User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID bivalent booster better vs. recent Omicron subvariants: Pfizer
the company reported on Nov. 4, supporting calls by public health officials for eligible people to get this booster before a potential COVID-19 surge this winter.
The company’s ongoing phase 2/3 study of their Omicron BA.4 and BA.5 bivalent – which targets both the virus’ original strain and the two subvariants – shows that the vaccine offered the strongest protection in people older than 55 years.
One month after receiving a 30-mcg booster with the bivalent vaccine, those older than 55 had four times more neutralizing antibodies against these Omicron subvariants, compared with people who received the original monovalent vaccine as a booster in the study.
Researchers compared the geometric mean titer (GMT) levels of these antibodies in three groups before and 1 month after boosting. The 36 people older than 55 years in the released study findings had an GMT level of 896 with the bivalent booster, a level 13 times higher than before this immunization.
For the 38 adults ages 18-55 in the study, the GMT level increased to 606 at 1 month after the bivalent booster, an increase of almost 10-fold, compared with baseline. In a comparator group of 40 people receiving the original vaccine as a fourth dose, the GMT level was 236, or threefold higher than before their booster shot.
The newly released data is “very encouraging and consistent now with three studies all showing a substantial 3-4 fold increased level of neutralizing antibodies versus BA.5 as compared with the original booster,” said Eric Topol, MD, director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News.
Pfizer and BioNTech announced the updated findings in a Nov. 4 press release.
A booster dose of the BA.4/BA.5-adapted bivalent vaccine is authorized for emergency use by the Food and Drug Administration for ages 5 years and older. The safety and tolerability profile of the Pfizer/BioNTech bivalent booster remains favorable and similar to the original COVID-19 vaccine, the company reported.
Until recently, the BA.5 Omicron variant was the dominant strain in the United States, but is now getting elbowed out by the subvariants BQ.1.1, BQ.1, and BA.4.6, which together make up almost 45% of the circulating virus.
Some skepticism
“It is important to note that these data are press-release level, which does not allow a view of the data totality,” Hana El Sahly, MD, professor of molecular virology and microbiology, Baylor College of Medicine, Houston, said in an interview.
“For example, there may be significant differences between the groups, and the release mentions at least one difference that is of importance: the interval since the last vaccination which often affects the response to subsequent boosting,” she said.
Dr. El Sahly added that the findings are not surprising. “In the short term, a variant-specific vaccine produces a higher level of antibody against the variant in the vaccine than the vaccines based on the ancestral strains.”
More researcher results are warranted. “These data do not indicate that these differences between the two vaccines translate into a meaningful clinical benefit at a population level,” Dr. El Sahly said.
An uncertain winter ahead
“As we head into the holiday season, we hope these updated data will encourage people to seek out a COVID-19 bivalent booster as soon as they are eligible in order to maintain high levels of protection against the widely circulating Omicron BA.4 and BA.5 sublineages,” Albert Bourla, Pfizer chairman and CEO, stated in the release.
The updated data from the Pfizer/BioNTech study are “all the more reason to get a booster, with added protection also versus BQ.1.1, which will soon become dominant in the U.S.,” Dr. Topol predicted.
It is unclear when the next surge will happen, as COVID-19 does not always follow a seasonal pattern, at least not yet, Dr. El Sahly said. “Regardless, it is reasonable to recommend additional vaccine doses to immunocompromised and frail or older persons. More importantly, influenza vaccination and being up to date on pneumococcal vaccines are highly recommended as soon as feasible, given the early and intense flu season.”
A version of this article first appeared on Medscape.com.
the company reported on Nov. 4, supporting calls by public health officials for eligible people to get this booster before a potential COVID-19 surge this winter.
The company’s ongoing phase 2/3 study of their Omicron BA.4 and BA.5 bivalent – which targets both the virus’ original strain and the two subvariants – shows that the vaccine offered the strongest protection in people older than 55 years.
One month after receiving a 30-mcg booster with the bivalent vaccine, those older than 55 had four times more neutralizing antibodies against these Omicron subvariants, compared with people who received the original monovalent vaccine as a booster in the study.
Researchers compared the geometric mean titer (GMT) levels of these antibodies in three groups before and 1 month after boosting. The 36 people older than 55 years in the released study findings had an GMT level of 896 with the bivalent booster, a level 13 times higher than before this immunization.
For the 38 adults ages 18-55 in the study, the GMT level increased to 606 at 1 month after the bivalent booster, an increase of almost 10-fold, compared with baseline. In a comparator group of 40 people receiving the original vaccine as a fourth dose, the GMT level was 236, or threefold higher than before their booster shot.
The newly released data is “very encouraging and consistent now with three studies all showing a substantial 3-4 fold increased level of neutralizing antibodies versus BA.5 as compared with the original booster,” said Eric Topol, MD, director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News.
Pfizer and BioNTech announced the updated findings in a Nov. 4 press release.
A booster dose of the BA.4/BA.5-adapted bivalent vaccine is authorized for emergency use by the Food and Drug Administration for ages 5 years and older. The safety and tolerability profile of the Pfizer/BioNTech bivalent booster remains favorable and similar to the original COVID-19 vaccine, the company reported.
Until recently, the BA.5 Omicron variant was the dominant strain in the United States, but is now getting elbowed out by the subvariants BQ.1.1, BQ.1, and BA.4.6, which together make up almost 45% of the circulating virus.
Some skepticism
“It is important to note that these data are press-release level, which does not allow a view of the data totality,” Hana El Sahly, MD, professor of molecular virology and microbiology, Baylor College of Medicine, Houston, said in an interview.
“For example, there may be significant differences between the groups, and the release mentions at least one difference that is of importance: the interval since the last vaccination which often affects the response to subsequent boosting,” she said.
Dr. El Sahly added that the findings are not surprising. “In the short term, a variant-specific vaccine produces a higher level of antibody against the variant in the vaccine than the vaccines based on the ancestral strains.”
More researcher results are warranted. “These data do not indicate that these differences between the two vaccines translate into a meaningful clinical benefit at a population level,” Dr. El Sahly said.
An uncertain winter ahead
“As we head into the holiday season, we hope these updated data will encourage people to seek out a COVID-19 bivalent booster as soon as they are eligible in order to maintain high levels of protection against the widely circulating Omicron BA.4 and BA.5 sublineages,” Albert Bourla, Pfizer chairman and CEO, stated in the release.
The updated data from the Pfizer/BioNTech study are “all the more reason to get a booster, with added protection also versus BQ.1.1, which will soon become dominant in the U.S.,” Dr. Topol predicted.
It is unclear when the next surge will happen, as COVID-19 does not always follow a seasonal pattern, at least not yet, Dr. El Sahly said. “Regardless, it is reasonable to recommend additional vaccine doses to immunocompromised and frail or older persons. More importantly, influenza vaccination and being up to date on pneumococcal vaccines are highly recommended as soon as feasible, given the early and intense flu season.”
A version of this article first appeared on Medscape.com.
the company reported on Nov. 4, supporting calls by public health officials for eligible people to get this booster before a potential COVID-19 surge this winter.
The company’s ongoing phase 2/3 study of their Omicron BA.4 and BA.5 bivalent – which targets both the virus’ original strain and the two subvariants – shows that the vaccine offered the strongest protection in people older than 55 years.
One month after receiving a 30-mcg booster with the bivalent vaccine, those older than 55 had four times more neutralizing antibodies against these Omicron subvariants, compared with people who received the original monovalent vaccine as a booster in the study.
Researchers compared the geometric mean titer (GMT) levels of these antibodies in three groups before and 1 month after boosting. The 36 people older than 55 years in the released study findings had an GMT level of 896 with the bivalent booster, a level 13 times higher than before this immunization.
For the 38 adults ages 18-55 in the study, the GMT level increased to 606 at 1 month after the bivalent booster, an increase of almost 10-fold, compared with baseline. In a comparator group of 40 people receiving the original vaccine as a fourth dose, the GMT level was 236, or threefold higher than before their booster shot.
The newly released data is “very encouraging and consistent now with three studies all showing a substantial 3-4 fold increased level of neutralizing antibodies versus BA.5 as compared with the original booster,” said Eric Topol, MD, director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News.
Pfizer and BioNTech announced the updated findings in a Nov. 4 press release.
A booster dose of the BA.4/BA.5-adapted bivalent vaccine is authorized for emergency use by the Food and Drug Administration for ages 5 years and older. The safety and tolerability profile of the Pfizer/BioNTech bivalent booster remains favorable and similar to the original COVID-19 vaccine, the company reported.
Until recently, the BA.5 Omicron variant was the dominant strain in the United States, but is now getting elbowed out by the subvariants BQ.1.1, BQ.1, and BA.4.6, which together make up almost 45% of the circulating virus.
Some skepticism
“It is important to note that these data are press-release level, which does not allow a view of the data totality,” Hana El Sahly, MD, professor of molecular virology and microbiology, Baylor College of Medicine, Houston, said in an interview.
“For example, there may be significant differences between the groups, and the release mentions at least one difference that is of importance: the interval since the last vaccination which often affects the response to subsequent boosting,” she said.
Dr. El Sahly added that the findings are not surprising. “In the short term, a variant-specific vaccine produces a higher level of antibody against the variant in the vaccine than the vaccines based on the ancestral strains.”
More researcher results are warranted. “These data do not indicate that these differences between the two vaccines translate into a meaningful clinical benefit at a population level,” Dr. El Sahly said.
An uncertain winter ahead
“As we head into the holiday season, we hope these updated data will encourage people to seek out a COVID-19 bivalent booster as soon as they are eligible in order to maintain high levels of protection against the widely circulating Omicron BA.4 and BA.5 sublineages,” Albert Bourla, Pfizer chairman and CEO, stated in the release.
The updated data from the Pfizer/BioNTech study are “all the more reason to get a booster, with added protection also versus BQ.1.1, which will soon become dominant in the U.S.,” Dr. Topol predicted.
It is unclear when the next surge will happen, as COVID-19 does not always follow a seasonal pattern, at least not yet, Dr. El Sahly said. “Regardless, it is reasonable to recommend additional vaccine doses to immunocompromised and frail or older persons. More importantly, influenza vaccination and being up to date on pneumococcal vaccines are highly recommended as soon as feasible, given the early and intense flu season.”
A version of this article first appeared on Medscape.com.
Avoid routine early ECMO in severe cardiogenic shock: ECMO-CS
CHICAGO – Routine early, expeditious use of extracorporeal membrane oxygenation (ECMO) is a common strategy in patients with severe cardiogenic shock, but a less aggressive initial approach may be just as effective, a randomized trial suggests.
In the study that assigned patients with “rapidly deteriorating or severe” cardiogenic shock to one or the other approach, clinical outcomes were no better for those who received immediate ECMO than for those initially managed with inotropes and vasopressors, researchers said.
The conservative strategy, importantly, allowed for downstream ECMO in the event of hemodynamic deterioration, which occurred in a substantial 39% of cases, observed Petr Ostadal, MD, PhD, when presenting the results at the American Heart Association scientific sessions.
Dr. Ostadal of Na Homolce Hospital, Prague, is also first author on the published report of the study, called Extracorporeal Membrane Oxygenation in the Therapy of Cardiogenic Shock (ECMO-CS), which was published the same day in Circulation.
The trial makes a firm case for preferring the conservative initial approach over routine early ECMO in the kind of patients it entered, Larry A. Allen, MD, MHS, University of Coloradoat Denver, Aurora, told this news organization.
More than 60% of the trial’s 117 patients had shock secondary to an acute coronary syndrome; another 23% were in heart failure decompensation.
A preference for the conservative initial approach would be welcome, he said. The early aggressive ECMO approach is resource intensive and carries some important risks, such as stroke or coagulopathy, said Dr. Allen, who is not connected with ECMO-CS. Yet it is increasingly the go-to approach in such patients, based primarily on observational data.
Although early ECMO apparently didn’t benefit patients in this study in their specific stage of cardiogenic shock, Dr. Allen observed, it would presumably help some, but identifying them in practice presents challenges. “Defining where people are in the spectrum of early versus middle versus late cardiogenic shock is actually very tricky.”
It will therefore be important, he said, to identify ways to predict which conservatively managed patients do well with the strategy, and which are most at risk for hemodynamic deterioration and for whom ECMO should be readily available.
“I think part of what ECMO-CS tells us is that, if a patient is stable on intravenous inotropic and vasopressor support, you can defer ECMO while you’re thinking about the patient – about their larger context and the right medical decision-making for them.”
The trial randomly assigned 122 patients with rapidly deteriorating or severe cardiogenic shock to the immediate-ECMO or the conservative strategy at four centers in the Czech Republic. The 117 patients for whom informed consent could be obtained were included in the analysis, 58 and 59 patients, respectively. Their mean age was about 65 years and three-fourths were male.
The primary endpoint, the only endpoint for which the study was powered, consisted of death from any cause, resuscitated circulatory arrest, or use of a different form of mechanical circulatory support (MCS) by 30 days.
It occurred in 63.8% of patients assigned to immediate ECMO and 71.2% of those in the conservative strategy group, for a hazard ratio of 0.72 (95% confidence interval, 0.46-1.12; P = .21).
As individual endpoints, rates of death from any cause and resuscitated arrest did not significantly differ between the groups, but conservatively managed patients more often used another form of MCS. The HRs were 1.11 (95% CI, 0.66-1.87) for death from any cause, 0.79 (95% CI, 0.27-2.28) for resuscitated cardiac arrest, and 0.38 (95% CI, 0.18-0.79) for use of another MCS device.
The rates for serious adverse events – including bleeding, ischemia, stroke, pneumonia, or sepsis – were similar at 60.3% in the early-ECMO group and 61% in group with conservative initial management, Dr. Ostadal reported.
Other than the 23 patients in the conservative initial strategy group who went on to receive ECMO (1.9 days after randomization, on average), 1 went on to undergo implantation with a HeartMate (Abbott) ventricular assist device and 3 received an Impella pump (Abiomed).
Six patients in the early-ECMO group were already receiving intra-aortic balloon pump (IABP) support at randomization, two underwent temporary implantation with a Centrimag device (Abbott), and three went on to receive a HeartMate device, the published report notes.
ECMO is the optimal first choice for MCS in such patients with cardiogenic shock who need a circulatory support device, especially because it also oxygenates the blood, Dr. Ostadal told this news organization.
But ECMO doesn’t help with ventricular unloading. Indeed, it can sometimes reduce ventricular preload, especially if right-heart pressures are low. So MCS devices that unload the ventricle, typically an IABP, can complement ECMO.
Dr. Ostadal speculates, however, that there may be a better pairing option. “Impella plus ECMO, I think, is the combination which has a future,” he said, for patients in cardiogenic shock who need a short-term percutaneous hemodynamic support device. Impella “supports the whole circulation” and unloads the left ventricle.
“A balloon pump in combination with ECMO is still not a bad choice. It’s very cheap in comparison with Impella.” But in his opinion, Dr. Ostadal said, “The combination of Impella plus ECMO is more efficient from a hemodynamic point of view.”
As the published report notes, ongoing randomized trials looking at ECMO plus other MCS devices in cardiogenic shock include ECLS-SHOCK, with a projected enrollment of 420 patients, and EURO-SHOCK, aiming for a similar number of patients; both compare routine ECMO to conservative management.
In addition, ANCHOR, in which ECMO is combined with IABP, and DanShock, which looks at early use of Impella rather than ECMO, are enrolling patients with shock secondary to acute coronary syndromes.
Dr. Ostadal disclosed consulting for Getinge, Edwards, Medtronic, Biomedica, and Xenios/Fresenius, and receiving research support from Xenios/Fresenius. Dr. Allen disclosed modest or significant relationships with ACI Clinical, Novartis, UpToDate, Boston Scientific, and Cytokinetics.
A version of this article first appeared on Medscape.com.
CHICAGO – Routine early, expeditious use of extracorporeal membrane oxygenation (ECMO) is a common strategy in patients with severe cardiogenic shock, but a less aggressive initial approach may be just as effective, a randomized trial suggests.
In the study that assigned patients with “rapidly deteriorating or severe” cardiogenic shock to one or the other approach, clinical outcomes were no better for those who received immediate ECMO than for those initially managed with inotropes and vasopressors, researchers said.
The conservative strategy, importantly, allowed for downstream ECMO in the event of hemodynamic deterioration, which occurred in a substantial 39% of cases, observed Petr Ostadal, MD, PhD, when presenting the results at the American Heart Association scientific sessions.
Dr. Ostadal of Na Homolce Hospital, Prague, is also first author on the published report of the study, called Extracorporeal Membrane Oxygenation in the Therapy of Cardiogenic Shock (ECMO-CS), which was published the same day in Circulation.
The trial makes a firm case for preferring the conservative initial approach over routine early ECMO in the kind of patients it entered, Larry A. Allen, MD, MHS, University of Coloradoat Denver, Aurora, told this news organization.
More than 60% of the trial’s 117 patients had shock secondary to an acute coronary syndrome; another 23% were in heart failure decompensation.
A preference for the conservative initial approach would be welcome, he said. The early aggressive ECMO approach is resource intensive and carries some important risks, such as stroke or coagulopathy, said Dr. Allen, who is not connected with ECMO-CS. Yet it is increasingly the go-to approach in such patients, based primarily on observational data.
Although early ECMO apparently didn’t benefit patients in this study in their specific stage of cardiogenic shock, Dr. Allen observed, it would presumably help some, but identifying them in practice presents challenges. “Defining where people are in the spectrum of early versus middle versus late cardiogenic shock is actually very tricky.”
It will therefore be important, he said, to identify ways to predict which conservatively managed patients do well with the strategy, and which are most at risk for hemodynamic deterioration and for whom ECMO should be readily available.
“I think part of what ECMO-CS tells us is that, if a patient is stable on intravenous inotropic and vasopressor support, you can defer ECMO while you’re thinking about the patient – about their larger context and the right medical decision-making for them.”
The trial randomly assigned 122 patients with rapidly deteriorating or severe cardiogenic shock to the immediate-ECMO or the conservative strategy at four centers in the Czech Republic. The 117 patients for whom informed consent could be obtained were included in the analysis, 58 and 59 patients, respectively. Their mean age was about 65 years and three-fourths were male.
The primary endpoint, the only endpoint for which the study was powered, consisted of death from any cause, resuscitated circulatory arrest, or use of a different form of mechanical circulatory support (MCS) by 30 days.
It occurred in 63.8% of patients assigned to immediate ECMO and 71.2% of those in the conservative strategy group, for a hazard ratio of 0.72 (95% confidence interval, 0.46-1.12; P = .21).
As individual endpoints, rates of death from any cause and resuscitated arrest did not significantly differ between the groups, but conservatively managed patients more often used another form of MCS. The HRs were 1.11 (95% CI, 0.66-1.87) for death from any cause, 0.79 (95% CI, 0.27-2.28) for resuscitated cardiac arrest, and 0.38 (95% CI, 0.18-0.79) for use of another MCS device.
The rates for serious adverse events – including bleeding, ischemia, stroke, pneumonia, or sepsis – were similar at 60.3% in the early-ECMO group and 61% in group with conservative initial management, Dr. Ostadal reported.
Other than the 23 patients in the conservative initial strategy group who went on to receive ECMO (1.9 days after randomization, on average), 1 went on to undergo implantation with a HeartMate (Abbott) ventricular assist device and 3 received an Impella pump (Abiomed).
Six patients in the early-ECMO group were already receiving intra-aortic balloon pump (IABP) support at randomization, two underwent temporary implantation with a Centrimag device (Abbott), and three went on to receive a HeartMate device, the published report notes.
ECMO is the optimal first choice for MCS in such patients with cardiogenic shock who need a circulatory support device, especially because it also oxygenates the blood, Dr. Ostadal told this news organization.
But ECMO doesn’t help with ventricular unloading. Indeed, it can sometimes reduce ventricular preload, especially if right-heart pressures are low. So MCS devices that unload the ventricle, typically an IABP, can complement ECMO.
Dr. Ostadal speculates, however, that there may be a better pairing option. “Impella plus ECMO, I think, is the combination which has a future,” he said, for patients in cardiogenic shock who need a short-term percutaneous hemodynamic support device. Impella “supports the whole circulation” and unloads the left ventricle.
“A balloon pump in combination with ECMO is still not a bad choice. It’s very cheap in comparison with Impella.” But in his opinion, Dr. Ostadal said, “The combination of Impella plus ECMO is more efficient from a hemodynamic point of view.”
As the published report notes, ongoing randomized trials looking at ECMO plus other MCS devices in cardiogenic shock include ECLS-SHOCK, with a projected enrollment of 420 patients, and EURO-SHOCK, aiming for a similar number of patients; both compare routine ECMO to conservative management.
In addition, ANCHOR, in which ECMO is combined with IABP, and DanShock, which looks at early use of Impella rather than ECMO, are enrolling patients with shock secondary to acute coronary syndromes.
Dr. Ostadal disclosed consulting for Getinge, Edwards, Medtronic, Biomedica, and Xenios/Fresenius, and receiving research support from Xenios/Fresenius. Dr. Allen disclosed modest or significant relationships with ACI Clinical, Novartis, UpToDate, Boston Scientific, and Cytokinetics.
A version of this article first appeared on Medscape.com.
CHICAGO – Routine early, expeditious use of extracorporeal membrane oxygenation (ECMO) is a common strategy in patients with severe cardiogenic shock, but a less aggressive initial approach may be just as effective, a randomized trial suggests.
In the study that assigned patients with “rapidly deteriorating or severe” cardiogenic shock to one or the other approach, clinical outcomes were no better for those who received immediate ECMO than for those initially managed with inotropes and vasopressors, researchers said.
The conservative strategy, importantly, allowed for downstream ECMO in the event of hemodynamic deterioration, which occurred in a substantial 39% of cases, observed Petr Ostadal, MD, PhD, when presenting the results at the American Heart Association scientific sessions.
Dr. Ostadal of Na Homolce Hospital, Prague, is also first author on the published report of the study, called Extracorporeal Membrane Oxygenation in the Therapy of Cardiogenic Shock (ECMO-CS), which was published the same day in Circulation.
The trial makes a firm case for preferring the conservative initial approach over routine early ECMO in the kind of patients it entered, Larry A. Allen, MD, MHS, University of Coloradoat Denver, Aurora, told this news organization.
More than 60% of the trial’s 117 patients had shock secondary to an acute coronary syndrome; another 23% were in heart failure decompensation.
A preference for the conservative initial approach would be welcome, he said. The early aggressive ECMO approach is resource intensive and carries some important risks, such as stroke or coagulopathy, said Dr. Allen, who is not connected with ECMO-CS. Yet it is increasingly the go-to approach in such patients, based primarily on observational data.
Although early ECMO apparently didn’t benefit patients in this study in their specific stage of cardiogenic shock, Dr. Allen observed, it would presumably help some, but identifying them in practice presents challenges. “Defining where people are in the spectrum of early versus middle versus late cardiogenic shock is actually very tricky.”
It will therefore be important, he said, to identify ways to predict which conservatively managed patients do well with the strategy, and which are most at risk for hemodynamic deterioration and for whom ECMO should be readily available.
“I think part of what ECMO-CS tells us is that, if a patient is stable on intravenous inotropic and vasopressor support, you can defer ECMO while you’re thinking about the patient – about their larger context and the right medical decision-making for them.”
The trial randomly assigned 122 patients with rapidly deteriorating or severe cardiogenic shock to the immediate-ECMO or the conservative strategy at four centers in the Czech Republic. The 117 patients for whom informed consent could be obtained were included in the analysis, 58 and 59 patients, respectively. Their mean age was about 65 years and three-fourths were male.
The primary endpoint, the only endpoint for which the study was powered, consisted of death from any cause, resuscitated circulatory arrest, or use of a different form of mechanical circulatory support (MCS) by 30 days.
It occurred in 63.8% of patients assigned to immediate ECMO and 71.2% of those in the conservative strategy group, for a hazard ratio of 0.72 (95% confidence interval, 0.46-1.12; P = .21).
As individual endpoints, rates of death from any cause and resuscitated arrest did not significantly differ between the groups, but conservatively managed patients more often used another form of MCS. The HRs were 1.11 (95% CI, 0.66-1.87) for death from any cause, 0.79 (95% CI, 0.27-2.28) for resuscitated cardiac arrest, and 0.38 (95% CI, 0.18-0.79) for use of another MCS device.
The rates for serious adverse events – including bleeding, ischemia, stroke, pneumonia, or sepsis – were similar at 60.3% in the early-ECMO group and 61% in group with conservative initial management, Dr. Ostadal reported.
Other than the 23 patients in the conservative initial strategy group who went on to receive ECMO (1.9 days after randomization, on average), 1 went on to undergo implantation with a HeartMate (Abbott) ventricular assist device and 3 received an Impella pump (Abiomed).
Six patients in the early-ECMO group were already receiving intra-aortic balloon pump (IABP) support at randomization, two underwent temporary implantation with a Centrimag device (Abbott), and three went on to receive a HeartMate device, the published report notes.
ECMO is the optimal first choice for MCS in such patients with cardiogenic shock who need a circulatory support device, especially because it also oxygenates the blood, Dr. Ostadal told this news organization.
But ECMO doesn’t help with ventricular unloading. Indeed, it can sometimes reduce ventricular preload, especially if right-heart pressures are low. So MCS devices that unload the ventricle, typically an IABP, can complement ECMO.
Dr. Ostadal speculates, however, that there may be a better pairing option. “Impella plus ECMO, I think, is the combination which has a future,” he said, for patients in cardiogenic shock who need a short-term percutaneous hemodynamic support device. Impella “supports the whole circulation” and unloads the left ventricle.
“A balloon pump in combination with ECMO is still not a bad choice. It’s very cheap in comparison with Impella.” But in his opinion, Dr. Ostadal said, “The combination of Impella plus ECMO is more efficient from a hemodynamic point of view.”
As the published report notes, ongoing randomized trials looking at ECMO plus other MCS devices in cardiogenic shock include ECLS-SHOCK, with a projected enrollment of 420 patients, and EURO-SHOCK, aiming for a similar number of patients; both compare routine ECMO to conservative management.
In addition, ANCHOR, in which ECMO is combined with IABP, and DanShock, which looks at early use of Impella rather than ECMO, are enrolling patients with shock secondary to acute coronary syndromes.
Dr. Ostadal disclosed consulting for Getinge, Edwards, Medtronic, Biomedica, and Xenios/Fresenius, and receiving research support from Xenios/Fresenius. Dr. Allen disclosed modest or significant relationships with ACI Clinical, Novartis, UpToDate, Boston Scientific, and Cytokinetics.
A version of this article first appeared on Medscape.com.
AT AHA 2022
No survival advantage for either torsemide or furosemide in HF: TRANSFORM-HF
CHICAGO – The choice of loop diuretic for decongestion in patients hospitalized with heart failure (HF) may make little difference to survival or readmission risk over the next year, at least when deciding between furosemide or torsemide, a randomized trial suggests.
Both drugs are old and widely used, but differences between the two loop diuretics in bioavailability, effects on potassium levels, and other features have led some clinicians to sometimes prefer torsemide. Until now, however, no randomized HF trials have compared the two drugs.
The new findings suggest clinicians can continue starting such patients with HF on either agent, at their discretion, without concern that the choice may compromise outcomes, say researchers from the TRANSFORM-HF trial, which compared furosemide-first and torsemide-first diuretic strategies in a diverse population of patients with HF.
Given that the two strategies were similarly effective for survival and rehospitalization, clinicians caring for patients with HF can focus more on “getting patients on the right dose for their loop diuretic, and prioritizing those therapies proven to improve clinical outcomes,” said Robert J. Mentz, MD, of Duke University Clinical Research Institute, Durham, N.C.
Dr. Mentz, a TRANSFORM-HF principal investigator, presented the primary results November 5 at the American Heart Association scientific sessions.
The trial had randomly assigned 2,859 patients hospitalized with HF and with a plan for oral loop diuretic therapy to initiate treatment with furosemide or torsemide. Clinicians were encouraged to maintain patients on the assigned diuretic, but crossovers to the other drug or other diuretic changes were allowed.
Rates of death from any cause, the primary endpoint, were about 26% in both groups over a median 17-month follow-up, regardless of ejection fraction (EF).
The composite rates of all-cause death or hospitalization at 12 months were also not significantly different, about 49% for those started on furosemide and about 47% for patients initially prescribed torsemide.
As a pragmatic comparative effectiveness trial, TRANSFORM-HF entered diverse patients with HF, broadly representative of actual clinical practice, who were managed according to routine practice and a streamlined study protocol at more than 60 U.S. centers, Dr. Mentz observed.
One of the pragmatic design’s advantages, he told this news organization, was “how efficient it was” as a randomized comparison of treatment strategies for clinical outcomes. It was “relatively low cost” and recruited patients quickly, compared with conventional randomized trials, “and we answered the question clearly.” The trial’s results, Dr. Mentz said, reflect “what happens in the real world.”
When might torsemide have the edge?
Although furosemide is the most commonly used loop diuretic in HF, and there are others besides it and torsemide, the latter has both known and theoretical advantages that set it apart. Torsemide is more than twice as potent as furosemide and more bioavailable, and its treatment effect lasts longer, the TRANSFORM-HF investigators have noted.
In addition, preclinical and small clinical studies suggest torsemide may have pleiotropic effects that might be theoretical advantages for patients with HF. For example, it appears to downregulate the renin-angiotensin-aldosterone system (RAAS) and reduce myocardial fibrosis and promote reverse ventricular remodeling, the group writes.
In practice, therefore, torsemide may be preferred in patients with furosemide resistance or “challenges with bioavailability, especially those with very advanced heart failure with congestion who may have gut edema, where oral furosemide and other loop diuretics are not effectively absorbed,” Biykem Bozkurt, MD, PhD, Baylor College of Medicine, Houston, told this news organization.
In such patients, she said, torsemide “is considered to be a better choice for individuals who have diuretic resistance with advanced congestion.”
The drug’s apparent pleiotropic effects, such as RAAS inhibition, may have less relevance to the TRANSFORM-HF primary endpoint of all-cause mortality than to clinical outcomes more likely associated with successful decongestion, such as HF hospitalization, Dr. Bozkurt proposed.
The trial’s pragmatic design, however, made it more feasible to focus on all-cause mortality and less practical to use measures of successful decongestion, such as volume loss or reduction in natriuretic peptide levels, she observed. Those are endpoints of special interest when diuretics are compared, “especially for the subgroup of patients who are diuretic resistant.”
Over the last 20 years or so, “we’ve learned that hospitalized heart failure is a very different disease process with a different natural history,” observed Clyde W. Yancy, MD, MSc, Northwestern University, Chicago, who was not part of the current study.
“So, the idea that something as nuanced as choice of one loop diuretic over the other, in that setting, would be sufficient to change the natural history, may be still a high bar for us,” he said in an interview.
“Based on these data, one would have to argue that whichever loop diuretic you select for the hospitalized patient – and a lot of that is driven by market exigencies right now – it turns out that the response is indistinguishable,” Dr. Yancy said. “That means if your hospital happens to have furosemide on the formulary, use it. If furosemide is not available but torsemide is available, use it.”
Dr. Yancy said he’d like to see a trial similar to TRANSFORM-HF but in outpatients receiving today’s guideline-directed medical therapy, which includes the sodium-glucose cotransporter 2 (SGLT2) inhibitors, drugs that increase the fractional excretion of sodium and have a “diureticlike” effect.
Such a trial, he said, would explore “the combination of not one, or two, but three agents with a diuretic effect – a loop diuretic, a mineralocorticoid antagonist, and an SGLT2 inhibitor – in ambulatory, optimized patients. It might make a difference.”
HF regardless of EF
The trial enrolled patients hospitalized with worsening or new-onset HF with a plan for long-term loop diuretic therapy who had either an EF of 40% or lower or, regardless of EF, elevated natriuretic peptide levels when hospitalized.
Of the 2,859 participants, whose mean age was about 65 years, about 36% were women and 34% African American. Overall, 1,428 were assigned to receive furosemide as their initial oral diuretic and 1,431 patients were assigned to the torsemide-first strategy.
The rate of death from any cause in both groups was 17 per 100 patient-years at a median of 17.4 months. The hazard ratio for torsemide vs. furosemide was 1.02 (95% confidence interval, 0.89-1.18; P = .77).
The corresponding HR at 12 months for all-cause death or hospitalization was 0.92 (95% CI, 0.83-1.02; P = .11). The relative risk for any hospitalization was 0.94 (95% CI, 0.84-1.07).
Pragmatic design: Other implications
Dosing was left to clinician discretion in the open-label study, as was whether patients maintained their assigned drug or switched over to the other agent. Indeed, 5.4% of patients crossed over to the other loop diuretic, and 2.8% went off loop diuretics entirely between in-hospital randomization and discharge, Dr. Mentz reported. By day 30, 6.7% had crossed over, and 7% had stopped taking loop diuretics.
The diuretic crossovers and discontinuations, Dr. Mentz said, likely biased the trial’s outcomes, such that the two strategies performed about equally well. Efforts were made, however, to at least partially overcome that limitation.
“We put measures in place to support adherence – sending letters to their primary doctors, giving them a wallet card so they would know which therapy they were on, having conversations about the importance of trying to stay on the randomized therapy,” Dr. Mentz said in an interview. Still, some clinicians saw differences between the two agents that prompted them, at some point, to switch patients from one loop diuretic to the other.
But interestingly, Dr. Mentz reported, the two strategies did not significantly differ in all-cause mortality or the composite of all-cause mortality or hospitalization in analysis by intention to treat.
Dr. Mentz discloses receiving honoraria from AstraZeneca, Bayer/Merck, Boehringer Ingelheim/Lilly, Cytokinetics, Pharmacosmos, Respicardia, Windtree Therapeutics, and Zoll; and research grants from American Regent and Novartis. Dr. Bozkurt discloses receiving honoraria from AstraZeneca, Baxter Health Care, and Sanofi Aventis and having other relationships with Renovacor, Respicardia, Abbott Vascular, Liva Nova, Vifor, and Cardurion. Dr. Yancy discloses a modest relationship with Abbott.
A version of this article first appeared on Medscape.com.
CHICAGO – The choice of loop diuretic for decongestion in patients hospitalized with heart failure (HF) may make little difference to survival or readmission risk over the next year, at least when deciding between furosemide or torsemide, a randomized trial suggests.
Both drugs are old and widely used, but differences between the two loop diuretics in bioavailability, effects on potassium levels, and other features have led some clinicians to sometimes prefer torsemide. Until now, however, no randomized HF trials have compared the two drugs.
The new findings suggest clinicians can continue starting such patients with HF on either agent, at their discretion, without concern that the choice may compromise outcomes, say researchers from the TRANSFORM-HF trial, which compared furosemide-first and torsemide-first diuretic strategies in a diverse population of patients with HF.
Given that the two strategies were similarly effective for survival and rehospitalization, clinicians caring for patients with HF can focus more on “getting patients on the right dose for their loop diuretic, and prioritizing those therapies proven to improve clinical outcomes,” said Robert J. Mentz, MD, of Duke University Clinical Research Institute, Durham, N.C.
Dr. Mentz, a TRANSFORM-HF principal investigator, presented the primary results November 5 at the American Heart Association scientific sessions.
The trial had randomly assigned 2,859 patients hospitalized with HF and with a plan for oral loop diuretic therapy to initiate treatment with furosemide or torsemide. Clinicians were encouraged to maintain patients on the assigned diuretic, but crossovers to the other drug or other diuretic changes were allowed.
Rates of death from any cause, the primary endpoint, were about 26% in both groups over a median 17-month follow-up, regardless of ejection fraction (EF).
The composite rates of all-cause death or hospitalization at 12 months were also not significantly different, about 49% for those started on furosemide and about 47% for patients initially prescribed torsemide.
As a pragmatic comparative effectiveness trial, TRANSFORM-HF entered diverse patients with HF, broadly representative of actual clinical practice, who were managed according to routine practice and a streamlined study protocol at more than 60 U.S. centers, Dr. Mentz observed.
One of the pragmatic design’s advantages, he told this news organization, was “how efficient it was” as a randomized comparison of treatment strategies for clinical outcomes. It was “relatively low cost” and recruited patients quickly, compared with conventional randomized trials, “and we answered the question clearly.” The trial’s results, Dr. Mentz said, reflect “what happens in the real world.”
When might torsemide have the edge?
Although furosemide is the most commonly used loop diuretic in HF, and there are others besides it and torsemide, the latter has both known and theoretical advantages that set it apart. Torsemide is more than twice as potent as furosemide and more bioavailable, and its treatment effect lasts longer, the TRANSFORM-HF investigators have noted.
In addition, preclinical and small clinical studies suggest torsemide may have pleiotropic effects that might be theoretical advantages for patients with HF. For example, it appears to downregulate the renin-angiotensin-aldosterone system (RAAS) and reduce myocardial fibrosis and promote reverse ventricular remodeling, the group writes.
In practice, therefore, torsemide may be preferred in patients with furosemide resistance or “challenges with bioavailability, especially those with very advanced heart failure with congestion who may have gut edema, where oral furosemide and other loop diuretics are not effectively absorbed,” Biykem Bozkurt, MD, PhD, Baylor College of Medicine, Houston, told this news organization.
In such patients, she said, torsemide “is considered to be a better choice for individuals who have diuretic resistance with advanced congestion.”
The drug’s apparent pleiotropic effects, such as RAAS inhibition, may have less relevance to the TRANSFORM-HF primary endpoint of all-cause mortality than to clinical outcomes more likely associated with successful decongestion, such as HF hospitalization, Dr. Bozkurt proposed.
The trial’s pragmatic design, however, made it more feasible to focus on all-cause mortality and less practical to use measures of successful decongestion, such as volume loss or reduction in natriuretic peptide levels, she observed. Those are endpoints of special interest when diuretics are compared, “especially for the subgroup of patients who are diuretic resistant.”
Over the last 20 years or so, “we’ve learned that hospitalized heart failure is a very different disease process with a different natural history,” observed Clyde W. Yancy, MD, MSc, Northwestern University, Chicago, who was not part of the current study.
“So, the idea that something as nuanced as choice of one loop diuretic over the other, in that setting, would be sufficient to change the natural history, may be still a high bar for us,” he said in an interview.
“Based on these data, one would have to argue that whichever loop diuretic you select for the hospitalized patient – and a lot of that is driven by market exigencies right now – it turns out that the response is indistinguishable,” Dr. Yancy said. “That means if your hospital happens to have furosemide on the formulary, use it. If furosemide is not available but torsemide is available, use it.”
Dr. Yancy said he’d like to see a trial similar to TRANSFORM-HF but in outpatients receiving today’s guideline-directed medical therapy, which includes the sodium-glucose cotransporter 2 (SGLT2) inhibitors, drugs that increase the fractional excretion of sodium and have a “diureticlike” effect.
Such a trial, he said, would explore “the combination of not one, or two, but three agents with a diuretic effect – a loop diuretic, a mineralocorticoid antagonist, and an SGLT2 inhibitor – in ambulatory, optimized patients. It might make a difference.”
HF regardless of EF
The trial enrolled patients hospitalized with worsening or new-onset HF with a plan for long-term loop diuretic therapy who had either an EF of 40% or lower or, regardless of EF, elevated natriuretic peptide levels when hospitalized.
Of the 2,859 participants, whose mean age was about 65 years, about 36% were women and 34% African American. Overall, 1,428 were assigned to receive furosemide as their initial oral diuretic and 1,431 patients were assigned to the torsemide-first strategy.
The rate of death from any cause in both groups was 17 per 100 patient-years at a median of 17.4 months. The hazard ratio for torsemide vs. furosemide was 1.02 (95% confidence interval, 0.89-1.18; P = .77).
The corresponding HR at 12 months for all-cause death or hospitalization was 0.92 (95% CI, 0.83-1.02; P = .11). The relative risk for any hospitalization was 0.94 (95% CI, 0.84-1.07).
Pragmatic design: Other implications
Dosing was left to clinician discretion in the open-label study, as was whether patients maintained their assigned drug or switched over to the other agent. Indeed, 5.4% of patients crossed over to the other loop diuretic, and 2.8% went off loop diuretics entirely between in-hospital randomization and discharge, Dr. Mentz reported. By day 30, 6.7% had crossed over, and 7% had stopped taking loop diuretics.
The diuretic crossovers and discontinuations, Dr. Mentz said, likely biased the trial’s outcomes, such that the two strategies performed about equally well. Efforts were made, however, to at least partially overcome that limitation.
“We put measures in place to support adherence – sending letters to their primary doctors, giving them a wallet card so they would know which therapy they were on, having conversations about the importance of trying to stay on the randomized therapy,” Dr. Mentz said in an interview. Still, some clinicians saw differences between the two agents that prompted them, at some point, to switch patients from one loop diuretic to the other.
But interestingly, Dr. Mentz reported, the two strategies did not significantly differ in all-cause mortality or the composite of all-cause mortality or hospitalization in analysis by intention to treat.
Dr. Mentz discloses receiving honoraria from AstraZeneca, Bayer/Merck, Boehringer Ingelheim/Lilly, Cytokinetics, Pharmacosmos, Respicardia, Windtree Therapeutics, and Zoll; and research grants from American Regent and Novartis. Dr. Bozkurt discloses receiving honoraria from AstraZeneca, Baxter Health Care, and Sanofi Aventis and having other relationships with Renovacor, Respicardia, Abbott Vascular, Liva Nova, Vifor, and Cardurion. Dr. Yancy discloses a modest relationship with Abbott.
A version of this article first appeared on Medscape.com.
CHICAGO – The choice of loop diuretic for decongestion in patients hospitalized with heart failure (HF) may make little difference to survival or readmission risk over the next year, at least when deciding between furosemide or torsemide, a randomized trial suggests.
Both drugs are old and widely used, but differences between the two loop diuretics in bioavailability, effects on potassium levels, and other features have led some clinicians to sometimes prefer torsemide. Until now, however, no randomized HF trials have compared the two drugs.
The new findings suggest clinicians can continue starting such patients with HF on either agent, at their discretion, without concern that the choice may compromise outcomes, say researchers from the TRANSFORM-HF trial, which compared furosemide-first and torsemide-first diuretic strategies in a diverse population of patients with HF.
Given that the two strategies were similarly effective for survival and rehospitalization, clinicians caring for patients with HF can focus more on “getting patients on the right dose for their loop diuretic, and prioritizing those therapies proven to improve clinical outcomes,” said Robert J. Mentz, MD, of Duke University Clinical Research Institute, Durham, N.C.
Dr. Mentz, a TRANSFORM-HF principal investigator, presented the primary results November 5 at the American Heart Association scientific sessions.
The trial had randomly assigned 2,859 patients hospitalized with HF and with a plan for oral loop diuretic therapy to initiate treatment with furosemide or torsemide. Clinicians were encouraged to maintain patients on the assigned diuretic, but crossovers to the other drug or other diuretic changes were allowed.
Rates of death from any cause, the primary endpoint, were about 26% in both groups over a median 17-month follow-up, regardless of ejection fraction (EF).
The composite rates of all-cause death or hospitalization at 12 months were also not significantly different, about 49% for those started on furosemide and about 47% for patients initially prescribed torsemide.
As a pragmatic comparative effectiveness trial, TRANSFORM-HF entered diverse patients with HF, broadly representative of actual clinical practice, who were managed according to routine practice and a streamlined study protocol at more than 60 U.S. centers, Dr. Mentz observed.
One of the pragmatic design’s advantages, he told this news organization, was “how efficient it was” as a randomized comparison of treatment strategies for clinical outcomes. It was “relatively low cost” and recruited patients quickly, compared with conventional randomized trials, “and we answered the question clearly.” The trial’s results, Dr. Mentz said, reflect “what happens in the real world.”
When might torsemide have the edge?
Although furosemide is the most commonly used loop diuretic in HF, and there are others besides it and torsemide, the latter has both known and theoretical advantages that set it apart. Torsemide is more than twice as potent as furosemide and more bioavailable, and its treatment effect lasts longer, the TRANSFORM-HF investigators have noted.
In addition, preclinical and small clinical studies suggest torsemide may have pleiotropic effects that might be theoretical advantages for patients with HF. For example, it appears to downregulate the renin-angiotensin-aldosterone system (RAAS) and reduce myocardial fibrosis and promote reverse ventricular remodeling, the group writes.
In practice, therefore, torsemide may be preferred in patients with furosemide resistance or “challenges with bioavailability, especially those with very advanced heart failure with congestion who may have gut edema, where oral furosemide and other loop diuretics are not effectively absorbed,” Biykem Bozkurt, MD, PhD, Baylor College of Medicine, Houston, told this news organization.
In such patients, she said, torsemide “is considered to be a better choice for individuals who have diuretic resistance with advanced congestion.”
The drug’s apparent pleiotropic effects, such as RAAS inhibition, may have less relevance to the TRANSFORM-HF primary endpoint of all-cause mortality than to clinical outcomes more likely associated with successful decongestion, such as HF hospitalization, Dr. Bozkurt proposed.
The trial’s pragmatic design, however, made it more feasible to focus on all-cause mortality and less practical to use measures of successful decongestion, such as volume loss or reduction in natriuretic peptide levels, she observed. Those are endpoints of special interest when diuretics are compared, “especially for the subgroup of patients who are diuretic resistant.”
Over the last 20 years or so, “we’ve learned that hospitalized heart failure is a very different disease process with a different natural history,” observed Clyde W. Yancy, MD, MSc, Northwestern University, Chicago, who was not part of the current study.
“So, the idea that something as nuanced as choice of one loop diuretic over the other, in that setting, would be sufficient to change the natural history, may be still a high bar for us,” he said in an interview.
“Based on these data, one would have to argue that whichever loop diuretic you select for the hospitalized patient – and a lot of that is driven by market exigencies right now – it turns out that the response is indistinguishable,” Dr. Yancy said. “That means if your hospital happens to have furosemide on the formulary, use it. If furosemide is not available but torsemide is available, use it.”
Dr. Yancy said he’d like to see a trial similar to TRANSFORM-HF but in outpatients receiving today’s guideline-directed medical therapy, which includes the sodium-glucose cotransporter 2 (SGLT2) inhibitors, drugs that increase the fractional excretion of sodium and have a “diureticlike” effect.
Such a trial, he said, would explore “the combination of not one, or two, but three agents with a diuretic effect – a loop diuretic, a mineralocorticoid antagonist, and an SGLT2 inhibitor – in ambulatory, optimized patients. It might make a difference.”
HF regardless of EF
The trial enrolled patients hospitalized with worsening or new-onset HF with a plan for long-term loop diuretic therapy who had either an EF of 40% or lower or, regardless of EF, elevated natriuretic peptide levels when hospitalized.
Of the 2,859 participants, whose mean age was about 65 years, about 36% were women and 34% African American. Overall, 1,428 were assigned to receive furosemide as their initial oral diuretic and 1,431 patients were assigned to the torsemide-first strategy.
The rate of death from any cause in both groups was 17 per 100 patient-years at a median of 17.4 months. The hazard ratio for torsemide vs. furosemide was 1.02 (95% confidence interval, 0.89-1.18; P = .77).
The corresponding HR at 12 months for all-cause death or hospitalization was 0.92 (95% CI, 0.83-1.02; P = .11). The relative risk for any hospitalization was 0.94 (95% CI, 0.84-1.07).
Pragmatic design: Other implications
Dosing was left to clinician discretion in the open-label study, as was whether patients maintained their assigned drug or switched over to the other agent. Indeed, 5.4% of patients crossed over to the other loop diuretic, and 2.8% went off loop diuretics entirely between in-hospital randomization and discharge, Dr. Mentz reported. By day 30, 6.7% had crossed over, and 7% had stopped taking loop diuretics.
The diuretic crossovers and discontinuations, Dr. Mentz said, likely biased the trial’s outcomes, such that the two strategies performed about equally well. Efforts were made, however, to at least partially overcome that limitation.
“We put measures in place to support adherence – sending letters to their primary doctors, giving them a wallet card so they would know which therapy they were on, having conversations about the importance of trying to stay on the randomized therapy,” Dr. Mentz said in an interview. Still, some clinicians saw differences between the two agents that prompted them, at some point, to switch patients from one loop diuretic to the other.
But interestingly, Dr. Mentz reported, the two strategies did not significantly differ in all-cause mortality or the composite of all-cause mortality or hospitalization in analysis by intention to treat.
Dr. Mentz discloses receiving honoraria from AstraZeneca, Bayer/Merck, Boehringer Ingelheim/Lilly, Cytokinetics, Pharmacosmos, Respicardia, Windtree Therapeutics, and Zoll; and research grants from American Regent and Novartis. Dr. Bozkurt discloses receiving honoraria from AstraZeneca, Baxter Health Care, and Sanofi Aventis and having other relationships with Renovacor, Respicardia, Abbott Vascular, Liva Nova, Vifor, and Cardurion. Dr. Yancy discloses a modest relationship with Abbott.
A version of this article first appeared on Medscape.com.
AT AHA 2022
Acute heart failure risk assessment in ED improves outcomes: COACH
CHICAGO – Systematic mortality-risk assessment of patients who presented to hospital emergency departments for acute heart failure led to better patient outcomes in a controlled Canadian trial with more than 5,000 patients.
Thirty days after patients presented, the incidence of death from any cause or hospitalization for cardiovascular causes – one of two primary endpoints in the COACH study – was 12.1% among patients who underwent acute risk assessment and 14.5% in control patients who did not undergo this assessment, which translated into an adjusted, significant 12% relative risk reduction for the patients who underwent systematic assessment, Douglas S. Lee, MD, PhD, said at the American Heart Association scientific sessions.
The study’s second primary endpoint, the incidence of the same combined outcome 20 months after initial presentation, was 54.4% among the 2,480 patients assessed with the risk-assessment tool and 56.2% in the 2,972 controls, a significant, adjusted relative risk reduction of 5%.
This benefit was primarily driven by reductions in cardiovascular hospitalizations, which fell by an adjusted 16% in the intervention group compared with controls, and more specifically by hospitalizations for heart failure, which tallied a relative 20% less with the intervention. Both were significant between-group differences.
The other portion of the combined endpoint, all-cause mortality, was not significantly different between the patients who underwent the systematic emergency department assessment and the controls who were managed using usual emergency-department protocols.
Simultaneous with the report, the results also appeared online in the New England Journal of Medicine.
A pathway for early discharge and improved outcomes
“Implementation of this approach may lead to a pathway for early discharge from the hospital or emergency department, and improved patient outcomes,” said Dr. Lee, a professor at the University of Toronto, and a senior core scientist at the ICES Cardiovascular Research Program in Toronto.
“The treatment effect on the primary process outcome – patients admitted or discharged – will add useful insights into how intervention may improve,” commented Harriette Van Spall, MD, who was designated discussant for the report. The findings “fill an important knowledge gap,” added Dr. Van Spall, a cardiologist at McMaster University in Hamilton, Ont. The results “have important implications for health resource utilization,” she said.
The risk assessment tool used in the study is called the Emergency Heart failure Mortality Risk Grade for 30-day mortality (EHMRG30-ST), which was devised and validated by Dr. Lee and his associates. The assessment tool uses 11 clinical variables that include age, systolic blood pressure, heart rate, oxygen saturation, potassium and creatinine levels, and presence of ST depression on a 12-lead ECG.
The study design recommended that patients be discharged early and receive standardized transitional care as outpatients if they had a low risk of death within 7 days and within 30 days as estimated by the EHMRG30-ST. The protocol recommended that patients scored as high risk should be admitted to the hospital, and that clinicians use their clinical judgment for intermediate-risk patients but favor admission for intermediate to high risk and discharge for low to intermediate risk. The study ran at 10 hospitals in Ontario. Initially, all 10 hospitals assessed patients by usual care, and then, over time, each hospital began using the tool so that by the end of the study all 10 hospitals employed it. Among the 2,480 patients seen during the active phase, 2,442 actually underwent assessment, with 24% rated as low risk, 32% rated as intermediate risk, and 44% judged to have high risk.
The researchers also ran risk assessments retrospectively on the controls, who showed a roughly similar risk distribution, with 18% low risk, 28% intermediate risk, and 54% high risk.
The patients averaged 78 years of age, 55% were men, about 40% had diabetes, and about 64% had a prior heart failure diagnosis.
Heart failure admissions have become ‘a big deal’
Emergency department clinicians and heart failure cardiologists “have worked together for a long time” when making decisions about which patients with acute heart failure need hospital admission, commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation programs at Ascension St. Vincent Heart Center in Indianapolis. These decisions “became a big deal” a decade ago when the U.S. Centers for Medicare & Medicaid Services launched the Hospital Readmissions Reduction Program that began to penalize hospitals for high rates of hospital readmissions for several conditions including heart failure, she said in an interview.
“If a heart failure patient is not admitted, they can’t be readmitted,” Dr. Walsh noted.
“Many risk-assessment tools exist for patients once they are hospitalized, but these tools have not been used in emergency departments. The take-home message is that we need to start risk assessment sooner, in the emergency department,” she said.
But the specific approach tested in the COACH trial needs more study and may need further tweaking to work in the United States, where it is not clear who would pay for a program like the one tested in the trial. Canada’s unified health care payment system makes the COACH approach more financially feasible, Dr. Walsh commented.
COACH received no commercial funding. Dr. Lee, Dr. Van Spall, and Dr. Walsh had no disclosures.
CHICAGO – Systematic mortality-risk assessment of patients who presented to hospital emergency departments for acute heart failure led to better patient outcomes in a controlled Canadian trial with more than 5,000 patients.
Thirty days after patients presented, the incidence of death from any cause or hospitalization for cardiovascular causes – one of two primary endpoints in the COACH study – was 12.1% among patients who underwent acute risk assessment and 14.5% in control patients who did not undergo this assessment, which translated into an adjusted, significant 12% relative risk reduction for the patients who underwent systematic assessment, Douglas S. Lee, MD, PhD, said at the American Heart Association scientific sessions.
The study’s second primary endpoint, the incidence of the same combined outcome 20 months after initial presentation, was 54.4% among the 2,480 patients assessed with the risk-assessment tool and 56.2% in the 2,972 controls, a significant, adjusted relative risk reduction of 5%.
This benefit was primarily driven by reductions in cardiovascular hospitalizations, which fell by an adjusted 16% in the intervention group compared with controls, and more specifically by hospitalizations for heart failure, which tallied a relative 20% less with the intervention. Both were significant between-group differences.
The other portion of the combined endpoint, all-cause mortality, was not significantly different between the patients who underwent the systematic emergency department assessment and the controls who were managed using usual emergency-department protocols.
Simultaneous with the report, the results also appeared online in the New England Journal of Medicine.
A pathway for early discharge and improved outcomes
“Implementation of this approach may lead to a pathway for early discharge from the hospital or emergency department, and improved patient outcomes,” said Dr. Lee, a professor at the University of Toronto, and a senior core scientist at the ICES Cardiovascular Research Program in Toronto.
“The treatment effect on the primary process outcome – patients admitted or discharged – will add useful insights into how intervention may improve,” commented Harriette Van Spall, MD, who was designated discussant for the report. The findings “fill an important knowledge gap,” added Dr. Van Spall, a cardiologist at McMaster University in Hamilton, Ont. The results “have important implications for health resource utilization,” she said.
The risk assessment tool used in the study is called the Emergency Heart failure Mortality Risk Grade for 30-day mortality (EHMRG30-ST), which was devised and validated by Dr. Lee and his associates. The assessment tool uses 11 clinical variables that include age, systolic blood pressure, heart rate, oxygen saturation, potassium and creatinine levels, and presence of ST depression on a 12-lead ECG.
The study design recommended that patients be discharged early and receive standardized transitional care as outpatients if they had a low risk of death within 7 days and within 30 days as estimated by the EHMRG30-ST. The protocol recommended that patients scored as high risk should be admitted to the hospital, and that clinicians use their clinical judgment for intermediate-risk patients but favor admission for intermediate to high risk and discharge for low to intermediate risk. The study ran at 10 hospitals in Ontario. Initially, all 10 hospitals assessed patients by usual care, and then, over time, each hospital began using the tool so that by the end of the study all 10 hospitals employed it. Among the 2,480 patients seen during the active phase, 2,442 actually underwent assessment, with 24% rated as low risk, 32% rated as intermediate risk, and 44% judged to have high risk.
The researchers also ran risk assessments retrospectively on the controls, who showed a roughly similar risk distribution, with 18% low risk, 28% intermediate risk, and 54% high risk.
The patients averaged 78 years of age, 55% were men, about 40% had diabetes, and about 64% had a prior heart failure diagnosis.
Heart failure admissions have become ‘a big deal’
Emergency department clinicians and heart failure cardiologists “have worked together for a long time” when making decisions about which patients with acute heart failure need hospital admission, commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation programs at Ascension St. Vincent Heart Center in Indianapolis. These decisions “became a big deal” a decade ago when the U.S. Centers for Medicare & Medicaid Services launched the Hospital Readmissions Reduction Program that began to penalize hospitals for high rates of hospital readmissions for several conditions including heart failure, she said in an interview.
“If a heart failure patient is not admitted, they can’t be readmitted,” Dr. Walsh noted.
“Many risk-assessment tools exist for patients once they are hospitalized, but these tools have not been used in emergency departments. The take-home message is that we need to start risk assessment sooner, in the emergency department,” she said.
But the specific approach tested in the COACH trial needs more study and may need further tweaking to work in the United States, where it is not clear who would pay for a program like the one tested in the trial. Canada’s unified health care payment system makes the COACH approach more financially feasible, Dr. Walsh commented.
COACH received no commercial funding. Dr. Lee, Dr. Van Spall, and Dr. Walsh had no disclosures.
CHICAGO – Systematic mortality-risk assessment of patients who presented to hospital emergency departments for acute heart failure led to better patient outcomes in a controlled Canadian trial with more than 5,000 patients.
Thirty days after patients presented, the incidence of death from any cause or hospitalization for cardiovascular causes – one of two primary endpoints in the COACH study – was 12.1% among patients who underwent acute risk assessment and 14.5% in control patients who did not undergo this assessment, which translated into an adjusted, significant 12% relative risk reduction for the patients who underwent systematic assessment, Douglas S. Lee, MD, PhD, said at the American Heart Association scientific sessions.
The study’s second primary endpoint, the incidence of the same combined outcome 20 months after initial presentation, was 54.4% among the 2,480 patients assessed with the risk-assessment tool and 56.2% in the 2,972 controls, a significant, adjusted relative risk reduction of 5%.
This benefit was primarily driven by reductions in cardiovascular hospitalizations, which fell by an adjusted 16% in the intervention group compared with controls, and more specifically by hospitalizations for heart failure, which tallied a relative 20% less with the intervention. Both were significant between-group differences.
The other portion of the combined endpoint, all-cause mortality, was not significantly different between the patients who underwent the systematic emergency department assessment and the controls who were managed using usual emergency-department protocols.
Simultaneous with the report, the results also appeared online in the New England Journal of Medicine.
A pathway for early discharge and improved outcomes
“Implementation of this approach may lead to a pathway for early discharge from the hospital or emergency department, and improved patient outcomes,” said Dr. Lee, a professor at the University of Toronto, and a senior core scientist at the ICES Cardiovascular Research Program in Toronto.
“The treatment effect on the primary process outcome – patients admitted or discharged – will add useful insights into how intervention may improve,” commented Harriette Van Spall, MD, who was designated discussant for the report. The findings “fill an important knowledge gap,” added Dr. Van Spall, a cardiologist at McMaster University in Hamilton, Ont. The results “have important implications for health resource utilization,” she said.
The risk assessment tool used in the study is called the Emergency Heart failure Mortality Risk Grade for 30-day mortality (EHMRG30-ST), which was devised and validated by Dr. Lee and his associates. The assessment tool uses 11 clinical variables that include age, systolic blood pressure, heart rate, oxygen saturation, potassium and creatinine levels, and presence of ST depression on a 12-lead ECG.
The study design recommended that patients be discharged early and receive standardized transitional care as outpatients if they had a low risk of death within 7 days and within 30 days as estimated by the EHMRG30-ST. The protocol recommended that patients scored as high risk should be admitted to the hospital, and that clinicians use their clinical judgment for intermediate-risk patients but favor admission for intermediate to high risk and discharge for low to intermediate risk. The study ran at 10 hospitals in Ontario. Initially, all 10 hospitals assessed patients by usual care, and then, over time, each hospital began using the tool so that by the end of the study all 10 hospitals employed it. Among the 2,480 patients seen during the active phase, 2,442 actually underwent assessment, with 24% rated as low risk, 32% rated as intermediate risk, and 44% judged to have high risk.
The researchers also ran risk assessments retrospectively on the controls, who showed a roughly similar risk distribution, with 18% low risk, 28% intermediate risk, and 54% high risk.
The patients averaged 78 years of age, 55% were men, about 40% had diabetes, and about 64% had a prior heart failure diagnosis.
Heart failure admissions have become ‘a big deal’
Emergency department clinicians and heart failure cardiologists “have worked together for a long time” when making decisions about which patients with acute heart failure need hospital admission, commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation programs at Ascension St. Vincent Heart Center in Indianapolis. These decisions “became a big deal” a decade ago when the U.S. Centers for Medicare & Medicaid Services launched the Hospital Readmissions Reduction Program that began to penalize hospitals for high rates of hospital readmissions for several conditions including heart failure, she said in an interview.
“If a heart failure patient is not admitted, they can’t be readmitted,” Dr. Walsh noted.
“Many risk-assessment tools exist for patients once they are hospitalized, but these tools have not been used in emergency departments. The take-home message is that we need to start risk assessment sooner, in the emergency department,” she said.
But the specific approach tested in the COACH trial needs more study and may need further tweaking to work in the United States, where it is not clear who would pay for a program like the one tested in the trial. Canada’s unified health care payment system makes the COACH approach more financially feasible, Dr. Walsh commented.
COACH received no commercial funding. Dr. Lee, Dr. Van Spall, and Dr. Walsh had no disclosures.
AT AHA 2022
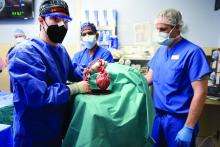
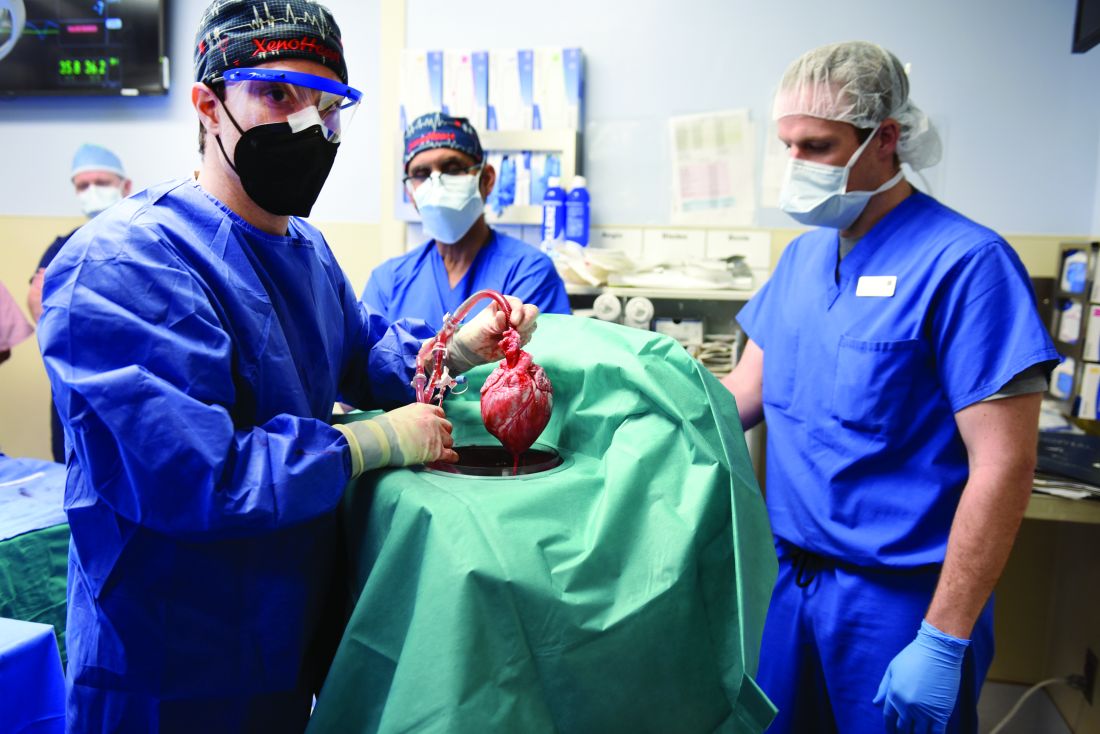
Puzzling, unique ECG from pig-to-human transplanted heart
In the first transplant of a genetically altered pig heart into a human in January, initial unexpected, prolonged ECG readings apparently did not affect the heart’s function, although the organ suddenly began to fail at day 50.
A study of these ECG changes, scheduled for presentation by Calvin Kagan, MD, and colleagues at the American Heart Association scientific sessions, offers insight into this novel operation.
As widely reported, the patient, 57-year-old David Bennett of Maryland, had end-stage heart disease and was a poor candidate for a ventricular assist device and was ineligible for a human heart, when he consented to be the first human to be transplanted with a pig heart that had a number of genes added or subtracted with the goal, in part, to prevent rejection.
The heart initially performed well after it was transplanted in an operation at the University of Maryland School of Medicine (UMSOM) in Baltimore on Jan. 7, but failed in the second month, and Mr. Bennett died on March 9.
The Food and Drug Administration had granted emergency authorization for the surgery through its expanded access (compassionate use) program, coauthor Muhammad Mohiuddin, MD, said in an interview.
“We have learned a lot and hope we can do more,” said Dr. Mohiuddin, scientific and program director of the cardiac xenotransplantation program at UMSOM.
“Suddenly on day 50, the heart started to get thicker and was not relaxing enough,” explained senior author Timm-Michael Dickfeld, MD, PhD, director of electrophysiology research at UMSOM. A biopsy revealed substantial buildup of interstitial fluid that restricted movement. The fluid was replaced by fibrous tissue, leading to irreversible damage.
Persistent, prolonged ECG parameters
In the heart from a genetically modified pig, three genes associated with antibody-mediated rejection and a gene associated with pig heart tissue growth had been inactivated and six human genes associated with immune acceptance had been added. The donor pig was supplied by Revivicor (Blacksburg, Va.).
The patient’s immunosuppressant therapy included an experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Mass.).
The patient had daily 12-lead ECGs after the transplant.
In prior research using a pig heart transplanted into a pig body, ECG readings showed a short PR interval (50-120 ms), short QRS duration (70-90 ms) and short QT intervals (260-380 ms).
However, in the transplanted xenograft heart, the initial ECG readings showed a longer PR interval of 190 ms, QRS duration of 138 ms, and QT of 538 ms.
Prolonged intrinsic PR intervals remained stable during the postoperative course (210 ms, range 142-246 ms).
QRS duration also remained prolonged (145 ms, range 116-192 ms), but shortened during the postoperative course (days 21-40 vs. 41-60: 148 ms vs. 132 ms; P < .001).
Increased QT persisted (509 ms, range 384-650 ms) with dynamic fluctuations. The shortest QT duration was observed on day 14 (P < .001).
“In a human heart, when those parameters get longer, this can indicate signs of electrical or myocardial disease,” Dr. Dickfeld explained in a press release from the AHA.
“The QRS duration may prolong when, for example, the muscle and the electrical system itself is diseased, and that is why it takes a long time for electricity to travel from cell to cell and travel from one side of the heart to the other,” he said.
“In the human heart, the QT duration is correlated with an increased risk of abnormal heart rhythms,” he noted. “In our patient, it was concerning that the QT measure was prolonged. While we saw some fluctuations, the QT measure remained prolonged during the whole 61 days.”
‘Interesting study’
Two experts who were not involved with this research weighed in on the findings for this news organization.
“This very interesting study reinforces the difficulties in xenotransplantation, and the need for more research to be able to safely monitor recipients, as baseline values are unknown,” said Edward Vigmond, PhD.
Dr. Vigmond, from the Electrophysiology and Heart Modeling Institute at the University of Bordeaux in France, published a related study about a model of translation of pig to human electrophysiology.
The ECG is sensitive to the electrical activation pattern of the heart, along with the cellular and tissue electrical properties, he noted.
“Although pigs and humans may be similar in size, there are many differences between them,” Dr. Vigmond observed, including “the extent of the rapid conduction system of the heart, the number of nuclei in the muscle cells, the proteins in the cell membrane which control electrical activity, the orientation of the heart and thorax, and the handling of calcium inside the cell.”
“On top of this,” he continued, “donor hearts are denervated, so they no longer respond to nervous modulation, and circulating compounds in the blood which affect heart function vary between species.
“With all these differences, it is not surprising that the ECG of a pig heart transplanted into a human resembles neither that of a human nor that of a pig,” Dr. Vigmond said.
“It is interesting to note that the humanized-gene-edited porcine heart exhibited abnormal electrical conduction parameters from the outset,” said Mandeep R. Mehra, MD.
“Whether these changes were due to the gene modifications (i.e., already inherent in the pig ECG prior to transplant) or a result of the transplant operation challenges (such as the ischemia reperfusion injury and early immunological interactions) is uncertain and should be clarified,” said Dr. Mehra, of Harvard Medical School and Brigham and Women’s Medicine in Boston.
“Knowledge of these changes is important to determine whether a simple ECG parameter may be useful to identify changes that could indicate developing pathology,” Dr. Mehra added.
“In the older days of human transplantation, we often used ECG parameters such as a change in voltage amplitude to identify signals for rejection,” he continued. “Whether such changes occurred in this case could be another interesting aspect to explore as changes occurred in cardiac performance in response to the physiological and pathological challenges that were encountered in this sentinel case.”
The study authors reported having no outside sources of funding.
A version of this article first appeared on Medscape.com.
In the first transplant of a genetically altered pig heart into a human in January, initial unexpected, prolonged ECG readings apparently did not affect the heart’s function, although the organ suddenly began to fail at day 50.
A study of these ECG changes, scheduled for presentation by Calvin Kagan, MD, and colleagues at the American Heart Association scientific sessions, offers insight into this novel operation.
As widely reported, the patient, 57-year-old David Bennett of Maryland, had end-stage heart disease and was a poor candidate for a ventricular assist device and was ineligible for a human heart, when he consented to be the first human to be transplanted with a pig heart that had a number of genes added or subtracted with the goal, in part, to prevent rejection.
The heart initially performed well after it was transplanted in an operation at the University of Maryland School of Medicine (UMSOM) in Baltimore on Jan. 7, but failed in the second month, and Mr. Bennett died on March 9.
The Food and Drug Administration had granted emergency authorization for the surgery through its expanded access (compassionate use) program, coauthor Muhammad Mohiuddin, MD, said in an interview.
“We have learned a lot and hope we can do more,” said Dr. Mohiuddin, scientific and program director of the cardiac xenotransplantation program at UMSOM.
“Suddenly on day 50, the heart started to get thicker and was not relaxing enough,” explained senior author Timm-Michael Dickfeld, MD, PhD, director of electrophysiology research at UMSOM. A biopsy revealed substantial buildup of interstitial fluid that restricted movement. The fluid was replaced by fibrous tissue, leading to irreversible damage.
Persistent, prolonged ECG parameters
In the heart from a genetically modified pig, three genes associated with antibody-mediated rejection and a gene associated with pig heart tissue growth had been inactivated and six human genes associated with immune acceptance had been added. The donor pig was supplied by Revivicor (Blacksburg, Va.).
The patient’s immunosuppressant therapy included an experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Mass.).
The patient had daily 12-lead ECGs after the transplant.
In prior research using a pig heart transplanted into a pig body, ECG readings showed a short PR interval (50-120 ms), short QRS duration (70-90 ms) and short QT intervals (260-380 ms).
However, in the transplanted xenograft heart, the initial ECG readings showed a longer PR interval of 190 ms, QRS duration of 138 ms, and QT of 538 ms.
Prolonged intrinsic PR intervals remained stable during the postoperative course (210 ms, range 142-246 ms).
QRS duration also remained prolonged (145 ms, range 116-192 ms), but shortened during the postoperative course (days 21-40 vs. 41-60: 148 ms vs. 132 ms; P < .001).
Increased QT persisted (509 ms, range 384-650 ms) with dynamic fluctuations. The shortest QT duration was observed on day 14 (P < .001).
“In a human heart, when those parameters get longer, this can indicate signs of electrical or myocardial disease,” Dr. Dickfeld explained in a press release from the AHA.
“The QRS duration may prolong when, for example, the muscle and the electrical system itself is diseased, and that is why it takes a long time for electricity to travel from cell to cell and travel from one side of the heart to the other,” he said.
“In the human heart, the QT duration is correlated with an increased risk of abnormal heart rhythms,” he noted. “In our patient, it was concerning that the QT measure was prolonged. While we saw some fluctuations, the QT measure remained prolonged during the whole 61 days.”
‘Interesting study’
Two experts who were not involved with this research weighed in on the findings for this news organization.
“This very interesting study reinforces the difficulties in xenotransplantation, and the need for more research to be able to safely monitor recipients, as baseline values are unknown,” said Edward Vigmond, PhD.
Dr. Vigmond, from the Electrophysiology and Heart Modeling Institute at the University of Bordeaux in France, published a related study about a model of translation of pig to human electrophysiology.
The ECG is sensitive to the electrical activation pattern of the heart, along with the cellular and tissue electrical properties, he noted.
“Although pigs and humans may be similar in size, there are many differences between them,” Dr. Vigmond observed, including “the extent of the rapid conduction system of the heart, the number of nuclei in the muscle cells, the proteins in the cell membrane which control electrical activity, the orientation of the heart and thorax, and the handling of calcium inside the cell.”
“On top of this,” he continued, “donor hearts are denervated, so they no longer respond to nervous modulation, and circulating compounds in the blood which affect heart function vary between species.
“With all these differences, it is not surprising that the ECG of a pig heart transplanted into a human resembles neither that of a human nor that of a pig,” Dr. Vigmond said.
“It is interesting to note that the humanized-gene-edited porcine heart exhibited abnormal electrical conduction parameters from the outset,” said Mandeep R. Mehra, MD.
“Whether these changes were due to the gene modifications (i.e., already inherent in the pig ECG prior to transplant) or a result of the transplant operation challenges (such as the ischemia reperfusion injury and early immunological interactions) is uncertain and should be clarified,” said Dr. Mehra, of Harvard Medical School and Brigham and Women’s Medicine in Boston.
“Knowledge of these changes is important to determine whether a simple ECG parameter may be useful to identify changes that could indicate developing pathology,” Dr. Mehra added.
“In the older days of human transplantation, we often used ECG parameters such as a change in voltage amplitude to identify signals for rejection,” he continued. “Whether such changes occurred in this case could be another interesting aspect to explore as changes occurred in cardiac performance in response to the physiological and pathological challenges that were encountered in this sentinel case.”
The study authors reported having no outside sources of funding.
A version of this article first appeared on Medscape.com.
In the first transplant of a genetically altered pig heart into a human in January, initial unexpected, prolonged ECG readings apparently did not affect the heart’s function, although the organ suddenly began to fail at day 50.
A study of these ECG changes, scheduled for presentation by Calvin Kagan, MD, and colleagues at the American Heart Association scientific sessions, offers insight into this novel operation.
As widely reported, the patient, 57-year-old David Bennett of Maryland, had end-stage heart disease and was a poor candidate for a ventricular assist device and was ineligible for a human heart, when he consented to be the first human to be transplanted with a pig heart that had a number of genes added or subtracted with the goal, in part, to prevent rejection.
The heart initially performed well after it was transplanted in an operation at the University of Maryland School of Medicine (UMSOM) in Baltimore on Jan. 7, but failed in the second month, and Mr. Bennett died on March 9.
The Food and Drug Administration had granted emergency authorization for the surgery through its expanded access (compassionate use) program, coauthor Muhammad Mohiuddin, MD, said in an interview.
“We have learned a lot and hope we can do more,” said Dr. Mohiuddin, scientific and program director of the cardiac xenotransplantation program at UMSOM.
“Suddenly on day 50, the heart started to get thicker and was not relaxing enough,” explained senior author Timm-Michael Dickfeld, MD, PhD, director of electrophysiology research at UMSOM. A biopsy revealed substantial buildup of interstitial fluid that restricted movement. The fluid was replaced by fibrous tissue, leading to irreversible damage.
Persistent, prolonged ECG parameters
In the heart from a genetically modified pig, three genes associated with antibody-mediated rejection and a gene associated with pig heart tissue growth had been inactivated and six human genes associated with immune acceptance had been added. The donor pig was supplied by Revivicor (Blacksburg, Va.).
The patient’s immunosuppressant therapy included an experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Mass.).
The patient had daily 12-lead ECGs after the transplant.
In prior research using a pig heart transplanted into a pig body, ECG readings showed a short PR interval (50-120 ms), short QRS duration (70-90 ms) and short QT intervals (260-380 ms).
However, in the transplanted xenograft heart, the initial ECG readings showed a longer PR interval of 190 ms, QRS duration of 138 ms, and QT of 538 ms.
Prolonged intrinsic PR intervals remained stable during the postoperative course (210 ms, range 142-246 ms).
QRS duration also remained prolonged (145 ms, range 116-192 ms), but shortened during the postoperative course (days 21-40 vs. 41-60: 148 ms vs. 132 ms; P < .001).
Increased QT persisted (509 ms, range 384-650 ms) with dynamic fluctuations. The shortest QT duration was observed on day 14 (P < .001).
“In a human heart, when those parameters get longer, this can indicate signs of electrical or myocardial disease,” Dr. Dickfeld explained in a press release from the AHA.
“The QRS duration may prolong when, for example, the muscle and the electrical system itself is diseased, and that is why it takes a long time for electricity to travel from cell to cell and travel from one side of the heart to the other,” he said.
“In the human heart, the QT duration is correlated with an increased risk of abnormal heart rhythms,” he noted. “In our patient, it was concerning that the QT measure was prolonged. While we saw some fluctuations, the QT measure remained prolonged during the whole 61 days.”
‘Interesting study’
Two experts who were not involved with this research weighed in on the findings for this news organization.
“This very interesting study reinforces the difficulties in xenotransplantation, and the need for more research to be able to safely monitor recipients, as baseline values are unknown,” said Edward Vigmond, PhD.
Dr. Vigmond, from the Electrophysiology and Heart Modeling Institute at the University of Bordeaux in France, published a related study about a model of translation of pig to human electrophysiology.
The ECG is sensitive to the electrical activation pattern of the heart, along with the cellular and tissue electrical properties, he noted.
“Although pigs and humans may be similar in size, there are many differences between them,” Dr. Vigmond observed, including “the extent of the rapid conduction system of the heart, the number of nuclei in the muscle cells, the proteins in the cell membrane which control electrical activity, the orientation of the heart and thorax, and the handling of calcium inside the cell.”
“On top of this,” he continued, “donor hearts are denervated, so they no longer respond to nervous modulation, and circulating compounds in the blood which affect heart function vary between species.
“With all these differences, it is not surprising that the ECG of a pig heart transplanted into a human resembles neither that of a human nor that of a pig,” Dr. Vigmond said.
“It is interesting to note that the humanized-gene-edited porcine heart exhibited abnormal electrical conduction parameters from the outset,” said Mandeep R. Mehra, MD.
“Whether these changes were due to the gene modifications (i.e., already inherent in the pig ECG prior to transplant) or a result of the transplant operation challenges (such as the ischemia reperfusion injury and early immunological interactions) is uncertain and should be clarified,” said Dr. Mehra, of Harvard Medical School and Brigham and Women’s Medicine in Boston.
“Knowledge of these changes is important to determine whether a simple ECG parameter may be useful to identify changes that could indicate developing pathology,” Dr. Mehra added.
“In the older days of human transplantation, we often used ECG parameters such as a change in voltage amplitude to identify signals for rejection,” he continued. “Whether such changes occurred in this case could be another interesting aspect to explore as changes occurred in cardiac performance in response to the physiological and pathological challenges that were encountered in this sentinel case.”
The study authors reported having no outside sources of funding.
A version of this article first appeared on Medscape.com.
FROM AHA 2022
Diuretic agents equal to prevent CV events in hypertension: DCP
There was no difference in major cardiovascular outcomes with the use of two different diuretics – chlorthalidone or hydrochlorothiazide – in the treatment of hypertension in a new large randomized real-world study.
The Diuretic Comparison Project (DCP), which was conducted in more than 13,500 U.S. veterans age 65 years or over, showed almost identical rates of the primary composite endpoint, including myocardial infarction (MI), stroke, noncancer death, hospitalization for acute heart failure, or urgent revascularization, after a median of 2.4 years of follow-up.
There was no difference in any of the individual endpoints or other secondary cardiovascular outcomes.
However, in the subgroup of patients who had a history of MI or stroke (who made up about 10% of the study population), there was a significant reduction in the primary endpoint with chlorthalidone, whereas those without a history of MI or stroke appeared to have an increased risk for primary outcome events while receiving chlorthalidone compared with those receiving hydrochlorothiazide.
The DCP trial was presented at the American Heart Association scientific sessions by Areef Ishani, MD, director of the Minneapolis Primary Care and Specialty Care Integrated Care Community and director of the Veterans Affairs (VA) Midwest Health Care Network.
Asked how to interpret the result for clinical practice, Dr. Ishani said, “I think we can now say that either of these two drugs is appropriate to use for the treatment of hypertension.”
But he added that the decision on what to do with the subgroup of patients with previous MI or stroke was more “challenging.”
“We saw a highly significant benefit in this subgroup, but this was in the context of an overall negative trial,” he noted. “I think this is a discussion with the patients on how they want to hedge their bets. Because these two drugs are so similar, if they wanted to take one or the other because of this subgroup result I think that is a conversation to have, but I think we now need to conduct another trial specifically in this subgroup of patients to see if chlorthalidone really is of benefit in that group.”
Dr. Ishani explained that both chlorthalidone and hydrochlorothiazide have been around for more than 50 years and are considered first-line treatments for hypertension. Early studies suggested better cardiovascular outcomes and 24-hour blood pressure control with chlorthalidone, but recent observational studies have not shown more benefit with chlorthalidone. These studies have suggested that chlorthalidone may be associated with an increase in adverse events, such as hypokalemia, acute kidney injury, and chronic kidney disease.
Pragmatic study
The DCP trial was conducted to try to definitively answer this question of whether chlorthalidone is superior to hydrochlorothiazide. The pragmatic study had a “point-of-care” design that allowed participants and health care professionals to know which medication was being prescribed and to administer the medication in a real-world setting.
“Patients can continue with their normal care with their usual care team because we integrated this trial into primary care clinics,” Dr. Ishani said. “We followed participant results using their electronic health record. This study was nonintrusive, cost-effective, and inexpensive. Plus, we were able to recruit a large rural population, which is unusual for large, randomized trials, where we usually rely on big academic medical centers.”
Using VA electronic medical records, the investigators recruited primary care physicians who identified patients older than age 65 years who were receiving hydrochlorothiazide (25 mg or 50 mg) for hypertension. These patients (97% of whom were male) were then randomly assigned to continue receiving hydrochlorothiazide or to switch to an equivalent dose of chlorthalidone. Patients were followed through the electronic medical record as well as Medicare claims and the National Death Index.
Results after a median follow-up of 2.4 years showed no difference in blood pressure control between the two groups.
In terms of clinical events, the primary composite outcome of MI, stroke, noncancer death, hospitalization for acute heart failure, or urgent revascularization occurred in 10.4% of the chlorthalidone group and in 10.0% of the hydrochlorothiazide group (hazard ratio [HR], 1.04; 95% confidence interval [CI], 0.94-1.16; P = .4).
There was no difference in any individual components of the primary endpoint or the secondary outcomes of all-cause mortality, any revascularization, or erectile dysfunction.
In terms of adverse events, chlorthalidone was associated with an increase in hypokalemia (6% vs. 4.4%; HR, 1.38), but there was no difference in hospitalization for acute kidney injury.
Benefit in MI, stroke subgroup?
In the subgroup analysis, patients with a history of MI or stroke who were receiving chlorthalidone had a significant 27% reduction in the primary endpoint (HR, 0.73; 95% CI, 0.57-0.94). Conversely, patients without a history of MI or stroke appeared to do worse while taking chlorthalidone (HR, 1.12; 95% CI, 1.00-1.26).
“We were surprised by these results,” Dr. Ishani said. “We expected chlorthalidone to be more effective overall. However, learning about these differences in patients who have a history of cardiovascular disease may affect patient care. It’s best for people to talk with their health care clinicians about which of these medications is better for their individual needs.”
He added: “More research is needed to explore these results further because we don’t know how they may fit into treating the general population.”
Dr. Ishani noted that a limitations of this study was that most patients were receiving the low dose of chlorthalidone, and previous studies that suggested benefits with chlorthalidone used the higher dose.
“But the world has voted – we had 4,000 clinicians involved in this study, and the vast majority are using the low dose of hydrochlorothiazide. And this is a definitively negative study,” he said. “The world has also voted in that 10 times more patients were on hydrochlorothiazide than on chlorthalidone.”
Commenting on the study at an AHA press conference, Biykem Bozkurt, MD, PhD, Baylor College of Medicine, Houston, pointed out that in all of the landmark National Institutes of Health hypertension trials, there was a signal for benefit with chlorthalidone compared with other antihypertensives.
“We’ve always had this concept that chlorthalidone is better,” she said. “But this study shows no difference in major cardiovascular endpoints. There was more hypokalemia with chlorthalidone, but that’s recognizable as chlorthalidone is a more potent diuretic.”
Other limitations of the DCP trial are its open-label design, which could interject some bias; the enduring effects of hydrochlorothiazide – most of these patients were receiving this agent as background therapy; and inability to look at the effectiveness of decongestion of the agents in such a pragmatic study, Dr. Bozkurt noted.
She said she would like to see more analysis in the subgroup of patients with previous MI or stroke. “Does this result mean that chlorthalidone is better for sicker patients or is this result just due to chance?” she asked.
“While this study demonstrates equal effectiveness of these two diuretics in the targeted population, the question of subgroups of patients for which we use a more potent diuretic I think remains unanswered,” she concluded.
Designated discussant of the DCP trial at the late-breaking trial session, Daniel Levy, MD, director of the Framingham Heart Study at the National Heart, Lung, and Blood Institute, reminded attendees that chlorthalidone had shown impressive results in previous important hypertension studies including SHEP and ALLHAT.
He said the current DCP was a pragmatic study addressing a knowledge gap that “would never have been performed by industry.”
Dr. Levy concluded that the results showing no difference in outcomes between the two diuretics were “compelling,” although a few questions remain.
These include a possible bias toward hydrochlorothiazide – patients were selected who were already taking that drug and so would have already had a favorable response to it. In addition, because the trial was conducted in an older male population, he questioned whether the results could be generalized to women and younger patients.
The DCP study was funded by the VA Cooperative Studies Program. Dr. Ishani reported no disclosures.
A version of this article first appeared on Medscape.com.
There was no difference in major cardiovascular outcomes with the use of two different diuretics – chlorthalidone or hydrochlorothiazide – in the treatment of hypertension in a new large randomized real-world study.
The Diuretic Comparison Project (DCP), which was conducted in more than 13,500 U.S. veterans age 65 years or over, showed almost identical rates of the primary composite endpoint, including myocardial infarction (MI), stroke, noncancer death, hospitalization for acute heart failure, or urgent revascularization, after a median of 2.4 years of follow-up.
There was no difference in any of the individual endpoints or other secondary cardiovascular outcomes.
However, in the subgroup of patients who had a history of MI or stroke (who made up about 10% of the study population), there was a significant reduction in the primary endpoint with chlorthalidone, whereas those without a history of MI or stroke appeared to have an increased risk for primary outcome events while receiving chlorthalidone compared with those receiving hydrochlorothiazide.
The DCP trial was presented at the American Heart Association scientific sessions by Areef Ishani, MD, director of the Minneapolis Primary Care and Specialty Care Integrated Care Community and director of the Veterans Affairs (VA) Midwest Health Care Network.
Asked how to interpret the result for clinical practice, Dr. Ishani said, “I think we can now say that either of these two drugs is appropriate to use for the treatment of hypertension.”
But he added that the decision on what to do with the subgroup of patients with previous MI or stroke was more “challenging.”
“We saw a highly significant benefit in this subgroup, but this was in the context of an overall negative trial,” he noted. “I think this is a discussion with the patients on how they want to hedge their bets. Because these two drugs are so similar, if they wanted to take one or the other because of this subgroup result I think that is a conversation to have, but I think we now need to conduct another trial specifically in this subgroup of patients to see if chlorthalidone really is of benefit in that group.”
Dr. Ishani explained that both chlorthalidone and hydrochlorothiazide have been around for more than 50 years and are considered first-line treatments for hypertension. Early studies suggested better cardiovascular outcomes and 24-hour blood pressure control with chlorthalidone, but recent observational studies have not shown more benefit with chlorthalidone. These studies have suggested that chlorthalidone may be associated with an increase in adverse events, such as hypokalemia, acute kidney injury, and chronic kidney disease.
Pragmatic study
The DCP trial was conducted to try to definitively answer this question of whether chlorthalidone is superior to hydrochlorothiazide. The pragmatic study had a “point-of-care” design that allowed participants and health care professionals to know which medication was being prescribed and to administer the medication in a real-world setting.
“Patients can continue with their normal care with their usual care team because we integrated this trial into primary care clinics,” Dr. Ishani said. “We followed participant results using their electronic health record. This study was nonintrusive, cost-effective, and inexpensive. Plus, we were able to recruit a large rural population, which is unusual for large, randomized trials, where we usually rely on big academic medical centers.”
Using VA electronic medical records, the investigators recruited primary care physicians who identified patients older than age 65 years who were receiving hydrochlorothiazide (25 mg or 50 mg) for hypertension. These patients (97% of whom were male) were then randomly assigned to continue receiving hydrochlorothiazide or to switch to an equivalent dose of chlorthalidone. Patients were followed through the electronic medical record as well as Medicare claims and the National Death Index.
Results after a median follow-up of 2.4 years showed no difference in blood pressure control between the two groups.
In terms of clinical events, the primary composite outcome of MI, stroke, noncancer death, hospitalization for acute heart failure, or urgent revascularization occurred in 10.4% of the chlorthalidone group and in 10.0% of the hydrochlorothiazide group (hazard ratio [HR], 1.04; 95% confidence interval [CI], 0.94-1.16; P = .4).
There was no difference in any individual components of the primary endpoint or the secondary outcomes of all-cause mortality, any revascularization, or erectile dysfunction.
In terms of adverse events, chlorthalidone was associated with an increase in hypokalemia (6% vs. 4.4%; HR, 1.38), but there was no difference in hospitalization for acute kidney injury.
Benefit in MI, stroke subgroup?
In the subgroup analysis, patients with a history of MI or stroke who were receiving chlorthalidone had a significant 27% reduction in the primary endpoint (HR, 0.73; 95% CI, 0.57-0.94). Conversely, patients without a history of MI or stroke appeared to do worse while taking chlorthalidone (HR, 1.12; 95% CI, 1.00-1.26).
“We were surprised by these results,” Dr. Ishani said. “We expected chlorthalidone to be more effective overall. However, learning about these differences in patients who have a history of cardiovascular disease may affect patient care. It’s best for people to talk with their health care clinicians about which of these medications is better for their individual needs.”
He added: “More research is needed to explore these results further because we don’t know how they may fit into treating the general population.”
Dr. Ishani noted that a limitations of this study was that most patients were receiving the low dose of chlorthalidone, and previous studies that suggested benefits with chlorthalidone used the higher dose.
“But the world has voted – we had 4,000 clinicians involved in this study, and the vast majority are using the low dose of hydrochlorothiazide. And this is a definitively negative study,” he said. “The world has also voted in that 10 times more patients were on hydrochlorothiazide than on chlorthalidone.”
Commenting on the study at an AHA press conference, Biykem Bozkurt, MD, PhD, Baylor College of Medicine, Houston, pointed out that in all of the landmark National Institutes of Health hypertension trials, there was a signal for benefit with chlorthalidone compared with other antihypertensives.
“We’ve always had this concept that chlorthalidone is better,” she said. “But this study shows no difference in major cardiovascular endpoints. There was more hypokalemia with chlorthalidone, but that’s recognizable as chlorthalidone is a more potent diuretic.”
Other limitations of the DCP trial are its open-label design, which could interject some bias; the enduring effects of hydrochlorothiazide – most of these patients were receiving this agent as background therapy; and inability to look at the effectiveness of decongestion of the agents in such a pragmatic study, Dr. Bozkurt noted.
She said she would like to see more analysis in the subgroup of patients with previous MI or stroke. “Does this result mean that chlorthalidone is better for sicker patients or is this result just due to chance?” she asked.
“While this study demonstrates equal effectiveness of these two diuretics in the targeted population, the question of subgroups of patients for which we use a more potent diuretic I think remains unanswered,” she concluded.
Designated discussant of the DCP trial at the late-breaking trial session, Daniel Levy, MD, director of the Framingham Heart Study at the National Heart, Lung, and Blood Institute, reminded attendees that chlorthalidone had shown impressive results in previous important hypertension studies including SHEP and ALLHAT.
He said the current DCP was a pragmatic study addressing a knowledge gap that “would never have been performed by industry.”
Dr. Levy concluded that the results showing no difference in outcomes between the two diuretics were “compelling,” although a few questions remain.
These include a possible bias toward hydrochlorothiazide – patients were selected who were already taking that drug and so would have already had a favorable response to it. In addition, because the trial was conducted in an older male population, he questioned whether the results could be generalized to women and younger patients.
The DCP study was funded by the VA Cooperative Studies Program. Dr. Ishani reported no disclosures.
A version of this article first appeared on Medscape.com.
There was no difference in major cardiovascular outcomes with the use of two different diuretics – chlorthalidone or hydrochlorothiazide – in the treatment of hypertension in a new large randomized real-world study.
The Diuretic Comparison Project (DCP), which was conducted in more than 13,500 U.S. veterans age 65 years or over, showed almost identical rates of the primary composite endpoint, including myocardial infarction (MI), stroke, noncancer death, hospitalization for acute heart failure, or urgent revascularization, after a median of 2.4 years of follow-up.
There was no difference in any of the individual endpoints or other secondary cardiovascular outcomes.
However, in the subgroup of patients who had a history of MI or stroke (who made up about 10% of the study population), there was a significant reduction in the primary endpoint with chlorthalidone, whereas those without a history of MI or stroke appeared to have an increased risk for primary outcome events while receiving chlorthalidone compared with those receiving hydrochlorothiazide.
The DCP trial was presented at the American Heart Association scientific sessions by Areef Ishani, MD, director of the Minneapolis Primary Care and Specialty Care Integrated Care Community and director of the Veterans Affairs (VA) Midwest Health Care Network.
Asked how to interpret the result for clinical practice, Dr. Ishani said, “I think we can now say that either of these two drugs is appropriate to use for the treatment of hypertension.”
But he added that the decision on what to do with the subgroup of patients with previous MI or stroke was more “challenging.”
“We saw a highly significant benefit in this subgroup, but this was in the context of an overall negative trial,” he noted. “I think this is a discussion with the patients on how they want to hedge their bets. Because these two drugs are so similar, if they wanted to take one or the other because of this subgroup result I think that is a conversation to have, but I think we now need to conduct another trial specifically in this subgroup of patients to see if chlorthalidone really is of benefit in that group.”
Dr. Ishani explained that both chlorthalidone and hydrochlorothiazide have been around for more than 50 years and are considered first-line treatments for hypertension. Early studies suggested better cardiovascular outcomes and 24-hour blood pressure control with chlorthalidone, but recent observational studies have not shown more benefit with chlorthalidone. These studies have suggested that chlorthalidone may be associated with an increase in adverse events, such as hypokalemia, acute kidney injury, and chronic kidney disease.
Pragmatic study
The DCP trial was conducted to try to definitively answer this question of whether chlorthalidone is superior to hydrochlorothiazide. The pragmatic study had a “point-of-care” design that allowed participants and health care professionals to know which medication was being prescribed and to administer the medication in a real-world setting.
“Patients can continue with their normal care with their usual care team because we integrated this trial into primary care clinics,” Dr. Ishani said. “We followed participant results using their electronic health record. This study was nonintrusive, cost-effective, and inexpensive. Plus, we were able to recruit a large rural population, which is unusual for large, randomized trials, where we usually rely on big academic medical centers.”
Using VA electronic medical records, the investigators recruited primary care physicians who identified patients older than age 65 years who were receiving hydrochlorothiazide (25 mg or 50 mg) for hypertension. These patients (97% of whom were male) were then randomly assigned to continue receiving hydrochlorothiazide or to switch to an equivalent dose of chlorthalidone. Patients were followed through the electronic medical record as well as Medicare claims and the National Death Index.
Results after a median follow-up of 2.4 years showed no difference in blood pressure control between the two groups.
In terms of clinical events, the primary composite outcome of MI, stroke, noncancer death, hospitalization for acute heart failure, or urgent revascularization occurred in 10.4% of the chlorthalidone group and in 10.0% of the hydrochlorothiazide group (hazard ratio [HR], 1.04; 95% confidence interval [CI], 0.94-1.16; P = .4).
There was no difference in any individual components of the primary endpoint or the secondary outcomes of all-cause mortality, any revascularization, or erectile dysfunction.
In terms of adverse events, chlorthalidone was associated with an increase in hypokalemia (6% vs. 4.4%; HR, 1.38), but there was no difference in hospitalization for acute kidney injury.
Benefit in MI, stroke subgroup?
In the subgroup analysis, patients with a history of MI or stroke who were receiving chlorthalidone had a significant 27% reduction in the primary endpoint (HR, 0.73; 95% CI, 0.57-0.94). Conversely, patients without a history of MI or stroke appeared to do worse while taking chlorthalidone (HR, 1.12; 95% CI, 1.00-1.26).
“We were surprised by these results,” Dr. Ishani said. “We expected chlorthalidone to be more effective overall. However, learning about these differences in patients who have a history of cardiovascular disease may affect patient care. It’s best for people to talk with their health care clinicians about which of these medications is better for their individual needs.”
He added: “More research is needed to explore these results further because we don’t know how they may fit into treating the general population.”
Dr. Ishani noted that a limitations of this study was that most patients were receiving the low dose of chlorthalidone, and previous studies that suggested benefits with chlorthalidone used the higher dose.
“But the world has voted – we had 4,000 clinicians involved in this study, and the vast majority are using the low dose of hydrochlorothiazide. And this is a definitively negative study,” he said. “The world has also voted in that 10 times more patients were on hydrochlorothiazide than on chlorthalidone.”
Commenting on the study at an AHA press conference, Biykem Bozkurt, MD, PhD, Baylor College of Medicine, Houston, pointed out that in all of the landmark National Institutes of Health hypertension trials, there was a signal for benefit with chlorthalidone compared with other antihypertensives.
“We’ve always had this concept that chlorthalidone is better,” she said. “But this study shows no difference in major cardiovascular endpoints. There was more hypokalemia with chlorthalidone, but that’s recognizable as chlorthalidone is a more potent diuretic.”
Other limitations of the DCP trial are its open-label design, which could interject some bias; the enduring effects of hydrochlorothiazide – most of these patients were receiving this agent as background therapy; and inability to look at the effectiveness of decongestion of the agents in such a pragmatic study, Dr. Bozkurt noted.
She said she would like to see more analysis in the subgroup of patients with previous MI or stroke. “Does this result mean that chlorthalidone is better for sicker patients or is this result just due to chance?” she asked.
“While this study demonstrates equal effectiveness of these two diuretics in the targeted population, the question of subgroups of patients for which we use a more potent diuretic I think remains unanswered,” she concluded.
Designated discussant of the DCP trial at the late-breaking trial session, Daniel Levy, MD, director of the Framingham Heart Study at the National Heart, Lung, and Blood Institute, reminded attendees that chlorthalidone had shown impressive results in previous important hypertension studies including SHEP and ALLHAT.
He said the current DCP was a pragmatic study addressing a knowledge gap that “would never have been performed by industry.”
Dr. Levy concluded that the results showing no difference in outcomes between the two diuretics were “compelling,” although a few questions remain.
These include a possible bias toward hydrochlorothiazide – patients were selected who were already taking that drug and so would have already had a favorable response to it. In addition, because the trial was conducted in an older male population, he questioned whether the results could be generalized to women and younger patients.
The DCP study was funded by the VA Cooperative Studies Program. Dr. Ishani reported no disclosures.
A version of this article first appeared on Medscape.com.
FROM AHA 2022
Triglyceride-lowering fails to show CV benefit in large fibrate trial
Twenty-five percent reduction has no effect
CHICAGO – Despite a 25% reduction in triglycerides (TGs) along with similar reductions in very-low-density lipoprotein (VLDL), and remnant cholesterol, a novel agent failed to provide any protection in a multinational trial against a composite endpoint of major adverse cardiovascular events (MACE) in patients with type 2 diabetes.
“Our data further highlight the complexity of lipid mediators of residual risk among patients with insulin resistance who are receiving statin therapy,” reported Aruna Das Pradhan, MD, of Harvard Medical School, Boston, and Queen Mary University, London.
It is the most recent in a series of trials that have failed to associate a meaningful reduction in TGs with protection from a composite MACE endpoint. This is a pattern that dates back 20 years, even though earlier trials did suggest that hypertriglyceridemia was a targetable risk factor.
No benefit from fibrates seen in statin era
“We have not seen a significant cardiovascular event reduction with a fibrate in the statin era,” according to Karol Watson, MD, PhD, director of the UCLA Women’s Cardiovascular Health Center, Los Angeles.
In the statin era, which began soon after the Helsinki Heart Study was published in 1987, Dr. Watson counted at least five studies with fibrates that had a null result.
In the setting of good control of LDL cholesterol, “fibrates have not been shown to further lower CV risk,” said Dr. Watson, who was invited by the AHA to discuss the PROMINENT trial.
In PROMINENT, 10,497 patients with type 2 diabetes were randomized to pemafibrate, a peroxisome proliferator-activated receptor a (PPAR-a) agonist, or placebo. Pemafibrate is not currently available in North America or Europe, but it is licensed in Japan for the treatment of hypertriglyceridemia.
The primary efficacy endpoint of the double-blind trial was a composite endpoint of nonfatal myocardial infarction, ischemic stroke, coronary revascularization, or death.
The patients were eligible if they had TG levels from 200 to 400 mg/dL and HDL cholesterol levels of 40 mg/dL or below. Pemafibrate in a dose of 0.2 mg or placebo were taken twice daily. About two-thirds had a prior history of coronary heart disease. The goal was primary prevention in the remainder.
After a median follow-up of 3.4 years when the study was stopped for futility, the proportion of patients reaching a primary endpoint was slightly greater in the experimental arm (3.60 vs. 3.51 events per 100 patient-years). The hazard ratio, although not significant, was nominally in favor of placebo (hazard ratio, 1.03; P = .67).
When events within the composite endpoint were assessed individually, there was no signal of benefit for any outcome. The rates of death from any cause, although numerically higher in the pemafibrate group (2.44 vs. 2.34 per 100 patient years), were also comparable.
Lipid profile improved as predicted
Yet, in regard to an improvement in the lipid profile, pemafibrate performed as predicted. When compared to placebo 4 months into the trial, pemafibrate was associated with median reductions of 26.2% in TGs, 25.8% in VLDL, and 25.6% in remnant cholesterol, which is cholesterol transported in TG-rich lipoproteins after lipolysis and lipoprotein remodeling.
Furthermore, pemafibrate was associated with a median 27.6% reduction relative to placebo in apolipoprotein C-III and a median 4.8% reduction in apolipoprotein E, all of which would be expected to reduce CV risk.
The findings of PROMINENT were published online in the New England Journal of Medicine immediately after their presentation.
The findings of this study do not eliminate any hope for lowering residual CV risk with TG reductions, but they do suggest the relationship with other lipid subfractions is complex, according to Salim S. Virani, MD, PhD, a professor of cardiology at Baylor College of Medicine, Houston.
“I think that the lack of efficacy despite TG lowering may be largely due to a lack of an overall decrease in the apolipoprotein B level,” speculated Dr. Virani, who wrote an editorial that accompanied publication of the PROMINENT results.
He noted that pemafibrate is implicated in converting remnant cholesterol to LDL cholesterol, which might be one reason for a counterproductive effect on CV risk.
“In order for therapies that lower TG levels to be effective, they probably have to have mechanisms to increase clearance of TG-rich remnant lipoprotein cholesterol particles rather than just converting remnant lipoproteins to LDL,” Dr. Virani explained in an attempt to unravel the interplay of these variables.
Although this study enrolled patients “who would be predicted to have the most benefit from a TG-lowering strategy,” Dr. Watson agreed that these results do not necessarily extend to other means of lowering TG. However, it might draw into question the value of pemafibrate and perhaps other drugs in this class for treatment of hypertriglyceridemia. In addition to a lack of CV benefit, treatment was not without risks, including a higher rate of thromboembolism and adverse renal events.
Dr. Das Pradhan reported financial relationships with Denka, Medtelligence, Optum, Novo Nordisk, and Kowa, which provided funding for this trial. Dr. Watson reported financial relationships with Amarin, Amgen, Boehringer-Ingelheim, and Esperion.
Twenty-five percent reduction has no effect
Twenty-five percent reduction has no effect
CHICAGO – Despite a 25% reduction in triglycerides (TGs) along with similar reductions in very-low-density lipoprotein (VLDL), and remnant cholesterol, a novel agent failed to provide any protection in a multinational trial against a composite endpoint of major adverse cardiovascular events (MACE) in patients with type 2 diabetes.
“Our data further highlight the complexity of lipid mediators of residual risk among patients with insulin resistance who are receiving statin therapy,” reported Aruna Das Pradhan, MD, of Harvard Medical School, Boston, and Queen Mary University, London.
It is the most recent in a series of trials that have failed to associate a meaningful reduction in TGs with protection from a composite MACE endpoint. This is a pattern that dates back 20 years, even though earlier trials did suggest that hypertriglyceridemia was a targetable risk factor.
No benefit from fibrates seen in statin era
“We have not seen a significant cardiovascular event reduction with a fibrate in the statin era,” according to Karol Watson, MD, PhD, director of the UCLA Women’s Cardiovascular Health Center, Los Angeles.
In the statin era, which began soon after the Helsinki Heart Study was published in 1987, Dr. Watson counted at least five studies with fibrates that had a null result.
In the setting of good control of LDL cholesterol, “fibrates have not been shown to further lower CV risk,” said Dr. Watson, who was invited by the AHA to discuss the PROMINENT trial.
In PROMINENT, 10,497 patients with type 2 diabetes were randomized to pemafibrate, a peroxisome proliferator-activated receptor a (PPAR-a) agonist, or placebo. Pemafibrate is not currently available in North America or Europe, but it is licensed in Japan for the treatment of hypertriglyceridemia.
The primary efficacy endpoint of the double-blind trial was a composite endpoint of nonfatal myocardial infarction, ischemic stroke, coronary revascularization, or death.
The patients were eligible if they had TG levels from 200 to 400 mg/dL and HDL cholesterol levels of 40 mg/dL or below. Pemafibrate in a dose of 0.2 mg or placebo were taken twice daily. About two-thirds had a prior history of coronary heart disease. The goal was primary prevention in the remainder.
After a median follow-up of 3.4 years when the study was stopped for futility, the proportion of patients reaching a primary endpoint was slightly greater in the experimental arm (3.60 vs. 3.51 events per 100 patient-years). The hazard ratio, although not significant, was nominally in favor of placebo (hazard ratio, 1.03; P = .67).
When events within the composite endpoint were assessed individually, there was no signal of benefit for any outcome. The rates of death from any cause, although numerically higher in the pemafibrate group (2.44 vs. 2.34 per 100 patient years), were also comparable.
Lipid profile improved as predicted
Yet, in regard to an improvement in the lipid profile, pemafibrate performed as predicted. When compared to placebo 4 months into the trial, pemafibrate was associated with median reductions of 26.2% in TGs, 25.8% in VLDL, and 25.6% in remnant cholesterol, which is cholesterol transported in TG-rich lipoproteins after lipolysis and lipoprotein remodeling.
Furthermore, pemafibrate was associated with a median 27.6% reduction relative to placebo in apolipoprotein C-III and a median 4.8% reduction in apolipoprotein E, all of which would be expected to reduce CV risk.
The findings of PROMINENT were published online in the New England Journal of Medicine immediately after their presentation.
The findings of this study do not eliminate any hope for lowering residual CV risk with TG reductions, but they do suggest the relationship with other lipid subfractions is complex, according to Salim S. Virani, MD, PhD, a professor of cardiology at Baylor College of Medicine, Houston.
“I think that the lack of efficacy despite TG lowering may be largely due to a lack of an overall decrease in the apolipoprotein B level,” speculated Dr. Virani, who wrote an editorial that accompanied publication of the PROMINENT results.
He noted that pemafibrate is implicated in converting remnant cholesterol to LDL cholesterol, which might be one reason for a counterproductive effect on CV risk.
“In order for therapies that lower TG levels to be effective, they probably have to have mechanisms to increase clearance of TG-rich remnant lipoprotein cholesterol particles rather than just converting remnant lipoproteins to LDL,” Dr. Virani explained in an attempt to unravel the interplay of these variables.
Although this study enrolled patients “who would be predicted to have the most benefit from a TG-lowering strategy,” Dr. Watson agreed that these results do not necessarily extend to other means of lowering TG. However, it might draw into question the value of pemafibrate and perhaps other drugs in this class for treatment of hypertriglyceridemia. In addition to a lack of CV benefit, treatment was not without risks, including a higher rate of thromboembolism and adverse renal events.
Dr. Das Pradhan reported financial relationships with Denka, Medtelligence, Optum, Novo Nordisk, and Kowa, which provided funding for this trial. Dr. Watson reported financial relationships with Amarin, Amgen, Boehringer-Ingelheim, and Esperion.
CHICAGO – Despite a 25% reduction in triglycerides (TGs) along with similar reductions in very-low-density lipoprotein (VLDL), and remnant cholesterol, a novel agent failed to provide any protection in a multinational trial against a composite endpoint of major adverse cardiovascular events (MACE) in patients with type 2 diabetes.
“Our data further highlight the complexity of lipid mediators of residual risk among patients with insulin resistance who are receiving statin therapy,” reported Aruna Das Pradhan, MD, of Harvard Medical School, Boston, and Queen Mary University, London.
It is the most recent in a series of trials that have failed to associate a meaningful reduction in TGs with protection from a composite MACE endpoint. This is a pattern that dates back 20 years, even though earlier trials did suggest that hypertriglyceridemia was a targetable risk factor.
No benefit from fibrates seen in statin era
“We have not seen a significant cardiovascular event reduction with a fibrate in the statin era,” according to Karol Watson, MD, PhD, director of the UCLA Women’s Cardiovascular Health Center, Los Angeles.
In the statin era, which began soon after the Helsinki Heart Study was published in 1987, Dr. Watson counted at least five studies with fibrates that had a null result.
In the setting of good control of LDL cholesterol, “fibrates have not been shown to further lower CV risk,” said Dr. Watson, who was invited by the AHA to discuss the PROMINENT trial.
In PROMINENT, 10,497 patients with type 2 diabetes were randomized to pemafibrate, a peroxisome proliferator-activated receptor a (PPAR-a) agonist, or placebo. Pemafibrate is not currently available in North America or Europe, but it is licensed in Japan for the treatment of hypertriglyceridemia.
The primary efficacy endpoint of the double-blind trial was a composite endpoint of nonfatal myocardial infarction, ischemic stroke, coronary revascularization, or death.
The patients were eligible if they had TG levels from 200 to 400 mg/dL and HDL cholesterol levels of 40 mg/dL or below. Pemafibrate in a dose of 0.2 mg or placebo were taken twice daily. About two-thirds had a prior history of coronary heart disease. The goal was primary prevention in the remainder.
After a median follow-up of 3.4 years when the study was stopped for futility, the proportion of patients reaching a primary endpoint was slightly greater in the experimental arm (3.60 vs. 3.51 events per 100 patient-years). The hazard ratio, although not significant, was nominally in favor of placebo (hazard ratio, 1.03; P = .67).
When events within the composite endpoint were assessed individually, there was no signal of benefit for any outcome. The rates of death from any cause, although numerically higher in the pemafibrate group (2.44 vs. 2.34 per 100 patient years), were also comparable.
Lipid profile improved as predicted
Yet, in regard to an improvement in the lipid profile, pemafibrate performed as predicted. When compared to placebo 4 months into the trial, pemafibrate was associated with median reductions of 26.2% in TGs, 25.8% in VLDL, and 25.6% in remnant cholesterol, which is cholesterol transported in TG-rich lipoproteins after lipolysis and lipoprotein remodeling.
Furthermore, pemafibrate was associated with a median 27.6% reduction relative to placebo in apolipoprotein C-III and a median 4.8% reduction in apolipoprotein E, all of which would be expected to reduce CV risk.
The findings of PROMINENT were published online in the New England Journal of Medicine immediately after their presentation.
The findings of this study do not eliminate any hope for lowering residual CV risk with TG reductions, but they do suggest the relationship with other lipid subfractions is complex, according to Salim S. Virani, MD, PhD, a professor of cardiology at Baylor College of Medicine, Houston.
“I think that the lack of efficacy despite TG lowering may be largely due to a lack of an overall decrease in the apolipoprotein B level,” speculated Dr. Virani, who wrote an editorial that accompanied publication of the PROMINENT results.
He noted that pemafibrate is implicated in converting remnant cholesterol to LDL cholesterol, which might be one reason for a counterproductive effect on CV risk.
“In order for therapies that lower TG levels to be effective, they probably have to have mechanisms to increase clearance of TG-rich remnant lipoprotein cholesterol particles rather than just converting remnant lipoproteins to LDL,” Dr. Virani explained in an attempt to unravel the interplay of these variables.
Although this study enrolled patients “who would be predicted to have the most benefit from a TG-lowering strategy,” Dr. Watson agreed that these results do not necessarily extend to other means of lowering TG. However, it might draw into question the value of pemafibrate and perhaps other drugs in this class for treatment of hypertriglyceridemia. In addition to a lack of CV benefit, treatment was not without risks, including a higher rate of thromboembolism and adverse renal events.
Dr. Das Pradhan reported financial relationships with Denka, Medtelligence, Optum, Novo Nordisk, and Kowa, which provided funding for this trial. Dr. Watson reported financial relationships with Amarin, Amgen, Boehringer-Ingelheim, and Esperion.
AT AHA 2022
Pulmonologist consult at COPD admission reduces risk of return
according to a retrospective cohort review presented at the annual meeting of the American College of Chest Physicians (CHEST).
“When stratified by severity of COPD at the time of admission, the difference in the readmission rate was even greater,” reported Nakisa Hekmat-Joo, MD, a third-year resident at Staten Island University Hospital, New York.
Just as protocols have been developed for prompt initiation of antibiotics in patients with septicemia or prompt revascularization in patients with ST-segment elevated myocardial infarction (STEMI), Dr. Hekmat-Joo said the data from this study warrant a larger trial to evaluate whether an AECOPD admission protocol is warranted to improve outcomes and lower costs.
In this study, all AECOPD admissions were included from a recent 2-year period at two Staten Island hospitals. Of these, 198 patients received a pulmonologist consult within 24 hours. The remaining 92 patients were not evaluated by pulmonologists but were admitted and then managed by residents, internists, or others.
The primary outcome was length of stay (LOS). Although the slightly lower LOS in pulmonologist-treated group did not approach significance (4.16 vs. 4.21 days; P = .88), the readmission rate at 90 days, which was a secondary outcome, was reduced by almost half (30.1% vs. 57.6%; P < .0001).
At admission, there was no significant difference between those receiving a pulmonologist consult and those who did not. The average O2 saturation was lower in the group seen by a pulmonologist (93% vs. 95.4%; P < .0001), but the most striking difference was the low relative readmission rate, which remained significant after controlling for severity and pulmonary function.
“When we stratified patients for baseline severity, the advantage of a pulmonologist consult was even greater for those with the most severe disease,” Dr. Hekmat-Joo said. Among those with the greatest severity, the 90-day readmission rate was nearly three times greater in the absence of a pulmonologist consult (72% vs. 28%).
Although the comparison of outcomes for those receiving a pulmonologist consult vs. those who did not was adjusted for COPD severity, the potential for pulmonologist consults to be ordered for those patients who looked the sickest would have likely worked against the study result.
“We speculate that pulmonologists were more likely than internists to treat beyond standard guidelines, particularly in the event of greater severity,” Dr. Hekmat-Joo explained. These steps might include earlier use of noninvasive positive pressure ventilation or earlier initiation of rehabilitation strategies.
There were several signals that a pulmonologist consult led to more rigorous care.
“The average time to follow-up after hospitalization was 23 days for the pulmonologist group and 66 days for the nonpulmonologist group,” said Dr. Hekmat-Joo, noting this difference was highly significant (P = .0052).
Based on these results, Dr. Hekmat-Joo and her co-investigators are now working on a protocol for COPD admissions that involves a pulmonologist consult within 24 hours of admission. She hopes to test this protocol in a prospective trial.
“COPD remains a major cause of death and consumes enormous health care resources. About 30% of the cost of COPD care is due to readmissions,” she said, noting that readmissions adversely impact quality of life.
Asked if there was sufficient staff at her institution to allow for a pulmonologist consult with every COPD admission, Dr. Hekmat-Joo acknowledged that this has to be demonstrated, but compelling evidence of a benefit might prompt a redistribution of resources.
“If we can show that readmissions are substantially reduced, adding staff to perform these consults would be a good investment,” said Dr. Hekmat-Joo, indicating that improved outcomes could also attract the attention of third-party payers and those tracking quality-of-care metrics.
There is a strong rationale for a randomized prospective trial to confirm the value of a pulmonologist consultation following admission for an acute exacerbation of COPD, according to Nicola A. Hanania, MD, director, Airways Clinical Research Center, Baylor College of Medicine, Houston.
The potential for benefit as seen in this retrospective study is a rational expectation and might be related to more appropriate therapy upon discharge as well as to earlier and more rigorous follow-up, according to Dr. Hanania. Although he cautioned that there is a meaningful risk of selection bias in a retrospective study, he thinks this study “is certainly probing an important issue.”
“Mortality from a hospitalized COPD exacerbation exceeds that of a myocardial infarction,” Dr. Hanania pointed out. Noting that all patients with an MI are evaluated by a cardiologist, he sees the logic of a pulmonologist consult – although he acknowledged that evidence is needed.
“I strongly believe that a prospective study is feasible and will answer the question in an unbiased manner if done properly,” he said in an interview. If a multicenter, well-controlled study was positive, it could change practice.
In the event of a study showing major clinical benefits, particularly a reduction in mortality, “I believe it is feasible to have a pulmonary consult to see every COPD exacerbation patient admitted to the hospital,” Dr. Hanania said.
Dr. Hekmat-Joo reports no relevant financial relationships. Dr. Hanania has financial relationships with AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Mylan, Novartis, Regeneron, Sanofi, and Sunovion.
A version of this article first appeared on Medscape.com.
according to a retrospective cohort review presented at the annual meeting of the American College of Chest Physicians (CHEST).
“When stratified by severity of COPD at the time of admission, the difference in the readmission rate was even greater,” reported Nakisa Hekmat-Joo, MD, a third-year resident at Staten Island University Hospital, New York.
Just as protocols have been developed for prompt initiation of antibiotics in patients with septicemia or prompt revascularization in patients with ST-segment elevated myocardial infarction (STEMI), Dr. Hekmat-Joo said the data from this study warrant a larger trial to evaluate whether an AECOPD admission protocol is warranted to improve outcomes and lower costs.
In this study, all AECOPD admissions were included from a recent 2-year period at two Staten Island hospitals. Of these, 198 patients received a pulmonologist consult within 24 hours. The remaining 92 patients were not evaluated by pulmonologists but were admitted and then managed by residents, internists, or others.
The primary outcome was length of stay (LOS). Although the slightly lower LOS in pulmonologist-treated group did not approach significance (4.16 vs. 4.21 days; P = .88), the readmission rate at 90 days, which was a secondary outcome, was reduced by almost half (30.1% vs. 57.6%; P < .0001).
At admission, there was no significant difference between those receiving a pulmonologist consult and those who did not. The average O2 saturation was lower in the group seen by a pulmonologist (93% vs. 95.4%; P < .0001), but the most striking difference was the low relative readmission rate, which remained significant after controlling for severity and pulmonary function.
“When we stratified patients for baseline severity, the advantage of a pulmonologist consult was even greater for those with the most severe disease,” Dr. Hekmat-Joo said. Among those with the greatest severity, the 90-day readmission rate was nearly three times greater in the absence of a pulmonologist consult (72% vs. 28%).
Although the comparison of outcomes for those receiving a pulmonologist consult vs. those who did not was adjusted for COPD severity, the potential for pulmonologist consults to be ordered for those patients who looked the sickest would have likely worked against the study result.
“We speculate that pulmonologists were more likely than internists to treat beyond standard guidelines, particularly in the event of greater severity,” Dr. Hekmat-Joo explained. These steps might include earlier use of noninvasive positive pressure ventilation or earlier initiation of rehabilitation strategies.
There were several signals that a pulmonologist consult led to more rigorous care.
“The average time to follow-up after hospitalization was 23 days for the pulmonologist group and 66 days for the nonpulmonologist group,” said Dr. Hekmat-Joo, noting this difference was highly significant (P = .0052).
Based on these results, Dr. Hekmat-Joo and her co-investigators are now working on a protocol for COPD admissions that involves a pulmonologist consult within 24 hours of admission. She hopes to test this protocol in a prospective trial.
“COPD remains a major cause of death and consumes enormous health care resources. About 30% of the cost of COPD care is due to readmissions,” she said, noting that readmissions adversely impact quality of life.
Asked if there was sufficient staff at her institution to allow for a pulmonologist consult with every COPD admission, Dr. Hekmat-Joo acknowledged that this has to be demonstrated, but compelling evidence of a benefit might prompt a redistribution of resources.
“If we can show that readmissions are substantially reduced, adding staff to perform these consults would be a good investment,” said Dr. Hekmat-Joo, indicating that improved outcomes could also attract the attention of third-party payers and those tracking quality-of-care metrics.
There is a strong rationale for a randomized prospective trial to confirm the value of a pulmonologist consultation following admission for an acute exacerbation of COPD, according to Nicola A. Hanania, MD, director, Airways Clinical Research Center, Baylor College of Medicine, Houston.
The potential for benefit as seen in this retrospective study is a rational expectation and might be related to more appropriate therapy upon discharge as well as to earlier and more rigorous follow-up, according to Dr. Hanania. Although he cautioned that there is a meaningful risk of selection bias in a retrospective study, he thinks this study “is certainly probing an important issue.”
“Mortality from a hospitalized COPD exacerbation exceeds that of a myocardial infarction,” Dr. Hanania pointed out. Noting that all patients with an MI are evaluated by a cardiologist, he sees the logic of a pulmonologist consult – although he acknowledged that evidence is needed.
“I strongly believe that a prospective study is feasible and will answer the question in an unbiased manner if done properly,” he said in an interview. If a multicenter, well-controlled study was positive, it could change practice.
In the event of a study showing major clinical benefits, particularly a reduction in mortality, “I believe it is feasible to have a pulmonary consult to see every COPD exacerbation patient admitted to the hospital,” Dr. Hanania said.
Dr. Hekmat-Joo reports no relevant financial relationships. Dr. Hanania has financial relationships with AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Mylan, Novartis, Regeneron, Sanofi, and Sunovion.
A version of this article first appeared on Medscape.com.
according to a retrospective cohort review presented at the annual meeting of the American College of Chest Physicians (CHEST).
“When stratified by severity of COPD at the time of admission, the difference in the readmission rate was even greater,” reported Nakisa Hekmat-Joo, MD, a third-year resident at Staten Island University Hospital, New York.
Just as protocols have been developed for prompt initiation of antibiotics in patients with septicemia or prompt revascularization in patients with ST-segment elevated myocardial infarction (STEMI), Dr. Hekmat-Joo said the data from this study warrant a larger trial to evaluate whether an AECOPD admission protocol is warranted to improve outcomes and lower costs.
In this study, all AECOPD admissions were included from a recent 2-year period at two Staten Island hospitals. Of these, 198 patients received a pulmonologist consult within 24 hours. The remaining 92 patients were not evaluated by pulmonologists but were admitted and then managed by residents, internists, or others.
The primary outcome was length of stay (LOS). Although the slightly lower LOS in pulmonologist-treated group did not approach significance (4.16 vs. 4.21 days; P = .88), the readmission rate at 90 days, which was a secondary outcome, was reduced by almost half (30.1% vs. 57.6%; P < .0001).
At admission, there was no significant difference between those receiving a pulmonologist consult and those who did not. The average O2 saturation was lower in the group seen by a pulmonologist (93% vs. 95.4%; P < .0001), but the most striking difference was the low relative readmission rate, which remained significant after controlling for severity and pulmonary function.
“When we stratified patients for baseline severity, the advantage of a pulmonologist consult was even greater for those with the most severe disease,” Dr. Hekmat-Joo said. Among those with the greatest severity, the 90-day readmission rate was nearly three times greater in the absence of a pulmonologist consult (72% vs. 28%).
Although the comparison of outcomes for those receiving a pulmonologist consult vs. those who did not was adjusted for COPD severity, the potential for pulmonologist consults to be ordered for those patients who looked the sickest would have likely worked against the study result.
“We speculate that pulmonologists were more likely than internists to treat beyond standard guidelines, particularly in the event of greater severity,” Dr. Hekmat-Joo explained. These steps might include earlier use of noninvasive positive pressure ventilation or earlier initiation of rehabilitation strategies.
There were several signals that a pulmonologist consult led to more rigorous care.
“The average time to follow-up after hospitalization was 23 days for the pulmonologist group and 66 days for the nonpulmonologist group,” said Dr. Hekmat-Joo, noting this difference was highly significant (P = .0052).
Based on these results, Dr. Hekmat-Joo and her co-investigators are now working on a protocol for COPD admissions that involves a pulmonologist consult within 24 hours of admission. She hopes to test this protocol in a prospective trial.
“COPD remains a major cause of death and consumes enormous health care resources. About 30% of the cost of COPD care is due to readmissions,” she said, noting that readmissions adversely impact quality of life.
Asked if there was sufficient staff at her institution to allow for a pulmonologist consult with every COPD admission, Dr. Hekmat-Joo acknowledged that this has to be demonstrated, but compelling evidence of a benefit might prompt a redistribution of resources.
“If we can show that readmissions are substantially reduced, adding staff to perform these consults would be a good investment,” said Dr. Hekmat-Joo, indicating that improved outcomes could also attract the attention of third-party payers and those tracking quality-of-care metrics.
There is a strong rationale for a randomized prospective trial to confirm the value of a pulmonologist consultation following admission for an acute exacerbation of COPD, according to Nicola A. Hanania, MD, director, Airways Clinical Research Center, Baylor College of Medicine, Houston.
The potential for benefit as seen in this retrospective study is a rational expectation and might be related to more appropriate therapy upon discharge as well as to earlier and more rigorous follow-up, according to Dr. Hanania. Although he cautioned that there is a meaningful risk of selection bias in a retrospective study, he thinks this study “is certainly probing an important issue.”
“Mortality from a hospitalized COPD exacerbation exceeds that of a myocardial infarction,” Dr. Hanania pointed out. Noting that all patients with an MI are evaluated by a cardiologist, he sees the logic of a pulmonologist consult – although he acknowledged that evidence is needed.
“I strongly believe that a prospective study is feasible and will answer the question in an unbiased manner if done properly,” he said in an interview. If a multicenter, well-controlled study was positive, it could change practice.
In the event of a study showing major clinical benefits, particularly a reduction in mortality, “I believe it is feasible to have a pulmonary consult to see every COPD exacerbation patient admitted to the hospital,” Dr. Hanania said.
Dr. Hekmat-Joo reports no relevant financial relationships. Dr. Hanania has financial relationships with AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Mylan, Novartis, Regeneron, Sanofi, and Sunovion.
A version of this article first appeared on Medscape.com.
FROM CHEST 2022
Working while sick: Why doctors don’t stay home when ill
The reasons are likely as varied as, “you weren’t feeling bad enough to miss work,” “you couldn’t afford to miss pay,” “you had too many patients to see,” or “too much work to do.”
In Medscape’s Employed Physicians Report: Loving the Focus, Hating the Bureaucracy, 61% of physicians reported that they sometimes or often come to work sick. Only 2% of respondents said they never come to work unwell.
Medscape wanted to know more about how often you call in sick, how often you come to work feeling unwell, what symptoms you have, and the dogma of your workplace culture regarding sick days. Not to mention the brutal ethos that starts in medical school, in which calling in sick shows weakness or is unacceptable.
So, we polled 2,347 physicians in the United States and abroad and asked them about their sniffling, sneezing, cold, flu, and fever symptoms, and, of course, COVID. Results were split about 50-50 among male and female physicians. The poll ran from Sept. 28 through Oct. 11.
Coming to work sick
It’s no surprise that the majority of physicians who were polled (85%) have come to work sick in 2022. In the last prepandemic year (2019), about 70% came to work feeling sick one to five times, and 13% worked while sick six to ten times.
When asked about the symptoms that they’ve previously come to work with, 48% of U.S. physicians said multiple symptoms. They gave high marks for runny nose, cough, congestion, and sore throat. Only 27% have worked with a fever, 22% have worked with other symptoms, and 7% have worked with both strep throat and COVID.
“My workplace, especially in the COVID years, accommodates persons who honestly do not feel well enough to report. Sooner or later, everyone covers for someone else who has to be out,” says Kenneth Abbott, MD, an oncologist in Maryland.
The culture of working while sick
Why doctors come to work when they’re sick is complicated. The overwhelming majority of U.S. respondents cited professional obligations; 73% noted that they feel a professional obligation to their patients, and 72% feel a professional obligation to their co-workers. Half of the polled U.S. physicians said they didn’t feel bad enough to stay home, while 48% said they had too much work to do to stay home.
Some 45% said the expectation at their workplace is to come to work unless seriously ill; 43% had too many patients to see; and 18% didn’t think they were contagious when they headed to work sick. Unfortunately, 15% chose to work while sick because otherwise they would lose pay.
In light of these responses, it’s not surprising that 93% reported they’d seen other medical professionals working when sick.
“My schedule is almost always booked weeks in advance. If someone misses or has to cancel their appointment, they typically have 2-4 weeks to wait to get back in. If I was sick and a full day of patients (or God forbid more than a day) had to be canceled because I called in, it’s so much more work when I return,” says Caitlin Briggs, MD, a psychiatrist in Lexington, Ky.
Doctors’ workplace sick day policy
Most employees’ benefits allow at least a few sick days, but doctors who treat society’s ill patients don’t seem to stay home from work when they’re suffering. So, we asked physicians, official policy aside, whether they thought going to work sick was expected in their workplace. The majority (76%) said yes, while 24% said no.
“Unless I’m dying or extremely contagious, I usually work. At least now, I have the telehealth option. Not saying any of this is right, but it’s the reality we deal with and the choice we must make,” says Dr. Briggs.
Additionally, 58% of polled physicians said their workplace did not have a clearly defined policy against coming to work sick, while 20% said theirs did, and 22% weren’t sure.
“The first thing I heard on the subject as a medical student was that sick people come to the hospital, so if you’re sick, then you come to the hospital too ... to work. If you can’t work, then you will be admitted. Another aphorism was from Churchill, that ‘most of the world’s work is done by people who don’t feel very well,’ ” says Paul Andreason, MD, a psychiatrist in Bethesda, Md.
Working in the time of COVID
Working while ill during ordinary times is one thing, but what about working in the time of COVID? Has the pandemic changed the culture of coming to work sick because medical facilities, such as doctor’s offices and hospitals, don’t want their staff coming in when they have COVID?
Surprisingly, when we asked physicians whether the pandemic has made it more or less acceptable to come to work sick, only 61% thought COVID has made it less acceptable to work while sick, while 16% thought it made it more acceptable, and 23% said there’s no change.
“I draw the line at fevers/chills, feeling like you’ve just been run over, or significant enteritis,” says Dr. Abbott. “Also, if I have to take palliative meds that interfere with alertness, I’m not doing my patients any favors.”
While a minority of physicians may call in sick, most still suffer through their sneezing, coughing, chills, and fever while seeing patients as usual.
A version of this article first appeared on Medscape.com.
The reasons are likely as varied as, “you weren’t feeling bad enough to miss work,” “you couldn’t afford to miss pay,” “you had too many patients to see,” or “too much work to do.”
In Medscape’s Employed Physicians Report: Loving the Focus, Hating the Bureaucracy, 61% of physicians reported that they sometimes or often come to work sick. Only 2% of respondents said they never come to work unwell.
Medscape wanted to know more about how often you call in sick, how often you come to work feeling unwell, what symptoms you have, and the dogma of your workplace culture regarding sick days. Not to mention the brutal ethos that starts in medical school, in which calling in sick shows weakness or is unacceptable.
So, we polled 2,347 physicians in the United States and abroad and asked them about their sniffling, sneezing, cold, flu, and fever symptoms, and, of course, COVID. Results were split about 50-50 among male and female physicians. The poll ran from Sept. 28 through Oct. 11.
Coming to work sick
It’s no surprise that the majority of physicians who were polled (85%) have come to work sick in 2022. In the last prepandemic year (2019), about 70% came to work feeling sick one to five times, and 13% worked while sick six to ten times.
When asked about the symptoms that they’ve previously come to work with, 48% of U.S. physicians said multiple symptoms. They gave high marks for runny nose, cough, congestion, and sore throat. Only 27% have worked with a fever, 22% have worked with other symptoms, and 7% have worked with both strep throat and COVID.
“My workplace, especially in the COVID years, accommodates persons who honestly do not feel well enough to report. Sooner or later, everyone covers for someone else who has to be out,” says Kenneth Abbott, MD, an oncologist in Maryland.
The culture of working while sick
Why doctors come to work when they’re sick is complicated. The overwhelming majority of U.S. respondents cited professional obligations; 73% noted that they feel a professional obligation to their patients, and 72% feel a professional obligation to their co-workers. Half of the polled U.S. physicians said they didn’t feel bad enough to stay home, while 48% said they had too much work to do to stay home.
Some 45% said the expectation at their workplace is to come to work unless seriously ill; 43% had too many patients to see; and 18% didn’t think they were contagious when they headed to work sick. Unfortunately, 15% chose to work while sick because otherwise they would lose pay.
In light of these responses, it’s not surprising that 93% reported they’d seen other medical professionals working when sick.
“My schedule is almost always booked weeks in advance. If someone misses or has to cancel their appointment, they typically have 2-4 weeks to wait to get back in. If I was sick and a full day of patients (or God forbid more than a day) had to be canceled because I called in, it’s so much more work when I return,” says Caitlin Briggs, MD, a psychiatrist in Lexington, Ky.
Doctors’ workplace sick day policy
Most employees’ benefits allow at least a few sick days, but doctors who treat society’s ill patients don’t seem to stay home from work when they’re suffering. So, we asked physicians, official policy aside, whether they thought going to work sick was expected in their workplace. The majority (76%) said yes, while 24% said no.
“Unless I’m dying or extremely contagious, I usually work. At least now, I have the telehealth option. Not saying any of this is right, but it’s the reality we deal with and the choice we must make,” says Dr. Briggs.
Additionally, 58% of polled physicians said their workplace did not have a clearly defined policy against coming to work sick, while 20% said theirs did, and 22% weren’t sure.
“The first thing I heard on the subject as a medical student was that sick people come to the hospital, so if you’re sick, then you come to the hospital too ... to work. If you can’t work, then you will be admitted. Another aphorism was from Churchill, that ‘most of the world’s work is done by people who don’t feel very well,’ ” says Paul Andreason, MD, a psychiatrist in Bethesda, Md.
Working in the time of COVID
Working while ill during ordinary times is one thing, but what about working in the time of COVID? Has the pandemic changed the culture of coming to work sick because medical facilities, such as doctor’s offices and hospitals, don’t want their staff coming in when they have COVID?
Surprisingly, when we asked physicians whether the pandemic has made it more or less acceptable to come to work sick, only 61% thought COVID has made it less acceptable to work while sick, while 16% thought it made it more acceptable, and 23% said there’s no change.
“I draw the line at fevers/chills, feeling like you’ve just been run over, or significant enteritis,” says Dr. Abbott. “Also, if I have to take palliative meds that interfere with alertness, I’m not doing my patients any favors.”
While a minority of physicians may call in sick, most still suffer through their sneezing, coughing, chills, and fever while seeing patients as usual.
A version of this article first appeared on Medscape.com.
The reasons are likely as varied as, “you weren’t feeling bad enough to miss work,” “you couldn’t afford to miss pay,” “you had too many patients to see,” or “too much work to do.”
In Medscape’s Employed Physicians Report: Loving the Focus, Hating the Bureaucracy, 61% of physicians reported that they sometimes or often come to work sick. Only 2% of respondents said they never come to work unwell.
Medscape wanted to know more about how often you call in sick, how often you come to work feeling unwell, what symptoms you have, and the dogma of your workplace culture regarding sick days. Not to mention the brutal ethos that starts in medical school, in which calling in sick shows weakness or is unacceptable.
So, we polled 2,347 physicians in the United States and abroad and asked them about their sniffling, sneezing, cold, flu, and fever symptoms, and, of course, COVID. Results were split about 50-50 among male and female physicians. The poll ran from Sept. 28 through Oct. 11.
Coming to work sick
It’s no surprise that the majority of physicians who were polled (85%) have come to work sick in 2022. In the last prepandemic year (2019), about 70% came to work feeling sick one to five times, and 13% worked while sick six to ten times.
When asked about the symptoms that they’ve previously come to work with, 48% of U.S. physicians said multiple symptoms. They gave high marks for runny nose, cough, congestion, and sore throat. Only 27% have worked with a fever, 22% have worked with other symptoms, and 7% have worked with both strep throat and COVID.
“My workplace, especially in the COVID years, accommodates persons who honestly do not feel well enough to report. Sooner or later, everyone covers for someone else who has to be out,” says Kenneth Abbott, MD, an oncologist in Maryland.
The culture of working while sick
Why doctors come to work when they’re sick is complicated. The overwhelming majority of U.S. respondents cited professional obligations; 73% noted that they feel a professional obligation to their patients, and 72% feel a professional obligation to their co-workers. Half of the polled U.S. physicians said they didn’t feel bad enough to stay home, while 48% said they had too much work to do to stay home.
Some 45% said the expectation at their workplace is to come to work unless seriously ill; 43% had too many patients to see; and 18% didn’t think they were contagious when they headed to work sick. Unfortunately, 15% chose to work while sick because otherwise they would lose pay.
In light of these responses, it’s not surprising that 93% reported they’d seen other medical professionals working when sick.
“My schedule is almost always booked weeks in advance. If someone misses or has to cancel their appointment, they typically have 2-4 weeks to wait to get back in. If I was sick and a full day of patients (or God forbid more than a day) had to be canceled because I called in, it’s so much more work when I return,” says Caitlin Briggs, MD, a psychiatrist in Lexington, Ky.
Doctors’ workplace sick day policy
Most employees’ benefits allow at least a few sick days, but doctors who treat society’s ill patients don’t seem to stay home from work when they’re suffering. So, we asked physicians, official policy aside, whether they thought going to work sick was expected in their workplace. The majority (76%) said yes, while 24% said no.
“Unless I’m dying or extremely contagious, I usually work. At least now, I have the telehealth option. Not saying any of this is right, but it’s the reality we deal with and the choice we must make,” says Dr. Briggs.
Additionally, 58% of polled physicians said their workplace did not have a clearly defined policy against coming to work sick, while 20% said theirs did, and 22% weren’t sure.
“The first thing I heard on the subject as a medical student was that sick people come to the hospital, so if you’re sick, then you come to the hospital too ... to work. If you can’t work, then you will be admitted. Another aphorism was from Churchill, that ‘most of the world’s work is done by people who don’t feel very well,’ ” says Paul Andreason, MD, a psychiatrist in Bethesda, Md.
Working in the time of COVID
Working while ill during ordinary times is one thing, but what about working in the time of COVID? Has the pandemic changed the culture of coming to work sick because medical facilities, such as doctor’s offices and hospitals, don’t want their staff coming in when they have COVID?
Surprisingly, when we asked physicians whether the pandemic has made it more or less acceptable to come to work sick, only 61% thought COVID has made it less acceptable to work while sick, while 16% thought it made it more acceptable, and 23% said there’s no change.
“I draw the line at fevers/chills, feeling like you’ve just been run over, or significant enteritis,” says Dr. Abbott. “Also, if I have to take palliative meds that interfere with alertness, I’m not doing my patients any favors.”
While a minority of physicians may call in sick, most still suffer through their sneezing, coughing, chills, and fever while seeing patients as usual.
A version of this article first appeared on Medscape.com.
New CDC guidance on prescribing opioids for pain
The 2022 Clinical Practice Guideline provides guidance on determining whether to initiate opioids for pain; selecting opioids and determining opioid dosages; deciding duration of initial opioid prescription and conducting follow-up; and assessing risk and addressing potential harms of opioid use.
“Patients with pain should receive compassionate, safe, and effective pain care. We want clinicians and patients to have the information they need to weigh the benefits of different approaches to pain care, with the goal of helping people reduce their pain and improve their quality of life,” Christopher M. Jones, PharmD, DrPH, acting director for the CDC’s National Center for Injury Prevention and Control, said in a news release.
How to taper safely
The last guideline on the topic was released by CDC in 2016. Since then, new evidence has emerged regarding the benefits and risks of prescription opioids for acute and chronic pain, comparisons with nonopioid pain treatments, dosing strategies, opioid dose-dependent effects, risk mitigation strategies, and opioid tapering and discontinuation, the CDC says.
A “critical” addition to the 2022 guideline is advice on tapering opioids, Dr. Jones said during a press briefing.
“Practical tips on how to taper in an individualized patient-centered manner have been added to help clinicians if the decision is made to taper opioids, and the guideline explicitly advises against abrupt discontinuation or rapid dose reductions of opioids,” Dr. Jones said.
“That is based on lessons learned over the last several years as well as new science about how we approach tapering and the real harms that can result when patients are abruptly discontinued or rapidly tapered,” he added.
The updated guideline was published online Nov. 3 in the Morbidity and Mortality Weekly Report.
Key recommendations in the 100-page document include the following:
- In determining whether or not to initiate opioids, nonopioid therapies are at least as effective as opioids for many common types of acute pain. Use of nondrug and nonopioid drug therapies should be maximized as appropriate, and opioid therapy should only be considered for acute pain if it is anticipated that benefits outweigh risks to the patient.
- Before starting opioid therapy, providers should discuss with patients the realistic benefits and known risks of opioid therapy.
- Before starting ongoing opioid therapy for patients with subacute pain lasting 1 to 3 months or chronic pain lasting more than 3 months, providers should work with patients to establish treatment goals for pain and function, and consideration should be given as to how opioid therapy will be discontinued if benefits do not outweigh risks.
- Once opioids are started, the lowest effective dose of immediate-release opioids should be prescribed for no longer than needed for the expected duration of pain severe enough to require opioids.
- Within 1 to 4 weeks of starting opioid therapy for subacute or chronic pain, providers should work with patients to evaluate and carefully weigh benefits and risks of continuing opioid therapy; care should be exercised when increasing, continuing, or reducing opioid dosage.
- Before starting and periodically during ongoing opioid therapy, providers should evaluate risk for opioid-related harms and should work with patients to incorporate relevant strategies to mitigate risk, including offering naloxone and reviewing potential interactions with any other prescribed medications or substance used.
- Abrupt discontinuation of opioids should be avoided, especially for patients receiving high doses.
- For treating patients with opioid use disorder, treatment with evidence-based medications should be provided, or arrangements for such treatment should be made.
Dr. Jones emphasized that the guideline is “voluntary and meant to guide shared decision-making between a clinician and patient. It’s not meant to be implemented as absolute limits of policy or practice by clinicians, health systems, insurance companies, governmental entities.”
He also noted that the “current state of the overdose crisis, which is very much driven by illicit synthetic opioids, is not the aim of this guideline.
“The release of this guideline is really about advancing pain care and improving the lives of patients living with pain,” he said.
“We know that at least 1 in 5 people in the country have chronic pain. It’s one of the most common reasons why people present to their health care provider, and the goal here is to advance pain care, function, and quality of life for that patient population, while also reducing misuse, diversion, and consequences of prescription opioid misuse,” Dr. Jones added.
A version of this article first appeared on Medscape.com.
The 2022 Clinical Practice Guideline provides guidance on determining whether to initiate opioids for pain; selecting opioids and determining opioid dosages; deciding duration of initial opioid prescription and conducting follow-up; and assessing risk and addressing potential harms of opioid use.
“Patients with pain should receive compassionate, safe, and effective pain care. We want clinicians and patients to have the information they need to weigh the benefits of different approaches to pain care, with the goal of helping people reduce their pain and improve their quality of life,” Christopher M. Jones, PharmD, DrPH, acting director for the CDC’s National Center for Injury Prevention and Control, said in a news release.
How to taper safely
The last guideline on the topic was released by CDC in 2016. Since then, new evidence has emerged regarding the benefits and risks of prescription opioids for acute and chronic pain, comparisons with nonopioid pain treatments, dosing strategies, opioid dose-dependent effects, risk mitigation strategies, and opioid tapering and discontinuation, the CDC says.
A “critical” addition to the 2022 guideline is advice on tapering opioids, Dr. Jones said during a press briefing.
“Practical tips on how to taper in an individualized patient-centered manner have been added to help clinicians if the decision is made to taper opioids, and the guideline explicitly advises against abrupt discontinuation or rapid dose reductions of opioids,” Dr. Jones said.
“That is based on lessons learned over the last several years as well as new science about how we approach tapering and the real harms that can result when patients are abruptly discontinued or rapidly tapered,” he added.
The updated guideline was published online Nov. 3 in the Morbidity and Mortality Weekly Report.
Key recommendations in the 100-page document include the following:
- In determining whether or not to initiate opioids, nonopioid therapies are at least as effective as opioids for many common types of acute pain. Use of nondrug and nonopioid drug therapies should be maximized as appropriate, and opioid therapy should only be considered for acute pain if it is anticipated that benefits outweigh risks to the patient.
- Before starting opioid therapy, providers should discuss with patients the realistic benefits and known risks of opioid therapy.
- Before starting ongoing opioid therapy for patients with subacute pain lasting 1 to 3 months or chronic pain lasting more than 3 months, providers should work with patients to establish treatment goals for pain and function, and consideration should be given as to how opioid therapy will be discontinued if benefits do not outweigh risks.
- Once opioids are started, the lowest effective dose of immediate-release opioids should be prescribed for no longer than needed for the expected duration of pain severe enough to require opioids.
- Within 1 to 4 weeks of starting opioid therapy for subacute or chronic pain, providers should work with patients to evaluate and carefully weigh benefits and risks of continuing opioid therapy; care should be exercised when increasing, continuing, or reducing opioid dosage.
- Before starting and periodically during ongoing opioid therapy, providers should evaluate risk for opioid-related harms and should work with patients to incorporate relevant strategies to mitigate risk, including offering naloxone and reviewing potential interactions with any other prescribed medications or substance used.
- Abrupt discontinuation of opioids should be avoided, especially for patients receiving high doses.
- For treating patients with opioid use disorder, treatment with evidence-based medications should be provided, or arrangements for such treatment should be made.
Dr. Jones emphasized that the guideline is “voluntary and meant to guide shared decision-making between a clinician and patient. It’s not meant to be implemented as absolute limits of policy or practice by clinicians, health systems, insurance companies, governmental entities.”
He also noted that the “current state of the overdose crisis, which is very much driven by illicit synthetic opioids, is not the aim of this guideline.
“The release of this guideline is really about advancing pain care and improving the lives of patients living with pain,” he said.
“We know that at least 1 in 5 people in the country have chronic pain. It’s one of the most common reasons why people present to their health care provider, and the goal here is to advance pain care, function, and quality of life for that patient population, while also reducing misuse, diversion, and consequences of prescription opioid misuse,” Dr. Jones added.
A version of this article first appeared on Medscape.com.
The 2022 Clinical Practice Guideline provides guidance on determining whether to initiate opioids for pain; selecting opioids and determining opioid dosages; deciding duration of initial opioid prescription and conducting follow-up; and assessing risk and addressing potential harms of opioid use.
“Patients with pain should receive compassionate, safe, and effective pain care. We want clinicians and patients to have the information they need to weigh the benefits of different approaches to pain care, with the goal of helping people reduce their pain and improve their quality of life,” Christopher M. Jones, PharmD, DrPH, acting director for the CDC’s National Center for Injury Prevention and Control, said in a news release.
How to taper safely
The last guideline on the topic was released by CDC in 2016. Since then, new evidence has emerged regarding the benefits and risks of prescription opioids for acute and chronic pain, comparisons with nonopioid pain treatments, dosing strategies, opioid dose-dependent effects, risk mitigation strategies, and opioid tapering and discontinuation, the CDC says.
A “critical” addition to the 2022 guideline is advice on tapering opioids, Dr. Jones said during a press briefing.
“Practical tips on how to taper in an individualized patient-centered manner have been added to help clinicians if the decision is made to taper opioids, and the guideline explicitly advises against abrupt discontinuation or rapid dose reductions of opioids,” Dr. Jones said.
“That is based on lessons learned over the last several years as well as new science about how we approach tapering and the real harms that can result when patients are abruptly discontinued or rapidly tapered,” he added.
The updated guideline was published online Nov. 3 in the Morbidity and Mortality Weekly Report.
Key recommendations in the 100-page document include the following:
- In determining whether or not to initiate opioids, nonopioid therapies are at least as effective as opioids for many common types of acute pain. Use of nondrug and nonopioid drug therapies should be maximized as appropriate, and opioid therapy should only be considered for acute pain if it is anticipated that benefits outweigh risks to the patient.
- Before starting opioid therapy, providers should discuss with patients the realistic benefits and known risks of opioid therapy.
- Before starting ongoing opioid therapy for patients with subacute pain lasting 1 to 3 months or chronic pain lasting more than 3 months, providers should work with patients to establish treatment goals for pain and function, and consideration should be given as to how opioid therapy will be discontinued if benefits do not outweigh risks.
- Once opioids are started, the lowest effective dose of immediate-release opioids should be prescribed for no longer than needed for the expected duration of pain severe enough to require opioids.
- Within 1 to 4 weeks of starting opioid therapy for subacute or chronic pain, providers should work with patients to evaluate and carefully weigh benefits and risks of continuing opioid therapy; care should be exercised when increasing, continuing, or reducing opioid dosage.
- Before starting and periodically during ongoing opioid therapy, providers should evaluate risk for opioid-related harms and should work with patients to incorporate relevant strategies to mitigate risk, including offering naloxone and reviewing potential interactions with any other prescribed medications or substance used.
- Abrupt discontinuation of opioids should be avoided, especially for patients receiving high doses.
- For treating patients with opioid use disorder, treatment with evidence-based medications should be provided, or arrangements for such treatment should be made.
Dr. Jones emphasized that the guideline is “voluntary and meant to guide shared decision-making between a clinician and patient. It’s not meant to be implemented as absolute limits of policy or practice by clinicians, health systems, insurance companies, governmental entities.”
He also noted that the “current state of the overdose crisis, which is very much driven by illicit synthetic opioids, is not the aim of this guideline.
“The release of this guideline is really about advancing pain care and improving the lives of patients living with pain,” he said.
“We know that at least 1 in 5 people in the country have chronic pain. It’s one of the most common reasons why people present to their health care provider, and the goal here is to advance pain care, function, and quality of life for that patient population, while also reducing misuse, diversion, and consequences of prescription opioid misuse,” Dr. Jones added.
A version of this article first appeared on Medscape.com.