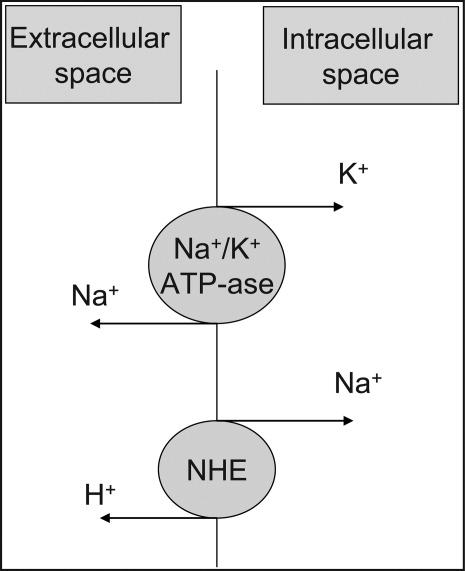
User login
Gainsharing: A Bigger Piece of the Pie
HM leaders are in a position to advocate for the potential cost savings and care efficiencies associated with gainsharing, according to a hospitalist who coauthored a study on the topic in this month’s Journal of Hospital Medicine. Gainsharing is a pay-for-performance model that tabulates the cost savings achieved via the adoption of best practices, then pays physicians bonuses with a portion of the savings.
The study found that in a three-year period ending June 2009, Beth Israel Medical Center in New York City reported a $25.1 million reduction in hospital costs, $16 million of which was attributed to physicians participating in the gainsharing program and $9.1 million from nonparticipating doctors (P<0.01) (DOI: 10.1002/jhm.788). In the same time frame, delinquent medical records dropped an average of 43% (P<0.0001).
Latha Sivaprasad, MD, FACP, FHM, medical director of quality management and patient safety and an internal-medicine attending at Beth Israel, says the data shows the viability of pay-for-performance programs.
“Gainsharing essentially aligns the incentives of physicians and hospitals to provide cost-efficient care without compromising patient safety,” says Dr. Sivaprasad. “Who better in the hospital to understand those principles than the hospitalist?”
Dr. Sivaprasad, who has been a hospitalist for eight years and is also an assistant professor at Albert Einstein College of Medicine in New York, says the majority of eligible physicians are now participating in Beth Israel’s gainsharing program, which started in 2006. She says that the validation by the Centers for Medicare & Medicaid Services (CMS)—evidenced by the Medicare demonstration project, which started in 2008—counters arguments about ethical concerns over pay for performance, as does the level of buy-in by physicians.
As it relates to HM groups, she adds, most already have some level of pay-for-performance budgeting in place.
“Pieces of it are there, even though they don’t call it gainsharing,” Dr. Sivaprasad says. “If hospitalists are incentivized for appropriate testing or streamlining throughput, pieces of this program are in place because efficient utilization of healthcare dollars is the heart of gainsharing. … Don’t excessively use precious resources you don’t need to in order to deliver quality medical care.”
HM leaders are in a position to advocate for the potential cost savings and care efficiencies associated with gainsharing, according to a hospitalist who coauthored a study on the topic in this month’s Journal of Hospital Medicine. Gainsharing is a pay-for-performance model that tabulates the cost savings achieved via the adoption of best practices, then pays physicians bonuses with a portion of the savings.
The study found that in a three-year period ending June 2009, Beth Israel Medical Center in New York City reported a $25.1 million reduction in hospital costs, $16 million of which was attributed to physicians participating in the gainsharing program and $9.1 million from nonparticipating doctors (P<0.01) (DOI: 10.1002/jhm.788). In the same time frame, delinquent medical records dropped an average of 43% (P<0.0001).
Latha Sivaprasad, MD, FACP, FHM, medical director of quality management and patient safety and an internal-medicine attending at Beth Israel, says the data shows the viability of pay-for-performance programs.
“Gainsharing essentially aligns the incentives of physicians and hospitals to provide cost-efficient care without compromising patient safety,” says Dr. Sivaprasad. “Who better in the hospital to understand those principles than the hospitalist?”
Dr. Sivaprasad, who has been a hospitalist for eight years and is also an assistant professor at Albert Einstein College of Medicine in New York, says the majority of eligible physicians are now participating in Beth Israel’s gainsharing program, which started in 2006. She says that the validation by the Centers for Medicare & Medicaid Services (CMS)—evidenced by the Medicare demonstration project, which started in 2008—counters arguments about ethical concerns over pay for performance, as does the level of buy-in by physicians.
As it relates to HM groups, she adds, most already have some level of pay-for-performance budgeting in place.
“Pieces of it are there, even though they don’t call it gainsharing,” Dr. Sivaprasad says. “If hospitalists are incentivized for appropriate testing or streamlining throughput, pieces of this program are in place because efficient utilization of healthcare dollars is the heart of gainsharing. … Don’t excessively use precious resources you don’t need to in order to deliver quality medical care.”
HM leaders are in a position to advocate for the potential cost savings and care efficiencies associated with gainsharing, according to a hospitalist who coauthored a study on the topic in this month’s Journal of Hospital Medicine. Gainsharing is a pay-for-performance model that tabulates the cost savings achieved via the adoption of best practices, then pays physicians bonuses with a portion of the savings.
The study found that in a three-year period ending June 2009, Beth Israel Medical Center in New York City reported a $25.1 million reduction in hospital costs, $16 million of which was attributed to physicians participating in the gainsharing program and $9.1 million from nonparticipating doctors (P<0.01) (DOI: 10.1002/jhm.788). In the same time frame, delinquent medical records dropped an average of 43% (P<0.0001).
Latha Sivaprasad, MD, FACP, FHM, medical director of quality management and patient safety and an internal-medicine attending at Beth Israel, says the data shows the viability of pay-for-performance programs.
“Gainsharing essentially aligns the incentives of physicians and hospitals to provide cost-efficient care without compromising patient safety,” says Dr. Sivaprasad. “Who better in the hospital to understand those principles than the hospitalist?”
Dr. Sivaprasad, who has been a hospitalist for eight years and is also an assistant professor at Albert Einstein College of Medicine in New York, says the majority of eligible physicians are now participating in Beth Israel’s gainsharing program, which started in 2006. She says that the validation by the Centers for Medicare & Medicaid Services (CMS)—evidenced by the Medicare demonstration project, which started in 2008—counters arguments about ethical concerns over pay for performance, as does the level of buy-in by physicians.
As it relates to HM groups, she adds, most already have some level of pay-for-performance budgeting in place.
“Pieces of it are there, even though they don’t call it gainsharing,” Dr. Sivaprasad says. “If hospitalists are incentivized for appropriate testing or streamlining throughput, pieces of this program are in place because efficient utilization of healthcare dollars is the heart of gainsharing. … Don’t excessively use precious resources you don’t need to in order to deliver quality medical care.”
Sobering News on Quality Front
Last month, the Office of Inspector General (OIG) issued a report (PDF) that estimates 15,000 Medicare patient deaths each month are attributable at least in part to the care they received in hospitals.
The federal watchdog agency tallied adverse events from the National Quality Forum’s list of serious reportable events and other hospital-acquired conditions in the charts of 780 Medicare patients from 2008, then extrapolated the proportions harmed through hospital care (13.5%) or who die as a result of that care (1.5%).
“Because many adverse events we identified were preventable, our study confirms the need and opportunity for hospitals to significantly reduce the incidence of events,” the report concludes. It recommends that the Agency for Healthcare Research and Quality (AHRQ) broadens patient-safety efforts and that the Centers for Medicaid & Medicare Services (CMS) provides further incentives for hospitals to reduce their incidences through its payment and oversight functions.
Confirmation of hospital safety concerns comes from a study published in the New England Journal of Medicine (2010;2363:2124-2134) that found harm to patients in North Carolina hospitals was common and did not decrease from 2002 to 2007.
Christopher Landrigan, MD, of Harvard Medical School and coauthors concluded that 18% of hospitalized patients were harmed through their medical care and, for 2.4%, it caused or contributed to their deaths.
The results of the OIG study are not surprising and might even underestimate the extent of the problem, says Gregory Seymann, MD, a hospitalist at the University of California at San Diego and a member of the Society of Hospital Medicine’s Performance and Standards Committee. The report doesn’t address what proportion of the harmed patients was on a service managed by hospitalists, “but we are in the best position to impact quality and safety—to go to our hospital administrators and get resources earmarked for quality,” he says.
Such results also mirror findings from the Institute of Medicine’s landmark 1999 report To Err is Human, adds Andrew Dunn, MD, a hospitalist at Mount Sinai Medical Center in New York City. “They suggest that medical errors are rampant in hospitals,” he says. “Because the incidence of harm is so broad across the elderly population, quality-improvement efforts in hospitals need to be across the board.”
Every hospitalist should be involved with these efforts, Dr. Dunn says. “There’s no putting your feet up. There’s always room to improve quality,” he adds. He predicts that safety outcomes will increasingly be tied to hospital reimbursement, “which is a good thing. It’s very motivational.”
Last month, the Office of Inspector General (OIG) issued a report (PDF) that estimates 15,000 Medicare patient deaths each month are attributable at least in part to the care they received in hospitals.
The federal watchdog agency tallied adverse events from the National Quality Forum’s list of serious reportable events and other hospital-acquired conditions in the charts of 780 Medicare patients from 2008, then extrapolated the proportions harmed through hospital care (13.5%) or who die as a result of that care (1.5%).
“Because many adverse events we identified were preventable, our study confirms the need and opportunity for hospitals to significantly reduce the incidence of events,” the report concludes. It recommends that the Agency for Healthcare Research and Quality (AHRQ) broadens patient-safety efforts and that the Centers for Medicaid & Medicare Services (CMS) provides further incentives for hospitals to reduce their incidences through its payment and oversight functions.
Confirmation of hospital safety concerns comes from a study published in the New England Journal of Medicine (2010;2363:2124-2134) that found harm to patients in North Carolina hospitals was common and did not decrease from 2002 to 2007.
Christopher Landrigan, MD, of Harvard Medical School and coauthors concluded that 18% of hospitalized patients were harmed through their medical care and, for 2.4%, it caused or contributed to their deaths.
The results of the OIG study are not surprising and might even underestimate the extent of the problem, says Gregory Seymann, MD, a hospitalist at the University of California at San Diego and a member of the Society of Hospital Medicine’s Performance and Standards Committee. The report doesn’t address what proportion of the harmed patients was on a service managed by hospitalists, “but we are in the best position to impact quality and safety—to go to our hospital administrators and get resources earmarked for quality,” he says.
Such results also mirror findings from the Institute of Medicine’s landmark 1999 report To Err is Human, adds Andrew Dunn, MD, a hospitalist at Mount Sinai Medical Center in New York City. “They suggest that medical errors are rampant in hospitals,” he says. “Because the incidence of harm is so broad across the elderly population, quality-improvement efforts in hospitals need to be across the board.”
Every hospitalist should be involved with these efforts, Dr. Dunn says. “There’s no putting your feet up. There’s always room to improve quality,” he adds. He predicts that safety outcomes will increasingly be tied to hospital reimbursement, “which is a good thing. It’s very motivational.”
Last month, the Office of Inspector General (OIG) issued a report (PDF) that estimates 15,000 Medicare patient deaths each month are attributable at least in part to the care they received in hospitals.
The federal watchdog agency tallied adverse events from the National Quality Forum’s list of serious reportable events and other hospital-acquired conditions in the charts of 780 Medicare patients from 2008, then extrapolated the proportions harmed through hospital care (13.5%) or who die as a result of that care (1.5%).
“Because many adverse events we identified were preventable, our study confirms the need and opportunity for hospitals to significantly reduce the incidence of events,” the report concludes. It recommends that the Agency for Healthcare Research and Quality (AHRQ) broadens patient-safety efforts and that the Centers for Medicaid & Medicare Services (CMS) provides further incentives for hospitals to reduce their incidences through its payment and oversight functions.
Confirmation of hospital safety concerns comes from a study published in the New England Journal of Medicine (2010;2363:2124-2134) that found harm to patients in North Carolina hospitals was common and did not decrease from 2002 to 2007.
Christopher Landrigan, MD, of Harvard Medical School and coauthors concluded that 18% of hospitalized patients were harmed through their medical care and, for 2.4%, it caused or contributed to their deaths.
The results of the OIG study are not surprising and might even underestimate the extent of the problem, says Gregory Seymann, MD, a hospitalist at the University of California at San Diego and a member of the Society of Hospital Medicine’s Performance and Standards Committee. The report doesn’t address what proportion of the harmed patients was on a service managed by hospitalists, “but we are in the best position to impact quality and safety—to go to our hospital administrators and get resources earmarked for quality,” he says.
Such results also mirror findings from the Institute of Medicine’s landmark 1999 report To Err is Human, adds Andrew Dunn, MD, a hospitalist at Mount Sinai Medical Center in New York City. “They suggest that medical errors are rampant in hospitals,” he says. “Because the incidence of harm is so broad across the elderly population, quality-improvement efforts in hospitals need to be across the board.”
Every hospitalist should be involved with these efforts, Dr. Dunn says. “There’s no putting your feet up. There’s always room to improve quality,” he adds. He predicts that safety outcomes will increasingly be tied to hospital reimbursement, “which is a good thing. It’s very motivational.”
Intra‐Hospital Transfer to a Higher Level of Care
Considerable research and public attention is being paid to the quantification, risk adjustment, and reporting of inpatient mortality.15 Inpatient mortality is reported as aggregate mortality (for all hospitalized patients or those with a specific diagnosis3, 6) or intensive care unit (ICU) mortality.7, 8 While reporting aggregate hospital or aggregate ICU mortality rates is useful, it is also important to develop reporting strategies that go beyond simply using data elements found in administrative databases (eg, diagnosis and procedure codes) to quantify practice variation. Ideally, such strategies would permit delineating processes of careparticularly those potentially under the control of hospitalists, not only intensiviststo identify improvement opportunities. One such process, which can be tracked using the bed history component of a patient's electronic medical record, is the transfer of patients between different units within the same hospital.
Several studies have documented that risk of ICU death is highest among patients transferred from general medical‐surgical wards, intermediate among direct admissions from the emergency department, and lowest among surgical admissions.911 Opportunities to reduce subsequent ICU mortality have been studied among ward patients who develop sepsis and are then transferred to the ICU,12 among patients who experience cardiac arrest,13, 14 as well as among patients with any physiological deterioration (eg, through the use of rapid response teams).1517 Most of these studies have been single‐center studies and/or studies reporting only an ICU denominator. While useful in some respects, such studies are less helpful to hospitalists, who would benefit from better understanding of the types of patients transferred and the total impact that transfers to a higher level of care make on general medical‐surgical wards. In addition, entities such as the Institute for Healthcare Improvement recommend the manual review of records of patients who were transferred from the ward to the ICU18 to identify performance improvement opportunities. While laudable, such approaches do not lend themselves to automated reporting strategies.
We recently described a new risk adjustment methodology for inpatient mortality based entirely on automated data preceding hospital admission and not restricted to ICU patients. This methodology, which has been externally validated in Ottawa, Canada, after development in the Kaiser Permanente Medical Care Program (KPMCP), permits quantification of a patient's pre‐existing comorbidity burden, physiologic derangement at the time of admission, and overall inpatient mortality risk.19, 20 The primary purpose of this study was to combine this methodology with bed history analysis to quantify the in‐hospital mortality and length of stay (LOS) of patients who experienced intra‐hospital transfers in a large, multihospital system. As a secondary goal, we also wanted to assess the degree to which these transfers could be predicted based on information available prior to a patient's admission.
ABBREVIATIONS AND TERMS USED IN TEXT
COPS: COmorbidity Point Score. Point score based on a patient's health care utilization diagnoses (during the year preceding admission to the hospital. Analogous to POA (present on admission) coding. Scores can range from 0 to a theoretical maximum of 701 but scores >200 are rare. With respect to a patient's pre‐existing comorbidity burden, the unadjusted relationship of COPS and inpatient mortality is as follows: a COPS <50 is associated with a mortality risk of <1%, <100 with a mortality risk of <5%, 100 to 145 with a mortality risk of 5% to 10%, and >145 with a mortality risk of 10% or more.
ICU: Intensive Care Unit. In this study, all ICUs have a minimum registered nurse to patient ratio of 1:2.
LAPS: Laboratory Acute Physiology Score. Point score based on 14 laboratory test results obtained in the 72 hours preceding hospitalization. With respect to a patient's physiologic derangement, the unadjusted relationship of LAPS and inpatient mortality is as follows: a LAPS <7 is associated with a mortality risk of <1%, <7 to 30 with a mortality risk of <5%, 30 to 60 with a mortality risk of 5% to 9%, and >60 with a mortality risk of 10% or more.
LOS: Exact hospital Length Of Stay. LOS is calculated from admission until first discharge home (i.e., it may span more than one hospital stay if a patient experienced inter‐hospital transport).
Predicted (expected) mortality risk: the % risk of death for a given patient based on his/her age, sex, admission diagnosis, COPS, and LAPS.
OEMR: Observed to Expected Mortality Ratio. For a given patient subset, the ratio of the actual mortality experienced by the subset to the expected (predicted) mortality for the subset. Predicted mortality is based on patients' age, sex, admission diagnosis, COPS, and LAPS.
OMELOS: Observed Minus Expected LOS. For a given patient subset, the difference between the actual number of hospital days experienced by the subset and the expected (predicted) number of hospital days for the subset. Predicted LOS is based on patients' age, sex, admission diagnosis, COPS, and LAPS.
TCU: Transitional Care Unit (also called intermediate care unit or stepdown unit). In this study, TCUs have variable nurse to patient ratios ranging from 1:2.5 to 1:3 and did not provide assisted ventilation, continuous pressor infusions, or invasive monitoring.
Materials and Methods
This project was approved by the Northern California KPMCP Institutional Review Board for the Protection of Human Subjects.
The Northern California KPMCP serves a total population of approximately 3.3 million members. Under a mutual exclusivity arrangement, physicians of The Permanente Medical Group, Inc., care for Kaiser Foundation Health Plan, Inc. members at facilities owned by Kaiser Foundation Hospitals, Inc. All Northern California KPMCP hospitals and clinics employ the same information systems with a common medical record number and can track care covered by the plan but delivered elsewhere. Databases maintained by the KPMCP capture admission and discharge times, admission and discharge diagnoses and procedures (assigned by professional coders), bed histories, inter‐hospital transfers, as well as the results of all inpatient and outpatient laboratory tests. The use of these databases for research has been described in multiple reports.2124
Our setting consisted of all 19 hospitals owned and operated by the KPMCP, whose characteristics are summarized in the Supporting Information Appendix available to interested readers. These include the 17 described in our previous report19 as well as 2 new hospitals (Antioch and Manteca) which are similar in size and type of population served. Our study population consisted of all patients admitted to these 19 hospitals who met these criteria: 1) hospitalization began from November 1st, 2006 through January 31st, 2008; 2) initial hospitalization occurred at a Northern California KPMCP hospital (ie, for inter‐hospital transfers, the first hospital stay occurred within the KPMCP); 3) age 15 years; and 4) hospitalization was not for childbirth.
We defined a linked hospitalization as the time period that began with a patient's admission to the hospital and ended with the patient's discharge (home, to a nursing home, or death). Linked hospitalizations can thus involve more than 1 hospital stay and could include a patient transfer from one hospital to another prior to definitive discharge. For linked hospitalizations, mortality was attributed to the admitting KPMCP hospital (ie, if a patient was admitted to hospital A, transferred to B, and died at hospital B, mortality was attributed to hospital A). We defined total LOS as the exact time in hours from when a patient was first admitted to the hospital until death or final discharge home or to a nursing home, while total ICU or transitional care unit (TCU, referred to as stepdown unit in some hospitals) LOS was calculated for all individual ICU or TCU stays during the hospital stay.
Intra‐Hospital Transfers
We grouped all possible hospital units into four types: general medical‐surgical ward (henceforth, ward); operating room (OR)/post‐anesthesia recovery (PAR); TCU; and ICU. In 2003, the KPMCP implemented a mandatory minimum staffing ratio of one registered nurse for every four patients in all its hospital units; in addition, staffing levels for designated ICUs adhered to the previously mandated minimum of one nurse for every 2 patients. So long as they adhere to these minimum ratios, individual hospitals have considerable autonomy with respect to how they staff or designate individual hospital units. Registered nurse‐to‐patient ratios during the time of this study were as follows: ward patients, 1:3.5 to 1:4; TCU patients, 1:2.5 to 1:3; and ICU patients, 1:1 to 1:2. Staffing ratios for the OR and PAR are more variable, depending on the surgical procedures involved. Current KPMCP databases do not permit accurate quantification of physician staffing. All 19 study hospitals had designated ICUs, 6 were teaching hospitals, and 11 had designated TCUs. None of the study hospitals had closed ICUs (units where only intensivists admit patients) and none had continuous coverage of the ICU by intensivists. While we were not able to employ electronic data to determine who made the decision to transfer, we did find considerable variation with respect to how intensivists covered the ICUs and how they interfaced with hospitalists. Staffing levels for specialized coronary care units and non‐ICU monitored beds were not standardized. All study hospitals had rapid response teams as well as code blue teams during the time period covered by this report. Respiratory care practitioners were available to patients in all hospital units, but considerable variation existed with respect to other services available (eg, cardiac catheterization units, provision of noninvasive positive pressure ventilation outside the ICU, etc.).
This report focuses on intra‐hospital transfers to the ICU and TCU, with special emphasis on nonsurgical transfers (due to space limitations, we are not reporting on the outcomes of patients whose first hospital unit was the OR; additional details on these patients are provided in the Supporting Information Appendix). For the purposes of this report, we defined the following admission types: direct admits (patients admitted to the ICU or TCU whose first hospital unit on admission was the ICU or TCU); and nonsurgical transfers to a higher level of care. These latter transfers could be of 3 types: ward to ICU, ward to TCU, and TCU to ICU. We also quantified the effect of inter‐hospital transfers.
Independent Variables
In addition to patients' age and sex, we employed the following independent variables to predict transfer to a higher level of care. These variables are part of the risk adjustment model described in greater detail in our previous report19 and were available electronically for all patients in the cohort. We grouped admission diagnoses into 44 broad diagnostic categories (Primary Conditions), and admission types into 4 groups (emergency medical, emergency surgical, elective medical, and elective surgical). We quantified patients' degree of physiologic derangement using a Laboratory‐based Acute Physiology Score (LAPS) using laboratory test results prior to hospitalization. We quantified patients' comorbid illness burden using a Comorbidity Point Score (COPS) based on patients' pre‐existing diagnoses over the 12‐month period preceding hospitalization. Lastly, we assigned each patient a predicted mortality risk (%) and LOS based on the above predictors,19 permitting calculation of observed to expected mortality ratios (OEMRs) and observed minus expected LOS (OMELOS).
Statistical Methods
All analyses were performed in SAS.25 We calculated standard descriptive statistics (medians, means, standard deviations) and compared different patient groupings using t and chi‐square tests. We employed a similar approach to that reported by Render et al.7 to calculate OEMR and OMELOS.
To determine the degree to which transfers to a higher level of care from the ward or TCU would be predictable using information available at the time of admission, we performed 4 sets of logistic regression analyses using the above‐mentioned predictors in which the outcome variables were as follows: 1) transfer occurring in the first 48 hours after admission (time frame by which point approximately half of the transferred patients experienced a transfer) among ward or TCU patients and 2) transfer occurring after 48 hours among ward or TCU patients. We evaluated the discrimination and calibration of these models using the same methods described in our original report (measuring the area under the receiver operator characteristic curve, or c statistic, and visually examining observed and expected mortality rates among predicted risk bands as well as risk deciles) as well as additional statistical tests recommended by Cook.19, 26
Results
During the study period, a total of 249,129 individual hospital stays involving 170,151 patients occurred at these 19 hospitals. After concatenation of inter‐hospital transfers, we were left with 237,208 linked hospitalizations. We excluded 26,738 linked hospitalizations that began at a non‐KPMCP hospital (ie, they were transported in), leaving a total of 210,470 linked hospitalizations involving 150,495 patients. The overall linked hospitalization mortality rate was 3.30%.
Table 1 summarizes cohort characteristics based on initial hospital location. On admission, ICU patients had the highest degree of physiologic derangement as well as the highest predicted mortality. Considerable inter‐hospital variation was present in both predictors and outcomes; details on these variations are provided in the Supporting Information Appendix.
| Ward | TCU | ICU | All* | |
|---|---|---|---|---|
| ||||
| n | 121,237 | 20,556 | 16,001 | 210,470 |
| Admitted via emergency department, n (%) | 99,909 (82.4) | 18,612 (90.5) | 13,847 (86.5) | 139,036 (66.1) |
| % range across hospitals | 55.0‐94.2 | 64.7‐97.6 | 49.5‐97.4 | 53.6‐76.9 |
| Male, n (%) | 53,744 (44.3) | 10,362 (50.4) | 8,378 (52.4) | 94,451 (44.9) |
| Age in years (mean SD) | 64.5 19.2 | 69.0 15.6 | 63.7 17.8 | 63.2 18.6 |
| LAPS (mean SD) | 19.2 18.0 | 23.3 19.5 | 31.7 25.7 | 16.7 19.0 |
| COPS (mean SD) | 90.4 64.0 | 99.2 65.9 | 94.5 67.5 | 84.7 61.8 |
| % predicted mortality (mean SD) | 4.0 7.1 | 4.6 7.3 | 8.7 12.8 | 3.6 7.3 |
| Observed in‐hospital deaths (n, %) | 3,793 (3.1) | 907 (4.4) | 1,995 (12.5) | 6,952 (3.3) |
| Observed to expected mortality ratio | 0.79 (0.77‐0.82) | 0.95 (0.89‐1.02) | 1.43 (1.36‐1.49) | 0.92 (0.89‐0.94) |
| Total hospital LOS, days (mean SD) | 4.6 7.5 | 5.3 10.0 | 7.8 14.0 | 4.6 8.1 |
Table 2 summarizes data from 3 groups of patients: patients initially admitted to the ward, or TCU, who did not experience a transfer to a higher level of care and patients admitted to these 2 units who did experience such a transfer. Patients who experienced a transfer constituted 5.3% (6,484/121,237) of ward patients and 6.7% (1,384/20,556) of TCU patients. Transferred patients tended to be older, have more acute physiologic derangement (higher LAPS), a greater pre‐existing illness burden (higher COPS), and a higher predicted mortality risk. Among ward patients, those with the following admission diagnoses were most likely to experience a transfer to a higher level of care: gastrointestinal bleeding (10.8% of all transfers), pneumonia (8.7%), and other infections (8.2%). The diagnoses most likely to be associated with death following transfer were cancer (death rate among transferred patients, 48%), renal disease (death rate, 36%), and liver disease (33%). Similar distributions were observed for TCU patients.
| Patients Initially Admitted to Ward, Remained There | Patients Initially Admitted to TCU, Remained There | Patients Transferred to Higher Level of Care | All | |
|---|---|---|---|---|
| ||||
| n | 114,753 | 19,172 | 7,868 | 141,793 |
| Male, n (%) | 50,586 (44.1) | 9,626 (50.2) | 3,894 (49.5) | 64,106 (45.2) |
| Age (mean SD) | 64.3 19.4 | 69.0 15.7 | 68.1 16.1 | 65.2 18.8 |
| LAPS (mean SD) | 18.9 17.8 | 22.7 19.1 | 26.7 21.0 | 19.8 18.3 |
| COPS (mean SD) | 89.4 63.7 | 98.3 65.5 | 107.9 67.6 | 91.7 64.4 |
| % predicted mortality risk (mean SD) | 3.8 7.0 | 4.4 7.0 | 6.5 8.8 | 4.1 7.1 |
| Admission diagnosis of pneumonia, n (%) | 5,624 (4.9) | 865 (4.5) | 684 (8.7) | 7,173 (5.1) |
| Admission diagnosis of sepsis, n (%) | 1,181 (1.0) | 227 (1.2) | 168 (2.1) | 1,576 (1.1) |
| Admission diagnosis of GI bleed, n (%) | 13,615 (11.9) | 1,448 (7.6) | 851 (10.8) | 15,914 (11.2) |
| Admission diagnosis of cancer, n (%) | 2,406 (2.1) | 80 (0.4) | 186 (2.4) | 2,672 (1.9) |
Table 3 compares outcomes among ward and TCU patients who did and did not experience a transfer to a higher level of care. The table shows that transferred patients were almost 3 times as likely to die, even after controlling for severity of illness, and that their hospital LOS was 9 days higher than expected. This increased risk was seen in all hospitals and among all transfer types (ward to ICU, ward to TCU, and TCU to ICU).
| Patients Initially Admitted to Ward, Remained There | Patients Initially Admitted to TCU, Remained There | Patients Transferred to Higher Level of Care | |
|---|---|---|---|
| |||
| n | 114,753 | 19,172 | 7,868 |
| Admitted to ICU, n (%) | 0 (0.0) | 0 (0.0) | 5,245 (66.7) |
| Ventilated, n (%) | 0 (0.0) | 0 (0.0) | 1,346 (17.1) |
| Died in the hospital, n (%) | 2,619 (2.3) | 572 (3.0) | 1,509 (19.2) |
| Length of stay, in days, at time of death (mean SD) | 7.0 11.9 | 8.3 12.4 | 16.2 23.7 |
| Observed to expected mortality ratio (95% CI) | 0.60 (0.57‐0.62) | 0.68 (0.63‐0.74) | 2.93 (2.79‐3.09) |
| Total hospital length of stay, days (mean SD) | 4.0 5.7 | 4.4 6.9 | 14.3 21.3 |
| Observed minus expected length of stay (95% CI) | 0.4 (0.3‐0.4) | 0.8 (0.7‐0.9) | 9.1 (8.6‐9.5) |
| Length of stay, in hours, at time of transfer (mean SD) | 80.8 167.2 | ||
Table 3 also shows that, among decedent patients, those who never left the ward or TCU died much sooner than those who died following transfer. Among direct admits to the ICU, the median LOS at time of death was 3.9 days, with a mean of 9.4 standard deviation of 19.9 days, while the corresponding times for TCU direct admits were a median and mean LOS of 6.5 and 11.7 19.5 days.
Table 4 summarizes outcomes among different patient subgroups that did and did not experience a transfer to a higher level of care. Based on location, patients who experienced a transfer from the TCU to the ICU had the highest crude death rate, but patients transferred from the ward to the ICU had the highest OEMR. On the other hand, if one divides patients by the degree of physiologic derangement, patients with low LAPS who experienced a transfer had the highest OEMR. With respect to LOS, patients transferred from the TCU to the ICU had the highest OMELOS (13.4 extra days).
| n (%)* | Death Rate (%) | OEMR | LOS (mean SD) | OMELOS | |
|---|---|---|---|---|---|
| |||||
| Never admitted to TCU or ICU | 157,632 (74.9) | 1.6 | 0.55 (0.53‐0.57) | 3.6 4.6 | 0.04 (0.02‐0.07) |
| Direct admit to TCU | 18,464 (8.8) | 2.9 | 0.66 (0.61‐0.72) | 4.2 5.8 | 0.60 (0.52‐0.68) |
| Direct admit to ICU | 14,655 (7.0) | 11.9 | 1.38 (1.32‐1.45) | 6.4 9.4 | 2.28 (2.14‐2.43) |
| Transferred from ward to ICU | 5,145 (2.4) | 21.5 | 3.23 (3.04‐3.42) | 15.7 21.6 | 10.33 (9.70‐10.96) |
| Transferred from ward to TCU | 3,144 (1.5) | 11.9 | 1.99 (1.79‐2.20) | 13.6 23.2 | 8.02 (7.23‐8.82) |
| Transferred from TCU to ICU | 1,107 (0.5) | 25.7 | 2.94 (2.61‐3.31) | 18.0 28.2 | 13.35 (11.49‐15.21) |
| Admitted to ward, COPS 80, no transfer to ICU or TCU | 55,405 (26.3) | 3.4 | 0.59 (0.56‐0.62) | 4.5 5.9 | 0.29 (0.24‐0.34) |
| Admitted to ward, COPS 80, did experience transfer to ICU or TCU | 4,851 (2.3) | 19.3 | 2.72 (2.55‐2.90) | 14.2 20.0 | 8.14 (7.56‐8.71) |
| Admitted to ward, COPS <80, no transfer to ICU or TCU | 57,421 (27.3) | 1.1 | 0.55 (0.51‐0.59) | 3.4 4.2 | 0.23 (0.19‐0.26) |
| Admitted to ward, COPS <80, did experience transfer to ICU or TCU | 3,560 (1.7) | 9.8 | 2.93 (2.63‐3.26) | 12.0 19.0 | 7.52 (6.89‐8.15) |
| Admitted to ward, LAPS 20, no transfer to ICU or TCU | 46,492 (22.1) | 4.2 | 0.59 (0.56‐0.61) | 4.6 5.4 | 0.16 (0.12‐0.21) |
| Admitted to ward, LAPS 20, did experience transfer to ICU or TCU | 4,070 (1.9) | 21.4 | 2.37 (2.22‐2.54) | 14.8 21.0 | 8.76 (8.06‐9.47) |
| Admitted to ward, LAPS <20, no transfer to ICU or TCU | 66,334 (31.5) | 0.9 | 0.55 (0.51‐0.60) | 3.5 4.9 | 0.32 (0.28‐0.36) |
| Admitted to ward, LAPS <20, did experience transfer to ICU or TCU | 4,341 (2.1) | 9.5 | 4.31 (3.90‐4.74) | 11.8 18.1 | 7.12 (6.61‐7.64) |
Transfers to a higher level of care at a different hospital, which in the KPMCP are usually planned, experienced lower mortality than transfers within the same hospital. For ward to TCU transfers, intra‐hospital transfers had a mortality of 12.1% while inter‐hospital transfers had a mortality of 5.7%. Corresponding rates for ward to ICU transfers were 21.7% and 11.2%, and for TCU to ICU transfers the rates were 25.9% and 12.5%, respectively.
Among patients initially admitted to the ward, a model to predict the occurrence of a transfer to a higher level of care (within 48 hours after admission) that included age, sex, admission type, primary condition, LAPS, COPS, and interaction terms had poor discrimination, with an area under the receiver operator characteristic (c statistic) of only 0.64. The c statistic for a model to predict transfer after 48 hours was 0.66. The corresponding models for TCU admits had c statistics of 0.67 and 0.68. All four models had poor calibration.
Discussion
Using automated bed history data permits characterizing a patient population with disproportionate mortality and LOS: intra‐hospital transfers to special care units (ICUs or TCUs). Indeed, the largest subset of these patients (those initially admitted to the ward or TCU) constituted only 3.7% of all admissions, but accounted for 24.2% of all ICU admissions, 21.7% of all hospital deaths, and 13.2% of all hospital days. These patients also had very elevated OEMRs and OMELOS. Models based on age, sex, preadmission laboratory test results, and comorbidities did not predict the occurrence of these transfers.
We performed multivariate analyses to explore the degree to which electronically assigned preadmission severity scores could predict these transfers. These analyses found that, compared to our ability to predict inpatient or 30‐day mortality at the time of admission, which is excellent, our ability to predict the occurrence of transfer after admission is much more limited. These results highlight the limitations of severity scores that rely on automated data, which may not have adequate discrimination when it comes to determining the risk of an adverse outcome within a narrow time frame. For example, among the 121,237 patients initially admitted to the ward who did not experience an intra‐hospital transfer, the mean LAPS was 18.9, while the mean LAPS among the 6,484 ward patients who did experience a transfer was 25.5. Differences between the mean and median LAPS, COPS, and predicted mortality risk among transferred and non‐transferred patients were significant (P < 0.0001 for all comparisons). However, examination of the distribution of LAPS, COPS, and predicted mortality risk between these two groups of patients showed considerable overlap.
Our methodology resembles Silber et al.'s27, 28 concept of failure to rescue in that it focuses on events occurring after hospitalization. Silber et al. argue that a hospital's quality can be measured by quantifying the degree to which patients who experience new problems are successfully rescued. Furthermore, quantification of those situations where rescue attempts are unsuccessful is felt to be superior to simply comparing raw or adjusted mortality rates because these are primarily determined by underlying case mix. The primary difference between Silber et al.'s approach and ours is at the level of detailthey specified a specific set of complications, whereas our measure is more generic and would include patients with many of the complications specified by Silber et al.27, 28
Most of the patients transferred to a higher level of care in our cohort survived (ie, were rescued), indicating that intensive care is beneficial. However, the fact that these patients had elevated OEMRs and OMELOS indicates that the real challenge facing hospitalists involves the timing of provision of a beneficial intervention. In theory, improved timing could result from earlier detection of problems, which is the underlying rationale for employing rapid response teams. However, the fact that our electronic tools (LAPS, COPS) cannot predict patient deteriorations within a narrow time frame suggests that early detection will remain a major challenge. Manually assigned vital signs scores designed for this purpose do not have good discrimination either.29, 30 This raises the possibility that, though patient groups may differ in terms of overall illness severity and mortality risk, differences at the individual patient level may be too subtle for clinicians to detect. Future research may thus need to focus on scores that combine laboratory data, vital signs, trends in data,31, 32 and newer proteomic markers (eg, procalcitonin).33 We also found that most transfers occurred early (within <72 hours), raising the possibility that at least some of these transfers may involve issues around triage rather than sudden deterioration.
Our study has important limitations. Due to resource constraints and limited data availability, we could not characterize the patients as well as might be desirable; in particular, we could not make full determinations of the actual reasons for patients' transfer for all patients. Broadly speaking, transfer to a higher level of care could be due to inappropriate triage, appropriate (preventive) transfer (which could include transfer to a more richly staffed unit for a specific procedure), relentless progression of disease despite maximal therapy, the occurrence of management errors, patient and family uncertainty about goals of care or inadequate understanding of treatment options and prognoses, or a combination of these factors. We could not make these distinctions with currently available electronic data. This is also true of postsurgical patients, in whom it is difficult to determine which transfers to intensive care might be planned (eg, in the case of surgical procedures where ICU care is anticipated) as opposed to the occurrence of a deterioration during or following surgery. Another major limitation of this study is our inability to identify code or no code status electronically. The elapsed LOS at time of death among patients who experienced a transfer to a higher level of care (as compared to patients who died in the ward without ever experiencing intra‐hospital transfer) suggests, but does not prove, that prolonged efforts were being made to keep them alive. We were also limited in terms of having access to other process data (eg, physician staffing levels, provision and timing of palliative care). Having ICU severity of illness scores would have permitted us to compare our cohort to those of other recent studies showing elevated mortality rates among transfer patients,911 but we have not yet developed that capability.
Consideration of our study findings suggests a possible research agenda that could be implemented by hospitalist researchers. This agenda should emphasize three areas: detection, intervention, and reflection.
With respect to detection, attention needs to be paid to better tools for quantifying patient risk at the time a decision to admit to the ward is made. It is likely that such tools will need to combine the attributes of our severity score (LAPS) with those of the manually assigned scores.30, 34 In some cases, use of these tools could lead a physician to change the locus of admission from the ward to the TCU or ICU, which could improve outcomes by ensuring more timely provision of intensive care. Since problems with initial triage could be due to factors other than the failure to suspect or anticipate impending instability, future research should also include a cognitive component (eg, quantifying what proportion of subsequent patient deteriorations could be ascribed to missed diagnoses35). Additional work also needs to be done on developing mathematical models that can inform electronic monitoring of ward (not just ICU) patients.
Research on interventions that hospitalists can use to prevent the need for intensive care or to improve the rescue rate should take two routes. The first is a disease‐specific route, which builds on the fact that a relatively small set of conditions (pneumonia, sepsis, gastrointestinal bleeding) account for most transfers to a higher level of care. Condition‐specific protocols, checklists, and bundles36 tailored to a ward environment (as opposed to the ICU or to the entire hospital) might prevent deteriorations in these patients, as has been reported for sepsis.37 The second route is to improve the overall capabilities of rapid response and code blue teams. Such research would need to include a more careful assessment of what commonalities exist among patients who were and were not successfully rescued by these teams. This approach would probably yield more insights than the current literature, which focuses on whether rapid response teams are a good thing or not.
Finally, research also needs to be performed on how hospitalists reflect on adverse outcomes among ward patients. Greater emphasis needs to be placed on moving beyond trigger tool approaches that rely on manual chart review. In an era of expanding use of electronic medical record systems, more work needs to be done on how to harness these to provide hospitalists with better quantitative and risk‐adjusted information. This information should not be limited to simply reporting rates of transfers and deaths. Rather, finer distinctions must be provided with respect of the type of patients (ie, more diagnostic detail), the clinical status of patients (ie, more physiologic detail), as well as the effects of including or excluding patients in whom therapeutic options may be limited (ie, do not resuscitate and comfort care patients) on reported rates. Ideally, researchers should develop better process and outcomes measures that could be tested in collaborative networks that include multiple nonacademic general medical‐surgical wards.
Acknowledgements
The authors thank Drs. Paul Feigenbaum, Alan Whippy, Joseph V. Selby, and Philip Madvig for reviewing the manuscript and Ms. Jennifer Calhoun for formatting the manuscript.
- ,,.To Err is Human: Building a Safer Health System.Washington, D. C.:National Academy Press;2000.
- Institute for Healthcare Improvement. Protecting 5 million lives from harm. Available at: http://www.ihi.org/IHI/Programs/Campaign. Accessed June2010.
- ,.Identifying poor‐quality hospitals. Can hospital mortality rates detect quality problems for medical diagnoses?Med Care.1996;34(8):737–753.
- ,,.Surgical mortality as an indicator of hospital quality: the problem with small sample size.JAMA.2004;292(7):847–851.
- State of California Office of Statewide Health Planning and Development. AHRQ ‐ Inpatient quality indicators (IQIs) hospital inpatient mortality indicators for California. Available at: http://www.oshpd.ca.gov/HID/Products/PatDischargeData/AHRQ/iqi‐imi_overview.html. Accessed June2010.
- ,,, et al.Enhancement of claims data to improve risk adjustment of hospital mortality.JAMA.2007;297(1):71–76.
- ,,, et al.Variation in outcomes in Veterans Affairs intensive care units with a computerized severity measure.Crit Care Med.2005;33(5):930–939.
- ,,,.Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today's critically ill patients.Crit Care Med.2006;34(5):1297–1310.
- ,,,.Day of the week of intensive care admission and patient outcomes: a multisite regional evaluation.Med Care.2002;40(6):530–539.
- ,,, et al.The hospital mortality of patients admitted to the ICU on weekends.Chest.2004;126(4):1292–1298.
- ,,, et al.Mortality among patients admitted to intensive care units during weekday day shifts compared with “off” hours.Crit Care Med.2007;35(1):3–11.
- ,,, et al.Septic shock: an analysis of outcomes for patients with onset on hospital wards versus intensive care units.Crit Care Med.1998;26(6):1020–1024.
- ,,,,.Clinical antecedents to in‐hospital cardiopulmonary arrest.Chest.1990;98(6):1388–1392.
- ,.Developing strategies to prevent inhospital cardiac arrest: analyzing responses of physicians and nurses in the hours before the event.Crit Care Med.1994;22(2):244–247.
- MERIT Study Investigators.Introduction of the medical emergency team (MET) system: a cluster‐randomized controlled trial.Lancet.2005;365(9477):2091–2097.
- Institute for Healthcare Improvement.The “MERIT” Trial of Medical Emergency Teams in Australia: An Analysis of Findings and Implications.Boston, MA:2005. Available on www.ihi.org
- ,,.Rapid response teams‐‐walk, don't run.JAMA.2006;296(13):1645–1647.
- ,.IHI Global Trigger Tool for Measuring Adverse Events.2nd ed.Cambridge, Massachusetts:Institute for Healthcare Improvement;2009.
- ,,,,,.Risk adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases.Medical Care.2008;46(3):232–239.
- ,,,.The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population.J Clin Epidemiol.2010;63(7):798–803.
- .Linking automated databases for research in managed care settings.Ann Intern Med.1997;127(8 Pt 2):719–724.
- ,,, et al.Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice?JAMA.2003;290(20):2685–2692.
- ,,, et al.Richardson score predicts short‐term adverse respiratory outcomes in newborns >/=34 weeks gestation.J Pediatr.2004;145(6):754–760.
- ,,, et al.Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases.Am J Manag Care.2008;14(3):158–166.
- Statistical Analysis Software [computer program]. Version 8.Cary, NC:SAS Institute, Inc.;2000.
- .Use and misuse of the receiver operating characteristic curve in risk prediction.Circulation.2007;115(7):928–935.
- ,,,.Hospital and patient characteristics associated with death after surgery. A study of adverse occurrence and failure to rescue.Med Care.1992;30(7):615–629.
- ,,.Comparing the contributions of groups of predictors: which outcomes vary with hospital rather than patient characteristics?J Am Stat Assoc.1995;90(429):7–18.
- ,.Beyond the intensive care unit: A review of interventions aimed at anticipating and preventing in‐hospital cardiopulmonary arrest.Resuscitation.2005;67(1):13–23.
- ,,.Reproducibility of physiological track‐and‐trigger warning systems for identifying at‐risk patients on the ward.Intensive Care Med.2007;33(4):619–624.
- ,,,,.Serial evaluation of the SOFA score to predict outcome in critically ill patients.JAMA.2001;286(14):1754–1758.
- ,,.Incorporation of Physiologic Trend and Interaction Effects in Neonatal Severity of Illness Scores: An Experiment Using a Variant of the Richardson Score.Intensive Care Med.2007;33(9):1602–1608.
- ,,, et al.Diagnostic and prognostic value of procalcitonin in patients with septic shock.Crit Care Med.2004;32(5):1166–1169.
- ,,, et al.Identifying the sick: can biochemical measurements be used to aid decision making on presentation to the accident and emergency department.Br J Anaesth.2005;94(6):735–741.
- .Improving patient care. The cognitive psychology of missed diagnoses.Ann Intern Med.2005;142(2):115–120.
- ,,,,,.Using care bundles to reduce in‐hospital mortality: quantitative survey.BMJ.2010;340:c1234.
- ,,, et al.Effect of a rapid response system for patients in shock on time to treatment and mortality during 5 years.Crit Care Med.2007;35(11):2568–2575.
Considerable research and public attention is being paid to the quantification, risk adjustment, and reporting of inpatient mortality.15 Inpatient mortality is reported as aggregate mortality (for all hospitalized patients or those with a specific diagnosis3, 6) or intensive care unit (ICU) mortality.7, 8 While reporting aggregate hospital or aggregate ICU mortality rates is useful, it is also important to develop reporting strategies that go beyond simply using data elements found in administrative databases (eg, diagnosis and procedure codes) to quantify practice variation. Ideally, such strategies would permit delineating processes of careparticularly those potentially under the control of hospitalists, not only intensiviststo identify improvement opportunities. One such process, which can be tracked using the bed history component of a patient's electronic medical record, is the transfer of patients between different units within the same hospital.
Several studies have documented that risk of ICU death is highest among patients transferred from general medical‐surgical wards, intermediate among direct admissions from the emergency department, and lowest among surgical admissions.911 Opportunities to reduce subsequent ICU mortality have been studied among ward patients who develop sepsis and are then transferred to the ICU,12 among patients who experience cardiac arrest,13, 14 as well as among patients with any physiological deterioration (eg, through the use of rapid response teams).1517 Most of these studies have been single‐center studies and/or studies reporting only an ICU denominator. While useful in some respects, such studies are less helpful to hospitalists, who would benefit from better understanding of the types of patients transferred and the total impact that transfers to a higher level of care make on general medical‐surgical wards. In addition, entities such as the Institute for Healthcare Improvement recommend the manual review of records of patients who were transferred from the ward to the ICU18 to identify performance improvement opportunities. While laudable, such approaches do not lend themselves to automated reporting strategies.
We recently described a new risk adjustment methodology for inpatient mortality based entirely on automated data preceding hospital admission and not restricted to ICU patients. This methodology, which has been externally validated in Ottawa, Canada, after development in the Kaiser Permanente Medical Care Program (KPMCP), permits quantification of a patient's pre‐existing comorbidity burden, physiologic derangement at the time of admission, and overall inpatient mortality risk.19, 20 The primary purpose of this study was to combine this methodology with bed history analysis to quantify the in‐hospital mortality and length of stay (LOS) of patients who experienced intra‐hospital transfers in a large, multihospital system. As a secondary goal, we also wanted to assess the degree to which these transfers could be predicted based on information available prior to a patient's admission.
ABBREVIATIONS AND TERMS USED IN TEXT
COPS: COmorbidity Point Score. Point score based on a patient's health care utilization diagnoses (during the year preceding admission to the hospital. Analogous to POA (present on admission) coding. Scores can range from 0 to a theoretical maximum of 701 but scores >200 are rare. With respect to a patient's pre‐existing comorbidity burden, the unadjusted relationship of COPS and inpatient mortality is as follows: a COPS <50 is associated with a mortality risk of <1%, <100 with a mortality risk of <5%, 100 to 145 with a mortality risk of 5% to 10%, and >145 with a mortality risk of 10% or more.
ICU: Intensive Care Unit. In this study, all ICUs have a minimum registered nurse to patient ratio of 1:2.
LAPS: Laboratory Acute Physiology Score. Point score based on 14 laboratory test results obtained in the 72 hours preceding hospitalization. With respect to a patient's physiologic derangement, the unadjusted relationship of LAPS and inpatient mortality is as follows: a LAPS <7 is associated with a mortality risk of <1%, <7 to 30 with a mortality risk of <5%, 30 to 60 with a mortality risk of 5% to 9%, and >60 with a mortality risk of 10% or more.
LOS: Exact hospital Length Of Stay. LOS is calculated from admission until first discharge home (i.e., it may span more than one hospital stay if a patient experienced inter‐hospital transport).
Predicted (expected) mortality risk: the % risk of death for a given patient based on his/her age, sex, admission diagnosis, COPS, and LAPS.
OEMR: Observed to Expected Mortality Ratio. For a given patient subset, the ratio of the actual mortality experienced by the subset to the expected (predicted) mortality for the subset. Predicted mortality is based on patients' age, sex, admission diagnosis, COPS, and LAPS.
OMELOS: Observed Minus Expected LOS. For a given patient subset, the difference between the actual number of hospital days experienced by the subset and the expected (predicted) number of hospital days for the subset. Predicted LOS is based on patients' age, sex, admission diagnosis, COPS, and LAPS.
TCU: Transitional Care Unit (also called intermediate care unit or stepdown unit). In this study, TCUs have variable nurse to patient ratios ranging from 1:2.5 to 1:3 and did not provide assisted ventilation, continuous pressor infusions, or invasive monitoring.
Materials and Methods
This project was approved by the Northern California KPMCP Institutional Review Board for the Protection of Human Subjects.
The Northern California KPMCP serves a total population of approximately 3.3 million members. Under a mutual exclusivity arrangement, physicians of The Permanente Medical Group, Inc., care for Kaiser Foundation Health Plan, Inc. members at facilities owned by Kaiser Foundation Hospitals, Inc. All Northern California KPMCP hospitals and clinics employ the same information systems with a common medical record number and can track care covered by the plan but delivered elsewhere. Databases maintained by the KPMCP capture admission and discharge times, admission and discharge diagnoses and procedures (assigned by professional coders), bed histories, inter‐hospital transfers, as well as the results of all inpatient and outpatient laboratory tests. The use of these databases for research has been described in multiple reports.2124
Our setting consisted of all 19 hospitals owned and operated by the KPMCP, whose characteristics are summarized in the Supporting Information Appendix available to interested readers. These include the 17 described in our previous report19 as well as 2 new hospitals (Antioch and Manteca) which are similar in size and type of population served. Our study population consisted of all patients admitted to these 19 hospitals who met these criteria: 1) hospitalization began from November 1st, 2006 through January 31st, 2008; 2) initial hospitalization occurred at a Northern California KPMCP hospital (ie, for inter‐hospital transfers, the first hospital stay occurred within the KPMCP); 3) age 15 years; and 4) hospitalization was not for childbirth.
We defined a linked hospitalization as the time period that began with a patient's admission to the hospital and ended with the patient's discharge (home, to a nursing home, or death). Linked hospitalizations can thus involve more than 1 hospital stay and could include a patient transfer from one hospital to another prior to definitive discharge. For linked hospitalizations, mortality was attributed to the admitting KPMCP hospital (ie, if a patient was admitted to hospital A, transferred to B, and died at hospital B, mortality was attributed to hospital A). We defined total LOS as the exact time in hours from when a patient was first admitted to the hospital until death or final discharge home or to a nursing home, while total ICU or transitional care unit (TCU, referred to as stepdown unit in some hospitals) LOS was calculated for all individual ICU or TCU stays during the hospital stay.
Intra‐Hospital Transfers
We grouped all possible hospital units into four types: general medical‐surgical ward (henceforth, ward); operating room (OR)/post‐anesthesia recovery (PAR); TCU; and ICU. In 2003, the KPMCP implemented a mandatory minimum staffing ratio of one registered nurse for every four patients in all its hospital units; in addition, staffing levels for designated ICUs adhered to the previously mandated minimum of one nurse for every 2 patients. So long as they adhere to these minimum ratios, individual hospitals have considerable autonomy with respect to how they staff or designate individual hospital units. Registered nurse‐to‐patient ratios during the time of this study were as follows: ward patients, 1:3.5 to 1:4; TCU patients, 1:2.5 to 1:3; and ICU patients, 1:1 to 1:2. Staffing ratios for the OR and PAR are more variable, depending on the surgical procedures involved. Current KPMCP databases do not permit accurate quantification of physician staffing. All 19 study hospitals had designated ICUs, 6 were teaching hospitals, and 11 had designated TCUs. None of the study hospitals had closed ICUs (units where only intensivists admit patients) and none had continuous coverage of the ICU by intensivists. While we were not able to employ electronic data to determine who made the decision to transfer, we did find considerable variation with respect to how intensivists covered the ICUs and how they interfaced with hospitalists. Staffing levels for specialized coronary care units and non‐ICU monitored beds were not standardized. All study hospitals had rapid response teams as well as code blue teams during the time period covered by this report. Respiratory care practitioners were available to patients in all hospital units, but considerable variation existed with respect to other services available (eg, cardiac catheterization units, provision of noninvasive positive pressure ventilation outside the ICU, etc.).
This report focuses on intra‐hospital transfers to the ICU and TCU, with special emphasis on nonsurgical transfers (due to space limitations, we are not reporting on the outcomes of patients whose first hospital unit was the OR; additional details on these patients are provided in the Supporting Information Appendix). For the purposes of this report, we defined the following admission types: direct admits (patients admitted to the ICU or TCU whose first hospital unit on admission was the ICU or TCU); and nonsurgical transfers to a higher level of care. These latter transfers could be of 3 types: ward to ICU, ward to TCU, and TCU to ICU. We also quantified the effect of inter‐hospital transfers.
Independent Variables
In addition to patients' age and sex, we employed the following independent variables to predict transfer to a higher level of care. These variables are part of the risk adjustment model described in greater detail in our previous report19 and were available electronically for all patients in the cohort. We grouped admission diagnoses into 44 broad diagnostic categories (Primary Conditions), and admission types into 4 groups (emergency medical, emergency surgical, elective medical, and elective surgical). We quantified patients' degree of physiologic derangement using a Laboratory‐based Acute Physiology Score (LAPS) using laboratory test results prior to hospitalization. We quantified patients' comorbid illness burden using a Comorbidity Point Score (COPS) based on patients' pre‐existing diagnoses over the 12‐month period preceding hospitalization. Lastly, we assigned each patient a predicted mortality risk (%) and LOS based on the above predictors,19 permitting calculation of observed to expected mortality ratios (OEMRs) and observed minus expected LOS (OMELOS).
Statistical Methods
All analyses were performed in SAS.25 We calculated standard descriptive statistics (medians, means, standard deviations) and compared different patient groupings using t and chi‐square tests. We employed a similar approach to that reported by Render et al.7 to calculate OEMR and OMELOS.
To determine the degree to which transfers to a higher level of care from the ward or TCU would be predictable using information available at the time of admission, we performed 4 sets of logistic regression analyses using the above‐mentioned predictors in which the outcome variables were as follows: 1) transfer occurring in the first 48 hours after admission (time frame by which point approximately half of the transferred patients experienced a transfer) among ward or TCU patients and 2) transfer occurring after 48 hours among ward or TCU patients. We evaluated the discrimination and calibration of these models using the same methods described in our original report (measuring the area under the receiver operator characteristic curve, or c statistic, and visually examining observed and expected mortality rates among predicted risk bands as well as risk deciles) as well as additional statistical tests recommended by Cook.19, 26
Results
During the study period, a total of 249,129 individual hospital stays involving 170,151 patients occurred at these 19 hospitals. After concatenation of inter‐hospital transfers, we were left with 237,208 linked hospitalizations. We excluded 26,738 linked hospitalizations that began at a non‐KPMCP hospital (ie, they were transported in), leaving a total of 210,470 linked hospitalizations involving 150,495 patients. The overall linked hospitalization mortality rate was 3.30%.
Table 1 summarizes cohort characteristics based on initial hospital location. On admission, ICU patients had the highest degree of physiologic derangement as well as the highest predicted mortality. Considerable inter‐hospital variation was present in both predictors and outcomes; details on these variations are provided in the Supporting Information Appendix.
| Ward | TCU | ICU | All* | |
|---|---|---|---|---|
| ||||
| n | 121,237 | 20,556 | 16,001 | 210,470 |
| Admitted via emergency department, n (%) | 99,909 (82.4) | 18,612 (90.5) | 13,847 (86.5) | 139,036 (66.1) |
| % range across hospitals | 55.0‐94.2 | 64.7‐97.6 | 49.5‐97.4 | 53.6‐76.9 |
| Male, n (%) | 53,744 (44.3) | 10,362 (50.4) | 8,378 (52.4) | 94,451 (44.9) |
| Age in years (mean SD) | 64.5 19.2 | 69.0 15.6 | 63.7 17.8 | 63.2 18.6 |
| LAPS (mean SD) | 19.2 18.0 | 23.3 19.5 | 31.7 25.7 | 16.7 19.0 |
| COPS (mean SD) | 90.4 64.0 | 99.2 65.9 | 94.5 67.5 | 84.7 61.8 |
| % predicted mortality (mean SD) | 4.0 7.1 | 4.6 7.3 | 8.7 12.8 | 3.6 7.3 |
| Observed in‐hospital deaths (n, %) | 3,793 (3.1) | 907 (4.4) | 1,995 (12.5) | 6,952 (3.3) |
| Observed to expected mortality ratio | 0.79 (0.77‐0.82) | 0.95 (0.89‐1.02) | 1.43 (1.36‐1.49) | 0.92 (0.89‐0.94) |
| Total hospital LOS, days (mean SD) | 4.6 7.5 | 5.3 10.0 | 7.8 14.0 | 4.6 8.1 |
Table 2 summarizes data from 3 groups of patients: patients initially admitted to the ward, or TCU, who did not experience a transfer to a higher level of care and patients admitted to these 2 units who did experience such a transfer. Patients who experienced a transfer constituted 5.3% (6,484/121,237) of ward patients and 6.7% (1,384/20,556) of TCU patients. Transferred patients tended to be older, have more acute physiologic derangement (higher LAPS), a greater pre‐existing illness burden (higher COPS), and a higher predicted mortality risk. Among ward patients, those with the following admission diagnoses were most likely to experience a transfer to a higher level of care: gastrointestinal bleeding (10.8% of all transfers), pneumonia (8.7%), and other infections (8.2%). The diagnoses most likely to be associated with death following transfer were cancer (death rate among transferred patients, 48%), renal disease (death rate, 36%), and liver disease (33%). Similar distributions were observed for TCU patients.
| Patients Initially Admitted to Ward, Remained There | Patients Initially Admitted to TCU, Remained There | Patients Transferred to Higher Level of Care | All | |
|---|---|---|---|---|
| ||||
| n | 114,753 | 19,172 | 7,868 | 141,793 |
| Male, n (%) | 50,586 (44.1) | 9,626 (50.2) | 3,894 (49.5) | 64,106 (45.2) |
| Age (mean SD) | 64.3 19.4 | 69.0 15.7 | 68.1 16.1 | 65.2 18.8 |
| LAPS (mean SD) | 18.9 17.8 | 22.7 19.1 | 26.7 21.0 | 19.8 18.3 |
| COPS (mean SD) | 89.4 63.7 | 98.3 65.5 | 107.9 67.6 | 91.7 64.4 |
| % predicted mortality risk (mean SD) | 3.8 7.0 | 4.4 7.0 | 6.5 8.8 | 4.1 7.1 |
| Admission diagnosis of pneumonia, n (%) | 5,624 (4.9) | 865 (4.5) | 684 (8.7) | 7,173 (5.1) |
| Admission diagnosis of sepsis, n (%) | 1,181 (1.0) | 227 (1.2) | 168 (2.1) | 1,576 (1.1) |
| Admission diagnosis of GI bleed, n (%) | 13,615 (11.9) | 1,448 (7.6) | 851 (10.8) | 15,914 (11.2) |
| Admission diagnosis of cancer, n (%) | 2,406 (2.1) | 80 (0.4) | 186 (2.4) | 2,672 (1.9) |
Table 3 compares outcomes among ward and TCU patients who did and did not experience a transfer to a higher level of care. The table shows that transferred patients were almost 3 times as likely to die, even after controlling for severity of illness, and that their hospital LOS was 9 days higher than expected. This increased risk was seen in all hospitals and among all transfer types (ward to ICU, ward to TCU, and TCU to ICU).
| Patients Initially Admitted to Ward, Remained There | Patients Initially Admitted to TCU, Remained There | Patients Transferred to Higher Level of Care | |
|---|---|---|---|
| |||
| n | 114,753 | 19,172 | 7,868 |
| Admitted to ICU, n (%) | 0 (0.0) | 0 (0.0) | 5,245 (66.7) |
| Ventilated, n (%) | 0 (0.0) | 0 (0.0) | 1,346 (17.1) |
| Died in the hospital, n (%) | 2,619 (2.3) | 572 (3.0) | 1,509 (19.2) |
| Length of stay, in days, at time of death (mean SD) | 7.0 11.9 | 8.3 12.4 | 16.2 23.7 |
| Observed to expected mortality ratio (95% CI) | 0.60 (0.57‐0.62) | 0.68 (0.63‐0.74) | 2.93 (2.79‐3.09) |
| Total hospital length of stay, days (mean SD) | 4.0 5.7 | 4.4 6.9 | 14.3 21.3 |
| Observed minus expected length of stay (95% CI) | 0.4 (0.3‐0.4) | 0.8 (0.7‐0.9) | 9.1 (8.6‐9.5) |
| Length of stay, in hours, at time of transfer (mean SD) | 80.8 167.2 | ||
Table 3 also shows that, among decedent patients, those who never left the ward or TCU died much sooner than those who died following transfer. Among direct admits to the ICU, the median LOS at time of death was 3.9 days, with a mean of 9.4 standard deviation of 19.9 days, while the corresponding times for TCU direct admits were a median and mean LOS of 6.5 and 11.7 19.5 days.
Table 4 summarizes outcomes among different patient subgroups that did and did not experience a transfer to a higher level of care. Based on location, patients who experienced a transfer from the TCU to the ICU had the highest crude death rate, but patients transferred from the ward to the ICU had the highest OEMR. On the other hand, if one divides patients by the degree of physiologic derangement, patients with low LAPS who experienced a transfer had the highest OEMR. With respect to LOS, patients transferred from the TCU to the ICU had the highest OMELOS (13.4 extra days).
| n (%)* | Death Rate (%) | OEMR | LOS (mean SD) | OMELOS | |
|---|---|---|---|---|---|
| |||||
| Never admitted to TCU or ICU | 157,632 (74.9) | 1.6 | 0.55 (0.53‐0.57) | 3.6 4.6 | 0.04 (0.02‐0.07) |
| Direct admit to TCU | 18,464 (8.8) | 2.9 | 0.66 (0.61‐0.72) | 4.2 5.8 | 0.60 (0.52‐0.68) |
| Direct admit to ICU | 14,655 (7.0) | 11.9 | 1.38 (1.32‐1.45) | 6.4 9.4 | 2.28 (2.14‐2.43) |
| Transferred from ward to ICU | 5,145 (2.4) | 21.5 | 3.23 (3.04‐3.42) | 15.7 21.6 | 10.33 (9.70‐10.96) |
| Transferred from ward to TCU | 3,144 (1.5) | 11.9 | 1.99 (1.79‐2.20) | 13.6 23.2 | 8.02 (7.23‐8.82) |
| Transferred from TCU to ICU | 1,107 (0.5) | 25.7 | 2.94 (2.61‐3.31) | 18.0 28.2 | 13.35 (11.49‐15.21) |
| Admitted to ward, COPS 80, no transfer to ICU or TCU | 55,405 (26.3) | 3.4 | 0.59 (0.56‐0.62) | 4.5 5.9 | 0.29 (0.24‐0.34) |
| Admitted to ward, COPS 80, did experience transfer to ICU or TCU | 4,851 (2.3) | 19.3 | 2.72 (2.55‐2.90) | 14.2 20.0 | 8.14 (7.56‐8.71) |
| Admitted to ward, COPS <80, no transfer to ICU or TCU | 57,421 (27.3) | 1.1 | 0.55 (0.51‐0.59) | 3.4 4.2 | 0.23 (0.19‐0.26) |
| Admitted to ward, COPS <80, did experience transfer to ICU or TCU | 3,560 (1.7) | 9.8 | 2.93 (2.63‐3.26) | 12.0 19.0 | 7.52 (6.89‐8.15) |
| Admitted to ward, LAPS 20, no transfer to ICU or TCU | 46,492 (22.1) | 4.2 | 0.59 (0.56‐0.61) | 4.6 5.4 | 0.16 (0.12‐0.21) |
| Admitted to ward, LAPS 20, did experience transfer to ICU or TCU | 4,070 (1.9) | 21.4 | 2.37 (2.22‐2.54) | 14.8 21.0 | 8.76 (8.06‐9.47) |
| Admitted to ward, LAPS <20, no transfer to ICU or TCU | 66,334 (31.5) | 0.9 | 0.55 (0.51‐0.60) | 3.5 4.9 | 0.32 (0.28‐0.36) |
| Admitted to ward, LAPS <20, did experience transfer to ICU or TCU | 4,341 (2.1) | 9.5 | 4.31 (3.90‐4.74) | 11.8 18.1 | 7.12 (6.61‐7.64) |
Transfers to a higher level of care at a different hospital, which in the KPMCP are usually planned, experienced lower mortality than transfers within the same hospital. For ward to TCU transfers, intra‐hospital transfers had a mortality of 12.1% while inter‐hospital transfers had a mortality of 5.7%. Corresponding rates for ward to ICU transfers were 21.7% and 11.2%, and for TCU to ICU transfers the rates were 25.9% and 12.5%, respectively.
Among patients initially admitted to the ward, a model to predict the occurrence of a transfer to a higher level of care (within 48 hours after admission) that included age, sex, admission type, primary condition, LAPS, COPS, and interaction terms had poor discrimination, with an area under the receiver operator characteristic (c statistic) of only 0.64. The c statistic for a model to predict transfer after 48 hours was 0.66. The corresponding models for TCU admits had c statistics of 0.67 and 0.68. All four models had poor calibration.
Discussion
Using automated bed history data permits characterizing a patient population with disproportionate mortality and LOS: intra‐hospital transfers to special care units (ICUs or TCUs). Indeed, the largest subset of these patients (those initially admitted to the ward or TCU) constituted only 3.7% of all admissions, but accounted for 24.2% of all ICU admissions, 21.7% of all hospital deaths, and 13.2% of all hospital days. These patients also had very elevated OEMRs and OMELOS. Models based on age, sex, preadmission laboratory test results, and comorbidities did not predict the occurrence of these transfers.
We performed multivariate analyses to explore the degree to which electronically assigned preadmission severity scores could predict these transfers. These analyses found that, compared to our ability to predict inpatient or 30‐day mortality at the time of admission, which is excellent, our ability to predict the occurrence of transfer after admission is much more limited. These results highlight the limitations of severity scores that rely on automated data, which may not have adequate discrimination when it comes to determining the risk of an adverse outcome within a narrow time frame. For example, among the 121,237 patients initially admitted to the ward who did not experience an intra‐hospital transfer, the mean LAPS was 18.9, while the mean LAPS among the 6,484 ward patients who did experience a transfer was 25.5. Differences between the mean and median LAPS, COPS, and predicted mortality risk among transferred and non‐transferred patients were significant (P < 0.0001 for all comparisons). However, examination of the distribution of LAPS, COPS, and predicted mortality risk between these two groups of patients showed considerable overlap.
Our methodology resembles Silber et al.'s27, 28 concept of failure to rescue in that it focuses on events occurring after hospitalization. Silber et al. argue that a hospital's quality can be measured by quantifying the degree to which patients who experience new problems are successfully rescued. Furthermore, quantification of those situations where rescue attempts are unsuccessful is felt to be superior to simply comparing raw or adjusted mortality rates because these are primarily determined by underlying case mix. The primary difference between Silber et al.'s approach and ours is at the level of detailthey specified a specific set of complications, whereas our measure is more generic and would include patients with many of the complications specified by Silber et al.27, 28
Most of the patients transferred to a higher level of care in our cohort survived (ie, were rescued), indicating that intensive care is beneficial. However, the fact that these patients had elevated OEMRs and OMELOS indicates that the real challenge facing hospitalists involves the timing of provision of a beneficial intervention. In theory, improved timing could result from earlier detection of problems, which is the underlying rationale for employing rapid response teams. However, the fact that our electronic tools (LAPS, COPS) cannot predict patient deteriorations within a narrow time frame suggests that early detection will remain a major challenge. Manually assigned vital signs scores designed for this purpose do not have good discrimination either.29, 30 This raises the possibility that, though patient groups may differ in terms of overall illness severity and mortality risk, differences at the individual patient level may be too subtle for clinicians to detect. Future research may thus need to focus on scores that combine laboratory data, vital signs, trends in data,31, 32 and newer proteomic markers (eg, procalcitonin).33 We also found that most transfers occurred early (within <72 hours), raising the possibility that at least some of these transfers may involve issues around triage rather than sudden deterioration.
Our study has important limitations. Due to resource constraints and limited data availability, we could not characterize the patients as well as might be desirable; in particular, we could not make full determinations of the actual reasons for patients' transfer for all patients. Broadly speaking, transfer to a higher level of care could be due to inappropriate triage, appropriate (preventive) transfer (which could include transfer to a more richly staffed unit for a specific procedure), relentless progression of disease despite maximal therapy, the occurrence of management errors, patient and family uncertainty about goals of care or inadequate understanding of treatment options and prognoses, or a combination of these factors. We could not make these distinctions with currently available electronic data. This is also true of postsurgical patients, in whom it is difficult to determine which transfers to intensive care might be planned (eg, in the case of surgical procedures where ICU care is anticipated) as opposed to the occurrence of a deterioration during or following surgery. Another major limitation of this study is our inability to identify code or no code status electronically. The elapsed LOS at time of death among patients who experienced a transfer to a higher level of care (as compared to patients who died in the ward without ever experiencing intra‐hospital transfer) suggests, but does not prove, that prolonged efforts were being made to keep them alive. We were also limited in terms of having access to other process data (eg, physician staffing levels, provision and timing of palliative care). Having ICU severity of illness scores would have permitted us to compare our cohort to those of other recent studies showing elevated mortality rates among transfer patients,911 but we have not yet developed that capability.
Consideration of our study findings suggests a possible research agenda that could be implemented by hospitalist researchers. This agenda should emphasize three areas: detection, intervention, and reflection.
With respect to detection, attention needs to be paid to better tools for quantifying patient risk at the time a decision to admit to the ward is made. It is likely that such tools will need to combine the attributes of our severity score (LAPS) with those of the manually assigned scores.30, 34 In some cases, use of these tools could lead a physician to change the locus of admission from the ward to the TCU or ICU, which could improve outcomes by ensuring more timely provision of intensive care. Since problems with initial triage could be due to factors other than the failure to suspect or anticipate impending instability, future research should also include a cognitive component (eg, quantifying what proportion of subsequent patient deteriorations could be ascribed to missed diagnoses35). Additional work also needs to be done on developing mathematical models that can inform electronic monitoring of ward (not just ICU) patients.
Research on interventions that hospitalists can use to prevent the need for intensive care or to improve the rescue rate should take two routes. The first is a disease‐specific route, which builds on the fact that a relatively small set of conditions (pneumonia, sepsis, gastrointestinal bleeding) account for most transfers to a higher level of care. Condition‐specific protocols, checklists, and bundles36 tailored to a ward environment (as opposed to the ICU or to the entire hospital) might prevent deteriorations in these patients, as has been reported for sepsis.37 The second route is to improve the overall capabilities of rapid response and code blue teams. Such research would need to include a more careful assessment of what commonalities exist among patients who were and were not successfully rescued by these teams. This approach would probably yield more insights than the current literature, which focuses on whether rapid response teams are a good thing or not.
Finally, research also needs to be performed on how hospitalists reflect on adverse outcomes among ward patients. Greater emphasis needs to be placed on moving beyond trigger tool approaches that rely on manual chart review. In an era of expanding use of electronic medical record systems, more work needs to be done on how to harness these to provide hospitalists with better quantitative and risk‐adjusted information. This information should not be limited to simply reporting rates of transfers and deaths. Rather, finer distinctions must be provided with respect of the type of patients (ie, more diagnostic detail), the clinical status of patients (ie, more physiologic detail), as well as the effects of including or excluding patients in whom therapeutic options may be limited (ie, do not resuscitate and comfort care patients) on reported rates. Ideally, researchers should develop better process and outcomes measures that could be tested in collaborative networks that include multiple nonacademic general medical‐surgical wards.
Acknowledgements
The authors thank Drs. Paul Feigenbaum, Alan Whippy, Joseph V. Selby, and Philip Madvig for reviewing the manuscript and Ms. Jennifer Calhoun for formatting the manuscript.
Considerable research and public attention is being paid to the quantification, risk adjustment, and reporting of inpatient mortality.15 Inpatient mortality is reported as aggregate mortality (for all hospitalized patients or those with a specific diagnosis3, 6) or intensive care unit (ICU) mortality.7, 8 While reporting aggregate hospital or aggregate ICU mortality rates is useful, it is also important to develop reporting strategies that go beyond simply using data elements found in administrative databases (eg, diagnosis and procedure codes) to quantify practice variation. Ideally, such strategies would permit delineating processes of careparticularly those potentially under the control of hospitalists, not only intensiviststo identify improvement opportunities. One such process, which can be tracked using the bed history component of a patient's electronic medical record, is the transfer of patients between different units within the same hospital.
Several studies have documented that risk of ICU death is highest among patients transferred from general medical‐surgical wards, intermediate among direct admissions from the emergency department, and lowest among surgical admissions.911 Opportunities to reduce subsequent ICU mortality have been studied among ward patients who develop sepsis and are then transferred to the ICU,12 among patients who experience cardiac arrest,13, 14 as well as among patients with any physiological deterioration (eg, through the use of rapid response teams).1517 Most of these studies have been single‐center studies and/or studies reporting only an ICU denominator. While useful in some respects, such studies are less helpful to hospitalists, who would benefit from better understanding of the types of patients transferred and the total impact that transfers to a higher level of care make on general medical‐surgical wards. In addition, entities such as the Institute for Healthcare Improvement recommend the manual review of records of patients who were transferred from the ward to the ICU18 to identify performance improvement opportunities. While laudable, such approaches do not lend themselves to automated reporting strategies.
We recently described a new risk adjustment methodology for inpatient mortality based entirely on automated data preceding hospital admission and not restricted to ICU patients. This methodology, which has been externally validated in Ottawa, Canada, after development in the Kaiser Permanente Medical Care Program (KPMCP), permits quantification of a patient's pre‐existing comorbidity burden, physiologic derangement at the time of admission, and overall inpatient mortality risk.19, 20 The primary purpose of this study was to combine this methodology with bed history analysis to quantify the in‐hospital mortality and length of stay (LOS) of patients who experienced intra‐hospital transfers in a large, multihospital system. As a secondary goal, we also wanted to assess the degree to which these transfers could be predicted based on information available prior to a patient's admission.
ABBREVIATIONS AND TERMS USED IN TEXT
COPS: COmorbidity Point Score. Point score based on a patient's health care utilization diagnoses (during the year preceding admission to the hospital. Analogous to POA (present on admission) coding. Scores can range from 0 to a theoretical maximum of 701 but scores >200 are rare. With respect to a patient's pre‐existing comorbidity burden, the unadjusted relationship of COPS and inpatient mortality is as follows: a COPS <50 is associated with a mortality risk of <1%, <100 with a mortality risk of <5%, 100 to 145 with a mortality risk of 5% to 10%, and >145 with a mortality risk of 10% or more.
ICU: Intensive Care Unit. In this study, all ICUs have a minimum registered nurse to patient ratio of 1:2.
LAPS: Laboratory Acute Physiology Score. Point score based on 14 laboratory test results obtained in the 72 hours preceding hospitalization. With respect to a patient's physiologic derangement, the unadjusted relationship of LAPS and inpatient mortality is as follows: a LAPS <7 is associated with a mortality risk of <1%, <7 to 30 with a mortality risk of <5%, 30 to 60 with a mortality risk of 5% to 9%, and >60 with a mortality risk of 10% or more.
LOS: Exact hospital Length Of Stay. LOS is calculated from admission until first discharge home (i.e., it may span more than one hospital stay if a patient experienced inter‐hospital transport).
Predicted (expected) mortality risk: the % risk of death for a given patient based on his/her age, sex, admission diagnosis, COPS, and LAPS.
OEMR: Observed to Expected Mortality Ratio. For a given patient subset, the ratio of the actual mortality experienced by the subset to the expected (predicted) mortality for the subset. Predicted mortality is based on patients' age, sex, admission diagnosis, COPS, and LAPS.
OMELOS: Observed Minus Expected LOS. For a given patient subset, the difference between the actual number of hospital days experienced by the subset and the expected (predicted) number of hospital days for the subset. Predicted LOS is based on patients' age, sex, admission diagnosis, COPS, and LAPS.
TCU: Transitional Care Unit (also called intermediate care unit or stepdown unit). In this study, TCUs have variable nurse to patient ratios ranging from 1:2.5 to 1:3 and did not provide assisted ventilation, continuous pressor infusions, or invasive monitoring.
Materials and Methods
This project was approved by the Northern California KPMCP Institutional Review Board for the Protection of Human Subjects.
The Northern California KPMCP serves a total population of approximately 3.3 million members. Under a mutual exclusivity arrangement, physicians of The Permanente Medical Group, Inc., care for Kaiser Foundation Health Plan, Inc. members at facilities owned by Kaiser Foundation Hospitals, Inc. All Northern California KPMCP hospitals and clinics employ the same information systems with a common medical record number and can track care covered by the plan but delivered elsewhere. Databases maintained by the KPMCP capture admission and discharge times, admission and discharge diagnoses and procedures (assigned by professional coders), bed histories, inter‐hospital transfers, as well as the results of all inpatient and outpatient laboratory tests. The use of these databases for research has been described in multiple reports.2124
Our setting consisted of all 19 hospitals owned and operated by the KPMCP, whose characteristics are summarized in the Supporting Information Appendix available to interested readers. These include the 17 described in our previous report19 as well as 2 new hospitals (Antioch and Manteca) which are similar in size and type of population served. Our study population consisted of all patients admitted to these 19 hospitals who met these criteria: 1) hospitalization began from November 1st, 2006 through January 31st, 2008; 2) initial hospitalization occurred at a Northern California KPMCP hospital (ie, for inter‐hospital transfers, the first hospital stay occurred within the KPMCP); 3) age 15 years; and 4) hospitalization was not for childbirth.
We defined a linked hospitalization as the time period that began with a patient's admission to the hospital and ended with the patient's discharge (home, to a nursing home, or death). Linked hospitalizations can thus involve more than 1 hospital stay and could include a patient transfer from one hospital to another prior to definitive discharge. For linked hospitalizations, mortality was attributed to the admitting KPMCP hospital (ie, if a patient was admitted to hospital A, transferred to B, and died at hospital B, mortality was attributed to hospital A). We defined total LOS as the exact time in hours from when a patient was first admitted to the hospital until death or final discharge home or to a nursing home, while total ICU or transitional care unit (TCU, referred to as stepdown unit in some hospitals) LOS was calculated for all individual ICU or TCU stays during the hospital stay.
Intra‐Hospital Transfers
We grouped all possible hospital units into four types: general medical‐surgical ward (henceforth, ward); operating room (OR)/post‐anesthesia recovery (PAR); TCU; and ICU. In 2003, the KPMCP implemented a mandatory minimum staffing ratio of one registered nurse for every four patients in all its hospital units; in addition, staffing levels for designated ICUs adhered to the previously mandated minimum of one nurse for every 2 patients. So long as they adhere to these minimum ratios, individual hospitals have considerable autonomy with respect to how they staff or designate individual hospital units. Registered nurse‐to‐patient ratios during the time of this study were as follows: ward patients, 1:3.5 to 1:4; TCU patients, 1:2.5 to 1:3; and ICU patients, 1:1 to 1:2. Staffing ratios for the OR and PAR are more variable, depending on the surgical procedures involved. Current KPMCP databases do not permit accurate quantification of physician staffing. All 19 study hospitals had designated ICUs, 6 were teaching hospitals, and 11 had designated TCUs. None of the study hospitals had closed ICUs (units where only intensivists admit patients) and none had continuous coverage of the ICU by intensivists. While we were not able to employ electronic data to determine who made the decision to transfer, we did find considerable variation with respect to how intensivists covered the ICUs and how they interfaced with hospitalists. Staffing levels for specialized coronary care units and non‐ICU monitored beds were not standardized. All study hospitals had rapid response teams as well as code blue teams during the time period covered by this report. Respiratory care practitioners were available to patients in all hospital units, but considerable variation existed with respect to other services available (eg, cardiac catheterization units, provision of noninvasive positive pressure ventilation outside the ICU, etc.).
This report focuses on intra‐hospital transfers to the ICU and TCU, with special emphasis on nonsurgical transfers (due to space limitations, we are not reporting on the outcomes of patients whose first hospital unit was the OR; additional details on these patients are provided in the Supporting Information Appendix). For the purposes of this report, we defined the following admission types: direct admits (patients admitted to the ICU or TCU whose first hospital unit on admission was the ICU or TCU); and nonsurgical transfers to a higher level of care. These latter transfers could be of 3 types: ward to ICU, ward to TCU, and TCU to ICU. We also quantified the effect of inter‐hospital transfers.
Independent Variables
In addition to patients' age and sex, we employed the following independent variables to predict transfer to a higher level of care. These variables are part of the risk adjustment model described in greater detail in our previous report19 and were available electronically for all patients in the cohort. We grouped admission diagnoses into 44 broad diagnostic categories (Primary Conditions), and admission types into 4 groups (emergency medical, emergency surgical, elective medical, and elective surgical). We quantified patients' degree of physiologic derangement using a Laboratory‐based Acute Physiology Score (LAPS) using laboratory test results prior to hospitalization. We quantified patients' comorbid illness burden using a Comorbidity Point Score (COPS) based on patients' pre‐existing diagnoses over the 12‐month period preceding hospitalization. Lastly, we assigned each patient a predicted mortality risk (%) and LOS based on the above predictors,19 permitting calculation of observed to expected mortality ratios (OEMRs) and observed minus expected LOS (OMELOS).
Statistical Methods
All analyses were performed in SAS.25 We calculated standard descriptive statistics (medians, means, standard deviations) and compared different patient groupings using t and chi‐square tests. We employed a similar approach to that reported by Render et al.7 to calculate OEMR and OMELOS.
To determine the degree to which transfers to a higher level of care from the ward or TCU would be predictable using information available at the time of admission, we performed 4 sets of logistic regression analyses using the above‐mentioned predictors in which the outcome variables were as follows: 1) transfer occurring in the first 48 hours after admission (time frame by which point approximately half of the transferred patients experienced a transfer) among ward or TCU patients and 2) transfer occurring after 48 hours among ward or TCU patients. We evaluated the discrimination and calibration of these models using the same methods described in our original report (measuring the area under the receiver operator characteristic curve, or c statistic, and visually examining observed and expected mortality rates among predicted risk bands as well as risk deciles) as well as additional statistical tests recommended by Cook.19, 26
Results
During the study period, a total of 249,129 individual hospital stays involving 170,151 patients occurred at these 19 hospitals. After concatenation of inter‐hospital transfers, we were left with 237,208 linked hospitalizations. We excluded 26,738 linked hospitalizations that began at a non‐KPMCP hospital (ie, they were transported in), leaving a total of 210,470 linked hospitalizations involving 150,495 patients. The overall linked hospitalization mortality rate was 3.30%.
Table 1 summarizes cohort characteristics based on initial hospital location. On admission, ICU patients had the highest degree of physiologic derangement as well as the highest predicted mortality. Considerable inter‐hospital variation was present in both predictors and outcomes; details on these variations are provided in the Supporting Information Appendix.
| Ward | TCU | ICU | All* | |
|---|---|---|---|---|
| ||||
| n | 121,237 | 20,556 | 16,001 | 210,470 |
| Admitted via emergency department, n (%) | 99,909 (82.4) | 18,612 (90.5) | 13,847 (86.5) | 139,036 (66.1) |
| % range across hospitals | 55.0‐94.2 | 64.7‐97.6 | 49.5‐97.4 | 53.6‐76.9 |
| Male, n (%) | 53,744 (44.3) | 10,362 (50.4) | 8,378 (52.4) | 94,451 (44.9) |
| Age in years (mean SD) | 64.5 19.2 | 69.0 15.6 | 63.7 17.8 | 63.2 18.6 |
| LAPS (mean SD) | 19.2 18.0 | 23.3 19.5 | 31.7 25.7 | 16.7 19.0 |
| COPS (mean SD) | 90.4 64.0 | 99.2 65.9 | 94.5 67.5 | 84.7 61.8 |
| % predicted mortality (mean SD) | 4.0 7.1 | 4.6 7.3 | 8.7 12.8 | 3.6 7.3 |
| Observed in‐hospital deaths (n, %) | 3,793 (3.1) | 907 (4.4) | 1,995 (12.5) | 6,952 (3.3) |
| Observed to expected mortality ratio | 0.79 (0.77‐0.82) | 0.95 (0.89‐1.02) | 1.43 (1.36‐1.49) | 0.92 (0.89‐0.94) |
| Total hospital LOS, days (mean SD) | 4.6 7.5 | 5.3 10.0 | 7.8 14.0 | 4.6 8.1 |
Table 2 summarizes data from 3 groups of patients: patients initially admitted to the ward, or TCU, who did not experience a transfer to a higher level of care and patients admitted to these 2 units who did experience such a transfer. Patients who experienced a transfer constituted 5.3% (6,484/121,237) of ward patients and 6.7% (1,384/20,556) of TCU patients. Transferred patients tended to be older, have more acute physiologic derangement (higher LAPS), a greater pre‐existing illness burden (higher COPS), and a higher predicted mortality risk. Among ward patients, those with the following admission diagnoses were most likely to experience a transfer to a higher level of care: gastrointestinal bleeding (10.8% of all transfers), pneumonia (8.7%), and other infections (8.2%). The diagnoses most likely to be associated with death following transfer were cancer (death rate among transferred patients, 48%), renal disease (death rate, 36%), and liver disease (33%). Similar distributions were observed for TCU patients.
| Patients Initially Admitted to Ward, Remained There | Patients Initially Admitted to TCU, Remained There | Patients Transferred to Higher Level of Care | All | |
|---|---|---|---|---|
| ||||
| n | 114,753 | 19,172 | 7,868 | 141,793 |
| Male, n (%) | 50,586 (44.1) | 9,626 (50.2) | 3,894 (49.5) | 64,106 (45.2) |
| Age (mean SD) | 64.3 19.4 | 69.0 15.7 | 68.1 16.1 | 65.2 18.8 |
| LAPS (mean SD) | 18.9 17.8 | 22.7 19.1 | 26.7 21.0 | 19.8 18.3 |
| COPS (mean SD) | 89.4 63.7 | 98.3 65.5 | 107.9 67.6 | 91.7 64.4 |
| % predicted mortality risk (mean SD) | 3.8 7.0 | 4.4 7.0 | 6.5 8.8 | 4.1 7.1 |
| Admission diagnosis of pneumonia, n (%) | 5,624 (4.9) | 865 (4.5) | 684 (8.7) | 7,173 (5.1) |
| Admission diagnosis of sepsis, n (%) | 1,181 (1.0) | 227 (1.2) | 168 (2.1) | 1,576 (1.1) |
| Admission diagnosis of GI bleed, n (%) | 13,615 (11.9) | 1,448 (7.6) | 851 (10.8) | 15,914 (11.2) |
| Admission diagnosis of cancer, n (%) | 2,406 (2.1) | 80 (0.4) | 186 (2.4) | 2,672 (1.9) |
Table 3 compares outcomes among ward and TCU patients who did and did not experience a transfer to a higher level of care. The table shows that transferred patients were almost 3 times as likely to die, even after controlling for severity of illness, and that their hospital LOS was 9 days higher than expected. This increased risk was seen in all hospitals and among all transfer types (ward to ICU, ward to TCU, and TCU to ICU).
| Patients Initially Admitted to Ward, Remained There | Patients Initially Admitted to TCU, Remained There | Patients Transferred to Higher Level of Care | |
|---|---|---|---|
| |||
| n | 114,753 | 19,172 | 7,868 |
| Admitted to ICU, n (%) | 0 (0.0) | 0 (0.0) | 5,245 (66.7) |
| Ventilated, n (%) | 0 (0.0) | 0 (0.0) | 1,346 (17.1) |
| Died in the hospital, n (%) | 2,619 (2.3) | 572 (3.0) | 1,509 (19.2) |
| Length of stay, in days, at time of death (mean SD) | 7.0 11.9 | 8.3 12.4 | 16.2 23.7 |
| Observed to expected mortality ratio (95% CI) | 0.60 (0.57‐0.62) | 0.68 (0.63‐0.74) | 2.93 (2.79‐3.09) |
| Total hospital length of stay, days (mean SD) | 4.0 5.7 | 4.4 6.9 | 14.3 21.3 |
| Observed minus expected length of stay (95% CI) | 0.4 (0.3‐0.4) | 0.8 (0.7‐0.9) | 9.1 (8.6‐9.5) |
| Length of stay, in hours, at time of transfer (mean SD) | 80.8 167.2 | ||
Table 3 also shows that, among decedent patients, those who never left the ward or TCU died much sooner than those who died following transfer. Among direct admits to the ICU, the median LOS at time of death was 3.9 days, with a mean of 9.4 standard deviation of 19.9 days, while the corresponding times for TCU direct admits were a median and mean LOS of 6.5 and 11.7 19.5 days.
Table 4 summarizes outcomes among different patient subgroups that did and did not experience a transfer to a higher level of care. Based on location, patients who experienced a transfer from the TCU to the ICU had the highest crude death rate, but patients transferred from the ward to the ICU had the highest OEMR. On the other hand, if one divides patients by the degree of physiologic derangement, patients with low LAPS who experienced a transfer had the highest OEMR. With respect to LOS, patients transferred from the TCU to the ICU had the highest OMELOS (13.4 extra days).
| n (%)* | Death Rate (%) | OEMR | LOS (mean SD) | OMELOS | |
|---|---|---|---|---|---|
| |||||
| Never admitted to TCU or ICU | 157,632 (74.9) | 1.6 | 0.55 (0.53‐0.57) | 3.6 4.6 | 0.04 (0.02‐0.07) |
| Direct admit to TCU | 18,464 (8.8) | 2.9 | 0.66 (0.61‐0.72) | 4.2 5.8 | 0.60 (0.52‐0.68) |
| Direct admit to ICU | 14,655 (7.0) | 11.9 | 1.38 (1.32‐1.45) | 6.4 9.4 | 2.28 (2.14‐2.43) |
| Transferred from ward to ICU | 5,145 (2.4) | 21.5 | 3.23 (3.04‐3.42) | 15.7 21.6 | 10.33 (9.70‐10.96) |
| Transferred from ward to TCU | 3,144 (1.5) | 11.9 | 1.99 (1.79‐2.20) | 13.6 23.2 | 8.02 (7.23‐8.82) |
| Transferred from TCU to ICU | 1,107 (0.5) | 25.7 | 2.94 (2.61‐3.31) | 18.0 28.2 | 13.35 (11.49‐15.21) |
| Admitted to ward, COPS 80, no transfer to ICU or TCU | 55,405 (26.3) | 3.4 | 0.59 (0.56‐0.62) | 4.5 5.9 | 0.29 (0.24‐0.34) |
| Admitted to ward, COPS 80, did experience transfer to ICU or TCU | 4,851 (2.3) | 19.3 | 2.72 (2.55‐2.90) | 14.2 20.0 | 8.14 (7.56‐8.71) |
| Admitted to ward, COPS <80, no transfer to ICU or TCU | 57,421 (27.3) | 1.1 | 0.55 (0.51‐0.59) | 3.4 4.2 | 0.23 (0.19‐0.26) |
| Admitted to ward, COPS <80, did experience transfer to ICU or TCU | 3,560 (1.7) | 9.8 | 2.93 (2.63‐3.26) | 12.0 19.0 | 7.52 (6.89‐8.15) |
| Admitted to ward, LAPS 20, no transfer to ICU or TCU | 46,492 (22.1) | 4.2 | 0.59 (0.56‐0.61) | 4.6 5.4 | 0.16 (0.12‐0.21) |
| Admitted to ward, LAPS 20, did experience transfer to ICU or TCU | 4,070 (1.9) | 21.4 | 2.37 (2.22‐2.54) | 14.8 21.0 | 8.76 (8.06‐9.47) |
| Admitted to ward, LAPS <20, no transfer to ICU or TCU | 66,334 (31.5) | 0.9 | 0.55 (0.51‐0.60) | 3.5 4.9 | 0.32 (0.28‐0.36) |
| Admitted to ward, LAPS <20, did experience transfer to ICU or TCU | 4,341 (2.1) | 9.5 | 4.31 (3.90‐4.74) | 11.8 18.1 | 7.12 (6.61‐7.64) |
Transfers to a higher level of care at a different hospital, which in the KPMCP are usually planned, experienced lower mortality than transfers within the same hospital. For ward to TCU transfers, intra‐hospital transfers had a mortality of 12.1% while inter‐hospital transfers had a mortality of 5.7%. Corresponding rates for ward to ICU transfers were 21.7% and 11.2%, and for TCU to ICU transfers the rates were 25.9% and 12.5%, respectively.
Among patients initially admitted to the ward, a model to predict the occurrence of a transfer to a higher level of care (within 48 hours after admission) that included age, sex, admission type, primary condition, LAPS, COPS, and interaction terms had poor discrimination, with an area under the receiver operator characteristic (c statistic) of only 0.64. The c statistic for a model to predict transfer after 48 hours was 0.66. The corresponding models for TCU admits had c statistics of 0.67 and 0.68. All four models had poor calibration.
Discussion
Using automated bed history data permits characterizing a patient population with disproportionate mortality and LOS: intra‐hospital transfers to special care units (ICUs or TCUs). Indeed, the largest subset of these patients (those initially admitted to the ward or TCU) constituted only 3.7% of all admissions, but accounted for 24.2% of all ICU admissions, 21.7% of all hospital deaths, and 13.2% of all hospital days. These patients also had very elevated OEMRs and OMELOS. Models based on age, sex, preadmission laboratory test results, and comorbidities did not predict the occurrence of these transfers.
We performed multivariate analyses to explore the degree to which electronically assigned preadmission severity scores could predict these transfers. These analyses found that, compared to our ability to predict inpatient or 30‐day mortality at the time of admission, which is excellent, our ability to predict the occurrence of transfer after admission is much more limited. These results highlight the limitations of severity scores that rely on automated data, which may not have adequate discrimination when it comes to determining the risk of an adverse outcome within a narrow time frame. For example, among the 121,237 patients initially admitted to the ward who did not experience an intra‐hospital transfer, the mean LAPS was 18.9, while the mean LAPS among the 6,484 ward patients who did experience a transfer was 25.5. Differences between the mean and median LAPS, COPS, and predicted mortality risk among transferred and non‐transferred patients were significant (P < 0.0001 for all comparisons). However, examination of the distribution of LAPS, COPS, and predicted mortality risk between these two groups of patients showed considerable overlap.
Our methodology resembles Silber et al.'s27, 28 concept of failure to rescue in that it focuses on events occurring after hospitalization. Silber et al. argue that a hospital's quality can be measured by quantifying the degree to which patients who experience new problems are successfully rescued. Furthermore, quantification of those situations where rescue attempts are unsuccessful is felt to be superior to simply comparing raw or adjusted mortality rates because these are primarily determined by underlying case mix. The primary difference between Silber et al.'s approach and ours is at the level of detailthey specified a specific set of complications, whereas our measure is more generic and would include patients with many of the complications specified by Silber et al.27, 28
Most of the patients transferred to a higher level of care in our cohort survived (ie, were rescued), indicating that intensive care is beneficial. However, the fact that these patients had elevated OEMRs and OMELOS indicates that the real challenge facing hospitalists involves the timing of provision of a beneficial intervention. In theory, improved timing could result from earlier detection of problems, which is the underlying rationale for employing rapid response teams. However, the fact that our electronic tools (LAPS, COPS) cannot predict patient deteriorations within a narrow time frame suggests that early detection will remain a major challenge. Manually assigned vital signs scores designed for this purpose do not have good discrimination either.29, 30 This raises the possibility that, though patient groups may differ in terms of overall illness severity and mortality risk, differences at the individual patient level may be too subtle for clinicians to detect. Future research may thus need to focus on scores that combine laboratory data, vital signs, trends in data,31, 32 and newer proteomic markers (eg, procalcitonin).33 We also found that most transfers occurred early (within <72 hours), raising the possibility that at least some of these transfers may involve issues around triage rather than sudden deterioration.
Our study has important limitations. Due to resource constraints and limited data availability, we could not characterize the patients as well as might be desirable; in particular, we could not make full determinations of the actual reasons for patients' transfer for all patients. Broadly speaking, transfer to a higher level of care could be due to inappropriate triage, appropriate (preventive) transfer (which could include transfer to a more richly staffed unit for a specific procedure), relentless progression of disease despite maximal therapy, the occurrence of management errors, patient and family uncertainty about goals of care or inadequate understanding of treatment options and prognoses, or a combination of these factors. We could not make these distinctions with currently available electronic data. This is also true of postsurgical patients, in whom it is difficult to determine which transfers to intensive care might be planned (eg, in the case of surgical procedures where ICU care is anticipated) as opposed to the occurrence of a deterioration during or following surgery. Another major limitation of this study is our inability to identify code or no code status electronically. The elapsed LOS at time of death among patients who experienced a transfer to a higher level of care (as compared to patients who died in the ward without ever experiencing intra‐hospital transfer) suggests, but does not prove, that prolonged efforts were being made to keep them alive. We were also limited in terms of having access to other process data (eg, physician staffing levels, provision and timing of palliative care). Having ICU severity of illness scores would have permitted us to compare our cohort to those of other recent studies showing elevated mortality rates among transfer patients,911 but we have not yet developed that capability.
Consideration of our study findings suggests a possible research agenda that could be implemented by hospitalist researchers. This agenda should emphasize three areas: detection, intervention, and reflection.
With respect to detection, attention needs to be paid to better tools for quantifying patient risk at the time a decision to admit to the ward is made. It is likely that such tools will need to combine the attributes of our severity score (LAPS) with those of the manually assigned scores.30, 34 In some cases, use of these tools could lead a physician to change the locus of admission from the ward to the TCU or ICU, which could improve outcomes by ensuring more timely provision of intensive care. Since problems with initial triage could be due to factors other than the failure to suspect or anticipate impending instability, future research should also include a cognitive component (eg, quantifying what proportion of subsequent patient deteriorations could be ascribed to missed diagnoses35). Additional work also needs to be done on developing mathematical models that can inform electronic monitoring of ward (not just ICU) patients.
Research on interventions that hospitalists can use to prevent the need for intensive care or to improve the rescue rate should take two routes. The first is a disease‐specific route, which builds on the fact that a relatively small set of conditions (pneumonia, sepsis, gastrointestinal bleeding) account for most transfers to a higher level of care. Condition‐specific protocols, checklists, and bundles36 tailored to a ward environment (as opposed to the ICU or to the entire hospital) might prevent deteriorations in these patients, as has been reported for sepsis.37 The second route is to improve the overall capabilities of rapid response and code blue teams. Such research would need to include a more careful assessment of what commonalities exist among patients who were and were not successfully rescued by these teams. This approach would probably yield more insights than the current literature, which focuses on whether rapid response teams are a good thing or not.
Finally, research also needs to be performed on how hospitalists reflect on adverse outcomes among ward patients. Greater emphasis needs to be placed on moving beyond trigger tool approaches that rely on manual chart review. In an era of expanding use of electronic medical record systems, more work needs to be done on how to harness these to provide hospitalists with better quantitative and risk‐adjusted information. This information should not be limited to simply reporting rates of transfers and deaths. Rather, finer distinctions must be provided with respect of the type of patients (ie, more diagnostic detail), the clinical status of patients (ie, more physiologic detail), as well as the effects of including or excluding patients in whom therapeutic options may be limited (ie, do not resuscitate and comfort care patients) on reported rates. Ideally, researchers should develop better process and outcomes measures that could be tested in collaborative networks that include multiple nonacademic general medical‐surgical wards.
Acknowledgements
The authors thank Drs. Paul Feigenbaum, Alan Whippy, Joseph V. Selby, and Philip Madvig for reviewing the manuscript and Ms. Jennifer Calhoun for formatting the manuscript.
- ,,.To Err is Human: Building a Safer Health System.Washington, D. C.:National Academy Press;2000.
- Institute for Healthcare Improvement. Protecting 5 million lives from harm. Available at: http://www.ihi.org/IHI/Programs/Campaign. Accessed June2010.
- ,.Identifying poor‐quality hospitals. Can hospital mortality rates detect quality problems for medical diagnoses?Med Care.1996;34(8):737–753.
- ,,.Surgical mortality as an indicator of hospital quality: the problem with small sample size.JAMA.2004;292(7):847–851.
- State of California Office of Statewide Health Planning and Development. AHRQ ‐ Inpatient quality indicators (IQIs) hospital inpatient mortality indicators for California. Available at: http://www.oshpd.ca.gov/HID/Products/PatDischargeData/AHRQ/iqi‐imi_overview.html. Accessed June2010.
- ,,, et al.Enhancement of claims data to improve risk adjustment of hospital mortality.JAMA.2007;297(1):71–76.
- ,,, et al.Variation in outcomes in Veterans Affairs intensive care units with a computerized severity measure.Crit Care Med.2005;33(5):930–939.
- ,,,.Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today's critically ill patients.Crit Care Med.2006;34(5):1297–1310.
- ,,,.Day of the week of intensive care admission and patient outcomes: a multisite regional evaluation.Med Care.2002;40(6):530–539.
- ,,, et al.The hospital mortality of patients admitted to the ICU on weekends.Chest.2004;126(4):1292–1298.
- ,,, et al.Mortality among patients admitted to intensive care units during weekday day shifts compared with “off” hours.Crit Care Med.2007;35(1):3–11.
- ,,, et al.Septic shock: an analysis of outcomes for patients with onset on hospital wards versus intensive care units.Crit Care Med.1998;26(6):1020–1024.
- ,,,,.Clinical antecedents to in‐hospital cardiopulmonary arrest.Chest.1990;98(6):1388–1392.
- ,.Developing strategies to prevent inhospital cardiac arrest: analyzing responses of physicians and nurses in the hours before the event.Crit Care Med.1994;22(2):244–247.
- MERIT Study Investigators.Introduction of the medical emergency team (MET) system: a cluster‐randomized controlled trial.Lancet.2005;365(9477):2091–2097.
- Institute for Healthcare Improvement.The “MERIT” Trial of Medical Emergency Teams in Australia: An Analysis of Findings and Implications.Boston, MA:2005. Available on www.ihi.org
- ,,.Rapid response teams‐‐walk, don't run.JAMA.2006;296(13):1645–1647.
- ,.IHI Global Trigger Tool for Measuring Adverse Events.2nd ed.Cambridge, Massachusetts:Institute for Healthcare Improvement;2009.
- ,,,,,.Risk adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases.Medical Care.2008;46(3):232–239.
- ,,,.The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population.J Clin Epidemiol.2010;63(7):798–803.
- .Linking automated databases for research in managed care settings.Ann Intern Med.1997;127(8 Pt 2):719–724.
- ,,, et al.Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice?JAMA.2003;290(20):2685–2692.
- ,,, et al.Richardson score predicts short‐term adverse respiratory outcomes in newborns >/=34 weeks gestation.J Pediatr.2004;145(6):754–760.
- ,,, et al.Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases.Am J Manag Care.2008;14(3):158–166.
- Statistical Analysis Software [computer program]. Version 8.Cary, NC:SAS Institute, Inc.;2000.
- .Use and misuse of the receiver operating characteristic curve in risk prediction.Circulation.2007;115(7):928–935.
- ,,,.Hospital and patient characteristics associated with death after surgery. A study of adverse occurrence and failure to rescue.Med Care.1992;30(7):615–629.
- ,,.Comparing the contributions of groups of predictors: which outcomes vary with hospital rather than patient characteristics?J Am Stat Assoc.1995;90(429):7–18.
- ,.Beyond the intensive care unit: A review of interventions aimed at anticipating and preventing in‐hospital cardiopulmonary arrest.Resuscitation.2005;67(1):13–23.
- ,,.Reproducibility of physiological track‐and‐trigger warning systems for identifying at‐risk patients on the ward.Intensive Care Med.2007;33(4):619–624.
- ,,,,.Serial evaluation of the SOFA score to predict outcome in critically ill patients.JAMA.2001;286(14):1754–1758.
- ,,.Incorporation of Physiologic Trend and Interaction Effects in Neonatal Severity of Illness Scores: An Experiment Using a Variant of the Richardson Score.Intensive Care Med.2007;33(9):1602–1608.
- ,,, et al.Diagnostic and prognostic value of procalcitonin in patients with septic shock.Crit Care Med.2004;32(5):1166–1169.
- ,,, et al.Identifying the sick: can biochemical measurements be used to aid decision making on presentation to the accident and emergency department.Br J Anaesth.2005;94(6):735–741.
- .Improving patient care. The cognitive psychology of missed diagnoses.Ann Intern Med.2005;142(2):115–120.
- ,,,,,.Using care bundles to reduce in‐hospital mortality: quantitative survey.BMJ.2010;340:c1234.
- ,,, et al.Effect of a rapid response system for patients in shock on time to treatment and mortality during 5 years.Crit Care Med.2007;35(11):2568–2575.
- ,,.To Err is Human: Building a Safer Health System.Washington, D. C.:National Academy Press;2000.
- Institute for Healthcare Improvement. Protecting 5 million lives from harm. Available at: http://www.ihi.org/IHI/Programs/Campaign. Accessed June2010.
- ,.Identifying poor‐quality hospitals. Can hospital mortality rates detect quality problems for medical diagnoses?Med Care.1996;34(8):737–753.
- ,,.Surgical mortality as an indicator of hospital quality: the problem with small sample size.JAMA.2004;292(7):847–851.
- State of California Office of Statewide Health Planning and Development. AHRQ ‐ Inpatient quality indicators (IQIs) hospital inpatient mortality indicators for California. Available at: http://www.oshpd.ca.gov/HID/Products/PatDischargeData/AHRQ/iqi‐imi_overview.html. Accessed June2010.
- ,,, et al.Enhancement of claims data to improve risk adjustment of hospital mortality.JAMA.2007;297(1):71–76.
- ,,, et al.Variation in outcomes in Veterans Affairs intensive care units with a computerized severity measure.Crit Care Med.2005;33(5):930–939.
- ,,,.Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today's critically ill patients.Crit Care Med.2006;34(5):1297–1310.
- ,,,.Day of the week of intensive care admission and patient outcomes: a multisite regional evaluation.Med Care.2002;40(6):530–539.
- ,,, et al.The hospital mortality of patients admitted to the ICU on weekends.Chest.2004;126(4):1292–1298.
- ,,, et al.Mortality among patients admitted to intensive care units during weekday day shifts compared with “off” hours.Crit Care Med.2007;35(1):3–11.
- ,,, et al.Septic shock: an analysis of outcomes for patients with onset on hospital wards versus intensive care units.Crit Care Med.1998;26(6):1020–1024.
- ,,,,.Clinical antecedents to in‐hospital cardiopulmonary arrest.Chest.1990;98(6):1388–1392.
- ,.Developing strategies to prevent inhospital cardiac arrest: analyzing responses of physicians and nurses in the hours before the event.Crit Care Med.1994;22(2):244–247.
- MERIT Study Investigators.Introduction of the medical emergency team (MET) system: a cluster‐randomized controlled trial.Lancet.2005;365(9477):2091–2097.
- Institute for Healthcare Improvement.The “MERIT” Trial of Medical Emergency Teams in Australia: An Analysis of Findings and Implications.Boston, MA:2005. Available on www.ihi.org
- ,,.Rapid response teams‐‐walk, don't run.JAMA.2006;296(13):1645–1647.
- ,.IHI Global Trigger Tool for Measuring Adverse Events.2nd ed.Cambridge, Massachusetts:Institute for Healthcare Improvement;2009.
- ,,,,,.Risk adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases.Medical Care.2008;46(3):232–239.
- ,,,.The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population.J Clin Epidemiol.2010;63(7):798–803.
- .Linking automated databases for research in managed care settings.Ann Intern Med.1997;127(8 Pt 2):719–724.
- ,,, et al.Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice?JAMA.2003;290(20):2685–2692.
- ,,, et al.Richardson score predicts short‐term adverse respiratory outcomes in newborns >/=34 weeks gestation.J Pediatr.2004;145(6):754–760.
- ,,, et al.Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases.Am J Manag Care.2008;14(3):158–166.
- Statistical Analysis Software [computer program]. Version 8.Cary, NC:SAS Institute, Inc.;2000.
- .Use and misuse of the receiver operating characteristic curve in risk prediction.Circulation.2007;115(7):928–935.
- ,,,.Hospital and patient characteristics associated with death after surgery. A study of adverse occurrence and failure to rescue.Med Care.1992;30(7):615–629.
- ,,.Comparing the contributions of groups of predictors: which outcomes vary with hospital rather than patient characteristics?J Am Stat Assoc.1995;90(429):7–18.
- ,.Beyond the intensive care unit: A review of interventions aimed at anticipating and preventing in‐hospital cardiopulmonary arrest.Resuscitation.2005;67(1):13–23.
- ,,.Reproducibility of physiological track‐and‐trigger warning systems for identifying at‐risk patients on the ward.Intensive Care Med.2007;33(4):619–624.
- ,,,,.Serial evaluation of the SOFA score to predict outcome in critically ill patients.JAMA.2001;286(14):1754–1758.
- ,,.Incorporation of Physiologic Trend and Interaction Effects in Neonatal Severity of Illness Scores: An Experiment Using a Variant of the Richardson Score.Intensive Care Med.2007;33(9):1602–1608.
- ,,, et al.Diagnostic and prognostic value of procalcitonin in patients with septic shock.Crit Care Med.2004;32(5):1166–1169.
- ,,, et al.Identifying the sick: can biochemical measurements be used to aid decision making on presentation to the accident and emergency department.Br J Anaesth.2005;94(6):735–741.
- .Improving patient care. The cognitive psychology of missed diagnoses.Ann Intern Med.2005;142(2):115–120.
- ,,,,,.Using care bundles to reduce in‐hospital mortality: quantitative survey.BMJ.2010;340:c1234.
- ,,, et al.Effect of a rapid response system for patients in shock on time to treatment and mortality during 5 years.Crit Care Med.2007;35(11):2568–2575.
Copyright © 2010 Society of Hospital Medicine
Four Nephrology Myths Debunked
There are many controversial topics relating to renal disease in hospitalized patients. The aim of this review is to shed light on some important and often debated issues. We will first discuss topics related to electrolytes disorder commonly seen in hospitalized patients (hyponatremia, hyperkalemia, metabolic acidosis) then the use of diuretics in patients with allergy to sulfa containing antibiotics.
Hypothyroidism and Hyponatremia
Hyponatremia is common in hospitalized patients and is associated with worse outcomes.1 It can be seen with a variety of conditions ranging from congestive heart failure to volume depletion. Careful history and physical examination are paramount and the initial work‐up usually includes serum and urine osmolality and urine sodium concentration.
For euvolemic hyponatremia, the differential diagnosis includes the syndrome of inappropriate adenine dinucleotide (ADH) secretion (SIADH), hypoadrenalism, and beer potomania. Additionally, many authorities also include hypothyroidism.
Although the simultaneous finding of hypothyroidism and hyponatremia can occur in patients as both diseases are widely prevalent in the general population, causation has yet to be convincingly demonstrated.
ADH is released in response to effective volume depletion; consequently when hypothyroidism is encountered in the setting of complete pituitary failure there is often hyponatremia.2, 3 Alternatively, with myxedema, the ability of the kidney to handle a water load and concentrate urine can be impaired.4
However, the observation that thyroid hormone administration did not raise sodium values in newborns with congenital hypothyroidism or in adults supports the absence of causal effect.5, 6And in addition, large studies done in the hospital and outpatient setting showed no differences between the serum sodium values of hypothyroid patients and that of controls.7, 8 In the study of outpatients, among those with hypothyroidism, for every increase of 10 mU/L of thyroid‐stimulating hormone (TSH), there was a drop of only 0.14 mmol/L of Na concentration.8 Thus, the elevation of TSH required for a clinically meaningful drop in sodium to occur was considerable.
Hence in patients with hyponatremia, the hospitalist should look for etiologies other than hypothyroidism and should only consider thyroid hypofunction as a culprit in cases of myxedema, or panhypopituitarism.
Sodium Bicarbonate for Hyperkalemia
Hyperkalemia is one of the most feared electrolyte disorders encountered in hospitalized patients and can lead to dire outcomes.9, 10
Potassium (K+) homeostasis is maintained in the body by 2 complimentary systems: a short‐term system that regulates K+ variation by modifying translocation across the cellular membrane and a long‐term system that adjusts overall K+ balance. The translocation system is regulated primarily by insulin and ‐2 stimulation. Overall K+ balance is mainly controlled by the kidney (90‐95%) although the gastrointestinal (GI) tract can have a more preponderant role in anephric patients.
Hyperkalemia can ensue by either a dysregulation of the translocation system (as in diabetic Ketoacidosis secondary to insulin deficiency) or impairment of K+ elimination.
Acid‐base status was previously thought to have a prominent influence on K+ concentration, based on studies that demonstrated that. However, studies looking at metabolic acidosis revealed that contrary to the effect of mineral acidosis (excess of nonmetabolizable anions)11 where there is an inverse correlation between potassium concentration and pH; organic acidosis (excess of metabolizable anions)11 was not associated with hyperkalemia.1214 However, organic acidosis can be seen simultaneously and induced by a same underlying disease (such as organ ischemia with lactic acidosis or insulin deficiency complicated by ketoacidosis). Also, when changes in pH are induced by respiratory variations or with alkalosis, the impact on serum K+ concentration is less remarkable.15 Hence, it seems that it is the nature of the acid‐base disturbance that impacts K+ concentration more than the change in pH itself.
In the kidney, the main site for regulation of K+ balance is the collecting duct. Factors that affect elimination include urinary sodium delivery, urine flow, and aldosterone.16 In order to adequately eliminate K+ these factors must be optimized in conjunction.
Treatment of hyperkalemia includes the sequential administration of agents that stabilize the cardiac membrane (calcium gluconate), shift the potassium intracellularly (insulin, ‐2 agonists), and remove the potassium (diuretics, sodium polystyrene, or dialysis).
The use of sodium bicarbonate for treatment of hyperkalemia has been long advocated.17 It was thought to act by translocation of potassium hence could be used to quickly lower K+ concentration. However, this dogma has been challenged recently.
To assess the true impact of sodium bicarbonate on potassium translocation, studies have been conducted on anephric patients with hyperkalemia. Bicarbonate infusion failed to elicit a significant rapid change in serum K+ concentration despite increase in bicarbonate concentration, arguing against a translocation mechanism.1821 After 60 minutes of treatment, neither isotonic nor hypertonic bicarbonate infusion affected Serum K+ levels in end‐stage renal disease (ESRD) patients.19, 20 On the contrary, hypertonic sodium bicarbonate increased the K+ concentration after 180 minutes of treatment,20 and it took a prolonged infusion of 4 hours to see a significant decrease in K+ concentration (0.6 mmol/L); half of which could be accounted for solely by volume administration. Moreover, this reduction was highly variable.
Rather, sodium bicarbonate seems to enhance potassium elimination by increasing sodium delivery to the distal tubule, increasing urinary pH and negative luminal charge and potentiating the action of diuretics.23 In an elegant study on normovolemic patients, the induction of bicarbonaturia practically doubled potassium excretion.23 However, such an effect is heterogeneous and usually takes place over 4 hours to 6 hours.17
At the cellular level, 2 ion exchange pumps cooperate to handle Na/K/H movement across the cellular membrane: an Na+/H+ exchanger (NHE) and the Na+/K+ ATP‐ase pump (Figure 1). The NHE is normally inactive and is only upregulated in cases of severe intracellular acidosis.24 The infusion of sodium bicarbonate to patients with severe metabolic acidosis could possibly decrease the serum potassium concentration by translocation if the NHE was significantly upregulated. However, this treatment can be associated with a drop in the ionized calcium level, a worsening of the intracellular acidosis, and a decreased peripheral oxygen delivery.25 Thus, the benefits should be balanced with the potential adverse effects and, even in cases of severe metabolic acidosis with hyperkalemia, we would advise the clinician to restrictively administer sodium bicarbonate.
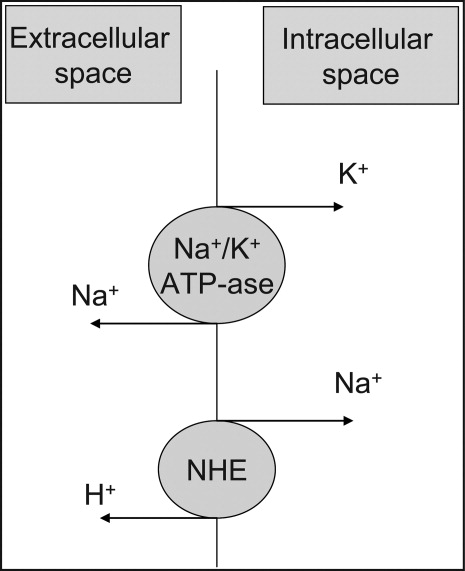
In addition, in ESRD patients, the administration of sodium bicarbonate can be problematic owing to the osmotic and volume burden it carries. It should also be avoided in patients who are volume overload or in those with decreased ability to eliminate potassium.
When treating hyperkalemic patients, hospitalists should use sodium bicarbonate to potentiate urinary elimination of potassium and should consider administering it either with acetazolamide or a loop diuretic, anticipating a lowering effect after a few hours.26 It should be avoided in patients with volume overload and anuria. Immediate translocation of potassium into cells is best achieved by insulin and ‐2 agonists.
5‐Oxoprolinuria: A Newly Recognized Cause of High Anion Gap Metabolic Acidosis
There are several causes of metabolic high anion gap acidosis in hospitalized patients. However, despite careful investigations, the cause of that disorder is not always apparent.27 Recently, 5‐oxoprolinuria (also called pyroglutamic acidosis) has become increasingly recognized as a potential etiology.2832
The metabolism of glutamate (though the ‐Glutamyl cycle) generates glutathione which provides negative feedback to the ‐Glutamyl‐cysteine synthetase enzyme. The depletion of glutathione increases 5‐oxoproline production owing to the loss of that inhibition (Figures 2 and 3). Low glutathione levels can be seen with liver disease,33 chronic alcohol intake,34 acetaminophen use,35 malnutrition,36 renal dysfunction,29 use of vigabatrim (an antiepileptic that received Food and Drug Administration [FDA] approval for use in April 2009)37 and sepsis.38 Most of the reported cases were female and had more than one risk factor.39
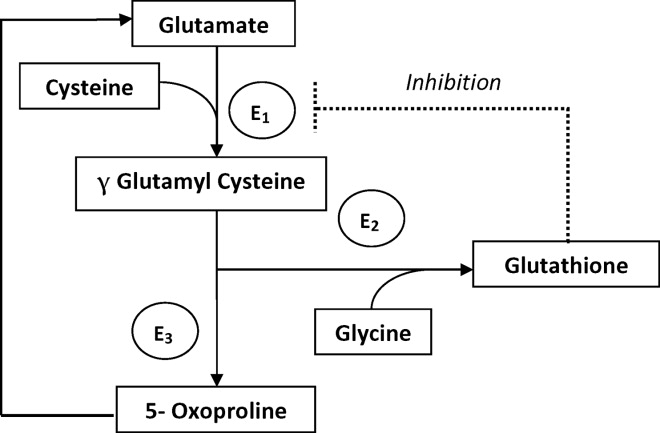
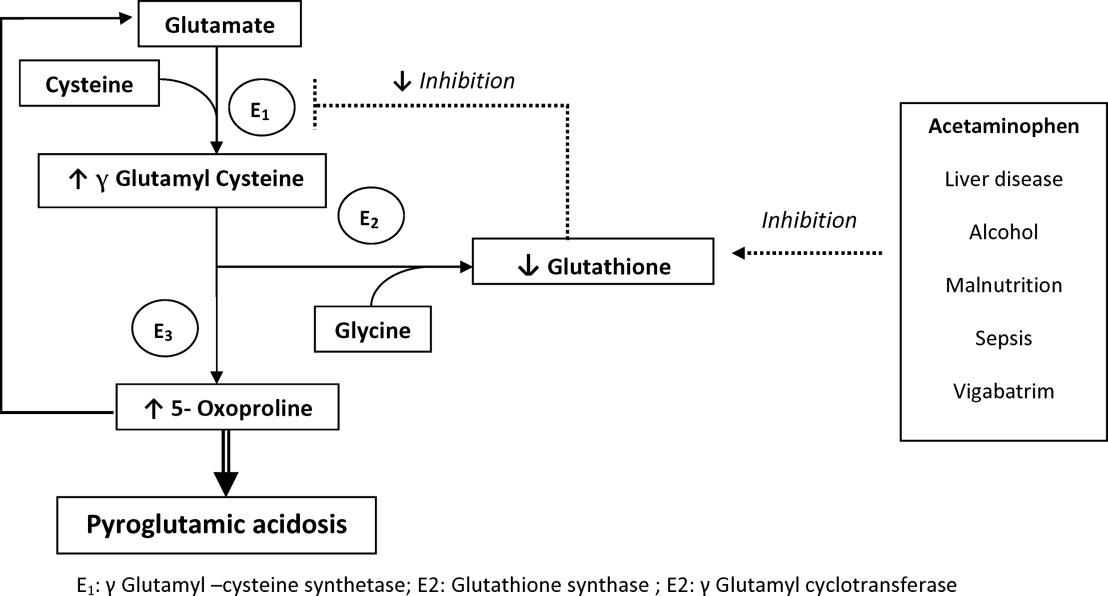
Typically, patients present with high anion gap acidosis (often more than 20)28 with normal acetaminophen levels and all usual tests being negative. A history of chronic acetaminophen use with or without other risk factors can frequently be found. The true independent impact of this type of acidosis on outcomes is difficult to determine as all of the reported cases had many confounding factors.
A urinary organic acid level is diagnostic and will reveal increased levels of pyroglutamic acid. Alternatively, the finding of a positive urinary anion gap (UNa +UK UCl) with a positive urinary osmolar gap (Uosmmeasured‐Uosmcalculated) in the appropriate clinical setting (unexplained high anion gap acidosis with negative workup and presence of risk factors for 5‐oxoproliniuria) can point towards the diagnosis.40
A study of patients with unexplained metabolic acidosis did not find any cases of 5‐oxoprolinuria.41 Although this might suggest that the incidence of this disease is low, very few of those patients were actually taking acetaminophen (therefore had a reduced propensity for developing pyroglutamic acidosis).41 Thus, the actual incidence of 5‐oxoprolinuria is hard to determine.
Once recognized, acetaminophen should be withheld and N‐acetylcysteine (NAC) can be used to replete glutathione levels although there is no convincing evidence for this use.42 It is important for hospitalists to be aware of this disorder as it can pose a diagnostic challenge (negative usual work‐up), is easy to treat by stopping acetaminophen, and can (possibly) negatively affect outcomes.
Furosemide and Patients With Sulfa Allergy
Allergic reactions are a common occurrence with sulfa‐containing antibiotics (SCA) and reports estimates the incidence to be approximately 3% to 5%.43, 44
One misbelief is that patients who are allergic to SCAs should not receive sulfa containing diuretics or other sulfa‐containing medications.45 This leads some physicians to substitute commonly used diuretics (such as furosemide or thiazides) for ethacrynic acid. The use of ethacrynic acid has several challenges: the limited supply of the intravenous form, the discontinuation of the oral form, the increased cost, and the risk of permanent ototoxicity.
The evidence for potential allergic cross‐reactivity among medications containing the sulfa moiety has been primarily derived from Case Reports.4649
The molecular structures of sulfamethoxazole and furosemide are shown below. The allergic antigen is most often the N1 component,45 and sometimes N4 but not the sulfa moiety. Both of the incriminated antigens are not present in the furosemide structure (as well as all other sulfa containing diuretics) (Figures 4 and 5).
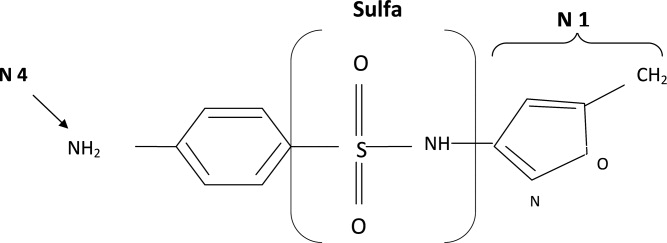
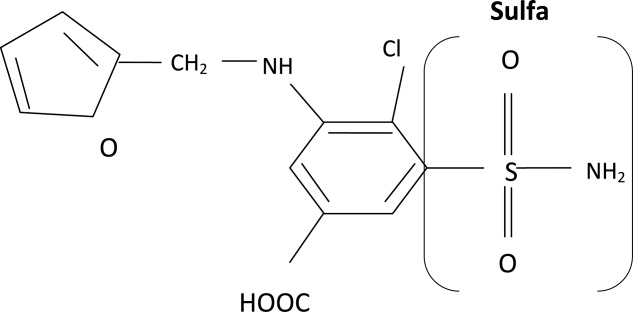
Experimental data showed that serum from patients allergic to SCAs did not bind to diuretics.50 In addition, clinical reports failed to demonstrate cross‐reactivity.5153 In a large clinical trial, Strom et al.53 showed that although there was a higher risk for allergic reaction to sulfa containing medications (SCM) in patients allergic to SCA (compared to those who were not), it was lower among patients with an allergy to sulfa antibiotic than among patients with a history of hypersensitivity to penicillins, suggesting this was due to a predisposition to allergic reactions in general rather than true cross‐reactivity. In another report, patients who were receiving ethacrynic acid for many years were successfully and uneventfully switched to furosemide.54
Taken together, these findings suggest that there is no evidence for withholding sulfa nonantibiotics in patients allergic to sulfa containing antibiotics.
Conclusion
Hypothyroidism, unlike myxedema, is not a cause of hyponatremia (although it can be sometimes seen in conjunction with the latter) and additional investigations should be done to determine its etiology. Sodium bicarbonate is effective for treatment of hyperkalemia by enhancing renal potassium elimination, rather than from shifting potassium into cells. The 5‐oxoprolinuria is a newly recognized cause of high anion‐gap metabolic acidosis and should be considered in patients who have taken acetaminophen. Furosemide (and sulfa containing diuretics) can be used safely in patients with an allergy to SCA.
- ,,.Mortality after hospitalization with mild, moderate, and severe hyponatremia.Am J Med.2009;122(9):857–865.
- ,.Hyponatremia as the presenting manifestation of Sheehan's syndrome in elderly patients.Aging Clin Exp Res.2006;18(6):536–539.
- ,,.Sodium and water disturbances in patients with Sheehan's syndrome.Am J Kidney Dis.2001;38(3):E14.
- ,,,,,.Effect of acute water loading on plasma levels of antidiuretic hormone AVP aldosterone, ANP fractional excretion of sodium and plasma and urine osmolalities in myxedema.Chin Med J (Engl).1990;103(9):704–708.
- ,.Sodium handling in congenitally hypothyroid neonates.Acta Paediatr.2004;93(1):22–24.
- ,,.Prevalence and severity of hyponatremia and hypercreatininemia in short‐term uncomplicated hypothyroidism.J Endocrinol Invest.1999;22(1):35–39.
- ,,,,.Absence of relation between hyponatraemia and hypothyroidism.Lancet.1997;350(9088):1402.
- ,,.The effect of newly diagnosed hypothyroidism on serum sodium concentrations: a retrospective study.Clin Endocrinol (Oxf).2006;64(5):598–599.
- .Disorders of potassium homeostasis. Hypokalemia and hyperkalemia.Crit Care Clin.2002;18(2):273–288,vi.
- ,.Hyperkalemia in hospitalized patients.Int Urol Nephrol.2000;32(2):177–180.
- ,.Treatment of metabolic acidosis.Curr Opin Crit Care.2003;9(4):260–265.
- ,,,.Natural history of lactic acidosis after grand‐mal seizures. A model for the study of an anion‐gap acidosis not associated with hyperkalemia.N Engl J Med.1977;297(15):796–799.
- ,,,,.The plasma potassium concentration in metabolic acidosis: a re‐evaluation.Am J Kidney Dis.1988;11(3):220–224.
- ,,,.Determinants of plasma potassium levels in diabetic ketoacidosis.Medicine (Baltimore).1986;65(3):163–172.
- ,.Changes in plasma potassium concentration during acute acid‐base disturbances.Am J Med.1981;71(3):456–467.
- ,,.New aspects of renal potassium transport.Pflugers Arch.2003;446(3):289–297.
- ,.Correction of hyperkalemia by bicarbonate despite constant blood pH.Kidney Int.1977;12(5):354–360.
- ,,,.Effect of various therapeutic approaches on plasma potassium and major regulating factors in terminal renal failure.Am J Med.1988;85(4):507–512.
- ,.Effect of bicarbonate administration on plasma potassium in dialysis patients: interactions with insulin and albuterol.Am J Kidney Dis.1996;28(4):508–514.
- ,,,,.Effect of hypertonic versus isotonic sodium bicarbonate on plasma potassium concentration in patients with end‐stage renal disease.Miner Electrolyte Metab.1991;17(5):297–302.
- .Combined effect of bicarbonate and insulin with glucose in acute therapy of hyperkalemia in end‐stage renal disease patients.Nephron.1996;72(3):476–482.
- ,,.Effect of prolonged bicarbonate administration on plasma potassium in terminal renal failure.Kidney Int.1992;41(2):369–374.
- ,,, et al.Modulation of the secretion of potassium by accompanying anions in humans.Kidney Int.1991;39(6):1206–1212.
- ,.Controversial issues in the treatment of hyperkalaemia.Nephrol Dial Transplant.2003;18(11):2215–2218.
- ,.Sodium bicarbonate for the treatment of lactic acidosis.Chest.2000;117(1):260–267.
- .Management of severe hyperkalemia.Crit Care Med.2008;36(12):3246–3251.
- ,.Pyroglutamic acid and high anion gap: looking through the keyhole?Crit Care Med.2000;28(6):2140–2141.
- ,,,,.Increased anion gap metabolic acidosis as a result of 5‐oxoproline (pyroglutamic acid): a role for acetaminophen.Clin J Am Soc Nephrol.2006;1(3):441–447.
- ,,,.Pyroglutamic acidemia: a cause of high anion gap metabolic acidosis.Crit Care Med.2000;28(6):1803–1807.
- ,,,,,.Acetaminophen‐induced anion gap metabolic acidosis and 5‐oxoprolinuria (pyroglutamic aciduria) acquired in hospital.Am J Kidney Dis.2005;46(1):143–146.
- ,.Anion gap acidosis associated with acetaminophen.Ann Intern Med.2000;133(9):752–753.
- ,,.Pyroglutamic acidosis in a renal transplant patient.Nephrol Dial Transplant.2005;20(12):2836–2838.
- ,,.Hepatic glutathione content in patients with alcoholic and non alcoholic liver diseases.Life Sci.1988;43(12):991–998.
- ,,,,,.Decreased hepatic glutathione in chronic alcoholic patients.J Hepatol.1986;3(1):1–6.
- ,.Intracellular signaling mechanisms of acetaminophen‐induced liver cell death.Toxicol Sci.2006;89(1):31–41.
- ,,.Urinary excretion of 5‐L‐oxoproline (pyroglutamic acid) is increased during recovery from severe childhood malnutrition and responds to supplemental glycine.J Nutr.1996;126(11):2823–2830.
- ,,,.Pyroglutamicaciduria from vigabatrin.Lancet.1989;1(8652):1452–1453.
- ,,, et al.Cysteine metabolism and whole blood glutathione synthesis in septic pediatric patients.Crit Care Med.2001;29(4):870–877.
- ,.Transient 5‐oxoprolinuria and high anion gap metabolic acidosis: clinical and biochemical findings in eleven subjects.Clin Chem.1998;44(7):1497–1503.
- ,,,.Guilty as charged: unmeasured urinary anions in a case of pyroglutamic acidosis.Neth J Med.2008;66(8):351–353.
- ,,.Unexplained metabolic acidosis in critically ill patients: the role of pyroglutamic acid.Intensive Care Med.2004;30(3):502–505.
- ,,.A therapeutic trial with N‐acetylcysteine in subjects with hereditary glutathione synthetase deficiency (5‐oxoprolinuria).J Inherit Metab Dis.1989;12(2):120–130.
- .Diagnosis of allergic reactions to sulfonamides.Allergy.1999;54Suppl 58:28–32.
- .Practical issues in the management of hypersensitivity reactions: sulfonamides.South Med J.2001;94(8):817–824.
- ,,.Should celecoxib be contraindicated in patients who are allergic to sulfonamides? Revisiting the meaning of ‘sulfa’ allergy.Drug Saf.2001;24(4):239–247.
- .Thrombocytopenia due to sulfonamide cross‐sensitivity.Wis Med J.1982;81(6):21–23.
- ,,.Leukocytoclastic vasculitis induced by use of glyburide: a case of possible cross‐reaction of a sulfonamide and a sulfonylurea.Cutis.2003;71(3):235–238.
- ,.Celecoxib‐induced erythema multiforme with glyburide cross‐reactivity.Pharmacotherapy.2002;22(5):637–640.
- ,.Vesiculobullous rash in a patient with systemic lupus erythematosus.Ann Allergy.1993;70(3):196–203.
- ,,, et al.Use of optical biosensor technology to study immunological cross‐reactivity between different sulfonamide drugs.Anal Biochem.2002;300(2):177–184.
- ,,,.Adverse reactions to sulphonamide and sulphonamide‐trimethoprim antimicrobials: clinical syndromes and pathogenesis.Adverse Drug React Toxicol Rev.1996;15(1):9–50.
- ,,.Cross‐reactivity in HIV‐infected patients switched from trimethoprim‐sulfamethoxazole to dapsone.Pharmacotherapy.1998;18(4):831–835.
- ,,, et al.Absence of cross‐reactivity between sulfonamide antibiotics and sulfonamide nonantibiotics.N Engl J Med.2003;349(17):1628–1635.
- ,,.Furosemide challenge in patients with heart failure and adverse reactions to sulfa‐containing diuretics.Ann Intern Med.2003;138(4):358–359.
There are many controversial topics relating to renal disease in hospitalized patients. The aim of this review is to shed light on some important and often debated issues. We will first discuss topics related to electrolytes disorder commonly seen in hospitalized patients (hyponatremia, hyperkalemia, metabolic acidosis) then the use of diuretics in patients with allergy to sulfa containing antibiotics.
Hypothyroidism and Hyponatremia
Hyponatremia is common in hospitalized patients and is associated with worse outcomes.1 It can be seen with a variety of conditions ranging from congestive heart failure to volume depletion. Careful history and physical examination are paramount and the initial work‐up usually includes serum and urine osmolality and urine sodium concentration.
For euvolemic hyponatremia, the differential diagnosis includes the syndrome of inappropriate adenine dinucleotide (ADH) secretion (SIADH), hypoadrenalism, and beer potomania. Additionally, many authorities also include hypothyroidism.
Although the simultaneous finding of hypothyroidism and hyponatremia can occur in patients as both diseases are widely prevalent in the general population, causation has yet to be convincingly demonstrated.
ADH is released in response to effective volume depletion; consequently when hypothyroidism is encountered in the setting of complete pituitary failure there is often hyponatremia.2, 3 Alternatively, with myxedema, the ability of the kidney to handle a water load and concentrate urine can be impaired.4
However, the observation that thyroid hormone administration did not raise sodium values in newborns with congenital hypothyroidism or in adults supports the absence of causal effect.5, 6And in addition, large studies done in the hospital and outpatient setting showed no differences between the serum sodium values of hypothyroid patients and that of controls.7, 8 In the study of outpatients, among those with hypothyroidism, for every increase of 10 mU/L of thyroid‐stimulating hormone (TSH), there was a drop of only 0.14 mmol/L of Na concentration.8 Thus, the elevation of TSH required for a clinically meaningful drop in sodium to occur was considerable.
Hence in patients with hyponatremia, the hospitalist should look for etiologies other than hypothyroidism and should only consider thyroid hypofunction as a culprit in cases of myxedema, or panhypopituitarism.
Sodium Bicarbonate for Hyperkalemia
Hyperkalemia is one of the most feared electrolyte disorders encountered in hospitalized patients and can lead to dire outcomes.9, 10
Potassium (K+) homeostasis is maintained in the body by 2 complimentary systems: a short‐term system that regulates K+ variation by modifying translocation across the cellular membrane and a long‐term system that adjusts overall K+ balance. The translocation system is regulated primarily by insulin and ‐2 stimulation. Overall K+ balance is mainly controlled by the kidney (90‐95%) although the gastrointestinal (GI) tract can have a more preponderant role in anephric patients.
Hyperkalemia can ensue by either a dysregulation of the translocation system (as in diabetic Ketoacidosis secondary to insulin deficiency) or impairment of K+ elimination.
Acid‐base status was previously thought to have a prominent influence on K+ concentration, based on studies that demonstrated that. However, studies looking at metabolic acidosis revealed that contrary to the effect of mineral acidosis (excess of nonmetabolizable anions)11 where there is an inverse correlation between potassium concentration and pH; organic acidosis (excess of metabolizable anions)11 was not associated with hyperkalemia.1214 However, organic acidosis can be seen simultaneously and induced by a same underlying disease (such as organ ischemia with lactic acidosis or insulin deficiency complicated by ketoacidosis). Also, when changes in pH are induced by respiratory variations or with alkalosis, the impact on serum K+ concentration is less remarkable.15 Hence, it seems that it is the nature of the acid‐base disturbance that impacts K+ concentration more than the change in pH itself.
In the kidney, the main site for regulation of K+ balance is the collecting duct. Factors that affect elimination include urinary sodium delivery, urine flow, and aldosterone.16 In order to adequately eliminate K+ these factors must be optimized in conjunction.
Treatment of hyperkalemia includes the sequential administration of agents that stabilize the cardiac membrane (calcium gluconate), shift the potassium intracellularly (insulin, ‐2 agonists), and remove the potassium (diuretics, sodium polystyrene, or dialysis).
The use of sodium bicarbonate for treatment of hyperkalemia has been long advocated.17 It was thought to act by translocation of potassium hence could be used to quickly lower K+ concentration. However, this dogma has been challenged recently.
To assess the true impact of sodium bicarbonate on potassium translocation, studies have been conducted on anephric patients with hyperkalemia. Bicarbonate infusion failed to elicit a significant rapid change in serum K+ concentration despite increase in bicarbonate concentration, arguing against a translocation mechanism.1821 After 60 minutes of treatment, neither isotonic nor hypertonic bicarbonate infusion affected Serum K+ levels in end‐stage renal disease (ESRD) patients.19, 20 On the contrary, hypertonic sodium bicarbonate increased the K+ concentration after 180 minutes of treatment,20 and it took a prolonged infusion of 4 hours to see a significant decrease in K+ concentration (0.6 mmol/L); half of which could be accounted for solely by volume administration. Moreover, this reduction was highly variable.
Rather, sodium bicarbonate seems to enhance potassium elimination by increasing sodium delivery to the distal tubule, increasing urinary pH and negative luminal charge and potentiating the action of diuretics.23 In an elegant study on normovolemic patients, the induction of bicarbonaturia practically doubled potassium excretion.23 However, such an effect is heterogeneous and usually takes place over 4 hours to 6 hours.17
At the cellular level, 2 ion exchange pumps cooperate to handle Na/K/H movement across the cellular membrane: an Na+/H+ exchanger (NHE) and the Na+/K+ ATP‐ase pump (Figure 1). The NHE is normally inactive and is only upregulated in cases of severe intracellular acidosis.24 The infusion of sodium bicarbonate to patients with severe metabolic acidosis could possibly decrease the serum potassium concentration by translocation if the NHE was significantly upregulated. However, this treatment can be associated with a drop in the ionized calcium level, a worsening of the intracellular acidosis, and a decreased peripheral oxygen delivery.25 Thus, the benefits should be balanced with the potential adverse effects and, even in cases of severe metabolic acidosis with hyperkalemia, we would advise the clinician to restrictively administer sodium bicarbonate.

In addition, in ESRD patients, the administration of sodium bicarbonate can be problematic owing to the osmotic and volume burden it carries. It should also be avoided in patients who are volume overload or in those with decreased ability to eliminate potassium.
When treating hyperkalemic patients, hospitalists should use sodium bicarbonate to potentiate urinary elimination of potassium and should consider administering it either with acetazolamide or a loop diuretic, anticipating a lowering effect after a few hours.26 It should be avoided in patients with volume overload and anuria. Immediate translocation of potassium into cells is best achieved by insulin and ‐2 agonists.
5‐Oxoprolinuria: A Newly Recognized Cause of High Anion Gap Metabolic Acidosis
There are several causes of metabolic high anion gap acidosis in hospitalized patients. However, despite careful investigations, the cause of that disorder is not always apparent.27 Recently, 5‐oxoprolinuria (also called pyroglutamic acidosis) has become increasingly recognized as a potential etiology.2832
The metabolism of glutamate (though the ‐Glutamyl cycle) generates glutathione which provides negative feedback to the ‐Glutamyl‐cysteine synthetase enzyme. The depletion of glutathione increases 5‐oxoproline production owing to the loss of that inhibition (Figures 2 and 3). Low glutathione levels can be seen with liver disease,33 chronic alcohol intake,34 acetaminophen use,35 malnutrition,36 renal dysfunction,29 use of vigabatrim (an antiepileptic that received Food and Drug Administration [FDA] approval for use in April 2009)37 and sepsis.38 Most of the reported cases were female and had more than one risk factor.39


Typically, patients present with high anion gap acidosis (often more than 20)28 with normal acetaminophen levels and all usual tests being negative. A history of chronic acetaminophen use with or without other risk factors can frequently be found. The true independent impact of this type of acidosis on outcomes is difficult to determine as all of the reported cases had many confounding factors.
A urinary organic acid level is diagnostic and will reveal increased levels of pyroglutamic acid. Alternatively, the finding of a positive urinary anion gap (UNa +UK UCl) with a positive urinary osmolar gap (Uosmmeasured‐Uosmcalculated) in the appropriate clinical setting (unexplained high anion gap acidosis with negative workup and presence of risk factors for 5‐oxoproliniuria) can point towards the diagnosis.40
A study of patients with unexplained metabolic acidosis did not find any cases of 5‐oxoprolinuria.41 Although this might suggest that the incidence of this disease is low, very few of those patients were actually taking acetaminophen (therefore had a reduced propensity for developing pyroglutamic acidosis).41 Thus, the actual incidence of 5‐oxoprolinuria is hard to determine.
Once recognized, acetaminophen should be withheld and N‐acetylcysteine (NAC) can be used to replete glutathione levels although there is no convincing evidence for this use.42 It is important for hospitalists to be aware of this disorder as it can pose a diagnostic challenge (negative usual work‐up), is easy to treat by stopping acetaminophen, and can (possibly) negatively affect outcomes.
Furosemide and Patients With Sulfa Allergy
Allergic reactions are a common occurrence with sulfa‐containing antibiotics (SCA) and reports estimates the incidence to be approximately 3% to 5%.43, 44
One misbelief is that patients who are allergic to SCAs should not receive sulfa containing diuretics or other sulfa‐containing medications.45 This leads some physicians to substitute commonly used diuretics (such as furosemide or thiazides) for ethacrynic acid. The use of ethacrynic acid has several challenges: the limited supply of the intravenous form, the discontinuation of the oral form, the increased cost, and the risk of permanent ototoxicity.
The evidence for potential allergic cross‐reactivity among medications containing the sulfa moiety has been primarily derived from Case Reports.4649
The molecular structures of sulfamethoxazole and furosemide are shown below. The allergic antigen is most often the N1 component,45 and sometimes N4 but not the sulfa moiety. Both of the incriminated antigens are not present in the furosemide structure (as well as all other sulfa containing diuretics) (Figures 4 and 5).


Experimental data showed that serum from patients allergic to SCAs did not bind to diuretics.50 In addition, clinical reports failed to demonstrate cross‐reactivity.5153 In a large clinical trial, Strom et al.53 showed that although there was a higher risk for allergic reaction to sulfa containing medications (SCM) in patients allergic to SCA (compared to those who were not), it was lower among patients with an allergy to sulfa antibiotic than among patients with a history of hypersensitivity to penicillins, suggesting this was due to a predisposition to allergic reactions in general rather than true cross‐reactivity. In another report, patients who were receiving ethacrynic acid for many years were successfully and uneventfully switched to furosemide.54
Taken together, these findings suggest that there is no evidence for withholding sulfa nonantibiotics in patients allergic to sulfa containing antibiotics.
Conclusion
Hypothyroidism, unlike myxedema, is not a cause of hyponatremia (although it can be sometimes seen in conjunction with the latter) and additional investigations should be done to determine its etiology. Sodium bicarbonate is effective for treatment of hyperkalemia by enhancing renal potassium elimination, rather than from shifting potassium into cells. The 5‐oxoprolinuria is a newly recognized cause of high anion‐gap metabolic acidosis and should be considered in patients who have taken acetaminophen. Furosemide (and sulfa containing diuretics) can be used safely in patients with an allergy to SCA.
There are many controversial topics relating to renal disease in hospitalized patients. The aim of this review is to shed light on some important and often debated issues. We will first discuss topics related to electrolytes disorder commonly seen in hospitalized patients (hyponatremia, hyperkalemia, metabolic acidosis) then the use of diuretics in patients with allergy to sulfa containing antibiotics.
Hypothyroidism and Hyponatremia
Hyponatremia is common in hospitalized patients and is associated with worse outcomes.1 It can be seen with a variety of conditions ranging from congestive heart failure to volume depletion. Careful history and physical examination are paramount and the initial work‐up usually includes serum and urine osmolality and urine sodium concentration.
For euvolemic hyponatremia, the differential diagnosis includes the syndrome of inappropriate adenine dinucleotide (ADH) secretion (SIADH), hypoadrenalism, and beer potomania. Additionally, many authorities also include hypothyroidism.
Although the simultaneous finding of hypothyroidism and hyponatremia can occur in patients as both diseases are widely prevalent in the general population, causation has yet to be convincingly demonstrated.
ADH is released in response to effective volume depletion; consequently when hypothyroidism is encountered in the setting of complete pituitary failure there is often hyponatremia.2, 3 Alternatively, with myxedema, the ability of the kidney to handle a water load and concentrate urine can be impaired.4
However, the observation that thyroid hormone administration did not raise sodium values in newborns with congenital hypothyroidism or in adults supports the absence of causal effect.5, 6And in addition, large studies done in the hospital and outpatient setting showed no differences between the serum sodium values of hypothyroid patients and that of controls.7, 8 In the study of outpatients, among those with hypothyroidism, for every increase of 10 mU/L of thyroid‐stimulating hormone (TSH), there was a drop of only 0.14 mmol/L of Na concentration.8 Thus, the elevation of TSH required for a clinically meaningful drop in sodium to occur was considerable.
Hence in patients with hyponatremia, the hospitalist should look for etiologies other than hypothyroidism and should only consider thyroid hypofunction as a culprit in cases of myxedema, or panhypopituitarism.
Sodium Bicarbonate for Hyperkalemia
Hyperkalemia is one of the most feared electrolyte disorders encountered in hospitalized patients and can lead to dire outcomes.9, 10
Potassium (K+) homeostasis is maintained in the body by 2 complimentary systems: a short‐term system that regulates K+ variation by modifying translocation across the cellular membrane and a long‐term system that adjusts overall K+ balance. The translocation system is regulated primarily by insulin and ‐2 stimulation. Overall K+ balance is mainly controlled by the kidney (90‐95%) although the gastrointestinal (GI) tract can have a more preponderant role in anephric patients.
Hyperkalemia can ensue by either a dysregulation of the translocation system (as in diabetic Ketoacidosis secondary to insulin deficiency) or impairment of K+ elimination.
Acid‐base status was previously thought to have a prominent influence on K+ concentration, based on studies that demonstrated that. However, studies looking at metabolic acidosis revealed that contrary to the effect of mineral acidosis (excess of nonmetabolizable anions)11 where there is an inverse correlation between potassium concentration and pH; organic acidosis (excess of metabolizable anions)11 was not associated with hyperkalemia.1214 However, organic acidosis can be seen simultaneously and induced by a same underlying disease (such as organ ischemia with lactic acidosis or insulin deficiency complicated by ketoacidosis). Also, when changes in pH are induced by respiratory variations or with alkalosis, the impact on serum K+ concentration is less remarkable.15 Hence, it seems that it is the nature of the acid‐base disturbance that impacts K+ concentration more than the change in pH itself.
In the kidney, the main site for regulation of K+ balance is the collecting duct. Factors that affect elimination include urinary sodium delivery, urine flow, and aldosterone.16 In order to adequately eliminate K+ these factors must be optimized in conjunction.
Treatment of hyperkalemia includes the sequential administration of agents that stabilize the cardiac membrane (calcium gluconate), shift the potassium intracellularly (insulin, ‐2 agonists), and remove the potassium (diuretics, sodium polystyrene, or dialysis).
The use of sodium bicarbonate for treatment of hyperkalemia has been long advocated.17 It was thought to act by translocation of potassium hence could be used to quickly lower K+ concentration. However, this dogma has been challenged recently.
To assess the true impact of sodium bicarbonate on potassium translocation, studies have been conducted on anephric patients with hyperkalemia. Bicarbonate infusion failed to elicit a significant rapid change in serum K+ concentration despite increase in bicarbonate concentration, arguing against a translocation mechanism.1821 After 60 minutes of treatment, neither isotonic nor hypertonic bicarbonate infusion affected Serum K+ levels in end‐stage renal disease (ESRD) patients.19, 20 On the contrary, hypertonic sodium bicarbonate increased the K+ concentration after 180 minutes of treatment,20 and it took a prolonged infusion of 4 hours to see a significant decrease in K+ concentration (0.6 mmol/L); half of which could be accounted for solely by volume administration. Moreover, this reduction was highly variable.
Rather, sodium bicarbonate seems to enhance potassium elimination by increasing sodium delivery to the distal tubule, increasing urinary pH and negative luminal charge and potentiating the action of diuretics.23 In an elegant study on normovolemic patients, the induction of bicarbonaturia practically doubled potassium excretion.23 However, such an effect is heterogeneous and usually takes place over 4 hours to 6 hours.17
At the cellular level, 2 ion exchange pumps cooperate to handle Na/K/H movement across the cellular membrane: an Na+/H+ exchanger (NHE) and the Na+/K+ ATP‐ase pump (Figure 1). The NHE is normally inactive and is only upregulated in cases of severe intracellular acidosis.24 The infusion of sodium bicarbonate to patients with severe metabolic acidosis could possibly decrease the serum potassium concentration by translocation if the NHE was significantly upregulated. However, this treatment can be associated with a drop in the ionized calcium level, a worsening of the intracellular acidosis, and a decreased peripheral oxygen delivery.25 Thus, the benefits should be balanced with the potential adverse effects and, even in cases of severe metabolic acidosis with hyperkalemia, we would advise the clinician to restrictively administer sodium bicarbonate.

In addition, in ESRD patients, the administration of sodium bicarbonate can be problematic owing to the osmotic and volume burden it carries. It should also be avoided in patients who are volume overload or in those with decreased ability to eliminate potassium.
When treating hyperkalemic patients, hospitalists should use sodium bicarbonate to potentiate urinary elimination of potassium and should consider administering it either with acetazolamide or a loop diuretic, anticipating a lowering effect after a few hours.26 It should be avoided in patients with volume overload and anuria. Immediate translocation of potassium into cells is best achieved by insulin and ‐2 agonists.
5‐Oxoprolinuria: A Newly Recognized Cause of High Anion Gap Metabolic Acidosis
There are several causes of metabolic high anion gap acidosis in hospitalized patients. However, despite careful investigations, the cause of that disorder is not always apparent.27 Recently, 5‐oxoprolinuria (also called pyroglutamic acidosis) has become increasingly recognized as a potential etiology.2832
The metabolism of glutamate (though the ‐Glutamyl cycle) generates glutathione which provides negative feedback to the ‐Glutamyl‐cysteine synthetase enzyme. The depletion of glutathione increases 5‐oxoproline production owing to the loss of that inhibition (Figures 2 and 3). Low glutathione levels can be seen with liver disease,33 chronic alcohol intake,34 acetaminophen use,35 malnutrition,36 renal dysfunction,29 use of vigabatrim (an antiepileptic that received Food and Drug Administration [FDA] approval for use in April 2009)37 and sepsis.38 Most of the reported cases were female and had more than one risk factor.39


Typically, patients present with high anion gap acidosis (often more than 20)28 with normal acetaminophen levels and all usual tests being negative. A history of chronic acetaminophen use with or without other risk factors can frequently be found. The true independent impact of this type of acidosis on outcomes is difficult to determine as all of the reported cases had many confounding factors.
A urinary organic acid level is diagnostic and will reveal increased levels of pyroglutamic acid. Alternatively, the finding of a positive urinary anion gap (UNa +UK UCl) with a positive urinary osmolar gap (Uosmmeasured‐Uosmcalculated) in the appropriate clinical setting (unexplained high anion gap acidosis with negative workup and presence of risk factors for 5‐oxoproliniuria) can point towards the diagnosis.40
A study of patients with unexplained metabolic acidosis did not find any cases of 5‐oxoprolinuria.41 Although this might suggest that the incidence of this disease is low, very few of those patients were actually taking acetaminophen (therefore had a reduced propensity for developing pyroglutamic acidosis).41 Thus, the actual incidence of 5‐oxoprolinuria is hard to determine.
Once recognized, acetaminophen should be withheld and N‐acetylcysteine (NAC) can be used to replete glutathione levels although there is no convincing evidence for this use.42 It is important for hospitalists to be aware of this disorder as it can pose a diagnostic challenge (negative usual work‐up), is easy to treat by stopping acetaminophen, and can (possibly) negatively affect outcomes.
Furosemide and Patients With Sulfa Allergy
Allergic reactions are a common occurrence with sulfa‐containing antibiotics (SCA) and reports estimates the incidence to be approximately 3% to 5%.43, 44
One misbelief is that patients who are allergic to SCAs should not receive sulfa containing diuretics or other sulfa‐containing medications.45 This leads some physicians to substitute commonly used diuretics (such as furosemide or thiazides) for ethacrynic acid. The use of ethacrynic acid has several challenges: the limited supply of the intravenous form, the discontinuation of the oral form, the increased cost, and the risk of permanent ototoxicity.
The evidence for potential allergic cross‐reactivity among medications containing the sulfa moiety has been primarily derived from Case Reports.4649
The molecular structures of sulfamethoxazole and furosemide are shown below. The allergic antigen is most often the N1 component,45 and sometimes N4 but not the sulfa moiety. Both of the incriminated antigens are not present in the furosemide structure (as well as all other sulfa containing diuretics) (Figures 4 and 5).


Experimental data showed that serum from patients allergic to SCAs did not bind to diuretics.50 In addition, clinical reports failed to demonstrate cross‐reactivity.5153 In a large clinical trial, Strom et al.53 showed that although there was a higher risk for allergic reaction to sulfa containing medications (SCM) in patients allergic to SCA (compared to those who were not), it was lower among patients with an allergy to sulfa antibiotic than among patients with a history of hypersensitivity to penicillins, suggesting this was due to a predisposition to allergic reactions in general rather than true cross‐reactivity. In another report, patients who were receiving ethacrynic acid for many years were successfully and uneventfully switched to furosemide.54
Taken together, these findings suggest that there is no evidence for withholding sulfa nonantibiotics in patients allergic to sulfa containing antibiotics.
Conclusion
Hypothyroidism, unlike myxedema, is not a cause of hyponatremia (although it can be sometimes seen in conjunction with the latter) and additional investigations should be done to determine its etiology. Sodium bicarbonate is effective for treatment of hyperkalemia by enhancing renal potassium elimination, rather than from shifting potassium into cells. The 5‐oxoprolinuria is a newly recognized cause of high anion‐gap metabolic acidosis and should be considered in patients who have taken acetaminophen. Furosemide (and sulfa containing diuretics) can be used safely in patients with an allergy to SCA.
- ,,.Mortality after hospitalization with mild, moderate, and severe hyponatremia.Am J Med.2009;122(9):857–865.
- ,.Hyponatremia as the presenting manifestation of Sheehan's syndrome in elderly patients.Aging Clin Exp Res.2006;18(6):536–539.
- ,,.Sodium and water disturbances in patients with Sheehan's syndrome.Am J Kidney Dis.2001;38(3):E14.
- ,,,,,.Effect of acute water loading on plasma levels of antidiuretic hormone AVP aldosterone, ANP fractional excretion of sodium and plasma and urine osmolalities in myxedema.Chin Med J (Engl).1990;103(9):704–708.
- ,.Sodium handling in congenitally hypothyroid neonates.Acta Paediatr.2004;93(1):22–24.
- ,,.Prevalence and severity of hyponatremia and hypercreatininemia in short‐term uncomplicated hypothyroidism.J Endocrinol Invest.1999;22(1):35–39.
- ,,,,.Absence of relation between hyponatraemia and hypothyroidism.Lancet.1997;350(9088):1402.
- ,,.The effect of newly diagnosed hypothyroidism on serum sodium concentrations: a retrospective study.Clin Endocrinol (Oxf).2006;64(5):598–599.
- .Disorders of potassium homeostasis. Hypokalemia and hyperkalemia.Crit Care Clin.2002;18(2):273–288,vi.
- ,.Hyperkalemia in hospitalized patients.Int Urol Nephrol.2000;32(2):177–180.
- ,.Treatment of metabolic acidosis.Curr Opin Crit Care.2003;9(4):260–265.
- ,,,.Natural history of lactic acidosis after grand‐mal seizures. A model for the study of an anion‐gap acidosis not associated with hyperkalemia.N Engl J Med.1977;297(15):796–799.
- ,,,,.The plasma potassium concentration in metabolic acidosis: a re‐evaluation.Am J Kidney Dis.1988;11(3):220–224.
- ,,,.Determinants of plasma potassium levels in diabetic ketoacidosis.Medicine (Baltimore).1986;65(3):163–172.
- ,.Changes in plasma potassium concentration during acute acid‐base disturbances.Am J Med.1981;71(3):456–467.
- ,,.New aspects of renal potassium transport.Pflugers Arch.2003;446(3):289–297.
- ,.Correction of hyperkalemia by bicarbonate despite constant blood pH.Kidney Int.1977;12(5):354–360.
- ,,,.Effect of various therapeutic approaches on plasma potassium and major regulating factors in terminal renal failure.Am J Med.1988;85(4):507–512.
- ,.Effect of bicarbonate administration on plasma potassium in dialysis patients: interactions with insulin and albuterol.Am J Kidney Dis.1996;28(4):508–514.
- ,,,,.Effect of hypertonic versus isotonic sodium bicarbonate on plasma potassium concentration in patients with end‐stage renal disease.Miner Electrolyte Metab.1991;17(5):297–302.
- .Combined effect of bicarbonate and insulin with glucose in acute therapy of hyperkalemia in end‐stage renal disease patients.Nephron.1996;72(3):476–482.
- ,,.Effect of prolonged bicarbonate administration on plasma potassium in terminal renal failure.Kidney Int.1992;41(2):369–374.
- ,,, et al.Modulation of the secretion of potassium by accompanying anions in humans.Kidney Int.1991;39(6):1206–1212.
- ,.Controversial issues in the treatment of hyperkalaemia.Nephrol Dial Transplant.2003;18(11):2215–2218.
- ,.Sodium bicarbonate for the treatment of lactic acidosis.Chest.2000;117(1):260–267.
- .Management of severe hyperkalemia.Crit Care Med.2008;36(12):3246–3251.
- ,.Pyroglutamic acid and high anion gap: looking through the keyhole?Crit Care Med.2000;28(6):2140–2141.
- ,,,,.Increased anion gap metabolic acidosis as a result of 5‐oxoproline (pyroglutamic acid): a role for acetaminophen.Clin J Am Soc Nephrol.2006;1(3):441–447.
- ,,,.Pyroglutamic acidemia: a cause of high anion gap metabolic acidosis.Crit Care Med.2000;28(6):1803–1807.
- ,,,,,.Acetaminophen‐induced anion gap metabolic acidosis and 5‐oxoprolinuria (pyroglutamic aciduria) acquired in hospital.Am J Kidney Dis.2005;46(1):143–146.
- ,.Anion gap acidosis associated with acetaminophen.Ann Intern Med.2000;133(9):752–753.
- ,,.Pyroglutamic acidosis in a renal transplant patient.Nephrol Dial Transplant.2005;20(12):2836–2838.
- ,,.Hepatic glutathione content in patients with alcoholic and non alcoholic liver diseases.Life Sci.1988;43(12):991–998.
- ,,,,,.Decreased hepatic glutathione in chronic alcoholic patients.J Hepatol.1986;3(1):1–6.
- ,.Intracellular signaling mechanisms of acetaminophen‐induced liver cell death.Toxicol Sci.2006;89(1):31–41.
- ,,.Urinary excretion of 5‐L‐oxoproline (pyroglutamic acid) is increased during recovery from severe childhood malnutrition and responds to supplemental glycine.J Nutr.1996;126(11):2823–2830.
- ,,,.Pyroglutamicaciduria from vigabatrin.Lancet.1989;1(8652):1452–1453.
- ,,, et al.Cysteine metabolism and whole blood glutathione synthesis in septic pediatric patients.Crit Care Med.2001;29(4):870–877.
- ,.Transient 5‐oxoprolinuria and high anion gap metabolic acidosis: clinical and biochemical findings in eleven subjects.Clin Chem.1998;44(7):1497–1503.
- ,,,.Guilty as charged: unmeasured urinary anions in a case of pyroglutamic acidosis.Neth J Med.2008;66(8):351–353.
- ,,.Unexplained metabolic acidosis in critically ill patients: the role of pyroglutamic acid.Intensive Care Med.2004;30(3):502–505.
- ,,.A therapeutic trial with N‐acetylcysteine in subjects with hereditary glutathione synthetase deficiency (5‐oxoprolinuria).J Inherit Metab Dis.1989;12(2):120–130.
- .Diagnosis of allergic reactions to sulfonamides.Allergy.1999;54Suppl 58:28–32.
- .Practical issues in the management of hypersensitivity reactions: sulfonamides.South Med J.2001;94(8):817–824.
- ,,.Should celecoxib be contraindicated in patients who are allergic to sulfonamides? Revisiting the meaning of ‘sulfa’ allergy.Drug Saf.2001;24(4):239–247.
- .Thrombocytopenia due to sulfonamide cross‐sensitivity.Wis Med J.1982;81(6):21–23.
- ,,.Leukocytoclastic vasculitis induced by use of glyburide: a case of possible cross‐reaction of a sulfonamide and a sulfonylurea.Cutis.2003;71(3):235–238.
- ,.Celecoxib‐induced erythema multiforme with glyburide cross‐reactivity.Pharmacotherapy.2002;22(5):637–640.
- ,.Vesiculobullous rash in a patient with systemic lupus erythematosus.Ann Allergy.1993;70(3):196–203.
- ,,, et al.Use of optical biosensor technology to study immunological cross‐reactivity between different sulfonamide drugs.Anal Biochem.2002;300(2):177–184.
- ,,,.Adverse reactions to sulphonamide and sulphonamide‐trimethoprim antimicrobials: clinical syndromes and pathogenesis.Adverse Drug React Toxicol Rev.1996;15(1):9–50.
- ,,.Cross‐reactivity in HIV‐infected patients switched from trimethoprim‐sulfamethoxazole to dapsone.Pharmacotherapy.1998;18(4):831–835.
- ,,, et al.Absence of cross‐reactivity between sulfonamide antibiotics and sulfonamide nonantibiotics.N Engl J Med.2003;349(17):1628–1635.
- ,,.Furosemide challenge in patients with heart failure and adverse reactions to sulfa‐containing diuretics.Ann Intern Med.2003;138(4):358–359.
- ,,.Mortality after hospitalization with mild, moderate, and severe hyponatremia.Am J Med.2009;122(9):857–865.
- ,.Hyponatremia as the presenting manifestation of Sheehan's syndrome in elderly patients.Aging Clin Exp Res.2006;18(6):536–539.
- ,,.Sodium and water disturbances in patients with Sheehan's syndrome.Am J Kidney Dis.2001;38(3):E14.
- ,,,,,.Effect of acute water loading on plasma levels of antidiuretic hormone AVP aldosterone, ANP fractional excretion of sodium and plasma and urine osmolalities in myxedema.Chin Med J (Engl).1990;103(9):704–708.
- ,.Sodium handling in congenitally hypothyroid neonates.Acta Paediatr.2004;93(1):22–24.
- ,,.Prevalence and severity of hyponatremia and hypercreatininemia in short‐term uncomplicated hypothyroidism.J Endocrinol Invest.1999;22(1):35–39.
- ,,,,.Absence of relation between hyponatraemia and hypothyroidism.Lancet.1997;350(9088):1402.
- ,,.The effect of newly diagnosed hypothyroidism on serum sodium concentrations: a retrospective study.Clin Endocrinol (Oxf).2006;64(5):598–599.
- .Disorders of potassium homeostasis. Hypokalemia and hyperkalemia.Crit Care Clin.2002;18(2):273–288,vi.
- ,.Hyperkalemia in hospitalized patients.Int Urol Nephrol.2000;32(2):177–180.
- ,.Treatment of metabolic acidosis.Curr Opin Crit Care.2003;9(4):260–265.
- ,,,.Natural history of lactic acidosis after grand‐mal seizures. A model for the study of an anion‐gap acidosis not associated with hyperkalemia.N Engl J Med.1977;297(15):796–799.
- ,,,,.The plasma potassium concentration in metabolic acidosis: a re‐evaluation.Am J Kidney Dis.1988;11(3):220–224.
- ,,,.Determinants of plasma potassium levels in diabetic ketoacidosis.Medicine (Baltimore).1986;65(3):163–172.
- ,.Changes in plasma potassium concentration during acute acid‐base disturbances.Am J Med.1981;71(3):456–467.
- ,,.New aspects of renal potassium transport.Pflugers Arch.2003;446(3):289–297.
- ,.Correction of hyperkalemia by bicarbonate despite constant blood pH.Kidney Int.1977;12(5):354–360.
- ,,,.Effect of various therapeutic approaches on plasma potassium and major regulating factors in terminal renal failure.Am J Med.1988;85(4):507–512.
- ,.Effect of bicarbonate administration on plasma potassium in dialysis patients: interactions with insulin and albuterol.Am J Kidney Dis.1996;28(4):508–514.
- ,,,,.Effect of hypertonic versus isotonic sodium bicarbonate on plasma potassium concentration in patients with end‐stage renal disease.Miner Electrolyte Metab.1991;17(5):297–302.
- .Combined effect of bicarbonate and insulin with glucose in acute therapy of hyperkalemia in end‐stage renal disease patients.Nephron.1996;72(3):476–482.
- ,,.Effect of prolonged bicarbonate administration on plasma potassium in terminal renal failure.Kidney Int.1992;41(2):369–374.
- ,,, et al.Modulation of the secretion of potassium by accompanying anions in humans.Kidney Int.1991;39(6):1206–1212.
- ,.Controversial issues in the treatment of hyperkalaemia.Nephrol Dial Transplant.2003;18(11):2215–2218.
- ,.Sodium bicarbonate for the treatment of lactic acidosis.Chest.2000;117(1):260–267.
- .Management of severe hyperkalemia.Crit Care Med.2008;36(12):3246–3251.
- ,.Pyroglutamic acid and high anion gap: looking through the keyhole?Crit Care Med.2000;28(6):2140–2141.
- ,,,,.Increased anion gap metabolic acidosis as a result of 5‐oxoproline (pyroglutamic acid): a role for acetaminophen.Clin J Am Soc Nephrol.2006;1(3):441–447.
- ,,,.Pyroglutamic acidemia: a cause of high anion gap metabolic acidosis.Crit Care Med.2000;28(6):1803–1807.
- ,,,,,.Acetaminophen‐induced anion gap metabolic acidosis and 5‐oxoprolinuria (pyroglutamic aciduria) acquired in hospital.Am J Kidney Dis.2005;46(1):143–146.
- ,.Anion gap acidosis associated with acetaminophen.Ann Intern Med.2000;133(9):752–753.
- ,,.Pyroglutamic acidosis in a renal transplant patient.Nephrol Dial Transplant.2005;20(12):2836–2838.
- ,,.Hepatic glutathione content in patients with alcoholic and non alcoholic liver diseases.Life Sci.1988;43(12):991–998.
- ,,,,,.Decreased hepatic glutathione in chronic alcoholic patients.J Hepatol.1986;3(1):1–6.
- ,.Intracellular signaling mechanisms of acetaminophen‐induced liver cell death.Toxicol Sci.2006;89(1):31–41.
- ,,.Urinary excretion of 5‐L‐oxoproline (pyroglutamic acid) is increased during recovery from severe childhood malnutrition and responds to supplemental glycine.J Nutr.1996;126(11):2823–2830.
- ,,,.Pyroglutamicaciduria from vigabatrin.Lancet.1989;1(8652):1452–1453.
- ,,, et al.Cysteine metabolism and whole blood glutathione synthesis in septic pediatric patients.Crit Care Med.2001;29(4):870–877.
- ,.Transient 5‐oxoprolinuria and high anion gap metabolic acidosis: clinical and biochemical findings in eleven subjects.Clin Chem.1998;44(7):1497–1503.
- ,,,.Guilty as charged: unmeasured urinary anions in a case of pyroglutamic acidosis.Neth J Med.2008;66(8):351–353.
- ,,.Unexplained metabolic acidosis in critically ill patients: the role of pyroglutamic acid.Intensive Care Med.2004;30(3):502–505.
- ,,.A therapeutic trial with N‐acetylcysteine in subjects with hereditary glutathione synthetase deficiency (5‐oxoprolinuria).J Inherit Metab Dis.1989;12(2):120–130.
- .Diagnosis of allergic reactions to sulfonamides.Allergy.1999;54Suppl 58:28–32.
- .Practical issues in the management of hypersensitivity reactions: sulfonamides.South Med J.2001;94(8):817–824.
- ,,.Should celecoxib be contraindicated in patients who are allergic to sulfonamides? Revisiting the meaning of ‘sulfa’ allergy.Drug Saf.2001;24(4):239–247.
- .Thrombocytopenia due to sulfonamide cross‐sensitivity.Wis Med J.1982;81(6):21–23.
- ,,.Leukocytoclastic vasculitis induced by use of glyburide: a case of possible cross‐reaction of a sulfonamide and a sulfonylurea.Cutis.2003;71(3):235–238.
- ,.Celecoxib‐induced erythema multiforme with glyburide cross‐reactivity.Pharmacotherapy.2002;22(5):637–640.
- ,.Vesiculobullous rash in a patient with systemic lupus erythematosus.Ann Allergy.1993;70(3):196–203.
- ,,, et al.Use of optical biosensor technology to study immunological cross‐reactivity between different sulfonamide drugs.Anal Biochem.2002;300(2):177–184.
- ,,,.Adverse reactions to sulphonamide and sulphonamide‐trimethoprim antimicrobials: clinical syndromes and pathogenesis.Adverse Drug React Toxicol Rev.1996;15(1):9–50.
- ,,.Cross‐reactivity in HIV‐infected patients switched from trimethoprim‐sulfamethoxazole to dapsone.Pharmacotherapy.1998;18(4):831–835.
- ,,, et al.Absence of cross‐reactivity between sulfonamide antibiotics and sulfonamide nonantibiotics.N Engl J Med.2003;349(17):1628–1635.
- ,,.Furosemide challenge in patients with heart failure and adverse reactions to sulfa‐containing diuretics.Ann Intern Med.2003;138(4):358–359.
Report Supports Telemedicine Use in ICU
A new report that outlines the potential care delivery improvements and cost savings of telemedicine might also be a road map to increased efficiency for hospitalists, according to one of the study’s authors and a former SHM president.
Telemedicine "simplifies their life," says Mitchell Adams, AB, MBA, executive director of the Massachusetts Technology Collaborative (MTC) in Boston, which coauthored "Critical Care, Critical Choices: The Case for Tele-ICUs in Intensive Care" with the New England Healthcare Institute (NEHI) in Cambridge. "It means you have you less complications, you have less work. You have an intensivist looking over your shoulder, making sure you’re doing it right."
As the population ages and hospitalists become more entrenched in their institutions, they could end up spending more time on ICU cases. The use of telemedicine—where an intensivist at a remote “command center” oversees the delivery of care—could foster higher-quality and more efficient care, which would subsequently allow HM practitioners to focus on the rest of the census, according to Mary Jo Gorman, MD, MBA, FHM, former SHM president and CEO of St. Louis-based Advanced ICU Care, which provides intensivists to community hospitals using telemedicine.
The study, based on a demonstration project at the University of Massachusetts Memorial Medical Center and two associated community hospitals, reported a 20% drop in ICU mortality at the academic medical center (P=0.01). When adjusted for the severity of ICU illnesses, one community hospital reported a 36% drop in mortality (P=0.83); the other reported a 142% increase (P<0.001). All three centers also reported a reduction in length of stay (LOS) of at least 12 hours.
Dr. Gorman and Adams agree that it will take more evidence-based studies showing the efficacy of telemedicine before the practice becomes widespread, a phenomenon Adams attributes to the "inherent inertia and viscosity in the system to maintain the status quo."
"Everybody might know the right answer," Dr. Gorman adds. "It's still going to take a long time. That’s just the pace at which we move in healthcare."
A new report that outlines the potential care delivery improvements and cost savings of telemedicine might also be a road map to increased efficiency for hospitalists, according to one of the study’s authors and a former SHM president.
Telemedicine "simplifies their life," says Mitchell Adams, AB, MBA, executive director of the Massachusetts Technology Collaborative (MTC) in Boston, which coauthored "Critical Care, Critical Choices: The Case for Tele-ICUs in Intensive Care" with the New England Healthcare Institute (NEHI) in Cambridge. "It means you have you less complications, you have less work. You have an intensivist looking over your shoulder, making sure you’re doing it right."
As the population ages and hospitalists become more entrenched in their institutions, they could end up spending more time on ICU cases. The use of telemedicine—where an intensivist at a remote “command center” oversees the delivery of care—could foster higher-quality and more efficient care, which would subsequently allow HM practitioners to focus on the rest of the census, according to Mary Jo Gorman, MD, MBA, FHM, former SHM president and CEO of St. Louis-based Advanced ICU Care, which provides intensivists to community hospitals using telemedicine.
The study, based on a demonstration project at the University of Massachusetts Memorial Medical Center and two associated community hospitals, reported a 20% drop in ICU mortality at the academic medical center (P=0.01). When adjusted for the severity of ICU illnesses, one community hospital reported a 36% drop in mortality (P=0.83); the other reported a 142% increase (P<0.001). All three centers also reported a reduction in length of stay (LOS) of at least 12 hours.
Dr. Gorman and Adams agree that it will take more evidence-based studies showing the efficacy of telemedicine before the practice becomes widespread, a phenomenon Adams attributes to the "inherent inertia and viscosity in the system to maintain the status quo."
"Everybody might know the right answer," Dr. Gorman adds. "It's still going to take a long time. That’s just the pace at which we move in healthcare."
A new report that outlines the potential care delivery improvements and cost savings of telemedicine might also be a road map to increased efficiency for hospitalists, according to one of the study’s authors and a former SHM president.
Telemedicine "simplifies their life," says Mitchell Adams, AB, MBA, executive director of the Massachusetts Technology Collaborative (MTC) in Boston, which coauthored "Critical Care, Critical Choices: The Case for Tele-ICUs in Intensive Care" with the New England Healthcare Institute (NEHI) in Cambridge. "It means you have you less complications, you have less work. You have an intensivist looking over your shoulder, making sure you’re doing it right."
As the population ages and hospitalists become more entrenched in their institutions, they could end up spending more time on ICU cases. The use of telemedicine—where an intensivist at a remote “command center” oversees the delivery of care—could foster higher-quality and more efficient care, which would subsequently allow HM practitioners to focus on the rest of the census, according to Mary Jo Gorman, MD, MBA, FHM, former SHM president and CEO of St. Louis-based Advanced ICU Care, which provides intensivists to community hospitals using telemedicine.
The study, based on a demonstration project at the University of Massachusetts Memorial Medical Center and two associated community hospitals, reported a 20% drop in ICU mortality at the academic medical center (P=0.01). When adjusted for the severity of ICU illnesses, one community hospital reported a 36% drop in mortality (P=0.83); the other reported a 142% increase (P<0.001). All three centers also reported a reduction in length of stay (LOS) of at least 12 hours.
Dr. Gorman and Adams agree that it will take more evidence-based studies showing the efficacy of telemedicine before the practice becomes widespread, a phenomenon Adams attributes to the "inherent inertia and viscosity in the system to maintain the status quo."
"Everybody might know the right answer," Dr. Gorman adds. "It's still going to take a long time. That’s just the pace at which we move in healthcare."
CMS Announces Guidelines for $500M Care Transitions Program
An eagerly awaited federal grant program that will allocate $500 million for improving care transitions and reducing rehospitalizations will soon release a solicitation and application instructions, possibly by the end of December.
The Community-Based Care Transitions Program (CCTP), funded for five years starting in January through the Affordable Care Act, is open to hospitals with higher-than-average rehospitalization rates and their community-based partners, and to coalitions of community-based organizations that include hospitals, CMS research analyst Juliana Tiongson explained during an all-day conference Dec. 3 in Baltimore.
Hospitalists and HM groups likely would not qualify directly for these grants, but they can start identifying and partnering with interested hospital leaders and relevant community-based providers. CCTP is designed to encourage communities to work together in ongoing learning collaboratives, drawing on evidence-based models for improving care transitions, Tiongson said. Examples of evidence-based strategies, such as Eric Coleman’s Community Care Transitions Program, Mary Naylor’s Transitional Care Model, and SHM’s Project BOOST, were described during the teleconference.
“We will require applicants to do their homework, including a thorough root cause analysis” of current care-transitions processes and their limitations, Tiongson said. Applications that include multiple stakeholders across the healthcare continuum, such as consumer representation, links to accountable-care organizations (ACOs), and medical homes, and those that are based in the communities they propose to serve, will be favored in the rolling application process. Applicants should demonstrate organizational readiness in terms of staffing, training, and preparation for better managing such transitions as hospital discharges.
An eagerly awaited federal grant program that will allocate $500 million for improving care transitions and reducing rehospitalizations will soon release a solicitation and application instructions, possibly by the end of December.
The Community-Based Care Transitions Program (CCTP), funded for five years starting in January through the Affordable Care Act, is open to hospitals with higher-than-average rehospitalization rates and their community-based partners, and to coalitions of community-based organizations that include hospitals, CMS research analyst Juliana Tiongson explained during an all-day conference Dec. 3 in Baltimore.
Hospitalists and HM groups likely would not qualify directly for these grants, but they can start identifying and partnering with interested hospital leaders and relevant community-based providers. CCTP is designed to encourage communities to work together in ongoing learning collaboratives, drawing on evidence-based models for improving care transitions, Tiongson said. Examples of evidence-based strategies, such as Eric Coleman’s Community Care Transitions Program, Mary Naylor’s Transitional Care Model, and SHM’s Project BOOST, were described during the teleconference.
“We will require applicants to do their homework, including a thorough root cause analysis” of current care-transitions processes and their limitations, Tiongson said. Applications that include multiple stakeholders across the healthcare continuum, such as consumer representation, links to accountable-care organizations (ACOs), and medical homes, and those that are based in the communities they propose to serve, will be favored in the rolling application process. Applicants should demonstrate organizational readiness in terms of staffing, training, and preparation for better managing such transitions as hospital discharges.
An eagerly awaited federal grant program that will allocate $500 million for improving care transitions and reducing rehospitalizations will soon release a solicitation and application instructions, possibly by the end of December.
The Community-Based Care Transitions Program (CCTP), funded for five years starting in January through the Affordable Care Act, is open to hospitals with higher-than-average rehospitalization rates and their community-based partners, and to coalitions of community-based organizations that include hospitals, CMS research analyst Juliana Tiongson explained during an all-day conference Dec. 3 in Baltimore.
Hospitalists and HM groups likely would not qualify directly for these grants, but they can start identifying and partnering with interested hospital leaders and relevant community-based providers. CCTP is designed to encourage communities to work together in ongoing learning collaboratives, drawing on evidence-based models for improving care transitions, Tiongson said. Examples of evidence-based strategies, such as Eric Coleman’s Community Care Transitions Program, Mary Naylor’s Transitional Care Model, and SHM’s Project BOOST, were described during the teleconference.
“We will require applicants to do their homework, including a thorough root cause analysis” of current care-transitions processes and their limitations, Tiongson said. Applications that include multiple stakeholders across the healthcare continuum, such as consumer representation, links to accountable-care organizations (ACOs), and medical homes, and those that are based in the communities they propose to serve, will be favored in the rolling application process. Applicants should demonstrate organizational readiness in terms of staffing, training, and preparation for better managing such transitions as hospital discharges.
Patient Dissatisfaction
The United States spends more money per capita on healthcare than any other industrialized nation,1 yet patients are the least satisfied with their care.2 Patient satisfaction is associated in both cross‐sectional3 and longitudinal studies4 with improved physical and mental health outcomes. Conversely, dissatisfaction with care hampers future medical interactions, prevents sharing of information, and impairs the building of trust.5 The increasing recognition that a patient's experience of care affects patient outcomes has furthered efforts to evaluate satisfaction with care.6, 7
However, patient satisfaction is challenging to define and understand. Even the definition of satisfaction is ambiguous, for to satisfy can mean both to make happy and the lesser, to be adequate. To dissatisfy is to displease or disappoint, but dissatisfaction is not the opposite of satisfaction: qualitative studies give little if any indication that patients evaluate satisfaction on a continuum ranging from dissatisfied at one end to very satisfied at the other.8 Instead, it appears that satisfaction and dissatisfaction are different constructs, such that patients may simultaneously be both satisfied and dissatisfied.9, 10 Patients often express overall satisfaction with a service or encounter while also reporting specific criticisms about its shortcomings.11, 12 Alternatively, consumers may be generally satisfied unless something unpleasant or improper happens.13 Thus, dissatisfaction and satisfaction may require different methods of measurement.
The most common model for measuring patient satisfaction is a quantitative survey of patients' experiences in specific predetermined domains. Of 54 hospital satisfaction surveys in common use, only 11 included patient input in their development,14 casting doubt on the relevance of these attributes to patients' priorities of care. Since it is well recognized that patient expectations influence satisfaction,8, 13, 15 it is important to identify patient expectations and priorities up front. However, these have not been clearly established. Furthermore, focusing purely on satisfaction with particular domains of care may miss the separate but illuminating construct of patient dissatisfaction.
In this study we therefore aim to understand patient dissatisfaction with hospitalization more fully as a means of elucidating implicit expectations for hospital care. Using qualitative techniques, we analyzed a large volume of patient responses to a single open‐ended study question to identify determinants and patterns of patient dissatisfaction.
Methods
Study Design
We conducted a qualitative analysis of telephone survey data obtained from adults recently discharged after an acute care hospitalization. Survey participants were asked five questions, including: If there was one thing we could have done to improve your experience in the hospital, what would it have been? Answers to this open‐ended question were included in this study.
Setting and Participants
The hospital is a 944‐bed, urban academic medical center. Patients or patient representatives were routinely surveyed in a telephone interview conducted by trained hospital staff 1‐5 days after hospital discharge. Calls were attempted to 90% of adult discharged patients, and approximately 50% of them were reached. For this study, we included patients who were age 18 or older, spoke English, and were discharged to home from a medical, surgical, gynecology‐oncology, neurology, neurosurgery, or intensive care unit. Of those patients, we randomly selected 10% of those surveyed between July 1, 2007 and June 30, 2008 for inclusion.
Primary Data Analysis
Qualitative data analysis was used to classify patient suggestions. The study team included internal medicine physicians (J.P.M., L.I.H.), a medical student (A.V.L.), and a recent college graduate (C.P.B.). Codes were generated using a mixed inductive and deductive approach by reading and rereading the primary data.16 A set of 100 interview responses were first read individually by three investigators (J.P.M., A.V.L., C.P.B.), after which investigators met to discuss themes and ideas. A preliminary list of coding categories was then generated. Each investigator then assigned these coding categories to additional survey responses in sets of 100. Subsequent meetings were held to refine codes using the constant comparative method.16 Disagreements were resolved by negotiated consensus. The full study group met periodically to review the code structure for logic and breadth. Once thematic saturation was achieved, the entire dataset was recoded by two investigators using the final coding structure. The final coding structure contained 42 unique codes organized into six broader themes. We used descriptive statistics to characterize the coding category results. The score for intercoder reliability was 0.91.
This study was approved by the Yale Human Investigation Committee, which granted a waiver of informed consent.
Results
A total of 976 surveys was randomly selected from 9,764 postdischarge phone interviews completed between July 1, 2007 and June 30, 2008. A total of 56.3% of patients was female. Nearly half the patients were discharged from medical units (Table 1). Of the 976 patients, 439 (45.0%) gave at least one suggestion for improvement, yielding a total of 579 suggestions. Patients also offered numerous positive comments about their care, but these comments were not included in the analysis.
| No. | % of Total Surveyed | |
|---|---|---|
| Total surveys | 976 | |
| Male | 427 | 43.7 |
| Female | 549 | 56.3 |
| Discharge Unit | ||
| Medical | 434 | 44.5 |
| Surgical | 303 | 31.0 |
| Gynecology/Oncology | 103 | 10.6 |
| ICU/CCU/Step‐down | 71 | 7.3 |
| Neurology/Neurosurgery | 65 | 6.6 |
| No suggestions for improvement | 537 | 55.0 |
| At least one suggestion for improvement | 439 | 45.0 |
Through qualitative analysis, we assigned suggestions for improvement to six major categories of dissatisfaction: 1) ineptitude, 2) disrespect, 3) prolonged waits, 4) ineffective communication, 5) lack of environmental control, and 6) substandard amenities. We considered the inverse of these problems to represent six implicit expectations of good hospital care: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities (Table 2). The number of patient suggestions related to each domain is detailed in Table 3.
| Domain of Dissatisfaction | Implicit Expectations | Example |
|---|---|---|
| Ineptitude | Safety | The only thing was that when I was getting ready to get discharged, one of Dr. H*'s associates came in and said, We have to readmit you for a further procedure. I said, Well, that's strange because Dr. H* put in a stent yesterday, and I'm supposed to leave today. Well, he checked, and he had the wrong guy. I'm glad I said something or else they probably would have hauled me off. |
| Disrespect | Treatment with respect and dignity | Transport was rude due to me being a heavy person. They were saying they didn't want to move me and snickering. |
| Prolonged waits | Prompt and efficient care | I called for someone because I had to use the bathroom really bad, but I had those things stuck to my legs and needed help walking to the bathroom but no one came. Well, I had to go so bad that I had a panic attack. Then all these people came rushing in to help. I felt so embarrassed. |
| Ineffective communication | Successful exchange of information | There were a few days that [were] a little confusing to me. I didn't know if I was going to have surgery or go home. The communication wasn't that great. |
| Lack of environmental control | Environmental autonomy and control | I was put in a room with a man who had many issues. He was loud and yelling all night. It was a very disturbing experience. |
| Substandard amenities | High‐quality amenities | In that ICU they should put a TV on the ceiling for when you're lying flat on your back looking at the ceiling tiles for 4 days. |
| Domain of Dissatisfaction | No. (N = 579 suggestionsa) | % of Total Surveyed | % Within Domain |
|---|---|---|---|
| |||
| Perceived ineptitude | 75 | 7.7 | 100 |
| Adverse events | 18 | 1.8 | 24.0 |
| Cleanliness | 36 | 3.7 | 48.0 |
| Perceived lack of knowledge/skill | 12 | 1.2 | 16.0 |
| Rushed out | 9 | 0.9 | 12.0 |
| Disrespect | 59 | 6.0 | 100 |
| Unprofessional staff behavior | 55 | 5.6 | 93.2 |
| Lack of privacy/confidentiality | 4 | 0.4 | 6.8 |
| Prolonged waits | 154 | 15.8 | 100 |
| Response to call bell | |||
| Bathing/toileting/distress | 24 | 2.5 | 15.6 |
| General | 41 | 4.2 | 26.6 |
| Wait for physician | 12 | 1.2 | 7.8 |
| Wait for admission bed | 29 | 3.0 | 18.8 |
| Wait for transport | 16 | 1.6 | 10.4 |
| Wait for food | 7 | 0.7 | 4.6 |
| Wait for medication | 11 | 1.1 | 7.1 |
| Wait for diagnostic test/procedures | 6 | 0.6 | 3.9 |
| Wait for discharge | 8 | 0.8 | 5.2 |
| Ineffective communication | 72 | 7.4 | 100 |
| Communication with patients | 33 | 3.4 | 45.8 |
| Communication with family | 3 | 0.3 | 4.2 |
| Translation | 2 | 0.2 | 2.8 |
| Communication between providers | 13 | 1.3 | 18.1 |
| Coordination of care (inpatient) | 11 | 1.1 | 15.3 |
| Medication reconciliation | 5 | 0.5 | 6.9 |
| Continuity inpatient to outpatient | 5 | 0.5 | 6.9 |
| Lack of environmental control | 152 | 15.6 | 100 |
| Physical environment | |||
| Roommates | 38 | 3.9 | 25.0 |
| Noise | 24 | 2.5 | 15.8 |
| Temperature | 12 | 1.2 | 7.9 |
| Smell | 1 | 0.1 | 0.7 |
| Interruption by staff | 15 | 1.5 | 9.9 |
| Lighting | 2 | 0.2 | 1.3 |
| Chaos/hectic | 4 | 0.4 | 2.6 |
| Shorter Stay | 8 | 0.8 | 5.3 |
| General | 3 | 0.3 | 2.0 |
| Facilities | |||
| Pain control | 10 | 1.0 | 6.6 |
| Painful procedures | 17 | 1.7 | 11.2 |
| Facilities | |||
| Bathrooms | 7 | 0.7 | 4.6 |
| Maintenance response | 5 | 0.5 | 3.3 |
| Traffic/parking | 6 | 0.6 | 3.9 |
| Substandard amenities | 67 | 6.9 | 100 |
| Food quality | 26 | 2.7 | 38.8 |
| Food variety | 5 | 0.5 | 7.5 |
| Food service | 16 | 1.6 | 23.9 |
| TV | 8 | 0.8 | 11.9 |
| Beds | 8 | 0.8 | 11.9 |
| Gowns | 4 | 0.4 | 6.0 |
Ineptitude
A total of 7.7% of interviewed patients reported experiencing a situation that made them feel unsafe. Dissatisfaction with safety included adverse events or near misses, uncleanliness, and a perceived lack of knowledge or skill. The implicit expectation that emerged from this domain was that the hospital would be safe, and that medical staff would be knowledgeable and skillful.
Adverse events or near misses were experienced in several areas, including diet, medication administration, patient identification, and equipment. Patients were particularly troubled when they or a family member caught the error:
There was one male nurse in training, C*, who was about to give my mother an injection. I asked what he was doing because she was about to go into surgery. He said he thought she was going home. He looked at the chart again and it turns out he was holding her roommate's chart. I don't know what would have happened if I wasn't there.
Dissatisfaction with the cleanliness of the hospital environment was also frequently expressed as a safety concern:
The rooms are dirtyThe floors are dirty. They don't sweep unless you ask them to. It took three different people to come and clean the bathroom right. I have to come back for surgery and I'm scared to death with all that bacteria and uncleanliness.
In this category, patients also described care by not too knowledgeable trainees or other staff as a safety hazard.
Disrespect
A total of 6.0% of surveyed patients suggested improvements that reflected disrespectful treatment, including poor work ethic, lack of warmth, rudeness, and a lack of attention to privacy and confidentiality. This type of dissatisfaction suggested an implicit expectation for treatment with respect and dignity that was clearly distinct from the expectation of technical quality:
[Hospital name] has always been like [this] since I started going there in 1982. They're very good technically but their bedside manner kind of sucks. You survive but you don't walk away with a warm fuzzy feeling.
Underprivileged patients were particularly sensitive to the need for respect:
I feel like the doctor that saw me that last night there was trying to get me out of there as fast as possible, saying not in so many words that it was because I don't have any insurance. I just feel like they treated me like an animal.
Violations of privacy and confidentiality were not only perceived as disrespectful, but also as a direct impediment to high‐quality care:
In the ER, I didn't like that I had no privacy especially talking with the doctor because I was in the hallway. I didn't have any privacy therefore I wasn't completely truthful with the doctor because everyone could hear.
Prolonged Waits
A total of 15.8% of patients noted dissatisfaction with wait times in the hospital. Waits for admission, transport, or discharge were frequently cited as anxiety‐provoking or frustrating:
The ER wait is too long. I was there from 8:00 AM to 2:00 AM the next day. I was there the whole day and night. When someone is in pain, they just want to be taken care of, not waiting around.
Waits related to receiving patient care, for example the inability to access nurses or physicians, more often caused feelings of fear and abandonment:
Every patient is different, I understand, but when you're there at night it can be a little scary. I was not only scared but in pain. The nurse tried to get a hold of the doctor that was on call, but the doctor took hours to respond. That was very scary.
It was also distressing to patients to watch roommates experience a delay in help for urgent needs:
The lady next to me was an elderly woman with a brace on her neck, and she couldn't speak very well. She had diarrhea at night and she would ask for a bedpan. The nurses would take forever bringing it to her. I just think when there are elderly people they should be more attentive to them because they tend to not be as vocal, you know?
Together, these comments represented an implicit expectation for prompt and efficient care.
Ineffective Communication
Communication during hospitalization was a source of dissatisfaction in 7.4% of surveyed patients. Communication failures occurred in several areas. Most common was the ineffective transfer of medical information to patients:
For days I thought I was having surgery on Friday. So all that day I ate and drank nothing and got prepped for surgery. Finally later that night I was told I was going to have it on Saturday. Saturday comes and still nothing. I never saw a surgeon or talked to anyone. Then later after that I was told I'm not having the surgery. That was the most frustrating thing.
Patients were also dissatisfied with their ability to communicate with their doctors:
I was sent home on a Friday and was sent right back on Friday night because my blood count was low and I ended up needing a blood transfusion. I tried to tell them this but they didn't listen. They need to listen to the patients.
Failed communication between care providers in the hospital was a third inadequacy noted by patients:
The only problem I had was all the different doctors coming in and out. There's so many that it confuses the patient, and a lot of them would contradict each other. One doctor said I could go home and another doctor said, No, you need to stay.
Finally, patients were dissatisfied when there was ineffective communication between inpatient and outpatient providers.
They said the VNA [Visiting Nurse Association] is supposed to come. The nurse hasn't come to see me and she hasn't called. My daughter and I have been waiting.
Thus, patients had an implicit expectation for effective communication between all parties in the hospital and were dissatisfied when any type of communication was inadequate.
Lack of Environmental Control
A total of 15.4% of surveyed patients reported dissatisfaction with the inability to control the physical environment. The inability to control noise levels, roommate behavior, temperature, smells, pain, lighting, staff interruptions, food service, smoking, and even humidity were all anxiety‐producing for different patients. The feeling of being imposed upon by an uncomfortable physical environment also extended to hospital facilities such as inaccessible bathrooms, traffic, and parking. Dissatisfaction with rooming arrangements was common:
I was in a triple room and one of my roommates had at least six visitors in the room at a time every day including two infant twins. Someone really should have said something about that. It became very disturbing, and I even left a day early because of that.
An expectation for quiet, especially during the night, was also repeatedly expressed:
The night shift could have been more considerate of people trying to rest. There was a lot of noise and bangs. I know people have to laugh and have fun but it could have been a little more quiet.
Related was the inability to control interruptions by staff members:
It's hard enough to get sleep, but then those blood suckers come in the middle of the night.
This category of dissatisfaction reflected an implicit expectation for autonomy and control over the environment so that it was conducive to rest and healing.
Substandard Amenities
A total of 6.9% of surveyed patients suggested improvements to amenities such as food, bedding, gowns, and television. Moving beyond the expectation of having peaceful surroundings, these comments reflected an expectation of a well‐appointed hospital environment with high‐quality amenities. A typical example was this comment about the food and service:
You never get what you order from the kitchen. Your tray either has something missing from it or it's the wrong tray or not the right diet. It's very frustrating and hard to get the orders the way you want.
Discussion
We analyzed 439 patient suggestions for improving hospital care and found that dissatisfaction resulted from six categories of negative experiences: 1) ineptitude, 2) disrespect, 3) prolonged wait times, 4) ineffective communication, 5) lack of environmental control, and 6) substandard amenities. These domains represented a corresponding set of implicit patient expectations for: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities. Each of these categories suggests avenues by which both the assessment and provision of hospital care can be made more patient‐centered.
The most widely used patient satisfaction survey in use in the United States today is the Hospital Consumer Assessment of Healthcare Providers & Systems (HCAHPS), which includes eight domains: communication with doctors, communication with nurses, responsiveness of hospital staff, pain management, communication about medicines, discharge information, cleanliness of the hospital environment, and quietness of the hospital environment.17 The dissatisfaction domains found in this study closely overlap the HCAHPS satisfaction domains, but with a few key differences.
First, dissatisfaction with ineptitude in our study encompassed concerns over adverse events and near misses, in addition to the cleanliness of the environment. Other research has shown that dissatisfaction with hospitalization can be predicted by the number of reported problems18 and the perception of receiving incorrect treatment.19 While elaborate methods have been devised to assess and compare the hospital quality and safety, patient satisfaction surveys including the HCAHPS survey often fail to ask patients directly about their perceptions of safety. In fact, this study and others20, 21 show that patients are able to recognize adverse events during hospitalization. Patient report may be a useful adjunct to other methods of adverse event case finding and outcomes reporting.
Second, while HCAHPS and others identify warmth, courtesy, concern, and respect as dimensions of patient‐centered care,14, 17, 22, 23 the ability of quantitative satisfaction surveys to capture the experience of disrespectful treatment may be limited, especially during hospitalization. Most respondents who commented on feeling disrespected identified only a single encounter, which can be masked by otherwise satisfying interactions with numerous care providers. Directly asking patients whether any experience during hospitalization caused them to feel disrespected, and allowing room for explanation, might more efficiently identify problem areas. This is particularly important because even one episode of disrespectful treatment, particularly when perceived to be racially motivated, increases the likelihood of not following a doctor's advice or putting off care.24
Third, HCAHPS emphasizes two aspects of communication: that between patients and doctors, and that between patients and nurses. Our patients confirmed that these are important, but they also noted a third dimension of communication contributing to dissatisfaction: provider‐provider communication. Communication and coordination failures among providers are key contributors to adverse events or near misses,2528 but their influence on patient satisfaction has not been widely assessed. Furthermore, patient input is rarely utilized to identify poor interprovider communication. Our study suggests that, just as patients can identify adverse events, they are also able to recognize poor provider‐provider communication.
Patients' reports of dissatisfying events also highlight areas in which small changes in hospital practice might greatly improve the patient experience. For instance, concerns over environment, food, sleep, hygiene, and pain appeared to be representative of a broader dissatisfaction with loss of autonomy and control. Hospitalized patients are often obliged to room with strangers, are subject to noise and interruptions, and cede control of their medication management at a time when they are feeling particularly vulnerable. The importance of this lack of autonomy to patients suggests a variety of small interventions that could improve satisfaction, such as individual control of noise and temperature, a visible commitment to a quiet hospital environment, and minimized interruptions and sleep disturbance.2932 Single‐occupancy hospital rooms have been associated with lower rates of nosocomial infection, medication errors, and patient stress, as well as increased privacy, rest, visitor involvement, and doctor‐patient communication.33, 34 The most sophisticated intervention, acuity‐adaptable private hospital rooms, allows hospitals to maintain patients in the same private hospital room during an entire admission, regardless of changes to level of acuity.35
In‐depth analysis of suggestions for improvement, as gathered by telephone surveys of recently discharged patients, was a particularly well‐suited approach to identifying explicit expectations for care that were violated by dissatisfying incidents. When allowed to express dissatisfaction in terms of suggestions for improvement, patients talked freely about specific dissatisfying experiences. Using telephone interviews allowed a large volume of patient responses to be included, unlike smaller focus groups. Our study was oral and did not rely on the literacy level of patients. Additionally, the open‐ended nature of questioning avoided some of the usual pitfalls of satisfaction surveys. We did not rely on predetermined satisfaction categories or presume the inherent value of particular attributes of care. Nonetheless, our study does have important limitations.
Patient perceptions were not compared with chart data or clinician report. Caregivers were allowed to participate in lieu of patients, which may have reduced identification of some dissatisfying events. Likewise, patients discharged to nursing homes or who were not English or Spanish speaking were excluded and may have had different dissatisfying experiences. Interviews were brief and dissatisfying events were not explored in detail. Although nearly half of respondents reported dissatisfying events, some patients may have been reluctant to criticize their care directly to a hospital representative. Finally, patients generally confined their comments to one or two dissatisfying events, even though there may have been others. We therefore cannot draw any conclusions about the relative frequency of dissatisfying events by domain.
Conclusions
All hospitalized patients bring expectations for their hospital experience. While specific expectations vary between patients, expectations for: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities were found in this study to encompass core expectations for hospitalization. It may be useful to ensure that postdischarge surveys explicitly address these expectations. Efforts to address and manage these core expectations of hospital care may help to reduce patient dissatisfaction with hospitalization and improve the delivery and quality of hospital care.
- ,.Multinational Comparisons of Health Systems Data, 2006.Washington, DC:The Commonwealth Fund;2007.
- ,,,,,.Toward higher‐performance health systems: Adults' health care experiences in seven countries, 2007.Health Aff.2007;26:w717–734.
- ,,, et al.Patient satisfaction and its relationship with clinical quality and inpatient mortality in acute myocardial infarction.Circ Cardiovasc Qual Outcomes.2010;3:188–195.
- ,.A longitudinal analysis of patient satisfaction and subsequent quality of life in Hong Kong Chinese breast and nasopharyngeal cancer patients.Med Care.2009;47:875–881.
- .Understanding dissatisfied users: developing a framework for comprehending criticisms of health care work.J Adv Nurs.1999;30:723–731.
- ,.Patient satisfaction: a review of issues and concepts.Soc Sci Med.1997;45:1829–1843.
- Institute of Medicine.Crossing the Quality Chasm: A New Health System for the 21st Century.Washington, DC:National Academy Press;2001.
- .Patient satisfaction: a valid concept?Soc Sci Med.1994;38:509–516.
- .Exploring the meaning of ‘dissatisfaction’ with health care: the importance of ‘personal identity threat’.Sociol Health Illn.1999;21:95–123.
- ,.Pathways, pyramids and icebergs? Mapping the links between dissatisfaction and complaints.Sociol Health Illn.1998;20:825–847.
- ,.Convergence and divergence: assessing criteria of consumer satisfaction across general practice, dental and hospital care settings.Soc Sci Med.1991;33:707–716.
- ,,,,,.National survey of hospital patients [see comment].BMJ.1994;309:1542–1546.
- ,,, et al.The measurement of satisfaction with healthcare: implications for practice from a systematic review of the literature.Health Technol Assess.2002;6:1–244.
- ,,,.Review of the literature on survey instruments used to collect data on hospital patients' perceptions of care.Health Serv Res.2005;40:1996–2017.
- ,,.Satisfying solutions? A review of some unresolved issues in the measurement of patient satisfaction.J Adv Nurs.1995;22:316–322.
- ,.The Discovery of Grounded Theory: Strategies for Qualitative Research.Chicago, IL:Aldine;1967.
- Hospital Consumer Assessment of Healthcare Providers 25:25–36.
- ,,,.Patient experiences in relation to respondent and health service delivery characteristics: a survey of 26,938 patients attending 62 hospitals throughout Norway.Scand J Public Health.2007;35:70–77.
- ,,, et al.What can hospitalized patients tell us about adverse events? Learning from patient‐reported incidents.J Gen Intern Med.2005;20:830–836.
- .A hospitalization from hell: a patient's perspective on quality.Ann Intern Med.2003;138:33–39.
- ,,,.Through the Patient's Eyes: Understanding and Promoting Patient‐Centered Care.San Francisco, CA:Jossey‐Bass;1993.
- ,,,,.What do consumers want to know about the quality of care in hospitals?Health Serv Res.2005;40:2018–2036.
- ,.R‐e‐s‐p‐e‐c‐t: patient reports of disrespect in the health care setting and its impact on care.J Fam Pract.2004;53:721–730.
- ,,,,.Perceived information needs and communication difficulties of inpatient physicians and nurses.J Am Med Inform Assoc.2002;9(6 suppl 1):S64–S69.
- ,,.The human factor: the critical importance of effective teamwork and communication in providing safe care.Qual Saf Health Care.2004;13:i85–i90.
- ,,,,.Consequences of inadequate sign‐out for patient care.Arch Intern Med.2008;168:1755–1760.
- ,,,,,.Dropping the baton: a qualitative analysis of failures during the transition from emergency department to inpatient care.Ann Emerg Med.2009;53:701–710 e704.
- ,.Interactive relationships between hospital patients' noise‐induced stress and other stress with sleep.Heart Lung.2001;30:237–243.
- .The influence of subjective reactions to noise on health effects of the noise.Environ Int.1996;22:93–104.
- .A novel PACU design for noise reduction.J Perianesth Nurs.2008;23:226–229.
- ,,,.The Planetree Model Hospital Project: an example of the patient as partner. (Pacific Presbyterian Medical Center, San Francisco).Hosp Health Serv Admin.1990;35:591–601.
- ,,.Physician‐patient communication in single‐bedded versus four‐bedded hospital rooms.Patient Educ Couns.2008;73:215–219.
- ,,.Advantages and disadvantages of single‐versus multiple‐occupancy rooms in acute care environments: a review and analysis of the literature.Environ Behav.2005;37:760–786.
- ,.Impacting patient outcomes through design: acuity adaptable care/universal room design.Crit Care Nurs Q.2006;29:326–341.
The United States spends more money per capita on healthcare than any other industrialized nation,1 yet patients are the least satisfied with their care.2 Patient satisfaction is associated in both cross‐sectional3 and longitudinal studies4 with improved physical and mental health outcomes. Conversely, dissatisfaction with care hampers future medical interactions, prevents sharing of information, and impairs the building of trust.5 The increasing recognition that a patient's experience of care affects patient outcomes has furthered efforts to evaluate satisfaction with care.6, 7
However, patient satisfaction is challenging to define and understand. Even the definition of satisfaction is ambiguous, for to satisfy can mean both to make happy and the lesser, to be adequate. To dissatisfy is to displease or disappoint, but dissatisfaction is not the opposite of satisfaction: qualitative studies give little if any indication that patients evaluate satisfaction on a continuum ranging from dissatisfied at one end to very satisfied at the other.8 Instead, it appears that satisfaction and dissatisfaction are different constructs, such that patients may simultaneously be both satisfied and dissatisfied.9, 10 Patients often express overall satisfaction with a service or encounter while also reporting specific criticisms about its shortcomings.11, 12 Alternatively, consumers may be generally satisfied unless something unpleasant or improper happens.13 Thus, dissatisfaction and satisfaction may require different methods of measurement.
The most common model for measuring patient satisfaction is a quantitative survey of patients' experiences in specific predetermined domains. Of 54 hospital satisfaction surveys in common use, only 11 included patient input in their development,14 casting doubt on the relevance of these attributes to patients' priorities of care. Since it is well recognized that patient expectations influence satisfaction,8, 13, 15 it is important to identify patient expectations and priorities up front. However, these have not been clearly established. Furthermore, focusing purely on satisfaction with particular domains of care may miss the separate but illuminating construct of patient dissatisfaction.
In this study we therefore aim to understand patient dissatisfaction with hospitalization more fully as a means of elucidating implicit expectations for hospital care. Using qualitative techniques, we analyzed a large volume of patient responses to a single open‐ended study question to identify determinants and patterns of patient dissatisfaction.
Methods
Study Design
We conducted a qualitative analysis of telephone survey data obtained from adults recently discharged after an acute care hospitalization. Survey participants were asked five questions, including: If there was one thing we could have done to improve your experience in the hospital, what would it have been? Answers to this open‐ended question were included in this study.
Setting and Participants
The hospital is a 944‐bed, urban academic medical center. Patients or patient representatives were routinely surveyed in a telephone interview conducted by trained hospital staff 1‐5 days after hospital discharge. Calls were attempted to 90% of adult discharged patients, and approximately 50% of them were reached. For this study, we included patients who were age 18 or older, spoke English, and were discharged to home from a medical, surgical, gynecology‐oncology, neurology, neurosurgery, or intensive care unit. Of those patients, we randomly selected 10% of those surveyed between July 1, 2007 and June 30, 2008 for inclusion.
Primary Data Analysis
Qualitative data analysis was used to classify patient suggestions. The study team included internal medicine physicians (J.P.M., L.I.H.), a medical student (A.V.L.), and a recent college graduate (C.P.B.). Codes were generated using a mixed inductive and deductive approach by reading and rereading the primary data.16 A set of 100 interview responses were first read individually by three investigators (J.P.M., A.V.L., C.P.B.), after which investigators met to discuss themes and ideas. A preliminary list of coding categories was then generated. Each investigator then assigned these coding categories to additional survey responses in sets of 100. Subsequent meetings were held to refine codes using the constant comparative method.16 Disagreements were resolved by negotiated consensus. The full study group met periodically to review the code structure for logic and breadth. Once thematic saturation was achieved, the entire dataset was recoded by two investigators using the final coding structure. The final coding structure contained 42 unique codes organized into six broader themes. We used descriptive statistics to characterize the coding category results. The score for intercoder reliability was 0.91.
This study was approved by the Yale Human Investigation Committee, which granted a waiver of informed consent.
Results
A total of 976 surveys was randomly selected from 9,764 postdischarge phone interviews completed between July 1, 2007 and June 30, 2008. A total of 56.3% of patients was female. Nearly half the patients were discharged from medical units (Table 1). Of the 976 patients, 439 (45.0%) gave at least one suggestion for improvement, yielding a total of 579 suggestions. Patients also offered numerous positive comments about their care, but these comments were not included in the analysis.
| No. | % of Total Surveyed | |
|---|---|---|
| Total surveys | 976 | |
| Male | 427 | 43.7 |
| Female | 549 | 56.3 |
| Discharge Unit | ||
| Medical | 434 | 44.5 |
| Surgical | 303 | 31.0 |
| Gynecology/Oncology | 103 | 10.6 |
| ICU/CCU/Step‐down | 71 | 7.3 |
| Neurology/Neurosurgery | 65 | 6.6 |
| No suggestions for improvement | 537 | 55.0 |
| At least one suggestion for improvement | 439 | 45.0 |
Through qualitative analysis, we assigned suggestions for improvement to six major categories of dissatisfaction: 1) ineptitude, 2) disrespect, 3) prolonged waits, 4) ineffective communication, 5) lack of environmental control, and 6) substandard amenities. We considered the inverse of these problems to represent six implicit expectations of good hospital care: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities (Table 2). The number of patient suggestions related to each domain is detailed in Table 3.
| Domain of Dissatisfaction | Implicit Expectations | Example |
|---|---|---|
| Ineptitude | Safety | The only thing was that when I was getting ready to get discharged, one of Dr. H*'s associates came in and said, We have to readmit you for a further procedure. I said, Well, that's strange because Dr. H* put in a stent yesterday, and I'm supposed to leave today. Well, he checked, and he had the wrong guy. I'm glad I said something or else they probably would have hauled me off. |
| Disrespect | Treatment with respect and dignity | Transport was rude due to me being a heavy person. They were saying they didn't want to move me and snickering. |
| Prolonged waits | Prompt and efficient care | I called for someone because I had to use the bathroom really bad, but I had those things stuck to my legs and needed help walking to the bathroom but no one came. Well, I had to go so bad that I had a panic attack. Then all these people came rushing in to help. I felt so embarrassed. |
| Ineffective communication | Successful exchange of information | There were a few days that [were] a little confusing to me. I didn't know if I was going to have surgery or go home. The communication wasn't that great. |
| Lack of environmental control | Environmental autonomy and control | I was put in a room with a man who had many issues. He was loud and yelling all night. It was a very disturbing experience. |
| Substandard amenities | High‐quality amenities | In that ICU they should put a TV on the ceiling for when you're lying flat on your back looking at the ceiling tiles for 4 days. |
| Domain of Dissatisfaction | No. (N = 579 suggestionsa) | % of Total Surveyed | % Within Domain |
|---|---|---|---|
| |||
| Perceived ineptitude | 75 | 7.7 | 100 |
| Adverse events | 18 | 1.8 | 24.0 |
| Cleanliness | 36 | 3.7 | 48.0 |
| Perceived lack of knowledge/skill | 12 | 1.2 | 16.0 |
| Rushed out | 9 | 0.9 | 12.0 |
| Disrespect | 59 | 6.0 | 100 |
| Unprofessional staff behavior | 55 | 5.6 | 93.2 |
| Lack of privacy/confidentiality | 4 | 0.4 | 6.8 |
| Prolonged waits | 154 | 15.8 | 100 |
| Response to call bell | |||
| Bathing/toileting/distress | 24 | 2.5 | 15.6 |
| General | 41 | 4.2 | 26.6 |
| Wait for physician | 12 | 1.2 | 7.8 |
| Wait for admission bed | 29 | 3.0 | 18.8 |
| Wait for transport | 16 | 1.6 | 10.4 |
| Wait for food | 7 | 0.7 | 4.6 |
| Wait for medication | 11 | 1.1 | 7.1 |
| Wait for diagnostic test/procedures | 6 | 0.6 | 3.9 |
| Wait for discharge | 8 | 0.8 | 5.2 |
| Ineffective communication | 72 | 7.4 | 100 |
| Communication with patients | 33 | 3.4 | 45.8 |
| Communication with family | 3 | 0.3 | 4.2 |
| Translation | 2 | 0.2 | 2.8 |
| Communication between providers | 13 | 1.3 | 18.1 |
| Coordination of care (inpatient) | 11 | 1.1 | 15.3 |
| Medication reconciliation | 5 | 0.5 | 6.9 |
| Continuity inpatient to outpatient | 5 | 0.5 | 6.9 |
| Lack of environmental control | 152 | 15.6 | 100 |
| Physical environment | |||
| Roommates | 38 | 3.9 | 25.0 |
| Noise | 24 | 2.5 | 15.8 |
| Temperature | 12 | 1.2 | 7.9 |
| Smell | 1 | 0.1 | 0.7 |
| Interruption by staff | 15 | 1.5 | 9.9 |
| Lighting | 2 | 0.2 | 1.3 |
| Chaos/hectic | 4 | 0.4 | 2.6 |
| Shorter Stay | 8 | 0.8 | 5.3 |
| General | 3 | 0.3 | 2.0 |
| Facilities | |||
| Pain control | 10 | 1.0 | 6.6 |
| Painful procedures | 17 | 1.7 | 11.2 |
| Facilities | |||
| Bathrooms | 7 | 0.7 | 4.6 |
| Maintenance response | 5 | 0.5 | 3.3 |
| Traffic/parking | 6 | 0.6 | 3.9 |
| Substandard amenities | 67 | 6.9 | 100 |
| Food quality | 26 | 2.7 | 38.8 |
| Food variety | 5 | 0.5 | 7.5 |
| Food service | 16 | 1.6 | 23.9 |
| TV | 8 | 0.8 | 11.9 |
| Beds | 8 | 0.8 | 11.9 |
| Gowns | 4 | 0.4 | 6.0 |
Ineptitude
A total of 7.7% of interviewed patients reported experiencing a situation that made them feel unsafe. Dissatisfaction with safety included adverse events or near misses, uncleanliness, and a perceived lack of knowledge or skill. The implicit expectation that emerged from this domain was that the hospital would be safe, and that medical staff would be knowledgeable and skillful.
Adverse events or near misses were experienced in several areas, including diet, medication administration, patient identification, and equipment. Patients were particularly troubled when they or a family member caught the error:
There was one male nurse in training, C*, who was about to give my mother an injection. I asked what he was doing because she was about to go into surgery. He said he thought she was going home. He looked at the chart again and it turns out he was holding her roommate's chart. I don't know what would have happened if I wasn't there.
Dissatisfaction with the cleanliness of the hospital environment was also frequently expressed as a safety concern:
The rooms are dirtyThe floors are dirty. They don't sweep unless you ask them to. It took three different people to come and clean the bathroom right. I have to come back for surgery and I'm scared to death with all that bacteria and uncleanliness.
In this category, patients also described care by not too knowledgeable trainees or other staff as a safety hazard.
Disrespect
A total of 6.0% of surveyed patients suggested improvements that reflected disrespectful treatment, including poor work ethic, lack of warmth, rudeness, and a lack of attention to privacy and confidentiality. This type of dissatisfaction suggested an implicit expectation for treatment with respect and dignity that was clearly distinct from the expectation of technical quality:
[Hospital name] has always been like [this] since I started going there in 1982. They're very good technically but their bedside manner kind of sucks. You survive but you don't walk away with a warm fuzzy feeling.
Underprivileged patients were particularly sensitive to the need for respect:
I feel like the doctor that saw me that last night there was trying to get me out of there as fast as possible, saying not in so many words that it was because I don't have any insurance. I just feel like they treated me like an animal.
Violations of privacy and confidentiality were not only perceived as disrespectful, but also as a direct impediment to high‐quality care:
In the ER, I didn't like that I had no privacy especially talking with the doctor because I was in the hallway. I didn't have any privacy therefore I wasn't completely truthful with the doctor because everyone could hear.
Prolonged Waits
A total of 15.8% of patients noted dissatisfaction with wait times in the hospital. Waits for admission, transport, or discharge were frequently cited as anxiety‐provoking or frustrating:
The ER wait is too long. I was there from 8:00 AM to 2:00 AM the next day. I was there the whole day and night. When someone is in pain, they just want to be taken care of, not waiting around.
Waits related to receiving patient care, for example the inability to access nurses or physicians, more often caused feelings of fear and abandonment:
Every patient is different, I understand, but when you're there at night it can be a little scary. I was not only scared but in pain. The nurse tried to get a hold of the doctor that was on call, but the doctor took hours to respond. That was very scary.
It was also distressing to patients to watch roommates experience a delay in help for urgent needs:
The lady next to me was an elderly woman with a brace on her neck, and she couldn't speak very well. She had diarrhea at night and she would ask for a bedpan. The nurses would take forever bringing it to her. I just think when there are elderly people they should be more attentive to them because they tend to not be as vocal, you know?
Together, these comments represented an implicit expectation for prompt and efficient care.
Ineffective Communication
Communication during hospitalization was a source of dissatisfaction in 7.4% of surveyed patients. Communication failures occurred in several areas. Most common was the ineffective transfer of medical information to patients:
For days I thought I was having surgery on Friday. So all that day I ate and drank nothing and got prepped for surgery. Finally later that night I was told I was going to have it on Saturday. Saturday comes and still nothing. I never saw a surgeon or talked to anyone. Then later after that I was told I'm not having the surgery. That was the most frustrating thing.
Patients were also dissatisfied with their ability to communicate with their doctors:
I was sent home on a Friday and was sent right back on Friday night because my blood count was low and I ended up needing a blood transfusion. I tried to tell them this but they didn't listen. They need to listen to the patients.
Failed communication between care providers in the hospital was a third inadequacy noted by patients:
The only problem I had was all the different doctors coming in and out. There's so many that it confuses the patient, and a lot of them would contradict each other. One doctor said I could go home and another doctor said, No, you need to stay.
Finally, patients were dissatisfied when there was ineffective communication between inpatient and outpatient providers.
They said the VNA [Visiting Nurse Association] is supposed to come. The nurse hasn't come to see me and she hasn't called. My daughter and I have been waiting.
Thus, patients had an implicit expectation for effective communication between all parties in the hospital and were dissatisfied when any type of communication was inadequate.
Lack of Environmental Control
A total of 15.4% of surveyed patients reported dissatisfaction with the inability to control the physical environment. The inability to control noise levels, roommate behavior, temperature, smells, pain, lighting, staff interruptions, food service, smoking, and even humidity were all anxiety‐producing for different patients. The feeling of being imposed upon by an uncomfortable physical environment also extended to hospital facilities such as inaccessible bathrooms, traffic, and parking. Dissatisfaction with rooming arrangements was common:
I was in a triple room and one of my roommates had at least six visitors in the room at a time every day including two infant twins. Someone really should have said something about that. It became very disturbing, and I even left a day early because of that.
An expectation for quiet, especially during the night, was also repeatedly expressed:
The night shift could have been more considerate of people trying to rest. There was a lot of noise and bangs. I know people have to laugh and have fun but it could have been a little more quiet.
Related was the inability to control interruptions by staff members:
It's hard enough to get sleep, but then those blood suckers come in the middle of the night.
This category of dissatisfaction reflected an implicit expectation for autonomy and control over the environment so that it was conducive to rest and healing.
Substandard Amenities
A total of 6.9% of surveyed patients suggested improvements to amenities such as food, bedding, gowns, and television. Moving beyond the expectation of having peaceful surroundings, these comments reflected an expectation of a well‐appointed hospital environment with high‐quality amenities. A typical example was this comment about the food and service:
You never get what you order from the kitchen. Your tray either has something missing from it or it's the wrong tray or not the right diet. It's very frustrating and hard to get the orders the way you want.
Discussion
We analyzed 439 patient suggestions for improving hospital care and found that dissatisfaction resulted from six categories of negative experiences: 1) ineptitude, 2) disrespect, 3) prolonged wait times, 4) ineffective communication, 5) lack of environmental control, and 6) substandard amenities. These domains represented a corresponding set of implicit patient expectations for: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities. Each of these categories suggests avenues by which both the assessment and provision of hospital care can be made more patient‐centered.
The most widely used patient satisfaction survey in use in the United States today is the Hospital Consumer Assessment of Healthcare Providers & Systems (HCAHPS), which includes eight domains: communication with doctors, communication with nurses, responsiveness of hospital staff, pain management, communication about medicines, discharge information, cleanliness of the hospital environment, and quietness of the hospital environment.17 The dissatisfaction domains found in this study closely overlap the HCAHPS satisfaction domains, but with a few key differences.
First, dissatisfaction with ineptitude in our study encompassed concerns over adverse events and near misses, in addition to the cleanliness of the environment. Other research has shown that dissatisfaction with hospitalization can be predicted by the number of reported problems18 and the perception of receiving incorrect treatment.19 While elaborate methods have been devised to assess and compare the hospital quality and safety, patient satisfaction surveys including the HCAHPS survey often fail to ask patients directly about their perceptions of safety. In fact, this study and others20, 21 show that patients are able to recognize adverse events during hospitalization. Patient report may be a useful adjunct to other methods of adverse event case finding and outcomes reporting.
Second, while HCAHPS and others identify warmth, courtesy, concern, and respect as dimensions of patient‐centered care,14, 17, 22, 23 the ability of quantitative satisfaction surveys to capture the experience of disrespectful treatment may be limited, especially during hospitalization. Most respondents who commented on feeling disrespected identified only a single encounter, which can be masked by otherwise satisfying interactions with numerous care providers. Directly asking patients whether any experience during hospitalization caused them to feel disrespected, and allowing room for explanation, might more efficiently identify problem areas. This is particularly important because even one episode of disrespectful treatment, particularly when perceived to be racially motivated, increases the likelihood of not following a doctor's advice or putting off care.24
Third, HCAHPS emphasizes two aspects of communication: that between patients and doctors, and that between patients and nurses. Our patients confirmed that these are important, but they also noted a third dimension of communication contributing to dissatisfaction: provider‐provider communication. Communication and coordination failures among providers are key contributors to adverse events or near misses,2528 but their influence on patient satisfaction has not been widely assessed. Furthermore, patient input is rarely utilized to identify poor interprovider communication. Our study suggests that, just as patients can identify adverse events, they are also able to recognize poor provider‐provider communication.
Patients' reports of dissatisfying events also highlight areas in which small changes in hospital practice might greatly improve the patient experience. For instance, concerns over environment, food, sleep, hygiene, and pain appeared to be representative of a broader dissatisfaction with loss of autonomy and control. Hospitalized patients are often obliged to room with strangers, are subject to noise and interruptions, and cede control of their medication management at a time when they are feeling particularly vulnerable. The importance of this lack of autonomy to patients suggests a variety of small interventions that could improve satisfaction, such as individual control of noise and temperature, a visible commitment to a quiet hospital environment, and minimized interruptions and sleep disturbance.2932 Single‐occupancy hospital rooms have been associated with lower rates of nosocomial infection, medication errors, and patient stress, as well as increased privacy, rest, visitor involvement, and doctor‐patient communication.33, 34 The most sophisticated intervention, acuity‐adaptable private hospital rooms, allows hospitals to maintain patients in the same private hospital room during an entire admission, regardless of changes to level of acuity.35
In‐depth analysis of suggestions for improvement, as gathered by telephone surveys of recently discharged patients, was a particularly well‐suited approach to identifying explicit expectations for care that were violated by dissatisfying incidents. When allowed to express dissatisfaction in terms of suggestions for improvement, patients talked freely about specific dissatisfying experiences. Using telephone interviews allowed a large volume of patient responses to be included, unlike smaller focus groups. Our study was oral and did not rely on the literacy level of patients. Additionally, the open‐ended nature of questioning avoided some of the usual pitfalls of satisfaction surveys. We did not rely on predetermined satisfaction categories or presume the inherent value of particular attributes of care. Nonetheless, our study does have important limitations.
Patient perceptions were not compared with chart data or clinician report. Caregivers were allowed to participate in lieu of patients, which may have reduced identification of some dissatisfying events. Likewise, patients discharged to nursing homes or who were not English or Spanish speaking were excluded and may have had different dissatisfying experiences. Interviews were brief and dissatisfying events were not explored in detail. Although nearly half of respondents reported dissatisfying events, some patients may have been reluctant to criticize their care directly to a hospital representative. Finally, patients generally confined their comments to one or two dissatisfying events, even though there may have been others. We therefore cannot draw any conclusions about the relative frequency of dissatisfying events by domain.
Conclusions
All hospitalized patients bring expectations for their hospital experience. While specific expectations vary between patients, expectations for: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities were found in this study to encompass core expectations for hospitalization. It may be useful to ensure that postdischarge surveys explicitly address these expectations. Efforts to address and manage these core expectations of hospital care may help to reduce patient dissatisfaction with hospitalization and improve the delivery and quality of hospital care.
The United States spends more money per capita on healthcare than any other industrialized nation,1 yet patients are the least satisfied with their care.2 Patient satisfaction is associated in both cross‐sectional3 and longitudinal studies4 with improved physical and mental health outcomes. Conversely, dissatisfaction with care hampers future medical interactions, prevents sharing of information, and impairs the building of trust.5 The increasing recognition that a patient's experience of care affects patient outcomes has furthered efforts to evaluate satisfaction with care.6, 7
However, patient satisfaction is challenging to define and understand. Even the definition of satisfaction is ambiguous, for to satisfy can mean both to make happy and the lesser, to be adequate. To dissatisfy is to displease or disappoint, but dissatisfaction is not the opposite of satisfaction: qualitative studies give little if any indication that patients evaluate satisfaction on a continuum ranging from dissatisfied at one end to very satisfied at the other.8 Instead, it appears that satisfaction and dissatisfaction are different constructs, such that patients may simultaneously be both satisfied and dissatisfied.9, 10 Patients often express overall satisfaction with a service or encounter while also reporting specific criticisms about its shortcomings.11, 12 Alternatively, consumers may be generally satisfied unless something unpleasant or improper happens.13 Thus, dissatisfaction and satisfaction may require different methods of measurement.
The most common model for measuring patient satisfaction is a quantitative survey of patients' experiences in specific predetermined domains. Of 54 hospital satisfaction surveys in common use, only 11 included patient input in their development,14 casting doubt on the relevance of these attributes to patients' priorities of care. Since it is well recognized that patient expectations influence satisfaction,8, 13, 15 it is important to identify patient expectations and priorities up front. However, these have not been clearly established. Furthermore, focusing purely on satisfaction with particular domains of care may miss the separate but illuminating construct of patient dissatisfaction.
In this study we therefore aim to understand patient dissatisfaction with hospitalization more fully as a means of elucidating implicit expectations for hospital care. Using qualitative techniques, we analyzed a large volume of patient responses to a single open‐ended study question to identify determinants and patterns of patient dissatisfaction.
Methods
Study Design
We conducted a qualitative analysis of telephone survey data obtained from adults recently discharged after an acute care hospitalization. Survey participants were asked five questions, including: If there was one thing we could have done to improve your experience in the hospital, what would it have been? Answers to this open‐ended question were included in this study.
Setting and Participants
The hospital is a 944‐bed, urban academic medical center. Patients or patient representatives were routinely surveyed in a telephone interview conducted by trained hospital staff 1‐5 days after hospital discharge. Calls were attempted to 90% of adult discharged patients, and approximately 50% of them were reached. For this study, we included patients who were age 18 or older, spoke English, and were discharged to home from a medical, surgical, gynecology‐oncology, neurology, neurosurgery, or intensive care unit. Of those patients, we randomly selected 10% of those surveyed between July 1, 2007 and June 30, 2008 for inclusion.
Primary Data Analysis
Qualitative data analysis was used to classify patient suggestions. The study team included internal medicine physicians (J.P.M., L.I.H.), a medical student (A.V.L.), and a recent college graduate (C.P.B.). Codes were generated using a mixed inductive and deductive approach by reading and rereading the primary data.16 A set of 100 interview responses were first read individually by three investigators (J.P.M., A.V.L., C.P.B.), after which investigators met to discuss themes and ideas. A preliminary list of coding categories was then generated. Each investigator then assigned these coding categories to additional survey responses in sets of 100. Subsequent meetings were held to refine codes using the constant comparative method.16 Disagreements were resolved by negotiated consensus. The full study group met periodically to review the code structure for logic and breadth. Once thematic saturation was achieved, the entire dataset was recoded by two investigators using the final coding structure. The final coding structure contained 42 unique codes organized into six broader themes. We used descriptive statistics to characterize the coding category results. The score for intercoder reliability was 0.91.
This study was approved by the Yale Human Investigation Committee, which granted a waiver of informed consent.
Results
A total of 976 surveys was randomly selected from 9,764 postdischarge phone interviews completed between July 1, 2007 and June 30, 2008. A total of 56.3% of patients was female. Nearly half the patients were discharged from medical units (Table 1). Of the 976 patients, 439 (45.0%) gave at least one suggestion for improvement, yielding a total of 579 suggestions. Patients also offered numerous positive comments about their care, but these comments were not included in the analysis.
| No. | % of Total Surveyed | |
|---|---|---|
| Total surveys | 976 | |
| Male | 427 | 43.7 |
| Female | 549 | 56.3 |
| Discharge Unit | ||
| Medical | 434 | 44.5 |
| Surgical | 303 | 31.0 |
| Gynecology/Oncology | 103 | 10.6 |
| ICU/CCU/Step‐down | 71 | 7.3 |
| Neurology/Neurosurgery | 65 | 6.6 |
| No suggestions for improvement | 537 | 55.0 |
| At least one suggestion for improvement | 439 | 45.0 |
Through qualitative analysis, we assigned suggestions for improvement to six major categories of dissatisfaction: 1) ineptitude, 2) disrespect, 3) prolonged waits, 4) ineffective communication, 5) lack of environmental control, and 6) substandard amenities. We considered the inverse of these problems to represent six implicit expectations of good hospital care: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities (Table 2). The number of patient suggestions related to each domain is detailed in Table 3.
| Domain of Dissatisfaction | Implicit Expectations | Example |
|---|---|---|
| Ineptitude | Safety | The only thing was that when I was getting ready to get discharged, one of Dr. H*'s associates came in and said, We have to readmit you for a further procedure. I said, Well, that's strange because Dr. H* put in a stent yesterday, and I'm supposed to leave today. Well, he checked, and he had the wrong guy. I'm glad I said something or else they probably would have hauled me off. |
| Disrespect | Treatment with respect and dignity | Transport was rude due to me being a heavy person. They were saying they didn't want to move me and snickering. |
| Prolonged waits | Prompt and efficient care | I called for someone because I had to use the bathroom really bad, but I had those things stuck to my legs and needed help walking to the bathroom but no one came. Well, I had to go so bad that I had a panic attack. Then all these people came rushing in to help. I felt so embarrassed. |
| Ineffective communication | Successful exchange of information | There were a few days that [were] a little confusing to me. I didn't know if I was going to have surgery or go home. The communication wasn't that great. |
| Lack of environmental control | Environmental autonomy and control | I was put in a room with a man who had many issues. He was loud and yelling all night. It was a very disturbing experience. |
| Substandard amenities | High‐quality amenities | In that ICU they should put a TV on the ceiling for when you're lying flat on your back looking at the ceiling tiles for 4 days. |
| Domain of Dissatisfaction | No. (N = 579 suggestionsa) | % of Total Surveyed | % Within Domain |
|---|---|---|---|
| |||
| Perceived ineptitude | 75 | 7.7 | 100 |
| Adverse events | 18 | 1.8 | 24.0 |
| Cleanliness | 36 | 3.7 | 48.0 |
| Perceived lack of knowledge/skill | 12 | 1.2 | 16.0 |
| Rushed out | 9 | 0.9 | 12.0 |
| Disrespect | 59 | 6.0 | 100 |
| Unprofessional staff behavior | 55 | 5.6 | 93.2 |
| Lack of privacy/confidentiality | 4 | 0.4 | 6.8 |
| Prolonged waits | 154 | 15.8 | 100 |
| Response to call bell | |||
| Bathing/toileting/distress | 24 | 2.5 | 15.6 |
| General | 41 | 4.2 | 26.6 |
| Wait for physician | 12 | 1.2 | 7.8 |
| Wait for admission bed | 29 | 3.0 | 18.8 |
| Wait for transport | 16 | 1.6 | 10.4 |
| Wait for food | 7 | 0.7 | 4.6 |
| Wait for medication | 11 | 1.1 | 7.1 |
| Wait for diagnostic test/procedures | 6 | 0.6 | 3.9 |
| Wait for discharge | 8 | 0.8 | 5.2 |
| Ineffective communication | 72 | 7.4 | 100 |
| Communication with patients | 33 | 3.4 | 45.8 |
| Communication with family | 3 | 0.3 | 4.2 |
| Translation | 2 | 0.2 | 2.8 |
| Communication between providers | 13 | 1.3 | 18.1 |
| Coordination of care (inpatient) | 11 | 1.1 | 15.3 |
| Medication reconciliation | 5 | 0.5 | 6.9 |
| Continuity inpatient to outpatient | 5 | 0.5 | 6.9 |
| Lack of environmental control | 152 | 15.6 | 100 |
| Physical environment | |||
| Roommates | 38 | 3.9 | 25.0 |
| Noise | 24 | 2.5 | 15.8 |
| Temperature | 12 | 1.2 | 7.9 |
| Smell | 1 | 0.1 | 0.7 |
| Interruption by staff | 15 | 1.5 | 9.9 |
| Lighting | 2 | 0.2 | 1.3 |
| Chaos/hectic | 4 | 0.4 | 2.6 |
| Shorter Stay | 8 | 0.8 | 5.3 |
| General | 3 | 0.3 | 2.0 |
| Facilities | |||
| Pain control | 10 | 1.0 | 6.6 |
| Painful procedures | 17 | 1.7 | 11.2 |
| Facilities | |||
| Bathrooms | 7 | 0.7 | 4.6 |
| Maintenance response | 5 | 0.5 | 3.3 |
| Traffic/parking | 6 | 0.6 | 3.9 |
| Substandard amenities | 67 | 6.9 | 100 |
| Food quality | 26 | 2.7 | 38.8 |
| Food variety | 5 | 0.5 | 7.5 |
| Food service | 16 | 1.6 | 23.9 |
| TV | 8 | 0.8 | 11.9 |
| Beds | 8 | 0.8 | 11.9 |
| Gowns | 4 | 0.4 | 6.0 |
Ineptitude
A total of 7.7% of interviewed patients reported experiencing a situation that made them feel unsafe. Dissatisfaction with safety included adverse events or near misses, uncleanliness, and a perceived lack of knowledge or skill. The implicit expectation that emerged from this domain was that the hospital would be safe, and that medical staff would be knowledgeable and skillful.
Adverse events or near misses were experienced in several areas, including diet, medication administration, patient identification, and equipment. Patients were particularly troubled when they or a family member caught the error:
There was one male nurse in training, C*, who was about to give my mother an injection. I asked what he was doing because she was about to go into surgery. He said he thought she was going home. He looked at the chart again and it turns out he was holding her roommate's chart. I don't know what would have happened if I wasn't there.
Dissatisfaction with the cleanliness of the hospital environment was also frequently expressed as a safety concern:
The rooms are dirtyThe floors are dirty. They don't sweep unless you ask them to. It took three different people to come and clean the bathroom right. I have to come back for surgery and I'm scared to death with all that bacteria and uncleanliness.
In this category, patients also described care by not too knowledgeable trainees or other staff as a safety hazard.
Disrespect
A total of 6.0% of surveyed patients suggested improvements that reflected disrespectful treatment, including poor work ethic, lack of warmth, rudeness, and a lack of attention to privacy and confidentiality. This type of dissatisfaction suggested an implicit expectation for treatment with respect and dignity that was clearly distinct from the expectation of technical quality:
[Hospital name] has always been like [this] since I started going there in 1982. They're very good technically but their bedside manner kind of sucks. You survive but you don't walk away with a warm fuzzy feeling.
Underprivileged patients were particularly sensitive to the need for respect:
I feel like the doctor that saw me that last night there was trying to get me out of there as fast as possible, saying not in so many words that it was because I don't have any insurance. I just feel like they treated me like an animal.
Violations of privacy and confidentiality were not only perceived as disrespectful, but also as a direct impediment to high‐quality care:
In the ER, I didn't like that I had no privacy especially talking with the doctor because I was in the hallway. I didn't have any privacy therefore I wasn't completely truthful with the doctor because everyone could hear.
Prolonged Waits
A total of 15.8% of patients noted dissatisfaction with wait times in the hospital. Waits for admission, transport, or discharge were frequently cited as anxiety‐provoking or frustrating:
The ER wait is too long. I was there from 8:00 AM to 2:00 AM the next day. I was there the whole day and night. When someone is in pain, they just want to be taken care of, not waiting around.
Waits related to receiving patient care, for example the inability to access nurses or physicians, more often caused feelings of fear and abandonment:
Every patient is different, I understand, but when you're there at night it can be a little scary. I was not only scared but in pain. The nurse tried to get a hold of the doctor that was on call, but the doctor took hours to respond. That was very scary.
It was also distressing to patients to watch roommates experience a delay in help for urgent needs:
The lady next to me was an elderly woman with a brace on her neck, and she couldn't speak very well. She had diarrhea at night and she would ask for a bedpan. The nurses would take forever bringing it to her. I just think when there are elderly people they should be more attentive to them because they tend to not be as vocal, you know?
Together, these comments represented an implicit expectation for prompt and efficient care.
Ineffective Communication
Communication during hospitalization was a source of dissatisfaction in 7.4% of surveyed patients. Communication failures occurred in several areas. Most common was the ineffective transfer of medical information to patients:
For days I thought I was having surgery on Friday. So all that day I ate and drank nothing and got prepped for surgery. Finally later that night I was told I was going to have it on Saturday. Saturday comes and still nothing. I never saw a surgeon or talked to anyone. Then later after that I was told I'm not having the surgery. That was the most frustrating thing.
Patients were also dissatisfied with their ability to communicate with their doctors:
I was sent home on a Friday and was sent right back on Friday night because my blood count was low and I ended up needing a blood transfusion. I tried to tell them this but they didn't listen. They need to listen to the patients.
Failed communication between care providers in the hospital was a third inadequacy noted by patients:
The only problem I had was all the different doctors coming in and out. There's so many that it confuses the patient, and a lot of them would contradict each other. One doctor said I could go home and another doctor said, No, you need to stay.
Finally, patients were dissatisfied when there was ineffective communication between inpatient and outpatient providers.
They said the VNA [Visiting Nurse Association] is supposed to come. The nurse hasn't come to see me and she hasn't called. My daughter and I have been waiting.
Thus, patients had an implicit expectation for effective communication between all parties in the hospital and were dissatisfied when any type of communication was inadequate.
Lack of Environmental Control
A total of 15.4% of surveyed patients reported dissatisfaction with the inability to control the physical environment. The inability to control noise levels, roommate behavior, temperature, smells, pain, lighting, staff interruptions, food service, smoking, and even humidity were all anxiety‐producing for different patients. The feeling of being imposed upon by an uncomfortable physical environment also extended to hospital facilities such as inaccessible bathrooms, traffic, and parking. Dissatisfaction with rooming arrangements was common:
I was in a triple room and one of my roommates had at least six visitors in the room at a time every day including two infant twins. Someone really should have said something about that. It became very disturbing, and I even left a day early because of that.
An expectation for quiet, especially during the night, was also repeatedly expressed:
The night shift could have been more considerate of people trying to rest. There was a lot of noise and bangs. I know people have to laugh and have fun but it could have been a little more quiet.
Related was the inability to control interruptions by staff members:
It's hard enough to get sleep, but then those blood suckers come in the middle of the night.
This category of dissatisfaction reflected an implicit expectation for autonomy and control over the environment so that it was conducive to rest and healing.
Substandard Amenities
A total of 6.9% of surveyed patients suggested improvements to amenities such as food, bedding, gowns, and television. Moving beyond the expectation of having peaceful surroundings, these comments reflected an expectation of a well‐appointed hospital environment with high‐quality amenities. A typical example was this comment about the food and service:
You never get what you order from the kitchen. Your tray either has something missing from it or it's the wrong tray or not the right diet. It's very frustrating and hard to get the orders the way you want.
Discussion
We analyzed 439 patient suggestions for improving hospital care and found that dissatisfaction resulted from six categories of negative experiences: 1) ineptitude, 2) disrespect, 3) prolonged wait times, 4) ineffective communication, 5) lack of environmental control, and 6) substandard amenities. These domains represented a corresponding set of implicit patient expectations for: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities. Each of these categories suggests avenues by which both the assessment and provision of hospital care can be made more patient‐centered.
The most widely used patient satisfaction survey in use in the United States today is the Hospital Consumer Assessment of Healthcare Providers & Systems (HCAHPS), which includes eight domains: communication with doctors, communication with nurses, responsiveness of hospital staff, pain management, communication about medicines, discharge information, cleanliness of the hospital environment, and quietness of the hospital environment.17 The dissatisfaction domains found in this study closely overlap the HCAHPS satisfaction domains, but with a few key differences.
First, dissatisfaction with ineptitude in our study encompassed concerns over adverse events and near misses, in addition to the cleanliness of the environment. Other research has shown that dissatisfaction with hospitalization can be predicted by the number of reported problems18 and the perception of receiving incorrect treatment.19 While elaborate methods have been devised to assess and compare the hospital quality and safety, patient satisfaction surveys including the HCAHPS survey often fail to ask patients directly about their perceptions of safety. In fact, this study and others20, 21 show that patients are able to recognize adverse events during hospitalization. Patient report may be a useful adjunct to other methods of adverse event case finding and outcomes reporting.
Second, while HCAHPS and others identify warmth, courtesy, concern, and respect as dimensions of patient‐centered care,14, 17, 22, 23 the ability of quantitative satisfaction surveys to capture the experience of disrespectful treatment may be limited, especially during hospitalization. Most respondents who commented on feeling disrespected identified only a single encounter, which can be masked by otherwise satisfying interactions with numerous care providers. Directly asking patients whether any experience during hospitalization caused them to feel disrespected, and allowing room for explanation, might more efficiently identify problem areas. This is particularly important because even one episode of disrespectful treatment, particularly when perceived to be racially motivated, increases the likelihood of not following a doctor's advice or putting off care.24
Third, HCAHPS emphasizes two aspects of communication: that between patients and doctors, and that between patients and nurses. Our patients confirmed that these are important, but they also noted a third dimension of communication contributing to dissatisfaction: provider‐provider communication. Communication and coordination failures among providers are key contributors to adverse events or near misses,2528 but their influence on patient satisfaction has not been widely assessed. Furthermore, patient input is rarely utilized to identify poor interprovider communication. Our study suggests that, just as patients can identify adverse events, they are also able to recognize poor provider‐provider communication.
Patients' reports of dissatisfying events also highlight areas in which small changes in hospital practice might greatly improve the patient experience. For instance, concerns over environment, food, sleep, hygiene, and pain appeared to be representative of a broader dissatisfaction with loss of autonomy and control. Hospitalized patients are often obliged to room with strangers, are subject to noise and interruptions, and cede control of their medication management at a time when they are feeling particularly vulnerable. The importance of this lack of autonomy to patients suggests a variety of small interventions that could improve satisfaction, such as individual control of noise and temperature, a visible commitment to a quiet hospital environment, and minimized interruptions and sleep disturbance.2932 Single‐occupancy hospital rooms have been associated with lower rates of nosocomial infection, medication errors, and patient stress, as well as increased privacy, rest, visitor involvement, and doctor‐patient communication.33, 34 The most sophisticated intervention, acuity‐adaptable private hospital rooms, allows hospitals to maintain patients in the same private hospital room during an entire admission, regardless of changes to level of acuity.35
In‐depth analysis of suggestions for improvement, as gathered by telephone surveys of recently discharged patients, was a particularly well‐suited approach to identifying explicit expectations for care that were violated by dissatisfying incidents. When allowed to express dissatisfaction in terms of suggestions for improvement, patients talked freely about specific dissatisfying experiences. Using telephone interviews allowed a large volume of patient responses to be included, unlike smaller focus groups. Our study was oral and did not rely on the literacy level of patients. Additionally, the open‐ended nature of questioning avoided some of the usual pitfalls of satisfaction surveys. We did not rely on predetermined satisfaction categories or presume the inherent value of particular attributes of care. Nonetheless, our study does have important limitations.
Patient perceptions were not compared with chart data or clinician report. Caregivers were allowed to participate in lieu of patients, which may have reduced identification of some dissatisfying events. Likewise, patients discharged to nursing homes or who were not English or Spanish speaking were excluded and may have had different dissatisfying experiences. Interviews were brief and dissatisfying events were not explored in detail. Although nearly half of respondents reported dissatisfying events, some patients may have been reluctant to criticize their care directly to a hospital representative. Finally, patients generally confined their comments to one or two dissatisfying events, even though there may have been others. We therefore cannot draw any conclusions about the relative frequency of dissatisfying events by domain.
Conclusions
All hospitalized patients bring expectations for their hospital experience. While specific expectations vary between patients, expectations for: 1) safety, 2) treatment with respect and dignity, 3) prompt and efficient care, 4) successful exchange of information, 5) environmental autonomy and control, and 6) high‐quality amenities were found in this study to encompass core expectations for hospitalization. It may be useful to ensure that postdischarge surveys explicitly address these expectations. Efforts to address and manage these core expectations of hospital care may help to reduce patient dissatisfaction with hospitalization and improve the delivery and quality of hospital care.
- ,.Multinational Comparisons of Health Systems Data, 2006.Washington, DC:The Commonwealth Fund;2007.
- ,,,,,.Toward higher‐performance health systems: Adults' health care experiences in seven countries, 2007.Health Aff.2007;26:w717–734.
- ,,, et al.Patient satisfaction and its relationship with clinical quality and inpatient mortality in acute myocardial infarction.Circ Cardiovasc Qual Outcomes.2010;3:188–195.
- ,.A longitudinal analysis of patient satisfaction and subsequent quality of life in Hong Kong Chinese breast and nasopharyngeal cancer patients.Med Care.2009;47:875–881.
- .Understanding dissatisfied users: developing a framework for comprehending criticisms of health care work.J Adv Nurs.1999;30:723–731.
- ,.Patient satisfaction: a review of issues and concepts.Soc Sci Med.1997;45:1829–1843.
- Institute of Medicine.Crossing the Quality Chasm: A New Health System for the 21st Century.Washington, DC:National Academy Press;2001.
- .Patient satisfaction: a valid concept?Soc Sci Med.1994;38:509–516.
- .Exploring the meaning of ‘dissatisfaction’ with health care: the importance of ‘personal identity threat’.Sociol Health Illn.1999;21:95–123.
- ,.Pathways, pyramids and icebergs? Mapping the links between dissatisfaction and complaints.Sociol Health Illn.1998;20:825–847.
- ,.Convergence and divergence: assessing criteria of consumer satisfaction across general practice, dental and hospital care settings.Soc Sci Med.1991;33:707–716.
- ,,,,,.National survey of hospital patients [see comment].BMJ.1994;309:1542–1546.
- ,,, et al.The measurement of satisfaction with healthcare: implications for practice from a systematic review of the literature.Health Technol Assess.2002;6:1–244.
- ,,,.Review of the literature on survey instruments used to collect data on hospital patients' perceptions of care.Health Serv Res.2005;40:1996–2017.
- ,,.Satisfying solutions? A review of some unresolved issues in the measurement of patient satisfaction.J Adv Nurs.1995;22:316–322.
- ,.The Discovery of Grounded Theory: Strategies for Qualitative Research.Chicago, IL:Aldine;1967.
- Hospital Consumer Assessment of Healthcare Providers 25:25–36.
- ,,,.Patient experiences in relation to respondent and health service delivery characteristics: a survey of 26,938 patients attending 62 hospitals throughout Norway.Scand J Public Health.2007;35:70–77.
- ,,, et al.What can hospitalized patients tell us about adverse events? Learning from patient‐reported incidents.J Gen Intern Med.2005;20:830–836.
- .A hospitalization from hell: a patient's perspective on quality.Ann Intern Med.2003;138:33–39.
- ,,,.Through the Patient's Eyes: Understanding and Promoting Patient‐Centered Care.San Francisco, CA:Jossey‐Bass;1993.
- ,,,,.What do consumers want to know about the quality of care in hospitals?Health Serv Res.2005;40:2018–2036.
- ,.R‐e‐s‐p‐e‐c‐t: patient reports of disrespect in the health care setting and its impact on care.J Fam Pract.2004;53:721–730.
- ,,,,.Perceived information needs and communication difficulties of inpatient physicians and nurses.J Am Med Inform Assoc.2002;9(6 suppl 1):S64–S69.
- ,,.The human factor: the critical importance of effective teamwork and communication in providing safe care.Qual Saf Health Care.2004;13:i85–i90.
- ,,,,.Consequences of inadequate sign‐out for patient care.Arch Intern Med.2008;168:1755–1760.
- ,,,,,.Dropping the baton: a qualitative analysis of failures during the transition from emergency department to inpatient care.Ann Emerg Med.2009;53:701–710 e704.
- ,.Interactive relationships between hospital patients' noise‐induced stress and other stress with sleep.Heart Lung.2001;30:237–243.
- .The influence of subjective reactions to noise on health effects of the noise.Environ Int.1996;22:93–104.
- .A novel PACU design for noise reduction.J Perianesth Nurs.2008;23:226–229.
- ,,,.The Planetree Model Hospital Project: an example of the patient as partner. (Pacific Presbyterian Medical Center, San Francisco).Hosp Health Serv Admin.1990;35:591–601.
- ,,.Physician‐patient communication in single‐bedded versus four‐bedded hospital rooms.Patient Educ Couns.2008;73:215–219.
- ,,.Advantages and disadvantages of single‐versus multiple‐occupancy rooms in acute care environments: a review and analysis of the literature.Environ Behav.2005;37:760–786.
- ,.Impacting patient outcomes through design: acuity adaptable care/universal room design.Crit Care Nurs Q.2006;29:326–341.
- ,.Multinational Comparisons of Health Systems Data, 2006.Washington, DC:The Commonwealth Fund;2007.
- ,,,,,.Toward higher‐performance health systems: Adults' health care experiences in seven countries, 2007.Health Aff.2007;26:w717–734.
- ,,, et al.Patient satisfaction and its relationship with clinical quality and inpatient mortality in acute myocardial infarction.Circ Cardiovasc Qual Outcomes.2010;3:188–195.
- ,.A longitudinal analysis of patient satisfaction and subsequent quality of life in Hong Kong Chinese breast and nasopharyngeal cancer patients.Med Care.2009;47:875–881.
- .Understanding dissatisfied users: developing a framework for comprehending criticisms of health care work.J Adv Nurs.1999;30:723–731.
- ,.Patient satisfaction: a review of issues and concepts.Soc Sci Med.1997;45:1829–1843.
- Institute of Medicine.Crossing the Quality Chasm: A New Health System for the 21st Century.Washington, DC:National Academy Press;2001.
- .Patient satisfaction: a valid concept?Soc Sci Med.1994;38:509–516.
- .Exploring the meaning of ‘dissatisfaction’ with health care: the importance of ‘personal identity threat’.Sociol Health Illn.1999;21:95–123.
- ,.Pathways, pyramids and icebergs? Mapping the links between dissatisfaction and complaints.Sociol Health Illn.1998;20:825–847.
- ,.Convergence and divergence: assessing criteria of consumer satisfaction across general practice, dental and hospital care settings.Soc Sci Med.1991;33:707–716.
- ,,,,,.National survey of hospital patients [see comment].BMJ.1994;309:1542–1546.
- ,,, et al.The measurement of satisfaction with healthcare: implications for practice from a systematic review of the literature.Health Technol Assess.2002;6:1–244.
- ,,,.Review of the literature on survey instruments used to collect data on hospital patients' perceptions of care.Health Serv Res.2005;40:1996–2017.
- ,,.Satisfying solutions? A review of some unresolved issues in the measurement of patient satisfaction.J Adv Nurs.1995;22:316–322.
- ,.The Discovery of Grounded Theory: Strategies for Qualitative Research.Chicago, IL:Aldine;1967.
- Hospital Consumer Assessment of Healthcare Providers 25:25–36.
- ,,,.Patient experiences in relation to respondent and health service delivery characteristics: a survey of 26,938 patients attending 62 hospitals throughout Norway.Scand J Public Health.2007;35:70–77.
- ,,, et al.What can hospitalized patients tell us about adverse events? Learning from patient‐reported incidents.J Gen Intern Med.2005;20:830–836.
- .A hospitalization from hell: a patient's perspective on quality.Ann Intern Med.2003;138:33–39.
- ,,,.Through the Patient's Eyes: Understanding and Promoting Patient‐Centered Care.San Francisco, CA:Jossey‐Bass;1993.
- ,,,,.What do consumers want to know about the quality of care in hospitals?Health Serv Res.2005;40:2018–2036.
- ,.R‐e‐s‐p‐e‐c‐t: patient reports of disrespect in the health care setting and its impact on care.J Fam Pract.2004;53:721–730.
- ,,,,.Perceived information needs and communication difficulties of inpatient physicians and nurses.J Am Med Inform Assoc.2002;9(6 suppl 1):S64–S69.
- ,,.The human factor: the critical importance of effective teamwork and communication in providing safe care.Qual Saf Health Care.2004;13:i85–i90.
- ,,,,.Consequences of inadequate sign‐out for patient care.Arch Intern Med.2008;168:1755–1760.
- ,,,,,.Dropping the baton: a qualitative analysis of failures during the transition from emergency department to inpatient care.Ann Emerg Med.2009;53:701–710 e704.
- ,.Interactive relationships between hospital patients' noise‐induced stress and other stress with sleep.Heart Lung.2001;30:237–243.
- .The influence of subjective reactions to noise on health effects of the noise.Environ Int.1996;22:93–104.
- .A novel PACU design for noise reduction.J Perianesth Nurs.2008;23:226–229.
- ,,,.The Planetree Model Hospital Project: an example of the patient as partner. (Pacific Presbyterian Medical Center, San Francisco).Hosp Health Serv Admin.1990;35:591–601.
- ,,.Physician‐patient communication in single‐bedded versus four‐bedded hospital rooms.Patient Educ Couns.2008;73:215–219.
- ,,.Advantages and disadvantages of single‐versus multiple‐occupancy rooms in acute care environments: a review and analysis of the literature.Environ Behav.2005;37:760–786.
- ,.Impacting patient outcomes through design: acuity adaptable care/universal room design.Crit Care Nurs Q.2006;29:326–341.
Copyright © 2010 Society of Hospital Medicine
Assessing Hospitalist Communication
Effective communication between patients and physicians improves a number of important outcomes including patient adherence to treatment,1‐3 quality of the medical history4 and clinical outcomes.1, 5, 6 Recognizing the importance of physician communication skills, the American Board of Medical Specialties, American Council for Graduate Medical Education and The Joint Commission all identify communication as a core competency for physicians.7‐9 For hospitalists and their patients, building a therapeutic partnership is challenged by the lack of a preexisting relationship and potential lack of patient history information, particularly psychosocial history.10 Other factors that complicate the relationships between hospitalists and their patients include acuity of illness, limited time course, and absence of or lack of input from patients' primary physicians.11
As a rapidly increasing percentage of hospitalized patients are cared for by hospitalists,12, 13 communication skills need to be directly assessed and addressed. As of 2006, at least 37% of all Medicare claims for inpatient evaluation and management services by general internists were attributed to hospitalists, and more than half of hospitalized Medicare patients are seen by hospitalists.14 Yet, a search of the MEDLINE database for articles published between 1965 and September 2009, querying hospitalist AND patient AND communication within the article title and abstract, yielded only 2 studies assessing hospitalist‐patient communication. A 1998 study15 compared patient‐reported communication problems with hospitalists versus continuity physicians involved with hospital care, and found that patients whose continuity physicians remained involved with care during the hospitalization were less likely to report communication problems than those patients who were cared for by a hospitalist alone. A 2004 study16 utilized chart documentation to compare the end‐of‐life care and communication provided by continuity physicians and hospitalists. Hospitalists were found to document end‐of‐life care discussions more often than continuity physicians, and were more likely to be present for these meetings, which may suggest improved end‐of‐life care. Neither of these hospitalist‐patient communication studies directly assessed patient perceptions of communication with hospitalists.
We undertook this study to explore patient perceptions of communication with hospitalists using the Communication Assessment Tool (CAT), a psychometrically validated instrument for patient assessment of physician communication skills.17 The CAT was initially field tested in outpatient offices, omitting the inpatient experience. A 2008 study18 successfully adapted the CAT tool for use in assessing emergency department (ED) teams. Given the importance of physician‐patient communication when patients are sickest and most vulnerable in the hospital setting, we sought to establish a baseline assessment of patient perceptions of communication with hospitalists in our group. Second, we compared results of our CAT implementation with published results examining communication in other physician groups.
Methods
Between September 2008 and August 2009 we performed a cross‐sectional study of patients admitted to the hospital medicine service at an urban, academic medical center with 873 beds. This busy service was responsible for 10,225 admissions in 2008. Patients of age 18 years or older and cared for by a hospitalist or teaching team led by a hospitalist were eligible to participate. Exclusion criteria included patient confusion, physiological instability, non‐English speaking, patient unable to communicate, or patient in isolation status. Interviews were conducted in the patient's private room with no other staff present.
Patient perception of communication with hospitalists was measured with the CAT.17 This 15‐item survey is written at a fourth grade reading level, and measures responses along a 5‐point scale (1 = poor, 2 = fair, 3 = good, 4 = very good, 5 = excellent). The CAT was originally field tested with a convenience sample of 38 physicians from various regions within the US, across 6 specialties (Dermatology, Family Medicine, Neurosurgery, Ophthalmology, Orthopedic Surgery, and Physical Medicine & Rehabilitation). Each physician's office recruited 25 patients to complete the CAT through a phone or Internet‐based system.
The 14 core items of the CAT, which focus on communication with the individual physician, were used in this study. The 15th item, The doctor's staff treated me with respect, was dropped as it does not reflect the inpatient setting. Results for each physician are reported as the percentage of excellent responses. This dichotomized scoring is consistent with the development study, where analysis with Andrich's rating scale model19, 20 indicated that excellent scores correspond to a yes response while poor through very good scores correspond to a no response. This method of reporting scores as a percentage of excellent responses was found to be more useful for summarizing physician scores than reporting mean scores, which are highly skewed towards positive performance.17
Interviews were conducted by trained research assistants during hospitalists' weekday shifts. Hospitalists were not told which patients would be recruited, but were aware that patients on the service were being interviewed to assess communication. A list of patient names, room numbers, dates of admission, and assigned hospitalists was obtained daily from the electronic medical record system. Patients were approached on the second or third day of the hospital admission, and only if they had been assigned to the same hospitalist for at least 2 consecutive days. After explaining the study to patients and receiving verbal consent, researchers verified that the patient recognized the hospitalist, providing a photo if necessary. Patients who were not confident of their hospitalist's identity were excluded.
The 14 core items of the CAT survey were read aloud to the patient, who was provided with a copy of the instrument's scale and asked to respond with a number or word description (1 = poor to 5 = excellent). Patients were allowed to skip any questions they did not wish to answer. At the conclusion of the survey, patients were asked if they had any further comments to add. Patient demographics as well as hospitalist service (general or teaching) and unit were recorded. Most interviews were completed in less than 5 minutes. Based on the recommendations of the original development and validation of the CAT,17 we collected 20 patient surveys for each hospitalist. For CAT items that the patients skipped, we did not impute values; rather the percentage of excellent responses was calculated based on the number of questions the patient answered. To examine basic psychometric characteristics, we assessed scale reliability and performed a factor analysis using the principal components method of extraction with Varimax rotation.
This project was determined exempt by the Northwestern University Institutional Review Board.
Results
We identified 1,137 patients as potentially eligible for the study. Figure 1 shows a flowchart of patient exclusion. Of note, 107 patients consenting to participate (13% overall) were unable to identify their hospitalist by name or photo. More specifically, 70 teaching service patients (25% of 275 eligible patients) were unable to identify their hospitalist, compared to 37 patients on general service (7% of 553 eligible patients); (z = 7.58, P < 0.001). Another 21 (3%) declined to participate because they had not talked enough with their doctor to render an assessment.
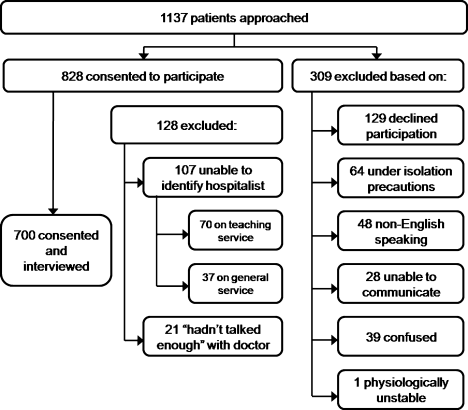
We analyzed 700 patient surveys (20 patients for each of 35 hospitalists; 62% of patients identified). Patient and hospitalist characteristics are presented in Table 1. The proportion of excellent ratings for each hospitalist ranged from 38.5% to 73.5% with an average of 59.1% excellent (standard deviation [SD] = 9.5). See Figure 2 for the distribution of hospitalist scores. For the group as a whole, highest ratings on individual CAT items were for treating the patient with respect (66% excellent), letting the patient talk without interruptions (66%), and talking in terms the patient can understand (64%). Lowest ratings were for involving the patient in decisions as much as he or she wanted (53%), encouraging the patient to ask questions (53%), and greeting the patient in a way that made him or her feel comfortable (55%). Table 2 contains a full ranking of individual item scores.
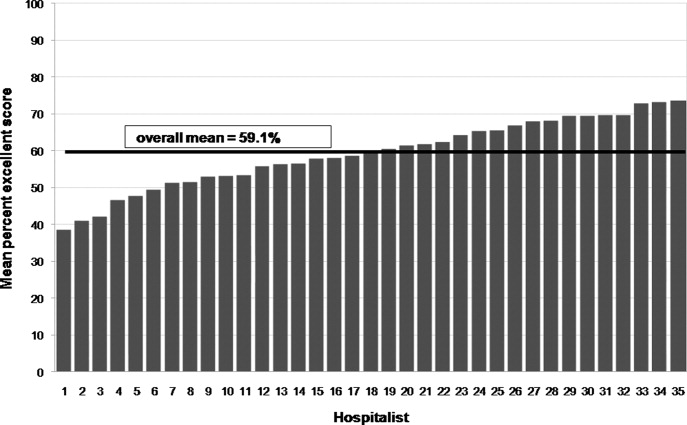
| Characteristics | |
|---|---|
| Patients (n =700), n (%) | |
| Sex, female | 378 (54) |
| Age, years | |
| 44 and younger | 189 (27) |
| 45‐64 | 266 (38) |
| 65 and older | 245 (35) |
| Race | |
| Caucasian | 357 (51) |
| African American | 266 (38) |
| Hispanic | 49 (7) |
| Other | 28 (4) |
| Hospitalists (n = 35), n (%) | |
| Sex, female | 18 (51) |
| Age, years | |
| Range | 3039 |
| Mean (SD) | 33 (2.4) |
| Race | |
| Caucasian | 14 (40) |
| South Asian | 11 (31) |
| Asian | 7 (20) |
| African American | 3 (9) |
| Non‐native English speaker | 5 (14) |
| Foreign medical graduate | 3 (9) |
| Communication Assessment Tool Item | Percent Excellent Scores |
|---|---|
| 1. Greeted me in a way that made me feel comfortable | 54.9 |
| 2. Treated me with respect | 66.3 |
| 3. Showed interest in my ideas about my health | 58.2 |
| 4. Understood my main health concerns | 57.4 |
| 5. Paid attention to me (looked at me, listened carefully) | 64.1 |
| 6. Let me talk without interruptions | 66.3 |
| 7. Gave me as much information as I wanted | 56.0 |
| 8. Talked in terms I could understand | 64.2 |
| 9. Checked to be sure I understood everything | 57.1 |
| 10. Encouraged me to ask questions | 53.2 |
| 11. Involved me in decisions as much as I wanted | 52.9 |
| 12. Discussed next steps including any follow‐up plans | 58.2 |
| 13. Showed care and concern | 63.8 |
| 14. Spent the right amount of time with me | 57.0 |
Overall scale reliability proved to be high (Cronbach's alpha = 0.97) in this sample. The factor analysis showed that scores for each of the 14 items load onto 1 factor. These results are consistent with the high reliability and single‐factor loading found in Makoul's original scale reliability and validity testing.17
The ad hoc comments made by patients at the conclusion of the CAT survey were categorized as positive or negative. Although many positive comments were made, they tended to be general in nature (eg, She is a great doctor). Negative comments were more explicit. A total of 110 patients (16%) made specific negative comments, which fell into 7 general domains: lack of information (35 comments), not enough time spent with the patient (27 comments), poor listening to the patient (24 comments), ineffective care delivery (7 comments), issues of care, concern, and respect (6 comments), ineffective communication with other staff (5 comments), and unclear role of physician (3 comments). Three patient comments were not related to these domains.
Patient age, race or gender did not correlate with CAT results. Hospitalist factors of age, race, gender, years of experience also were not associated with differences in ratings. However, race concordance between the patient and hospitalist was associated with improved CAT ratings. Patients of the same race as their hospitalist rated the hospitalist's communication significantly higher (M = 64.9%, SD = 39.1) than did patients who were of a different race than their hospitalist (M = 57.3%, SD = 40.3), P < 0.05. Gender concordance was not associated with improved CAT ratings. No score differences were found between patients cared for by a hospitalist on teaching service and direct care, and there were no differences between nursing units.
Discussion
To the best of our knowledge, this is the first study to explicitly measure patient perceptions of communication with hospitalists. The results yielded a wide distribution of scores for physicians within a single, large hospital medicine group. Comparing their own scores to those of peers may allow low‐scoring hospitalists to grasp the potential for improving their communication with patients. Our reliability testing matched the results of the original development study,17 indicating very high overall scale reliability. This suggests that the CAT could be streamlined by dropping some of the survey items. However we agree with Makoul et al.17 that it is best to keep the full set as it provides specific information for physicians without placing undue burden on patients (ie, the CAT takes only 1‐2 min to complete). Individual item scores for each of the 14 CAT items highlight specific communication tasks where intervention may be targeted for individual hospitalists and the group as a whole. It may be feasible to utilize CAT results as an individual report card for physicians. While program leaders should be aware that implementation of the CAT requires standardized data collection, it may be possible to build this into existing structures such as the discharge process.
Interestingly, many patients could not recognize the hospitalist caring for them by name or photo. More than 1 in 10 patients (107 of 828; 13%) were unable to identify their hospitalist. This was more than 3 times as common on the teaching service, where the hospitalist is accompanied by house staff and the intern or resident is the primary physician for patient contact, compared to the service on which hospitalists directly take care of patients without residents. It is also troubling that another 3% of patients (21 of 828) stated they hadn't talked enough with their hospitalist to answer basic communication questions, when approached 2 or 3 days into the relationship. It may be telling that Greeted me in a way that made me feel comfortable was one of the lowest‐rated survey items. Hospitalists should recognize that patients, in addition to facing their own physical and emotional stressors, see many hospital staff members throughout the day; all of whom may be strangers to them. Thus it becomes vital for hospitalists to not only establish an initial rapport with the patient, but to reintroduce themselves each time they enter the room.
An examination of the ad hoc negative comments made by survey respondents reinforces and extends findings related to the CAT items, particularly about those areas of communication valued by patients. The majority of comments fell into categories of failing to give enough information (eg, Sometimes I was left confused when the doctor was ready to leave), not spending enough time with the patient (eg, He was just in and out), and not listening to the patient's own ideas (eg, When giving my history, she cut me off at some points when I had more to say). The information and time categories may directly relate to scores on the CAT items Gave me as much information as I wanted and Spent the right amount of time with me, which are among the lowest‐scoring items. Listening to the patient may reflect broader issues of considering the patient's own experience, questions, concerns and goals.
In this study, patient‐physician race concordance was associated with CAT ratings. Patients who were of the same race as their hospitalist rated the hospitalist higher compared to patients who were of a different race than their hospitalist. This effect is consistent with previous research describing higher patient ratings of communication and care when the patient and physician are of the same race or ethnicity.21
A number of factors limit interpretation of the results of this study. The data were collected at a single site, thus limiting generalizability to other hospitalist practice environments. We used a retrospective, patient assessment of hospitalist communication which may have inherent biases different from a study using direct researcher observation or recording of patient‐hospitalist interactions to assess communication. This methodology allowed us to examine the patient's own perceptions and expectations of communication, but certainly leaves room for selection bias in recruitment and recall bias. Patients were interviewed on the second or third day of their admission. This controlled the length of exposure to the hospitalist, but the course of treatment might vary considerably; at the time of interview, some patients may not yet have had a clear diagnosis and plan while others may have been ready for discharge. Future work should examine how stage of evaluation and management might affect patients' perception of communication with hospitalists. Severity of condition is another factor that may affect patients' ratings, and was not examined in this study.
When compared to physicians from the CAT development study's field test, this study sample of hospitalists scored much lower, 59.1% excellent vs. 76.3% (P < 0.001). A number of factors may account for some of these differences. The majority of patients in the original field test had multiple interactions with their physician, and rated their health status as good or very good. In contrast, hospitalized patients usually lack previous exposure to the hospitalist, and likely have poorer health status. Also, physicians in the original field test volunteered to participate, and patients completed the CAT survey through the Internet or phone response system, rather than through a face‐to‐face interview by trained research assistants. Another key difference is that field‐test patients answered the CAT within 1 day of their outpatient visit, while in this study patients were interviewed in the midst of their hospital admission and prior to completion of their hospital course. Finally, patients commonly choose their outpatient physician and can select someone else if dissatisfied with their communication skills, while hospitalized patients are assigned hospitalists based on availability. Thus, given this potential selection bias, outpatients could be expected to rate their personal physician higher.
Another possibility is that hospitalists are on average less skilled in patient communication than outpatient physicians. Given the transient nature of the inpatient relationship, hospitalists may not value developing rapport with patients, and may not make this a goal of patient care or seek extensive training in communication skills. In future research, evaluating hospitalists' training in and attitudes towards patient communication could be paired with communication assessment results.
Although it is beyond the scope of this study to assess precisely how these environmental and survey implementation factors may affect CAT summary scores, their importance is evident. Another hospital‐based implementation of the CAT tool, an evaluation of ED teams,18 utilized face‐to‐face interviews with trained research assistants. The study yielded results similar to our findings: the average percent excellent score for ED teams was 62.3%, vs. 58.2% percent excellent for our hospitalist group. Taken together, these study comparisons between the original field‐test, our hospitalist implementation, and the ED team implementation support the argument that factors of setting (inpatient vs. outpatient), mode of survey administration (face‐to‐face interview vs. self‐administration through phone or Internet), and shorter duration or course of patient‐physician interaction may be important considerations when implementing the CAT tool to assess physician communication skills, or attempting to set standards of minimally acceptable or desired scoring.
More work must be done to establish norms and/or minimally acceptable scores for hospitalists. Numerous factors of specialty, practice setting, survey implementation, patient variables, and even the expertise of who is setting the communication standards22 may strongly influence comparisons between physician groups, even within a single institution. Organizations seeking to establish norms or minimally acceptable scores for physician‐patient communication should be aware of these factors. As the original development study points out, standard‐setting studies could establish specialty‐specific and country‐specific norms as well as norms or standards for level‐of‐training (eg, medical students versus attending physicians).17
Conclusion
The previously validated CAT instrument appears to have reliable test characteristics and can be used to gauge patient perceptions of hospitalist communication skills. Comparative scores between physicians of different specialties and settings should be interpreted cautiously as there may be confounding variables. Within our single institution, comparative scores between hospitalists, along with an examination of the hospitalist's individual item scores, may offer useful feedback for efforts aimed at enhancing communication. Many hospitalists in this study may benefit from targeted training to improve patient communication skills, particularly in the areas of encouraging questions and involving patients in decision making. Future qualitative research in the context of hospital medicine could identify specific communication techniques used by highly‐rated physicians, with the goal of developing tools for targeted improvement and determining impact on outcomes.
Acknowledgements
The authors thank Christie Edwards, Rachel Grayer and Caitlin Lawes for assistance with data collection, and Jie Peng for help with the analysis.
- ,,.Is the quality of the patient‐provider relationship associated with better adherence and health outcomes for patients with HIV?J Gen Intern Med.2006;21(6):661–665.
- ,,,,.Does physician communication influence older patients' diabetes self‐management and glycemic control? Results from the Health and Retirement Study (HRS).J Gerontol A Biol Sci Med Sci.2007;62(12):1435–1442.
- ,.Physician communication and patient adherence to treatment: a meta‐analysis.Med Care.2009;47(8):826–834.
- ,.The effect of physician behavior on the collection of data.Ann Intern Med.1984;101(5):692–696.
- ,,,,.Does physician‐patient communication that aims at empowering patients improve clinical outcome? A case study.Patient Educ Couns.2006;61(2):299–306.
- .Effective physician‐patient communication and health outcomes: a review.CMAJ.1995;152(9):1423–1433.
- .Evaluation of clinical competencies: basic certification, subspecialty certification, and recertification.Am J Phys Med Rehabil.2000;79(5):478–480.
- ,,,,.General competencies and accreditation in graduate medical education.Health Aff (Millwood).2002;21(5):103–111.
- Joint Commission on Accreditation of Healthcare Organizations. The Joint Commission Standards supporting effective communication, cultural competence, and patient‐centered care.2009:44.
- .Rapport and the hospitalist.Am J Med.2001;111(9B):31S–35S.
- ,.The emerging role of “hospitalists” in the American health care system.N Engl J Med.1996;335(7):514–517.
- ,,,.Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians.Ann Intern Med.1999;130(4 Pt 2):343–349.
- ,,,.The status of hospital medicine groups in the United States.J Hosp Med.2006;1:75–80.
- ,,,.Growth in the care of older patients by hospitalists in the United States.N Engl J Med.2009;360(11):1102–1112.
- ,,,,.Communication problems for patients hospitalized with chest pain.J Gen Intern Med.1998;13(12):836–838.
- ,.End‐of‐life care in a voluntary hospitalist model: effects on communication, processes of care, and patient symptoms.Am J Med.2004;116(10):669–675.
- ,,.Measuring patient views of physician communication skills: development and testing of the Communication Assessment Tool.Patient Educ Couns.2007;67(3):333–342.
- ,,, et al.Patient perspectives on communication with the medical team: pilot study using the Communication Assessment Tool‐Team (CAT‐T).Patient Educ Couns.2008;73(2):220–223.
- .Understanding resistance to the data‐model relationship in Rasch's paradigm: a reflection for the next generation.J Appl Meas.2002;3(3):325–359.
- ,.Conditional pairwise estimation in the Rasch model for ordered response categories using principal components.J Appl Meas.2003;4(3):205–221.
- ,,,,,.Patient‐centered communication, ratings of care, and concordance of patient and physician race.Ann Intern Med.2003;139(11):907–915.
- ,,,.The impact of judge selection on standard setting for a patient survey of physician communication skills.Acad Med.2008;83(10 Suppl):S17–S20.
Effective communication between patients and physicians improves a number of important outcomes including patient adherence to treatment,1‐3 quality of the medical history4 and clinical outcomes.1, 5, 6 Recognizing the importance of physician communication skills, the American Board of Medical Specialties, American Council for Graduate Medical Education and The Joint Commission all identify communication as a core competency for physicians.7‐9 For hospitalists and their patients, building a therapeutic partnership is challenged by the lack of a preexisting relationship and potential lack of patient history information, particularly psychosocial history.10 Other factors that complicate the relationships between hospitalists and their patients include acuity of illness, limited time course, and absence of or lack of input from patients' primary physicians.11
As a rapidly increasing percentage of hospitalized patients are cared for by hospitalists,12, 13 communication skills need to be directly assessed and addressed. As of 2006, at least 37% of all Medicare claims for inpatient evaluation and management services by general internists were attributed to hospitalists, and more than half of hospitalized Medicare patients are seen by hospitalists.14 Yet, a search of the MEDLINE database for articles published between 1965 and September 2009, querying hospitalist AND patient AND communication within the article title and abstract, yielded only 2 studies assessing hospitalist‐patient communication. A 1998 study15 compared patient‐reported communication problems with hospitalists versus continuity physicians involved with hospital care, and found that patients whose continuity physicians remained involved with care during the hospitalization were less likely to report communication problems than those patients who were cared for by a hospitalist alone. A 2004 study16 utilized chart documentation to compare the end‐of‐life care and communication provided by continuity physicians and hospitalists. Hospitalists were found to document end‐of‐life care discussions more often than continuity physicians, and were more likely to be present for these meetings, which may suggest improved end‐of‐life care. Neither of these hospitalist‐patient communication studies directly assessed patient perceptions of communication with hospitalists.
We undertook this study to explore patient perceptions of communication with hospitalists using the Communication Assessment Tool (CAT), a psychometrically validated instrument for patient assessment of physician communication skills.17 The CAT was initially field tested in outpatient offices, omitting the inpatient experience. A 2008 study18 successfully adapted the CAT tool for use in assessing emergency department (ED) teams. Given the importance of physician‐patient communication when patients are sickest and most vulnerable in the hospital setting, we sought to establish a baseline assessment of patient perceptions of communication with hospitalists in our group. Second, we compared results of our CAT implementation with published results examining communication in other physician groups.
Methods
Between September 2008 and August 2009 we performed a cross‐sectional study of patients admitted to the hospital medicine service at an urban, academic medical center with 873 beds. This busy service was responsible for 10,225 admissions in 2008. Patients of age 18 years or older and cared for by a hospitalist or teaching team led by a hospitalist were eligible to participate. Exclusion criteria included patient confusion, physiological instability, non‐English speaking, patient unable to communicate, or patient in isolation status. Interviews were conducted in the patient's private room with no other staff present.
Patient perception of communication with hospitalists was measured with the CAT.17 This 15‐item survey is written at a fourth grade reading level, and measures responses along a 5‐point scale (1 = poor, 2 = fair, 3 = good, 4 = very good, 5 = excellent). The CAT was originally field tested with a convenience sample of 38 physicians from various regions within the US, across 6 specialties (Dermatology, Family Medicine, Neurosurgery, Ophthalmology, Orthopedic Surgery, and Physical Medicine & Rehabilitation). Each physician's office recruited 25 patients to complete the CAT through a phone or Internet‐based system.
The 14 core items of the CAT, which focus on communication with the individual physician, were used in this study. The 15th item, The doctor's staff treated me with respect, was dropped as it does not reflect the inpatient setting. Results for each physician are reported as the percentage of excellent responses. This dichotomized scoring is consistent with the development study, where analysis with Andrich's rating scale model19, 20 indicated that excellent scores correspond to a yes response while poor through very good scores correspond to a no response. This method of reporting scores as a percentage of excellent responses was found to be more useful for summarizing physician scores than reporting mean scores, which are highly skewed towards positive performance.17
Interviews were conducted by trained research assistants during hospitalists' weekday shifts. Hospitalists were not told which patients would be recruited, but were aware that patients on the service were being interviewed to assess communication. A list of patient names, room numbers, dates of admission, and assigned hospitalists was obtained daily from the electronic medical record system. Patients were approached on the second or third day of the hospital admission, and only if they had been assigned to the same hospitalist for at least 2 consecutive days. After explaining the study to patients and receiving verbal consent, researchers verified that the patient recognized the hospitalist, providing a photo if necessary. Patients who were not confident of their hospitalist's identity were excluded.
The 14 core items of the CAT survey were read aloud to the patient, who was provided with a copy of the instrument's scale and asked to respond with a number or word description (1 = poor to 5 = excellent). Patients were allowed to skip any questions they did not wish to answer. At the conclusion of the survey, patients were asked if they had any further comments to add. Patient demographics as well as hospitalist service (general or teaching) and unit were recorded. Most interviews were completed in less than 5 minutes. Based on the recommendations of the original development and validation of the CAT,17 we collected 20 patient surveys for each hospitalist. For CAT items that the patients skipped, we did not impute values; rather the percentage of excellent responses was calculated based on the number of questions the patient answered. To examine basic psychometric characteristics, we assessed scale reliability and performed a factor analysis using the principal components method of extraction with Varimax rotation.
This project was determined exempt by the Northwestern University Institutional Review Board.
Results
We identified 1,137 patients as potentially eligible for the study. Figure 1 shows a flowchart of patient exclusion. Of note, 107 patients consenting to participate (13% overall) were unable to identify their hospitalist by name or photo. More specifically, 70 teaching service patients (25% of 275 eligible patients) were unable to identify their hospitalist, compared to 37 patients on general service (7% of 553 eligible patients); (z = 7.58, P < 0.001). Another 21 (3%) declined to participate because they had not talked enough with their doctor to render an assessment.

We analyzed 700 patient surveys (20 patients for each of 35 hospitalists; 62% of patients identified). Patient and hospitalist characteristics are presented in Table 1. The proportion of excellent ratings for each hospitalist ranged from 38.5% to 73.5% with an average of 59.1% excellent (standard deviation [SD] = 9.5). See Figure 2 for the distribution of hospitalist scores. For the group as a whole, highest ratings on individual CAT items were for treating the patient with respect (66% excellent), letting the patient talk without interruptions (66%), and talking in terms the patient can understand (64%). Lowest ratings were for involving the patient in decisions as much as he or she wanted (53%), encouraging the patient to ask questions (53%), and greeting the patient in a way that made him or her feel comfortable (55%). Table 2 contains a full ranking of individual item scores.

| Characteristics | |
|---|---|
| Patients (n =700), n (%) | |
| Sex, female | 378 (54) |
| Age, years | |
| 44 and younger | 189 (27) |
| 45‐64 | 266 (38) |
| 65 and older | 245 (35) |
| Race | |
| Caucasian | 357 (51) |
| African American | 266 (38) |
| Hispanic | 49 (7) |
| Other | 28 (4) |
| Hospitalists (n = 35), n (%) | |
| Sex, female | 18 (51) |
| Age, years | |
| Range | 3039 |
| Mean (SD) | 33 (2.4) |
| Race | |
| Caucasian | 14 (40) |
| South Asian | 11 (31) |
| Asian | 7 (20) |
| African American | 3 (9) |
| Non‐native English speaker | 5 (14) |
| Foreign medical graduate | 3 (9) |
| Communication Assessment Tool Item | Percent Excellent Scores |
|---|---|
| 1. Greeted me in a way that made me feel comfortable | 54.9 |
| 2. Treated me with respect | 66.3 |
| 3. Showed interest in my ideas about my health | 58.2 |
| 4. Understood my main health concerns | 57.4 |
| 5. Paid attention to me (looked at me, listened carefully) | 64.1 |
| 6. Let me talk without interruptions | 66.3 |
| 7. Gave me as much information as I wanted | 56.0 |
| 8. Talked in terms I could understand | 64.2 |
| 9. Checked to be sure I understood everything | 57.1 |
| 10. Encouraged me to ask questions | 53.2 |
| 11. Involved me in decisions as much as I wanted | 52.9 |
| 12. Discussed next steps including any follow‐up plans | 58.2 |
| 13. Showed care and concern | 63.8 |
| 14. Spent the right amount of time with me | 57.0 |
Overall scale reliability proved to be high (Cronbach's alpha = 0.97) in this sample. The factor analysis showed that scores for each of the 14 items load onto 1 factor. These results are consistent with the high reliability and single‐factor loading found in Makoul's original scale reliability and validity testing.17
The ad hoc comments made by patients at the conclusion of the CAT survey were categorized as positive or negative. Although many positive comments were made, they tended to be general in nature (eg, She is a great doctor). Negative comments were more explicit. A total of 110 patients (16%) made specific negative comments, which fell into 7 general domains: lack of information (35 comments), not enough time spent with the patient (27 comments), poor listening to the patient (24 comments), ineffective care delivery (7 comments), issues of care, concern, and respect (6 comments), ineffective communication with other staff (5 comments), and unclear role of physician (3 comments). Three patient comments were not related to these domains.
Patient age, race or gender did not correlate with CAT results. Hospitalist factors of age, race, gender, years of experience also were not associated with differences in ratings. However, race concordance between the patient and hospitalist was associated with improved CAT ratings. Patients of the same race as their hospitalist rated the hospitalist's communication significantly higher (M = 64.9%, SD = 39.1) than did patients who were of a different race than their hospitalist (M = 57.3%, SD = 40.3), P < 0.05. Gender concordance was not associated with improved CAT ratings. No score differences were found between patients cared for by a hospitalist on teaching service and direct care, and there were no differences between nursing units.
Discussion
To the best of our knowledge, this is the first study to explicitly measure patient perceptions of communication with hospitalists. The results yielded a wide distribution of scores for physicians within a single, large hospital medicine group. Comparing their own scores to those of peers may allow low‐scoring hospitalists to grasp the potential for improving their communication with patients. Our reliability testing matched the results of the original development study,17 indicating very high overall scale reliability. This suggests that the CAT could be streamlined by dropping some of the survey items. However we agree with Makoul et al.17 that it is best to keep the full set as it provides specific information for physicians without placing undue burden on patients (ie, the CAT takes only 1‐2 min to complete). Individual item scores for each of the 14 CAT items highlight specific communication tasks where intervention may be targeted for individual hospitalists and the group as a whole. It may be feasible to utilize CAT results as an individual report card for physicians. While program leaders should be aware that implementation of the CAT requires standardized data collection, it may be possible to build this into existing structures such as the discharge process.
Interestingly, many patients could not recognize the hospitalist caring for them by name or photo. More than 1 in 10 patients (107 of 828; 13%) were unable to identify their hospitalist. This was more than 3 times as common on the teaching service, where the hospitalist is accompanied by house staff and the intern or resident is the primary physician for patient contact, compared to the service on which hospitalists directly take care of patients without residents. It is also troubling that another 3% of patients (21 of 828) stated they hadn't talked enough with their hospitalist to answer basic communication questions, when approached 2 or 3 days into the relationship. It may be telling that Greeted me in a way that made me feel comfortable was one of the lowest‐rated survey items. Hospitalists should recognize that patients, in addition to facing their own physical and emotional stressors, see many hospital staff members throughout the day; all of whom may be strangers to them. Thus it becomes vital for hospitalists to not only establish an initial rapport with the patient, but to reintroduce themselves each time they enter the room.
An examination of the ad hoc negative comments made by survey respondents reinforces and extends findings related to the CAT items, particularly about those areas of communication valued by patients. The majority of comments fell into categories of failing to give enough information (eg, Sometimes I was left confused when the doctor was ready to leave), not spending enough time with the patient (eg, He was just in and out), and not listening to the patient's own ideas (eg, When giving my history, she cut me off at some points when I had more to say). The information and time categories may directly relate to scores on the CAT items Gave me as much information as I wanted and Spent the right amount of time with me, which are among the lowest‐scoring items. Listening to the patient may reflect broader issues of considering the patient's own experience, questions, concerns and goals.
In this study, patient‐physician race concordance was associated with CAT ratings. Patients who were of the same race as their hospitalist rated the hospitalist higher compared to patients who were of a different race than their hospitalist. This effect is consistent with previous research describing higher patient ratings of communication and care when the patient and physician are of the same race or ethnicity.21
A number of factors limit interpretation of the results of this study. The data were collected at a single site, thus limiting generalizability to other hospitalist practice environments. We used a retrospective, patient assessment of hospitalist communication which may have inherent biases different from a study using direct researcher observation or recording of patient‐hospitalist interactions to assess communication. This methodology allowed us to examine the patient's own perceptions and expectations of communication, but certainly leaves room for selection bias in recruitment and recall bias. Patients were interviewed on the second or third day of their admission. This controlled the length of exposure to the hospitalist, but the course of treatment might vary considerably; at the time of interview, some patients may not yet have had a clear diagnosis and plan while others may have been ready for discharge. Future work should examine how stage of evaluation and management might affect patients' perception of communication with hospitalists. Severity of condition is another factor that may affect patients' ratings, and was not examined in this study.
When compared to physicians from the CAT development study's field test, this study sample of hospitalists scored much lower, 59.1% excellent vs. 76.3% (P < 0.001). A number of factors may account for some of these differences. The majority of patients in the original field test had multiple interactions with their physician, and rated their health status as good or very good. In contrast, hospitalized patients usually lack previous exposure to the hospitalist, and likely have poorer health status. Also, physicians in the original field test volunteered to participate, and patients completed the CAT survey through the Internet or phone response system, rather than through a face‐to‐face interview by trained research assistants. Another key difference is that field‐test patients answered the CAT within 1 day of their outpatient visit, while in this study patients were interviewed in the midst of their hospital admission and prior to completion of their hospital course. Finally, patients commonly choose their outpatient physician and can select someone else if dissatisfied with their communication skills, while hospitalized patients are assigned hospitalists based on availability. Thus, given this potential selection bias, outpatients could be expected to rate their personal physician higher.
Another possibility is that hospitalists are on average less skilled in patient communication than outpatient physicians. Given the transient nature of the inpatient relationship, hospitalists may not value developing rapport with patients, and may not make this a goal of patient care or seek extensive training in communication skills. In future research, evaluating hospitalists' training in and attitudes towards patient communication could be paired with communication assessment results.
Although it is beyond the scope of this study to assess precisely how these environmental and survey implementation factors may affect CAT summary scores, their importance is evident. Another hospital‐based implementation of the CAT tool, an evaluation of ED teams,18 utilized face‐to‐face interviews with trained research assistants. The study yielded results similar to our findings: the average percent excellent score for ED teams was 62.3%, vs. 58.2% percent excellent for our hospitalist group. Taken together, these study comparisons between the original field‐test, our hospitalist implementation, and the ED team implementation support the argument that factors of setting (inpatient vs. outpatient), mode of survey administration (face‐to‐face interview vs. self‐administration through phone or Internet), and shorter duration or course of patient‐physician interaction may be important considerations when implementing the CAT tool to assess physician communication skills, or attempting to set standards of minimally acceptable or desired scoring.
More work must be done to establish norms and/or minimally acceptable scores for hospitalists. Numerous factors of specialty, practice setting, survey implementation, patient variables, and even the expertise of who is setting the communication standards22 may strongly influence comparisons between physician groups, even within a single institution. Organizations seeking to establish norms or minimally acceptable scores for physician‐patient communication should be aware of these factors. As the original development study points out, standard‐setting studies could establish specialty‐specific and country‐specific norms as well as norms or standards for level‐of‐training (eg, medical students versus attending physicians).17
Conclusion
The previously validated CAT instrument appears to have reliable test characteristics and can be used to gauge patient perceptions of hospitalist communication skills. Comparative scores between physicians of different specialties and settings should be interpreted cautiously as there may be confounding variables. Within our single institution, comparative scores between hospitalists, along with an examination of the hospitalist's individual item scores, may offer useful feedback for efforts aimed at enhancing communication. Many hospitalists in this study may benefit from targeted training to improve patient communication skills, particularly in the areas of encouraging questions and involving patients in decision making. Future qualitative research in the context of hospital medicine could identify specific communication techniques used by highly‐rated physicians, with the goal of developing tools for targeted improvement and determining impact on outcomes.
Acknowledgements
The authors thank Christie Edwards, Rachel Grayer and Caitlin Lawes for assistance with data collection, and Jie Peng for help with the analysis.
Effective communication between patients and physicians improves a number of important outcomes including patient adherence to treatment,1‐3 quality of the medical history4 and clinical outcomes.1, 5, 6 Recognizing the importance of physician communication skills, the American Board of Medical Specialties, American Council for Graduate Medical Education and The Joint Commission all identify communication as a core competency for physicians.7‐9 For hospitalists and their patients, building a therapeutic partnership is challenged by the lack of a preexisting relationship and potential lack of patient history information, particularly psychosocial history.10 Other factors that complicate the relationships between hospitalists and their patients include acuity of illness, limited time course, and absence of or lack of input from patients' primary physicians.11
As a rapidly increasing percentage of hospitalized patients are cared for by hospitalists,12, 13 communication skills need to be directly assessed and addressed. As of 2006, at least 37% of all Medicare claims for inpatient evaluation and management services by general internists were attributed to hospitalists, and more than half of hospitalized Medicare patients are seen by hospitalists.14 Yet, a search of the MEDLINE database for articles published between 1965 and September 2009, querying hospitalist AND patient AND communication within the article title and abstract, yielded only 2 studies assessing hospitalist‐patient communication. A 1998 study15 compared patient‐reported communication problems with hospitalists versus continuity physicians involved with hospital care, and found that patients whose continuity physicians remained involved with care during the hospitalization were less likely to report communication problems than those patients who were cared for by a hospitalist alone. A 2004 study16 utilized chart documentation to compare the end‐of‐life care and communication provided by continuity physicians and hospitalists. Hospitalists were found to document end‐of‐life care discussions more often than continuity physicians, and were more likely to be present for these meetings, which may suggest improved end‐of‐life care. Neither of these hospitalist‐patient communication studies directly assessed patient perceptions of communication with hospitalists.
We undertook this study to explore patient perceptions of communication with hospitalists using the Communication Assessment Tool (CAT), a psychometrically validated instrument for patient assessment of physician communication skills.17 The CAT was initially field tested in outpatient offices, omitting the inpatient experience. A 2008 study18 successfully adapted the CAT tool for use in assessing emergency department (ED) teams. Given the importance of physician‐patient communication when patients are sickest and most vulnerable in the hospital setting, we sought to establish a baseline assessment of patient perceptions of communication with hospitalists in our group. Second, we compared results of our CAT implementation with published results examining communication in other physician groups.
Methods
Between September 2008 and August 2009 we performed a cross‐sectional study of patients admitted to the hospital medicine service at an urban, academic medical center with 873 beds. This busy service was responsible for 10,225 admissions in 2008. Patients of age 18 years or older and cared for by a hospitalist or teaching team led by a hospitalist were eligible to participate. Exclusion criteria included patient confusion, physiological instability, non‐English speaking, patient unable to communicate, or patient in isolation status. Interviews were conducted in the patient's private room with no other staff present.
Patient perception of communication with hospitalists was measured with the CAT.17 This 15‐item survey is written at a fourth grade reading level, and measures responses along a 5‐point scale (1 = poor, 2 = fair, 3 = good, 4 = very good, 5 = excellent). The CAT was originally field tested with a convenience sample of 38 physicians from various regions within the US, across 6 specialties (Dermatology, Family Medicine, Neurosurgery, Ophthalmology, Orthopedic Surgery, and Physical Medicine & Rehabilitation). Each physician's office recruited 25 patients to complete the CAT through a phone or Internet‐based system.
The 14 core items of the CAT, which focus on communication with the individual physician, were used in this study. The 15th item, The doctor's staff treated me with respect, was dropped as it does not reflect the inpatient setting. Results for each physician are reported as the percentage of excellent responses. This dichotomized scoring is consistent with the development study, where analysis with Andrich's rating scale model19, 20 indicated that excellent scores correspond to a yes response while poor through very good scores correspond to a no response. This method of reporting scores as a percentage of excellent responses was found to be more useful for summarizing physician scores than reporting mean scores, which are highly skewed towards positive performance.17
Interviews were conducted by trained research assistants during hospitalists' weekday shifts. Hospitalists were not told which patients would be recruited, but were aware that patients on the service were being interviewed to assess communication. A list of patient names, room numbers, dates of admission, and assigned hospitalists was obtained daily from the electronic medical record system. Patients were approached on the second or third day of the hospital admission, and only if they had been assigned to the same hospitalist for at least 2 consecutive days. After explaining the study to patients and receiving verbal consent, researchers verified that the patient recognized the hospitalist, providing a photo if necessary. Patients who were not confident of their hospitalist's identity were excluded.
The 14 core items of the CAT survey were read aloud to the patient, who was provided with a copy of the instrument's scale and asked to respond with a number or word description (1 = poor to 5 = excellent). Patients were allowed to skip any questions they did not wish to answer. At the conclusion of the survey, patients were asked if they had any further comments to add. Patient demographics as well as hospitalist service (general or teaching) and unit were recorded. Most interviews were completed in less than 5 minutes. Based on the recommendations of the original development and validation of the CAT,17 we collected 20 patient surveys for each hospitalist. For CAT items that the patients skipped, we did not impute values; rather the percentage of excellent responses was calculated based on the number of questions the patient answered. To examine basic psychometric characteristics, we assessed scale reliability and performed a factor analysis using the principal components method of extraction with Varimax rotation.
This project was determined exempt by the Northwestern University Institutional Review Board.
Results
We identified 1,137 patients as potentially eligible for the study. Figure 1 shows a flowchart of patient exclusion. Of note, 107 patients consenting to participate (13% overall) were unable to identify their hospitalist by name or photo. More specifically, 70 teaching service patients (25% of 275 eligible patients) were unable to identify their hospitalist, compared to 37 patients on general service (7% of 553 eligible patients); (z = 7.58, P < 0.001). Another 21 (3%) declined to participate because they had not talked enough with their doctor to render an assessment.

We analyzed 700 patient surveys (20 patients for each of 35 hospitalists; 62% of patients identified). Patient and hospitalist characteristics are presented in Table 1. The proportion of excellent ratings for each hospitalist ranged from 38.5% to 73.5% with an average of 59.1% excellent (standard deviation [SD] = 9.5). See Figure 2 for the distribution of hospitalist scores. For the group as a whole, highest ratings on individual CAT items were for treating the patient with respect (66% excellent), letting the patient talk without interruptions (66%), and talking in terms the patient can understand (64%). Lowest ratings were for involving the patient in decisions as much as he or she wanted (53%), encouraging the patient to ask questions (53%), and greeting the patient in a way that made him or her feel comfortable (55%). Table 2 contains a full ranking of individual item scores.

| Characteristics | |
|---|---|
| Patients (n =700), n (%) | |
| Sex, female | 378 (54) |
| Age, years | |
| 44 and younger | 189 (27) |
| 45‐64 | 266 (38) |
| 65 and older | 245 (35) |
| Race | |
| Caucasian | 357 (51) |
| African American | 266 (38) |
| Hispanic | 49 (7) |
| Other | 28 (4) |
| Hospitalists (n = 35), n (%) | |
| Sex, female | 18 (51) |
| Age, years | |
| Range | 3039 |
| Mean (SD) | 33 (2.4) |
| Race | |
| Caucasian | 14 (40) |
| South Asian | 11 (31) |
| Asian | 7 (20) |
| African American | 3 (9) |
| Non‐native English speaker | 5 (14) |
| Foreign medical graduate | 3 (9) |
| Communication Assessment Tool Item | Percent Excellent Scores |
|---|---|
| 1. Greeted me in a way that made me feel comfortable | 54.9 |
| 2. Treated me with respect | 66.3 |
| 3. Showed interest in my ideas about my health | 58.2 |
| 4. Understood my main health concerns | 57.4 |
| 5. Paid attention to me (looked at me, listened carefully) | 64.1 |
| 6. Let me talk without interruptions | 66.3 |
| 7. Gave me as much information as I wanted | 56.0 |
| 8. Talked in terms I could understand | 64.2 |
| 9. Checked to be sure I understood everything | 57.1 |
| 10. Encouraged me to ask questions | 53.2 |
| 11. Involved me in decisions as much as I wanted | 52.9 |
| 12. Discussed next steps including any follow‐up plans | 58.2 |
| 13. Showed care and concern | 63.8 |
| 14. Spent the right amount of time with me | 57.0 |
Overall scale reliability proved to be high (Cronbach's alpha = 0.97) in this sample. The factor analysis showed that scores for each of the 14 items load onto 1 factor. These results are consistent with the high reliability and single‐factor loading found in Makoul's original scale reliability and validity testing.17
The ad hoc comments made by patients at the conclusion of the CAT survey were categorized as positive or negative. Although many positive comments were made, they tended to be general in nature (eg, She is a great doctor). Negative comments were more explicit. A total of 110 patients (16%) made specific negative comments, which fell into 7 general domains: lack of information (35 comments), not enough time spent with the patient (27 comments), poor listening to the patient (24 comments), ineffective care delivery (7 comments), issues of care, concern, and respect (6 comments), ineffective communication with other staff (5 comments), and unclear role of physician (3 comments). Three patient comments were not related to these domains.
Patient age, race or gender did not correlate with CAT results. Hospitalist factors of age, race, gender, years of experience also were not associated with differences in ratings. However, race concordance between the patient and hospitalist was associated with improved CAT ratings. Patients of the same race as their hospitalist rated the hospitalist's communication significantly higher (M = 64.9%, SD = 39.1) than did patients who were of a different race than their hospitalist (M = 57.3%, SD = 40.3), P < 0.05. Gender concordance was not associated with improved CAT ratings. No score differences were found between patients cared for by a hospitalist on teaching service and direct care, and there were no differences between nursing units.
Discussion
To the best of our knowledge, this is the first study to explicitly measure patient perceptions of communication with hospitalists. The results yielded a wide distribution of scores for physicians within a single, large hospital medicine group. Comparing their own scores to those of peers may allow low‐scoring hospitalists to grasp the potential for improving their communication with patients. Our reliability testing matched the results of the original development study,17 indicating very high overall scale reliability. This suggests that the CAT could be streamlined by dropping some of the survey items. However we agree with Makoul et al.17 that it is best to keep the full set as it provides specific information for physicians without placing undue burden on patients (ie, the CAT takes only 1‐2 min to complete). Individual item scores for each of the 14 CAT items highlight specific communication tasks where intervention may be targeted for individual hospitalists and the group as a whole. It may be feasible to utilize CAT results as an individual report card for physicians. While program leaders should be aware that implementation of the CAT requires standardized data collection, it may be possible to build this into existing structures such as the discharge process.
Interestingly, many patients could not recognize the hospitalist caring for them by name or photo. More than 1 in 10 patients (107 of 828; 13%) were unable to identify their hospitalist. This was more than 3 times as common on the teaching service, where the hospitalist is accompanied by house staff and the intern or resident is the primary physician for patient contact, compared to the service on which hospitalists directly take care of patients without residents. It is also troubling that another 3% of patients (21 of 828) stated they hadn't talked enough with their hospitalist to answer basic communication questions, when approached 2 or 3 days into the relationship. It may be telling that Greeted me in a way that made me feel comfortable was one of the lowest‐rated survey items. Hospitalists should recognize that patients, in addition to facing their own physical and emotional stressors, see many hospital staff members throughout the day; all of whom may be strangers to them. Thus it becomes vital for hospitalists to not only establish an initial rapport with the patient, but to reintroduce themselves each time they enter the room.
An examination of the ad hoc negative comments made by survey respondents reinforces and extends findings related to the CAT items, particularly about those areas of communication valued by patients. The majority of comments fell into categories of failing to give enough information (eg, Sometimes I was left confused when the doctor was ready to leave), not spending enough time with the patient (eg, He was just in and out), and not listening to the patient's own ideas (eg, When giving my history, she cut me off at some points when I had more to say). The information and time categories may directly relate to scores on the CAT items Gave me as much information as I wanted and Spent the right amount of time with me, which are among the lowest‐scoring items. Listening to the patient may reflect broader issues of considering the patient's own experience, questions, concerns and goals.
In this study, patient‐physician race concordance was associated with CAT ratings. Patients who were of the same race as their hospitalist rated the hospitalist higher compared to patients who were of a different race than their hospitalist. This effect is consistent with previous research describing higher patient ratings of communication and care when the patient and physician are of the same race or ethnicity.21
A number of factors limit interpretation of the results of this study. The data were collected at a single site, thus limiting generalizability to other hospitalist practice environments. We used a retrospective, patient assessment of hospitalist communication which may have inherent biases different from a study using direct researcher observation or recording of patient‐hospitalist interactions to assess communication. This methodology allowed us to examine the patient's own perceptions and expectations of communication, but certainly leaves room for selection bias in recruitment and recall bias. Patients were interviewed on the second or third day of their admission. This controlled the length of exposure to the hospitalist, but the course of treatment might vary considerably; at the time of interview, some patients may not yet have had a clear diagnosis and plan while others may have been ready for discharge. Future work should examine how stage of evaluation and management might affect patients' perception of communication with hospitalists. Severity of condition is another factor that may affect patients' ratings, and was not examined in this study.
When compared to physicians from the CAT development study's field test, this study sample of hospitalists scored much lower, 59.1% excellent vs. 76.3% (P < 0.001). A number of factors may account for some of these differences. The majority of patients in the original field test had multiple interactions with their physician, and rated their health status as good or very good. In contrast, hospitalized patients usually lack previous exposure to the hospitalist, and likely have poorer health status. Also, physicians in the original field test volunteered to participate, and patients completed the CAT survey through the Internet or phone response system, rather than through a face‐to‐face interview by trained research assistants. Another key difference is that field‐test patients answered the CAT within 1 day of their outpatient visit, while in this study patients were interviewed in the midst of their hospital admission and prior to completion of their hospital course. Finally, patients commonly choose their outpatient physician and can select someone else if dissatisfied with their communication skills, while hospitalized patients are assigned hospitalists based on availability. Thus, given this potential selection bias, outpatients could be expected to rate their personal physician higher.
Another possibility is that hospitalists are on average less skilled in patient communication than outpatient physicians. Given the transient nature of the inpatient relationship, hospitalists may not value developing rapport with patients, and may not make this a goal of patient care or seek extensive training in communication skills. In future research, evaluating hospitalists' training in and attitudes towards patient communication could be paired with communication assessment results.
Although it is beyond the scope of this study to assess precisely how these environmental and survey implementation factors may affect CAT summary scores, their importance is evident. Another hospital‐based implementation of the CAT tool, an evaluation of ED teams,18 utilized face‐to‐face interviews with trained research assistants. The study yielded results similar to our findings: the average percent excellent score for ED teams was 62.3%, vs. 58.2% percent excellent for our hospitalist group. Taken together, these study comparisons between the original field‐test, our hospitalist implementation, and the ED team implementation support the argument that factors of setting (inpatient vs. outpatient), mode of survey administration (face‐to‐face interview vs. self‐administration through phone or Internet), and shorter duration or course of patient‐physician interaction may be important considerations when implementing the CAT tool to assess physician communication skills, or attempting to set standards of minimally acceptable or desired scoring.
More work must be done to establish norms and/or minimally acceptable scores for hospitalists. Numerous factors of specialty, practice setting, survey implementation, patient variables, and even the expertise of who is setting the communication standards22 may strongly influence comparisons between physician groups, even within a single institution. Organizations seeking to establish norms or minimally acceptable scores for physician‐patient communication should be aware of these factors. As the original development study points out, standard‐setting studies could establish specialty‐specific and country‐specific norms as well as norms or standards for level‐of‐training (eg, medical students versus attending physicians).17
Conclusion
The previously validated CAT instrument appears to have reliable test characteristics and can be used to gauge patient perceptions of hospitalist communication skills. Comparative scores between physicians of different specialties and settings should be interpreted cautiously as there may be confounding variables. Within our single institution, comparative scores between hospitalists, along with an examination of the hospitalist's individual item scores, may offer useful feedback for efforts aimed at enhancing communication. Many hospitalists in this study may benefit from targeted training to improve patient communication skills, particularly in the areas of encouraging questions and involving patients in decision making. Future qualitative research in the context of hospital medicine could identify specific communication techniques used by highly‐rated physicians, with the goal of developing tools for targeted improvement and determining impact on outcomes.
Acknowledgements
The authors thank Christie Edwards, Rachel Grayer and Caitlin Lawes for assistance with data collection, and Jie Peng for help with the analysis.
- ,,.Is the quality of the patient‐provider relationship associated with better adherence and health outcomes for patients with HIV?J Gen Intern Med.2006;21(6):661–665.
- ,,,,.Does physician communication influence older patients' diabetes self‐management and glycemic control? Results from the Health and Retirement Study (HRS).J Gerontol A Biol Sci Med Sci.2007;62(12):1435–1442.
- ,.Physician communication and patient adherence to treatment: a meta‐analysis.Med Care.2009;47(8):826–834.
- ,.The effect of physician behavior on the collection of data.Ann Intern Med.1984;101(5):692–696.
- ,,,,.Does physician‐patient communication that aims at empowering patients improve clinical outcome? A case study.Patient Educ Couns.2006;61(2):299–306.
- .Effective physician‐patient communication and health outcomes: a review.CMAJ.1995;152(9):1423–1433.
- .Evaluation of clinical competencies: basic certification, subspecialty certification, and recertification.Am J Phys Med Rehabil.2000;79(5):478–480.
- ,,,,.General competencies and accreditation in graduate medical education.Health Aff (Millwood).2002;21(5):103–111.
- Joint Commission on Accreditation of Healthcare Organizations. The Joint Commission Standards supporting effective communication, cultural competence, and patient‐centered care.2009:44.
- .Rapport and the hospitalist.Am J Med.2001;111(9B):31S–35S.
- ,.The emerging role of “hospitalists” in the American health care system.N Engl J Med.1996;335(7):514–517.
- ,,,.Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians.Ann Intern Med.1999;130(4 Pt 2):343–349.
- ,,,.The status of hospital medicine groups in the United States.J Hosp Med.2006;1:75–80.
- ,,,.Growth in the care of older patients by hospitalists in the United States.N Engl J Med.2009;360(11):1102–1112.
- ,,,,.Communication problems for patients hospitalized with chest pain.J Gen Intern Med.1998;13(12):836–838.
- ,.End‐of‐life care in a voluntary hospitalist model: effects on communication, processes of care, and patient symptoms.Am J Med.2004;116(10):669–675.
- ,,.Measuring patient views of physician communication skills: development and testing of the Communication Assessment Tool.Patient Educ Couns.2007;67(3):333–342.
- ,,, et al.Patient perspectives on communication with the medical team: pilot study using the Communication Assessment Tool‐Team (CAT‐T).Patient Educ Couns.2008;73(2):220–223.
- .Understanding resistance to the data‐model relationship in Rasch's paradigm: a reflection for the next generation.J Appl Meas.2002;3(3):325–359.
- ,.Conditional pairwise estimation in the Rasch model for ordered response categories using principal components.J Appl Meas.2003;4(3):205–221.
- ,,,,,.Patient‐centered communication, ratings of care, and concordance of patient and physician race.Ann Intern Med.2003;139(11):907–915.
- ,,,.The impact of judge selection on standard setting for a patient survey of physician communication skills.Acad Med.2008;83(10 Suppl):S17–S20.
- ,,.Is the quality of the patient‐provider relationship associated with better adherence and health outcomes for patients with HIV?J Gen Intern Med.2006;21(6):661–665.
- ,,,,.Does physician communication influence older patients' diabetes self‐management and glycemic control? Results from the Health and Retirement Study (HRS).J Gerontol A Biol Sci Med Sci.2007;62(12):1435–1442.
- ,.Physician communication and patient adherence to treatment: a meta‐analysis.Med Care.2009;47(8):826–834.
- ,.The effect of physician behavior on the collection of data.Ann Intern Med.1984;101(5):692–696.
- ,,,,.Does physician‐patient communication that aims at empowering patients improve clinical outcome? A case study.Patient Educ Couns.2006;61(2):299–306.
- .Effective physician‐patient communication and health outcomes: a review.CMAJ.1995;152(9):1423–1433.
- .Evaluation of clinical competencies: basic certification, subspecialty certification, and recertification.Am J Phys Med Rehabil.2000;79(5):478–480.
- ,,,,.General competencies and accreditation in graduate medical education.Health Aff (Millwood).2002;21(5):103–111.
- Joint Commission on Accreditation of Healthcare Organizations. The Joint Commission Standards supporting effective communication, cultural competence, and patient‐centered care.2009:44.
- .Rapport and the hospitalist.Am J Med.2001;111(9B):31S–35S.
- ,.The emerging role of “hospitalists” in the American health care system.N Engl J Med.1996;335(7):514–517.
- ,,,.Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians.Ann Intern Med.1999;130(4 Pt 2):343–349.
- ,,,.The status of hospital medicine groups in the United States.J Hosp Med.2006;1:75–80.
- ,,,.Growth in the care of older patients by hospitalists in the United States.N Engl J Med.2009;360(11):1102–1112.
- ,,,,.Communication problems for patients hospitalized with chest pain.J Gen Intern Med.1998;13(12):836–838.
- ,.End‐of‐life care in a voluntary hospitalist model: effects on communication, processes of care, and patient symptoms.Am J Med.2004;116(10):669–675.
- ,,.Measuring patient views of physician communication skills: development and testing of the Communication Assessment Tool.Patient Educ Couns.2007;67(3):333–342.
- ,,, et al.Patient perspectives on communication with the medical team: pilot study using the Communication Assessment Tool‐Team (CAT‐T).Patient Educ Couns.2008;73(2):220–223.
- .Understanding resistance to the data‐model relationship in Rasch's paradigm: a reflection for the next generation.J Appl Meas.2002;3(3):325–359.
- ,.Conditional pairwise estimation in the Rasch model for ordered response categories using principal components.J Appl Meas.2003;4(3):205–221.
- ,,,,,.Patient‐centered communication, ratings of care, and concordance of patient and physician race.Ann Intern Med.2003;139(11):907–915.
- ,,,.The impact of judge selection on standard setting for a patient survey of physician communication skills.Acad Med.2008;83(10 Suppl):S17–S20.
Copyright © 2010 Society of Hospital Medicine
Elements of Confusion
A 23‐year‐old man presented to his family physician's office with a 2‐week history of fever, chills, night sweats, anorexia, and fatigue. This was associated with a 4‐month history of a nonproductive cough and a 20‐pound involuntary weight loss. He denied shortness of breath, chest pain, headaches, abdominal pain, vomiting, diarrhea, dysuria, and rash. There was no recent travel, sick contacts, or animal exposures.
This patient's symptoms could represent an underlying infectious, neoplastic, or inflammatory process. I would ascertain any relevant personal or family history and explore whether the patient has risk factors for human immunodeficiency virus (HIV) infection or tuberculosis (TB). On physical examination, I would listen for a heart murmur and look for lymphadenopathy, hepatosplenomegaly, and arthritis. Investigations including cultures, urinalysis, and a chest radiograph would be indicated at this time.
During the 2 weeks after his initial presentation, he experienced persistent fever, and further weight loss. He was admitted to the hospital to determine the etiology of his symptoms. The patient had no previous medical problems. On initial examination, his temperature was 102 degrees Fahrenheit, blood pressure was 100/65 mmHg, heart rate was 105 per minute, respiratory rate was 22 breaths per minute and oxygen saturation was normal on ambient air. He appeared cachectic. He was oriented to person, place, and time. Head and neck examination revealed no intraoral pathology, lymphadenopathy or scleral icterus, but did reveal conjunctival pallor. The chest was clear to auscultation, and the cardiovascular examination revealed a normal apical impulse and heart sounds with no murmurs. There was peripheral edema to the level of the mid‐shins bilaterally. The abdomen was soft and non‐tender with no appreciable hepatosplenomegaly. There were no stigmata of chronic liver disease. There was no axillary or inguinal lymphadenopathy. The remainder of the examination was normal. A complete blood count showed a hemoglobin concentration of 5.2 g/dL with a mean corpuscular volume (MCV) of 89fL, white blood cells were 1,400 cells/mm3 with an absolute neutrophil count (ANC) of 800 cells/mm3 and a platelet count of 90,000 cells/mm3 The serum sodium was 124 mmol/L, potassium 3.0 mmol/L, chloride 91 mmol/L, bicarbonate 26 mmol/L, and the creatinine 1.36 mg/dL His liver enzyme profile showed aspartate aminotransferase (AST) 68 U/L (normal 35), alanine aminotransferase (ALT) 25 U/L (normal 40), alkaline phosphatase (ALP) 210 U/L (normal 110) and a total bilirubin of 1.64 mg/dL.
The patient is clearly very unwell and requires admission to the hospital for treatment and further investigation. Emergent management includes administration of intravenous fluids to correct his electrolyte abnormalities, empiric broad spectrum antibiotics (given his relative neutropenia and fever), and a transfusion for his profound anemia. I would be very concerned that he has an underlying malignancy such as lymphoma or leukemia. Pancytopenia related to decreased cell production may be secondary to infiltration (malignant or granulomatous), infection (HIV, TB, fungal, viral), or aplasia (primary or drug‐related). Less likely etiologies include B12 or folate deficiency (unlikely given the normal MCV), systemic lupus erythematosus, paroxysmal nocturnal hemoglobinuria or cell sequestration due to hypersplenism. A history of recent exposure to drugs or toxins should be elicited. The patient's pulmonary symptoms may relate to the primary disorder or may represent an infection secondary to myelosuppression. I would want an immediate review of the peripheral blood smear, a hemolysis work‐up (drawn prior to transfusion including lactate dehydrogenase [LDH], haptoglobin, fractionated bilirubin, reticulocyte count and direct antiglobulin testing), antinuclear antibody (ANA), B12 and folate levels, imaging of the chest and blood cultures.
With evidence of fever and pancytopenia, acute leukemia was suspected and the patient was admitted to a hematology service. Over the next two weeks an extensive investigation including blood and urine cultures, and computed tomograms (CT) of the chest and abdomen were performed. A bone marrow aspirate and biopsy were also were done and were submitted for histopathologic examination and culture. The CT scan of the chest revealed left axillary and supraclavicular lymphadenopathy (Figure 1), and the abdominal imaging revealed splenomegaly. The blood, urine and bone marrow cultures were all negative. A peripheral blood smear showed pancytopenia with a hematologist interpretation suggesting that an intrinsic bone marrow process may be resulting in impaired cell production. The corresponding bone marrow biopsy and aspirate showed no evidence of malignancy, but there were numerous granulomata, and the periodic acid‐Schiff (PAS) and silver staining showed cells that resembled fungal elements (Figure 2).
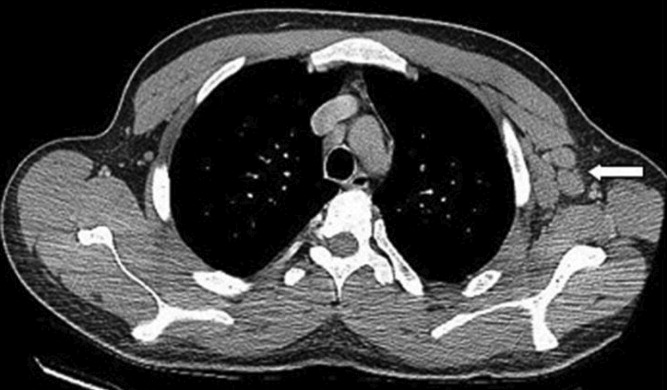
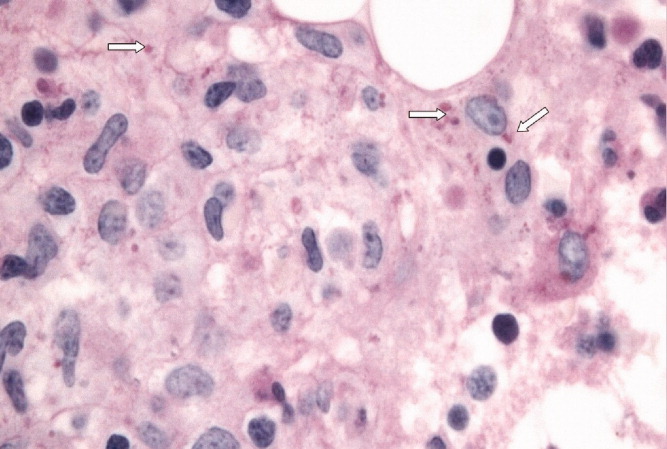
The absence of malignant cells in the bone marrow leaves us to consider infectious and inflammatory causes of this patient's presentation. Infectious etiologies associated with bone marrow granulomata include fungal, mycobacterial, bacterial (brucellosis, typhoid and Q fever) and viral pathogens including HIV, Epstein‐Barr virus (EBV), and cytomegalovirus (CMV). Noninfectious causes include sarcoidosis, drug effects, and autoimmune conditions. The PAS and silver staining suggests this patient has a disseminated fungal infection. Disseminated Histoplasma capsulatum is the most likely organism but blastomycosis and coccidioidomycosis should be considered. HIV and occult lymphoma are considerations as is a primary immune disorder such as common variable immunodeficiency (CVID) which can present in this age group. While there is no recent travel history, it will be critical to determine where the patient currently lives and previously resided, review the medical record for prior infections and HIV risk factors, and take a thorough occupational history.
At this point, the following investigations should be undertaken: blood, sputum, and bone marrow culture; fungal and acid‐fast bacilli (AFB) stains on sputum and bone marrow; Histoplasma urine antigen; tuberculin skin test; serology for HIV and histoplasmosis; and serum protein electrophoresis with immunofixation and quantitation of immunoglobulins.
Acid fast staining of the bone marrow as well as mycobacterial and fungal cultures were negative. He lived in eastern Ontario and worked in construction. He reported helping tear down an old cabin in a wooded area, but denied any insect bites. This project coincided with the onset of his cough. He had no history of high risk sexual activity, intravenous drug use, tattoos or blood transfusions previous to his presentation. The HIV test was negative. His clinicians at this point considered a disseminated fungal infection as a cause for his symptoms and started him empirically on itraconazole He was discharged from the hospital with a plan for close outpatient followup. Within three days of discharge on the itraconazole, the patient's fever began to diminish, but did not completely resolve.
The clinical picture including cough, geography, and recent occupational exposure is entirely consistent with disseminated histoplasmosis. However, we are still lacking microbiologic confirmation of the diagnosis. Sarcoidosis and occult malignancy must still be considered. In the absence of a definitive diagnosis, I would consider bronchoscopy with bronchoalveolar lavage (BAL) and obtaining a lymph node or liver biopsy for microbiologic and pathologic examination. With the patient now receiving antifungal therapy, a diagnosis of histoplasmosis would be supported by a response to therapy, declining Histoplasma antigen levels and clinical improvement including recovery of his bone marrow.
The urine specimen was negative for Histoplasma antigen. Seven days after initiating itraconazole, he developed jaundice and confusion and was taken back to the hospital. On presentation, he was disoriented but awake. His temperature was 103.1 degrees Fahrenheit, blood pressure was 90/60 mm Hg, heart rate was 115 per minute, and oxygen saturation was normal on room air. He was obviously jaundiced, and more cachectic than previous. The neurologic examination demonstrated disorientation with no localizing findings. The chest and cardiovascular examinations were normal. His abdomen was soft and non‐tender with no evidence of hepatomegaly, but the spleen tip was palpable. There was no ascites or any other signs of portal hypertension, but his peripheral edema was worse than before and asterixis was present. The remainder of the examination was unchanged from previous. His laboratory investigations at this point showed a bilirubin of 18.5 mg/dL, AST 269 U/L, ALT 76 U/L, ALP 165 U/L, albumin 18 g/L, fibrinogen 1.53 g/L (normal 1.5‐3.5), triglycerides 2.4 mmol/L (normal 2), ferritin 59415 ug/L (normal 22‐275) an international normalized ratio (INR) of 2.65. His complete blood count still showed pancytopenia.
The patient has now developed fulminant hepatic failure. He requires volume resuscitation, drawing of repeat cultures, initiation of empiric broad spectrum antibiotics, urgent hepatology consultation and intensive care unit (ICU) support. The most common causes of acute liver failure are drug toxicity (including acetaminophen), viral hepatitis, Wilson's disease, Budd‐Chiari syndrome, cryptogenic liver disease and fatty infiltration. The critical diagnostic issue at this point is to determine if the liver failure is a secondary process (in which case drug toxicity due to itraconazole would be the most likely cause) or if this represents evolution of his primary disease with extensive hepatic involvement. Liver failure due to itraconazole has been reported and given the lack of microbiologic confirmation of a fungal infection, this agent should clearly be discontinued. Returning to our initial differential diagnosis of this man's granulomatous bone marrow infiltration and pancytopenia, etiologies which may progress to hepatic failure include viral infections (EBV or CMV) and malignancy. This patient's presentation could be an unusual manifestation of a common illness such as EBV or a rapidly progressive lymphoma. An abdominal Doppler ultrasound is required to rule out Budd‐Chiari syndrome. Given his abrupt change in clinical status, I would repeat a CT scan of his chest and abdomen to evaluate for evidence of infection, infiltration, or malignancy. Owing to the uncertainty regarding this patient's diagnosis and the rapidly progressive nature of his disease, serious consideration must be given to a transjugular liver biopsy.
Soon after admission, he developed hematemesis. He was given multiple blood transfusions, and then intravenous fluids, broad spectrum antibiotics and lactulose. Upper gastrointestinal endoscopy showed no varices, but did reveal multiple esophageal and gastric ulcerations. He was then transferred to a liver transplant center where repeat bone marrow biopsy and a liver biopsy were done. Both revealed extensive granulomatosis and the bone marrow biopsy showed evidence of hemophagocytosis (Figure 3).
The finding of hemophagocytosis in the setting of fever, hepatosplenomegaly, and pancytopenia is consistent with a diagnosis of hemophagocytic lymphohistiocytosis (HLH). The cornerstone of therapy for patients with HLH is suppression of the severe inflammatory response with corticosteroids, etoposide and cyclosporin. Patients who respond to this are candidates for allogeneic stem cell transplant with curative intent. This patient's hepatic dysfunction precludes the use of etoposide and initial therapy should therefore include dexamethasone and cyclosporin.
All bacterial, fungal, and mycobacterial cultures again demonstrated no growth. Broad spectrum antibiotics were continued, and empiric intravenous amphotericin B was added. He became hemodynamically unstable, was intubated, put on mechanical ventilation and required vasoactive medications to maintain his blood pressure. An empiric course of pulse corticosteroids was given for the possibility of sarcoidosis. His blood pressure stabilized, though he continued to require vasopressors.
While HLH has been very rarely reported in association with sarcoidosis, the underlying pathogenesis of his clinical presentation (infectious, neoplastic, or inflammatory) has not yet been confirmed. In the meantime, I would continue with supportive care and intravenous corticosteroids.
Immunohistochemical studies of the liver biopsy returned showing CD15/30+ cells with weak‐to‐negative CD45 expression cells typical of Hodgkin lymphoma (HL) (Figure 4). He was started on chemotherapy, but over 48 hours became progressively more hypotensive. The patient died of Klebsiella and Pseudomonas sepsis on the 7th hospital day. Post‐mortem immunohistochemical examination revealed evidence of Hodgkin disease in the axillary lymph nodes, bone marrow and liver. The bone marrow showed evidence of hemophagocytosis and was also positive for Epstein‐Barr encoded RNA (EBER). Serologic studies were subsequently available and revealed positive EBV IgM against the viral capsid antigen (VCA) as well as EBV IgG VCA, which in conjunction with the marrow findings, was highly suggestive of reactivation EBV disease.
Discussion
This patient's diagnostic course led both the clinical team and discussant down a winding path, which ultimately ended in the finding of Hodgkin lymphoma, a relatively common diagnosis that had been clouded by seemingly contradictory clinical and laboratory data. The provisional diagnosis of disseminated histoplasmosis was reasonable given that H. capsulatum is endemic in Ontario and that the patient's occupation placed him at risk of infection. Given the acuity of his illness, empiric antifungal therapy based on the report of fungal elements on bone marrow examination seemed reasonable. However, Histoplasma urinary antigen testing has been shown in the literature to be 98% sensitive in immunosuppressed populations, and the negative result prompted a re‐examination of the marrow specimen. The previously described fungal elements were felt to be most likely artifact, and the underlying diagnosis was reconsidered.1 This is when the repeat bone marrow examination pointed towards the diagnosis of HLH.
Hemophagocytic lymphohistiocytosis (HLH) is a severe, systemic hyperinflammatory disorder characterized by histiocytic proliferation that may be primary or can be triggered by infection, connective tissue diseases or malignancy.25 The central pathogenesis involves dysregulated Th1 cytokine secretion. This results in an uncontrolled accumulation of activated T‐lymphocytes and histiocytes in various organs including the liver, spleen and bone marrow. The infiltration of histiocytes into major organs can lead to disruption of function and multiorgan failure.6 Viruses are the most common infectious triggers of HLH, particularly EBV, and lymphoma is the most common associated malignancy.25 It is hypothesized that EBV can interfere with normal lymphocyte signaling pathways leading to the aforementioned over‐expression of Th1 cytokines, which can then trigger HLH.7 The diagnosis of HLH is based on a combination of clinical and laboratory parameters as outlined in Table 1.8 Our patient met all five of the major criteria.
| |
| Major criteria | 1. Fever |
| 2. Splenomegaly | |
| 3. Cytopenia in two or more cell lines | |
| 4. Hypertriglyceridemia or hypofibrinogenemia | |
| 5. Hemophagocytosis on histopathologic examination | |
| Alternative criteria | A. Low or absent natural killer cell activity |
| B. Serum ferritin level >500 ug/L | |
| C. Soluble CD‐25 level >2400 U/mL | |
The recommended treatment of HLH involves the administration of the HLH‐94 protocol consisting of corticosteroids, cyclosporine and etoposide.9, 10 Those who survive this initial phase are recommended for hematopoetic stem cell transplantation producing an overall 3‐year survival rate of 64%. However, those who do not receive early etoposide therapy fare much worse, with a mortality rate of 92%.10 This patient was not able to receive etoposide because of his decompensated liver disease.
In this case, the development of Hodgkin lymphoma involving the bone marrow and liver may have resulted in a state of immune suppression. The loss of immune function likely allowed the reactivation of Epstein‐Barr virus which triggered HLH and his fulminant presentation.35,9 Indeed, both the liver and bone marrow samples showed evidence of EBV reactivation as evidenced by the presence of EBER. The diagnosis of Hodgkin lymphoma was made from a liver biopsy specimen rather than bone marrow examination. The diagnosis of Hodgkin lymphoma is based on the presence of Reed‐Sternberg cells surrounded by an inflammatory milieu of cells including variable numbers of small lymphocytes, neutrophils eosinophils and fibroblasts. The HLH‐induced pancytopenia depleted the aforementioned inflammatory milieu in the bone marrow, which obscured the diagnosis of Hodgkin lymphoma. Unfortunately, lymphoma‐associated HLH has a very poor prognosis with a mortality rate of up to 60%.4 At the outset of this case, the reported fungal elements proved to be a source of confusion which delayed the diagnosis of Hodgkin lymphoma. Given the poor prognosis of lymphoma‐associated HLH, it is unlikely this would have had any effect on the ultimate outcome.
Teaching Points
-
HLH is a rare and complex hyperinflammatory disorder which may present as pancytopenia.
-
Triggers of HLH can include infections (particularly EBV), malignancy (particularly lymphoma) and connective tissue diseases.
-
The diagnosis of HLH is based on clinical and laboratory criteria, including cytopenias that may make the evaluation for triggering conditions such as HL more difficult.
-
Lymphoma should be included in the differential diagnosis of granulomatous inflammation.
Acknowledgements
The authors acknowledge Ralph Meyer, MD (Queen's University, Kingston, Ontario) for his comments on a draft of this paper and Drs. David Barth and Maha Guindi (Department of Laboratory Medicine, University Health Network) for their reviews of the pathology specimens.
- ,Antigen detection for diagnosis of the endemic mycoses.Curr Fung Infect Rep.2008;4:189–193.
- ,,, et al.Lymphoma‐associated hemophagocytic syndrome: Clinical features and treatment outcome.Ann Hematol.2007;86:493–498.
- ,,, et al.Hodgkin lymphoma‐associated hemophagocytic syndrome: A disorder strongly correlated with Epstein‐Barr virus.Clin Inf Dis.2008;47:531–534.
- ,.Hemophagocytic syndrome associated with Hodgkin lymphoma and pneumocystis jiroveci pneumonitis.Br J Hematol.2007;138:672.
- ,,, et al.Infections associated with haemophagocytic syndrome.Lancet Infect Dis.2007;12:814–822.
- ,,,.Hemophagocytic lymphohistiocytic syndrome: Unrecognized cause of multiple organ failure.Pediatr Crit Care Med.2000;1:51–54.
- ,,, et al.Epstein‐Barr virus LMP1 Inhibits the expression of SAP gene and upregulates Th1 cytokines in the pathogenesis of hemophagocytic syndrome.Blood.2005;106:3090–3096.
- ,,.Diagnostic guidelines for hemophagocytic lymphohistiocytosis.Semin Oncol.1991;18:29–33
- ,,, et al.Treatment of Epstein‐Barr virus‐associated hemophagocytic lymphohistiocytosis (EBV‐HLH) in young adults: A report from the HLH Study Center.Med Pediatr Oncol.2003;41:103–109.
- ,,, et al.Haematopoietic stem cell transplantation in haemophagocytic lymphohistiocytosis.Br J Haematol.2005;129:622–630.
A 23‐year‐old man presented to his family physician's office with a 2‐week history of fever, chills, night sweats, anorexia, and fatigue. This was associated with a 4‐month history of a nonproductive cough and a 20‐pound involuntary weight loss. He denied shortness of breath, chest pain, headaches, abdominal pain, vomiting, diarrhea, dysuria, and rash. There was no recent travel, sick contacts, or animal exposures.
This patient's symptoms could represent an underlying infectious, neoplastic, or inflammatory process. I would ascertain any relevant personal or family history and explore whether the patient has risk factors for human immunodeficiency virus (HIV) infection or tuberculosis (TB). On physical examination, I would listen for a heart murmur and look for lymphadenopathy, hepatosplenomegaly, and arthritis. Investigations including cultures, urinalysis, and a chest radiograph would be indicated at this time.
During the 2 weeks after his initial presentation, he experienced persistent fever, and further weight loss. He was admitted to the hospital to determine the etiology of his symptoms. The patient had no previous medical problems. On initial examination, his temperature was 102 degrees Fahrenheit, blood pressure was 100/65 mmHg, heart rate was 105 per minute, respiratory rate was 22 breaths per minute and oxygen saturation was normal on ambient air. He appeared cachectic. He was oriented to person, place, and time. Head and neck examination revealed no intraoral pathology, lymphadenopathy or scleral icterus, but did reveal conjunctival pallor. The chest was clear to auscultation, and the cardiovascular examination revealed a normal apical impulse and heart sounds with no murmurs. There was peripheral edema to the level of the mid‐shins bilaterally. The abdomen was soft and non‐tender with no appreciable hepatosplenomegaly. There were no stigmata of chronic liver disease. There was no axillary or inguinal lymphadenopathy. The remainder of the examination was normal. A complete blood count showed a hemoglobin concentration of 5.2 g/dL with a mean corpuscular volume (MCV) of 89fL, white blood cells were 1,400 cells/mm3 with an absolute neutrophil count (ANC) of 800 cells/mm3 and a platelet count of 90,000 cells/mm3 The serum sodium was 124 mmol/L, potassium 3.0 mmol/L, chloride 91 mmol/L, bicarbonate 26 mmol/L, and the creatinine 1.36 mg/dL His liver enzyme profile showed aspartate aminotransferase (AST) 68 U/L (normal 35), alanine aminotransferase (ALT) 25 U/L (normal 40), alkaline phosphatase (ALP) 210 U/L (normal 110) and a total bilirubin of 1.64 mg/dL.
The patient is clearly very unwell and requires admission to the hospital for treatment and further investigation. Emergent management includes administration of intravenous fluids to correct his electrolyte abnormalities, empiric broad spectrum antibiotics (given his relative neutropenia and fever), and a transfusion for his profound anemia. I would be very concerned that he has an underlying malignancy such as lymphoma or leukemia. Pancytopenia related to decreased cell production may be secondary to infiltration (malignant or granulomatous), infection (HIV, TB, fungal, viral), or aplasia (primary or drug‐related). Less likely etiologies include B12 or folate deficiency (unlikely given the normal MCV), systemic lupus erythematosus, paroxysmal nocturnal hemoglobinuria or cell sequestration due to hypersplenism. A history of recent exposure to drugs or toxins should be elicited. The patient's pulmonary symptoms may relate to the primary disorder or may represent an infection secondary to myelosuppression. I would want an immediate review of the peripheral blood smear, a hemolysis work‐up (drawn prior to transfusion including lactate dehydrogenase [LDH], haptoglobin, fractionated bilirubin, reticulocyte count and direct antiglobulin testing), antinuclear antibody (ANA), B12 and folate levels, imaging of the chest and blood cultures.
With evidence of fever and pancytopenia, acute leukemia was suspected and the patient was admitted to a hematology service. Over the next two weeks an extensive investigation including blood and urine cultures, and computed tomograms (CT) of the chest and abdomen were performed. A bone marrow aspirate and biopsy were also were done and were submitted for histopathologic examination and culture. The CT scan of the chest revealed left axillary and supraclavicular lymphadenopathy (Figure 1), and the abdominal imaging revealed splenomegaly. The blood, urine and bone marrow cultures were all negative. A peripheral blood smear showed pancytopenia with a hematologist interpretation suggesting that an intrinsic bone marrow process may be resulting in impaired cell production. The corresponding bone marrow biopsy and aspirate showed no evidence of malignancy, but there were numerous granulomata, and the periodic acid‐Schiff (PAS) and silver staining showed cells that resembled fungal elements (Figure 2).


The absence of malignant cells in the bone marrow leaves us to consider infectious and inflammatory causes of this patient's presentation. Infectious etiologies associated with bone marrow granulomata include fungal, mycobacterial, bacterial (brucellosis, typhoid and Q fever) and viral pathogens including HIV, Epstein‐Barr virus (EBV), and cytomegalovirus (CMV). Noninfectious causes include sarcoidosis, drug effects, and autoimmune conditions. The PAS and silver staining suggests this patient has a disseminated fungal infection. Disseminated Histoplasma capsulatum is the most likely organism but blastomycosis and coccidioidomycosis should be considered. HIV and occult lymphoma are considerations as is a primary immune disorder such as common variable immunodeficiency (CVID) which can present in this age group. While there is no recent travel history, it will be critical to determine where the patient currently lives and previously resided, review the medical record for prior infections and HIV risk factors, and take a thorough occupational history.
At this point, the following investigations should be undertaken: blood, sputum, and bone marrow culture; fungal and acid‐fast bacilli (AFB) stains on sputum and bone marrow; Histoplasma urine antigen; tuberculin skin test; serology for HIV and histoplasmosis; and serum protein electrophoresis with immunofixation and quantitation of immunoglobulins.
Acid fast staining of the bone marrow as well as mycobacterial and fungal cultures were negative. He lived in eastern Ontario and worked in construction. He reported helping tear down an old cabin in a wooded area, but denied any insect bites. This project coincided with the onset of his cough. He had no history of high risk sexual activity, intravenous drug use, tattoos or blood transfusions previous to his presentation. The HIV test was negative. His clinicians at this point considered a disseminated fungal infection as a cause for his symptoms and started him empirically on itraconazole He was discharged from the hospital with a plan for close outpatient followup. Within three days of discharge on the itraconazole, the patient's fever began to diminish, but did not completely resolve.
The clinical picture including cough, geography, and recent occupational exposure is entirely consistent with disseminated histoplasmosis. However, we are still lacking microbiologic confirmation of the diagnosis. Sarcoidosis and occult malignancy must still be considered. In the absence of a definitive diagnosis, I would consider bronchoscopy with bronchoalveolar lavage (BAL) and obtaining a lymph node or liver biopsy for microbiologic and pathologic examination. With the patient now receiving antifungal therapy, a diagnosis of histoplasmosis would be supported by a response to therapy, declining Histoplasma antigen levels and clinical improvement including recovery of his bone marrow.
The urine specimen was negative for Histoplasma antigen. Seven days after initiating itraconazole, he developed jaundice and confusion and was taken back to the hospital. On presentation, he was disoriented but awake. His temperature was 103.1 degrees Fahrenheit, blood pressure was 90/60 mm Hg, heart rate was 115 per minute, and oxygen saturation was normal on room air. He was obviously jaundiced, and more cachectic than previous. The neurologic examination demonstrated disorientation with no localizing findings. The chest and cardiovascular examinations were normal. His abdomen was soft and non‐tender with no evidence of hepatomegaly, but the spleen tip was palpable. There was no ascites or any other signs of portal hypertension, but his peripheral edema was worse than before and asterixis was present. The remainder of the examination was unchanged from previous. His laboratory investigations at this point showed a bilirubin of 18.5 mg/dL, AST 269 U/L, ALT 76 U/L, ALP 165 U/L, albumin 18 g/L, fibrinogen 1.53 g/L (normal 1.5‐3.5), triglycerides 2.4 mmol/L (normal 2), ferritin 59415 ug/L (normal 22‐275) an international normalized ratio (INR) of 2.65. His complete blood count still showed pancytopenia.
The patient has now developed fulminant hepatic failure. He requires volume resuscitation, drawing of repeat cultures, initiation of empiric broad spectrum antibiotics, urgent hepatology consultation and intensive care unit (ICU) support. The most common causes of acute liver failure are drug toxicity (including acetaminophen), viral hepatitis, Wilson's disease, Budd‐Chiari syndrome, cryptogenic liver disease and fatty infiltration. The critical diagnostic issue at this point is to determine if the liver failure is a secondary process (in which case drug toxicity due to itraconazole would be the most likely cause) or if this represents evolution of his primary disease with extensive hepatic involvement. Liver failure due to itraconazole has been reported and given the lack of microbiologic confirmation of a fungal infection, this agent should clearly be discontinued. Returning to our initial differential diagnosis of this man's granulomatous bone marrow infiltration and pancytopenia, etiologies which may progress to hepatic failure include viral infections (EBV or CMV) and malignancy. This patient's presentation could be an unusual manifestation of a common illness such as EBV or a rapidly progressive lymphoma. An abdominal Doppler ultrasound is required to rule out Budd‐Chiari syndrome. Given his abrupt change in clinical status, I would repeat a CT scan of his chest and abdomen to evaluate for evidence of infection, infiltration, or malignancy. Owing to the uncertainty regarding this patient's diagnosis and the rapidly progressive nature of his disease, serious consideration must be given to a transjugular liver biopsy.
Soon after admission, he developed hematemesis. He was given multiple blood transfusions, and then intravenous fluids, broad spectrum antibiotics and lactulose. Upper gastrointestinal endoscopy showed no varices, but did reveal multiple esophageal and gastric ulcerations. He was then transferred to a liver transplant center where repeat bone marrow biopsy and a liver biopsy were done. Both revealed extensive granulomatosis and the bone marrow biopsy showed evidence of hemophagocytosis (Figure 3).

The finding of hemophagocytosis in the setting of fever, hepatosplenomegaly, and pancytopenia is consistent with a diagnosis of hemophagocytic lymphohistiocytosis (HLH). The cornerstone of therapy for patients with HLH is suppression of the severe inflammatory response with corticosteroids, etoposide and cyclosporin. Patients who respond to this are candidates for allogeneic stem cell transplant with curative intent. This patient's hepatic dysfunction precludes the use of etoposide and initial therapy should therefore include dexamethasone and cyclosporin.
All bacterial, fungal, and mycobacterial cultures again demonstrated no growth. Broad spectrum antibiotics were continued, and empiric intravenous amphotericin B was added. He became hemodynamically unstable, was intubated, put on mechanical ventilation and required vasoactive medications to maintain his blood pressure. An empiric course of pulse corticosteroids was given for the possibility of sarcoidosis. His blood pressure stabilized, though he continued to require vasopressors.
While HLH has been very rarely reported in association with sarcoidosis, the underlying pathogenesis of his clinical presentation (infectious, neoplastic, or inflammatory) has not yet been confirmed. In the meantime, I would continue with supportive care and intravenous corticosteroids.
Immunohistochemical studies of the liver biopsy returned showing CD15/30+ cells with weak‐to‐negative CD45 expression cells typical of Hodgkin lymphoma (HL) (Figure 4). He was started on chemotherapy, but over 48 hours became progressively more hypotensive. The patient died of Klebsiella and Pseudomonas sepsis on the 7th hospital day. Post‐mortem immunohistochemical examination revealed evidence of Hodgkin disease in the axillary lymph nodes, bone marrow and liver. The bone marrow showed evidence of hemophagocytosis and was also positive for Epstein‐Barr encoded RNA (EBER). Serologic studies were subsequently available and revealed positive EBV IgM against the viral capsid antigen (VCA) as well as EBV IgG VCA, which in conjunction with the marrow findings, was highly suggestive of reactivation EBV disease.

Discussion
This patient's diagnostic course led both the clinical team and discussant down a winding path, which ultimately ended in the finding of Hodgkin lymphoma, a relatively common diagnosis that had been clouded by seemingly contradictory clinical and laboratory data. The provisional diagnosis of disseminated histoplasmosis was reasonable given that H. capsulatum is endemic in Ontario and that the patient's occupation placed him at risk of infection. Given the acuity of his illness, empiric antifungal therapy based on the report of fungal elements on bone marrow examination seemed reasonable. However, Histoplasma urinary antigen testing has been shown in the literature to be 98% sensitive in immunosuppressed populations, and the negative result prompted a re‐examination of the marrow specimen. The previously described fungal elements were felt to be most likely artifact, and the underlying diagnosis was reconsidered.1 This is when the repeat bone marrow examination pointed towards the diagnosis of HLH.
Hemophagocytic lymphohistiocytosis (HLH) is a severe, systemic hyperinflammatory disorder characterized by histiocytic proliferation that may be primary or can be triggered by infection, connective tissue diseases or malignancy.25 The central pathogenesis involves dysregulated Th1 cytokine secretion. This results in an uncontrolled accumulation of activated T‐lymphocytes and histiocytes in various organs including the liver, spleen and bone marrow. The infiltration of histiocytes into major organs can lead to disruption of function and multiorgan failure.6 Viruses are the most common infectious triggers of HLH, particularly EBV, and lymphoma is the most common associated malignancy.25 It is hypothesized that EBV can interfere with normal lymphocyte signaling pathways leading to the aforementioned over‐expression of Th1 cytokines, which can then trigger HLH.7 The diagnosis of HLH is based on a combination of clinical and laboratory parameters as outlined in Table 1.8 Our patient met all five of the major criteria.
| |
| Major criteria | 1. Fever |
| 2. Splenomegaly | |
| 3. Cytopenia in two or more cell lines | |
| 4. Hypertriglyceridemia or hypofibrinogenemia | |
| 5. Hemophagocytosis on histopathologic examination | |
| Alternative criteria | A. Low or absent natural killer cell activity |
| B. Serum ferritin level >500 ug/L | |
| C. Soluble CD‐25 level >2400 U/mL | |
The recommended treatment of HLH involves the administration of the HLH‐94 protocol consisting of corticosteroids, cyclosporine and etoposide.9, 10 Those who survive this initial phase are recommended for hematopoetic stem cell transplantation producing an overall 3‐year survival rate of 64%. However, those who do not receive early etoposide therapy fare much worse, with a mortality rate of 92%.10 This patient was not able to receive etoposide because of his decompensated liver disease.
In this case, the development of Hodgkin lymphoma involving the bone marrow and liver may have resulted in a state of immune suppression. The loss of immune function likely allowed the reactivation of Epstein‐Barr virus which triggered HLH and his fulminant presentation.35,9 Indeed, both the liver and bone marrow samples showed evidence of EBV reactivation as evidenced by the presence of EBER. The diagnosis of Hodgkin lymphoma was made from a liver biopsy specimen rather than bone marrow examination. The diagnosis of Hodgkin lymphoma is based on the presence of Reed‐Sternberg cells surrounded by an inflammatory milieu of cells including variable numbers of small lymphocytes, neutrophils eosinophils and fibroblasts. The HLH‐induced pancytopenia depleted the aforementioned inflammatory milieu in the bone marrow, which obscured the diagnosis of Hodgkin lymphoma. Unfortunately, lymphoma‐associated HLH has a very poor prognosis with a mortality rate of up to 60%.4 At the outset of this case, the reported fungal elements proved to be a source of confusion which delayed the diagnosis of Hodgkin lymphoma. Given the poor prognosis of lymphoma‐associated HLH, it is unlikely this would have had any effect on the ultimate outcome.
Teaching Points
-
HLH is a rare and complex hyperinflammatory disorder which may present as pancytopenia.
-
Triggers of HLH can include infections (particularly EBV), malignancy (particularly lymphoma) and connective tissue diseases.
-
The diagnosis of HLH is based on clinical and laboratory criteria, including cytopenias that may make the evaluation for triggering conditions such as HL more difficult.
-
Lymphoma should be included in the differential diagnosis of granulomatous inflammation.
Acknowledgements
The authors acknowledge Ralph Meyer, MD (Queen's University, Kingston, Ontario) for his comments on a draft of this paper and Drs. David Barth and Maha Guindi (Department of Laboratory Medicine, University Health Network) for their reviews of the pathology specimens.
A 23‐year‐old man presented to his family physician's office with a 2‐week history of fever, chills, night sweats, anorexia, and fatigue. This was associated with a 4‐month history of a nonproductive cough and a 20‐pound involuntary weight loss. He denied shortness of breath, chest pain, headaches, abdominal pain, vomiting, diarrhea, dysuria, and rash. There was no recent travel, sick contacts, or animal exposures.
This patient's symptoms could represent an underlying infectious, neoplastic, or inflammatory process. I would ascertain any relevant personal or family history and explore whether the patient has risk factors for human immunodeficiency virus (HIV) infection or tuberculosis (TB). On physical examination, I would listen for a heart murmur and look for lymphadenopathy, hepatosplenomegaly, and arthritis. Investigations including cultures, urinalysis, and a chest radiograph would be indicated at this time.
During the 2 weeks after his initial presentation, he experienced persistent fever, and further weight loss. He was admitted to the hospital to determine the etiology of his symptoms. The patient had no previous medical problems. On initial examination, his temperature was 102 degrees Fahrenheit, blood pressure was 100/65 mmHg, heart rate was 105 per minute, respiratory rate was 22 breaths per minute and oxygen saturation was normal on ambient air. He appeared cachectic. He was oriented to person, place, and time. Head and neck examination revealed no intraoral pathology, lymphadenopathy or scleral icterus, but did reveal conjunctival pallor. The chest was clear to auscultation, and the cardiovascular examination revealed a normal apical impulse and heart sounds with no murmurs. There was peripheral edema to the level of the mid‐shins bilaterally. The abdomen was soft and non‐tender with no appreciable hepatosplenomegaly. There were no stigmata of chronic liver disease. There was no axillary or inguinal lymphadenopathy. The remainder of the examination was normal. A complete blood count showed a hemoglobin concentration of 5.2 g/dL with a mean corpuscular volume (MCV) of 89fL, white blood cells were 1,400 cells/mm3 with an absolute neutrophil count (ANC) of 800 cells/mm3 and a platelet count of 90,000 cells/mm3 The serum sodium was 124 mmol/L, potassium 3.0 mmol/L, chloride 91 mmol/L, bicarbonate 26 mmol/L, and the creatinine 1.36 mg/dL His liver enzyme profile showed aspartate aminotransferase (AST) 68 U/L (normal 35), alanine aminotransferase (ALT) 25 U/L (normal 40), alkaline phosphatase (ALP) 210 U/L (normal 110) and a total bilirubin of 1.64 mg/dL.
The patient is clearly very unwell and requires admission to the hospital for treatment and further investigation. Emergent management includes administration of intravenous fluids to correct his electrolyte abnormalities, empiric broad spectrum antibiotics (given his relative neutropenia and fever), and a transfusion for his profound anemia. I would be very concerned that he has an underlying malignancy such as lymphoma or leukemia. Pancytopenia related to decreased cell production may be secondary to infiltration (malignant or granulomatous), infection (HIV, TB, fungal, viral), or aplasia (primary or drug‐related). Less likely etiologies include B12 or folate deficiency (unlikely given the normal MCV), systemic lupus erythematosus, paroxysmal nocturnal hemoglobinuria or cell sequestration due to hypersplenism. A history of recent exposure to drugs or toxins should be elicited. The patient's pulmonary symptoms may relate to the primary disorder or may represent an infection secondary to myelosuppression. I would want an immediate review of the peripheral blood smear, a hemolysis work‐up (drawn prior to transfusion including lactate dehydrogenase [LDH], haptoglobin, fractionated bilirubin, reticulocyte count and direct antiglobulin testing), antinuclear antibody (ANA), B12 and folate levels, imaging of the chest and blood cultures.
With evidence of fever and pancytopenia, acute leukemia was suspected and the patient was admitted to a hematology service. Over the next two weeks an extensive investigation including blood and urine cultures, and computed tomograms (CT) of the chest and abdomen were performed. A bone marrow aspirate and biopsy were also were done and were submitted for histopathologic examination and culture. The CT scan of the chest revealed left axillary and supraclavicular lymphadenopathy (Figure 1), and the abdominal imaging revealed splenomegaly. The blood, urine and bone marrow cultures were all negative. A peripheral blood smear showed pancytopenia with a hematologist interpretation suggesting that an intrinsic bone marrow process may be resulting in impaired cell production. The corresponding bone marrow biopsy and aspirate showed no evidence of malignancy, but there were numerous granulomata, and the periodic acid‐Schiff (PAS) and silver staining showed cells that resembled fungal elements (Figure 2).


The absence of malignant cells in the bone marrow leaves us to consider infectious and inflammatory causes of this patient's presentation. Infectious etiologies associated with bone marrow granulomata include fungal, mycobacterial, bacterial (brucellosis, typhoid and Q fever) and viral pathogens including HIV, Epstein‐Barr virus (EBV), and cytomegalovirus (CMV). Noninfectious causes include sarcoidosis, drug effects, and autoimmune conditions. The PAS and silver staining suggests this patient has a disseminated fungal infection. Disseminated Histoplasma capsulatum is the most likely organism but blastomycosis and coccidioidomycosis should be considered. HIV and occult lymphoma are considerations as is a primary immune disorder such as common variable immunodeficiency (CVID) which can present in this age group. While there is no recent travel history, it will be critical to determine where the patient currently lives and previously resided, review the medical record for prior infections and HIV risk factors, and take a thorough occupational history.
At this point, the following investigations should be undertaken: blood, sputum, and bone marrow culture; fungal and acid‐fast bacilli (AFB) stains on sputum and bone marrow; Histoplasma urine antigen; tuberculin skin test; serology for HIV and histoplasmosis; and serum protein electrophoresis with immunofixation and quantitation of immunoglobulins.
Acid fast staining of the bone marrow as well as mycobacterial and fungal cultures were negative. He lived in eastern Ontario and worked in construction. He reported helping tear down an old cabin in a wooded area, but denied any insect bites. This project coincided with the onset of his cough. He had no history of high risk sexual activity, intravenous drug use, tattoos or blood transfusions previous to his presentation. The HIV test was negative. His clinicians at this point considered a disseminated fungal infection as a cause for his symptoms and started him empirically on itraconazole He was discharged from the hospital with a plan for close outpatient followup. Within three days of discharge on the itraconazole, the patient's fever began to diminish, but did not completely resolve.
The clinical picture including cough, geography, and recent occupational exposure is entirely consistent with disseminated histoplasmosis. However, we are still lacking microbiologic confirmation of the diagnosis. Sarcoidosis and occult malignancy must still be considered. In the absence of a definitive diagnosis, I would consider bronchoscopy with bronchoalveolar lavage (BAL) and obtaining a lymph node or liver biopsy for microbiologic and pathologic examination. With the patient now receiving antifungal therapy, a diagnosis of histoplasmosis would be supported by a response to therapy, declining Histoplasma antigen levels and clinical improvement including recovery of his bone marrow.
The urine specimen was negative for Histoplasma antigen. Seven days after initiating itraconazole, he developed jaundice and confusion and was taken back to the hospital. On presentation, he was disoriented but awake. His temperature was 103.1 degrees Fahrenheit, blood pressure was 90/60 mm Hg, heart rate was 115 per minute, and oxygen saturation was normal on room air. He was obviously jaundiced, and more cachectic than previous. The neurologic examination demonstrated disorientation with no localizing findings. The chest and cardiovascular examinations were normal. His abdomen was soft and non‐tender with no evidence of hepatomegaly, but the spleen tip was palpable. There was no ascites or any other signs of portal hypertension, but his peripheral edema was worse than before and asterixis was present. The remainder of the examination was unchanged from previous. His laboratory investigations at this point showed a bilirubin of 18.5 mg/dL, AST 269 U/L, ALT 76 U/L, ALP 165 U/L, albumin 18 g/L, fibrinogen 1.53 g/L (normal 1.5‐3.5), triglycerides 2.4 mmol/L (normal 2), ferritin 59415 ug/L (normal 22‐275) an international normalized ratio (INR) of 2.65. His complete blood count still showed pancytopenia.
The patient has now developed fulminant hepatic failure. He requires volume resuscitation, drawing of repeat cultures, initiation of empiric broad spectrum antibiotics, urgent hepatology consultation and intensive care unit (ICU) support. The most common causes of acute liver failure are drug toxicity (including acetaminophen), viral hepatitis, Wilson's disease, Budd‐Chiari syndrome, cryptogenic liver disease and fatty infiltration. The critical diagnostic issue at this point is to determine if the liver failure is a secondary process (in which case drug toxicity due to itraconazole would be the most likely cause) or if this represents evolution of his primary disease with extensive hepatic involvement. Liver failure due to itraconazole has been reported and given the lack of microbiologic confirmation of a fungal infection, this agent should clearly be discontinued. Returning to our initial differential diagnosis of this man's granulomatous bone marrow infiltration and pancytopenia, etiologies which may progress to hepatic failure include viral infections (EBV or CMV) and malignancy. This patient's presentation could be an unusual manifestation of a common illness such as EBV or a rapidly progressive lymphoma. An abdominal Doppler ultrasound is required to rule out Budd‐Chiari syndrome. Given his abrupt change in clinical status, I would repeat a CT scan of his chest and abdomen to evaluate for evidence of infection, infiltration, or malignancy. Owing to the uncertainty regarding this patient's diagnosis and the rapidly progressive nature of his disease, serious consideration must be given to a transjugular liver biopsy.
Soon after admission, he developed hematemesis. He was given multiple blood transfusions, and then intravenous fluids, broad spectrum antibiotics and lactulose. Upper gastrointestinal endoscopy showed no varices, but did reveal multiple esophageal and gastric ulcerations. He was then transferred to a liver transplant center where repeat bone marrow biopsy and a liver biopsy were done. Both revealed extensive granulomatosis and the bone marrow biopsy showed evidence of hemophagocytosis (Figure 3).

The finding of hemophagocytosis in the setting of fever, hepatosplenomegaly, and pancytopenia is consistent with a diagnosis of hemophagocytic lymphohistiocytosis (HLH). The cornerstone of therapy for patients with HLH is suppression of the severe inflammatory response with corticosteroids, etoposide and cyclosporin. Patients who respond to this are candidates for allogeneic stem cell transplant with curative intent. This patient's hepatic dysfunction precludes the use of etoposide and initial therapy should therefore include dexamethasone and cyclosporin.
All bacterial, fungal, and mycobacterial cultures again demonstrated no growth. Broad spectrum antibiotics were continued, and empiric intravenous amphotericin B was added. He became hemodynamically unstable, was intubated, put on mechanical ventilation and required vasoactive medications to maintain his blood pressure. An empiric course of pulse corticosteroids was given for the possibility of sarcoidosis. His blood pressure stabilized, though he continued to require vasopressors.
While HLH has been very rarely reported in association with sarcoidosis, the underlying pathogenesis of his clinical presentation (infectious, neoplastic, or inflammatory) has not yet been confirmed. In the meantime, I would continue with supportive care and intravenous corticosteroids.
Immunohistochemical studies of the liver biopsy returned showing CD15/30+ cells with weak‐to‐negative CD45 expression cells typical of Hodgkin lymphoma (HL) (Figure 4). He was started on chemotherapy, but over 48 hours became progressively more hypotensive. The patient died of Klebsiella and Pseudomonas sepsis on the 7th hospital day. Post‐mortem immunohistochemical examination revealed evidence of Hodgkin disease in the axillary lymph nodes, bone marrow and liver. The bone marrow showed evidence of hemophagocytosis and was also positive for Epstein‐Barr encoded RNA (EBER). Serologic studies were subsequently available and revealed positive EBV IgM against the viral capsid antigen (VCA) as well as EBV IgG VCA, which in conjunction with the marrow findings, was highly suggestive of reactivation EBV disease.

Discussion
This patient's diagnostic course led both the clinical team and discussant down a winding path, which ultimately ended in the finding of Hodgkin lymphoma, a relatively common diagnosis that had been clouded by seemingly contradictory clinical and laboratory data. The provisional diagnosis of disseminated histoplasmosis was reasonable given that H. capsulatum is endemic in Ontario and that the patient's occupation placed him at risk of infection. Given the acuity of his illness, empiric antifungal therapy based on the report of fungal elements on bone marrow examination seemed reasonable. However, Histoplasma urinary antigen testing has been shown in the literature to be 98% sensitive in immunosuppressed populations, and the negative result prompted a re‐examination of the marrow specimen. The previously described fungal elements were felt to be most likely artifact, and the underlying diagnosis was reconsidered.1 This is when the repeat bone marrow examination pointed towards the diagnosis of HLH.
Hemophagocytic lymphohistiocytosis (HLH) is a severe, systemic hyperinflammatory disorder characterized by histiocytic proliferation that may be primary or can be triggered by infection, connective tissue diseases or malignancy.25 The central pathogenesis involves dysregulated Th1 cytokine secretion. This results in an uncontrolled accumulation of activated T‐lymphocytes and histiocytes in various organs including the liver, spleen and bone marrow. The infiltration of histiocytes into major organs can lead to disruption of function and multiorgan failure.6 Viruses are the most common infectious triggers of HLH, particularly EBV, and lymphoma is the most common associated malignancy.25 It is hypothesized that EBV can interfere with normal lymphocyte signaling pathways leading to the aforementioned over‐expression of Th1 cytokines, which can then trigger HLH.7 The diagnosis of HLH is based on a combination of clinical and laboratory parameters as outlined in Table 1.8 Our patient met all five of the major criteria.
| |
| Major criteria | 1. Fever |
| 2. Splenomegaly | |
| 3. Cytopenia in two or more cell lines | |
| 4. Hypertriglyceridemia or hypofibrinogenemia | |
| 5. Hemophagocytosis on histopathologic examination | |
| Alternative criteria | A. Low or absent natural killer cell activity |
| B. Serum ferritin level >500 ug/L | |
| C. Soluble CD‐25 level >2400 U/mL | |
The recommended treatment of HLH involves the administration of the HLH‐94 protocol consisting of corticosteroids, cyclosporine and etoposide.9, 10 Those who survive this initial phase are recommended for hematopoetic stem cell transplantation producing an overall 3‐year survival rate of 64%. However, those who do not receive early etoposide therapy fare much worse, with a mortality rate of 92%.10 This patient was not able to receive etoposide because of his decompensated liver disease.
In this case, the development of Hodgkin lymphoma involving the bone marrow and liver may have resulted in a state of immune suppression. The loss of immune function likely allowed the reactivation of Epstein‐Barr virus which triggered HLH and his fulminant presentation.35,9 Indeed, both the liver and bone marrow samples showed evidence of EBV reactivation as evidenced by the presence of EBER. The diagnosis of Hodgkin lymphoma was made from a liver biopsy specimen rather than bone marrow examination. The diagnosis of Hodgkin lymphoma is based on the presence of Reed‐Sternberg cells surrounded by an inflammatory milieu of cells including variable numbers of small lymphocytes, neutrophils eosinophils and fibroblasts. The HLH‐induced pancytopenia depleted the aforementioned inflammatory milieu in the bone marrow, which obscured the diagnosis of Hodgkin lymphoma. Unfortunately, lymphoma‐associated HLH has a very poor prognosis with a mortality rate of up to 60%.4 At the outset of this case, the reported fungal elements proved to be a source of confusion which delayed the diagnosis of Hodgkin lymphoma. Given the poor prognosis of lymphoma‐associated HLH, it is unlikely this would have had any effect on the ultimate outcome.
Teaching Points
-
HLH is a rare and complex hyperinflammatory disorder which may present as pancytopenia.
-
Triggers of HLH can include infections (particularly EBV), malignancy (particularly lymphoma) and connective tissue diseases.
-
The diagnosis of HLH is based on clinical and laboratory criteria, including cytopenias that may make the evaluation for triggering conditions such as HL more difficult.
-
Lymphoma should be included in the differential diagnosis of granulomatous inflammation.
Acknowledgements
The authors acknowledge Ralph Meyer, MD (Queen's University, Kingston, Ontario) for his comments on a draft of this paper and Drs. David Barth and Maha Guindi (Department of Laboratory Medicine, University Health Network) for their reviews of the pathology specimens.
- ,Antigen detection for diagnosis of the endemic mycoses.Curr Fung Infect Rep.2008;4:189–193.
- ,,, et al.Lymphoma‐associated hemophagocytic syndrome: Clinical features and treatment outcome.Ann Hematol.2007;86:493–498.
- ,,, et al.Hodgkin lymphoma‐associated hemophagocytic syndrome: A disorder strongly correlated with Epstein‐Barr virus.Clin Inf Dis.2008;47:531–534.
- ,.Hemophagocytic syndrome associated with Hodgkin lymphoma and pneumocystis jiroveci pneumonitis.Br J Hematol.2007;138:672.
- ,,, et al.Infections associated with haemophagocytic syndrome.Lancet Infect Dis.2007;12:814–822.
- ,,,.Hemophagocytic lymphohistiocytic syndrome: Unrecognized cause of multiple organ failure.Pediatr Crit Care Med.2000;1:51–54.
- ,,, et al.Epstein‐Barr virus LMP1 Inhibits the expression of SAP gene and upregulates Th1 cytokines in the pathogenesis of hemophagocytic syndrome.Blood.2005;106:3090–3096.
- ,,.Diagnostic guidelines for hemophagocytic lymphohistiocytosis.Semin Oncol.1991;18:29–33
- ,,, et al.Treatment of Epstein‐Barr virus‐associated hemophagocytic lymphohistiocytosis (EBV‐HLH) in young adults: A report from the HLH Study Center.Med Pediatr Oncol.2003;41:103–109.
- ,,, et al.Haematopoietic stem cell transplantation in haemophagocytic lymphohistiocytosis.Br J Haematol.2005;129:622–630.
- ,Antigen detection for diagnosis of the endemic mycoses.Curr Fung Infect Rep.2008;4:189–193.
- ,,, et al.Lymphoma‐associated hemophagocytic syndrome: Clinical features and treatment outcome.Ann Hematol.2007;86:493–498.
- ,,, et al.Hodgkin lymphoma‐associated hemophagocytic syndrome: A disorder strongly correlated with Epstein‐Barr virus.Clin Inf Dis.2008;47:531–534.
- ,.Hemophagocytic syndrome associated with Hodgkin lymphoma and pneumocystis jiroveci pneumonitis.Br J Hematol.2007;138:672.
- ,,, et al.Infections associated with haemophagocytic syndrome.Lancet Infect Dis.2007;12:814–822.
- ,,,.Hemophagocytic lymphohistiocytic syndrome: Unrecognized cause of multiple organ failure.Pediatr Crit Care Med.2000;1:51–54.
- ,,, et al.Epstein‐Barr virus LMP1 Inhibits the expression of SAP gene and upregulates Th1 cytokines in the pathogenesis of hemophagocytic syndrome.Blood.2005;106:3090–3096.
- ,,.Diagnostic guidelines for hemophagocytic lymphohistiocytosis.Semin Oncol.1991;18:29–33
- ,,, et al.Treatment of Epstein‐Barr virus‐associated hemophagocytic lymphohistiocytosis (EBV‐HLH) in young adults: A report from the HLH Study Center.Med Pediatr Oncol.2003;41:103–109.
- ,,, et al.Haematopoietic stem cell transplantation in haemophagocytic lymphohistiocytosis.Br J Haematol.2005;129:622–630.
Continuing Medical Education Program in
If you wish to receive credit for this activity, which begins on the next page, please refer to the website: www. blackwellpublishing.com/cme.
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to www.blackwellpublishing.com/cme.
-
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which begins on the next page, please refer to the website: www. blackwellpublishing.com/cme.
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to www.blackwellpublishing.com/cme.
-
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.
If you wish to receive credit for this activity, which begins on the next page, please refer to the website: www. blackwellpublishing.com/cme.
Accreditation and Designation Statement
Blackwell Futura Media Services designates this educational activity for a 1 AMA PRA Category 1 Credit. Physicians should only claim credit commensurate with the extent of their participation in the activity.
Blackwell Futura Media Services is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Educational Objectives
Continuous participation in the Journal of Hospital Medicine CME program will enable learners to be better able to:
-
Interpret clinical guidelines and their applications for higher quality and more efficient care for all hospitalized patients.
-
Describe the standard of care for common illnesses and conditions treated in the hospital; such as pneumonia, COPD exacerbation, acute coronary syndrome, HF exacerbation, glycemic control, venous thromboembolic disease, stroke, etc.
-
Discuss evidence‐based recommendations involving transitions of care, including the hospital discharge process.
-
Gain insights into the roles of hospitalists as medical educators, researchers, medical ethicists, palliative care providers, and hospital‐based geriatricians.
-
Incorporate best practices for hospitalist administration, including quality improvement, patient safety, practice management, leadership, and demonstrating hospitalist value.
-
Identify evidence‐based best practices and trends for both adult and pediatric hospital medicine.
Instructions on Receiving Credit
For information on applicability and acceptance of continuing medical education credit for this activity, please consult your professional licensing board.
This activity is designed to be completed within the time designated on the title page; physicians should claim only those credits that reflect the time actually spent in the activity. To successfully earn credit, participants must complete the activity during the valid credit period that is noted on the title page.
Follow these steps to earn credit:
-
Log on to www.blackwellpublishing.com/cme.
-
Read the target audience, learning objectives, and author disclosures.
-
Read the article in print or online format.
-
Reflect on the article.
-
Access the CME Exam, and choose the best answer to each question.
-
Complete the required evaluation component of the activity.