User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Biden to end COVID emergencies in May
Doing so will have many effects, including the end of free vaccines and health services to fight the pandemic. The public health emergency has been renewed every 90 days since it was declared by the Trump administration in January 2020.
The declaration allowed major changes throughout the health care system to deal with the pandemic, including the free distribution of vaccines, testing, and treatments. In addition, telehealth services were expanded, and Medicaid and the Children’s Health Insurance Program were extended to millions more Americans.
Biden said the COVID-19 national emergency is set to expire March 1 while the declared public health emergency would currently expire on April 11. The president said both will be extended to end May 11.
There were nearly 300,000 newly reported COVID-19 cases in the United States for the week ending Jan. 25, according to CDC data, as well as more than 3,750 deaths.
A version of this article first appeared on WebMD.com.
Doing so will have many effects, including the end of free vaccines and health services to fight the pandemic. The public health emergency has been renewed every 90 days since it was declared by the Trump administration in January 2020.
The declaration allowed major changes throughout the health care system to deal with the pandemic, including the free distribution of vaccines, testing, and treatments. In addition, telehealth services were expanded, and Medicaid and the Children’s Health Insurance Program were extended to millions more Americans.
Biden said the COVID-19 national emergency is set to expire March 1 while the declared public health emergency would currently expire on April 11. The president said both will be extended to end May 11.
There were nearly 300,000 newly reported COVID-19 cases in the United States for the week ending Jan. 25, according to CDC data, as well as more than 3,750 deaths.
A version of this article first appeared on WebMD.com.
Doing so will have many effects, including the end of free vaccines and health services to fight the pandemic. The public health emergency has been renewed every 90 days since it was declared by the Trump administration in January 2020.
The declaration allowed major changes throughout the health care system to deal with the pandemic, including the free distribution of vaccines, testing, and treatments. In addition, telehealth services were expanded, and Medicaid and the Children’s Health Insurance Program were extended to millions more Americans.
Biden said the COVID-19 national emergency is set to expire March 1 while the declared public health emergency would currently expire on April 11. The president said both will be extended to end May 11.
There were nearly 300,000 newly reported COVID-19 cases in the United States for the week ending Jan. 25, according to CDC data, as well as more than 3,750 deaths.
A version of this article first appeared on WebMD.com.
Healthy habits lower T2D microvascular risks: Cohort study
People with diabetes who adhere to a healthy diet, exercise regularly, and follow other healthy lifestyle practices have a significantly lower risk of microvascular complications from the disease, such as diabetic neuropathy, retinopathy, and nephropathy, as well as foot disorders, than counterparts with diabetes who don’t, a prospective cohort study of more than 7,000 patients with type 2 diabetes has found.
“We believe this is one of the first large-scale analyses among diabetes patients that specifically examined an overall healthy lifestyle in relation to the risk of developing microvascular complications,” senior study author Qi Sun, MD, ScD, said in an interview. “The results are not surprising that the healthy lifestyle is associated with lower risk of developing these complications and the enhanced adherence to the healthy lifestyle is associated with lower risk as well. And these findings bear lots of public health significance as they suggest the important role of living a healthy lifestyle in the prevention of diabetes complications, on top of the clinical treatment.”
Dr. Sun is an associate professor of nutrition and epidemiology at the Harvard T.H. Chan School of Public Health, Boston.
The study stated that the findings “lend support” for the American Diabetes Association guidelines for healthy lifestyle practices in people with diabetes.
The study used a cohort from two large prospective cohort studies, the Nurses’ Health Study (NHS) and the Health Professionals Follow-up Study (HPFS), comprising 4,982 women and 2,095 men who were diagnosed with type 2 diabetes during follow-up. They had no cardiovascular disease or cancer at the time of their diabetes diagnosis. Both NHS and HPFS used validated questionnaires to gather information on diet, lifestyle, medical history, and newly diagnosed diseases every 2-4 years. The latter study included NHS and HPFS participants who also completed supplementary questionnaires about their diabetes.
The latest study took into account five modifiable lifestyle-related factors: diet, body weight, smoking status, alcohol, and physical activity. For diet, both large studies used the 2010 Alternate Healthy Eating Index to assess diet quality; those in the upper 40th percentile of the study population were defined as healthy diet. Healthy body weight was defined at a body mass index of 18.5-25 kg/m2.
Among the latter study cohort, 2,878 incident cases of diabetic microvascular complications were documented during follow-up. Patients who adhered to a healthy lifestyle before their diabetes diagnosis, defined as having four or more low-risk lifestyle factors, had a 27% lower relative risk of developing any microvascular complication than counterparts with no low-risk lifestyle factors (relative risk, 0.73; 95% confidence interval, 0.35-1; P = .006).
The study found similar outcomes for those who adopted a healthy lifestyle after their diabetes diagnosis, with a 32% reduction in relative risk compared with those who didn’t adopt any healthy lifestyle practices (RR, 0.68; 95% CI, 0.55-0.83; P < .001).
Dr. Sun noted what was noteworthy about his group’s cohort study. “The unique design is truly the prospective follow-up over time so that we could examine the lifestyle at diabetes diagnosis as well as changes in lifestyle before and after diabetes in relation to the future risk of developing the complications,” he said.
A randomized trial would be a more rigorous way to evaluate the impact of a healthy lifestyle, he added, “although it’s much more expensive than a cohort study like what we did with this investigation.”
As for future research, Dr. Sun said, “It will be interesting to understand mechanisms underlying these observations. It’s also critical to understand why certain diabetes patients may not benefit from a healthy lifestyle, since some of them, even when living a healthy lifestyle, still develop the complications.”
This trial shows in a new light the benefits of healthy lifestyle practices on microvascular complications of type 2 diabetes, Paul S. Jellinger, MD, of the Center for Diabetes and Endocrine Care in Hollywood, Fla., and a professor at the University of Miami, said in a comment. “These benefits have always been surmised and demonstrated in a limited way in previous trials, but not subject to the level of analysis seen in this prospective cohort trial.”
He called the study design “excellent,” adding, “ ‘Validated’ self-reported questionnaires were used widely, although minimal detail is provided about the validation process.” One limitation, he noted, was “the homogeneity of the participants; all were health professionals.”
The study “affirms” and “quantitates” the benefits of a healthy lifestyle in type 2 diabetes. “The issue is not unawareness but rather application,” Dr. Jellinger said. “Modifying long-held lifestyle habits is a real challenge. Perhaps by ‘quantitating’ the benefit, as shown in this trial and hopefully additional studies, impetus will be provided to refocus on this approach, which is too often simply given lip service.”
The National Institutes of Health provided funding for the study. Dr. Sun has no relevant disclosures. Dr. Jellinger disclosed relationships with Amgen and Esperion.
People with diabetes who adhere to a healthy diet, exercise regularly, and follow other healthy lifestyle practices have a significantly lower risk of microvascular complications from the disease, such as diabetic neuropathy, retinopathy, and nephropathy, as well as foot disorders, than counterparts with diabetes who don’t, a prospective cohort study of more than 7,000 patients with type 2 diabetes has found.
“We believe this is one of the first large-scale analyses among diabetes patients that specifically examined an overall healthy lifestyle in relation to the risk of developing microvascular complications,” senior study author Qi Sun, MD, ScD, said in an interview. “The results are not surprising that the healthy lifestyle is associated with lower risk of developing these complications and the enhanced adherence to the healthy lifestyle is associated with lower risk as well. And these findings bear lots of public health significance as they suggest the important role of living a healthy lifestyle in the prevention of diabetes complications, on top of the clinical treatment.”
Dr. Sun is an associate professor of nutrition and epidemiology at the Harvard T.H. Chan School of Public Health, Boston.
The study stated that the findings “lend support” for the American Diabetes Association guidelines for healthy lifestyle practices in people with diabetes.
The study used a cohort from two large prospective cohort studies, the Nurses’ Health Study (NHS) and the Health Professionals Follow-up Study (HPFS), comprising 4,982 women and 2,095 men who were diagnosed with type 2 diabetes during follow-up. They had no cardiovascular disease or cancer at the time of their diabetes diagnosis. Both NHS and HPFS used validated questionnaires to gather information on diet, lifestyle, medical history, and newly diagnosed diseases every 2-4 years. The latter study included NHS and HPFS participants who also completed supplementary questionnaires about their diabetes.
The latest study took into account five modifiable lifestyle-related factors: diet, body weight, smoking status, alcohol, and physical activity. For diet, both large studies used the 2010 Alternate Healthy Eating Index to assess diet quality; those in the upper 40th percentile of the study population were defined as healthy diet. Healthy body weight was defined at a body mass index of 18.5-25 kg/m2.
Among the latter study cohort, 2,878 incident cases of diabetic microvascular complications were documented during follow-up. Patients who adhered to a healthy lifestyle before their diabetes diagnosis, defined as having four or more low-risk lifestyle factors, had a 27% lower relative risk of developing any microvascular complication than counterparts with no low-risk lifestyle factors (relative risk, 0.73; 95% confidence interval, 0.35-1; P = .006).
The study found similar outcomes for those who adopted a healthy lifestyle after their diabetes diagnosis, with a 32% reduction in relative risk compared with those who didn’t adopt any healthy lifestyle practices (RR, 0.68; 95% CI, 0.55-0.83; P < .001).
Dr. Sun noted what was noteworthy about his group’s cohort study. “The unique design is truly the prospective follow-up over time so that we could examine the lifestyle at diabetes diagnosis as well as changes in lifestyle before and after diabetes in relation to the future risk of developing the complications,” he said.
A randomized trial would be a more rigorous way to evaluate the impact of a healthy lifestyle, he added, “although it’s much more expensive than a cohort study like what we did with this investigation.”
As for future research, Dr. Sun said, “It will be interesting to understand mechanisms underlying these observations. It’s also critical to understand why certain diabetes patients may not benefit from a healthy lifestyle, since some of them, even when living a healthy lifestyle, still develop the complications.”
This trial shows in a new light the benefits of healthy lifestyle practices on microvascular complications of type 2 diabetes, Paul S. Jellinger, MD, of the Center for Diabetes and Endocrine Care in Hollywood, Fla., and a professor at the University of Miami, said in a comment. “These benefits have always been surmised and demonstrated in a limited way in previous trials, but not subject to the level of analysis seen in this prospective cohort trial.”
He called the study design “excellent,” adding, “ ‘Validated’ self-reported questionnaires were used widely, although minimal detail is provided about the validation process.” One limitation, he noted, was “the homogeneity of the participants; all were health professionals.”
The study “affirms” and “quantitates” the benefits of a healthy lifestyle in type 2 diabetes. “The issue is not unawareness but rather application,” Dr. Jellinger said. “Modifying long-held lifestyle habits is a real challenge. Perhaps by ‘quantitating’ the benefit, as shown in this trial and hopefully additional studies, impetus will be provided to refocus on this approach, which is too often simply given lip service.”
The National Institutes of Health provided funding for the study. Dr. Sun has no relevant disclosures. Dr. Jellinger disclosed relationships with Amgen and Esperion.
People with diabetes who adhere to a healthy diet, exercise regularly, and follow other healthy lifestyle practices have a significantly lower risk of microvascular complications from the disease, such as diabetic neuropathy, retinopathy, and nephropathy, as well as foot disorders, than counterparts with diabetes who don’t, a prospective cohort study of more than 7,000 patients with type 2 diabetes has found.
“We believe this is one of the first large-scale analyses among diabetes patients that specifically examined an overall healthy lifestyle in relation to the risk of developing microvascular complications,” senior study author Qi Sun, MD, ScD, said in an interview. “The results are not surprising that the healthy lifestyle is associated with lower risk of developing these complications and the enhanced adherence to the healthy lifestyle is associated with lower risk as well. And these findings bear lots of public health significance as they suggest the important role of living a healthy lifestyle in the prevention of diabetes complications, on top of the clinical treatment.”
Dr. Sun is an associate professor of nutrition and epidemiology at the Harvard T.H. Chan School of Public Health, Boston.
The study stated that the findings “lend support” for the American Diabetes Association guidelines for healthy lifestyle practices in people with diabetes.
The study used a cohort from two large prospective cohort studies, the Nurses’ Health Study (NHS) and the Health Professionals Follow-up Study (HPFS), comprising 4,982 women and 2,095 men who were diagnosed with type 2 diabetes during follow-up. They had no cardiovascular disease or cancer at the time of their diabetes diagnosis. Both NHS and HPFS used validated questionnaires to gather information on diet, lifestyle, medical history, and newly diagnosed diseases every 2-4 years. The latter study included NHS and HPFS participants who also completed supplementary questionnaires about their diabetes.
The latest study took into account five modifiable lifestyle-related factors: diet, body weight, smoking status, alcohol, and physical activity. For diet, both large studies used the 2010 Alternate Healthy Eating Index to assess diet quality; those in the upper 40th percentile of the study population were defined as healthy diet. Healthy body weight was defined at a body mass index of 18.5-25 kg/m2.
Among the latter study cohort, 2,878 incident cases of diabetic microvascular complications were documented during follow-up. Patients who adhered to a healthy lifestyle before their diabetes diagnosis, defined as having four or more low-risk lifestyle factors, had a 27% lower relative risk of developing any microvascular complication than counterparts with no low-risk lifestyle factors (relative risk, 0.73; 95% confidence interval, 0.35-1; P = .006).
The study found similar outcomes for those who adopted a healthy lifestyle after their diabetes diagnosis, with a 32% reduction in relative risk compared with those who didn’t adopt any healthy lifestyle practices (RR, 0.68; 95% CI, 0.55-0.83; P < .001).
Dr. Sun noted what was noteworthy about his group’s cohort study. “The unique design is truly the prospective follow-up over time so that we could examine the lifestyle at diabetes diagnosis as well as changes in lifestyle before and after diabetes in relation to the future risk of developing the complications,” he said.
A randomized trial would be a more rigorous way to evaluate the impact of a healthy lifestyle, he added, “although it’s much more expensive than a cohort study like what we did with this investigation.”
As for future research, Dr. Sun said, “It will be interesting to understand mechanisms underlying these observations. It’s also critical to understand why certain diabetes patients may not benefit from a healthy lifestyle, since some of them, even when living a healthy lifestyle, still develop the complications.”
This trial shows in a new light the benefits of healthy lifestyle practices on microvascular complications of type 2 diabetes, Paul S. Jellinger, MD, of the Center for Diabetes and Endocrine Care in Hollywood, Fla., and a professor at the University of Miami, said in a comment. “These benefits have always been surmised and demonstrated in a limited way in previous trials, but not subject to the level of analysis seen in this prospective cohort trial.”
He called the study design “excellent,” adding, “ ‘Validated’ self-reported questionnaires were used widely, although minimal detail is provided about the validation process.” One limitation, he noted, was “the homogeneity of the participants; all were health professionals.”
The study “affirms” and “quantitates” the benefits of a healthy lifestyle in type 2 diabetes. “The issue is not unawareness but rather application,” Dr. Jellinger said. “Modifying long-held lifestyle habits is a real challenge. Perhaps by ‘quantitating’ the benefit, as shown in this trial and hopefully additional studies, impetus will be provided to refocus on this approach, which is too often simply given lip service.”
The National Institutes of Health provided funding for the study. Dr. Sun has no relevant disclosures. Dr. Jellinger disclosed relationships with Amgen and Esperion.
FROM JAMA NETWORK OPEN
Six healthy lifestyle habits linked to slowed memory decline
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.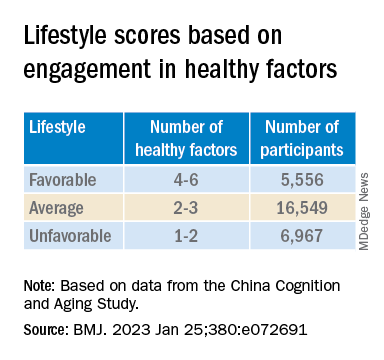
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.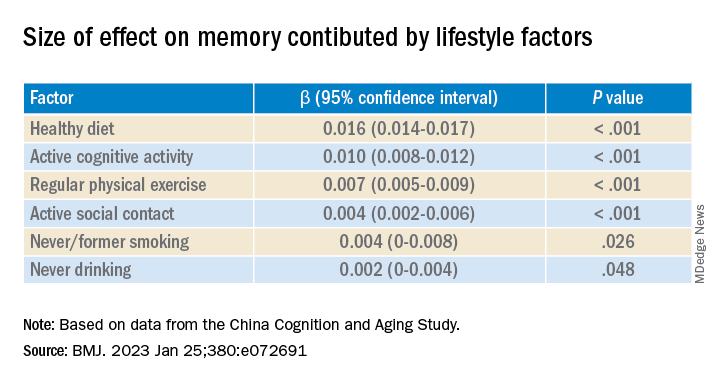
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
Angioedema risk jumps when switching HF meds
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
‘Sugar tax’ prevented thousands of girls becoming obese
The introduction of the soft drinks industry levy (SDIL) – dubbed the ‘sugar tax’ – in England was followed by a drop in the number of older primary school girls succumbing to obesity, according to researchers from the Universities of Cambridge, Oxford, and Bath, with colleagues at the London School of Hygiene and Tropical Medicine.
The study, published in PLOS Medicine, has led to calls to extend the levy to other unhealthy foods and drinks
Obesity has become a global public health problem, the researchers said. In England, around 10% of 4- to 5-year-old children and 20% of 10- to 11-year-olds were recorded as obese in 2020. Childhood obesity is associated with depression in children and the adults into which they maturate, as well as with serious health problems in later life including high blood pressure and type 2 diabetes.
In the United Kingdom, young people consume significantly more added sugars than are recommended – by late adolescence, typically 70 g of added sugar per day, more than double the recommended 30g. The team said that sugar-sweetened beverages (SSB) are the primary sources of dietary added sugars in children, with high consumption commonly observed in more deprived areas where obesity prevalence is also highest.
Protecting children from excessive sugar
The two-tier SDIL on drinks manufacturers was implemented in April 2018 and aimed to protect children from excessive sugar consumption and tackle childhood obesity by incentivizing reformulation of SSBs in the U.K. with reduced sugar content.
To assess the effects of SDIL, the researchers used data from the National Child Measurement Programme on over 1 million children at ages 4 to 5 years (reception class) and 10 to 11 years (school year 6) in state-maintained English primary schools. The surveillance program includes annual repeat cross-sectional measurements, enabling the researchers to examine trajectories in monthly prevalence of obesity from September 2013 to November 2019, 19 months after the implementation of the SDIL.
Taking account of previous trends in obesity levels, they estimated both absolute and relative changes in obesity prevalence, both overall and by sex and deprivation, and compared obesity levels after the SDIL with predicted levels had the tax not been introduced, controlling for children’s sex and the level of deprivation of their school area.
Although they found no significant association with obesity levels in reception-age children or year-6 boys, they noted an overall absolute reduction in obesity prevalence of 1.6 percentage points (PPs) (95% confidence interval, 1.1-2.1) in 10- to 11-year-old (year 6) girls. This equated to an 8% relative reduction in obesity rates compared with a counterfactual estimated from the trend prior to the SDIL announcement in March 2016, adjusted for temporal variations in obesity prevalence.
The researchers estimated that this was equivalent to preventing 5,234 cases of obesity per year in this group of year-6 girls alone.
Obesity reductions greatest in most deprived areas
Reductions were greatest in girls whose schools were in the most deprived areas, where children are known to consume the largest amount of sugary drinks. The greatest reductions in obesity were observed in the two most deprived quintiles – such that in the lowest quintile the absolute obesity prevalence reduction was 2.4 PP (95% CI, 1.6-3.2), equivalent to a 9% reduction in those living in the most deprived areas.
There are several reasons why the sugar tax did not lead to changes in levels of obesity among the younger children, the researchers said. Very young children consume fewer sugar-sweetened drinks than older children, so the soft drinks levy would have had a smaller effect. Also, fruit juices are not included in the levy, but contribute similar amounts of sugar in young children’s diets as do sugar-sweetened beverages.
Advertising may impact consumption in boys
It’s also unclear why the sugar tax might affect obesity prevalence in girls and boys differently, they said, especially since boys are higher consumers of sugar-sweetened beverages. One explanation is the possible impact of advertising – numerous studies have found that boys are often exposed to more food advertising than girls, both through higher levels of TV viewing and in how adverts are framed. Physical activity is often used to promote junk food and boys, compared with girls, have been shown to be more likely to believe that energy-dense junk foods depicted in adverts will boost physical performance, and so are more likely to choose energy-dense, nutrient-poor products following celebrity endorsements.
Tax ‘led to positive health impacts’
“Our findings suggest that the U.K. SDIL led to positive health impacts in the form of reduced obesity levels in girls aged 10-11 years,” the authors said. However: “Additional strategies beyond SSB taxation will be needed to reduce obesity prevalence overall, and particularly in older boys and younger children.”
Dr. Nina Rogers from the MRC Epidemiology Unit at Cambridge (England), who led the study, said: “We urgently need to find ways to tackle the increasing numbers of children living with obesity, otherwise we risk our children growing up to face significant health problems. That was one reason why the U.K.’s SDIL was introduced, and the evidence so far is promising. We’ve shown for the first time that it is likely to have helped prevent thousands of children each year becoming obese.
“It isn’t a straightforward picture, though, as it was mainly older girls who benefited. But the fact that we saw the biggest difference among girls from areas of high deprivation is important and is a step towards reducing the health inequalities they face.”
Although the researchers found an association rather than a causal link, this study adds to previous findings that the levy was associated with a substantial reduction in the amount of sugar in soft drinks.
Senior author Professor Jean Adams from the MRC Epidemiology Unit said: “We know that consuming too many sugary drinks contributes to obesity and that the U.K. soft drinks levy led to a drop in the amount of sugar in soft drinks available in the U.K., so it makes sense that we also see a drop in cases of obesity, although we only found this in girls. Children from more deprived backgrounds tend to consume the largest amount of sugary drinks, and it was among girls in this group that we saw the biggest change.”
Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, said: “The claim that the soft drink levy might have prevented 5,000 children from becoming obese is speculative because it is based on an association not actual measurements of consumption.”
He added that: “As well as continuing to discourage the consumption of sugar sweetened beverages and sweets, wider recognition should be given to foods such as biscuits [and] deep-fried foods (crisps, corn snacks, chips) that make [a] bigger contribution to excess calorie intake in children. Tackling poverty, however, is probably [the] best way to improve the diets of socially deprived children.”
Government ‘should learn from this success’
Asked to comment by this news organization, Katharine Jenner, director of the Obesity Health Alliance, said: “Government should be heartened that their soft drinks policy is already improving the health of young girls, regardless of where they live. The government should learn from this success, especially when compared with the many unsuccessful attempts to persuade industry to change their products voluntarily. They must now press ahead with policies that make it easier for everyone to eat a healthier diet, including extending the soft drinks industry levy to include other less healthy foods and drinks and measures to take junk food out of the spotlight.
“The research notes that numerous studies have found that boys are often exposed to more food advertising content than girls, negating the impact of the soft drinks levy [so] we need restriction on junk food marketing now, to put healthy food back in the spotlight.”
The research was supported by the National Institute of Health and Care Research and the Medical Research Council.
A version of this article originally appeared on MedscapeUK.
The introduction of the soft drinks industry levy (SDIL) – dubbed the ‘sugar tax’ – in England was followed by a drop in the number of older primary school girls succumbing to obesity, according to researchers from the Universities of Cambridge, Oxford, and Bath, with colleagues at the London School of Hygiene and Tropical Medicine.
The study, published in PLOS Medicine, has led to calls to extend the levy to other unhealthy foods and drinks
Obesity has become a global public health problem, the researchers said. In England, around 10% of 4- to 5-year-old children and 20% of 10- to 11-year-olds were recorded as obese in 2020. Childhood obesity is associated with depression in children and the adults into which they maturate, as well as with serious health problems in later life including high blood pressure and type 2 diabetes.
In the United Kingdom, young people consume significantly more added sugars than are recommended – by late adolescence, typically 70 g of added sugar per day, more than double the recommended 30g. The team said that sugar-sweetened beverages (SSB) are the primary sources of dietary added sugars in children, with high consumption commonly observed in more deprived areas where obesity prevalence is also highest.
Protecting children from excessive sugar
The two-tier SDIL on drinks manufacturers was implemented in April 2018 and aimed to protect children from excessive sugar consumption and tackle childhood obesity by incentivizing reformulation of SSBs in the U.K. with reduced sugar content.
To assess the effects of SDIL, the researchers used data from the National Child Measurement Programme on over 1 million children at ages 4 to 5 years (reception class) and 10 to 11 years (school year 6) in state-maintained English primary schools. The surveillance program includes annual repeat cross-sectional measurements, enabling the researchers to examine trajectories in monthly prevalence of obesity from September 2013 to November 2019, 19 months after the implementation of the SDIL.
Taking account of previous trends in obesity levels, they estimated both absolute and relative changes in obesity prevalence, both overall and by sex and deprivation, and compared obesity levels after the SDIL with predicted levels had the tax not been introduced, controlling for children’s sex and the level of deprivation of their school area.
Although they found no significant association with obesity levels in reception-age children or year-6 boys, they noted an overall absolute reduction in obesity prevalence of 1.6 percentage points (PPs) (95% confidence interval, 1.1-2.1) in 10- to 11-year-old (year 6) girls. This equated to an 8% relative reduction in obesity rates compared with a counterfactual estimated from the trend prior to the SDIL announcement in March 2016, adjusted for temporal variations in obesity prevalence.
The researchers estimated that this was equivalent to preventing 5,234 cases of obesity per year in this group of year-6 girls alone.
Obesity reductions greatest in most deprived areas
Reductions were greatest in girls whose schools were in the most deprived areas, where children are known to consume the largest amount of sugary drinks. The greatest reductions in obesity were observed in the two most deprived quintiles – such that in the lowest quintile the absolute obesity prevalence reduction was 2.4 PP (95% CI, 1.6-3.2), equivalent to a 9% reduction in those living in the most deprived areas.
There are several reasons why the sugar tax did not lead to changes in levels of obesity among the younger children, the researchers said. Very young children consume fewer sugar-sweetened drinks than older children, so the soft drinks levy would have had a smaller effect. Also, fruit juices are not included in the levy, but contribute similar amounts of sugar in young children’s diets as do sugar-sweetened beverages.
Advertising may impact consumption in boys
It’s also unclear why the sugar tax might affect obesity prevalence in girls and boys differently, they said, especially since boys are higher consumers of sugar-sweetened beverages. One explanation is the possible impact of advertising – numerous studies have found that boys are often exposed to more food advertising than girls, both through higher levels of TV viewing and in how adverts are framed. Physical activity is often used to promote junk food and boys, compared with girls, have been shown to be more likely to believe that energy-dense junk foods depicted in adverts will boost physical performance, and so are more likely to choose energy-dense, nutrient-poor products following celebrity endorsements.
Tax ‘led to positive health impacts’
“Our findings suggest that the U.K. SDIL led to positive health impacts in the form of reduced obesity levels in girls aged 10-11 years,” the authors said. However: “Additional strategies beyond SSB taxation will be needed to reduce obesity prevalence overall, and particularly in older boys and younger children.”
Dr. Nina Rogers from the MRC Epidemiology Unit at Cambridge (England), who led the study, said: “We urgently need to find ways to tackle the increasing numbers of children living with obesity, otherwise we risk our children growing up to face significant health problems. That was one reason why the U.K.’s SDIL was introduced, and the evidence so far is promising. We’ve shown for the first time that it is likely to have helped prevent thousands of children each year becoming obese.
“It isn’t a straightforward picture, though, as it was mainly older girls who benefited. But the fact that we saw the biggest difference among girls from areas of high deprivation is important and is a step towards reducing the health inequalities they face.”
Although the researchers found an association rather than a causal link, this study adds to previous findings that the levy was associated with a substantial reduction in the amount of sugar in soft drinks.
Senior author Professor Jean Adams from the MRC Epidemiology Unit said: “We know that consuming too many sugary drinks contributes to obesity and that the U.K. soft drinks levy led to a drop in the amount of sugar in soft drinks available in the U.K., so it makes sense that we also see a drop in cases of obesity, although we only found this in girls. Children from more deprived backgrounds tend to consume the largest amount of sugary drinks, and it was among girls in this group that we saw the biggest change.”
Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, said: “The claim that the soft drink levy might have prevented 5,000 children from becoming obese is speculative because it is based on an association not actual measurements of consumption.”
He added that: “As well as continuing to discourage the consumption of sugar sweetened beverages and sweets, wider recognition should be given to foods such as biscuits [and] deep-fried foods (crisps, corn snacks, chips) that make [a] bigger contribution to excess calorie intake in children. Tackling poverty, however, is probably [the] best way to improve the diets of socially deprived children.”
Government ‘should learn from this success’
Asked to comment by this news organization, Katharine Jenner, director of the Obesity Health Alliance, said: “Government should be heartened that their soft drinks policy is already improving the health of young girls, regardless of where they live. The government should learn from this success, especially when compared with the many unsuccessful attempts to persuade industry to change their products voluntarily. They must now press ahead with policies that make it easier for everyone to eat a healthier diet, including extending the soft drinks industry levy to include other less healthy foods and drinks and measures to take junk food out of the spotlight.
“The research notes that numerous studies have found that boys are often exposed to more food advertising content than girls, negating the impact of the soft drinks levy [so] we need restriction on junk food marketing now, to put healthy food back in the spotlight.”
The research was supported by the National Institute of Health and Care Research and the Medical Research Council.
A version of this article originally appeared on MedscapeUK.
The introduction of the soft drinks industry levy (SDIL) – dubbed the ‘sugar tax’ – in England was followed by a drop in the number of older primary school girls succumbing to obesity, according to researchers from the Universities of Cambridge, Oxford, and Bath, with colleagues at the London School of Hygiene and Tropical Medicine.
The study, published in PLOS Medicine, has led to calls to extend the levy to other unhealthy foods and drinks
Obesity has become a global public health problem, the researchers said. In England, around 10% of 4- to 5-year-old children and 20% of 10- to 11-year-olds were recorded as obese in 2020. Childhood obesity is associated with depression in children and the adults into which they maturate, as well as with serious health problems in later life including high blood pressure and type 2 diabetes.
In the United Kingdom, young people consume significantly more added sugars than are recommended – by late adolescence, typically 70 g of added sugar per day, more than double the recommended 30g. The team said that sugar-sweetened beverages (SSB) are the primary sources of dietary added sugars in children, with high consumption commonly observed in more deprived areas where obesity prevalence is also highest.
Protecting children from excessive sugar
The two-tier SDIL on drinks manufacturers was implemented in April 2018 and aimed to protect children from excessive sugar consumption and tackle childhood obesity by incentivizing reformulation of SSBs in the U.K. with reduced sugar content.
To assess the effects of SDIL, the researchers used data from the National Child Measurement Programme on over 1 million children at ages 4 to 5 years (reception class) and 10 to 11 years (school year 6) in state-maintained English primary schools. The surveillance program includes annual repeat cross-sectional measurements, enabling the researchers to examine trajectories in monthly prevalence of obesity from September 2013 to November 2019, 19 months after the implementation of the SDIL.
Taking account of previous trends in obesity levels, they estimated both absolute and relative changes in obesity prevalence, both overall and by sex and deprivation, and compared obesity levels after the SDIL with predicted levels had the tax not been introduced, controlling for children’s sex and the level of deprivation of their school area.
Although they found no significant association with obesity levels in reception-age children or year-6 boys, they noted an overall absolute reduction in obesity prevalence of 1.6 percentage points (PPs) (95% confidence interval, 1.1-2.1) in 10- to 11-year-old (year 6) girls. This equated to an 8% relative reduction in obesity rates compared with a counterfactual estimated from the trend prior to the SDIL announcement in March 2016, adjusted for temporal variations in obesity prevalence.
The researchers estimated that this was equivalent to preventing 5,234 cases of obesity per year in this group of year-6 girls alone.
Obesity reductions greatest in most deprived areas
Reductions were greatest in girls whose schools were in the most deprived areas, where children are known to consume the largest amount of sugary drinks. The greatest reductions in obesity were observed in the two most deprived quintiles – such that in the lowest quintile the absolute obesity prevalence reduction was 2.4 PP (95% CI, 1.6-3.2), equivalent to a 9% reduction in those living in the most deprived areas.
There are several reasons why the sugar tax did not lead to changes in levels of obesity among the younger children, the researchers said. Very young children consume fewer sugar-sweetened drinks than older children, so the soft drinks levy would have had a smaller effect. Also, fruit juices are not included in the levy, but contribute similar amounts of sugar in young children’s diets as do sugar-sweetened beverages.
Advertising may impact consumption in boys
It’s also unclear why the sugar tax might affect obesity prevalence in girls and boys differently, they said, especially since boys are higher consumers of sugar-sweetened beverages. One explanation is the possible impact of advertising – numerous studies have found that boys are often exposed to more food advertising than girls, both through higher levels of TV viewing and in how adverts are framed. Physical activity is often used to promote junk food and boys, compared with girls, have been shown to be more likely to believe that energy-dense junk foods depicted in adverts will boost physical performance, and so are more likely to choose energy-dense, nutrient-poor products following celebrity endorsements.
Tax ‘led to positive health impacts’
“Our findings suggest that the U.K. SDIL led to positive health impacts in the form of reduced obesity levels in girls aged 10-11 years,” the authors said. However: “Additional strategies beyond SSB taxation will be needed to reduce obesity prevalence overall, and particularly in older boys and younger children.”
Dr. Nina Rogers from the MRC Epidemiology Unit at Cambridge (England), who led the study, said: “We urgently need to find ways to tackle the increasing numbers of children living with obesity, otherwise we risk our children growing up to face significant health problems. That was one reason why the U.K.’s SDIL was introduced, and the evidence so far is promising. We’ve shown for the first time that it is likely to have helped prevent thousands of children each year becoming obese.
“It isn’t a straightforward picture, though, as it was mainly older girls who benefited. But the fact that we saw the biggest difference among girls from areas of high deprivation is important and is a step towards reducing the health inequalities they face.”
Although the researchers found an association rather than a causal link, this study adds to previous findings that the levy was associated with a substantial reduction in the amount of sugar in soft drinks.
Senior author Professor Jean Adams from the MRC Epidemiology Unit said: “We know that consuming too many sugary drinks contributes to obesity and that the U.K. soft drinks levy led to a drop in the amount of sugar in soft drinks available in the U.K., so it makes sense that we also see a drop in cases of obesity, although we only found this in girls. Children from more deprived backgrounds tend to consume the largest amount of sugary drinks, and it was among girls in this group that we saw the biggest change.”
Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, said: “The claim that the soft drink levy might have prevented 5,000 children from becoming obese is speculative because it is based on an association not actual measurements of consumption.”
He added that: “As well as continuing to discourage the consumption of sugar sweetened beverages and sweets, wider recognition should be given to foods such as biscuits [and] deep-fried foods (crisps, corn snacks, chips) that make [a] bigger contribution to excess calorie intake in children. Tackling poverty, however, is probably [the] best way to improve the diets of socially deprived children.”
Government ‘should learn from this success’
Asked to comment by this news organization, Katharine Jenner, director of the Obesity Health Alliance, said: “Government should be heartened that their soft drinks policy is already improving the health of young girls, regardless of where they live. The government should learn from this success, especially when compared with the many unsuccessful attempts to persuade industry to change their products voluntarily. They must now press ahead with policies that make it easier for everyone to eat a healthier diet, including extending the soft drinks industry levy to include other less healthy foods and drinks and measures to take junk food out of the spotlight.
“The research notes that numerous studies have found that boys are often exposed to more food advertising content than girls, negating the impact of the soft drinks levy [so] we need restriction on junk food marketing now, to put healthy food back in the spotlight.”
The research was supported by the National Institute of Health and Care Research and the Medical Research Council.
A version of this article originally appeared on MedscapeUK.
Weight bias affects views of kids’ obesity recommendations
Apparently, offering children effective treatments for a chronic disease that markedly increases their risk for other chronic diseases, regularly erodes their quality of life, and is the No. 1 target of school-based bullying is wrong.
At least that’s my take watching the coverage of the recent American Academy of Pediatrics new pediatric obesity treatment guidelines that, gasp, suggest that children whose severity of obesity warrants medication or surgeries be offered medication or surgery. Because it’s wiser to not try to treat the obesity that›s contributing to a child’s type 2 diabetes, hypertension, fatty liver disease, or reduced quality of life?
The reaction isn’t surprising. Some of those who are up in arms about it have clinical or research careers dependent on championing their own favorite dietary strategies as if they are more effective and reproducible than decades of uniformly disappointing studies proving that they’re not. Others are upset because, for reasons that at times may be personal and at times may be conflicted, they believe that obesity should not be treated and/or that sustained weight loss is impossible. But overarchingly, probably the bulk of the hoopla stems from obesity being seen as a moral failing. Because the notion that those who suffer with obesity are themselves to blame has been the prevailing societal view for decades, if not centuries.
Working with families of children with obesity severe enough for them to seek help, it’s clear that if desire were sufficient to will it away, we wouldn’t need treatment guidelines let alone medications or surgery. Near uniformly, parents describe their children being bullied consequent to and being deeply self-conscious of their weight.
And what would those who think children shouldn’t be offered reproducibly effective treatment for obesity have them do about it? Many seem to think it would be preferable for kids to be placed on formal diets and, of course, that they should go out and play more. And though I’m all for encouraging the improvement of a child’s dietary quality and activity level, anyone suggesting those as panaceas for childhood obesity haven’t a clue. Not to mention the fact that, in most cases, improving overall dietary quality, something worthwhile at any weight, isn’t the dietary goal being recommended. Instead, the prescription seems to be restrictive dieting coupled with overexercising, which, unlike appropriately and thoughtfully informed and utilized medication, may increase a child’s risk of maladaptive thinking around food and fitness as well as disordered eating, not to mention challenge their self-esteem if their lifestyle results are underwhelming.
This brings us to one of the most bizarre takes on this whole business – that medications will be pushed and used when not necessary. No doubt that at times, that may occur, but the issue is that of a clinician’s overzealous prescribing and not of the treatment options or indications. Consider childhood asthma. There is no worry or uproar that children with mild asthma that isn’t having an impact on their quality of life or markedly risking their health will be placed on multiple inhaled steroids and treatments. Why? Because clinicians have been taught how to dispassionately evaluate treatment needs for asthma, monitor disease course, and not simply prescribe everything in our armamentarium.
Shocking, I know, but as is the case with every other medical condition, I think doctors are capable of learning and following an algorithm covering the indications and options for the treatment of childhood obesity.
How that looks also mirrors what’s seen with any other chronic noncommunicable disease with varied severity and impact. Doctors will evaluate each child with obesity to see whether it’s having a detrimental effect on their health or quality of life. They will monitor their patients’ obesity to see if it’s worsening and will, when necessary, undertake investigations to rule out its potential contribution to common comorbidities like type 2 diabetes, hypertension, and fatty liver disease. And, when appropriate, they will provide information on available treatment options – from lifestyle to medication to surgery and the risks, benefits, and realistic expectations associated with each – and then, without judgment, support their patients’ treatment choices because blame-free informed discussion and supportive prescription of care is, in fact, the distillation of our jobs.
If people are looking to be outraged rather than focusing their outrage on what we now need to do about childhood obesity, they should instead look to what got us here: our obesogenic environment. We and our children are swimming against a torrential current of cheap ultraprocessed calories being pushed upon us by a broken societal food culture that values convenience and simultaneously embraces the notion that knowledge is a match versus the thousands of genes and dozens of hormones that increasingly sophisticated food industry marketers and scientists prey upon. When dealing with torrential currents, we need to do more than just recommend swimming lessons.
Like asthma, which may be exacerbated by pollution in our environment both outdoors and indoors, childhood obesity is a modern-day environmentally influenced disease with varied penetrance that does not always require active treatment. Like asthma, childhood obesity is not a disease that children choose to have; it’s not a disease that can be willed away; and it’s not a disease that responds uniformly, dramatically, or enduringly to diet and exercise. Finally, literally and figuratively, like asthma, for childhood obesity, we thankfully now have a number of effective treatment options that we can offer, and it’s only our societal weight bias that leads to thinking that’s anything but great.
A version of this article first appeared on Medscape.com.
Apparently, offering children effective treatments for a chronic disease that markedly increases their risk for other chronic diseases, regularly erodes their quality of life, and is the No. 1 target of school-based bullying is wrong.
At least that’s my take watching the coverage of the recent American Academy of Pediatrics new pediatric obesity treatment guidelines that, gasp, suggest that children whose severity of obesity warrants medication or surgeries be offered medication or surgery. Because it’s wiser to not try to treat the obesity that›s contributing to a child’s type 2 diabetes, hypertension, fatty liver disease, or reduced quality of life?
The reaction isn’t surprising. Some of those who are up in arms about it have clinical or research careers dependent on championing their own favorite dietary strategies as if they are more effective and reproducible than decades of uniformly disappointing studies proving that they’re not. Others are upset because, for reasons that at times may be personal and at times may be conflicted, they believe that obesity should not be treated and/or that sustained weight loss is impossible. But overarchingly, probably the bulk of the hoopla stems from obesity being seen as a moral failing. Because the notion that those who suffer with obesity are themselves to blame has been the prevailing societal view for decades, if not centuries.
Working with families of children with obesity severe enough for them to seek help, it’s clear that if desire were sufficient to will it away, we wouldn’t need treatment guidelines let alone medications or surgery. Near uniformly, parents describe their children being bullied consequent to and being deeply self-conscious of their weight.
And what would those who think children shouldn’t be offered reproducibly effective treatment for obesity have them do about it? Many seem to think it would be preferable for kids to be placed on formal diets and, of course, that they should go out and play more. And though I’m all for encouraging the improvement of a child’s dietary quality and activity level, anyone suggesting those as panaceas for childhood obesity haven’t a clue. Not to mention the fact that, in most cases, improving overall dietary quality, something worthwhile at any weight, isn’t the dietary goal being recommended. Instead, the prescription seems to be restrictive dieting coupled with overexercising, which, unlike appropriately and thoughtfully informed and utilized medication, may increase a child’s risk of maladaptive thinking around food and fitness as well as disordered eating, not to mention challenge their self-esteem if their lifestyle results are underwhelming.
This brings us to one of the most bizarre takes on this whole business – that medications will be pushed and used when not necessary. No doubt that at times, that may occur, but the issue is that of a clinician’s overzealous prescribing and not of the treatment options or indications. Consider childhood asthma. There is no worry or uproar that children with mild asthma that isn’t having an impact on their quality of life or markedly risking their health will be placed on multiple inhaled steroids and treatments. Why? Because clinicians have been taught how to dispassionately evaluate treatment needs for asthma, monitor disease course, and not simply prescribe everything in our armamentarium.
Shocking, I know, but as is the case with every other medical condition, I think doctors are capable of learning and following an algorithm covering the indications and options for the treatment of childhood obesity.
How that looks also mirrors what’s seen with any other chronic noncommunicable disease with varied severity and impact. Doctors will evaluate each child with obesity to see whether it’s having a detrimental effect on their health or quality of life. They will monitor their patients’ obesity to see if it’s worsening and will, when necessary, undertake investigations to rule out its potential contribution to common comorbidities like type 2 diabetes, hypertension, and fatty liver disease. And, when appropriate, they will provide information on available treatment options – from lifestyle to medication to surgery and the risks, benefits, and realistic expectations associated with each – and then, without judgment, support their patients’ treatment choices because blame-free informed discussion and supportive prescription of care is, in fact, the distillation of our jobs.
If people are looking to be outraged rather than focusing their outrage on what we now need to do about childhood obesity, they should instead look to what got us here: our obesogenic environment. We and our children are swimming against a torrential current of cheap ultraprocessed calories being pushed upon us by a broken societal food culture that values convenience and simultaneously embraces the notion that knowledge is a match versus the thousands of genes and dozens of hormones that increasingly sophisticated food industry marketers and scientists prey upon. When dealing with torrential currents, we need to do more than just recommend swimming lessons.
Like asthma, which may be exacerbated by pollution in our environment both outdoors and indoors, childhood obesity is a modern-day environmentally influenced disease with varied penetrance that does not always require active treatment. Like asthma, childhood obesity is not a disease that children choose to have; it’s not a disease that can be willed away; and it’s not a disease that responds uniformly, dramatically, or enduringly to diet and exercise. Finally, literally and figuratively, like asthma, for childhood obesity, we thankfully now have a number of effective treatment options that we can offer, and it’s only our societal weight bias that leads to thinking that’s anything but great.
A version of this article first appeared on Medscape.com.
Apparently, offering children effective treatments for a chronic disease that markedly increases their risk for other chronic diseases, regularly erodes their quality of life, and is the No. 1 target of school-based bullying is wrong.
At least that’s my take watching the coverage of the recent American Academy of Pediatrics new pediatric obesity treatment guidelines that, gasp, suggest that children whose severity of obesity warrants medication or surgeries be offered medication or surgery. Because it’s wiser to not try to treat the obesity that›s contributing to a child’s type 2 diabetes, hypertension, fatty liver disease, or reduced quality of life?
The reaction isn’t surprising. Some of those who are up in arms about it have clinical or research careers dependent on championing their own favorite dietary strategies as if they are more effective and reproducible than decades of uniformly disappointing studies proving that they’re not. Others are upset because, for reasons that at times may be personal and at times may be conflicted, they believe that obesity should not be treated and/or that sustained weight loss is impossible. But overarchingly, probably the bulk of the hoopla stems from obesity being seen as a moral failing. Because the notion that those who suffer with obesity are themselves to blame has been the prevailing societal view for decades, if not centuries.
Working with families of children with obesity severe enough for them to seek help, it’s clear that if desire were sufficient to will it away, we wouldn’t need treatment guidelines let alone medications or surgery. Near uniformly, parents describe their children being bullied consequent to and being deeply self-conscious of their weight.
And what would those who think children shouldn’t be offered reproducibly effective treatment for obesity have them do about it? Many seem to think it would be preferable for kids to be placed on formal diets and, of course, that they should go out and play more. And though I’m all for encouraging the improvement of a child’s dietary quality and activity level, anyone suggesting those as panaceas for childhood obesity haven’t a clue. Not to mention the fact that, in most cases, improving overall dietary quality, something worthwhile at any weight, isn’t the dietary goal being recommended. Instead, the prescription seems to be restrictive dieting coupled with overexercising, which, unlike appropriately and thoughtfully informed and utilized medication, may increase a child’s risk of maladaptive thinking around food and fitness as well as disordered eating, not to mention challenge their self-esteem if their lifestyle results are underwhelming.
This brings us to one of the most bizarre takes on this whole business – that medications will be pushed and used when not necessary. No doubt that at times, that may occur, but the issue is that of a clinician’s overzealous prescribing and not of the treatment options or indications. Consider childhood asthma. There is no worry or uproar that children with mild asthma that isn’t having an impact on their quality of life or markedly risking their health will be placed on multiple inhaled steroids and treatments. Why? Because clinicians have been taught how to dispassionately evaluate treatment needs for asthma, monitor disease course, and not simply prescribe everything in our armamentarium.
Shocking, I know, but as is the case with every other medical condition, I think doctors are capable of learning and following an algorithm covering the indications and options for the treatment of childhood obesity.
How that looks also mirrors what’s seen with any other chronic noncommunicable disease with varied severity and impact. Doctors will evaluate each child with obesity to see whether it’s having a detrimental effect on their health or quality of life. They will monitor their patients’ obesity to see if it’s worsening and will, when necessary, undertake investigations to rule out its potential contribution to common comorbidities like type 2 diabetes, hypertension, and fatty liver disease. And, when appropriate, they will provide information on available treatment options – from lifestyle to medication to surgery and the risks, benefits, and realistic expectations associated with each – and then, without judgment, support their patients’ treatment choices because blame-free informed discussion and supportive prescription of care is, in fact, the distillation of our jobs.
If people are looking to be outraged rather than focusing their outrage on what we now need to do about childhood obesity, they should instead look to what got us here: our obesogenic environment. We and our children are swimming against a torrential current of cheap ultraprocessed calories being pushed upon us by a broken societal food culture that values convenience and simultaneously embraces the notion that knowledge is a match versus the thousands of genes and dozens of hormones that increasingly sophisticated food industry marketers and scientists prey upon. When dealing with torrential currents, we need to do more than just recommend swimming lessons.
Like asthma, which may be exacerbated by pollution in our environment both outdoors and indoors, childhood obesity is a modern-day environmentally influenced disease with varied penetrance that does not always require active treatment. Like asthma, childhood obesity is not a disease that children choose to have; it’s not a disease that can be willed away; and it’s not a disease that responds uniformly, dramatically, or enduringly to diet and exercise. Finally, literally and figuratively, like asthma, for childhood obesity, we thankfully now have a number of effective treatment options that we can offer, and it’s only our societal weight bias that leads to thinking that’s anything but great.
A version of this article first appeared on Medscape.com.
Don’t cross the friends line with patients
All that moving can make it hard to maintain friendships. Factor in the challenges from the pandemic, and a physician’s life can be lonely. So, when a patient invites you for coffee or a game of pickleball, do you accept? For almost one-third of the physicians who responded to the Medscape Physician Friendships: The Joys and Challenges 2022, the answer might be yes.
About 29% said they develop friendships with patients. However, a lot depends on the circumstances. As one physician in the report said: “I have been a pediatrician for 35 years, and my patients have grown up and become productive adults in our small, rural, isolated area. You can’t help but know almost everyone.”
As the daughter of a cardiologist, Nishi Mehta, MD, a radiologist and founder of the largest physician-only Facebook group in the country, grew up with that small-town-everyone-knows-the-doctor model.
“When I was a kid, I’d go to the mall, and my friends and I would play a game: How long before a patient [of my dad’s] comes up to me?” she said. At the time, Dr. Mehta was embarrassed, but now she marvels that her dad knew his patients so well that they would recognize his daughter in crowded suburban mall.
In other instances, a physician may develop a friendly relationship after a patient leaves their care. For example, Leo Nissola, MD, now a full-time researcher and immunotherapy scientist in San Francisco, has stayed in touch with some of the patients he treated while at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Nissola said it was important to stay connected with the patients he had meaningful relationships with. “It becomes challenging, though, when a former patient asks for medical advice.” At that moment, “you have to be explicitly clear that the relationship has changed.”
A hard line in the sand
The blurring of lines is one reason many doctors refuse to befriend patients, even after they are no longer treating them. The American College of Physicians Ethics Manual advises against treating anyone with whom you have a close relationship, including family and friends.
“Friendships can get in the way of patients being honest with you, which can interfere with medical care,” Dr. Mehta said. “If a patient has a concern related to something they wouldn’t want you to know as friends, it can get awkward. They may elect not to tell you.”
And on the flip side, friendship can provide a view into your private life that you may not welcome in the exam room.
“Let’s say you go out for drinks [with a patient], and you’re up late, but you have surgery the next day,” said Brandi Ring, MD, an ob.gyn. and the associate medical director at the Center for Children and Women in Houston. Now, one of your patients knows you were out until midnight when you had to be in the OR at 5:00 a.m.
Worse still, your relationship could color your decisions about a patient’s care, even unconsciously. It can be hard to maintain objectivity when you have an emotional investment in someone’s well-being.
“We don’t necessarily treat family and friends to the standards of medical care,” said Dr. Ring. “We go above and beyond. We might order more tests and more scans. We don’t always follow the guidelines, especially in critical illness.”
For all these reasons and more, the ACP advises against treating friends.
Put physician before friend
But adhering to those guidelines can lead physicians to make some painful decisions. Cutting yourself off from the possibility of friendship is never easy, and the Medscape report found that physicians tend to have fewer friends than the average American.
“Especially earlier in my practice, when I was a young parent, and I would see a lot of other young parents in the same stage in life, I’d think, ‘In other circumstances, I would be hanging out at the park with this person,’ “ said Kathleen Rowland, MD, a family medicine physician and vice chair of education in the department of family medicine at Rush University, Chicago. “But the hard part is, the doctor-patient relationship always comes first.”
To a certain extent, one’s specialty may determine the feasibility of becoming friends with a patient. While Dr. Mehta has never done so, as a radiologist, she doesn’t usually see patients repeatedly. Likewise, a young gerontologist may have little in common with his octogenarian patients. And an older pediatrician is not in the same life stage as his patients’ sleep-deprived new parents, possibly making them less attractive friends.
However, practicing family medicine is all about long-term physician-patient relationships. Getting to know patients and their families over many years can lead to a certain intimacy. Dr. Rowland said that, while a wonderful part of being a physician is getting that unique trust whereby patients tell you all sorts of things about their lives, she’s never gone down the friendship path.
“There’s the assumption I’ll take care of someone for a long period of time, and their partner and their kids, maybe another generation or two,” Dr. Rowland said. “People really do rely on that relationship to contribute to their health.”
Worse, nowadays, when people may be starved for connection, many patients want to feel emotionally close and cared for by their doctor, so it’d be easy to cross the line. While patients deserve a compassionate, caring doctor, the physician is left to walk the line between those boundaries. Dr. Rowland said, “It’s up to the clinician to say: ‘My role is as a doctor. You deserve caring friends, but I have to order your mammogram and your blood counts. My role is different.’ ”
Friendly but not friends
It can be tricky to navigate the boundary between a cordial, warm relationship with a patient and that patient inviting you to their daughter’s wedding.
“People may mistake being pleasant and friendly for being friends,” said Larry Blosser, MD, chief medical officer at Central Ohio Primary Care, Westerville. In his position, he sometimes hears from patients who have misunderstood their relationship with a doctor in the practice. When that happens, he advises the physician to consider the persona they’re presenting to the patient. If you’re overly friendly, there’s the potential for confusion, but you can’t be aloof and cold, he said.
Maintaining that awareness helps to prevent a patient’s offhand invitation to catch a movie or go on a hike. And verbalizing it to your patients can make your relationship clear from the get-go.
“I tell patients we’re a team. I’m the captain, and they’re my MVP. When the match is over, whatever the results, we’re done,” said Karenne Fru, MD, PhD, a fertility specialist at Oma Fertility Atlanta. Making deep connections is essential to her practice, so Dr. Fru structures her patient interactions carefully. “Infertility is such an isolating experience. While you’re with us, we care about what’s going on in your life, your pets, and your mom’s chemo. We need mutual trust for you to be compliant with the care.”
However, that approach won’t work when you see patients regularly, as with family practice or specialties that see the same patients repeatedly throughout the year. In those circumstances, the match is never over but one in which the onus is on the physician to establish a friendly yet professional rapport without letting your self-interest, loneliness, or lack of friends interfere.
“It’s been a very difficult couple of years for a lot of us. Depending on what kind of clinical work we do, some of us took care of healthy people that got very sick or passed away,” Dr. Rowland said. “Having the chance to reconnect with people and reestablish some of that closeness, both physical and emotional, is going to be good for us.”
Just continue conveying warm, trusting compassion for your patients without blurring the friend lines.
A version of this article first appeared on Medscape.com.
All that moving can make it hard to maintain friendships. Factor in the challenges from the pandemic, and a physician’s life can be lonely. So, when a patient invites you for coffee or a game of pickleball, do you accept? For almost one-third of the physicians who responded to the Medscape Physician Friendships: The Joys and Challenges 2022, the answer might be yes.
About 29% said they develop friendships with patients. However, a lot depends on the circumstances. As one physician in the report said: “I have been a pediatrician for 35 years, and my patients have grown up and become productive adults in our small, rural, isolated area. You can’t help but know almost everyone.”
As the daughter of a cardiologist, Nishi Mehta, MD, a radiologist and founder of the largest physician-only Facebook group in the country, grew up with that small-town-everyone-knows-the-doctor model.
“When I was a kid, I’d go to the mall, and my friends and I would play a game: How long before a patient [of my dad’s] comes up to me?” she said. At the time, Dr. Mehta was embarrassed, but now she marvels that her dad knew his patients so well that they would recognize his daughter in crowded suburban mall.
In other instances, a physician may develop a friendly relationship after a patient leaves their care. For example, Leo Nissola, MD, now a full-time researcher and immunotherapy scientist in San Francisco, has stayed in touch with some of the patients he treated while at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Nissola said it was important to stay connected with the patients he had meaningful relationships with. “It becomes challenging, though, when a former patient asks for medical advice.” At that moment, “you have to be explicitly clear that the relationship has changed.”
A hard line in the sand
The blurring of lines is one reason many doctors refuse to befriend patients, even after they are no longer treating them. The American College of Physicians Ethics Manual advises against treating anyone with whom you have a close relationship, including family and friends.
“Friendships can get in the way of patients being honest with you, which can interfere with medical care,” Dr. Mehta said. “If a patient has a concern related to something they wouldn’t want you to know as friends, it can get awkward. They may elect not to tell you.”
And on the flip side, friendship can provide a view into your private life that you may not welcome in the exam room.
“Let’s say you go out for drinks [with a patient], and you’re up late, but you have surgery the next day,” said Brandi Ring, MD, an ob.gyn. and the associate medical director at the Center for Children and Women in Houston. Now, one of your patients knows you were out until midnight when you had to be in the OR at 5:00 a.m.
Worse still, your relationship could color your decisions about a patient’s care, even unconsciously. It can be hard to maintain objectivity when you have an emotional investment in someone’s well-being.
“We don’t necessarily treat family and friends to the standards of medical care,” said Dr. Ring. “We go above and beyond. We might order more tests and more scans. We don’t always follow the guidelines, especially in critical illness.”
For all these reasons and more, the ACP advises against treating friends.
Put physician before friend
But adhering to those guidelines can lead physicians to make some painful decisions. Cutting yourself off from the possibility of friendship is never easy, and the Medscape report found that physicians tend to have fewer friends than the average American.
“Especially earlier in my practice, when I was a young parent, and I would see a lot of other young parents in the same stage in life, I’d think, ‘In other circumstances, I would be hanging out at the park with this person,’ “ said Kathleen Rowland, MD, a family medicine physician and vice chair of education in the department of family medicine at Rush University, Chicago. “But the hard part is, the doctor-patient relationship always comes first.”
To a certain extent, one’s specialty may determine the feasibility of becoming friends with a patient. While Dr. Mehta has never done so, as a radiologist, she doesn’t usually see patients repeatedly. Likewise, a young gerontologist may have little in common with his octogenarian patients. And an older pediatrician is not in the same life stage as his patients’ sleep-deprived new parents, possibly making them less attractive friends.
However, practicing family medicine is all about long-term physician-patient relationships. Getting to know patients and their families over many years can lead to a certain intimacy. Dr. Rowland said that, while a wonderful part of being a physician is getting that unique trust whereby patients tell you all sorts of things about their lives, she’s never gone down the friendship path.
“There’s the assumption I’ll take care of someone for a long period of time, and their partner and their kids, maybe another generation or two,” Dr. Rowland said. “People really do rely on that relationship to contribute to their health.”
Worse, nowadays, when people may be starved for connection, many patients want to feel emotionally close and cared for by their doctor, so it’d be easy to cross the line. While patients deserve a compassionate, caring doctor, the physician is left to walk the line between those boundaries. Dr. Rowland said, “It’s up to the clinician to say: ‘My role is as a doctor. You deserve caring friends, but I have to order your mammogram and your blood counts. My role is different.’ ”
Friendly but not friends
It can be tricky to navigate the boundary between a cordial, warm relationship with a patient and that patient inviting you to their daughter’s wedding.
“People may mistake being pleasant and friendly for being friends,” said Larry Blosser, MD, chief medical officer at Central Ohio Primary Care, Westerville. In his position, he sometimes hears from patients who have misunderstood their relationship with a doctor in the practice. When that happens, he advises the physician to consider the persona they’re presenting to the patient. If you’re overly friendly, there’s the potential for confusion, but you can’t be aloof and cold, he said.
Maintaining that awareness helps to prevent a patient’s offhand invitation to catch a movie or go on a hike. And verbalizing it to your patients can make your relationship clear from the get-go.
“I tell patients we’re a team. I’m the captain, and they’re my MVP. When the match is over, whatever the results, we’re done,” said Karenne Fru, MD, PhD, a fertility specialist at Oma Fertility Atlanta. Making deep connections is essential to her practice, so Dr. Fru structures her patient interactions carefully. “Infertility is such an isolating experience. While you’re with us, we care about what’s going on in your life, your pets, and your mom’s chemo. We need mutual trust for you to be compliant with the care.”
However, that approach won’t work when you see patients regularly, as with family practice or specialties that see the same patients repeatedly throughout the year. In those circumstances, the match is never over but one in which the onus is on the physician to establish a friendly yet professional rapport without letting your self-interest, loneliness, or lack of friends interfere.
“It’s been a very difficult couple of years for a lot of us. Depending on what kind of clinical work we do, some of us took care of healthy people that got very sick or passed away,” Dr. Rowland said. “Having the chance to reconnect with people and reestablish some of that closeness, both physical and emotional, is going to be good for us.”
Just continue conveying warm, trusting compassion for your patients without blurring the friend lines.
A version of this article first appeared on Medscape.com.
All that moving can make it hard to maintain friendships. Factor in the challenges from the pandemic, and a physician’s life can be lonely. So, when a patient invites you for coffee or a game of pickleball, do you accept? For almost one-third of the physicians who responded to the Medscape Physician Friendships: The Joys and Challenges 2022, the answer might be yes.
About 29% said they develop friendships with patients. However, a lot depends on the circumstances. As one physician in the report said: “I have been a pediatrician for 35 years, and my patients have grown up and become productive adults in our small, rural, isolated area. You can’t help but know almost everyone.”
As the daughter of a cardiologist, Nishi Mehta, MD, a radiologist and founder of the largest physician-only Facebook group in the country, grew up with that small-town-everyone-knows-the-doctor model.
“When I was a kid, I’d go to the mall, and my friends and I would play a game: How long before a patient [of my dad’s] comes up to me?” she said. At the time, Dr. Mehta was embarrassed, but now she marvels that her dad knew his patients so well that they would recognize his daughter in crowded suburban mall.
In other instances, a physician may develop a friendly relationship after a patient leaves their care. For example, Leo Nissola, MD, now a full-time researcher and immunotherapy scientist in San Francisco, has stayed in touch with some of the patients he treated while at the University of Texas MD Anderson Cancer Center, Houston.
Dr. Nissola said it was important to stay connected with the patients he had meaningful relationships with. “It becomes challenging, though, when a former patient asks for medical advice.” At that moment, “you have to be explicitly clear that the relationship has changed.”
A hard line in the sand
The blurring of lines is one reason many doctors refuse to befriend patients, even after they are no longer treating them. The American College of Physicians Ethics Manual advises against treating anyone with whom you have a close relationship, including family and friends.
“Friendships can get in the way of patients being honest with you, which can interfere with medical care,” Dr. Mehta said. “If a patient has a concern related to something they wouldn’t want you to know as friends, it can get awkward. They may elect not to tell you.”
And on the flip side, friendship can provide a view into your private life that you may not welcome in the exam room.
“Let’s say you go out for drinks [with a patient], and you’re up late, but you have surgery the next day,” said Brandi Ring, MD, an ob.gyn. and the associate medical director at the Center for Children and Women in Houston. Now, one of your patients knows you were out until midnight when you had to be in the OR at 5:00 a.m.
Worse still, your relationship could color your decisions about a patient’s care, even unconsciously. It can be hard to maintain objectivity when you have an emotional investment in someone’s well-being.
“We don’t necessarily treat family and friends to the standards of medical care,” said Dr. Ring. “We go above and beyond. We might order more tests and more scans. We don’t always follow the guidelines, especially in critical illness.”
For all these reasons and more, the ACP advises against treating friends.
Put physician before friend
But adhering to those guidelines can lead physicians to make some painful decisions. Cutting yourself off from the possibility of friendship is never easy, and the Medscape report found that physicians tend to have fewer friends than the average American.
“Especially earlier in my practice, when I was a young parent, and I would see a lot of other young parents in the same stage in life, I’d think, ‘In other circumstances, I would be hanging out at the park with this person,’ “ said Kathleen Rowland, MD, a family medicine physician and vice chair of education in the department of family medicine at Rush University, Chicago. “But the hard part is, the doctor-patient relationship always comes first.”
To a certain extent, one’s specialty may determine the feasibility of becoming friends with a patient. While Dr. Mehta has never done so, as a radiologist, she doesn’t usually see patients repeatedly. Likewise, a young gerontologist may have little in common with his octogenarian patients. And an older pediatrician is not in the same life stage as his patients’ sleep-deprived new parents, possibly making them less attractive friends.
However, practicing family medicine is all about long-term physician-patient relationships. Getting to know patients and their families over many years can lead to a certain intimacy. Dr. Rowland said that, while a wonderful part of being a physician is getting that unique trust whereby patients tell you all sorts of things about their lives, she’s never gone down the friendship path.
“There’s the assumption I’ll take care of someone for a long period of time, and their partner and their kids, maybe another generation or two,” Dr. Rowland said. “People really do rely on that relationship to contribute to their health.”
Worse, nowadays, when people may be starved for connection, many patients want to feel emotionally close and cared for by their doctor, so it’d be easy to cross the line. While patients deserve a compassionate, caring doctor, the physician is left to walk the line between those boundaries. Dr. Rowland said, “It’s up to the clinician to say: ‘My role is as a doctor. You deserve caring friends, but I have to order your mammogram and your blood counts. My role is different.’ ”
Friendly but not friends
It can be tricky to navigate the boundary between a cordial, warm relationship with a patient and that patient inviting you to their daughter’s wedding.
“People may mistake being pleasant and friendly for being friends,” said Larry Blosser, MD, chief medical officer at Central Ohio Primary Care, Westerville. In his position, he sometimes hears from patients who have misunderstood their relationship with a doctor in the practice. When that happens, he advises the physician to consider the persona they’re presenting to the patient. If you’re overly friendly, there’s the potential for confusion, but you can’t be aloof and cold, he said.
Maintaining that awareness helps to prevent a patient’s offhand invitation to catch a movie or go on a hike. And verbalizing it to your patients can make your relationship clear from the get-go.
“I tell patients we’re a team. I’m the captain, and they’re my MVP. When the match is over, whatever the results, we’re done,” said Karenne Fru, MD, PhD, a fertility specialist at Oma Fertility Atlanta. Making deep connections is essential to her practice, so Dr. Fru structures her patient interactions carefully. “Infertility is such an isolating experience. While you’re with us, we care about what’s going on in your life, your pets, and your mom’s chemo. We need mutual trust for you to be compliant with the care.”
However, that approach won’t work when you see patients regularly, as with family practice or specialties that see the same patients repeatedly throughout the year. In those circumstances, the match is never over but one in which the onus is on the physician to establish a friendly yet professional rapport without letting your self-interest, loneliness, or lack of friends interfere.
“It’s been a very difficult couple of years for a lot of us. Depending on what kind of clinical work we do, some of us took care of healthy people that got very sick or passed away,” Dr. Rowland said. “Having the chance to reconnect with people and reestablish some of that closeness, both physical and emotional, is going to be good for us.”
Just continue conveying warm, trusting compassion for your patients without blurring the friend lines.
A version of this article first appeared on Medscape.com.
More type 2 diabetes deaths from cancer than heart disease
Cancer appears to have overtaken cardiovascular disease (CVD) as a leading cause of death in adults with type 2 diabetes, a 20-year population study in England suggests.
The researchers found that, from 1998 to 2018, in more than 130,000 adults aged 35 and older with type 2 diabetes, all-cause mortality declined for all ages, but cancer mortality increased for those aged 75 and older; people with type 2 diabetes who were smokers had higher and steadily increasing cancer mortality rates; and people with type 2 diabetes had more than twice the rate of colorectal, pancreatic, liver, and endometrial cancer mortality than age- and sex-matched individuals in the general population.
The findings suggest that “cancer prevention strategies therefore deserve at least a similar level of attention as cardiovascular disease prevention, particularly in older people and for some cancers such as liver, colorectal, and pancreatic cancer,” the researchers wrote.
Tailored cancer prevention and early-detection strategies are needed to address persistent inequalities in the older population, the most deprived, and smokers, they added.
Breast cancer rates in younger women with type 2 diabetes rising
According to the researchers, “early cancer detection through changes to existing screening [programs], or more in-depth investigations for suspected/nonspecific symptoms, may reduce the number of avoidable cancer deaths in people with type 2 diabetes.”
Moreover, breast cancer rates in younger women with type 2 diabetes are rising by 4.1% per year, they wrote, which suggests such women are high risk and should be screened at a younger age, but screening age would need to be determined in cost-effectiveness analyses.
The study by Suping Ling, PhD, and colleagues was published online in Diabetologia.
Results challenge belief that preventing CVD is priority in type 2 diabetes
“The prevention of cardiovascular disease has been, and is still considered, a priority in people with diabetes,” the researchers wrote.
“Our results challenge this view by showing that cancer may have overtaken cardiovascular disease as a leading cause of death in people with type 2 diabetes.”
“The proportion of cancer deaths out of all-cause deaths remains high (> 30%) in young ages, and it was steadily increasing in older ages,” Dr. Ling, from the department of noncommunicable disease epidemiology, London School of Hygiene & Tropical Medicine, said in a comment.
“Combined with previous studies reporting decreasing CVD mortality rates,” she said, “we concluded that cancer might have overtaken CVD as the leading cause of death in people with type 2 diabetes.”
Many evidence-based cancer-prevention strategies related to lifestyle (such as being physically active, being a healthy weight, eating a better diet, stopping smoking, as summarized by the World Cancer Research Fund), are helpful for preventing both cancer and CVD, Ling observed.
However, in the medical community, many additional efforts were made for monitoring, early detection, and innovating medications for CVD, she noted. “Therefore, we would like to propose a similar level of attention and effort for cancer in people with type 2 diabetes.”
Deaths from cancer vs. all causes in patients with diabetes
The researchers identified 137,804 patients aged 35 and older who were newly diagnosed with type 2 diabetes from 1998 to 2018 in general practices in the UK that were part of the Clinical Practice Research Datalink.
Patients were a median age of 64 years and 45% were women. Most (83%) were White, followed by South Asian (3.5%), Black (2.0%), and other (3%); 8.4% had missing information for race. Patients had a median body mass index (BMI) of 30.6 kg/m2.
Researchers divided patients into socioeconomic quintiles of most to least deprived based on income, employment, education, and other factors. During a median follow-up of 8.4 years, there were 39,212 deaths (28.5%).
Cancer mortality in subgroups of patients with type 2 diabetes
Researchers analyzed annual deaths from cancer and from all causes over 20 years in subgroups of patients with type 2 diabetes.
In adults with type 2 diabetes, the average percentage change in cancer mortality per year, from 1998 to 2018 decreased in people aged 55 and 65 (–1.4% and –0.2%, respectively), but increased in people aged 75 and 85 (1.2% and 1.6%, respectively); increased more in women than in men (1.5% vs 1.0%), although women had lower cancer mortality than men; and increased more in the least deprived (wealthiest) individuals than in the most deprived (1.5% vs 1.0%). Cancer mortality rates were consistently higher in the most deprived individuals, Dr. Ling noted.
Cancer mortality also increased more in people with class III obesity (BMI ≥ 35) versus normal weight (5.8% vs 0.7%) and versus other weights. In addition, there was an upward trend in cancer mortality in people who were White or former/current smokers.
Deaths from specific cancers in diabetes vs. general population
Next, researchers determined cancer mortality ratios – the cancer mortality of the patients with diabetes divided by the cancer mortality of the general population.
They determined this for all cancers, the four most common cancers in the United Kingdom (lung, colorectal, breast, and prostate), and cancers caused by type 2 diabetes (pancreatic, liver, gallbladder, and endometrial cancer), standardized by sex and age.
Mortality from all cancer was 18% higher in patients with type 2 diabetes, compared with the general population.
Overall, mortality from colorectal cancer, pancreatic cancer, and liver cancer was 2.4 times, 2.12 times, and 2.13 times higher, respectively, in patients with type 2 diabetes than in the general population.
Mortality from breast cancer was 9% higher and mortality from endometrial cancer was 2.08 times higher in women with type 2 diabetes than in women in the general population.
There was a constant upward trend for mortality rates for pancreatic, liver, and lung cancer at all ages, colorectal cancer at most ages, breast cancer at younger ages, and prostate and endometrial cancer at older ages.
The study was funded by Hope Against Cancer. Dr. Ling reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cancer appears to have overtaken cardiovascular disease (CVD) as a leading cause of death in adults with type 2 diabetes, a 20-year population study in England suggests.
The researchers found that, from 1998 to 2018, in more than 130,000 adults aged 35 and older with type 2 diabetes, all-cause mortality declined for all ages, but cancer mortality increased for those aged 75 and older; people with type 2 diabetes who were smokers had higher and steadily increasing cancer mortality rates; and people with type 2 diabetes had more than twice the rate of colorectal, pancreatic, liver, and endometrial cancer mortality than age- and sex-matched individuals in the general population.
The findings suggest that “cancer prevention strategies therefore deserve at least a similar level of attention as cardiovascular disease prevention, particularly in older people and for some cancers such as liver, colorectal, and pancreatic cancer,” the researchers wrote.
Tailored cancer prevention and early-detection strategies are needed to address persistent inequalities in the older population, the most deprived, and smokers, they added.
Breast cancer rates in younger women with type 2 diabetes rising
According to the researchers, “early cancer detection through changes to existing screening [programs], or more in-depth investigations for suspected/nonspecific symptoms, may reduce the number of avoidable cancer deaths in people with type 2 diabetes.”
Moreover, breast cancer rates in younger women with type 2 diabetes are rising by 4.1% per year, they wrote, which suggests such women are high risk and should be screened at a younger age, but screening age would need to be determined in cost-effectiveness analyses.
The study by Suping Ling, PhD, and colleagues was published online in Diabetologia.
Results challenge belief that preventing CVD is priority in type 2 diabetes
“The prevention of cardiovascular disease has been, and is still considered, a priority in people with diabetes,” the researchers wrote.
“Our results challenge this view by showing that cancer may have overtaken cardiovascular disease as a leading cause of death in people with type 2 diabetes.”
“The proportion of cancer deaths out of all-cause deaths remains high (> 30%) in young ages, and it was steadily increasing in older ages,” Dr. Ling, from the department of noncommunicable disease epidemiology, London School of Hygiene & Tropical Medicine, said in a comment.
“Combined with previous studies reporting decreasing CVD mortality rates,” she said, “we concluded that cancer might have overtaken CVD as the leading cause of death in people with type 2 diabetes.”
Many evidence-based cancer-prevention strategies related to lifestyle (such as being physically active, being a healthy weight, eating a better diet, stopping smoking, as summarized by the World Cancer Research Fund), are helpful for preventing both cancer and CVD, Ling observed.
However, in the medical community, many additional efforts were made for monitoring, early detection, and innovating medications for CVD, she noted. “Therefore, we would like to propose a similar level of attention and effort for cancer in people with type 2 diabetes.”
Deaths from cancer vs. all causes in patients with diabetes
The researchers identified 137,804 patients aged 35 and older who were newly diagnosed with type 2 diabetes from 1998 to 2018 in general practices in the UK that were part of the Clinical Practice Research Datalink.
Patients were a median age of 64 years and 45% were women. Most (83%) were White, followed by South Asian (3.5%), Black (2.0%), and other (3%); 8.4% had missing information for race. Patients had a median body mass index (BMI) of 30.6 kg/m2.
Researchers divided patients into socioeconomic quintiles of most to least deprived based on income, employment, education, and other factors. During a median follow-up of 8.4 years, there were 39,212 deaths (28.5%).
Cancer mortality in subgroups of patients with type 2 diabetes
Researchers analyzed annual deaths from cancer and from all causes over 20 years in subgroups of patients with type 2 diabetes.

In adults with type 2 diabetes, the average percentage change in cancer mortality per year, from 1998 to 2018 decreased in people aged 55 and 65 (–1.4% and –0.2%, respectively), but increased in people aged 75 and 85 (1.2% and 1.6%, respectively); increased more in women than in men (1.5% vs 1.0%), although women had lower cancer mortality than men; and increased more in the least deprived (wealthiest) individuals than in the most deprived (1.5% vs 1.0%). Cancer mortality rates were consistently higher in the most deprived individuals, Dr. Ling noted.
Cancer mortality also increased more in people with class III obesity (BMI ≥ 35) versus normal weight (5.8% vs 0.7%) and versus other weights. In addition, there was an upward trend in cancer mortality in people who were White or former/current smokers.
Deaths from specific cancers in diabetes vs. general population
Next, researchers determined cancer mortality ratios – the cancer mortality of the patients with diabetes divided by the cancer mortality of the general population.
They determined this for all cancers, the four most common cancers in the United Kingdom (lung, colorectal, breast, and prostate), and cancers caused by type 2 diabetes (pancreatic, liver, gallbladder, and endometrial cancer), standardized by sex and age.
Mortality from all cancer was 18% higher in patients with type 2 diabetes, compared with the general population.
Overall, mortality from colorectal cancer, pancreatic cancer, and liver cancer was 2.4 times, 2.12 times, and 2.13 times higher, respectively, in patients with type 2 diabetes than in the general population.
Mortality from breast cancer was 9% higher and mortality from endometrial cancer was 2.08 times higher in women with type 2 diabetes than in women in the general population.
There was a constant upward trend for mortality rates for pancreatic, liver, and lung cancer at all ages, colorectal cancer at most ages, breast cancer at younger ages, and prostate and endometrial cancer at older ages.
The study was funded by Hope Against Cancer. Dr. Ling reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cancer appears to have overtaken cardiovascular disease (CVD) as a leading cause of death in adults with type 2 diabetes, a 20-year population study in England suggests.
The researchers found that, from 1998 to 2018, in more than 130,000 adults aged 35 and older with type 2 diabetes, all-cause mortality declined for all ages, but cancer mortality increased for those aged 75 and older; people with type 2 diabetes who were smokers had higher and steadily increasing cancer mortality rates; and people with type 2 diabetes had more than twice the rate of colorectal, pancreatic, liver, and endometrial cancer mortality than age- and sex-matched individuals in the general population.
The findings suggest that “cancer prevention strategies therefore deserve at least a similar level of attention as cardiovascular disease prevention, particularly in older people and for some cancers such as liver, colorectal, and pancreatic cancer,” the researchers wrote.
Tailored cancer prevention and early-detection strategies are needed to address persistent inequalities in the older population, the most deprived, and smokers, they added.
Breast cancer rates in younger women with type 2 diabetes rising
According to the researchers, “early cancer detection through changes to existing screening [programs], or more in-depth investigations for suspected/nonspecific symptoms, may reduce the number of avoidable cancer deaths in people with type 2 diabetes.”
Moreover, breast cancer rates in younger women with type 2 diabetes are rising by 4.1% per year, they wrote, which suggests such women are high risk and should be screened at a younger age, but screening age would need to be determined in cost-effectiveness analyses.
The study by Suping Ling, PhD, and colleagues was published online in Diabetologia.
Results challenge belief that preventing CVD is priority in type 2 diabetes
“The prevention of cardiovascular disease has been, and is still considered, a priority in people with diabetes,” the researchers wrote.
“Our results challenge this view by showing that cancer may have overtaken cardiovascular disease as a leading cause of death in people with type 2 diabetes.”
“The proportion of cancer deaths out of all-cause deaths remains high (> 30%) in young ages, and it was steadily increasing in older ages,” Dr. Ling, from the department of noncommunicable disease epidemiology, London School of Hygiene & Tropical Medicine, said in a comment.
“Combined with previous studies reporting decreasing CVD mortality rates,” she said, “we concluded that cancer might have overtaken CVD as the leading cause of death in people with type 2 diabetes.”
Many evidence-based cancer-prevention strategies related to lifestyle (such as being physically active, being a healthy weight, eating a better diet, stopping smoking, as summarized by the World Cancer Research Fund), are helpful for preventing both cancer and CVD, Ling observed.
However, in the medical community, many additional efforts were made for monitoring, early detection, and innovating medications for CVD, she noted. “Therefore, we would like to propose a similar level of attention and effort for cancer in people with type 2 diabetes.”
Deaths from cancer vs. all causes in patients with diabetes
The researchers identified 137,804 patients aged 35 and older who were newly diagnosed with type 2 diabetes from 1998 to 2018 in general practices in the UK that were part of the Clinical Practice Research Datalink.
Patients were a median age of 64 years and 45% were women. Most (83%) were White, followed by South Asian (3.5%), Black (2.0%), and other (3%); 8.4% had missing information for race. Patients had a median body mass index (BMI) of 30.6 kg/m2.
Researchers divided patients into socioeconomic quintiles of most to least deprived based on income, employment, education, and other factors. During a median follow-up of 8.4 years, there were 39,212 deaths (28.5%).
Cancer mortality in subgroups of patients with type 2 diabetes
Researchers analyzed annual deaths from cancer and from all causes over 20 years in subgroups of patients with type 2 diabetes.

In adults with type 2 diabetes, the average percentage change in cancer mortality per year, from 1998 to 2018 decreased in people aged 55 and 65 (–1.4% and –0.2%, respectively), but increased in people aged 75 and 85 (1.2% and 1.6%, respectively); increased more in women than in men (1.5% vs 1.0%), although women had lower cancer mortality than men; and increased more in the least deprived (wealthiest) individuals than in the most deprived (1.5% vs 1.0%). Cancer mortality rates were consistently higher in the most deprived individuals, Dr. Ling noted.
Cancer mortality also increased more in people with class III obesity (BMI ≥ 35) versus normal weight (5.8% vs 0.7%) and versus other weights. In addition, there was an upward trend in cancer mortality in people who were White or former/current smokers.
Deaths from specific cancers in diabetes vs. general population
Next, researchers determined cancer mortality ratios – the cancer mortality of the patients with diabetes divided by the cancer mortality of the general population.
They determined this for all cancers, the four most common cancers in the United Kingdom (lung, colorectal, breast, and prostate), and cancers caused by type 2 diabetes (pancreatic, liver, gallbladder, and endometrial cancer), standardized by sex and age.
Mortality from all cancer was 18% higher in patients with type 2 diabetes, compared with the general population.
Overall, mortality from colorectal cancer, pancreatic cancer, and liver cancer was 2.4 times, 2.12 times, and 2.13 times higher, respectively, in patients with type 2 diabetes than in the general population.
Mortality from breast cancer was 9% higher and mortality from endometrial cancer was 2.08 times higher in women with type 2 diabetes than in women in the general population.
There was a constant upward trend for mortality rates for pancreatic, liver, and lung cancer at all ages, colorectal cancer at most ages, breast cancer at younger ages, and prostate and endometrial cancer at older ages.
The study was funded by Hope Against Cancer. Dr. Ling reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DIABETOLOGIA
High HDL-C levels linked to increased fracture risk
High levels of high-density lipoprotein cholesterol (HDL-C) in older adults are associated with a higher risk of sustaining a fracture than lower HDL-C levels, a new study suggests.
“Two animal studies showing that HDL-C reduces bone mineral density by reducing osteoblast number and function provide a plausible explanation for why high HDL-C may increase the risk of fractures,” Monira Hussain, MBBS, MPH, PhD, of Monash University in Melbourne, told this news organization. “So, it was not surprising when our analyses provided evidence that amongst those in the highest quintile of HDL-C (> 74 mg/dL), there was a [33%] increased risk of fractures.”
After adjustment, one standard deviation increment in HDL-C level was associated with a 14% higher risk of fracture during a 4-year follow-up.
Based on this and other studies, Dr. Hussain said, “I believe that the finding of a very high HDL-C [should] alert clinicians to a higher risk of mortality, fractures, and possibly other threats to their patient’s health.”
The study was published online in JAMA Cardiology.
Independent risk factor
For this report, the researchers conducted a post hoc analysis of data from the Aspirin in Reducing Events in the Elderly (ASPREE) clinical trial and the ASPREE-Fracture substudy.
ASPREE was a double-blind, randomized, placebo-controlled primary prevention trial of aspirin. Participants were 16,703 community-dwelling Australians and 2,411 individuals from the United States with a mean age of 75 and without evident cardiovascular disease, dementia, physical disability, or life-limiting chronic illness.
The ASPREE-Fracture substudy collected data on fractures reported post randomization from the Australian participants. Fractures were confirmed by imaging and adjudicated by an expert panel and included both traumatic and minimal trauma fractures.
Of the 16,262 participants who had a plasma HDL-C measurement at baseline (55% women), 1,659 (10.2%) experienced at least one fracture over a median of 4 years. This included 711 minimal trauma fractures (for example, falls from standing height) and 948 other trauma fractures, mainly falls on stairs, ladders, or stools.
Higher rates of fractures occurred in the highest quintile of HDL-C level where the mean level was 89 mg/dL. At baseline, participants in that quintile had a lower BMI, a high prevalence of current/former smoking and current alcohol use, 12 years or longer of school, more physical activity, and higher use of antiosteoporosis medication. They also had less chronic kidney disease, diabetes, prefrailty/frailty, or treatment with lipid-lowering drugs.
In a fully adjusted model, each standard deviation increment in HDL-C level was associated with a 14% higher risk of fractures (hazard ratio, 1.14). When analyzed in quintiles, compared with participants in Q1, those in Q5 had a 33% higher risk for fracture (HR, 1.33).
Prevalence rates were similar between the sexes. The increase in fracture risk appeared to be independent of traditional risk factors for fractures, including age, sex, physical activity, alcohol use, frailty, BMI, smoking status, diabetes, chronic kidney disease, use of lipid-lowering or antiosteoporosis drugs, and education, the authors note.
The results persisted in sensitivity analyses in restricted subgroups of interest and in stratified analyses – including, for example, only minimal fractures; participants not taking antiosteoporosis drugs or statins; never smokers; nondrinkers; and those engaging in minimal physical activity (walking less than 30 minutes per day).
No association was observed between non–HDL-C levels and fractures.
The authors conclude that the study “provides robust evidence that higher levels of HDL-C are associated with incident fractures in both male and female individuals, independent of conventional risk factors.”
Clinically useful?
Commenting on the study for this news organization, Marilyn Tan, MD, clinic chief of the Endocrine Clinic and clinical associate professor of medicine at Stanford (Calif.) University, said, “I certainly would not recommend anyone do anything to actively lower their HDL levels. HDL levels are largely determined by genetics, diet, and lifestyle, with some effects from certain medications/supplements. Studies have demonstrated that moderately higher HDL levels may be protective for atherosclerosis.”
In the current study, she said, “Causation has not been proven, and importantly there is no evidence that reducing HDL levels reduces fracture risk. Also, this association between raised HDL levels and fracture risk has not been demonstrated consistently in other studies.”
Furthermore, she noted, the preclinical trials on which the authors based their hypothesis – that is, an association between HDL and a reduction in the number and function of osteoblasts – “has not been demonstrated widely in human subjects.”
“We have a large armamentarium of FDA-approved treatments for osteoporosis that have been clinically proven to reduce fracture risk very significantly, and these are the tools [in addition to lifestyle changes] we should use to reduce fracture risk,” Dr. Tan concluded.
John Wilkins, MD, of Northwestern University, Chicago, and Anand Rohatgi, MD, MSCS, of UT Southwestern Medical Center, Dallas, also point out some limitations of the study in a related editorial.
They note the inclusion of predominantly healthy adults with a mean age of 75, a population that could yield different findings from middle-aged cohorts with chronic illnesses, as well as a lack of clarity regarding the possible role of alcohol intake among the study participants.
Furthermore, the editorialists write, although significant associations were shown in this study, “models were not adjusted for detailed measures of exercise/activity, triglycerides, or any other lipids, including other HDL compositional measures such as HDL-P or ApoA-I levels. There was no assessment of whether HDL-C improved discrimination, reclassification, or any other validated measures of risk prediction performance.
“Taken together,” they conclude, “this study alone leaves several unanswered questions as to whether high HDL-C could be a useful biomarker to detect fracture risk.”
No commercial funding was disclosed. The authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
High levels of high-density lipoprotein cholesterol (HDL-C) in older adults are associated with a higher risk of sustaining a fracture than lower HDL-C levels, a new study suggests.
“Two animal studies showing that HDL-C reduces bone mineral density by reducing osteoblast number and function provide a plausible explanation for why high HDL-C may increase the risk of fractures,” Monira Hussain, MBBS, MPH, PhD, of Monash University in Melbourne, told this news organization. “So, it was not surprising when our analyses provided evidence that amongst those in the highest quintile of HDL-C (> 74 mg/dL), there was a [33%] increased risk of fractures.”
After adjustment, one standard deviation increment in HDL-C level was associated with a 14% higher risk of fracture during a 4-year follow-up.
Based on this and other studies, Dr. Hussain said, “I believe that the finding of a very high HDL-C [should] alert clinicians to a higher risk of mortality, fractures, and possibly other threats to their patient’s health.”
The study was published online in JAMA Cardiology.
Independent risk factor
For this report, the researchers conducted a post hoc analysis of data from the Aspirin in Reducing Events in the Elderly (ASPREE) clinical trial and the ASPREE-Fracture substudy.
ASPREE was a double-blind, randomized, placebo-controlled primary prevention trial of aspirin. Participants were 16,703 community-dwelling Australians and 2,411 individuals from the United States with a mean age of 75 and without evident cardiovascular disease, dementia, physical disability, or life-limiting chronic illness.
The ASPREE-Fracture substudy collected data on fractures reported post randomization from the Australian participants. Fractures were confirmed by imaging and adjudicated by an expert panel and included both traumatic and minimal trauma fractures.
Of the 16,262 participants who had a plasma HDL-C measurement at baseline (55% women), 1,659 (10.2%) experienced at least one fracture over a median of 4 years. This included 711 minimal trauma fractures (for example, falls from standing height) and 948 other trauma fractures, mainly falls on stairs, ladders, or stools.
Higher rates of fractures occurred in the highest quintile of HDL-C level where the mean level was 89 mg/dL. At baseline, participants in that quintile had a lower BMI, a high prevalence of current/former smoking and current alcohol use, 12 years or longer of school, more physical activity, and higher use of antiosteoporosis medication. They also had less chronic kidney disease, diabetes, prefrailty/frailty, or treatment with lipid-lowering drugs.
In a fully adjusted model, each standard deviation increment in HDL-C level was associated with a 14% higher risk of fractures (hazard ratio, 1.14). When analyzed in quintiles, compared with participants in Q1, those in Q5 had a 33% higher risk for fracture (HR, 1.33).
Prevalence rates were similar between the sexes. The increase in fracture risk appeared to be independent of traditional risk factors for fractures, including age, sex, physical activity, alcohol use, frailty, BMI, smoking status, diabetes, chronic kidney disease, use of lipid-lowering or antiosteoporosis drugs, and education, the authors note.
The results persisted in sensitivity analyses in restricted subgroups of interest and in stratified analyses – including, for example, only minimal fractures; participants not taking antiosteoporosis drugs or statins; never smokers; nondrinkers; and those engaging in minimal physical activity (walking less than 30 minutes per day).
No association was observed between non–HDL-C levels and fractures.
The authors conclude that the study “provides robust evidence that higher levels of HDL-C are associated with incident fractures in both male and female individuals, independent of conventional risk factors.”
Clinically useful?
Commenting on the study for this news organization, Marilyn Tan, MD, clinic chief of the Endocrine Clinic and clinical associate professor of medicine at Stanford (Calif.) University, said, “I certainly would not recommend anyone do anything to actively lower their HDL levels. HDL levels are largely determined by genetics, diet, and lifestyle, with some effects from certain medications/supplements. Studies have demonstrated that moderately higher HDL levels may be protective for atherosclerosis.”
In the current study, she said, “Causation has not been proven, and importantly there is no evidence that reducing HDL levels reduces fracture risk. Also, this association between raised HDL levels and fracture risk has not been demonstrated consistently in other studies.”
Furthermore, she noted, the preclinical trials on which the authors based their hypothesis – that is, an association between HDL and a reduction in the number and function of osteoblasts – “has not been demonstrated widely in human subjects.”
“We have a large armamentarium of FDA-approved treatments for osteoporosis that have been clinically proven to reduce fracture risk very significantly, and these are the tools [in addition to lifestyle changes] we should use to reduce fracture risk,” Dr. Tan concluded.
John Wilkins, MD, of Northwestern University, Chicago, and Anand Rohatgi, MD, MSCS, of UT Southwestern Medical Center, Dallas, also point out some limitations of the study in a related editorial.
They note the inclusion of predominantly healthy adults with a mean age of 75, a population that could yield different findings from middle-aged cohorts with chronic illnesses, as well as a lack of clarity regarding the possible role of alcohol intake among the study participants.
Furthermore, the editorialists write, although significant associations were shown in this study, “models were not adjusted for detailed measures of exercise/activity, triglycerides, or any other lipids, including other HDL compositional measures such as HDL-P or ApoA-I levels. There was no assessment of whether HDL-C improved discrimination, reclassification, or any other validated measures of risk prediction performance.
“Taken together,” they conclude, “this study alone leaves several unanswered questions as to whether high HDL-C could be a useful biomarker to detect fracture risk.”
No commercial funding was disclosed. The authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
High levels of high-density lipoprotein cholesterol (HDL-C) in older adults are associated with a higher risk of sustaining a fracture than lower HDL-C levels, a new study suggests.
“Two animal studies showing that HDL-C reduces bone mineral density by reducing osteoblast number and function provide a plausible explanation for why high HDL-C may increase the risk of fractures,” Monira Hussain, MBBS, MPH, PhD, of Monash University in Melbourne, told this news organization. “So, it was not surprising when our analyses provided evidence that amongst those in the highest quintile of HDL-C (> 74 mg/dL), there was a [33%] increased risk of fractures.”
After adjustment, one standard deviation increment in HDL-C level was associated with a 14% higher risk of fracture during a 4-year follow-up.
Based on this and other studies, Dr. Hussain said, “I believe that the finding of a very high HDL-C [should] alert clinicians to a higher risk of mortality, fractures, and possibly other threats to their patient’s health.”
The study was published online in JAMA Cardiology.
Independent risk factor
For this report, the researchers conducted a post hoc analysis of data from the Aspirin in Reducing Events in the Elderly (ASPREE) clinical trial and the ASPREE-Fracture substudy.
ASPREE was a double-blind, randomized, placebo-controlled primary prevention trial of aspirin. Participants were 16,703 community-dwelling Australians and 2,411 individuals from the United States with a mean age of 75 and without evident cardiovascular disease, dementia, physical disability, or life-limiting chronic illness.
The ASPREE-Fracture substudy collected data on fractures reported post randomization from the Australian participants. Fractures were confirmed by imaging and adjudicated by an expert panel and included both traumatic and minimal trauma fractures.
Of the 16,262 participants who had a plasma HDL-C measurement at baseline (55% women), 1,659 (10.2%) experienced at least one fracture over a median of 4 years. This included 711 minimal trauma fractures (for example, falls from standing height) and 948 other trauma fractures, mainly falls on stairs, ladders, or stools.
Higher rates of fractures occurred in the highest quintile of HDL-C level where the mean level was 89 mg/dL. At baseline, participants in that quintile had a lower BMI, a high prevalence of current/former smoking and current alcohol use, 12 years or longer of school, more physical activity, and higher use of antiosteoporosis medication. They also had less chronic kidney disease, diabetes, prefrailty/frailty, or treatment with lipid-lowering drugs.
In a fully adjusted model, each standard deviation increment in HDL-C level was associated with a 14% higher risk of fractures (hazard ratio, 1.14). When analyzed in quintiles, compared with participants in Q1, those in Q5 had a 33% higher risk for fracture (HR, 1.33).
Prevalence rates were similar between the sexes. The increase in fracture risk appeared to be independent of traditional risk factors for fractures, including age, sex, physical activity, alcohol use, frailty, BMI, smoking status, diabetes, chronic kidney disease, use of lipid-lowering or antiosteoporosis drugs, and education, the authors note.
The results persisted in sensitivity analyses in restricted subgroups of interest and in stratified analyses – including, for example, only minimal fractures; participants not taking antiosteoporosis drugs or statins; never smokers; nondrinkers; and those engaging in minimal physical activity (walking less than 30 minutes per day).
No association was observed between non–HDL-C levels and fractures.
The authors conclude that the study “provides robust evidence that higher levels of HDL-C are associated with incident fractures in both male and female individuals, independent of conventional risk factors.”
Clinically useful?
Commenting on the study for this news organization, Marilyn Tan, MD, clinic chief of the Endocrine Clinic and clinical associate professor of medicine at Stanford (Calif.) University, said, “I certainly would not recommend anyone do anything to actively lower their HDL levels. HDL levels are largely determined by genetics, diet, and lifestyle, with some effects from certain medications/supplements. Studies have demonstrated that moderately higher HDL levels may be protective for atherosclerosis.”
In the current study, she said, “Causation has not been proven, and importantly there is no evidence that reducing HDL levels reduces fracture risk. Also, this association between raised HDL levels and fracture risk has not been demonstrated consistently in other studies.”
Furthermore, she noted, the preclinical trials on which the authors based their hypothesis – that is, an association between HDL and a reduction in the number and function of osteoblasts – “has not been demonstrated widely in human subjects.”
“We have a large armamentarium of FDA-approved treatments for osteoporosis that have been clinically proven to reduce fracture risk very significantly, and these are the tools [in addition to lifestyle changes] we should use to reduce fracture risk,” Dr. Tan concluded.
John Wilkins, MD, of Northwestern University, Chicago, and Anand Rohatgi, MD, MSCS, of UT Southwestern Medical Center, Dallas, also point out some limitations of the study in a related editorial.
They note the inclusion of predominantly healthy adults with a mean age of 75, a population that could yield different findings from middle-aged cohorts with chronic illnesses, as well as a lack of clarity regarding the possible role of alcohol intake among the study participants.
Furthermore, the editorialists write, although significant associations were shown in this study, “models were not adjusted for detailed measures of exercise/activity, triglycerides, or any other lipids, including other HDL compositional measures such as HDL-P or ApoA-I levels. There was no assessment of whether HDL-C improved discrimination, reclassification, or any other validated measures of risk prediction performance.
“Taken together,” they conclude, “this study alone leaves several unanswered questions as to whether high HDL-C could be a useful biomarker to detect fracture risk.”
No commercial funding was disclosed. The authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Eye check important before starting semaglutide for diabetes
A small potential increased risk of retinopathy worsening at 1 year with injected semaglutide (Ozempic, Novo Nordisk), a glucagon-like peptide 1 (GLP-1) agonist approved for type 2 diabetes, doesn’t outweigh the drug’s cardiovascular benefits but does highlight the need for baseline ophthalmologic evaluation before initiating treatment and ongoing retinal monitoring, researchers say.
That conclusion was based on data from a meta-analysis of the seven major cardiovascular outcomes trials of GLP-1 agonists currently on the market.
The findings were recently published in Diabetes & Metabolic Syndrome: Clinical Research & Reviews, by Stewart G. Albert, MD, and colleagues.
Concerns about retinopathy worsening with the GLP-1 agonist drug class first arose from the SUSTAIN-6 cardiovascular outcomes trial for injectable semaglutide, although a subsequent analysis of data from that trial appeared to suggest the problem is likely due to rapid glucose-lowering in already vulnerable patients rather than a drug-specific effect. This effect had been previously reported, most notably in the landmark Diabetes Control and Complications Trial.
In this new meta-analysis, “we showed that with improvements in A1c there were correlations with decreases in the rate of cardiovascular events but increases in the rate of retinopathy,” Dr. Albert, of St. Louis University, told this news organization.
“As a class of drugs, we did not find an increased rate of retinopathy. The effect of GLP-1 agonists on retinopathy did not appear to be due to an immediate direct toxic effect of the drug. The worsening of the rate of retinopathy was seen with semaglutide after 1 year of therapy and when there was a decrease in A1c of 1%,” he explained.
He noted that because the increased risk was seen primarily among those who already had retinopathy at baseline, “it would seem prudent to know the level of retinopathy either before or plan for close ophthalmologic monitoring around the time of drug initiation ... We routinely evaluate patients with known type 2 diabetes mellitus at yearly intervals for retinopathy. From our data, we saw worsening at 1 year of drug exposure, but we do not know the exact time when the changes occurred during that year.”
The Ozempic label advises that “patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy” but doesn’t specifically mention baseline assessment at the time of drug initiation.
No increase in retinopathy risk for GLP-1 agonist class overall
The seven trials in the meta-analysis comprised 56,004 participants, with baseline retinopathy prevalence ranging from 9% to 31%.
For the GLP-1 agonist class overall, there was no significant increase in the relative rate (RR) of retinopathy (RR, 1.09; P = .36), while there were significant reductions in relative rates of major adverse cardiac events, overall deaths, and cardiovascular deaths (all P < .001 or P = .001).
The increased retinopathy risk was seen only in the subcutaneous semaglutide group (RR, 1.73; P = .02).
The overall number needed to harm was 1,000 and the number to treat was 77. For semaglutide, those values were 77 and 43, respectively.
There was a significant correlation between a decrease in major adverse cardiac events and a decrease in A1c (P = .014), while for retinopathy, the risk increased with improved A1c (P = .076).
Semaglutide subanalysis finds increased retinopathy worsening
Dr. Albert and colleagues conducted a separate subanalysis of 11 studies of semaglutide that enrolled 11,894 patients, of which 6 studies (n = 5,610) were of oral semaglutide (Rybelsus) and 5 studies were of subcutaneous semaglutide (Ozempic; n = 6,284).
In the subanalysis, there was an overall increase in relative rates of new or worsening retinopathy (RR, 1.218; P = .049).
The change in relative rate of retinopathy was predominantly found for subcutaneous semaglutide given for longer than 1 year (RR, 1.559; P = .022) and decreases in A1c of more than 1.0% (RR, 1.590; P = .016). No such differences were seen with oral semaglutide.
A further evaluation of the data without the SUSTAIN 6 trial showed no effect on retinopathy but the analysis lacked power.
Dr. Albert told this news organization: “We did not find an immediate toxic effect of any drug. However, we cannot rule out that there was a cumulative effect of the dose over longer times.”
No disclosures were given.
A version of this article first appeared on Medscape.com.
A small potential increased risk of retinopathy worsening at 1 year with injected semaglutide (Ozempic, Novo Nordisk), a glucagon-like peptide 1 (GLP-1) agonist approved for type 2 diabetes, doesn’t outweigh the drug’s cardiovascular benefits but does highlight the need for baseline ophthalmologic evaluation before initiating treatment and ongoing retinal monitoring, researchers say.
That conclusion was based on data from a meta-analysis of the seven major cardiovascular outcomes trials of GLP-1 agonists currently on the market.
The findings were recently published in Diabetes & Metabolic Syndrome: Clinical Research & Reviews, by Stewart G. Albert, MD, and colleagues.
Concerns about retinopathy worsening with the GLP-1 agonist drug class first arose from the SUSTAIN-6 cardiovascular outcomes trial for injectable semaglutide, although a subsequent analysis of data from that trial appeared to suggest the problem is likely due to rapid glucose-lowering in already vulnerable patients rather than a drug-specific effect. This effect had been previously reported, most notably in the landmark Diabetes Control and Complications Trial.
In this new meta-analysis, “we showed that with improvements in A1c there were correlations with decreases in the rate of cardiovascular events but increases in the rate of retinopathy,” Dr. Albert, of St. Louis University, told this news organization.
“As a class of drugs, we did not find an increased rate of retinopathy. The effect of GLP-1 agonists on retinopathy did not appear to be due to an immediate direct toxic effect of the drug. The worsening of the rate of retinopathy was seen with semaglutide after 1 year of therapy and when there was a decrease in A1c of 1%,” he explained.
He noted that because the increased risk was seen primarily among those who already had retinopathy at baseline, “it would seem prudent to know the level of retinopathy either before or plan for close ophthalmologic monitoring around the time of drug initiation ... We routinely evaluate patients with known type 2 diabetes mellitus at yearly intervals for retinopathy. From our data, we saw worsening at 1 year of drug exposure, but we do not know the exact time when the changes occurred during that year.”
The Ozempic label advises that “patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy” but doesn’t specifically mention baseline assessment at the time of drug initiation.
No increase in retinopathy risk for GLP-1 agonist class overall
The seven trials in the meta-analysis comprised 56,004 participants, with baseline retinopathy prevalence ranging from 9% to 31%.
For the GLP-1 agonist class overall, there was no significant increase in the relative rate (RR) of retinopathy (RR, 1.09; P = .36), while there were significant reductions in relative rates of major adverse cardiac events, overall deaths, and cardiovascular deaths (all P < .001 or P = .001).
The increased retinopathy risk was seen only in the subcutaneous semaglutide group (RR, 1.73; P = .02).
The overall number needed to harm was 1,000 and the number to treat was 77. For semaglutide, those values were 77 and 43, respectively.
There was a significant correlation between a decrease in major adverse cardiac events and a decrease in A1c (P = .014), while for retinopathy, the risk increased with improved A1c (P = .076).
Semaglutide subanalysis finds increased retinopathy worsening
Dr. Albert and colleagues conducted a separate subanalysis of 11 studies of semaglutide that enrolled 11,894 patients, of which 6 studies (n = 5,610) were of oral semaglutide (Rybelsus) and 5 studies were of subcutaneous semaglutide (Ozempic; n = 6,284).
In the subanalysis, there was an overall increase in relative rates of new or worsening retinopathy (RR, 1.218; P = .049).
The change in relative rate of retinopathy was predominantly found for subcutaneous semaglutide given for longer than 1 year (RR, 1.559; P = .022) and decreases in A1c of more than 1.0% (RR, 1.590; P = .016). No such differences were seen with oral semaglutide.
A further evaluation of the data without the SUSTAIN 6 trial showed no effect on retinopathy but the analysis lacked power.
Dr. Albert told this news organization: “We did not find an immediate toxic effect of any drug. However, we cannot rule out that there was a cumulative effect of the dose over longer times.”
No disclosures were given.
A version of this article first appeared on Medscape.com.
A small potential increased risk of retinopathy worsening at 1 year with injected semaglutide (Ozempic, Novo Nordisk), a glucagon-like peptide 1 (GLP-1) agonist approved for type 2 diabetes, doesn’t outweigh the drug’s cardiovascular benefits but does highlight the need for baseline ophthalmologic evaluation before initiating treatment and ongoing retinal monitoring, researchers say.
That conclusion was based on data from a meta-analysis of the seven major cardiovascular outcomes trials of GLP-1 agonists currently on the market.
The findings were recently published in Diabetes & Metabolic Syndrome: Clinical Research & Reviews, by Stewart G. Albert, MD, and colleagues.
Concerns about retinopathy worsening with the GLP-1 agonist drug class first arose from the SUSTAIN-6 cardiovascular outcomes trial for injectable semaglutide, although a subsequent analysis of data from that trial appeared to suggest the problem is likely due to rapid glucose-lowering in already vulnerable patients rather than a drug-specific effect. This effect had been previously reported, most notably in the landmark Diabetes Control and Complications Trial.
In this new meta-analysis, “we showed that with improvements in A1c there were correlations with decreases in the rate of cardiovascular events but increases in the rate of retinopathy,” Dr. Albert, of St. Louis University, told this news organization.
“As a class of drugs, we did not find an increased rate of retinopathy. The effect of GLP-1 agonists on retinopathy did not appear to be due to an immediate direct toxic effect of the drug. The worsening of the rate of retinopathy was seen with semaglutide after 1 year of therapy and when there was a decrease in A1c of 1%,” he explained.
He noted that because the increased risk was seen primarily among those who already had retinopathy at baseline, “it would seem prudent to know the level of retinopathy either before or plan for close ophthalmologic monitoring around the time of drug initiation ... We routinely evaluate patients with known type 2 diabetes mellitus at yearly intervals for retinopathy. From our data, we saw worsening at 1 year of drug exposure, but we do not know the exact time when the changes occurred during that year.”
The Ozempic label advises that “patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy” but doesn’t specifically mention baseline assessment at the time of drug initiation.
No increase in retinopathy risk for GLP-1 agonist class overall
The seven trials in the meta-analysis comprised 56,004 participants, with baseline retinopathy prevalence ranging from 9% to 31%.
For the GLP-1 agonist class overall, there was no significant increase in the relative rate (RR) of retinopathy (RR, 1.09; P = .36), while there were significant reductions in relative rates of major adverse cardiac events, overall deaths, and cardiovascular deaths (all P < .001 or P = .001).
The increased retinopathy risk was seen only in the subcutaneous semaglutide group (RR, 1.73; P = .02).
The overall number needed to harm was 1,000 and the number to treat was 77. For semaglutide, those values were 77 and 43, respectively.
There was a significant correlation between a decrease in major adverse cardiac events and a decrease in A1c (P = .014), while for retinopathy, the risk increased with improved A1c (P = .076).
Semaglutide subanalysis finds increased retinopathy worsening
Dr. Albert and colleagues conducted a separate subanalysis of 11 studies of semaglutide that enrolled 11,894 patients, of which 6 studies (n = 5,610) were of oral semaglutide (Rybelsus) and 5 studies were of subcutaneous semaglutide (Ozempic; n = 6,284).
In the subanalysis, there was an overall increase in relative rates of new or worsening retinopathy (RR, 1.218; P = .049).
The change in relative rate of retinopathy was predominantly found for subcutaneous semaglutide given for longer than 1 year (RR, 1.559; P = .022) and decreases in A1c of more than 1.0% (RR, 1.590; P = .016). No such differences were seen with oral semaglutide.
A further evaluation of the data without the SUSTAIN 6 trial showed no effect on retinopathy but the analysis lacked power.
Dr. Albert told this news organization: “We did not find an immediate toxic effect of any drug. However, we cannot rule out that there was a cumulative effect of the dose over longer times.”
No disclosures were given.
A version of this article first appeared on Medscape.com.
FROM DIABETES & METABOLIC SYNDROME: CLINICAL RESEARCH & REVIEWS