User login
New PET tracer detects more metastases in cancer patients
leading to predictions of a “paradigm shift” in this field.
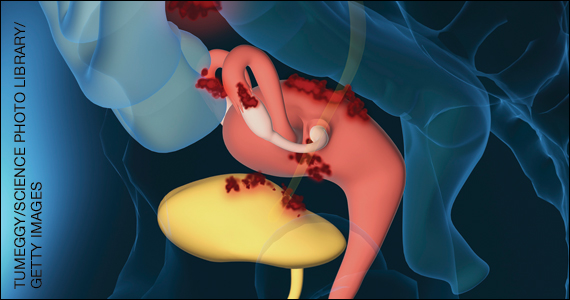
The new tracer, 68Ga-FAPI (fibroblast activation protein inhibitor), detected more metastases in patients with lung cancer than the standard tracer, 18F-FDG (fluorodeoxyglucose), which has been in use for years.
The study by Chinese researchers was published in Radiology.
The team imaged 34 lung cancer patients with both 68Ga-FAPI and 18F-FDG. Performance was similar for primary tumors and for lung, liver, and adrenal gland metastases. However, FAPI imaging detected more metastases in the lymph nodes (356 vs. 320), brain (23 vs. 10), bone (109 vs. 91), and pleura (66 vs. 35). However, neither modality outperformed MRI for brain metastases, the researchers note.
An accompanying editorial concluded that 68Ga-FAPI PET/CT scanning marks “an important paradigm shift to more specific identification and characterization of a variety of cancers.”
“This may also mark the arrival of a new era in nuclear medicine where molecular imaging helps visualize and characterize the entire tumor burden in one setting,” write editorialists Francine Jacobson, MD, and Annick Van den Abbeele, MD, from Harvard University and Brigham and Women’s Hospital and the Dana Farber Cancer Center, in Boston.
This study was the one of the latest in a fast-growing body of literature reporting that tracers targeting FAP with a small-molecule inhibitor (FAPI) outperform FDG tracers, not just in lung cancer but across a broad range of cancers, including breast, hepatic, gastrointestinal, head-neck, gynecologic, and many other tumor types.
The possibilities aren’t limited to imaging, either. Several companies are planning trials to target FAP with radiopharmaceuticals.
FAP is associated with wound repair and is highly expressed by the fibroblasts tightly packed in with cancer cells, particularly in stroma-dense tumors. FAP is rarely expressed by healthy tissue.
The underlying idea is to deliver a radionuclide to cancer-associated fibroblasts, using either a positron emitter, such as gallium-68 (68Ga), for PET imaging or a beta particle or other short-radiation emitter to kill nearby cancer cells as part of treatment.
Targeting FAP holds the promise of PET imaging that is more selective for cancer than FDG. FDG resolution depends on glucose uptake, which is high in active tumors but is also high in inflamed tissues as well as in the brain, gastrointestinal tract, and other areas. Uptake by background tissue can make it difficult to distinguish tumors from their surroundings. FDG uptake can also be lower in small and indolent tumors.
On the therapy side, there’s hope that FAP targeting will lead to radiopharmaceuticals that work across tumor types, not just in specific cancers.
High interest in FAP
Overall, FAP “is a target of high interest for the whole medical oncology community. The preliminary data are good, but this will take a while” to get to market, said Jeremie Calais, MD, a nuclear medicine specialist and FAP researcher at the University of California, Los Angeles.
Interest in FAP as a radiopharmaceutical target is being driven by the success of two agents that have served as a kind of proof of concept, Dr. Calais said.
The first is Novartis’s 177Lu-PSMA-617, which was granted priority review by the U.S. Food and Drug Administration in September 2021 following phase 3 results that showed a progression-free survival benefit of about 5 months when added to standard of care for metastatic castration-resistant prostate cancer, as well as an overall survival benefit of 4 months.
PSMA-617 binds prostate cancer cells that express prostate-specific membrane antigen. The lutetium-177 (177Lu) bombards them with beta particles and gamma radiation.
FAP researchers are also encouraged by the success of 177Lu dotatate (Lutathera), from Advanced Accelerator Applications, which delivers the radionucleotide to gastroenteropancreatic neuroendocrine tumors that express somatostatin receptors.
The FDA approved this agent in 2018 in part on the basis of phase 3 results that found a 20-month progression-free survival of 65.2% when Lutathera was added to octreotide for metastatic disease vs. 10.8% when it wasn’t.
Novartis is now looking into developing FAP-targeted radiopharmaceuticals, along with Clovis and Point Biopharma, among others.
“That’s the key goal” of industry research, “more so than FAP as a diagnostic tool,” Dr. Calais commented to this news organization. There’s “huge potential” if it works out, he said, in part because it won’t be limited to one tumor type.
Clovis recently launched a phase 1/2 trial of its candidate, 177Lu-FAP-2286, for advanced/metastatic solid tumors.
In the company’s “luMIERE” trial, subjects will be infused with 68Ga-FAP-2286 to image the tumor. Once uptake is confirmed, they’ll be infused with 177Lu-FAP-2286 for treatment.
The two-step process – uptake confirmation, then treatment – is dubbed “theranostics” and is the standard approach for radiopharmaceutical therapy, Dr. Calais said.
His own team is working to confirm that imaging accurately reflects FAP expression in tumors by comparing preoperative imaging results with FAP expression on surgical specimens. So far, his team has found that they are strongly correlated.
FAPI PET imaging research is much farther along than therapeutic applications, with almost 200 research articles listed on PubMed in 2021, up from just 3 in 2018. One 2019 paper reported “remarkably high uptake and image contrast” across 28 cancers in 80 patients, including breast, esophagus, lung, pancreatic, head-neck, and colorectal tumors.
Imaging studies so far have tended to be small, with many currently focused on identifying the optimal molecule for targeting FAP and the best positron emitter to combine with it.
FAPI tracers are not available yet commercially, so researchers are creating them themselves. One team recently reported it’s recipe for automated synthesis using commercially available synthesis modules.
Sofie, a maker of FDG and other tracers, hopes to change that and is working to bring FAP tracers to market. The company announced in November 2021 a phase 2 study of 68Ga FAPI-46 to image pancreatic ductal adenocarcinoma. It’s the first step in a broader development program for oncologic and nononcologic indications, Sofie said in a press release.
Dr. Calais sees potential for indications where FAPI has already outperformed FDG in the literature, particularly for gastrointestinal cancers. He doesn’t think it will ever replace FDG for indications such as lymphoma, where it “works perfectly well.”
“On the other hand, you have lesions located in a tissue that has some background level” of FDG uptake. “These things are okay with FDG, but I think maybe FAP can help” because of the improved signal-to-noise ratio, Dr. Calais commented. Unlike FDG, “you mostly never see background uptake with FAP-imaging agents,” he said.
Other pluses include quicker distribution throughout the body than FDG, so scan times are shorter, and also patients do not need to fast beforehand.
Dr. Calais predicts that FAPI tracers will reach the market within 5 years.
A version of this article first appeared on Medscape.com.
leading to predictions of a “paradigm shift” in this field.
The new tracer, 68Ga-FAPI (fibroblast activation protein inhibitor), detected more metastases in patients with lung cancer than the standard tracer, 18F-FDG (fluorodeoxyglucose), which has been in use for years.
The study by Chinese researchers was published in Radiology.
The team imaged 34 lung cancer patients with both 68Ga-FAPI and 18F-FDG. Performance was similar for primary tumors and for lung, liver, and adrenal gland metastases. However, FAPI imaging detected more metastases in the lymph nodes (356 vs. 320), brain (23 vs. 10), bone (109 vs. 91), and pleura (66 vs. 35). However, neither modality outperformed MRI for brain metastases, the researchers note.
An accompanying editorial concluded that 68Ga-FAPI PET/CT scanning marks “an important paradigm shift to more specific identification and characterization of a variety of cancers.”
“This may also mark the arrival of a new era in nuclear medicine where molecular imaging helps visualize and characterize the entire tumor burden in one setting,” write editorialists Francine Jacobson, MD, and Annick Van den Abbeele, MD, from Harvard University and Brigham and Women’s Hospital and the Dana Farber Cancer Center, in Boston.
This study was the one of the latest in a fast-growing body of literature reporting that tracers targeting FAP with a small-molecule inhibitor (FAPI) outperform FDG tracers, not just in lung cancer but across a broad range of cancers, including breast, hepatic, gastrointestinal, head-neck, gynecologic, and many other tumor types.
The possibilities aren’t limited to imaging, either. Several companies are planning trials to target FAP with radiopharmaceuticals.
FAP is associated with wound repair and is highly expressed by the fibroblasts tightly packed in with cancer cells, particularly in stroma-dense tumors. FAP is rarely expressed by healthy tissue.
The underlying idea is to deliver a radionuclide to cancer-associated fibroblasts, using either a positron emitter, such as gallium-68 (68Ga), for PET imaging or a beta particle or other short-radiation emitter to kill nearby cancer cells as part of treatment.
Targeting FAP holds the promise of PET imaging that is more selective for cancer than FDG. FDG resolution depends on glucose uptake, which is high in active tumors but is also high in inflamed tissues as well as in the brain, gastrointestinal tract, and other areas. Uptake by background tissue can make it difficult to distinguish tumors from their surroundings. FDG uptake can also be lower in small and indolent tumors.
On the therapy side, there’s hope that FAP targeting will lead to radiopharmaceuticals that work across tumor types, not just in specific cancers.
High interest in FAP
Overall, FAP “is a target of high interest for the whole medical oncology community. The preliminary data are good, but this will take a while” to get to market, said Jeremie Calais, MD, a nuclear medicine specialist and FAP researcher at the University of California, Los Angeles.
Interest in FAP as a radiopharmaceutical target is being driven by the success of two agents that have served as a kind of proof of concept, Dr. Calais said.
The first is Novartis’s 177Lu-PSMA-617, which was granted priority review by the U.S. Food and Drug Administration in September 2021 following phase 3 results that showed a progression-free survival benefit of about 5 months when added to standard of care for metastatic castration-resistant prostate cancer, as well as an overall survival benefit of 4 months.
PSMA-617 binds prostate cancer cells that express prostate-specific membrane antigen. The lutetium-177 (177Lu) bombards them with beta particles and gamma radiation.
FAP researchers are also encouraged by the success of 177Lu dotatate (Lutathera), from Advanced Accelerator Applications, which delivers the radionucleotide to gastroenteropancreatic neuroendocrine tumors that express somatostatin receptors.
The FDA approved this agent in 2018 in part on the basis of phase 3 results that found a 20-month progression-free survival of 65.2% when Lutathera was added to octreotide for metastatic disease vs. 10.8% when it wasn’t.
Novartis is now looking into developing FAP-targeted radiopharmaceuticals, along with Clovis and Point Biopharma, among others.
“That’s the key goal” of industry research, “more so than FAP as a diagnostic tool,” Dr. Calais commented to this news organization. There’s “huge potential” if it works out, he said, in part because it won’t be limited to one tumor type.
Clovis recently launched a phase 1/2 trial of its candidate, 177Lu-FAP-2286, for advanced/metastatic solid tumors.
In the company’s “luMIERE” trial, subjects will be infused with 68Ga-FAP-2286 to image the tumor. Once uptake is confirmed, they’ll be infused with 177Lu-FAP-2286 for treatment.
The two-step process – uptake confirmation, then treatment – is dubbed “theranostics” and is the standard approach for radiopharmaceutical therapy, Dr. Calais said.
His own team is working to confirm that imaging accurately reflects FAP expression in tumors by comparing preoperative imaging results with FAP expression on surgical specimens. So far, his team has found that they are strongly correlated.
FAPI PET imaging research is much farther along than therapeutic applications, with almost 200 research articles listed on PubMed in 2021, up from just 3 in 2018. One 2019 paper reported “remarkably high uptake and image contrast” across 28 cancers in 80 patients, including breast, esophagus, lung, pancreatic, head-neck, and colorectal tumors.
Imaging studies so far have tended to be small, with many currently focused on identifying the optimal molecule for targeting FAP and the best positron emitter to combine with it.
FAPI tracers are not available yet commercially, so researchers are creating them themselves. One team recently reported it’s recipe for automated synthesis using commercially available synthesis modules.
Sofie, a maker of FDG and other tracers, hopes to change that and is working to bring FAP tracers to market. The company announced in November 2021 a phase 2 study of 68Ga FAPI-46 to image pancreatic ductal adenocarcinoma. It’s the first step in a broader development program for oncologic and nononcologic indications, Sofie said in a press release.
Dr. Calais sees potential for indications where FAPI has already outperformed FDG in the literature, particularly for gastrointestinal cancers. He doesn’t think it will ever replace FDG for indications such as lymphoma, where it “works perfectly well.”
“On the other hand, you have lesions located in a tissue that has some background level” of FDG uptake. “These things are okay with FDG, but I think maybe FAP can help” because of the improved signal-to-noise ratio, Dr. Calais commented. Unlike FDG, “you mostly never see background uptake with FAP-imaging agents,” he said.
Other pluses include quicker distribution throughout the body than FDG, so scan times are shorter, and also patients do not need to fast beforehand.
Dr. Calais predicts that FAPI tracers will reach the market within 5 years.
A version of this article first appeared on Medscape.com.
leading to predictions of a “paradigm shift” in this field.
The new tracer, 68Ga-FAPI (fibroblast activation protein inhibitor), detected more metastases in patients with lung cancer than the standard tracer, 18F-FDG (fluorodeoxyglucose), which has been in use for years.
The study by Chinese researchers was published in Radiology.
The team imaged 34 lung cancer patients with both 68Ga-FAPI and 18F-FDG. Performance was similar for primary tumors and for lung, liver, and adrenal gland metastases. However, FAPI imaging detected more metastases in the lymph nodes (356 vs. 320), brain (23 vs. 10), bone (109 vs. 91), and pleura (66 vs. 35). However, neither modality outperformed MRI for brain metastases, the researchers note.
An accompanying editorial concluded that 68Ga-FAPI PET/CT scanning marks “an important paradigm shift to more specific identification and characterization of a variety of cancers.”
“This may also mark the arrival of a new era in nuclear medicine where molecular imaging helps visualize and characterize the entire tumor burden in one setting,” write editorialists Francine Jacobson, MD, and Annick Van den Abbeele, MD, from Harvard University and Brigham and Women’s Hospital and the Dana Farber Cancer Center, in Boston.
This study was the one of the latest in a fast-growing body of literature reporting that tracers targeting FAP with a small-molecule inhibitor (FAPI) outperform FDG tracers, not just in lung cancer but across a broad range of cancers, including breast, hepatic, gastrointestinal, head-neck, gynecologic, and many other tumor types.
The possibilities aren’t limited to imaging, either. Several companies are planning trials to target FAP with radiopharmaceuticals.
FAP is associated with wound repair and is highly expressed by the fibroblasts tightly packed in with cancer cells, particularly in stroma-dense tumors. FAP is rarely expressed by healthy tissue.
The underlying idea is to deliver a radionuclide to cancer-associated fibroblasts, using either a positron emitter, such as gallium-68 (68Ga), for PET imaging or a beta particle or other short-radiation emitter to kill nearby cancer cells as part of treatment.
Targeting FAP holds the promise of PET imaging that is more selective for cancer than FDG. FDG resolution depends on glucose uptake, which is high in active tumors but is also high in inflamed tissues as well as in the brain, gastrointestinal tract, and other areas. Uptake by background tissue can make it difficult to distinguish tumors from their surroundings. FDG uptake can also be lower in small and indolent tumors.
On the therapy side, there’s hope that FAP targeting will lead to radiopharmaceuticals that work across tumor types, not just in specific cancers.
High interest in FAP
Overall, FAP “is a target of high interest for the whole medical oncology community. The preliminary data are good, but this will take a while” to get to market, said Jeremie Calais, MD, a nuclear medicine specialist and FAP researcher at the University of California, Los Angeles.
Interest in FAP as a radiopharmaceutical target is being driven by the success of two agents that have served as a kind of proof of concept, Dr. Calais said.
The first is Novartis’s 177Lu-PSMA-617, which was granted priority review by the U.S. Food and Drug Administration in September 2021 following phase 3 results that showed a progression-free survival benefit of about 5 months when added to standard of care for metastatic castration-resistant prostate cancer, as well as an overall survival benefit of 4 months.
PSMA-617 binds prostate cancer cells that express prostate-specific membrane antigen. The lutetium-177 (177Lu) bombards them with beta particles and gamma radiation.
FAP researchers are also encouraged by the success of 177Lu dotatate (Lutathera), from Advanced Accelerator Applications, which delivers the radionucleotide to gastroenteropancreatic neuroendocrine tumors that express somatostatin receptors.
The FDA approved this agent in 2018 in part on the basis of phase 3 results that found a 20-month progression-free survival of 65.2% when Lutathera was added to octreotide for metastatic disease vs. 10.8% when it wasn’t.
Novartis is now looking into developing FAP-targeted radiopharmaceuticals, along with Clovis and Point Biopharma, among others.
“That’s the key goal” of industry research, “more so than FAP as a diagnostic tool,” Dr. Calais commented to this news organization. There’s “huge potential” if it works out, he said, in part because it won’t be limited to one tumor type.
Clovis recently launched a phase 1/2 trial of its candidate, 177Lu-FAP-2286, for advanced/metastatic solid tumors.
In the company’s “luMIERE” trial, subjects will be infused with 68Ga-FAP-2286 to image the tumor. Once uptake is confirmed, they’ll be infused with 177Lu-FAP-2286 for treatment.
The two-step process – uptake confirmation, then treatment – is dubbed “theranostics” and is the standard approach for radiopharmaceutical therapy, Dr. Calais said.
His own team is working to confirm that imaging accurately reflects FAP expression in tumors by comparing preoperative imaging results with FAP expression on surgical specimens. So far, his team has found that they are strongly correlated.
FAPI PET imaging research is much farther along than therapeutic applications, with almost 200 research articles listed on PubMed in 2021, up from just 3 in 2018. One 2019 paper reported “remarkably high uptake and image contrast” across 28 cancers in 80 patients, including breast, esophagus, lung, pancreatic, head-neck, and colorectal tumors.
Imaging studies so far have tended to be small, with many currently focused on identifying the optimal molecule for targeting FAP and the best positron emitter to combine with it.
FAPI tracers are not available yet commercially, so researchers are creating them themselves. One team recently reported it’s recipe for automated synthesis using commercially available synthesis modules.
Sofie, a maker of FDG and other tracers, hopes to change that and is working to bring FAP tracers to market. The company announced in November 2021 a phase 2 study of 68Ga FAPI-46 to image pancreatic ductal adenocarcinoma. It’s the first step in a broader development program for oncologic and nononcologic indications, Sofie said in a press release.
Dr. Calais sees potential for indications where FAPI has already outperformed FDG in the literature, particularly for gastrointestinal cancers. He doesn’t think it will ever replace FDG for indications such as lymphoma, where it “works perfectly well.”
“On the other hand, you have lesions located in a tissue that has some background level” of FDG uptake. “These things are okay with FDG, but I think maybe FAP can help” because of the improved signal-to-noise ratio, Dr. Calais commented. Unlike FDG, “you mostly never see background uptake with FAP-imaging agents,” he said.
Other pluses include quicker distribution throughout the body than FDG, so scan times are shorter, and also patients do not need to fast beforehand.
Dr. Calais predicts that FAPI tracers will reach the market within 5 years.
A version of this article first appeared on Medscape.com.
FROM RADIOLOGY
LGBTQ parents fare worse giving birth
Members of the LGBTQ community who give birth appear to have a greater risk of hypertensive disorders of pregnancy and postpartum hemorrhage, according to new research presented at the annual meeting sponsored by the Society for Maternal-Fetal Medicine.
“Our study found that birthing patients in likely sexual and gender minority partnerships experienced disparities in clinical outcomes,” Stephanie Leonard, PhD, an epidemiology and biostatistics instructor at the Stanford (Calif.) University division of maternal-fetal medicine and obstetrics, told attendees at the meeting. The disparities are likely because of various social determinants and possibly higher use of assisted reproductive technology (ART). The findings establish “how these are significant disparities that have been largely overlooked and set the groundwork for doing further research on maybe ways that we can improve the inclusivity of obstetric care.”
Jenny Mei, MD, a maternal-fetal medicine fellow at the University of California, Los Angeles, who attended the presentation but was not involved in the research, said the findings were “overall unfortunate but not surprising given the existing studies looking at LGBTQ patients and their poorer health outcomes, largely due to lack of access to health care and discrimination in the health care setting.”
Dr. Leonard described the societal, interpersonal, and individual factors that can contribute to health disparities among gender and sexual minority patients.
“At the societal level, there are expectations of what it means to be pregnant, to give birth, and to be a parent. At the community level, there’s the clinical care environment, and at the interpersonal level, there’s an obstetrician’s relationship with the patient,” Dr. Leonard said. “At the individual level, most notably is minority stress, the biological effects of the chronic experience of discrimination.”
It has historically been difficult to collect data on this patient population, but a change in the design of the California birth certificate made it possible to gather more data than previously possible. The updated California birth certificate, issued in 2016, allows the parent not giving birth to check off whether they are the child’s mother, father, parent, or “not specified” instead of defaulting to “father.” In addition, the parent giving birth can select mother, father, parent or not specified instead of being “mother” by default.
The researchers classified sexual and gender minority (SGM) partnerships as those in which the parent giving birth was identified as the father and those where both parents were identified as mothers. Non-SGM minority partnerships were those in which the birthing parent was identified as the mother and the nonbirthing parent was identified as the father.
The population-based cohort study included data from all live birth hospitalizations from 2016-2019 in California, whose annual births represent one in eight babies born each year in the United States. The population of SGM patients different significantly from the non-SGM population in nearly every demographic and clinical factor except rates of pre-existing diabetes. For example, 42% of the SGM birthing patients were age 35 or older, compared with 23% of the non-SGM patients.
SGM patients were more likely to be born in the United States, were more likely to be White, and were less likely to be Asian or Hispanic. SGM patients had higher education levels and were more likely to have private insurance. They were also more likely to be nulliparous and have chronic hypertension. Average body mass index for SGM patients was 33 kg/m2, compared with 30 for non-SGM patients. SGM patients were also much more likely to have multifetal gestation: 7.1% of SGM patients versus 1.5% of non-SGM patients.
In terms of clinical outcomes, 14% of SGM patients had hypertensive disorders of pregnancy, compared with 8% of non-SGM patients. Before adjustment for potential confounders, SGM patients were also twice as likely to have postpartum hemorrhage (8% vs. 4% in non-SGM patients) and postterm birth at 42-44 weeks (0.6% vs 0.3% in non-SGM patients).
“Having increased postterm birth is a matter of declining induction of labor, as it is recommended to have an induction by 41 weeks of gestation in general,” Dr. Mei said in an interview. “It is also possible this patient cohort faces more barriers in access to care and possible discrimination as sexual/gender minority patients.”
Rates of severe preeclampsia, induction of labor, cesarean delivery, preterm birth, low birth weight, and a low Apgar score were also higher among SGM patients, but these associations were no longer significant after adjustment for age, education, payment method, parity, prepregnancy weight, comorbidities, and multifetal gestation. The difference in hypertensive disorders of pregnancy, postpartum hemorrhage, and postterm birth remained statistically significant after adjustment.
Past research has shown that only about a third of cisgender female same-sex marriages used ART, so the disparities cannot be completely explained by ART use, Dr. Leonard said.
“I think the main drivers are structural disparities,” Dr. Leonard said. “Every obstetric clinic is focused in a way that’s about mother-father, and many people who don’t feel like they fit into that paradigm feel excluded and disengage with health care.”
Elliott Main, MD, a clinical professor of obstetrics and gynecology at Stanford University and coauthor of the study noted that discrimination and stigma likely play a substantial role in the disparities.
“Sexual and/or gender minority people face this discrimination at structural and interpersonal levels on a regular basis, which can lead to chronic stress and its harmful physical effects as well as lower-quality health care,” Dr. Main said in an interview.
Another coauthor, Juno Obedin-Maliver, MD, an assistant professor of obstetrics and gynecology at Stanford, emphasized how much room for improvement exists in care for SGM obstetric patients.
“We hope that this study brings needed attention to the disparities in perinatal health experienced by sexual and/or gender minority people,” Dr. Obedin-Maliver said. “There is much we can do to better understand the family building goals of sexual and/or gender minority people and help those to be achieved with healthy outcomes for parents and their children.”
One limitation of the study is that it’s possible to misclassify individuals using the birth certificate data, and not everyone may be comfortable selecting the box that accurately represents their identity, particularly if they aren’t “out” or fear discrimination or stigma, so the population may underrepresent the actual numbers of sexual and gender minority individuals giving birth. Dr. Mei added that it would be helpful to see data on neonatal ICU admissions and use of ART.
It’s difficult to say how generalizable the findings are, Dr. Mei said. “It is possible the findings would be more exaggerated in the rest of the country outside of California, if we assume there is potentially lower health access and more stigma.” The fact that California offers different gender options for the birthing and nonbirthing parent is, by itself, an indication of a potentially more accepting social environment than might be found in other states.
”The take-home message is that this patient population is higher risk, likely partially due to baseline increased risk factors, such as older maternal age and likely use of ART, and partially due to possible lack of health access and stigma,” Dr. Mei said. “Health care providers should be notably cognizant of these increased risks, particularly in the psychosocial context and make efforts to reduce those burdens as much as possible.”
The research was funded by the Stanford Maternal and Child Health Research Institute. Dr. Obedin-Maliver has consulted for Sage Therapeutics, Ibis Reproductive Health, and Hims. Dr. Mei and the other authors had no disclosures.
Members of the LGBTQ community who give birth appear to have a greater risk of hypertensive disorders of pregnancy and postpartum hemorrhage, according to new research presented at the annual meeting sponsored by the Society for Maternal-Fetal Medicine.
“Our study found that birthing patients in likely sexual and gender minority partnerships experienced disparities in clinical outcomes,” Stephanie Leonard, PhD, an epidemiology and biostatistics instructor at the Stanford (Calif.) University division of maternal-fetal medicine and obstetrics, told attendees at the meeting. The disparities are likely because of various social determinants and possibly higher use of assisted reproductive technology (ART). The findings establish “how these are significant disparities that have been largely overlooked and set the groundwork for doing further research on maybe ways that we can improve the inclusivity of obstetric care.”
Jenny Mei, MD, a maternal-fetal medicine fellow at the University of California, Los Angeles, who attended the presentation but was not involved in the research, said the findings were “overall unfortunate but not surprising given the existing studies looking at LGBTQ patients and their poorer health outcomes, largely due to lack of access to health care and discrimination in the health care setting.”
Dr. Leonard described the societal, interpersonal, and individual factors that can contribute to health disparities among gender and sexual minority patients.
“At the societal level, there are expectations of what it means to be pregnant, to give birth, and to be a parent. At the community level, there’s the clinical care environment, and at the interpersonal level, there’s an obstetrician’s relationship with the patient,” Dr. Leonard said. “At the individual level, most notably is minority stress, the biological effects of the chronic experience of discrimination.”
It has historically been difficult to collect data on this patient population, but a change in the design of the California birth certificate made it possible to gather more data than previously possible. The updated California birth certificate, issued in 2016, allows the parent not giving birth to check off whether they are the child’s mother, father, parent, or “not specified” instead of defaulting to “father.” In addition, the parent giving birth can select mother, father, parent or not specified instead of being “mother” by default.
The researchers classified sexual and gender minority (SGM) partnerships as those in which the parent giving birth was identified as the father and those where both parents were identified as mothers. Non-SGM minority partnerships were those in which the birthing parent was identified as the mother and the nonbirthing parent was identified as the father.
The population-based cohort study included data from all live birth hospitalizations from 2016-2019 in California, whose annual births represent one in eight babies born each year in the United States. The population of SGM patients different significantly from the non-SGM population in nearly every demographic and clinical factor except rates of pre-existing diabetes. For example, 42% of the SGM birthing patients were age 35 or older, compared with 23% of the non-SGM patients.
SGM patients were more likely to be born in the United States, were more likely to be White, and were less likely to be Asian or Hispanic. SGM patients had higher education levels and were more likely to have private insurance. They were also more likely to be nulliparous and have chronic hypertension. Average body mass index for SGM patients was 33 kg/m2, compared with 30 for non-SGM patients. SGM patients were also much more likely to have multifetal gestation: 7.1% of SGM patients versus 1.5% of non-SGM patients.
In terms of clinical outcomes, 14% of SGM patients had hypertensive disorders of pregnancy, compared with 8% of non-SGM patients. Before adjustment for potential confounders, SGM patients were also twice as likely to have postpartum hemorrhage (8% vs. 4% in non-SGM patients) and postterm birth at 42-44 weeks (0.6% vs 0.3% in non-SGM patients).
“Having increased postterm birth is a matter of declining induction of labor, as it is recommended to have an induction by 41 weeks of gestation in general,” Dr. Mei said in an interview. “It is also possible this patient cohort faces more barriers in access to care and possible discrimination as sexual/gender minority patients.”
Rates of severe preeclampsia, induction of labor, cesarean delivery, preterm birth, low birth weight, and a low Apgar score were also higher among SGM patients, but these associations were no longer significant after adjustment for age, education, payment method, parity, prepregnancy weight, comorbidities, and multifetal gestation. The difference in hypertensive disorders of pregnancy, postpartum hemorrhage, and postterm birth remained statistically significant after adjustment.
Past research has shown that only about a third of cisgender female same-sex marriages used ART, so the disparities cannot be completely explained by ART use, Dr. Leonard said.
“I think the main drivers are structural disparities,” Dr. Leonard said. “Every obstetric clinic is focused in a way that’s about mother-father, and many people who don’t feel like they fit into that paradigm feel excluded and disengage with health care.”
Elliott Main, MD, a clinical professor of obstetrics and gynecology at Stanford University and coauthor of the study noted that discrimination and stigma likely play a substantial role in the disparities.
“Sexual and/or gender minority people face this discrimination at structural and interpersonal levels on a regular basis, which can lead to chronic stress and its harmful physical effects as well as lower-quality health care,” Dr. Main said in an interview.
Another coauthor, Juno Obedin-Maliver, MD, an assistant professor of obstetrics and gynecology at Stanford, emphasized how much room for improvement exists in care for SGM obstetric patients.
“We hope that this study brings needed attention to the disparities in perinatal health experienced by sexual and/or gender minority people,” Dr. Obedin-Maliver said. “There is much we can do to better understand the family building goals of sexual and/or gender minority people and help those to be achieved with healthy outcomes for parents and their children.”
One limitation of the study is that it’s possible to misclassify individuals using the birth certificate data, and not everyone may be comfortable selecting the box that accurately represents their identity, particularly if they aren’t “out” or fear discrimination or stigma, so the population may underrepresent the actual numbers of sexual and gender minority individuals giving birth. Dr. Mei added that it would be helpful to see data on neonatal ICU admissions and use of ART.
It’s difficult to say how generalizable the findings are, Dr. Mei said. “It is possible the findings would be more exaggerated in the rest of the country outside of California, if we assume there is potentially lower health access and more stigma.” The fact that California offers different gender options for the birthing and nonbirthing parent is, by itself, an indication of a potentially more accepting social environment than might be found in other states.
”The take-home message is that this patient population is higher risk, likely partially due to baseline increased risk factors, such as older maternal age and likely use of ART, and partially due to possible lack of health access and stigma,” Dr. Mei said. “Health care providers should be notably cognizant of these increased risks, particularly in the psychosocial context and make efforts to reduce those burdens as much as possible.”
The research was funded by the Stanford Maternal and Child Health Research Institute. Dr. Obedin-Maliver has consulted for Sage Therapeutics, Ibis Reproductive Health, and Hims. Dr. Mei and the other authors had no disclosures.
Members of the LGBTQ community who give birth appear to have a greater risk of hypertensive disorders of pregnancy and postpartum hemorrhage, according to new research presented at the annual meeting sponsored by the Society for Maternal-Fetal Medicine.
“Our study found that birthing patients in likely sexual and gender minority partnerships experienced disparities in clinical outcomes,” Stephanie Leonard, PhD, an epidemiology and biostatistics instructor at the Stanford (Calif.) University division of maternal-fetal medicine and obstetrics, told attendees at the meeting. The disparities are likely because of various social determinants and possibly higher use of assisted reproductive technology (ART). The findings establish “how these are significant disparities that have been largely overlooked and set the groundwork for doing further research on maybe ways that we can improve the inclusivity of obstetric care.”
Jenny Mei, MD, a maternal-fetal medicine fellow at the University of California, Los Angeles, who attended the presentation but was not involved in the research, said the findings were “overall unfortunate but not surprising given the existing studies looking at LGBTQ patients and their poorer health outcomes, largely due to lack of access to health care and discrimination in the health care setting.”
Dr. Leonard described the societal, interpersonal, and individual factors that can contribute to health disparities among gender and sexual minority patients.
“At the societal level, there are expectations of what it means to be pregnant, to give birth, and to be a parent. At the community level, there’s the clinical care environment, and at the interpersonal level, there’s an obstetrician’s relationship with the patient,” Dr. Leonard said. “At the individual level, most notably is minority stress, the biological effects of the chronic experience of discrimination.”
It has historically been difficult to collect data on this patient population, but a change in the design of the California birth certificate made it possible to gather more data than previously possible. The updated California birth certificate, issued in 2016, allows the parent not giving birth to check off whether they are the child’s mother, father, parent, or “not specified” instead of defaulting to “father.” In addition, the parent giving birth can select mother, father, parent or not specified instead of being “mother” by default.
The researchers classified sexual and gender minority (SGM) partnerships as those in which the parent giving birth was identified as the father and those where both parents were identified as mothers. Non-SGM minority partnerships were those in which the birthing parent was identified as the mother and the nonbirthing parent was identified as the father.
The population-based cohort study included data from all live birth hospitalizations from 2016-2019 in California, whose annual births represent one in eight babies born each year in the United States. The population of SGM patients different significantly from the non-SGM population in nearly every demographic and clinical factor except rates of pre-existing diabetes. For example, 42% of the SGM birthing patients were age 35 or older, compared with 23% of the non-SGM patients.
SGM patients were more likely to be born in the United States, were more likely to be White, and were less likely to be Asian or Hispanic. SGM patients had higher education levels and were more likely to have private insurance. They were also more likely to be nulliparous and have chronic hypertension. Average body mass index for SGM patients was 33 kg/m2, compared with 30 for non-SGM patients. SGM patients were also much more likely to have multifetal gestation: 7.1% of SGM patients versus 1.5% of non-SGM patients.
In terms of clinical outcomes, 14% of SGM patients had hypertensive disorders of pregnancy, compared with 8% of non-SGM patients. Before adjustment for potential confounders, SGM patients were also twice as likely to have postpartum hemorrhage (8% vs. 4% in non-SGM patients) and postterm birth at 42-44 weeks (0.6% vs 0.3% in non-SGM patients).
“Having increased postterm birth is a matter of declining induction of labor, as it is recommended to have an induction by 41 weeks of gestation in general,” Dr. Mei said in an interview. “It is also possible this patient cohort faces more barriers in access to care and possible discrimination as sexual/gender minority patients.”
Rates of severe preeclampsia, induction of labor, cesarean delivery, preterm birth, low birth weight, and a low Apgar score were also higher among SGM patients, but these associations were no longer significant after adjustment for age, education, payment method, parity, prepregnancy weight, comorbidities, and multifetal gestation. The difference in hypertensive disorders of pregnancy, postpartum hemorrhage, and postterm birth remained statistically significant after adjustment.
Past research has shown that only about a third of cisgender female same-sex marriages used ART, so the disparities cannot be completely explained by ART use, Dr. Leonard said.
“I think the main drivers are structural disparities,” Dr. Leonard said. “Every obstetric clinic is focused in a way that’s about mother-father, and many people who don’t feel like they fit into that paradigm feel excluded and disengage with health care.”
Elliott Main, MD, a clinical professor of obstetrics and gynecology at Stanford University and coauthor of the study noted that discrimination and stigma likely play a substantial role in the disparities.
“Sexual and/or gender minority people face this discrimination at structural and interpersonal levels on a regular basis, which can lead to chronic stress and its harmful physical effects as well as lower-quality health care,” Dr. Main said in an interview.
Another coauthor, Juno Obedin-Maliver, MD, an assistant professor of obstetrics and gynecology at Stanford, emphasized how much room for improvement exists in care for SGM obstetric patients.
“We hope that this study brings needed attention to the disparities in perinatal health experienced by sexual and/or gender minority people,” Dr. Obedin-Maliver said. “There is much we can do to better understand the family building goals of sexual and/or gender minority people and help those to be achieved with healthy outcomes for parents and their children.”
One limitation of the study is that it’s possible to misclassify individuals using the birth certificate data, and not everyone may be comfortable selecting the box that accurately represents their identity, particularly if they aren’t “out” or fear discrimination or stigma, so the population may underrepresent the actual numbers of sexual and gender minority individuals giving birth. Dr. Mei added that it would be helpful to see data on neonatal ICU admissions and use of ART.
It’s difficult to say how generalizable the findings are, Dr. Mei said. “It is possible the findings would be more exaggerated in the rest of the country outside of California, if we assume there is potentially lower health access and more stigma.” The fact that California offers different gender options for the birthing and nonbirthing parent is, by itself, an indication of a potentially more accepting social environment than might be found in other states.
”The take-home message is that this patient population is higher risk, likely partially due to baseline increased risk factors, such as older maternal age and likely use of ART, and partially due to possible lack of health access and stigma,” Dr. Mei said. “Health care providers should be notably cognizant of these increased risks, particularly in the psychosocial context and make efforts to reduce those burdens as much as possible.”
The research was funded by the Stanford Maternal and Child Health Research Institute. Dr. Obedin-Maliver has consulted for Sage Therapeutics, Ibis Reproductive Health, and Hims. Dr. Mei and the other authors had no disclosures.
FROM THE PREGNANCY MEETING
Picture warnings on sugary drinks could help fight childhood obesity
Sugary beverages – juice, soda, decadent lattes, sports drinks, and more – are the leading source of both calories and added sugar in the American diet, according to the Harvard T. H. Chan School of Public Health. But new research published in PLoS Medicine has found that
The new study, from researchers at the University of North Carolina at Chapel Hill, found that parents were 17% less likely to buy sugary drinks for their children when the beverages had the graphical – and graphic – warnings on the products.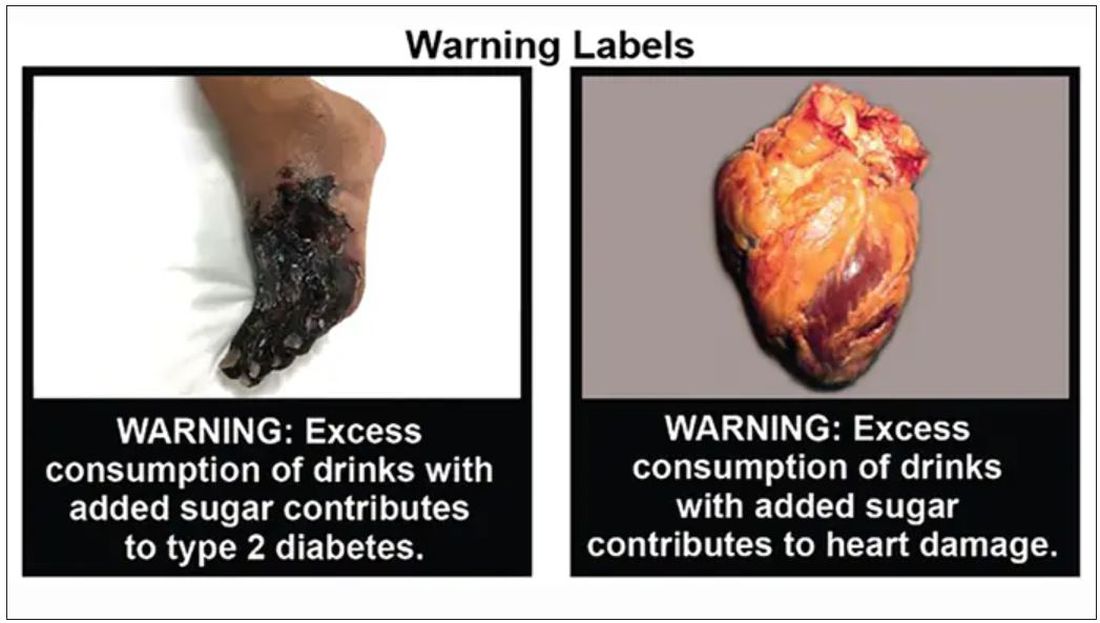
The researchers turned a laboratory setting into a “minimart,” and parents were told to choose one drink and snack for their children, along with one household item (to disguise the purpose of the study).
Some parents were presented with sweetened drinks with images on the products reflecting type 2 diabetes and heart damage. Others were shown sugary drinks with a barcode label and no picture warning.
Forty-five percent of parents chose sugary drinks for their children when the products had no picture warning, but only 28% of parents chose sugary beverages with the cautionary images.
“When people make choices about what food to buy, they are juggling dozens of factors, like taste, cost, and advertising, and are looking at many products at once,” said Lindsey Smith Taillie, PhD, a nutrition researcher and the senior author of the paper.
“Showing that warnings can cut through the noise of everything else that’s happening in a food store is powerful evidence that they would help reduce sugary drink purchases in the real world.”
Children are particularly prone to overindulging on sugar, largely due to companies’ frequent marketing displays of pleasurable-looking and seemingly “thirst-quenching” sweet beverages.
Drink packaging also can be misleading.
Fruits and vegetables displayed on the front of many beverages often lead parents to buy what they believe are “healthy” options, when these drinks could be packed with sugar, according to a study published online Jan. 22 in the journal Appetite.
Parents are often “doing the best with what information they have,” so more education about nutrition, through picture warning labels, for example, would make a difference, said Caroline Fausel, a paleo food blogger, podcaster, and author of “Prep, Cook, Freeze: A Paleo Meal Planning Cookbook.”
Healthier choices on the rise
The American Beverage Association, an industry trade group, shared the current steps that major companies are taking to help lower Americans’ sugar intake.
Pepsi, Coca-Cola, and Keurig Dr Pepper joined forces in 2014 to create the Balance Calories Initiative, which aims to reduce beverage calories in the national diet.
Coca-Cola now offers 250 beverages with zero to low calories, and Keurig Dr Pepper has 158 products with 40 calories or less. Pepsi sells 7.5-ounce mini-cans, along with various other sizes, to encourage portion control.
“Beverage companies are fully transparent about the calories and sugar in our products, and we are offering more choices with less sugar than ever before,” William Dermody, vice president of media and public affairs for the American Beverage Association, said in a statement. “We agree that too much sugar is not good for anyone, and clear information about beverages is most helpful to consumers.”
Other big companies also are taking strides to lower sugar content in their products.
Kraft Heinz, which owns the popular line of Capri Sun drinks, has publicly shared its efforts to ramp up the nutritional value of its products.
The company has a goal to slash 60 million pounds of total sugar in Kraft Heinz products globally by 2025.
“As more people become aware of the harm that excessive sugar can cause in the body, my hope is that they continue to choose healthier alternatives,” Ms. Fausel said.
Creating new patterns
For children who consume sweetened juices and sodas regularly, the transition to healthier options might be challenging at first.
“Change can involve tantrums and unhappiness, and right now parents are at their max living pandemic parenting life,” said Jennifer Anderson, a registered dietitian and CEO of Kids Eat in Color, LLC, a resource for improving child nutrition and health through innovative education, meal plans, and tools. “Kids can get used to having sugary drinks, and they don’t want to give them up.”
One way to help make the switch is by having only water and milk as options while children are up and about, a technique that works particularly well for younger children, she said.
“This sort of ‘quiet restriction’ helps kids learn to love the healthier option without feeling deprived,” Ms. Anderson said. “They will eventually learn about juice, soda, chocolate milk, sports drinks, and more, but you can let them learn about those foods at a slower pace when you rarely or don’t serve them at home.”
The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sugary beverages – juice, soda, decadent lattes, sports drinks, and more – are the leading source of both calories and added sugar in the American diet, according to the Harvard T. H. Chan School of Public Health. But new research published in PLoS Medicine has found that
The new study, from researchers at the University of North Carolina at Chapel Hill, found that parents were 17% less likely to buy sugary drinks for their children when the beverages had the graphical – and graphic – warnings on the products.
The researchers turned a laboratory setting into a “minimart,” and parents were told to choose one drink and snack for their children, along with one household item (to disguise the purpose of the study).
Some parents were presented with sweetened drinks with images on the products reflecting type 2 diabetes and heart damage. Others were shown sugary drinks with a barcode label and no picture warning.
Forty-five percent of parents chose sugary drinks for their children when the products had no picture warning, but only 28% of parents chose sugary beverages with the cautionary images.
“When people make choices about what food to buy, they are juggling dozens of factors, like taste, cost, and advertising, and are looking at many products at once,” said Lindsey Smith Taillie, PhD, a nutrition researcher and the senior author of the paper.
“Showing that warnings can cut through the noise of everything else that’s happening in a food store is powerful evidence that they would help reduce sugary drink purchases in the real world.”
Children are particularly prone to overindulging on sugar, largely due to companies’ frequent marketing displays of pleasurable-looking and seemingly “thirst-quenching” sweet beverages.
Drink packaging also can be misleading.
Fruits and vegetables displayed on the front of many beverages often lead parents to buy what they believe are “healthy” options, when these drinks could be packed with sugar, according to a study published online Jan. 22 in the journal Appetite.
Parents are often “doing the best with what information they have,” so more education about nutrition, through picture warning labels, for example, would make a difference, said Caroline Fausel, a paleo food blogger, podcaster, and author of “Prep, Cook, Freeze: A Paleo Meal Planning Cookbook.”
Healthier choices on the rise
The American Beverage Association, an industry trade group, shared the current steps that major companies are taking to help lower Americans’ sugar intake.
Pepsi, Coca-Cola, and Keurig Dr Pepper joined forces in 2014 to create the Balance Calories Initiative, which aims to reduce beverage calories in the national diet.
Coca-Cola now offers 250 beverages with zero to low calories, and Keurig Dr Pepper has 158 products with 40 calories or less. Pepsi sells 7.5-ounce mini-cans, along with various other sizes, to encourage portion control.
“Beverage companies are fully transparent about the calories and sugar in our products, and we are offering more choices with less sugar than ever before,” William Dermody, vice president of media and public affairs for the American Beverage Association, said in a statement. “We agree that too much sugar is not good for anyone, and clear information about beverages is most helpful to consumers.”
Other big companies also are taking strides to lower sugar content in their products.
Kraft Heinz, which owns the popular line of Capri Sun drinks, has publicly shared its efforts to ramp up the nutritional value of its products.
The company has a goal to slash 60 million pounds of total sugar in Kraft Heinz products globally by 2025.
“As more people become aware of the harm that excessive sugar can cause in the body, my hope is that they continue to choose healthier alternatives,” Ms. Fausel said.
Creating new patterns
For children who consume sweetened juices and sodas regularly, the transition to healthier options might be challenging at first.
“Change can involve tantrums and unhappiness, and right now parents are at their max living pandemic parenting life,” said Jennifer Anderson, a registered dietitian and CEO of Kids Eat in Color, LLC, a resource for improving child nutrition and health through innovative education, meal plans, and tools. “Kids can get used to having sugary drinks, and they don’t want to give them up.”
One way to help make the switch is by having only water and milk as options while children are up and about, a technique that works particularly well for younger children, she said.
“This sort of ‘quiet restriction’ helps kids learn to love the healthier option without feeling deprived,” Ms. Anderson said. “They will eventually learn about juice, soda, chocolate milk, sports drinks, and more, but you can let them learn about those foods at a slower pace when you rarely or don’t serve them at home.”
The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sugary beverages – juice, soda, decadent lattes, sports drinks, and more – are the leading source of both calories and added sugar in the American diet, according to the Harvard T. H. Chan School of Public Health. But new research published in PLoS Medicine has found that
The new study, from researchers at the University of North Carolina at Chapel Hill, found that parents were 17% less likely to buy sugary drinks for their children when the beverages had the graphical – and graphic – warnings on the products.
The researchers turned a laboratory setting into a “minimart,” and parents were told to choose one drink and snack for their children, along with one household item (to disguise the purpose of the study).
Some parents were presented with sweetened drinks with images on the products reflecting type 2 diabetes and heart damage. Others were shown sugary drinks with a barcode label and no picture warning.
Forty-five percent of parents chose sugary drinks for their children when the products had no picture warning, but only 28% of parents chose sugary beverages with the cautionary images.
“When people make choices about what food to buy, they are juggling dozens of factors, like taste, cost, and advertising, and are looking at many products at once,” said Lindsey Smith Taillie, PhD, a nutrition researcher and the senior author of the paper.
“Showing that warnings can cut through the noise of everything else that’s happening in a food store is powerful evidence that they would help reduce sugary drink purchases in the real world.”
Children are particularly prone to overindulging on sugar, largely due to companies’ frequent marketing displays of pleasurable-looking and seemingly “thirst-quenching” sweet beverages.
Drink packaging also can be misleading.
Fruits and vegetables displayed on the front of many beverages often lead parents to buy what they believe are “healthy” options, when these drinks could be packed with sugar, according to a study published online Jan. 22 in the journal Appetite.
Parents are often “doing the best with what information they have,” so more education about nutrition, through picture warning labels, for example, would make a difference, said Caroline Fausel, a paleo food blogger, podcaster, and author of “Prep, Cook, Freeze: A Paleo Meal Planning Cookbook.”
Healthier choices on the rise
The American Beverage Association, an industry trade group, shared the current steps that major companies are taking to help lower Americans’ sugar intake.
Pepsi, Coca-Cola, and Keurig Dr Pepper joined forces in 2014 to create the Balance Calories Initiative, which aims to reduce beverage calories in the national diet.
Coca-Cola now offers 250 beverages with zero to low calories, and Keurig Dr Pepper has 158 products with 40 calories or less. Pepsi sells 7.5-ounce mini-cans, along with various other sizes, to encourage portion control.
“Beverage companies are fully transparent about the calories and sugar in our products, and we are offering more choices with less sugar than ever before,” William Dermody, vice president of media and public affairs for the American Beverage Association, said in a statement. “We agree that too much sugar is not good for anyone, and clear information about beverages is most helpful to consumers.”
Other big companies also are taking strides to lower sugar content in their products.
Kraft Heinz, which owns the popular line of Capri Sun drinks, has publicly shared its efforts to ramp up the nutritional value of its products.
The company has a goal to slash 60 million pounds of total sugar in Kraft Heinz products globally by 2025.
“As more people become aware of the harm that excessive sugar can cause in the body, my hope is that they continue to choose healthier alternatives,” Ms. Fausel said.
Creating new patterns
For children who consume sweetened juices and sodas regularly, the transition to healthier options might be challenging at first.
“Change can involve tantrums and unhappiness, and right now parents are at their max living pandemic parenting life,” said Jennifer Anderson, a registered dietitian and CEO of Kids Eat in Color, LLC, a resource for improving child nutrition and health through innovative education, meal plans, and tools. “Kids can get used to having sugary drinks, and they don’t want to give them up.”
One way to help make the switch is by having only water and milk as options while children are up and about, a technique that works particularly well for younger children, she said.
“This sort of ‘quiet restriction’ helps kids learn to love the healthier option without feeling deprived,” Ms. Anderson said. “They will eventually learn about juice, soda, chocolate milk, sports drinks, and more, but you can let them learn about those foods at a slower pace when you rarely or don’t serve them at home.”
The researchers reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PLOS MEDICINE
2022 Update on fertility
In this Update, the authors discuss 2 important areas that impact fertility. First, with in vitro fertilization (IVF), successful implantation that leads to live birth requires a normal embryo and a receptive endometrium. While research using advanced molecular array technology has resulted in a clinical test to identify the optimal window of implantation, recent evidence has questioned its clinical effectiveness. Second, recognizing the importance of endometriosis—a common disease with high burden that causes pain, infertility, and other symptoms—the World Health Organization (WHO) last year published an informative fact sheet that highlights the diagnosis, treatment options, and challenges of this significant disease.
Endometrial receptivity array and the quest for optimal endometrial preparation prior to embryo transfer in IVF
Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
A successful pregnancy requires optimal crosstalk between the embryo and the endometrium. Over the past several decades, research efforts to improve IVF outcomes have been focused mainly on the embryo factor and methods to improve embryo selection, such as extended culture to blastocyst, time-lapse imaging (morphokinetic assessment), and more notably, preimplantation genetic testing for aneuploidy (PGT-A). However, the other half of the equation, the endometrium, has not garnered the attention that it deserves. Effort has therefore been renewed to optimize the endometrial factor by better diagnosing and treating various forms of endometrial dysfunction that could lead to infertility in general and lack of success with IVF and euploid embryo transfers in particular.
Historical background on endometrial function
Progesterone has long been recognized as the main effector that transforms the estrogen-primed endometrium into a receptive state that results in successful embryo implantation. Progesterone exposure is required at appropriate levels and duration before the endometrium becomes receptive to the embryo. If implantation does not occur soon after the endometrium has attained receptive status (7–10 days after ovulation), further progesterone exposure results in progression of endometrial changes that no longer permit successful implantation.
As early as the 1950s, “luteal phase deficiency” was defined as due to inadequate progesterone secretion and resulted in a short luteal phase. In the 1970s, histologic “dating” of the endometrium became the gold standard for diagnosing luteal phase defects; this relied on a classic histologic appearance of secretory phase endometrium and its changes throughout the luteal phase. Subsequently, however, results of prospective randomized controlled trials published in 2004 cast significant doubt on the accuracy and reproducibility of these endometrial biopsies and did not show any clinical diagnostic benefit or correlation with pregnancy outcomes.
21st century advances: Endometrial dating 2.0
A decade later, with the advancement of molecular biology tools such as microarray technology, researchers were able to study endometrial gene expression patterns at different stages of the menstrual cycle. They identified different phases of endometrial development with molecular profiles, or “signatures,” for the luteal phase, endometriosis, polycystic ovary syndrome, and uterine fibroids.
In 2013, researchers in Spain introduced a diagnostic test called endometrial receptivity array (ERA) with the stated goal of being able to temporally define the receptive endometrium and identify prereceptive as well as postreceptive states.1 In other words, instead of the histologic dating of the endometrium used in the 1970s, it represented “molecular dating” of the endometrium. Although the initial studies were conducted among women who experienced prior unsuccessful embryo transfers (the so-called recurrent implantation failure, or RIF), the test’s scope was subsequently expanded to include any individual planning on a frozen embryo transfer (FET), regardless of any prior attempts. The term personalized embryo transfer (pET) was coined to suggest the ability to define the best time (up to hours) for embryo transfers on an individual basis. Despite lack of independent validation studies, ERA was then widely adopted by many clinicians (and requested by some patients) with the hope of improving IVF outcomes.
However, not unlike many other novel innovations in assisted reproductive technology, ERA regrettably did not withstand the test of time. Three independent studies in 2021, 1 randomized clinical trial and 2 observational cohort studies, did not show any benefit with regard to implantation rates, pregnancy rates, or live birth rates when ERA was performed in the general infertility population.2-4
Continue to: Study results...
Study results
The cohort study that matched 133 ERA patients with 353 non-ERA patients showed live birth rates of 49.62% for the ERA group and 54.96% for the non-ERA group (odds ratio [OR], 0.8074; 95% confidence interval [CI], 0.5424–1.2018).2 Of note, no difference occurred between subgroups based on the prior number of FETs or the receptivity status (TABLE 1).
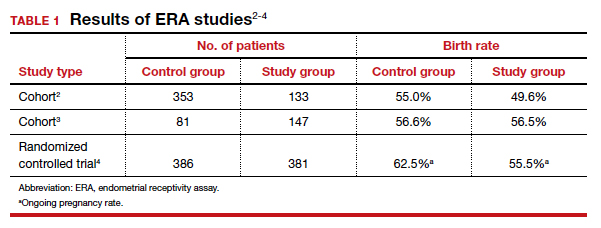
Another cohort study from the University of California, Los Angeles, published in 2021 analyzed 228 single euploid FET cycles.3 This study did not show any benefit for routine ERA testing, with a live birth rate of 56.6% in the non-ERA group and 56.5% in the ERA group.
Still, the most convincing evidence for the lack of benefit from routine ERA was noted from the results of the randomized clinical trial.4 A total of 767 patients were randomly allocated, 381 to the ERA group and 386 to the control group. There was no difference in ongoing pregnancy rates between the 2 groups. Perhaps more important, even after limiting the analysis to individuals with a nonreceptive ERA result, there was no difference in ongoing pregnancy rates between the 2 groups: 62.5% in the control group (default timing of transfer) and 55.5% in the study group (transfer timing adjusted based on ERA) (rate ratio [RR], 0.9; 95% CI, 0.70–1.14).
ERA usefulness is unsupported in general infertility population
The studies discussed collectively suggest with a high degree of certainty that there is no indication for routine ERA testing in the general infertility population prior to frozen embryo transfers.
Although these studies all were conducted in the general infertility population and did not specifically evaluate the performance of ERA in women with recurrent pregnancy loss or recurrent implantation failure, it is important to acknowledge that if ERA were truly able to define the window of receptivity, one would expect a lower implantation rate if the embryos were transferred outside of the window suggested by the ERA. This was not the case in these studies, as they all showed equivalent pregnancy rates in the control (nonadjusted) groups even when ERA suggested a nonreceptive status.
This observation seriously questions the validity of ERA regarding its ability to temporally define the window of receptivity. On the other hand, as stated earlier, there is still a possibility for ERA to be beneficial for a small subgroup of patients whose window of receptivity may not be as wide as expected in the general population. The challenging question would be how best to identify the particular group with a narrow, or displaced, window of receptivity.
The optimal timing for implantation of a normal embryo requires a receptive endometrium. The endometrial biopsy was used widely for many years before research showed it was not clinically useful. More recently, the endometrial receptivity array has been suggested to help time the frozen embryo transfer. Unfortunately, recent studies have shown that this test is not clinically useful for the general infertility population.
Continue to: WHO raises awareness of endometriosis burden and...
WHO raises awareness of endometriosis burden and highlights need to address diagnosis and treatment for women’s reproductive health
World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room /fact-sheets/detail/endometriosis. Accessed January 3, 2022.
The WHO published its first fact sheet on endometriosis in March 2021, recognizing endometriosis as a severe disease that affects almost 190 million women with life-impacting pain, infertility, other symptoms, and especially with chronic, significant emotional sequelae (TABLE 2).5 The disease’s variable and broad symptoms result in a lack of awareness and diagnosis by both women and health care providers, especially in low- and middle-income countries and in disadvantaged populations in developed countries. Increased awareness to promote earlier diagnosis, improved training for better management, expanded research for greater understanding, and policies that increase access to quality care are needed to ensure the reproductive health and rights of tens of millions of women with endometriosis.

Endometriosis characteristics and symptoms
Endometriosis is characterized by the presence of tissue resembling endometrium outside the uterus, where it causes a chronic inflammatory reaction that may result in the formation of scar tissue. Endometriotic lesions may be superficial, cystic ovarian endometriomas, or deep lesions, causing a myriad of pain and related symptoms.6.7
Chronic pain may occur because pain centers in the brain become hyperresponsive over time (central sensitization); this can occur at any point throughout the life course of endometriosis, even when endometriosis lesions are no longer visible. Sometimes, endometriosis is asymptomatic. In addition, endometriosis can cause infertility through anatomic distortion and inflammatory, endocrinologic, and other pathways.
The origins of endometriosis are thought to be multifactorial and include retrograde menstruation, cellular metaplasia, and/or stem cells that spread through blood and lymphatic vessels. Endometriosis is estrogen dependent, but lesion growth also is affected by altered or impaired immunity, localized complex hormonal influences, genetics, and possibly environmental contaminants.
Impact on public health and reproductive rights
Endometriosis has significant social, public health, and economic implications. It can decrease quality of life and prevent girls and women from attending work or school.8 Painful sex can affect sexual health. The WHO states that, “Addressing endometriosis will empower those affected by it, by supporting their human right to the highest standard of sexual and reproductive health, quality of life, and overall well-being.”5
At present, no known way is available to prevent or cure endometriosis. Early diagnosis and treatment, however, may slow or halt its natural progression and associated symptoms.
Diagnostic steps and treatment options
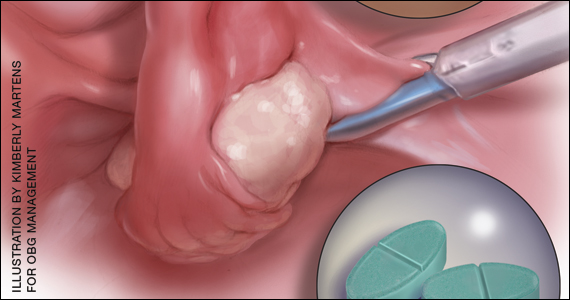
Early suspicion of endometriosis is the most important factor, followed by a careful history of menstrual symptoms and chronic pelvic pain, early referral to specialists for ultrasonography or other imaging, and sometimes surgical or laparoscopic visualization. Empirical treatment can be begun without histologic or laparoscopic confirmation.
Endometriosis can be treated with medications and/or surgery depending on symptoms, lesions, desired outcome, and patient choice.5,6 Common therapies include contraceptive steroids, nonsteroidal anti-inflammatory medications, and analgesics. Medical treatments focus on either lowering estrogen or increasing progesterone levels.
Surgery can remove endometriosis lesions, adhesions, and scar tissue. However, success in reducing pain symptoms and increasing pregnancy rates often depends on the extent of disease.
For infertility due to endometriosis, treatment options include laparoscopic surgical removal of endometriosis, ovarian stimulation with intrauterine insemination (IUI), and IVF. Multidisciplinary treatment addressing different symptoms and overall health often requires referral to pain experts and other specialists.9
The WHO perspective on endometriosis
Recognizing the importance of endometriosis and its impact on people’s sexual and reproductive health, quality of life, and overall well-being, the WHO is taking action to improve awareness, diagnosis, and treatment of endometriosis (TABLE 3).5 ●

Endometriosis is now recognized as a disease with significant burden for women everywhere. Widespread lack of awareness of presenting symptoms and management options means that all women’s health care clinicians need to become better informed about endometriosis so they can improve the quality of care they provide.
- Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100:818-824.
- Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
- Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
- Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
- World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room/fact-sheets/detail /endometriosis. Accessed January 3, 2022.
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382:1244-1256.
- Johnson NP, Hummelshoj L, Adamson GD, et al. World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 2017;32:315-324.
- Nnoaham K, Hummelshoj L, Webster P, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96:366-373.e8.
- Carey ET, Till SR, As-Sanie S. Pharmacological management of chronic pelvic pain in women. Drugs. 2017;77:285-301.
In this Update, the authors discuss 2 important areas that impact fertility. First, with in vitro fertilization (IVF), successful implantation that leads to live birth requires a normal embryo and a receptive endometrium. While research using advanced molecular array technology has resulted in a clinical test to identify the optimal window of implantation, recent evidence has questioned its clinical effectiveness. Second, recognizing the importance of endometriosis—a common disease with high burden that causes pain, infertility, and other symptoms—the World Health Organization (WHO) last year published an informative fact sheet that highlights the diagnosis, treatment options, and challenges of this significant disease.
Endometrial receptivity array and the quest for optimal endometrial preparation prior to embryo transfer in IVF
Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
A successful pregnancy requires optimal crosstalk between the embryo and the endometrium. Over the past several decades, research efforts to improve IVF outcomes have been focused mainly on the embryo factor and methods to improve embryo selection, such as extended culture to blastocyst, time-lapse imaging (morphokinetic assessment), and more notably, preimplantation genetic testing for aneuploidy (PGT-A). However, the other half of the equation, the endometrium, has not garnered the attention that it deserves. Effort has therefore been renewed to optimize the endometrial factor by better diagnosing and treating various forms of endometrial dysfunction that could lead to infertility in general and lack of success with IVF and euploid embryo transfers in particular.
Historical background on endometrial function
Progesterone has long been recognized as the main effector that transforms the estrogen-primed endometrium into a receptive state that results in successful embryo implantation. Progesterone exposure is required at appropriate levels and duration before the endometrium becomes receptive to the embryo. If implantation does not occur soon after the endometrium has attained receptive status (7–10 days after ovulation), further progesterone exposure results in progression of endometrial changes that no longer permit successful implantation.
As early as the 1950s, “luteal phase deficiency” was defined as due to inadequate progesterone secretion and resulted in a short luteal phase. In the 1970s, histologic “dating” of the endometrium became the gold standard for diagnosing luteal phase defects; this relied on a classic histologic appearance of secretory phase endometrium and its changes throughout the luteal phase. Subsequently, however, results of prospective randomized controlled trials published in 2004 cast significant doubt on the accuracy and reproducibility of these endometrial biopsies and did not show any clinical diagnostic benefit or correlation with pregnancy outcomes.
21st century advances: Endometrial dating 2.0
A decade later, with the advancement of molecular biology tools such as microarray technology, researchers were able to study endometrial gene expression patterns at different stages of the menstrual cycle. They identified different phases of endometrial development with molecular profiles, or “signatures,” for the luteal phase, endometriosis, polycystic ovary syndrome, and uterine fibroids.
In 2013, researchers in Spain introduced a diagnostic test called endometrial receptivity array (ERA) with the stated goal of being able to temporally define the receptive endometrium and identify prereceptive as well as postreceptive states.1 In other words, instead of the histologic dating of the endometrium used in the 1970s, it represented “molecular dating” of the endometrium. Although the initial studies were conducted among women who experienced prior unsuccessful embryo transfers (the so-called recurrent implantation failure, or RIF), the test’s scope was subsequently expanded to include any individual planning on a frozen embryo transfer (FET), regardless of any prior attempts. The term personalized embryo transfer (pET) was coined to suggest the ability to define the best time (up to hours) for embryo transfers on an individual basis. Despite lack of independent validation studies, ERA was then widely adopted by many clinicians (and requested by some patients) with the hope of improving IVF outcomes.
However, not unlike many other novel innovations in assisted reproductive technology, ERA regrettably did not withstand the test of time. Three independent studies in 2021, 1 randomized clinical trial and 2 observational cohort studies, did not show any benefit with regard to implantation rates, pregnancy rates, or live birth rates when ERA was performed in the general infertility population.2-4
Continue to: Study results...
Study results
The cohort study that matched 133 ERA patients with 353 non-ERA patients showed live birth rates of 49.62% for the ERA group and 54.96% for the non-ERA group (odds ratio [OR], 0.8074; 95% confidence interval [CI], 0.5424–1.2018).2 Of note, no difference occurred between subgroups based on the prior number of FETs or the receptivity status (TABLE 1).

Another cohort study from the University of California, Los Angeles, published in 2021 analyzed 228 single euploid FET cycles.3 This study did not show any benefit for routine ERA testing, with a live birth rate of 56.6% in the non-ERA group and 56.5% in the ERA group.
Still, the most convincing evidence for the lack of benefit from routine ERA was noted from the results of the randomized clinical trial.4 A total of 767 patients were randomly allocated, 381 to the ERA group and 386 to the control group. There was no difference in ongoing pregnancy rates between the 2 groups. Perhaps more important, even after limiting the analysis to individuals with a nonreceptive ERA result, there was no difference in ongoing pregnancy rates between the 2 groups: 62.5% in the control group (default timing of transfer) and 55.5% in the study group (transfer timing adjusted based on ERA) (rate ratio [RR], 0.9; 95% CI, 0.70–1.14).
ERA usefulness is unsupported in general infertility population
The studies discussed collectively suggest with a high degree of certainty that there is no indication for routine ERA testing in the general infertility population prior to frozen embryo transfers.
Although these studies all were conducted in the general infertility population and did not specifically evaluate the performance of ERA in women with recurrent pregnancy loss or recurrent implantation failure, it is important to acknowledge that if ERA were truly able to define the window of receptivity, one would expect a lower implantation rate if the embryos were transferred outside of the window suggested by the ERA. This was not the case in these studies, as they all showed equivalent pregnancy rates in the control (nonadjusted) groups even when ERA suggested a nonreceptive status.
This observation seriously questions the validity of ERA regarding its ability to temporally define the window of receptivity. On the other hand, as stated earlier, there is still a possibility for ERA to be beneficial for a small subgroup of patients whose window of receptivity may not be as wide as expected in the general population. The challenging question would be how best to identify the particular group with a narrow, or displaced, window of receptivity.
The optimal timing for implantation of a normal embryo requires a receptive endometrium. The endometrial biopsy was used widely for many years before research showed it was not clinically useful. More recently, the endometrial receptivity array has been suggested to help time the frozen embryo transfer. Unfortunately, recent studies have shown that this test is not clinically useful for the general infertility population.
Continue to: WHO raises awareness of endometriosis burden and...
WHO raises awareness of endometriosis burden and highlights need to address diagnosis and treatment for women’s reproductive health
World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room /fact-sheets/detail/endometriosis. Accessed January 3, 2022.
The WHO published its first fact sheet on endometriosis in March 2021, recognizing endometriosis as a severe disease that affects almost 190 million women with life-impacting pain, infertility, other symptoms, and especially with chronic, significant emotional sequelae (TABLE 2).5 The disease’s variable and broad symptoms result in a lack of awareness and diagnosis by both women and health care providers, especially in low- and middle-income countries and in disadvantaged populations in developed countries. Increased awareness to promote earlier diagnosis, improved training for better management, expanded research for greater understanding, and policies that increase access to quality care are needed to ensure the reproductive health and rights of tens of millions of women with endometriosis.

Endometriosis characteristics and symptoms
Endometriosis is characterized by the presence of tissue resembling endometrium outside the uterus, where it causes a chronic inflammatory reaction that may result in the formation of scar tissue. Endometriotic lesions may be superficial, cystic ovarian endometriomas, or deep lesions, causing a myriad of pain and related symptoms.6.7
Chronic pain may occur because pain centers in the brain become hyperresponsive over time (central sensitization); this can occur at any point throughout the life course of endometriosis, even when endometriosis lesions are no longer visible. Sometimes, endometriosis is asymptomatic. In addition, endometriosis can cause infertility through anatomic distortion and inflammatory, endocrinologic, and other pathways.
The origins of endometriosis are thought to be multifactorial and include retrograde menstruation, cellular metaplasia, and/or stem cells that spread through blood and lymphatic vessels. Endometriosis is estrogen dependent, but lesion growth also is affected by altered or impaired immunity, localized complex hormonal influences, genetics, and possibly environmental contaminants.
Impact on public health and reproductive rights
Endometriosis has significant social, public health, and economic implications. It can decrease quality of life and prevent girls and women from attending work or school.8 Painful sex can affect sexual health. The WHO states that, “Addressing endometriosis will empower those affected by it, by supporting their human right to the highest standard of sexual and reproductive health, quality of life, and overall well-being.”5
At present, no known way is available to prevent or cure endometriosis. Early diagnosis and treatment, however, may slow or halt its natural progression and associated symptoms.
Diagnostic steps and treatment options
Early suspicion of endometriosis is the most important factor, followed by a careful history of menstrual symptoms and chronic pelvic pain, early referral to specialists for ultrasonography or other imaging, and sometimes surgical or laparoscopic visualization. Empirical treatment can be begun without histologic or laparoscopic confirmation.
Endometriosis can be treated with medications and/or surgery depending on symptoms, lesions, desired outcome, and patient choice.5,6 Common therapies include contraceptive steroids, nonsteroidal anti-inflammatory medications, and analgesics. Medical treatments focus on either lowering estrogen or increasing progesterone levels.
Surgery can remove endometriosis lesions, adhesions, and scar tissue. However, success in reducing pain symptoms and increasing pregnancy rates often depends on the extent of disease.
For infertility due to endometriosis, treatment options include laparoscopic surgical removal of endometriosis, ovarian stimulation with intrauterine insemination (IUI), and IVF. Multidisciplinary treatment addressing different symptoms and overall health often requires referral to pain experts and other specialists.9
The WHO perspective on endometriosis
Recognizing the importance of endometriosis and its impact on people’s sexual and reproductive health, quality of life, and overall well-being, the WHO is taking action to improve awareness, diagnosis, and treatment of endometriosis (TABLE 3).5 ●

Endometriosis is now recognized as a disease with significant burden for women everywhere. Widespread lack of awareness of presenting symptoms and management options means that all women’s health care clinicians need to become better informed about endometriosis so they can improve the quality of care they provide.
In this Update, the authors discuss 2 important areas that impact fertility. First, with in vitro fertilization (IVF), successful implantation that leads to live birth requires a normal embryo and a receptive endometrium. While research using advanced molecular array technology has resulted in a clinical test to identify the optimal window of implantation, recent evidence has questioned its clinical effectiveness. Second, recognizing the importance of endometriosis—a common disease with high burden that causes pain, infertility, and other symptoms—the World Health Organization (WHO) last year published an informative fact sheet that highlights the diagnosis, treatment options, and challenges of this significant disease.
Endometrial receptivity array and the quest for optimal endometrial preparation prior to embryo transfer in IVF
Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
A successful pregnancy requires optimal crosstalk between the embryo and the endometrium. Over the past several decades, research efforts to improve IVF outcomes have been focused mainly on the embryo factor and methods to improve embryo selection, such as extended culture to blastocyst, time-lapse imaging (morphokinetic assessment), and more notably, preimplantation genetic testing for aneuploidy (PGT-A). However, the other half of the equation, the endometrium, has not garnered the attention that it deserves. Effort has therefore been renewed to optimize the endometrial factor by better diagnosing and treating various forms of endometrial dysfunction that could lead to infertility in general and lack of success with IVF and euploid embryo transfers in particular.
Historical background on endometrial function
Progesterone has long been recognized as the main effector that transforms the estrogen-primed endometrium into a receptive state that results in successful embryo implantation. Progesterone exposure is required at appropriate levels and duration before the endometrium becomes receptive to the embryo. If implantation does not occur soon after the endometrium has attained receptive status (7–10 days after ovulation), further progesterone exposure results in progression of endometrial changes that no longer permit successful implantation.
As early as the 1950s, “luteal phase deficiency” was defined as due to inadequate progesterone secretion and resulted in a short luteal phase. In the 1970s, histologic “dating” of the endometrium became the gold standard for diagnosing luteal phase defects; this relied on a classic histologic appearance of secretory phase endometrium and its changes throughout the luteal phase. Subsequently, however, results of prospective randomized controlled trials published in 2004 cast significant doubt on the accuracy and reproducibility of these endometrial biopsies and did not show any clinical diagnostic benefit or correlation with pregnancy outcomes.
21st century advances: Endometrial dating 2.0
A decade later, with the advancement of molecular biology tools such as microarray technology, researchers were able to study endometrial gene expression patterns at different stages of the menstrual cycle. They identified different phases of endometrial development with molecular profiles, or “signatures,” for the luteal phase, endometriosis, polycystic ovary syndrome, and uterine fibroids.
In 2013, researchers in Spain introduced a diagnostic test called endometrial receptivity array (ERA) with the stated goal of being able to temporally define the receptive endometrium and identify prereceptive as well as postreceptive states.1 In other words, instead of the histologic dating of the endometrium used in the 1970s, it represented “molecular dating” of the endometrium. Although the initial studies were conducted among women who experienced prior unsuccessful embryo transfers (the so-called recurrent implantation failure, or RIF), the test’s scope was subsequently expanded to include any individual planning on a frozen embryo transfer (FET), regardless of any prior attempts. The term personalized embryo transfer (pET) was coined to suggest the ability to define the best time (up to hours) for embryo transfers on an individual basis. Despite lack of independent validation studies, ERA was then widely adopted by many clinicians (and requested by some patients) with the hope of improving IVF outcomes.
However, not unlike many other novel innovations in assisted reproductive technology, ERA regrettably did not withstand the test of time. Three independent studies in 2021, 1 randomized clinical trial and 2 observational cohort studies, did not show any benefit with regard to implantation rates, pregnancy rates, or live birth rates when ERA was performed in the general infertility population.2-4
Continue to: Study results...
Study results
The cohort study that matched 133 ERA patients with 353 non-ERA patients showed live birth rates of 49.62% for the ERA group and 54.96% for the non-ERA group (odds ratio [OR], 0.8074; 95% confidence interval [CI], 0.5424–1.2018).2 Of note, no difference occurred between subgroups based on the prior number of FETs or the receptivity status (TABLE 1).

Another cohort study from the University of California, Los Angeles, published in 2021 analyzed 228 single euploid FET cycles.3 This study did not show any benefit for routine ERA testing, with a live birth rate of 56.6% in the non-ERA group and 56.5% in the ERA group.
Still, the most convincing evidence for the lack of benefit from routine ERA was noted from the results of the randomized clinical trial.4 A total of 767 patients were randomly allocated, 381 to the ERA group and 386 to the control group. There was no difference in ongoing pregnancy rates between the 2 groups. Perhaps more important, even after limiting the analysis to individuals with a nonreceptive ERA result, there was no difference in ongoing pregnancy rates between the 2 groups: 62.5% in the control group (default timing of transfer) and 55.5% in the study group (transfer timing adjusted based on ERA) (rate ratio [RR], 0.9; 95% CI, 0.70–1.14).
ERA usefulness is unsupported in general infertility population
The studies discussed collectively suggest with a high degree of certainty that there is no indication for routine ERA testing in the general infertility population prior to frozen embryo transfers.
Although these studies all were conducted in the general infertility population and did not specifically evaluate the performance of ERA in women with recurrent pregnancy loss or recurrent implantation failure, it is important to acknowledge that if ERA were truly able to define the window of receptivity, one would expect a lower implantation rate if the embryos were transferred outside of the window suggested by the ERA. This was not the case in these studies, as they all showed equivalent pregnancy rates in the control (nonadjusted) groups even when ERA suggested a nonreceptive status.
This observation seriously questions the validity of ERA regarding its ability to temporally define the window of receptivity. On the other hand, as stated earlier, there is still a possibility for ERA to be beneficial for a small subgroup of patients whose window of receptivity may not be as wide as expected in the general population. The challenging question would be how best to identify the particular group with a narrow, or displaced, window of receptivity.
The optimal timing for implantation of a normal embryo requires a receptive endometrium. The endometrial biopsy was used widely for many years before research showed it was not clinically useful. More recently, the endometrial receptivity array has been suggested to help time the frozen embryo transfer. Unfortunately, recent studies have shown that this test is not clinically useful for the general infertility population.
Continue to: WHO raises awareness of endometriosis burden and...
WHO raises awareness of endometriosis burden and highlights need to address diagnosis and treatment for women’s reproductive health
World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room /fact-sheets/detail/endometriosis. Accessed January 3, 2022.
The WHO published its first fact sheet on endometriosis in March 2021, recognizing endometriosis as a severe disease that affects almost 190 million women with life-impacting pain, infertility, other symptoms, and especially with chronic, significant emotional sequelae (TABLE 2).5 The disease’s variable and broad symptoms result in a lack of awareness and diagnosis by both women and health care providers, especially in low- and middle-income countries and in disadvantaged populations in developed countries. Increased awareness to promote earlier diagnosis, improved training for better management, expanded research for greater understanding, and policies that increase access to quality care are needed to ensure the reproductive health and rights of tens of millions of women with endometriosis.

Endometriosis characteristics and symptoms
Endometriosis is characterized by the presence of tissue resembling endometrium outside the uterus, where it causes a chronic inflammatory reaction that may result in the formation of scar tissue. Endometriotic lesions may be superficial, cystic ovarian endometriomas, or deep lesions, causing a myriad of pain and related symptoms.6.7
Chronic pain may occur because pain centers in the brain become hyperresponsive over time (central sensitization); this can occur at any point throughout the life course of endometriosis, even when endometriosis lesions are no longer visible. Sometimes, endometriosis is asymptomatic. In addition, endometriosis can cause infertility through anatomic distortion and inflammatory, endocrinologic, and other pathways.
The origins of endometriosis are thought to be multifactorial and include retrograde menstruation, cellular metaplasia, and/or stem cells that spread through blood and lymphatic vessels. Endometriosis is estrogen dependent, but lesion growth also is affected by altered or impaired immunity, localized complex hormonal influences, genetics, and possibly environmental contaminants.
Impact on public health and reproductive rights
Endometriosis has significant social, public health, and economic implications. It can decrease quality of life and prevent girls and women from attending work or school.8 Painful sex can affect sexual health. The WHO states that, “Addressing endometriosis will empower those affected by it, by supporting their human right to the highest standard of sexual and reproductive health, quality of life, and overall well-being.”5
At present, no known way is available to prevent or cure endometriosis. Early diagnosis and treatment, however, may slow or halt its natural progression and associated symptoms.
Diagnostic steps and treatment options
Early suspicion of endometriosis is the most important factor, followed by a careful history of menstrual symptoms and chronic pelvic pain, early referral to specialists for ultrasonography or other imaging, and sometimes surgical or laparoscopic visualization. Empirical treatment can be begun without histologic or laparoscopic confirmation.
Endometriosis can be treated with medications and/or surgery depending on symptoms, lesions, desired outcome, and patient choice.5,6 Common therapies include contraceptive steroids, nonsteroidal anti-inflammatory medications, and analgesics. Medical treatments focus on either lowering estrogen or increasing progesterone levels.
Surgery can remove endometriosis lesions, adhesions, and scar tissue. However, success in reducing pain symptoms and increasing pregnancy rates often depends on the extent of disease.
For infertility due to endometriosis, treatment options include laparoscopic surgical removal of endometriosis, ovarian stimulation with intrauterine insemination (IUI), and IVF. Multidisciplinary treatment addressing different symptoms and overall health often requires referral to pain experts and other specialists.9
The WHO perspective on endometriosis
Recognizing the importance of endometriosis and its impact on people’s sexual and reproductive health, quality of life, and overall well-being, the WHO is taking action to improve awareness, diagnosis, and treatment of endometriosis (TABLE 3).5 ●

Endometriosis is now recognized as a disease with significant burden for women everywhere. Widespread lack of awareness of presenting symptoms and management options means that all women’s health care clinicians need to become better informed about endometriosis so they can improve the quality of care they provide.
- Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100:818-824.
- Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
- Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
- Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
- World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room/fact-sheets/detail /endometriosis. Accessed January 3, 2022.
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382:1244-1256.
- Johnson NP, Hummelshoj L, Adamson GD, et al. World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 2017;32:315-324.
- Nnoaham K, Hummelshoj L, Webster P, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96:366-373.e8.
- Carey ET, Till SR, As-Sanie S. Pharmacological management of chronic pelvic pain in women. Drugs. 2017;77:285-301.
- Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, et al. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil Steril. 2013;100:818-824.
- Bergin K, Eliner Y, Duvall DW Jr, et al. The use of propensity score matching to assess the benefit of the endometrial receptivity analysis in frozen embryo transfers. Fertil Steril. 2021;116:396-403.
- Riestenberg C, Kroener L, Quinn M, et al. Routine endometrial receptivity array in first embryo transfer cycles does not improve live birth rate. Fertil Steril. 2021;115:1001-1006.
- Doyle N, Jahandideh S, Hill MJ, et al. A randomized controlled trial comparing live birth from single euploid frozen blastocyst transfer using standardized timing versus timing by endometrial receptivity analysis. Fertil Steril. 2021;116(suppl):e101.
- World Health Organization. Endometriosis fact sheet. March 31, 2021. https://www.who.int/news-room/fact-sheets/detail /endometriosis. Accessed January 3, 2022.
- Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382:1244-1256.
- Johnson NP, Hummelshoj L, Adamson GD, et al. World Endometriosis Society consensus on the classification of endometriosis. Hum Reprod. 2017;32:315-324.
- Nnoaham K, Hummelshoj L, Webster P, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96:366-373.e8.
- Carey ET, Till SR, As-Sanie S. Pharmacological management of chronic pelvic pain in women. Drugs. 2017;77:285-301.
Embryo mix-up debacles: Is there liability?
CASE Embryo mix-up with 2 couples
A lawsuit was recently filed in California by a couple after the woman carried and gave birth to “the wrong child.” This was the second full-term pregnancy for the couple. The couple had undergone an unsuccessful in vitro fertilization (IVF) cycle in October 2018. The next IVF cycle in 2019 led to the birth of a daughter on September 24, 2019, who is the subject of this case.1
At the time of birth, the couple suspected something was wrong because the baby had “jet-black hair and a complexion that was darker” than their complexions. The couple eventually obtained a DNA test, which confirmed in November 2019 that this was not their biological child.1
A few weeks later, they learned that another woman who went to the same IVF clinic gave birth to a female baby 1 week after their daughter was born. Similarly, that baby did not resemble the parents, and DNA testing confirmed the baby belonged to the first couple. The couples ultimately exchanged the babies.1
The legal claim filed against the IVF center and its owner (an obstetrician) was for breach of contract, medical malpractice, and infliction of emotional distress, including experiencing “disassociation” on the part of the couple(s). Each couple felt they did not get to experience the birth of their biological child, and, of course there was considerable distress in the process of learning that the child was not theirs and exchanging the birth child for the biological child. In addition, the couple who filed the suit had another child (now age 7 years), who begged them to keep the baby to whom they gave birth. The couple also reported experiencing panic attacks as a result of the events.1
Medical considerations
As of 2018, more than 8 million IVF babies had been born, with the first in 1978 in the United Kingdom.2 Advances in science and technology have improved the process. Storage tanks now have alarms and several safeguards to monitor the level of liquid nitrogen and immediately notify key personnel if levels are low (FIGURES 1 and 2). Preimplantation genetic testing is also readily available to assess the embryo prior to transfer into the uterus and identify various genetic problems.


Guidelines for embryo straw labelling are provided by the College of American Pathologists and the Centers for Disease Control and Prevention. The American Society for Reproductive Medicine (ASRM) also provides guidelines. When an error occurs, disclosure is recommended and ethical and legal counsel should be involved. Failing to disclose can lead to professional penalties.4
Unfortunately, despite these advances and guidelines, embryo mix-ups like the one in the above case do occur and receive public notice (See “Cross country embryo mix-up cases”).5,6 A report from the University of Nevada assessed liability for embryo mix-ups in US fertility practices from 2000 to 2020.7 They evaluated 184,015 IVF cycles with 176 claims. Payments were made to plaintiffs in 21 cases, resulting in $15 million of awarded damages (average award was $199,188).7 The most common problem was in the embryology laboratory with an overall incidence of 0.03% of the total number of IVF cycles.7 To avoid damages, the authors emphasized the importance of following labeling guidelines when storing embryos, considering a 2-step read-back method prior to embryo transfer, and offering genetic testing when a discrepancy is noted in the record (TABLE).7

Other medical liability considerations
Embryo mix-ups are not the only source of problems and potential liability in IVF. At the 2021 Association of Sexual and Reproductive Medicine Annual Meeting, Applebaum et al presented results from a comprehensive review of malpractice litigation involving IVF in the United States.8 Using the legal database NEXIS Uni they identified 50 cases between 1986 and 2020 (32% of which were filed in New York state). Common thematic elements among patient allegations were embryology errors (eg, lost or destroyed embryos or incorrect sperm or egg donor), errors in preimplantation genetics, surgical or medical errors/complications, or misdiagnosis (eg, sexually transmitted disease screening or malignancy).8 Overall, the most common plaintiff complaint was negligence (26 cases) due to informed consent–related issues (9 cases), wrongful life or birth (9 cases), or negligent or intentional infliction of emotional distress (5 cases).8
In 48% of cases, the verdict was in favor of the defendant; it was for the plaintiff in 36% of cases and ongoing proceedings or partial judgement accounted for the remaining cases.8 Damages ranged from $4,171.45 to $50 million. The authors emphasized specific defense strategies, including the importance of careful labeling and handling of embryos, prompt disclosure when an error does occur, and awareness of the specific state statute(s) of limitations for medical malpractice claims.8
Continue to: Legal considerations...
Legal considerations
The case at the beginning of this article is a “mix-up” case, in which an IVF center implanted the wrong embryo, resulting in the birth parents not being the biological parents.1 As in that case, there may be (but are not always)6,9 2 mix-ups, so that 2 couples have each other’s biological children. These cases may go unnoticed by the birth parent if the physical appearance is not unexpected and the parents never do genetic testing, or if the IVF center does not discover the error and inform the parents. Infrequently the cases make the news or the courts.10,11
News accounts are not trials, and we do not suggest that all the facts discussed in news reports on the case described here are complete—or even accurate in the details reported. They are generally 1-sided, so there are other perspectives. To consider the legal issues, however, we will assume for discussion only that the facts are as they have been reported in the news coverage—with the understanding that the discovery and trial processes would undoubtedly bring to light many other important facts or corrections.
Negligence
Although there are several potential bases for liability (ie, contract or warranty claims, a form of product liability/defect) in mix-up and other artificial reproductive technology (ART), negligence or malpractice seem most likely.12 “Negligence” here is intended to be simple negligence but may also include gross negligence or recklessness.
Although the incidence of errors in ART is unknown, there is limited evidence that suggests it is not a rare event. One study suggested >20% of fertility clinics knew of errors in processing or handling donor samples and embryos for implantation.13 Another study in the United Kingdom found that 1 in 1,000 IVF embryos were implanted in the wrong woman.14
Was there negligence? The first question in a malpractice or negligence-type action is, was there a professional relationship between the plaintiff who is claiming harm and the professional or organization defendant? The next question is whether the defendant was reasonably careful given the circumstances—that is, did the physician meet the “standard of care”? This is sometimes described as whether the professional’s actions would be acceptable (ie, reasonably prudent within the profession or specialty). If there was negligence, then the next question is, did that negligence cause an injury to the plaintiff?15
Determining the standard of care. The nature of the expected standard of care is dependent, in part, on the potential consequences of an error. For example, the care required when there is a significant risk of death from an error would be considerably more cautious than for an error that might result in small property damage. In this case study, a mix-up error is likely to be less severe than death, but is very substantial in terms of emotional harm and disruption. Thus, considerable care and attention would be expected to avoid these errors. They should be a “never” event. Institutions and physicians should give considerable attention to their processes and procedures to avoid the possibility of a mix-up error.16
Where did the negligence occur? There is an old tort doctrine “Res ipsa loquitor” (RIL) that means, “The thing speaks for itself.” Although there are several technical rules around the application of RIL as a presumption of negligence, it comes down to the proposition that some injuries do not occur without negligence. A traditional medical example is the sponge left in a patient during surgery—ordinarily that does not happen without some negligence. For RIL to be applied, usually the mechanism by which the injury occurred had to be under the control of the defendant (or the agents of the defendant).
The “mix-up” of embryos is an example of the kind of error that would not likely occur without negligence.17 But the embryo may not be in the exclusive control of any 1 institution. For example, the mistake could be made by the IVF center (or its employees), a separate facility that has processed or cryogenically stored the genetic materials, and independent physicians (not employees or agents of the center). Therefore, it is necessary to pinpoint where the negligence occurred and who is legally responsible. In some cases, a health care provider must take steps to ensure that its contractors have sufficient safeguards to avoid unnecessary harms. For example, an IVF center that uses an external cryogenic storage facility may have some obligation to know that the genetic material returned to the center is the same material that the center provided the storage facility in the first place and is properly identified.18
Assessing damages
From the facts as we have them, it appears that there must have been negligence that caused the mix-up of the embryos in the original case. It also appears reasonably clear that the negligence resulted in harm to both sets of parents and their families. This would suggest that the families should recover substantial damages. But that, somewhat surprisingly, may not be the case.19 Several legal principles may limit the availability or size of damages in mix-up cases. Also, it is worth remembering that there are differences in how states treat the different types of damages in these cases. Although the case was filed in California, we’ll take a more national view of the damages issue.
Not all harm is treated as equal. The first problem facing plaintiffs in mix-up cases may be the fact that they have suffered only emotional harm, without any physical injury. Traditionally, the courts have been reluctant to allow recovery in negligence for purely emotional injuries. Also “intentional” infliction of emotional distress does permit financial recovery, but generally “negligent” infliction of emotional harm traditionally has not. In part, this was because of the fear of unwarranted (and difficult-to-assess) claims of emotional harm that are not related to a physical harm. Some states developed a “zone of danger” exception (eg, where someone was almost hit by a car) or allowed some emotional injury recovery if there were “physical manifestations” of the emotional harm. In short, depending on the state’s rules, negligence that causes purely emotional harm may not be compensable.20
State-based malpractice “caps.” Another limitation on emotional injuries is the “caps” on malpractice damages enacted by several states (including California, where this mix-up case occurred). Therefore, if a mix-up case is determined to be a malpractice case under state law, emotional suffering damages (which are non-economic damages) may be limited to the cap—$250,000 in California, for example—even if the state allows damages for emotional injuries without physical injuries.
The rare exception. Very careless labeling or handling of the identity of the embryo could at the extreme be considered gross negligence or recklessness. There are relatively inexpensive and easy procedures that could easily avoid what is likely to be significant harm to families (including emotional upset).21 Institutions that callously fail to use those procedures might be seen by some courts as reckless, or in outrageous cases, even intentional. An example would be the University of California Irvine Center for Reproductive Health case, in which physicians intentionally (without consent) used patients’ ova, fertilized them, and then implanted them in other patients, with at least 15 births, many lawsuits, and multimillion dollar settlements.22 In “intentional” cases, limitations on emotional injuries would usually not be major barriers to recovery of damages. However, those are legal stretches, and recovery is the exception rather than the rule.23
Continue to: Additional legal concerns with IVF...
Additional legal concerns with IVF
Reproduction negligence cases include a large range of errors and injuries—not just embryo mix-ups. Courts have struggled with when it is appropriate to allow damages, even when there have been clear injuries. For the most part courts have been reluctant to find liability in many areas of new IVF technology.12 One problem in determining how to assess damages is determining how incidental benefits should be used to offset some or all of the damages. For example, how should the joy of having a child offset the costs of raising the child?
There are more than a dozen kinds of current and likely future claims arising from problems with ART. It is tempting to conclude, “Oh, what a tangled legal web we weave when first we practice to artificially conceive.” There are various groupings of such claims, with several examples of cases presented in this article. It is not possible to consider those in detail in this article. As a general proposition, however, “our legal system treats wrongfully disrupted plans concerning reproduction like one of those life adversities that people are expected to abide without remedy.”24
This is not to say, however, that there is no compensation for IVF-related injuries. Applebaum and colleagues found more than 100 cases in the 35 years covered by the study (1984-2020).8 However, only 50 of those cases fit the criteria for inclusion in their data. The successful cases for the plaintiffs involved medical or surgical error, while it appeared that various forms of wrongful life or birth were much less successful. It would be a mistake to conclude from these data that there are not, and will not be, meaningful risks of liability in the areas of IVF and ART more generally.
First, claims that fit with existing legal doctrine are producing liability. About half of the claims (25 over the 34 years) examined by Applebaum et al resulted in liability. Admittedly, that number was small because ART use was increasing. Where the claims fit well-recognized legal forms of damages and forms of action (primarily negligence), the liability could be substantial. A remarkable example of this is the case of Wuth v Lab. Corp (see “Liability for genetic testing errors”),25 which was the largest verdict ($50 million) in the Applebaum and colleagues’ study.8 The large verdict was due to the failure of the testing company and a medical center to properly perform and assess a genetic test, which resulted in the birth of a child with an unbalanced chromosome translocation.8,25 The child’s serious disabilities would require a great deal of expensive care. Although the jury held the testing laboratory and medical center liable, they did not find liability against the physician.25 Ultimately, this case would be considered a failure of genetic testing rather than an IVF case.
More than 2 couples
In a second case from California, a couples’ son was born to another couple in New York—along with another boy from a third couple. The woman in New York thought she had carried biological twins but genetic testing confirmed the twins were not related to the couple or to each other (the second couple filed a separate medical malpractice and negligence lawsuit in New York). All 3 couples had sought care at the same IVF clinic. The babies were eventually returned to their biological parents.1
Different races
In a New York case, a Korean couple had twin White boys after consenting to a single embryo transfer. Meanwhile a couple in Los Angeles who went to the same in vitro fertilization clinic gave birth to a child that did not match their appearance. Both couples had undergone embryo transfers on the same day. The court arranged for the Korean couple to surrender their twins to their biological parents when they were 6 months of age in exchange for their biological child.2
References
1. Couple claims clinic implanted their embryo in wrong woman. Associated Press. July 10, 2019. https://apnews.com/article/de32d537c6e34808b28834c23f00e272. Accessed January 6, 2022.
2. In the matter of accusation against Steven L. Katz. Case no. 03-20001-122617.OAH no. N2004080093. Sacramento, CA. Medical Board of California Department of Consumer Affairs 2005.
Future challenges
The future is likely to bring substantially expanded IVF/ART liability for several reasons. ART is becoming more common. Although courts have struggled with how to apply existing liability rules to the new technologies and related novel legal claims, the absence of established legal principles into which IVF injuries fit will not last forever. The legal system eventually finds ways of adjusting old rules or adopting new ones to cover injuries from new technology.
Although IVF injuries that most people feel deserve compensation currently are not cognizable in law, that will undoubtedly change. Either the courts will find new ways of assessing ART claims, or state legislatures and Congress will step in with legislation. To date, Congress has been relatively “hands off” on the ART processes, with the Fertility Clinic Success Rate and Certification Act of 1992 being a notable exception.24 This law requires ART programs to report success rates and directs the Centers for Disease Control and Prevention (CDC) to publish reported success rates and laboratory incidents. It also establishes a model state laboratory certification program.24 The CDC has an outline of the work under the statute,26 as well as state-specific data regarding ART27 and lists of publications in key areas.28 In addition there are various state laws related to recordkeeping, donor qualifications, licensing, and family law issues.29 Ultimately, physicians, scientists, and legal professionals can perform a valuable role in helping to fashion IVF liability principles that are workable and reasonable, that will not interfere with the progress of medicine, and that will ensure that those injured through carelessness or bad medicine receive compensation. ●
Although not technically an in vitro fertilization (IVF) case, Wuth v Lab. Corp. involved an infant born through IVF with a translocation defect chromosome 2 (ie, deleted material) and extra chromatin on 9. The father’s family history included birth defects, including a female cousin with profound developmental disabilities, seizures, and antisocial behavior. He had undergone genetic testing that revealed an asymptomatic balanced, 2;9 translocation. As part of the IVF process, the couple had a genetic consultation and were told there was a 50% chance that the fetus would have an unbalanced 2;9 translocation given the father’s family history and that chorionic villus sampling or amniocentesis could detect this in the fetus.1
Amniocentesis had been performed, with the specimen sent to Lab. Corp. The result was “normal male karyotype.” However, when the baby was born, it was immediately apparent that he had severe physical defects and subsequently cognitive defects. Genetic testing of the child revealed an unbalanced 2;9 translocation. The couple filed a suit for wrongful birth and wrongful life, which went to a jury. The child was awarded $25 million and the parents/family were awarded another $25 million in general damages. The verdict reflected errors in genetic (laboratory) testing.
Reference
1. Wuth v Lab. Corp. of Am., 189 Wash. App. 660, 359 P.3d 841 (2015).
- Mark J. California couple sues fertility clinic following IVF embryo mix-up. Washington Post. November 9, 2021. https://www.washingtonpost.com/nation/2021/11/09/in-vitro-fertilization-ivf-mix-up-daphna-cardinale. Accessed January 5, 2022.
- More than 8 million babies born from IVF since the world’s first in 1978. Science Daily. July 3, 2018. https://www.sciencedaily.com/releases/2018/07/180703084127.htm. Accessed January 11, 2022.
- ESCO Medical. In vitro fertilization (IVF) as fertility treatment. https://www.esco-medical.com/resource/in-vitro-fertilization-ivf-as-fertility-treatment.
- Vigdor N. “We had their baby, and they had our baby”: couple sues over embryo “mix-up.” NY Times. November 9, 2021. https://www.nytimes.com/2021/11/09/us/fertility-clinic-embryo-mixup.html. Accessed January 11, 2022.
- Couple claims clinic implanted their embryo in wrong woman. Associated Press. July 10, 2019. https://apnews.com/article/de32d537c6e34808b28834c23f00e2728. Accessed January 6, 2022.
- In the matter of accusation against Steven L. Katz. Case no. 03-20001-122617.OAH no. N2004080093. Sacramento, CA. Medical Board of California Department of Consumer Affairs 2005
- Rasouli MA, Moutos CP, Phelps JY. Liability for embryo mix-ups in fertility practices in the USA. J Assist Reprod Genet. 2021;38:1101-1107. doi:10.1007/s10815-021-02108-1
- Applebaum J, Berger D, O’Neill K. Can a reproductive endocrinologist be sued for 50 million dollars? A comprehensive review of malpractice litigation involving in vitro fertilization in the U.S. Fertil Steril. 2021;116(3s):e19. doi:10.1016/j.fertnstert.2021.07.059
- Andrews v Keltz, 838 N.Y.S.2d 363, 365 (Sup. Ct. 2007).
- Chichi DV. In vitro fertilization, fertility frustrations, and the lack of regulation. Hofstra L Rev. 2021;49:535-568. https://www.hofstralawreview.org/wp-content/uploads/2021/04/bb.2.chichi.pdf. Accessed January 11, 2022.
- Lewin T. Sperm banks accused of losing samples and lying about donors. NY Times. July 21, 2016. https://www.nytimes.com/2016/07/22/us/sperm-banks-accused-of-losing-samples-and-lying-about-donors.html. Accessed January 11, 2022.
- Bender L. To err is human ART mix-ups: labor-based, relational proposal. J Gender Race Justice. 2006;9:443-508. https://surface.syr.edu/cgi/viewcontent.cgi?article=1050&context=lawpub. Accessed January 11, 2022.
- Baruch S, Kaufman D, Hudson KL. Genetic testing of embryos: practices and perspectives of U.S. in vitro fertilization clinics. Fertil Steril. 2007;89:1053-1058. doi:10.1016/j.fertnstert.2007.05.048
- Liebler R. Are you my parent? Are you my child? The role of genetics and race in defining relationships after reproductive technological mistakes. DePaul J Health Care Law. 2002;5:15-56. https://via.library.depaul.edu/cgi/viewcontent.cgi?article=1202&context=jhcl. Accessed January 11, 2022.
- Crockin SL, Altman AB, Edmonds MA. The history and future trends of art medicine and law. Fam Court Rev. 2021;59:22-45. doi:10.1111/fcre.12550
- Fernandes JS. Perfecting pregnancy via preimplantation genetic screening: the quest for an elusive standard of care. UC Irvine L Rev. 2014;4:1295-1326. https://www.law.uci.edu/lawreview/vol4/no4/Fernandes.pdf. Accessed January 11, 2022.
- VanGessel MM. Wrongful surrogacy: the need for right of action in cases of clear negligence. U Toledo L Rev. 2015;46:681-706.
- Reich J, Swink D. Outsourcing human reproduction: embryos and surrogacy services in the cyberprocreation era. J Health Care L Policy. 2011;14:241-298. https://core.ac.uk/download/pdf/217156567.pdf. Accessed January 11, 2022.
- Strasser M. Prenatal tort slippage. Health Matrix. 2021;31:221-262. https://scholarlycommons.law.case.edu/healthmatrix/vol31/iss1/9. Accessed January 11, 2022.
- Heide IH. Negligence in the creation of healthy babies: negligent infliction of emotional distress in cases of alternative reproductive technology malpractice without physical injury. J Med L. 2005;9:55-94.
- Novo S, Nogués C, Penon O, et al. Barcode tagging of human oocytes and embryos to prevent mix-ups in assisted reproduction technologies. Hum Reprod. 2014;29:18-28. doi: 10.1093/humrep/det409
- Yoshino K. UCI Settles Dozens of Fertility Suits. LA Times. September 11, 2009. https://www.latimes.com/archives/la-xpm-2009-sep-11-me-uci-fertility11-story.html. Accessed January 11, 2022.
- Fox D. Reproductive negligence. Columbia L Rev. 2017;117:149-242. https://columbialawreview.org/wp-content/uploads/2017/01/149.pdf. Accessed January 11, 2022.
- 42 U.S.C.S. §263a-1-263a-7; Public Law 102-493. https://www.govinfo.gov/content/pkg/STATUTE-106/pdf/STATUTE-106-Pg3146.pdf. Accessed January 11, 2022.
- Wuth v Lab. Corp. of Am., 189 Wash. App. 660, 359 P.3d 841 (2015).
- Centers for Disease Control and Prevention. The Fertility Clinic Success Rate and Certification Act. December 14, 2020. https://www.cdc.gov/art/nass/policy.html#act. Accessed January 11, 2022.
- Centers for Disease Control and Prevention. State-specific assisted reproductive technology surveillance. December 17, 2020. https://www.cdc.gov/art/state-specific-surveillance/index.html. Accessed January 11, 2022.
- Centers for Disease Control and Prevention. Key findings. March 12, 2021. https://www.cdc.gov/art/key-findings/index.html. Accessed January 11, 2022.
- Cohen EN. 5 Treatise on Health Care Law §22.04, (ed. Hooper, Lundy & Bookman, & Robert W. Lundy, Jr. RW.) (Matthew Bender-LexisNexis)
CASE Embryo mix-up with 2 couples
A lawsuit was recently filed in California by a couple after the woman carried and gave birth to “the wrong child.” This was the second full-term pregnancy for the couple. The couple had undergone an unsuccessful in vitro fertilization (IVF) cycle in October 2018. The next IVF cycle in 2019 led to the birth of a daughter on September 24, 2019, who is the subject of this case.1
At the time of birth, the couple suspected something was wrong because the baby had “jet-black hair and a complexion that was darker” than their complexions. The couple eventually obtained a DNA test, which confirmed in November 2019 that this was not their biological child.1
A few weeks later, they learned that another woman who went to the same IVF clinic gave birth to a female baby 1 week after their daughter was born. Similarly, that baby did not resemble the parents, and DNA testing confirmed the baby belonged to the first couple. The couples ultimately exchanged the babies.1
The legal claim filed against the IVF center and its owner (an obstetrician) was for breach of contract, medical malpractice, and infliction of emotional distress, including experiencing “disassociation” on the part of the couple(s). Each couple felt they did not get to experience the birth of their biological child, and, of course there was considerable distress in the process of learning that the child was not theirs and exchanging the birth child for the biological child. In addition, the couple who filed the suit had another child (now age 7 years), who begged them to keep the baby to whom they gave birth. The couple also reported experiencing panic attacks as a result of the events.1
Medical considerations
As of 2018, more than 8 million IVF babies had been born, with the first in 1978 in the United Kingdom.2 Advances in science and technology have improved the process. Storage tanks now have alarms and several safeguards to monitor the level of liquid nitrogen and immediately notify key personnel if levels are low (FIGURES 1 and 2). Preimplantation genetic testing is also readily available to assess the embryo prior to transfer into the uterus and identify various genetic problems.


Guidelines for embryo straw labelling are provided by the College of American Pathologists and the Centers for Disease Control and Prevention. The American Society for Reproductive Medicine (ASRM) also provides guidelines. When an error occurs, disclosure is recommended and ethical and legal counsel should be involved. Failing to disclose can lead to professional penalties.4
Unfortunately, despite these advances and guidelines, embryo mix-ups like the one in the above case do occur and receive public notice (See “Cross country embryo mix-up cases”).5,6 A report from the University of Nevada assessed liability for embryo mix-ups in US fertility practices from 2000 to 2020.7 They evaluated 184,015 IVF cycles with 176 claims. Payments were made to plaintiffs in 21 cases, resulting in $15 million of awarded damages (average award was $199,188).7 The most common problem was in the embryology laboratory with an overall incidence of 0.03% of the total number of IVF cycles.7 To avoid damages, the authors emphasized the importance of following labeling guidelines when storing embryos, considering a 2-step read-back method prior to embryo transfer, and offering genetic testing when a discrepancy is noted in the record (TABLE).7

Other medical liability considerations
Embryo mix-ups are not the only source of problems and potential liability in IVF. At the 2021 Association of Sexual and Reproductive Medicine Annual Meeting, Applebaum et al presented results from a comprehensive review of malpractice litigation involving IVF in the United States.8 Using the legal database NEXIS Uni they identified 50 cases between 1986 and 2020 (32% of which were filed in New York state). Common thematic elements among patient allegations were embryology errors (eg, lost or destroyed embryos or incorrect sperm or egg donor), errors in preimplantation genetics, surgical or medical errors/complications, or misdiagnosis (eg, sexually transmitted disease screening or malignancy).8 Overall, the most common plaintiff complaint was negligence (26 cases) due to informed consent–related issues (9 cases), wrongful life or birth (9 cases), or negligent or intentional infliction of emotional distress (5 cases).8
In 48% of cases, the verdict was in favor of the defendant; it was for the plaintiff in 36% of cases and ongoing proceedings or partial judgement accounted for the remaining cases.8 Damages ranged from $4,171.45 to $50 million. The authors emphasized specific defense strategies, including the importance of careful labeling and handling of embryos, prompt disclosure when an error does occur, and awareness of the specific state statute(s) of limitations for medical malpractice claims.8
Continue to: Legal considerations...
Legal considerations
The case at the beginning of this article is a “mix-up” case, in which an IVF center implanted the wrong embryo, resulting in the birth parents not being the biological parents.1 As in that case, there may be (but are not always)6,9 2 mix-ups, so that 2 couples have each other’s biological children. These cases may go unnoticed by the birth parent if the physical appearance is not unexpected and the parents never do genetic testing, or if the IVF center does not discover the error and inform the parents. Infrequently the cases make the news or the courts.10,11
News accounts are not trials, and we do not suggest that all the facts discussed in news reports on the case described here are complete—or even accurate in the details reported. They are generally 1-sided, so there are other perspectives. To consider the legal issues, however, we will assume for discussion only that the facts are as they have been reported in the news coverage—with the understanding that the discovery and trial processes would undoubtedly bring to light many other important facts or corrections.
Negligence
Although there are several potential bases for liability (ie, contract or warranty claims, a form of product liability/defect) in mix-up and other artificial reproductive technology (ART), negligence or malpractice seem most likely.12 “Negligence” here is intended to be simple negligence but may also include gross negligence or recklessness.
Although the incidence of errors in ART is unknown, there is limited evidence that suggests it is not a rare event. One study suggested >20% of fertility clinics knew of errors in processing or handling donor samples and embryos for implantation.13 Another study in the United Kingdom found that 1 in 1,000 IVF embryos were implanted in the wrong woman.14
Was there negligence? The first question in a malpractice or negligence-type action is, was there a professional relationship between the plaintiff who is claiming harm and the professional or organization defendant? The next question is whether the defendant was reasonably careful given the circumstances—that is, did the physician meet the “standard of care”? This is sometimes described as whether the professional’s actions would be acceptable (ie, reasonably prudent within the profession or specialty). If there was negligence, then the next question is, did that negligence cause an injury to the plaintiff?15
Determining the standard of care. The nature of the expected standard of care is dependent, in part, on the potential consequences of an error. For example, the care required when there is a significant risk of death from an error would be considerably more cautious than for an error that might result in small property damage. In this case study, a mix-up error is likely to be less severe than death, but is very substantial in terms of emotional harm and disruption. Thus, considerable care and attention would be expected to avoid these errors. They should be a “never” event. Institutions and physicians should give considerable attention to their processes and procedures to avoid the possibility of a mix-up error.16
Where did the negligence occur? There is an old tort doctrine “Res ipsa loquitor” (RIL) that means, “The thing speaks for itself.” Although there are several technical rules around the application of RIL as a presumption of negligence, it comes down to the proposition that some injuries do not occur without negligence. A traditional medical example is the sponge left in a patient during surgery—ordinarily that does not happen without some negligence. For RIL to be applied, usually the mechanism by which the injury occurred had to be under the control of the defendant (or the agents of the defendant).
The “mix-up” of embryos is an example of the kind of error that would not likely occur without negligence.17 But the embryo may not be in the exclusive control of any 1 institution. For example, the mistake could be made by the IVF center (or its employees), a separate facility that has processed or cryogenically stored the genetic materials, and independent physicians (not employees or agents of the center). Therefore, it is necessary to pinpoint where the negligence occurred and who is legally responsible. In some cases, a health care provider must take steps to ensure that its contractors have sufficient safeguards to avoid unnecessary harms. For example, an IVF center that uses an external cryogenic storage facility may have some obligation to know that the genetic material returned to the center is the same material that the center provided the storage facility in the first place and is properly identified.18
Assessing damages
From the facts as we have them, it appears that there must have been negligence that caused the mix-up of the embryos in the original case. It also appears reasonably clear that the negligence resulted in harm to both sets of parents and their families. This would suggest that the families should recover substantial damages. But that, somewhat surprisingly, may not be the case.19 Several legal principles may limit the availability or size of damages in mix-up cases. Also, it is worth remembering that there are differences in how states treat the different types of damages in these cases. Although the case was filed in California, we’ll take a more national view of the damages issue.
Not all harm is treated as equal. The first problem facing plaintiffs in mix-up cases may be the fact that they have suffered only emotional harm, without any physical injury. Traditionally, the courts have been reluctant to allow recovery in negligence for purely emotional injuries. Also “intentional” infliction of emotional distress does permit financial recovery, but generally “negligent” infliction of emotional harm traditionally has not. In part, this was because of the fear of unwarranted (and difficult-to-assess) claims of emotional harm that are not related to a physical harm. Some states developed a “zone of danger” exception (eg, where someone was almost hit by a car) or allowed some emotional injury recovery if there were “physical manifestations” of the emotional harm. In short, depending on the state’s rules, negligence that causes purely emotional harm may not be compensable.20
State-based malpractice “caps.” Another limitation on emotional injuries is the “caps” on malpractice damages enacted by several states (including California, where this mix-up case occurred). Therefore, if a mix-up case is determined to be a malpractice case under state law, emotional suffering damages (which are non-economic damages) may be limited to the cap—$250,000 in California, for example—even if the state allows damages for emotional injuries without physical injuries.
The rare exception. Very careless labeling or handling of the identity of the embryo could at the extreme be considered gross negligence or recklessness. There are relatively inexpensive and easy procedures that could easily avoid what is likely to be significant harm to families (including emotional upset).21 Institutions that callously fail to use those procedures might be seen by some courts as reckless, or in outrageous cases, even intentional. An example would be the University of California Irvine Center for Reproductive Health case, in which physicians intentionally (without consent) used patients’ ova, fertilized them, and then implanted them in other patients, with at least 15 births, many lawsuits, and multimillion dollar settlements.22 In “intentional” cases, limitations on emotional injuries would usually not be major barriers to recovery of damages. However, those are legal stretches, and recovery is the exception rather than the rule.23
Continue to: Additional legal concerns with IVF...
Additional legal concerns with IVF
Reproduction negligence cases include a large range of errors and injuries—not just embryo mix-ups. Courts have struggled with when it is appropriate to allow damages, even when there have been clear injuries. For the most part courts have been reluctant to find liability in many areas of new IVF technology.12 One problem in determining how to assess damages is determining how incidental benefits should be used to offset some or all of the damages. For example, how should the joy of having a child offset the costs of raising the child?
There are more than a dozen kinds of current and likely future claims arising from problems with ART. It is tempting to conclude, “Oh, what a tangled legal web we weave when first we practice to artificially conceive.” There are various groupings of such claims, with several examples of cases presented in this article. It is not possible to consider those in detail in this article. As a general proposition, however, “our legal system treats wrongfully disrupted plans concerning reproduction like one of those life adversities that people are expected to abide without remedy.”24
This is not to say, however, that there is no compensation for IVF-related injuries. Applebaum and colleagues found more than 100 cases in the 35 years covered by the study (1984-2020).8 However, only 50 of those cases fit the criteria for inclusion in their data. The successful cases for the plaintiffs involved medical or surgical error, while it appeared that various forms of wrongful life or birth were much less successful. It would be a mistake to conclude from these data that there are not, and will not be, meaningful risks of liability in the areas of IVF and ART more generally.
First, claims that fit with existing legal doctrine are producing liability. About half of the claims (25 over the 34 years) examined by Applebaum et al resulted in liability. Admittedly, that number was small because ART use was increasing. Where the claims fit well-recognized legal forms of damages and forms of action (primarily negligence), the liability could be substantial. A remarkable example of this is the case of Wuth v Lab. Corp (see “Liability for genetic testing errors”),25 which was the largest verdict ($50 million) in the Applebaum and colleagues’ study.8 The large verdict was due to the failure of the testing company and a medical center to properly perform and assess a genetic test, which resulted in the birth of a child with an unbalanced chromosome translocation.8,25 The child’s serious disabilities would require a great deal of expensive care. Although the jury held the testing laboratory and medical center liable, they did not find liability against the physician.25 Ultimately, this case would be considered a failure of genetic testing rather than an IVF case.
More than 2 couples
In a second case from California, a couples’ son was born to another couple in New York—along with another boy from a third couple. The woman in New York thought she had carried biological twins but genetic testing confirmed the twins were not related to the couple or to each other (the second couple filed a separate medical malpractice and negligence lawsuit in New York). All 3 couples had sought care at the same IVF clinic. The babies were eventually returned to their biological parents.1
Different races
In a New York case, a Korean couple had twin White boys after consenting to a single embryo transfer. Meanwhile a couple in Los Angeles who went to the same in vitro fertilization clinic gave birth to a child that did not match their appearance. Both couples had undergone embryo transfers on the same day. The court arranged for the Korean couple to surrender their twins to their biological parents when they were 6 months of age in exchange for their biological child.2
References
1. Couple claims clinic implanted their embryo in wrong woman. Associated Press. July 10, 2019. https://apnews.com/article/de32d537c6e34808b28834c23f00e272. Accessed January 6, 2022.
2. In the matter of accusation against Steven L. Katz. Case no. 03-20001-122617.OAH no. N2004080093. Sacramento, CA. Medical Board of California Department of Consumer Affairs 2005.
Future challenges
The future is likely to bring substantially expanded IVF/ART liability for several reasons. ART is becoming more common. Although courts have struggled with how to apply existing liability rules to the new technologies and related novel legal claims, the absence of established legal principles into which IVF injuries fit will not last forever. The legal system eventually finds ways of adjusting old rules or adopting new ones to cover injuries from new technology.
Although IVF injuries that most people feel deserve compensation currently are not cognizable in law, that will undoubtedly change. Either the courts will find new ways of assessing ART claims, or state legislatures and Congress will step in with legislation. To date, Congress has been relatively “hands off” on the ART processes, with the Fertility Clinic Success Rate and Certification Act of 1992 being a notable exception.24 This law requires ART programs to report success rates and directs the Centers for Disease Control and Prevention (CDC) to publish reported success rates and laboratory incidents. It also establishes a model state laboratory certification program.24 The CDC has an outline of the work under the statute,26 as well as state-specific data regarding ART27 and lists of publications in key areas.28 In addition there are various state laws related to recordkeeping, donor qualifications, licensing, and family law issues.29 Ultimately, physicians, scientists, and legal professionals can perform a valuable role in helping to fashion IVF liability principles that are workable and reasonable, that will not interfere with the progress of medicine, and that will ensure that those injured through carelessness or bad medicine receive compensation. ●
Although not technically an in vitro fertilization (IVF) case, Wuth v Lab. Corp. involved an infant born through IVF with a translocation defect chromosome 2 (ie, deleted material) and extra chromatin on 9. The father’s family history included birth defects, including a female cousin with profound developmental disabilities, seizures, and antisocial behavior. He had undergone genetic testing that revealed an asymptomatic balanced, 2;9 translocation. As part of the IVF process, the couple had a genetic consultation and were told there was a 50% chance that the fetus would have an unbalanced 2;9 translocation given the father’s family history and that chorionic villus sampling or amniocentesis could detect this in the fetus.1
Amniocentesis had been performed, with the specimen sent to Lab. Corp. The result was “normal male karyotype.” However, when the baby was born, it was immediately apparent that he had severe physical defects and subsequently cognitive defects. Genetic testing of the child revealed an unbalanced 2;9 translocation. The couple filed a suit for wrongful birth and wrongful life, which went to a jury. The child was awarded $25 million and the parents/family were awarded another $25 million in general damages. The verdict reflected errors in genetic (laboratory) testing.
Reference
1. Wuth v Lab. Corp. of Am., 189 Wash. App. 660, 359 P.3d 841 (2015).
CASE Embryo mix-up with 2 couples
A lawsuit was recently filed in California by a couple after the woman carried and gave birth to “the wrong child.” This was the second full-term pregnancy for the couple. The couple had undergone an unsuccessful in vitro fertilization (IVF) cycle in October 2018. The next IVF cycle in 2019 led to the birth of a daughter on September 24, 2019, who is the subject of this case.1
At the time of birth, the couple suspected something was wrong because the baby had “jet-black hair and a complexion that was darker” than their complexions. The couple eventually obtained a DNA test, which confirmed in November 2019 that this was not their biological child.1
A few weeks later, they learned that another woman who went to the same IVF clinic gave birth to a female baby 1 week after their daughter was born. Similarly, that baby did not resemble the parents, and DNA testing confirmed the baby belonged to the first couple. The couples ultimately exchanged the babies.1
The legal claim filed against the IVF center and its owner (an obstetrician) was for breach of contract, medical malpractice, and infliction of emotional distress, including experiencing “disassociation” on the part of the couple(s). Each couple felt they did not get to experience the birth of their biological child, and, of course there was considerable distress in the process of learning that the child was not theirs and exchanging the birth child for the biological child. In addition, the couple who filed the suit had another child (now age 7 years), who begged them to keep the baby to whom they gave birth. The couple also reported experiencing panic attacks as a result of the events.1
Medical considerations
As of 2018, more than 8 million IVF babies had been born, with the first in 1978 in the United Kingdom.2 Advances in science and technology have improved the process. Storage tanks now have alarms and several safeguards to monitor the level of liquid nitrogen and immediately notify key personnel if levels are low (FIGURES 1 and 2). Preimplantation genetic testing is also readily available to assess the embryo prior to transfer into the uterus and identify various genetic problems.


Guidelines for embryo straw labelling are provided by the College of American Pathologists and the Centers for Disease Control and Prevention. The American Society for Reproductive Medicine (ASRM) also provides guidelines. When an error occurs, disclosure is recommended and ethical and legal counsel should be involved. Failing to disclose can lead to professional penalties.4
Unfortunately, despite these advances and guidelines, embryo mix-ups like the one in the above case do occur and receive public notice (See “Cross country embryo mix-up cases”).5,6 A report from the University of Nevada assessed liability for embryo mix-ups in US fertility practices from 2000 to 2020.7 They evaluated 184,015 IVF cycles with 176 claims. Payments were made to plaintiffs in 21 cases, resulting in $15 million of awarded damages (average award was $199,188).7 The most common problem was in the embryology laboratory with an overall incidence of 0.03% of the total number of IVF cycles.7 To avoid damages, the authors emphasized the importance of following labeling guidelines when storing embryos, considering a 2-step read-back method prior to embryo transfer, and offering genetic testing when a discrepancy is noted in the record (TABLE).7

Other medical liability considerations
Embryo mix-ups are not the only source of problems and potential liability in IVF. At the 2021 Association of Sexual and Reproductive Medicine Annual Meeting, Applebaum et al presented results from a comprehensive review of malpractice litigation involving IVF in the United States.8 Using the legal database NEXIS Uni they identified 50 cases between 1986 and 2020 (32% of which were filed in New York state). Common thematic elements among patient allegations were embryology errors (eg, lost or destroyed embryos or incorrect sperm or egg donor), errors in preimplantation genetics, surgical or medical errors/complications, or misdiagnosis (eg, sexually transmitted disease screening or malignancy).8 Overall, the most common plaintiff complaint was negligence (26 cases) due to informed consent–related issues (9 cases), wrongful life or birth (9 cases), or negligent or intentional infliction of emotional distress (5 cases).8
In 48% of cases, the verdict was in favor of the defendant; it was for the plaintiff in 36% of cases and ongoing proceedings or partial judgement accounted for the remaining cases.8 Damages ranged from $4,171.45 to $50 million. The authors emphasized specific defense strategies, including the importance of careful labeling and handling of embryos, prompt disclosure when an error does occur, and awareness of the specific state statute(s) of limitations for medical malpractice claims.8
Continue to: Legal considerations...
Legal considerations
The case at the beginning of this article is a “mix-up” case, in which an IVF center implanted the wrong embryo, resulting in the birth parents not being the biological parents.1 As in that case, there may be (but are not always)6,9 2 mix-ups, so that 2 couples have each other’s biological children. These cases may go unnoticed by the birth parent if the physical appearance is not unexpected and the parents never do genetic testing, or if the IVF center does not discover the error and inform the parents. Infrequently the cases make the news or the courts.10,11
News accounts are not trials, and we do not suggest that all the facts discussed in news reports on the case described here are complete—or even accurate in the details reported. They are generally 1-sided, so there are other perspectives. To consider the legal issues, however, we will assume for discussion only that the facts are as they have been reported in the news coverage—with the understanding that the discovery and trial processes would undoubtedly bring to light many other important facts or corrections.
Negligence
Although there are several potential bases for liability (ie, contract or warranty claims, a form of product liability/defect) in mix-up and other artificial reproductive technology (ART), negligence or malpractice seem most likely.12 “Negligence” here is intended to be simple negligence but may also include gross negligence or recklessness.
Although the incidence of errors in ART is unknown, there is limited evidence that suggests it is not a rare event. One study suggested >20% of fertility clinics knew of errors in processing or handling donor samples and embryos for implantation.13 Another study in the United Kingdom found that 1 in 1,000 IVF embryos were implanted in the wrong woman.14
Was there negligence? The first question in a malpractice or negligence-type action is, was there a professional relationship between the plaintiff who is claiming harm and the professional or organization defendant? The next question is whether the defendant was reasonably careful given the circumstances—that is, did the physician meet the “standard of care”? This is sometimes described as whether the professional’s actions would be acceptable (ie, reasonably prudent within the profession or specialty). If there was negligence, then the next question is, did that negligence cause an injury to the plaintiff?15
Determining the standard of care. The nature of the expected standard of care is dependent, in part, on the potential consequences of an error. For example, the care required when there is a significant risk of death from an error would be considerably more cautious than for an error that might result in small property damage. In this case study, a mix-up error is likely to be less severe than death, but is very substantial in terms of emotional harm and disruption. Thus, considerable care and attention would be expected to avoid these errors. They should be a “never” event. Institutions and physicians should give considerable attention to their processes and procedures to avoid the possibility of a mix-up error.16
Where did the negligence occur? There is an old tort doctrine “Res ipsa loquitor” (RIL) that means, “The thing speaks for itself.” Although there are several technical rules around the application of RIL as a presumption of negligence, it comes down to the proposition that some injuries do not occur without negligence. A traditional medical example is the sponge left in a patient during surgery—ordinarily that does not happen without some negligence. For RIL to be applied, usually the mechanism by which the injury occurred had to be under the control of the defendant (or the agents of the defendant).
The “mix-up” of embryos is an example of the kind of error that would not likely occur without negligence.17 But the embryo may not be in the exclusive control of any 1 institution. For example, the mistake could be made by the IVF center (or its employees), a separate facility that has processed or cryogenically stored the genetic materials, and independent physicians (not employees or agents of the center). Therefore, it is necessary to pinpoint where the negligence occurred and who is legally responsible. In some cases, a health care provider must take steps to ensure that its contractors have sufficient safeguards to avoid unnecessary harms. For example, an IVF center that uses an external cryogenic storage facility may have some obligation to know that the genetic material returned to the center is the same material that the center provided the storage facility in the first place and is properly identified.18
Assessing damages
From the facts as we have them, it appears that there must have been negligence that caused the mix-up of the embryos in the original case. It also appears reasonably clear that the negligence resulted in harm to both sets of parents and their families. This would suggest that the families should recover substantial damages. But that, somewhat surprisingly, may not be the case.19 Several legal principles may limit the availability or size of damages in mix-up cases. Also, it is worth remembering that there are differences in how states treat the different types of damages in these cases. Although the case was filed in California, we’ll take a more national view of the damages issue.
Not all harm is treated as equal. The first problem facing plaintiffs in mix-up cases may be the fact that they have suffered only emotional harm, without any physical injury. Traditionally, the courts have been reluctant to allow recovery in negligence for purely emotional injuries. Also “intentional” infliction of emotional distress does permit financial recovery, but generally “negligent” infliction of emotional harm traditionally has not. In part, this was because of the fear of unwarranted (and difficult-to-assess) claims of emotional harm that are not related to a physical harm. Some states developed a “zone of danger” exception (eg, where someone was almost hit by a car) or allowed some emotional injury recovery if there were “physical manifestations” of the emotional harm. In short, depending on the state’s rules, negligence that causes purely emotional harm may not be compensable.20
State-based malpractice “caps.” Another limitation on emotional injuries is the “caps” on malpractice damages enacted by several states (including California, where this mix-up case occurred). Therefore, if a mix-up case is determined to be a malpractice case under state law, emotional suffering damages (which are non-economic damages) may be limited to the cap—$250,000 in California, for example—even if the state allows damages for emotional injuries without physical injuries.
The rare exception. Very careless labeling or handling of the identity of the embryo could at the extreme be considered gross negligence or recklessness. There are relatively inexpensive and easy procedures that could easily avoid what is likely to be significant harm to families (including emotional upset).21 Institutions that callously fail to use those procedures might be seen by some courts as reckless, or in outrageous cases, even intentional. An example would be the University of California Irvine Center for Reproductive Health case, in which physicians intentionally (without consent) used patients’ ova, fertilized them, and then implanted them in other patients, with at least 15 births, many lawsuits, and multimillion dollar settlements.22 In “intentional” cases, limitations on emotional injuries would usually not be major barriers to recovery of damages. However, those are legal stretches, and recovery is the exception rather than the rule.23
Continue to: Additional legal concerns with IVF...
Additional legal concerns with IVF
Reproduction negligence cases include a large range of errors and injuries—not just embryo mix-ups. Courts have struggled with when it is appropriate to allow damages, even when there have been clear injuries. For the most part courts have been reluctant to find liability in many areas of new IVF technology.12 One problem in determining how to assess damages is determining how incidental benefits should be used to offset some or all of the damages. For example, how should the joy of having a child offset the costs of raising the child?
There are more than a dozen kinds of current and likely future claims arising from problems with ART. It is tempting to conclude, “Oh, what a tangled legal web we weave when first we practice to artificially conceive.” There are various groupings of such claims, with several examples of cases presented in this article. It is not possible to consider those in detail in this article. As a general proposition, however, “our legal system treats wrongfully disrupted plans concerning reproduction like one of those life adversities that people are expected to abide without remedy.”24
This is not to say, however, that there is no compensation for IVF-related injuries. Applebaum and colleagues found more than 100 cases in the 35 years covered by the study (1984-2020).8 However, only 50 of those cases fit the criteria for inclusion in their data. The successful cases for the plaintiffs involved medical or surgical error, while it appeared that various forms of wrongful life or birth were much less successful. It would be a mistake to conclude from these data that there are not, and will not be, meaningful risks of liability in the areas of IVF and ART more generally.
First, claims that fit with existing legal doctrine are producing liability. About half of the claims (25 over the 34 years) examined by Applebaum et al resulted in liability. Admittedly, that number was small because ART use was increasing. Where the claims fit well-recognized legal forms of damages and forms of action (primarily negligence), the liability could be substantial. A remarkable example of this is the case of Wuth v Lab. Corp (see “Liability for genetic testing errors”),25 which was the largest verdict ($50 million) in the Applebaum and colleagues’ study.8 The large verdict was due to the failure of the testing company and a medical center to properly perform and assess a genetic test, which resulted in the birth of a child with an unbalanced chromosome translocation.8,25 The child’s serious disabilities would require a great deal of expensive care. Although the jury held the testing laboratory and medical center liable, they did not find liability against the physician.25 Ultimately, this case would be considered a failure of genetic testing rather than an IVF case.
More than 2 couples
In a second case from California, a couples’ son was born to another couple in New York—along with another boy from a third couple. The woman in New York thought she had carried biological twins but genetic testing confirmed the twins were not related to the couple or to each other (the second couple filed a separate medical malpractice and negligence lawsuit in New York). All 3 couples had sought care at the same IVF clinic. The babies were eventually returned to their biological parents.1
Different races
In a New York case, a Korean couple had twin White boys after consenting to a single embryo transfer. Meanwhile a couple in Los Angeles who went to the same in vitro fertilization clinic gave birth to a child that did not match their appearance. Both couples had undergone embryo transfers on the same day. The court arranged for the Korean couple to surrender their twins to their biological parents when they were 6 months of age in exchange for their biological child.2
References
1. Couple claims clinic implanted their embryo in wrong woman. Associated Press. July 10, 2019. https://apnews.com/article/de32d537c6e34808b28834c23f00e272. Accessed January 6, 2022.
2. In the matter of accusation against Steven L. Katz. Case no. 03-20001-122617.OAH no. N2004080093. Sacramento, CA. Medical Board of California Department of Consumer Affairs 2005.
Future challenges
The future is likely to bring substantially expanded IVF/ART liability for several reasons. ART is becoming more common. Although courts have struggled with how to apply existing liability rules to the new technologies and related novel legal claims, the absence of established legal principles into which IVF injuries fit will not last forever. The legal system eventually finds ways of adjusting old rules or adopting new ones to cover injuries from new technology.
Although IVF injuries that most people feel deserve compensation currently are not cognizable in law, that will undoubtedly change. Either the courts will find new ways of assessing ART claims, or state legislatures and Congress will step in with legislation. To date, Congress has been relatively “hands off” on the ART processes, with the Fertility Clinic Success Rate and Certification Act of 1992 being a notable exception.24 This law requires ART programs to report success rates and directs the Centers for Disease Control and Prevention (CDC) to publish reported success rates and laboratory incidents. It also establishes a model state laboratory certification program.24 The CDC has an outline of the work under the statute,26 as well as state-specific data regarding ART27 and lists of publications in key areas.28 In addition there are various state laws related to recordkeeping, donor qualifications, licensing, and family law issues.29 Ultimately, physicians, scientists, and legal professionals can perform a valuable role in helping to fashion IVF liability principles that are workable and reasonable, that will not interfere with the progress of medicine, and that will ensure that those injured through carelessness or bad medicine receive compensation. ●
Although not technically an in vitro fertilization (IVF) case, Wuth v Lab. Corp. involved an infant born through IVF with a translocation defect chromosome 2 (ie, deleted material) and extra chromatin on 9. The father’s family history included birth defects, including a female cousin with profound developmental disabilities, seizures, and antisocial behavior. He had undergone genetic testing that revealed an asymptomatic balanced, 2;9 translocation. As part of the IVF process, the couple had a genetic consultation and were told there was a 50% chance that the fetus would have an unbalanced 2;9 translocation given the father’s family history and that chorionic villus sampling or amniocentesis could detect this in the fetus.1
Amniocentesis had been performed, with the specimen sent to Lab. Corp. The result was “normal male karyotype.” However, when the baby was born, it was immediately apparent that he had severe physical defects and subsequently cognitive defects. Genetic testing of the child revealed an unbalanced 2;9 translocation. The couple filed a suit for wrongful birth and wrongful life, which went to a jury. The child was awarded $25 million and the parents/family were awarded another $25 million in general damages. The verdict reflected errors in genetic (laboratory) testing.
Reference
1. Wuth v Lab. Corp. of Am., 189 Wash. App. 660, 359 P.3d 841 (2015).
- Mark J. California couple sues fertility clinic following IVF embryo mix-up. Washington Post. November 9, 2021. https://www.washingtonpost.com/nation/2021/11/09/in-vitro-fertilization-ivf-mix-up-daphna-cardinale. Accessed January 5, 2022.
- More than 8 million babies born from IVF since the world’s first in 1978. Science Daily. July 3, 2018. https://www.sciencedaily.com/releases/2018/07/180703084127.htm. Accessed January 11, 2022.
- ESCO Medical. In vitro fertilization (IVF) as fertility treatment. https://www.esco-medical.com/resource/in-vitro-fertilization-ivf-as-fertility-treatment.
- Vigdor N. “We had their baby, and they had our baby”: couple sues over embryo “mix-up.” NY Times. November 9, 2021. https://www.nytimes.com/2021/11/09/us/fertility-clinic-embryo-mixup.html. Accessed January 11, 2022.
- Couple claims clinic implanted their embryo in wrong woman. Associated Press. July 10, 2019. https://apnews.com/article/de32d537c6e34808b28834c23f00e2728. Accessed January 6, 2022.
- In the matter of accusation against Steven L. Katz. Case no. 03-20001-122617.OAH no. N2004080093. Sacramento, CA. Medical Board of California Department of Consumer Affairs 2005
- Rasouli MA, Moutos CP, Phelps JY. Liability for embryo mix-ups in fertility practices in the USA. J Assist Reprod Genet. 2021;38:1101-1107. doi:10.1007/s10815-021-02108-1
- Applebaum J, Berger D, O’Neill K. Can a reproductive endocrinologist be sued for 50 million dollars? A comprehensive review of malpractice litigation involving in vitro fertilization in the U.S. Fertil Steril. 2021;116(3s):e19. doi:10.1016/j.fertnstert.2021.07.059
- Andrews v Keltz, 838 N.Y.S.2d 363, 365 (Sup. Ct. 2007).
- Chichi DV. In vitro fertilization, fertility frustrations, and the lack of regulation. Hofstra L Rev. 2021;49:535-568. https://www.hofstralawreview.org/wp-content/uploads/2021/04/bb.2.chichi.pdf. Accessed January 11, 2022.
- Lewin T. Sperm banks accused of losing samples and lying about donors. NY Times. July 21, 2016. https://www.nytimes.com/2016/07/22/us/sperm-banks-accused-of-losing-samples-and-lying-about-donors.html. Accessed January 11, 2022.
- Bender L. To err is human ART mix-ups: labor-based, relational proposal. J Gender Race Justice. 2006;9:443-508. https://surface.syr.edu/cgi/viewcontent.cgi?article=1050&context=lawpub. Accessed January 11, 2022.
- Baruch S, Kaufman D, Hudson KL. Genetic testing of embryos: practices and perspectives of U.S. in vitro fertilization clinics. Fertil Steril. 2007;89:1053-1058. doi:10.1016/j.fertnstert.2007.05.048
- Liebler R. Are you my parent? Are you my child? The role of genetics and race in defining relationships after reproductive technological mistakes. DePaul J Health Care Law. 2002;5:15-56. https://via.library.depaul.edu/cgi/viewcontent.cgi?article=1202&context=jhcl. Accessed January 11, 2022.
- Crockin SL, Altman AB, Edmonds MA. The history and future trends of art medicine and law. Fam Court Rev. 2021;59:22-45. doi:10.1111/fcre.12550
- Fernandes JS. Perfecting pregnancy via preimplantation genetic screening: the quest for an elusive standard of care. UC Irvine L Rev. 2014;4:1295-1326. https://www.law.uci.edu/lawreview/vol4/no4/Fernandes.pdf. Accessed January 11, 2022.
- VanGessel MM. Wrongful surrogacy: the need for right of action in cases of clear negligence. U Toledo L Rev. 2015;46:681-706.
- Reich J, Swink D. Outsourcing human reproduction: embryos and surrogacy services in the cyberprocreation era. J Health Care L Policy. 2011;14:241-298. https://core.ac.uk/download/pdf/217156567.pdf. Accessed January 11, 2022.
- Strasser M. Prenatal tort slippage. Health Matrix. 2021;31:221-262. https://scholarlycommons.law.case.edu/healthmatrix/vol31/iss1/9. Accessed January 11, 2022.
- Heide IH. Negligence in the creation of healthy babies: negligent infliction of emotional distress in cases of alternative reproductive technology malpractice without physical injury. J Med L. 2005;9:55-94.
- Novo S, Nogués C, Penon O, et al. Barcode tagging of human oocytes and embryos to prevent mix-ups in assisted reproduction technologies. Hum Reprod. 2014;29:18-28. doi: 10.1093/humrep/det409
- Yoshino K. UCI Settles Dozens of Fertility Suits. LA Times. September 11, 2009. https://www.latimes.com/archives/la-xpm-2009-sep-11-me-uci-fertility11-story.html. Accessed January 11, 2022.
- Fox D. Reproductive negligence. Columbia L Rev. 2017;117:149-242. https://columbialawreview.org/wp-content/uploads/2017/01/149.pdf. Accessed January 11, 2022.
- 42 U.S.C.S. §263a-1-263a-7; Public Law 102-493. https://www.govinfo.gov/content/pkg/STATUTE-106/pdf/STATUTE-106-Pg3146.pdf. Accessed January 11, 2022.
- Wuth v Lab. Corp. of Am., 189 Wash. App. 660, 359 P.3d 841 (2015).
- Centers for Disease Control and Prevention. The Fertility Clinic Success Rate and Certification Act. December 14, 2020. https://www.cdc.gov/art/nass/policy.html#act. Accessed January 11, 2022.
- Centers for Disease Control and Prevention. State-specific assisted reproductive technology surveillance. December 17, 2020. https://www.cdc.gov/art/state-specific-surveillance/index.html. Accessed January 11, 2022.
- Centers for Disease Control and Prevention. Key findings. March 12, 2021. https://www.cdc.gov/art/key-findings/index.html. Accessed January 11, 2022.
- Cohen EN. 5 Treatise on Health Care Law §22.04, (ed. Hooper, Lundy & Bookman, & Robert W. Lundy, Jr. RW.) (Matthew Bender-LexisNexis)
- Mark J. California couple sues fertility clinic following IVF embryo mix-up. Washington Post. November 9, 2021. https://www.washingtonpost.com/nation/2021/11/09/in-vitro-fertilization-ivf-mix-up-daphna-cardinale. Accessed January 5, 2022.
- More than 8 million babies born from IVF since the world’s first in 1978. Science Daily. July 3, 2018. https://www.sciencedaily.com/releases/2018/07/180703084127.htm. Accessed January 11, 2022.
- ESCO Medical. In vitro fertilization (IVF) as fertility treatment. https://www.esco-medical.com/resource/in-vitro-fertilization-ivf-as-fertility-treatment.
- Vigdor N. “We had their baby, and they had our baby”: couple sues over embryo “mix-up.” NY Times. November 9, 2021. https://www.nytimes.com/2021/11/09/us/fertility-clinic-embryo-mixup.html. Accessed January 11, 2022.
- Couple claims clinic implanted their embryo in wrong woman. Associated Press. July 10, 2019. https://apnews.com/article/de32d537c6e34808b28834c23f00e2728. Accessed January 6, 2022.
- In the matter of accusation against Steven L. Katz. Case no. 03-20001-122617.OAH no. N2004080093. Sacramento, CA. Medical Board of California Department of Consumer Affairs 2005
- Rasouli MA, Moutos CP, Phelps JY. Liability for embryo mix-ups in fertility practices in the USA. J Assist Reprod Genet. 2021;38:1101-1107. doi:10.1007/s10815-021-02108-1
- Applebaum J, Berger D, O’Neill K. Can a reproductive endocrinologist be sued for 50 million dollars? A comprehensive review of malpractice litigation involving in vitro fertilization in the U.S. Fertil Steril. 2021;116(3s):e19. doi:10.1016/j.fertnstert.2021.07.059
- Andrews v Keltz, 838 N.Y.S.2d 363, 365 (Sup. Ct. 2007).
- Chichi DV. In vitro fertilization, fertility frustrations, and the lack of regulation. Hofstra L Rev. 2021;49:535-568. https://www.hofstralawreview.org/wp-content/uploads/2021/04/bb.2.chichi.pdf. Accessed January 11, 2022.
- Lewin T. Sperm banks accused of losing samples and lying about donors. NY Times. July 21, 2016. https://www.nytimes.com/2016/07/22/us/sperm-banks-accused-of-losing-samples-and-lying-about-donors.html. Accessed January 11, 2022.
- Bender L. To err is human ART mix-ups: labor-based, relational proposal. J Gender Race Justice. 2006;9:443-508. https://surface.syr.edu/cgi/viewcontent.cgi?article=1050&context=lawpub. Accessed January 11, 2022.
- Baruch S, Kaufman D, Hudson KL. Genetic testing of embryos: practices and perspectives of U.S. in vitro fertilization clinics. Fertil Steril. 2007;89:1053-1058. doi:10.1016/j.fertnstert.2007.05.048
- Liebler R. Are you my parent? Are you my child? The role of genetics and race in defining relationships after reproductive technological mistakes. DePaul J Health Care Law. 2002;5:15-56. https://via.library.depaul.edu/cgi/viewcontent.cgi?article=1202&context=jhcl. Accessed January 11, 2022.
- Crockin SL, Altman AB, Edmonds MA. The history and future trends of art medicine and law. Fam Court Rev. 2021;59:22-45. doi:10.1111/fcre.12550
- Fernandes JS. Perfecting pregnancy via preimplantation genetic screening: the quest for an elusive standard of care. UC Irvine L Rev. 2014;4:1295-1326. https://www.law.uci.edu/lawreview/vol4/no4/Fernandes.pdf. Accessed January 11, 2022.
- VanGessel MM. Wrongful surrogacy: the need for right of action in cases of clear negligence. U Toledo L Rev. 2015;46:681-706.
- Reich J, Swink D. Outsourcing human reproduction: embryos and surrogacy services in the cyberprocreation era. J Health Care L Policy. 2011;14:241-298. https://core.ac.uk/download/pdf/217156567.pdf. Accessed January 11, 2022.
- Strasser M. Prenatal tort slippage. Health Matrix. 2021;31:221-262. https://scholarlycommons.law.case.edu/healthmatrix/vol31/iss1/9. Accessed January 11, 2022.
- Heide IH. Negligence in the creation of healthy babies: negligent infliction of emotional distress in cases of alternative reproductive technology malpractice without physical injury. J Med L. 2005;9:55-94.
- Novo S, Nogués C, Penon O, et al. Barcode tagging of human oocytes and embryos to prevent mix-ups in assisted reproduction technologies. Hum Reprod. 2014;29:18-28. doi: 10.1093/humrep/det409
- Yoshino K. UCI Settles Dozens of Fertility Suits. LA Times. September 11, 2009. https://www.latimes.com/archives/la-xpm-2009-sep-11-me-uci-fertility11-story.html. Accessed January 11, 2022.
- Fox D. Reproductive negligence. Columbia L Rev. 2017;117:149-242. https://columbialawreview.org/wp-content/uploads/2017/01/149.pdf. Accessed January 11, 2022.
- 42 U.S.C.S. §263a-1-263a-7; Public Law 102-493. https://www.govinfo.gov/content/pkg/STATUTE-106/pdf/STATUTE-106-Pg3146.pdf. Accessed January 11, 2022.
- Wuth v Lab. Corp. of Am., 189 Wash. App. 660, 359 P.3d 841 (2015).
- Centers for Disease Control and Prevention. The Fertility Clinic Success Rate and Certification Act. December 14, 2020. https://www.cdc.gov/art/nass/policy.html#act. Accessed January 11, 2022.
- Centers for Disease Control and Prevention. State-specific assisted reproductive technology surveillance. December 17, 2020. https://www.cdc.gov/art/state-specific-surveillance/index.html. Accessed January 11, 2022.
- Centers for Disease Control and Prevention. Key findings. March 12, 2021. https://www.cdc.gov/art/key-findings/index.html. Accessed January 11, 2022.
- Cohen EN. 5 Treatise on Health Care Law §22.04, (ed. Hooper, Lundy & Bookman, & Robert W. Lundy, Jr. RW.) (Matthew Bender-LexisNexis)
HT for women who have had BSO before the age of natural menopause: Discerning the nuances
Women who undergo bilateral salpingo-oophorectomy (BSO) for various indications prior to menopause experience a rapid decline in ovarian hormone levels and consequent vasomotor and other menopausal symptoms. In addition, the resulting estrogen deprivation is associated with such long-term adverse outcomes as osteoporosis and cardiovascular morbidity.
OBG M
Surgical vs natural menopause
Stephanie Faubion, MD, MBA, NCMP: Since the Women’s Health Initiative study was published in 2002,2 many clinicians have been fearful of using systemic HT in menopausal women, and HT use has declined dramatically such that only about 4% to 6% of menopausal women are now receiving systemic HT. Importantly, however, a group of younger menopausal women also are not receiving HT, and that is women who undergo BSO before they reach the average age of menopause, which in the United States is about age 52; this is sometimes referred to as surgical menopause or early surgical menopause. Early surgical menopause has different connotations for long-term health risks than natural menopause at the average age, and we are here to discuss these health effects and their management.
My name is Stephanie Faubion, and I am a women’s health internist and the Chair of the Department of Medicine at Mayo Clinic in Jacksonville, Florida, and Director of Mayo Clinic Women’s Health. I am here with 2 of my esteemed colleagues, Dr. Andrew Kaunitz and Dr. Ekta Kapoor.
Andrew M. Kaunitz, MD, NCMP: Hello, I am an ObGyn with the University of Florida College of Medicine in Jacksonville, with particular interests in contraception, menopause, and gynecologic ultrasonography.
Ekta Kapoor, MBBS, NCMP: And I am an endocrinologist at Mayo Clinic in Rochester with a specific interest in menopause and hormone therapy. I am also the Assistant Director for Mayo Clinic Women’s Health.
Higher-than-standard estrogen doses needed in younger menopausal women
Dr. Faubion: Let’s consider a couple of cases so that we can illustrate some important points regarding hormone management in women who have undergone BSO before the age of natural menopause.
Our first case patient is a woman who is 41 years of age and, because of adenomyosis, she will undergo a hysterectomy. She tells her clinician that she is very concerned about ovarian cancer risk because one of her good friends recently was diagnosed with ovarian cancer, and together they decide to remove her ovaries at the time of hysterectomy. Notably, her ovaries were healthy.
The patient is now menopausal postsurgery, and she is having significant hot flashes and night sweats. She visits her local internist, who is concerned about initiating HT. She is otherwise a healthy woman and does not have any contraindications to HT. Dr. Kaunitz, what would you tell her internist?
Dr. Kaunitz: We are dealing with 2 different issues in terms of decision making about systemic HT for this 41-year-old who has undergone BSO. First, as you mentioned, Dr. Faubion, she has bothersome hot flashes, or vasomotor symptoms. Unless there are contraindications, systemic HT would be appropriate. Although I might start treatment at standard doses, and the accompanying TABLE depicts standard doses for the 2 most common oral estrogen formulations as well as transdermal estradiol, it’s important to recognize that younger menopausal women often will need to use higher-than-standard doses.
For example, for a 53-year-old woman who has been menopausal for a year or 2 and now has bothersome symptoms, I might start her on estradiol 1 mg tablets with progestin if a uterus is present. However, in this 41-year-old case patient, while I might start treatment at a standard dose, I would anticipate increasing to higher doses, such as 1.5 or 2 mg of daily estradiol until she feels her menopausal symptoms are adequately addressed.
Dr. Faubion: It is important to note that sometimes women with early BSO tend to have more severe vasomotor symptoms. Do you find that sometimes a higher dose is required just to manage symptoms, Dr. Kaunitz?
Dr. Kaunitz: Absolutely, yes. The decision whether or not to use systemic HT might be considered discretionary or elective in the classic 53-year-old woman recently menopausal with hot flashes, a so-called spontaneously or naturally menopausal woman. But my perspective is that unless there are clear contraindications, the decision to start systemic HT in the 41-year-old BSO case patient is actually not discretionary. Unless contraindications are present, it is important not only to treat symptoms but also to prevent an array of chronic major health concerns that are more likely if we don’t prescribe systemic HT.
Continue to: Health effects of not using HT...
Health effects of not using HT
Dr. Faubion: Dr. Kapoor, can you describe the potential long-term adverse health consequences of not using estrogen therapy? Say the same 41-year-old woman does not have many bothersome symptoms. What would you do?
Dr. Kapoor: Thank you for that important question. Building on what Dr. Kaunitz said, in these patients there are really 2 issues that can seem to be independent but are not: The first relates to the immediate consequences of lack of estrogen, ie, the menopause-related symptoms, but the second and perhaps the bigger issue is the long-term risk associated with estrogen deprivation.
The symptoms in these women are often obvious as they can be quite severe and abrupt; one day these women have normal hormone levels and the next day, after BSO, suddenly their hormones are very low. So if symptoms occur, they are usually hard to miss, simply because they are very drastic and very severe.
Historically, patients and their clinicians have targeted these symptoms. Patients experience menopausal symptoms, they seek treatment, and then the clinicians basically titrate the treatment to manage these symptoms. That misses the bigger issue, however, which is that premature estrogen deprivation leads to a host of chronic health conditions, as Dr. Kaunitz mentioned. These mainly include increased risk for cardiovascular disease, diabetes, hypertension, dyslipidemia, increased risk of mortality, dementia, and osteoporosis.
Fairly strong observational evidence suggests that use of estrogen therapy given in replacement doses—doses higher than those typically used in women after natural menopause, therefore considered replacement doses—helps mitigate the risk of some of these adverse health conditions.
In these women, the bigger goal really is to reinstate the hormonal milieu that exists prior to menopause. To your point, Dr. Faubion, if I have a patient who is younger than 46 years, who has her ovaries taken out, and even if she has zero symptoms (and sometimes that does happen), I would still make a case for this patient to utilize hormone therapy unless there is a contraindication such as breast cancer or other estrogen-sensitive cancers.
Dr. Faubion: Again, would you aim for those higher doses rather than treat with the “lowest dose”?
Dr. Kapoor: Absolutely. My punchline to the patients and clinicians in these discussions is that the rules of the game are different for these women. We cannot extrapolate the risks and benefits of HT use in women after natural menopause to younger women who have surgical menopause. Those rules just do not apply with respect to both benefits and risks.
Dr. Faubion: I think it’s important to say that these same “rules” would apply if the women were to go through premature menopause for any other reason, too, such as chemotherapy, radiation therapy, or premature ovarian insufficiency for any number of reasons, including toxic, metabolic, or genetic causes and so on. Would that be true?
Dr. Kapoor: Yes, absolutely so.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: In terms of practical or clinical issues regarding systemic HT management, for the woman in her early 50s who has experienced normal or natural spontaneous menopause, a starting dose of transdermal estradiol would be, for instance, a 0.05-mg patch, which is a patch that over 24 hours releases 0.05 mg of estradiol daily; or standard oral estrogen, including conjugated equine estrogen, a 0.625-mg tablet daily, or estradiol, a 1-mg tablet daily.
But in younger patients, we want to use higher doses. For a patch, for instance, I would aim for a 0.075- or 0.1-mg estradiol patch, which releases a higher daily dose of estradiol than the standard dose. For oral estrogen, the dose would be 0.9- or even 1.25-mg tablets of conjugated equine estrogen or 1.5 mg, which is a 1-mg plus a 0.5-mg estradiol tablet, or a 2-mg estradiol tablet. Estradiol does come in a 2-mg strength.
For oral estrogen, I prefer estradiol because it’s available as a generic medication and often available at a very low cost, sometimes as low as $4 a month from chain pharmacies.
Continue to: Usefulness of monitoring estradiol levels for dosage adjustment...
Usefulness of monitoring estradiol levels for dosage adjustment
Dr. Faubion: That’s a great point, and again it is important to emphasize that we are aiming to recreate the premenopausal hormonal milieu. If you were to check estradiol levels, that would be aiming for a premenopausal range of approximately 80 to 120 pg per mL. Dr. Kapoor, is there utility in monitoring estrogen levels?
Dr. Kapoor: Great question, Dr. Faubion, and as you know it’s a loaded one. We base this on empiric evidence. We know that if the hormonal milieu in a young patient is changed to a postmenopausal one, her risk for many chronic conditions is increased. So if we were to reinstate a premenopausal hormonal milieu, that risk would probably be reduced. It makes good sense to target an empiric goal of 80 to 120 pg per mL of estradiol, which is the average estradiol level in a premenopausal woman. If you were to ask me, however, are there randomized, controlled trial data to support this practice—that is, if you target that level, can you make sure that the risk of diabetes is lower or that the risk of heart disease is lower—that study has yet to be done, and it may not ever be done on a large scale. However, it intuitively makes good sense to target premenopausal estradiol levels.
Dr. Faubion: When might you check an estradiol level in this population? For example, if you are treating a patient with a 0.1-mg estradiol patch and she still has significant hot flashes, would it be useful to check the level?
Dr. Kapoor: It would. In my practice, I check estradiol levels on these patients on an annual basis, regardless of symptoms, but definitely in the patient who has symptoms. It makes good sense, because sometimes these patients don’t absorb the estrogen well, particularly if administered by the transdermal route.
A general rule of thumb is that in the average population, if a patient is on the 0.1-mg patch, for example, you would expect her level to be around 100. If it is much lower than that, which sometimes happens, that speaks for poor absorption. Options at that point would be to treat her with a higher dose patch, depending on what the level is, or switch to a different formulation, such as oral.
In instances in which I have treated patients with a 0.1-mg patch for example, and their estradiol levels are undetectable, that speaks for very poor absorption. For such patients I make a case for switching them to oral therapy. Most definitely that makes sense in a patient who is symptomatic despite treatment. But even for patients who don’t have symptoms, I like to target that level, acknowledging that there is no evidence as such to support this practice.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: Yes, a few practical points. Although patches are available in a wider array of doses than oral estrogen formulations, the highest dose available is 0.1 mg. It’s important for clinicians to recognize that while checking serum levels when indicated can be performed in women using transdermal estradiol or patches, in women who are using oral estrogen, checking blood levels is not going to work well because serum estrogen levels have a daily peak and valley in women who use oral versus transdermal estradiol.
I also wanted to talk about progestins. Although many patients who have had a BSO prior to spontaneous menopause also have had a hysterectomy, others have an intact uterus associated with their BSO, so progestins must be used along with estrogen. And if we are using higher-than-standard doses of estrogen, we also need to use higher-than-standard doses of progestin.
In that classic 53-year-old woman I referred to who had spontaneous normal menopause, if she is taking 1 mg of estradiol daily, or a 0.05-mg patch, or 0.625 mg of conjugated equine estrogen, 2.5 mg of medroxyprogesterone is fine. In fact, that showed excellent progestational protection of the endometrium in the Women’s Health Initiative and in other studies.
However, if we are going to use double the estrogen dose, we should increase the progestin dose too. In some of my patients on higher estrogen doses who have an intact uterus, I’ll use 5 or even 10 mg of daily medroxyprogesterone acetate to ensure adequate progestational suppression.
Dr. Faubion: Another practical tip is that if one is using conjugated equine estrogens, measuring the serum estradiol levels is not useful either.
Dr. Kaunitz: I agree.
Continue to: Oral contraceptives as replacement HT...
Oral contraceptives as replacement HT
Dr. Faubion: Would you comment on use of a birth control pill in this circumstance? Would it be optimal to use a postmenopausal HT regimen as opposed to a birth control pill or combined hormonal contraception?
Dr. Kapoor: In this younger population, sometimes it seems like a more socially acceptable decision to be on a birth control option than on menopausal HT. But there are some issues with being on a contraceptive regimen. One is that we end up using estrogen doses much higher than what is really needed for replacement purposes. It is also a nonphysiologic way of replacement in another sense—as opposed to estradiol, which is the main hormone made by the ovaries, the hormonal contraceptive regimens contain the synthetic estrogen ethinyl estradiol for the most part.
The other issue that is based on some weak evidence is that it appears that the bone health outcomes are probably inferior with combined hormonal contraception. For these reasons, regimens that are based on replacement doses of estradiol are preferred.
Dr. Faubion: Right, although the data are somewhat weak, I agree that thus far it seems optimal to utilize a postmenopausal regimen for various reasons. Dr. Kaunitz, anything to add?
Dr. Kaunitz: Yes, to underscore Dr. Kapoor’s point, a common oral contraceptive that contains 20 µg of ethinyl estradiol is substantially more estrogenic than 1.0 or 2.0 mg of micronized oral estradiol.
Also consider that a 20-µg ethinyl estradiol oral contraceptive may increase the risk of venous thromboembolism more than menopausal doses of oral estradiol, whether it be a micronized estradiol or conjugated equine estrogen.
Dr. Faubion: So the risk may be greater with oral combined hormonal contraception as well?
Dr. Kaunitz: One thing we can do is explain to our patients that their ovaries, prior to surgery or prior to induced menopause, were making substantial quantities of estradiol. Whether we prescribe a patch or oral micronized estradiol, this estrogen is identical to the hormone that their ovaries were making prior to surgery or induced menopause.
Breast cancer concerns
Dr. Faubion: Let’s consider a more complicated case. A 35-year-old woman has an identified BRCA1 mutation; she has not had any cancers but has undergone risk-reducing BSO and her uterus remains. Is this woman a candidate for HT? At what dose, and for how long? Dr. Kaunitz, why don’t you start.
Dr. Kaunitz: That is a challenging case but one that I think our readers will find interesting and maybe even provocative.
We know that women with BRCA1 mutations, the more common of the 2 BRCA mutations, have a very high risk of developing epithelial ovarian cancer at a young age. For this reason, our colleagues in medical oncology who specialize in hereditary ovarian/breast cancer syndromes recommend prophylactic risk-reducing—and I would also say lifesaving—BSO with or without hysterectomy for women with BRCA1 mutations.
However, over the years there has been tremendous reluctance among physicians caring for BRCA patients and the women themselves—I use the term “previvors” to describe BRCA carriers who have not been diagnosed with breast or ovarian cancer—to use HT after BSO because of concerns that HT might increase breast cancer risk in women who are already at high risk for breast cancer.
I assume, Dr. Faubion, that in this case the woman had gynecologic surgery but continues to have intact breasts. Is that correct?
Dr. Faubion: That is correct.
Dr. Kaunitz: Although the assumption has been that it is not safe to prescribe HT in this setting, in fact, the reported cohort studies that have looked at this issue have not found an elevated risk of breast cancer when replacement estrogen, with or without progestin, is prescribed to BRCA1 previvors with intact breasts.
Given what Dr. Kapoor said regarding the morbidity that is associated with BSO without replacement of physiologic estrogen, and also given the severe symptoms that so many of these young menopausal women experience, in my practice I do prescribe estrogen or estrogen-progestin therapy and focus on the higher target doses that we discussed for the earlier case patient who had a hysterectomy for abnormal uterine bleeding with adenomyosis.
Dr. Faubion: Dr. Kapoor, do you agree with this approach? How long would you continue therapy?
Dr. Kapoor: First, in this BRCA1 case we need to appreciate that the indication for the BSO is a legitimate one, in contrast to the first case in which the ovaries were removed in a patient whose average risk of ovarian cancer was low. It is important to recognize that surgery performed in this context is the right thing to do because it does significantly reduce the risk of ovarian cancer.
The second thing to appreciate is that while we reduce the risk of ovarian cancer significantly and make sure that these patients survive longer, it’s striking a fine balance in that you want to make sure that their morbidity is not increased as a result of premature estrogen deprivation.
As Dr. Kaunitz told us, the evidence that we have so far, which granted is not very robust but is fairly strong observational evidence, suggests that the risk of breast cancer is not elevated when these patients are treated with replacement doses of HT.
Having said that, I do have very strong discussions with my patients in this category about having the risk-reducing bilateral mastectomy also, because if they were to get breast cancer because of their increased genetic predisposition, the cancer is likely to grow faster if the patient is on HT. So one of my counseling points to patients is that they strongly consider bilateral mastectomy, which reduces their breast cancer risk by more than 90%. At the same time, I also strongly endorse using HT in replacement doses for the reasons that we have already stated.
Dr. Faubion: Continue HT until age 50 or 52?
Dr. Kapoor: Definitely until that age, and possibly longer, depending on their symptoms. The indications for treating beyond the age of natural menopause are much the same as for women who experience natural menopause.
Dr. Faubion: That is assuming they had a bilateral mastectomy?
Dr. Kapoor: Yes.
Continue to: Continuing HT until the age of natural menopause...
Continuing HT until the age of natural menopause
Dr. Kaunitz: Dr. Kapoor brings up the important point of duration of systemic HT. I agree that similar considerations apply both to the healthy 41-year-old who had a hysterectomy for abnormal uterine bleeding and to the 35-year-old who had risk-reducing surgery because of her BRCA1 mutation.
In the 2 cases, both to treat symptoms and to prevent chronic diseases, it makes sense to continue HT at least until the age of natural menopause. That is consistent with 2017 guidance from The North American Menopause Society (NAMS) position statement on the use of systemic HT, that is, continuing systemic HT at least until the age of natural menopause.3 Then at that point, continuing or discontinuing systemic HT becomes discretionary, and that would be true for both cases. If the patient is slender or has a strong family history of osteoporosis, that tends to push the patient more in terms of continuing systemic HT. Those are just some examples, and Dr. Kapoor may want to detail other relevant considerations.
Dr. Kapoor: I completely agree. The decision is driven by symptoms that are not otherwise well managed, for example, with nonhormone strategies. If we have any concerns utilizing HT beyond the age of natural menopause, then nonhormonal options can be considered; but sometimes those are not as effective. And bone health is very important. You want to avoid using bisphosphonates in younger women and reserve them for older patients in their late 60s and 70s. Hormone therapy use is a very reasonable strategy to prevent bone loss.
Dr. Kaunitz: It is also worth mentioning that sometimes the woman involved in shared decision making with her clinician decides to stop systemic HT. In that setting, should the patient start developing new-onset dyspareunia, vaginal dryness, or other genital or sexuality-related concerns, it takes very little for me to advise that she start low-dose local vaginal estrogen therapy.
Dr. Faubion: In either scenario, if a woman were to develop symptoms consistent with genitourinary syndrome of menopause (GSM), would you use vaginal estrogen in addition to the systemic estrogen or alone after the woman elected to discontinue systemic therapy?
Dr. Kapoor: Yes to both, I would say.
Dr. Kaunitz: As my patients using systemic HT age, often I will lower the dose. For instance, the dose I use in a 53-year-old will be higher than when she is 59 or 62. At the same time, as we lower the dose of systemic estrogen therapy, symptoms of vaginal atrophy or GSM often will appear, and these can be effectively treated by adding low-dose vaginal estrogen therapy. A number of my patients, particularly those who are on lower-than-standard doses of systemic HT, are also using low-dose vaginal estrogen therapy.
There is a “hybrid” product available: the 90-day estradiol vaginal ring. Estring is a low-dose, 2-mg, 90-day estradiol ring that is very useful, but it is effective only for treating GSM or vaginal atrophy. A second menopausal vaginal estradiol ring, Femring, is available in 2 doses: 0.05 mg/day and 0.1 mg/day. These are very effective in treating both systemic issues, such as vasomotor symptoms or prevention of osteoporosis, and very effective in treating GSM or vaginal atrophy. One problem is that Femring, depending on insurance coverage, can be very expensive. It’s not available as a generic, so for insurance or financial reasons I don’t often prescribe it. If I could remove those financial barriers, I would prescribe Femring more often because it is very useful.
Dr. Faubion: You raise an important point, and that is, for women who have been on HT for some time, clinicians often feel the need to slowly reduce the dose. Would you do that same thing, Dr. Kapoor, for a 40-year-old woman? Would you reduce the dose as she approaches age 50? Is there pressure that “she shouldn’t be on that much estrogen”?
Dr. Kapoor: No, I would not feel pressured until the patient turns at least 46. I bring up age 46 because the average age range for menopause is 46 to 55. After that, if there is any concern, we can decrease the dose to half and keep the patient on that until she turns 50 or 51. But most of my patients are on replacement doses until the average age of menopause, which is around 51 years, and that’s when you reduce the dose to that of the typical HT regimens used after natural menopause.
Sometimes patients are told something by a friend or they have read something and they worry about the risk of 2 things. One is breast cancer and the other is venous thromboembolism (VTE), and that may be why they want to be on a lower dose. I counsel patients that while the risk of VTE is real with HT, it is the women after natural menopause who are at risk—because age itself is a risk for VTE—and it also has to do with the kind of HT regimen that a patient is on. High doses of oral estrogens and certain progestogens increase the risk. But again, for estradiol used in replacement doses and the more common progestogens that we now use in practice, such as micronized progesterone, the risk is not the same. The same goes for breast cancer. My biggest message to patients and clinicians who take care of these patients is that the rules that apply to women after natural menopause just do not apply to this very different patient population.
Dr. Faubion: Thank you, Dr. Kaunitz and Dr. Kapoor, for sharing your knowledge and experience. ●
Systemic HT past the age of 65
Dr. Kaunitz: Another practical issue relates to long-term or extended use of systemic HT. It’s not infrequent in my practice to receive mail and faxes from insurance carriers of systemic HT users who are age 65 and older in which the company refers to the American Geriatrics Society’s Beers criteria for potentially inappropriate medication use in older adults,1 suggesting that systemic HT is inappropriate for all women over age 65. In this age group, I use lower doses if I am continuing systemic HT. But the good news is that both NAMS and the American College Obstetricians and Gynecologists indicate that arbitrarily stopping systemic HT at age 65 or for any other arbitrary reason is inappropriate, and that decisions about continuing or discontinuing therapy should be made on an individualized basis using shared decision making. That’s an important message for our readers.
Counseling regarding elective BSO
Dr. Faubion: One final note about elective BSO in the absence of a genetic mutation that predisposes to increased ovarian or breast cancer risk. Fortunately, we have seen rates of oophorectomy before the age of natural menopause decline, but what would your advice be to women or clinicians of these women who say they are “just afraid of ovarian cancer and would like to have their ovaries removed before the age of natural menopause”?
Dr. Kaunitz: If patients have increased anxiety about ovarian cancer and yet they themselves are not known to be at elevated risk, I emphasize that, fortunately, ovarian cancer is uncommon. It is much less common than other cancers the patient might be familiar with, such as breast or colon or lung cancer. I also emphasize that women who have given birth, particularly multiple times; women who nursed their infants; and women who have used combination hormonal contraceptives, particularly if long term, are at markedly lower risk for ovarian cancer as they get older. We are talking about an uncommon cancer that is even less common if women have given birth, nursed their infants, or used combination contraceptives long term.
Dr. Faubion: Dr. Kapoor, what would you say regarding the increased risk they might incur if they do have their ovaries out?
Dr. Kapoor: As Dr. Kaunitz said, this is an uncommon cancer, and pursuing something to reduce the risk of an uncommon cancer does not benefit the community. That is also my counseling point to patients.
I also talk to them extensively about the risk associated with the ovaries being removed, and I tell them that although we have the option of giving them HT, it is hard to replicate the magic of nature. No matter what concoction or regimen we use, we cannot ensure reinstating health to what it was in the premenopausal state, because estrogen has such myriad effects on the body in so many different organ systems.
Reference
1. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227-2246.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430.
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
- North American Menopause Society. The 2017 hormone therapy position statement of The North American Menopause Society. J North Am Menopause Soc. 2017;24: 728-753.

Women who undergo bilateral salpingo-oophorectomy (BSO) for various indications prior to menopause experience a rapid decline in ovarian hormone levels and consequent vasomotor and other menopausal symptoms. In addition, the resulting estrogen deprivation is associated with such long-term adverse outcomes as osteoporosis and cardiovascular morbidity.
OBG M
Surgical vs natural menopause
Stephanie Faubion, MD, MBA, NCMP: Since the Women’s Health Initiative study was published in 2002,2 many clinicians have been fearful of using systemic HT in menopausal women, and HT use has declined dramatically such that only about 4% to 6% of menopausal women are now receiving systemic HT. Importantly, however, a group of younger menopausal women also are not receiving HT, and that is women who undergo BSO before they reach the average age of menopause, which in the United States is about age 52; this is sometimes referred to as surgical menopause or early surgical menopause. Early surgical menopause has different connotations for long-term health risks than natural menopause at the average age, and we are here to discuss these health effects and their management.
My name is Stephanie Faubion, and I am a women’s health internist and the Chair of the Department of Medicine at Mayo Clinic in Jacksonville, Florida, and Director of Mayo Clinic Women’s Health. I am here with 2 of my esteemed colleagues, Dr. Andrew Kaunitz and Dr. Ekta Kapoor.
Andrew M. Kaunitz, MD, NCMP: Hello, I am an ObGyn with the University of Florida College of Medicine in Jacksonville, with particular interests in contraception, menopause, and gynecologic ultrasonography.
Ekta Kapoor, MBBS, NCMP: And I am an endocrinologist at Mayo Clinic in Rochester with a specific interest in menopause and hormone therapy. I am also the Assistant Director for Mayo Clinic Women’s Health.
Higher-than-standard estrogen doses needed in younger menopausal women
Dr. Faubion: Let’s consider a couple of cases so that we can illustrate some important points regarding hormone management in women who have undergone BSO before the age of natural menopause.
Our first case patient is a woman who is 41 years of age and, because of adenomyosis, she will undergo a hysterectomy. She tells her clinician that she is very concerned about ovarian cancer risk because one of her good friends recently was diagnosed with ovarian cancer, and together they decide to remove her ovaries at the time of hysterectomy. Notably, her ovaries were healthy.
The patient is now menopausal postsurgery, and she is having significant hot flashes and night sweats. She visits her local internist, who is concerned about initiating HT. She is otherwise a healthy woman and does not have any contraindications to HT. Dr. Kaunitz, what would you tell her internist?
Dr. Kaunitz: We are dealing with 2 different issues in terms of decision making about systemic HT for this 41-year-old who has undergone BSO. First, as you mentioned, Dr. Faubion, she has bothersome hot flashes, or vasomotor symptoms. Unless there are contraindications, systemic HT would be appropriate. Although I might start treatment at standard doses, and the accompanying TABLE depicts standard doses for the 2 most common oral estrogen formulations as well as transdermal estradiol, it’s important to recognize that younger menopausal women often will need to use higher-than-standard doses.

For example, for a 53-year-old woman who has been menopausal for a year or 2 and now has bothersome symptoms, I might start her on estradiol 1 mg tablets with progestin if a uterus is present. However, in this 41-year-old case patient, while I might start treatment at a standard dose, I would anticipate increasing to higher doses, such as 1.5 or 2 mg of daily estradiol until she feels her menopausal symptoms are adequately addressed.
Dr. Faubion: It is important to note that sometimes women with early BSO tend to have more severe vasomotor symptoms. Do you find that sometimes a higher dose is required just to manage symptoms, Dr. Kaunitz?
Dr. Kaunitz: Absolutely, yes. The decision whether or not to use systemic HT might be considered discretionary or elective in the classic 53-year-old woman recently menopausal with hot flashes, a so-called spontaneously or naturally menopausal woman. But my perspective is that unless there are clear contraindications, the decision to start systemic HT in the 41-year-old BSO case patient is actually not discretionary. Unless contraindications are present, it is important not only to treat symptoms but also to prevent an array of chronic major health concerns that are more likely if we don’t prescribe systemic HT.
Continue to: Health effects of not using HT...
Health effects of not using HT
Dr. Faubion: Dr. Kapoor, can you describe the potential long-term adverse health consequences of not using estrogen therapy? Say the same 41-year-old woman does not have many bothersome symptoms. What would you do?
Dr. Kapoor: Thank you for that important question. Building on what Dr. Kaunitz said, in these patients there are really 2 issues that can seem to be independent but are not: The first relates to the immediate consequences of lack of estrogen, ie, the menopause-related symptoms, but the second and perhaps the bigger issue is the long-term risk associated with estrogen deprivation.
The symptoms in these women are often obvious as they can be quite severe and abrupt; one day these women have normal hormone levels and the next day, after BSO, suddenly their hormones are very low. So if symptoms occur, they are usually hard to miss, simply because they are very drastic and very severe.
Historically, patients and their clinicians have targeted these symptoms. Patients experience menopausal symptoms, they seek treatment, and then the clinicians basically titrate the treatment to manage these symptoms. That misses the bigger issue, however, which is that premature estrogen deprivation leads to a host of chronic health conditions, as Dr. Kaunitz mentioned. These mainly include increased risk for cardiovascular disease, diabetes, hypertension, dyslipidemia, increased risk of mortality, dementia, and osteoporosis.
Fairly strong observational evidence suggests that use of estrogen therapy given in replacement doses—doses higher than those typically used in women after natural menopause, therefore considered replacement doses—helps mitigate the risk of some of these adverse health conditions.
In these women, the bigger goal really is to reinstate the hormonal milieu that exists prior to menopause. To your point, Dr. Faubion, if I have a patient who is younger than 46 years, who has her ovaries taken out, and even if she has zero symptoms (and sometimes that does happen), I would still make a case for this patient to utilize hormone therapy unless there is a contraindication such as breast cancer or other estrogen-sensitive cancers.
Dr. Faubion: Again, would you aim for those higher doses rather than treat with the “lowest dose”?
Dr. Kapoor: Absolutely. My punchline to the patients and clinicians in these discussions is that the rules of the game are different for these women. We cannot extrapolate the risks and benefits of HT use in women after natural menopause to younger women who have surgical menopause. Those rules just do not apply with respect to both benefits and risks.
Dr. Faubion: I think it’s important to say that these same “rules” would apply if the women were to go through premature menopause for any other reason, too, such as chemotherapy, radiation therapy, or premature ovarian insufficiency for any number of reasons, including toxic, metabolic, or genetic causes and so on. Would that be true?
Dr. Kapoor: Yes, absolutely so.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: In terms of practical or clinical issues regarding systemic HT management, for the woman in her early 50s who has experienced normal or natural spontaneous menopause, a starting dose of transdermal estradiol would be, for instance, a 0.05-mg patch, which is a patch that over 24 hours releases 0.05 mg of estradiol daily; or standard oral estrogen, including conjugated equine estrogen, a 0.625-mg tablet daily, or estradiol, a 1-mg tablet daily.
But in younger patients, we want to use higher doses. For a patch, for instance, I would aim for a 0.075- or 0.1-mg estradiol patch, which releases a higher daily dose of estradiol than the standard dose. For oral estrogen, the dose would be 0.9- or even 1.25-mg tablets of conjugated equine estrogen or 1.5 mg, which is a 1-mg plus a 0.5-mg estradiol tablet, or a 2-mg estradiol tablet. Estradiol does come in a 2-mg strength.
For oral estrogen, I prefer estradiol because it’s available as a generic medication and often available at a very low cost, sometimes as low as $4 a month from chain pharmacies.
Continue to: Usefulness of monitoring estradiol levels for dosage adjustment...
Usefulness of monitoring estradiol levels for dosage adjustment
Dr. Faubion: That’s a great point, and again it is important to emphasize that we are aiming to recreate the premenopausal hormonal milieu. If you were to check estradiol levels, that would be aiming for a premenopausal range of approximately 80 to 120 pg per mL. Dr. Kapoor, is there utility in monitoring estrogen levels?
Dr. Kapoor: Great question, Dr. Faubion, and as you know it’s a loaded one. We base this on empiric evidence. We know that if the hormonal milieu in a young patient is changed to a postmenopausal one, her risk for many chronic conditions is increased. So if we were to reinstate a premenopausal hormonal milieu, that risk would probably be reduced. It makes good sense to target an empiric goal of 80 to 120 pg per mL of estradiol, which is the average estradiol level in a premenopausal woman. If you were to ask me, however, are there randomized, controlled trial data to support this practice—that is, if you target that level, can you make sure that the risk of diabetes is lower or that the risk of heart disease is lower—that study has yet to be done, and it may not ever be done on a large scale. However, it intuitively makes good sense to target premenopausal estradiol levels.
Dr. Faubion: When might you check an estradiol level in this population? For example, if you are treating a patient with a 0.1-mg estradiol patch and she still has significant hot flashes, would it be useful to check the level?
Dr. Kapoor: It would. In my practice, I check estradiol levels on these patients on an annual basis, regardless of symptoms, but definitely in the patient who has symptoms. It makes good sense, because sometimes these patients don’t absorb the estrogen well, particularly if administered by the transdermal route.
A general rule of thumb is that in the average population, if a patient is on the 0.1-mg patch, for example, you would expect her level to be around 100. If it is much lower than that, which sometimes happens, that speaks for poor absorption. Options at that point would be to treat her with a higher dose patch, depending on what the level is, or switch to a different formulation, such as oral.
In instances in which I have treated patients with a 0.1-mg patch for example, and their estradiol levels are undetectable, that speaks for very poor absorption. For such patients I make a case for switching them to oral therapy. Most definitely that makes sense in a patient who is symptomatic despite treatment. But even for patients who don’t have symptoms, I like to target that level, acknowledging that there is no evidence as such to support this practice.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: Yes, a few practical points. Although patches are available in a wider array of doses than oral estrogen formulations, the highest dose available is 0.1 mg. It’s important for clinicians to recognize that while checking serum levels when indicated can be performed in women using transdermal estradiol or patches, in women who are using oral estrogen, checking blood levels is not going to work well because serum estrogen levels have a daily peak and valley in women who use oral versus transdermal estradiol.
I also wanted to talk about progestins. Although many patients who have had a BSO prior to spontaneous menopause also have had a hysterectomy, others have an intact uterus associated with their BSO, so progestins must be used along with estrogen. And if we are using higher-than-standard doses of estrogen, we also need to use higher-than-standard doses of progestin.
In that classic 53-year-old woman I referred to who had spontaneous normal menopause, if she is taking 1 mg of estradiol daily, or a 0.05-mg patch, or 0.625 mg of conjugated equine estrogen, 2.5 mg of medroxyprogesterone is fine. In fact, that showed excellent progestational protection of the endometrium in the Women’s Health Initiative and in other studies.
However, if we are going to use double the estrogen dose, we should increase the progestin dose too. In some of my patients on higher estrogen doses who have an intact uterus, I’ll use 5 or even 10 mg of daily medroxyprogesterone acetate to ensure adequate progestational suppression.
Dr. Faubion: Another practical tip is that if one is using conjugated equine estrogens, measuring the serum estradiol levels is not useful either.
Dr. Kaunitz: I agree.
Continue to: Oral contraceptives as replacement HT...
Oral contraceptives as replacement HT
Dr. Faubion: Would you comment on use of a birth control pill in this circumstance? Would it be optimal to use a postmenopausal HT regimen as opposed to a birth control pill or combined hormonal contraception?
Dr. Kapoor: In this younger population, sometimes it seems like a more socially acceptable decision to be on a birth control option than on menopausal HT. But there are some issues with being on a contraceptive regimen. One is that we end up using estrogen doses much higher than what is really needed for replacement purposes. It is also a nonphysiologic way of replacement in another sense—as opposed to estradiol, which is the main hormone made by the ovaries, the hormonal contraceptive regimens contain the synthetic estrogen ethinyl estradiol for the most part.
The other issue that is based on some weak evidence is that it appears that the bone health outcomes are probably inferior with combined hormonal contraception. For these reasons, regimens that are based on replacement doses of estradiol are preferred.
Dr. Faubion: Right, although the data are somewhat weak, I agree that thus far it seems optimal to utilize a postmenopausal regimen for various reasons. Dr. Kaunitz, anything to add?
Dr. Kaunitz: Yes, to underscore Dr. Kapoor’s point, a common oral contraceptive that contains 20 µg of ethinyl estradiol is substantially more estrogenic than 1.0 or 2.0 mg of micronized oral estradiol.
Also consider that a 20-µg ethinyl estradiol oral contraceptive may increase the risk of venous thromboembolism more than menopausal doses of oral estradiol, whether it be a micronized estradiol or conjugated equine estrogen.
Dr. Faubion: So the risk may be greater with oral combined hormonal contraception as well?
Dr. Kaunitz: One thing we can do is explain to our patients that their ovaries, prior to surgery or prior to induced menopause, were making substantial quantities of estradiol. Whether we prescribe a patch or oral micronized estradiol, this estrogen is identical to the hormone that their ovaries were making prior to surgery or induced menopause.
Breast cancer concerns
Dr. Faubion: Let’s consider a more complicated case. A 35-year-old woman has an identified BRCA1 mutation; she has not had any cancers but has undergone risk-reducing BSO and her uterus remains. Is this woman a candidate for HT? At what dose, and for how long? Dr. Kaunitz, why don’t you start.
Dr. Kaunitz: That is a challenging case but one that I think our readers will find interesting and maybe even provocative.
We know that women with BRCA1 mutations, the more common of the 2 BRCA mutations, have a very high risk of developing epithelial ovarian cancer at a young age. For this reason, our colleagues in medical oncology who specialize in hereditary ovarian/breast cancer syndromes recommend prophylactic risk-reducing—and I would also say lifesaving—BSO with or without hysterectomy for women with BRCA1 mutations.
However, over the years there has been tremendous reluctance among physicians caring for BRCA patients and the women themselves—I use the term “previvors” to describe BRCA carriers who have not been diagnosed with breast or ovarian cancer—to use HT after BSO because of concerns that HT might increase breast cancer risk in women who are already at high risk for breast cancer.
I assume, Dr. Faubion, that in this case the woman had gynecologic surgery but continues to have intact breasts. Is that correct?
Dr. Faubion: That is correct.
Dr. Kaunitz: Although the assumption has been that it is not safe to prescribe HT in this setting, in fact, the reported cohort studies that have looked at this issue have not found an elevated risk of breast cancer when replacement estrogen, with or without progestin, is prescribed to BRCA1 previvors with intact breasts.
Given what Dr. Kapoor said regarding the morbidity that is associated with BSO without replacement of physiologic estrogen, and also given the severe symptoms that so many of these young menopausal women experience, in my practice I do prescribe estrogen or estrogen-progestin therapy and focus on the higher target doses that we discussed for the earlier case patient who had a hysterectomy for abnormal uterine bleeding with adenomyosis.
Dr. Faubion: Dr. Kapoor, do you agree with this approach? How long would you continue therapy?
Dr. Kapoor: First, in this BRCA1 case we need to appreciate that the indication for the BSO is a legitimate one, in contrast to the first case in which the ovaries were removed in a patient whose average risk of ovarian cancer was low. It is important to recognize that surgery performed in this context is the right thing to do because it does significantly reduce the risk of ovarian cancer.
The second thing to appreciate is that while we reduce the risk of ovarian cancer significantly and make sure that these patients survive longer, it’s striking a fine balance in that you want to make sure that their morbidity is not increased as a result of premature estrogen deprivation.
As Dr. Kaunitz told us, the evidence that we have so far, which granted is not very robust but is fairly strong observational evidence, suggests that the risk of breast cancer is not elevated when these patients are treated with replacement doses of HT.
Having said that, I do have very strong discussions with my patients in this category about having the risk-reducing bilateral mastectomy also, because if they were to get breast cancer because of their increased genetic predisposition, the cancer is likely to grow faster if the patient is on HT. So one of my counseling points to patients is that they strongly consider bilateral mastectomy, which reduces their breast cancer risk by more than 90%. At the same time, I also strongly endorse using HT in replacement doses for the reasons that we have already stated.
Dr. Faubion: Continue HT until age 50 or 52?
Dr. Kapoor: Definitely until that age, and possibly longer, depending on their symptoms. The indications for treating beyond the age of natural menopause are much the same as for women who experience natural menopause.
Dr. Faubion: That is assuming they had a bilateral mastectomy?
Dr. Kapoor: Yes.
Continue to: Continuing HT until the age of natural menopause...
Continuing HT until the age of natural menopause
Dr. Kaunitz: Dr. Kapoor brings up the important point of duration of systemic HT. I agree that similar considerations apply both to the healthy 41-year-old who had a hysterectomy for abnormal uterine bleeding and to the 35-year-old who had risk-reducing surgery because of her BRCA1 mutation.
In the 2 cases, both to treat symptoms and to prevent chronic diseases, it makes sense to continue HT at least until the age of natural menopause. That is consistent with 2017 guidance from The North American Menopause Society (NAMS) position statement on the use of systemic HT, that is, continuing systemic HT at least until the age of natural menopause.3 Then at that point, continuing or discontinuing systemic HT becomes discretionary, and that would be true for both cases. If the patient is slender or has a strong family history of osteoporosis, that tends to push the patient more in terms of continuing systemic HT. Those are just some examples, and Dr. Kapoor may want to detail other relevant considerations.
Dr. Kapoor: I completely agree. The decision is driven by symptoms that are not otherwise well managed, for example, with nonhormone strategies. If we have any concerns utilizing HT beyond the age of natural menopause, then nonhormonal options can be considered; but sometimes those are not as effective. And bone health is very important. You want to avoid using bisphosphonates in younger women and reserve them for older patients in their late 60s and 70s. Hormone therapy use is a very reasonable strategy to prevent bone loss.
Dr. Kaunitz: It is also worth mentioning that sometimes the woman involved in shared decision making with her clinician decides to stop systemic HT. In that setting, should the patient start developing new-onset dyspareunia, vaginal dryness, or other genital or sexuality-related concerns, it takes very little for me to advise that she start low-dose local vaginal estrogen therapy.
Dr. Faubion: In either scenario, if a woman were to develop symptoms consistent with genitourinary syndrome of menopause (GSM), would you use vaginal estrogen in addition to the systemic estrogen or alone after the woman elected to discontinue systemic therapy?
Dr. Kapoor: Yes to both, I would say.
Dr. Kaunitz: As my patients using systemic HT age, often I will lower the dose. For instance, the dose I use in a 53-year-old will be higher than when she is 59 or 62. At the same time, as we lower the dose of systemic estrogen therapy, symptoms of vaginal atrophy or GSM often will appear, and these can be effectively treated by adding low-dose vaginal estrogen therapy. A number of my patients, particularly those who are on lower-than-standard doses of systemic HT, are also using low-dose vaginal estrogen therapy.
There is a “hybrid” product available: the 90-day estradiol vaginal ring. Estring is a low-dose, 2-mg, 90-day estradiol ring that is very useful, but it is effective only for treating GSM or vaginal atrophy. A second menopausal vaginal estradiol ring, Femring, is available in 2 doses: 0.05 mg/day and 0.1 mg/day. These are very effective in treating both systemic issues, such as vasomotor symptoms or prevention of osteoporosis, and very effective in treating GSM or vaginal atrophy. One problem is that Femring, depending on insurance coverage, can be very expensive. It’s not available as a generic, so for insurance or financial reasons I don’t often prescribe it. If I could remove those financial barriers, I would prescribe Femring more often because it is very useful.
Dr. Faubion: You raise an important point, and that is, for women who have been on HT for some time, clinicians often feel the need to slowly reduce the dose. Would you do that same thing, Dr. Kapoor, for a 40-year-old woman? Would you reduce the dose as she approaches age 50? Is there pressure that “she shouldn’t be on that much estrogen”?
Dr. Kapoor: No, I would not feel pressured until the patient turns at least 46. I bring up age 46 because the average age range for menopause is 46 to 55. After that, if there is any concern, we can decrease the dose to half and keep the patient on that until she turns 50 or 51. But most of my patients are on replacement doses until the average age of menopause, which is around 51 years, and that’s when you reduce the dose to that of the typical HT regimens used after natural menopause.
Sometimes patients are told something by a friend or they have read something and they worry about the risk of 2 things. One is breast cancer and the other is venous thromboembolism (VTE), and that may be why they want to be on a lower dose. I counsel patients that while the risk of VTE is real with HT, it is the women after natural menopause who are at risk—because age itself is a risk for VTE—and it also has to do with the kind of HT regimen that a patient is on. High doses of oral estrogens and certain progestogens increase the risk. But again, for estradiol used in replacement doses and the more common progestogens that we now use in practice, such as micronized progesterone, the risk is not the same. The same goes for breast cancer. My biggest message to patients and clinicians who take care of these patients is that the rules that apply to women after natural menopause just do not apply to this very different patient population.
Dr. Faubion: Thank you, Dr. Kaunitz and Dr. Kapoor, for sharing your knowledge and experience. ●
Systemic HT past the age of 65
Dr. Kaunitz: Another practical issue relates to long-term or extended use of systemic HT. It’s not infrequent in my practice to receive mail and faxes from insurance carriers of systemic HT users who are age 65 and older in which the company refers to the American Geriatrics Society’s Beers criteria for potentially inappropriate medication use in older adults,1 suggesting that systemic HT is inappropriate for all women over age 65. In this age group, I use lower doses if I am continuing systemic HT. But the good news is that both NAMS and the American College Obstetricians and Gynecologists indicate that arbitrarily stopping systemic HT at age 65 or for any other arbitrary reason is inappropriate, and that decisions about continuing or discontinuing therapy should be made on an individualized basis using shared decision making. That’s an important message for our readers.
Counseling regarding elective BSO
Dr. Faubion: One final note about elective BSO in the absence of a genetic mutation that predisposes to increased ovarian or breast cancer risk. Fortunately, we have seen rates of oophorectomy before the age of natural menopause decline, but what would your advice be to women or clinicians of these women who say they are “just afraid of ovarian cancer and would like to have their ovaries removed before the age of natural menopause”?
Dr. Kaunitz: If patients have increased anxiety about ovarian cancer and yet they themselves are not known to be at elevated risk, I emphasize that, fortunately, ovarian cancer is uncommon. It is much less common than other cancers the patient might be familiar with, such as breast or colon or lung cancer. I also emphasize that women who have given birth, particularly multiple times; women who nursed their infants; and women who have used combination hormonal contraceptives, particularly if long term, are at markedly lower risk for ovarian cancer as they get older. We are talking about an uncommon cancer that is even less common if women have given birth, nursed their infants, or used combination contraceptives long term.
Dr. Faubion: Dr. Kapoor, what would you say regarding the increased risk they might incur if they do have their ovaries out?
Dr. Kapoor: As Dr. Kaunitz said, this is an uncommon cancer, and pursuing something to reduce the risk of an uncommon cancer does not benefit the community. That is also my counseling point to patients.
I also talk to them extensively about the risk associated with the ovaries being removed, and I tell them that although we have the option of giving them HT, it is hard to replicate the magic of nature. No matter what concoction or regimen we use, we cannot ensure reinstating health to what it was in the premenopausal state, because estrogen has such myriad effects on the body in so many different organ systems.
Reference
1. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227-2246.

Women who undergo bilateral salpingo-oophorectomy (BSO) for various indications prior to menopause experience a rapid decline in ovarian hormone levels and consequent vasomotor and other menopausal symptoms. In addition, the resulting estrogen deprivation is associated with such long-term adverse outcomes as osteoporosis and cardiovascular morbidity.
OBG M
Surgical vs natural menopause
Stephanie Faubion, MD, MBA, NCMP: Since the Women’s Health Initiative study was published in 2002,2 many clinicians have been fearful of using systemic HT in menopausal women, and HT use has declined dramatically such that only about 4% to 6% of menopausal women are now receiving systemic HT. Importantly, however, a group of younger menopausal women also are not receiving HT, and that is women who undergo BSO before they reach the average age of menopause, which in the United States is about age 52; this is sometimes referred to as surgical menopause or early surgical menopause. Early surgical menopause has different connotations for long-term health risks than natural menopause at the average age, and we are here to discuss these health effects and their management.
My name is Stephanie Faubion, and I am a women’s health internist and the Chair of the Department of Medicine at Mayo Clinic in Jacksonville, Florida, and Director of Mayo Clinic Women’s Health. I am here with 2 of my esteemed colleagues, Dr. Andrew Kaunitz and Dr. Ekta Kapoor.
Andrew M. Kaunitz, MD, NCMP: Hello, I am an ObGyn with the University of Florida College of Medicine in Jacksonville, with particular interests in contraception, menopause, and gynecologic ultrasonography.
Ekta Kapoor, MBBS, NCMP: And I am an endocrinologist at Mayo Clinic in Rochester with a specific interest in menopause and hormone therapy. I am also the Assistant Director for Mayo Clinic Women’s Health.
Higher-than-standard estrogen doses needed in younger menopausal women
Dr. Faubion: Let’s consider a couple of cases so that we can illustrate some important points regarding hormone management in women who have undergone BSO before the age of natural menopause.
Our first case patient is a woman who is 41 years of age and, because of adenomyosis, she will undergo a hysterectomy. She tells her clinician that she is very concerned about ovarian cancer risk because one of her good friends recently was diagnosed with ovarian cancer, and together they decide to remove her ovaries at the time of hysterectomy. Notably, her ovaries were healthy.
The patient is now menopausal postsurgery, and she is having significant hot flashes and night sweats. She visits her local internist, who is concerned about initiating HT. She is otherwise a healthy woman and does not have any contraindications to HT. Dr. Kaunitz, what would you tell her internist?
Dr. Kaunitz: We are dealing with 2 different issues in terms of decision making about systemic HT for this 41-year-old who has undergone BSO. First, as you mentioned, Dr. Faubion, she has bothersome hot flashes, or vasomotor symptoms. Unless there are contraindications, systemic HT would be appropriate. Although I might start treatment at standard doses, and the accompanying TABLE depicts standard doses for the 2 most common oral estrogen formulations as well as transdermal estradiol, it’s important to recognize that younger menopausal women often will need to use higher-than-standard doses.

For example, for a 53-year-old woman who has been menopausal for a year or 2 and now has bothersome symptoms, I might start her on estradiol 1 mg tablets with progestin if a uterus is present. However, in this 41-year-old case patient, while I might start treatment at a standard dose, I would anticipate increasing to higher doses, such as 1.5 or 2 mg of daily estradiol until she feels her menopausal symptoms are adequately addressed.
Dr. Faubion: It is important to note that sometimes women with early BSO tend to have more severe vasomotor symptoms. Do you find that sometimes a higher dose is required just to manage symptoms, Dr. Kaunitz?
Dr. Kaunitz: Absolutely, yes. The decision whether or not to use systemic HT might be considered discretionary or elective in the classic 53-year-old woman recently menopausal with hot flashes, a so-called spontaneously or naturally menopausal woman. But my perspective is that unless there are clear contraindications, the decision to start systemic HT in the 41-year-old BSO case patient is actually not discretionary. Unless contraindications are present, it is important not only to treat symptoms but also to prevent an array of chronic major health concerns that are more likely if we don’t prescribe systemic HT.
Continue to: Health effects of not using HT...
Health effects of not using HT
Dr. Faubion: Dr. Kapoor, can you describe the potential long-term adverse health consequences of not using estrogen therapy? Say the same 41-year-old woman does not have many bothersome symptoms. What would you do?
Dr. Kapoor: Thank you for that important question. Building on what Dr. Kaunitz said, in these patients there are really 2 issues that can seem to be independent but are not: The first relates to the immediate consequences of lack of estrogen, ie, the menopause-related symptoms, but the second and perhaps the bigger issue is the long-term risk associated with estrogen deprivation.
The symptoms in these women are often obvious as they can be quite severe and abrupt; one day these women have normal hormone levels and the next day, after BSO, suddenly their hormones are very low. So if symptoms occur, they are usually hard to miss, simply because they are very drastic and very severe.
Historically, patients and their clinicians have targeted these symptoms. Patients experience menopausal symptoms, they seek treatment, and then the clinicians basically titrate the treatment to manage these symptoms. That misses the bigger issue, however, which is that premature estrogen deprivation leads to a host of chronic health conditions, as Dr. Kaunitz mentioned. These mainly include increased risk for cardiovascular disease, diabetes, hypertension, dyslipidemia, increased risk of mortality, dementia, and osteoporosis.
Fairly strong observational evidence suggests that use of estrogen therapy given in replacement doses—doses higher than those typically used in women after natural menopause, therefore considered replacement doses—helps mitigate the risk of some of these adverse health conditions.
In these women, the bigger goal really is to reinstate the hormonal milieu that exists prior to menopause. To your point, Dr. Faubion, if I have a patient who is younger than 46 years, who has her ovaries taken out, and even if she has zero symptoms (and sometimes that does happen), I would still make a case for this patient to utilize hormone therapy unless there is a contraindication such as breast cancer or other estrogen-sensitive cancers.
Dr. Faubion: Again, would you aim for those higher doses rather than treat with the “lowest dose”?
Dr. Kapoor: Absolutely. My punchline to the patients and clinicians in these discussions is that the rules of the game are different for these women. We cannot extrapolate the risks and benefits of HT use in women after natural menopause to younger women who have surgical menopause. Those rules just do not apply with respect to both benefits and risks.
Dr. Faubion: I think it’s important to say that these same “rules” would apply if the women were to go through premature menopause for any other reason, too, such as chemotherapy, radiation therapy, or premature ovarian insufficiency for any number of reasons, including toxic, metabolic, or genetic causes and so on. Would that be true?
Dr. Kapoor: Yes, absolutely so.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: In terms of practical or clinical issues regarding systemic HT management, for the woman in her early 50s who has experienced normal or natural spontaneous menopause, a starting dose of transdermal estradiol would be, for instance, a 0.05-mg patch, which is a patch that over 24 hours releases 0.05 mg of estradiol daily; or standard oral estrogen, including conjugated equine estrogen, a 0.625-mg tablet daily, or estradiol, a 1-mg tablet daily.
But in younger patients, we want to use higher doses. For a patch, for instance, I would aim for a 0.075- or 0.1-mg estradiol patch, which releases a higher daily dose of estradiol than the standard dose. For oral estrogen, the dose would be 0.9- or even 1.25-mg tablets of conjugated equine estrogen or 1.5 mg, which is a 1-mg plus a 0.5-mg estradiol tablet, or a 2-mg estradiol tablet. Estradiol does come in a 2-mg strength.
For oral estrogen, I prefer estradiol because it’s available as a generic medication and often available at a very low cost, sometimes as low as $4 a month from chain pharmacies.
Continue to: Usefulness of monitoring estradiol levels for dosage adjustment...
Usefulness of monitoring estradiol levels for dosage adjustment
Dr. Faubion: That’s a great point, and again it is important to emphasize that we are aiming to recreate the premenopausal hormonal milieu. If you were to check estradiol levels, that would be aiming for a premenopausal range of approximately 80 to 120 pg per mL. Dr. Kapoor, is there utility in monitoring estrogen levels?
Dr. Kapoor: Great question, Dr. Faubion, and as you know it’s a loaded one. We base this on empiric evidence. We know that if the hormonal milieu in a young patient is changed to a postmenopausal one, her risk for many chronic conditions is increased. So if we were to reinstate a premenopausal hormonal milieu, that risk would probably be reduced. It makes good sense to target an empiric goal of 80 to 120 pg per mL of estradiol, which is the average estradiol level in a premenopausal woman. If you were to ask me, however, are there randomized, controlled trial data to support this practice—that is, if you target that level, can you make sure that the risk of diabetes is lower or that the risk of heart disease is lower—that study has yet to be done, and it may not ever be done on a large scale. However, it intuitively makes good sense to target premenopausal estradiol levels.
Dr. Faubion: When might you check an estradiol level in this population? For example, if you are treating a patient with a 0.1-mg estradiol patch and she still has significant hot flashes, would it be useful to check the level?
Dr. Kapoor: It would. In my practice, I check estradiol levels on these patients on an annual basis, regardless of symptoms, but definitely in the patient who has symptoms. It makes good sense, because sometimes these patients don’t absorb the estrogen well, particularly if administered by the transdermal route.
A general rule of thumb is that in the average population, if a patient is on the 0.1-mg patch, for example, you would expect her level to be around 100. If it is much lower than that, which sometimes happens, that speaks for poor absorption. Options at that point would be to treat her with a higher dose patch, depending on what the level is, or switch to a different formulation, such as oral.
In instances in which I have treated patients with a 0.1-mg patch for example, and their estradiol levels are undetectable, that speaks for very poor absorption. For such patients I make a case for switching them to oral therapy. Most definitely that makes sense in a patient who is symptomatic despite treatment. But even for patients who don’t have symptoms, I like to target that level, acknowledging that there is no evidence as such to support this practice.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: Yes, a few practical points. Although patches are available in a wider array of doses than oral estrogen formulations, the highest dose available is 0.1 mg. It’s important for clinicians to recognize that while checking serum levels when indicated can be performed in women using transdermal estradiol or patches, in women who are using oral estrogen, checking blood levels is not going to work well because serum estrogen levels have a daily peak and valley in women who use oral versus transdermal estradiol.
I also wanted to talk about progestins. Although many patients who have had a BSO prior to spontaneous menopause also have had a hysterectomy, others have an intact uterus associated with their BSO, so progestins must be used along with estrogen. And if we are using higher-than-standard doses of estrogen, we also need to use higher-than-standard doses of progestin.
In that classic 53-year-old woman I referred to who had spontaneous normal menopause, if she is taking 1 mg of estradiol daily, or a 0.05-mg patch, or 0.625 mg of conjugated equine estrogen, 2.5 mg of medroxyprogesterone is fine. In fact, that showed excellent progestational protection of the endometrium in the Women’s Health Initiative and in other studies.
However, if we are going to use double the estrogen dose, we should increase the progestin dose too. In some of my patients on higher estrogen doses who have an intact uterus, I’ll use 5 or even 10 mg of daily medroxyprogesterone acetate to ensure adequate progestational suppression.
Dr. Faubion: Another practical tip is that if one is using conjugated equine estrogens, measuring the serum estradiol levels is not useful either.
Dr. Kaunitz: I agree.
Continue to: Oral contraceptives as replacement HT...
Oral contraceptives as replacement HT
Dr. Faubion: Would you comment on use of a birth control pill in this circumstance? Would it be optimal to use a postmenopausal HT regimen as opposed to a birth control pill or combined hormonal contraception?
Dr. Kapoor: In this younger population, sometimes it seems like a more socially acceptable decision to be on a birth control option than on menopausal HT. But there are some issues with being on a contraceptive regimen. One is that we end up using estrogen doses much higher than what is really needed for replacement purposes. It is also a nonphysiologic way of replacement in another sense—as opposed to estradiol, which is the main hormone made by the ovaries, the hormonal contraceptive regimens contain the synthetic estrogen ethinyl estradiol for the most part.
The other issue that is based on some weak evidence is that it appears that the bone health outcomes are probably inferior with combined hormonal contraception. For these reasons, regimens that are based on replacement doses of estradiol are preferred.
Dr. Faubion: Right, although the data are somewhat weak, I agree that thus far it seems optimal to utilize a postmenopausal regimen for various reasons. Dr. Kaunitz, anything to add?
Dr. Kaunitz: Yes, to underscore Dr. Kapoor’s point, a common oral contraceptive that contains 20 µg of ethinyl estradiol is substantially more estrogenic than 1.0 or 2.0 mg of micronized oral estradiol.
Also consider that a 20-µg ethinyl estradiol oral contraceptive may increase the risk of venous thromboembolism more than menopausal doses of oral estradiol, whether it be a micronized estradiol or conjugated equine estrogen.
Dr. Faubion: So the risk may be greater with oral combined hormonal contraception as well?
Dr. Kaunitz: One thing we can do is explain to our patients that their ovaries, prior to surgery or prior to induced menopause, were making substantial quantities of estradiol. Whether we prescribe a patch or oral micronized estradiol, this estrogen is identical to the hormone that their ovaries were making prior to surgery or induced menopause.
Breast cancer concerns
Dr. Faubion: Let’s consider a more complicated case. A 35-year-old woman has an identified BRCA1 mutation; she has not had any cancers but has undergone risk-reducing BSO and her uterus remains. Is this woman a candidate for HT? At what dose, and for how long? Dr. Kaunitz, why don’t you start.
Dr. Kaunitz: That is a challenging case but one that I think our readers will find interesting and maybe even provocative.
We know that women with BRCA1 mutations, the more common of the 2 BRCA mutations, have a very high risk of developing epithelial ovarian cancer at a young age. For this reason, our colleagues in medical oncology who specialize in hereditary ovarian/breast cancer syndromes recommend prophylactic risk-reducing—and I would also say lifesaving—BSO with or without hysterectomy for women with BRCA1 mutations.
However, over the years there has been tremendous reluctance among physicians caring for BRCA patients and the women themselves—I use the term “previvors” to describe BRCA carriers who have not been diagnosed with breast or ovarian cancer—to use HT after BSO because of concerns that HT might increase breast cancer risk in women who are already at high risk for breast cancer.
I assume, Dr. Faubion, that in this case the woman had gynecologic surgery but continues to have intact breasts. Is that correct?
Dr. Faubion: That is correct.
Dr. Kaunitz: Although the assumption has been that it is not safe to prescribe HT in this setting, in fact, the reported cohort studies that have looked at this issue have not found an elevated risk of breast cancer when replacement estrogen, with or without progestin, is prescribed to BRCA1 previvors with intact breasts.
Given what Dr. Kapoor said regarding the morbidity that is associated with BSO without replacement of physiologic estrogen, and also given the severe symptoms that so many of these young menopausal women experience, in my practice I do prescribe estrogen or estrogen-progestin therapy and focus on the higher target doses that we discussed for the earlier case patient who had a hysterectomy for abnormal uterine bleeding with adenomyosis.
Dr. Faubion: Dr. Kapoor, do you agree with this approach? How long would you continue therapy?
Dr. Kapoor: First, in this BRCA1 case we need to appreciate that the indication for the BSO is a legitimate one, in contrast to the first case in which the ovaries were removed in a patient whose average risk of ovarian cancer was low. It is important to recognize that surgery performed in this context is the right thing to do because it does significantly reduce the risk of ovarian cancer.
The second thing to appreciate is that while we reduce the risk of ovarian cancer significantly and make sure that these patients survive longer, it’s striking a fine balance in that you want to make sure that their morbidity is not increased as a result of premature estrogen deprivation.
As Dr. Kaunitz told us, the evidence that we have so far, which granted is not very robust but is fairly strong observational evidence, suggests that the risk of breast cancer is not elevated when these patients are treated with replacement doses of HT.
Having said that, I do have very strong discussions with my patients in this category about having the risk-reducing bilateral mastectomy also, because if they were to get breast cancer because of their increased genetic predisposition, the cancer is likely to grow faster if the patient is on HT. So one of my counseling points to patients is that they strongly consider bilateral mastectomy, which reduces their breast cancer risk by more than 90%. At the same time, I also strongly endorse using HT in replacement doses for the reasons that we have already stated.
Dr. Faubion: Continue HT until age 50 or 52?
Dr. Kapoor: Definitely until that age, and possibly longer, depending on their symptoms. The indications for treating beyond the age of natural menopause are much the same as for women who experience natural menopause.
Dr. Faubion: That is assuming they had a bilateral mastectomy?
Dr. Kapoor: Yes.
Continue to: Continuing HT until the age of natural menopause...
Continuing HT until the age of natural menopause
Dr. Kaunitz: Dr. Kapoor brings up the important point of duration of systemic HT. I agree that similar considerations apply both to the healthy 41-year-old who had a hysterectomy for abnormal uterine bleeding and to the 35-year-old who had risk-reducing surgery because of her BRCA1 mutation.
In the 2 cases, both to treat symptoms and to prevent chronic diseases, it makes sense to continue HT at least until the age of natural menopause. That is consistent with 2017 guidance from The North American Menopause Society (NAMS) position statement on the use of systemic HT, that is, continuing systemic HT at least until the age of natural menopause.3 Then at that point, continuing or discontinuing systemic HT becomes discretionary, and that would be true for both cases. If the patient is slender or has a strong family history of osteoporosis, that tends to push the patient more in terms of continuing systemic HT. Those are just some examples, and Dr. Kapoor may want to detail other relevant considerations.
Dr. Kapoor: I completely agree. The decision is driven by symptoms that are not otherwise well managed, for example, with nonhormone strategies. If we have any concerns utilizing HT beyond the age of natural menopause, then nonhormonal options can be considered; but sometimes those are not as effective. And bone health is very important. You want to avoid using bisphosphonates in younger women and reserve them for older patients in their late 60s and 70s. Hormone therapy use is a very reasonable strategy to prevent bone loss.
Dr. Kaunitz: It is also worth mentioning that sometimes the woman involved in shared decision making with her clinician decides to stop systemic HT. In that setting, should the patient start developing new-onset dyspareunia, vaginal dryness, or other genital or sexuality-related concerns, it takes very little for me to advise that she start low-dose local vaginal estrogen therapy.
Dr. Faubion: In either scenario, if a woman were to develop symptoms consistent with genitourinary syndrome of menopause (GSM), would you use vaginal estrogen in addition to the systemic estrogen or alone after the woman elected to discontinue systemic therapy?
Dr. Kapoor: Yes to both, I would say.
Dr. Kaunitz: As my patients using systemic HT age, often I will lower the dose. For instance, the dose I use in a 53-year-old will be higher than when she is 59 or 62. At the same time, as we lower the dose of systemic estrogen therapy, symptoms of vaginal atrophy or GSM often will appear, and these can be effectively treated by adding low-dose vaginal estrogen therapy. A number of my patients, particularly those who are on lower-than-standard doses of systemic HT, are also using low-dose vaginal estrogen therapy.
There is a “hybrid” product available: the 90-day estradiol vaginal ring. Estring is a low-dose, 2-mg, 90-day estradiol ring that is very useful, but it is effective only for treating GSM or vaginal atrophy. A second menopausal vaginal estradiol ring, Femring, is available in 2 doses: 0.05 mg/day and 0.1 mg/day. These are very effective in treating both systemic issues, such as vasomotor symptoms or prevention of osteoporosis, and very effective in treating GSM or vaginal atrophy. One problem is that Femring, depending on insurance coverage, can be very expensive. It’s not available as a generic, so for insurance or financial reasons I don’t often prescribe it. If I could remove those financial barriers, I would prescribe Femring more often because it is very useful.
Dr. Faubion: You raise an important point, and that is, for women who have been on HT for some time, clinicians often feel the need to slowly reduce the dose. Would you do that same thing, Dr. Kapoor, for a 40-year-old woman? Would you reduce the dose as she approaches age 50? Is there pressure that “she shouldn’t be on that much estrogen”?
Dr. Kapoor: No, I would not feel pressured until the patient turns at least 46. I bring up age 46 because the average age range for menopause is 46 to 55. After that, if there is any concern, we can decrease the dose to half and keep the patient on that until she turns 50 or 51. But most of my patients are on replacement doses until the average age of menopause, which is around 51 years, and that’s when you reduce the dose to that of the typical HT regimens used after natural menopause.
Sometimes patients are told something by a friend or they have read something and they worry about the risk of 2 things. One is breast cancer and the other is venous thromboembolism (VTE), and that may be why they want to be on a lower dose. I counsel patients that while the risk of VTE is real with HT, it is the women after natural menopause who are at risk—because age itself is a risk for VTE—and it also has to do with the kind of HT regimen that a patient is on. High doses of oral estrogens and certain progestogens increase the risk. But again, for estradiol used in replacement doses and the more common progestogens that we now use in practice, such as micronized progesterone, the risk is not the same. The same goes for breast cancer. My biggest message to patients and clinicians who take care of these patients is that the rules that apply to women after natural menopause just do not apply to this very different patient population.
Dr. Faubion: Thank you, Dr. Kaunitz and Dr. Kapoor, for sharing your knowledge and experience. ●
Systemic HT past the age of 65
Dr. Kaunitz: Another practical issue relates to long-term or extended use of systemic HT. It’s not infrequent in my practice to receive mail and faxes from insurance carriers of systemic HT users who are age 65 and older in which the company refers to the American Geriatrics Society’s Beers criteria for potentially inappropriate medication use in older adults,1 suggesting that systemic HT is inappropriate for all women over age 65. In this age group, I use lower doses if I am continuing systemic HT. But the good news is that both NAMS and the American College Obstetricians and Gynecologists indicate that arbitrarily stopping systemic HT at age 65 or for any other arbitrary reason is inappropriate, and that decisions about continuing or discontinuing therapy should be made on an individualized basis using shared decision making. That’s an important message for our readers.
Counseling regarding elective BSO
Dr. Faubion: One final note about elective BSO in the absence of a genetic mutation that predisposes to increased ovarian or breast cancer risk. Fortunately, we have seen rates of oophorectomy before the age of natural menopause decline, but what would your advice be to women or clinicians of these women who say they are “just afraid of ovarian cancer and would like to have their ovaries removed before the age of natural menopause”?
Dr. Kaunitz: If patients have increased anxiety about ovarian cancer and yet they themselves are not known to be at elevated risk, I emphasize that, fortunately, ovarian cancer is uncommon. It is much less common than other cancers the patient might be familiar with, such as breast or colon or lung cancer. I also emphasize that women who have given birth, particularly multiple times; women who nursed their infants; and women who have used combination hormonal contraceptives, particularly if long term, are at markedly lower risk for ovarian cancer as they get older. We are talking about an uncommon cancer that is even less common if women have given birth, nursed their infants, or used combination contraceptives long term.
Dr. Faubion: Dr. Kapoor, what would you say regarding the increased risk they might incur if they do have their ovaries out?
Dr. Kapoor: As Dr. Kaunitz said, this is an uncommon cancer, and pursuing something to reduce the risk of an uncommon cancer does not benefit the community. That is also my counseling point to patients.
I also talk to them extensively about the risk associated with the ovaries being removed, and I tell them that although we have the option of giving them HT, it is hard to replicate the magic of nature. No matter what concoction or regimen we use, we cannot ensure reinstating health to what it was in the premenopausal state, because estrogen has such myriad effects on the body in so many different organ systems.
Reference
1. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227-2246.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430.
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
- North American Menopause Society. The 2017 hormone therapy position statement of The North American Menopause Society. J North Am Menopause Soc. 2017;24: 728-753.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430.
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
- North American Menopause Society. The 2017 hormone therapy position statement of The North American Menopause Society. J North Am Menopause Soc. 2017;24: 728-753.
3D no-compression breast imaging, new STI treatment resources
Koning 3D Breast CT
Koning expects to submit trial data for their ongoing screening study to the FDA in Q1 2022.
For more information, visit https://www.koninghealth.com/en/
New STI treatment resources
Healthcare Effectiveness and Data Information Set (HEDIS) measures are performance improvement measures used for health plans to track various dimensions of care. In 2019, the HEDIS measure for chlamydia screening showed that commercial and Medicaid health plans had an average 52% screening rate among sexually active 16- to 24-year-old women. In an effort to increase screening rates among young women, the Centers for Disease Control and Prevention has implemented opt-out, or universal screening, for chlamydia. In order to aid clinicians in implementing this opt-out screening into their practices, the American Sexual Health Association and the National Chlamydia Coalition created resources that offer guidance, including using normalizing language with patients to explain the screening strategy. Providers can access these resources online (http://chlamydiacoalition.org/opt-out-screening/). Videos are offered and include case examples of how to speak with patients about universal screening, and printable documents are included that expand on ways that practices can improve screening rates.
For more information, visit http://chlamydiacoalition.org/opt-out-screening/.
Koning 3D Breast CT
Koning expects to submit trial data for their ongoing screening study to the FDA in Q1 2022.
For more information, visit https://www.koninghealth.com/en/
New STI treatment resources
Healthcare Effectiveness and Data Information Set (HEDIS) measures are performance improvement measures used for health plans to track various dimensions of care. In 2019, the HEDIS measure for chlamydia screening showed that commercial and Medicaid health plans had an average 52% screening rate among sexually active 16- to 24-year-old women. In an effort to increase screening rates among young women, the Centers for Disease Control and Prevention has implemented opt-out, or universal screening, for chlamydia. In order to aid clinicians in implementing this opt-out screening into their practices, the American Sexual Health Association and the National Chlamydia Coalition created resources that offer guidance, including using normalizing language with patients to explain the screening strategy. Providers can access these resources online (http://chlamydiacoalition.org/opt-out-screening/). Videos are offered and include case examples of how to speak with patients about universal screening, and printable documents are included that expand on ways that practices can improve screening rates.
For more information, visit http://chlamydiacoalition.org/opt-out-screening/.
Koning 3D Breast CT
Koning expects to submit trial data for their ongoing screening study to the FDA in Q1 2022.
For more information, visit https://www.koninghealth.com/en/
New STI treatment resources
Healthcare Effectiveness and Data Information Set (HEDIS) measures are performance improvement measures used for health plans to track various dimensions of care. In 2019, the HEDIS measure for chlamydia screening showed that commercial and Medicaid health plans had an average 52% screening rate among sexually active 16- to 24-year-old women. In an effort to increase screening rates among young women, the Centers for Disease Control and Prevention has implemented opt-out, or universal screening, for chlamydia. In order to aid clinicians in implementing this opt-out screening into their practices, the American Sexual Health Association and the National Chlamydia Coalition created resources that offer guidance, including using normalizing language with patients to explain the screening strategy. Providers can access these resources online (http://chlamydiacoalition.org/opt-out-screening/). Videos are offered and include case examples of how to speak with patients about universal screening, and printable documents are included that expand on ways that practices can improve screening rates.
For more information, visit http://chlamydiacoalition.org/opt-out-screening/.
Drospirenone vs norethindrone progestin-only pills. Is there a clear winner?
Contraception and family planning have improved the health of all people by reducing maternal mortality, improving maternal and child health through birth spacing, supporting full education attainment, and advancing workforce participation.1 Contraception is cost-effective and should be supported by all health insurers. One economic study reported that depending on the contraceptive method utilized, up to $7 of health care costs were saved for each dollar spent on contraceptive services and supplies.2
Progestin-only pills (POPs) are an important contraceptive option for people in the following situations who3:
- have a contraindication to estrogen-containing contraceptives
- are actively breastfeeding
- are less than 21 days since birth
- have a preference to avoid estrogen.
POPs are contraindicated for women who have breast cancer, abnormal uterine bleeding, or active liver disease and for women who are pregnant. A history of bariatric surgery with a malabsorption procedure (Roux-en-Y and biliopancreatic diversion) and the use of antiepileptic medications that are strong enzyme inducers are additional situations where the risk of POP may outweigh the benefit.3 Alternative progestin-only options include the subdermal etonogestrel implant, depot medroxyprogesterone acetate, and levonorgestrel-releasing intrauterine devices. These 3 options provide superior contraceptive efficacy to POP.
As a contraceptive, norethindrone at a dose of 0.35 mg daily has two major flaws:
- it does not reliably inhibit ovulation
- it has a short half-life.
In clinical studies, norethindrone inhibits ovulation in approximately 50% of cycles.4,5 Because norethindrone at a dose of 0.35 mg does not reliably inhibit ovulation it relies on additional mechanisms for contraceptive efficacy, including thickening of the cervical mucus to block sperm entry into the upper reproductive tract, reduced fallopian tube motility, and thinning of the endometrium.6
Norethindrone POP is formulated in packs of 28 pills containing 0.35 mg intended for daily continuous administration and no medication-free intervals. One rationale for the low dose of 0.35 mg in norethindrone POP is that it approximates the lowest dose with contraceptive efficacy for breastfeeding women, which has the benefit of minimizing exposure of the baby to the medication. Estrogen-progestin birth control pills containing norethindrone as the progestin reliably inhibit ovulation and have a minimum of 1 mg of norethindrone in each hormone pill. A POP with 1 mg of norethindrone per pill would likely have greater contraceptive efficacy. When taken daily, norethindrone acetate 5 mg (Aygestin) suppresses ovarian estrogen production, ovulation, and often causes cessation of uterine bleeding.7 The short half-life of norethindrone (7.7 hours) further exacerbates the problem of an insufficient daily dose.6 The standard guidance is that norethindrone must be taken at the same time every day, a goal that is nearly impossible to achieve. If a dose of norethindrone is taken >3 hours late, backup contraception is recommended for 48 hours.6
Drospirenone is a chemical analogue of spironolactone. Drospirenone is a progestin that suppresses LH and FSH and has anti-androgenic and partial anti-mineralocorticoid effects.8 Drospirenone POP contains 4 mg of a nonmicronized formulation that is believed to provide a pharmacologically similar area under the curve in drug metabolism studies to the 3 mg of micronized drospirenone, present in drospirenone-containing estrogen-progestin contraceptives.8 It is provided in a pack of 28 pills with 24 drospirenone pills and 4 pills without hormone. Drospirenone has a long half-life of 30 to 34 hours.8 If ≥2 drospirenone pills are missed, backup contraception is recommended for 7 days.9 The contraceptive effectiveness of drospirenone POP is thought to be similar to estrogen-progestin pills.8 Theoretically, drospirenone, acting as an anti-mineralocorticoid, can cause hyperkalemia. People with renal and adrenal insufficiency are most vulnerable to this adverse effect and should not be prescribed drospirenone. Women taking drospirenone and a medication that strongly inhibits CYP3A4, an enzyme involved in drospirenone degradation—including ketoconazole, indinavir, boceprevir, and clarithromycin—may have increased circulating levels of drospirenone and be at an increased risk of hyperkalemia. The US Food and Drug Administration (FDA) suggests that clinicians consider monitoring potassium concentration in women taking drospirenone who are also prescribed a strong CYP3A4 inhibitor.9 In people with normal renal and adrenal function, drospirenone-induced hyperkalemia is not commonly observed.9
Drospirenone 4 mg has been reported to not affect the natural balance of pro- and anti-coagulation factors in women.10 Drospirenone 4 mg daily has been reported to cause a modest decrease in systolic (-8 mm Hg) and diastolic (-5 mm Hg) blood pressure for women with a baseline blood pressure ≥130 mm Hg. Drospirenone 4 mg daily did not change blood pressure measurement in women with a baseline systolic blood pressure <130 mm Hg.11 For women using drospirenone POP, circulating estradiol concentration is usually >30 pg/mL, with a mean concentration of 51 pg/mL.12,13 Drospirenone POP does not result in a significant change in body weight.14 Preliminary studies suggest that drospirenone is an effective contraceptive in women with a BMI >30 kg/m2.14,15 Drospirenone enters breast milk and the relative infant dose is reported to be 1.5%.9 In general, breastfeeding is considered reasonably safe when the relative infant dose of a medication is <10%.16
The most common adverse effect reported with both norethindrone and drospirenone POP is unscheduled uterine bleeding. With norethindrone POP about 50% of users have a relatively preserved monthly bleeding pattern and approximately 50% have bleeding between periods, spotting and/or prolonged bleeding.17,18 A similar frequency of unscheduled uterine bleeding has been reported with drospirenone POP.14,19 Unscheduled and bothersome uterine bleeding is a common reason people discontinue POP. For drospirenone POP, the FDA reports a Pearl Index of 4.9 Other studies report a Pearl Index of 0.73 (95% confidence interval [CI], 0.31 to 1.43) for drospirenone POP.14 For norethindrone POP, the FDA reports that in typical use about 5% of people using the contraceptive method would become pregnant.6 The TABLE provides a comparison of the key features of the two available POP contraceptives. My assessment is that drospirenone has superior contraceptive properties over norethindrone POP. However, a head-to-head clinical trial would be necessary to determine the relative contraceptive effectiveness of drospirenone versus norethindrone POP.
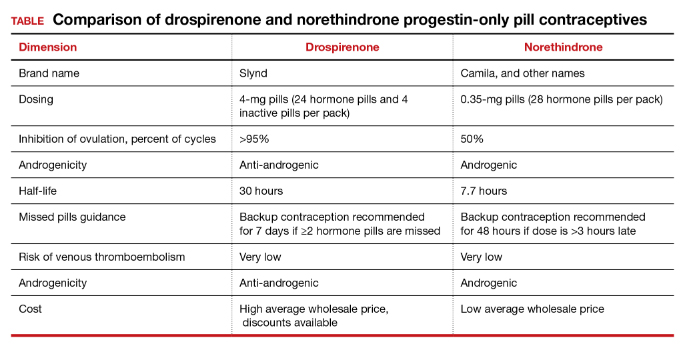
Maintaining contraception access
Access to contraception without a copayment is an important component of a comprehensive and equitable insurance program.20 The American College of Obstetricians and Gynecologists (ACOG) advocates that all people “should have unhindered and affordable access to all U.S. Food and Drug Administration-approved contraceptives.”21 ACOG also calls for the “full implementation of the Affordable Care Act requirement that new and revised private health insurance plans cover all U.S. Food and Drug Administration approved contraceptives without cost sharing, including nonequivalent options within one method category.” The National Women’s Law Center22 provides helpful resources to ensure access to legislated contraceptive benefits, including a phone script for speaking with an insurance benefits agent23 and a toolkit for advocating for your contraceptive choice.24 We need to ensure that people have unfettered access to all FDA-approved contraceptives because access to contraception is an important component of public health. Although drospirenone is more costly than norethindrone POP, drospirenone contraception should be available to all patients seeking POP contraception. ●
- Kavanaugh ML, Andreson RM. Contraception and beyond: the health benefits of services provided at family planning centers, NY. Guttmacher Institute. 2013. www.gutmacher.org/pubs/helth-benefits.pdf. Accessed January 13, 2022.
- Foster DG, Rostovtseva DP, Brindis CD, et al. Cost savings from the provision of specific methods of contraception in a publicly funded program. Am J Pub Health. 2009;99:446-451.
- Curtis M, Tepper NK, Jatlaoui TC, et al. U.S. Medical eligibility criteria for contraceptive use, 2016. MMWR Recomm Rep. 2016;65:1-103.
- Rice CF, Killick SR, Dieben T, et al. A comparison of the inhibition of ovulation achieved by desogestrel 75 µg and levonorgestrel 30 µg daily. Human Reprod. 1999;14:982-985.
- Milsom I, Korver T. Ovulation incidence with oral contraceptives: a literature review. J Fam Plann Reprod Health Care. 2008;34:237-246.
- OrthoMicronor [package insert]. OrthoMcNeil: Raritan, New Jersey. June 2008.
- Brown JB, Fotherby K, Loraine JA. The effect of norethisterone and its acetate on ovarian and pituitary function during the menstrual cycle. J Endocrinol. 1962;25:331-341.
- Romer T, Bitzer J, Egarter C, et al. Oral progestins in hormonal contraception: importance and future perspectives of a new progestin only-pill containing 4 mg drospirenone. Geburtsch Frauenheilk. 2021;81:1021-1030.
- Slynd [package insert]. Exeltis: Florham Park, New Jersey. May 2019.
- Regidor PA, Colli E, Schindlre AE. Drospirenone as estrogen-free pill and hemostasis: coagulatory study results comparing a novel 4 mg formulation in a 24+4 cycle with desogestrel 75 µg per day. Gynecol Endocrinol. 2016;32:749-751.
- Palacios S, Colli E, Regidor PA. Efficacy and cardiovascular safety of the new estrogen-free contraceptive pill containing 4 mg drospirenone alone in a 24/4 regime. BMC Womens Health. 2020;20:218.
- Hadji P, Colli E, Regidor PA. Bone health in estrogen-free contraception. Osteoporosis Int. 2019;30:2391-2400.
- Mitchell VE, Welling LM. Not all progestins are created equally: considering unique progestins individually in psychobehavioral research. Adapt Human Behav Physiol. 2020;6:381-412.
- Palacios S, Colli E, Regidor PA. Multicenter, phase III trials on the contraceptive efficacy, tolerability and safety of a new drospirenone-only pill. Acta Obstet Gynecol Scand. 2019;98:1549-1557.
- Archer DF, Ahrendt HJ, Drouin D. Drospirenone-only oral contraceptive: results from a multicenter noncomparative trial of efficacy, safety and tolerability. Contraception. 2015;92:439-444.
- Anderson PO, Sauberan JB. Modeling drug passage into human milk. Clin Pharmacol Ther. 2016;100:42-52. doi: 10.1002/cpt.377.
- Belsey EM. Vaginal bleeding patterns among women using one natural and eight hormonal methods of contraception. Contraception. 1988;38:181-206.
- Broome M, Fotherby K. Clinical experience with the progestin-only pill. Contraception. 1990;42:489-495.
- Apter D, Colli E, Gemzell-Danielsson K, et al. Multicenter, open-label trial to assess the safety and tolerability of drospirenone 4.0 mg over 6 cycles in female adolescents with a 7-cycle extension phase. Contraception. 2020;101:412.
- Birth control benefits. Healthcare.gov website. https://www.healthcare.gov/coverage/birth-control-benefits/. Accessed January 13, 2022.
- American College of Obstetricians and Gynecologists. Access to contraception. Committee Opinion No. 615. Obstet Gynecol. 2015;125:250-256.
- Health care and reproductive rights. National Women’s Law Center website. https://nwlc.org/issue/health-care. Accessed January 13, 2022.
- How to find out if your health plan covers birth control at no cost to you. National Women’s Law Center website. https://nwlc.org/sites/default/files/072014-insuranceflowchart_vupdated.pdf. Accessed January 13, 2022.
- Toolkit: Getting the coverage you deserve. National Women’s Law Center website. https://nwlc.org/sites/default/files/pdfs/final_nwlclogo_preventive servicestoolkit_9-25-13.pdf. Accessed January 13, 2022.
Contraception and family planning have improved the health of all people by reducing maternal mortality, improving maternal and child health through birth spacing, supporting full education attainment, and advancing workforce participation.1 Contraception is cost-effective and should be supported by all health insurers. One economic study reported that depending on the contraceptive method utilized, up to $7 of health care costs were saved for each dollar spent on contraceptive services and supplies.2
Progestin-only pills (POPs) are an important contraceptive option for people in the following situations who3:
- have a contraindication to estrogen-containing contraceptives
- are actively breastfeeding
- are less than 21 days since birth
- have a preference to avoid estrogen.
POPs are contraindicated for women who have breast cancer, abnormal uterine bleeding, or active liver disease and for women who are pregnant. A history of bariatric surgery with a malabsorption procedure (Roux-en-Y and biliopancreatic diversion) and the use of antiepileptic medications that are strong enzyme inducers are additional situations where the risk of POP may outweigh the benefit.3 Alternative progestin-only options include the subdermal etonogestrel implant, depot medroxyprogesterone acetate, and levonorgestrel-releasing intrauterine devices. These 3 options provide superior contraceptive efficacy to POP.
As a contraceptive, norethindrone at a dose of 0.35 mg daily has two major flaws:
- it does not reliably inhibit ovulation
- it has a short half-life.
In clinical studies, norethindrone inhibits ovulation in approximately 50% of cycles.4,5 Because norethindrone at a dose of 0.35 mg does not reliably inhibit ovulation it relies on additional mechanisms for contraceptive efficacy, including thickening of the cervical mucus to block sperm entry into the upper reproductive tract, reduced fallopian tube motility, and thinning of the endometrium.6
Norethindrone POP is formulated in packs of 28 pills containing 0.35 mg intended for daily continuous administration and no medication-free intervals. One rationale for the low dose of 0.35 mg in norethindrone POP is that it approximates the lowest dose with contraceptive efficacy for breastfeeding women, which has the benefit of minimizing exposure of the baby to the medication. Estrogen-progestin birth control pills containing norethindrone as the progestin reliably inhibit ovulation and have a minimum of 1 mg of norethindrone in each hormone pill. A POP with 1 mg of norethindrone per pill would likely have greater contraceptive efficacy. When taken daily, norethindrone acetate 5 mg (Aygestin) suppresses ovarian estrogen production, ovulation, and often causes cessation of uterine bleeding.7 The short half-life of norethindrone (7.7 hours) further exacerbates the problem of an insufficient daily dose.6 The standard guidance is that norethindrone must be taken at the same time every day, a goal that is nearly impossible to achieve. If a dose of norethindrone is taken >3 hours late, backup contraception is recommended for 48 hours.6
Drospirenone is a chemical analogue of spironolactone. Drospirenone is a progestin that suppresses LH and FSH and has anti-androgenic and partial anti-mineralocorticoid effects.8 Drospirenone POP contains 4 mg of a nonmicronized formulation that is believed to provide a pharmacologically similar area under the curve in drug metabolism studies to the 3 mg of micronized drospirenone, present in drospirenone-containing estrogen-progestin contraceptives.8 It is provided in a pack of 28 pills with 24 drospirenone pills and 4 pills without hormone. Drospirenone has a long half-life of 30 to 34 hours.8 If ≥2 drospirenone pills are missed, backup contraception is recommended for 7 days.9 The contraceptive effectiveness of drospirenone POP is thought to be similar to estrogen-progestin pills.8 Theoretically, drospirenone, acting as an anti-mineralocorticoid, can cause hyperkalemia. People with renal and adrenal insufficiency are most vulnerable to this adverse effect and should not be prescribed drospirenone. Women taking drospirenone and a medication that strongly inhibits CYP3A4, an enzyme involved in drospirenone degradation—including ketoconazole, indinavir, boceprevir, and clarithromycin—may have increased circulating levels of drospirenone and be at an increased risk of hyperkalemia. The US Food and Drug Administration (FDA) suggests that clinicians consider monitoring potassium concentration in women taking drospirenone who are also prescribed a strong CYP3A4 inhibitor.9 In people with normal renal and adrenal function, drospirenone-induced hyperkalemia is not commonly observed.9
Drospirenone 4 mg has been reported to not affect the natural balance of pro- and anti-coagulation factors in women.10 Drospirenone 4 mg daily has been reported to cause a modest decrease in systolic (-8 mm Hg) and diastolic (-5 mm Hg) blood pressure for women with a baseline blood pressure ≥130 mm Hg. Drospirenone 4 mg daily did not change blood pressure measurement in women with a baseline systolic blood pressure <130 mm Hg.11 For women using drospirenone POP, circulating estradiol concentration is usually >30 pg/mL, with a mean concentration of 51 pg/mL.12,13 Drospirenone POP does not result in a significant change in body weight.14 Preliminary studies suggest that drospirenone is an effective contraceptive in women with a BMI >30 kg/m2.14,15 Drospirenone enters breast milk and the relative infant dose is reported to be 1.5%.9 In general, breastfeeding is considered reasonably safe when the relative infant dose of a medication is <10%.16
The most common adverse effect reported with both norethindrone and drospirenone POP is unscheduled uterine bleeding. With norethindrone POP about 50% of users have a relatively preserved monthly bleeding pattern and approximately 50% have bleeding between periods, spotting and/or prolonged bleeding.17,18 A similar frequency of unscheduled uterine bleeding has been reported with drospirenone POP.14,19 Unscheduled and bothersome uterine bleeding is a common reason people discontinue POP. For drospirenone POP, the FDA reports a Pearl Index of 4.9 Other studies report a Pearl Index of 0.73 (95% confidence interval [CI], 0.31 to 1.43) for drospirenone POP.14 For norethindrone POP, the FDA reports that in typical use about 5% of people using the contraceptive method would become pregnant.6 The TABLE provides a comparison of the key features of the two available POP contraceptives. My assessment is that drospirenone has superior contraceptive properties over norethindrone POP. However, a head-to-head clinical trial would be necessary to determine the relative contraceptive effectiveness of drospirenone versus norethindrone POP.

Maintaining contraception access
Access to contraception without a copayment is an important component of a comprehensive and equitable insurance program.20 The American College of Obstetricians and Gynecologists (ACOG) advocates that all people “should have unhindered and affordable access to all U.S. Food and Drug Administration-approved contraceptives.”21 ACOG also calls for the “full implementation of the Affordable Care Act requirement that new and revised private health insurance plans cover all U.S. Food and Drug Administration approved contraceptives without cost sharing, including nonequivalent options within one method category.” The National Women’s Law Center22 provides helpful resources to ensure access to legislated contraceptive benefits, including a phone script for speaking with an insurance benefits agent23 and a toolkit for advocating for your contraceptive choice.24 We need to ensure that people have unfettered access to all FDA-approved contraceptives because access to contraception is an important component of public health. Although drospirenone is more costly than norethindrone POP, drospirenone contraception should be available to all patients seeking POP contraception. ●
Contraception and family planning have improved the health of all people by reducing maternal mortality, improving maternal and child health through birth spacing, supporting full education attainment, and advancing workforce participation.1 Contraception is cost-effective and should be supported by all health insurers. One economic study reported that depending on the contraceptive method utilized, up to $7 of health care costs were saved for each dollar spent on contraceptive services and supplies.2
Progestin-only pills (POPs) are an important contraceptive option for people in the following situations who3:
- have a contraindication to estrogen-containing contraceptives
- are actively breastfeeding
- are less than 21 days since birth
- have a preference to avoid estrogen.
POPs are contraindicated for women who have breast cancer, abnormal uterine bleeding, or active liver disease and for women who are pregnant. A history of bariatric surgery with a malabsorption procedure (Roux-en-Y and biliopancreatic diversion) and the use of antiepileptic medications that are strong enzyme inducers are additional situations where the risk of POP may outweigh the benefit.3 Alternative progestin-only options include the subdermal etonogestrel implant, depot medroxyprogesterone acetate, and levonorgestrel-releasing intrauterine devices. These 3 options provide superior contraceptive efficacy to POP.
As a contraceptive, norethindrone at a dose of 0.35 mg daily has two major flaws:
- it does not reliably inhibit ovulation
- it has a short half-life.
In clinical studies, norethindrone inhibits ovulation in approximately 50% of cycles.4,5 Because norethindrone at a dose of 0.35 mg does not reliably inhibit ovulation it relies on additional mechanisms for contraceptive efficacy, including thickening of the cervical mucus to block sperm entry into the upper reproductive tract, reduced fallopian tube motility, and thinning of the endometrium.6
Norethindrone POP is formulated in packs of 28 pills containing 0.35 mg intended for daily continuous administration and no medication-free intervals. One rationale for the low dose of 0.35 mg in norethindrone POP is that it approximates the lowest dose with contraceptive efficacy for breastfeeding women, which has the benefit of minimizing exposure of the baby to the medication. Estrogen-progestin birth control pills containing norethindrone as the progestin reliably inhibit ovulation and have a minimum of 1 mg of norethindrone in each hormone pill. A POP with 1 mg of norethindrone per pill would likely have greater contraceptive efficacy. When taken daily, norethindrone acetate 5 mg (Aygestin) suppresses ovarian estrogen production, ovulation, and often causes cessation of uterine bleeding.7 The short half-life of norethindrone (7.7 hours) further exacerbates the problem of an insufficient daily dose.6 The standard guidance is that norethindrone must be taken at the same time every day, a goal that is nearly impossible to achieve. If a dose of norethindrone is taken >3 hours late, backup contraception is recommended for 48 hours.6
Drospirenone is a chemical analogue of spironolactone. Drospirenone is a progestin that suppresses LH and FSH and has anti-androgenic and partial anti-mineralocorticoid effects.8 Drospirenone POP contains 4 mg of a nonmicronized formulation that is believed to provide a pharmacologically similar area under the curve in drug metabolism studies to the 3 mg of micronized drospirenone, present in drospirenone-containing estrogen-progestin contraceptives.8 It is provided in a pack of 28 pills with 24 drospirenone pills and 4 pills without hormone. Drospirenone has a long half-life of 30 to 34 hours.8 If ≥2 drospirenone pills are missed, backup contraception is recommended for 7 days.9 The contraceptive effectiveness of drospirenone POP is thought to be similar to estrogen-progestin pills.8 Theoretically, drospirenone, acting as an anti-mineralocorticoid, can cause hyperkalemia. People with renal and adrenal insufficiency are most vulnerable to this adverse effect and should not be prescribed drospirenone. Women taking drospirenone and a medication that strongly inhibits CYP3A4, an enzyme involved in drospirenone degradation—including ketoconazole, indinavir, boceprevir, and clarithromycin—may have increased circulating levels of drospirenone and be at an increased risk of hyperkalemia. The US Food and Drug Administration (FDA) suggests that clinicians consider monitoring potassium concentration in women taking drospirenone who are also prescribed a strong CYP3A4 inhibitor.9 In people with normal renal and adrenal function, drospirenone-induced hyperkalemia is not commonly observed.9
Drospirenone 4 mg has been reported to not affect the natural balance of pro- and anti-coagulation factors in women.10 Drospirenone 4 mg daily has been reported to cause a modest decrease in systolic (-8 mm Hg) and diastolic (-5 mm Hg) blood pressure for women with a baseline blood pressure ≥130 mm Hg. Drospirenone 4 mg daily did not change blood pressure measurement in women with a baseline systolic blood pressure <130 mm Hg.11 For women using drospirenone POP, circulating estradiol concentration is usually >30 pg/mL, with a mean concentration of 51 pg/mL.12,13 Drospirenone POP does not result in a significant change in body weight.14 Preliminary studies suggest that drospirenone is an effective contraceptive in women with a BMI >30 kg/m2.14,15 Drospirenone enters breast milk and the relative infant dose is reported to be 1.5%.9 In general, breastfeeding is considered reasonably safe when the relative infant dose of a medication is <10%.16
The most common adverse effect reported with both norethindrone and drospirenone POP is unscheduled uterine bleeding. With norethindrone POP about 50% of users have a relatively preserved monthly bleeding pattern and approximately 50% have bleeding between periods, spotting and/or prolonged bleeding.17,18 A similar frequency of unscheduled uterine bleeding has been reported with drospirenone POP.14,19 Unscheduled and bothersome uterine bleeding is a common reason people discontinue POP. For drospirenone POP, the FDA reports a Pearl Index of 4.9 Other studies report a Pearl Index of 0.73 (95% confidence interval [CI], 0.31 to 1.43) for drospirenone POP.14 For norethindrone POP, the FDA reports that in typical use about 5% of people using the contraceptive method would become pregnant.6 The TABLE provides a comparison of the key features of the two available POP contraceptives. My assessment is that drospirenone has superior contraceptive properties over norethindrone POP. However, a head-to-head clinical trial would be necessary to determine the relative contraceptive effectiveness of drospirenone versus norethindrone POP.

Maintaining contraception access
Access to contraception without a copayment is an important component of a comprehensive and equitable insurance program.20 The American College of Obstetricians and Gynecologists (ACOG) advocates that all people “should have unhindered and affordable access to all U.S. Food and Drug Administration-approved contraceptives.”21 ACOG also calls for the “full implementation of the Affordable Care Act requirement that new and revised private health insurance plans cover all U.S. Food and Drug Administration approved contraceptives without cost sharing, including nonequivalent options within one method category.” The National Women’s Law Center22 provides helpful resources to ensure access to legislated contraceptive benefits, including a phone script for speaking with an insurance benefits agent23 and a toolkit for advocating for your contraceptive choice.24 We need to ensure that people have unfettered access to all FDA-approved contraceptives because access to contraception is an important component of public health. Although drospirenone is more costly than norethindrone POP, drospirenone contraception should be available to all patients seeking POP contraception. ●
- Kavanaugh ML, Andreson RM. Contraception and beyond: the health benefits of services provided at family planning centers, NY. Guttmacher Institute. 2013. www.gutmacher.org/pubs/helth-benefits.pdf. Accessed January 13, 2022.
- Foster DG, Rostovtseva DP, Brindis CD, et al. Cost savings from the provision of specific methods of contraception in a publicly funded program. Am J Pub Health. 2009;99:446-451.
- Curtis M, Tepper NK, Jatlaoui TC, et al. U.S. Medical eligibility criteria for contraceptive use, 2016. MMWR Recomm Rep. 2016;65:1-103.
- Rice CF, Killick SR, Dieben T, et al. A comparison of the inhibition of ovulation achieved by desogestrel 75 µg and levonorgestrel 30 µg daily. Human Reprod. 1999;14:982-985.
- Milsom I, Korver T. Ovulation incidence with oral contraceptives: a literature review. J Fam Plann Reprod Health Care. 2008;34:237-246.
- OrthoMicronor [package insert]. OrthoMcNeil: Raritan, New Jersey. June 2008.
- Brown JB, Fotherby K, Loraine JA. The effect of norethisterone and its acetate on ovarian and pituitary function during the menstrual cycle. J Endocrinol. 1962;25:331-341.
- Romer T, Bitzer J, Egarter C, et al. Oral progestins in hormonal contraception: importance and future perspectives of a new progestin only-pill containing 4 mg drospirenone. Geburtsch Frauenheilk. 2021;81:1021-1030.
- Slynd [package insert]. Exeltis: Florham Park, New Jersey. May 2019.
- Regidor PA, Colli E, Schindlre AE. Drospirenone as estrogen-free pill and hemostasis: coagulatory study results comparing a novel 4 mg formulation in a 24+4 cycle with desogestrel 75 µg per day. Gynecol Endocrinol. 2016;32:749-751.
- Palacios S, Colli E, Regidor PA. Efficacy and cardiovascular safety of the new estrogen-free contraceptive pill containing 4 mg drospirenone alone in a 24/4 regime. BMC Womens Health. 2020;20:218.
- Hadji P, Colli E, Regidor PA. Bone health in estrogen-free contraception. Osteoporosis Int. 2019;30:2391-2400.
- Mitchell VE, Welling LM. Not all progestins are created equally: considering unique progestins individually in psychobehavioral research. Adapt Human Behav Physiol. 2020;6:381-412.
- Palacios S, Colli E, Regidor PA. Multicenter, phase III trials on the contraceptive efficacy, tolerability and safety of a new drospirenone-only pill. Acta Obstet Gynecol Scand. 2019;98:1549-1557.
- Archer DF, Ahrendt HJ, Drouin D. Drospirenone-only oral contraceptive: results from a multicenter noncomparative trial of efficacy, safety and tolerability. Contraception. 2015;92:439-444.
- Anderson PO, Sauberan JB. Modeling drug passage into human milk. Clin Pharmacol Ther. 2016;100:42-52. doi: 10.1002/cpt.377.
- Belsey EM. Vaginal bleeding patterns among women using one natural and eight hormonal methods of contraception. Contraception. 1988;38:181-206.
- Broome M, Fotherby K. Clinical experience with the progestin-only pill. Contraception. 1990;42:489-495.
- Apter D, Colli E, Gemzell-Danielsson K, et al. Multicenter, open-label trial to assess the safety and tolerability of drospirenone 4.0 mg over 6 cycles in female adolescents with a 7-cycle extension phase. Contraception. 2020;101:412.
- Birth control benefits. Healthcare.gov website. https://www.healthcare.gov/coverage/birth-control-benefits/. Accessed January 13, 2022.
- American College of Obstetricians and Gynecologists. Access to contraception. Committee Opinion No. 615. Obstet Gynecol. 2015;125:250-256.
- Health care and reproductive rights. National Women’s Law Center website. https://nwlc.org/issue/health-care. Accessed January 13, 2022.
- How to find out if your health plan covers birth control at no cost to you. National Women’s Law Center website. https://nwlc.org/sites/default/files/072014-insuranceflowchart_vupdated.pdf. Accessed January 13, 2022.
- Toolkit: Getting the coverage you deserve. National Women’s Law Center website. https://nwlc.org/sites/default/files/pdfs/final_nwlclogo_preventive servicestoolkit_9-25-13.pdf. Accessed January 13, 2022.
- Kavanaugh ML, Andreson RM. Contraception and beyond: the health benefits of services provided at family planning centers, NY. Guttmacher Institute. 2013. www.gutmacher.org/pubs/helth-benefits.pdf. Accessed January 13, 2022.
- Foster DG, Rostovtseva DP, Brindis CD, et al. Cost savings from the provision of specific methods of contraception in a publicly funded program. Am J Pub Health. 2009;99:446-451.
- Curtis M, Tepper NK, Jatlaoui TC, et al. U.S. Medical eligibility criteria for contraceptive use, 2016. MMWR Recomm Rep. 2016;65:1-103.
- Rice CF, Killick SR, Dieben T, et al. A comparison of the inhibition of ovulation achieved by desogestrel 75 µg and levonorgestrel 30 µg daily. Human Reprod. 1999;14:982-985.
- Milsom I, Korver T. Ovulation incidence with oral contraceptives: a literature review. J Fam Plann Reprod Health Care. 2008;34:237-246.
- OrthoMicronor [package insert]. OrthoMcNeil: Raritan, New Jersey. June 2008.
- Brown JB, Fotherby K, Loraine JA. The effect of norethisterone and its acetate on ovarian and pituitary function during the menstrual cycle. J Endocrinol. 1962;25:331-341.
- Romer T, Bitzer J, Egarter C, et al. Oral progestins in hormonal contraception: importance and future perspectives of a new progestin only-pill containing 4 mg drospirenone. Geburtsch Frauenheilk. 2021;81:1021-1030.
- Slynd [package insert]. Exeltis: Florham Park, New Jersey. May 2019.
- Regidor PA, Colli E, Schindlre AE. Drospirenone as estrogen-free pill and hemostasis: coagulatory study results comparing a novel 4 mg formulation in a 24+4 cycle with desogestrel 75 µg per day. Gynecol Endocrinol. 2016;32:749-751.
- Palacios S, Colli E, Regidor PA. Efficacy and cardiovascular safety of the new estrogen-free contraceptive pill containing 4 mg drospirenone alone in a 24/4 regime. BMC Womens Health. 2020;20:218.
- Hadji P, Colli E, Regidor PA. Bone health in estrogen-free contraception. Osteoporosis Int. 2019;30:2391-2400.
- Mitchell VE, Welling LM. Not all progestins are created equally: considering unique progestins individually in psychobehavioral research. Adapt Human Behav Physiol. 2020;6:381-412.
- Palacios S, Colli E, Regidor PA. Multicenter, phase III trials on the contraceptive efficacy, tolerability and safety of a new drospirenone-only pill. Acta Obstet Gynecol Scand. 2019;98:1549-1557.
- Archer DF, Ahrendt HJ, Drouin D. Drospirenone-only oral contraceptive: results from a multicenter noncomparative trial of efficacy, safety and tolerability. Contraception. 2015;92:439-444.
- Anderson PO, Sauberan JB. Modeling drug passage into human milk. Clin Pharmacol Ther. 2016;100:42-52. doi: 10.1002/cpt.377.
- Belsey EM. Vaginal bleeding patterns among women using one natural and eight hormonal methods of contraception. Contraception. 1988;38:181-206.
- Broome M, Fotherby K. Clinical experience with the progestin-only pill. Contraception. 1990;42:489-495.
- Apter D, Colli E, Gemzell-Danielsson K, et al. Multicenter, open-label trial to assess the safety and tolerability of drospirenone 4.0 mg over 6 cycles in female adolescents with a 7-cycle extension phase. Contraception. 2020;101:412.
- Birth control benefits. Healthcare.gov website. https://www.healthcare.gov/coverage/birth-control-benefits/. Accessed January 13, 2022.
- American College of Obstetricians and Gynecologists. Access to contraception. Committee Opinion No. 615. Obstet Gynecol. 2015;125:250-256.
- Health care and reproductive rights. National Women’s Law Center website. https://nwlc.org/issue/health-care. Accessed January 13, 2022.
- How to find out if your health plan covers birth control at no cost to you. National Women’s Law Center website. https://nwlc.org/sites/default/files/072014-insuranceflowchart_vupdated.pdf. Accessed January 13, 2022.
- Toolkit: Getting the coverage you deserve. National Women’s Law Center website. https://nwlc.org/sites/default/files/pdfs/final_nwlclogo_preventive servicestoolkit_9-25-13.pdf. Accessed January 13, 2022.
Real-world data reinforce stem cell transplant for progressive systemic sclerosis
Current selection criteria for autologous hematopoietic stem cell transplant (AHSCT) in patients with rapidly progressing systemic sclerosis were validated in a study presented at the annual meeting of the Canadian Rheumatology Association.
The study, which associated AHSCT with improvement in overall survival and an acceptable risk of adverse events, “provides valuable real-world, long-term data pertaining to key clinical outcomes to support the use of AHSCT in patients with rapidly progressing systemic sclerosis,” reported Nancy Maltez, MD, a rheumatologist and clinical investigator who is on the faculty of the University of Ottawa.
The prospective study enrolled 85 patients in Canada and 41 patients in France with rapidly progressing systemic sclerosis. The patients in both countries were enrolled with the same eligibility criteria for AHSCT, but patients in France underwent AHSCT while the patients in Canada were treated with conventional therapies, such as cyclophosphamide.
On the primary outcome of overall survival, the Kaplan-Meier curve split almost immediately in favor of AHSCT. At 4 years, more than 25% of patients in the conventional therapy group had died versus less than 5% of those who underwent AHSCT. Although the mortality curve did slope downwards in the AHSCT group over the subsequent 6 years of follow-up, it largely paralleled and remained superior to convention therapy.
About 50% survival advantage seen for AHSCT
In this nonrandomized study, the statistical survival advantage of AHSCT was not provided, but the survival graph showed about 75% survival at 8 years of follow-up in the AHSCT group, compared with about 50% survival in the conventional-therapy group.
Many of the secondary outcomes, including those evaluating skin involvement, preservation of lung function, and absence of renal complications also favored AHSCT, according to Dr. Maltez.
On the modified Rodnan skin score, a significant difference (P < .001) observed at 12 months was sustained at 36 months, when the score was 4.48 points lower among patients treated with AHSCT. The difference in forced vital capacity (FVC) was about 10% higher (P < .0001) in the AHSCT group.
Over long-term follow-up, the incidence of scleroderma renal crisis per 100 person-years was 6.02 cases in the conventional therapy group versus 0.58 cases (P < .001) in the AHSCT group. There was no significant difference in the proportion of patients in the two groups receiving a pacemaker over the course of follow-up, but the rate of new malignancies per 100 person-years was 3.71 in the conventional care group versus 0.58 (P < .001) in the AHSCT group.
Significant complications attributed to AHSCT were uncommon. This is important, because AHSCT was not uniformly well tolerated in the initial trials. The first of three randomized trials with AHSCT in progressive systemic sclerosis was published more than 10 years ago after a series of promising early phase trials. Each associated AHSCT with benefit, but patient selection appeared to be important.
In the ASSIST trial of 2011, AHSCT was associated with significant reductions in skin involvement and improvements in pulmonary function relative to cyclophosphamide, but enrollment was stopped after only 19 patients, and follow-up extended to only 2 years.
Substantial AHSCT-related mortality in ASTIS
In the second trial, called ASTIS, AHSCT was associated with a higher rate of mortality than cyclophosphamide after 1 year of follow-up, although there was a significantly greater long-term event-free survival for AHSCT when patients were followed out to 4 years. This study reinforced the need for cardiac screening because of because of concern that severe cardiac compromise contributed to the increased risk of AHSCT-related mortality.
The SCOT trial employed a high-intensity myeloablative conditioning regimen and total body irradiation prior to AHSCT. It is not clear that these contributed to improved survival, particularly because of the risk for irradiation to exacerbate complications in the lung and kidney, but AHSCT-related mortality was only 3% at 54 months. Patient enrollment criteria in this trial were also suspected of having played a role in the favorable results.
In the Canadian-French collaborative study, patients were considered eligible for AHSCT if they met the enrollment criteria used in the ASTIS trial, according to Dr. Maltez. She attributed the low rates of early mortality and relative absence of transplant-related death to the lessons learned in the published trials.
Overall, the data support the routine but selective use of AHSCT in rapidly progressing systemic sclerosis, Dr. Maltez concluded.
Maria Carolina Oliveira, MD, of the department of internal medicine at the University of São Paulo, generally agreed. A coauthor of a recent review of AHSCT for systemic sclerosis, Dr. Oliveira emphasized that patient selection is critical.
“AHSCT for systemic sclerosis has very specific inclusion criteria. Indeed, it is indicated for patients with severe and progressive disease but under two specific conditions: severe and progressive diffuse skin involvement and/or progressive interstitial lung disease,” she said in an interview.
Because of the thin line between benefit and risk according to disease subtypes and comorbidities, she said that it is important to be aware of relative contraindications and to recognize the risks of AHSCT.
At this time, and in the absence of better biomarkers to identify those most likely to benefit, “patients with other forms of severe scleroderma, such as those with pulmonary hypertension, scleroderma renal crisis, or severe cardiac involvement, for example, are not eligible,” she said.
Dr. Maltez and Dr. Oliveira reported having no potential conflicts of interest.
Current selection criteria for autologous hematopoietic stem cell transplant (AHSCT) in patients with rapidly progressing systemic sclerosis were validated in a study presented at the annual meeting of the Canadian Rheumatology Association.
The study, which associated AHSCT with improvement in overall survival and an acceptable risk of adverse events, “provides valuable real-world, long-term data pertaining to key clinical outcomes to support the use of AHSCT in patients with rapidly progressing systemic sclerosis,” reported Nancy Maltez, MD, a rheumatologist and clinical investigator who is on the faculty of the University of Ottawa.
The prospective study enrolled 85 patients in Canada and 41 patients in France with rapidly progressing systemic sclerosis. The patients in both countries were enrolled with the same eligibility criteria for AHSCT, but patients in France underwent AHSCT while the patients in Canada were treated with conventional therapies, such as cyclophosphamide.
On the primary outcome of overall survival, the Kaplan-Meier curve split almost immediately in favor of AHSCT. At 4 years, more than 25% of patients in the conventional therapy group had died versus less than 5% of those who underwent AHSCT. Although the mortality curve did slope downwards in the AHSCT group over the subsequent 6 years of follow-up, it largely paralleled and remained superior to convention therapy.
About 50% survival advantage seen for AHSCT
In this nonrandomized study, the statistical survival advantage of AHSCT was not provided, but the survival graph showed about 75% survival at 8 years of follow-up in the AHSCT group, compared with about 50% survival in the conventional-therapy group.
Many of the secondary outcomes, including those evaluating skin involvement, preservation of lung function, and absence of renal complications also favored AHSCT, according to Dr. Maltez.
On the modified Rodnan skin score, a significant difference (P < .001) observed at 12 months was sustained at 36 months, when the score was 4.48 points lower among patients treated with AHSCT. The difference in forced vital capacity (FVC) was about 10% higher (P < .0001) in the AHSCT group.
Over long-term follow-up, the incidence of scleroderma renal crisis per 100 person-years was 6.02 cases in the conventional therapy group versus 0.58 cases (P < .001) in the AHSCT group. There was no significant difference in the proportion of patients in the two groups receiving a pacemaker over the course of follow-up, but the rate of new malignancies per 100 person-years was 3.71 in the conventional care group versus 0.58 (P < .001) in the AHSCT group.
Significant complications attributed to AHSCT were uncommon. This is important, because AHSCT was not uniformly well tolerated in the initial trials. The first of three randomized trials with AHSCT in progressive systemic sclerosis was published more than 10 years ago after a series of promising early phase trials. Each associated AHSCT with benefit, but patient selection appeared to be important.
In the ASSIST trial of 2011, AHSCT was associated with significant reductions in skin involvement and improvements in pulmonary function relative to cyclophosphamide, but enrollment was stopped after only 19 patients, and follow-up extended to only 2 years.
Substantial AHSCT-related mortality in ASTIS
In the second trial, called ASTIS, AHSCT was associated with a higher rate of mortality than cyclophosphamide after 1 year of follow-up, although there was a significantly greater long-term event-free survival for AHSCT when patients were followed out to 4 years. This study reinforced the need for cardiac screening because of because of concern that severe cardiac compromise contributed to the increased risk of AHSCT-related mortality.
The SCOT trial employed a high-intensity myeloablative conditioning regimen and total body irradiation prior to AHSCT. It is not clear that these contributed to improved survival, particularly because of the risk for irradiation to exacerbate complications in the lung and kidney, but AHSCT-related mortality was only 3% at 54 months. Patient enrollment criteria in this trial were also suspected of having played a role in the favorable results.
In the Canadian-French collaborative study, patients were considered eligible for AHSCT if they met the enrollment criteria used in the ASTIS trial, according to Dr. Maltez. She attributed the low rates of early mortality and relative absence of transplant-related death to the lessons learned in the published trials.
Overall, the data support the routine but selective use of AHSCT in rapidly progressing systemic sclerosis, Dr. Maltez concluded.
Maria Carolina Oliveira, MD, of the department of internal medicine at the University of São Paulo, generally agreed. A coauthor of a recent review of AHSCT for systemic sclerosis, Dr. Oliveira emphasized that patient selection is critical.
“AHSCT for systemic sclerosis has very specific inclusion criteria. Indeed, it is indicated for patients with severe and progressive disease but under two specific conditions: severe and progressive diffuse skin involvement and/or progressive interstitial lung disease,” she said in an interview.
Because of the thin line between benefit and risk according to disease subtypes and comorbidities, she said that it is important to be aware of relative contraindications and to recognize the risks of AHSCT.
At this time, and in the absence of better biomarkers to identify those most likely to benefit, “patients with other forms of severe scleroderma, such as those with pulmonary hypertension, scleroderma renal crisis, or severe cardiac involvement, for example, are not eligible,” she said.
Dr. Maltez and Dr. Oliveira reported having no potential conflicts of interest.
Current selection criteria for autologous hematopoietic stem cell transplant (AHSCT) in patients with rapidly progressing systemic sclerosis were validated in a study presented at the annual meeting of the Canadian Rheumatology Association.
The study, which associated AHSCT with improvement in overall survival and an acceptable risk of adverse events, “provides valuable real-world, long-term data pertaining to key clinical outcomes to support the use of AHSCT in patients with rapidly progressing systemic sclerosis,” reported Nancy Maltez, MD, a rheumatologist and clinical investigator who is on the faculty of the University of Ottawa.
The prospective study enrolled 85 patients in Canada and 41 patients in France with rapidly progressing systemic sclerosis. The patients in both countries were enrolled with the same eligibility criteria for AHSCT, but patients in France underwent AHSCT while the patients in Canada were treated with conventional therapies, such as cyclophosphamide.
On the primary outcome of overall survival, the Kaplan-Meier curve split almost immediately in favor of AHSCT. At 4 years, more than 25% of patients in the conventional therapy group had died versus less than 5% of those who underwent AHSCT. Although the mortality curve did slope downwards in the AHSCT group over the subsequent 6 years of follow-up, it largely paralleled and remained superior to convention therapy.
About 50% survival advantage seen for AHSCT
In this nonrandomized study, the statistical survival advantage of AHSCT was not provided, but the survival graph showed about 75% survival at 8 years of follow-up in the AHSCT group, compared with about 50% survival in the conventional-therapy group.
Many of the secondary outcomes, including those evaluating skin involvement, preservation of lung function, and absence of renal complications also favored AHSCT, according to Dr. Maltez.
On the modified Rodnan skin score, a significant difference (P < .001) observed at 12 months was sustained at 36 months, when the score was 4.48 points lower among patients treated with AHSCT. The difference in forced vital capacity (FVC) was about 10% higher (P < .0001) in the AHSCT group.
Over long-term follow-up, the incidence of scleroderma renal crisis per 100 person-years was 6.02 cases in the conventional therapy group versus 0.58 cases (P < .001) in the AHSCT group. There was no significant difference in the proportion of patients in the two groups receiving a pacemaker over the course of follow-up, but the rate of new malignancies per 100 person-years was 3.71 in the conventional care group versus 0.58 (P < .001) in the AHSCT group.
Significant complications attributed to AHSCT were uncommon. This is important, because AHSCT was not uniformly well tolerated in the initial trials. The first of three randomized trials with AHSCT in progressive systemic sclerosis was published more than 10 years ago after a series of promising early phase trials. Each associated AHSCT with benefit, but patient selection appeared to be important.
In the ASSIST trial of 2011, AHSCT was associated with significant reductions in skin involvement and improvements in pulmonary function relative to cyclophosphamide, but enrollment was stopped after only 19 patients, and follow-up extended to only 2 years.
Substantial AHSCT-related mortality in ASTIS
In the second trial, called ASTIS, AHSCT was associated with a higher rate of mortality than cyclophosphamide after 1 year of follow-up, although there was a significantly greater long-term event-free survival for AHSCT when patients were followed out to 4 years. This study reinforced the need for cardiac screening because of because of concern that severe cardiac compromise contributed to the increased risk of AHSCT-related mortality.
The SCOT trial employed a high-intensity myeloablative conditioning regimen and total body irradiation prior to AHSCT. It is not clear that these contributed to improved survival, particularly because of the risk for irradiation to exacerbate complications in the lung and kidney, but AHSCT-related mortality was only 3% at 54 months. Patient enrollment criteria in this trial were also suspected of having played a role in the favorable results.
In the Canadian-French collaborative study, patients were considered eligible for AHSCT if they met the enrollment criteria used in the ASTIS trial, according to Dr. Maltez. She attributed the low rates of early mortality and relative absence of transplant-related death to the lessons learned in the published trials.
Overall, the data support the routine but selective use of AHSCT in rapidly progressing systemic sclerosis, Dr. Maltez concluded.
Maria Carolina Oliveira, MD, of the department of internal medicine at the University of São Paulo, generally agreed. A coauthor of a recent review of AHSCT for systemic sclerosis, Dr. Oliveira emphasized that patient selection is critical.
“AHSCT for systemic sclerosis has very specific inclusion criteria. Indeed, it is indicated for patients with severe and progressive disease but under two specific conditions: severe and progressive diffuse skin involvement and/or progressive interstitial lung disease,” she said in an interview.
Because of the thin line between benefit and risk according to disease subtypes and comorbidities, she said that it is important to be aware of relative contraindications and to recognize the risks of AHSCT.
At this time, and in the absence of better biomarkers to identify those most likely to benefit, “patients with other forms of severe scleroderma, such as those with pulmonary hypertension, scleroderma renal crisis, or severe cardiac involvement, for example, are not eligible,” she said.
Dr. Maltez and Dr. Oliveira reported having no potential conflicts of interest.
FROM THE ANNUAL MEETING OF THE CANADIAN RHEUMATOLOGY ASSOCIATION
How to advance equity, diversity, and inclusion in dermatology: Recommendations from an expert panel
– so he launched community outreach efforts with local businesses to attract patients from diverse backgrounds.
“For instance, I worked with U.S. Bank to give lectures on minorities in medicine and talked about outreach options and possible ways to include more ethnicities in medicine overall,” Dr. Qutub said during a panel discussion on equity, diversity, and inclusion (EDI) that took place at the ODAC Dermatology, Aesthetic & Surgical Conference. “I also did outreach with medical clinics in the area. Once patients are referred to you, they start to talk to people in their communities about you, and before you know it, you get people from their church and family members in your clinic.”
Dr. Qutub, who is ODAC’s director of Equity, Diversity, and Inclusion, kept EDI in mind when hiring staff for his practice, “to include candidates with varying experiences and backgrounds,” he said. “The idea was to make sure that when patients came into the clinic, they saw a varied group of individuals that were working together to help improve their health care outcomes. I found that made patients more comfortable in the clinic. It’s also important to have that representation daily in a larger setting like residency programs or multispecialty groups.”
Educational resources
Another panelist, Adam Friedman, MD, emphasized inclusivity of educational resources to ensure a dermatology workforce that can take care of all patients. “How can we expect the dermatology community to be able to treat anyone who comes through the door of their clinic if we don’t provide the resources that highlight and showcase the nuances and the diversity that skin disease has to offer?” asked Dr. Friedman, professor and chair of dermatology at George Washington University, Washington. “It comes down to educational tools and being purposeful when you’re putting together a talk or writing a paper, to be inclusive and have that on the top of your mind. It’s about saying right here, right now, we have to purposefully make a decision to be inclusive, to be welcoming to all so that we can practice at the highest level of our calling to treat everyone effectively and equitably.”
A unique feature of the atlas “is that we have taken multiple skin conditions, even common features such as erythema, and placed different skin tones side by side at the same angle to appreciate the full spectrum, and highlight those nuances,” Dr. Friedman said. “When you’re in clinic, when you see even common things like acne or seborrheic dermatitis,” he recommended taking photos to create a repository, “because you never know when that will be helpful when you want to show a medical student or a patient what something can look like on someone with a similar skin tone, or even to share with them how diverse skin conditions can appear across populations.”
Clinical research
Another way to help close racial gaps in dermatology is to improve access to mentorships and clinical research, according to panelist Chesahna Kindred, MD, of Kindred Hair & Skin Center in Columbia, Md. “We should be thoroughly embarrassed by the lack of diversity in our clinical trials,” she said.
In her role as chair of the dermatology section of the National Medical Association (NMA Derm), Dr. Kindred helped launch the NMA Derm research committee, which trains members to run clinical trials in their practices – an undertaking that was largely prompted by claims from pharmaceutical industry representatives that they struggle to find Black participants for clinical trials. “The truth of the matter is, if a Black patient doesn’t choose to go to Dr. Smith as a patient, they’re certainly not going to choose to go to Dr. Smith as a research participant,” Dr. Kindred said. “We have to meet those diverse populations where they are. By and large for Black patients, those are Black dermatologists.
In addition to meeting with primary investigators, she has been meeting with industry representatives, who she said are very interested in improving clinical trial diversity. “When a trial does not include a diverse population, we can call it out and say it is subpar,” she said.
In 2020, the Food and Drug Administration announced the availability of a guidance document, “Enhancing the Diversity of Clinical Trial Populations – Eligibility Criteria, Enrollment Practices, and Trial Designs,” which includes recommendations for sponsors on how to increase enrollment of underrepresented populations in their clinical trials of medical products.
Dr. Kindred has created a clinical research unit in her own practice, in partnership with Howard University’s department of dermatology and NMA Dermatology.
Studies Dr. Kindred is involved with include those looking at the relationship between hair care products targeted to Black women and the development of central centrifugal cicatricial alopecia (CCCA). CCCA is getting worse with each generation, “and we think the cause might be environmental,” she said. “Studies show that there are almost zero percent carcinogens in hair care products that target Whites. But close to 100% of hair care products that target Blacks contain carcinogens and endocrine disrupting chemicals, the most common being phthalates, which are found in relaxers, chemicals that patients use to straighten their hair.”
Urinary phthalate concentrations have been found to be much higher in Black women than in White women, and one of the pilot studies she is involved with is checking the urinary phthalate levels in Black women with and without CCCA, to see if there is a correlation.
Mentorships
DiAnne S. Davis, MD, of North Dallas Dermatology Associates, rounded out the panel discussion by underscoring the importance of mentorships for underrepresented minority medical students, which includes providing guidance through the application process. “Mentorship is key to closing some of these gaps, particularly in our field of dermatology,” Dr. Davis said.
Through NMA Derm, Dr. Davis was tasked by one of her mentors, Dr. Kindred, to spearhead a mentorship program that pairs medical students with a mentor in the dermatology field, “so we can help guide them not only on their medical school process but help in coordinating research projects, and make them successful in matching to dermatology,” she said. “When students reach out to you, it’s important to take them under your wing or connect them to somebody you know so that we can increase the number of minority dermatologists.”
None of the panelists reported having disclosures relevant to their presentations.
– so he launched community outreach efforts with local businesses to attract patients from diverse backgrounds.
“For instance, I worked with U.S. Bank to give lectures on minorities in medicine and talked about outreach options and possible ways to include more ethnicities in medicine overall,” Dr. Qutub said during a panel discussion on equity, diversity, and inclusion (EDI) that took place at the ODAC Dermatology, Aesthetic & Surgical Conference. “I also did outreach with medical clinics in the area. Once patients are referred to you, they start to talk to people in their communities about you, and before you know it, you get people from their church and family members in your clinic.”
Dr. Qutub, who is ODAC’s director of Equity, Diversity, and Inclusion, kept EDI in mind when hiring staff for his practice, “to include candidates with varying experiences and backgrounds,” he said. “The idea was to make sure that when patients came into the clinic, they saw a varied group of individuals that were working together to help improve their health care outcomes. I found that made patients more comfortable in the clinic. It’s also important to have that representation daily in a larger setting like residency programs or multispecialty groups.”
Educational resources
Another panelist, Adam Friedman, MD, emphasized inclusivity of educational resources to ensure a dermatology workforce that can take care of all patients. “How can we expect the dermatology community to be able to treat anyone who comes through the door of their clinic if we don’t provide the resources that highlight and showcase the nuances and the diversity that skin disease has to offer?” asked Dr. Friedman, professor and chair of dermatology at George Washington University, Washington. “It comes down to educational tools and being purposeful when you’re putting together a talk or writing a paper, to be inclusive and have that on the top of your mind. It’s about saying right here, right now, we have to purposefully make a decision to be inclusive, to be welcoming to all so that we can practice at the highest level of our calling to treat everyone effectively and equitably.”
A unique feature of the atlas “is that we have taken multiple skin conditions, even common features such as erythema, and placed different skin tones side by side at the same angle to appreciate the full spectrum, and highlight those nuances,” Dr. Friedman said. “When you’re in clinic, when you see even common things like acne or seborrheic dermatitis,” he recommended taking photos to create a repository, “because you never know when that will be helpful when you want to show a medical student or a patient what something can look like on someone with a similar skin tone, or even to share with them how diverse skin conditions can appear across populations.”
Clinical research
Another way to help close racial gaps in dermatology is to improve access to mentorships and clinical research, according to panelist Chesahna Kindred, MD, of Kindred Hair & Skin Center in Columbia, Md. “We should be thoroughly embarrassed by the lack of diversity in our clinical trials,” she said.
In her role as chair of the dermatology section of the National Medical Association (NMA Derm), Dr. Kindred helped launch the NMA Derm research committee, which trains members to run clinical trials in their practices – an undertaking that was largely prompted by claims from pharmaceutical industry representatives that they struggle to find Black participants for clinical trials. “The truth of the matter is, if a Black patient doesn’t choose to go to Dr. Smith as a patient, they’re certainly not going to choose to go to Dr. Smith as a research participant,” Dr. Kindred said. “We have to meet those diverse populations where they are. By and large for Black patients, those are Black dermatologists.
In addition to meeting with primary investigators, she has been meeting with industry representatives, who she said are very interested in improving clinical trial diversity. “When a trial does not include a diverse population, we can call it out and say it is subpar,” she said.
In 2020, the Food and Drug Administration announced the availability of a guidance document, “Enhancing the Diversity of Clinical Trial Populations – Eligibility Criteria, Enrollment Practices, and Trial Designs,” which includes recommendations for sponsors on how to increase enrollment of underrepresented populations in their clinical trials of medical products.
Dr. Kindred has created a clinical research unit in her own practice, in partnership with Howard University’s department of dermatology and NMA Dermatology.
Studies Dr. Kindred is involved with include those looking at the relationship between hair care products targeted to Black women and the development of central centrifugal cicatricial alopecia (CCCA). CCCA is getting worse with each generation, “and we think the cause might be environmental,” she said. “Studies show that there are almost zero percent carcinogens in hair care products that target Whites. But close to 100% of hair care products that target Blacks contain carcinogens and endocrine disrupting chemicals, the most common being phthalates, which are found in relaxers, chemicals that patients use to straighten their hair.”
Urinary phthalate concentrations have been found to be much higher in Black women than in White women, and one of the pilot studies she is involved with is checking the urinary phthalate levels in Black women with and without CCCA, to see if there is a correlation.
Mentorships
DiAnne S. Davis, MD, of North Dallas Dermatology Associates, rounded out the panel discussion by underscoring the importance of mentorships for underrepresented minority medical students, which includes providing guidance through the application process. “Mentorship is key to closing some of these gaps, particularly in our field of dermatology,” Dr. Davis said.
Through NMA Derm, Dr. Davis was tasked by one of her mentors, Dr. Kindred, to spearhead a mentorship program that pairs medical students with a mentor in the dermatology field, “so we can help guide them not only on their medical school process but help in coordinating research projects, and make them successful in matching to dermatology,” she said. “When students reach out to you, it’s important to take them under your wing or connect them to somebody you know so that we can increase the number of minority dermatologists.”
None of the panelists reported having disclosures relevant to their presentations.
– so he launched community outreach efforts with local businesses to attract patients from diverse backgrounds.
“For instance, I worked with U.S. Bank to give lectures on minorities in medicine and talked about outreach options and possible ways to include more ethnicities in medicine overall,” Dr. Qutub said during a panel discussion on equity, diversity, and inclusion (EDI) that took place at the ODAC Dermatology, Aesthetic & Surgical Conference. “I also did outreach with medical clinics in the area. Once patients are referred to you, they start to talk to people in their communities about you, and before you know it, you get people from their church and family members in your clinic.”
Dr. Qutub, who is ODAC’s director of Equity, Diversity, and Inclusion, kept EDI in mind when hiring staff for his practice, “to include candidates with varying experiences and backgrounds,” he said. “The idea was to make sure that when patients came into the clinic, they saw a varied group of individuals that were working together to help improve their health care outcomes. I found that made patients more comfortable in the clinic. It’s also important to have that representation daily in a larger setting like residency programs or multispecialty groups.”
Educational resources
Another panelist, Adam Friedman, MD, emphasized inclusivity of educational resources to ensure a dermatology workforce that can take care of all patients. “How can we expect the dermatology community to be able to treat anyone who comes through the door of their clinic if we don’t provide the resources that highlight and showcase the nuances and the diversity that skin disease has to offer?” asked Dr. Friedman, professor and chair of dermatology at George Washington University, Washington. “It comes down to educational tools and being purposeful when you’re putting together a talk or writing a paper, to be inclusive and have that on the top of your mind. It’s about saying right here, right now, we have to purposefully make a decision to be inclusive, to be welcoming to all so that we can practice at the highest level of our calling to treat everyone effectively and equitably.”
A unique feature of the atlas “is that we have taken multiple skin conditions, even common features such as erythema, and placed different skin tones side by side at the same angle to appreciate the full spectrum, and highlight those nuances,” Dr. Friedman said. “When you’re in clinic, when you see even common things like acne or seborrheic dermatitis,” he recommended taking photos to create a repository, “because you never know when that will be helpful when you want to show a medical student or a patient what something can look like on someone with a similar skin tone, or even to share with them how diverse skin conditions can appear across populations.”
Clinical research
Another way to help close racial gaps in dermatology is to improve access to mentorships and clinical research, according to panelist Chesahna Kindred, MD, of Kindred Hair & Skin Center in Columbia, Md. “We should be thoroughly embarrassed by the lack of diversity in our clinical trials,” she said.
In her role as chair of the dermatology section of the National Medical Association (NMA Derm), Dr. Kindred helped launch the NMA Derm research committee, which trains members to run clinical trials in their practices – an undertaking that was largely prompted by claims from pharmaceutical industry representatives that they struggle to find Black participants for clinical trials. “The truth of the matter is, if a Black patient doesn’t choose to go to Dr. Smith as a patient, they’re certainly not going to choose to go to Dr. Smith as a research participant,” Dr. Kindred said. “We have to meet those diverse populations where they are. By and large for Black patients, those are Black dermatologists.
In addition to meeting with primary investigators, she has been meeting with industry representatives, who she said are very interested in improving clinical trial diversity. “When a trial does not include a diverse population, we can call it out and say it is subpar,” she said.
In 2020, the Food and Drug Administration announced the availability of a guidance document, “Enhancing the Diversity of Clinical Trial Populations – Eligibility Criteria, Enrollment Practices, and Trial Designs,” which includes recommendations for sponsors on how to increase enrollment of underrepresented populations in their clinical trials of medical products.
Dr. Kindred has created a clinical research unit in her own practice, in partnership with Howard University’s department of dermatology and NMA Dermatology.
Studies Dr. Kindred is involved with include those looking at the relationship between hair care products targeted to Black women and the development of central centrifugal cicatricial alopecia (CCCA). CCCA is getting worse with each generation, “and we think the cause might be environmental,” she said. “Studies show that there are almost zero percent carcinogens in hair care products that target Whites. But close to 100% of hair care products that target Blacks contain carcinogens and endocrine disrupting chemicals, the most common being phthalates, which are found in relaxers, chemicals that patients use to straighten their hair.”
Urinary phthalate concentrations have been found to be much higher in Black women than in White women, and one of the pilot studies she is involved with is checking the urinary phthalate levels in Black women with and without CCCA, to see if there is a correlation.
Mentorships
DiAnne S. Davis, MD, of North Dallas Dermatology Associates, rounded out the panel discussion by underscoring the importance of mentorships for underrepresented minority medical students, which includes providing guidance through the application process. “Mentorship is key to closing some of these gaps, particularly in our field of dermatology,” Dr. Davis said.
Through NMA Derm, Dr. Davis was tasked by one of her mentors, Dr. Kindred, to spearhead a mentorship program that pairs medical students with a mentor in the dermatology field, “so we can help guide them not only on their medical school process but help in coordinating research projects, and make them successful in matching to dermatology,” she said. “When students reach out to you, it’s important to take them under your wing or connect them to somebody you know so that we can increase the number of minority dermatologists.”
None of the panelists reported having disclosures relevant to their presentations.
FROM ODAC 2022