User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.
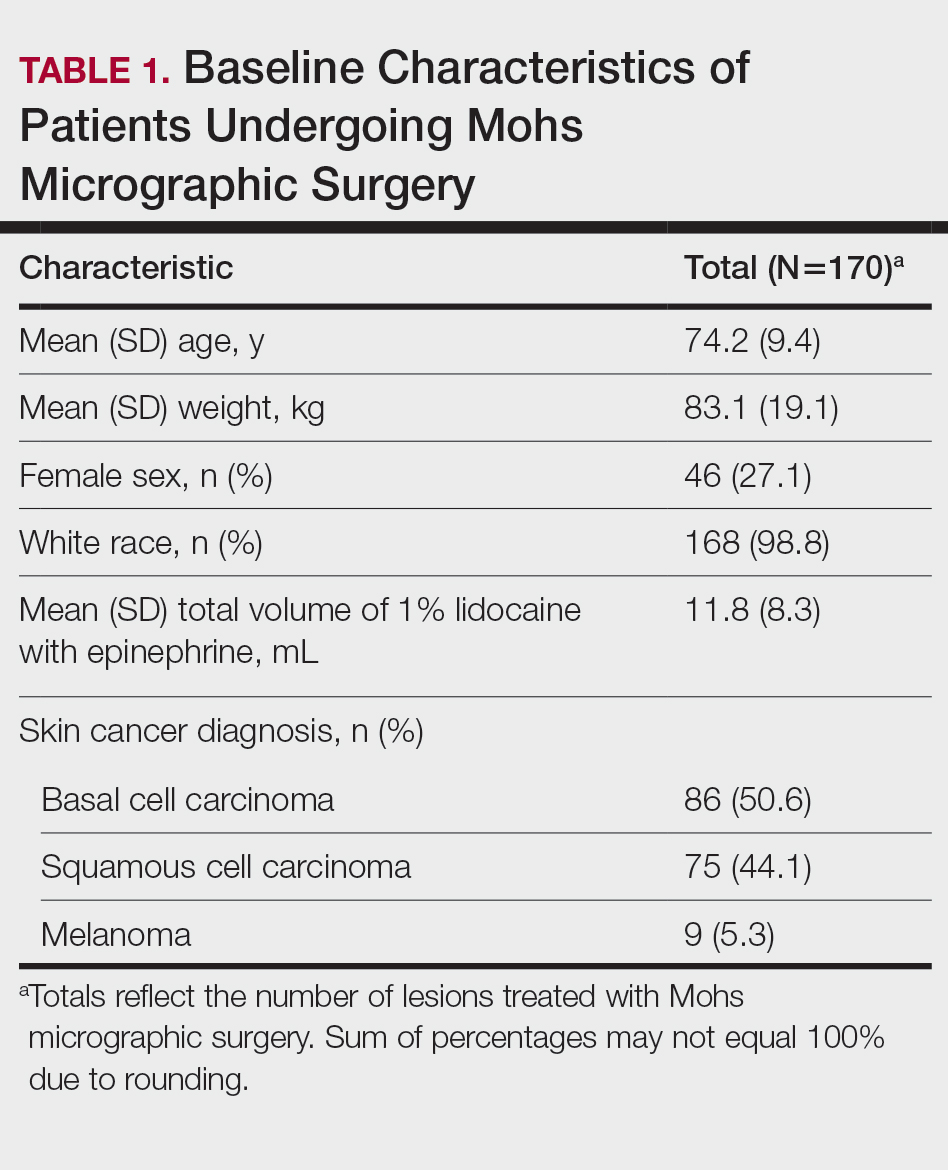
Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.
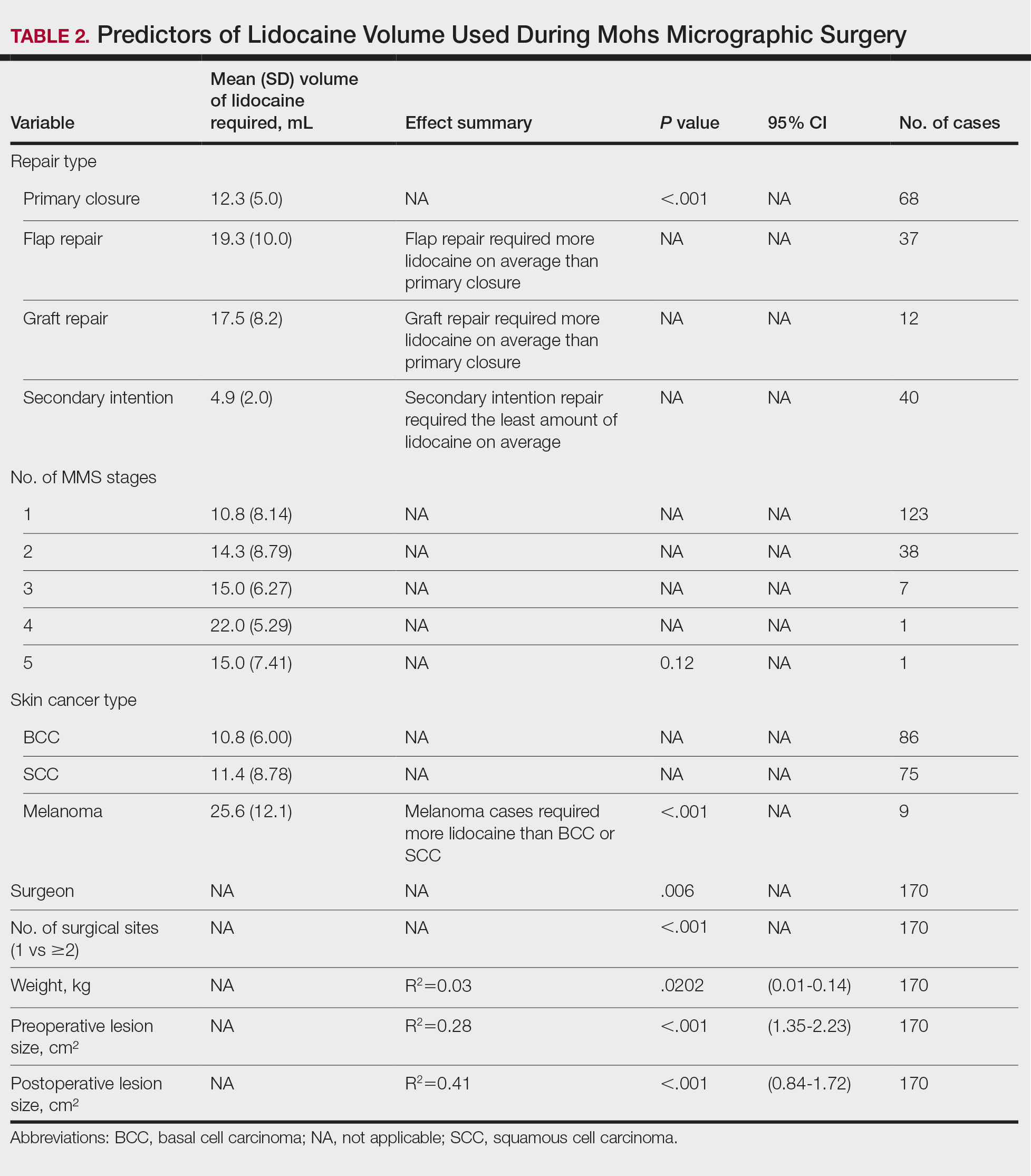
Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.

Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.

Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.

Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.

Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Practice Points
- Larger lesion size, melanoma diagnosis, and multiple surgical sites are associated with higher lidocaine volume requirements during Mohs micrographic surgery.
- Melanomas required more than twice the average lidocaine volume compared with basal cell carcinomas and squamous cell carcinomas.
- Flap and graft repairs require substantially more lidocaine than primary closures, while secondary-intention healing uses the least, making reconstruction type an important predictor of total anesthetic needs.
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).
In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).

In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
Chromoblastomycosis is a neglected tropical implantation mycosis caused by dematiaceous fungi that leads to substantial morbidity. This condition is diagnosed microscopically by visualizing the characteristic thick-walled, single, or multicellular clusters of pigmented fungal cells (also known as medlar bodies, muriform cells, or sclerotic bodies).1 The main causative fungi varies by geographic region, but most commonly is caused by Cladophialophora carrionii, Fonsecaea species, Phialophora verrucosa species complex, and Rhinocladiella aquaspersa.2-4 Standardized treatment guidelines have not been established, but itraconazole typically is considered first-line regardless of causative fungi.5 Terbinafine, other azoles, and topical immunomodulators, either as monotherapy or in combination, may be appropriate alternative or adjunctive options for refractory disease, although supporting data are limited.6-9
Complications from chromoblastomycosis are common, particularly in long-standing, severe, or refractory disease. An analysis using billing codes in the United States found 14% (35/255) of hospitalized patients with chromoblastomycosis had lymphedema.10 In Mexico, 63% (32/51) of patients with chromoblastomycosis developed secondary bacterial infections.11 Skin fibrosis and ankylosis also can occur and cause mobility issues and decreased quality of life. An infrequent but potentially life-threatening complication12 is the development of squamous cell carcinoma (SCC) associated with chronic lesions, representing a preventable end-stage complication of delayed diagnosis and treatment (Figure).

In this review, we summarize reported epidemiology and clinical risk factors for SCC complicating chromoblastomycosis. We also discuss plausible inflammatory mechanisms of malignant transformation and propose pragmatic clinical and public health interventions, including decentralized microscopy-based diagnosis, timely antifungal access, and biopsy-triggered surveillance of chronically inflamed lesions, to reduce preventable morbidity.
Epidemiology and Risk Factors
The epidemiology of SCC developing from chromoblastomycosis is not well understood due to gaps in national and global surveillance. Some studies have found that 2% to 13% of patients with chromoblastomycosis developed SCC.4,11,13-15 Based on case reports and case series, a symptom duration of more than 10 years appears to be the most substantial risk factor for the development of SCC rather than host immune status.16-18 Severity, specifically the size of the injury, and vegetating lesions also have been suggested as risk factors for the development of SCC.16 Additionally, the appearance of new lesions (mainly ulcers not related to secondary infection) that appear during the healing phase should raise the suspicion of SCC and warrant a biopsy for evaluation.16
Pathophysiology
The exact mechanism of malignant transformation has not been elucidated, but histopathologic features suggest substantial epidermal proliferation. In some cases, this leads to pseudoepitheliomatous hyperplasia, a nonmalignant hyperproliferative state that is an important differential HPV to leishmaniasis and lupus vulgaris.19 The chronic inflammation from long-standing chromoblastomycosis likely contributes to the further malignant transformation to SCC.
Polymorphonuclear cells and activated macrophages seen in chronic inflammation can promote the release of enzymes and free radicals that has led to malignant transformation in vitro but has not been investigated specifically in chromoblastomycosis.16 Additionally, chronic inflammation and metabolic products of phagocytosis often are accompanied by excessive production of reactive oxygen and nitrogen species, which can damage DNA, lipoproteins, and cell membranes. Other potential contributors include the expression of cyclooxygenase 2 and release of arachidonic acid metabolites (eg, prostaglandins, leukotrienes), which can damage the cell and promote carcinogenesis. It is not clear whether similar mechanisms account for the development of SCCs in other chronic skin inflammations or infections such as cutaneous tuberculosis or Marjolin ulcers.20
Clinical and Public Health Interventions
Squamous cell carcinoma arising in the setting of chromoblastomycosis warrants prompt oncologic evaluation and definitive surgical management, which may require extensive surgical excision and, in advanced disease, amputation.14,17,18 Advanced malignant tumors can be difficult to manage and can result in death.21,22 Additionally, clinicians should maintain a low threshold for biopsy in long-standing chromoblastomycosis, particularly when lesions demonstrate new ulceration, rapid growth, bleeding, pain, malodor, or failure to improve with appropriate antifungal therapy.16 Recurrent or new lesions after amputation may indicate persistent or recurrent infection and may require continued antifungal management alongside cancer care.16
Squamous cell carcinoma arising from chromoblastomycosis results after substantial diagnostic delays, allowing chronic inflammation to transform infection into malignancy. Separating benign inflammation-associated epidermal proliferation from transformation to SCC requires histopathologic skill. An assay based on increased expression of chromosome 15 open reading frame 48 (C15orf48), an immune regulatory protein, has been developed to aid in this distinction; however, it is not widely available.23
Raising awareness of chromoblastomycosis among clinicians and communities, particularly in rural areas where the disease is more common, is critical to improve health care–seeking behaviors and expedite access to care pathways.2 Furthermore, access and training on microscopy to diagnose chromoblastomycosis in decentralized areas can facilitate earlier diagnosis in primary health care settings rather than waiting for diagnosis in tertiary care settings, at which point disease usually is advanced. Global implementation of existing programs that use microscopy (eg, malaria in rural areas) can be partnered with frontline health worker cross-training on chromoblastomycosis diagnosis to improve appropriate identification of disease.24 Finally, improving access to affordable antifungals, particularly itraconazole, is necessary along with further research into novel therapeutic strategies. Approaches that utilize local manufacturing and pooled procurement could help expand treatment availability in parallel with diagnostic improvement initiatives.25
Final Thoughts
Squamous cell carcinoma resulting from chromoblastomycosis is a devastating complication, often leading to limb amputation. The true prevalence is unknown, but it occurs more commonly in long-standing disease without appropriate therapy. The appearance of new lesions or ulcers after initial improvement should increase suspicion and lead to biopsy and careful pathologic evaluation. Prevention of SCC requires increased clinical awareness, early diagnosis, and timely initiation of antifungal treatment. Enhanced surveillance among individuals with chromoblastomycosis would help to better understand its prevalence, associated risk factors, and impact on quality of life.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
- Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276.
- Smith DJ, Queiroz-Telles F, Rabenja FR, et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl Trop Dis. 2024;18:E0012562.
- Yen JS, Shih IH, Chung WH, et al. Chromoblastomycosis in northern Taiwan from 2017 to 2024: unique characteristics. Clin Exp Dermatol. Published online July 18, 2025. doi:10.1093/ced/llaf329
- Santos DWCL, Vicente VA, Weiss VA, et al. Chromoblastomycosis in an endemic area of Brazil: a clinical-epidemiological analysis and a worldwide haplotype network. J Fungi. 2020;6:204.
- Smith DJ, Melhem MSC, Dirven J, et al. Establishment of epidemiological cutoff values for Fonsecaea pedrosoi, the primary etiologic agent of chromoblastomycosis, and eight antifungal medications. J Clin Microbiol. 2025;63:E01903-24.
- Logan C, Singh M, Fox N, et al. Chromoblastomycosis treated with posaconazole and adjunctive imiquimod: lending innate immunity a helping hand. Open Forum Infect Dis. 2023;10:ofad124.
- de Sousa M da GT, Belda W, Spina R, et al. Topical application of imiquimod as a treatment for chromoblastomycosis. Clin Infect Dis. 2014;58:1734-1737.
- Criado PR, Careta MF, Valente NYS, et al. Extensive long-standing chromomycosis due to Fonsecaea pedrosoi: three cases with relevant improvement under voriconazole therapy. J Dermatol Treat. 2011;22:167-174.
- Esterre P, Inzan CK, Ramarcel ER, et al. Treatment of chromomycosis with terbinafine: preliminary results of an open pilot study. Br J Dermatol. 1996;134:33-36.
- Smith DJ, Benedict K, Lockhart SR, et al. Chromoblastomycosis and phaeohyphomycotic abscess-associated hospitalizations, United States, 2016–2021. PLoS Negl Trop Dis. 2025;19:E0013499.
- Bonifaz A, Carrasco‐Gerard E, Saúl A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses. 2001;44:1-7.
- Torres E, Beristain JG, Lievanos Z, et al. Chromoblastomycosis associated with a lethal squamous cell carcinoma. An Bras Dermatol. 2010;85:267-270.
- Verma S, Thakur BK, Raphael V, et al. Epidemiology of subcutaneous mycoses in northeast India: a retrospective study. Indian J Dermatol. 2018;63:496-501.
- Siregar GO, Harianja M, Rinonce HT, et al. Chromoblastomycosis: a case series from Sumba, eastern Indonesia. Clin Exp Dermatol. 2025;50:1447-1450.
- Valentin J, Grotta G, Muller T, et al. Chromoblastomycosis in French Guiana: epidemiology and practices, 1955-2023. J Fungi. 2024;10:168.
- Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60:1500-1504.
- Belda Jr W, Criado PR, Casteleti P, et al. Chromoblastomycosis evolving to sarcomatoid squamous cell carcinoma: a case report. Dermatol Rep. 2021;13:9009.
- Jamil A, Lee YY, Thevarajah S. Invasive squamous cell carcinoma arising from chromoblastomycosis. Med Mycol. 2012;50:99-102.
- Delahaye T, Orduz-Robledo M, Beltran A M, et al. Pseudo-epitheliomatous hyperplasia and skin infections. Open Dermatol J. 2024;18:E18743722304513.
- Fania L, Didona D, Di Pietro FR, et al. Cutaneous squamous cell carcinoma: from pathophysiology to novel therapeutic approaches. Biomedicines. 2021;9:171.
- Torres E, Beristain JG, Lievanos Z, et al. Carcinoma epidermoide como complicação letal de lesões crônicas de cromoblastomicose. An Bras Dermatol. 2010;85:267-270.
- Rojas OC, González GM, Moreno-Treviño M, et al. Chromoblastomycosis by Cladophialophora carrionii associated with squamous cell carcinoma and review of published reports. Mycopathologia. 2015;179:153-157.
- Su A, Ra S, Li X, et al. Differentiating cutaneous squamous cell carcinoma and pseudoepitheliomatous hyperplasia by multiplex qRT-PCR. Mod Pathol. 2013;26:1433-1437.
- Siregar GO, Harianja M, Smith DJ, et al. Leveraging malaria microscopy infrastructure to diagnose common and neglected skin diseases using direct microscopy in Sumba, Indonesia. Lancet Reg Health - West Pac. 2025;64:101739.
- Smith DJ, Soebono H, Parajuli N, et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg Health Southeast Asia. 2025;35:100561.
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Chromoblastomycosis Leading to Squamous Cell Carcinoma: An Overlooked Outcome of a Neglected Tropical Disease
Practice Points
- Chromoblastomycosis is recognized by the World Health Organization as a neglected tropical disease and principally affects agricultural workers in tropical and subtropical regions.
- Long-standing or refractory chromoblastomycosis can lead to substantial morbidity, including lymphedema, secondary bacterial infections, extensive scarring, functional impairment, and squamous cell carcinoma (SCC).
- The development of SCC is thought to be related to chronic inflammation and prolonged disease duration (Mathematical Pi LT Std>10 years). Advanced cases may require extensive surgical excision or amputation.
- Early recognition with support of direct microscopy or histopathology, timely antifungal treatment (often with itraconazole), and a low threshold for repeat biopsy of new ulceration or rapidly changing lesions may prevent malignant transformation and disability.
Enlarging Mass on the Scalp
Enlarging Mass on the Scalp
THE DIAGNOSIS: Malignant Proliferating Trichilemmal Tumor
Histologic examination revealed atypical keratinocytes, nuclear pleomorphism, and lobulating epithelial masses with trichilemmal keratinization (Figure). The presence of CD34 positivity, a marker of outer follicular root sheath–derived cells, supported the diagnosis of a malignant proliferating trichilemmal tumor (MPTT). Imaging also revealed signs of bone invasion, further supporting a malignant process. Based on these findings, the patient underwent complete excision of the mass with scalp reconstruction, lymph node dissection, and systemic evaluation for metastases. Final pathology confirmed negative surgical margins and no lymph node involvement. Adjuvant radiation was not required, given the absence of skull invasion or confirmed distant metastasis.
The differential diagnosis for rapidly enlarging scalp tumors can be broad and includes both benign and malignant processes. In this patient, the differential diagnoses included trichilemmal carcinoma, cutaneous squamous cell carcinoma (SCC), sebaceous carcinoma (SC), proliferating trichilemmal tumor (PTT), and MPTT. Due to the notable clinical and histologic overlap among these lesions, definitive diagnosis required histopathologic evaluation in our patient.
Proliferating trichilemmal tumors were first described in 1966 by Wilson-Jones,1 who used the term proliferating epidermoid cysts, noting their distinct histologic features and resemblance to SCC.2 These tumors generally are benign and arise from the isthmus of the outer root sheath of the hair follicle; however, malignant transformation can occur, resulting in a rare entity known as MPTT. This malignant variant was first described in 1983 by Saida et al,3 who emphasized its distinct clinical behavior, including infiltrative growth, high mitotic activity, and potential for local recurrence and metastasis.
A recent literature review identified 60 reported cases of MPTT, with an average patient age of 57 years and a female predominance.4 Clinically, MPTTs often manifest as large (>5 cm) lobulated masses located on sun-exposed, hair-bearing areas of the skin, especially the scalp. These lesions may be flesh-colored to pink and often exhibit ulceration, necrosis, or calcification.5 Typically, MPTTs follow a biphasic course, beginning with a slow-growing phase followed by a period of rapid growth. Due to their aggressive behavior and resemblance to other cutaneous malignancies, accurate differentiation of MPTT from benign PTTs, cutaneous SCCs, SCs, and trichilemmal carcinomas is critical.
Malignant proliferating trichilemmal tumors demonstrate a substantially higher metastatic potential than either benign PTTs or cutaneous SCCs. While cutaneous SCCs carry a metastasis rate of approximately 1.9% to 2.6%, MPTTs carry a considerably higher rate of approximately 25.0%.6 Regional lymphatic spread is the most common route of dissemination, making comprehensive lymph node assessment—both radiographic and clinical—an important component of tumor staging. When lymph node involvement is suspected, surgical dissection may be indicated, along with consideration of adjuvant therapies.
Histopathologically, MPTT is characterized by nuclear atypia, mitotic figures, and lobulated masses of proliferating epithelium showing trichilemmal differentiation and infiltrative growth.4 The presence of CD34 positivity, reflecting outer follicular root sheath differentiation, helps distinguish MPTT from cutaneous SCC and SC, which typically lack this marker.6,7 Immunohistochemistry is therefore a valuable adjunct in differentiating these lesions.
The mainstay of treatment for MPTT is wide local excision with clear margins. Margins of at least 1 cm generally are recommended. Although Mohs micrographic surgery may be used in anatomically sensitive areas, it typically is not preferred due to the potential for skip lesions in MPTT, which may lead to incomplete excision and recurrence.8 In cases with evidence of regional or distant metastasis or when clear margins cannot be achieved confidently, adjunctive treatments such as radiation therapy and systemic chemotherapy may be indicated. Preoperative imaging is used to evaluate for local invasion (skull or bone involvement) and regional lymph node status, which may inform adjuvant therapy postoperatively.
The prognosis for MPTT is variable and depends largely on early recognition, accurate histopathologic diagnosis, complete surgical excision with clear margins, and the presence or absence of metastasis. When the tumor is fully excised with negative margins and no lymph node involvement, the risk for recurrence is substantially reduced; however, MPTT is known for its potential aggressive behavior. Delays in diagnosis or incomplete resection can lead to local recurrence, regional spread, or even distant metastasis. In the literature review discussed previously, the mortality rate of patients with MPTT was 11.7%,4 which is notably higher than that of more common cutaneous malignancies such as cutaneous SCC, which is reported at 1.2%.9
The clinical course of MPTT remains difficult to predict due to its rarity and the limited availability of large-scale studies. Most published data are derived from isolated case reports or small case series, making standardized treatment guidelines challenging. Given this uncertainty, long-term follow-up is strongly recommended to monitor for recurrence or metastatic progression.2
This case highlights the critical role of clinicopathologic correlation in the evaluation of atypical or rapidly growing scalp lesions. The expertise of dermatologists in recognizing atypical presentations, combined with precise histopathologic analysis, including immunohistochemical staining, is vital to ensuring accurate diagnosis and optimal treatment. Early intervention can improve patient outcomes by reducing the risk for local recurrence and metastatic progression as well as the need for more intensive therapies.
- Jones EW. Proliferating epidermoid cysts. Arch Dermatol. 1966;94:11-19.
- Kemaloglu CA, Öztürk M, Aydın B, et al. Malignant proliferating trichilemmal tumor of the scalp: report of 4 cases and a short review of the literature. Case Reports Plast Surg Hand Surg. 2022;9:158-164. doi:10.1080/23320885.2022.2077208
- Saida T, Oohard K, Hori Y, et al. Development of a malignant proliferating trichilemmal cyst in a patient with multiple trichilemmal cysts. Dermatology. 1983;166:203-208. doi:10.1159/000249868
- Abdelhammed MH, Siatecka H, Diwan AH, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology (Basel). 2024;11:354-363. doi:10.3390 /dermatopathology11040038
- Fronek L, Brahs A, Farsi M, et al. A rare case of trichilemmal carcinoma: histology and management. J Clin Aesthet Dermatol. 2021;14:25-30.
- Osto M, Parry N, Rehman R, et al. Malignant proliferating trichilemmal tumor of the scalp: a systematic review. Am J Dermatopathol. 2021;43:851-866. doi:10.1097/DAD.0000000000001991
- Plaza JA, Mackinnon A, Carrillo L, et al. Role of immunohistochemistry in the diagnosis of sebaceous carcinoma: a clinicopathologic and immunohistochemical study. Am J Dermatopathol. 2015;37:809-821. doi:10.1097/DAD.0000000000000255
- Singh P, Usman A, Motta L, et al. Malignant proliferating trichilemmal tumour. BMJ Case Rep. Published online August 17, 2018. doi:10.1136/bcr-2018-224460
- Ran NA, Granger EE, Brodland DG, et al. Risk factor number and recurrence, metastasis, and disease-related death in cutaneous squamous cell carcinoma. JAMA Dermatol. 2025;161:597-604. doi:10.1001/jamadermatol.2025.0128
THE DIAGNOSIS: Malignant Proliferating Trichilemmal Tumor
Histologic examination revealed atypical keratinocytes, nuclear pleomorphism, and lobulating epithelial masses with trichilemmal keratinization (Figure). The presence of CD34 positivity, a marker of outer follicular root sheath–derived cells, supported the diagnosis of a malignant proliferating trichilemmal tumor (MPTT). Imaging also revealed signs of bone invasion, further supporting a malignant process. Based on these findings, the patient underwent complete excision of the mass with scalp reconstruction, lymph node dissection, and systemic evaluation for metastases. Final pathology confirmed negative surgical margins and no lymph node involvement. Adjuvant radiation was not required, given the absence of skull invasion or confirmed distant metastasis.

The differential diagnosis for rapidly enlarging scalp tumors can be broad and includes both benign and malignant processes. In this patient, the differential diagnoses included trichilemmal carcinoma, cutaneous squamous cell carcinoma (SCC), sebaceous carcinoma (SC), proliferating trichilemmal tumor (PTT), and MPTT. Due to the notable clinical and histologic overlap among these lesions, definitive diagnosis required histopathologic evaluation in our patient.
Proliferating trichilemmal tumors were first described in 1966 by Wilson-Jones,1 who used the term proliferating epidermoid cysts, noting their distinct histologic features and resemblance to SCC.2 These tumors generally are benign and arise from the isthmus of the outer root sheath of the hair follicle; however, malignant transformation can occur, resulting in a rare entity known as MPTT. This malignant variant was first described in 1983 by Saida et al,3 who emphasized its distinct clinical behavior, including infiltrative growth, high mitotic activity, and potential for local recurrence and metastasis.
A recent literature review identified 60 reported cases of MPTT, with an average patient age of 57 years and a female predominance.4 Clinically, MPTTs often manifest as large (>5 cm) lobulated masses located on sun-exposed, hair-bearing areas of the skin, especially the scalp. These lesions may be flesh-colored to pink and often exhibit ulceration, necrosis, or calcification.5 Typically, MPTTs follow a biphasic course, beginning with a slow-growing phase followed by a period of rapid growth. Due to their aggressive behavior and resemblance to other cutaneous malignancies, accurate differentiation of MPTT from benign PTTs, cutaneous SCCs, SCs, and trichilemmal carcinomas is critical.
Malignant proliferating trichilemmal tumors demonstrate a substantially higher metastatic potential than either benign PTTs or cutaneous SCCs. While cutaneous SCCs carry a metastasis rate of approximately 1.9% to 2.6%, MPTTs carry a considerably higher rate of approximately 25.0%.6 Regional lymphatic spread is the most common route of dissemination, making comprehensive lymph node assessment—both radiographic and clinical—an important component of tumor staging. When lymph node involvement is suspected, surgical dissection may be indicated, along with consideration of adjuvant therapies.
Histopathologically, MPTT is characterized by nuclear atypia, mitotic figures, and lobulated masses of proliferating epithelium showing trichilemmal differentiation and infiltrative growth.4 The presence of CD34 positivity, reflecting outer follicular root sheath differentiation, helps distinguish MPTT from cutaneous SCC and SC, which typically lack this marker.6,7 Immunohistochemistry is therefore a valuable adjunct in differentiating these lesions.
The mainstay of treatment for MPTT is wide local excision with clear margins. Margins of at least 1 cm generally are recommended. Although Mohs micrographic surgery may be used in anatomically sensitive areas, it typically is not preferred due to the potential for skip lesions in MPTT, which may lead to incomplete excision and recurrence.8 In cases with evidence of regional or distant metastasis or when clear margins cannot be achieved confidently, adjunctive treatments such as radiation therapy and systemic chemotherapy may be indicated. Preoperative imaging is used to evaluate for local invasion (skull or bone involvement) and regional lymph node status, which may inform adjuvant therapy postoperatively.
The prognosis for MPTT is variable and depends largely on early recognition, accurate histopathologic diagnosis, complete surgical excision with clear margins, and the presence or absence of metastasis. When the tumor is fully excised with negative margins and no lymph node involvement, the risk for recurrence is substantially reduced; however, MPTT is known for its potential aggressive behavior. Delays in diagnosis or incomplete resection can lead to local recurrence, regional spread, or even distant metastasis. In the literature review discussed previously, the mortality rate of patients with MPTT was 11.7%,4 which is notably higher than that of more common cutaneous malignancies such as cutaneous SCC, which is reported at 1.2%.9
The clinical course of MPTT remains difficult to predict due to its rarity and the limited availability of large-scale studies. Most published data are derived from isolated case reports or small case series, making standardized treatment guidelines challenging. Given this uncertainty, long-term follow-up is strongly recommended to monitor for recurrence or metastatic progression.2
This case highlights the critical role of clinicopathologic correlation in the evaluation of atypical or rapidly growing scalp lesions. The expertise of dermatologists in recognizing atypical presentations, combined with precise histopathologic analysis, including immunohistochemical staining, is vital to ensuring accurate diagnosis and optimal treatment. Early intervention can improve patient outcomes by reducing the risk for local recurrence and metastatic progression as well as the need for more intensive therapies.
THE DIAGNOSIS: Malignant Proliferating Trichilemmal Tumor
Histologic examination revealed atypical keratinocytes, nuclear pleomorphism, and lobulating epithelial masses with trichilemmal keratinization (Figure). The presence of CD34 positivity, a marker of outer follicular root sheath–derived cells, supported the diagnosis of a malignant proliferating trichilemmal tumor (MPTT). Imaging also revealed signs of bone invasion, further supporting a malignant process. Based on these findings, the patient underwent complete excision of the mass with scalp reconstruction, lymph node dissection, and systemic evaluation for metastases. Final pathology confirmed negative surgical margins and no lymph node involvement. Adjuvant radiation was not required, given the absence of skull invasion or confirmed distant metastasis.

The differential diagnosis for rapidly enlarging scalp tumors can be broad and includes both benign and malignant processes. In this patient, the differential diagnoses included trichilemmal carcinoma, cutaneous squamous cell carcinoma (SCC), sebaceous carcinoma (SC), proliferating trichilemmal tumor (PTT), and MPTT. Due to the notable clinical and histologic overlap among these lesions, definitive diagnosis required histopathologic evaluation in our patient.
Proliferating trichilemmal tumors were first described in 1966 by Wilson-Jones,1 who used the term proliferating epidermoid cysts, noting their distinct histologic features and resemblance to SCC.2 These tumors generally are benign and arise from the isthmus of the outer root sheath of the hair follicle; however, malignant transformation can occur, resulting in a rare entity known as MPTT. This malignant variant was first described in 1983 by Saida et al,3 who emphasized its distinct clinical behavior, including infiltrative growth, high mitotic activity, and potential for local recurrence and metastasis.
A recent literature review identified 60 reported cases of MPTT, with an average patient age of 57 years and a female predominance.4 Clinically, MPTTs often manifest as large (>5 cm) lobulated masses located on sun-exposed, hair-bearing areas of the skin, especially the scalp. These lesions may be flesh-colored to pink and often exhibit ulceration, necrosis, or calcification.5 Typically, MPTTs follow a biphasic course, beginning with a slow-growing phase followed by a period of rapid growth. Due to their aggressive behavior and resemblance to other cutaneous malignancies, accurate differentiation of MPTT from benign PTTs, cutaneous SCCs, SCs, and trichilemmal carcinomas is critical.
Malignant proliferating trichilemmal tumors demonstrate a substantially higher metastatic potential than either benign PTTs or cutaneous SCCs. While cutaneous SCCs carry a metastasis rate of approximately 1.9% to 2.6%, MPTTs carry a considerably higher rate of approximately 25.0%.6 Regional lymphatic spread is the most common route of dissemination, making comprehensive lymph node assessment—both radiographic and clinical—an important component of tumor staging. When lymph node involvement is suspected, surgical dissection may be indicated, along with consideration of adjuvant therapies.
Histopathologically, MPTT is characterized by nuclear atypia, mitotic figures, and lobulated masses of proliferating epithelium showing trichilemmal differentiation and infiltrative growth.4 The presence of CD34 positivity, reflecting outer follicular root sheath differentiation, helps distinguish MPTT from cutaneous SCC and SC, which typically lack this marker.6,7 Immunohistochemistry is therefore a valuable adjunct in differentiating these lesions.
The mainstay of treatment for MPTT is wide local excision with clear margins. Margins of at least 1 cm generally are recommended. Although Mohs micrographic surgery may be used in anatomically sensitive areas, it typically is not preferred due to the potential for skip lesions in MPTT, which may lead to incomplete excision and recurrence.8 In cases with evidence of regional or distant metastasis or when clear margins cannot be achieved confidently, adjunctive treatments such as radiation therapy and systemic chemotherapy may be indicated. Preoperative imaging is used to evaluate for local invasion (skull or bone involvement) and regional lymph node status, which may inform adjuvant therapy postoperatively.
The prognosis for MPTT is variable and depends largely on early recognition, accurate histopathologic diagnosis, complete surgical excision with clear margins, and the presence or absence of metastasis. When the tumor is fully excised with negative margins and no lymph node involvement, the risk for recurrence is substantially reduced; however, MPTT is known for its potential aggressive behavior. Delays in diagnosis or incomplete resection can lead to local recurrence, regional spread, or even distant metastasis. In the literature review discussed previously, the mortality rate of patients with MPTT was 11.7%,4 which is notably higher than that of more common cutaneous malignancies such as cutaneous SCC, which is reported at 1.2%.9
The clinical course of MPTT remains difficult to predict due to its rarity and the limited availability of large-scale studies. Most published data are derived from isolated case reports or small case series, making standardized treatment guidelines challenging. Given this uncertainty, long-term follow-up is strongly recommended to monitor for recurrence or metastatic progression.2
This case highlights the critical role of clinicopathologic correlation in the evaluation of atypical or rapidly growing scalp lesions. The expertise of dermatologists in recognizing atypical presentations, combined with precise histopathologic analysis, including immunohistochemical staining, is vital to ensuring accurate diagnosis and optimal treatment. Early intervention can improve patient outcomes by reducing the risk for local recurrence and metastatic progression as well as the need for more intensive therapies.
- Jones EW. Proliferating epidermoid cysts. Arch Dermatol. 1966;94:11-19.
- Kemaloglu CA, Öztürk M, Aydın B, et al. Malignant proliferating trichilemmal tumor of the scalp: report of 4 cases and a short review of the literature. Case Reports Plast Surg Hand Surg. 2022;9:158-164. doi:10.1080/23320885.2022.2077208
- Saida T, Oohard K, Hori Y, et al. Development of a malignant proliferating trichilemmal cyst in a patient with multiple trichilemmal cysts. Dermatology. 1983;166:203-208. doi:10.1159/000249868
- Abdelhammed MH, Siatecka H, Diwan AH, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology (Basel). 2024;11:354-363. doi:10.3390 /dermatopathology11040038
- Fronek L, Brahs A, Farsi M, et al. A rare case of trichilemmal carcinoma: histology and management. J Clin Aesthet Dermatol. 2021;14:25-30.
- Osto M, Parry N, Rehman R, et al. Malignant proliferating trichilemmal tumor of the scalp: a systematic review. Am J Dermatopathol. 2021;43:851-866. doi:10.1097/DAD.0000000000001991
- Plaza JA, Mackinnon A, Carrillo L, et al. Role of immunohistochemistry in the diagnosis of sebaceous carcinoma: a clinicopathologic and immunohistochemical study. Am J Dermatopathol. 2015;37:809-821. doi:10.1097/DAD.0000000000000255
- Singh P, Usman A, Motta L, et al. Malignant proliferating trichilemmal tumour. BMJ Case Rep. Published online August 17, 2018. doi:10.1136/bcr-2018-224460
- Ran NA, Granger EE, Brodland DG, et al. Risk factor number and recurrence, metastasis, and disease-related death in cutaneous squamous cell carcinoma. JAMA Dermatol. 2025;161:597-604. doi:10.1001/jamadermatol.2025.0128
- Jones EW. Proliferating epidermoid cysts. Arch Dermatol. 1966;94:11-19.
- Kemaloglu CA, Öztürk M, Aydın B, et al. Malignant proliferating trichilemmal tumor of the scalp: report of 4 cases and a short review of the literature. Case Reports Plast Surg Hand Surg. 2022;9:158-164. doi:10.1080/23320885.2022.2077208
- Saida T, Oohard K, Hori Y, et al. Development of a malignant proliferating trichilemmal cyst in a patient with multiple trichilemmal cysts. Dermatology. 1983;166:203-208. doi:10.1159/000249868
- Abdelhammed MH, Siatecka H, Diwan AH, et al. A rare case of a malignant proliferating trichilemmal tumor: a molecular study harboring potential therapeutic significance and a review of literature. Dermatopathology (Basel). 2024;11:354-363. doi:10.3390 /dermatopathology11040038
- Fronek L, Brahs A, Farsi M, et al. A rare case of trichilemmal carcinoma: histology and management. J Clin Aesthet Dermatol. 2021;14:25-30.
- Osto M, Parry N, Rehman R, et al. Malignant proliferating trichilemmal tumor of the scalp: a systematic review. Am J Dermatopathol. 2021;43:851-866. doi:10.1097/DAD.0000000000001991
- Plaza JA, Mackinnon A, Carrillo L, et al. Role of immunohistochemistry in the diagnosis of sebaceous carcinoma: a clinicopathologic and immunohistochemical study. Am J Dermatopathol. 2015;37:809-821. doi:10.1097/DAD.0000000000000255
- Singh P, Usman A, Motta L, et al. Malignant proliferating trichilemmal tumour. BMJ Case Rep. Published online August 17, 2018. doi:10.1136/bcr-2018-224460
- Ran NA, Granger EE, Brodland DG, et al. Risk factor number and recurrence, metastasis, and disease-related death in cutaneous squamous cell carcinoma. JAMA Dermatol. 2025;161:597-604. doi:10.1001/jamadermatol.2025.0128
Enlarging Mass on the Scalp
Enlarging Mass on the Scalp
A 61-year-old woman presented to the emergency department with worsening pain and bleeding from a scalp tumor of 16 years’ duration. Initially noted as a small nodule on the left parietal scalp on computed tomography of the head, the mass had grown rapidly in recent years and currently measured 22×10×15 cm. At prior consultations with plastic and general surgery, the patient had declined surgical intervention. At the current presentation, biopsies were performed by plastic surgery, and a dermatopathology consultation was ordered. Histopathology revealed atypical keratinocytes, nuclear pleomorphism, lobulating epithelial masses with trichilemmal keratinization, and CD34 positivity. Subsequent computed tomography and positron emission tomography of the head showed occipital skull erosion and bilateral cervical lymphadenopathy, suggesting metastasis.
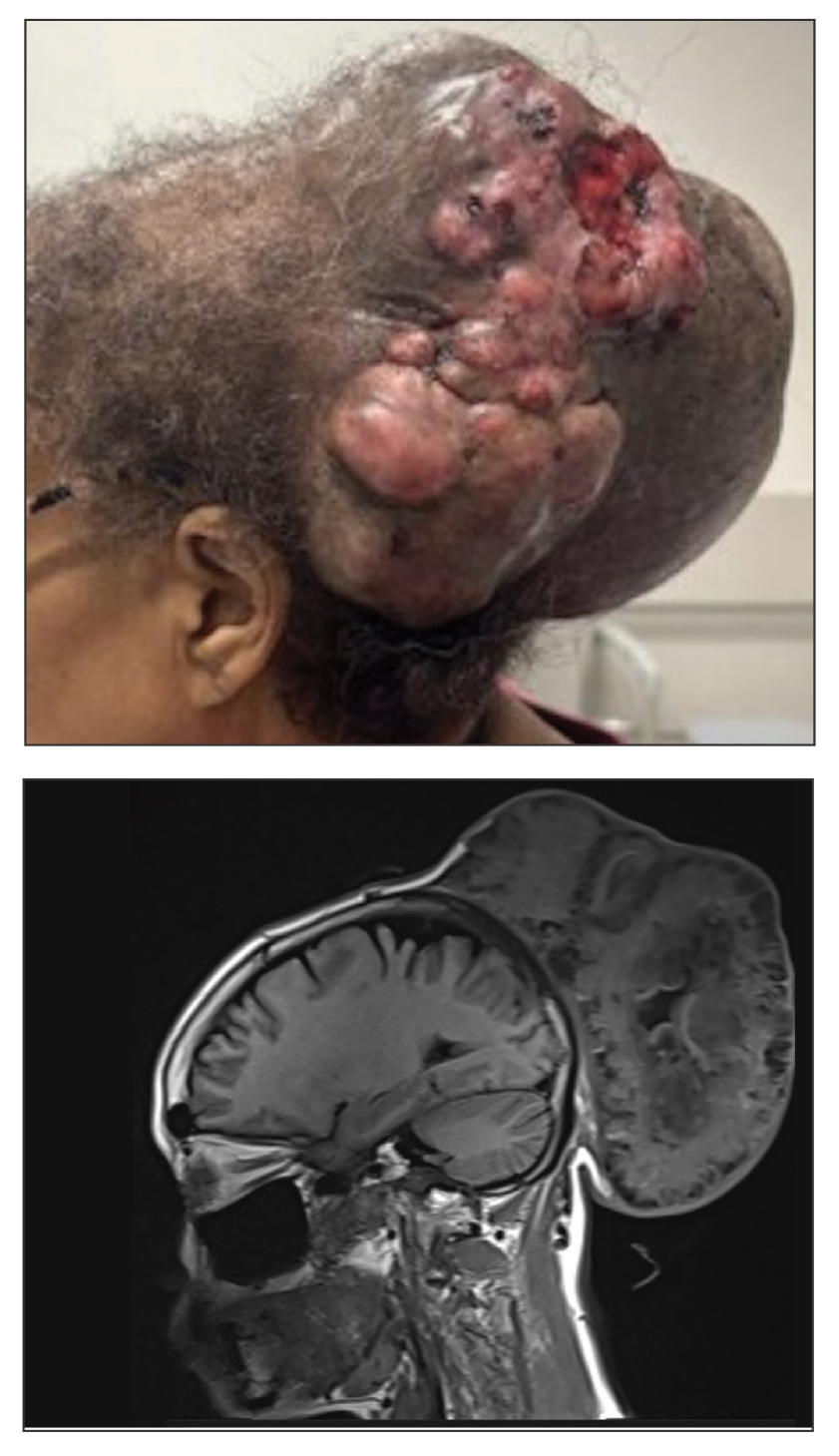
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)
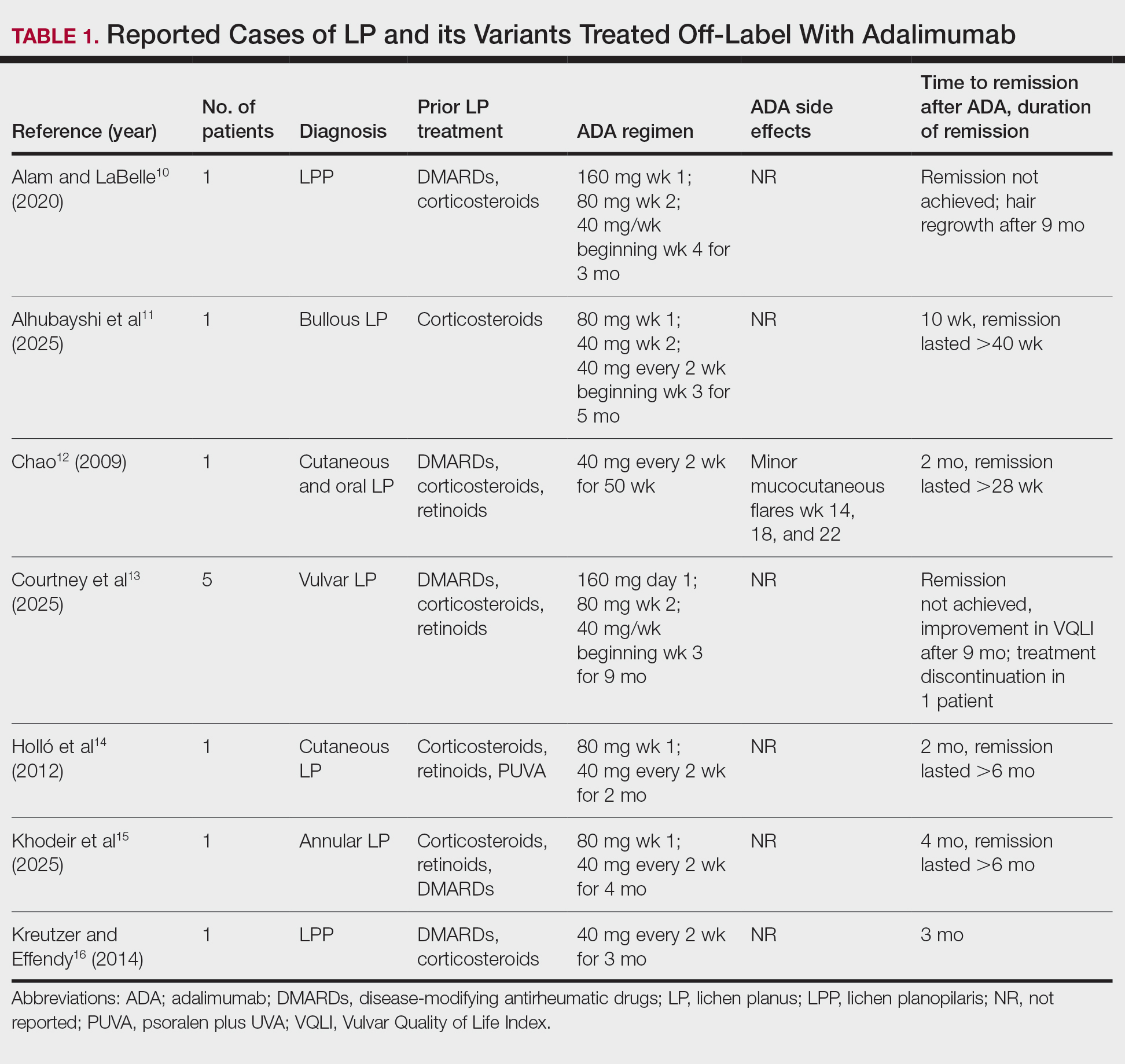
Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22
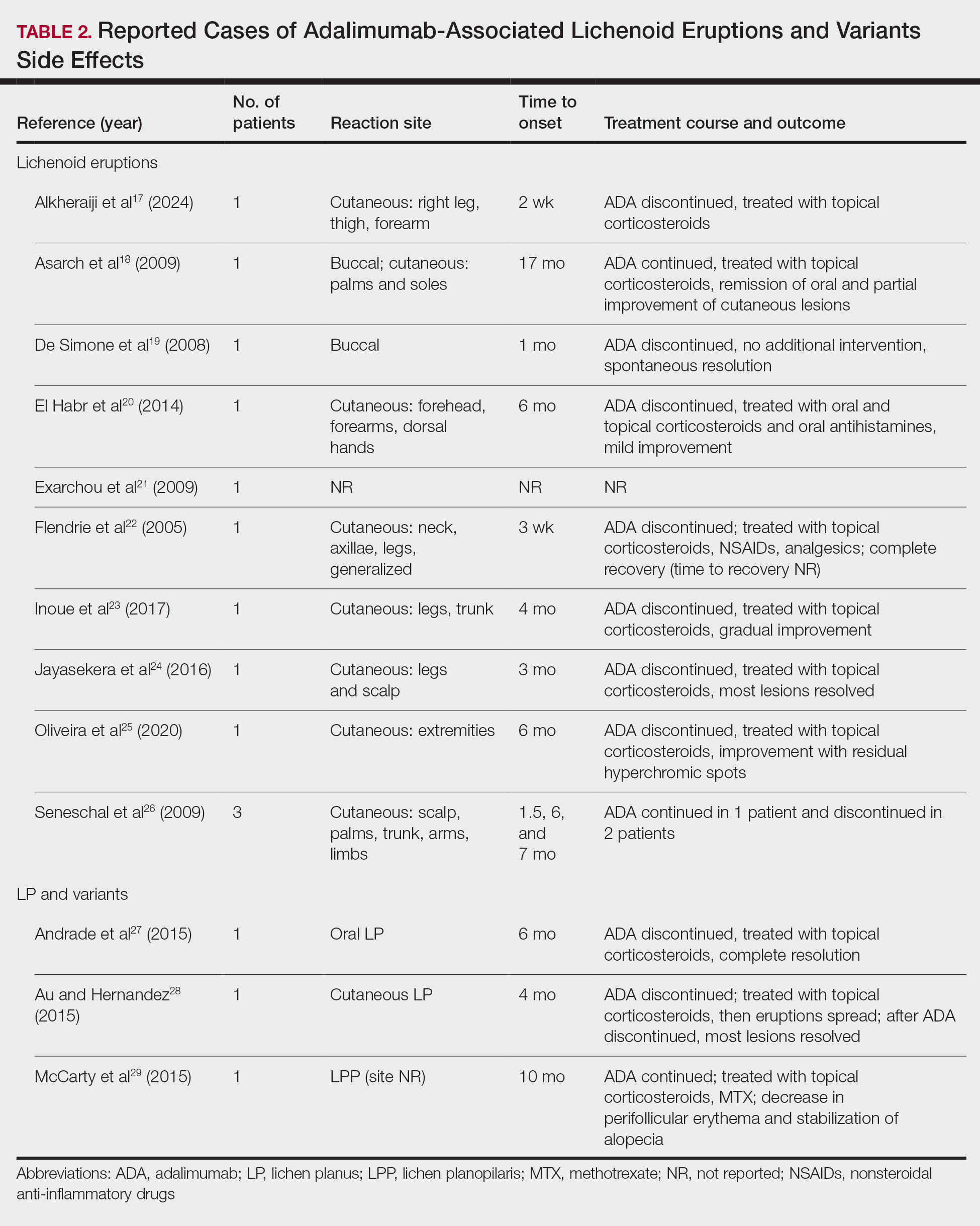
In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Practice Points
- Adalimumab can be beneficial when used off label for treatment of lichen planus in patients who do not respond to conventional therapies, including corticosteroids and immunosuppressants.
- Clinicians should be aware that adalimumab could potentially lead to paradoxical lichenoid eruptions and should monitor patients closely during treatment.
Metastatic Primary Extramammary Paget Disease: A Case Series
Metastatic Primary Extramammary Paget Disease: A Case Series
Extramammary Paget disease (EMPD) is a rare cutaneous malignancy typically seen in apocrine-rich areas, including the axillae and anogenital region. It presents as a slow-growing, erythematous patch or plaque that commonly is misdiagnosed as an infectious or inflammatory condition.1,2 Primary EMPD occurs as a intraepithelial neoplasm, whereas secondary EMPD occurs due to epidermotropic metastases or direct extension of an underlying adenocarcinoma into the skin.1 Most commonly, primary EMPD occurs in situ; however, when present, dermal invasion and metastases from the skin are associated with poorer outcomes.3 Given the rarity of metastatic disease, existing literature is limited to case reports and case series.
We present 2 patients with metastatic primary EMPD who had evidence of invasion on initial biopsy and died secondary to metastatic EMPD. We conducted a comprehensive review of the literature for invasive and metastatic EMPD to highlight key clinicopathologic features, treatment considerations, and the potential for rapid disease progression in cases of invasive EMPD.
Case Series
Patient 1—A 68-year-old White man with a history of breast cancer (in remission) presented to our clinic for further management of biopsy-proven scrotal EMPD. Prior to biopsy, he described a 6-month history of worsening scrotal rash treated with topical antifungals, oral antibiotics, and topical steroids due to presumed diagnosis of intertrigo, cellulitis, and dermatitis, respectively. Clinical examination showed indurated, erythematous, ulcerated plaques involving the bilateral groin, genitalia, and perineum (Figure 1). Skin biopsy confirmed a diagnosis of EMPD with both dermal and lymphovascular invasion. An immunohistochemical profile was positive for CK7 and carcinoembryonic antigen (CEA) and negative for CK20 (Figure 2).
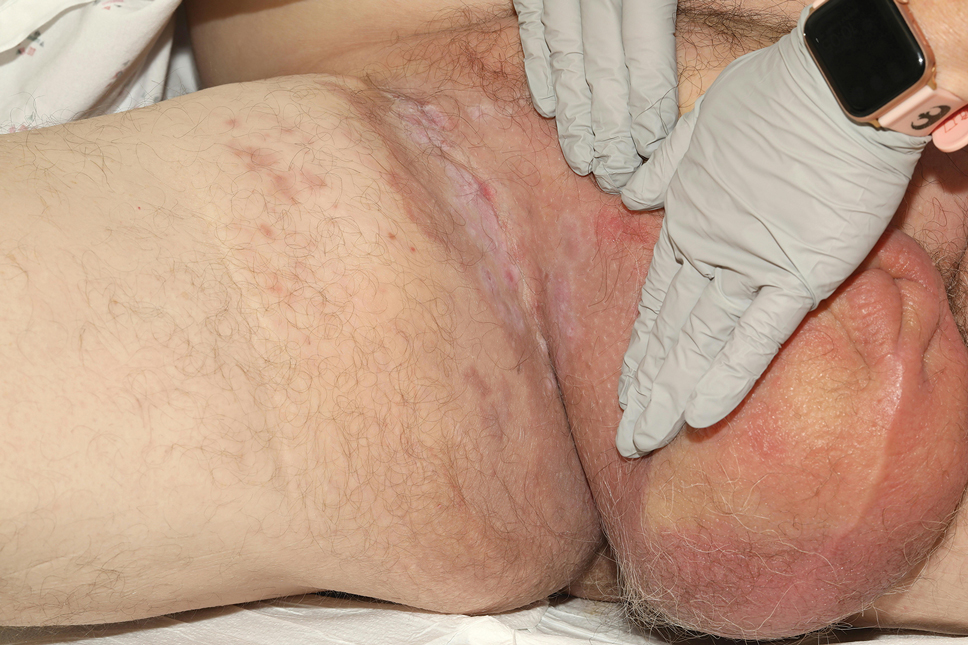
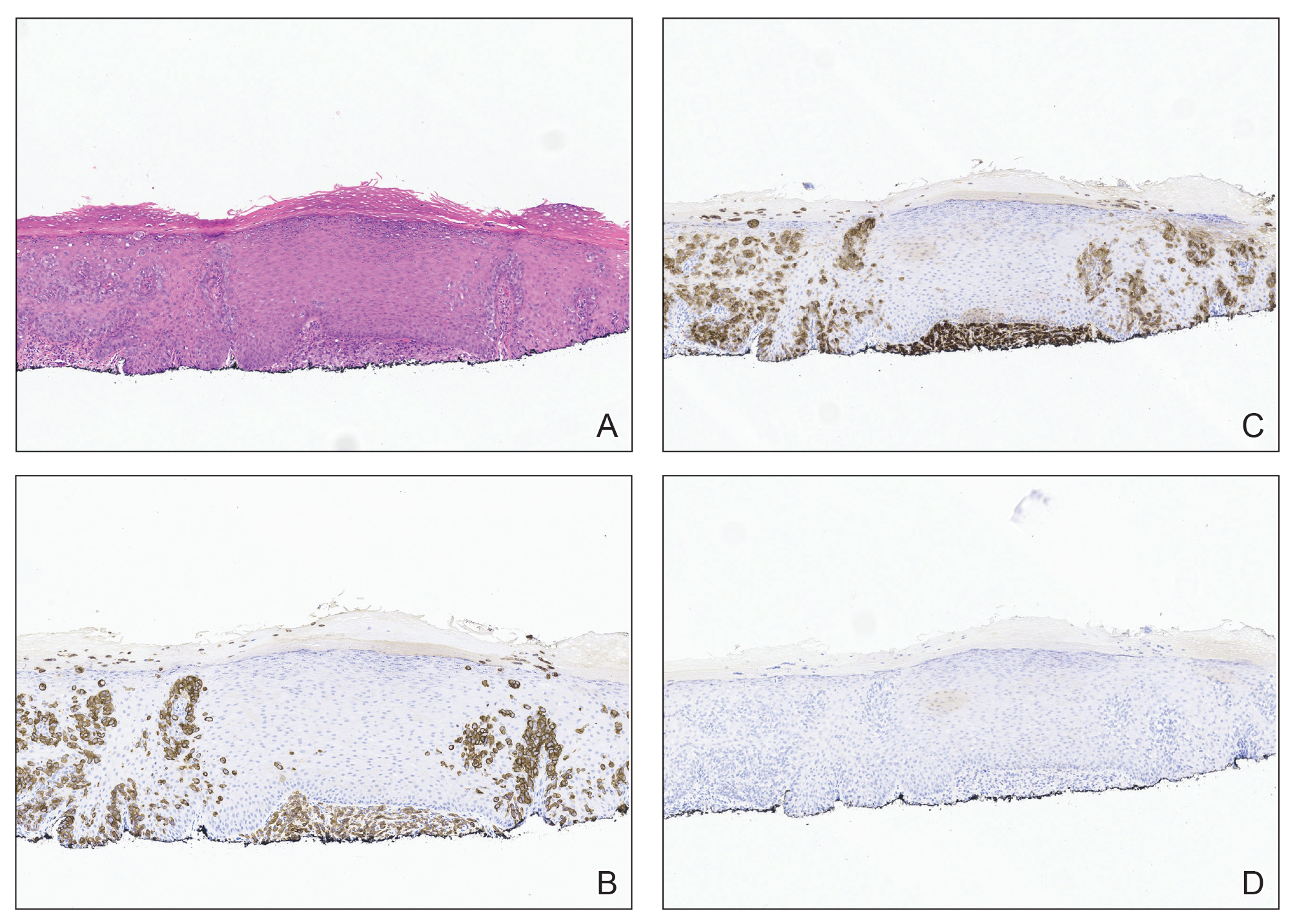
At presentation, the patient had palpable lymphadenopathy and scrotal edema concerning for inguinal and iliac lymph node metastases. Workup for an underlying adenocarcinoma included computed tomography (CT) of the chest, abdomen, and pelvis; urologic consultation with cystoscopy; and a screening colonoscopy. The CT scan revealed multiple enlarged inguinal and external iliac lymph nodes. Fine-needle aspiration revealed CK7- and CEA-positive neoplastic cells consistent with metastatic EMPD. The patient was treated with 6 cycles of carboplatin-paclitaxel, palliative radiation therapy, and pembrolizumab with minimal response to treatment and development of osteolytic vertebral lesions concerning for disease progression. He died 1 year after the initial diagnosis secondary to the disease.
Patient 2—A 79-year-old White man presented for further management of an outside diagnosis of superficially invasive primary EMPD of the bilateral inguinal folds and scrotum that had been present for 5 months prior to biopsy and diagnosis. Clinical examination at initial presentation revealed erythematous patches of the bilateral inguinal folds and scrotum, as well as an erythematous scaling plaque in the right axilla. There was no palpable clinical lymphadenopathy. Biopsy of the axilla and groin were both consistent with invasive EMPD with positive staining for CK7 and negative staining for CK20 and CDX2. Workup for underlying adenocarcinoma with whole-body positron emission tomography/CT, mammography, esophagogastroduodenoscopy, serum CEA, colonoscopy, and cystoscopy were all negative for a metastatic adenocarcinoma. There was no imaging or clinical evidence of lymphadenopathy. Complete circumferential peripheral and deep-margin assessment was performed in a staged manner on both sites, and negative margins were obtained.
Surveillance imaging 6 months after surgery revealed suspicious hepatic lesions. Fine-needle aspiration of the hepatic lesions demonstrated positive staining for CK7 and negative staining for CK20, CDX2, prostate-specific antigen, and thyroid transcription factor 1, consistent with metastatic EMPD. Oncology recommended carboplatin and docetaxel or docetaxel monotherapy chemotherapy. The patient was further managed by an outside oncologist due to ease of travel but died secondary to the disease 15 months following the initial diagnosis.
Comment
Extramammary Paget disease is an uncommon cutaneous malignancy that manifests as pruritic erythematous plaques within apocrine-rich areas such as the genitalia, axillae, or anal region. It most commonly occurs in patients older than 65 years, with White women and Asian men being affected at disproportionately higher rates.1,4 Delay in diagnosis is common, as EMPD can mimic other benign inflammatory or infectious conditions, including contact dermatitis, seborrheic dermatitis, tinea, candidiasis, and eczema.1
Metastatic and multifocal cases of primary EMPD are especially rare. According to a search of PubMed articles indexed for MEDLINE published through December 2023 using the terms extramammary Paget disease, EMPD, neoplasm metastasis, invasive extramammary, and neoplasm invasiveness, we identified 5040 cases of invasive EMPD and 477 cases of metastatic EMPD.5-37 Of the reports that disclosed patient demographic information, 3627 patients were female 1410 were male, and the mean age was 67 years. Sites of metastases included regional lymph nodes, liver, lungs, cervix, bladder, bone, brain, skin, kidney, and adrenal glands
Workup for EMPD—The initial steps for workup of EMPD include a thorough physical examination and lymph node assessment. A skin biopsy also should be performed for patients presenting with refractory, pruritic, and eczematous rashes in apocrine-rich areas to evaluate for EMPD.1 Characterization of large and complex tumors is better achieved through multiple biopsies with particular focus on nodular or thickened areas, as these may indicate invasive disease.2 Primary EMPD is characterized by pagetoid cells with abundant pale cytoplasm proliferating in a single-cell or nested pattern within the epidermis or dermis in invasive disease and often is accompanied by dermal lymphocytic inflammation.1 Immunohistochemistry demonstrates positive staining for CEA, CK7, and CK8, with negative staining for indicators of secondary EMPD including CK20 and CDX2.1,2
As part of the workup, it is critical to distinguish between primary disease and secondary EMPD.1 Beyond skin and clinical lymph node examination, additional workup should be based on age-appropriate and location-directed malignant neoplasm screenings, including colonoscopy, cystoscopy, prostate examination, mammography, and Papanicolaou test. Advanced imaging such as CT, positron emission tomography, or magnetic resonance imaging can be used to assess for metastatic disease if internal malignant neoplasms are present on initial screening or clinical lymphadenopathy is identified.2 Additionally, it can be helpful in the evaluation for nodal disease in cases of invasive EMPD.
The likelihood of associated underlying carcinomas varies depending on the site of involvement.38,39 For example, vulvar involvement constitutes approximately 65% of EMPD cases, with 11% to 20% of cases being associated with underlying gastrointestinal or genitourinary carcinomas. Involvement of the male genitalia, as in our 2 patients, is rare, accounting for approximately 14% of cases, 11% of which are associated with prostate, testicular, and bladder carcinoma. Perianal involvement comprises 20% of EMPD cases and has the greatest risk for underlying malignancy with an incidence of 33% to 86%, the majority of which are rectal or tubo-ovarian cancers.38,39 Consideration of the frequency and types of underlying carcinoma of respective sites of involvement can be helpful when ruling out secondary EMPD.
In both of our patients, palpable lymphadenopathy at the time of original diagnosis and histologic invasive disease on initial biopsy warranted thorough imaging and laboratory workup; there was no evidence of primary malignancy. Given the absence of an underlying carcinoma, both patients were classified as having metastatic primary EMPD.
Assessment of lymphadenopathy is an essential aspect of disease workup, as it is associated with a statistically higher rate of lymph node metastases. A study by Fujisawa et al20 demonstrated that 80% of patients with lymphadenopathy had regional metastases compared to only 15% of patients without clinical lymphadenopathy. The presence of invasive disease also has been shown to correspond with lymph node metastases.40 Ogata et al40 showed that 0% of cases with in situ EMPD had a positive sentinel lymph node biopsy (SLNB) compared to 4% and 43% in cases that showed evidence of microinvasion and dermal invasion, respectively. Lymph node metastases are associated with poor prognosis, with increasingly worse prognosis when there are multiple lymph nodes affected.41 In our case series, patient 1 had lymphadenopathy and both patients had invasive EMPD; they both later developed metastases and died.
Lymphadenopathy should be further investigated with imaging and biopsy or fine-needle aspiration.42 Recent expert consensus guidelines recommended this method of investigation over routine use of SLNB, as there is no evidence that a positive SLNB affects treatment that changes disease-specific survival.2
Treatment of EMPD—Surgical excision of the primary lesion is the first-line treatment of EMPD,1,2 which can be performed by wide local excision; however, studies have demonstrated higher recurrence-free survival with margin-controlled surgery (complete circumferential peripheral and deep margin assessment) or Mohs micrographic surgery (MMS), especially with CK7 immunostaining.2,37,43 The literature on MMS of invasive EMPD is sparse, accounting for 57 patients.25,37,44 Other reports describe management with surgical excision, wide local excision, regional resection, or vulvectomy, in addition to lymph node dissection, radiation therapy (RT), and/or chemotherapy.1-36,39,43-46 Despite the improved outcomes with MMS, the predominance of other surgical approaches in our search suggests that MMS may be currently underutilized for the treatment of invasive or locally advanced EMPD.
Among patients with unresectable disease or distant metastases, management includes RT with curative intent, chemotherapy, or a combination of both.1,2 In our review, 267 cases were treated using RT and 77 with chemotherapy. Radiation therapy is an effective therapeutic option with a reported response rate of 62% to 100% and can be employed as either primary or adjuvant treatment.3 For patients with lymph node metastasis the combination of RT and lymph node dissection has been shown to have improved outcomes compared to lymph node dissection alone, with 1 study showing a 5-year survival of 75% for patients who received adjuvant RT compared to 0% for lymph node dissection alone.45
There are currently no consensus guidelines on the best chemotherapeutic regimen for metastatic EMPD. Several regimens have been reported, including docetaxel monotherapy; low-dose 5-fluorouracil and cisplatin; combination chemotherapy FECOM (5-fluorouracil, epirubicin, carboplatin, vincristine, mitomycin); or combination therapy with cisplatin, epirubicin, and paclitaxel.1
Prognosis of Metastatic EMPD—Because invasive and metastatic EMPD is rare, its natural history is hard to predict. Poor prognosis is associated with nodule formation, tumor thickness, perianal or vaginal involvement, lymphovascular invasion, nodal metastasis, and distant metastasis. The 5-year survival for metastatic EMPD has been reported to be less than 10%.46 Our cases underscore the poor prognostic risk associated with metastatic EMPD.
For all cases of EMPD, close follow-up is warranted. Guidelines recommend physical examination with lymph node assessment every 3 to 6 months for 3 years and every 6 to 12 months for the subsequent 5 years.2 Specific recommendations for follow-up in invasive disease have not yet been described, though the 20% probability of developing an internal malignancy within 5 years after diagnosis and poor prognostic outcomes associated with invasive and metastatic disease support the need for close monitoring.2
Conclusion
Although in situ EMPD often is a slow-growing tumor with good prognosis, invasive disease has high potential to behave aggressively with high morbidity and mortality. Increased awareness and prompt identification of invasive EMPD, expedited comprehensive workup, and early multidisciplinary management might impact patient outcomes.
Acknowledgment—The authors would like to thank Ellen Aaronson, MLIS, AHIP (Mayo Clinic Libraries [Jacksonville, FL]), for creating and conducting the narrative literature search in the MEDLINE database.
- Hashimoto H, Ito T. Current management and treatment of extramammary Paget’s disease. Curr Treat Options Oncol. 2022;23:818-830. doi:10.1007/s11864-021-00923-3
- Kibbi N, Owen JL, Worley B, et al. Evidence-based clinical practice guidelines for extramammary Paget disease. JAMA Oncol. 2022;8:618-628. doi:10.1001/jamaoncol.2021.7148
- Morris CR, Hurst EA. Extramammary Paget’s disease: a review of the literature part II: treatment and prognosis. Dermatol Surg. 2020;46:305-311. doi:10.1097/DSS.0000000000002240
- Merritt BG, Degesys CA, Brodland DG. Extramammary Paget disease. Dermatol Clin. 2019;37:261-267. doi:10.1016/j.det.2019.02.002
- Aroche Gutierrez LL, Holloway SB, Donthi D, et al. Docetaxel treatment for widely metastatic invasive vulvar extramammary Paget’s disease with multifocal bone metastasis. Gynecol Oncol Rep. 2022;45:101114. doi:10.1016/j.gore.2022.101114
- Ueda M, Omori M, Sakai A. Invasive extramammary Paget’s disease with lymph node metastases and high-grade B-cell lymphoma. An Bras Dermatol. 2023;98:414-418. doi:10.1016/j.abd.2022.04.012
- Rathore R, Yadav D, Agarwal S, et al. Primary extra mammary Paget’s disease of vulva, with apocrine adenocarcinoma, signet ring cell differentiation and distant metastasis. J Family Reprod Health. 2020;14:276-280. doi:10.18502/jfrh.v14i4.5213
- Kawahara Y, Umeda Y, Yamaguchi B, et al. Long-term resolution of invasive extramammary Paget’s disease with multiple regional lymph node metastases solely with regional lymph node dissection. J Dermatol. 2021;48:E452-E453. doi:10.1111/1346-8138.16007
- Hanyu T, Fujitani S, Ito A, et al. Brain metastasis from extramammary Paget’s disease. Nagoya J Med Sci. 2020;82:791-798. doi:10.18999/nagjms.82.4.791
- Waki Y, Nobeyama Y, Ogawa T, et al. Case of extramammary Paget’s disease causing pulmonary tumor embolism. J Dermatol. 2020;47:E133-E134. doi:10.1111/1346-8138.15267
- Li ZG, Qin XJ. Extensive invasive extramammary Paget disease evaluated by F-18 FDG PET/CT: a case report. Medicine (Baltimore). 2015;94:E371. doi:10.1097/MD.0000000000000371
- Kato N, Matsue K, Sotodate A, et al. Extramammary Paget’s disease with distant skin metastasis. J Dermatol. 1996;23:408-414. doi:10.1111/j.1346-8138.1996.tb04043.x
- Hosomi M, Miyake O, Matsumiya K, et al. Extramammary Paget’s disease with a large mass in male genitalia: a case report. Article in Japanese. Hinyokika Kiyo. 1989;35:1981-1984.
- Hardy LE, Baxter L, Wan K, et al. Invasive cervical adenocarcinoma arising from extension of recurrent vulval Paget’s disease. BMJ Case Rep. 2020;13e232424. doi:10.1136/bcr-2019-232424
- Onaiwu CO, Ramirez PT, Kamat A, et al. Invasive extramammary Paget’s disease of the bladder diagnosed 18 years after noninvasive extramammary Paget’s disease of the vulva. Gynecol Oncol Case Rep. 2014;8:27-29. doi:10.1016/j.gynor.2014.03.004
- Yao H, Xie M, Fu S, et al. Survival analysis of patients with invasive extramammary Paget disease: implications of anatomic sites. BMC Cancer. 2018;18:403. doi:10.1186/s12885-018-4257-1
- Kato H, Watanabe S, Kariya K, et al. Efficacy of low-dose 5-fluorouracil/cisplatin therapy for invasive extramammary Paget’s disease. J Dermatol. 2018;45:560-563. doi:10.1111/1346-8138.14247
- Yoshino K, Fujisawa Y, Kiyohara Y, et al. Usefulness of docetaxel as first-line chemotherapy for metastatic extramammary Paget’s disease. J Dermatol. 2016;43:633-637. doi:10.1111/1346-8138.13200
- Shu B, Shen XX, Chen P, et al. Primary invasive extramammary Paget disease on penoscrotum: a clinicopathological analysis of 41 cases. Hum Pathol. 2016;47:70-77. doi:10.1016/j.humpath.2015.09.005References
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Dai B, Kong YY, Chang K, et al. Primary invasive carcinoma associated with penoscrotal extramammary Paget’s disease: a clinicopathological analysis of 56 cases. BJU Int. 2015;115:153-160. doi:10.1111/bju.12776
- Shiomi T, Noguchi T, Nakayama H, et al. Clinicopathological study of invasive extramammary Paget’s disease: subgroup comparison according to invasion depth. J Eur Acad Dermatol Venereol. 2013;27:589-592. doi:10.1111/j.1468-3083.2012.04489.x
- Hatta N, Morita R, Yamada M, et al. Sentinel lymph node biopsy in patients with extramammary Paget’s disease. Dermatol Surg. 2004;30:1329-1334. doi:10.1111/j.1524-4725.2004.30377.x
- Karam A, Dorigo O. Treatment outcomes in a large cohort of patients with invasive extramammary Paget’s disease. Gynecol Oncol. 2012;125:346-351. doi:10.1016/j.ygyno.2012.01.032
- Guo L, Liu X, Li H, et al. Clinicopathological features of extramammary Paget’s disease: a report of 75 cases. Article in Chinese. Zhonghua Yi Xue Za Zhi. 2015;95:1751-1754.
- Kilts TP, Long B, Glasgow AE, et al. Invasive vulvar extramammary Paget’s disease in the United States. Gynecol Oncol. 2020;157:649-655. doi:10.1016/j.ygyno.2020.03.018
- Kusatake K, Harada Y, Mizumoto K, et al. Usefulness of sentinel lymph node biopsy for the detection of metastasis in the early stage of extramammary Paget’s disease. Eur J Dermatol. 2015;25:156-161. doi:10.1684/ejd.2015.2534
- Jeong BK, Kim KR. Invasive extramammary Paget disease of the vulva with signet ring cell morphology in a patient with signet ring cell carcinoma of the stomach: report of a case. Int J Gynecol Pathol. 2018;37:147-151. doi:10.1097/PGP.0000000000000405
- Pagnanelli M, De Nardi P, Martella S, et al. Local excision of a mucinous adenocarcinoma of the anal margin (extramammary Paget’s disease) and reconstruction with a bilateral V-Y flap. Case Rep Surg. 2019;2019:9073982. doi:10.1155/2019/9073982
- Sopracordevole F, Di Giuseppe J, De Piero G, et al. Surgical treatment of Paget disease of the vulva: prognostic significance of stromal invasion and surgical margin status. J Low Genit Tract Dis. 2016;20:184-188. doi:10.1097/LGT.0000000000000191
- Evans AT, Neven P. Invasive adenocarcinoma arising in extramammary Paget’s disease of the vulva. Histopathology. 1991;18:355-360. doi:10.1111/j.1365-2559.1991.tb00857.x
- Kitano A, Izumi M, Tamura K, et al. Brain metastasis from cutaneous squamous cell carcinoma coexistent with extramammary Paget’s disease: a case report. Pathol Int. 2019;69:619-625. doi:10.1111/pin.12846
- Miracco C, Francini E, Torre P, et al. Extramammary invasive Paget’s disease and apocrine angiomatous hamartoma: an unusual association. Eur J Dermatol. 2018;28:853-855. doi:10.1684/ejd.2018.3438
- Kambayashi Y, Fujimura T, Ohuchi K, et al. Advanced invasive extramammary Paget’s disease concomitant with cecal cancer possessing rare variant of TP53 single nucleotide polymorphism. Case Rep Oncol. 2019;12:855-860. doi:10.1159/000504339
- Fujimura T, Furudate S, Kambayashi Y, et al. Potential use of bisphosphonates in invasive extramammary Paget’s disease: an immunohistochemical investigation. Clin Dev Immunol. 2013;2013:164982. doi:10.1155/2013/164982
- Kawamura H, Ogata K, Miura H, et al. Patellar metastases. A report of two cases. Int Orthop. 1993;17:57-59. doi:10.1007/BF00195227
- Damavandy AA, Terushkin V, Zitelli JA, et al. Intraoperative immunostaining for cytokeratin-7 during Mohs micrographic surgery demonstrates low local recurrence rates in extramammary Paget’s disease. Dermatol Surg. 2018;44:354-364. doi:10.1097/DSS.0000000000001355
- Morris CR, Hurst EA. Extramammary Paget disease: a review of the literature-part I: history, epidemiology, pathogenesis, presentation, histopathology, and diagnostic work-up. Dermatol Surg. 2020;46:151-158. doi:10.1097/DSS.0000000000002064
- Simonds RM, Segal RJ, Sharma A. Extramammary Paget’s disease: a review of the literature. Int J Dermatol. 2019;58:871-879. doi:10.1111/ijd.14328
- Ogata D, Kiyohara Y, Yoshikawa S, et al. Usefulness of sentinel lymph node biopsy for prognostic prediction in extramammary Paget’s disease. Eur J Dermatol. 2016;26:254-259. doi:10.1684/ejd.2016.2744
- Ohara K, Fujisawa Y, Yoshino K, et al. A proposal for a TNM staging system for extramammary Paget disease: retrospective analysis of 301 patients with invasive primary tumors. J Dermatol Sci. 2016;83:234-239. doi:10.1016/j.jdermsci.2016.06.004
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Kim SJ, Thompson AK, Zubair AS, et al. Surgical treatment and outcomes of patients with extramammary Paget disease: a cohort study. Dermatol Surg. 2017;43:708-714. doi:10.1097/DSS.0000000000001051
- Wollina U. Extensive invasive extramammary Paget’s disease: surgical treatment. J Cutan Aesthet Surg. 2013;6:41-44. doi:10.4103/0974-2077.110098
- Tsutsui K, Takahashi A, Muto Y, et al. Outcomes of lymph node dissection in the treatment of extramammary Paget’s disease: a single-institution study. J Dermatol. 2020;47:512-517. doi:10.1111/1346-8138.15285
- Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: a case report. Case Rep Oncol. 2021;14:430-438. doi:10.1159/000514345
Extramammary Paget disease (EMPD) is a rare cutaneous malignancy typically seen in apocrine-rich areas, including the axillae and anogenital region. It presents as a slow-growing, erythematous patch or plaque that commonly is misdiagnosed as an infectious or inflammatory condition.1,2 Primary EMPD occurs as a intraepithelial neoplasm, whereas secondary EMPD occurs due to epidermotropic metastases or direct extension of an underlying adenocarcinoma into the skin.1 Most commonly, primary EMPD occurs in situ; however, when present, dermal invasion and metastases from the skin are associated with poorer outcomes.3 Given the rarity of metastatic disease, existing literature is limited to case reports and case series.
We present 2 patients with metastatic primary EMPD who had evidence of invasion on initial biopsy and died secondary to metastatic EMPD. We conducted a comprehensive review of the literature for invasive and metastatic EMPD to highlight key clinicopathologic features, treatment considerations, and the potential for rapid disease progression in cases of invasive EMPD.
Case Series
Patient 1—A 68-year-old White man with a history of breast cancer (in remission) presented to our clinic for further management of biopsy-proven scrotal EMPD. Prior to biopsy, he described a 6-month history of worsening scrotal rash treated with topical antifungals, oral antibiotics, and topical steroids due to presumed diagnosis of intertrigo, cellulitis, and dermatitis, respectively. Clinical examination showed indurated, erythematous, ulcerated plaques involving the bilateral groin, genitalia, and perineum (Figure 1). Skin biopsy confirmed a diagnosis of EMPD with both dermal and lymphovascular invasion. An immunohistochemical profile was positive for CK7 and carcinoembryonic antigen (CEA) and negative for CK20 (Figure 2).


At presentation, the patient had palpable lymphadenopathy and scrotal edema concerning for inguinal and iliac lymph node metastases. Workup for an underlying adenocarcinoma included computed tomography (CT) of the chest, abdomen, and pelvis; urologic consultation with cystoscopy; and a screening colonoscopy. The CT scan revealed multiple enlarged inguinal and external iliac lymph nodes. Fine-needle aspiration revealed CK7- and CEA-positive neoplastic cells consistent with metastatic EMPD. The patient was treated with 6 cycles of carboplatin-paclitaxel, palliative radiation therapy, and pembrolizumab with minimal response to treatment and development of osteolytic vertebral lesions concerning for disease progression. He died 1 year after the initial diagnosis secondary to the disease.
Patient 2—A 79-year-old White man presented for further management of an outside diagnosis of superficially invasive primary EMPD of the bilateral inguinal folds and scrotum that had been present for 5 months prior to biopsy and diagnosis. Clinical examination at initial presentation revealed erythematous patches of the bilateral inguinal folds and scrotum, as well as an erythematous scaling plaque in the right axilla. There was no palpable clinical lymphadenopathy. Biopsy of the axilla and groin were both consistent with invasive EMPD with positive staining for CK7 and negative staining for CK20 and CDX2. Workup for underlying adenocarcinoma with whole-body positron emission tomography/CT, mammography, esophagogastroduodenoscopy, serum CEA, colonoscopy, and cystoscopy were all negative for a metastatic adenocarcinoma. There was no imaging or clinical evidence of lymphadenopathy. Complete circumferential peripheral and deep-margin assessment was performed in a staged manner on both sites, and negative margins were obtained.
Surveillance imaging 6 months after surgery revealed suspicious hepatic lesions. Fine-needle aspiration of the hepatic lesions demonstrated positive staining for CK7 and negative staining for CK20, CDX2, prostate-specific antigen, and thyroid transcription factor 1, consistent with metastatic EMPD. Oncology recommended carboplatin and docetaxel or docetaxel monotherapy chemotherapy. The patient was further managed by an outside oncologist due to ease of travel but died secondary to the disease 15 months following the initial diagnosis.
Comment
Extramammary Paget disease is an uncommon cutaneous malignancy that manifests as pruritic erythematous plaques within apocrine-rich areas such as the genitalia, axillae, or anal region. It most commonly occurs in patients older than 65 years, with White women and Asian men being affected at disproportionately higher rates.1,4 Delay in diagnosis is common, as EMPD can mimic other benign inflammatory or infectious conditions, including contact dermatitis, seborrheic dermatitis, tinea, candidiasis, and eczema.1
Metastatic and multifocal cases of primary EMPD are especially rare. According to a search of PubMed articles indexed for MEDLINE published through December 2023 using the terms extramammary Paget disease, EMPD, neoplasm metastasis, invasive extramammary, and neoplasm invasiveness, we identified 5040 cases of invasive EMPD and 477 cases of metastatic EMPD.5-37 Of the reports that disclosed patient demographic information, 3627 patients were female 1410 were male, and the mean age was 67 years. Sites of metastases included regional lymph nodes, liver, lungs, cervix, bladder, bone, brain, skin, kidney, and adrenal glands
Workup for EMPD—The initial steps for workup of EMPD include a thorough physical examination and lymph node assessment. A skin biopsy also should be performed for patients presenting with refractory, pruritic, and eczematous rashes in apocrine-rich areas to evaluate for EMPD.1 Characterization of large and complex tumors is better achieved through multiple biopsies with particular focus on nodular or thickened areas, as these may indicate invasive disease.2 Primary EMPD is characterized by pagetoid cells with abundant pale cytoplasm proliferating in a single-cell or nested pattern within the epidermis or dermis in invasive disease and often is accompanied by dermal lymphocytic inflammation.1 Immunohistochemistry demonstrates positive staining for CEA, CK7, and CK8, with negative staining for indicators of secondary EMPD including CK20 and CDX2.1,2
As part of the workup, it is critical to distinguish between primary disease and secondary EMPD.1 Beyond skin and clinical lymph node examination, additional workup should be based on age-appropriate and location-directed malignant neoplasm screenings, including colonoscopy, cystoscopy, prostate examination, mammography, and Papanicolaou test. Advanced imaging such as CT, positron emission tomography, or magnetic resonance imaging can be used to assess for metastatic disease if internal malignant neoplasms are present on initial screening or clinical lymphadenopathy is identified.2 Additionally, it can be helpful in the evaluation for nodal disease in cases of invasive EMPD.
The likelihood of associated underlying carcinomas varies depending on the site of involvement.38,39 For example, vulvar involvement constitutes approximately 65% of EMPD cases, with 11% to 20% of cases being associated with underlying gastrointestinal or genitourinary carcinomas. Involvement of the male genitalia, as in our 2 patients, is rare, accounting for approximately 14% of cases, 11% of which are associated with prostate, testicular, and bladder carcinoma. Perianal involvement comprises 20% of EMPD cases and has the greatest risk for underlying malignancy with an incidence of 33% to 86%, the majority of which are rectal or tubo-ovarian cancers.38,39 Consideration of the frequency and types of underlying carcinoma of respective sites of involvement can be helpful when ruling out secondary EMPD.
In both of our patients, palpable lymphadenopathy at the time of original diagnosis and histologic invasive disease on initial biopsy warranted thorough imaging and laboratory workup; there was no evidence of primary malignancy. Given the absence of an underlying carcinoma, both patients were classified as having metastatic primary EMPD.
Assessment of lymphadenopathy is an essential aspect of disease workup, as it is associated with a statistically higher rate of lymph node metastases. A study by Fujisawa et al20 demonstrated that 80% of patients with lymphadenopathy had regional metastases compared to only 15% of patients without clinical lymphadenopathy. The presence of invasive disease also has been shown to correspond with lymph node metastases.40 Ogata et al40 showed that 0% of cases with in situ EMPD had a positive sentinel lymph node biopsy (SLNB) compared to 4% and 43% in cases that showed evidence of microinvasion and dermal invasion, respectively. Lymph node metastases are associated with poor prognosis, with increasingly worse prognosis when there are multiple lymph nodes affected.41 In our case series, patient 1 had lymphadenopathy and both patients had invasive EMPD; they both later developed metastases and died.
Lymphadenopathy should be further investigated with imaging and biopsy or fine-needle aspiration.42 Recent expert consensus guidelines recommended this method of investigation over routine use of SLNB, as there is no evidence that a positive SLNB affects treatment that changes disease-specific survival.2
Treatment of EMPD—Surgical excision of the primary lesion is the first-line treatment of EMPD,1,2 which can be performed by wide local excision; however, studies have demonstrated higher recurrence-free survival with margin-controlled surgery (complete circumferential peripheral and deep margin assessment) or Mohs micrographic surgery (MMS), especially with CK7 immunostaining.2,37,43 The literature on MMS of invasive EMPD is sparse, accounting for 57 patients.25,37,44 Other reports describe management with surgical excision, wide local excision, regional resection, or vulvectomy, in addition to lymph node dissection, radiation therapy (RT), and/or chemotherapy.1-36,39,43-46 Despite the improved outcomes with MMS, the predominance of other surgical approaches in our search suggests that MMS may be currently underutilized for the treatment of invasive or locally advanced EMPD.
Among patients with unresectable disease or distant metastases, management includes RT with curative intent, chemotherapy, or a combination of both.1,2 In our review, 267 cases were treated using RT and 77 with chemotherapy. Radiation therapy is an effective therapeutic option with a reported response rate of 62% to 100% and can be employed as either primary or adjuvant treatment.3 For patients with lymph node metastasis the combination of RT and lymph node dissection has been shown to have improved outcomes compared to lymph node dissection alone, with 1 study showing a 5-year survival of 75% for patients who received adjuvant RT compared to 0% for lymph node dissection alone.45
There are currently no consensus guidelines on the best chemotherapeutic regimen for metastatic EMPD. Several regimens have been reported, including docetaxel monotherapy; low-dose 5-fluorouracil and cisplatin; combination chemotherapy FECOM (5-fluorouracil, epirubicin, carboplatin, vincristine, mitomycin); or combination therapy with cisplatin, epirubicin, and paclitaxel.1
Prognosis of Metastatic EMPD—Because invasive and metastatic EMPD is rare, its natural history is hard to predict. Poor prognosis is associated with nodule formation, tumor thickness, perianal or vaginal involvement, lymphovascular invasion, nodal metastasis, and distant metastasis. The 5-year survival for metastatic EMPD has been reported to be less than 10%.46 Our cases underscore the poor prognostic risk associated with metastatic EMPD.
For all cases of EMPD, close follow-up is warranted. Guidelines recommend physical examination with lymph node assessment every 3 to 6 months for 3 years and every 6 to 12 months for the subsequent 5 years.2 Specific recommendations for follow-up in invasive disease have not yet been described, though the 20% probability of developing an internal malignancy within 5 years after diagnosis and poor prognostic outcomes associated with invasive and metastatic disease support the need for close monitoring.2
Conclusion
Although in situ EMPD often is a slow-growing tumor with good prognosis, invasive disease has high potential to behave aggressively with high morbidity and mortality. Increased awareness and prompt identification of invasive EMPD, expedited comprehensive workup, and early multidisciplinary management might impact patient outcomes.
Acknowledgment—The authors would like to thank Ellen Aaronson, MLIS, AHIP (Mayo Clinic Libraries [Jacksonville, FL]), for creating and conducting the narrative literature search in the MEDLINE database.
Extramammary Paget disease (EMPD) is a rare cutaneous malignancy typically seen in apocrine-rich areas, including the axillae and anogenital region. It presents as a slow-growing, erythematous patch or plaque that commonly is misdiagnosed as an infectious or inflammatory condition.1,2 Primary EMPD occurs as a intraepithelial neoplasm, whereas secondary EMPD occurs due to epidermotropic metastases or direct extension of an underlying adenocarcinoma into the skin.1 Most commonly, primary EMPD occurs in situ; however, when present, dermal invasion and metastases from the skin are associated with poorer outcomes.3 Given the rarity of metastatic disease, existing literature is limited to case reports and case series.
We present 2 patients with metastatic primary EMPD who had evidence of invasion on initial biopsy and died secondary to metastatic EMPD. We conducted a comprehensive review of the literature for invasive and metastatic EMPD to highlight key clinicopathologic features, treatment considerations, and the potential for rapid disease progression in cases of invasive EMPD.
Case Series
Patient 1—A 68-year-old White man with a history of breast cancer (in remission) presented to our clinic for further management of biopsy-proven scrotal EMPD. Prior to biopsy, he described a 6-month history of worsening scrotal rash treated with topical antifungals, oral antibiotics, and topical steroids due to presumed diagnosis of intertrigo, cellulitis, and dermatitis, respectively. Clinical examination showed indurated, erythematous, ulcerated plaques involving the bilateral groin, genitalia, and perineum (Figure 1). Skin biopsy confirmed a diagnosis of EMPD with both dermal and lymphovascular invasion. An immunohistochemical profile was positive for CK7 and carcinoembryonic antigen (CEA) and negative for CK20 (Figure 2).


At presentation, the patient had palpable lymphadenopathy and scrotal edema concerning for inguinal and iliac lymph node metastases. Workup for an underlying adenocarcinoma included computed tomography (CT) of the chest, abdomen, and pelvis; urologic consultation with cystoscopy; and a screening colonoscopy. The CT scan revealed multiple enlarged inguinal and external iliac lymph nodes. Fine-needle aspiration revealed CK7- and CEA-positive neoplastic cells consistent with metastatic EMPD. The patient was treated with 6 cycles of carboplatin-paclitaxel, palliative radiation therapy, and pembrolizumab with minimal response to treatment and development of osteolytic vertebral lesions concerning for disease progression. He died 1 year after the initial diagnosis secondary to the disease.
Patient 2—A 79-year-old White man presented for further management of an outside diagnosis of superficially invasive primary EMPD of the bilateral inguinal folds and scrotum that had been present for 5 months prior to biopsy and diagnosis. Clinical examination at initial presentation revealed erythematous patches of the bilateral inguinal folds and scrotum, as well as an erythematous scaling plaque in the right axilla. There was no palpable clinical lymphadenopathy. Biopsy of the axilla and groin were both consistent with invasive EMPD with positive staining for CK7 and negative staining for CK20 and CDX2. Workup for underlying adenocarcinoma with whole-body positron emission tomography/CT, mammography, esophagogastroduodenoscopy, serum CEA, colonoscopy, and cystoscopy were all negative for a metastatic adenocarcinoma. There was no imaging or clinical evidence of lymphadenopathy. Complete circumferential peripheral and deep-margin assessment was performed in a staged manner on both sites, and negative margins were obtained.
Surveillance imaging 6 months after surgery revealed suspicious hepatic lesions. Fine-needle aspiration of the hepatic lesions demonstrated positive staining for CK7 and negative staining for CK20, CDX2, prostate-specific antigen, and thyroid transcription factor 1, consistent with metastatic EMPD. Oncology recommended carboplatin and docetaxel or docetaxel monotherapy chemotherapy. The patient was further managed by an outside oncologist due to ease of travel but died secondary to the disease 15 months following the initial diagnosis.
Comment
Extramammary Paget disease is an uncommon cutaneous malignancy that manifests as pruritic erythematous plaques within apocrine-rich areas such as the genitalia, axillae, or anal region. It most commonly occurs in patients older than 65 years, with White women and Asian men being affected at disproportionately higher rates.1,4 Delay in diagnosis is common, as EMPD can mimic other benign inflammatory or infectious conditions, including contact dermatitis, seborrheic dermatitis, tinea, candidiasis, and eczema.1
Metastatic and multifocal cases of primary EMPD are especially rare. According to a search of PubMed articles indexed for MEDLINE published through December 2023 using the terms extramammary Paget disease, EMPD, neoplasm metastasis, invasive extramammary, and neoplasm invasiveness, we identified 5040 cases of invasive EMPD and 477 cases of metastatic EMPD.5-37 Of the reports that disclosed patient demographic information, 3627 patients were female 1410 were male, and the mean age was 67 years. Sites of metastases included regional lymph nodes, liver, lungs, cervix, bladder, bone, brain, skin, kidney, and adrenal glands
Workup for EMPD—The initial steps for workup of EMPD include a thorough physical examination and lymph node assessment. A skin biopsy also should be performed for patients presenting with refractory, pruritic, and eczematous rashes in apocrine-rich areas to evaluate for EMPD.1 Characterization of large and complex tumors is better achieved through multiple biopsies with particular focus on nodular or thickened areas, as these may indicate invasive disease.2 Primary EMPD is characterized by pagetoid cells with abundant pale cytoplasm proliferating in a single-cell or nested pattern within the epidermis or dermis in invasive disease and often is accompanied by dermal lymphocytic inflammation.1 Immunohistochemistry demonstrates positive staining for CEA, CK7, and CK8, with negative staining for indicators of secondary EMPD including CK20 and CDX2.1,2
As part of the workup, it is critical to distinguish between primary disease and secondary EMPD.1 Beyond skin and clinical lymph node examination, additional workup should be based on age-appropriate and location-directed malignant neoplasm screenings, including colonoscopy, cystoscopy, prostate examination, mammography, and Papanicolaou test. Advanced imaging such as CT, positron emission tomography, or magnetic resonance imaging can be used to assess for metastatic disease if internal malignant neoplasms are present on initial screening or clinical lymphadenopathy is identified.2 Additionally, it can be helpful in the evaluation for nodal disease in cases of invasive EMPD.
The likelihood of associated underlying carcinomas varies depending on the site of involvement.38,39 For example, vulvar involvement constitutes approximately 65% of EMPD cases, with 11% to 20% of cases being associated with underlying gastrointestinal or genitourinary carcinomas. Involvement of the male genitalia, as in our 2 patients, is rare, accounting for approximately 14% of cases, 11% of which are associated with prostate, testicular, and bladder carcinoma. Perianal involvement comprises 20% of EMPD cases and has the greatest risk for underlying malignancy with an incidence of 33% to 86%, the majority of which are rectal or tubo-ovarian cancers.38,39 Consideration of the frequency and types of underlying carcinoma of respective sites of involvement can be helpful when ruling out secondary EMPD.
In both of our patients, palpable lymphadenopathy at the time of original diagnosis and histologic invasive disease on initial biopsy warranted thorough imaging and laboratory workup; there was no evidence of primary malignancy. Given the absence of an underlying carcinoma, both patients were classified as having metastatic primary EMPD.
Assessment of lymphadenopathy is an essential aspect of disease workup, as it is associated with a statistically higher rate of lymph node metastases. A study by Fujisawa et al20 demonstrated that 80% of patients with lymphadenopathy had regional metastases compared to only 15% of patients without clinical lymphadenopathy. The presence of invasive disease also has been shown to correspond with lymph node metastases.40 Ogata et al40 showed that 0% of cases with in situ EMPD had a positive sentinel lymph node biopsy (SLNB) compared to 4% and 43% in cases that showed evidence of microinvasion and dermal invasion, respectively. Lymph node metastases are associated with poor prognosis, with increasingly worse prognosis when there are multiple lymph nodes affected.41 In our case series, patient 1 had lymphadenopathy and both patients had invasive EMPD; they both later developed metastases and died.
Lymphadenopathy should be further investigated with imaging and biopsy or fine-needle aspiration.42 Recent expert consensus guidelines recommended this method of investigation over routine use of SLNB, as there is no evidence that a positive SLNB affects treatment that changes disease-specific survival.2
Treatment of EMPD—Surgical excision of the primary lesion is the first-line treatment of EMPD,1,2 which can be performed by wide local excision; however, studies have demonstrated higher recurrence-free survival with margin-controlled surgery (complete circumferential peripheral and deep margin assessment) or Mohs micrographic surgery (MMS), especially with CK7 immunostaining.2,37,43 The literature on MMS of invasive EMPD is sparse, accounting for 57 patients.25,37,44 Other reports describe management with surgical excision, wide local excision, regional resection, or vulvectomy, in addition to lymph node dissection, radiation therapy (RT), and/or chemotherapy.1-36,39,43-46 Despite the improved outcomes with MMS, the predominance of other surgical approaches in our search suggests that MMS may be currently underutilized for the treatment of invasive or locally advanced EMPD.
Among patients with unresectable disease or distant metastases, management includes RT with curative intent, chemotherapy, or a combination of both.1,2 In our review, 267 cases were treated using RT and 77 with chemotherapy. Radiation therapy is an effective therapeutic option with a reported response rate of 62% to 100% and can be employed as either primary or adjuvant treatment.3 For patients with lymph node metastasis the combination of RT and lymph node dissection has been shown to have improved outcomes compared to lymph node dissection alone, with 1 study showing a 5-year survival of 75% for patients who received adjuvant RT compared to 0% for lymph node dissection alone.45
There are currently no consensus guidelines on the best chemotherapeutic regimen for metastatic EMPD. Several regimens have been reported, including docetaxel monotherapy; low-dose 5-fluorouracil and cisplatin; combination chemotherapy FECOM (5-fluorouracil, epirubicin, carboplatin, vincristine, mitomycin); or combination therapy with cisplatin, epirubicin, and paclitaxel.1
Prognosis of Metastatic EMPD—Because invasive and metastatic EMPD is rare, its natural history is hard to predict. Poor prognosis is associated with nodule formation, tumor thickness, perianal or vaginal involvement, lymphovascular invasion, nodal metastasis, and distant metastasis. The 5-year survival for metastatic EMPD has been reported to be less than 10%.46 Our cases underscore the poor prognostic risk associated with metastatic EMPD.
For all cases of EMPD, close follow-up is warranted. Guidelines recommend physical examination with lymph node assessment every 3 to 6 months for 3 years and every 6 to 12 months for the subsequent 5 years.2 Specific recommendations for follow-up in invasive disease have not yet been described, though the 20% probability of developing an internal malignancy within 5 years after diagnosis and poor prognostic outcomes associated with invasive and metastatic disease support the need for close monitoring.2
Conclusion
Although in situ EMPD often is a slow-growing tumor with good prognosis, invasive disease has high potential to behave aggressively with high morbidity and mortality. Increased awareness and prompt identification of invasive EMPD, expedited comprehensive workup, and early multidisciplinary management might impact patient outcomes.
Acknowledgment—The authors would like to thank Ellen Aaronson, MLIS, AHIP (Mayo Clinic Libraries [Jacksonville, FL]), for creating and conducting the narrative literature search in the MEDLINE database.
- Hashimoto H, Ito T. Current management and treatment of extramammary Paget’s disease. Curr Treat Options Oncol. 2022;23:818-830. doi:10.1007/s11864-021-00923-3
- Kibbi N, Owen JL, Worley B, et al. Evidence-based clinical practice guidelines for extramammary Paget disease. JAMA Oncol. 2022;8:618-628. doi:10.1001/jamaoncol.2021.7148
- Morris CR, Hurst EA. Extramammary Paget’s disease: a review of the literature part II: treatment and prognosis. Dermatol Surg. 2020;46:305-311. doi:10.1097/DSS.0000000000002240
- Merritt BG, Degesys CA, Brodland DG. Extramammary Paget disease. Dermatol Clin. 2019;37:261-267. doi:10.1016/j.det.2019.02.002
- Aroche Gutierrez LL, Holloway SB, Donthi D, et al. Docetaxel treatment for widely metastatic invasive vulvar extramammary Paget’s disease with multifocal bone metastasis. Gynecol Oncol Rep. 2022;45:101114. doi:10.1016/j.gore.2022.101114
- Ueda M, Omori M, Sakai A. Invasive extramammary Paget’s disease with lymph node metastases and high-grade B-cell lymphoma. An Bras Dermatol. 2023;98:414-418. doi:10.1016/j.abd.2022.04.012
- Rathore R, Yadav D, Agarwal S, et al. Primary extra mammary Paget’s disease of vulva, with apocrine adenocarcinoma, signet ring cell differentiation and distant metastasis. J Family Reprod Health. 2020;14:276-280. doi:10.18502/jfrh.v14i4.5213
- Kawahara Y, Umeda Y, Yamaguchi B, et al. Long-term resolution of invasive extramammary Paget’s disease with multiple regional lymph node metastases solely with regional lymph node dissection. J Dermatol. 2021;48:E452-E453. doi:10.1111/1346-8138.16007
- Hanyu T, Fujitani S, Ito A, et al. Brain metastasis from extramammary Paget’s disease. Nagoya J Med Sci. 2020;82:791-798. doi:10.18999/nagjms.82.4.791
- Waki Y, Nobeyama Y, Ogawa T, et al. Case of extramammary Paget’s disease causing pulmonary tumor embolism. J Dermatol. 2020;47:E133-E134. doi:10.1111/1346-8138.15267
- Li ZG, Qin XJ. Extensive invasive extramammary Paget disease evaluated by F-18 FDG PET/CT: a case report. Medicine (Baltimore). 2015;94:E371. doi:10.1097/MD.0000000000000371
- Kato N, Matsue K, Sotodate A, et al. Extramammary Paget’s disease with distant skin metastasis. J Dermatol. 1996;23:408-414. doi:10.1111/j.1346-8138.1996.tb04043.x
- Hosomi M, Miyake O, Matsumiya K, et al. Extramammary Paget’s disease with a large mass in male genitalia: a case report. Article in Japanese. Hinyokika Kiyo. 1989;35:1981-1984.
- Hardy LE, Baxter L, Wan K, et al. Invasive cervical adenocarcinoma arising from extension of recurrent vulval Paget’s disease. BMJ Case Rep. 2020;13e232424. doi:10.1136/bcr-2019-232424
- Onaiwu CO, Ramirez PT, Kamat A, et al. Invasive extramammary Paget’s disease of the bladder diagnosed 18 years after noninvasive extramammary Paget’s disease of the vulva. Gynecol Oncol Case Rep. 2014;8:27-29. doi:10.1016/j.gynor.2014.03.004
- Yao H, Xie M, Fu S, et al. Survival analysis of patients with invasive extramammary Paget disease: implications of anatomic sites. BMC Cancer. 2018;18:403. doi:10.1186/s12885-018-4257-1
- Kato H, Watanabe S, Kariya K, et al. Efficacy of low-dose 5-fluorouracil/cisplatin therapy for invasive extramammary Paget’s disease. J Dermatol. 2018;45:560-563. doi:10.1111/1346-8138.14247
- Yoshino K, Fujisawa Y, Kiyohara Y, et al. Usefulness of docetaxel as first-line chemotherapy for metastatic extramammary Paget’s disease. J Dermatol. 2016;43:633-637. doi:10.1111/1346-8138.13200
- Shu B, Shen XX, Chen P, et al. Primary invasive extramammary Paget disease on penoscrotum: a clinicopathological analysis of 41 cases. Hum Pathol. 2016;47:70-77. doi:10.1016/j.humpath.2015.09.005References
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Dai B, Kong YY, Chang K, et al. Primary invasive carcinoma associated with penoscrotal extramammary Paget’s disease: a clinicopathological analysis of 56 cases. BJU Int. 2015;115:153-160. doi:10.1111/bju.12776
- Shiomi T, Noguchi T, Nakayama H, et al. Clinicopathological study of invasive extramammary Paget’s disease: subgroup comparison according to invasion depth. J Eur Acad Dermatol Venereol. 2013;27:589-592. doi:10.1111/j.1468-3083.2012.04489.x
- Hatta N, Morita R, Yamada M, et al. Sentinel lymph node biopsy in patients with extramammary Paget’s disease. Dermatol Surg. 2004;30:1329-1334. doi:10.1111/j.1524-4725.2004.30377.x
- Karam A, Dorigo O. Treatment outcomes in a large cohort of patients with invasive extramammary Paget’s disease. Gynecol Oncol. 2012;125:346-351. doi:10.1016/j.ygyno.2012.01.032
- Guo L, Liu X, Li H, et al. Clinicopathological features of extramammary Paget’s disease: a report of 75 cases. Article in Chinese. Zhonghua Yi Xue Za Zhi. 2015;95:1751-1754.
- Kilts TP, Long B, Glasgow AE, et al. Invasive vulvar extramammary Paget’s disease in the United States. Gynecol Oncol. 2020;157:649-655. doi:10.1016/j.ygyno.2020.03.018
- Kusatake K, Harada Y, Mizumoto K, et al. Usefulness of sentinel lymph node biopsy for the detection of metastasis in the early stage of extramammary Paget’s disease. Eur J Dermatol. 2015;25:156-161. doi:10.1684/ejd.2015.2534
- Jeong BK, Kim KR. Invasive extramammary Paget disease of the vulva with signet ring cell morphology in a patient with signet ring cell carcinoma of the stomach: report of a case. Int J Gynecol Pathol. 2018;37:147-151. doi:10.1097/PGP.0000000000000405
- Pagnanelli M, De Nardi P, Martella S, et al. Local excision of a mucinous adenocarcinoma of the anal margin (extramammary Paget’s disease) and reconstruction with a bilateral V-Y flap. Case Rep Surg. 2019;2019:9073982. doi:10.1155/2019/9073982
- Sopracordevole F, Di Giuseppe J, De Piero G, et al. Surgical treatment of Paget disease of the vulva: prognostic significance of stromal invasion and surgical margin status. J Low Genit Tract Dis. 2016;20:184-188. doi:10.1097/LGT.0000000000000191
- Evans AT, Neven P. Invasive adenocarcinoma arising in extramammary Paget’s disease of the vulva. Histopathology. 1991;18:355-360. doi:10.1111/j.1365-2559.1991.tb00857.x
- Kitano A, Izumi M, Tamura K, et al. Brain metastasis from cutaneous squamous cell carcinoma coexistent with extramammary Paget’s disease: a case report. Pathol Int. 2019;69:619-625. doi:10.1111/pin.12846
- Miracco C, Francini E, Torre P, et al. Extramammary invasive Paget’s disease and apocrine angiomatous hamartoma: an unusual association. Eur J Dermatol. 2018;28:853-855. doi:10.1684/ejd.2018.3438
- Kambayashi Y, Fujimura T, Ohuchi K, et al. Advanced invasive extramammary Paget’s disease concomitant with cecal cancer possessing rare variant of TP53 single nucleotide polymorphism. Case Rep Oncol. 2019;12:855-860. doi:10.1159/000504339
- Fujimura T, Furudate S, Kambayashi Y, et al. Potential use of bisphosphonates in invasive extramammary Paget’s disease: an immunohistochemical investigation. Clin Dev Immunol. 2013;2013:164982. doi:10.1155/2013/164982
- Kawamura H, Ogata K, Miura H, et al. Patellar metastases. A report of two cases. Int Orthop. 1993;17:57-59. doi:10.1007/BF00195227
- Damavandy AA, Terushkin V, Zitelli JA, et al. Intraoperative immunostaining for cytokeratin-7 during Mohs micrographic surgery demonstrates low local recurrence rates in extramammary Paget’s disease. Dermatol Surg. 2018;44:354-364. doi:10.1097/DSS.0000000000001355
- Morris CR, Hurst EA. Extramammary Paget disease: a review of the literature-part I: history, epidemiology, pathogenesis, presentation, histopathology, and diagnostic work-up. Dermatol Surg. 2020;46:151-158. doi:10.1097/DSS.0000000000002064
- Simonds RM, Segal RJ, Sharma A. Extramammary Paget’s disease: a review of the literature. Int J Dermatol. 2019;58:871-879. doi:10.1111/ijd.14328
- Ogata D, Kiyohara Y, Yoshikawa S, et al. Usefulness of sentinel lymph node biopsy for prognostic prediction in extramammary Paget’s disease. Eur J Dermatol. 2016;26:254-259. doi:10.1684/ejd.2016.2744
- Ohara K, Fujisawa Y, Yoshino K, et al. A proposal for a TNM staging system for extramammary Paget disease: retrospective analysis of 301 patients with invasive primary tumors. J Dermatol Sci. 2016;83:234-239. doi:10.1016/j.jdermsci.2016.06.004
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Kim SJ, Thompson AK, Zubair AS, et al. Surgical treatment and outcomes of patients with extramammary Paget disease: a cohort study. Dermatol Surg. 2017;43:708-714. doi:10.1097/DSS.0000000000001051
- Wollina U. Extensive invasive extramammary Paget’s disease: surgical treatment. J Cutan Aesthet Surg. 2013;6:41-44. doi:10.4103/0974-2077.110098
- Tsutsui K, Takahashi A, Muto Y, et al. Outcomes of lymph node dissection in the treatment of extramammary Paget’s disease: a single-institution study. J Dermatol. 2020;47:512-517. doi:10.1111/1346-8138.15285
- Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: a case report. Case Rep Oncol. 2021;14:430-438. doi:10.1159/000514345
- Hashimoto H, Ito T. Current management and treatment of extramammary Paget’s disease. Curr Treat Options Oncol. 2022;23:818-830. doi:10.1007/s11864-021-00923-3
- Kibbi N, Owen JL, Worley B, et al. Evidence-based clinical practice guidelines for extramammary Paget disease. JAMA Oncol. 2022;8:618-628. doi:10.1001/jamaoncol.2021.7148
- Morris CR, Hurst EA. Extramammary Paget’s disease: a review of the literature part II: treatment and prognosis. Dermatol Surg. 2020;46:305-311. doi:10.1097/DSS.0000000000002240
- Merritt BG, Degesys CA, Brodland DG. Extramammary Paget disease. Dermatol Clin. 2019;37:261-267. doi:10.1016/j.det.2019.02.002
- Aroche Gutierrez LL, Holloway SB, Donthi D, et al. Docetaxel treatment for widely metastatic invasive vulvar extramammary Paget’s disease with multifocal bone metastasis. Gynecol Oncol Rep. 2022;45:101114. doi:10.1016/j.gore.2022.101114
- Ueda M, Omori M, Sakai A. Invasive extramammary Paget’s disease with lymph node metastases and high-grade B-cell lymphoma. An Bras Dermatol. 2023;98:414-418. doi:10.1016/j.abd.2022.04.012
- Rathore R, Yadav D, Agarwal S, et al. Primary extra mammary Paget’s disease of vulva, with apocrine adenocarcinoma, signet ring cell differentiation and distant metastasis. J Family Reprod Health. 2020;14:276-280. doi:10.18502/jfrh.v14i4.5213
- Kawahara Y, Umeda Y, Yamaguchi B, et al. Long-term resolution of invasive extramammary Paget’s disease with multiple regional lymph node metastases solely with regional lymph node dissection. J Dermatol. 2021;48:E452-E453. doi:10.1111/1346-8138.16007
- Hanyu T, Fujitani S, Ito A, et al. Brain metastasis from extramammary Paget’s disease. Nagoya J Med Sci. 2020;82:791-798. doi:10.18999/nagjms.82.4.791
- Waki Y, Nobeyama Y, Ogawa T, et al. Case of extramammary Paget’s disease causing pulmonary tumor embolism. J Dermatol. 2020;47:E133-E134. doi:10.1111/1346-8138.15267
- Li ZG, Qin XJ. Extensive invasive extramammary Paget disease evaluated by F-18 FDG PET/CT: a case report. Medicine (Baltimore). 2015;94:E371. doi:10.1097/MD.0000000000000371
- Kato N, Matsue K, Sotodate A, et al. Extramammary Paget’s disease with distant skin metastasis. J Dermatol. 1996;23:408-414. doi:10.1111/j.1346-8138.1996.tb04043.x
- Hosomi M, Miyake O, Matsumiya K, et al. Extramammary Paget’s disease with a large mass in male genitalia: a case report. Article in Japanese. Hinyokika Kiyo. 1989;35:1981-1984.
- Hardy LE, Baxter L, Wan K, et al. Invasive cervical adenocarcinoma arising from extension of recurrent vulval Paget’s disease. BMJ Case Rep. 2020;13e232424. doi:10.1136/bcr-2019-232424
- Onaiwu CO, Ramirez PT, Kamat A, et al. Invasive extramammary Paget’s disease of the bladder diagnosed 18 years after noninvasive extramammary Paget’s disease of the vulva. Gynecol Oncol Case Rep. 2014;8:27-29. doi:10.1016/j.gynor.2014.03.004
- Yao H, Xie M, Fu S, et al. Survival analysis of patients with invasive extramammary Paget disease: implications of anatomic sites. BMC Cancer. 2018;18:403. doi:10.1186/s12885-018-4257-1
- Kato H, Watanabe S, Kariya K, et al. Efficacy of low-dose 5-fluorouracil/cisplatin therapy for invasive extramammary Paget’s disease. J Dermatol. 2018;45:560-563. doi:10.1111/1346-8138.14247
- Yoshino K, Fujisawa Y, Kiyohara Y, et al. Usefulness of docetaxel as first-line chemotherapy for metastatic extramammary Paget’s disease. J Dermatol. 2016;43:633-637. doi:10.1111/1346-8138.13200
- Shu B, Shen XX, Chen P, et al. Primary invasive extramammary Paget disease on penoscrotum: a clinicopathological analysis of 41 cases. Hum Pathol. 2016;47:70-77. doi:10.1016/j.humpath.2015.09.005References
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Dai B, Kong YY, Chang K, et al. Primary invasive carcinoma associated with penoscrotal extramammary Paget’s disease: a clinicopathological analysis of 56 cases. BJU Int. 2015;115:153-160. doi:10.1111/bju.12776
- Shiomi T, Noguchi T, Nakayama H, et al. Clinicopathological study of invasive extramammary Paget’s disease: subgroup comparison according to invasion depth. J Eur Acad Dermatol Venereol. 2013;27:589-592. doi:10.1111/j.1468-3083.2012.04489.x
- Hatta N, Morita R, Yamada M, et al. Sentinel lymph node biopsy in patients with extramammary Paget’s disease. Dermatol Surg. 2004;30:1329-1334. doi:10.1111/j.1524-4725.2004.30377.x
- Karam A, Dorigo O. Treatment outcomes in a large cohort of patients with invasive extramammary Paget’s disease. Gynecol Oncol. 2012;125:346-351. doi:10.1016/j.ygyno.2012.01.032
- Guo L, Liu X, Li H, et al. Clinicopathological features of extramammary Paget’s disease: a report of 75 cases. Article in Chinese. Zhonghua Yi Xue Za Zhi. 2015;95:1751-1754.
- Kilts TP, Long B, Glasgow AE, et al. Invasive vulvar extramammary Paget’s disease in the United States. Gynecol Oncol. 2020;157:649-655. doi:10.1016/j.ygyno.2020.03.018
- Kusatake K, Harada Y, Mizumoto K, et al. Usefulness of sentinel lymph node biopsy for the detection of metastasis in the early stage of extramammary Paget’s disease. Eur J Dermatol. 2015;25:156-161. doi:10.1684/ejd.2015.2534
- Jeong BK, Kim KR. Invasive extramammary Paget disease of the vulva with signet ring cell morphology in a patient with signet ring cell carcinoma of the stomach: report of a case. Int J Gynecol Pathol. 2018;37:147-151. doi:10.1097/PGP.0000000000000405
- Pagnanelli M, De Nardi P, Martella S, et al. Local excision of a mucinous adenocarcinoma of the anal margin (extramammary Paget’s disease) and reconstruction with a bilateral V-Y flap. Case Rep Surg. 2019;2019:9073982. doi:10.1155/2019/9073982
- Sopracordevole F, Di Giuseppe J, De Piero G, et al. Surgical treatment of Paget disease of the vulva: prognostic significance of stromal invasion and surgical margin status. J Low Genit Tract Dis. 2016;20:184-188. doi:10.1097/LGT.0000000000000191
- Evans AT, Neven P. Invasive adenocarcinoma arising in extramammary Paget’s disease of the vulva. Histopathology. 1991;18:355-360. doi:10.1111/j.1365-2559.1991.tb00857.x
- Kitano A, Izumi M, Tamura K, et al. Brain metastasis from cutaneous squamous cell carcinoma coexistent with extramammary Paget’s disease: a case report. Pathol Int. 2019;69:619-625. doi:10.1111/pin.12846
- Miracco C, Francini E, Torre P, et al. Extramammary invasive Paget’s disease and apocrine angiomatous hamartoma: an unusual association. Eur J Dermatol. 2018;28:853-855. doi:10.1684/ejd.2018.3438
- Kambayashi Y, Fujimura T, Ohuchi K, et al. Advanced invasive extramammary Paget’s disease concomitant with cecal cancer possessing rare variant of TP53 single nucleotide polymorphism. Case Rep Oncol. 2019;12:855-860. doi:10.1159/000504339
- Fujimura T, Furudate S, Kambayashi Y, et al. Potential use of bisphosphonates in invasive extramammary Paget’s disease: an immunohistochemical investigation. Clin Dev Immunol. 2013;2013:164982. doi:10.1155/2013/164982
- Kawamura H, Ogata K, Miura H, et al. Patellar metastases. A report of two cases. Int Orthop. 1993;17:57-59. doi:10.1007/BF00195227
- Damavandy AA, Terushkin V, Zitelli JA, et al. Intraoperative immunostaining for cytokeratin-7 during Mohs micrographic surgery demonstrates low local recurrence rates in extramammary Paget’s disease. Dermatol Surg. 2018;44:354-364. doi:10.1097/DSS.0000000000001355
- Morris CR, Hurst EA. Extramammary Paget disease: a review of the literature-part I: history, epidemiology, pathogenesis, presentation, histopathology, and diagnostic work-up. Dermatol Surg. 2020;46:151-158. doi:10.1097/DSS.0000000000002064
- Simonds RM, Segal RJ, Sharma A. Extramammary Paget’s disease: a review of the literature. Int J Dermatol. 2019;58:871-879. doi:10.1111/ijd.14328
- Ogata D, Kiyohara Y, Yoshikawa S, et al. Usefulness of sentinel lymph node biopsy for prognostic prediction in extramammary Paget’s disease. Eur J Dermatol. 2016;26:254-259. doi:10.1684/ejd.2016.2744
- Ohara K, Fujisawa Y, Yoshino K, et al. A proposal for a TNM staging system for extramammary Paget disease: retrospective analysis of 301 patients with invasive primary tumors. J Dermatol Sci. 2016;83:234-239. doi:10.1016/j.jdermsci.2016.06.004
- Fujisawa Y, Yoshino K, Kiyohara Y, et al. The role of sentinel lymph node biopsy in the management of invasive extramammary Paget’s disease: multi-center, retrospective study of 151 patients. J Dermatol Sci. 2015;79:38-42. doi:10.1016/j.jdermsci.2015.03.014
- Kim SJ, Thompson AK, Zubair AS, et al. Surgical treatment and outcomes of patients with extramammary Paget disease: a cohort study. Dermatol Surg. 2017;43:708-714. doi:10.1097/DSS.0000000000001051
- Wollina U. Extensive invasive extramammary Paget’s disease: surgical treatment. J Cutan Aesthet Surg. 2013;6:41-44. doi:10.4103/0974-2077.110098
- Tsutsui K, Takahashi A, Muto Y, et al. Outcomes of lymph node dissection in the treatment of extramammary Paget’s disease: a single-institution study. J Dermatol. 2020;47:512-517. doi:10.1111/1346-8138.15285
- Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: a case report. Case Rep Oncol. 2021;14:430-438. doi:10.1159/000514345
Metastatic Primary Extramammary Paget Disease: A Case Series
Metastatic Primary Extramammary Paget Disease: A Case Series
Practice Points
- Invasive primary extramammary Paget disease has a higher risk for lymph node metastasis.
- Consider extramammary Paget disease in patients presenting with erythematous pruritic plaques in apocrine-rich areas that fail to respond to topical steroids or antifungals.
- Prompt diagnosis can expedite comprehensive malignancy work-up and multidisciplinary management, potentially impacting patient outcomes.
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Practice Points
- Clinicians should be aware of the occurrence of severe cutaneous adverse reactions (SCARs) in patients on antineoplastic therapy to prevent delays in treatment and improve patient outcomes.
- Rapid initiation of treatment can be effective in resolving SCARs and ensuring full recovery.
- Close coordination between dermatology and oncology teams is crucial to manage SCARs while minimizing cancer treatment interruptions.
Adverse Events Associated With At-Home Microcurrent Facial Devices
Adverse Events Associated With At-Home Microcurrent Facial Devices
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
Adverse Events Associated With At-Home Microcurrent Facial Devices
Adverse Events Associated With At-Home Microcurrent Facial Devices
PRACTICE POINTS
- At-home microcurrent facial devices have been associated with serious adverse events, including arrhythmia, pain, dizziness, and retinal detachment, based on US Food and Drug Administration Manufacturer and User Facility Device Experience database reports, underscoring the importance of counseling patients about potential risks prior to use.
- Existing randomized controlled trials of microcurrent devices are limited by small sample sizes and short follow-up periods (maximum 12 weeks), making it difficult to characterize the long-term safety profile of these increasingly popular devices.
- Dermatologists should be aware that the largely unregulated at-home microcurrent device market lacks robust, large-scale safety data. Patients, particularly those with cardiac conditions or implanted electrical devices, should be advised to consult a physician before use.
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
What evidence exists on the impact of systemic immunosuppressants, biologics, and small-molecule inhibitors on patch test sensitivity and specificity?
DR. ATWATER: Guidance on this topic recently was published by the North American Contact Dermatitis Group (NACDG) in the Journal of the American Academy of Dermatology in June 2025. The article outlined expert recommendations on whether systemic immunosuppressants, biologics, and small-molecule inhibitors should be held before patch testing, how long they should be withheld, and the maximum recommended doses that can be used during testing.
How can dermatologists perform patch testing or use alternative diagnostic strategies when systemic therapy cannot safely be withheld?
DR. ATWATER: When systemic therapy cannot safely be withheld and patch testing is needed for diagnostic purposes, I typically proceed with the understanding that there is risk of false-negative reactions. If the patient has dermatitis on systemic therapy, it suggests that an allergic response on patch testing is also possible. I generally follow the NACDG guidelines mentioned above, and I hold systemic medications during the week of testing, when possible. If the patient has diffuse dermatitis on systemic therapy and their skin is not clear enough to proceed, I prescribe a prednisone taper and patch test on 10 mg per day for the entire week of testing. In patients taking systemic medications, I typically consider doubtful (+/−) patch test reactions to be the equivalent of a positive (1+) reaction.
One alternative diagnostic strategy is to create a safe list that avoids common allergens and have the patient use only products on this list. If their skin clears with avoidance, it suggests that they may have a contact allergy, and you can proceed with patch testing.
In patients with a convincing history of contact dermatitis but a negative patch test, what are the most common causes of false negatives, and how do you distinguish those from true negatives?
DR. ATWATER: In this setting, the most common cause of a false-negative patch test is not testing the correct allergens. This may occur when too few allergens are tested or when relevant allergens are not tested. Other potential causes of false negatives are incorrect timing of allergen exposure and readings, inadequate allergen adherence, expired allergens, and testing with the incorrect vehicle or concentration of allergen. Some immunosuppressant medications also can cause a false-negative patch test. The only way to distinguish false negatives from true negatives is to be aware of these potential pitfalls and continuously work to avoid errors whenever possible.
What technical and practical factors most influence false negatives/positives, and what steps do you recommend to standardize and improve test yield?
DR. ATWATER: Not testing the correct allergens is a potential pitfall in patch testing. For example, when comparing the 35 allergens in the T.R.U.E. Test (thin layer rapid-use epicutaneous test) to the 80 allergens tested by the NACDG in 2021 to 2022, up to 48% of NACDG allergens are missed when testing with only the T.R.U.E. Test. This argues for comprehensive patch testing and testing of at least 80 to 90 allergens whenever possible. Another example is failure to test allergens relevant to occupational exposures, such as in the case of a hairdresser or nail technician. When patches are not applied for the correct period (48 hours) and the final reading isn’t completed in the recommended timeframe (96 to 168 hours), there is increased risk for false negatives and positives. Both false negatives and positives can occur if you complete your final reading too early, whereas false negatives may be more likely if you complete your final reading late. Poor allergen adherence, which can be caused by hair, sweat, poor tape application, water, and exercise, also can result in false negatives. Allergen concentration that is too low to elicit a reaction, as well as too little allergen placed into the chamber, also could increase the risk of false negatives. Too much allergen in the chamber or too high a concentration of the allergen can result in false positives.
Topical medications applied to the patch test site prior to or during patch testing, as well as phototherapy, sun exposure, intramuscular triamcinolone, immunosuppressants, biologics, and small-molecule inhibitors can suppress the immune response to allergen exposure.
To identify true positive patch test reactions, use side lighting and palpate the skin. Be aware of the appearance of irritant reactions, patch test reaction variants such as follicular reactions, and the poral reaction, which can be seen with cobalt. Strong knowledge of how to read patch test reactions will decrease your risk of false-positive and -negative reactions. Training and protocols are vital for standardization and accurate patch testing. We train our staff on the technicalities of patch testing and utilize patch test orders and checklists in our office. We take photos to confirm application sites and visually track reactions between visits. We also provide verbal and written patch test care instructions for our patients and reinforce instructions at each clinic visit.
What are your top practical tips for dermatologists to maximize diagnostic accuracy and patient safety?
DR. ATWATER: My first tip is to develop patch test protocols that are followed by staff and physicians—every time—for every patient. My second tip is to make sure you understand and are comfortable with the patch test process. There are several great patch test resources that can help, including Introduction to Patch Testing, a recently developed CME module in the AAD Learning Center (https://learning.aad.org/Listing/Introduction-to-Patch- Testing-20366). There also are patch test training courses and other resources offered by the American Contact Dermatitis Society (https://www.contactderm.org/).
What single guideline change would most improve patch testing?
DR. ATWATER: The single guideline change that would most improve the practice of patch testing is removal of payer limitations on the number of patches that can be applied per day. For many payers in the United States, this limit is 80 to 90 allergens, and it is sometimes lower. Limits on the number of allergens that can be applied per day may result in false-negative patch testing (when limits are applied), patient inconvenience (if testing is completed over 2 different application days), and insufficient reimbursement (if more allergens are tested than the limit allows).
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD
Optimizing Patch Testing in Clinical Practice: Insights From Amber Reck Atwater, MD