User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Alcohol may curb stress signaling in brain to protect heart
The study shows that light to moderate drinking was associated with lower major adverse cardiovascular events (MACE), and this was partly mediated by decreased stress signaling in the brain.
In addition, the benefit of light to moderate drinking with respect to MACE was most pronounced among people with a history of anxiety, a condition known to be associated with higher stress signaling in the brain.
However, the apparent CVD benefits of light to moderate drinking were counterbalanced by an increased risk of cancer.
“There is no safe level of alcohol consumption,” senior author and cardiologist Ahmed Tawakol, MD, codirector of the Cardiovascular Imaging Research Center at Massachusetts General Hospital, Boston, said in an interview.
“We see cancer risk even at the level that we see some protection from heart disease. And higher amounts of alcohol clearly increase heart disease risk,” Dr. Tawakol said.
The study was published online in the Journal of the American College of Cardiology.
Clear mechanistic link
Chronic stress is associated with MACE via stress-related neural network activity (SNA). Light to moderate alcohol consumption has been linked to lower MACE risk, but the mechanisms behind this connection remain unclear.
“We know that when the neural centers of stress are activated, they trigger downstream changes that result in heart disease. And we’ve long appreciated that alcohol in the short term reduces stress, so we hypothesized that maybe alcohol impacts those stress systems chronically and that might explain its cardiovascular effects,” Dr. Tawakol explained.
The study included roughly 53,000 adults (mean age, 60 years; 60% women) from the Mass General Brigham Biobank. The researchers first evaluated the relationship between light to moderate alcohol consumption and MACE after adjusting for a range of genetic, clinical, lifestyle, and socioeconomic factors.
During mean follow-up of 3.4 years, 1,914 individuals experienced MACE. Light to moderate alcohol consumption (compared to none/minimal) was associated with lower MACE risk (hazard ratio [HR], 0.786; 95% confidence interval [CI], 0.717-0.862; P < .0001) after adjustment for cardiovascular risk factors.
The researchers then studied a subset of 713 individuals who had undergone previous PET/CT brain imaging (primarily for cancer surveillance) to determine the effect of light to moderate alcohol consumption on resting SNA.
They found that light to moderate alcohol consumption correlated with decreased SNA (standardized beta, –0.192; 95% CI, –0.338 to 0.046; P = .01). Lower SNA partially mediated the beneficial effect of light to moderate alcohol intake on MACE risk (odds ratio [OR], –0.040; 95% CI, –0.097 to –0.003; P < .05).
Light to moderate alcohol consumption was associated with larger decreases in MACE risk among individuals with a history of anxiety (HR, 0.60; 95% CI, 0.50-0.72, vs. HR, 1.78; 95% CI, 0.73-0.80; P = .003).
The coauthors of an editorial say the discovery of a “new possible mechanism of action” for why light to moderate alcohol consumption might protect the heart “deserves closer attention in future investigations.”
However, Giovanni de Gaetano, MD, PhD, department of epidemiology and prevention, IRCCS NEUROMED, Pozzilli, Italy, one of the authors, emphasized that individuals who consume alcohol should not “exceed the recommended daily dose limits suggested in many countries and that no abstainer should start to drink, even in moderation, solely for the purpose of improving his/her health outcomes.”
Dr. Tawakol and colleagues said that, given alcohol’s adverse health effects, such as heightened cancer risk, new interventions that have positive effects on the neurobiology of stress but without the harmful effects of alcohol are needed.
To that end, they are studying the effect of exercise, stress-reduction interventions such as meditation, and pharmacologic therapies on stress-associated neural networks, and how they might induce CV benefits.
Dr. Tawakol said in an interview that one “additional important message is that anxiety and other related conditions like depression have really substantial health consequences, including increased MACE. Safer interventions that reduce anxiety may yet prove to reduce the risk of heart disease very nicely.”
The study was supported by the National Institutes of Health. Dr. Tawakol and Dr. de Gaetano have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The study shows that light to moderate drinking was associated with lower major adverse cardiovascular events (MACE), and this was partly mediated by decreased stress signaling in the brain.
In addition, the benefit of light to moderate drinking with respect to MACE was most pronounced among people with a history of anxiety, a condition known to be associated with higher stress signaling in the brain.
However, the apparent CVD benefits of light to moderate drinking were counterbalanced by an increased risk of cancer.
“There is no safe level of alcohol consumption,” senior author and cardiologist Ahmed Tawakol, MD, codirector of the Cardiovascular Imaging Research Center at Massachusetts General Hospital, Boston, said in an interview.
“We see cancer risk even at the level that we see some protection from heart disease. And higher amounts of alcohol clearly increase heart disease risk,” Dr. Tawakol said.
The study was published online in the Journal of the American College of Cardiology.
Clear mechanistic link
Chronic stress is associated with MACE via stress-related neural network activity (SNA). Light to moderate alcohol consumption has been linked to lower MACE risk, but the mechanisms behind this connection remain unclear.
“We know that when the neural centers of stress are activated, they trigger downstream changes that result in heart disease. And we’ve long appreciated that alcohol in the short term reduces stress, so we hypothesized that maybe alcohol impacts those stress systems chronically and that might explain its cardiovascular effects,” Dr. Tawakol explained.
The study included roughly 53,000 adults (mean age, 60 years; 60% women) from the Mass General Brigham Biobank. The researchers first evaluated the relationship between light to moderate alcohol consumption and MACE after adjusting for a range of genetic, clinical, lifestyle, and socioeconomic factors.
During mean follow-up of 3.4 years, 1,914 individuals experienced MACE. Light to moderate alcohol consumption (compared to none/minimal) was associated with lower MACE risk (hazard ratio [HR], 0.786; 95% confidence interval [CI], 0.717-0.862; P < .0001) after adjustment for cardiovascular risk factors.
The researchers then studied a subset of 713 individuals who had undergone previous PET/CT brain imaging (primarily for cancer surveillance) to determine the effect of light to moderate alcohol consumption on resting SNA.
They found that light to moderate alcohol consumption correlated with decreased SNA (standardized beta, –0.192; 95% CI, –0.338 to 0.046; P = .01). Lower SNA partially mediated the beneficial effect of light to moderate alcohol intake on MACE risk (odds ratio [OR], –0.040; 95% CI, –0.097 to –0.003; P < .05).
Light to moderate alcohol consumption was associated with larger decreases in MACE risk among individuals with a history of anxiety (HR, 0.60; 95% CI, 0.50-0.72, vs. HR, 1.78; 95% CI, 0.73-0.80; P = .003).
The coauthors of an editorial say the discovery of a “new possible mechanism of action” for why light to moderate alcohol consumption might protect the heart “deserves closer attention in future investigations.”
However, Giovanni de Gaetano, MD, PhD, department of epidemiology and prevention, IRCCS NEUROMED, Pozzilli, Italy, one of the authors, emphasized that individuals who consume alcohol should not “exceed the recommended daily dose limits suggested in many countries and that no abstainer should start to drink, even in moderation, solely for the purpose of improving his/her health outcomes.”
Dr. Tawakol and colleagues said that, given alcohol’s adverse health effects, such as heightened cancer risk, new interventions that have positive effects on the neurobiology of stress but without the harmful effects of alcohol are needed.
To that end, they are studying the effect of exercise, stress-reduction interventions such as meditation, and pharmacologic therapies on stress-associated neural networks, and how they might induce CV benefits.
Dr. Tawakol said in an interview that one “additional important message is that anxiety and other related conditions like depression have really substantial health consequences, including increased MACE. Safer interventions that reduce anxiety may yet prove to reduce the risk of heart disease very nicely.”
The study was supported by the National Institutes of Health. Dr. Tawakol and Dr. de Gaetano have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The study shows that light to moderate drinking was associated with lower major adverse cardiovascular events (MACE), and this was partly mediated by decreased stress signaling in the brain.
In addition, the benefit of light to moderate drinking with respect to MACE was most pronounced among people with a history of anxiety, a condition known to be associated with higher stress signaling in the brain.
However, the apparent CVD benefits of light to moderate drinking were counterbalanced by an increased risk of cancer.
“There is no safe level of alcohol consumption,” senior author and cardiologist Ahmed Tawakol, MD, codirector of the Cardiovascular Imaging Research Center at Massachusetts General Hospital, Boston, said in an interview.
“We see cancer risk even at the level that we see some protection from heart disease. And higher amounts of alcohol clearly increase heart disease risk,” Dr. Tawakol said.
The study was published online in the Journal of the American College of Cardiology.
Clear mechanistic link
Chronic stress is associated with MACE via stress-related neural network activity (SNA). Light to moderate alcohol consumption has been linked to lower MACE risk, but the mechanisms behind this connection remain unclear.
“We know that when the neural centers of stress are activated, they trigger downstream changes that result in heart disease. And we’ve long appreciated that alcohol in the short term reduces stress, so we hypothesized that maybe alcohol impacts those stress systems chronically and that might explain its cardiovascular effects,” Dr. Tawakol explained.
The study included roughly 53,000 adults (mean age, 60 years; 60% women) from the Mass General Brigham Biobank. The researchers first evaluated the relationship between light to moderate alcohol consumption and MACE after adjusting for a range of genetic, clinical, lifestyle, and socioeconomic factors.
During mean follow-up of 3.4 years, 1,914 individuals experienced MACE. Light to moderate alcohol consumption (compared to none/minimal) was associated with lower MACE risk (hazard ratio [HR], 0.786; 95% confidence interval [CI], 0.717-0.862; P < .0001) after adjustment for cardiovascular risk factors.
The researchers then studied a subset of 713 individuals who had undergone previous PET/CT brain imaging (primarily for cancer surveillance) to determine the effect of light to moderate alcohol consumption on resting SNA.
They found that light to moderate alcohol consumption correlated with decreased SNA (standardized beta, –0.192; 95% CI, –0.338 to 0.046; P = .01). Lower SNA partially mediated the beneficial effect of light to moderate alcohol intake on MACE risk (odds ratio [OR], –0.040; 95% CI, –0.097 to –0.003; P < .05).
Light to moderate alcohol consumption was associated with larger decreases in MACE risk among individuals with a history of anxiety (HR, 0.60; 95% CI, 0.50-0.72, vs. HR, 1.78; 95% CI, 0.73-0.80; P = .003).
The coauthors of an editorial say the discovery of a “new possible mechanism of action” for why light to moderate alcohol consumption might protect the heart “deserves closer attention in future investigations.”
However, Giovanni de Gaetano, MD, PhD, department of epidemiology and prevention, IRCCS NEUROMED, Pozzilli, Italy, one of the authors, emphasized that individuals who consume alcohol should not “exceed the recommended daily dose limits suggested in many countries and that no abstainer should start to drink, even in moderation, solely for the purpose of improving his/her health outcomes.”
Dr. Tawakol and colleagues said that, given alcohol’s adverse health effects, such as heightened cancer risk, new interventions that have positive effects on the neurobiology of stress but without the harmful effects of alcohol are needed.
To that end, they are studying the effect of exercise, stress-reduction interventions such as meditation, and pharmacologic therapies on stress-associated neural networks, and how they might induce CV benefits.
Dr. Tawakol said in an interview that one “additional important message is that anxiety and other related conditions like depression have really substantial health consequences, including increased MACE. Safer interventions that reduce anxiety may yet prove to reduce the risk of heart disease very nicely.”
The study was supported by the National Institutes of Health. Dr. Tawakol and Dr. de Gaetano have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
The cardiopulmonary effects of mask wearing
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
There was a time when I would have had to explain to you what an N95 mask is, how it is designed to filter out 95% of fine particles, defined as stuff in the air less than 2.5 microns in size.
But of course, you know that now. The N95 had its moment – a moment that seemed to be passing as the concentration of airborne coronavirus particles decreased.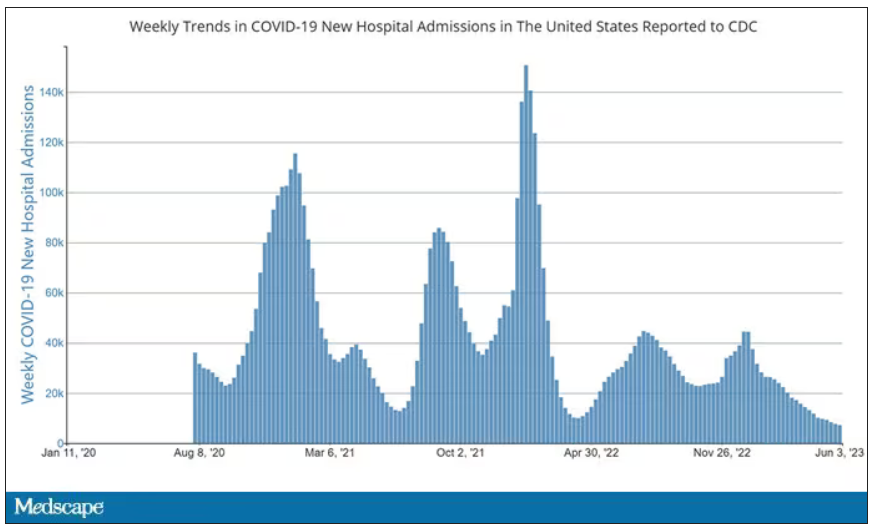
But, as the poet said, all that is less than 2.5 microns in size is not coronavirus. Wildfire smoke is also chock full of fine particulate matter. And so, N95s are having something of a comeback.
That’s why an article that took a deep look at what happens to our cardiovascular system when we wear N95 masks caught my eye.
Mask wearing has been the subject of intense debate around the country. While the vast majority of evidence, as well as the personal experience of thousands of doctors, suggests that wearing a mask has no significant physiologic effects, it’s not hard to find those who suggest that mask wearing depletes oxygen levels, or leads to infection, or has other bizarre effects.
In a world of conflicting opinions, a controlled study is a wonderful thing, and that’s what appeared in JAMA Network Open.
This isn’t a huge study, but it’s big enough to make some important conclusions. Thirty individuals, all young and healthy, half female, were enrolled. Each participant spent 3 days in a metabolic chamber; this is essentially a giant, airtight room where all the inputs (oxygen levels and so on) and outputs (carbon dioxide levels and so on) can be precisely measured.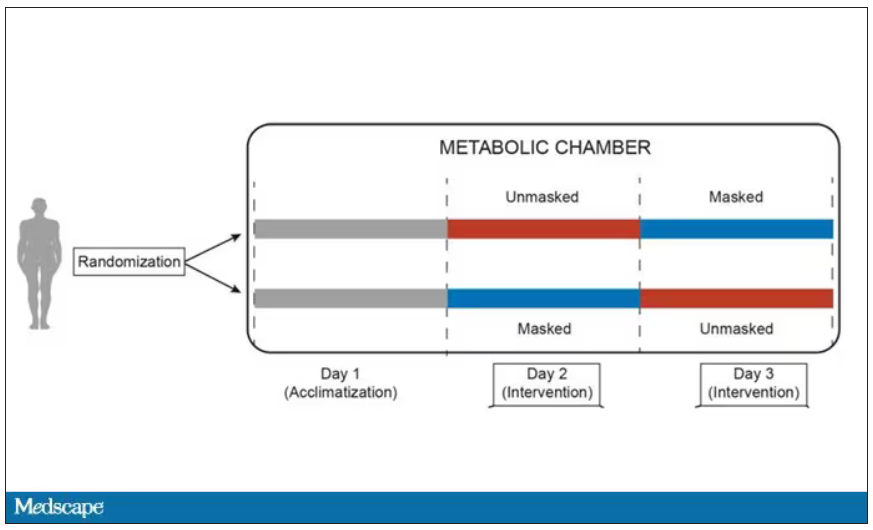
After a day of getting used to the environment, the participants spent a day either wearing an N95 mask or not for 16 waking hours. On the next day, they switched. Every other variable was controlled, from the calories in their diet to the temperature of the room itself.
They engaged in light exercise twice during the day – riding a stationary bike – and a host of physiologic parameters were measured. The question being, would the wearing of the mask for 16 hours straight change anything?
And the answer is yes, some things changed, but not by much.
Here’s a graph of the heart rate over time. You can see some separation, with higher heart rates during the mask-wearing day, particularly around 11 a.m. – when light exercise was scheduled.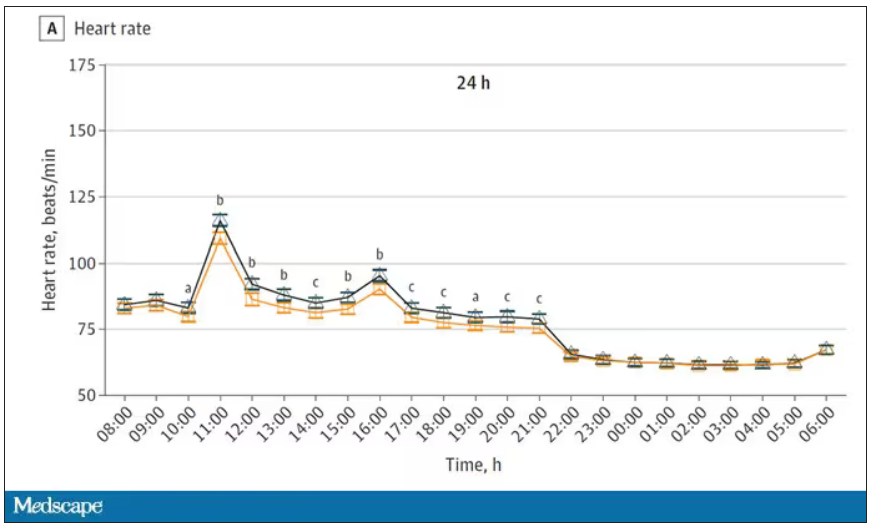
Zooming in on the exercise period makes the difference more clear. The heart rate was about eight beats/min higher while masked and engaging in exercise. Systolic blood pressure was about 6 mm Hg higher. Oxygen saturation was lower by 0.7%.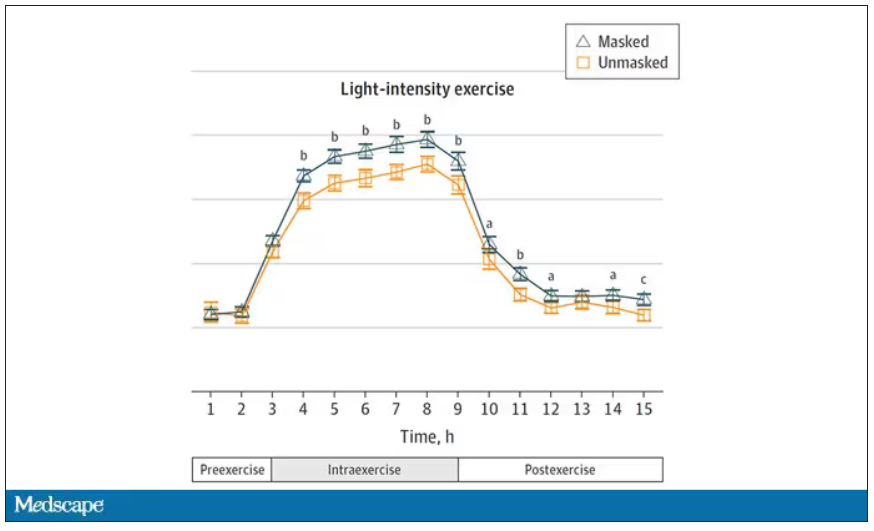
So yes, exercising while wearing an N95 mask might be different from exercising without an N95 mask. But nothing here looks dangerous to me. The 0.7% decrease in oxygen saturation is smaller than the typical measurement error of a pulse oximeter. The authors write that venous pH decreased during the masked day, which is of more interest to me as a nephrologist, but they don’t show that data even in the supplement. I suspect it didn’t decrease much.
They also showed that respiratory rate during exercise decreased in the masked condition. That doesn’t really make sense when you think about it in the context of the other findings, which are all suggestive of increased metabolic rate and sympathetic drive. Does that call the whole procedure into question? No, but it’s worth noting.
These were young, healthy people. You could certainly argue that those with more vulnerable cardiopulmonary status might have had different effects from mask wearing, but without a specific study in those people, it’s just conjecture. Clearly, this study lets us conclude that mask wearing at rest has less of an effect than mask wearing during exercise.
But remember that, in reality, we are wearing masks for a reason. One could imagine a study where this metabolic chamber was filled with wildfire smoke at a concentration similar to what we saw in New York. In that situation, we might find that wearing an N95 is quite helpful. The thing is, studying masks in isolation is useful because you can control so many variables. But masks aren’t used in isolation. In fact, that’s sort of their defining characteristic.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
There was a time when I would have had to explain to you what an N95 mask is, how it is designed to filter out 95% of fine particles, defined as stuff in the air less than 2.5 microns in size.
But of course, you know that now. The N95 had its moment – a moment that seemed to be passing as the concentration of airborne coronavirus particles decreased.
But, as the poet said, all that is less than 2.5 microns in size is not coronavirus. Wildfire smoke is also chock full of fine particulate matter. And so, N95s are having something of a comeback.
That’s why an article that took a deep look at what happens to our cardiovascular system when we wear N95 masks caught my eye.
Mask wearing has been the subject of intense debate around the country. While the vast majority of evidence, as well as the personal experience of thousands of doctors, suggests that wearing a mask has no significant physiologic effects, it’s not hard to find those who suggest that mask wearing depletes oxygen levels, or leads to infection, or has other bizarre effects.
In a world of conflicting opinions, a controlled study is a wonderful thing, and that’s what appeared in JAMA Network Open.
This isn’t a huge study, but it’s big enough to make some important conclusions. Thirty individuals, all young and healthy, half female, were enrolled. Each participant spent 3 days in a metabolic chamber; this is essentially a giant, airtight room where all the inputs (oxygen levels and so on) and outputs (carbon dioxide levels and so on) can be precisely measured.
After a day of getting used to the environment, the participants spent a day either wearing an N95 mask or not for 16 waking hours. On the next day, they switched. Every other variable was controlled, from the calories in their diet to the temperature of the room itself.
They engaged in light exercise twice during the day – riding a stationary bike – and a host of physiologic parameters were measured. The question being, would the wearing of the mask for 16 hours straight change anything?
And the answer is yes, some things changed, but not by much.
Here’s a graph of the heart rate over time. You can see some separation, with higher heart rates during the mask-wearing day, particularly around 11 a.m. – when light exercise was scheduled.
Zooming in on the exercise period makes the difference more clear. The heart rate was about eight beats/min higher while masked and engaging in exercise. Systolic blood pressure was about 6 mm Hg higher. Oxygen saturation was lower by 0.7%.
So yes, exercising while wearing an N95 mask might be different from exercising without an N95 mask. But nothing here looks dangerous to me. The 0.7% decrease in oxygen saturation is smaller than the typical measurement error of a pulse oximeter. The authors write that venous pH decreased during the masked day, which is of more interest to me as a nephrologist, but they don’t show that data even in the supplement. I suspect it didn’t decrease much.
They also showed that respiratory rate during exercise decreased in the masked condition. That doesn’t really make sense when you think about it in the context of the other findings, which are all suggestive of increased metabolic rate and sympathetic drive. Does that call the whole procedure into question? No, but it’s worth noting.
These were young, healthy people. You could certainly argue that those with more vulnerable cardiopulmonary status might have had different effects from mask wearing, but without a specific study in those people, it’s just conjecture. Clearly, this study lets us conclude that mask wearing at rest has less of an effect than mask wearing during exercise.
But remember that, in reality, we are wearing masks for a reason. One could imagine a study where this metabolic chamber was filled with wildfire smoke at a concentration similar to what we saw in New York. In that situation, we might find that wearing an N95 is quite helpful. The thing is, studying masks in isolation is useful because you can control so many variables. But masks aren’t used in isolation. In fact, that’s sort of their defining characteristic.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
There was a time when I would have had to explain to you what an N95 mask is, how it is designed to filter out 95% of fine particles, defined as stuff in the air less than 2.5 microns in size.
But of course, you know that now. The N95 had its moment – a moment that seemed to be passing as the concentration of airborne coronavirus particles decreased.
But, as the poet said, all that is less than 2.5 microns in size is not coronavirus. Wildfire smoke is also chock full of fine particulate matter. And so, N95s are having something of a comeback.
That’s why an article that took a deep look at what happens to our cardiovascular system when we wear N95 masks caught my eye.
Mask wearing has been the subject of intense debate around the country. While the vast majority of evidence, as well as the personal experience of thousands of doctors, suggests that wearing a mask has no significant physiologic effects, it’s not hard to find those who suggest that mask wearing depletes oxygen levels, or leads to infection, or has other bizarre effects.
In a world of conflicting opinions, a controlled study is a wonderful thing, and that’s what appeared in JAMA Network Open.
This isn’t a huge study, but it’s big enough to make some important conclusions. Thirty individuals, all young and healthy, half female, were enrolled. Each participant spent 3 days in a metabolic chamber; this is essentially a giant, airtight room where all the inputs (oxygen levels and so on) and outputs (carbon dioxide levels and so on) can be precisely measured.
After a day of getting used to the environment, the participants spent a day either wearing an N95 mask or not for 16 waking hours. On the next day, they switched. Every other variable was controlled, from the calories in their diet to the temperature of the room itself.
They engaged in light exercise twice during the day – riding a stationary bike – and a host of physiologic parameters were measured. The question being, would the wearing of the mask for 16 hours straight change anything?
And the answer is yes, some things changed, but not by much.
Here’s a graph of the heart rate over time. You can see some separation, with higher heart rates during the mask-wearing day, particularly around 11 a.m. – when light exercise was scheduled.
Zooming in on the exercise period makes the difference more clear. The heart rate was about eight beats/min higher while masked and engaging in exercise. Systolic blood pressure was about 6 mm Hg higher. Oxygen saturation was lower by 0.7%.
So yes, exercising while wearing an N95 mask might be different from exercising without an N95 mask. But nothing here looks dangerous to me. The 0.7% decrease in oxygen saturation is smaller than the typical measurement error of a pulse oximeter. The authors write that venous pH decreased during the masked day, which is of more interest to me as a nephrologist, but they don’t show that data even in the supplement. I suspect it didn’t decrease much.
They also showed that respiratory rate during exercise decreased in the masked condition. That doesn’t really make sense when you think about it in the context of the other findings, which are all suggestive of increased metabolic rate and sympathetic drive. Does that call the whole procedure into question? No, but it’s worth noting.
These were young, healthy people. You could certainly argue that those with more vulnerable cardiopulmonary status might have had different effects from mask wearing, but without a specific study in those people, it’s just conjecture. Clearly, this study lets us conclude that mask wearing at rest has less of an effect than mask wearing during exercise.
But remember that, in reality, we are wearing masks for a reason. One could imagine a study where this metabolic chamber was filled with wildfire smoke at a concentration similar to what we saw in New York. In that situation, we might find that wearing an N95 is quite helpful. The thing is, studying masks in isolation is useful because you can control so many variables. But masks aren’t used in isolation. In fact, that’s sort of their defining characteristic.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Good COP, bad COP. Is this cardiorespiratory measure the best predictor of early death?
according to clinicians who champion the assessment. The COP is easier to obtain than cardiorespiratory measures that require people to exercise to their limit, advocates say; rather than running full speed, someone can walk or lightly jog on a treadmill, with a COP value obtained easily.
But other clinicians argue that maximal exercise tests have many prognostic benefits, and that physicians should do everything in their power to push patients to exercise as hard as possible. In particular, the VO2 max test captures the maximum amount of oxygen someone uses when exercising at their capacity and is the preferred method for measuring cardiovascular endurance.
The COP is a measure of the minimum number of liters of air during breathing required to move one liter of oxygen through the bloodstream. The lower the COP the better, because this means that someone is working less strenuously than someone else to transport the same amount of oxygen, denoting a more efficient interaction between their heart and lungs.
The COP for a fit person might be 15, about 20-25 for a healthy person, and 35 for someone with heart failure, according to Claudio Gil Araújo, MD, PhD, director of research and education at CLINIMEX, an exercise medicine clinic in Rio de Janeiro.
“Max VO2 is very important, that’s indisputable. But when do you use max VO2 in your daily life? Never,” Dr. Araújo said. But almost anyone can generate a COP.
Emerging uses for the COP
“I can put someone on the treadmill or bike, and after 3 or 4 minutes I have the COP. It’s like a walking pace,” Dr. Araújo said. Yet the values are obtained with roughly half the effort as VO2 max. Other clinicians argue exercising to the limits of endurance offers unique clinical insights.
“We should do everything in our power to exercise our patients to maximum. How long a patient is able to go is really important,” said Anu Lala, MD, a cardiologist who specializes in heart failure treatment at Mount Sinai Hospital in New York. A full-capacity exercise test gives useful insights into someone’s heart rate, heart rate recovery, blood pressure, and ECG response to vigorous exercise, Dr. Lala added, all of which are important clues to someone’s overall health.
In 2012 Dr. Araújo coauthored a study that first defined the COP, which is calculated by measuring expired gasses people produce while gently exercising, perhaps to the point where they begin to perspire, and then dividing their breathing capacity by their oxygen uptake every minute. The lowest value obtained during any exercise session is the COP.
Various studies show that higher COP values are associated with more severe heart lesions in patients with congenital heart disease; higher levels of mortality in seemingly healthy male adults; and with worse prognoses in patients with heart failure. These studies all appeared within the last 7 months.
The mortality study, which Dr. Araújo coauthored, compared COP in more than 3,000 U.S. men and women who completed an exercise test from 1973 to 2018 and were tracked for an average of 23 years. Although COP was introduced as an assessment in 2012, calculating the value from tests prior to that date was possible because those tests had captured the relevant breathing rate and oxygen uptake. In males aged 18-85 years, a worse COP was significantly associated with an increased risk for earlier death. This finding did not hold for females, however; Dr. Araújo noted that more research is needed to understand the discrepancy in COP’s predictive power by sex.
In the heart failure study, everyone enrolled had heart failure and completed a COP test. People with the worse COPs also had the worst symptoms of heart failure, but completing an exercise rehabilitation program improved COP values when researchers measured them again. Dr. Araújo was also part of this study, based in the Netherlands.
“I think the COP could become a novel parameter in clinical care,” for most people, said Thijs Eijsvogels, PhD, an exercise physiologist at Radboud University in Nijmegen, the Netherlands, and the senior author of the heart failure study. That said, Dr. Eijsvogels said elite athletes will always be more interested in measuring VO2 max.
Dr. Lala agreed that tests such as the COP have some value. Her own work has shown that measuring the efficiency of someone’s breathing patterns for exhaling carbon dioxide, which can also be done without making people exercise full strength, has prognostic value for patients with advanced heart failure. Even so, she said she would like to see maximal effort tests used as much as possible.
“I worry about saying we’re going to settle for a parameter that can be achieved at 50% of peak VO2 and then we don’t exercise our patients,” Dr. Lala said.
Dr. Araújo said he plans to continue to measure VO2 max but he believes COP has utility – even for elite athletes. One of his patients is a frequent Ironman competitor who competes well despite having a solid but not amazing VO2 max level. But her COP is quite low, Dr. Araújo said, which to him suggests an especially efficient interaction between her respiratory and cardiovascular systems.
“We have a new player in the game,” Dr. Araújo said.
The sources in this study report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to clinicians who champion the assessment. The COP is easier to obtain than cardiorespiratory measures that require people to exercise to their limit, advocates say; rather than running full speed, someone can walk or lightly jog on a treadmill, with a COP value obtained easily.
But other clinicians argue that maximal exercise tests have many prognostic benefits, and that physicians should do everything in their power to push patients to exercise as hard as possible. In particular, the VO2 max test captures the maximum amount of oxygen someone uses when exercising at their capacity and is the preferred method for measuring cardiovascular endurance.
The COP is a measure of the minimum number of liters of air during breathing required to move one liter of oxygen through the bloodstream. The lower the COP the better, because this means that someone is working less strenuously than someone else to transport the same amount of oxygen, denoting a more efficient interaction between their heart and lungs.
The COP for a fit person might be 15, about 20-25 for a healthy person, and 35 for someone with heart failure, according to Claudio Gil Araújo, MD, PhD, director of research and education at CLINIMEX, an exercise medicine clinic in Rio de Janeiro.
“Max VO2 is very important, that’s indisputable. But when do you use max VO2 in your daily life? Never,” Dr. Araújo said. But almost anyone can generate a COP.
Emerging uses for the COP
“I can put someone on the treadmill or bike, and after 3 or 4 minutes I have the COP. It’s like a walking pace,” Dr. Araújo said. Yet the values are obtained with roughly half the effort as VO2 max. Other clinicians argue exercising to the limits of endurance offers unique clinical insights.
“We should do everything in our power to exercise our patients to maximum. How long a patient is able to go is really important,” said Anu Lala, MD, a cardiologist who specializes in heart failure treatment at Mount Sinai Hospital in New York. A full-capacity exercise test gives useful insights into someone’s heart rate, heart rate recovery, blood pressure, and ECG response to vigorous exercise, Dr. Lala added, all of which are important clues to someone’s overall health.
In 2012 Dr. Araújo coauthored a study that first defined the COP, which is calculated by measuring expired gasses people produce while gently exercising, perhaps to the point where they begin to perspire, and then dividing their breathing capacity by their oxygen uptake every minute. The lowest value obtained during any exercise session is the COP.
Various studies show that higher COP values are associated with more severe heart lesions in patients with congenital heart disease; higher levels of mortality in seemingly healthy male adults; and with worse prognoses in patients with heart failure. These studies all appeared within the last 7 months.
The mortality study, which Dr. Araújo coauthored, compared COP in more than 3,000 U.S. men and women who completed an exercise test from 1973 to 2018 and were tracked for an average of 23 years. Although COP was introduced as an assessment in 2012, calculating the value from tests prior to that date was possible because those tests had captured the relevant breathing rate and oxygen uptake. In males aged 18-85 years, a worse COP was significantly associated with an increased risk for earlier death. This finding did not hold for females, however; Dr. Araújo noted that more research is needed to understand the discrepancy in COP’s predictive power by sex.
In the heart failure study, everyone enrolled had heart failure and completed a COP test. People with the worse COPs also had the worst symptoms of heart failure, but completing an exercise rehabilitation program improved COP values when researchers measured them again. Dr. Araújo was also part of this study, based in the Netherlands.
“I think the COP could become a novel parameter in clinical care,” for most people, said Thijs Eijsvogels, PhD, an exercise physiologist at Radboud University in Nijmegen, the Netherlands, and the senior author of the heart failure study. That said, Dr. Eijsvogels said elite athletes will always be more interested in measuring VO2 max.
Dr. Lala agreed that tests such as the COP have some value. Her own work has shown that measuring the efficiency of someone’s breathing patterns for exhaling carbon dioxide, which can also be done without making people exercise full strength, has prognostic value for patients with advanced heart failure. Even so, she said she would like to see maximal effort tests used as much as possible.
“I worry about saying we’re going to settle for a parameter that can be achieved at 50% of peak VO2 and then we don’t exercise our patients,” Dr. Lala said.
Dr. Araújo said he plans to continue to measure VO2 max but he believes COP has utility – even for elite athletes. One of his patients is a frequent Ironman competitor who competes well despite having a solid but not amazing VO2 max level. But her COP is quite low, Dr. Araújo said, which to him suggests an especially efficient interaction between her respiratory and cardiovascular systems.
“We have a new player in the game,” Dr. Araújo said.
The sources in this study report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to clinicians who champion the assessment. The COP is easier to obtain than cardiorespiratory measures that require people to exercise to their limit, advocates say; rather than running full speed, someone can walk or lightly jog on a treadmill, with a COP value obtained easily.
But other clinicians argue that maximal exercise tests have many prognostic benefits, and that physicians should do everything in their power to push patients to exercise as hard as possible. In particular, the VO2 max test captures the maximum amount of oxygen someone uses when exercising at their capacity and is the preferred method for measuring cardiovascular endurance.
The COP is a measure of the minimum number of liters of air during breathing required to move one liter of oxygen through the bloodstream. The lower the COP the better, because this means that someone is working less strenuously than someone else to transport the same amount of oxygen, denoting a more efficient interaction between their heart and lungs.
The COP for a fit person might be 15, about 20-25 for a healthy person, and 35 for someone with heart failure, according to Claudio Gil Araújo, MD, PhD, director of research and education at CLINIMEX, an exercise medicine clinic in Rio de Janeiro.
“Max VO2 is very important, that’s indisputable. But when do you use max VO2 in your daily life? Never,” Dr. Araújo said. But almost anyone can generate a COP.
Emerging uses for the COP
“I can put someone on the treadmill or bike, and after 3 or 4 minutes I have the COP. It’s like a walking pace,” Dr. Araújo said. Yet the values are obtained with roughly half the effort as VO2 max. Other clinicians argue exercising to the limits of endurance offers unique clinical insights.
“We should do everything in our power to exercise our patients to maximum. How long a patient is able to go is really important,” said Anu Lala, MD, a cardiologist who specializes in heart failure treatment at Mount Sinai Hospital in New York. A full-capacity exercise test gives useful insights into someone’s heart rate, heart rate recovery, blood pressure, and ECG response to vigorous exercise, Dr. Lala added, all of which are important clues to someone’s overall health.
In 2012 Dr. Araújo coauthored a study that first defined the COP, which is calculated by measuring expired gasses people produce while gently exercising, perhaps to the point where they begin to perspire, and then dividing their breathing capacity by their oxygen uptake every minute. The lowest value obtained during any exercise session is the COP.
Various studies show that higher COP values are associated with more severe heart lesions in patients with congenital heart disease; higher levels of mortality in seemingly healthy male adults; and with worse prognoses in patients with heart failure. These studies all appeared within the last 7 months.
The mortality study, which Dr. Araújo coauthored, compared COP in more than 3,000 U.S. men and women who completed an exercise test from 1973 to 2018 and were tracked for an average of 23 years. Although COP was introduced as an assessment in 2012, calculating the value from tests prior to that date was possible because those tests had captured the relevant breathing rate and oxygen uptake. In males aged 18-85 years, a worse COP was significantly associated with an increased risk for earlier death. This finding did not hold for females, however; Dr. Araújo noted that more research is needed to understand the discrepancy in COP’s predictive power by sex.
In the heart failure study, everyone enrolled had heart failure and completed a COP test. People with the worse COPs also had the worst symptoms of heart failure, but completing an exercise rehabilitation program improved COP values when researchers measured them again. Dr. Araújo was also part of this study, based in the Netherlands.
“I think the COP could become a novel parameter in clinical care,” for most people, said Thijs Eijsvogels, PhD, an exercise physiologist at Radboud University in Nijmegen, the Netherlands, and the senior author of the heart failure study. That said, Dr. Eijsvogels said elite athletes will always be more interested in measuring VO2 max.
Dr. Lala agreed that tests such as the COP have some value. Her own work has shown that measuring the efficiency of someone’s breathing patterns for exhaling carbon dioxide, which can also be done without making people exercise full strength, has prognostic value for patients with advanced heart failure. Even so, she said she would like to see maximal effort tests used as much as possible.
“I worry about saying we’re going to settle for a parameter that can be achieved at 50% of peak VO2 and then we don’t exercise our patients,” Dr. Lala said.
Dr. Araújo said he plans to continue to measure VO2 max but he believes COP has utility – even for elite athletes. One of his patients is a frequent Ironman competitor who competes well despite having a solid but not amazing VO2 max level. But her COP is quite low, Dr. Araújo said, which to him suggests an especially efficient interaction between her respiratory and cardiovascular systems.
“We have a new player in the game,” Dr. Araújo said.
The sources in this study report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Low-dose oral minoxidil for hair loss soars after NYT article
.
The weekly rate of first-time low-dose oral minoxidil (LDOM) prescriptions per 10,000 outpatient encounters was “significantly higher 8 weeks after vs. 8 weeks before article publication,” at 0.9 prescriptions, compared with 0.5 per 10,000, wrote the authors of the research letter, published in JAMA Network Open. There was no similar bump for first-time finasteride or hypertension prescriptions, wrote the authors, from Harvard Medical School and Massachusetts General Hospital, Boston, and Truveta, a company that provides EHR data from U.S. health care systems.
The New York Times article noted that LDOM was relatively unknown to patients and doctors – and not approved by the Food and Drug Administration for treating hair loss – but that it was inexpensive, safe, and very effective for many individuals. “The article did not report new research findings or large-scale randomized evidence,” wrote the authors of the JAMA study.
Rodney Sinclair, MD, professor of dermatology at the University of Melbourne, who conducted the original research on LDOM and hair loss and was quoted in the Times story, told this news organization that “the sharp uplift after the New York Times article was on the back of a gradual increase.” He added that “the momentum for minoxidil prescriptions is increasing,” so much so that it has led to a global shortage of LDOM. The drug appears to still be widely available in the United States, however. It is not on the ASHP shortages list.
“There has been growing momentum for minoxidil use since I first presented our data about 6 years ago,” Dr. Sinclair said. He noted that 2022 International Society of Hair Restoration Surgery survey data found that 26% of treating physicians always or often prescribed off-label oral minoxidil, up from 10% in 2019 and 0% in 2017, while another 20% said they prescribed it sometimes.
The authors of the new study looked at prescriptions for patients at eight health care systems before and after the Times article was published in August 2022. They calculated the rate of first-time oral minoxidil prescriptions for 2.5 mg and 5 mg tablets, excluding 10 mg tablets, which are prescribed for hypertension.
Among those receiving first-time prescriptions, 2,846 received them in the 7 months before the article and 3,695 in the 5 months after publication. Men (43.6% after vs. 37.7% before publication) and White individuals (68.6% after vs. 60.8% before publication) accounted for a higher proportion of prescriptions after the article was published. There was a 2.4-fold increase in first-time prescriptions among men, and a 1.7-fold increase among females, while people with comorbidities accounted for a smaller proportion after the publication.
“Socioeconomic factors, such as access to health care and education and income levels, may be associated with individuals seeking low-dose oral minoxidil after article publication,” wrote the authors.
In an interview, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said that he was not surprised to see an uptick in prescriptions after the Times article.
He and his colleagues were curious as to whether the article might have prompted newfound interest in LDOM. They experienced an uptick at George Washington, which Dr. Friedman thought could have been because he was quoted in the Times story. He and colleagues conducted a national survey of dermatologists asking if more patients had called, emailed, or come in to the office asking about LDOM after the article’s publication. “Over 85% said yes,” Dr. Friedman said in the interview. He and his coauthors also found a huge increase in Google searches for terms such as hair loss, alopecia, and minoxidil in the weeks after the article, he said.
The results are expected to published soon in the Journal of Drugs in Dermatology.
“I think a lot of people know about [LDOM] and it’s certainly has gained a lot more attention and acceptance in recent years,” said Dr. Friedman, but he added that “there’s no question” that the Times article increased interest.
That is not necessarily a bad thing, he said. “With one article, education on a common disease was disseminated worldwide in a way that no one doctor can do,” he said. The article was truthful, evidence-based, and included expert dermatologists, he noted.
“It probably got people who never thought twice about their hair thinning to actually think that there’s hope,” he said, adding that it also likely prompted them to seek care, and, more importantly, “to seek care from the person who should be taking care of this, which is the dermatologist.”
However, the article might also inspire some people to think LDOM can help when it can’t, or they might insist on a prescription when another medication is more appropriate, said Dr. Friedman.
Both he and Dr. Sinclair expect demand for LDOM to continue increasing.
“Word of mouth will drive the next wave of prescriptions,” said Dr. Sinclair. “We are continuing to do work to improve safety, to understand its mechanism of action, and identify ways to improve equity of access to treatment for men and women who are concerned about their hair loss and motivated to treat it,” he said.
Dr. Sinclair and Dr. Friedman report no relevant financial relationships.
.
The weekly rate of first-time low-dose oral minoxidil (LDOM) prescriptions per 10,000 outpatient encounters was “significantly higher 8 weeks after vs. 8 weeks before article publication,” at 0.9 prescriptions, compared with 0.5 per 10,000, wrote the authors of the research letter, published in JAMA Network Open. There was no similar bump for first-time finasteride or hypertension prescriptions, wrote the authors, from Harvard Medical School and Massachusetts General Hospital, Boston, and Truveta, a company that provides EHR data from U.S. health care systems.
The New York Times article noted that LDOM was relatively unknown to patients and doctors – and not approved by the Food and Drug Administration for treating hair loss – but that it was inexpensive, safe, and very effective for many individuals. “The article did not report new research findings or large-scale randomized evidence,” wrote the authors of the JAMA study.
Rodney Sinclair, MD, professor of dermatology at the University of Melbourne, who conducted the original research on LDOM and hair loss and was quoted in the Times story, told this news organization that “the sharp uplift after the New York Times article was on the back of a gradual increase.” He added that “the momentum for minoxidil prescriptions is increasing,” so much so that it has led to a global shortage of LDOM. The drug appears to still be widely available in the United States, however. It is not on the ASHP shortages list.
“There has been growing momentum for minoxidil use since I first presented our data about 6 years ago,” Dr. Sinclair said. He noted that 2022 International Society of Hair Restoration Surgery survey data found that 26% of treating physicians always or often prescribed off-label oral minoxidil, up from 10% in 2019 and 0% in 2017, while another 20% said they prescribed it sometimes.
The authors of the new study looked at prescriptions for patients at eight health care systems before and after the Times article was published in August 2022. They calculated the rate of first-time oral minoxidil prescriptions for 2.5 mg and 5 mg tablets, excluding 10 mg tablets, which are prescribed for hypertension.
Among those receiving first-time prescriptions, 2,846 received them in the 7 months before the article and 3,695 in the 5 months after publication. Men (43.6% after vs. 37.7% before publication) and White individuals (68.6% after vs. 60.8% before publication) accounted for a higher proportion of prescriptions after the article was published. There was a 2.4-fold increase in first-time prescriptions among men, and a 1.7-fold increase among females, while people with comorbidities accounted for a smaller proportion after the publication.
“Socioeconomic factors, such as access to health care and education and income levels, may be associated with individuals seeking low-dose oral minoxidil after article publication,” wrote the authors.
In an interview, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said that he was not surprised to see an uptick in prescriptions after the Times article.
He and his colleagues were curious as to whether the article might have prompted newfound interest in LDOM. They experienced an uptick at George Washington, which Dr. Friedman thought could have been because he was quoted in the Times story. He and colleagues conducted a national survey of dermatologists asking if more patients had called, emailed, or come in to the office asking about LDOM after the article’s publication. “Over 85% said yes,” Dr. Friedman said in the interview. He and his coauthors also found a huge increase in Google searches for terms such as hair loss, alopecia, and minoxidil in the weeks after the article, he said.
The results are expected to published soon in the Journal of Drugs in Dermatology.
“I think a lot of people know about [LDOM] and it’s certainly has gained a lot more attention and acceptance in recent years,” said Dr. Friedman, but he added that “there’s no question” that the Times article increased interest.
That is not necessarily a bad thing, he said. “With one article, education on a common disease was disseminated worldwide in a way that no one doctor can do,” he said. The article was truthful, evidence-based, and included expert dermatologists, he noted.
“It probably got people who never thought twice about their hair thinning to actually think that there’s hope,” he said, adding that it also likely prompted them to seek care, and, more importantly, “to seek care from the person who should be taking care of this, which is the dermatologist.”
However, the article might also inspire some people to think LDOM can help when it can’t, or they might insist on a prescription when another medication is more appropriate, said Dr. Friedman.
Both he and Dr. Sinclair expect demand for LDOM to continue increasing.
“Word of mouth will drive the next wave of prescriptions,” said Dr. Sinclair. “We are continuing to do work to improve safety, to understand its mechanism of action, and identify ways to improve equity of access to treatment for men and women who are concerned about their hair loss and motivated to treat it,” he said.
Dr. Sinclair and Dr. Friedman report no relevant financial relationships.
.
The weekly rate of first-time low-dose oral minoxidil (LDOM) prescriptions per 10,000 outpatient encounters was “significantly higher 8 weeks after vs. 8 weeks before article publication,” at 0.9 prescriptions, compared with 0.5 per 10,000, wrote the authors of the research letter, published in JAMA Network Open. There was no similar bump for first-time finasteride or hypertension prescriptions, wrote the authors, from Harvard Medical School and Massachusetts General Hospital, Boston, and Truveta, a company that provides EHR data from U.S. health care systems.
The New York Times article noted that LDOM was relatively unknown to patients and doctors – and not approved by the Food and Drug Administration for treating hair loss – but that it was inexpensive, safe, and very effective for many individuals. “The article did not report new research findings or large-scale randomized evidence,” wrote the authors of the JAMA study.
Rodney Sinclair, MD, professor of dermatology at the University of Melbourne, who conducted the original research on LDOM and hair loss and was quoted in the Times story, told this news organization that “the sharp uplift after the New York Times article was on the back of a gradual increase.” He added that “the momentum for minoxidil prescriptions is increasing,” so much so that it has led to a global shortage of LDOM. The drug appears to still be widely available in the United States, however. It is not on the ASHP shortages list.
“There has been growing momentum for minoxidil use since I first presented our data about 6 years ago,” Dr. Sinclair said. He noted that 2022 International Society of Hair Restoration Surgery survey data found that 26% of treating physicians always or often prescribed off-label oral minoxidil, up from 10% in 2019 and 0% in 2017, while another 20% said they prescribed it sometimes.
The authors of the new study looked at prescriptions for patients at eight health care systems before and after the Times article was published in August 2022. They calculated the rate of first-time oral minoxidil prescriptions for 2.5 mg and 5 mg tablets, excluding 10 mg tablets, which are prescribed for hypertension.
Among those receiving first-time prescriptions, 2,846 received them in the 7 months before the article and 3,695 in the 5 months after publication. Men (43.6% after vs. 37.7% before publication) and White individuals (68.6% after vs. 60.8% before publication) accounted for a higher proportion of prescriptions after the article was published. There was a 2.4-fold increase in first-time prescriptions among men, and a 1.7-fold increase among females, while people with comorbidities accounted for a smaller proportion after the publication.
“Socioeconomic factors, such as access to health care and education and income levels, may be associated with individuals seeking low-dose oral minoxidil after article publication,” wrote the authors.
In an interview, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said that he was not surprised to see an uptick in prescriptions after the Times article.
He and his colleagues were curious as to whether the article might have prompted newfound interest in LDOM. They experienced an uptick at George Washington, which Dr. Friedman thought could have been because he was quoted in the Times story. He and colleagues conducted a national survey of dermatologists asking if more patients had called, emailed, or come in to the office asking about LDOM after the article’s publication. “Over 85% said yes,” Dr. Friedman said in the interview. He and his coauthors also found a huge increase in Google searches for terms such as hair loss, alopecia, and minoxidil in the weeks after the article, he said.
The results are expected to published soon in the Journal of Drugs in Dermatology.
“I think a lot of people know about [LDOM] and it’s certainly has gained a lot more attention and acceptance in recent years,” said Dr. Friedman, but he added that “there’s no question” that the Times article increased interest.
That is not necessarily a bad thing, he said. “With one article, education on a common disease was disseminated worldwide in a way that no one doctor can do,” he said. The article was truthful, evidence-based, and included expert dermatologists, he noted.
“It probably got people who never thought twice about their hair thinning to actually think that there’s hope,” he said, adding that it also likely prompted them to seek care, and, more importantly, “to seek care from the person who should be taking care of this, which is the dermatologist.”
However, the article might also inspire some people to think LDOM can help when it can’t, or they might insist on a prescription when another medication is more appropriate, said Dr. Friedman.
Both he and Dr. Sinclair expect demand for LDOM to continue increasing.
“Word of mouth will drive the next wave of prescriptions,” said Dr. Sinclair. “We are continuing to do work to improve safety, to understand its mechanism of action, and identify ways to improve equity of access to treatment for men and women who are concerned about their hair loss and motivated to treat it,” he said.
Dr. Sinclair and Dr. Friedman report no relevant financial relationships.
FROM JAMA NETWORK OPEN
Cuffless blood pressure monitors: Still a numbers game
Medscape’s Editor-in-Chief Eric Topol, MD, referred to continual noninvasive, cuffless, accurate blood pressure devices as “a holy grail in sensor technology.”
He personally tested a cuff-calibrated, over-the-counter device available in Europe that claims to monitor daily blood pressure changes and produce data that can help physicians titrate medications.
Dr. Topol does not believe that it is ready for prime time. Yes, cuffless devices are easy to use, and generate lots of data. But are those data accurate?
Many experts say not yet, even as the market continues to grow and more devices are introduced and highlighted at high-profile consumer events.
Burned before
Limitations of cuffed devices are well known, including errors related to cuff size, patient positioning, patient habits or behaviors (for example, caffeine/nicotine use, acute meal digestion, full bladder, very recent physical activity) and clinicians’ failure to take accurate measurements.
Like many clinicians, Timothy B. Plante, MD, MHS, assistant professor at the University of Vermont Medical Center thrombosis & hemostasis program in Burlington, is very excited about cuffless technology. However, “we’ve been burned by it before,” he said in an interview.
Dr. Plante’s 2016 validation study of an instant blood pressure smartphone app found that its measurements were “highly inaccurate,” with such low sensitivity that more than three-quarters of individuals with hypertensive blood levels would be falsely reassured that their blood pressure was in the normal range.
His team’s 2023 review of the current landscape, which includes more sophisticated devices, concluded that accuracy remains an issue: “Unfortunately, the pace of regulation of these devices has failed to match the speed of innovation and direct availability to patient consumers. There is an urgent need to develop a consensus on standards by which cuffless BP devices can be tested for accuracy.”
Devices, indications differ
Cuffless devices estimate blood pressure indirectly. Most operate based on pulse wave analysis and pulse arrival time (PWA-PAT), explained Ramakrishna Mukkamala, PhD, in a commentary. Dr. Mukkamala is a professor in the departments of bioengineering and anesthesiology and perioperative medicine at the University of Pittsburgh.
PWA involves measuring a peripheral arterial waveform using an optical sensor such as the green lights on the back of a wrist-worn device, or a ‘force sensor’ such as a finger cuff or pressing on a smartphone. Certain features are extracted from the waveform using machine learning and calibrated to blood pressure values.
PAT techniques work together with PWA; they record the ECG and extract features from that signal as well as the arterial waveform for calibration to blood pressure values.
The algorithm used to generate the BP numbers comprises a proprietary baseline model that may include demographics and other patient characteristics. A cuff measurement is often part of the baseline model because most cuffless devices require periodic (typically weekly or monthly) calibration using a cuffed device.
Cuffless devices that require cuff calibration compare the estimate they get to the cuff-calibrated number. In this scenario, the cuffless device may come up with the same blood pressure numbers simply because the baseline model – which is made up of thousands of data points relevant to the patient – has not changed.
This has led some experts to question whether PWA-PAT cuffless device readings actually add anything to the baseline model.
They don’t, according to Microsoft Research in what Dr. Mukkamala and coauthors referred to (in a review published in Hypertension) as “a complex article describing perhaps the most important and highest resource project to date (Aurora Project) on assessing the accuracy of PWA and PWA devices.”
The Microsoft article was written for bioengineers. The review in Hypertension explains the project for clinicians, and concludes that, “Cuffless BP devices based on PWA and PWA-PAT, which are similar to some regulatory-cleared devices, were of no additional value in measuring auscultatory or 24-hour ambulatory cuff BP when compared with a baseline model in which BP was predicted without an actual measurement.”
IEEE and FDA validation
Despite these concerns, several cuffless devices using PWA and PAT have been cleared by the Food and Drug Administration.
Validating cuffless devices is no simple matter. The Institute of Electrical and Electronics Engineers published a validation protocol for cuffless blood pressure devices in 2014 that was amended in 2019 to include a requirement to evaluate performance in different positions and in the presence of motion with varying degrees of noise artifact.
However, Daichi Shimbo, MD, codirector of the Columbia Hypertension Center in New York and vice chair of the American Heart Association Statement on blood pressure monitoring, and colleagues point out limitations, even in the updated standard. These include not requiring evaluation for drift over time; lack of specific dynamic testing protocols for stressors such as exercise or environmental temperatures; and an unsuitable reference standard (oscillometric cuff-based devices) during movement.
Dr. Shimbo said in an interview that, although he is excited about them, “these cuffless devices are not aligned with regulatory bodies. If a device gives someone a wrong blood pressure, they might be diagnosed with hypertension when they don’t have it or might miss the fact that they’re hypertensive because they get a normal blood pressure reading. If there’s no yardstick by which you say these devices are good, what are we really doing – helping, or causing a problem?”
“The specifics of how a device estimates blood pressure can determine what testing is needed to ensure that it is providing accurate performance in the intended conditions of use,” Jeremy Kahn, an FDA press officer, said in an interview. “For example, for cuffless devices that are calibrated initially with a cuff-based blood pressure device, the cuffless device needs to specify the period over which it can provide accurate readings and have testing to demonstrate that it provides accurate results over that period of use.”
The FDA said its testing is different from what the Microsoft Aurora Project used in their study.
“The intent of that testing, as the agency understands it, is to evaluate whether the device is providing useful input based on the current physiology of the patient rather than relying on predetermined values based on calibration or patient attributes. We evaluate this clinically in two separate tests: an induced change in blood pressure test and tracking of natural blood pressure changes with longer term device use,” Mr. Kahn explained.
Analyzing a device’s performance on individuals who have had natural changes in blood pressure as compared to a calibration value or initial reading “can also help discern if the device is using physiological data from the patient to determine their blood pressure accurately,” he said.
Experts interviewed for this article who remain skeptical about cuffless BP monitoring question whether the numbers that appear during the induced blood pressure change, and with the natural blood pressure changes that may occur over time, accurately reflect a patient’s blood pressure.
“The FDA doesn’t approve these devices; they clear them,” Dr. Shimbo pointed out. “Clearing them means they can be sold to the general public in the U.S. It’s not a strong statement that they’re accurate.”
Moving toward validation, standards
Ultimately, cuffless BP monitors may require more than one validation protocol and standard, depending on their technology, how and where they will be used, and by whom.
And as Dr. Plante and colleagues write, “Importantly, validation should be performed in diverse and special populations, including pregnant women and individuals across a range of heart rates, skin tones, wrist sizes, common arrhythmias, and beta-blocker use.”
Organizations that might be expected to help move validation and standards forward have mostly remained silent. The American Medical Association’s US Blood Pressure Validated Device Listing website includes only cuffed devices, as does the website of the international scientific nonprofit STRIDE BP.
The European Society of Hypertension 2022 consensus statement on cuffless devices concluded that, until there is an internationally accepted accuracy standard and the devices have been tested in healthy people and those with suspected or diagnosed hypertension, “cuffless BP devices should not be used for the evaluation or management of hypertension in clinical practice.”
This month, ESH published recommendations for “specific, clinically meaningful, and pragmatic validation procedures for different types of intermittent cuffless devices” that will be presented at their upcoming annual meeting June 26.
Updated protocols from IEEE “are coming out soon,” according to Dr. Shimbo. The FDA says currently cleared devices won’t need to revalidate according to new standards unless the sponsor makes significant modifications in software algorithms, device hardware, or targeted patient populations.
Device makers take the initiative
In the face of conflicting reports on accuracy and lack of a robust standard, some device makers are publishing their own tests or encouraging validation by potential customers.
For example, institutions that are considering using the Biobeat cuffless blood pressure monitor watch “usually start with small pilots with our devices to do internal validation,” Lior Ben Shettrit, the company’s vice president of business development, said in an interview. “Only after they complete the internal validation are they willing to move forward to full implementation.”
Cardiologist Dean Nachman, MD, is leading validation studies of the Biobeat device at the Hadassah Ein Kerem Medical Center in Jerusalem. For the first validation, the team recruited 1,057 volunteers who did a single blood pressure measurement with the cuffless device and with a cuffed device.
“We found 96.3% agreement in identifying hypertension and an interclass correlation coefficient of 0.99 and 0.97 for systolic and diastolic measurements, respectively,” he said. “Then we took it to the next level and compared the device to ambulatory 24-hour blood pressure monitoring and found comparable measurements.”
The investigators are not done yet. “We need data from thousands of patients, with subgroups, to not have any concerns,” he says. “Right now, we are using the device as a general monitor – as an EKG plus heart rate plus oxygen saturation level monitor – and as a blood pressure monitor for 24-hour blood pressure monitoring.”
The developers of the Aktiia device, which is the one Dr. Topol tested, take a different perspective. “When somebody introduces a new technology that is disrupting something that has been in place for over 100 years, there will always be some grumblings, ruffling of feathers, people saying it’s not ready, it’s not ready, it’s not ready,” Aktiia’s chief medical officer Jay Shah, MD, noted.
“But a lot of those comments are coming from the isolation of an ivory tower,” he said.
Aktiia cofounder and chief technology officer Josep Solà said that “no device is probably as accurate as if you have an invasive catheter,” adding that “we engage patients to look at their blood pressure day by day. … If each individual measurement of each of those patient is slightly less accurate than a cuff, who cares? We have 40 measurements per day on each patient. The accuracy and precision of each of those is good.”
Researchers from the George Institute for Global Health recently compared the Aktiia device to conventional ambulatory monitoring in 41 patients and found that “it did not accurately track night-time BP decline and results suggested it was unable to track medication-induced BP changes.”
“In the context of 24/7 monitoring of hypertensive patients,” Mr. Solà said, “whatever you do, if it’s better than a sham device or a baseline model and you track the blood pressure changes, it’s a hundred times much better than doing nothing.”
Dr. Nachman and Dr. Plante reported no relevant financial relationships. Dr. Shimbo reported that he received funding from NIH and has consulted for Abbott Vascular, Edward Lifesciences, Medtronic, and Tryton Medical.
A version of this article first appeared on Medscape.com.
Medscape’s Editor-in-Chief Eric Topol, MD, referred to continual noninvasive, cuffless, accurate blood pressure devices as “a holy grail in sensor technology.”
He personally tested a cuff-calibrated, over-the-counter device available in Europe that claims to monitor daily blood pressure changes and produce data that can help physicians titrate medications.
Dr. Topol does not believe that it is ready for prime time. Yes, cuffless devices are easy to use, and generate lots of data. But are those data accurate?
Many experts say not yet, even as the market continues to grow and more devices are introduced and highlighted at high-profile consumer events.
Burned before
Limitations of cuffed devices are well known, including errors related to cuff size, patient positioning, patient habits or behaviors (for example, caffeine/nicotine use, acute meal digestion, full bladder, very recent physical activity) and clinicians’ failure to take accurate measurements.
Like many clinicians, Timothy B. Plante, MD, MHS, assistant professor at the University of Vermont Medical Center thrombosis & hemostasis program in Burlington, is very excited about cuffless technology. However, “we’ve been burned by it before,” he said in an interview.
Dr. Plante’s 2016 validation study of an instant blood pressure smartphone app found that its measurements were “highly inaccurate,” with such low sensitivity that more than three-quarters of individuals with hypertensive blood levels would be falsely reassured that their blood pressure was in the normal range.
His team’s 2023 review of the current landscape, which includes more sophisticated devices, concluded that accuracy remains an issue: “Unfortunately, the pace of regulation of these devices has failed to match the speed of innovation and direct availability to patient consumers. There is an urgent need to develop a consensus on standards by which cuffless BP devices can be tested for accuracy.”
Devices, indications differ
Cuffless devices estimate blood pressure indirectly. Most operate based on pulse wave analysis and pulse arrival time (PWA-PAT), explained Ramakrishna Mukkamala, PhD, in a commentary. Dr. Mukkamala is a professor in the departments of bioengineering and anesthesiology and perioperative medicine at the University of Pittsburgh.
PWA involves measuring a peripheral arterial waveform using an optical sensor such as the green lights on the back of a wrist-worn device, or a ‘force sensor’ such as a finger cuff or pressing on a smartphone. Certain features are extracted from the waveform using machine learning and calibrated to blood pressure values.
PAT techniques work together with PWA; they record the ECG and extract features from that signal as well as the arterial waveform for calibration to blood pressure values.
The algorithm used to generate the BP numbers comprises a proprietary baseline model that may include demographics and other patient characteristics. A cuff measurement is often part of the baseline model because most cuffless devices require periodic (typically weekly or monthly) calibration using a cuffed device.
Cuffless devices that require cuff calibration compare the estimate they get to the cuff-calibrated number. In this scenario, the cuffless device may come up with the same blood pressure numbers simply because the baseline model – which is made up of thousands of data points relevant to the patient – has not changed.
This has led some experts to question whether PWA-PAT cuffless device readings actually add anything to the baseline model.
They don’t, according to Microsoft Research in what Dr. Mukkamala and coauthors referred to (in a review published in Hypertension) as “a complex article describing perhaps the most important and highest resource project to date (Aurora Project) on assessing the accuracy of PWA and PWA devices.”
The Microsoft article was written for bioengineers. The review in Hypertension explains the project for clinicians, and concludes that, “Cuffless BP devices based on PWA and PWA-PAT, which are similar to some regulatory-cleared devices, were of no additional value in measuring auscultatory or 24-hour ambulatory cuff BP when compared with a baseline model in which BP was predicted without an actual measurement.”
IEEE and FDA validation
Despite these concerns, several cuffless devices using PWA and PAT have been cleared by the Food and Drug Administration.
Validating cuffless devices is no simple matter. The Institute of Electrical and Electronics Engineers published a validation protocol for cuffless blood pressure devices in 2014 that was amended in 2019 to include a requirement to evaluate performance in different positions and in the presence of motion with varying degrees of noise artifact.
However, Daichi Shimbo, MD, codirector of the Columbia Hypertension Center in New York and vice chair of the American Heart Association Statement on blood pressure monitoring, and colleagues point out limitations, even in the updated standard. These include not requiring evaluation for drift over time; lack of specific dynamic testing protocols for stressors such as exercise or environmental temperatures; and an unsuitable reference standard (oscillometric cuff-based devices) during movement.
Dr. Shimbo said in an interview that, although he is excited about them, “these cuffless devices are not aligned with regulatory bodies. If a device gives someone a wrong blood pressure, they might be diagnosed with hypertension when they don’t have it or might miss the fact that they’re hypertensive because they get a normal blood pressure reading. If there’s no yardstick by which you say these devices are good, what are we really doing – helping, or causing a problem?”
“The specifics of how a device estimates blood pressure can determine what testing is needed to ensure that it is providing accurate performance in the intended conditions of use,” Jeremy Kahn, an FDA press officer, said in an interview. “For example, for cuffless devices that are calibrated initially with a cuff-based blood pressure device, the cuffless device needs to specify the period over which it can provide accurate readings and have testing to demonstrate that it provides accurate results over that period of use.”
The FDA said its testing is different from what the Microsoft Aurora Project used in their study.
“The intent of that testing, as the agency understands it, is to evaluate whether the device is providing useful input based on the current physiology of the patient rather than relying on predetermined values based on calibration or patient attributes. We evaluate this clinically in two separate tests: an induced change in blood pressure test and tracking of natural blood pressure changes with longer term device use,” Mr. Kahn explained.
Analyzing a device’s performance on individuals who have had natural changes in blood pressure as compared to a calibration value or initial reading “can also help discern if the device is using physiological data from the patient to determine their blood pressure accurately,” he said.
Experts interviewed for this article who remain skeptical about cuffless BP monitoring question whether the numbers that appear during the induced blood pressure change, and with the natural blood pressure changes that may occur over time, accurately reflect a patient’s blood pressure.
“The FDA doesn’t approve these devices; they clear them,” Dr. Shimbo pointed out. “Clearing them means they can be sold to the general public in the U.S. It’s not a strong statement that they’re accurate.”
Moving toward validation, standards
Ultimately, cuffless BP monitors may require more than one validation protocol and standard, depending on their technology, how and where they will be used, and by whom.
And as Dr. Plante and colleagues write, “Importantly, validation should be performed in diverse and special populations, including pregnant women and individuals across a range of heart rates, skin tones, wrist sizes, common arrhythmias, and beta-blocker use.”
Organizations that might be expected to help move validation and standards forward have mostly remained silent. The American Medical Association’s US Blood Pressure Validated Device Listing website includes only cuffed devices, as does the website of the international scientific nonprofit STRIDE BP.
The European Society of Hypertension 2022 consensus statement on cuffless devices concluded that, until there is an internationally accepted accuracy standard and the devices have been tested in healthy people and those with suspected or diagnosed hypertension, “cuffless BP devices should not be used for the evaluation or management of hypertension in clinical practice.”
This month, ESH published recommendations for “specific, clinically meaningful, and pragmatic validation procedures for different types of intermittent cuffless devices” that will be presented at their upcoming annual meeting June 26.
Updated protocols from IEEE “are coming out soon,” according to Dr. Shimbo. The FDA says currently cleared devices won’t need to revalidate according to new standards unless the sponsor makes significant modifications in software algorithms, device hardware, or targeted patient populations.
Device makers take the initiative
In the face of conflicting reports on accuracy and lack of a robust standard, some device makers are publishing their own tests or encouraging validation by potential customers.
For example, institutions that are considering using the Biobeat cuffless blood pressure monitor watch “usually start with small pilots with our devices to do internal validation,” Lior Ben Shettrit, the company’s vice president of business development, said in an interview. “Only after they complete the internal validation are they willing to move forward to full implementation.”
Cardiologist Dean Nachman, MD, is leading validation studies of the Biobeat device at the Hadassah Ein Kerem Medical Center in Jerusalem. For the first validation, the team recruited 1,057 volunteers who did a single blood pressure measurement with the cuffless device and with a cuffed device.
“We found 96.3% agreement in identifying hypertension and an interclass correlation coefficient of 0.99 and 0.97 for systolic and diastolic measurements, respectively,” he said. “Then we took it to the next level and compared the device to ambulatory 24-hour blood pressure monitoring and found comparable measurements.”
The investigators are not done yet. “We need data from thousands of patients, with subgroups, to not have any concerns,” he says. “Right now, we are using the device as a general monitor – as an EKG plus heart rate plus oxygen saturation level monitor – and as a blood pressure monitor for 24-hour blood pressure monitoring.”
The developers of the Aktiia device, which is the one Dr. Topol tested, take a different perspective. “When somebody introduces a new technology that is disrupting something that has been in place for over 100 years, there will always be some grumblings, ruffling of feathers, people saying it’s not ready, it’s not ready, it’s not ready,” Aktiia’s chief medical officer Jay Shah, MD, noted.
“But a lot of those comments are coming from the isolation of an ivory tower,” he said.
Aktiia cofounder and chief technology officer Josep Solà said that “no device is probably as accurate as if you have an invasive catheter,” adding that “we engage patients to look at their blood pressure day by day. … If each individual measurement of each of those patient is slightly less accurate than a cuff, who cares? We have 40 measurements per day on each patient. The accuracy and precision of each of those is good.”
Researchers from the George Institute for Global Health recently compared the Aktiia device to conventional ambulatory monitoring in 41 patients and found that “it did not accurately track night-time BP decline and results suggested it was unable to track medication-induced BP changes.”
“In the context of 24/7 monitoring of hypertensive patients,” Mr. Solà said, “whatever you do, if it’s better than a sham device or a baseline model and you track the blood pressure changes, it’s a hundred times much better than doing nothing.”
Dr. Nachman and Dr. Plante reported no relevant financial relationships. Dr. Shimbo reported that he received funding from NIH and has consulted for Abbott Vascular, Edward Lifesciences, Medtronic, and Tryton Medical.
A version of this article first appeared on Medscape.com.
Medscape’s Editor-in-Chief Eric Topol, MD, referred to continual noninvasive, cuffless, accurate blood pressure devices as “a holy grail in sensor technology.”
He personally tested a cuff-calibrated, over-the-counter device available in Europe that claims to monitor daily blood pressure changes and produce data that can help physicians titrate medications.
Dr. Topol does not believe that it is ready for prime time. Yes, cuffless devices are easy to use, and generate lots of data. But are those data accurate?
Many experts say not yet, even as the market continues to grow and more devices are introduced and highlighted at high-profile consumer events.
Burned before
Limitations of cuffed devices are well known, including errors related to cuff size, patient positioning, patient habits or behaviors (for example, caffeine/nicotine use, acute meal digestion, full bladder, very recent physical activity) and clinicians’ failure to take accurate measurements.
Like many clinicians, Timothy B. Plante, MD, MHS, assistant professor at the University of Vermont Medical Center thrombosis & hemostasis program in Burlington, is very excited about cuffless technology. However, “we’ve been burned by it before,” he said in an interview.
Dr. Plante’s 2016 validation study of an instant blood pressure smartphone app found that its measurements were “highly inaccurate,” with such low sensitivity that more than three-quarters of individuals with hypertensive blood levels would be falsely reassured that their blood pressure was in the normal range.
His team’s 2023 review of the current landscape, which includes more sophisticated devices, concluded that accuracy remains an issue: “Unfortunately, the pace of regulation of these devices has failed to match the speed of innovation and direct availability to patient consumers. There is an urgent need to develop a consensus on standards by which cuffless BP devices can be tested for accuracy.”
Devices, indications differ
Cuffless devices estimate blood pressure indirectly. Most operate based on pulse wave analysis and pulse arrival time (PWA-PAT), explained Ramakrishna Mukkamala, PhD, in a commentary. Dr. Mukkamala is a professor in the departments of bioengineering and anesthesiology and perioperative medicine at the University of Pittsburgh.
PWA involves measuring a peripheral arterial waveform using an optical sensor such as the green lights on the back of a wrist-worn device, or a ‘force sensor’ such as a finger cuff or pressing on a smartphone. Certain features are extracted from the waveform using machine learning and calibrated to blood pressure values.
PAT techniques work together with PWA; they record the ECG and extract features from that signal as well as the arterial waveform for calibration to blood pressure values.
The algorithm used to generate the BP numbers comprises a proprietary baseline model that may include demographics and other patient characteristics. A cuff measurement is often part of the baseline model because most cuffless devices require periodic (typically weekly or monthly) calibration using a cuffed device.
Cuffless devices that require cuff calibration compare the estimate they get to the cuff-calibrated number. In this scenario, the cuffless device may come up with the same blood pressure numbers simply because the baseline model – which is made up of thousands of data points relevant to the patient – has not changed.
This has led some experts to question whether PWA-PAT cuffless device readings actually add anything to the baseline model.
They don’t, according to Microsoft Research in what Dr. Mukkamala and coauthors referred to (in a review published in Hypertension) as “a complex article describing perhaps the most important and highest resource project to date (Aurora Project) on assessing the accuracy of PWA and PWA devices.”
The Microsoft article was written for bioengineers. The review in Hypertension explains the project for clinicians, and concludes that, “Cuffless BP devices based on PWA and PWA-PAT, which are similar to some regulatory-cleared devices, were of no additional value in measuring auscultatory or 24-hour ambulatory cuff BP when compared with a baseline model in which BP was predicted without an actual measurement.”
IEEE and FDA validation
Despite these concerns, several cuffless devices using PWA and PAT have been cleared by the Food and Drug Administration.
Validating cuffless devices is no simple matter. The Institute of Electrical and Electronics Engineers published a validation protocol for cuffless blood pressure devices in 2014 that was amended in 2019 to include a requirement to evaluate performance in different positions and in the presence of motion with varying degrees of noise artifact.
However, Daichi Shimbo, MD, codirector of the Columbia Hypertension Center in New York and vice chair of the American Heart Association Statement on blood pressure monitoring, and colleagues point out limitations, even in the updated standard. These include not requiring evaluation for drift over time; lack of specific dynamic testing protocols for stressors such as exercise or environmental temperatures; and an unsuitable reference standard (oscillometric cuff-based devices) during movement.
Dr. Shimbo said in an interview that, although he is excited about them, “these cuffless devices are not aligned with regulatory bodies. If a device gives someone a wrong blood pressure, they might be diagnosed with hypertension when they don’t have it or might miss the fact that they’re hypertensive because they get a normal blood pressure reading. If there’s no yardstick by which you say these devices are good, what are we really doing – helping, or causing a problem?”
“The specifics of how a device estimates blood pressure can determine what testing is needed to ensure that it is providing accurate performance in the intended conditions of use,” Jeremy Kahn, an FDA press officer, said in an interview. “For example, for cuffless devices that are calibrated initially with a cuff-based blood pressure device, the cuffless device needs to specify the period over which it can provide accurate readings and have testing to demonstrate that it provides accurate results over that period of use.”
The FDA said its testing is different from what the Microsoft Aurora Project used in their study.
“The intent of that testing, as the agency understands it, is to evaluate whether the device is providing useful input based on the current physiology of the patient rather than relying on predetermined values based on calibration or patient attributes. We evaluate this clinically in two separate tests: an induced change in blood pressure test and tracking of natural blood pressure changes with longer term device use,” Mr. Kahn explained.
Analyzing a device’s performance on individuals who have had natural changes in blood pressure as compared to a calibration value or initial reading “can also help discern if the device is using physiological data from the patient to determine their blood pressure accurately,” he said.
Experts interviewed for this article who remain skeptical about cuffless BP monitoring question whether the numbers that appear during the induced blood pressure change, and with the natural blood pressure changes that may occur over time, accurately reflect a patient’s blood pressure.
“The FDA doesn’t approve these devices; they clear them,” Dr. Shimbo pointed out. “Clearing them means they can be sold to the general public in the U.S. It’s not a strong statement that they’re accurate.”
Moving toward validation, standards
Ultimately, cuffless BP monitors may require more than one validation protocol and standard, depending on their technology, how and where they will be used, and by whom.
And as Dr. Plante and colleagues write, “Importantly, validation should be performed in diverse and special populations, including pregnant women and individuals across a range of heart rates, skin tones, wrist sizes, common arrhythmias, and beta-blocker use.”
Organizations that might be expected to help move validation and standards forward have mostly remained silent. The American Medical Association’s US Blood Pressure Validated Device Listing website includes only cuffed devices, as does the website of the international scientific nonprofit STRIDE BP.
The European Society of Hypertension 2022 consensus statement on cuffless devices concluded that, until there is an internationally accepted accuracy standard and the devices have been tested in healthy people and those with suspected or diagnosed hypertension, “cuffless BP devices should not be used for the evaluation or management of hypertension in clinical practice.”
This month, ESH published recommendations for “specific, clinically meaningful, and pragmatic validation procedures for different types of intermittent cuffless devices” that will be presented at their upcoming annual meeting June 26.
Updated protocols from IEEE “are coming out soon,” according to Dr. Shimbo. The FDA says currently cleared devices won’t need to revalidate according to new standards unless the sponsor makes significant modifications in software algorithms, device hardware, or targeted patient populations.
Device makers take the initiative
In the face of conflicting reports on accuracy and lack of a robust standard, some device makers are publishing their own tests or encouraging validation by potential customers.
For example, institutions that are considering using the Biobeat cuffless blood pressure monitor watch “usually start with small pilots with our devices to do internal validation,” Lior Ben Shettrit, the company’s vice president of business development, said in an interview. “Only after they complete the internal validation are they willing to move forward to full implementation.”
Cardiologist Dean Nachman, MD, is leading validation studies of the Biobeat device at the Hadassah Ein Kerem Medical Center in Jerusalem. For the first validation, the team recruited 1,057 volunteers who did a single blood pressure measurement with the cuffless device and with a cuffed device.
“We found 96.3% agreement in identifying hypertension and an interclass correlation coefficient of 0.99 and 0.97 for systolic and diastolic measurements, respectively,” he said. “Then we took it to the next level and compared the device to ambulatory 24-hour blood pressure monitoring and found comparable measurements.”
The investigators are not done yet. “We need data from thousands of patients, with subgroups, to not have any concerns,” he says. “Right now, we are using the device as a general monitor – as an EKG plus heart rate plus oxygen saturation level monitor – and as a blood pressure monitor for 24-hour blood pressure monitoring.”
The developers of the Aktiia device, which is the one Dr. Topol tested, take a different perspective. “When somebody introduces a new technology that is disrupting something that has been in place for over 100 years, there will always be some grumblings, ruffling of feathers, people saying it’s not ready, it’s not ready, it’s not ready,” Aktiia’s chief medical officer Jay Shah, MD, noted.
“But a lot of those comments are coming from the isolation of an ivory tower,” he said.
Aktiia cofounder and chief technology officer Josep Solà said that “no device is probably as accurate as if you have an invasive catheter,” adding that “we engage patients to look at their blood pressure day by day. … If each individual measurement of each of those patient is slightly less accurate than a cuff, who cares? We have 40 measurements per day on each patient. The accuracy and precision of each of those is good.”
Researchers from the George Institute for Global Health recently compared the Aktiia device to conventional ambulatory monitoring in 41 patients and found that “it did not accurately track night-time BP decline and results suggested it was unable to track medication-induced BP changes.”
“In the context of 24/7 monitoring of hypertensive patients,” Mr. Solà said, “whatever you do, if it’s better than a sham device or a baseline model and you track the blood pressure changes, it’s a hundred times much better than doing nothing.”
Dr. Nachman and Dr. Plante reported no relevant financial relationships. Dr. Shimbo reported that he received funding from NIH and has consulted for Abbott Vascular, Edward Lifesciences, Medtronic, and Tryton Medical.
A version of this article first appeared on Medscape.com.
Early hysterectomy linked to higher CVD, stroke risk
TOPLINE:
METHODOLOGY:
- Risk of CVD rapidly increases after menopause, possibly owing to loss of protective effects of female sex hormones and hemorheologic changes.
- Results of previous studies of the association between hysterectomy and CVD were mixed.
- Using national health insurance data, this cohort study included 55,539 South Korean women (median age, 45 years) who underwent a hysterectomy and a propensity-matched group of women.
- The primary outcome was CVD, including myocardial infarction (MI), coronary artery revascularization, and stroke.
TAKEAWAY:
- During follow-up of just under 8 years, the hysterectomy group had an increased risk of CVD compared with the non-hysterectomy group (hazard ratio [HR] 1.25; 95% confidence interval [CI], 1.09-1.44; P = .002)
- The incidence of MI and coronary revascularization was comparable between groups, but the risk of stroke was significantly higher among those who had had a hysterectomy (HR, 1.31; 95% CI, 1.12-1.53; P < .001)
- This increase in risk was similar after excluding patients who also underwent adnexal surgery.
IN PRACTICE:
Early hysterectomy was linked to higher CVD risk, especially stroke, but since the CVD incidence wasn’t high, a change in clinical practice may not be needed, said the authors.
STUDY DETAILS:
The study was conducted by Jin-Sung Yuk, MD, PhD, Department of Obstetrics and Gynecology, Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Republic of Korea, and colleagues. It was published online June 12 in JAMA Network Open.
LIMITATIONS:
The study was retrospective and observational and used administrative databases that may be prone to inaccurate coding. The findings may not be generalizable outside Korea.
DISCLOSURES:
The study was supported by a National Research Foundation of Korea grant funded by the Korea government. The authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Risk of CVD rapidly increases after menopause, possibly owing to loss of protective effects of female sex hormones and hemorheologic changes.
- Results of previous studies of the association between hysterectomy and CVD were mixed.
- Using national health insurance data, this cohort study included 55,539 South Korean women (median age, 45 years) who underwent a hysterectomy and a propensity-matched group of women.
- The primary outcome was CVD, including myocardial infarction (MI), coronary artery revascularization, and stroke.
TAKEAWAY:
- During follow-up of just under 8 years, the hysterectomy group had an increased risk of CVD compared with the non-hysterectomy group (hazard ratio [HR] 1.25; 95% confidence interval [CI], 1.09-1.44; P = .002)
- The incidence of MI and coronary revascularization was comparable between groups, but the risk of stroke was significantly higher among those who had had a hysterectomy (HR, 1.31; 95% CI, 1.12-1.53; P < .001)
- This increase in risk was similar after excluding patients who also underwent adnexal surgery.
IN PRACTICE:
Early hysterectomy was linked to higher CVD risk, especially stroke, but since the CVD incidence wasn’t high, a change in clinical practice may not be needed, said the authors.
STUDY DETAILS:
The study was conducted by Jin-Sung Yuk, MD, PhD, Department of Obstetrics and Gynecology, Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Republic of Korea, and colleagues. It was published online June 12 in JAMA Network Open.
LIMITATIONS:
The study was retrospective and observational and used administrative databases that may be prone to inaccurate coding. The findings may not be generalizable outside Korea.
DISCLOSURES:
The study was supported by a National Research Foundation of Korea grant funded by the Korea government. The authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Risk of CVD rapidly increases after menopause, possibly owing to loss of protective effects of female sex hormones and hemorheologic changes.
- Results of previous studies of the association between hysterectomy and CVD were mixed.
- Using national health insurance data, this cohort study included 55,539 South Korean women (median age, 45 years) who underwent a hysterectomy and a propensity-matched group of women.
- The primary outcome was CVD, including myocardial infarction (MI), coronary artery revascularization, and stroke.
TAKEAWAY:
- During follow-up of just under 8 years, the hysterectomy group had an increased risk of CVD compared with the non-hysterectomy group (hazard ratio [HR] 1.25; 95% confidence interval [CI], 1.09-1.44; P = .002)
- The incidence of MI and coronary revascularization was comparable between groups, but the risk of stroke was significantly higher among those who had had a hysterectomy (HR, 1.31; 95% CI, 1.12-1.53; P < .001)
- This increase in risk was similar after excluding patients who also underwent adnexal surgery.
IN PRACTICE:
Early hysterectomy was linked to higher CVD risk, especially stroke, but since the CVD incidence wasn’t high, a change in clinical practice may not be needed, said the authors.
STUDY DETAILS:
The study was conducted by Jin-Sung Yuk, MD, PhD, Department of Obstetrics and Gynecology, Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Republic of Korea, and colleagues. It was published online June 12 in JAMA Network Open.
LIMITATIONS:
The study was retrospective and observational and used administrative databases that may be prone to inaccurate coding. The findings may not be generalizable outside Korea.
DISCLOSURES:
The study was supported by a National Research Foundation of Korea grant funded by the Korea government. The authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Immediate statin after acute stroke reduces disability
MUNICH, GERMANY – without compromising safety, results of the INSPIRES trial show.
The research, presented at the annual European Stroke Organisation Conference, also showed that intensive antiplatelet therapy reduced the risk for recurrent stroke albeit at an increased in bleeding risk versus standard treatment.
The study involved more than 6,000 patients with acute mild ischemic stroke or TIA and intracranial or extracranial atherosclerosis (ICAS/ECAS), who were randomly assigned in a 2 x 2 factorial design to compare intensive versus standard antiplatelet therapy and intensive statin therapy within 24 hours versus waiting up to 72 hours after onset.
Intensive antiplatelet therapy with clopidogrel plus aspirin reduced the risk for recurrent stroke within 90 days by 21% versus standard single-agent therapy, although it also doubled the risk for moderate to severe bleeding.
Starting intensive statin therapy with atorvastatin within 24 hours of onset had no impact on recurrent stroke risk but did reduce the risk for a poor functional outcome versus waiting up to 72 hours by 16%.
Moreover, it was “safe, with no increased risk of bleeding, hepatotoxicity, or muscle toxicity,” said study presenter Yilong Wang, MD, department of neurology, Beijing Tiantan Hospital, National Clinical Research Center.
There was, however, a suggestion of an interaction between intensive antiplatelet therapy and immediate intensive statin therapy, he noted, with a trend toward increased bleeding vs delaying the start of statin therapy.
Approached for comment, session cochair Carlos Molina, MD, director of the stroke unit and brain hemodynamics in Hospital Universitari Vall d’Hebron, Barcelona, said that the study is “important because when we look at studies of minor stroke and TIA, they are just focused on long-term outcomes in terms of recurrent stroke.”
He said in an interview that “putting statins in the equation and looking at their impact on long-term outcomes, the study demonstrates that statins are associated ... in particular with reductions in disabling stoke, and that’s good.”
Recurrence and progression
Dr. Wang began by highlighting that acute mild stroke and high-risk TIA are common and underestimated, with a relatively high risk for recurrence and progression, often caused by ICAS/ECAS.
Numerous guidelines recommend intensive antiplatelet therapy in the first 24 hours after the event, but Wang pointed out that there is little evidence to support this, and a meta-analysis suggested the window for effective treatment may be up to 72 hours.
In addition, intense statin therapy appears to be beneficial for the secondary prevention of atherosclerotic stroke in the nonacute phase, although there is no evidence for any neuroprotective effects in the acute phase nor for the optimal timing of starting the drugs.
Dr. Wang also noted that there is the potential for an interaction between intensive antiplatelet and statin therapy that could increase the risk for bleeding.
To investigate further, the researchers conducted a multicenter study involving patients aged 35-80 years with acute ischemic stroke or TIA.
The former was defined as an acute single infarction with 50% or greater stenosis of a major intracranial or extracranial artery that “probably account for the infarction and symptoms,” or multiple infarctions of large artery origin, including nonstenotic vulnerable plaques.
Patients were required to have a National Institutes of Health Stroke Scale score of 4-5 24 hours or less from acute stoke onset or 0-5 between 24 and 72 hours of onset.
TIA was defined as 50% or more stenosis of major intracranial or extracranial arteries that probably account for the symptoms, and an ABCD2 score for stroke risk of 4 or more within 24-72 hours of onset.
Patients were excluded if they had received dual antiplatelet therapy with aspirin and clopidogrel or high-intensity statin therapy within 14 days of random assignment or had intravenous thrombolysis or endovascular therapy after acute stroke or TIA onset.
Those included in the trial were randomly assigned in a 2 x 2 factorial design to receive:
- Intensive or dual antiplatelet therapy with clopidogrel and aspirin plus immediate high-intensity statin therapy with atorvastatin
- Intensive antiplatelet therapy plus delayed high-intensity statin therapy
- Standard antiplatelet therapy with aspirin alone plus immediate high-intensity statin therapy
- Standard antiplatelet therapy plus delayed high-intensity statin therapy
In all, 6,100 patients were enrolled from 222 hospitals in 99 cities across 25 provinces in China. The mean age was 65 years, and 34.6%-37.0% were women. TIA was recorded in 12.2%-14.1% of patients; 19.5%-19.7% had a single acute infarction, and 66.4%-68.1% had acute multiple infarctions.
The time to randomization was 24 hours or less after event onset in 12.5%-13.2% of cases versus 24-48 hours in 41.2%-42.5% and 48 hours or more in 44.9%-45.7% of patients.
The primary efficacy outcome, defined as stroke at 90 days, was significantly less common with intensive versus standard antiplatelet therapy, at a cumulative probability of 9.2% versus 7.3% (hazard ratio, 0.79; 95% confidence interval, 0.66-0.94; P = .007).
Clopidogrel plus aspirin was also associated with a significant reduction in a composite vascular event of stroke, myocardial infarction, or vascular death versus aspirin alone, at 7.5% versus 9.3% (HR, 0.80; 95% CI, 0.67-0.95, P = .01), as well as a reduction in rates of ischemic stroke (P = .002), and TIA (P = .02).
The primary safety outcome, defined as moderate to severe bleeding on the GUSTO criteria, was increased with intensive antiplatelet therapy, at 0.9% versus 0.4% for aspirin alone (HR, 2.08; 95% CI, 1.07-4.03; P = .02).
Turning to statin use, Dr. Wang showed that there was no significant difference in rates of stroke at 90 days between delayed and immediate intensive therapy, at a cumulative probability of 8.4% versus 8.1% (HR, 0.95; P = .58).
There was also no difference in rates of moderate to severe bleeding, at 0.8% with immediate versus 0.6% for delayed intensive statin therapy (HR, 1.36; 95% CI, 0.73-2.54; P = .34).
Dr. Wang reported that there were no significant differences in key secondary efficacy and safety outcomes.
Analysis of the distribution of modified Rankin Scale scores at 90 days, however, indicated that there was a significant reduction in the risk for poor functional outcome, defined as a score of 2-6, with immediate versus delayed statin therapy (odds ratio, 0.84; 95% CI, 0.72-0.99; P = .04).
Finally, it was found that combining dual antiplatelet therapy with immediate intensive statin therapy was associated with an increase in moderate to severe bleeding versus delayed statin therapy, affecting 1.1% versus 0.7% of patients. The association nonetheless did not reach statistical significance (HR, 1.70; 95% CI, 0.78-3.71; P = .18).
The study was funded by the National Natural Science Foundation of China, the National Key R&D Program of China, the Beijing Outstanding Young Scientist Program, the Beijing Youth Scholar Program, and the Beijing Talent Project. The drug was provided by Sanofi and Jialin Pharmaceutical. No relevant financial relationships were declared.
A version of this article originally appeared on Medscape.com.
MUNICH, GERMANY – without compromising safety, results of the INSPIRES trial show.
The research, presented at the annual European Stroke Organisation Conference, also showed that intensive antiplatelet therapy reduced the risk for recurrent stroke albeit at an increased in bleeding risk versus standard treatment.
The study involved more than 6,000 patients with acute mild ischemic stroke or TIA and intracranial or extracranial atherosclerosis (ICAS/ECAS), who were randomly assigned in a 2 x 2 factorial design to compare intensive versus standard antiplatelet therapy and intensive statin therapy within 24 hours versus waiting up to 72 hours after onset.
Intensive antiplatelet therapy with clopidogrel plus aspirin reduced the risk for recurrent stroke within 90 days by 21% versus standard single-agent therapy, although it also doubled the risk for moderate to severe bleeding.
Starting intensive statin therapy with atorvastatin within 24 hours of onset had no impact on recurrent stroke risk but did reduce the risk for a poor functional outcome versus waiting up to 72 hours by 16%.
Moreover, it was “safe, with no increased risk of bleeding, hepatotoxicity, or muscle toxicity,” said study presenter Yilong Wang, MD, department of neurology, Beijing Tiantan Hospital, National Clinical Research Center.
There was, however, a suggestion of an interaction between intensive antiplatelet therapy and immediate intensive statin therapy, he noted, with a trend toward increased bleeding vs delaying the start of statin therapy.
Approached for comment, session cochair Carlos Molina, MD, director of the stroke unit and brain hemodynamics in Hospital Universitari Vall d’Hebron, Barcelona, said that the study is “important because when we look at studies of minor stroke and TIA, they are just focused on long-term outcomes in terms of recurrent stroke.”
He said in an interview that “putting statins in the equation and looking at their impact on long-term outcomes, the study demonstrates that statins are associated ... in particular with reductions in disabling stoke, and that’s good.”
Recurrence and progression
Dr. Wang began by highlighting that acute mild stroke and high-risk TIA are common and underestimated, with a relatively high risk for recurrence and progression, often caused by ICAS/ECAS.
Numerous guidelines recommend intensive antiplatelet therapy in the first 24 hours after the event, but Wang pointed out that there is little evidence to support this, and a meta-analysis suggested the window for effective treatment may be up to 72 hours.
In addition, intense statin therapy appears to be beneficial for the secondary prevention of atherosclerotic stroke in the nonacute phase, although there is no evidence for any neuroprotective effects in the acute phase nor for the optimal timing of starting the drugs.
Dr. Wang also noted that there is the potential for an interaction between intensive antiplatelet and statin therapy that could increase the risk for bleeding.
To investigate further, the researchers conducted a multicenter study involving patients aged 35-80 years with acute ischemic stroke or TIA.
The former was defined as an acute single infarction with 50% or greater stenosis of a major intracranial or extracranial artery that “probably account for the infarction and symptoms,” or multiple infarctions of large artery origin, including nonstenotic vulnerable plaques.
Patients were required to have a National Institutes of Health Stroke Scale score of 4-5 24 hours or less from acute stoke onset or 0-5 between 24 and 72 hours of onset.
TIA was defined as 50% or more stenosis of major intracranial or extracranial arteries that probably account for the symptoms, and an ABCD2 score for stroke risk of 4 or more within 24-72 hours of onset.
Patients were excluded if they had received dual antiplatelet therapy with aspirin and clopidogrel or high-intensity statin therapy within 14 days of random assignment or had intravenous thrombolysis or endovascular therapy after acute stroke or TIA onset.
Those included in the trial were randomly assigned in a 2 x 2 factorial design to receive:
- Intensive or dual antiplatelet therapy with clopidogrel and aspirin plus immediate high-intensity statin therapy with atorvastatin
- Intensive antiplatelet therapy plus delayed high-intensity statin therapy
- Standard antiplatelet therapy with aspirin alone plus immediate high-intensity statin therapy
- Standard antiplatelet therapy plus delayed high-intensity statin therapy
In all, 6,100 patients were enrolled from 222 hospitals in 99 cities across 25 provinces in China. The mean age was 65 years, and 34.6%-37.0% were women. TIA was recorded in 12.2%-14.1% of patients; 19.5%-19.7% had a single acute infarction, and 66.4%-68.1% had acute multiple infarctions.
The time to randomization was 24 hours or less after event onset in 12.5%-13.2% of cases versus 24-48 hours in 41.2%-42.5% and 48 hours or more in 44.9%-45.7% of patients.
The primary efficacy outcome, defined as stroke at 90 days, was significantly less common with intensive versus standard antiplatelet therapy, at a cumulative probability of 9.2% versus 7.3% (hazard ratio, 0.79; 95% confidence interval, 0.66-0.94; P = .007).
Clopidogrel plus aspirin was also associated with a significant reduction in a composite vascular event of stroke, myocardial infarction, or vascular death versus aspirin alone, at 7.5% versus 9.3% (HR, 0.80; 95% CI, 0.67-0.95, P = .01), as well as a reduction in rates of ischemic stroke (P = .002), and TIA (P = .02).
The primary safety outcome, defined as moderate to severe bleeding on the GUSTO criteria, was increased with intensive antiplatelet therapy, at 0.9% versus 0.4% for aspirin alone (HR, 2.08; 95% CI, 1.07-4.03; P = .02).
Turning to statin use, Dr. Wang showed that there was no significant difference in rates of stroke at 90 days between delayed and immediate intensive therapy, at a cumulative probability of 8.4% versus 8.1% (HR, 0.95; P = .58).
There was also no difference in rates of moderate to severe bleeding, at 0.8% with immediate versus 0.6% for delayed intensive statin therapy (HR, 1.36; 95% CI, 0.73-2.54; P = .34).
Dr. Wang reported that there were no significant differences in key secondary efficacy and safety outcomes.
Analysis of the distribution of modified Rankin Scale scores at 90 days, however, indicated that there was a significant reduction in the risk for poor functional outcome, defined as a score of 2-6, with immediate versus delayed statin therapy (odds ratio, 0.84; 95% CI, 0.72-0.99; P = .04).
Finally, it was found that combining dual antiplatelet therapy with immediate intensive statin therapy was associated with an increase in moderate to severe bleeding versus delayed statin therapy, affecting 1.1% versus 0.7% of patients. The association nonetheless did not reach statistical significance (HR, 1.70; 95% CI, 0.78-3.71; P = .18).
The study was funded by the National Natural Science Foundation of China, the National Key R&D Program of China, the Beijing Outstanding Young Scientist Program, the Beijing Youth Scholar Program, and the Beijing Talent Project. The drug was provided by Sanofi and Jialin Pharmaceutical. No relevant financial relationships were declared.
A version of this article originally appeared on Medscape.com.
MUNICH, GERMANY – without compromising safety, results of the INSPIRES trial show.
The research, presented at the annual European Stroke Organisation Conference, also showed that intensive antiplatelet therapy reduced the risk for recurrent stroke albeit at an increased in bleeding risk versus standard treatment.
The study involved more than 6,000 patients with acute mild ischemic stroke or TIA and intracranial or extracranial atherosclerosis (ICAS/ECAS), who were randomly assigned in a 2 x 2 factorial design to compare intensive versus standard antiplatelet therapy and intensive statin therapy within 24 hours versus waiting up to 72 hours after onset.
Intensive antiplatelet therapy with clopidogrel plus aspirin reduced the risk for recurrent stroke within 90 days by 21% versus standard single-agent therapy, although it also doubled the risk for moderate to severe bleeding.
Starting intensive statin therapy with atorvastatin within 24 hours of onset had no impact on recurrent stroke risk but did reduce the risk for a poor functional outcome versus waiting up to 72 hours by 16%.
Moreover, it was “safe, with no increased risk of bleeding, hepatotoxicity, or muscle toxicity,” said study presenter Yilong Wang, MD, department of neurology, Beijing Tiantan Hospital, National Clinical Research Center.
There was, however, a suggestion of an interaction between intensive antiplatelet therapy and immediate intensive statin therapy, he noted, with a trend toward increased bleeding vs delaying the start of statin therapy.
Approached for comment, session cochair Carlos Molina, MD, director of the stroke unit and brain hemodynamics in Hospital Universitari Vall d’Hebron, Barcelona, said that the study is “important because when we look at studies of minor stroke and TIA, they are just focused on long-term outcomes in terms of recurrent stroke.”
He said in an interview that “putting statins in the equation and looking at their impact on long-term outcomes, the study demonstrates that statins are associated ... in particular with reductions in disabling stoke, and that’s good.”
Recurrence and progression
Dr. Wang began by highlighting that acute mild stroke and high-risk TIA are common and underestimated, with a relatively high risk for recurrence and progression, often caused by ICAS/ECAS.
Numerous guidelines recommend intensive antiplatelet therapy in the first 24 hours after the event, but Wang pointed out that there is little evidence to support this, and a meta-analysis suggested the window for effective treatment may be up to 72 hours.
In addition, intense statin therapy appears to be beneficial for the secondary prevention of atherosclerotic stroke in the nonacute phase, although there is no evidence for any neuroprotective effects in the acute phase nor for the optimal timing of starting the drugs.
Dr. Wang also noted that there is the potential for an interaction between intensive antiplatelet and statin therapy that could increase the risk for bleeding.
To investigate further, the researchers conducted a multicenter study involving patients aged 35-80 years with acute ischemic stroke or TIA.
The former was defined as an acute single infarction with 50% or greater stenosis of a major intracranial or extracranial artery that “probably account for the infarction and symptoms,” or multiple infarctions of large artery origin, including nonstenotic vulnerable plaques.
Patients were required to have a National Institutes of Health Stroke Scale score of 4-5 24 hours or less from acute stoke onset or 0-5 between 24 and 72 hours of onset.
TIA was defined as 50% or more stenosis of major intracranial or extracranial arteries that probably account for the symptoms, and an ABCD2 score for stroke risk of 4 or more within 24-72 hours of onset.
Patients were excluded if they had received dual antiplatelet therapy with aspirin and clopidogrel or high-intensity statin therapy within 14 days of random assignment or had intravenous thrombolysis or endovascular therapy after acute stroke or TIA onset.
Those included in the trial were randomly assigned in a 2 x 2 factorial design to receive:
- Intensive or dual antiplatelet therapy with clopidogrel and aspirin plus immediate high-intensity statin therapy with atorvastatin
- Intensive antiplatelet therapy plus delayed high-intensity statin therapy
- Standard antiplatelet therapy with aspirin alone plus immediate high-intensity statin therapy
- Standard antiplatelet therapy plus delayed high-intensity statin therapy
In all, 6,100 patients were enrolled from 222 hospitals in 99 cities across 25 provinces in China. The mean age was 65 years, and 34.6%-37.0% were women. TIA was recorded in 12.2%-14.1% of patients; 19.5%-19.7% had a single acute infarction, and 66.4%-68.1% had acute multiple infarctions.
The time to randomization was 24 hours or less after event onset in 12.5%-13.2% of cases versus 24-48 hours in 41.2%-42.5% and 48 hours or more in 44.9%-45.7% of patients.
The primary efficacy outcome, defined as stroke at 90 days, was significantly less common with intensive versus standard antiplatelet therapy, at a cumulative probability of 9.2% versus 7.3% (hazard ratio, 0.79; 95% confidence interval, 0.66-0.94; P = .007).
Clopidogrel plus aspirin was also associated with a significant reduction in a composite vascular event of stroke, myocardial infarction, or vascular death versus aspirin alone, at 7.5% versus 9.3% (HR, 0.80; 95% CI, 0.67-0.95, P = .01), as well as a reduction in rates of ischemic stroke (P = .002), and TIA (P = .02).
The primary safety outcome, defined as moderate to severe bleeding on the GUSTO criteria, was increased with intensive antiplatelet therapy, at 0.9% versus 0.4% for aspirin alone (HR, 2.08; 95% CI, 1.07-4.03; P = .02).
Turning to statin use, Dr. Wang showed that there was no significant difference in rates of stroke at 90 days between delayed and immediate intensive therapy, at a cumulative probability of 8.4% versus 8.1% (HR, 0.95; P = .58).
There was also no difference in rates of moderate to severe bleeding, at 0.8% with immediate versus 0.6% for delayed intensive statin therapy (HR, 1.36; 95% CI, 0.73-2.54; P = .34).
Dr. Wang reported that there were no significant differences in key secondary efficacy and safety outcomes.
Analysis of the distribution of modified Rankin Scale scores at 90 days, however, indicated that there was a significant reduction in the risk for poor functional outcome, defined as a score of 2-6, with immediate versus delayed statin therapy (odds ratio, 0.84; 95% CI, 0.72-0.99; P = .04).
Finally, it was found that combining dual antiplatelet therapy with immediate intensive statin therapy was associated with an increase in moderate to severe bleeding versus delayed statin therapy, affecting 1.1% versus 0.7% of patients. The association nonetheless did not reach statistical significance (HR, 1.70; 95% CI, 0.78-3.71; P = .18).
The study was funded by the National Natural Science Foundation of China, the National Key R&D Program of China, the Beijing Outstanding Young Scientist Program, the Beijing Youth Scholar Program, and the Beijing Talent Project. The drug was provided by Sanofi and Jialin Pharmaceutical. No relevant financial relationships were declared.
A version of this article originally appeared on Medscape.com.
AT ESOC 2023
Antibody linked to spontaneous reversal of ATTR-CM
Cardiac transthyretin amyloidosis (also called ATTR amyloidosis cardiomyopathy or ATTR-CM) is a progressive disease and a cause of heart failure resulting from accumulation of the protein transthyretin, which misfolds and forms amyloid deposits on the walls of the heart, causing both systolic and diastolic dysfunction.
The condition is progressive and normally fatal within a few years of diagnosis. Treatment options are limited and aimed at slowing progression; nothing has been shown to reverse the course of the disease.
However, an international team of researchers is now reporting the discovery of three patients with ATTR-CM–associated heart failure in whom the condition resolved spontaneously, with reversion to near normal cardiac structure and function. On further investigation, it was found that these three patients had developed circulating polyclonal IgG antibodies to human ATTR amyloid.
They are hopeful that a monoclonal form of these antibodies could be developed and may represent a novel treatment, or even a cure, for the condition.
The researchers report their findings in a letter to the New England Journal of Medicine.
“We are very optimistic about this discovery of these antibodies. They could become the first treatment to clear the amyloid that causes this horribly progressive and fatal condition,” senior author Julian Gillmore, MD, head of the University College London Centre for Amyloidosis, based at the Royal Free Hospital, said in an interview.
“Obviously, there is a lot of work to do before we can say this is the case, but it is very exciting,” he added.
Dr. Gillmore explained how the antibodies were discovered. “This disease has a universally progressive course, but we had one patient who on a repeat appointment said he felt better and on detailed cardiac MRI imaging, we found that the amyloid in his heart had reduced. That is totally unheard of,” he said.
“We then looked back at our cohort of 1,663 patients with ATTR-cardiomyopathy, and we discovered two others who had also improved both on imaging and clinically,” Dr. Gillmore said.
Each of these three patients reported a reduction in symptoms, although they had not received any new or potentially disease-modifying treatments. None of the patients had had recent vaccinations, notable infections, or any clinical suggestion of myocarditis.
Clinical recovery was corroborated by substantial improvement or normalization of findings on echocardiography, serum biomarker levels, and results of cardiopulmonary exercise tests and scintigraphy.
Serial cardiac MRI scans confirmed near-complete regression of myocardial extracellular volume, coupled with remodeling to near-normal cardiac structure and function without scarring.
The researchers wondered whether the changes in these patients may have been brought about by an antibody response. On further investigation, they found antibodies in the three patients that bound specifically to ATTR amyloid deposits in a transgenic mouse model of the condition, and to synthetic ATTR amyloid. No such antibodies were present in the other 350 patients in the cohort with a typical clinical course.
“The cause and clinical significance of the anti-ATTR amyloid antibodies are intriguing and presently unclear. However, the clinical recovery of these patients establishes the unanticipated potential for reversibility of ATTR-CM and raises expectations for its treatment,” the researchers conclude.
Dr. Gillmore said they didn’t know why these three patients had these antibodies, while all the other patients did not. “There must be something different about these patients. We don’t know what that is at present, but we are looking hard.”
The researchers are hoping that after this publication, other centers caring for patients with ATTR-cardiomyopathy will look in their cohorts and see if they can identify other cases where there has been improvement.
“It is very plausible that they do have such cases, but they will be rare, as we all think of this disease as universally progressive and fatal,” Dr. Gillmore noted.
“We haven’t absolutely proven that the antibodies have caused the clearance of amyloid in these patients, but we strongly suspect this to be the case,” Dr. Gillmore said. The researchers are planning to try to confirm this by isolating the antibodies and treating the transgenic mice.
Dr. Gillmore attributed the current discovery to the development of novel imaging cardiac MRI techniques. “This allowed us to monitor closely the amyloid burden in the heart. The observation that this had diminished in these three patients was the breakthrough that led us to look for antibodies.”
Another antibody product directed against ATTR cardiomyopathy is also in development by Neurimmune, a Swiss biopharmaceutical company. A phase 1 study of this agent was recently published, suggesting that it appeared to reduce the amount of amyloid protein deposited in the heart.
Dr. Gillmore said the antibody they have detected is different from the Neurimmune product.
The research was supported by a British Heart Foundation Intermediate Clinical Research Fellowship, a Medical Research Council Career Development Award, and a project grant from the British Heart Foundation. Dr. Gillmore reports being a consultant or expert advisory board member for Alnylam Pharmaceuticals, AstraZeneca, ATTRalus, Eidos Therapeutics, Intellia Therapeutics, Ionis Pharmaceuticals, and Pfizer.
A version of this article originally appeared on Medscape.com.
Cardiac transthyretin amyloidosis (also called ATTR amyloidosis cardiomyopathy or ATTR-CM) is a progressive disease and a cause of heart failure resulting from accumulation of the protein transthyretin, which misfolds and forms amyloid deposits on the walls of the heart, causing both systolic and diastolic dysfunction.
The condition is progressive and normally fatal within a few years of diagnosis. Treatment options are limited and aimed at slowing progression; nothing has been shown to reverse the course of the disease.
However, an international team of researchers is now reporting the discovery of three patients with ATTR-CM–associated heart failure in whom the condition resolved spontaneously, with reversion to near normal cardiac structure and function. On further investigation, it was found that these three patients had developed circulating polyclonal IgG antibodies to human ATTR amyloid.
They are hopeful that a monoclonal form of these antibodies could be developed and may represent a novel treatment, or even a cure, for the condition.
The researchers report their findings in a letter to the New England Journal of Medicine.
“We are very optimistic about this discovery of these antibodies. They could become the first treatment to clear the amyloid that causes this horribly progressive and fatal condition,” senior author Julian Gillmore, MD, head of the University College London Centre for Amyloidosis, based at the Royal Free Hospital, said in an interview.
“Obviously, there is a lot of work to do before we can say this is the case, but it is very exciting,” he added.
Dr. Gillmore explained how the antibodies were discovered. “This disease has a universally progressive course, but we had one patient who on a repeat appointment said he felt better and on detailed cardiac MRI imaging, we found that the amyloid in his heart had reduced. That is totally unheard of,” he said.
“We then looked back at our cohort of 1,663 patients with ATTR-cardiomyopathy, and we discovered two others who had also improved both on imaging and clinically,” Dr. Gillmore said.
Each of these three patients reported a reduction in symptoms, although they had not received any new or potentially disease-modifying treatments. None of the patients had had recent vaccinations, notable infections, or any clinical suggestion of myocarditis.
Clinical recovery was corroborated by substantial improvement or normalization of findings on echocardiography, serum biomarker levels, and results of cardiopulmonary exercise tests and scintigraphy.
Serial cardiac MRI scans confirmed near-complete regression of myocardial extracellular volume, coupled with remodeling to near-normal cardiac structure and function without scarring.
The researchers wondered whether the changes in these patients may have been brought about by an antibody response. On further investigation, they found antibodies in the three patients that bound specifically to ATTR amyloid deposits in a transgenic mouse model of the condition, and to synthetic ATTR amyloid. No such antibodies were present in the other 350 patients in the cohort with a typical clinical course.
“The cause and clinical significance of the anti-ATTR amyloid antibodies are intriguing and presently unclear. However, the clinical recovery of these patients establishes the unanticipated potential for reversibility of ATTR-CM and raises expectations for its treatment,” the researchers conclude.
Dr. Gillmore said they didn’t know why these three patients had these antibodies, while all the other patients did not. “There must be something different about these patients. We don’t know what that is at present, but we are looking hard.”
The researchers are hoping that after this publication, other centers caring for patients with ATTR-cardiomyopathy will look in their cohorts and see if they can identify other cases where there has been improvement.
“It is very plausible that they do have such cases, but they will be rare, as we all think of this disease as universally progressive and fatal,” Dr. Gillmore noted.
“We haven’t absolutely proven that the antibodies have caused the clearance of amyloid in these patients, but we strongly suspect this to be the case,” Dr. Gillmore said. The researchers are planning to try to confirm this by isolating the antibodies and treating the transgenic mice.
Dr. Gillmore attributed the current discovery to the development of novel imaging cardiac MRI techniques. “This allowed us to monitor closely the amyloid burden in the heart. The observation that this had diminished in these three patients was the breakthrough that led us to look for antibodies.”
Another antibody product directed against ATTR cardiomyopathy is also in development by Neurimmune, a Swiss biopharmaceutical company. A phase 1 study of this agent was recently published, suggesting that it appeared to reduce the amount of amyloid protein deposited in the heart.
Dr. Gillmore said the antibody they have detected is different from the Neurimmune product.
The research was supported by a British Heart Foundation Intermediate Clinical Research Fellowship, a Medical Research Council Career Development Award, and a project grant from the British Heart Foundation. Dr. Gillmore reports being a consultant or expert advisory board member for Alnylam Pharmaceuticals, AstraZeneca, ATTRalus, Eidos Therapeutics, Intellia Therapeutics, Ionis Pharmaceuticals, and Pfizer.
A version of this article originally appeared on Medscape.com.
Cardiac transthyretin amyloidosis (also called ATTR amyloidosis cardiomyopathy or ATTR-CM) is a progressive disease and a cause of heart failure resulting from accumulation of the protein transthyretin, which misfolds and forms amyloid deposits on the walls of the heart, causing both systolic and diastolic dysfunction.
The condition is progressive and normally fatal within a few years of diagnosis. Treatment options are limited and aimed at slowing progression; nothing has been shown to reverse the course of the disease.
However, an international team of researchers is now reporting the discovery of three patients with ATTR-CM–associated heart failure in whom the condition resolved spontaneously, with reversion to near normal cardiac structure and function. On further investigation, it was found that these three patients had developed circulating polyclonal IgG antibodies to human ATTR amyloid.
They are hopeful that a monoclonal form of these antibodies could be developed and may represent a novel treatment, or even a cure, for the condition.
The researchers report their findings in a letter to the New England Journal of Medicine.
“We are very optimistic about this discovery of these antibodies. They could become the first treatment to clear the amyloid that causes this horribly progressive and fatal condition,” senior author Julian Gillmore, MD, head of the University College London Centre for Amyloidosis, based at the Royal Free Hospital, said in an interview.
“Obviously, there is a lot of work to do before we can say this is the case, but it is very exciting,” he added.
Dr. Gillmore explained how the antibodies were discovered. “This disease has a universally progressive course, but we had one patient who on a repeat appointment said he felt better and on detailed cardiac MRI imaging, we found that the amyloid in his heart had reduced. That is totally unheard of,” he said.
“We then looked back at our cohort of 1,663 patients with ATTR-cardiomyopathy, and we discovered two others who had also improved both on imaging and clinically,” Dr. Gillmore said.
Each of these three patients reported a reduction in symptoms, although they had not received any new or potentially disease-modifying treatments. None of the patients had had recent vaccinations, notable infections, or any clinical suggestion of myocarditis.
Clinical recovery was corroborated by substantial improvement or normalization of findings on echocardiography, serum biomarker levels, and results of cardiopulmonary exercise tests and scintigraphy.
Serial cardiac MRI scans confirmed near-complete regression of myocardial extracellular volume, coupled with remodeling to near-normal cardiac structure and function without scarring.
The researchers wondered whether the changes in these patients may have been brought about by an antibody response. On further investigation, they found antibodies in the three patients that bound specifically to ATTR amyloid deposits in a transgenic mouse model of the condition, and to synthetic ATTR amyloid. No such antibodies were present in the other 350 patients in the cohort with a typical clinical course.
“The cause and clinical significance of the anti-ATTR amyloid antibodies are intriguing and presently unclear. However, the clinical recovery of these patients establishes the unanticipated potential for reversibility of ATTR-CM and raises expectations for its treatment,” the researchers conclude.
Dr. Gillmore said they didn’t know why these three patients had these antibodies, while all the other patients did not. “There must be something different about these patients. We don’t know what that is at present, but we are looking hard.”
The researchers are hoping that after this publication, other centers caring for patients with ATTR-cardiomyopathy will look in their cohorts and see if they can identify other cases where there has been improvement.
“It is very plausible that they do have such cases, but they will be rare, as we all think of this disease as universally progressive and fatal,” Dr. Gillmore noted.
“We haven’t absolutely proven that the antibodies have caused the clearance of amyloid in these patients, but we strongly suspect this to be the case,” Dr. Gillmore said. The researchers are planning to try to confirm this by isolating the antibodies and treating the transgenic mice.
Dr. Gillmore attributed the current discovery to the development of novel imaging cardiac MRI techniques. “This allowed us to monitor closely the amyloid burden in the heart. The observation that this had diminished in these three patients was the breakthrough that led us to look for antibodies.”
Another antibody product directed against ATTR cardiomyopathy is also in development by Neurimmune, a Swiss biopharmaceutical company. A phase 1 study of this agent was recently published, suggesting that it appeared to reduce the amount of amyloid protein deposited in the heart.
Dr. Gillmore said the antibody they have detected is different from the Neurimmune product.
The research was supported by a British Heart Foundation Intermediate Clinical Research Fellowship, a Medical Research Council Career Development Award, and a project grant from the British Heart Foundation. Dr. Gillmore reports being a consultant or expert advisory board member for Alnylam Pharmaceuticals, AstraZeneca, ATTRalus, Eidos Therapeutics, Intellia Therapeutics, Ionis Pharmaceuticals, and Pfizer.
A version of this article originally appeared on Medscape.com.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Big boost in sodium excretion with HF diuretic protocol
In patients with acute heart failure, a urine sodium-guided diuretic protocol, currently recommended in guidelines from the Heart Failure Association of the European Society of Cardiology (HFA-ESC), led to significant increases in natriuresis and diuresis over 2 days in the prospective ENACT-HF clinical trial.
The guideline protocol was based on a 2019 HFA position paper with expert consensus, but it had not been tested prospectively, Jeroen Dauw, MD, of AZ Sint-Lucas Ghent (Belgium), explained in a presentation at HFA-ESC 2023.
“We had 282 millimoles of sodium excretion after one day, which is an increase of 64%, compared with standard of care,” Dr. Dauw told meeting attendees. “We wanted to power for 15%, so we’re way above it, with a P value of lower than 0.001.”
The effect was consistent across predefined subgroups, he said. “In addition, there’s an even higher benefit in patients with a lower eGFR [estimated glomerular filtration rate] and a higher home dose of loop diuretics, which might signal more diuretic resistance and more benefit of the protocol.”
After 2 days, the investigators saw 52% higher natriuresis and 33% higher diuresis, compared with usual care.
In an interview, Dr. Dauw said, “The protocol is feasible, safe, and very effective. Cardiologists might consider how to implement a similar protocol in their center to improve the care of their acute heart failure patients.”
Twice the oral home dose
The investigators conducted a multicenter, open-label, nonrandomized pragmatic trial at 29 centers in 18 countries globally. “We aimed to recruit 500 to detect a 15% difference in natriuresis,” Dr. Dauw said in his presentation, “but because we were a really low-budget trial, we had to stop after 3 years of recruitment.”
Therefore, 401 patients participated, 254 in the SOC arm and 147 in the protocol arm, because of the sequential nature of the study; that is, patients in the SOC arm of the two-phase study were recruited first.
Patients’ mean age was 70 years, 38% were women, and they all had at least one sign of volume overload. They were on a maintenance daily diuretic dose of 40 mg of furosemide for a month or more, and the NT-proBNP was above 1,000.
In phase 1 of the study, all centers treated 10 consecutive patients according to the local standard of care, at the discretion of the physician. In phase 2, the centers again recruited and treated at least 10 consecutive patients, this time according to the standardized diuretic protocol.
In the protocol phase, patients were treated with twice the oral home dose as an IV bolus. “This meant if, for example, you have 40 mg of furosemide at home, then you receive 80 mg as a first bolus,” Dr. Dauw told attendees. A spot urine sample was taken after 2 hours, and the response was evaluated after 6 hours. A urine sodium above 50 millimoles per liter was considered a good response.
On the second day, patients were reevaluated in the morning using urine output as a measure of diuretic response. If it was above 3 L, then the same bolus was repeated again twice daily, with 6-12 hours between administrations.
As noted, after one day, natriuresis was 174 millimoles in the SOC arm versus 282 millimoles in the protocol group – an increase of 64%. The effect was consistent across subgroups, and those with a lower eGFR and a higher home dose of loop diuretics benefited more.
Furthermore, Dr. Dauw said, there was no interaction on the endpoints with SGLT2 inhibitor use at baseline.
After two days, natriuresis was 52% higher in the protocol group and diuresis was 33% higher.
However, there was no significant difference in weight loss and no difference in the congestion score.
“We did expect to see a difference in weight loss between the study groups, as higher natriuresis and diuresis would normally be associated with higher weight loss in the protocol group,” Dr. Dauw told this news organization. “However, looking back at the study design, weight was collected from the electronic health records and not rigorously collected by study nurses. Previous studies have shown discrepancies between fluid loss and weight loss, so this is an ‘explainable’ finding.”
Participants also had a relatively high congestion score at baseline, with edema above the knee and also some pleural effusion, he told meeting attendees. Therefore, it might take more time to see a change in congestion score in those patients.
The protocol also led to a shorter length of stay – one day less in the hospital – and was very safe on renal endpoints, Dr. Dauw concluded.
A session chair asked why only patients already on diuretics were included in the study, noting that in his clinic, about half of the admissions are de novo.
Dr. Dauw said that patients already taking diuretics chronically would benefit most from the protocol. “If patients are diuretic-naive, they probably will respond well to whatever you do; if you just give a higher dose, they will respond well,” he said. “We expected that the largest benefit would be in patients already taking diuretics because they have a higher chance of not responding well.”
“There also was a big difference in the starting dose,” he added. “In the SOC arm, the baseline dose was about 60 mg, whereas we gave 120 mg, and we could already see a high difference in the effect. So, in those patients, I think the gain is bigger if you follow the protocol.”
More data coming
Looking ahead, “we only showed efficacy in the first 2 days of treatment and a shorter length of stay, probably reflecting a faster decongestion, but we don’t know for sure,” Dr. Dauw told this news organization.
“It would be important to have a study where the protocol is followed until full decongestion is reached,” he said. “That way, we can directly prove that decongestion is better and/or faster with the protocol.”
“A good decongestive strategy is one that is fast, safe and effective in decreasing signs and symptoms that patients suffer from,” he added. “We believe our protocol can achieve that, but our study is only one piece of the puzzle.”
More data on natriuresis-guided decongestion is coming this year, he said, with the PUSH-AHF study from Groningen, the European DECONGEST study, and the U.S. ESCALATE study.
The study had no funding. Dr. Dauw declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In patients with acute heart failure, a urine sodium-guided diuretic protocol, currently recommended in guidelines from the Heart Failure Association of the European Society of Cardiology (HFA-ESC), led to significant increases in natriuresis and diuresis over 2 days in the prospective ENACT-HF clinical trial.
The guideline protocol was based on a 2019 HFA position paper with expert consensus, but it had not been tested prospectively, Jeroen Dauw, MD, of AZ Sint-Lucas Ghent (Belgium), explained in a presentation at HFA-ESC 2023.
“We had 282 millimoles of sodium excretion after one day, which is an increase of 64%, compared with standard of care,” Dr. Dauw told meeting attendees. “We wanted to power for 15%, so we’re way above it, with a P value of lower than 0.001.”
The effect was consistent across predefined subgroups, he said. “In addition, there’s an even higher benefit in patients with a lower eGFR [estimated glomerular filtration rate] and a higher home dose of loop diuretics, which might signal more diuretic resistance and more benefit of the protocol.”
After 2 days, the investigators saw 52% higher natriuresis and 33% higher diuresis, compared with usual care.
In an interview, Dr. Dauw said, “The protocol is feasible, safe, and very effective. Cardiologists might consider how to implement a similar protocol in their center to improve the care of their acute heart failure patients.”
Twice the oral home dose
The investigators conducted a multicenter, open-label, nonrandomized pragmatic trial at 29 centers in 18 countries globally. “We aimed to recruit 500 to detect a 15% difference in natriuresis,” Dr. Dauw said in his presentation, “but because we were a really low-budget trial, we had to stop after 3 years of recruitment.”
Therefore, 401 patients participated, 254 in the SOC arm and 147 in the protocol arm, because of the sequential nature of the study; that is, patients in the SOC arm of the two-phase study were recruited first.
Patients’ mean age was 70 years, 38% were women, and they all had at least one sign of volume overload. They were on a maintenance daily diuretic dose of 40 mg of furosemide for a month or more, and the NT-proBNP was above 1,000.
In phase 1 of the study, all centers treated 10 consecutive patients according to the local standard of care, at the discretion of the physician. In phase 2, the centers again recruited and treated at least 10 consecutive patients, this time according to the standardized diuretic protocol.
In the protocol phase, patients were treated with twice the oral home dose as an IV bolus. “This meant if, for example, you have 40 mg of furosemide at home, then you receive 80 mg as a first bolus,” Dr. Dauw told attendees. A spot urine sample was taken after 2 hours, and the response was evaluated after 6 hours. A urine sodium above 50 millimoles per liter was considered a good response.
On the second day, patients were reevaluated in the morning using urine output as a measure of diuretic response. If it was above 3 L, then the same bolus was repeated again twice daily, with 6-12 hours between administrations.
As noted, after one day, natriuresis was 174 millimoles in the SOC arm versus 282 millimoles in the protocol group – an increase of 64%. The effect was consistent across subgroups, and those with a lower eGFR and a higher home dose of loop diuretics benefited more.
Furthermore, Dr. Dauw said, there was no interaction on the endpoints with SGLT2 inhibitor use at baseline.
After two days, natriuresis was 52% higher in the protocol group and diuresis was 33% higher.
However, there was no significant difference in weight loss and no difference in the congestion score.
“We did expect to see a difference in weight loss between the study groups, as higher natriuresis and diuresis would normally be associated with higher weight loss in the protocol group,” Dr. Dauw told this news organization. “However, looking back at the study design, weight was collected from the electronic health records and not rigorously collected by study nurses. Previous studies have shown discrepancies between fluid loss and weight loss, so this is an ‘explainable’ finding.”
Participants also had a relatively high congestion score at baseline, with edema above the knee and also some pleural effusion, he told meeting attendees. Therefore, it might take more time to see a change in congestion score in those patients.
The protocol also led to a shorter length of stay – one day less in the hospital – and was very safe on renal endpoints, Dr. Dauw concluded.
A session chair asked why only patients already on diuretics were included in the study, noting that in his clinic, about half of the admissions are de novo.
Dr. Dauw said that patients already taking diuretics chronically would benefit most from the protocol. “If patients are diuretic-naive, they probably will respond well to whatever you do; if you just give a higher dose, they will respond well,” he said. “We expected that the largest benefit would be in patients already taking diuretics because they have a higher chance of not responding well.”
“There also was a big difference in the starting dose,” he added. “In the SOC arm, the baseline dose was about 60 mg, whereas we gave 120 mg, and we could already see a high difference in the effect. So, in those patients, I think the gain is bigger if you follow the protocol.”
More data coming
Looking ahead, “we only showed efficacy in the first 2 days of treatment and a shorter length of stay, probably reflecting a faster decongestion, but we don’t know for sure,” Dr. Dauw told this news organization.
“It would be important to have a study where the protocol is followed until full decongestion is reached,” he said. “That way, we can directly prove that decongestion is better and/or faster with the protocol.”
“A good decongestive strategy is one that is fast, safe and effective in decreasing signs and symptoms that patients suffer from,” he added. “We believe our protocol can achieve that, but our study is only one piece of the puzzle.”
More data on natriuresis-guided decongestion is coming this year, he said, with the PUSH-AHF study from Groningen, the European DECONGEST study, and the U.S. ESCALATE study.
The study had no funding. Dr. Dauw declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In patients with acute heart failure, a urine sodium-guided diuretic protocol, currently recommended in guidelines from the Heart Failure Association of the European Society of Cardiology (HFA-ESC), led to significant increases in natriuresis and diuresis over 2 days in the prospective ENACT-HF clinical trial.
The guideline protocol was based on a 2019 HFA position paper with expert consensus, but it had not been tested prospectively, Jeroen Dauw, MD, of AZ Sint-Lucas Ghent (Belgium), explained in a presentation at HFA-ESC 2023.
“We had 282 millimoles of sodium excretion after one day, which is an increase of 64%, compared with standard of care,” Dr. Dauw told meeting attendees. “We wanted to power for 15%, so we’re way above it, with a P value of lower than 0.001.”
The effect was consistent across predefined subgroups, he said. “In addition, there’s an even higher benefit in patients with a lower eGFR [estimated glomerular filtration rate] and a higher home dose of loop diuretics, which might signal more diuretic resistance and more benefit of the protocol.”
After 2 days, the investigators saw 52% higher natriuresis and 33% higher diuresis, compared with usual care.
In an interview, Dr. Dauw said, “The protocol is feasible, safe, and very effective. Cardiologists might consider how to implement a similar protocol in their center to improve the care of their acute heart failure patients.”
Twice the oral home dose
The investigators conducted a multicenter, open-label, nonrandomized pragmatic trial at 29 centers in 18 countries globally. “We aimed to recruit 500 to detect a 15% difference in natriuresis,” Dr. Dauw said in his presentation, “but because we were a really low-budget trial, we had to stop after 3 years of recruitment.”
Therefore, 401 patients participated, 254 in the SOC arm and 147 in the protocol arm, because of the sequential nature of the study; that is, patients in the SOC arm of the two-phase study were recruited first.
Patients’ mean age was 70 years, 38% were women, and they all had at least one sign of volume overload. They were on a maintenance daily diuretic dose of 40 mg of furosemide for a month or more, and the NT-proBNP was above 1,000.
In phase 1 of the study, all centers treated 10 consecutive patients according to the local standard of care, at the discretion of the physician. In phase 2, the centers again recruited and treated at least 10 consecutive patients, this time according to the standardized diuretic protocol.
In the protocol phase, patients were treated with twice the oral home dose as an IV bolus. “This meant if, for example, you have 40 mg of furosemide at home, then you receive 80 mg as a first bolus,” Dr. Dauw told attendees. A spot urine sample was taken after 2 hours, and the response was evaluated after 6 hours. A urine sodium above 50 millimoles per liter was considered a good response.
On the second day, patients were reevaluated in the morning using urine output as a measure of diuretic response. If it was above 3 L, then the same bolus was repeated again twice daily, with 6-12 hours between administrations.
As noted, after one day, natriuresis was 174 millimoles in the SOC arm versus 282 millimoles in the protocol group – an increase of 64%. The effect was consistent across subgroups, and those with a lower eGFR and a higher home dose of loop diuretics benefited more.
Furthermore, Dr. Dauw said, there was no interaction on the endpoints with SGLT2 inhibitor use at baseline.
After two days, natriuresis was 52% higher in the protocol group and diuresis was 33% higher.
However, there was no significant difference in weight loss and no difference in the congestion score.
“We did expect to see a difference in weight loss between the study groups, as higher natriuresis and diuresis would normally be associated with higher weight loss in the protocol group,” Dr. Dauw told this news organization. “However, looking back at the study design, weight was collected from the electronic health records and not rigorously collected by study nurses. Previous studies have shown discrepancies between fluid loss and weight loss, so this is an ‘explainable’ finding.”
Participants also had a relatively high congestion score at baseline, with edema above the knee and also some pleural effusion, he told meeting attendees. Therefore, it might take more time to see a change in congestion score in those patients.
The protocol also led to a shorter length of stay – one day less in the hospital – and was very safe on renal endpoints, Dr. Dauw concluded.
A session chair asked why only patients already on diuretics were included in the study, noting that in his clinic, about half of the admissions are de novo.
Dr. Dauw said that patients already taking diuretics chronically would benefit most from the protocol. “If patients are diuretic-naive, they probably will respond well to whatever you do; if you just give a higher dose, they will respond well,” he said. “We expected that the largest benefit would be in patients already taking diuretics because they have a higher chance of not responding well.”
“There also was a big difference in the starting dose,” he added. “In the SOC arm, the baseline dose was about 60 mg, whereas we gave 120 mg, and we could already see a high difference in the effect. So, in those patients, I think the gain is bigger if you follow the protocol.”
More data coming
Looking ahead, “we only showed efficacy in the first 2 days of treatment and a shorter length of stay, probably reflecting a faster decongestion, but we don’t know for sure,” Dr. Dauw told this news organization.
“It would be important to have a study where the protocol is followed until full decongestion is reached,” he said. “That way, we can directly prove that decongestion is better and/or faster with the protocol.”
“A good decongestive strategy is one that is fast, safe and effective in decreasing signs and symptoms that patients suffer from,” he added. “We believe our protocol can achieve that, but our study is only one piece of the puzzle.”
More data on natriuresis-guided decongestion is coming this year, he said, with the PUSH-AHF study from Groningen, the European DECONGEST study, and the U.S. ESCALATE study.
The study had no funding. Dr. Dauw declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM HFA-ESC 2023
High Lp(a) tied to higher coronary plaque volume, progression
MANNHEIM, GERMANY – , an observational imaging study shows.
This could explain the greater risk for major adverse cardiovascular events seen in patients with high Lp(a) levels, suggests the research, presented during the annual European Atherosclerosis Society Congress.
The team performed follow-up coronary CT angiography (CCTA) on almost 275 patients who had undergone imaging approximately 10 years earlier, finding that almost one-third had high Lp(a) levels.
At baseline, per cent plaque volumes were 1.8 times greater in high Lp(a) patients versus those with low levels of the protein. After 10 years, plaque volumes were 3.3 times larger in patients with high Lp(a) levels.
Over this period, the rate of increase of plaque volume was 1.9 times greater in patients with high Lp(a) levels.
Study presenter Nick S. Nurmohamed, MD, PhD candidate, department of vascular medicine, Amsterdam University Medical Centers, also showed that high Lp(a) levels were associated with a 2.1-fold increase in rates of MACE.
He said in an interview that this finding could be related to Lp(a) increasing inflammatory signaling in the plaque, “making it more prone to rupture, and we saw that on the CCTA scans,” where high Lp(a) levels were associated with the presence of more high-risk plaques.
He added that in the absence of drugs that target Lp(a) levels directly, the results underline the need to focus on other means of lipid-lowering, as well as “creating awareness that Lp(a) is associated with plaque formation.”
Dr. Nurmohamed said that “for the moment, we have to treat patients with high Lp(a) with other risk-lowering therapies, such as low-density lipoprotein [LDL] cholesterol–lowering drugs, and the management of other risk factors.”
However, he noted that “there are a couple of Lp(a)-lowering medications in trials,” with results expected in the next 2-3 years.
“Then we will have the means to treat those patients, and with CCTA we can identify the patients with the biggest risk,” Dr. Nurmohamed added.
Plaque burden
Philippe Moulin, MD, PhD, head of endocrinology and professor of human nutrition at Faculté Lyon Est, Claude Bernard Lyon (France) 1 University, said that the association between Lp(a) and plaque burden has been seen previously in the literature in a very similar study but with only 1-year follow-up.
Similarly, registry data have suggested that Lp(a) is associated with worsening plaque progression over time.
“Here, with 10-year follow-up, [the study] is much more interesting,” due to its greater statistical power, he said in an interview. It is also “well-documented” and uses an “appropriate” methodology.
But Dr. Moulin underlined that the number of patients with high Lp(a) levels included in the study is relatively small.
Consequently, the researchers were not able to look at the level and rate of progression of atherosclerosis between different quartiles of Lp(a), “so you have no dose-response analysis.”
It also does not “establish causality,” as it remains an observational study, despite being longitudinal, “well done, and so on.”
Dr. Moulin added that the study nevertheless adds “one more stone” to the construct of the idea of high risk around high Lp(a) levels, and “prepares the ground” for the availability of two drugs to decrease Lp(a) levels, expected in 2026 and 2027.
These are expected to substantially reduce Lp(a) levels and achieve a reduction in cardiovascular risk of around 20%-40%, “which would be interesting,” especially as “we have patients who have Lp(a) levels four times above the upper normal value.”
Crucially, they may already have normal LDL cholesterol levels, meaning that, for some patients, “there is clearly a need for such treatment, as long as it is proven that it will decrease cardiovascular risk.”
For the moment, however, the strategy for managing patients with high Lp(a) remains to increase the dose of statin and to have more stringent targets, although Dr. Moulin pointed out that, “when you give statins, you raise slightly Lp(a) levels.”
Dr. Nurmohamed said in an interview that “we know from largely genetic and observational studies that Lp(a) is causally associated with atherosclerotic cardiovascular disease.”
What is less clear is the exact underlying mechanism, he said, noting that there have been several imaging studies in high and low Lp(a) patients that have yielded conflicting results in terms of the relationship with plaque burden.
To investigate the impact of Lp(a) levels on long-term coronary plaque progression, the team invited patients who had taken part in a previous CCTA study to undergo repeat CCTA, regardless of their underlying symptoms.
In all, 299 patients underwent follow-up imaging a median of 10.2 years after their original scan. Plaque volumes were quantified and adjusted for vessel volumes, and the patients were classified as having high (≥ 70 nmol/L) or low (< 70 nmol/L) Lp(a) levels.
After excluding patients who had undergone coronary artery bypass grafting, the team analyzed 274 patients with a mean age at baseline of 57 years. Of these, 159 (58%) were men. High Lp(a) levels were identified in 87 (32%) patients.
The team found that at baseline, patients with high Lp(a) levels had significantly larger percent atheroma volumes than those with low levels, at 3.92% versus 2.17%, or an absolute difference of 1.75% (P = .013).
The difference between the two groups was even greater at the follow-up, when percent atheroma volumes reached 8.75% in patients with high Lp(a) levels versus 3.90% for those with low levels, or an absolute difference of 4.85% (P = .005).
Similar findings were seen when looking separately at percentage of noncalcified and calcified plaque volumes as well as when analyzing for the presence of low-density plaques.
Multivariate analysis taking into account clinical risk factors, statin use, and CT tube voltage found that high Lp(a) levels were associated with a greater percent atheroma volume at baseline, at an odds ratio versus low Lp(a) of 1.83 (95% confidence interval, 0.12-3.54; P = .037).
High Lp(a) levels were also linked to a larger percent atheroma volume on follow-up imaging, at an odds ratio of 3.25 (95% CI, 0.80-5.71; P = .010), and a significantly greater change in atheroma volume from baseline to follow-up imaging, at an odds ratio of 1.86 (95% CI, 0.59-3.14; P = .005)
Finally, the team showed that, after adjusting for clinical risk factors, high baseline Lp(a) levels were associated with an increased risk of MACE during the follow-up period, at a hazard ratio versus low Lp(a) levels of 2.10 (95% CI, 1.01-4.29, P = .048).
No funding was declared. Dr. Nurmohamed is cofounder of Lipid Tools. Other authors declare relationships with Amgen, Novartis, Esperion, Sanofi-Regeneron, Ackee, Cleerly, GW Heart and Vascular Institute, Siemens Healthineers, and HeartFlow.
A version of this article first appeared on Medscape.com.
MANNHEIM, GERMANY – , an observational imaging study shows.
This could explain the greater risk for major adverse cardiovascular events seen in patients with high Lp(a) levels, suggests the research, presented during the annual European Atherosclerosis Society Congress.
The team performed follow-up coronary CT angiography (CCTA) on almost 275 patients who had undergone imaging approximately 10 years earlier, finding that almost one-third had high Lp(a) levels.
At baseline, per cent plaque volumes were 1.8 times greater in high Lp(a) patients versus those with low levels of the protein. After 10 years, plaque volumes were 3.3 times larger in patients with high Lp(a) levels.
Over this period, the rate of increase of plaque volume was 1.9 times greater in patients with high Lp(a) levels.
Study presenter Nick S. Nurmohamed, MD, PhD candidate, department of vascular medicine, Amsterdam University Medical Centers, also showed that high Lp(a) levels were associated with a 2.1-fold increase in rates of MACE.
He said in an interview that this finding could be related to Lp(a) increasing inflammatory signaling in the plaque, “making it more prone to rupture, and we saw that on the CCTA scans,” where high Lp(a) levels were associated with the presence of more high-risk plaques.
He added that in the absence of drugs that target Lp(a) levels directly, the results underline the need to focus on other means of lipid-lowering, as well as “creating awareness that Lp(a) is associated with plaque formation.”
Dr. Nurmohamed said that “for the moment, we have to treat patients with high Lp(a) with other risk-lowering therapies, such as low-density lipoprotein [LDL] cholesterol–lowering drugs, and the management of other risk factors.”
However, he noted that “there are a couple of Lp(a)-lowering medications in trials,” with results expected in the next 2-3 years.
“Then we will have the means to treat those patients, and with CCTA we can identify the patients with the biggest risk,” Dr. Nurmohamed added.
Plaque burden
Philippe Moulin, MD, PhD, head of endocrinology and professor of human nutrition at Faculté Lyon Est, Claude Bernard Lyon (France) 1 University, said that the association between Lp(a) and plaque burden has been seen previously in the literature in a very similar study but with only 1-year follow-up.
Similarly, registry data have suggested that Lp(a) is associated with worsening plaque progression over time.
“Here, with 10-year follow-up, [the study] is much more interesting,” due to its greater statistical power, he said in an interview. It is also “well-documented” and uses an “appropriate” methodology.
But Dr. Moulin underlined that the number of patients with high Lp(a) levels included in the study is relatively small.
Consequently, the researchers were not able to look at the level and rate of progression of atherosclerosis between different quartiles of Lp(a), “so you have no dose-response analysis.”
It also does not “establish causality,” as it remains an observational study, despite being longitudinal, “well done, and so on.”
Dr. Moulin added that the study nevertheless adds “one more stone” to the construct of the idea of high risk around high Lp(a) levels, and “prepares the ground” for the availability of two drugs to decrease Lp(a) levels, expected in 2026 and 2027.
These are expected to substantially reduce Lp(a) levels and achieve a reduction in cardiovascular risk of around 20%-40%, “which would be interesting,” especially as “we have patients who have Lp(a) levels four times above the upper normal value.”
Crucially, they may already have normal LDL cholesterol levels, meaning that, for some patients, “there is clearly a need for such treatment, as long as it is proven that it will decrease cardiovascular risk.”
For the moment, however, the strategy for managing patients with high Lp(a) remains to increase the dose of statin and to have more stringent targets, although Dr. Moulin pointed out that, “when you give statins, you raise slightly Lp(a) levels.”
Dr. Nurmohamed said in an interview that “we know from largely genetic and observational studies that Lp(a) is causally associated with atherosclerotic cardiovascular disease.”
What is less clear is the exact underlying mechanism, he said, noting that there have been several imaging studies in high and low Lp(a) patients that have yielded conflicting results in terms of the relationship with plaque burden.
To investigate the impact of Lp(a) levels on long-term coronary plaque progression, the team invited patients who had taken part in a previous CCTA study to undergo repeat CCTA, regardless of their underlying symptoms.
In all, 299 patients underwent follow-up imaging a median of 10.2 years after their original scan. Plaque volumes were quantified and adjusted for vessel volumes, and the patients were classified as having high (≥ 70 nmol/L) or low (< 70 nmol/L) Lp(a) levels.
After excluding patients who had undergone coronary artery bypass grafting, the team analyzed 274 patients with a mean age at baseline of 57 years. Of these, 159 (58%) were men. High Lp(a) levels were identified in 87 (32%) patients.
The team found that at baseline, patients with high Lp(a) levels had significantly larger percent atheroma volumes than those with low levels, at 3.92% versus 2.17%, or an absolute difference of 1.75% (P = .013).
The difference between the two groups was even greater at the follow-up, when percent atheroma volumes reached 8.75% in patients with high Lp(a) levels versus 3.90% for those with low levels, or an absolute difference of 4.85% (P = .005).
Similar findings were seen when looking separately at percentage of noncalcified and calcified plaque volumes as well as when analyzing for the presence of low-density plaques.
Multivariate analysis taking into account clinical risk factors, statin use, and CT tube voltage found that high Lp(a) levels were associated with a greater percent atheroma volume at baseline, at an odds ratio versus low Lp(a) of 1.83 (95% confidence interval, 0.12-3.54; P = .037).
High Lp(a) levels were also linked to a larger percent atheroma volume on follow-up imaging, at an odds ratio of 3.25 (95% CI, 0.80-5.71; P = .010), and a significantly greater change in atheroma volume from baseline to follow-up imaging, at an odds ratio of 1.86 (95% CI, 0.59-3.14; P = .005)
Finally, the team showed that, after adjusting for clinical risk factors, high baseline Lp(a) levels were associated with an increased risk of MACE during the follow-up period, at a hazard ratio versus low Lp(a) levels of 2.10 (95% CI, 1.01-4.29, P = .048).
No funding was declared. Dr. Nurmohamed is cofounder of Lipid Tools. Other authors declare relationships with Amgen, Novartis, Esperion, Sanofi-Regeneron, Ackee, Cleerly, GW Heart and Vascular Institute, Siemens Healthineers, and HeartFlow.
A version of this article first appeared on Medscape.com.
MANNHEIM, GERMANY – , an observational imaging study shows.
This could explain the greater risk for major adverse cardiovascular events seen in patients with high Lp(a) levels, suggests the research, presented during the annual European Atherosclerosis Society Congress.
The team performed follow-up coronary CT angiography (CCTA) on almost 275 patients who had undergone imaging approximately 10 years earlier, finding that almost one-third had high Lp(a) levels.
At baseline, per cent plaque volumes were 1.8 times greater in high Lp(a) patients versus those with low levels of the protein. After 10 years, plaque volumes were 3.3 times larger in patients with high Lp(a) levels.
Over this period, the rate of increase of plaque volume was 1.9 times greater in patients with high Lp(a) levels.
Study presenter Nick S. Nurmohamed, MD, PhD candidate, department of vascular medicine, Amsterdam University Medical Centers, also showed that high Lp(a) levels were associated with a 2.1-fold increase in rates of MACE.
He said in an interview that this finding could be related to Lp(a) increasing inflammatory signaling in the plaque, “making it more prone to rupture, and we saw that on the CCTA scans,” where high Lp(a) levels were associated with the presence of more high-risk plaques.
He added that in the absence of drugs that target Lp(a) levels directly, the results underline the need to focus on other means of lipid-lowering, as well as “creating awareness that Lp(a) is associated with plaque formation.”
Dr. Nurmohamed said that “for the moment, we have to treat patients with high Lp(a) with other risk-lowering therapies, such as low-density lipoprotein [LDL] cholesterol–lowering drugs, and the management of other risk factors.”
However, he noted that “there are a couple of Lp(a)-lowering medications in trials,” with results expected in the next 2-3 years.
“Then we will have the means to treat those patients, and with CCTA we can identify the patients with the biggest risk,” Dr. Nurmohamed added.
Plaque burden
Philippe Moulin, MD, PhD, head of endocrinology and professor of human nutrition at Faculté Lyon Est, Claude Bernard Lyon (France) 1 University, said that the association between Lp(a) and plaque burden has been seen previously in the literature in a very similar study but with only 1-year follow-up.
Similarly, registry data have suggested that Lp(a) is associated with worsening plaque progression over time.
“Here, with 10-year follow-up, [the study] is much more interesting,” due to its greater statistical power, he said in an interview. It is also “well-documented” and uses an “appropriate” methodology.
But Dr. Moulin underlined that the number of patients with high Lp(a) levels included in the study is relatively small.
Consequently, the researchers were not able to look at the level and rate of progression of atherosclerosis between different quartiles of Lp(a), “so you have no dose-response analysis.”
It also does not “establish causality,” as it remains an observational study, despite being longitudinal, “well done, and so on.”
Dr. Moulin added that the study nevertheless adds “one more stone” to the construct of the idea of high risk around high Lp(a) levels, and “prepares the ground” for the availability of two drugs to decrease Lp(a) levels, expected in 2026 and 2027.
These are expected to substantially reduce Lp(a) levels and achieve a reduction in cardiovascular risk of around 20%-40%, “which would be interesting,” especially as “we have patients who have Lp(a) levels four times above the upper normal value.”
Crucially, they may already have normal LDL cholesterol levels, meaning that, for some patients, “there is clearly a need for such treatment, as long as it is proven that it will decrease cardiovascular risk.”
For the moment, however, the strategy for managing patients with high Lp(a) remains to increase the dose of statin and to have more stringent targets, although Dr. Moulin pointed out that, “when you give statins, you raise slightly Lp(a) levels.”
Dr. Nurmohamed said in an interview that “we know from largely genetic and observational studies that Lp(a) is causally associated with atherosclerotic cardiovascular disease.”
What is less clear is the exact underlying mechanism, he said, noting that there have been several imaging studies in high and low Lp(a) patients that have yielded conflicting results in terms of the relationship with plaque burden.
To investigate the impact of Lp(a) levels on long-term coronary plaque progression, the team invited patients who had taken part in a previous CCTA study to undergo repeat CCTA, regardless of their underlying symptoms.
In all, 299 patients underwent follow-up imaging a median of 10.2 years after their original scan. Plaque volumes were quantified and adjusted for vessel volumes, and the patients were classified as having high (≥ 70 nmol/L) or low (< 70 nmol/L) Lp(a) levels.
After excluding patients who had undergone coronary artery bypass grafting, the team analyzed 274 patients with a mean age at baseline of 57 years. Of these, 159 (58%) were men. High Lp(a) levels were identified in 87 (32%) patients.
The team found that at baseline, patients with high Lp(a) levels had significantly larger percent atheroma volumes than those with low levels, at 3.92% versus 2.17%, or an absolute difference of 1.75% (P = .013).
The difference between the two groups was even greater at the follow-up, when percent atheroma volumes reached 8.75% in patients with high Lp(a) levels versus 3.90% for those with low levels, or an absolute difference of 4.85% (P = .005).
Similar findings were seen when looking separately at percentage of noncalcified and calcified plaque volumes as well as when analyzing for the presence of low-density plaques.
Multivariate analysis taking into account clinical risk factors, statin use, and CT tube voltage found that high Lp(a) levels were associated with a greater percent atheroma volume at baseline, at an odds ratio versus low Lp(a) of 1.83 (95% confidence interval, 0.12-3.54; P = .037).
High Lp(a) levels were also linked to a larger percent atheroma volume on follow-up imaging, at an odds ratio of 3.25 (95% CI, 0.80-5.71; P = .010), and a significantly greater change in atheroma volume from baseline to follow-up imaging, at an odds ratio of 1.86 (95% CI, 0.59-3.14; P = .005)
Finally, the team showed that, after adjusting for clinical risk factors, high baseline Lp(a) levels were associated with an increased risk of MACE during the follow-up period, at a hazard ratio versus low Lp(a) levels of 2.10 (95% CI, 1.01-4.29, P = .048).
No funding was declared. Dr. Nurmohamed is cofounder of Lipid Tools. Other authors declare relationships with Amgen, Novartis, Esperion, Sanofi-Regeneron, Ackee, Cleerly, GW Heart and Vascular Institute, Siemens Healthineers, and HeartFlow.
A version of this article first appeared on Medscape.com.
AT EAS 2023