User login
Localizing General Medical Teams
Localizing inpatient general medical teams to nursing units has high intuitive validity for improving physician productivity, hospital efficiency, and patient outcomes. Motion or the moving of personnel between tasksso prominent if teams are not localizedis 1 of the 7 wastes in lean thinking.1 In a timemotion study, where hospitalists cared for patients on up to 5 different wards, O'Leary et al2 have reported large parts of hospitalists' workdays spent in indirect patient care (69%), paging (13%), and travel (3%). Localization could increase the amount of time available for direct patient care, decrease time spent for (and interruptions due to) paging, and decrease travel time, all leading to greater productivity.
O'Leary et al3 have also reported the beneficial effects of localization of medical inpatients on communication between nurses and physicians, who could identify each other more often, and reported greater communication (specifically face‐to‐face communication) with each other following localization. This improvement in communication and effective multidisciplinary rounds could lead to safer care4 and better outcomes.
Further investigations about the effect of localization are limited. Roy et al5 have compared the outcomes of patients localized to 2 inpatient pods medically staffed by hospitalists and physician assistants (PAs) to geographically dispersed, but structurally different, house staff teams. They noticed significantly lower costs, slight but nonsignificant increase in length of stay, and no difference in mortality or readmissions, but it is impossible to tease out the affect of localization versus the affect of team composition. In a before‐and‐after study, Findlay et al6 have reported a decrease in mortality and complication rates in clinically homogenous surgical patients (proximal hip fractures) when cared for by junior trainee physicians localized to a unit, but their experience cannot be extrapolated to the much more diverse general medical population.
In our hospital, each general medical team could admit patients dispersed over 14 different units. An internal group, commissioned to evaluate our hospitalist practice, recommended reducing this dispersal to improve physician productivity, hospital efficiency, and outcomes of care. We therefore conducted a project to evaluate the impact of localizing general medical inpatient teams to a single nursing unit.
METHODS
Setting
We conducted our project at a 490 bed, urban academic medical center in the midwestern United States where of the 10 total general medical teams, 6 were traditional resident‐based teams and 4 consisted of a hospitalist paired with a PA (H‐PA teams). We focused our study on the 4 H‐PA teams. The hospitalists could be assigned to any H‐PA team and staffed them for 2 weeks (including weekends). The PAs were always assigned to the same team but took weekends off. An in‐house hospitalist provided overnight cross‐coverage for the H‐PA teams. Prior to our intervention, these teams could admit patients to any of the 14 nursing units at our hospital. They admitted patients from 7 AM to 3 PM, and also accepted care of patients admitted overnight after the resident teams had reached their admission limits (overflow). A Faculty Admitting Medical Officer (AMO) balanced the existing workload of the teams against the number and complexity of incoming patients to decide team assignment for the patients. The AMO was given guidelines (soft caps) to limit total admissions to H‐PA teams to 5 per team per day (3 on a weekend), and to not exceed a total patient census of 16 for an H‐PA team.
Intervention
Starting April 1, 2010, until July 15, 2010, we localized patients admitted to 2 of our 4 H‐PA teams on a single 32‐bed nursing unit. The patients of the other 2 H‐PA teams remained dispersed throughout the hospital.
Transition
April 1, 2010 was a scheduled switch day for the hospitalists on the H‐PA teams. We took advantage of this switch day and reassigned all patients cared for by H‐PA teams on our localized unit to the 2 localized teams. Similarly, all patients on nonlocalized units cared for by H‐PA teams were reassigned to the 2 nonlocalized teams. All patients cared for by resident teams on the localized unit, that were anticipated to be discharged soon, stayed until discharge; those that had a longer stay anticipated were transferred to a nonlocalized unit.
Patient Assignment
The 4 H‐PA teams continued to accept patients between 7 AM and 3 PM, as well as overflow patients. Patients with sickle cell crises were admitted exclusively to the nonlocalized teams, as they were cared for on a specialized nursing unit. No other patient characteristic was used to decide team assignment.
The AMO balanced the existing workload of the teams against the number and complexity of incoming patients to decide team assignment for the patients, but if these factors were equivocal, the AMO was now asked to preferentially admit to the localized teams. The admission soft cap for the H‐PA teams remained the same (5 on weekdays and 3 on weekends). The soft cap on the total census of 16 patients for the nonlocalized teams remained, but we imposed hard caps on the total census for the localized teams. These hard caps were 16 for each localized team for the month of April (to fill a 32‐bed unit), then decreased to 12 for the month of May, as informal feedback from the teams suggested a need to decrease workload, and then rebalanced to 14 for the remaining study period.
Evaluation
Clinical Outcomes
Using both concurrent and historical controls, we evaluated the impact of localization on the following clinical outcome measures: length of stay (LOS), charges, and 30‐day readmission rates.
Inclusion Criteria
We included all patients assigned to localized and nonlocalized teams between the period April 1, 2010 to July 15, 2010, and discharged before July 16, 2010, in our intervention group and concurrent control group, respectively. We included all patients assigned to any of the 4 H‐PA teams during the period January 1, 2010 and March 31, 2010 in the historical control group.
Exclusion Criteria
From the historical control group, we excluded patients assigned to one particular H‐PA team during the period January 1, 2010 to February 28, 2010, during which the PA assigned to that team was on leave. We excluded, from all groups, patients with a diagnosis of sickle cell disease and hospitalizations that straddled the start of the intervention. Further, we excluded repeat admissions for each patient.
Data Collection
We used admission logs to determine team assignment and linked them to our hospital's discharge abstract database to get patient level data. We grouped the principal diagnosis, International Classification of Diseases, Ninth Revision, Clinical Modification (ICD‐9‐CM) codes into clinically relevant categories using the Healthcare Cost and Utilization Project Clinical Classification Software for ICD‐9‐CM (Rockville, MD, www.hcup‐us.ahrq.gov/toolssoftware/ccs/ccs.jsp). We created comorbidity measures using Healthcare Cost and Utilization Project Comorbidity Software, version 3.4 (Rockville, MD, www.hcup‐us.ahrq.gov/toolssoftware/comorbidity/comorbidity.jsp).
We calculated LOS by subtracting the discharge day and time from the admission day and time. We summed all charges accrued during the entire hospital stay, but did not include professional fees. The LOS and charges included time spent and charges accrued in the intensive care unit (ICU). As ICU care was not under the control of the general medical teams and could have a significant impact on outcomes reflecting resource utilization, we compared LOS and charges only for 2 subsets of patients: patients not initially admitted to ICU before care by medical teams, and patients never requiring ICU care. We considered any repeat hospitalization to our hospital within 30 days following a discharge to be a readmission, except those for a planned procedure or for inpatient rehabilitation. We compared readmission rates for all patients irrespective of ICU stay, as discharge planning for all patients was under the direct control of the general medical teams.
Data Analysis
We performed unadjusted descriptive statistics using medians and interquartile ranges for continuous variables, and frequencies and percentages for categorical variables. We used chi‐square tests of association, and KruskalWallis analysis of variance, to compare baseline characteristics of patients assigned to localized and control teams.
We used regression models with random effects to risk adjust for a wide variety of variables. We included age, gender, race, insurance, admission source, time, day of week, discharge time, and total number of comorbidities as fixed effects in all models. We then added individual comorbidity measures one by one as fixed effects, including them only if significant at P < 0.01. We always added a variable identifying the admitting physician as a random effect, to account for dependence between admissions to the same physician. We log transformed LOS and charges because they were extremely skewed in nature. We analyzed readmissions after excluding patients who died. We evaluated the affect of our intervention on clinical outcomes using both historical and concurrent controls. We report P values for both overall 3‐way comparisons, as well as each of the 2‐way comparisonsintervention versus historical control and intervention versus concurrent control.
Productivity and Workflow Measures
We also evaluated the impact of localization on the following productivity and workflow measures: number of pages received, number of patient encounters, relative value units (RVUs) generated, and steps walked by PAs.
Data Collection
We queried our in‐house paging systems for the number of pages received by intervention and concurrent control teams between 7 AM and 6 PM (usual workday). We queried our professional billing data to determine the number of encounters per day and RVUs generated by the intervention, as well as historical and concurrent control teams, as a measure of productivity.
During the last 15 days of our intervention (July 1 July 15, 2010), we received 4 pedometers and we asked the PAs to record the number of steps taken during their workday. We chose PAs, rather than physicians, as the PAs had purely clinical duties and their walking activity would reflect activity for solely clinical purposes.
Data Analysis
For productivity and workflow measures, we adjusted for the day of the week and used random effects models to adjust for clustering of data by physician and physician assistant.
Statistical Software
We performed the statistical analysis using R software, versions 2.9.0 (The R Project for Statistical Computing, Vienna, Austria, http://www.R‐project.org).
Ethical Concerns
The study protocol was approved by our institutional review board.
RESULTS
Study Population
There were 2431 hospitalizations to the 4 H‐PA teams during the study period. Data from 37 hospitalizations was excluded because of missing data. After applying all exclusion criteria, our final study sample consisted of a total of 1826 first hospitalizations for patients: 783 historical controls, 478 concurrent controls, and 565 localized patients.
Patients in the control groups and intervention group were similar in age, gender, race, and insurance status. Patients in the intervention group were more likely to be admitted over the weekend, but had similar probability of being discharged over the weekend or having had an ICU stay. Historical controls were admitted more often between 6 AM and 12 noon, while during the intervention period, patients were more likely to be admitted between midnight and 6 AM. The discharge time was similar across all groups. The 5 most common diagnoses were similar across the groups (Table 1).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| Patients | 783 | 565 | 478 | |
| Age median (IQR) | 57 (4575) | 57 (4573) | 56 (4470) | 0.186 |
| Age groups, n (%) | ||||
| <30 | 65 (8.3) | 37 (6.6) | 46 (9.6) | |
| 3039 | 76 (9.7) | 62 (11.0) | 47 (9.8) | |
| 4049 | 114 (14.6) | 85 (15.0) | 68 (14.2) | |
| 5059 | 162 (20.7) | 124 (22.0) | 118 (24.7) | 0.145 |
| 6069 | 119 (15.2) | 84 (14.9) | 76 (16.0) | |
| 7079 | 100 (12.8) | 62 (11.0) | 58 (12.1) | |
| 8089 | 113 (14.4) | 95 (16.8) | 51 (10.7) | |
| >89 | 34 (4.3) | 16 (2.88) | 14 (2.9) | |
| Female gender, n (%) | 434 (55.4) | 327 (57.9) | 264 (55.2) | 0.602 |
| Race: Black, n (%) | 285 (36.4) | 229 (40.5) | 200 (41.8) | 0.111 |
| Observation status, n (%) | 165 (21.1) | 108 (19.1) | 108 (22.6) | 0.380 |
| Insurance, n (%) | ||||
| Commercial | 171 (21.8) | 101 (17.9) | 101 (21.1) | |
| Medicare | 376 (48.0) | 278 (49.2) | 218 (45.6) | 0.225 |
| Medicaid | 179 (22.8) | 126 (22.3) | 117 (24.5) | |
| Uninsured | 54 (7.3) | 60 (10.6) | 42 (8.8) | |
| Weekend admission, n (%) | 137 (17.5) | 116 (20.5) | 65 (13.6) | 0.013 |
| Weekend discharge, n (%) | 132 (16.9) | 107 (18.9) | 91 (19.0) | 0.505 |
| Source of admission | ||||
| ED, n (%) | 654 (83.5) | 450 (79.7) | 370 (77.4) | 0.022 |
| No ICU stay, n (%) | 600 (76.6) | 440 (77.9) | 383 (80.1) | 0.348 |
| Admission time, n (%) | ||||
| 00000559 | 239 (30.5) | 208 (36.8) | 172 (36.0) | |
| 06001159 | 296 (37.8) | 157 (27.8) | 154 (32.2) | 0.007 |
| 12001759 | 183 (23.4) | 147 (26.0) | 105 (22.0) | |
| 18002359 | 65 (8.3) | 53 (9.4) | 47 (9.8) | |
| Discharge time, n (%) | ||||
| 00001159 | 67 (8.6) | 45 (8.0) | 43 (9.0) | |
| 12001759 | 590 (75.4) | 417 (73.8) | 364 (76.2) | 0.658 |
| 18002359 | 126 (16.1) | 103 (18.2) | 71 (14.9) | |
| Inpatient deaths, n | 13 | 13 | 6 | |
| Top 5 primary diagnoses (%) | ||||
| 1 | Chest pain (11.5) | Chest pain (13.3) | Chest pain (11.9) | |
| 2 | Septicemia (6.4) | Septicemia (5.1) | Septicemia (3.8) | |
| 3 | Diabetes w/cm (4.6) | Pneumonia (4.9) | Diabetes w/cm (3.3) | n/a |
| 4 | Pneumonia (2.8) | Diabetes w/cm (4.1) | Pneumonia (3.3) | |
| 5 | UTI (2.7) | COPD (3.2) | UTI (2.9) | |
Clinical Outcomes
Unadjusted Analyses
The risk of 30‐day readmission was no different between the intervention and control groups. In patients without an initial ICU stay, and without any ICU stay, charges incurred and LOS were no different between the intervention and control groups (Table 2).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| 30‐day readmissions n (%) | 118 (15.3) | 69 (12.5) | 66 (14.0) | 0.346 |
| Charges: excluding patients initially admitted to ICU | ||||
| Median (IQR) in $ | 9346 (621614,520) | 9724 (665715,390) | 9902 (661115,670) | 0.393 |
| Charges: excluding all patients with an ICU stay | ||||
| Median (IQR) in $ | 9270 (618713,990) | 9509 (660114,940) | 9846 (658015,400) | 0.283 |
| Length of stay: excluding patients initially admitted to ICU | ||||
| Median (IQR) in days | 1.81 (1.223.35) | 2.16 (1.214.02) | 1.89 (1.193.50) | 0.214 |
| Length of stay: excluding all patients with an ICU stay | ||||
| Median (IQR) in days | 1.75 (1.203.26) | 2.12 (1.203.74) | 1.84 (1.193.42) | 0.236 |
Adjusted Analysis
The risk of 30‐day readmission was no different between the intervention and control groups. In patients without an initial ICU stay, and without any ICU stay, charges incurred were no different between the intervention and control groups; LOS was about 11% higher in the localized group as compared to historical controls, and about 9% higher as compared to the concurrent control group. The difference in LOS was not statistically significant on an overall 3‐way comparison (Table 3).
| Localized Teams in Comparison to | |||
|---|---|---|---|
| Historical Control | Concurrent Control | Overall P Value | |
| |||
| 30‐day risk of readmission OR (CI) | 0.85 (0.611.19) | 0.94 (0.651.37) | 0.630 |
| P value | 0.351 | 0.751 | |
| Charges: excluding patients initially admitted to ICU | |||
| % change | 2% higher | 4% lower | 0.367 |
| (CI) | (6% lower to 11% higher) | (12% lower to 5%higher) | |
| P value | 0.572 | 0.427 | |
| Charges: excluding all patients with an ICU stay | |||
| % change | 2% higher | 5% lower | 0.314 |
| (CI) | (6% lower to 10% higher) | (13% lower to 4% higher) | |
| P value | 0.695 | 0.261 | |
| Length of stay: excluding patients initially admitted to ICU | |||
| % change | 11% higher | 9% higher | 0.105 |
| (CI) | (1% to 22% higher) | (3% lower to 21% higher) | |
| P value | 0.038 | 0.138 | |
| Length of stay: excluding all patients with an ICU stay | |||
| % change | 10% higher | 8% higher | 0.133 |
| (CI) | (0% to 22% higher) | (3% lower to 20% higher) | |
| P value | 0.047 | 0.171 | |
Productivity and Workflow Measures
Unadjusted Analyses
The localized teams received fewer pages as compared to concurrently nonlocalized teams. Localized teams had more patient encounters per day and generated more RVUs per day as compared to both historical and concurrent control groups. Physician assistants on localized teams took fewer steps during their work day (Table 4).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| Pages received/day (7 AM6 PM) Median (IQR) | No data | 15 (921) | 28 (12.540) | <0.001 |
| Total encounters/day Median (IQR) | 10 (813) | 12 (1013) | 11 (913) | <0.001 |
| RVU/day | ||||
| Mean (SD) | 19.9 (6.76) | 22.6 (5.6) | 21.2 (6.7) | <0.001 |
| Steps/day Median (IQR) | No data | 4661 (3922 5166) | 5554 (50606544) | <0.001 |
Adjusted Analysis
On adjusting for clustering by physician and day of week, the significant differences in pages received, total patient encounters, and RVUs generated persisted, while the difference in steps walked by PAs was attenuated to a statistically nonsignificant level (Table 5). The increase in RVU productivity was sustained through various periods of hard caps (data not shown).
| Localized Teams in Comparison to | |||
|---|---|---|---|
| Historical Control | Concurrent Control | Overall P Value | |
| |||
| Pages received (7 AM 6 PM) %(CI) | No data | 51% fewer (4854) | |
| P value | P < 0.001 | ||
| Total encounters | 0.89 more | 1.02 more | |
| N (CI) | (0.371.41) | (0.461.58) | |
| P value | P < 0.001 | P < 0.001 | P < 0.001 |
| RVU/day | 2.20 more | 1.36 more | |
| N (CI) | (1.103.29) | (0.172.55) | |
| P value | P < 0.001 | P = 0.024 | P < 0.001 |
| Steps/day | 1186 fewer (791 more to | ||
| N (CI) | No data | 3164 fewer) | |
| P value | P = 0.240 | ||
DISCUSSION
We found that general medical patients admitted to H‐PA teams and localized to a single nursing unit had similar risk of 30‐day readmission and charges, but may have had a higher length of stay compared to historical and concurrent controls. The localized teams received far fewer pages, had more patient encounters, generated more RVUs, and walked less during their work day. Taken together, these findings imply that in our study, localization led to greater team productivity and a possible decrease in hospital efficiency, with no significant impact on readmissions or charges incurred.
The higher productivity was likely mediated by the preferential assignments of more patients to the localized teams, and improvements in workflow (such as fewer pages and fewer steps walked), which allowed them to provide more care with the same resources as the control teams. Kocher and Sahni7 recently pointed out that the healthcare sector has experienced no gains in labor productivity in the past 20 years. Our intervention fits their prescription for redesigning healthcare delivery models to achieve higher productivity.
The possibility of a higher LOS associated with localization was a counterintuitive finding, and similar to that reported by Roy et al.5 We propose 3 hypotheses to explain this:
Selection bias: Higher workload of the localized teams led to compromised efficiency and a higher length of stay (eg, localized teams had fewer observation admissions, more hospitalizations with an ICU stay, and the AMO was asked to preferentially admit patients to localized teams).
Localization provided teams the opportunity to spend more time with their patients (by decreasing nonvalue‐added tasks) and to consequently address more issues before transitioning to outpatient care, or to provide higher quality of care.
Gaming: By having a hard cap on total number of occupied beds, we provided a perverse incentive to the localized teams to retain patients longer to keep assigned beds occupied, thereby delaying new admissions to avoid higher workload.
Our study cannot tell us which of these hypotheses represents the dominant phenomenon that led to this surprising finding. Hypothesis 3 is most worrying, and we suggest that others looking to localize their medical teams consider the possibility of unintended perverse incentives.
Differences were more pronounced between the historical control group and the intervention group, as opposed to the intervention group and concurrent controls. This may have occurred if we contaminated the concurrent control by decreasing the number of units they had to go to, by sequestering 1 unit for the intervention team.
Our report has limitations. It is a nonrandomized, quasi‐experimental investigation using a single institution's administrative databases. Our intervention was small in scale (localizing 2 out of 10 general medical teams on 1 out of 14 nursing units). What impact a wider implementation of localization may have on emergency department throughput and hospital occupancy remains to be studied. Nevertheless, our research is the first report, to our knowledge, investigating a wide variety of outcomes of localizing inpatient medical teams, and adds significantly to the limited research on this topic. It also provides significant operational details for other institutions to use when localizing medical teams.
We conclude that our intervention of localization of medical teams to a single nursing unit led to higher productivity and better workflow, but did not impact readmissions or charges incurred. We caution others designing similar localization interventions to protect against possible perverse incentives for inefficient care.
Acknowledgements
Disclosure: Nothing to report.
- . Reducing waste in US health care systems. JAMA. 2007;297(8):871–874.
- , , . How hospitalists spend their time: insights on efficiency and safety. J Hosp Med. 2006;1(2):88–93.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 2009;24(11):1223–1227.
- , , , et al. Structured interdisciplinary rounds in a medical teaching unit: improving patient safety. Arch Intern Med. 2011;171(7):678–684.
- , , , et al. Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes. J Hosp Med. 2008;3(5):361–368.
- , , , , . Ward‐based rather than team‐based junior surgical doctors reduce mortality for patients with a fracture of the proximal femur: results from a two‐year observational study. J Bone Joint Surg Br. 2011;93‐B(3):393–398.
- , . Rethinking health care labor. N Engl J Med. 2011;365(15):1370–1372.
Localizing inpatient general medical teams to nursing units has high intuitive validity for improving physician productivity, hospital efficiency, and patient outcomes. Motion or the moving of personnel between tasksso prominent if teams are not localizedis 1 of the 7 wastes in lean thinking.1 In a timemotion study, where hospitalists cared for patients on up to 5 different wards, O'Leary et al2 have reported large parts of hospitalists' workdays spent in indirect patient care (69%), paging (13%), and travel (3%). Localization could increase the amount of time available for direct patient care, decrease time spent for (and interruptions due to) paging, and decrease travel time, all leading to greater productivity.
O'Leary et al3 have also reported the beneficial effects of localization of medical inpatients on communication between nurses and physicians, who could identify each other more often, and reported greater communication (specifically face‐to‐face communication) with each other following localization. This improvement in communication and effective multidisciplinary rounds could lead to safer care4 and better outcomes.
Further investigations about the effect of localization are limited. Roy et al5 have compared the outcomes of patients localized to 2 inpatient pods medically staffed by hospitalists and physician assistants (PAs) to geographically dispersed, but structurally different, house staff teams. They noticed significantly lower costs, slight but nonsignificant increase in length of stay, and no difference in mortality or readmissions, but it is impossible to tease out the affect of localization versus the affect of team composition. In a before‐and‐after study, Findlay et al6 have reported a decrease in mortality and complication rates in clinically homogenous surgical patients (proximal hip fractures) when cared for by junior trainee physicians localized to a unit, but their experience cannot be extrapolated to the much more diverse general medical population.
In our hospital, each general medical team could admit patients dispersed over 14 different units. An internal group, commissioned to evaluate our hospitalist practice, recommended reducing this dispersal to improve physician productivity, hospital efficiency, and outcomes of care. We therefore conducted a project to evaluate the impact of localizing general medical inpatient teams to a single nursing unit.
METHODS
Setting
We conducted our project at a 490 bed, urban academic medical center in the midwestern United States where of the 10 total general medical teams, 6 were traditional resident‐based teams and 4 consisted of a hospitalist paired with a PA (H‐PA teams). We focused our study on the 4 H‐PA teams. The hospitalists could be assigned to any H‐PA team and staffed them for 2 weeks (including weekends). The PAs were always assigned to the same team but took weekends off. An in‐house hospitalist provided overnight cross‐coverage for the H‐PA teams. Prior to our intervention, these teams could admit patients to any of the 14 nursing units at our hospital. They admitted patients from 7 AM to 3 PM, and also accepted care of patients admitted overnight after the resident teams had reached their admission limits (overflow). A Faculty Admitting Medical Officer (AMO) balanced the existing workload of the teams against the number and complexity of incoming patients to decide team assignment for the patients. The AMO was given guidelines (soft caps) to limit total admissions to H‐PA teams to 5 per team per day (3 on a weekend), and to not exceed a total patient census of 16 for an H‐PA team.
Intervention
Starting April 1, 2010, until July 15, 2010, we localized patients admitted to 2 of our 4 H‐PA teams on a single 32‐bed nursing unit. The patients of the other 2 H‐PA teams remained dispersed throughout the hospital.
Transition
April 1, 2010 was a scheduled switch day for the hospitalists on the H‐PA teams. We took advantage of this switch day and reassigned all patients cared for by H‐PA teams on our localized unit to the 2 localized teams. Similarly, all patients on nonlocalized units cared for by H‐PA teams were reassigned to the 2 nonlocalized teams. All patients cared for by resident teams on the localized unit, that were anticipated to be discharged soon, stayed until discharge; those that had a longer stay anticipated were transferred to a nonlocalized unit.
Patient Assignment
The 4 H‐PA teams continued to accept patients between 7 AM and 3 PM, as well as overflow patients. Patients with sickle cell crises were admitted exclusively to the nonlocalized teams, as they were cared for on a specialized nursing unit. No other patient characteristic was used to decide team assignment.
The AMO balanced the existing workload of the teams against the number and complexity of incoming patients to decide team assignment for the patients, but if these factors were equivocal, the AMO was now asked to preferentially admit to the localized teams. The admission soft cap for the H‐PA teams remained the same (5 on weekdays and 3 on weekends). The soft cap on the total census of 16 patients for the nonlocalized teams remained, but we imposed hard caps on the total census for the localized teams. These hard caps were 16 for each localized team for the month of April (to fill a 32‐bed unit), then decreased to 12 for the month of May, as informal feedback from the teams suggested a need to decrease workload, and then rebalanced to 14 for the remaining study period.
Evaluation
Clinical Outcomes
Using both concurrent and historical controls, we evaluated the impact of localization on the following clinical outcome measures: length of stay (LOS), charges, and 30‐day readmission rates.
Inclusion Criteria
We included all patients assigned to localized and nonlocalized teams between the period April 1, 2010 to July 15, 2010, and discharged before July 16, 2010, in our intervention group and concurrent control group, respectively. We included all patients assigned to any of the 4 H‐PA teams during the period January 1, 2010 and March 31, 2010 in the historical control group.
Exclusion Criteria
From the historical control group, we excluded patients assigned to one particular H‐PA team during the period January 1, 2010 to February 28, 2010, during which the PA assigned to that team was on leave. We excluded, from all groups, patients with a diagnosis of sickle cell disease and hospitalizations that straddled the start of the intervention. Further, we excluded repeat admissions for each patient.
Data Collection
We used admission logs to determine team assignment and linked them to our hospital's discharge abstract database to get patient level data. We grouped the principal diagnosis, International Classification of Diseases, Ninth Revision, Clinical Modification (ICD‐9‐CM) codes into clinically relevant categories using the Healthcare Cost and Utilization Project Clinical Classification Software for ICD‐9‐CM (Rockville, MD, www.hcup‐us.ahrq.gov/toolssoftware/ccs/ccs.jsp). We created comorbidity measures using Healthcare Cost and Utilization Project Comorbidity Software, version 3.4 (Rockville, MD, www.hcup‐us.ahrq.gov/toolssoftware/comorbidity/comorbidity.jsp).
We calculated LOS by subtracting the discharge day and time from the admission day and time. We summed all charges accrued during the entire hospital stay, but did not include professional fees. The LOS and charges included time spent and charges accrued in the intensive care unit (ICU). As ICU care was not under the control of the general medical teams and could have a significant impact on outcomes reflecting resource utilization, we compared LOS and charges only for 2 subsets of patients: patients not initially admitted to ICU before care by medical teams, and patients never requiring ICU care. We considered any repeat hospitalization to our hospital within 30 days following a discharge to be a readmission, except those for a planned procedure or for inpatient rehabilitation. We compared readmission rates for all patients irrespective of ICU stay, as discharge planning for all patients was under the direct control of the general medical teams.
Data Analysis
We performed unadjusted descriptive statistics using medians and interquartile ranges for continuous variables, and frequencies and percentages for categorical variables. We used chi‐square tests of association, and KruskalWallis analysis of variance, to compare baseline characteristics of patients assigned to localized and control teams.
We used regression models with random effects to risk adjust for a wide variety of variables. We included age, gender, race, insurance, admission source, time, day of week, discharge time, and total number of comorbidities as fixed effects in all models. We then added individual comorbidity measures one by one as fixed effects, including them only if significant at P < 0.01. We always added a variable identifying the admitting physician as a random effect, to account for dependence between admissions to the same physician. We log transformed LOS and charges because they were extremely skewed in nature. We analyzed readmissions after excluding patients who died. We evaluated the affect of our intervention on clinical outcomes using both historical and concurrent controls. We report P values for both overall 3‐way comparisons, as well as each of the 2‐way comparisonsintervention versus historical control and intervention versus concurrent control.
Productivity and Workflow Measures
We also evaluated the impact of localization on the following productivity and workflow measures: number of pages received, number of patient encounters, relative value units (RVUs) generated, and steps walked by PAs.
Data Collection
We queried our in‐house paging systems for the number of pages received by intervention and concurrent control teams between 7 AM and 6 PM (usual workday). We queried our professional billing data to determine the number of encounters per day and RVUs generated by the intervention, as well as historical and concurrent control teams, as a measure of productivity.
During the last 15 days of our intervention (July 1 July 15, 2010), we received 4 pedometers and we asked the PAs to record the number of steps taken during their workday. We chose PAs, rather than physicians, as the PAs had purely clinical duties and their walking activity would reflect activity for solely clinical purposes.
Data Analysis
For productivity and workflow measures, we adjusted for the day of the week and used random effects models to adjust for clustering of data by physician and physician assistant.
Statistical Software
We performed the statistical analysis using R software, versions 2.9.0 (The R Project for Statistical Computing, Vienna, Austria, http://www.R‐project.org).
Ethical Concerns
The study protocol was approved by our institutional review board.
RESULTS
Study Population
There were 2431 hospitalizations to the 4 H‐PA teams during the study period. Data from 37 hospitalizations was excluded because of missing data. After applying all exclusion criteria, our final study sample consisted of a total of 1826 first hospitalizations for patients: 783 historical controls, 478 concurrent controls, and 565 localized patients.
Patients in the control groups and intervention group were similar in age, gender, race, and insurance status. Patients in the intervention group were more likely to be admitted over the weekend, but had similar probability of being discharged over the weekend or having had an ICU stay. Historical controls were admitted more often between 6 AM and 12 noon, while during the intervention period, patients were more likely to be admitted between midnight and 6 AM. The discharge time was similar across all groups. The 5 most common diagnoses were similar across the groups (Table 1).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| Patients | 783 | 565 | 478 | |
| Age median (IQR) | 57 (4575) | 57 (4573) | 56 (4470) | 0.186 |
| Age groups, n (%) | ||||
| <30 | 65 (8.3) | 37 (6.6) | 46 (9.6) | |
| 3039 | 76 (9.7) | 62 (11.0) | 47 (9.8) | |
| 4049 | 114 (14.6) | 85 (15.0) | 68 (14.2) | |
| 5059 | 162 (20.7) | 124 (22.0) | 118 (24.7) | 0.145 |
| 6069 | 119 (15.2) | 84 (14.9) | 76 (16.0) | |
| 7079 | 100 (12.8) | 62 (11.0) | 58 (12.1) | |
| 8089 | 113 (14.4) | 95 (16.8) | 51 (10.7) | |
| >89 | 34 (4.3) | 16 (2.88) | 14 (2.9) | |
| Female gender, n (%) | 434 (55.4) | 327 (57.9) | 264 (55.2) | 0.602 |
| Race: Black, n (%) | 285 (36.4) | 229 (40.5) | 200 (41.8) | 0.111 |
| Observation status, n (%) | 165 (21.1) | 108 (19.1) | 108 (22.6) | 0.380 |
| Insurance, n (%) | ||||
| Commercial | 171 (21.8) | 101 (17.9) | 101 (21.1) | |
| Medicare | 376 (48.0) | 278 (49.2) | 218 (45.6) | 0.225 |
| Medicaid | 179 (22.8) | 126 (22.3) | 117 (24.5) | |
| Uninsured | 54 (7.3) | 60 (10.6) | 42 (8.8) | |
| Weekend admission, n (%) | 137 (17.5) | 116 (20.5) | 65 (13.6) | 0.013 |
| Weekend discharge, n (%) | 132 (16.9) | 107 (18.9) | 91 (19.0) | 0.505 |
| Source of admission | ||||
| ED, n (%) | 654 (83.5) | 450 (79.7) | 370 (77.4) | 0.022 |
| No ICU stay, n (%) | 600 (76.6) | 440 (77.9) | 383 (80.1) | 0.348 |
| Admission time, n (%) | ||||
| 00000559 | 239 (30.5) | 208 (36.8) | 172 (36.0) | |
| 06001159 | 296 (37.8) | 157 (27.8) | 154 (32.2) | 0.007 |
| 12001759 | 183 (23.4) | 147 (26.0) | 105 (22.0) | |
| 18002359 | 65 (8.3) | 53 (9.4) | 47 (9.8) | |
| Discharge time, n (%) | ||||
| 00001159 | 67 (8.6) | 45 (8.0) | 43 (9.0) | |
| 12001759 | 590 (75.4) | 417 (73.8) | 364 (76.2) | 0.658 |
| 18002359 | 126 (16.1) | 103 (18.2) | 71 (14.9) | |
| Inpatient deaths, n | 13 | 13 | 6 | |
| Top 5 primary diagnoses (%) | ||||
| 1 | Chest pain (11.5) | Chest pain (13.3) | Chest pain (11.9) | |
| 2 | Septicemia (6.4) | Septicemia (5.1) | Septicemia (3.8) | |
| 3 | Diabetes w/cm (4.6) | Pneumonia (4.9) | Diabetes w/cm (3.3) | n/a |
| 4 | Pneumonia (2.8) | Diabetes w/cm (4.1) | Pneumonia (3.3) | |
| 5 | UTI (2.7) | COPD (3.2) | UTI (2.9) | |
Clinical Outcomes
Unadjusted Analyses
The risk of 30‐day readmission was no different between the intervention and control groups. In patients without an initial ICU stay, and without any ICU stay, charges incurred and LOS were no different between the intervention and control groups (Table 2).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| 30‐day readmissions n (%) | 118 (15.3) | 69 (12.5) | 66 (14.0) | 0.346 |
| Charges: excluding patients initially admitted to ICU | ||||
| Median (IQR) in $ | 9346 (621614,520) | 9724 (665715,390) | 9902 (661115,670) | 0.393 |
| Charges: excluding all patients with an ICU stay | ||||
| Median (IQR) in $ | 9270 (618713,990) | 9509 (660114,940) | 9846 (658015,400) | 0.283 |
| Length of stay: excluding patients initially admitted to ICU | ||||
| Median (IQR) in days | 1.81 (1.223.35) | 2.16 (1.214.02) | 1.89 (1.193.50) | 0.214 |
| Length of stay: excluding all patients with an ICU stay | ||||
| Median (IQR) in days | 1.75 (1.203.26) | 2.12 (1.203.74) | 1.84 (1.193.42) | 0.236 |
Adjusted Analysis
The risk of 30‐day readmission was no different between the intervention and control groups. In patients without an initial ICU stay, and without any ICU stay, charges incurred were no different between the intervention and control groups; LOS was about 11% higher in the localized group as compared to historical controls, and about 9% higher as compared to the concurrent control group. The difference in LOS was not statistically significant on an overall 3‐way comparison (Table 3).
| Localized Teams in Comparison to | |||
|---|---|---|---|
| Historical Control | Concurrent Control | Overall P Value | |
| |||
| 30‐day risk of readmission OR (CI) | 0.85 (0.611.19) | 0.94 (0.651.37) | 0.630 |
| P value | 0.351 | 0.751 | |
| Charges: excluding patients initially admitted to ICU | |||
| % change | 2% higher | 4% lower | 0.367 |
| (CI) | (6% lower to 11% higher) | (12% lower to 5%higher) | |
| P value | 0.572 | 0.427 | |
| Charges: excluding all patients with an ICU stay | |||
| % change | 2% higher | 5% lower | 0.314 |
| (CI) | (6% lower to 10% higher) | (13% lower to 4% higher) | |
| P value | 0.695 | 0.261 | |
| Length of stay: excluding patients initially admitted to ICU | |||
| % change | 11% higher | 9% higher | 0.105 |
| (CI) | (1% to 22% higher) | (3% lower to 21% higher) | |
| P value | 0.038 | 0.138 | |
| Length of stay: excluding all patients with an ICU stay | |||
| % change | 10% higher | 8% higher | 0.133 |
| (CI) | (0% to 22% higher) | (3% lower to 20% higher) | |
| P value | 0.047 | 0.171 | |
Productivity and Workflow Measures
Unadjusted Analyses
The localized teams received fewer pages as compared to concurrently nonlocalized teams. Localized teams had more patient encounters per day and generated more RVUs per day as compared to both historical and concurrent control groups. Physician assistants on localized teams took fewer steps during their work day (Table 4).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| Pages received/day (7 AM6 PM) Median (IQR) | No data | 15 (921) | 28 (12.540) | <0.001 |
| Total encounters/day Median (IQR) | 10 (813) | 12 (1013) | 11 (913) | <0.001 |
| RVU/day | ||||
| Mean (SD) | 19.9 (6.76) | 22.6 (5.6) | 21.2 (6.7) | <0.001 |
| Steps/day Median (IQR) | No data | 4661 (3922 5166) | 5554 (50606544) | <0.001 |
Adjusted Analysis
On adjusting for clustering by physician and day of week, the significant differences in pages received, total patient encounters, and RVUs generated persisted, while the difference in steps walked by PAs was attenuated to a statistically nonsignificant level (Table 5). The increase in RVU productivity was sustained through various periods of hard caps (data not shown).
| Localized Teams in Comparison to | |||
|---|---|---|---|
| Historical Control | Concurrent Control | Overall P Value | |
| |||
| Pages received (7 AM 6 PM) %(CI) | No data | 51% fewer (4854) | |
| P value | P < 0.001 | ||
| Total encounters | 0.89 more | 1.02 more | |
| N (CI) | (0.371.41) | (0.461.58) | |
| P value | P < 0.001 | P < 0.001 | P < 0.001 |
| RVU/day | 2.20 more | 1.36 more | |
| N (CI) | (1.103.29) | (0.172.55) | |
| P value | P < 0.001 | P = 0.024 | P < 0.001 |
| Steps/day | 1186 fewer (791 more to | ||
| N (CI) | No data | 3164 fewer) | |
| P value | P = 0.240 | ||
DISCUSSION
We found that general medical patients admitted to H‐PA teams and localized to a single nursing unit had similar risk of 30‐day readmission and charges, but may have had a higher length of stay compared to historical and concurrent controls. The localized teams received far fewer pages, had more patient encounters, generated more RVUs, and walked less during their work day. Taken together, these findings imply that in our study, localization led to greater team productivity and a possible decrease in hospital efficiency, with no significant impact on readmissions or charges incurred.
The higher productivity was likely mediated by the preferential assignments of more patients to the localized teams, and improvements in workflow (such as fewer pages and fewer steps walked), which allowed them to provide more care with the same resources as the control teams. Kocher and Sahni7 recently pointed out that the healthcare sector has experienced no gains in labor productivity in the past 20 years. Our intervention fits their prescription for redesigning healthcare delivery models to achieve higher productivity.
The possibility of a higher LOS associated with localization was a counterintuitive finding, and similar to that reported by Roy et al.5 We propose 3 hypotheses to explain this:
Selection bias: Higher workload of the localized teams led to compromised efficiency and a higher length of stay (eg, localized teams had fewer observation admissions, more hospitalizations with an ICU stay, and the AMO was asked to preferentially admit patients to localized teams).
Localization provided teams the opportunity to spend more time with their patients (by decreasing nonvalue‐added tasks) and to consequently address more issues before transitioning to outpatient care, or to provide higher quality of care.
Gaming: By having a hard cap on total number of occupied beds, we provided a perverse incentive to the localized teams to retain patients longer to keep assigned beds occupied, thereby delaying new admissions to avoid higher workload.
Our study cannot tell us which of these hypotheses represents the dominant phenomenon that led to this surprising finding. Hypothesis 3 is most worrying, and we suggest that others looking to localize their medical teams consider the possibility of unintended perverse incentives.
Differences were more pronounced between the historical control group and the intervention group, as opposed to the intervention group and concurrent controls. This may have occurred if we contaminated the concurrent control by decreasing the number of units they had to go to, by sequestering 1 unit for the intervention team.
Our report has limitations. It is a nonrandomized, quasi‐experimental investigation using a single institution's administrative databases. Our intervention was small in scale (localizing 2 out of 10 general medical teams on 1 out of 14 nursing units). What impact a wider implementation of localization may have on emergency department throughput and hospital occupancy remains to be studied. Nevertheless, our research is the first report, to our knowledge, investigating a wide variety of outcomes of localizing inpatient medical teams, and adds significantly to the limited research on this topic. It also provides significant operational details for other institutions to use when localizing medical teams.
We conclude that our intervention of localization of medical teams to a single nursing unit led to higher productivity and better workflow, but did not impact readmissions or charges incurred. We caution others designing similar localization interventions to protect against possible perverse incentives for inefficient care.
Acknowledgements
Disclosure: Nothing to report.
Localizing inpatient general medical teams to nursing units has high intuitive validity for improving physician productivity, hospital efficiency, and patient outcomes. Motion or the moving of personnel between tasksso prominent if teams are not localizedis 1 of the 7 wastes in lean thinking.1 In a timemotion study, where hospitalists cared for patients on up to 5 different wards, O'Leary et al2 have reported large parts of hospitalists' workdays spent in indirect patient care (69%), paging (13%), and travel (3%). Localization could increase the amount of time available for direct patient care, decrease time spent for (and interruptions due to) paging, and decrease travel time, all leading to greater productivity.
O'Leary et al3 have also reported the beneficial effects of localization of medical inpatients on communication between nurses and physicians, who could identify each other more often, and reported greater communication (specifically face‐to‐face communication) with each other following localization. This improvement in communication and effective multidisciplinary rounds could lead to safer care4 and better outcomes.
Further investigations about the effect of localization are limited. Roy et al5 have compared the outcomes of patients localized to 2 inpatient pods medically staffed by hospitalists and physician assistants (PAs) to geographically dispersed, but structurally different, house staff teams. They noticed significantly lower costs, slight but nonsignificant increase in length of stay, and no difference in mortality or readmissions, but it is impossible to tease out the affect of localization versus the affect of team composition. In a before‐and‐after study, Findlay et al6 have reported a decrease in mortality and complication rates in clinically homogenous surgical patients (proximal hip fractures) when cared for by junior trainee physicians localized to a unit, but their experience cannot be extrapolated to the much more diverse general medical population.
In our hospital, each general medical team could admit patients dispersed over 14 different units. An internal group, commissioned to evaluate our hospitalist practice, recommended reducing this dispersal to improve physician productivity, hospital efficiency, and outcomes of care. We therefore conducted a project to evaluate the impact of localizing general medical inpatient teams to a single nursing unit.
METHODS
Setting
We conducted our project at a 490 bed, urban academic medical center in the midwestern United States where of the 10 total general medical teams, 6 were traditional resident‐based teams and 4 consisted of a hospitalist paired with a PA (H‐PA teams). We focused our study on the 4 H‐PA teams. The hospitalists could be assigned to any H‐PA team and staffed them for 2 weeks (including weekends). The PAs were always assigned to the same team but took weekends off. An in‐house hospitalist provided overnight cross‐coverage for the H‐PA teams. Prior to our intervention, these teams could admit patients to any of the 14 nursing units at our hospital. They admitted patients from 7 AM to 3 PM, and also accepted care of patients admitted overnight after the resident teams had reached their admission limits (overflow). A Faculty Admitting Medical Officer (AMO) balanced the existing workload of the teams against the number and complexity of incoming patients to decide team assignment for the patients. The AMO was given guidelines (soft caps) to limit total admissions to H‐PA teams to 5 per team per day (3 on a weekend), and to not exceed a total patient census of 16 for an H‐PA team.
Intervention
Starting April 1, 2010, until July 15, 2010, we localized patients admitted to 2 of our 4 H‐PA teams on a single 32‐bed nursing unit. The patients of the other 2 H‐PA teams remained dispersed throughout the hospital.
Transition
April 1, 2010 was a scheduled switch day for the hospitalists on the H‐PA teams. We took advantage of this switch day and reassigned all patients cared for by H‐PA teams on our localized unit to the 2 localized teams. Similarly, all patients on nonlocalized units cared for by H‐PA teams were reassigned to the 2 nonlocalized teams. All patients cared for by resident teams on the localized unit, that were anticipated to be discharged soon, stayed until discharge; those that had a longer stay anticipated were transferred to a nonlocalized unit.
Patient Assignment
The 4 H‐PA teams continued to accept patients between 7 AM and 3 PM, as well as overflow patients. Patients with sickle cell crises were admitted exclusively to the nonlocalized teams, as they were cared for on a specialized nursing unit. No other patient characteristic was used to decide team assignment.
The AMO balanced the existing workload of the teams against the number and complexity of incoming patients to decide team assignment for the patients, but if these factors were equivocal, the AMO was now asked to preferentially admit to the localized teams. The admission soft cap for the H‐PA teams remained the same (5 on weekdays and 3 on weekends). The soft cap on the total census of 16 patients for the nonlocalized teams remained, but we imposed hard caps on the total census for the localized teams. These hard caps were 16 for each localized team for the month of April (to fill a 32‐bed unit), then decreased to 12 for the month of May, as informal feedback from the teams suggested a need to decrease workload, and then rebalanced to 14 for the remaining study period.
Evaluation
Clinical Outcomes
Using both concurrent and historical controls, we evaluated the impact of localization on the following clinical outcome measures: length of stay (LOS), charges, and 30‐day readmission rates.
Inclusion Criteria
We included all patients assigned to localized and nonlocalized teams between the period April 1, 2010 to July 15, 2010, and discharged before July 16, 2010, in our intervention group and concurrent control group, respectively. We included all patients assigned to any of the 4 H‐PA teams during the period January 1, 2010 and March 31, 2010 in the historical control group.
Exclusion Criteria
From the historical control group, we excluded patients assigned to one particular H‐PA team during the period January 1, 2010 to February 28, 2010, during which the PA assigned to that team was on leave. We excluded, from all groups, patients with a diagnosis of sickle cell disease and hospitalizations that straddled the start of the intervention. Further, we excluded repeat admissions for each patient.
Data Collection
We used admission logs to determine team assignment and linked them to our hospital's discharge abstract database to get patient level data. We grouped the principal diagnosis, International Classification of Diseases, Ninth Revision, Clinical Modification (ICD‐9‐CM) codes into clinically relevant categories using the Healthcare Cost and Utilization Project Clinical Classification Software for ICD‐9‐CM (Rockville, MD, www.hcup‐us.ahrq.gov/toolssoftware/ccs/ccs.jsp). We created comorbidity measures using Healthcare Cost and Utilization Project Comorbidity Software, version 3.4 (Rockville, MD, www.hcup‐us.ahrq.gov/toolssoftware/comorbidity/comorbidity.jsp).
We calculated LOS by subtracting the discharge day and time from the admission day and time. We summed all charges accrued during the entire hospital stay, but did not include professional fees. The LOS and charges included time spent and charges accrued in the intensive care unit (ICU). As ICU care was not under the control of the general medical teams and could have a significant impact on outcomes reflecting resource utilization, we compared LOS and charges only for 2 subsets of patients: patients not initially admitted to ICU before care by medical teams, and patients never requiring ICU care. We considered any repeat hospitalization to our hospital within 30 days following a discharge to be a readmission, except those for a planned procedure or for inpatient rehabilitation. We compared readmission rates for all patients irrespective of ICU stay, as discharge planning for all patients was under the direct control of the general medical teams.
Data Analysis
We performed unadjusted descriptive statistics using medians and interquartile ranges for continuous variables, and frequencies and percentages for categorical variables. We used chi‐square tests of association, and KruskalWallis analysis of variance, to compare baseline characteristics of patients assigned to localized and control teams.
We used regression models with random effects to risk adjust for a wide variety of variables. We included age, gender, race, insurance, admission source, time, day of week, discharge time, and total number of comorbidities as fixed effects in all models. We then added individual comorbidity measures one by one as fixed effects, including them only if significant at P < 0.01. We always added a variable identifying the admitting physician as a random effect, to account for dependence between admissions to the same physician. We log transformed LOS and charges because they were extremely skewed in nature. We analyzed readmissions after excluding patients who died. We evaluated the affect of our intervention on clinical outcomes using both historical and concurrent controls. We report P values for both overall 3‐way comparisons, as well as each of the 2‐way comparisonsintervention versus historical control and intervention versus concurrent control.
Productivity and Workflow Measures
We also evaluated the impact of localization on the following productivity and workflow measures: number of pages received, number of patient encounters, relative value units (RVUs) generated, and steps walked by PAs.
Data Collection
We queried our in‐house paging systems for the number of pages received by intervention and concurrent control teams between 7 AM and 6 PM (usual workday). We queried our professional billing data to determine the number of encounters per day and RVUs generated by the intervention, as well as historical and concurrent control teams, as a measure of productivity.
During the last 15 days of our intervention (July 1 July 15, 2010), we received 4 pedometers and we asked the PAs to record the number of steps taken during their workday. We chose PAs, rather than physicians, as the PAs had purely clinical duties and their walking activity would reflect activity for solely clinical purposes.
Data Analysis
For productivity and workflow measures, we adjusted for the day of the week and used random effects models to adjust for clustering of data by physician and physician assistant.
Statistical Software
We performed the statistical analysis using R software, versions 2.9.0 (The R Project for Statistical Computing, Vienna, Austria, http://www.R‐project.org).
Ethical Concerns
The study protocol was approved by our institutional review board.
RESULTS
Study Population
There were 2431 hospitalizations to the 4 H‐PA teams during the study period. Data from 37 hospitalizations was excluded because of missing data. After applying all exclusion criteria, our final study sample consisted of a total of 1826 first hospitalizations for patients: 783 historical controls, 478 concurrent controls, and 565 localized patients.
Patients in the control groups and intervention group were similar in age, gender, race, and insurance status. Patients in the intervention group were more likely to be admitted over the weekend, but had similar probability of being discharged over the weekend or having had an ICU stay. Historical controls were admitted more often between 6 AM and 12 noon, while during the intervention period, patients were more likely to be admitted between midnight and 6 AM. The discharge time was similar across all groups. The 5 most common diagnoses were similar across the groups (Table 1).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| Patients | 783 | 565 | 478 | |
| Age median (IQR) | 57 (4575) | 57 (4573) | 56 (4470) | 0.186 |
| Age groups, n (%) | ||||
| <30 | 65 (8.3) | 37 (6.6) | 46 (9.6) | |
| 3039 | 76 (9.7) | 62 (11.0) | 47 (9.8) | |
| 4049 | 114 (14.6) | 85 (15.0) | 68 (14.2) | |
| 5059 | 162 (20.7) | 124 (22.0) | 118 (24.7) | 0.145 |
| 6069 | 119 (15.2) | 84 (14.9) | 76 (16.0) | |
| 7079 | 100 (12.8) | 62 (11.0) | 58 (12.1) | |
| 8089 | 113 (14.4) | 95 (16.8) | 51 (10.7) | |
| >89 | 34 (4.3) | 16 (2.88) | 14 (2.9) | |
| Female gender, n (%) | 434 (55.4) | 327 (57.9) | 264 (55.2) | 0.602 |
| Race: Black, n (%) | 285 (36.4) | 229 (40.5) | 200 (41.8) | 0.111 |
| Observation status, n (%) | 165 (21.1) | 108 (19.1) | 108 (22.6) | 0.380 |
| Insurance, n (%) | ||||
| Commercial | 171 (21.8) | 101 (17.9) | 101 (21.1) | |
| Medicare | 376 (48.0) | 278 (49.2) | 218 (45.6) | 0.225 |
| Medicaid | 179 (22.8) | 126 (22.3) | 117 (24.5) | |
| Uninsured | 54 (7.3) | 60 (10.6) | 42 (8.8) | |
| Weekend admission, n (%) | 137 (17.5) | 116 (20.5) | 65 (13.6) | 0.013 |
| Weekend discharge, n (%) | 132 (16.9) | 107 (18.9) | 91 (19.0) | 0.505 |
| Source of admission | ||||
| ED, n (%) | 654 (83.5) | 450 (79.7) | 370 (77.4) | 0.022 |
| No ICU stay, n (%) | 600 (76.6) | 440 (77.9) | 383 (80.1) | 0.348 |
| Admission time, n (%) | ||||
| 00000559 | 239 (30.5) | 208 (36.8) | 172 (36.0) | |
| 06001159 | 296 (37.8) | 157 (27.8) | 154 (32.2) | 0.007 |
| 12001759 | 183 (23.4) | 147 (26.0) | 105 (22.0) | |
| 18002359 | 65 (8.3) | 53 (9.4) | 47 (9.8) | |
| Discharge time, n (%) | ||||
| 00001159 | 67 (8.6) | 45 (8.0) | 43 (9.0) | |
| 12001759 | 590 (75.4) | 417 (73.8) | 364 (76.2) | 0.658 |
| 18002359 | 126 (16.1) | 103 (18.2) | 71 (14.9) | |
| Inpatient deaths, n | 13 | 13 | 6 | |
| Top 5 primary diagnoses (%) | ||||
| 1 | Chest pain (11.5) | Chest pain (13.3) | Chest pain (11.9) | |
| 2 | Septicemia (6.4) | Septicemia (5.1) | Septicemia (3.8) | |
| 3 | Diabetes w/cm (4.6) | Pneumonia (4.9) | Diabetes w/cm (3.3) | n/a |
| 4 | Pneumonia (2.8) | Diabetes w/cm (4.1) | Pneumonia (3.3) | |
| 5 | UTI (2.7) | COPD (3.2) | UTI (2.9) | |
Clinical Outcomes
Unadjusted Analyses
The risk of 30‐day readmission was no different between the intervention and control groups. In patients without an initial ICU stay, and without any ICU stay, charges incurred and LOS were no different between the intervention and control groups (Table 2).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| 30‐day readmissions n (%) | 118 (15.3) | 69 (12.5) | 66 (14.0) | 0.346 |
| Charges: excluding patients initially admitted to ICU | ||||
| Median (IQR) in $ | 9346 (621614,520) | 9724 (665715,390) | 9902 (661115,670) | 0.393 |
| Charges: excluding all patients with an ICU stay | ||||
| Median (IQR) in $ | 9270 (618713,990) | 9509 (660114,940) | 9846 (658015,400) | 0.283 |
| Length of stay: excluding patients initially admitted to ICU | ||||
| Median (IQR) in days | 1.81 (1.223.35) | 2.16 (1.214.02) | 1.89 (1.193.50) | 0.214 |
| Length of stay: excluding all patients with an ICU stay | ||||
| Median (IQR) in days | 1.75 (1.203.26) | 2.12 (1.203.74) | 1.84 (1.193.42) | 0.236 |
Adjusted Analysis
The risk of 30‐day readmission was no different between the intervention and control groups. In patients without an initial ICU stay, and without any ICU stay, charges incurred were no different between the intervention and control groups; LOS was about 11% higher in the localized group as compared to historical controls, and about 9% higher as compared to the concurrent control group. The difference in LOS was not statistically significant on an overall 3‐way comparison (Table 3).
| Localized Teams in Comparison to | |||
|---|---|---|---|
| Historical Control | Concurrent Control | Overall P Value | |
| |||
| 30‐day risk of readmission OR (CI) | 0.85 (0.611.19) | 0.94 (0.651.37) | 0.630 |
| P value | 0.351 | 0.751 | |
| Charges: excluding patients initially admitted to ICU | |||
| % change | 2% higher | 4% lower | 0.367 |
| (CI) | (6% lower to 11% higher) | (12% lower to 5%higher) | |
| P value | 0.572 | 0.427 | |
| Charges: excluding all patients with an ICU stay | |||
| % change | 2% higher | 5% lower | 0.314 |
| (CI) | (6% lower to 10% higher) | (13% lower to 4% higher) | |
| P value | 0.695 | 0.261 | |
| Length of stay: excluding patients initially admitted to ICU | |||
| % change | 11% higher | 9% higher | 0.105 |
| (CI) | (1% to 22% higher) | (3% lower to 21% higher) | |
| P value | 0.038 | 0.138 | |
| Length of stay: excluding all patients with an ICU stay | |||
| % change | 10% higher | 8% higher | 0.133 |
| (CI) | (0% to 22% higher) | (3% lower to 20% higher) | |
| P value | 0.047 | 0.171 | |
Productivity and Workflow Measures
Unadjusted Analyses
The localized teams received fewer pages as compared to concurrently nonlocalized teams. Localized teams had more patient encounters per day and generated more RVUs per day as compared to both historical and concurrent control groups. Physician assistants on localized teams took fewer steps during their work day (Table 4).
| Historical Control | Intervention Localized Teams | Concurrent Control | P Value | |
|---|---|---|---|---|
| ||||
| Pages received/day (7 AM6 PM) Median (IQR) | No data | 15 (921) | 28 (12.540) | <0.001 |
| Total encounters/day Median (IQR) | 10 (813) | 12 (1013) | 11 (913) | <0.001 |
| RVU/day | ||||
| Mean (SD) | 19.9 (6.76) | 22.6 (5.6) | 21.2 (6.7) | <0.001 |
| Steps/day Median (IQR) | No data | 4661 (3922 5166) | 5554 (50606544) | <0.001 |
Adjusted Analysis
On adjusting for clustering by physician and day of week, the significant differences in pages received, total patient encounters, and RVUs generated persisted, while the difference in steps walked by PAs was attenuated to a statistically nonsignificant level (Table 5). The increase in RVU productivity was sustained through various periods of hard caps (data not shown).
| Localized Teams in Comparison to | |||
|---|---|---|---|
| Historical Control | Concurrent Control | Overall P Value | |
| |||
| Pages received (7 AM 6 PM) %(CI) | No data | 51% fewer (4854) | |
| P value | P < 0.001 | ||
| Total encounters | 0.89 more | 1.02 more | |
| N (CI) | (0.371.41) | (0.461.58) | |
| P value | P < 0.001 | P < 0.001 | P < 0.001 |
| RVU/day | 2.20 more | 1.36 more | |
| N (CI) | (1.103.29) | (0.172.55) | |
| P value | P < 0.001 | P = 0.024 | P < 0.001 |
| Steps/day | 1186 fewer (791 more to | ||
| N (CI) | No data | 3164 fewer) | |
| P value | P = 0.240 | ||
DISCUSSION
We found that general medical patients admitted to H‐PA teams and localized to a single nursing unit had similar risk of 30‐day readmission and charges, but may have had a higher length of stay compared to historical and concurrent controls. The localized teams received far fewer pages, had more patient encounters, generated more RVUs, and walked less during their work day. Taken together, these findings imply that in our study, localization led to greater team productivity and a possible decrease in hospital efficiency, with no significant impact on readmissions or charges incurred.
The higher productivity was likely mediated by the preferential assignments of more patients to the localized teams, and improvements in workflow (such as fewer pages and fewer steps walked), which allowed them to provide more care with the same resources as the control teams. Kocher and Sahni7 recently pointed out that the healthcare sector has experienced no gains in labor productivity in the past 20 years. Our intervention fits their prescription for redesigning healthcare delivery models to achieve higher productivity.
The possibility of a higher LOS associated with localization was a counterintuitive finding, and similar to that reported by Roy et al.5 We propose 3 hypotheses to explain this:
Selection bias: Higher workload of the localized teams led to compromised efficiency and a higher length of stay (eg, localized teams had fewer observation admissions, more hospitalizations with an ICU stay, and the AMO was asked to preferentially admit patients to localized teams).
Localization provided teams the opportunity to spend more time with their patients (by decreasing nonvalue‐added tasks) and to consequently address more issues before transitioning to outpatient care, or to provide higher quality of care.
Gaming: By having a hard cap on total number of occupied beds, we provided a perverse incentive to the localized teams to retain patients longer to keep assigned beds occupied, thereby delaying new admissions to avoid higher workload.
Our study cannot tell us which of these hypotheses represents the dominant phenomenon that led to this surprising finding. Hypothesis 3 is most worrying, and we suggest that others looking to localize their medical teams consider the possibility of unintended perverse incentives.
Differences were more pronounced between the historical control group and the intervention group, as opposed to the intervention group and concurrent controls. This may have occurred if we contaminated the concurrent control by decreasing the number of units they had to go to, by sequestering 1 unit for the intervention team.
Our report has limitations. It is a nonrandomized, quasi‐experimental investigation using a single institution's administrative databases. Our intervention was small in scale (localizing 2 out of 10 general medical teams on 1 out of 14 nursing units). What impact a wider implementation of localization may have on emergency department throughput and hospital occupancy remains to be studied. Nevertheless, our research is the first report, to our knowledge, investigating a wide variety of outcomes of localizing inpatient medical teams, and adds significantly to the limited research on this topic. It also provides significant operational details for other institutions to use when localizing medical teams.
We conclude that our intervention of localization of medical teams to a single nursing unit led to higher productivity and better workflow, but did not impact readmissions or charges incurred. We caution others designing similar localization interventions to protect against possible perverse incentives for inefficient care.
Acknowledgements
Disclosure: Nothing to report.
- . Reducing waste in US health care systems. JAMA. 2007;297(8):871–874.
- , , . How hospitalists spend their time: insights on efficiency and safety. J Hosp Med. 2006;1(2):88–93.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 2009;24(11):1223–1227.
- , , , et al. Structured interdisciplinary rounds in a medical teaching unit: improving patient safety. Arch Intern Med. 2011;171(7):678–684.
- , , , et al. Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes. J Hosp Med. 2008;3(5):361–368.
- , , , , . Ward‐based rather than team‐based junior surgical doctors reduce mortality for patients with a fracture of the proximal femur: results from a two‐year observational study. J Bone Joint Surg Br. 2011;93‐B(3):393–398.
- , . Rethinking health care labor. N Engl J Med. 2011;365(15):1370–1372.
- . Reducing waste in US health care systems. JAMA. 2007;297(8):871–874.
- , , . How hospitalists spend their time: insights on efficiency and safety. J Hosp Med. 2006;1(2):88–93.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 2009;24(11):1223–1227.
- , , , et al. Structured interdisciplinary rounds in a medical teaching unit: improving patient safety. Arch Intern Med. 2011;171(7):678–684.
- , , , et al. Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes. J Hosp Med. 2008;3(5):361–368.
- , , , , . Ward‐based rather than team‐based junior surgical doctors reduce mortality for patients with a fracture of the proximal femur: results from a two‐year observational study. J Bone Joint Surg Br. 2011;93‐B(3):393–398.
- , . Rethinking health care labor. N Engl J Med. 2011;365(15):1370–1372.
Copyright © 2012 Society of Hospital Medicine
Alcohol Withdrawal Admissions
Many patients are admitted and readmitted to acute care hospitals with alcohol‐related diagnoses, including alcohol withdrawal syndrome (AWS), and experience significant morbidity and mortality. In patients with septic shock or at risk for acute respiratory distress syndrome (ARDS), chronic alcohol abuse is associated with increased ARDS and severity of multiple organ dysfunction.1 Among intensive care unit (ICU) patients, those with alcohol dependence have higher morbidity, including septic shock, and higher hospital mortality.2 Patients who experience AWS as a result of alcohol dependence may experience life‐threatening complications, such as seizures and delirium tremens.3,4 In‐hospital mortality from AWS is historically high,5 but with benzodiazepines used in a symptom‐driven manner to treat the complications of alcohol use, hospital mortality rates are more recently reported at 2.4%.6
As inpatient outcomes1,2,7 and hospital mortality810 are negatively affected by alcohol abuse, the post‐hospital course of these patients is also of interest. Specifically, patients are often admitted and readmitted with alcohol‐related diagnoses or AWS to acute care hospitals, but relatively little quantitative data exist on readmission factors in this population.11 Patients readmitted to detoxification units or alcohol and substance abuse units have been studied, and factors associated with readmission include psychiatric disorder,1217 female gender,14,15 and delay in rehabilitation aftercare.18
These results cannot be generalized to patients with AWS who are admitted and readmitted to acute‐care hospitals. First, patients hospitalized for alcohol withdrawal symptoms are often medically ill with more severe symptoms, and more frequent coexisting medical and psychiatric illnesses, that complicate the withdrawal syndrome. Detoxification units and substance abuse units require patients to be medically stable before admission, because they do not have the ability to provide a high level of supervision and treatment. Second, much of what we know regarding risk factors for readmission to detoxification centers and substance abuse units comes from studies of administrative data of the Veterans Health Administration,12,13 Medicare Provider Analysis and Review file,16 privately owned outpatient substance abuse centers,14 and publicly funded detoxification centers.18 These results may be difficult to generalize to other patient populations. Accordingly, the objective of this study was to identify demographic and clinical factors associated with multiple admissions to a general medicine service for treatment of AWS over a 3‐year period. Characterization of these high‐risk patients and their hospital course may help focus intervention and reduce these revolving door admissions.
METHODS
The Mayo Clinic Institutional Review Board deemed the study exempt.
Patient Selection
The study was conducted at an 1157‐bed academic tertiary referral hospital, located in the Midwest, that has approximately 15,000 inpatient admissions to general medicine services annually and serves as the main referral center for the region. Patients included in this study were adults admitted to general medicine services and treated with symptom‐triggered Clinical Institute Withdrawal AssessmentAlcohol Revised (CIWA‐Ar) protocol19 between January 1, 2006 and December 31, 2008. Patients were identified using the Mayo Clinic Quality Improvement Resource Center database, as done in a previous CIWA‐Ar study.20 Patients were excluded if the primary diagnosis was a nonalcohol‐related diagnosis (Figure 1).
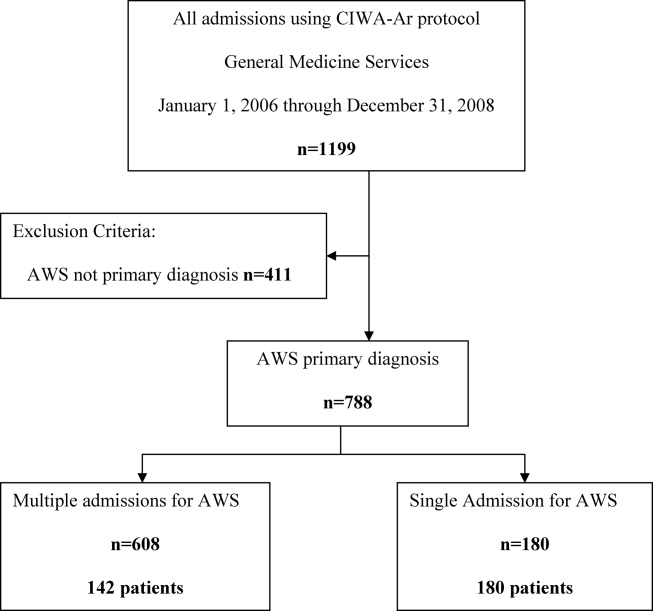
Patients were placed in 1 of 2 groups based on number of admissions during the study period, either a single‐admission group or a multiple‐admissions group. While most readmission studies use a 30‐day mark from discharge, we used 3 years to better capture relapse and recidivism in this patient population. The 2 groups were then compared retrospectively. To insure that a single admission was not arbitrarily created by the December 2008 cutoff, we reviewed the single‐admission group for additional admissions through June of 2009. If a patient did have a subsequent admission, then the patient was moved to the multiple‐admissions group.
Clinical Variables
Demographic and clinical data was obtained using the Mayo Data Retrieval System (DRS), the Mayo Clinic Life Sciences System (MCLSS) database, and electronic medical records. Clinical data for the multiple‐admissions group was derived from the first admission of the study period, and subsequently referred to as index admission. Specific demographic information collected included age, race, gender, marital status, employment status, and education. Clinical data collected included admitting diagnosis, comorbid medical disorders, psychiatric disorders, and CIWA‐Ar evaluations including highest total score (CIWA‐Ar score [max] and component scores). The CIWA‐Ar protocol is a scale to assess the severity of alcohol withdrawal, based on 10 symptoms of alcohol withdrawal ranging from 0 (not present) to 7 (extremely severe). The protocol requires the scale to be administered hourly, and total scores guide the medication dosing and administration of benzodiazepines to control withdrawal symptoms. Laboratory data collected included serum ammonia, alanine aminotransferase(ALT), and admission urine drug screen. For the purposes of this study, a urine drug screen was considered positive if a substance other than alcohol was present. Length of stay (LOS) and adverse events during hospitalization (delirium tremens, intubations, rapid response team [RRT] calls, ICU transfers, and in‐hospital mortality) were also collected.
Medical comorbidity was measured using the Charlson Comorbidity Index (CCI).21 The CCI was scored electronically using diagnoses in the institution's medical index database dating back 5 years from patient's first, or index, admission. Originally validated as a prognostic tool for mortality 1 year after admission in medical patients, the CCI was chosen as it accounts for most medical comorbidities.21 Data was validated, by another investigator not involved in the initial abstracting process, by randomly verifying 5% of the abstracted data.
Statistical Analysis
Standard descriptive statistics were used for patient characteristics and demographics. Comparing the multiple‐admissions group and single‐admission group, categorical variables were evaluated using the Fisher exact test or Pearson chi‐square test. Continuous variables were evaluated using 2‐sample t test. Multivariate logistic model analyses with stepwise elimination method were used to identify risk factors that were associated with multiple admissions. Age, gender, and variables that were statistically significant in the univariate analysis were used in stepwise regression to get to the final model. A P value of 0.05 was considered statistically significant. All statistical analyses were performed using SAS version 9.3 software (SAS Institute, Cary, NC).
RESULTS
The CIWA‐Ar protocol was ordered on 1199 admissions during the study period. Of these, 411 (34.3%) admissions were excluded because AWS was not the primary diagnosis, leaving 788 (65.7%) admissions for 322 patients, which formed the study population. Of the 322 patients, 180 (56%) had a single admission and 142 (44%) had multiple admissions.
Univariate analyses of demographic and clinical variables are shown in Tables 1 and 2, respectively. Patients with multiple admissions were more likely divorced (P = 0.028), have a high school education or less (P = 0.002), have a higher CCI score (P < 0.0001), a higher CIWA‐Ar score (max) (P < 0.0001), a higher ALT level (P = 0.050), more psychiatric comorbidity (P < 0.026), and a positive urine drug screen (P < 0.001). Adverse events were not significantly different between the 2 groups (Table 2).
| Variable | Single Admission N = 180 | Multiple Admissions N = 142 | P Value |
|---|---|---|---|
| |||
| Age, years (SD) | 47.85 (12.84) | 45.94 (12) | 0.170 |
| Male, No. (%) | 122 (68) | 109 (77) | 0.080 |
| Race/Ethnicity, No. (%) | 0.270 | ||
| White | 168 (93) | 132 (93) | |
| African American | 6 (3) | 3 (2) | |
| Asian | 0 (0) | 1 (1) | |
| Middle Eastern | 3 (2) | 0 (0) | |
| Other | 3 (2) | 6 (4) | |
| Relationship status, No. (%) | 0.160 | ||
| Divorced | 49 (27) | 55 (39) | 0.028* |
| Married | 54 (30) | 34 (24) | 0.230 |
| Separated | 9 (5) | 4 (3) | 0.323 |
| Single | 59 (33) | 38 (27) | 0.243 |
| Widowed | 5 (3) | 3 (2) | 0.703 |
| Committed | 4 (2) | 7 (5) | 0.188 |
| Unknown | 0 (0) | 1 (1) | 0.259 |
| Education, No. (%) | 0.002* | ||
| High school graduate, GED, or less | 49 (28) | 67 (47) | |
| Some college or above | 89 (49) | 60 (42) | |
| Unknown | 41 (23) | 15 (11) | |
| Employment, No. (%) | 0.290 | ||
| Retired | 26 (14) | 12 (8) | |
| Employed | 72 (40) | 51 (36) | |
| Unemployed | 51 (28) | 51 (36) | |
| Homemaker | 9 (5) | 4 (3) | |
| Work disabled | 20 (11) | 23 (16) | |
| Student | 1 (1) | 0 (0) | |
| Unknown | 1 (1) | 1 (1) | |
| Variable | Single Admission N = 180 | Multiple Admissions N = 142 | P Value |
|---|---|---|---|
| |||
| LOS, mean (SD) | 3.71 (7.10) | 2.72 (3.40) | 0.130 |
| Charlson Comorbidity Index, mean (SD) | 1.7 (2.23) | 2.51 (2.90) | 0.005* |
| Medical comorbidity, No. (%) | |||
| Diabetes mellitus | 6 (3) | 16 (11) | 0.005* |
| Cardiovascular disease | 6 (3) | 15 (11) | 0.050* |
| Cerebrovascular disease | 0 (0) | 3 (2) | 0.009* |
| Hypertension | 53 (30) | 36 (25) | 0.400 |
| Cancer | 17 (7) | 10 (9) | 0.440 |
| Psychiatric comorbidity, No. (%) | 97 (54) | 94 (66) | 0.026* |
| Adjustment disorder | 0 (0) | 6 (4) | 0.005* |
| Depressive disorder | 85 (47) | 76 (54) | 0.260 |
| Bipolar disorder | 6 (3) | 10 (7) | 0.130 |
| Psychotic disorder | 4 (2) | 6 (4) | 0.030* |
| Anxiety disorder | 30 (17) | 25 (18) | 0.820 |
| Drug abuse | 4 (2) | 4 (3) | 0.730 |
| Eating disorder | 0 (0) | 3 (2) | 0.050* |
| CIWA‐Ar scores | |||
| CIWA‐Ar score (max), mean (SD) | 15 (8) | 20 (9) | <0.000* |
| Component, mean (SD) | |||
| Agitation | 20 (11) | 36 (25) | 0.001* |
| Anxiety | 23 (13) | 38 (27) | 0.001* |
| Auditory disturbance | 4 (2) | 9 (6) | 0.110 |
| Headache | 11 (6) | 26 (18) | 0.001* |
| Nausea/vomiting | 5 (3) | 17 (12) | 0.003* |
| Orientation | 52 (29) | 72 (51) | 0.001* |
| Paroxysm/sweats | 9 (5) | 17 (12) | 0.023* |
| Tactile disturbance | 25 (14) | 54 (38) | 0.001* |
| Tremor | 35 (19) | 47 (33) | 0.004* |
| Visual disturbance | 54 (30) | 77 (54) | 0.001* |
| ALT (U/L), mean (SD) | 76 (85) | 101 (71) | 0.050* |
| Ammonia (mcg N/dl), mean (SD) | 25 (14) | 29 (29) | 0.530 |
| Positive urine drug screen, No. (%) | 25 (14) | 49 (35) | <0.001* |
| Tetrahydrocannabinol | 14 (56) | 19 (39) | |
| Cocaine | 8 (32) | 8 (16) | |
| Benzodiazepine | 6 (24) | 11 (22) | |
| Opiate | 4 (16) | 13 (26) | |
| Amphetamine | 2 (8) | 2 (4) | |
| Barbiturate | 1 (4) | 0 (0) | |
| Adverse event, No. (%) | |||
| RRT | 1 (1) | 1 (1) | 0.866 |
| ICU transfer | 32 (18) | 20 (14) | 0.550 |
| Intubation | 12 (7) | 4 (3) | 0.890 |
| Delirium tremens | 7 (4) | 4 (3) | 0.600 |
| In‐hospital mortality | 0 (0) | 0 (0) | |
Multivariate logistic model analysis was performed using the variables age, male gender, divorced marital status, high school education or less, CIWA‐Ar score (max), CCI score, psychiatric comorbidity, and positive urine drug screen. With a stepwise elimination process, the final model showed that multiple admissions were associated with high school education or less (P = 0.0071), higher CCI score (P = 0.0010), higher CIWA‐Ar score (max) (P < 0.0001), a positive urine drug screen (P = 0.0002), and psychiatric comorbidity (P = 0.0303) (Table 3).
| Variable | Adjusted Odds Ratio (95% CI) | P Value |
|---|---|---|
| ||
| High school education or less | 2.074 (1.219, 3.529) | 0.0071* |
| CIWA‐Ar score (max) | 1.074 (1.042, 1.107) | <0.0001* |
| Charlson Comorbidity Index | 1.232 (1.088, 1.396) | 0.0010* |
| Psychiatric comorbidity | 1.757 (1.055, 2.928) | 0.0303* |
| Positive urine drug screen | 3.180 (1.740, 5.812) | 0.0002* |
DISCUSSION
We provide important information regarding identification of individuals at high risk for multiple admissions to general medicine services for treatment of AWS. This study found that patients with multiple admissions for AWS had more medical comorbidity. They had more cases of diabetes mellitus, cardiovascular disease, and cerebrovascular disease, and their CCI scores were higher. They also had higher CIWA‐Ar (max) scores, as well as higher CIWA‐Ar component scores, indicating a more severe withdrawal.
Further, psychiatric comorbidity was also associated with multiple admissions. Consistent with the high prevalence in alcoholic patients, psychiatric comorbidity was common in both patients with a single admission and multiple admissions. We also found that a positive urine drug screen was associated with multiple admissions. Interestingly, few patients in each group had a diagnosis recorded in the medical record of an additional substance abuse disorder, yet 14% of patients with a single admission and 29% of patients with multiple admissions had a positive urine drug screen for a non‐alcohol substance. Psychiatric comorbidity, including additional substance abuse, is a well‐established risk factor for readmission to detoxification centers.1215, 17,22,23 Also, people with either an alcohol or non‐alcohol drug addiction, are known to be 7 times more likely to have another addiction than the rest of the population.24 This study suggests clinicians may underrecognize additional substance abuse disorders which are common in this patient population.
In contrast to studies of patients readmitted to detoxification units and substance abuse units,14,15,18,22 we found level of education, specifically a high school education or less, to be associated with multiple admissions. In a study of alcoholics, Greenfield and colleagues found that lower education in alcoholics predicts shorter time to relapse.25 A lack of education may result in inadequate healthcare literacy. Poor health behavior choices that follow may lead to relapse and subsequent admissions for AWS. With respect to other demographic variables, patients in our study population were predominantly men, which is not surprising. Gender differences in alcoholism are well established, with alcohol abuse and dependence more prevalent in men.26 We did not find gender associated with multiple admissions.
Our findings have management and treatment implications. First, providers who care for patients with AWS should not simply focus on treating withdrawal signs and symptoms, but also screen for and address other medical issues, which may not be apparent or seem stable at first glance. While a comorbid medical condition may not be the primary reason for hospital admission, comorbid medical conditions are known to be a source of psychological distress27 and have a negative effect on abstinence. Second, all patients should be screened for additional substance abuse. Initial laboratory testing should include a urine drug screen. Third, before discharging the patient, providers should establish primary care follow‐up for continued surveillance of medical issues. There is evidence that primary care services are predictive of better substance abuse treatment outcomes for those with medical problems.28,29
Finally, inpatient psychiatric consultation, upon admission, is essential for several reasons. First, the psychiatric team can help with initial management and treatment of the alcohol withdrawal regardless of stage and severity, obtain a more comprehensive psychiatric history, and assess for the presence of psychiatric comorbidities that may contribute to, aggravate, or complicate the clinical picture. The team can also address other substance abuse issues when detected by drug screen or clinical history. The psychiatric team, along with chemical‐dependency counselors and social workers, can provide valuable input regarding chemical‐dependency resources available on discharge and help instruct the patient in healthy behaviors. Because healthcare illiteracy may be an issue in this patient population, these instructions should be tailored to the patient's educational level. Prior to discharge, the psychiatry team, social workers, or chemical‐dependency counselors can also assist with, or arrange, rehabilitation aftercare for patients. Recent work shows that patients were less likely to be readmitted to crisis detoxification if they entered rehabilitation care within 3 days of discharge.18
Our study has significant limitations. This study was performed with data at a single academic medical center with an ethnically homogeneous patient population, limiting the external validity of its results. Because this is a retrospective study, data analyses are limited by the quality and accuracy of data in the electronic medical record. Also, our follow‐up period may not have been long enough to detect additional admissions, and we did not screen for patient admissions prior to the study period. By limiting data collection to admissions for AWS to general medical services, we may have missed cases of AWS when admitted for other reasons or to subspecialty services, and we may have missed severe cases requiring admission to an intensive care unit. While we believe we were able to capture most admissions, we may underreport this number since we cannot account for those events that may have occurred at other facilities and locations. Lastly, without a control group, this study is limited in its ability to show an association between any variable and readmission.
In our study, 142 patients accounted for 608 admissions during the 3‐year study period, which speaks to the high recidivism rates for patients with AWS. This disease is associated with high morbidity, high medical costs, and high utilization of healthcare. Our study provides insight regarding identification of patients at high risk of multiple admissions with respect to demographic (lower level of education) and clinical characteristics (worse withdrawal severity, more medical and psychiatric comorbidity, and polysubstance abuse). We believe collaboration between social services, chemical‐dependency counselors, psychiatry, and medicine is necessary to effectively treat this population of patients and assist with the crucial transition to the outpatient setting. Future studies should include key social factors, such as health literacy in the readmission risk assessment, as well as primary care follow‐up and rehabilitation aftercare.
- , . Chronic alcohol abuse, acute respiratory distress syndrome, and multiple organ dysfunction. Crit Care Med. 2003;31(4 suppl):S207–S212.
- , , et al. Alcohol dependence is independently associated with sepsis, septic shock, and hospital mortality among adult intensive care unit patients. Crit Care Med. 2007;35(2):345–350.
- , , . Use of an objective clinical scale in the assessment and management of alcohol withdrawal in a large general hospital. Alcohol Clin Exp Res. 1988;12(3):360–364.
- , . Alcohol withdrawal syndromes in the intensive care unit. Crit Care Med. 2010;38(9 suppl):S494–S501.
- , Alcoholism at the Boston City Hospital—V. The causes of death among alcoholic patients at the Haymarket Square Relief Station, 1923–year="1938"1938. N Engl J Med. year="1939"1939;221(July 13):58–59.
- , , . Epidemiology of alcohol withdrawal syndrome: mortality and factors of poor prognosis [in Spanish]. An Med Interna (Madrid). 2006;23(7):307–309.
- , , . The impact of alcohol‐related diagnoses on pneumonia outcomes. Arch Intern Med. 1997;157(13):1446–1452.
- , , , , , . Analysis of the factors determining survival of alcoholic withdrawal syndrome patients in a general hospital. Alcohol Alcohol. 2010;45(2):151–158.
- , , , , . Predictors of mortality in patients with delirium tremens. Acad Emerg Med. 2008;15(8):788–790.
- , , , . Long‐term mortality of patients admitted to the hospital with alcohol withdrawal syndrome. Alcohol Clin Exp Res. 2011;35(6):1180–1186.
- , , , , , . Substance use treatment barriers for patients with frequent hospital admissions. J Subst Abuse Treat. 2010;38(1):22–30.
- , , . Diagnostic subgroups and predictors of one‐year re‐admission among late‐middle‐aged and older substance abuse patients. J Stud Alcohol. 1994;55(2):173–183.
- , , . Rates and predictors of four‐year readmission among late‐middle‐aged and older substance abuse patients. J Stud Alcohol. 1994;55(5):561–570.
- , , . Readmission among chemical dependency patients in private, outpatient treatment: patterns, correlates and role in long‐term outcome. J Stud Alcohol. 2005;66(6):842–847.
- , , , . Predicting readmission to substance abuse treatment using state information systems. The impact of client and treatment characteristics. J Subst Abuse. 2000;12(3):255–270.
- , , , . Elderly Medicare inpatients with substance use disorders: characteristics and predictors of hospital readmissions over a four‐year interval. J Stud Alcohol. 2000;61(6):891–895.
- , . The role of psychiatric comorbidity in the prediction of readmission for detoxification. Compr Psychiatry. 1998;39(3):129–136.
- , , , , , . Factors associated with frequent utilization of crisis substance use detoxification services. J Addict Dis. 2011;30(2):116–122.
- , , , , . Assessment of alcohol withdrawal: the revised Clinical Institute Withdrawal Assessment for Alcohol scale (CIWA‐Ar). Br J Addict. 1989;84(11):1353–1357.
- , , , Inappropriate use of symptom‐triggered therapy for alcohol withdrawal in the general hospital. Mayo Clin Proc. 2008;83(3):274–279.
- , , , . A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , . A four‐year follow‐up study of male alcoholics: factors affecting the risk of readmission. Alcohol. 2002;27(2):83–88.
- , , , et al. Diagnosis and hospital readmission rates of female veterans with substance‐related disorders. Psychiatr Serv. 1995;46(9):932–937.
- , , , et al. Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiologic Catchment Area (ECA) Study. JAMA. 1990;264(19):2511–2518.
- , , , , , . The relationship between educational attainment and relapse among alcohol‐dependent men and women: a prospective study. Alcohol Clin Exp Res. 2003;27(8):1278–1285.
- , , , . Prevalence, correlates, disability, and comorbidity of DSM‐IV alcohol abuse and dependence in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry. 2007;64(7):830–842.
- , . Health‐related quality of life among adults with serious psychological distress and chronic medical conditions. Qual Life Res. 2008;17(4):521–528.
- , , , , . Primary medical care and reductions in addiction severity: a prospective cohort study. Addiction. 2005;100(1):70–78.
- , , , . The role of medical conditions and primary care services in 5‐year substance use outcomes among chemical dependency treatment patients. Drug Alcohol Depend. 2008;98(1–2):45–53.
Many patients are admitted and readmitted to acute care hospitals with alcohol‐related diagnoses, including alcohol withdrawal syndrome (AWS), and experience significant morbidity and mortality. In patients with septic shock or at risk for acute respiratory distress syndrome (ARDS), chronic alcohol abuse is associated with increased ARDS and severity of multiple organ dysfunction.1 Among intensive care unit (ICU) patients, those with alcohol dependence have higher morbidity, including septic shock, and higher hospital mortality.2 Patients who experience AWS as a result of alcohol dependence may experience life‐threatening complications, such as seizures and delirium tremens.3,4 In‐hospital mortality from AWS is historically high,5 but with benzodiazepines used in a symptom‐driven manner to treat the complications of alcohol use, hospital mortality rates are more recently reported at 2.4%.6
As inpatient outcomes1,2,7 and hospital mortality810 are negatively affected by alcohol abuse, the post‐hospital course of these patients is also of interest. Specifically, patients are often admitted and readmitted with alcohol‐related diagnoses or AWS to acute care hospitals, but relatively little quantitative data exist on readmission factors in this population.11 Patients readmitted to detoxification units or alcohol and substance abuse units have been studied, and factors associated with readmission include psychiatric disorder,1217 female gender,14,15 and delay in rehabilitation aftercare.18
These results cannot be generalized to patients with AWS who are admitted and readmitted to acute‐care hospitals. First, patients hospitalized for alcohol withdrawal symptoms are often medically ill with more severe symptoms, and more frequent coexisting medical and psychiatric illnesses, that complicate the withdrawal syndrome. Detoxification units and substance abuse units require patients to be medically stable before admission, because they do not have the ability to provide a high level of supervision and treatment. Second, much of what we know regarding risk factors for readmission to detoxification centers and substance abuse units comes from studies of administrative data of the Veterans Health Administration,12,13 Medicare Provider Analysis and Review file,16 privately owned outpatient substance abuse centers,14 and publicly funded detoxification centers.18 These results may be difficult to generalize to other patient populations. Accordingly, the objective of this study was to identify demographic and clinical factors associated with multiple admissions to a general medicine service for treatment of AWS over a 3‐year period. Characterization of these high‐risk patients and their hospital course may help focus intervention and reduce these revolving door admissions.
METHODS
The Mayo Clinic Institutional Review Board deemed the study exempt.
Patient Selection
The study was conducted at an 1157‐bed academic tertiary referral hospital, located in the Midwest, that has approximately 15,000 inpatient admissions to general medicine services annually and serves as the main referral center for the region. Patients included in this study were adults admitted to general medicine services and treated with symptom‐triggered Clinical Institute Withdrawal AssessmentAlcohol Revised (CIWA‐Ar) protocol19 between January 1, 2006 and December 31, 2008. Patients were identified using the Mayo Clinic Quality Improvement Resource Center database, as done in a previous CIWA‐Ar study.20 Patients were excluded if the primary diagnosis was a nonalcohol‐related diagnosis (Figure 1).

Patients were placed in 1 of 2 groups based on number of admissions during the study period, either a single‐admission group or a multiple‐admissions group. While most readmission studies use a 30‐day mark from discharge, we used 3 years to better capture relapse and recidivism in this patient population. The 2 groups were then compared retrospectively. To insure that a single admission was not arbitrarily created by the December 2008 cutoff, we reviewed the single‐admission group for additional admissions through June of 2009. If a patient did have a subsequent admission, then the patient was moved to the multiple‐admissions group.
Clinical Variables
Demographic and clinical data was obtained using the Mayo Data Retrieval System (DRS), the Mayo Clinic Life Sciences System (MCLSS) database, and electronic medical records. Clinical data for the multiple‐admissions group was derived from the first admission of the study period, and subsequently referred to as index admission. Specific demographic information collected included age, race, gender, marital status, employment status, and education. Clinical data collected included admitting diagnosis, comorbid medical disorders, psychiatric disorders, and CIWA‐Ar evaluations including highest total score (CIWA‐Ar score [max] and component scores). The CIWA‐Ar protocol is a scale to assess the severity of alcohol withdrawal, based on 10 symptoms of alcohol withdrawal ranging from 0 (not present) to 7 (extremely severe). The protocol requires the scale to be administered hourly, and total scores guide the medication dosing and administration of benzodiazepines to control withdrawal symptoms. Laboratory data collected included serum ammonia, alanine aminotransferase(ALT), and admission urine drug screen. For the purposes of this study, a urine drug screen was considered positive if a substance other than alcohol was present. Length of stay (LOS) and adverse events during hospitalization (delirium tremens, intubations, rapid response team [RRT] calls, ICU transfers, and in‐hospital mortality) were also collected.
Medical comorbidity was measured using the Charlson Comorbidity Index (CCI).21 The CCI was scored electronically using diagnoses in the institution's medical index database dating back 5 years from patient's first, or index, admission. Originally validated as a prognostic tool for mortality 1 year after admission in medical patients, the CCI was chosen as it accounts for most medical comorbidities.21 Data was validated, by another investigator not involved in the initial abstracting process, by randomly verifying 5% of the abstracted data.
Statistical Analysis
Standard descriptive statistics were used for patient characteristics and demographics. Comparing the multiple‐admissions group and single‐admission group, categorical variables were evaluated using the Fisher exact test or Pearson chi‐square test. Continuous variables were evaluated using 2‐sample t test. Multivariate logistic model analyses with stepwise elimination method were used to identify risk factors that were associated with multiple admissions. Age, gender, and variables that were statistically significant in the univariate analysis were used in stepwise regression to get to the final model. A P value of 0.05 was considered statistically significant. All statistical analyses were performed using SAS version 9.3 software (SAS Institute, Cary, NC).
RESULTS
The CIWA‐Ar protocol was ordered on 1199 admissions during the study period. Of these, 411 (34.3%) admissions were excluded because AWS was not the primary diagnosis, leaving 788 (65.7%) admissions for 322 patients, which formed the study population. Of the 322 patients, 180 (56%) had a single admission and 142 (44%) had multiple admissions.
Univariate analyses of demographic and clinical variables are shown in Tables 1 and 2, respectively. Patients with multiple admissions were more likely divorced (P = 0.028), have a high school education or less (P = 0.002), have a higher CCI score (P < 0.0001), a higher CIWA‐Ar score (max) (P < 0.0001), a higher ALT level (P = 0.050), more psychiatric comorbidity (P < 0.026), and a positive urine drug screen (P < 0.001). Adverse events were not significantly different between the 2 groups (Table 2).
| Variable | Single Admission N = 180 | Multiple Admissions N = 142 | P Value |
|---|---|---|---|
| |||
| Age, years (SD) | 47.85 (12.84) | 45.94 (12) | 0.170 |
| Male, No. (%) | 122 (68) | 109 (77) | 0.080 |
| Race/Ethnicity, No. (%) | 0.270 | ||
| White | 168 (93) | 132 (93) | |
| African American | 6 (3) | 3 (2) | |
| Asian | 0 (0) | 1 (1) | |
| Middle Eastern | 3 (2) | 0 (0) | |
| Other | 3 (2) | 6 (4) | |
| Relationship status, No. (%) | 0.160 | ||
| Divorced | 49 (27) | 55 (39) | 0.028* |
| Married | 54 (30) | 34 (24) | 0.230 |
| Separated | 9 (5) | 4 (3) | 0.323 |
| Single | 59 (33) | 38 (27) | 0.243 |
| Widowed | 5 (3) | 3 (2) | 0.703 |
| Committed | 4 (2) | 7 (5) | 0.188 |
| Unknown | 0 (0) | 1 (1) | 0.259 |
| Education, No. (%) | 0.002* | ||
| High school graduate, GED, or less | 49 (28) | 67 (47) | |
| Some college or above | 89 (49) | 60 (42) | |
| Unknown | 41 (23) | 15 (11) | |
| Employment, No. (%) | 0.290 | ||
| Retired | 26 (14) | 12 (8) | |
| Employed | 72 (40) | 51 (36) | |
| Unemployed | 51 (28) | 51 (36) | |
| Homemaker | 9 (5) | 4 (3) | |
| Work disabled | 20 (11) | 23 (16) | |
| Student | 1 (1) | 0 (0) | |
| Unknown | 1 (1) | 1 (1) | |
| Variable | Single Admission N = 180 | Multiple Admissions N = 142 | P Value |
|---|---|---|---|
| |||
| LOS, mean (SD) | 3.71 (7.10) | 2.72 (3.40) | 0.130 |
| Charlson Comorbidity Index, mean (SD) | 1.7 (2.23) | 2.51 (2.90) | 0.005* |
| Medical comorbidity, No. (%) | |||
| Diabetes mellitus | 6 (3) | 16 (11) | 0.005* |
| Cardiovascular disease | 6 (3) | 15 (11) | 0.050* |
| Cerebrovascular disease | 0 (0) | 3 (2) | 0.009* |
| Hypertension | 53 (30) | 36 (25) | 0.400 |
| Cancer | 17 (7) | 10 (9) | 0.440 |
| Psychiatric comorbidity, No. (%) | 97 (54) | 94 (66) | 0.026* |
| Adjustment disorder | 0 (0) | 6 (4) | 0.005* |
| Depressive disorder | 85 (47) | 76 (54) | 0.260 |
| Bipolar disorder | 6 (3) | 10 (7) | 0.130 |
| Psychotic disorder | 4 (2) | 6 (4) | 0.030* |
| Anxiety disorder | 30 (17) | 25 (18) | 0.820 |
| Drug abuse | 4 (2) | 4 (3) | 0.730 |
| Eating disorder | 0 (0) | 3 (2) | 0.050* |
| CIWA‐Ar scores | |||
| CIWA‐Ar score (max), mean (SD) | 15 (8) | 20 (9) | <0.000* |
| Component, mean (SD) | |||
| Agitation | 20 (11) | 36 (25) | 0.001* |
| Anxiety | 23 (13) | 38 (27) | 0.001* |
| Auditory disturbance | 4 (2) | 9 (6) | 0.110 |
| Headache | 11 (6) | 26 (18) | 0.001* |
| Nausea/vomiting | 5 (3) | 17 (12) | 0.003* |
| Orientation | 52 (29) | 72 (51) | 0.001* |
| Paroxysm/sweats | 9 (5) | 17 (12) | 0.023* |
| Tactile disturbance | 25 (14) | 54 (38) | 0.001* |
| Tremor | 35 (19) | 47 (33) | 0.004* |
| Visual disturbance | 54 (30) | 77 (54) | 0.001* |
| ALT (U/L), mean (SD) | 76 (85) | 101 (71) | 0.050* |
| Ammonia (mcg N/dl), mean (SD) | 25 (14) | 29 (29) | 0.530 |
| Positive urine drug screen, No. (%) | 25 (14) | 49 (35) | <0.001* |
| Tetrahydrocannabinol | 14 (56) | 19 (39) | |
| Cocaine | 8 (32) | 8 (16) | |
| Benzodiazepine | 6 (24) | 11 (22) | |
| Opiate | 4 (16) | 13 (26) | |
| Amphetamine | 2 (8) | 2 (4) | |
| Barbiturate | 1 (4) | 0 (0) | |
| Adverse event, No. (%) | |||
| RRT | 1 (1) | 1 (1) | 0.866 |
| ICU transfer | 32 (18) | 20 (14) | 0.550 |
| Intubation | 12 (7) | 4 (3) | 0.890 |
| Delirium tremens | 7 (4) | 4 (3) | 0.600 |
| In‐hospital mortality | 0 (0) | 0 (0) | |
Multivariate logistic model analysis was performed using the variables age, male gender, divorced marital status, high school education or less, CIWA‐Ar score (max), CCI score, psychiatric comorbidity, and positive urine drug screen. With a stepwise elimination process, the final model showed that multiple admissions were associated with high school education or less (P = 0.0071), higher CCI score (P = 0.0010), higher CIWA‐Ar score (max) (P < 0.0001), a positive urine drug screen (P = 0.0002), and psychiatric comorbidity (P = 0.0303) (Table 3).
| Variable | Adjusted Odds Ratio (95% CI) | P Value |
|---|---|---|
| ||
| High school education or less | 2.074 (1.219, 3.529) | 0.0071* |
| CIWA‐Ar score (max) | 1.074 (1.042, 1.107) | <0.0001* |
| Charlson Comorbidity Index | 1.232 (1.088, 1.396) | 0.0010* |
| Psychiatric comorbidity | 1.757 (1.055, 2.928) | 0.0303* |
| Positive urine drug screen | 3.180 (1.740, 5.812) | 0.0002* |
DISCUSSION
We provide important information regarding identification of individuals at high risk for multiple admissions to general medicine services for treatment of AWS. This study found that patients with multiple admissions for AWS had more medical comorbidity. They had more cases of diabetes mellitus, cardiovascular disease, and cerebrovascular disease, and their CCI scores were higher. They also had higher CIWA‐Ar (max) scores, as well as higher CIWA‐Ar component scores, indicating a more severe withdrawal.
Further, psychiatric comorbidity was also associated with multiple admissions. Consistent with the high prevalence in alcoholic patients, psychiatric comorbidity was common in both patients with a single admission and multiple admissions. We also found that a positive urine drug screen was associated with multiple admissions. Interestingly, few patients in each group had a diagnosis recorded in the medical record of an additional substance abuse disorder, yet 14% of patients with a single admission and 29% of patients with multiple admissions had a positive urine drug screen for a non‐alcohol substance. Psychiatric comorbidity, including additional substance abuse, is a well‐established risk factor for readmission to detoxification centers.1215, 17,22,23 Also, people with either an alcohol or non‐alcohol drug addiction, are known to be 7 times more likely to have another addiction than the rest of the population.24 This study suggests clinicians may underrecognize additional substance abuse disorders which are common in this patient population.
In contrast to studies of patients readmitted to detoxification units and substance abuse units,14,15,18,22 we found level of education, specifically a high school education or less, to be associated with multiple admissions. In a study of alcoholics, Greenfield and colleagues found that lower education in alcoholics predicts shorter time to relapse.25 A lack of education may result in inadequate healthcare literacy. Poor health behavior choices that follow may lead to relapse and subsequent admissions for AWS. With respect to other demographic variables, patients in our study population were predominantly men, which is not surprising. Gender differences in alcoholism are well established, with alcohol abuse and dependence more prevalent in men.26 We did not find gender associated with multiple admissions.
Our findings have management and treatment implications. First, providers who care for patients with AWS should not simply focus on treating withdrawal signs and symptoms, but also screen for and address other medical issues, which may not be apparent or seem stable at first glance. While a comorbid medical condition may not be the primary reason for hospital admission, comorbid medical conditions are known to be a source of psychological distress27 and have a negative effect on abstinence. Second, all patients should be screened for additional substance abuse. Initial laboratory testing should include a urine drug screen. Third, before discharging the patient, providers should establish primary care follow‐up for continued surveillance of medical issues. There is evidence that primary care services are predictive of better substance abuse treatment outcomes for those with medical problems.28,29
Finally, inpatient psychiatric consultation, upon admission, is essential for several reasons. First, the psychiatric team can help with initial management and treatment of the alcohol withdrawal regardless of stage and severity, obtain a more comprehensive psychiatric history, and assess for the presence of psychiatric comorbidities that may contribute to, aggravate, or complicate the clinical picture. The team can also address other substance abuse issues when detected by drug screen or clinical history. The psychiatric team, along with chemical‐dependency counselors and social workers, can provide valuable input regarding chemical‐dependency resources available on discharge and help instruct the patient in healthy behaviors. Because healthcare illiteracy may be an issue in this patient population, these instructions should be tailored to the patient's educational level. Prior to discharge, the psychiatry team, social workers, or chemical‐dependency counselors can also assist with, or arrange, rehabilitation aftercare for patients. Recent work shows that patients were less likely to be readmitted to crisis detoxification if they entered rehabilitation care within 3 days of discharge.18
Our study has significant limitations. This study was performed with data at a single academic medical center with an ethnically homogeneous patient population, limiting the external validity of its results. Because this is a retrospective study, data analyses are limited by the quality and accuracy of data in the electronic medical record. Also, our follow‐up period may not have been long enough to detect additional admissions, and we did not screen for patient admissions prior to the study period. By limiting data collection to admissions for AWS to general medical services, we may have missed cases of AWS when admitted for other reasons or to subspecialty services, and we may have missed severe cases requiring admission to an intensive care unit. While we believe we were able to capture most admissions, we may underreport this number since we cannot account for those events that may have occurred at other facilities and locations. Lastly, without a control group, this study is limited in its ability to show an association between any variable and readmission.
In our study, 142 patients accounted for 608 admissions during the 3‐year study period, which speaks to the high recidivism rates for patients with AWS. This disease is associated with high morbidity, high medical costs, and high utilization of healthcare. Our study provides insight regarding identification of patients at high risk of multiple admissions with respect to demographic (lower level of education) and clinical characteristics (worse withdrawal severity, more medical and psychiatric comorbidity, and polysubstance abuse). We believe collaboration between social services, chemical‐dependency counselors, psychiatry, and medicine is necessary to effectively treat this population of patients and assist with the crucial transition to the outpatient setting. Future studies should include key social factors, such as health literacy in the readmission risk assessment, as well as primary care follow‐up and rehabilitation aftercare.
Many patients are admitted and readmitted to acute care hospitals with alcohol‐related diagnoses, including alcohol withdrawal syndrome (AWS), and experience significant morbidity and mortality. In patients with septic shock or at risk for acute respiratory distress syndrome (ARDS), chronic alcohol abuse is associated with increased ARDS and severity of multiple organ dysfunction.1 Among intensive care unit (ICU) patients, those with alcohol dependence have higher morbidity, including septic shock, and higher hospital mortality.2 Patients who experience AWS as a result of alcohol dependence may experience life‐threatening complications, such as seizures and delirium tremens.3,4 In‐hospital mortality from AWS is historically high,5 but with benzodiazepines used in a symptom‐driven manner to treat the complications of alcohol use, hospital mortality rates are more recently reported at 2.4%.6
As inpatient outcomes1,2,7 and hospital mortality810 are negatively affected by alcohol abuse, the post‐hospital course of these patients is also of interest. Specifically, patients are often admitted and readmitted with alcohol‐related diagnoses or AWS to acute care hospitals, but relatively little quantitative data exist on readmission factors in this population.11 Patients readmitted to detoxification units or alcohol and substance abuse units have been studied, and factors associated with readmission include psychiatric disorder,1217 female gender,14,15 and delay in rehabilitation aftercare.18
These results cannot be generalized to patients with AWS who are admitted and readmitted to acute‐care hospitals. First, patients hospitalized for alcohol withdrawal symptoms are often medically ill with more severe symptoms, and more frequent coexisting medical and psychiatric illnesses, that complicate the withdrawal syndrome. Detoxification units and substance abuse units require patients to be medically stable before admission, because they do not have the ability to provide a high level of supervision and treatment. Second, much of what we know regarding risk factors for readmission to detoxification centers and substance abuse units comes from studies of administrative data of the Veterans Health Administration,12,13 Medicare Provider Analysis and Review file,16 privately owned outpatient substance abuse centers,14 and publicly funded detoxification centers.18 These results may be difficult to generalize to other patient populations. Accordingly, the objective of this study was to identify demographic and clinical factors associated with multiple admissions to a general medicine service for treatment of AWS over a 3‐year period. Characterization of these high‐risk patients and their hospital course may help focus intervention and reduce these revolving door admissions.
METHODS
The Mayo Clinic Institutional Review Board deemed the study exempt.
Patient Selection
The study was conducted at an 1157‐bed academic tertiary referral hospital, located in the Midwest, that has approximately 15,000 inpatient admissions to general medicine services annually and serves as the main referral center for the region. Patients included in this study were adults admitted to general medicine services and treated with symptom‐triggered Clinical Institute Withdrawal AssessmentAlcohol Revised (CIWA‐Ar) protocol19 between January 1, 2006 and December 31, 2008. Patients were identified using the Mayo Clinic Quality Improvement Resource Center database, as done in a previous CIWA‐Ar study.20 Patients were excluded if the primary diagnosis was a nonalcohol‐related diagnosis (Figure 1).

Patients were placed in 1 of 2 groups based on number of admissions during the study period, either a single‐admission group or a multiple‐admissions group. While most readmission studies use a 30‐day mark from discharge, we used 3 years to better capture relapse and recidivism in this patient population. The 2 groups were then compared retrospectively. To insure that a single admission was not arbitrarily created by the December 2008 cutoff, we reviewed the single‐admission group for additional admissions through June of 2009. If a patient did have a subsequent admission, then the patient was moved to the multiple‐admissions group.
Clinical Variables
Demographic and clinical data was obtained using the Mayo Data Retrieval System (DRS), the Mayo Clinic Life Sciences System (MCLSS) database, and electronic medical records. Clinical data for the multiple‐admissions group was derived from the first admission of the study period, and subsequently referred to as index admission. Specific demographic information collected included age, race, gender, marital status, employment status, and education. Clinical data collected included admitting diagnosis, comorbid medical disorders, psychiatric disorders, and CIWA‐Ar evaluations including highest total score (CIWA‐Ar score [max] and component scores). The CIWA‐Ar protocol is a scale to assess the severity of alcohol withdrawal, based on 10 symptoms of alcohol withdrawal ranging from 0 (not present) to 7 (extremely severe). The protocol requires the scale to be administered hourly, and total scores guide the medication dosing and administration of benzodiazepines to control withdrawal symptoms. Laboratory data collected included serum ammonia, alanine aminotransferase(ALT), and admission urine drug screen. For the purposes of this study, a urine drug screen was considered positive if a substance other than alcohol was present. Length of stay (LOS) and adverse events during hospitalization (delirium tremens, intubations, rapid response team [RRT] calls, ICU transfers, and in‐hospital mortality) were also collected.
Medical comorbidity was measured using the Charlson Comorbidity Index (CCI).21 The CCI was scored electronically using diagnoses in the institution's medical index database dating back 5 years from patient's first, or index, admission. Originally validated as a prognostic tool for mortality 1 year after admission in medical patients, the CCI was chosen as it accounts for most medical comorbidities.21 Data was validated, by another investigator not involved in the initial abstracting process, by randomly verifying 5% of the abstracted data.
Statistical Analysis
Standard descriptive statistics were used for patient characteristics and demographics. Comparing the multiple‐admissions group and single‐admission group, categorical variables were evaluated using the Fisher exact test or Pearson chi‐square test. Continuous variables were evaluated using 2‐sample t test. Multivariate logistic model analyses with stepwise elimination method were used to identify risk factors that were associated with multiple admissions. Age, gender, and variables that were statistically significant in the univariate analysis were used in stepwise regression to get to the final model. A P value of 0.05 was considered statistically significant. All statistical analyses were performed using SAS version 9.3 software (SAS Institute, Cary, NC).
RESULTS
The CIWA‐Ar protocol was ordered on 1199 admissions during the study period. Of these, 411 (34.3%) admissions were excluded because AWS was not the primary diagnosis, leaving 788 (65.7%) admissions for 322 patients, which formed the study population. Of the 322 patients, 180 (56%) had a single admission and 142 (44%) had multiple admissions.
Univariate analyses of demographic and clinical variables are shown in Tables 1 and 2, respectively. Patients with multiple admissions were more likely divorced (P = 0.028), have a high school education or less (P = 0.002), have a higher CCI score (P < 0.0001), a higher CIWA‐Ar score (max) (P < 0.0001), a higher ALT level (P = 0.050), more psychiatric comorbidity (P < 0.026), and a positive urine drug screen (P < 0.001). Adverse events were not significantly different between the 2 groups (Table 2).
| Variable | Single Admission N = 180 | Multiple Admissions N = 142 | P Value |
|---|---|---|---|
| |||
| Age, years (SD) | 47.85 (12.84) | 45.94 (12) | 0.170 |
| Male, No. (%) | 122 (68) | 109 (77) | 0.080 |
| Race/Ethnicity, No. (%) | 0.270 | ||
| White | 168 (93) | 132 (93) | |
| African American | 6 (3) | 3 (2) | |
| Asian | 0 (0) | 1 (1) | |
| Middle Eastern | 3 (2) | 0 (0) | |
| Other | 3 (2) | 6 (4) | |
| Relationship status, No. (%) | 0.160 | ||
| Divorced | 49 (27) | 55 (39) | 0.028* |
| Married | 54 (30) | 34 (24) | 0.230 |
| Separated | 9 (5) | 4 (3) | 0.323 |
| Single | 59 (33) | 38 (27) | 0.243 |
| Widowed | 5 (3) | 3 (2) | 0.703 |
| Committed | 4 (2) | 7 (5) | 0.188 |
| Unknown | 0 (0) | 1 (1) | 0.259 |
| Education, No. (%) | 0.002* | ||
| High school graduate, GED, or less | 49 (28) | 67 (47) | |
| Some college or above | 89 (49) | 60 (42) | |
| Unknown | 41 (23) | 15 (11) | |
| Employment, No. (%) | 0.290 | ||
| Retired | 26 (14) | 12 (8) | |
| Employed | 72 (40) | 51 (36) | |
| Unemployed | 51 (28) | 51 (36) | |
| Homemaker | 9 (5) | 4 (3) | |
| Work disabled | 20 (11) | 23 (16) | |
| Student | 1 (1) | 0 (0) | |
| Unknown | 1 (1) | 1 (1) | |
| Variable | Single Admission N = 180 | Multiple Admissions N = 142 | P Value |
|---|---|---|---|
| |||
| LOS, mean (SD) | 3.71 (7.10) | 2.72 (3.40) | 0.130 |
| Charlson Comorbidity Index, mean (SD) | 1.7 (2.23) | 2.51 (2.90) | 0.005* |
| Medical comorbidity, No. (%) | |||
| Diabetes mellitus | 6 (3) | 16 (11) | 0.005* |
| Cardiovascular disease | 6 (3) | 15 (11) | 0.050* |
| Cerebrovascular disease | 0 (0) | 3 (2) | 0.009* |
| Hypertension | 53 (30) | 36 (25) | 0.400 |
| Cancer | 17 (7) | 10 (9) | 0.440 |
| Psychiatric comorbidity, No. (%) | 97 (54) | 94 (66) | 0.026* |
| Adjustment disorder | 0 (0) | 6 (4) | 0.005* |
| Depressive disorder | 85 (47) | 76 (54) | 0.260 |
| Bipolar disorder | 6 (3) | 10 (7) | 0.130 |
| Psychotic disorder | 4 (2) | 6 (4) | 0.030* |
| Anxiety disorder | 30 (17) | 25 (18) | 0.820 |
| Drug abuse | 4 (2) | 4 (3) | 0.730 |
| Eating disorder | 0 (0) | 3 (2) | 0.050* |
| CIWA‐Ar scores | |||
| CIWA‐Ar score (max), mean (SD) | 15 (8) | 20 (9) | <0.000* |
| Component, mean (SD) | |||
| Agitation | 20 (11) | 36 (25) | 0.001* |
| Anxiety | 23 (13) | 38 (27) | 0.001* |
| Auditory disturbance | 4 (2) | 9 (6) | 0.110 |
| Headache | 11 (6) | 26 (18) | 0.001* |
| Nausea/vomiting | 5 (3) | 17 (12) | 0.003* |
| Orientation | 52 (29) | 72 (51) | 0.001* |
| Paroxysm/sweats | 9 (5) | 17 (12) | 0.023* |
| Tactile disturbance | 25 (14) | 54 (38) | 0.001* |
| Tremor | 35 (19) | 47 (33) | 0.004* |
| Visual disturbance | 54 (30) | 77 (54) | 0.001* |
| ALT (U/L), mean (SD) | 76 (85) | 101 (71) | 0.050* |
| Ammonia (mcg N/dl), mean (SD) | 25 (14) | 29 (29) | 0.530 |
| Positive urine drug screen, No. (%) | 25 (14) | 49 (35) | <0.001* |
| Tetrahydrocannabinol | 14 (56) | 19 (39) | |
| Cocaine | 8 (32) | 8 (16) | |
| Benzodiazepine | 6 (24) | 11 (22) | |
| Opiate | 4 (16) | 13 (26) | |
| Amphetamine | 2 (8) | 2 (4) | |
| Barbiturate | 1 (4) | 0 (0) | |
| Adverse event, No. (%) | |||
| RRT | 1 (1) | 1 (1) | 0.866 |
| ICU transfer | 32 (18) | 20 (14) | 0.550 |
| Intubation | 12 (7) | 4 (3) | 0.890 |
| Delirium tremens | 7 (4) | 4 (3) | 0.600 |
| In‐hospital mortality | 0 (0) | 0 (0) | |
Multivariate logistic model analysis was performed using the variables age, male gender, divorced marital status, high school education or less, CIWA‐Ar score (max), CCI score, psychiatric comorbidity, and positive urine drug screen. With a stepwise elimination process, the final model showed that multiple admissions were associated with high school education or less (P = 0.0071), higher CCI score (P = 0.0010), higher CIWA‐Ar score (max) (P < 0.0001), a positive urine drug screen (P = 0.0002), and psychiatric comorbidity (P = 0.0303) (Table 3).
| Variable | Adjusted Odds Ratio (95% CI) | P Value |
|---|---|---|
| ||
| High school education or less | 2.074 (1.219, 3.529) | 0.0071* |
| CIWA‐Ar score (max) | 1.074 (1.042, 1.107) | <0.0001* |
| Charlson Comorbidity Index | 1.232 (1.088, 1.396) | 0.0010* |
| Psychiatric comorbidity | 1.757 (1.055, 2.928) | 0.0303* |
| Positive urine drug screen | 3.180 (1.740, 5.812) | 0.0002* |
DISCUSSION
We provide important information regarding identification of individuals at high risk for multiple admissions to general medicine services for treatment of AWS. This study found that patients with multiple admissions for AWS had more medical comorbidity. They had more cases of diabetes mellitus, cardiovascular disease, and cerebrovascular disease, and their CCI scores were higher. They also had higher CIWA‐Ar (max) scores, as well as higher CIWA‐Ar component scores, indicating a more severe withdrawal.
Further, psychiatric comorbidity was also associated with multiple admissions. Consistent with the high prevalence in alcoholic patients, psychiatric comorbidity was common in both patients with a single admission and multiple admissions. We also found that a positive urine drug screen was associated with multiple admissions. Interestingly, few patients in each group had a diagnosis recorded in the medical record of an additional substance abuse disorder, yet 14% of patients with a single admission and 29% of patients with multiple admissions had a positive urine drug screen for a non‐alcohol substance. Psychiatric comorbidity, including additional substance abuse, is a well‐established risk factor for readmission to detoxification centers.1215, 17,22,23 Also, people with either an alcohol or non‐alcohol drug addiction, are known to be 7 times more likely to have another addiction than the rest of the population.24 This study suggests clinicians may underrecognize additional substance abuse disorders which are common in this patient population.
In contrast to studies of patients readmitted to detoxification units and substance abuse units,14,15,18,22 we found level of education, specifically a high school education or less, to be associated with multiple admissions. In a study of alcoholics, Greenfield and colleagues found that lower education in alcoholics predicts shorter time to relapse.25 A lack of education may result in inadequate healthcare literacy. Poor health behavior choices that follow may lead to relapse and subsequent admissions for AWS. With respect to other demographic variables, patients in our study population were predominantly men, which is not surprising. Gender differences in alcoholism are well established, with alcohol abuse and dependence more prevalent in men.26 We did not find gender associated with multiple admissions.
Our findings have management and treatment implications. First, providers who care for patients with AWS should not simply focus on treating withdrawal signs and symptoms, but also screen for and address other medical issues, which may not be apparent or seem stable at first glance. While a comorbid medical condition may not be the primary reason for hospital admission, comorbid medical conditions are known to be a source of psychological distress27 and have a negative effect on abstinence. Second, all patients should be screened for additional substance abuse. Initial laboratory testing should include a urine drug screen. Third, before discharging the patient, providers should establish primary care follow‐up for continued surveillance of medical issues. There is evidence that primary care services are predictive of better substance abuse treatment outcomes for those with medical problems.28,29
Finally, inpatient psychiatric consultation, upon admission, is essential for several reasons. First, the psychiatric team can help with initial management and treatment of the alcohol withdrawal regardless of stage and severity, obtain a more comprehensive psychiatric history, and assess for the presence of psychiatric comorbidities that may contribute to, aggravate, or complicate the clinical picture. The team can also address other substance abuse issues when detected by drug screen or clinical history. The psychiatric team, along with chemical‐dependency counselors and social workers, can provide valuable input regarding chemical‐dependency resources available on discharge and help instruct the patient in healthy behaviors. Because healthcare illiteracy may be an issue in this patient population, these instructions should be tailored to the patient's educational level. Prior to discharge, the psychiatry team, social workers, or chemical‐dependency counselors can also assist with, or arrange, rehabilitation aftercare for patients. Recent work shows that patients were less likely to be readmitted to crisis detoxification if they entered rehabilitation care within 3 days of discharge.18
Our study has significant limitations. This study was performed with data at a single academic medical center with an ethnically homogeneous patient population, limiting the external validity of its results. Because this is a retrospective study, data analyses are limited by the quality and accuracy of data in the electronic medical record. Also, our follow‐up period may not have been long enough to detect additional admissions, and we did not screen for patient admissions prior to the study period. By limiting data collection to admissions for AWS to general medical services, we may have missed cases of AWS when admitted for other reasons or to subspecialty services, and we may have missed severe cases requiring admission to an intensive care unit. While we believe we were able to capture most admissions, we may underreport this number since we cannot account for those events that may have occurred at other facilities and locations. Lastly, without a control group, this study is limited in its ability to show an association between any variable and readmission.
In our study, 142 patients accounted for 608 admissions during the 3‐year study period, which speaks to the high recidivism rates for patients with AWS. This disease is associated with high morbidity, high medical costs, and high utilization of healthcare. Our study provides insight regarding identification of patients at high risk of multiple admissions with respect to demographic (lower level of education) and clinical characteristics (worse withdrawal severity, more medical and psychiatric comorbidity, and polysubstance abuse). We believe collaboration between social services, chemical‐dependency counselors, psychiatry, and medicine is necessary to effectively treat this population of patients and assist with the crucial transition to the outpatient setting. Future studies should include key social factors, such as health literacy in the readmission risk assessment, as well as primary care follow‐up and rehabilitation aftercare.
- , . Chronic alcohol abuse, acute respiratory distress syndrome, and multiple organ dysfunction. Crit Care Med. 2003;31(4 suppl):S207–S212.
- , , et al. Alcohol dependence is independently associated with sepsis, septic shock, and hospital mortality among adult intensive care unit patients. Crit Care Med. 2007;35(2):345–350.
- , , . Use of an objective clinical scale in the assessment and management of alcohol withdrawal in a large general hospital. Alcohol Clin Exp Res. 1988;12(3):360–364.
- , . Alcohol withdrawal syndromes in the intensive care unit. Crit Care Med. 2010;38(9 suppl):S494–S501.
- , Alcoholism at the Boston City Hospital—V. The causes of death among alcoholic patients at the Haymarket Square Relief Station, 1923–year="1938"1938. N Engl J Med. year="1939"1939;221(July 13):58–59.
- , , . Epidemiology of alcohol withdrawal syndrome: mortality and factors of poor prognosis [in Spanish]. An Med Interna (Madrid). 2006;23(7):307–309.
- , , . The impact of alcohol‐related diagnoses on pneumonia outcomes. Arch Intern Med. 1997;157(13):1446–1452.
- , , , , , . Analysis of the factors determining survival of alcoholic withdrawal syndrome patients in a general hospital. Alcohol Alcohol. 2010;45(2):151–158.
- , , , , . Predictors of mortality in patients with delirium tremens. Acad Emerg Med. 2008;15(8):788–790.
- , , , . Long‐term mortality of patients admitted to the hospital with alcohol withdrawal syndrome. Alcohol Clin Exp Res. 2011;35(6):1180–1186.
- , , , , , . Substance use treatment barriers for patients with frequent hospital admissions. J Subst Abuse Treat. 2010;38(1):22–30.
- , , . Diagnostic subgroups and predictors of one‐year re‐admission among late‐middle‐aged and older substance abuse patients. J Stud Alcohol. 1994;55(2):173–183.
- , , . Rates and predictors of four‐year readmission among late‐middle‐aged and older substance abuse patients. J Stud Alcohol. 1994;55(5):561–570.
- , , . Readmission among chemical dependency patients in private, outpatient treatment: patterns, correlates and role in long‐term outcome. J Stud Alcohol. 2005;66(6):842–847.
- , , , . Predicting readmission to substance abuse treatment using state information systems. The impact of client and treatment characteristics. J Subst Abuse. 2000;12(3):255–270.
- , , , . Elderly Medicare inpatients with substance use disorders: characteristics and predictors of hospital readmissions over a four‐year interval. J Stud Alcohol. 2000;61(6):891–895.
- , . The role of psychiatric comorbidity in the prediction of readmission for detoxification. Compr Psychiatry. 1998;39(3):129–136.
- , , , , , . Factors associated with frequent utilization of crisis substance use detoxification services. J Addict Dis. 2011;30(2):116–122.
- , , , , . Assessment of alcohol withdrawal: the revised Clinical Institute Withdrawal Assessment for Alcohol scale (CIWA‐Ar). Br J Addict. 1989;84(11):1353–1357.
- , , , Inappropriate use of symptom‐triggered therapy for alcohol withdrawal in the general hospital. Mayo Clin Proc. 2008;83(3):274–279.
- , , , . A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , . A four‐year follow‐up study of male alcoholics: factors affecting the risk of readmission. Alcohol. 2002;27(2):83–88.
- , , , et al. Diagnosis and hospital readmission rates of female veterans with substance‐related disorders. Psychiatr Serv. 1995;46(9):932–937.
- , , , et al. Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiologic Catchment Area (ECA) Study. JAMA. 1990;264(19):2511–2518.
- , , , , , . The relationship between educational attainment and relapse among alcohol‐dependent men and women: a prospective study. Alcohol Clin Exp Res. 2003;27(8):1278–1285.
- , , , . Prevalence, correlates, disability, and comorbidity of DSM‐IV alcohol abuse and dependence in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry. 2007;64(7):830–842.
- , . Health‐related quality of life among adults with serious psychological distress and chronic medical conditions. Qual Life Res. 2008;17(4):521–528.
- , , , , . Primary medical care and reductions in addiction severity: a prospective cohort study. Addiction. 2005;100(1):70–78.
- , , , . The role of medical conditions and primary care services in 5‐year substance use outcomes among chemical dependency treatment patients. Drug Alcohol Depend. 2008;98(1–2):45–53.
- , . Chronic alcohol abuse, acute respiratory distress syndrome, and multiple organ dysfunction. Crit Care Med. 2003;31(4 suppl):S207–S212.
- , , et al. Alcohol dependence is independently associated with sepsis, septic shock, and hospital mortality among adult intensive care unit patients. Crit Care Med. 2007;35(2):345–350.
- , , . Use of an objective clinical scale in the assessment and management of alcohol withdrawal in a large general hospital. Alcohol Clin Exp Res. 1988;12(3):360–364.
- , . Alcohol withdrawal syndromes in the intensive care unit. Crit Care Med. 2010;38(9 suppl):S494–S501.
- , Alcoholism at the Boston City Hospital—V. The causes of death among alcoholic patients at the Haymarket Square Relief Station, 1923–year="1938"1938. N Engl J Med. year="1939"1939;221(July 13):58–59.
- , , . Epidemiology of alcohol withdrawal syndrome: mortality and factors of poor prognosis [in Spanish]. An Med Interna (Madrid). 2006;23(7):307–309.
- , , . The impact of alcohol‐related diagnoses on pneumonia outcomes. Arch Intern Med. 1997;157(13):1446–1452.
- , , , , , . Analysis of the factors determining survival of alcoholic withdrawal syndrome patients in a general hospital. Alcohol Alcohol. 2010;45(2):151–158.
- , , , , . Predictors of mortality in patients with delirium tremens. Acad Emerg Med. 2008;15(8):788–790.
- , , , . Long‐term mortality of patients admitted to the hospital with alcohol withdrawal syndrome. Alcohol Clin Exp Res. 2011;35(6):1180–1186.
- , , , , , . Substance use treatment barriers for patients with frequent hospital admissions. J Subst Abuse Treat. 2010;38(1):22–30.
- , , . Diagnostic subgroups and predictors of one‐year re‐admission among late‐middle‐aged and older substance abuse patients. J Stud Alcohol. 1994;55(2):173–183.
- , , . Rates and predictors of four‐year readmission among late‐middle‐aged and older substance abuse patients. J Stud Alcohol. 1994;55(5):561–570.
- , , . Readmission among chemical dependency patients in private, outpatient treatment: patterns, correlates and role in long‐term outcome. J Stud Alcohol. 2005;66(6):842–847.
- , , , . Predicting readmission to substance abuse treatment using state information systems. The impact of client and treatment characteristics. J Subst Abuse. 2000;12(3):255–270.
- , , , . Elderly Medicare inpatients with substance use disorders: characteristics and predictors of hospital readmissions over a four‐year interval. J Stud Alcohol. 2000;61(6):891–895.
- , . The role of psychiatric comorbidity in the prediction of readmission for detoxification. Compr Psychiatry. 1998;39(3):129–136.
- , , , , , . Factors associated with frequent utilization of crisis substance use detoxification services. J Addict Dis. 2011;30(2):116–122.
- , , , , . Assessment of alcohol withdrawal: the revised Clinical Institute Withdrawal Assessment for Alcohol scale (CIWA‐Ar). Br J Addict. 1989;84(11):1353–1357.
- , , , Inappropriate use of symptom‐triggered therapy for alcohol withdrawal in the general hospital. Mayo Clin Proc. 2008;83(3):274–279.
- , , , . A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383.
- , , . A four‐year follow‐up study of male alcoholics: factors affecting the risk of readmission. Alcohol. 2002;27(2):83–88.
- , , , et al. Diagnosis and hospital readmission rates of female veterans with substance‐related disorders. Psychiatr Serv. 1995;46(9):932–937.
- , , , et al. Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiologic Catchment Area (ECA) Study. JAMA. 1990;264(19):2511–2518.
- , , , , , . The relationship between educational attainment and relapse among alcohol‐dependent men and women: a prospective study. Alcohol Clin Exp Res. 2003;27(8):1278–1285.
- , , , . Prevalence, correlates, disability, and comorbidity of DSM‐IV alcohol abuse and dependence in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry. 2007;64(7):830–842.
- , . Health‐related quality of life among adults with serious psychological distress and chronic medical conditions. Qual Life Res. 2008;17(4):521–528.
- , , , , . Primary medical care and reductions in addiction severity: a prospective cohort study. Addiction. 2005;100(1):70–78.
- , , , . The role of medical conditions and primary care services in 5‐year substance use outcomes among chemical dependency treatment patients. Drug Alcohol Depend. 2008;98(1–2):45–53.
Copyright © 2012 Society of Hospital Medicine
Evolving Practice of Hospital Medicine
Hospitalists are physicians whose primary focus is the general medical care of hospitalized patients. Hospitalists are uniquely positioned to implement strategies to improve patient flow and efficiency.1 With emergency department (ED) diversion reaching rates upward of 70%, lack of access to inpatient beds leads to delayed care with worsened outcomes.25
To improve access to hospital beds, hospitals may increase capacity by either adding beds or by more efficiently using existing beds. Operations management principles have been applied to healthcare to ensure efficient use of beds. These include: reducing variability of scheduled admissions, remeasuring length of stay (LOS) and bed demand after implementing strategies to reduce practice variation, and employing queuing theory to generate predictions of optimal beds needed.6 The Joint Commission implemented a leadership standard (LD 04.03.11) that hospitals develop and implement plans to identify and mitigate impediments to efficient patient flow through the hospital.
To improve access, hospital leaders expect hospitalists to staff in inpatient medicine programs, surgical comanagement, short stay and chest pain units, and active bed management.7 In the following review, we define hospitalists' roles in the aforementioned programs and their effect on patient flow. We also touch on preoperative clinics, palliative care, geographic rounding, and flexible staffing models.
ACUTE INPATIENT CARE
Hospitalists are one of the fastest growing physician groups in the United States.810 Hospitalists improve efficiency and quality of care across a variety of demographic, geographic, and healthcare settings.11, 12 A 2002 retrospective cohort study in a community‐based urban teaching hospital showed that hospitalists decreased LOS by 0.61 days and lowered risk for death in the hospital (adjusted relative hazard, 0.71; 95% confidence interval [CI], 0.540.93).13 A 2004 prospective quasi‐experimental observational study done at an academic teaching hospital showed an adjusted LOS that was 16.2% lower, and adjusted cost 9.7% lower, for patients on the hospitalists' service.14 In 2007, Lindenauer and colleagues found that a national sample of hospitalists decreased LOS by 0.4 days and lowered cost by $286 per patient.15 The findings of these individual studies were supported in a 2009 systematic review of 33 studies by Peterson which showed that hospitalists decrease LOS.16 In a recent study, Kuo and Goodwin showed that while hospitalists decrease LOS and cost, the patients they care for have higher Medicare costs after discharge by $322 per patient, and are more likely to be readmitted (odds ratio, 1.08; CI, 1.041.14).17
The hospitalist model of care continues to grow, and hospitalists will soon number as many as 30,000.18 For acute medical inpatients, the evidence suggests that hospitalists improve patient flow by decreasing LOS while improving other aspects of quality of care. However, Kuo and Goodwin's findings suggest that the transition of care from inpatient to outpatient settings still requires attention.17
SURGICAL COMANAGEMENT
The Society of Hospital Medicine (SHM) core competencies include perioperative medicine.19, 20 In the 2006 SHM national survey, 85% of hospital medicine groups indicated that they participated in surgical comanagement.21
Hospitalists have improved patient flow and outcomes for orthopedic patients. Hospitalist management of hip fracture patients decreases time to surgery and LOS compared to standard care.2224 Phy and colleagues studied 466 patients for 2 years after the inception of hospital medicine comanagement of surgical patients, and found that care by hospitalists decreased LOS by 2.2 days.22 In a retrospective study of 118 patients, Roy and colleagues found that hospitalist‐managed patients had shorter time to consultation and surgery, decreased LOS, and lower costs.23 In a retrospective cohort study, Batsis looked at mortality in 466 patients with hip fracture, and found no difference between hospitalist management and standard care.24 In patients undergoing elective hip and knee arthroplasty, Huddleston and colleagues reported that patients managed by hospitalists had fewer complications and shorter LOS. The nurses and orthopedic surgeons preferred the hospitalistorthopedist comanagement model.25
The benefits of hospitalist comanagement are not limited to adult patients undergoing orthopedic surgery. For high‐risk patients undergoing lower extremity reconstruction surgery, Pinzur and colleagues noted that LOS was shorter for a cohort of patients managed by hospitalists than for a group of historical controls not treated by hospitalists.26 Simon and colleagues studied comanagement for pediatric spinal fusion patients, and found a decrease in LOS from 6.5 to 4.8 days.27
Several factors should be considered in developing and implementing a successful comanagement program. Since comanagement duties may fall upon hospitalists in order to protect surgeons' time,28 hospital medicine groups should ensure adequate staffing prior to taking on additional services. Clear guidelines to delineate roles and responsibilities of the comanaging groups also need to be developed and implemented.29, 30
Comanaging may also involve additional training. Hospitalists who manage neurologic, neurosurgical, trauma, and psychiatric patients report being undertrained for such conditions.31, 32 Hospital medicine groups need to ensure training needs are met and supported. Given the successes of comanagement and the increasing complexity of surgical patients,33 this practice will likely expand to a greater variety of non‐medical patients.
SHORT STAY UNITS
In 2003, short stay units (SSU) were present in approximately 20% of US hospitals, with 11% of hospitals planning on opening one in the next year.34 SSU are designed to manage acute, self‐limited medical conditions that require brief staysusually less than 72 hours. Approximately 80% of SSU patients are discharged home, avoiding hospitalization.35 Historically, SSU have been under the domain of the ED; however, there is an emerging role for hospitalist‐run SSU.36
Despite demand for SSU, little research has been performed on hospitalist‐led SSU. In 2000, Abenhaim and colleagues showed that a hospitalist‐run SSU at a university‐affiliated teaching hospital had a shorter LOS and lower rates of complications and readmissions when compared to medicine teaching services.37 In 2008, Northwestern Memorial Hospital opened a 30‐bed hospitalist‐run SSU; for those patients, LOS decreased by 2 days.38 In 2010, Leykum and colleagues showed that a hospitalist‐run observation unit can decrease LOS from 2.4 days to 2.2 days.39 Careful selection of SSU patients is needed to obtain these results. Lucas and colleagues found that whether or not SSU patients required assistance of specialists was the strongest predictor of unsuccessful stays (>72 hours or inpatient conversion) in SSU.36
Whether SSU are run by hospital medicine or emergency medicine is decided at an institutional level. Location of SSU in a specifically designated area is crucial, as it allows physicians to round efficiently on patients and to work with staff trained in observation services. Development of admission criteria that include specific diagnoses which match hospitalists' scope of practice is also important (Table 1).32
| Evaluation of Diagnostic Syndromes | Treatment of Emergent Conditions |
|---|---|
| |
| Chest pain | Asthma |
| Abdominal pain | Congestive heart failure |
| Fever | Dehydration |
| Gastrointestinal bleed | Hypoglycemia or hyperglycemia |
| Syncope | Hypercalcemia |
| Dizziness | Atrial fibrillation |
| Headache | |
| Chest trauma | |
| Abdominal trauma | |
The protocol‐based and diagnosis‐specific nature of SSU may enhance quality of care through standardization. Future research may delineate the utility of SSU.
CHEST PAIN UNITS
In the United States, in 2004, approximately 6 million patients present annually to EDs with chest pain.40 Cost of care of patients unnecessarily admitted to coronary care units has been estimated to be nearly $3 billion annually.41 Still, as many as 3% of patients with acute myocardial infarction are discharged home.42 Chest pain units (CPU) were developed to facilitate evaluation of patients with chest pain, at low risk for acute coronary syndrome, without requiring inpatient admission. A number of studies have suggested that admission to a CPU is a safe and cost‐effective alternative to hospital admission.4348
CPU have traditionally been staffed by ED physicians and/or cardiologists. In a prepost study, Krantz and colleagues found that a CPU model, incorporating hospitalists at an academic public safety‐net hospital, decreased ED LOS with no difference in 30‐day cardiac event rate.49 Myers and colleagues created a hospitalist‐directed nonteaching service in an academic medical center to admit low‐risk chest pain patients. Patients admitted to the hospitalist service had a statistically significant lower median LOS (23 hours vs 33 hours) and approximately half the median hospital charges than those admitted to teaching services.50 At the same academic medical center, Bayley and colleagues showed that 91% of patients admitted for chest pain waited more than 3 hours for a bed. This adversely affected ED revenue by tying up beds, resulting in an estimated annual loss of $168,300 of hospital revenue. Creation of a hospitalist‐managed service for low‐acuity chest pain patients reduced hospital LOS by 7 hours.51 Somekh and colleagues demonstrated that a protocol‐driven, cardiologist‐run CPU results in a decreased LOS and readmission rate compared to usual care.52 In a non‐peer reviewed case study, Cox Health opened an 8‐bed, hospitalist‐led CPU in 2003. They decreased LOS from 72 to 18 hours, while increasing revenue by $2.5 million a year.53 These studies suggest that hospitalist‐run CPU can decrease LOS, increase revenue, and relieve ED overcrowding.
Development of a successful CPU depends upon clear inclusion/exclusion criteria; close collaboration among ED physicians, hospitalists, and cardiologists; the development of evidence‐based protocols, and the availability of stress testing.
ACTIVE BED MANAGEMENT
As of 2007, 90% of EDs were crowded beyond their capacity.2 ED crowding leads to ambulance diversion,54 which can delay care and increase mortality rates.55 One of the main causes of ED crowding is the boarding of admitted patients.56 Boarded, admitted patients have been shown to have decreased quality of care and patient satisfaction.35
Active bed management (ABM) by hospitalists can decrease ED diversion. Howell and colleagues instituted ABM where hospitalists, as active bed managers, facilitate placement of patients to their inpatient destinations to assist ED flow.57 This 24‐hour, hospitalist‐led, active bed management service decreased both ED LOS and ambulance diversion. The bed manager collaborated real‐time with medicine and ED attending physicians, nursing supervisors, and charge nurses to change patient care status, and assign and facilitate transfer of patients to appropriate units. These hospitalist bed managers were also empowered to activate additional resources when pre‐diversion rounds identified resource limitations and impending ED divert. They found overall ED LOS for admitted patients decreased by 98 minutes, while LOS for non‐admitted patients stayed the same. AMB decreased diversion due to critically ill and telemetry patients by 28% (786 hours), and diversion due to lower acuity patients by 6% (182 hours). This intervention proved cost‐effective. Three full‐time equivalent (FTE) hospitalists' salaries staff 1 active bed manager working 24/7. Nearly 1000 hours of diversion were avoided at an annual savings of $1086 per hour of diversion decreased.
ABM is a new frontier for hospitals in general, and hospitalists in particular. Chadaga and colleagues found that a hospital medicine‐ED team participating in active bed management, while caring for admitted patients boarded in the ED, can decrease ED diversion and improve patient flow. The percentage of patients transferred to a medicine floor and discharged within 8 hours was reduced by 67% (P 0.01), while the number of discharges from the ED of admitted medicine patients increased by 61% (P 0.001).58
To decrease initial investment, components of ABM (ED triage, bed assignment, discharge facilitation) can be instituted in parts. Hospital medicine groups with limited resources may only provide a triage service by phone for difficult ED cases. Bedside evaluations and collaboration with nursing staff to improve bed placement may be a next step, with floor and/or intensive care unit (ICU) rounds to facilitate early discharges as a final component.
OTHER AREAS
Preoperative Clinics
In 2005, SHM cited preoperative clinics as an important aspect of preoperative care.59 Sehgal and Wachter included preoperative clinics as an area for expanding the role of hospitalists in the United States.60 These clinics can decrease delays to surgery, LOS, and cancellations on the day of surgery.61 The Cleveland Clinic established the Internal Medicine Preoperative Assessment, Consultation, and Treatment (IMPACT) Center in 1997, and has decreased surgery delay rate by 49%.59 At Kaiser Bellflower Medical Center, a preoperative medicine service that provides preoperative screening decreased the number of surgical procedures cancelled on the day of surgery by more than half.62 Gates Hospitalists LLC's perioperative care decreased delay to surgery and lost operating room time.63 In order for a preoperative service to be successful, there must be buy‐in from hospitalists, surgeons, and primary care physicians, as well as adequate staffing and clinical support.59
Palliative Care
Palliative care has been identified by SHM as a core competency in hospital medicine.64 There are several key components in delivery of quality palliative care, including communication about prognosis, pain and symptom control, and hospice eligibility.65 Hospitalists are in a unique position to offer and improve palliative care for hospitalized patients. The majority of hospitalists report spending significant amounts of time caring for dying patients; thereby, hospitalists frequently provide end‐of‐life care.66, 67 Compared to community‐based physicians, patients cared for by hospitalists have higher odds of having documented family discussions regarding end‐of‐life care, and have fewer or no key symptoms (pain, anxiety, or dyspnea).66 In addition, hospitalists' availability improves response time when a patient's clinical status changes or deteriorates, leading to prompter delivery of symptom alleviation.65 Hospitalists are becoming more experienced with end‐of‐life care, as they are exposed to terminally ill patients on a daily basis. More experience leads to improved recognition of patients with limited prognosis, which leads to earlier discussions about goals of care and faster delivery of palliative care. Perhaps this could decrease LOS and be a future area of study.
Geographic Rounding
In the last 5 years, hospital administrators have promoted geographic rounding, where hospitalists see all their patients in 1 geographic location.69 The driving forces behind this include poor patient satisfaction with physician availability, large amounts of time spent by hospitalists in transit to and from patient locations, and frustrations regarding communication with nursing.70 Several groups have instituted this with success. Cleveland Clinic and Virtua Memorial Hospital have found improved patient satisfaction and decreased LOS.69, 70 O'Leary and colleagues found improved awareness of care plans by the entire team.71 Caution should be taken to assure proper physician‐to‐patient ratios, avoid physician isolation, and coordinate physician shifts with bed assignments.69 To address some of these issues, groups have used a hybrid model where a hospitalist is primarily located on one unit but can flex or overflow onto another unit.70 Steps to success with geographic rounding include buy‐in from the institution and nursing, assuring a safe physician‐to‐patient ratio, avoiding wasted beds, and facilitating multidisciplinary rounds.69
Flexible Staffing Models
In SHM's 2010 State of Hospital Medicine Report, 70% of hospitalist groups used a fixed shift‐based staffing model (ie, 7 days on/7 days off).72 Flexible staffing models in which physician coverage is adjusted to patient volume are growing in popularity. This model can be tailored for each institution by examining admission and patient volume trends to increase coverage during busy periods and decrease coverage during slower periods. Potential benefits include alleviating burn out, reducing LOS, and improving patient outcomes. Nursing data suggests that a higher patient‐to‐nursing ratio is associated with increased 30‐day mortality,73 and an ED study found that increasing physician coverage during the evening shift shortened ED LOS by 20%.74 To date, none of these endpoints have been studied for hospital medicine.
CONCLUSION
While many hospital medicine groups were started to provide acute inpatient medical care, most have found that their value to hospitals reaches beyond bedside care. With an epidemic of ED diversion and lack of access to hospital beds and services, optimizing throughput has become imperative for hospital systems. While hospital access can be improved with addition of new beds, improving throughput by decreasing LOS maximizes utilization of existing resources.
We have reviewed how hospitalists improve patient flow in acute inpatient medicine, surgical comanagement, short stay units, chest pain units, and active bed management. In each instance, the literature supports measures for decreasing LOS while maintaining or improving quality of care. Hinami and colleagues showed physician satisfaction with hospitalist‐provided patient care.75 Most studies have been limited by tracking upstream effects of improved efficiency. As there is now some evidence that decreasing LOS may increase readmissions,17 future studies should incorporate this metric into their outcomes. The effect of formal operations management principles on patient flow and bed efficiency is not well known and should be further examined.
In addition, we have touched on other areas (perioperative clinics, palliative care, geographic rounding, and flexible staffing models) where hospitalists may impact patient throughput. These areas represent excellent opportunities for future research.
Hospitalist participation in many of these areas is in its infancy. Hospital medicine programs interested in expanding their services, beyond acute inpatient care, have the opportunity to develop standards and continue research on the effect of hospital medicine‐led services on patient care and flow.
Acknowledgements
Disclosure: All authors disclose no relevant or financial conflicts of interest.
- SHM Benchmarks Committee. Maximizing throughput and improving patient flow. The Hospitalist, Supplement: How Hospitalists Add Value. Philadelphia, PA: Society of Hospital Medicine; 2005. Available online at http://www.the‐hospitalist.org/details/article/279433/Maximizing_Throughput_and_Improving_Patient_Flow.html. Accessed on July 2009.
- Institute of Medicine, Committee on the Future of Emergency Care in the United States Health System. Hospital‐Based Emergency Care: At the Breaking Point. Washington, DC: National Academies Press; 2007.
- , . Emergency department crowding is associated with poor care for patients with severe pain. Ann Emerg Med. 2008;51:1–5.
- , , , et al. The impact of emergency department crowding measures on time to antibiotics for patients with community‐acquired pneumonia. Ann Emerg Med. 2007;50:510–516.
- , , , , ; for the DELAYED‐ED Study Group. Impact of delayed transfer of critically ill patients from the emergency department to the intensive care unit. Crit Care Med. 2007;35:1477–1483.
- . Managing Patient Flow in Hospitals: Strategies and Solutions, 2nd ed. In: Beurhaus P, Rudolph M, Prenney B, et al, eds. Joint Commission Resources, Joint Commission Resources, Inc., 2009.
- , , , . California hospital leader's view of hospitalists: meeting needs of the present and the future. J Hosp Med. 2009;4(9):528–534.
- , . The emerging role of “hospitalists” in the American health care system. N Engl J Med. 1996;335:514–517.
- , , , et al. Trends in market demand for internal medicine 1999–2004: an analysis of physician job advertisements. J Gen Intern Med. 2006;21:1079–1085.
- , , , et al. Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians. Ann Intern Med. 1999;130:343–349.
- , , , et al. Reorganizing an academic medical service impact on cost, quality, patient satisfaction, and education. JAMA. 1998;279:1560–1565.
- , . The hospitalist movement 5 years later. JAMA. 2002;287:487–494.
- , , , , , . Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes. Ann Intern Med. 2002;137(11):859–865.
- , , . Associations with reduced length of stay and costs on an academic hospitalist service. Am J Manag Care. 2004;10(8):561–568.
- , , , et al. Outcomes of care by hospitalists, general internists and family physicians. N Engl J Med. 2007;357(25):2589–2600.
- . Systematic review of outcomes and quality measures in adult patients cared for by hospitalists vs nonhospitalists. Mayo Clin Proc. 2009;84(3):248–254.
- , . Association of hospitalist care with medical utilization after discharge: evidence of cost shift from a cohort study. Ann Intern Med. 2011;155:152–159.
- , , , et al. The potential size of the hospitalist workforce in the United States. Am Med. 1999;106(4):441–445.
- , . Surgical co‐management: a natural evolution of hospitalist practice. J Hosp Med. 2008;3:394–397.
- , , , et al. The core competencies in hospital medicine: a framework for curriculum development. J Hosp Med. 2006;1(suppl 1):1–30.
- Society of Hospital Medicine. Co‐Management Task Force Page. Available at: http://www.hospitalmedicine.org/AM/Template.cfm? Section=Home165(7):796–801.
- , , , et al. Associations between the hospitalist model of care and quality‐of‐care‐related outcomes in patients undergoing hip fracture surgery. Mayo Clin Proc. 2006;81(1):28–31.
- . Effects of a hospitalist care model on mortality of elderly patients with hip fractures. J Hosp Med. 2007;2(4):219–225.
- , , , et al; for the Hospital‐Orthopedic Team Trial Investigators. Medical and surgical co‐management after elective hip and knee arthroplasty: a randomized, controlled trial. Ann Intern Med. 2004;141(1):28–38.
- , , , et al. Hospitalist‐orthopedic co‐management of high‐risk patients undergoing lower extremity reconstruction surgery. Orthopedics. 2009;32(7):495.
- , , , et al. Pediatric hospitalist co‐management of spinal fusion surgery patients. J Hosp Med. 2007;2:23–29.
- . Just because you can, doesn't mean that you should: a call for the rational application of hospitalist co‐management. J Hosp Med. 2008;3:398–402.
- Society of Hospital Medicine. SHM White Paper: Co‐Management White Paper. Philadelphia, PA: 2010.
- American Medical Association, Council on Ethical and Judicial Affairs. CEJA Report 5–I‐99. Ethical Implications of Surgical Co‐Management. Available at: http://www.ama‐assn.org/resources/doc/code‐medical‐ethics/8043a.pdf. Accessed November 17, 2011.
- , , , et al. Hospitalist care and length of stay in patients requiring complex discharge planning and close clinical monitoring. Arch Intern Med. 2007;167:1869–1874.
- , , , et al. Hospitalist's perceptions of their residency training needs: results of a national survey. Am J Med. 2001;111:247–254.
- , . Why perioperative medicine matters more than ever. Cleve Clin J Med. 2006;73(supp 1):S1.
- , , , et al. A national survey of observation units in the United States. Am J Emerg Med. 2003;12:529–533.
- Society of Hospital Medicine. SHM White Paper: Observation Unit White Paper. Philadelphia, PA: 2009.
- , , , et al. A hospitalist‐run short‐stay unit: features that predict length‐of‐stay and eventual admission to traditional inpatient services. J Hosp Med. 2009;4(5):276–284.
- , , , . Program description: a hospitalist‐run medical short‐stay unit in a teaching hospital. Can Med Assoc J. 2000:163(11):1477–1480.
- . Hospitalists can cut ED overcrowding. ACEP News. 2010.
- , , . Implementation of a hospitalist‐run observation unit and impact on length of stay (LOS): a brief report. J Hosp Med. 2010;5(9):E2–E5.
- , . National Hospital Ambulatory Medical Care survey: 2004 emergency department summary. Adv Data. 2006;23:1–29.
- , . Identification of chest pain patients appropriate for an emergency department observation unit. Emerg Med Clin North Am. 2001;19:35–66.
- , , . Stress testing for risk stratification of patients with low to moderate probability of acute cardiac ischemia. Emerg Med Clin North Am. 2001;19:87–103.
- , , , et al. An evaluation of a chest pain diagnostic protocol to exclude acute cardiac ischemia in the emergency department. Arch Intern Med. 1997;157:1085–1091.
- , , . The Chest Pain Evaluation Center at the University of Maryland Medical Center. Md Med J. 1994;43:1047–1052.
- , , , , . Cost effectiveness of mandatory stress testing in chest pain center patients. Ann Emerg Med. 1997;29:88–98.
- , , , et al. A rapid diagnostic and treatment center for patients with chest pain in the emergency department. Ann Emerg Med. 1995;25:1–8.
- , , , , . An emergency department‐based protocol for rapidly ruling out myocardial ischemia reduces hospital time and expense: results of a randomized study (ROMIO). J Am Coll Cardiol. 1996;28:25–33.
- , , , et al. Randomized controlled trial and economic evaluation of a chest pain observation unit compared with routine care. BMJ. 2004;328:254.
- , , , et al. A cooperative care model: cardiologists and hospitalists reduce length of stay in a chest pain observation unit. Crit Pathw Cardiol. 2005;4(2):55–58.
- , , , et al. Improving resource utilization in a teaching hospital: development of a nonteaching service for chest pain admissions. Acad Med. 2006;81(5):432–435.
- , , , et al. The financial burden of emergency department congestion and hospital crowding for chest pain patients awaiting admission. Ann Emerg Med. 2005;45(2):110–117.
- , , , , . Differences in diagnostic evaluation and clinical outcomes in the care of patients with chest pain based on admitting service: the benefits of a dedicated chest pain unit. J Nucl Cardiol. 2008;15(2):186–192.
- . Taking charge of observation units. Today's Hospitalist. July 2007.
- , , . Access block cause emergency department overcrowding and ambulance diversion in Perth, Western Australia. Emerg Med J. 2005;22:351–354.
- , , , . The relationship between distance to hospital and patient mortality in emergencies: an observational study. Emerg Med J. 2007;24:665–668.
- , . Systematic review of emergency department crowding: causes, effects, and solutions. Ann Emerg Med. 2008;52:126–136.
- , , , , , . Active bed management by hospitalists and emergency department throughput. Ann Intern Med. 2008;149:804–810.
- , , , et al. A hospitalist‐led emergency department team improves hospital bed efficiency. J Hosp Med. 2010;5(suppl 1):17–18.
- Society of Hospital Medicine. Perioperative care (a special supplement to The Hospitalist). Philadelphia, PA: Society of Hospital Medicine; 2005. Available at: http://www.hospitalmedicine.org/AM/Template.cfm?Section=Home136:591–596.
- Hospitalist Management Advisor. Hospitalist branch into preoperative medicine with preop assessments. Marblehead, MA: HCPro, 2006. Available at: http://www.hcpro.com/HOM‐57460–3615/Hospitalists‐branch‐into‐perioperative‐medicine‐with‐preop‐assessments.html. Accessed February 15, 2012.
- . The preoperative medicine service: an innovative practice at Kaiser Bellflower Medical Center. The Permanente Journal. 2002;6:13–16.
- . A preop evaluation service delivers unexpected benefits. Today's Hospitalist. January 2008.
- , , , , . The core competencies in hospital medicine: a framework for curriculum development. J Hosp Med. 2006;1:1–67.
- , , , , , . Common myths about caring for patients with terminal illness: opportunities to improve care in the hospital setting. J Hosp Med. 2007;2:357–365.
- . End‐of‐life care in a voluntary hospitalist model: effects on communication, processes of care, and patient symptoms. Am J Med. 2004;116:669–675.
- , , , . Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians. Ann Intern Med. 1999;130:343–349.
- , . Palliative care and hospitalist: an opportunity for cross‐fertilization. Am J Med. 2001;111(suppl):10S–14S.
- . Giving hospitalists their space. ACP Hospitalist. February 2008.
- . Having problems findings your patients? Today's Hospitalists. June 2010.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 24(11):1223–1227.
- Medical Group Management Association and Society of Hospital Medicine (SHM). State of Hospital Medicine 2010 Report Based on 2009. Available online at http://www.mgma.com/store/Surveys‐and‐Benchmarking/State‐of‐Hospital‐Medicine‐2010‐Report‐Based‐on‐2009 ‐Data‐Print‐Edition/.
- , , , et al. Hospital nurse staffing and patient mortality, nurse burnout, and job dissatisfaction. JAMA. 2002;288(16):1987–1993.
- , . Reduced length of stay in medical emergency department patients: a prospective controlled study on emergency physician staffing. Eur J Emerg Med. 2004;11(1):29–34.
- , , , . Provider expectations and experiences of comanagement. J Hosp Med. 2011;6(7):401–404.
Hospitalists are physicians whose primary focus is the general medical care of hospitalized patients. Hospitalists are uniquely positioned to implement strategies to improve patient flow and efficiency.1 With emergency department (ED) diversion reaching rates upward of 70%, lack of access to inpatient beds leads to delayed care with worsened outcomes.25
To improve access to hospital beds, hospitals may increase capacity by either adding beds or by more efficiently using existing beds. Operations management principles have been applied to healthcare to ensure efficient use of beds. These include: reducing variability of scheduled admissions, remeasuring length of stay (LOS) and bed demand after implementing strategies to reduce practice variation, and employing queuing theory to generate predictions of optimal beds needed.6 The Joint Commission implemented a leadership standard (LD 04.03.11) that hospitals develop and implement plans to identify and mitigate impediments to efficient patient flow through the hospital.
To improve access, hospital leaders expect hospitalists to staff in inpatient medicine programs, surgical comanagement, short stay and chest pain units, and active bed management.7 In the following review, we define hospitalists' roles in the aforementioned programs and their effect on patient flow. We also touch on preoperative clinics, palliative care, geographic rounding, and flexible staffing models.
ACUTE INPATIENT CARE
Hospitalists are one of the fastest growing physician groups in the United States.810 Hospitalists improve efficiency and quality of care across a variety of demographic, geographic, and healthcare settings.11, 12 A 2002 retrospective cohort study in a community‐based urban teaching hospital showed that hospitalists decreased LOS by 0.61 days and lowered risk for death in the hospital (adjusted relative hazard, 0.71; 95% confidence interval [CI], 0.540.93).13 A 2004 prospective quasi‐experimental observational study done at an academic teaching hospital showed an adjusted LOS that was 16.2% lower, and adjusted cost 9.7% lower, for patients on the hospitalists' service.14 In 2007, Lindenauer and colleagues found that a national sample of hospitalists decreased LOS by 0.4 days and lowered cost by $286 per patient.15 The findings of these individual studies were supported in a 2009 systematic review of 33 studies by Peterson which showed that hospitalists decrease LOS.16 In a recent study, Kuo and Goodwin showed that while hospitalists decrease LOS and cost, the patients they care for have higher Medicare costs after discharge by $322 per patient, and are more likely to be readmitted (odds ratio, 1.08; CI, 1.041.14).17
The hospitalist model of care continues to grow, and hospitalists will soon number as many as 30,000.18 For acute medical inpatients, the evidence suggests that hospitalists improve patient flow by decreasing LOS while improving other aspects of quality of care. However, Kuo and Goodwin's findings suggest that the transition of care from inpatient to outpatient settings still requires attention.17
SURGICAL COMANAGEMENT
The Society of Hospital Medicine (SHM) core competencies include perioperative medicine.19, 20 In the 2006 SHM national survey, 85% of hospital medicine groups indicated that they participated in surgical comanagement.21
Hospitalists have improved patient flow and outcomes for orthopedic patients. Hospitalist management of hip fracture patients decreases time to surgery and LOS compared to standard care.2224 Phy and colleagues studied 466 patients for 2 years after the inception of hospital medicine comanagement of surgical patients, and found that care by hospitalists decreased LOS by 2.2 days.22 In a retrospective study of 118 patients, Roy and colleagues found that hospitalist‐managed patients had shorter time to consultation and surgery, decreased LOS, and lower costs.23 In a retrospective cohort study, Batsis looked at mortality in 466 patients with hip fracture, and found no difference between hospitalist management and standard care.24 In patients undergoing elective hip and knee arthroplasty, Huddleston and colleagues reported that patients managed by hospitalists had fewer complications and shorter LOS. The nurses and orthopedic surgeons preferred the hospitalistorthopedist comanagement model.25
The benefits of hospitalist comanagement are not limited to adult patients undergoing orthopedic surgery. For high‐risk patients undergoing lower extremity reconstruction surgery, Pinzur and colleagues noted that LOS was shorter for a cohort of patients managed by hospitalists than for a group of historical controls not treated by hospitalists.26 Simon and colleagues studied comanagement for pediatric spinal fusion patients, and found a decrease in LOS from 6.5 to 4.8 days.27
Several factors should be considered in developing and implementing a successful comanagement program. Since comanagement duties may fall upon hospitalists in order to protect surgeons' time,28 hospital medicine groups should ensure adequate staffing prior to taking on additional services. Clear guidelines to delineate roles and responsibilities of the comanaging groups also need to be developed and implemented.29, 30
Comanaging may also involve additional training. Hospitalists who manage neurologic, neurosurgical, trauma, and psychiatric patients report being undertrained for such conditions.31, 32 Hospital medicine groups need to ensure training needs are met and supported. Given the successes of comanagement and the increasing complexity of surgical patients,33 this practice will likely expand to a greater variety of non‐medical patients.
SHORT STAY UNITS
In 2003, short stay units (SSU) were present in approximately 20% of US hospitals, with 11% of hospitals planning on opening one in the next year.34 SSU are designed to manage acute, self‐limited medical conditions that require brief staysusually less than 72 hours. Approximately 80% of SSU patients are discharged home, avoiding hospitalization.35 Historically, SSU have been under the domain of the ED; however, there is an emerging role for hospitalist‐run SSU.36
Despite demand for SSU, little research has been performed on hospitalist‐led SSU. In 2000, Abenhaim and colleagues showed that a hospitalist‐run SSU at a university‐affiliated teaching hospital had a shorter LOS and lower rates of complications and readmissions when compared to medicine teaching services.37 In 2008, Northwestern Memorial Hospital opened a 30‐bed hospitalist‐run SSU; for those patients, LOS decreased by 2 days.38 In 2010, Leykum and colleagues showed that a hospitalist‐run observation unit can decrease LOS from 2.4 days to 2.2 days.39 Careful selection of SSU patients is needed to obtain these results. Lucas and colleagues found that whether or not SSU patients required assistance of specialists was the strongest predictor of unsuccessful stays (>72 hours or inpatient conversion) in SSU.36
Whether SSU are run by hospital medicine or emergency medicine is decided at an institutional level. Location of SSU in a specifically designated area is crucial, as it allows physicians to round efficiently on patients and to work with staff trained in observation services. Development of admission criteria that include specific diagnoses which match hospitalists' scope of practice is also important (Table 1).32
| Evaluation of Diagnostic Syndromes | Treatment of Emergent Conditions |
|---|---|
| |
| Chest pain | Asthma |
| Abdominal pain | Congestive heart failure |
| Fever | Dehydration |
| Gastrointestinal bleed | Hypoglycemia or hyperglycemia |
| Syncope | Hypercalcemia |
| Dizziness | Atrial fibrillation |
| Headache | |
| Chest trauma | |
| Abdominal trauma | |
The protocol‐based and diagnosis‐specific nature of SSU may enhance quality of care through standardization. Future research may delineate the utility of SSU.
CHEST PAIN UNITS
In the United States, in 2004, approximately 6 million patients present annually to EDs with chest pain.40 Cost of care of patients unnecessarily admitted to coronary care units has been estimated to be nearly $3 billion annually.41 Still, as many as 3% of patients with acute myocardial infarction are discharged home.42 Chest pain units (CPU) were developed to facilitate evaluation of patients with chest pain, at low risk for acute coronary syndrome, without requiring inpatient admission. A number of studies have suggested that admission to a CPU is a safe and cost‐effective alternative to hospital admission.4348
CPU have traditionally been staffed by ED physicians and/or cardiologists. In a prepost study, Krantz and colleagues found that a CPU model, incorporating hospitalists at an academic public safety‐net hospital, decreased ED LOS with no difference in 30‐day cardiac event rate.49 Myers and colleagues created a hospitalist‐directed nonteaching service in an academic medical center to admit low‐risk chest pain patients. Patients admitted to the hospitalist service had a statistically significant lower median LOS (23 hours vs 33 hours) and approximately half the median hospital charges than those admitted to teaching services.50 At the same academic medical center, Bayley and colleagues showed that 91% of patients admitted for chest pain waited more than 3 hours for a bed. This adversely affected ED revenue by tying up beds, resulting in an estimated annual loss of $168,300 of hospital revenue. Creation of a hospitalist‐managed service for low‐acuity chest pain patients reduced hospital LOS by 7 hours.51 Somekh and colleagues demonstrated that a protocol‐driven, cardiologist‐run CPU results in a decreased LOS and readmission rate compared to usual care.52 In a non‐peer reviewed case study, Cox Health opened an 8‐bed, hospitalist‐led CPU in 2003. They decreased LOS from 72 to 18 hours, while increasing revenue by $2.5 million a year.53 These studies suggest that hospitalist‐run CPU can decrease LOS, increase revenue, and relieve ED overcrowding.
Development of a successful CPU depends upon clear inclusion/exclusion criteria; close collaboration among ED physicians, hospitalists, and cardiologists; the development of evidence‐based protocols, and the availability of stress testing.
ACTIVE BED MANAGEMENT
As of 2007, 90% of EDs were crowded beyond their capacity.2 ED crowding leads to ambulance diversion,54 which can delay care and increase mortality rates.55 One of the main causes of ED crowding is the boarding of admitted patients.56 Boarded, admitted patients have been shown to have decreased quality of care and patient satisfaction.35
Active bed management (ABM) by hospitalists can decrease ED diversion. Howell and colleagues instituted ABM where hospitalists, as active bed managers, facilitate placement of patients to their inpatient destinations to assist ED flow.57 This 24‐hour, hospitalist‐led, active bed management service decreased both ED LOS and ambulance diversion. The bed manager collaborated real‐time with medicine and ED attending physicians, nursing supervisors, and charge nurses to change patient care status, and assign and facilitate transfer of patients to appropriate units. These hospitalist bed managers were also empowered to activate additional resources when pre‐diversion rounds identified resource limitations and impending ED divert. They found overall ED LOS for admitted patients decreased by 98 minutes, while LOS for non‐admitted patients stayed the same. AMB decreased diversion due to critically ill and telemetry patients by 28% (786 hours), and diversion due to lower acuity patients by 6% (182 hours). This intervention proved cost‐effective. Three full‐time equivalent (FTE) hospitalists' salaries staff 1 active bed manager working 24/7. Nearly 1000 hours of diversion were avoided at an annual savings of $1086 per hour of diversion decreased.
ABM is a new frontier for hospitals in general, and hospitalists in particular. Chadaga and colleagues found that a hospital medicine‐ED team participating in active bed management, while caring for admitted patients boarded in the ED, can decrease ED diversion and improve patient flow. The percentage of patients transferred to a medicine floor and discharged within 8 hours was reduced by 67% (P 0.01), while the number of discharges from the ED of admitted medicine patients increased by 61% (P 0.001).58
To decrease initial investment, components of ABM (ED triage, bed assignment, discharge facilitation) can be instituted in parts. Hospital medicine groups with limited resources may only provide a triage service by phone for difficult ED cases. Bedside evaluations and collaboration with nursing staff to improve bed placement may be a next step, with floor and/or intensive care unit (ICU) rounds to facilitate early discharges as a final component.
OTHER AREAS
Preoperative Clinics
In 2005, SHM cited preoperative clinics as an important aspect of preoperative care.59 Sehgal and Wachter included preoperative clinics as an area for expanding the role of hospitalists in the United States.60 These clinics can decrease delays to surgery, LOS, and cancellations on the day of surgery.61 The Cleveland Clinic established the Internal Medicine Preoperative Assessment, Consultation, and Treatment (IMPACT) Center in 1997, and has decreased surgery delay rate by 49%.59 At Kaiser Bellflower Medical Center, a preoperative medicine service that provides preoperative screening decreased the number of surgical procedures cancelled on the day of surgery by more than half.62 Gates Hospitalists LLC's perioperative care decreased delay to surgery and lost operating room time.63 In order for a preoperative service to be successful, there must be buy‐in from hospitalists, surgeons, and primary care physicians, as well as adequate staffing and clinical support.59
Palliative Care
Palliative care has been identified by SHM as a core competency in hospital medicine.64 There are several key components in delivery of quality palliative care, including communication about prognosis, pain and symptom control, and hospice eligibility.65 Hospitalists are in a unique position to offer and improve palliative care for hospitalized patients. The majority of hospitalists report spending significant amounts of time caring for dying patients; thereby, hospitalists frequently provide end‐of‐life care.66, 67 Compared to community‐based physicians, patients cared for by hospitalists have higher odds of having documented family discussions regarding end‐of‐life care, and have fewer or no key symptoms (pain, anxiety, or dyspnea).66 In addition, hospitalists' availability improves response time when a patient's clinical status changes or deteriorates, leading to prompter delivery of symptom alleviation.65 Hospitalists are becoming more experienced with end‐of‐life care, as they are exposed to terminally ill patients on a daily basis. More experience leads to improved recognition of patients with limited prognosis, which leads to earlier discussions about goals of care and faster delivery of palliative care. Perhaps this could decrease LOS and be a future area of study.
Geographic Rounding
In the last 5 years, hospital administrators have promoted geographic rounding, where hospitalists see all their patients in 1 geographic location.69 The driving forces behind this include poor patient satisfaction with physician availability, large amounts of time spent by hospitalists in transit to and from patient locations, and frustrations regarding communication with nursing.70 Several groups have instituted this with success. Cleveland Clinic and Virtua Memorial Hospital have found improved patient satisfaction and decreased LOS.69, 70 O'Leary and colleagues found improved awareness of care plans by the entire team.71 Caution should be taken to assure proper physician‐to‐patient ratios, avoid physician isolation, and coordinate physician shifts with bed assignments.69 To address some of these issues, groups have used a hybrid model where a hospitalist is primarily located on one unit but can flex or overflow onto another unit.70 Steps to success with geographic rounding include buy‐in from the institution and nursing, assuring a safe physician‐to‐patient ratio, avoiding wasted beds, and facilitating multidisciplinary rounds.69
Flexible Staffing Models
In SHM's 2010 State of Hospital Medicine Report, 70% of hospitalist groups used a fixed shift‐based staffing model (ie, 7 days on/7 days off).72 Flexible staffing models in which physician coverage is adjusted to patient volume are growing in popularity. This model can be tailored for each institution by examining admission and patient volume trends to increase coverage during busy periods and decrease coverage during slower periods. Potential benefits include alleviating burn out, reducing LOS, and improving patient outcomes. Nursing data suggests that a higher patient‐to‐nursing ratio is associated with increased 30‐day mortality,73 and an ED study found that increasing physician coverage during the evening shift shortened ED LOS by 20%.74 To date, none of these endpoints have been studied for hospital medicine.
CONCLUSION
While many hospital medicine groups were started to provide acute inpatient medical care, most have found that their value to hospitals reaches beyond bedside care. With an epidemic of ED diversion and lack of access to hospital beds and services, optimizing throughput has become imperative for hospital systems. While hospital access can be improved with addition of new beds, improving throughput by decreasing LOS maximizes utilization of existing resources.
We have reviewed how hospitalists improve patient flow in acute inpatient medicine, surgical comanagement, short stay units, chest pain units, and active bed management. In each instance, the literature supports measures for decreasing LOS while maintaining or improving quality of care. Hinami and colleagues showed physician satisfaction with hospitalist‐provided patient care.75 Most studies have been limited by tracking upstream effects of improved efficiency. As there is now some evidence that decreasing LOS may increase readmissions,17 future studies should incorporate this metric into their outcomes. The effect of formal operations management principles on patient flow and bed efficiency is not well known and should be further examined.
In addition, we have touched on other areas (perioperative clinics, palliative care, geographic rounding, and flexible staffing models) where hospitalists may impact patient throughput. These areas represent excellent opportunities for future research.
Hospitalist participation in many of these areas is in its infancy. Hospital medicine programs interested in expanding their services, beyond acute inpatient care, have the opportunity to develop standards and continue research on the effect of hospital medicine‐led services on patient care and flow.
Acknowledgements
Disclosure: All authors disclose no relevant or financial conflicts of interest.
Hospitalists are physicians whose primary focus is the general medical care of hospitalized patients. Hospitalists are uniquely positioned to implement strategies to improve patient flow and efficiency.1 With emergency department (ED) diversion reaching rates upward of 70%, lack of access to inpatient beds leads to delayed care with worsened outcomes.25
To improve access to hospital beds, hospitals may increase capacity by either adding beds or by more efficiently using existing beds. Operations management principles have been applied to healthcare to ensure efficient use of beds. These include: reducing variability of scheduled admissions, remeasuring length of stay (LOS) and bed demand after implementing strategies to reduce practice variation, and employing queuing theory to generate predictions of optimal beds needed.6 The Joint Commission implemented a leadership standard (LD 04.03.11) that hospitals develop and implement plans to identify and mitigate impediments to efficient patient flow through the hospital.
To improve access, hospital leaders expect hospitalists to staff in inpatient medicine programs, surgical comanagement, short stay and chest pain units, and active bed management.7 In the following review, we define hospitalists' roles in the aforementioned programs and their effect on patient flow. We also touch on preoperative clinics, palliative care, geographic rounding, and flexible staffing models.
ACUTE INPATIENT CARE
Hospitalists are one of the fastest growing physician groups in the United States.810 Hospitalists improve efficiency and quality of care across a variety of demographic, geographic, and healthcare settings.11, 12 A 2002 retrospective cohort study in a community‐based urban teaching hospital showed that hospitalists decreased LOS by 0.61 days and lowered risk for death in the hospital (adjusted relative hazard, 0.71; 95% confidence interval [CI], 0.540.93).13 A 2004 prospective quasi‐experimental observational study done at an academic teaching hospital showed an adjusted LOS that was 16.2% lower, and adjusted cost 9.7% lower, for patients on the hospitalists' service.14 In 2007, Lindenauer and colleagues found that a national sample of hospitalists decreased LOS by 0.4 days and lowered cost by $286 per patient.15 The findings of these individual studies were supported in a 2009 systematic review of 33 studies by Peterson which showed that hospitalists decrease LOS.16 In a recent study, Kuo and Goodwin showed that while hospitalists decrease LOS and cost, the patients they care for have higher Medicare costs after discharge by $322 per patient, and are more likely to be readmitted (odds ratio, 1.08; CI, 1.041.14).17
The hospitalist model of care continues to grow, and hospitalists will soon number as many as 30,000.18 For acute medical inpatients, the evidence suggests that hospitalists improve patient flow by decreasing LOS while improving other aspects of quality of care. However, Kuo and Goodwin's findings suggest that the transition of care from inpatient to outpatient settings still requires attention.17
SURGICAL COMANAGEMENT
The Society of Hospital Medicine (SHM) core competencies include perioperative medicine.19, 20 In the 2006 SHM national survey, 85% of hospital medicine groups indicated that they participated in surgical comanagement.21
Hospitalists have improved patient flow and outcomes for orthopedic patients. Hospitalist management of hip fracture patients decreases time to surgery and LOS compared to standard care.2224 Phy and colleagues studied 466 patients for 2 years after the inception of hospital medicine comanagement of surgical patients, and found that care by hospitalists decreased LOS by 2.2 days.22 In a retrospective study of 118 patients, Roy and colleagues found that hospitalist‐managed patients had shorter time to consultation and surgery, decreased LOS, and lower costs.23 In a retrospective cohort study, Batsis looked at mortality in 466 patients with hip fracture, and found no difference between hospitalist management and standard care.24 In patients undergoing elective hip and knee arthroplasty, Huddleston and colleagues reported that patients managed by hospitalists had fewer complications and shorter LOS. The nurses and orthopedic surgeons preferred the hospitalistorthopedist comanagement model.25
The benefits of hospitalist comanagement are not limited to adult patients undergoing orthopedic surgery. For high‐risk patients undergoing lower extremity reconstruction surgery, Pinzur and colleagues noted that LOS was shorter for a cohort of patients managed by hospitalists than for a group of historical controls not treated by hospitalists.26 Simon and colleagues studied comanagement for pediatric spinal fusion patients, and found a decrease in LOS from 6.5 to 4.8 days.27
Several factors should be considered in developing and implementing a successful comanagement program. Since comanagement duties may fall upon hospitalists in order to protect surgeons' time,28 hospital medicine groups should ensure adequate staffing prior to taking on additional services. Clear guidelines to delineate roles and responsibilities of the comanaging groups also need to be developed and implemented.29, 30
Comanaging may also involve additional training. Hospitalists who manage neurologic, neurosurgical, trauma, and psychiatric patients report being undertrained for such conditions.31, 32 Hospital medicine groups need to ensure training needs are met and supported. Given the successes of comanagement and the increasing complexity of surgical patients,33 this practice will likely expand to a greater variety of non‐medical patients.
SHORT STAY UNITS
In 2003, short stay units (SSU) were present in approximately 20% of US hospitals, with 11% of hospitals planning on opening one in the next year.34 SSU are designed to manage acute, self‐limited medical conditions that require brief staysusually less than 72 hours. Approximately 80% of SSU patients are discharged home, avoiding hospitalization.35 Historically, SSU have been under the domain of the ED; however, there is an emerging role for hospitalist‐run SSU.36
Despite demand for SSU, little research has been performed on hospitalist‐led SSU. In 2000, Abenhaim and colleagues showed that a hospitalist‐run SSU at a university‐affiliated teaching hospital had a shorter LOS and lower rates of complications and readmissions when compared to medicine teaching services.37 In 2008, Northwestern Memorial Hospital opened a 30‐bed hospitalist‐run SSU; for those patients, LOS decreased by 2 days.38 In 2010, Leykum and colleagues showed that a hospitalist‐run observation unit can decrease LOS from 2.4 days to 2.2 days.39 Careful selection of SSU patients is needed to obtain these results. Lucas and colleagues found that whether or not SSU patients required assistance of specialists was the strongest predictor of unsuccessful stays (>72 hours or inpatient conversion) in SSU.36
Whether SSU are run by hospital medicine or emergency medicine is decided at an institutional level. Location of SSU in a specifically designated area is crucial, as it allows physicians to round efficiently on patients and to work with staff trained in observation services. Development of admission criteria that include specific diagnoses which match hospitalists' scope of practice is also important (Table 1).32
| Evaluation of Diagnostic Syndromes | Treatment of Emergent Conditions |
|---|---|
| |
| Chest pain | Asthma |
| Abdominal pain | Congestive heart failure |
| Fever | Dehydration |
| Gastrointestinal bleed | Hypoglycemia or hyperglycemia |
| Syncope | Hypercalcemia |
| Dizziness | Atrial fibrillation |
| Headache | |
| Chest trauma | |
| Abdominal trauma | |
The protocol‐based and diagnosis‐specific nature of SSU may enhance quality of care through standardization. Future research may delineate the utility of SSU.
CHEST PAIN UNITS
In the United States, in 2004, approximately 6 million patients present annually to EDs with chest pain.40 Cost of care of patients unnecessarily admitted to coronary care units has been estimated to be nearly $3 billion annually.41 Still, as many as 3% of patients with acute myocardial infarction are discharged home.42 Chest pain units (CPU) were developed to facilitate evaluation of patients with chest pain, at low risk for acute coronary syndrome, without requiring inpatient admission. A number of studies have suggested that admission to a CPU is a safe and cost‐effective alternative to hospital admission.4348
CPU have traditionally been staffed by ED physicians and/or cardiologists. In a prepost study, Krantz and colleagues found that a CPU model, incorporating hospitalists at an academic public safety‐net hospital, decreased ED LOS with no difference in 30‐day cardiac event rate.49 Myers and colleagues created a hospitalist‐directed nonteaching service in an academic medical center to admit low‐risk chest pain patients. Patients admitted to the hospitalist service had a statistically significant lower median LOS (23 hours vs 33 hours) and approximately half the median hospital charges than those admitted to teaching services.50 At the same academic medical center, Bayley and colleagues showed that 91% of patients admitted for chest pain waited more than 3 hours for a bed. This adversely affected ED revenue by tying up beds, resulting in an estimated annual loss of $168,300 of hospital revenue. Creation of a hospitalist‐managed service for low‐acuity chest pain patients reduced hospital LOS by 7 hours.51 Somekh and colleagues demonstrated that a protocol‐driven, cardiologist‐run CPU results in a decreased LOS and readmission rate compared to usual care.52 In a non‐peer reviewed case study, Cox Health opened an 8‐bed, hospitalist‐led CPU in 2003. They decreased LOS from 72 to 18 hours, while increasing revenue by $2.5 million a year.53 These studies suggest that hospitalist‐run CPU can decrease LOS, increase revenue, and relieve ED overcrowding.
Development of a successful CPU depends upon clear inclusion/exclusion criteria; close collaboration among ED physicians, hospitalists, and cardiologists; the development of evidence‐based protocols, and the availability of stress testing.
ACTIVE BED MANAGEMENT
As of 2007, 90% of EDs were crowded beyond their capacity.2 ED crowding leads to ambulance diversion,54 which can delay care and increase mortality rates.55 One of the main causes of ED crowding is the boarding of admitted patients.56 Boarded, admitted patients have been shown to have decreased quality of care and patient satisfaction.35
Active bed management (ABM) by hospitalists can decrease ED diversion. Howell and colleagues instituted ABM where hospitalists, as active bed managers, facilitate placement of patients to their inpatient destinations to assist ED flow.57 This 24‐hour, hospitalist‐led, active bed management service decreased both ED LOS and ambulance diversion. The bed manager collaborated real‐time with medicine and ED attending physicians, nursing supervisors, and charge nurses to change patient care status, and assign and facilitate transfer of patients to appropriate units. These hospitalist bed managers were also empowered to activate additional resources when pre‐diversion rounds identified resource limitations and impending ED divert. They found overall ED LOS for admitted patients decreased by 98 minutes, while LOS for non‐admitted patients stayed the same. AMB decreased diversion due to critically ill and telemetry patients by 28% (786 hours), and diversion due to lower acuity patients by 6% (182 hours). This intervention proved cost‐effective. Three full‐time equivalent (FTE) hospitalists' salaries staff 1 active bed manager working 24/7. Nearly 1000 hours of diversion were avoided at an annual savings of $1086 per hour of diversion decreased.
ABM is a new frontier for hospitals in general, and hospitalists in particular. Chadaga and colleagues found that a hospital medicine‐ED team participating in active bed management, while caring for admitted patients boarded in the ED, can decrease ED diversion and improve patient flow. The percentage of patients transferred to a medicine floor and discharged within 8 hours was reduced by 67% (P 0.01), while the number of discharges from the ED of admitted medicine patients increased by 61% (P 0.001).58
To decrease initial investment, components of ABM (ED triage, bed assignment, discharge facilitation) can be instituted in parts. Hospital medicine groups with limited resources may only provide a triage service by phone for difficult ED cases. Bedside evaluations and collaboration with nursing staff to improve bed placement may be a next step, with floor and/or intensive care unit (ICU) rounds to facilitate early discharges as a final component.
OTHER AREAS
Preoperative Clinics
In 2005, SHM cited preoperative clinics as an important aspect of preoperative care.59 Sehgal and Wachter included preoperative clinics as an area for expanding the role of hospitalists in the United States.60 These clinics can decrease delays to surgery, LOS, and cancellations on the day of surgery.61 The Cleveland Clinic established the Internal Medicine Preoperative Assessment, Consultation, and Treatment (IMPACT) Center in 1997, and has decreased surgery delay rate by 49%.59 At Kaiser Bellflower Medical Center, a preoperative medicine service that provides preoperative screening decreased the number of surgical procedures cancelled on the day of surgery by more than half.62 Gates Hospitalists LLC's perioperative care decreased delay to surgery and lost operating room time.63 In order for a preoperative service to be successful, there must be buy‐in from hospitalists, surgeons, and primary care physicians, as well as adequate staffing and clinical support.59
Palliative Care
Palliative care has been identified by SHM as a core competency in hospital medicine.64 There are several key components in delivery of quality palliative care, including communication about prognosis, pain and symptom control, and hospice eligibility.65 Hospitalists are in a unique position to offer and improve palliative care for hospitalized patients. The majority of hospitalists report spending significant amounts of time caring for dying patients; thereby, hospitalists frequently provide end‐of‐life care.66, 67 Compared to community‐based physicians, patients cared for by hospitalists have higher odds of having documented family discussions regarding end‐of‐life care, and have fewer or no key symptoms (pain, anxiety, or dyspnea).66 In addition, hospitalists' availability improves response time when a patient's clinical status changes or deteriorates, leading to prompter delivery of symptom alleviation.65 Hospitalists are becoming more experienced with end‐of‐life care, as they are exposed to terminally ill patients on a daily basis. More experience leads to improved recognition of patients with limited prognosis, which leads to earlier discussions about goals of care and faster delivery of palliative care. Perhaps this could decrease LOS and be a future area of study.
Geographic Rounding
In the last 5 years, hospital administrators have promoted geographic rounding, where hospitalists see all their patients in 1 geographic location.69 The driving forces behind this include poor patient satisfaction with physician availability, large amounts of time spent by hospitalists in transit to and from patient locations, and frustrations regarding communication with nursing.70 Several groups have instituted this with success. Cleveland Clinic and Virtua Memorial Hospital have found improved patient satisfaction and decreased LOS.69, 70 O'Leary and colleagues found improved awareness of care plans by the entire team.71 Caution should be taken to assure proper physician‐to‐patient ratios, avoid physician isolation, and coordinate physician shifts with bed assignments.69 To address some of these issues, groups have used a hybrid model where a hospitalist is primarily located on one unit but can flex or overflow onto another unit.70 Steps to success with geographic rounding include buy‐in from the institution and nursing, assuring a safe physician‐to‐patient ratio, avoiding wasted beds, and facilitating multidisciplinary rounds.69
Flexible Staffing Models
In SHM's 2010 State of Hospital Medicine Report, 70% of hospitalist groups used a fixed shift‐based staffing model (ie, 7 days on/7 days off).72 Flexible staffing models in which physician coverage is adjusted to patient volume are growing in popularity. This model can be tailored for each institution by examining admission and patient volume trends to increase coverage during busy periods and decrease coverage during slower periods. Potential benefits include alleviating burn out, reducing LOS, and improving patient outcomes. Nursing data suggests that a higher patient‐to‐nursing ratio is associated with increased 30‐day mortality,73 and an ED study found that increasing physician coverage during the evening shift shortened ED LOS by 20%.74 To date, none of these endpoints have been studied for hospital medicine.
CONCLUSION
While many hospital medicine groups were started to provide acute inpatient medical care, most have found that their value to hospitals reaches beyond bedside care. With an epidemic of ED diversion and lack of access to hospital beds and services, optimizing throughput has become imperative for hospital systems. While hospital access can be improved with addition of new beds, improving throughput by decreasing LOS maximizes utilization of existing resources.
We have reviewed how hospitalists improve patient flow in acute inpatient medicine, surgical comanagement, short stay units, chest pain units, and active bed management. In each instance, the literature supports measures for decreasing LOS while maintaining or improving quality of care. Hinami and colleagues showed physician satisfaction with hospitalist‐provided patient care.75 Most studies have been limited by tracking upstream effects of improved efficiency. As there is now some evidence that decreasing LOS may increase readmissions,17 future studies should incorporate this metric into their outcomes. The effect of formal operations management principles on patient flow and bed efficiency is not well known and should be further examined.
In addition, we have touched on other areas (perioperative clinics, palliative care, geographic rounding, and flexible staffing models) where hospitalists may impact patient throughput. These areas represent excellent opportunities for future research.
Hospitalist participation in many of these areas is in its infancy. Hospital medicine programs interested in expanding their services, beyond acute inpatient care, have the opportunity to develop standards and continue research on the effect of hospital medicine‐led services on patient care and flow.
Acknowledgements
Disclosure: All authors disclose no relevant or financial conflicts of interest.
- SHM Benchmarks Committee. Maximizing throughput and improving patient flow. The Hospitalist, Supplement: How Hospitalists Add Value. Philadelphia, PA: Society of Hospital Medicine; 2005. Available online at http://www.the‐hospitalist.org/details/article/279433/Maximizing_Throughput_and_Improving_Patient_Flow.html. Accessed on July 2009.
- Institute of Medicine, Committee on the Future of Emergency Care in the United States Health System. Hospital‐Based Emergency Care: At the Breaking Point. Washington, DC: National Academies Press; 2007.
- , . Emergency department crowding is associated with poor care for patients with severe pain. Ann Emerg Med. 2008;51:1–5.
- , , , et al. The impact of emergency department crowding measures on time to antibiotics for patients with community‐acquired pneumonia. Ann Emerg Med. 2007;50:510–516.
- , , , , ; for the DELAYED‐ED Study Group. Impact of delayed transfer of critically ill patients from the emergency department to the intensive care unit. Crit Care Med. 2007;35:1477–1483.
- . Managing Patient Flow in Hospitals: Strategies and Solutions, 2nd ed. In: Beurhaus P, Rudolph M, Prenney B, et al, eds. Joint Commission Resources, Joint Commission Resources, Inc., 2009.
- , , , . California hospital leader's view of hospitalists: meeting needs of the present and the future. J Hosp Med. 2009;4(9):528–534.
- , . The emerging role of “hospitalists” in the American health care system. N Engl J Med. 1996;335:514–517.
- , , , et al. Trends in market demand for internal medicine 1999–2004: an analysis of physician job advertisements. J Gen Intern Med. 2006;21:1079–1085.
- , , , et al. Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians. Ann Intern Med. 1999;130:343–349.
- , , , et al. Reorganizing an academic medical service impact on cost, quality, patient satisfaction, and education. JAMA. 1998;279:1560–1565.
- , . The hospitalist movement 5 years later. JAMA. 2002;287:487–494.
- , , , , , . Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes. Ann Intern Med. 2002;137(11):859–865.
- , , . Associations with reduced length of stay and costs on an academic hospitalist service. Am J Manag Care. 2004;10(8):561–568.
- , , , et al. Outcomes of care by hospitalists, general internists and family physicians. N Engl J Med. 2007;357(25):2589–2600.
- . Systematic review of outcomes and quality measures in adult patients cared for by hospitalists vs nonhospitalists. Mayo Clin Proc. 2009;84(3):248–254.
- , . Association of hospitalist care with medical utilization after discharge: evidence of cost shift from a cohort study. Ann Intern Med. 2011;155:152–159.
- , , , et al. The potential size of the hospitalist workforce in the United States. Am Med. 1999;106(4):441–445.
- , . Surgical co‐management: a natural evolution of hospitalist practice. J Hosp Med. 2008;3:394–397.
- , , , et al. The core competencies in hospital medicine: a framework for curriculum development. J Hosp Med. 2006;1(suppl 1):1–30.
- Society of Hospital Medicine. Co‐Management Task Force Page. Available at: http://www.hospitalmedicine.org/AM/Template.cfm? Section=Home165(7):796–801.
- , , , et al. Associations between the hospitalist model of care and quality‐of‐care‐related outcomes in patients undergoing hip fracture surgery. Mayo Clin Proc. 2006;81(1):28–31.
- . Effects of a hospitalist care model on mortality of elderly patients with hip fractures. J Hosp Med. 2007;2(4):219–225.
- , , , et al; for the Hospital‐Orthopedic Team Trial Investigators. Medical and surgical co‐management after elective hip and knee arthroplasty: a randomized, controlled trial. Ann Intern Med. 2004;141(1):28–38.
- , , , et al. Hospitalist‐orthopedic co‐management of high‐risk patients undergoing lower extremity reconstruction surgery. Orthopedics. 2009;32(7):495.
- , , , et al. Pediatric hospitalist co‐management of spinal fusion surgery patients. J Hosp Med. 2007;2:23–29.
- . Just because you can, doesn't mean that you should: a call for the rational application of hospitalist co‐management. J Hosp Med. 2008;3:398–402.
- Society of Hospital Medicine. SHM White Paper: Co‐Management White Paper. Philadelphia, PA: 2010.
- American Medical Association, Council on Ethical and Judicial Affairs. CEJA Report 5–I‐99. Ethical Implications of Surgical Co‐Management. Available at: http://www.ama‐assn.org/resources/doc/code‐medical‐ethics/8043a.pdf. Accessed November 17, 2011.
- , , , et al. Hospitalist care and length of stay in patients requiring complex discharge planning and close clinical monitoring. Arch Intern Med. 2007;167:1869–1874.
- , , , et al. Hospitalist's perceptions of their residency training needs: results of a national survey. Am J Med. 2001;111:247–254.
- , . Why perioperative medicine matters more than ever. Cleve Clin J Med. 2006;73(supp 1):S1.
- , , , et al. A national survey of observation units in the United States. Am J Emerg Med. 2003;12:529–533.
- Society of Hospital Medicine. SHM White Paper: Observation Unit White Paper. Philadelphia, PA: 2009.
- , , , et al. A hospitalist‐run short‐stay unit: features that predict length‐of‐stay and eventual admission to traditional inpatient services. J Hosp Med. 2009;4(5):276–284.
- , , , . Program description: a hospitalist‐run medical short‐stay unit in a teaching hospital. Can Med Assoc J. 2000:163(11):1477–1480.
- . Hospitalists can cut ED overcrowding. ACEP News. 2010.
- , , . Implementation of a hospitalist‐run observation unit and impact on length of stay (LOS): a brief report. J Hosp Med. 2010;5(9):E2–E5.
- , . National Hospital Ambulatory Medical Care survey: 2004 emergency department summary. Adv Data. 2006;23:1–29.
- , . Identification of chest pain patients appropriate for an emergency department observation unit. Emerg Med Clin North Am. 2001;19:35–66.
- , , . Stress testing for risk stratification of patients with low to moderate probability of acute cardiac ischemia. Emerg Med Clin North Am. 2001;19:87–103.
- , , , et al. An evaluation of a chest pain diagnostic protocol to exclude acute cardiac ischemia in the emergency department. Arch Intern Med. 1997;157:1085–1091.
- , , . The Chest Pain Evaluation Center at the University of Maryland Medical Center. Md Med J. 1994;43:1047–1052.
- , , , , . Cost effectiveness of mandatory stress testing in chest pain center patients. Ann Emerg Med. 1997;29:88–98.
- , , , et al. A rapid diagnostic and treatment center for patients with chest pain in the emergency department. Ann Emerg Med. 1995;25:1–8.
- , , , , . An emergency department‐based protocol for rapidly ruling out myocardial ischemia reduces hospital time and expense: results of a randomized study (ROMIO). J Am Coll Cardiol. 1996;28:25–33.
- , , , et al. Randomized controlled trial and economic evaluation of a chest pain observation unit compared with routine care. BMJ. 2004;328:254.
- , , , et al. A cooperative care model: cardiologists and hospitalists reduce length of stay in a chest pain observation unit. Crit Pathw Cardiol. 2005;4(2):55–58.
- , , , et al. Improving resource utilization in a teaching hospital: development of a nonteaching service for chest pain admissions. Acad Med. 2006;81(5):432–435.
- , , , et al. The financial burden of emergency department congestion and hospital crowding for chest pain patients awaiting admission. Ann Emerg Med. 2005;45(2):110–117.
- , , , , . Differences in diagnostic evaluation and clinical outcomes in the care of patients with chest pain based on admitting service: the benefits of a dedicated chest pain unit. J Nucl Cardiol. 2008;15(2):186–192.
- . Taking charge of observation units. Today's Hospitalist. July 2007.
- , , . Access block cause emergency department overcrowding and ambulance diversion in Perth, Western Australia. Emerg Med J. 2005;22:351–354.
- , , , . The relationship between distance to hospital and patient mortality in emergencies: an observational study. Emerg Med J. 2007;24:665–668.
- , . Systematic review of emergency department crowding: causes, effects, and solutions. Ann Emerg Med. 2008;52:126–136.
- , , , , , . Active bed management by hospitalists and emergency department throughput. Ann Intern Med. 2008;149:804–810.
- , , , et al. A hospitalist‐led emergency department team improves hospital bed efficiency. J Hosp Med. 2010;5(suppl 1):17–18.
- Society of Hospital Medicine. Perioperative care (a special supplement to The Hospitalist). Philadelphia, PA: Society of Hospital Medicine; 2005. Available at: http://www.hospitalmedicine.org/AM/Template.cfm?Section=Home136:591–596.
- Hospitalist Management Advisor. Hospitalist branch into preoperative medicine with preop assessments. Marblehead, MA: HCPro, 2006. Available at: http://www.hcpro.com/HOM‐57460–3615/Hospitalists‐branch‐into‐perioperative‐medicine‐with‐preop‐assessments.html. Accessed February 15, 2012.
- . The preoperative medicine service: an innovative practice at Kaiser Bellflower Medical Center. The Permanente Journal. 2002;6:13–16.
- . A preop evaluation service delivers unexpected benefits. Today's Hospitalist. January 2008.
- , , , , . The core competencies in hospital medicine: a framework for curriculum development. J Hosp Med. 2006;1:1–67.
- , , , , , . Common myths about caring for patients with terminal illness: opportunities to improve care in the hospital setting. J Hosp Med. 2007;2:357–365.
- . End‐of‐life care in a voluntary hospitalist model: effects on communication, processes of care, and patient symptoms. Am J Med. 2004;116:669–675.
- , , , . Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians. Ann Intern Med. 1999;130:343–349.
- , . Palliative care and hospitalist: an opportunity for cross‐fertilization. Am J Med. 2001;111(suppl):10S–14S.
- . Giving hospitalists their space. ACP Hospitalist. February 2008.
- . Having problems findings your patients? Today's Hospitalists. June 2010.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 24(11):1223–1227.
- Medical Group Management Association and Society of Hospital Medicine (SHM). State of Hospital Medicine 2010 Report Based on 2009. Available online at http://www.mgma.com/store/Surveys‐and‐Benchmarking/State‐of‐Hospital‐Medicine‐2010‐Report‐Based‐on‐2009 ‐Data‐Print‐Edition/.
- , , , et al. Hospital nurse staffing and patient mortality, nurse burnout, and job dissatisfaction. JAMA. 2002;288(16):1987–1993.
- , . Reduced length of stay in medical emergency department patients: a prospective controlled study on emergency physician staffing. Eur J Emerg Med. 2004;11(1):29–34.
- , , , . Provider expectations and experiences of comanagement. J Hosp Med. 2011;6(7):401–404.
- SHM Benchmarks Committee. Maximizing throughput and improving patient flow. The Hospitalist, Supplement: How Hospitalists Add Value. Philadelphia, PA: Society of Hospital Medicine; 2005. Available online at http://www.the‐hospitalist.org/details/article/279433/Maximizing_Throughput_and_Improving_Patient_Flow.html. Accessed on July 2009.
- Institute of Medicine, Committee on the Future of Emergency Care in the United States Health System. Hospital‐Based Emergency Care: At the Breaking Point. Washington, DC: National Academies Press; 2007.
- , . Emergency department crowding is associated with poor care for patients with severe pain. Ann Emerg Med. 2008;51:1–5.
- , , , et al. The impact of emergency department crowding measures on time to antibiotics for patients with community‐acquired pneumonia. Ann Emerg Med. 2007;50:510–516.
- , , , , ; for the DELAYED‐ED Study Group. Impact of delayed transfer of critically ill patients from the emergency department to the intensive care unit. Crit Care Med. 2007;35:1477–1483.
- . Managing Patient Flow in Hospitals: Strategies and Solutions, 2nd ed. In: Beurhaus P, Rudolph M, Prenney B, et al, eds. Joint Commission Resources, Joint Commission Resources, Inc., 2009.
- , , , . California hospital leader's view of hospitalists: meeting needs of the present and the future. J Hosp Med. 2009;4(9):528–534.
- , . The emerging role of “hospitalists” in the American health care system. N Engl J Med. 1996;335:514–517.
- , , , et al. Trends in market demand for internal medicine 1999–2004: an analysis of physician job advertisements. J Gen Intern Med. 2006;21:1079–1085.
- , , , et al. Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians. Ann Intern Med. 1999;130:343–349.
- , , , et al. Reorganizing an academic medical service impact on cost, quality, patient satisfaction, and education. JAMA. 1998;279:1560–1565.
- , . The hospitalist movement 5 years later. JAMA. 2002;287:487–494.
- , , , , , . Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes. Ann Intern Med. 2002;137(11):859–865.
- , , . Associations with reduced length of stay and costs on an academic hospitalist service. Am J Manag Care. 2004;10(8):561–568.
- , , , et al. Outcomes of care by hospitalists, general internists and family physicians. N Engl J Med. 2007;357(25):2589–2600.
- . Systematic review of outcomes and quality measures in adult patients cared for by hospitalists vs nonhospitalists. Mayo Clin Proc. 2009;84(3):248–254.
- , . Association of hospitalist care with medical utilization after discharge: evidence of cost shift from a cohort study. Ann Intern Med. 2011;155:152–159.
- , , , et al. The potential size of the hospitalist workforce in the United States. Am Med. 1999;106(4):441–445.
- , . Surgical co‐management: a natural evolution of hospitalist practice. J Hosp Med. 2008;3:394–397.
- , , , et al. The core competencies in hospital medicine: a framework for curriculum development. J Hosp Med. 2006;1(suppl 1):1–30.
- Society of Hospital Medicine. Co‐Management Task Force Page. Available at: http://www.hospitalmedicine.org/AM/Template.cfm? Section=Home165(7):796–801.
- , , , et al. Associations between the hospitalist model of care and quality‐of‐care‐related outcomes in patients undergoing hip fracture surgery. Mayo Clin Proc. 2006;81(1):28–31.
- . Effects of a hospitalist care model on mortality of elderly patients with hip fractures. J Hosp Med. 2007;2(4):219–225.
- , , , et al; for the Hospital‐Orthopedic Team Trial Investigators. Medical and surgical co‐management after elective hip and knee arthroplasty: a randomized, controlled trial. Ann Intern Med. 2004;141(1):28–38.
- , , , et al. Hospitalist‐orthopedic co‐management of high‐risk patients undergoing lower extremity reconstruction surgery. Orthopedics. 2009;32(7):495.
- , , , et al. Pediatric hospitalist co‐management of spinal fusion surgery patients. J Hosp Med. 2007;2:23–29.
- . Just because you can, doesn't mean that you should: a call for the rational application of hospitalist co‐management. J Hosp Med. 2008;3:398–402.
- Society of Hospital Medicine. SHM White Paper: Co‐Management White Paper. Philadelphia, PA: 2010.
- American Medical Association, Council on Ethical and Judicial Affairs. CEJA Report 5–I‐99. Ethical Implications of Surgical Co‐Management. Available at: http://www.ama‐assn.org/resources/doc/code‐medical‐ethics/8043a.pdf. Accessed November 17, 2011.
- , , , et al. Hospitalist care and length of stay in patients requiring complex discharge planning and close clinical monitoring. Arch Intern Med. 2007;167:1869–1874.
- , , , et al. Hospitalist's perceptions of their residency training needs: results of a national survey. Am J Med. 2001;111:247–254.
- , . Why perioperative medicine matters more than ever. Cleve Clin J Med. 2006;73(supp 1):S1.
- , , , et al. A national survey of observation units in the United States. Am J Emerg Med. 2003;12:529–533.
- Society of Hospital Medicine. SHM White Paper: Observation Unit White Paper. Philadelphia, PA: 2009.
- , , , et al. A hospitalist‐run short‐stay unit: features that predict length‐of‐stay and eventual admission to traditional inpatient services. J Hosp Med. 2009;4(5):276–284.
- , , , . Program description: a hospitalist‐run medical short‐stay unit in a teaching hospital. Can Med Assoc J. 2000:163(11):1477–1480.
- . Hospitalists can cut ED overcrowding. ACEP News. 2010.
- , , . Implementation of a hospitalist‐run observation unit and impact on length of stay (LOS): a brief report. J Hosp Med. 2010;5(9):E2–E5.
- , . National Hospital Ambulatory Medical Care survey: 2004 emergency department summary. Adv Data. 2006;23:1–29.
- , . Identification of chest pain patients appropriate for an emergency department observation unit. Emerg Med Clin North Am. 2001;19:35–66.
- , , . Stress testing for risk stratification of patients with low to moderate probability of acute cardiac ischemia. Emerg Med Clin North Am. 2001;19:87–103.
- , , , et al. An evaluation of a chest pain diagnostic protocol to exclude acute cardiac ischemia in the emergency department. Arch Intern Med. 1997;157:1085–1091.
- , , . The Chest Pain Evaluation Center at the University of Maryland Medical Center. Md Med J. 1994;43:1047–1052.
- , , , , . Cost effectiveness of mandatory stress testing in chest pain center patients. Ann Emerg Med. 1997;29:88–98.
- , , , et al. A rapid diagnostic and treatment center for patients with chest pain in the emergency department. Ann Emerg Med. 1995;25:1–8.
- , , , , . An emergency department‐based protocol for rapidly ruling out myocardial ischemia reduces hospital time and expense: results of a randomized study (ROMIO). J Am Coll Cardiol. 1996;28:25–33.
- , , , et al. Randomized controlled trial and economic evaluation of a chest pain observation unit compared with routine care. BMJ. 2004;328:254.
- , , , et al. A cooperative care model: cardiologists and hospitalists reduce length of stay in a chest pain observation unit. Crit Pathw Cardiol. 2005;4(2):55–58.
- , , , et al. Improving resource utilization in a teaching hospital: development of a nonteaching service for chest pain admissions. Acad Med. 2006;81(5):432–435.
- , , , et al. The financial burden of emergency department congestion and hospital crowding for chest pain patients awaiting admission. Ann Emerg Med. 2005;45(2):110–117.
- , , , , . Differences in diagnostic evaluation and clinical outcomes in the care of patients with chest pain based on admitting service: the benefits of a dedicated chest pain unit. J Nucl Cardiol. 2008;15(2):186–192.
- . Taking charge of observation units. Today's Hospitalist. July 2007.
- , , . Access block cause emergency department overcrowding and ambulance diversion in Perth, Western Australia. Emerg Med J. 2005;22:351–354.
- , , , . The relationship between distance to hospital and patient mortality in emergencies: an observational study. Emerg Med J. 2007;24:665–668.
- , . Systematic review of emergency department crowding: causes, effects, and solutions. Ann Emerg Med. 2008;52:126–136.
- , , , , , . Active bed management by hospitalists and emergency department throughput. Ann Intern Med. 2008;149:804–810.
- , , , et al. A hospitalist‐led emergency department team improves hospital bed efficiency. J Hosp Med. 2010;5(suppl 1):17–18.
- Society of Hospital Medicine. Perioperative care (a special supplement to The Hospitalist). Philadelphia, PA: Society of Hospital Medicine; 2005. Available at: http://www.hospitalmedicine.org/AM/Template.cfm?Section=Home136:591–596.
- Hospitalist Management Advisor. Hospitalist branch into preoperative medicine with preop assessments. Marblehead, MA: HCPro, 2006. Available at: http://www.hcpro.com/HOM‐57460–3615/Hospitalists‐branch‐into‐perioperative‐medicine‐with‐preop‐assessments.html. Accessed February 15, 2012.
- . The preoperative medicine service: an innovative practice at Kaiser Bellflower Medical Center. The Permanente Journal. 2002;6:13–16.
- . A preop evaluation service delivers unexpected benefits. Today's Hospitalist. January 2008.
- , , , , . The core competencies in hospital medicine: a framework for curriculum development. J Hosp Med. 2006;1:1–67.
- , , , , , . Common myths about caring for patients with terminal illness: opportunities to improve care in the hospital setting. J Hosp Med. 2007;2:357–365.
- . End‐of‐life care in a voluntary hospitalist model: effects on communication, processes of care, and patient symptoms. Am J Med. 2004;116:669–675.
- , , , . Hospitalists and the practice of inpatient medicine: results of a survey of the National Association of Inpatient Physicians. Ann Intern Med. 1999;130:343–349.
- , . Palliative care and hospitalist: an opportunity for cross‐fertilization. Am J Med. 2001;111(suppl):10S–14S.
- . Giving hospitalists their space. ACP Hospitalist. February 2008.
- . Having problems findings your patients? Today's Hospitalists. June 2010.
- , , , et al. Impact of localizing physicians to hospital units on nurse–physician communication and agreement on the plan of care. J Gen Intern Med. 24(11):1223–1227.
- Medical Group Management Association and Society of Hospital Medicine (SHM). State of Hospital Medicine 2010 Report Based on 2009. Available online at http://www.mgma.com/store/Surveys‐and‐Benchmarking/State‐of‐Hospital‐Medicine‐2010‐Report‐Based‐on‐2009 ‐Data‐Print‐Edition/.
- , , , et al. Hospital nurse staffing and patient mortality, nurse burnout, and job dissatisfaction. JAMA. 2002;288(16):1987–1993.
- , . Reduced length of stay in medical emergency department patients: a prospective controlled study on emergency physician staffing. Eur J Emerg Med. 2004;11(1):29–34.
- , , , . Provider expectations and experiences of comanagement. J Hosp Med. 2011;6(7):401–404.
Hospitalist Physical Diagnosis Curricula
Deficiencies in physical examination skills among medical students, housestaff, and even faculty have been reported for decades. For example, cardiac examination skills have been shown to improve during the early years of medical school and then plateau by the third year, with no measurable improvement through residency or beyond without fellowship training.13
Despite efforts by residency programs to promote bedside teaching, many general medicine faculty shy away due to lack of confidence and comfort in teaching physical diagnosis.4 The presence of hospitalists increases satisfaction among trainees with inpatient teaching,5 and medical faculty's ability has been shown to positively impact medical students' test scores.6 This lack of bedside teaching is a missed opportunity, as general medicine faculty are assuming more teaching responsibility.
The Merrin Bedside Teaching Program was founded in 2004 at New York University (NYU) School of Medicine to improve the quality of bedside teaching in the Department of Medicine/Division of General Internal Medicine. In this article, we describe the development process and early outcomes of this program.
METHODS
The principal teaching institution of NYU is Bellevue Hospital, an 800‐bed tertiary care safety‐net facility located in New York City. Approximately 30 general medicine faculty members at Bellevue Hospital attend on the inpatient teaching service 12 weeks per year each, with the remaining time spent in the hospital‐based clinics and other inpatient services.
Prior to the initiation of this program, our faculty did not receive formal instruction in bedside teaching, in general, or in teaching physical diagnosis, in particular. Housestaff and students were reporting that bedside rounds were not being conducted consistently and were of variable educational quality. Among a number of our residency and fellowship programs, there were concerns about meeting Accreditation Counsel for Graduate Medical Education (ACGME) requirements for quantity and quality of bedside rounds and faculty development.
To inform program development, we conducted a literature review, a series of focus groups with residents and faculty, and a survey of 39 general medicine hospital and ambulatory‐based clinician teachers to determine the perceived bedside teaching faculty development needs of our department. Then, over the next 2 years, we recruited 32 hospitalists and fellowship faculty to participate in a videotaped bedside teaching simulation. Each attending reviewed the videotape one‐on‐one with an experienced facilitator (A.K.), who elicited the attending's goals and instructional models and methods used for clinical teaching. To address the wide variety in teaching approaches identified in the assessment, 4 to 6 attendings met weekly for 1 month to observe each other conducting rounds with their teams. The facilitator of these groups used materials derived from relevant medical education theory79 and related conceptual models to frame the debriefing. Participants enthusiastically supported the educational value of this learner‐centered, experiential teaching approach.
We integrated this needs assessment with an individualized approach, incorporating learner goal‐setting with interactive and highly experiential teaching strategies,7,9,10 to create the Merrin Bedside Teaching Program in 2004. This program recruits faculty, with reputations as excellent teachers, to design a program to develop their own bedside teaching skills and disseminate what they learn to their peers. Faculty fellows are recruited through an open call for applications, which includes a letter of support from a supervisor and a detailed independent learning plan, including an identified mentor. Fellows are selected by the program's executive committee based on the likelihood of the success of their proposed program. A stipend equivalent of between 5% and 10% of base salary is provided to each fellow for a period of 2 years. Selected faculty fellows are encouraged to focus on an aspect of the physical examination, work in groups of 2 or 3, and to identify and recruit a mentor who is considered a master clinician in the target specialty. Master clinicians are given an honorarium to acknowledge their selection and incentivize them to spend time with the faculty fellows. This is funded with philanthropic support from the Merrin Family Foundation.
Fellows are guided by program leadership in their independent study, development of clinical teaching skills, and curriculum development using the same theory‐driven, systematic approach that framed program inception.911 Bedside rounds are the core instructional method used by each group of fellows and are supplemented by lectures, interactive small‐group seminars, and Web‐based modules in certain cases. Bedside sessions are run by the master‐clinician mentor until the faculty fellows are deemed competent by the mentor and feel confident enough to lead independently.
Since 2004, there have been 14 fellows who have developed programs focused on the examination of the heart, skin, knee, and shoulder. Program development is underway in motivational interviewing, the pulmonary examination, and the examination of the critically ill patient. We describe the work of the first 4 fellows as an example of how this fellowship creates value for the individual fellows, our departmental teaching programs, and the medical school.
Our first cohort of fellows chose, out of personal interest, to concentrate on the cardiac examination. They spent the first year working with highly respected cardiologists to hone their own clinical skills, reviewing the literature on the evidence‐based cardiac physical examination and effective teaching methods,12,13 researching the use of electronic stethoscopes and related technology for teaching at the bedside, and piloting a variety of approaches to teaching their busy colleagues these skills.
Bedside rounds focus on pertinent physical findings with an emphasis on an evidence‐based approach. We find we are most effective when the patient's diagnosis is unknown by the group leader to avoid bias when formulating the differential diagnosis. Sessions include a discussion of how the exam correlates with the diagnosis, relevant pathophysiology, imaging, and treatment options.
Two, 1‐hour‐long lectures in cardiac examination are delivered: the first reviews basics of heart sounds, both normal and abnormal, and the second reviews the most common systolic and diastolic murmurs. These lectures, scheduled into routine faculty conference time, utilize a PowerPoint format, with an overview of basic physiology and pathophysiology, aided by phonocardiograms, frequency spectrographs, and audio recordings delivered via a loudspeaker. Interactive cases offered by Blaufuss Multimedia (Rolling Hills Estates, CA) are an excellent teaching tool that incorporate case presentations, videos of key physical findings, auscultatory recordings, and relevant pathophysiology. We initially used this resource because of its high quality and ease of use; we now use our own interactive case presentations, which allow for flexibility with content and style, and which reinforce the prevalence of interesting cases at our institution to the audience members.
Technology has proven to be an invaluable tool in teaching cardiac physical diagnosis, both at the bedside and in the classroom. Electronic stethoscopes provide the ability to record heart sounds for use in teaching venues on short notice, such as morning report, and for use in creating the interactive case presentations described above. The electronic stethoscopes we use can be wired to peripheral devices, such as camcorders, iPods, and speaker pads. Speaker pads are devices, approximately the size of a stethoscope head, that can be connected by wires in series, each attached to a stethoscope, allowing a group of people to listen to the same sounds simultaneously with excellent sound reproduction. This technology allows each person standing around the bedside to listen to a patient while the group leader auscultates and explains the findings in real time. There are distinct advantages of simultaneous auscultation both for describing auditory findings and minimizing discomfort to the patient.
Applications are available for the iPod (Stethoscope App, Thinklabs Technology, LLC, Centennial, CO) which can record and display real‐time phonocardiography when attached to an electronic stethoscope, even at the terminus of a speaker pad chain. This application also allows recorded sounds to be played directly through a speaker, or transferred to a computer with the corresponding phonocardiographic and spectrographic images, that can all be incorporated into an interactive case presentation. Frequency spectrographs allow visualization of differences between low‐ and high‐frequency sounds, which, in conjunction with the timing and amplitude displayed by phonocardiography, can aid in teaching subtle findings, such as shapes of murmurs, patterns of splitting, gallups, etc.14 Playback of heart sounds in a conference room setting can be challenging, given the often subtle and low‐frequency findings typical of cardiac pathology, and is effectively achieved by using a musician‐quality loudspeaker. We have found that speaker pads offer the best sound quality at the bedside, although they are inconvenient for larger groups.
DISCUSSION
A new framework has been proposed for considering faculty development programs that focuses on the participants, program, content, facilitator, and the context in which the program occurs.15 We have effectively addressed and synthesized these components in a rich, high impact, learner‐centered faculty development program that also responds to challenges raised by changes in the health delivery system, concerns about accreditation requirements, and targeted local needs assessment.
We have been fortunate to recruit specialty faculty who are outstanding teachers, have welcomed the fellows into their clinics, and have dedicated countless hours to supervision and education. An unintended, but important, outcome of the program is that we are able to highlight the exceptional skills of our senior, experienced clinicians. These are colleagues who all too often do not receive adequate recognition in the modern‐day academic medical center environment, but who are undoubtedly invaluable to the education mission of these centers.
The existence of the program has resulted in our general medicine faculty showing great enthusiasm, both to develop an area of expertise and to participate as learners in the programs developed by peers. The faculty fellows in each specialty have become a valuable resource to peer faculty, residents, and medical students alike, who are now less dependent on consultants to identify and explain physical findings. The faculty teaching the knee and shoulder exams started a Sports Medicine Clinic within primary care, and assist with joint injections throughout the clinic. In addition to providing clinical support, their educational curriculum is included in both the attending and housestaff conference schedules. The cardiac lectures, both didactic and interactive case presentations, are included in the attending conference schedule, intern and resident core curricula, and the third‐year medicine clerkship lecture series. The dermatology group has created a series of comprehensive online modules that provide content tailored to general medicine. All this durable material is available broadly to trainees of all professions in our medical center.
Given the ever‐growing burden of patient care and extra‐clinical responsibilities, the principal factor limiting the effectiveness of bedside rounds is faculty availability. Despite this, all of our hospitalists have attended at least 1 bedside cardiac session, and the majority have attended multiple times. Varying the time and day of the sessions, offering to join attending rounds, and being available for impromptu diagnostic consultations have maximized the fellows' contact with faculty, residents, and students.
Although funding for evaluation of the program has been limited, a research agenda is emerging. Both the pulmonary physical exam and critical care groups are in the process of evaluating the effectiveness of their programs on the quality of bedside rounds, student and resident learning, and, to the extent possible, on patient care.
CONCLUSION
We believe wholeheartedly that bedside instruction both in physical diagnosis and interview skills must not become a lost art. General medicine faculty are ideally situated to take on this challenge. An educational program targeting hospitalists and general medicine faculty energizes faculty and leverages local resources to fill in gaps in skills for faculty and then for trainees. Generalist faculty relish the opportunity to champion a particular element of the doctorpatient encounter, which has contributed to our ultimate goal of strengthening the core diagnostic skills of our faculty who are at the forefront of clinical care and medical education.
Acknowledgements
The authors thank the Merrin Family for their generous support of the program; Drs Gregory Mints, Tanping Wong, and Sabrina Felson for their initial work in developing the Merrin Faculty Development Program; and Dr Martin Kahn for his tireless dedication to mentorship and bedside teaching.
Disclosure: Drs Janjigian, Charap, and Kalet report receiving funding from the Merrin Family Foundation.
- , . Cardiac auscultatory skills of internal medicine and family practice trainees. A comparison of diagnostic proficiency. JAMA. 1997;278(9):717–722.
- , , , et al. Competency in cardiac examination skills in medical students, trainees, physicians, and faculty: a multicenter study. Arch Intern Med. 2006;166(6):610–616.
- , , , et al. Confidential testing of cardiac examination competency in cardiology and noncardiology faculty and trainees: a multicenter study. Clin Cardiol. 2010;33(12):738–745.
- , , , . Whither bedside teaching? A focus‐group study of clinical teachers. Acad Med. 2003;78(4):384–390.
- , , , . Effect of hospitalist attending physicians on trainee educational experiences: a systematic review. J Hosp Med. 2009;4(8):490–498.
- , , , , , . Is there a relationship between attending physicians' and residents' teaching skills and students' examination scores? Acad Med. 2000;75(11):1144–1146.
- , , , . A five‐step “microskills” model of clinical teaching. J Am Board Fam Pract. 1992;5(4):419–424.
- , , , , . Attributes of excellent attending‐physician role models. N Engl J Med. 1998;339(27):1986–1993.
- , . Teaching at the bedside: a new model. Med Teach. 2003;25(2):127–130.
- . A theory‐based faculty development program for clinician‐educators. Acad Med. 2000;75(5):498–501.
- Kern D, Thomas P, Howard D, Bass E, ed. Curriculum Development for Medical Education: A Six‐Step Approach. Baltimore, MD: The Johns Hopkins University Press; 1998.
- , , , , . Innovative web‐based multimedia curriculum improves cardiac examination competency of residents. J Hosp Med. 2008;3(2):124–133.
- , , , , . Using virtual patients to improve cardiac examination competency in medical students. Clin Cardiol. 2008;31(7):334–339.
- . Cardiac auscultation: a glorious past—and it does have a future! Circulation. 2006;113(9):1255–1259.
- , . Reframing research on faculty development. Acad Med. 2011;86(4):421–428.
Deficiencies in physical examination skills among medical students, housestaff, and even faculty have been reported for decades. For example, cardiac examination skills have been shown to improve during the early years of medical school and then plateau by the third year, with no measurable improvement through residency or beyond without fellowship training.13
Despite efforts by residency programs to promote bedside teaching, many general medicine faculty shy away due to lack of confidence and comfort in teaching physical diagnosis.4 The presence of hospitalists increases satisfaction among trainees with inpatient teaching,5 and medical faculty's ability has been shown to positively impact medical students' test scores.6 This lack of bedside teaching is a missed opportunity, as general medicine faculty are assuming more teaching responsibility.
The Merrin Bedside Teaching Program was founded in 2004 at New York University (NYU) School of Medicine to improve the quality of bedside teaching in the Department of Medicine/Division of General Internal Medicine. In this article, we describe the development process and early outcomes of this program.
METHODS
The principal teaching institution of NYU is Bellevue Hospital, an 800‐bed tertiary care safety‐net facility located in New York City. Approximately 30 general medicine faculty members at Bellevue Hospital attend on the inpatient teaching service 12 weeks per year each, with the remaining time spent in the hospital‐based clinics and other inpatient services.
Prior to the initiation of this program, our faculty did not receive formal instruction in bedside teaching, in general, or in teaching physical diagnosis, in particular. Housestaff and students were reporting that bedside rounds were not being conducted consistently and were of variable educational quality. Among a number of our residency and fellowship programs, there were concerns about meeting Accreditation Counsel for Graduate Medical Education (ACGME) requirements for quantity and quality of bedside rounds and faculty development.
To inform program development, we conducted a literature review, a series of focus groups with residents and faculty, and a survey of 39 general medicine hospital and ambulatory‐based clinician teachers to determine the perceived bedside teaching faculty development needs of our department. Then, over the next 2 years, we recruited 32 hospitalists and fellowship faculty to participate in a videotaped bedside teaching simulation. Each attending reviewed the videotape one‐on‐one with an experienced facilitator (A.K.), who elicited the attending's goals and instructional models and methods used for clinical teaching. To address the wide variety in teaching approaches identified in the assessment, 4 to 6 attendings met weekly for 1 month to observe each other conducting rounds with their teams. The facilitator of these groups used materials derived from relevant medical education theory79 and related conceptual models to frame the debriefing. Participants enthusiastically supported the educational value of this learner‐centered, experiential teaching approach.
We integrated this needs assessment with an individualized approach, incorporating learner goal‐setting with interactive and highly experiential teaching strategies,7,9,10 to create the Merrin Bedside Teaching Program in 2004. This program recruits faculty, with reputations as excellent teachers, to design a program to develop their own bedside teaching skills and disseminate what they learn to their peers. Faculty fellows are recruited through an open call for applications, which includes a letter of support from a supervisor and a detailed independent learning plan, including an identified mentor. Fellows are selected by the program's executive committee based on the likelihood of the success of their proposed program. A stipend equivalent of between 5% and 10% of base salary is provided to each fellow for a period of 2 years. Selected faculty fellows are encouraged to focus on an aspect of the physical examination, work in groups of 2 or 3, and to identify and recruit a mentor who is considered a master clinician in the target specialty. Master clinicians are given an honorarium to acknowledge their selection and incentivize them to spend time with the faculty fellows. This is funded with philanthropic support from the Merrin Family Foundation.
Fellows are guided by program leadership in their independent study, development of clinical teaching skills, and curriculum development using the same theory‐driven, systematic approach that framed program inception.911 Bedside rounds are the core instructional method used by each group of fellows and are supplemented by lectures, interactive small‐group seminars, and Web‐based modules in certain cases. Bedside sessions are run by the master‐clinician mentor until the faculty fellows are deemed competent by the mentor and feel confident enough to lead independently.
Since 2004, there have been 14 fellows who have developed programs focused on the examination of the heart, skin, knee, and shoulder. Program development is underway in motivational interviewing, the pulmonary examination, and the examination of the critically ill patient. We describe the work of the first 4 fellows as an example of how this fellowship creates value for the individual fellows, our departmental teaching programs, and the medical school.
Our first cohort of fellows chose, out of personal interest, to concentrate on the cardiac examination. They spent the first year working with highly respected cardiologists to hone their own clinical skills, reviewing the literature on the evidence‐based cardiac physical examination and effective teaching methods,12,13 researching the use of electronic stethoscopes and related technology for teaching at the bedside, and piloting a variety of approaches to teaching their busy colleagues these skills.
Bedside rounds focus on pertinent physical findings with an emphasis on an evidence‐based approach. We find we are most effective when the patient's diagnosis is unknown by the group leader to avoid bias when formulating the differential diagnosis. Sessions include a discussion of how the exam correlates with the diagnosis, relevant pathophysiology, imaging, and treatment options.
Two, 1‐hour‐long lectures in cardiac examination are delivered: the first reviews basics of heart sounds, both normal and abnormal, and the second reviews the most common systolic and diastolic murmurs. These lectures, scheduled into routine faculty conference time, utilize a PowerPoint format, with an overview of basic physiology and pathophysiology, aided by phonocardiograms, frequency spectrographs, and audio recordings delivered via a loudspeaker. Interactive cases offered by Blaufuss Multimedia (Rolling Hills Estates, CA) are an excellent teaching tool that incorporate case presentations, videos of key physical findings, auscultatory recordings, and relevant pathophysiology. We initially used this resource because of its high quality and ease of use; we now use our own interactive case presentations, which allow for flexibility with content and style, and which reinforce the prevalence of interesting cases at our institution to the audience members.
Technology has proven to be an invaluable tool in teaching cardiac physical diagnosis, both at the bedside and in the classroom. Electronic stethoscopes provide the ability to record heart sounds for use in teaching venues on short notice, such as morning report, and for use in creating the interactive case presentations described above. The electronic stethoscopes we use can be wired to peripheral devices, such as camcorders, iPods, and speaker pads. Speaker pads are devices, approximately the size of a stethoscope head, that can be connected by wires in series, each attached to a stethoscope, allowing a group of people to listen to the same sounds simultaneously with excellent sound reproduction. This technology allows each person standing around the bedside to listen to a patient while the group leader auscultates and explains the findings in real time. There are distinct advantages of simultaneous auscultation both for describing auditory findings and minimizing discomfort to the patient.
Applications are available for the iPod (Stethoscope App, Thinklabs Technology, LLC, Centennial, CO) which can record and display real‐time phonocardiography when attached to an electronic stethoscope, even at the terminus of a speaker pad chain. This application also allows recorded sounds to be played directly through a speaker, or transferred to a computer with the corresponding phonocardiographic and spectrographic images, that can all be incorporated into an interactive case presentation. Frequency spectrographs allow visualization of differences between low‐ and high‐frequency sounds, which, in conjunction with the timing and amplitude displayed by phonocardiography, can aid in teaching subtle findings, such as shapes of murmurs, patterns of splitting, gallups, etc.14 Playback of heart sounds in a conference room setting can be challenging, given the often subtle and low‐frequency findings typical of cardiac pathology, and is effectively achieved by using a musician‐quality loudspeaker. We have found that speaker pads offer the best sound quality at the bedside, although they are inconvenient for larger groups.
DISCUSSION
A new framework has been proposed for considering faculty development programs that focuses on the participants, program, content, facilitator, and the context in which the program occurs.15 We have effectively addressed and synthesized these components in a rich, high impact, learner‐centered faculty development program that also responds to challenges raised by changes in the health delivery system, concerns about accreditation requirements, and targeted local needs assessment.
We have been fortunate to recruit specialty faculty who are outstanding teachers, have welcomed the fellows into their clinics, and have dedicated countless hours to supervision and education. An unintended, but important, outcome of the program is that we are able to highlight the exceptional skills of our senior, experienced clinicians. These are colleagues who all too often do not receive adequate recognition in the modern‐day academic medical center environment, but who are undoubtedly invaluable to the education mission of these centers.
The existence of the program has resulted in our general medicine faculty showing great enthusiasm, both to develop an area of expertise and to participate as learners in the programs developed by peers. The faculty fellows in each specialty have become a valuable resource to peer faculty, residents, and medical students alike, who are now less dependent on consultants to identify and explain physical findings. The faculty teaching the knee and shoulder exams started a Sports Medicine Clinic within primary care, and assist with joint injections throughout the clinic. In addition to providing clinical support, their educational curriculum is included in both the attending and housestaff conference schedules. The cardiac lectures, both didactic and interactive case presentations, are included in the attending conference schedule, intern and resident core curricula, and the third‐year medicine clerkship lecture series. The dermatology group has created a series of comprehensive online modules that provide content tailored to general medicine. All this durable material is available broadly to trainees of all professions in our medical center.
Given the ever‐growing burden of patient care and extra‐clinical responsibilities, the principal factor limiting the effectiveness of bedside rounds is faculty availability. Despite this, all of our hospitalists have attended at least 1 bedside cardiac session, and the majority have attended multiple times. Varying the time and day of the sessions, offering to join attending rounds, and being available for impromptu diagnostic consultations have maximized the fellows' contact with faculty, residents, and students.
Although funding for evaluation of the program has been limited, a research agenda is emerging. Both the pulmonary physical exam and critical care groups are in the process of evaluating the effectiveness of their programs on the quality of bedside rounds, student and resident learning, and, to the extent possible, on patient care.
CONCLUSION
We believe wholeheartedly that bedside instruction both in physical diagnosis and interview skills must not become a lost art. General medicine faculty are ideally situated to take on this challenge. An educational program targeting hospitalists and general medicine faculty energizes faculty and leverages local resources to fill in gaps in skills for faculty and then for trainees. Generalist faculty relish the opportunity to champion a particular element of the doctorpatient encounter, which has contributed to our ultimate goal of strengthening the core diagnostic skills of our faculty who are at the forefront of clinical care and medical education.
Acknowledgements
The authors thank the Merrin Family for their generous support of the program; Drs Gregory Mints, Tanping Wong, and Sabrina Felson for their initial work in developing the Merrin Faculty Development Program; and Dr Martin Kahn for his tireless dedication to mentorship and bedside teaching.
Disclosure: Drs Janjigian, Charap, and Kalet report receiving funding from the Merrin Family Foundation.
Deficiencies in physical examination skills among medical students, housestaff, and even faculty have been reported for decades. For example, cardiac examination skills have been shown to improve during the early years of medical school and then plateau by the third year, with no measurable improvement through residency or beyond without fellowship training.13
Despite efforts by residency programs to promote bedside teaching, many general medicine faculty shy away due to lack of confidence and comfort in teaching physical diagnosis.4 The presence of hospitalists increases satisfaction among trainees with inpatient teaching,5 and medical faculty's ability has been shown to positively impact medical students' test scores.6 This lack of bedside teaching is a missed opportunity, as general medicine faculty are assuming more teaching responsibility.
The Merrin Bedside Teaching Program was founded in 2004 at New York University (NYU) School of Medicine to improve the quality of bedside teaching in the Department of Medicine/Division of General Internal Medicine. In this article, we describe the development process and early outcomes of this program.
METHODS
The principal teaching institution of NYU is Bellevue Hospital, an 800‐bed tertiary care safety‐net facility located in New York City. Approximately 30 general medicine faculty members at Bellevue Hospital attend on the inpatient teaching service 12 weeks per year each, with the remaining time spent in the hospital‐based clinics and other inpatient services.
Prior to the initiation of this program, our faculty did not receive formal instruction in bedside teaching, in general, or in teaching physical diagnosis, in particular. Housestaff and students were reporting that bedside rounds were not being conducted consistently and were of variable educational quality. Among a number of our residency and fellowship programs, there were concerns about meeting Accreditation Counsel for Graduate Medical Education (ACGME) requirements for quantity and quality of bedside rounds and faculty development.
To inform program development, we conducted a literature review, a series of focus groups with residents and faculty, and a survey of 39 general medicine hospital and ambulatory‐based clinician teachers to determine the perceived bedside teaching faculty development needs of our department. Then, over the next 2 years, we recruited 32 hospitalists and fellowship faculty to participate in a videotaped bedside teaching simulation. Each attending reviewed the videotape one‐on‐one with an experienced facilitator (A.K.), who elicited the attending's goals and instructional models and methods used for clinical teaching. To address the wide variety in teaching approaches identified in the assessment, 4 to 6 attendings met weekly for 1 month to observe each other conducting rounds with their teams. The facilitator of these groups used materials derived from relevant medical education theory79 and related conceptual models to frame the debriefing. Participants enthusiastically supported the educational value of this learner‐centered, experiential teaching approach.
We integrated this needs assessment with an individualized approach, incorporating learner goal‐setting with interactive and highly experiential teaching strategies,7,9,10 to create the Merrin Bedside Teaching Program in 2004. This program recruits faculty, with reputations as excellent teachers, to design a program to develop their own bedside teaching skills and disseminate what they learn to their peers. Faculty fellows are recruited through an open call for applications, which includes a letter of support from a supervisor and a detailed independent learning plan, including an identified mentor. Fellows are selected by the program's executive committee based on the likelihood of the success of their proposed program. A stipend equivalent of between 5% and 10% of base salary is provided to each fellow for a period of 2 years. Selected faculty fellows are encouraged to focus on an aspect of the physical examination, work in groups of 2 or 3, and to identify and recruit a mentor who is considered a master clinician in the target specialty. Master clinicians are given an honorarium to acknowledge their selection and incentivize them to spend time with the faculty fellows. This is funded with philanthropic support from the Merrin Family Foundation.
Fellows are guided by program leadership in their independent study, development of clinical teaching skills, and curriculum development using the same theory‐driven, systematic approach that framed program inception.911 Bedside rounds are the core instructional method used by each group of fellows and are supplemented by lectures, interactive small‐group seminars, and Web‐based modules in certain cases. Bedside sessions are run by the master‐clinician mentor until the faculty fellows are deemed competent by the mentor and feel confident enough to lead independently.
Since 2004, there have been 14 fellows who have developed programs focused on the examination of the heart, skin, knee, and shoulder. Program development is underway in motivational interviewing, the pulmonary examination, and the examination of the critically ill patient. We describe the work of the first 4 fellows as an example of how this fellowship creates value for the individual fellows, our departmental teaching programs, and the medical school.
Our first cohort of fellows chose, out of personal interest, to concentrate on the cardiac examination. They spent the first year working with highly respected cardiologists to hone their own clinical skills, reviewing the literature on the evidence‐based cardiac physical examination and effective teaching methods,12,13 researching the use of electronic stethoscopes and related technology for teaching at the bedside, and piloting a variety of approaches to teaching their busy colleagues these skills.
Bedside rounds focus on pertinent physical findings with an emphasis on an evidence‐based approach. We find we are most effective when the patient's diagnosis is unknown by the group leader to avoid bias when formulating the differential diagnosis. Sessions include a discussion of how the exam correlates with the diagnosis, relevant pathophysiology, imaging, and treatment options.
Two, 1‐hour‐long lectures in cardiac examination are delivered: the first reviews basics of heart sounds, both normal and abnormal, and the second reviews the most common systolic and diastolic murmurs. These lectures, scheduled into routine faculty conference time, utilize a PowerPoint format, with an overview of basic physiology and pathophysiology, aided by phonocardiograms, frequency spectrographs, and audio recordings delivered via a loudspeaker. Interactive cases offered by Blaufuss Multimedia (Rolling Hills Estates, CA) are an excellent teaching tool that incorporate case presentations, videos of key physical findings, auscultatory recordings, and relevant pathophysiology. We initially used this resource because of its high quality and ease of use; we now use our own interactive case presentations, which allow for flexibility with content and style, and which reinforce the prevalence of interesting cases at our institution to the audience members.
Technology has proven to be an invaluable tool in teaching cardiac physical diagnosis, both at the bedside and in the classroom. Electronic stethoscopes provide the ability to record heart sounds for use in teaching venues on short notice, such as morning report, and for use in creating the interactive case presentations described above. The electronic stethoscopes we use can be wired to peripheral devices, such as camcorders, iPods, and speaker pads. Speaker pads are devices, approximately the size of a stethoscope head, that can be connected by wires in series, each attached to a stethoscope, allowing a group of people to listen to the same sounds simultaneously with excellent sound reproduction. This technology allows each person standing around the bedside to listen to a patient while the group leader auscultates and explains the findings in real time. There are distinct advantages of simultaneous auscultation both for describing auditory findings and minimizing discomfort to the patient.
Applications are available for the iPod (Stethoscope App, Thinklabs Technology, LLC, Centennial, CO) which can record and display real‐time phonocardiography when attached to an electronic stethoscope, even at the terminus of a speaker pad chain. This application also allows recorded sounds to be played directly through a speaker, or transferred to a computer with the corresponding phonocardiographic and spectrographic images, that can all be incorporated into an interactive case presentation. Frequency spectrographs allow visualization of differences between low‐ and high‐frequency sounds, which, in conjunction with the timing and amplitude displayed by phonocardiography, can aid in teaching subtle findings, such as shapes of murmurs, patterns of splitting, gallups, etc.14 Playback of heart sounds in a conference room setting can be challenging, given the often subtle and low‐frequency findings typical of cardiac pathology, and is effectively achieved by using a musician‐quality loudspeaker. We have found that speaker pads offer the best sound quality at the bedside, although they are inconvenient for larger groups.
DISCUSSION
A new framework has been proposed for considering faculty development programs that focuses on the participants, program, content, facilitator, and the context in which the program occurs.15 We have effectively addressed and synthesized these components in a rich, high impact, learner‐centered faculty development program that also responds to challenges raised by changes in the health delivery system, concerns about accreditation requirements, and targeted local needs assessment.
We have been fortunate to recruit specialty faculty who are outstanding teachers, have welcomed the fellows into their clinics, and have dedicated countless hours to supervision and education. An unintended, but important, outcome of the program is that we are able to highlight the exceptional skills of our senior, experienced clinicians. These are colleagues who all too often do not receive adequate recognition in the modern‐day academic medical center environment, but who are undoubtedly invaluable to the education mission of these centers.
The existence of the program has resulted in our general medicine faculty showing great enthusiasm, both to develop an area of expertise and to participate as learners in the programs developed by peers. The faculty fellows in each specialty have become a valuable resource to peer faculty, residents, and medical students alike, who are now less dependent on consultants to identify and explain physical findings. The faculty teaching the knee and shoulder exams started a Sports Medicine Clinic within primary care, and assist with joint injections throughout the clinic. In addition to providing clinical support, their educational curriculum is included in both the attending and housestaff conference schedules. The cardiac lectures, both didactic and interactive case presentations, are included in the attending conference schedule, intern and resident core curricula, and the third‐year medicine clerkship lecture series. The dermatology group has created a series of comprehensive online modules that provide content tailored to general medicine. All this durable material is available broadly to trainees of all professions in our medical center.
Given the ever‐growing burden of patient care and extra‐clinical responsibilities, the principal factor limiting the effectiveness of bedside rounds is faculty availability. Despite this, all of our hospitalists have attended at least 1 bedside cardiac session, and the majority have attended multiple times. Varying the time and day of the sessions, offering to join attending rounds, and being available for impromptu diagnostic consultations have maximized the fellows' contact with faculty, residents, and students.
Although funding for evaluation of the program has been limited, a research agenda is emerging. Both the pulmonary physical exam and critical care groups are in the process of evaluating the effectiveness of their programs on the quality of bedside rounds, student and resident learning, and, to the extent possible, on patient care.
CONCLUSION
We believe wholeheartedly that bedside instruction both in physical diagnosis and interview skills must not become a lost art. General medicine faculty are ideally situated to take on this challenge. An educational program targeting hospitalists and general medicine faculty energizes faculty and leverages local resources to fill in gaps in skills for faculty and then for trainees. Generalist faculty relish the opportunity to champion a particular element of the doctorpatient encounter, which has contributed to our ultimate goal of strengthening the core diagnostic skills of our faculty who are at the forefront of clinical care and medical education.
Acknowledgements
The authors thank the Merrin Family for their generous support of the program; Drs Gregory Mints, Tanping Wong, and Sabrina Felson for their initial work in developing the Merrin Faculty Development Program; and Dr Martin Kahn for his tireless dedication to mentorship and bedside teaching.
Disclosure: Drs Janjigian, Charap, and Kalet report receiving funding from the Merrin Family Foundation.
- , . Cardiac auscultatory skills of internal medicine and family practice trainees. A comparison of diagnostic proficiency. JAMA. 1997;278(9):717–722.
- , , , et al. Competency in cardiac examination skills in medical students, trainees, physicians, and faculty: a multicenter study. Arch Intern Med. 2006;166(6):610–616.
- , , , et al. Confidential testing of cardiac examination competency in cardiology and noncardiology faculty and trainees: a multicenter study. Clin Cardiol. 2010;33(12):738–745.
- , , , . Whither bedside teaching? A focus‐group study of clinical teachers. Acad Med. 2003;78(4):384–390.
- , , , . Effect of hospitalist attending physicians on trainee educational experiences: a systematic review. J Hosp Med. 2009;4(8):490–498.
- , , , , , . Is there a relationship between attending physicians' and residents' teaching skills and students' examination scores? Acad Med. 2000;75(11):1144–1146.
- , , , . A five‐step “microskills” model of clinical teaching. J Am Board Fam Pract. 1992;5(4):419–424.
- , , , , . Attributes of excellent attending‐physician role models. N Engl J Med. 1998;339(27):1986–1993.
- , . Teaching at the bedside: a new model. Med Teach. 2003;25(2):127–130.
- . A theory‐based faculty development program for clinician‐educators. Acad Med. 2000;75(5):498–501.
- Kern D, Thomas P, Howard D, Bass E, ed. Curriculum Development for Medical Education: A Six‐Step Approach. Baltimore, MD: The Johns Hopkins University Press; 1998.
- , , , , . Innovative web‐based multimedia curriculum improves cardiac examination competency of residents. J Hosp Med. 2008;3(2):124–133.
- , , , , . Using virtual patients to improve cardiac examination competency in medical students. Clin Cardiol. 2008;31(7):334–339.
- . Cardiac auscultation: a glorious past—and it does have a future! Circulation. 2006;113(9):1255–1259.
- , . Reframing research on faculty development. Acad Med. 2011;86(4):421–428.
- , . Cardiac auscultatory skills of internal medicine and family practice trainees. A comparison of diagnostic proficiency. JAMA. 1997;278(9):717–722.
- , , , et al. Competency in cardiac examination skills in medical students, trainees, physicians, and faculty: a multicenter study. Arch Intern Med. 2006;166(6):610–616.
- , , , et al. Confidential testing of cardiac examination competency in cardiology and noncardiology faculty and trainees: a multicenter study. Clin Cardiol. 2010;33(12):738–745.
- , , , . Whither bedside teaching? A focus‐group study of clinical teachers. Acad Med. 2003;78(4):384–390.
- , , , . Effect of hospitalist attending physicians on trainee educational experiences: a systematic review. J Hosp Med. 2009;4(8):490–498.
- , , , , , . Is there a relationship between attending physicians' and residents' teaching skills and students' examination scores? Acad Med. 2000;75(11):1144–1146.
- , , , . A five‐step “microskills” model of clinical teaching. J Am Board Fam Pract. 1992;5(4):419–424.
- , , , , . Attributes of excellent attending‐physician role models. N Engl J Med. 1998;339(27):1986–1993.
- , . Teaching at the bedside: a new model. Med Teach. 2003;25(2):127–130.
- . A theory‐based faculty development program for clinician‐educators. Acad Med. 2000;75(5):498–501.
- Kern D, Thomas P, Howard D, Bass E, ed. Curriculum Development for Medical Education: A Six‐Step Approach. Baltimore, MD: The Johns Hopkins University Press; 1998.
- , , , , . Innovative web‐based multimedia curriculum improves cardiac examination competency of residents. J Hosp Med. 2008;3(2):124–133.
- , , , , . Using virtual patients to improve cardiac examination competency in medical students. Clin Cardiol. 2008;31(7):334–339.
- . Cardiac auscultation: a glorious past—and it does have a future! Circulation. 2006;113(9):1255–1259.
- , . Reframing research on faculty development. Acad Med. 2011;86(4):421–428.
FDA Clears First Test to ID Bacteria Associated with Bloodstream Infections
Last month, the Food and Drug Administration cleared a test designed to quickly identify a dozen types of bacteria that can lead to bloodstream infections.
The Verigene GP Blood Culture Nucleic Acid Test, developed by molecular diagnostic firm Nanosphere Inc. of Northbrook, Ill., can identify Staphylococcus (including methicillin-resistant S. aureus, or MRSA), Streptococcus, Enterococcus (including vancomycin-resistant enterococci), and Listeria.
"The current standard of treatment is to provide broad-spectrum antibiotics, including some last-line therapies, such as vancomycin, in order to get coverage for everything," says Mike McGarrity, a Nanosphere executive. "With antibiotic stewardship programs in the majority of hospitals, there is an understanding of the overuse of these last-line therapies and the development of resistance."
Currently, blood cultures can take two to four days to identify certain types of bacteria and determine whether any present are resistant to certain therapies. Once a blood culture is positive, the Nanosphere test can identify bacteria and antimicrobial resistance genes in roughly two and half hours. In a pitch that McGarrity believes will resonate with HM groups, he positions the product as a cost-saver that can reduce length of stay (LOS) for hospitalized patients, as physicians don’t have to wait two days for test results. Quicker identification can also lead to lower mortality rates, he says.
McGarrity, who says Nanosphere will submit an application to the FDA this year for a similar rapid-results test for Clostridium difficile and a broad enteric panel, adds that the test is $75 per use. With LOS reduction and cost savings for targeted de-escalated therapies, he says, there is clear value in the test.
"This gets the attention of stakeholders," he says.
Last month, the Food and Drug Administration cleared a test designed to quickly identify a dozen types of bacteria that can lead to bloodstream infections.
The Verigene GP Blood Culture Nucleic Acid Test, developed by molecular diagnostic firm Nanosphere Inc. of Northbrook, Ill., can identify Staphylococcus (including methicillin-resistant S. aureus, or MRSA), Streptococcus, Enterococcus (including vancomycin-resistant enterococci), and Listeria.
"The current standard of treatment is to provide broad-spectrum antibiotics, including some last-line therapies, such as vancomycin, in order to get coverage for everything," says Mike McGarrity, a Nanosphere executive. "With antibiotic stewardship programs in the majority of hospitals, there is an understanding of the overuse of these last-line therapies and the development of resistance."
Currently, blood cultures can take two to four days to identify certain types of bacteria and determine whether any present are resistant to certain therapies. Once a blood culture is positive, the Nanosphere test can identify bacteria and antimicrobial resistance genes in roughly two and half hours. In a pitch that McGarrity believes will resonate with HM groups, he positions the product as a cost-saver that can reduce length of stay (LOS) for hospitalized patients, as physicians don’t have to wait two days for test results. Quicker identification can also lead to lower mortality rates, he says.
McGarrity, who says Nanosphere will submit an application to the FDA this year for a similar rapid-results test for Clostridium difficile and a broad enteric panel, adds that the test is $75 per use. With LOS reduction and cost savings for targeted de-escalated therapies, he says, there is clear value in the test.
"This gets the attention of stakeholders," he says.
Last month, the Food and Drug Administration cleared a test designed to quickly identify a dozen types of bacteria that can lead to bloodstream infections.
The Verigene GP Blood Culture Nucleic Acid Test, developed by molecular diagnostic firm Nanosphere Inc. of Northbrook, Ill., can identify Staphylococcus (including methicillin-resistant S. aureus, or MRSA), Streptococcus, Enterococcus (including vancomycin-resistant enterococci), and Listeria.
"The current standard of treatment is to provide broad-spectrum antibiotics, including some last-line therapies, such as vancomycin, in order to get coverage for everything," says Mike McGarrity, a Nanosphere executive. "With antibiotic stewardship programs in the majority of hospitals, there is an understanding of the overuse of these last-line therapies and the development of resistance."
Currently, blood cultures can take two to four days to identify certain types of bacteria and determine whether any present are resistant to certain therapies. Once a blood culture is positive, the Nanosphere test can identify bacteria and antimicrobial resistance genes in roughly two and half hours. In a pitch that McGarrity believes will resonate with HM groups, he positions the product as a cost-saver that can reduce length of stay (LOS) for hospitalized patients, as physicians don’t have to wait two days for test results. Quicker identification can also lead to lower mortality rates, he says.
McGarrity, who says Nanosphere will submit an application to the FDA this year for a similar rapid-results test for Clostridium difficile and a broad enteric panel, adds that the test is $75 per use. With LOS reduction and cost savings for targeted de-escalated therapies, he says, there is clear value in the test.
"This gets the attention of stakeholders," he says.
ITL: Physician Reviews of HM-Relevant Research
Clinical question: Do oral fluoroquinolones increase the risk of retinal detachment?
Background: Fluoroquinolones are increasingly used in both inpatient and outpatient settings, given their broad antimicrobial coverage. However, adverse effects, including those related to connective tissue and the eye, are increasingly reported. Whether that also includes retinal detachment is not yet known.
Study design: Nested case control study.
Setting: Canadian province.
Synopsis: Using data from administrative databases to identify patients who visited ophthalmologists in British Columbia between 2000 and 2007, the investigators identified 4,384 cases of retinal detachment, and matched those cases to controls at a rate of 10:1. Current, recent, and past fluoroquinolone usage was the exposure of interest.
Patients actively taking a fluoroquinolone had a higher risk of retinal detachment compared with those not taking the drug (adjusted RR of 4.5, 95% CI of 3.56-5.70). Prior or recent use of a fluoroquinolone did not increase the rate of retinal detachment. The patients were more likely to be male, myopic, diabetic, and have a prior history of cataract surgery. Ciprofloxacin was the drug most frequently involved, but this is not adjusted by frequency of prescription. Despite this association, the actual outcome is quite rare (approximately 1,440 cases per year in the U.S.).
This study has the benefit of a large amount of data and captures prescription data well. It relied on coding to identify the cases and might have missed or inappropriately categorized some cases. Despite these caveats, this study adds to the concerning adverse events due to the increasing use of fluoroquinolone therapy, and hospitalists should use appropriate clinical judgment when prescribing and educating patients about the risks and benefits.
Bottom line: Fluoroquinolone use might increase the rate of retinal detachment in patients, but the absolute risk of the event is low.
Citation: Etminan M, Forooghian F, Brophy JM, Bird ST, Maberley D. Oral fluoroquinolones and the risk of retinal detachment. JAMA. 2012;307:1414-1419.
Clinical question: Do oral fluoroquinolones increase the risk of retinal detachment?
Background: Fluoroquinolones are increasingly used in both inpatient and outpatient settings, given their broad antimicrobial coverage. However, adverse effects, including those related to connective tissue and the eye, are increasingly reported. Whether that also includes retinal detachment is not yet known.
Study design: Nested case control study.
Setting: Canadian province.
Synopsis: Using data from administrative databases to identify patients who visited ophthalmologists in British Columbia between 2000 and 2007, the investigators identified 4,384 cases of retinal detachment, and matched those cases to controls at a rate of 10:1. Current, recent, and past fluoroquinolone usage was the exposure of interest.
Patients actively taking a fluoroquinolone had a higher risk of retinal detachment compared with those not taking the drug (adjusted RR of 4.5, 95% CI of 3.56-5.70). Prior or recent use of a fluoroquinolone did not increase the rate of retinal detachment. The patients were more likely to be male, myopic, diabetic, and have a prior history of cataract surgery. Ciprofloxacin was the drug most frequently involved, but this is not adjusted by frequency of prescription. Despite this association, the actual outcome is quite rare (approximately 1,440 cases per year in the U.S.).
This study has the benefit of a large amount of data and captures prescription data well. It relied on coding to identify the cases and might have missed or inappropriately categorized some cases. Despite these caveats, this study adds to the concerning adverse events due to the increasing use of fluoroquinolone therapy, and hospitalists should use appropriate clinical judgment when prescribing and educating patients about the risks and benefits.
Bottom line: Fluoroquinolone use might increase the rate of retinal detachment in patients, but the absolute risk of the event is low.
Citation: Etminan M, Forooghian F, Brophy JM, Bird ST, Maberley D. Oral fluoroquinolones and the risk of retinal detachment. JAMA. 2012;307:1414-1419.
Clinical question: Do oral fluoroquinolones increase the risk of retinal detachment?
Background: Fluoroquinolones are increasingly used in both inpatient and outpatient settings, given their broad antimicrobial coverage. However, adverse effects, including those related to connective tissue and the eye, are increasingly reported. Whether that also includes retinal detachment is not yet known.
Study design: Nested case control study.
Setting: Canadian province.
Synopsis: Using data from administrative databases to identify patients who visited ophthalmologists in British Columbia between 2000 and 2007, the investigators identified 4,384 cases of retinal detachment, and matched those cases to controls at a rate of 10:1. Current, recent, and past fluoroquinolone usage was the exposure of interest.
Patients actively taking a fluoroquinolone had a higher risk of retinal detachment compared with those not taking the drug (adjusted RR of 4.5, 95% CI of 3.56-5.70). Prior or recent use of a fluoroquinolone did not increase the rate of retinal detachment. The patients were more likely to be male, myopic, diabetic, and have a prior history of cataract surgery. Ciprofloxacin was the drug most frequently involved, but this is not adjusted by frequency of prescription. Despite this association, the actual outcome is quite rare (approximately 1,440 cases per year in the U.S.).
This study has the benefit of a large amount of data and captures prescription data well. It relied on coding to identify the cases and might have missed or inappropriately categorized some cases. Despite these caveats, this study adds to the concerning adverse events due to the increasing use of fluoroquinolone therapy, and hospitalists should use appropriate clinical judgment when prescribing and educating patients about the risks and benefits.
Bottom line: Fluoroquinolone use might increase the rate of retinal detachment in patients, but the absolute risk of the event is low.
Citation: Etminan M, Forooghian F, Brophy JM, Bird ST, Maberley D. Oral fluoroquinolones and the risk of retinal detachment. JAMA. 2012;307:1414-1419.
Hemophilia patients plagued by professional challenges
PARIS—Results of a large study suggest that 8 in 10 hemophilia patients feel
their disorder has had a negative impact on their career.
In fact, 1 in 5
patients said they have lost a job because of their condition. However, only 8% said they were unable to find employment because of it.
These findings were presented at the World Federation of Hemophilia 2012 World Congress. The data are part of the HERO study, which
evaluated the psychosocial aspects of living with hemophilia. The study
was sponsored by Novo Nordisk.
“Treatment of hemophilia has advanced greatly over the past decades, but psychosocial issues can still be a challenge for people with hemophilia . . . ,” said Alfonso Iorio, MD, PhD, a member of the HERO International Advisory Board and a professor at McMaster University in Canada.
With this in mind, HERO researchers examined hemophilia’s effects on relationships, sexual health, and quality of life. The investigators also assessed treatment and management of the disorder, patients’ knowledge and education levels, and how hemophilia has impacted their professional life.
For the career analysis, researchers interviewed 605 patients with hemophilia. Forty percent of patients said their hemophilia played a major role in their choice of profession or job training, but 21% of patients said their disorder had no impact on their job choice.
Thirty percent of patients said their current treatment allows them to work in most situations. But 21% of patients said they have to restrict their hours due to their hemophilia, and 17% said they must work flexible hours.
Twenty-four percent of patients said they have voluntarily left a job because of their hemophilia, and 22% thought they lost a job because of their disorder.
Twenty-two percent of patients believed they were not hired for a job because of their hemophilia, and 14% felt they had not received a promotion because of it. But 10% of patients said their disorder actually helped them land a job.
These results were presented at the World Federation of Hemophilia 2012 World Congress on Monday as poster 199. Other results from the HERO study will also be presented at the meeting, which is taking place July 8-12.
“The insights from the HERO study will provide much-needed evidence to support advocating for better comprehensive hemophilia care,” Dr Iorio said. ![]()
PARIS—Results of a large study suggest that 8 in 10 hemophilia patients feel
their disorder has had a negative impact on their career.
In fact, 1 in 5
patients said they have lost a job because of their condition. However, only 8% said they were unable to find employment because of it.
These findings were presented at the World Federation of Hemophilia 2012 World Congress. The data are part of the HERO study, which
evaluated the psychosocial aspects of living with hemophilia. The study
was sponsored by Novo Nordisk.
“Treatment of hemophilia has advanced greatly over the past decades, but psychosocial issues can still be a challenge for people with hemophilia . . . ,” said Alfonso Iorio, MD, PhD, a member of the HERO International Advisory Board and a professor at McMaster University in Canada.
With this in mind, HERO researchers examined hemophilia’s effects on relationships, sexual health, and quality of life. The investigators also assessed treatment and management of the disorder, patients’ knowledge and education levels, and how hemophilia has impacted their professional life.
For the career analysis, researchers interviewed 605 patients with hemophilia. Forty percent of patients said their hemophilia played a major role in their choice of profession or job training, but 21% of patients said their disorder had no impact on their job choice.
Thirty percent of patients said their current treatment allows them to work in most situations. But 21% of patients said they have to restrict their hours due to their hemophilia, and 17% said they must work flexible hours.
Twenty-four percent of patients said they have voluntarily left a job because of their hemophilia, and 22% thought they lost a job because of their disorder.
Twenty-two percent of patients believed they were not hired for a job because of their hemophilia, and 14% felt they had not received a promotion because of it. But 10% of patients said their disorder actually helped them land a job.
These results were presented at the World Federation of Hemophilia 2012 World Congress on Monday as poster 199. Other results from the HERO study will also be presented at the meeting, which is taking place July 8-12.
“The insights from the HERO study will provide much-needed evidence to support advocating for better comprehensive hemophilia care,” Dr Iorio said. ![]()
PARIS—Results of a large study suggest that 8 in 10 hemophilia patients feel
their disorder has had a negative impact on their career.
In fact, 1 in 5
patients said they have lost a job because of their condition. However, only 8% said they were unable to find employment because of it.
These findings were presented at the World Federation of Hemophilia 2012 World Congress. The data are part of the HERO study, which
evaluated the psychosocial aspects of living with hemophilia. The study
was sponsored by Novo Nordisk.
“Treatment of hemophilia has advanced greatly over the past decades, but psychosocial issues can still be a challenge for people with hemophilia . . . ,” said Alfonso Iorio, MD, PhD, a member of the HERO International Advisory Board and a professor at McMaster University in Canada.
With this in mind, HERO researchers examined hemophilia’s effects on relationships, sexual health, and quality of life. The investigators also assessed treatment and management of the disorder, patients’ knowledge and education levels, and how hemophilia has impacted their professional life.
For the career analysis, researchers interviewed 605 patients with hemophilia. Forty percent of patients said their hemophilia played a major role in their choice of profession or job training, but 21% of patients said their disorder had no impact on their job choice.
Thirty percent of patients said their current treatment allows them to work in most situations. But 21% of patients said they have to restrict their hours due to their hemophilia, and 17% said they must work flexible hours.
Twenty-four percent of patients said they have voluntarily left a job because of their hemophilia, and 22% thought they lost a job because of their disorder.
Twenty-two percent of patients believed they were not hired for a job because of their hemophilia, and 14% felt they had not received a promotion because of it. But 10% of patients said their disorder actually helped them land a job.
These results were presented at the World Federation of Hemophilia 2012 World Congress on Monday as poster 199. Other results from the HERO study will also be presented at the meeting, which is taking place July 8-12.
“The insights from the HERO study will provide much-needed evidence to support advocating for better comprehensive hemophilia care,” Dr Iorio said. ![]()
Tech Takes Off: Videoconferences in medical settings is more acceptable and affordable, but hurdles remain
Picture this likely scenario: You’re a hospitalist in a remote setting, and a patient with stroke symptoms is rushed in by ambulance. Numbness has overcome one side of his body. Dizziness disrupts his balance, his speech becomes slurred, and his vision is blurred. Treatment must be started swiftly to halt irreversible brain damage. The nearest neurologist is located hours away, but thanks to advanced video technology, you’re able to instantly consult face to face with that specialist to help ensure optimal recovery for the patient.
Such applications of telemedicine are becoming more mainstream and affordable, facilitating discussions and decisions between healthcare providers while improving patient access to specialty care in emergencies and other situations.
Remote hospitalist services include videoconferencing for patient monitoring and assessment of various clinical services, says Jona
Advantages and Challenges
Remote patient monitoring in ICUs is on the upswing, filling gaps in the shortage of physicians specializing in critical care. Some unit administrators have established off-site command centers for these specialists to follow multiple facilities with the assistance of video technology and to intervene at urgent times.1
In a neonatal ICU, this type of live-feed technology allows for a face-to-face interaction with a pediatric pulmonologist, for example, when a premature infant is exhibiting symptoms of respiratory distress in the middle of the night, says David Cattell-Gordon, MSW, director of the Office of Telemedicine at the University of Virginia in Charlottesville.
Similarly, in rural areas where women don’t have immediate access to high-risk obstetricians, telemedicine makes it possible to consult with maternal-fetal medicine specialists from a distance, boosting the chances for pregnant mothers with complex conditions to carry healthy babies to term, says Cattell-Gordon. “Our approach has been to bring telemedicine to hospitals and clinics in communities where that resource [specialists] otherwise is unavailable,” he adds.
—Matthew Harbison, MD, medical director, Sound Physicians hospitalist services, Memorial Hermann-Texas Medical Center
Compared with telephone conversations, the advantages of video consultations are multifold: They display a patient’s facial expressions, gestures, and other body language, which might assist with the diagnosis and prescribed treatment, says Kerry Weiner, MD, chief clinical officer for IPC: The Hospitalist Company in North Hollywood, Calif., which has a presence in about 900 facilities in 25 states.
When the strength of that assessment depends on visual inspection, the technology can be particularly helpful. “The weak part of it is when you need to touch” to guide that assessment, Dr. Weiner says. That’s when the technology isn’t as useful. Still, he adds, “We use teleconferencing all over the place in a Skype-like manner, only more sophisticated. It’s more encrypted.”
Interacting within a secure network is crucial to protect privacy, says Peter Kragel, MD, clinical director of the Telemedicine Center at East Carolina University’s Brody School of Medicine in Greenville, N.C. As with any form of communication that transmits identifiable patient information, healthcare providers must comply with HIPAA guidelines when employing videoconferencing services similar to Skype.
“Because of concerns about compliance with encryption and confidentiality regulations, we do not use [videoconferencing] here,” Dr. Kragel says.
Additionally, “telemedicine isn’t always appropriate for patient care,” Linkous says. “All of this depends on the circumstances and needs of the patient. Obviously, surgery requires a direct physician-patient interaction, except for robotic surgery.” For hospitals that don’t have any neurology coverage, telemedicine robots can assist with outside consults for time-sensitive stroke care.
—Jonathan D. Linkous, CEO, American Telemedicine Association
Videoconferencing isn’t necessary for all telemedicine encounters, Linkous says. Teledermatology and retinal screening use “store and forward” communication of images, which allows for the electronic transmission of images and documents in non-emergent situations in which immediate video isn’t necessary.
“As a society, we’ve become more comfortable with the technology,” says Matthew Harbison, MD, medical director of Sound Physicians hospitalist services at Memorial Hermann-Texas Medical Center in Houston. “And as the technology continues to develop, ultimately there will be [more of] a role, but how large that will be is difficult to predict.” He adds that “the advantages are obviously in low-staffed places or staffing-challenged sites.”
Moving Ahead
As experts continue to iron out the kinks and as communities obtain greater access to broadband signals, telemedicine equipment is moving to advanced high-definition platforms. Meanwhile, the expense has come down considerably since its inception in the mid-1990s. A high-definition setup that once cost upward of $130,000 is now available for less than $10,000, Cattell-Gordon says.
The digital transmission also can assist in patient follow-up after discharge from the hospital and in monitoring various chronic diseases from home. It’s an effective tool for medical staff meetings and training purposes as well.
IPC's hospitalists have been using the technology to communicate with each other, brainstorming across regions of the country. “Because we’re a national company,” Dr. Weiner says, “this has changed the game in terms of being able to collaborate.”
Susan Kreimer is a freelance medical writer based in New York.
Reference
1. Thomas EJ, Lucke JF, Wueste L, Weavind L, Patel B. Association of telemedicine for remote monitoring of intensive care patients with mortality, complications, and length of stay. JAMA. 2009;302:2671-2678.
Picture this likely scenario: You’re a hospitalist in a remote setting, and a patient with stroke symptoms is rushed in by ambulance. Numbness has overcome one side of his body. Dizziness disrupts his balance, his speech becomes slurred, and his vision is blurred. Treatment must be started swiftly to halt irreversible brain damage. The nearest neurologist is located hours away, but thanks to advanced video technology, you’re able to instantly consult face to face with that specialist to help ensure optimal recovery for the patient.
Such applications of telemedicine are becoming more mainstream and affordable, facilitating discussions and decisions between healthcare providers while improving patient access to specialty care in emergencies and other situations.
Remote hospitalist services include videoconferencing for patient monitoring and assessment of various clinical services, says Jona
Advantages and Challenges
Remote patient monitoring in ICUs is on the upswing, filling gaps in the shortage of physicians specializing in critical care. Some unit administrators have established off-site command centers for these specialists to follow multiple facilities with the assistance of video technology and to intervene at urgent times.1
In a neonatal ICU, this type of live-feed technology allows for a face-to-face interaction with a pediatric pulmonologist, for example, when a premature infant is exhibiting symptoms of respiratory distress in the middle of the night, says David Cattell-Gordon, MSW, director of the Office of Telemedicine at the University of Virginia in Charlottesville.
Similarly, in rural areas where women don’t have immediate access to high-risk obstetricians, telemedicine makes it possible to consult with maternal-fetal medicine specialists from a distance, boosting the chances for pregnant mothers with complex conditions to carry healthy babies to term, says Cattell-Gordon. “Our approach has been to bring telemedicine to hospitals and clinics in communities where that resource [specialists] otherwise is unavailable,” he adds.
—Matthew Harbison, MD, medical director, Sound Physicians hospitalist services, Memorial Hermann-Texas Medical Center
Compared with telephone conversations, the advantages of video consultations are multifold: They display a patient’s facial expressions, gestures, and other body language, which might assist with the diagnosis and prescribed treatment, says Kerry Weiner, MD, chief clinical officer for IPC: The Hospitalist Company in North Hollywood, Calif., which has a presence in about 900 facilities in 25 states.
When the strength of that assessment depends on visual inspection, the technology can be particularly helpful. “The weak part of it is when you need to touch” to guide that assessment, Dr. Weiner says. That’s when the technology isn’t as useful. Still, he adds, “We use teleconferencing all over the place in a Skype-like manner, only more sophisticated. It’s more encrypted.”
Interacting within a secure network is crucial to protect privacy, says Peter Kragel, MD, clinical director of the Telemedicine Center at East Carolina University’s Brody School of Medicine in Greenville, N.C. As with any form of communication that transmits identifiable patient information, healthcare providers must comply with HIPAA guidelines when employing videoconferencing services similar to Skype.
“Because of concerns about compliance with encryption and confidentiality regulations, we do not use [videoconferencing] here,” Dr. Kragel says.
Additionally, “telemedicine isn’t always appropriate for patient care,” Linkous says. “All of this depends on the circumstances and needs of the patient. Obviously, surgery requires a direct physician-patient interaction, except for robotic surgery.” For hospitals that don’t have any neurology coverage, telemedicine robots can assist with outside consults for time-sensitive stroke care.
—Jonathan D. Linkous, CEO, American Telemedicine Association
Videoconferencing isn’t necessary for all telemedicine encounters, Linkous says. Teledermatology and retinal screening use “store and forward” communication of images, which allows for the electronic transmission of images and documents in non-emergent situations in which immediate video isn’t necessary.
“As a society, we’ve become more comfortable with the technology,” says Matthew Harbison, MD, medical director of Sound Physicians hospitalist services at Memorial Hermann-Texas Medical Center in Houston. “And as the technology continues to develop, ultimately there will be [more of] a role, but how large that will be is difficult to predict.” He adds that “the advantages are obviously in low-staffed places or staffing-challenged sites.”
Moving Ahead
As experts continue to iron out the kinks and as communities obtain greater access to broadband signals, telemedicine equipment is moving to advanced high-definition platforms. Meanwhile, the expense has come down considerably since its inception in the mid-1990s. A high-definition setup that once cost upward of $130,000 is now available for less than $10,000, Cattell-Gordon says.
The digital transmission also can assist in patient follow-up after discharge from the hospital and in monitoring various chronic diseases from home. It’s an effective tool for medical staff meetings and training purposes as well.
IPC's hospitalists have been using the technology to communicate with each other, brainstorming across regions of the country. “Because we’re a national company,” Dr. Weiner says, “this has changed the game in terms of being able to collaborate.”
Susan Kreimer is a freelance medical writer based in New York.
Reference
1. Thomas EJ, Lucke JF, Wueste L, Weavind L, Patel B. Association of telemedicine for remote monitoring of intensive care patients with mortality, complications, and length of stay. JAMA. 2009;302:2671-2678.
Picture this likely scenario: You’re a hospitalist in a remote setting, and a patient with stroke symptoms is rushed in by ambulance. Numbness has overcome one side of his body. Dizziness disrupts his balance, his speech becomes slurred, and his vision is blurred. Treatment must be started swiftly to halt irreversible brain damage. The nearest neurologist is located hours away, but thanks to advanced video technology, you’re able to instantly consult face to face with that specialist to help ensure optimal recovery for the patient.
Such applications of telemedicine are becoming more mainstream and affordable, facilitating discussions and decisions between healthcare providers while improving patient access to specialty care in emergencies and other situations.
Remote hospitalist services include videoconferencing for patient monitoring and assessment of various clinical services, says Jona
Advantages and Challenges
Remote patient monitoring in ICUs is on the upswing, filling gaps in the shortage of physicians specializing in critical care. Some unit administrators have established off-site command centers for these specialists to follow multiple facilities with the assistance of video technology and to intervene at urgent times.1
In a neonatal ICU, this type of live-feed technology allows for a face-to-face interaction with a pediatric pulmonologist, for example, when a premature infant is exhibiting symptoms of respiratory distress in the middle of the night, says David Cattell-Gordon, MSW, director of the Office of Telemedicine at the University of Virginia in Charlottesville.
Similarly, in rural areas where women don’t have immediate access to high-risk obstetricians, telemedicine makes it possible to consult with maternal-fetal medicine specialists from a distance, boosting the chances for pregnant mothers with complex conditions to carry healthy babies to term, says Cattell-Gordon. “Our approach has been to bring telemedicine to hospitals and clinics in communities where that resource [specialists] otherwise is unavailable,” he adds.
—Matthew Harbison, MD, medical director, Sound Physicians hospitalist services, Memorial Hermann-Texas Medical Center
Compared with telephone conversations, the advantages of video consultations are multifold: They display a patient’s facial expressions, gestures, and other body language, which might assist with the diagnosis and prescribed treatment, says Kerry Weiner, MD, chief clinical officer for IPC: The Hospitalist Company in North Hollywood, Calif., which has a presence in about 900 facilities in 25 states.
When the strength of that assessment depends on visual inspection, the technology can be particularly helpful. “The weak part of it is when you need to touch” to guide that assessment, Dr. Weiner says. That’s when the technology isn’t as useful. Still, he adds, “We use teleconferencing all over the place in a Skype-like manner, only more sophisticated. It’s more encrypted.”
Interacting within a secure network is crucial to protect privacy, says Peter Kragel, MD, clinical director of the Telemedicine Center at East Carolina University’s Brody School of Medicine in Greenville, N.C. As with any form of communication that transmits identifiable patient information, healthcare providers must comply with HIPAA guidelines when employing videoconferencing services similar to Skype.
“Because of concerns about compliance with encryption and confidentiality regulations, we do not use [videoconferencing] here,” Dr. Kragel says.
Additionally, “telemedicine isn’t always appropriate for patient care,” Linkous says. “All of this depends on the circumstances and needs of the patient. Obviously, surgery requires a direct physician-patient interaction, except for robotic surgery.” For hospitals that don’t have any neurology coverage, telemedicine robots can assist with outside consults for time-sensitive stroke care.
—Jonathan D. Linkous, CEO, American Telemedicine Association
Videoconferencing isn’t necessary for all telemedicine encounters, Linkous says. Teledermatology and retinal screening use “store and forward” communication of images, which allows for the electronic transmission of images and documents in non-emergent situations in which immediate video isn’t necessary.
“As a society, we’ve become more comfortable with the technology,” says Matthew Harbison, MD, medical director of Sound Physicians hospitalist services at Memorial Hermann-Texas Medical Center in Houston. “And as the technology continues to develop, ultimately there will be [more of] a role, but how large that will be is difficult to predict.” He adds that “the advantages are obviously in low-staffed places or staffing-challenged sites.”
Moving Ahead
As experts continue to iron out the kinks and as communities obtain greater access to broadband signals, telemedicine equipment is moving to advanced high-definition platforms. Meanwhile, the expense has come down considerably since its inception in the mid-1990s. A high-definition setup that once cost upward of $130,000 is now available for less than $10,000, Cattell-Gordon says.
The digital transmission also can assist in patient follow-up after discharge from the hospital and in monitoring various chronic diseases from home. It’s an effective tool for medical staff meetings and training purposes as well.
IPC's hospitalists have been using the technology to communicate with each other, brainstorming across regions of the country. “Because we’re a national company,” Dr. Weiner says, “this has changed the game in terms of being able to collaborate.”
Susan Kreimer is a freelance medical writer based in New York.
Reference
1. Thomas EJ, Lucke JF, Wueste L, Weavind L, Patel B. Association of telemedicine for remote monitoring of intensive care patients with mortality, complications, and length of stay. JAMA. 2009;302:2671-2678.
What's the Dose?
Physicians struggle every day to pick the right drug dosage for the treatment and prevention of disease. For the acute illnesses, efficacy is evident within hours or days. For the prevention of chronic disease, however, the outcome is uncertain at best. Therefore, we rely on randomized clinical trials to provide evidence that a specific drug and dosage are safe and effective.
Unfortunately, because of the limited average follow-up of 3-5 years, randomized clinical trials (RCTs) do not provide efficacy and safety information for lifetime therapy that is often advocated for the prevention of chronic disease.
For both the patient and physician, the side effects become the deciding factor. The physician usually chooses the smallest dose in order to avoid toxicity and presumably to achieve some benefit. The patient takes the drug irregularly at best.
As an example, consider the appropriate dosage for statin therapy for the prevention of atherosclerotic cardiovascular disease. Although numerous RCTs have defined the effective dose of a number of statins, recent trends in therapeutics have advocated that rather than using the dose that was used in RCTs, clinicians should increase the dose in order to reach a specific LDL cholesterol blood level.
Choosing the dosage of a drug in an RCT is a less-than-perfect exercise. Here’s how it usually goes:
Phase I trials – often based on pharmacokinetic data derived from animal studies – examine the physiological characteristics of the drug in healthy human volunteers in order to determine an effective and safe dosage prior to a phase II trial.
Phase II trials are larger; they usually examine the effect of several different dosages on a target population, and are focused not on physiological effects but on clinical outcomes and safety, in order to choose the best dosage for a phase III study. Because of their small size, these phase II studies are underpowered and prone to providing misleading dose choices.
Nevertheless, one or two doses are chosen to be used in the definitive phase III RCT, which includes enough patients to provide proof of benefit and safety of the drug based solely on its effect on mortality and morbidity.
Information is often collected in regard to the physiological effects of the drug on, for example, LDL cholesterol (in the case of statins) or heart rate (in the case of beta-blocking drugs). The proof of benefit, however, is determined by clinical outcomes, not on the physiological or "surrogate" measurements.
In the process of designing an RCT, we often make presumptions about mechanisms and will identify certain parameters that theoretically provide insight into the presumed benefit. However, many of the drugs we use have physiological effects that extend beyond the specific therapeutic target. We often remain ignorant about the mechanism by which drugs express their benefit long after their proof of benefit is demonstrated.
Statins, for instance, have a variety of pleiotropic effects. One of the most interesting is their ability to modulate inflammation, a process that is thought to be central to the progression of atherosclerotic disease. Although we presume that their effect is on LDL cholesterol, that presumption may be incorrect. Similarly, beta-blockers have well-known effects on heart rate and blood pressure, but their effect on modulating the up-regulated sympathetic nervous system in heart failure has presumed importance well beyond their effect on heart rate and blood pressure.
It is tempting to make presumptions about the effect of a drug intervention on the basis of surrogate measures like heart rate or LDL cholesterol effects, but their mechanisms of action on mortality and morbidity of disease may be unrelated to that measure.
RCTs have come a long way from relying on "surrogate" end points as the basis for making therapeutic decisions. More than 20 years ago, the CAST (Cardiac Arrhythmia Suppression Trial) was the watershed RCT that excluded the surrogate as a measure of therapeutic efficacy (J. Am. Coll. Cardiol. 1991;18:14-9). At a time when ventricular premature contraction (VPC) suppression was the "mantra" to prevent sudden death, CAST examined the pharmacologic suppression of VPCs in post–MI patients and found that, as the drugs decreased ventricular ectopy, mortality increased.
The use of the seemingly appropriate and obvious "surrogate" of LDL cholesterol lowering as a measure of therapeutic efficacy may be just as illusory. As enticing as surrogates are, the contemporary drive to lower LDL cholesterol may be as misdirected as the target to decrease the frequency of VPCs to prevent sudden death.
Like many things in life and science, things may not be what they seem.
Dr. Goldstein, the medical editor of Cardiology News, is a professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Physicians struggle every day to pick the right drug dosage for the treatment and prevention of disease. For the acute illnesses, efficacy is evident within hours or days. For the prevention of chronic disease, however, the outcome is uncertain at best. Therefore, we rely on randomized clinical trials to provide evidence that a specific drug and dosage are safe and effective.
Unfortunately, because of the limited average follow-up of 3-5 years, randomized clinical trials (RCTs) do not provide efficacy and safety information for lifetime therapy that is often advocated for the prevention of chronic disease.
For both the patient and physician, the side effects become the deciding factor. The physician usually chooses the smallest dose in order to avoid toxicity and presumably to achieve some benefit. The patient takes the drug irregularly at best.
As an example, consider the appropriate dosage for statin therapy for the prevention of atherosclerotic cardiovascular disease. Although numerous RCTs have defined the effective dose of a number of statins, recent trends in therapeutics have advocated that rather than using the dose that was used in RCTs, clinicians should increase the dose in order to reach a specific LDL cholesterol blood level.
Choosing the dosage of a drug in an RCT is a less-than-perfect exercise. Here’s how it usually goes:
Phase I trials – often based on pharmacokinetic data derived from animal studies – examine the physiological characteristics of the drug in healthy human volunteers in order to determine an effective and safe dosage prior to a phase II trial.
Phase II trials are larger; they usually examine the effect of several different dosages on a target population, and are focused not on physiological effects but on clinical outcomes and safety, in order to choose the best dosage for a phase III study. Because of their small size, these phase II studies are underpowered and prone to providing misleading dose choices.
Nevertheless, one or two doses are chosen to be used in the definitive phase III RCT, which includes enough patients to provide proof of benefit and safety of the drug based solely on its effect on mortality and morbidity.
Information is often collected in regard to the physiological effects of the drug on, for example, LDL cholesterol (in the case of statins) or heart rate (in the case of beta-blocking drugs). The proof of benefit, however, is determined by clinical outcomes, not on the physiological or "surrogate" measurements.
In the process of designing an RCT, we often make presumptions about mechanisms and will identify certain parameters that theoretically provide insight into the presumed benefit. However, many of the drugs we use have physiological effects that extend beyond the specific therapeutic target. We often remain ignorant about the mechanism by which drugs express their benefit long after their proof of benefit is demonstrated.
Statins, for instance, have a variety of pleiotropic effects. One of the most interesting is their ability to modulate inflammation, a process that is thought to be central to the progression of atherosclerotic disease. Although we presume that their effect is on LDL cholesterol, that presumption may be incorrect. Similarly, beta-blockers have well-known effects on heart rate and blood pressure, but their effect on modulating the up-regulated sympathetic nervous system in heart failure has presumed importance well beyond their effect on heart rate and blood pressure.
It is tempting to make presumptions about the effect of a drug intervention on the basis of surrogate measures like heart rate or LDL cholesterol effects, but their mechanisms of action on mortality and morbidity of disease may be unrelated to that measure.
RCTs have come a long way from relying on "surrogate" end points as the basis for making therapeutic decisions. More than 20 years ago, the CAST (Cardiac Arrhythmia Suppression Trial) was the watershed RCT that excluded the surrogate as a measure of therapeutic efficacy (J. Am. Coll. Cardiol. 1991;18:14-9). At a time when ventricular premature contraction (VPC) suppression was the "mantra" to prevent sudden death, CAST examined the pharmacologic suppression of VPCs in post–MI patients and found that, as the drugs decreased ventricular ectopy, mortality increased.
The use of the seemingly appropriate and obvious "surrogate" of LDL cholesterol lowering as a measure of therapeutic efficacy may be just as illusory. As enticing as surrogates are, the contemporary drive to lower LDL cholesterol may be as misdirected as the target to decrease the frequency of VPCs to prevent sudden death.
Like many things in life and science, things may not be what they seem.
Dr. Goldstein, the medical editor of Cardiology News, is a professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Physicians struggle every day to pick the right drug dosage for the treatment and prevention of disease. For the acute illnesses, efficacy is evident within hours or days. For the prevention of chronic disease, however, the outcome is uncertain at best. Therefore, we rely on randomized clinical trials to provide evidence that a specific drug and dosage are safe and effective.
Unfortunately, because of the limited average follow-up of 3-5 years, randomized clinical trials (RCTs) do not provide efficacy and safety information for lifetime therapy that is often advocated for the prevention of chronic disease.
For both the patient and physician, the side effects become the deciding factor. The physician usually chooses the smallest dose in order to avoid toxicity and presumably to achieve some benefit. The patient takes the drug irregularly at best.
As an example, consider the appropriate dosage for statin therapy for the prevention of atherosclerotic cardiovascular disease. Although numerous RCTs have defined the effective dose of a number of statins, recent trends in therapeutics have advocated that rather than using the dose that was used in RCTs, clinicians should increase the dose in order to reach a specific LDL cholesterol blood level.
Choosing the dosage of a drug in an RCT is a less-than-perfect exercise. Here’s how it usually goes:
Phase I trials – often based on pharmacokinetic data derived from animal studies – examine the physiological characteristics of the drug in healthy human volunteers in order to determine an effective and safe dosage prior to a phase II trial.
Phase II trials are larger; they usually examine the effect of several different dosages on a target population, and are focused not on physiological effects but on clinical outcomes and safety, in order to choose the best dosage for a phase III study. Because of their small size, these phase II studies are underpowered and prone to providing misleading dose choices.
Nevertheless, one or two doses are chosen to be used in the definitive phase III RCT, which includes enough patients to provide proof of benefit and safety of the drug based solely on its effect on mortality and morbidity.
Information is often collected in regard to the physiological effects of the drug on, for example, LDL cholesterol (in the case of statins) or heart rate (in the case of beta-blocking drugs). The proof of benefit, however, is determined by clinical outcomes, not on the physiological or "surrogate" measurements.
In the process of designing an RCT, we often make presumptions about mechanisms and will identify certain parameters that theoretically provide insight into the presumed benefit. However, many of the drugs we use have physiological effects that extend beyond the specific therapeutic target. We often remain ignorant about the mechanism by which drugs express their benefit long after their proof of benefit is demonstrated.
Statins, for instance, have a variety of pleiotropic effects. One of the most interesting is their ability to modulate inflammation, a process that is thought to be central to the progression of atherosclerotic disease. Although we presume that their effect is on LDL cholesterol, that presumption may be incorrect. Similarly, beta-blockers have well-known effects on heart rate and blood pressure, but their effect on modulating the up-regulated sympathetic nervous system in heart failure has presumed importance well beyond their effect on heart rate and blood pressure.
It is tempting to make presumptions about the effect of a drug intervention on the basis of surrogate measures like heart rate or LDL cholesterol effects, but their mechanisms of action on mortality and morbidity of disease may be unrelated to that measure.
RCTs have come a long way from relying on "surrogate" end points as the basis for making therapeutic decisions. More than 20 years ago, the CAST (Cardiac Arrhythmia Suppression Trial) was the watershed RCT that excluded the surrogate as a measure of therapeutic efficacy (J. Am. Coll. Cardiol. 1991;18:14-9). At a time when ventricular premature contraction (VPC) suppression was the "mantra" to prevent sudden death, CAST examined the pharmacologic suppression of VPCs in post–MI patients and found that, as the drugs decreased ventricular ectopy, mortality increased.
The use of the seemingly appropriate and obvious "surrogate" of LDL cholesterol lowering as a measure of therapeutic efficacy may be just as illusory. As enticing as surrogates are, the contemporary drive to lower LDL cholesterol may be as misdirected as the target to decrease the frequency of VPCs to prevent sudden death.
Like many things in life and science, things may not be what they seem.
Dr. Goldstein, the medical editor of Cardiology News, is a professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Early Data Find No Adalimumab Teratogenicity
BALTIMORE – Exposure to adalimumab was not associated with any specific pattern of minor or major birth defects in women with rheumatoid arthritis taking the biologic drug during pregnancy, according to preliminary data from an ongoing prospective cohort study.
Between November 2004 and January 2012, 312 pregnant women in the United States and Canada – 69 women with RA exposed to adalimumab, 80 women with RA who had not taken adalimumab, and 163 healthy controls – were enrolled before 20 weeks’ gestation. Their mean age was 32-33 years, and about two-thirds were white.
Major birth defects among the live births were identified in 5% of the babies born to women exposed to adalimumab, compared with about 4% among disease-matched controls who did not take adalimumab, and about 7% among healthy controls, Christina Chambers, Ph.D., of the University of California, San Diego, reported at the annual meeting of the Teratology Society.
The rate of minor structural abnormalities was similar in the three groups, at about 22%-24%, and there was no pattern of major or minor structural defects noted among the adalimumab-exposed group. (The three major malformations in the adalimumab-exposed group were one ventricular septal defect, one unilateral cryptorchidism, and one case of microcephaly.)
There were no stillbirths. The rate of spontaneous abortions was not significantly different between the three groups, nor were the rates of preterm delivery or birth weights, said Dr. Chambers, director of the California Teratogen Information Service and Clinical Research Program.
Through 1-year of follow-up, there were no malignancies among the infants and the rates of serious infections in the three groups were similar (about 3% in the two RA groups and 2% in the healthy comparison group).
The teratogenic effects of adalimumab, a tumor necrosis factor blocker, are being evaluated in the pregnancy registry, which is part of the Organization of Teratology Information Specialists (OTIS) Autoimmune Diseases in Pregnancy Project.
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved in the United States in 2002 as a treatment for people with moderately to severely active RA, and has since been approved for other autoimmune diseases, including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and psoriasis.
The registry study is comparing outcomes in women with RA who are treated with adalimumab during pregnancy, in women with RA not treated with adalimumab during pregnancy, and in women who do not have an autoimmune disease and have not been exposed to adalimumab or any known teratogenic drug during pregnancy. The study includes medical record reviews, examination of infants for major and minor structural abnormalities, and follow-up for 1 year post partum. It is expected to continue through 2017; the pregnant women are recruited from OTIS member services and from rheumatologists, and other clinicians who care for these patients.
Although little to no placental transfer of adalimumab is expected during early pregnancy, limited information on the safety of adalimumab during pregnancy has been published, Dr. Chambers said.
Abbott Laboratories is among the sponsors of the OTIS Autoimmune Diseases in Pregnancy Project, which is also evaluating safety of medications in women with ankylosing spondylitis, psoriasis and psoriatic arthritis, and Crohn’s disease. Dr. Chambers and her coauthors have received or receive grant funding for research on medications for autoimmune diseases from Abbott and other manufacturers: Amgen, Bristol Myers Squibb, Roche Genentech, Sanofi, Teva, Par, Sandoz, and Apotex.
Information for women and clinicians interested in enrolling in the OTIS Autoimmune Diseases in Pregnancy Project is available at www.otispregnancy.org/autoimmune-studies-s13049.
BALTIMORE – Exposure to adalimumab was not associated with any specific pattern of minor or major birth defects in women with rheumatoid arthritis taking the biologic drug during pregnancy, according to preliminary data from an ongoing prospective cohort study.
Between November 2004 and January 2012, 312 pregnant women in the United States and Canada – 69 women with RA exposed to adalimumab, 80 women with RA who had not taken adalimumab, and 163 healthy controls – were enrolled before 20 weeks’ gestation. Their mean age was 32-33 years, and about two-thirds were white.
Major birth defects among the live births were identified in 5% of the babies born to women exposed to adalimumab, compared with about 4% among disease-matched controls who did not take adalimumab, and about 7% among healthy controls, Christina Chambers, Ph.D., of the University of California, San Diego, reported at the annual meeting of the Teratology Society.
The rate of minor structural abnormalities was similar in the three groups, at about 22%-24%, and there was no pattern of major or minor structural defects noted among the adalimumab-exposed group. (The three major malformations in the adalimumab-exposed group were one ventricular septal defect, one unilateral cryptorchidism, and one case of microcephaly.)
There were no stillbirths. The rate of spontaneous abortions was not significantly different between the three groups, nor were the rates of preterm delivery or birth weights, said Dr. Chambers, director of the California Teratogen Information Service and Clinical Research Program.
Through 1-year of follow-up, there were no malignancies among the infants and the rates of serious infections in the three groups were similar (about 3% in the two RA groups and 2% in the healthy comparison group).
The teratogenic effects of adalimumab, a tumor necrosis factor blocker, are being evaluated in the pregnancy registry, which is part of the Organization of Teratology Information Specialists (OTIS) Autoimmune Diseases in Pregnancy Project.
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved in the United States in 2002 as a treatment for people with moderately to severely active RA, and has since been approved for other autoimmune diseases, including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and psoriasis.
The registry study is comparing outcomes in women with RA who are treated with adalimumab during pregnancy, in women with RA not treated with adalimumab during pregnancy, and in women who do not have an autoimmune disease and have not been exposed to adalimumab or any known teratogenic drug during pregnancy. The study includes medical record reviews, examination of infants for major and minor structural abnormalities, and follow-up for 1 year post partum. It is expected to continue through 2017; the pregnant women are recruited from OTIS member services and from rheumatologists, and other clinicians who care for these patients.
Although little to no placental transfer of adalimumab is expected during early pregnancy, limited information on the safety of adalimumab during pregnancy has been published, Dr. Chambers said.
Abbott Laboratories is among the sponsors of the OTIS Autoimmune Diseases in Pregnancy Project, which is also evaluating safety of medications in women with ankylosing spondylitis, psoriasis and psoriatic arthritis, and Crohn’s disease. Dr. Chambers and her coauthors have received or receive grant funding for research on medications for autoimmune diseases from Abbott and other manufacturers: Amgen, Bristol Myers Squibb, Roche Genentech, Sanofi, Teva, Par, Sandoz, and Apotex.
Information for women and clinicians interested in enrolling in the OTIS Autoimmune Diseases in Pregnancy Project is available at www.otispregnancy.org/autoimmune-studies-s13049.
BALTIMORE – Exposure to adalimumab was not associated with any specific pattern of minor or major birth defects in women with rheumatoid arthritis taking the biologic drug during pregnancy, according to preliminary data from an ongoing prospective cohort study.
Between November 2004 and January 2012, 312 pregnant women in the United States and Canada – 69 women with RA exposed to adalimumab, 80 women with RA who had not taken adalimumab, and 163 healthy controls – were enrolled before 20 weeks’ gestation. Their mean age was 32-33 years, and about two-thirds were white.
Major birth defects among the live births were identified in 5% of the babies born to women exposed to adalimumab, compared with about 4% among disease-matched controls who did not take adalimumab, and about 7% among healthy controls, Christina Chambers, Ph.D., of the University of California, San Diego, reported at the annual meeting of the Teratology Society.
The rate of minor structural abnormalities was similar in the three groups, at about 22%-24%, and there was no pattern of major or minor structural defects noted among the adalimumab-exposed group. (The three major malformations in the adalimumab-exposed group were one ventricular septal defect, one unilateral cryptorchidism, and one case of microcephaly.)
There were no stillbirths. The rate of spontaneous abortions was not significantly different between the three groups, nor were the rates of preterm delivery or birth weights, said Dr. Chambers, director of the California Teratogen Information Service and Clinical Research Program.
Through 1-year of follow-up, there were no malignancies among the infants and the rates of serious infections in the three groups were similar (about 3% in the two RA groups and 2% in the healthy comparison group).
The teratogenic effects of adalimumab, a tumor necrosis factor blocker, are being evaluated in the pregnancy registry, which is part of the Organization of Teratology Information Specialists (OTIS) Autoimmune Diseases in Pregnancy Project.
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved in the United States in 2002 as a treatment for people with moderately to severely active RA, and has since been approved for other autoimmune diseases, including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and psoriasis.
The registry study is comparing outcomes in women with RA who are treated with adalimumab during pregnancy, in women with RA not treated with adalimumab during pregnancy, and in women who do not have an autoimmune disease and have not been exposed to adalimumab or any known teratogenic drug during pregnancy. The study includes medical record reviews, examination of infants for major and minor structural abnormalities, and follow-up for 1 year post partum. It is expected to continue through 2017; the pregnant women are recruited from OTIS member services and from rheumatologists, and other clinicians who care for these patients.
Although little to no placental transfer of adalimumab is expected during early pregnancy, limited information on the safety of adalimumab during pregnancy has been published, Dr. Chambers said.
Abbott Laboratories is among the sponsors of the OTIS Autoimmune Diseases in Pregnancy Project, which is also evaluating safety of medications in women with ankylosing spondylitis, psoriasis and psoriatic arthritis, and Crohn’s disease. Dr. Chambers and her coauthors have received or receive grant funding for research on medications for autoimmune diseases from Abbott and other manufacturers: Amgen, Bristol Myers Squibb, Roche Genentech, Sanofi, Teva, Par, Sandoz, and Apotex.
Information for women and clinicians interested in enrolling in the OTIS Autoimmune Diseases in Pregnancy Project is available at www.otispregnancy.org/autoimmune-studies-s13049.
AT THE ANNUAL MEETING OF THE TERATOLOGY SOCIETY