIM 631: Operations Management.Wharton School, University of Pennsylvania.Philadelphia, PA.2008.
- Agency for Healthcare Research and Quality. HCUP Databases, Healthcare Cost and Utilization Project (HCUP). 2008. Available at: www.hcup‐us.ahrq.gov/kidoverview.jsp. Accessed June2010.
Copyright © 2010 Society of Hospital Medicine
Hospitalist Invention Aims for Infection Prevention
Stethoscopes can be magnets for infectious agents. As many as 1 in 3 stethoscopes used in EDs carry methicillin-resistant Staphylococcus aureus (Prehospital Emergency Care. 2009;13:71-74). Cleaning them with alcohol rubs can be cumbersome, however, and the alcohol doesn’t kill such infections as Clostridium difficile, which is common in hospitals and causes colitis, says Richard Ma, MD, chair of hospital medicine at Saints Memorial Medical Center in Lowell, Mass.
“Being a hospitalist, I see a lot around me that is wasteful and inefficient,” Dr. Ma says. Four years ago, he set out to create a product that would protect against C. diff, and that would meet the challenge of keeping the neck, as well as the business end, of the stethoscope clean.
The solution: a disposable, lightweight, slip-on cover about 12 inches in length, which resembles the transparent plastic bags on rollers found in supermarket produce departments. The cover has a built-in, V-shaped seal into which the stethoscope tip is wedged. Dr. Ma plans to distribute his recently patented invention, called the Stethguard, at Saints, where all staff will be trained in its use, and to other hospitals in the state. He also hopes to license it to a medical supply distributor for wider distribution. He says it only costs pennies per bag, even less when mass-produced.
Demand for the Stethguard could be aided by the current national focus on preventing hospital-acquired infections. But will hospitalists embrace his invention? Perhaps not, Dr. Ma says, given that many doctors resist washing their hands before entering patients’ rooms. But if his idea catches on, consumers will eventually learn to demand it, he adds.
Stethoscopes can be magnets for infectious agents. As many as 1 in 3 stethoscopes used in EDs carry methicillin-resistant Staphylococcus aureus (Prehospital Emergency Care. 2009;13:71-74). Cleaning them with alcohol rubs can be cumbersome, however, and the alcohol doesn’t kill such infections as Clostridium difficile, which is common in hospitals and causes colitis, says Richard Ma, MD, chair of hospital medicine at Saints Memorial Medical Center in Lowell, Mass.
“Being a hospitalist, I see a lot around me that is wasteful and inefficient,” Dr. Ma says. Four years ago, he set out to create a product that would protect against C. diff, and that would meet the challenge of keeping the neck, as well as the business end, of the stethoscope clean.
The solution: a disposable, lightweight, slip-on cover about 12 inches in length, which resembles the transparent plastic bags on rollers found in supermarket produce departments. The cover has a built-in, V-shaped seal into which the stethoscope tip is wedged. Dr. Ma plans to distribute his recently patented invention, called the Stethguard, at Saints, where all staff will be trained in its use, and to other hospitals in the state. He also hopes to license it to a medical supply distributor for wider distribution. He says it only costs pennies per bag, even less when mass-produced.
Demand for the Stethguard could be aided by the current national focus on preventing hospital-acquired infections. But will hospitalists embrace his invention? Perhaps not, Dr. Ma says, given that many doctors resist washing their hands before entering patients’ rooms. But if his idea catches on, consumers will eventually learn to demand it, he adds.
Stethoscopes can be magnets for infectious agents. As many as 1 in 3 stethoscopes used in EDs carry methicillin-resistant Staphylococcus aureus (Prehospital Emergency Care. 2009;13:71-74). Cleaning them with alcohol rubs can be cumbersome, however, and the alcohol doesn’t kill such infections as Clostridium difficile, which is common in hospitals and causes colitis, says Richard Ma, MD, chair of hospital medicine at Saints Memorial Medical Center in Lowell, Mass.
“Being a hospitalist, I see a lot around me that is wasteful and inefficient,” Dr. Ma says. Four years ago, he set out to create a product that would protect against C. diff, and that would meet the challenge of keeping the neck, as well as the business end, of the stethoscope clean.
The solution: a disposable, lightweight, slip-on cover about 12 inches in length, which resembles the transparent plastic bags on rollers found in supermarket produce departments. The cover has a built-in, V-shaped seal into which the stethoscope tip is wedged. Dr. Ma plans to distribute his recently patented invention, called the Stethguard, at Saints, where all staff will be trained in its use, and to other hospitals in the state. He also hopes to license it to a medical supply distributor for wider distribution. He says it only costs pennies per bag, even less when mass-produced.
Demand for the Stethguard could be aided by the current national focus on preventing hospital-acquired infections. But will hospitalists embrace his invention? Perhaps not, Dr. Ma says, given that many doctors resist washing their hands before entering patients’ rooms. But if his idea catches on, consumers will eventually learn to demand it, he adds.
Business Blueprint
When your group needed a director, everybody stepped back except you. Or maybe you’re thinking about transitioning into a hospital administrative leadership role like department chair, patient safety officer, vice president of medical affairs, chief medical officer, or even CEO.
“Over the next 10 years, the single largest source of new CMOs might be hospitalists,” says John Nelson, MD, MHM, medical director at Overlake Hospital in Bellevue, Wash., and principal of the consulting firm Nelson Flores Hospital Medicine Consultants.
You might already have discovered that these responsibilities require skills that weren't taught in medical school, and you could be struggling. The trick is figuring out which skills you need to strengthen, as well as selecting the right training venues.
You'll need financial and business literacy, technical savvy for process and system improvements, planning ability, and emotional intelligence to engineer cooperative relationships between multiple stakeholders. Successful career advancement ultimately requires leadership acumen: proof that you can run a business, manage upstream and downstream communication, and handle administrative and liaison duties within the hospital.
“As healthcare reform begins to financially incentivize things like safe patient handoffs and more evidence-based medicine, the business part of running a practice is going to quickly align with quality and safety outcomes. That’s what hospital medicine leaders should be focusing on,” says Lakshmi K. Halasyamani, MD, SFHM, vice president of quality and systems improvement at Saint Joseph Mercy Hospital in Ann Arbor, Mich., and an SHM board member.
There is no shortage of training options, including books, mentorships, hospital committee membership, workshops, courses, and master’s-level programs in business leadership. And as the healthcare landscape continues to evolve, there will be a growing demand for physicians, particularly hospitalists, with greater procedural and conceptual understanding of healthcare systems.
For more about hospitalists becoming the business leaders in healthcare, check out this month’s cover story, "Business Blueprint."
When your group needed a director, everybody stepped back except you. Or maybe you’re thinking about transitioning into a hospital administrative leadership role like department chair, patient safety officer, vice president of medical affairs, chief medical officer, or even CEO.
“Over the next 10 years, the single largest source of new CMOs might be hospitalists,” says John Nelson, MD, MHM, medical director at Overlake Hospital in Bellevue, Wash., and principal of the consulting firm Nelson Flores Hospital Medicine Consultants.
You might already have discovered that these responsibilities require skills that weren't taught in medical school, and you could be struggling. The trick is figuring out which skills you need to strengthen, as well as selecting the right training venues.
You'll need financial and business literacy, technical savvy for process and system improvements, planning ability, and emotional intelligence to engineer cooperative relationships between multiple stakeholders. Successful career advancement ultimately requires leadership acumen: proof that you can run a business, manage upstream and downstream communication, and handle administrative and liaison duties within the hospital.
“As healthcare reform begins to financially incentivize things like safe patient handoffs and more evidence-based medicine, the business part of running a practice is going to quickly align with quality and safety outcomes. That’s what hospital medicine leaders should be focusing on,” says Lakshmi K. Halasyamani, MD, SFHM, vice president of quality and systems improvement at Saint Joseph Mercy Hospital in Ann Arbor, Mich., and an SHM board member.
There is no shortage of training options, including books, mentorships, hospital committee membership, workshops, courses, and master’s-level programs in business leadership. And as the healthcare landscape continues to evolve, there will be a growing demand for physicians, particularly hospitalists, with greater procedural and conceptual understanding of healthcare systems.
For more about hospitalists becoming the business leaders in healthcare, check out this month’s cover story, "Business Blueprint."
When your group needed a director, everybody stepped back except you. Or maybe you’re thinking about transitioning into a hospital administrative leadership role like department chair, patient safety officer, vice president of medical affairs, chief medical officer, or even CEO.
“Over the next 10 years, the single largest source of new CMOs might be hospitalists,” says John Nelson, MD, MHM, medical director at Overlake Hospital in Bellevue, Wash., and principal of the consulting firm Nelson Flores Hospital Medicine Consultants.
You might already have discovered that these responsibilities require skills that weren't taught in medical school, and you could be struggling. The trick is figuring out which skills you need to strengthen, as well as selecting the right training venues.
You'll need financial and business literacy, technical savvy for process and system improvements, planning ability, and emotional intelligence to engineer cooperative relationships between multiple stakeholders. Successful career advancement ultimately requires leadership acumen: proof that you can run a business, manage upstream and downstream communication, and handle administrative and liaison duties within the hospital.
“As healthcare reform begins to financially incentivize things like safe patient handoffs and more evidence-based medicine, the business part of running a practice is going to quickly align with quality and safety outcomes. That’s what hospital medicine leaders should be focusing on,” says Lakshmi K. Halasyamani, MD, SFHM, vice president of quality and systems improvement at Saint Joseph Mercy Hospital in Ann Arbor, Mich., and an SHM board member.
There is no shortage of training options, including books, mentorships, hospital committee membership, workshops, courses, and master’s-level programs in business leadership. And as the healthcare landscape continues to evolve, there will be a growing demand for physicians, particularly hospitalists, with greater procedural and conceptual understanding of healthcare systems.
For more about hospitalists becoming the business leaders in healthcare, check out this month’s cover story, "Business Blueprint."
Brentuximab and Refractory Hodgkin's Lymphoma
Brentuximab vedotin, an investigational agent, was associated with complete remission in 34% of patients whose Hodgkin's lymphoma recurred after autologous stem cell transplants, based on data from a phase II trial of 102 patients. For the full story, see http://tinyurl.com/2937bbx
Brentuximab vedotin, an investigational agent, was associated with complete remission in 34% of patients whose Hodgkin's lymphoma recurred after autologous stem cell transplants, based on data from a phase II trial of 102 patients. For the full story, see http://tinyurl.com/2937bbx
Brentuximab vedotin, an investigational agent, was associated with complete remission in 34% of patients whose Hodgkin's lymphoma recurred after autologous stem cell transplants, based on data from a phase II trial of 102 patients. For the full story, see http://tinyurl.com/2937bbx
Nilotinib for Myeloid Leukemia
Chronic myeloid leukemia patients who took 400 mg of nilotinib twice daily had a 97% overall survival rate after 3 years, based on data from 73 patients, explains Dr. Gianantonio Rosti. See the Internal Medicine News story at http://tinyurl.com/27nwkcb
Chronic myeloid leukemia patients who took 400 mg of nilotinib twice daily had a 97% overall survival rate after 3 years, based on data from 73 patients, explains Dr. Gianantonio Rosti. See the Internal Medicine News story at http://tinyurl.com/27nwkcb
Chronic myeloid leukemia patients who took 400 mg of nilotinib twice daily had a 97% overall survival rate after 3 years, based on data from 73 patients, explains Dr. Gianantonio Rosti. See the Internal Medicine News story at http://tinyurl.com/27nwkcb